User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
High cholesterol in seniors: Use statins for primary prevention?
LONG BEACH, CALIF. – For years, clinicians have debated whether prescribing statins to patients older than 75 for the prevention of cardiovascular events is appropriate.
In 2022, the U.S. Preventive Services Task Force concluded that scientific evidence was insufficient to assess the balance between the benefits and harms of the therapy for this older population.
At a session of the annual meeting of the American Geriatrics Society, experts laid out new preliminary recommendations of the AGS and the National Lipid Association on assessing risk and deciding on treatment.
The group concluded that LDL cholesterol levels are associated with incident arteriosclerotic cardiovascular disease (ASCVD), that the coronary artery calcium (CAC) score can be a valuable measure, and that statins may be reasonable to prescribe, even given the risks that have been linked to statins, such as that for muscle pain. Final recommendations are expected by fall 2023.
“This is still a work in progress,” said Daniel E. Forman, MD, professor of medicine and chair of geriatric cardiology at the University of Pittsburgh.
The AGS-NLA panel concluded that, for those aged 75 or older without established ASCVD, LDL cholesterol is associated with incident ASCVD, the only recommendation to be given a class I (strong) rating; others were classed as moderate or weak.
Dr. Forman reviewed the evidence for lowering LDL cholesterol to decrease ASCVD, citing a 2018 study that concluded, “Reverse causation may contribute to the association of lower TC with higher mortality in nonrandomized studies.”
However, research overall shows that, as LDL cholesterol levels increase, patients are more likely to experience a heart event.
Dr. Forman noted that the utility of equations for assessing 5- or 10-year risk of ASCVD is uncertain. However, he said, traditional risk factors, such as family history and ethnicity, still have value.
Assessing risk “has been enriched in the past few years by the introduction of the coronary artery calcium [CAC] score,” he said.
Lower scores predict lower rates of CVD events, Forman said. The AGS-NLA recommends measuring CAC if clinical uncertainty exists about the value of statins.
“It’s reasonable to measure CAC and to withhold statins when the CAC is zero,” Dr. Forman said. “When the CAC score is zero ... the risk of having a cardiovascular event is really next to nil. Patients are happy to know they have a CAC of zero.”
Likewise, patients appreciate knowing whether their score is high, which would indicate increased risk. He said the CAC score is underused by geriatric physicians.
The group also determined, after reviewing the research, that starting treatment is reasonable for patients with an LDL cholesterol level of 70-189 if they have no life-limiting illness and their life expectancy is over 5 years.
Other preliminary recommendations include the use of statins for those aged 75 and older, irrespective of risk for statin-associated muscle symptoms, type 2 diabetes, or impaired cognition. These associations are often weak, Dr. Forman added.
Focusing on person-centered decisions
Ariel Green, MD, MPH, PhD, associate professor of medicine at Johns Hopkins University, Baltimore, said statin therapy “should be individualized” to weigh benefits, noncardiac risks, and other considerations.
Clinicians can incorporate life expectancy into prevention decisions using tools such as ePrognosis, from the University of California, San Francisco, Dr. Green said.
If life expectancy is greater than the time to benefit, statin therapy may help. Dr. Green cited research that showed that 2.5 years of statin therapy was needed to prevent one major adverse cardiovascular event (MACE) per 100 patients in a population aged 50-75. Other data show reductions in MACE for those older than 75, but overall, the data are limited in this population.
The proposed recommendation is to use tools such as life tables that include comorbid conditions and functional status to guide clinical decisions.
“Another aspect of assessing net benefits of statin therapy is to consider competing health risks,” Dr. Green said.
The group recommends considering using competing risk-adjusted CVD models, though these are not widely used.
The group also recommends integrating screenings for frailty (Clinical Frailty Scale), dementia (Mini-Cog), and functional status (Vulnerable Elders Scale–13) into assessments.
“The presence of these syndromes should prompt elicitation of patient values and preferences related to prevention and medication use,” Dr. Green said.
Clinicians can use decision aids, but these are not always practical, owing to obstacles such as patients’ cognitive problems, Dr. Green said.
“Another approach is asking people to prioritize a set of universal health outcomes that apply across health conditions, such as maintaining independence, staying alive, reducing, or eliminating symptoms and focusing on comfort,” Dr. Green said.
She addressed the evidence about deprescribing statins, with a focus on those with a life expectancy of less than a year. Researchers have found an increase in quality of life and no increases in cardiovascular events or death when statins were deprescribed.
A welcome framework
Cory Krueger, MD, an internal medicine and geriatric physician in Cornville, Ariz., who attended the talk, said he welcomed the presentation, in which preliminary recommendations were explained.
“This has been a controversial area in geriatrics,” Dr. Krueger said. “At least this gave me a framework for discussing this with my patients in a reasonable way.”
Dr. Forman and Dr. Krueger disclosed no relevant financial relationships. Dr. Green receives funding from the National Institute of Aging and Impact Collaboratory.
A version of this article first appeared on Medscape.com.
LONG BEACH, CALIF. – For years, clinicians have debated whether prescribing statins to patients older than 75 for the prevention of cardiovascular events is appropriate.
In 2022, the U.S. Preventive Services Task Force concluded that scientific evidence was insufficient to assess the balance between the benefits and harms of the therapy for this older population.
At a session of the annual meeting of the American Geriatrics Society, experts laid out new preliminary recommendations of the AGS and the National Lipid Association on assessing risk and deciding on treatment.
The group concluded that LDL cholesterol levels are associated with incident arteriosclerotic cardiovascular disease (ASCVD), that the coronary artery calcium (CAC) score can be a valuable measure, and that statins may be reasonable to prescribe, even given the risks that have been linked to statins, such as that for muscle pain. Final recommendations are expected by fall 2023.
“This is still a work in progress,” said Daniel E. Forman, MD, professor of medicine and chair of geriatric cardiology at the University of Pittsburgh.
The AGS-NLA panel concluded that, for those aged 75 or older without established ASCVD, LDL cholesterol is associated with incident ASCVD, the only recommendation to be given a class I (strong) rating; others were classed as moderate or weak.
Dr. Forman reviewed the evidence for lowering LDL cholesterol to decrease ASCVD, citing a 2018 study that concluded, “Reverse causation may contribute to the association of lower TC with higher mortality in nonrandomized studies.”
However, research overall shows that, as LDL cholesterol levels increase, patients are more likely to experience a heart event.
Dr. Forman noted that the utility of equations for assessing 5- or 10-year risk of ASCVD is uncertain. However, he said, traditional risk factors, such as family history and ethnicity, still have value.
Assessing risk “has been enriched in the past few years by the introduction of the coronary artery calcium [CAC] score,” he said.
Lower scores predict lower rates of CVD events, Forman said. The AGS-NLA recommends measuring CAC if clinical uncertainty exists about the value of statins.
“It’s reasonable to measure CAC and to withhold statins when the CAC is zero,” Dr. Forman said. “When the CAC score is zero ... the risk of having a cardiovascular event is really next to nil. Patients are happy to know they have a CAC of zero.”
Likewise, patients appreciate knowing whether their score is high, which would indicate increased risk. He said the CAC score is underused by geriatric physicians.
The group also determined, after reviewing the research, that starting treatment is reasonable for patients with an LDL cholesterol level of 70-189 if they have no life-limiting illness and their life expectancy is over 5 years.
Other preliminary recommendations include the use of statins for those aged 75 and older, irrespective of risk for statin-associated muscle symptoms, type 2 diabetes, or impaired cognition. These associations are often weak, Dr. Forman added.
Focusing on person-centered decisions
Ariel Green, MD, MPH, PhD, associate professor of medicine at Johns Hopkins University, Baltimore, said statin therapy “should be individualized” to weigh benefits, noncardiac risks, and other considerations.
Clinicians can incorporate life expectancy into prevention decisions using tools such as ePrognosis, from the University of California, San Francisco, Dr. Green said.
If life expectancy is greater than the time to benefit, statin therapy may help. Dr. Green cited research that showed that 2.5 years of statin therapy was needed to prevent one major adverse cardiovascular event (MACE) per 100 patients in a population aged 50-75. Other data show reductions in MACE for those older than 75, but overall, the data are limited in this population.
The proposed recommendation is to use tools such as life tables that include comorbid conditions and functional status to guide clinical decisions.
“Another aspect of assessing net benefits of statin therapy is to consider competing health risks,” Dr. Green said.
The group recommends considering using competing risk-adjusted CVD models, though these are not widely used.
The group also recommends integrating screenings for frailty (Clinical Frailty Scale), dementia (Mini-Cog), and functional status (Vulnerable Elders Scale–13) into assessments.
“The presence of these syndromes should prompt elicitation of patient values and preferences related to prevention and medication use,” Dr. Green said.
Clinicians can use decision aids, but these are not always practical, owing to obstacles such as patients’ cognitive problems, Dr. Green said.
“Another approach is asking people to prioritize a set of universal health outcomes that apply across health conditions, such as maintaining independence, staying alive, reducing, or eliminating symptoms and focusing on comfort,” Dr. Green said.
She addressed the evidence about deprescribing statins, with a focus on those with a life expectancy of less than a year. Researchers have found an increase in quality of life and no increases in cardiovascular events or death when statins were deprescribed.
A welcome framework
Cory Krueger, MD, an internal medicine and geriatric physician in Cornville, Ariz., who attended the talk, said he welcomed the presentation, in which preliminary recommendations were explained.
“This has been a controversial area in geriatrics,” Dr. Krueger said. “At least this gave me a framework for discussing this with my patients in a reasonable way.”
Dr. Forman and Dr. Krueger disclosed no relevant financial relationships. Dr. Green receives funding from the National Institute of Aging and Impact Collaboratory.
A version of this article first appeared on Medscape.com.
LONG BEACH, CALIF. – For years, clinicians have debated whether prescribing statins to patients older than 75 for the prevention of cardiovascular events is appropriate.
In 2022, the U.S. Preventive Services Task Force concluded that scientific evidence was insufficient to assess the balance between the benefits and harms of the therapy for this older population.
At a session of the annual meeting of the American Geriatrics Society, experts laid out new preliminary recommendations of the AGS and the National Lipid Association on assessing risk and deciding on treatment.
The group concluded that LDL cholesterol levels are associated with incident arteriosclerotic cardiovascular disease (ASCVD), that the coronary artery calcium (CAC) score can be a valuable measure, and that statins may be reasonable to prescribe, even given the risks that have been linked to statins, such as that for muscle pain. Final recommendations are expected by fall 2023.
“This is still a work in progress,” said Daniel E. Forman, MD, professor of medicine and chair of geriatric cardiology at the University of Pittsburgh.
The AGS-NLA panel concluded that, for those aged 75 or older without established ASCVD, LDL cholesterol is associated with incident ASCVD, the only recommendation to be given a class I (strong) rating; others were classed as moderate or weak.
Dr. Forman reviewed the evidence for lowering LDL cholesterol to decrease ASCVD, citing a 2018 study that concluded, “Reverse causation may contribute to the association of lower TC with higher mortality in nonrandomized studies.”
However, research overall shows that, as LDL cholesterol levels increase, patients are more likely to experience a heart event.
Dr. Forman noted that the utility of equations for assessing 5- or 10-year risk of ASCVD is uncertain. However, he said, traditional risk factors, such as family history and ethnicity, still have value.
Assessing risk “has been enriched in the past few years by the introduction of the coronary artery calcium [CAC] score,” he said.
Lower scores predict lower rates of CVD events, Forman said. The AGS-NLA recommends measuring CAC if clinical uncertainty exists about the value of statins.
“It’s reasonable to measure CAC and to withhold statins when the CAC is zero,” Dr. Forman said. “When the CAC score is zero ... the risk of having a cardiovascular event is really next to nil. Patients are happy to know they have a CAC of zero.”
Likewise, patients appreciate knowing whether their score is high, which would indicate increased risk. He said the CAC score is underused by geriatric physicians.
The group also determined, after reviewing the research, that starting treatment is reasonable for patients with an LDL cholesterol level of 70-189 if they have no life-limiting illness and their life expectancy is over 5 years.
Other preliminary recommendations include the use of statins for those aged 75 and older, irrespective of risk for statin-associated muscle symptoms, type 2 diabetes, or impaired cognition. These associations are often weak, Dr. Forman added.
Focusing on person-centered decisions
Ariel Green, MD, MPH, PhD, associate professor of medicine at Johns Hopkins University, Baltimore, said statin therapy “should be individualized” to weigh benefits, noncardiac risks, and other considerations.
Clinicians can incorporate life expectancy into prevention decisions using tools such as ePrognosis, from the University of California, San Francisco, Dr. Green said.
If life expectancy is greater than the time to benefit, statin therapy may help. Dr. Green cited research that showed that 2.5 years of statin therapy was needed to prevent one major adverse cardiovascular event (MACE) per 100 patients in a population aged 50-75. Other data show reductions in MACE for those older than 75, but overall, the data are limited in this population.
The proposed recommendation is to use tools such as life tables that include comorbid conditions and functional status to guide clinical decisions.
“Another aspect of assessing net benefits of statin therapy is to consider competing health risks,” Dr. Green said.
The group recommends considering using competing risk-adjusted CVD models, though these are not widely used.
The group also recommends integrating screenings for frailty (Clinical Frailty Scale), dementia (Mini-Cog), and functional status (Vulnerable Elders Scale–13) into assessments.
“The presence of these syndromes should prompt elicitation of patient values and preferences related to prevention and medication use,” Dr. Green said.
Clinicians can use decision aids, but these are not always practical, owing to obstacles such as patients’ cognitive problems, Dr. Green said.
“Another approach is asking people to prioritize a set of universal health outcomes that apply across health conditions, such as maintaining independence, staying alive, reducing, or eliminating symptoms and focusing on comfort,” Dr. Green said.
She addressed the evidence about deprescribing statins, with a focus on those with a life expectancy of less than a year. Researchers have found an increase in quality of life and no increases in cardiovascular events or death when statins were deprescribed.
A welcome framework
Cory Krueger, MD, an internal medicine and geriatric physician in Cornville, Ariz., who attended the talk, said he welcomed the presentation, in which preliminary recommendations were explained.
“This has been a controversial area in geriatrics,” Dr. Krueger said. “At least this gave me a framework for discussing this with my patients in a reasonable way.”
Dr. Forman and Dr. Krueger disclosed no relevant financial relationships. Dr. Green receives funding from the National Institute of Aging and Impact Collaboratory.
A version of this article first appeared on Medscape.com.
AT AGS 2023
Medical students gain momentum in effort to ban legacy admissions
, which they say offer preferential treatment to applicants based on their association with donors or alumni.
While an estimated 25% of public colleges and universities still use legacy admissions, a growing list of top medical schools have moved away from the practice over the last decade, including Johns Hopkins University, Baltimore, and Tufts University, Medford, Mass.
Legacy admissions contradict schools’ more inclusive policies, Senila Yasmin, MPH, a second-year medical student at Tufts University, said in an interview. While Tufts maintains legacy admissions for its undergraduate applicants, the medical school stopped the practice in 2021, said Ms. Yasmin, a member of a student group that lobbied against the school’s legacy preferences.
Describing herself as a low-income, first-generation Muslim-Pakistani American, Ms. Yasmin wants to use her experience at Tufts to improve accessibility for students like herself.
As a member of the American Medical Association (AMA) Medical Student Section, she coauthored a resolution stating that legacy admissions go against the AMA’s strategic plan to advance racial justice and health equity. The Student Section passed the resolution in November, and in June, the AMA House of Delegates will vote on whether to adopt the policy.
Along with a Supreme Court decision that could strike down race-conscious college admissions, an AMA policy could convince medical schools to rethink legacy admissions and how to maintain diverse student bodies. In June, the court is expected to issue a decision in the Students for Fair Admissions lawsuit against Harvard University, Cambridge, Mass., and the University of North Carolina, Chapel Hill, which alleges that considering race in holistic admissions constitutes racial discrimination and violates the Equal Protection Clause.
Opponents of legacy admissions, like Ms. Yasmin, say it penalizes students from racial minorities and lower socioeconomic backgrounds, hampering a fair and equitable admissions process that attracts diverse medical school admissions.
Diversity of medical applicants
Diversity in medical schools continued to increase last year with more Black, Hispanic, and female students applying and enrolling, according to a recent report by the Association of American Medical Colleges (AAMC). However, universities often include nonacademic criteria in their admission assessments to improve educational access for underrepresented minorities.
Medical schools carefully consider each applicant’s background “to yield a diverse class of students,” Geoffrey Young, PhD, AAMC’s senior director of transforming the health care workforce, told this news organization.
Some schools, such as Morehouse School of Medicine, Atlanta, the University of Virginia School of Medicine, Charlottesville, and the University of Arizona College of Medicine, Tucson, perform a thorough review of candidates while offering admissions practices designed specifically for legacy applicants. The schools assert that legacy designation doesn’t factor into the student’s likelihood of acceptance.
The arrangement may show that schools want to commit to equity and fairness but have trouble moving away from entrenched traditions, two professors from Penn State College of Medicine, Hershey, Pa., who sit on separate medical admissions subcommittees, wrote last year in Bioethics Today.
Legislation may hasten legacies’ end
In December, Ms. Yasmin and a group of Massachusetts Medical Society student-members presented another resolution to the state medical society, which adopted it.
The society’s new policy opposes the use of legacy status in medical school admissions and supports mechanisms to eliminate its inclusion from the application process, Theodore Calianos II, MD, FACS, president of the Massachusetts Medical Society, said in an interview.
“Legacy preferences limit racial and socioeconomic diversity on campuses, so we asked, ‘What can we do so that everyone has equal access to medical education?’ It is exciting to see the students and young physicians – the future of medicine – become involved in policymaking.”
Proposed laws may also hasten the end of legacy admissions. Last year, the U.S. Senate began considering a bill prohibiting colleges receiving federal financial aid from giving preferential treatment to students based on their relations to donors or alumni. However, the bill allows the Department of Education to make exceptions for institutions serving historically underrepresented groups.
The New York State Senate and the New York State Assembly also are reviewing bills that ban legacy and early admissions policies at public and private universities. Connecticut announced similar legislation last year. Massachusetts legislators are considering two bills: one that would ban the practice at the state’s public universities and another that would require all schools using legacy status to pay a “public service fee” equal to a percentage of its endowment. Colleges with endowment assets exceeding $2 billion must pay at least $2 million, according to the bill’s text.
At schools like Harvard, whose endowment surpasses $50 billion, the option to pay the penalty will make the law moot, Michael Walls, DO, MPH, president of the American Medical Student Association (AMSA), said in an interview. “Smaller schools wouldn’t be able to afford the fine and are less likely to be doing [legacy admissions] anyway,” he said. “The schools that want to continue doing it could just pay the fine.”
Dr. Walls said AMSA supports race-conscious admissions processes and anything that increases fairness for medical school applicants. “Whatever [fair] means is up for interpretation, but it would be great to eliminate legacy admissions,” he said.
A version of this article originally appeared on Medscape.com.
, which they say offer preferential treatment to applicants based on their association with donors or alumni.
While an estimated 25% of public colleges and universities still use legacy admissions, a growing list of top medical schools have moved away from the practice over the last decade, including Johns Hopkins University, Baltimore, and Tufts University, Medford, Mass.
Legacy admissions contradict schools’ more inclusive policies, Senila Yasmin, MPH, a second-year medical student at Tufts University, said in an interview. While Tufts maintains legacy admissions for its undergraduate applicants, the medical school stopped the practice in 2021, said Ms. Yasmin, a member of a student group that lobbied against the school’s legacy preferences.
Describing herself as a low-income, first-generation Muslim-Pakistani American, Ms. Yasmin wants to use her experience at Tufts to improve accessibility for students like herself.
As a member of the American Medical Association (AMA) Medical Student Section, she coauthored a resolution stating that legacy admissions go against the AMA’s strategic plan to advance racial justice and health equity. The Student Section passed the resolution in November, and in June, the AMA House of Delegates will vote on whether to adopt the policy.
Along with a Supreme Court decision that could strike down race-conscious college admissions, an AMA policy could convince medical schools to rethink legacy admissions and how to maintain diverse student bodies. In June, the court is expected to issue a decision in the Students for Fair Admissions lawsuit against Harvard University, Cambridge, Mass., and the University of North Carolina, Chapel Hill, which alleges that considering race in holistic admissions constitutes racial discrimination and violates the Equal Protection Clause.
Opponents of legacy admissions, like Ms. Yasmin, say it penalizes students from racial minorities and lower socioeconomic backgrounds, hampering a fair and equitable admissions process that attracts diverse medical school admissions.
Diversity of medical applicants
Diversity in medical schools continued to increase last year with more Black, Hispanic, and female students applying and enrolling, according to a recent report by the Association of American Medical Colleges (AAMC). However, universities often include nonacademic criteria in their admission assessments to improve educational access for underrepresented minorities.
Medical schools carefully consider each applicant’s background “to yield a diverse class of students,” Geoffrey Young, PhD, AAMC’s senior director of transforming the health care workforce, told this news organization.
Some schools, such as Morehouse School of Medicine, Atlanta, the University of Virginia School of Medicine, Charlottesville, and the University of Arizona College of Medicine, Tucson, perform a thorough review of candidates while offering admissions practices designed specifically for legacy applicants. The schools assert that legacy designation doesn’t factor into the student’s likelihood of acceptance.
The arrangement may show that schools want to commit to equity and fairness but have trouble moving away from entrenched traditions, two professors from Penn State College of Medicine, Hershey, Pa., who sit on separate medical admissions subcommittees, wrote last year in Bioethics Today.
Legislation may hasten legacies’ end
In December, Ms. Yasmin and a group of Massachusetts Medical Society student-members presented another resolution to the state medical society, which adopted it.
The society’s new policy opposes the use of legacy status in medical school admissions and supports mechanisms to eliminate its inclusion from the application process, Theodore Calianos II, MD, FACS, president of the Massachusetts Medical Society, said in an interview.
“Legacy preferences limit racial and socioeconomic diversity on campuses, so we asked, ‘What can we do so that everyone has equal access to medical education?’ It is exciting to see the students and young physicians – the future of medicine – become involved in policymaking.”
Proposed laws may also hasten the end of legacy admissions. Last year, the U.S. Senate began considering a bill prohibiting colleges receiving federal financial aid from giving preferential treatment to students based on their relations to donors or alumni. However, the bill allows the Department of Education to make exceptions for institutions serving historically underrepresented groups.
The New York State Senate and the New York State Assembly also are reviewing bills that ban legacy and early admissions policies at public and private universities. Connecticut announced similar legislation last year. Massachusetts legislators are considering two bills: one that would ban the practice at the state’s public universities and another that would require all schools using legacy status to pay a “public service fee” equal to a percentage of its endowment. Colleges with endowment assets exceeding $2 billion must pay at least $2 million, according to the bill’s text.
At schools like Harvard, whose endowment surpasses $50 billion, the option to pay the penalty will make the law moot, Michael Walls, DO, MPH, president of the American Medical Student Association (AMSA), said in an interview. “Smaller schools wouldn’t be able to afford the fine and are less likely to be doing [legacy admissions] anyway,” he said. “The schools that want to continue doing it could just pay the fine.”
Dr. Walls said AMSA supports race-conscious admissions processes and anything that increases fairness for medical school applicants. “Whatever [fair] means is up for interpretation, but it would be great to eliminate legacy admissions,” he said.
A version of this article originally appeared on Medscape.com.
, which they say offer preferential treatment to applicants based on their association with donors or alumni.
While an estimated 25% of public colleges and universities still use legacy admissions, a growing list of top medical schools have moved away from the practice over the last decade, including Johns Hopkins University, Baltimore, and Tufts University, Medford, Mass.
Legacy admissions contradict schools’ more inclusive policies, Senila Yasmin, MPH, a second-year medical student at Tufts University, said in an interview. While Tufts maintains legacy admissions for its undergraduate applicants, the medical school stopped the practice in 2021, said Ms. Yasmin, a member of a student group that lobbied against the school’s legacy preferences.
Describing herself as a low-income, first-generation Muslim-Pakistani American, Ms. Yasmin wants to use her experience at Tufts to improve accessibility for students like herself.
As a member of the American Medical Association (AMA) Medical Student Section, she coauthored a resolution stating that legacy admissions go against the AMA’s strategic plan to advance racial justice and health equity. The Student Section passed the resolution in November, and in June, the AMA House of Delegates will vote on whether to adopt the policy.
Along with a Supreme Court decision that could strike down race-conscious college admissions, an AMA policy could convince medical schools to rethink legacy admissions and how to maintain diverse student bodies. In June, the court is expected to issue a decision in the Students for Fair Admissions lawsuit against Harvard University, Cambridge, Mass., and the University of North Carolina, Chapel Hill, which alleges that considering race in holistic admissions constitutes racial discrimination and violates the Equal Protection Clause.
Opponents of legacy admissions, like Ms. Yasmin, say it penalizes students from racial minorities and lower socioeconomic backgrounds, hampering a fair and equitable admissions process that attracts diverse medical school admissions.
Diversity of medical applicants
Diversity in medical schools continued to increase last year with more Black, Hispanic, and female students applying and enrolling, according to a recent report by the Association of American Medical Colleges (AAMC). However, universities often include nonacademic criteria in their admission assessments to improve educational access for underrepresented minorities.
Medical schools carefully consider each applicant’s background “to yield a diverse class of students,” Geoffrey Young, PhD, AAMC’s senior director of transforming the health care workforce, told this news organization.
Some schools, such as Morehouse School of Medicine, Atlanta, the University of Virginia School of Medicine, Charlottesville, and the University of Arizona College of Medicine, Tucson, perform a thorough review of candidates while offering admissions practices designed specifically for legacy applicants. The schools assert that legacy designation doesn’t factor into the student’s likelihood of acceptance.
The arrangement may show that schools want to commit to equity and fairness but have trouble moving away from entrenched traditions, two professors from Penn State College of Medicine, Hershey, Pa., who sit on separate medical admissions subcommittees, wrote last year in Bioethics Today.
Legislation may hasten legacies’ end
In December, Ms. Yasmin and a group of Massachusetts Medical Society student-members presented another resolution to the state medical society, which adopted it.
The society’s new policy opposes the use of legacy status in medical school admissions and supports mechanisms to eliminate its inclusion from the application process, Theodore Calianos II, MD, FACS, president of the Massachusetts Medical Society, said in an interview.
“Legacy preferences limit racial and socioeconomic diversity on campuses, so we asked, ‘What can we do so that everyone has equal access to medical education?’ It is exciting to see the students and young physicians – the future of medicine – become involved in policymaking.”
Proposed laws may also hasten the end of legacy admissions. Last year, the U.S. Senate began considering a bill prohibiting colleges receiving federal financial aid from giving preferential treatment to students based on their relations to donors or alumni. However, the bill allows the Department of Education to make exceptions for institutions serving historically underrepresented groups.
The New York State Senate and the New York State Assembly also are reviewing bills that ban legacy and early admissions policies at public and private universities. Connecticut announced similar legislation last year. Massachusetts legislators are considering two bills: one that would ban the practice at the state’s public universities and another that would require all schools using legacy status to pay a “public service fee” equal to a percentage of its endowment. Colleges with endowment assets exceeding $2 billion must pay at least $2 million, according to the bill’s text.
At schools like Harvard, whose endowment surpasses $50 billion, the option to pay the penalty will make the law moot, Michael Walls, DO, MPH, president of the American Medical Student Association (AMSA), said in an interview. “Smaller schools wouldn’t be able to afford the fine and are less likely to be doing [legacy admissions] anyway,” he said. “The schools that want to continue doing it could just pay the fine.”
Dr. Walls said AMSA supports race-conscious admissions processes and anything that increases fairness for medical school applicants. “Whatever [fair] means is up for interpretation, but it would be great to eliminate legacy admissions,” he said.
A version of this article originally appeared on Medscape.com.
Five ways docs may qualify for discounts on medical malpractice premiums
Getting a better deal might simply mean taking advantage of incentives and discounts your insurer may already offer. These include claims-free, new-to-practice, and working part-time discounts.
However, if you decide to shop around, keep in mind that discounts are just one factor that can affect your premium price – insurers look at your specialty, location, and claims history.
One of the most common ways physicians can earn discounts is by participating in risk management programs. With this type of program, physicians evaluate elements of their practice and documentation practices and identify areas that might leave them at risk for a lawsuit. While they save money, physician risk management programs also are designed to reduce malpractice claims, which ultimately minimizes the potential for bigger financial losses, insurance experts say.
“It’s a win-win situation when liability insurers and physicians work together to minimize risk, and it’s a win for patients,” said Gary Price, MD, president of The Physicians Foundation.
Doctors in private practice or employed by small hospitals that are not self-insured can qualify for these discounts, said David Zetter, president of Zetter HealthCare Management Consultants.
“I do a lot of work with medical malpractice companies trying to find clients policies. All the carriers are transparent about what physicians have to do to lower their premiums. Physicians can receive the discounts if they follow through and meet the insurer’s requirements,” said Mr. Zetter.
State insurance departments regulate medical malpractice insurance, including the premium credits insurers offer. Most states cap discounts at 25%, but some go as high as 70%, according to The Doctors Company, a national physician-owned medical malpractice insurer.
Insurers typically offer doctors several ways to earn discounts. The size of the discount also can depend on whether a doctor is new to a practice, remains claims free, or takes risk management courses.
In addition to the premium discount, some online risk management classes and webinars are eligible for CME credits.
“The credits can add up and they can be used for recertification or relicensure,” said Susan Boisvert, senior patient safety risk manager at The Doctors Company.
Here are five ways you may qualify for discounts with your insurer.
1. Make use of discounts available to new doctors
Doctors can earn hefty discounts on their premiums when they are no longer interns or residents and start practicing medicine. The Doctors Company usually gives a 50% discount on member premiums the first year they’re in practice and a 25% discount credit in their second year. The discounts end after that.
Other insurance carriers offer similar discounts to doctors starting to practice medicine. The deepest one is offered in the first year (at least 50%) and a smaller one (20%-25%) the second year, according to medical malpractice brokers.
“The new-to-practice discount is based solely on when the physician left their formal training to begin their practice for the first time; it is not based on claim-free history,” explained Mr. Zetter.
This is a very common discount used by different insurer carriers, said Dr. Price. “New physicians don’t have the same amount of risk of a lawsuit when they’re starting out. It’s unlikely they will have a claim and most liability actions have a 2-year time limit from the date of injury to be filed.”
2. Take advantage of being claims free
If you’ve been claims free for at least a few years, you may be eligible for a large discount.
“Doctors without claims are a better risk. Once a doctor has one claim, they’re likely to have a second, which the research shows,” said Mr. Zetter.
The most common credit The Doctors Company offers is 3 years of being claim free – this earns doctors up to 25%, he said. Mr. Zetter explained that the criteria and size of The Doctors Company credit may depend on the state where physicians practice.
“We allowed insurance carriers that we acquired to continue with their own claim-free discount program such as Florida’s First Professionals Insurance Company we acquired in 2011,” he said.
Doctors with other medical malpractice insurers may also be eligible for a credit up to 25%. In some instances, they may have to be claims free for 5 or 10 years, say insurance experts.
It pays to shop around before purchasing insurance.
3. If you work part time, make sure your premium reflects that
Physicians who see patients part time can receive up to a 75% discount on their medical liability insurance premiums.
The discounts are based on the hours the physician works per week. The fewer hours worked, the larger the discount. This type of discount does not vary by specialty.
According to The Doctors Company, working 10 hours or less per week may entitle doctors to a 75% discount; working 11-20 hours per week may entitle them to a 50% discount, and working 21-30 hours per week may entitle them to a 25% discount. If you are in this situation, it pays to ask your insurer if there is a discount available to you.
4. Look into your professional medical society insurance company
“I would look at your state medical association [or] state specialty society and talk to your colleagues to learn what premiums they’re paying and about any discounts they’re getting,” advised Mr. Zetter.
Some state medical societies have formed their own liability companies and offer lower premiums to their members because “they’re organized and managed by doctors, which makes their premiums more competitive,” Dr. Price said.
Other state medical societies endorse specific insurance carriers and offer their members a 5% discount for enrolling with them.
5. Enroll in a risk management program
Most insurers offer online educational activities designed to improve patient safety and reduce the risk of a lawsuit. Physicians may be eligible for both premium discounts and CME credits.
Medical Liability Mutual Insurance Company, owned by Berkshire Hathaway, operates in New York and offers physicians a premium discount of up to 5%, CME credit, and maintenance of certification credit for successfully completing its risk management program every other year.
ProAssurance members nationwide can earn 5% in premium discounts if they complete a 2-hour video series called “Back to Basics: Loss Prevention and Navigating Everyday Risks: Using Data to Drive Change.”
They can earn one credit for completing each webinar on topics such as “Medication Management: Minimizing Errors and Improving Safety” and “Opioid Prescribing: Keeping Patients Safe.”
MagMutual offers its insured physicians 1 CME credit for completing their specialty’s risk assessment and courses, which may be applied toward their premium discounts.
The Doctors Company offers its members a 5% premium discount if they complete 4 CME credits. One of its most popular courses is “How To Get Rid of a Difficult Patient.”
“Busy residents like the shorter case studies worth one-quarter credit that they can complete in 15 minutes,” said Ms. Boisvert.
“This is a good bargain from the physician’s standpoint and the fact that risk management education is offered online makes it a lot easier than going to a seminar in person,” said Dr. Price.
A version of this article first appeared on Medscape.com.
Getting a better deal might simply mean taking advantage of incentives and discounts your insurer may already offer. These include claims-free, new-to-practice, and working part-time discounts.
However, if you decide to shop around, keep in mind that discounts are just one factor that can affect your premium price – insurers look at your specialty, location, and claims history.
One of the most common ways physicians can earn discounts is by participating in risk management programs. With this type of program, physicians evaluate elements of their practice and documentation practices and identify areas that might leave them at risk for a lawsuit. While they save money, physician risk management programs also are designed to reduce malpractice claims, which ultimately minimizes the potential for bigger financial losses, insurance experts say.
“It’s a win-win situation when liability insurers and physicians work together to minimize risk, and it’s a win for patients,” said Gary Price, MD, president of The Physicians Foundation.
Doctors in private practice or employed by small hospitals that are not self-insured can qualify for these discounts, said David Zetter, president of Zetter HealthCare Management Consultants.
“I do a lot of work with medical malpractice companies trying to find clients policies. All the carriers are transparent about what physicians have to do to lower their premiums. Physicians can receive the discounts if they follow through and meet the insurer’s requirements,” said Mr. Zetter.
State insurance departments regulate medical malpractice insurance, including the premium credits insurers offer. Most states cap discounts at 25%, but some go as high as 70%, according to The Doctors Company, a national physician-owned medical malpractice insurer.
Insurers typically offer doctors several ways to earn discounts. The size of the discount also can depend on whether a doctor is new to a practice, remains claims free, or takes risk management courses.
In addition to the premium discount, some online risk management classes and webinars are eligible for CME credits.
“The credits can add up and they can be used for recertification or relicensure,” said Susan Boisvert, senior patient safety risk manager at The Doctors Company.
Here are five ways you may qualify for discounts with your insurer.
1. Make use of discounts available to new doctors
Doctors can earn hefty discounts on their premiums when they are no longer interns or residents and start practicing medicine. The Doctors Company usually gives a 50% discount on member premiums the first year they’re in practice and a 25% discount credit in their second year. The discounts end after that.
Other insurance carriers offer similar discounts to doctors starting to practice medicine. The deepest one is offered in the first year (at least 50%) and a smaller one (20%-25%) the second year, according to medical malpractice brokers.
“The new-to-practice discount is based solely on when the physician left their formal training to begin their practice for the first time; it is not based on claim-free history,” explained Mr. Zetter.
This is a very common discount used by different insurer carriers, said Dr. Price. “New physicians don’t have the same amount of risk of a lawsuit when they’re starting out. It’s unlikely they will have a claim and most liability actions have a 2-year time limit from the date of injury to be filed.”
2. Take advantage of being claims free
If you’ve been claims free for at least a few years, you may be eligible for a large discount.
“Doctors without claims are a better risk. Once a doctor has one claim, they’re likely to have a second, which the research shows,” said Mr. Zetter.
The most common credit The Doctors Company offers is 3 years of being claim free – this earns doctors up to 25%, he said. Mr. Zetter explained that the criteria and size of The Doctors Company credit may depend on the state where physicians practice.
“We allowed insurance carriers that we acquired to continue with their own claim-free discount program such as Florida’s First Professionals Insurance Company we acquired in 2011,” he said.
Doctors with other medical malpractice insurers may also be eligible for a credit up to 25%. In some instances, they may have to be claims free for 5 or 10 years, say insurance experts.
It pays to shop around before purchasing insurance.
3. If you work part time, make sure your premium reflects that
Physicians who see patients part time can receive up to a 75% discount on their medical liability insurance premiums.
The discounts are based on the hours the physician works per week. The fewer hours worked, the larger the discount. This type of discount does not vary by specialty.
According to The Doctors Company, working 10 hours or less per week may entitle doctors to a 75% discount; working 11-20 hours per week may entitle them to a 50% discount, and working 21-30 hours per week may entitle them to a 25% discount. If you are in this situation, it pays to ask your insurer if there is a discount available to you.
4. Look into your professional medical society insurance company
“I would look at your state medical association [or] state specialty society and talk to your colleagues to learn what premiums they’re paying and about any discounts they’re getting,” advised Mr. Zetter.
Some state medical societies have formed their own liability companies and offer lower premiums to their members because “they’re organized and managed by doctors, which makes their premiums more competitive,” Dr. Price said.
Other state medical societies endorse specific insurance carriers and offer their members a 5% discount for enrolling with them.
5. Enroll in a risk management program
Most insurers offer online educational activities designed to improve patient safety and reduce the risk of a lawsuit. Physicians may be eligible for both premium discounts and CME credits.
Medical Liability Mutual Insurance Company, owned by Berkshire Hathaway, operates in New York and offers physicians a premium discount of up to 5%, CME credit, and maintenance of certification credit for successfully completing its risk management program every other year.
ProAssurance members nationwide can earn 5% in premium discounts if they complete a 2-hour video series called “Back to Basics: Loss Prevention and Navigating Everyday Risks: Using Data to Drive Change.”
They can earn one credit for completing each webinar on topics such as “Medication Management: Minimizing Errors and Improving Safety” and “Opioid Prescribing: Keeping Patients Safe.”
MagMutual offers its insured physicians 1 CME credit for completing their specialty’s risk assessment and courses, which may be applied toward their premium discounts.
The Doctors Company offers its members a 5% premium discount if they complete 4 CME credits. One of its most popular courses is “How To Get Rid of a Difficult Patient.”
“Busy residents like the shorter case studies worth one-quarter credit that they can complete in 15 minutes,” said Ms. Boisvert.
“This is a good bargain from the physician’s standpoint and the fact that risk management education is offered online makes it a lot easier than going to a seminar in person,” said Dr. Price.
A version of this article first appeared on Medscape.com.
Getting a better deal might simply mean taking advantage of incentives and discounts your insurer may already offer. These include claims-free, new-to-practice, and working part-time discounts.
However, if you decide to shop around, keep in mind that discounts are just one factor that can affect your premium price – insurers look at your specialty, location, and claims history.
One of the most common ways physicians can earn discounts is by participating in risk management programs. With this type of program, physicians evaluate elements of their practice and documentation practices and identify areas that might leave them at risk for a lawsuit. While they save money, physician risk management programs also are designed to reduce malpractice claims, which ultimately minimizes the potential for bigger financial losses, insurance experts say.
“It’s a win-win situation when liability insurers and physicians work together to minimize risk, and it’s a win for patients,” said Gary Price, MD, president of The Physicians Foundation.
Doctors in private practice or employed by small hospitals that are not self-insured can qualify for these discounts, said David Zetter, president of Zetter HealthCare Management Consultants.
“I do a lot of work with medical malpractice companies trying to find clients policies. All the carriers are transparent about what physicians have to do to lower their premiums. Physicians can receive the discounts if they follow through and meet the insurer’s requirements,” said Mr. Zetter.
State insurance departments regulate medical malpractice insurance, including the premium credits insurers offer. Most states cap discounts at 25%, but some go as high as 70%, according to The Doctors Company, a national physician-owned medical malpractice insurer.
Insurers typically offer doctors several ways to earn discounts. The size of the discount also can depend on whether a doctor is new to a practice, remains claims free, or takes risk management courses.
In addition to the premium discount, some online risk management classes and webinars are eligible for CME credits.
“The credits can add up and they can be used for recertification or relicensure,” said Susan Boisvert, senior patient safety risk manager at The Doctors Company.
Here are five ways you may qualify for discounts with your insurer.
1. Make use of discounts available to new doctors
Doctors can earn hefty discounts on their premiums when they are no longer interns or residents and start practicing medicine. The Doctors Company usually gives a 50% discount on member premiums the first year they’re in practice and a 25% discount credit in their second year. The discounts end after that.
Other insurance carriers offer similar discounts to doctors starting to practice medicine. The deepest one is offered in the first year (at least 50%) and a smaller one (20%-25%) the second year, according to medical malpractice brokers.
“The new-to-practice discount is based solely on when the physician left their formal training to begin their practice for the first time; it is not based on claim-free history,” explained Mr. Zetter.
This is a very common discount used by different insurer carriers, said Dr. Price. “New physicians don’t have the same amount of risk of a lawsuit when they’re starting out. It’s unlikely they will have a claim and most liability actions have a 2-year time limit from the date of injury to be filed.”
2. Take advantage of being claims free
If you’ve been claims free for at least a few years, you may be eligible for a large discount.
“Doctors without claims are a better risk. Once a doctor has one claim, they’re likely to have a second, which the research shows,” said Mr. Zetter.
The most common credit The Doctors Company offers is 3 years of being claim free – this earns doctors up to 25%, he said. Mr. Zetter explained that the criteria and size of The Doctors Company credit may depend on the state where physicians practice.
“We allowed insurance carriers that we acquired to continue with their own claim-free discount program such as Florida’s First Professionals Insurance Company we acquired in 2011,” he said.
Doctors with other medical malpractice insurers may also be eligible for a credit up to 25%. In some instances, they may have to be claims free for 5 or 10 years, say insurance experts.
It pays to shop around before purchasing insurance.
3. If you work part time, make sure your premium reflects that
Physicians who see patients part time can receive up to a 75% discount on their medical liability insurance premiums.
The discounts are based on the hours the physician works per week. The fewer hours worked, the larger the discount. This type of discount does not vary by specialty.
According to The Doctors Company, working 10 hours or less per week may entitle doctors to a 75% discount; working 11-20 hours per week may entitle them to a 50% discount, and working 21-30 hours per week may entitle them to a 25% discount. If you are in this situation, it pays to ask your insurer if there is a discount available to you.
4. Look into your professional medical society insurance company
“I would look at your state medical association [or] state specialty society and talk to your colleagues to learn what premiums they’re paying and about any discounts they’re getting,” advised Mr. Zetter.
Some state medical societies have formed their own liability companies and offer lower premiums to their members because “they’re organized and managed by doctors, which makes their premiums more competitive,” Dr. Price said.
Other state medical societies endorse specific insurance carriers and offer their members a 5% discount for enrolling with them.
5. Enroll in a risk management program
Most insurers offer online educational activities designed to improve patient safety and reduce the risk of a lawsuit. Physicians may be eligible for both premium discounts and CME credits.
Medical Liability Mutual Insurance Company, owned by Berkshire Hathaway, operates in New York and offers physicians a premium discount of up to 5%, CME credit, and maintenance of certification credit for successfully completing its risk management program every other year.
ProAssurance members nationwide can earn 5% in premium discounts if they complete a 2-hour video series called “Back to Basics: Loss Prevention and Navigating Everyday Risks: Using Data to Drive Change.”
They can earn one credit for completing each webinar on topics such as “Medication Management: Minimizing Errors and Improving Safety” and “Opioid Prescribing: Keeping Patients Safe.”
MagMutual offers its insured physicians 1 CME credit for completing their specialty’s risk assessment and courses, which may be applied toward their premium discounts.
The Doctors Company offers its members a 5% premium discount if they complete 4 CME credits. One of its most popular courses is “How To Get Rid of a Difficult Patient.”
“Busy residents like the shorter case studies worth one-quarter credit that they can complete in 15 minutes,” said Ms. Boisvert.
“This is a good bargain from the physician’s standpoint and the fact that risk management education is offered online makes it a lot easier than going to a seminar in person,” said Dr. Price.
A version of this article first appeared on Medscape.com.
Bundled strategy increased preteen lipid screening
WASHINGTON – A bundled intervention combining point-of-care testing, electronic medical record support, and provider education significantly improved lipid screening rates in children aged 9-11 years, according to data from approximately 100 monthly visits over a 3-year period.
Guidelines from the National Heart, Lung, and Blood Institute currently recommend universal lipid screening for children aged 9-11 years, but screening rates in clinical practice remain low, according to Ruth E. Gardner, MD, of Penn State University, Hershey, and colleagues.
In a poster presented at the Pediatric Academic Societies annual meeting, Dr. Gardner and colleagues shared results of the implementation of a bundled testing protocol designed to improve screening.
The researchers reviewed data on lipid testing within 30 days for all 9- to 11-year-old well child visits at a single center between May 2019 and February 2022. The bundled intervention was introduced in May 2021.
The bundled protocol included in-office capillary testing and provider education. In addition, electronic medical record templates were modified to include prompts for lipid screening at relevant ages, and EMR orders were adjusted to include lipid testing. The researchers also collected targeted provider feedback on individualized screening rates in February 2022.
Screening rates were plotted monthly. For the period from May 2019 through May 2021, the rates averaged 6.5%. However, after the introduction of the bundled intervention, the rate increased to 29.9%. Following targeted provider feedback in February 2022, the researchers found an additional shift to 52.1% through March and April 2022.
The findings were limited by the use of data from a single center, and the researchers used an extended study period to account for disruptions to well-child care in the spring of 2020 related to the COVID-19 pandemic.
However, the results support the effectiveness of a bundled intervention for improving lipid screening rates in children aged 9-11 years, the researchers said, and targeted provider feedback and education could yield additional improvements, they concluded.
Preteen years are an optimal time for screening
“The current study is important because atherosclerosis begins in childhood, and screening at ages 9-11 is an optimal time to begin lifestyle changes to improve overall health and reduce risks of heart disease,” said Margaret Thew, DNP, FNP-BC, of the Medical College of Wisconsin, Milwaukee, in an interview.
Ms. Thew, who was not involved in the study, said, “The number of recommended and required screening items needed in pediatrics is vast, so many providers have to select which items to focus on for their health screenings with these ages.”
Overall, “I was impressed with the improvements that were made in this quality improvement study,” said Ms. Thew.
Barriers to lipid screening in this population include the reduced number of health screenings and immunizations recommended for this age group; the consequence is that access is limited to discuss preventive care opportunities, said Ms. Thew in an interview. Steps to overcome these barriers could include the use of many of the screening tools introduced in the current study, such as point-of-care testing in the office, use of the EMR to remind providers of testing, which can be done during well visits or school physicals, and educating providers about the current guidelines, she noted.
Other strategies to increase screening include moving the immunization series to provide more frequent appointments to children aged 9-11 years to offer education and preventive care, Ms. Thew added.
The study received no outside funding. The researchers had no financial conflicts to disclose. Ms. Thew had no financial conflicts to disclose and serves on the Editorial Advisory Board of Pediatric News.
WASHINGTON – A bundled intervention combining point-of-care testing, electronic medical record support, and provider education significantly improved lipid screening rates in children aged 9-11 years, according to data from approximately 100 monthly visits over a 3-year period.
Guidelines from the National Heart, Lung, and Blood Institute currently recommend universal lipid screening for children aged 9-11 years, but screening rates in clinical practice remain low, according to Ruth E. Gardner, MD, of Penn State University, Hershey, and colleagues.
In a poster presented at the Pediatric Academic Societies annual meeting, Dr. Gardner and colleagues shared results of the implementation of a bundled testing protocol designed to improve screening.
The researchers reviewed data on lipid testing within 30 days for all 9- to 11-year-old well child visits at a single center between May 2019 and February 2022. The bundled intervention was introduced in May 2021.
The bundled protocol included in-office capillary testing and provider education. In addition, electronic medical record templates were modified to include prompts for lipid screening at relevant ages, and EMR orders were adjusted to include lipid testing. The researchers also collected targeted provider feedback on individualized screening rates in February 2022.
Screening rates were plotted monthly. For the period from May 2019 through May 2021, the rates averaged 6.5%. However, after the introduction of the bundled intervention, the rate increased to 29.9%. Following targeted provider feedback in February 2022, the researchers found an additional shift to 52.1% through March and April 2022.
The findings were limited by the use of data from a single center, and the researchers used an extended study period to account for disruptions to well-child care in the spring of 2020 related to the COVID-19 pandemic.
However, the results support the effectiveness of a bundled intervention for improving lipid screening rates in children aged 9-11 years, the researchers said, and targeted provider feedback and education could yield additional improvements, they concluded.
Preteen years are an optimal time for screening
“The current study is important because atherosclerosis begins in childhood, and screening at ages 9-11 is an optimal time to begin lifestyle changes to improve overall health and reduce risks of heart disease,” said Margaret Thew, DNP, FNP-BC, of the Medical College of Wisconsin, Milwaukee, in an interview.
Ms. Thew, who was not involved in the study, said, “The number of recommended and required screening items needed in pediatrics is vast, so many providers have to select which items to focus on for their health screenings with these ages.”
Overall, “I was impressed with the improvements that were made in this quality improvement study,” said Ms. Thew.
Barriers to lipid screening in this population include the reduced number of health screenings and immunizations recommended for this age group; the consequence is that access is limited to discuss preventive care opportunities, said Ms. Thew in an interview. Steps to overcome these barriers could include the use of many of the screening tools introduced in the current study, such as point-of-care testing in the office, use of the EMR to remind providers of testing, which can be done during well visits or school physicals, and educating providers about the current guidelines, she noted.
Other strategies to increase screening include moving the immunization series to provide more frequent appointments to children aged 9-11 years to offer education and preventive care, Ms. Thew added.
The study received no outside funding. The researchers had no financial conflicts to disclose. Ms. Thew had no financial conflicts to disclose and serves on the Editorial Advisory Board of Pediatric News.
WASHINGTON – A bundled intervention combining point-of-care testing, electronic medical record support, and provider education significantly improved lipid screening rates in children aged 9-11 years, according to data from approximately 100 monthly visits over a 3-year period.
Guidelines from the National Heart, Lung, and Blood Institute currently recommend universal lipid screening for children aged 9-11 years, but screening rates in clinical practice remain low, according to Ruth E. Gardner, MD, of Penn State University, Hershey, and colleagues.
In a poster presented at the Pediatric Academic Societies annual meeting, Dr. Gardner and colleagues shared results of the implementation of a bundled testing protocol designed to improve screening.
The researchers reviewed data on lipid testing within 30 days for all 9- to 11-year-old well child visits at a single center between May 2019 and February 2022. The bundled intervention was introduced in May 2021.
The bundled protocol included in-office capillary testing and provider education. In addition, electronic medical record templates were modified to include prompts for lipid screening at relevant ages, and EMR orders were adjusted to include lipid testing. The researchers also collected targeted provider feedback on individualized screening rates in February 2022.
Screening rates were plotted monthly. For the period from May 2019 through May 2021, the rates averaged 6.5%. However, after the introduction of the bundled intervention, the rate increased to 29.9%. Following targeted provider feedback in February 2022, the researchers found an additional shift to 52.1% through March and April 2022.
The findings were limited by the use of data from a single center, and the researchers used an extended study period to account for disruptions to well-child care in the spring of 2020 related to the COVID-19 pandemic.
However, the results support the effectiveness of a bundled intervention for improving lipid screening rates in children aged 9-11 years, the researchers said, and targeted provider feedback and education could yield additional improvements, they concluded.
Preteen years are an optimal time for screening
“The current study is important because atherosclerosis begins in childhood, and screening at ages 9-11 is an optimal time to begin lifestyle changes to improve overall health and reduce risks of heart disease,” said Margaret Thew, DNP, FNP-BC, of the Medical College of Wisconsin, Milwaukee, in an interview.
Ms. Thew, who was not involved in the study, said, “The number of recommended and required screening items needed in pediatrics is vast, so many providers have to select which items to focus on for their health screenings with these ages.”
Overall, “I was impressed with the improvements that were made in this quality improvement study,” said Ms. Thew.
Barriers to lipid screening in this population include the reduced number of health screenings and immunizations recommended for this age group; the consequence is that access is limited to discuss preventive care opportunities, said Ms. Thew in an interview. Steps to overcome these barriers could include the use of many of the screening tools introduced in the current study, such as point-of-care testing in the office, use of the EMR to remind providers of testing, which can be done during well visits or school physicals, and educating providers about the current guidelines, she noted.
Other strategies to increase screening include moving the immunization series to provide more frequent appointments to children aged 9-11 years to offer education and preventive care, Ms. Thew added.
The study received no outside funding. The researchers had no financial conflicts to disclose. Ms. Thew had no financial conflicts to disclose and serves on the Editorial Advisory Board of Pediatric News.
FROM PAS 2023
Boys may carry the weight, or overweight, of adults’ infertility
Overweight boy, infertile man?
When it comes to causes of infertility, history and science have generally focused on women. A lot of the research overlooks men, but some previous studies have suggested that male infertility contributes to about half of the cases of couple infertility. The reason for much of that male infertility, however, has been a mystery. Until now.
A group of Italian investigators looked at the declining trend in sperm counts over the past 40 years and the increase of childhood obesity. Is there a correlation? The researchers think so. Childhood obesity can be linked to multiple causes, but the researchers zeroed in on the effect that obesity has on metabolic rates and, therefore, testicular growth.
Collecting data on testicular volume, body mass index (BMI), and insulin resistance from 268 boys aged 2-18 years, the researchers discovered that those with normal weight and normal insulin levels had testicular volumes 1.5 times higher than their overweight counterparts and 1.5-2 times higher than those with hyperinsulinemia, building a case for obesity being a factor for infertility later in life.
Since low testicular volume is associated with lower sperm count and production as an adult, putting two and two together makes a compelling argument for childhood obesity being a major male infertility culprit. It also creates even more urgency for the health care industry and community decision makers to focus on childhood obesity.
It sure would be nice to be able to take one of the many risk factors for future human survival off the table. Maybe by taking something, like cake, off the table.
Fecal transplantation moves to the kitchen
Fecal microbiota transplantation is an effective way to treat Clostridioides difficile infection, but, in the end, it’s still a transplantation procedure involving a nasogastric or colorectal tube or rather large oral capsules with a demanding (30-40 capsules over 2 days) dosage. Please, Science, tell us there’s a better way.
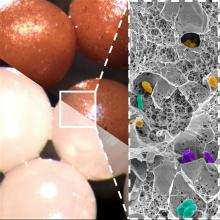
Science, in the form of investigators at the University of Geneva and Lausanne University Hospital in Switzerland, has spoken, and there may be a better way. Presenting fecal beads: All the bacterial goodness of donor stool without the tubal insertions or massive quantities of giant capsules.
We know you’re scoffing out there, but it’s true. All you need is a little alginate, which is a “biocompatible polysaccharide isolated from brown algae” of the Phaeophyceae family. The donor feces is microencapsulated by mixing it with the alginate, dropping that mixture into water containing calcium chloride, turning it into a gel, and then freeze-drying the gel into small (just 2 mm), solid beads.
Sounds plausible enough, but what do you do with them? “These brownish beads can be easily dispersed in a liquid or food that is pleasant to eat. They also have no taste,” senior author Eric Allémann, PhD, said in a statement released by the University of Geneva.
Pleasant to eat? No taste? So which is it? If you really want to know, watch fecal beads week on the new season of “The Great British Baking Show,” when Paul and Prue judge poop baked into crumpets, crepes, and crostatas. Yum.
We’re on the low-oxygen diet
Nine out of ten doctors agree: Oxygen is more important to your continued well-being than food. After all, a human can go weeks without food, but just minutes without oxygen. However, ten out of ten doctors agree that the United States has an obesity problem. They all also agree that previous research has shown soldiers who train at high altitudes lose more weight than those training at lower altitudes.
So, on the one hand, we have a country full of overweight people, and on the other, we have low oxygen levels causing weight loss. The solution, then, is obvious: Stop breathing.
More specifically (and somewhat less facetiously), researchers from Louisiana have launched the Low Oxygen and Weight Status trial and are currently recruiting individuals with BMIs of 30-40 to, uh, suffocate themselves. No, no, it’s okay, it’s just when they’re sleeping.
Fine, straight face. Participants in the LOWS trial will undergo an 8-week period when they will consume a controlled weight-loss diet and spend their nights in a hypoxic sealed tent, where they will sleep in an environment with an oxygen level equivalent to 8,500 feet above sea level (roughly equivalent to Aspen, Colo.). They will be compared with people on the same diet who sleep in a normal, sea-level oxygen environment.
The study’s goal is to determine whether or not spending time in a low-oxygen environment will suppress appetite, increase energy expenditure, and improve weight loss and insulin sensitivity. Excessive weight loss in high-altitude environments isn’t a good thing for soldiers – they kind of need their muscles and body weight to do the whole soldiering thing – but it could be great for people struggling to lose those last few pounds. And it also may prove LOTME’s previous thesis: Air is not good.
Overweight boy, infertile man?
When it comes to causes of infertility, history and science have generally focused on women. A lot of the research overlooks men, but some previous studies have suggested that male infertility contributes to about half of the cases of couple infertility. The reason for much of that male infertility, however, has been a mystery. Until now.
A group of Italian investigators looked at the declining trend in sperm counts over the past 40 years and the increase of childhood obesity. Is there a correlation? The researchers think so. Childhood obesity can be linked to multiple causes, but the researchers zeroed in on the effect that obesity has on metabolic rates and, therefore, testicular growth.
Collecting data on testicular volume, body mass index (BMI), and insulin resistance from 268 boys aged 2-18 years, the researchers discovered that those with normal weight and normal insulin levels had testicular volumes 1.5 times higher than their overweight counterparts and 1.5-2 times higher than those with hyperinsulinemia, building a case for obesity being a factor for infertility later in life.
Since low testicular volume is associated with lower sperm count and production as an adult, putting two and two together makes a compelling argument for childhood obesity being a major male infertility culprit. It also creates even more urgency for the health care industry and community decision makers to focus on childhood obesity.
It sure would be nice to be able to take one of the many risk factors for future human survival off the table. Maybe by taking something, like cake, off the table.
Fecal transplantation moves to the kitchen
Fecal microbiota transplantation is an effective way to treat Clostridioides difficile infection, but, in the end, it’s still a transplantation procedure involving a nasogastric or colorectal tube or rather large oral capsules with a demanding (30-40 capsules over 2 days) dosage. Please, Science, tell us there’s a better way.
Science, in the form of investigators at the University of Geneva and Lausanne University Hospital in Switzerland, has spoken, and there may be a better way. Presenting fecal beads: All the bacterial goodness of donor stool without the tubal insertions or massive quantities of giant capsules.
We know you’re scoffing out there, but it’s true. All you need is a little alginate, which is a “biocompatible polysaccharide isolated from brown algae” of the Phaeophyceae family. The donor feces is microencapsulated by mixing it with the alginate, dropping that mixture into water containing calcium chloride, turning it into a gel, and then freeze-drying the gel into small (just 2 mm), solid beads.
Sounds plausible enough, but what do you do with them? “These brownish beads can be easily dispersed in a liquid or food that is pleasant to eat. They also have no taste,” senior author Eric Allémann, PhD, said in a statement released by the University of Geneva.
Pleasant to eat? No taste? So which is it? If you really want to know, watch fecal beads week on the new season of “The Great British Baking Show,” when Paul and Prue judge poop baked into crumpets, crepes, and crostatas. Yum.
We’re on the low-oxygen diet
Nine out of ten doctors agree: Oxygen is more important to your continued well-being than food. After all, a human can go weeks without food, but just minutes without oxygen. However, ten out of ten doctors agree that the United States has an obesity problem. They all also agree that previous research has shown soldiers who train at high altitudes lose more weight than those training at lower altitudes.
So, on the one hand, we have a country full of overweight people, and on the other, we have low oxygen levels causing weight loss. The solution, then, is obvious: Stop breathing.
More specifically (and somewhat less facetiously), researchers from Louisiana have launched the Low Oxygen and Weight Status trial and are currently recruiting individuals with BMIs of 30-40 to, uh, suffocate themselves. No, no, it’s okay, it’s just when they’re sleeping.
Fine, straight face. Participants in the LOWS trial will undergo an 8-week period when they will consume a controlled weight-loss diet and spend their nights in a hypoxic sealed tent, where they will sleep in an environment with an oxygen level equivalent to 8,500 feet above sea level (roughly equivalent to Aspen, Colo.). They will be compared with people on the same diet who sleep in a normal, sea-level oxygen environment.
The study’s goal is to determine whether or not spending time in a low-oxygen environment will suppress appetite, increase energy expenditure, and improve weight loss and insulin sensitivity. Excessive weight loss in high-altitude environments isn’t a good thing for soldiers – they kind of need their muscles and body weight to do the whole soldiering thing – but it could be great for people struggling to lose those last few pounds. And it also may prove LOTME’s previous thesis: Air is not good.
Overweight boy, infertile man?
When it comes to causes of infertility, history and science have generally focused on women. A lot of the research overlooks men, but some previous studies have suggested that male infertility contributes to about half of the cases of couple infertility. The reason for much of that male infertility, however, has been a mystery. Until now.
A group of Italian investigators looked at the declining trend in sperm counts over the past 40 years and the increase of childhood obesity. Is there a correlation? The researchers think so. Childhood obesity can be linked to multiple causes, but the researchers zeroed in on the effect that obesity has on metabolic rates and, therefore, testicular growth.
Collecting data on testicular volume, body mass index (BMI), and insulin resistance from 268 boys aged 2-18 years, the researchers discovered that those with normal weight and normal insulin levels had testicular volumes 1.5 times higher than their overweight counterparts and 1.5-2 times higher than those with hyperinsulinemia, building a case for obesity being a factor for infertility later in life.
Since low testicular volume is associated with lower sperm count and production as an adult, putting two and two together makes a compelling argument for childhood obesity being a major male infertility culprit. It also creates even more urgency for the health care industry and community decision makers to focus on childhood obesity.
It sure would be nice to be able to take one of the many risk factors for future human survival off the table. Maybe by taking something, like cake, off the table.
Fecal transplantation moves to the kitchen
Fecal microbiota transplantation is an effective way to treat Clostridioides difficile infection, but, in the end, it’s still a transplantation procedure involving a nasogastric or colorectal tube or rather large oral capsules with a demanding (30-40 capsules over 2 days) dosage. Please, Science, tell us there’s a better way.
Science, in the form of investigators at the University of Geneva and Lausanne University Hospital in Switzerland, has spoken, and there may be a better way. Presenting fecal beads: All the bacterial goodness of donor stool without the tubal insertions or massive quantities of giant capsules.
We know you’re scoffing out there, but it’s true. All you need is a little alginate, which is a “biocompatible polysaccharide isolated from brown algae” of the Phaeophyceae family. The donor feces is microencapsulated by mixing it with the alginate, dropping that mixture into water containing calcium chloride, turning it into a gel, and then freeze-drying the gel into small (just 2 mm), solid beads.
Sounds plausible enough, but what do you do with them? “These brownish beads can be easily dispersed in a liquid or food that is pleasant to eat. They also have no taste,” senior author Eric Allémann, PhD, said in a statement released by the University of Geneva.
Pleasant to eat? No taste? So which is it? If you really want to know, watch fecal beads week on the new season of “The Great British Baking Show,” when Paul and Prue judge poop baked into crumpets, crepes, and crostatas. Yum.
We’re on the low-oxygen diet
Nine out of ten doctors agree: Oxygen is more important to your continued well-being than food. After all, a human can go weeks without food, but just minutes without oxygen. However, ten out of ten doctors agree that the United States has an obesity problem. They all also agree that previous research has shown soldiers who train at high altitudes lose more weight than those training at lower altitudes.
So, on the one hand, we have a country full of overweight people, and on the other, we have low oxygen levels causing weight loss. The solution, then, is obvious: Stop breathing.
More specifically (and somewhat less facetiously), researchers from Louisiana have launched the Low Oxygen and Weight Status trial and are currently recruiting individuals with BMIs of 30-40 to, uh, suffocate themselves. No, no, it’s okay, it’s just when they’re sleeping.
Fine, straight face. Participants in the LOWS trial will undergo an 8-week period when they will consume a controlled weight-loss diet and spend their nights in a hypoxic sealed tent, where they will sleep in an environment with an oxygen level equivalent to 8,500 feet above sea level (roughly equivalent to Aspen, Colo.). They will be compared with people on the same diet who sleep in a normal, sea-level oxygen environment.
The study’s goal is to determine whether or not spending time in a low-oxygen environment will suppress appetite, increase energy expenditure, and improve weight loss and insulin sensitivity. Excessive weight loss in high-altitude environments isn’t a good thing for soldiers – they kind of need their muscles and body weight to do the whole soldiering thing – but it could be great for people struggling to lose those last few pounds. And it also may prove LOTME’s previous thesis: Air is not good.
Surprisingly more nonsustained VT shown in HCM using extended ECG monitoring
BARCELONA – , suggests a study that questions current risk stratification practices in HCM.
In the registry study, such arrythmias were observed in about six times as many HCM patients during 30 days of ambulatory electrocardiographic monitoring as would have been identified based on the first 24 hours of the monitoring period: 65% vs. 11% of the cohort.
Also, about 62% of the patients showed NSVT at “extended” 30-day monitoring, compared with an 8% prevalence of the arrhythmia based on the more conventional ECG monitoring period of 24 hours.
Nonsustained ventricular tachycardia, an important arrhythmia used every day in clinical practice to make decisions, is “much, much more prevalent than we thought” in patients with HCM, Juan Caro Codón, MD, the study’s principal investigator, said in an interview. “We should invest in further research regarding extended ECG monitoring in these patients.”
Dr. Caro Codón, of La Paz University Hospital, Madrid, presented the findings from the TEMPO-HCM study at the European Heart Rhythm Association 2023 Congress, held in Barcelona and virtually.
Its results, he said, have implications for stratifying HCM patients according to their risk for sudden cardiac death in deciding who should be offered an implantable cardioverter-defibrillator (ICD).
The life-incidence of atrial fibrillation (AF) in patients like those in the current analysis has previously been found to be about 20%, and the life-prevalence of NSVT about 20%-30%, using traditional 24- or 48-hour Holter monitoring, Dr. Caro Codón said.
“These arrhythmias are clinically relevant events because they are linked to very meaningful clinical endpoints,” including stroke and thromboembolism, he said, “but also for sudden cardiac death.”
Extended ECG monitoring has been shown useful in the setting of cryptogenic stroke and after AF ablation, but similar findings have been scarce in HCM. Patients using personal wearable monitors such as smart watches, Dr. Caro Codón said, have come to his clinic with concerns that the devices may have signaled a problem. But the lack of relevant data leaves them without a sufficient answer.
In other findings, invited discussant Isabelle van Gelder, MD, PhD, observed after Dr. Caro Codón’s presentation that the number of patients with AF almost doubled based on extended monitoring, compared with the first 24 hours of monitoring.
Based on European Society of Cardiology guidelines from 2020, “Once clinical AF has been documented, there is a class IIA recommendation to start anticoagulation,” said Dr. van Gelder, University of Groningen, the Netherlands. “Therefore, your data really are a call for more data on screening for AF in hypertrophic cardiomyopathy patients.”
Prospective multicenter registry
The TEMPO-HCM registry includes patients with HCM and a clinical indication for standard Holter monitoring at five hospitals in Spain. It excludes patients with an HCM-like phenotype but who lack the telltale genotype, as well as those already implanted with an ICD.
Those in the current analysis underwent 30-day ECG monitoring with a small, wearable device that Dr. Caro Codón described as about 7 cm long, worn in what is essentially a T-shirt with a pocket. Patients could remove the shirt and device to bathe or go swimming, for example, and still be monitored for most of the day.
The analysis included the registry’s first 100 patients (mean age, 57 years; 78% male). Hypertension was present in 47%, 58% were on beta-blockers, 16% had prior AF or atrial flutter, and 19% were taking anticoagulants. Only 8% were on antiarrhythmic drugs, Dr. Caro Codón reported.
The patients had good functional status (68% and 29% were in NYHA class 1 and 2, respectively) and their left ventricular ejection fraction averaged 66%. Of the 71 patients who underwent MRI, 28.2% showed late gadolinium enhancement suggesting myocardial scarring.
More arrhythmias on 30-day monitoring
The primary endpoint of clinically relevant arrhythmia (AF, atrial flutter, or NSVT) was identified during the first 24 hours of monitoring in 11% of patients. The prevalence rose to 65% (P < .001) based on 30-day monitoring.
Similarly, prevalences of the composite primary endpoint components grew on extended monitoring, but the increases reached statistical significance only for NSVT; its prevalence went from 8% to 62% (P < .001). Prevalences rose nonsignificantly from 6% to 10% for AF and 0% to 1% for sustained ventricular tachycardia.
The incidence of NSVT during monitoring climbed fastest from day 0 through about day 19 and then rose more slowly through day 30, Dr. Caro Codón said. “It actually didn’t reach a plateau during this time period, so there is the possibility that if we had continued monitoring patients, the difference between both periods may have been even higher.”
Three variables predicted the incidence of nonsustained VT during monitoring, he said: age, atrial wall thickness, and whether there was late gadolinium enhancement at MRI.
An exploratory analysis looked at the 5-year risk of sudden cardiac death using the European Society of Cardiology HCM-SCD risk calculator recommended in guidelines. Risk assessment based on the 30-day extended monitoring period, compared with the first 24 hours of monitoring alone, predicted a significantly higher 5-year risk of sudden death, Dr. Caro Codón said.
“Even more importantly,” he added, “over 20%” of patients would have been reclassified into a higher-risk group and possibly considered for an ICD based on extended monitoring, compared to 24-hour monitoring.
However, given that more than 50% of patients were found to have NSVT during extended monitoring, Dr. Caro Codón proposed that decisions on whether to implant an ICD should not be so “binary” based on the presence or absence of symptoms, and proposed further investigations be conducted into the complete phenotype of these arrhythmias.
The study has limitations, he observed, including a relatively small size; but it was able to detect important differences between 24-hour and 30-day monitoring outcomes even with only 100 patients. It was also limited by a lack of clinical follow-up for information on endpoints like stroke, thromboembolism, and sudden cardiac death.
Extended monitoring detected more cases of NSVT in the study’s relatively low-risk HCM patients who would not generally have an indication for ICD implantation, observed Dr. van Gelder. Also, at present the prognostic value of NSVT for SCD “seems to be more important at younger age” – that is, younger than 30 years – in patients with HCM.
Dr. van Gelder echoed Dr. Caro Codón’s call for more data from prolonged monitoring to help stratify patients according to risk; she proposed NSVT frequency, duration, and rate as possible targets.
The study was supported by an unrestricted grant from Nuubo, which provided the ECG monitoring systems. Dr. Caro Codón and Dr. van Gelder reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BARCELONA – , suggests a study that questions current risk stratification practices in HCM.
In the registry study, such arrythmias were observed in about six times as many HCM patients during 30 days of ambulatory electrocardiographic monitoring as would have been identified based on the first 24 hours of the monitoring period: 65% vs. 11% of the cohort.
Also, about 62% of the patients showed NSVT at “extended” 30-day monitoring, compared with an 8% prevalence of the arrhythmia based on the more conventional ECG monitoring period of 24 hours.
Nonsustained ventricular tachycardia, an important arrhythmia used every day in clinical practice to make decisions, is “much, much more prevalent than we thought” in patients with HCM, Juan Caro Codón, MD, the study’s principal investigator, said in an interview. “We should invest in further research regarding extended ECG monitoring in these patients.”
Dr. Caro Codón, of La Paz University Hospital, Madrid, presented the findings from the TEMPO-HCM study at the European Heart Rhythm Association 2023 Congress, held in Barcelona and virtually.
Its results, he said, have implications for stratifying HCM patients according to their risk for sudden cardiac death in deciding who should be offered an implantable cardioverter-defibrillator (ICD).
The life-incidence of atrial fibrillation (AF) in patients like those in the current analysis has previously been found to be about 20%, and the life-prevalence of NSVT about 20%-30%, using traditional 24- or 48-hour Holter monitoring, Dr. Caro Codón said.
“These arrhythmias are clinically relevant events because they are linked to very meaningful clinical endpoints,” including stroke and thromboembolism, he said, “but also for sudden cardiac death.”
Extended ECG monitoring has been shown useful in the setting of cryptogenic stroke and after AF ablation, but similar findings have been scarce in HCM. Patients using personal wearable monitors such as smart watches, Dr. Caro Codón said, have come to his clinic with concerns that the devices may have signaled a problem. But the lack of relevant data leaves them without a sufficient answer.
In other findings, invited discussant Isabelle van Gelder, MD, PhD, observed after Dr. Caro Codón’s presentation that the number of patients with AF almost doubled based on extended monitoring, compared with the first 24 hours of monitoring.
Based on European Society of Cardiology guidelines from 2020, “Once clinical AF has been documented, there is a class IIA recommendation to start anticoagulation,” said Dr. van Gelder, University of Groningen, the Netherlands. “Therefore, your data really are a call for more data on screening for AF in hypertrophic cardiomyopathy patients.”
Prospective multicenter registry
The TEMPO-HCM registry includes patients with HCM and a clinical indication for standard Holter monitoring at five hospitals in Spain. It excludes patients with an HCM-like phenotype but who lack the telltale genotype, as well as those already implanted with an ICD.
Those in the current analysis underwent 30-day ECG monitoring with a small, wearable device that Dr. Caro Codón described as about 7 cm long, worn in what is essentially a T-shirt with a pocket. Patients could remove the shirt and device to bathe or go swimming, for example, and still be monitored for most of the day.
The analysis included the registry’s first 100 patients (mean age, 57 years; 78% male). Hypertension was present in 47%, 58% were on beta-blockers, 16% had prior AF or atrial flutter, and 19% were taking anticoagulants. Only 8% were on antiarrhythmic drugs, Dr. Caro Codón reported.
The patients had good functional status (68% and 29% were in NYHA class 1 and 2, respectively) and their left ventricular ejection fraction averaged 66%. Of the 71 patients who underwent MRI, 28.2% showed late gadolinium enhancement suggesting myocardial scarring.
More arrhythmias on 30-day monitoring
The primary endpoint of clinically relevant arrhythmia (AF, atrial flutter, or NSVT) was identified during the first 24 hours of monitoring in 11% of patients. The prevalence rose to 65% (P < .001) based on 30-day monitoring.
Similarly, prevalences of the composite primary endpoint components grew on extended monitoring, but the increases reached statistical significance only for NSVT; its prevalence went from 8% to 62% (P < .001). Prevalences rose nonsignificantly from 6% to 10% for AF and 0% to 1% for sustained ventricular tachycardia.
The incidence of NSVT during monitoring climbed fastest from day 0 through about day 19 and then rose more slowly through day 30, Dr. Caro Codón said. “It actually didn’t reach a plateau during this time period, so there is the possibility that if we had continued monitoring patients, the difference between both periods may have been even higher.”
Three variables predicted the incidence of nonsustained VT during monitoring, he said: age, atrial wall thickness, and whether there was late gadolinium enhancement at MRI.
An exploratory analysis looked at the 5-year risk of sudden cardiac death using the European Society of Cardiology HCM-SCD risk calculator recommended in guidelines. Risk assessment based on the 30-day extended monitoring period, compared with the first 24 hours of monitoring alone, predicted a significantly higher 5-year risk of sudden death, Dr. Caro Codón said.
“Even more importantly,” he added, “over 20%” of patients would have been reclassified into a higher-risk group and possibly considered for an ICD based on extended monitoring, compared to 24-hour monitoring.
However, given that more than 50% of patients were found to have NSVT during extended monitoring, Dr. Caro Codón proposed that decisions on whether to implant an ICD should not be so “binary” based on the presence or absence of symptoms, and proposed further investigations be conducted into the complete phenotype of these arrhythmias.
The study has limitations, he observed, including a relatively small size; but it was able to detect important differences between 24-hour and 30-day monitoring outcomes even with only 100 patients. It was also limited by a lack of clinical follow-up for information on endpoints like stroke, thromboembolism, and sudden cardiac death.
Extended monitoring detected more cases of NSVT in the study’s relatively low-risk HCM patients who would not generally have an indication for ICD implantation, observed Dr. van Gelder. Also, at present the prognostic value of NSVT for SCD “seems to be more important at younger age” – that is, younger than 30 years – in patients with HCM.
Dr. van Gelder echoed Dr. Caro Codón’s call for more data from prolonged monitoring to help stratify patients according to risk; she proposed NSVT frequency, duration, and rate as possible targets.
The study was supported by an unrestricted grant from Nuubo, which provided the ECG monitoring systems. Dr. Caro Codón and Dr. van Gelder reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BARCELONA – , suggests a study that questions current risk stratification practices in HCM.
In the registry study, such arrythmias were observed in about six times as many HCM patients during 30 days of ambulatory electrocardiographic monitoring as would have been identified based on the first 24 hours of the monitoring period: 65% vs. 11% of the cohort.
Also, about 62% of the patients showed NSVT at “extended” 30-day monitoring, compared with an 8% prevalence of the arrhythmia based on the more conventional ECG monitoring period of 24 hours.
Nonsustained ventricular tachycardia, an important arrhythmia used every day in clinical practice to make decisions, is “much, much more prevalent than we thought” in patients with HCM, Juan Caro Codón, MD, the study’s principal investigator, said in an interview. “We should invest in further research regarding extended ECG monitoring in these patients.”
Dr. Caro Codón, of La Paz University Hospital, Madrid, presented the findings from the TEMPO-HCM study at the European Heart Rhythm Association 2023 Congress, held in Barcelona and virtually.
Its results, he said, have implications for stratifying HCM patients according to their risk for sudden cardiac death in deciding who should be offered an implantable cardioverter-defibrillator (ICD).
The life-incidence of atrial fibrillation (AF) in patients like those in the current analysis has previously been found to be about 20%, and the life-prevalence of NSVT about 20%-30%, using traditional 24- or 48-hour Holter monitoring, Dr. Caro Codón said.
“These arrhythmias are clinically relevant events because they are linked to very meaningful clinical endpoints,” including stroke and thromboembolism, he said, “but also for sudden cardiac death.”
Extended ECG monitoring has been shown useful in the setting of cryptogenic stroke and after AF ablation, but similar findings have been scarce in HCM. Patients using personal wearable monitors such as smart watches, Dr. Caro Codón said, have come to his clinic with concerns that the devices may have signaled a problem. But the lack of relevant data leaves them without a sufficient answer.
In other findings, invited discussant Isabelle van Gelder, MD, PhD, observed after Dr. Caro Codón’s presentation that the number of patients with AF almost doubled based on extended monitoring, compared with the first 24 hours of monitoring.
Based on European Society of Cardiology guidelines from 2020, “Once clinical AF has been documented, there is a class IIA recommendation to start anticoagulation,” said Dr. van Gelder, University of Groningen, the Netherlands. “Therefore, your data really are a call for more data on screening for AF in hypertrophic cardiomyopathy patients.”
Prospective multicenter registry
The TEMPO-HCM registry includes patients with HCM and a clinical indication for standard Holter monitoring at five hospitals in Spain. It excludes patients with an HCM-like phenotype but who lack the telltale genotype, as well as those already implanted with an ICD.
Those in the current analysis underwent 30-day ECG monitoring with a small, wearable device that Dr. Caro Codón described as about 7 cm long, worn in what is essentially a T-shirt with a pocket. Patients could remove the shirt and device to bathe or go swimming, for example, and still be monitored for most of the day.
The analysis included the registry’s first 100 patients (mean age, 57 years; 78% male). Hypertension was present in 47%, 58% were on beta-blockers, 16% had prior AF or atrial flutter, and 19% were taking anticoagulants. Only 8% were on antiarrhythmic drugs, Dr. Caro Codón reported.
The patients had good functional status (68% and 29% were in NYHA class 1 and 2, respectively) and their left ventricular ejection fraction averaged 66%. Of the 71 patients who underwent MRI, 28.2% showed late gadolinium enhancement suggesting myocardial scarring.
More arrhythmias on 30-day monitoring
The primary endpoint of clinically relevant arrhythmia (AF, atrial flutter, or NSVT) was identified during the first 24 hours of monitoring in 11% of patients. The prevalence rose to 65% (P < .001) based on 30-day monitoring.
Similarly, prevalences of the composite primary endpoint components grew on extended monitoring, but the increases reached statistical significance only for NSVT; its prevalence went from 8% to 62% (P < .001). Prevalences rose nonsignificantly from 6% to 10% for AF and 0% to 1% for sustained ventricular tachycardia.
The incidence of NSVT during monitoring climbed fastest from day 0 through about day 19 and then rose more slowly through day 30, Dr. Caro Codón said. “It actually didn’t reach a plateau during this time period, so there is the possibility that if we had continued monitoring patients, the difference between both periods may have been even higher.”
Three variables predicted the incidence of nonsustained VT during monitoring, he said: age, atrial wall thickness, and whether there was late gadolinium enhancement at MRI.
An exploratory analysis looked at the 5-year risk of sudden cardiac death using the European Society of Cardiology HCM-SCD risk calculator recommended in guidelines. Risk assessment based on the 30-day extended monitoring period, compared with the first 24 hours of monitoring alone, predicted a significantly higher 5-year risk of sudden death, Dr. Caro Codón said.
“Even more importantly,” he added, “over 20%” of patients would have been reclassified into a higher-risk group and possibly considered for an ICD based on extended monitoring, compared to 24-hour monitoring.
However, given that more than 50% of patients were found to have NSVT during extended monitoring, Dr. Caro Codón proposed that decisions on whether to implant an ICD should not be so “binary” based on the presence or absence of symptoms, and proposed further investigations be conducted into the complete phenotype of these arrhythmias.
The study has limitations, he observed, including a relatively small size; but it was able to detect important differences between 24-hour and 30-day monitoring outcomes even with only 100 patients. It was also limited by a lack of clinical follow-up for information on endpoints like stroke, thromboembolism, and sudden cardiac death.
Extended monitoring detected more cases of NSVT in the study’s relatively low-risk HCM patients who would not generally have an indication for ICD implantation, observed Dr. van Gelder. Also, at present the prognostic value of NSVT for SCD “seems to be more important at younger age” – that is, younger than 30 years – in patients with HCM.
Dr. van Gelder echoed Dr. Caro Codón’s call for more data from prolonged monitoring to help stratify patients according to risk; she proposed NSVT frequency, duration, and rate as possible targets.
The study was supported by an unrestricted grant from Nuubo, which provided the ECG monitoring systems. Dr. Caro Codón and Dr. van Gelder reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM EHRA
Clinical trials: Top priority for long COVID
The Centers for Disease Control and Prevention and the U.S. Census Bureau estimate that 6.1% of the U.S. adult population is living with long COVID, with millions more debilitated worldwide. The demand for substantial treatment is enormous, but the urgency to fund and begin the necessary range of clinical trials has not met the severity of the problem.
While trials are slowly beginning to happen, the treatment choices and trial design require crucial nuances and understanding of viral-onset illnesses, and few research groups are creating strong trials that fully reflect the complexities of this landscape.
These recommendations recognize that roughly half of long COVID patients have new-onset myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and dysautonomia from COVID, which must be at the forefront of how trials are designed and conducted, and are additionally based on the current hypotheses about long COVID’s pathophysiologies.
1: Drugs proposed by experts in postviral fields should be prioritized
Upward of 50 drugs for viral-onset conditions like ME/CFS, dysautonomia, AIDS, and others have been waiting for years to go to trial, but have not had the funding to do so.
Treatments proposed by experts in viral-onset illnesses (such as ME/CFS and dysautonomia) should be prioritized (PM R. 2022 Oct;14[10]:1270-91), as outside researchers are not familiar with these fields and their potential treatment options.
2: Drugs targeting a wide range of mechanisms should be trialed
Treatments that should be trialed include anticoagulants/antiplatelets for clotting and vascular functioning, immunomodulators including JAK-STAT inhibitors, COVID-specific antivirals and antivirals against reactivated herpesviruses (Valcyte, Valacyclovir, EBV vaccine).
Other options include prescription mast cell stabilizers (ketotifen, cromolyn sodium), drugs that regulate microglial activation (low-dose naltrexone, low-dose aripiprazole), anti-CGRP medications, beta-blockers, and intravenous immunoglobulin.
Others include medications that target mitochondrial dysfunction; ivabradine; pyridostigmine;, DRP1 inhibitors; supplements showing success in patient communities including lactoferrin, ubiquinone, and nattokinase; and therapies targeting glymphatic/lymphatic dysfunction, microbiome therapies, and therapeutic peptides.
3: Use appropriate long COVID subtypes
Long COVID is an umbrella term that encompasses multiple new-onset and worsened conditions and symptoms after COVID. Roughly half of long COVID patients likely meet the criteria for ME/CFS and/or dysautonomia. Others may have new-onset diabetes, major clotting events, lung damage, neurological disorders, loss of smell or taste, and other manifestations.
Patients in different categories likely have different responses to treatments. It’s critical to identify appropriate subtypes for each trial, ideally performing detailed analyses to identify the treatments that work best, and don’t, for each subtype.
4: Behavioral treatments, especially those that have harmed similar populations, should not be trialed
Behavioral treatments including exercise, graded exercise therapy (GET), and cognitive-behavioral therapy (CBT) should not be trialed, let alone prioritized, for long COVID.
In patients with postexertional malaise (PEM), one of the most common long COVID symptoms, exercise is actively harmful and causes dysfunctional metabolic patterns, cardiac preload failure, impaired systemic oxygen extraction, and more. GET and CBT have failed similar populations , and exercise is explicitly contraindicated by the World Health Organization, the British National Institute for Health and Care Excellence, the CDC, and other organizations.
Resources should instead be put toward the wide range of medications that have not yet adequately undergone clinical trials.
5: PCR and antibody tests should not be used as inclusion criteria for trial participants
Only an estimated 1%-3% of cases in the first wave of COVID were documented, and the CDC estimates that only 25% of cases through September 2021 were documented. Similarly, antibody tests are unreliable to determine past infection, as roughly a third of patients don’t seroconvert, and a similar proportion serorevert within a few months. Using polymerase chain reaction (PCR) and antibody testing to determine who should be included in clinical trials limits who is eligible to participate in research, particularly those who have been ill for longer. Additionally, the majority of those who serorevert are women, so using antibody tests for inclusion introduces a selection bias and may miss mechanisms of immune system functioning that are part of long COVID.
PCR tests also have high false-negative rates and requiring them in research excludes people with lower viral loads with long COVID, which would confound findings.
These issues with testing also lead to COVID-infected people accidentally being included in control groups, which ruins the credibility of the research findings completely.
6: Include comparator groups
There are several common diagnoses that occur in people with long COVID, including ME/CFS, postural orthostatic tachycardia syndrome, small-fiber neuropathy, mast cell activation syndrome, and Ehlers-Danlos syndrome.
Identifying people with these conditions within the trial cohort improves research across all fields, benefiting all groups, and helps clarify what types of patients benefit most from certain medications.
7: Identify the right endpoints; avoid the wrong ones
Even though our understanding of the pathophysiology of long COVID is still evolving, it’s still possible to do clinical trials by identifying strong endpoints and outcome measures.
Several tools have been designed for viral-onset conditions and should be used alongside other endpoints. Postexertional malaise and autonomic symptoms, which are some of the most common symptoms of long COVID, can be measured with the validated DSQ-PEM and COMPASS-31, respectively. Tools for cognitive dysfunction trials should capture specific and common types of impairment, like processing speed.
Endpoints should be high-impact and aim for large improvements that have clinical significance over small improvements that do not have clinical significance.
Objective tests should be incorporated where possible; some to consider include natural killer cell functioning, cerebral blood flow, T-cell functioning, levels of reactivated herpesviruses, blood lactate levels, and microclots, as testing becomes available.
Mental health outcomes shouldn’t be primary endpoints, except where a trial is targeting a specific mental health condition because of COVID (for example, premenstrual dysphoric disorder).
If mental health conditions are tracked secondarily, it’s vital not to use questionnaires that include physical symptoms like fatigue, difficulty concentrating, difficulty sleeping, or palpitations, as these artificially increase depression and anxiety scores in chronically ill respondents. Tools that include physical symptoms (Patient Health Questionnaire–9, Beck Anxiety Inventory, Beck Depression Inventory) can be replaced with scales like the PHQ-2, General Anxiety Disorder–7, Hospital Anxiety and Depression Scale, or PROMIS-29 subscales.
Because certain cytokines and other inflammatory markers may naturally decrease over time without corresponding improvement in the ME/CFS subtype, caution should be taken when using cytokines as endpoints.
8: Consider enrollment and objectives carefully
A proportion of people with long COVID will recover in the early months after infection. Ideally, clinical trials will primarily study treatments in patients who have been ill 6 months or longer, as some natural recovery will happen before that can bias studies.
But where resources are abundant, it is ideal for trials to additionally look at whether the treatments can help patients in the early months recover and prevent progression to the later stage.
9: Tracking illness duration is crucial
Research from ME/CFS shows that there may be an immune change in the first few years of the illness, where cytokines decrease without any corresponding change in symptom improvement.
Because of this and the possibility that other markers follow the same pattern, disease duration should be a core feature of all analyses and trial designs. Trial outcomes should be designed to answer the question of whether the medication helps patients at different durations of illness.
10: Prioritize patient populations less likely to recover without intervention
Some long COVID phenotypes seem less likely to recover without intervention. Trials should take care to focus on these patient populations, which include those with neurologic symptoms and those meeting ME/CFS criteria.
11: Account for the relapsing/remitting nature
Outcome measures need to be assessed in a way that can distinguish a temporary remission, which is part of the natural course of the disease, from a permanent cure.
Factors that can contribute to the relapsing/remitting nature include physical and cognitive postexertional malaise, menstrual cycle changes, and seasonal changes.
12: Trial participants should reflect the diversity of the long COVID population
Certain demographics are more likely to be affected by acute and long COVID and need to be appropriately recruited and reflected in research, including in patient engagement.
Trials must include high numbers of Hispanic/Latinx, Black, and indigenous communities, queer and transgender populations, and women. Trial materials and design need to incorporate linguistic diversity in addition to racial/ethnic diversity.
Upward of 75% of long COVID cases happen after mild acute cases; clinical researchers should ensure that nonhospitalized patients make up the bulk of trial participants.
13: Utilize meaningful engagement of patients, especially in treatment selection and study design
Meaningful patient engagement means engaging multiple patients at every step of the trial process, from treatment selection to study design to analysis to communication of the results.
Patient experiences are extremely valuable and contain information that researchers may not be familiar with, including the nature and patterns of the illness, insights into possible treatments, and barriers to documentation and care that may also impact research. Tapping into those patient experiences will make trials stronger.
Overall, the landscape of long COVID clinical trials is ripe for discovery, and researchers choosing to go down this path will be deeply appreciated by the patient community.
Hannah Davis is a long COVID patient-researcher and cofounder of the Patient-Led Research Collaborative, an organization studying the long-term effects of COVID.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention and the U.S. Census Bureau estimate that 6.1% of the U.S. adult population is living with long COVID, with millions more debilitated worldwide. The demand for substantial treatment is enormous, but the urgency to fund and begin the necessary range of clinical trials has not met the severity of the problem.
While trials are slowly beginning to happen, the treatment choices and trial design require crucial nuances and understanding of viral-onset illnesses, and few research groups are creating strong trials that fully reflect the complexities of this landscape.
These recommendations recognize that roughly half of long COVID patients have new-onset myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and dysautonomia from COVID, which must be at the forefront of how trials are designed and conducted, and are additionally based on the current hypotheses about long COVID’s pathophysiologies.
1: Drugs proposed by experts in postviral fields should be prioritized
Upward of 50 drugs for viral-onset conditions like ME/CFS, dysautonomia, AIDS, and others have been waiting for years to go to trial, but have not had the funding to do so.
Treatments proposed by experts in viral-onset illnesses (such as ME/CFS and dysautonomia) should be prioritized (PM R. 2022 Oct;14[10]:1270-91), as outside researchers are not familiar with these fields and their potential treatment options.
2: Drugs targeting a wide range of mechanisms should be trialed
Treatments that should be trialed include anticoagulants/antiplatelets for clotting and vascular functioning, immunomodulators including JAK-STAT inhibitors, COVID-specific antivirals and antivirals against reactivated herpesviruses (Valcyte, Valacyclovir, EBV vaccine).
Other options include prescription mast cell stabilizers (ketotifen, cromolyn sodium), drugs that regulate microglial activation (low-dose naltrexone, low-dose aripiprazole), anti-CGRP medications, beta-blockers, and intravenous immunoglobulin.
Others include medications that target mitochondrial dysfunction; ivabradine; pyridostigmine;, DRP1 inhibitors; supplements showing success in patient communities including lactoferrin, ubiquinone, and nattokinase; and therapies targeting glymphatic/lymphatic dysfunction, microbiome therapies, and therapeutic peptides.
3: Use appropriate long COVID subtypes
Long COVID is an umbrella term that encompasses multiple new-onset and worsened conditions and symptoms after COVID. Roughly half of long COVID patients likely meet the criteria for ME/CFS and/or dysautonomia. Others may have new-onset diabetes, major clotting events, lung damage, neurological disorders, loss of smell or taste, and other manifestations.
Patients in different categories likely have different responses to treatments. It’s critical to identify appropriate subtypes for each trial, ideally performing detailed analyses to identify the treatments that work best, and don’t, for each subtype.
4: Behavioral treatments, especially those that have harmed similar populations, should not be trialed
Behavioral treatments including exercise, graded exercise therapy (GET), and cognitive-behavioral therapy (CBT) should not be trialed, let alone prioritized, for long COVID.
In patients with postexertional malaise (PEM), one of the most common long COVID symptoms, exercise is actively harmful and causes dysfunctional metabolic patterns, cardiac preload failure, impaired systemic oxygen extraction, and more. GET and CBT have failed similar populations , and exercise is explicitly contraindicated by the World Health Organization, the British National Institute for Health and Care Excellence, the CDC, and other organizations.
Resources should instead be put toward the wide range of medications that have not yet adequately undergone clinical trials.
5: PCR and antibody tests should not be used as inclusion criteria for trial participants
Only an estimated 1%-3% of cases in the first wave of COVID were documented, and the CDC estimates that only 25% of cases through September 2021 were documented. Similarly, antibody tests are unreliable to determine past infection, as roughly a third of patients don’t seroconvert, and a similar proportion serorevert within a few months. Using polymerase chain reaction (PCR) and antibody testing to determine who should be included in clinical trials limits who is eligible to participate in research, particularly those who have been ill for longer. Additionally, the majority of those who serorevert are women, so using antibody tests for inclusion introduces a selection bias and may miss mechanisms of immune system functioning that are part of long COVID.
PCR tests also have high false-negative rates and requiring them in research excludes people with lower viral loads with long COVID, which would confound findings.
These issues with testing also lead to COVID-infected people accidentally being included in control groups, which ruins the credibility of the research findings completely.
6: Include comparator groups
There are several common diagnoses that occur in people with long COVID, including ME/CFS, postural orthostatic tachycardia syndrome, small-fiber neuropathy, mast cell activation syndrome, and Ehlers-Danlos syndrome.
Identifying people with these conditions within the trial cohort improves research across all fields, benefiting all groups, and helps clarify what types of patients benefit most from certain medications.
7: Identify the right endpoints; avoid the wrong ones
Even though our understanding of the pathophysiology of long COVID is still evolving, it’s still possible to do clinical trials by identifying strong endpoints and outcome measures.
Several tools have been designed for viral-onset conditions and should be used alongside other endpoints. Postexertional malaise and autonomic symptoms, which are some of the most common symptoms of long COVID, can be measured with the validated DSQ-PEM and COMPASS-31, respectively. Tools for cognitive dysfunction trials should capture specific and common types of impairment, like processing speed.
Endpoints should be high-impact and aim for large improvements that have clinical significance over small improvements that do not have clinical significance.
Objective tests should be incorporated where possible; some to consider include natural killer cell functioning, cerebral blood flow, T-cell functioning, levels of reactivated herpesviruses, blood lactate levels, and microclots, as testing becomes available.
Mental health outcomes shouldn’t be primary endpoints, except where a trial is targeting a specific mental health condition because of COVID (for example, premenstrual dysphoric disorder).
If mental health conditions are tracked secondarily, it’s vital not to use questionnaires that include physical symptoms like fatigue, difficulty concentrating, difficulty sleeping, or palpitations, as these artificially increase depression and anxiety scores in chronically ill respondents. Tools that include physical symptoms (Patient Health Questionnaire–9, Beck Anxiety Inventory, Beck Depression Inventory) can be replaced with scales like the PHQ-2, General Anxiety Disorder–7, Hospital Anxiety and Depression Scale, or PROMIS-29 subscales.
Because certain cytokines and other inflammatory markers may naturally decrease over time without corresponding improvement in the ME/CFS subtype, caution should be taken when using cytokines as endpoints.
8: Consider enrollment and objectives carefully
A proportion of people with long COVID will recover in the early months after infection. Ideally, clinical trials will primarily study treatments in patients who have been ill 6 months or longer, as some natural recovery will happen before that can bias studies.
But where resources are abundant, it is ideal for trials to additionally look at whether the treatments can help patients in the early months recover and prevent progression to the later stage.
9: Tracking illness duration is crucial
Research from ME/CFS shows that there may be an immune change in the first few years of the illness, where cytokines decrease without any corresponding change in symptom improvement.
Because of this and the possibility that other markers follow the same pattern, disease duration should be a core feature of all analyses and trial designs. Trial outcomes should be designed to answer the question of whether the medication helps patients at different durations of illness.
10: Prioritize patient populations less likely to recover without intervention
Some long COVID phenotypes seem less likely to recover without intervention. Trials should take care to focus on these patient populations, which include those with neurologic symptoms and those meeting ME/CFS criteria.
11: Account for the relapsing/remitting nature
Outcome measures need to be assessed in a way that can distinguish a temporary remission, which is part of the natural course of the disease, from a permanent cure.
Factors that can contribute to the relapsing/remitting nature include physical and cognitive postexertional malaise, menstrual cycle changes, and seasonal changes.
12: Trial participants should reflect the diversity of the long COVID population
Certain demographics are more likely to be affected by acute and long COVID and need to be appropriately recruited and reflected in research, including in patient engagement.
Trials must include high numbers of Hispanic/Latinx, Black, and indigenous communities, queer and transgender populations, and women. Trial materials and design need to incorporate linguistic diversity in addition to racial/ethnic diversity.
Upward of 75% of long COVID cases happen after mild acute cases; clinical researchers should ensure that nonhospitalized patients make up the bulk of trial participants.
13: Utilize meaningful engagement of patients, especially in treatment selection and study design
Meaningful patient engagement means engaging multiple patients at every step of the trial process, from treatment selection to study design to analysis to communication of the results.
Patient experiences are extremely valuable and contain information that researchers may not be familiar with, including the nature and patterns of the illness, insights into possible treatments, and barriers to documentation and care that may also impact research. Tapping into those patient experiences will make trials stronger.
Overall, the landscape of long COVID clinical trials is ripe for discovery, and researchers choosing to go down this path will be deeply appreciated by the patient community.
Hannah Davis is a long COVID patient-researcher and cofounder of the Patient-Led Research Collaborative, an organization studying the long-term effects of COVID.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention and the U.S. Census Bureau estimate that 6.1% of the U.S. adult population is living with long COVID, with millions more debilitated worldwide. The demand for substantial treatment is enormous, but the urgency to fund and begin the necessary range of clinical trials has not met the severity of the problem.
While trials are slowly beginning to happen, the treatment choices and trial design require crucial nuances and understanding of viral-onset illnesses, and few research groups are creating strong trials that fully reflect the complexities of this landscape.
These recommendations recognize that roughly half of long COVID patients have new-onset myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and dysautonomia from COVID, which must be at the forefront of how trials are designed and conducted, and are additionally based on the current hypotheses about long COVID’s pathophysiologies.
1: Drugs proposed by experts in postviral fields should be prioritized
Upward of 50 drugs for viral-onset conditions like ME/CFS, dysautonomia, AIDS, and others have been waiting for years to go to trial, but have not had the funding to do so.
Treatments proposed by experts in viral-onset illnesses (such as ME/CFS and dysautonomia) should be prioritized (PM R. 2022 Oct;14[10]:1270-91), as outside researchers are not familiar with these fields and their potential treatment options.
2: Drugs targeting a wide range of mechanisms should be trialed
Treatments that should be trialed include anticoagulants/antiplatelets for clotting and vascular functioning, immunomodulators including JAK-STAT inhibitors, COVID-specific antivirals and antivirals against reactivated herpesviruses (Valcyte, Valacyclovir, EBV vaccine).
Other options include prescription mast cell stabilizers (ketotifen, cromolyn sodium), drugs that regulate microglial activation (low-dose naltrexone, low-dose aripiprazole), anti-CGRP medications, beta-blockers, and intravenous immunoglobulin.
Others include medications that target mitochondrial dysfunction; ivabradine; pyridostigmine;, DRP1 inhibitors; supplements showing success in patient communities including lactoferrin, ubiquinone, and nattokinase; and therapies targeting glymphatic/lymphatic dysfunction, microbiome therapies, and therapeutic peptides.
3: Use appropriate long COVID subtypes
Long COVID is an umbrella term that encompasses multiple new-onset and worsened conditions and symptoms after COVID. Roughly half of long COVID patients likely meet the criteria for ME/CFS and/or dysautonomia. Others may have new-onset diabetes, major clotting events, lung damage, neurological disorders, loss of smell or taste, and other manifestations.
Patients in different categories likely have different responses to treatments. It’s critical to identify appropriate subtypes for each trial, ideally performing detailed analyses to identify the treatments that work best, and don’t, for each subtype.
4: Behavioral treatments, especially those that have harmed similar populations, should not be trialed
Behavioral treatments including exercise, graded exercise therapy (GET), and cognitive-behavioral therapy (CBT) should not be trialed, let alone prioritized, for long COVID.
In patients with postexertional malaise (PEM), one of the most common long COVID symptoms, exercise is actively harmful and causes dysfunctional metabolic patterns, cardiac preload failure, impaired systemic oxygen extraction, and more. GET and CBT have failed similar populations , and exercise is explicitly contraindicated by the World Health Organization, the British National Institute for Health and Care Excellence, the CDC, and other organizations.
Resources should instead be put toward the wide range of medications that have not yet adequately undergone clinical trials.
5: PCR and antibody tests should not be used as inclusion criteria for trial participants
Only an estimated 1%-3% of cases in the first wave of COVID were documented, and the CDC estimates that only 25% of cases through September 2021 were documented. Similarly, antibody tests are unreliable to determine past infection, as roughly a third of patients don’t seroconvert, and a similar proportion serorevert within a few months. Using polymerase chain reaction (PCR) and antibody testing to determine who should be included in clinical trials limits who is eligible to participate in research, particularly those who have been ill for longer. Additionally, the majority of those who serorevert are women, so using antibody tests for inclusion introduces a selection bias and may miss mechanisms of immune system functioning that are part of long COVID.
PCR tests also have high false-negative rates and requiring them in research excludes people with lower viral loads with long COVID, which would confound findings.
These issues with testing also lead to COVID-infected people accidentally being included in control groups, which ruins the credibility of the research findings completely.
6: Include comparator groups
There are several common diagnoses that occur in people with long COVID, including ME/CFS, postural orthostatic tachycardia syndrome, small-fiber neuropathy, mast cell activation syndrome, and Ehlers-Danlos syndrome.
Identifying people with these conditions within the trial cohort improves research across all fields, benefiting all groups, and helps clarify what types of patients benefit most from certain medications.
7: Identify the right endpoints; avoid the wrong ones
Even though our understanding of the pathophysiology of long COVID is still evolving, it’s still possible to do clinical trials by identifying strong endpoints and outcome measures.
Several tools have been designed for viral-onset conditions and should be used alongside other endpoints. Postexertional malaise and autonomic symptoms, which are some of the most common symptoms of long COVID, can be measured with the validated DSQ-PEM and COMPASS-31, respectively. Tools for cognitive dysfunction trials should capture specific and common types of impairment, like processing speed.
Endpoints should be high-impact and aim for large improvements that have clinical significance over small improvements that do not have clinical significance.
Objective tests should be incorporated where possible; some to consider include natural killer cell functioning, cerebral blood flow, T-cell functioning, levels of reactivated herpesviruses, blood lactate levels, and microclots, as testing becomes available.
Mental health outcomes shouldn’t be primary endpoints, except where a trial is targeting a specific mental health condition because of COVID (for example, premenstrual dysphoric disorder).
If mental health conditions are tracked secondarily, it’s vital not to use questionnaires that include physical symptoms like fatigue, difficulty concentrating, difficulty sleeping, or palpitations, as these artificially increase depression and anxiety scores in chronically ill respondents. Tools that include physical symptoms (Patient Health Questionnaire–9, Beck Anxiety Inventory, Beck Depression Inventory) can be replaced with scales like the PHQ-2, General Anxiety Disorder–7, Hospital Anxiety and Depression Scale, or PROMIS-29 subscales.
Because certain cytokines and other inflammatory markers may naturally decrease over time without corresponding improvement in the ME/CFS subtype, caution should be taken when using cytokines as endpoints.
8: Consider enrollment and objectives carefully
A proportion of people with long COVID will recover in the early months after infection. Ideally, clinical trials will primarily study treatments in patients who have been ill 6 months or longer, as some natural recovery will happen before that can bias studies.
But where resources are abundant, it is ideal for trials to additionally look at whether the treatments can help patients in the early months recover and prevent progression to the later stage.
9: Tracking illness duration is crucial
Research from ME/CFS shows that there may be an immune change in the first few years of the illness, where cytokines decrease without any corresponding change in symptom improvement.
Because of this and the possibility that other markers follow the same pattern, disease duration should be a core feature of all analyses and trial designs. Trial outcomes should be designed to answer the question of whether the medication helps patients at different durations of illness.
10: Prioritize patient populations less likely to recover without intervention
Some long COVID phenotypes seem less likely to recover without intervention. Trials should take care to focus on these patient populations, which include those with neurologic symptoms and those meeting ME/CFS criteria.
11: Account for the relapsing/remitting nature
Outcome measures need to be assessed in a way that can distinguish a temporary remission, which is part of the natural course of the disease, from a permanent cure.
Factors that can contribute to the relapsing/remitting nature include physical and cognitive postexertional malaise, menstrual cycle changes, and seasonal changes.
12: Trial participants should reflect the diversity of the long COVID population
Certain demographics are more likely to be affected by acute and long COVID and need to be appropriately recruited and reflected in research, including in patient engagement.
Trials must include high numbers of Hispanic/Latinx, Black, and indigenous communities, queer and transgender populations, and women. Trial materials and design need to incorporate linguistic diversity in addition to racial/ethnic diversity.
Upward of 75% of long COVID cases happen after mild acute cases; clinical researchers should ensure that nonhospitalized patients make up the bulk of trial participants.
13: Utilize meaningful engagement of patients, especially in treatment selection and study design
Meaningful patient engagement means engaging multiple patients at every step of the trial process, from treatment selection to study design to analysis to communication of the results.
Patient experiences are extremely valuable and contain information that researchers may not be familiar with, including the nature and patterns of the illness, insights into possible treatments, and barriers to documentation and care that may also impact research. Tapping into those patient experiences will make trials stronger.
Overall, the landscape of long COVID clinical trials is ripe for discovery, and researchers choosing to go down this path will be deeply appreciated by the patient community.
Hannah Davis is a long COVID patient-researcher and cofounder of the Patient-Led Research Collaborative, an organization studying the long-term effects of COVID.
A version of this article first appeared on Medscape.com.
FDA expands use of dapagliflozin to broader range of HF
– including HF with mildly reduced ejection fraction (HFmrEF) and with preserved ejection fraction (HFpEF).
The sodium-glucose cotransporter 2 (SGLT2) inhibitor was previously approved in the United States for adults with heart failure with reduced ejection fraction (HFrEF).
The expanded indication is based on data from the phase 3 DELIVER trial, which showed clear clinical benefits of the SGLT2 inhibitor for patients with HF regardless of left ventricular function.
In the trial, which included more than 6,200 patients, dapagliflozin led to a statistically significant and clinically meaningful early reduction in the primary composite endpoint of cardiovascular (CV) death or worsening HF for patients with HFmrEF or HFpEFF.
In addition, results of a pooled analysis of the DAPA-HF and DELIVER phase 3 trials showed a consistent benefit from dapagliflozin treatment in significantly reducing the combined endpoint of CV death or HF hospitalization across the range of LVEF.
The European Commission expanded the indication for dapagliflozin (Forxiga) to include HF across the full spectrum of LVEF in February.
The SGLT2 inhibitor is also approved for use by patients with chronic kidney disease. It was first approved in 2014 to improve glycemic control for patients with diabetes mellitus.
A version of this article first appeared on Medscape.com.
– including HF with mildly reduced ejection fraction (HFmrEF) and with preserved ejection fraction (HFpEF).
The sodium-glucose cotransporter 2 (SGLT2) inhibitor was previously approved in the United States for adults with heart failure with reduced ejection fraction (HFrEF).
The expanded indication is based on data from the phase 3 DELIVER trial, which showed clear clinical benefits of the SGLT2 inhibitor for patients with HF regardless of left ventricular function.
In the trial, which included more than 6,200 patients, dapagliflozin led to a statistically significant and clinically meaningful early reduction in the primary composite endpoint of cardiovascular (CV) death or worsening HF for patients with HFmrEF or HFpEFF.
In addition, results of a pooled analysis of the DAPA-HF and DELIVER phase 3 trials showed a consistent benefit from dapagliflozin treatment in significantly reducing the combined endpoint of CV death or HF hospitalization across the range of LVEF.
The European Commission expanded the indication for dapagliflozin (Forxiga) to include HF across the full spectrum of LVEF in February.
The SGLT2 inhibitor is also approved for use by patients with chronic kidney disease. It was first approved in 2014 to improve glycemic control for patients with diabetes mellitus.
A version of this article first appeared on Medscape.com.
– including HF with mildly reduced ejection fraction (HFmrEF) and with preserved ejection fraction (HFpEF).
The sodium-glucose cotransporter 2 (SGLT2) inhibitor was previously approved in the United States for adults with heart failure with reduced ejection fraction (HFrEF).
The expanded indication is based on data from the phase 3 DELIVER trial, which showed clear clinical benefits of the SGLT2 inhibitor for patients with HF regardless of left ventricular function.
In the trial, which included more than 6,200 patients, dapagliflozin led to a statistically significant and clinically meaningful early reduction in the primary composite endpoint of cardiovascular (CV) death or worsening HF for patients with HFmrEF or HFpEFF.
In addition, results of a pooled analysis of the DAPA-HF and DELIVER phase 3 trials showed a consistent benefit from dapagliflozin treatment in significantly reducing the combined endpoint of CV death or HF hospitalization across the range of LVEF.
The European Commission expanded the indication for dapagliflozin (Forxiga) to include HF across the full spectrum of LVEF in February.
The SGLT2 inhibitor is also approved for use by patients with chronic kidney disease. It was first approved in 2014 to improve glycemic control for patients with diabetes mellitus.
A version of this article first appeared on Medscape.com.
AHA flags differing CVD risk in Asian American subgroups
Asian Americans have significant differences in genetics, socioeconomic factors, culture, diet, lifestyle, and acculturation levels based on the Asian region of their ancestry that likely have unique effects on their risk for type 2 diabetes and heart disease, the statement noted.
“Examining Asian subgroups separately is crucial to better understand the distinctions among them, how these differences translate into their risk of type 2 diabetes and atherosclerotic disease, and how health care professionals may provide care and support in a culturally appropriate manner,” writing group chair Tak W. Kwan, MD, chief of cardiology, Lenox Health Greenwich Village, and clinical professor of medicine, Northwell Health, New York City, said in a news release.
The statement was published online in the journal Circulation.
Impact on health outcomes
Asian American subgroups are broadly categorized by the geographic region of Asian descent and include South Asia (India, Pakistan, Sri Lanka, Bangladesh, Nepal, or Bhutan); East Asia (Japan, China, or Korea); Southeast Asia (Philippines, Vietnam, Thailand, Cambodia, Laos, Indonesia, Malaysia, Singapore, Hmong); and Native Hawaiian/Pacific Islander (Hawaii, Guam, Samoa, or other Pacific islands).
Asian Americans make up the fastest growing racial and ethnic group in the United States. Together, type 2 diabetes (T2D) and atherosclerotic cardiovascular disease (ASCVD) are the leading causes of illness and death among Asian American adults.
Yet, there is significant variability in prevalence and risk factors within the different subgroups, the writing group pointed out.
For example, based on available data, rates of coronary artery disease (CAD) among Asian Americans indicate an overall prevalence of 8% in men and about 3% in women.
However, available data for subgroups suggest higher CAD rates among Asian Indian Americans (13% for men and 4.4% for women) and Filipino Americans (about 9% and 4%, respectively).
Available data on T2D among Asian American subgroups also show varied prevalence and risk.
A study from California found overall, Asian American adults had higher rates of T2D (range of 15.6%-34.5%) compared with non-Hispanic White adults (12.8%). Among Chinese Americans, the rate was 15.8%. Among Korean and Japanese Americans, rates were about 18% and among Americans with Filipino ancestry, the rate was nearly 32%.
Yet most studies to date aggregate Asian Americans in a single group and do not examine the subgroups individually, which is a challenge to providing evidence-based recommendations, the writing group said.
“Particular attention should focus on the T2D and ASCVD risk differences among the different Asian American subgroups because they may affect the precision in clinical and health outcomes,” the group suggested.
“Culturally specific recommendations and interventions across the different Asian American subgroups related to T2D and ASCVD will help improve primary and secondary prevention and health outcomes in this population,” they added.
The writing group noted that existing CVD risk calculators, which are based on data validated in non-Hispanic Black adults and non-Hispanic White adults and less extensively studied in Asian Americans, may underestimate the risk of T2D and heart disease in South Asian adults, those of lower socioeconomic status, or those with chronic inflammatory diseases.
On the other hand, these tools may overestimate CVD risk among East Asians, those with higher socioeconomic status or those who are already participating in preventive healthcare services.
Advances in epidemiology and data analysis and the availability of larger, representative cohorts will allow for refinement of pooled cohort equations to better gauge ASCVD risk in Asian American subgroups, the group said.
Filling in the gaps
The writing group outlined several key areas to consider for strengthening the data about Asian American adults. Chief among them is the need to include disaggregated data on Asian American subgroups in clinical trials and government-sponsored studies.
Another is to standardize ways of collecting ethnic and subgroup data for Asian Americans for national health systems, surveys, and registries. National surveillance surveys should consider oversampling Asian Americans to increase representation for the various subgroups, the writing group suggested.
“All of us – health care professionals, policymakers, community leaders and patients – must advocate for more health research funding for Asian Americans and demand inclusion of Asian American subgroup information in clinical trials and government-sponsored research,” Dr. Kwan said.
“Having a platform to share and disseminate data on Asian Americans for the scientific and research community would also be an asset for the health care professionals who care for this population,” Dr. Kwan added.
The new scientific statement is a follow-up to a 2010 AHA “call to action” to seek data on health disparities among Asian American subgroups and a 2018 scientific statement addressing CVD risk in South Asians (Asian Indian, Pakistani, Sri Lankan, Bangladeshi, Nepali, or Bhutanese).
This scientific statement was prepared by the volunteer writing group on behalf of the AHA Council on Epidemiology and Prevention; the Council on Lifestyle and Cardiometabolic Health; the Council on Arteriosclerosis, Thrombosis and Vascular Biology; the Council on Clinical Cardiology; the Council on Cardiovascular and Stroke Nursing; and the Council on Genomic and Precision Medicine.
A version of this article first appeared on Medscape.com.
Asian Americans have significant differences in genetics, socioeconomic factors, culture, diet, lifestyle, and acculturation levels based on the Asian region of their ancestry that likely have unique effects on their risk for type 2 diabetes and heart disease, the statement noted.
“Examining Asian subgroups separately is crucial to better understand the distinctions among them, how these differences translate into their risk of type 2 diabetes and atherosclerotic disease, and how health care professionals may provide care and support in a culturally appropriate manner,” writing group chair Tak W. Kwan, MD, chief of cardiology, Lenox Health Greenwich Village, and clinical professor of medicine, Northwell Health, New York City, said in a news release.
The statement was published online in the journal Circulation.
Impact on health outcomes
Asian American subgroups are broadly categorized by the geographic region of Asian descent and include South Asia (India, Pakistan, Sri Lanka, Bangladesh, Nepal, or Bhutan); East Asia (Japan, China, or Korea); Southeast Asia (Philippines, Vietnam, Thailand, Cambodia, Laos, Indonesia, Malaysia, Singapore, Hmong); and Native Hawaiian/Pacific Islander (Hawaii, Guam, Samoa, or other Pacific islands).
Asian Americans make up the fastest growing racial and ethnic group in the United States. Together, type 2 diabetes (T2D) and atherosclerotic cardiovascular disease (ASCVD) are the leading causes of illness and death among Asian American adults.
Yet, there is significant variability in prevalence and risk factors within the different subgroups, the writing group pointed out.
For example, based on available data, rates of coronary artery disease (CAD) among Asian Americans indicate an overall prevalence of 8% in men and about 3% in women.
However, available data for subgroups suggest higher CAD rates among Asian Indian Americans (13% for men and 4.4% for women) and Filipino Americans (about 9% and 4%, respectively).
Available data on T2D among Asian American subgroups also show varied prevalence and risk.
A study from California found overall, Asian American adults had higher rates of T2D (range of 15.6%-34.5%) compared with non-Hispanic White adults (12.8%). Among Chinese Americans, the rate was 15.8%. Among Korean and Japanese Americans, rates were about 18% and among Americans with Filipino ancestry, the rate was nearly 32%.
Yet most studies to date aggregate Asian Americans in a single group and do not examine the subgroups individually, which is a challenge to providing evidence-based recommendations, the writing group said.
“Particular attention should focus on the T2D and ASCVD risk differences among the different Asian American subgroups because they may affect the precision in clinical and health outcomes,” the group suggested.
“Culturally specific recommendations and interventions across the different Asian American subgroups related to T2D and ASCVD will help improve primary and secondary prevention and health outcomes in this population,” they added.
The writing group noted that existing CVD risk calculators, which are based on data validated in non-Hispanic Black adults and non-Hispanic White adults and less extensively studied in Asian Americans, may underestimate the risk of T2D and heart disease in South Asian adults, those of lower socioeconomic status, or those with chronic inflammatory diseases.
On the other hand, these tools may overestimate CVD risk among East Asians, those with higher socioeconomic status or those who are already participating in preventive healthcare services.
Advances in epidemiology and data analysis and the availability of larger, representative cohorts will allow for refinement of pooled cohort equations to better gauge ASCVD risk in Asian American subgroups, the group said.
Filling in the gaps
The writing group outlined several key areas to consider for strengthening the data about Asian American adults. Chief among them is the need to include disaggregated data on Asian American subgroups in clinical trials and government-sponsored studies.
Another is to standardize ways of collecting ethnic and subgroup data for Asian Americans for national health systems, surveys, and registries. National surveillance surveys should consider oversampling Asian Americans to increase representation for the various subgroups, the writing group suggested.
“All of us – health care professionals, policymakers, community leaders and patients – must advocate for more health research funding for Asian Americans and demand inclusion of Asian American subgroup information in clinical trials and government-sponsored research,” Dr. Kwan said.
“Having a platform to share and disseminate data on Asian Americans for the scientific and research community would also be an asset for the health care professionals who care for this population,” Dr. Kwan added.
The new scientific statement is a follow-up to a 2010 AHA “call to action” to seek data on health disparities among Asian American subgroups and a 2018 scientific statement addressing CVD risk in South Asians (Asian Indian, Pakistani, Sri Lankan, Bangladeshi, Nepali, or Bhutanese).
This scientific statement was prepared by the volunteer writing group on behalf of the AHA Council on Epidemiology and Prevention; the Council on Lifestyle and Cardiometabolic Health; the Council on Arteriosclerosis, Thrombosis and Vascular Biology; the Council on Clinical Cardiology; the Council on Cardiovascular and Stroke Nursing; and the Council on Genomic and Precision Medicine.
A version of this article first appeared on Medscape.com.
Asian Americans have significant differences in genetics, socioeconomic factors, culture, diet, lifestyle, and acculturation levels based on the Asian region of their ancestry that likely have unique effects on their risk for type 2 diabetes and heart disease, the statement noted.
“Examining Asian subgroups separately is crucial to better understand the distinctions among them, how these differences translate into their risk of type 2 diabetes and atherosclerotic disease, and how health care professionals may provide care and support in a culturally appropriate manner,” writing group chair Tak W. Kwan, MD, chief of cardiology, Lenox Health Greenwich Village, and clinical professor of medicine, Northwell Health, New York City, said in a news release.
The statement was published online in the journal Circulation.
Impact on health outcomes
Asian American subgroups are broadly categorized by the geographic region of Asian descent and include South Asia (India, Pakistan, Sri Lanka, Bangladesh, Nepal, or Bhutan); East Asia (Japan, China, or Korea); Southeast Asia (Philippines, Vietnam, Thailand, Cambodia, Laos, Indonesia, Malaysia, Singapore, Hmong); and Native Hawaiian/Pacific Islander (Hawaii, Guam, Samoa, or other Pacific islands).
Asian Americans make up the fastest growing racial and ethnic group in the United States. Together, type 2 diabetes (T2D) and atherosclerotic cardiovascular disease (ASCVD) are the leading causes of illness and death among Asian American adults.
Yet, there is significant variability in prevalence and risk factors within the different subgroups, the writing group pointed out.
For example, based on available data, rates of coronary artery disease (CAD) among Asian Americans indicate an overall prevalence of 8% in men and about 3% in women.
However, available data for subgroups suggest higher CAD rates among Asian Indian Americans (13% for men and 4.4% for women) and Filipino Americans (about 9% and 4%, respectively).
Available data on T2D among Asian American subgroups also show varied prevalence and risk.
A study from California found overall, Asian American adults had higher rates of T2D (range of 15.6%-34.5%) compared with non-Hispanic White adults (12.8%). Among Chinese Americans, the rate was 15.8%. Among Korean and Japanese Americans, rates were about 18% and among Americans with Filipino ancestry, the rate was nearly 32%.
Yet most studies to date aggregate Asian Americans in a single group and do not examine the subgroups individually, which is a challenge to providing evidence-based recommendations, the writing group said.
“Particular attention should focus on the T2D and ASCVD risk differences among the different Asian American subgroups because they may affect the precision in clinical and health outcomes,” the group suggested.
“Culturally specific recommendations and interventions across the different Asian American subgroups related to T2D and ASCVD will help improve primary and secondary prevention and health outcomes in this population,” they added.
The writing group noted that existing CVD risk calculators, which are based on data validated in non-Hispanic Black adults and non-Hispanic White adults and less extensively studied in Asian Americans, may underestimate the risk of T2D and heart disease in South Asian adults, those of lower socioeconomic status, or those with chronic inflammatory diseases.
On the other hand, these tools may overestimate CVD risk among East Asians, those with higher socioeconomic status or those who are already participating in preventive healthcare services.
Advances in epidemiology and data analysis and the availability of larger, representative cohorts will allow for refinement of pooled cohort equations to better gauge ASCVD risk in Asian American subgroups, the group said.
Filling in the gaps
The writing group outlined several key areas to consider for strengthening the data about Asian American adults. Chief among them is the need to include disaggregated data on Asian American subgroups in clinical trials and government-sponsored studies.
Another is to standardize ways of collecting ethnic and subgroup data for Asian Americans for national health systems, surveys, and registries. National surveillance surveys should consider oversampling Asian Americans to increase representation for the various subgroups, the writing group suggested.
“All of us – health care professionals, policymakers, community leaders and patients – must advocate for more health research funding for Asian Americans and demand inclusion of Asian American subgroup information in clinical trials and government-sponsored research,” Dr. Kwan said.
“Having a platform to share and disseminate data on Asian Americans for the scientific and research community would also be an asset for the health care professionals who care for this population,” Dr. Kwan added.
The new scientific statement is a follow-up to a 2010 AHA “call to action” to seek data on health disparities among Asian American subgroups and a 2018 scientific statement addressing CVD risk in South Asians (Asian Indian, Pakistani, Sri Lankan, Bangladeshi, Nepali, or Bhutanese).
This scientific statement was prepared by the volunteer writing group on behalf of the AHA Council on Epidemiology and Prevention; the Council on Lifestyle and Cardiometabolic Health; the Council on Arteriosclerosis, Thrombosis and Vascular Biology; the Council on Clinical Cardiology; the Council on Cardiovascular and Stroke Nursing; and the Council on Genomic and Precision Medicine.
A version of this article first appeared on Medscape.com.
FROM CIRCULATION
New AACE type 2 diabetes algorithm individualizes care
SEATTLE – The latest American Association of Clinical Endocrinology type 2 diabetes management algorithm uses graphics to focus on individualized care while adding newly compiled information about medication access and affordability, vaccinations, and weight loss drugs.
The clinical guidance document was presented at the annual scientific & clinical congress of the American Association of Clinical Endocrinology and simultaneously published in Endocrine Practice.
Using text and colorful graphics, the document summarizes information from last year’s update and other recent AACE documents, including those addressing dyslipidemia and use of diabetes technology.
lead author Susan L. Samson, MD, PhD, chair of endocrinology, diabetes & metabolism at the Mayo Clinic Florida, Jacksonville, said in an interview.
Asked to comment, Anne L. Peters, MD, professor of clinical medicine at the University of Southern California, Los Angeles, said: “I like their simple graphics. For the Department of Health Services in Los Angeles County, we have been painstakingly trying to create our own flow diagrams. ... These will help.”
Eleven separate algorithms with text and graphics
Included are 11 visual management algorithms, with accompanying text for each one. The first lists 10 overall management principles, including “lifestyle modification underlies all therapy,” “maintain or achieve optimal weight,” “choice of therapy includes ease of use and access,” “individualize all glucose targets,” “avoid hypoglycemia,” and “comorbidities must be managed for comprehensive care.”
Three more algorithms cover the diabetes-adjacent topics of adiposity-based chronic disease, prediabetes, dyslipidemia, and hypertension.
Four separate graphics address glucose-lowering. Two are “complications-centric” and “glucose-centric” algorithms, another covers insulin initiation and titration, and a table summarizes the benefits and risks of currently available glucose-lowering medications, as well as cost.
Splitting the glucose-lowering algorithms into “complications-centric” and “glucose-centric” graphics is new, Dr. Samson said. “The complications one comes first, deliberately. You need to think about: Does my patient have a history of or high risk for cardiovascular disease, heart failure, stroke, or diabetic kidney disease? And, you want to prioritize those medications that have evidence to improve outcomes with those different diabetes complications versus a one-size-fits-all approach.”
And for patients without those complications, the glucose-centric algorithm considers obesity, hypoglycemia risk, and access/cost issues. “So, overall the diabetes medication algorithm has been split in order to emphasize that personalized approach to decision-making,” Dr. Samson explained.
Also new is a table listing the benefits and risks of weight-loss medications, and another covering immunization guidance for people with diabetes based on recommendations from the U.S. Centers for Disease Control and Prevention. “Coming out of the pandemic, we’re thinking about how can we protect our patients from infectious disease and all the comorbidities. In some cases, people with diabetes can have a much higher risk for adverse events,” Dr. Samson noted.
Regarding the weight-loss medications table, she pointed out that the task force couldn’t include the blockbuster twincretin tirzepatide because it’s not yet approved for weight loss by the U.S. Food and Drug Administration. However, it is included in the glucose-lowering drug table with weight loss listed among its benefits.
“We want this to be a living document that should be updated in a timely fashion, and so, as these new indications are approved and we see more evidence supporting their different uses, this should be updated in a really timely fashion to reflect that,” Dr. Samson said.
The end of the document includes a full page of each graphic, meant for wall posting.
Dr. Peters noted that for the most part, the AACE guidelines and algorithm align with joint guidance by the American Diabetes Association and European Association for the Study of Diabetes.
“For many years there seemed to be big differences between the AACE and ADA guidelines for the management of type 2 diabetes. Although small differences still exist ... the ADA and AACE guidelines have become quite similar,” she said.
Dr. Peters also praised the AACE algorithm for providing “a pathway for people who have issues with access and cost.”
“I am incredibly proud that in the County of Los Angeles you can get a [glucagon-like peptide-1 receptor agonist] and/or a [sodium-glucose cotransporter-2 inhibitor] even with the most restricted MediCal insurance if indications are met. But there remain many people in many places where access and cost limit options, and I am grateful that AACE includes this in their algorithms,” she said.
Dr. Samson has reported receiving research support to the Mayo Clinic from Corcept, serving on a steering committee and being a national or overall principal investigator for Chiasma and Novartis, and being a committee chair for the American Board of Internal Medicine. Dr. Peters has reported relationships with Blue Circle Health, Vertex, and Abbott Diabetes Care, receiving research grants from Abbott Diabetes Care and Insulet, and holding stock options in Teladoc and Omada Health.
A version of this article originally appeared on Medscape.com.
SEATTLE – The latest American Association of Clinical Endocrinology type 2 diabetes management algorithm uses graphics to focus on individualized care while adding newly compiled information about medication access and affordability, vaccinations, and weight loss drugs.
The clinical guidance document was presented at the annual scientific & clinical congress of the American Association of Clinical Endocrinology and simultaneously published in Endocrine Practice.
Using text and colorful graphics, the document summarizes information from last year’s update and other recent AACE documents, including those addressing dyslipidemia and use of diabetes technology.
lead author Susan L. Samson, MD, PhD, chair of endocrinology, diabetes & metabolism at the Mayo Clinic Florida, Jacksonville, said in an interview.
Asked to comment, Anne L. Peters, MD, professor of clinical medicine at the University of Southern California, Los Angeles, said: “I like their simple graphics. For the Department of Health Services in Los Angeles County, we have been painstakingly trying to create our own flow diagrams. ... These will help.”
Eleven separate algorithms with text and graphics
Included are 11 visual management algorithms, with accompanying text for each one. The first lists 10 overall management principles, including “lifestyle modification underlies all therapy,” “maintain or achieve optimal weight,” “choice of therapy includes ease of use and access,” “individualize all glucose targets,” “avoid hypoglycemia,” and “comorbidities must be managed for comprehensive care.”
Three more algorithms cover the diabetes-adjacent topics of adiposity-based chronic disease, prediabetes, dyslipidemia, and hypertension.
Four separate graphics address glucose-lowering. Two are “complications-centric” and “glucose-centric” algorithms, another covers insulin initiation and titration, and a table summarizes the benefits and risks of currently available glucose-lowering medications, as well as cost.
Splitting the glucose-lowering algorithms into “complications-centric” and “glucose-centric” graphics is new, Dr. Samson said. “The complications one comes first, deliberately. You need to think about: Does my patient have a history of or high risk for cardiovascular disease, heart failure, stroke, or diabetic kidney disease? And, you want to prioritize those medications that have evidence to improve outcomes with those different diabetes complications versus a one-size-fits-all approach.”
And for patients without those complications, the glucose-centric algorithm considers obesity, hypoglycemia risk, and access/cost issues. “So, overall the diabetes medication algorithm has been split in order to emphasize that personalized approach to decision-making,” Dr. Samson explained.
Also new is a table listing the benefits and risks of weight-loss medications, and another covering immunization guidance for people with diabetes based on recommendations from the U.S. Centers for Disease Control and Prevention. “Coming out of the pandemic, we’re thinking about how can we protect our patients from infectious disease and all the comorbidities. In some cases, people with diabetes can have a much higher risk for adverse events,” Dr. Samson noted.
Regarding the weight-loss medications table, she pointed out that the task force couldn’t include the blockbuster twincretin tirzepatide because it’s not yet approved for weight loss by the U.S. Food and Drug Administration. However, it is included in the glucose-lowering drug table with weight loss listed among its benefits.
“We want this to be a living document that should be updated in a timely fashion, and so, as these new indications are approved and we see more evidence supporting their different uses, this should be updated in a really timely fashion to reflect that,” Dr. Samson said.
The end of the document includes a full page of each graphic, meant for wall posting.
Dr. Peters noted that for the most part, the AACE guidelines and algorithm align with joint guidance by the American Diabetes Association and European Association for the Study of Diabetes.
“For many years there seemed to be big differences between the AACE and ADA guidelines for the management of type 2 diabetes. Although small differences still exist ... the ADA and AACE guidelines have become quite similar,” she said.
Dr. Peters also praised the AACE algorithm for providing “a pathway for people who have issues with access and cost.”
“I am incredibly proud that in the County of Los Angeles you can get a [glucagon-like peptide-1 receptor agonist] and/or a [sodium-glucose cotransporter-2 inhibitor] even with the most restricted MediCal insurance if indications are met. But there remain many people in many places where access and cost limit options, and I am grateful that AACE includes this in their algorithms,” she said.
Dr. Samson has reported receiving research support to the Mayo Clinic from Corcept, serving on a steering committee and being a national or overall principal investigator for Chiasma and Novartis, and being a committee chair for the American Board of Internal Medicine. Dr. Peters has reported relationships with Blue Circle Health, Vertex, and Abbott Diabetes Care, receiving research grants from Abbott Diabetes Care and Insulet, and holding stock options in Teladoc and Omada Health.
A version of this article originally appeared on Medscape.com.
SEATTLE – The latest American Association of Clinical Endocrinology type 2 diabetes management algorithm uses graphics to focus on individualized care while adding newly compiled information about medication access and affordability, vaccinations, and weight loss drugs.
The clinical guidance document was presented at the annual scientific & clinical congress of the American Association of Clinical Endocrinology and simultaneously published in Endocrine Practice.
Using text and colorful graphics, the document summarizes information from last year’s update and other recent AACE documents, including those addressing dyslipidemia and use of diabetes technology.
lead author Susan L. Samson, MD, PhD, chair of endocrinology, diabetes & metabolism at the Mayo Clinic Florida, Jacksonville, said in an interview.
Asked to comment, Anne L. Peters, MD, professor of clinical medicine at the University of Southern California, Los Angeles, said: “I like their simple graphics. For the Department of Health Services in Los Angeles County, we have been painstakingly trying to create our own flow diagrams. ... These will help.”
Eleven separate algorithms with text and graphics
Included are 11 visual management algorithms, with accompanying text for each one. The first lists 10 overall management principles, including “lifestyle modification underlies all therapy,” “maintain or achieve optimal weight,” “choice of therapy includes ease of use and access,” “individualize all glucose targets,” “avoid hypoglycemia,” and “comorbidities must be managed for comprehensive care.”
Three more algorithms cover the diabetes-adjacent topics of adiposity-based chronic disease, prediabetes, dyslipidemia, and hypertension.
Four separate graphics address glucose-lowering. Two are “complications-centric” and “glucose-centric” algorithms, another covers insulin initiation and titration, and a table summarizes the benefits and risks of currently available glucose-lowering medications, as well as cost.
Splitting the glucose-lowering algorithms into “complications-centric” and “glucose-centric” graphics is new, Dr. Samson said. “The complications one comes first, deliberately. You need to think about: Does my patient have a history of or high risk for cardiovascular disease, heart failure, stroke, or diabetic kidney disease? And, you want to prioritize those medications that have evidence to improve outcomes with those different diabetes complications versus a one-size-fits-all approach.”
And for patients without those complications, the glucose-centric algorithm considers obesity, hypoglycemia risk, and access/cost issues. “So, overall the diabetes medication algorithm has been split in order to emphasize that personalized approach to decision-making,” Dr. Samson explained.
Also new is a table listing the benefits and risks of weight-loss medications, and another covering immunization guidance for people with diabetes based on recommendations from the U.S. Centers for Disease Control and Prevention. “Coming out of the pandemic, we’re thinking about how can we protect our patients from infectious disease and all the comorbidities. In some cases, people with diabetes can have a much higher risk for adverse events,” Dr. Samson noted.
Regarding the weight-loss medications table, she pointed out that the task force couldn’t include the blockbuster twincretin tirzepatide because it’s not yet approved for weight loss by the U.S. Food and Drug Administration. However, it is included in the glucose-lowering drug table with weight loss listed among its benefits.
“We want this to be a living document that should be updated in a timely fashion, and so, as these new indications are approved and we see more evidence supporting their different uses, this should be updated in a really timely fashion to reflect that,” Dr. Samson said.
The end of the document includes a full page of each graphic, meant for wall posting.
Dr. Peters noted that for the most part, the AACE guidelines and algorithm align with joint guidance by the American Diabetes Association and European Association for the Study of Diabetes.
“For many years there seemed to be big differences between the AACE and ADA guidelines for the management of type 2 diabetes. Although small differences still exist ... the ADA and AACE guidelines have become quite similar,” she said.
Dr. Peters also praised the AACE algorithm for providing “a pathway for people who have issues with access and cost.”
“I am incredibly proud that in the County of Los Angeles you can get a [glucagon-like peptide-1 receptor agonist] and/or a [sodium-glucose cotransporter-2 inhibitor] even with the most restricted MediCal insurance if indications are met. But there remain many people in many places where access and cost limit options, and I am grateful that AACE includes this in their algorithms,” she said.
Dr. Samson has reported receiving research support to the Mayo Clinic from Corcept, serving on a steering committee and being a national or overall principal investigator for Chiasma and Novartis, and being a committee chair for the American Board of Internal Medicine. Dr. Peters has reported relationships with Blue Circle Health, Vertex, and Abbott Diabetes Care, receiving research grants from Abbott Diabetes Care and Insulet, and holding stock options in Teladoc and Omada Health.
A version of this article originally appeared on Medscape.com.
AT AACE 2023