User login
Low vitamin D – poor ICU outcomes
NAPLES, FLA. – Vitamin D deficiency is common in critically ill trauma patients and portends worse outcomes, a retrospective study suggests.
Among 200 trauma patients with available vitamin D levels, 26% were vitamin D deficient on ICU admission.
"These patients have a higher APACHE II score, have a longer ICU stay, and will likely be hospitalized greater than 2 weeks," Dr. Joseph Ibrahim reported at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
Long known to be essential for bone development and wound healing, recent studies have demonstrated that vitamin D deficiency is a significant predictor of 30- and 90-day all-cause mortality in critically ill patients, even after adjustment for such factors as age, Charlson/Deyo index, sepsis, and season (Crit. Care Med. 2012;40:63-72). It also has been shown to significantly predict acute kidney injury in the critically ill (Crit. Care Med. 2012;40:3170-9).
For the current analysis, vitamin D levels were drawn upon ICU admission, at 72 hours, and every 7 days until hospital discharge in 200 of 234 consecutive adult trauma patients admitted to the ICU at the Level 1 Orlando Regional Medical Center during a 4-month period. Deficiency was defined as 25-hydroxyvitamin D of 20 ng/mL or less. All patients received nutritional support using a standard protocol, but not vitamin D supplementation.
Median vitamin D ICU admission levels in the 51 vitamin D–deficient patients were significantly lower than for nondeficient patients (16 ng/mL vs. 28 ng/mL; P less than .001). Levels decreased a median of 4 ng/mL at 72 hours in both groups, but only the sufficient group returned to admission baseline levels at week 2, reported Dr. Ibrahim, a critical care surgeon with the medical center.
"This demonstrates that if we wish to obtain normal vitamin D levels in these patients, we will have to supplement them with much higher doses than what we are providing with standard enteral formulas," he said in an interview.
Patients with vitamin D deficiency spent more time than did nondeficient patients in the ICU (median 3 days vs. 2.7 days) and hospital (median 8.4 days vs. 7.1 days), but these trends did not reach statistical significance.
Significantly more deficient patients, however, remained in the hospital for at least 2 weeks (37% vs. 20%).
The investigators were unable to show a difference in mortality between the deficient and nondeficient groups (16% vs. 12%; P = .51), possibly because the study was underpowered, he said.
Deficient and sufficient patients did not differ in age (median 48 years vs. 44 years), body mass index (26.2 kg/m2 vs. 25.7 kg/m2), admission ionized calcium (1.06 mmol/L for both), or Injury Severity Score (14 vs. 13). Only APACHE II scores were significantly higher in deficient patients (20 vs. 15).
"It makes sense that with the significant difference in APACHE II score, one would expect to see a similar difference in mortality, but again we were unable to show this with this study," Dr. Ibrahim said.
Prehospital factors significantly associated with low vitamin D status were African American race, diabetes, and lack of vitamin D supplementation.
Vitamin D supplementation may be helpful in critically ill trauma patients during hospitalization, but more research is needed, Dr. Ibrahim said. The group is planning a supplementation study, looking at vitamin D dosing and frequency of testing.
"Our first goal was to demonstrate a significant incidence, which we did," he said. "It should be noted that the incidence was in a location with probably one of the highest amounts of sunshine in the country and that the findings may underestimate what one would find in other areas of the United States."
Dr. Oscar Guillamondegui, of Vanderbilt University Medical Center in Nashville, Tenn., who proctored the poster session, said he would expect vitamin D levels to be lower in acutely sick patients requiring ICU management because production of vitamin D–binding protein, a subprotein in the albumin family of proteins involved in vitamin D transport and storage, is decreased in high stress situations to allow for the increase in acute phase protein production.
"Although the data are intriguing, as a retrospective study, it is too early to suggest that supplementation is essential," he said.
Dr. Steven Q. Simpson, FCCP, comments: This is an interesting and provocative report associating vitamin D deficiency with worse outcome among trauma patients. As noted in the article, we have currently observed only an association, not cause and effect, so it is too soon to suggest routine measurement and/or supplementation of vitamin D in this patient population. However, given the known immunomodulating effects of vitamin D, there is biological plausibility in these findings, and I suspect that a story is about to unfold.
Dr. Ibrahim and Dr. Guillamondegui reported having no financial disclosures.
If you’re interested in more about these topics, you can join a discussion on this topic within the Critical Care e-Community. Simply log in to ecommunity.chestnet.org and find the Critical Care group. If you’re not part of the Critical Care NetWork, log in to chestnet.org and add the Critical Care NetWork to your profile.
Questions? Contact communityadmin@chestnet.org.
Dr. Steven Q. Simpson, FCCP, comments: This is an interesting and provocative report associating vitamin D deficiency with worse outcome among trauma patients. As noted in the article, we have currently observed only an association, not cause and effect, so it is too soon to suggest routine measurement and/or supplementation of vitamin D in this patient population. However, given the known immunomodulating effects of vitamin D, there is biological plausibility in these findings, and I suspect that a story is about to unfold.
Dr. Steven Q. Simpson, FCCP, comments: This is an interesting and provocative report associating vitamin D deficiency with worse outcome among trauma patients. As noted in the article, we have currently observed only an association, not cause and effect, so it is too soon to suggest routine measurement and/or supplementation of vitamin D in this patient population. However, given the known immunomodulating effects of vitamin D, there is biological plausibility in these findings, and I suspect that a story is about to unfold.
Dr. Steven Q. Simpson, FCCP, comments: This is an interesting and provocative report associating vitamin D deficiency with worse outcome among trauma patients. As noted in the article, we have currently observed only an association, not cause and effect, so it is too soon to suggest routine measurement and/or supplementation of vitamin D in this patient population. However, given the known immunomodulating effects of vitamin D, there is biological plausibility in these findings, and I suspect that a story is about to unfold.
NAPLES, FLA. – Vitamin D deficiency is common in critically ill trauma patients and portends worse outcomes, a retrospective study suggests.
Among 200 trauma patients with available vitamin D levels, 26% were vitamin D deficient on ICU admission.
"These patients have a higher APACHE II score, have a longer ICU stay, and will likely be hospitalized greater than 2 weeks," Dr. Joseph Ibrahim reported at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
Long known to be essential for bone development and wound healing, recent studies have demonstrated that vitamin D deficiency is a significant predictor of 30- and 90-day all-cause mortality in critically ill patients, even after adjustment for such factors as age, Charlson/Deyo index, sepsis, and season (Crit. Care Med. 2012;40:63-72). It also has been shown to significantly predict acute kidney injury in the critically ill (Crit. Care Med. 2012;40:3170-9).
For the current analysis, vitamin D levels were drawn upon ICU admission, at 72 hours, and every 7 days until hospital discharge in 200 of 234 consecutive adult trauma patients admitted to the ICU at the Level 1 Orlando Regional Medical Center during a 4-month period. Deficiency was defined as 25-hydroxyvitamin D of 20 ng/mL or less. All patients received nutritional support using a standard protocol, but not vitamin D supplementation.
Median vitamin D ICU admission levels in the 51 vitamin D–deficient patients were significantly lower than for nondeficient patients (16 ng/mL vs. 28 ng/mL; P less than .001). Levels decreased a median of 4 ng/mL at 72 hours in both groups, but only the sufficient group returned to admission baseline levels at week 2, reported Dr. Ibrahim, a critical care surgeon with the medical center.
"This demonstrates that if we wish to obtain normal vitamin D levels in these patients, we will have to supplement them with much higher doses than what we are providing with standard enteral formulas," he said in an interview.
Patients with vitamin D deficiency spent more time than did nondeficient patients in the ICU (median 3 days vs. 2.7 days) and hospital (median 8.4 days vs. 7.1 days), but these trends did not reach statistical significance.
Significantly more deficient patients, however, remained in the hospital for at least 2 weeks (37% vs. 20%).
The investigators were unable to show a difference in mortality between the deficient and nondeficient groups (16% vs. 12%; P = .51), possibly because the study was underpowered, he said.
Deficient and sufficient patients did not differ in age (median 48 years vs. 44 years), body mass index (26.2 kg/m2 vs. 25.7 kg/m2), admission ionized calcium (1.06 mmol/L for both), or Injury Severity Score (14 vs. 13). Only APACHE II scores were significantly higher in deficient patients (20 vs. 15).
"It makes sense that with the significant difference in APACHE II score, one would expect to see a similar difference in mortality, but again we were unable to show this with this study," Dr. Ibrahim said.
Prehospital factors significantly associated with low vitamin D status were African American race, diabetes, and lack of vitamin D supplementation.
Vitamin D supplementation may be helpful in critically ill trauma patients during hospitalization, but more research is needed, Dr. Ibrahim said. The group is planning a supplementation study, looking at vitamin D dosing and frequency of testing.
"Our first goal was to demonstrate a significant incidence, which we did," he said. "It should be noted that the incidence was in a location with probably one of the highest amounts of sunshine in the country and that the findings may underestimate what one would find in other areas of the United States."
Dr. Oscar Guillamondegui, of Vanderbilt University Medical Center in Nashville, Tenn., who proctored the poster session, said he would expect vitamin D levels to be lower in acutely sick patients requiring ICU management because production of vitamin D–binding protein, a subprotein in the albumin family of proteins involved in vitamin D transport and storage, is decreased in high stress situations to allow for the increase in acute phase protein production.
"Although the data are intriguing, as a retrospective study, it is too early to suggest that supplementation is essential," he said.
Dr. Steven Q. Simpson, FCCP, comments: This is an interesting and provocative report associating vitamin D deficiency with worse outcome among trauma patients. As noted in the article, we have currently observed only an association, not cause and effect, so it is too soon to suggest routine measurement and/or supplementation of vitamin D in this patient population. However, given the known immunomodulating effects of vitamin D, there is biological plausibility in these findings, and I suspect that a story is about to unfold.
Dr. Ibrahim and Dr. Guillamondegui reported having no financial disclosures.
If you’re interested in more about these topics, you can join a discussion on this topic within the Critical Care e-Community. Simply log in to ecommunity.chestnet.org and find the Critical Care group. If you’re not part of the Critical Care NetWork, log in to chestnet.org and add the Critical Care NetWork to your profile.
Questions? Contact communityadmin@chestnet.org.
NAPLES, FLA. – Vitamin D deficiency is common in critically ill trauma patients and portends worse outcomes, a retrospective study suggests.
Among 200 trauma patients with available vitamin D levels, 26% were vitamin D deficient on ICU admission.
"These patients have a higher APACHE II score, have a longer ICU stay, and will likely be hospitalized greater than 2 weeks," Dr. Joseph Ibrahim reported at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
Long known to be essential for bone development and wound healing, recent studies have demonstrated that vitamin D deficiency is a significant predictor of 30- and 90-day all-cause mortality in critically ill patients, even after adjustment for such factors as age, Charlson/Deyo index, sepsis, and season (Crit. Care Med. 2012;40:63-72). It also has been shown to significantly predict acute kidney injury in the critically ill (Crit. Care Med. 2012;40:3170-9).
For the current analysis, vitamin D levels were drawn upon ICU admission, at 72 hours, and every 7 days until hospital discharge in 200 of 234 consecutive adult trauma patients admitted to the ICU at the Level 1 Orlando Regional Medical Center during a 4-month period. Deficiency was defined as 25-hydroxyvitamin D of 20 ng/mL or less. All patients received nutritional support using a standard protocol, but not vitamin D supplementation.
Median vitamin D ICU admission levels in the 51 vitamin D–deficient patients were significantly lower than for nondeficient patients (16 ng/mL vs. 28 ng/mL; P less than .001). Levels decreased a median of 4 ng/mL at 72 hours in both groups, but only the sufficient group returned to admission baseline levels at week 2, reported Dr. Ibrahim, a critical care surgeon with the medical center.
"This demonstrates that if we wish to obtain normal vitamin D levels in these patients, we will have to supplement them with much higher doses than what we are providing with standard enteral formulas," he said in an interview.
Patients with vitamin D deficiency spent more time than did nondeficient patients in the ICU (median 3 days vs. 2.7 days) and hospital (median 8.4 days vs. 7.1 days), but these trends did not reach statistical significance.
Significantly more deficient patients, however, remained in the hospital for at least 2 weeks (37% vs. 20%).
The investigators were unable to show a difference in mortality between the deficient and nondeficient groups (16% vs. 12%; P = .51), possibly because the study was underpowered, he said.
Deficient and sufficient patients did not differ in age (median 48 years vs. 44 years), body mass index (26.2 kg/m2 vs. 25.7 kg/m2), admission ionized calcium (1.06 mmol/L for both), or Injury Severity Score (14 vs. 13). Only APACHE II scores were significantly higher in deficient patients (20 vs. 15).
"It makes sense that with the significant difference in APACHE II score, one would expect to see a similar difference in mortality, but again we were unable to show this with this study," Dr. Ibrahim said.
Prehospital factors significantly associated with low vitamin D status were African American race, diabetes, and lack of vitamin D supplementation.
Vitamin D supplementation may be helpful in critically ill trauma patients during hospitalization, but more research is needed, Dr. Ibrahim said. The group is planning a supplementation study, looking at vitamin D dosing and frequency of testing.
"Our first goal was to demonstrate a significant incidence, which we did," he said. "It should be noted that the incidence was in a location with probably one of the highest amounts of sunshine in the country and that the findings may underestimate what one would find in other areas of the United States."
Dr. Oscar Guillamondegui, of Vanderbilt University Medical Center in Nashville, Tenn., who proctored the poster session, said he would expect vitamin D levels to be lower in acutely sick patients requiring ICU management because production of vitamin D–binding protein, a subprotein in the albumin family of proteins involved in vitamin D transport and storage, is decreased in high stress situations to allow for the increase in acute phase protein production.
"Although the data are intriguing, as a retrospective study, it is too early to suggest that supplementation is essential," he said.
Dr. Steven Q. Simpson, FCCP, comments: This is an interesting and provocative report associating vitamin D deficiency with worse outcome among trauma patients. As noted in the article, we have currently observed only an association, not cause and effect, so it is too soon to suggest routine measurement and/or supplementation of vitamin D in this patient population. However, given the known immunomodulating effects of vitamin D, there is biological plausibility in these findings, and I suspect that a story is about to unfold.
Dr. Ibrahim and Dr. Guillamondegui reported having no financial disclosures.
If you’re interested in more about these topics, you can join a discussion on this topic within the Critical Care e-Community. Simply log in to ecommunity.chestnet.org and find the Critical Care group. If you’re not part of the Critical Care NetWork, log in to chestnet.org and add the Critical Care NetWork to your profile.
Questions? Contact communityadmin@chestnet.org.
Major finding: In all, 26% of patients were vitamin D deficient on ICU admission.
Data source: A retrospective study of 200 ICU trauma patients.
Disclosures: Dr. Ibrahim and Dr. Guillamondegui reported having no financial disclosures.
No VAERS safety signal with Tdap in pregnancy
No unexpected adverse events were seen with the administration of Tdap vaccine during pregnancy, although vaccine surveillance data show a shift toward later pregnancy administration.
Serious adverse events, defined as death, life threatening, hospitalization, prolonged hospitalization, and permanent disability, occurred in 6 of 132 (5%) Tdap pregnancies before the recommendation for routine Tdap vaccination during pregnancy and in 14 of 90 (16%) Tdap pregnancies after the recommendation.
The proportion of preterm births (2 vs. 5) and stillbirths (2 vs. 4) increased after the recommendation, while spontaneous abortions decreased (22 vs. 4), Dr. Pedro L. Moro reported at the winter meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
There was one major birth defect before and two after ACIP’s October 2011 recommendation for routine Tdap vaccination during pregnancy.
One of the biggest changes observed in the Vaccine Adverse Event Reporting System (VAERS) reports was the number of women receiving Tdap during the third trimester, up from just 4% to 55% now, said Dr. Moro, an epidemiologist with the CDC.
The most common non–pregnancy-related outcome in the postrecommendation cohort (Oct. 11, 2011-Jan. 31, 2014) was injection site reactions in 19, compared with 6 reported in the prerecommendation cohort (January 2005-June 2010).
"Changes in reporting patterns are likely due to the new routine Tdap recommendation, increased awareness, and differences in the trimester of vaccination," he said.
Pediatric infectious disease specialist and vaccine developer Dr. Stanley Plotkin said he was troubled that 45% of women are still receiving Tdap before the third trimester because this fails to derive maximum benefit from the vaccine in terms of transmitting passive antibodies to the infant.
"It seems to me the recommendation should be stronger for a third-trimester vaccination than for early [vaccination]," he said. "Also, the issue of confusion with congenital anomalies if the vaccine is given in the first trimester, could damage the idea of safety of vaccinations during pregnancy."
The issue of high antibody levels at birth is important, but "We shouldn’t assume that 45% of the Tdap doses are being given before the third trimester because that is not what this VAERS can tell us," commented Dr. Anne Schuchat, director of the CDC’s National Center for Immunization and Respiratory Diseases.
During his presentation, Dr. Moro emphasized that VAERS looks for safety signals and generates hypotheses, but has inconsistent data quality and completeness and was not designed to assess whether a vaccine caused an adverse event.
Dr. Moro reported having no financial disclosures.
No unexpected adverse events were seen with the administration of Tdap vaccine during pregnancy, although vaccine surveillance data show a shift toward later pregnancy administration.
Serious adverse events, defined as death, life threatening, hospitalization, prolonged hospitalization, and permanent disability, occurred in 6 of 132 (5%) Tdap pregnancies before the recommendation for routine Tdap vaccination during pregnancy and in 14 of 90 (16%) Tdap pregnancies after the recommendation.
The proportion of preterm births (2 vs. 5) and stillbirths (2 vs. 4) increased after the recommendation, while spontaneous abortions decreased (22 vs. 4), Dr. Pedro L. Moro reported at the winter meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
There was one major birth defect before and two after ACIP’s October 2011 recommendation for routine Tdap vaccination during pregnancy.
One of the biggest changes observed in the Vaccine Adverse Event Reporting System (VAERS) reports was the number of women receiving Tdap during the third trimester, up from just 4% to 55% now, said Dr. Moro, an epidemiologist with the CDC.
The most common non–pregnancy-related outcome in the postrecommendation cohort (Oct. 11, 2011-Jan. 31, 2014) was injection site reactions in 19, compared with 6 reported in the prerecommendation cohort (January 2005-June 2010).
"Changes in reporting patterns are likely due to the new routine Tdap recommendation, increased awareness, and differences in the trimester of vaccination," he said.
Pediatric infectious disease specialist and vaccine developer Dr. Stanley Plotkin said he was troubled that 45% of women are still receiving Tdap before the third trimester because this fails to derive maximum benefit from the vaccine in terms of transmitting passive antibodies to the infant.
"It seems to me the recommendation should be stronger for a third-trimester vaccination than for early [vaccination]," he said. "Also, the issue of confusion with congenital anomalies if the vaccine is given in the first trimester, could damage the idea of safety of vaccinations during pregnancy."
The issue of high antibody levels at birth is important, but "We shouldn’t assume that 45% of the Tdap doses are being given before the third trimester because that is not what this VAERS can tell us," commented Dr. Anne Schuchat, director of the CDC’s National Center for Immunization and Respiratory Diseases.
During his presentation, Dr. Moro emphasized that VAERS looks for safety signals and generates hypotheses, but has inconsistent data quality and completeness and was not designed to assess whether a vaccine caused an adverse event.
Dr. Moro reported having no financial disclosures.
No unexpected adverse events were seen with the administration of Tdap vaccine during pregnancy, although vaccine surveillance data show a shift toward later pregnancy administration.
Serious adverse events, defined as death, life threatening, hospitalization, prolonged hospitalization, and permanent disability, occurred in 6 of 132 (5%) Tdap pregnancies before the recommendation for routine Tdap vaccination during pregnancy and in 14 of 90 (16%) Tdap pregnancies after the recommendation.
The proportion of preterm births (2 vs. 5) and stillbirths (2 vs. 4) increased after the recommendation, while spontaneous abortions decreased (22 vs. 4), Dr. Pedro L. Moro reported at the winter meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
There was one major birth defect before and two after ACIP’s October 2011 recommendation for routine Tdap vaccination during pregnancy.
One of the biggest changes observed in the Vaccine Adverse Event Reporting System (VAERS) reports was the number of women receiving Tdap during the third trimester, up from just 4% to 55% now, said Dr. Moro, an epidemiologist with the CDC.
The most common non–pregnancy-related outcome in the postrecommendation cohort (Oct. 11, 2011-Jan. 31, 2014) was injection site reactions in 19, compared with 6 reported in the prerecommendation cohort (January 2005-June 2010).
"Changes in reporting patterns are likely due to the new routine Tdap recommendation, increased awareness, and differences in the trimester of vaccination," he said.
Pediatric infectious disease specialist and vaccine developer Dr. Stanley Plotkin said he was troubled that 45% of women are still receiving Tdap before the third trimester because this fails to derive maximum benefit from the vaccine in terms of transmitting passive antibodies to the infant.
"It seems to me the recommendation should be stronger for a third-trimester vaccination than for early [vaccination]," he said. "Also, the issue of confusion with congenital anomalies if the vaccine is given in the first trimester, could damage the idea of safety of vaccinations during pregnancy."
The issue of high antibody levels at birth is important, but "We shouldn’t assume that 45% of the Tdap doses are being given before the third trimester because that is not what this VAERS can tell us," commented Dr. Anne Schuchat, director of the CDC’s National Center for Immunization and Respiratory Diseases.
During his presentation, Dr. Moro emphasized that VAERS looks for safety signals and generates hypotheses, but has inconsistent data quality and completeness and was not designed to assess whether a vaccine caused an adverse event.
Dr. Moro reported having no financial disclosures.
AT AN ACIP MEETING
CDC sounds alarm on hospital antibiotic use
A scathing new report by the Centers for Disease Control and Prevention found ample room for improvement in inpatient antibiotic prescribing.
Findings include continued overuse of antibiotics in hospitals, errors in prescribing, and the lifesaving potential of efforts to reduce antibiotic use:
• Physicians in some hospitals prescribed three times as many antibiotics as doctors in other hospitals, even though patients were being cared for in similar areas of each hospital.
• Antibiotic prescriptions contained an error in 37% of cases involving treatment for urinary tract infections or use of the common and critical drug, vancomycin (Vancocin).
• Models predicted that a 30% decrease in the use of broad-spectrum antibiotics would lead to a 26% reduction in Clostridium difficile infections, which kill roughly 14,000 hospitalized patients each year.
"Antibiotics are often lifesaving, and we have to protect them before our medicine chests run empty," CDC director Tom Frieden said during a press conference highlighting the report, released in the CDC’s March 4 Morbidity and Mortality Weekly Report (MMWR 2014 March 4;63:1-7).
Dr. Frieden announced that the CDC’s fiscal 2015 budget, part of President Obama’s budget initiative rolled out today, contains a $30 million increase in funds to establish a robust infrastructure in the United States to detect antibiotic threats and protect patients and communities.
The new monies would allow the CDC to extend the "detect and protect" strategy to combat antibiotic resistance outlined last year, help support state and hospital efforts to implement antibiotic stewardship programs, and improve rapid detection of antimicrobial threats and outbreaks.
"One of the things that makes us so focused on antimicrobial resistance is that not only is it a really serious problem, but [also] it’s not too late," Dr. Frieden said.
If funded, he anticipates the CDC and other stakeholders will be able to reverse drug resistance and cut in half the rate of C. difficile and the "nightmare" carbapenem-resistant Enterobacteriaceae infections.
It was noted that robust efforts to improve the use of antibiotics associated with C. difficile in the United Kingdom have resulted in more than a 50% reduction in use of those targeted agents and a roughly 70% reduction in C. difficile infections over the past 6 to 7 years.
The CDC is strongly recommending that every hospital in the United States have an antibiotic stewardship program and is providing a new checklist to help facilities with the task. The checklist contains seven core elements of an effective program: leadership commitment; accountability for outcomes under a single leader; drug expertise under a single pharmacist leader; taking action on at least one prescribing improvement practice; tracking antibiotic prescribing and resistance patterns; reporting regularly to staff about these patterns; and educating staff on antibiotic resistance and improving prescribing practices.
Specific advice was also given to clinicians to order recommended cultures before antibiotics are given and to start drugs promptly; make sure the indication, dose, and expected duration are specified in the patient record; and reassess patients within 48 hours and adjust treatment, if necessary, or stop treatment, if indicated.
Concerns were raised during the briefing over whether voluntary strategies will curb interfacility transmission caused by transfers of patients with multidrug-resistant infections and the failure to report outbreaks between facilities. Dr. John R. Combes, the American Hospital Association’s senior vice president said several groups are working to smooth out these transfers and that the AHA’s "Hospitals in Pursuit of Excellence" program provides best practices to facilitate transfers and foster cooperation with surrounding facilities to prevent infections.
The new CDC report is based on a review of data from all 323 hospitals in the MarketScan Hospital Drug Database and from hospitals in the CDC’s Emerging Infections Program.
Antibiotics were prescribed for 55.7% of patients hospitalized in 2010 in the MarketScan Hospital Drug Database, with 30% receiving at least one dose of broad-spectrum antibiotics.
One or more antibiotics were used to treat active infections in 37% of 11,282 patients treated in 2011 at 183 acute care hospitals evaluated by the Emerging Infections Program. Half of the antibiotics were prescribed for one of three scenarios: lower respiratory tract infections (22.2%), urinary tract infections (14%), and suspected drug-resistant Gram-positive infections such as methicillin-resistant Staphylococcus aureus (17.6%).
The CDC previously called on physicians to address antibiotic resistance in its Antibiotic Threats in the United States, 2013 report and the 2013 Get Smart About Antibiotics Week. The issue also will be tackled in the CDC’s forthcoming Transatlantic Taskforce on Antimicrobial Resistance 2013 report, with additional research expected to focus on contributing factors that led to such wide variances in antibiotic use between hospitals.
Dr. Frieden and Dr. Combes reported having no financial disclosures.

|
| Dr. Franklin A. Michota |
Dr. Franklin A. Michota is director of academic affairs, department of hospital medicine, Cleveland Clinic. He reports having no disclosures.

|
| Dr. Franklin A. Michota |
Dr. Franklin A. Michota is director of academic affairs, department of hospital medicine, Cleveland Clinic. He reports having no disclosures.

|
| Dr. Franklin A. Michota |
Dr. Franklin A. Michota is director of academic affairs, department of hospital medicine, Cleveland Clinic. He reports having no disclosures.
A scathing new report by the Centers for Disease Control and Prevention found ample room for improvement in inpatient antibiotic prescribing.
Findings include continued overuse of antibiotics in hospitals, errors in prescribing, and the lifesaving potential of efforts to reduce antibiotic use:
• Physicians in some hospitals prescribed three times as many antibiotics as doctors in other hospitals, even though patients were being cared for in similar areas of each hospital.
• Antibiotic prescriptions contained an error in 37% of cases involving treatment for urinary tract infections or use of the common and critical drug, vancomycin (Vancocin).
• Models predicted that a 30% decrease in the use of broad-spectrum antibiotics would lead to a 26% reduction in Clostridium difficile infections, which kill roughly 14,000 hospitalized patients each year.
"Antibiotics are often lifesaving, and we have to protect them before our medicine chests run empty," CDC director Tom Frieden said during a press conference highlighting the report, released in the CDC’s March 4 Morbidity and Mortality Weekly Report (MMWR 2014 March 4;63:1-7).
Dr. Frieden announced that the CDC’s fiscal 2015 budget, part of President Obama’s budget initiative rolled out today, contains a $30 million increase in funds to establish a robust infrastructure in the United States to detect antibiotic threats and protect patients and communities.
The new monies would allow the CDC to extend the "detect and protect" strategy to combat antibiotic resistance outlined last year, help support state and hospital efforts to implement antibiotic stewardship programs, and improve rapid detection of antimicrobial threats and outbreaks.
"One of the things that makes us so focused on antimicrobial resistance is that not only is it a really serious problem, but [also] it’s not too late," Dr. Frieden said.
If funded, he anticipates the CDC and other stakeholders will be able to reverse drug resistance and cut in half the rate of C. difficile and the "nightmare" carbapenem-resistant Enterobacteriaceae infections.
It was noted that robust efforts to improve the use of antibiotics associated with C. difficile in the United Kingdom have resulted in more than a 50% reduction in use of those targeted agents and a roughly 70% reduction in C. difficile infections over the past 6 to 7 years.
The CDC is strongly recommending that every hospital in the United States have an antibiotic stewardship program and is providing a new checklist to help facilities with the task. The checklist contains seven core elements of an effective program: leadership commitment; accountability for outcomes under a single leader; drug expertise under a single pharmacist leader; taking action on at least one prescribing improvement practice; tracking antibiotic prescribing and resistance patterns; reporting regularly to staff about these patterns; and educating staff on antibiotic resistance and improving prescribing practices.
Specific advice was also given to clinicians to order recommended cultures before antibiotics are given and to start drugs promptly; make sure the indication, dose, and expected duration are specified in the patient record; and reassess patients within 48 hours and adjust treatment, if necessary, or stop treatment, if indicated.
Concerns were raised during the briefing over whether voluntary strategies will curb interfacility transmission caused by transfers of patients with multidrug-resistant infections and the failure to report outbreaks between facilities. Dr. John R. Combes, the American Hospital Association’s senior vice president said several groups are working to smooth out these transfers and that the AHA’s "Hospitals in Pursuit of Excellence" program provides best practices to facilitate transfers and foster cooperation with surrounding facilities to prevent infections.
The new CDC report is based on a review of data from all 323 hospitals in the MarketScan Hospital Drug Database and from hospitals in the CDC’s Emerging Infections Program.
Antibiotics were prescribed for 55.7% of patients hospitalized in 2010 in the MarketScan Hospital Drug Database, with 30% receiving at least one dose of broad-spectrum antibiotics.
One or more antibiotics were used to treat active infections in 37% of 11,282 patients treated in 2011 at 183 acute care hospitals evaluated by the Emerging Infections Program. Half of the antibiotics were prescribed for one of three scenarios: lower respiratory tract infections (22.2%), urinary tract infections (14%), and suspected drug-resistant Gram-positive infections such as methicillin-resistant Staphylococcus aureus (17.6%).
The CDC previously called on physicians to address antibiotic resistance in its Antibiotic Threats in the United States, 2013 report and the 2013 Get Smart About Antibiotics Week. The issue also will be tackled in the CDC’s forthcoming Transatlantic Taskforce on Antimicrobial Resistance 2013 report, with additional research expected to focus on contributing factors that led to such wide variances in antibiotic use between hospitals.
Dr. Frieden and Dr. Combes reported having no financial disclosures.
A scathing new report by the Centers for Disease Control and Prevention found ample room for improvement in inpatient antibiotic prescribing.
Findings include continued overuse of antibiotics in hospitals, errors in prescribing, and the lifesaving potential of efforts to reduce antibiotic use:
• Physicians in some hospitals prescribed three times as many antibiotics as doctors in other hospitals, even though patients were being cared for in similar areas of each hospital.
• Antibiotic prescriptions contained an error in 37% of cases involving treatment for urinary tract infections or use of the common and critical drug, vancomycin (Vancocin).
• Models predicted that a 30% decrease in the use of broad-spectrum antibiotics would lead to a 26% reduction in Clostridium difficile infections, which kill roughly 14,000 hospitalized patients each year.
"Antibiotics are often lifesaving, and we have to protect them before our medicine chests run empty," CDC director Tom Frieden said during a press conference highlighting the report, released in the CDC’s March 4 Morbidity and Mortality Weekly Report (MMWR 2014 March 4;63:1-7).
Dr. Frieden announced that the CDC’s fiscal 2015 budget, part of President Obama’s budget initiative rolled out today, contains a $30 million increase in funds to establish a robust infrastructure in the United States to detect antibiotic threats and protect patients and communities.
The new monies would allow the CDC to extend the "detect and protect" strategy to combat antibiotic resistance outlined last year, help support state and hospital efforts to implement antibiotic stewardship programs, and improve rapid detection of antimicrobial threats and outbreaks.
"One of the things that makes us so focused on antimicrobial resistance is that not only is it a really serious problem, but [also] it’s not too late," Dr. Frieden said.
If funded, he anticipates the CDC and other stakeholders will be able to reverse drug resistance and cut in half the rate of C. difficile and the "nightmare" carbapenem-resistant Enterobacteriaceae infections.
It was noted that robust efforts to improve the use of antibiotics associated with C. difficile in the United Kingdom have resulted in more than a 50% reduction in use of those targeted agents and a roughly 70% reduction in C. difficile infections over the past 6 to 7 years.
The CDC is strongly recommending that every hospital in the United States have an antibiotic stewardship program and is providing a new checklist to help facilities with the task. The checklist contains seven core elements of an effective program: leadership commitment; accountability for outcomes under a single leader; drug expertise under a single pharmacist leader; taking action on at least one prescribing improvement practice; tracking antibiotic prescribing and resistance patterns; reporting regularly to staff about these patterns; and educating staff on antibiotic resistance and improving prescribing practices.
Specific advice was also given to clinicians to order recommended cultures before antibiotics are given and to start drugs promptly; make sure the indication, dose, and expected duration are specified in the patient record; and reassess patients within 48 hours and adjust treatment, if necessary, or stop treatment, if indicated.
Concerns were raised during the briefing over whether voluntary strategies will curb interfacility transmission caused by transfers of patients with multidrug-resistant infections and the failure to report outbreaks between facilities. Dr. John R. Combes, the American Hospital Association’s senior vice president said several groups are working to smooth out these transfers and that the AHA’s "Hospitals in Pursuit of Excellence" program provides best practices to facilitate transfers and foster cooperation with surrounding facilities to prevent infections.
The new CDC report is based on a review of data from all 323 hospitals in the MarketScan Hospital Drug Database and from hospitals in the CDC’s Emerging Infections Program.
Antibiotics were prescribed for 55.7% of patients hospitalized in 2010 in the MarketScan Hospital Drug Database, with 30% receiving at least one dose of broad-spectrum antibiotics.
One or more antibiotics were used to treat active infections in 37% of 11,282 patients treated in 2011 at 183 acute care hospitals evaluated by the Emerging Infections Program. Half of the antibiotics were prescribed for one of three scenarios: lower respiratory tract infections (22.2%), urinary tract infections (14%), and suspected drug-resistant Gram-positive infections such as methicillin-resistant Staphylococcus aureus (17.6%).
The CDC previously called on physicians to address antibiotic resistance in its Antibiotic Threats in the United States, 2013 report and the 2013 Get Smart About Antibiotics Week. The issue also will be tackled in the CDC’s forthcoming Transatlantic Taskforce on Antimicrobial Resistance 2013 report, with additional research expected to focus on contributing factors that led to such wide variances in antibiotic use between hospitals.
Dr. Frieden and Dr. Combes reported having no financial disclosures.
FROM MORBIDITY AND MORTALITY WEEKLY REPORT
Tdap vaccination during pregnancy not linked to adverse birth outcomes
Receipt of the Tdap vaccine during pregnancy was not associated with increased risks for adverse birth outcomes in a safety study, although there was a weak association with chorioamnionitis.
In a separate study, tetanus, diphtheria, and pertussis coverage during pregnancy increased eightfold among women in five states in 2012, while dipping slightly in California.
The safety cohort included 26,224 women who received Tdap in pregnancy and 97,265 unvaccinated women with live births during 2010-2012 at two California sites in the Vaccine Safety Datalink (VSD) program.
Inpatient chorioamnionitis was diagnosed in 6.1% of vaccinated women and 5.5% of unvaccinated women (adjusted relative risk, 1.19; 95% confidence interval, 1.13-1.27), Dr. Elyse Olshen Kharbanda reported at the winter meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP).
In contrast, rates of preterm birth (less than 37 weeks’ gestation) were lower with receipt of Tdap (6.3% vs. 7.8%; hazard ratio, 0.88; 95% CI, 0.83-0.93), while rates were similar for small for gestational age births (8.4% vs. 8.3%; RR, 1.00; 95% CI, 0.96-1.06).
A new diagnosis of hypertensive disorders, examined only in those vaccinated before 20 weeks’ gestation, occurred in 8.2% of vaccinated and 8.0% of unvaccinated women (RR, 1.00; 95% CI, 0.99-1.20), she said.
Secondary analyses restricted to women vaccinated between 27 and 36 weeks’ gestation, consistent with the preferential timing for Tdap vaccination, appeared to confirm the results. Chorioamnionitis occurred in 5.6% of vaccinated and 5.5% of unvaccinated women (RR, 1.11; 95% CI, 1.03-1.21), preterm birth in 5.3% vs. 7.8% (HR, 0.83; 95% CI, 0.77-0.90), and small for gestational age births in 8.6% vs. 8.3% (RR, 1.03; 95% CI, 0.96-1.10).
"The chorioamnionitis findings merit further discussion and possibly further investigation," said Dr. Kharbanda of the HealthPartners Institute for Education and Research, Minneapolis.
The findings in the current study persisted after researchers adjusted for risk factors such as maternal age and comorbidities, although data were not available for many of the most important chorioamnionitis risk factors, she said. Dr. Kharbanda went on to say that the magnitude of the potential risk detected was small, the risk was largely attributable to differences in chorioamnionitis rates seen only for the year 2012, and that there was no associated risk for preterm birth.
Several ACIP representatives questioned the choice of chorioamnionitis as a safety outcome, including American College of Obstetricians and Gynecologists representative Dr. Laura Riley of Harvard Medical School, Boston, who said, "It’s probably one of the messiest diagnoses there is, and it’s really hard to make any sense out of what it really means."
Carol Hayes, MPH, a certified nurse-midwife and a representative from the American Nurses Association, suggested the data be stratified by gestational age at birth because chorioamnionitis is a link for preterm birth, adding "What made you pick chorioamnionitis? It’s such a bizarre thing to look at with a vaccine."
Dr. Kharbanda said chorioamnionitis previously had been studied in relation to trivalent inactivated influenza vaccine, where the risk was slightly increased among pregnant women (Obstet. Gynecol. 2013;1211:519-25). She acknowledged that their obstetrical consultant had also "brought up that in maternal-fetal medicine, often chorioamnionitis is on the pathway for preterm delivery, but not considered an outcome in itself. I think we were sort of trying to look at common diagnoses that occur in women, and it is a common diagnosis."
Tdap coverage
Tdap coverage estimates were based on pregnancies ending between Jan. 1, 2007, and Nov. 15, 2012 at seven sites in the VSD, a collaboration between the CDC and nine managed-care organizations. Among 371,539 live births from 2007 to 2012, overall, 9.5% of women received Tdap during pregnancy and 14.4% post partum. In 2012, the corresponding rates were 13.7% and 8.8%.
In Colorado, Minnesota, Oregon, Washington, and Wisconsin, 16% of women were vaccinated with Tdap during pregnancy in 2012, compared with 2.2% in 2011, Dr. Kharbanda said. Prepregnancy Tdap vaccination rates were 46% and 56.2%, respectively
The increase in Tdap coverage during pregnancy is likely in response to ACIP’s June 2011 recommendation that health care providers administer Tdap to pregnant women who had not previously been vaccinated, preferably after 20 weeks’ gestation, she said.
ACIP updated its guidelines again in October 2012 because of persistent increases in whooping cough in the United States, this time calling for Tdap administration during each pregnancy, irrespective of prior vaccination history, and preferably between 27 and 36 weeks’ gestation.
In California, Tdap coverage during pregnancy increased substantially in 2010 and 2011, but decreased in 2012 (16% vs. 30% vs. 19.5%), likely because many women had received Tdap prepregnancy, she said. Prepregnancy vaccinations climbed steadily from 21.5% in 2010 and 35.3% in 2011 to 53.8% in 2012.
In 2010, the California Department of Public Health began recommending that all women of childbearing age be vaccinated with Tdap.
A protocol looking at repeat Tdap vaccinations and repeat pregnancies has already been written and is being led by CDC investigators, Dr. Kharbanda said.
Dr. Kharbanda reported study support from the CDC VSD project.
Receipt of the Tdap vaccine during pregnancy was not associated with increased risks for adverse birth outcomes in a safety study, although there was a weak association with chorioamnionitis.
In a separate study, tetanus, diphtheria, and pertussis coverage during pregnancy increased eightfold among women in five states in 2012, while dipping slightly in California.
The safety cohort included 26,224 women who received Tdap in pregnancy and 97,265 unvaccinated women with live births during 2010-2012 at two California sites in the Vaccine Safety Datalink (VSD) program.
Inpatient chorioamnionitis was diagnosed in 6.1% of vaccinated women and 5.5% of unvaccinated women (adjusted relative risk, 1.19; 95% confidence interval, 1.13-1.27), Dr. Elyse Olshen Kharbanda reported at the winter meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP).
In contrast, rates of preterm birth (less than 37 weeks’ gestation) were lower with receipt of Tdap (6.3% vs. 7.8%; hazard ratio, 0.88; 95% CI, 0.83-0.93), while rates were similar for small for gestational age births (8.4% vs. 8.3%; RR, 1.00; 95% CI, 0.96-1.06).
A new diagnosis of hypertensive disorders, examined only in those vaccinated before 20 weeks’ gestation, occurred in 8.2% of vaccinated and 8.0% of unvaccinated women (RR, 1.00; 95% CI, 0.99-1.20), she said.
Secondary analyses restricted to women vaccinated between 27 and 36 weeks’ gestation, consistent with the preferential timing for Tdap vaccination, appeared to confirm the results. Chorioamnionitis occurred in 5.6% of vaccinated and 5.5% of unvaccinated women (RR, 1.11; 95% CI, 1.03-1.21), preterm birth in 5.3% vs. 7.8% (HR, 0.83; 95% CI, 0.77-0.90), and small for gestational age births in 8.6% vs. 8.3% (RR, 1.03; 95% CI, 0.96-1.10).
"The chorioamnionitis findings merit further discussion and possibly further investigation," said Dr. Kharbanda of the HealthPartners Institute for Education and Research, Minneapolis.
The findings in the current study persisted after researchers adjusted for risk factors such as maternal age and comorbidities, although data were not available for many of the most important chorioamnionitis risk factors, she said. Dr. Kharbanda went on to say that the magnitude of the potential risk detected was small, the risk was largely attributable to differences in chorioamnionitis rates seen only for the year 2012, and that there was no associated risk for preterm birth.
Several ACIP representatives questioned the choice of chorioamnionitis as a safety outcome, including American College of Obstetricians and Gynecologists representative Dr. Laura Riley of Harvard Medical School, Boston, who said, "It’s probably one of the messiest diagnoses there is, and it’s really hard to make any sense out of what it really means."
Carol Hayes, MPH, a certified nurse-midwife and a representative from the American Nurses Association, suggested the data be stratified by gestational age at birth because chorioamnionitis is a link for preterm birth, adding "What made you pick chorioamnionitis? It’s such a bizarre thing to look at with a vaccine."
Dr. Kharbanda said chorioamnionitis previously had been studied in relation to trivalent inactivated influenza vaccine, where the risk was slightly increased among pregnant women (Obstet. Gynecol. 2013;1211:519-25). She acknowledged that their obstetrical consultant had also "brought up that in maternal-fetal medicine, often chorioamnionitis is on the pathway for preterm delivery, but not considered an outcome in itself. I think we were sort of trying to look at common diagnoses that occur in women, and it is a common diagnosis."
Tdap coverage
Tdap coverage estimates were based on pregnancies ending between Jan. 1, 2007, and Nov. 15, 2012 at seven sites in the VSD, a collaboration between the CDC and nine managed-care organizations. Among 371,539 live births from 2007 to 2012, overall, 9.5% of women received Tdap during pregnancy and 14.4% post partum. In 2012, the corresponding rates were 13.7% and 8.8%.
In Colorado, Minnesota, Oregon, Washington, and Wisconsin, 16% of women were vaccinated with Tdap during pregnancy in 2012, compared with 2.2% in 2011, Dr. Kharbanda said. Prepregnancy Tdap vaccination rates were 46% and 56.2%, respectively
The increase in Tdap coverage during pregnancy is likely in response to ACIP’s June 2011 recommendation that health care providers administer Tdap to pregnant women who had not previously been vaccinated, preferably after 20 weeks’ gestation, she said.
ACIP updated its guidelines again in October 2012 because of persistent increases in whooping cough in the United States, this time calling for Tdap administration during each pregnancy, irrespective of prior vaccination history, and preferably between 27 and 36 weeks’ gestation.
In California, Tdap coverage during pregnancy increased substantially in 2010 and 2011, but decreased in 2012 (16% vs. 30% vs. 19.5%), likely because many women had received Tdap prepregnancy, she said. Prepregnancy vaccinations climbed steadily from 21.5% in 2010 and 35.3% in 2011 to 53.8% in 2012.
In 2010, the California Department of Public Health began recommending that all women of childbearing age be vaccinated with Tdap.
A protocol looking at repeat Tdap vaccinations and repeat pregnancies has already been written and is being led by CDC investigators, Dr. Kharbanda said.
Dr. Kharbanda reported study support from the CDC VSD project.
Receipt of the Tdap vaccine during pregnancy was not associated with increased risks for adverse birth outcomes in a safety study, although there was a weak association with chorioamnionitis.
In a separate study, tetanus, diphtheria, and pertussis coverage during pregnancy increased eightfold among women in five states in 2012, while dipping slightly in California.
The safety cohort included 26,224 women who received Tdap in pregnancy and 97,265 unvaccinated women with live births during 2010-2012 at two California sites in the Vaccine Safety Datalink (VSD) program.
Inpatient chorioamnionitis was diagnosed in 6.1% of vaccinated women and 5.5% of unvaccinated women (adjusted relative risk, 1.19; 95% confidence interval, 1.13-1.27), Dr. Elyse Olshen Kharbanda reported at the winter meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP).
In contrast, rates of preterm birth (less than 37 weeks’ gestation) were lower with receipt of Tdap (6.3% vs. 7.8%; hazard ratio, 0.88; 95% CI, 0.83-0.93), while rates were similar for small for gestational age births (8.4% vs. 8.3%; RR, 1.00; 95% CI, 0.96-1.06).
A new diagnosis of hypertensive disorders, examined only in those vaccinated before 20 weeks’ gestation, occurred in 8.2% of vaccinated and 8.0% of unvaccinated women (RR, 1.00; 95% CI, 0.99-1.20), she said.
Secondary analyses restricted to women vaccinated between 27 and 36 weeks’ gestation, consistent with the preferential timing for Tdap vaccination, appeared to confirm the results. Chorioamnionitis occurred in 5.6% of vaccinated and 5.5% of unvaccinated women (RR, 1.11; 95% CI, 1.03-1.21), preterm birth in 5.3% vs. 7.8% (HR, 0.83; 95% CI, 0.77-0.90), and small for gestational age births in 8.6% vs. 8.3% (RR, 1.03; 95% CI, 0.96-1.10).
"The chorioamnionitis findings merit further discussion and possibly further investigation," said Dr. Kharbanda of the HealthPartners Institute for Education and Research, Minneapolis.
The findings in the current study persisted after researchers adjusted for risk factors such as maternal age and comorbidities, although data were not available for many of the most important chorioamnionitis risk factors, she said. Dr. Kharbanda went on to say that the magnitude of the potential risk detected was small, the risk was largely attributable to differences in chorioamnionitis rates seen only for the year 2012, and that there was no associated risk for preterm birth.
Several ACIP representatives questioned the choice of chorioamnionitis as a safety outcome, including American College of Obstetricians and Gynecologists representative Dr. Laura Riley of Harvard Medical School, Boston, who said, "It’s probably one of the messiest diagnoses there is, and it’s really hard to make any sense out of what it really means."
Carol Hayes, MPH, a certified nurse-midwife and a representative from the American Nurses Association, suggested the data be stratified by gestational age at birth because chorioamnionitis is a link for preterm birth, adding "What made you pick chorioamnionitis? It’s such a bizarre thing to look at with a vaccine."
Dr. Kharbanda said chorioamnionitis previously had been studied in relation to trivalent inactivated influenza vaccine, where the risk was slightly increased among pregnant women (Obstet. Gynecol. 2013;1211:519-25). She acknowledged that their obstetrical consultant had also "brought up that in maternal-fetal medicine, often chorioamnionitis is on the pathway for preterm delivery, but not considered an outcome in itself. I think we were sort of trying to look at common diagnoses that occur in women, and it is a common diagnosis."
Tdap coverage
Tdap coverage estimates were based on pregnancies ending between Jan. 1, 2007, and Nov. 15, 2012 at seven sites in the VSD, a collaboration between the CDC and nine managed-care organizations. Among 371,539 live births from 2007 to 2012, overall, 9.5% of women received Tdap during pregnancy and 14.4% post partum. In 2012, the corresponding rates were 13.7% and 8.8%.
In Colorado, Minnesota, Oregon, Washington, and Wisconsin, 16% of women were vaccinated with Tdap during pregnancy in 2012, compared with 2.2% in 2011, Dr. Kharbanda said. Prepregnancy Tdap vaccination rates were 46% and 56.2%, respectively
The increase in Tdap coverage during pregnancy is likely in response to ACIP’s June 2011 recommendation that health care providers administer Tdap to pregnant women who had not previously been vaccinated, preferably after 20 weeks’ gestation, she said.
ACIP updated its guidelines again in October 2012 because of persistent increases in whooping cough in the United States, this time calling for Tdap administration during each pregnancy, irrespective of prior vaccination history, and preferably between 27 and 36 weeks’ gestation.
In California, Tdap coverage during pregnancy increased substantially in 2010 and 2011, but decreased in 2012 (16% vs. 30% vs. 19.5%), likely because many women had received Tdap prepregnancy, she said. Prepregnancy vaccinations climbed steadily from 21.5% in 2010 and 35.3% in 2011 to 53.8% in 2012.
In 2010, the California Department of Public Health began recommending that all women of childbearing age be vaccinated with Tdap.
A protocol looking at repeat Tdap vaccinations and repeat pregnancies has already been written and is being led by CDC investigators, Dr. Kharbanda said.
Dr. Kharbanda reported study support from the CDC VSD project.
FROM AN ACIP MEETING
Major finding: Inpatient chorioamnionitis was diagnosed in 6.1% of vaccinated women and 5.5% of unvaccinated women (adjusted RR, 1.19; 95% CI, 1.13-1.27).
Data source: CDC Vaccine Safety Datalink.
Disclosures: Dr. Kharbanda reported study support from the CDC VSD project.
New takeaways from the Boston Marathon bombings
NAPLES, FLA. – Without a single life lost at area hospitals, the medical response to the Boston Marathon bombings was by all accounts remarkable. But what if police and other first-responders had tourniquets and were trained in their use?
As it was, advanced topical hemostatic agents and tourniquets were MIA in the field, forcing all but one prehospital tourniquet to be improvised, according to Massachusetts General Hospital trauma surgeon Lt. Col. David King, U. S. Army Reserve.
"It’s abundantly clear from the literature that improvised tourniquets almost never work," he said. "The fact that every soldier down range has one in his pocket and we couldn’t muster up more than one in the city of Boston is a shame and we need to rectify that."
In all, there were 66 limb injuries, with 29 patients having recognized limb exsanguination at the scene. Of the 27 tourniquets applied, 26 were improvised.
Failure to translate this important lesson from military experience meant that these homemade devices were most likely wildly ineffective and, in some cases, may have even exacerbated blood loss, he said. Surprisingly, there’s no formal protocol for tourniquet application or training paradigm in Massachusetts, such that exists in the Army’s prehospital Tactical Combat Casualty Care handbook.
"If you look across the paramedic medical guidelines, there is no protocol on the right, evidence-based way to put on a tourniquet or what the indications are for a tourniquet," Dr. King said at the annual scientific assembly of the Eastern Association for the Surgery of Trauma (EAST). "We don’t have it, and we need to" have it nationwide.
EAST is hoping to provide some guidance in this area with the development of practice management guidelines (PMG) for tourniquet use in extremity trauma. Dr. King, who heads this PMG committee, reported at the meeting that the guidelines, due out later this year, will emphasize that commercially made tourniquets should be available in the prehospital setting, improvised tourniquets rarely work and cannot be recommended; essentially all commercially made tourniquets are equieffective; and that nonmedical personnel can be trained to correctly apply a proper tourniquet.
The need for bleeding control equipment and training for the public has not gone unnoticed by other stakeholders and is the central focus of the newest statement from the Hartford Consensus, a collaborative group of trauma surgeons, law enforcement officers, and emergency responders led by the American College of Surgeons (J. Am. Coll. Surg. 2014;218:467-75).
In dissecting the medical response to the Boston bombings, much has been made of the fact that Boston is home to six Level 1 trauma centers, and that it was race day, a city holiday, 3 p.m., and low-yield devices were used. Dr. King, however, gave high praise to an unsung hero, the Boston EMS loading officer who managed most of the transport destination decisions and smartly distributed critical cases throughout the area’s hospitals.
All critical patients were evacuated within 1 hour, but some have argued this could have been accomplished more quickly were it not for a "stay-and-play" mentality in the medical tent. The after-action review suggests there probably were enough ambulances to do the job in 30 minutes, said Dr. King, who remarked that the only thing that matters in critical trauma cases is time to surgical hemorrhage control.
"This will remain a controversial issue for Boston, whether staying and playing in the medical tent was a good idea," he said. "I don’t know what the right answer is."
Credit was also given to Massachusetts General, and many of the other city hospitals, for their insistence on regular full-scale drills that allowed quick lock-down of the facility to control access and manage the surge capacity in the emergency department.
"It’s more than just a tabletop exercise," Dr. King said. "When we test the surge capacity, we actually empty the emergency room for real. Usually, it occurs between 5:00 and 7:00 or 4:30 and 7:30 in the morning, but actually doing these things was useful, because when it came time to do it for real, no one was tripping over their own feet."
ED volume on that April afternoon was reduced from 97 patients to just 39 within 1.5 hours. Nontrauma physicians, including psychiatrists, did their part by writing "barebones orders" to get patients transferred to the wards.
Staff at Massachusetts General treated 43 patients, performing nine emergent operations, six amputations, two laparotomies, and one thoracotomy, with two traumatic arrests within the first 72 hours. As with other Boston hospitals, there were no in-hospital deaths, he said.
Patients arrived in such rapid succession, however, that it slowed the electronic medical record system and forced staff to use sequential record numbers, which can be dangerous. Registration couldn’t keep up and orders couldn’t be put in fast enough with 43 patients arriving in roughly 35 minutes, Dr. King explained. Workarounds were often old-school, and reminiscent of his days as a green trauma surgeon serving at Ibn Sina Hospital, Baghdad, Iraq.
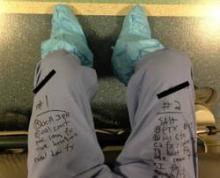
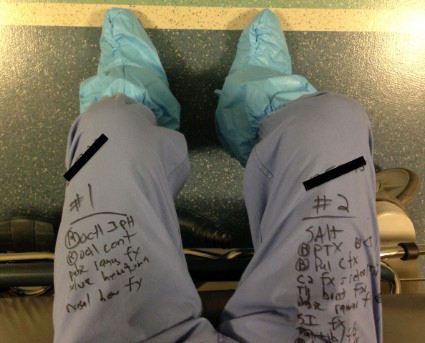
"In the midst of this entire event, I turned to my nurse practitioner and asked for x, y, z information on a particular patient," he said. "She didn’t turn to her tablet or desktop computer in the operating room. She pulled out a piece of paper where she’d written all the relevant information. At the end of the event, I looked down and realized I’d done the same thing. I was keeping track of my patients on my pants with a Sharpie."
King describes this as a failure of technology and translation from the battlefield, but noted that the hospital did use the military’s practice of reviewing a critical event. Once they’d finished in the operating room and achieved hemorrhage and contamination control, the entire trauma team was reassembled. They did detailed tertiary trauma surveys and went back through every single patient to determine what additional studies they needed, what injuries had been missed.
"It was remarkable the number of things that were missed," Dr. King said. "No one was bleeding to death, but tons and tons of [small, non–life threatening] missed injuries [like ruptured ear drums], and to me this was one of the more important events that surrounded our response to the entire bombing – sitting down after the dust had settled and carefully going over every patient’s medical record."
Dr. King reported having no financial disclosures.
Lt. Col. King makes some excellent points. However, despite Dr. King's combat medicine experience, his contribution to this article reveals a gap that still exists between field medicine and hospital medicine.
The case against improvised tourniquets
There is no consensus on the definition of an "improvised" tourniquet. Homemade applications such as neckties and sticks are defined as improvised, but the Boston EMS kits (surgical tubing and Kelly clamps) also could be considered improvised. All of these methods may sound primitive since the advent of Combat-Application Tourniquets; however CATS are relatively new to the scene. Should we consider every tourniquet applied prior to their introduction to have been improvised? For the price of one CAT, we can assemble 10 tourniquet kits. And if Dr. King's initial contention was that tourniquets were in short supply, then it is a better system to deploy as many as possible. This point may be moot, however, because since the Boston bombing incident, federal grant money has allowed the purchase of CAT units in great numbers within the Metro-Boston area.
Tourniquet efficacy
"It is abundantly clear from the literature that improvised tourniquets almost never work." I feel this study had the wrong focus. Rather than ask which tourniquets have a better success rate, the study should have asked whose tourniquets have a better success rate.
Study after study has shown that successful intubation rates have little to do with the level of training of the practitioner, and everything to do with the frequency of intubation. More tubes equal more successful passes. I propose that this applies to tourniquet application as well. Field personnel who do not practice or have the opportunity to apply tourniquets frequently (which is the great majority of us) may not be applying tourniquets properly. This is why we instituted a massive tourniquet training and review program almost immediately following the 2013 bombing at the Boston Marathon.
Another point not addressed in studies is the effectiveness of nonresponder tourniquets. I would contend that an experienced practitioner with a necktie and a stick would be more successful in hemorrhage control that a civilian with a CAT, but those data have not been explored yet either.
The Loading Officer as unsung hero
All our Action Area Officers performed brilliantly (full disclosure, I was the Loading Officer). However the term Loading Officer is a misnomer. Yes, there is a Loading Officer, but there is actually a Loading Team. It consists of staff at the scene who liaise with the treatment areas and the true unsung heroes, the staff in the Operations Division or Dispatch Center. Dispatchers are in constant contact with the hospitals, scene operations, as well as continuing city service. They perform the hospital destination designations because they have a global sense of traffic, while at the scene we have only a worm's-eye view.
The stay-and-play critique
"All critical patients were evacuated within 1 hour, but some argued this could have been accomplished more quickly were it not for a 'stay-and-play' mentality." I take exception to this assertion. First, the point is contradictory. All critical patients were evacuated within 1 hour, but there was a stay-and-play mentality? Every EMT and paramedic knows that transport times translate into positive results with trauma.
Emergency personnel are taught that what matters most in trauma survival is not basic life support, advance life support, or wheelbarrow, but getting them to the ED fast. Yes, there were slowdowns in the medical tents, but most of delays came from non-EMS personnel who were not well versed in field medicine. We questioned, pleaded, and ultimately ordered some practitioners away from patients as they were attempting to perform procedures that we would never attempt on a trauma patient in the field. This is by no means a condemnation on my part. The staff we worked with was exceptional, and I witnessed true skill, heroism, and compassion. But it also demonstrated to me that a disconnect exists between field medicine and hospital medicine.
I see an opportunity here for training to better acclimate responders to working together with our hospital counterparts. We also need hospital personnel to be oriented to realities and limitations of field medicine.
Lt. Brian Pomodoro is a member of the Boston EMS.
Lt. Col. King makes some excellent points. However, despite Dr. King's combat medicine experience, his contribution to this article reveals a gap that still exists between field medicine and hospital medicine.
The case against improvised tourniquets
There is no consensus on the definition of an "improvised" tourniquet. Homemade applications such as neckties and sticks are defined as improvised, but the Boston EMS kits (surgical tubing and Kelly clamps) also could be considered improvised. All of these methods may sound primitive since the advent of Combat-Application Tourniquets; however CATS are relatively new to the scene. Should we consider every tourniquet applied prior to their introduction to have been improvised? For the price of one CAT, we can assemble 10 tourniquet kits. And if Dr. King's initial contention was that tourniquets were in short supply, then it is a better system to deploy as many as possible. This point may be moot, however, because since the Boston bombing incident, federal grant money has allowed the purchase of CAT units in great numbers within the Metro-Boston area.
Tourniquet efficacy
"It is abundantly clear from the literature that improvised tourniquets almost never work." I feel this study had the wrong focus. Rather than ask which tourniquets have a better success rate, the study should have asked whose tourniquets have a better success rate.
Study after study has shown that successful intubation rates have little to do with the level of training of the practitioner, and everything to do with the frequency of intubation. More tubes equal more successful passes. I propose that this applies to tourniquet application as well. Field personnel who do not practice or have the opportunity to apply tourniquets frequently (which is the great majority of us) may not be applying tourniquets properly. This is why we instituted a massive tourniquet training and review program almost immediately following the 2013 bombing at the Boston Marathon.
Another point not addressed in studies is the effectiveness of nonresponder tourniquets. I would contend that an experienced practitioner with a necktie and a stick would be more successful in hemorrhage control that a civilian with a CAT, but those data have not been explored yet either.
The Loading Officer as unsung hero
All our Action Area Officers performed brilliantly (full disclosure, I was the Loading Officer). However the term Loading Officer is a misnomer. Yes, there is a Loading Officer, but there is actually a Loading Team. It consists of staff at the scene who liaise with the treatment areas and the true unsung heroes, the staff in the Operations Division or Dispatch Center. Dispatchers are in constant contact with the hospitals, scene operations, as well as continuing city service. They perform the hospital destination designations because they have a global sense of traffic, while at the scene we have only a worm's-eye view.
The stay-and-play critique
"All critical patients were evacuated within 1 hour, but some argued this could have been accomplished more quickly were it not for a 'stay-and-play' mentality." I take exception to this assertion. First, the point is contradictory. All critical patients were evacuated within 1 hour, but there was a stay-and-play mentality? Every EMT and paramedic knows that transport times translate into positive results with trauma.
Emergency personnel are taught that what matters most in trauma survival is not basic life support, advance life support, or wheelbarrow, but getting them to the ED fast. Yes, there were slowdowns in the medical tents, but most of delays came from non-EMS personnel who were not well versed in field medicine. We questioned, pleaded, and ultimately ordered some practitioners away from patients as they were attempting to perform procedures that we would never attempt on a trauma patient in the field. This is by no means a condemnation on my part. The staff we worked with was exceptional, and I witnessed true skill, heroism, and compassion. But it also demonstrated to me that a disconnect exists between field medicine and hospital medicine.
I see an opportunity here for training to better acclimate responders to working together with our hospital counterparts. We also need hospital personnel to be oriented to realities and limitations of field medicine.
Lt. Brian Pomodoro is a member of the Boston EMS.
Lt. Col. King makes some excellent points. However, despite Dr. King's combat medicine experience, his contribution to this article reveals a gap that still exists between field medicine and hospital medicine.
The case against improvised tourniquets
There is no consensus on the definition of an "improvised" tourniquet. Homemade applications such as neckties and sticks are defined as improvised, but the Boston EMS kits (surgical tubing and Kelly clamps) also could be considered improvised. All of these methods may sound primitive since the advent of Combat-Application Tourniquets; however CATS are relatively new to the scene. Should we consider every tourniquet applied prior to their introduction to have been improvised? For the price of one CAT, we can assemble 10 tourniquet kits. And if Dr. King's initial contention was that tourniquets were in short supply, then it is a better system to deploy as many as possible. This point may be moot, however, because since the Boston bombing incident, federal grant money has allowed the purchase of CAT units in great numbers within the Metro-Boston area.
Tourniquet efficacy
"It is abundantly clear from the literature that improvised tourniquets almost never work." I feel this study had the wrong focus. Rather than ask which tourniquets have a better success rate, the study should have asked whose tourniquets have a better success rate.
Study after study has shown that successful intubation rates have little to do with the level of training of the practitioner, and everything to do with the frequency of intubation. More tubes equal more successful passes. I propose that this applies to tourniquet application as well. Field personnel who do not practice or have the opportunity to apply tourniquets frequently (which is the great majority of us) may not be applying tourniquets properly. This is why we instituted a massive tourniquet training and review program almost immediately following the 2013 bombing at the Boston Marathon.
Another point not addressed in studies is the effectiveness of nonresponder tourniquets. I would contend that an experienced practitioner with a necktie and a stick would be more successful in hemorrhage control that a civilian with a CAT, but those data have not been explored yet either.
The Loading Officer as unsung hero
All our Action Area Officers performed brilliantly (full disclosure, I was the Loading Officer). However the term Loading Officer is a misnomer. Yes, there is a Loading Officer, but there is actually a Loading Team. It consists of staff at the scene who liaise with the treatment areas and the true unsung heroes, the staff in the Operations Division or Dispatch Center. Dispatchers are in constant contact with the hospitals, scene operations, as well as continuing city service. They perform the hospital destination designations because they have a global sense of traffic, while at the scene we have only a worm's-eye view.
The stay-and-play critique
"All critical patients were evacuated within 1 hour, but some argued this could have been accomplished more quickly were it not for a 'stay-and-play' mentality." I take exception to this assertion. First, the point is contradictory. All critical patients were evacuated within 1 hour, but there was a stay-and-play mentality? Every EMT and paramedic knows that transport times translate into positive results with trauma.
Emergency personnel are taught that what matters most in trauma survival is not basic life support, advance life support, or wheelbarrow, but getting them to the ED fast. Yes, there were slowdowns in the medical tents, but most of delays came from non-EMS personnel who were not well versed in field medicine. We questioned, pleaded, and ultimately ordered some practitioners away from patients as they were attempting to perform procedures that we would never attempt on a trauma patient in the field. This is by no means a condemnation on my part. The staff we worked with was exceptional, and I witnessed true skill, heroism, and compassion. But it also demonstrated to me that a disconnect exists between field medicine and hospital medicine.
I see an opportunity here for training to better acclimate responders to working together with our hospital counterparts. We also need hospital personnel to be oriented to realities and limitations of field medicine.
Lt. Brian Pomodoro is a member of the Boston EMS.
NAPLES, FLA. – Without a single life lost at area hospitals, the medical response to the Boston Marathon bombings was by all accounts remarkable. But what if police and other first-responders had tourniquets and were trained in their use?
As it was, advanced topical hemostatic agents and tourniquets were MIA in the field, forcing all but one prehospital tourniquet to be improvised, according to Massachusetts General Hospital trauma surgeon Lt. Col. David King, U. S. Army Reserve.
"It’s abundantly clear from the literature that improvised tourniquets almost never work," he said. "The fact that every soldier down range has one in his pocket and we couldn’t muster up more than one in the city of Boston is a shame and we need to rectify that."
In all, there were 66 limb injuries, with 29 patients having recognized limb exsanguination at the scene. Of the 27 tourniquets applied, 26 were improvised.
Failure to translate this important lesson from military experience meant that these homemade devices were most likely wildly ineffective and, in some cases, may have even exacerbated blood loss, he said. Surprisingly, there’s no formal protocol for tourniquet application or training paradigm in Massachusetts, such that exists in the Army’s prehospital Tactical Combat Casualty Care handbook.
"If you look across the paramedic medical guidelines, there is no protocol on the right, evidence-based way to put on a tourniquet or what the indications are for a tourniquet," Dr. King said at the annual scientific assembly of the Eastern Association for the Surgery of Trauma (EAST). "We don’t have it, and we need to" have it nationwide.
EAST is hoping to provide some guidance in this area with the development of practice management guidelines (PMG) for tourniquet use in extremity trauma. Dr. King, who heads this PMG committee, reported at the meeting that the guidelines, due out later this year, will emphasize that commercially made tourniquets should be available in the prehospital setting, improvised tourniquets rarely work and cannot be recommended; essentially all commercially made tourniquets are equieffective; and that nonmedical personnel can be trained to correctly apply a proper tourniquet.
The need for bleeding control equipment and training for the public has not gone unnoticed by other stakeholders and is the central focus of the newest statement from the Hartford Consensus, a collaborative group of trauma surgeons, law enforcement officers, and emergency responders led by the American College of Surgeons (J. Am. Coll. Surg. 2014;218:467-75).
In dissecting the medical response to the Boston bombings, much has been made of the fact that Boston is home to six Level 1 trauma centers, and that it was race day, a city holiday, 3 p.m., and low-yield devices were used. Dr. King, however, gave high praise to an unsung hero, the Boston EMS loading officer who managed most of the transport destination decisions and smartly distributed critical cases throughout the area’s hospitals.
All critical patients were evacuated within 1 hour, but some have argued this could have been accomplished more quickly were it not for a "stay-and-play" mentality in the medical tent. The after-action review suggests there probably were enough ambulances to do the job in 30 minutes, said Dr. King, who remarked that the only thing that matters in critical trauma cases is time to surgical hemorrhage control.
"This will remain a controversial issue for Boston, whether staying and playing in the medical tent was a good idea," he said. "I don’t know what the right answer is."
Credit was also given to Massachusetts General, and many of the other city hospitals, for their insistence on regular full-scale drills that allowed quick lock-down of the facility to control access and manage the surge capacity in the emergency department.
"It’s more than just a tabletop exercise," Dr. King said. "When we test the surge capacity, we actually empty the emergency room for real. Usually, it occurs between 5:00 and 7:00 or 4:30 and 7:30 in the morning, but actually doing these things was useful, because when it came time to do it for real, no one was tripping over their own feet."
ED volume on that April afternoon was reduced from 97 patients to just 39 within 1.5 hours. Nontrauma physicians, including psychiatrists, did their part by writing "barebones orders" to get patients transferred to the wards.
Staff at Massachusetts General treated 43 patients, performing nine emergent operations, six amputations, two laparotomies, and one thoracotomy, with two traumatic arrests within the first 72 hours. As with other Boston hospitals, there were no in-hospital deaths, he said.
Patients arrived in such rapid succession, however, that it slowed the electronic medical record system and forced staff to use sequential record numbers, which can be dangerous. Registration couldn’t keep up and orders couldn’t be put in fast enough with 43 patients arriving in roughly 35 minutes, Dr. King explained. Workarounds were often old-school, and reminiscent of his days as a green trauma surgeon serving at Ibn Sina Hospital, Baghdad, Iraq.
"In the midst of this entire event, I turned to my nurse practitioner and asked for x, y, z information on a particular patient," he said. "She didn’t turn to her tablet or desktop computer in the operating room. She pulled out a piece of paper where she’d written all the relevant information. At the end of the event, I looked down and realized I’d done the same thing. I was keeping track of my patients on my pants with a Sharpie."
King describes this as a failure of technology and translation from the battlefield, but noted that the hospital did use the military’s practice of reviewing a critical event. Once they’d finished in the operating room and achieved hemorrhage and contamination control, the entire trauma team was reassembled. They did detailed tertiary trauma surveys and went back through every single patient to determine what additional studies they needed, what injuries had been missed.
"It was remarkable the number of things that were missed," Dr. King said. "No one was bleeding to death, but tons and tons of [small, non–life threatening] missed injuries [like ruptured ear drums], and to me this was one of the more important events that surrounded our response to the entire bombing – sitting down after the dust had settled and carefully going over every patient’s medical record."
Dr. King reported having no financial disclosures.
NAPLES, FLA. – Without a single life lost at area hospitals, the medical response to the Boston Marathon bombings was by all accounts remarkable. But what if police and other first-responders had tourniquets and were trained in their use?
As it was, advanced topical hemostatic agents and tourniquets were MIA in the field, forcing all but one prehospital tourniquet to be improvised, according to Massachusetts General Hospital trauma surgeon Lt. Col. David King, U. S. Army Reserve.
"It’s abundantly clear from the literature that improvised tourniquets almost never work," he said. "The fact that every soldier down range has one in his pocket and we couldn’t muster up more than one in the city of Boston is a shame and we need to rectify that."
In all, there were 66 limb injuries, with 29 patients having recognized limb exsanguination at the scene. Of the 27 tourniquets applied, 26 were improvised.
Failure to translate this important lesson from military experience meant that these homemade devices were most likely wildly ineffective and, in some cases, may have even exacerbated blood loss, he said. Surprisingly, there’s no formal protocol for tourniquet application or training paradigm in Massachusetts, such that exists in the Army’s prehospital Tactical Combat Casualty Care handbook.
"If you look across the paramedic medical guidelines, there is no protocol on the right, evidence-based way to put on a tourniquet or what the indications are for a tourniquet," Dr. King said at the annual scientific assembly of the Eastern Association for the Surgery of Trauma (EAST). "We don’t have it, and we need to" have it nationwide.
EAST is hoping to provide some guidance in this area with the development of practice management guidelines (PMG) for tourniquet use in extremity trauma. Dr. King, who heads this PMG committee, reported at the meeting that the guidelines, due out later this year, will emphasize that commercially made tourniquets should be available in the prehospital setting, improvised tourniquets rarely work and cannot be recommended; essentially all commercially made tourniquets are equieffective; and that nonmedical personnel can be trained to correctly apply a proper tourniquet.
The need for bleeding control equipment and training for the public has not gone unnoticed by other stakeholders and is the central focus of the newest statement from the Hartford Consensus, a collaborative group of trauma surgeons, law enforcement officers, and emergency responders led by the American College of Surgeons (J. Am. Coll. Surg. 2014;218:467-75).
In dissecting the medical response to the Boston bombings, much has been made of the fact that Boston is home to six Level 1 trauma centers, and that it was race day, a city holiday, 3 p.m., and low-yield devices were used. Dr. King, however, gave high praise to an unsung hero, the Boston EMS loading officer who managed most of the transport destination decisions and smartly distributed critical cases throughout the area’s hospitals.
All critical patients were evacuated within 1 hour, but some have argued this could have been accomplished more quickly were it not for a "stay-and-play" mentality in the medical tent. The after-action review suggests there probably were enough ambulances to do the job in 30 minutes, said Dr. King, who remarked that the only thing that matters in critical trauma cases is time to surgical hemorrhage control.
"This will remain a controversial issue for Boston, whether staying and playing in the medical tent was a good idea," he said. "I don’t know what the right answer is."
Credit was also given to Massachusetts General, and many of the other city hospitals, for their insistence on regular full-scale drills that allowed quick lock-down of the facility to control access and manage the surge capacity in the emergency department.
"It’s more than just a tabletop exercise," Dr. King said. "When we test the surge capacity, we actually empty the emergency room for real. Usually, it occurs between 5:00 and 7:00 or 4:30 and 7:30 in the morning, but actually doing these things was useful, because when it came time to do it for real, no one was tripping over their own feet."
ED volume on that April afternoon was reduced from 97 patients to just 39 within 1.5 hours. Nontrauma physicians, including psychiatrists, did their part by writing "barebones orders" to get patients transferred to the wards.
Staff at Massachusetts General treated 43 patients, performing nine emergent operations, six amputations, two laparotomies, and one thoracotomy, with two traumatic arrests within the first 72 hours. As with other Boston hospitals, there were no in-hospital deaths, he said.
Patients arrived in such rapid succession, however, that it slowed the electronic medical record system and forced staff to use sequential record numbers, which can be dangerous. Registration couldn’t keep up and orders couldn’t be put in fast enough with 43 patients arriving in roughly 35 minutes, Dr. King explained. Workarounds were often old-school, and reminiscent of his days as a green trauma surgeon serving at Ibn Sina Hospital, Baghdad, Iraq.
"In the midst of this entire event, I turned to my nurse practitioner and asked for x, y, z information on a particular patient," he said. "She didn’t turn to her tablet or desktop computer in the operating room. She pulled out a piece of paper where she’d written all the relevant information. At the end of the event, I looked down and realized I’d done the same thing. I was keeping track of my patients on my pants with a Sharpie."
King describes this as a failure of technology and translation from the battlefield, but noted that the hospital did use the military’s practice of reviewing a critical event. Once they’d finished in the operating room and achieved hemorrhage and contamination control, the entire trauma team was reassembled. They did detailed tertiary trauma surveys and went back through every single patient to determine what additional studies they needed, what injuries had been missed.
"It was remarkable the number of things that were missed," Dr. King said. "No one was bleeding to death, but tons and tons of [small, non–life threatening] missed injuries [like ruptured ear drums], and to me this was one of the more important events that surrounded our response to the entire bombing – sitting down after the dust had settled and carefully going over every patient’s medical record."
Dr. King reported having no financial disclosures.
EXPERT ANALYSIS FROM EAST SCIENTIFIC ASSEMBLY 2014
Campus meningitis outbreaks tamed but can our approach last?
The incidence of all meningococcal serotypes is in decline, except for the spike in serogroup B on college campuses.
"Although we do not expect an increase in MenB outbreaks on college campuses, the CDC has recently formed a meningococcal outbreak work group," said Dr. Manisha Patel, medical officer with the Centers for Disease Control and Prevention’s Meningitis and Vaccine Preventable Diseases Branch.
Five school-based meningitis B (MenB) outbreaks have been reported since 2009, involving four undergraduates at the University of California–Santa Barbara (UCSB) since November 2013 and eight cases from March to November 2013 among students or persons with links to Princeton University.
There is no licensed MenB vaccine in the United States, but the Princeton isolates expressed two of the four antigens (fHBP and NHBA) in sufficient quantities to suggest protection with Bexsero, a recombinant MenB vaccine (Novartis) recently licensed in Europe, Australia, and Canada, Dr. Patel said at the CDC’s most recent Advisory Committee on Immunization Practices meeting.
An on-campus vaccination campaign that continued through Feb. 20, 2014, resulted in 95% coverage for dose 1 and 88% coverage for dose 2 among 5,772 at-risk persons.
Based on mandatory follow-up reporting to the Food and Drug Administration, the serious adverse event rate was 2.0/1,000 vaccinees following the first dose and 0.2/1,000 vaccinees after the second. None of these events have been determined to be causally related to the vaccine, and there have been no concerning patterns among other types of reported adverse events, she said.
The target population at Princeton includes all undergraduates, graduate students living in dormitories, at-risk students, faculty, and staff, as well as spouses or parents living with undergraduates in dorms.
The attack rate during the outbreak was 134/100,000 among undergraduates. No deaths occurred, but two cases had sequelae of neurocognitive deficit or hearing loss, Dr. Patel said.
The attack rate at UCSB was 21.1/100,000 among 17-22 years olds, or 234-fold higher than the incidence rate for this age group in the general population. All four students recovered, but one had bilateral foot amputation.
Procurement of the Bexsero vaccine began in August 2013 with the initial proposal to the FDA to explore its use in an outbreak under an expanded access Investigational New Drug (IND) protocol, Dr. Patel explained. Other steps along the way included isolate testing by Novartis, epidemiologic investigations, CDC Institutional Review Board approval, and issuance of an FDA safe to proceed letter before contractual agreements between the CDC, Novartis, and Princeton University were finalized in December 2013.
A CDC-sponsored expanded–access IND protocol was subsequently approved by the FDA for Bexsero use at UCSB. First-dose vaccinations began Feb. 24 – this time with an estimated 20,000 persons eligible for vaccination, Dr. Patel said.
"Vaccination is now possible in response to meningococcal B outbreaks," she said.
Dr. Peter Dull, head of meningitis cluster, Novartis Vaccines and Diagnostics, Cambridge, Mass., agreed that the vaccinations are a great example of collaboration among entities, but he cautioned ACIP members to take a step back.
"If you look at the timeline Dr. Patel laid out as to how long it took to walk through the steps, yes, there’s work with the FDA approving the IND, and yes, there’s set up of the implementation campaign," he said. "But also on the company side, we’re not in surge capacity that you can pick up the phone, and we can label and ship unlicensed vaccines in the U.S.A. very quickly.
"These outbreaks clearly happen fast, and we do have to have a more sustained solution to this."
Dr. Patel pointed to the challenges of using an unlicensed vaccine including the logistics of vaccine procurement and implementation, with the mandate to meet safety follow-up requirements. Plus the IND preparation process must address specifics of the outbreak.
The new meningococcal outbreak work group will bring together federal vaccine experts, state public and college health officials, university administrators, and insurance industry representatives. The group is tasked with reviewing recent epidemiology data of meningococcal disease and outbreaks, considering options for updating the current meningococcal outbreak guidelines, and developing guidance for the use of meningococcal vaccines, both licensed and unlicensed in an outbreak, she said.
Dr. Patel reported having no financial disclosures.
The incidence of all meningococcal serotypes is in decline, except for the spike in serogroup B on college campuses.
"Although we do not expect an increase in MenB outbreaks on college campuses, the CDC has recently formed a meningococcal outbreak work group," said Dr. Manisha Patel, medical officer with the Centers for Disease Control and Prevention’s Meningitis and Vaccine Preventable Diseases Branch.
Five school-based meningitis B (MenB) outbreaks have been reported since 2009, involving four undergraduates at the University of California–Santa Barbara (UCSB) since November 2013 and eight cases from March to November 2013 among students or persons with links to Princeton University.
There is no licensed MenB vaccine in the United States, but the Princeton isolates expressed two of the four antigens (fHBP and NHBA) in sufficient quantities to suggest protection with Bexsero, a recombinant MenB vaccine (Novartis) recently licensed in Europe, Australia, and Canada, Dr. Patel said at the CDC’s most recent Advisory Committee on Immunization Practices meeting.
An on-campus vaccination campaign that continued through Feb. 20, 2014, resulted in 95% coverage for dose 1 and 88% coverage for dose 2 among 5,772 at-risk persons.
Based on mandatory follow-up reporting to the Food and Drug Administration, the serious adverse event rate was 2.0/1,000 vaccinees following the first dose and 0.2/1,000 vaccinees after the second. None of these events have been determined to be causally related to the vaccine, and there have been no concerning patterns among other types of reported adverse events, she said.
The target population at Princeton includes all undergraduates, graduate students living in dormitories, at-risk students, faculty, and staff, as well as spouses or parents living with undergraduates in dorms.
The attack rate during the outbreak was 134/100,000 among undergraduates. No deaths occurred, but two cases had sequelae of neurocognitive deficit or hearing loss, Dr. Patel said.
The attack rate at UCSB was 21.1/100,000 among 17-22 years olds, or 234-fold higher than the incidence rate for this age group in the general population. All four students recovered, but one had bilateral foot amputation.
Procurement of the Bexsero vaccine began in August 2013 with the initial proposal to the FDA to explore its use in an outbreak under an expanded access Investigational New Drug (IND) protocol, Dr. Patel explained. Other steps along the way included isolate testing by Novartis, epidemiologic investigations, CDC Institutional Review Board approval, and issuance of an FDA safe to proceed letter before contractual agreements between the CDC, Novartis, and Princeton University were finalized in December 2013.
A CDC-sponsored expanded–access IND protocol was subsequently approved by the FDA for Bexsero use at UCSB. First-dose vaccinations began Feb. 24 – this time with an estimated 20,000 persons eligible for vaccination, Dr. Patel said.
"Vaccination is now possible in response to meningococcal B outbreaks," she said.
Dr. Peter Dull, head of meningitis cluster, Novartis Vaccines and Diagnostics, Cambridge, Mass., agreed that the vaccinations are a great example of collaboration among entities, but he cautioned ACIP members to take a step back.
"If you look at the timeline Dr. Patel laid out as to how long it took to walk through the steps, yes, there’s work with the FDA approving the IND, and yes, there’s set up of the implementation campaign," he said. "But also on the company side, we’re not in surge capacity that you can pick up the phone, and we can label and ship unlicensed vaccines in the U.S.A. very quickly.
"These outbreaks clearly happen fast, and we do have to have a more sustained solution to this."
Dr. Patel pointed to the challenges of using an unlicensed vaccine including the logistics of vaccine procurement and implementation, with the mandate to meet safety follow-up requirements. Plus the IND preparation process must address specifics of the outbreak.
The new meningococcal outbreak work group will bring together federal vaccine experts, state public and college health officials, university administrators, and insurance industry representatives. The group is tasked with reviewing recent epidemiology data of meningococcal disease and outbreaks, considering options for updating the current meningococcal outbreak guidelines, and developing guidance for the use of meningococcal vaccines, both licensed and unlicensed in an outbreak, she said.
Dr. Patel reported having no financial disclosures.
The incidence of all meningococcal serotypes is in decline, except for the spike in serogroup B on college campuses.
"Although we do not expect an increase in MenB outbreaks on college campuses, the CDC has recently formed a meningococcal outbreak work group," said Dr. Manisha Patel, medical officer with the Centers for Disease Control and Prevention’s Meningitis and Vaccine Preventable Diseases Branch.
Five school-based meningitis B (MenB) outbreaks have been reported since 2009, involving four undergraduates at the University of California–Santa Barbara (UCSB) since November 2013 and eight cases from March to November 2013 among students or persons with links to Princeton University.
There is no licensed MenB vaccine in the United States, but the Princeton isolates expressed two of the four antigens (fHBP and NHBA) in sufficient quantities to suggest protection with Bexsero, a recombinant MenB vaccine (Novartis) recently licensed in Europe, Australia, and Canada, Dr. Patel said at the CDC’s most recent Advisory Committee on Immunization Practices meeting.
An on-campus vaccination campaign that continued through Feb. 20, 2014, resulted in 95% coverage for dose 1 and 88% coverage for dose 2 among 5,772 at-risk persons.
Based on mandatory follow-up reporting to the Food and Drug Administration, the serious adverse event rate was 2.0/1,000 vaccinees following the first dose and 0.2/1,000 vaccinees after the second. None of these events have been determined to be causally related to the vaccine, and there have been no concerning patterns among other types of reported adverse events, she said.
The target population at Princeton includes all undergraduates, graduate students living in dormitories, at-risk students, faculty, and staff, as well as spouses or parents living with undergraduates in dorms.
The attack rate during the outbreak was 134/100,000 among undergraduates. No deaths occurred, but two cases had sequelae of neurocognitive deficit or hearing loss, Dr. Patel said.
The attack rate at UCSB was 21.1/100,000 among 17-22 years olds, or 234-fold higher than the incidence rate for this age group in the general population. All four students recovered, but one had bilateral foot amputation.
Procurement of the Bexsero vaccine began in August 2013 with the initial proposal to the FDA to explore its use in an outbreak under an expanded access Investigational New Drug (IND) protocol, Dr. Patel explained. Other steps along the way included isolate testing by Novartis, epidemiologic investigations, CDC Institutional Review Board approval, and issuance of an FDA safe to proceed letter before contractual agreements between the CDC, Novartis, and Princeton University were finalized in December 2013.
A CDC-sponsored expanded–access IND protocol was subsequently approved by the FDA for Bexsero use at UCSB. First-dose vaccinations began Feb. 24 – this time with an estimated 20,000 persons eligible for vaccination, Dr. Patel said.
"Vaccination is now possible in response to meningococcal B outbreaks," she said.
Dr. Peter Dull, head of meningitis cluster, Novartis Vaccines and Diagnostics, Cambridge, Mass., agreed that the vaccinations are a great example of collaboration among entities, but he cautioned ACIP members to take a step back.
"If you look at the timeline Dr. Patel laid out as to how long it took to walk through the steps, yes, there’s work with the FDA approving the IND, and yes, there’s set up of the implementation campaign," he said. "But also on the company side, we’re not in surge capacity that you can pick up the phone, and we can label and ship unlicensed vaccines in the U.S.A. very quickly.
"These outbreaks clearly happen fast, and we do have to have a more sustained solution to this."
Dr. Patel pointed to the challenges of using an unlicensed vaccine including the logistics of vaccine procurement and implementation, with the mandate to meet safety follow-up requirements. Plus the IND preparation process must address specifics of the outbreak.
The new meningococcal outbreak work group will bring together federal vaccine experts, state public and college health officials, university administrators, and insurance industry representatives. The group is tasked with reviewing recent epidemiology data of meningococcal disease and outbreaks, considering options for updating the current meningococcal outbreak guidelines, and developing guidance for the use of meningococcal vaccines, both licensed and unlicensed in an outbreak, she said.
Dr. Patel reported having no financial disclosures.
FROM AN ACIP MEETING
Sizzle magnets: a worrisome buzz in the emergency department
NAPLES, FLA. – They sizzle, they buzz, they vibrate. And they’re sending some children to the emergency department.
"One of the newest things we’re seeing a fair amount of are magnets, those sizzle magnets," reported Karen Macauley, DHA, R.N., trauma program director, All Children’s Hospital, St. Petersburg, Fla., and secretary of the Society of Trauma Nurses.
So-called "sizzle" magnets make a sound when they connect and are being sold as novelty toys, jewelry, and even for purported health benefits. One website specializing in gifts for the visually impaired notes that the magnetic field of these hematite magnets is "so strong," they’ll stay put if placed on the front and back of hands or ear lobes.
That strong magnetic force, however, can create havoc if children ingest magnets of any kind, Dr. Macauley said at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
"If the magnets stay in a string, they’re probably okay; but if they swallow one and wait a little while and swallow another, you can start to imagine what happens as it starts to pass through," she said. "One magnet stays, another comes in another part of the bowel, and they sizzle together. Now all of a sudden you have magnets that aren’t moving and cause a lot of necrosis. They’re actually very dangerous, and we don’t think too much about how dangerous they can be."
Between 2009 and 2011, an estimated 1,700 emergency department visits occurred because of magnet ingestion, with more than 70% of those cases involving children between age 4 and 12 years, according to researchers at the U.S. Public Interest Research Group, which highlighted magnets in its most recent annual toy safety survey.
Dr. Macauley also cautioned that button-size batteries, now in everything from remote controls to Grandma’s singing greeting card and hearing aid, are also a concern because they can cause burns or erode through tissue if ingested or put into body orifices.
"These batteries are everywhere, and kids do crazy stuff with them," she said.
The National Capital Poison Center, which operates a 24-hour National Battery Ingestion Hotline (202-625-3333)* for swallowed battery cases, estimates 3,500 Americans of all ages swallow miniature disc or button batteries each year.
The problem has been recognized for some time, but what few parents or providers realize is how quickly burns can occur, Dr. Macauley said.
She described a case involving a 6-year-old girl who picked up a small battery off the playground and amazed her friends by making it disappear in her ear. The child complained of severe pain overnight and presented to the emergency department in the morning, where surgery to remove the battery revealed third-degree burns to the ear canal and 65% perforation of the eardrum.
"It seemed like it should have been nothing; it was in there maybe 12 hours, but she had very severe injuries to her ear," Dr. Macauley said. "She’s been in the operating room probably a total of six different times trying to get that repaired, has a fair amount of hearing loss that stays, and ended up at a specialist hospital, out of network, to get a graft put in that ear."
Greeting card makers such as Hallmark have taken steps to secure button batteries, such as enclosing them underneath metal caps or in modules in which the cap is secured with screws. And the card makers warn consumers to properly dispose of old batteries after they’ve been replaced.
Dr. Macauley reported having no financial disclosures.
Correction, 3/5/2014: An earlier version of this article misstated the phone number for the Battery Ingestion hotline.
NAPLES, FLA. – They sizzle, they buzz, they vibrate. And they’re sending some children to the emergency department.
"One of the newest things we’re seeing a fair amount of are magnets, those sizzle magnets," reported Karen Macauley, DHA, R.N., trauma program director, All Children’s Hospital, St. Petersburg, Fla., and secretary of the Society of Trauma Nurses.
So-called "sizzle" magnets make a sound when they connect and are being sold as novelty toys, jewelry, and even for purported health benefits. One website specializing in gifts for the visually impaired notes that the magnetic field of these hematite magnets is "so strong," they’ll stay put if placed on the front and back of hands or ear lobes.
That strong magnetic force, however, can create havoc if children ingest magnets of any kind, Dr. Macauley said at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
"If the magnets stay in a string, they’re probably okay; but if they swallow one and wait a little while and swallow another, you can start to imagine what happens as it starts to pass through," she said. "One magnet stays, another comes in another part of the bowel, and they sizzle together. Now all of a sudden you have magnets that aren’t moving and cause a lot of necrosis. They’re actually very dangerous, and we don’t think too much about how dangerous they can be."
Between 2009 and 2011, an estimated 1,700 emergency department visits occurred because of magnet ingestion, with more than 70% of those cases involving children between age 4 and 12 years, according to researchers at the U.S. Public Interest Research Group, which highlighted magnets in its most recent annual toy safety survey.
Dr. Macauley also cautioned that button-size batteries, now in everything from remote controls to Grandma’s singing greeting card and hearing aid, are also a concern because they can cause burns or erode through tissue if ingested or put into body orifices.
"These batteries are everywhere, and kids do crazy stuff with them," she said.
The National Capital Poison Center, which operates a 24-hour National Battery Ingestion Hotline (202-625-3333)* for swallowed battery cases, estimates 3,500 Americans of all ages swallow miniature disc or button batteries each year.
The problem has been recognized for some time, but what few parents or providers realize is how quickly burns can occur, Dr. Macauley said.
She described a case involving a 6-year-old girl who picked up a small battery off the playground and amazed her friends by making it disappear in her ear. The child complained of severe pain overnight and presented to the emergency department in the morning, where surgery to remove the battery revealed third-degree burns to the ear canal and 65% perforation of the eardrum.
"It seemed like it should have been nothing; it was in there maybe 12 hours, but she had very severe injuries to her ear," Dr. Macauley said. "She’s been in the operating room probably a total of six different times trying to get that repaired, has a fair amount of hearing loss that stays, and ended up at a specialist hospital, out of network, to get a graft put in that ear."
Greeting card makers such as Hallmark have taken steps to secure button batteries, such as enclosing them underneath metal caps or in modules in which the cap is secured with screws. And the card makers warn consumers to properly dispose of old batteries after they’ve been replaced.
Dr. Macauley reported having no financial disclosures.
Correction, 3/5/2014: An earlier version of this article misstated the phone number for the Battery Ingestion hotline.
NAPLES, FLA. – They sizzle, they buzz, they vibrate. And they’re sending some children to the emergency department.
"One of the newest things we’re seeing a fair amount of are magnets, those sizzle magnets," reported Karen Macauley, DHA, R.N., trauma program director, All Children’s Hospital, St. Petersburg, Fla., and secretary of the Society of Trauma Nurses.
So-called "sizzle" magnets make a sound when they connect and are being sold as novelty toys, jewelry, and even for purported health benefits. One website specializing in gifts for the visually impaired notes that the magnetic field of these hematite magnets is "so strong," they’ll stay put if placed on the front and back of hands or ear lobes.
That strong magnetic force, however, can create havoc if children ingest magnets of any kind, Dr. Macauley said at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
"If the magnets stay in a string, they’re probably okay; but if they swallow one and wait a little while and swallow another, you can start to imagine what happens as it starts to pass through," she said. "One magnet stays, another comes in another part of the bowel, and they sizzle together. Now all of a sudden you have magnets that aren’t moving and cause a lot of necrosis. They’re actually very dangerous, and we don’t think too much about how dangerous they can be."
Between 2009 and 2011, an estimated 1,700 emergency department visits occurred because of magnet ingestion, with more than 70% of those cases involving children between age 4 and 12 years, according to researchers at the U.S. Public Interest Research Group, which highlighted magnets in its most recent annual toy safety survey.
Dr. Macauley also cautioned that button-size batteries, now in everything from remote controls to Grandma’s singing greeting card and hearing aid, are also a concern because they can cause burns or erode through tissue if ingested or put into body orifices.
"These batteries are everywhere, and kids do crazy stuff with them," she said.
The National Capital Poison Center, which operates a 24-hour National Battery Ingestion Hotline (202-625-3333)* for swallowed battery cases, estimates 3,500 Americans of all ages swallow miniature disc or button batteries each year.
The problem has been recognized for some time, but what few parents or providers realize is how quickly burns can occur, Dr. Macauley said.
She described a case involving a 6-year-old girl who picked up a small battery off the playground and amazed her friends by making it disappear in her ear. The child complained of severe pain overnight and presented to the emergency department in the morning, where surgery to remove the battery revealed third-degree burns to the ear canal and 65% perforation of the eardrum.
"It seemed like it should have been nothing; it was in there maybe 12 hours, but she had very severe injuries to her ear," Dr. Macauley said. "She’s been in the operating room probably a total of six different times trying to get that repaired, has a fair amount of hearing loss that stays, and ended up at a specialist hospital, out of network, to get a graft put in that ear."
Greeting card makers such as Hallmark have taken steps to secure button batteries, such as enclosing them underneath metal caps or in modules in which the cap is secured with screws. And the card makers warn consumers to properly dispose of old batteries after they’ve been replaced.
Dr. Macauley reported having no financial disclosures.
Correction, 3/5/2014: An earlier version of this article misstated the phone number for the Battery Ingestion hotline.
AT EAST 2014
ACIP balks at recommending LAIV
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted to retain its current recommendations and patient populations for annual influenza vaccination Feb. 26 at its meeting.
The vote was unanimous, but it belies the desire by some for ACIP to call for vote on whether live-attenuated influenza vaccine (LAIV) should be recommended preferentially over inactivated influenza vaccine (IIV) for healthy children.
"Many of us feel it’s long overdue," said Dr. Jeff Duchin, a member of the ACIP influenza working group. "We have a vaccine that even the CDC has acknowledged on a public Web page is superior in young children. Yet we haven’t made that step to make that recommendation, which does influence a lot of health care providers."
LAIV is recommended preferentially for various age groups in the United Kingdom, Canada, Israel, and Germany, as well as in the states of Oregon and Washington.
Several studies indicate that LAIV may have advantages over IIV in younger populations, but this is not clear in older populations, said influenza work group member Dr. Lisa Grohskopf of the influenza division in the CDC National Center for Immunization and Respiratory Diseases (NCIRD). The work group also struggled with the lower age limit for children when evaluating the efficacy and safety evidence because LAIV is not licensed for children under 2 years of age.
Based on review of the efficacy data, LAIV was associated with a decreased risk of lab-confirmed influenza among 2- to 8-year-olds based on high-quality randomized evidence, but it showed no difference over IIV among 9- to 18-year-olds based on very-low-quality observational data, Dr. Grohskopf said. Among 2- to 8-year-olds, LAIV also was associated with a decreased risk of otitis media, but there was no difference between vaccines with regard to hospitalization, medically attended acute respiratory illness, or influenzalike illness.
As ACIP goes forward, it will need to "harmonize" any possible changes to its recommendations with the American Academy of Pediatrics, which doesn’t meet on the issue until April, Dr. Grohskopf said. It also will have to consider the LAIV supply and the safety of quadrivalent vaccines since all of the studies in the evidence review were done when only trivalent vaccines were available.
Four new, recently licensed vaccines are now available for the 2013-1014 flu season, including FluMist Quadrivalent nasal LAIV (LAIV4) from MedImmune for persons aged 2-49 years; Fluarix Quadrivalent inactivated influenza vaccine (IIV4) from GlaxoSmithKline for those 3 years and older; Flucelvax, a cell culture–based inactivated influenza vaccine (ccIIV3) from Novartis for patients aged 18 years and older; and Flublok, a recombinant hemagglutinin vaccine (RIV3) from Protein Sciences for adults aged 18-49 years.
No new safety concerns have been detected for LAIV4, IIV4, or IIV3 vaccines during the 2013-14 influenza season in patients less than 18 years of age based on interim Vaccine Adverse Event Reporting System surveillance data and Vaccine Safety Datalink data, said Dr. Maria Cano of the CDC National Center for Emerging and Infectious Zoonotic Diseases. Uptake has been limited for the IIV4, cell culture–based IIV3, and recombinant IIV3 vaccines, compared with 194,080 LAIV4 doses and 3.1 million IIV3 doses.
The influenza work group also assessed influenza vaccine safety data in children aged 2-8 years and found no evidence for an increased risk of serious adverse events or medically attended wheezing after LAIV vs. trivalent influenza vaccine (TIV), reported Dr. Emmanuel Walter, director of the Duke Translational Medicine Institute’s clinical vaccine unit at Duke University Medical Center, Durham, N.C. There was evidence for a transient increased risk of mild fever after LAIV vs. TIV, but it was seen only in one study during a single influenza season.
In a pooled safety analysis of LAIV vs. IIV in healthy children, there was no difference between vaccines in medically attended wheezing, fever, or vaccine-related serious adverse events, Dr. Grohskopf said. Both she and Dr. Walter observed that the analyses were limited by several factors, including few studies directly comparing LAIV and IIV, lack of standardized outcome definitions across studies, and difficulty judging the risk of serious rare adverse events.
This led some in attendance to call for additional data on adverse events like seizures at the June meeting, while others tasked the work group with clarifying and separating contraindications from precautions before opening up use of the LAIV to a wider population.
Dr. Walter reported previously serving as a clinical investigator within the last 12 months for GlaxoSmithKline, Merck, and Pfizer and as a data monitoring board member and consultant for Novartis. No other presenters reported having financial disclosures.
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted to retain its current recommendations and patient populations for annual influenza vaccination Feb. 26 at its meeting.
The vote was unanimous, but it belies the desire by some for ACIP to call for vote on whether live-attenuated influenza vaccine (LAIV) should be recommended preferentially over inactivated influenza vaccine (IIV) for healthy children.
"Many of us feel it’s long overdue," said Dr. Jeff Duchin, a member of the ACIP influenza working group. "We have a vaccine that even the CDC has acknowledged on a public Web page is superior in young children. Yet we haven’t made that step to make that recommendation, which does influence a lot of health care providers."
LAIV is recommended preferentially for various age groups in the United Kingdom, Canada, Israel, and Germany, as well as in the states of Oregon and Washington.
Several studies indicate that LAIV may have advantages over IIV in younger populations, but this is not clear in older populations, said influenza work group member Dr. Lisa Grohskopf of the influenza division in the CDC National Center for Immunization and Respiratory Diseases (NCIRD). The work group also struggled with the lower age limit for children when evaluating the efficacy and safety evidence because LAIV is not licensed for children under 2 years of age.
Based on review of the efficacy data, LAIV was associated with a decreased risk of lab-confirmed influenza among 2- to 8-year-olds based on high-quality randomized evidence, but it showed no difference over IIV among 9- to 18-year-olds based on very-low-quality observational data, Dr. Grohskopf said. Among 2- to 8-year-olds, LAIV also was associated with a decreased risk of otitis media, but there was no difference between vaccines with regard to hospitalization, medically attended acute respiratory illness, or influenzalike illness.
As ACIP goes forward, it will need to "harmonize" any possible changes to its recommendations with the American Academy of Pediatrics, which doesn’t meet on the issue until April, Dr. Grohskopf said. It also will have to consider the LAIV supply and the safety of quadrivalent vaccines since all of the studies in the evidence review were done when only trivalent vaccines were available.
Four new, recently licensed vaccines are now available for the 2013-1014 flu season, including FluMist Quadrivalent nasal LAIV (LAIV4) from MedImmune for persons aged 2-49 years; Fluarix Quadrivalent inactivated influenza vaccine (IIV4) from GlaxoSmithKline for those 3 years and older; Flucelvax, a cell culture–based inactivated influenza vaccine (ccIIV3) from Novartis for patients aged 18 years and older; and Flublok, a recombinant hemagglutinin vaccine (RIV3) from Protein Sciences for adults aged 18-49 years.
No new safety concerns have been detected for LAIV4, IIV4, or IIV3 vaccines during the 2013-14 influenza season in patients less than 18 years of age based on interim Vaccine Adverse Event Reporting System surveillance data and Vaccine Safety Datalink data, said Dr. Maria Cano of the CDC National Center for Emerging and Infectious Zoonotic Diseases. Uptake has been limited for the IIV4, cell culture–based IIV3, and recombinant IIV3 vaccines, compared with 194,080 LAIV4 doses and 3.1 million IIV3 doses.
The influenza work group also assessed influenza vaccine safety data in children aged 2-8 years and found no evidence for an increased risk of serious adverse events or medically attended wheezing after LAIV vs. trivalent influenza vaccine (TIV), reported Dr. Emmanuel Walter, director of the Duke Translational Medicine Institute’s clinical vaccine unit at Duke University Medical Center, Durham, N.C. There was evidence for a transient increased risk of mild fever after LAIV vs. TIV, but it was seen only in one study during a single influenza season.
In a pooled safety analysis of LAIV vs. IIV in healthy children, there was no difference between vaccines in medically attended wheezing, fever, or vaccine-related serious adverse events, Dr. Grohskopf said. Both she and Dr. Walter observed that the analyses were limited by several factors, including few studies directly comparing LAIV and IIV, lack of standardized outcome definitions across studies, and difficulty judging the risk of serious rare adverse events.
This led some in attendance to call for additional data on adverse events like seizures at the June meeting, while others tasked the work group with clarifying and separating contraindications from precautions before opening up use of the LAIV to a wider population.
Dr. Walter reported previously serving as a clinical investigator within the last 12 months for GlaxoSmithKline, Merck, and Pfizer and as a data monitoring board member and consultant for Novartis. No other presenters reported having financial disclosures.
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted to retain its current recommendations and patient populations for annual influenza vaccination Feb. 26 at its meeting.
The vote was unanimous, but it belies the desire by some for ACIP to call for vote on whether live-attenuated influenza vaccine (LAIV) should be recommended preferentially over inactivated influenza vaccine (IIV) for healthy children.
"Many of us feel it’s long overdue," said Dr. Jeff Duchin, a member of the ACIP influenza working group. "We have a vaccine that even the CDC has acknowledged on a public Web page is superior in young children. Yet we haven’t made that step to make that recommendation, which does influence a lot of health care providers."
LAIV is recommended preferentially for various age groups in the United Kingdom, Canada, Israel, and Germany, as well as in the states of Oregon and Washington.
Several studies indicate that LAIV may have advantages over IIV in younger populations, but this is not clear in older populations, said influenza work group member Dr. Lisa Grohskopf of the influenza division in the CDC National Center for Immunization and Respiratory Diseases (NCIRD). The work group also struggled with the lower age limit for children when evaluating the efficacy and safety evidence because LAIV is not licensed for children under 2 years of age.
Based on review of the efficacy data, LAIV was associated with a decreased risk of lab-confirmed influenza among 2- to 8-year-olds based on high-quality randomized evidence, but it showed no difference over IIV among 9- to 18-year-olds based on very-low-quality observational data, Dr. Grohskopf said. Among 2- to 8-year-olds, LAIV also was associated with a decreased risk of otitis media, but there was no difference between vaccines with regard to hospitalization, medically attended acute respiratory illness, or influenzalike illness.
As ACIP goes forward, it will need to "harmonize" any possible changes to its recommendations with the American Academy of Pediatrics, which doesn’t meet on the issue until April, Dr. Grohskopf said. It also will have to consider the LAIV supply and the safety of quadrivalent vaccines since all of the studies in the evidence review were done when only trivalent vaccines were available.
Four new, recently licensed vaccines are now available for the 2013-1014 flu season, including FluMist Quadrivalent nasal LAIV (LAIV4) from MedImmune for persons aged 2-49 years; Fluarix Quadrivalent inactivated influenza vaccine (IIV4) from GlaxoSmithKline for those 3 years and older; Flucelvax, a cell culture–based inactivated influenza vaccine (ccIIV3) from Novartis for patients aged 18 years and older; and Flublok, a recombinant hemagglutinin vaccine (RIV3) from Protein Sciences for adults aged 18-49 years.
No new safety concerns have been detected for LAIV4, IIV4, or IIV3 vaccines during the 2013-14 influenza season in patients less than 18 years of age based on interim Vaccine Adverse Event Reporting System surveillance data and Vaccine Safety Datalink data, said Dr. Maria Cano of the CDC National Center for Emerging and Infectious Zoonotic Diseases. Uptake has been limited for the IIV4, cell culture–based IIV3, and recombinant IIV3 vaccines, compared with 194,080 LAIV4 doses and 3.1 million IIV3 doses.
The influenza work group also assessed influenza vaccine safety data in children aged 2-8 years and found no evidence for an increased risk of serious adverse events or medically attended wheezing after LAIV vs. trivalent influenza vaccine (TIV), reported Dr. Emmanuel Walter, director of the Duke Translational Medicine Institute’s clinical vaccine unit at Duke University Medical Center, Durham, N.C. There was evidence for a transient increased risk of mild fever after LAIV vs. TIV, but it was seen only in one study during a single influenza season.
In a pooled safety analysis of LAIV vs. IIV in healthy children, there was no difference between vaccines in medically attended wheezing, fever, or vaccine-related serious adverse events, Dr. Grohskopf said. Both she and Dr. Walter observed that the analyses were limited by several factors, including few studies directly comparing LAIV and IIV, lack of standardized outcome definitions across studies, and difficulty judging the risk of serious rare adverse events.
This led some in attendance to call for additional data on adverse events like seizures at the June meeting, while others tasked the work group with clarifying and separating contraindications from precautions before opening up use of the LAIV to a wider population.
Dr. Walter reported previously serving as a clinical investigator within the last 12 months for GlaxoSmithKline, Merck, and Pfizer and as a data monitoring board member and consultant for Novartis. No other presenters reported having financial disclosures.
FROM AN ACIP MEETING
Curb Vaccine-preventable Diseases in Patients with Diabetes
SAN FRANCISCO – Patients with diabetes have an increased incidence for a number of infections, but vaccine-preventable diseases need not be among them.
"In the risk/benefit category, there’s a lot of potential benefit to these" immunizations, Dr. David Parenti said at the annual advanced postgraduate course held by the American Diabetes Association.
Studies have shown that patients with diabetes who are aged 23-59 years have a twofold higher relative risk of acute hepatitis B virus, compared with those without diabetes. In addition, the diabetic case fatality rate is more than double, at about 5%.
Individual glucose monitoring has reduced the transmission of hepatitis B virus by contaminated medical equipment, but outbreaks still occur in a variety of settings because of lapses in infection control, said Dr. Parenti, professor of medicine at George Washington University, Washington.
Still, vaccination rates are no better among patients with diabetes than those without. From 1999-2004 to 2005-2008, vaccination rates inched up only slightly among diabetics for both hepatitis A (9.3% to 15.4%) and hepatitis B (15.2% to 22.4%), according to National Health and Nutrition Examination Surveys (Hepatology 2011;54:1167-78).
Indeed, a show of hands revealed that less than a dozen of the roughly 500 attendees at the meeting had vaccinated their patients.
"I do think hepatitis B vaccination is important, but the question is: How do you know whether it works and how do you monitor it?" Dr. Parenti said.
A variety of factors can impair the immunogenicity of the hepatitis B vaccine, including gluteal or intradermal administration, increased age, higher body mass index, and genetics, such as human leukocyte antigen-DR3, which is present in about 95% of people with type 1 diabetes.
Unpublished data from the Centers for Disease Control and Prevention suggest that diabetes patients experience a similar drop in seroprotection with age (80% at 41-59 years, 65% at 60-69 years, and less than 40% at 70 years and older), he noted.
For patients who fail to respond to the first or second series of vaccinations, based on postimmunization serologic testing, using a higher 40 mcg-dose or four doses at 0, 1, 2, and 6 months has been shown to improve immunologic responses. There is no need, however, to restart a series if it was interrupted, regardless of the duration between vaccinations, Dr. Parenti said.
Only about 15% of individuals receive the herpes zoster vaccination, approved for persons aged 50 years and older, despite a third of the population expected to develop shingles within their lifetime. The risk is nearly doubled among diabetics under age 49 years with a hemoglobin A1c exceeding 8%, he said.
For those who do get vaccinated, however, a small Japanese study (J. Infect. 2013;67:215-9) reported similar humoral and cellular responses 3 and 6 months postvaccination among patients aged 60-70 years, with and without diabetes, he noted.
One attendee said that Medicare often refuses to cover herpes zoster and hepatitis vaccinations, leading her to remark, "It’s kind of turning into a class issue. My patients with money will get the vaccine."
It’s not just Medicare, but a significant number of other insurers who aren’t paying for the vaccines, particularly the pricey $300-$400 zoster vaccine, Dr. Parenti replied. Much depends on the patient’s plan. For example, the shingles vaccine is covered by Medicare Part D, but not Part A or Part B, while Medicare Part B covers recipients at "high or medium risk" for hepatitis B, including those with diabetes or end-stage renal disease. At this point, prior authorization is needed for all insurers, including Medicare, and appeals may be necessary, he said.
Dr. Parenti reported serving on a data safety monitoring board for the National Institute of Allergy and Infectious Diseases and receiving research support from Merck, Pfizer, Cubist Pharmaceuticals, Janssen Pharmaceuticals, Insight, and the National Institutes of Health.
SAN FRANCISCO – Patients with diabetes have an increased incidence for a number of infections, but vaccine-preventable diseases need not be among them.
"In the risk/benefit category, there’s a lot of potential benefit to these" immunizations, Dr. David Parenti said at the annual advanced postgraduate course held by the American Diabetes Association.
Studies have shown that patients with diabetes who are aged 23-59 years have a twofold higher relative risk of acute hepatitis B virus, compared with those without diabetes. In addition, the diabetic case fatality rate is more than double, at about 5%.
Individual glucose monitoring has reduced the transmission of hepatitis B virus by contaminated medical equipment, but outbreaks still occur in a variety of settings because of lapses in infection control, said Dr. Parenti, professor of medicine at George Washington University, Washington.
Still, vaccination rates are no better among patients with diabetes than those without. From 1999-2004 to 2005-2008, vaccination rates inched up only slightly among diabetics for both hepatitis A (9.3% to 15.4%) and hepatitis B (15.2% to 22.4%), according to National Health and Nutrition Examination Surveys (Hepatology 2011;54:1167-78).
Indeed, a show of hands revealed that less than a dozen of the roughly 500 attendees at the meeting had vaccinated their patients.
"I do think hepatitis B vaccination is important, but the question is: How do you know whether it works and how do you monitor it?" Dr. Parenti said.
A variety of factors can impair the immunogenicity of the hepatitis B vaccine, including gluteal or intradermal administration, increased age, higher body mass index, and genetics, such as human leukocyte antigen-DR3, which is present in about 95% of people with type 1 diabetes.
Unpublished data from the Centers for Disease Control and Prevention suggest that diabetes patients experience a similar drop in seroprotection with age (80% at 41-59 years, 65% at 60-69 years, and less than 40% at 70 years and older), he noted.
For patients who fail to respond to the first or second series of vaccinations, based on postimmunization serologic testing, using a higher 40 mcg-dose or four doses at 0, 1, 2, and 6 months has been shown to improve immunologic responses. There is no need, however, to restart a series if it was interrupted, regardless of the duration between vaccinations, Dr. Parenti said.
Only about 15% of individuals receive the herpes zoster vaccination, approved for persons aged 50 years and older, despite a third of the population expected to develop shingles within their lifetime. The risk is nearly doubled among diabetics under age 49 years with a hemoglobin A1c exceeding 8%, he said.
For those who do get vaccinated, however, a small Japanese study (J. Infect. 2013;67:215-9) reported similar humoral and cellular responses 3 and 6 months postvaccination among patients aged 60-70 years, with and without diabetes, he noted.
One attendee said that Medicare often refuses to cover herpes zoster and hepatitis vaccinations, leading her to remark, "It’s kind of turning into a class issue. My patients with money will get the vaccine."
It’s not just Medicare, but a significant number of other insurers who aren’t paying for the vaccines, particularly the pricey $300-$400 zoster vaccine, Dr. Parenti replied. Much depends on the patient’s plan. For example, the shingles vaccine is covered by Medicare Part D, but not Part A or Part B, while Medicare Part B covers recipients at "high or medium risk" for hepatitis B, including those with diabetes or end-stage renal disease. At this point, prior authorization is needed for all insurers, including Medicare, and appeals may be necessary, he said.
Dr. Parenti reported serving on a data safety monitoring board for the National Institute of Allergy and Infectious Diseases and receiving research support from Merck, Pfizer, Cubist Pharmaceuticals, Janssen Pharmaceuticals, Insight, and the National Institutes of Health.
SAN FRANCISCO – Patients with diabetes have an increased incidence for a number of infections, but vaccine-preventable diseases need not be among them.
"In the risk/benefit category, there’s a lot of potential benefit to these" immunizations, Dr. David Parenti said at the annual advanced postgraduate course held by the American Diabetes Association.
Studies have shown that patients with diabetes who are aged 23-59 years have a twofold higher relative risk of acute hepatitis B virus, compared with those without diabetes. In addition, the diabetic case fatality rate is more than double, at about 5%.
Individual glucose monitoring has reduced the transmission of hepatitis B virus by contaminated medical equipment, but outbreaks still occur in a variety of settings because of lapses in infection control, said Dr. Parenti, professor of medicine at George Washington University, Washington.
Still, vaccination rates are no better among patients with diabetes than those without. From 1999-2004 to 2005-2008, vaccination rates inched up only slightly among diabetics for both hepatitis A (9.3% to 15.4%) and hepatitis B (15.2% to 22.4%), according to National Health and Nutrition Examination Surveys (Hepatology 2011;54:1167-78).
Indeed, a show of hands revealed that less than a dozen of the roughly 500 attendees at the meeting had vaccinated their patients.
"I do think hepatitis B vaccination is important, but the question is: How do you know whether it works and how do you monitor it?" Dr. Parenti said.
A variety of factors can impair the immunogenicity of the hepatitis B vaccine, including gluteal or intradermal administration, increased age, higher body mass index, and genetics, such as human leukocyte antigen-DR3, which is present in about 95% of people with type 1 diabetes.
Unpublished data from the Centers for Disease Control and Prevention suggest that diabetes patients experience a similar drop in seroprotection with age (80% at 41-59 years, 65% at 60-69 years, and less than 40% at 70 years and older), he noted.
For patients who fail to respond to the first or second series of vaccinations, based on postimmunization serologic testing, using a higher 40 mcg-dose or four doses at 0, 1, 2, and 6 months has been shown to improve immunologic responses. There is no need, however, to restart a series if it was interrupted, regardless of the duration between vaccinations, Dr. Parenti said.
Only about 15% of individuals receive the herpes zoster vaccination, approved for persons aged 50 years and older, despite a third of the population expected to develop shingles within their lifetime. The risk is nearly doubled among diabetics under age 49 years with a hemoglobin A1c exceeding 8%, he said.
For those who do get vaccinated, however, a small Japanese study (J. Infect. 2013;67:215-9) reported similar humoral and cellular responses 3 and 6 months postvaccination among patients aged 60-70 years, with and without diabetes, he noted.
One attendee said that Medicare often refuses to cover herpes zoster and hepatitis vaccinations, leading her to remark, "It’s kind of turning into a class issue. My patients with money will get the vaccine."
It’s not just Medicare, but a significant number of other insurers who aren’t paying for the vaccines, particularly the pricey $300-$400 zoster vaccine, Dr. Parenti replied. Much depends on the patient’s plan. For example, the shingles vaccine is covered by Medicare Part D, but not Part A or Part B, while Medicare Part B covers recipients at "high or medium risk" for hepatitis B, including those with diabetes or end-stage renal disease. At this point, prior authorization is needed for all insurers, including Medicare, and appeals may be necessary, he said.
Dr. Parenti reported serving on a data safety monitoring board for the National Institute of Allergy and Infectious Diseases and receiving research support from Merck, Pfizer, Cubist Pharmaceuticals, Janssen Pharmaceuticals, Insight, and the National Institutes of Health.
EXPERT ANALYSIS FROM THE ADA ADVANCED POSTGRADUATE COURSE
Curb vaccine-preventable diseases in diabetes patients
SAN FRANCISCO – Patients with diabetes have an increased incidence for a number of infections, but vaccine-preventable diseases need not be among them.
"In the risk/benefit category, there’s a lot of potential benefit to these" immunizations, Dr. David Parenti said at the annual advanced postgraduate course held by the American Diabetes Association.
Studies have shown that patients with diabetes who are aged 23-59 years have a twofold higher relative risk of acute hepatitis B virus, compared with those without diabetes. In addition, the diabetic case fatality rate is more than double, at about 5%.
Individual glucose monitoring has reduced the transmission of hepatitis B virus by contaminated medical equipment, but outbreaks still occur in a variety of settings because of lapses in infection control, said Dr. Parenti, professor of medicine at George Washington University, Washington.
Still, vaccination rates are no better among patients with diabetes than those without. From 1999-2004 to 2005-2008, vaccination rates inched up only slightly among diabetics for both hepatitis A (9.3% to 15.4%) and hepatitis B (15.2% to 22.4%), according to National Health and Nutrition Examination Surveys (Hepatology 2011;54:1167-78).
Indeed, a show of hands revealed that less than a dozen of the roughly 500 attendees at the meeting had vaccinated their patients.
"I do think hepatitis B vaccination is important, but the question is: How do you know whether it works and how do you monitor it?" Dr. Parenti said.
A variety of factors can impair the immunogenicity of the hepatitis B vaccine, including gluteal or intradermal administration, increased age, higher body mass index, and genetics, such as human leukocyte antigen-DR3, which is present in about 95% of people with type 1 diabetes.
Unpublished data from the Centers for Disease Control and Prevention suggest that diabetes patients experience a similar drop in seroprotection with age (80% at 41-59 years, 65% at 60-69 years, and less than 40% at 70 years and older), he noted.
For patients who fail to respond to the first or second series of vaccinations, based on postimmunization serologic testing, using a higher 40 mcg-dose or four doses at 0, 1, 2, and 6 months has been shown to improve immunologic responses. There is no need, however, to restart a series if it was interrupted, regardless of the duration between vaccinations, Dr. Parenti said.
Only about 15% of individuals receive the herpes zoster vaccination, approved for persons aged 50 years and older, despite a third of the population expected to develop shingles within their lifetime. The risk is nearly doubled among diabetics under age 49 years with a hemoglobin A1c exceeding 8%, he said.
For those who do get vaccinated, however, a small Japanese study (J. Infect. 2013;67:215-9) reported similar humoral and cellular responses 3 and 6 months postvaccination among patients aged 60-70 years, with and without diabetes, he noted.
One attendee said that Medicare often refuses to cover herpes zoster and hepatitis vaccinations, leading her to remark, "It’s kind of turning into a class issue. My patients with money will get the vaccine."
It’s not just Medicare, but a significant number of other insurers who aren’t paying for the vaccines, particularly the pricey $300-$400 zoster vaccine, Dr. Parenti replied. Much depends on the patient’s plan. For example, the shingles vaccine is covered by Medicare Part D, but not Part A or Part B, while Medicare Part B covers recipients at "high or medium risk" for hepatitis B, including those with diabetes or end-stage renal disease. At this point, prior authorization is needed for all insurers, including Medicare, and appeals may be necessary, he said.
Dr. Parenti reported serving on a data safety monitoring board for the National Institute of Allergy and Infectious Diseases and receiving research support from Merck, Pfizer, Cubist Pharmaceuticals, Janssen Pharmaceuticals, Insight, and the National Institutes of Health.
SAN FRANCISCO – Patients with diabetes have an increased incidence for a number of infections, but vaccine-preventable diseases need not be among them.
"In the risk/benefit category, there’s a lot of potential benefit to these" immunizations, Dr. David Parenti said at the annual advanced postgraduate course held by the American Diabetes Association.
Studies have shown that patients with diabetes who are aged 23-59 years have a twofold higher relative risk of acute hepatitis B virus, compared with those without diabetes. In addition, the diabetic case fatality rate is more than double, at about 5%.
Individual glucose monitoring has reduced the transmission of hepatitis B virus by contaminated medical equipment, but outbreaks still occur in a variety of settings because of lapses in infection control, said Dr. Parenti, professor of medicine at George Washington University, Washington.
Still, vaccination rates are no better among patients with diabetes than those without. From 1999-2004 to 2005-2008, vaccination rates inched up only slightly among diabetics for both hepatitis A (9.3% to 15.4%) and hepatitis B (15.2% to 22.4%), according to National Health and Nutrition Examination Surveys (Hepatology 2011;54:1167-78).
Indeed, a show of hands revealed that less than a dozen of the roughly 500 attendees at the meeting had vaccinated their patients.
"I do think hepatitis B vaccination is important, but the question is: How do you know whether it works and how do you monitor it?" Dr. Parenti said.
A variety of factors can impair the immunogenicity of the hepatitis B vaccine, including gluteal or intradermal administration, increased age, higher body mass index, and genetics, such as human leukocyte antigen-DR3, which is present in about 95% of people with type 1 diabetes.
Unpublished data from the Centers for Disease Control and Prevention suggest that diabetes patients experience a similar drop in seroprotection with age (80% at 41-59 years, 65% at 60-69 years, and less than 40% at 70 years and older), he noted.
For patients who fail to respond to the first or second series of vaccinations, based on postimmunization serologic testing, using a higher 40 mcg-dose or four doses at 0, 1, 2, and 6 months has been shown to improve immunologic responses. There is no need, however, to restart a series if it was interrupted, regardless of the duration between vaccinations, Dr. Parenti said.
Only about 15% of individuals receive the herpes zoster vaccination, approved for persons aged 50 years and older, despite a third of the population expected to develop shingles within their lifetime. The risk is nearly doubled among diabetics under age 49 years with a hemoglobin A1c exceeding 8%, he said.
For those who do get vaccinated, however, a small Japanese study (J. Infect. 2013;67:215-9) reported similar humoral and cellular responses 3 and 6 months postvaccination among patients aged 60-70 years, with and without diabetes, he noted.
One attendee said that Medicare often refuses to cover herpes zoster and hepatitis vaccinations, leading her to remark, "It’s kind of turning into a class issue. My patients with money will get the vaccine."
It’s not just Medicare, but a significant number of other insurers who aren’t paying for the vaccines, particularly the pricey $300-$400 zoster vaccine, Dr. Parenti replied. Much depends on the patient’s plan. For example, the shingles vaccine is covered by Medicare Part D, but not Part A or Part B, while Medicare Part B covers recipients at "high or medium risk" for hepatitis B, including those with diabetes or end-stage renal disease. At this point, prior authorization is needed for all insurers, including Medicare, and appeals may be necessary, he said.
Dr. Parenti reported serving on a data safety monitoring board for the National Institute of Allergy and Infectious Diseases and receiving research support from Merck, Pfizer, Cubist Pharmaceuticals, Janssen Pharmaceuticals, Insight, and the National Institutes of Health.
SAN FRANCISCO – Patients with diabetes have an increased incidence for a number of infections, but vaccine-preventable diseases need not be among them.
"In the risk/benefit category, there’s a lot of potential benefit to these" immunizations, Dr. David Parenti said at the annual advanced postgraduate course held by the American Diabetes Association.
Studies have shown that patients with diabetes who are aged 23-59 years have a twofold higher relative risk of acute hepatitis B virus, compared with those without diabetes. In addition, the diabetic case fatality rate is more than double, at about 5%.
Individual glucose monitoring has reduced the transmission of hepatitis B virus by contaminated medical equipment, but outbreaks still occur in a variety of settings because of lapses in infection control, said Dr. Parenti, professor of medicine at George Washington University, Washington.
Still, vaccination rates are no better among patients with diabetes than those without. From 1999-2004 to 2005-2008, vaccination rates inched up only slightly among diabetics for both hepatitis A (9.3% to 15.4%) and hepatitis B (15.2% to 22.4%), according to National Health and Nutrition Examination Surveys (Hepatology 2011;54:1167-78).
Indeed, a show of hands revealed that less than a dozen of the roughly 500 attendees at the meeting had vaccinated their patients.
"I do think hepatitis B vaccination is important, but the question is: How do you know whether it works and how do you monitor it?" Dr. Parenti said.
A variety of factors can impair the immunogenicity of the hepatitis B vaccine, including gluteal or intradermal administration, increased age, higher body mass index, and genetics, such as human leukocyte antigen-DR3, which is present in about 95% of people with type 1 diabetes.
Unpublished data from the Centers for Disease Control and Prevention suggest that diabetes patients experience a similar drop in seroprotection with age (80% at 41-59 years, 65% at 60-69 years, and less than 40% at 70 years and older), he noted.
For patients who fail to respond to the first or second series of vaccinations, based on postimmunization serologic testing, using a higher 40 mcg-dose or four doses at 0, 1, 2, and 6 months has been shown to improve immunologic responses. There is no need, however, to restart a series if it was interrupted, regardless of the duration between vaccinations, Dr. Parenti said.
Only about 15% of individuals receive the herpes zoster vaccination, approved for persons aged 50 years and older, despite a third of the population expected to develop shingles within their lifetime. The risk is nearly doubled among diabetics under age 49 years with a hemoglobin A1c exceeding 8%, he said.
For those who do get vaccinated, however, a small Japanese study (J. Infect. 2013;67:215-9) reported similar humoral and cellular responses 3 and 6 months postvaccination among patients aged 60-70 years, with and without diabetes, he noted.
One attendee said that Medicare often refuses to cover herpes zoster and hepatitis vaccinations, leading her to remark, "It’s kind of turning into a class issue. My patients with money will get the vaccine."
It’s not just Medicare, but a significant number of other insurers who aren’t paying for the vaccines, particularly the pricey $300-$400 zoster vaccine, Dr. Parenti replied. Much depends on the patient’s plan. For example, the shingles vaccine is covered by Medicare Part D, but not Part A or Part B, while Medicare Part B covers recipients at "high or medium risk" for hepatitis B, including those with diabetes or end-stage renal disease. At this point, prior authorization is needed for all insurers, including Medicare, and appeals may be necessary, he said.
Dr. Parenti reported serving on a data safety monitoring board for the National Institute of Allergy and Infectious Diseases and receiving research support from Merck, Pfizer, Cubist Pharmaceuticals, Janssen Pharmaceuticals, Insight, and the National Institutes of Health.
EXPERT ANALYSIS FROM THE ADA ADVANCED POSTGRADUATE COURSE