User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Diffuse Papular Eruption With Erosions and Ulcerations
The Diagnosis: Immunotherapy-Related Lichenoid Drug Eruption
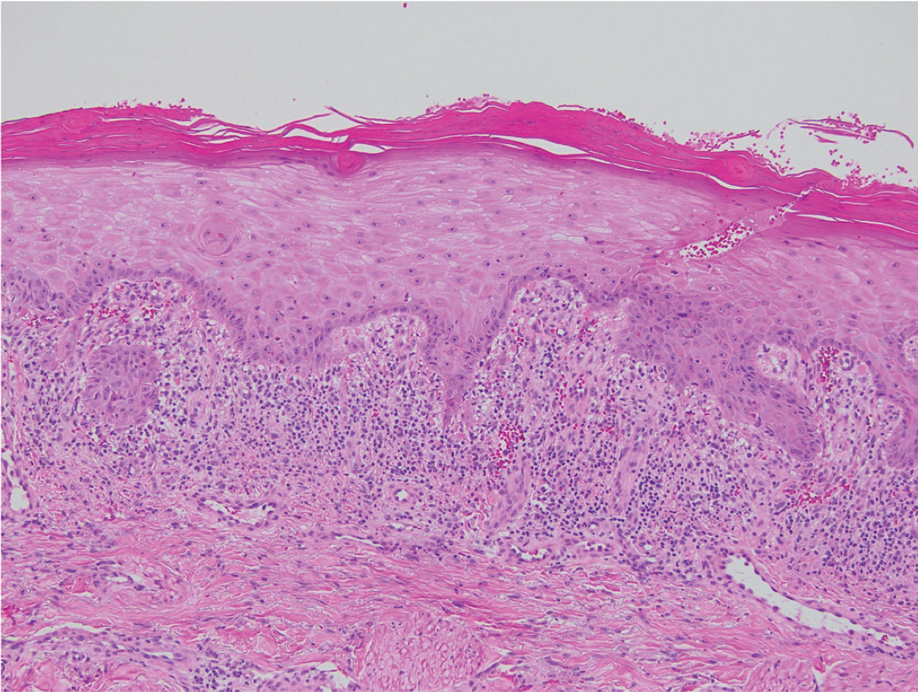
Direct immunofluorescence was negative, and histopathology revealed a lichenoid interface dermatitis, minimal parakeratosis, and saw-toothed rete ridges (Figure 1). He was diagnosed with an immunotherapyrelated lichenoid drug eruption based on the morphology of the skin lesions and clinicopathologic correlation. Bullous pemphigoid and lichen planus pemphigoides were ruled out given the negative direct immunofluorescence findings. Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) was not consistent with the clinical presentation, especially given the lack of mucosal findings. The histology also was not consistent, as the biopsy specimen lacked apoptotic and necrotic keratinocytes to the degree seen in SJS/TEN and also had a greater degree of inflammatory infiltrate. Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome was ruled out given the lack of systemic findings, including facial swelling and lymphadenopathy and the clinical appearance of the rash. No morbilliform features were present, which is the most common presentation of DRESS syndrome.
Checkpoint inhibitor (CPI) therapy has become the cornerstone in management of certain advanced malignancies.1 Checkpoint inhibitors block cytotoxic T lymphocyte–associated protein 4, programmed cell death-1, and/or programmed cell death ligand-1, allowing activated T cells to infiltrate the tumor microenvironment and destroy malignant cells. Checkpoint inhibitors are approved for the treatment of melanoma, cutaneous squamous cell carcinoma, and Merkel cell carcinoma and are being investigated in various other cutaneous and soft tissue malignancies.1-3
Although CPIs have shown substantial efficacy in the management of advanced malignancies, immune-related adverse events (AEs) are common due to nonspecific immune activation.2 Immune-related cutaneous AEs are the most common immune-related AEs, occurring in 30% to 50% of patients who undergo treatment.2-5 Common immune-related cutaneous AEs include maculopapular, psoriasiform, and lichenoid dermatitis, as well as pruritus without dermatitis.2,3,6 Other reactions include but are not limited to bullous pemphigoid, vitiligolike depigmentation, and alopecia.2,3 Immune-related cutaneous AEs usually are self-limited; however, severe life-threatening reactions such as the spectrum of SJS/TEN and DRESS syndrome also can occur.2-4 Immune-related cutaneous AEs are graded based on the Common Terminology Criteria for Adverse Events: grade 1 reactions are asymptomatic and cover less than 10% of the patient’s body surface area (BSA), grade 2 reactions have mild symptoms and cover 10% to 30% of the patient’s BSA, grade 3 reactions have moderate to severe symptoms and cover greater than 30% of the patient’s BSA, and grade 4 reactions are life-threatening.2,3 With prompt recognition and adequate treatment, mild to moderate immune-related cutaneous AEs—grades 1 and 2—largely are reversible, and less than 5% require discontinuation of therapy.2,3,6 It has been suggested that immune-related cutaneous AEs may be a positive prognostic factor in the treatment of underlying malignancy, indicating adequate immune activation targeting the malignant cells.6
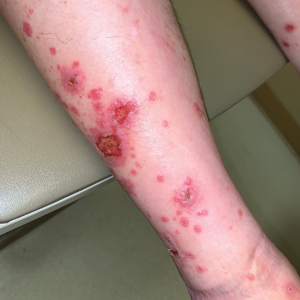
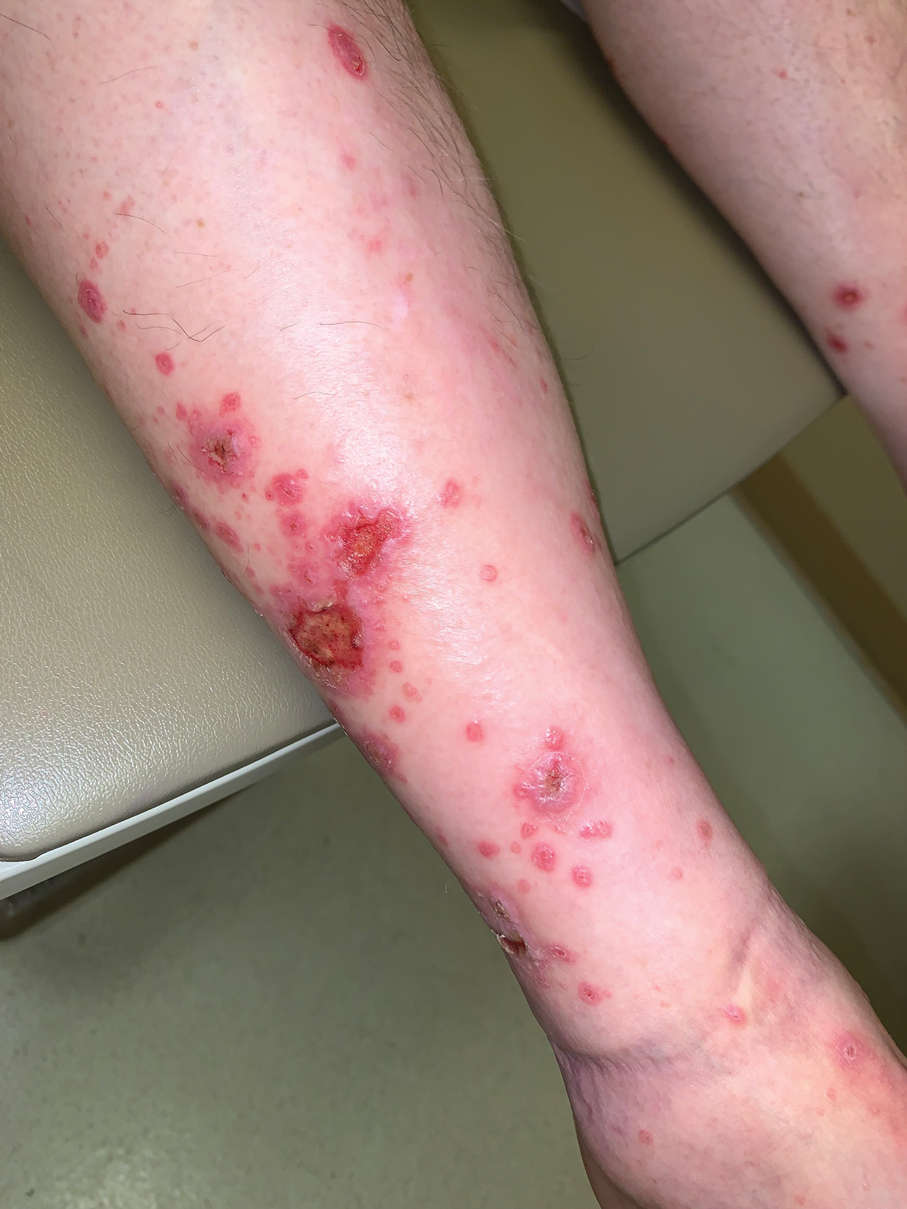
Although our patient had some typical violaceous, flat-topped papules and plaques with Wickham striae, he also had atypical findings for a lichenoid reaction. Given the endorsement of blisters, it is possible that some of these lesions initially were bullous and subsequently ruptured, leaving behind erosions. However, in other areas, there also were eroded papules and ulcerations without a reported history of excoriation, scratching, picking, or prior bullae, including difficult-to-reach areas such as the back. It is favored that these lesions represented a robust lichenoid dermatitis leading to erosive and ulcerated lesions, similar to the formation of bullous lichen planus. Lichenoid eruptions secondary to immunotherapy are well-known phenomena, but a PubMed search of articles indexed for MEDLINE using the terms ulcer, lichenoid, and immunotherapy revealed only 2 cases of ulcerative lichenoid eruptions: a localized digital erosive lichenoid dermatitis and a widespread ulcerative lichenoid drug eruption without true erosions.7,8 However, widespread erosive and ulcerated lichenoid reactions are rare.
Lichenoid eruptions most strongly are associated with anti–programmed cell death-1/ programmed cell death ligand-1 therapy, occurring in 20% of patients undergoing treatment.3 Lichenoid eruptions present as discrete, pruritic, erythematous, violaceous papules and plaques on the chest and back and rarely may involve the limbs, palmoplantar surfaces, and oral mucosa.2,3,6 Histopathologic features include a dense bandlike lymphocytic infiltrate in the dermis with scattered apoptotic keratinocytes in the basal layer of the epidermis.2,4,6 Grades 1 to 2 lesions can be managed with high-potency topical corticosteroids without CPI dose interruption, with more extensive grade 2 lesions requiring systemic corticosteroids.2,6,9 Lichenoid eruptions grade 3 or higher also require systemic corticosteroid therapy CPI therapy cessation until the eruption has receded to grade 0 to 1.2 Alternative treatment options for high-grade toxicity include phototherapy and acitretin.2,4,9
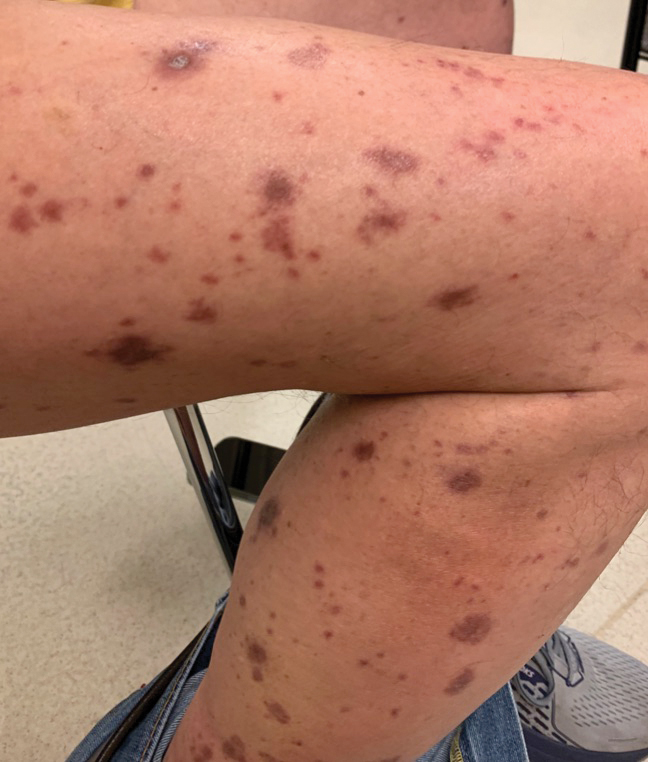
Our patient was treated with cessation of immunotherapy and initiation of a systemic corticosteroid taper, acitretin, and narrowband UVB therapy. After 6 weeks of treatment, the pain and pruritus improved and the rash had resolved in some areas while it had taken on a more classic lichenoid appearance with violaceous scaly papules and plaques (Figure 2) in areas of prior ulcers and erosions. He no longer had any bullae, erosions, or ulcers.
- Barrios DM, Do MH, Phillips GS, et al. Immune checkpoint inhibitors to treat cutaneous malignancies. J Am Acad Dermatol. 2020;83:1239-1253. doi:10.1016/j.jaad.2020.03.131
- Geisler AN, Phillips GS, Barrios DM, et al. Immune checkpoint inhibitor-related dermatologic adverse events. J Am Acad Dermatol. 2020;83:1255-1268. doi:10.1016/j.jaad.2020.03.132
- Tattersall IW, Leventhal JS. Cutaneous toxicities of immune checkpoint inhibitors: the role of the dermatologist. Yale J Biol Med. 2020;93:123-132.
- Si X, He C, Zhang L, et al. Management of immune checkpoint inhibitor-related dermatologic adverse events. Thorac Cancer. 2020;11:488-492. doi:10.1111/1759-7714.13275
- Eggermont AMM, Kicinski M, Blank CU, et al. Association between immune-related adverse events and recurrence-free survival among patients with stage III melanoma randomized to receive pembrolizumab or placebo: a secondary analysis of a randomized clinical trial. JAMA Oncol. 2020;6:519-527. doi:10.1001 /jamaoncol.2019.5570
- Sibaud V, Meyer N, Lamant L, et al. Dermatologic complications of anti-PD-1/PD-L1 immune checkpoint antibodies. Curr Opin Oncol. 2016;28:254-263. doi:10.1097/CCO.0000000000000290
- Martínez-Doménech Á, García-Legaz Martínez M, Magdaleno-Tapial J, et al. Digital ulcerative lichenoid dermatitis in a patient receiving anti-PD-1 therapy. Dermatol Online J. 2019;25:13030/qt8sm0j7t7.
- Davis MJ, Wilken R, Fung MA, et al. Debilitating erosive lichenoid interface dermatitis from checkpoint inhibitor therapy. Dermatol Online J. 2018;24:13030/qt3vq6b04v.
- Apalla Z, Papageorgiou C, Lallas A, et al. Cutaneous adverse events of immune checkpoint inhibitors: a literature review [published online January 29, 2021]. Dermatol Pract Concept. 2021;11:E2021155. doi:10.5826/dpc.1101a155
The Diagnosis: Immunotherapy-Related Lichenoid Drug Eruption
Direct immunofluorescence was negative, and histopathology revealed a lichenoid interface dermatitis, minimal parakeratosis, and saw-toothed rete ridges (Figure 1). He was diagnosed with an immunotherapyrelated lichenoid drug eruption based on the morphology of the skin lesions and clinicopathologic correlation. Bullous pemphigoid and lichen planus pemphigoides were ruled out given the negative direct immunofluorescence findings. Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) was not consistent with the clinical presentation, especially given the lack of mucosal findings. The histology also was not consistent, as the biopsy specimen lacked apoptotic and necrotic keratinocytes to the degree seen in SJS/TEN and also had a greater degree of inflammatory infiltrate. Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome was ruled out given the lack of systemic findings, including facial swelling and lymphadenopathy and the clinical appearance of the rash. No morbilliform features were present, which is the most common presentation of DRESS syndrome.

Checkpoint inhibitor (CPI) therapy has become the cornerstone in management of certain advanced malignancies.1 Checkpoint inhibitors block cytotoxic T lymphocyte–associated protein 4, programmed cell death-1, and/or programmed cell death ligand-1, allowing activated T cells to infiltrate the tumor microenvironment and destroy malignant cells. Checkpoint inhibitors are approved for the treatment of melanoma, cutaneous squamous cell carcinoma, and Merkel cell carcinoma and are being investigated in various other cutaneous and soft tissue malignancies.1-3
Although CPIs have shown substantial efficacy in the management of advanced malignancies, immune-related adverse events (AEs) are common due to nonspecific immune activation.2 Immune-related cutaneous AEs are the most common immune-related AEs, occurring in 30% to 50% of patients who undergo treatment.2-5 Common immune-related cutaneous AEs include maculopapular, psoriasiform, and lichenoid dermatitis, as well as pruritus without dermatitis.2,3,6 Other reactions include but are not limited to bullous pemphigoid, vitiligolike depigmentation, and alopecia.2,3 Immune-related cutaneous AEs usually are self-limited; however, severe life-threatening reactions such as the spectrum of SJS/TEN and DRESS syndrome also can occur.2-4 Immune-related cutaneous AEs are graded based on the Common Terminology Criteria for Adverse Events: grade 1 reactions are asymptomatic and cover less than 10% of the patient’s body surface area (BSA), grade 2 reactions have mild symptoms and cover 10% to 30% of the patient’s BSA, grade 3 reactions have moderate to severe symptoms and cover greater than 30% of the patient’s BSA, and grade 4 reactions are life-threatening.2,3 With prompt recognition and adequate treatment, mild to moderate immune-related cutaneous AEs—grades 1 and 2—largely are reversible, and less than 5% require discontinuation of therapy.2,3,6 It has been suggested that immune-related cutaneous AEs may be a positive prognostic factor in the treatment of underlying malignancy, indicating adequate immune activation targeting the malignant cells.6
Although our patient had some typical violaceous, flat-topped papules and plaques with Wickham striae, he also had atypical findings for a lichenoid reaction. Given the endorsement of blisters, it is possible that some of these lesions initially were bullous and subsequently ruptured, leaving behind erosions. However, in other areas, there also were eroded papules and ulcerations without a reported history of excoriation, scratching, picking, or prior bullae, including difficult-to-reach areas such as the back. It is favored that these lesions represented a robust lichenoid dermatitis leading to erosive and ulcerated lesions, similar to the formation of bullous lichen planus. Lichenoid eruptions secondary to immunotherapy are well-known phenomena, but a PubMed search of articles indexed for MEDLINE using the terms ulcer, lichenoid, and immunotherapy revealed only 2 cases of ulcerative lichenoid eruptions: a localized digital erosive lichenoid dermatitis and a widespread ulcerative lichenoid drug eruption without true erosions.7,8 However, widespread erosive and ulcerated lichenoid reactions are rare.
Lichenoid eruptions most strongly are associated with anti–programmed cell death-1/ programmed cell death ligand-1 therapy, occurring in 20% of patients undergoing treatment.3 Lichenoid eruptions present as discrete, pruritic, erythematous, violaceous papules and plaques on the chest and back and rarely may involve the limbs, palmoplantar surfaces, and oral mucosa.2,3,6 Histopathologic features include a dense bandlike lymphocytic infiltrate in the dermis with scattered apoptotic keratinocytes in the basal layer of the epidermis.2,4,6 Grades 1 to 2 lesions can be managed with high-potency topical corticosteroids without CPI dose interruption, with more extensive grade 2 lesions requiring systemic corticosteroids.2,6,9 Lichenoid eruptions grade 3 or higher also require systemic corticosteroid therapy CPI therapy cessation until the eruption has receded to grade 0 to 1.2 Alternative treatment options for high-grade toxicity include phototherapy and acitretin.2,4,9
Our patient was treated with cessation of immunotherapy and initiation of a systemic corticosteroid taper, acitretin, and narrowband UVB therapy. After 6 weeks of treatment, the pain and pruritus improved and the rash had resolved in some areas while it had taken on a more classic lichenoid appearance with violaceous scaly papules and plaques (Figure 2) in areas of prior ulcers and erosions. He no longer had any bullae, erosions, or ulcers.

The Diagnosis: Immunotherapy-Related Lichenoid Drug Eruption
Direct immunofluorescence was negative, and histopathology revealed a lichenoid interface dermatitis, minimal parakeratosis, and saw-toothed rete ridges (Figure 1). He was diagnosed with an immunotherapyrelated lichenoid drug eruption based on the morphology of the skin lesions and clinicopathologic correlation. Bullous pemphigoid and lichen planus pemphigoides were ruled out given the negative direct immunofluorescence findings. Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) was not consistent with the clinical presentation, especially given the lack of mucosal findings. The histology also was not consistent, as the biopsy specimen lacked apoptotic and necrotic keratinocytes to the degree seen in SJS/TEN and also had a greater degree of inflammatory infiltrate. Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome was ruled out given the lack of systemic findings, including facial swelling and lymphadenopathy and the clinical appearance of the rash. No morbilliform features were present, which is the most common presentation of DRESS syndrome.

Checkpoint inhibitor (CPI) therapy has become the cornerstone in management of certain advanced malignancies.1 Checkpoint inhibitors block cytotoxic T lymphocyte–associated protein 4, programmed cell death-1, and/or programmed cell death ligand-1, allowing activated T cells to infiltrate the tumor microenvironment and destroy malignant cells. Checkpoint inhibitors are approved for the treatment of melanoma, cutaneous squamous cell carcinoma, and Merkel cell carcinoma and are being investigated in various other cutaneous and soft tissue malignancies.1-3
Although CPIs have shown substantial efficacy in the management of advanced malignancies, immune-related adverse events (AEs) are common due to nonspecific immune activation.2 Immune-related cutaneous AEs are the most common immune-related AEs, occurring in 30% to 50% of patients who undergo treatment.2-5 Common immune-related cutaneous AEs include maculopapular, psoriasiform, and lichenoid dermatitis, as well as pruritus without dermatitis.2,3,6 Other reactions include but are not limited to bullous pemphigoid, vitiligolike depigmentation, and alopecia.2,3 Immune-related cutaneous AEs usually are self-limited; however, severe life-threatening reactions such as the spectrum of SJS/TEN and DRESS syndrome also can occur.2-4 Immune-related cutaneous AEs are graded based on the Common Terminology Criteria for Adverse Events: grade 1 reactions are asymptomatic and cover less than 10% of the patient’s body surface area (BSA), grade 2 reactions have mild symptoms and cover 10% to 30% of the patient’s BSA, grade 3 reactions have moderate to severe symptoms and cover greater than 30% of the patient’s BSA, and grade 4 reactions are life-threatening.2,3 With prompt recognition and adequate treatment, mild to moderate immune-related cutaneous AEs—grades 1 and 2—largely are reversible, and less than 5% require discontinuation of therapy.2,3,6 It has been suggested that immune-related cutaneous AEs may be a positive prognostic factor in the treatment of underlying malignancy, indicating adequate immune activation targeting the malignant cells.6
Although our patient had some typical violaceous, flat-topped papules and plaques with Wickham striae, he also had atypical findings for a lichenoid reaction. Given the endorsement of blisters, it is possible that some of these lesions initially were bullous and subsequently ruptured, leaving behind erosions. However, in other areas, there also were eroded papules and ulcerations without a reported history of excoriation, scratching, picking, or prior bullae, including difficult-to-reach areas such as the back. It is favored that these lesions represented a robust lichenoid dermatitis leading to erosive and ulcerated lesions, similar to the formation of bullous lichen planus. Lichenoid eruptions secondary to immunotherapy are well-known phenomena, but a PubMed search of articles indexed for MEDLINE using the terms ulcer, lichenoid, and immunotherapy revealed only 2 cases of ulcerative lichenoid eruptions: a localized digital erosive lichenoid dermatitis and a widespread ulcerative lichenoid drug eruption without true erosions.7,8 However, widespread erosive and ulcerated lichenoid reactions are rare.
Lichenoid eruptions most strongly are associated with anti–programmed cell death-1/ programmed cell death ligand-1 therapy, occurring in 20% of patients undergoing treatment.3 Lichenoid eruptions present as discrete, pruritic, erythematous, violaceous papules and plaques on the chest and back and rarely may involve the limbs, palmoplantar surfaces, and oral mucosa.2,3,6 Histopathologic features include a dense bandlike lymphocytic infiltrate in the dermis with scattered apoptotic keratinocytes in the basal layer of the epidermis.2,4,6 Grades 1 to 2 lesions can be managed with high-potency topical corticosteroids without CPI dose interruption, with more extensive grade 2 lesions requiring systemic corticosteroids.2,6,9 Lichenoid eruptions grade 3 or higher also require systemic corticosteroid therapy CPI therapy cessation until the eruption has receded to grade 0 to 1.2 Alternative treatment options for high-grade toxicity include phototherapy and acitretin.2,4,9
Our patient was treated with cessation of immunotherapy and initiation of a systemic corticosteroid taper, acitretin, and narrowband UVB therapy. After 6 weeks of treatment, the pain and pruritus improved and the rash had resolved in some areas while it had taken on a more classic lichenoid appearance with violaceous scaly papules and plaques (Figure 2) in areas of prior ulcers and erosions. He no longer had any bullae, erosions, or ulcers.

- Barrios DM, Do MH, Phillips GS, et al. Immune checkpoint inhibitors to treat cutaneous malignancies. J Am Acad Dermatol. 2020;83:1239-1253. doi:10.1016/j.jaad.2020.03.131
- Geisler AN, Phillips GS, Barrios DM, et al. Immune checkpoint inhibitor-related dermatologic adverse events. J Am Acad Dermatol. 2020;83:1255-1268. doi:10.1016/j.jaad.2020.03.132
- Tattersall IW, Leventhal JS. Cutaneous toxicities of immune checkpoint inhibitors: the role of the dermatologist. Yale J Biol Med. 2020;93:123-132.
- Si X, He C, Zhang L, et al. Management of immune checkpoint inhibitor-related dermatologic adverse events. Thorac Cancer. 2020;11:488-492. doi:10.1111/1759-7714.13275
- Eggermont AMM, Kicinski M, Blank CU, et al. Association between immune-related adverse events and recurrence-free survival among patients with stage III melanoma randomized to receive pembrolizumab or placebo: a secondary analysis of a randomized clinical trial. JAMA Oncol. 2020;6:519-527. doi:10.1001 /jamaoncol.2019.5570
- Sibaud V, Meyer N, Lamant L, et al. Dermatologic complications of anti-PD-1/PD-L1 immune checkpoint antibodies. Curr Opin Oncol. 2016;28:254-263. doi:10.1097/CCO.0000000000000290
- Martínez-Doménech Á, García-Legaz Martínez M, Magdaleno-Tapial J, et al. Digital ulcerative lichenoid dermatitis in a patient receiving anti-PD-1 therapy. Dermatol Online J. 2019;25:13030/qt8sm0j7t7.
- Davis MJ, Wilken R, Fung MA, et al. Debilitating erosive lichenoid interface dermatitis from checkpoint inhibitor therapy. Dermatol Online J. 2018;24:13030/qt3vq6b04v.
- Apalla Z, Papageorgiou C, Lallas A, et al. Cutaneous adverse events of immune checkpoint inhibitors: a literature review [published online January 29, 2021]. Dermatol Pract Concept. 2021;11:E2021155. doi:10.5826/dpc.1101a155
- Barrios DM, Do MH, Phillips GS, et al. Immune checkpoint inhibitors to treat cutaneous malignancies. J Am Acad Dermatol. 2020;83:1239-1253. doi:10.1016/j.jaad.2020.03.131
- Geisler AN, Phillips GS, Barrios DM, et al. Immune checkpoint inhibitor-related dermatologic adverse events. J Am Acad Dermatol. 2020;83:1255-1268. doi:10.1016/j.jaad.2020.03.132
- Tattersall IW, Leventhal JS. Cutaneous toxicities of immune checkpoint inhibitors: the role of the dermatologist. Yale J Biol Med. 2020;93:123-132.
- Si X, He C, Zhang L, et al. Management of immune checkpoint inhibitor-related dermatologic adverse events. Thorac Cancer. 2020;11:488-492. doi:10.1111/1759-7714.13275
- Eggermont AMM, Kicinski M, Blank CU, et al. Association between immune-related adverse events and recurrence-free survival among patients with stage III melanoma randomized to receive pembrolizumab or placebo: a secondary analysis of a randomized clinical trial. JAMA Oncol. 2020;6:519-527. doi:10.1001 /jamaoncol.2019.5570
- Sibaud V, Meyer N, Lamant L, et al. Dermatologic complications of anti-PD-1/PD-L1 immune checkpoint antibodies. Curr Opin Oncol. 2016;28:254-263. doi:10.1097/CCO.0000000000000290
- Martínez-Doménech Á, García-Legaz Martínez M, Magdaleno-Tapial J, et al. Digital ulcerative lichenoid dermatitis in a patient receiving anti-PD-1 therapy. Dermatol Online J. 2019;25:13030/qt8sm0j7t7.
- Davis MJ, Wilken R, Fung MA, et al. Debilitating erosive lichenoid interface dermatitis from checkpoint inhibitor therapy. Dermatol Online J. 2018;24:13030/qt3vq6b04v.
- Apalla Z, Papageorgiou C, Lallas A, et al. Cutaneous adverse events of immune checkpoint inhibitors: a literature review [published online January 29, 2021]. Dermatol Pract Concept. 2021;11:E2021155. doi:10.5826/dpc.1101a155
A 70-year-old man presented with a painful, pruritic, diffuse eruption on the trunk, legs, and arms of 2 months’ duration. He had a history of stage IV pleomorphic cell sarcoma of the retroperitoneum and was started on pembrolizumab therapy 6 weeks prior to the eruption. Physical examination revealed violaceous papules and plaques with shiny reticulated scaling as well as multiple scattered eroded papules and shallow ulcerations. The oral mucosa and genitals were spared. The patient endorsed blisters followed by open sores that were both itchy and painful. He denied self-infliction. Both the patient and his wife denied scratching. Two biopsies for direct immunofluorescence and histopathology were performed.
Mortality after acute stroke worsened by accompanying acute AFib
The study covered in this summary was published on ResearchSquare.com as a preprint and has not yet been peer-reviewed.
Key takeaway
Why this matters
- A comprehensive understanding of the relationship between acute AF and risk for acute ischemic stroke and prognosis will help improve management and treatment of patients with acute ischemic stroke.
Study design
- The retrospective study included patients with acute ischemic stroke within the prior 24 hours; 12-lead electrocardiogram in the emergency department; and hospitalization and treatment at the hospital stroke center.
- The cohort of 706 patients admitted to a single center in Shanghai, China, from December 2019 to December 2021, included 142 with episodes of acute AF and 564 without such episodes.
- Patients with acute ischemic stroke and acute AF – including AF of new onset, paroxysmal, persistent, or permanent with symptoms such as palpitations or dizziness attributed to rapid ventricular rates – were identified.
- Neurological deficits were assessed using the 7-day National Institutes of Health Stroke Scale/Score (NIHSS). Patients with a 7-day NIHSS score of at least 16 were considered to have moderate to severe stroke.
- Associations between acute AF onset and the severity of early neurological deficits were assessed and related to all-cause mortality within 30 days of the stroke.
Key results
- Patients with acute AF were older than those without acute AF (80.3 years vs. 71.0 years; P < .001).
- Baseline NIHSS scores averaged 16.09 for the stroke patients with acute AF and 8.65 for those without acute AF (P < .001).
- Significantly more patients with acute AF than without acute AF had a 7-day NIHSS score of at least 16 (45.1% vs. 14.4%; P < .001).
- More patients with than without acute AF underwent transcatheter thrombectomy (44.4% vs. 24.5%; P < .001) or received thrombolytic therapy (31.6% vs. 19.7%; P = .005).
- Patients aged 73 years or older showed baseline NIHSS score and acute AF as independent risk factors for early neurological deficits in stroke patients admitted to the emergency department.
- Mortality at 30 days was significantly higher in patients with acute AF than in those without acute AF (30.3% vs. 10.1%; P < .001).
- Baseline NIHSS had an adjusted odds ratio for 30-day mortality of 1.18 (95% confidence interval, 1.15-1.22; P < .001).
- Other independent predictors included acute AF (1.87 [95% CI, 1.09-3.19; P = .022]) and age 73 or older (2.00 [95% CI, 1.18-3.37; P = .01]).
Limitations
- The study was retrospective and didn’t have access to some potentially relevant data, such as duration of AF.
- The single-center study with limited generalizability does not necessarily represent the broad population of stroke patients in China or elsewhere.
Disclosures
- This study was supported by the Cardiovascular Multidisciplinary Integrated Research Fund and Construction of Shanghai Municipal Health Commission.
- The authors report no relevant financial relationships.
This is a summary of a preprint research study, “Acute Atrial Fibrillation During Onset of Stroke Indicates Higher Probability of Post-Stroke Death Outcomes,” written by Yongxia Li, from the Shanghai Sixth People’s Hospital, and colleagues, on ResearchSquare.com. This study has not yet been peer reviewed. The full text of the study can be found on ResearchSquare.com.A version of this article first appeared on Medscape.com.
The study covered in this summary was published on ResearchSquare.com as a preprint and has not yet been peer-reviewed.
Key takeaway
Why this matters
- A comprehensive understanding of the relationship between acute AF and risk for acute ischemic stroke and prognosis will help improve management and treatment of patients with acute ischemic stroke.
Study design
- The retrospective study included patients with acute ischemic stroke within the prior 24 hours; 12-lead electrocardiogram in the emergency department; and hospitalization and treatment at the hospital stroke center.
- The cohort of 706 patients admitted to a single center in Shanghai, China, from December 2019 to December 2021, included 142 with episodes of acute AF and 564 without such episodes.
- Patients with acute ischemic stroke and acute AF – including AF of new onset, paroxysmal, persistent, or permanent with symptoms such as palpitations or dizziness attributed to rapid ventricular rates – were identified.
- Neurological deficits were assessed using the 7-day National Institutes of Health Stroke Scale/Score (NIHSS). Patients with a 7-day NIHSS score of at least 16 were considered to have moderate to severe stroke.
- Associations between acute AF onset and the severity of early neurological deficits were assessed and related to all-cause mortality within 30 days of the stroke.
Key results
- Patients with acute AF were older than those without acute AF (80.3 years vs. 71.0 years; P < .001).
- Baseline NIHSS scores averaged 16.09 for the stroke patients with acute AF and 8.65 for those without acute AF (P < .001).
- Significantly more patients with acute AF than without acute AF had a 7-day NIHSS score of at least 16 (45.1% vs. 14.4%; P < .001).
- More patients with than without acute AF underwent transcatheter thrombectomy (44.4% vs. 24.5%; P < .001) or received thrombolytic therapy (31.6% vs. 19.7%; P = .005).
- Patients aged 73 years or older showed baseline NIHSS score and acute AF as independent risk factors for early neurological deficits in stroke patients admitted to the emergency department.
- Mortality at 30 days was significantly higher in patients with acute AF than in those without acute AF (30.3% vs. 10.1%; P < .001).
- Baseline NIHSS had an adjusted odds ratio for 30-day mortality of 1.18 (95% confidence interval, 1.15-1.22; P < .001).
- Other independent predictors included acute AF (1.87 [95% CI, 1.09-3.19; P = .022]) and age 73 or older (2.00 [95% CI, 1.18-3.37; P = .01]).
Limitations
- The study was retrospective and didn’t have access to some potentially relevant data, such as duration of AF.
- The single-center study with limited generalizability does not necessarily represent the broad population of stroke patients in China or elsewhere.
Disclosures
- This study was supported by the Cardiovascular Multidisciplinary Integrated Research Fund and Construction of Shanghai Municipal Health Commission.
- The authors report no relevant financial relationships.
This is a summary of a preprint research study, “Acute Atrial Fibrillation During Onset of Stroke Indicates Higher Probability of Post-Stroke Death Outcomes,” written by Yongxia Li, from the Shanghai Sixth People’s Hospital, and colleagues, on ResearchSquare.com. This study has not yet been peer reviewed. The full text of the study can be found on ResearchSquare.com.A version of this article first appeared on Medscape.com.
The study covered in this summary was published on ResearchSquare.com as a preprint and has not yet been peer-reviewed.
Key takeaway
Why this matters
- A comprehensive understanding of the relationship between acute AF and risk for acute ischemic stroke and prognosis will help improve management and treatment of patients with acute ischemic stroke.
Study design
- The retrospective study included patients with acute ischemic stroke within the prior 24 hours; 12-lead electrocardiogram in the emergency department; and hospitalization and treatment at the hospital stroke center.
- The cohort of 706 patients admitted to a single center in Shanghai, China, from December 2019 to December 2021, included 142 with episodes of acute AF and 564 without such episodes.
- Patients with acute ischemic stroke and acute AF – including AF of new onset, paroxysmal, persistent, or permanent with symptoms such as palpitations or dizziness attributed to rapid ventricular rates – were identified.
- Neurological deficits were assessed using the 7-day National Institutes of Health Stroke Scale/Score (NIHSS). Patients with a 7-day NIHSS score of at least 16 were considered to have moderate to severe stroke.
- Associations between acute AF onset and the severity of early neurological deficits were assessed and related to all-cause mortality within 30 days of the stroke.
Key results
- Patients with acute AF were older than those without acute AF (80.3 years vs. 71.0 years; P < .001).
- Baseline NIHSS scores averaged 16.09 for the stroke patients with acute AF and 8.65 for those without acute AF (P < .001).
- Significantly more patients with acute AF than without acute AF had a 7-day NIHSS score of at least 16 (45.1% vs. 14.4%; P < .001).
- More patients with than without acute AF underwent transcatheter thrombectomy (44.4% vs. 24.5%; P < .001) or received thrombolytic therapy (31.6% vs. 19.7%; P = .005).
- Patients aged 73 years or older showed baseline NIHSS score and acute AF as independent risk factors for early neurological deficits in stroke patients admitted to the emergency department.
- Mortality at 30 days was significantly higher in patients with acute AF than in those without acute AF (30.3% vs. 10.1%; P < .001).
- Baseline NIHSS had an adjusted odds ratio for 30-day mortality of 1.18 (95% confidence interval, 1.15-1.22; P < .001).
- Other independent predictors included acute AF (1.87 [95% CI, 1.09-3.19; P = .022]) and age 73 or older (2.00 [95% CI, 1.18-3.37; P = .01]).
Limitations
- The study was retrospective and didn’t have access to some potentially relevant data, such as duration of AF.
- The single-center study with limited generalizability does not necessarily represent the broad population of stroke patients in China or elsewhere.
Disclosures
- This study was supported by the Cardiovascular Multidisciplinary Integrated Research Fund and Construction of Shanghai Municipal Health Commission.
- The authors report no relevant financial relationships.
This is a summary of a preprint research study, “Acute Atrial Fibrillation During Onset of Stroke Indicates Higher Probability of Post-Stroke Death Outcomes,” written by Yongxia Li, from the Shanghai Sixth People’s Hospital, and colleagues, on ResearchSquare.com. This study has not yet been peer reviewed. The full text of the study can be found on ResearchSquare.com.A version of this article first appeared on Medscape.com.
Major life stressors ‘strongly predictive’ of long COVID symptoms
new research suggests.
Major life stressors in the year after hospital discharge for COVID-19 are “strongly predictive of a lot of the important outcomes that people may face after COVID,” lead investigator Jennifer A. Frontera, MD, a professor in the department of neurology at New York University Langone Health, said in an interview.
These outcomes include depression, brain fog, fatigue, trouble sleeping, and other long COVID symptoms.
The findings were published online in the Journal of the Neurological Sciences.
Major stressful events common
Dr. Frontera and the NYU Neurology COVID-19 study team evaluated 451 adults who survived a COVID hospital stay. Of these, 383 completed a 6-month follow-up, 242 completed a 12-month follow-up, and 174 completed follow-up at both time points.
Within 1 year of discharge, 77 (17%) patients died and 51% suffered a major stressful life event.
In multivariable analyses, major life stressors – including financial insecurity, food insecurity, death of a close contact, and new disability – were strong independent predictors of disability, trouble with activities of daily living, depression, fatigue, sleep problems, and prolonged post-acute COVID symptoms. The adjusted odds ratios for these outcomes ranged from 2.5 to 20.8.
The research also confirmed the contribution of traditional risk factors for long COVID symptoms, as shown in past studies. These include older age, poor pre-COVID functional status, and more severe initial COVID-19 infection.
Long-term sequelae of COVID are increasingly recognized as major public health issues.
It has been estimated that roughly 16 million U.S. adults aged 18-65 years ave long COVID, with the often debilitating symptoms keeping up to 4 million out of work.
Holistic approach
Dr. Frontera said it’s important to realize that “sleep, fatigue, anxiety, depression, even cognition are so interwoven with each other that anything that impacts any one of them could have repercussions on the other.”
She added that it “certainly makes sense that there is an interplay or even a bidirectional relationship between the stressors that people face and how well they can recover after COVID.”
Therapies that lessen the trauma of the most stress-inducing life events need to be a central part of treatment for long COVID, with more research needed to validate the best approaches, Dr. Frontera said.
She also noted that social services or case management resources may be able to help address at least some of the stressors that individuals are under – and it is important to refer them to these resources. Referral to mental health services is also important.
“I think it’s really important to take a holistic approach and try to deal with whatever the problem may be,” said Dr. Frontera.
“I’m a neurologist, but as part of my evaluation, I really need to address if there are life stressors or mental health issues that may be impacting this person’s function,” she added.
The study had no commercial funding. The investigators reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
Major life stressors in the year after hospital discharge for COVID-19 are “strongly predictive of a lot of the important outcomes that people may face after COVID,” lead investigator Jennifer A. Frontera, MD, a professor in the department of neurology at New York University Langone Health, said in an interview.
These outcomes include depression, brain fog, fatigue, trouble sleeping, and other long COVID symptoms.
The findings were published online in the Journal of the Neurological Sciences.
Major stressful events common
Dr. Frontera and the NYU Neurology COVID-19 study team evaluated 451 adults who survived a COVID hospital stay. Of these, 383 completed a 6-month follow-up, 242 completed a 12-month follow-up, and 174 completed follow-up at both time points.
Within 1 year of discharge, 77 (17%) patients died and 51% suffered a major stressful life event.
In multivariable analyses, major life stressors – including financial insecurity, food insecurity, death of a close contact, and new disability – were strong independent predictors of disability, trouble with activities of daily living, depression, fatigue, sleep problems, and prolonged post-acute COVID symptoms. The adjusted odds ratios for these outcomes ranged from 2.5 to 20.8.
The research also confirmed the contribution of traditional risk factors for long COVID symptoms, as shown in past studies. These include older age, poor pre-COVID functional status, and more severe initial COVID-19 infection.
Long-term sequelae of COVID are increasingly recognized as major public health issues.
It has been estimated that roughly 16 million U.S. adults aged 18-65 years ave long COVID, with the often debilitating symptoms keeping up to 4 million out of work.
Holistic approach
Dr. Frontera said it’s important to realize that “sleep, fatigue, anxiety, depression, even cognition are so interwoven with each other that anything that impacts any one of them could have repercussions on the other.”
She added that it “certainly makes sense that there is an interplay or even a bidirectional relationship between the stressors that people face and how well they can recover after COVID.”
Therapies that lessen the trauma of the most stress-inducing life events need to be a central part of treatment for long COVID, with more research needed to validate the best approaches, Dr. Frontera said.
She also noted that social services or case management resources may be able to help address at least some of the stressors that individuals are under – and it is important to refer them to these resources. Referral to mental health services is also important.
“I think it’s really important to take a holistic approach and try to deal with whatever the problem may be,” said Dr. Frontera.
“I’m a neurologist, but as part of my evaluation, I really need to address if there are life stressors or mental health issues that may be impacting this person’s function,” she added.
The study had no commercial funding. The investigators reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
Major life stressors in the year after hospital discharge for COVID-19 are “strongly predictive of a lot of the important outcomes that people may face after COVID,” lead investigator Jennifer A. Frontera, MD, a professor in the department of neurology at New York University Langone Health, said in an interview.
These outcomes include depression, brain fog, fatigue, trouble sleeping, and other long COVID symptoms.
The findings were published online in the Journal of the Neurological Sciences.
Major stressful events common
Dr. Frontera and the NYU Neurology COVID-19 study team evaluated 451 adults who survived a COVID hospital stay. Of these, 383 completed a 6-month follow-up, 242 completed a 12-month follow-up, and 174 completed follow-up at both time points.
Within 1 year of discharge, 77 (17%) patients died and 51% suffered a major stressful life event.
In multivariable analyses, major life stressors – including financial insecurity, food insecurity, death of a close contact, and new disability – were strong independent predictors of disability, trouble with activities of daily living, depression, fatigue, sleep problems, and prolonged post-acute COVID symptoms. The adjusted odds ratios for these outcomes ranged from 2.5 to 20.8.
The research also confirmed the contribution of traditional risk factors for long COVID symptoms, as shown in past studies. These include older age, poor pre-COVID functional status, and more severe initial COVID-19 infection.
Long-term sequelae of COVID are increasingly recognized as major public health issues.
It has been estimated that roughly 16 million U.S. adults aged 18-65 years ave long COVID, with the often debilitating symptoms keeping up to 4 million out of work.
Holistic approach
Dr. Frontera said it’s important to realize that “sleep, fatigue, anxiety, depression, even cognition are so interwoven with each other that anything that impacts any one of them could have repercussions on the other.”
She added that it “certainly makes sense that there is an interplay or even a bidirectional relationship between the stressors that people face and how well they can recover after COVID.”
Therapies that lessen the trauma of the most stress-inducing life events need to be a central part of treatment for long COVID, with more research needed to validate the best approaches, Dr. Frontera said.
She also noted that social services or case management resources may be able to help address at least some of the stressors that individuals are under – and it is important to refer them to these resources. Referral to mental health services is also important.
“I think it’s really important to take a holistic approach and try to deal with whatever the problem may be,” said Dr. Frontera.
“I’m a neurologist, but as part of my evaluation, I really need to address if there are life stressors or mental health issues that may be impacting this person’s function,” she added.
The study had no commercial funding. The investigators reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE NEUROLOGICAL SCIENCES
Experts explain the ‘perfect storm’ of rampant RSV and flu
Headlines over the past few weeks are ringing the alarm about earlier and more serious influenza (flu) and respiratory syncytial virus (RSV) outbreaks compared with previous years. Add COVID-19 to the mix and you have a dangerous mash of viruses that have many experts calling for caution and searching for explanations.
RSV and the flu “are certainly getting more attention, and they’re getting more attention for two reasons,” said William Schaffner, MD, professor of preventive medicine and infectious diseases at Vanderbilt University, Nashville, Tenn.
“The first is that they’re both extraordinarily early. The second is that they’re both out there spreading very, very rapidly,” he told this news organization.
RSV usually follows a seasonal pattern with cases peaking in January and February. Both viruses tend to hit different regions of the country at different times, and that’s not the case in 2022.
“This is particularly striking for RSV, which usually doesn’t affect the entire country simultaneously,” Dr. Schaffner said.
“Yes, RSV is causing many more hospitalizations and earlier than any previously recorded season in the U.S.,” according to figures from the Centers for Disease Control and Prevention on RSV hospitalizations, said Kevin Messacar, MD, PhD, associate professor at the University of Colorado at Denver, Aurora, and a pediatric infectious disease specialist at Children’s Hospital Colorado in Aurora.
Although there could be some increase in diagnoses because of increased awareness, the jump in RSV and flu cases “is a real phenomenon for multiple reasons,” said Peter Chin-Hong, MD, professor in the division of infectious diseases at the University of California, San Francisco.
With fewer COVID-related restrictions, people are moving around more. Also, during fall and winter, people tend to gather indoors. Colder temperatures and lower humidity contribute as well, Dr. Chin-Hong said, because “the droplets are just simply lighter.
“I think those are all factors,” he told this news organization.
Paul Auwaerter, MD, agreed that there are likely multiple causes for the unusual timing and severity of RSV and flu this year.
“Change in behaviors is a leading cause,” said the clinical director for the division of infectious diseases at the Johns Hopkins University, Baltimore. More people returning to the workplace and children going to school without masks are examples, he added.
Less exposure to these three viruses also means there was less immune boosting among existing populations, he said. This can lead to “larger susceptible populations, especially infants and younger children, due to the relative absence of circulating virus in past years.”
A leading theory
Are we paying a price now for people following the edicts from officials to mask up, stand apart, and take other personal and public health precautions during the COVID-19 pandemic?
It’s possible, but that may not be the whole story.
“When it comes to RSV, I think that theory of isolation, social distancing, mask wearing, and not attending schools is a very valid one,” Dr. Schaffner said. “That’s everybody’s favorite [reason].”
He said he is confident that the jump in RSV cases is being driven by previous COVID public health protections. However, he’s “a little more cautious about influenza, in part because influenza is so variable.
“Like people in influenza say, if you’ve seen one influenza season, you’ve seen one influenza season,” Dr. Schaffner said.
“There’s a lot of debate,” he added. “Nobody can say definitively whether the immune deficit or debt is a consequence of not being stimulated and restimulated by the influenza virus over the past two seasons.”
‘A perfect storm’
“Now you kind of have the perfect storm,” Dr. Chin-Hong said. “It’s not a good situation for COVID with the variants that are emerging. For influenza, not having seen a lot of influenza the last 2 years, we’re probably more susceptible to getting infected.”
RSV cases rose during summer 2021, but now the weather is colder, and people are interacting more closely. “And it’s very, very transmissible,” he said.
Dr. Chin-Hong also predicted that “even though we don’t have a lot of COVID now, COVID will probably pick up.”
The rise in RSV was unexpected by some experts. “This early influenza is also a bit of a surprise and may be influenced by the fact that lots of us are going back and seeing each other again close-to-close, face-to-face in many enclosed environments,” Dr. Schaffner said.
He estimated the 2022-2023 flu season started 4-6 weeks early “and it’s taken off like a rocket. It started in the Southeast, quickly went to the Southwest and up the East Coast. Now it’s moving dramatically through the Midwest and will continue. It’s quite sure to hit the West Coast if it isn’t there already.”
A phenomenon by any other name
Some are calling the situation an “immunity debt,” while others dub it an “immunity pause” or an “immunity deficit.” Many physicians and immunologists have taken to social media to push back on the term “immunity debt,” saying it’s a mischaracterization that is being used to vilify COVID precautions, such as masking, social distancing, and other protective measures taken during the pandemic.
“I prefer the term ‘immunity gap’ ... which is more established in the epidemiology literature, especially given the politicization of the term ‘immunity debt’ by folks recently,” Dr. Messacar said.
“To me, the immunity gap is a scientific observation, not a political argument,” he added.
In a July 2022 publication in The Lancet, Dr. Messacar and his colleagues stated that “decreased exposure to endemic viruses created an immunity gap – a group of susceptible individuals who avoided infection and therefore lack pathogen-specific immunity to protect against future infection. Decreases in childhood vaccinations with pandemic disruptions to health care delivery contribute to this immunity gap for vaccine-preventable diseases, such as influenza,measles, and polio.”
The researchers noted that because of isolation during the pandemic, older children and newborns are being exposed to RSV for the first time. Returning to birthday parties, playing with friends, and going to school without masks means “children are being exposed to RSV, and that’s likely the reason that RSV is moving early and very, very substantially through this now expanded pool of susceptible children,” Dr. Schaffner said.
How likely are coinfections?
With peaks in RSV, flu, and COVID-19 cases each predicted in the coming months, how likely is it that someone could get sick with more than one infection at the same time?
Early in the pandemic, coinfection with COVID and the flu was reported in people at some centers on the West Coast, Dr. Auwaerter said. Now, however, “the unpredictable nature of the Omicron subvariants and the potential for further change, along with the never-before-seen significant lessening of influenza over 2 years, leave little for predictability.
“I do think it is less likely, given the extent of immunity now to SARS-CoV-2 in the population,” Dr. Auwaerter said.
“I most worry about viral coinfections ... in people with suppressed immune systems if we have high community rates of the SARS-CoV-2 and influenza circulating this fall and winter,” he added.
Studies during the pandemic suggest that coinfection with the SARS-CoV-2 virus and another respiratory virus were either rare or nonexistent.
Dr. Schaffner said these findings align with his experience at Vanderbilt University, which is part of a CDC-sponsored network that tracks laboratory-confirmed RSV, flu, and COVID cases among people in the hospital. “Coinfections are, at least to date, very unusual.”
There needs to be an asterisk next to that, Dr. Schaffner added. “Looking back over the last 2 years, we’ve had very little influenza, and we’ve had curtailed RSV seasons. So there hasn’t been a whole lot of opportunity for dual infections to occur.
“So this year may be more revelatory as we go forward,” he said.
Future concerns
The future is uncertain, Dr. Messacar and colleagues wrote in The Lancet: “Crucially, the patterns of these returning viral outbreaks have been heterogeneous across locations, populations, and pathogens, making predictions and preparations challenging.”
Dr. Chin-Hong used a horse race analogy to illustrate the situation now and going forward. RSV is the front-running horse, and influenza is running behind but trying to catch up. “And then COVID is the dark horse. It’s trailing the race right now – but all these variants are giving the horse extra supplements.
“And the COVID horse is probably going to be very competitive with the front-runner,” he said.
“We’re just at the beginning of the race right now,” Dr. Chin-Hong said, “so that’s why we’re worried that these three [viruses] will be even more pronounced come later in the year.”
A version of this article first appeared on Medscape.com.
Headlines over the past few weeks are ringing the alarm about earlier and more serious influenza (flu) and respiratory syncytial virus (RSV) outbreaks compared with previous years. Add COVID-19 to the mix and you have a dangerous mash of viruses that have many experts calling for caution and searching for explanations.
RSV and the flu “are certainly getting more attention, and they’re getting more attention for two reasons,” said William Schaffner, MD, professor of preventive medicine and infectious diseases at Vanderbilt University, Nashville, Tenn.
“The first is that they’re both extraordinarily early. The second is that they’re both out there spreading very, very rapidly,” he told this news organization.
RSV usually follows a seasonal pattern with cases peaking in January and February. Both viruses tend to hit different regions of the country at different times, and that’s not the case in 2022.
“This is particularly striking for RSV, which usually doesn’t affect the entire country simultaneously,” Dr. Schaffner said.
“Yes, RSV is causing many more hospitalizations and earlier than any previously recorded season in the U.S.,” according to figures from the Centers for Disease Control and Prevention on RSV hospitalizations, said Kevin Messacar, MD, PhD, associate professor at the University of Colorado at Denver, Aurora, and a pediatric infectious disease specialist at Children’s Hospital Colorado in Aurora.
Although there could be some increase in diagnoses because of increased awareness, the jump in RSV and flu cases “is a real phenomenon for multiple reasons,” said Peter Chin-Hong, MD, professor in the division of infectious diseases at the University of California, San Francisco.
With fewer COVID-related restrictions, people are moving around more. Also, during fall and winter, people tend to gather indoors. Colder temperatures and lower humidity contribute as well, Dr. Chin-Hong said, because “the droplets are just simply lighter.
“I think those are all factors,” he told this news organization.
Paul Auwaerter, MD, agreed that there are likely multiple causes for the unusual timing and severity of RSV and flu this year.
“Change in behaviors is a leading cause,” said the clinical director for the division of infectious diseases at the Johns Hopkins University, Baltimore. More people returning to the workplace and children going to school without masks are examples, he added.
Less exposure to these three viruses also means there was less immune boosting among existing populations, he said. This can lead to “larger susceptible populations, especially infants and younger children, due to the relative absence of circulating virus in past years.”
A leading theory
Are we paying a price now for people following the edicts from officials to mask up, stand apart, and take other personal and public health precautions during the COVID-19 pandemic?
It’s possible, but that may not be the whole story.
“When it comes to RSV, I think that theory of isolation, social distancing, mask wearing, and not attending schools is a very valid one,” Dr. Schaffner said. “That’s everybody’s favorite [reason].”
He said he is confident that the jump in RSV cases is being driven by previous COVID public health protections. However, he’s “a little more cautious about influenza, in part because influenza is so variable.
“Like people in influenza say, if you’ve seen one influenza season, you’ve seen one influenza season,” Dr. Schaffner said.
“There’s a lot of debate,” he added. “Nobody can say definitively whether the immune deficit or debt is a consequence of not being stimulated and restimulated by the influenza virus over the past two seasons.”
‘A perfect storm’
“Now you kind of have the perfect storm,” Dr. Chin-Hong said. “It’s not a good situation for COVID with the variants that are emerging. For influenza, not having seen a lot of influenza the last 2 years, we’re probably more susceptible to getting infected.”
RSV cases rose during summer 2021, but now the weather is colder, and people are interacting more closely. “And it’s very, very transmissible,” he said.
Dr. Chin-Hong also predicted that “even though we don’t have a lot of COVID now, COVID will probably pick up.”
The rise in RSV was unexpected by some experts. “This early influenza is also a bit of a surprise and may be influenced by the fact that lots of us are going back and seeing each other again close-to-close, face-to-face in many enclosed environments,” Dr. Schaffner said.
He estimated the 2022-2023 flu season started 4-6 weeks early “and it’s taken off like a rocket. It started in the Southeast, quickly went to the Southwest and up the East Coast. Now it’s moving dramatically through the Midwest and will continue. It’s quite sure to hit the West Coast if it isn’t there already.”
A phenomenon by any other name
Some are calling the situation an “immunity debt,” while others dub it an “immunity pause” or an “immunity deficit.” Many physicians and immunologists have taken to social media to push back on the term “immunity debt,” saying it’s a mischaracterization that is being used to vilify COVID precautions, such as masking, social distancing, and other protective measures taken during the pandemic.
“I prefer the term ‘immunity gap’ ... which is more established in the epidemiology literature, especially given the politicization of the term ‘immunity debt’ by folks recently,” Dr. Messacar said.
“To me, the immunity gap is a scientific observation, not a political argument,” he added.
In a July 2022 publication in The Lancet, Dr. Messacar and his colleagues stated that “decreased exposure to endemic viruses created an immunity gap – a group of susceptible individuals who avoided infection and therefore lack pathogen-specific immunity to protect against future infection. Decreases in childhood vaccinations with pandemic disruptions to health care delivery contribute to this immunity gap for vaccine-preventable diseases, such as influenza,measles, and polio.”
The researchers noted that because of isolation during the pandemic, older children and newborns are being exposed to RSV for the first time. Returning to birthday parties, playing with friends, and going to school without masks means “children are being exposed to RSV, and that’s likely the reason that RSV is moving early and very, very substantially through this now expanded pool of susceptible children,” Dr. Schaffner said.
How likely are coinfections?
With peaks in RSV, flu, and COVID-19 cases each predicted in the coming months, how likely is it that someone could get sick with more than one infection at the same time?
Early in the pandemic, coinfection with COVID and the flu was reported in people at some centers on the West Coast, Dr. Auwaerter said. Now, however, “the unpredictable nature of the Omicron subvariants and the potential for further change, along with the never-before-seen significant lessening of influenza over 2 years, leave little for predictability.
“I do think it is less likely, given the extent of immunity now to SARS-CoV-2 in the population,” Dr. Auwaerter said.
“I most worry about viral coinfections ... in people with suppressed immune systems if we have high community rates of the SARS-CoV-2 and influenza circulating this fall and winter,” he added.
Studies during the pandemic suggest that coinfection with the SARS-CoV-2 virus and another respiratory virus were either rare or nonexistent.
Dr. Schaffner said these findings align with his experience at Vanderbilt University, which is part of a CDC-sponsored network that tracks laboratory-confirmed RSV, flu, and COVID cases among people in the hospital. “Coinfections are, at least to date, very unusual.”
There needs to be an asterisk next to that, Dr. Schaffner added. “Looking back over the last 2 years, we’ve had very little influenza, and we’ve had curtailed RSV seasons. So there hasn’t been a whole lot of opportunity for dual infections to occur.
“So this year may be more revelatory as we go forward,” he said.
Future concerns
The future is uncertain, Dr. Messacar and colleagues wrote in The Lancet: “Crucially, the patterns of these returning viral outbreaks have been heterogeneous across locations, populations, and pathogens, making predictions and preparations challenging.”
Dr. Chin-Hong used a horse race analogy to illustrate the situation now and going forward. RSV is the front-running horse, and influenza is running behind but trying to catch up. “And then COVID is the dark horse. It’s trailing the race right now – but all these variants are giving the horse extra supplements.
“And the COVID horse is probably going to be very competitive with the front-runner,” he said.
“We’re just at the beginning of the race right now,” Dr. Chin-Hong said, “so that’s why we’re worried that these three [viruses] will be even more pronounced come later in the year.”
A version of this article first appeared on Medscape.com.
Headlines over the past few weeks are ringing the alarm about earlier and more serious influenza (flu) and respiratory syncytial virus (RSV) outbreaks compared with previous years. Add COVID-19 to the mix and you have a dangerous mash of viruses that have many experts calling for caution and searching for explanations.
RSV and the flu “are certainly getting more attention, and they’re getting more attention for two reasons,” said William Schaffner, MD, professor of preventive medicine and infectious diseases at Vanderbilt University, Nashville, Tenn.
“The first is that they’re both extraordinarily early. The second is that they’re both out there spreading very, very rapidly,” he told this news organization.
RSV usually follows a seasonal pattern with cases peaking in January and February. Both viruses tend to hit different regions of the country at different times, and that’s not the case in 2022.
“This is particularly striking for RSV, which usually doesn’t affect the entire country simultaneously,” Dr. Schaffner said.
“Yes, RSV is causing many more hospitalizations and earlier than any previously recorded season in the U.S.,” according to figures from the Centers for Disease Control and Prevention on RSV hospitalizations, said Kevin Messacar, MD, PhD, associate professor at the University of Colorado at Denver, Aurora, and a pediatric infectious disease specialist at Children’s Hospital Colorado in Aurora.
Although there could be some increase in diagnoses because of increased awareness, the jump in RSV and flu cases “is a real phenomenon for multiple reasons,” said Peter Chin-Hong, MD, professor in the division of infectious diseases at the University of California, San Francisco.
With fewer COVID-related restrictions, people are moving around more. Also, during fall and winter, people tend to gather indoors. Colder temperatures and lower humidity contribute as well, Dr. Chin-Hong said, because “the droplets are just simply lighter.
“I think those are all factors,” he told this news organization.
Paul Auwaerter, MD, agreed that there are likely multiple causes for the unusual timing and severity of RSV and flu this year.
“Change in behaviors is a leading cause,” said the clinical director for the division of infectious diseases at the Johns Hopkins University, Baltimore. More people returning to the workplace and children going to school without masks are examples, he added.
Less exposure to these three viruses also means there was less immune boosting among existing populations, he said. This can lead to “larger susceptible populations, especially infants and younger children, due to the relative absence of circulating virus in past years.”
A leading theory
Are we paying a price now for people following the edicts from officials to mask up, stand apart, and take other personal and public health precautions during the COVID-19 pandemic?
It’s possible, but that may not be the whole story.
“When it comes to RSV, I think that theory of isolation, social distancing, mask wearing, and not attending schools is a very valid one,” Dr. Schaffner said. “That’s everybody’s favorite [reason].”
He said he is confident that the jump in RSV cases is being driven by previous COVID public health protections. However, he’s “a little more cautious about influenza, in part because influenza is so variable.
“Like people in influenza say, if you’ve seen one influenza season, you’ve seen one influenza season,” Dr. Schaffner said.
“There’s a lot of debate,” he added. “Nobody can say definitively whether the immune deficit or debt is a consequence of not being stimulated and restimulated by the influenza virus over the past two seasons.”
‘A perfect storm’
“Now you kind of have the perfect storm,” Dr. Chin-Hong said. “It’s not a good situation for COVID with the variants that are emerging. For influenza, not having seen a lot of influenza the last 2 years, we’re probably more susceptible to getting infected.”
RSV cases rose during summer 2021, but now the weather is colder, and people are interacting more closely. “And it’s very, very transmissible,” he said.
Dr. Chin-Hong also predicted that “even though we don’t have a lot of COVID now, COVID will probably pick up.”
The rise in RSV was unexpected by some experts. “This early influenza is also a bit of a surprise and may be influenced by the fact that lots of us are going back and seeing each other again close-to-close, face-to-face in many enclosed environments,” Dr. Schaffner said.
He estimated the 2022-2023 flu season started 4-6 weeks early “and it’s taken off like a rocket. It started in the Southeast, quickly went to the Southwest and up the East Coast. Now it’s moving dramatically through the Midwest and will continue. It’s quite sure to hit the West Coast if it isn’t there already.”
A phenomenon by any other name
Some are calling the situation an “immunity debt,” while others dub it an “immunity pause” or an “immunity deficit.” Many physicians and immunologists have taken to social media to push back on the term “immunity debt,” saying it’s a mischaracterization that is being used to vilify COVID precautions, such as masking, social distancing, and other protective measures taken during the pandemic.
“I prefer the term ‘immunity gap’ ... which is more established in the epidemiology literature, especially given the politicization of the term ‘immunity debt’ by folks recently,” Dr. Messacar said.
“To me, the immunity gap is a scientific observation, not a political argument,” he added.
In a July 2022 publication in The Lancet, Dr. Messacar and his colleagues stated that “decreased exposure to endemic viruses created an immunity gap – a group of susceptible individuals who avoided infection and therefore lack pathogen-specific immunity to protect against future infection. Decreases in childhood vaccinations with pandemic disruptions to health care delivery contribute to this immunity gap for vaccine-preventable diseases, such as influenza,measles, and polio.”
The researchers noted that because of isolation during the pandemic, older children and newborns are being exposed to RSV for the first time. Returning to birthday parties, playing with friends, and going to school without masks means “children are being exposed to RSV, and that’s likely the reason that RSV is moving early and very, very substantially through this now expanded pool of susceptible children,” Dr. Schaffner said.
How likely are coinfections?
With peaks in RSV, flu, and COVID-19 cases each predicted in the coming months, how likely is it that someone could get sick with more than one infection at the same time?
Early in the pandemic, coinfection with COVID and the flu was reported in people at some centers on the West Coast, Dr. Auwaerter said. Now, however, “the unpredictable nature of the Omicron subvariants and the potential for further change, along with the never-before-seen significant lessening of influenza over 2 years, leave little for predictability.
“I do think it is less likely, given the extent of immunity now to SARS-CoV-2 in the population,” Dr. Auwaerter said.
“I most worry about viral coinfections ... in people with suppressed immune systems if we have high community rates of the SARS-CoV-2 and influenza circulating this fall and winter,” he added.
Studies during the pandemic suggest that coinfection with the SARS-CoV-2 virus and another respiratory virus were either rare or nonexistent.
Dr. Schaffner said these findings align with his experience at Vanderbilt University, which is part of a CDC-sponsored network that tracks laboratory-confirmed RSV, flu, and COVID cases among people in the hospital. “Coinfections are, at least to date, very unusual.”
There needs to be an asterisk next to that, Dr. Schaffner added. “Looking back over the last 2 years, we’ve had very little influenza, and we’ve had curtailed RSV seasons. So there hasn’t been a whole lot of opportunity for dual infections to occur.
“So this year may be more revelatory as we go forward,” he said.
Future concerns
The future is uncertain, Dr. Messacar and colleagues wrote in The Lancet: “Crucially, the patterns of these returning viral outbreaks have been heterogeneous across locations, populations, and pathogens, making predictions and preparations challenging.”
Dr. Chin-Hong used a horse race analogy to illustrate the situation now and going forward. RSV is the front-running horse, and influenza is running behind but trying to catch up. “And then COVID is the dark horse. It’s trailing the race right now – but all these variants are giving the horse extra supplements.
“And the COVID horse is probably going to be very competitive with the front-runner,” he said.
“We’re just at the beginning of the race right now,” Dr. Chin-Hong said, “so that’s why we’re worried that these three [viruses] will be even more pronounced come later in the year.”
A version of this article first appeared on Medscape.com.
Be aware, mindfulness training can lower systolic BP: MB-BP
CHICAGO – It’s been said that one can observe a lot just by watching. Turning such observation inward, new evidence suggests, might lead to blood pressure (BP) reductions that approach what’s possible from an antihypertensive agent.
Systolic BP fell over 6 months by almost 6 mm Hg, on average, in people with elevated BP who participated in an 8-week mindful awareness program as part of a randomized trial that included a usual-care control group.
The program taught established mindfulness-training techniques aimed at modifying behaviors regarding diet, exercise, and other controllable influences on the success of antihypertensive therapy.
Participants in the program, called Mindfulness-Based Blood Pressure Reduction (MB-BP), also the name of the single-center study, “showed potentially clinically relevant reductions in systolic blood pressure,” said principal investigator Eric B. Loucks, PhD, Brown University, Providence, R.I.
The phase 2 trial has some limitations, he observed, including on generalizability. For example, it entered about 200 mostly White, college-educated adults from one metropolitan area.
But if these findings are replicated in further studies, “preferably by other research groups, in a larger and broader population, and with longer follow-up,” Dr. Loucks said, the MB-BP intervention could become “an appealing approach to help control blood pressure.”
Dr. Loucks made the comments at a press conference prior to his formal presentation of MB-BP Nov. 6 at American Heart Association (AHA) Scientific Sessions 2022, held in Chicago and virtually.
Mindfulness-based interventions for elevated BP have not been widely studied, “so this is exactly what we need: a well-done trial with a control group to show that it actually works,” Amit Khera, MD, not connected with MB-BP, told this news organization.
The trial is “really important for proof of concept, but it had only 200 people. You need a larger one, and you need longer-term data,” agreed Dr. Khera, who directs the preventive cardiology program at the University of Texas Southwestern Medical Center, in Dallas. “Six months is good, but we want to see if it’s durable.”
Rhian M. Touyz, MBBCh, also not part of MB-BP, agreed that the nearly 6 mm Hg mean systolic BP reduction among program participants is clinically relevant. “I think in the context of global risk and reduction of target organ damage and cardiovascular events, it is significant in terms of events at a population level,” Dr. Touyz, McGill University Health Centre, Montreal, told this news organization.
Many patients on antihypertensive therapy that’s falling short resist the addition of another such agent, she observed, and instead might show further BP reduction from mindfulness training. The intervention probably also “would benefit health in general.” Mindfulness-based approaches could therefore be useful additions to treatment protocols for elevated BP, Dr. Touyz said.
How the training works
The MB-BP program used validated mindfulness-based stress-management techniques, adapted to address elevated BP, that included “personalized feedback and education about hypertension risk factors, mindful awareness training of participants’ relationships with hypertension risk factors, and support for behavior change,” Dr. Loucks and colleagues reported.
Participants were trained in mindfulness skills that included “self-awareness and emotion regulation,” Dr. Loucks said, which they then could apply to their “relationships with the things that we know influence blood pressure, like physical activity, diet, antihypertensive medication adherence, or alcohol consumption.”
One goal is to promote greater “attention control,” he said, “so that there’s some self-awareness that arises in terms of how we feel the next day, after a lot of alcohol consumption, for example, or lack of physical activity.” The process can provide insights that inspire patients to modify behaviors and risk factors that elevate BP, Dr. Loucks explained.
Effects on medication use
Systolic BP responses led some program participants to be managed on fewer or reduced dosages of antihypertensive meds, he told this news organization. Physicians seen outside of the trial could adjust their prescriptions, intensifying or pulling back on meds depending on their assessments of the patient. Any prescription changes would be documented by the researchers at the patient’s next class or trial-clinic visit.
The group that did the training, Dr. Loucks said, was 33% less likely to increase and 30% more likely to decrease their use of BP-lowering medications compared with the control group.
Elevated BP is so common and undertreated that “there is a need for every possible level of intervention, starting from the population level to the individual and everything else in between,” nephrologist Janani Rangaswami, MD, George Washington University, Washington, said at the press conference.
Therefore, “this mindfulness-based approach, in addition to standard of care with pharmacotherapy, is a really welcome addition to the hypertension literature,” said Dr. Rangaswami, who directs her center’s cardiorenal program. The systolic BP reduction seen in the intervention group, she agreed, was “clinically important and meaningful.”
Blinded assessments
The trial entered 201 patients with systolic and diastolic BP greater than 120 mm Hg and 80 mm Hg, respectively; 58.7% were women, 81% were White, and 73% were college-educated, Dr. Loucks reported.
The 100 assigned to the “enhanced usual care” control group received educational materials on controlling high BP. They and the 101 who followed the mindfulness-based program were given and trained on a home BP-monitoring device. They were then followed for the primary endpoint of change in systolic BP at 6 months.
Data management and outcomes assessments were conducted by trialists not involved in the training intervention who were blinded to randomization assignment.
In a prespecified unadjusted analysis by intention-to-treat, systolic BP in the intervention group dropped by a mean of 5.9 mm Hg (P < .001) compared with baseline and 4.5 mm Hg (P = .045), compared with the control group.
A post hoc analysis adjusted for sex and baseline BP showed an average 4.3 mm Hg reduction (P = .056) in those following the MB-BP program, compared with controls.
There were no observed significant effects on diastolic BP.
The study offered clues to how engagement in the MB-BP program might promote reductions in systolic BP, Dr. Loucks observed. For example, it may have led to increased activity levels, reduced sodium intake, and other dietary improvements.
Indeed, program participants averaged about 351 minutes less sedentary time (P = .02) and showed a 0.32-point improvement in Dietary Approaches to Stop Hypertension scores (P = .08), compared with the control group, Dr. Loucks reported. Other modifiable risk factors for elevated BP that could have responded to the mindfulness-based training, he proposed, include obesity, alcohol intake, and reaction to stress.
Dr. Loucks reports that he developed the MB-BP training and was a program instructor but did not receive related financial compensation; he had no other disclosures. Dr. Khera, Dr. Touyz, and Dr. Rangaswami had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
CHICAGO – It’s been said that one can observe a lot just by watching. Turning such observation inward, new evidence suggests, might lead to blood pressure (BP) reductions that approach what’s possible from an antihypertensive agent.
Systolic BP fell over 6 months by almost 6 mm Hg, on average, in people with elevated BP who participated in an 8-week mindful awareness program as part of a randomized trial that included a usual-care control group.
The program taught established mindfulness-training techniques aimed at modifying behaviors regarding diet, exercise, and other controllable influences on the success of antihypertensive therapy.
Participants in the program, called Mindfulness-Based Blood Pressure Reduction (MB-BP), also the name of the single-center study, “showed potentially clinically relevant reductions in systolic blood pressure,” said principal investigator Eric B. Loucks, PhD, Brown University, Providence, R.I.
The phase 2 trial has some limitations, he observed, including on generalizability. For example, it entered about 200 mostly White, college-educated adults from one metropolitan area.
But if these findings are replicated in further studies, “preferably by other research groups, in a larger and broader population, and with longer follow-up,” Dr. Loucks said, the MB-BP intervention could become “an appealing approach to help control blood pressure.”
Dr. Loucks made the comments at a press conference prior to his formal presentation of MB-BP Nov. 6 at American Heart Association (AHA) Scientific Sessions 2022, held in Chicago and virtually.
Mindfulness-based interventions for elevated BP have not been widely studied, “so this is exactly what we need: a well-done trial with a control group to show that it actually works,” Amit Khera, MD, not connected with MB-BP, told this news organization.
The trial is “really important for proof of concept, but it had only 200 people. You need a larger one, and you need longer-term data,” agreed Dr. Khera, who directs the preventive cardiology program at the University of Texas Southwestern Medical Center, in Dallas. “Six months is good, but we want to see if it’s durable.”
Rhian M. Touyz, MBBCh, also not part of MB-BP, agreed that the nearly 6 mm Hg mean systolic BP reduction among program participants is clinically relevant. “I think in the context of global risk and reduction of target organ damage and cardiovascular events, it is significant in terms of events at a population level,” Dr. Touyz, McGill University Health Centre, Montreal, told this news organization.
Many patients on antihypertensive therapy that’s falling short resist the addition of another such agent, she observed, and instead might show further BP reduction from mindfulness training. The intervention probably also “would benefit health in general.” Mindfulness-based approaches could therefore be useful additions to treatment protocols for elevated BP, Dr. Touyz said.
How the training works
The MB-BP program used validated mindfulness-based stress-management techniques, adapted to address elevated BP, that included “personalized feedback and education about hypertension risk factors, mindful awareness training of participants’ relationships with hypertension risk factors, and support for behavior change,” Dr. Loucks and colleagues reported.
Participants were trained in mindfulness skills that included “self-awareness and emotion regulation,” Dr. Loucks said, which they then could apply to their “relationships with the things that we know influence blood pressure, like physical activity, diet, antihypertensive medication adherence, or alcohol consumption.”
One goal is to promote greater “attention control,” he said, “so that there’s some self-awareness that arises in terms of how we feel the next day, after a lot of alcohol consumption, for example, or lack of physical activity.” The process can provide insights that inspire patients to modify behaviors and risk factors that elevate BP, Dr. Loucks explained.
Effects on medication use
Systolic BP responses led some program participants to be managed on fewer or reduced dosages of antihypertensive meds, he told this news organization. Physicians seen outside of the trial could adjust their prescriptions, intensifying or pulling back on meds depending on their assessments of the patient. Any prescription changes would be documented by the researchers at the patient’s next class or trial-clinic visit.
The group that did the training, Dr. Loucks said, was 33% less likely to increase and 30% more likely to decrease their use of BP-lowering medications compared with the control group.
Elevated BP is so common and undertreated that “there is a need for every possible level of intervention, starting from the population level to the individual and everything else in between,” nephrologist Janani Rangaswami, MD, George Washington University, Washington, said at the press conference.
Therefore, “this mindfulness-based approach, in addition to standard of care with pharmacotherapy, is a really welcome addition to the hypertension literature,” said Dr. Rangaswami, who directs her center’s cardiorenal program. The systolic BP reduction seen in the intervention group, she agreed, was “clinically important and meaningful.”
Blinded assessments
The trial entered 201 patients with systolic and diastolic BP greater than 120 mm Hg and 80 mm Hg, respectively; 58.7% were women, 81% were White, and 73% were college-educated, Dr. Loucks reported.
The 100 assigned to the “enhanced usual care” control group received educational materials on controlling high BP. They and the 101 who followed the mindfulness-based program were given and trained on a home BP-monitoring device. They were then followed for the primary endpoint of change in systolic BP at 6 months.
Data management and outcomes assessments were conducted by trialists not involved in the training intervention who were blinded to randomization assignment.
In a prespecified unadjusted analysis by intention-to-treat, systolic BP in the intervention group dropped by a mean of 5.9 mm Hg (P < .001) compared with baseline and 4.5 mm Hg (P = .045), compared with the control group.
A post hoc analysis adjusted for sex and baseline BP showed an average 4.3 mm Hg reduction (P = .056) in those following the MB-BP program, compared with controls.
There were no observed significant effects on diastolic BP.
The study offered clues to how engagement in the MB-BP program might promote reductions in systolic BP, Dr. Loucks observed. For example, it may have led to increased activity levels, reduced sodium intake, and other dietary improvements.
Indeed, program participants averaged about 351 minutes less sedentary time (P = .02) and showed a 0.32-point improvement in Dietary Approaches to Stop Hypertension scores (P = .08), compared with the control group, Dr. Loucks reported. Other modifiable risk factors for elevated BP that could have responded to the mindfulness-based training, he proposed, include obesity, alcohol intake, and reaction to stress.
Dr. Loucks reports that he developed the MB-BP training and was a program instructor but did not receive related financial compensation; he had no other disclosures. Dr. Khera, Dr. Touyz, and Dr. Rangaswami had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
CHICAGO – It’s been said that one can observe a lot just by watching. Turning such observation inward, new evidence suggests, might lead to blood pressure (BP) reductions that approach what’s possible from an antihypertensive agent.
Systolic BP fell over 6 months by almost 6 mm Hg, on average, in people with elevated BP who participated in an 8-week mindful awareness program as part of a randomized trial that included a usual-care control group.
The program taught established mindfulness-training techniques aimed at modifying behaviors regarding diet, exercise, and other controllable influences on the success of antihypertensive therapy.
Participants in the program, called Mindfulness-Based Blood Pressure Reduction (MB-BP), also the name of the single-center study, “showed potentially clinically relevant reductions in systolic blood pressure,” said principal investigator Eric B. Loucks, PhD, Brown University, Providence, R.I.
The phase 2 trial has some limitations, he observed, including on generalizability. For example, it entered about 200 mostly White, college-educated adults from one metropolitan area.
But if these findings are replicated in further studies, “preferably by other research groups, in a larger and broader population, and with longer follow-up,” Dr. Loucks said, the MB-BP intervention could become “an appealing approach to help control blood pressure.”
Dr. Loucks made the comments at a press conference prior to his formal presentation of MB-BP Nov. 6 at American Heart Association (AHA) Scientific Sessions 2022, held in Chicago and virtually.
Mindfulness-based interventions for elevated BP have not been widely studied, “so this is exactly what we need: a well-done trial with a control group to show that it actually works,” Amit Khera, MD, not connected with MB-BP, told this news organization.
The trial is “really important for proof of concept, but it had only 200 people. You need a larger one, and you need longer-term data,” agreed Dr. Khera, who directs the preventive cardiology program at the University of Texas Southwestern Medical Center, in Dallas. “Six months is good, but we want to see if it’s durable.”
Rhian M. Touyz, MBBCh, also not part of MB-BP, agreed that the nearly 6 mm Hg mean systolic BP reduction among program participants is clinically relevant. “I think in the context of global risk and reduction of target organ damage and cardiovascular events, it is significant in terms of events at a population level,” Dr. Touyz, McGill University Health Centre, Montreal, told this news organization.
Many patients on antihypertensive therapy that’s falling short resist the addition of another such agent, she observed, and instead might show further BP reduction from mindfulness training. The intervention probably also “would benefit health in general.” Mindfulness-based approaches could therefore be useful additions to treatment protocols for elevated BP, Dr. Touyz said.
How the training works
The MB-BP program used validated mindfulness-based stress-management techniques, adapted to address elevated BP, that included “personalized feedback and education about hypertension risk factors, mindful awareness training of participants’ relationships with hypertension risk factors, and support for behavior change,” Dr. Loucks and colleagues reported.
Participants were trained in mindfulness skills that included “self-awareness and emotion regulation,” Dr. Loucks said, which they then could apply to their “relationships with the things that we know influence blood pressure, like physical activity, diet, antihypertensive medication adherence, or alcohol consumption.”
One goal is to promote greater “attention control,” he said, “so that there’s some self-awareness that arises in terms of how we feel the next day, after a lot of alcohol consumption, for example, or lack of physical activity.” The process can provide insights that inspire patients to modify behaviors and risk factors that elevate BP, Dr. Loucks explained.
Effects on medication use
Systolic BP responses led some program participants to be managed on fewer or reduced dosages of antihypertensive meds, he told this news organization. Physicians seen outside of the trial could adjust their prescriptions, intensifying or pulling back on meds depending on their assessments of the patient. Any prescription changes would be documented by the researchers at the patient’s next class or trial-clinic visit.
The group that did the training, Dr. Loucks said, was 33% less likely to increase and 30% more likely to decrease their use of BP-lowering medications compared with the control group.
Elevated BP is so common and undertreated that “there is a need for every possible level of intervention, starting from the population level to the individual and everything else in between,” nephrologist Janani Rangaswami, MD, George Washington University, Washington, said at the press conference.
Therefore, “this mindfulness-based approach, in addition to standard of care with pharmacotherapy, is a really welcome addition to the hypertension literature,” said Dr. Rangaswami, who directs her center’s cardiorenal program. The systolic BP reduction seen in the intervention group, she agreed, was “clinically important and meaningful.”
Blinded assessments
The trial entered 201 patients with systolic and diastolic BP greater than 120 mm Hg and 80 mm Hg, respectively; 58.7% were women, 81% were White, and 73% were college-educated, Dr. Loucks reported.
The 100 assigned to the “enhanced usual care” control group received educational materials on controlling high BP. They and the 101 who followed the mindfulness-based program were given and trained on a home BP-monitoring device. They were then followed for the primary endpoint of change in systolic BP at 6 months.
Data management and outcomes assessments were conducted by trialists not involved in the training intervention who were blinded to randomization assignment.
In a prespecified unadjusted analysis by intention-to-treat, systolic BP in the intervention group dropped by a mean of 5.9 mm Hg (P < .001) compared with baseline and 4.5 mm Hg (P = .045), compared with the control group.
A post hoc analysis adjusted for sex and baseline BP showed an average 4.3 mm Hg reduction (P = .056) in those following the MB-BP program, compared with controls.
There were no observed significant effects on diastolic BP.
The study offered clues to how engagement in the MB-BP program might promote reductions in systolic BP, Dr. Loucks observed. For example, it may have led to increased activity levels, reduced sodium intake, and other dietary improvements.
Indeed, program participants averaged about 351 minutes less sedentary time (P = .02) and showed a 0.32-point improvement in Dietary Approaches to Stop Hypertension scores (P = .08), compared with the control group, Dr. Loucks reported. Other modifiable risk factors for elevated BP that could have responded to the mindfulness-based training, he proposed, include obesity, alcohol intake, and reaction to stress.
Dr. Loucks reports that he developed the MB-BP training and was a program instructor but did not receive related financial compensation; he had no other disclosures. Dr. Khera, Dr. Touyz, and Dr. Rangaswami had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT AHA 2022
Hypertension linked to risk of severe COVID
U.K. researchers have established that hypertension is associated with a 22% greater risk of severe COVID-19, with the odds of severe COVID-19 unaffected by medication type.
Hypertension “appears to be one of the commonest comorbidities in COVID-19 patients”, explained the authors of a new study, published in PLOS ONE. The authors highlighted that previous research had shown that hypertension was more prevalent in severe and fatal cases compared with all cases of COVID-19.
They pointed out, however, that whether hypertensive individuals have a higher risk of severe COVID-19, compared with nonhypertensives, and whether the absolute level of systolic blood pressure or the type of antihypertensive medication is related to this risk, remained “unclear.”
To try to answer these questions, the research team, led by University of Cambridge researchers, analyzed data from 16,134 individuals who tested positive for COVID-19 (mean age 65.3 years, 47% male, 90% white), 40% were diagnosed with essential hypertension at the analysis baseline – 22% of whom had developed severe COVID-19.
Systolic blood pressure (SBP) was categorized by 10–mm Hg ranges, starting from < 120 mm Hg up to 180+ mm Hg, with the reference category defined as 120-129 mm Hg, based on data from the SPRINT study, which demonstrated that intensive SBP lowering to below 120 mm Hg, as compared with the traditional threshold of 140 mm Hg, was beneficial. Diastolic blood pressure was categorized by 10–mm Hg ranges, starting from < 60 mm Hg up to 100+ mm Hg with 80-90 mm Hg being the reference category.
In their analyses the researchers adjusted for age, sex, body mass index, ethnicity, smoking status, diabetes status, socioeconomic status, and inflammation (C-reactive protein [CRP]), as these were proposed as potential confounders. To assess the direct effect of hypertension on COVID-19, they also adjusted for intermediate variables, including cardiovascular comorbidities and stroke, on the causal pathway between hypertension and severe COVID-19.
Majority of effect of hypertension on severe COVID-19 was direct
The unadjusted odds ratio of the association between hypertension and severe COVID-19 was 2.33 (95% confidence interval, 2.16-2.51), the authors emphasized. They found that, after adjusting for all confounding variables, hypertension was associated with 22% higher odds of severe COVID-19 (OR, 1.22; 95% CI, 1.12-1.33), compared with normotension.
Individuals with severe COVID-19 were marginally older, more likely to be male, and more deprived, the authors said. “They were also more likely to be hypertensive, compared with individuals without severe COVID-19, and a greater proportion of individuals with severe COVID-19 had cardiovascular comorbidities.”
The majority of the effect of hypertension on development of severe COVID-19 was “direct,” they said. However, a modest proportion of the effect was mediated via cardiovascular comorbidities such as peripheral vascular disease, MI, coronary heart disease, arrhythmias, and stroke. Of note, those with a history of stroke had a 47% higher risk of severe COVID-19 and those with a history of other cardiovascular comorbidities had a 30% higher risk of severe COVID-19, the authors commented.
J-shaped relationship
Of the total of 6,517 (40%) individuals who had a diagnosis of essential hypertension at baseline, 67% were treated (41% with monotherapy, 59% with combination therapy), and 33% were untreated.
There were similar numbers of severe COVID-19 in each medication group: ACE inhibitors, 34%; angiotensin receptor blockers (ARBs), 36%; and “other” medications 34%.
In hypertensive individuals receiving antihypertensive medications, there was a “J-shaped relationship” between the level of blood pressure and risk of severe COVID-19 when using a systolic blood pressure level of 120-129 mm Hg as a reference – 150-159 mm Hg versus 120-129 mm Hg (OR 1.91; 95% CI, 1.44-2.53), > 180+ mm Hg versus 120-129 mm Hg (OR 1.93; 95% CI, 1.06-3.51).
The authors commented that there was no evidence of a higher risk of severe COVID-19 until systolic blood pressure “exceeded 150 mm Hg.”
They said it was an interesting finding that “very well-controlled” systolic blood pressure < 120 mm Hg was associated with a 40% (OR, 1.40; 95% CI, 1.11-1.78) greater odds of severe COVID-19. “This may be due to reverse causality, where low systolic blood pressure levels may indicate poorer health, such that the occurrence of severe COVID-19 may be related to underlying disease rather than the level of SBP per se,” they suggested.
The J-shaped association observed remained after multiple adjustments, including presence of known cardiovascular comorbidities, which suggested a possible “real effect” of low SBP on severe COVID-19, “at least in treated hypertensive individuals.”
Their analyses also identified that, compared with a “normal” diastolic blood pressure (80-90 mm Hg), having a diastolic blood pressure higher than 90 mm Hg was associated with higher odds of severe COVID-19.
The association between hypertension and COVID-19 was “amplified” if the individuals were treated and their BP remained uncontrolled, the authors pointed out.
There did not appear to be any difference in the risk of severe COVID-19 between individuals taking ACE inhibitors and those taking ARBs or other antihypertensive medications, the authors said.
Better understanding of underlying mechanisms needed
Individuals with hypertension who tested positive for COVID-19 had “over twice” the risk of developing severe COVID-19, compared with nonhypertensive individuals, the authors said.
They highlighted that their findings also suggest that there are “further effects” influencing the severity of COVID-19 beyond a “dichotomous” diagnosis of hypertension.
“Individuals with a higher-than-target systolic blood pressure may be less healthy, less active, suffering more severe hypertension, or have developed drug-resistant hypertension, all suggesting that the effects of hypertension have already had detrimental physiological effects on the cardiovascular system, which in turn may offer some explanation for the higher risk of severe COVID-19 with uncontrolled SBP,” they explained.
“Hypertension is an important risk factor for COVID-19,” reiterated the authors, who emphasized that a better understanding of the underlying mechanisms driving this increased risk is warranted in case of “more severe strains or other viruses” in the future.
The authors have declared no competing interests.
A version of this article first appeared on Medscape UK.
U.K. researchers have established that hypertension is associated with a 22% greater risk of severe COVID-19, with the odds of severe COVID-19 unaffected by medication type.
Hypertension “appears to be one of the commonest comorbidities in COVID-19 patients”, explained the authors of a new study, published in PLOS ONE. The authors highlighted that previous research had shown that hypertension was more prevalent in severe and fatal cases compared with all cases of COVID-19.
They pointed out, however, that whether hypertensive individuals have a higher risk of severe COVID-19, compared with nonhypertensives, and whether the absolute level of systolic blood pressure or the type of antihypertensive medication is related to this risk, remained “unclear.”
To try to answer these questions, the research team, led by University of Cambridge researchers, analyzed data from 16,134 individuals who tested positive for COVID-19 (mean age 65.3 years, 47% male, 90% white), 40% were diagnosed with essential hypertension at the analysis baseline – 22% of whom had developed severe COVID-19.
Systolic blood pressure (SBP) was categorized by 10–mm Hg ranges, starting from < 120 mm Hg up to 180+ mm Hg, with the reference category defined as 120-129 mm Hg, based on data from the SPRINT study, which demonstrated that intensive SBP lowering to below 120 mm Hg, as compared with the traditional threshold of 140 mm Hg, was beneficial. Diastolic blood pressure was categorized by 10–mm Hg ranges, starting from < 60 mm Hg up to 100+ mm Hg with 80-90 mm Hg being the reference category.
In their analyses the researchers adjusted for age, sex, body mass index, ethnicity, smoking status, diabetes status, socioeconomic status, and inflammation (C-reactive protein [CRP]), as these were proposed as potential confounders. To assess the direct effect of hypertension on COVID-19, they also adjusted for intermediate variables, including cardiovascular comorbidities and stroke, on the causal pathway between hypertension and severe COVID-19.
Majority of effect of hypertension on severe COVID-19 was direct
The unadjusted odds ratio of the association between hypertension and severe COVID-19 was 2.33 (95% confidence interval, 2.16-2.51), the authors emphasized. They found that, after adjusting for all confounding variables, hypertension was associated with 22% higher odds of severe COVID-19 (OR, 1.22; 95% CI, 1.12-1.33), compared with normotension.
Individuals with severe COVID-19 were marginally older, more likely to be male, and more deprived, the authors said. “They were also more likely to be hypertensive, compared with individuals without severe COVID-19, and a greater proportion of individuals with severe COVID-19 had cardiovascular comorbidities.”
The majority of the effect of hypertension on development of severe COVID-19 was “direct,” they said. However, a modest proportion of the effect was mediated via cardiovascular comorbidities such as peripheral vascular disease, MI, coronary heart disease, arrhythmias, and stroke. Of note, those with a history of stroke had a 47% higher risk of severe COVID-19 and those with a history of other cardiovascular comorbidities had a 30% higher risk of severe COVID-19, the authors commented.
J-shaped relationship
Of the total of 6,517 (40%) individuals who had a diagnosis of essential hypertension at baseline, 67% were treated (41% with monotherapy, 59% with combination therapy), and 33% were untreated.
There were similar numbers of severe COVID-19 in each medication group: ACE inhibitors, 34%; angiotensin receptor blockers (ARBs), 36%; and “other” medications 34%.
In hypertensive individuals receiving antihypertensive medications, there was a “J-shaped relationship” between the level of blood pressure and risk of severe COVID-19 when using a systolic blood pressure level of 120-129 mm Hg as a reference – 150-159 mm Hg versus 120-129 mm Hg (OR 1.91; 95% CI, 1.44-2.53), > 180+ mm Hg versus 120-129 mm Hg (OR 1.93; 95% CI, 1.06-3.51).
The authors commented that there was no evidence of a higher risk of severe COVID-19 until systolic blood pressure “exceeded 150 mm Hg.”
They said it was an interesting finding that “very well-controlled” systolic blood pressure < 120 mm Hg was associated with a 40% (OR, 1.40; 95% CI, 1.11-1.78) greater odds of severe COVID-19. “This may be due to reverse causality, where low systolic blood pressure levels may indicate poorer health, such that the occurrence of severe COVID-19 may be related to underlying disease rather than the level of SBP per se,” they suggested.
The J-shaped association observed remained after multiple adjustments, including presence of known cardiovascular comorbidities, which suggested a possible “real effect” of low SBP on severe COVID-19, “at least in treated hypertensive individuals.”
Their analyses also identified that, compared with a “normal” diastolic blood pressure (80-90 mm Hg), having a diastolic blood pressure higher than 90 mm Hg was associated with higher odds of severe COVID-19.
The association between hypertension and COVID-19 was “amplified” if the individuals were treated and their BP remained uncontrolled, the authors pointed out.
There did not appear to be any difference in the risk of severe COVID-19 between individuals taking ACE inhibitors and those taking ARBs or other antihypertensive medications, the authors said.
Better understanding of underlying mechanisms needed
Individuals with hypertension who tested positive for COVID-19 had “over twice” the risk of developing severe COVID-19, compared with nonhypertensive individuals, the authors said.
They highlighted that their findings also suggest that there are “further effects” influencing the severity of COVID-19 beyond a “dichotomous” diagnosis of hypertension.
“Individuals with a higher-than-target systolic blood pressure may be less healthy, less active, suffering more severe hypertension, or have developed drug-resistant hypertension, all suggesting that the effects of hypertension have already had detrimental physiological effects on the cardiovascular system, which in turn may offer some explanation for the higher risk of severe COVID-19 with uncontrolled SBP,” they explained.
“Hypertension is an important risk factor for COVID-19,” reiterated the authors, who emphasized that a better understanding of the underlying mechanisms driving this increased risk is warranted in case of “more severe strains or other viruses” in the future.
The authors have declared no competing interests.
A version of this article first appeared on Medscape UK.
U.K. researchers have established that hypertension is associated with a 22% greater risk of severe COVID-19, with the odds of severe COVID-19 unaffected by medication type.
Hypertension “appears to be one of the commonest comorbidities in COVID-19 patients”, explained the authors of a new study, published in PLOS ONE. The authors highlighted that previous research had shown that hypertension was more prevalent in severe and fatal cases compared with all cases of COVID-19.
They pointed out, however, that whether hypertensive individuals have a higher risk of severe COVID-19, compared with nonhypertensives, and whether the absolute level of systolic blood pressure or the type of antihypertensive medication is related to this risk, remained “unclear.”
To try to answer these questions, the research team, led by University of Cambridge researchers, analyzed data from 16,134 individuals who tested positive for COVID-19 (mean age 65.3 years, 47% male, 90% white), 40% were diagnosed with essential hypertension at the analysis baseline – 22% of whom had developed severe COVID-19.
Systolic blood pressure (SBP) was categorized by 10–mm Hg ranges, starting from < 120 mm Hg up to 180+ mm Hg, with the reference category defined as 120-129 mm Hg, based on data from the SPRINT study, which demonstrated that intensive SBP lowering to below 120 mm Hg, as compared with the traditional threshold of 140 mm Hg, was beneficial. Diastolic blood pressure was categorized by 10–mm Hg ranges, starting from < 60 mm Hg up to 100+ mm Hg with 80-90 mm Hg being the reference category.
In their analyses the researchers adjusted for age, sex, body mass index, ethnicity, smoking status, diabetes status, socioeconomic status, and inflammation (C-reactive protein [CRP]), as these were proposed as potential confounders. To assess the direct effect of hypertension on COVID-19, they also adjusted for intermediate variables, including cardiovascular comorbidities and stroke, on the causal pathway between hypertension and severe COVID-19.
Majority of effect of hypertension on severe COVID-19 was direct
The unadjusted odds ratio of the association between hypertension and severe COVID-19 was 2.33 (95% confidence interval, 2.16-2.51), the authors emphasized. They found that, after adjusting for all confounding variables, hypertension was associated with 22% higher odds of severe COVID-19 (OR, 1.22; 95% CI, 1.12-1.33), compared with normotension.
Individuals with severe COVID-19 were marginally older, more likely to be male, and more deprived, the authors said. “They were also more likely to be hypertensive, compared with individuals without severe COVID-19, and a greater proportion of individuals with severe COVID-19 had cardiovascular comorbidities.”
The majority of the effect of hypertension on development of severe COVID-19 was “direct,” they said. However, a modest proportion of the effect was mediated via cardiovascular comorbidities such as peripheral vascular disease, MI, coronary heart disease, arrhythmias, and stroke. Of note, those with a history of stroke had a 47% higher risk of severe COVID-19 and those with a history of other cardiovascular comorbidities had a 30% higher risk of severe COVID-19, the authors commented.
J-shaped relationship
Of the total of 6,517 (40%) individuals who had a diagnosis of essential hypertension at baseline, 67% were treated (41% with monotherapy, 59% with combination therapy), and 33% were untreated.
There were similar numbers of severe COVID-19 in each medication group: ACE inhibitors, 34%; angiotensin receptor blockers (ARBs), 36%; and “other” medications 34%.
In hypertensive individuals receiving antihypertensive medications, there was a “J-shaped relationship” between the level of blood pressure and risk of severe COVID-19 when using a systolic blood pressure level of 120-129 mm Hg as a reference – 150-159 mm Hg versus 120-129 mm Hg (OR 1.91; 95% CI, 1.44-2.53), > 180+ mm Hg versus 120-129 mm Hg (OR 1.93; 95% CI, 1.06-3.51).
The authors commented that there was no evidence of a higher risk of severe COVID-19 until systolic blood pressure “exceeded 150 mm Hg.”
They said it was an interesting finding that “very well-controlled” systolic blood pressure < 120 mm Hg was associated with a 40% (OR, 1.40; 95% CI, 1.11-1.78) greater odds of severe COVID-19. “This may be due to reverse causality, where low systolic blood pressure levels may indicate poorer health, such that the occurrence of severe COVID-19 may be related to underlying disease rather than the level of SBP per se,” they suggested.
The J-shaped association observed remained after multiple adjustments, including presence of known cardiovascular comorbidities, which suggested a possible “real effect” of low SBP on severe COVID-19, “at least in treated hypertensive individuals.”
Their analyses also identified that, compared with a “normal” diastolic blood pressure (80-90 mm Hg), having a diastolic blood pressure higher than 90 mm Hg was associated with higher odds of severe COVID-19.
The association between hypertension and COVID-19 was “amplified” if the individuals were treated and their BP remained uncontrolled, the authors pointed out.
There did not appear to be any difference in the risk of severe COVID-19 between individuals taking ACE inhibitors and those taking ARBs or other antihypertensive medications, the authors said.
Better understanding of underlying mechanisms needed
Individuals with hypertension who tested positive for COVID-19 had “over twice” the risk of developing severe COVID-19, compared with nonhypertensive individuals, the authors said.
They highlighted that their findings also suggest that there are “further effects” influencing the severity of COVID-19 beyond a “dichotomous” diagnosis of hypertension.
“Individuals with a higher-than-target systolic blood pressure may be less healthy, less active, suffering more severe hypertension, or have developed drug-resistant hypertension, all suggesting that the effects of hypertension have already had detrimental physiological effects on the cardiovascular system, which in turn may offer some explanation for the higher risk of severe COVID-19 with uncontrolled SBP,” they explained.
“Hypertension is an important risk factor for COVID-19,” reiterated the authors, who emphasized that a better understanding of the underlying mechanisms driving this increased risk is warranted in case of “more severe strains or other viruses” in the future.
The authors have declared no competing interests.
A version of this article first appeared on Medscape UK.
FROM PLOS ONE
Novel vaccine approach halts disease after 23 years of breast cancer
A recent 6-month follow-up showed no evidence of new or recurrent disease, and scans showed regression of a distant bulky left adrenal metastasis, as well as at other sites.
A small site of residual hypermetabolism remains in the sternum, but this is thought to be related to scar tissue.
The patient, Stephanie Gangi, told Medscape Medical News that, before she entered into the trial for the novel cancer vaccine, she was “mentally and physically exhausted.” She had benefited from being diagnosed with hormone-positive breast cancer just as its treatment was evolving and progressing, which meant that, every time a treatment failed, “there was the next thing to try, which was great and kept me going.”
“But I will admit that, by age 66, and more than 20 years of cancer treatments, I was exhausted.”
Ms. Gangi, a New York City-based poet, essayist, and fiction writer, said she was “cautiously optimistic” about the cancer vaccine, but the “overriding thought was I wanted to avoid chemotherapy.”
“I was not really signing on for great outcomes, I was signing on for something that might keep chemo at bay. The biggest impact so far for me has been that, for the first time in more than a decade, I am not on any medication. That’s really amazing…and that means no side effects,” she said.
Ms. Gangi stopped the vaccine treatment this past July, and just over 3 months later, she is still “wrapping her head around” the fact that her cancer has regressed. “I’ve had breast cancer a long time,” she said, “and you can’t just snap your fingers and be fine.”
Although the two scans that she has had since the trial ended have been “astonishing,” she underlined that this is not about a ‘cure,’ but rather “clearing tumors for the first time in many years.”
“Cancer is sneaky and sinister, and it figures out how to circumvent all kinds of treatments,” she said, adding nevertheless that she is “happy and hopeful, and my family is thrilled, of course.”
Ms. Gangi was classed as having had a partial response to the cancer vaccine, one of a few in a small phase 1/2 trial at the Icahn School of Medicine at Mount Sinai in New York. One other patient also had a partial response, and one patient had a complete response.
However, six patients have progressive disease, and one has stable disease.
These results come from an interim analysis of 10 patients from the trial, and show a 30% response rate. They were presented at the recent annual meeting of the Society for Immunotherapy of Cancer.
The vaccine that was being tested combines local low-dose radiation, intramural Flt3L, which stimulates dendritic cells, and intravenous poly-ICLC, an immune stimulating factor, with the PD-1 inhibitor pembrolizumab (Keytruda).
The result is that, instead of making a vaccine in a laboratory and administering it, “we’re actually formulating it within the body,” lead author Thomas Marron, MD, PhD, professor of medicine (hematology and medical oncology) at Mount Sinai, said in an interview.
“What people don’t realize,” he said, is that bulky tumor sites contain “a lot of dead tumor, because they grow so fast and in a haphazard way.” This means that the immune system can be recruited to recognize the dead tumor and “gobble up the dead stuff that’s already there,” he added.
The hope is that the immune system will then kill not only “the tumor you are injecting into, but also tumors elsewhere in the body,” Dr. Marron said. “So you’re basically using your body’s own immune system and on and off switches to vaccinate the patient against their cancer.”
Another patient in the trial who had a complete response to the vaccine was William Morrison, with non-Hodgkin lymphoma (NHL).
Mr. Morrison was diagnosed in 2017, at which time he was enrolled onto a phase 1 trial of an earlier version of this novel vaccine treatment regimen. “Basically, they didn’t get the results they were hoping for, and I still had the lymphoma,” he said. In 2018, his indolent follicular lymphoma transformed into an aggressive diffuse large B-cell lymphoma, for which Mr. Morrison was given six cycles of chemotherapy. This put him into remission and cleared his lymphoma.
“But the remission lasted for maybe a little over a year,” he said.
The cancer came back, and at that point he was given the opportunity to enroll in the Mount Sinai trial. At the end of the treatment, “everything was clear.”
“I’ve been for PET scans every 6 months, and I just had a scan done the other week, and everything has been fine…I’ve been pretty excited. I was pretty lucky.”
“This recent one really has worked wonders,” he said, “When they gave me the good news the other day. I felt like a big weight had been lifted.”
Mr. Morrison also said that he did not experience any serious adverse events while being treated with the vaccine. “Other than a few minor things, I tolerated it pretty well,” he said.
In contrast, Ms. Gangi said she experienced “intense” flu-like symptoms that started in the first few days after the treatment and lasted for a couple of days.
Need to improve response rate
The current trial achieved responses in 30% of patients, which “is great, [but] we want to be at 100%,” said Dr. Marron.
“What we’re doing in the laboratory right now is using this as an opportunity to study what it is that’s special about those three people who responded and what’s not happening in the other seven people, and we have some initial data that we’re analyzing,” he said.
“We are seeing that the patients who responded have a much more robust response to the Ft3L in particular…and that could suggest that maybe we need a better Ft3L, or we could think about other ways to potentially manipulate this vaccine.
“Most of the patients who are referred to me are people who have run out of options…and that usually means they’ve had many different types of chemotherapy,” Dr. Marron commented. For example, Ms. Gangi had already been through 12 different chemotherapy regimens.
Chemotherapy suppresses the immune system, but it’s not only that — also having an effect are all the other treatments aimed at reducing nausea and allergic reactions to the anti-cancer therapy, Dr. Marron explained.
“By the time that I see a patient,” Dr. Marron said, “oftentimes their immune system is not optimal. So another way in which we would hope to see better responses is by moving this vaccine earlier in the treatment paradigm, and administering it to patients as their first or second treatment.”
Senior author Joshua Brody, MD, director of the Lymphoma Immunotherapy Program at Mount Sinai’s Tisch Cancer Institute, added that it “might be easy” to incorporate the vaccine into earlier lines of therapy.
He said in an interview that both immunotherapy and radiation therapy are “standard” treatments, and the key is “adding multiple ingredients together that don’t have cumulative toxicity.”
“You can’t just chemo one plus chemo two, because they have some of the same toxicities, but the delightful thing here is this therapy had been quite safe.
“So in theory it would be fairly easy to incorporate this into earlier lines of therapy, once we can get a bit more proof of principle,” Dr. Brody said.
Approached for comment, Ann W. Silk, MD, said that the results are “particularly impressive because we know anti-PD-1 plus radiation therapy does not work in hormone-positive breast cancer or lymphoma.”
Dr. Silk, an oncologist at the Dana-Farber Cancer Institute and assistant professor of medicine at Harvard Medical School in Boston, said in an interview that one advantage of this vaccine is that it “is not restricted to a certain number of antigens and does not rely on an algorithm.”
“I would love to see more data in hormone-positive metastatic breast cancer patients,” she added. “I would use this approach after the hormonal treatments stop working, but before chemotherapy.”
Dr. Silk also said that the safety profile “looks quite good, and I imagine this approach would result in a much better quality of life for patients as compared to chemotherapy.”
Details of the trial and results
The Mount Sinai researchers had previously developed a personalized genomic cancer vaccine, PGV-001, which showed promise in a phase 1 trial in 13 patients with solid tumors or multiple myeloma and a high risk of recurrence after surgery or autologous stem cell transplant.
Next, they worked to develop the concept further to turn the tumor into its own vaccine, which involved inducing anti-tumor responses in indolent NHL, which typically responds poorly to checkpoint blockade, by combining Ft3L, low-dose irradiation, and poly-ICLC.
The next phase 1 trial showed that this approach was feasible, but preclinical modeling suggested that the addition of PD-1 blockade could improve the cure rates. The researchers therefore conducted the current trial, recruiting 10 patients with indolent NHL, metastatic breast cancer, or head and neck squamous cell carcinoma (HNSCC).
Patients were given local radiation therapy on days 1 and 2, and intramural Ft3L to the same tumor on day 9, followed by eight intravenous injections of poly-ICLC over 6 weeks. On day 23, they received their first of eight doses of pembrolizumab.
Dr. Marron explained that the radiotherapy increases the amount of dead material for the immune system to work on by “killing some of the tumor cells,” adding: “We’re not trying to kill the whole tumor with the radiation…it just starts the process of releasing some more of that dead stuff.”
He explained that Ft3L is a human growth factor that simulates dendritic cells, “which I always say are the professor cells of the immune system,” as they tell the body “what’s good and what’s bad.”
The poly-ICLC is “basically like a fake virus,” Dr. Marron said, as it “turns on those immune cells that have taken up the tumor antigen in the neighborhood” of the tumor, so they “teach the immune system that there is something bad”.
Finally, the pembrolizumab is there to “take the foot off the brake of the immune system” and “grease the wheels a bit more,” he added, even though it does not work in all patients, or in all tumor types, including indolent NHL.
The trial was planned in two phases. In the first part, six patients were enrolled to assess the safety of the approach; the phase 2 stage of the trial followed a Simon’s Two-Stage design, with the aim of recruiting seven patients of each tumor type, followed by a further 12 patients if they showed a response.
The current interim analysis that was presented at the SITC meeting focused on the first 10 patients in the phase 2 part, who were enrolled between April 2019 and July 2022. This included six patients with metastatic breast cancer, three with indolent NHL, and one with HNSCC, all of whom completed their first disease response assessment.
All patients experienced treatment-related adverse events, largely comprising low-grade injection site reactions and flu-like symptoms linked to the poly-ICLC injections.
One patient experienced grade 3 pembrolizumab-related colitis, while another had self-resolving grade 3 fever following poly-ICLC injection.
The study was sponsored by Icahn School of Medicine at Mount Sinai and conducted in collaboration with Merck Sharp & Dohme LLC and Celldex Therapeutics. No relevant financial relationships were reported.
A version of this article first appeared on Medscape.com.
A recent 6-month follow-up showed no evidence of new or recurrent disease, and scans showed regression of a distant bulky left adrenal metastasis, as well as at other sites.
A small site of residual hypermetabolism remains in the sternum, but this is thought to be related to scar tissue.
The patient, Stephanie Gangi, told Medscape Medical News that, before she entered into the trial for the novel cancer vaccine, she was “mentally and physically exhausted.” She had benefited from being diagnosed with hormone-positive breast cancer just as its treatment was evolving and progressing, which meant that, every time a treatment failed, “there was the next thing to try, which was great and kept me going.”
“But I will admit that, by age 66, and more than 20 years of cancer treatments, I was exhausted.”
Ms. Gangi, a New York City-based poet, essayist, and fiction writer, said she was “cautiously optimistic” about the cancer vaccine, but the “overriding thought was I wanted to avoid chemotherapy.”
“I was not really signing on for great outcomes, I was signing on for something that might keep chemo at bay. The biggest impact so far for me has been that, for the first time in more than a decade, I am not on any medication. That’s really amazing…and that means no side effects,” she said.
Ms. Gangi stopped the vaccine treatment this past July, and just over 3 months later, she is still “wrapping her head around” the fact that her cancer has regressed. “I’ve had breast cancer a long time,” she said, “and you can’t just snap your fingers and be fine.”
Although the two scans that she has had since the trial ended have been “astonishing,” she underlined that this is not about a ‘cure,’ but rather “clearing tumors for the first time in many years.”
“Cancer is sneaky and sinister, and it figures out how to circumvent all kinds of treatments,” she said, adding nevertheless that she is “happy and hopeful, and my family is thrilled, of course.”
Ms. Gangi was classed as having had a partial response to the cancer vaccine, one of a few in a small phase 1/2 trial at the Icahn School of Medicine at Mount Sinai in New York. One other patient also had a partial response, and one patient had a complete response.
However, six patients have progressive disease, and one has stable disease.
These results come from an interim analysis of 10 patients from the trial, and show a 30% response rate. They were presented at the recent annual meeting of the Society for Immunotherapy of Cancer.
The vaccine that was being tested combines local low-dose radiation, intramural Flt3L, which stimulates dendritic cells, and intravenous poly-ICLC, an immune stimulating factor, with the PD-1 inhibitor pembrolizumab (Keytruda).
The result is that, instead of making a vaccine in a laboratory and administering it, “we’re actually formulating it within the body,” lead author Thomas Marron, MD, PhD, professor of medicine (hematology and medical oncology) at Mount Sinai, said in an interview.
“What people don’t realize,” he said, is that bulky tumor sites contain “a lot of dead tumor, because they grow so fast and in a haphazard way.” This means that the immune system can be recruited to recognize the dead tumor and “gobble up the dead stuff that’s already there,” he added.
The hope is that the immune system will then kill not only “the tumor you are injecting into, but also tumors elsewhere in the body,” Dr. Marron said. “So you’re basically using your body’s own immune system and on and off switches to vaccinate the patient against their cancer.”
Another patient in the trial who had a complete response to the vaccine was William Morrison, with non-Hodgkin lymphoma (NHL).
Mr. Morrison was diagnosed in 2017, at which time he was enrolled onto a phase 1 trial of an earlier version of this novel vaccine treatment regimen. “Basically, they didn’t get the results they were hoping for, and I still had the lymphoma,” he said. In 2018, his indolent follicular lymphoma transformed into an aggressive diffuse large B-cell lymphoma, for which Mr. Morrison was given six cycles of chemotherapy. This put him into remission and cleared his lymphoma.
“But the remission lasted for maybe a little over a year,” he said.
The cancer came back, and at that point he was given the opportunity to enroll in the Mount Sinai trial. At the end of the treatment, “everything was clear.”
“I’ve been for PET scans every 6 months, and I just had a scan done the other week, and everything has been fine…I’ve been pretty excited. I was pretty lucky.”
“This recent one really has worked wonders,” he said, “When they gave me the good news the other day. I felt like a big weight had been lifted.”
Mr. Morrison also said that he did not experience any serious adverse events while being treated with the vaccine. “Other than a few minor things, I tolerated it pretty well,” he said.
In contrast, Ms. Gangi said she experienced “intense” flu-like symptoms that started in the first few days after the treatment and lasted for a couple of days.
Need to improve response rate
The current trial achieved responses in 30% of patients, which “is great, [but] we want to be at 100%,” said Dr. Marron.
“What we’re doing in the laboratory right now is using this as an opportunity to study what it is that’s special about those three people who responded and what’s not happening in the other seven people, and we have some initial data that we’re analyzing,” he said.
“We are seeing that the patients who responded have a much more robust response to the Ft3L in particular…and that could suggest that maybe we need a better Ft3L, or we could think about other ways to potentially manipulate this vaccine.
“Most of the patients who are referred to me are people who have run out of options…and that usually means they’ve had many different types of chemotherapy,” Dr. Marron commented. For example, Ms. Gangi had already been through 12 different chemotherapy regimens.
Chemotherapy suppresses the immune system, but it’s not only that — also having an effect are all the other treatments aimed at reducing nausea and allergic reactions to the anti-cancer therapy, Dr. Marron explained.
“By the time that I see a patient,” Dr. Marron said, “oftentimes their immune system is not optimal. So another way in which we would hope to see better responses is by moving this vaccine earlier in the treatment paradigm, and administering it to patients as their first or second treatment.”
Senior author Joshua Brody, MD, director of the Lymphoma Immunotherapy Program at Mount Sinai’s Tisch Cancer Institute, added that it “might be easy” to incorporate the vaccine into earlier lines of therapy.
He said in an interview that both immunotherapy and radiation therapy are “standard” treatments, and the key is “adding multiple ingredients together that don’t have cumulative toxicity.”
“You can’t just chemo one plus chemo two, because they have some of the same toxicities, but the delightful thing here is this therapy had been quite safe.
“So in theory it would be fairly easy to incorporate this into earlier lines of therapy, once we can get a bit more proof of principle,” Dr. Brody said.
Approached for comment, Ann W. Silk, MD, said that the results are “particularly impressive because we know anti-PD-1 plus radiation therapy does not work in hormone-positive breast cancer or lymphoma.”
Dr. Silk, an oncologist at the Dana-Farber Cancer Institute and assistant professor of medicine at Harvard Medical School in Boston, said in an interview that one advantage of this vaccine is that it “is not restricted to a certain number of antigens and does not rely on an algorithm.”
“I would love to see more data in hormone-positive metastatic breast cancer patients,” she added. “I would use this approach after the hormonal treatments stop working, but before chemotherapy.”
Dr. Silk also said that the safety profile “looks quite good, and I imagine this approach would result in a much better quality of life for patients as compared to chemotherapy.”
Details of the trial and results
The Mount Sinai researchers had previously developed a personalized genomic cancer vaccine, PGV-001, which showed promise in a phase 1 trial in 13 patients with solid tumors or multiple myeloma and a high risk of recurrence after surgery or autologous stem cell transplant.
Next, they worked to develop the concept further to turn the tumor into its own vaccine, which involved inducing anti-tumor responses in indolent NHL, which typically responds poorly to checkpoint blockade, by combining Ft3L, low-dose irradiation, and poly-ICLC.
The next phase 1 trial showed that this approach was feasible, but preclinical modeling suggested that the addition of PD-1 blockade could improve the cure rates. The researchers therefore conducted the current trial, recruiting 10 patients with indolent NHL, metastatic breast cancer, or head and neck squamous cell carcinoma (HNSCC).
Patients were given local radiation therapy on days 1 and 2, and intramural Ft3L to the same tumor on day 9, followed by eight intravenous injections of poly-ICLC over 6 weeks. On day 23, they received their first of eight doses of pembrolizumab.
Dr. Marron explained that the radiotherapy increases the amount of dead material for the immune system to work on by “killing some of the tumor cells,” adding: “We’re not trying to kill the whole tumor with the radiation…it just starts the process of releasing some more of that dead stuff.”
He explained that Ft3L is a human growth factor that simulates dendritic cells, “which I always say are the professor cells of the immune system,” as they tell the body “what’s good and what’s bad.”
The poly-ICLC is “basically like a fake virus,” Dr. Marron said, as it “turns on those immune cells that have taken up the tumor antigen in the neighborhood” of the tumor, so they “teach the immune system that there is something bad”.
Finally, the pembrolizumab is there to “take the foot off the brake of the immune system” and “grease the wheels a bit more,” he added, even though it does not work in all patients, or in all tumor types, including indolent NHL.
The trial was planned in two phases. In the first part, six patients were enrolled to assess the safety of the approach; the phase 2 stage of the trial followed a Simon’s Two-Stage design, with the aim of recruiting seven patients of each tumor type, followed by a further 12 patients if they showed a response.
The current interim analysis that was presented at the SITC meeting focused on the first 10 patients in the phase 2 part, who were enrolled between April 2019 and July 2022. This included six patients with metastatic breast cancer, three with indolent NHL, and one with HNSCC, all of whom completed their first disease response assessment.
All patients experienced treatment-related adverse events, largely comprising low-grade injection site reactions and flu-like symptoms linked to the poly-ICLC injections.
One patient experienced grade 3 pembrolizumab-related colitis, while another had self-resolving grade 3 fever following poly-ICLC injection.
The study was sponsored by Icahn School of Medicine at Mount Sinai and conducted in collaboration with Merck Sharp & Dohme LLC and Celldex Therapeutics. No relevant financial relationships were reported.
A version of this article first appeared on Medscape.com.
A recent 6-month follow-up showed no evidence of new or recurrent disease, and scans showed regression of a distant bulky left adrenal metastasis, as well as at other sites.
A small site of residual hypermetabolism remains in the sternum, but this is thought to be related to scar tissue.
The patient, Stephanie Gangi, told Medscape Medical News that, before she entered into the trial for the novel cancer vaccine, she was “mentally and physically exhausted.” She had benefited from being diagnosed with hormone-positive breast cancer just as its treatment was evolving and progressing, which meant that, every time a treatment failed, “there was the next thing to try, which was great and kept me going.”
“But I will admit that, by age 66, and more than 20 years of cancer treatments, I was exhausted.”
Ms. Gangi, a New York City-based poet, essayist, and fiction writer, said she was “cautiously optimistic” about the cancer vaccine, but the “overriding thought was I wanted to avoid chemotherapy.”
“I was not really signing on for great outcomes, I was signing on for something that might keep chemo at bay. The biggest impact so far for me has been that, for the first time in more than a decade, I am not on any medication. That’s really amazing…and that means no side effects,” she said.
Ms. Gangi stopped the vaccine treatment this past July, and just over 3 months later, she is still “wrapping her head around” the fact that her cancer has regressed. “I’ve had breast cancer a long time,” she said, “and you can’t just snap your fingers and be fine.”
Although the two scans that she has had since the trial ended have been “astonishing,” she underlined that this is not about a ‘cure,’ but rather “clearing tumors for the first time in many years.”
“Cancer is sneaky and sinister, and it figures out how to circumvent all kinds of treatments,” she said, adding nevertheless that she is “happy and hopeful, and my family is thrilled, of course.”
Ms. Gangi was classed as having had a partial response to the cancer vaccine, one of a few in a small phase 1/2 trial at the Icahn School of Medicine at Mount Sinai in New York. One other patient also had a partial response, and one patient had a complete response.
However, six patients have progressive disease, and one has stable disease.
These results come from an interim analysis of 10 patients from the trial, and show a 30% response rate. They were presented at the recent annual meeting of the Society for Immunotherapy of Cancer.
The vaccine that was being tested combines local low-dose radiation, intramural Flt3L, which stimulates dendritic cells, and intravenous poly-ICLC, an immune stimulating factor, with the PD-1 inhibitor pembrolizumab (Keytruda).
The result is that, instead of making a vaccine in a laboratory and administering it, “we’re actually formulating it within the body,” lead author Thomas Marron, MD, PhD, professor of medicine (hematology and medical oncology) at Mount Sinai, said in an interview.
“What people don’t realize,” he said, is that bulky tumor sites contain “a lot of dead tumor, because they grow so fast and in a haphazard way.” This means that the immune system can be recruited to recognize the dead tumor and “gobble up the dead stuff that’s already there,” he added.
The hope is that the immune system will then kill not only “the tumor you are injecting into, but also tumors elsewhere in the body,” Dr. Marron said. “So you’re basically using your body’s own immune system and on and off switches to vaccinate the patient against their cancer.”
Another patient in the trial who had a complete response to the vaccine was William Morrison, with non-Hodgkin lymphoma (NHL).
Mr. Morrison was diagnosed in 2017, at which time he was enrolled onto a phase 1 trial of an earlier version of this novel vaccine treatment regimen. “Basically, they didn’t get the results they were hoping for, and I still had the lymphoma,” he said. In 2018, his indolent follicular lymphoma transformed into an aggressive diffuse large B-cell lymphoma, for which Mr. Morrison was given six cycles of chemotherapy. This put him into remission and cleared his lymphoma.
“But the remission lasted for maybe a little over a year,” he said.
The cancer came back, and at that point he was given the opportunity to enroll in the Mount Sinai trial. At the end of the treatment, “everything was clear.”
“I’ve been for PET scans every 6 months, and I just had a scan done the other week, and everything has been fine…I’ve been pretty excited. I was pretty lucky.”
“This recent one really has worked wonders,” he said, “When they gave me the good news the other day. I felt like a big weight had been lifted.”
Mr. Morrison also said that he did not experience any serious adverse events while being treated with the vaccine. “Other than a few minor things, I tolerated it pretty well,” he said.
In contrast, Ms. Gangi said she experienced “intense” flu-like symptoms that started in the first few days after the treatment and lasted for a couple of days.
Need to improve response rate
The current trial achieved responses in 30% of patients, which “is great, [but] we want to be at 100%,” said Dr. Marron.
“What we’re doing in the laboratory right now is using this as an opportunity to study what it is that’s special about those three people who responded and what’s not happening in the other seven people, and we have some initial data that we’re analyzing,” he said.
“We are seeing that the patients who responded have a much more robust response to the Ft3L in particular…and that could suggest that maybe we need a better Ft3L, or we could think about other ways to potentially manipulate this vaccine.
“Most of the patients who are referred to me are people who have run out of options…and that usually means they’ve had many different types of chemotherapy,” Dr. Marron commented. For example, Ms. Gangi had already been through 12 different chemotherapy regimens.
Chemotherapy suppresses the immune system, but it’s not only that — also having an effect are all the other treatments aimed at reducing nausea and allergic reactions to the anti-cancer therapy, Dr. Marron explained.
“By the time that I see a patient,” Dr. Marron said, “oftentimes their immune system is not optimal. So another way in which we would hope to see better responses is by moving this vaccine earlier in the treatment paradigm, and administering it to patients as their first or second treatment.”
Senior author Joshua Brody, MD, director of the Lymphoma Immunotherapy Program at Mount Sinai’s Tisch Cancer Institute, added that it “might be easy” to incorporate the vaccine into earlier lines of therapy.
He said in an interview that both immunotherapy and radiation therapy are “standard” treatments, and the key is “adding multiple ingredients together that don’t have cumulative toxicity.”
“You can’t just chemo one plus chemo two, because they have some of the same toxicities, but the delightful thing here is this therapy had been quite safe.
“So in theory it would be fairly easy to incorporate this into earlier lines of therapy, once we can get a bit more proof of principle,” Dr. Brody said.
Approached for comment, Ann W. Silk, MD, said that the results are “particularly impressive because we know anti-PD-1 plus radiation therapy does not work in hormone-positive breast cancer or lymphoma.”
Dr. Silk, an oncologist at the Dana-Farber Cancer Institute and assistant professor of medicine at Harvard Medical School in Boston, said in an interview that one advantage of this vaccine is that it “is not restricted to a certain number of antigens and does not rely on an algorithm.”
“I would love to see more data in hormone-positive metastatic breast cancer patients,” she added. “I would use this approach after the hormonal treatments stop working, but before chemotherapy.”
Dr. Silk also said that the safety profile “looks quite good, and I imagine this approach would result in a much better quality of life for patients as compared to chemotherapy.”
Details of the trial and results
The Mount Sinai researchers had previously developed a personalized genomic cancer vaccine, PGV-001, which showed promise in a phase 1 trial in 13 patients with solid tumors or multiple myeloma and a high risk of recurrence after surgery or autologous stem cell transplant.
Next, they worked to develop the concept further to turn the tumor into its own vaccine, which involved inducing anti-tumor responses in indolent NHL, which typically responds poorly to checkpoint blockade, by combining Ft3L, low-dose irradiation, and poly-ICLC.
The next phase 1 trial showed that this approach was feasible, but preclinical modeling suggested that the addition of PD-1 blockade could improve the cure rates. The researchers therefore conducted the current trial, recruiting 10 patients with indolent NHL, metastatic breast cancer, or head and neck squamous cell carcinoma (HNSCC).
Patients were given local radiation therapy on days 1 and 2, and intramural Ft3L to the same tumor on day 9, followed by eight intravenous injections of poly-ICLC over 6 weeks. On day 23, they received their first of eight doses of pembrolizumab.
Dr. Marron explained that the radiotherapy increases the amount of dead material for the immune system to work on by “killing some of the tumor cells,” adding: “We’re not trying to kill the whole tumor with the radiation…it just starts the process of releasing some more of that dead stuff.”
He explained that Ft3L is a human growth factor that simulates dendritic cells, “which I always say are the professor cells of the immune system,” as they tell the body “what’s good and what’s bad.”
The poly-ICLC is “basically like a fake virus,” Dr. Marron said, as it “turns on those immune cells that have taken up the tumor antigen in the neighborhood” of the tumor, so they “teach the immune system that there is something bad”.
Finally, the pembrolizumab is there to “take the foot off the brake of the immune system” and “grease the wheels a bit more,” he added, even though it does not work in all patients, or in all tumor types, including indolent NHL.
The trial was planned in two phases. In the first part, six patients were enrolled to assess the safety of the approach; the phase 2 stage of the trial followed a Simon’s Two-Stage design, with the aim of recruiting seven patients of each tumor type, followed by a further 12 patients if they showed a response.
The current interim analysis that was presented at the SITC meeting focused on the first 10 patients in the phase 2 part, who were enrolled between April 2019 and July 2022. This included six patients with metastatic breast cancer, three with indolent NHL, and one with HNSCC, all of whom completed their first disease response assessment.
All patients experienced treatment-related adverse events, largely comprising low-grade injection site reactions and flu-like symptoms linked to the poly-ICLC injections.
One patient experienced grade 3 pembrolizumab-related colitis, while another had self-resolving grade 3 fever following poly-ICLC injection.
The study was sponsored by Icahn School of Medicine at Mount Sinai and conducted in collaboration with Merck Sharp & Dohme LLC and Celldex Therapeutics. No relevant financial relationships were reported.
A version of this article first appeared on Medscape.com.
FROM SITC 2022
EHR reminders boost well-child visits, vax rates
Reminder messages sent through electronic health records (EHRs) to patient portals increased rates of scheduling and completion of well-child visits for those overdue for well care, as well as vaccination rates, according to data published today in JAMA Network Open.
Anne E. Berset, BA, with general and community pediatrics at Cincinnati Children’s Hospital, led a randomized clinical trial conducted at three academic primary care practices from July 30 to Oct. 4, 2021.
The practices serve a mostly non-Hispanic Black, low-income population and provide more than 60,000 visits a year to 30,000 patients.
The study population included 945 patients, 62.4% of them non-Hispanic Black and 807 (85.4%) covered by public insurance.
Standard message and tailored message compared
The study population was randomized to get either a standard reminder message, a tailored message, or no message (control group).
The standard messages referenced the patient’s first name and reminded parents their child was due for a well-child visit, and asked them to schedule it using the portal or by calling the number provided.
The tailored message added the date of the last well-child check-up “to address distortions of time perception experienced during the pandemic,” the authors write.
The primary outcome was whether the well-child visit was completed within 8 weeks. Other outcomes included whether a well-child visit was scheduled within 2 weeks of the first reminder message and whether the patient received a COVID-19 vaccine within 8 weeks of the reminder.
Reminders outperform control group
There was a significant increase in scheduling and completion of appointments after parents received the standard and tailored messages compared with controls.
The results with the COVID reminders were particularly striking. While only 3.7% of the eligible patients got a vaccine when they got no message, 17.3% got the vaccine in that time frame when they got the standard message. Interestingly, only 2.7% received the shot after the tailored message.
J. Howard Smart, MD, with Sharp HealthCare in San Diego, told this publication he was not surprised that a patient portal reminder would increase the rate of scheduling overdue well-child visits. “Parents may indeed have lost track of when their children need to have check-ups” during the pandemic, he said.
Reminders without tailored information more effective
He said he was surprised, just as the investigators were, that the reminders without the tailored information seemed to be more effective for some outcomes.
The authors explained that a focus group found “that families with patient portal accounts prefer straightforward, brief, and user-friendly messages.”
That may help explain why the standard message outperformed the tailored message, the authors write.
“Families may have been distracted by the additional information related to the child’s age and date of last WCC [well-child check-up] in the tailored message,” they write.
Dr. Smart said, “Their discussion of why this might be the case sounds reasonable. Simple, short messages may be better received.”
“Evidence like this should stimulate portal software vendors to make this kind of outgoing messaging easy to set up for providers,” he said.
Simple measure, large effect
Tim Joos, MD, a pediatrician in Seattle, told this publication he “was impressed by how such a simple measure increased rates of well-child care completion.”
Minority and low-income patients have traditionally had more access challenges to completing health care visits and vaccinations on schedule, he noted, so he was glad to see positive results from the intervention in this study population.
The authors note that research on portal messages have largely looked at use in non-Hispanic Whites and the privately insured.
Dr. Joos said the intervention also offers convenience in that once patients get into the portal, they can schedule an appointment there without having to wait on the phone or leave a message.
Funding/support was received from the Center for Clinical and Translational Science and Training at the University of Cincinnati, which is funded by the National Institutes of Health. William B. Brinkman, MD, MEd, MSc, one of the study authors, holds common stock in Pfizer, Merck, Abbott Laboratories, Viatris, and Johnson & Johnson outside the submitted work. No other author disclosures were reported. Dr. Smart and Dr. Joos declared no relevant financial relationships.
Reminder messages sent through electronic health records (EHRs) to patient portals increased rates of scheduling and completion of well-child visits for those overdue for well care, as well as vaccination rates, according to data published today in JAMA Network Open.
Anne E. Berset, BA, with general and community pediatrics at Cincinnati Children’s Hospital, led a randomized clinical trial conducted at three academic primary care practices from July 30 to Oct. 4, 2021.
The practices serve a mostly non-Hispanic Black, low-income population and provide more than 60,000 visits a year to 30,000 patients.
The study population included 945 patients, 62.4% of them non-Hispanic Black and 807 (85.4%) covered by public insurance.
Standard message and tailored message compared
The study population was randomized to get either a standard reminder message, a tailored message, or no message (control group).
The standard messages referenced the patient’s first name and reminded parents their child was due for a well-child visit, and asked them to schedule it using the portal or by calling the number provided.
The tailored message added the date of the last well-child check-up “to address distortions of time perception experienced during the pandemic,” the authors write.
The primary outcome was whether the well-child visit was completed within 8 weeks. Other outcomes included whether a well-child visit was scheduled within 2 weeks of the first reminder message and whether the patient received a COVID-19 vaccine within 8 weeks of the reminder.
Reminders outperform control group
There was a significant increase in scheduling and completion of appointments after parents received the standard and tailored messages compared with controls.
The results with the COVID reminders were particularly striking. While only 3.7% of the eligible patients got a vaccine when they got no message, 17.3% got the vaccine in that time frame when they got the standard message. Interestingly, only 2.7% received the shot after the tailored message.
J. Howard Smart, MD, with Sharp HealthCare in San Diego, told this publication he was not surprised that a patient portal reminder would increase the rate of scheduling overdue well-child visits. “Parents may indeed have lost track of when their children need to have check-ups” during the pandemic, he said.
Reminders without tailored information more effective
He said he was surprised, just as the investigators were, that the reminders without the tailored information seemed to be more effective for some outcomes.
The authors explained that a focus group found “that families with patient portal accounts prefer straightforward, brief, and user-friendly messages.”
That may help explain why the standard message outperformed the tailored message, the authors write.
“Families may have been distracted by the additional information related to the child’s age and date of last WCC [well-child check-up] in the tailored message,” they write.
Dr. Smart said, “Their discussion of why this might be the case sounds reasonable. Simple, short messages may be better received.”
“Evidence like this should stimulate portal software vendors to make this kind of outgoing messaging easy to set up for providers,” he said.
Simple measure, large effect
Tim Joos, MD, a pediatrician in Seattle, told this publication he “was impressed by how such a simple measure increased rates of well-child care completion.”
Minority and low-income patients have traditionally had more access challenges to completing health care visits and vaccinations on schedule, he noted, so he was glad to see positive results from the intervention in this study population.
The authors note that research on portal messages have largely looked at use in non-Hispanic Whites and the privately insured.
Dr. Joos said the intervention also offers convenience in that once patients get into the portal, they can schedule an appointment there without having to wait on the phone or leave a message.
Funding/support was received from the Center for Clinical and Translational Science and Training at the University of Cincinnati, which is funded by the National Institutes of Health. William B. Brinkman, MD, MEd, MSc, one of the study authors, holds common stock in Pfizer, Merck, Abbott Laboratories, Viatris, and Johnson & Johnson outside the submitted work. No other author disclosures were reported. Dr. Smart and Dr. Joos declared no relevant financial relationships.
Reminder messages sent through electronic health records (EHRs) to patient portals increased rates of scheduling and completion of well-child visits for those overdue for well care, as well as vaccination rates, according to data published today in JAMA Network Open.
Anne E. Berset, BA, with general and community pediatrics at Cincinnati Children’s Hospital, led a randomized clinical trial conducted at three academic primary care practices from July 30 to Oct. 4, 2021.
The practices serve a mostly non-Hispanic Black, low-income population and provide more than 60,000 visits a year to 30,000 patients.
The study population included 945 patients, 62.4% of them non-Hispanic Black and 807 (85.4%) covered by public insurance.
Standard message and tailored message compared
The study population was randomized to get either a standard reminder message, a tailored message, or no message (control group).
The standard messages referenced the patient’s first name and reminded parents their child was due for a well-child visit, and asked them to schedule it using the portal or by calling the number provided.
The tailored message added the date of the last well-child check-up “to address distortions of time perception experienced during the pandemic,” the authors write.
The primary outcome was whether the well-child visit was completed within 8 weeks. Other outcomes included whether a well-child visit was scheduled within 2 weeks of the first reminder message and whether the patient received a COVID-19 vaccine within 8 weeks of the reminder.
Reminders outperform control group
There was a significant increase in scheduling and completion of appointments after parents received the standard and tailored messages compared with controls.
The results with the COVID reminders were particularly striking. While only 3.7% of the eligible patients got a vaccine when they got no message, 17.3% got the vaccine in that time frame when they got the standard message. Interestingly, only 2.7% received the shot after the tailored message.
J. Howard Smart, MD, with Sharp HealthCare in San Diego, told this publication he was not surprised that a patient portal reminder would increase the rate of scheduling overdue well-child visits. “Parents may indeed have lost track of when their children need to have check-ups” during the pandemic, he said.
Reminders without tailored information more effective
He said he was surprised, just as the investigators were, that the reminders without the tailored information seemed to be more effective for some outcomes.
The authors explained that a focus group found “that families with patient portal accounts prefer straightforward, brief, and user-friendly messages.”
That may help explain why the standard message outperformed the tailored message, the authors write.
“Families may have been distracted by the additional information related to the child’s age and date of last WCC [well-child check-up] in the tailored message,” they write.
Dr. Smart said, “Their discussion of why this might be the case sounds reasonable. Simple, short messages may be better received.”
“Evidence like this should stimulate portal software vendors to make this kind of outgoing messaging easy to set up for providers,” he said.
Simple measure, large effect
Tim Joos, MD, a pediatrician in Seattle, told this publication he “was impressed by how such a simple measure increased rates of well-child care completion.”
Minority and low-income patients have traditionally had more access challenges to completing health care visits and vaccinations on schedule, he noted, so he was glad to see positive results from the intervention in this study population.
The authors note that research on portal messages have largely looked at use in non-Hispanic Whites and the privately insured.
Dr. Joos said the intervention also offers convenience in that once patients get into the portal, they can schedule an appointment there without having to wait on the phone or leave a message.
Funding/support was received from the Center for Clinical and Translational Science and Training at the University of Cincinnati, which is funded by the National Institutes of Health. William B. Brinkman, MD, MEd, MSc, one of the study authors, holds common stock in Pfizer, Merck, Abbott Laboratories, Viatris, and Johnson & Johnson outside the submitted work. No other author disclosures were reported. Dr. Smart and Dr. Joos declared no relevant financial relationships.
FROM JAMA NETWORK OPEN
FDA approves first-ever agent to delay type 1 diabetes onset
“Today’s approval of a first-in-class therapy adds an important new treatment option for certain at-risk patients,” said John Sharretts, MD, director of the Division of Diabetes, Lipid Disorders, and Obesity in the FDA’s Center for Drug Evaluation and Research. “The drug’s potential to delay clinical diagnosis of type 1 diabetes may provide patients with months to years without the burdens of disease.”
The agent, which interferes with T-cell-mediated autoimmune destruction of pancreatic beta cells, is the first disease-modifying therapy for impeding progression of type 1 diabetes. It is administered by intravenous infusion once daily for 14 consecutive days.
The specific indication is “to delay the onset of stage 3 type 1 diabetes in adults and pediatric patients 8 years and older who currently have stage 2 type 1 diabetes.” In type 1 diabetes staging, adopted in 2015, stage 1 is defined as the presence of beta cell autoimmunity with two or more islet autoantibodies with normoglycemia, stage 2 is beta-cell autoimmunity with dysglycemia yet asymptomatic, and stage 3 is the onset of symptomatic type 1 diabetes.
Stage 2 type 1 diabetes is associated with a nearly 100% lifetime risk of progression to clinical (stage 3) type 1 diabetes and a 75% risk of developing the condition within 5 years.
The FDA had previously rejected teplizumab for this indication in July 2021, despite a prior endorsement from an advisory panel in May 2021.
Now, with the FDA approval, Provention Bio cofounder and CEO Ashleigh Palmer said in a statement, “This is a historic occasion for the T1D community and a paradigm shifting breakthrough ... It cannot be emphasized enough how precious a delay in the onset of stage 3 T1D can be from a patient and family perspective; more time to live without and, when necessary, prepare for the burdens, complications, and risks associated with stage 3 disease.”
T1D onset delayed by 2 years
In 2019, a pivotal phase 2, randomized, placebo-controlled trial involving 76 at-risk children and adults aged 8 years and older showed that a single 14-day treatment of daily intravenous infusions of teplizumab in 44 patients resulted in a significant median 2-year delay to onset of clinical type 1 diabetes compared with 32 who received placebo.
Those “game changer” data were presented at the American Diabetes Association (ADA) annual meeting in June 2019 and simultaneously published in the New England Journal of Medicine.
Three-year data were presented at the June 2020 ADA meeting and published in March 2021 in Science Translational Medicine, by Emily K. Sims, MD, department of pediatrics, Indiana University, Indianapolis, and colleagues.
At a median follow-up of 923 days, 50% of those randomly assigned to teplizumab remained diabetes free, compared with 22% of those who received placebo infusions (hazard ratio, 0.457; P = .01). The teplizumab group had a greater average C-peptide area under the curve compared with placebo, reflecting improved beta-cell function (1.96 vs. 1.68 pmol/mL; P = .006).
C-peptide levels declined over time in the placebo group but stabilized in those receiving teplizumab (P = .0015).
“The mid-range time from randomization to stage 3 type 1 diabetes diagnosis was 50 months for the patients who received Tzield and 25 months for those who received a placebo. This represents a statistically significant delay in the development of stage 3 type 1 diabetes,” according to the FDA statement.
The most common side effects of Tzield include lymphopenia (73% teplizumab vs. 6% placebo), rash (36% vs. 0%), leukopenia (221% vs. 0%), and headache (11% vs. 6%). Label warnings and precautions include monitoring for cytokine release syndrome, risk for serious infections, and avoidance of live, inactivated, and mRNA vaccines.
This approval is likely to accelerate discussion about universal autoantibody screening. Currently, most individuals identified as having preclinical type 1 diabetes are first-degree relatives of people with type 1 diabetes identified through the federally funded TrialNet program. In December 2020, the type 1 diabetes research and advocacy organization JDRF began offering a $55 home blood test to screen for the antibodies, and other screening programs have been launched in the United States and Europe.
Previous studies have examined cost-effectiveness of universal screening in children and the optimal ages that such screening should take place.
In October, Provention Bio announced a co-promotion agreement with Sanofi for the U.S. launch of Tzield for delay in onset of clinical T1D in at-risk individuals. Provention Bio offers financial assistance options (e.g., copay assistance) to eligible patients for out-of-pocket costs.
A version of this article first appeared on Medscape.com.
“Today’s approval of a first-in-class therapy adds an important new treatment option for certain at-risk patients,” said John Sharretts, MD, director of the Division of Diabetes, Lipid Disorders, and Obesity in the FDA’s Center for Drug Evaluation and Research. “The drug’s potential to delay clinical diagnosis of type 1 diabetes may provide patients with months to years without the burdens of disease.”
The agent, which interferes with T-cell-mediated autoimmune destruction of pancreatic beta cells, is the first disease-modifying therapy for impeding progression of type 1 diabetes. It is administered by intravenous infusion once daily for 14 consecutive days.
The specific indication is “to delay the onset of stage 3 type 1 diabetes in adults and pediatric patients 8 years and older who currently have stage 2 type 1 diabetes.” In type 1 diabetes staging, adopted in 2015, stage 1 is defined as the presence of beta cell autoimmunity with two or more islet autoantibodies with normoglycemia, stage 2 is beta-cell autoimmunity with dysglycemia yet asymptomatic, and stage 3 is the onset of symptomatic type 1 diabetes.
Stage 2 type 1 diabetes is associated with a nearly 100% lifetime risk of progression to clinical (stage 3) type 1 diabetes and a 75% risk of developing the condition within 5 years.
The FDA had previously rejected teplizumab for this indication in July 2021, despite a prior endorsement from an advisory panel in May 2021.
Now, with the FDA approval, Provention Bio cofounder and CEO Ashleigh Palmer said in a statement, “This is a historic occasion for the T1D community and a paradigm shifting breakthrough ... It cannot be emphasized enough how precious a delay in the onset of stage 3 T1D can be from a patient and family perspective; more time to live without and, when necessary, prepare for the burdens, complications, and risks associated with stage 3 disease.”
T1D onset delayed by 2 years
In 2019, a pivotal phase 2, randomized, placebo-controlled trial involving 76 at-risk children and adults aged 8 years and older showed that a single 14-day treatment of daily intravenous infusions of teplizumab in 44 patients resulted in a significant median 2-year delay to onset of clinical type 1 diabetes compared with 32 who received placebo.
Those “game changer” data were presented at the American Diabetes Association (ADA) annual meeting in June 2019 and simultaneously published in the New England Journal of Medicine.
Three-year data were presented at the June 2020 ADA meeting and published in March 2021 in Science Translational Medicine, by Emily K. Sims, MD, department of pediatrics, Indiana University, Indianapolis, and colleagues.
At a median follow-up of 923 days, 50% of those randomly assigned to teplizumab remained diabetes free, compared with 22% of those who received placebo infusions (hazard ratio, 0.457; P = .01). The teplizumab group had a greater average C-peptide area under the curve compared with placebo, reflecting improved beta-cell function (1.96 vs. 1.68 pmol/mL; P = .006).
C-peptide levels declined over time in the placebo group but stabilized in those receiving teplizumab (P = .0015).
“The mid-range time from randomization to stage 3 type 1 diabetes diagnosis was 50 months for the patients who received Tzield and 25 months for those who received a placebo. This represents a statistically significant delay in the development of stage 3 type 1 diabetes,” according to the FDA statement.
The most common side effects of Tzield include lymphopenia (73% teplizumab vs. 6% placebo), rash (36% vs. 0%), leukopenia (221% vs. 0%), and headache (11% vs. 6%). Label warnings and precautions include monitoring for cytokine release syndrome, risk for serious infections, and avoidance of live, inactivated, and mRNA vaccines.
This approval is likely to accelerate discussion about universal autoantibody screening. Currently, most individuals identified as having preclinical type 1 diabetes are first-degree relatives of people with type 1 diabetes identified through the federally funded TrialNet program. In December 2020, the type 1 diabetes research and advocacy organization JDRF began offering a $55 home blood test to screen for the antibodies, and other screening programs have been launched in the United States and Europe.
Previous studies have examined cost-effectiveness of universal screening in children and the optimal ages that such screening should take place.
In October, Provention Bio announced a co-promotion agreement with Sanofi for the U.S. launch of Tzield for delay in onset of clinical T1D in at-risk individuals. Provention Bio offers financial assistance options (e.g., copay assistance) to eligible patients for out-of-pocket costs.
A version of this article first appeared on Medscape.com.
“Today’s approval of a first-in-class therapy adds an important new treatment option for certain at-risk patients,” said John Sharretts, MD, director of the Division of Diabetes, Lipid Disorders, and Obesity in the FDA’s Center for Drug Evaluation and Research. “The drug’s potential to delay clinical diagnosis of type 1 diabetes may provide patients with months to years without the burdens of disease.”
The agent, which interferes with T-cell-mediated autoimmune destruction of pancreatic beta cells, is the first disease-modifying therapy for impeding progression of type 1 diabetes. It is administered by intravenous infusion once daily for 14 consecutive days.
The specific indication is “to delay the onset of stage 3 type 1 diabetes in adults and pediatric patients 8 years and older who currently have stage 2 type 1 diabetes.” In type 1 diabetes staging, adopted in 2015, stage 1 is defined as the presence of beta cell autoimmunity with two or more islet autoantibodies with normoglycemia, stage 2 is beta-cell autoimmunity with dysglycemia yet asymptomatic, and stage 3 is the onset of symptomatic type 1 diabetes.
Stage 2 type 1 diabetes is associated with a nearly 100% lifetime risk of progression to clinical (stage 3) type 1 diabetes and a 75% risk of developing the condition within 5 years.
The FDA had previously rejected teplizumab for this indication in July 2021, despite a prior endorsement from an advisory panel in May 2021.
Now, with the FDA approval, Provention Bio cofounder and CEO Ashleigh Palmer said in a statement, “This is a historic occasion for the T1D community and a paradigm shifting breakthrough ... It cannot be emphasized enough how precious a delay in the onset of stage 3 T1D can be from a patient and family perspective; more time to live without and, when necessary, prepare for the burdens, complications, and risks associated with stage 3 disease.”
T1D onset delayed by 2 years
In 2019, a pivotal phase 2, randomized, placebo-controlled trial involving 76 at-risk children and adults aged 8 years and older showed that a single 14-day treatment of daily intravenous infusions of teplizumab in 44 patients resulted in a significant median 2-year delay to onset of clinical type 1 diabetes compared with 32 who received placebo.
Those “game changer” data were presented at the American Diabetes Association (ADA) annual meeting in June 2019 and simultaneously published in the New England Journal of Medicine.
Three-year data were presented at the June 2020 ADA meeting and published in March 2021 in Science Translational Medicine, by Emily K. Sims, MD, department of pediatrics, Indiana University, Indianapolis, and colleagues.
At a median follow-up of 923 days, 50% of those randomly assigned to teplizumab remained diabetes free, compared with 22% of those who received placebo infusions (hazard ratio, 0.457; P = .01). The teplizumab group had a greater average C-peptide area under the curve compared with placebo, reflecting improved beta-cell function (1.96 vs. 1.68 pmol/mL; P = .006).
C-peptide levels declined over time in the placebo group but stabilized in those receiving teplizumab (P = .0015).
“The mid-range time from randomization to stage 3 type 1 diabetes diagnosis was 50 months for the patients who received Tzield and 25 months for those who received a placebo. This represents a statistically significant delay in the development of stage 3 type 1 diabetes,” according to the FDA statement.
The most common side effects of Tzield include lymphopenia (73% teplizumab vs. 6% placebo), rash (36% vs. 0%), leukopenia (221% vs. 0%), and headache (11% vs. 6%). Label warnings and precautions include monitoring for cytokine release syndrome, risk for serious infections, and avoidance of live, inactivated, and mRNA vaccines.
This approval is likely to accelerate discussion about universal autoantibody screening. Currently, most individuals identified as having preclinical type 1 diabetes are first-degree relatives of people with type 1 diabetes identified through the federally funded TrialNet program. In December 2020, the type 1 diabetes research and advocacy organization JDRF began offering a $55 home blood test to screen for the antibodies, and other screening programs have been launched in the United States and Europe.
Previous studies have examined cost-effectiveness of universal screening in children and the optimal ages that such screening should take place.
In October, Provention Bio announced a co-promotion agreement with Sanofi for the U.S. launch of Tzield for delay in onset of clinical T1D in at-risk individuals. Provention Bio offers financial assistance options (e.g., copay assistance) to eligible patients for out-of-pocket costs.
A version of this article first appeared on Medscape.com.
FDA grants accelerated approval for new treatment of female cancers
The Food and Drug Administration has granted accelerated approval to mirvetuximab soravtansine (Elahere) for use in pretreated patients with folate receptor (FR) alpha–positive, platinum-resistant epithelial ovarian, fallopian tube, and primary peritoneal cancer. These patients can have received one to three prior lines of treatment.
“ according to labeling.
Mirvetuximab soravtansine is an antibody-drug conjugate (ADC) with an antibody directed against FR alpha that is linked to a microtubule inhibitor conjugate.
This product is a first-in-class ADC directed against FR alpha, a cell-surface protein highly expressed in ovarian cancer, and is the first FDA-approved ADC for platinum-resistant disease, said the manufacturer, ImmunoGen.
Patients are selected for treatment with this drug using a diagnostic test that the FDA approved along with the agent: the VENTANA FOLR1 (FOLR-2.1) RxDx Assay.
FR alpha–positive platinum-resistant ovarian cancer is characterized by limited treatment options and poor outcomes, commented Ursula Matulonis, MD, chief of the division of gynecologic oncology at the Dana-Farber Cancer Institute, Boston, and co–principal investigator of the SORAYA trial that led to the approval. In a company press release, she said results from this trial show that mirvetuximab soravtansine has “impressive antitumor activity, durability of response, and overall tolerability ... [which] demonstrate the benefit of this new therapeutic option.”
The SORAYA trial (also known as Study 0417 [NCT04296890]) was a single-arm trial of 106 patients with FR alpha–positive, platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer.
These patients were identified as FR alpha positive by using the assay. They were permitted to receive up to three prior lines of systemic therapy, and all patients were required to have received bevacizumab.
All patients received mirvetuximab soravtansine-gynx 6 mg/kg (based on adjusted ideal body weight) as an intravenous infusion every 3 weeks until disease progression or unacceptable toxicity.
The approval was based on an investigator-assessed overall response rate of 31.7%, which included five complete responses, and a median duration of response of 6.9 months.
Safety was evaluated in a pooled analysis from three studies among a total of 464 patients with FR alpha–positive, platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer who received at least one dose of the drug.
The most common adverse events, occurring in 20% or more of study participants, were vision impairment, fatigue, increased AST level, nausea, increased alanine aminotransferase level, keratopathy, abdominal pain, decreased lymphocytes, peripheral neuropathy, diarrhea, decreased albumin, constipation, increased alkaline phosphatase level, dry eye, decreased magnesium level, decreased leukocyte count, decreased neutrophil count, and decreased hemoglobin level.
Potential participants were excluded if they had corneal disorders, ocular conditions requiring ongoing treatment, peripheral neuropathy above grade 1, or noninfectious interstitial lung disease.
The product labeling contains a boxed warning of ocular toxicity. Full prescribing information is available.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has granted accelerated approval to mirvetuximab soravtansine (Elahere) for use in pretreated patients with folate receptor (FR) alpha–positive, platinum-resistant epithelial ovarian, fallopian tube, and primary peritoneal cancer. These patients can have received one to three prior lines of treatment.
“ according to labeling.
Mirvetuximab soravtansine is an antibody-drug conjugate (ADC) with an antibody directed against FR alpha that is linked to a microtubule inhibitor conjugate.
This product is a first-in-class ADC directed against FR alpha, a cell-surface protein highly expressed in ovarian cancer, and is the first FDA-approved ADC for platinum-resistant disease, said the manufacturer, ImmunoGen.
Patients are selected for treatment with this drug using a diagnostic test that the FDA approved along with the agent: the VENTANA FOLR1 (FOLR-2.1) RxDx Assay.
FR alpha–positive platinum-resistant ovarian cancer is characterized by limited treatment options and poor outcomes, commented Ursula Matulonis, MD, chief of the division of gynecologic oncology at the Dana-Farber Cancer Institute, Boston, and co–principal investigator of the SORAYA trial that led to the approval. In a company press release, she said results from this trial show that mirvetuximab soravtansine has “impressive antitumor activity, durability of response, and overall tolerability ... [which] demonstrate the benefit of this new therapeutic option.”
The SORAYA trial (also known as Study 0417 [NCT04296890]) was a single-arm trial of 106 patients with FR alpha–positive, platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer.
These patients were identified as FR alpha positive by using the assay. They were permitted to receive up to three prior lines of systemic therapy, and all patients were required to have received bevacizumab.
All patients received mirvetuximab soravtansine-gynx 6 mg/kg (based on adjusted ideal body weight) as an intravenous infusion every 3 weeks until disease progression or unacceptable toxicity.
The approval was based on an investigator-assessed overall response rate of 31.7%, which included five complete responses, and a median duration of response of 6.9 months.
Safety was evaluated in a pooled analysis from three studies among a total of 464 patients with FR alpha–positive, platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer who received at least one dose of the drug.
The most common adverse events, occurring in 20% or more of study participants, were vision impairment, fatigue, increased AST level, nausea, increased alanine aminotransferase level, keratopathy, abdominal pain, decreased lymphocytes, peripheral neuropathy, diarrhea, decreased albumin, constipation, increased alkaline phosphatase level, dry eye, decreased magnesium level, decreased leukocyte count, decreased neutrophil count, and decreased hemoglobin level.
Potential participants were excluded if they had corneal disorders, ocular conditions requiring ongoing treatment, peripheral neuropathy above grade 1, or noninfectious interstitial lung disease.
The product labeling contains a boxed warning of ocular toxicity. Full prescribing information is available.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has granted accelerated approval to mirvetuximab soravtansine (Elahere) for use in pretreated patients with folate receptor (FR) alpha–positive, platinum-resistant epithelial ovarian, fallopian tube, and primary peritoneal cancer. These patients can have received one to three prior lines of treatment.
“ according to labeling.
Mirvetuximab soravtansine is an antibody-drug conjugate (ADC) with an antibody directed against FR alpha that is linked to a microtubule inhibitor conjugate.
This product is a first-in-class ADC directed against FR alpha, a cell-surface protein highly expressed in ovarian cancer, and is the first FDA-approved ADC for platinum-resistant disease, said the manufacturer, ImmunoGen.
Patients are selected for treatment with this drug using a diagnostic test that the FDA approved along with the agent: the VENTANA FOLR1 (FOLR-2.1) RxDx Assay.
FR alpha–positive platinum-resistant ovarian cancer is characterized by limited treatment options and poor outcomes, commented Ursula Matulonis, MD, chief of the division of gynecologic oncology at the Dana-Farber Cancer Institute, Boston, and co–principal investigator of the SORAYA trial that led to the approval. In a company press release, she said results from this trial show that mirvetuximab soravtansine has “impressive antitumor activity, durability of response, and overall tolerability ... [which] demonstrate the benefit of this new therapeutic option.”
The SORAYA trial (also known as Study 0417 [NCT04296890]) was a single-arm trial of 106 patients with FR alpha–positive, platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer.
These patients were identified as FR alpha positive by using the assay. They were permitted to receive up to three prior lines of systemic therapy, and all patients were required to have received bevacizumab.
All patients received mirvetuximab soravtansine-gynx 6 mg/kg (based on adjusted ideal body weight) as an intravenous infusion every 3 weeks until disease progression or unacceptable toxicity.
The approval was based on an investigator-assessed overall response rate of 31.7%, which included five complete responses, and a median duration of response of 6.9 months.
Safety was evaluated in a pooled analysis from three studies among a total of 464 patients with FR alpha–positive, platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer who received at least one dose of the drug.
The most common adverse events, occurring in 20% or more of study participants, were vision impairment, fatigue, increased AST level, nausea, increased alanine aminotransferase level, keratopathy, abdominal pain, decreased lymphocytes, peripheral neuropathy, diarrhea, decreased albumin, constipation, increased alkaline phosphatase level, dry eye, decreased magnesium level, decreased leukocyte count, decreased neutrophil count, and decreased hemoglobin level.
Potential participants were excluded if they had corneal disorders, ocular conditions requiring ongoing treatment, peripheral neuropathy above grade 1, or noninfectious interstitial lung disease.
The product labeling contains a boxed warning of ocular toxicity. Full prescribing information is available.
A version of this article first appeared on Medscape.com.