User login
Ultrasonic renal denervation passes 2-month test in uncontrolled HTN: RADIANCE II
Systolic blood pressure went down safely and consistently 2 months after renal denervation achieved by ultrasound ablation in patients with uncontrolled, mild to moderate hypertension (HTN) in a key sham-controlled test of the balloon-equipped catheter.
The BP reductions were significant almost regardless of how they were measured – at home, in the office, during the day, at night, or over 24 hours – and weren’t dependent on baseline BP levels.
The 224-patient RADIANCE II Pivotal Study follows two earlier successful sham-controlled trials that used the same renal denervation catheter in other types of patients with HTN. They were RADIANCE-HTN SOLO, which entered patients with mild to moderate HTN not taking medication, and RADIANCE-HTN TRIO, which included patients with HTN despite fixed-dose, single-pill, triple-antihypertensive therapy.
The consistent results of all three trials suggest that the ultrasound renal denervation (uRDN) technique “lowers blood pressure across the spectrum of hypertension,” concluded co–principal investigator Ajay J. Kirtane, MD, SM, Columbia University Irving Medical Center, New York–Presbyterian Hospital, when presenting RADIANCE II at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
RADIANCE II, the largest of the three studies, met its prespecified primary efficacy endpoint of change in daytime ambulatory systolic BP at 2 months by showing a significant 6.3–mm Hg greater reduction in the uRDN group, compared with the sham-control group. There were no major adverse events at 30 days in either group.
The trial was similarly successful for the secondary endpoints of change in systolic BP measured in various other settings, including over 24 hours. Reductions after uRDN averaged 5-7 mm Hg greater than in the control group.
Sparse top-line results of the RADIANCE II pivotal trial were announced in July by the study’s sponsor, ReCor Medical.
Dr. Kirtane stressed in an interview that uRDN and likely any form of HTN renal denervation therapy is not a substitute for standard management. “This is really for patients in whom you’ve made best efforts to do the traditional things – lifestyle modification, medications, all of that – and yet they’re still uncontrolled.” At that point, assuming denervation therapy is available in practice, “it would be something to potentially consider.”
As a panelist after Dr. Kirtane’s formal presentation of RADIANCE II at the conference, Naomi D. Fisher, MD, who was a RADIANCE-HTN TRIO investigator, described how the treatment’s perceived intended patient population evolved over time.
“We all began with the idea that we were going to treat patients with resistant hypertension, that was going to be the first target. We have learned that those patients are far fewer than we thought,” said Dr. Fisher, who directs the hypertension service at Brigham and Women’s Hospital, Boston.
Initial estimates were that such patients with the resistant form, “meaning they require more than three drugs to control their blood pressure,” would represent 15%-20% of patients with HTN.
“We learned from our TRIO data that if you give these patients one single combined pill, lo and behold, many of them become controlled,” she said. “There is so much nonadherence out there in the world, about 50% of our patients aren’t taking their pills. It’s a hard and true fact.”
Exclude patients who aren’t adherent and “our true resistance population becomes minuscule. So, I don’t think that’s going to be the main population” for renal denervation therapy.
More likely, she said, it would be “patients who are uncontrolled and unable to take their medications. So that is going to include nonadherence, intolerance. It’s a very large category of patients. And the priorities can be stacked in favor of those who have higher cardiovascular risk.”
RADIANCE II can show the persistence of uRDN’s BP-lowering effect only out to 2 months so far, but the effect’s durability based on the RADIANCE program’s combined experience appears to be at least 2 years, Dr. Kirtane said in an interview.
“The RADIANCE II pivotal trial is a powerful, well-designed study attesting to the efficacy of renal denervation in BP lowering,” Franz H. Messerli, MD, Swiss Cardiovascular Center, University Hospital Bern, said in an interview.
The trial “shows the well-known unpredictability of antihypertensive response. We cannot predict who responds to renal denervation and who does not, and who even has a paradoxical increase in BP,” Dr. Messerli, an international hypertension expert not associated with the trial, said in an interview.
“As long as we cannot predict the antihypertensive response to renal denervation therapy, potential synergism/antagonism with drug therapy remains an educated guess,” he said.
“Hypertension is a disease that lasts years and decades. As impressive as RADIANCE II’s 2-month snapshot is, I look forward to similar or better BP data 12 and 24 months after renal denervation,” Dr. Messerli added.
RADIANCE II entered patients with mild to moderate uncontrolled HTN, that is, a systolic BP at least 140/90 mm Hg and less than 180/120 mm Hg, who were receiving no more than two antihypertensive medications. They could have no history of cardiovascular or cerebrovascular events or uncontrolled diabetes, and their estimated glomerular filtration rate (eGFR) had to be at least 40 mL/min per 1.73 m2.
After a 4-week drug washout period, patients who were clinically stable with an ambulatory BP of at least 135/85 mm Hg and less than 170/105 mm Hg underwent CT and renal angiography. Then, the 224 patients still anatomically eligible for the procedure were randomly assigned 2:1 to uRDN or a sham-control procedure: 150 and 74 patients, respectively.
At 2 months, daytime ambulatory systolic BP on average fell 7.9 mm Hg in the uRDN group and 1.8 mm Hg in the sham-control group, for a drop that was steeper by 6.3 mm Hg (P < .0001) after uRDN.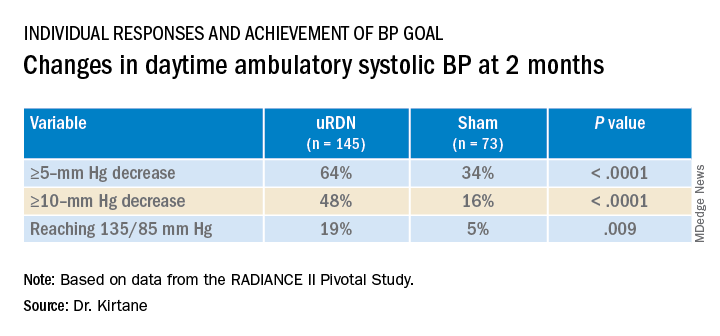
Also in the uRDN group, there was a 6.2–mm Hg larger decrease in 24-hour ambulatory systolic BP (P < .0001), a 5.8–mm Hg greater decline in nighttime ambulatory systolic BP (P < .0004), a 7.6–mm Hg steeper drop in mean home systolic BP (P < .0001), and 5.4 mm Hg more of a decrease in office-based systolic BP (P = .0035).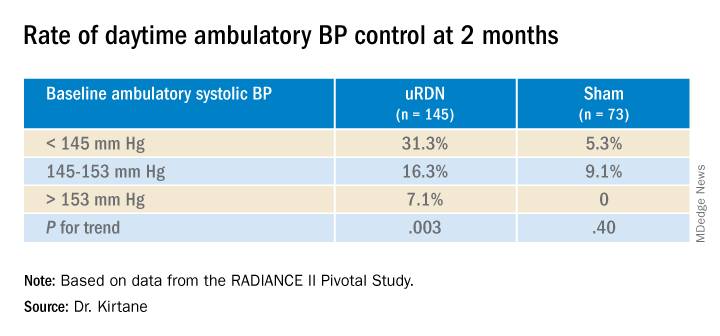
No significant differences were seen in subgroup analyses by sex, age, higher versus lower baseline systolic pressures, high versus low baseline eGFR, degree of abdominal obesity, U.S. versus European site, or whether patients entered before or during the COVID pandemic
Regulators have been accepting change in systolic BP as a surrogate for clinical endpoints in trials of antihypertensive therapy, whether pharmacologic or interventional, under consideration for approval. “That’s why safety endpoints are important to investigate” in these clinical trials, especially for invasive therapies like renal denervation, Dr. Kirtane observed.
That said, “in the longer-term follow-ups of the renal denervation therapies that are out there, including this one, there does not appear to be an appreciable decline in glomerular filtration rate, or any adverse safety signals that we see to date,” Dr. Kirtane said in an interview. “But we know that these are low-frequency events, so we have to be very vigilant, and we can’t get complacent about it.”
In RADIANCE II, there were zero adverse events within 30 days in both groups; the endpoint included death, new myocardial infarction, renal artery complications requiring invasive intervention, and hospitalization for major cardiovascular or hemodynamic-related events. Nor were there instances of new-onset renal artery stenosis greater than 70% documented by imaging at 6 months.
The ReCor uRDN catheter uses ultrasound energy to disrupt renal nerve signaling, a technology thought to deliver safer “burns,” compared with other renal denervation catheter technologies. It features an axially stabilizing balloon that transmits ultrasound energy – two to three sonications, each lasting 7 seconds, Dr. Kirtane said – outward through the arterial wall. The design is intended to ensure consistently circumferential ablation. Circulating saline within the balloon, Kirtane noted, directly cools the adjacent vessel wall to help it avoid thermal damage.
Dr. Kirtane reported receiving institutional funding from Medtronic, Boston Scientific, Abbott Vascular, Amgen, CSI, Philips, ReCor Medical, Neurotronic, Biotronik, Chiesi, Bolt Medical, Magenta Medical, Canon, SoniVie, Shockwave Medical, and Merck; consulting for IMDS; and receiving travel and meal expenses from Medtronic, Boston Scientific, Abbott Vascular, CSI, Siemens, Philips, ReCor Medical, Chiesi, OpSens, Zoll, and Regeneron. Dr. Fisher disclosed receiving honoraria or fees for consulting or serving on a speaker’s bureau for Medtronic, ReCor Medical, and Aktiia and receiving grant support or holding research contracts for Recor Medical and Aktiia. Dr. Messerli disclosed receiving honoraria from Medtronic, Menarini, Krka, and Ipca.
A version of this article first appeared on Medscape.com.
Systolic blood pressure went down safely and consistently 2 months after renal denervation achieved by ultrasound ablation in patients with uncontrolled, mild to moderate hypertension (HTN) in a key sham-controlled test of the balloon-equipped catheter.
The BP reductions were significant almost regardless of how they were measured – at home, in the office, during the day, at night, or over 24 hours – and weren’t dependent on baseline BP levels.
The 224-patient RADIANCE II Pivotal Study follows two earlier successful sham-controlled trials that used the same renal denervation catheter in other types of patients with HTN. They were RADIANCE-HTN SOLO, which entered patients with mild to moderate HTN not taking medication, and RADIANCE-HTN TRIO, which included patients with HTN despite fixed-dose, single-pill, triple-antihypertensive therapy.
The consistent results of all three trials suggest that the ultrasound renal denervation (uRDN) technique “lowers blood pressure across the spectrum of hypertension,” concluded co–principal investigator Ajay J. Kirtane, MD, SM, Columbia University Irving Medical Center, New York–Presbyterian Hospital, when presenting RADIANCE II at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
RADIANCE II, the largest of the three studies, met its prespecified primary efficacy endpoint of change in daytime ambulatory systolic BP at 2 months by showing a significant 6.3–mm Hg greater reduction in the uRDN group, compared with the sham-control group. There were no major adverse events at 30 days in either group.
The trial was similarly successful for the secondary endpoints of change in systolic BP measured in various other settings, including over 24 hours. Reductions after uRDN averaged 5-7 mm Hg greater than in the control group.
Sparse top-line results of the RADIANCE II pivotal trial were announced in July by the study’s sponsor, ReCor Medical.
Dr. Kirtane stressed in an interview that uRDN and likely any form of HTN renal denervation therapy is not a substitute for standard management. “This is really for patients in whom you’ve made best efforts to do the traditional things – lifestyle modification, medications, all of that – and yet they’re still uncontrolled.” At that point, assuming denervation therapy is available in practice, “it would be something to potentially consider.”
As a panelist after Dr. Kirtane’s formal presentation of RADIANCE II at the conference, Naomi D. Fisher, MD, who was a RADIANCE-HTN TRIO investigator, described how the treatment’s perceived intended patient population evolved over time.
“We all began with the idea that we were going to treat patients with resistant hypertension, that was going to be the first target. We have learned that those patients are far fewer than we thought,” said Dr. Fisher, who directs the hypertension service at Brigham and Women’s Hospital, Boston.
Initial estimates were that such patients with the resistant form, “meaning they require more than three drugs to control their blood pressure,” would represent 15%-20% of patients with HTN.
“We learned from our TRIO data that if you give these patients one single combined pill, lo and behold, many of them become controlled,” she said. “There is so much nonadherence out there in the world, about 50% of our patients aren’t taking their pills. It’s a hard and true fact.”
Exclude patients who aren’t adherent and “our true resistance population becomes minuscule. So, I don’t think that’s going to be the main population” for renal denervation therapy.
More likely, she said, it would be “patients who are uncontrolled and unable to take their medications. So that is going to include nonadherence, intolerance. It’s a very large category of patients. And the priorities can be stacked in favor of those who have higher cardiovascular risk.”
RADIANCE II can show the persistence of uRDN’s BP-lowering effect only out to 2 months so far, but the effect’s durability based on the RADIANCE program’s combined experience appears to be at least 2 years, Dr. Kirtane said in an interview.
“The RADIANCE II pivotal trial is a powerful, well-designed study attesting to the efficacy of renal denervation in BP lowering,” Franz H. Messerli, MD, Swiss Cardiovascular Center, University Hospital Bern, said in an interview.
The trial “shows the well-known unpredictability of antihypertensive response. We cannot predict who responds to renal denervation and who does not, and who even has a paradoxical increase in BP,” Dr. Messerli, an international hypertension expert not associated with the trial, said in an interview.
“As long as we cannot predict the antihypertensive response to renal denervation therapy, potential synergism/antagonism with drug therapy remains an educated guess,” he said.
“Hypertension is a disease that lasts years and decades. As impressive as RADIANCE II’s 2-month snapshot is, I look forward to similar or better BP data 12 and 24 months after renal denervation,” Dr. Messerli added.
RADIANCE II entered patients with mild to moderate uncontrolled HTN, that is, a systolic BP at least 140/90 mm Hg and less than 180/120 mm Hg, who were receiving no more than two antihypertensive medications. They could have no history of cardiovascular or cerebrovascular events or uncontrolled diabetes, and their estimated glomerular filtration rate (eGFR) had to be at least 40 mL/min per 1.73 m2.
After a 4-week drug washout period, patients who were clinically stable with an ambulatory BP of at least 135/85 mm Hg and less than 170/105 mm Hg underwent CT and renal angiography. Then, the 224 patients still anatomically eligible for the procedure were randomly assigned 2:1 to uRDN or a sham-control procedure: 150 and 74 patients, respectively.
At 2 months, daytime ambulatory systolic BP on average fell 7.9 mm Hg in the uRDN group and 1.8 mm Hg in the sham-control group, for a drop that was steeper by 6.3 mm Hg (P < .0001) after uRDN.
Also in the uRDN group, there was a 6.2–mm Hg larger decrease in 24-hour ambulatory systolic BP (P < .0001), a 5.8–mm Hg greater decline in nighttime ambulatory systolic BP (P < .0004), a 7.6–mm Hg steeper drop in mean home systolic BP (P < .0001), and 5.4 mm Hg more of a decrease in office-based systolic BP (P = .0035).
No significant differences were seen in subgroup analyses by sex, age, higher versus lower baseline systolic pressures, high versus low baseline eGFR, degree of abdominal obesity, U.S. versus European site, or whether patients entered before or during the COVID pandemic
Regulators have been accepting change in systolic BP as a surrogate for clinical endpoints in trials of antihypertensive therapy, whether pharmacologic or interventional, under consideration for approval. “That’s why safety endpoints are important to investigate” in these clinical trials, especially for invasive therapies like renal denervation, Dr. Kirtane observed.
That said, “in the longer-term follow-ups of the renal denervation therapies that are out there, including this one, there does not appear to be an appreciable decline in glomerular filtration rate, or any adverse safety signals that we see to date,” Dr. Kirtane said in an interview. “But we know that these are low-frequency events, so we have to be very vigilant, and we can’t get complacent about it.”
In RADIANCE II, there were zero adverse events within 30 days in both groups; the endpoint included death, new myocardial infarction, renal artery complications requiring invasive intervention, and hospitalization for major cardiovascular or hemodynamic-related events. Nor were there instances of new-onset renal artery stenosis greater than 70% documented by imaging at 6 months.
The ReCor uRDN catheter uses ultrasound energy to disrupt renal nerve signaling, a technology thought to deliver safer “burns,” compared with other renal denervation catheter technologies. It features an axially stabilizing balloon that transmits ultrasound energy – two to three sonications, each lasting 7 seconds, Dr. Kirtane said – outward through the arterial wall. The design is intended to ensure consistently circumferential ablation. Circulating saline within the balloon, Kirtane noted, directly cools the adjacent vessel wall to help it avoid thermal damage.
Dr. Kirtane reported receiving institutional funding from Medtronic, Boston Scientific, Abbott Vascular, Amgen, CSI, Philips, ReCor Medical, Neurotronic, Biotronik, Chiesi, Bolt Medical, Magenta Medical, Canon, SoniVie, Shockwave Medical, and Merck; consulting for IMDS; and receiving travel and meal expenses from Medtronic, Boston Scientific, Abbott Vascular, CSI, Siemens, Philips, ReCor Medical, Chiesi, OpSens, Zoll, and Regeneron. Dr. Fisher disclosed receiving honoraria or fees for consulting or serving on a speaker’s bureau for Medtronic, ReCor Medical, and Aktiia and receiving grant support or holding research contracts for Recor Medical and Aktiia. Dr. Messerli disclosed receiving honoraria from Medtronic, Menarini, Krka, and Ipca.
A version of this article first appeared on Medscape.com.
Systolic blood pressure went down safely and consistently 2 months after renal denervation achieved by ultrasound ablation in patients with uncontrolled, mild to moderate hypertension (HTN) in a key sham-controlled test of the balloon-equipped catheter.
The BP reductions were significant almost regardless of how they were measured – at home, in the office, during the day, at night, or over 24 hours – and weren’t dependent on baseline BP levels.
The 224-patient RADIANCE II Pivotal Study follows two earlier successful sham-controlled trials that used the same renal denervation catheter in other types of patients with HTN. They were RADIANCE-HTN SOLO, which entered patients with mild to moderate HTN not taking medication, and RADIANCE-HTN TRIO, which included patients with HTN despite fixed-dose, single-pill, triple-antihypertensive therapy.
The consistent results of all three trials suggest that the ultrasound renal denervation (uRDN) technique “lowers blood pressure across the spectrum of hypertension,” concluded co–principal investigator Ajay J. Kirtane, MD, SM, Columbia University Irving Medical Center, New York–Presbyterian Hospital, when presenting RADIANCE II at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
RADIANCE II, the largest of the three studies, met its prespecified primary efficacy endpoint of change in daytime ambulatory systolic BP at 2 months by showing a significant 6.3–mm Hg greater reduction in the uRDN group, compared with the sham-control group. There were no major adverse events at 30 days in either group.
The trial was similarly successful for the secondary endpoints of change in systolic BP measured in various other settings, including over 24 hours. Reductions after uRDN averaged 5-7 mm Hg greater than in the control group.
Sparse top-line results of the RADIANCE II pivotal trial were announced in July by the study’s sponsor, ReCor Medical.
Dr. Kirtane stressed in an interview that uRDN and likely any form of HTN renal denervation therapy is not a substitute for standard management. “This is really for patients in whom you’ve made best efforts to do the traditional things – lifestyle modification, medications, all of that – and yet they’re still uncontrolled.” At that point, assuming denervation therapy is available in practice, “it would be something to potentially consider.”
As a panelist after Dr. Kirtane’s formal presentation of RADIANCE II at the conference, Naomi D. Fisher, MD, who was a RADIANCE-HTN TRIO investigator, described how the treatment’s perceived intended patient population evolved over time.
“We all began with the idea that we were going to treat patients with resistant hypertension, that was going to be the first target. We have learned that those patients are far fewer than we thought,” said Dr. Fisher, who directs the hypertension service at Brigham and Women’s Hospital, Boston.
Initial estimates were that such patients with the resistant form, “meaning they require more than three drugs to control their blood pressure,” would represent 15%-20% of patients with HTN.
“We learned from our TRIO data that if you give these patients one single combined pill, lo and behold, many of them become controlled,” she said. “There is so much nonadherence out there in the world, about 50% of our patients aren’t taking their pills. It’s a hard and true fact.”
Exclude patients who aren’t adherent and “our true resistance population becomes minuscule. So, I don’t think that’s going to be the main population” for renal denervation therapy.
More likely, she said, it would be “patients who are uncontrolled and unable to take their medications. So that is going to include nonadherence, intolerance. It’s a very large category of patients. And the priorities can be stacked in favor of those who have higher cardiovascular risk.”
RADIANCE II can show the persistence of uRDN’s BP-lowering effect only out to 2 months so far, but the effect’s durability based on the RADIANCE program’s combined experience appears to be at least 2 years, Dr. Kirtane said in an interview.
“The RADIANCE II pivotal trial is a powerful, well-designed study attesting to the efficacy of renal denervation in BP lowering,” Franz H. Messerli, MD, Swiss Cardiovascular Center, University Hospital Bern, said in an interview.
The trial “shows the well-known unpredictability of antihypertensive response. We cannot predict who responds to renal denervation and who does not, and who even has a paradoxical increase in BP,” Dr. Messerli, an international hypertension expert not associated with the trial, said in an interview.
“As long as we cannot predict the antihypertensive response to renal denervation therapy, potential synergism/antagonism with drug therapy remains an educated guess,” he said.
“Hypertension is a disease that lasts years and decades. As impressive as RADIANCE II’s 2-month snapshot is, I look forward to similar or better BP data 12 and 24 months after renal denervation,” Dr. Messerli added.
RADIANCE II entered patients with mild to moderate uncontrolled HTN, that is, a systolic BP at least 140/90 mm Hg and less than 180/120 mm Hg, who were receiving no more than two antihypertensive medications. They could have no history of cardiovascular or cerebrovascular events or uncontrolled diabetes, and their estimated glomerular filtration rate (eGFR) had to be at least 40 mL/min per 1.73 m2.
After a 4-week drug washout period, patients who were clinically stable with an ambulatory BP of at least 135/85 mm Hg and less than 170/105 mm Hg underwent CT and renal angiography. Then, the 224 patients still anatomically eligible for the procedure were randomly assigned 2:1 to uRDN or a sham-control procedure: 150 and 74 patients, respectively.
At 2 months, daytime ambulatory systolic BP on average fell 7.9 mm Hg in the uRDN group and 1.8 mm Hg in the sham-control group, for a drop that was steeper by 6.3 mm Hg (P < .0001) after uRDN.
Also in the uRDN group, there was a 6.2–mm Hg larger decrease in 24-hour ambulatory systolic BP (P < .0001), a 5.8–mm Hg greater decline in nighttime ambulatory systolic BP (P < .0004), a 7.6–mm Hg steeper drop in mean home systolic BP (P < .0001), and 5.4 mm Hg more of a decrease in office-based systolic BP (P = .0035).
No significant differences were seen in subgroup analyses by sex, age, higher versus lower baseline systolic pressures, high versus low baseline eGFR, degree of abdominal obesity, U.S. versus European site, or whether patients entered before or during the COVID pandemic
Regulators have been accepting change in systolic BP as a surrogate for clinical endpoints in trials of antihypertensive therapy, whether pharmacologic or interventional, under consideration for approval. “That’s why safety endpoints are important to investigate” in these clinical trials, especially for invasive therapies like renal denervation, Dr. Kirtane observed.
That said, “in the longer-term follow-ups of the renal denervation therapies that are out there, including this one, there does not appear to be an appreciable decline in glomerular filtration rate, or any adverse safety signals that we see to date,” Dr. Kirtane said in an interview. “But we know that these are low-frequency events, so we have to be very vigilant, and we can’t get complacent about it.”
In RADIANCE II, there were zero adverse events within 30 days in both groups; the endpoint included death, new myocardial infarction, renal artery complications requiring invasive intervention, and hospitalization for major cardiovascular or hemodynamic-related events. Nor were there instances of new-onset renal artery stenosis greater than 70% documented by imaging at 6 months.
The ReCor uRDN catheter uses ultrasound energy to disrupt renal nerve signaling, a technology thought to deliver safer “burns,” compared with other renal denervation catheter technologies. It features an axially stabilizing balloon that transmits ultrasound energy – two to three sonications, each lasting 7 seconds, Dr. Kirtane said – outward through the arterial wall. The design is intended to ensure consistently circumferential ablation. Circulating saline within the balloon, Kirtane noted, directly cools the adjacent vessel wall to help it avoid thermal damage.
Dr. Kirtane reported receiving institutional funding from Medtronic, Boston Scientific, Abbott Vascular, Amgen, CSI, Philips, ReCor Medical, Neurotronic, Biotronik, Chiesi, Bolt Medical, Magenta Medical, Canon, SoniVie, Shockwave Medical, and Merck; consulting for IMDS; and receiving travel and meal expenses from Medtronic, Boston Scientific, Abbott Vascular, CSI, Siemens, Philips, ReCor Medical, Chiesi, OpSens, Zoll, and Regeneron. Dr. Fisher disclosed receiving honoraria or fees for consulting or serving on a speaker’s bureau for Medtronic, ReCor Medical, and Aktiia and receiving grant support or holding research contracts for Recor Medical and Aktiia. Dr. Messerli disclosed receiving honoraria from Medtronic, Menarini, Krka, and Ipca.
A version of this article first appeared on Medscape.com.
FROM TCT 2022
Royal family affliction or not, porphyria is treatable
European royal families may be enormously rich, but being a blueblood doesn’t always mean your blood is pristine. Queen Victoria’s DNA is famously believed to have silently bequeathed hemophilia to many of her descendants, including a great-grandson whose severe illness played a tragic role in spurring the Russian Revolution.
And that’s not all.
There’s plenty of skepticism about this theory, which seeks to explain the “madness” of King George III. But one thing is clear. If porphyria does indeed haunt the imperial bloodline that stretches to a new generation – the late Queen Elizabeth II’s great-grandchildren – any royal who’s afflicted going forward is likely to benefit mightily from modern treatment. While this disease may require lifelong vigilance, experts said in interviews that porphyria can often be controlled.
“If patients know they have the diagnosis, and they do the right things and avoid alcohol and risky drugs, most people will have few acute attacks,” said gastroenterologist Herbert Lloyd Bonkovsky, MD, of Wake Forest University, Winston-Salem, N.C., a leading porphyria specialist.
Heme infusions can also be helpful, he added, and the revolutionary new drug givosiran is available for those who suffer recurrent attacks. And “if all else fails, a successful liver transplant is curative” – as long as the transplanted liver doesn’t have porphyria, as happened in at least one case.
But, Dr. Bonkovsky cautioned, the diagnosis is often missed, in some cases for 15 years or more.
Diagnosing porphyria: Awareness and tests are crucial
Porphyria is caused when porphyins – essential components of hemoglobin – build up in the body, disrupting systems such as the nerves, skin, and gut. The urine can turn purplish, hence the condition’s name. (Porphyrus is the Greek word for purple.)
According to hematologist Danielle Nance, MD, of Banner MD Anderson Cancer Center in Gilbert, Ariz., acute intermittent porphyria “should be suspected in persons who have recurrent severe attacks of abdominal pain requiring strong pain medication to control symptoms, and there is no obvious physical cause.”
In such cases, practitioners should send out blood and urine for porphobilinogen (PBG) and delta-aminolevulinic acid (dALA or Delta-ALA) testing, Dr. Nance said. “These are almost always elevated, even between attacks, in persons with diagnoses of acute intermittent porphyria. Other types of porphyria, such as erythropoietic porphyria, may require additional testing. Genetic testing should be offered when a patient is suspected of having porphyria, as this can speed the diagnosis.”
The typical patient is a woman from age 18 to 55, often a young woman with recurrent abdominal pain that may occur during the second half of the menstrual cycle, Wake Forest’s Dr. Bonkovsky said. Constipation is common.
“She keeps coming to the clinic or emergency department, and no one knows what’s going on. Eventually, she tends to undergo an appendectomy, often a cholecystectomy, or sometimes gynecologic procedures without cure of the disease. Only after this long and arduous road of misdiagnosis does someone think it’s porphyria and do the correct tests.”
Dr. Bonkovsky led a 2014 study of 108 subjects (81% female) with acute porphyrias and found that the average time to a correct diagnosis was a whopping 15 years. Pain in the abdomen was the most common symptom (74%), followed by nausea/vomiting (73%), weakness (63%), and constipation (60%).
While underdiagnosis is common, porphyrias can also be overdiagnosed. According to Dr. Bonkovsky, a mild increase in urinary porphyrins is often misdiagnosed as porphyria when it may be a sign of liver disease or alcohol use, instead.
Hematologist Kleber Y. Fertrin, MD, PhD, of the University of Washington, Seattle, emphasized the importance of ordering the correct tests. “Urinary porphyrins are often inappropriately ordered because of their name. They are not diagnostic for acute hepatic porphyrias and may be nonspecifically elevated. It is paramount to get the labs from a reliable lab test experienced at performing diagnostic testing for porphyrias and make sure the urine sample needed is correctly obtained and preserved.”
New drug can control attacks, but it’s costly
Treatment should begin right away if porphyria is suspected, without waiting for confirmation, Dr. Nance said. “There are porphyria experts in the U.S. and in many other countries who can help practitioners interpret symptoms and testing results if there is uncertainty or if treatment options are unclear.”
Patients are counseled to avoid attack triggers and eat healthy diets, she said. “Persons with porphyria are encouraged to have a plan for home management of an attack and when to come to the hospital for symptoms that don’t respond to home therapy,” she said.
Dr. Fertrin said “the typical treatment for an acute porphyric attack is the use of intravenous glucose, as well as intravenous hemin infusions, along with pain medications and fluids as needed. Some patients may need medication for high blood pressure. For patients with recurrent attacks, prophylactic doses of hemin can be used.”
Another option is givosiran (Givlaari), the biggest recent advance in porphyria treatment. It was approved by the Food and Drug Administration for acute hepatic porphyria in 2019. “It’s an inhibitory mRNA that shuts down heme synthesis by downregulating aminolevulinic acid synthase,” said hematologist Thomas DeLoughery, MD, of Oregon Health & Science University, Portland.
A 2020 study coauthored by Dr. Bonkovsky found that the drug reduced the mean annualized attack rate in acute intermittent porphyria by 74%, compared with placebo (P < .001). “Givosiran led to lower levels of urinary ALA and porphobilinogen, fewer days of hemin use, and better daily scores for pain than placebo,” the study authors write. “Key adverse events that were observed more frequently in the givosiran group were elevations in serum aminotransferase levels, changes in serum creatinine levels and the estimated glomerular filtration rate, and injection-site reactions.”
Dr. Bonkovsky said the drug is appropriate in patients whose disease is not controlled by lifestyle interventions regarding alcohol, smoking, and substance use. However, while givosiran is helpful, “it’s not a cure,” he said. “Probably about 40% of patients have found the side effects such as nausea, fatigue, headaches, and liver injury to be too adverse and have not decided to take it in the long term.”
In addition, the drug costs about $500,000 a year, and insurers may balk at the expense. Still, specialists say it can be a good option. “Before givosiran, the standard therapy was hematin, which decreased the activity of the ALA synthase,” Dr. DeLoughery said. “This is a nasty drug that can inflame the blood vessels and is not all that effective.”
The royal family may be able to breathe easy
There are other forms of porphyria besides the acute type. Cutaneous porphryias can cause blistering from sunlight exposure, and treatment is avoidance of such triggers, Dr. Fertin said. “For a rare form called erythropoietic protoporphyria, there is a subcutaneous implant with a medication called afamelanotide that increases tolerance to sunlight.”
There’s another type: variegate porphyria, which UpToDate says is “characterized by cutaneous blistering and/or acute neurovisceral attacks.” In 1966, a mother-and-son psychiatrist team theorized that Britain’s King George III suffered from this type of porphyria.
However, researchers have sharply criticized the porphyria theory with regard to that king, and a recent theory now suggests that he actually had bipolar disorder. Dr. Fertin is among the skeptics, noting the high prevalence of porphyria in affected families.
“Since King George III would now have a living number of relatives of about 900 people, we would expect around 180 cases of variegate porphyria to be connected to him, and those are nowhere to be found,” he said. “The idea that it could cause progressive psychosis and dementia is also a misconception. We do not find that porphyria is the underlying cause of psychiatric conditions on their own. Many atypical symptoms of the king were attributed to variegate porphyria, and the few more typical symptoms he reportedly had, of abdominal pain and urine discoloration, can be more easily explained by common diseases, such as kidney stones or gallstones. Finally, attempts at obtaining genetic or biochemical evidence of porphyria from relatives of King George III have been negative or inconclusive.”
That’s good news for the royals, who have many other concerns these days.
Dr. Nance discloses research study funding from Alnylam (maker of Givlaari) and payments to participate in educational activities for patients and practitioners. Dr. Bonkovsky discloses relationships with drugmakers who have developed products to treat porphyria including Alnylam, Mitsubishi Tanabe, Recordati, and Disc. Dr. Fertin is subinvestigator in a clinical trial for a novel treatment for erythropoietic protoporphyria sponsored by Mitsubishi Tanabe. Dr. DeLoughery reports no disclosures.
European royal families may be enormously rich, but being a blueblood doesn’t always mean your blood is pristine. Queen Victoria’s DNA is famously believed to have silently bequeathed hemophilia to many of her descendants, including a great-grandson whose severe illness played a tragic role in spurring the Russian Revolution.
And that’s not all.
There’s plenty of skepticism about this theory, which seeks to explain the “madness” of King George III. But one thing is clear. If porphyria does indeed haunt the imperial bloodline that stretches to a new generation – the late Queen Elizabeth II’s great-grandchildren – any royal who’s afflicted going forward is likely to benefit mightily from modern treatment. While this disease may require lifelong vigilance, experts said in interviews that porphyria can often be controlled.
“If patients know they have the diagnosis, and they do the right things and avoid alcohol and risky drugs, most people will have few acute attacks,” said gastroenterologist Herbert Lloyd Bonkovsky, MD, of Wake Forest University, Winston-Salem, N.C., a leading porphyria specialist.
Heme infusions can also be helpful, he added, and the revolutionary new drug givosiran is available for those who suffer recurrent attacks. And “if all else fails, a successful liver transplant is curative” – as long as the transplanted liver doesn’t have porphyria, as happened in at least one case.
But, Dr. Bonkovsky cautioned, the diagnosis is often missed, in some cases for 15 years or more.
Diagnosing porphyria: Awareness and tests are crucial
Porphyria is caused when porphyins – essential components of hemoglobin – build up in the body, disrupting systems such as the nerves, skin, and gut. The urine can turn purplish, hence the condition’s name. (Porphyrus is the Greek word for purple.)
According to hematologist Danielle Nance, MD, of Banner MD Anderson Cancer Center in Gilbert, Ariz., acute intermittent porphyria “should be suspected in persons who have recurrent severe attacks of abdominal pain requiring strong pain medication to control symptoms, and there is no obvious physical cause.”
In such cases, practitioners should send out blood and urine for porphobilinogen (PBG) and delta-aminolevulinic acid (dALA or Delta-ALA) testing, Dr. Nance said. “These are almost always elevated, even between attacks, in persons with diagnoses of acute intermittent porphyria. Other types of porphyria, such as erythropoietic porphyria, may require additional testing. Genetic testing should be offered when a patient is suspected of having porphyria, as this can speed the diagnosis.”
The typical patient is a woman from age 18 to 55, often a young woman with recurrent abdominal pain that may occur during the second half of the menstrual cycle, Wake Forest’s Dr. Bonkovsky said. Constipation is common.
“She keeps coming to the clinic or emergency department, and no one knows what’s going on. Eventually, she tends to undergo an appendectomy, often a cholecystectomy, or sometimes gynecologic procedures without cure of the disease. Only after this long and arduous road of misdiagnosis does someone think it’s porphyria and do the correct tests.”
Dr. Bonkovsky led a 2014 study of 108 subjects (81% female) with acute porphyrias and found that the average time to a correct diagnosis was a whopping 15 years. Pain in the abdomen was the most common symptom (74%), followed by nausea/vomiting (73%), weakness (63%), and constipation (60%).
While underdiagnosis is common, porphyrias can also be overdiagnosed. According to Dr. Bonkovsky, a mild increase in urinary porphyrins is often misdiagnosed as porphyria when it may be a sign of liver disease or alcohol use, instead.
Hematologist Kleber Y. Fertrin, MD, PhD, of the University of Washington, Seattle, emphasized the importance of ordering the correct tests. “Urinary porphyrins are often inappropriately ordered because of their name. They are not diagnostic for acute hepatic porphyrias and may be nonspecifically elevated. It is paramount to get the labs from a reliable lab test experienced at performing diagnostic testing for porphyrias and make sure the urine sample needed is correctly obtained and preserved.”
New drug can control attacks, but it’s costly
Treatment should begin right away if porphyria is suspected, without waiting for confirmation, Dr. Nance said. “There are porphyria experts in the U.S. and in many other countries who can help practitioners interpret symptoms and testing results if there is uncertainty or if treatment options are unclear.”
Patients are counseled to avoid attack triggers and eat healthy diets, she said. “Persons with porphyria are encouraged to have a plan for home management of an attack and when to come to the hospital for symptoms that don’t respond to home therapy,” she said.
Dr. Fertrin said “the typical treatment for an acute porphyric attack is the use of intravenous glucose, as well as intravenous hemin infusions, along with pain medications and fluids as needed. Some patients may need medication for high blood pressure. For patients with recurrent attacks, prophylactic doses of hemin can be used.”
Another option is givosiran (Givlaari), the biggest recent advance in porphyria treatment. It was approved by the Food and Drug Administration for acute hepatic porphyria in 2019. “It’s an inhibitory mRNA that shuts down heme synthesis by downregulating aminolevulinic acid synthase,” said hematologist Thomas DeLoughery, MD, of Oregon Health & Science University, Portland.
A 2020 study coauthored by Dr. Bonkovsky found that the drug reduced the mean annualized attack rate in acute intermittent porphyria by 74%, compared with placebo (P < .001). “Givosiran led to lower levels of urinary ALA and porphobilinogen, fewer days of hemin use, and better daily scores for pain than placebo,” the study authors write. “Key adverse events that were observed more frequently in the givosiran group were elevations in serum aminotransferase levels, changes in serum creatinine levels and the estimated glomerular filtration rate, and injection-site reactions.”
Dr. Bonkovsky said the drug is appropriate in patients whose disease is not controlled by lifestyle interventions regarding alcohol, smoking, and substance use. However, while givosiran is helpful, “it’s not a cure,” he said. “Probably about 40% of patients have found the side effects such as nausea, fatigue, headaches, and liver injury to be too adverse and have not decided to take it in the long term.”
In addition, the drug costs about $500,000 a year, and insurers may balk at the expense. Still, specialists say it can be a good option. “Before givosiran, the standard therapy was hematin, which decreased the activity of the ALA synthase,” Dr. DeLoughery said. “This is a nasty drug that can inflame the blood vessels and is not all that effective.”
The royal family may be able to breathe easy
There are other forms of porphyria besides the acute type. Cutaneous porphryias can cause blistering from sunlight exposure, and treatment is avoidance of such triggers, Dr. Fertin said. “For a rare form called erythropoietic protoporphyria, there is a subcutaneous implant with a medication called afamelanotide that increases tolerance to sunlight.”
There’s another type: variegate porphyria, which UpToDate says is “characterized by cutaneous blistering and/or acute neurovisceral attacks.” In 1966, a mother-and-son psychiatrist team theorized that Britain’s King George III suffered from this type of porphyria.
However, researchers have sharply criticized the porphyria theory with regard to that king, and a recent theory now suggests that he actually had bipolar disorder. Dr. Fertin is among the skeptics, noting the high prevalence of porphyria in affected families.
“Since King George III would now have a living number of relatives of about 900 people, we would expect around 180 cases of variegate porphyria to be connected to him, and those are nowhere to be found,” he said. “The idea that it could cause progressive psychosis and dementia is also a misconception. We do not find that porphyria is the underlying cause of psychiatric conditions on their own. Many atypical symptoms of the king were attributed to variegate porphyria, and the few more typical symptoms he reportedly had, of abdominal pain and urine discoloration, can be more easily explained by common diseases, such as kidney stones or gallstones. Finally, attempts at obtaining genetic or biochemical evidence of porphyria from relatives of King George III have been negative or inconclusive.”
That’s good news for the royals, who have many other concerns these days.
Dr. Nance discloses research study funding from Alnylam (maker of Givlaari) and payments to participate in educational activities for patients and practitioners. Dr. Bonkovsky discloses relationships with drugmakers who have developed products to treat porphyria including Alnylam, Mitsubishi Tanabe, Recordati, and Disc. Dr. Fertin is subinvestigator in a clinical trial for a novel treatment for erythropoietic protoporphyria sponsored by Mitsubishi Tanabe. Dr. DeLoughery reports no disclosures.
European royal families may be enormously rich, but being a blueblood doesn’t always mean your blood is pristine. Queen Victoria’s DNA is famously believed to have silently bequeathed hemophilia to many of her descendants, including a great-grandson whose severe illness played a tragic role in spurring the Russian Revolution.
And that’s not all.
There’s plenty of skepticism about this theory, which seeks to explain the “madness” of King George III. But one thing is clear. If porphyria does indeed haunt the imperial bloodline that stretches to a new generation – the late Queen Elizabeth II’s great-grandchildren – any royal who’s afflicted going forward is likely to benefit mightily from modern treatment. While this disease may require lifelong vigilance, experts said in interviews that porphyria can often be controlled.
“If patients know they have the diagnosis, and they do the right things and avoid alcohol and risky drugs, most people will have few acute attacks,” said gastroenterologist Herbert Lloyd Bonkovsky, MD, of Wake Forest University, Winston-Salem, N.C., a leading porphyria specialist.
Heme infusions can also be helpful, he added, and the revolutionary new drug givosiran is available for those who suffer recurrent attacks. And “if all else fails, a successful liver transplant is curative” – as long as the transplanted liver doesn’t have porphyria, as happened in at least one case.
But, Dr. Bonkovsky cautioned, the diagnosis is often missed, in some cases for 15 years or more.
Diagnosing porphyria: Awareness and tests are crucial
Porphyria is caused when porphyins – essential components of hemoglobin – build up in the body, disrupting systems such as the nerves, skin, and gut. The urine can turn purplish, hence the condition’s name. (Porphyrus is the Greek word for purple.)
According to hematologist Danielle Nance, MD, of Banner MD Anderson Cancer Center in Gilbert, Ariz., acute intermittent porphyria “should be suspected in persons who have recurrent severe attacks of abdominal pain requiring strong pain medication to control symptoms, and there is no obvious physical cause.”
In such cases, practitioners should send out blood and urine for porphobilinogen (PBG) and delta-aminolevulinic acid (dALA or Delta-ALA) testing, Dr. Nance said. “These are almost always elevated, even between attacks, in persons with diagnoses of acute intermittent porphyria. Other types of porphyria, such as erythropoietic porphyria, may require additional testing. Genetic testing should be offered when a patient is suspected of having porphyria, as this can speed the diagnosis.”
The typical patient is a woman from age 18 to 55, often a young woman with recurrent abdominal pain that may occur during the second half of the menstrual cycle, Wake Forest’s Dr. Bonkovsky said. Constipation is common.
“She keeps coming to the clinic or emergency department, and no one knows what’s going on. Eventually, she tends to undergo an appendectomy, often a cholecystectomy, or sometimes gynecologic procedures without cure of the disease. Only after this long and arduous road of misdiagnosis does someone think it’s porphyria and do the correct tests.”
Dr. Bonkovsky led a 2014 study of 108 subjects (81% female) with acute porphyrias and found that the average time to a correct diagnosis was a whopping 15 years. Pain in the abdomen was the most common symptom (74%), followed by nausea/vomiting (73%), weakness (63%), and constipation (60%).
While underdiagnosis is common, porphyrias can also be overdiagnosed. According to Dr. Bonkovsky, a mild increase in urinary porphyrins is often misdiagnosed as porphyria when it may be a sign of liver disease or alcohol use, instead.
Hematologist Kleber Y. Fertrin, MD, PhD, of the University of Washington, Seattle, emphasized the importance of ordering the correct tests. “Urinary porphyrins are often inappropriately ordered because of their name. They are not diagnostic for acute hepatic porphyrias and may be nonspecifically elevated. It is paramount to get the labs from a reliable lab test experienced at performing diagnostic testing for porphyrias and make sure the urine sample needed is correctly obtained and preserved.”
New drug can control attacks, but it’s costly
Treatment should begin right away if porphyria is suspected, without waiting for confirmation, Dr. Nance said. “There are porphyria experts in the U.S. and in many other countries who can help practitioners interpret symptoms and testing results if there is uncertainty or if treatment options are unclear.”
Patients are counseled to avoid attack triggers and eat healthy diets, she said. “Persons with porphyria are encouraged to have a plan for home management of an attack and when to come to the hospital for symptoms that don’t respond to home therapy,” she said.
Dr. Fertrin said “the typical treatment for an acute porphyric attack is the use of intravenous glucose, as well as intravenous hemin infusions, along with pain medications and fluids as needed. Some patients may need medication for high blood pressure. For patients with recurrent attacks, prophylactic doses of hemin can be used.”
Another option is givosiran (Givlaari), the biggest recent advance in porphyria treatment. It was approved by the Food and Drug Administration for acute hepatic porphyria in 2019. “It’s an inhibitory mRNA that shuts down heme synthesis by downregulating aminolevulinic acid synthase,” said hematologist Thomas DeLoughery, MD, of Oregon Health & Science University, Portland.
A 2020 study coauthored by Dr. Bonkovsky found that the drug reduced the mean annualized attack rate in acute intermittent porphyria by 74%, compared with placebo (P < .001). “Givosiran led to lower levels of urinary ALA and porphobilinogen, fewer days of hemin use, and better daily scores for pain than placebo,” the study authors write. “Key adverse events that were observed more frequently in the givosiran group were elevations in serum aminotransferase levels, changes in serum creatinine levels and the estimated glomerular filtration rate, and injection-site reactions.”
Dr. Bonkovsky said the drug is appropriate in patients whose disease is not controlled by lifestyle interventions regarding alcohol, smoking, and substance use. However, while givosiran is helpful, “it’s not a cure,” he said. “Probably about 40% of patients have found the side effects such as nausea, fatigue, headaches, and liver injury to be too adverse and have not decided to take it in the long term.”
In addition, the drug costs about $500,000 a year, and insurers may balk at the expense. Still, specialists say it can be a good option. “Before givosiran, the standard therapy was hematin, which decreased the activity of the ALA synthase,” Dr. DeLoughery said. “This is a nasty drug that can inflame the blood vessels and is not all that effective.”
The royal family may be able to breathe easy
There are other forms of porphyria besides the acute type. Cutaneous porphryias can cause blistering from sunlight exposure, and treatment is avoidance of such triggers, Dr. Fertin said. “For a rare form called erythropoietic protoporphyria, there is a subcutaneous implant with a medication called afamelanotide that increases tolerance to sunlight.”
There’s another type: variegate porphyria, which UpToDate says is “characterized by cutaneous blistering and/or acute neurovisceral attacks.” In 1966, a mother-and-son psychiatrist team theorized that Britain’s King George III suffered from this type of porphyria.
However, researchers have sharply criticized the porphyria theory with regard to that king, and a recent theory now suggests that he actually had bipolar disorder. Dr. Fertin is among the skeptics, noting the high prevalence of porphyria in affected families.
“Since King George III would now have a living number of relatives of about 900 people, we would expect around 180 cases of variegate porphyria to be connected to him, and those are nowhere to be found,” he said. “The idea that it could cause progressive psychosis and dementia is also a misconception. We do not find that porphyria is the underlying cause of psychiatric conditions on their own. Many atypical symptoms of the king were attributed to variegate porphyria, and the few more typical symptoms he reportedly had, of abdominal pain and urine discoloration, can be more easily explained by common diseases, such as kidney stones or gallstones. Finally, attempts at obtaining genetic or biochemical evidence of porphyria from relatives of King George III have been negative or inconclusive.”
That’s good news for the royals, who have many other concerns these days.
Dr. Nance discloses research study funding from Alnylam (maker of Givlaari) and payments to participate in educational activities for patients and practitioners. Dr. Bonkovsky discloses relationships with drugmakers who have developed products to treat porphyria including Alnylam, Mitsubishi Tanabe, Recordati, and Disc. Dr. Fertin is subinvestigator in a clinical trial for a novel treatment for erythropoietic protoporphyria sponsored by Mitsubishi Tanabe. Dr. DeLoughery reports no disclosures.
Horse hockey notwithstanding
He’s 24 years younger than I am, recently married, no kids. Just starting out as a neurologist. He also has a full head of hair, something I’m admittedly jealous of.
He’s always in a sweater, something that seems oddly out of place in Phoenix, Arizona.
He’s the picture on my hospital ID.
I don’t go to the hospital much anymore, but he still sits in my car, greeting me whenever I open the center console to get my sunglasses or phone charger. He looks very enthusiastic about starting his career. I clearly remember the day I had the picture taken, as a newly-minted attending getting his first hospital privileges.
Sometimes I talk to him. Usually it’s just silly advice (“bet on the ’16 Cubs”). Other times I wonder what he’d do in certain situations, with all his youthful enthusiasm. I’m sure he wonders the same about me, with my 24 years of experience.
To a large extent we are the same people we started out as, but time changes us, in ways besides the obvious (like my hairs jumping off like lemmings).
Looking back at him (or even the older pic on my medical school application) I have no complaints about where life and my career have taken me. Would there be a few things I might have changed if I could go back?
Realistically, maybe one or two, both involving my father, but neither of them would likely change where I am.
But as far as medicine goes? Not really. The things I liked then, that got me into the field? I still enjoy them. The horse hockey? Yeah, it’s always there, probably has gotten worse over time, and it still bothers me. But there isn’t a job that doesn’t have its share of cow patties. It’s just a matter of trying not to step in them more than necessary as you do the parts you enjoy.
Sometimes I look at my younger self, and wonder what I’d really say to him if we actually met.
Probably just “good luck, enjoy the ride, and ditch the sweater.”
We measure our gains out in luck and coincidence
Lanterns to turn back the night.
And put our defeats down to chance or experience
And try once again for the light.
– Al Stewart
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
He’s 24 years younger than I am, recently married, no kids. Just starting out as a neurologist. He also has a full head of hair, something I’m admittedly jealous of.
He’s always in a sweater, something that seems oddly out of place in Phoenix, Arizona.
He’s the picture on my hospital ID.
I don’t go to the hospital much anymore, but he still sits in my car, greeting me whenever I open the center console to get my sunglasses or phone charger. He looks very enthusiastic about starting his career. I clearly remember the day I had the picture taken, as a newly-minted attending getting his first hospital privileges.
Sometimes I talk to him. Usually it’s just silly advice (“bet on the ’16 Cubs”). Other times I wonder what he’d do in certain situations, with all his youthful enthusiasm. I’m sure he wonders the same about me, with my 24 years of experience.
To a large extent we are the same people we started out as, but time changes us, in ways besides the obvious (like my hairs jumping off like lemmings).
Looking back at him (or even the older pic on my medical school application) I have no complaints about where life and my career have taken me. Would there be a few things I might have changed if I could go back?
Realistically, maybe one or two, both involving my father, but neither of them would likely change where I am.
But as far as medicine goes? Not really. The things I liked then, that got me into the field? I still enjoy them. The horse hockey? Yeah, it’s always there, probably has gotten worse over time, and it still bothers me. But there isn’t a job that doesn’t have its share of cow patties. It’s just a matter of trying not to step in them more than necessary as you do the parts you enjoy.
Sometimes I look at my younger self, and wonder what I’d really say to him if we actually met.
Probably just “good luck, enjoy the ride, and ditch the sweater.”
We measure our gains out in luck and coincidence
Lanterns to turn back the night.
And put our defeats down to chance or experience
And try once again for the light.
– Al Stewart
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
He’s 24 years younger than I am, recently married, no kids. Just starting out as a neurologist. He also has a full head of hair, something I’m admittedly jealous of.
He’s always in a sweater, something that seems oddly out of place in Phoenix, Arizona.
He’s the picture on my hospital ID.
I don’t go to the hospital much anymore, but he still sits in my car, greeting me whenever I open the center console to get my sunglasses or phone charger. He looks very enthusiastic about starting his career. I clearly remember the day I had the picture taken, as a newly-minted attending getting his first hospital privileges.
Sometimes I talk to him. Usually it’s just silly advice (“bet on the ’16 Cubs”). Other times I wonder what he’d do in certain situations, with all his youthful enthusiasm. I’m sure he wonders the same about me, with my 24 years of experience.
To a large extent we are the same people we started out as, but time changes us, in ways besides the obvious (like my hairs jumping off like lemmings).
Looking back at him (or even the older pic on my medical school application) I have no complaints about where life and my career have taken me. Would there be a few things I might have changed if I could go back?
Realistically, maybe one or two, both involving my father, but neither of them would likely change where I am.
But as far as medicine goes? Not really. The things I liked then, that got me into the field? I still enjoy them. The horse hockey? Yeah, it’s always there, probably has gotten worse over time, and it still bothers me. But there isn’t a job that doesn’t have its share of cow patties. It’s just a matter of trying not to step in them more than necessary as you do the parts you enjoy.
Sometimes I look at my younger self, and wonder what I’d really say to him if we actually met.
Probably just “good luck, enjoy the ride, and ditch the sweater.”
We measure our gains out in luck and coincidence
Lanterns to turn back the night.
And put our defeats down to chance or experience
And try once again for the light.
– Al Stewart
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Experts sound alarm on ruling threatening preventive cancer care
In a statement, the groups highlighted that the decision “would result in a return to financial and other barriers proven to discourage Americans from obtaining lifesaving, preventive care.”
The ruling, issued earlier in September by a federal district judge in Texas, essentially says that the U.S. Preventive Services Task Force has no authority to determine which preventive care services must be fully covered by insurance companies – an ability granted by the Affordable Care Act (ACA).
Judge Reed O’Connor ruled that the method of appointing officers to the USPSTF is unconstitutional, which means the task force’s recommendations for no-cost preventive health care may no longer be guaranteed under the ACA for millions of insured Americans.
The judgment, however, is not yet final, and individuals still have access to these preventive services. The judge must first make the scope of the ruling clear, and the decision will likely be appealed. In addition, the decision does not affect the authority of two other entities that make recommendations about vaccinations and preventive care for infants, children, and adolescents.
But experts are concerned that the ruling will force some individuals to pay out of pocket for preventive cancer screenings and other care that would otherwise have been fully covered by insurance.
After the ruling, a group of 26 patient groups, including the American Cancer Society Cancer Action Network and the Leukemia and Lymphoma Society, took a stand.
In a statement, the groups highlighted that “access to preventive health care can prevent both disease and early death.” Under the ACA, more than 150 million Americans have benefited from expanded access to these services, and research reveals that high-quality coverage – which includes preventive services – improves health, reduces health disparities, and lowers health care costs. “This ruling directly threatens these benefits,” they explained.
Lisa Lacasse, MBA, president of ACS CAN, agreed that the ruling “threatens to erode more than a decade of progress reducing cancer deaths and suffering.”
In a statement, the ACS CAN urged the government to “swiftly appeal” the decision.
“We cannot risk returning to a system wherein every individual has to interpret their complex insurance plans to determine if a recommended mammogram will be covered or to determine how much their colonoscopy may cost,” Ms. Lacasse told this news organization.
For now, Ms. Lacasse urged patients and providers to remember that no changes to coverage requirements will occur while litigation continues.
“All preventive services required under the Affordable Care Act remain in place with no cost sharing for enrollees,” she said. “ACS CAN will continue to support and advocate for coverage of preventive services at no cost sharing.”
A version of this article first appeared on Medscape.com.
In a statement, the groups highlighted that the decision “would result in a return to financial and other barriers proven to discourage Americans from obtaining lifesaving, preventive care.”
The ruling, issued earlier in September by a federal district judge in Texas, essentially says that the U.S. Preventive Services Task Force has no authority to determine which preventive care services must be fully covered by insurance companies – an ability granted by the Affordable Care Act (ACA).
Judge Reed O’Connor ruled that the method of appointing officers to the USPSTF is unconstitutional, which means the task force’s recommendations for no-cost preventive health care may no longer be guaranteed under the ACA for millions of insured Americans.
The judgment, however, is not yet final, and individuals still have access to these preventive services. The judge must first make the scope of the ruling clear, and the decision will likely be appealed. In addition, the decision does not affect the authority of two other entities that make recommendations about vaccinations and preventive care for infants, children, and adolescents.
But experts are concerned that the ruling will force some individuals to pay out of pocket for preventive cancer screenings and other care that would otherwise have been fully covered by insurance.
After the ruling, a group of 26 patient groups, including the American Cancer Society Cancer Action Network and the Leukemia and Lymphoma Society, took a stand.
In a statement, the groups highlighted that “access to preventive health care can prevent both disease and early death.” Under the ACA, more than 150 million Americans have benefited from expanded access to these services, and research reveals that high-quality coverage – which includes preventive services – improves health, reduces health disparities, and lowers health care costs. “This ruling directly threatens these benefits,” they explained.
Lisa Lacasse, MBA, president of ACS CAN, agreed that the ruling “threatens to erode more than a decade of progress reducing cancer deaths and suffering.”
In a statement, the ACS CAN urged the government to “swiftly appeal” the decision.
“We cannot risk returning to a system wherein every individual has to interpret their complex insurance plans to determine if a recommended mammogram will be covered or to determine how much their colonoscopy may cost,” Ms. Lacasse told this news organization.
For now, Ms. Lacasse urged patients and providers to remember that no changes to coverage requirements will occur while litigation continues.
“All preventive services required under the Affordable Care Act remain in place with no cost sharing for enrollees,” she said. “ACS CAN will continue to support and advocate for coverage of preventive services at no cost sharing.”
A version of this article first appeared on Medscape.com.
In a statement, the groups highlighted that the decision “would result in a return to financial and other barriers proven to discourage Americans from obtaining lifesaving, preventive care.”
The ruling, issued earlier in September by a federal district judge in Texas, essentially says that the U.S. Preventive Services Task Force has no authority to determine which preventive care services must be fully covered by insurance companies – an ability granted by the Affordable Care Act (ACA).
Judge Reed O’Connor ruled that the method of appointing officers to the USPSTF is unconstitutional, which means the task force’s recommendations for no-cost preventive health care may no longer be guaranteed under the ACA for millions of insured Americans.
The judgment, however, is not yet final, and individuals still have access to these preventive services. The judge must first make the scope of the ruling clear, and the decision will likely be appealed. In addition, the decision does not affect the authority of two other entities that make recommendations about vaccinations and preventive care for infants, children, and adolescents.
But experts are concerned that the ruling will force some individuals to pay out of pocket for preventive cancer screenings and other care that would otherwise have been fully covered by insurance.
After the ruling, a group of 26 patient groups, including the American Cancer Society Cancer Action Network and the Leukemia and Lymphoma Society, took a stand.
In a statement, the groups highlighted that “access to preventive health care can prevent both disease and early death.” Under the ACA, more than 150 million Americans have benefited from expanded access to these services, and research reveals that high-quality coverage – which includes preventive services – improves health, reduces health disparities, and lowers health care costs. “This ruling directly threatens these benefits,” they explained.
Lisa Lacasse, MBA, president of ACS CAN, agreed that the ruling “threatens to erode more than a decade of progress reducing cancer deaths and suffering.”
In a statement, the ACS CAN urged the government to “swiftly appeal” the decision.
“We cannot risk returning to a system wherein every individual has to interpret their complex insurance plans to determine if a recommended mammogram will be covered or to determine how much their colonoscopy may cost,” Ms. Lacasse told this news organization.
For now, Ms. Lacasse urged patients and providers to remember that no changes to coverage requirements will occur while litigation continues.
“All preventive services required under the Affordable Care Act remain in place with no cost sharing for enrollees,” she said. “ACS CAN will continue to support and advocate for coverage of preventive services at no cost sharing.”
A version of this article first appeared on Medscape.com.
TAVR now used in almost 50% of younger severe aortic stenosis patients
Among patients with severe isolated aortic stenosis younger than 65, the rate of transcatheter aortic valve replacement (TAVR) now almost matches that of surgical aortic valve replacement (SAVR), despite guideline recommendations to the contrary, a study in a national U.S. population shows.
The 2020 American Heart Association/American College of Cardiology (AHA/ACC) valve guideline recommends SAVR for patients younger than 65 with severe aortic stenosis, the researchers note, but their study showed “near equal utilization between TAVR and SAVR in these younger patients by 2021,” at 48% and 52% respectively.
Toishi Sharma, MD, and colleagues presented these findings in an oral poster session at Transcatheter Cardiovascular Therapeutics 2022, and the study was simultaneously published as a Research Letter in the Journal of the American College of Cardiology (JACC).
“To our knowledge, the current findings represent the first national temporal trends study stratifying [aortic stenosis] therapies according to guideline-recommended age groups: our observations demonstrate the dramatic growth of TAVR in all age groups, including young patients,” the researchers conclude.
They analyzed changes in rates of TAVR and SAVR in a U.S. sample stratified by age: younger than 65 years, 65-80, and older than 80 years.
These findings have implications for lifetime management of younger patients who undergo TAVR, they write, “including issues related to lifetime coronary access, valve durability, and the potential for subsequent TAVR procedures over time.”
Three age groups
In a study published in JACC, this group examined changes in uptake of TAVR versus SAVR in 4,161 patients with aortic stenosis in Vermont, New Hampshire, and Maine, senior author Harold L. Dauerman, MD, said in an interview.
The greatest rate of rise of TAVR was in the group younger than 65, but that study ended in 2019, said Dr. Dauerman, from the University of Vermont Health Network, Burlington.
The 2020 guideline stratifies TAVR and SAVR recommendations such that “less than 65 should primarily be a surgical approach and greater than 80 primarily a TAVR approach, while 65 to 80 is a gray zone, and shared decision-making becomes important,” he noted.
The group hypothesized that recent trials and technology have led to a national increase in TAVR in people younger than 65.
From the Vizient clinical database, including more than 250 U.S. academic centers that perform both TAVR and SAVR, the researchers identified 142,953 patients who underwent TAVR or SAVR for isolated aortic stenosis from Oct. 1, 2015, to Dec. 31, 2021. From 2015 to 2021, the valve replacement rates in the three age groups changed as follows:
- Age less than 65: TAVR rose from 17% to 48%; SAVR fell from 83% to 52%.
- Age 65-80: TAVR rose from 46% to 87%; SAVR fell from 54% to 12%.
- Age greater than 80: TAVR rose from 83% to 99%; SAVR fell from 16% to 1%.
“All ages have grown in the last 7 years in TAVR,” Dr. Dauerman summarized. “The one that’s surprising, and in contradiction to the guideline, is the growth of TAVR in young patients less than 65.”
Among patients younger than 65, prior bypass surgery and congestive heart failure predicted the use of TAVR instead of surgery, whereas bicuspid aortic valve disease was the biggest predictor of surgery instead of TAVR.
Most studies on TAVR valve durability are limited to patients in the randomized trials who were primarily in their mid-70s to mid-80s, some of whom died before a 10-year follow-up, Dr. Dauerman noted.
European guidelines recommend surgery for patients younger than 70, and it would be interesting to see if clinicians there follow this recommendation or if TAVR is now the preferred approach, he added.
There is a need for further, longer study of TAVR in younger patients, he said, to determine whether there are long-term clinical issues of concern.
Strategy depends on more than age
The “findings are not too surprising,” John Carroll, MD, who was not involved in this research, said in an email.
“Age is only one of multiple patient characteristics that enter into consideration of TAVR versus SAVR,” said Dr. Carroll, from Anschutz Medical Campus, University of Colorado, Aurora.
“As the article reports,” he noted, “those less than 65 having TAVR are more likely to have comorbid conditions that likely made the risk of SAVR higher.”
Dr. Carroll was lead author of a review article published in 2020 based on data from the ACC–Society of Thoracic Surgeons (STS)–Transcatheter Valve Therapy (TVT) registry on 276,316 patients who had TAVR in the United States from 2011-2019.
He pointed out that Figure 2 in that review shows that “SAVR is often performed in conjunction with other surgical procedures – another major reason why SAVR remains an important treatment for valvular heart disease.”
“We are awaiting long-term data comparing TAVR to SAVR durability,” Dr. Carroll added, echoing Dr. Dauerman. “So far [there are] no major differences, but it remains a key need to fully understand TAVR and the various models in commercial use.”
“Both TAVR and SAVR used in adults are tissue valves (SAVR with mechanical valves is used in younger patients),” Dr. Carroll noted, “and all tissue valves will eventually fail if the patient lives long enough.”
Patient management strategies need to consider what treatment options exist when the first valve fails. “If the first valve is SAVR, there is now extensive experience with placing a TAVR valve inside a failing SAVR valve, so called Valve-in-Valve or TAVR-in-SAVR. This is the preferred treatment in most patients with failing SAVR valves,” he said.
“On the other hand,” he continued, “we are just beginning to see more and more patients with failing TAVR valves, and the TAVR-in-TAVR procedure is less well understood.”
“Issues such as acute coronary occlusion and long-term difficulty in accessing coronary arteries are being encountered in some patients having TAVR-in-TAVR,” Dr. Carroll noted, which he discusses in a recent editorial he coauthored about the complexities of redo TAVR, published in JACC: Cardiovascular Interventions.
The study received no funding. Dr. Dauerman has research grants and is a consultant for Medtronic and Boston Scientific. Dr. Carroll is a local principal investigator in trials sponsored by Medtronic, Abbott, and Edwards Lifesciences.
A version of this article first appeared on Medscape.com.
Among patients with severe isolated aortic stenosis younger than 65, the rate of transcatheter aortic valve replacement (TAVR) now almost matches that of surgical aortic valve replacement (SAVR), despite guideline recommendations to the contrary, a study in a national U.S. population shows.
The 2020 American Heart Association/American College of Cardiology (AHA/ACC) valve guideline recommends SAVR for patients younger than 65 with severe aortic stenosis, the researchers note, but their study showed “near equal utilization between TAVR and SAVR in these younger patients by 2021,” at 48% and 52% respectively.
Toishi Sharma, MD, and colleagues presented these findings in an oral poster session at Transcatheter Cardiovascular Therapeutics 2022, and the study was simultaneously published as a Research Letter in the Journal of the American College of Cardiology (JACC).
“To our knowledge, the current findings represent the first national temporal trends study stratifying [aortic stenosis] therapies according to guideline-recommended age groups: our observations demonstrate the dramatic growth of TAVR in all age groups, including young patients,” the researchers conclude.
They analyzed changes in rates of TAVR and SAVR in a U.S. sample stratified by age: younger than 65 years, 65-80, and older than 80 years.
These findings have implications for lifetime management of younger patients who undergo TAVR, they write, “including issues related to lifetime coronary access, valve durability, and the potential for subsequent TAVR procedures over time.”
Three age groups
In a study published in JACC, this group examined changes in uptake of TAVR versus SAVR in 4,161 patients with aortic stenosis in Vermont, New Hampshire, and Maine, senior author Harold L. Dauerman, MD, said in an interview.
The greatest rate of rise of TAVR was in the group younger than 65, but that study ended in 2019, said Dr. Dauerman, from the University of Vermont Health Network, Burlington.
The 2020 guideline stratifies TAVR and SAVR recommendations such that “less than 65 should primarily be a surgical approach and greater than 80 primarily a TAVR approach, while 65 to 80 is a gray zone, and shared decision-making becomes important,” he noted.
The group hypothesized that recent trials and technology have led to a national increase in TAVR in people younger than 65.
From the Vizient clinical database, including more than 250 U.S. academic centers that perform both TAVR and SAVR, the researchers identified 142,953 patients who underwent TAVR or SAVR for isolated aortic stenosis from Oct. 1, 2015, to Dec. 31, 2021. From 2015 to 2021, the valve replacement rates in the three age groups changed as follows:
- Age less than 65: TAVR rose from 17% to 48%; SAVR fell from 83% to 52%.
- Age 65-80: TAVR rose from 46% to 87%; SAVR fell from 54% to 12%.
- Age greater than 80: TAVR rose from 83% to 99%; SAVR fell from 16% to 1%.
“All ages have grown in the last 7 years in TAVR,” Dr. Dauerman summarized. “The one that’s surprising, and in contradiction to the guideline, is the growth of TAVR in young patients less than 65.”
Among patients younger than 65, prior bypass surgery and congestive heart failure predicted the use of TAVR instead of surgery, whereas bicuspid aortic valve disease was the biggest predictor of surgery instead of TAVR.
Most studies on TAVR valve durability are limited to patients in the randomized trials who were primarily in their mid-70s to mid-80s, some of whom died before a 10-year follow-up, Dr. Dauerman noted.
European guidelines recommend surgery for patients younger than 70, and it would be interesting to see if clinicians there follow this recommendation or if TAVR is now the preferred approach, he added.
There is a need for further, longer study of TAVR in younger patients, he said, to determine whether there are long-term clinical issues of concern.
Strategy depends on more than age
The “findings are not too surprising,” John Carroll, MD, who was not involved in this research, said in an email.
“Age is only one of multiple patient characteristics that enter into consideration of TAVR versus SAVR,” said Dr. Carroll, from Anschutz Medical Campus, University of Colorado, Aurora.
“As the article reports,” he noted, “those less than 65 having TAVR are more likely to have comorbid conditions that likely made the risk of SAVR higher.”
Dr. Carroll was lead author of a review article published in 2020 based on data from the ACC–Society of Thoracic Surgeons (STS)–Transcatheter Valve Therapy (TVT) registry on 276,316 patients who had TAVR in the United States from 2011-2019.
He pointed out that Figure 2 in that review shows that “SAVR is often performed in conjunction with other surgical procedures – another major reason why SAVR remains an important treatment for valvular heart disease.”
“We are awaiting long-term data comparing TAVR to SAVR durability,” Dr. Carroll added, echoing Dr. Dauerman. “So far [there are] no major differences, but it remains a key need to fully understand TAVR and the various models in commercial use.”
“Both TAVR and SAVR used in adults are tissue valves (SAVR with mechanical valves is used in younger patients),” Dr. Carroll noted, “and all tissue valves will eventually fail if the patient lives long enough.”
Patient management strategies need to consider what treatment options exist when the first valve fails. “If the first valve is SAVR, there is now extensive experience with placing a TAVR valve inside a failing SAVR valve, so called Valve-in-Valve or TAVR-in-SAVR. This is the preferred treatment in most patients with failing SAVR valves,” he said.
“On the other hand,” he continued, “we are just beginning to see more and more patients with failing TAVR valves, and the TAVR-in-TAVR procedure is less well understood.”
“Issues such as acute coronary occlusion and long-term difficulty in accessing coronary arteries are being encountered in some patients having TAVR-in-TAVR,” Dr. Carroll noted, which he discusses in a recent editorial he coauthored about the complexities of redo TAVR, published in JACC: Cardiovascular Interventions.
The study received no funding. Dr. Dauerman has research grants and is a consultant for Medtronic and Boston Scientific. Dr. Carroll is a local principal investigator in trials sponsored by Medtronic, Abbott, and Edwards Lifesciences.
A version of this article first appeared on Medscape.com.
Among patients with severe isolated aortic stenosis younger than 65, the rate of transcatheter aortic valve replacement (TAVR) now almost matches that of surgical aortic valve replacement (SAVR), despite guideline recommendations to the contrary, a study in a national U.S. population shows.
The 2020 American Heart Association/American College of Cardiology (AHA/ACC) valve guideline recommends SAVR for patients younger than 65 with severe aortic stenosis, the researchers note, but their study showed “near equal utilization between TAVR and SAVR in these younger patients by 2021,” at 48% and 52% respectively.
Toishi Sharma, MD, and colleagues presented these findings in an oral poster session at Transcatheter Cardiovascular Therapeutics 2022, and the study was simultaneously published as a Research Letter in the Journal of the American College of Cardiology (JACC).
“To our knowledge, the current findings represent the first national temporal trends study stratifying [aortic stenosis] therapies according to guideline-recommended age groups: our observations demonstrate the dramatic growth of TAVR in all age groups, including young patients,” the researchers conclude.
They analyzed changes in rates of TAVR and SAVR in a U.S. sample stratified by age: younger than 65 years, 65-80, and older than 80 years.
These findings have implications for lifetime management of younger patients who undergo TAVR, they write, “including issues related to lifetime coronary access, valve durability, and the potential for subsequent TAVR procedures over time.”
Three age groups
In a study published in JACC, this group examined changes in uptake of TAVR versus SAVR in 4,161 patients with aortic stenosis in Vermont, New Hampshire, and Maine, senior author Harold L. Dauerman, MD, said in an interview.
The greatest rate of rise of TAVR was in the group younger than 65, but that study ended in 2019, said Dr. Dauerman, from the University of Vermont Health Network, Burlington.
The 2020 guideline stratifies TAVR and SAVR recommendations such that “less than 65 should primarily be a surgical approach and greater than 80 primarily a TAVR approach, while 65 to 80 is a gray zone, and shared decision-making becomes important,” he noted.
The group hypothesized that recent trials and technology have led to a national increase in TAVR in people younger than 65.
From the Vizient clinical database, including more than 250 U.S. academic centers that perform both TAVR and SAVR, the researchers identified 142,953 patients who underwent TAVR or SAVR for isolated aortic stenosis from Oct. 1, 2015, to Dec. 31, 2021. From 2015 to 2021, the valve replacement rates in the three age groups changed as follows:
- Age less than 65: TAVR rose from 17% to 48%; SAVR fell from 83% to 52%.
- Age 65-80: TAVR rose from 46% to 87%; SAVR fell from 54% to 12%.
- Age greater than 80: TAVR rose from 83% to 99%; SAVR fell from 16% to 1%.
“All ages have grown in the last 7 years in TAVR,” Dr. Dauerman summarized. “The one that’s surprising, and in contradiction to the guideline, is the growth of TAVR in young patients less than 65.”
Among patients younger than 65, prior bypass surgery and congestive heart failure predicted the use of TAVR instead of surgery, whereas bicuspid aortic valve disease was the biggest predictor of surgery instead of TAVR.
Most studies on TAVR valve durability are limited to patients in the randomized trials who were primarily in their mid-70s to mid-80s, some of whom died before a 10-year follow-up, Dr. Dauerman noted.
European guidelines recommend surgery for patients younger than 70, and it would be interesting to see if clinicians there follow this recommendation or if TAVR is now the preferred approach, he added.
There is a need for further, longer study of TAVR in younger patients, he said, to determine whether there are long-term clinical issues of concern.
Strategy depends on more than age
The “findings are not too surprising,” John Carroll, MD, who was not involved in this research, said in an email.
“Age is only one of multiple patient characteristics that enter into consideration of TAVR versus SAVR,” said Dr. Carroll, from Anschutz Medical Campus, University of Colorado, Aurora.
“As the article reports,” he noted, “those less than 65 having TAVR are more likely to have comorbid conditions that likely made the risk of SAVR higher.”
Dr. Carroll was lead author of a review article published in 2020 based on data from the ACC–Society of Thoracic Surgeons (STS)–Transcatheter Valve Therapy (TVT) registry on 276,316 patients who had TAVR in the United States from 2011-2019.
He pointed out that Figure 2 in that review shows that “SAVR is often performed in conjunction with other surgical procedures – another major reason why SAVR remains an important treatment for valvular heart disease.”
“We are awaiting long-term data comparing TAVR to SAVR durability,” Dr. Carroll added, echoing Dr. Dauerman. “So far [there are] no major differences, but it remains a key need to fully understand TAVR and the various models in commercial use.”
“Both TAVR and SAVR used in adults are tissue valves (SAVR with mechanical valves is used in younger patients),” Dr. Carroll noted, “and all tissue valves will eventually fail if the patient lives long enough.”
Patient management strategies need to consider what treatment options exist when the first valve fails. “If the first valve is SAVR, there is now extensive experience with placing a TAVR valve inside a failing SAVR valve, so called Valve-in-Valve or TAVR-in-SAVR. This is the preferred treatment in most patients with failing SAVR valves,” he said.
“On the other hand,” he continued, “we are just beginning to see more and more patients with failing TAVR valves, and the TAVR-in-TAVR procedure is less well understood.”
“Issues such as acute coronary occlusion and long-term difficulty in accessing coronary arteries are being encountered in some patients having TAVR-in-TAVR,” Dr. Carroll noted, which he discusses in a recent editorial he coauthored about the complexities of redo TAVR, published in JACC: Cardiovascular Interventions.
The study received no funding. Dr. Dauerman has research grants and is a consultant for Medtronic and Boston Scientific. Dr. Carroll is a local principal investigator in trials sponsored by Medtronic, Abbott, and Edwards Lifesciences.
A version of this article first appeared on Medscape.com.
FROM TCT 2022
Novel blood test for early-stage liver cancer shows promise
, increasing the likelihood of potentially curative therapy and improved patient prognosis.
HCC accounts for the majority of primary liver cancers and mainly occurs in patients with cirrhosis or chronic hepatitis B virus infection. The prognosis of HCC is poor, largely owing to advanced disease stage at diagnosis.
The current guidelines recommend surveillance with twice-yearly liver ultrasound, with or without serum alpha-fetoprotein, for patients at risk for HCC, although the diagnostic performance is suboptimal.
The new “liquid biopsy” uses HCC-associated extracellular vesicles (EVs) to establish an HCC EV ECG score for distinguishing patients with early-stage HCC from at-risk controls with cirrhosis from a 400-mcL plasma sample.
“We’re the first team looking at extracellular vesicles as a detection biomarker for early-stage liver cancer, and our study showed it had outstanding performance,” study investigator Ju Dong Yang, MD, with Cedars-Sinai Medical Center, Los Angeles, said in a news release.
The study was published online in Hepatology.
In a phase 2 biomarker (case-control) study, the investigators tested their blood test in a training cohort of 106 individuals (45 patients with treatment-naive early-stage HCC and 61 with cirrhosis) and an independent validation cohort with 72 participants (35 patients with treatment-naive early-stage HCC and 37 with cirrhosis).
The HCC EV ECG score had “excellent accuracy” for discriminating between HCC and cirrhosis, with an area under the receiver operating characteristic curve (AUROC) of 0.95 and 0.93 in the training and validation cohorts, respectively, they report.
The diagnostic performance “remained excellent” among the subpopulations of HCC etiology and those with tumors within the Milan criteria.
Adding the serum alpha-fetoprotein level to the HCC EV ECG score did not improve its performance.
The researchers say that further validation of the blood test in a larger phase 2 study and a subsequent phase 3 study are needed to confirm its utility in clinical settings.
“We are planning on doing larger-scale studies to further validate this test and bring it into routine clinical practice here – and globally,” Dr. Yang said.
“In addition to its excellent performance, this marker has the advantages of being user friendly, cost efficient, and having a fast turnaround time – within 6 hours from sample collection to result,” Dan Theodorescu, MD, PhD, director of Cedars-Sinai Cancer, who is not an author on the study, said in the news release.
“Once this marker has been validated in subsequent studies, it can be easily adopted by existing PCR [polymerase chain reaction] facilities,” Dr. Theodorescu added.
The study was supported by an American College of Gastroenterology Junior Faculty Development Award, a Department of Defense Peer Reviewed Cancer Research Program Career Development Award, and the National Institutes of Health. Dr. Yang provides a consulting service for Exact Sciences, Gilead Sciences, and Eisai. Dr. Theodorescu reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, increasing the likelihood of potentially curative therapy and improved patient prognosis.
HCC accounts for the majority of primary liver cancers and mainly occurs in patients with cirrhosis or chronic hepatitis B virus infection. The prognosis of HCC is poor, largely owing to advanced disease stage at diagnosis.
The current guidelines recommend surveillance with twice-yearly liver ultrasound, with or without serum alpha-fetoprotein, for patients at risk for HCC, although the diagnostic performance is suboptimal.
The new “liquid biopsy” uses HCC-associated extracellular vesicles (EVs) to establish an HCC EV ECG score for distinguishing patients with early-stage HCC from at-risk controls with cirrhosis from a 400-mcL plasma sample.
“We’re the first team looking at extracellular vesicles as a detection biomarker for early-stage liver cancer, and our study showed it had outstanding performance,” study investigator Ju Dong Yang, MD, with Cedars-Sinai Medical Center, Los Angeles, said in a news release.
The study was published online in Hepatology.
In a phase 2 biomarker (case-control) study, the investigators tested their blood test in a training cohort of 106 individuals (45 patients with treatment-naive early-stage HCC and 61 with cirrhosis) and an independent validation cohort with 72 participants (35 patients with treatment-naive early-stage HCC and 37 with cirrhosis).
The HCC EV ECG score had “excellent accuracy” for discriminating between HCC and cirrhosis, with an area under the receiver operating characteristic curve (AUROC) of 0.95 and 0.93 in the training and validation cohorts, respectively, they report.
The diagnostic performance “remained excellent” among the subpopulations of HCC etiology and those with tumors within the Milan criteria.
Adding the serum alpha-fetoprotein level to the HCC EV ECG score did not improve its performance.
The researchers say that further validation of the blood test in a larger phase 2 study and a subsequent phase 3 study are needed to confirm its utility in clinical settings.
“We are planning on doing larger-scale studies to further validate this test and bring it into routine clinical practice here – and globally,” Dr. Yang said.
“In addition to its excellent performance, this marker has the advantages of being user friendly, cost efficient, and having a fast turnaround time – within 6 hours from sample collection to result,” Dan Theodorescu, MD, PhD, director of Cedars-Sinai Cancer, who is not an author on the study, said in the news release.
“Once this marker has been validated in subsequent studies, it can be easily adopted by existing PCR [polymerase chain reaction] facilities,” Dr. Theodorescu added.
The study was supported by an American College of Gastroenterology Junior Faculty Development Award, a Department of Defense Peer Reviewed Cancer Research Program Career Development Award, and the National Institutes of Health. Dr. Yang provides a consulting service for Exact Sciences, Gilead Sciences, and Eisai. Dr. Theodorescu reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, increasing the likelihood of potentially curative therapy and improved patient prognosis.
HCC accounts for the majority of primary liver cancers and mainly occurs in patients with cirrhosis or chronic hepatitis B virus infection. The prognosis of HCC is poor, largely owing to advanced disease stage at diagnosis.
The current guidelines recommend surveillance with twice-yearly liver ultrasound, with or without serum alpha-fetoprotein, for patients at risk for HCC, although the diagnostic performance is suboptimal.
The new “liquid biopsy” uses HCC-associated extracellular vesicles (EVs) to establish an HCC EV ECG score for distinguishing patients with early-stage HCC from at-risk controls with cirrhosis from a 400-mcL plasma sample.
“We’re the first team looking at extracellular vesicles as a detection biomarker for early-stage liver cancer, and our study showed it had outstanding performance,” study investigator Ju Dong Yang, MD, with Cedars-Sinai Medical Center, Los Angeles, said in a news release.
The study was published online in Hepatology.
In a phase 2 biomarker (case-control) study, the investigators tested their blood test in a training cohort of 106 individuals (45 patients with treatment-naive early-stage HCC and 61 with cirrhosis) and an independent validation cohort with 72 participants (35 patients with treatment-naive early-stage HCC and 37 with cirrhosis).
The HCC EV ECG score had “excellent accuracy” for discriminating between HCC and cirrhosis, with an area under the receiver operating characteristic curve (AUROC) of 0.95 and 0.93 in the training and validation cohorts, respectively, they report.
The diagnostic performance “remained excellent” among the subpopulations of HCC etiology and those with tumors within the Milan criteria.
Adding the serum alpha-fetoprotein level to the HCC EV ECG score did not improve its performance.
The researchers say that further validation of the blood test in a larger phase 2 study and a subsequent phase 3 study are needed to confirm its utility in clinical settings.
“We are planning on doing larger-scale studies to further validate this test and bring it into routine clinical practice here – and globally,” Dr. Yang said.
“In addition to its excellent performance, this marker has the advantages of being user friendly, cost efficient, and having a fast turnaround time – within 6 hours from sample collection to result,” Dan Theodorescu, MD, PhD, director of Cedars-Sinai Cancer, who is not an author on the study, said in the news release.
“Once this marker has been validated in subsequent studies, it can be easily adopted by existing PCR [polymerase chain reaction] facilities,” Dr. Theodorescu added.
The study was supported by an American College of Gastroenterology Junior Faculty Development Award, a Department of Defense Peer Reviewed Cancer Research Program Career Development Award, and the National Institutes of Health. Dr. Yang provides a consulting service for Exact Sciences, Gilead Sciences, and Eisai. Dr. Theodorescu reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM HEPATOLOGY
Meta-analysis demonstrates potential of probiotics in reducing atopic dermatitis disease severity
Key clinical point: The use of probiotic supplementation reduced disease severity in adult patients with atopic dermatitis (AD).
Major finding: Probiotic supplementation vs placebo led to a significant reduction in the Scoring AD index (mean difference −7.90; 95% CI −7.25 to−6.92), but no significant improvements in skin severity and itch severity.
Study details: Findings are from a meta-analysis of six randomized controlled trials including 241 adults with AD, of which 128 received probiotics and 113 received placebo.
Disclosures: This study was funded by a grant from Universitas Airlangga, Indonesia. The authors declared no conflicts of interest.
Source: Umborowati MA et al. The role of probiotics in the treatment of adult atopic dermatitis: a meta-analysis of randomized controlled trials. J Health Popul Nutr. 2022;41:37 (Aug 17). Doi: 10.1186/s41043-022-00318-6
Key clinical point: The use of probiotic supplementation reduced disease severity in adult patients with atopic dermatitis (AD).
Major finding: Probiotic supplementation vs placebo led to a significant reduction in the Scoring AD index (mean difference −7.90; 95% CI −7.25 to−6.92), but no significant improvements in skin severity and itch severity.
Study details: Findings are from a meta-analysis of six randomized controlled trials including 241 adults with AD, of which 128 received probiotics and 113 received placebo.
Disclosures: This study was funded by a grant from Universitas Airlangga, Indonesia. The authors declared no conflicts of interest.
Source: Umborowati MA et al. The role of probiotics in the treatment of adult atopic dermatitis: a meta-analysis of randomized controlled trials. J Health Popul Nutr. 2022;41:37 (Aug 17). Doi: 10.1186/s41043-022-00318-6
Key clinical point: The use of probiotic supplementation reduced disease severity in adult patients with atopic dermatitis (AD).
Major finding: Probiotic supplementation vs placebo led to a significant reduction in the Scoring AD index (mean difference −7.90; 95% CI −7.25 to−6.92), but no significant improvements in skin severity and itch severity.
Study details: Findings are from a meta-analysis of six randomized controlled trials including 241 adults with AD, of which 128 received probiotics and 113 received placebo.
Disclosures: This study was funded by a grant from Universitas Airlangga, Indonesia. The authors declared no conflicts of interest.
Source: Umborowati MA et al. The role of probiotics in the treatment of adult atopic dermatitis: a meta-analysis of randomized controlled trials. J Health Popul Nutr. 2022;41:37 (Aug 17). Doi: 10.1186/s41043-022-00318-6
Real-world characteristics of patients with moderate-to-severe atopic dermatitis receiving dupilumab
Key clinical point: Patients who received dupilumab for atopic dermatitis (AD) had moderate-to-severe disease, long medical history, and high prevalence of coexisting type 2 inflammatory diseases.
Major finding: A majority of patients (66.6%) were diagnosed with AD in childhood, and most patients presented with bordering moderate-to-severe AD (Eczema Area and Severity Index > 21), high prevalence of pruritus (99.6%), and coexisting atopic and type 2 inflammatory diseases (51.8%).
Study details: Findings are from an analysis of PROLEAD, a national, multicenter, prospective, non-interventional study, including 817 patients with moderate-to-severe AD who received dupilumab.
Disclosures: This study was funded by Sanofi. Three authors declared being employees of or holding stocks in Sanofi. The other authors reported ties with several sources, including Sanofi.
Source: Thaci D et al. Dupilumab treatment of atopic dermatitis in routine clinical care: Baseline characteristics of patients in the PROLEAD prospective, observational study. Dermatol Ther (Heidelb). 2022;12(9):2145-2160 (Aug 19). Doi: 10.1007/s13555-022-00791-1
Key clinical point: Patients who received dupilumab for atopic dermatitis (AD) had moderate-to-severe disease, long medical history, and high prevalence of coexisting type 2 inflammatory diseases.
Major finding: A majority of patients (66.6%) were diagnosed with AD in childhood, and most patients presented with bordering moderate-to-severe AD (Eczema Area and Severity Index > 21), high prevalence of pruritus (99.6%), and coexisting atopic and type 2 inflammatory diseases (51.8%).
Study details: Findings are from an analysis of PROLEAD, a national, multicenter, prospective, non-interventional study, including 817 patients with moderate-to-severe AD who received dupilumab.
Disclosures: This study was funded by Sanofi. Three authors declared being employees of or holding stocks in Sanofi. The other authors reported ties with several sources, including Sanofi.
Source: Thaci D et al. Dupilumab treatment of atopic dermatitis in routine clinical care: Baseline characteristics of patients in the PROLEAD prospective, observational study. Dermatol Ther (Heidelb). 2022;12(9):2145-2160 (Aug 19). Doi: 10.1007/s13555-022-00791-1
Key clinical point: Patients who received dupilumab for atopic dermatitis (AD) had moderate-to-severe disease, long medical history, and high prevalence of coexisting type 2 inflammatory diseases.
Major finding: A majority of patients (66.6%) were diagnosed with AD in childhood, and most patients presented with bordering moderate-to-severe AD (Eczema Area and Severity Index > 21), high prevalence of pruritus (99.6%), and coexisting atopic and type 2 inflammatory diseases (51.8%).
Study details: Findings are from an analysis of PROLEAD, a national, multicenter, prospective, non-interventional study, including 817 patients with moderate-to-severe AD who received dupilumab.
Disclosures: This study was funded by Sanofi. Three authors declared being employees of or holding stocks in Sanofi. The other authors reported ties with several sources, including Sanofi.
Source: Thaci D et al. Dupilumab treatment of atopic dermatitis in routine clinical care: Baseline characteristics of patients in the PROLEAD prospective, observational study. Dermatol Ther (Heidelb). 2022;12(9):2145-2160 (Aug 19). Doi: 10.1007/s13555-022-00791-1
Pediatric atopic dermatitis and neuropsychiatric disorders: What is the link?
Key clinical point: Atopic dermatitis (AD) did not increase the incidence risk for most neuropsychiatric disorders in a pediatric cohort.
Major finding: The risks for attention deficit hyperactivity disorder (hazard ratio [HR] 1.02; 95% CI 0.97-1.06), autism (HR 1.02; 95% CI 0.98-1.06), anxiety (HR 1.01; 95% CI 0.99-1.03), and bipolar disorder (HR 1.08; 95% CI 0.85-1.36) were comparable in the AD and non-AD groups. Participants with vs without AD were less likely to develop depression (HR 0.93; 95% CI 0.91-0.95) or schizophrenia (HR 0.72; 95% CI 0.54-0.95) but more likely to develop obsessive compulsive disorder (HR 1.26; 95% CI 1.16-1.37). However, the risks varied with disease severity and patient’s age.
Study details: Findings are from a retrospective population-based cohort study including 409,431 children with AD and 1,809,029 matched children without AD.
Disclosures: This study was supported by a contract from Pfizer, Inc. One author declared being an employee of Pfizer. The other authors reported ties with several sources, including Pfizer.
Source: Wan J et al. Atopic dermatitis and risk of major neuropsychiatric disorders in children: A population-based cohort study. J Eur Acad Dermatol Venereol. 2022 (Aug 26). Doi: 10.1111/jdv.18564
Key clinical point: Atopic dermatitis (AD) did not increase the incidence risk for most neuropsychiatric disorders in a pediatric cohort.
Major finding: The risks for attention deficit hyperactivity disorder (hazard ratio [HR] 1.02; 95% CI 0.97-1.06), autism (HR 1.02; 95% CI 0.98-1.06), anxiety (HR 1.01; 95% CI 0.99-1.03), and bipolar disorder (HR 1.08; 95% CI 0.85-1.36) were comparable in the AD and non-AD groups. Participants with vs without AD were less likely to develop depression (HR 0.93; 95% CI 0.91-0.95) or schizophrenia (HR 0.72; 95% CI 0.54-0.95) but more likely to develop obsessive compulsive disorder (HR 1.26; 95% CI 1.16-1.37). However, the risks varied with disease severity and patient’s age.
Study details: Findings are from a retrospective population-based cohort study including 409,431 children with AD and 1,809,029 matched children without AD.
Disclosures: This study was supported by a contract from Pfizer, Inc. One author declared being an employee of Pfizer. The other authors reported ties with several sources, including Pfizer.
Source: Wan J et al. Atopic dermatitis and risk of major neuropsychiatric disorders in children: A population-based cohort study. J Eur Acad Dermatol Venereol. 2022 (Aug 26). Doi: 10.1111/jdv.18564
Key clinical point: Atopic dermatitis (AD) did not increase the incidence risk for most neuropsychiatric disorders in a pediatric cohort.
Major finding: The risks for attention deficit hyperactivity disorder (hazard ratio [HR] 1.02; 95% CI 0.97-1.06), autism (HR 1.02; 95% CI 0.98-1.06), anxiety (HR 1.01; 95% CI 0.99-1.03), and bipolar disorder (HR 1.08; 95% CI 0.85-1.36) were comparable in the AD and non-AD groups. Participants with vs without AD were less likely to develop depression (HR 0.93; 95% CI 0.91-0.95) or schizophrenia (HR 0.72; 95% CI 0.54-0.95) but more likely to develop obsessive compulsive disorder (HR 1.26; 95% CI 1.16-1.37). However, the risks varied with disease severity and patient’s age.
Study details: Findings are from a retrospective population-based cohort study including 409,431 children with AD and 1,809,029 matched children without AD.
Disclosures: This study was supported by a contract from Pfizer, Inc. One author declared being an employee of Pfizer. The other authors reported ties with several sources, including Pfizer.
Source: Wan J et al. Atopic dermatitis and risk of major neuropsychiatric disorders in children: A population-based cohort study. J Eur Acad Dermatol Venereol. 2022 (Aug 26). Doi: 10.1111/jdv.18564
Early initiation of emollient reduces risk for atopic dermatitis in high risk infants
Key clinical point: Initiation of daily application of a specialized emollient from the first to the eighth week of life reduced the risk for atopic dermatitis (AD) incidence for 12 months in infants with high risk for AD.
Major finding: At 12 months, the cumulative incidence of AD was significantly lower in the emollient vs standard routine skin care group (32.8% vs 46.4%; relative risk 0.707; P = .036). The rate of patient-reported skin infections was similar between both the treatment groups during the 8-week intervention period (5.0% vs 5.7%).
Study details: Findings are from the STOP AD trial including 321 newborn infants at high risk for AD who were randomly assigned to receive twice-daily emollient for the first 8 weeks of life or standard routine skin care.
Disclosures: This study was supported by The City of Dublin Skin and Cancer Hospital Charity and the Skin Research Institute of Singapore. Some authors declared being managing directors, employees, shareholders, or consultants or receiving research funding, speaker fees, or consulting fees from several sources.
Source: Ní Chaoimh C, Lad D, et al. Early initiation of short-term emollient use for the prevention of atopic dermatitis in high risk infants - the STOP AD randomised controlled trial. Allergy. 2022 (Aug 23). Doi: 10.1111/all.15491
Key clinical point: Initiation of daily application of a specialized emollient from the first to the eighth week of life reduced the risk for atopic dermatitis (AD) incidence for 12 months in infants with high risk for AD.
Major finding: At 12 months, the cumulative incidence of AD was significantly lower in the emollient vs standard routine skin care group (32.8% vs 46.4%; relative risk 0.707; P = .036). The rate of patient-reported skin infections was similar between both the treatment groups during the 8-week intervention period (5.0% vs 5.7%).
Study details: Findings are from the STOP AD trial including 321 newborn infants at high risk for AD who were randomly assigned to receive twice-daily emollient for the first 8 weeks of life or standard routine skin care.
Disclosures: This study was supported by The City of Dublin Skin and Cancer Hospital Charity and the Skin Research Institute of Singapore. Some authors declared being managing directors, employees, shareholders, or consultants or receiving research funding, speaker fees, or consulting fees from several sources.
Source: Ní Chaoimh C, Lad D, et al. Early initiation of short-term emollient use for the prevention of atopic dermatitis in high risk infants - the STOP AD randomised controlled trial. Allergy. 2022 (Aug 23). Doi: 10.1111/all.15491
Key clinical point: Initiation of daily application of a specialized emollient from the first to the eighth week of life reduced the risk for atopic dermatitis (AD) incidence for 12 months in infants with high risk for AD.
Major finding: At 12 months, the cumulative incidence of AD was significantly lower in the emollient vs standard routine skin care group (32.8% vs 46.4%; relative risk 0.707; P = .036). The rate of patient-reported skin infections was similar between both the treatment groups during the 8-week intervention period (5.0% vs 5.7%).
Study details: Findings are from the STOP AD trial including 321 newborn infants at high risk for AD who were randomly assigned to receive twice-daily emollient for the first 8 weeks of life or standard routine skin care.
Disclosures: This study was supported by The City of Dublin Skin and Cancer Hospital Charity and the Skin Research Institute of Singapore. Some authors declared being managing directors, employees, shareholders, or consultants or receiving research funding, speaker fees, or consulting fees from several sources.
Source: Ní Chaoimh C, Lad D, et al. Early initiation of short-term emollient use for the prevention of atopic dermatitis in high risk infants - the STOP AD randomised controlled trial. Allergy. 2022 (Aug 23). Doi: 10.1111/all.15491





