User login
Octogenarians hard hit by insulin adverse events
CHICAGO – Older adults with diabetes are particularly vulnerable to insulin adverse event–related emergency department visits and hospitalizations, an analysis of nationally representative surveillance data has shown.
Compared with diabetes patients aged 45-64 years, those 80 years or older were 2.5 times more likely to present to the emergency department with an insulin adverse event (34.9 vs. 13.7/1,000 insulin-treated diabetes patients) and about five times more likely to be emergently hospitalized for such an event (16.1 vs. 3.3/1,000 persons).
"These data support setting glycemic targets based on risk/benefit," Dr. Andrew I. Geller of the Centers for Disease Control and Prevention said at the annual scientific sessions of the American Diabetes Association.
National guidelines are evolving in that direction. Earlier this year, the ADA recommended that older adults who are "functional, cognitively intact, and have significant life expectancy" should not have glycemic targets different from those of younger adults, but acknowledged that for some older adults, glycemic targets "might reasonably be relaxed, using individual criteria."
The American Association of Clinical Endocrinologists Diabetes Management Algorithm 2013 says that a hemoglobin A1c level "of 6.5% or less is still considered optimal if it can be achieved in a safe and affordable manner," but adds that the HbA1c target "must be individualized, based on numerous factors, such as age."
Dr. Geller suggested that reducing insulin adverse events in older adults may have a significant public health impact. Insulin is the most commonly implicated medicine in U.S. ED visits, and it’s linked to about 14,000 hospitalizations annually among adults 65 years or older. Insulins and synthetic analogues also accounted for 71% of spending growth on antidiabetes medications in 2011.
Dr. Geller and his colleagues estimated annual insulin use frequency with National Health Interview Survey data, and annual insulin adverse events with data from the National Electronic Injury Surveillance System–Cooperative Adverse Drug Event Surveillance (NEISS-CADES), an ongoing public health surveillance project based on chart abstraction, not ICD codes.
Insulin adverse events were defined as supratherapeutic effects (hypoglycemia) and therapeutic misadventures (for example, errors). Allergic reactions, local effects, and accidental exposure were excluded.
Insulin adverse events led to an estimated 97,745 emergency department visits annually from 2007 to 2011, accounting for 6.5% of all adverse drug event–related ED visits, Dr. Geller said.
While the sample had a slight predominance of males (50.4%) and a majority of whites (60.5%), there was no difference in rates among genders or races.
Patients aged 45-64 years had the highest proportion of ED visits (35%), followed by those 65-79 years (25.3%), 18-44 years (21.7%), 80 years or older (15.8%), and younger than 18 years (2.1%).
Still, patients 80 years or older had a rate of 34.9 ED visits per 1,000 diabetic persons, far surpassing rates observed among those younger than 18 years (13.7), 18-44 years (24.3), 45-64 years (13.7), and 65-79 years (16.3).
Patients aged 65-79 years had the highest proportion of hospitalizations (32.9%), but once again, those 80 years and older had the highest rate of hospitalizations per 1,000 diabetic persons (16.1), compared with the other age groups: younger than 18 years (2.5), 18-44 years (3.9), 45-64 years (3.3), and 65-79 years (6.2).
Based on clinical and laboratory findings, about 65% of patients presented with shock, loss of consciousness, or seizure (23.2%); fall or other injury (5%); and altered mental status (37%), Dr. Geller said. More than half of patients had a blood glucose level less than 50 mg/dL.
More than half (56%) of patients received intravenous dextrose or glucagon during their ED visit, while 29.3% of visits overall resulted in hospitalization, he said.
Precipitating factors were documented in about 21% of charts. The three most common were:
• Meal-related factors in 45.6%, such as a 19-year-old driver who took insulin 2 hours before hitting a tree, but had "no time to eat."
• Product mix-ups in 22%, including a 51-year-old injected by his spouse with 50 U of NovoLog instead of 50 U of Lantus.
• Incorrect dose or units in 12%, such as a 62-year-old who took 40 U of regular insulin instead of 4.
In cases where the patient took the wrong type of insulin, 52.3% of mix-ups involved a patient taking a rapid-acting agent instead of a long-acting one, Dr. Geller said.
"Perhaps the burden of insulin adverse events may be impacted by targeting at-risk individuals such as older adults, bolstering diabetes education such as meal planning, and minimizing the risk of product mix-ups, perhaps through packaging," he concluded.
Dr. Geller reported having no relevant financial disclosures.
CHICAGO – Older adults with diabetes are particularly vulnerable to insulin adverse event–related emergency department visits and hospitalizations, an analysis of nationally representative surveillance data has shown.
Compared with diabetes patients aged 45-64 years, those 80 years or older were 2.5 times more likely to present to the emergency department with an insulin adverse event (34.9 vs. 13.7/1,000 insulin-treated diabetes patients) and about five times more likely to be emergently hospitalized for such an event (16.1 vs. 3.3/1,000 persons).
"These data support setting glycemic targets based on risk/benefit," Dr. Andrew I. Geller of the Centers for Disease Control and Prevention said at the annual scientific sessions of the American Diabetes Association.
National guidelines are evolving in that direction. Earlier this year, the ADA recommended that older adults who are "functional, cognitively intact, and have significant life expectancy" should not have glycemic targets different from those of younger adults, but acknowledged that for some older adults, glycemic targets "might reasonably be relaxed, using individual criteria."
The American Association of Clinical Endocrinologists Diabetes Management Algorithm 2013 says that a hemoglobin A1c level "of 6.5% or less is still considered optimal if it can be achieved in a safe and affordable manner," but adds that the HbA1c target "must be individualized, based on numerous factors, such as age."
Dr. Geller suggested that reducing insulin adverse events in older adults may have a significant public health impact. Insulin is the most commonly implicated medicine in U.S. ED visits, and it’s linked to about 14,000 hospitalizations annually among adults 65 years or older. Insulins and synthetic analogues also accounted for 71% of spending growth on antidiabetes medications in 2011.
Dr. Geller and his colleagues estimated annual insulin use frequency with National Health Interview Survey data, and annual insulin adverse events with data from the National Electronic Injury Surveillance System–Cooperative Adverse Drug Event Surveillance (NEISS-CADES), an ongoing public health surveillance project based on chart abstraction, not ICD codes.
Insulin adverse events were defined as supratherapeutic effects (hypoglycemia) and therapeutic misadventures (for example, errors). Allergic reactions, local effects, and accidental exposure were excluded.
Insulin adverse events led to an estimated 97,745 emergency department visits annually from 2007 to 2011, accounting for 6.5% of all adverse drug event–related ED visits, Dr. Geller said.
While the sample had a slight predominance of males (50.4%) and a majority of whites (60.5%), there was no difference in rates among genders or races.
Patients aged 45-64 years had the highest proportion of ED visits (35%), followed by those 65-79 years (25.3%), 18-44 years (21.7%), 80 years or older (15.8%), and younger than 18 years (2.1%).
Still, patients 80 years or older had a rate of 34.9 ED visits per 1,000 diabetic persons, far surpassing rates observed among those younger than 18 years (13.7), 18-44 years (24.3), 45-64 years (13.7), and 65-79 years (16.3).
Patients aged 65-79 years had the highest proportion of hospitalizations (32.9%), but once again, those 80 years and older had the highest rate of hospitalizations per 1,000 diabetic persons (16.1), compared with the other age groups: younger than 18 years (2.5), 18-44 years (3.9), 45-64 years (3.3), and 65-79 years (6.2).
Based on clinical and laboratory findings, about 65% of patients presented with shock, loss of consciousness, or seizure (23.2%); fall or other injury (5%); and altered mental status (37%), Dr. Geller said. More than half of patients had a blood glucose level less than 50 mg/dL.
More than half (56%) of patients received intravenous dextrose or glucagon during their ED visit, while 29.3% of visits overall resulted in hospitalization, he said.
Precipitating factors were documented in about 21% of charts. The three most common were:
• Meal-related factors in 45.6%, such as a 19-year-old driver who took insulin 2 hours before hitting a tree, but had "no time to eat."
• Product mix-ups in 22%, including a 51-year-old injected by his spouse with 50 U of NovoLog instead of 50 U of Lantus.
• Incorrect dose or units in 12%, such as a 62-year-old who took 40 U of regular insulin instead of 4.
In cases where the patient took the wrong type of insulin, 52.3% of mix-ups involved a patient taking a rapid-acting agent instead of a long-acting one, Dr. Geller said.
"Perhaps the burden of insulin adverse events may be impacted by targeting at-risk individuals such as older adults, bolstering diabetes education such as meal planning, and minimizing the risk of product mix-ups, perhaps through packaging," he concluded.
Dr. Geller reported having no relevant financial disclosures.
CHICAGO – Older adults with diabetes are particularly vulnerable to insulin adverse event–related emergency department visits and hospitalizations, an analysis of nationally representative surveillance data has shown.
Compared with diabetes patients aged 45-64 years, those 80 years or older were 2.5 times more likely to present to the emergency department with an insulin adverse event (34.9 vs. 13.7/1,000 insulin-treated diabetes patients) and about five times more likely to be emergently hospitalized for such an event (16.1 vs. 3.3/1,000 persons).
"These data support setting glycemic targets based on risk/benefit," Dr. Andrew I. Geller of the Centers for Disease Control and Prevention said at the annual scientific sessions of the American Diabetes Association.
National guidelines are evolving in that direction. Earlier this year, the ADA recommended that older adults who are "functional, cognitively intact, and have significant life expectancy" should not have glycemic targets different from those of younger adults, but acknowledged that for some older adults, glycemic targets "might reasonably be relaxed, using individual criteria."
The American Association of Clinical Endocrinologists Diabetes Management Algorithm 2013 says that a hemoglobin A1c level "of 6.5% or less is still considered optimal if it can be achieved in a safe and affordable manner," but adds that the HbA1c target "must be individualized, based on numerous factors, such as age."
Dr. Geller suggested that reducing insulin adverse events in older adults may have a significant public health impact. Insulin is the most commonly implicated medicine in U.S. ED visits, and it’s linked to about 14,000 hospitalizations annually among adults 65 years or older. Insulins and synthetic analogues also accounted for 71% of spending growth on antidiabetes medications in 2011.
Dr. Geller and his colleagues estimated annual insulin use frequency with National Health Interview Survey data, and annual insulin adverse events with data from the National Electronic Injury Surveillance System–Cooperative Adverse Drug Event Surveillance (NEISS-CADES), an ongoing public health surveillance project based on chart abstraction, not ICD codes.
Insulin adverse events were defined as supratherapeutic effects (hypoglycemia) and therapeutic misadventures (for example, errors). Allergic reactions, local effects, and accidental exposure were excluded.
Insulin adverse events led to an estimated 97,745 emergency department visits annually from 2007 to 2011, accounting for 6.5% of all adverse drug event–related ED visits, Dr. Geller said.
While the sample had a slight predominance of males (50.4%) and a majority of whites (60.5%), there was no difference in rates among genders or races.
Patients aged 45-64 years had the highest proportion of ED visits (35%), followed by those 65-79 years (25.3%), 18-44 years (21.7%), 80 years or older (15.8%), and younger than 18 years (2.1%).
Still, patients 80 years or older had a rate of 34.9 ED visits per 1,000 diabetic persons, far surpassing rates observed among those younger than 18 years (13.7), 18-44 years (24.3), 45-64 years (13.7), and 65-79 years (16.3).
Patients aged 65-79 years had the highest proportion of hospitalizations (32.9%), but once again, those 80 years and older had the highest rate of hospitalizations per 1,000 diabetic persons (16.1), compared with the other age groups: younger than 18 years (2.5), 18-44 years (3.9), 45-64 years (3.3), and 65-79 years (6.2).
Based on clinical and laboratory findings, about 65% of patients presented with shock, loss of consciousness, or seizure (23.2%); fall or other injury (5%); and altered mental status (37%), Dr. Geller said. More than half of patients had a blood glucose level less than 50 mg/dL.
More than half (56%) of patients received intravenous dextrose or glucagon during their ED visit, while 29.3% of visits overall resulted in hospitalization, he said.
Precipitating factors were documented in about 21% of charts. The three most common were:
• Meal-related factors in 45.6%, such as a 19-year-old driver who took insulin 2 hours before hitting a tree, but had "no time to eat."
• Product mix-ups in 22%, including a 51-year-old injected by his spouse with 50 U of NovoLog instead of 50 U of Lantus.
• Incorrect dose or units in 12%, such as a 62-year-old who took 40 U of regular insulin instead of 4.
In cases where the patient took the wrong type of insulin, 52.3% of mix-ups involved a patient taking a rapid-acting agent instead of a long-acting one, Dr. Geller said.
"Perhaps the burden of insulin adverse events may be impacted by targeting at-risk individuals such as older adults, bolstering diabetes education such as meal planning, and minimizing the risk of product mix-ups, perhaps through packaging," he concluded.
Dr. Geller reported having no relevant financial disclosures.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major finding: Diabetes patients aged 80 years or older were 2.5 times more likely to present to the emergency department for an insulin adverse event and five times more likely to be hospitalized than diabetes patients aged 45-64 years.
Data source: An analysis of national surveillance data from 2007 to 2011.
Disclosures: Dr. Geller reported having no relevant financial disclosures.
Wet wipes linked to contact dermatitis in children
MILWAUKEE – Despite extensive testing, seemingly innocuous wet wipes have been linked to allergic contact dermatitis in children.
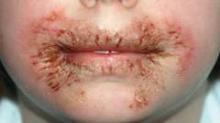
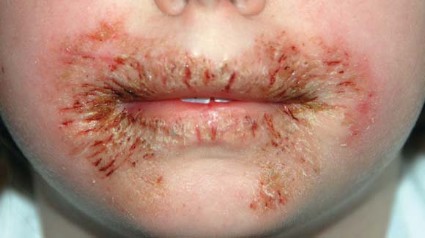
The first reported case occurred in a healthy 8-year-old girl who presented with chronic, recalcitrant erythematous, eczematous patches and plaques with crusting and fissuring around the mouth and perianal area. She had been using wipes containing methylisothiazolinone (MI), without methylchloroisothiazolinone (MCI).
Within 22 months, an additional five children had been diagnosed with allergic contact dermatitis to MI in wet wipes, Dr. Mary Wu Chang and Ms. Radhika Nakrani reported at the annual meeting of the Society for Pediatric Dermatology.
All cases were confirmed on patch testing, and all patients had rapid, complete resolution within about 2 days after discontinuing use of the wipes. Ms. Nakrani also interviewed the parents to determine the type of wipes used, identified as Cottonelle and Huggies brands (both manufactured by Kimberly-Clark). The preservative MI was identified in these brands.
"This is going to explode," Dr. Chang, a pediatric dermatologist at the University of Connecticut, West Hartford, said in an interview. "There’s more marketing [of wipes] now for non–diaper wearing children, and older people use wipes out of convenience."
The case series was described as the first report of pediatric allergic contact dermatitis to MI in wet wipes in the United States.
Although the MCI/MI patch test (T.R.U.E. Test) detected contact allergy to MI in the six children, identifying MI contact sensitization may require specialized MI patch tests with higher concentrations of MI or the use of small squares of the wipes themselves, Dr. Chang observed. Patch testing containing 100 ppm of MCI/MI mixture consists of only 25 ppm of MI and may be inadequate to detect MI alone, missing 33%-60% of cases.
Acute contact dermatitis was suspected in the index case, when the 8-year-old girl presented to pediatric dermatology after suffering for 11 months. She was initially misdiagnosed with impetiginized eczema and had received numerous oral and topical corticosteroids and antibiotics, said Dr. Chang. Questioning revealed that the patient used wet wipes for toileting and facial cleansing, and the rash resolved after the wipes were discontinued. MI sensitivity was confirmed on patch testing. Ms. Nakrani also interviewed the parents to determine the type of wipes used, and the preservative MI was identified in these brands.
"Remember to ask about wet wipe use, even in individuals who do not wear diapers and even if the rash in on the face," Dr. Chang said.
In the five other cases, allergic reactions to MI in the wet wipes were misdiagnosed as impetigo, psoriasis, diaper dermatitis, or atopic dermatitis, and unsuccessfully treated with a multitude of medications including steroids, antifungals, and topical tacrolimus (Protopic). The children, aged 3-8 years, had experienced symptoms for 1-12 months, according to the authors, who earned top honors for their poster presentation at the meeting.
The index patient also suffered from chronic retroauricular dermatitis, which resolved after discontinuing a shampoo containing MI.
MI was named the 2013 contact allergen of the year by the American Contact Dermatitis Society. In another recent study, Australian researchers reported 23 reactions to MI from a variety of personal care products, including 7 cases of hand dermatitis in parents of young children that was caused by an allergic reaction to MI in baby wipes (Australas. J. Dermatol. 2013 May 29 [doi: 10.1111/ajd.12062]).
The preservative MI was previously used only in a 3:1 MCI/MI combination (Kathon CG) that is widely known to cause allergic contact dermatitis. In an attempt to minimize such reactions, MI has been used alone, explained Ms. Nakrani, a medical student at the University of Connecticut.
However, its permitted concentration has increased by more than 25 times – from 3.7 ppm to 100 ppm – because it was thought to be a weaker sensitizer than MCI, she said.
"Once we found out the culprit was MI in the wet wipes and instructed parents to read labels and avoid MI, parents became more vigilant about the other products they were using like shampoos and soaps, and it really turned things right around once they stopped using these products," Ms. Nakrani said. "One mom, very grateful that the rashes finally cleared, said her child was going to school and everyone thought it was contagious."
The investigators have not contacted Kimberly-Clark regarding their findings. Bob Brand, director of external communications for Kimberly-Clark, would not specifically say whether they had received complaints of allergic contact dermatitis to MI in their wet wipes, but said all of their products undergo a thorough safety review prior to commercialization.
"While we understand there might be concern regarding a potential reaction to one of our products, consumers can use our wipes with confidence and know that the concentration levels of MI in our products are considered safe and well within the recommended levels as established by scientific and regulatory bodies such as the Cosmetic Ingredient Review Expert Panel in the U.S.A. and the European Commission Scientific Committee on Consumer Safety," he told Skin & Allergy News.
The formulation of the wipes appears to vary by product. Ms. Dianna Kenneally, principal scientist, baby care scientific communication, for Procter & Gamble, said in a separate interview that Procter & Gamble baby wipe brands do not contain MI.
Dr. Chang and Ms. Nakrani reported no relevant disclosures.
MILWAUKEE – Despite extensive testing, seemingly innocuous wet wipes have been linked to allergic contact dermatitis in children.
The first reported case occurred in a healthy 8-year-old girl who presented with chronic, recalcitrant erythematous, eczematous patches and plaques with crusting and fissuring around the mouth and perianal area. She had been using wipes containing methylisothiazolinone (MI), without methylchloroisothiazolinone (MCI).
Within 22 months, an additional five children had been diagnosed with allergic contact dermatitis to MI in wet wipes, Dr. Mary Wu Chang and Ms. Radhika Nakrani reported at the annual meeting of the Society for Pediatric Dermatology.
All cases were confirmed on patch testing, and all patients had rapid, complete resolution within about 2 days after discontinuing use of the wipes. Ms. Nakrani also interviewed the parents to determine the type of wipes used, identified as Cottonelle and Huggies brands (both manufactured by Kimberly-Clark). The preservative MI was identified in these brands.
"This is going to explode," Dr. Chang, a pediatric dermatologist at the University of Connecticut, West Hartford, said in an interview. "There’s more marketing [of wipes] now for non–diaper wearing children, and older people use wipes out of convenience."
The case series was described as the first report of pediatric allergic contact dermatitis to MI in wet wipes in the United States.
Although the MCI/MI patch test (T.R.U.E. Test) detected contact allergy to MI in the six children, identifying MI contact sensitization may require specialized MI patch tests with higher concentrations of MI or the use of small squares of the wipes themselves, Dr. Chang observed. Patch testing containing 100 ppm of MCI/MI mixture consists of only 25 ppm of MI and may be inadequate to detect MI alone, missing 33%-60% of cases.
Acute contact dermatitis was suspected in the index case, when the 8-year-old girl presented to pediatric dermatology after suffering for 11 months. She was initially misdiagnosed with impetiginized eczema and had received numerous oral and topical corticosteroids and antibiotics, said Dr. Chang. Questioning revealed that the patient used wet wipes for toileting and facial cleansing, and the rash resolved after the wipes were discontinued. MI sensitivity was confirmed on patch testing. Ms. Nakrani also interviewed the parents to determine the type of wipes used, and the preservative MI was identified in these brands.
"Remember to ask about wet wipe use, even in individuals who do not wear diapers and even if the rash in on the face," Dr. Chang said.
In the five other cases, allergic reactions to MI in the wet wipes were misdiagnosed as impetigo, psoriasis, diaper dermatitis, or atopic dermatitis, and unsuccessfully treated with a multitude of medications including steroids, antifungals, and topical tacrolimus (Protopic). The children, aged 3-8 years, had experienced symptoms for 1-12 months, according to the authors, who earned top honors for their poster presentation at the meeting.
The index patient also suffered from chronic retroauricular dermatitis, which resolved after discontinuing a shampoo containing MI.
MI was named the 2013 contact allergen of the year by the American Contact Dermatitis Society. In another recent study, Australian researchers reported 23 reactions to MI from a variety of personal care products, including 7 cases of hand dermatitis in parents of young children that was caused by an allergic reaction to MI in baby wipes (Australas. J. Dermatol. 2013 May 29 [doi: 10.1111/ajd.12062]).
The preservative MI was previously used only in a 3:1 MCI/MI combination (Kathon CG) that is widely known to cause allergic contact dermatitis. In an attempt to minimize such reactions, MI has been used alone, explained Ms. Nakrani, a medical student at the University of Connecticut.
However, its permitted concentration has increased by more than 25 times – from 3.7 ppm to 100 ppm – because it was thought to be a weaker sensitizer than MCI, she said.
"Once we found out the culprit was MI in the wet wipes and instructed parents to read labels and avoid MI, parents became more vigilant about the other products they were using like shampoos and soaps, and it really turned things right around once they stopped using these products," Ms. Nakrani said. "One mom, very grateful that the rashes finally cleared, said her child was going to school and everyone thought it was contagious."
The investigators have not contacted Kimberly-Clark regarding their findings. Bob Brand, director of external communications for Kimberly-Clark, would not specifically say whether they had received complaints of allergic contact dermatitis to MI in their wet wipes, but said all of their products undergo a thorough safety review prior to commercialization.
"While we understand there might be concern regarding a potential reaction to one of our products, consumers can use our wipes with confidence and know that the concentration levels of MI in our products are considered safe and well within the recommended levels as established by scientific and regulatory bodies such as the Cosmetic Ingredient Review Expert Panel in the U.S.A. and the European Commission Scientific Committee on Consumer Safety," he told Skin & Allergy News.
The formulation of the wipes appears to vary by product. Ms. Dianna Kenneally, principal scientist, baby care scientific communication, for Procter & Gamble, said in a separate interview that Procter & Gamble baby wipe brands do not contain MI.
Dr. Chang and Ms. Nakrani reported no relevant disclosures.
MILWAUKEE – Despite extensive testing, seemingly innocuous wet wipes have been linked to allergic contact dermatitis in children.
The first reported case occurred in a healthy 8-year-old girl who presented with chronic, recalcitrant erythematous, eczematous patches and plaques with crusting and fissuring around the mouth and perianal area. She had been using wipes containing methylisothiazolinone (MI), without methylchloroisothiazolinone (MCI).
Within 22 months, an additional five children had been diagnosed with allergic contact dermatitis to MI in wet wipes, Dr. Mary Wu Chang and Ms. Radhika Nakrani reported at the annual meeting of the Society for Pediatric Dermatology.
All cases were confirmed on patch testing, and all patients had rapid, complete resolution within about 2 days after discontinuing use of the wipes. Ms. Nakrani also interviewed the parents to determine the type of wipes used, identified as Cottonelle and Huggies brands (both manufactured by Kimberly-Clark). The preservative MI was identified in these brands.
"This is going to explode," Dr. Chang, a pediatric dermatologist at the University of Connecticut, West Hartford, said in an interview. "There’s more marketing [of wipes] now for non–diaper wearing children, and older people use wipes out of convenience."
The case series was described as the first report of pediatric allergic contact dermatitis to MI in wet wipes in the United States.
Although the MCI/MI patch test (T.R.U.E. Test) detected contact allergy to MI in the six children, identifying MI contact sensitization may require specialized MI patch tests with higher concentrations of MI or the use of small squares of the wipes themselves, Dr. Chang observed. Patch testing containing 100 ppm of MCI/MI mixture consists of only 25 ppm of MI and may be inadequate to detect MI alone, missing 33%-60% of cases.
Acute contact dermatitis was suspected in the index case, when the 8-year-old girl presented to pediatric dermatology after suffering for 11 months. She was initially misdiagnosed with impetiginized eczema and had received numerous oral and topical corticosteroids and antibiotics, said Dr. Chang. Questioning revealed that the patient used wet wipes for toileting and facial cleansing, and the rash resolved after the wipes were discontinued. MI sensitivity was confirmed on patch testing. Ms. Nakrani also interviewed the parents to determine the type of wipes used, and the preservative MI was identified in these brands.
"Remember to ask about wet wipe use, even in individuals who do not wear diapers and even if the rash in on the face," Dr. Chang said.
In the five other cases, allergic reactions to MI in the wet wipes were misdiagnosed as impetigo, psoriasis, diaper dermatitis, or atopic dermatitis, and unsuccessfully treated with a multitude of medications including steroids, antifungals, and topical tacrolimus (Protopic). The children, aged 3-8 years, had experienced symptoms for 1-12 months, according to the authors, who earned top honors for their poster presentation at the meeting.
The index patient also suffered from chronic retroauricular dermatitis, which resolved after discontinuing a shampoo containing MI.
MI was named the 2013 contact allergen of the year by the American Contact Dermatitis Society. In another recent study, Australian researchers reported 23 reactions to MI from a variety of personal care products, including 7 cases of hand dermatitis in parents of young children that was caused by an allergic reaction to MI in baby wipes (Australas. J. Dermatol. 2013 May 29 [doi: 10.1111/ajd.12062]).
The preservative MI was previously used only in a 3:1 MCI/MI combination (Kathon CG) that is widely known to cause allergic contact dermatitis. In an attempt to minimize such reactions, MI has been used alone, explained Ms. Nakrani, a medical student at the University of Connecticut.
However, its permitted concentration has increased by more than 25 times – from 3.7 ppm to 100 ppm – because it was thought to be a weaker sensitizer than MCI, she said.
"Once we found out the culprit was MI in the wet wipes and instructed parents to read labels and avoid MI, parents became more vigilant about the other products they were using like shampoos and soaps, and it really turned things right around once they stopped using these products," Ms. Nakrani said. "One mom, very grateful that the rashes finally cleared, said her child was going to school and everyone thought it was contagious."
The investigators have not contacted Kimberly-Clark regarding their findings. Bob Brand, director of external communications for Kimberly-Clark, would not specifically say whether they had received complaints of allergic contact dermatitis to MI in their wet wipes, but said all of their products undergo a thorough safety review prior to commercialization.
"While we understand there might be concern regarding a potential reaction to one of our products, consumers can use our wipes with confidence and know that the concentration levels of MI in our products are considered safe and well within the recommended levels as established by scientific and regulatory bodies such as the Cosmetic Ingredient Review Expert Panel in the U.S.A. and the European Commission Scientific Committee on Consumer Safety," he told Skin & Allergy News.
The formulation of the wipes appears to vary by product. Ms. Dianna Kenneally, principal scientist, baby care scientific communication, for Procter & Gamble, said in a separate interview that Procter & Gamble baby wipe brands do not contain MI.
Dr. Chang and Ms. Nakrani reported no relevant disclosures.
AT THE SPD ANNUAL MEETING
Major finding: Six cases of pediatric allergic contact dermatitis to methylisothiazolinone in wet wipes were confirmed on patch testing, and resolved within 1-2 days after discontinuing the wipes.
Data source: A case series of six children with allergic contact dermatitis to methylisothiazolinone.
Disclosures: Dr. Chang and Ms. Nakrani reported no relevant disclosures.
Methotrexate assay useful in severe pediatric dermatitis, psoriasis
MILWAUKEE – In children with severe atopic dermatitis or psoriasis, the methotrexate polyglutamate assay appears useful in guiding dose modification in those failing to respond to methotrexate at 12 weeks, a retrospective study has shown.
The methotrexate polyglutamate 3 assay (MTX PG3) measures concentrations of methotrexate polyglutamates, the active metabolites of methotrexate, Syed Rahman said at the annual meeting of the Society for Pediatric Dermatology.
Methotrexate, a prodrug, requires enzymatic conversion via sequential addition of glutamic acid residues. Those residues form a glutamate "tail" that enhances intracellular methotrexate retention. The 3-tail metabolite, MTX PG3, is the predominant species in most patients, explained Mr. Rahman of St. Louis University.
"This study supports the usefulness of a commercially available tool for customizing optimal methotrexate dosing, coauthor Dr. Elaine Siegfried, professor of pediatrics and dermatology at the university, said in an interview.
The MTX PG3 assay has been validated in adults with rheumatoid arthritis where patients with a therapeutic methotrexate polyglutamate level of more than 60 nmol/L were fivefold more likely to have a good response to methotrexate than those with levels below this threshold, according to poster results presented at the 2008 American College of Rheumatology annual meeting (cited on the company website). However, the test was not predictive of response in 36% of patients whose arthritis improved with treatment despite subtherapeutic levels.
For the current analysis, the investigators reviewed the charts of 46 children with severe atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap, who were treated with a starting dose of oral methotrexate 0.5 mg/kg per week (maximum, 15 mg) and assessed by MTX PG3 assay at week 12. In a subset of children who failed to respond, the dose was modified and the level rechecked at 8 weeks. Disease severity was rated using a 5-point physician global assessment scale, with a good to excellent response defined by a 2-point improvement.
In all, 83% of children achieved a good to excellent response, with 27 patients doing so within 12 weeks and 11 responding after dose adjustment involving either a higher oral dose or switching to subcutaneous administration, Mr. Rahman reported.
Response rates to methotrexate were slightly higher in children with psoriasis or overlap (the two groups were combined) than those with atopic dermatitis (94% vs. 77%; no P value given). Overall, 8 children failed to respond.
The mean MTX PG3 level significantly correlated with efficacy in patients with atopic dermatitis (responders, 34.8 nmol/L, vs. 17.8 nmol/L, nonresponders; P = .018), but not in those with psoriasis–atopic dermatitis overlap (responders, 26.5 nmol/L, vs. 20.7, nonresponders; P = .724), Mr. Rahman reported.
The data suggest that compared with the therapeutic level of 60 nmol/L identified in adults, "pediatric patients have a lower therapeutic value," he said.
A subset analysis of MTX PG3 levels based on response identified a significantly higher mean maximum MTX PG3 level for responders than nonresponders (31.5 vs. 18.1 nmol/L; P = .035).
More importantly, late responders had the highest methotrexate levels. All late responders achieved levels greater than 30 nmol/L, and mean levels were significantly higher than for early responders (41.9 vs. 27.3 nmol/L; P = .024), suggesting that the test is most valuable for the subset of children requiring dose adjustment, Mr. Rahman said.
"Patients who respond won’t even need the test, but for those at the 12-week juncture who do not respond and their levels are less than 30 nmol/L, this [study] suggests that if we increase the PO [dose] or change to subcutaneous [administration], we will eventually have a response," he said.
During a discussion of the results, an audience member questioned whether the test, with its added expense, really enhances clinical judgment because most clinicians would already adjust the dose in their patients failing to respond at 12 weeks.
Mr. Rahman replied that not all patients will respond to continued treatment with methotrexate, and that the test could differentiate between responders and nonresponders, thereby directing the patient to alternative therapy earlier.
Session moderator Dr. Amy Paller, chair of dermatology and professor of pediatrics and dermatology at Northwestern University in Chicago, agreed that the test is most valuable for the subset of children who are nonresponders at 12 weeks.
"In these children, this test can guide dose modification," she said in an interview. "Pharmacogenetic variation can require relatively higher oral doses or subcutaneous administration."
In contrast to a therapeutic level of more than 60 nmol/L established for adults, a level of 30 nmol/L may be sufficient in children, Dr. Paller said.
"A MTX PG3 less than 30 nmol/L in a child who has failed to respond within 12 weeks suggests that an increase in oral dose, or even a change to subcutaneous [administration], will boost efficacy," she commented.
Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
MILWAUKEE – In children with severe atopic dermatitis or psoriasis, the methotrexate polyglutamate assay appears useful in guiding dose modification in those failing to respond to methotrexate at 12 weeks, a retrospective study has shown.
The methotrexate polyglutamate 3 assay (MTX PG3) measures concentrations of methotrexate polyglutamates, the active metabolites of methotrexate, Syed Rahman said at the annual meeting of the Society for Pediatric Dermatology.
Methotrexate, a prodrug, requires enzymatic conversion via sequential addition of glutamic acid residues. Those residues form a glutamate "tail" that enhances intracellular methotrexate retention. The 3-tail metabolite, MTX PG3, is the predominant species in most patients, explained Mr. Rahman of St. Louis University.
"This study supports the usefulness of a commercially available tool for customizing optimal methotrexate dosing, coauthor Dr. Elaine Siegfried, professor of pediatrics and dermatology at the university, said in an interview.
The MTX PG3 assay has been validated in adults with rheumatoid arthritis where patients with a therapeutic methotrexate polyglutamate level of more than 60 nmol/L were fivefold more likely to have a good response to methotrexate than those with levels below this threshold, according to poster results presented at the 2008 American College of Rheumatology annual meeting (cited on the company website). However, the test was not predictive of response in 36% of patients whose arthritis improved with treatment despite subtherapeutic levels.
For the current analysis, the investigators reviewed the charts of 46 children with severe atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap, who were treated with a starting dose of oral methotrexate 0.5 mg/kg per week (maximum, 15 mg) and assessed by MTX PG3 assay at week 12. In a subset of children who failed to respond, the dose was modified and the level rechecked at 8 weeks. Disease severity was rated using a 5-point physician global assessment scale, with a good to excellent response defined by a 2-point improvement.
In all, 83% of children achieved a good to excellent response, with 27 patients doing so within 12 weeks and 11 responding after dose adjustment involving either a higher oral dose or switching to subcutaneous administration, Mr. Rahman reported.
Response rates to methotrexate were slightly higher in children with psoriasis or overlap (the two groups were combined) than those with atopic dermatitis (94% vs. 77%; no P value given). Overall, 8 children failed to respond.
The mean MTX PG3 level significantly correlated with efficacy in patients with atopic dermatitis (responders, 34.8 nmol/L, vs. 17.8 nmol/L, nonresponders; P = .018), but not in those with psoriasis–atopic dermatitis overlap (responders, 26.5 nmol/L, vs. 20.7, nonresponders; P = .724), Mr. Rahman reported.
The data suggest that compared with the therapeutic level of 60 nmol/L identified in adults, "pediatric patients have a lower therapeutic value," he said.
A subset analysis of MTX PG3 levels based on response identified a significantly higher mean maximum MTX PG3 level for responders than nonresponders (31.5 vs. 18.1 nmol/L; P = .035).
More importantly, late responders had the highest methotrexate levels. All late responders achieved levels greater than 30 nmol/L, and mean levels were significantly higher than for early responders (41.9 vs. 27.3 nmol/L; P = .024), suggesting that the test is most valuable for the subset of children requiring dose adjustment, Mr. Rahman said.
"Patients who respond won’t even need the test, but for those at the 12-week juncture who do not respond and their levels are less than 30 nmol/L, this [study] suggests that if we increase the PO [dose] or change to subcutaneous [administration], we will eventually have a response," he said.
During a discussion of the results, an audience member questioned whether the test, with its added expense, really enhances clinical judgment because most clinicians would already adjust the dose in their patients failing to respond at 12 weeks.
Mr. Rahman replied that not all patients will respond to continued treatment with methotrexate, and that the test could differentiate between responders and nonresponders, thereby directing the patient to alternative therapy earlier.
Session moderator Dr. Amy Paller, chair of dermatology and professor of pediatrics and dermatology at Northwestern University in Chicago, agreed that the test is most valuable for the subset of children who are nonresponders at 12 weeks.
"In these children, this test can guide dose modification," she said in an interview. "Pharmacogenetic variation can require relatively higher oral doses or subcutaneous administration."
In contrast to a therapeutic level of more than 60 nmol/L established for adults, a level of 30 nmol/L may be sufficient in children, Dr. Paller said.
"A MTX PG3 less than 30 nmol/L in a child who has failed to respond within 12 weeks suggests that an increase in oral dose, or even a change to subcutaneous [administration], will boost efficacy," she commented.
Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
MILWAUKEE – In children with severe atopic dermatitis or psoriasis, the methotrexate polyglutamate assay appears useful in guiding dose modification in those failing to respond to methotrexate at 12 weeks, a retrospective study has shown.
The methotrexate polyglutamate 3 assay (MTX PG3) measures concentrations of methotrexate polyglutamates, the active metabolites of methotrexate, Syed Rahman said at the annual meeting of the Society for Pediatric Dermatology.
Methotrexate, a prodrug, requires enzymatic conversion via sequential addition of glutamic acid residues. Those residues form a glutamate "tail" that enhances intracellular methotrexate retention. The 3-tail metabolite, MTX PG3, is the predominant species in most patients, explained Mr. Rahman of St. Louis University.
"This study supports the usefulness of a commercially available tool for customizing optimal methotrexate dosing, coauthor Dr. Elaine Siegfried, professor of pediatrics and dermatology at the university, said in an interview.
The MTX PG3 assay has been validated in adults with rheumatoid arthritis where patients with a therapeutic methotrexate polyglutamate level of more than 60 nmol/L were fivefold more likely to have a good response to methotrexate than those with levels below this threshold, according to poster results presented at the 2008 American College of Rheumatology annual meeting (cited on the company website). However, the test was not predictive of response in 36% of patients whose arthritis improved with treatment despite subtherapeutic levels.
For the current analysis, the investigators reviewed the charts of 46 children with severe atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap, who were treated with a starting dose of oral methotrexate 0.5 mg/kg per week (maximum, 15 mg) and assessed by MTX PG3 assay at week 12. In a subset of children who failed to respond, the dose was modified and the level rechecked at 8 weeks. Disease severity was rated using a 5-point physician global assessment scale, with a good to excellent response defined by a 2-point improvement.
In all, 83% of children achieved a good to excellent response, with 27 patients doing so within 12 weeks and 11 responding after dose adjustment involving either a higher oral dose or switching to subcutaneous administration, Mr. Rahman reported.
Response rates to methotrexate were slightly higher in children with psoriasis or overlap (the two groups were combined) than those with atopic dermatitis (94% vs. 77%; no P value given). Overall, 8 children failed to respond.
The mean MTX PG3 level significantly correlated with efficacy in patients with atopic dermatitis (responders, 34.8 nmol/L, vs. 17.8 nmol/L, nonresponders; P = .018), but not in those with psoriasis–atopic dermatitis overlap (responders, 26.5 nmol/L, vs. 20.7, nonresponders; P = .724), Mr. Rahman reported.
The data suggest that compared with the therapeutic level of 60 nmol/L identified in adults, "pediatric patients have a lower therapeutic value," he said.
A subset analysis of MTX PG3 levels based on response identified a significantly higher mean maximum MTX PG3 level for responders than nonresponders (31.5 vs. 18.1 nmol/L; P = .035).
More importantly, late responders had the highest methotrexate levels. All late responders achieved levels greater than 30 nmol/L, and mean levels were significantly higher than for early responders (41.9 vs. 27.3 nmol/L; P = .024), suggesting that the test is most valuable for the subset of children requiring dose adjustment, Mr. Rahman said.
"Patients who respond won’t even need the test, but for those at the 12-week juncture who do not respond and their levels are less than 30 nmol/L, this [study] suggests that if we increase the PO [dose] or change to subcutaneous [administration], we will eventually have a response," he said.
During a discussion of the results, an audience member questioned whether the test, with its added expense, really enhances clinical judgment because most clinicians would already adjust the dose in their patients failing to respond at 12 weeks.
Mr. Rahman replied that not all patients will respond to continued treatment with methotrexate, and that the test could differentiate between responders and nonresponders, thereby directing the patient to alternative therapy earlier.
Session moderator Dr. Amy Paller, chair of dermatology and professor of pediatrics and dermatology at Northwestern University in Chicago, agreed that the test is most valuable for the subset of children who are nonresponders at 12 weeks.
"In these children, this test can guide dose modification," she said in an interview. "Pharmacogenetic variation can require relatively higher oral doses or subcutaneous administration."
In contrast to a therapeutic level of more than 60 nmol/L established for adults, a level of 30 nmol/L may be sufficient in children, Dr. Paller said.
"A MTX PG3 less than 30 nmol/L in a child who has failed to respond within 12 weeks suggests that an increase in oral dose, or even a change to subcutaneous [administration], will boost efficacy," she commented.
Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
AT THE ANNUAL MEETING OF THE SOCIETY FOR PEDIATRIC DERMATOLOGY
Methotrexate assay useful in severe pediatric dermatitis, psoriasis
MILWAUKEE – In children with severe atopic dermatitis or psoriasis, the methotrexate polyglutamate assay appears useful in guiding dose modification in those failing to respond to methotrexate at 12 weeks, a retrospective study has shown.
The methotrexate polyglutamate 3 assay (MTX PG3) measures concentrations of methotrexate polyglutamates, the active metabolites of methotrexate, Syed Rahman said at the annual meeting of the Society for Pediatric Dermatology.
Methotrexate, a prodrug, requires enzymatic conversion via sequential addition of glutamic acid residues. Those residues form a glutamate "tail" that enhances intracellular methotrexate retention. The 3-tail metabolite, MTX PG3, is the predominant species in most patients, explained Mr. Rahman of St. Louis University.
"This study supports the usefulness of a commercially available tool for customizing optimal methotrexate dosing, coauthor Dr. Elaine Siegfried, professor of pediatrics and dermatology at the university, said in an interview.
The MTX PG3 assay has been validated in adults with rheumatoid arthritis where patients with a therapeutic methotrexate polyglutamate level of more than 60 nmol/L were fivefold more likely to have a good response to methotrexate than those with levels below this threshold, according to poster results presented at the 2008 American College of Rheumatology annual meeting (cited on the company website). However, the test was not predictive of response in 36% of patients whose arthritis improved with treatment despite subtherapeutic levels.
For the current analysis, the investigators reviewed the charts of 46 children with severe atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap, who were treated with a starting dose of oral methotrexate 0.5 mg/kg per week (maximum, 15 mg) and assessed by MTX PG3 assay at week 12. In a subset of children who failed to respond, the dose was modified and the level rechecked at 8 weeks. Disease severity was rated using a 5-point physician global assessment scale, with a good to excellent response defined by a 2-point improvement.
In all, 83% of children achieved a good to excellent response, with 27 patients doing so within 12 weeks and 11 responding after dose adjustment involving either a higher oral dose or switching to subcutaneous administration, Mr. Rahman reported.
Response rates to methotrexate were slightly higher in children with psoriasis or overlap (the two groups were combined) than those with atopic dermatitis (94% vs. 77%; no P value given). Overall, 8 children failed to respond.
The mean MTX PG3 level significantly correlated with efficacy in patients with atopic dermatitis (responders, 34.8 nmol/L, vs. 17.8 nmol/L, nonresponders; P = .018), but not in those with psoriasis–atopic dermatitis overlap (responders, 26.5 nmol/L, vs. 20.7, nonresponders; P = .724), Mr. Rahman reported.
The data suggest that compared with the therapeutic level of 60 nmol/L identified in adults, "pediatric patients have a lower therapeutic value," he said.
A subset analysis of MTX PG3 levels based on response identified a significantly higher mean maximum MTX PG3 level for responders than nonresponders (31.5 vs. 18.1 nmol/L; P = .035).
More importantly, late responders had the highest methotrexate levels. All late responders achieved levels greater than 30 nmol/L, and mean levels were significantly higher than for early responders (41.9 vs. 27.3 nmol/L; P = .024), suggesting that the test is most valuable for the subset of children requiring dose adjustment, Mr. Rahman said.
"Patients who respond won’t even need the test, but for those at the 12-week juncture who do not respond and their levels are less than 30 nmol/L, this [study] suggests that if we increase the PO [dose] or change to subcutaneous [administration], we will eventually have a response," he said.
During a discussion of the results, an audience member questioned whether the test, with its added expense, really enhances clinical judgment because most clinicians would already adjust the dose in their patients failing to respond at 12 weeks.
Mr. Rahman replied that not all patients will respond to continued treatment with methotrexate, and that the test could differentiate between responders and nonresponders, thereby directing the patient to alternative therapy earlier.
Session moderator Dr. Amy Paller, chair of dermatology and professor of pediatrics and dermatology at Northwestern University in Chicago, agreed that the test is most valuable for the subset of children who are nonresponders at 12 weeks.
"In these children, this test can guide dose modification," she said in an interview. "Pharmacogenetic variation can require relatively higher oral doses or subcutaneous administration."
In contrast to a therapeutic level of more than 60 nmol/L established for adults, a level of 30 nmol/L may be sufficient in children, Dr. Paller said.
"A MTX PG3 less than 30 nmol/L in a child who has failed to respond within 12 weeks suggests that an increase in oral dose, or even a change to subcutaneous [administration], will boost efficacy," she commented.
Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
MILWAUKEE – In children with severe atopic dermatitis or psoriasis, the methotrexate polyglutamate assay appears useful in guiding dose modification in those failing to respond to methotrexate at 12 weeks, a retrospective study has shown.
The methotrexate polyglutamate 3 assay (MTX PG3) measures concentrations of methotrexate polyglutamates, the active metabolites of methotrexate, Syed Rahman said at the annual meeting of the Society for Pediatric Dermatology.
Methotrexate, a prodrug, requires enzymatic conversion via sequential addition of glutamic acid residues. Those residues form a glutamate "tail" that enhances intracellular methotrexate retention. The 3-tail metabolite, MTX PG3, is the predominant species in most patients, explained Mr. Rahman of St. Louis University.
"This study supports the usefulness of a commercially available tool for customizing optimal methotrexate dosing, coauthor Dr. Elaine Siegfried, professor of pediatrics and dermatology at the university, said in an interview.
The MTX PG3 assay has been validated in adults with rheumatoid arthritis where patients with a therapeutic methotrexate polyglutamate level of more than 60 nmol/L were fivefold more likely to have a good response to methotrexate than those with levels below this threshold, according to poster results presented at the 2008 American College of Rheumatology annual meeting (cited on the company website). However, the test was not predictive of response in 36% of patients whose arthritis improved with treatment despite subtherapeutic levels.
For the current analysis, the investigators reviewed the charts of 46 children with severe atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap, who were treated with a starting dose of oral methotrexate 0.5 mg/kg per week (maximum, 15 mg) and assessed by MTX PG3 assay at week 12. In a subset of children who failed to respond, the dose was modified and the level rechecked at 8 weeks. Disease severity was rated using a 5-point physician global assessment scale, with a good to excellent response defined by a 2-point improvement.
In all, 83% of children achieved a good to excellent response, with 27 patients doing so within 12 weeks and 11 responding after dose adjustment involving either a higher oral dose or switching to subcutaneous administration, Mr. Rahman reported.
Response rates to methotrexate were slightly higher in children with psoriasis or overlap (the two groups were combined) than those with atopic dermatitis (94% vs. 77%; no P value given). Overall, 8 children failed to respond.
The mean MTX PG3 level significantly correlated with efficacy in patients with atopic dermatitis (responders, 34.8 nmol/L, vs. 17.8 nmol/L, nonresponders; P = .018), but not in those with psoriasis–atopic dermatitis overlap (responders, 26.5 nmol/L, vs. 20.7, nonresponders; P = .724), Mr. Rahman reported.
The data suggest that compared with the therapeutic level of 60 nmol/L identified in adults, "pediatric patients have a lower therapeutic value," he said.
A subset analysis of MTX PG3 levels based on response identified a significantly higher mean maximum MTX PG3 level for responders than nonresponders (31.5 vs. 18.1 nmol/L; P = .035).
More importantly, late responders had the highest methotrexate levels. All late responders achieved levels greater than 30 nmol/L, and mean levels were significantly higher than for early responders (41.9 vs. 27.3 nmol/L; P = .024), suggesting that the test is most valuable for the subset of children requiring dose adjustment, Mr. Rahman said.
"Patients who respond won’t even need the test, but for those at the 12-week juncture who do not respond and their levels are less than 30 nmol/L, this [study] suggests that if we increase the PO [dose] or change to subcutaneous [administration], we will eventually have a response," he said.
During a discussion of the results, an audience member questioned whether the test, with its added expense, really enhances clinical judgment because most clinicians would already adjust the dose in their patients failing to respond at 12 weeks.
Mr. Rahman replied that not all patients will respond to continued treatment with methotrexate, and that the test could differentiate between responders and nonresponders, thereby directing the patient to alternative therapy earlier.
Session moderator Dr. Amy Paller, chair of dermatology and professor of pediatrics and dermatology at Northwestern University in Chicago, agreed that the test is most valuable for the subset of children who are nonresponders at 12 weeks.
"In these children, this test can guide dose modification," she said in an interview. "Pharmacogenetic variation can require relatively higher oral doses or subcutaneous administration."
In contrast to a therapeutic level of more than 60 nmol/L established for adults, a level of 30 nmol/L may be sufficient in children, Dr. Paller said.
"A MTX PG3 less than 30 nmol/L in a child who has failed to respond within 12 weeks suggests that an increase in oral dose, or even a change to subcutaneous [administration], will boost efficacy," she commented.
Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
MILWAUKEE – In children with severe atopic dermatitis or psoriasis, the methotrexate polyglutamate assay appears useful in guiding dose modification in those failing to respond to methotrexate at 12 weeks, a retrospective study has shown.
The methotrexate polyglutamate 3 assay (MTX PG3) measures concentrations of methotrexate polyglutamates, the active metabolites of methotrexate, Syed Rahman said at the annual meeting of the Society for Pediatric Dermatology.
Methotrexate, a prodrug, requires enzymatic conversion via sequential addition of glutamic acid residues. Those residues form a glutamate "tail" that enhances intracellular methotrexate retention. The 3-tail metabolite, MTX PG3, is the predominant species in most patients, explained Mr. Rahman of St. Louis University.
"This study supports the usefulness of a commercially available tool for customizing optimal methotrexate dosing, coauthor Dr. Elaine Siegfried, professor of pediatrics and dermatology at the university, said in an interview.
The MTX PG3 assay has been validated in adults with rheumatoid arthritis where patients with a therapeutic methotrexate polyglutamate level of more than 60 nmol/L were fivefold more likely to have a good response to methotrexate than those with levels below this threshold, according to poster results presented at the 2008 American College of Rheumatology annual meeting (cited on the company website). However, the test was not predictive of response in 36% of patients whose arthritis improved with treatment despite subtherapeutic levels.
For the current analysis, the investigators reviewed the charts of 46 children with severe atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap, who were treated with a starting dose of oral methotrexate 0.5 mg/kg per week (maximum, 15 mg) and assessed by MTX PG3 assay at week 12. In a subset of children who failed to respond, the dose was modified and the level rechecked at 8 weeks. Disease severity was rated using a 5-point physician global assessment scale, with a good to excellent response defined by a 2-point improvement.
In all, 83% of children achieved a good to excellent response, with 27 patients doing so within 12 weeks and 11 responding after dose adjustment involving either a higher oral dose or switching to subcutaneous administration, Mr. Rahman reported.
Response rates to methotrexate were slightly higher in children with psoriasis or overlap (the two groups were combined) than those with atopic dermatitis (94% vs. 77%; no P value given). Overall, 8 children failed to respond.
The mean MTX PG3 level significantly correlated with efficacy in patients with atopic dermatitis (responders, 34.8 nmol/L, vs. 17.8 nmol/L, nonresponders; P = .018), but not in those with psoriasis–atopic dermatitis overlap (responders, 26.5 nmol/L, vs. 20.7, nonresponders; P = .724), Mr. Rahman reported.
The data suggest that compared with the therapeutic level of 60 nmol/L identified in adults, "pediatric patients have a lower therapeutic value," he said.
A subset analysis of MTX PG3 levels based on response identified a significantly higher mean maximum MTX PG3 level for responders than nonresponders (31.5 vs. 18.1 nmol/L; P = .035).
More importantly, late responders had the highest methotrexate levels. All late responders achieved levels greater than 30 nmol/L, and mean levels were significantly higher than for early responders (41.9 vs. 27.3 nmol/L; P = .024), suggesting that the test is most valuable for the subset of children requiring dose adjustment, Mr. Rahman said.
"Patients who respond won’t even need the test, but for those at the 12-week juncture who do not respond and their levels are less than 30 nmol/L, this [study] suggests that if we increase the PO [dose] or change to subcutaneous [administration], we will eventually have a response," he said.
During a discussion of the results, an audience member questioned whether the test, with its added expense, really enhances clinical judgment because most clinicians would already adjust the dose in their patients failing to respond at 12 weeks.
Mr. Rahman replied that not all patients will respond to continued treatment with methotrexate, and that the test could differentiate between responders and nonresponders, thereby directing the patient to alternative therapy earlier.
Session moderator Dr. Amy Paller, chair of dermatology and professor of pediatrics and dermatology at Northwestern University in Chicago, agreed that the test is most valuable for the subset of children who are nonresponders at 12 weeks.
"In these children, this test can guide dose modification," she said in an interview. "Pharmacogenetic variation can require relatively higher oral doses or subcutaneous administration."
In contrast to a therapeutic level of more than 60 nmol/L established for adults, a level of 30 nmol/L may be sufficient in children, Dr. Paller said.
"A MTX PG3 less than 30 nmol/L in a child who has failed to respond within 12 weeks suggests that an increase in oral dose, or even a change to subcutaneous [administration], will boost efficacy," she commented.
Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
AT THE ANNUAL MEETING OF THE SOCIETY FOR PEDIATRIC DERMATOLOGY
Major finding: Late responders had the highest methotrexate levels (41.9 nmol/L vs. 27.3 nmol/L in early responders; P = .024) on the MTX PG3 assay.
Data source: A retrospective study of 46 children on methotrexate for atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap who were assessed by MTX PG3 assay.
Disclosures: Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
Case builds for counting fat, protein alongside carbs in T1DM
CHICAGO – Meals high in fat or protein cause hyperglycemia among children using intensive insulin therapy for type 1 diabetes, a study has shown.
Further, protein and fat have an additive effect on postprandial glycemic rise, based on results of the randomized study involving 33 children.
"Protein and fat should be considered in prandial insulin dose and distribution," Carmel Smart, Ph.D., reported at the annual scientific sessions of the American Diabetes Association.
In intensive insulin therapy, the mealtime insulin dose is calculated based exclusively on counting carbohydrates. A growing body of evidence, however, suggests that macronutrients other than carbohydrates influence postprandial glucose levels and insulin requirements, said Dr. Smart, a specialist diabetes dietitian at the John Hunter Children’s Hospital, Newcastle, Australia.
She highlighted a recent study showing that high-fat meals with identical carbohydrate and protein content, required more insulin than low-fat meals and, despite the additional insulin, caused more hyperglycemia in type 1 diabetics (Diabetes Care 2013;36:810-6). An early study reported that meals with added protein significantly increased late glucose responses and insulin requirements, compared with standard or fat-added meals (Am. J. Clin. Nutr. 1993;58:555-60).
For the current experiment, 33 children (aged 8-17) ate four test pancake breakfasts on different days with identical carbohydrate content (30 g), but varying protein and fat: low fat (4 g)/low protein (5 g), high fat (35 g)/low protein, low fat/high protein (40 g), and high fat/high protein. A protein supplement and double cream (50% fat) were used to vary the protein and fat content.
The meals were given after the same individually standardized insulin dose, and no activity or snacks were allowed for 5 hours after the meal. Glucose excursions or fluctuations were measured at 30-minute increments for 5 hours using continuous glucose monitoring.
At baseline, the 17 girls and 16 boys had a mean A1c level of 7.2% and a mean duration of diabetes of 4.9 years; 27 were on continuous subcutaneous insulin infusion, and 6 were on a multiple daily insulin regimen. Their average age was 12.2 years.
Compared with the low-protein/low-fat meal, postprandial glucose excursions were significantly greater at 210-300 minutes after the high-fat/low-protein meal (1.78 mmol/L vs. –0.50 mmol/L; P = .01), Dr. Smart said.
Postprandial glucose excursions also were significantly greater at 180 minutes after the high-protein/low-fat meal (2.40 mmol/L vs. 0.54 mmol/L; P = .02).
The high-fat/high-protein meal resulted in significantly higher and sustained glucose excursions from 180 to 300 minutes compared with all the other meals (P less than .04).
The effect of fat and protein was additive at all time points after 150 minutes, she said. Compared with the low-fat/low-protein meal, the average glucose excursion was 4.2 mmol/L greater at 180 minutes after the high-fat/high-protein meal and 5.4 mmol/L greater at 5 hours.
Hypoglycemia, defined by a blood glucose level of less than 3.6 mmol/L, occurred in 29 children in the 5-hour postprandial period, and was significantly different between meal types (P = .003), Dr. Smart said.
There was a significant reduction in hypoglycemia after high-protein meals (odds ratio, 0.16; P less than .0001), but no reduction after high-fat meals (OR, 0.50; P = .08).
While meals high in protein and fat cause hyperglycemia, "our results suggest that protein may have a protective effect on hypoglycemia," she said.
During a discussion of the study, an audience member thanked the investigators for conducting the study "because this is what I’ve been seeing in patients eating high-fat diets, and I haven’t known exactly what to do about their insulin when they aren’t on the pump."
Dr. Smart agreed that they’ve been seeing this phenomenon for a long time, and said clinicians can now have the confidence to tell patients that it’s not inaccurate carbohydrate counting causing the glucose fluctuations, as previously believed. Currently, the investigators are advising patients on pump therapy to use the dual-wave bolus, although novel algorithms to determine prandial glucose dosing are being refined for high-fat/high-protein diets, she added.
A study led by Kirstine Bell, Ph.D., detailing one of those algorithms, called the food insulin index, was presented during the same session, by Dr. Stephen Colagiuri.
The study was supported by grants from Australian Pediatric Endocrine Care and Hunter Children’s Research Foundation. Medtronic provided the glucose-monitoring equipment. Dr. Smart reported having no relevant financial disclosures.
At the American Diabetes Association scientific sessions, two studies were presented in adolescents with type 1 diabetes that show that postmeal glucose excursions were influenced by more than carbohydrates.
In adolescents with type 1 diabetes, higher fat and protein intake contributed to the rise in postmeal glucose levels using a continual glucose monitoring system, with less hypoglycemia after a higher-protein meal. Suggestions included using a dual wave bolus in pump patients before they eat a high fat meal or developing a new way besides carbohydrate counting to guesstimate insulin bolus requirements.
Similar in concept to the glycemic index, the food insulin demand (FID) is an algorithm for ranking foods based on their insulin demand and portion size relative to a reference food, and it takes into consideration protein intake.
It's likely that calculation of premeal insulin needs will consider a variety of factors to decrease the glucose excursions throughout the day: total food composition (carbohydrate, fat, and protein content) of anticipated meal, portion size, timing of when to bolus, adjustments for the premeal level of glucose (currently called the "correction factor"), anticipated exercise and stress post meal.
In other words, we have a long way to go to accurately predict glucose excursions and anticipate insulin requirements.
Dr. Jay Cohen is medical director of the Endocrine Clinic, Memphis, Tenn. He said that he had no financial disclosures relevant to this article.
At the American Diabetes Association scientific sessions, two studies were presented in adolescents with type 1 diabetes that show that postmeal glucose excursions were influenced by more than carbohydrates.
In adolescents with type 1 diabetes, higher fat and protein intake contributed to the rise in postmeal glucose levels using a continual glucose monitoring system, with less hypoglycemia after a higher-protein meal. Suggestions included using a dual wave bolus in pump patients before they eat a high fat meal or developing a new way besides carbohydrate counting to guesstimate insulin bolus requirements.
Similar in concept to the glycemic index, the food insulin demand (FID) is an algorithm for ranking foods based on their insulin demand and portion size relative to a reference food, and it takes into consideration protein intake.
It's likely that calculation of premeal insulin needs will consider a variety of factors to decrease the glucose excursions throughout the day: total food composition (carbohydrate, fat, and protein content) of anticipated meal, portion size, timing of when to bolus, adjustments for the premeal level of glucose (currently called the "correction factor"), anticipated exercise and stress post meal.
In other words, we have a long way to go to accurately predict glucose excursions and anticipate insulin requirements.
Dr. Jay Cohen is medical director of the Endocrine Clinic, Memphis, Tenn. He said that he had no financial disclosures relevant to this article.
At the American Diabetes Association scientific sessions, two studies were presented in adolescents with type 1 diabetes that show that postmeal glucose excursions were influenced by more than carbohydrates.
In adolescents with type 1 diabetes, higher fat and protein intake contributed to the rise in postmeal glucose levels using a continual glucose monitoring system, with less hypoglycemia after a higher-protein meal. Suggestions included using a dual wave bolus in pump patients before they eat a high fat meal or developing a new way besides carbohydrate counting to guesstimate insulin bolus requirements.
Similar in concept to the glycemic index, the food insulin demand (FID) is an algorithm for ranking foods based on their insulin demand and portion size relative to a reference food, and it takes into consideration protein intake.
It's likely that calculation of premeal insulin needs will consider a variety of factors to decrease the glucose excursions throughout the day: total food composition (carbohydrate, fat, and protein content) of anticipated meal, portion size, timing of when to bolus, adjustments for the premeal level of glucose (currently called the "correction factor"), anticipated exercise and stress post meal.
In other words, we have a long way to go to accurately predict glucose excursions and anticipate insulin requirements.
Dr. Jay Cohen is medical director of the Endocrine Clinic, Memphis, Tenn. He said that he had no financial disclosures relevant to this article.
CHICAGO – Meals high in fat or protein cause hyperglycemia among children using intensive insulin therapy for type 1 diabetes, a study has shown.
Further, protein and fat have an additive effect on postprandial glycemic rise, based on results of the randomized study involving 33 children.
"Protein and fat should be considered in prandial insulin dose and distribution," Carmel Smart, Ph.D., reported at the annual scientific sessions of the American Diabetes Association.
In intensive insulin therapy, the mealtime insulin dose is calculated based exclusively on counting carbohydrates. A growing body of evidence, however, suggests that macronutrients other than carbohydrates influence postprandial glucose levels and insulin requirements, said Dr. Smart, a specialist diabetes dietitian at the John Hunter Children’s Hospital, Newcastle, Australia.
She highlighted a recent study showing that high-fat meals with identical carbohydrate and protein content, required more insulin than low-fat meals and, despite the additional insulin, caused more hyperglycemia in type 1 diabetics (Diabetes Care 2013;36:810-6). An early study reported that meals with added protein significantly increased late glucose responses and insulin requirements, compared with standard or fat-added meals (Am. J. Clin. Nutr. 1993;58:555-60).
For the current experiment, 33 children (aged 8-17) ate four test pancake breakfasts on different days with identical carbohydrate content (30 g), but varying protein and fat: low fat (4 g)/low protein (5 g), high fat (35 g)/low protein, low fat/high protein (40 g), and high fat/high protein. A protein supplement and double cream (50% fat) were used to vary the protein and fat content.
The meals were given after the same individually standardized insulin dose, and no activity or snacks were allowed for 5 hours after the meal. Glucose excursions or fluctuations were measured at 30-minute increments for 5 hours using continuous glucose monitoring.
At baseline, the 17 girls and 16 boys had a mean A1c level of 7.2% and a mean duration of diabetes of 4.9 years; 27 were on continuous subcutaneous insulin infusion, and 6 were on a multiple daily insulin regimen. Their average age was 12.2 years.
Compared with the low-protein/low-fat meal, postprandial glucose excursions were significantly greater at 210-300 minutes after the high-fat/low-protein meal (1.78 mmol/L vs. –0.50 mmol/L; P = .01), Dr. Smart said.
Postprandial glucose excursions also were significantly greater at 180 minutes after the high-protein/low-fat meal (2.40 mmol/L vs. 0.54 mmol/L; P = .02).
The high-fat/high-protein meal resulted in significantly higher and sustained glucose excursions from 180 to 300 minutes compared with all the other meals (P less than .04).
The effect of fat and protein was additive at all time points after 150 minutes, she said. Compared with the low-fat/low-protein meal, the average glucose excursion was 4.2 mmol/L greater at 180 minutes after the high-fat/high-protein meal and 5.4 mmol/L greater at 5 hours.
Hypoglycemia, defined by a blood glucose level of less than 3.6 mmol/L, occurred in 29 children in the 5-hour postprandial period, and was significantly different between meal types (P = .003), Dr. Smart said.
There was a significant reduction in hypoglycemia after high-protein meals (odds ratio, 0.16; P less than .0001), but no reduction after high-fat meals (OR, 0.50; P = .08).
While meals high in protein and fat cause hyperglycemia, "our results suggest that protein may have a protective effect on hypoglycemia," she said.
During a discussion of the study, an audience member thanked the investigators for conducting the study "because this is what I’ve been seeing in patients eating high-fat diets, and I haven’t known exactly what to do about their insulin when they aren’t on the pump."
Dr. Smart agreed that they’ve been seeing this phenomenon for a long time, and said clinicians can now have the confidence to tell patients that it’s not inaccurate carbohydrate counting causing the glucose fluctuations, as previously believed. Currently, the investigators are advising patients on pump therapy to use the dual-wave bolus, although novel algorithms to determine prandial glucose dosing are being refined for high-fat/high-protein diets, she added.
A study led by Kirstine Bell, Ph.D., detailing one of those algorithms, called the food insulin index, was presented during the same session, by Dr. Stephen Colagiuri.
The study was supported by grants from Australian Pediatric Endocrine Care and Hunter Children’s Research Foundation. Medtronic provided the glucose-monitoring equipment. Dr. Smart reported having no relevant financial disclosures.
CHICAGO – Meals high in fat or protein cause hyperglycemia among children using intensive insulin therapy for type 1 diabetes, a study has shown.
Further, protein and fat have an additive effect on postprandial glycemic rise, based on results of the randomized study involving 33 children.
"Protein and fat should be considered in prandial insulin dose and distribution," Carmel Smart, Ph.D., reported at the annual scientific sessions of the American Diabetes Association.
In intensive insulin therapy, the mealtime insulin dose is calculated based exclusively on counting carbohydrates. A growing body of evidence, however, suggests that macronutrients other than carbohydrates influence postprandial glucose levels and insulin requirements, said Dr. Smart, a specialist diabetes dietitian at the John Hunter Children’s Hospital, Newcastle, Australia.
She highlighted a recent study showing that high-fat meals with identical carbohydrate and protein content, required more insulin than low-fat meals and, despite the additional insulin, caused more hyperglycemia in type 1 diabetics (Diabetes Care 2013;36:810-6). An early study reported that meals with added protein significantly increased late glucose responses and insulin requirements, compared with standard or fat-added meals (Am. J. Clin. Nutr. 1993;58:555-60).
For the current experiment, 33 children (aged 8-17) ate four test pancake breakfasts on different days with identical carbohydrate content (30 g), but varying protein and fat: low fat (4 g)/low protein (5 g), high fat (35 g)/low protein, low fat/high protein (40 g), and high fat/high protein. A protein supplement and double cream (50% fat) were used to vary the protein and fat content.
The meals were given after the same individually standardized insulin dose, and no activity or snacks were allowed for 5 hours after the meal. Glucose excursions or fluctuations were measured at 30-minute increments for 5 hours using continuous glucose monitoring.
At baseline, the 17 girls and 16 boys had a mean A1c level of 7.2% and a mean duration of diabetes of 4.9 years; 27 were on continuous subcutaneous insulin infusion, and 6 were on a multiple daily insulin regimen. Their average age was 12.2 years.
Compared with the low-protein/low-fat meal, postprandial glucose excursions were significantly greater at 210-300 minutes after the high-fat/low-protein meal (1.78 mmol/L vs. –0.50 mmol/L; P = .01), Dr. Smart said.
Postprandial glucose excursions also were significantly greater at 180 minutes after the high-protein/low-fat meal (2.40 mmol/L vs. 0.54 mmol/L; P = .02).
The high-fat/high-protein meal resulted in significantly higher and sustained glucose excursions from 180 to 300 minutes compared with all the other meals (P less than .04).
The effect of fat and protein was additive at all time points after 150 minutes, she said. Compared with the low-fat/low-protein meal, the average glucose excursion was 4.2 mmol/L greater at 180 minutes after the high-fat/high-protein meal and 5.4 mmol/L greater at 5 hours.
Hypoglycemia, defined by a blood glucose level of less than 3.6 mmol/L, occurred in 29 children in the 5-hour postprandial period, and was significantly different between meal types (P = .003), Dr. Smart said.
There was a significant reduction in hypoglycemia after high-protein meals (odds ratio, 0.16; P less than .0001), but no reduction after high-fat meals (OR, 0.50; P = .08).
While meals high in protein and fat cause hyperglycemia, "our results suggest that protein may have a protective effect on hypoglycemia," she said.
During a discussion of the study, an audience member thanked the investigators for conducting the study "because this is what I’ve been seeing in patients eating high-fat diets, and I haven’t known exactly what to do about their insulin when they aren’t on the pump."
Dr. Smart agreed that they’ve been seeing this phenomenon for a long time, and said clinicians can now have the confidence to tell patients that it’s not inaccurate carbohydrate counting causing the glucose fluctuations, as previously believed. Currently, the investigators are advising patients on pump therapy to use the dual-wave bolus, although novel algorithms to determine prandial glucose dosing are being refined for high-fat/high-protein diets, she added.
A study led by Kirstine Bell, Ph.D., detailing one of those algorithms, called the food insulin index, was presented during the same session, by Dr. Stephen Colagiuri.
The study was supported by grants from Australian Pediatric Endocrine Care and Hunter Children’s Research Foundation. Medtronic provided the glucose-monitoring equipment. Dr. Smart reported having no relevant financial disclosures.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major finding: A high-fat/high-protein meal resulted in significantly higher glucose excursions from 180 to 300 minutes, compared with all the other meals (P less than .04).
Data source: A randomized study in 33 children using intensive insulin therapy for type 1 diabetes.
Disclosures: The study was supported by grants from Australian Pediatric Endocrine Care and Hunter Children’s Research Foundation. Medtronic provided the glucose-monitoring equipment. Dr. Smart reported having no relevant financial disclosures.
Two meals per day better than six in T2DM
CHICAGO – Overweight patients with type 2 diabetes who ate only breakfast and lunch lost more weight than did those eating six meals a day in a randomized, crossover study.
They also had greater decreases in fasting plasma glucose, C-peptide, glucagon, and hepatic fat content, and bigger improvements in insulin sensitivity.
"Our data suggest that consuming a hearty breakfast and lunch may be more beneficial for patients with type 2 diabetes than eating more, smaller meals during the day," Dr. Hana Kahleová, said at the annual scientific sessions of the American Diabetes Association.
When asked whether eating a big lunch and dinner would produce the same benefits given the difficulty clinicians would face convincing patients to give up their evening meal, Dr. Kahleová agreed it would be a hard sell, but said the breakfast/lunch schedule is more beneficial. She highlighted research during the presentation showing fat deposition is greater following dinner (J. Clin. Endocrinol. Metab. 2009;94:1781-8), and that diabetes-free people of Mediterranean heritage who ate most of their daily intake early lost more weight and did so at a faster rate than did late eaters (Int. J. Obes. [Lond.] 2013;37:604-11).
"Our results support the ancient proverb: Eat breakfast like a king, lunch like a prince, and dinner like a pauper," said Dr. Kahleová of the Diabetes Centre, Institute for Clinical and Experimental Medicine, Prague.
Session moderator Dr. Anastassios Pittas, codirector of the Diabetes Center at Tufts Medical Center in Boston, said in an interview that few clinicians are in favor of six small meals a day because invariably patients will eat six moderate-size meals. Insulin dosing with such frequent meals is also more onerous.
"I agree with the finding that two is better than six, but that does not imply that, in general, the concept of fewer meals is better than more meals," he said. "It’s just that the comparison groups were so extreme that it’s logical to me what happened. I think the better, clinically applicable comparison would have been two vs. four or maybe five meals."
The six-meal diet was chosen as the comparator because it has been usually recommended that patients with type 2 diabetes eat five to six small meals during the day, Dr. Kahleová said in an interview. Although diabetic patients who eat small regular meals are reported to have better control of their blood glucose concentrations, the current results are strongly supported by animal studies showing the antidiabetic effects of caloric restriction and intermittent fasting diets. These effects include reduced blood glucose and insulin concentrations and improved glucose tolerance in rodents and are thought to be driven largely by increased insulin sensitivity.
Dr. Kahleová and her associates evenly randomized 54 patients with type 2 diabetes for more than 1 year to follow a six meals/day or two meals/day diet for 12 weeks. The diets were then switched for a subsequent 12 weeks. Both diets had the same caloric restriction (–500 kcal/day), carbohydrates (50%-55%), proteins (20%-25%), fats (25%-30%), cholesterol (less than 200 mg/day), and fiber (30g-40g/day).
For the two-meal diet, breakfast was eaten between hours 6-10 and lunch between hours 12-16. In the six-meal diet, breakfast was eaten during hours 6-8, snack hours 8-10, lunch hours 11-13, second snack hours 14-15, dinner hours 17-18 and second dinner hours 20-21.
To improve compliance, each regimen was started with a 4-day tutorial on how to compose and prepare their diet; group meetings with a dietician; individual counseling and meals provided for half of the patients. Patients were also asked not to alter their exercise habits during the study.
At baseline, 54% of patients were female, average diabetes duration 8.1 years, average body mass index 32.6 kg/m2, and average hemoglobin A1c 7.2%. Their average age was 59.4 years.
At 12 weeks, the mean change in BMI significantly favored the two-meal diet over the six-meal diet (–1.23 kg/m2 vs.–0.82 kg/m2; P less than .001), Dr. Kahleová said. Waist circumference shrunk significantly more as well (–5.14 cm vs. –1.37 cm; P less than .001).
Fasting plasma glucose decreased in response to both diets, but the decrease was significantly greater with two meals per day (–0.78 mmol/L vs. –0.47 mmol/L; P = .004), she said. C-peptide followed the same pattern (P = .04).
Hepatic fat content, as measured by proton magnetic resonance spectroscopy, decreased significantly more with two meals per day (–4.2% vs. –3.4%; P less than .001).
HbA1c decreased comparably in both diets, Dr. Kahleová said. Fasting plasma glucagon decreased with two daily meals (P less than .001), while it increased with six meals (P = .04).
Both parameters of beta-cell function – insulin secretion and glucose sensitivity – increased comparably with the two diets, Dr. Kahleová said. Changes in glucose sensitivity (P = .02) and oral glucose insulin sensitivity (P less than .001) correlated negatively with the change in hepatic fat content. The correlations were no longer significant, however, after adjustment for changes in BMI.
Dr. Kahleová said the results were as expected, and she declined to speculate as to whether they would be the same if the cohort had been heavier than allowed with the BMI inclusion criteria of 27-40 kg/m2.
The investigators are currently trying to find a link between postprandial oxidative stress and gastrointestinal peptides in patients with type 2 diabetes.
The study was supported by a grant from the Ministry of Health, Prague, and by the Grant Agency of Charles University, also in Prague. Dr. Kahleová reported having no financial disclosures.
CHICAGO – Overweight patients with type 2 diabetes who ate only breakfast and lunch lost more weight than did those eating six meals a day in a randomized, crossover study.
They also had greater decreases in fasting plasma glucose, C-peptide, glucagon, and hepatic fat content, and bigger improvements in insulin sensitivity.
"Our data suggest that consuming a hearty breakfast and lunch may be more beneficial for patients with type 2 diabetes than eating more, smaller meals during the day," Dr. Hana Kahleová, said at the annual scientific sessions of the American Diabetes Association.
When asked whether eating a big lunch and dinner would produce the same benefits given the difficulty clinicians would face convincing patients to give up their evening meal, Dr. Kahleová agreed it would be a hard sell, but said the breakfast/lunch schedule is more beneficial. She highlighted research during the presentation showing fat deposition is greater following dinner (J. Clin. Endocrinol. Metab. 2009;94:1781-8), and that diabetes-free people of Mediterranean heritage who ate most of their daily intake early lost more weight and did so at a faster rate than did late eaters (Int. J. Obes. [Lond.] 2013;37:604-11).
"Our results support the ancient proverb: Eat breakfast like a king, lunch like a prince, and dinner like a pauper," said Dr. Kahleová of the Diabetes Centre, Institute for Clinical and Experimental Medicine, Prague.
Session moderator Dr. Anastassios Pittas, codirector of the Diabetes Center at Tufts Medical Center in Boston, said in an interview that few clinicians are in favor of six small meals a day because invariably patients will eat six moderate-size meals. Insulin dosing with such frequent meals is also more onerous.
"I agree with the finding that two is better than six, but that does not imply that, in general, the concept of fewer meals is better than more meals," he said. "It’s just that the comparison groups were so extreme that it’s logical to me what happened. I think the better, clinically applicable comparison would have been two vs. four or maybe five meals."
The six-meal diet was chosen as the comparator because it has been usually recommended that patients with type 2 diabetes eat five to six small meals during the day, Dr. Kahleová said in an interview. Although diabetic patients who eat small regular meals are reported to have better control of their blood glucose concentrations, the current results are strongly supported by animal studies showing the antidiabetic effects of caloric restriction and intermittent fasting diets. These effects include reduced blood glucose and insulin concentrations and improved glucose tolerance in rodents and are thought to be driven largely by increased insulin sensitivity.
Dr. Kahleová and her associates evenly randomized 54 patients with type 2 diabetes for more than 1 year to follow a six meals/day or two meals/day diet for 12 weeks. The diets were then switched for a subsequent 12 weeks. Both diets had the same caloric restriction (–500 kcal/day), carbohydrates (50%-55%), proteins (20%-25%), fats (25%-30%), cholesterol (less than 200 mg/day), and fiber (30g-40g/day).
For the two-meal diet, breakfast was eaten between hours 6-10 and lunch between hours 12-16. In the six-meal diet, breakfast was eaten during hours 6-8, snack hours 8-10, lunch hours 11-13, second snack hours 14-15, dinner hours 17-18 and second dinner hours 20-21.
To improve compliance, each regimen was started with a 4-day tutorial on how to compose and prepare their diet; group meetings with a dietician; individual counseling and meals provided for half of the patients. Patients were also asked not to alter their exercise habits during the study.
At baseline, 54% of patients were female, average diabetes duration 8.1 years, average body mass index 32.6 kg/m2, and average hemoglobin A1c 7.2%. Their average age was 59.4 years.
At 12 weeks, the mean change in BMI significantly favored the two-meal diet over the six-meal diet (–1.23 kg/m2 vs.–0.82 kg/m2; P less than .001), Dr. Kahleová said. Waist circumference shrunk significantly more as well (–5.14 cm vs. –1.37 cm; P less than .001).
Fasting plasma glucose decreased in response to both diets, but the decrease was significantly greater with two meals per day (–0.78 mmol/L vs. –0.47 mmol/L; P = .004), she said. C-peptide followed the same pattern (P = .04).
Hepatic fat content, as measured by proton magnetic resonance spectroscopy, decreased significantly more with two meals per day (–4.2% vs. –3.4%; P less than .001).
HbA1c decreased comparably in both diets, Dr. Kahleová said. Fasting plasma glucagon decreased with two daily meals (P less than .001), while it increased with six meals (P = .04).
Both parameters of beta-cell function – insulin secretion and glucose sensitivity – increased comparably with the two diets, Dr. Kahleová said. Changes in glucose sensitivity (P = .02) and oral glucose insulin sensitivity (P less than .001) correlated negatively with the change in hepatic fat content. The correlations were no longer significant, however, after adjustment for changes in BMI.
Dr. Kahleová said the results were as expected, and she declined to speculate as to whether they would be the same if the cohort had been heavier than allowed with the BMI inclusion criteria of 27-40 kg/m2.
The investigators are currently trying to find a link between postprandial oxidative stress and gastrointestinal peptides in patients with type 2 diabetes.
The study was supported by a grant from the Ministry of Health, Prague, and by the Grant Agency of Charles University, also in Prague. Dr. Kahleová reported having no financial disclosures.
CHICAGO – Overweight patients with type 2 diabetes who ate only breakfast and lunch lost more weight than did those eating six meals a day in a randomized, crossover study.
They also had greater decreases in fasting plasma glucose, C-peptide, glucagon, and hepatic fat content, and bigger improvements in insulin sensitivity.
"Our data suggest that consuming a hearty breakfast and lunch may be more beneficial for patients with type 2 diabetes than eating more, smaller meals during the day," Dr. Hana Kahleová, said at the annual scientific sessions of the American Diabetes Association.
When asked whether eating a big lunch and dinner would produce the same benefits given the difficulty clinicians would face convincing patients to give up their evening meal, Dr. Kahleová agreed it would be a hard sell, but said the breakfast/lunch schedule is more beneficial. She highlighted research during the presentation showing fat deposition is greater following dinner (J. Clin. Endocrinol. Metab. 2009;94:1781-8), and that diabetes-free people of Mediterranean heritage who ate most of their daily intake early lost more weight and did so at a faster rate than did late eaters (Int. J. Obes. [Lond.] 2013;37:604-11).
"Our results support the ancient proverb: Eat breakfast like a king, lunch like a prince, and dinner like a pauper," said Dr. Kahleová of the Diabetes Centre, Institute for Clinical and Experimental Medicine, Prague.
Session moderator Dr. Anastassios Pittas, codirector of the Diabetes Center at Tufts Medical Center in Boston, said in an interview that few clinicians are in favor of six small meals a day because invariably patients will eat six moderate-size meals. Insulin dosing with such frequent meals is also more onerous.
"I agree with the finding that two is better than six, but that does not imply that, in general, the concept of fewer meals is better than more meals," he said. "It’s just that the comparison groups were so extreme that it’s logical to me what happened. I think the better, clinically applicable comparison would have been two vs. four or maybe five meals."
The six-meal diet was chosen as the comparator because it has been usually recommended that patients with type 2 diabetes eat five to six small meals during the day, Dr. Kahleová said in an interview. Although diabetic patients who eat small regular meals are reported to have better control of their blood glucose concentrations, the current results are strongly supported by animal studies showing the antidiabetic effects of caloric restriction and intermittent fasting diets. These effects include reduced blood glucose and insulin concentrations and improved glucose tolerance in rodents and are thought to be driven largely by increased insulin sensitivity.
Dr. Kahleová and her associates evenly randomized 54 patients with type 2 diabetes for more than 1 year to follow a six meals/day or two meals/day diet for 12 weeks. The diets were then switched for a subsequent 12 weeks. Both diets had the same caloric restriction (–500 kcal/day), carbohydrates (50%-55%), proteins (20%-25%), fats (25%-30%), cholesterol (less than 200 mg/day), and fiber (30g-40g/day).
For the two-meal diet, breakfast was eaten between hours 6-10 and lunch between hours 12-16. In the six-meal diet, breakfast was eaten during hours 6-8, snack hours 8-10, lunch hours 11-13, second snack hours 14-15, dinner hours 17-18 and second dinner hours 20-21.
To improve compliance, each regimen was started with a 4-day tutorial on how to compose and prepare their diet; group meetings with a dietician; individual counseling and meals provided for half of the patients. Patients were also asked not to alter their exercise habits during the study.
At baseline, 54% of patients were female, average diabetes duration 8.1 years, average body mass index 32.6 kg/m2, and average hemoglobin A1c 7.2%. Their average age was 59.4 years.
At 12 weeks, the mean change in BMI significantly favored the two-meal diet over the six-meal diet (–1.23 kg/m2 vs.–0.82 kg/m2; P less than .001), Dr. Kahleová said. Waist circumference shrunk significantly more as well (–5.14 cm vs. –1.37 cm; P less than .001).
Fasting plasma glucose decreased in response to both diets, but the decrease was significantly greater with two meals per day (–0.78 mmol/L vs. –0.47 mmol/L; P = .004), she said. C-peptide followed the same pattern (P = .04).
Hepatic fat content, as measured by proton magnetic resonance spectroscopy, decreased significantly more with two meals per day (–4.2% vs. –3.4%; P less than .001).
HbA1c decreased comparably in both diets, Dr. Kahleová said. Fasting plasma glucagon decreased with two daily meals (P less than .001), while it increased with six meals (P = .04).
Both parameters of beta-cell function – insulin secretion and glucose sensitivity – increased comparably with the two diets, Dr. Kahleová said. Changes in glucose sensitivity (P = .02) and oral glucose insulin sensitivity (P less than .001) correlated negatively with the change in hepatic fat content. The correlations were no longer significant, however, after adjustment for changes in BMI.
Dr. Kahleová said the results were as expected, and she declined to speculate as to whether they would be the same if the cohort had been heavier than allowed with the BMI inclusion criteria of 27-40 kg/m2.
The investigators are currently trying to find a link between postprandial oxidative stress and gastrointestinal peptides in patients with type 2 diabetes.
The study was supported by a grant from the Ministry of Health, Prague, and by the Grant Agency of Charles University, also in Prague. Dr. Kahleová reported having no financial disclosures.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major finding: The mean change in BMI at 12 weeks with two meals was –1.23 kg/m2 vs. –0.82 kg/m2 with six meals (P less than .001).
Data source: Randomized, cross-over study in 54 patients with type 2 diabetes.
Disclosures: The study was supported by a grant from the Ministry of Health, Prague, and by the Grant Agency of Charles University, also in Prague. Dr. Kahleová reported having no financial disclosures.
Ketoacidosis rates remain troubling in juvenile type 1 diabetes
CHICAGO – The prevalence of diabetic ketoacidosis has fallen by about 10% annually over time in youth with type 2 diabetes, but the life-threatening condition continues to plague roughly one in three children with type 1 diabetes, according to Dr. Dana Dabelea.
"The frequency of diabetic ketoacidosis in youth with type 1 diabetes, while stable, remains high among U.S. children at one in three children, which indicates a persistent need for increased awareness and better parental education, but also better access to health care," Dr. Dana Dabelea said at the annual scientific sessions of the American Diabetes Association.
She reported on an analysis involving 5,618 youth, aged less than 20 years, with newly diagnosed diabetes in three time periods (2002-2003, 2004-2005, and 2008-2009) and enrolled in the SEARCH for Diabetes in Youth study, a national, multicenter study designed to learn more about diabetes among American children and young adults.
Diabetic ketoacidosis (DKA) was defined by ICD-9 code and/or bicarbonate levels less than 15 mmol/L and/or a venous pH less than 7.25. Based on provider assessment, 4,537 patients had type 1 diabetes and 1,081 had type 2.
U.S. trend data on DKA prevalence in type 1 diabetes are limited, while no data are available in youth with type 2 diabetes, said Dr. Dabelea, professor of epidemiology, Colorado School for Public Health, Aurora. In Europe, decreases in DKA prevalence at onset of type 1 diabetes have been reported in Sweden and Finland, but not in Germany and Austria.
In the current analysis, DKA prevalence among youth with type 1 diabetes remained high at 29.8%, 28.8%, and 28.2% between 2002 and 2003, 2004 and 2005, and 2008 and 2009 (P = .43), she said. The number of patients diagnosed was 434, 453, and 425, respectively.
For type 2 diabetes, DKA prevalence declined from 11.3% to 6.5%, and then 6.1% over the same time periods. The difference was statistically significant (P = .02), but the number of patients is very small at 37, 21, and 26, Dr. Dabelea reported.
"In youth with type 2 diabetes, DKA at onset is less common and seems to be decreasing over time, suggesting perhaps improved diagnosis in at-risk individuals," Dr. Dabelea said.
The number of patients was too small for stratified type 2 analyses. For type 1 diabetes, DKA prevalence did not vary significantly by age at onset, race, or gender, although children aged 0-4 years in addition to blacks and Hispanics had significantly higher rates in all three time periods, she said.
In multivariate analyses, higher DKA prevalence in type 1 diabetes was independently associated with younger age at onset (age 5-9, odds ratio 0.57; age 10-14, OR 0.75; age 15-19, OR 0.38; all P less than .0001 compared with age 0-4), minority race (OR 1.33; P = .002), and lack of private insurance (OR 1.33; P = .010). Gender, education, and income were not significantly associated with a higher DKA, Dr. Dabelea reported.
Independent correlates of DKA in type 2 diabetes were younger age at onset (age 15-19 vs. age 10-14: OR 0.48; P = .002), minority race (OR 2.45; P = .013), and male gender (OR 0.54; P = .008).
"With changes in health care access on the horizon through health care reform, we might begin to see future decreases in DKA prevalence over time, but we believe that focused efforts to help the minority and underserved populations will be needed to achieve this goal," Dr. Dabelea concluded.
SEARCH is funded by the Centers for Disease Control and Prevention and the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Dabelea reported having no relevant financial disclosures.
CHICAGO – The prevalence of diabetic ketoacidosis has fallen by about 10% annually over time in youth with type 2 diabetes, but the life-threatening condition continues to plague roughly one in three children with type 1 diabetes, according to Dr. Dana Dabelea.
"The frequency of diabetic ketoacidosis in youth with type 1 diabetes, while stable, remains high among U.S. children at one in three children, which indicates a persistent need for increased awareness and better parental education, but also better access to health care," Dr. Dana Dabelea said at the annual scientific sessions of the American Diabetes Association.
She reported on an analysis involving 5,618 youth, aged less than 20 years, with newly diagnosed diabetes in three time periods (2002-2003, 2004-2005, and 2008-2009) and enrolled in the SEARCH for Diabetes in Youth study, a national, multicenter study designed to learn more about diabetes among American children and young adults.
Diabetic ketoacidosis (DKA) was defined by ICD-9 code and/or bicarbonate levels less than 15 mmol/L and/or a venous pH less than 7.25. Based on provider assessment, 4,537 patients had type 1 diabetes and 1,081 had type 2.
U.S. trend data on DKA prevalence in type 1 diabetes are limited, while no data are available in youth with type 2 diabetes, said Dr. Dabelea, professor of epidemiology, Colorado School for Public Health, Aurora. In Europe, decreases in DKA prevalence at onset of type 1 diabetes have been reported in Sweden and Finland, but not in Germany and Austria.
In the current analysis, DKA prevalence among youth with type 1 diabetes remained high at 29.8%, 28.8%, and 28.2% between 2002 and 2003, 2004 and 2005, and 2008 and 2009 (P = .43), she said. The number of patients diagnosed was 434, 453, and 425, respectively.
For type 2 diabetes, DKA prevalence declined from 11.3% to 6.5%, and then 6.1% over the same time periods. The difference was statistically significant (P = .02), but the number of patients is very small at 37, 21, and 26, Dr. Dabelea reported.
"In youth with type 2 diabetes, DKA at onset is less common and seems to be decreasing over time, suggesting perhaps improved diagnosis in at-risk individuals," Dr. Dabelea said.
The number of patients was too small for stratified type 2 analyses. For type 1 diabetes, DKA prevalence did not vary significantly by age at onset, race, or gender, although children aged 0-4 years in addition to blacks and Hispanics had significantly higher rates in all three time periods, she said.
In multivariate analyses, higher DKA prevalence in type 1 diabetes was independently associated with younger age at onset (age 5-9, odds ratio 0.57; age 10-14, OR 0.75; age 15-19, OR 0.38; all P less than .0001 compared with age 0-4), minority race (OR 1.33; P = .002), and lack of private insurance (OR 1.33; P = .010). Gender, education, and income were not significantly associated with a higher DKA, Dr. Dabelea reported.
Independent correlates of DKA in type 2 diabetes were younger age at onset (age 15-19 vs. age 10-14: OR 0.48; P = .002), minority race (OR 2.45; P = .013), and male gender (OR 0.54; P = .008).
"With changes in health care access on the horizon through health care reform, we might begin to see future decreases in DKA prevalence over time, but we believe that focused efforts to help the minority and underserved populations will be needed to achieve this goal," Dr. Dabelea concluded.
SEARCH is funded by the Centers for Disease Control and Prevention and the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Dabelea reported having no relevant financial disclosures.
CHICAGO – The prevalence of diabetic ketoacidosis has fallen by about 10% annually over time in youth with type 2 diabetes, but the life-threatening condition continues to plague roughly one in three children with type 1 diabetes, according to Dr. Dana Dabelea.
"The frequency of diabetic ketoacidosis in youth with type 1 diabetes, while stable, remains high among U.S. children at one in three children, which indicates a persistent need for increased awareness and better parental education, but also better access to health care," Dr. Dana Dabelea said at the annual scientific sessions of the American Diabetes Association.
She reported on an analysis involving 5,618 youth, aged less than 20 years, with newly diagnosed diabetes in three time periods (2002-2003, 2004-2005, and 2008-2009) and enrolled in the SEARCH for Diabetes in Youth study, a national, multicenter study designed to learn more about diabetes among American children and young adults.
Diabetic ketoacidosis (DKA) was defined by ICD-9 code and/or bicarbonate levels less than 15 mmol/L and/or a venous pH less than 7.25. Based on provider assessment, 4,537 patients had type 1 diabetes and 1,081 had type 2.
U.S. trend data on DKA prevalence in type 1 diabetes are limited, while no data are available in youth with type 2 diabetes, said Dr. Dabelea, professor of epidemiology, Colorado School for Public Health, Aurora. In Europe, decreases in DKA prevalence at onset of type 1 diabetes have been reported in Sweden and Finland, but not in Germany and Austria.
In the current analysis, DKA prevalence among youth with type 1 diabetes remained high at 29.8%, 28.8%, and 28.2% between 2002 and 2003, 2004 and 2005, and 2008 and 2009 (P = .43), she said. The number of patients diagnosed was 434, 453, and 425, respectively.
For type 2 diabetes, DKA prevalence declined from 11.3% to 6.5%, and then 6.1% over the same time periods. The difference was statistically significant (P = .02), but the number of patients is very small at 37, 21, and 26, Dr. Dabelea reported.
"In youth with type 2 diabetes, DKA at onset is less common and seems to be decreasing over time, suggesting perhaps improved diagnosis in at-risk individuals," Dr. Dabelea said.
The number of patients was too small for stratified type 2 analyses. For type 1 diabetes, DKA prevalence did not vary significantly by age at onset, race, or gender, although children aged 0-4 years in addition to blacks and Hispanics had significantly higher rates in all three time periods, she said.
In multivariate analyses, higher DKA prevalence in type 1 diabetes was independently associated with younger age at onset (age 5-9, odds ratio 0.57; age 10-14, OR 0.75; age 15-19, OR 0.38; all P less than .0001 compared with age 0-4), minority race (OR 1.33; P = .002), and lack of private insurance (OR 1.33; P = .010). Gender, education, and income were not significantly associated with a higher DKA, Dr. Dabelea reported.
Independent correlates of DKA in type 2 diabetes were younger age at onset (age 15-19 vs. age 10-14: OR 0.48; P = .002), minority race (OR 2.45; P = .013), and male gender (OR 0.54; P = .008).
"With changes in health care access on the horizon through health care reform, we might begin to see future decreases in DKA prevalence over time, but we believe that focused efforts to help the minority and underserved populations will be needed to achieve this goal," Dr. Dabelea concluded.
SEARCH is funded by the Centers for Disease Control and Prevention and the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Dabelea reported having no relevant financial disclosures.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Major finding: DKA prevalence for type 1 diabetes remained high at 29.8%, 28.8%, and 28.2% between 2002 and 2003, 2004 and 2005, and 2008 and 2009 (P = .43).
Data source: An analysis of 5,618 youth, aged less than 20 years, with newly diagnosed diabetes in the SEARCH for Diabetes in Youth study.
Disclosures: SEARCH is funded by the Centers for Disease Control and Prevention and the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Dabelea reported having no relevant financial disclosures.
Erythropoietin cuts heart surgery transfusions
MINNEAPOLIS – A single high dose of human recombinant erythropoietin given 2 days before heart surgery reduced blood transfusions by 65%, without substantially increasing mortality or morbidity in the prospective, randomized SHOT trial.
Patients who received 80,000 IU of erythropoietin (Eprex) in a single bolus plus iron supplementation until discharge needed 0.39 blood units/patient, compared with 1.12 units/patient for those receiving standard care involving thromboelastography and tranexamic acid (P less than .001; risk ratio, 0.338).
All-cause mortality at 45 days postoperative was 3.0% with erythropoietin and 3.33%
without it (P = .26), Dr. Luca Weltert said at the annual meeting of the American Association for Thoracic Surgery.
The investigators undertook the SHOT (Blood Sparing Strategies: Single Shot High Dose Erythropoietin Two Days Before Heart Surgery) trial because their original erythropoietin protocol proved too compli- cated for everyday use. The protocol was incorporated
into the 2012 Society of
Thoracic Surgeons and Society of Cardiovascular Anesthesiologists blood conservation clinical practice guidelines, but consisted of five different doses of erythropoietin in 5 days, totaling 52,000 IU, he explained.
Invited discussant Dr. Victor Ferraris, of the Lexington (Ky.) Veterans Affairs Medical Center, questioned whether the high dose of erythropoietin increased thromboembolic events, in light of the black box warnings added to erythropoiesis-stimulating agents due to an increased risk for thromboembolic events in patients with malignancy and an increased risk of serious cardiovascular events in patients with chronic kidney disease when the hemoglobin level exceeds 12 g/dL.
Thromboembolic events were not significantly different between groups, and the trial did not include cancer patients, said Dr. Weltert of the European Hospital in Rome.
Major nonfatal adverse events at 45 days were reported in 4.33% of erythropoietin and 5.67% of control patients (P = .13). Specifically, there were no differences in neurologic complications (4 vs. 5 events, respectively), long-term wound infection (4 in both groups), deep vein thrombosis (2 vs. 5), onset of acute hypertension (1 vs. 2), and new-onset renal failure (2 vs. 1).
Attendees continued this line of questioning, asking how reliable the negative finding is and whether the investigators could have missed a population with a positive result. The efficacy endpoint was based on a minimal sample size of 400 patients, and the trial enrolled 600 consecutive "all comers," Dr. Weltert said, but he added that 3,000 - 4,000 patients would be needed for the safety endpoint, "so the samples are ridiculously small to really state strongly that there is no harm for a subgroup."
The two study arms were well matched, with 35% of all patients undergoing coronary artery bypass grafting, 31% valve surgery, roughly 20% repair of the ascending aorta, and other in about 14%. The mean logistic EuroScore was 9.94 in the erythropoietin group and 10.12 in the control group, and the mean ages were 72.9 and 70.4 years, respectively.
Mean hemoglobin levels on day 4 postoperative was significantly higher in the erythropoietin group than in controls (10.21 vs. 9.01 g/dL; P = .02), Dr. Weltert said.
There were no significant differences between the erythropoietin and control groups in intensive care unit stay (2.71 vs. 2.60 days, respectively), perioperative myocardial infarction (7 vs. 7), cardiac tamponade (3 vs. 5), or need for reintubation (13 vs. 15).
An attendee asked whether hemoglobin levels drifted between days 3 and 4 postoperatively, as this can confound the decision to transfuse after cardiac surgery, and whether the investigators looked at another potential confounder, hemoglobin trigger, since no less than five randomized controlled trials have shown that a hemoglobin level of 7-8 g/dL should be used as a transfusion trigger.
Dr. Weltert said the team picked day 4 to check hemoglobin levels for practical reasons because that’s the day when you see the biggest drift and that the erythropoietin group fared better. He went on to say, "The trigger is extremely important, and I think it will be the very next step in bloodless surgery."
The investigators did compare costs for the two strategies, with standard care ringing up at 336 euros (U.S.$434), vs. 297 euros (U.S.$383) for the erythropoietin protocol (P = .05).
The European Hospital sponsored the study. Dr. Weltert reported no relevant disclosures.
MINNEAPOLIS – A single high dose of human recombinant erythropoietin given 2 days before heart surgery reduced blood transfusions by 65%, without substantially increasing mortality or morbidity in the prospective, randomized SHOT trial.
Patients who received 80,000 IU of erythropoietin (Eprex) in a single bolus plus iron supplementation until discharge needed 0.39 blood units/patient, compared with 1.12 units/patient for those receiving standard care involving thromboelastography and tranexamic acid (P less than .001; risk ratio, 0.338).
All-cause mortality at 45 days postoperative was 3.0% with erythropoietin and 3.33%
without it (P = .26), Dr. Luca Weltert said at the annual meeting of the American Association for Thoracic Surgery.
The investigators undertook the SHOT (Blood Sparing Strategies: Single Shot High Dose Erythropoietin Two Days Before Heart Surgery) trial because their original erythropoietin protocol proved too compli- cated for everyday use. The protocol was incorporated
into the 2012 Society of
Thoracic Surgeons and Society of Cardiovascular Anesthesiologists blood conservation clinical practice guidelines, but consisted of five different doses of erythropoietin in 5 days, totaling 52,000 IU, he explained.
Invited discussant Dr. Victor Ferraris, of the Lexington (Ky.) Veterans Affairs Medical Center, questioned whether the high dose of erythropoietin increased thromboembolic events, in light of the black box warnings added to erythropoiesis-stimulating agents due to an increased risk for thromboembolic events in patients with malignancy and an increased risk of serious cardiovascular events in patients with chronic kidney disease when the hemoglobin level exceeds 12 g/dL.
Thromboembolic events were not significantly different between groups, and the trial did not include cancer patients, said Dr. Weltert of the European Hospital in Rome.
Major nonfatal adverse events at 45 days were reported in 4.33% of erythropoietin and 5.67% of control patients (P = .13). Specifically, there were no differences in neurologic complications (4 vs. 5 events, respectively), long-term wound infection (4 in both groups), deep vein thrombosis (2 vs. 5), onset of acute hypertension (1 vs. 2), and new-onset renal failure (2 vs. 1).
Attendees continued this line of questioning, asking how reliable the negative finding is and whether the investigators could have missed a population with a positive result. The efficacy endpoint was based on a minimal sample size of 400 patients, and the trial enrolled 600 consecutive "all comers," Dr. Weltert said, but he added that 3,000 - 4,000 patients would be needed for the safety endpoint, "so the samples are ridiculously small to really state strongly that there is no harm for a subgroup."
The two study arms were well matched, with 35% of all patients undergoing coronary artery bypass grafting, 31% valve surgery, roughly 20% repair of the ascending aorta, and other in about 14%. The mean logistic EuroScore was 9.94 in the erythropoietin group and 10.12 in the control group, and the mean ages were 72.9 and 70.4 years, respectively.
Mean hemoglobin levels on day 4 postoperative was significantly higher in the erythropoietin group than in controls (10.21 vs. 9.01 g/dL; P = .02), Dr. Weltert said.
There were no significant differences between the erythropoietin and control groups in intensive care unit stay (2.71 vs. 2.60 days, respectively), perioperative myocardial infarction (7 vs. 7), cardiac tamponade (3 vs. 5), or need for reintubation (13 vs. 15).
An attendee asked whether hemoglobin levels drifted between days 3 and 4 postoperatively, as this can confound the decision to transfuse after cardiac surgery, and whether the investigators looked at another potential confounder, hemoglobin trigger, since no less than five randomized controlled trials have shown that a hemoglobin level of 7-8 g/dL should be used as a transfusion trigger.
Dr. Weltert said the team picked day 4 to check hemoglobin levels for practical reasons because that’s the day when you see the biggest drift and that the erythropoietin group fared better. He went on to say, "The trigger is extremely important, and I think it will be the very next step in bloodless surgery."
The investigators did compare costs for the two strategies, with standard care ringing up at 336 euros (U.S.$434), vs. 297 euros (U.S.$383) for the erythropoietin protocol (P = .05).
The European Hospital sponsored the study. Dr. Weltert reported no relevant disclosures.
MINNEAPOLIS – A single high dose of human recombinant erythropoietin given 2 days before heart surgery reduced blood transfusions by 65%, without substantially increasing mortality or morbidity in the prospective, randomized SHOT trial.
Patients who received 80,000 IU of erythropoietin (Eprex) in a single bolus plus iron supplementation until discharge needed 0.39 blood units/patient, compared with 1.12 units/patient for those receiving standard care involving thromboelastography and tranexamic acid (P less than .001; risk ratio, 0.338).
All-cause mortality at 45 days postoperative was 3.0% with erythropoietin and 3.33%
without it (P = .26), Dr. Luca Weltert said at the annual meeting of the American Association for Thoracic Surgery.
The investigators undertook the SHOT (Blood Sparing Strategies: Single Shot High Dose Erythropoietin Two Days Before Heart Surgery) trial because their original erythropoietin protocol proved too compli- cated for everyday use. The protocol was incorporated
into the 2012 Society of
Thoracic Surgeons and Society of Cardiovascular Anesthesiologists blood conservation clinical practice guidelines, but consisted of five different doses of erythropoietin in 5 days, totaling 52,000 IU, he explained.
Invited discussant Dr. Victor Ferraris, of the Lexington (Ky.) Veterans Affairs Medical Center, questioned whether the high dose of erythropoietin increased thromboembolic events, in light of the black box warnings added to erythropoiesis-stimulating agents due to an increased risk for thromboembolic events in patients with malignancy and an increased risk of serious cardiovascular events in patients with chronic kidney disease when the hemoglobin level exceeds 12 g/dL.
Thromboembolic events were not significantly different between groups, and the trial did not include cancer patients, said Dr. Weltert of the European Hospital in Rome.
Major nonfatal adverse events at 45 days were reported in 4.33% of erythropoietin and 5.67% of control patients (P = .13). Specifically, there were no differences in neurologic complications (4 vs. 5 events, respectively), long-term wound infection (4 in both groups), deep vein thrombosis (2 vs. 5), onset of acute hypertension (1 vs. 2), and new-onset renal failure (2 vs. 1).
Attendees continued this line of questioning, asking how reliable the negative finding is and whether the investigators could have missed a population with a positive result. The efficacy endpoint was based on a minimal sample size of 400 patients, and the trial enrolled 600 consecutive "all comers," Dr. Weltert said, but he added that 3,000 - 4,000 patients would be needed for the safety endpoint, "so the samples are ridiculously small to really state strongly that there is no harm for a subgroup."
The two study arms were well matched, with 35% of all patients undergoing coronary artery bypass grafting, 31% valve surgery, roughly 20% repair of the ascending aorta, and other in about 14%. The mean logistic EuroScore was 9.94 in the erythropoietin group and 10.12 in the control group, and the mean ages were 72.9 and 70.4 years, respectively.
Mean hemoglobin levels on day 4 postoperative was significantly higher in the erythropoietin group than in controls (10.21 vs. 9.01 g/dL; P = .02), Dr. Weltert said.
There were no significant differences between the erythropoietin and control groups in intensive care unit stay (2.71 vs. 2.60 days, respectively), perioperative myocardial infarction (7 vs. 7), cardiac tamponade (3 vs. 5), or need for reintubation (13 vs. 15).
An attendee asked whether hemoglobin levels drifted between days 3 and 4 postoperatively, as this can confound the decision to transfuse after cardiac surgery, and whether the investigators looked at another potential confounder, hemoglobin trigger, since no less than five randomized controlled trials have shown that a hemoglobin level of 7-8 g/dL should be used as a transfusion trigger.
Dr. Weltert said the team picked day 4 to check hemoglobin levels for practical reasons because that’s the day when you see the biggest drift and that the erythropoietin group fared better. He went on to say, "The trigger is extremely important, and I think it will be the very next step in bloodless surgery."
The investigators did compare costs for the two strategies, with standard care ringing up at 336 euros (U.S.$434), vs. 297 euros (U.S.$383) for the erythropoietin protocol (P = .05).
The European Hospital sponsored the study. Dr. Weltert reported no relevant disclosures.
AT THE AATS ANNUAL MEETING
Survival equivalent in sublobar, lobar resection of NSCLC
MINNEAPOLIS – Sublobar resection and lobectomy resulted in equivalent lung cancer survival and overall recurrence rates in a screen-detected cohort of 348 stage IA non–small cell lung cancer patients.
In all, 7% of patients (4/54) who underwent sublobar resection and 10% of those (29/294) who underwent lobectomy died of lung cancer after a median follow-up of 73 months (P = .64). All-cause mortality was also statistically similar at 17% and 22%, said Dr. Nasser Altorki, professor of cardiothoracic surgery and director of thoracic surgery at New York Presbyterian–Weill Cornell Medical Center in New York.
Although lobectomy has been the standard of care for resection of early-stage non–small cell lung cancer (NSCLC) since 1994, several studies support sublobar resection in patients with small peripheral tumors and the elderly with compromised pulmonary reserve.
The best surgical treatment for younger patients with adequate cardiopulmonary function remains controversial, with two large, ongoing trials in the U.S. and Japan evaluating lobar vs. sublobar resection in this setting. A recent best evidence paper (Interact. CardioVasc. Thorac. Surg. 2012;14:816-20) concluded that lobectomy is still the best surgical option for these patients, citing evidence of lower survival and higher recurrence rates with wedge resections than with anatomic segmentectomies. The two sublobar techniques are often lumped together in comparisons with lobectomy, but are not technically or oncologically the same.
Wedge resection in the current study seemed to be associated with a higher rate of recurrence than segmentectomy, Dr. Altorki said at the annual meeting of the American Association for Thoracic Surgery.
Recurrence occurred in 32 patients after lobectomy and 8 after sublobar resection (11% vs. 15%; P = .40), with all of the sublobar resection recurrences occurring after wedge resection (8/38 or 21% vs. 0%).
"I don’t want to say this is a practice-changing study; however, it is a study that calls for more technical equipoise in our approach to the [surgical] treatment of lung cancer," he said during a discussion of the results. "Clearly, we can apply this operation to patients who would be candidates for both lobectomy and sublobar resection, but it does not extend to those patients, for example, that would have been poor candidates for any surgical resection."
The 348 patients had c1A non–small cell lung cancer that presented as a solid nodule on computed tomography screening and underwent surgery as part of the International Early Lung Cancer Action Program from 1993 to 2011. Comorbidities were similar among the lobectomy and sublobar patients including cardiac disease (6% vs. 11%), chronic obstructive pulmonary disease (15% vs. 26%), and diabetes (9% vs. 7%). Their median age was 63 vs. 65 years, and the median number of pack-years of smoking was 48 vs. 49, respectively.
The lobectomy group had significantly larger tumors (13 mm vs. 11 mm), more frequent sampling of mediastinal nodes (78% vs. 56%), and more resected lymph nodes (mean 8 vs. 5).
The overall rate of hospital mortality in the multicenter study was low at 0.9% (3/348) and "represents what is achievable in screening centers of excellence," Dr. Altorki said.
Ten-year survival was 88% with lobectomy and 90% with sublobar resection (log rank P = .64). There was no difference in patients with tumors less than 2 cm (88% vs. 89%), who represented the majority or 86% of the cohort.
Cox regression analysis, adjusted for the above potential confounders, showed that only age (hazard ratio, 2.9; P less than .0001) and severe emphysema (HR, 4.2; P = .005) significantly predicted survival, whereas sublobar resection did not (HR, 0.8; P = .60), he said.
Invited discussant Dr. Joseph S. Friedberg, chief of thoracic surgery at the University of Pennsylvania Health System-Presbyterian in Philadelphia, expressed surprise that despite undergoing surgery by highly qualified general thoracic surgeons, 70% of sublobar resections were performed as wedge and not segmentectomies, and that more than 40% of sublobar resection patients and nearly a quarter of lobectomy patients did not have one mediastinal node biopsied.
"One would expect, based on the Lung Cancer Study Group analysis and common sense, that some of these patients, especially sublobar resection patients, were understaged and/or undertreated and yet the results are as good as anything in the literature," he said. "How do you reconcile that?"
Dr. Altorki said assessment of the mediastinal field was disappointing and that further education is needed on the prognostic and therapeutic benefits of such assessment, but that the rate far exceeds what is in the published literature. He also speculated that most of the patients were treated by video-assisted thoracic surgery and that mediastinal node assessment may not be as straightforward with VATS as it is with open surgery.
Dr. Altorki said that, going in, many of the surgeons may have thought that a 1- to 1.2-cm tumor may be equally resected with wedge resection and anatomic segmentectomy, and that this "is a job we have to address in ongoing randomized trials."
Dr. Altorki reported no relevant financial disclosures.
MINNEAPOLIS – Sublobar resection and lobectomy resulted in equivalent lung cancer survival and overall recurrence rates in a screen-detected cohort of 348 stage IA non–small cell lung cancer patients.
In all, 7% of patients (4/54) who underwent sublobar resection and 10% of those (29/294) who underwent lobectomy died of lung cancer after a median follow-up of 73 months (P = .64). All-cause mortality was also statistically similar at 17% and 22%, said Dr. Nasser Altorki, professor of cardiothoracic surgery and director of thoracic surgery at New York Presbyterian–Weill Cornell Medical Center in New York.
Although lobectomy has been the standard of care for resection of early-stage non–small cell lung cancer (NSCLC) since 1994, several studies support sublobar resection in patients with small peripheral tumors and the elderly with compromised pulmonary reserve.
The best surgical treatment for younger patients with adequate cardiopulmonary function remains controversial, with two large, ongoing trials in the U.S. and Japan evaluating lobar vs. sublobar resection in this setting. A recent best evidence paper (Interact. CardioVasc. Thorac. Surg. 2012;14:816-20) concluded that lobectomy is still the best surgical option for these patients, citing evidence of lower survival and higher recurrence rates with wedge resections than with anatomic segmentectomies. The two sublobar techniques are often lumped together in comparisons with lobectomy, but are not technically or oncologically the same.
Wedge resection in the current study seemed to be associated with a higher rate of recurrence than segmentectomy, Dr. Altorki said at the annual meeting of the American Association for Thoracic Surgery.
Recurrence occurred in 32 patients after lobectomy and 8 after sublobar resection (11% vs. 15%; P = .40), with all of the sublobar resection recurrences occurring after wedge resection (8/38 or 21% vs. 0%).
"I don’t want to say this is a practice-changing study; however, it is a study that calls for more technical equipoise in our approach to the [surgical] treatment of lung cancer," he said during a discussion of the results. "Clearly, we can apply this operation to patients who would be candidates for both lobectomy and sublobar resection, but it does not extend to those patients, for example, that would have been poor candidates for any surgical resection."
The 348 patients had c1A non–small cell lung cancer that presented as a solid nodule on computed tomography screening and underwent surgery as part of the International Early Lung Cancer Action Program from 1993 to 2011. Comorbidities were similar among the lobectomy and sublobar patients including cardiac disease (6% vs. 11%), chronic obstructive pulmonary disease (15% vs. 26%), and diabetes (9% vs. 7%). Their median age was 63 vs. 65 years, and the median number of pack-years of smoking was 48 vs. 49, respectively.
The lobectomy group had significantly larger tumors (13 mm vs. 11 mm), more frequent sampling of mediastinal nodes (78% vs. 56%), and more resected lymph nodes (mean 8 vs. 5).
The overall rate of hospital mortality in the multicenter study was low at 0.9% (3/348) and "represents what is achievable in screening centers of excellence," Dr. Altorki said.
Ten-year survival was 88% with lobectomy and 90% with sublobar resection (log rank P = .64). There was no difference in patients with tumors less than 2 cm (88% vs. 89%), who represented the majority or 86% of the cohort.
Cox regression analysis, adjusted for the above potential confounders, showed that only age (hazard ratio, 2.9; P less than .0001) and severe emphysema (HR, 4.2; P = .005) significantly predicted survival, whereas sublobar resection did not (HR, 0.8; P = .60), he said.
Invited discussant Dr. Joseph S. Friedberg, chief of thoracic surgery at the University of Pennsylvania Health System-Presbyterian in Philadelphia, expressed surprise that despite undergoing surgery by highly qualified general thoracic surgeons, 70% of sublobar resections were performed as wedge and not segmentectomies, and that more than 40% of sublobar resection patients and nearly a quarter of lobectomy patients did not have one mediastinal node biopsied.
"One would expect, based on the Lung Cancer Study Group analysis and common sense, that some of these patients, especially sublobar resection patients, were understaged and/or undertreated and yet the results are as good as anything in the literature," he said. "How do you reconcile that?"
Dr. Altorki said assessment of the mediastinal field was disappointing and that further education is needed on the prognostic and therapeutic benefits of such assessment, but that the rate far exceeds what is in the published literature. He also speculated that most of the patients were treated by video-assisted thoracic surgery and that mediastinal node assessment may not be as straightforward with VATS as it is with open surgery.
Dr. Altorki said that, going in, many of the surgeons may have thought that a 1- to 1.2-cm tumor may be equally resected with wedge resection and anatomic segmentectomy, and that this "is a job we have to address in ongoing randomized trials."
Dr. Altorki reported no relevant financial disclosures.
MINNEAPOLIS – Sublobar resection and lobectomy resulted in equivalent lung cancer survival and overall recurrence rates in a screen-detected cohort of 348 stage IA non–small cell lung cancer patients.
In all, 7% of patients (4/54) who underwent sublobar resection and 10% of those (29/294) who underwent lobectomy died of lung cancer after a median follow-up of 73 months (P = .64). All-cause mortality was also statistically similar at 17% and 22%, said Dr. Nasser Altorki, professor of cardiothoracic surgery and director of thoracic surgery at New York Presbyterian–Weill Cornell Medical Center in New York.
Although lobectomy has been the standard of care for resection of early-stage non–small cell lung cancer (NSCLC) since 1994, several studies support sublobar resection in patients with small peripheral tumors and the elderly with compromised pulmonary reserve.
The best surgical treatment for younger patients with adequate cardiopulmonary function remains controversial, with two large, ongoing trials in the U.S. and Japan evaluating lobar vs. sublobar resection in this setting. A recent best evidence paper (Interact. CardioVasc. Thorac. Surg. 2012;14:816-20) concluded that lobectomy is still the best surgical option for these patients, citing evidence of lower survival and higher recurrence rates with wedge resections than with anatomic segmentectomies. The two sublobar techniques are often lumped together in comparisons with lobectomy, but are not technically or oncologically the same.
Wedge resection in the current study seemed to be associated with a higher rate of recurrence than segmentectomy, Dr. Altorki said at the annual meeting of the American Association for Thoracic Surgery.
Recurrence occurred in 32 patients after lobectomy and 8 after sublobar resection (11% vs. 15%; P = .40), with all of the sublobar resection recurrences occurring after wedge resection (8/38 or 21% vs. 0%).
"I don’t want to say this is a practice-changing study; however, it is a study that calls for more technical equipoise in our approach to the [surgical] treatment of lung cancer," he said during a discussion of the results. "Clearly, we can apply this operation to patients who would be candidates for both lobectomy and sublobar resection, but it does not extend to those patients, for example, that would have been poor candidates for any surgical resection."
The 348 patients had c1A non–small cell lung cancer that presented as a solid nodule on computed tomography screening and underwent surgery as part of the International Early Lung Cancer Action Program from 1993 to 2011. Comorbidities were similar among the lobectomy and sublobar patients including cardiac disease (6% vs. 11%), chronic obstructive pulmonary disease (15% vs. 26%), and diabetes (9% vs. 7%). Their median age was 63 vs. 65 years, and the median number of pack-years of smoking was 48 vs. 49, respectively.
The lobectomy group had significantly larger tumors (13 mm vs. 11 mm), more frequent sampling of mediastinal nodes (78% vs. 56%), and more resected lymph nodes (mean 8 vs. 5).
The overall rate of hospital mortality in the multicenter study was low at 0.9% (3/348) and "represents what is achievable in screening centers of excellence," Dr. Altorki said.
Ten-year survival was 88% with lobectomy and 90% with sublobar resection (log rank P = .64). There was no difference in patients with tumors less than 2 cm (88% vs. 89%), who represented the majority or 86% of the cohort.
Cox regression analysis, adjusted for the above potential confounders, showed that only age (hazard ratio, 2.9; P less than .0001) and severe emphysema (HR, 4.2; P = .005) significantly predicted survival, whereas sublobar resection did not (HR, 0.8; P = .60), he said.
Invited discussant Dr. Joseph S. Friedberg, chief of thoracic surgery at the University of Pennsylvania Health System-Presbyterian in Philadelphia, expressed surprise that despite undergoing surgery by highly qualified general thoracic surgeons, 70% of sublobar resections were performed as wedge and not segmentectomies, and that more than 40% of sublobar resection patients and nearly a quarter of lobectomy patients did not have one mediastinal node biopsied.
"One would expect, based on the Lung Cancer Study Group analysis and common sense, that some of these patients, especially sublobar resection patients, were understaged and/or undertreated and yet the results are as good as anything in the literature," he said. "How do you reconcile that?"
Dr. Altorki said assessment of the mediastinal field was disappointing and that further education is needed on the prognostic and therapeutic benefits of such assessment, but that the rate far exceeds what is in the published literature. He also speculated that most of the patients were treated by video-assisted thoracic surgery and that mediastinal node assessment may not be as straightforward with VATS as it is with open surgery.
Dr. Altorki said that, going in, many of the surgeons may have thought that a 1- to 1.2-cm tumor may be equally resected with wedge resection and anatomic segmentectomy, and that this "is a job we have to address in ongoing randomized trials."
Dr. Altorki reported no relevant financial disclosures.
AT THE AATS ANNUAL MEETING
Major finding: Lung cancer–specific mortality was 7% with sublobar resection and 10% with lobar resection.
Data source: Retrospective analysis of 348 patients with stage IA non–small-cell lung cancer in the prospective International Early Lung Cancer Action Program.
Disclosures: Dr. Altorki reported no relevant financial disclosures.
Enteral nutrition linked to in-hospital mortality
CHICAGO – Hyperglycemia is common in patients receiving enteral nutrition and is a significant risk factor for hospital mortality, according to a retrospective study involving 157 patients.
Nearly 60% of patients had capillary blood glucose (CBG) values that exceeded 140 mg/dL, and nearly one-third exceeded 180 mg/dL.
More important, almost 40% of patients had a mean CBG of more than 180 mg/dL for the entire duration of their tube feedings.
At the threshold glucose of 180 mg/dL or more, the odds of dying in the hospital during tube feeding are approximately three times higher than for patients who had better glycemic control, Dr. Michael Jakoby said at the annual scientific sessions of the American Diabetes Association.
"We look at this body of work as just a stepping point to the next logical extension, which is to investigate ways that we can improve glycemic control for patients receiving any artificial nutrition, whether it’s parenteral or enteral," said Dr. Jakoby, chief of endocrinology at St. John’s Hospital in Springfield, Ill.
He previously reported that using an insulin protocol that determined insulin doses based on carbohydrate delivery and CBG was superior to ad hoc insulin dosing in the management of parenteral nutrition–induced hyperglycemia (JPEN J. Parenter. Enteral. Nutr. 2012;36:183-8).
Malnutrition among nonsurgical patients is high, and more than 40% of general surgery service patients have been reported to be malnourished (Am. J. Clin. Nutr. 1997;66:1232-9). Five studies since 2005 have established a link between parenteral nutrition and increased morbidity and in-hospital mortality, but little is available on outcomes with enteral nutrition, Dr. Jakoby explained.
The current analysis involved 157 patients receiving enteral nutrition in 2011 at St. John’s, a 350-bed hospital where each year an estimated 11,000 patients receive artificial nutrition for 2 weeks.
Of those patients, 58 were diagnosed with hyperglycemia (37%), defined as a mean CBG of 180 mg/dL or more for the duration of tube feedings.
Hyperglycemic patients were significantly older than controls (75 vs. 67), twice as likely to have a preexisting diagnosis of diabetes (81% vs. 41%), had 2 fewer days of enteral nutrition (7 vs. 9), and were less likely to be in the ICU during their tube feedings (62% vs. 83%), he said.
The two groups were well matched with regard to the amount of protein, fat, and total energy they received; however, hyperglycemic patients received significantly fewer carbohydrates than did controls (2.2 g/kg per day vs. 2.7 g/kg per day).
"With the documentation available to us, it seemed that this was an adaptive response to help combat the hyperglycemia that was happening during pure enteral nutrition," Dr. Jakoby said.
Cardiac complications – a composite of myocardial infarction, arrhythmia, and cardiac arrest – were increased in hyperglycemic patients (34% vs. 22%), but this difference was not significant in univariate analysis (odds ratio, 1.84; P = .13).
The risk of hospital mortality, however, was significantly higher in hyperglycemic patients in univariate analysis (36% vs. 16%; OR, 2.94), and remained significant in multivariate analysis after adjustment for age, sex, and preexisting diagnosis of diabetes (OR, 3.28), he noted.
During a discussion of the results, Dr. Jakoby said analyses are forthcoming looking at the impact of glycemic variability during hospitalization, observing that in a prior study he conducted, glycemic variability in the noncritical care setting strongly correlated with hospital stay and a greater likelihood of going from hospital to nursing home or the morgue.
Dr. Jakoby reported serving as a speaker for Sanofi-Aventis, Merck, and Novo Nordisk.
CHICAGO – Hyperglycemia is common in patients receiving enteral nutrition and is a significant risk factor for hospital mortality, according to a retrospective study involving 157 patients.
Nearly 60% of patients had capillary blood glucose (CBG) values that exceeded 140 mg/dL, and nearly one-third exceeded 180 mg/dL.
More important, almost 40% of patients had a mean CBG of more than 180 mg/dL for the entire duration of their tube feedings.
At the threshold glucose of 180 mg/dL or more, the odds of dying in the hospital during tube feeding are approximately three times higher than for patients who had better glycemic control, Dr. Michael Jakoby said at the annual scientific sessions of the American Diabetes Association.
"We look at this body of work as just a stepping point to the next logical extension, which is to investigate ways that we can improve glycemic control for patients receiving any artificial nutrition, whether it’s parenteral or enteral," said Dr. Jakoby, chief of endocrinology at St. John’s Hospital in Springfield, Ill.
He previously reported that using an insulin protocol that determined insulin doses based on carbohydrate delivery and CBG was superior to ad hoc insulin dosing in the management of parenteral nutrition–induced hyperglycemia (JPEN J. Parenter. Enteral. Nutr. 2012;36:183-8).
Malnutrition among nonsurgical patients is high, and more than 40% of general surgery service patients have been reported to be malnourished (Am. J. Clin. Nutr. 1997;66:1232-9). Five studies since 2005 have established a link between parenteral nutrition and increased morbidity and in-hospital mortality, but little is available on outcomes with enteral nutrition, Dr. Jakoby explained.
The current analysis involved 157 patients receiving enteral nutrition in 2011 at St. John’s, a 350-bed hospital where each year an estimated 11,000 patients receive artificial nutrition for 2 weeks.
Of those patients, 58 were diagnosed with hyperglycemia (37%), defined as a mean CBG of 180 mg/dL or more for the duration of tube feedings.
Hyperglycemic patients were significantly older than controls (75 vs. 67), twice as likely to have a preexisting diagnosis of diabetes (81% vs. 41%), had 2 fewer days of enteral nutrition (7 vs. 9), and were less likely to be in the ICU during their tube feedings (62% vs. 83%), he said.
The two groups were well matched with regard to the amount of protein, fat, and total energy they received; however, hyperglycemic patients received significantly fewer carbohydrates than did controls (2.2 g/kg per day vs. 2.7 g/kg per day).
"With the documentation available to us, it seemed that this was an adaptive response to help combat the hyperglycemia that was happening during pure enteral nutrition," Dr. Jakoby said.
Cardiac complications – a composite of myocardial infarction, arrhythmia, and cardiac arrest – were increased in hyperglycemic patients (34% vs. 22%), but this difference was not significant in univariate analysis (odds ratio, 1.84; P = .13).
The risk of hospital mortality, however, was significantly higher in hyperglycemic patients in univariate analysis (36% vs. 16%; OR, 2.94), and remained significant in multivariate analysis after adjustment for age, sex, and preexisting diagnosis of diabetes (OR, 3.28), he noted.
During a discussion of the results, Dr. Jakoby said analyses are forthcoming looking at the impact of glycemic variability during hospitalization, observing that in a prior study he conducted, glycemic variability in the noncritical care setting strongly correlated with hospital stay and a greater likelihood of going from hospital to nursing home or the morgue.
Dr. Jakoby reported serving as a speaker for Sanofi-Aventis, Merck, and Novo Nordisk.
CHICAGO – Hyperglycemia is common in patients receiving enteral nutrition and is a significant risk factor for hospital mortality, according to a retrospective study involving 157 patients.
Nearly 60% of patients had capillary blood glucose (CBG) values that exceeded 140 mg/dL, and nearly one-third exceeded 180 mg/dL.
More important, almost 40% of patients had a mean CBG of more than 180 mg/dL for the entire duration of their tube feedings.
At the threshold glucose of 180 mg/dL or more, the odds of dying in the hospital during tube feeding are approximately three times higher than for patients who had better glycemic control, Dr. Michael Jakoby said at the annual scientific sessions of the American Diabetes Association.
"We look at this body of work as just a stepping point to the next logical extension, which is to investigate ways that we can improve glycemic control for patients receiving any artificial nutrition, whether it’s parenteral or enteral," said Dr. Jakoby, chief of endocrinology at St. John’s Hospital in Springfield, Ill.
He previously reported that using an insulin protocol that determined insulin doses based on carbohydrate delivery and CBG was superior to ad hoc insulin dosing in the management of parenteral nutrition–induced hyperglycemia (JPEN J. Parenter. Enteral. Nutr. 2012;36:183-8).
Malnutrition among nonsurgical patients is high, and more than 40% of general surgery service patients have been reported to be malnourished (Am. J. Clin. Nutr. 1997;66:1232-9). Five studies since 2005 have established a link between parenteral nutrition and increased morbidity and in-hospital mortality, but little is available on outcomes with enteral nutrition, Dr. Jakoby explained.
The current analysis involved 157 patients receiving enteral nutrition in 2011 at St. John’s, a 350-bed hospital where each year an estimated 11,000 patients receive artificial nutrition for 2 weeks.
Of those patients, 58 were diagnosed with hyperglycemia (37%), defined as a mean CBG of 180 mg/dL or more for the duration of tube feedings.
Hyperglycemic patients were significantly older than controls (75 vs. 67), twice as likely to have a preexisting diagnosis of diabetes (81% vs. 41%), had 2 fewer days of enteral nutrition (7 vs. 9), and were less likely to be in the ICU during their tube feedings (62% vs. 83%), he said.
The two groups were well matched with regard to the amount of protein, fat, and total energy they received; however, hyperglycemic patients received significantly fewer carbohydrates than did controls (2.2 g/kg per day vs. 2.7 g/kg per day).
"With the documentation available to us, it seemed that this was an adaptive response to help combat the hyperglycemia that was happening during pure enteral nutrition," Dr. Jakoby said.
Cardiac complications – a composite of myocardial infarction, arrhythmia, and cardiac arrest – were increased in hyperglycemic patients (34% vs. 22%), but this difference was not significant in univariate analysis (odds ratio, 1.84; P = .13).
The risk of hospital mortality, however, was significantly higher in hyperglycemic patients in univariate analysis (36% vs. 16%; OR, 2.94), and remained significant in multivariate analysis after adjustment for age, sex, and preexisting diagnosis of diabetes (OR, 3.28), he noted.
During a discussion of the results, Dr. Jakoby said analyses are forthcoming looking at the impact of glycemic variability during hospitalization, observing that in a prior study he conducted, glycemic variability in the noncritical care setting strongly correlated with hospital stay and a greater likelihood of going from hospital to nursing home or the morgue.
Dr. Jakoby reported serving as a speaker for Sanofi-Aventis, Merck, and Novo Nordisk.
AT THE ANNUAL ADA SCIENTIFIC SESSIONS
Major finding: The mortality rate was 36% in hyperglycemic patients vs. 16% in controls (unadjusted OR 2.94; adjusted 3.28), a significant difference.
Data source: Retrospective analysis of 157 hospitalized patients receiving enteral nutrition.
Disclosures: Dr. Jakoby reported serving as a speaker for Sanofi-Aventis, Merck, and Novo Nordisk.