User login
How Should Hospitalized Patients with Long QT Syndrome Be Managed?
Case
You are asked to admit a 63-year-old male with a history of hypertension and osteoarthritis. The patient, who fell at home, is scheduled for open repair of his femoral neck fracture the following day. The patient reports tripping over his granddaughter’s toys and denies any associated symptoms around the time of his fall. An electrocardiogram (ECG) reveals a QTc (QT) interval of 480 ms. How should this hospitalized patient’s prolonged QT interval be managed?
Overview
Patients with a prolonged QT interval on routine ECG present an interesting dilemma for clinicians. Although QT prolongation—either congenital or acquired—has been associated with dysrhythmias, the risk of torsades de pointes and sudden cardiac death varies considerably based on myriad underlying factors.1 Therefore, the principle job of the clinician who has recognized QT prolongation is to assess and minimize the risk of the development of clinically significant dysrhythmias, and to be prepared to manage them should they arise.
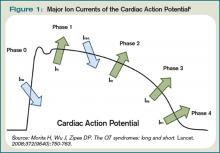
The QT interval encompasses ventricular depolarization and repolarization. This ventricular action potential proceeds through five phases. The initial upstroke (phase 0) of depolarization occurs with the opening of Na+ channels, triggering the inward Na+ current (INa), and causes the interior of the myocytes to become positively charged. This is followed by initial repolarization (phase 1) when the opening of K+ channels causes an outward K+ current (Ito). Next, the plateau phase (phase 2) of the action potential follows with a balance of inward current through Ca2+channels (Ica-L) and outward current through slow rectifier K+ channels (IKs), and then later through delayed, rapid K+ rectifier channels (IKr). Then, the inward current is deactivated, while the outward current increases through the rapid delayed rectifier (IKr) and opening of inward rectifier channels (IK1) to complete repolarization (phase 3). Finally, the action potential returns to baseline (phase 4) and Na+ begins to enter the cell again (see Figure 1, above).
The long QT syndrome (LQTS) is defined by a defect in these cardiac ion channels, which leads to abnormal repolarization, usually lengthening the QT interval and thus predisposing to ventricular dysrhythmias.2 It is estimated that as many as 85% of these syndromes are inherited, and up to 15% are acquired or sporadic.3 Depending on the underlying etiology of the LQTS, manifestations might first be appreciated at any time from in utero through adulthood.4 Symptoms including palpitations, syncope, seizures, or cardiac arrest bring these patients to medical attention.3 These symptoms frequently elicit physical or emotional stress, but they can occur without obvious inciting triggers.5 A 20% mortality risk exists in patients who are symptomatic and untreated in the first year following diagnosis, and up to 50% within 10 years following diagnosis.4
How is Long QT Syndrome Diagnosed?
The LQTS diagnosis is based on clinical history in combination with ECG abnormalities.6 Important historical elements include symptoms of palpitations, syncope, seizures, or cardiac arrest.3 In addition, a family history of unexplained syncope or sudden death, especially at a young age, should raise LQTS suspicion.5
A variety of ECG findings can be witnessed in LQTS patients.4,5 Although the majority of patients have a QTc >440 ms, approximately one-third have a QTc ≤460 ms, and about 10% have normal QTc intervals.5 Other ECG abnormalities include notched, biphasic, or prolonged T-waves, and the presence of U-waves.4,5 Schwartz et al used these elements to publish criteria (see Table 1, right) that physicians can use to assess the probability that a patient has LQTS.7
Types of Long QT Syndromes
Because the risk of developing significant dysrhythmias with LQTS is dependent on both the actual QT interval, with risk for sudden cardiac death increased two to three times with QT >440 ms compared with QT <440 ms and the specific underlying genotype, it is important to have an understanding of congenital and acquired LQTS and the associated triggers for torsades de pointes.
Congenital LQTS
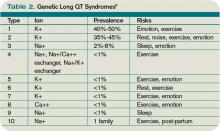
Congenital LQTS is caused by mutations in cardiac ion channel proteins, primarily sodium, and potassium channels.5,6 These defects either slow depolarization or lengthen repolarization, leading to heterogeneity of repolarization of the membrane.5 This, in turn, predisposes to ventricular dysrhythmias, including torsades de pointes and subsequent ventricular fibrillation and death.2 Currently, 12 genetic defects have been identified in LQTS. Hundreds of mutations have been described to cause these defects (see Table 2, right).8 Approximately 70% of congenital LQTS are caused by mutations in three genes and are classified as LQTS 1, LQTS 2, and LQTS 3.8 The other seven mutation types account for about 5% of cases; a quarter of LQTS cases have no identified genetic mutations.8
LQTS usually can be distinguished by clinical features and some ECG characteristics, but diagnosis of the specific type requires genetic testing.8,9 The most common types of LQTS are discussed below.
- Long QT1 is the most common type, occurring in approximately 40% to 50% of patients diagnosed with LQTS. It is characterized by a defect in the potassium channel alpha subunit leading to IKs reduction.9 These patients typically present with syncope or adrenergic-induced torsades, might have wide, broad-based T-waves on ECG, and respond well to beta-blocker therapy.6 Triggers for these patients include physical exertion or emotional stressors, particularly exercise and swimming. These patients typically present in early childhood.1
- Long QT2 occurs in 35% to 40% of patients and is characterized by a different defect in the alpha subunit of the potassium channel, which leads to reduced IKr.9 ECGs in LQTS2 can demonstrate low-amplitude and notched T-waves. Sudden catecholamine surges related to emotional stress or loud noises and bradycardia can trigger dysrhythmias in Long QT2.6 Thus, beta blockers reduce overall cardiac events in LQTS2 but less effectively than in LQTS1.6 These patients also present in childhood but typically are older than patients with LQTS1.6
- Long QT3 is less common than LQTS1 or LQTS2, at 2% to 8% of LQTS patients, but carries a higher mortality and is not treated effectively with beta blockers. LQTS3 is characterized by a defect in a sodium channel, causing a gain-of-function in the INa.4,9 These patients are more likely to have a fatal dysrhythmia while sleeping, are less susceptible to exercise-induced events, and have increased morbidity and mortality associated with bradycardia.4,9 ECG frequently reveals a relatively long ST segment, followed by a peaked and tall T-wave. Beta-blocker therapy can predispose to dysrhythmias in these patients; therefore, many of these patients will have pacemakers implanted as first-line therapy.6
While less common, Jervell and Lange Nielson syndrome is an autosomal recessive form of LQTS in which affected patients have homozygous mutations in the KCNQ1 or KCNE1 genes. This syndrome occurs in approximately 1% to 7% of LQTS patients, displays a typical QTc >550 ms, can be triggered by exercise and emotional stress, is associated with deafness, and carries a high risk of cardiac events at a young age.6
Acquired Syndromes
In addition to congenital LQTS, certain patients can acquire LQTS after being treated with particular drugs or having metabolic abnormalities, namely hypomagnesemia, hypocalcemia, and hypokalemia. Most experts think patients who “acquire” LQTS that predisposes to torsades de pointes have underlying structural heart disease or LQTS genetic carrier mutations that combine with transient initiating events (e.g., drugs or metabolic abnormalities) to induce dysrhythmias.1 In addition to certain drugs, cardiac ischemia, and electrolyte abnormalities, cocaine abuse, HIV, and subarachnoid hemorrhage can induce dysrhythmias in susceptible patients.5
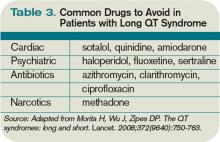
Many types of drugs can cause a prolonged QT interval, and others should be avoided in patients with pre-existing prolonged QT (see Table 3, p. 17). Potentially offending drugs that are frequently encountered by inpatient physicians include amiodarone, diltiazem, erythromycin, clarithromycin, ciprofloxacin, fluoxetine, paroxetine, sertraline, haloperidol, ritonavir, and methadone.1 Additionally, drugs that cause electrolyte abnormalities (e.g., diuretics and lithium) should be monitored closely.
Overall, the goals of therapy in LQTS are:
- Decrease the risk of dysrhythmic events;
- Minimize adrenergic response;
- Shorten the QTc;
- Decrease the dispersion of refractoriness; and
- Improve the function of the ion channels.3
Supportive measures should be taken for patients who are acutely symptomatic from LQTS and associated torsades de pointes. In addition to immediate cardioversion for ongoing and hemodynamically significant torsades, intravenous magnesium should be administered, electrolytes corrected, and offending drugs discontinued.5 Temporary transvenous pacing at rates of approximately 100 beats per minute is highly effective in preventing short-term recurrence of torsades in congenital and acquired LQTS, especially in bradycardic patients.5 Isoproterenol infusion increases the heart rate and effectively prevents acute recurrence of torsades in patients with acquired LQTS, but it should be used with caution in patients with structural heart disease.5
Long-term strategies to manage LQTS include:
- Minimizing the risk of triggering cardiac events via adrenergic stimulation;
- Preventing ongoing dysrhythmias;
- Avoiding medications known to prolong the QT interval; and
- Maintaining normal electrolytes and minerals.5
Most patients with congenital long QT should be discouraged from participating in competitive sports, and patients should attempt to eliminate exposures to stress or sudden awakening, though this is not practical in all cases.5 Beta blockers generally are the first-line therapy and are more effective for LQT1 than LQT2 or LQT3.4,5 If patients are still symptomatic despite adequate medical therapy, or have survived cardiac arrest, they should be considered for ICD therapy.4,5 In addition, patients with profound bradycardia benefit from pacemaker implantation.5 Patients who remain symptomatic despite both beta blockade and ICD placement might find cervicothoracic sympathectomy curative.4,5
Perioperative Considerations
Although little data is available to guide physicians in the prevention of torsades de pointes during the course of anesthesia, there are a number of considerations that may reduce the chances of symptomatic dysrhythmias.
First, care should be taken to avoid dysrhythmia triggers in LQTS by providing a calm, quiet environment during induction, monitoring, and treating metabolic abnormalities, and providing an appropriate level of anesthesia.10 Beta-blocker therapy should be continued and potentially measured preoperatively by assessing heart rate response during stress testing.5 An implantable cardioverter-defibrillator (AICD) should be interrogated prior to surgery and inactivated during the operation.5
Finally, Kies et al have recommended general anesthesia with propofol for induction (or throughout), isoflurane as the volatile agent, vecuronium for muscle relaxation, and intravenous fentanyl for analgesia when possible.10
Back to the Case
While the patient had no genetic testing for LQTS, evaluation of previous ECGs demonstrated a prolonged QT interval. The hip fracture repair was considered an urgent procedure, which precluded the ability to undertake genetic testing and consideration for device implantation. The only medication that was known to increase the risk for dysrhythmias in this patient was his diuretic, with the attendant risk of electrolyte abnormalities.
Thus, the patient’s hydrochlorothiazide was discontinued and his pre-existing atenolol continued. The patient’s electrolytes and minerals were monitored closely, and magnesium was administered on the day of surgery. Anesthesia was made aware of the prolonged QT interval, such that they were able to minimize the risk for and anticipate the treatment of dysrhythmias. The patient tolerated the surgery and post-operative period without complication and was scheduled for an outpatient workup and management for his prolonged QT interval.
Bottom Line
Long QT syndrome is frequently genetic in origin, but it can be caused by certain medications and perturbations of electrolytes. Beta blockers are the first-line therapy for the majority of LQTS cases, along with discontinuation of drugs that might induce or aggravate the QT prolongation.
Patients who have had cardiac arrest or who remain symptomatic despite beta-blocker therapy should have an ICD implanted.
In the perioperative period, patients’ electrolytes should be monitored and kept within normal limits. If the patient is on a beta blocker, it should be continued, and the anesthesiologist should be made aware of the diagnosis so that the anesthethic plan can be optimized to prevent arrhythmic complications. TH
Dr. Kamali is a medical resident at the University of Colorado Denver. Dr. Stickrath is a hospitalist at the Veterans Affairs Medical Center in Denver and an instructor of medicine at UC Denver. Dr. Prochazka is director of ambulatory care at the Denver VA and professor of medicine at UC Denver. Dr. Varosy is director of cardiac electrophysiology at the Denver VA and assistant professor of medicine at UC Denver.
References
- Kao LW, Furbee BR. Drug-induced q-T prolongation. Med Clin North Am. 2005;89(6):1125-1144.
- Marchlinski F. Chapter 226, The Tachyarrhythmias; Harrison's Principles of Internal Medicine, 17e. Available at: www.accessmedicine.com/resourceTOC .aspx?resourceID=4. Accessed Nov. 21, 2009.
- Zareba W, Cygankiewicz I. Long QT syndrome and short QT syndrome. Prog Cardiovasc Dis. 2008; 51(3):264-278.
- Booker PD, Whyte SD, Ladusans EJ. Long QT syndrome and anaesthesia. Br J Anaesth. 2003;90(3):349-366.
- Khan IA. Long QT syndrome: diagnosis and management. Am Heart J. 2002;143(1):7-14.
- Morita H, Wu J, Zipes DP. The QT syndromes: long and short. Lancet. 2008;372(9640):750-763.
- Schwartz PJ, Moss AJ, Vincent GM, Crampton RS. Diagnostic criteria for the long QT syndrome. An update. Circulation. 1993;88(2):782-784.
- Kapa S, Tester DJ, Salisbury BA, et al. Genetic testing for long-QT syndrome: distinguishing pathogenic mutations from benign variants. Circulation. 2009;120(18):1752-1760.
- Modell SM, Lehmann MH. The long QT syndrome family of cardiac ion channelopathies: a HuGE review. Genet Med. 2006;8(3):143-155.
- Kies SJ, Pabelick CM, Hurley HA, White RD, Ackerman MJ. Anesthesia for patients with congenital long QT syndrome. Anesthesiology. 2005;102(1):204-210.
- Wisely NA, Shipton EA. Long QT syndrome and anaesthesia. Eur J Anaesthesiol. 2002;19(12):853-859.
Case
You are asked to admit a 63-year-old male with a history of hypertension and osteoarthritis. The patient, who fell at home, is scheduled for open repair of his femoral neck fracture the following day. The patient reports tripping over his granddaughter’s toys and denies any associated symptoms around the time of his fall. An electrocardiogram (ECG) reveals a QTc (QT) interval of 480 ms. How should this hospitalized patient’s prolonged QT interval be managed?
Overview
Patients with a prolonged QT interval on routine ECG present an interesting dilemma for clinicians. Although QT prolongation—either congenital or acquired—has been associated with dysrhythmias, the risk of torsades de pointes and sudden cardiac death varies considerably based on myriad underlying factors.1 Therefore, the principle job of the clinician who has recognized QT prolongation is to assess and minimize the risk of the development of clinically significant dysrhythmias, and to be prepared to manage them should they arise.
The QT interval encompasses ventricular depolarization and repolarization. This ventricular action potential proceeds through five phases. The initial upstroke (phase 0) of depolarization occurs with the opening of Na+ channels, triggering the inward Na+ current (INa), and causes the interior of the myocytes to become positively charged. This is followed by initial repolarization (phase 1) when the opening of K+ channels causes an outward K+ current (Ito). Next, the plateau phase (phase 2) of the action potential follows with a balance of inward current through Ca2+channels (Ica-L) and outward current through slow rectifier K+ channels (IKs), and then later through delayed, rapid K+ rectifier channels (IKr). Then, the inward current is deactivated, while the outward current increases through the rapid delayed rectifier (IKr) and opening of inward rectifier channels (IK1) to complete repolarization (phase 3). Finally, the action potential returns to baseline (phase 4) and Na+ begins to enter the cell again (see Figure 1, above).
The long QT syndrome (LQTS) is defined by a defect in these cardiac ion channels, which leads to abnormal repolarization, usually lengthening the QT interval and thus predisposing to ventricular dysrhythmias.2 It is estimated that as many as 85% of these syndromes are inherited, and up to 15% are acquired or sporadic.3 Depending on the underlying etiology of the LQTS, manifestations might first be appreciated at any time from in utero through adulthood.4 Symptoms including palpitations, syncope, seizures, or cardiac arrest bring these patients to medical attention.3 These symptoms frequently elicit physical or emotional stress, but they can occur without obvious inciting triggers.5 A 20% mortality risk exists in patients who are symptomatic and untreated in the first year following diagnosis, and up to 50% within 10 years following diagnosis.4
How is Long QT Syndrome Diagnosed?
The LQTS diagnosis is based on clinical history in combination with ECG abnormalities.6 Important historical elements include symptoms of palpitations, syncope, seizures, or cardiac arrest.3 In addition, a family history of unexplained syncope or sudden death, especially at a young age, should raise LQTS suspicion.5
A variety of ECG findings can be witnessed in LQTS patients.4,5 Although the majority of patients have a QTc >440 ms, approximately one-third have a QTc ≤460 ms, and about 10% have normal QTc intervals.5 Other ECG abnormalities include notched, biphasic, or prolonged T-waves, and the presence of U-waves.4,5 Schwartz et al used these elements to publish criteria (see Table 1, right) that physicians can use to assess the probability that a patient has LQTS.7
Types of Long QT Syndromes
Because the risk of developing significant dysrhythmias with LQTS is dependent on both the actual QT interval, with risk for sudden cardiac death increased two to three times with QT >440 ms compared with QT <440 ms and the specific underlying genotype, it is important to have an understanding of congenital and acquired LQTS and the associated triggers for torsades de pointes.
Congenital LQTS
Congenital LQTS is caused by mutations in cardiac ion channel proteins, primarily sodium, and potassium channels.5,6 These defects either slow depolarization or lengthen repolarization, leading to heterogeneity of repolarization of the membrane.5 This, in turn, predisposes to ventricular dysrhythmias, including torsades de pointes and subsequent ventricular fibrillation and death.2 Currently, 12 genetic defects have been identified in LQTS. Hundreds of mutations have been described to cause these defects (see Table 2, right).8 Approximately 70% of congenital LQTS are caused by mutations in three genes and are classified as LQTS 1, LQTS 2, and LQTS 3.8 The other seven mutation types account for about 5% of cases; a quarter of LQTS cases have no identified genetic mutations.8
LQTS usually can be distinguished by clinical features and some ECG characteristics, but diagnosis of the specific type requires genetic testing.8,9 The most common types of LQTS are discussed below.
- Long QT1 is the most common type, occurring in approximately 40% to 50% of patients diagnosed with LQTS. It is characterized by a defect in the potassium channel alpha subunit leading to IKs reduction.9 These patients typically present with syncope or adrenergic-induced torsades, might have wide, broad-based T-waves on ECG, and respond well to beta-blocker therapy.6 Triggers for these patients include physical exertion or emotional stressors, particularly exercise and swimming. These patients typically present in early childhood.1
- Long QT2 occurs in 35% to 40% of patients and is characterized by a different defect in the alpha subunit of the potassium channel, which leads to reduced IKr.9 ECGs in LQTS2 can demonstrate low-amplitude and notched T-waves. Sudden catecholamine surges related to emotional stress or loud noises and bradycardia can trigger dysrhythmias in Long QT2.6 Thus, beta blockers reduce overall cardiac events in LQTS2 but less effectively than in LQTS1.6 These patients also present in childhood but typically are older than patients with LQTS1.6
- Long QT3 is less common than LQTS1 or LQTS2, at 2% to 8% of LQTS patients, but carries a higher mortality and is not treated effectively with beta blockers. LQTS3 is characterized by a defect in a sodium channel, causing a gain-of-function in the INa.4,9 These patients are more likely to have a fatal dysrhythmia while sleeping, are less susceptible to exercise-induced events, and have increased morbidity and mortality associated with bradycardia.4,9 ECG frequently reveals a relatively long ST segment, followed by a peaked and tall T-wave. Beta-blocker therapy can predispose to dysrhythmias in these patients; therefore, many of these patients will have pacemakers implanted as first-line therapy.6
While less common, Jervell and Lange Nielson syndrome is an autosomal recessive form of LQTS in which affected patients have homozygous mutations in the KCNQ1 or KCNE1 genes. This syndrome occurs in approximately 1% to 7% of LQTS patients, displays a typical QTc >550 ms, can be triggered by exercise and emotional stress, is associated with deafness, and carries a high risk of cardiac events at a young age.6
Acquired Syndromes
In addition to congenital LQTS, certain patients can acquire LQTS after being treated with particular drugs or having metabolic abnormalities, namely hypomagnesemia, hypocalcemia, and hypokalemia. Most experts think patients who “acquire” LQTS that predisposes to torsades de pointes have underlying structural heart disease or LQTS genetic carrier mutations that combine with transient initiating events (e.g., drugs or metabolic abnormalities) to induce dysrhythmias.1 In addition to certain drugs, cardiac ischemia, and electrolyte abnormalities, cocaine abuse, HIV, and subarachnoid hemorrhage can induce dysrhythmias in susceptible patients.5
Many types of drugs can cause a prolonged QT interval, and others should be avoided in patients with pre-existing prolonged QT (see Table 3, p. 17). Potentially offending drugs that are frequently encountered by inpatient physicians include amiodarone, diltiazem, erythromycin, clarithromycin, ciprofloxacin, fluoxetine, paroxetine, sertraline, haloperidol, ritonavir, and methadone.1 Additionally, drugs that cause electrolyte abnormalities (e.g., diuretics and lithium) should be monitored closely.
Overall, the goals of therapy in LQTS are:
- Decrease the risk of dysrhythmic events;
- Minimize adrenergic response;
- Shorten the QTc;
- Decrease the dispersion of refractoriness; and
- Improve the function of the ion channels.3
Supportive measures should be taken for patients who are acutely symptomatic from LQTS and associated torsades de pointes. In addition to immediate cardioversion for ongoing and hemodynamically significant torsades, intravenous magnesium should be administered, electrolytes corrected, and offending drugs discontinued.5 Temporary transvenous pacing at rates of approximately 100 beats per minute is highly effective in preventing short-term recurrence of torsades in congenital and acquired LQTS, especially in bradycardic patients.5 Isoproterenol infusion increases the heart rate and effectively prevents acute recurrence of torsades in patients with acquired LQTS, but it should be used with caution in patients with structural heart disease.5
Long-term strategies to manage LQTS include:
- Minimizing the risk of triggering cardiac events via adrenergic stimulation;
- Preventing ongoing dysrhythmias;
- Avoiding medications known to prolong the QT interval; and
- Maintaining normal electrolytes and minerals.5
Most patients with congenital long QT should be discouraged from participating in competitive sports, and patients should attempt to eliminate exposures to stress or sudden awakening, though this is not practical in all cases.5 Beta blockers generally are the first-line therapy and are more effective for LQT1 than LQT2 or LQT3.4,5 If patients are still symptomatic despite adequate medical therapy, or have survived cardiac arrest, they should be considered for ICD therapy.4,5 In addition, patients with profound bradycardia benefit from pacemaker implantation.5 Patients who remain symptomatic despite both beta blockade and ICD placement might find cervicothoracic sympathectomy curative.4,5
Perioperative Considerations
Although little data is available to guide physicians in the prevention of torsades de pointes during the course of anesthesia, there are a number of considerations that may reduce the chances of symptomatic dysrhythmias.
First, care should be taken to avoid dysrhythmia triggers in LQTS by providing a calm, quiet environment during induction, monitoring, and treating metabolic abnormalities, and providing an appropriate level of anesthesia.10 Beta-blocker therapy should be continued and potentially measured preoperatively by assessing heart rate response during stress testing.5 An implantable cardioverter-defibrillator (AICD) should be interrogated prior to surgery and inactivated during the operation.5
Finally, Kies et al have recommended general anesthesia with propofol for induction (or throughout), isoflurane as the volatile agent, vecuronium for muscle relaxation, and intravenous fentanyl for analgesia when possible.10
Back to the Case
While the patient had no genetic testing for LQTS, evaluation of previous ECGs demonstrated a prolonged QT interval. The hip fracture repair was considered an urgent procedure, which precluded the ability to undertake genetic testing and consideration for device implantation. The only medication that was known to increase the risk for dysrhythmias in this patient was his diuretic, with the attendant risk of electrolyte abnormalities.
Thus, the patient’s hydrochlorothiazide was discontinued and his pre-existing atenolol continued. The patient’s electrolytes and minerals were monitored closely, and magnesium was administered on the day of surgery. Anesthesia was made aware of the prolonged QT interval, such that they were able to minimize the risk for and anticipate the treatment of dysrhythmias. The patient tolerated the surgery and post-operative period without complication and was scheduled for an outpatient workup and management for his prolonged QT interval.
Bottom Line
Long QT syndrome is frequently genetic in origin, but it can be caused by certain medications and perturbations of electrolytes. Beta blockers are the first-line therapy for the majority of LQTS cases, along with discontinuation of drugs that might induce or aggravate the QT prolongation.
Patients who have had cardiac arrest or who remain symptomatic despite beta-blocker therapy should have an ICD implanted.
In the perioperative period, patients’ electrolytes should be monitored and kept within normal limits. If the patient is on a beta blocker, it should be continued, and the anesthesiologist should be made aware of the diagnosis so that the anesthethic plan can be optimized to prevent arrhythmic complications. TH
Dr. Kamali is a medical resident at the University of Colorado Denver. Dr. Stickrath is a hospitalist at the Veterans Affairs Medical Center in Denver and an instructor of medicine at UC Denver. Dr. Prochazka is director of ambulatory care at the Denver VA and professor of medicine at UC Denver. Dr. Varosy is director of cardiac electrophysiology at the Denver VA and assistant professor of medicine at UC Denver.
References
- Kao LW, Furbee BR. Drug-induced q-T prolongation. Med Clin North Am. 2005;89(6):1125-1144.
- Marchlinski F. Chapter 226, The Tachyarrhythmias; Harrison's Principles of Internal Medicine, 17e. Available at: www.accessmedicine.com/resourceTOC .aspx?resourceID=4. Accessed Nov. 21, 2009.
- Zareba W, Cygankiewicz I. Long QT syndrome and short QT syndrome. Prog Cardiovasc Dis. 2008; 51(3):264-278.
- Booker PD, Whyte SD, Ladusans EJ. Long QT syndrome and anaesthesia. Br J Anaesth. 2003;90(3):349-366.
- Khan IA. Long QT syndrome: diagnosis and management. Am Heart J. 2002;143(1):7-14.
- Morita H, Wu J, Zipes DP. The QT syndromes: long and short. Lancet. 2008;372(9640):750-763.
- Schwartz PJ, Moss AJ, Vincent GM, Crampton RS. Diagnostic criteria for the long QT syndrome. An update. Circulation. 1993;88(2):782-784.
- Kapa S, Tester DJ, Salisbury BA, et al. Genetic testing for long-QT syndrome: distinguishing pathogenic mutations from benign variants. Circulation. 2009;120(18):1752-1760.
- Modell SM, Lehmann MH. The long QT syndrome family of cardiac ion channelopathies: a HuGE review. Genet Med. 2006;8(3):143-155.
- Kies SJ, Pabelick CM, Hurley HA, White RD, Ackerman MJ. Anesthesia for patients with congenital long QT syndrome. Anesthesiology. 2005;102(1):204-210.
- Wisely NA, Shipton EA. Long QT syndrome and anaesthesia. Eur J Anaesthesiol. 2002;19(12):853-859.
Case
You are asked to admit a 63-year-old male with a history of hypertension and osteoarthritis. The patient, who fell at home, is scheduled for open repair of his femoral neck fracture the following day. The patient reports tripping over his granddaughter’s toys and denies any associated symptoms around the time of his fall. An electrocardiogram (ECG) reveals a QTc (QT) interval of 480 ms. How should this hospitalized patient’s prolonged QT interval be managed?
Overview
Patients with a prolonged QT interval on routine ECG present an interesting dilemma for clinicians. Although QT prolongation—either congenital or acquired—has been associated with dysrhythmias, the risk of torsades de pointes and sudden cardiac death varies considerably based on myriad underlying factors.1 Therefore, the principle job of the clinician who has recognized QT prolongation is to assess and minimize the risk of the development of clinically significant dysrhythmias, and to be prepared to manage them should they arise.
The QT interval encompasses ventricular depolarization and repolarization. This ventricular action potential proceeds through five phases. The initial upstroke (phase 0) of depolarization occurs with the opening of Na+ channels, triggering the inward Na+ current (INa), and causes the interior of the myocytes to become positively charged. This is followed by initial repolarization (phase 1) when the opening of K+ channels causes an outward K+ current (Ito). Next, the plateau phase (phase 2) of the action potential follows with a balance of inward current through Ca2+channels (Ica-L) and outward current through slow rectifier K+ channels (IKs), and then later through delayed, rapid K+ rectifier channels (IKr). Then, the inward current is deactivated, while the outward current increases through the rapid delayed rectifier (IKr) and opening of inward rectifier channels (IK1) to complete repolarization (phase 3). Finally, the action potential returns to baseline (phase 4) and Na+ begins to enter the cell again (see Figure 1, above).
The long QT syndrome (LQTS) is defined by a defect in these cardiac ion channels, which leads to abnormal repolarization, usually lengthening the QT interval and thus predisposing to ventricular dysrhythmias.2 It is estimated that as many as 85% of these syndromes are inherited, and up to 15% are acquired or sporadic.3 Depending on the underlying etiology of the LQTS, manifestations might first be appreciated at any time from in utero through adulthood.4 Symptoms including palpitations, syncope, seizures, or cardiac arrest bring these patients to medical attention.3 These symptoms frequently elicit physical or emotional stress, but they can occur without obvious inciting triggers.5 A 20% mortality risk exists in patients who are symptomatic and untreated in the first year following diagnosis, and up to 50% within 10 years following diagnosis.4
How is Long QT Syndrome Diagnosed?
The LQTS diagnosis is based on clinical history in combination with ECG abnormalities.6 Important historical elements include symptoms of palpitations, syncope, seizures, or cardiac arrest.3 In addition, a family history of unexplained syncope or sudden death, especially at a young age, should raise LQTS suspicion.5
A variety of ECG findings can be witnessed in LQTS patients.4,5 Although the majority of patients have a QTc >440 ms, approximately one-third have a QTc ≤460 ms, and about 10% have normal QTc intervals.5 Other ECG abnormalities include notched, biphasic, or prolonged T-waves, and the presence of U-waves.4,5 Schwartz et al used these elements to publish criteria (see Table 1, right) that physicians can use to assess the probability that a patient has LQTS.7
Types of Long QT Syndromes
Because the risk of developing significant dysrhythmias with LQTS is dependent on both the actual QT interval, with risk for sudden cardiac death increased two to three times with QT >440 ms compared with QT <440 ms and the specific underlying genotype, it is important to have an understanding of congenital and acquired LQTS and the associated triggers for torsades de pointes.
Congenital LQTS
Congenital LQTS is caused by mutations in cardiac ion channel proteins, primarily sodium, and potassium channels.5,6 These defects either slow depolarization or lengthen repolarization, leading to heterogeneity of repolarization of the membrane.5 This, in turn, predisposes to ventricular dysrhythmias, including torsades de pointes and subsequent ventricular fibrillation and death.2 Currently, 12 genetic defects have been identified in LQTS. Hundreds of mutations have been described to cause these defects (see Table 2, right).8 Approximately 70% of congenital LQTS are caused by mutations in three genes and are classified as LQTS 1, LQTS 2, and LQTS 3.8 The other seven mutation types account for about 5% of cases; a quarter of LQTS cases have no identified genetic mutations.8
LQTS usually can be distinguished by clinical features and some ECG characteristics, but diagnosis of the specific type requires genetic testing.8,9 The most common types of LQTS are discussed below.
- Long QT1 is the most common type, occurring in approximately 40% to 50% of patients diagnosed with LQTS. It is characterized by a defect in the potassium channel alpha subunit leading to IKs reduction.9 These patients typically present with syncope or adrenergic-induced torsades, might have wide, broad-based T-waves on ECG, and respond well to beta-blocker therapy.6 Triggers for these patients include physical exertion or emotional stressors, particularly exercise and swimming. These patients typically present in early childhood.1
- Long QT2 occurs in 35% to 40% of patients and is characterized by a different defect in the alpha subunit of the potassium channel, which leads to reduced IKr.9 ECGs in LQTS2 can demonstrate low-amplitude and notched T-waves. Sudden catecholamine surges related to emotional stress or loud noises and bradycardia can trigger dysrhythmias in Long QT2.6 Thus, beta blockers reduce overall cardiac events in LQTS2 but less effectively than in LQTS1.6 These patients also present in childhood but typically are older than patients with LQTS1.6
- Long QT3 is less common than LQTS1 or LQTS2, at 2% to 8% of LQTS patients, but carries a higher mortality and is not treated effectively with beta blockers. LQTS3 is characterized by a defect in a sodium channel, causing a gain-of-function in the INa.4,9 These patients are more likely to have a fatal dysrhythmia while sleeping, are less susceptible to exercise-induced events, and have increased morbidity and mortality associated with bradycardia.4,9 ECG frequently reveals a relatively long ST segment, followed by a peaked and tall T-wave. Beta-blocker therapy can predispose to dysrhythmias in these patients; therefore, many of these patients will have pacemakers implanted as first-line therapy.6
While less common, Jervell and Lange Nielson syndrome is an autosomal recessive form of LQTS in which affected patients have homozygous mutations in the KCNQ1 or KCNE1 genes. This syndrome occurs in approximately 1% to 7% of LQTS patients, displays a typical QTc >550 ms, can be triggered by exercise and emotional stress, is associated with deafness, and carries a high risk of cardiac events at a young age.6
Acquired Syndromes
In addition to congenital LQTS, certain patients can acquire LQTS after being treated with particular drugs or having metabolic abnormalities, namely hypomagnesemia, hypocalcemia, and hypokalemia. Most experts think patients who “acquire” LQTS that predisposes to torsades de pointes have underlying structural heart disease or LQTS genetic carrier mutations that combine with transient initiating events (e.g., drugs or metabolic abnormalities) to induce dysrhythmias.1 In addition to certain drugs, cardiac ischemia, and electrolyte abnormalities, cocaine abuse, HIV, and subarachnoid hemorrhage can induce dysrhythmias in susceptible patients.5
Many types of drugs can cause a prolonged QT interval, and others should be avoided in patients with pre-existing prolonged QT (see Table 3, p. 17). Potentially offending drugs that are frequently encountered by inpatient physicians include amiodarone, diltiazem, erythromycin, clarithromycin, ciprofloxacin, fluoxetine, paroxetine, sertraline, haloperidol, ritonavir, and methadone.1 Additionally, drugs that cause electrolyte abnormalities (e.g., diuretics and lithium) should be monitored closely.
Overall, the goals of therapy in LQTS are:
- Decrease the risk of dysrhythmic events;
- Minimize adrenergic response;
- Shorten the QTc;
- Decrease the dispersion of refractoriness; and
- Improve the function of the ion channels.3
Supportive measures should be taken for patients who are acutely symptomatic from LQTS and associated torsades de pointes. In addition to immediate cardioversion for ongoing and hemodynamically significant torsades, intravenous magnesium should be administered, electrolytes corrected, and offending drugs discontinued.5 Temporary transvenous pacing at rates of approximately 100 beats per minute is highly effective in preventing short-term recurrence of torsades in congenital and acquired LQTS, especially in bradycardic patients.5 Isoproterenol infusion increases the heart rate and effectively prevents acute recurrence of torsades in patients with acquired LQTS, but it should be used with caution in patients with structural heart disease.5
Long-term strategies to manage LQTS include:
- Minimizing the risk of triggering cardiac events via adrenergic stimulation;
- Preventing ongoing dysrhythmias;
- Avoiding medications known to prolong the QT interval; and
- Maintaining normal electrolytes and minerals.5
Most patients with congenital long QT should be discouraged from participating in competitive sports, and patients should attempt to eliminate exposures to stress or sudden awakening, though this is not practical in all cases.5 Beta blockers generally are the first-line therapy and are more effective for LQT1 than LQT2 or LQT3.4,5 If patients are still symptomatic despite adequate medical therapy, or have survived cardiac arrest, they should be considered for ICD therapy.4,5 In addition, patients with profound bradycardia benefit from pacemaker implantation.5 Patients who remain symptomatic despite both beta blockade and ICD placement might find cervicothoracic sympathectomy curative.4,5
Perioperative Considerations
Although little data is available to guide physicians in the prevention of torsades de pointes during the course of anesthesia, there are a number of considerations that may reduce the chances of symptomatic dysrhythmias.
First, care should be taken to avoid dysrhythmia triggers in LQTS by providing a calm, quiet environment during induction, monitoring, and treating metabolic abnormalities, and providing an appropriate level of anesthesia.10 Beta-blocker therapy should be continued and potentially measured preoperatively by assessing heart rate response during stress testing.5 An implantable cardioverter-defibrillator (AICD) should be interrogated prior to surgery and inactivated during the operation.5
Finally, Kies et al have recommended general anesthesia with propofol for induction (or throughout), isoflurane as the volatile agent, vecuronium for muscle relaxation, and intravenous fentanyl for analgesia when possible.10
Back to the Case
While the patient had no genetic testing for LQTS, evaluation of previous ECGs demonstrated a prolonged QT interval. The hip fracture repair was considered an urgent procedure, which precluded the ability to undertake genetic testing and consideration for device implantation. The only medication that was known to increase the risk for dysrhythmias in this patient was his diuretic, with the attendant risk of electrolyte abnormalities.
Thus, the patient’s hydrochlorothiazide was discontinued and his pre-existing atenolol continued. The patient’s electrolytes and minerals were monitored closely, and magnesium was administered on the day of surgery. Anesthesia was made aware of the prolonged QT interval, such that they were able to minimize the risk for and anticipate the treatment of dysrhythmias. The patient tolerated the surgery and post-operative period without complication and was scheduled for an outpatient workup and management for his prolonged QT interval.
Bottom Line
Long QT syndrome is frequently genetic in origin, but it can be caused by certain medications and perturbations of electrolytes. Beta blockers are the first-line therapy for the majority of LQTS cases, along with discontinuation of drugs that might induce or aggravate the QT prolongation.
Patients who have had cardiac arrest or who remain symptomatic despite beta-blocker therapy should have an ICD implanted.
In the perioperative period, patients’ electrolytes should be monitored and kept within normal limits. If the patient is on a beta blocker, it should be continued, and the anesthesiologist should be made aware of the diagnosis so that the anesthethic plan can be optimized to prevent arrhythmic complications. TH
Dr. Kamali is a medical resident at the University of Colorado Denver. Dr. Stickrath is a hospitalist at the Veterans Affairs Medical Center in Denver and an instructor of medicine at UC Denver. Dr. Prochazka is director of ambulatory care at the Denver VA and professor of medicine at UC Denver. Dr. Varosy is director of cardiac electrophysiology at the Denver VA and assistant professor of medicine at UC Denver.
References
- Kao LW, Furbee BR. Drug-induced q-T prolongation. Med Clin North Am. 2005;89(6):1125-1144.
- Marchlinski F. Chapter 226, The Tachyarrhythmias; Harrison's Principles of Internal Medicine, 17e. Available at: www.accessmedicine.com/resourceTOC .aspx?resourceID=4. Accessed Nov. 21, 2009.
- Zareba W, Cygankiewicz I. Long QT syndrome and short QT syndrome. Prog Cardiovasc Dis. 2008; 51(3):264-278.
- Booker PD, Whyte SD, Ladusans EJ. Long QT syndrome and anaesthesia. Br J Anaesth. 2003;90(3):349-366.
- Khan IA. Long QT syndrome: diagnosis and management. Am Heart J. 2002;143(1):7-14.
- Morita H, Wu J, Zipes DP. The QT syndromes: long and short. Lancet. 2008;372(9640):750-763.
- Schwartz PJ, Moss AJ, Vincent GM, Crampton RS. Diagnostic criteria for the long QT syndrome. An update. Circulation. 1993;88(2):782-784.
- Kapa S, Tester DJ, Salisbury BA, et al. Genetic testing for long-QT syndrome: distinguishing pathogenic mutations from benign variants. Circulation. 2009;120(18):1752-1760.
- Modell SM, Lehmann MH. The long QT syndrome family of cardiac ion channelopathies: a HuGE review. Genet Med. 2006;8(3):143-155.
- Kies SJ, Pabelick CM, Hurley HA, White RD, Ackerman MJ. Anesthesia for patients with congenital long QT syndrome. Anesthesiology. 2005;102(1):204-210.
- Wisely NA, Shipton EA. Long QT syndrome and anaesthesia. Eur J Anaesthesiol. 2002;19(12):853-859.
Market Watch
New Generics
- Fentanyl citrate troche/lozenge (generic Actiq) transmucosal lollipop1,2
- Perindopril erbumine (generic Aceon) tablets3
New Drugs, Indications, Label Changes, and Dosage Forms
- Capsaicin 8% patch (Qutenza) has been approved by the U.S. Food and Drug Administration (FDA) as a prescription medication to treat pain associated with post-herpetic neuralgia.4 The patch is a higher concentration than over-the-counter (OTC) products. The most common side effects in clinical trials were pain, swelling, itching, redness, and bumps at the application site, as well as blood pressure increases.5 Patch placement requires the use of a topical anesthetic and additional pain relief (ice or opioid pain relievers). Therefore, patch placement must be performed by a healthcare professional. Once the patch is applied, the patient must be observed for at least an hour, as there could be a significant increase in blood pressure.
- Diclofenac sodium topical (Pennsaid) has been approved by the FDA as a topical treatment to manage knee osteoarthritis.6 The transdermal carrier dimethylsulfoxide (DMSO) is utilized to deliver active diclofenac sodium through the skin to the pain site.7
- Exenatide injection (Byetta) has been approved by the FDA as monotherapy for treating patients with Type 2 diabetes mellitus, along with diet and exercise.8
- Human papilloma virus (HPV) vaccine (Gardasil) has been approved by the FDA for preventing condyloma acuminata due to HPV types 6 and 11 in males ages 9 to 26.9 The makers hope that this vaccine will decrease the need to treat genital warts. Clinical studies showed that in males not infected with HPV at the beginning of the study, the vaccine was close to 90% effective in preventing genital warts caused by infection of HPV types 6 and 11. The manufacturer plans to obtain additional safety and effectiveness information in this patient population.
- Lansoprazole 24HR (Prevacid OTC) 15-mg delayed-release capsules are available for treating frequent heartburn.10 The capsules will be available by prescription in both the 15-mg and 30-mg dosage strengths.
- Metoclopramide HCl orally disintegrating tablet (Metozolv ODT) has been approved by the FDA for treating both acute and recurrent diabetic gastroparesis and for the short-term management (four to 12 weeks) of adults with documented symptomatic gastroesophageal reflux disease who do not respond to conventional therapy.11
- Peramivir intravenous will be made available by the Centers for Disease Control (CDC) as an emergency treatment for children and adult patients who develop H1N1 influenza and are nonresponsive to oral or inhaled antiviral therapies.12
- Rosuvastatin (Crestor) has been approved by the FDA for treating heterozygous familial cholesterolemia in children ages 10-17.13
- Telmisartan 80-mg tablets (Micardis) have been approved by the FDA for risk reduction of myocardial infarction, stroke, or death from cardiovascular causes in patients 55 years and older who are at high risk for major cardiovascular events, or who are not able to take angiotensin-converting enzyme inhibitors.14
- Telmisartan/amlodipine tablets (Twynsta) have been approved by the FDA as a new combination for treating hypertension, either alone or in combination with other antihypertensive agents. This combination is not indicated for cardiovascular risk reduction. The tablets are available in the following strengths of telmisartan/amlodipine, respectively: 40 mg/5 mg, 40 mg/10 mg, 80 mg/5 mg, and 80 mg/10 mg.
- Tranexamic acid (Lysteda) has been approved by the FDA for the treatment of menorrhagia.15 This is the first nonhormonal, oral therapeutic agent approved to treat this condition.16
Pipeline
- Indacaterol is being investigated as a once-daily bronchodilator for treating adults with chronic obstructive pulmonary disease (COPD).17 Novartis has received a complete response letter from the FDA requesting additional data on the dosing of the agent. The company is working with the FDA to resolve these issues.
- Rituximab injection (Rituxan) is approved for treating moderate to severe rheumatoid arthritis after patients have been treated with methotrexate, as well as non-Hodgkin’s lymphoma.18 Genentech/Biogen is attempting to expand rituximab use to treat patients earlier in the course of their disease. The FDA recently rejected this application, citing the rare but serious safety risk of developing progressive multifocal leukoencephalopathy.
Safety Information
Use of omeprazole and clopidogrel combination therapy should be avoided, according to new data from the FDA.18 Current data suggest that clopidogrel’s antiplatelet effect may be hindered by 50% with concomitant omeprazole therapy. Omeprazole blocks the conversion of clopidogrel to its active, antiplatelet form, thus significantly decreasing its effectiveness. It is not known if other proton-pump inhibitors interfere with clopidogrel’s effectiveness. Other drugs that should not be used with clopidogrel include esomeprazole (Nexium), cimetidine (Tagamet and Tagamet HB), fluconazole (Diflucan), ketoconazole (Nizoral), voriconazole (VFEND), etravirine (Intelence), felbamate (Felbatol), fluoxetine (Prozac, Sarafem, and Symbyax), fluvoxamine (Luvox), and ticlopidine (Ticlid). TH
Michele B. Kaufman is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. FDA Web site. Available at: www.accessdata.fda.gov/scripts/cder/ob/docs/obdetail.cfm?Appl_No=077312&TABLE1=OB_Rx. Accessed Nov. 19, 2009.
- Covidien gets approval of generic pain drug. Forbes.com Web site. Available at: http://www.forbes.com/feeds/ap/2009/10/30/business-health-care-us-covidien-actiq_7069457.html. Accessed Nov. 19, 2009.
- Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. FDA Web site. Available at: www.accessdata.fda.gov/scripts/cder/ob/default.cfm Accessed Nov. 19, 2009.
- Riley K. FDA Approves New Drug Treatment for Long-Term Pain Relief after Shingles Attacks. FDA Web site. Available at: www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm191003.htm. Accessed Nov. 19, 2009.
- Qutenza Approved for Post-Shingles Nerve Pain. U.S. News and World Report Web site. Available at: health.usnews.com/articles/health/healthday/2009/11/17/qutenza-approved-for-post-shingles-nerve-pain.html. Accessed Nov. 19, 2009.
- Nuvo shares surge on FDA pain cream approval. Reuters Web site. Available at: www.reuters.com/article/email/idUSN0543459820091105. Accessed Nov. 19, 2009.
- Pennsaid. Nuvo Web site. Available at: www.nuvoresearch.com/pipeline/pennsaid.asp. Accessed Nov. 19, 2009.
- BYETTA Approved For Expanded Use As First-Line Treatment For Type 2 Diabetes. Medical News Today Web site. Available at: www.medicalnewstoday.com/articles/169396.php. Accessed Nov. 19, 2009.
- Burgess S. FDA Approves New Indication for Gardasil to Prevent Genital Warts in Men and Boys. FDA Web site. Available at: www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm187003.htm Accessed Nov. 19, 2009.
- Prevacid 24hr available over-the-counter. Monthly Prescribing Reference Web site. Available at: www.empr.com/prevacid-24hr-available-over-the-counter/article/157592/. Accessed Nov. 19, 2009.
- FDA Approves New Salix Product Exclusively in Catalent’s Zydis Fast Dissolve Technology. Catalent Web site. Available at: www.catalent.com/about-us/news/45. Accessed Nov. 19, 2009.
- Peramivir to be Used for Special Swine Flu Cases. Pharmaceutical-Technology.com Web site. Available at: www.pharmaceutical-technology.com/News/News68102.html. Accessed Nov. 19, 2009.
- Dennis M. AstraZeneca’s Crestor approved in US for paediatric use; Vimovo filed in Europe. FirstWord Web site. Available at: www.firstwordplus.com/Fws.do?articleid=41E54BF434C54C8E903A9EB4EF0839AA&logRowId=332380. Accessed Nov. 19, 2009.
- FDA Approves New Use for Micardis in Cardiovascular Risk Reduction and Twynsta as New Combination Treatment for High Blood Pressure. Boehringer Ingelheim Web site. Available at: us.boehringer-ingelheim.com/newsroom/2009/files/micardis_twynsta_approve_10-19-09.pdf. Accessed Nov. 19, 2009.
- Todoruk M. FDA approves Xanodyne’s Lysteda for menorrhaegia. FirstWord Web site. Available at: www.firstwordplus.com/Fws.do?articleid=8FCB424A8E7147A08CEC9BFCD8A450D2&logRowId=337054. Accessed Nov. 19, 2009.
- Xanodyne announces FDA approval of Lysteda for treatment of women with heavy menstrual bleeding. Xanodyne Web site. Available at: www.xanodyne.com/newsroom_details.asp?NewsId=61. Accessed Nov. 19, 2009.
- Todoruk M. FDA requests additional data for Novartis’ COPD drug indacaterol. FirstWord Web site. Available at: www.firstwordplus.com/Fws.do?articleid=B426F5ED30AB43A89338DAD0124168A7&logRowId=332718. Accessed Nov. 19, 2009.
- Genentech and Biogen Idec Receive a Complete Response from FDA for Earlier Use of Rituxan for Rheumatoid Arthritis. Genentech Web site. Available at: www.gene.com/gene/news/press-releases/display.do?method=detail&id=12407. Accessed Nov. 19, 2009.
- Walsh S. FDA Announces New Warning on Plavix: Avoid Use with Prilosec/Prilosec OTC. FDA Web site. Available at www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm191169.htm. Accessed Nov. 19, 2009.
New Generics
- Fentanyl citrate troche/lozenge (generic Actiq) transmucosal lollipop1,2
- Perindopril erbumine (generic Aceon) tablets3
New Drugs, Indications, Label Changes, and Dosage Forms
- Capsaicin 8% patch (Qutenza) has been approved by the U.S. Food and Drug Administration (FDA) as a prescription medication to treat pain associated with post-herpetic neuralgia.4 The patch is a higher concentration than over-the-counter (OTC) products. The most common side effects in clinical trials were pain, swelling, itching, redness, and bumps at the application site, as well as blood pressure increases.5 Patch placement requires the use of a topical anesthetic and additional pain relief (ice or opioid pain relievers). Therefore, patch placement must be performed by a healthcare professional. Once the patch is applied, the patient must be observed for at least an hour, as there could be a significant increase in blood pressure.
- Diclofenac sodium topical (Pennsaid) has been approved by the FDA as a topical treatment to manage knee osteoarthritis.6 The transdermal carrier dimethylsulfoxide (DMSO) is utilized to deliver active diclofenac sodium through the skin to the pain site.7
- Exenatide injection (Byetta) has been approved by the FDA as monotherapy for treating patients with Type 2 diabetes mellitus, along with diet and exercise.8
- Human papilloma virus (HPV) vaccine (Gardasil) has been approved by the FDA for preventing condyloma acuminata due to HPV types 6 and 11 in males ages 9 to 26.9 The makers hope that this vaccine will decrease the need to treat genital warts. Clinical studies showed that in males not infected with HPV at the beginning of the study, the vaccine was close to 90% effective in preventing genital warts caused by infection of HPV types 6 and 11. The manufacturer plans to obtain additional safety and effectiveness information in this patient population.
- Lansoprazole 24HR (Prevacid OTC) 15-mg delayed-release capsules are available for treating frequent heartburn.10 The capsules will be available by prescription in both the 15-mg and 30-mg dosage strengths.
- Metoclopramide HCl orally disintegrating tablet (Metozolv ODT) has been approved by the FDA for treating both acute and recurrent diabetic gastroparesis and for the short-term management (four to 12 weeks) of adults with documented symptomatic gastroesophageal reflux disease who do not respond to conventional therapy.11
- Peramivir intravenous will be made available by the Centers for Disease Control (CDC) as an emergency treatment for children and adult patients who develop H1N1 influenza and are nonresponsive to oral or inhaled antiviral therapies.12
- Rosuvastatin (Crestor) has been approved by the FDA for treating heterozygous familial cholesterolemia in children ages 10-17.13
- Telmisartan 80-mg tablets (Micardis) have been approved by the FDA for risk reduction of myocardial infarction, stroke, or death from cardiovascular causes in patients 55 years and older who are at high risk for major cardiovascular events, or who are not able to take angiotensin-converting enzyme inhibitors.14
- Telmisartan/amlodipine tablets (Twynsta) have been approved by the FDA as a new combination for treating hypertension, either alone or in combination with other antihypertensive agents. This combination is not indicated for cardiovascular risk reduction. The tablets are available in the following strengths of telmisartan/amlodipine, respectively: 40 mg/5 mg, 40 mg/10 mg, 80 mg/5 mg, and 80 mg/10 mg.
- Tranexamic acid (Lysteda) has been approved by the FDA for the treatment of menorrhagia.15 This is the first nonhormonal, oral therapeutic agent approved to treat this condition.16
Pipeline
- Indacaterol is being investigated as a once-daily bronchodilator for treating adults with chronic obstructive pulmonary disease (COPD).17 Novartis has received a complete response letter from the FDA requesting additional data on the dosing of the agent. The company is working with the FDA to resolve these issues.
- Rituximab injection (Rituxan) is approved for treating moderate to severe rheumatoid arthritis after patients have been treated with methotrexate, as well as non-Hodgkin’s lymphoma.18 Genentech/Biogen is attempting to expand rituximab use to treat patients earlier in the course of their disease. The FDA recently rejected this application, citing the rare but serious safety risk of developing progressive multifocal leukoencephalopathy.
Safety Information
Use of omeprazole and clopidogrel combination therapy should be avoided, according to new data from the FDA.18 Current data suggest that clopidogrel’s antiplatelet effect may be hindered by 50% with concomitant omeprazole therapy. Omeprazole blocks the conversion of clopidogrel to its active, antiplatelet form, thus significantly decreasing its effectiveness. It is not known if other proton-pump inhibitors interfere with clopidogrel’s effectiveness. Other drugs that should not be used with clopidogrel include esomeprazole (Nexium), cimetidine (Tagamet and Tagamet HB), fluconazole (Diflucan), ketoconazole (Nizoral), voriconazole (VFEND), etravirine (Intelence), felbamate (Felbatol), fluoxetine (Prozac, Sarafem, and Symbyax), fluvoxamine (Luvox), and ticlopidine (Ticlid). TH
Michele B. Kaufman is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. FDA Web site. Available at: www.accessdata.fda.gov/scripts/cder/ob/docs/obdetail.cfm?Appl_No=077312&TABLE1=OB_Rx. Accessed Nov. 19, 2009.
- Covidien gets approval of generic pain drug. Forbes.com Web site. Available at: http://www.forbes.com/feeds/ap/2009/10/30/business-health-care-us-covidien-actiq_7069457.html. Accessed Nov. 19, 2009.
- Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. FDA Web site. Available at: www.accessdata.fda.gov/scripts/cder/ob/default.cfm Accessed Nov. 19, 2009.
- Riley K. FDA Approves New Drug Treatment for Long-Term Pain Relief after Shingles Attacks. FDA Web site. Available at: www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm191003.htm. Accessed Nov. 19, 2009.
- Qutenza Approved for Post-Shingles Nerve Pain. U.S. News and World Report Web site. Available at: health.usnews.com/articles/health/healthday/2009/11/17/qutenza-approved-for-post-shingles-nerve-pain.html. Accessed Nov. 19, 2009.
- Nuvo shares surge on FDA pain cream approval. Reuters Web site. Available at: www.reuters.com/article/email/idUSN0543459820091105. Accessed Nov. 19, 2009.
- Pennsaid. Nuvo Web site. Available at: www.nuvoresearch.com/pipeline/pennsaid.asp. Accessed Nov. 19, 2009.
- BYETTA Approved For Expanded Use As First-Line Treatment For Type 2 Diabetes. Medical News Today Web site. Available at: www.medicalnewstoday.com/articles/169396.php. Accessed Nov. 19, 2009.
- Burgess S. FDA Approves New Indication for Gardasil to Prevent Genital Warts in Men and Boys. FDA Web site. Available at: www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm187003.htm Accessed Nov. 19, 2009.
- Prevacid 24hr available over-the-counter. Monthly Prescribing Reference Web site. Available at: www.empr.com/prevacid-24hr-available-over-the-counter/article/157592/. Accessed Nov. 19, 2009.
- FDA Approves New Salix Product Exclusively in Catalent’s Zydis Fast Dissolve Technology. Catalent Web site. Available at: www.catalent.com/about-us/news/45. Accessed Nov. 19, 2009.
- Peramivir to be Used for Special Swine Flu Cases. Pharmaceutical-Technology.com Web site. Available at: www.pharmaceutical-technology.com/News/News68102.html. Accessed Nov. 19, 2009.
- Dennis M. AstraZeneca’s Crestor approved in US for paediatric use; Vimovo filed in Europe. FirstWord Web site. Available at: www.firstwordplus.com/Fws.do?articleid=41E54BF434C54C8E903A9EB4EF0839AA&logRowId=332380. Accessed Nov. 19, 2009.
- FDA Approves New Use for Micardis in Cardiovascular Risk Reduction and Twynsta as New Combination Treatment for High Blood Pressure. Boehringer Ingelheim Web site. Available at: us.boehringer-ingelheim.com/newsroom/2009/files/micardis_twynsta_approve_10-19-09.pdf. Accessed Nov. 19, 2009.
- Todoruk M. FDA approves Xanodyne’s Lysteda for menorrhaegia. FirstWord Web site. Available at: www.firstwordplus.com/Fws.do?articleid=8FCB424A8E7147A08CEC9BFCD8A450D2&logRowId=337054. Accessed Nov. 19, 2009.
- Xanodyne announces FDA approval of Lysteda for treatment of women with heavy menstrual bleeding. Xanodyne Web site. Available at: www.xanodyne.com/newsroom_details.asp?NewsId=61. Accessed Nov. 19, 2009.
- Todoruk M. FDA requests additional data for Novartis’ COPD drug indacaterol. FirstWord Web site. Available at: www.firstwordplus.com/Fws.do?articleid=B426F5ED30AB43A89338DAD0124168A7&logRowId=332718. Accessed Nov. 19, 2009.
- Genentech and Biogen Idec Receive a Complete Response from FDA for Earlier Use of Rituxan for Rheumatoid Arthritis. Genentech Web site. Available at: www.gene.com/gene/news/press-releases/display.do?method=detail&id=12407. Accessed Nov. 19, 2009.
- Walsh S. FDA Announces New Warning on Plavix: Avoid Use with Prilosec/Prilosec OTC. FDA Web site. Available at www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm191169.htm. Accessed Nov. 19, 2009.
New Generics
- Fentanyl citrate troche/lozenge (generic Actiq) transmucosal lollipop1,2
- Perindopril erbumine (generic Aceon) tablets3
New Drugs, Indications, Label Changes, and Dosage Forms
- Capsaicin 8% patch (Qutenza) has been approved by the U.S. Food and Drug Administration (FDA) as a prescription medication to treat pain associated with post-herpetic neuralgia.4 The patch is a higher concentration than over-the-counter (OTC) products. The most common side effects in clinical trials were pain, swelling, itching, redness, and bumps at the application site, as well as blood pressure increases.5 Patch placement requires the use of a topical anesthetic and additional pain relief (ice or opioid pain relievers). Therefore, patch placement must be performed by a healthcare professional. Once the patch is applied, the patient must be observed for at least an hour, as there could be a significant increase in blood pressure.
- Diclofenac sodium topical (Pennsaid) has been approved by the FDA as a topical treatment to manage knee osteoarthritis.6 The transdermal carrier dimethylsulfoxide (DMSO) is utilized to deliver active diclofenac sodium through the skin to the pain site.7
- Exenatide injection (Byetta) has been approved by the FDA as monotherapy for treating patients with Type 2 diabetes mellitus, along with diet and exercise.8
- Human papilloma virus (HPV) vaccine (Gardasil) has been approved by the FDA for preventing condyloma acuminata due to HPV types 6 and 11 in males ages 9 to 26.9 The makers hope that this vaccine will decrease the need to treat genital warts. Clinical studies showed that in males not infected with HPV at the beginning of the study, the vaccine was close to 90% effective in preventing genital warts caused by infection of HPV types 6 and 11. The manufacturer plans to obtain additional safety and effectiveness information in this patient population.
- Lansoprazole 24HR (Prevacid OTC) 15-mg delayed-release capsules are available for treating frequent heartburn.10 The capsules will be available by prescription in both the 15-mg and 30-mg dosage strengths.
- Metoclopramide HCl orally disintegrating tablet (Metozolv ODT) has been approved by the FDA for treating both acute and recurrent diabetic gastroparesis and for the short-term management (four to 12 weeks) of adults with documented symptomatic gastroesophageal reflux disease who do not respond to conventional therapy.11
- Peramivir intravenous will be made available by the Centers for Disease Control (CDC) as an emergency treatment for children and adult patients who develop H1N1 influenza and are nonresponsive to oral or inhaled antiviral therapies.12
- Rosuvastatin (Crestor) has been approved by the FDA for treating heterozygous familial cholesterolemia in children ages 10-17.13
- Telmisartan 80-mg tablets (Micardis) have been approved by the FDA for risk reduction of myocardial infarction, stroke, or death from cardiovascular causes in patients 55 years and older who are at high risk for major cardiovascular events, or who are not able to take angiotensin-converting enzyme inhibitors.14
- Telmisartan/amlodipine tablets (Twynsta) have been approved by the FDA as a new combination for treating hypertension, either alone or in combination with other antihypertensive agents. This combination is not indicated for cardiovascular risk reduction. The tablets are available in the following strengths of telmisartan/amlodipine, respectively: 40 mg/5 mg, 40 mg/10 mg, 80 mg/5 mg, and 80 mg/10 mg.
- Tranexamic acid (Lysteda) has been approved by the FDA for the treatment of menorrhagia.15 This is the first nonhormonal, oral therapeutic agent approved to treat this condition.16
Pipeline
- Indacaterol is being investigated as a once-daily bronchodilator for treating adults with chronic obstructive pulmonary disease (COPD).17 Novartis has received a complete response letter from the FDA requesting additional data on the dosing of the agent. The company is working with the FDA to resolve these issues.
- Rituximab injection (Rituxan) is approved for treating moderate to severe rheumatoid arthritis after patients have been treated with methotrexate, as well as non-Hodgkin’s lymphoma.18 Genentech/Biogen is attempting to expand rituximab use to treat patients earlier in the course of their disease. The FDA recently rejected this application, citing the rare but serious safety risk of developing progressive multifocal leukoencephalopathy.
Safety Information
Use of omeprazole and clopidogrel combination therapy should be avoided, according to new data from the FDA.18 Current data suggest that clopidogrel’s antiplatelet effect may be hindered by 50% with concomitant omeprazole therapy. Omeprazole blocks the conversion of clopidogrel to its active, antiplatelet form, thus significantly decreasing its effectiveness. It is not known if other proton-pump inhibitors interfere with clopidogrel’s effectiveness. Other drugs that should not be used with clopidogrel include esomeprazole (Nexium), cimetidine (Tagamet and Tagamet HB), fluconazole (Diflucan), ketoconazole (Nizoral), voriconazole (VFEND), etravirine (Intelence), felbamate (Felbatol), fluoxetine (Prozac, Sarafem, and Symbyax), fluvoxamine (Luvox), and ticlopidine (Ticlid). TH
Michele B. Kaufman is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. FDA Web site. Available at: www.accessdata.fda.gov/scripts/cder/ob/docs/obdetail.cfm?Appl_No=077312&TABLE1=OB_Rx. Accessed Nov. 19, 2009.
- Covidien gets approval of generic pain drug. Forbes.com Web site. Available at: http://www.forbes.com/feeds/ap/2009/10/30/business-health-care-us-covidien-actiq_7069457.html. Accessed Nov. 19, 2009.
- Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. FDA Web site. Available at: www.accessdata.fda.gov/scripts/cder/ob/default.cfm Accessed Nov. 19, 2009.
- Riley K. FDA Approves New Drug Treatment for Long-Term Pain Relief after Shingles Attacks. FDA Web site. Available at: www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm191003.htm. Accessed Nov. 19, 2009.
- Qutenza Approved for Post-Shingles Nerve Pain. U.S. News and World Report Web site. Available at: health.usnews.com/articles/health/healthday/2009/11/17/qutenza-approved-for-post-shingles-nerve-pain.html. Accessed Nov. 19, 2009.
- Nuvo shares surge on FDA pain cream approval. Reuters Web site. Available at: www.reuters.com/article/email/idUSN0543459820091105. Accessed Nov. 19, 2009.
- Pennsaid. Nuvo Web site. Available at: www.nuvoresearch.com/pipeline/pennsaid.asp. Accessed Nov. 19, 2009.
- BYETTA Approved For Expanded Use As First-Line Treatment For Type 2 Diabetes. Medical News Today Web site. Available at: www.medicalnewstoday.com/articles/169396.php. Accessed Nov. 19, 2009.
- Burgess S. FDA Approves New Indication for Gardasil to Prevent Genital Warts in Men and Boys. FDA Web site. Available at: www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm187003.htm Accessed Nov. 19, 2009.
- Prevacid 24hr available over-the-counter. Monthly Prescribing Reference Web site. Available at: www.empr.com/prevacid-24hr-available-over-the-counter/article/157592/. Accessed Nov. 19, 2009.
- FDA Approves New Salix Product Exclusively in Catalent’s Zydis Fast Dissolve Technology. Catalent Web site. Available at: www.catalent.com/about-us/news/45. Accessed Nov. 19, 2009.
- Peramivir to be Used for Special Swine Flu Cases. Pharmaceutical-Technology.com Web site. Available at: www.pharmaceutical-technology.com/News/News68102.html. Accessed Nov. 19, 2009.
- Dennis M. AstraZeneca’s Crestor approved in US for paediatric use; Vimovo filed in Europe. FirstWord Web site. Available at: www.firstwordplus.com/Fws.do?articleid=41E54BF434C54C8E903A9EB4EF0839AA&logRowId=332380. Accessed Nov. 19, 2009.
- FDA Approves New Use for Micardis in Cardiovascular Risk Reduction and Twynsta as New Combination Treatment for High Blood Pressure. Boehringer Ingelheim Web site. Available at: us.boehringer-ingelheim.com/newsroom/2009/files/micardis_twynsta_approve_10-19-09.pdf. Accessed Nov. 19, 2009.
- Todoruk M. FDA approves Xanodyne’s Lysteda for menorrhaegia. FirstWord Web site. Available at: www.firstwordplus.com/Fws.do?articleid=8FCB424A8E7147A08CEC9BFCD8A450D2&logRowId=337054. Accessed Nov. 19, 2009.
- Xanodyne announces FDA approval of Lysteda for treatment of women with heavy menstrual bleeding. Xanodyne Web site. Available at: www.xanodyne.com/newsroom_details.asp?NewsId=61. Accessed Nov. 19, 2009.
- Todoruk M. FDA requests additional data for Novartis’ COPD drug indacaterol. FirstWord Web site. Available at: www.firstwordplus.com/Fws.do?articleid=B426F5ED30AB43A89338DAD0124168A7&logRowId=332718. Accessed Nov. 19, 2009.
- Genentech and Biogen Idec Receive a Complete Response from FDA for Earlier Use of Rituxan for Rheumatoid Arthritis. Genentech Web site. Available at: www.gene.com/gene/news/press-releases/display.do?method=detail&id=12407. Accessed Nov. 19, 2009.
- Walsh S. FDA Announces New Warning on Plavix: Avoid Use with Prilosec/Prilosec OTC. FDA Web site. Available at www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm191169.htm. Accessed Nov. 19, 2009.
In the Literature: February 2010
In This Edition
Literature at a Glance
A guide to this month’s studies
- Perioperative oxygen use and infection rate.
- Effects of adverse events and healthcare costs
- Impact of DVT on PE rates in trauma
- VTE prevention and treatment in cancer patients
- Effect of perioperative beta-blocker discontinuation
- Endovascular vs. open AAA repair
- OTC analgesics in patients with hepatic dysfunction
- Cardiovascular disease and risk of hip fracture
High Perioperative Oxygen Fraction Does Not Improve Surgical-Site Infection Frequency after Abdominal Surgery
Clinical question: Does the use of 80% oxygen perioperatively in abdominal surgery decrease the frequency of surgical-site infection within 14 days without increasing the rate of pulmonary complications?
Background: Low oxygen tension in wounds can negatively impact immune response and healing. Increasing inspiratory oxygen fraction during the perioperative period translates into higher wound oxygen tension. However, the benefit of increased oxygen fraction therapy in abdominal surgery healing and complications is not clear, nor is the frequency of pulmonary complications.
Study design: Patient- and observer-blinded clinical trial.
Setting: Fourteen Danish hospitals from October 2006 to October 2008.
Synopsis: Patients were randomized to receive a fraction of inspired oxygen (FIO2) of 0.80 or 0.30. The primary outcome—surgical-site infection in the superficial or deep wound or intra-abdominal cavity within 14 days of surgery—was defined using Centers for Disease Control and Prevention (CDC) criteria. Secondary outcomes included pulmonary complications within 14 days (pneumonia, atelectasis, or respiratory failure), 30-day mortality, duration of post-op course, ICU stay within 14 days post-op, and any abdominal operation within 14 days. The 1,386 patients were enrolled in the intention-to-treat analysis.
Infection occurred in 19.1% of patients given 0.80 FIO2 and in 20.1% of patients given 0.30 FIO2; odds ratio of 0.94 (95% CI 0.72 to 1.22; P=0.64). Numbers of pulmonary complications were not significantly different between the groups.
This trial included acute and nonacute laparotomies with followup for adverse outcomes. Study limitations included the inability to ensure that both groups received timely antibiotics and prevention for hypothermia. Of patients in the 30% FIO2 group, 7.3% required higher oxygen administration. Additionally, infection might have been underestimated in 11.3% of patients who were not followed up on between days 13 and 30.
Bottom line: High oxygen concentration administered during and after laparotomy did not lead to fewer surgical site infections, nor did it significantly increase the frequency of pulmonary complications or death.
Citation: Meyhoff CS, Wetterslev J, Jorgensen LN, et al. Effect of high perioperative oxygen fraction on surgical site infection and pulmonary complications after abdominal surgery: the PROXI randomized clinical trial. JAMA. 2009;302(14):1543-1550.
Eliminating Adverse Events and Redundant Tests Could Generate U.S. Healthcare Savings
Clinical question: Using available data, what is the estimated cost savings of eliminating adverse events and avoiding redundant tests?
Background: Reimbursement schemes are changing such that hospitals are reimbursed less for some adverse events. This financial disincentive is expected to spark interest in improved patient safety. The authors sought to model the cost savings generated by eliminating redundant testing and adverse events from literature-based estimates.
Study design: Development of conceptual model to identify common or costly adverse events, redundant tests, and simulated costs.
Setting: Literature review, expert opinion, data from safety organizations and epidemiologic studies, and patient data from the 2004 National Inpatient Data Sample.
Synopsis: The conceptual model identified 5.7 million adverse events in U.S. hospitals, of which 3 million were considered preventable. The most common events included hospital-acquired infections (82% preventable), adverse drug events (26%), falls (33%), and iatrogenic thromboembolic events (62%). The calculated cost savings totaled $16.6 billion (5.5% of total inpatient costs) for adverse events and $8.2 billion for the elimination of redundant tests. When looking at hospital subtypes, the greatest savings would come from major teaching hospitals.
This study is limited by its use of published and heterogeneous data spanning a 15-year period. The authors did not include events for which there was no epidemiologic or cost data. As hospital-care changes and technology is adopted, it is uncertain how this changes the costs, prevalence, and the preventable nature of these events. The model was not consistently able to identifying high- and low-risk patients. For instance, in some models, all patients were considered at risk for events.
Bottom line: Based on a conceptual model of 2004 hospitalized patients, eliminating preventable adverse events could have saved $16.6 billion, while eliminating redundant tests could have saved another $8 billion.
Citation: Jha AK, Chan DC, Ridgway AB, Franz C, Bates DW. Improving safety and eliminating redundant tests: cutting costs in U.S. hospitals. Health Aff (Millwood). 2009;28(5):1475-1484.
Trauma Patients with Pulmonary Embolism Might Not Have DVT on Imaging of Lower Extremities
Clinical question: What is the relationship between acute DVT and pulmonary embolism (PE) in trauma patients?
Background: Major trauma is associated with an increased risk of acute DVT and PE. It is assumed that the majority of PEs arise from DVTs in the lower extremities. Definitive evidence demonstrating that PEs form in situ rather than embolize from leg veins could impact indications for inferior vena cava filters.
Study design: Retrospective chart review.
Setting: Academic Level 1 trauma center in Boston.
Synopsis: The medical records of 247 trauma patients with suspected PE who underwent CT angiography of the lungs and simultaneous CT venography of the pelvis and lower extremities from January 2004 to December 2007 were reviewed. High-risk patients also underwent weekly screening with duplex ultrasonagraphy of the legs.
PE was diagnosed in 46 patients (19%) and DVT in 18 patients (7%). Anticoagulant prophylaxis had been administered to 96% and 78% of the patients with PE and DVT, respectively. PE was diagnosed a median of 5.5 days after admission (range 0-40 days) and the majority (61%) were in segmental or subsegmental branches, rather than in the main or lobar pulmonary arteries (39%). Only seven of the 46 patients (15%) diagnosed with PE also had a pelvic or lower-extremity DVT on simultaneous imaging with CT venography.
Bottom line: Trauma patients with PE often do not have a DVT at the time of diagnosis, though it remains unknown whether this is due to in-situ pulmonary thrombosis or complete embolization from the lower extremities.
Citation: Velmahos GC, Spaniolas K, Tabbara M, et al. Pulmonary embolism and deep venous thrombosis in trauma: are they related? Arch Surg. 2009;144:928-932.
Cancer Guideline for VTE Prophylaxis for Inpatients and Long-Term Treatment With Low-Molecular-Weight Heparin for Acute VTE
Clinical question: On what aspects of VTE management in cancer patients are there consensus among the major guideline panels?
Background: VTE is a common and serious complication of cancer. Patients might be hypercoagulable due to prothrombotic mediators released or mediated by tumor cells, chemotherapeutic agents, debility, central venous catheters, hospitalizations, or surgical procedures. The optimal management often is problematic due to uncertain benefit and risk of bleeding.
Study design: Review of major guideline statements.
Synopsis: The authors examined five VTE guidelines of American and European cancer societies. Each guideline was reviewed to determine the main recommendations and whether there was consensus on key aspects of anticoagulant management.
The study authors concluded that consensus was reached on most key recommendations:
- VTE prophylaxis in hospitalized medical patients. All five guidelines recommend the use of prophylaxis, though some guidelines recommend anticoagulant prophylaxis for all inpatients in the absence of contraindications and some recommend limiting prophylaxis to immobilized patients. All five recommend the use of either unfractionated heparin, low-molecular-weight heparin (LMWH), or fondaparinux.
- VTE prevention in cancer patients undergoing surgery. All five guidelines recommend anticoagulant prophylaxis in the absence of contraindications and extending prophylaxis approximately four weeks after major surgery.
- VTE prophylaxis in cancer patients with central venous catheters. Not recommended.
- VTE prophylaxis in ambulatory cancer patients without central venous catheters. Recommended only for multiple myeloma patients receiving a thalidomide-lenalidomide regimen.
- Long-term treatment of acute VTE in cancer patients. All five guidelines recommend initial treatment with LMWH for at least three to six months, followed by indefinite treatment with LMWH or a vitamin K antagonist.
Bottom line: Major guideline panels agree on key aspects of VTE management for cancer patients, including the use of prophylaxis for hospitalized medical and surgical patients and the use of long-term LMWH treatment for cancer patients with acute VTE.
Citation: Khorana AA, Streiff MB, Farge D, et al. Venous thromboembolism prophylaxis and treatment in cancer: a consensus statement of major guidelines panels and call to action. J Clin Oncol. 2009; 27(29):4919-4926.
Discontinuation of Beta Blockers Increases Risk of Postoperative Myocardial Infarction and Death
Clinical question: Does perioperative beta-blocker discontinuation affect postoperative myocardial infarction (MI) in low-risk patients undergoing joint arthroplasty?
Background: Recent trials show no benefit of perioperative beta blockers in reducing the incidence of perioperative myocardial infarctions (POMI) in low-risk patients. This retrospective study examined the impact of continuing or discontinuing beta blockers and the occurrence of POMI in patients undergoing elective joint arthroplasties.
Study design: Retrospective chart review.
Setting: Large academic center in Ottawa, Canada.
Synopsis: Medical records for 5,178 patients undergoing elective hip or knee arthroplasty from January 2002 to June 2006 were included in the review. The primary outcome was POMI, defined as an increased troponin level. Patients were divided into three groups: beta blocker prescribed on post-operative day (POD) zero and continued for one week or until discharge; beta blocker prescribed on POD zero and discontinued at any time in the first week; and no beta blocker on POD 0.
Beta blockers were continued in 992 patients and discontinued in 252 patients. The rate of POMI and death increased in the beta-blocker discontinuation group (odds ratio 2.0 [1.1-3.9] and 2.0 [1.1-3.9], respectively). This association persisted after adjustment for cardiac risk using a validated risk score.
The study was limited by the fact that the control group did not include patients who were on a beta blocker at home, thus potentially increasing the number of events in this group. The discontinuation beta blocker group had an increased baseline risk for POMI. The reason for discontinuing the beta blocker was not known, and cessation of beta blocker could have been due to an acute event.
Bottom line: This study adds support to the American College of Cardiology and American Heart Association (ACC/AHA) guidelines, which recommend continuation of beta-blocker therapy in the perioperative period.
Citation: Van Klei WA, Bryson GL, Yang H, Forster AJ. Effect of beta-blocker prescription on the incidence of postoperative myocardial infarction after hip and knee arthroplasty. Anesthesiology. 2009;111(4):717-724.
Lower Perioperative Mortality with Endovascular Vs. Open Abdominal Aortic Aneurysm Repair
Clinical question: How do perioperative and long-term morbidity and mortality compare in endovascular and open repair of abdominal aortic aneurysm (AAA)?
Background: Open AAA repair has relatively high perioperative mortality. Endovascular repair was developed as a less-invasive option and has been shown to reduce inpatient perioperative mortality, length of hospital stay, and ICU requirement. However, data suggest it leads to more frequent reinterventions and the same mortality rate as open repair at two years.
Study design: Randomized clinical trial.
Setting: Veterans Affairs medical centers.
Synopsis: The study randomized 881 veterans who planned to have elective AAA repair and were eligible for both endovascular and open repair. This is a planned, two-year interim report in a nine-year study.
Perioperative mortality was 0.5% in the endovascular repair group, compared with 3.0% in the open repair group. However, this difference in mortality was not statistically significant at two years. The endovascular repair group experienced shorter procedure and mechanical ventilation time, decreased hospital and ICU stay, and lower rate of blood transfusions.
Overall, there was no difference between the groups for major morbidity, procedure failure, need for secondary therapeutic intervention, quality of life, or erectile dysfunction. More data on long-term comparison of these two interventions will be available at the conclusion of this study.
Bottom line: Endovascular repair of AAA has lower perioperative mortality than open repair but did not lead to improved morbidity or mortality at two years.
Citation: Lederle FA, Freischlag JA, Kyriakides TC, et al. Outcomes following endovascular vs. open repair of abdominal aortic aneurysm: a randomized trial. JAMA. 2009;302 (14):1535-1542.
OTC Analgesics Not Associated with Acute Decompensation in Cirrhotic Patients
Clinical question: Do over-the-counter (OTC) analgesics lead to acute hepatic decompensation among patients with cirrhosis?
Background: In theory, intake of acetaminophen and/or nonsteroidal anti-inflammatory drugs (NSAIDs) can worsen hepatic function and lead to complications among cirrhotic patients. The role of OTC analgesics in potentially triggering acute hepatic decompensation among cirrhotic patients has not been studied.
Study design: Prospective case-control study.
Setting: Two tertiary-care hospitals.
Synopsis: Cirrhotic patients hospitalized for acute liver decompensation were compared with compensated cirrhotic patients in the liver clinic (cirrhotic controls) and with randomly selected, noncirrhotic patients who were simultaneously hospitalized (noncirrhotic controls). Data collected through questionnaires included quantity and dose of OTC analgesics used and alcohol consumption in the past 30 days.
Thirty-five percent of the hospitalized cirrhotic patients, 52% of the cirrhotic controls, and 70% of the noncirrhotic controls used OTC analgesics. At doses lower than those recommended, acetaminophen is not associated with acute liver decompensation among cirrhotic patients, even with recent alcohol use. However, NSAIDs taken by the cirrhotic patients, when compared to control subjects, were in larger doses and used for a longer duration, suggesting NSAIDs may have contributed to the acute decompensation.
Study limitations include the nature of the study design, reliance on the patient’s recall of OTC analgesic use, and obtaining other possible causes of decompensation, such as herbal supplement intake or compliance with diuretics or dietary indiscretion.
Bottom line: Acetaminophen at doses lower than recommended is not associated with adverse complications in cirrhotic patients, but NSAIDs are possibly associated with acute decompensation.
Citation: Khalid SK, Lane J, Navarro V, Garcia-Tsao G. Use of over-the-counter analgesics is not associated with acute decompensation in patients with cirrhosis. Clin Gastroenterol Hepatol. 2009;7(9):994-999.
Cardiovascular Disease and Risk of Hip Fracture
Clinical question: Is the diagnosis of cardiovascular disease (CVD) associated with the risk of subsequent hip fracture?
Background: Osteoporosis and CVD are regarded as independent, age-related conditions. However, recent research suggests that the bone and vascular systems share common regulatory mechanisms. Stroke is a known risk factor for hip fractures, and bisphosphonates have been shown to prevent atherosclerosis and reduce total mortality rate.
Study design: Cohort study.
Setting: Swedish National Patient Registry.
Synopsis: The study identified 31,936 Swedish twins born from 1914 to 1944. This cohort was followed up to age 50, and time-dependent exposures using Cox-proportional hazard regression models were evaluated.
Times to hip fracture after CVD diagnosis were isolated. Crude absolute rate of hip fractures (per 1,000 person-years) was 12.6 after diagnosis of heart failure, 12.6 after a stroke, 6.6 after peripheral atherosclerosis, and 5.2 after ischemic heart disease (IHD), compared with 1.2 per 1,000 person-years without a CVD diagnosis. Multivariable-adjusted hazard ratio (HR) of hip fracture after heart failure was 4.40 (95% CI, 3.43-5.63); after a stroke was 5.09 (95% CI, 4.18-6.20); after peripheral atherosclerosis was 3.20 (CI, 2.28-4.50); and after an IHD event was 2.32 (CI, 1.91-2.84).
Identical twins even without heart failure and stroke also had an increased risk of hip fracture if their twin had been diagnosed with these diseases.
Bottom line: Cardiovascular disease is significantly associated with risk of subsequent hip fracture, and genetic factors probably play a role in the association.
Citation: Sennerby U, Melhus H, Gedeborg R, et al. Cardiovascular diseases and risk of hip fracture. JAMA. 2009;302(15):1666-1673. TH
In This Edition
Literature at a Glance
A guide to this month’s studies
- Perioperative oxygen use and infection rate.
- Effects of adverse events and healthcare costs
- Impact of DVT on PE rates in trauma
- VTE prevention and treatment in cancer patients
- Effect of perioperative beta-blocker discontinuation
- Endovascular vs. open AAA repair
- OTC analgesics in patients with hepatic dysfunction
- Cardiovascular disease and risk of hip fracture
High Perioperative Oxygen Fraction Does Not Improve Surgical-Site Infection Frequency after Abdominal Surgery
Clinical question: Does the use of 80% oxygen perioperatively in abdominal surgery decrease the frequency of surgical-site infection within 14 days without increasing the rate of pulmonary complications?
Background: Low oxygen tension in wounds can negatively impact immune response and healing. Increasing inspiratory oxygen fraction during the perioperative period translates into higher wound oxygen tension. However, the benefit of increased oxygen fraction therapy in abdominal surgery healing and complications is not clear, nor is the frequency of pulmonary complications.
Study design: Patient- and observer-blinded clinical trial.
Setting: Fourteen Danish hospitals from October 2006 to October 2008.
Synopsis: Patients were randomized to receive a fraction of inspired oxygen (FIO2) of 0.80 or 0.30. The primary outcome—surgical-site infection in the superficial or deep wound or intra-abdominal cavity within 14 days of surgery—was defined using Centers for Disease Control and Prevention (CDC) criteria. Secondary outcomes included pulmonary complications within 14 days (pneumonia, atelectasis, or respiratory failure), 30-day mortality, duration of post-op course, ICU stay within 14 days post-op, and any abdominal operation within 14 days. The 1,386 patients were enrolled in the intention-to-treat analysis.
Infection occurred in 19.1% of patients given 0.80 FIO2 and in 20.1% of patients given 0.30 FIO2; odds ratio of 0.94 (95% CI 0.72 to 1.22; P=0.64). Numbers of pulmonary complications were not significantly different between the groups.
This trial included acute and nonacute laparotomies with followup for adverse outcomes. Study limitations included the inability to ensure that both groups received timely antibiotics and prevention for hypothermia. Of patients in the 30% FIO2 group, 7.3% required higher oxygen administration. Additionally, infection might have been underestimated in 11.3% of patients who were not followed up on between days 13 and 30.
Bottom line: High oxygen concentration administered during and after laparotomy did not lead to fewer surgical site infections, nor did it significantly increase the frequency of pulmonary complications or death.
Citation: Meyhoff CS, Wetterslev J, Jorgensen LN, et al. Effect of high perioperative oxygen fraction on surgical site infection and pulmonary complications after abdominal surgery: the PROXI randomized clinical trial. JAMA. 2009;302(14):1543-1550.
Eliminating Adverse Events and Redundant Tests Could Generate U.S. Healthcare Savings
Clinical question: Using available data, what is the estimated cost savings of eliminating adverse events and avoiding redundant tests?
Background: Reimbursement schemes are changing such that hospitals are reimbursed less for some adverse events. This financial disincentive is expected to spark interest in improved patient safety. The authors sought to model the cost savings generated by eliminating redundant testing and adverse events from literature-based estimates.
Study design: Development of conceptual model to identify common or costly adverse events, redundant tests, and simulated costs.
Setting: Literature review, expert opinion, data from safety organizations and epidemiologic studies, and patient data from the 2004 National Inpatient Data Sample.
Synopsis: The conceptual model identified 5.7 million adverse events in U.S. hospitals, of which 3 million were considered preventable. The most common events included hospital-acquired infections (82% preventable), adverse drug events (26%), falls (33%), and iatrogenic thromboembolic events (62%). The calculated cost savings totaled $16.6 billion (5.5% of total inpatient costs) for adverse events and $8.2 billion for the elimination of redundant tests. When looking at hospital subtypes, the greatest savings would come from major teaching hospitals.
This study is limited by its use of published and heterogeneous data spanning a 15-year period. The authors did not include events for which there was no epidemiologic or cost data. As hospital-care changes and technology is adopted, it is uncertain how this changes the costs, prevalence, and the preventable nature of these events. The model was not consistently able to identifying high- and low-risk patients. For instance, in some models, all patients were considered at risk for events.
Bottom line: Based on a conceptual model of 2004 hospitalized patients, eliminating preventable adverse events could have saved $16.6 billion, while eliminating redundant tests could have saved another $8 billion.
Citation: Jha AK, Chan DC, Ridgway AB, Franz C, Bates DW. Improving safety and eliminating redundant tests: cutting costs in U.S. hospitals. Health Aff (Millwood). 2009;28(5):1475-1484.
Trauma Patients with Pulmonary Embolism Might Not Have DVT on Imaging of Lower Extremities
Clinical question: What is the relationship between acute DVT and pulmonary embolism (PE) in trauma patients?
Background: Major trauma is associated with an increased risk of acute DVT and PE. It is assumed that the majority of PEs arise from DVTs in the lower extremities. Definitive evidence demonstrating that PEs form in situ rather than embolize from leg veins could impact indications for inferior vena cava filters.
Study design: Retrospective chart review.
Setting: Academic Level 1 trauma center in Boston.
Synopsis: The medical records of 247 trauma patients with suspected PE who underwent CT angiography of the lungs and simultaneous CT venography of the pelvis and lower extremities from January 2004 to December 2007 were reviewed. High-risk patients also underwent weekly screening with duplex ultrasonagraphy of the legs.
PE was diagnosed in 46 patients (19%) and DVT in 18 patients (7%). Anticoagulant prophylaxis had been administered to 96% and 78% of the patients with PE and DVT, respectively. PE was diagnosed a median of 5.5 days after admission (range 0-40 days) and the majority (61%) were in segmental or subsegmental branches, rather than in the main or lobar pulmonary arteries (39%). Only seven of the 46 patients (15%) diagnosed with PE also had a pelvic or lower-extremity DVT on simultaneous imaging with CT venography.
Bottom line: Trauma patients with PE often do not have a DVT at the time of diagnosis, though it remains unknown whether this is due to in-situ pulmonary thrombosis or complete embolization from the lower extremities.
Citation: Velmahos GC, Spaniolas K, Tabbara M, et al. Pulmonary embolism and deep venous thrombosis in trauma: are they related? Arch Surg. 2009;144:928-932.
Cancer Guideline for VTE Prophylaxis for Inpatients and Long-Term Treatment With Low-Molecular-Weight Heparin for Acute VTE
Clinical question: On what aspects of VTE management in cancer patients are there consensus among the major guideline panels?
Background: VTE is a common and serious complication of cancer. Patients might be hypercoagulable due to prothrombotic mediators released or mediated by tumor cells, chemotherapeutic agents, debility, central venous catheters, hospitalizations, or surgical procedures. The optimal management often is problematic due to uncertain benefit and risk of bleeding.
Study design: Review of major guideline statements.
Synopsis: The authors examined five VTE guidelines of American and European cancer societies. Each guideline was reviewed to determine the main recommendations and whether there was consensus on key aspects of anticoagulant management.
The study authors concluded that consensus was reached on most key recommendations:
- VTE prophylaxis in hospitalized medical patients. All five guidelines recommend the use of prophylaxis, though some guidelines recommend anticoagulant prophylaxis for all inpatients in the absence of contraindications and some recommend limiting prophylaxis to immobilized patients. All five recommend the use of either unfractionated heparin, low-molecular-weight heparin (LMWH), or fondaparinux.
- VTE prevention in cancer patients undergoing surgery. All five guidelines recommend anticoagulant prophylaxis in the absence of contraindications and extending prophylaxis approximately four weeks after major surgery.
- VTE prophylaxis in cancer patients with central venous catheters. Not recommended.
- VTE prophylaxis in ambulatory cancer patients without central venous catheters. Recommended only for multiple myeloma patients receiving a thalidomide-lenalidomide regimen.
- Long-term treatment of acute VTE in cancer patients. All five guidelines recommend initial treatment with LMWH for at least three to six months, followed by indefinite treatment with LMWH or a vitamin K antagonist.
Bottom line: Major guideline panels agree on key aspects of VTE management for cancer patients, including the use of prophylaxis for hospitalized medical and surgical patients and the use of long-term LMWH treatment for cancer patients with acute VTE.
Citation: Khorana AA, Streiff MB, Farge D, et al. Venous thromboembolism prophylaxis and treatment in cancer: a consensus statement of major guidelines panels and call to action. J Clin Oncol. 2009; 27(29):4919-4926.
Discontinuation of Beta Blockers Increases Risk of Postoperative Myocardial Infarction and Death
Clinical question: Does perioperative beta-blocker discontinuation affect postoperative myocardial infarction (MI) in low-risk patients undergoing joint arthroplasty?
Background: Recent trials show no benefit of perioperative beta blockers in reducing the incidence of perioperative myocardial infarctions (POMI) in low-risk patients. This retrospective study examined the impact of continuing or discontinuing beta blockers and the occurrence of POMI in patients undergoing elective joint arthroplasties.
Study design: Retrospective chart review.
Setting: Large academic center in Ottawa, Canada.
Synopsis: Medical records for 5,178 patients undergoing elective hip or knee arthroplasty from January 2002 to June 2006 were included in the review. The primary outcome was POMI, defined as an increased troponin level. Patients were divided into three groups: beta blocker prescribed on post-operative day (POD) zero and continued for one week or until discharge; beta blocker prescribed on POD zero and discontinued at any time in the first week; and no beta blocker on POD 0.
Beta blockers were continued in 992 patients and discontinued in 252 patients. The rate of POMI and death increased in the beta-blocker discontinuation group (odds ratio 2.0 [1.1-3.9] and 2.0 [1.1-3.9], respectively). This association persisted after adjustment for cardiac risk using a validated risk score.
The study was limited by the fact that the control group did not include patients who were on a beta blocker at home, thus potentially increasing the number of events in this group. The discontinuation beta blocker group had an increased baseline risk for POMI. The reason for discontinuing the beta blocker was not known, and cessation of beta blocker could have been due to an acute event.
Bottom line: This study adds support to the American College of Cardiology and American Heart Association (ACC/AHA) guidelines, which recommend continuation of beta-blocker therapy in the perioperative period.
Citation: Van Klei WA, Bryson GL, Yang H, Forster AJ. Effect of beta-blocker prescription on the incidence of postoperative myocardial infarction after hip and knee arthroplasty. Anesthesiology. 2009;111(4):717-724.
Lower Perioperative Mortality with Endovascular Vs. Open Abdominal Aortic Aneurysm Repair
Clinical question: How do perioperative and long-term morbidity and mortality compare in endovascular and open repair of abdominal aortic aneurysm (AAA)?
Background: Open AAA repair has relatively high perioperative mortality. Endovascular repair was developed as a less-invasive option and has been shown to reduce inpatient perioperative mortality, length of hospital stay, and ICU requirement. However, data suggest it leads to more frequent reinterventions and the same mortality rate as open repair at two years.
Study design: Randomized clinical trial.
Setting: Veterans Affairs medical centers.
Synopsis: The study randomized 881 veterans who planned to have elective AAA repair and were eligible for both endovascular and open repair. This is a planned, two-year interim report in a nine-year study.
Perioperative mortality was 0.5% in the endovascular repair group, compared with 3.0% in the open repair group. However, this difference in mortality was not statistically significant at two years. The endovascular repair group experienced shorter procedure and mechanical ventilation time, decreased hospital and ICU stay, and lower rate of blood transfusions.
Overall, there was no difference between the groups for major morbidity, procedure failure, need for secondary therapeutic intervention, quality of life, or erectile dysfunction. More data on long-term comparison of these two interventions will be available at the conclusion of this study.
Bottom line: Endovascular repair of AAA has lower perioperative mortality than open repair but did not lead to improved morbidity or mortality at two years.
Citation: Lederle FA, Freischlag JA, Kyriakides TC, et al. Outcomes following endovascular vs. open repair of abdominal aortic aneurysm: a randomized trial. JAMA. 2009;302 (14):1535-1542.
OTC Analgesics Not Associated with Acute Decompensation in Cirrhotic Patients
Clinical question: Do over-the-counter (OTC) analgesics lead to acute hepatic decompensation among patients with cirrhosis?
Background: In theory, intake of acetaminophen and/or nonsteroidal anti-inflammatory drugs (NSAIDs) can worsen hepatic function and lead to complications among cirrhotic patients. The role of OTC analgesics in potentially triggering acute hepatic decompensation among cirrhotic patients has not been studied.
Study design: Prospective case-control study.
Setting: Two tertiary-care hospitals.
Synopsis: Cirrhotic patients hospitalized for acute liver decompensation were compared with compensated cirrhotic patients in the liver clinic (cirrhotic controls) and with randomly selected, noncirrhotic patients who were simultaneously hospitalized (noncirrhotic controls). Data collected through questionnaires included quantity and dose of OTC analgesics used and alcohol consumption in the past 30 days.
Thirty-five percent of the hospitalized cirrhotic patients, 52% of the cirrhotic controls, and 70% of the noncirrhotic controls used OTC analgesics. At doses lower than those recommended, acetaminophen is not associated with acute liver decompensation among cirrhotic patients, even with recent alcohol use. However, NSAIDs taken by the cirrhotic patients, when compared to control subjects, were in larger doses and used for a longer duration, suggesting NSAIDs may have contributed to the acute decompensation.
Study limitations include the nature of the study design, reliance on the patient’s recall of OTC analgesic use, and obtaining other possible causes of decompensation, such as herbal supplement intake or compliance with diuretics or dietary indiscretion.
Bottom line: Acetaminophen at doses lower than recommended is not associated with adverse complications in cirrhotic patients, but NSAIDs are possibly associated with acute decompensation.
Citation: Khalid SK, Lane J, Navarro V, Garcia-Tsao G. Use of over-the-counter analgesics is not associated with acute decompensation in patients with cirrhosis. Clin Gastroenterol Hepatol. 2009;7(9):994-999.
Cardiovascular Disease and Risk of Hip Fracture
Clinical question: Is the diagnosis of cardiovascular disease (CVD) associated with the risk of subsequent hip fracture?
Background: Osteoporosis and CVD are regarded as independent, age-related conditions. However, recent research suggests that the bone and vascular systems share common regulatory mechanisms. Stroke is a known risk factor for hip fractures, and bisphosphonates have been shown to prevent atherosclerosis and reduce total mortality rate.
Study design: Cohort study.
Setting: Swedish National Patient Registry.
Synopsis: The study identified 31,936 Swedish twins born from 1914 to 1944. This cohort was followed up to age 50, and time-dependent exposures using Cox-proportional hazard regression models were evaluated.
Times to hip fracture after CVD diagnosis were isolated. Crude absolute rate of hip fractures (per 1,000 person-years) was 12.6 after diagnosis of heart failure, 12.6 after a stroke, 6.6 after peripheral atherosclerosis, and 5.2 after ischemic heart disease (IHD), compared with 1.2 per 1,000 person-years without a CVD diagnosis. Multivariable-adjusted hazard ratio (HR) of hip fracture after heart failure was 4.40 (95% CI, 3.43-5.63); after a stroke was 5.09 (95% CI, 4.18-6.20); after peripheral atherosclerosis was 3.20 (CI, 2.28-4.50); and after an IHD event was 2.32 (CI, 1.91-2.84).
Identical twins even without heart failure and stroke also had an increased risk of hip fracture if their twin had been diagnosed with these diseases.
Bottom line: Cardiovascular disease is significantly associated with risk of subsequent hip fracture, and genetic factors probably play a role in the association.
Citation: Sennerby U, Melhus H, Gedeborg R, et al. Cardiovascular diseases and risk of hip fracture. JAMA. 2009;302(15):1666-1673. TH
In This Edition
Literature at a Glance
A guide to this month’s studies
- Perioperative oxygen use and infection rate.
- Effects of adverse events and healthcare costs
- Impact of DVT on PE rates in trauma
- VTE prevention and treatment in cancer patients
- Effect of perioperative beta-blocker discontinuation
- Endovascular vs. open AAA repair
- OTC analgesics in patients with hepatic dysfunction
- Cardiovascular disease and risk of hip fracture
High Perioperative Oxygen Fraction Does Not Improve Surgical-Site Infection Frequency after Abdominal Surgery
Clinical question: Does the use of 80% oxygen perioperatively in abdominal surgery decrease the frequency of surgical-site infection within 14 days without increasing the rate of pulmonary complications?
Background: Low oxygen tension in wounds can negatively impact immune response and healing. Increasing inspiratory oxygen fraction during the perioperative period translates into higher wound oxygen tension. However, the benefit of increased oxygen fraction therapy in abdominal surgery healing and complications is not clear, nor is the frequency of pulmonary complications.
Study design: Patient- and observer-blinded clinical trial.
Setting: Fourteen Danish hospitals from October 2006 to October 2008.
Synopsis: Patients were randomized to receive a fraction of inspired oxygen (FIO2) of 0.80 or 0.30. The primary outcome—surgical-site infection in the superficial or deep wound or intra-abdominal cavity within 14 days of surgery—was defined using Centers for Disease Control and Prevention (CDC) criteria. Secondary outcomes included pulmonary complications within 14 days (pneumonia, atelectasis, or respiratory failure), 30-day mortality, duration of post-op course, ICU stay within 14 days post-op, and any abdominal operation within 14 days. The 1,386 patients were enrolled in the intention-to-treat analysis.
Infection occurred in 19.1% of patients given 0.80 FIO2 and in 20.1% of patients given 0.30 FIO2; odds ratio of 0.94 (95% CI 0.72 to 1.22; P=0.64). Numbers of pulmonary complications were not significantly different between the groups.
This trial included acute and nonacute laparotomies with followup for adverse outcomes. Study limitations included the inability to ensure that both groups received timely antibiotics and prevention for hypothermia. Of patients in the 30% FIO2 group, 7.3% required higher oxygen administration. Additionally, infection might have been underestimated in 11.3% of patients who were not followed up on between days 13 and 30.
Bottom line: High oxygen concentration administered during and after laparotomy did not lead to fewer surgical site infections, nor did it significantly increase the frequency of pulmonary complications or death.
Citation: Meyhoff CS, Wetterslev J, Jorgensen LN, et al. Effect of high perioperative oxygen fraction on surgical site infection and pulmonary complications after abdominal surgery: the PROXI randomized clinical trial. JAMA. 2009;302(14):1543-1550.
Eliminating Adverse Events and Redundant Tests Could Generate U.S. Healthcare Savings
Clinical question: Using available data, what is the estimated cost savings of eliminating adverse events and avoiding redundant tests?
Background: Reimbursement schemes are changing such that hospitals are reimbursed less for some adverse events. This financial disincentive is expected to spark interest in improved patient safety. The authors sought to model the cost savings generated by eliminating redundant testing and adverse events from literature-based estimates.
Study design: Development of conceptual model to identify common or costly adverse events, redundant tests, and simulated costs.
Setting: Literature review, expert opinion, data from safety organizations and epidemiologic studies, and patient data from the 2004 National Inpatient Data Sample.
Synopsis: The conceptual model identified 5.7 million adverse events in U.S. hospitals, of which 3 million were considered preventable. The most common events included hospital-acquired infections (82% preventable), adverse drug events (26%), falls (33%), and iatrogenic thromboembolic events (62%). The calculated cost savings totaled $16.6 billion (5.5% of total inpatient costs) for adverse events and $8.2 billion for the elimination of redundant tests. When looking at hospital subtypes, the greatest savings would come from major teaching hospitals.
This study is limited by its use of published and heterogeneous data spanning a 15-year period. The authors did not include events for which there was no epidemiologic or cost data. As hospital-care changes and technology is adopted, it is uncertain how this changes the costs, prevalence, and the preventable nature of these events. The model was not consistently able to identifying high- and low-risk patients. For instance, in some models, all patients were considered at risk for events.
Bottom line: Based on a conceptual model of 2004 hospitalized patients, eliminating preventable adverse events could have saved $16.6 billion, while eliminating redundant tests could have saved another $8 billion.
Citation: Jha AK, Chan DC, Ridgway AB, Franz C, Bates DW. Improving safety and eliminating redundant tests: cutting costs in U.S. hospitals. Health Aff (Millwood). 2009;28(5):1475-1484.
Trauma Patients with Pulmonary Embolism Might Not Have DVT on Imaging of Lower Extremities
Clinical question: What is the relationship between acute DVT and pulmonary embolism (PE) in trauma patients?
Background: Major trauma is associated with an increased risk of acute DVT and PE. It is assumed that the majority of PEs arise from DVTs in the lower extremities. Definitive evidence demonstrating that PEs form in situ rather than embolize from leg veins could impact indications for inferior vena cava filters.
Study design: Retrospective chart review.
Setting: Academic Level 1 trauma center in Boston.
Synopsis: The medical records of 247 trauma patients with suspected PE who underwent CT angiography of the lungs and simultaneous CT venography of the pelvis and lower extremities from January 2004 to December 2007 were reviewed. High-risk patients also underwent weekly screening with duplex ultrasonagraphy of the legs.
PE was diagnosed in 46 patients (19%) and DVT in 18 patients (7%). Anticoagulant prophylaxis had been administered to 96% and 78% of the patients with PE and DVT, respectively. PE was diagnosed a median of 5.5 days after admission (range 0-40 days) and the majority (61%) were in segmental or subsegmental branches, rather than in the main or lobar pulmonary arteries (39%). Only seven of the 46 patients (15%) diagnosed with PE also had a pelvic or lower-extremity DVT on simultaneous imaging with CT venography.
Bottom line: Trauma patients with PE often do not have a DVT at the time of diagnosis, though it remains unknown whether this is due to in-situ pulmonary thrombosis or complete embolization from the lower extremities.
Citation: Velmahos GC, Spaniolas K, Tabbara M, et al. Pulmonary embolism and deep venous thrombosis in trauma: are they related? Arch Surg. 2009;144:928-932.
Cancer Guideline for VTE Prophylaxis for Inpatients and Long-Term Treatment With Low-Molecular-Weight Heparin for Acute VTE
Clinical question: On what aspects of VTE management in cancer patients are there consensus among the major guideline panels?
Background: VTE is a common and serious complication of cancer. Patients might be hypercoagulable due to prothrombotic mediators released or mediated by tumor cells, chemotherapeutic agents, debility, central venous catheters, hospitalizations, or surgical procedures. The optimal management often is problematic due to uncertain benefit and risk of bleeding.
Study design: Review of major guideline statements.
Synopsis: The authors examined five VTE guidelines of American and European cancer societies. Each guideline was reviewed to determine the main recommendations and whether there was consensus on key aspects of anticoagulant management.
The study authors concluded that consensus was reached on most key recommendations:
- VTE prophylaxis in hospitalized medical patients. All five guidelines recommend the use of prophylaxis, though some guidelines recommend anticoagulant prophylaxis for all inpatients in the absence of contraindications and some recommend limiting prophylaxis to immobilized patients. All five recommend the use of either unfractionated heparin, low-molecular-weight heparin (LMWH), or fondaparinux.
- VTE prevention in cancer patients undergoing surgery. All five guidelines recommend anticoagulant prophylaxis in the absence of contraindications and extending prophylaxis approximately four weeks after major surgery.
- VTE prophylaxis in cancer patients with central venous catheters. Not recommended.
- VTE prophylaxis in ambulatory cancer patients without central venous catheters. Recommended only for multiple myeloma patients receiving a thalidomide-lenalidomide regimen.
- Long-term treatment of acute VTE in cancer patients. All five guidelines recommend initial treatment with LMWH for at least three to six months, followed by indefinite treatment with LMWH or a vitamin K antagonist.
Bottom line: Major guideline panels agree on key aspects of VTE management for cancer patients, including the use of prophylaxis for hospitalized medical and surgical patients and the use of long-term LMWH treatment for cancer patients with acute VTE.
Citation: Khorana AA, Streiff MB, Farge D, et al. Venous thromboembolism prophylaxis and treatment in cancer: a consensus statement of major guidelines panels and call to action. J Clin Oncol. 2009; 27(29):4919-4926.
Discontinuation of Beta Blockers Increases Risk of Postoperative Myocardial Infarction and Death
Clinical question: Does perioperative beta-blocker discontinuation affect postoperative myocardial infarction (MI) in low-risk patients undergoing joint arthroplasty?
Background: Recent trials show no benefit of perioperative beta blockers in reducing the incidence of perioperative myocardial infarctions (POMI) in low-risk patients. This retrospective study examined the impact of continuing or discontinuing beta blockers and the occurrence of POMI in patients undergoing elective joint arthroplasties.
Study design: Retrospective chart review.
Setting: Large academic center in Ottawa, Canada.
Synopsis: Medical records for 5,178 patients undergoing elective hip or knee arthroplasty from January 2002 to June 2006 were included in the review. The primary outcome was POMI, defined as an increased troponin level. Patients were divided into three groups: beta blocker prescribed on post-operative day (POD) zero and continued for one week or until discharge; beta blocker prescribed on POD zero and discontinued at any time in the first week; and no beta blocker on POD 0.
Beta blockers were continued in 992 patients and discontinued in 252 patients. The rate of POMI and death increased in the beta-blocker discontinuation group (odds ratio 2.0 [1.1-3.9] and 2.0 [1.1-3.9], respectively). This association persisted after adjustment for cardiac risk using a validated risk score.
The study was limited by the fact that the control group did not include patients who were on a beta blocker at home, thus potentially increasing the number of events in this group. The discontinuation beta blocker group had an increased baseline risk for POMI. The reason for discontinuing the beta blocker was not known, and cessation of beta blocker could have been due to an acute event.
Bottom line: This study adds support to the American College of Cardiology and American Heart Association (ACC/AHA) guidelines, which recommend continuation of beta-blocker therapy in the perioperative period.
Citation: Van Klei WA, Bryson GL, Yang H, Forster AJ. Effect of beta-blocker prescription on the incidence of postoperative myocardial infarction after hip and knee arthroplasty. Anesthesiology. 2009;111(4):717-724.
Lower Perioperative Mortality with Endovascular Vs. Open Abdominal Aortic Aneurysm Repair
Clinical question: How do perioperative and long-term morbidity and mortality compare in endovascular and open repair of abdominal aortic aneurysm (AAA)?
Background: Open AAA repair has relatively high perioperative mortality. Endovascular repair was developed as a less-invasive option and has been shown to reduce inpatient perioperative mortality, length of hospital stay, and ICU requirement. However, data suggest it leads to more frequent reinterventions and the same mortality rate as open repair at two years.
Study design: Randomized clinical trial.
Setting: Veterans Affairs medical centers.
Synopsis: The study randomized 881 veterans who planned to have elective AAA repair and were eligible for both endovascular and open repair. This is a planned, two-year interim report in a nine-year study.
Perioperative mortality was 0.5% in the endovascular repair group, compared with 3.0% in the open repair group. However, this difference in mortality was not statistically significant at two years. The endovascular repair group experienced shorter procedure and mechanical ventilation time, decreased hospital and ICU stay, and lower rate of blood transfusions.
Overall, there was no difference between the groups for major morbidity, procedure failure, need for secondary therapeutic intervention, quality of life, or erectile dysfunction. More data on long-term comparison of these two interventions will be available at the conclusion of this study.
Bottom line: Endovascular repair of AAA has lower perioperative mortality than open repair but did not lead to improved morbidity or mortality at two years.
Citation: Lederle FA, Freischlag JA, Kyriakides TC, et al. Outcomes following endovascular vs. open repair of abdominal aortic aneurysm: a randomized trial. JAMA. 2009;302 (14):1535-1542.
OTC Analgesics Not Associated with Acute Decompensation in Cirrhotic Patients
Clinical question: Do over-the-counter (OTC) analgesics lead to acute hepatic decompensation among patients with cirrhosis?
Background: In theory, intake of acetaminophen and/or nonsteroidal anti-inflammatory drugs (NSAIDs) can worsen hepatic function and lead to complications among cirrhotic patients. The role of OTC analgesics in potentially triggering acute hepatic decompensation among cirrhotic patients has not been studied.
Study design: Prospective case-control study.
Setting: Two tertiary-care hospitals.
Synopsis: Cirrhotic patients hospitalized for acute liver decompensation were compared with compensated cirrhotic patients in the liver clinic (cirrhotic controls) and with randomly selected, noncirrhotic patients who were simultaneously hospitalized (noncirrhotic controls). Data collected through questionnaires included quantity and dose of OTC analgesics used and alcohol consumption in the past 30 days.
Thirty-five percent of the hospitalized cirrhotic patients, 52% of the cirrhotic controls, and 70% of the noncirrhotic controls used OTC analgesics. At doses lower than those recommended, acetaminophen is not associated with acute liver decompensation among cirrhotic patients, even with recent alcohol use. However, NSAIDs taken by the cirrhotic patients, when compared to control subjects, were in larger doses and used for a longer duration, suggesting NSAIDs may have contributed to the acute decompensation.
Study limitations include the nature of the study design, reliance on the patient’s recall of OTC analgesic use, and obtaining other possible causes of decompensation, such as herbal supplement intake or compliance with diuretics or dietary indiscretion.
Bottom line: Acetaminophen at doses lower than recommended is not associated with adverse complications in cirrhotic patients, but NSAIDs are possibly associated with acute decompensation.
Citation: Khalid SK, Lane J, Navarro V, Garcia-Tsao G. Use of over-the-counter analgesics is not associated with acute decompensation in patients with cirrhosis. Clin Gastroenterol Hepatol. 2009;7(9):994-999.
Cardiovascular Disease and Risk of Hip Fracture
Clinical question: Is the diagnosis of cardiovascular disease (CVD) associated with the risk of subsequent hip fracture?
Background: Osteoporosis and CVD are regarded as independent, age-related conditions. However, recent research suggests that the bone and vascular systems share common regulatory mechanisms. Stroke is a known risk factor for hip fractures, and bisphosphonates have been shown to prevent atherosclerosis and reduce total mortality rate.
Study design: Cohort study.
Setting: Swedish National Patient Registry.
Synopsis: The study identified 31,936 Swedish twins born from 1914 to 1944. This cohort was followed up to age 50, and time-dependent exposures using Cox-proportional hazard regression models were evaluated.
Times to hip fracture after CVD diagnosis were isolated. Crude absolute rate of hip fractures (per 1,000 person-years) was 12.6 after diagnosis of heart failure, 12.6 after a stroke, 6.6 after peripheral atherosclerosis, and 5.2 after ischemic heart disease (IHD), compared with 1.2 per 1,000 person-years without a CVD diagnosis. Multivariable-adjusted hazard ratio (HR) of hip fracture after heart failure was 4.40 (95% CI, 3.43-5.63); after a stroke was 5.09 (95% CI, 4.18-6.20); after peripheral atherosclerosis was 3.20 (CI, 2.28-4.50); and after an IHD event was 2.32 (CI, 1.91-2.84).
Identical twins even without heart failure and stroke also had an increased risk of hip fracture if their twin had been diagnosed with these diseases.
Bottom line: Cardiovascular disease is significantly associated with risk of subsequent hip fracture, and genetic factors probably play a role in the association.
Citation: Sennerby U, Melhus H, Gedeborg R, et al. Cardiovascular diseases and risk of hip fracture. JAMA. 2009;302(15):1666-1673. TH
HM Heads to Washington
Hospital Medicine 2010—HM10, in hospitalist parlance—is coming soon. SHM’s annual meeting continues to exceed expectations for educational content, networking opportunities, professional advancement, and fun.
HM10, which runs April 8-11, has an additional attraction this year: With the conference center just miles from Washington, D.C., HM10 will bring hospitalists closer than ever to the heart of the continued national debate over healthcare reform and delivery. Between now and then, the details of healthcare policy will no doubt change, but the intensity and impact of the decisions made in our nation’s capital are unlikely to fade.
“Washington, D.C., is always an exciting place to visit, but it’s even more attractive now for hospitalists and others involved in healthcare,” says Geri Barnes, SHM’s senior director for education and meetings. “The discussions happening in Washington now are likely to affect every corner of the healthcare sector.”
As in years past, HM10 offerings will be wide-ranging enough to include topics that will satisfy physicians, nonphysician practitioners (NPPs), and HM administrators alike.
Registration for HM09 sold out in advance—an additional incentive for early HM10 registration.
“Last year’s conference set new records and generated real excitement within the specialty,” Barnes says. “We’re confident that the program we’ve created for 2010 will do it again.”
Featured Speakers: The Stars of Hospital Care
This year’s featured presenters represent a mix of fresh, outside perspectives and familiar favorites.
Kicking off the formal agenda will be Paul Levy, president and CEO of Beth Israel Deaconess Medical Center, a 621-bed academic hospital center in Boston. In 2009, it was one of three American Hospital Association McKesson Quest for Quality Prize honorees for its efforts in eliminating preventable harm.
Levy says he expects to expound on the patient-safety theme during his HM10 presentation. He’ll speak about his center’s “journey in preventable harm—what we’ve learned, ideas for the future, the role of transparency, and the different approaches to process improvement,” he says. Check out his blog at http://runningahospital.blogspot.com.
To wrap up the conference, HM pioneer Robert Wachter, MD, FHM, chief of the hospital medicine division, professor, and associate chair of the Department of Medicine at the University of California at San Francisco, a former SHM president, and author of the blog Wachter’s World (www.wachters world.com), will bring his perspective on HM and healthcare reform in a presentation called “How Health Care Reform Changes the Hospitalist Field . . . And Vice Versa.”
More Pre-Courses
In response to increased demand for educational content, HM10 will offer the most pre-courses ever. The pre-courses emphasize a hands-on approach to professional development. This year’s eight pre-courses—two more than last year—will run concurrently all day April 8. “Hospitalists are always looking for ways to enhance their knowledge of the specialty and sharpen their skills,” Barnes says. “The new pre-courses at HM10 were added specifically because of demand from hospitalists.”
The two new additions represent the changing needs within HM. The “Essential Neurology for the Hospitalist” pre-course, taught by David Likosky, MD, FHM, a hospitalist at Evergreen Hospital Medical Center in Kirkland, Wash., recognizes hospitalists often serve as the primary health providers for hospitalized patients with neurological disorders. The pre-course will cover the basics of neurological exams, diagnosis, and management of many of the conditions hospitalists encounter on a regular basis.
The second new pre-course addresses some of the daunting challenges that new hospitalists face. “Early Career Hospitalist: Skills for Success,” led by Efren Manjarrez, MD, FHM, of the University of Miami School of Medicine, will lead new hospitalists through such day-to-day issues as communicating with patients and families, coding, quality improvement (QI) efforts, and legal considerations in their practice.
Although the pre-course on the American Board of Internal Medicine’s (ABIM) Maintenance of Certification (MOC) isn’t new, it will include new information about the process for applying to ABIM’s new Recognition of Focused Practice in Hospital Medicine program.
Inaugural Year for Senior Fellows
SHM’s Fellows in Hospital Medicine Program will take center stage again, as the society welcomes the first class of Senior Fellows in Hospital Medicine (SFHM) and the second class of Fellows in Hospital Medicine (FHM). Now in its second year, the fellows program recognizes hospitalists for their commitment to excellence.
At HM09, SHM inducted more than 500 hospitalists as fellows. Candidates must have at least five years’ experience as a hospitalist and demonstrate their work in QI, teamwork, and leadership. This year, SHM expects to induct even more fellows.
The requirements for the SHFM are similar to those of the FHM program but demand more experience in each category. Hospitalists applying for SFHM also must be an SHM member in good standing for at least five years.
New Educational Options: Breakout Sessions and RIV Competition
The HM10 educational program features new breakout sessions and the annual Research, Innovations, and Clinical Vignettes (RIV) competition. The new tracks include:
- Academic;
- Clinical 1;
- Clinical 2;
- Evidence-Based Rapid Fire;
- Palliative Care;
- Pediatric;
- Practice Management;
- Quality;
- Research; and
- Workshops (educational format for the annual conference).
Hundreds of hospitalists will submit abstracts for the RIV competition. Of those, dozens will be chosen for the HM10 poster session; a panel of experts will judge the entries on Saturday, April 10. The winners will be announced at the conference and claim a $250 cash prize.
High-Visibility Exhibits
HM’s growth has spurred a burgeoning industry of products and service providers that help hospitalists do their jobs more effectively and efficiently. HM10 brings the best of the industry directly to hospitalists, and this year, SHM is making it easier than ever for hospitalists to find the experts on the exhibit floor. For the first time, HM10’s agenda includes time to allow attendees to browse the exhibit hall without competing workshops or plenary sessions. Plus, attendees will win prizes for visiting exhibit booths.
“HM10 is all about bringing the leaders in hospital medicine together. That includes the leaders in organizations that support hospital medicine,” says Todd Von Deak, vice president of membership and marketing for SHM. “Just like other parts of HM10, innovation and synergy happen on the exhibition floor.”
—Geri Barnes, SHM senior director of education and meetings
Bring the Family
Washington, D.C., is a prime destination for vacationers from around the world, and SHM has organized tours for families and spouses of hospitalists at HM10. Each tour departs directly from the Gaylord National Hotel & Convention Center and takes participants to some of the most famous attractions in the nation’s capital.
Never been to Washington? Then start with the all-day DC IT ALL! Tour, which takes visitors on a guided bus tour to many major monuments, museums, and other city sights.
For those more familiar with Washington, tours of the National Air & Space Museum, a Segway tour of Old Town Alexandria, Va., and George Washington’s Mount Vernon via water cruise are also scheduled.
For more information, visit the “Family Activities” section of the HM10 Web site. To register for a tour, call SHM at 800-843-3360. TH
Brendon Shank is a freelance writer based in Philadelphia.
Chapter Update
Piedmont Chapter
Hospital Medicine 2010—HM10, in hospitalist parlance—is coming soon. SHM’s annual meeting continues to exceed expectations for educational content, networking opportunities, professional advancement, and fun.
HM10, which runs April 8-11, has an additional attraction this year: With the conference center just miles from Washington, D.C., HM10 will bring hospitalists closer than ever to the heart of the continued national debate over healthcare reform and delivery. Between now and then, the details of healthcare policy will no doubt change, but the intensity and impact of the decisions made in our nation’s capital are unlikely to fade.
“Washington, D.C., is always an exciting place to visit, but it’s even more attractive now for hospitalists and others involved in healthcare,” says Geri Barnes, SHM’s senior director for education and meetings. “The discussions happening in Washington now are likely to affect every corner of the healthcare sector.”
As in years past, HM10 offerings will be wide-ranging enough to include topics that will satisfy physicians, nonphysician practitioners (NPPs), and HM administrators alike.
Registration for HM09 sold out in advance—an additional incentive for early HM10 registration.
“Last year’s conference set new records and generated real excitement within the specialty,” Barnes says. “We’re confident that the program we’ve created for 2010 will do it again.”
Featured Speakers: The Stars of Hospital Care
This year’s featured presenters represent a mix of fresh, outside perspectives and familiar favorites.
Kicking off the formal agenda will be Paul Levy, president and CEO of Beth Israel Deaconess Medical Center, a 621-bed academic hospital center in Boston. In 2009, it was one of three American Hospital Association McKesson Quest for Quality Prize honorees for its efforts in eliminating preventable harm.
Levy says he expects to expound on the patient-safety theme during his HM10 presentation. He’ll speak about his center’s “journey in preventable harm—what we’ve learned, ideas for the future, the role of transparency, and the different approaches to process improvement,” he says. Check out his blog at http://runningahospital.blogspot.com.
To wrap up the conference, HM pioneer Robert Wachter, MD, FHM, chief of the hospital medicine division, professor, and associate chair of the Department of Medicine at the University of California at San Francisco, a former SHM president, and author of the blog Wachter’s World (www.wachters world.com), will bring his perspective on HM and healthcare reform in a presentation called “How Health Care Reform Changes the Hospitalist Field . . . And Vice Versa.”
More Pre-Courses
In response to increased demand for educational content, HM10 will offer the most pre-courses ever. The pre-courses emphasize a hands-on approach to professional development. This year’s eight pre-courses—two more than last year—will run concurrently all day April 8. “Hospitalists are always looking for ways to enhance their knowledge of the specialty and sharpen their skills,” Barnes says. “The new pre-courses at HM10 were added specifically because of demand from hospitalists.”
The two new additions represent the changing needs within HM. The “Essential Neurology for the Hospitalist” pre-course, taught by David Likosky, MD, FHM, a hospitalist at Evergreen Hospital Medical Center in Kirkland, Wash., recognizes hospitalists often serve as the primary health providers for hospitalized patients with neurological disorders. The pre-course will cover the basics of neurological exams, diagnosis, and management of many of the conditions hospitalists encounter on a regular basis.
The second new pre-course addresses some of the daunting challenges that new hospitalists face. “Early Career Hospitalist: Skills for Success,” led by Efren Manjarrez, MD, FHM, of the University of Miami School of Medicine, will lead new hospitalists through such day-to-day issues as communicating with patients and families, coding, quality improvement (QI) efforts, and legal considerations in their practice.
Although the pre-course on the American Board of Internal Medicine’s (ABIM) Maintenance of Certification (MOC) isn’t new, it will include new information about the process for applying to ABIM’s new Recognition of Focused Practice in Hospital Medicine program.
Inaugural Year for Senior Fellows
SHM’s Fellows in Hospital Medicine Program will take center stage again, as the society welcomes the first class of Senior Fellows in Hospital Medicine (SFHM) and the second class of Fellows in Hospital Medicine (FHM). Now in its second year, the fellows program recognizes hospitalists for their commitment to excellence.
At HM09, SHM inducted more than 500 hospitalists as fellows. Candidates must have at least five years’ experience as a hospitalist and demonstrate their work in QI, teamwork, and leadership. This year, SHM expects to induct even more fellows.
The requirements for the SHFM are similar to those of the FHM program but demand more experience in each category. Hospitalists applying for SFHM also must be an SHM member in good standing for at least five years.
New Educational Options: Breakout Sessions and RIV Competition
The HM10 educational program features new breakout sessions and the annual Research, Innovations, and Clinical Vignettes (RIV) competition. The new tracks include:
- Academic;
- Clinical 1;
- Clinical 2;
- Evidence-Based Rapid Fire;
- Palliative Care;
- Pediatric;
- Practice Management;
- Quality;
- Research; and
- Workshops (educational format for the annual conference).
Hundreds of hospitalists will submit abstracts for the RIV competition. Of those, dozens will be chosen for the HM10 poster session; a panel of experts will judge the entries on Saturday, April 10. The winners will be announced at the conference and claim a $250 cash prize.
High-Visibility Exhibits
HM’s growth has spurred a burgeoning industry of products and service providers that help hospitalists do their jobs more effectively and efficiently. HM10 brings the best of the industry directly to hospitalists, and this year, SHM is making it easier than ever for hospitalists to find the experts on the exhibit floor. For the first time, HM10’s agenda includes time to allow attendees to browse the exhibit hall without competing workshops or plenary sessions. Plus, attendees will win prizes for visiting exhibit booths.
“HM10 is all about bringing the leaders in hospital medicine together. That includes the leaders in organizations that support hospital medicine,” says Todd Von Deak, vice president of membership and marketing for SHM. “Just like other parts of HM10, innovation and synergy happen on the exhibition floor.”
—Geri Barnes, SHM senior director of education and meetings
Bring the Family
Washington, D.C., is a prime destination for vacationers from around the world, and SHM has organized tours for families and spouses of hospitalists at HM10. Each tour departs directly from the Gaylord National Hotel & Convention Center and takes participants to some of the most famous attractions in the nation’s capital.
Never been to Washington? Then start with the all-day DC IT ALL! Tour, which takes visitors on a guided bus tour to many major monuments, museums, and other city sights.
For those more familiar with Washington, tours of the National Air & Space Museum, a Segway tour of Old Town Alexandria, Va., and George Washington’s Mount Vernon via water cruise are also scheduled.
For more information, visit the “Family Activities” section of the HM10 Web site. To register for a tour, call SHM at 800-843-3360. TH
Brendon Shank is a freelance writer based in Philadelphia.
Chapter Update
Piedmont Chapter
Hospital Medicine 2010—HM10, in hospitalist parlance—is coming soon. SHM’s annual meeting continues to exceed expectations for educational content, networking opportunities, professional advancement, and fun.
HM10, which runs April 8-11, has an additional attraction this year: With the conference center just miles from Washington, D.C., HM10 will bring hospitalists closer than ever to the heart of the continued national debate over healthcare reform and delivery. Between now and then, the details of healthcare policy will no doubt change, but the intensity and impact of the decisions made in our nation’s capital are unlikely to fade.
“Washington, D.C., is always an exciting place to visit, but it’s even more attractive now for hospitalists and others involved in healthcare,” says Geri Barnes, SHM’s senior director for education and meetings. “The discussions happening in Washington now are likely to affect every corner of the healthcare sector.”
As in years past, HM10 offerings will be wide-ranging enough to include topics that will satisfy physicians, nonphysician practitioners (NPPs), and HM administrators alike.
Registration for HM09 sold out in advance—an additional incentive for early HM10 registration.
“Last year’s conference set new records and generated real excitement within the specialty,” Barnes says. “We’re confident that the program we’ve created for 2010 will do it again.”
Featured Speakers: The Stars of Hospital Care
This year’s featured presenters represent a mix of fresh, outside perspectives and familiar favorites.
Kicking off the formal agenda will be Paul Levy, president and CEO of Beth Israel Deaconess Medical Center, a 621-bed academic hospital center in Boston. In 2009, it was one of three American Hospital Association McKesson Quest for Quality Prize honorees for its efforts in eliminating preventable harm.
Levy says he expects to expound on the patient-safety theme during his HM10 presentation. He’ll speak about his center’s “journey in preventable harm—what we’ve learned, ideas for the future, the role of transparency, and the different approaches to process improvement,” he says. Check out his blog at http://runningahospital.blogspot.com.
To wrap up the conference, HM pioneer Robert Wachter, MD, FHM, chief of the hospital medicine division, professor, and associate chair of the Department of Medicine at the University of California at San Francisco, a former SHM president, and author of the blog Wachter’s World (www.wachters world.com), will bring his perspective on HM and healthcare reform in a presentation called “How Health Care Reform Changes the Hospitalist Field . . . And Vice Versa.”
More Pre-Courses
In response to increased demand for educational content, HM10 will offer the most pre-courses ever. The pre-courses emphasize a hands-on approach to professional development. This year’s eight pre-courses—two more than last year—will run concurrently all day April 8. “Hospitalists are always looking for ways to enhance their knowledge of the specialty and sharpen their skills,” Barnes says. “The new pre-courses at HM10 were added specifically because of demand from hospitalists.”
The two new additions represent the changing needs within HM. The “Essential Neurology for the Hospitalist” pre-course, taught by David Likosky, MD, FHM, a hospitalist at Evergreen Hospital Medical Center in Kirkland, Wash., recognizes hospitalists often serve as the primary health providers for hospitalized patients with neurological disorders. The pre-course will cover the basics of neurological exams, diagnosis, and management of many of the conditions hospitalists encounter on a regular basis.
The second new pre-course addresses some of the daunting challenges that new hospitalists face. “Early Career Hospitalist: Skills for Success,” led by Efren Manjarrez, MD, FHM, of the University of Miami School of Medicine, will lead new hospitalists through such day-to-day issues as communicating with patients and families, coding, quality improvement (QI) efforts, and legal considerations in their practice.
Although the pre-course on the American Board of Internal Medicine’s (ABIM) Maintenance of Certification (MOC) isn’t new, it will include new information about the process for applying to ABIM’s new Recognition of Focused Practice in Hospital Medicine program.
Inaugural Year for Senior Fellows
SHM’s Fellows in Hospital Medicine Program will take center stage again, as the society welcomes the first class of Senior Fellows in Hospital Medicine (SFHM) and the second class of Fellows in Hospital Medicine (FHM). Now in its second year, the fellows program recognizes hospitalists for their commitment to excellence.
At HM09, SHM inducted more than 500 hospitalists as fellows. Candidates must have at least five years’ experience as a hospitalist and demonstrate their work in QI, teamwork, and leadership. This year, SHM expects to induct even more fellows.
The requirements for the SHFM are similar to those of the FHM program but demand more experience in each category. Hospitalists applying for SFHM also must be an SHM member in good standing for at least five years.
New Educational Options: Breakout Sessions and RIV Competition
The HM10 educational program features new breakout sessions and the annual Research, Innovations, and Clinical Vignettes (RIV) competition. The new tracks include:
- Academic;
- Clinical 1;
- Clinical 2;
- Evidence-Based Rapid Fire;
- Palliative Care;
- Pediatric;
- Practice Management;
- Quality;
- Research; and
- Workshops (educational format for the annual conference).
Hundreds of hospitalists will submit abstracts for the RIV competition. Of those, dozens will be chosen for the HM10 poster session; a panel of experts will judge the entries on Saturday, April 10. The winners will be announced at the conference and claim a $250 cash prize.
High-Visibility Exhibits
HM’s growth has spurred a burgeoning industry of products and service providers that help hospitalists do their jobs more effectively and efficiently. HM10 brings the best of the industry directly to hospitalists, and this year, SHM is making it easier than ever for hospitalists to find the experts on the exhibit floor. For the first time, HM10’s agenda includes time to allow attendees to browse the exhibit hall without competing workshops or plenary sessions. Plus, attendees will win prizes for visiting exhibit booths.
“HM10 is all about bringing the leaders in hospital medicine together. That includes the leaders in organizations that support hospital medicine,” says Todd Von Deak, vice president of membership and marketing for SHM. “Just like other parts of HM10, innovation and synergy happen on the exhibition floor.”
—Geri Barnes, SHM senior director of education and meetings
Bring the Family
Washington, D.C., is a prime destination for vacationers from around the world, and SHM has organized tours for families and spouses of hospitalists at HM10. Each tour departs directly from the Gaylord National Hotel & Convention Center and takes participants to some of the most famous attractions in the nation’s capital.
Never been to Washington? Then start with the all-day DC IT ALL! Tour, which takes visitors on a guided bus tour to many major monuments, museums, and other city sights.
For those more familiar with Washington, tours of the National Air & Space Museum, a Segway tour of Old Town Alexandria, Va., and George Washington’s Mount Vernon via water cruise are also scheduled.
For more information, visit the “Family Activities” section of the HM10 Web site. To register for a tour, call SHM at 800-843-3360. TH
Brendon Shank is a freelance writer based in Philadelphia.
Chapter Update
Piedmont Chapter
An Officer and a Physician
Imagine being transferred to a hospital where the temperature is 20 below outside, and 40 inches of snow fill the parking lot. Few physicians would sign on for such an assignment. For a brave few, it’s all in a day’s work.
Maj. Ramey Wilson, MD, is a U.S. Army physician who encountered such conditions during his 15-month experience in Afghanistan. “A couple of times, when we couldn’t get helicopters [for evacuation], we had to turn my aid station into a mini-hospital. There were no nurses, labs, or X-ray,” he says. “With only basic supplies and my combat medics, we had to provide all the patient care until the weather broke.”
Not quite the circumstances most hospitalists encounter in their daily practice.
Hospitalists in the military face daunting odds, and at the same time are blessed with some unexpected advantages. On the plus side, military physicians cite the camaraderie, teamwork, honor of caring for soldiers, and the opportunity to train other providers, both in traditional, U.S.-based residencies and while deployed. Among the minuses, they mention a lack of equipment and supplies when they are assigned to forward-deployed soldiers on foreign soil, the heartache of being separated from family, and lower compensation. Most military physicians, however, say that the lower compensation can be offset by generous government benefits and the absence of medical school debt.
All in all, hospitalists in the military have a unique—and sometimes adventurous—story to tell.
Challenges Met, Success Exemplified
Dr. Wilson is a hospitalist and Army physician assigned to Fort Bragg, N.C. Until this past summer, he was the chief of internal medicine at Womack Army Medical Center, one of eight full-service hospitals in the U.S. Army Medical Command. Because the Army is still familiarizing itself with the HM model and the role hospitalists play in the delivery of healthcare, resident house staff meet many of the operational needs, including night and weekend coverage. “The Army doesn’t have a good system for 24-hour continuous care at busy hospitals without residents,” Dr. Wilson says, “and we’ve worked hard to get hospitalists into our system.”
While other Army medical centers have internal-medicine residencies, Womack has only a family medicine residency program. Residents once provided extensive coverage for the hospital, but decreasing numbers (only four interns this year) and work-hour restrictions have shifted the inpatient responsibilities to the internal-medicine staff. “All of the military general internists have functionally become hospitalists to support the inpatient medicine and ICU services,” Dr. Wilson says. “Our family medicine house staff coverage has evaporated.”
The conditions he sees at Womack are similar to what he sees at FirstHealth Moore Regional Hospital, the civilian community hospital in Pinehurst, N.C., where he practices part time. Womack serves two major military populations: those on active duty and their family members, and those no longer on active duty or retired (and not a part of the Veterans Administration program).

—Col. Walt Franz, MD, U.S. Army Medical Corps, Amarah/Al Kut, Iraq
Dr. Wilson, who served in the Ghazni province in eastern Afghanistan, was the only American physician in an area of 8,800 square miles. He and his physician-assistant staff were tasked with keeping U.S. soldiers healthy, serving acute resuscitative trauma care and “basic sick call.” In addition to caring for U.S. and coalition soldiers, he partnered with the Ghazni Ministry of Health to improve the delivery of healthcare to residents of the province.
“Afghanistan has a great plan for medical care through its ‘basic’ package of health services and ‘essential’ package hospital services, developed with assistance from the U.S. Agency for International Development [USAID], and which we used as our road map for the Afghan public health service,” Dr. Wilson says. He and a nurse practitioner from the nearby provincial reconstruction team worked out of a forward operating base outside Ghazni’s provincial capital—the city of Ghazni—and the nearby provincial hospital. He says his hospitalist background was helpful when it came to working with and teaching the Afghan physicians and nurses at the hospital, which served as the referral center for several surrounding provinces.
“There was no infection-control program; their hospital and clinics were heated by wood stoves; and they were using the one endotracheal tube that had been left by the International Red Cross years earlier,” he says, noting that during his tour, the U.S. military dropped basic medical equipment and supplies—which were shared with the local hospital—into his forward operating base. “They were doing anesthesia without monitors. We trained them with an initial focus of making surgery safer. … To say that it was challenging is an understatement, and for many different reasons.”
Almost every other week, Dr. Wilson hosted a medical conference at his base for 10 to 20 Afghan physicians. Due to local customs, female providers were not allowed to travel without a male relative, so Dr. Wilson’s team videotaped the classes, had them translated into the Pashto language, and arranged with the hospital directors to distribute them to female physicians.
The needs in both army and civilian circumstances are huge in Afghanistan. Most Afghan hospitals and clinics are without Internet access, so decision support and telemedicine consultative service is out of the question. Textbooks are in short supply, too. Because the Taliban decreed that no image of a human being is allowed in print, they confiscated and destroyed all of the country’s anatomy books.
In terms of training, the key to success with locals is demonstrating what success can look like.
“Most of these providers have practiced in a system that they think is as good as it can be given the lack of advanced machinery and equipment,” Dr. Wilson says.
Physicians who visit U.S. military or Western hospitals and witness the successes possible in infection control, nursing care, medication administration, and medical documentation return to Afghanistan excited about the skills introduced to them. “They see that the provision of really good medical care is more dependent on having a clean space, a well-organized system, good communication, and solid basic medical care,” Dr. Wilson says.
Contrast to Care Continuity
Col. Walt Franz, MD, of U.S. Army Medical Corps headquartered in Amarah/Al Kut, Iraq, has just begun the work of partnering with Iraqi physicians and nurses for the first time since 2003. In 2004, as a public health team leader, his primary task was helping Iraqi providers with hospital and clinic projects. The projects ranged in cost from $40,000 (for securing an X-ray machine) to $5,000 for such smaller repairs and fix-ups as securing parts to make an elevator run. In fact, patients were being carried up several flights of stairs in the local, six-story hospital.
For about five months in 2008, Dr. Franz was deputy commander for clinical services for hospital and outpatient medical care at a combat support hospital. Since the beginning of 2009, he has been the commander of the 945th Forward Surgical Team at a small forward base in Amarah, near the Iraq-Iran border. “Our mission here is to provide urgent surgical resuscitation for the critically wounded and evac[uation] by helo [helicopter],” Dr. Franz says.
When he’s at home and working at the Mayo Clinic in Rochester, Minn., he practices primarily as a family physician. With nearly 30 years of clinical practice under his belt, Dr. Franz also puts in plenty of hours as a hospitalist. He has practiced during four deployments: three to Iraq and one to Germany.
“Active duty in a war zone presents experiences ranging from the inspiring to the absolutely tragic,” Dr. Franz says. “There is nothing worse than a casualty coming in on a medevac. It’s someone’s son or daughter or husband or wife, and nothing approaches the joy of helping a soldier. In fact, as a civilian, we scrupulously follow the Geneva Convention requirements.” (The treaty affords wounded and sick soldiers to be cared for and protected even though they may become prisoners of war.)
After you eliminate the dangers of enemy fire, there are still big differences between combat versus civilian medicine, he says. One is that combat medicine is usually acute care with little or no followup in the theater of operation, Dr. Franz says. Combat medicine has a strong foundation in echelons of care and evacuations away from the initial point of care. It runs concurrent to the civilian premise of continuity, and the limited number of specialists in theater usually means the Army relies on evacuation or electronic consults.
Maysan Province, where Dr. Franz is stationed, is the poorest part of Iraq. Because of its large Shia population, its citizens were devastated during the Iran-Iraq war and brutalized by Saddam Hussein. “The docs here are very street-smart; their work ethic is great and they have done without for a long time,” Dr. Franz says. Providers at the 540-bed hospital in Al Amarah see 200 patients per day in the ED; several hundred outpatients are triaged, and senior staff physicians see 75 or more cases daily. “One young doc told me it was not unusual to have 500 patients present to a regional ED in a 24-hour period, making triage and care almost overwhelming,” he says.
The biggest problem Dr. Franz witnesses in Iraqi hospitals is the lack of specialty nurses. His teams are teaching classes and training trainers in ED triage, basic ICU care, and the ultrasound FAST (Focused Assessment with Sonography in Trauma) exam skills Iraqi providers can use anywhere in the hospital.
Other issues include a lack of continuing medical education; poor infrastructure, which chokes the supply of pharmaceuticals and other medical equipment; and paucity of specialty nursing. Dr. Franz also cites critical staffing issues, such as the large number of physicians who have fled the country and the rising prominence of the private, fee-for-service care system, which can attract physicians and nurses away from the public system.
Care for Female Soldiers
With three other OB hospitalists, also known as laborists, Brook Thomson, MD, spent the summer organizing an OB/GYN hospital medicine program at Saint Alphonsus Regional Medical Center in Boise, Idaho. A veteran of military medicine, Dr. Thomson trained at Uniformed Services University of Health Sciences (USUHS) and completed an OB/GYN residency in 1997, then was stationed in Germany for four years. From 2001 to 2004, he served as chief of obstetrics at Madigan Army Medical Center in Tacoma, Wash., during which time he was deployed to Iraq for 10 months.
The OB/GYN expertise combined with the HM practice model that Dr. Thomson offers is a growing need in the military. “The number of women in the military is increasing, and there just aren’t a lot of people who understand female soldiers’ special needs,” he says.
Supporting women’s health has become an important aspect of battlefield medicine, namely the rooting out of potential sexual abuse. Dr. Thomson has published on the subject.1
In 2003, he was deployed as a general medical officer in Kuwait and assigned to the Basra area of Iraq, treating the gamut of patient needs. Recent Army policy changes, he says, ensure that OB/GYN military physicians now practice within their specialty.
A Canadian Perspective
Brendan James Hughes, MD, CCFP, returned from his military tour of duty and became a family practitioner in Lakefield, Ontario, a small community about 100 miles north of Toronto, and medical director of first-aid services for the Ontario Zone of the Canadian Red Cross.
In 2001, when Dr. Hughes was deployed as a hospitalist to Bosnia-Herzegovina for six months, the unrest from the civil war that involved Bosnians, Croatians, and Serbs (more than 100,000 were killed, and millions were injured or displaced), had settled, and his unit returned home without any loss of life. Upon his return, he transitioned from military life to become a full-time civilian hospitalist for six years in Ontario and Alberta. He now works as a part-time hospitalist.
Dr. Hughes says Canadian military practice is more acute and trauma-based now, as compared to his 2001 deployment in Eastern Europe. He notices many more deaths and major trauma cases in reports from Afghanistan, mostly blast injuries, limb amputations, and acute brain injuries, than there would be in a traditional, nonmilitary HM practice. He also notes that a lot of time and effort was placed on rehabilitation-focused practice that the patients required in the recovery phase.
Military practice differs from civilian hospitalist practice in other ways, he says. “In the military, every patient is essentially a workplace patient where the military is the employer,” Dr. Hughes says. Although clinicians maintain patient confidentiality, they are obliged to the chain of command to provide information on patient abilities. “We are careful not to relay a specific diagnosis without patient consent, but we have to dictate any needed restrictions on duty that are important in a combat situation, for themselves and for others,” he adds.
Such privacy and disclosure concerns are particularly difficult to navigate when it comes to diagnosis and treatment of alcohol and drug abuse, depression, post-traumatic stress, and suicide risk—issues that can lead soldiers to develop such long-term problems as substance abuse, marital discord, and marital abuse. TH
Andrea Sattinger is a freelance writer based in North Carolina.
Reference
- Thomson B, Nielsen P. Women’s healthcare in Operation Iraqi Freedom: a survey of camps with echelon one or two facilities. Mil Med. 2006;171:216-219.
Dr. Wilson’s photos from Afghanistan
Click images to enlarge

|
|

|

|

|

|

|

|
|

|

|

|

|

|

|
|

|

|

|

|

|

|

|
|

|

|

|

|

|
PHOTOS COURTESY OF MAJ. RAMEY WILSON
Imagine being transferred to a hospital where the temperature is 20 below outside, and 40 inches of snow fill the parking lot. Few physicians would sign on for such an assignment. For a brave few, it’s all in a day’s work.
Maj. Ramey Wilson, MD, is a U.S. Army physician who encountered such conditions during his 15-month experience in Afghanistan. “A couple of times, when we couldn’t get helicopters [for evacuation], we had to turn my aid station into a mini-hospital. There were no nurses, labs, or X-ray,” he says. “With only basic supplies and my combat medics, we had to provide all the patient care until the weather broke.”
Not quite the circumstances most hospitalists encounter in their daily practice.
Hospitalists in the military face daunting odds, and at the same time are blessed with some unexpected advantages. On the plus side, military physicians cite the camaraderie, teamwork, honor of caring for soldiers, and the opportunity to train other providers, both in traditional, U.S.-based residencies and while deployed. Among the minuses, they mention a lack of equipment and supplies when they are assigned to forward-deployed soldiers on foreign soil, the heartache of being separated from family, and lower compensation. Most military physicians, however, say that the lower compensation can be offset by generous government benefits and the absence of medical school debt.
All in all, hospitalists in the military have a unique—and sometimes adventurous—story to tell.
Challenges Met, Success Exemplified
Dr. Wilson is a hospitalist and Army physician assigned to Fort Bragg, N.C. Until this past summer, he was the chief of internal medicine at Womack Army Medical Center, one of eight full-service hospitals in the U.S. Army Medical Command. Because the Army is still familiarizing itself with the HM model and the role hospitalists play in the delivery of healthcare, resident house staff meet many of the operational needs, including night and weekend coverage. “The Army doesn’t have a good system for 24-hour continuous care at busy hospitals without residents,” Dr. Wilson says, “and we’ve worked hard to get hospitalists into our system.”
While other Army medical centers have internal-medicine residencies, Womack has only a family medicine residency program. Residents once provided extensive coverage for the hospital, but decreasing numbers (only four interns this year) and work-hour restrictions have shifted the inpatient responsibilities to the internal-medicine staff. “All of the military general internists have functionally become hospitalists to support the inpatient medicine and ICU services,” Dr. Wilson says. “Our family medicine house staff coverage has evaporated.”
The conditions he sees at Womack are similar to what he sees at FirstHealth Moore Regional Hospital, the civilian community hospital in Pinehurst, N.C., where he practices part time. Womack serves two major military populations: those on active duty and their family members, and those no longer on active duty or retired (and not a part of the Veterans Administration program).

—Col. Walt Franz, MD, U.S. Army Medical Corps, Amarah/Al Kut, Iraq
Dr. Wilson, who served in the Ghazni province in eastern Afghanistan, was the only American physician in an area of 8,800 square miles. He and his physician-assistant staff were tasked with keeping U.S. soldiers healthy, serving acute resuscitative trauma care and “basic sick call.” In addition to caring for U.S. and coalition soldiers, he partnered with the Ghazni Ministry of Health to improve the delivery of healthcare to residents of the province.
“Afghanistan has a great plan for medical care through its ‘basic’ package of health services and ‘essential’ package hospital services, developed with assistance from the U.S. Agency for International Development [USAID], and which we used as our road map for the Afghan public health service,” Dr. Wilson says. He and a nurse practitioner from the nearby provincial reconstruction team worked out of a forward operating base outside Ghazni’s provincial capital—the city of Ghazni—and the nearby provincial hospital. He says his hospitalist background was helpful when it came to working with and teaching the Afghan physicians and nurses at the hospital, which served as the referral center for several surrounding provinces.
“There was no infection-control program; their hospital and clinics were heated by wood stoves; and they were using the one endotracheal tube that had been left by the International Red Cross years earlier,” he says, noting that during his tour, the U.S. military dropped basic medical equipment and supplies—which were shared with the local hospital—into his forward operating base. “They were doing anesthesia without monitors. We trained them with an initial focus of making surgery safer. … To say that it was challenging is an understatement, and for many different reasons.”
Almost every other week, Dr. Wilson hosted a medical conference at his base for 10 to 20 Afghan physicians. Due to local customs, female providers were not allowed to travel without a male relative, so Dr. Wilson’s team videotaped the classes, had them translated into the Pashto language, and arranged with the hospital directors to distribute them to female physicians.
The needs in both army and civilian circumstances are huge in Afghanistan. Most Afghan hospitals and clinics are without Internet access, so decision support and telemedicine consultative service is out of the question. Textbooks are in short supply, too. Because the Taliban decreed that no image of a human being is allowed in print, they confiscated and destroyed all of the country’s anatomy books.
In terms of training, the key to success with locals is demonstrating what success can look like.
“Most of these providers have practiced in a system that they think is as good as it can be given the lack of advanced machinery and equipment,” Dr. Wilson says.
Physicians who visit U.S. military or Western hospitals and witness the successes possible in infection control, nursing care, medication administration, and medical documentation return to Afghanistan excited about the skills introduced to them. “They see that the provision of really good medical care is more dependent on having a clean space, a well-organized system, good communication, and solid basic medical care,” Dr. Wilson says.
Contrast to Care Continuity
Col. Walt Franz, MD, of U.S. Army Medical Corps headquartered in Amarah/Al Kut, Iraq, has just begun the work of partnering with Iraqi physicians and nurses for the first time since 2003. In 2004, as a public health team leader, his primary task was helping Iraqi providers with hospital and clinic projects. The projects ranged in cost from $40,000 (for securing an X-ray machine) to $5,000 for such smaller repairs and fix-ups as securing parts to make an elevator run. In fact, patients were being carried up several flights of stairs in the local, six-story hospital.
For about five months in 2008, Dr. Franz was deputy commander for clinical services for hospital and outpatient medical care at a combat support hospital. Since the beginning of 2009, he has been the commander of the 945th Forward Surgical Team at a small forward base in Amarah, near the Iraq-Iran border. “Our mission here is to provide urgent surgical resuscitation for the critically wounded and evac[uation] by helo [helicopter],” Dr. Franz says.
When he’s at home and working at the Mayo Clinic in Rochester, Minn., he practices primarily as a family physician. With nearly 30 years of clinical practice under his belt, Dr. Franz also puts in plenty of hours as a hospitalist. He has practiced during four deployments: three to Iraq and one to Germany.
“Active duty in a war zone presents experiences ranging from the inspiring to the absolutely tragic,” Dr. Franz says. “There is nothing worse than a casualty coming in on a medevac. It’s someone’s son or daughter or husband or wife, and nothing approaches the joy of helping a soldier. In fact, as a civilian, we scrupulously follow the Geneva Convention requirements.” (The treaty affords wounded and sick soldiers to be cared for and protected even though they may become prisoners of war.)
After you eliminate the dangers of enemy fire, there are still big differences between combat versus civilian medicine, he says. One is that combat medicine is usually acute care with little or no followup in the theater of operation, Dr. Franz says. Combat medicine has a strong foundation in echelons of care and evacuations away from the initial point of care. It runs concurrent to the civilian premise of continuity, and the limited number of specialists in theater usually means the Army relies on evacuation or electronic consults.
Maysan Province, where Dr. Franz is stationed, is the poorest part of Iraq. Because of its large Shia population, its citizens were devastated during the Iran-Iraq war and brutalized by Saddam Hussein. “The docs here are very street-smart; their work ethic is great and they have done without for a long time,” Dr. Franz says. Providers at the 540-bed hospital in Al Amarah see 200 patients per day in the ED; several hundred outpatients are triaged, and senior staff physicians see 75 or more cases daily. “One young doc told me it was not unusual to have 500 patients present to a regional ED in a 24-hour period, making triage and care almost overwhelming,” he says.
The biggest problem Dr. Franz witnesses in Iraqi hospitals is the lack of specialty nurses. His teams are teaching classes and training trainers in ED triage, basic ICU care, and the ultrasound FAST (Focused Assessment with Sonography in Trauma) exam skills Iraqi providers can use anywhere in the hospital.
Other issues include a lack of continuing medical education; poor infrastructure, which chokes the supply of pharmaceuticals and other medical equipment; and paucity of specialty nursing. Dr. Franz also cites critical staffing issues, such as the large number of physicians who have fled the country and the rising prominence of the private, fee-for-service care system, which can attract physicians and nurses away from the public system.
Care for Female Soldiers
With three other OB hospitalists, also known as laborists, Brook Thomson, MD, spent the summer organizing an OB/GYN hospital medicine program at Saint Alphonsus Regional Medical Center in Boise, Idaho. A veteran of military medicine, Dr. Thomson trained at Uniformed Services University of Health Sciences (USUHS) and completed an OB/GYN residency in 1997, then was stationed in Germany for four years. From 2001 to 2004, he served as chief of obstetrics at Madigan Army Medical Center in Tacoma, Wash., during which time he was deployed to Iraq for 10 months.
The OB/GYN expertise combined with the HM practice model that Dr. Thomson offers is a growing need in the military. “The number of women in the military is increasing, and there just aren’t a lot of people who understand female soldiers’ special needs,” he says.
Supporting women’s health has become an important aspect of battlefield medicine, namely the rooting out of potential sexual abuse. Dr. Thomson has published on the subject.1
In 2003, he was deployed as a general medical officer in Kuwait and assigned to the Basra area of Iraq, treating the gamut of patient needs. Recent Army policy changes, he says, ensure that OB/GYN military physicians now practice within their specialty.
A Canadian Perspective
Brendan James Hughes, MD, CCFP, returned from his military tour of duty and became a family practitioner in Lakefield, Ontario, a small community about 100 miles north of Toronto, and medical director of first-aid services for the Ontario Zone of the Canadian Red Cross.
In 2001, when Dr. Hughes was deployed as a hospitalist to Bosnia-Herzegovina for six months, the unrest from the civil war that involved Bosnians, Croatians, and Serbs (more than 100,000 were killed, and millions were injured or displaced), had settled, and his unit returned home without any loss of life. Upon his return, he transitioned from military life to become a full-time civilian hospitalist for six years in Ontario and Alberta. He now works as a part-time hospitalist.
Dr. Hughes says Canadian military practice is more acute and trauma-based now, as compared to his 2001 deployment in Eastern Europe. He notices many more deaths and major trauma cases in reports from Afghanistan, mostly blast injuries, limb amputations, and acute brain injuries, than there would be in a traditional, nonmilitary HM practice. He also notes that a lot of time and effort was placed on rehabilitation-focused practice that the patients required in the recovery phase.
Military practice differs from civilian hospitalist practice in other ways, he says. “In the military, every patient is essentially a workplace patient where the military is the employer,” Dr. Hughes says. Although clinicians maintain patient confidentiality, they are obliged to the chain of command to provide information on patient abilities. “We are careful not to relay a specific diagnosis without patient consent, but we have to dictate any needed restrictions on duty that are important in a combat situation, for themselves and for others,” he adds.
Such privacy and disclosure concerns are particularly difficult to navigate when it comes to diagnosis and treatment of alcohol and drug abuse, depression, post-traumatic stress, and suicide risk—issues that can lead soldiers to develop such long-term problems as substance abuse, marital discord, and marital abuse. TH
Andrea Sattinger is a freelance writer based in North Carolina.
Reference
- Thomson B, Nielsen P. Women’s healthcare in Operation Iraqi Freedom: a survey of camps with echelon one or two facilities. Mil Med. 2006;171:216-219.
Dr. Wilson’s photos from Afghanistan
Click images to enlarge

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|
PHOTOS COURTESY OF MAJ. RAMEY WILSON
Imagine being transferred to a hospital where the temperature is 20 below outside, and 40 inches of snow fill the parking lot. Few physicians would sign on for such an assignment. For a brave few, it’s all in a day’s work.
Maj. Ramey Wilson, MD, is a U.S. Army physician who encountered such conditions during his 15-month experience in Afghanistan. “A couple of times, when we couldn’t get helicopters [for evacuation], we had to turn my aid station into a mini-hospital. There were no nurses, labs, or X-ray,” he says. “With only basic supplies and my combat medics, we had to provide all the patient care until the weather broke.”
Not quite the circumstances most hospitalists encounter in their daily practice.
Hospitalists in the military face daunting odds, and at the same time are blessed with some unexpected advantages. On the plus side, military physicians cite the camaraderie, teamwork, honor of caring for soldiers, and the opportunity to train other providers, both in traditional, U.S.-based residencies and while deployed. Among the minuses, they mention a lack of equipment and supplies when they are assigned to forward-deployed soldiers on foreign soil, the heartache of being separated from family, and lower compensation. Most military physicians, however, say that the lower compensation can be offset by generous government benefits and the absence of medical school debt.
All in all, hospitalists in the military have a unique—and sometimes adventurous—story to tell.
Challenges Met, Success Exemplified
Dr. Wilson is a hospitalist and Army physician assigned to Fort Bragg, N.C. Until this past summer, he was the chief of internal medicine at Womack Army Medical Center, one of eight full-service hospitals in the U.S. Army Medical Command. Because the Army is still familiarizing itself with the HM model and the role hospitalists play in the delivery of healthcare, resident house staff meet many of the operational needs, including night and weekend coverage. “The Army doesn’t have a good system for 24-hour continuous care at busy hospitals without residents,” Dr. Wilson says, “and we’ve worked hard to get hospitalists into our system.”
While other Army medical centers have internal-medicine residencies, Womack has only a family medicine residency program. Residents once provided extensive coverage for the hospital, but decreasing numbers (only four interns this year) and work-hour restrictions have shifted the inpatient responsibilities to the internal-medicine staff. “All of the military general internists have functionally become hospitalists to support the inpatient medicine and ICU services,” Dr. Wilson says. “Our family medicine house staff coverage has evaporated.”
The conditions he sees at Womack are similar to what he sees at FirstHealth Moore Regional Hospital, the civilian community hospital in Pinehurst, N.C., where he practices part time. Womack serves two major military populations: those on active duty and their family members, and those no longer on active duty or retired (and not a part of the Veterans Administration program).

—Col. Walt Franz, MD, U.S. Army Medical Corps, Amarah/Al Kut, Iraq
Dr. Wilson, who served in the Ghazni province in eastern Afghanistan, was the only American physician in an area of 8,800 square miles. He and his physician-assistant staff were tasked with keeping U.S. soldiers healthy, serving acute resuscitative trauma care and “basic sick call.” In addition to caring for U.S. and coalition soldiers, he partnered with the Ghazni Ministry of Health to improve the delivery of healthcare to residents of the province.
“Afghanistan has a great plan for medical care through its ‘basic’ package of health services and ‘essential’ package hospital services, developed with assistance from the U.S. Agency for International Development [USAID], and which we used as our road map for the Afghan public health service,” Dr. Wilson says. He and a nurse practitioner from the nearby provincial reconstruction team worked out of a forward operating base outside Ghazni’s provincial capital—the city of Ghazni—and the nearby provincial hospital. He says his hospitalist background was helpful when it came to working with and teaching the Afghan physicians and nurses at the hospital, which served as the referral center for several surrounding provinces.
“There was no infection-control program; their hospital and clinics were heated by wood stoves; and they were using the one endotracheal tube that had been left by the International Red Cross years earlier,” he says, noting that during his tour, the U.S. military dropped basic medical equipment and supplies—which were shared with the local hospital—into his forward operating base. “They were doing anesthesia without monitors. We trained them with an initial focus of making surgery safer. … To say that it was challenging is an understatement, and for many different reasons.”
Almost every other week, Dr. Wilson hosted a medical conference at his base for 10 to 20 Afghan physicians. Due to local customs, female providers were not allowed to travel without a male relative, so Dr. Wilson’s team videotaped the classes, had them translated into the Pashto language, and arranged with the hospital directors to distribute them to female physicians.
The needs in both army and civilian circumstances are huge in Afghanistan. Most Afghan hospitals and clinics are without Internet access, so decision support and telemedicine consultative service is out of the question. Textbooks are in short supply, too. Because the Taliban decreed that no image of a human being is allowed in print, they confiscated and destroyed all of the country’s anatomy books.
In terms of training, the key to success with locals is demonstrating what success can look like.
“Most of these providers have practiced in a system that they think is as good as it can be given the lack of advanced machinery and equipment,” Dr. Wilson says.
Physicians who visit U.S. military or Western hospitals and witness the successes possible in infection control, nursing care, medication administration, and medical documentation return to Afghanistan excited about the skills introduced to them. “They see that the provision of really good medical care is more dependent on having a clean space, a well-organized system, good communication, and solid basic medical care,” Dr. Wilson says.
Contrast to Care Continuity
Col. Walt Franz, MD, of U.S. Army Medical Corps headquartered in Amarah/Al Kut, Iraq, has just begun the work of partnering with Iraqi physicians and nurses for the first time since 2003. In 2004, as a public health team leader, his primary task was helping Iraqi providers with hospital and clinic projects. The projects ranged in cost from $40,000 (for securing an X-ray machine) to $5,000 for such smaller repairs and fix-ups as securing parts to make an elevator run. In fact, patients were being carried up several flights of stairs in the local, six-story hospital.
For about five months in 2008, Dr. Franz was deputy commander for clinical services for hospital and outpatient medical care at a combat support hospital. Since the beginning of 2009, he has been the commander of the 945th Forward Surgical Team at a small forward base in Amarah, near the Iraq-Iran border. “Our mission here is to provide urgent surgical resuscitation for the critically wounded and evac[uation] by helo [helicopter],” Dr. Franz says.
When he’s at home and working at the Mayo Clinic in Rochester, Minn., he practices primarily as a family physician. With nearly 30 years of clinical practice under his belt, Dr. Franz also puts in plenty of hours as a hospitalist. He has practiced during four deployments: three to Iraq and one to Germany.
“Active duty in a war zone presents experiences ranging from the inspiring to the absolutely tragic,” Dr. Franz says. “There is nothing worse than a casualty coming in on a medevac. It’s someone’s son or daughter or husband or wife, and nothing approaches the joy of helping a soldier. In fact, as a civilian, we scrupulously follow the Geneva Convention requirements.” (The treaty affords wounded and sick soldiers to be cared for and protected even though they may become prisoners of war.)
After you eliminate the dangers of enemy fire, there are still big differences between combat versus civilian medicine, he says. One is that combat medicine is usually acute care with little or no followup in the theater of operation, Dr. Franz says. Combat medicine has a strong foundation in echelons of care and evacuations away from the initial point of care. It runs concurrent to the civilian premise of continuity, and the limited number of specialists in theater usually means the Army relies on evacuation or electronic consults.
Maysan Province, where Dr. Franz is stationed, is the poorest part of Iraq. Because of its large Shia population, its citizens were devastated during the Iran-Iraq war and brutalized by Saddam Hussein. “The docs here are very street-smart; their work ethic is great and they have done without for a long time,” Dr. Franz says. Providers at the 540-bed hospital in Al Amarah see 200 patients per day in the ED; several hundred outpatients are triaged, and senior staff physicians see 75 or more cases daily. “One young doc told me it was not unusual to have 500 patients present to a regional ED in a 24-hour period, making triage and care almost overwhelming,” he says.
The biggest problem Dr. Franz witnesses in Iraqi hospitals is the lack of specialty nurses. His teams are teaching classes and training trainers in ED triage, basic ICU care, and the ultrasound FAST (Focused Assessment with Sonography in Trauma) exam skills Iraqi providers can use anywhere in the hospital.
Other issues include a lack of continuing medical education; poor infrastructure, which chokes the supply of pharmaceuticals and other medical equipment; and paucity of specialty nursing. Dr. Franz also cites critical staffing issues, such as the large number of physicians who have fled the country and the rising prominence of the private, fee-for-service care system, which can attract physicians and nurses away from the public system.
Care for Female Soldiers
With three other OB hospitalists, also known as laborists, Brook Thomson, MD, spent the summer organizing an OB/GYN hospital medicine program at Saint Alphonsus Regional Medical Center in Boise, Idaho. A veteran of military medicine, Dr. Thomson trained at Uniformed Services University of Health Sciences (USUHS) and completed an OB/GYN residency in 1997, then was stationed in Germany for four years. From 2001 to 2004, he served as chief of obstetrics at Madigan Army Medical Center in Tacoma, Wash., during which time he was deployed to Iraq for 10 months.
The OB/GYN expertise combined with the HM practice model that Dr. Thomson offers is a growing need in the military. “The number of women in the military is increasing, and there just aren’t a lot of people who understand female soldiers’ special needs,” he says.
Supporting women’s health has become an important aspect of battlefield medicine, namely the rooting out of potential sexual abuse. Dr. Thomson has published on the subject.1
In 2003, he was deployed as a general medical officer in Kuwait and assigned to the Basra area of Iraq, treating the gamut of patient needs. Recent Army policy changes, he says, ensure that OB/GYN military physicians now practice within their specialty.
A Canadian Perspective
Brendan James Hughes, MD, CCFP, returned from his military tour of duty and became a family practitioner in Lakefield, Ontario, a small community about 100 miles north of Toronto, and medical director of first-aid services for the Ontario Zone of the Canadian Red Cross.
In 2001, when Dr. Hughes was deployed as a hospitalist to Bosnia-Herzegovina for six months, the unrest from the civil war that involved Bosnians, Croatians, and Serbs (more than 100,000 were killed, and millions were injured or displaced), had settled, and his unit returned home without any loss of life. Upon his return, he transitioned from military life to become a full-time civilian hospitalist for six years in Ontario and Alberta. He now works as a part-time hospitalist.
Dr. Hughes says Canadian military practice is more acute and trauma-based now, as compared to his 2001 deployment in Eastern Europe. He notices many more deaths and major trauma cases in reports from Afghanistan, mostly blast injuries, limb amputations, and acute brain injuries, than there would be in a traditional, nonmilitary HM practice. He also notes that a lot of time and effort was placed on rehabilitation-focused practice that the patients required in the recovery phase.
Military practice differs from civilian hospitalist practice in other ways, he says. “In the military, every patient is essentially a workplace patient where the military is the employer,” Dr. Hughes says. Although clinicians maintain patient confidentiality, they are obliged to the chain of command to provide information on patient abilities. “We are careful not to relay a specific diagnosis without patient consent, but we have to dictate any needed restrictions on duty that are important in a combat situation, for themselves and for others,” he adds.
Such privacy and disclosure concerns are particularly difficult to navigate when it comes to diagnosis and treatment of alcohol and drug abuse, depression, post-traumatic stress, and suicide risk—issues that can lead soldiers to develop such long-term problems as substance abuse, marital discord, and marital abuse. TH
Andrea Sattinger is a freelance writer based in North Carolina.
Reference
- Thomson B, Nielsen P. Women’s healthcare in Operation Iraqi Freedom: a survey of camps with echelon one or two facilities. Mil Med. 2006;171:216-219.
Dr. Wilson’s photos from Afghanistan
Click images to enlarge

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|

|
PHOTOS COURTESY OF MAJ. RAMEY WILSON
The Year Ahead
Rising pressure to contain healthcare costs, increasing demands for safety and quality improvement, more focus on institutional accountability: In 2010, healthcare experts expect several dominant themes to continue converging and moving hospitalists even more to the center of key policy debates.
Peter Pronovost, MD, PhD, medical director of the Center for Innovation in Quality Patient Care and director of the Quality and Safety Research Group at Johns Hopkins University in Baltimore, sees three big themes moving to the fore. One is a greater focus on outcome measurements and accountability for performance, and he expects both carrots and sticks to be wielded. “So, both payment reform and social humiliation, or making things public,” Dr. Pronovost says. “Two, I see a lot more focus on measures that are population-based rather than hospital-based, so looking more at episodes of care.” The shift will force hospitalists to expand their purview beyond the hospital and, he says, partner more with community physicians to develop and monitor performance in such areas as transitions of care and general benchmarks of care.
Dr. Pronovost also expects “significant pressure on both the provider organization and individual clinician being paid less for what they do.” Finding ways to minimize costs will be a priority as payors increase scrutiny on expenses like unnecessary hospital readmissions. But hospitalists, he says, are better positioned than many other physicians to play a key role in the drive toward efficiency while also improving healthcare quality and safety. “I think hospitalists’ roles are going to go up dramatically,” Dr. Pronovost adds, “and I hope the field responds by making sure they put out people who have the skills to lead.”
End-of-Life Issues
Nancy Berlinger, PhD, deputy director and research scholar at The Hastings Center in Garrison, N.Y., cites end-of-life care as another theme likely to gain traction in 2010. As project director of the center’s revised ethical guidelines for end-of-life care, Dr. Berlinger notes how often clinicians in her working group have invoked the hospitalist profession. It’s no accident. “Hospitalists are increasingly associated with the care of patients on Medicare,” she says, adding Medicare beneficiaries are far more likely to be nearing the end of life.
Demographics suggest that connection will continue to grow in 2010 and beyond. Dr. Berlinger points to a 2009 New England Journal of Medicine study showing that the odds of a hospitalized Medicare patient receiving care from a hospitalist increased at a brisk 29.2% annual clip from 1997 through 2006.1 And while the U.S. faces a shortage of geriatricians, HM is growing rapidly as a medical profession. “By default, whether or not hospitalists self-identify as caring for older Americans,” Dr. Berlinger says, “this is their area of practical specialization.”
With that specialization comes added responsibility to assist with advanced-care planning and helping patients to document their wishes. Similarly, she says, it means acknowledging that these patients are more likely to have comorbid conditions and identify with goals of care. “I don’t think there’s any way around this,” she says. “Medicare and hospitalists, whether by accident or design, are increasingly joined at the hip. That is something that hospitalists, as a profession, will always need to keep their eye on.”
A parallel trend is that other doctors increasingly view hospitalists as hospital specialists. “The hospitalist’s responsibilities are not just in terms of the patients they care for, but also in terms of the institution itself,” Dr. Berlinger says. Non-staff physicians, for example, expect hospitalists to know how a hospital’s in-patient care system works. Practically speaking, as electronic medical records (EMR) become more commonplace, hospitalists will be increasingly relied upon to understand a hospital’s information technology.
—Peter Pronovost, MD, PhD, medical director, Center for Innovation in Quality Patient Care, Johns Hopkins University, Baltimore
New Economy, New Hospital Landscape
Douglas Wood, MD, chair of the Division of Health Care Policy and Research at the Mayo Clinic in Rochester, Minn., points to language in the federal healthcare reform legislation as evidence that hospitals and hospitalists will need to be in sync in other ways to avoid future penalties. One provision, for example, would increase the penalties for hospital-acquired infections. Other language seeks to reduce unnecessary readmissions.
Likewise, Dr. Wood says, addressing geographical variations in healthcare payments driven largely by unnecessary overutilization—including excessive use of ICU care, in-patient care, imaging, and specialist services—might mean asking hospitalists to take on more aspects of patient care.
Meanwhile, increased interest in demonstration projects that might achieve savings (e.g., accountable care organizations and bundled payments) suggests that proactive hospitals should again look to hospitalists. The flurry of new proposals won’t fundamentally change hospitalists’ responsibilities to provide effective and efficient care, “but it will put more emphasis on what they’re doing,” Dr. Wood says, “to the degree that hospitalists could take a lead in demonstrating how you can provide better outcomes at a lower overall utilization of resources.”
Regardless of how slowly or quickly these initiatives proceed at the national level, he says, hospitalists should be mindful that several states are well ahead of the curve and are likely to be more aggressive in instituting policy changes.
The Bottom Line
If there’s a single, overriding theme for 2010, Bradley Flansbaum, DO, MPH, FACP, FHM, director of hospitalist services at Lenox Hill Hospital in New York City and a member of SHM’s Public Policy Committee, says it might be that of dealing with the unknown. Squeezing healthcare costs and more tightly regulating inflation will have a greater effect on a hospital’s bottom line and thus impact what’s required of hospitalists. Even so, the profession will have to wait and see whether and how various proposals are codified and implemented. “We don’t know exactly what things are going to look like,” he says.
Nor is there a good sense of how new standards for transparency, quality, and accountability might be measured. “While people want more measurement and they want more report-card-type information, the data that we can acquire right now and how we analyze that data are still fairly primitive,” Dr. Flansbaum says. Even current benchmarks are lacking in how to determine who’s doing a good job and who isn’t, he says.
One big question that must be answered, then: Are we even looking at the right measurements? “Or, do the right measurements exist, or do we have the databases, the registries, the sources, to make the decisions we need to make?” he says.
Any new proposals will require another round of such questions and filling-in of blanks to add workable details to vague and potentially confusing language.
“I think we know that change is afoot, and most smart hospitalists know that the system needs to run leaner,” Dr. Flansbaum says. “But how each one of us is going to function in our hospital, and the kinds of demands that will be placed on us, and what we’re going to need to do with the doctors in the community and the other nonphysician colleagues that we work with, is all really unknown.” TH
Bryn Nelson is a freelance medical writer based in Seattle.
Reference
- Kuo YF, Sharma G, Freeman JL, Goodwin JS. Growth in the care of older patients by hospitalists in the United States. N Engl J Med. 2009;360(11): 1102-1112.
Image Source: PAGADESIGN, OVERSNAP/ISTOCKPHOTO.COM
Rising pressure to contain healthcare costs, increasing demands for safety and quality improvement, more focus on institutional accountability: In 2010, healthcare experts expect several dominant themes to continue converging and moving hospitalists even more to the center of key policy debates.
Peter Pronovost, MD, PhD, medical director of the Center for Innovation in Quality Patient Care and director of the Quality and Safety Research Group at Johns Hopkins University in Baltimore, sees three big themes moving to the fore. One is a greater focus on outcome measurements and accountability for performance, and he expects both carrots and sticks to be wielded. “So, both payment reform and social humiliation, or making things public,” Dr. Pronovost says. “Two, I see a lot more focus on measures that are population-based rather than hospital-based, so looking more at episodes of care.” The shift will force hospitalists to expand their purview beyond the hospital and, he says, partner more with community physicians to develop and monitor performance in such areas as transitions of care and general benchmarks of care.
Dr. Pronovost also expects “significant pressure on both the provider organization and individual clinician being paid less for what they do.” Finding ways to minimize costs will be a priority as payors increase scrutiny on expenses like unnecessary hospital readmissions. But hospitalists, he says, are better positioned than many other physicians to play a key role in the drive toward efficiency while also improving healthcare quality and safety. “I think hospitalists’ roles are going to go up dramatically,” Dr. Pronovost adds, “and I hope the field responds by making sure they put out people who have the skills to lead.”
End-of-Life Issues
Nancy Berlinger, PhD, deputy director and research scholar at The Hastings Center in Garrison, N.Y., cites end-of-life care as another theme likely to gain traction in 2010. As project director of the center’s revised ethical guidelines for end-of-life care, Dr. Berlinger notes how often clinicians in her working group have invoked the hospitalist profession. It’s no accident. “Hospitalists are increasingly associated with the care of patients on Medicare,” she says, adding Medicare beneficiaries are far more likely to be nearing the end of life.
Demographics suggest that connection will continue to grow in 2010 and beyond. Dr. Berlinger points to a 2009 New England Journal of Medicine study showing that the odds of a hospitalized Medicare patient receiving care from a hospitalist increased at a brisk 29.2% annual clip from 1997 through 2006.1 And while the U.S. faces a shortage of geriatricians, HM is growing rapidly as a medical profession. “By default, whether or not hospitalists self-identify as caring for older Americans,” Dr. Berlinger says, “this is their area of practical specialization.”
With that specialization comes added responsibility to assist with advanced-care planning and helping patients to document their wishes. Similarly, she says, it means acknowledging that these patients are more likely to have comorbid conditions and identify with goals of care. “I don’t think there’s any way around this,” she says. “Medicare and hospitalists, whether by accident or design, are increasingly joined at the hip. That is something that hospitalists, as a profession, will always need to keep their eye on.”
A parallel trend is that other doctors increasingly view hospitalists as hospital specialists. “The hospitalist’s responsibilities are not just in terms of the patients they care for, but also in terms of the institution itself,” Dr. Berlinger says. Non-staff physicians, for example, expect hospitalists to know how a hospital’s in-patient care system works. Practically speaking, as electronic medical records (EMR) become more commonplace, hospitalists will be increasingly relied upon to understand a hospital’s information technology.
—Peter Pronovost, MD, PhD, medical director, Center for Innovation in Quality Patient Care, Johns Hopkins University, Baltimore
New Economy, New Hospital Landscape
Douglas Wood, MD, chair of the Division of Health Care Policy and Research at the Mayo Clinic in Rochester, Minn., points to language in the federal healthcare reform legislation as evidence that hospitals and hospitalists will need to be in sync in other ways to avoid future penalties. One provision, for example, would increase the penalties for hospital-acquired infections. Other language seeks to reduce unnecessary readmissions.
Likewise, Dr. Wood says, addressing geographical variations in healthcare payments driven largely by unnecessary overutilization—including excessive use of ICU care, in-patient care, imaging, and specialist services—might mean asking hospitalists to take on more aspects of patient care.
Meanwhile, increased interest in demonstration projects that might achieve savings (e.g., accountable care organizations and bundled payments) suggests that proactive hospitals should again look to hospitalists. The flurry of new proposals won’t fundamentally change hospitalists’ responsibilities to provide effective and efficient care, “but it will put more emphasis on what they’re doing,” Dr. Wood says, “to the degree that hospitalists could take a lead in demonstrating how you can provide better outcomes at a lower overall utilization of resources.”
Regardless of how slowly or quickly these initiatives proceed at the national level, he says, hospitalists should be mindful that several states are well ahead of the curve and are likely to be more aggressive in instituting policy changes.
The Bottom Line
If there’s a single, overriding theme for 2010, Bradley Flansbaum, DO, MPH, FACP, FHM, director of hospitalist services at Lenox Hill Hospital in New York City and a member of SHM’s Public Policy Committee, says it might be that of dealing with the unknown. Squeezing healthcare costs and more tightly regulating inflation will have a greater effect on a hospital’s bottom line and thus impact what’s required of hospitalists. Even so, the profession will have to wait and see whether and how various proposals are codified and implemented. “We don’t know exactly what things are going to look like,” he says.
Nor is there a good sense of how new standards for transparency, quality, and accountability might be measured. “While people want more measurement and they want more report-card-type information, the data that we can acquire right now and how we analyze that data are still fairly primitive,” Dr. Flansbaum says. Even current benchmarks are lacking in how to determine who’s doing a good job and who isn’t, he says.
One big question that must be answered, then: Are we even looking at the right measurements? “Or, do the right measurements exist, or do we have the databases, the registries, the sources, to make the decisions we need to make?” he says.
Any new proposals will require another round of such questions and filling-in of blanks to add workable details to vague and potentially confusing language.
“I think we know that change is afoot, and most smart hospitalists know that the system needs to run leaner,” Dr. Flansbaum says. “But how each one of us is going to function in our hospital, and the kinds of demands that will be placed on us, and what we’re going to need to do with the doctors in the community and the other nonphysician colleagues that we work with, is all really unknown.” TH
Bryn Nelson is a freelance medical writer based in Seattle.
Reference
- Kuo YF, Sharma G, Freeman JL, Goodwin JS. Growth in the care of older patients by hospitalists in the United States. N Engl J Med. 2009;360(11): 1102-1112.
Image Source: PAGADESIGN, OVERSNAP/ISTOCKPHOTO.COM
Rising pressure to contain healthcare costs, increasing demands for safety and quality improvement, more focus on institutional accountability: In 2010, healthcare experts expect several dominant themes to continue converging and moving hospitalists even more to the center of key policy debates.
Peter Pronovost, MD, PhD, medical director of the Center for Innovation in Quality Patient Care and director of the Quality and Safety Research Group at Johns Hopkins University in Baltimore, sees three big themes moving to the fore. One is a greater focus on outcome measurements and accountability for performance, and he expects both carrots and sticks to be wielded. “So, both payment reform and social humiliation, or making things public,” Dr. Pronovost says. “Two, I see a lot more focus on measures that are population-based rather than hospital-based, so looking more at episodes of care.” The shift will force hospitalists to expand their purview beyond the hospital and, he says, partner more with community physicians to develop and monitor performance in such areas as transitions of care and general benchmarks of care.
Dr. Pronovost also expects “significant pressure on both the provider organization and individual clinician being paid less for what they do.” Finding ways to minimize costs will be a priority as payors increase scrutiny on expenses like unnecessary hospital readmissions. But hospitalists, he says, are better positioned than many other physicians to play a key role in the drive toward efficiency while also improving healthcare quality and safety. “I think hospitalists’ roles are going to go up dramatically,” Dr. Pronovost adds, “and I hope the field responds by making sure they put out people who have the skills to lead.”
End-of-Life Issues
Nancy Berlinger, PhD, deputy director and research scholar at The Hastings Center in Garrison, N.Y., cites end-of-life care as another theme likely to gain traction in 2010. As project director of the center’s revised ethical guidelines for end-of-life care, Dr. Berlinger notes how often clinicians in her working group have invoked the hospitalist profession. It’s no accident. “Hospitalists are increasingly associated with the care of patients on Medicare,” she says, adding Medicare beneficiaries are far more likely to be nearing the end of life.
Demographics suggest that connection will continue to grow in 2010 and beyond. Dr. Berlinger points to a 2009 New England Journal of Medicine study showing that the odds of a hospitalized Medicare patient receiving care from a hospitalist increased at a brisk 29.2% annual clip from 1997 through 2006.1 And while the U.S. faces a shortage of geriatricians, HM is growing rapidly as a medical profession. “By default, whether or not hospitalists self-identify as caring for older Americans,” Dr. Berlinger says, “this is their area of practical specialization.”
With that specialization comes added responsibility to assist with advanced-care planning and helping patients to document their wishes. Similarly, she says, it means acknowledging that these patients are more likely to have comorbid conditions and identify with goals of care. “I don’t think there’s any way around this,” she says. “Medicare and hospitalists, whether by accident or design, are increasingly joined at the hip. That is something that hospitalists, as a profession, will always need to keep their eye on.”
A parallel trend is that other doctors increasingly view hospitalists as hospital specialists. “The hospitalist’s responsibilities are not just in terms of the patients they care for, but also in terms of the institution itself,” Dr. Berlinger says. Non-staff physicians, for example, expect hospitalists to know how a hospital’s in-patient care system works. Practically speaking, as electronic medical records (EMR) become more commonplace, hospitalists will be increasingly relied upon to understand a hospital’s information technology.
—Peter Pronovost, MD, PhD, medical director, Center for Innovation in Quality Patient Care, Johns Hopkins University, Baltimore
New Economy, New Hospital Landscape
Douglas Wood, MD, chair of the Division of Health Care Policy and Research at the Mayo Clinic in Rochester, Minn., points to language in the federal healthcare reform legislation as evidence that hospitals and hospitalists will need to be in sync in other ways to avoid future penalties. One provision, for example, would increase the penalties for hospital-acquired infections. Other language seeks to reduce unnecessary readmissions.
Likewise, Dr. Wood says, addressing geographical variations in healthcare payments driven largely by unnecessary overutilization—including excessive use of ICU care, in-patient care, imaging, and specialist services—might mean asking hospitalists to take on more aspects of patient care.
Meanwhile, increased interest in demonstration projects that might achieve savings (e.g., accountable care organizations and bundled payments) suggests that proactive hospitals should again look to hospitalists. The flurry of new proposals won’t fundamentally change hospitalists’ responsibilities to provide effective and efficient care, “but it will put more emphasis on what they’re doing,” Dr. Wood says, “to the degree that hospitalists could take a lead in demonstrating how you can provide better outcomes at a lower overall utilization of resources.”
Regardless of how slowly or quickly these initiatives proceed at the national level, he says, hospitalists should be mindful that several states are well ahead of the curve and are likely to be more aggressive in instituting policy changes.
The Bottom Line
If there’s a single, overriding theme for 2010, Bradley Flansbaum, DO, MPH, FACP, FHM, director of hospitalist services at Lenox Hill Hospital in New York City and a member of SHM’s Public Policy Committee, says it might be that of dealing with the unknown. Squeezing healthcare costs and more tightly regulating inflation will have a greater effect on a hospital’s bottom line and thus impact what’s required of hospitalists. Even so, the profession will have to wait and see whether and how various proposals are codified and implemented. “We don’t know exactly what things are going to look like,” he says.
Nor is there a good sense of how new standards for transparency, quality, and accountability might be measured. “While people want more measurement and they want more report-card-type information, the data that we can acquire right now and how we analyze that data are still fairly primitive,” Dr. Flansbaum says. Even current benchmarks are lacking in how to determine who’s doing a good job and who isn’t, he says.
One big question that must be answered, then: Are we even looking at the right measurements? “Or, do the right measurements exist, or do we have the databases, the registries, the sources, to make the decisions we need to make?” he says.
Any new proposals will require another round of such questions and filling-in of blanks to add workable details to vague and potentially confusing language.
“I think we know that change is afoot, and most smart hospitalists know that the system needs to run leaner,” Dr. Flansbaum says. “But how each one of us is going to function in our hospital, and the kinds of demands that will be placed on us, and what we’re going to need to do with the doctors in the community and the other nonphysician colleagues that we work with, is all really unknown.” TH
Bryn Nelson is a freelance medical writer based in Seattle.
Reference
- Kuo YF, Sharma G, Freeman JL, Goodwin JS. Growth in the care of older patients by hospitalists in the United States. N Engl J Med. 2009;360(11): 1102-1112.
Image Source: PAGADESIGN, OVERSNAP/ISTOCKPHOTO.COM
Recognition and Management of Nighttime Reflux Symptoms
A supplement to Internal Medicine News supported by Santarus, Inc. The supplement is based on a faculty interview.
To view the supplement, click the image above.
FACULTY
Colin W. Howden, MD
Professor, Division of Gastroenterology
Northwestern University Feinberg School of Medicine
A supplement to Internal Medicine News supported by Santarus, Inc. The supplement is based on a faculty interview.
To view the supplement, click the image above.
FACULTY
Colin W. Howden, MD
Professor, Division of Gastroenterology
Northwestern University Feinberg School of Medicine
A supplement to Internal Medicine News supported by Santarus, Inc. The supplement is based on a faculty interview.
To view the supplement, click the image above.
FACULTY
Colin W. Howden, MD
Professor, Division of Gastroenterology
Northwestern University Feinberg School of Medicine
ONLINE EXCLUSIVE: Audio interview with HM10 Course Director
ONLINE EXCLUSIVE: Training in Two Cultures: Medicine and Soldiering
Undergrads who choose the military and attend USUHS receive free tuition, books and other supplies; hand-held devices and related subscriptions, and basic medical equipment such as stethoscopes. In addition, USUHS medical students are paid as an active-duty second lieutenant (the going rate for the U.S. Army is about $1,900 per month).
Once they graduate, military residents work in uniform at military facilities and are afforded housing allowances. The government also covers the cost of medical malpractice insurance and supports them in any litigation while they are on active service. If they complete 20 years in active service, military physicians receive a generous retirement package, including a retained 40% to 50% pension for the rest of their lives, and they can seek work in the civilian sector after their military career.
For those who choose the military from the outset, the Department of Defense offers sign-on bonuses of $20,000 and a Health Service Professional Scholarship (HPSP) program for qualified applicants. It covers all medical school costs at a civilian medical school of the student’s choice. (Recent studies show the typical medical school grad has $120,000 of school load debt; $160,000 if they attended a private school.) The caveat is that after graduation, whether from USUHS or a civilian medical school, the physician works in uniform as a military physician for a pre-determined payback period (e.g., the Army obligation is one year of service for every year of scholarship).
The military offers training programs in medical, dental, optometry, veterinary, psychiatric nurse practitioner, and clinical and counseling psychology. Training at USUHS or with an HPSP requires each student before matriculation to choose his or her preferred branch of military service for the payback period. Whichever route a student takes, USUHS or HPSP, the student will end up as a doctor and a trained service member knowledgeable about areas including rank structure, military administration, and personal physical fitness.
Undergrads who choose the military and attend USUHS receive free tuition, books and other supplies; hand-held devices and related subscriptions, and basic medical equipment such as stethoscopes. In addition, USUHS medical students are paid as an active-duty second lieutenant (the going rate for the U.S. Army is about $1,900 per month).
Once they graduate, military residents work in uniform at military facilities and are afforded housing allowances. The government also covers the cost of medical malpractice insurance and supports them in any litigation while they are on active service. If they complete 20 years in active service, military physicians receive a generous retirement package, including a retained 40% to 50% pension for the rest of their lives, and they can seek work in the civilian sector after their military career.
For those who choose the military from the outset, the Department of Defense offers sign-on bonuses of $20,000 and a Health Service Professional Scholarship (HPSP) program for qualified applicants. It covers all medical school costs at a civilian medical school of the student’s choice. (Recent studies show the typical medical school grad has $120,000 of school load debt; $160,000 if they attended a private school.) The caveat is that after graduation, whether from USUHS or a civilian medical school, the physician works in uniform as a military physician for a pre-determined payback period (e.g., the Army obligation is one year of service for every year of scholarship).
The military offers training programs in medical, dental, optometry, veterinary, psychiatric nurse practitioner, and clinical and counseling psychology. Training at USUHS or with an HPSP requires each student before matriculation to choose his or her preferred branch of military service for the payback period. Whichever route a student takes, USUHS or HPSP, the student will end up as a doctor and a trained service member knowledgeable about areas including rank structure, military administration, and personal physical fitness.
Undergrads who choose the military and attend USUHS receive free tuition, books and other supplies; hand-held devices and related subscriptions, and basic medical equipment such as stethoscopes. In addition, USUHS medical students are paid as an active-duty second lieutenant (the going rate for the U.S. Army is about $1,900 per month).
Once they graduate, military residents work in uniform at military facilities and are afforded housing allowances. The government also covers the cost of medical malpractice insurance and supports them in any litigation while they are on active service. If they complete 20 years in active service, military physicians receive a generous retirement package, including a retained 40% to 50% pension for the rest of their lives, and they can seek work in the civilian sector after their military career.
For those who choose the military from the outset, the Department of Defense offers sign-on bonuses of $20,000 and a Health Service Professional Scholarship (HPSP) program for qualified applicants. It covers all medical school costs at a civilian medical school of the student’s choice. (Recent studies show the typical medical school grad has $120,000 of school load debt; $160,000 if they attended a private school.) The caveat is that after graduation, whether from USUHS or a civilian medical school, the physician works in uniform as a military physician for a pre-determined payback period (e.g., the Army obligation is one year of service for every year of scholarship).
The military offers training programs in medical, dental, optometry, veterinary, psychiatric nurse practitioner, and clinical and counseling psychology. Training at USUHS or with an HPSP requires each student before matriculation to choose his or her preferred branch of military service for the payback period. Whichever route a student takes, USUHS or HPSP, the student will end up as a doctor and a trained service member knowledgeable about areas including rank structure, military administration, and personal physical fitness.
Preventing venous thromboembolism in long-term care residents: Cautious advice based on limited data
Randomized trials that included more than 20,000 medical patients have shown that anticoagulant therapy is safe and effective in preventing venous thromboembolism (VTE), ie, deep vein thrombosis and pulmonary embolism.
However, these trials were done in hospitalized patients, who typically had an acute medical illness and who, if eligible, received a short (7- to 10-day) course of anticoagulant prophylaxis.
Little attention has been given to VTE prophylaxis in residents of long-term care facilities. These patients have risk profiles similar to those of hospitalized medical patients. Some of them may have been transferred from an acute care hospital. In addition, most are elderly, and many have reduced mobility and are at risk for illnesses such as stroke and cardiorespiratory insufficiency, which increase the risk of VTE.
VTE in residents of long-term care facilities is a growing concern. By some estimates, by the year 2030 more than 20% of the US population (70.2 million people) will be over 65 years of age.1 Of those who reached age 65 in 1990, an estimated 43% will enter a nursing home at least once before they die—32% for 3 months, 24% for at least a year, and 9% for at least 5 years.2
Against this background, the objectives of this review are to consider:
- The scope of the problem of VTE in long-term care residents
- Why VTE prophylaxis is often overlooked in medical patients
- Evidence—or lack of evidence—for the safety and efficacy of VTE prophylaxis in long-term care residents and other medical patients
- Available options for VTE prophylaxis
- Which long-term care residents should or should not be considered for prophylaxis.
THE TRUE SCOPE OF THE PROBLEM IS UNKNOWN
The incidence of acute VTE among nursing home residents is reported to be 1.3 events per 100 person-years.3 About 8% of cases of pulmonary embolism and 10% of cases of deep venous thrombosis in the elderly are in nursing home residents.4
However, only 20% of patients with VTE have typical symptoms such as leg pain and swelling or acute dyspnea and chest pain, while 80% have no symptoms.5
Furthermore, deep venous thrombosis is more likely to be clinically silent in patients whose mobility is impaired, such as nursing home residents, as the symptoms arising from obstruction of venous flow are more pronounced with walking.
Pulmonary embolism is also underdiagnosed in this group. An autopsy study of 234 nursing home residents found undiagnosed pulmonary embolism to be the cause of death in 8%, and 40% of cases of pulmonary embolism were not suspected before the patient died.6 Yet pulmonary embolism has a higher case-fatality rate in the elderly than in younger patients, particularly when elderly patients have comorbidities.7
A reason why the diagnosis is so often missed is that pulmonary embolism can present atypically in the elderly, with syncope being more common and tachycardia being less common than in younger patients.8
Since so many cases of VTE are clinically silent and most long-term care residents who die do not undergo autopsy, the true scope of VTE as a clinical problem in these patients is unknown. Consequently, the best way to diagnose, prevent, and treat VTE is also unclear.
WHY IS VTE PREVENTION SO OFTEN OVERLOOKED IN MEDICAL PATIENTS?
In general, nonsurgical patients receive suboptimal thromboprophylaxis. National and international chart audits and cross-sectional studies show that only 16% to 33% of hospitalized medical patients at risk for VTE receive appropriate anticoagulant prophylaxis.9 Though no audits in long-term care facilities have been published, the rate of appropriate prophylaxis is likely comparable to or possibly less than that in medical patients in the hospital. In contrast, in surgical patients the rate is much higher—up to 90%.10,11
Why is VTE prophylaxis so underused in medical patients?
One reason is that we do not really know the baseline risk of VTE in medical patients, particularly in those with chronic illness who require long-term care.12 This is relevant because, in the absence of data about patients’ baseline risk, anticoagulant prophylaxis should be ordered selectively, as it poses known risks of bleeding. The risk is greater in elderly people with comorbidities, as are the associated costs.
In addition, relatively few studies have assessed thromboprophylaxis in medical patients, especially in residents of long-term care facilities.
Another reason is that we lack practice guidelines for patients who need long-term care. The well-accepted guidelines from the American College of Chest Physicians (ACCP) cite advanced age and immobility as risk factors for VTE and strongly recommend prophylaxis in acutely ill medical patients who have limited mobility and an additional risk factor such as infection or cancer.13 Though elderly residents of long-term care facilities may share some of these risk factors, the ACCP guidelines make no specific recommendations for this group.
The attitudes of health care professionals may also pose a barrier. Lloyd et al (unpublished data, 2009) surveyed 1,601 health care professionals in Ontario, Canada, in 2007, to assess potential barriers to anticoagulant prophylaxis in hospitalized medical patients. Respondents cited concerns about the risk of bleeding from anticoagulants, lack of clear indications and contraindications for anticoagulant prophylaxis, and lack of time to consider VTE prophylaxis in every patient. (They did not, however, cite disagreement with guidelines or patient discomfort from subcutaneous anticoagulant injections as barriers.) It is reasonable to assume that these attitudes may also pose a problem in long-term care residents.
Finally, no randomized trials have evaluated the efficacy and safety of anticoagulant drugs or mechanical methods of prophylaxis in long-term care residents. Studies have shown that a short course (7–10 days) of an anticoagulant drug effectively prevents VTE in acutely ill patients, but the efficacy of an extended course in patients with chronic illness who require long-term care is not clear. Therefore, recommendations about thromboprophylaxis in long-term care residents should be made with the caveat that they are based on indirect evidence from other patient groups. This is a considerable limitation.
OPTIONS FOR THROMBOPROPHYLAXIS IN LONG-TERM CARE RESIDENTS
Options for thromboprophylaxis fall into two broad categories: anticoagulant drugs and mechanical devices.
Anticoagulant prophylactic drugs
These agents have been assessed in randomized trials in surgical or acutely ill medical patients, although fondaparinux and tinzaparin are not approved for use in medical patients. Furthermore, none of them has been evaluated in residents of long-term care facilities.
The choice of anticoagulant for prophylaxis is determined largely by clinical factors.
Low-molecular-weight heparins are popular both in and out of the hospital because they have predictable pharmacokinetic properties, they come in convenient prefilled syringes, and they can be given once daily. However, some of them may bioaccumulate in patients with impaired renal function, as they are cleared primarily by the kidney.
Unfractionated heparin is likely to be safer in patients with severe renal insufficiency (creatinine clearance < 30 mL/min), as it is cleared via nonrenal mechanisms.
However, a recent single-arm trial of dalteparin 5,000 IU once daily in critically ill patients with severe renal insufficiency found no evidence of an excessive anticoagulant effect or of drug bioaccumulation.15 Dalteparin may thus be an alternative to unfractionated heparin in medical patients with impaired renal function.
Fondaparinux, a newer anticoagulant, is also given once daily. It is the anticoagulant of choice in patients who have had heparin-induced thrombocytopenia because it is not derived from heparin and likely does not cross-react with heparin-induced thrombocytopenia antibodies.16,17
Limited data on benefit of prophylactic anticoagulant drugs
As mentioned, the trials that confirmed the efficacy and safety of anticoagulant prophylaxis were in surgical patients and hospitalized medical patients, not elderly long-term care residents. The poor evidence for anticoagulant prophylaxis in these patients may be strengthened if extended-duration, out-of-hospital prophylaxis were shown to be effective in medical patients. Long-term care residents could more reasonably be compared with medical patients discharged home with a chronic or resolving illness than with those who are hospitalized.
There is some evidence, although with caveats, that extended anticoagulant prophylaxis, started after an acute illness has resolved, confers a benefit. A recent randomized trial compared extended-duration and short-duration prophylaxis (5 weeks vs 10 days) with enoxaparin 40 mg once daily in 4,726 medical patients with impaired mobility.18 The risk of any VTE event was 44% lower with extended-duration prophylaxis (2.8% vs 4.9%; P = .001) and the risk of symptomatic VTE was 73% lower (0.3% vs 1.1%; P = .004), and this benefit persisted 2 months after treatment was stopped (3.0% vs 5.2%; P = .0015). However, extended treatment conferred a fourfold higher risk of major bleeding (0.6% vs 0.15%; P = .019).
These findings should also be considered in terms of absolute benefit and harm. Treating 1,000 patients for 5 weeks instead of 10 days would prevent eight episodes of symptomatic VTE (absolute risk reduction = 0.8%, number needed to treat = 125) at the cost of four to five episodes of major bleeding (absolute risk increase = 0.45%, number needed to harm = 222). This is a modest net therapeutic benefit.
The therapeutic benefit would be greater if we consider all episodes of VTE, both symptomatic and asymptomatic. Treating 1,000 patients for 5 weeks would prevent 20 episodes of symptomatic or asymptomatic VTE (absolute risk reduction = 2.1%, number needed to treat = 48). However, the clinical importance of asymptomatic VTE is questionable.
Given these considerations, if extended-duration anticoagulant prophylaxis is considered, it should be for patients at highest risk to optimize both its net therapeutic benefits and its cost-effectiveness.
Mechanical prophylaxis
Mechanical thromboprophylactic devices—graduated or elastic compression stockings and intermittent pneumatic compression devices—are effective when used by themselves in surgical patients.13 However, in a randomized controlled trial in patients with ischemic stroke, the rate of VTE was 10.0% with graduated compression stockings in addition to “usual care VTE prophylaxis” vs 10.5% with usual care alone, and patients in the stocking group had a fourfold higher risk of developing skin breaks, ulcers, blisters, or necrosis (5% vs 1%; odds ratio 4.18; 95% CI 2.4–7.3).19 Furthermore, improperly fitted stockings, especially those that are thigh-length, can be uncomfortable to wear and difficult to apply.
Overall, the role of mechanical thromboprophylaxis in long-term care facilities is not clear. If it is considered, there should be a compelling reason to use it—for example, for patients at high risk in whom anticoagulants are contraindicated because of ongoing bleeding or a higher risk of bleeding (eg, recent gastrointestinal bleeding, hemorrhagic stroke, coagulopathy, or thrombocytopenia). Furthermore, if stockings are used, they should be properly fitted and routinely monitored for adverse effects, since elderly patients are likely to be most susceptible to skin breakdown.
WHICH LONG-TERM CARE RESIDENTS SHOULD RECEIVE VTE PROPHYLAXIS?
Old age and immobility are not the only risk factors
The current ACCP guidelines suggest considering thromboprophylaxis for hospitalized medical patients over age 75 who cannot walk without assistance.13 However, we lack evidence to suggest a similar strategy in long-term care residents.
The ACCP guidelines are based on data on risk. Nearly 25% of elderly patients with confirmed pulmonary embolism had been immobile prior to their diagnosis.8 In addition, prolonged bed rest (> 14 days) has been reported to be the strongest independent risk factor for symptomatic deep venous thrombosis, increasing the risk more than fivefold.20 Advanced age is also considered a risk factor for VTE, as risk starts to increase at age 40 and doubles each decade of life thereafter.18
No study has assessed the impact of these factors on the risk of VTE in long-term care residents. Since most of such patients are elderly and have impaired mobility, we believe a more selective approach should be used in assigning VTE risk status, one that does not use advanced age and immobility as the only criteria for starting thromboprophylaxis.
Residents of long-term care facilities may be immobile because of underlying illness or disability, such as cognitive impairment, sensory impairment (eg, poor access to corrective lenses and hearing aids), or poor access to assist devices (eg, walkers, canes). In addition, iatrogenic factors that decrease mobility such as indwelling bladder catheters and physical restraints are also common in such patients.
Efforts to improve mobility should be encouraged. However, we recommend that thromboprophylaxis be considered only in patients who have both impaired mobility and an intercurrent acute medical illness such as an acute infection or acute inflammatory disease.13
A related issue is the difference between long-term care residents with a chronic but stable disease and those with acute disease. Patients with acute exacerbations of congestive heart failure or chronic obstructive lung disease may be considered for thromboprophylaxis, as they become more comparable to acutely ill medical patients in whom clinical trials have shown the effectiveness of anticoagulant prophylaxis. On the other hand, patients with these diseases who remain stable may not need prophylaxis.
This approach avoids giving long-term anticoagulant prophylaxis to patients who have irreversible diseases and limits the use of these drugs and devices to higher-risk periods.
Consider thromboprophylaxis if…
- An acute exacerbation of congestive heart failure or chronic obstructive pulmonary disease
- Acute infection (eg, urosepsis, pneumonia, cellulitis, infectious diarrhea)
- An acute exacerbation of an inflammatory disease (eg, rheumatoid arthritis)
- Active cancer (eg, patient receiving radiation therapy or chemotherapy)
- Immobility and prior VTE.
Do not routinely consider prophylaxis if…
We also believe patients should not be routinely considered for anticoagulant VTE prophylaxis if they have:
- Chronic but stable cardiorespiratory disease
- Chronic but stable infectious or inflammatory disease
- Terminal cancer with very limited life expectancy
- Any contraindication to anticoagulants (eg, active bleeding, recent bleeding, coagulopathy, thrombocytopenia).
ANTICOAGULANT PROPHYLAXIS POSES RISKS IN LONG-TERM CARE RESIDENTS
Bleeding is the principal risk
The incidence of clinically important bleeding associated with anticoagulant prophylaxis is 0.2% to 5.6%, and the risk of fatal bleeding is 0.02% to 0.5%.21–24
As no randomized trial has examined anticoagulant prophylaxis in elderly long-term care residents, their bleeding risk with this therapy is unclear. However, older patients are likely to be at higher risk than younger patients because they have more comorbidities, take more drugs that could interact with heparin and potentiate bleeding, and have fragile skin, predisposing to injury from subcutaneous injections.
Also, renal function tends to decline with age. In a retrospective study of 854 outpatients over age 65, 29% had moderate renal insufficiency (creatinine clearance 30–50 mL/min), and 6% had severe renal insufficiency (creatinine clearance < 30 mL/min).25 Recent evidence suggests that some low-molecular-weight heparins (dalteparin and tinzaparin) do not bioaccumulate in patients with impaired renal function. However, enoxaparin and fondaparinux should be used with caution in patients with moderate to severe renal impairment.
Though much attention has recently been paid to increasing anticoagulant doses if the patient is obese, residents of long-term care facilities are more likely to be underweight. Dose adjustment should be considered when a low-molecular-weight heparin or fondaparinux is given to patients weighing less than 50 kg.
Heparin-induced thrombocytopenia
The other major risk of anticoagulant prophylaxis is heparin-induced thrombocytopenia, an infrequent but life-threatening complication caused by the formation of antibodies to the heparin-derived anticoagulant and a platelet surface antigen. It is associated with moderate thrombocytopenia and an incidence of venous or arterial thrombosis that is over 50%.26
No study has assessed the incidence of heparin-induced thrombocytopenia in long-term care residents. A meta-analysis reported that the risk with anticoagulant prophylaxis was 1.6% with unfractionated heparin (95% confidence interval [CI] 1.2%–2.1%) and 0.6% with low-molecular-weight heparin (95% CI 0.4%–0.9%), and that this risk increased with the duration of prophylaxis.27 If anticoagulant prophylaxis were given to all long-term care residents for extended durations (eg, for the duration of reduced mobility), the incidence and prevalence of heparin-induced thrombocytopenia would likely become a major concern.
Whenever anticoagulant prophylaxis is considered, the risks of both thrombosis and bleeding should be considered. Patients who are receiving anticoagulant prophylaxis should also be monitored for bleeding and heparin-induced thrombocytopenia. This is particularly true in long-term care residents, in whom the risks and benefits of anticoagulant prophylaxis are extrapolated from data from other populations.
MORE RESEARCH IS NEEDED
To date, we lack audits of thromboprophylaxis, clinical practice guidelines, and clear indications and contraindications for anticoagulant prophylaxis in long-term care residents. In the absence of such data, extrapolating the efficacy and safety of thromboprophylaxis from hospitalized patients to long-term care residents is difficult.
Clearly, additional research is needed to identify which long-term care residents would benefit most from thromboprophylaxis. In the meantime, a selective approach to identifying patients who should be considered for thromboprophylaxis should be adopted.
- Cornman JM. Questions for societies with “third age” populations. The Extension-of-Life Working Group, The Gerontological Society of America. Acad Med 1997; 72:856–862.
- Kemper P, Murtaugh CM. Lifetime use of nursing home care. N Engl J Med 1991; 324:595–600.
- Gomes JP, Shaheen WH, Truong SV, Brown EF, Beasley BW, Gajewski BJ. Incidence of venous thromboembolic events among nursing home residents. J Gen Intern Med 2003; 18:934–936.
- Kniffin WD, Baron JA, Barrett J, Birkmeyer JD, Anderson FA. The epidemiology of diagnosed pulmonary embolism and deep venous thrombosis in the elderly. Arch Intern Med 1994; 154:861–866.
- Bounameaux H. Integrating pharmacologic and mechanical prophylaxis of venous thromboembolism. Thromb Haemost 1999; 82:931–937.
- Gross JS, Neufeld RR, Libow LS, Gerber I, Rodstein M. Autopsy study of the elderly institutionalized patient. Review of 234 autopsies. Arch Intern Med 1988; 148:173–176.
- Spyropoulos AC, Merli G. Management of venous thromboembolism in the elderly. Drugs Aging 2006; 23:651–671.
- Punukollu H, Khan IA, Punukollu G, Gowda RM, Mendoza C, Sacchi TJ. Acute pulmonary embolism in elderly: clinical characteristics and outcome. Int J Cardiol 2005; 99:213–216.
- Douketis JD. Prevention of venous thromboembolism in hospitalized medical patients: addressing some practical questions. Curr Opin Pulm Med 2008; 14:381–388.
- Cohen AT, Tapson VF, Bergmann JF, et al; ENDORSE Investigators. Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross-sectional study. Lancet 2008; 371:387–394.
- Kahn SR, Panju A, Geerts W, et al; CURVE study investigators. Multicenter evaluation of the use of venous thromboembolism prophylaxis in acutely ill medical patients in Canada. Thromb Res 2007; 119:145–155.
- Haas S, Spyropoulos AC. Primary prevention of venous thromboembolism in long-term care: identifying and managing the risk. Clin Appl Thromb Hemost 2008; 14:149–158.
- Geerts WH, Bergqvist D, Pineo GF, et al; American College of Chest Physicians. Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest 2008; 133( suppl 6):381S–453S.
- Francis CW. Clinical practice. Prophylaxis for thromboembolism in hospitalized medical patients. N Engl J Med 2007; 356:1438–1444.
- Douketis J, Cook D, Meade M, et al; Canadian Critical Care Trials Group. Prophylaxis against deep vein thrombosis in critically ill patients with severe renal insufficiency with the low-molecular-weight heparin dalteparin: an assessment of safety and pharmacodynamics: the DIRECT study. Arch Intern Med 2008; 168:1805–1812.
- Lobo B, Finch C, Howard A, Minhas S. Fondaparinux for the treatment of patients with acute heparin-induced thrombocytopenia. Thromb Haemost 2008; 99:208–214.
- Spinler SA. New concepts in heparin-induced thrombocytopenia: diagnosis and management. J Thromb Thrombolysis 2006; 21:17–21.
- Hull RD, Schellong SM, Tapson VF, et al. Extended-duration thromboprophylaxis in acutely ill medical patients with recent reduced mobility: methodology for the EXCLAIM study. J Thromb Thrombolysis 2006; 22:31–38.
- Dennis M, Sandercock PA, Reid J, et al; CLOTS Trials Collaboration Effectiveness of thigh-length graduated compression stockings to reduce the risk of deep vein thrombosis after stroke (CLOTS trial 1): a multicentre, randomised controlled trial. Lancet 2009; 373:1958–1965.
- Weill-Engerer S, Meaume S, Lahlou A, et al. Risk factors for deep vein thrombosis in inpatients aged 65 and older: a case-control multicenter study. J Am Geriatr Soc 2004; 52:1299–1304.
- Dentali F, Douketis JD, Gianni M, Lim W, Crowther MA. Meta-analysis: anticoagulant prophylaxis to prevent symptomatic venous thromboembolism in hospitalized medical patients. Ann Intern Med 2007; 146:278–288.
- Douketis JD, Arneklev K, Goldhaber SZ, Spandorfer J, Halperin F, Horrow J. Comparison of bleeding in patients with nonvalvular atrial fibrillation treated with ximelagatran or warfarin: assessment of incidence, case-fatality rate, time course and sites of bleeding, and risk factors for bleeding. Arch Intern Med 2006; 166:853–859.
- Linkins LA, Choi PT, Douketis JD. Clinical impact of bleeding in patients taking oral anticoagulant therapy for venous thromboembolism: a meta-analysis. Ann Intern Med 2003; 139:893–900.
- Lloyd NS, Douketis JD, Moinuddin I, Lim W, Crowther MA. Anticoagulant prophylaxis to prevent asymptomatic deep vein thrombosis in hospitalized medical patients: a systematic review and meta-analysis. J Thromb Haemost 2008; 6:405–414.
- Swedko PJ, Clark HD, Paramsothy K, Akbari A. Serum creatinine is an inadequate screening test for renal failure in elderly patients. Arch Intern Med 2003; 163:356–360.
- Martel N, Lee J, Wells PS. Risk for heparin-induced thrombocytopenia with unfractionated and low-molecular-weight heparin thromboprophylaxis: a meta-analysis. Blood 2005; 106:2710–2715.
- Stein PD, Hull RD, Matta F, Yaekoub AY, Liang J. Incidence of thrombocytopenia in hospitalized patients with venous thromboembolism. Am J Med 2009; 122:919–930.
Randomized trials that included more than 20,000 medical patients have shown that anticoagulant therapy is safe and effective in preventing venous thromboembolism (VTE), ie, deep vein thrombosis and pulmonary embolism.
However, these trials were done in hospitalized patients, who typically had an acute medical illness and who, if eligible, received a short (7- to 10-day) course of anticoagulant prophylaxis.
Little attention has been given to VTE prophylaxis in residents of long-term care facilities. These patients have risk profiles similar to those of hospitalized medical patients. Some of them may have been transferred from an acute care hospital. In addition, most are elderly, and many have reduced mobility and are at risk for illnesses such as stroke and cardiorespiratory insufficiency, which increase the risk of VTE.
VTE in residents of long-term care facilities is a growing concern. By some estimates, by the year 2030 more than 20% of the US population (70.2 million people) will be over 65 years of age.1 Of those who reached age 65 in 1990, an estimated 43% will enter a nursing home at least once before they die—32% for 3 months, 24% for at least a year, and 9% for at least 5 years.2
Against this background, the objectives of this review are to consider:
- The scope of the problem of VTE in long-term care residents
- Why VTE prophylaxis is often overlooked in medical patients
- Evidence—or lack of evidence—for the safety and efficacy of VTE prophylaxis in long-term care residents and other medical patients
- Available options for VTE prophylaxis
- Which long-term care residents should or should not be considered for prophylaxis.
THE TRUE SCOPE OF THE PROBLEM IS UNKNOWN
The incidence of acute VTE among nursing home residents is reported to be 1.3 events per 100 person-years.3 About 8% of cases of pulmonary embolism and 10% of cases of deep venous thrombosis in the elderly are in nursing home residents.4
However, only 20% of patients with VTE have typical symptoms such as leg pain and swelling or acute dyspnea and chest pain, while 80% have no symptoms.5
Furthermore, deep venous thrombosis is more likely to be clinically silent in patients whose mobility is impaired, such as nursing home residents, as the symptoms arising from obstruction of venous flow are more pronounced with walking.
Pulmonary embolism is also underdiagnosed in this group. An autopsy study of 234 nursing home residents found undiagnosed pulmonary embolism to be the cause of death in 8%, and 40% of cases of pulmonary embolism were not suspected before the patient died.6 Yet pulmonary embolism has a higher case-fatality rate in the elderly than in younger patients, particularly when elderly patients have comorbidities.7
A reason why the diagnosis is so often missed is that pulmonary embolism can present atypically in the elderly, with syncope being more common and tachycardia being less common than in younger patients.8
Since so many cases of VTE are clinically silent and most long-term care residents who die do not undergo autopsy, the true scope of VTE as a clinical problem in these patients is unknown. Consequently, the best way to diagnose, prevent, and treat VTE is also unclear.
WHY IS VTE PREVENTION SO OFTEN OVERLOOKED IN MEDICAL PATIENTS?
In general, nonsurgical patients receive suboptimal thromboprophylaxis. National and international chart audits and cross-sectional studies show that only 16% to 33% of hospitalized medical patients at risk for VTE receive appropriate anticoagulant prophylaxis.9 Though no audits in long-term care facilities have been published, the rate of appropriate prophylaxis is likely comparable to or possibly less than that in medical patients in the hospital. In contrast, in surgical patients the rate is much higher—up to 90%.10,11
Why is VTE prophylaxis so underused in medical patients?
One reason is that we do not really know the baseline risk of VTE in medical patients, particularly in those with chronic illness who require long-term care.12 This is relevant because, in the absence of data about patients’ baseline risk, anticoagulant prophylaxis should be ordered selectively, as it poses known risks of bleeding. The risk is greater in elderly people with comorbidities, as are the associated costs.
In addition, relatively few studies have assessed thromboprophylaxis in medical patients, especially in residents of long-term care facilities.
Another reason is that we lack practice guidelines for patients who need long-term care. The well-accepted guidelines from the American College of Chest Physicians (ACCP) cite advanced age and immobility as risk factors for VTE and strongly recommend prophylaxis in acutely ill medical patients who have limited mobility and an additional risk factor such as infection or cancer.13 Though elderly residents of long-term care facilities may share some of these risk factors, the ACCP guidelines make no specific recommendations for this group.
The attitudes of health care professionals may also pose a barrier. Lloyd et al (unpublished data, 2009) surveyed 1,601 health care professionals in Ontario, Canada, in 2007, to assess potential barriers to anticoagulant prophylaxis in hospitalized medical patients. Respondents cited concerns about the risk of bleeding from anticoagulants, lack of clear indications and contraindications for anticoagulant prophylaxis, and lack of time to consider VTE prophylaxis in every patient. (They did not, however, cite disagreement with guidelines or patient discomfort from subcutaneous anticoagulant injections as barriers.) It is reasonable to assume that these attitudes may also pose a problem in long-term care residents.
Finally, no randomized trials have evaluated the efficacy and safety of anticoagulant drugs or mechanical methods of prophylaxis in long-term care residents. Studies have shown that a short course (7–10 days) of an anticoagulant drug effectively prevents VTE in acutely ill patients, but the efficacy of an extended course in patients with chronic illness who require long-term care is not clear. Therefore, recommendations about thromboprophylaxis in long-term care residents should be made with the caveat that they are based on indirect evidence from other patient groups. This is a considerable limitation.
OPTIONS FOR THROMBOPROPHYLAXIS IN LONG-TERM CARE RESIDENTS
Options for thromboprophylaxis fall into two broad categories: anticoagulant drugs and mechanical devices.
Anticoagulant prophylactic drugs
These agents have been assessed in randomized trials in surgical or acutely ill medical patients, although fondaparinux and tinzaparin are not approved for use in medical patients. Furthermore, none of them has been evaluated in residents of long-term care facilities.
The choice of anticoagulant for prophylaxis is determined largely by clinical factors.
Low-molecular-weight heparins are popular both in and out of the hospital because they have predictable pharmacokinetic properties, they come in convenient prefilled syringes, and they can be given once daily. However, some of them may bioaccumulate in patients with impaired renal function, as they are cleared primarily by the kidney.
Unfractionated heparin is likely to be safer in patients with severe renal insufficiency (creatinine clearance < 30 mL/min), as it is cleared via nonrenal mechanisms.
However, a recent single-arm trial of dalteparin 5,000 IU once daily in critically ill patients with severe renal insufficiency found no evidence of an excessive anticoagulant effect or of drug bioaccumulation.15 Dalteparin may thus be an alternative to unfractionated heparin in medical patients with impaired renal function.
Fondaparinux, a newer anticoagulant, is also given once daily. It is the anticoagulant of choice in patients who have had heparin-induced thrombocytopenia because it is not derived from heparin and likely does not cross-react with heparin-induced thrombocytopenia antibodies.16,17
Limited data on benefit of prophylactic anticoagulant drugs
As mentioned, the trials that confirmed the efficacy and safety of anticoagulant prophylaxis were in surgical patients and hospitalized medical patients, not elderly long-term care residents. The poor evidence for anticoagulant prophylaxis in these patients may be strengthened if extended-duration, out-of-hospital prophylaxis were shown to be effective in medical patients. Long-term care residents could more reasonably be compared with medical patients discharged home with a chronic or resolving illness than with those who are hospitalized.
There is some evidence, although with caveats, that extended anticoagulant prophylaxis, started after an acute illness has resolved, confers a benefit. A recent randomized trial compared extended-duration and short-duration prophylaxis (5 weeks vs 10 days) with enoxaparin 40 mg once daily in 4,726 medical patients with impaired mobility.18 The risk of any VTE event was 44% lower with extended-duration prophylaxis (2.8% vs 4.9%; P = .001) and the risk of symptomatic VTE was 73% lower (0.3% vs 1.1%; P = .004), and this benefit persisted 2 months after treatment was stopped (3.0% vs 5.2%; P = .0015). However, extended treatment conferred a fourfold higher risk of major bleeding (0.6% vs 0.15%; P = .019).
These findings should also be considered in terms of absolute benefit and harm. Treating 1,000 patients for 5 weeks instead of 10 days would prevent eight episodes of symptomatic VTE (absolute risk reduction = 0.8%, number needed to treat = 125) at the cost of four to five episodes of major bleeding (absolute risk increase = 0.45%, number needed to harm = 222). This is a modest net therapeutic benefit.
The therapeutic benefit would be greater if we consider all episodes of VTE, both symptomatic and asymptomatic. Treating 1,000 patients for 5 weeks would prevent 20 episodes of symptomatic or asymptomatic VTE (absolute risk reduction = 2.1%, number needed to treat = 48). However, the clinical importance of asymptomatic VTE is questionable.
Given these considerations, if extended-duration anticoagulant prophylaxis is considered, it should be for patients at highest risk to optimize both its net therapeutic benefits and its cost-effectiveness.
Mechanical prophylaxis
Mechanical thromboprophylactic devices—graduated or elastic compression stockings and intermittent pneumatic compression devices—are effective when used by themselves in surgical patients.13 However, in a randomized controlled trial in patients with ischemic stroke, the rate of VTE was 10.0% with graduated compression stockings in addition to “usual care VTE prophylaxis” vs 10.5% with usual care alone, and patients in the stocking group had a fourfold higher risk of developing skin breaks, ulcers, blisters, or necrosis (5% vs 1%; odds ratio 4.18; 95% CI 2.4–7.3).19 Furthermore, improperly fitted stockings, especially those that are thigh-length, can be uncomfortable to wear and difficult to apply.
Overall, the role of mechanical thromboprophylaxis in long-term care facilities is not clear. If it is considered, there should be a compelling reason to use it—for example, for patients at high risk in whom anticoagulants are contraindicated because of ongoing bleeding or a higher risk of bleeding (eg, recent gastrointestinal bleeding, hemorrhagic stroke, coagulopathy, or thrombocytopenia). Furthermore, if stockings are used, they should be properly fitted and routinely monitored for adverse effects, since elderly patients are likely to be most susceptible to skin breakdown.
WHICH LONG-TERM CARE RESIDENTS SHOULD RECEIVE VTE PROPHYLAXIS?
Old age and immobility are not the only risk factors
The current ACCP guidelines suggest considering thromboprophylaxis for hospitalized medical patients over age 75 who cannot walk without assistance.13 However, we lack evidence to suggest a similar strategy in long-term care residents.
The ACCP guidelines are based on data on risk. Nearly 25% of elderly patients with confirmed pulmonary embolism had been immobile prior to their diagnosis.8 In addition, prolonged bed rest (> 14 days) has been reported to be the strongest independent risk factor for symptomatic deep venous thrombosis, increasing the risk more than fivefold.20 Advanced age is also considered a risk factor for VTE, as risk starts to increase at age 40 and doubles each decade of life thereafter.18
No study has assessed the impact of these factors on the risk of VTE in long-term care residents. Since most of such patients are elderly and have impaired mobility, we believe a more selective approach should be used in assigning VTE risk status, one that does not use advanced age and immobility as the only criteria for starting thromboprophylaxis.
Residents of long-term care facilities may be immobile because of underlying illness or disability, such as cognitive impairment, sensory impairment (eg, poor access to corrective lenses and hearing aids), or poor access to assist devices (eg, walkers, canes). In addition, iatrogenic factors that decrease mobility such as indwelling bladder catheters and physical restraints are also common in such patients.
Efforts to improve mobility should be encouraged. However, we recommend that thromboprophylaxis be considered only in patients who have both impaired mobility and an intercurrent acute medical illness such as an acute infection or acute inflammatory disease.13
A related issue is the difference between long-term care residents with a chronic but stable disease and those with acute disease. Patients with acute exacerbations of congestive heart failure or chronic obstructive lung disease may be considered for thromboprophylaxis, as they become more comparable to acutely ill medical patients in whom clinical trials have shown the effectiveness of anticoagulant prophylaxis. On the other hand, patients with these diseases who remain stable may not need prophylaxis.
This approach avoids giving long-term anticoagulant prophylaxis to patients who have irreversible diseases and limits the use of these drugs and devices to higher-risk periods.
Consider thromboprophylaxis if…
- An acute exacerbation of congestive heart failure or chronic obstructive pulmonary disease
- Acute infection (eg, urosepsis, pneumonia, cellulitis, infectious diarrhea)
- An acute exacerbation of an inflammatory disease (eg, rheumatoid arthritis)
- Active cancer (eg, patient receiving radiation therapy or chemotherapy)
- Immobility and prior VTE.
Do not routinely consider prophylaxis if…
We also believe patients should not be routinely considered for anticoagulant VTE prophylaxis if they have:
- Chronic but stable cardiorespiratory disease
- Chronic but stable infectious or inflammatory disease
- Terminal cancer with very limited life expectancy
- Any contraindication to anticoagulants (eg, active bleeding, recent bleeding, coagulopathy, thrombocytopenia).
ANTICOAGULANT PROPHYLAXIS POSES RISKS IN LONG-TERM CARE RESIDENTS
Bleeding is the principal risk
The incidence of clinically important bleeding associated with anticoagulant prophylaxis is 0.2% to 5.6%, and the risk of fatal bleeding is 0.02% to 0.5%.21–24
As no randomized trial has examined anticoagulant prophylaxis in elderly long-term care residents, their bleeding risk with this therapy is unclear. However, older patients are likely to be at higher risk than younger patients because they have more comorbidities, take more drugs that could interact with heparin and potentiate bleeding, and have fragile skin, predisposing to injury from subcutaneous injections.
Also, renal function tends to decline with age. In a retrospective study of 854 outpatients over age 65, 29% had moderate renal insufficiency (creatinine clearance 30–50 mL/min), and 6% had severe renal insufficiency (creatinine clearance < 30 mL/min).25 Recent evidence suggests that some low-molecular-weight heparins (dalteparin and tinzaparin) do not bioaccumulate in patients with impaired renal function. However, enoxaparin and fondaparinux should be used with caution in patients with moderate to severe renal impairment.
Though much attention has recently been paid to increasing anticoagulant doses if the patient is obese, residents of long-term care facilities are more likely to be underweight. Dose adjustment should be considered when a low-molecular-weight heparin or fondaparinux is given to patients weighing less than 50 kg.
Heparin-induced thrombocytopenia
The other major risk of anticoagulant prophylaxis is heparin-induced thrombocytopenia, an infrequent but life-threatening complication caused by the formation of antibodies to the heparin-derived anticoagulant and a platelet surface antigen. It is associated with moderate thrombocytopenia and an incidence of venous or arterial thrombosis that is over 50%.26
No study has assessed the incidence of heparin-induced thrombocytopenia in long-term care residents. A meta-analysis reported that the risk with anticoagulant prophylaxis was 1.6% with unfractionated heparin (95% confidence interval [CI] 1.2%–2.1%) and 0.6% with low-molecular-weight heparin (95% CI 0.4%–0.9%), and that this risk increased with the duration of prophylaxis.27 If anticoagulant prophylaxis were given to all long-term care residents for extended durations (eg, for the duration of reduced mobility), the incidence and prevalence of heparin-induced thrombocytopenia would likely become a major concern.
Whenever anticoagulant prophylaxis is considered, the risks of both thrombosis and bleeding should be considered. Patients who are receiving anticoagulant prophylaxis should also be monitored for bleeding and heparin-induced thrombocytopenia. This is particularly true in long-term care residents, in whom the risks and benefits of anticoagulant prophylaxis are extrapolated from data from other populations.
MORE RESEARCH IS NEEDED
To date, we lack audits of thromboprophylaxis, clinical practice guidelines, and clear indications and contraindications for anticoagulant prophylaxis in long-term care residents. In the absence of such data, extrapolating the efficacy and safety of thromboprophylaxis from hospitalized patients to long-term care residents is difficult.
Clearly, additional research is needed to identify which long-term care residents would benefit most from thromboprophylaxis. In the meantime, a selective approach to identifying patients who should be considered for thromboprophylaxis should be adopted.
Randomized trials that included more than 20,000 medical patients have shown that anticoagulant therapy is safe and effective in preventing venous thromboembolism (VTE), ie, deep vein thrombosis and pulmonary embolism.
However, these trials were done in hospitalized patients, who typically had an acute medical illness and who, if eligible, received a short (7- to 10-day) course of anticoagulant prophylaxis.
Little attention has been given to VTE prophylaxis in residents of long-term care facilities. These patients have risk profiles similar to those of hospitalized medical patients. Some of them may have been transferred from an acute care hospital. In addition, most are elderly, and many have reduced mobility and are at risk for illnesses such as stroke and cardiorespiratory insufficiency, which increase the risk of VTE.
VTE in residents of long-term care facilities is a growing concern. By some estimates, by the year 2030 more than 20% of the US population (70.2 million people) will be over 65 years of age.1 Of those who reached age 65 in 1990, an estimated 43% will enter a nursing home at least once before they die—32% for 3 months, 24% for at least a year, and 9% for at least 5 years.2
Against this background, the objectives of this review are to consider:
- The scope of the problem of VTE in long-term care residents
- Why VTE prophylaxis is often overlooked in medical patients
- Evidence—or lack of evidence—for the safety and efficacy of VTE prophylaxis in long-term care residents and other medical patients
- Available options for VTE prophylaxis
- Which long-term care residents should or should not be considered for prophylaxis.
THE TRUE SCOPE OF THE PROBLEM IS UNKNOWN
The incidence of acute VTE among nursing home residents is reported to be 1.3 events per 100 person-years.3 About 8% of cases of pulmonary embolism and 10% of cases of deep venous thrombosis in the elderly are in nursing home residents.4
However, only 20% of patients with VTE have typical symptoms such as leg pain and swelling or acute dyspnea and chest pain, while 80% have no symptoms.5
Furthermore, deep venous thrombosis is more likely to be clinically silent in patients whose mobility is impaired, such as nursing home residents, as the symptoms arising from obstruction of venous flow are more pronounced with walking.
Pulmonary embolism is also underdiagnosed in this group. An autopsy study of 234 nursing home residents found undiagnosed pulmonary embolism to be the cause of death in 8%, and 40% of cases of pulmonary embolism were not suspected before the patient died.6 Yet pulmonary embolism has a higher case-fatality rate in the elderly than in younger patients, particularly when elderly patients have comorbidities.7
A reason why the diagnosis is so often missed is that pulmonary embolism can present atypically in the elderly, with syncope being more common and tachycardia being less common than in younger patients.8
Since so many cases of VTE are clinically silent and most long-term care residents who die do not undergo autopsy, the true scope of VTE as a clinical problem in these patients is unknown. Consequently, the best way to diagnose, prevent, and treat VTE is also unclear.
WHY IS VTE PREVENTION SO OFTEN OVERLOOKED IN MEDICAL PATIENTS?
In general, nonsurgical patients receive suboptimal thromboprophylaxis. National and international chart audits and cross-sectional studies show that only 16% to 33% of hospitalized medical patients at risk for VTE receive appropriate anticoagulant prophylaxis.9 Though no audits in long-term care facilities have been published, the rate of appropriate prophylaxis is likely comparable to or possibly less than that in medical patients in the hospital. In contrast, in surgical patients the rate is much higher—up to 90%.10,11
Why is VTE prophylaxis so underused in medical patients?
One reason is that we do not really know the baseline risk of VTE in medical patients, particularly in those with chronic illness who require long-term care.12 This is relevant because, in the absence of data about patients’ baseline risk, anticoagulant prophylaxis should be ordered selectively, as it poses known risks of bleeding. The risk is greater in elderly people with comorbidities, as are the associated costs.
In addition, relatively few studies have assessed thromboprophylaxis in medical patients, especially in residents of long-term care facilities.
Another reason is that we lack practice guidelines for patients who need long-term care. The well-accepted guidelines from the American College of Chest Physicians (ACCP) cite advanced age and immobility as risk factors for VTE and strongly recommend prophylaxis in acutely ill medical patients who have limited mobility and an additional risk factor such as infection or cancer.13 Though elderly residents of long-term care facilities may share some of these risk factors, the ACCP guidelines make no specific recommendations for this group.
The attitudes of health care professionals may also pose a barrier. Lloyd et al (unpublished data, 2009) surveyed 1,601 health care professionals in Ontario, Canada, in 2007, to assess potential barriers to anticoagulant prophylaxis in hospitalized medical patients. Respondents cited concerns about the risk of bleeding from anticoagulants, lack of clear indications and contraindications for anticoagulant prophylaxis, and lack of time to consider VTE prophylaxis in every patient. (They did not, however, cite disagreement with guidelines or patient discomfort from subcutaneous anticoagulant injections as barriers.) It is reasonable to assume that these attitudes may also pose a problem in long-term care residents.
Finally, no randomized trials have evaluated the efficacy and safety of anticoagulant drugs or mechanical methods of prophylaxis in long-term care residents. Studies have shown that a short course (7–10 days) of an anticoagulant drug effectively prevents VTE in acutely ill patients, but the efficacy of an extended course in patients with chronic illness who require long-term care is not clear. Therefore, recommendations about thromboprophylaxis in long-term care residents should be made with the caveat that they are based on indirect evidence from other patient groups. This is a considerable limitation.
OPTIONS FOR THROMBOPROPHYLAXIS IN LONG-TERM CARE RESIDENTS
Options for thromboprophylaxis fall into two broad categories: anticoagulant drugs and mechanical devices.
Anticoagulant prophylactic drugs
These agents have been assessed in randomized trials in surgical or acutely ill medical patients, although fondaparinux and tinzaparin are not approved for use in medical patients. Furthermore, none of them has been evaluated in residents of long-term care facilities.
The choice of anticoagulant for prophylaxis is determined largely by clinical factors.
Low-molecular-weight heparins are popular both in and out of the hospital because they have predictable pharmacokinetic properties, they come in convenient prefilled syringes, and they can be given once daily. However, some of them may bioaccumulate in patients with impaired renal function, as they are cleared primarily by the kidney.
Unfractionated heparin is likely to be safer in patients with severe renal insufficiency (creatinine clearance < 30 mL/min), as it is cleared via nonrenal mechanisms.
However, a recent single-arm trial of dalteparin 5,000 IU once daily in critically ill patients with severe renal insufficiency found no evidence of an excessive anticoagulant effect or of drug bioaccumulation.15 Dalteparin may thus be an alternative to unfractionated heparin in medical patients with impaired renal function.
Fondaparinux, a newer anticoagulant, is also given once daily. It is the anticoagulant of choice in patients who have had heparin-induced thrombocytopenia because it is not derived from heparin and likely does not cross-react with heparin-induced thrombocytopenia antibodies.16,17
Limited data on benefit of prophylactic anticoagulant drugs
As mentioned, the trials that confirmed the efficacy and safety of anticoagulant prophylaxis were in surgical patients and hospitalized medical patients, not elderly long-term care residents. The poor evidence for anticoagulant prophylaxis in these patients may be strengthened if extended-duration, out-of-hospital prophylaxis were shown to be effective in medical patients. Long-term care residents could more reasonably be compared with medical patients discharged home with a chronic or resolving illness than with those who are hospitalized.
There is some evidence, although with caveats, that extended anticoagulant prophylaxis, started after an acute illness has resolved, confers a benefit. A recent randomized trial compared extended-duration and short-duration prophylaxis (5 weeks vs 10 days) with enoxaparin 40 mg once daily in 4,726 medical patients with impaired mobility.18 The risk of any VTE event was 44% lower with extended-duration prophylaxis (2.8% vs 4.9%; P = .001) and the risk of symptomatic VTE was 73% lower (0.3% vs 1.1%; P = .004), and this benefit persisted 2 months after treatment was stopped (3.0% vs 5.2%; P = .0015). However, extended treatment conferred a fourfold higher risk of major bleeding (0.6% vs 0.15%; P = .019).
These findings should also be considered in terms of absolute benefit and harm. Treating 1,000 patients for 5 weeks instead of 10 days would prevent eight episodes of symptomatic VTE (absolute risk reduction = 0.8%, number needed to treat = 125) at the cost of four to five episodes of major bleeding (absolute risk increase = 0.45%, number needed to harm = 222). This is a modest net therapeutic benefit.
The therapeutic benefit would be greater if we consider all episodes of VTE, both symptomatic and asymptomatic. Treating 1,000 patients for 5 weeks would prevent 20 episodes of symptomatic or asymptomatic VTE (absolute risk reduction = 2.1%, number needed to treat = 48). However, the clinical importance of asymptomatic VTE is questionable.
Given these considerations, if extended-duration anticoagulant prophylaxis is considered, it should be for patients at highest risk to optimize both its net therapeutic benefits and its cost-effectiveness.
Mechanical prophylaxis
Mechanical thromboprophylactic devices—graduated or elastic compression stockings and intermittent pneumatic compression devices—are effective when used by themselves in surgical patients.13 However, in a randomized controlled trial in patients with ischemic stroke, the rate of VTE was 10.0% with graduated compression stockings in addition to “usual care VTE prophylaxis” vs 10.5% with usual care alone, and patients in the stocking group had a fourfold higher risk of developing skin breaks, ulcers, blisters, or necrosis (5% vs 1%; odds ratio 4.18; 95% CI 2.4–7.3).19 Furthermore, improperly fitted stockings, especially those that are thigh-length, can be uncomfortable to wear and difficult to apply.
Overall, the role of mechanical thromboprophylaxis in long-term care facilities is not clear. If it is considered, there should be a compelling reason to use it—for example, for patients at high risk in whom anticoagulants are contraindicated because of ongoing bleeding or a higher risk of bleeding (eg, recent gastrointestinal bleeding, hemorrhagic stroke, coagulopathy, or thrombocytopenia). Furthermore, if stockings are used, they should be properly fitted and routinely monitored for adverse effects, since elderly patients are likely to be most susceptible to skin breakdown.
WHICH LONG-TERM CARE RESIDENTS SHOULD RECEIVE VTE PROPHYLAXIS?
Old age and immobility are not the only risk factors
The current ACCP guidelines suggest considering thromboprophylaxis for hospitalized medical patients over age 75 who cannot walk without assistance.13 However, we lack evidence to suggest a similar strategy in long-term care residents.
The ACCP guidelines are based on data on risk. Nearly 25% of elderly patients with confirmed pulmonary embolism had been immobile prior to their diagnosis.8 In addition, prolonged bed rest (> 14 days) has been reported to be the strongest independent risk factor for symptomatic deep venous thrombosis, increasing the risk more than fivefold.20 Advanced age is also considered a risk factor for VTE, as risk starts to increase at age 40 and doubles each decade of life thereafter.18
No study has assessed the impact of these factors on the risk of VTE in long-term care residents. Since most of such patients are elderly and have impaired mobility, we believe a more selective approach should be used in assigning VTE risk status, one that does not use advanced age and immobility as the only criteria for starting thromboprophylaxis.
Residents of long-term care facilities may be immobile because of underlying illness or disability, such as cognitive impairment, sensory impairment (eg, poor access to corrective lenses and hearing aids), or poor access to assist devices (eg, walkers, canes). In addition, iatrogenic factors that decrease mobility such as indwelling bladder catheters and physical restraints are also common in such patients.
Efforts to improve mobility should be encouraged. However, we recommend that thromboprophylaxis be considered only in patients who have both impaired mobility and an intercurrent acute medical illness such as an acute infection or acute inflammatory disease.13
A related issue is the difference between long-term care residents with a chronic but stable disease and those with acute disease. Patients with acute exacerbations of congestive heart failure or chronic obstructive lung disease may be considered for thromboprophylaxis, as they become more comparable to acutely ill medical patients in whom clinical trials have shown the effectiveness of anticoagulant prophylaxis. On the other hand, patients with these diseases who remain stable may not need prophylaxis.
This approach avoids giving long-term anticoagulant prophylaxis to patients who have irreversible diseases and limits the use of these drugs and devices to higher-risk periods.
Consider thromboprophylaxis if…
- An acute exacerbation of congestive heart failure or chronic obstructive pulmonary disease
- Acute infection (eg, urosepsis, pneumonia, cellulitis, infectious diarrhea)
- An acute exacerbation of an inflammatory disease (eg, rheumatoid arthritis)
- Active cancer (eg, patient receiving radiation therapy or chemotherapy)
- Immobility and prior VTE.
Do not routinely consider prophylaxis if…
We also believe patients should not be routinely considered for anticoagulant VTE prophylaxis if they have:
- Chronic but stable cardiorespiratory disease
- Chronic but stable infectious or inflammatory disease
- Terminal cancer with very limited life expectancy
- Any contraindication to anticoagulants (eg, active bleeding, recent bleeding, coagulopathy, thrombocytopenia).
ANTICOAGULANT PROPHYLAXIS POSES RISKS IN LONG-TERM CARE RESIDENTS
Bleeding is the principal risk
The incidence of clinically important bleeding associated with anticoagulant prophylaxis is 0.2% to 5.6%, and the risk of fatal bleeding is 0.02% to 0.5%.21–24
As no randomized trial has examined anticoagulant prophylaxis in elderly long-term care residents, their bleeding risk with this therapy is unclear. However, older patients are likely to be at higher risk than younger patients because they have more comorbidities, take more drugs that could interact with heparin and potentiate bleeding, and have fragile skin, predisposing to injury from subcutaneous injections.
Also, renal function tends to decline with age. In a retrospective study of 854 outpatients over age 65, 29% had moderate renal insufficiency (creatinine clearance 30–50 mL/min), and 6% had severe renal insufficiency (creatinine clearance < 30 mL/min).25 Recent evidence suggests that some low-molecular-weight heparins (dalteparin and tinzaparin) do not bioaccumulate in patients with impaired renal function. However, enoxaparin and fondaparinux should be used with caution in patients with moderate to severe renal impairment.
Though much attention has recently been paid to increasing anticoagulant doses if the patient is obese, residents of long-term care facilities are more likely to be underweight. Dose adjustment should be considered when a low-molecular-weight heparin or fondaparinux is given to patients weighing less than 50 kg.
Heparin-induced thrombocytopenia
The other major risk of anticoagulant prophylaxis is heparin-induced thrombocytopenia, an infrequent but life-threatening complication caused by the formation of antibodies to the heparin-derived anticoagulant and a platelet surface antigen. It is associated with moderate thrombocytopenia and an incidence of venous or arterial thrombosis that is over 50%.26
No study has assessed the incidence of heparin-induced thrombocytopenia in long-term care residents. A meta-analysis reported that the risk with anticoagulant prophylaxis was 1.6% with unfractionated heparin (95% confidence interval [CI] 1.2%–2.1%) and 0.6% with low-molecular-weight heparin (95% CI 0.4%–0.9%), and that this risk increased with the duration of prophylaxis.27 If anticoagulant prophylaxis were given to all long-term care residents for extended durations (eg, for the duration of reduced mobility), the incidence and prevalence of heparin-induced thrombocytopenia would likely become a major concern.
Whenever anticoagulant prophylaxis is considered, the risks of both thrombosis and bleeding should be considered. Patients who are receiving anticoagulant prophylaxis should also be monitored for bleeding and heparin-induced thrombocytopenia. This is particularly true in long-term care residents, in whom the risks and benefits of anticoagulant prophylaxis are extrapolated from data from other populations.
MORE RESEARCH IS NEEDED
To date, we lack audits of thromboprophylaxis, clinical practice guidelines, and clear indications and contraindications for anticoagulant prophylaxis in long-term care residents. In the absence of such data, extrapolating the efficacy and safety of thromboprophylaxis from hospitalized patients to long-term care residents is difficult.
Clearly, additional research is needed to identify which long-term care residents would benefit most from thromboprophylaxis. In the meantime, a selective approach to identifying patients who should be considered for thromboprophylaxis should be adopted.
- Cornman JM. Questions for societies with “third age” populations. The Extension-of-Life Working Group, The Gerontological Society of America. Acad Med 1997; 72:856–862.
- Kemper P, Murtaugh CM. Lifetime use of nursing home care. N Engl J Med 1991; 324:595–600.
- Gomes JP, Shaheen WH, Truong SV, Brown EF, Beasley BW, Gajewski BJ. Incidence of venous thromboembolic events among nursing home residents. J Gen Intern Med 2003; 18:934–936.
- Kniffin WD, Baron JA, Barrett J, Birkmeyer JD, Anderson FA. The epidemiology of diagnosed pulmonary embolism and deep venous thrombosis in the elderly. Arch Intern Med 1994; 154:861–866.
- Bounameaux H. Integrating pharmacologic and mechanical prophylaxis of venous thromboembolism. Thromb Haemost 1999; 82:931–937.
- Gross JS, Neufeld RR, Libow LS, Gerber I, Rodstein M. Autopsy study of the elderly institutionalized patient. Review of 234 autopsies. Arch Intern Med 1988; 148:173–176.
- Spyropoulos AC, Merli G. Management of venous thromboembolism in the elderly. Drugs Aging 2006; 23:651–671.
- Punukollu H, Khan IA, Punukollu G, Gowda RM, Mendoza C, Sacchi TJ. Acute pulmonary embolism in elderly: clinical characteristics and outcome. Int J Cardiol 2005; 99:213–216.
- Douketis JD. Prevention of venous thromboembolism in hospitalized medical patients: addressing some practical questions. Curr Opin Pulm Med 2008; 14:381–388.
- Cohen AT, Tapson VF, Bergmann JF, et al; ENDORSE Investigators. Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross-sectional study. Lancet 2008; 371:387–394.
- Kahn SR, Panju A, Geerts W, et al; CURVE study investigators. Multicenter evaluation of the use of venous thromboembolism prophylaxis in acutely ill medical patients in Canada. Thromb Res 2007; 119:145–155.
- Haas S, Spyropoulos AC. Primary prevention of venous thromboembolism in long-term care: identifying and managing the risk. Clin Appl Thromb Hemost 2008; 14:149–158.
- Geerts WH, Bergqvist D, Pineo GF, et al; American College of Chest Physicians. Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest 2008; 133( suppl 6):381S–453S.
- Francis CW. Clinical practice. Prophylaxis for thromboembolism in hospitalized medical patients. N Engl J Med 2007; 356:1438–1444.
- Douketis J, Cook D, Meade M, et al; Canadian Critical Care Trials Group. Prophylaxis against deep vein thrombosis in critically ill patients with severe renal insufficiency with the low-molecular-weight heparin dalteparin: an assessment of safety and pharmacodynamics: the DIRECT study. Arch Intern Med 2008; 168:1805–1812.
- Lobo B, Finch C, Howard A, Minhas S. Fondaparinux for the treatment of patients with acute heparin-induced thrombocytopenia. Thromb Haemost 2008; 99:208–214.
- Spinler SA. New concepts in heparin-induced thrombocytopenia: diagnosis and management. J Thromb Thrombolysis 2006; 21:17–21.
- Hull RD, Schellong SM, Tapson VF, et al. Extended-duration thromboprophylaxis in acutely ill medical patients with recent reduced mobility: methodology for the EXCLAIM study. J Thromb Thrombolysis 2006; 22:31–38.
- Dennis M, Sandercock PA, Reid J, et al; CLOTS Trials Collaboration Effectiveness of thigh-length graduated compression stockings to reduce the risk of deep vein thrombosis after stroke (CLOTS trial 1): a multicentre, randomised controlled trial. Lancet 2009; 373:1958–1965.
- Weill-Engerer S, Meaume S, Lahlou A, et al. Risk factors for deep vein thrombosis in inpatients aged 65 and older: a case-control multicenter study. J Am Geriatr Soc 2004; 52:1299–1304.
- Dentali F, Douketis JD, Gianni M, Lim W, Crowther MA. Meta-analysis: anticoagulant prophylaxis to prevent symptomatic venous thromboembolism in hospitalized medical patients. Ann Intern Med 2007; 146:278–288.
- Douketis JD, Arneklev K, Goldhaber SZ, Spandorfer J, Halperin F, Horrow J. Comparison of bleeding in patients with nonvalvular atrial fibrillation treated with ximelagatran or warfarin: assessment of incidence, case-fatality rate, time course and sites of bleeding, and risk factors for bleeding. Arch Intern Med 2006; 166:853–859.
- Linkins LA, Choi PT, Douketis JD. Clinical impact of bleeding in patients taking oral anticoagulant therapy for venous thromboembolism: a meta-analysis. Ann Intern Med 2003; 139:893–900.
- Lloyd NS, Douketis JD, Moinuddin I, Lim W, Crowther MA. Anticoagulant prophylaxis to prevent asymptomatic deep vein thrombosis in hospitalized medical patients: a systematic review and meta-analysis. J Thromb Haemost 2008; 6:405–414.
- Swedko PJ, Clark HD, Paramsothy K, Akbari A. Serum creatinine is an inadequate screening test for renal failure in elderly patients. Arch Intern Med 2003; 163:356–360.
- Martel N, Lee J, Wells PS. Risk for heparin-induced thrombocytopenia with unfractionated and low-molecular-weight heparin thromboprophylaxis: a meta-analysis. Blood 2005; 106:2710–2715.
- Stein PD, Hull RD, Matta F, Yaekoub AY, Liang J. Incidence of thrombocytopenia in hospitalized patients with venous thromboembolism. Am J Med 2009; 122:919–930.
- Cornman JM. Questions for societies with “third age” populations. The Extension-of-Life Working Group, The Gerontological Society of America. Acad Med 1997; 72:856–862.
- Kemper P, Murtaugh CM. Lifetime use of nursing home care. N Engl J Med 1991; 324:595–600.
- Gomes JP, Shaheen WH, Truong SV, Brown EF, Beasley BW, Gajewski BJ. Incidence of venous thromboembolic events among nursing home residents. J Gen Intern Med 2003; 18:934–936.
- Kniffin WD, Baron JA, Barrett J, Birkmeyer JD, Anderson FA. The epidemiology of diagnosed pulmonary embolism and deep venous thrombosis in the elderly. Arch Intern Med 1994; 154:861–866.
- Bounameaux H. Integrating pharmacologic and mechanical prophylaxis of venous thromboembolism. Thromb Haemost 1999; 82:931–937.
- Gross JS, Neufeld RR, Libow LS, Gerber I, Rodstein M. Autopsy study of the elderly institutionalized patient. Review of 234 autopsies. Arch Intern Med 1988; 148:173–176.
- Spyropoulos AC, Merli G. Management of venous thromboembolism in the elderly. Drugs Aging 2006; 23:651–671.
- Punukollu H, Khan IA, Punukollu G, Gowda RM, Mendoza C, Sacchi TJ. Acute pulmonary embolism in elderly: clinical characteristics and outcome. Int J Cardiol 2005; 99:213–216.
- Douketis JD. Prevention of venous thromboembolism in hospitalized medical patients: addressing some practical questions. Curr Opin Pulm Med 2008; 14:381–388.
- Cohen AT, Tapson VF, Bergmann JF, et al; ENDORSE Investigators. Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross-sectional study. Lancet 2008; 371:387–394.
- Kahn SR, Panju A, Geerts W, et al; CURVE study investigators. Multicenter evaluation of the use of venous thromboembolism prophylaxis in acutely ill medical patients in Canada. Thromb Res 2007; 119:145–155.
- Haas S, Spyropoulos AC. Primary prevention of venous thromboembolism in long-term care: identifying and managing the risk. Clin Appl Thromb Hemost 2008; 14:149–158.
- Geerts WH, Bergqvist D, Pineo GF, et al; American College of Chest Physicians. Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest 2008; 133( suppl 6):381S–453S.
- Francis CW. Clinical practice. Prophylaxis for thromboembolism in hospitalized medical patients. N Engl J Med 2007; 356:1438–1444.
- Douketis J, Cook D, Meade M, et al; Canadian Critical Care Trials Group. Prophylaxis against deep vein thrombosis in critically ill patients with severe renal insufficiency with the low-molecular-weight heparin dalteparin: an assessment of safety and pharmacodynamics: the DIRECT study. Arch Intern Med 2008; 168:1805–1812.
- Lobo B, Finch C, Howard A, Minhas S. Fondaparinux for the treatment of patients with acute heparin-induced thrombocytopenia. Thromb Haemost 2008; 99:208–214.
- Spinler SA. New concepts in heparin-induced thrombocytopenia: diagnosis and management. J Thromb Thrombolysis 2006; 21:17–21.
- Hull RD, Schellong SM, Tapson VF, et al. Extended-duration thromboprophylaxis in acutely ill medical patients with recent reduced mobility: methodology for the EXCLAIM study. J Thromb Thrombolysis 2006; 22:31–38.
- Dennis M, Sandercock PA, Reid J, et al; CLOTS Trials Collaboration Effectiveness of thigh-length graduated compression stockings to reduce the risk of deep vein thrombosis after stroke (CLOTS trial 1): a multicentre, randomised controlled trial. Lancet 2009; 373:1958–1965.
- Weill-Engerer S, Meaume S, Lahlou A, et al. Risk factors for deep vein thrombosis in inpatients aged 65 and older: a case-control multicenter study. J Am Geriatr Soc 2004; 52:1299–1304.
- Dentali F, Douketis JD, Gianni M, Lim W, Crowther MA. Meta-analysis: anticoagulant prophylaxis to prevent symptomatic venous thromboembolism in hospitalized medical patients. Ann Intern Med 2007; 146:278–288.
- Douketis JD, Arneklev K, Goldhaber SZ, Spandorfer J, Halperin F, Horrow J. Comparison of bleeding in patients with nonvalvular atrial fibrillation treated with ximelagatran or warfarin: assessment of incidence, case-fatality rate, time course and sites of bleeding, and risk factors for bleeding. Arch Intern Med 2006; 166:853–859.
- Linkins LA, Choi PT, Douketis JD. Clinical impact of bleeding in patients taking oral anticoagulant therapy for venous thromboembolism: a meta-analysis. Ann Intern Med 2003; 139:893–900.
- Lloyd NS, Douketis JD, Moinuddin I, Lim W, Crowther MA. Anticoagulant prophylaxis to prevent asymptomatic deep vein thrombosis in hospitalized medical patients: a systematic review and meta-analysis. J Thromb Haemost 2008; 6:405–414.
- Swedko PJ, Clark HD, Paramsothy K, Akbari A. Serum creatinine is an inadequate screening test for renal failure in elderly patients. Arch Intern Med 2003; 163:356–360.
- Martel N, Lee J, Wells PS. Risk for heparin-induced thrombocytopenia with unfractionated and low-molecular-weight heparin thromboprophylaxis: a meta-analysis. Blood 2005; 106:2710–2715.
- Stein PD, Hull RD, Matta F, Yaekoub AY, Liang J. Incidence of thrombocytopenia in hospitalized patients with venous thromboembolism. Am J Med 2009; 122:919–930.
KEY POINTS
- Assessment of VTE risk and consideration of need for anticoagulant prophylaxis in long-term care residents are based on indirect data, derived primarily from studies of acutely ill hospitalized medical patients.
- Drugs and devices for thromboprophylaxis have been studied in medical and surgical populations, but issues of efficacy and safety are likely to also pertain to long-term care residents.
- Thromboprophylaxis should be considered for long-term care residents if they are definitely at increased risk of VTE—ie, if they have an acute exacerbation of congestive heart failure or chronic obstructive pulmonary disease; acute inflammatory disease; acute infection; active cancer; or immobility and prior VTE.