User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
HPV infection during pregnancy ups risk of premature birth
Persistent human papillomavirus (HPV) 16 and HPV 18 during a pregnancy may be associated with an increased risk of premature birth.
Findings published online in JAMA Network Open found that 15.9% of individuals who had a persistent HPV 16 or 18 infection during the first and third trimesters of their pregnancy gave birth prematurely, compared with 5.6% of those who did not have an HPV infection at all.
The findings prompted the question of “the pathophysiology of HPV in pregnancy and how the virus is affecting the placenta,” said Lisette Davidson Tanner, MD, MPH, FACOG, who was not involved in the study.
Researchers said the findings are the first to show the association between preterm birth and HPV, which is an incurable virus that most sexually active individuals will get at some point in their lives, according to the Centers for Disease Control and Prevention.
“The results of this study are very important in helping us understand the burden caused by HPV in pregnancy,” study author Helen Trottier, MSc, PhD, researcher at the Centre Hospitalier Universitaire Sainte-Justine, said in an interview. “We may have just pinpointed an important cause of preterm birth that has so far been unexplained.”
Dr. Trottier and colleagues examined data from 1,052 pregnant women from three university-affiliated health care centers in Montreal between Nov. 8, 2010, and Oct. 16, 2016.
Only 5.6% of those who did not have an HPV infection had a premature birth, compared with 6.9% of those who tested positive for any HPV infection in the first trimester.
When looking at the first trimester, researchers found 12% of those diagnosed with HPV 16 and 18 had a preterm birth, compared to 4.9% of those who had a high-risk HPV infection other than HPV 16/18. When looking at the third trimester, researchers found that 15.9% of those with HPV 16/18 had an increased risk of giving birth prematurely, compared to those who had other high-risk HPV infections.
When researchers looked at the persistence of these HPV infections, they found that most HPV infections detected in the first trimester persist to the third trimester. The findings also revealed that persistent vaginal HPV 16/18 detection was significantly associated with all preterm births and spontaneous preterm births. This association was also found among those who had HPV infections detected in their placentas.
Meanwhile, 5.8% of those who had an HPV infection only during the first trimester experienced a preterm birth.
The researchers also found that HPV infections were frequent in pregnancy even among populations “considered to be at low risk based on sociodemographic and sexual history characteristics,” they wrote. Dr. Trottier said she hopes the findings will strengthen support for HPV vaccination.
Dr. Trottier’s study adds to a growing body of research regarding the adverse effects of HPV, according to Dr. Tanner, assistant professor of gynecology and obstetrics at Emory University, Atlanta. “It is already well known that HPV is associated with a number of anogenital and oropharyngeal cancers,” Dr. Tanner said in an interview. “The potential association with preterm birth only adds weight to the recommendations to screen for and prevent HPV infection.”
HPV 16 and 18 are high-risk types that cause about 70% of cervical cancers and precancerous cervical lesions, according to the World Health Organization. However, there are three HPV vaccines – 9-valent HPV vaccine (Gardasil), quadrivalent HPV vaccine (Gardasil®, 4vHPV), and bivalent HPV vaccine (Cervarix) – that help protect against HPV 16/18.
The findings strengthen the benefits of HPV vaccination, Dr. Trottier explained. “There is no cure when the HPV infection is present,” Dr. Trottier said. “If the link [between preterm birth and HPV infections] is indeed causal, we can expect a greater risk of preterm delivery in these women. The effective tool we have is the HPV vaccination, but it should ideally be received before the start of sexual activity in order to prevent future infections that could occur in women.”
The American College of Obstetricians and Gynecologists recommends HPV vaccination for girls and women between the ages of 11 and 26 years old. However, Dr. Tanner said, women aged 27-45 who were previously unvaccinated may still receive benefit from the vaccine.
“Despite the known efficacy of the vaccine, only 50% of patients are up to date with their HPV vaccination,” Dr. Tanner explained. “This study further highlights the need to educate and encourage patients to be vaccinated.”
The researchers said future studies should investigate the association of HPV vaccination and vaccination programs with the risk of preterm birth.
The experts disclosed no conflicts of interest.
Persistent human papillomavirus (HPV) 16 and HPV 18 during a pregnancy may be associated with an increased risk of premature birth.
Findings published online in JAMA Network Open found that 15.9% of individuals who had a persistent HPV 16 or 18 infection during the first and third trimesters of their pregnancy gave birth prematurely, compared with 5.6% of those who did not have an HPV infection at all.
The findings prompted the question of “the pathophysiology of HPV in pregnancy and how the virus is affecting the placenta,” said Lisette Davidson Tanner, MD, MPH, FACOG, who was not involved in the study.
Researchers said the findings are the first to show the association between preterm birth and HPV, which is an incurable virus that most sexually active individuals will get at some point in their lives, according to the Centers for Disease Control and Prevention.
“The results of this study are very important in helping us understand the burden caused by HPV in pregnancy,” study author Helen Trottier, MSc, PhD, researcher at the Centre Hospitalier Universitaire Sainte-Justine, said in an interview. “We may have just pinpointed an important cause of preterm birth that has so far been unexplained.”
Dr. Trottier and colleagues examined data from 1,052 pregnant women from three university-affiliated health care centers in Montreal between Nov. 8, 2010, and Oct. 16, 2016.
Only 5.6% of those who did not have an HPV infection had a premature birth, compared with 6.9% of those who tested positive for any HPV infection in the first trimester.
When looking at the first trimester, researchers found 12% of those diagnosed with HPV 16 and 18 had a preterm birth, compared to 4.9% of those who had a high-risk HPV infection other than HPV 16/18. When looking at the third trimester, researchers found that 15.9% of those with HPV 16/18 had an increased risk of giving birth prematurely, compared to those who had other high-risk HPV infections.
When researchers looked at the persistence of these HPV infections, they found that most HPV infections detected in the first trimester persist to the third trimester. The findings also revealed that persistent vaginal HPV 16/18 detection was significantly associated with all preterm births and spontaneous preterm births. This association was also found among those who had HPV infections detected in their placentas.
Meanwhile, 5.8% of those who had an HPV infection only during the first trimester experienced a preterm birth.
The researchers also found that HPV infections were frequent in pregnancy even among populations “considered to be at low risk based on sociodemographic and sexual history characteristics,” they wrote. Dr. Trottier said she hopes the findings will strengthen support for HPV vaccination.
Dr. Trottier’s study adds to a growing body of research regarding the adverse effects of HPV, according to Dr. Tanner, assistant professor of gynecology and obstetrics at Emory University, Atlanta. “It is already well known that HPV is associated with a number of anogenital and oropharyngeal cancers,” Dr. Tanner said in an interview. “The potential association with preterm birth only adds weight to the recommendations to screen for and prevent HPV infection.”
HPV 16 and 18 are high-risk types that cause about 70% of cervical cancers and precancerous cervical lesions, according to the World Health Organization. However, there are three HPV vaccines – 9-valent HPV vaccine (Gardasil), quadrivalent HPV vaccine (Gardasil®, 4vHPV), and bivalent HPV vaccine (Cervarix) – that help protect against HPV 16/18.
The findings strengthen the benefits of HPV vaccination, Dr. Trottier explained. “There is no cure when the HPV infection is present,” Dr. Trottier said. “If the link [between preterm birth and HPV infections] is indeed causal, we can expect a greater risk of preterm delivery in these women. The effective tool we have is the HPV vaccination, but it should ideally be received before the start of sexual activity in order to prevent future infections that could occur in women.”
The American College of Obstetricians and Gynecologists recommends HPV vaccination for girls and women between the ages of 11 and 26 years old. However, Dr. Tanner said, women aged 27-45 who were previously unvaccinated may still receive benefit from the vaccine.
“Despite the known efficacy of the vaccine, only 50% of patients are up to date with their HPV vaccination,” Dr. Tanner explained. “This study further highlights the need to educate and encourage patients to be vaccinated.”
The researchers said future studies should investigate the association of HPV vaccination and vaccination programs with the risk of preterm birth.
The experts disclosed no conflicts of interest.
Persistent human papillomavirus (HPV) 16 and HPV 18 during a pregnancy may be associated with an increased risk of premature birth.
Findings published online in JAMA Network Open found that 15.9% of individuals who had a persistent HPV 16 or 18 infection during the first and third trimesters of their pregnancy gave birth prematurely, compared with 5.6% of those who did not have an HPV infection at all.
The findings prompted the question of “the pathophysiology of HPV in pregnancy and how the virus is affecting the placenta,” said Lisette Davidson Tanner, MD, MPH, FACOG, who was not involved in the study.
Researchers said the findings are the first to show the association between preterm birth and HPV, which is an incurable virus that most sexually active individuals will get at some point in their lives, according to the Centers for Disease Control and Prevention.
“The results of this study are very important in helping us understand the burden caused by HPV in pregnancy,” study author Helen Trottier, MSc, PhD, researcher at the Centre Hospitalier Universitaire Sainte-Justine, said in an interview. “We may have just pinpointed an important cause of preterm birth that has so far been unexplained.”
Dr. Trottier and colleagues examined data from 1,052 pregnant women from three university-affiliated health care centers in Montreal between Nov. 8, 2010, and Oct. 16, 2016.
Only 5.6% of those who did not have an HPV infection had a premature birth, compared with 6.9% of those who tested positive for any HPV infection in the first trimester.
When looking at the first trimester, researchers found 12% of those diagnosed with HPV 16 and 18 had a preterm birth, compared to 4.9% of those who had a high-risk HPV infection other than HPV 16/18. When looking at the third trimester, researchers found that 15.9% of those with HPV 16/18 had an increased risk of giving birth prematurely, compared to those who had other high-risk HPV infections.
When researchers looked at the persistence of these HPV infections, they found that most HPV infections detected in the first trimester persist to the third trimester. The findings also revealed that persistent vaginal HPV 16/18 detection was significantly associated with all preterm births and spontaneous preterm births. This association was also found among those who had HPV infections detected in their placentas.
Meanwhile, 5.8% of those who had an HPV infection only during the first trimester experienced a preterm birth.
The researchers also found that HPV infections were frequent in pregnancy even among populations “considered to be at low risk based on sociodemographic and sexual history characteristics,” they wrote. Dr. Trottier said she hopes the findings will strengthen support for HPV vaccination.
Dr. Trottier’s study adds to a growing body of research regarding the adverse effects of HPV, according to Dr. Tanner, assistant professor of gynecology and obstetrics at Emory University, Atlanta. “It is already well known that HPV is associated with a number of anogenital and oropharyngeal cancers,” Dr. Tanner said in an interview. “The potential association with preterm birth only adds weight to the recommendations to screen for and prevent HPV infection.”
HPV 16 and 18 are high-risk types that cause about 70% of cervical cancers and precancerous cervical lesions, according to the World Health Organization. However, there are three HPV vaccines – 9-valent HPV vaccine (Gardasil), quadrivalent HPV vaccine (Gardasil®, 4vHPV), and bivalent HPV vaccine (Cervarix) – that help protect against HPV 16/18.
The findings strengthen the benefits of HPV vaccination, Dr. Trottier explained. “There is no cure when the HPV infection is present,” Dr. Trottier said. “If the link [between preterm birth and HPV infections] is indeed causal, we can expect a greater risk of preterm delivery in these women. The effective tool we have is the HPV vaccination, but it should ideally be received before the start of sexual activity in order to prevent future infections that could occur in women.”
The American College of Obstetricians and Gynecologists recommends HPV vaccination for girls and women between the ages of 11 and 26 years old. However, Dr. Tanner said, women aged 27-45 who were previously unvaccinated may still receive benefit from the vaccine.
“Despite the known efficacy of the vaccine, only 50% of patients are up to date with their HPV vaccination,” Dr. Tanner explained. “This study further highlights the need to educate and encourage patients to be vaccinated.”
The researchers said future studies should investigate the association of HPV vaccination and vaccination programs with the risk of preterm birth.
The experts disclosed no conflicts of interest.
FROM JAMA NETWORK OPEN
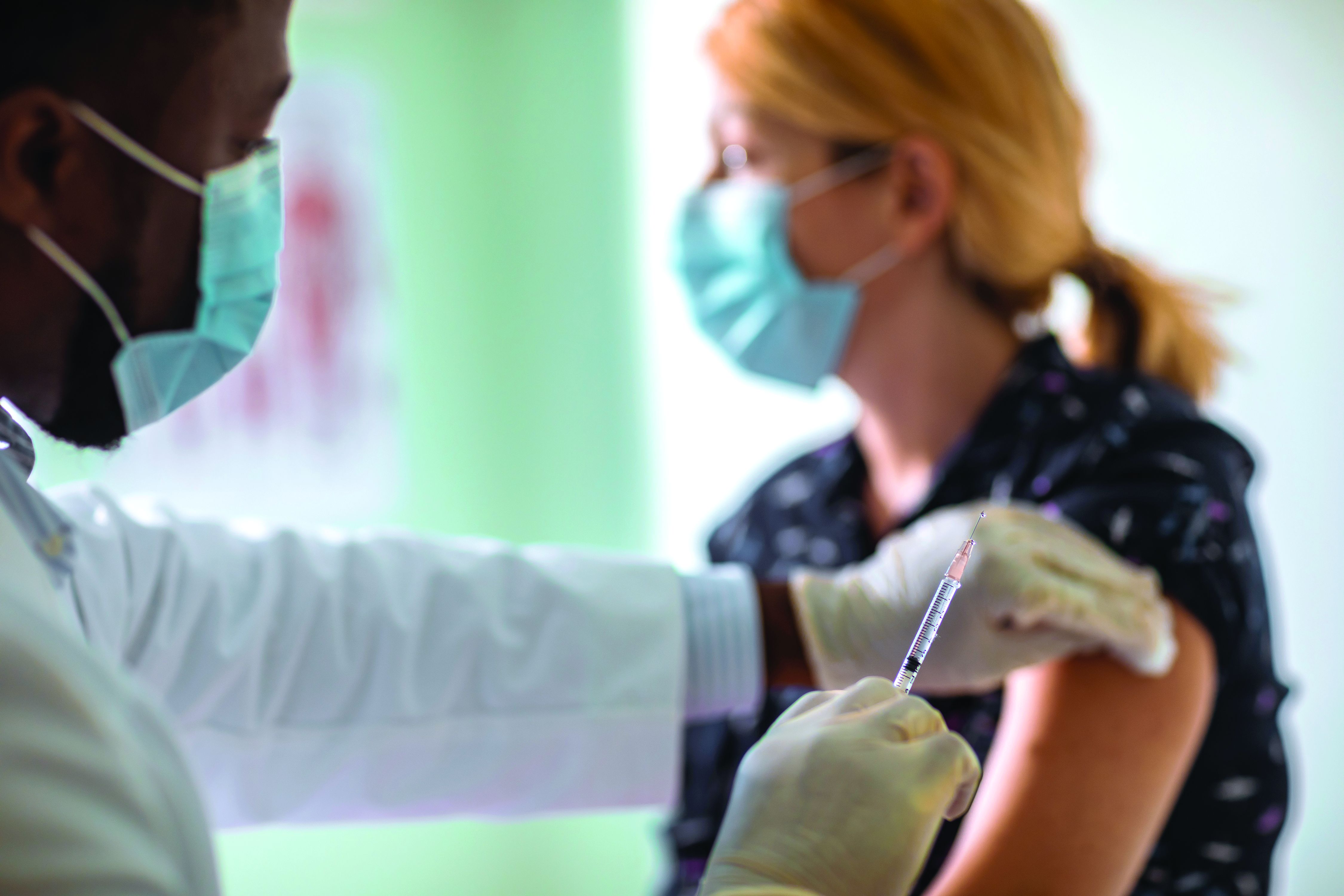
New Moderna vaccine data ‘support’ booster shot after 8 months
Moderna has released new data that it said support the argument for COVID-19 booster shots – specifically showing that people who received a first shot of their mRNA vaccine a median of 13 months ago are more likely to experience a breakthrough infection compared to individuals who received a first shot a median of 8 months ago.
The findings come from the ongoing phase 3 COVE clinical trial, the results of which the Food and Drug Administration considered in granting emergency use authorization for the vaccine. In the initial stage of the trial, people were randomly assigned to receive the company’s mRNA vaccine or placebo.
according to the analysis of the open-label extension of the study during which placebo participants could cross over and get immunized as well.
The updated COVE trial data show that 88 breakthrough cases of COVID-19 occurred among 11,431 participants vaccinated between December 2020 and March 2021 (49.0 cases per 1,000 person-years).
In contrast, there were 162 breakthrough cases among 14,746 people vaccinated between July and October 2020 (77.1 cases per 1,000 person-years).
The breakthrough infections include 19 severe cases. Although not statically different, there was a trend toward fewer severe cases among the more recently vaccinated, at a rate of 3.3 per 1,000 person-years, compared with 6.2 per 1,000 person-years in the group vaccinated in 2020
The findings were posted as a preprint to the medRxiv server and have not yet been peer reviewed.
“The increased risk of breakthrough infections in COVE study participants who were vaccinated last year compared to more recently illustrates the impact of waning immunity and supports the need for a booster to maintain high levels of protection,” Moderna CEO Stéphane Bancel said in a company statement.
An FDA advisory committee is meeting Sept. 17 to look at the available evidence on boosters to help the agency decide whether the additional shots are warranted.
There is still a lot of debate in the medical community about the need for boosters. U.S. physicians and nurses are divided about the need for them and about how the country should prioritize its vaccine supplies, according to a Medscape poll of more than 1,700 clinicians that collected responses from Aug. 25 to Sept. 6, 2020.
The research was funded by Moderna, and also supported by the Office of the Assistant Secretary for Preparedness and Response, Biomedical Advanced Research and Development Authority, and by the National Institute of Allergy and Infectious Diseases.
A version of this article first appeared on Medscape.com.
Moderna has released new data that it said support the argument for COVID-19 booster shots – specifically showing that people who received a first shot of their mRNA vaccine a median of 13 months ago are more likely to experience a breakthrough infection compared to individuals who received a first shot a median of 8 months ago.
The findings come from the ongoing phase 3 COVE clinical trial, the results of which the Food and Drug Administration considered in granting emergency use authorization for the vaccine. In the initial stage of the trial, people were randomly assigned to receive the company’s mRNA vaccine or placebo.
according to the analysis of the open-label extension of the study during which placebo participants could cross over and get immunized as well.
The updated COVE trial data show that 88 breakthrough cases of COVID-19 occurred among 11,431 participants vaccinated between December 2020 and March 2021 (49.0 cases per 1,000 person-years).
In contrast, there were 162 breakthrough cases among 14,746 people vaccinated between July and October 2020 (77.1 cases per 1,000 person-years).
The breakthrough infections include 19 severe cases. Although not statically different, there was a trend toward fewer severe cases among the more recently vaccinated, at a rate of 3.3 per 1,000 person-years, compared with 6.2 per 1,000 person-years in the group vaccinated in 2020
The findings were posted as a preprint to the medRxiv server and have not yet been peer reviewed.
“The increased risk of breakthrough infections in COVE study participants who were vaccinated last year compared to more recently illustrates the impact of waning immunity and supports the need for a booster to maintain high levels of protection,” Moderna CEO Stéphane Bancel said in a company statement.
An FDA advisory committee is meeting Sept. 17 to look at the available evidence on boosters to help the agency decide whether the additional shots are warranted.
There is still a lot of debate in the medical community about the need for boosters. U.S. physicians and nurses are divided about the need for them and about how the country should prioritize its vaccine supplies, according to a Medscape poll of more than 1,700 clinicians that collected responses from Aug. 25 to Sept. 6, 2020.
The research was funded by Moderna, and also supported by the Office of the Assistant Secretary for Preparedness and Response, Biomedical Advanced Research and Development Authority, and by the National Institute of Allergy and Infectious Diseases.
A version of this article first appeared on Medscape.com.
Moderna has released new data that it said support the argument for COVID-19 booster shots – specifically showing that people who received a first shot of their mRNA vaccine a median of 13 months ago are more likely to experience a breakthrough infection compared to individuals who received a first shot a median of 8 months ago.
The findings come from the ongoing phase 3 COVE clinical trial, the results of which the Food and Drug Administration considered in granting emergency use authorization for the vaccine. In the initial stage of the trial, people were randomly assigned to receive the company’s mRNA vaccine or placebo.
according to the analysis of the open-label extension of the study during which placebo participants could cross over and get immunized as well.
The updated COVE trial data show that 88 breakthrough cases of COVID-19 occurred among 11,431 participants vaccinated between December 2020 and March 2021 (49.0 cases per 1,000 person-years).
In contrast, there were 162 breakthrough cases among 14,746 people vaccinated between July and October 2020 (77.1 cases per 1,000 person-years).
The breakthrough infections include 19 severe cases. Although not statically different, there was a trend toward fewer severe cases among the more recently vaccinated, at a rate of 3.3 per 1,000 person-years, compared with 6.2 per 1,000 person-years in the group vaccinated in 2020
The findings were posted as a preprint to the medRxiv server and have not yet been peer reviewed.
“The increased risk of breakthrough infections in COVE study participants who were vaccinated last year compared to more recently illustrates the impact of waning immunity and supports the need for a booster to maintain high levels of protection,” Moderna CEO Stéphane Bancel said in a company statement.
An FDA advisory committee is meeting Sept. 17 to look at the available evidence on boosters to help the agency decide whether the additional shots are warranted.
There is still a lot of debate in the medical community about the need for boosters. U.S. physicians and nurses are divided about the need for them and about how the country should prioritize its vaccine supplies, according to a Medscape poll of more than 1,700 clinicians that collected responses from Aug. 25 to Sept. 6, 2020.
The research was funded by Moderna, and also supported by the Office of the Assistant Secretary for Preparedness and Response, Biomedical Advanced Research and Development Authority, and by the National Institute of Allergy and Infectious Diseases.
A version of this article first appeared on Medscape.com.
Three ‘bad news’ payment changes coming soon for physicians
Physicians are bracing for upcoming changes in reimbursement that may start within a few months. As doctors gear up for another wave of COVID, payment trends may not be the top priority, but some “uh oh” announcements in the fall of 2021 could have far-reaching implications that could affect your future.
The Centers for Medicare & Medicaid Services issued a proposed rule in the summer covering key aspects of physician payment. Although the rule contained some small bright lights, the most important changes proposed were far from welcome.
Here’s what could be in store:
1. The highly anticipated Medicare Physician Fee Schedule ruling confirmed a sweeping payment cut. The drive to maintain budget neutrality forced the federal agency to reduce Medicare payments, on average, by nearly 4%. Many physicians are outraged at the proposed cut.
2. More bad news for 2022: Sequestration will be back. Sequestration is the mandatory, pesky, negative 2% adjustment on all Medicare payments. It had been put on hold and is set to return at the beginning of 2022.
Essentially, sequestration reduces what Medicare pays its providers for health services, but Medicare beneficiaries bear no responsibility for the cost difference. To prevent further debt, CMS imposes financially on hospitals, physicians, and other health care providers.
The Health Resources and Services Administration has funds remaining to reimburse for all COVID-related testing, treatment, and vaccines provided to uninsured individuals. You can apply and be reimbursed at Medicare rates for these services when COVID is the primary diagnosis (or secondary in the case of pregnancy). Patients need not be American citizens for you to get paid.
3. Down to a nail-biter: The final ruling is expected in early November. The situation smacks of earlier days when physicians clung to a precipice, waiting in anticipation for a legislative body to save them from the dreaded income plunge. Indeed, we are slipping back to the decade-long period when Congress kept coming to the rescue simply to maintain the status quo.
Many anticipate a last-minute Congressional intervention to save the day, particularly in the midst of another COVID spike. The promises of a stable reimbursement system made possible by the Medicare Access and CHIP Reauthorization Act have been far from realized, and there are signs that the payment landscape is in the midst of a fundamental transformation.
Other changes proposed in the 1,747-page ruling include:
Positive:
- More telehealth services will be covered by Medicare, including home visits.
- Tele–mental health services got a big boost; many restrictions were removed so that now the patient’s home is considered a permissible originating site. It also allows for audio-only (no visual required) encounters; the audio-only allowance will extend to opioid use disorder treatment services. Phone treatment is covered.
- Permanent adoption of G2252: The 11- to 20-minute virtual check-in code wasn’t just a one-time payment but will be reimbursed in perpetuity.
- Boosts in reimbursement for chronic care and principal care management codes, which range on the basis of service but indicate a commitment to pay for care coordination.
- Clarification of roles and billing opportunities for split/shared visits, which occur if a physician and advanced practice provider see the same patient on a particular day. Prepare for new coding rules to include a modifier. Previously, the rules for billing were muddled, so transparency helps guide payment opportunities.
- Delay of the appropriate use criteria for advanced imaging for 1 (more) year, a welcome postponement of the ruling that carries a significant administrative burden.
- Physician assistants will be able to bill Medicare directly, and referrals to be made to medical nutrition therapy by a nontreating physician.
- A new approach to patient cost-sharing for colorectal cancer screenings will be phased in. This area has caused problems in the past when the physician identifies a need for additional services (for example, polyp removal by a gastroenterologist during routine colonoscopy).
Not positive:
- Which specialties benefit and which get zapped? The anticipated impact by specialty ranges from hits to interventional radiologists (–9%) and vascular surgeons (–8%), to increases for family practitioners, hand surgeons, endocrinologists, and geriatricians, each estimated to gain a modest 2%. (The exception is portable x-ray supplier, with an estimated increase of 10%.) All other specialties fall in between.
- The proposed conversion factor for 2022 is $33.58, a 3.75% drop from the 2021 conversion factor of $34.89.
The proposed ruling also covered the Quality Payment Program, the overarching program of which the Merit-based Incentive Payment System (MIPS) is the main track for participation. The proposal incorporates additional episode-based cost measures as well as updates to quality indicators and improvement activities.
MIPS penalties. The stakes are higher now, with 9% penalties on the table for nonparticipants. The government offers physicians the ability to officially get out of the program in 2021 because of the COVID-19 pandemic, thereby staving off the steep penalty. The option, which is available through the end of the year, requires a simple application that can be completed on behalf of the entire practice. If you want out, now is the time to find and fill out that application.
Exempt from technology requirements. If the proposal is accepted, small practices – defined by CMS as 15 eligible clinicians or fewer – won’t have to file an annual application to reweight the “promoting interoperability” portion of the program. If acknowledged, small practices will automatically be exempt from the program’s technology section. That’s a big plus, as one of the many chief complaints from small practices is the onus of meeting the technology requirements, which include a security risk analysis, bi-directional health information exchange, public health reporting, and patient access to health information. Meeting the requirements is no small feat. That will only affect future years, so be sure to apply in 2021 if applicable for your practice.
Changes in MIPS. MIPS Value Pathways (MVPs) are anticipated for 2023, with the government releasing details about proposed models for heart disease, rheumatology, joint repair, and more. The MVPs are slated to take over the traditional MIPS by 2027.
The program will shift to 30% of your score coming from the “cost” category, which is based on the government’s analysis of a physician’s claims – and, if attributed, the claims of the patients for whom you care. This area is tricky to manage, but recognize that the costs under scrutiny are the expenses paid by Medicare on behalf of its patients.
In essence, Medicare is measuring the cost of your patients as compared with your colleagues’ costs (in the form of specialty-based benchmarks). Therefore, if you’re referring, or ordering, a more costly set of diagnostic tests, assessments, or interventions than your peers, you’ll be dinged.
However, physicians are more likely this year to flat out reject participation in the federal payment program. Payouts have been paltry and dismal to date, and the buzz is that physicians just don’t consider it worth the effort. Of course, clearing the threshold (which is proposed at 70 points next year) is a must to avoid the penalty, but don’t go crazy to get a perfect score as it won’t count for much. 2022 is the final year that there are any monies for exceptional performance.
Considering that the payouts for exceptional performance have been less than 2% for several years now, it’s hard to justify dedicating resources to achieve perfection. Experts believe that even exceptional performance will only be worth pennies in bonus payments.
The fear of the stick, therefore, may be the only motivation. And that is subjective, as physicians weigh the effort required versus just taking the hit on the penalty. But the penalty is substantial, and so even without the incentive, it’s important to participate at least at the threshold.
Fewer cost-sharing waivers. While the federal government’s payment policies have a major impact on reimbursement, other forces may have broader implications. Commercial payers have rolled back cost-sharing waivers, bringing to light the significant financial responsibility that patients have for their health care in the form of deductibles, coinsurance, and so forth.
More than a third of Americans had trouble paying their health care bills before the pandemic; as patients catch up with services that were postponed or delayed because of the pandemic, this may expose challenges for you. Patients with unpaid bills translate into your financial burden.
Virtual-first health plans. Patients may be seeking alternatives to avoid the frustrating cycle of unpaid medical bills. This may be a factor propelling another trend: Lower-cost virtual-first health plans such as Alignment Health have taken hold in the market. As the name implies, insurance coverage features telehealth that extends to in-person services if necessary.
These disruptors may have their hands at least somewhat tied, however. The market may not be able to fully embrace telemedicine until state licensure is addressed. Despite the federal regulatory relaxations, states still control the distribution of medical care through licensure requirements. Many are rolling back their pandemic-based emergency orders and only allowing licensed physicians to see patients in their state, even over telemedicine.
While seemingly frustrating for physicians who want to see patients over state lines, the delays imposed by states may actually have a welcome effect. If licensure migrates to the federal level, there are many implications. For the purposes of this article, the competitive landscape will become incredibly aggressive. You will need to compete with Amazon Care, Walmart, Cigna, and many other well-funded national players that would love nothing more than to launch a campaign to target the entire nation. Investors are eager to capture part of the nearly quarter-trillion-dollar market, with telemedicine at 38 times prepandemic levels and no signs of abating.
Increased competition for insurers. While the proposed drop in Medicare reimbursement is frustrating, keep a pulse on the fact that your patients may soon be lured by vendors like Amazon and others eager to gain access to physician payments. Instead of analyzing Federal Registers in the future, we may be assessing stock prices.
Consider, therefore, how to ensure that your digital front door is at least available, if not wide open, in the meantime. The nature of physician payments is surely changing.
Ms. Woodcock is president of Woodcock & Associates, Atlanta. She has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
Physicians are bracing for upcoming changes in reimbursement that may start within a few months. As doctors gear up for another wave of COVID, payment trends may not be the top priority, but some “uh oh” announcements in the fall of 2021 could have far-reaching implications that could affect your future.
The Centers for Medicare & Medicaid Services issued a proposed rule in the summer covering key aspects of physician payment. Although the rule contained some small bright lights, the most important changes proposed were far from welcome.
Here’s what could be in store:
1. The highly anticipated Medicare Physician Fee Schedule ruling confirmed a sweeping payment cut. The drive to maintain budget neutrality forced the federal agency to reduce Medicare payments, on average, by nearly 4%. Many physicians are outraged at the proposed cut.
2. More bad news for 2022: Sequestration will be back. Sequestration is the mandatory, pesky, negative 2% adjustment on all Medicare payments. It had been put on hold and is set to return at the beginning of 2022.
Essentially, sequestration reduces what Medicare pays its providers for health services, but Medicare beneficiaries bear no responsibility for the cost difference. To prevent further debt, CMS imposes financially on hospitals, physicians, and other health care providers.
The Health Resources and Services Administration has funds remaining to reimburse for all COVID-related testing, treatment, and vaccines provided to uninsured individuals. You can apply and be reimbursed at Medicare rates for these services when COVID is the primary diagnosis (or secondary in the case of pregnancy). Patients need not be American citizens for you to get paid.
3. Down to a nail-biter: The final ruling is expected in early November. The situation smacks of earlier days when physicians clung to a precipice, waiting in anticipation for a legislative body to save them from the dreaded income plunge. Indeed, we are slipping back to the decade-long period when Congress kept coming to the rescue simply to maintain the status quo.
Many anticipate a last-minute Congressional intervention to save the day, particularly in the midst of another COVID spike. The promises of a stable reimbursement system made possible by the Medicare Access and CHIP Reauthorization Act have been far from realized, and there are signs that the payment landscape is in the midst of a fundamental transformation.
Other changes proposed in the 1,747-page ruling include:
Positive:
- More telehealth services will be covered by Medicare, including home visits.
- Tele–mental health services got a big boost; many restrictions were removed so that now the patient’s home is considered a permissible originating site. It also allows for audio-only (no visual required) encounters; the audio-only allowance will extend to opioid use disorder treatment services. Phone treatment is covered.
- Permanent adoption of G2252: The 11- to 20-minute virtual check-in code wasn’t just a one-time payment but will be reimbursed in perpetuity.
- Boosts in reimbursement for chronic care and principal care management codes, which range on the basis of service but indicate a commitment to pay for care coordination.
- Clarification of roles and billing opportunities for split/shared visits, which occur if a physician and advanced practice provider see the same patient on a particular day. Prepare for new coding rules to include a modifier. Previously, the rules for billing were muddled, so transparency helps guide payment opportunities.
- Delay of the appropriate use criteria for advanced imaging for 1 (more) year, a welcome postponement of the ruling that carries a significant administrative burden.
- Physician assistants will be able to bill Medicare directly, and referrals to be made to medical nutrition therapy by a nontreating physician.
- A new approach to patient cost-sharing for colorectal cancer screenings will be phased in. This area has caused problems in the past when the physician identifies a need for additional services (for example, polyp removal by a gastroenterologist during routine colonoscopy).
Not positive:
- Which specialties benefit and which get zapped? The anticipated impact by specialty ranges from hits to interventional radiologists (–9%) and vascular surgeons (–8%), to increases for family practitioners, hand surgeons, endocrinologists, and geriatricians, each estimated to gain a modest 2%. (The exception is portable x-ray supplier, with an estimated increase of 10%.) All other specialties fall in between.
- The proposed conversion factor for 2022 is $33.58, a 3.75% drop from the 2021 conversion factor of $34.89.
The proposed ruling also covered the Quality Payment Program, the overarching program of which the Merit-based Incentive Payment System (MIPS) is the main track for participation. The proposal incorporates additional episode-based cost measures as well as updates to quality indicators and improvement activities.
MIPS penalties. The stakes are higher now, with 9% penalties on the table for nonparticipants. The government offers physicians the ability to officially get out of the program in 2021 because of the COVID-19 pandemic, thereby staving off the steep penalty. The option, which is available through the end of the year, requires a simple application that can be completed on behalf of the entire practice. If you want out, now is the time to find and fill out that application.
Exempt from technology requirements. If the proposal is accepted, small practices – defined by CMS as 15 eligible clinicians or fewer – won’t have to file an annual application to reweight the “promoting interoperability” portion of the program. If acknowledged, small practices will automatically be exempt from the program’s technology section. That’s a big plus, as one of the many chief complaints from small practices is the onus of meeting the technology requirements, which include a security risk analysis, bi-directional health information exchange, public health reporting, and patient access to health information. Meeting the requirements is no small feat. That will only affect future years, so be sure to apply in 2021 if applicable for your practice.
Changes in MIPS. MIPS Value Pathways (MVPs) are anticipated for 2023, with the government releasing details about proposed models for heart disease, rheumatology, joint repair, and more. The MVPs are slated to take over the traditional MIPS by 2027.
The program will shift to 30% of your score coming from the “cost” category, which is based on the government’s analysis of a physician’s claims – and, if attributed, the claims of the patients for whom you care. This area is tricky to manage, but recognize that the costs under scrutiny are the expenses paid by Medicare on behalf of its patients.
In essence, Medicare is measuring the cost of your patients as compared with your colleagues’ costs (in the form of specialty-based benchmarks). Therefore, if you’re referring, or ordering, a more costly set of diagnostic tests, assessments, or interventions than your peers, you’ll be dinged.
However, physicians are more likely this year to flat out reject participation in the federal payment program. Payouts have been paltry and dismal to date, and the buzz is that physicians just don’t consider it worth the effort. Of course, clearing the threshold (which is proposed at 70 points next year) is a must to avoid the penalty, but don’t go crazy to get a perfect score as it won’t count for much. 2022 is the final year that there are any monies for exceptional performance.
Considering that the payouts for exceptional performance have been less than 2% for several years now, it’s hard to justify dedicating resources to achieve perfection. Experts believe that even exceptional performance will only be worth pennies in bonus payments.
The fear of the stick, therefore, may be the only motivation. And that is subjective, as physicians weigh the effort required versus just taking the hit on the penalty. But the penalty is substantial, and so even without the incentive, it’s important to participate at least at the threshold.
Fewer cost-sharing waivers. While the federal government’s payment policies have a major impact on reimbursement, other forces may have broader implications. Commercial payers have rolled back cost-sharing waivers, bringing to light the significant financial responsibility that patients have for their health care in the form of deductibles, coinsurance, and so forth.
More than a third of Americans had trouble paying their health care bills before the pandemic; as patients catch up with services that were postponed or delayed because of the pandemic, this may expose challenges for you. Patients with unpaid bills translate into your financial burden.
Virtual-first health plans. Patients may be seeking alternatives to avoid the frustrating cycle of unpaid medical bills. This may be a factor propelling another trend: Lower-cost virtual-first health plans such as Alignment Health have taken hold in the market. As the name implies, insurance coverage features telehealth that extends to in-person services if necessary.
These disruptors may have their hands at least somewhat tied, however. The market may not be able to fully embrace telemedicine until state licensure is addressed. Despite the federal regulatory relaxations, states still control the distribution of medical care through licensure requirements. Many are rolling back their pandemic-based emergency orders and only allowing licensed physicians to see patients in their state, even over telemedicine.
While seemingly frustrating for physicians who want to see patients over state lines, the delays imposed by states may actually have a welcome effect. If licensure migrates to the federal level, there are many implications. For the purposes of this article, the competitive landscape will become incredibly aggressive. You will need to compete with Amazon Care, Walmart, Cigna, and many other well-funded national players that would love nothing more than to launch a campaign to target the entire nation. Investors are eager to capture part of the nearly quarter-trillion-dollar market, with telemedicine at 38 times prepandemic levels and no signs of abating.
Increased competition for insurers. While the proposed drop in Medicare reimbursement is frustrating, keep a pulse on the fact that your patients may soon be lured by vendors like Amazon and others eager to gain access to physician payments. Instead of analyzing Federal Registers in the future, we may be assessing stock prices.
Consider, therefore, how to ensure that your digital front door is at least available, if not wide open, in the meantime. The nature of physician payments is surely changing.
Ms. Woodcock is president of Woodcock & Associates, Atlanta. She has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
Physicians are bracing for upcoming changes in reimbursement that may start within a few months. As doctors gear up for another wave of COVID, payment trends may not be the top priority, but some “uh oh” announcements in the fall of 2021 could have far-reaching implications that could affect your future.
The Centers for Medicare & Medicaid Services issued a proposed rule in the summer covering key aspects of physician payment. Although the rule contained some small bright lights, the most important changes proposed were far from welcome.
Here’s what could be in store:
1. The highly anticipated Medicare Physician Fee Schedule ruling confirmed a sweeping payment cut. The drive to maintain budget neutrality forced the federal agency to reduce Medicare payments, on average, by nearly 4%. Many physicians are outraged at the proposed cut.
2. More bad news for 2022: Sequestration will be back. Sequestration is the mandatory, pesky, negative 2% adjustment on all Medicare payments. It had been put on hold and is set to return at the beginning of 2022.
Essentially, sequestration reduces what Medicare pays its providers for health services, but Medicare beneficiaries bear no responsibility for the cost difference. To prevent further debt, CMS imposes financially on hospitals, physicians, and other health care providers.
The Health Resources and Services Administration has funds remaining to reimburse for all COVID-related testing, treatment, and vaccines provided to uninsured individuals. You can apply and be reimbursed at Medicare rates for these services when COVID is the primary diagnosis (or secondary in the case of pregnancy). Patients need not be American citizens for you to get paid.
3. Down to a nail-biter: The final ruling is expected in early November. The situation smacks of earlier days when physicians clung to a precipice, waiting in anticipation for a legislative body to save them from the dreaded income plunge. Indeed, we are slipping back to the decade-long period when Congress kept coming to the rescue simply to maintain the status quo.
Many anticipate a last-minute Congressional intervention to save the day, particularly in the midst of another COVID spike. The promises of a stable reimbursement system made possible by the Medicare Access and CHIP Reauthorization Act have been far from realized, and there are signs that the payment landscape is in the midst of a fundamental transformation.
Other changes proposed in the 1,747-page ruling include:
Positive:
- More telehealth services will be covered by Medicare, including home visits.
- Tele–mental health services got a big boost; many restrictions were removed so that now the patient’s home is considered a permissible originating site. It also allows for audio-only (no visual required) encounters; the audio-only allowance will extend to opioid use disorder treatment services. Phone treatment is covered.
- Permanent adoption of G2252: The 11- to 20-minute virtual check-in code wasn’t just a one-time payment but will be reimbursed in perpetuity.
- Boosts in reimbursement for chronic care and principal care management codes, which range on the basis of service but indicate a commitment to pay for care coordination.
- Clarification of roles and billing opportunities for split/shared visits, which occur if a physician and advanced practice provider see the same patient on a particular day. Prepare for new coding rules to include a modifier. Previously, the rules for billing were muddled, so transparency helps guide payment opportunities.
- Delay of the appropriate use criteria for advanced imaging for 1 (more) year, a welcome postponement of the ruling that carries a significant administrative burden.
- Physician assistants will be able to bill Medicare directly, and referrals to be made to medical nutrition therapy by a nontreating physician.
- A new approach to patient cost-sharing for colorectal cancer screenings will be phased in. This area has caused problems in the past when the physician identifies a need for additional services (for example, polyp removal by a gastroenterologist during routine colonoscopy).
Not positive:
- Which specialties benefit and which get zapped? The anticipated impact by specialty ranges from hits to interventional radiologists (–9%) and vascular surgeons (–8%), to increases for family practitioners, hand surgeons, endocrinologists, and geriatricians, each estimated to gain a modest 2%. (The exception is portable x-ray supplier, with an estimated increase of 10%.) All other specialties fall in between.
- The proposed conversion factor for 2022 is $33.58, a 3.75% drop from the 2021 conversion factor of $34.89.
The proposed ruling also covered the Quality Payment Program, the overarching program of which the Merit-based Incentive Payment System (MIPS) is the main track for participation. The proposal incorporates additional episode-based cost measures as well as updates to quality indicators and improvement activities.
MIPS penalties. The stakes are higher now, with 9% penalties on the table for nonparticipants. The government offers physicians the ability to officially get out of the program in 2021 because of the COVID-19 pandemic, thereby staving off the steep penalty. The option, which is available through the end of the year, requires a simple application that can be completed on behalf of the entire practice. If you want out, now is the time to find and fill out that application.
Exempt from technology requirements. If the proposal is accepted, small practices – defined by CMS as 15 eligible clinicians or fewer – won’t have to file an annual application to reweight the “promoting interoperability” portion of the program. If acknowledged, small practices will automatically be exempt from the program’s technology section. That’s a big plus, as one of the many chief complaints from small practices is the onus of meeting the technology requirements, which include a security risk analysis, bi-directional health information exchange, public health reporting, and patient access to health information. Meeting the requirements is no small feat. That will only affect future years, so be sure to apply in 2021 if applicable for your practice.
Changes in MIPS. MIPS Value Pathways (MVPs) are anticipated for 2023, with the government releasing details about proposed models for heart disease, rheumatology, joint repair, and more. The MVPs are slated to take over the traditional MIPS by 2027.
The program will shift to 30% of your score coming from the “cost” category, which is based on the government’s analysis of a physician’s claims – and, if attributed, the claims of the patients for whom you care. This area is tricky to manage, but recognize that the costs under scrutiny are the expenses paid by Medicare on behalf of its patients.
In essence, Medicare is measuring the cost of your patients as compared with your colleagues’ costs (in the form of specialty-based benchmarks). Therefore, if you’re referring, or ordering, a more costly set of diagnostic tests, assessments, or interventions than your peers, you’ll be dinged.
However, physicians are more likely this year to flat out reject participation in the federal payment program. Payouts have been paltry and dismal to date, and the buzz is that physicians just don’t consider it worth the effort. Of course, clearing the threshold (which is proposed at 70 points next year) is a must to avoid the penalty, but don’t go crazy to get a perfect score as it won’t count for much. 2022 is the final year that there are any monies for exceptional performance.
Considering that the payouts for exceptional performance have been less than 2% for several years now, it’s hard to justify dedicating resources to achieve perfection. Experts believe that even exceptional performance will only be worth pennies in bonus payments.
The fear of the stick, therefore, may be the only motivation. And that is subjective, as physicians weigh the effort required versus just taking the hit on the penalty. But the penalty is substantial, and so even without the incentive, it’s important to participate at least at the threshold.
Fewer cost-sharing waivers. While the federal government’s payment policies have a major impact on reimbursement, other forces may have broader implications. Commercial payers have rolled back cost-sharing waivers, bringing to light the significant financial responsibility that patients have for their health care in the form of deductibles, coinsurance, and so forth.
More than a third of Americans had trouble paying their health care bills before the pandemic; as patients catch up with services that were postponed or delayed because of the pandemic, this may expose challenges for you. Patients with unpaid bills translate into your financial burden.
Virtual-first health plans. Patients may be seeking alternatives to avoid the frustrating cycle of unpaid medical bills. This may be a factor propelling another trend: Lower-cost virtual-first health plans such as Alignment Health have taken hold in the market. As the name implies, insurance coverage features telehealth that extends to in-person services if necessary.
These disruptors may have their hands at least somewhat tied, however. The market may not be able to fully embrace telemedicine until state licensure is addressed. Despite the federal regulatory relaxations, states still control the distribution of medical care through licensure requirements. Many are rolling back their pandemic-based emergency orders and only allowing licensed physicians to see patients in their state, even over telemedicine.
While seemingly frustrating for physicians who want to see patients over state lines, the delays imposed by states may actually have a welcome effect. If licensure migrates to the federal level, there are many implications. For the purposes of this article, the competitive landscape will become incredibly aggressive. You will need to compete with Amazon Care, Walmart, Cigna, and many other well-funded national players that would love nothing more than to launch a campaign to target the entire nation. Investors are eager to capture part of the nearly quarter-trillion-dollar market, with telemedicine at 38 times prepandemic levels and no signs of abating.
Increased competition for insurers. While the proposed drop in Medicare reimbursement is frustrating, keep a pulse on the fact that your patients may soon be lured by vendors like Amazon and others eager to gain access to physician payments. Instead of analyzing Federal Registers in the future, we may be assessing stock prices.
Consider, therefore, how to ensure that your digital front door is at least available, if not wide open, in the meantime. The nature of physician payments is surely changing.
Ms. Woodcock is president of Woodcock & Associates, Atlanta. She has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
COVID wars, part nine: The rise of iodine
Onions and iodine and COVID, oh my!
As surely as the sun rises, anti-vaxxers will come up with some wacky and dangerous new idea to prevent COVID. While perhaps nothing will top horse medication, gargling iodine (or spraying it into the nose) is also not a great idea.
Multiple social media posts have extolled the virtues of gargling Betadine (povidone iodine), which is a TOPICAL disinfectant commonly used in EDs and operating rooms. One post cited a paper by a Bangladeshi plastic surgeon who hypothesized on the subject, and if that’s not a peer-reviewed, rigorously researched source, we don’t know what is.
Perhaps unsurprisingly, actual medical experts do not recommend using Betadine to prevent COVID. Ingesting it can cause iodine poisoning and plenty of nasty GI side effects; while Betadine does make a diluted product safe for gargling use (used for the treatment of sore throats), it has not shown any effectiveness against viruses or COVID in particular.
A New York ED doctor summed it up best in the Rolling Stone article when he was told anti-vaxxers were gargling iodine: He offered a choice four-letter expletive, then said, “Of course they are.”
But wait! We’ve got a two-for-one deal on dubious COVID cures this week. Health experts in Myanmar (Burma to all the “Seinfeld” fans) and Thailand have been combating social media posts claiming that onion fumes will cure COVID. All you need to do is slice an onion in half, sniff it for a while, then chew on a second onion, and your COVID will be cured!
In what is surely the most radical understatement of the year, a professor in the department of preventive and social medicine at Chulalongkorn University, Bangkok, said in the AFP article that there is “no solid evidence” to support onion sniffing from “any clinical research.”
We’re just going to assume the expletives that surely followed were kept off the record.
Pro-Trump state governor encourages vaccination
Clearly, the politics of COVID-19 have been working against the science of COVID-19. Politicians can’t, or won’t, agree on what to do about it, and many prominent Republicans have been actively resisting vaccine and mask mandates.
There is at least one Republican governor who has wholeheartedly encouraged vaccination in his pro-Trump state. We’re talking about Gov. Jim Justice of West Virginia, and not for the first time.
The Washington Post has detailed his efforts to promote the COVID vaccine, and we would like to share a couple of examples.
In June he suggested that people who didn’t get vaccinated were “entering the death drawing.” He followed that by saying, “If I knew for certain that there was going to be eight or nine people die by next Tuesday, and I could be one of them if I don’t take the vaccine ... What in the world do you think I would do? I mean, I would run over top of somebody.”
More recently, Gov. Justice took on vaccine conspiracy theories.
“For God’s sakes a livin’, how difficult is this to understand? Why in the world do we have to come up with these crazy ideas – and they’re crazy ideas – that the vaccine’s got something in it and it’s tracing people wherever they go? And the very same people that are saying that are carrying their cellphones around. I mean, come on. Come on.”
Nuff said.
Jet lag may be a gut feeling
After a week-long vacation halfway around the world, it’s time to go back to your usual routine and time zone. But don’t forget about that free souvenir, jet lag. A disrupted circadian rhythm can be a real bummer, but researchers may have found the fix in your belly.
In a study funded by the U.S. Navy, researchers at the University of Colorado, Boulder, looked into how the presence of a prebiotic in one’s diet can have on the disrupted biological clocks. They’re not the same as probiotics, which help you stay regular in another way. Prebiotics work as food to help the good gut bacteria you already have. An earlier study had suggested that prebiotics may have a positive effect on the brain.
To test the theory, the researchers gave one group of rats their regular food while another group received food with two different prebiotics. After manipulating the rats’ light-dark cycle for 8 weeks to give the illusion of traveling to a time zone 12 hours ahead every week, they found that the rats who ate the prebiotics were able to bounce back faster.
The possibility of ingesting something to keep your body clock regular sounds like a dream, but the researchers don’t really advise you to snatch all the supplements you can at your local pharmacy just yet.
“If you know you are going to come into a challenge, you could take a look at some of the prebiotics that are available. Just realize that they are not customized yet, so it might work for you but it won’t work for your neighbor,” said senior author Monika Fleshner.
Until there’s more conclusive research, just be good to your bacteria.
How to make stuff up and influence people
You’ve probably heard that we use only 10% of our brain. It’s right up there with “the Earth is flat” and “an apple a day keeps the doctor away.”
The idea that we use only 10% of our brains can probably be traced back to the early 1900s, suggests Discover magazine, when psychologist William James wrote, “Compared with what we ought to be, we are only half awake. Our fires are damped, our drafts are checked. We are making use of only a small part of our possible mental and physical resources.”
There are many different takes on it, but it is indeed a myth that we use only 10% of our brains. Dale Carnegie, the public speaking teacher, seems to be the one who put the specific number of 10% on James’ idea in his 1936 book, “How to Win Friends and Influence People.”
“We think that people are excited by this pseudo fact because it’s very optimistic,” neuroscientist Sandra Aamodt told Discover. “Wouldn’t we all love to think our brains had some giant pool of untapped potential that we’re not using?”
The reality is, we do use our whole brain. Functional MRI shows that different parts of the brain are used for different things such as language and memories. “Not all at the same time, of course. But every part of the brain has a job to do,” the Discover article explained.
There are many things we don’t know about how the brain works, but at least you know you use more than 10%. After all, a brain just told you so.
Onions and iodine and COVID, oh my!
As surely as the sun rises, anti-vaxxers will come up with some wacky and dangerous new idea to prevent COVID. While perhaps nothing will top horse medication, gargling iodine (or spraying it into the nose) is also not a great idea.
Multiple social media posts have extolled the virtues of gargling Betadine (povidone iodine), which is a TOPICAL disinfectant commonly used in EDs and operating rooms. One post cited a paper by a Bangladeshi plastic surgeon who hypothesized on the subject, and if that’s not a peer-reviewed, rigorously researched source, we don’t know what is.
Perhaps unsurprisingly, actual medical experts do not recommend using Betadine to prevent COVID. Ingesting it can cause iodine poisoning and plenty of nasty GI side effects; while Betadine does make a diluted product safe for gargling use (used for the treatment of sore throats), it has not shown any effectiveness against viruses or COVID in particular.
A New York ED doctor summed it up best in the Rolling Stone article when he was told anti-vaxxers were gargling iodine: He offered a choice four-letter expletive, then said, “Of course they are.”
But wait! We’ve got a two-for-one deal on dubious COVID cures this week. Health experts in Myanmar (Burma to all the “Seinfeld” fans) and Thailand have been combating social media posts claiming that onion fumes will cure COVID. All you need to do is slice an onion in half, sniff it for a while, then chew on a second onion, and your COVID will be cured!
In what is surely the most radical understatement of the year, a professor in the department of preventive and social medicine at Chulalongkorn University, Bangkok, said in the AFP article that there is “no solid evidence” to support onion sniffing from “any clinical research.”
We’re just going to assume the expletives that surely followed were kept off the record.
Pro-Trump state governor encourages vaccination
Clearly, the politics of COVID-19 have been working against the science of COVID-19. Politicians can’t, or won’t, agree on what to do about it, and many prominent Republicans have been actively resisting vaccine and mask mandates.
There is at least one Republican governor who has wholeheartedly encouraged vaccination in his pro-Trump state. We’re talking about Gov. Jim Justice of West Virginia, and not for the first time.
The Washington Post has detailed his efforts to promote the COVID vaccine, and we would like to share a couple of examples.
In June he suggested that people who didn’t get vaccinated were “entering the death drawing.” He followed that by saying, “If I knew for certain that there was going to be eight or nine people die by next Tuesday, and I could be one of them if I don’t take the vaccine ... What in the world do you think I would do? I mean, I would run over top of somebody.”
More recently, Gov. Justice took on vaccine conspiracy theories.
“For God’s sakes a livin’, how difficult is this to understand? Why in the world do we have to come up with these crazy ideas – and they’re crazy ideas – that the vaccine’s got something in it and it’s tracing people wherever they go? And the very same people that are saying that are carrying their cellphones around. I mean, come on. Come on.”
Nuff said.
Jet lag may be a gut feeling
After a week-long vacation halfway around the world, it’s time to go back to your usual routine and time zone. But don’t forget about that free souvenir, jet lag. A disrupted circadian rhythm can be a real bummer, but researchers may have found the fix in your belly.
In a study funded by the U.S. Navy, researchers at the University of Colorado, Boulder, looked into how the presence of a prebiotic in one’s diet can have on the disrupted biological clocks. They’re not the same as probiotics, which help you stay regular in another way. Prebiotics work as food to help the good gut bacteria you already have. An earlier study had suggested that prebiotics may have a positive effect on the brain.
To test the theory, the researchers gave one group of rats their regular food while another group received food with two different prebiotics. After manipulating the rats’ light-dark cycle for 8 weeks to give the illusion of traveling to a time zone 12 hours ahead every week, they found that the rats who ate the prebiotics were able to bounce back faster.
The possibility of ingesting something to keep your body clock regular sounds like a dream, but the researchers don’t really advise you to snatch all the supplements you can at your local pharmacy just yet.
“If you know you are going to come into a challenge, you could take a look at some of the prebiotics that are available. Just realize that they are not customized yet, so it might work for you but it won’t work for your neighbor,” said senior author Monika Fleshner.
Until there’s more conclusive research, just be good to your bacteria.
How to make stuff up and influence people
You’ve probably heard that we use only 10% of our brain. It’s right up there with “the Earth is flat” and “an apple a day keeps the doctor away.”
The idea that we use only 10% of our brains can probably be traced back to the early 1900s, suggests Discover magazine, when psychologist William James wrote, “Compared with what we ought to be, we are only half awake. Our fires are damped, our drafts are checked. We are making use of only a small part of our possible mental and physical resources.”
There are many different takes on it, but it is indeed a myth that we use only 10% of our brains. Dale Carnegie, the public speaking teacher, seems to be the one who put the specific number of 10% on James’ idea in his 1936 book, “How to Win Friends and Influence People.”
“We think that people are excited by this pseudo fact because it’s very optimistic,” neuroscientist Sandra Aamodt told Discover. “Wouldn’t we all love to think our brains had some giant pool of untapped potential that we’re not using?”
The reality is, we do use our whole brain. Functional MRI shows that different parts of the brain are used for different things such as language and memories. “Not all at the same time, of course. But every part of the brain has a job to do,” the Discover article explained.
There are many things we don’t know about how the brain works, but at least you know you use more than 10%. After all, a brain just told you so.
Onions and iodine and COVID, oh my!
As surely as the sun rises, anti-vaxxers will come up with some wacky and dangerous new idea to prevent COVID. While perhaps nothing will top horse medication, gargling iodine (or spraying it into the nose) is also not a great idea.
Multiple social media posts have extolled the virtues of gargling Betadine (povidone iodine), which is a TOPICAL disinfectant commonly used in EDs and operating rooms. One post cited a paper by a Bangladeshi plastic surgeon who hypothesized on the subject, and if that’s not a peer-reviewed, rigorously researched source, we don’t know what is.
Perhaps unsurprisingly, actual medical experts do not recommend using Betadine to prevent COVID. Ingesting it can cause iodine poisoning and plenty of nasty GI side effects; while Betadine does make a diluted product safe for gargling use (used for the treatment of sore throats), it has not shown any effectiveness against viruses or COVID in particular.
A New York ED doctor summed it up best in the Rolling Stone article when he was told anti-vaxxers were gargling iodine: He offered a choice four-letter expletive, then said, “Of course they are.”
But wait! We’ve got a two-for-one deal on dubious COVID cures this week. Health experts in Myanmar (Burma to all the “Seinfeld” fans) and Thailand have been combating social media posts claiming that onion fumes will cure COVID. All you need to do is slice an onion in half, sniff it for a while, then chew on a second onion, and your COVID will be cured!
In what is surely the most radical understatement of the year, a professor in the department of preventive and social medicine at Chulalongkorn University, Bangkok, said in the AFP article that there is “no solid evidence” to support onion sniffing from “any clinical research.”
We’re just going to assume the expletives that surely followed were kept off the record.
Pro-Trump state governor encourages vaccination
Clearly, the politics of COVID-19 have been working against the science of COVID-19. Politicians can’t, or won’t, agree on what to do about it, and many prominent Republicans have been actively resisting vaccine and mask mandates.
There is at least one Republican governor who has wholeheartedly encouraged vaccination in his pro-Trump state. We’re talking about Gov. Jim Justice of West Virginia, and not for the first time.
The Washington Post has detailed his efforts to promote the COVID vaccine, and we would like to share a couple of examples.
In June he suggested that people who didn’t get vaccinated were “entering the death drawing.” He followed that by saying, “If I knew for certain that there was going to be eight or nine people die by next Tuesday, and I could be one of them if I don’t take the vaccine ... What in the world do you think I would do? I mean, I would run over top of somebody.”
More recently, Gov. Justice took on vaccine conspiracy theories.
“For God’s sakes a livin’, how difficult is this to understand? Why in the world do we have to come up with these crazy ideas – and they’re crazy ideas – that the vaccine’s got something in it and it’s tracing people wherever they go? And the very same people that are saying that are carrying their cellphones around. I mean, come on. Come on.”
Nuff said.
Jet lag may be a gut feeling
After a week-long vacation halfway around the world, it’s time to go back to your usual routine and time zone. But don’t forget about that free souvenir, jet lag. A disrupted circadian rhythm can be a real bummer, but researchers may have found the fix in your belly.
In a study funded by the U.S. Navy, researchers at the University of Colorado, Boulder, looked into how the presence of a prebiotic in one’s diet can have on the disrupted biological clocks. They’re not the same as probiotics, which help you stay regular in another way. Prebiotics work as food to help the good gut bacteria you already have. An earlier study had suggested that prebiotics may have a positive effect on the brain.
To test the theory, the researchers gave one group of rats their regular food while another group received food with two different prebiotics. After manipulating the rats’ light-dark cycle for 8 weeks to give the illusion of traveling to a time zone 12 hours ahead every week, they found that the rats who ate the prebiotics were able to bounce back faster.
The possibility of ingesting something to keep your body clock regular sounds like a dream, but the researchers don’t really advise you to snatch all the supplements you can at your local pharmacy just yet.
“If you know you are going to come into a challenge, you could take a look at some of the prebiotics that are available. Just realize that they are not customized yet, so it might work for you but it won’t work for your neighbor,” said senior author Monika Fleshner.
Until there’s more conclusive research, just be good to your bacteria.
How to make stuff up and influence people
You’ve probably heard that we use only 10% of our brain. It’s right up there with “the Earth is flat” and “an apple a day keeps the doctor away.”
The idea that we use only 10% of our brains can probably be traced back to the early 1900s, suggests Discover magazine, when psychologist William James wrote, “Compared with what we ought to be, we are only half awake. Our fires are damped, our drafts are checked. We are making use of only a small part of our possible mental and physical resources.”
There are many different takes on it, but it is indeed a myth that we use only 10% of our brains. Dale Carnegie, the public speaking teacher, seems to be the one who put the specific number of 10% on James’ idea in his 1936 book, “How to Win Friends and Influence People.”
“We think that people are excited by this pseudo fact because it’s very optimistic,” neuroscientist Sandra Aamodt told Discover. “Wouldn’t we all love to think our brains had some giant pool of untapped potential that we’re not using?”
The reality is, we do use our whole brain. Functional MRI shows that different parts of the brain are used for different things such as language and memories. “Not all at the same time, of course. But every part of the brain has a job to do,” the Discover article explained.
There are many things we don’t know about how the brain works, but at least you know you use more than 10%. After all, a brain just told you so.
USPSTF: Continue gonorrhea, chlamydia screening in sexually active young women, teens
The U.S. Preventive Services Task Force (USPSTF) announced on Tuesday that it is standing by its 2014 recommendations that sexually active girls and young women be screened for chlamydia and gonorrhea. But the panel is not ready to provide guidance about screening males even amid an outbreak of gonorrhea infections among men who have sex with men (MSM).
“For men in general, there’s not enough evidence to determine whether screening will reduce the risk of complications or spreading infections to others,” said Marti Kubik, PhD, RN, in an interview. Dr. Kubik is a professor at the George Mason University School of Nursing, Fairfax, Va., and is a member of the task force. “We need further research so we will know how to make those recommendations,” she said.
The screening recommendations for chlamydia and gonorrhea were published Sept. 14 in the Journal of the American Medical Association. The guidance is identical to the panel’s 2014 recommendations. The task force recommends screening for chlamydia and gonorrhea in all sexually active females aged 24 years or younger and in sexually active women aged 25 and older if they are at higher risk because of factors such as new or multiple sex partners.
“We continue to see rising rates of these infections in spite of consistent screening recommendations,” Dr. Kubik said. “In 2019, the CDC recorded nearly 2 million cases of chlamydia and a half million cases of gonorrhea. The big clincher is that chlamydia and gonorrhea can occur without symptoms. It’s critical to screen if we’re going to prevent serious health complications.”
The report notes that chlamydia and gonorrhea may lead to pelvic inflammatory disease in women and to multiple complications in infants born to infected mothers. Men can develop urethritis and epididymitis. Both diseases can boost the risk for HIV infection and transmission.
“We want clinicians to review the new recommendation and feel confident about the evidence base that supports a need for us to be screening young women and older women who are at increased risk,” Dr. Kubik said. She noted that almost two-thirds of chlamydia cases and more than half of gonorrhea cases occur in men and women aged 15-24.
Unlike the CDC, which recommends annual chlamydia and gonorrhea screening in appropriate female patients, the task force provides no guidance on screening frequency. “We didn’t have the evidence base to make a recommendation about how often to screen,” Dr. Kubik said. “But recognizing that these often occur without symptoms, it’s reasonable for clinicians to screen patients whose sexual history reveals new or consistent risk factors.”
Philip A. Chan, MD, an associate professor at Brown University, Providence, R.I., who directs a sexually transmitted disease clinic, told this news organization that he found it frustrating that the task force didn’t make recommendations about screening of MSM. According to a commentary accompanying the new recommendations, the rate of gonorrhea in MSM – 5,166 cases per 100,000, or more than 5% – is at a historic high.
In contrast to the task force, the CDC recommends annual or more frequent testing for gonorrhea and chlamydia plus HIV and syphilis in sexually active MSM.
Dr. Chan noted that the task force’s guidance “tends to be the most evidence-based recommendations that exist. If the evidence isn’t there, they usually don’t make a recommendation.” Still, he said, “I would argue that there’s good evidence that in MSM, the risk for HIV acquisition warrants routine screening.”
Jeanne Marrazzo, MD, MPH, director of the division of infectious diseases at the University of Alabama at Birmingham, also noted the limits of the task force’s insistence on certain kinds of evidence. Dr. Marrazzo, who coauthored a commentary that accompanies the recommendations, said in an interview that the panel’s “reliance on randomized-controlled-trial-level evidence tends to limit its ability to evolve their recommendations in a way that could account for evolving epidemiology or advances in our understanding of pathophysiology of these infections.”
Dr. Chan noted that obstacles exist for patients even when screening recommendations are in place. Although insurers typically cover costs of chlamydia and gonorrhea screening tests, he said, the uninsured may have to pay $100 or more each.
The USPSTF is supported by the U.S. Agency for Healthcare Research and Quality. Dr. Kubik, Dr. Chan, and Dr. Marrazzo report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The U.S. Preventive Services Task Force (USPSTF) announced on Tuesday that it is standing by its 2014 recommendations that sexually active girls and young women be screened for chlamydia and gonorrhea. But the panel is not ready to provide guidance about screening males even amid an outbreak of gonorrhea infections among men who have sex with men (MSM).
“For men in general, there’s not enough evidence to determine whether screening will reduce the risk of complications or spreading infections to others,” said Marti Kubik, PhD, RN, in an interview. Dr. Kubik is a professor at the George Mason University School of Nursing, Fairfax, Va., and is a member of the task force. “We need further research so we will know how to make those recommendations,” she said.
The screening recommendations for chlamydia and gonorrhea were published Sept. 14 in the Journal of the American Medical Association. The guidance is identical to the panel’s 2014 recommendations. The task force recommends screening for chlamydia and gonorrhea in all sexually active females aged 24 years or younger and in sexually active women aged 25 and older if they are at higher risk because of factors such as new or multiple sex partners.
“We continue to see rising rates of these infections in spite of consistent screening recommendations,” Dr. Kubik said. “In 2019, the CDC recorded nearly 2 million cases of chlamydia and a half million cases of gonorrhea. The big clincher is that chlamydia and gonorrhea can occur without symptoms. It’s critical to screen if we’re going to prevent serious health complications.”
The report notes that chlamydia and gonorrhea may lead to pelvic inflammatory disease in women and to multiple complications in infants born to infected mothers. Men can develop urethritis and epididymitis. Both diseases can boost the risk for HIV infection and transmission.
“We want clinicians to review the new recommendation and feel confident about the evidence base that supports a need for us to be screening young women and older women who are at increased risk,” Dr. Kubik said. She noted that almost two-thirds of chlamydia cases and more than half of gonorrhea cases occur in men and women aged 15-24.
Unlike the CDC, which recommends annual chlamydia and gonorrhea screening in appropriate female patients, the task force provides no guidance on screening frequency. “We didn’t have the evidence base to make a recommendation about how often to screen,” Dr. Kubik said. “But recognizing that these often occur without symptoms, it’s reasonable for clinicians to screen patients whose sexual history reveals new or consistent risk factors.”
Philip A. Chan, MD, an associate professor at Brown University, Providence, R.I., who directs a sexually transmitted disease clinic, told this news organization that he found it frustrating that the task force didn’t make recommendations about screening of MSM. According to a commentary accompanying the new recommendations, the rate of gonorrhea in MSM – 5,166 cases per 100,000, or more than 5% – is at a historic high.
In contrast to the task force, the CDC recommends annual or more frequent testing for gonorrhea and chlamydia plus HIV and syphilis in sexually active MSM.
Dr. Chan noted that the task force’s guidance “tends to be the most evidence-based recommendations that exist. If the evidence isn’t there, they usually don’t make a recommendation.” Still, he said, “I would argue that there’s good evidence that in MSM, the risk for HIV acquisition warrants routine screening.”
Jeanne Marrazzo, MD, MPH, director of the division of infectious diseases at the University of Alabama at Birmingham, also noted the limits of the task force’s insistence on certain kinds of evidence. Dr. Marrazzo, who coauthored a commentary that accompanies the recommendations, said in an interview that the panel’s “reliance on randomized-controlled-trial-level evidence tends to limit its ability to evolve their recommendations in a way that could account for evolving epidemiology or advances in our understanding of pathophysiology of these infections.”
Dr. Chan noted that obstacles exist for patients even when screening recommendations are in place. Although insurers typically cover costs of chlamydia and gonorrhea screening tests, he said, the uninsured may have to pay $100 or more each.
The USPSTF is supported by the U.S. Agency for Healthcare Research and Quality. Dr. Kubik, Dr. Chan, and Dr. Marrazzo report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The U.S. Preventive Services Task Force (USPSTF) announced on Tuesday that it is standing by its 2014 recommendations that sexually active girls and young women be screened for chlamydia and gonorrhea. But the panel is not ready to provide guidance about screening males even amid an outbreak of gonorrhea infections among men who have sex with men (MSM).
“For men in general, there’s not enough evidence to determine whether screening will reduce the risk of complications or spreading infections to others,” said Marti Kubik, PhD, RN, in an interview. Dr. Kubik is a professor at the George Mason University School of Nursing, Fairfax, Va., and is a member of the task force. “We need further research so we will know how to make those recommendations,” she said.
The screening recommendations for chlamydia and gonorrhea were published Sept. 14 in the Journal of the American Medical Association. The guidance is identical to the panel’s 2014 recommendations. The task force recommends screening for chlamydia and gonorrhea in all sexually active females aged 24 years or younger and in sexually active women aged 25 and older if they are at higher risk because of factors such as new or multiple sex partners.
“We continue to see rising rates of these infections in spite of consistent screening recommendations,” Dr. Kubik said. “In 2019, the CDC recorded nearly 2 million cases of chlamydia and a half million cases of gonorrhea. The big clincher is that chlamydia and gonorrhea can occur without symptoms. It’s critical to screen if we’re going to prevent serious health complications.”
The report notes that chlamydia and gonorrhea may lead to pelvic inflammatory disease in women and to multiple complications in infants born to infected mothers. Men can develop urethritis and epididymitis. Both diseases can boost the risk for HIV infection and transmission.
“We want clinicians to review the new recommendation and feel confident about the evidence base that supports a need for us to be screening young women and older women who are at increased risk,” Dr. Kubik said. She noted that almost two-thirds of chlamydia cases and more than half of gonorrhea cases occur in men and women aged 15-24.
Unlike the CDC, which recommends annual chlamydia and gonorrhea screening in appropriate female patients, the task force provides no guidance on screening frequency. “We didn’t have the evidence base to make a recommendation about how often to screen,” Dr. Kubik said. “But recognizing that these often occur without symptoms, it’s reasonable for clinicians to screen patients whose sexual history reveals new or consistent risk factors.”
Philip A. Chan, MD, an associate professor at Brown University, Providence, R.I., who directs a sexually transmitted disease clinic, told this news organization that he found it frustrating that the task force didn’t make recommendations about screening of MSM. According to a commentary accompanying the new recommendations, the rate of gonorrhea in MSM – 5,166 cases per 100,000, or more than 5% – is at a historic high.
In contrast to the task force, the CDC recommends annual or more frequent testing for gonorrhea and chlamydia plus HIV and syphilis in sexually active MSM.
Dr. Chan noted that the task force’s guidance “tends to be the most evidence-based recommendations that exist. If the evidence isn’t there, they usually don’t make a recommendation.” Still, he said, “I would argue that there’s good evidence that in MSM, the risk for HIV acquisition warrants routine screening.”
Jeanne Marrazzo, MD, MPH, director of the division of infectious diseases at the University of Alabama at Birmingham, also noted the limits of the task force’s insistence on certain kinds of evidence. Dr. Marrazzo, who coauthored a commentary that accompanies the recommendations, said in an interview that the panel’s “reliance on randomized-controlled-trial-level evidence tends to limit its ability to evolve their recommendations in a way that could account for evolving epidemiology or advances in our understanding of pathophysiology of these infections.”
Dr. Chan noted that obstacles exist for patients even when screening recommendations are in place. Although insurers typically cover costs of chlamydia and gonorrhea screening tests, he said, the uninsured may have to pay $100 or more each.
The USPSTF is supported by the U.S. Agency for Healthcare Research and Quality. Dr. Kubik, Dr. Chan, and Dr. Marrazzo report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Candida auris transmission can be contained in postacute care settings
A new study from Orange County, California, shows how Candida auris, an emerging pathogen, was successfully identified and contained in long-term acute care hospitals (LTACHs) and ventilator-capable skilled-nursing facilities (vSNFs).
Lead author Ellora Karmarkar, MD, MSc, formerly an epidemic intelligence service officer with the Centers for Disease Control and Prevention and currently with the California Department of Public Health, said in an interview that the prospective surveillance of urine cultures for C. auris was prompted by “seeing what was happening in New York, New Jersey, and Illinois [being] pretty alarming for a lot of the health officials in California, [who] know that LTACHs are high-risk facilities because they take care of really sick people. Some of those people are there for a very long time.”
Therefore, the study authors decided to focus their investigations there, rather than in acute care hospitals, which were believed to be at lower risk for C. auris outbreaks.
The Orange County Health Department, working with the California Department of Health and the CDC, asked labs to prospectively identify all Candida isolates in urines from LTACHs between September 2018 and February 2019. Normally, labs do not speciate Candida from nonsterile body sites.
Dan Diekema, MD, an epidemiologist and clinical microbiologist at the University of Iowa, Iowa City, who was not involved in the study, told this news organization, “Acute care hospitals really ought to be moving toward doing species identification of Candida from nonsterile sites if they really want to have a better chance of detecting this early.”
The OCHD also screened LTACH and vSNF patients with composite cultures from the axilla-groin or nasal swabs. Screening was undertaken because 5%-10% of colonized patients later develop invasive infections, and 30%-60% die.
The first bloodstream infection was detected in May 2019. Per the report, published online Sept. 7 in Annals of Internal Medicine, “As of 1 January 2020, of 182 patients, 22 (12%) died within 30 days of C. auris identification; 47 (26%) died within 90 days. One of 47 deaths was attributed to C. auris.” Whole-genome sequencing showed that the isolates were all closely related in clade III.
Experts conducted extensive education in infection control at the LTACHs, and communication among the LTACHs and between the long-term facilities and acute care hospitals was improved. As a result, receiving facilities accepting transfers began culturing their newly admitted patients and quickly identified 4 of 99 patients with C. auris who had no known history of colonization. By October 2019, the outbreak was contained in two facilities, down from the nine where C. auris was initially found.
Dr. Diekema noted, “The challenge, of course, for a new emerging MDRO [multidrug-resistant organism] like Candida auris, is that the initial approach, in general, has to be almost passive, when you have not seen the organism. ... Passive surveillance means that you just carefully monitor your clinical cultures, and the first time you detect the MDRO of concern, then you begin doing the point prevalence surveys. ... This [prospective] kind of approach is really good for how we should move forward with both initial detection and containment of MDRO spread.”
Many outbreak studies are confined to a particular institution. Authors of an accompanying editorial commented that this study “underlines the importance of proactive protocols for outbreak investigations and containment measures across the entirety of the health care network serving at-risk patients.”
In her research, Dr. Karmarkar observed that, “some of these facilities don’t have the same infrastructure and infection prevention and control that an acute care hospital might.”
She said in an interview that, “one of the challenges was that people were so focused on COVID that they forgot about the MDROs. ... Some of the things that we recommend to help control Candida auris are also excellent practices for every other organism including COVID care. ... What I appreciated about this investigation is that every facility that we went to was so open to learning, so happy to have us there. They’re very interested in learning about Candida auris and understanding what they could do to control it.”
While recent attention has been on the frightening levels of multidrug resistance in C. auris, Dr. Karmarkar concluded that the “central message in our investigation is that with the right effort, the right approach, and the right team this is an intervenable issue. It’s not inevitable if the attention is focused on it to pick it up early and then try to contain it.”
Dr. Karmarkar reports no relevant financial relationships. Dr. Diekema reports research funding from bioMerieux and consulting fees from Opgen.
A version of this article first appeared on Medscape.com.
A new study from Orange County, California, shows how Candida auris, an emerging pathogen, was successfully identified and contained in long-term acute care hospitals (LTACHs) and ventilator-capable skilled-nursing facilities (vSNFs).
Lead author Ellora Karmarkar, MD, MSc, formerly an epidemic intelligence service officer with the Centers for Disease Control and Prevention and currently with the California Department of Public Health, said in an interview that the prospective surveillance of urine cultures for C. auris was prompted by “seeing what was happening in New York, New Jersey, and Illinois [being] pretty alarming for a lot of the health officials in California, [who] know that LTACHs are high-risk facilities because they take care of really sick people. Some of those people are there for a very long time.”
Therefore, the study authors decided to focus their investigations there, rather than in acute care hospitals, which were believed to be at lower risk for C. auris outbreaks.
The Orange County Health Department, working with the California Department of Health and the CDC, asked labs to prospectively identify all Candida isolates in urines from LTACHs between September 2018 and February 2019. Normally, labs do not speciate Candida from nonsterile body sites.
Dan Diekema, MD, an epidemiologist and clinical microbiologist at the University of Iowa, Iowa City, who was not involved in the study, told this news organization, “Acute care hospitals really ought to be moving toward doing species identification of Candida from nonsterile sites if they really want to have a better chance of detecting this early.”
The OCHD also screened LTACH and vSNF patients with composite cultures from the axilla-groin or nasal swabs. Screening was undertaken because 5%-10% of colonized patients later develop invasive infections, and 30%-60% die.
The first bloodstream infection was detected in May 2019. Per the report, published online Sept. 7 in Annals of Internal Medicine, “As of 1 January 2020, of 182 patients, 22 (12%) died within 30 days of C. auris identification; 47 (26%) died within 90 days. One of 47 deaths was attributed to C. auris.” Whole-genome sequencing showed that the isolates were all closely related in clade III.
Experts conducted extensive education in infection control at the LTACHs, and communication among the LTACHs and between the long-term facilities and acute care hospitals was improved. As a result, receiving facilities accepting transfers began culturing their newly admitted patients and quickly identified 4 of 99 patients with C. auris who had no known history of colonization. By October 2019, the outbreak was contained in two facilities, down from the nine where C. auris was initially found.
Dr. Diekema noted, “The challenge, of course, for a new emerging MDRO [multidrug-resistant organism] like Candida auris, is that the initial approach, in general, has to be almost passive, when you have not seen the organism. ... Passive surveillance means that you just carefully monitor your clinical cultures, and the first time you detect the MDRO of concern, then you begin doing the point prevalence surveys. ... This [prospective] kind of approach is really good for how we should move forward with both initial detection and containment of MDRO spread.”
Many outbreak studies are confined to a particular institution. Authors of an accompanying editorial commented that this study “underlines the importance of proactive protocols for outbreak investigations and containment measures across the entirety of the health care network serving at-risk patients.”
In her research, Dr. Karmarkar observed that, “some of these facilities don’t have the same infrastructure and infection prevention and control that an acute care hospital might.”
She said in an interview that, “one of the challenges was that people were so focused on COVID that they forgot about the MDROs. ... Some of the things that we recommend to help control Candida auris are also excellent practices for every other organism including COVID care. ... What I appreciated about this investigation is that every facility that we went to was so open to learning, so happy to have us there. They’re very interested in learning about Candida auris and understanding what they could do to control it.”
While recent attention has been on the frightening levels of multidrug resistance in C. auris, Dr. Karmarkar concluded that the “central message in our investigation is that with the right effort, the right approach, and the right team this is an intervenable issue. It’s not inevitable if the attention is focused on it to pick it up early and then try to contain it.”
Dr. Karmarkar reports no relevant financial relationships. Dr. Diekema reports research funding from bioMerieux and consulting fees from Opgen.
A version of this article first appeared on Medscape.com.
A new study from Orange County, California, shows how Candida auris, an emerging pathogen, was successfully identified and contained in long-term acute care hospitals (LTACHs) and ventilator-capable skilled-nursing facilities (vSNFs).
Lead author Ellora Karmarkar, MD, MSc, formerly an epidemic intelligence service officer with the Centers for Disease Control and Prevention and currently with the California Department of Public Health, said in an interview that the prospective surveillance of urine cultures for C. auris was prompted by “seeing what was happening in New York, New Jersey, and Illinois [being] pretty alarming for a lot of the health officials in California, [who] know that LTACHs are high-risk facilities because they take care of really sick people. Some of those people are there for a very long time.”
Therefore, the study authors decided to focus their investigations there, rather than in acute care hospitals, which were believed to be at lower risk for C. auris outbreaks.
The Orange County Health Department, working with the California Department of Health and the CDC, asked labs to prospectively identify all Candida isolates in urines from LTACHs between September 2018 and February 2019. Normally, labs do not speciate Candida from nonsterile body sites.
Dan Diekema, MD, an epidemiologist and clinical microbiologist at the University of Iowa, Iowa City, who was not involved in the study, told this news organization, “Acute care hospitals really ought to be moving toward doing species identification of Candida from nonsterile sites if they really want to have a better chance of detecting this early.”
The OCHD also screened LTACH and vSNF patients with composite cultures from the axilla-groin or nasal swabs. Screening was undertaken because 5%-10% of colonized patients later develop invasive infections, and 30%-60% die.
The first bloodstream infection was detected in May 2019. Per the report, published online Sept. 7 in Annals of Internal Medicine, “As of 1 January 2020, of 182 patients, 22 (12%) died within 30 days of C. auris identification; 47 (26%) died within 90 days. One of 47 deaths was attributed to C. auris.” Whole-genome sequencing showed that the isolates were all closely related in clade III.
Experts conducted extensive education in infection control at the LTACHs, and communication among the LTACHs and between the long-term facilities and acute care hospitals was improved. As a result, receiving facilities accepting transfers began culturing their newly admitted patients and quickly identified 4 of 99 patients with C. auris who had no known history of colonization. By October 2019, the outbreak was contained in two facilities, down from the nine where C. auris was initially found.
Dr. Diekema noted, “The challenge, of course, for a new emerging MDRO [multidrug-resistant organism] like Candida auris, is that the initial approach, in general, has to be almost passive, when you have not seen the organism. ... Passive surveillance means that you just carefully monitor your clinical cultures, and the first time you detect the MDRO of concern, then you begin doing the point prevalence surveys. ... This [prospective] kind of approach is really good for how we should move forward with both initial detection and containment of MDRO spread.”
Many outbreak studies are confined to a particular institution. Authors of an accompanying editorial commented that this study “underlines the importance of proactive protocols for outbreak investigations and containment measures across the entirety of the health care network serving at-risk patients.”
In her research, Dr. Karmarkar observed that, “some of these facilities don’t have the same infrastructure and infection prevention and control that an acute care hospital might.”
She said in an interview that, “one of the challenges was that people were so focused on COVID that they forgot about the MDROs. ... Some of the things that we recommend to help control Candida auris are also excellent practices for every other organism including COVID care. ... What I appreciated about this investigation is that every facility that we went to was so open to learning, so happy to have us there. They’re very interested in learning about Candida auris and understanding what they could do to control it.”
While recent attention has been on the frightening levels of multidrug resistance in C. auris, Dr. Karmarkar concluded that the “central message in our investigation is that with the right effort, the right approach, and the right team this is an intervenable issue. It’s not inevitable if the attention is focused on it to pick it up early and then try to contain it.”
Dr. Karmarkar reports no relevant financial relationships. Dr. Diekema reports research funding from bioMerieux and consulting fees from Opgen.
A version of this article first appeared on Medscape.com.
Want to see what COVID strain you have? The government says no
Every day, more than 140,000 people in the United States are diagnosed with COVID-19. But no matter how curious they are about which variant they are fighting, none of them will find out.
The country is dotted with labs that sequence the genomes of COVID-19 cases, and the Centers for Disease Control and Prevention tracks those results. But federal rules say those results are not allowed to make their way back to patients or doctors.
According to public health and infectious disease experts, this is unlikely to change any time soon.
“I know people want to know – I’ve had a lot of friends or family who’ve asked me how they can find out,” says Aubree Gordon, PhD, an epidemiology specialist at the University of Michigan, Ann Arbor. “I think it’s an interesting thing to find out, for sure. And it would certainly be nice to know. But because it probably isn’t necessary, there is little motivation to change the rules.”
Because the tests that are used have not been approved as diagnostic tools under the Clinical Laboratory Improvement Amendments program, which is overseen by the Centers for Medicare & Medicaid Services, they can only be used for research purposes.
In fact, the scientists doing the sequencing rarely have any patient information, Dr. Gordon says. For example, the Lauring Lab at University of Michigan – run by Adam Lauring, MD – focuses on viral evolution and currently tests for variants. But this is not done for the sake of the patient or the doctors treating the patient.
“The samples come in ... and they’ve been de-identified,”Dr. Gordon says. “This is just for research purposes. Not much patient information is shared with the researchers.”
But as of now, aside from sheer curiosity, there is not a reason to change this, says Timothy Brewer, MD, a professor of medicine and epidemiology at University of California, Los Angeles.
Although there are emerging variants – including the new Mu variant, also known as B.1.621 and recently classified as a “variant of interest” – the Delta variant accounts for about 99% of U.S. cases.
In addition, Dr. Brewer says, treatments are the same for all COVID-19 patients, regardless of the variant.
“There would have to be some clinical significance for there to be a good reason to give this information,” he says. “That would mean we would be doing something different treatment-wise depending on the variant. As of now, that is not the case.”
There is a loophole that allows labs to release variant information: They can develop their own tests. But they then must go through a lengthy validation process that proves their tests are as effective as the gold standard, says Mark Pandori, PhD, director of the Nevada State Public Health Laboratory.
But even with validation, it is too time-consuming and costly to sequence large numbers of cases, he says.
“The reason we’re not doing it routinely is there’s no way to do the genomic analysis on all the positives,” Dr. Pandori says. “It is about $110 dollars to do a sequence. It’s not like a standard PCR test.”
There is a hypothetical situation that may warrant the release of these results, Dr. Brewer says: If a variant emerges that evades vaccines.
“That would be a real public health issue,” he says. “You want to make sure there aren’t variants emerging somewhere that are escaping immunity.”
A version of this article first appeared on WebMD.com.
Every day, more than 140,000 people in the United States are diagnosed with COVID-19. But no matter how curious they are about which variant they are fighting, none of them will find out.

The country is dotted with labs that sequence the genomes of COVID-19 cases, and the Centers for Disease Control and Prevention tracks those results. But federal rules say those results are not allowed to make their way back to patients or doctors.
According to public health and infectious disease experts, this is unlikely to change any time soon.
“I know people want to know – I’ve had a lot of friends or family who’ve asked me how they can find out,” says Aubree Gordon, PhD, an epidemiology specialist at the University of Michigan, Ann Arbor. “I think it’s an interesting thing to find out, for sure. And it would certainly be nice to know. But because it probably isn’t necessary, there is little motivation to change the rules.”
Because the tests that are used have not been approved as diagnostic tools under the Clinical Laboratory Improvement Amendments program, which is overseen by the Centers for Medicare & Medicaid Services, they can only be used for research purposes.
In fact, the scientists doing the sequencing rarely have any patient information, Dr. Gordon says. For example, the Lauring Lab at University of Michigan – run by Adam Lauring, MD – focuses on viral evolution and currently tests for variants. But this is not done for the sake of the patient or the doctors treating the patient.
“The samples come in ... and they’ve been de-identified,”Dr. Gordon says. “This is just for research purposes. Not much patient information is shared with the researchers.”
But as of now, aside from sheer curiosity, there is not a reason to change this, says Timothy Brewer, MD, a professor of medicine and epidemiology at University of California, Los Angeles.
Although there are emerging variants – including the new Mu variant, also known as B.1.621 and recently classified as a “variant of interest” – the Delta variant accounts for about 99% of U.S. cases.
In addition, Dr. Brewer says, treatments are the same for all COVID-19 patients, regardless of the variant.
“There would have to be some clinical significance for there to be a good reason to give this information,” he says. “That would mean we would be doing something different treatment-wise depending on the variant. As of now, that is not the case.”
There is a loophole that allows labs to release variant information: They can develop their own tests. But they then must go through a lengthy validation process that proves their tests are as effective as the gold standard, says Mark Pandori, PhD, director of the Nevada State Public Health Laboratory.
But even with validation, it is too time-consuming and costly to sequence large numbers of cases, he says.
“The reason we’re not doing it routinely is there’s no way to do the genomic analysis on all the positives,” Dr. Pandori says. “It is about $110 dollars to do a sequence. It’s not like a standard PCR test.”
There is a hypothetical situation that may warrant the release of these results, Dr. Brewer says: If a variant emerges that evades vaccines.
“That would be a real public health issue,” he says. “You want to make sure there aren’t variants emerging somewhere that are escaping immunity.”
A version of this article first appeared on WebMD.com.
Every day, more than 140,000 people in the United States are diagnosed with COVID-19. But no matter how curious they are about which variant they are fighting, none of them will find out.

The country is dotted with labs that sequence the genomes of COVID-19 cases, and the Centers for Disease Control and Prevention tracks those results. But federal rules say those results are not allowed to make their way back to patients or doctors.
According to public health and infectious disease experts, this is unlikely to change any time soon.
“I know people want to know – I’ve had a lot of friends or family who’ve asked me how they can find out,” says Aubree Gordon, PhD, an epidemiology specialist at the University of Michigan, Ann Arbor. “I think it’s an interesting thing to find out, for sure. And it would certainly be nice to know. But because it probably isn’t necessary, there is little motivation to change the rules.”
Because the tests that are used have not been approved as diagnostic tools under the Clinical Laboratory Improvement Amendments program, which is overseen by the Centers for Medicare & Medicaid Services, they can only be used for research purposes.
In fact, the scientists doing the sequencing rarely have any patient information, Dr. Gordon says. For example, the Lauring Lab at University of Michigan – run by Adam Lauring, MD – focuses on viral evolution and currently tests for variants. But this is not done for the sake of the patient or the doctors treating the patient.
“The samples come in ... and they’ve been de-identified,”Dr. Gordon says. “This is just for research purposes. Not much patient information is shared with the researchers.”
But as of now, aside from sheer curiosity, there is not a reason to change this, says Timothy Brewer, MD, a professor of medicine and epidemiology at University of California, Los Angeles.
Although there are emerging variants – including the new Mu variant, also known as B.1.621 and recently classified as a “variant of interest” – the Delta variant accounts for about 99% of U.S. cases.
In addition, Dr. Brewer says, treatments are the same for all COVID-19 patients, regardless of the variant.
“There would have to be some clinical significance for there to be a good reason to give this information,” he says. “That would mean we would be doing something different treatment-wise depending on the variant. As of now, that is not the case.”
There is a loophole that allows labs to release variant information: They can develop their own tests. But they then must go through a lengthy validation process that proves their tests are as effective as the gold standard, says Mark Pandori, PhD, director of the Nevada State Public Health Laboratory.
But even with validation, it is too time-consuming and costly to sequence large numbers of cases, he says.
“The reason we’re not doing it routinely is there’s no way to do the genomic analysis on all the positives,” Dr. Pandori says. “It is about $110 dollars to do a sequence. It’s not like a standard PCR test.”
There is a hypothetical situation that may warrant the release of these results, Dr. Brewer says: If a variant emerges that evades vaccines.
“That would be a real public health issue,” he says. “You want to make sure there aren’t variants emerging somewhere that are escaping immunity.”
A version of this article first appeared on WebMD.com.
Children and COVID: New cases down slightly from record high
Weekly cases of COVID-19 in children dropped for the first time since June, and daily hospitalizations appear to be falling, even as the pace of vaccinations continues to slow among the youngest eligible recipients, according to new data.
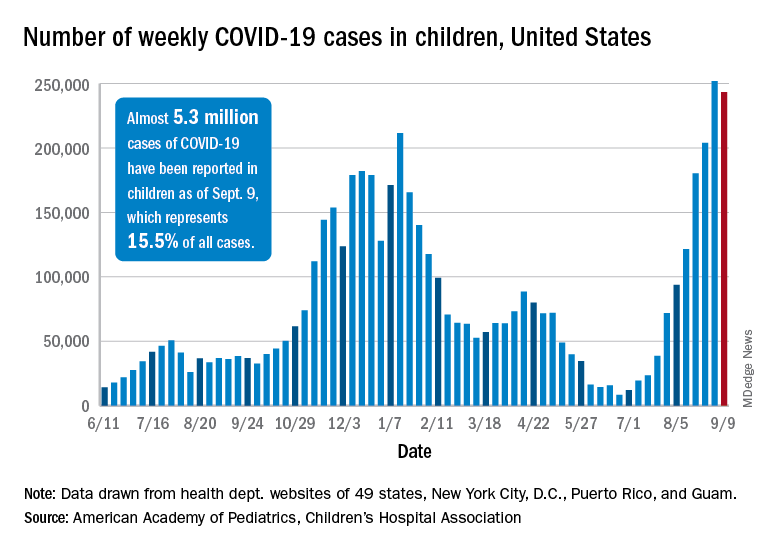
Despite the 3.3% decline from the previous week’s record high, the new-case count still topped 243,000 for the week of Sept. 3-9, putting the total number of cases in children at almost 5.3 million since the pandemic began.
Hospitalizations seem to have peaked on Sept. 4, when the rate for children aged 0-17 years reached 0.51 per 100,000 population. The admission rate for confirmed COVID-19 has dropped steadily since then and was down to 0.45 per 100,000 on Sept. 11, the last day for which preliminary data from the Centers for Disease Control and Prevention were available.
On the prevention side, fully vaccinated children aged 12-17 years represented 5.5% of all Americans who had completed the vaccine regimen as of Sept. 13. Vaccine initiation, however, has dropped for 5 consecutive weeks in 12- to 15-year-olds and in 4 of the last 5 weeks among 16- and 17-year-olds, the CDC said on its COVID Data Tracker.
Just under 199,000 children aged 12-15 received their first dose of the COVID-19 vaccine during the week of Sept. 7-13. That’s down by 18.5% from the week before and by 51.6% since Aug. 9, the last week that vaccine initiation increased for the age group. Among 16- and 17-year-olds, the 83,000 new recipients that week was a decrease of 25.7% from the previous week and a decline of 47% since the summer peak of Aug. 9, the CDC data show.
Those newest recipients bring at-least-one-dose status to 52.0% of those aged 12-15 and 59.9% of the 16- and 17-year-olds, while 40.3% and 48.9% were fully vaccinated as of Sept. 13. Corresponding figures for some of the older groups are 61.6%/49.7% (age 18-24 years), 73.8%/63.1% (40-49 years), and 95.1%/84.5% (65-74 years), the CDC said.
Vaccine coverage for children at the state level deviates considerably from the national averages. The highest rates for children aged 12-17 are to be found in Vermont, where 76% have received at least one dose, the AAP reported in a separate analysis. Massachusetts is just below that but also comes in at 76% by virtue of a rounding error. The other states in the top five are Connecticut (74%), Hawaii (73%), and Rhode Island (71%).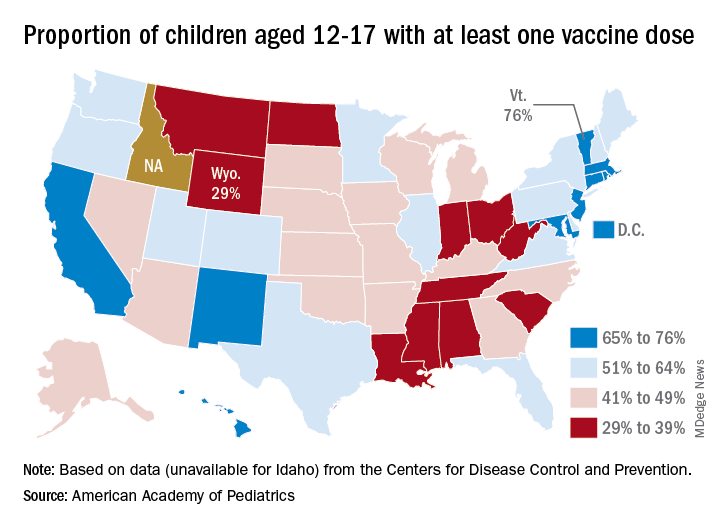
The lowest vaccination rate for children comes from Wyoming (29%), which is preceded by North Dakota (33%), West Virginia (33%), Alabama (33%), and Mississippi (34%). the AAP said based on data from the CDC, which does not include Idaho.
In a bit of a side note, West Virginia’s Republican governor, Jim Justice, recently said this about vaccine reluctance in his state: “For God’s sakes a livin’, how difficult is this to understand? Why in the world do we have to come up with these crazy ideas – and they’re crazy ideas – that the vaccine’s got something in it and it’s tracing people wherever they go? And the same very people that are saying that are carrying their cellphones around. I mean, come on. Come on.”
Over the last 3 weeks, the District of Columbia has had the largest increase in children having received at least one dose: 10 percentage points, as it went from 58% to 68%. The next-largest improvement – 7 percentage points – occurred in Georgia (34% to 41%), New Mexico (61% to 68%), New York (55% to 62%), and Washington (57% to 64%), the AAP said in its weekly vaccination trends report.
Weekly cases of COVID-19 in children dropped for the first time since June, and daily hospitalizations appear to be falling, even as the pace of vaccinations continues to slow among the youngest eligible recipients, according to new data.

Despite the 3.3% decline from the previous week’s record high, the new-case count still topped 243,000 for the week of Sept. 3-9, putting the total number of cases in children at almost 5.3 million since the pandemic began.
Hospitalizations seem to have peaked on Sept. 4, when the rate for children aged 0-17 years reached 0.51 per 100,000 population. The admission rate for confirmed COVID-19 has dropped steadily since then and was down to 0.45 per 100,000 on Sept. 11, the last day for which preliminary data from the Centers for Disease Control and Prevention were available.
On the prevention side, fully vaccinated children aged 12-17 years represented 5.5% of all Americans who had completed the vaccine regimen as of Sept. 13. Vaccine initiation, however, has dropped for 5 consecutive weeks in 12- to 15-year-olds and in 4 of the last 5 weeks among 16- and 17-year-olds, the CDC said on its COVID Data Tracker.
Just under 199,000 children aged 12-15 received their first dose of the COVID-19 vaccine during the week of Sept. 7-13. That’s down by 18.5% from the week before and by 51.6% since Aug. 9, the last week that vaccine initiation increased for the age group. Among 16- and 17-year-olds, the 83,000 new recipients that week was a decrease of 25.7% from the previous week and a decline of 47% since the summer peak of Aug. 9, the CDC data show.
Those newest recipients bring at-least-one-dose status to 52.0% of those aged 12-15 and 59.9% of the 16- and 17-year-olds, while 40.3% and 48.9% were fully vaccinated as of Sept. 13. Corresponding figures for some of the older groups are 61.6%/49.7% (age 18-24 years), 73.8%/63.1% (40-49 years), and 95.1%/84.5% (65-74 years), the CDC said.
Vaccine coverage for children at the state level deviates considerably from the national averages. The highest rates for children aged 12-17 are to be found in Vermont, where 76% have received at least one dose, the AAP reported in a separate analysis. Massachusetts is just below that but also comes in at 76% by virtue of a rounding error. The other states in the top five are Connecticut (74%), Hawaii (73%), and Rhode Island (71%).
The lowest vaccination rate for children comes from Wyoming (29%), which is preceded by North Dakota (33%), West Virginia (33%), Alabama (33%), and Mississippi (34%). the AAP said based on data from the CDC, which does not include Idaho.
In a bit of a side note, West Virginia’s Republican governor, Jim Justice, recently said this about vaccine reluctance in his state: “For God’s sakes a livin’, how difficult is this to understand? Why in the world do we have to come up with these crazy ideas – and they’re crazy ideas – that the vaccine’s got something in it and it’s tracing people wherever they go? And the same very people that are saying that are carrying their cellphones around. I mean, come on. Come on.”
Over the last 3 weeks, the District of Columbia has had the largest increase in children having received at least one dose: 10 percentage points, as it went from 58% to 68%. The next-largest improvement – 7 percentage points – occurred in Georgia (34% to 41%), New Mexico (61% to 68%), New York (55% to 62%), and Washington (57% to 64%), the AAP said in its weekly vaccination trends report.
Weekly cases of COVID-19 in children dropped for the first time since June, and daily hospitalizations appear to be falling, even as the pace of vaccinations continues to slow among the youngest eligible recipients, according to new data.

Despite the 3.3% decline from the previous week’s record high, the new-case count still topped 243,000 for the week of Sept. 3-9, putting the total number of cases in children at almost 5.3 million since the pandemic began.
Hospitalizations seem to have peaked on Sept. 4, when the rate for children aged 0-17 years reached 0.51 per 100,000 population. The admission rate for confirmed COVID-19 has dropped steadily since then and was down to 0.45 per 100,000 on Sept. 11, the last day for which preliminary data from the Centers for Disease Control and Prevention were available.
On the prevention side, fully vaccinated children aged 12-17 years represented 5.5% of all Americans who had completed the vaccine regimen as of Sept. 13. Vaccine initiation, however, has dropped for 5 consecutive weeks in 12- to 15-year-olds and in 4 of the last 5 weeks among 16- and 17-year-olds, the CDC said on its COVID Data Tracker.
Just under 199,000 children aged 12-15 received their first dose of the COVID-19 vaccine during the week of Sept. 7-13. That’s down by 18.5% from the week before and by 51.6% since Aug. 9, the last week that vaccine initiation increased for the age group. Among 16- and 17-year-olds, the 83,000 new recipients that week was a decrease of 25.7% from the previous week and a decline of 47% since the summer peak of Aug. 9, the CDC data show.
Those newest recipients bring at-least-one-dose status to 52.0% of those aged 12-15 and 59.9% of the 16- and 17-year-olds, while 40.3% and 48.9% were fully vaccinated as of Sept. 13. Corresponding figures for some of the older groups are 61.6%/49.7% (age 18-24 years), 73.8%/63.1% (40-49 years), and 95.1%/84.5% (65-74 years), the CDC said.
Vaccine coverage for children at the state level deviates considerably from the national averages. The highest rates for children aged 12-17 are to be found in Vermont, where 76% have received at least one dose, the AAP reported in a separate analysis. Massachusetts is just below that but also comes in at 76% by virtue of a rounding error. The other states in the top five are Connecticut (74%), Hawaii (73%), and Rhode Island (71%).
The lowest vaccination rate for children comes from Wyoming (29%), which is preceded by North Dakota (33%), West Virginia (33%), Alabama (33%), and Mississippi (34%). the AAP said based on data from the CDC, which does not include Idaho.
In a bit of a side note, West Virginia’s Republican governor, Jim Justice, recently said this about vaccine reluctance in his state: “For God’s sakes a livin’, how difficult is this to understand? Why in the world do we have to come up with these crazy ideas – and they’re crazy ideas – that the vaccine’s got something in it and it’s tracing people wherever they go? And the same very people that are saying that are carrying their cellphones around. I mean, come on. Come on.”
Over the last 3 weeks, the District of Columbia has had the largest increase in children having received at least one dose: 10 percentage points, as it went from 58% to 68%. The next-largest improvement – 7 percentage points – occurred in Georgia (34% to 41%), New Mexico (61% to 68%), New York (55% to 62%), and Washington (57% to 64%), the AAP said in its weekly vaccination trends report.
Antibiotic use and colon cancer: More evidence of link
The latest data come from a Swedish population study. Investigators analyzed data from more than 40,000 colorectal cancer patients and 200,000 cancer-free control persons.
They found that moderate use of antibiotics increased the risk for proximal colon cancer by 9% and that very high antibiotic use increased the risk by 17%.
In contrast, the risk for rectal cancer was reduced by 4% with moderate use and 9% with very high use, but this association was confined to women.
Antibiotic use was categorized as no use (no reported use of antibiotics during the study period), low (use during a period of 1-10 days), moderate (11-60 days), high (61-180 days), and very high (>180 days).
The study, led by Sophia Harlid, PhD, department of radiation sciences, oncology, Umeå University, Sweden, was published online on Sept. 1 in the Journal of the National Cancer Institute.
The results complement findings from a recent study from Scotland, which found that a history of antibiotic use among individuals younger than 50 appeared to increase the risk of developing colon cancer but not rectal cancer by 49%.
The new data from Sweden “strengthen prior evidence and provide new insights into site-specific carcinogenesis as well as indirect support for the role of gut microbiota,” lead author Dr. Dr. Harlid commented in an interview.
“The positive associations between antibiotics use and proximal colon cancer began at the lowest level of antibiotics use, providing a potential justification for reducing antibiotics prescriptions in clinical practice,” she added.
In their article, the team suggests that the increased risk could be a result of antibiotics having a “disruptive effect” on the gut microbiome.
The finding of an increased risk for cancer in the proximal colon but not further along the alimentary tract “is consistent with a high microbial impact in the proximal colon and a decreasing concentration of short-chain fatty acids along the colon,” the authors comment.
This results “in higher bacterial activity, biofilm formation, and fermentation in the proximal compared with the distal colon and rectum.”
A further analysis showed that the use of quinolones and sulfonamides and/or trimethoprims was associated with an increased risk for proximal colon cancer, whereas use of nitrofurantoins, macrolides and/or lincosamides, and metronidazoles and/or tinidazoles was inversely associated with rectal cancer.
Details of the study findings
For their study, the team analyzed complete-population data from Swedish national registers for the period July 1, 2005 to Dec. 31, 2016.
They matched case patients who were diagnosed with colorectal cancer from Jan. 1, 2010 to Dec. 31, 2016 with cancer-free control persons in a 1:5 ratio. Data on antibiotic use were extracted from the Swedish Prescribed Drug Register.
Other variables, such as socioeconomic factors and health care utilization, were obtained from the Swedish Inpatient Register and the Longitudinal Integration Database for Health Insurance and Labor Market Studies.
The team identified 40,545 patients with colorectal cancer cases; there were 202,720 control persons. Just over half (52.9%) of the participants were men; the mean age at cancer diagnosis was 72 years. Among the cases, 36.4% were proximal colon cancers, 29.3% were distal colon cancers, and 33.0% rectal cancers.
Control patients were more likely to have been prescribed no antibiotics, at 22.4% versus 18.7% for case patients. Case patients were more likely than control persons to have used antibiotics for more than 2 months, at 20.8% versus 19.3% (P < .001).
Overall, antibiotic use was positively associated with colorectal cancer. In comparison with no use, the odds ratio for moderate use was 1.15; for very high use, it was 1.17 (P < .001 for trend).
Excluding all antibiotic use during the 2 years prior to a colorectal cancer diagnosis attenuated the association, such that it was no longer significant for very high use versus no antibiotic use.
Applying this cutoff to the remaining analyses, the team found that the dose-response relationship between antibiotic use and colorectal cancer was largely confined to proximal colon cancer, at an odds ratio of 1.09 for moderate use and 1.17 for very high use in comparison with no use (P < .001 for trend).
For distal colon cancer, the relationship was “close to null.”
There was a slight inverse relationship between rectal cancer and antibiotic use, at an odds rate of 0.96 for moderate use and 0.91 for very high use versus no use (P < .001 for trend). This association was found in women only, whereas the other associations were seen in both men and women.
The study was supported by the Lion’s Cancer Research Foundation, Umeå University, and Region Västerbotten. Dr. Harlid has disclosed no relevant financial relationships. Three coauthors report various relationships with industry, as noted in the original article.
A version of this article first appeared on Medscape.com.
The latest data come from a Swedish population study. Investigators analyzed data from more than 40,000 colorectal cancer patients and 200,000 cancer-free control persons.
They found that moderate use of antibiotics increased the risk for proximal colon cancer by 9% and that very high antibiotic use increased the risk by 17%.
In contrast, the risk for rectal cancer was reduced by 4% with moderate use and 9% with very high use, but this association was confined to women.
Antibiotic use was categorized as no use (no reported use of antibiotics during the study period), low (use during a period of 1-10 days), moderate (11-60 days), high (61-180 days), and very high (>180 days).
The study, led by Sophia Harlid, PhD, department of radiation sciences, oncology, Umeå University, Sweden, was published online on Sept. 1 in the Journal of the National Cancer Institute.
The results complement findings from a recent study from Scotland, which found that a history of antibiotic use among individuals younger than 50 appeared to increase the risk of developing colon cancer but not rectal cancer by 49%.
The new data from Sweden “strengthen prior evidence and provide new insights into site-specific carcinogenesis as well as indirect support for the role of gut microbiota,” lead author Dr. Dr. Harlid commented in an interview.
“The positive associations between antibiotics use and proximal colon cancer began at the lowest level of antibiotics use, providing a potential justification for reducing antibiotics prescriptions in clinical practice,” she added.
In their article, the team suggests that the increased risk could be a result of antibiotics having a “disruptive effect” on the gut microbiome.
The finding of an increased risk for cancer in the proximal colon but not further along the alimentary tract “is consistent with a high microbial impact in the proximal colon and a decreasing concentration of short-chain fatty acids along the colon,” the authors comment.
This results “in higher bacterial activity, biofilm formation, and fermentation in the proximal compared with the distal colon and rectum.”
A further analysis showed that the use of quinolones and sulfonamides and/or trimethoprims was associated with an increased risk for proximal colon cancer, whereas use of nitrofurantoins, macrolides and/or lincosamides, and metronidazoles and/or tinidazoles was inversely associated with rectal cancer.
Details of the study findings
For their study, the team analyzed complete-population data from Swedish national registers for the period July 1, 2005 to Dec. 31, 2016.
They matched case patients who were diagnosed with colorectal cancer from Jan. 1, 2010 to Dec. 31, 2016 with cancer-free control persons in a 1:5 ratio. Data on antibiotic use were extracted from the Swedish Prescribed Drug Register.
Other variables, such as socioeconomic factors and health care utilization, were obtained from the Swedish Inpatient Register and the Longitudinal Integration Database for Health Insurance and Labor Market Studies.
The team identified 40,545 patients with colorectal cancer cases; there were 202,720 control persons. Just over half (52.9%) of the participants were men; the mean age at cancer diagnosis was 72 years. Among the cases, 36.4% were proximal colon cancers, 29.3% were distal colon cancers, and 33.0% rectal cancers.
Control patients were more likely to have been prescribed no antibiotics, at 22.4% versus 18.7% for case patients. Case patients were more likely than control persons to have used antibiotics for more than 2 months, at 20.8% versus 19.3% (P < .001).
Overall, antibiotic use was positively associated with colorectal cancer. In comparison with no use, the odds ratio for moderate use was 1.15; for very high use, it was 1.17 (P < .001 for trend).
Excluding all antibiotic use during the 2 years prior to a colorectal cancer diagnosis attenuated the association, such that it was no longer significant for very high use versus no antibiotic use.
Applying this cutoff to the remaining analyses, the team found that the dose-response relationship between antibiotic use and colorectal cancer was largely confined to proximal colon cancer, at an odds ratio of 1.09 for moderate use and 1.17 for very high use in comparison with no use (P < .001 for trend).
For distal colon cancer, the relationship was “close to null.”
There was a slight inverse relationship between rectal cancer and antibiotic use, at an odds rate of 0.96 for moderate use and 0.91 for very high use versus no use (P < .001 for trend). This association was found in women only, whereas the other associations were seen in both men and women.
The study was supported by the Lion’s Cancer Research Foundation, Umeå University, and Region Västerbotten. Dr. Harlid has disclosed no relevant financial relationships. Three coauthors report various relationships with industry, as noted in the original article.
A version of this article first appeared on Medscape.com.
The latest data come from a Swedish population study. Investigators analyzed data from more than 40,000 colorectal cancer patients and 200,000 cancer-free control persons.
They found that moderate use of antibiotics increased the risk for proximal colon cancer by 9% and that very high antibiotic use increased the risk by 17%.
In contrast, the risk for rectal cancer was reduced by 4% with moderate use and 9% with very high use, but this association was confined to women.
Antibiotic use was categorized as no use (no reported use of antibiotics during the study period), low (use during a period of 1-10 days), moderate (11-60 days), high (61-180 days), and very high (>180 days).
The study, led by Sophia Harlid, PhD, department of radiation sciences, oncology, Umeå University, Sweden, was published online on Sept. 1 in the Journal of the National Cancer Institute.
The results complement findings from a recent study from Scotland, which found that a history of antibiotic use among individuals younger than 50 appeared to increase the risk of developing colon cancer but not rectal cancer by 49%.
The new data from Sweden “strengthen prior evidence and provide new insights into site-specific carcinogenesis as well as indirect support for the role of gut microbiota,” lead author Dr. Dr. Harlid commented in an interview.
“The positive associations between antibiotics use and proximal colon cancer began at the lowest level of antibiotics use, providing a potential justification for reducing antibiotics prescriptions in clinical practice,” she added.
In their article, the team suggests that the increased risk could be a result of antibiotics having a “disruptive effect” on the gut microbiome.
The finding of an increased risk for cancer in the proximal colon but not further along the alimentary tract “is consistent with a high microbial impact in the proximal colon and a decreasing concentration of short-chain fatty acids along the colon,” the authors comment.
This results “in higher bacterial activity, biofilm formation, and fermentation in the proximal compared with the distal colon and rectum.”
A further analysis showed that the use of quinolones and sulfonamides and/or trimethoprims was associated with an increased risk for proximal colon cancer, whereas use of nitrofurantoins, macrolides and/or lincosamides, and metronidazoles and/or tinidazoles was inversely associated with rectal cancer.
Details of the study findings
For their study, the team analyzed complete-population data from Swedish national registers for the period July 1, 2005 to Dec. 31, 2016.
They matched case patients who were diagnosed with colorectal cancer from Jan. 1, 2010 to Dec. 31, 2016 with cancer-free control persons in a 1:5 ratio. Data on antibiotic use were extracted from the Swedish Prescribed Drug Register.
Other variables, such as socioeconomic factors and health care utilization, were obtained from the Swedish Inpatient Register and the Longitudinal Integration Database for Health Insurance and Labor Market Studies.
The team identified 40,545 patients with colorectal cancer cases; there were 202,720 control persons. Just over half (52.9%) of the participants were men; the mean age at cancer diagnosis was 72 years. Among the cases, 36.4% were proximal colon cancers, 29.3% were distal colon cancers, and 33.0% rectal cancers.
Control patients were more likely to have been prescribed no antibiotics, at 22.4% versus 18.7% for case patients. Case patients were more likely than control persons to have used antibiotics for more than 2 months, at 20.8% versus 19.3% (P < .001).
Overall, antibiotic use was positively associated with colorectal cancer. In comparison with no use, the odds ratio for moderate use was 1.15; for very high use, it was 1.17 (P < .001 for trend).
Excluding all antibiotic use during the 2 years prior to a colorectal cancer diagnosis attenuated the association, such that it was no longer significant for very high use versus no antibiotic use.
Applying this cutoff to the remaining analyses, the team found that the dose-response relationship between antibiotic use and colorectal cancer was largely confined to proximal colon cancer, at an odds ratio of 1.09 for moderate use and 1.17 for very high use in comparison with no use (P < .001 for trend).
For distal colon cancer, the relationship was “close to null.”
There was a slight inverse relationship between rectal cancer and antibiotic use, at an odds rate of 0.96 for moderate use and 0.91 for very high use versus no use (P < .001 for trend). This association was found in women only, whereas the other associations were seen in both men and women.
The study was supported by the Lion’s Cancer Research Foundation, Umeå University, and Region Västerbotten. Dr. Harlid has disclosed no relevant financial relationships. Three coauthors report various relationships with industry, as noted in the original article.
A version of this article first appeared on Medscape.com.
USPSTF update: Screen young asymptomatic women for chlamydia and gonorrhea
But evidence for screening men remains insufficient, task force says
The U.S. Preventive Services Task Force has updated its 2014 statement on screening asymptomatic individuals for chlamydia and gonorrhea infection.
Published online in JAMA, the 2021 version recommends that all sexually active women aged 24 years or younger and at-risk women 25 years or older should be screened for chlamydia and gonorrhea.
As in 2014, the task force made no screening recommendation for men owing to inconclusive evidence of benefit.
With cases of sexually transmitted infections reaching all-time highs, Amy G. Cantor, MD, MPH, of the Pacific Northwest Evidence-based Practice Center at Oregon Health & Science University, Portland, and colleagues noted that chlamydia and gonorrhea are among the most common STIs in this country. According to the Centers for Disease Control and Prevention, 2019 saw approximately 1.8 million reported cases of chlamydia and more than 600,000 of gonorrhea.
In the current analysis of 27 observational and randomized studies comprising 179,515 patients, the USPSTF panel found that, compared with no screening, chlamydia screening was significantly associated with a reduced risk of pelvic inflammatory disease (PID) in young women in 2 out of 4 trials.
The authors cautioned, however, that the magnitude of benefit was relatively small. No studies reported on screening effectiveness in men, except for one reporting rates of epididymitis, and no studies were done on pregnant women for any outcome.
The largest and newest study, the Australian Chlamydia Control Effectiveness Pilot trial of 2018, assessed chlamydia screening against usual care in 180,355 men and women aged 16-29 years in 130 rural Australian primary care clinics. Screening was associated with a reduced risk of hospital-diagnosed PID: the absolute risk was 0.24% for screening versus 0.38% for usual care (unadjusted risk ratio, 0.6; 95% confidence interval, 0.4-1.0). It was not, however, significantly associated with a reduced risk of clinic-diagnosed PID, with an absolute risk of 0.45% versus 0.39% (RR, 1.1; 95% CI, 0.7-18). Nor did it correlate with a risk reduction for clinic-diagnosed epididymitis: 0.26% vs. 0.27% (RR, 0.9; 95% CI, 0.6-1.4).
While risk prediction criteria apart from age were only minimally accurate, testing for asymptomatic chlamydial and gonococcal infections was highly accurate at most anatomical sites, including urine and self-collected specimens, the investigators observed. Age 22 years or younger alone versus multi-item risk criteria demonstrated similar discrimination in a study that included symptomatic and asymptomatic women.
Sensitivity of chlamydial testing was similar at endocervical (89%-100%) and self- and clinician-collected vaginal (90%-100%) sites for women and at meatal (100%), urethral (99%), and rectal (92%) sites for men. It was lower, however, at pharyngeal sites (69.2%) for men who have sex with men (MSM).
Sensitivity of gonococcal testing was 89% or greater for all anatomical samples. False-positive and false-negative testing rates were low across anatomical sites and collection methods.
“Effectiveness of screening in men and during pregnancy, optimal screening intervals, and adverse effects of screening require further evaluation, Dr. Cantor and associates concluded.
In an accompanying editorial, Jeanne Marrazzo, MD, MPH, and Jodie Dionne-Odom, MD, MSPH, of the division of infectious diseases at the University of Alabama at Birmingham, called the guidelines “timely” and “powerful agents of change” that “influence a wide spectrum of health-based metrics, from quality assurance measures to criteria for financial reimbursement.”
They pointed out that men who have sex with men are experiencing historically high rates of gonorrhea, with most infections occurring extragenitally at the pharynx or rectum. In 2019 CDC data, MSM had substantially higher rates of gonorrhea than men who had sex only with women. They recommended that guidelines for men consider STI risk because of sexual relations with men, women, or both.
“Comprehensive screening guidelines for common STIs like chlamydia and gonorrhea could incorporate the limited evidence base for MSM, whether it is regular practice or not,” they wrote, with the same approach for women who have sex with women but may be at risk for chlamydia, particularly if they also have sex with men.
In their view, these latest guidelines appropriately prioritize high-level clinically based data. They pointed, however, to recent progress in understanding the pathogenesis of upper reproductive tract infection in women and the sexual networks behind the current resurgence of STIs in the United States in the failure to manage exposed sex partners.
“Considering these critical advances in the evolution of clinic-based screening guidelines is a work in progress,” they wrote, “the dialogue among basic scientists, clinical trial investigators, and public health professionals to inform the next version of updated USPSTF chlamydia and gonorrhea screening guidelines should start now.”
In the opinion of Jennifer L. Reed, MD, MS, a professor of pediatrics and an emergency medicine physician at Cincinnati Children’s Hospital Medical Center and not involved in the updated statement, the recommendations are very reasonable. “The highest rates of infection occur in females 15-24 years of age, and therefore asymptomatic screening for chlamydia and gonorrhea is imperative at least annually or more often if they are high risk,” she said in an interview.
“I would hope that providers increase their asymptomatic screening as a result of these recommendations and highly consider it in the younger men,” Dr. Reed added. “I see a very high rate of gonorrhea and chlamydia infections.” Her center is studying the implementation of gonorrhea and chlamydia asymptomatic screening for adolescents in the pediatric emergency department, a high-risk patient population that will benefit from STI screening opportunities in nontraditional settings.
This research was funded by the Agency for Healthcare Research and Quality and the Department of Health & Human Services under a contract to support the USPSTF. One statement coauthor reported personal fees from Insmed, Paratek, RedHill, and Spero, as well as grants from Insmed. No other disclosures were reported. Dr. Dionne-Odom reported grants from the National Institutes of Health/National Institute of Child Health and Development. Dr. Reed reported a grant from NIH/NICHD for a pragmatic trial of improving STI detection in the pediatric ED.
But evidence for screening men remains insufficient, task force says
But evidence for screening men remains insufficient, task force says
The U.S. Preventive Services Task Force has updated its 2014 statement on screening asymptomatic individuals for chlamydia and gonorrhea infection.
Published online in JAMA, the 2021 version recommends that all sexually active women aged 24 years or younger and at-risk women 25 years or older should be screened for chlamydia and gonorrhea.
As in 2014, the task force made no screening recommendation for men owing to inconclusive evidence of benefit.
With cases of sexually transmitted infections reaching all-time highs, Amy G. Cantor, MD, MPH, of the Pacific Northwest Evidence-based Practice Center at Oregon Health & Science University, Portland, and colleagues noted that chlamydia and gonorrhea are among the most common STIs in this country. According to the Centers for Disease Control and Prevention, 2019 saw approximately 1.8 million reported cases of chlamydia and more than 600,000 of gonorrhea.
In the current analysis of 27 observational and randomized studies comprising 179,515 patients, the USPSTF panel found that, compared with no screening, chlamydia screening was significantly associated with a reduced risk of pelvic inflammatory disease (PID) in young women in 2 out of 4 trials.
The authors cautioned, however, that the magnitude of benefit was relatively small. No studies reported on screening effectiveness in men, except for one reporting rates of epididymitis, and no studies were done on pregnant women for any outcome.
The largest and newest study, the Australian Chlamydia Control Effectiveness Pilot trial of 2018, assessed chlamydia screening against usual care in 180,355 men and women aged 16-29 years in 130 rural Australian primary care clinics. Screening was associated with a reduced risk of hospital-diagnosed PID: the absolute risk was 0.24% for screening versus 0.38% for usual care (unadjusted risk ratio, 0.6; 95% confidence interval, 0.4-1.0). It was not, however, significantly associated with a reduced risk of clinic-diagnosed PID, with an absolute risk of 0.45% versus 0.39% (RR, 1.1; 95% CI, 0.7-18). Nor did it correlate with a risk reduction for clinic-diagnosed epididymitis: 0.26% vs. 0.27% (RR, 0.9; 95% CI, 0.6-1.4).
While risk prediction criteria apart from age were only minimally accurate, testing for asymptomatic chlamydial and gonococcal infections was highly accurate at most anatomical sites, including urine and self-collected specimens, the investigators observed. Age 22 years or younger alone versus multi-item risk criteria demonstrated similar discrimination in a study that included symptomatic and asymptomatic women.
Sensitivity of chlamydial testing was similar at endocervical (89%-100%) and self- and clinician-collected vaginal (90%-100%) sites for women and at meatal (100%), urethral (99%), and rectal (92%) sites for men. It was lower, however, at pharyngeal sites (69.2%) for men who have sex with men (MSM).
Sensitivity of gonococcal testing was 89% or greater for all anatomical samples. False-positive and false-negative testing rates were low across anatomical sites and collection methods.
“Effectiveness of screening in men and during pregnancy, optimal screening intervals, and adverse effects of screening require further evaluation, Dr. Cantor and associates concluded.
In an accompanying editorial, Jeanne Marrazzo, MD, MPH, and Jodie Dionne-Odom, MD, MSPH, of the division of infectious diseases at the University of Alabama at Birmingham, called the guidelines “timely” and “powerful agents of change” that “influence a wide spectrum of health-based metrics, from quality assurance measures to criteria for financial reimbursement.”
They pointed out that men who have sex with men are experiencing historically high rates of gonorrhea, with most infections occurring extragenitally at the pharynx or rectum. In 2019 CDC data, MSM had substantially higher rates of gonorrhea than men who had sex only with women. They recommended that guidelines for men consider STI risk because of sexual relations with men, women, or both.
“Comprehensive screening guidelines for common STIs like chlamydia and gonorrhea could incorporate the limited evidence base for MSM, whether it is regular practice or not,” they wrote, with the same approach for women who have sex with women but may be at risk for chlamydia, particularly if they also have sex with men.
In their view, these latest guidelines appropriately prioritize high-level clinically based data. They pointed, however, to recent progress in understanding the pathogenesis of upper reproductive tract infection in women and the sexual networks behind the current resurgence of STIs in the United States in the failure to manage exposed sex partners.
“Considering these critical advances in the evolution of clinic-based screening guidelines is a work in progress,” they wrote, “the dialogue among basic scientists, clinical trial investigators, and public health professionals to inform the next version of updated USPSTF chlamydia and gonorrhea screening guidelines should start now.”
In the opinion of Jennifer L. Reed, MD, MS, a professor of pediatrics and an emergency medicine physician at Cincinnati Children’s Hospital Medical Center and not involved in the updated statement, the recommendations are very reasonable. “The highest rates of infection occur in females 15-24 years of age, and therefore asymptomatic screening for chlamydia and gonorrhea is imperative at least annually or more often if they are high risk,” she said in an interview.
“I would hope that providers increase their asymptomatic screening as a result of these recommendations and highly consider it in the younger men,” Dr. Reed added. “I see a very high rate of gonorrhea and chlamydia infections.” Her center is studying the implementation of gonorrhea and chlamydia asymptomatic screening for adolescents in the pediatric emergency department, a high-risk patient population that will benefit from STI screening opportunities in nontraditional settings.
This research was funded by the Agency for Healthcare Research and Quality and the Department of Health & Human Services under a contract to support the USPSTF. One statement coauthor reported personal fees from Insmed, Paratek, RedHill, and Spero, as well as grants from Insmed. No other disclosures were reported. Dr. Dionne-Odom reported grants from the National Institutes of Health/National Institute of Child Health and Development. Dr. Reed reported a grant from NIH/NICHD for a pragmatic trial of improving STI detection in the pediatric ED.
The U.S. Preventive Services Task Force has updated its 2014 statement on screening asymptomatic individuals for chlamydia and gonorrhea infection.
Published online in JAMA, the 2021 version recommends that all sexually active women aged 24 years or younger and at-risk women 25 years or older should be screened for chlamydia and gonorrhea.
As in 2014, the task force made no screening recommendation for men owing to inconclusive evidence of benefit.
With cases of sexually transmitted infections reaching all-time highs, Amy G. Cantor, MD, MPH, of the Pacific Northwest Evidence-based Practice Center at Oregon Health & Science University, Portland, and colleagues noted that chlamydia and gonorrhea are among the most common STIs in this country. According to the Centers for Disease Control and Prevention, 2019 saw approximately 1.8 million reported cases of chlamydia and more than 600,000 of gonorrhea.
In the current analysis of 27 observational and randomized studies comprising 179,515 patients, the USPSTF panel found that, compared with no screening, chlamydia screening was significantly associated with a reduced risk of pelvic inflammatory disease (PID) in young women in 2 out of 4 trials.
The authors cautioned, however, that the magnitude of benefit was relatively small. No studies reported on screening effectiveness in men, except for one reporting rates of epididymitis, and no studies were done on pregnant women for any outcome.
The largest and newest study, the Australian Chlamydia Control Effectiveness Pilot trial of 2018, assessed chlamydia screening against usual care in 180,355 men and women aged 16-29 years in 130 rural Australian primary care clinics. Screening was associated with a reduced risk of hospital-diagnosed PID: the absolute risk was 0.24% for screening versus 0.38% for usual care (unadjusted risk ratio, 0.6; 95% confidence interval, 0.4-1.0). It was not, however, significantly associated with a reduced risk of clinic-diagnosed PID, with an absolute risk of 0.45% versus 0.39% (RR, 1.1; 95% CI, 0.7-18). Nor did it correlate with a risk reduction for clinic-diagnosed epididymitis: 0.26% vs. 0.27% (RR, 0.9; 95% CI, 0.6-1.4).
While risk prediction criteria apart from age were only minimally accurate, testing for asymptomatic chlamydial and gonococcal infections was highly accurate at most anatomical sites, including urine and self-collected specimens, the investigators observed. Age 22 years or younger alone versus multi-item risk criteria demonstrated similar discrimination in a study that included symptomatic and asymptomatic women.
Sensitivity of chlamydial testing was similar at endocervical (89%-100%) and self- and clinician-collected vaginal (90%-100%) sites for women and at meatal (100%), urethral (99%), and rectal (92%) sites for men. It was lower, however, at pharyngeal sites (69.2%) for men who have sex with men (MSM).
Sensitivity of gonococcal testing was 89% or greater for all anatomical samples. False-positive and false-negative testing rates were low across anatomical sites and collection methods.
“Effectiveness of screening in men and during pregnancy, optimal screening intervals, and adverse effects of screening require further evaluation, Dr. Cantor and associates concluded.
In an accompanying editorial, Jeanne Marrazzo, MD, MPH, and Jodie Dionne-Odom, MD, MSPH, of the division of infectious diseases at the University of Alabama at Birmingham, called the guidelines “timely” and “powerful agents of change” that “influence a wide spectrum of health-based metrics, from quality assurance measures to criteria for financial reimbursement.”
They pointed out that men who have sex with men are experiencing historically high rates of gonorrhea, with most infections occurring extragenitally at the pharynx or rectum. In 2019 CDC data, MSM had substantially higher rates of gonorrhea than men who had sex only with women. They recommended that guidelines for men consider STI risk because of sexual relations with men, women, or both.
“Comprehensive screening guidelines for common STIs like chlamydia and gonorrhea could incorporate the limited evidence base for MSM, whether it is regular practice or not,” they wrote, with the same approach for women who have sex with women but may be at risk for chlamydia, particularly if they also have sex with men.
In their view, these latest guidelines appropriately prioritize high-level clinically based data. They pointed, however, to recent progress in understanding the pathogenesis of upper reproductive tract infection in women and the sexual networks behind the current resurgence of STIs in the United States in the failure to manage exposed sex partners.
“Considering these critical advances in the evolution of clinic-based screening guidelines is a work in progress,” they wrote, “the dialogue among basic scientists, clinical trial investigators, and public health professionals to inform the next version of updated USPSTF chlamydia and gonorrhea screening guidelines should start now.”
In the opinion of Jennifer L. Reed, MD, MS, a professor of pediatrics and an emergency medicine physician at Cincinnati Children’s Hospital Medical Center and not involved in the updated statement, the recommendations are very reasonable. “The highest rates of infection occur in females 15-24 years of age, and therefore asymptomatic screening for chlamydia and gonorrhea is imperative at least annually or more often if they are high risk,” she said in an interview.
“I would hope that providers increase their asymptomatic screening as a result of these recommendations and highly consider it in the younger men,” Dr. Reed added. “I see a very high rate of gonorrhea and chlamydia infections.” Her center is studying the implementation of gonorrhea and chlamydia asymptomatic screening for adolescents in the pediatric emergency department, a high-risk patient population that will benefit from STI screening opportunities in nontraditional settings.
This research was funded by the Agency for Healthcare Research and Quality and the Department of Health & Human Services under a contract to support the USPSTF. One statement coauthor reported personal fees from Insmed, Paratek, RedHill, and Spero, as well as grants from Insmed. No other disclosures were reported. Dr. Dionne-Odom reported grants from the National Institutes of Health/National Institute of Child Health and Development. Dr. Reed reported a grant from NIH/NICHD for a pragmatic trial of improving STI detection in the pediatric ED.
FROM JAMA