User login
Practice-based asthma navigators trim asthma symptoms, HC utilization
MADRID – Use of practice-based asthma navigators significantly reduced symptoms and emergency room and inpatient visits among inner-city children with moderate to severe asthma in a prospective, case-matched study.
"We’ve seen with our home-visit studies that we’re able to reduce asthma morbidity, but what we hear from the parents all the time is that they feel their communication with providers is lacking, and that they really needed someone to help them navigate the clinical system," Dr. Tyra Bryant-Stephens, founder and director of the Community Asthma Prevention Program at Children’s Hospital of Philadelphia (CHOP), said in an interview.
To overcome this hurdle, CHOP began integrating community health workers as asthma navigators into the clinical team at its inner-city asthma clinics. Key tasks are to teach caregivers about the Asthma Care Plan and proper use of controller medications, facilitate appointment scheduling and set up reminders, contact and share asthma care plans with school nurses, work with social workers to identify appropriate resources for families, and set care coordination goals.
The study enrolled children, aged 2-17 years, on at least two asthma control medications. The participants had been hospitalized or had at least two emergency department (ED) visits in the past year. Their average age was 4.6 years.
After 12 months in the navigator program, preliminary data on 99 children revealed a clear 2- to 3-day reduction over the past 14 days in days using rescue asthma medications (5.28 vs. 3.07; P less than .01), days with symptoms (6.93 to 3.8; P less than .01), and nights with symptoms (6.11 to 3.34; P less than .01), Dr. Bryant-Stephens reported at the world congress of the American College of Chest Physicians.
Compared with baseline, the number of days not taking asthma medications at 12 months was not significantly different (1.48 vs. 1.22) nor was the number of days during which activities had to be slowed (4.41 vs. 3.73).
Enrollment in the navigator program, however, lowered the number of school days missed (16 vs. 2; P less than .05), workdays missed (9.11 vs. 1.13; P less than .01), unscheduled visits to the doctor (2.5 vs. 0.5; P less than .05), ED visits (3.65 vs. 1.31; P less than .01), and hospitalizations (1.73 vs. 0.4; P less than .01).
Although the final analysis comparing participants with matched controls receiving usual care is not yet complete, the data so far show a definite reduction in health care utilization by participants, despite controls being less sick at baseline, Dr. Bryant-Stephens said. This is particularly encouraging because earlier studies at CHOP showed that roughly 50% of asthmatic children never made it back to their primary care physician for follow-up between ED visits.
Making appointments for and keeping follow-up visits were listed by 146 of 157 (93%) caregivers as one of the most important care coordination goals, and were achieved by 80% at 12 months with the assistance of phone calls from the asthma navigator, transportation tokens, and insurance cotransportation, according to the poster presentation.
Success among the other top five caregiver goals was 92% for learning how to properly use asthma medications (126/136 caregivers), 98% for reducing asthma triggers (154/157), 54% for stopping smoking in the house and car (23/42), and 94% for the surprising goal of losing weight in hopes it would reduce asthma symptoms (34/36).
There is no "magic bullet," or single component of the navigator program responsible for the results, but "I think what the navigators have been most effective at is bringing them back to the office. It’s unbelievable compared with the control group," Dr. Bryant-Stephens said at the meeting.
Ultimately, the goal is to make the program self-sufficient, with a pilot program currently underway in which Pennsylvania’s largest Medicaid provider, Keystone First, will pay CHOP to have their clients assigned an asthma navigator when they enroll at an asthma clinic.
Dr. Bryant-Stephens reported funding from the Merck Childhood Asthma Network.
MADRID – Use of practice-based asthma navigators significantly reduced symptoms and emergency room and inpatient visits among inner-city children with moderate to severe asthma in a prospective, case-matched study.
"We’ve seen with our home-visit studies that we’re able to reduce asthma morbidity, but what we hear from the parents all the time is that they feel their communication with providers is lacking, and that they really needed someone to help them navigate the clinical system," Dr. Tyra Bryant-Stephens, founder and director of the Community Asthma Prevention Program at Children’s Hospital of Philadelphia (CHOP), said in an interview.
To overcome this hurdle, CHOP began integrating community health workers as asthma navigators into the clinical team at its inner-city asthma clinics. Key tasks are to teach caregivers about the Asthma Care Plan and proper use of controller medications, facilitate appointment scheduling and set up reminders, contact and share asthma care plans with school nurses, work with social workers to identify appropriate resources for families, and set care coordination goals.
The study enrolled children, aged 2-17 years, on at least two asthma control medications. The participants had been hospitalized or had at least two emergency department (ED) visits in the past year. Their average age was 4.6 years.
After 12 months in the navigator program, preliminary data on 99 children revealed a clear 2- to 3-day reduction over the past 14 days in days using rescue asthma medications (5.28 vs. 3.07; P less than .01), days with symptoms (6.93 to 3.8; P less than .01), and nights with symptoms (6.11 to 3.34; P less than .01), Dr. Bryant-Stephens reported at the world congress of the American College of Chest Physicians.
Compared with baseline, the number of days not taking asthma medications at 12 months was not significantly different (1.48 vs. 1.22) nor was the number of days during which activities had to be slowed (4.41 vs. 3.73).
Enrollment in the navigator program, however, lowered the number of school days missed (16 vs. 2; P less than .05), workdays missed (9.11 vs. 1.13; P less than .01), unscheduled visits to the doctor (2.5 vs. 0.5; P less than .05), ED visits (3.65 vs. 1.31; P less than .01), and hospitalizations (1.73 vs. 0.4; P less than .01).
Although the final analysis comparing participants with matched controls receiving usual care is not yet complete, the data so far show a definite reduction in health care utilization by participants, despite controls being less sick at baseline, Dr. Bryant-Stephens said. This is particularly encouraging because earlier studies at CHOP showed that roughly 50% of asthmatic children never made it back to their primary care physician for follow-up between ED visits.
Making appointments for and keeping follow-up visits were listed by 146 of 157 (93%) caregivers as one of the most important care coordination goals, and were achieved by 80% at 12 months with the assistance of phone calls from the asthma navigator, transportation tokens, and insurance cotransportation, according to the poster presentation.
Success among the other top five caregiver goals was 92% for learning how to properly use asthma medications (126/136 caregivers), 98% for reducing asthma triggers (154/157), 54% for stopping smoking in the house and car (23/42), and 94% for the surprising goal of losing weight in hopes it would reduce asthma symptoms (34/36).
There is no "magic bullet," or single component of the navigator program responsible for the results, but "I think what the navigators have been most effective at is bringing them back to the office. It’s unbelievable compared with the control group," Dr. Bryant-Stephens said at the meeting.
Ultimately, the goal is to make the program self-sufficient, with a pilot program currently underway in which Pennsylvania’s largest Medicaid provider, Keystone First, will pay CHOP to have their clients assigned an asthma navigator when they enroll at an asthma clinic.
Dr. Bryant-Stephens reported funding from the Merck Childhood Asthma Network.
MADRID – Use of practice-based asthma navigators significantly reduced symptoms and emergency room and inpatient visits among inner-city children with moderate to severe asthma in a prospective, case-matched study.
"We’ve seen with our home-visit studies that we’re able to reduce asthma morbidity, but what we hear from the parents all the time is that they feel their communication with providers is lacking, and that they really needed someone to help them navigate the clinical system," Dr. Tyra Bryant-Stephens, founder and director of the Community Asthma Prevention Program at Children’s Hospital of Philadelphia (CHOP), said in an interview.
To overcome this hurdle, CHOP began integrating community health workers as asthma navigators into the clinical team at its inner-city asthma clinics. Key tasks are to teach caregivers about the Asthma Care Plan and proper use of controller medications, facilitate appointment scheduling and set up reminders, contact and share asthma care plans with school nurses, work with social workers to identify appropriate resources for families, and set care coordination goals.
The study enrolled children, aged 2-17 years, on at least two asthma control medications. The participants had been hospitalized or had at least two emergency department (ED) visits in the past year. Their average age was 4.6 years.
After 12 months in the navigator program, preliminary data on 99 children revealed a clear 2- to 3-day reduction over the past 14 days in days using rescue asthma medications (5.28 vs. 3.07; P less than .01), days with symptoms (6.93 to 3.8; P less than .01), and nights with symptoms (6.11 to 3.34; P less than .01), Dr. Bryant-Stephens reported at the world congress of the American College of Chest Physicians.
Compared with baseline, the number of days not taking asthma medications at 12 months was not significantly different (1.48 vs. 1.22) nor was the number of days during which activities had to be slowed (4.41 vs. 3.73).
Enrollment in the navigator program, however, lowered the number of school days missed (16 vs. 2; P less than .05), workdays missed (9.11 vs. 1.13; P less than .01), unscheduled visits to the doctor (2.5 vs. 0.5; P less than .05), ED visits (3.65 vs. 1.31; P less than .01), and hospitalizations (1.73 vs. 0.4; P less than .01).
Although the final analysis comparing participants with matched controls receiving usual care is not yet complete, the data so far show a definite reduction in health care utilization by participants, despite controls being less sick at baseline, Dr. Bryant-Stephens said. This is particularly encouraging because earlier studies at CHOP showed that roughly 50% of asthmatic children never made it back to their primary care physician for follow-up between ED visits.
Making appointments for and keeping follow-up visits were listed by 146 of 157 (93%) caregivers as one of the most important care coordination goals, and were achieved by 80% at 12 months with the assistance of phone calls from the asthma navigator, transportation tokens, and insurance cotransportation, according to the poster presentation.
Success among the other top five caregiver goals was 92% for learning how to properly use asthma medications (126/136 caregivers), 98% for reducing asthma triggers (154/157), 54% for stopping smoking in the house and car (23/42), and 94% for the surprising goal of losing weight in hopes it would reduce asthma symptoms (34/36).
There is no "magic bullet," or single component of the navigator program responsible for the results, but "I think what the navigators have been most effective at is bringing them back to the office. It’s unbelievable compared with the control group," Dr. Bryant-Stephens said at the meeting.
Ultimately, the goal is to make the program self-sufficient, with a pilot program currently underway in which Pennsylvania’s largest Medicaid provider, Keystone First, will pay CHOP to have their clients assigned an asthma navigator when they enroll at an asthma clinic.
Dr. Bryant-Stephens reported funding from the Merck Childhood Asthma Network.
AT CHEST WORLD CONGRESS 2014
Major finding: After 12 months in the asthma navigator program, children had fewer unscheduled visits to the doctor (2.5 vs. 0.5; P less than .05), emergency department visits (3.65 vs. 1.31; P less than .01), and hospitalizations (1.73 vs. 0.4; P less than .01).
Data source: A prospective case-matched study in 99 children with moderate to severe asthma.
Disclosures: Dr. Bryant-Stephens reported funding from the Merck Childhood Asthma Network.
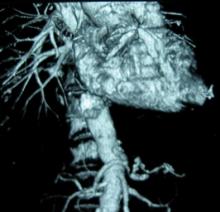
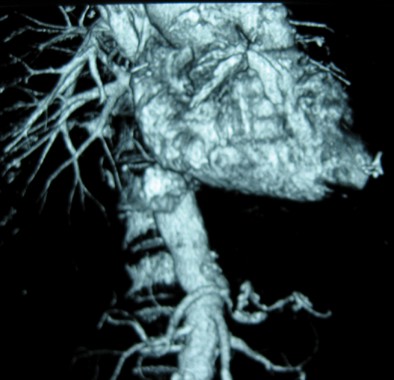
Failure to rescue drives AAA repair mortality
CHICAGO -- Failure to rescue patients from major complications drives much of the variation in hospital mortality for abdominal aortic aneurysm repair, an award-winning study suggests.
"Careful attention to early recognition and management of postoperative complications could be the key to improving mortality," Dr. Seth Waits reported at the annual meeting of the Midwestern Vascular Surgical Society.
Failure to rescue (FTR), or death after a complication, is increasing being recognized as a source of differences in hospital mortality. A recent study reported that women who experienced a major complication after ovarian cancer treatment at a low-volume hospital were 48% more likely to die than were their counterparts at a high-volume hospital. Complication rates, long thought to be the culprit behind higher hospital mortality, were similar at the hospitals, while FTR rates were almost double at the low-volume hospitals (J. Clin. Oncol. 2012;30:3976-82).
For the current analysis, Dr. Waits and his colleagues at the University of Michigan Frankel Cardiovascular Center in Ann Arbor, calculated risk-adjusted mortality rates for 3,215 patients who underwent open or endovascular abdominal aortic aneurysm (AAA) repair at 40 hospitals participating in the Michigan Surgical Quality Collaborative between 2007 and 2012.
For 2,440 patients undergoing endovascular repair, hospital mortality ranged from a low of 0.07% to a high of 6.14%.
Though low- and high-mortality hospitals had similar major complication rates (11.6% vs. 10.6%), FTR rates were 45 times greater in high-mortality hospitals (0.83% vs. 37.5%), Dr. Waits reported.
For 775 patients who underwent open AAA repair, hospital mortality ranged from 4.5% to 16.4%.
Once again, despite low- and high-mortality hospitals having nearly identical complication rates (45.1% vs. 45.8%), but FTR rates were three times higher at the high-mortality hospitals (10.3% vs. 33%), according to Dr. Waits.
An average of 2.85 and 2.66 severe complications occurred per FTR event for open and endovascular repair, respectively.
Transfusion was the most common postoperative complication leading to a FTR event for endovascular and open repair (5.8% and 29.8%, respectively), followed by prolonged intubation (2.4%; 18.2%) and reintubation (9.2%; 2%), according to the meeting’s best poster presentation.
No significant difference was seen in rupture/emergent repair between low- and high-mortality hospitals.
Dr. Waits called for preoperative identification of high-risk patients and use of FTR countermeasures, such as improved ICU admission, anesthesia alerts, and nurse/physician awareness to improve AAA mortality.
"Understanding the mechanisms that underlie failure to rescue offers the opportunity to move from a reactive to proactive approach in our management of complications following abdominal aortic aneurysm repair," he said in an interview.
Dr. Waits and his coauthors reported that they had no financial disclosures.
CHICAGO -- Failure to rescue patients from major complications drives much of the variation in hospital mortality for abdominal aortic aneurysm repair, an award-winning study suggests.
"Careful attention to early recognition and management of postoperative complications could be the key to improving mortality," Dr. Seth Waits reported at the annual meeting of the Midwestern Vascular Surgical Society.
Failure to rescue (FTR), or death after a complication, is increasing being recognized as a source of differences in hospital mortality. A recent study reported that women who experienced a major complication after ovarian cancer treatment at a low-volume hospital were 48% more likely to die than were their counterparts at a high-volume hospital. Complication rates, long thought to be the culprit behind higher hospital mortality, were similar at the hospitals, while FTR rates were almost double at the low-volume hospitals (J. Clin. Oncol. 2012;30:3976-82).
For the current analysis, Dr. Waits and his colleagues at the University of Michigan Frankel Cardiovascular Center in Ann Arbor, calculated risk-adjusted mortality rates for 3,215 patients who underwent open or endovascular abdominal aortic aneurysm (AAA) repair at 40 hospitals participating in the Michigan Surgical Quality Collaborative between 2007 and 2012.
For 2,440 patients undergoing endovascular repair, hospital mortality ranged from a low of 0.07% to a high of 6.14%.
Though low- and high-mortality hospitals had similar major complication rates (11.6% vs. 10.6%), FTR rates were 45 times greater in high-mortality hospitals (0.83% vs. 37.5%), Dr. Waits reported.
For 775 patients who underwent open AAA repair, hospital mortality ranged from 4.5% to 16.4%.
Once again, despite low- and high-mortality hospitals having nearly identical complication rates (45.1% vs. 45.8%), but FTR rates were three times higher at the high-mortality hospitals (10.3% vs. 33%), according to Dr. Waits.
An average of 2.85 and 2.66 severe complications occurred per FTR event for open and endovascular repair, respectively.
Transfusion was the most common postoperative complication leading to a FTR event for endovascular and open repair (5.8% and 29.8%, respectively), followed by prolonged intubation (2.4%; 18.2%) and reintubation (9.2%; 2%), according to the meeting’s best poster presentation.
No significant difference was seen in rupture/emergent repair between low- and high-mortality hospitals.
Dr. Waits called for preoperative identification of high-risk patients and use of FTR countermeasures, such as improved ICU admission, anesthesia alerts, and nurse/physician awareness to improve AAA mortality.
"Understanding the mechanisms that underlie failure to rescue offers the opportunity to move from a reactive to proactive approach in our management of complications following abdominal aortic aneurysm repair," he said in an interview.
Dr. Waits and his coauthors reported that they had no financial disclosures.
CHICAGO -- Failure to rescue patients from major complications drives much of the variation in hospital mortality for abdominal aortic aneurysm repair, an award-winning study suggests.
"Careful attention to early recognition and management of postoperative complications could be the key to improving mortality," Dr. Seth Waits reported at the annual meeting of the Midwestern Vascular Surgical Society.
Failure to rescue (FTR), or death after a complication, is increasing being recognized as a source of differences in hospital mortality. A recent study reported that women who experienced a major complication after ovarian cancer treatment at a low-volume hospital were 48% more likely to die than were their counterparts at a high-volume hospital. Complication rates, long thought to be the culprit behind higher hospital mortality, were similar at the hospitals, while FTR rates were almost double at the low-volume hospitals (J. Clin. Oncol. 2012;30:3976-82).
For the current analysis, Dr. Waits and his colleagues at the University of Michigan Frankel Cardiovascular Center in Ann Arbor, calculated risk-adjusted mortality rates for 3,215 patients who underwent open or endovascular abdominal aortic aneurysm (AAA) repair at 40 hospitals participating in the Michigan Surgical Quality Collaborative between 2007 and 2012.
For 2,440 patients undergoing endovascular repair, hospital mortality ranged from a low of 0.07% to a high of 6.14%.
Though low- and high-mortality hospitals had similar major complication rates (11.6% vs. 10.6%), FTR rates were 45 times greater in high-mortality hospitals (0.83% vs. 37.5%), Dr. Waits reported.
For 775 patients who underwent open AAA repair, hospital mortality ranged from 4.5% to 16.4%.
Once again, despite low- and high-mortality hospitals having nearly identical complication rates (45.1% vs. 45.8%), but FTR rates were three times higher at the high-mortality hospitals (10.3% vs. 33%), according to Dr. Waits.
An average of 2.85 and 2.66 severe complications occurred per FTR event for open and endovascular repair, respectively.
Transfusion was the most common postoperative complication leading to a FTR event for endovascular and open repair (5.8% and 29.8%, respectively), followed by prolonged intubation (2.4%; 18.2%) and reintubation (9.2%; 2%), according to the meeting’s best poster presentation.
No significant difference was seen in rupture/emergent repair between low- and high-mortality hospitals.
Dr. Waits called for preoperative identification of high-risk patients and use of FTR countermeasures, such as improved ICU admission, anesthesia alerts, and nurse/physician awareness to improve AAA mortality.
"Understanding the mechanisms that underlie failure to rescue offers the opportunity to move from a reactive to proactive approach in our management of complications following abdominal aortic aneurysm repair," he said in an interview.
Dr. Waits and his coauthors reported that they had no financial disclosures.
AT MIDWESTERN VASCULAR 2013
Major finding: FTR rates were 45 times higher in high AAA-mortality hospitals vs. low AAA-mortality hospitals (0.83% vs. 37.5%).
Data source: Retrospective study of 3,215 patients with abdominal aortic aneurysm repair.
Disclosures: Dr. Waits and his coauthors reported having no financial disclosures.
Anatomy dictates endo PAA repair
CHICAGO - Endovascular repair of popliteal artery aneurysms is a relatively safe and viable off-label alternative to open repair, but appropriate anatomy is essential. This includes a landing zone of at least 2 cm above and below the aneurysm, minimal discrepancy in size between the proximal and distal landing zones, and lack of extensive vessel tortuosity due to potential kinking of the endograft, Dr. Neal S. Cayne said at a symposium on vascular surgery sponsored by Northwestern University.
The risk of kinking and graft thrombosis also excludes patients who frequently flex their knee more than 90 degrees, such as carpenters and gardeners.
Patients with a contraindication to antiplatelet medication are also off limits, as clopidogrel (Plavix) has been shown to be a predictor of success, said Dr. Cayne, director of endovascular surgery at New York University Langone Medical Center.
In 2012, his team reported technical success in 25 of 26 endovascular popliteal artery aneurysm (PAA) repairs performed in 21 consecutive patients between January 2004 and January 2011, with the one technical failure due to stent graft infolding (J. Vasc. Surg. 2012;55:1647-53).
Primary and secondary patency rates were both 91.2% at 1 year, and were 85.5% and 91.2%, respectively, at 2 years. All patients were maintained on aspirin or clopidogrel.
No limb loss was reported, but three occlusions occurred during follow-up at 4, 14, and 26 months. One patient required a tibial artery bypass for a nonhealing wound, and two were successfully repaired with open thrombectomy. All three occlusion patients had single-vessel runoff.
Based on our data, in general, I will not stent someone "with single-vessel outflow, and I also will not stent someone who, for some reason, can't take antiplatelet agents," Dr. Cayne said.
Even when patients present urgently and the PAA has thrombosed, endovascular repair is not an option if there is only one outflow vessel. "I would do a bypass; there's nothing wrong with open surgery," he said.
A recent unpublished review of 79 PAAs treated at Langone from 1998 to 2012 with both approaches found 5-year primary patencies of 67% for open repair and 80% for endovascular repair (P less than .05). Secondary patency was 90% in both groups.
One amputation occurred in the open group, but occlusion rates were higher using endovascular repair with one-vessel runoff (P = .003), Dr. Cayne said.
As expected, length of stay was shorter with endovascular repair (1.9 vs. 6.4 days; P less than .001).
Follow-up was longer for the 36 open PAAs than the 43 endovascular PAAs (75 vs. 34 months), but patients in both groups were similar with respect to age, comorbidities, PAA size, runoff, and symptoms, he said.
During a discussion following the presentation, some attendees said they still prefer to use bypass for all patients with PAA, and asked how candidates are selected for stent placement.
"Number one and most important is the anatomy," Dr. Cayne said. "The one advantage of endovascular repair is that if you have a patient too sick to get general or even regional anesthesia, you can do it under local [anesthesia] almost all the time with a small cutdown or puncture. But you do have that long discussion with the patient that this is a non-FDA-approved, off-label use. We provide them with the data, but some patients in New York will come in with pages and pages of literature and say, 'Nope, I want a stent, I want this particular stent, and this is the way I want you to do it.'"
The stent of choice at Langone has been Gore's Viabahn covered stent graft, which is FDA approved for treating occlusive disease rather than PAA. The device is usually oversized by 10%-15%, but no more than that, because of the risk of graft infolding, Dr. Cayne said.
If more than one graft is needed, a maximum of no more than 1-mm size differential between grafts is suggested. A minimum overlap of 2-3 cm between grafts is also preferred.
Last year, surgeons reported a dismal 50% occlusion rate within just 6 weeks in the endovascular treatment of six PAAs using a novel, multilayer stent (Cardiatis's Multilayer Aneurysm Repair System) (J. Endovasc. Ther. 2013;20:381-8), he observed.
Dr. Cayne reported receiving honoraria as a consultant from Cook Medical.
CHICAGO - Endovascular repair of popliteal artery aneurysms is a relatively safe and viable off-label alternative to open repair, but appropriate anatomy is essential. This includes a landing zone of at least 2 cm above and below the aneurysm, minimal discrepancy in size between the proximal and distal landing zones, and lack of extensive vessel tortuosity due to potential kinking of the endograft, Dr. Neal S. Cayne said at a symposium on vascular surgery sponsored by Northwestern University.
The risk of kinking and graft thrombosis also excludes patients who frequently flex their knee more than 90 degrees, such as carpenters and gardeners.
Patients with a contraindication to antiplatelet medication are also off limits, as clopidogrel (Plavix) has been shown to be a predictor of success, said Dr. Cayne, director of endovascular surgery at New York University Langone Medical Center.
In 2012, his team reported technical success in 25 of 26 endovascular popliteal artery aneurysm (PAA) repairs performed in 21 consecutive patients between January 2004 and January 2011, with the one technical failure due to stent graft infolding (J. Vasc. Surg. 2012;55:1647-53).
Primary and secondary patency rates were both 91.2% at 1 year, and were 85.5% and 91.2%, respectively, at 2 years. All patients were maintained on aspirin or clopidogrel.
No limb loss was reported, but three occlusions occurred during follow-up at 4, 14, and 26 months. One patient required a tibial artery bypass for a nonhealing wound, and two were successfully repaired with open thrombectomy. All three occlusion patients had single-vessel runoff.
Based on our data, in general, I will not stent someone "with single-vessel outflow, and I also will not stent someone who, for some reason, can't take antiplatelet agents," Dr. Cayne said.
Even when patients present urgently and the PAA has thrombosed, endovascular repair is not an option if there is only one outflow vessel. "I would do a bypass; there's nothing wrong with open surgery," he said.
A recent unpublished review of 79 PAAs treated at Langone from 1998 to 2012 with both approaches found 5-year primary patencies of 67% for open repair and 80% for endovascular repair (P less than .05). Secondary patency was 90% in both groups.
One amputation occurred in the open group, but occlusion rates were higher using endovascular repair with one-vessel runoff (P = .003), Dr. Cayne said.
As expected, length of stay was shorter with endovascular repair (1.9 vs. 6.4 days; P less than .001).
Follow-up was longer for the 36 open PAAs than the 43 endovascular PAAs (75 vs. 34 months), but patients in both groups were similar with respect to age, comorbidities, PAA size, runoff, and symptoms, he said.
During a discussion following the presentation, some attendees said they still prefer to use bypass for all patients with PAA, and asked how candidates are selected for stent placement.
"Number one and most important is the anatomy," Dr. Cayne said. "The one advantage of endovascular repair is that if you have a patient too sick to get general or even regional anesthesia, you can do it under local [anesthesia] almost all the time with a small cutdown or puncture. But you do have that long discussion with the patient that this is a non-FDA-approved, off-label use. We provide them with the data, but some patients in New York will come in with pages and pages of literature and say, 'Nope, I want a stent, I want this particular stent, and this is the way I want you to do it.'"
The stent of choice at Langone has been Gore's Viabahn covered stent graft, which is FDA approved for treating occlusive disease rather than PAA. The device is usually oversized by 10%-15%, but no more than that, because of the risk of graft infolding, Dr. Cayne said.
If more than one graft is needed, a maximum of no more than 1-mm size differential between grafts is suggested. A minimum overlap of 2-3 cm between grafts is also preferred.
Last year, surgeons reported a dismal 50% occlusion rate within just 6 weeks in the endovascular treatment of six PAAs using a novel, multilayer stent (Cardiatis's Multilayer Aneurysm Repair System) (J. Endovasc. Ther. 2013;20:381-8), he observed.
Dr. Cayne reported receiving honoraria as a consultant from Cook Medical.
CHICAGO - Endovascular repair of popliteal artery aneurysms is a relatively safe and viable off-label alternative to open repair, but appropriate anatomy is essential. This includes a landing zone of at least 2 cm above and below the aneurysm, minimal discrepancy in size between the proximal and distal landing zones, and lack of extensive vessel tortuosity due to potential kinking of the endograft, Dr. Neal S. Cayne said at a symposium on vascular surgery sponsored by Northwestern University.
The risk of kinking and graft thrombosis also excludes patients who frequently flex their knee more than 90 degrees, such as carpenters and gardeners.
Patients with a contraindication to antiplatelet medication are also off limits, as clopidogrel (Plavix) has been shown to be a predictor of success, said Dr. Cayne, director of endovascular surgery at New York University Langone Medical Center.
In 2012, his team reported technical success in 25 of 26 endovascular popliteal artery aneurysm (PAA) repairs performed in 21 consecutive patients between January 2004 and January 2011, with the one technical failure due to stent graft infolding (J. Vasc. Surg. 2012;55:1647-53).
Primary and secondary patency rates were both 91.2% at 1 year, and were 85.5% and 91.2%, respectively, at 2 years. All patients were maintained on aspirin or clopidogrel.
No limb loss was reported, but three occlusions occurred during follow-up at 4, 14, and 26 months. One patient required a tibial artery bypass for a nonhealing wound, and two were successfully repaired with open thrombectomy. All three occlusion patients had single-vessel runoff.
Based on our data, in general, I will not stent someone "with single-vessel outflow, and I also will not stent someone who, for some reason, can't take antiplatelet agents," Dr. Cayne said.
Even when patients present urgently and the PAA has thrombosed, endovascular repair is not an option if there is only one outflow vessel. "I would do a bypass; there's nothing wrong with open surgery," he said.
A recent unpublished review of 79 PAAs treated at Langone from 1998 to 2012 with both approaches found 5-year primary patencies of 67% for open repair and 80% for endovascular repair (P less than .05). Secondary patency was 90% in both groups.
One amputation occurred in the open group, but occlusion rates were higher using endovascular repair with one-vessel runoff (P = .003), Dr. Cayne said.
As expected, length of stay was shorter with endovascular repair (1.9 vs. 6.4 days; P less than .001).
Follow-up was longer for the 36 open PAAs than the 43 endovascular PAAs (75 vs. 34 months), but patients in both groups were similar with respect to age, comorbidities, PAA size, runoff, and symptoms, he said.
During a discussion following the presentation, some attendees said they still prefer to use bypass for all patients with PAA, and asked how candidates are selected for stent placement.
"Number one and most important is the anatomy," Dr. Cayne said. "The one advantage of endovascular repair is that if you have a patient too sick to get general or even regional anesthesia, you can do it under local [anesthesia] almost all the time with a small cutdown or puncture. But you do have that long discussion with the patient that this is a non-FDA-approved, off-label use. We provide them with the data, but some patients in New York will come in with pages and pages of literature and say, 'Nope, I want a stent, I want this particular stent, and this is the way I want you to do it.'"
The stent of choice at Langone has been Gore's Viabahn covered stent graft, which is FDA approved for treating occlusive disease rather than PAA. The device is usually oversized by 10%-15%, but no more than that, because of the risk of graft infolding, Dr. Cayne said.
If more than one graft is needed, a maximum of no more than 1-mm size differential between grafts is suggested. A minimum overlap of 2-3 cm between grafts is also preferred.
Last year, surgeons reported a dismal 50% occlusion rate within just 6 weeks in the endovascular treatment of six PAAs using a novel, multilayer stent (Cardiatis's Multilayer Aneurysm Repair System) (J. Endovasc. Ther. 2013;20:381-8), he observed.
Dr. Cayne reported receiving honoraria as a consultant from Cook Medical.
AT THE NORTHWESTERN VASCULAR SYMPOSIUM
Major finding: Five-year primary patency was 67% for open repair and 80% for endovascular repair (P less than .05).
Data source: Expert opinion and retrospective study of 79 popliteal artery aneurysm repairs.
Disclosures: Dr. Cayne reported receiving honoraria as a consultant from Cook Medical.
Protocol-based resuscitation fails to beat usual care in early sepsis
Protocol-based resuscitation did not reduce sepsis deaths or morbidity, compared with usual care, in the Protocolized Care for Early Septic Shock study conducted at 31 U.S. academic hospitals.
Among 1,341 evaluable patients, the primary endpoint of 60-day in-hospital mortality was 21% with protocol-based early goal-directed therapy (EGDT), 18.2% with protocol-based standard therapy that did not require the placement of a central venous catheter, administration of inotropes, or blood transfusions, and 18.9% with usual care according to the treating physician’s judgment.
The difference was not statistically significant between the protocol-based interventions and usual care (relative risk, 1.04; P = .83) or between protocol-based EGDT and protocol-based standard therapy (RR, 1.15; P = .31), according to data presented at the International Symposium on Intensive Care and Emergency Medicine and simultaneously published in the New England Journal of Medicine (N. Engl. J. Med. 2014 March 18 [doi:10.1056/NEJMoa1401602]).
The three groups also did not differ significantly with regard to 90-day mortality, incidence of cardiovascular or respiratory failure, length of hospital stay, or discharge disposition.
The results from ProCESS differ from those reported more than a decade ago in the pivotal, single-center Rivers et al. study, in which mortality was significantly reduced for patients with severe sepsis or septic shock treated with 6 hours of EGDT therapy versus standard therapy (N. Engl. J. Med. 2001;345:1368-77).
Though both trials used the same EGDT protocol delivered by a trained, dedicated team at each site, possible reasons for the discordance are that the Rivers study showed nearly perfect adherence to protocol and that its cohort was slightly older, had higher rates of preexisting heart and liver disease, and had a higher initial serum lactate level, noted Dr. Derek Angus, ProCESS principal investigator and chair of critical care medicine at the University of Pittsburgh.
In addition, changes in the management of critically ill patient in the ensuing decade, including use of lower hemoglobin levels as a threshold for transfusion, use of lung-protection strategies, and tighter blood sugar control, "may have helped lower the overall mortality and may have reduced the marginal benefit of alternative resuscitation strategies," he observed.
Dr. Angus received grant support from the National Institutes of General Medical Sciences and nonfinancial support from Edwards Inc. during the trial, grant support to his university from Eisai, and personal fees from Pfizer and MedImmune outside this trial.
"The critical role of the clinician in the early recognition of
sepsis continues to this day to be fundamental to our efforts to improve
the rate of survival," said pulmonologist Dr. Craig M. Lilly of the University of Massachusetts Memorial Medical Center, Boston. (N. Engl. J. Med. 2014 March 18 [doi:10.1056/NEJMe1402564]). He
cautioned, however, on using the findings to come to the "dubious
conclusion that protocols and decision prompts do not have a role in the
treatment of septic shock."
An important contribution of ProCESS
is the evidence it provides on the early recognition of, and antibiotic
treatment for, sepsis in improving survival, Dr. Lilly observed. Shock
was recognized early in most patients (randomization occurred at an
average of 3 hours after ED arrival), 76% received antimicrobial agents
by randomization, and 97% received IV antimicrobials 6 hours after
randomization.
ProCESS also provides "transformative insights
about the treatments for septic shock that bring generalizable benefits"
when septic shock is recognized in the first hours after ED arrival, he
stated. Notably, that use of central hemodynamic and oxygen-saturation
monitoring in the protocol-based EGDT group did not result in better
outcomes than those achieved with clinical assessment of the adequacy of
circulation.
"State legislation and clinical guidelines,
including those endorsed by the National Quality Forum, should be
updated to remove the requirement for central hemodynamic monitoring and
to focus on less costly, lower-risk, and equally effective
alternatives," Dr. Lilly wrote. Dr. Lilly reported no conflicts.
"The critical role of the clinician in the early recognition of
sepsis continues to this day to be fundamental to our efforts to improve
the rate of survival," said pulmonologist Dr. Craig M. Lilly of the University of Massachusetts Memorial Medical Center, Boston. (N. Engl. J. Med. 2014 March 18 [doi:10.1056/NEJMe1402564]). He
cautioned, however, on using the findings to come to the "dubious
conclusion that protocols and decision prompts do not have a role in the
treatment of septic shock."
An important contribution of ProCESS
is the evidence it provides on the early recognition of, and antibiotic
treatment for, sepsis in improving survival, Dr. Lilly observed. Shock
was recognized early in most patients (randomization occurred at an
average of 3 hours after ED arrival), 76% received antimicrobial agents
by randomization, and 97% received IV antimicrobials 6 hours after
randomization.
ProCESS also provides "transformative insights
about the treatments for septic shock that bring generalizable benefits"
when septic shock is recognized in the first hours after ED arrival, he
stated. Notably, that use of central hemodynamic and oxygen-saturation
monitoring in the protocol-based EGDT group did not result in better
outcomes than those achieved with clinical assessment of the adequacy of
circulation.
"State legislation and clinical guidelines,
including those endorsed by the National Quality Forum, should be
updated to remove the requirement for central hemodynamic monitoring and
to focus on less costly, lower-risk, and equally effective
alternatives," Dr. Lilly wrote. Dr. Lilly reported no conflicts.
"The critical role of the clinician in the early recognition of
sepsis continues to this day to be fundamental to our efforts to improve
the rate of survival," said pulmonologist Dr. Craig M. Lilly of the University of Massachusetts Memorial Medical Center, Boston. (N. Engl. J. Med. 2014 March 18 [doi:10.1056/NEJMe1402564]). He
cautioned, however, on using the findings to come to the "dubious
conclusion that protocols and decision prompts do not have a role in the
treatment of septic shock."
An important contribution of ProCESS
is the evidence it provides on the early recognition of, and antibiotic
treatment for, sepsis in improving survival, Dr. Lilly observed. Shock
was recognized early in most patients (randomization occurred at an
average of 3 hours after ED arrival), 76% received antimicrobial agents
by randomization, and 97% received IV antimicrobials 6 hours after
randomization.
ProCESS also provides "transformative insights
about the treatments for septic shock that bring generalizable benefits"
when septic shock is recognized in the first hours after ED arrival, he
stated. Notably, that use of central hemodynamic and oxygen-saturation
monitoring in the protocol-based EGDT group did not result in better
outcomes than those achieved with clinical assessment of the adequacy of
circulation.
"State legislation and clinical guidelines,
including those endorsed by the National Quality Forum, should be
updated to remove the requirement for central hemodynamic monitoring and
to focus on less costly, lower-risk, and equally effective
alternatives," Dr. Lilly wrote. Dr. Lilly reported no conflicts.
Protocol-based resuscitation did not reduce sepsis deaths or morbidity, compared with usual care, in the Protocolized Care for Early Septic Shock study conducted at 31 U.S. academic hospitals.
Among 1,341 evaluable patients, the primary endpoint of 60-day in-hospital mortality was 21% with protocol-based early goal-directed therapy (EGDT), 18.2% with protocol-based standard therapy that did not require the placement of a central venous catheter, administration of inotropes, or blood transfusions, and 18.9% with usual care according to the treating physician’s judgment.
The difference was not statistically significant between the protocol-based interventions and usual care (relative risk, 1.04; P = .83) or between protocol-based EGDT and protocol-based standard therapy (RR, 1.15; P = .31), according to data presented at the International Symposium on Intensive Care and Emergency Medicine and simultaneously published in the New England Journal of Medicine (N. Engl. J. Med. 2014 March 18 [doi:10.1056/NEJMoa1401602]).
The three groups also did not differ significantly with regard to 90-day mortality, incidence of cardiovascular or respiratory failure, length of hospital stay, or discharge disposition.
The results from ProCESS differ from those reported more than a decade ago in the pivotal, single-center Rivers et al. study, in which mortality was significantly reduced for patients with severe sepsis or septic shock treated with 6 hours of EGDT therapy versus standard therapy (N. Engl. J. Med. 2001;345:1368-77).
Though both trials used the same EGDT protocol delivered by a trained, dedicated team at each site, possible reasons for the discordance are that the Rivers study showed nearly perfect adherence to protocol and that its cohort was slightly older, had higher rates of preexisting heart and liver disease, and had a higher initial serum lactate level, noted Dr. Derek Angus, ProCESS principal investigator and chair of critical care medicine at the University of Pittsburgh.
In addition, changes in the management of critically ill patient in the ensuing decade, including use of lower hemoglobin levels as a threshold for transfusion, use of lung-protection strategies, and tighter blood sugar control, "may have helped lower the overall mortality and may have reduced the marginal benefit of alternative resuscitation strategies," he observed.
Dr. Angus received grant support from the National Institutes of General Medical Sciences and nonfinancial support from Edwards Inc. during the trial, grant support to his university from Eisai, and personal fees from Pfizer and MedImmune outside this trial.
Protocol-based resuscitation did not reduce sepsis deaths or morbidity, compared with usual care, in the Protocolized Care for Early Septic Shock study conducted at 31 U.S. academic hospitals.
Among 1,341 evaluable patients, the primary endpoint of 60-day in-hospital mortality was 21% with protocol-based early goal-directed therapy (EGDT), 18.2% with protocol-based standard therapy that did not require the placement of a central venous catheter, administration of inotropes, or blood transfusions, and 18.9% with usual care according to the treating physician’s judgment.
The difference was not statistically significant between the protocol-based interventions and usual care (relative risk, 1.04; P = .83) or between protocol-based EGDT and protocol-based standard therapy (RR, 1.15; P = .31), according to data presented at the International Symposium on Intensive Care and Emergency Medicine and simultaneously published in the New England Journal of Medicine (N. Engl. J. Med. 2014 March 18 [doi:10.1056/NEJMoa1401602]).
The three groups also did not differ significantly with regard to 90-day mortality, incidence of cardiovascular or respiratory failure, length of hospital stay, or discharge disposition.
The results from ProCESS differ from those reported more than a decade ago in the pivotal, single-center Rivers et al. study, in which mortality was significantly reduced for patients with severe sepsis or septic shock treated with 6 hours of EGDT therapy versus standard therapy (N. Engl. J. Med. 2001;345:1368-77).
Though both trials used the same EGDT protocol delivered by a trained, dedicated team at each site, possible reasons for the discordance are that the Rivers study showed nearly perfect adherence to protocol and that its cohort was slightly older, had higher rates of preexisting heart and liver disease, and had a higher initial serum lactate level, noted Dr. Derek Angus, ProCESS principal investigator and chair of critical care medicine at the University of Pittsburgh.
In addition, changes in the management of critically ill patient in the ensuing decade, including use of lower hemoglobin levels as a threshold for transfusion, use of lung-protection strategies, and tighter blood sugar control, "may have helped lower the overall mortality and may have reduced the marginal benefit of alternative resuscitation strategies," he observed.
Dr. Angus received grant support from the National Institutes of General Medical Sciences and nonfinancial support from Edwards Inc. during the trial, grant support to his university from Eisai, and personal fees from Pfizer and MedImmune outside this trial.
FROM ISICEM 2014
Major finding: Sixty-day mortality was 21% with protocol-based EGDT, 18.2% with protocol-based standard therapy, and 18.9% with usual care.
Data source: A prospective study of 1,341 patients with septic shock.
Disclosures: Dr. Angus received grant support from the National Institutes of General Medical Sciences and nonfinancial support from Edwards Inc. during the trial, grant support to his university from Eisai, and personal fees from Pfizer and MedImmune outside this trial.
Higher MAP target fails to improve sepsis survival
Use of a high mean arterial pressure during initial resuscitation in patients with septic shock did not improve mortality at 28 or 90 days in the multicenter, open-label SEPSISPAM trial.
The Surviving Sepsis Campaign guidelines recommend targeting a mean arterial pressure (MAP) of at least 65 mm Hg, but suggest a higher target may be better for patients with atherosclerosis or previous hypertension. Retrospective data also suggest a MAP of more than 75 mm Hg may be needed to maintain kidney function during early sepsis.
For the current trial, investigators at 29 centers in France evenly randomized 776 patients to vasopressor treatment adjusted to maintain a MAP of 80-85 mm Hg (high-target group) or 65-70 mm Hg (low-target group).
The study’s primary endpoint of all-cause mortality at 28 days was 36.5% in the high-target group and 34% in the low-target group (nonsignificant hazard ratio in the high-target group, 1.07), according to data presented at the International Symposium on Intensive Care and Emergency Medicine and simultaneously published online (N. Engl. J. Med. 2014 March 18 [doi:10.1056/NEJMoa1312173]).
In addition, there were no significant differences between the high- and low-target groups in the secondary outcomes of 90-day mortality (43.7% vs. 42.3%; HR, 1.04), need for mechanical ventilation, ICU length of stay, or Sequential Organ Failure Assessment score at day 7.
Atrial fibrillation, however, was significantly more common in the high-target group than in the low-target group, at 6.7%, compared with 2.8%. This could be related to the high-target group receiving significantly higher doses of vasopressor catecholamines over a significantly longer time period, although other confounding factors cannot be ruled out, lead author Dr. Pierre Asfar of University Hospital of Angers (France), reported on behalf of SEPSISPAM investigators.
Among patients with chronic arterial hypertension, who comprised more than 40% of the study population, use of the high MAP target significantly reduced both the incidence of doubling of plasma creatinine (39% vs. 52%) and the rate of renal-replacement therapy (31.7% vs. 42.2%).
The authors noted that, although investigators were asked to treat patients to a MAP of 65-70 mm Hg in the low-target group, the observed pressures were for the most part between 70 and 75 mm Hg. The high-target group was likewise off goal, at a mean of 70 mm Hg. They also acknowledged that the lower-than-expected death rate, albeit in line with more recent trials, led to an underpowered study.
The French Ministry of Health funded the trial. Dr. Asfar reported lecture fees from LFB Biomedicaments.
Use of a high mean arterial pressure during initial resuscitation in patients with septic shock did not improve mortality at 28 or 90 days in the multicenter, open-label SEPSISPAM trial.
The Surviving Sepsis Campaign guidelines recommend targeting a mean arterial pressure (MAP) of at least 65 mm Hg, but suggest a higher target may be better for patients with atherosclerosis or previous hypertension. Retrospective data also suggest a MAP of more than 75 mm Hg may be needed to maintain kidney function during early sepsis.
For the current trial, investigators at 29 centers in France evenly randomized 776 patients to vasopressor treatment adjusted to maintain a MAP of 80-85 mm Hg (high-target group) or 65-70 mm Hg (low-target group).
The study’s primary endpoint of all-cause mortality at 28 days was 36.5% in the high-target group and 34% in the low-target group (nonsignificant hazard ratio in the high-target group, 1.07), according to data presented at the International Symposium on Intensive Care and Emergency Medicine and simultaneously published online (N. Engl. J. Med. 2014 March 18 [doi:10.1056/NEJMoa1312173]).
In addition, there were no significant differences between the high- and low-target groups in the secondary outcomes of 90-day mortality (43.7% vs. 42.3%; HR, 1.04), need for mechanical ventilation, ICU length of stay, or Sequential Organ Failure Assessment score at day 7.
Atrial fibrillation, however, was significantly more common in the high-target group than in the low-target group, at 6.7%, compared with 2.8%. This could be related to the high-target group receiving significantly higher doses of vasopressor catecholamines over a significantly longer time period, although other confounding factors cannot be ruled out, lead author Dr. Pierre Asfar of University Hospital of Angers (France), reported on behalf of SEPSISPAM investigators.
Among patients with chronic arterial hypertension, who comprised more than 40% of the study population, use of the high MAP target significantly reduced both the incidence of doubling of plasma creatinine (39% vs. 52%) and the rate of renal-replacement therapy (31.7% vs. 42.2%).
The authors noted that, although investigators were asked to treat patients to a MAP of 65-70 mm Hg in the low-target group, the observed pressures were for the most part between 70 and 75 mm Hg. The high-target group was likewise off goal, at a mean of 70 mm Hg. They also acknowledged that the lower-than-expected death rate, albeit in line with more recent trials, led to an underpowered study.
The French Ministry of Health funded the trial. Dr. Asfar reported lecture fees from LFB Biomedicaments.
Use of a high mean arterial pressure during initial resuscitation in patients with septic shock did not improve mortality at 28 or 90 days in the multicenter, open-label SEPSISPAM trial.
The Surviving Sepsis Campaign guidelines recommend targeting a mean arterial pressure (MAP) of at least 65 mm Hg, but suggest a higher target may be better for patients with atherosclerosis or previous hypertension. Retrospective data also suggest a MAP of more than 75 mm Hg may be needed to maintain kidney function during early sepsis.
For the current trial, investigators at 29 centers in France evenly randomized 776 patients to vasopressor treatment adjusted to maintain a MAP of 80-85 mm Hg (high-target group) or 65-70 mm Hg (low-target group).
The study’s primary endpoint of all-cause mortality at 28 days was 36.5% in the high-target group and 34% in the low-target group (nonsignificant hazard ratio in the high-target group, 1.07), according to data presented at the International Symposium on Intensive Care and Emergency Medicine and simultaneously published online (N. Engl. J. Med. 2014 March 18 [doi:10.1056/NEJMoa1312173]).
In addition, there were no significant differences between the high- and low-target groups in the secondary outcomes of 90-day mortality (43.7% vs. 42.3%; HR, 1.04), need for mechanical ventilation, ICU length of stay, or Sequential Organ Failure Assessment score at day 7.
Atrial fibrillation, however, was significantly more common in the high-target group than in the low-target group, at 6.7%, compared with 2.8%. This could be related to the high-target group receiving significantly higher doses of vasopressor catecholamines over a significantly longer time period, although other confounding factors cannot be ruled out, lead author Dr. Pierre Asfar of University Hospital of Angers (France), reported on behalf of SEPSISPAM investigators.
Among patients with chronic arterial hypertension, who comprised more than 40% of the study population, use of the high MAP target significantly reduced both the incidence of doubling of plasma creatinine (39% vs. 52%) and the rate of renal-replacement therapy (31.7% vs. 42.2%).
The authors noted that, although investigators were asked to treat patients to a MAP of 65-70 mm Hg in the low-target group, the observed pressures were for the most part between 70 and 75 mm Hg. The high-target group was likewise off goal, at a mean of 70 mm Hg. They also acknowledged that the lower-than-expected death rate, albeit in line with more recent trials, led to an underpowered study.
The French Ministry of Health funded the trial. Dr. Asfar reported lecture fees from LFB Biomedicaments.
FROM ISICEM 2014
Major finding: The 28-day mortality was 36.5% in the high-target group and 34% in the low-target group (P = .57).
Data source: A multicenter, open-label trial of 776 patients with septic shock who were treated to a low (60-65 mm Hg) or high (80-85 mm Hg) target mean arterial pressure.
Disclosures: The French Ministry of Health funded the trial. Dr. Asfar reported lecture fees from LFB Biomedicaments.
The push for smaller, smarter cancer trials
The American Society of Clinical Oncology is pressing cancer researchers to rethink the design of future clinical trials to achieve larger gains in four common cancers.
The final recommendations, which come after months of deliberations and public comment, try to hit the sweet spot between proposing guidelines that are not obtainable, and thus ignored, and having ambitious yet realistic goals.
For pancreatic cancer, for example, the experts recommended that clinical trials seek to improve median overall survival by 50%, or 4-5 months, for patients eligible for FOLFIRINOX (leucovorin, fluorouracil, irinotecan, and oxaliplatin) and by 3-4 months for those eligible for gemcitabine (Gemzar) with or without nab-paclitaxel (Abraxane).
Overall survival (OS) was selected over progression-free survival as the primary endpoint, although it was acknowledged that OS poses challenges such as the need for longer follow-up, the potential confounding effect of post-study therapies, and use of second-line therapies for secondary mutations identified after progression during first-line targeted therapy.
Ultimately, an improvement in median OS of 2.5-6 months, depending on the setting, was identified as the minimum incremental improvement over standard therapy that would define a clinically meaningful outcome.
The recommendations, published March 17 in the Journal of Clinical Oncology (J. Clin. Oncol. 2014 Mar. 17 [doi:10.1200/JCO.2013.53.8009]), also note that incremental improvements should be accompanied by little to no added toxicity over current treatments, and that a highly toxic regimen should produce the greatest OS gains to be considered clinically meaningful.
"We expect that sponsors will appreciate the need for raising the bar with regard to clinical trial goals, but that they will be conservative in their adoption of the recommendations," Dr. Lee M. Ellis, committee chair and professor of surgery at the University of Texas M.D. Anderson Cancer Center, Houston, said in an interview. "Trials designed with less ambitious goals may still be of benefit to individual patients if trial endpoints are met and if we can develop methods to identify patients most likely to benefit from the intervention."
Achieving the "smaller and smarter" trials envisioned by the committee rests on the ability to select patients for targeted therapy based on the molecular drivers of their tumors, rather than enrolling all comers. Unfortunately, in many cases, targeted agents continue to be developed without complete understanding of the drug target and, therefore, companion diagnostics to aid in patient selection, the experts observed.
"It is difficult to hit a target when it is not certain where it is or if it is valid," agreed Dr. David M. Dilts, codirector of the Center for Management Research in Healthcare, Oregon Health & Science University, Portland, in an accompanying editorial (J. Clin. Oncol. 2014 Mar. 17 [doi:10.1200/JCO.2013.54.5277]). "This, not insubstantial risk, should be ameliorated in the near future as major clinical research organizations are banking specimens, some of which are highly annotated, and as technology to analyze such specimens becomes faster, better, and cheaper."
To further this goal, the expert committee calls on trial sponsors to develop comprehensive biospecimen banks for each trial.
"Obstacles to developing these banks include cost and the willingness and ability of trial sponsors to foot the bill," Dr. Ellis said. "However, we believe the investment will pay off in increasing our ability to understand the molecular drivers of cancer and, as a result, more appropriate targeted therapies for people with cancer."
QOL
Though quality of life was a common theme that arose in all working group discussions, the recommendations lack hard targets in this area. Instead, the working groups cited the 2011 approval of the Janus kinase 1 and 2 inhibitor ruxolitinib (Jakavi) for myelofibrosis as an example of how serial assessment of specific cancer-related symptoms can define a clinically meaningful outcome for patients.
"It is not enough to just mention how important quality of life is. A clinical trial must be designed with a suite of thoughtful, feasible, validated patient-reported outcome measures that capture clinical benefit," Ms. Musa Mayer, a long-time advocate for patients with metastatic breast cancer, said in an interview. "Observed adverse events can never fully account for the lived experience of a given treatment."
Breast cancer
For breast cancer, the committee selected metastatic triple-negative breast cancer that was previously untreated for metastatic disease. They recommend clinical trials aim for an increase in OS of 4.5-6 months, although it was noted that consensus was not achieved by the breast cancer group on the magnitude of the benefit that would be considered clinically meaningful. The current median overall survival in this poor-prognosis population is 18 months.
Lung cancer
The committee addressed two lung cancer populations: nonsquamous cell carcinoma and squamous cell carcinoma. They recommend clinical trials seek to improve OS by 3.25-4 months and by 2.5-3 months, respectively. Current baseline median OS in these groups is 13 and 10 months.
Colon cancer
The recommendations for colon cancer target patients with disease progression with all prior therapies, or who are not candidates for standard second- or third-line options. Here, the goal is to improve OS by 3-5 months over the current baseline median OS of 4-6 months.
Notably, the cost of delivering the recommended targets for all four cancers was not addressed by the committee. The ASCO Value of Cancer Care Task Force, however, is already tasked with evaluating the efficacy, toxicity, and cost of specific oncology treatments.
"The working group provided thoughtful recommendations for the topics considered, although the specific recommendations were limited," Ms. Patricia Haugen, breast cancer survivor and current member and previous chair of the Department of Defense Congressionally Directed Breast Cancer Research Program Integration Panel, said in an interview.
She is hopeful that the new recommendations will be followed, but said there needs to be broad support and commitment to changes that produce more meaningful clinical benefit. "That commitment must be real and must come from all parties involved in the clinical trials process, so that clinical trials that do not meet a high bar are not considered, funded, nor implemented," she said.
Editorialist Dr. Dilts agreed that advocates from many areas are needed if the recommended goals are to be reached and suggested what might be required is "a more DARPA [Defense Advanced Research Projects Agency] approach, where answering high-risk questions are fostered and supported."
Dr. Ellis reported a consultant/advisory role with Genentech, Roche, Imclone, Eli Lilly, and Amgen. Ms. Mayer, Ms. Haugen, and Dr. Dilts reported no potential conflicts of interest.
The American Society of Clinical Oncology is pressing cancer researchers to rethink the design of future clinical trials to achieve larger gains in four common cancers.
The final recommendations, which come after months of deliberations and public comment, try to hit the sweet spot between proposing guidelines that are not obtainable, and thus ignored, and having ambitious yet realistic goals.
For pancreatic cancer, for example, the experts recommended that clinical trials seek to improve median overall survival by 50%, or 4-5 months, for patients eligible for FOLFIRINOX (leucovorin, fluorouracil, irinotecan, and oxaliplatin) and by 3-4 months for those eligible for gemcitabine (Gemzar) with or without nab-paclitaxel (Abraxane).
Overall survival (OS) was selected over progression-free survival as the primary endpoint, although it was acknowledged that OS poses challenges such as the need for longer follow-up, the potential confounding effect of post-study therapies, and use of second-line therapies for secondary mutations identified after progression during first-line targeted therapy.
Ultimately, an improvement in median OS of 2.5-6 months, depending on the setting, was identified as the minimum incremental improvement over standard therapy that would define a clinically meaningful outcome.
The recommendations, published March 17 in the Journal of Clinical Oncology (J. Clin. Oncol. 2014 Mar. 17 [doi:10.1200/JCO.2013.53.8009]), also note that incremental improvements should be accompanied by little to no added toxicity over current treatments, and that a highly toxic regimen should produce the greatest OS gains to be considered clinically meaningful.
"We expect that sponsors will appreciate the need for raising the bar with regard to clinical trial goals, but that they will be conservative in their adoption of the recommendations," Dr. Lee M. Ellis, committee chair and professor of surgery at the University of Texas M.D. Anderson Cancer Center, Houston, said in an interview. "Trials designed with less ambitious goals may still be of benefit to individual patients if trial endpoints are met and if we can develop methods to identify patients most likely to benefit from the intervention."
Achieving the "smaller and smarter" trials envisioned by the committee rests on the ability to select patients for targeted therapy based on the molecular drivers of their tumors, rather than enrolling all comers. Unfortunately, in many cases, targeted agents continue to be developed without complete understanding of the drug target and, therefore, companion diagnostics to aid in patient selection, the experts observed.
"It is difficult to hit a target when it is not certain where it is or if it is valid," agreed Dr. David M. Dilts, codirector of the Center for Management Research in Healthcare, Oregon Health & Science University, Portland, in an accompanying editorial (J. Clin. Oncol. 2014 Mar. 17 [doi:10.1200/JCO.2013.54.5277]). "This, not insubstantial risk, should be ameliorated in the near future as major clinical research organizations are banking specimens, some of which are highly annotated, and as technology to analyze such specimens becomes faster, better, and cheaper."
To further this goal, the expert committee calls on trial sponsors to develop comprehensive biospecimen banks for each trial.
"Obstacles to developing these banks include cost and the willingness and ability of trial sponsors to foot the bill," Dr. Ellis said. "However, we believe the investment will pay off in increasing our ability to understand the molecular drivers of cancer and, as a result, more appropriate targeted therapies for people with cancer."
QOL
Though quality of life was a common theme that arose in all working group discussions, the recommendations lack hard targets in this area. Instead, the working groups cited the 2011 approval of the Janus kinase 1 and 2 inhibitor ruxolitinib (Jakavi) for myelofibrosis as an example of how serial assessment of specific cancer-related symptoms can define a clinically meaningful outcome for patients.
"It is not enough to just mention how important quality of life is. A clinical trial must be designed with a suite of thoughtful, feasible, validated patient-reported outcome measures that capture clinical benefit," Ms. Musa Mayer, a long-time advocate for patients with metastatic breast cancer, said in an interview. "Observed adverse events can never fully account for the lived experience of a given treatment."
Breast cancer
For breast cancer, the committee selected metastatic triple-negative breast cancer that was previously untreated for metastatic disease. They recommend clinical trials aim for an increase in OS of 4.5-6 months, although it was noted that consensus was not achieved by the breast cancer group on the magnitude of the benefit that would be considered clinically meaningful. The current median overall survival in this poor-prognosis population is 18 months.
Lung cancer
The committee addressed two lung cancer populations: nonsquamous cell carcinoma and squamous cell carcinoma. They recommend clinical trials seek to improve OS by 3.25-4 months and by 2.5-3 months, respectively. Current baseline median OS in these groups is 13 and 10 months.
Colon cancer
The recommendations for colon cancer target patients with disease progression with all prior therapies, or who are not candidates for standard second- or third-line options. Here, the goal is to improve OS by 3-5 months over the current baseline median OS of 4-6 months.
Notably, the cost of delivering the recommended targets for all four cancers was not addressed by the committee. The ASCO Value of Cancer Care Task Force, however, is already tasked with evaluating the efficacy, toxicity, and cost of specific oncology treatments.
"The working group provided thoughtful recommendations for the topics considered, although the specific recommendations were limited," Ms. Patricia Haugen, breast cancer survivor and current member and previous chair of the Department of Defense Congressionally Directed Breast Cancer Research Program Integration Panel, said in an interview.
She is hopeful that the new recommendations will be followed, but said there needs to be broad support and commitment to changes that produce more meaningful clinical benefit. "That commitment must be real and must come from all parties involved in the clinical trials process, so that clinical trials that do not meet a high bar are not considered, funded, nor implemented," she said.
Editorialist Dr. Dilts agreed that advocates from many areas are needed if the recommended goals are to be reached and suggested what might be required is "a more DARPA [Defense Advanced Research Projects Agency] approach, where answering high-risk questions are fostered and supported."
Dr. Ellis reported a consultant/advisory role with Genentech, Roche, Imclone, Eli Lilly, and Amgen. Ms. Mayer, Ms. Haugen, and Dr. Dilts reported no potential conflicts of interest.
The American Society of Clinical Oncology is pressing cancer researchers to rethink the design of future clinical trials to achieve larger gains in four common cancers.
The final recommendations, which come after months of deliberations and public comment, try to hit the sweet spot between proposing guidelines that are not obtainable, and thus ignored, and having ambitious yet realistic goals.
For pancreatic cancer, for example, the experts recommended that clinical trials seek to improve median overall survival by 50%, or 4-5 months, for patients eligible for FOLFIRINOX (leucovorin, fluorouracil, irinotecan, and oxaliplatin) and by 3-4 months for those eligible for gemcitabine (Gemzar) with or without nab-paclitaxel (Abraxane).
Overall survival (OS) was selected over progression-free survival as the primary endpoint, although it was acknowledged that OS poses challenges such as the need for longer follow-up, the potential confounding effect of post-study therapies, and use of second-line therapies for secondary mutations identified after progression during first-line targeted therapy.
Ultimately, an improvement in median OS of 2.5-6 months, depending on the setting, was identified as the minimum incremental improvement over standard therapy that would define a clinically meaningful outcome.
The recommendations, published March 17 in the Journal of Clinical Oncology (J. Clin. Oncol. 2014 Mar. 17 [doi:10.1200/JCO.2013.53.8009]), also note that incremental improvements should be accompanied by little to no added toxicity over current treatments, and that a highly toxic regimen should produce the greatest OS gains to be considered clinically meaningful.
"We expect that sponsors will appreciate the need for raising the bar with regard to clinical trial goals, but that they will be conservative in their adoption of the recommendations," Dr. Lee M. Ellis, committee chair and professor of surgery at the University of Texas M.D. Anderson Cancer Center, Houston, said in an interview. "Trials designed with less ambitious goals may still be of benefit to individual patients if trial endpoints are met and if we can develop methods to identify patients most likely to benefit from the intervention."
Achieving the "smaller and smarter" trials envisioned by the committee rests on the ability to select patients for targeted therapy based on the molecular drivers of their tumors, rather than enrolling all comers. Unfortunately, in many cases, targeted agents continue to be developed without complete understanding of the drug target and, therefore, companion diagnostics to aid in patient selection, the experts observed.
"It is difficult to hit a target when it is not certain where it is or if it is valid," agreed Dr. David M. Dilts, codirector of the Center for Management Research in Healthcare, Oregon Health & Science University, Portland, in an accompanying editorial (J. Clin. Oncol. 2014 Mar. 17 [doi:10.1200/JCO.2013.54.5277]). "This, not insubstantial risk, should be ameliorated in the near future as major clinical research organizations are banking specimens, some of which are highly annotated, and as technology to analyze such specimens becomes faster, better, and cheaper."
To further this goal, the expert committee calls on trial sponsors to develop comprehensive biospecimen banks for each trial.
"Obstacles to developing these banks include cost and the willingness and ability of trial sponsors to foot the bill," Dr. Ellis said. "However, we believe the investment will pay off in increasing our ability to understand the molecular drivers of cancer and, as a result, more appropriate targeted therapies for people with cancer."
QOL
Though quality of life was a common theme that arose in all working group discussions, the recommendations lack hard targets in this area. Instead, the working groups cited the 2011 approval of the Janus kinase 1 and 2 inhibitor ruxolitinib (Jakavi) for myelofibrosis as an example of how serial assessment of specific cancer-related symptoms can define a clinically meaningful outcome for patients.
"It is not enough to just mention how important quality of life is. A clinical trial must be designed with a suite of thoughtful, feasible, validated patient-reported outcome measures that capture clinical benefit," Ms. Musa Mayer, a long-time advocate for patients with metastatic breast cancer, said in an interview. "Observed adverse events can never fully account for the lived experience of a given treatment."
Breast cancer
For breast cancer, the committee selected metastatic triple-negative breast cancer that was previously untreated for metastatic disease. They recommend clinical trials aim for an increase in OS of 4.5-6 months, although it was noted that consensus was not achieved by the breast cancer group on the magnitude of the benefit that would be considered clinically meaningful. The current median overall survival in this poor-prognosis population is 18 months.
Lung cancer
The committee addressed two lung cancer populations: nonsquamous cell carcinoma and squamous cell carcinoma. They recommend clinical trials seek to improve OS by 3.25-4 months and by 2.5-3 months, respectively. Current baseline median OS in these groups is 13 and 10 months.
Colon cancer
The recommendations for colon cancer target patients with disease progression with all prior therapies, or who are not candidates for standard second- or third-line options. Here, the goal is to improve OS by 3-5 months over the current baseline median OS of 4-6 months.
Notably, the cost of delivering the recommended targets for all four cancers was not addressed by the committee. The ASCO Value of Cancer Care Task Force, however, is already tasked with evaluating the efficacy, toxicity, and cost of specific oncology treatments.
"The working group provided thoughtful recommendations for the topics considered, although the specific recommendations were limited," Ms. Patricia Haugen, breast cancer survivor and current member and previous chair of the Department of Defense Congressionally Directed Breast Cancer Research Program Integration Panel, said in an interview.
She is hopeful that the new recommendations will be followed, but said there needs to be broad support and commitment to changes that produce more meaningful clinical benefit. "That commitment must be real and must come from all parties involved in the clinical trials process, so that clinical trials that do not meet a high bar are not considered, funded, nor implemented," she said.
Editorialist Dr. Dilts agreed that advocates from many areas are needed if the recommended goals are to be reached and suggested what might be required is "a more DARPA [Defense Advanced Research Projects Agency] approach, where answering high-risk questions are fostered and supported."
Dr. Ellis reported a consultant/advisory role with Genentech, Roche, Imclone, Eli Lilly, and Amgen. Ms. Mayer, Ms. Haugen, and Dr. Dilts reported no potential conflicts of interest.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Mindfulness Training Shifts Patients with Diabetes Off Autopilot
SAN FRANCISCO – Cultivating mindfulness in patients with diabetes is a useful self-help strategy to combat depression and foster healthy habits.
"How can you effectively manage yourself if you’re not aware of your self?" posited Dr. Laura Young at the annual advanced postgraduate course held by the American Diabetes Association.
One in four patients living with diabetes will develop depression in their lifetime, with 80% relapsing within 5 years of their depression going into remission, she said. Comorbid diabetes and depression increase mortality 2.5-fold, as well as raising the risk of all diabetes complications.
Mindfulness-based interventions teach individuals to be in the present moment, without judgment or emotional reactivity. The benefits are thought to be improved clarity, awareness, and acceptance.
Mindfulness also helps to weaken old, unhelpful, and automatic thinking habits that can potentially prevent positive action. This is critical for patients with diabetes, particularly those with type 1, who tend to go into autopilot when managing their disease, said Dr. Young, with the University of North Carolina in Durham.
"So many of our patients just get into a pattern of ‘It’s just what I’ve always done,’ " she said. "Mindfulness can help patients take a step back, evaluate those habits, and decide which are helping and which are not so great, and potentially change those habits."
Dr. Young recalled a recent patient who each evening had a bedtime snack that was sometimes healthful, but more often, not. The patient could offer no concrete reason for this snacking habit and admitted that 75% of the time he wasn’t even hungry.
"It hadn’t even occurred to him that it was something he shouldn’t be doing because it was something he’d always done," she said.
Mindfulness-based interventions have been studied for decades and have some of the best, most robust evidence behind them among self-help strategies, Dr. Young observed. In the Heidelberger Diabetes and Stress Study, patients with type 2 diabetes randomized to a mindfulness-based intervention had lower levels of depression, psychosocial distress, and improved health status over 5 years of follow-up compared with treatment-as-usual controls (Diabetes Care 2012;35:945-7).
A recent study found a mindful-eating intervention was as effective as a diabetes education self-management "Smart Choices" intervention in significantly improving depressive symptoms, cognitive control, and disinhibition of control regarding eating habits in adults, aged 35-65 years, with type 2 diabetes not on insulin therapy. Weight control at 3 months was also similar with either group-based intervention (Health Educ. Behav. 2014 April;41:145-54)
Mindfulness interventions have been readily adopted in some areas, often combined with yoga or cognitive-behavioral therapy, but these could be a hard sell for those who envision sitting cross-legged on yoga mats in the lotus position.
"I have a lot of patients from rural North Carolina, and they aren’t going to buy into that," Dr. Young said.
For these patients, her university offers a 2.5-hour class in which patients simply sit in a circle on chairs while being guided through mindfulness exercises. An ongoing study at the university is comparing this approach with a health education control group on physiological and psychological outcomes in patients with type 2 diabetes and high levels of diabetes-related distress, she said.
A show of hands at the meeting revealed that about a third of the audience is already using mindfulness training in their patients with diabetes. Dr. Young pointed out that the meeting host city of San Francisco had 386 therapists who use a combination of cognitive-behavioral therapy and mindfulness, whereas 40 therapists are available in Mississippi, as are a handful in rural North Carolina.
"So your patients can do this," she added.
Dr. Young reported research support paid to her university from numerous pharmaceutical firms and the National Institutes of Health.
SAN FRANCISCO – Cultivating mindfulness in patients with diabetes is a useful self-help strategy to combat depression and foster healthy habits.
"How can you effectively manage yourself if you’re not aware of your self?" posited Dr. Laura Young at the annual advanced postgraduate course held by the American Diabetes Association.
One in four patients living with diabetes will develop depression in their lifetime, with 80% relapsing within 5 years of their depression going into remission, she said. Comorbid diabetes and depression increase mortality 2.5-fold, as well as raising the risk of all diabetes complications.
Mindfulness-based interventions teach individuals to be in the present moment, without judgment or emotional reactivity. The benefits are thought to be improved clarity, awareness, and acceptance.
Mindfulness also helps to weaken old, unhelpful, and automatic thinking habits that can potentially prevent positive action. This is critical for patients with diabetes, particularly those with type 1, who tend to go into autopilot when managing their disease, said Dr. Young, with the University of North Carolina in Durham.
"So many of our patients just get into a pattern of ‘It’s just what I’ve always done,’ " she said. "Mindfulness can help patients take a step back, evaluate those habits, and decide which are helping and which are not so great, and potentially change those habits."
Dr. Young recalled a recent patient who each evening had a bedtime snack that was sometimes healthful, but more often, not. The patient could offer no concrete reason for this snacking habit and admitted that 75% of the time he wasn’t even hungry.
"It hadn’t even occurred to him that it was something he shouldn’t be doing because it was something he’d always done," she said.
Mindfulness-based interventions have been studied for decades and have some of the best, most robust evidence behind them among self-help strategies, Dr. Young observed. In the Heidelberger Diabetes and Stress Study, patients with type 2 diabetes randomized to a mindfulness-based intervention had lower levels of depression, psychosocial distress, and improved health status over 5 years of follow-up compared with treatment-as-usual controls (Diabetes Care 2012;35:945-7).
A recent study found a mindful-eating intervention was as effective as a diabetes education self-management "Smart Choices" intervention in significantly improving depressive symptoms, cognitive control, and disinhibition of control regarding eating habits in adults, aged 35-65 years, with type 2 diabetes not on insulin therapy. Weight control at 3 months was also similar with either group-based intervention (Health Educ. Behav. 2014 April;41:145-54)
Mindfulness interventions have been readily adopted in some areas, often combined with yoga or cognitive-behavioral therapy, but these could be a hard sell for those who envision sitting cross-legged on yoga mats in the lotus position.
"I have a lot of patients from rural North Carolina, and they aren’t going to buy into that," Dr. Young said.
For these patients, her university offers a 2.5-hour class in which patients simply sit in a circle on chairs while being guided through mindfulness exercises. An ongoing study at the university is comparing this approach with a health education control group on physiological and psychological outcomes in patients with type 2 diabetes and high levels of diabetes-related distress, she said.
A show of hands at the meeting revealed that about a third of the audience is already using mindfulness training in their patients with diabetes. Dr. Young pointed out that the meeting host city of San Francisco had 386 therapists who use a combination of cognitive-behavioral therapy and mindfulness, whereas 40 therapists are available in Mississippi, as are a handful in rural North Carolina.
"So your patients can do this," she added.
Dr. Young reported research support paid to her university from numerous pharmaceutical firms and the National Institutes of Health.
SAN FRANCISCO – Cultivating mindfulness in patients with diabetes is a useful self-help strategy to combat depression and foster healthy habits.
"How can you effectively manage yourself if you’re not aware of your self?" posited Dr. Laura Young at the annual advanced postgraduate course held by the American Diabetes Association.
One in four patients living with diabetes will develop depression in their lifetime, with 80% relapsing within 5 years of their depression going into remission, she said. Comorbid diabetes and depression increase mortality 2.5-fold, as well as raising the risk of all diabetes complications.
Mindfulness-based interventions teach individuals to be in the present moment, without judgment or emotional reactivity. The benefits are thought to be improved clarity, awareness, and acceptance.
Mindfulness also helps to weaken old, unhelpful, and automatic thinking habits that can potentially prevent positive action. This is critical for patients with diabetes, particularly those with type 1, who tend to go into autopilot when managing their disease, said Dr. Young, with the University of North Carolina in Durham.
"So many of our patients just get into a pattern of ‘It’s just what I’ve always done,’ " she said. "Mindfulness can help patients take a step back, evaluate those habits, and decide which are helping and which are not so great, and potentially change those habits."
Dr. Young recalled a recent patient who each evening had a bedtime snack that was sometimes healthful, but more often, not. The patient could offer no concrete reason for this snacking habit and admitted that 75% of the time he wasn’t even hungry.
"It hadn’t even occurred to him that it was something he shouldn’t be doing because it was something he’d always done," she said.
Mindfulness-based interventions have been studied for decades and have some of the best, most robust evidence behind them among self-help strategies, Dr. Young observed. In the Heidelberger Diabetes and Stress Study, patients with type 2 diabetes randomized to a mindfulness-based intervention had lower levels of depression, psychosocial distress, and improved health status over 5 years of follow-up compared with treatment-as-usual controls (Diabetes Care 2012;35:945-7).
A recent study found a mindful-eating intervention was as effective as a diabetes education self-management "Smart Choices" intervention in significantly improving depressive symptoms, cognitive control, and disinhibition of control regarding eating habits in adults, aged 35-65 years, with type 2 diabetes not on insulin therapy. Weight control at 3 months was also similar with either group-based intervention (Health Educ. Behav. 2014 April;41:145-54)
Mindfulness interventions have been readily adopted in some areas, often combined with yoga or cognitive-behavioral therapy, but these could be a hard sell for those who envision sitting cross-legged on yoga mats in the lotus position.
"I have a lot of patients from rural North Carolina, and they aren’t going to buy into that," Dr. Young said.
For these patients, her university offers a 2.5-hour class in which patients simply sit in a circle on chairs while being guided through mindfulness exercises. An ongoing study at the university is comparing this approach with a health education control group on physiological and psychological outcomes in patients with type 2 diabetes and high levels of diabetes-related distress, she said.
A show of hands at the meeting revealed that about a third of the audience is already using mindfulness training in their patients with diabetes. Dr. Young pointed out that the meeting host city of San Francisco had 386 therapists who use a combination of cognitive-behavioral therapy and mindfulness, whereas 40 therapists are available in Mississippi, as are a handful in rural North Carolina.
"So your patients can do this," she added.
Dr. Young reported research support paid to her university from numerous pharmaceutical firms and the National Institutes of Health.
EXPERT ANALYSIS FROM THE ADA ADVANCED POSTGRADUATE COURSE
Mindfulness training shifts diabetic patients off autopilot
SAN FRANCISCO – Cultivating mindfulness in patients with diabetes is a useful self-help strategy to combat depression and foster healthy habits.
"How can you effectively manage yourself if you’re not aware of your self?" posited Dr. Laura Young at the annual advanced postgraduate course held by the American Diabetes Association.
One in four patients living with diabetes will develop depression in their lifetime, with 80% relapsing within 5 years of their depression going into remission, she said. Comorbid diabetes and depression increase mortality 2.5-fold, as well as raising the risk of all diabetes complications.
Mindfulness-based interventions teach individuals to be in the present moment, without judgment or emotional reactivity. The benefits are thought to be improved clarity, awareness, and acceptance.
Mindfulness also helps to weaken old, unhelpful, and automatic thinking habits that can potentially prevent positive action. This is critical for patients with diabetes, particularly those with type 1, who tend to go into autopilot when managing their disease, said Dr. Young, with the University of North Carolina in Durham.
"So many of our patients just get into a pattern of ‘It’s just what I’ve always done,’ " she said. "Mindfulness can help patients take a step back, evaluate those habits, and decide which are helping and which are not so great, and potentially change those habits."
Dr. Young recalled a recent patient who each evening had a bedtime snack that was sometimes healthful, but more often, not. The patient could offer no concrete reason for this snacking habit and admitted that 75% of the time he wasn’t even hungry.
"It hadn’t even occurred to him that it was something he shouldn’t be doing because it was something he’d always done," she said.
Mindfulness-based interventions have been studied for decades and have some of the best, most robust evidence behind them among self-help strategies, Dr. Young observed. In the Heidelberger Diabetes and Stress Study, patients with type 2 diabetes randomized to a mindfulness-based intervention had lower levels of depression, psychosocial distress, and improved health status over 5 years of follow-up compared with treatment-as-usual controls (Diabetes Care 2012;35:945-7).
A recent study found a mindful-eating intervention was as effective as a diabetes education self-management "Smart Choices" intervention in significantly improving depressive symptoms, cognitive control, and disinhibition of control regarding eating habits in adults, aged 35-65 years, with type 2 diabetes not on insulin therapy. Weight control at 3 months was also similar with either group-based intervention (Health Educ. Behav. 2014 April;41:145-54)
Mindfulness interventions have been readily adopted in some areas, often combined with yoga or cognitive-behavioral therapy, but these could be a hard sell for those who envision sitting cross-legged on yoga mats in the lotus position.
"I have a lot of patients from rural North Carolina, and they aren’t going to buy into that," Dr. Young said.
For these patients, her university offers a 2.5-hour class in which patients simply sit in a circle on chairs while being guided through mindfulness exercises. An ongoing study at the university is comparing this approach with a health education control group on physiological and psychological outcomes in patients with type 2 diabetes and high levels of diabetes-related distress, she said.
A show of hands at the meeting revealed that about a third of the audience is already using mindfulness training in their patients with diabetes. Dr. Young pointed out that the meeting host city of San Francisco had 386 therapists who use a combination of cognitive-behavioral therapy and mindfulness, whereas 40 therapists are available in Mississippi, as are a handful in rural North Carolina.
"So your patients can do this," she added.
Dr. Young reported research support paid to her university from numerous pharmaceutical firms and the National Institutes of Health.
SAN FRANCISCO – Cultivating mindfulness in patients with diabetes is a useful self-help strategy to combat depression and foster healthy habits.
"How can you effectively manage yourself if you’re not aware of your self?" posited Dr. Laura Young at the annual advanced postgraduate course held by the American Diabetes Association.
One in four patients living with diabetes will develop depression in their lifetime, with 80% relapsing within 5 years of their depression going into remission, she said. Comorbid diabetes and depression increase mortality 2.5-fold, as well as raising the risk of all diabetes complications.
Mindfulness-based interventions teach individuals to be in the present moment, without judgment or emotional reactivity. The benefits are thought to be improved clarity, awareness, and acceptance.
Mindfulness also helps to weaken old, unhelpful, and automatic thinking habits that can potentially prevent positive action. This is critical for patients with diabetes, particularly those with type 1, who tend to go into autopilot when managing their disease, said Dr. Young, with the University of North Carolina in Durham.
"So many of our patients just get into a pattern of ‘It’s just what I’ve always done,’ " she said. "Mindfulness can help patients take a step back, evaluate those habits, and decide which are helping and which are not so great, and potentially change those habits."
Dr. Young recalled a recent patient who each evening had a bedtime snack that was sometimes healthful, but more often, not. The patient could offer no concrete reason for this snacking habit and admitted that 75% of the time he wasn’t even hungry.
"It hadn’t even occurred to him that it was something he shouldn’t be doing because it was something he’d always done," she said.
Mindfulness-based interventions have been studied for decades and have some of the best, most robust evidence behind them among self-help strategies, Dr. Young observed. In the Heidelberger Diabetes and Stress Study, patients with type 2 diabetes randomized to a mindfulness-based intervention had lower levels of depression, psychosocial distress, and improved health status over 5 years of follow-up compared with treatment-as-usual controls (Diabetes Care 2012;35:945-7).
A recent study found a mindful-eating intervention was as effective as a diabetes education self-management "Smart Choices" intervention in significantly improving depressive symptoms, cognitive control, and disinhibition of control regarding eating habits in adults, aged 35-65 years, with type 2 diabetes not on insulin therapy. Weight control at 3 months was also similar with either group-based intervention (Health Educ. Behav. 2014 April;41:145-54)
Mindfulness interventions have been readily adopted in some areas, often combined with yoga or cognitive-behavioral therapy, but these could be a hard sell for those who envision sitting cross-legged on yoga mats in the lotus position.
"I have a lot of patients from rural North Carolina, and they aren’t going to buy into that," Dr. Young said.
For these patients, her university offers a 2.5-hour class in which patients simply sit in a circle on chairs while being guided through mindfulness exercises. An ongoing study at the university is comparing this approach with a health education control group on physiological and psychological outcomes in patients with type 2 diabetes and high levels of diabetes-related distress, she said.
A show of hands at the meeting revealed that about a third of the audience is already using mindfulness training in their patients with diabetes. Dr. Young pointed out that the meeting host city of San Francisco had 386 therapists who use a combination of cognitive-behavioral therapy and mindfulness, whereas 40 therapists are available in Mississippi, as are a handful in rural North Carolina.
"So your patients can do this," she added.
Dr. Young reported research support paid to her university from numerous pharmaceutical firms and the National Institutes of Health.
SAN FRANCISCO – Cultivating mindfulness in patients with diabetes is a useful self-help strategy to combat depression and foster healthy habits.
"How can you effectively manage yourself if you’re not aware of your self?" posited Dr. Laura Young at the annual advanced postgraduate course held by the American Diabetes Association.
One in four patients living with diabetes will develop depression in their lifetime, with 80% relapsing within 5 years of their depression going into remission, she said. Comorbid diabetes and depression increase mortality 2.5-fold, as well as raising the risk of all diabetes complications.
Mindfulness-based interventions teach individuals to be in the present moment, without judgment or emotional reactivity. The benefits are thought to be improved clarity, awareness, and acceptance.
Mindfulness also helps to weaken old, unhelpful, and automatic thinking habits that can potentially prevent positive action. This is critical for patients with diabetes, particularly those with type 1, who tend to go into autopilot when managing their disease, said Dr. Young, with the University of North Carolina in Durham.
"So many of our patients just get into a pattern of ‘It’s just what I’ve always done,’ " she said. "Mindfulness can help patients take a step back, evaluate those habits, and decide which are helping and which are not so great, and potentially change those habits."
Dr. Young recalled a recent patient who each evening had a bedtime snack that was sometimes healthful, but more often, not. The patient could offer no concrete reason for this snacking habit and admitted that 75% of the time he wasn’t even hungry.
"It hadn’t even occurred to him that it was something he shouldn’t be doing because it was something he’d always done," she said.
Mindfulness-based interventions have been studied for decades and have some of the best, most robust evidence behind them among self-help strategies, Dr. Young observed. In the Heidelberger Diabetes and Stress Study, patients with type 2 diabetes randomized to a mindfulness-based intervention had lower levels of depression, psychosocial distress, and improved health status over 5 years of follow-up compared with treatment-as-usual controls (Diabetes Care 2012;35:945-7).
A recent study found a mindful-eating intervention was as effective as a diabetes education self-management "Smart Choices" intervention in significantly improving depressive symptoms, cognitive control, and disinhibition of control regarding eating habits in adults, aged 35-65 years, with type 2 diabetes not on insulin therapy. Weight control at 3 months was also similar with either group-based intervention (Health Educ. Behav. 2014 April;41:145-54)
Mindfulness interventions have been readily adopted in some areas, often combined with yoga or cognitive-behavioral therapy, but these could be a hard sell for those who envision sitting cross-legged on yoga mats in the lotus position.
"I have a lot of patients from rural North Carolina, and they aren’t going to buy into that," Dr. Young said.
For these patients, her university offers a 2.5-hour class in which patients simply sit in a circle on chairs while being guided through mindfulness exercises. An ongoing study at the university is comparing this approach with a health education control group on physiological and psychological outcomes in patients with type 2 diabetes and high levels of diabetes-related distress, she said.
A show of hands at the meeting revealed that about a third of the audience is already using mindfulness training in their patients with diabetes. Dr. Young pointed out that the meeting host city of San Francisco had 386 therapists who use a combination of cognitive-behavioral therapy and mindfulness, whereas 40 therapists are available in Mississippi, as are a handful in rural North Carolina.
"So your patients can do this," she added.
Dr. Young reported research support paid to her university from numerous pharmaceutical firms and the National Institutes of Health.
EXPERT ANALYSIS FROM THE ADA ADVANCED POSTGRADUATE COURSE
U.S. tracking of HPV in cervical precancers confirms half due to 16/18
U.S. surveillance data show that half of high-grade cervical lesions are caused by human papillomavirus 16 and 18.
Another 25% are attributable to the five additional HPV types 31/33/45/52/58 included in the investigational 9-valent vaccine.
The analysis fills in some knowledge gaps regarding U.S. women and was based on 5,189 specimens drawn from women, aged 21-39 years, diagnosed with cervical intraepithelial neoplasia (CIN) 2 and 3 and adenocarcinoma in situ (AIS) (CIN2+) from 2008 through 2011 at five U.S. sites in the HPV-IMPACT sentinel surveillance project.
HPV 16 was the most commonly detected type among all lesions, while type 18 was relatively uncommon, accounting for only about 5% of lesions, Dr. Susan Hariri said at a meeting of the Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices (ACIP).
HPV 16 and 18 increased with lesion severity from 40% in CIN2 to 66% in CIN3/AIS.
Type 31 was the most common of the five additional HPV types in the 9-valent vaccine, followed by types 52 and 58, both of which are more frequent in CIN2 than CIN3. Types 33 and 45 were detected in less than 5% of specimens across all histologic types.
The most common other high-risk, oncogenic types not found in any vaccine were 35 and 51, identified in 7% and 9% of CIN2 lesions, said Dr. Hariri of the CDC’s National Center for HIV, Viral Hepatitis, STD, and TB Prevention.
American Nurses Association representative Carol Hayes, M.P.H., a certified nurse-midwife, questioned this finding and asked why types 33 and 45 were included in the 9-valent vaccine, but 35 and 51 were not.
A representative from Merck responded that the five additional types in the 9-valent vaccine were selected based on their attribution to cervical cancer lesions, not precancers.
Proportional type attribution data analyses by race and age revealed HPV 16 and 18 were most common across all age and racial/ethnic groups, although some differences were identified. Types 16 and 18 declined from 50% for all groups to about 44% among the oldest age group (55-99 years), where the five additional HPV vaccine types were on the rise.
Types 31/33/45/52/58 were more common in racial and ethnic groups (28%-32%) than in whites (22%), while other high-risk HPV types were more common in blacks (20%) than other racial groups (14%-16%).
"The reasons for these differences are not clear and are probably multifactorial, but may be due to differences in the underlying prevalence of HPV in the subpopulations or to differences in screening and treatment," Dr. Hariri said.
Dr. Alain Luxembourg of Merck, the vaccine developer, said the 9-valent vaccine has the potential not only to prevent cancer, but to prevent a lot of "precancerous lesions, which in countries with cervical cancer screening programs, means a lower need for invasive procedures."
He presented a summary of 9-valent clinical data including the pivotal efficacy trial among females, aged 16-26 years, which showed noninferior immunogenicity for HPV 16 and 18 compared with the quadrivalent vaccine, and about 97% protection against disease related to the five additional strains. An immunobridging study also showed noninferior immunogenicity in adolescents compared with adults.
Regulatory action is expected by the Food and Drug Administration on the 9-valent vaccine within the next 3 months, he said.
ACIP is not expected to vote on whether to recommend the 9-valent vaccine until February 2015 at the earliest, following further review of clinical and health economics data and regulatory approval, said Dr. Lauri Markowitz of the CDC’s National Center for HIV, Viral Hepatitis, STD, and TB Prevention.
Considerations for the candidate vaccine include routine vaccination at age 11 or 12 years, vaccination of older females and males who were not vaccinated at the recommended age, vaccination of persons fully or partially vaccinated with quadrivalent vaccine, and the timing for consideration of males, aged 16-26 years, because it’s anticipated that the vaccine will not be licensed in this age group at the time of first licensure.
ACIP’s HPV work group also plans to review reduced-dose vaccination schedules in more detail. Some jurisdictions are already using two-dose schedules in their national or provincial immunization programs, and GlaxoSmithKline’s Cervarix received European marketing approval in December 2013 as a two-dose schedule (0, 6 months) for girls aged 9-14 years, Dr. Markowitz said.
As ACIP members met, Sanofi Pasteur announced that its quadrivalent HPV vaccine, Gardasil, was one step closer to receiving European approval for a two-dose schedule (0, 6 months) in girls and boys aged 9-13 years.
Finally, ACIP’s members unanimously affirmed efforts to update its 2007 HPV vaccine statement. The new statement, which still needs to go through editing before a final vote, consolidates and clarifies existing recommendations and updates background and clinical trial data. It also adds a variety of new sections including postlicensure safety data, areas of ongoing research, and future priorities, including the 9-valent vaccine and reduced-dose schedules, she said.
Dr. Hariri and Dr. Markowitz reported having no financial disclosures. Dr. Luxembourg is an employee of Merck.
U.S. surveillance data show that half of high-grade cervical lesions are caused by human papillomavirus 16 and 18.
Another 25% are attributable to the five additional HPV types 31/33/45/52/58 included in the investigational 9-valent vaccine.
The analysis fills in some knowledge gaps regarding U.S. women and was based on 5,189 specimens drawn from women, aged 21-39 years, diagnosed with cervical intraepithelial neoplasia (CIN) 2 and 3 and adenocarcinoma in situ (AIS) (CIN2+) from 2008 through 2011 at five U.S. sites in the HPV-IMPACT sentinel surveillance project.
HPV 16 was the most commonly detected type among all lesions, while type 18 was relatively uncommon, accounting for only about 5% of lesions, Dr. Susan Hariri said at a meeting of the Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices (ACIP).
HPV 16 and 18 increased with lesion severity from 40% in CIN2 to 66% in CIN3/AIS.
Type 31 was the most common of the five additional HPV types in the 9-valent vaccine, followed by types 52 and 58, both of which are more frequent in CIN2 than CIN3. Types 33 and 45 were detected in less than 5% of specimens across all histologic types.
The most common other high-risk, oncogenic types not found in any vaccine were 35 and 51, identified in 7% and 9% of CIN2 lesions, said Dr. Hariri of the CDC’s National Center for HIV, Viral Hepatitis, STD, and TB Prevention.
American Nurses Association representative Carol Hayes, M.P.H., a certified nurse-midwife, questioned this finding and asked why types 33 and 45 were included in the 9-valent vaccine, but 35 and 51 were not.
A representative from Merck responded that the five additional types in the 9-valent vaccine were selected based on their attribution to cervical cancer lesions, not precancers.
Proportional type attribution data analyses by race and age revealed HPV 16 and 18 were most common across all age and racial/ethnic groups, although some differences were identified. Types 16 and 18 declined from 50% for all groups to about 44% among the oldest age group (55-99 years), where the five additional HPV vaccine types were on the rise.
Types 31/33/45/52/58 were more common in racial and ethnic groups (28%-32%) than in whites (22%), while other high-risk HPV types were more common in blacks (20%) than other racial groups (14%-16%).
"The reasons for these differences are not clear and are probably multifactorial, but may be due to differences in the underlying prevalence of HPV in the subpopulations or to differences in screening and treatment," Dr. Hariri said.
Dr. Alain Luxembourg of Merck, the vaccine developer, said the 9-valent vaccine has the potential not only to prevent cancer, but to prevent a lot of "precancerous lesions, which in countries with cervical cancer screening programs, means a lower need for invasive procedures."
He presented a summary of 9-valent clinical data including the pivotal efficacy trial among females, aged 16-26 years, which showed noninferior immunogenicity for HPV 16 and 18 compared with the quadrivalent vaccine, and about 97% protection against disease related to the five additional strains. An immunobridging study also showed noninferior immunogenicity in adolescents compared with adults.
Regulatory action is expected by the Food and Drug Administration on the 9-valent vaccine within the next 3 months, he said.
ACIP is not expected to vote on whether to recommend the 9-valent vaccine until February 2015 at the earliest, following further review of clinical and health economics data and regulatory approval, said Dr. Lauri Markowitz of the CDC’s National Center for HIV, Viral Hepatitis, STD, and TB Prevention.
Considerations for the candidate vaccine include routine vaccination at age 11 or 12 years, vaccination of older females and males who were not vaccinated at the recommended age, vaccination of persons fully or partially vaccinated with quadrivalent vaccine, and the timing for consideration of males, aged 16-26 years, because it’s anticipated that the vaccine will not be licensed in this age group at the time of first licensure.
ACIP’s HPV work group also plans to review reduced-dose vaccination schedules in more detail. Some jurisdictions are already using two-dose schedules in their national or provincial immunization programs, and GlaxoSmithKline’s Cervarix received European marketing approval in December 2013 as a two-dose schedule (0, 6 months) for girls aged 9-14 years, Dr. Markowitz said.
As ACIP members met, Sanofi Pasteur announced that its quadrivalent HPV vaccine, Gardasil, was one step closer to receiving European approval for a two-dose schedule (0, 6 months) in girls and boys aged 9-13 years.
Finally, ACIP’s members unanimously affirmed efforts to update its 2007 HPV vaccine statement. The new statement, which still needs to go through editing before a final vote, consolidates and clarifies existing recommendations and updates background and clinical trial data. It also adds a variety of new sections including postlicensure safety data, areas of ongoing research, and future priorities, including the 9-valent vaccine and reduced-dose schedules, she said.
Dr. Hariri and Dr. Markowitz reported having no financial disclosures. Dr. Luxembourg is an employee of Merck.
U.S. surveillance data show that half of high-grade cervical lesions are caused by human papillomavirus 16 and 18.
Another 25% are attributable to the five additional HPV types 31/33/45/52/58 included in the investigational 9-valent vaccine.
The analysis fills in some knowledge gaps regarding U.S. women and was based on 5,189 specimens drawn from women, aged 21-39 years, diagnosed with cervical intraepithelial neoplasia (CIN) 2 and 3 and adenocarcinoma in situ (AIS) (CIN2+) from 2008 through 2011 at five U.S. sites in the HPV-IMPACT sentinel surveillance project.
HPV 16 was the most commonly detected type among all lesions, while type 18 was relatively uncommon, accounting for only about 5% of lesions, Dr. Susan Hariri said at a meeting of the Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices (ACIP).
HPV 16 and 18 increased with lesion severity from 40% in CIN2 to 66% in CIN3/AIS.
Type 31 was the most common of the five additional HPV types in the 9-valent vaccine, followed by types 52 and 58, both of which are more frequent in CIN2 than CIN3. Types 33 and 45 were detected in less than 5% of specimens across all histologic types.
The most common other high-risk, oncogenic types not found in any vaccine were 35 and 51, identified in 7% and 9% of CIN2 lesions, said Dr. Hariri of the CDC’s National Center for HIV, Viral Hepatitis, STD, and TB Prevention.
American Nurses Association representative Carol Hayes, M.P.H., a certified nurse-midwife, questioned this finding and asked why types 33 and 45 were included in the 9-valent vaccine, but 35 and 51 were not.
A representative from Merck responded that the five additional types in the 9-valent vaccine were selected based on their attribution to cervical cancer lesions, not precancers.
Proportional type attribution data analyses by race and age revealed HPV 16 and 18 were most common across all age and racial/ethnic groups, although some differences were identified. Types 16 and 18 declined from 50% for all groups to about 44% among the oldest age group (55-99 years), where the five additional HPV vaccine types were on the rise.
Types 31/33/45/52/58 were more common in racial and ethnic groups (28%-32%) than in whites (22%), while other high-risk HPV types were more common in blacks (20%) than other racial groups (14%-16%).
"The reasons for these differences are not clear and are probably multifactorial, but may be due to differences in the underlying prevalence of HPV in the subpopulations or to differences in screening and treatment," Dr. Hariri said.
Dr. Alain Luxembourg of Merck, the vaccine developer, said the 9-valent vaccine has the potential not only to prevent cancer, but to prevent a lot of "precancerous lesions, which in countries with cervical cancer screening programs, means a lower need for invasive procedures."
He presented a summary of 9-valent clinical data including the pivotal efficacy trial among females, aged 16-26 years, which showed noninferior immunogenicity for HPV 16 and 18 compared with the quadrivalent vaccine, and about 97% protection against disease related to the five additional strains. An immunobridging study also showed noninferior immunogenicity in adolescents compared with adults.
Regulatory action is expected by the Food and Drug Administration on the 9-valent vaccine within the next 3 months, he said.
ACIP is not expected to vote on whether to recommend the 9-valent vaccine until February 2015 at the earliest, following further review of clinical and health economics data and regulatory approval, said Dr. Lauri Markowitz of the CDC’s National Center for HIV, Viral Hepatitis, STD, and TB Prevention.
Considerations for the candidate vaccine include routine vaccination at age 11 or 12 years, vaccination of older females and males who were not vaccinated at the recommended age, vaccination of persons fully or partially vaccinated with quadrivalent vaccine, and the timing for consideration of males, aged 16-26 years, because it’s anticipated that the vaccine will not be licensed in this age group at the time of first licensure.
ACIP’s HPV work group also plans to review reduced-dose vaccination schedules in more detail. Some jurisdictions are already using two-dose schedules in their national or provincial immunization programs, and GlaxoSmithKline’s Cervarix received European marketing approval in December 2013 as a two-dose schedule (0, 6 months) for girls aged 9-14 years, Dr. Markowitz said.
As ACIP members met, Sanofi Pasteur announced that its quadrivalent HPV vaccine, Gardasil, was one step closer to receiving European approval for a two-dose schedule (0, 6 months) in girls and boys aged 9-13 years.
Finally, ACIP’s members unanimously affirmed efforts to update its 2007 HPV vaccine statement. The new statement, which still needs to go through editing before a final vote, consolidates and clarifies existing recommendations and updates background and clinical trial data. It also adds a variety of new sections including postlicensure safety data, areas of ongoing research, and future priorities, including the 9-valent vaccine and reduced-dose schedules, she said.
Dr. Hariri and Dr. Markowitz reported having no financial disclosures. Dr. Luxembourg is an employee of Merck.
FROM AN ACIP MEETING
Major finding: HPV 16 was the most commonly detected type among all lesions, while type 18 was relatively uncommon, accounting for only about 5% of lesions.
Data source: A retrospective analysis of 5,189 cervical specimens in the HPV-IMPACT project.
Disclosures: Dr. Hariri and Dr. Markowitz reported having no financial disclosures. Dr. Luxembourg is an employee of Merck.
CRP levels signal cytokine storm in CAR therapy
C-reactive protein levels can serve as a reliable indicator for severe cytokine release syndrome in patients receiving chimeric antigen receptor T-cell therapy, according to additional analyses from a phase I trial.
"We have found that daily monitoring of CRP [C-reactive protein] in combination with simple clinical parameters allows us to identify patients in need of intensive medical monitoring and potentially pharmacologic management," reported Dr. Marco Davila in Science Translational Medicine.
The observations were based on analysis of clinical data and serum cytokine levels over the first 21 days after infusing CD19-targeted 19-28z chimeric antigen receptor (CAR) T cells into 16 patients with relapsed or refractory B-cell acute lymphoblastic leukemia (B-ALL) (Sci. Transl. Med. 2014;6:1-9).
CAR therapy has demonstrated activity against several treatment-refractory leukemias, but in some patients has induced cytokine release syndrome (CRS), a clinical syndrome of fevers, hypotension, hypoxia, and neurologic changes including seizures.
Patients with CRS typically have fevers that start about 24 hours after infusion with 19-28z CAR T cells and can persist for several days. These fevers, however, aren’t always the harbinger for more clinically relevant toxicities, and premature intervention to curb the CRS can diminish T-cell persistence or efficacy, explained Dr. Davila of Memorial Sloan-Kettering Cancer Center in New York.
In an effort to distinguish between patients whose fevers would spontaneously resolve and those with clinically meaningful or severe CRS, the investigators retrospectively analyzed cytokine increases in serum samples from all 16 patients on the phase I trial. Of the 39 cytokines screened, 7 inflammatory cytokines correlated with pretreatment tumor burden and severe CRS. They are interferon-gamma, interleukin-5, IL-6, IL-10, Flt-3L, granulocyte-macrophage colony-stimulating factor, and fractalkine.
In addition, patients requiring intensive medical intervention had a 75-fold increase over pretreatment baseline levels in two of the seven cytokines, and universally exhibited at least one clinical sign of toxicity such as hypotension, hypoxia, or neurologic disorders.
"Thus, on the basis of the combined clinical and cytokine data, we could accurately define a severe CRS in those patients with the triad of persistent fevers (38° C) for more than 3 days, selected cytokine elevations, and additional clinical evidence of toxicity," Dr. Davila reported.
CRP as a surrogate
Cytokine monitoring, however, is unlikely to be performed daily because of cost and time constraints. CRP was selected as a surrogate for cytokine elevation based on the well-known association between CRP levels and IL-6 and the clinical amelioration of severe CRS afforded by IL-6 receptor blockade.
"We observed a clear difference between the CRP levels of patients with a severe CRS vs. patients classified as having either mild or no CRS," he noted.
Only those patients who met the criteria for severe CRS had a CRP level of 20 mg/dL or more. Patients whose CRP exceeded this threshold were at particularly high risk for CRS (sensitivity, 86%; specificity, 100%).
"CRP, a routinely available lab test, can serve as a surrogate for predicting development of the CRS," coauthor and colleague Dr. Renier J. Brentjens said in an interview. "This allows for sensitive day-to-day assessment of cytokine release in treated patients, which will allow for better management of CRS in CAR T-cell-treated patients," said Dr. Brentjens, director of cellular therapeutics at Memorial Sloan-Kettering Cancer Center.
Dr. David L. Porter, director of blood and marrow transplantation at the University of Pennsylvania Hospital, Philadelphia, said they too have found that CRP tracks with severity of CRS. However, at least at this point, while it may help predict which patients have a more severe reaction, it does not allow for early or different intervention.
"It remains completely unknown whether early intervention for CRS will inhibit the T-cell activity," he said in an interview. "We have developed a grading system for CRS that was designed to help determine when patients require intervention for CRS based on clinical criteria rather than laboratory values. But ultimately, if CRP or other measurements predict when intervention is appropriate, this will be a very useful assay."
CRS intervention
Treating severe CRS with high-dose steroids resulted in rapid reversal of fevers, cytokines, and other clinical symptoms, but abrogated T-cell expansion and persistence, whereas IL-6 receptor blockade with tocilizumab (Actemra) did not, Dr. Avila reported. Indeed, two patients demonstrated an almost 7,000-fold expansion after first-line tocilizumab.
In addition, the manner of treating severe CRS may affect clinical outcomes. All three patients treated with high-dose steroids relapsed despite previously achieving a complete molecular remission whereas untreated patients or those treated with tocilizumab alone had no evidence of recurrent disease after achieving a complete molecular remission.
Efficacy
The complete response rate in the study was 88%, with 12 of these 14 patients further classified as having minimal residual disease. Seven (44%) patients were successfully bridged to allogeneic stem cell transplantation. This is "especially meaningful" when compared with the historic low of just 5% of relapsed/refractory adult B-ALL patients transitioning to transplant after salvage chemotherapy, Dr. Davila noted.
Still, the persistence of the 19-28z CAR T cells was about 3 months, compared with several months to even more than one year in at least one B-ALL patient and several CLL patients reported by the University of Pennsylvania researchers. Dr. Davila and his colleagues hypothesize that the longer persistence of CD19-targeted CARs incorporating a 4-1BB moiety, rather than CD28, may be due, in part, to antigen-independent signaling through the 4-1BB CAR, as previously shown in preclinical studies. They are also currently developing a human anti-mouse antibody assay to determine whether immune-mediated rejection might be a contributing factor to the limited persistence of the 19-28z CAR T cells.
Dr. Porter said it’s not known why their T cells persist longer, but agreed it may have to do with the novel signal domain of their CAR (4-1BB). "This may be important for high levels of expansion, but may also provide a survival signal to the T cells allowing for longer persistence," he said.
The study was supported by MSKCC. Dr. Davila reported having no financial disclosures. Dr. Brentjens holds a patent that covers the 19-28z receptor and is a cofounder of Juno Therapeutics, which holds the license.
C-reactive protein levels can serve as a reliable indicator for severe cytokine release syndrome in patients receiving chimeric antigen receptor T-cell therapy, according to additional analyses from a phase I trial.
"We have found that daily monitoring of CRP [C-reactive protein] in combination with simple clinical parameters allows us to identify patients in need of intensive medical monitoring and potentially pharmacologic management," reported Dr. Marco Davila in Science Translational Medicine.
The observations were based on analysis of clinical data and serum cytokine levels over the first 21 days after infusing CD19-targeted 19-28z chimeric antigen receptor (CAR) T cells into 16 patients with relapsed or refractory B-cell acute lymphoblastic leukemia (B-ALL) (Sci. Transl. Med. 2014;6:1-9).
CAR therapy has demonstrated activity against several treatment-refractory leukemias, but in some patients has induced cytokine release syndrome (CRS), a clinical syndrome of fevers, hypotension, hypoxia, and neurologic changes including seizures.
Patients with CRS typically have fevers that start about 24 hours after infusion with 19-28z CAR T cells and can persist for several days. These fevers, however, aren’t always the harbinger for more clinically relevant toxicities, and premature intervention to curb the CRS can diminish T-cell persistence or efficacy, explained Dr. Davila of Memorial Sloan-Kettering Cancer Center in New York.
In an effort to distinguish between patients whose fevers would spontaneously resolve and those with clinically meaningful or severe CRS, the investigators retrospectively analyzed cytokine increases in serum samples from all 16 patients on the phase I trial. Of the 39 cytokines screened, 7 inflammatory cytokines correlated with pretreatment tumor burden and severe CRS. They are interferon-gamma, interleukin-5, IL-6, IL-10, Flt-3L, granulocyte-macrophage colony-stimulating factor, and fractalkine.
In addition, patients requiring intensive medical intervention had a 75-fold increase over pretreatment baseline levels in two of the seven cytokines, and universally exhibited at least one clinical sign of toxicity such as hypotension, hypoxia, or neurologic disorders.
"Thus, on the basis of the combined clinical and cytokine data, we could accurately define a severe CRS in those patients with the triad of persistent fevers (38° C) for more than 3 days, selected cytokine elevations, and additional clinical evidence of toxicity," Dr. Davila reported.
CRP as a surrogate
Cytokine monitoring, however, is unlikely to be performed daily because of cost and time constraints. CRP was selected as a surrogate for cytokine elevation based on the well-known association between CRP levels and IL-6 and the clinical amelioration of severe CRS afforded by IL-6 receptor blockade.
"We observed a clear difference between the CRP levels of patients with a severe CRS vs. patients classified as having either mild or no CRS," he noted.
Only those patients who met the criteria for severe CRS had a CRP level of 20 mg/dL or more. Patients whose CRP exceeded this threshold were at particularly high risk for CRS (sensitivity, 86%; specificity, 100%).
"CRP, a routinely available lab test, can serve as a surrogate for predicting development of the CRS," coauthor and colleague Dr. Renier J. Brentjens said in an interview. "This allows for sensitive day-to-day assessment of cytokine release in treated patients, which will allow for better management of CRS in CAR T-cell-treated patients," said Dr. Brentjens, director of cellular therapeutics at Memorial Sloan-Kettering Cancer Center.
Dr. David L. Porter, director of blood and marrow transplantation at the University of Pennsylvania Hospital, Philadelphia, said they too have found that CRP tracks with severity of CRS. However, at least at this point, while it may help predict which patients have a more severe reaction, it does not allow for early or different intervention.
"It remains completely unknown whether early intervention for CRS will inhibit the T-cell activity," he said in an interview. "We have developed a grading system for CRS that was designed to help determine when patients require intervention for CRS based on clinical criteria rather than laboratory values. But ultimately, if CRP or other measurements predict when intervention is appropriate, this will be a very useful assay."
CRS intervention
Treating severe CRS with high-dose steroids resulted in rapid reversal of fevers, cytokines, and other clinical symptoms, but abrogated T-cell expansion and persistence, whereas IL-6 receptor blockade with tocilizumab (Actemra) did not, Dr. Avila reported. Indeed, two patients demonstrated an almost 7,000-fold expansion after first-line tocilizumab.
In addition, the manner of treating severe CRS may affect clinical outcomes. All three patients treated with high-dose steroids relapsed despite previously achieving a complete molecular remission whereas untreated patients or those treated with tocilizumab alone had no evidence of recurrent disease after achieving a complete molecular remission.
Efficacy
The complete response rate in the study was 88%, with 12 of these 14 patients further classified as having minimal residual disease. Seven (44%) patients were successfully bridged to allogeneic stem cell transplantation. This is "especially meaningful" when compared with the historic low of just 5% of relapsed/refractory adult B-ALL patients transitioning to transplant after salvage chemotherapy, Dr. Davila noted.
Still, the persistence of the 19-28z CAR T cells was about 3 months, compared with several months to even more than one year in at least one B-ALL patient and several CLL patients reported by the University of Pennsylvania researchers. Dr. Davila and his colleagues hypothesize that the longer persistence of CD19-targeted CARs incorporating a 4-1BB moiety, rather than CD28, may be due, in part, to antigen-independent signaling through the 4-1BB CAR, as previously shown in preclinical studies. They are also currently developing a human anti-mouse antibody assay to determine whether immune-mediated rejection might be a contributing factor to the limited persistence of the 19-28z CAR T cells.
Dr. Porter said it’s not known why their T cells persist longer, but agreed it may have to do with the novel signal domain of their CAR (4-1BB). "This may be important for high levels of expansion, but may also provide a survival signal to the T cells allowing for longer persistence," he said.
The study was supported by MSKCC. Dr. Davila reported having no financial disclosures. Dr. Brentjens holds a patent that covers the 19-28z receptor and is a cofounder of Juno Therapeutics, which holds the license.
C-reactive protein levels can serve as a reliable indicator for severe cytokine release syndrome in patients receiving chimeric antigen receptor T-cell therapy, according to additional analyses from a phase I trial.
"We have found that daily monitoring of CRP [C-reactive protein] in combination with simple clinical parameters allows us to identify patients in need of intensive medical monitoring and potentially pharmacologic management," reported Dr. Marco Davila in Science Translational Medicine.
The observations were based on analysis of clinical data and serum cytokine levels over the first 21 days after infusing CD19-targeted 19-28z chimeric antigen receptor (CAR) T cells into 16 patients with relapsed or refractory B-cell acute lymphoblastic leukemia (B-ALL) (Sci. Transl. Med. 2014;6:1-9).
CAR therapy has demonstrated activity against several treatment-refractory leukemias, but in some patients has induced cytokine release syndrome (CRS), a clinical syndrome of fevers, hypotension, hypoxia, and neurologic changes including seizures.
Patients with CRS typically have fevers that start about 24 hours after infusion with 19-28z CAR T cells and can persist for several days. These fevers, however, aren’t always the harbinger for more clinically relevant toxicities, and premature intervention to curb the CRS can diminish T-cell persistence or efficacy, explained Dr. Davila of Memorial Sloan-Kettering Cancer Center in New York.
In an effort to distinguish between patients whose fevers would spontaneously resolve and those with clinically meaningful or severe CRS, the investigators retrospectively analyzed cytokine increases in serum samples from all 16 patients on the phase I trial. Of the 39 cytokines screened, 7 inflammatory cytokines correlated with pretreatment tumor burden and severe CRS. They are interferon-gamma, interleukin-5, IL-6, IL-10, Flt-3L, granulocyte-macrophage colony-stimulating factor, and fractalkine.
In addition, patients requiring intensive medical intervention had a 75-fold increase over pretreatment baseline levels in two of the seven cytokines, and universally exhibited at least one clinical sign of toxicity such as hypotension, hypoxia, or neurologic disorders.
"Thus, on the basis of the combined clinical and cytokine data, we could accurately define a severe CRS in those patients with the triad of persistent fevers (38° C) for more than 3 days, selected cytokine elevations, and additional clinical evidence of toxicity," Dr. Davila reported.
CRP as a surrogate
Cytokine monitoring, however, is unlikely to be performed daily because of cost and time constraints. CRP was selected as a surrogate for cytokine elevation based on the well-known association between CRP levels and IL-6 and the clinical amelioration of severe CRS afforded by IL-6 receptor blockade.
"We observed a clear difference between the CRP levels of patients with a severe CRS vs. patients classified as having either mild or no CRS," he noted.
Only those patients who met the criteria for severe CRS had a CRP level of 20 mg/dL or more. Patients whose CRP exceeded this threshold were at particularly high risk for CRS (sensitivity, 86%; specificity, 100%).
"CRP, a routinely available lab test, can serve as a surrogate for predicting development of the CRS," coauthor and colleague Dr. Renier J. Brentjens said in an interview. "This allows for sensitive day-to-day assessment of cytokine release in treated patients, which will allow for better management of CRS in CAR T-cell-treated patients," said Dr. Brentjens, director of cellular therapeutics at Memorial Sloan-Kettering Cancer Center.
Dr. David L. Porter, director of blood and marrow transplantation at the University of Pennsylvania Hospital, Philadelphia, said they too have found that CRP tracks with severity of CRS. However, at least at this point, while it may help predict which patients have a more severe reaction, it does not allow for early or different intervention.
"It remains completely unknown whether early intervention for CRS will inhibit the T-cell activity," he said in an interview. "We have developed a grading system for CRS that was designed to help determine when patients require intervention for CRS based on clinical criteria rather than laboratory values. But ultimately, if CRP or other measurements predict when intervention is appropriate, this will be a very useful assay."
CRS intervention
Treating severe CRS with high-dose steroids resulted in rapid reversal of fevers, cytokines, and other clinical symptoms, but abrogated T-cell expansion and persistence, whereas IL-6 receptor blockade with tocilizumab (Actemra) did not, Dr. Avila reported. Indeed, two patients demonstrated an almost 7,000-fold expansion after first-line tocilizumab.
In addition, the manner of treating severe CRS may affect clinical outcomes. All three patients treated with high-dose steroids relapsed despite previously achieving a complete molecular remission whereas untreated patients or those treated with tocilizumab alone had no evidence of recurrent disease after achieving a complete molecular remission.
Efficacy
The complete response rate in the study was 88%, with 12 of these 14 patients further classified as having minimal residual disease. Seven (44%) patients were successfully bridged to allogeneic stem cell transplantation. This is "especially meaningful" when compared with the historic low of just 5% of relapsed/refractory adult B-ALL patients transitioning to transplant after salvage chemotherapy, Dr. Davila noted.
Still, the persistence of the 19-28z CAR T cells was about 3 months, compared with several months to even more than one year in at least one B-ALL patient and several CLL patients reported by the University of Pennsylvania researchers. Dr. Davila and his colleagues hypothesize that the longer persistence of CD19-targeted CARs incorporating a 4-1BB moiety, rather than CD28, may be due, in part, to antigen-independent signaling through the 4-1BB CAR, as previously shown in preclinical studies. They are also currently developing a human anti-mouse antibody assay to determine whether immune-mediated rejection might be a contributing factor to the limited persistence of the 19-28z CAR T cells.
Dr. Porter said it’s not known why their T cells persist longer, but agreed it may have to do with the novel signal domain of their CAR (4-1BB). "This may be important for high levels of expansion, but may also provide a survival signal to the T cells allowing for longer persistence," he said.
The study was supported by MSKCC. Dr. Davila reported having no financial disclosures. Dr. Brentjens holds a patent that covers the 19-28z receptor and is a cofounder of Juno Therapeutics, which holds the license.
FROM SCIENCE TRANSLATIONAL MEDICINE
Major finding: CRP levels of at least 20 mg/dL had a sensitivity and specificity to predict severe CRS of 86% and 100%, respectively.
Data source: A retrospective study of 16 patients with relapsed or refractory B-cell acute lymphoblastic leukemia.
Disclosures: The study was supported by MSKCC. Dr. Davila reported having no financial disclosures. Dr. Brentjens holds a patent that covers the 19-28z receptor and is a cofounder of Juno Therapeutics, which holds the license.