User login
Survival Better in Women Post TAVI
PARIS – Although female sex is a risk factor for worse outcomes after conventional cardiac surgery, the opposite appears to be true following transcatheter aortic valve implantation.
Among 260 consecutive patients undergoing TAVI for severe, symptomatic aortic stenosis, female sex was associated with significantly better 1-year survival (76% vs. 65%).
The study, described as the first analysis of sex difference with this emerging technique, also identified female sex as an independent predictor of long-term survival, lead author Dr. Kentaro Hayashida and his colleagues reported at the annual congress of the European Society of Cardiology.
The increased survival rate among women who were treated with TAVI may represent a paradox in the cardiovascular disease gender gap.
"Surgical aortic valve replacement in female patients is technically demanding because of their smaller stature and body surface area, higher body mass index, and smaller aortic root," Dr. Hayashida of the Institut Cardiovasculaire Paris-Sud (ICPS) in Massy, France, said in an interview. "In our study cohort, TAVI was performed with a similar device success rate, compared to males, because of the procedural feasibility inherent in this novel technique, despite the specific characteristics associated" with female sex.
TAVI was successfully achieved in 91% of women and 88.4% of men, a nonsignificant difference. Similarly, no significant sex differences were observed for 30-day mortality (12% for women and 18% for men).
Longer life expectancy and early detection of aortic stenosis in women may, in part, have contributed to their improved survival, coauthor and colleague Dr. Philippe Garot said. He pointed out that despite both sexes having made gains in cardiovascular disease mortality from 1950 to 1999, an 80-year-old man in France can expect to live 8.3 more years, compared with 10.5 additional years for an 80-year-old French woman.
The average age at the time of surgery was 83 years in both study groups.
At baseline, women had a significantly lower logistic EuroSCORE (European System for Cardiac Operative Risk Evaluation) (22% vs. 26%), higher left ventricular ejection fraction (54% vs. 47%) and less coronary artery disease (49% vs. 79%), peripheral artery disease (27% vs. 40%) and previous cardiac surgery (14% vs. 26.4%) than did men.
Women, however, also had a significantly smaller femoral artery size (7.74 mm vs. 8.55 mm), annulus size (20.9 mm vs. 22.9 mm) and valve size (23.9 mm vs. 26.3 mm) than did men, Dr. Garot said.
TAVI is emerging as the standard of care for patients with severe aortic stenosis who cannot undergo surgery, after the procedure demonstrated similar 1-year survival rates as did conventional aortic valve replacement among older, high-risk patients in the recent PARTNER (Placement of Aortic Transcatheter Valve) trial (N. Engl. J. Med. 2010;363;1597-607). A preliminary subgroup analysis suggested a benefit with the less-invasive TAVR among women and in those without prior coronary artery bypass surgery.
In a multivariate analysis of the current cohort, male sex was identified as an independent predictor of long-term mortality (odds ratio, 1.80), the authors reported. Other significant risk factors were previous cardiac surgery (OR, 2.3), postprocedural aortic regurgitation (OR, 2.3), transfusion of four or more units (OR, 2.5), acute kidney injury (OR, 6.9), and conversion to open surgery (OR, 5.1).
Notably, vascular complications were not associated with mortality in the study, Dr. Garot said.
Based on the current results, Dr. Hayashida said it seems too early to conclude that TAVI should be routinely recommended in women who are not at high risk for conventional surgery.
"After confirmation by other studies of larger patient populations in the future, maybe we can say ‘yes,’ " he added.
Data were prospectively collected on 131 women and 129 men with severe aortic stenosis who were treated at the ICPS from September 2006 through December 2010. TAVI was performed using the Edwards Sapien or Sapien XT valves (85%) or the third-generation CoreValve Revalving system (15%), with 65% of valves placed via the transfemoral approach.
Dr. Hayashida and his coauthors report no conflicts.
PARIS – Although female sex is a risk factor for worse outcomes after conventional cardiac surgery, the opposite appears to be true following transcatheter aortic valve implantation.
Among 260 consecutive patients undergoing TAVI for severe, symptomatic aortic stenosis, female sex was associated with significantly better 1-year survival (76% vs. 65%).
The study, described as the first analysis of sex difference with this emerging technique, also identified female sex as an independent predictor of long-term survival, lead author Dr. Kentaro Hayashida and his colleagues reported at the annual congress of the European Society of Cardiology.
The increased survival rate among women who were treated with TAVI may represent a paradox in the cardiovascular disease gender gap.
"Surgical aortic valve replacement in female patients is technically demanding because of their smaller stature and body surface area, higher body mass index, and smaller aortic root," Dr. Hayashida of the Institut Cardiovasculaire Paris-Sud (ICPS) in Massy, France, said in an interview. "In our study cohort, TAVI was performed with a similar device success rate, compared to males, because of the procedural feasibility inherent in this novel technique, despite the specific characteristics associated" with female sex.
TAVI was successfully achieved in 91% of women and 88.4% of men, a nonsignificant difference. Similarly, no significant sex differences were observed for 30-day mortality (12% for women and 18% for men).
Longer life expectancy and early detection of aortic stenosis in women may, in part, have contributed to their improved survival, coauthor and colleague Dr. Philippe Garot said. He pointed out that despite both sexes having made gains in cardiovascular disease mortality from 1950 to 1999, an 80-year-old man in France can expect to live 8.3 more years, compared with 10.5 additional years for an 80-year-old French woman.
The average age at the time of surgery was 83 years in both study groups.
At baseline, women had a significantly lower logistic EuroSCORE (European System for Cardiac Operative Risk Evaluation) (22% vs. 26%), higher left ventricular ejection fraction (54% vs. 47%) and less coronary artery disease (49% vs. 79%), peripheral artery disease (27% vs. 40%) and previous cardiac surgery (14% vs. 26.4%) than did men.
Women, however, also had a significantly smaller femoral artery size (7.74 mm vs. 8.55 mm), annulus size (20.9 mm vs. 22.9 mm) and valve size (23.9 mm vs. 26.3 mm) than did men, Dr. Garot said.
TAVI is emerging as the standard of care for patients with severe aortic stenosis who cannot undergo surgery, after the procedure demonstrated similar 1-year survival rates as did conventional aortic valve replacement among older, high-risk patients in the recent PARTNER (Placement of Aortic Transcatheter Valve) trial (N. Engl. J. Med. 2010;363;1597-607). A preliminary subgroup analysis suggested a benefit with the less-invasive TAVR among women and in those without prior coronary artery bypass surgery.
In a multivariate analysis of the current cohort, male sex was identified as an independent predictor of long-term mortality (odds ratio, 1.80), the authors reported. Other significant risk factors were previous cardiac surgery (OR, 2.3), postprocedural aortic regurgitation (OR, 2.3), transfusion of four or more units (OR, 2.5), acute kidney injury (OR, 6.9), and conversion to open surgery (OR, 5.1).
Notably, vascular complications were not associated with mortality in the study, Dr. Garot said.
Based on the current results, Dr. Hayashida said it seems too early to conclude that TAVI should be routinely recommended in women who are not at high risk for conventional surgery.
"After confirmation by other studies of larger patient populations in the future, maybe we can say ‘yes,’ " he added.
Data were prospectively collected on 131 women and 129 men with severe aortic stenosis who were treated at the ICPS from September 2006 through December 2010. TAVI was performed using the Edwards Sapien or Sapien XT valves (85%) or the third-generation CoreValve Revalving system (15%), with 65% of valves placed via the transfemoral approach.
Dr. Hayashida and his coauthors report no conflicts.
PARIS – Although female sex is a risk factor for worse outcomes after conventional cardiac surgery, the opposite appears to be true following transcatheter aortic valve implantation.
Among 260 consecutive patients undergoing TAVI for severe, symptomatic aortic stenosis, female sex was associated with significantly better 1-year survival (76% vs. 65%).
The study, described as the first analysis of sex difference with this emerging technique, also identified female sex as an independent predictor of long-term survival, lead author Dr. Kentaro Hayashida and his colleagues reported at the annual congress of the European Society of Cardiology.
The increased survival rate among women who were treated with TAVI may represent a paradox in the cardiovascular disease gender gap.
"Surgical aortic valve replacement in female patients is technically demanding because of their smaller stature and body surface area, higher body mass index, and smaller aortic root," Dr. Hayashida of the Institut Cardiovasculaire Paris-Sud (ICPS) in Massy, France, said in an interview. "In our study cohort, TAVI was performed with a similar device success rate, compared to males, because of the procedural feasibility inherent in this novel technique, despite the specific characteristics associated" with female sex.
TAVI was successfully achieved in 91% of women and 88.4% of men, a nonsignificant difference. Similarly, no significant sex differences were observed for 30-day mortality (12% for women and 18% for men).
Longer life expectancy and early detection of aortic stenosis in women may, in part, have contributed to their improved survival, coauthor and colleague Dr. Philippe Garot said. He pointed out that despite both sexes having made gains in cardiovascular disease mortality from 1950 to 1999, an 80-year-old man in France can expect to live 8.3 more years, compared with 10.5 additional years for an 80-year-old French woman.
The average age at the time of surgery was 83 years in both study groups.
At baseline, women had a significantly lower logistic EuroSCORE (European System for Cardiac Operative Risk Evaluation) (22% vs. 26%), higher left ventricular ejection fraction (54% vs. 47%) and less coronary artery disease (49% vs. 79%), peripheral artery disease (27% vs. 40%) and previous cardiac surgery (14% vs. 26.4%) than did men.
Women, however, also had a significantly smaller femoral artery size (7.74 mm vs. 8.55 mm), annulus size (20.9 mm vs. 22.9 mm) and valve size (23.9 mm vs. 26.3 mm) than did men, Dr. Garot said.
TAVI is emerging as the standard of care for patients with severe aortic stenosis who cannot undergo surgery, after the procedure demonstrated similar 1-year survival rates as did conventional aortic valve replacement among older, high-risk patients in the recent PARTNER (Placement of Aortic Transcatheter Valve) trial (N. Engl. J. Med. 2010;363;1597-607). A preliminary subgroup analysis suggested a benefit with the less-invasive TAVR among women and in those without prior coronary artery bypass surgery.
In a multivariate analysis of the current cohort, male sex was identified as an independent predictor of long-term mortality (odds ratio, 1.80), the authors reported. Other significant risk factors were previous cardiac surgery (OR, 2.3), postprocedural aortic regurgitation (OR, 2.3), transfusion of four or more units (OR, 2.5), acute kidney injury (OR, 6.9), and conversion to open surgery (OR, 5.1).
Notably, vascular complications were not associated with mortality in the study, Dr. Garot said.
Based on the current results, Dr. Hayashida said it seems too early to conclude that TAVI should be routinely recommended in women who are not at high risk for conventional surgery.
"After confirmation by other studies of larger patient populations in the future, maybe we can say ‘yes,’ " he added.
Data were prospectively collected on 131 women and 129 men with severe aortic stenosis who were treated at the ICPS from September 2006 through December 2010. TAVI was performed using the Edwards Sapien or Sapien XT valves (85%) or the third-generation CoreValve Revalving system (15%), with 65% of valves placed via the transfemoral approach.
Dr. Hayashida and his coauthors report no conflicts.
FROM THE ANNUAL CONGRESS OF THE EUROPEAN SOCIETY OF CARDIOLOGY
Major Finding: The 1-year survival was 76% for women vs. 65% for men, a significant difference.
Data Source: A review of prospectively collected data on 131 women and 129 men who were treated with TAVI.
Disclosures: Dr. Hayashida and his coauthors report no conflicts.
Eplerenone Effective Across All High-Risk Heart Failure Patients
PARIS – The aldosterone antagonist eplerenone cut cardiovascular events and the need for hospitalization significantly across all risk levels in patients with mild heart failure, according to a subanalysis of the EMPHASIS-HF trial.
Eplerenone (Inspra) was also shown to trim troublesome and costly repeat heart failure hospitalizations in a subset of patients followed for up to 10 additional months after the pivotal, phase III trial closed, Dr. Bertram Pitt reported at the annual congress of the European Society of Cardiology.
"Overall efficacy, no matter where we looked, was about the same, and we had the same safety," he told reporters.
EMPHASIS-HF (Eplerenone in Mild Patients Hospitalization and Survival Study in Heart Failure) was stopped prematurely last spring after eplerenone in addition to standard therapy demonstrated a 37% (hazard ratio, 0.63) improvement over placebo in the primary end point of death from cardiovascular causes or heart failure hospitalization in 2,637 patients with mild New York Heart Association class II systolic heart failure (N. Engl. J. Med. 2011;364:11-21).
Among 1,597 patients who remained on double-blind therapy after study closure, the primary end point occurred in 21% on eplerenone and 29% on placebo (HR, 0.66, P value less than .0001), said Dr. Pitt, with the University of Michigan, Ann Arbor.
Repeat hospitalization for heart failure was significantly reduced with eplerenone (rate ratio, 0.62, P less than .001).
"This suggests, to us at least, that this is going to have important cost implications, quality of life implications, as well as important implications on survival, since we know that heart failure hospitalization relates to target-organ damage and survival," he said.
The benefits of eplerenone were particularly compelling in elderly patients and those with diabetes and renal dysfunction, three high-risk populations in whom clinicians are hesitant to use adjuvant aldosterone blockade because of fears of inducing hyperkalemia, Dr. Pitt said. He pointed out that recent head-to-head data show that the aldosterone antagonist spironolactone (Aldactone) raises hemoglobin A1C and cortisol levels and reduces adiponectin in patients with diabetes, whereas eplerenone does not.
When Dr. Pitt and colleagues looked at patients with diabetes in the subanalysis, the benefit was better than that observed in the overall EMPHASIS-HF cohort, with a 46% reduction in the primary end point (HR, 0.54, P value less than .0001).
Dr. Pitt noted that the current American Heart Association, American College of Cardiology and European guidelines for aldosterone blockade don’t specify a particular agent, but look at the agents as a class.
"We believe with [these] data, that in the next guidelines there should at least be some consideration for the specific use of eplerenone, at least in the subset of patients with diabetes," he said.
Among patients with an estimated glomerular filtration rate less than 60 mL per minute per 1.73 m2, the improvement in the primary outcome reached 38% with eplerenone over placebo (HR, 0.62, P = .0001).
The study excluded patients with a baseline serum potassium level above 5.0 mmol/L and estimated GFR below 30 mL per minute per 1.73 m2 in an attempt to minimize the risk of hyperkalemia. Despite this, the positive results remain applicable, Dr. Pitt said in an interview.
Among elderly patients at least 75 years old, the benefit on the primary end point with eplerenone reached 34% (HR, 0.66, P = .004).
Significant benefits were also observed in patients with a left ventricular ejection fraction less than 30% (HR, 0.65, P less than .0001) and with a median systolic blood pressure less than 123 mmHg (HR, 0.63, P less than .0001).
As seen in the overall EMPHASIS-HF cohort, the benefits of eplerenone were accompanied by a significant increase in the incidence of serum potassium more than 5.5 mmol/L in each of the high-risk subgroups.
There were, however, no significant increases in the incidence of serum potassium more than 6 mmol/L, hospitalization leading to treatment discontinuation, hospitalization for/or deaths due to hyperkalemia, or hospitalizations for worsening renal function, Dr. Pitt said.
Invited discussant Dr. Piotr Ponikowski, with the 4th Military Hospital in Wroclaw, Poland, highlighted as important the benefits of eplerenone in people with diabetes and the reduction in repeat hospitalizations. He noted that up to 50% of heart failure patients are rehospitalized and these visits are associated with significant costs and reduced quality of life and mortality. Still, aldosterone antagonists remain underutilized in heart failure patients in Europe as well as the United States.
Physicians frequently question whether clinical trial data are real, consistent, and clinically meaningful, he said. "With the presented data, we can now say that the answer to all these questions is ‘yes.’ "
Pfizer sponsored the trial. Dr. Pitt reports financial relationships with several firms excluding Pfizer. His coauthors report similar relationships including employment with Pfizer.
PARIS – The aldosterone antagonist eplerenone cut cardiovascular events and the need for hospitalization significantly across all risk levels in patients with mild heart failure, according to a subanalysis of the EMPHASIS-HF trial.
Eplerenone (Inspra) was also shown to trim troublesome and costly repeat heart failure hospitalizations in a subset of patients followed for up to 10 additional months after the pivotal, phase III trial closed, Dr. Bertram Pitt reported at the annual congress of the European Society of Cardiology.
"Overall efficacy, no matter where we looked, was about the same, and we had the same safety," he told reporters.
EMPHASIS-HF (Eplerenone in Mild Patients Hospitalization and Survival Study in Heart Failure) was stopped prematurely last spring after eplerenone in addition to standard therapy demonstrated a 37% (hazard ratio, 0.63) improvement over placebo in the primary end point of death from cardiovascular causes or heart failure hospitalization in 2,637 patients with mild New York Heart Association class II systolic heart failure (N. Engl. J. Med. 2011;364:11-21).
Among 1,597 patients who remained on double-blind therapy after study closure, the primary end point occurred in 21% on eplerenone and 29% on placebo (HR, 0.66, P value less than .0001), said Dr. Pitt, with the University of Michigan, Ann Arbor.
Repeat hospitalization for heart failure was significantly reduced with eplerenone (rate ratio, 0.62, P less than .001).
"This suggests, to us at least, that this is going to have important cost implications, quality of life implications, as well as important implications on survival, since we know that heart failure hospitalization relates to target-organ damage and survival," he said.
The benefits of eplerenone were particularly compelling in elderly patients and those with diabetes and renal dysfunction, three high-risk populations in whom clinicians are hesitant to use adjuvant aldosterone blockade because of fears of inducing hyperkalemia, Dr. Pitt said. He pointed out that recent head-to-head data show that the aldosterone antagonist spironolactone (Aldactone) raises hemoglobin A1C and cortisol levels and reduces adiponectin in patients with diabetes, whereas eplerenone does not.
When Dr. Pitt and colleagues looked at patients with diabetes in the subanalysis, the benefit was better than that observed in the overall EMPHASIS-HF cohort, with a 46% reduction in the primary end point (HR, 0.54, P value less than .0001).
Dr. Pitt noted that the current American Heart Association, American College of Cardiology and European guidelines for aldosterone blockade don’t specify a particular agent, but look at the agents as a class.
"We believe with [these] data, that in the next guidelines there should at least be some consideration for the specific use of eplerenone, at least in the subset of patients with diabetes," he said.
Among patients with an estimated glomerular filtration rate less than 60 mL per minute per 1.73 m2, the improvement in the primary outcome reached 38% with eplerenone over placebo (HR, 0.62, P = .0001).
The study excluded patients with a baseline serum potassium level above 5.0 mmol/L and estimated GFR below 30 mL per minute per 1.73 m2 in an attempt to minimize the risk of hyperkalemia. Despite this, the positive results remain applicable, Dr. Pitt said in an interview.
Among elderly patients at least 75 years old, the benefit on the primary end point with eplerenone reached 34% (HR, 0.66, P = .004).
Significant benefits were also observed in patients with a left ventricular ejection fraction less than 30% (HR, 0.65, P less than .0001) and with a median systolic blood pressure less than 123 mmHg (HR, 0.63, P less than .0001).
As seen in the overall EMPHASIS-HF cohort, the benefits of eplerenone were accompanied by a significant increase in the incidence of serum potassium more than 5.5 mmol/L in each of the high-risk subgroups.
There were, however, no significant increases in the incidence of serum potassium more than 6 mmol/L, hospitalization leading to treatment discontinuation, hospitalization for/or deaths due to hyperkalemia, or hospitalizations for worsening renal function, Dr. Pitt said.
Invited discussant Dr. Piotr Ponikowski, with the 4th Military Hospital in Wroclaw, Poland, highlighted as important the benefits of eplerenone in people with diabetes and the reduction in repeat hospitalizations. He noted that up to 50% of heart failure patients are rehospitalized and these visits are associated with significant costs and reduced quality of life and mortality. Still, aldosterone antagonists remain underutilized in heart failure patients in Europe as well as the United States.
Physicians frequently question whether clinical trial data are real, consistent, and clinically meaningful, he said. "With the presented data, we can now say that the answer to all these questions is ‘yes.’ "
Pfizer sponsored the trial. Dr. Pitt reports financial relationships with several firms excluding Pfizer. His coauthors report similar relationships including employment with Pfizer.
PARIS – The aldosterone antagonist eplerenone cut cardiovascular events and the need for hospitalization significantly across all risk levels in patients with mild heart failure, according to a subanalysis of the EMPHASIS-HF trial.
Eplerenone (Inspra) was also shown to trim troublesome and costly repeat heart failure hospitalizations in a subset of patients followed for up to 10 additional months after the pivotal, phase III trial closed, Dr. Bertram Pitt reported at the annual congress of the European Society of Cardiology.
"Overall efficacy, no matter where we looked, was about the same, and we had the same safety," he told reporters.
EMPHASIS-HF (Eplerenone in Mild Patients Hospitalization and Survival Study in Heart Failure) was stopped prematurely last spring after eplerenone in addition to standard therapy demonstrated a 37% (hazard ratio, 0.63) improvement over placebo in the primary end point of death from cardiovascular causes or heart failure hospitalization in 2,637 patients with mild New York Heart Association class II systolic heart failure (N. Engl. J. Med. 2011;364:11-21).
Among 1,597 patients who remained on double-blind therapy after study closure, the primary end point occurred in 21% on eplerenone and 29% on placebo (HR, 0.66, P value less than .0001), said Dr. Pitt, with the University of Michigan, Ann Arbor.
Repeat hospitalization for heart failure was significantly reduced with eplerenone (rate ratio, 0.62, P less than .001).
"This suggests, to us at least, that this is going to have important cost implications, quality of life implications, as well as important implications on survival, since we know that heart failure hospitalization relates to target-organ damage and survival," he said.
The benefits of eplerenone were particularly compelling in elderly patients and those with diabetes and renal dysfunction, three high-risk populations in whom clinicians are hesitant to use adjuvant aldosterone blockade because of fears of inducing hyperkalemia, Dr. Pitt said. He pointed out that recent head-to-head data show that the aldosterone antagonist spironolactone (Aldactone) raises hemoglobin A1C and cortisol levels and reduces adiponectin in patients with diabetes, whereas eplerenone does not.
When Dr. Pitt and colleagues looked at patients with diabetes in the subanalysis, the benefit was better than that observed in the overall EMPHASIS-HF cohort, with a 46% reduction in the primary end point (HR, 0.54, P value less than .0001).
Dr. Pitt noted that the current American Heart Association, American College of Cardiology and European guidelines for aldosterone blockade don’t specify a particular agent, but look at the agents as a class.
"We believe with [these] data, that in the next guidelines there should at least be some consideration for the specific use of eplerenone, at least in the subset of patients with diabetes," he said.
Among patients with an estimated glomerular filtration rate less than 60 mL per minute per 1.73 m2, the improvement in the primary outcome reached 38% with eplerenone over placebo (HR, 0.62, P = .0001).
The study excluded patients with a baseline serum potassium level above 5.0 mmol/L and estimated GFR below 30 mL per minute per 1.73 m2 in an attempt to minimize the risk of hyperkalemia. Despite this, the positive results remain applicable, Dr. Pitt said in an interview.
Among elderly patients at least 75 years old, the benefit on the primary end point with eplerenone reached 34% (HR, 0.66, P = .004).
Significant benefits were also observed in patients with a left ventricular ejection fraction less than 30% (HR, 0.65, P less than .0001) and with a median systolic blood pressure less than 123 mmHg (HR, 0.63, P less than .0001).
As seen in the overall EMPHASIS-HF cohort, the benefits of eplerenone were accompanied by a significant increase in the incidence of serum potassium more than 5.5 mmol/L in each of the high-risk subgroups.
There were, however, no significant increases in the incidence of serum potassium more than 6 mmol/L, hospitalization leading to treatment discontinuation, hospitalization for/or deaths due to hyperkalemia, or hospitalizations for worsening renal function, Dr. Pitt said.
Invited discussant Dr. Piotr Ponikowski, with the 4th Military Hospital in Wroclaw, Poland, highlighted as important the benefits of eplerenone in people with diabetes and the reduction in repeat hospitalizations. He noted that up to 50% of heart failure patients are rehospitalized and these visits are associated with significant costs and reduced quality of life and mortality. Still, aldosterone antagonists remain underutilized in heart failure patients in Europe as well as the United States.
Physicians frequently question whether clinical trial data are real, consistent, and clinically meaningful, he said. "With the presented data, we can now say that the answer to all these questions is ‘yes.’ "
Pfizer sponsored the trial. Dr. Pitt reports financial relationships with several firms excluding Pfizer. His coauthors report similar relationships including employment with Pfizer.
FROM THE ANNUAL CONGRESS OF THE EUROPEAN SOCIETY OF CARDIOLOGY
Major Finding: Among patients with diabetes who took eplerenone, there was a 46% reduction in the primary end point of death from cardiovascular causes or heart failure hospitalization, compared with those who took placebo.
Data Source: Subanalysis of high-risk groups and follow-up analyses of the phase III EMPHASIS-HF trial.
Disclosures: Pfizer sponsored the trial. Dr. Pitt reports financial relationships with several firms excluding Pfizer. His coauthors report similar relationships including employment with Pfizer.
Dalcetrapib Boosts HDL Without Bumping BP
PARIS – The investigational cholesteryl ester transfer protein inhibitor dalcetrapib increased HDL cholesterol without the toxic effects that have raised safety concerns over the use of CETP inhibitors as a cardiovascular therapy.
Dalcetrapib reduced CETP activity by 49% and increased HDL cholesterol by 31% without affecting LDL cholesterol among patients with or at risk of coronary heart disease in the randomized phase II dal-VESSEL trial.
Dalcetrapib did not cause endothelial dysfunction nor did it improve it. Unlike the CETP inhibitor torcetrapib, dalcetrapib did not raise blood pressure, providing further reassurance regarding the safety of the compound, Dr. Thomas Lüscher said at the annual congress of the European Society of Cardiology.
"I think we can say it’s safe and has no untoward effects like the others, and it does the job as far as the lipid profile is concerned," he told reporters. "It’s a bit less potent than torcetrapib, which increased HDL by about 60% to 70%, but [it] may be that’s even an advantage. We’ll see."
Dr. Lüscher said it was a bit disappointing that dalcetrapib (manufactured by Hoffmann-La Roche) did not improve vascular function, but noted that it wasn’t worsened either.
Whether the novel CETP inhibitor will have an effect on cardiovascular events will be determined by the phase III dal-OUTCOMES trial, with results expected sometime in 2013, said Dr. Lüscher, professor and chair of cardiology at University Hospital Zurich, Switzerland.
A total of 15,872 patients with stable heart disease following a recent acute coronary event have been enrolled, according to the drug maker’s Web site.
Invited discussant Dr. Keith A. A. Fox, with the University of Edinburgh/British Heart Foundation Centre for Cardiovascular Science, said the positive dal-VESSEL data represent a "clear advance," but urged caution because of the limited size of the trial.
"We need to know whether the modest changes in blood pressure are real or have an impact on outcomes," he said.
The recent DEFINE trial of Merck’s CETP inhibitor anacetrapib, however, illustrates the very real difficulties in achieving this goal. Anacetrapib raised HDL cholesterol 138% and reduced LDL cholesterol by 40% compared with placebo, but did not reduce cardiovascular events in 1,623 patients with or at high risk for coronary heart disease (N. Engl. J. Med. 2010;363:2406-15).
Torcetrapib, the first CTEP to be tested, increased HDL cholesterol by 61% and decreased LDL cholesterol by 20%, but was associated with a roughly 4.6 mm Hg increase in blood pressure (N. Engl. J. Med. 2007;356:1304-16). Development of the agent came to a screeching halt, however, when the ILLUMINATE trial subsequently reported that torcetrapib also increased the risk of cardiovascular events by 25% and all-cause mortality by 58% in patients at high cardiovascular risk (N. Engl. J. Med. 2007:357:2109-22).
Further research suggested that the negative effects were caused not by reducing CTEP activity, but by off-target effects on renal glands, diminished nitrous oxide levels and increased endothelin-1, Dr. Lüscher explained.
The dal-VESSEL study was conducted at 19 centers in Europe and randomized 476 patients with coronary heart disease or CHD risk equivalents and HDL cholesterol of less than 50 mg/dL to dalcetrapib 600 mg/day or placebo plus their standard medication for a total of 36 weeks. Endothelial function was measured at 12 weeks using brachial flow-mediated dilation (FMD), a validated marker of endothelial dysfunction. Their mean age was 62 years, and roughly 97% were on statins.
The mean change in HDL cholesterol from baseline at week 4 was 2.7% for placebo and 27.5% for dalcetrapib; at week 36 it was -0.14% vs. 30.7%, respectively (P less than .0001 for both), Dr. Lüscher said.
At 36 weeks lipoprotein A-1 levels were significantly increased with dalcetrapib, but lipoprotein B levels were not affected.
There was no significant change with dalcetrapib in the co-primary safety endpoints of 24-hour ambulatory BP at week 4 and FMD at week 12.
Death from coronary heart disease occurred in one patient given placebo and none given dalcetrapib. Non-fatal myocardial infarctions were reported in 3 and 2 patients, respectively.
The trial was sponsored by Hoffmann-La Roche. Dr. Lüscher reports receiving research grants from Pfizer, Eli Lilly and Merck and consultancy or lecture fees from CSL, Merck, Pfizer and F. Hoffmann-La Roche.
PARIS – The investigational cholesteryl ester transfer protein inhibitor dalcetrapib increased HDL cholesterol without the toxic effects that have raised safety concerns over the use of CETP inhibitors as a cardiovascular therapy.
Dalcetrapib reduced CETP activity by 49% and increased HDL cholesterol by 31% without affecting LDL cholesterol among patients with or at risk of coronary heart disease in the randomized phase II dal-VESSEL trial.
Dalcetrapib did not cause endothelial dysfunction nor did it improve it. Unlike the CETP inhibitor torcetrapib, dalcetrapib did not raise blood pressure, providing further reassurance regarding the safety of the compound, Dr. Thomas Lüscher said at the annual congress of the European Society of Cardiology.
"I think we can say it’s safe and has no untoward effects like the others, and it does the job as far as the lipid profile is concerned," he told reporters. "It’s a bit less potent than torcetrapib, which increased HDL by about 60% to 70%, but [it] may be that’s even an advantage. We’ll see."
Dr. Lüscher said it was a bit disappointing that dalcetrapib (manufactured by Hoffmann-La Roche) did not improve vascular function, but noted that it wasn’t worsened either.
Whether the novel CETP inhibitor will have an effect on cardiovascular events will be determined by the phase III dal-OUTCOMES trial, with results expected sometime in 2013, said Dr. Lüscher, professor and chair of cardiology at University Hospital Zurich, Switzerland.
A total of 15,872 patients with stable heart disease following a recent acute coronary event have been enrolled, according to the drug maker’s Web site.
Invited discussant Dr. Keith A. A. Fox, with the University of Edinburgh/British Heart Foundation Centre for Cardiovascular Science, said the positive dal-VESSEL data represent a "clear advance," but urged caution because of the limited size of the trial.
"We need to know whether the modest changes in blood pressure are real or have an impact on outcomes," he said.
The recent DEFINE trial of Merck’s CETP inhibitor anacetrapib, however, illustrates the very real difficulties in achieving this goal. Anacetrapib raised HDL cholesterol 138% and reduced LDL cholesterol by 40% compared with placebo, but did not reduce cardiovascular events in 1,623 patients with or at high risk for coronary heart disease (N. Engl. J. Med. 2010;363:2406-15).
Torcetrapib, the first CTEP to be tested, increased HDL cholesterol by 61% and decreased LDL cholesterol by 20%, but was associated with a roughly 4.6 mm Hg increase in blood pressure (N. Engl. J. Med. 2007;356:1304-16). Development of the agent came to a screeching halt, however, when the ILLUMINATE trial subsequently reported that torcetrapib also increased the risk of cardiovascular events by 25% and all-cause mortality by 58% in patients at high cardiovascular risk (N. Engl. J. Med. 2007:357:2109-22).
Further research suggested that the negative effects were caused not by reducing CTEP activity, but by off-target effects on renal glands, diminished nitrous oxide levels and increased endothelin-1, Dr. Lüscher explained.
The dal-VESSEL study was conducted at 19 centers in Europe and randomized 476 patients with coronary heart disease or CHD risk equivalents and HDL cholesterol of less than 50 mg/dL to dalcetrapib 600 mg/day or placebo plus their standard medication for a total of 36 weeks. Endothelial function was measured at 12 weeks using brachial flow-mediated dilation (FMD), a validated marker of endothelial dysfunction. Their mean age was 62 years, and roughly 97% were on statins.
The mean change in HDL cholesterol from baseline at week 4 was 2.7% for placebo and 27.5% for dalcetrapib; at week 36 it was -0.14% vs. 30.7%, respectively (P less than .0001 for both), Dr. Lüscher said.
At 36 weeks lipoprotein A-1 levels were significantly increased with dalcetrapib, but lipoprotein B levels were not affected.
There was no significant change with dalcetrapib in the co-primary safety endpoints of 24-hour ambulatory BP at week 4 and FMD at week 12.
Death from coronary heart disease occurred in one patient given placebo and none given dalcetrapib. Non-fatal myocardial infarctions were reported in 3 and 2 patients, respectively.
The trial was sponsored by Hoffmann-La Roche. Dr. Lüscher reports receiving research grants from Pfizer, Eli Lilly and Merck and consultancy or lecture fees from CSL, Merck, Pfizer and F. Hoffmann-La Roche.
PARIS – The investigational cholesteryl ester transfer protein inhibitor dalcetrapib increased HDL cholesterol without the toxic effects that have raised safety concerns over the use of CETP inhibitors as a cardiovascular therapy.
Dalcetrapib reduced CETP activity by 49% and increased HDL cholesterol by 31% without affecting LDL cholesterol among patients with or at risk of coronary heart disease in the randomized phase II dal-VESSEL trial.
Dalcetrapib did not cause endothelial dysfunction nor did it improve it. Unlike the CETP inhibitor torcetrapib, dalcetrapib did not raise blood pressure, providing further reassurance regarding the safety of the compound, Dr. Thomas Lüscher said at the annual congress of the European Society of Cardiology.
"I think we can say it’s safe and has no untoward effects like the others, and it does the job as far as the lipid profile is concerned," he told reporters. "It’s a bit less potent than torcetrapib, which increased HDL by about 60% to 70%, but [it] may be that’s even an advantage. We’ll see."
Dr. Lüscher said it was a bit disappointing that dalcetrapib (manufactured by Hoffmann-La Roche) did not improve vascular function, but noted that it wasn’t worsened either.
Whether the novel CETP inhibitor will have an effect on cardiovascular events will be determined by the phase III dal-OUTCOMES trial, with results expected sometime in 2013, said Dr. Lüscher, professor and chair of cardiology at University Hospital Zurich, Switzerland.
A total of 15,872 patients with stable heart disease following a recent acute coronary event have been enrolled, according to the drug maker’s Web site.
Invited discussant Dr. Keith A. A. Fox, with the University of Edinburgh/British Heart Foundation Centre for Cardiovascular Science, said the positive dal-VESSEL data represent a "clear advance," but urged caution because of the limited size of the trial.
"We need to know whether the modest changes in blood pressure are real or have an impact on outcomes," he said.
The recent DEFINE trial of Merck’s CETP inhibitor anacetrapib, however, illustrates the very real difficulties in achieving this goal. Anacetrapib raised HDL cholesterol 138% and reduced LDL cholesterol by 40% compared with placebo, but did not reduce cardiovascular events in 1,623 patients with or at high risk for coronary heart disease (N. Engl. J. Med. 2010;363:2406-15).
Torcetrapib, the first CTEP to be tested, increased HDL cholesterol by 61% and decreased LDL cholesterol by 20%, but was associated with a roughly 4.6 mm Hg increase in blood pressure (N. Engl. J. Med. 2007;356:1304-16). Development of the agent came to a screeching halt, however, when the ILLUMINATE trial subsequently reported that torcetrapib also increased the risk of cardiovascular events by 25% and all-cause mortality by 58% in patients at high cardiovascular risk (N. Engl. J. Med. 2007:357:2109-22).
Further research suggested that the negative effects were caused not by reducing CTEP activity, but by off-target effects on renal glands, diminished nitrous oxide levels and increased endothelin-1, Dr. Lüscher explained.
The dal-VESSEL study was conducted at 19 centers in Europe and randomized 476 patients with coronary heart disease or CHD risk equivalents and HDL cholesterol of less than 50 mg/dL to dalcetrapib 600 mg/day or placebo plus their standard medication for a total of 36 weeks. Endothelial function was measured at 12 weeks using brachial flow-mediated dilation (FMD), a validated marker of endothelial dysfunction. Their mean age was 62 years, and roughly 97% were on statins.
The mean change in HDL cholesterol from baseline at week 4 was 2.7% for placebo and 27.5% for dalcetrapib; at week 36 it was -0.14% vs. 30.7%, respectively (P less than .0001 for both), Dr. Lüscher said.
At 36 weeks lipoprotein A-1 levels were significantly increased with dalcetrapib, but lipoprotein B levels were not affected.
There was no significant change with dalcetrapib in the co-primary safety endpoints of 24-hour ambulatory BP at week 4 and FMD at week 12.
Death from coronary heart disease occurred in one patient given placebo and none given dalcetrapib. Non-fatal myocardial infarctions were reported in 3 and 2 patients, respectively.
The trial was sponsored by Hoffmann-La Roche. Dr. Lüscher reports receiving research grants from Pfizer, Eli Lilly and Merck and consultancy or lecture fees from CSL, Merck, Pfizer and F. Hoffmann-La Roche.
FROM THE ANNUAL CONGRESS OF THE EUROPEAN SOCIETY OF CARDIOLOGY
Major Finding: Dalcetrapib increased HDL cholesterol levels by 31%, compared with placebo.
Data Source: Phase IIb randomized trial in 476 patients with or at risk of coronary heart disease.
Disclosures: The trial was sponsored by F. Hoffmann-La Roche. Dr. Lüscher reports receiving research grants from Pfizer, Eli Lilly and Merck and consultancy or lecture fees from CSL, Merck, Pfizer and F. Hoffmann-La Roche.
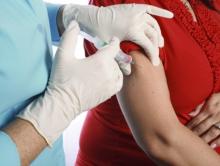
Pediatricians Push Tdap More Than Ob.Gyns.
CHICAGO – Pregnant women were significantly more likely to receive information on pertussis vaccination from their pediatrician than from their obstetrician in a survey of 314 women.
"Multiple opportunities exist for education of obstetricians and gynecologists to improve Tdap vaccination rates in the United States," Dr. Rachel Gutkin said at the annual meeting of the Infectious Diseases Society for Obstetrics and Gynecology.
She reported on 314 pregnant women presenting to an academic perinatal center between March and June 2011 who answered an anonymous, multiple-choice questionnaire regarding their knowledge and opinions on vaccination in general, and Tdap (tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis adsorbed) specifically.
Overall, 218 (69%) of the women had heard about the Tdap booster vaccine, with 76 (24%) women learning about it at their pediatrician’s office and 54 (17%) at their obstetrician’s office.
Just 8% of women learned about Tdap from the Internet, while 19% did so from friends or family, and 17% from TV or radio, said Dr. Gutkin, a resident in the department of obstetrics and gynecology at the University of California, Los Angeles. The remaining 15% learned of it from other sources.
The majority of respondents knew that pertussis is a significant health risk for children (76%) and newborns (86%), and 50% also thought it was a significant health risk for fetuses.
When asked whether they would receive a Tdap vaccination during pregnancy to protect their newborn from whooping cough, 11% said they would if their doctor recommended it, 12% said yes if they knew it was safe, and 66% said they would if both conditions were true. Additionally, 11% said they would not receive Tdap during pregnancy under any circumstances, she said.
Women who discussed Tdap with their obstetrician were nearly five times more likely to be vaccinated than those who did not (odds ratio, 4.93). Only 13% of women who did not discuss Tdap with their ob.gyn. were vaccinated.
Although women were significantly more likely to discuss Tdap with their pediatrician, women were nearly three times more likely to be vaccinated if they were counseled by their ob.gyn. versus their pediatrician (OR, 2.9), Dr. Gutkin reported.
The survey was conducted in California, which in 2010 experienced the largest outbreak of pertussis in 65 years, with 9,120 cases reported, including 10 deaths. Cases peaked in some parts of California in the summer of 2010, but public health officials anticipate that numbers will remain high in 2011.
The respondents were well-educated, with 54% completing college and 32% graduate school. Most had PPO (Preferred Provider Organizations) insurance (91%), and the majority were aged 25-44 years (95%).
Still, almost one-quarter of women were not sure if vaccines in general are safe (22%). Roughly two-thirds were not sure if vaccines are safe in pregnancy (68%) and a full 10% were not sure if vaccines are effective in preventing illness, Dr. Gutkin said.
Women were significantly more likely to have discussed the influenza vaccine with their ob.gyn. than Tdap (47% vs. 19%), and to be vaccinated for influenza than pertussis (42% vs. 18%).
Of note, women were three times more likely to receive Tdap if they had received a flu shot (OR, 3.68).
"Discussion with their ob.gyn. was a significant factor in the acceptance of Tdap and influenza vaccines during pregnancy," she said.
Dr. Gutkin and her colleagues reported no relevant financial disclosures.
CHICAGO – Pregnant women were significantly more likely to receive information on pertussis vaccination from their pediatrician than from their obstetrician in a survey of 314 women.
"Multiple opportunities exist for education of obstetricians and gynecologists to improve Tdap vaccination rates in the United States," Dr. Rachel Gutkin said at the annual meeting of the Infectious Diseases Society for Obstetrics and Gynecology.
She reported on 314 pregnant women presenting to an academic perinatal center between March and June 2011 who answered an anonymous, multiple-choice questionnaire regarding their knowledge and opinions on vaccination in general, and Tdap (tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis adsorbed) specifically.
Overall, 218 (69%) of the women had heard about the Tdap booster vaccine, with 76 (24%) women learning about it at their pediatrician’s office and 54 (17%) at their obstetrician’s office.
Just 8% of women learned about Tdap from the Internet, while 19% did so from friends or family, and 17% from TV or radio, said Dr. Gutkin, a resident in the department of obstetrics and gynecology at the University of California, Los Angeles. The remaining 15% learned of it from other sources.
The majority of respondents knew that pertussis is a significant health risk for children (76%) and newborns (86%), and 50% also thought it was a significant health risk for fetuses.
When asked whether they would receive a Tdap vaccination during pregnancy to protect their newborn from whooping cough, 11% said they would if their doctor recommended it, 12% said yes if they knew it was safe, and 66% said they would if both conditions were true. Additionally, 11% said they would not receive Tdap during pregnancy under any circumstances, she said.
Women who discussed Tdap with their obstetrician were nearly five times more likely to be vaccinated than those who did not (odds ratio, 4.93). Only 13% of women who did not discuss Tdap with their ob.gyn. were vaccinated.
Although women were significantly more likely to discuss Tdap with their pediatrician, women were nearly three times more likely to be vaccinated if they were counseled by their ob.gyn. versus their pediatrician (OR, 2.9), Dr. Gutkin reported.
The survey was conducted in California, which in 2010 experienced the largest outbreak of pertussis in 65 years, with 9,120 cases reported, including 10 deaths. Cases peaked in some parts of California in the summer of 2010, but public health officials anticipate that numbers will remain high in 2011.
The respondents were well-educated, with 54% completing college and 32% graduate school. Most had PPO (Preferred Provider Organizations) insurance (91%), and the majority were aged 25-44 years (95%).
Still, almost one-quarter of women were not sure if vaccines in general are safe (22%). Roughly two-thirds were not sure if vaccines are safe in pregnancy (68%) and a full 10% were not sure if vaccines are effective in preventing illness, Dr. Gutkin said.
Women were significantly more likely to have discussed the influenza vaccine with their ob.gyn. than Tdap (47% vs. 19%), and to be vaccinated for influenza than pertussis (42% vs. 18%).
Of note, women were three times more likely to receive Tdap if they had received a flu shot (OR, 3.68).
"Discussion with their ob.gyn. was a significant factor in the acceptance of Tdap and influenza vaccines during pregnancy," she said.
Dr. Gutkin and her colleagues reported no relevant financial disclosures.
CHICAGO – Pregnant women were significantly more likely to receive information on pertussis vaccination from their pediatrician than from their obstetrician in a survey of 314 women.
"Multiple opportunities exist for education of obstetricians and gynecologists to improve Tdap vaccination rates in the United States," Dr. Rachel Gutkin said at the annual meeting of the Infectious Diseases Society for Obstetrics and Gynecology.
She reported on 314 pregnant women presenting to an academic perinatal center between March and June 2011 who answered an anonymous, multiple-choice questionnaire regarding their knowledge and opinions on vaccination in general, and Tdap (tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis adsorbed) specifically.
Overall, 218 (69%) of the women had heard about the Tdap booster vaccine, with 76 (24%) women learning about it at their pediatrician’s office and 54 (17%) at their obstetrician’s office.
Just 8% of women learned about Tdap from the Internet, while 19% did so from friends or family, and 17% from TV or radio, said Dr. Gutkin, a resident in the department of obstetrics and gynecology at the University of California, Los Angeles. The remaining 15% learned of it from other sources.
The majority of respondents knew that pertussis is a significant health risk for children (76%) and newborns (86%), and 50% also thought it was a significant health risk for fetuses.
When asked whether they would receive a Tdap vaccination during pregnancy to protect their newborn from whooping cough, 11% said they would if their doctor recommended it, 12% said yes if they knew it was safe, and 66% said they would if both conditions were true. Additionally, 11% said they would not receive Tdap during pregnancy under any circumstances, she said.
Women who discussed Tdap with their obstetrician were nearly five times more likely to be vaccinated than those who did not (odds ratio, 4.93). Only 13% of women who did not discuss Tdap with their ob.gyn. were vaccinated.
Although women were significantly more likely to discuss Tdap with their pediatrician, women were nearly three times more likely to be vaccinated if they were counseled by their ob.gyn. versus their pediatrician (OR, 2.9), Dr. Gutkin reported.
The survey was conducted in California, which in 2010 experienced the largest outbreak of pertussis in 65 years, with 9,120 cases reported, including 10 deaths. Cases peaked in some parts of California in the summer of 2010, but public health officials anticipate that numbers will remain high in 2011.
The respondents were well-educated, with 54% completing college and 32% graduate school. Most had PPO (Preferred Provider Organizations) insurance (91%), and the majority were aged 25-44 years (95%).
Still, almost one-quarter of women were not sure if vaccines in general are safe (22%). Roughly two-thirds were not sure if vaccines are safe in pregnancy (68%) and a full 10% were not sure if vaccines are effective in preventing illness, Dr. Gutkin said.
Women were significantly more likely to have discussed the influenza vaccine with their ob.gyn. than Tdap (47% vs. 19%), and to be vaccinated for influenza than pertussis (42% vs. 18%).
Of note, women were three times more likely to receive Tdap if they had received a flu shot (OR, 3.68).
"Discussion with their ob.gyn. was a significant factor in the acceptance of Tdap and influenza vaccines during pregnancy," she said.
Dr. Gutkin and her colleagues reported no relevant financial disclosures.
FROM THE ANNUAL MEETING OF THE INFECTIOUS DISEASES SOCIETY FOR OBSTETRICS AND GYNECOLOGY
Major Finding: Pregnant women were significantly more likely to receive information on pertussis vaccination from their pediatrician than from their obstetrician (24% vs. 17%).
Data Source: Survey of 314 pregnant women presenting to an academic perinatal center between March and June 2011.
Disclosures: Dr. Gutkin and her colleagues reported no relevant financial disclosures.
Flu Vaccine in Pregnancy Benefits Baby, Too
CHICAGO – Influenza vaccination appears to improve neonatal outcomes, but coverage remains inadequate among pregnant women.
Among 1,641 evaluable women delivering at Duke University Hospital during the 2009-2010 influenza season, receipt of any flu vaccination was significantly associated with higher infant birth weight (3,178 grams vs. 2,903 grams) and longer gestational age (38.3 weeks vs. 36.8 weeks, both P values less than .0001).
Women who received at least one flu vaccine also were significantly less likely to require an antepartum visit or hospital admission than those who did not (39% vs. 44%, P = .005).
"This information supports prior accumulating data that receipt of a flu vaccine improves not only maternal outcomes, but also birth outcomes," Dr. Kimberly Fortner said at the annual meeting of the Infectious Diseases Society for Obstetrics and Gynecology.
In all, 44% of women in the preliminary analysis received both vaccines in compliance with recommendations, far higher than historical influenza vaccination rates of 12%-34% and comparable to other reports from the season. Another 7% elected no vaccine at all, and 24% of the population had no documented receipt of vaccine in obstetrical records or other electronic medical records.
Uptake of seasonal influenza vaccine was 58% vs. 55% for the 2009 H1N1 influenza vaccine, which is unique among published prior literature.
Even though rates were nearly equal, 24% of women elected to receive only one of the two recommended vaccines, resulting in inadequate coverage, said Dr. Fortner of Duke University Medical Center in Durham, N.C.
The researchers hypothesized that pregnant women may have inappropriate or inadequate vaccination during the 2009-2010 flu season due to issues of vaccine distribution, sensationalism of the H1N1 influenza pandemic, and recommendations by the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices that pregnant women receive both the seasonal and H1N1 influenza monovalent vaccines.
The earlier women went in for prenatal care, however, the more likely they were to receive a vaccine, she said.
Mean gestational age at first prenatal visit was significantly lower at 14.8 weeks among women who received the vaccine, compared with 18.6 weeks for women who did not receive any vaccine and 21.2 weeks for those with unknown vaccine status (P less than .0001).
Black women and those with public insurance or no insurance were significantly less likely to receive any vaccine.
In multivariate analysis that adjusted for maternal age, black race, less than a high school education, Medicaid or no insurance, and medical comorbidities, receipt of any influenza vaccine during that season was significantly associated with an estimated 133.7 g increase in birth weight (P = .0003).
The association between flu vaccination and preterm birth is unclear but might be explained by the healthy user effect, unidentified confounders or perhaps influenza behaves differently than other viruses. It may activate the inflammatory cascade in different ways, or perhaps the vaccine translates into different mechanisms that are protective, Dr. Fortner said.
Demographic data available on 1,642 women showed that the study population was diverse, with 31.5% black, 31.5% white, 25% Hispanic, 7% Asian/Pacific Islander, and 5% other. The mean maternal age was 28.7 years, 16% had less than a high school education, 28% had a chronic medical condition such as hypertension, diabetes, asthma, or a thrombotic disorder, and 37% received prenatal care at a public health department. Outcome data was not available for one patient.
The study was funded by the 2010 American College of Obstetricians and Gynecologists/Merck & Company Inc. Research Award on Immunization. Dr. Fortner and her colleagues reported no relevant financial disclosures.
CHICAGO – Influenza vaccination appears to improve neonatal outcomes, but coverage remains inadequate among pregnant women.
Among 1,641 evaluable women delivering at Duke University Hospital during the 2009-2010 influenza season, receipt of any flu vaccination was significantly associated with higher infant birth weight (3,178 grams vs. 2,903 grams) and longer gestational age (38.3 weeks vs. 36.8 weeks, both P values less than .0001).
Women who received at least one flu vaccine also were significantly less likely to require an antepartum visit or hospital admission than those who did not (39% vs. 44%, P = .005).
"This information supports prior accumulating data that receipt of a flu vaccine improves not only maternal outcomes, but also birth outcomes," Dr. Kimberly Fortner said at the annual meeting of the Infectious Diseases Society for Obstetrics and Gynecology.
In all, 44% of women in the preliminary analysis received both vaccines in compliance with recommendations, far higher than historical influenza vaccination rates of 12%-34% and comparable to other reports from the season. Another 7% elected no vaccine at all, and 24% of the population had no documented receipt of vaccine in obstetrical records or other electronic medical records.
Uptake of seasonal influenza vaccine was 58% vs. 55% for the 2009 H1N1 influenza vaccine, which is unique among published prior literature.
Even though rates were nearly equal, 24% of women elected to receive only one of the two recommended vaccines, resulting in inadequate coverage, said Dr. Fortner of Duke University Medical Center in Durham, N.C.
The researchers hypothesized that pregnant women may have inappropriate or inadequate vaccination during the 2009-2010 flu season due to issues of vaccine distribution, sensationalism of the H1N1 influenza pandemic, and recommendations by the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices that pregnant women receive both the seasonal and H1N1 influenza monovalent vaccines.
The earlier women went in for prenatal care, however, the more likely they were to receive a vaccine, she said.
Mean gestational age at first prenatal visit was significantly lower at 14.8 weeks among women who received the vaccine, compared with 18.6 weeks for women who did not receive any vaccine and 21.2 weeks for those with unknown vaccine status (P less than .0001).
Black women and those with public insurance or no insurance were significantly less likely to receive any vaccine.
In multivariate analysis that adjusted for maternal age, black race, less than a high school education, Medicaid or no insurance, and medical comorbidities, receipt of any influenza vaccine during that season was significantly associated with an estimated 133.7 g increase in birth weight (P = .0003).
The association between flu vaccination and preterm birth is unclear but might be explained by the healthy user effect, unidentified confounders or perhaps influenza behaves differently than other viruses. It may activate the inflammatory cascade in different ways, or perhaps the vaccine translates into different mechanisms that are protective, Dr. Fortner said.
Demographic data available on 1,642 women showed that the study population was diverse, with 31.5% black, 31.5% white, 25% Hispanic, 7% Asian/Pacific Islander, and 5% other. The mean maternal age was 28.7 years, 16% had less than a high school education, 28% had a chronic medical condition such as hypertension, diabetes, asthma, or a thrombotic disorder, and 37% received prenatal care at a public health department. Outcome data was not available for one patient.
The study was funded by the 2010 American College of Obstetricians and Gynecologists/Merck & Company Inc. Research Award on Immunization. Dr. Fortner and her colleagues reported no relevant financial disclosures.
CHICAGO – Influenza vaccination appears to improve neonatal outcomes, but coverage remains inadequate among pregnant women.
Among 1,641 evaluable women delivering at Duke University Hospital during the 2009-2010 influenza season, receipt of any flu vaccination was significantly associated with higher infant birth weight (3,178 grams vs. 2,903 grams) and longer gestational age (38.3 weeks vs. 36.8 weeks, both P values less than .0001).
Women who received at least one flu vaccine also were significantly less likely to require an antepartum visit or hospital admission than those who did not (39% vs. 44%, P = .005).
"This information supports prior accumulating data that receipt of a flu vaccine improves not only maternal outcomes, but also birth outcomes," Dr. Kimberly Fortner said at the annual meeting of the Infectious Diseases Society for Obstetrics and Gynecology.
In all, 44% of women in the preliminary analysis received both vaccines in compliance with recommendations, far higher than historical influenza vaccination rates of 12%-34% and comparable to other reports from the season. Another 7% elected no vaccine at all, and 24% of the population had no documented receipt of vaccine in obstetrical records or other electronic medical records.
Uptake of seasonal influenza vaccine was 58% vs. 55% for the 2009 H1N1 influenza vaccine, which is unique among published prior literature.
Even though rates were nearly equal, 24% of women elected to receive only one of the two recommended vaccines, resulting in inadequate coverage, said Dr. Fortner of Duke University Medical Center in Durham, N.C.
The researchers hypothesized that pregnant women may have inappropriate or inadequate vaccination during the 2009-2010 flu season due to issues of vaccine distribution, sensationalism of the H1N1 influenza pandemic, and recommendations by the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices that pregnant women receive both the seasonal and H1N1 influenza monovalent vaccines.
The earlier women went in for prenatal care, however, the more likely they were to receive a vaccine, she said.
Mean gestational age at first prenatal visit was significantly lower at 14.8 weeks among women who received the vaccine, compared with 18.6 weeks for women who did not receive any vaccine and 21.2 weeks for those with unknown vaccine status (P less than .0001).
Black women and those with public insurance or no insurance were significantly less likely to receive any vaccine.
In multivariate analysis that adjusted for maternal age, black race, less than a high school education, Medicaid or no insurance, and medical comorbidities, receipt of any influenza vaccine during that season was significantly associated with an estimated 133.7 g increase in birth weight (P = .0003).
The association between flu vaccination and preterm birth is unclear but might be explained by the healthy user effect, unidentified confounders or perhaps influenza behaves differently than other viruses. It may activate the inflammatory cascade in different ways, or perhaps the vaccine translates into different mechanisms that are protective, Dr. Fortner said.
Demographic data available on 1,642 women showed that the study population was diverse, with 31.5% black, 31.5% white, 25% Hispanic, 7% Asian/Pacific Islander, and 5% other. The mean maternal age was 28.7 years, 16% had less than a high school education, 28% had a chronic medical condition such as hypertension, diabetes, asthma, or a thrombotic disorder, and 37% received prenatal care at a public health department. Outcome data was not available for one patient.
The study was funded by the 2010 American College of Obstetricians and Gynecologists/Merck & Company Inc. Research Award on Immunization. Dr. Fortner and her colleagues reported no relevant financial disclosures.
FROM THE ANNUAL MEETING OF THE INFECTIOUS DISEASES SOCIETY FOR OBSTETRICS AND GYNECOLOGY
Major Finding: Receipt of any flu vaccination was significantly associated with higher infant birth weight (3,178 gram vs. 2,903 grams) and longer gestational age (38.3 weeks vs. 36.8 weeks, both P less than .0001).
Data Source: Preliminary analysis of 1,641 women delivering at Duke University Hospital during the 2009-2010 influenza season.
Disclosures: The study was funded by the 2010 American College of Obstetricians and Gynecologists/Merck & Company Inc. Research Award on Immunization. Dr. Fortner and her colleagues reported no relevant financial disclosures.
Handle Perioperative Management of Morbidly Obese With Care
KANSAS CITY, MO. – Seventeen percent of American children and adolescents are obese, and an increasing number of them are landing in operating rooms for related issues. They bring to the table particular perioperative concerns.
Dr. Moises Auron of the Cleveland Clinic offered an example during a talk on pediatric perioperative management at the Pediatric Hospital Medicine 2011 meeting: A 15-year-old African American with a body mass index of 60 kg/m2, fasting blood glucose of 115 mg/dL, and glycated hemoglobin of 6.4%, and who smokes, requires pinning of his hip for slipped capital femoral epiphysis.
For this high-risk patient, he said, cardiovascular concerns top the list, because obese children and adolescents are predisposed to hypertension at roughly a threefold higher risk than nonobese children. As a result, they can develop left ventricular hypertrophy that can produce a hypertrophic cardiomyopathy–like scenario, as well as diastolic dysfunction that also can predispose them to develop heart failure.
Childhood obesity is typically defined as a BMI at or above the 95th percentile for age and sex, while morbid obesity, as in the above patient, is a BMI that exceeds the 99th percentile.
Three times as many U.S. youth are obese now as were just one generation ago, the most recent data from the Centers for Disease Control and Prevention suggest. In addition, one in seven low-income, preschool-aged children is obese.
"Unfortunately, we are seeing more and more and more of these cases," said Dr. Auron, a pediatric hospitalist with the Center for Pediatric Hospital Medicine at the Cleveland Clinic.
Obese patients may require surgery not only for a variety of common ailments such as tonsillitis, but also for obesity-related conditions such as slipped capital femoral epiphysis, Blount’s disease, cholelithiasis, and polycystic ovary syndrome. In addition, they present with comorbid conditions such as insulin resistance, hypertension, and idiopathic intracranial hypertension.
What to Look for in Obese Patients
In the examination of these patients, Dr. Auron suggests that the history include any symptoms of sleep apnea or hypoventilation, such as falling asleep while watching television or when talking with friends, and poor tolerance to exercise, including breathlessness and asthma, because these may in fact represent symptoms of left ventricular dysfunction.
Documentation of recent weight loss or gain, as well as a careful history of current medications – especially herbs or special mixtures taken to lose weight – is paramount. Weight loss may be associated with malnutrition, while the use of common herbs can interfere with anesthesia or hemostasis and even be associated with tachycardia, hypertension, or dysrhythmias.
"Garlic and ginger are very good antiplatelet agents," he said. "It’s like taking baby aspirin, but it’s not good if you’re having surgery. You can bleed to death.
"Medications that contain ephedra can trigger a severe sympathetic response, including hypertensive crises and tachyarrhythmias."
Other perioperative considerations to be wary of in the morbidly obese pediatric patient are diabetes/insulin resistance; gastroesophageal reflux; and nonalcoholic fatty liver disease (NAFLD), including its progressive form, nonalcoholic steatohepatitis (NASH).
"When they develop NASH, it can evolve to cirrhosis, horribly," Dr. Auron said at the meeting sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association. "It’s even worse than alcoholic cirrhosis or viral hepatitis cirrhosis. I’m not sure why this is, but it causes substantial morbidity in the obese population who develops it."
The prevalence of NAFLD and NASH varies by setting. Among 41 morbidly obese adolescents aged 13-19 years undergoing gastric bypass surgery, 83% had NAFLD and 20% had NASH. Mean fasting glucose was significantly higher in those with NASH, although the prevalence of the metabolic syndrome was not (Gastroenterol. Hepatol. 2006;4:226-32).
A second study, however, recently reported that fatty liver disease, independent of visceral fat and intramyocellular lipid content, plays a central role in the pathogenesis of insulin resistance in obese adolescents (Diabetes Care 2010;33:1817-22).
Dr. Auron said he recommends echocardiography for hypertensive patients, although he noted that there is not a lot of evidence, even in the adult literature, to support an echocardiogram for every patient. The one exception is patients with cardiomegaly on x-ray; echocardiography is recommended for this patient group (Circulation 2009;120:86-95).
Preoperative fasting should follow the same rules as for nonobese patients, albeit perhaps a bit more fasting would not lead obese patients astray, he said, tongue in cheek.
Dr. Auron reported no relevant financial relationships.
Dr. Moises Auron, pediatric perioperative management, Pediatric Hospital Medicine, hypertension,
KANSAS CITY, MO. – Seventeen percent of American children and adolescents are obese, and an increasing number of them are landing in operating rooms for related issues. They bring to the table particular perioperative concerns.
Dr. Moises Auron of the Cleveland Clinic offered an example during a talk on pediatric perioperative management at the Pediatric Hospital Medicine 2011 meeting: A 15-year-old African American with a body mass index of 60 kg/m2, fasting blood glucose of 115 mg/dL, and glycated hemoglobin of 6.4%, and who smokes, requires pinning of his hip for slipped capital femoral epiphysis.
For this high-risk patient, he said, cardiovascular concerns top the list, because obese children and adolescents are predisposed to hypertension at roughly a threefold higher risk than nonobese children. As a result, they can develop left ventricular hypertrophy that can produce a hypertrophic cardiomyopathy–like scenario, as well as diastolic dysfunction that also can predispose them to develop heart failure.
Childhood obesity is typically defined as a BMI at or above the 95th percentile for age and sex, while morbid obesity, as in the above patient, is a BMI that exceeds the 99th percentile.
Three times as many U.S. youth are obese now as were just one generation ago, the most recent data from the Centers for Disease Control and Prevention suggest. In addition, one in seven low-income, preschool-aged children is obese.
"Unfortunately, we are seeing more and more and more of these cases," said Dr. Auron, a pediatric hospitalist with the Center for Pediatric Hospital Medicine at the Cleveland Clinic.
Obese patients may require surgery not only for a variety of common ailments such as tonsillitis, but also for obesity-related conditions such as slipped capital femoral epiphysis, Blount’s disease, cholelithiasis, and polycystic ovary syndrome. In addition, they present with comorbid conditions such as insulin resistance, hypertension, and idiopathic intracranial hypertension.
What to Look for in Obese Patients
In the examination of these patients, Dr. Auron suggests that the history include any symptoms of sleep apnea or hypoventilation, such as falling asleep while watching television or when talking with friends, and poor tolerance to exercise, including breathlessness and asthma, because these may in fact represent symptoms of left ventricular dysfunction.
Documentation of recent weight loss or gain, as well as a careful history of current medications – especially herbs or special mixtures taken to lose weight – is paramount. Weight loss may be associated with malnutrition, while the use of common herbs can interfere with anesthesia or hemostasis and even be associated with tachycardia, hypertension, or dysrhythmias.
"Garlic and ginger are very good antiplatelet agents," he said. "It’s like taking baby aspirin, but it’s not good if you’re having surgery. You can bleed to death.
"Medications that contain ephedra can trigger a severe sympathetic response, including hypertensive crises and tachyarrhythmias."
Other perioperative considerations to be wary of in the morbidly obese pediatric patient are diabetes/insulin resistance; gastroesophageal reflux; and nonalcoholic fatty liver disease (NAFLD), including its progressive form, nonalcoholic steatohepatitis (NASH).
"When they develop NASH, it can evolve to cirrhosis, horribly," Dr. Auron said at the meeting sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association. "It’s even worse than alcoholic cirrhosis or viral hepatitis cirrhosis. I’m not sure why this is, but it causes substantial morbidity in the obese population who develops it."
The prevalence of NAFLD and NASH varies by setting. Among 41 morbidly obese adolescents aged 13-19 years undergoing gastric bypass surgery, 83% had NAFLD and 20% had NASH. Mean fasting glucose was significantly higher in those with NASH, although the prevalence of the metabolic syndrome was not (Gastroenterol. Hepatol. 2006;4:226-32).
A second study, however, recently reported that fatty liver disease, independent of visceral fat and intramyocellular lipid content, plays a central role in the pathogenesis of insulin resistance in obese adolescents (Diabetes Care 2010;33:1817-22).
Dr. Auron said he recommends echocardiography for hypertensive patients, although he noted that there is not a lot of evidence, even in the adult literature, to support an echocardiogram for every patient. The one exception is patients with cardiomegaly on x-ray; echocardiography is recommended for this patient group (Circulation 2009;120:86-95).
Preoperative fasting should follow the same rules as for nonobese patients, albeit perhaps a bit more fasting would not lead obese patients astray, he said, tongue in cheek.
Dr. Auron reported no relevant financial relationships.
KANSAS CITY, MO. – Seventeen percent of American children and adolescents are obese, and an increasing number of them are landing in operating rooms for related issues. They bring to the table particular perioperative concerns.
Dr. Moises Auron of the Cleveland Clinic offered an example during a talk on pediatric perioperative management at the Pediatric Hospital Medicine 2011 meeting: A 15-year-old African American with a body mass index of 60 kg/m2, fasting blood glucose of 115 mg/dL, and glycated hemoglobin of 6.4%, and who smokes, requires pinning of his hip for slipped capital femoral epiphysis.
For this high-risk patient, he said, cardiovascular concerns top the list, because obese children and adolescents are predisposed to hypertension at roughly a threefold higher risk than nonobese children. As a result, they can develop left ventricular hypertrophy that can produce a hypertrophic cardiomyopathy–like scenario, as well as diastolic dysfunction that also can predispose them to develop heart failure.
Childhood obesity is typically defined as a BMI at or above the 95th percentile for age and sex, while morbid obesity, as in the above patient, is a BMI that exceeds the 99th percentile.
Three times as many U.S. youth are obese now as were just one generation ago, the most recent data from the Centers for Disease Control and Prevention suggest. In addition, one in seven low-income, preschool-aged children is obese.
"Unfortunately, we are seeing more and more and more of these cases," said Dr. Auron, a pediatric hospitalist with the Center for Pediatric Hospital Medicine at the Cleveland Clinic.
Obese patients may require surgery not only for a variety of common ailments such as tonsillitis, but also for obesity-related conditions such as slipped capital femoral epiphysis, Blount’s disease, cholelithiasis, and polycystic ovary syndrome. In addition, they present with comorbid conditions such as insulin resistance, hypertension, and idiopathic intracranial hypertension.
What to Look for in Obese Patients
In the examination of these patients, Dr. Auron suggests that the history include any symptoms of sleep apnea or hypoventilation, such as falling asleep while watching television or when talking with friends, and poor tolerance to exercise, including breathlessness and asthma, because these may in fact represent symptoms of left ventricular dysfunction.
Documentation of recent weight loss or gain, as well as a careful history of current medications – especially herbs or special mixtures taken to lose weight – is paramount. Weight loss may be associated with malnutrition, while the use of common herbs can interfere with anesthesia or hemostasis and even be associated with tachycardia, hypertension, or dysrhythmias.
"Garlic and ginger are very good antiplatelet agents," he said. "It’s like taking baby aspirin, but it’s not good if you’re having surgery. You can bleed to death.
"Medications that contain ephedra can trigger a severe sympathetic response, including hypertensive crises and tachyarrhythmias."
Other perioperative considerations to be wary of in the morbidly obese pediatric patient are diabetes/insulin resistance; gastroesophageal reflux; and nonalcoholic fatty liver disease (NAFLD), including its progressive form, nonalcoholic steatohepatitis (NASH).
"When they develop NASH, it can evolve to cirrhosis, horribly," Dr. Auron said at the meeting sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association. "It’s even worse than alcoholic cirrhosis or viral hepatitis cirrhosis. I’m not sure why this is, but it causes substantial morbidity in the obese population who develops it."
The prevalence of NAFLD and NASH varies by setting. Among 41 morbidly obese adolescents aged 13-19 years undergoing gastric bypass surgery, 83% had NAFLD and 20% had NASH. Mean fasting glucose was significantly higher in those with NASH, although the prevalence of the metabolic syndrome was not (Gastroenterol. Hepatol. 2006;4:226-32).
A second study, however, recently reported that fatty liver disease, independent of visceral fat and intramyocellular lipid content, plays a central role in the pathogenesis of insulin resistance in obese adolescents (Diabetes Care 2010;33:1817-22).
Dr. Auron said he recommends echocardiography for hypertensive patients, although he noted that there is not a lot of evidence, even in the adult literature, to support an echocardiogram for every patient. The one exception is patients with cardiomegaly on x-ray; echocardiography is recommended for this patient group (Circulation 2009;120:86-95).
Preoperative fasting should follow the same rules as for nonobese patients, albeit perhaps a bit more fasting would not lead obese patients astray, he said, tongue in cheek.
Dr. Auron reported no relevant financial relationships.
Dr. Moises Auron, pediatric perioperative management, Pediatric Hospital Medicine, hypertension,
Dr. Moises Auron, pediatric perioperative management, Pediatric Hospital Medicine, hypertension,
EXPERT ANALYSIS FROM THE PEDIATRIC HOSPITAL MEDICINE 2011 MEETING
'Mild Association' Found Between Autism, Intra-amniotic Infection
CHICAGO – The risk of autism is mildly increased in women who experience an intra-amniotic infection during labor, according to a study reported at the annual meeting of the Infectious Diseases Society for Obstetrics and Gynecology. A separate study, also reported at the meeting, found no such link, however.
In the first study, a retrospective birth-cohort analysis, the rate of intra-amniotic infection (IAI) was 7.27% among mothers whose children developed autism and 6.51% among those with typically developing children. After adjustment for maternal age, parity, and insurance payer, the risk of having a child with autism was increased 6% in mothers with an intra-amniotic infection (odds ratio, 1.06).
"There’s probably something small going on there," Dr. Cheryl Walker of the University of California, Davis, MIND (Medical Investigation of Neurodevelopmental Disorders) Institute reported.
"We’re putting it out there because there is very little, if any, connection between these specific perinatal birth-related events and autism, but a lot of people are very concerned about them," she added.
The analysis was based on administrative data from 20,206 children with autism and 6,028,353 controls born from January 1991 through December 2001 in California and recorded in the state’s massive Office of Statewide Health Planning and Development (OSHPD) database. IAI, which complicates up to 4% of term deliveries, was identified using the ICD-9 diagnostic code 658.4.
Dr. Walker pointed out that IAI is often underdiagnosed and poorly documented in medical records, leading to under-reporting to OSHPD. In addition, subtle subclinical states cannot be assessed from administrative data.
"Our results likely underestimate a mild association between intra-amniotic infection and autism," she acknowledged.
In contrast, analysis of data from a case-control study called CHARGE found no association between autism and IAI. CHARGE (Childhood Autism Risks from Genetics and the Environment) was launched in 2003, and includes nearly 1,500 patients from northern California, some of whom have autism spectrum disorders and others who are typically developing or have developmental delays but not ASD.
This substudy of CHARGE included only those for whom medical records were available from their births, and analyzed 509 mothers of children with ASD and 325 controls. IAI was evaluated as overt IAI documented in the maternal medical record; covert IAI defined as the presence of at least one IAI symptom of uterine tenderness, foul vaginal discharge, purulent amniotic fluid, and maternal or fetal tachycardia; IAI risk defined on a four-point scale ranging from no risk to overt IAI; and finally, IAI risk with or without the use of antibiotics in labor.
No significant differences were observed between 396 evaluable mothers of children with autism/ASD and 247 mothers of typically developing children in rates of overt IAI (3.9% vs. 4.6%), presence of at least one IAI symptom (9.8% vs. 9.9%), or any IAI risk (20.2% vs. 18.6%), Dr. Walker reported.
Rates also were similar between the autism and typically developing groups for rupture of membranes greater than 18 hours (6.5% vs. 4.1%), antibiotic use in labor (26.3% vs. 22.4%), and any IAI risk without antibiotics (12% vs. 10%).
After adjustment for maternal age, payer, and birth order, IAI risk was similar among mothers of children with autism and those without (adjusted OR, 1.08).
"The neurobiologists tell us that we should be focusing more on the first and second trimesters, so maybe exposure to infection at the time of birth is too late to have much of an effect," Dr. Walker said in an interview. "Most of the neuronal migration has probably occurred by the time of the infection."
In addition, there was the potential for a type II statistical error in the CHARGE study and suboptimal classification of predictors and outcomes in the OSHPD study.
"Case-control designs are great for rare outcomes, but don’t help with rare exposures; and huge administrative data sets help with power at the cost of covariate misclassification," she said.
The studies were prompted by a growing body of evidence that IAI is associated with adverse neurodevelopment and cerebral palsy, and that inflammation plays a role in autism. Many children with autism have altered immune systems; lower levels of circulating immunoglobulin G antibodies; impaired responses to a variety of stimulation assays in vitro, including immunizations; and very unusual natural killer cell function on a variety of levels, Dr. Walker explained.
Dr. Walker reported financial relationships with Merck and Graceway. Her coauthors reported no conflicts.
CHICAGO – The risk of autism is mildly increased in women who experience an intra-amniotic infection during labor, according to a study reported at the annual meeting of the Infectious Diseases Society for Obstetrics and Gynecology. A separate study, also reported at the meeting, found no such link, however.
In the first study, a retrospective birth-cohort analysis, the rate of intra-amniotic infection (IAI) was 7.27% among mothers whose children developed autism and 6.51% among those with typically developing children. After adjustment for maternal age, parity, and insurance payer, the risk of having a child with autism was increased 6% in mothers with an intra-amniotic infection (odds ratio, 1.06).
"There’s probably something small going on there," Dr. Cheryl Walker of the University of California, Davis, MIND (Medical Investigation of Neurodevelopmental Disorders) Institute reported.
"We’re putting it out there because there is very little, if any, connection between these specific perinatal birth-related events and autism, but a lot of people are very concerned about them," she added.
The analysis was based on administrative data from 20,206 children with autism and 6,028,353 controls born from January 1991 through December 2001 in California and recorded in the state’s massive Office of Statewide Health Planning and Development (OSHPD) database. IAI, which complicates up to 4% of term deliveries, was identified using the ICD-9 diagnostic code 658.4.
Dr. Walker pointed out that IAI is often underdiagnosed and poorly documented in medical records, leading to under-reporting to OSHPD. In addition, subtle subclinical states cannot be assessed from administrative data.
"Our results likely underestimate a mild association between intra-amniotic infection and autism," she acknowledged.
In contrast, analysis of data from a case-control study called CHARGE found no association between autism and IAI. CHARGE (Childhood Autism Risks from Genetics and the Environment) was launched in 2003, and includes nearly 1,500 patients from northern California, some of whom have autism spectrum disorders and others who are typically developing or have developmental delays but not ASD.
This substudy of CHARGE included only those for whom medical records were available from their births, and analyzed 509 mothers of children with ASD and 325 controls. IAI was evaluated as overt IAI documented in the maternal medical record; covert IAI defined as the presence of at least one IAI symptom of uterine tenderness, foul vaginal discharge, purulent amniotic fluid, and maternal or fetal tachycardia; IAI risk defined on a four-point scale ranging from no risk to overt IAI; and finally, IAI risk with or without the use of antibiotics in labor.
No significant differences were observed between 396 evaluable mothers of children with autism/ASD and 247 mothers of typically developing children in rates of overt IAI (3.9% vs. 4.6%), presence of at least one IAI symptom (9.8% vs. 9.9%), or any IAI risk (20.2% vs. 18.6%), Dr. Walker reported.
Rates also were similar between the autism and typically developing groups for rupture of membranes greater than 18 hours (6.5% vs. 4.1%), antibiotic use in labor (26.3% vs. 22.4%), and any IAI risk without antibiotics (12% vs. 10%).
After adjustment for maternal age, payer, and birth order, IAI risk was similar among mothers of children with autism and those without (adjusted OR, 1.08).
"The neurobiologists tell us that we should be focusing more on the first and second trimesters, so maybe exposure to infection at the time of birth is too late to have much of an effect," Dr. Walker said in an interview. "Most of the neuronal migration has probably occurred by the time of the infection."
In addition, there was the potential for a type II statistical error in the CHARGE study and suboptimal classification of predictors and outcomes in the OSHPD study.
"Case-control designs are great for rare outcomes, but don’t help with rare exposures; and huge administrative data sets help with power at the cost of covariate misclassification," she said.
The studies were prompted by a growing body of evidence that IAI is associated with adverse neurodevelopment and cerebral palsy, and that inflammation plays a role in autism. Many children with autism have altered immune systems; lower levels of circulating immunoglobulin G antibodies; impaired responses to a variety of stimulation assays in vitro, including immunizations; and very unusual natural killer cell function on a variety of levels, Dr. Walker explained.
Dr. Walker reported financial relationships with Merck and Graceway. Her coauthors reported no conflicts.
CHICAGO – The risk of autism is mildly increased in women who experience an intra-amniotic infection during labor, according to a study reported at the annual meeting of the Infectious Diseases Society for Obstetrics and Gynecology. A separate study, also reported at the meeting, found no such link, however.
In the first study, a retrospective birth-cohort analysis, the rate of intra-amniotic infection (IAI) was 7.27% among mothers whose children developed autism and 6.51% among those with typically developing children. After adjustment for maternal age, parity, and insurance payer, the risk of having a child with autism was increased 6% in mothers with an intra-amniotic infection (odds ratio, 1.06).
"There’s probably something small going on there," Dr. Cheryl Walker of the University of California, Davis, MIND (Medical Investigation of Neurodevelopmental Disorders) Institute reported.
"We’re putting it out there because there is very little, if any, connection between these specific perinatal birth-related events and autism, but a lot of people are very concerned about them," she added.
The analysis was based on administrative data from 20,206 children with autism and 6,028,353 controls born from January 1991 through December 2001 in California and recorded in the state’s massive Office of Statewide Health Planning and Development (OSHPD) database. IAI, which complicates up to 4% of term deliveries, was identified using the ICD-9 diagnostic code 658.4.
Dr. Walker pointed out that IAI is often underdiagnosed and poorly documented in medical records, leading to under-reporting to OSHPD. In addition, subtle subclinical states cannot be assessed from administrative data.
"Our results likely underestimate a mild association between intra-amniotic infection and autism," she acknowledged.
In contrast, analysis of data from a case-control study called CHARGE found no association between autism and IAI. CHARGE (Childhood Autism Risks from Genetics and the Environment) was launched in 2003, and includes nearly 1,500 patients from northern California, some of whom have autism spectrum disorders and others who are typically developing or have developmental delays but not ASD.
This substudy of CHARGE included only those for whom medical records were available from their births, and analyzed 509 mothers of children with ASD and 325 controls. IAI was evaluated as overt IAI documented in the maternal medical record; covert IAI defined as the presence of at least one IAI symptom of uterine tenderness, foul vaginal discharge, purulent amniotic fluid, and maternal or fetal tachycardia; IAI risk defined on a four-point scale ranging from no risk to overt IAI; and finally, IAI risk with or without the use of antibiotics in labor.
No significant differences were observed between 396 evaluable mothers of children with autism/ASD and 247 mothers of typically developing children in rates of overt IAI (3.9% vs. 4.6%), presence of at least one IAI symptom (9.8% vs. 9.9%), or any IAI risk (20.2% vs. 18.6%), Dr. Walker reported.
Rates also were similar between the autism and typically developing groups for rupture of membranes greater than 18 hours (6.5% vs. 4.1%), antibiotic use in labor (26.3% vs. 22.4%), and any IAI risk without antibiotics (12% vs. 10%).
After adjustment for maternal age, payer, and birth order, IAI risk was similar among mothers of children with autism and those without (adjusted OR, 1.08).
"The neurobiologists tell us that we should be focusing more on the first and second trimesters, so maybe exposure to infection at the time of birth is too late to have much of an effect," Dr. Walker said in an interview. "Most of the neuronal migration has probably occurred by the time of the infection."
In addition, there was the potential for a type II statistical error in the CHARGE study and suboptimal classification of predictors and outcomes in the OSHPD study.
"Case-control designs are great for rare outcomes, but don’t help with rare exposures; and huge administrative data sets help with power at the cost of covariate misclassification," she said.
The studies were prompted by a growing body of evidence that IAI is associated with adverse neurodevelopment and cerebral palsy, and that inflammation plays a role in autism. Many children with autism have altered immune systems; lower levels of circulating immunoglobulin G antibodies; impaired responses to a variety of stimulation assays in vitro, including immunizations; and very unusual natural killer cell function on a variety of levels, Dr. Walker explained.
Dr. Walker reported financial relationships with Merck and Graceway. Her coauthors reported no conflicts.
FROM THE ANNUAL MEETING OF THE INFECTIOUS DISEASES SOCIETY FOR OBSTETRICS AND GYNECOLOGY
Major Finding: The adjusted risk of having a child with autism was increased 6% in mothers with an intra-amniotic infection during labor in one study. A second study found no such association.
Data Source: The first study was a retrospective birth-cohort analysis of 20,206 children with autism and 6,028,353 controls. The second study was a case-control study of 509 mothers of children with autism spectrum disorders and 325 controls.
Disclosures: Dr. Walker reported financial relationships with Merck and Graceway. Her coauthors reported no conflicts.
Computer Wizardry Not Crucial to Cutting Hospital Errors
KANSAS CITY, MO. – The use of a resident handoff bundle was associated with significant reductions in overall medical errors, regardless of whether a computerized tool linking the electronic medical record was present or not.
Rates of errors, including those that harmed patients, near misses, and minor errors, significantly decreased from 32% to 19% in a unit with the full intervention and from 45% to 25% in a unit with all but the computerized tool. The rate of decline between the two units was not significantly different.
The two general inpatient units at Children’s Hospital Boston had relatively similar patient populations and size, the same resident call system, and the identical preexisting system for verbal and written handoffs, lead author Dr. Amy J. Starmer explained during the plenary session at the Pediatric Hospital Medicine 2011 meeting.
A review of randomly selected sign-out documents also found that several key elements were present significantly more often after implementation of the resident handoff bundle (RHB).
The research intervention featured three components, including communication and handoff skills training for resident physicians, adapted in large part from TeamSTEPPS, a communication and teamwork skills program developed by the Department of Defense and the Agency for Healthcare Research and Quality.
Secondly, the investigators attempted to standardize verbal handoffs by putting a verbal mnemonic in place, restructuring the process so that all residents and interns were brought together as a team and enforcing the process with intermittent evaluations and feedback from the chief resident.
Finally, a computerized handoff tool was introduced in Unit 1 that allowed for pre-text entry of key information fields and automatic importing of information such as demographics, medications, or patient code status, said Dr. Starmer, a pediatrician at Children’s Hospital. Unit 2 used a Microsoft Word document for these written tasks.
Outcomes were measured 3 months pre- and post-intervention using previously standardized error surveillance methods, a review of 107 electronic copies of daily printed sign-outs, and a written miscommunication abstraction tool with 14 key data elements. In all, 529 patient entries were reviewed for Unit 1 and 204 for Unit 2.
Based on the review of Unit 1, certain items were uniformly included preintervention such as name (97%), medical record number (97%), chief complaint (97%), allergies (95%), weight (94%), and patient summary (75%), while others such as medication list (3%), code status (2%), lab results (1%), and vital signs with date (0%) were rarely present, Dr. Starmer said.
All of the aforementioned items, except for chief complaint (99%) and patient summary (95%), were present in 100% of reports postintervention, likely because the data were being automatically imported, she said. Improvements also were observed for more complex items such as to-do lists (29% pre vs. 82% post) and contingency plans (18% vs. 47%), likely because of computer prompting. All comparisons, except chief complaint, were statistically significant (P less than .05).
In Unit 2, significant gains were observed in the inclusion of contingency plans (25% pre vs. 40% post), lab results with date (7% vs. 17%), and vital signs with date (2% vs. 9%) (P less than .05 for all), Dr. Starmer said.
Upward trends were noted for the routinely included items of chief complaint (96% vs. 100%), weight (99% vs. 100%), and patient summary (96% vs. 100%), with slight declines in allergies (94% vs. 91%) and code status (3% vs. 2%). Name and medical record number were included in 100% of records pre- and postintervention.
Limitations of the study include the inability to control for resident learning over time and seasonal variations, its single-site design, and the inability to sustain the use of the verbal mnemonic following the research period, said Dr. Starmer at the meeting, sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
She went on to say that these limitations will be addressed in I-PASS (IIPE-PRIS Accelerating Safe Sign-Outs), a multicenter study supported by the Initiative for Innovation in Pediatric Education (IIPE) and the Pediatric Research in Inpatient Settings (PRIS) network.
An attendee asked how the team approach to resident-intern verbal signoffs has been impacted by new resident work hours. Dr. Starmer said there was some concern that the new work rules would affect the amount of time spent with each patient, but that this has not changed.
Dr. Starmer and her coauthors reported no relevant financial relationships.
KANSAS CITY, MO. – The use of a resident handoff bundle was associated with significant reductions in overall medical errors, regardless of whether a computerized tool linking the electronic medical record was present or not.
Rates of errors, including those that harmed patients, near misses, and minor errors, significantly decreased from 32% to 19% in a unit with the full intervention and from 45% to 25% in a unit with all but the computerized tool. The rate of decline between the two units was not significantly different.
The two general inpatient units at Children’s Hospital Boston had relatively similar patient populations and size, the same resident call system, and the identical preexisting system for verbal and written handoffs, lead author Dr. Amy J. Starmer explained during the plenary session at the Pediatric Hospital Medicine 2011 meeting.
A review of randomly selected sign-out documents also found that several key elements were present significantly more often after implementation of the resident handoff bundle (RHB).
The research intervention featured three components, including communication and handoff skills training for resident physicians, adapted in large part from TeamSTEPPS, a communication and teamwork skills program developed by the Department of Defense and the Agency for Healthcare Research and Quality.
Secondly, the investigators attempted to standardize verbal handoffs by putting a verbal mnemonic in place, restructuring the process so that all residents and interns were brought together as a team and enforcing the process with intermittent evaluations and feedback from the chief resident.
Finally, a computerized handoff tool was introduced in Unit 1 that allowed for pre-text entry of key information fields and automatic importing of information such as demographics, medications, or patient code status, said Dr. Starmer, a pediatrician at Children’s Hospital. Unit 2 used a Microsoft Word document for these written tasks.
Outcomes were measured 3 months pre- and post-intervention using previously standardized error surveillance methods, a review of 107 electronic copies of daily printed sign-outs, and a written miscommunication abstraction tool with 14 key data elements. In all, 529 patient entries were reviewed for Unit 1 and 204 for Unit 2.
Based on the review of Unit 1, certain items were uniformly included preintervention such as name (97%), medical record number (97%), chief complaint (97%), allergies (95%), weight (94%), and patient summary (75%), while others such as medication list (3%), code status (2%), lab results (1%), and vital signs with date (0%) were rarely present, Dr. Starmer said.
All of the aforementioned items, except for chief complaint (99%) and patient summary (95%), were present in 100% of reports postintervention, likely because the data were being automatically imported, she said. Improvements also were observed for more complex items such as to-do lists (29% pre vs. 82% post) and contingency plans (18% vs. 47%), likely because of computer prompting. All comparisons, except chief complaint, were statistically significant (P less than .05).
In Unit 2, significant gains were observed in the inclusion of contingency plans (25% pre vs. 40% post), lab results with date (7% vs. 17%), and vital signs with date (2% vs. 9%) (P less than .05 for all), Dr. Starmer said.
Upward trends were noted for the routinely included items of chief complaint (96% vs. 100%), weight (99% vs. 100%), and patient summary (96% vs. 100%), with slight declines in allergies (94% vs. 91%) and code status (3% vs. 2%). Name and medical record number were included in 100% of records pre- and postintervention.
Limitations of the study include the inability to control for resident learning over time and seasonal variations, its single-site design, and the inability to sustain the use of the verbal mnemonic following the research period, said Dr. Starmer at the meeting, sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
She went on to say that these limitations will be addressed in I-PASS (IIPE-PRIS Accelerating Safe Sign-Outs), a multicenter study supported by the Initiative for Innovation in Pediatric Education (IIPE) and the Pediatric Research in Inpatient Settings (PRIS) network.
An attendee asked how the team approach to resident-intern verbal signoffs has been impacted by new resident work hours. Dr. Starmer said there was some concern that the new work rules would affect the amount of time spent with each patient, but that this has not changed.
Dr. Starmer and her coauthors reported no relevant financial relationships.
KANSAS CITY, MO. – The use of a resident handoff bundle was associated with significant reductions in overall medical errors, regardless of whether a computerized tool linking the electronic medical record was present or not.
Rates of errors, including those that harmed patients, near misses, and minor errors, significantly decreased from 32% to 19% in a unit with the full intervention and from 45% to 25% in a unit with all but the computerized tool. The rate of decline between the two units was not significantly different.
The two general inpatient units at Children’s Hospital Boston had relatively similar patient populations and size, the same resident call system, and the identical preexisting system for verbal and written handoffs, lead author Dr. Amy J. Starmer explained during the plenary session at the Pediatric Hospital Medicine 2011 meeting.
A review of randomly selected sign-out documents also found that several key elements were present significantly more often after implementation of the resident handoff bundle (RHB).
The research intervention featured three components, including communication and handoff skills training for resident physicians, adapted in large part from TeamSTEPPS, a communication and teamwork skills program developed by the Department of Defense and the Agency for Healthcare Research and Quality.
Secondly, the investigators attempted to standardize verbal handoffs by putting a verbal mnemonic in place, restructuring the process so that all residents and interns were brought together as a team and enforcing the process with intermittent evaluations and feedback from the chief resident.
Finally, a computerized handoff tool was introduced in Unit 1 that allowed for pre-text entry of key information fields and automatic importing of information such as demographics, medications, or patient code status, said Dr. Starmer, a pediatrician at Children’s Hospital. Unit 2 used a Microsoft Word document for these written tasks.
Outcomes were measured 3 months pre- and post-intervention using previously standardized error surveillance methods, a review of 107 electronic copies of daily printed sign-outs, and a written miscommunication abstraction tool with 14 key data elements. In all, 529 patient entries were reviewed for Unit 1 and 204 for Unit 2.
Based on the review of Unit 1, certain items were uniformly included preintervention such as name (97%), medical record number (97%), chief complaint (97%), allergies (95%), weight (94%), and patient summary (75%), while others such as medication list (3%), code status (2%), lab results (1%), and vital signs with date (0%) were rarely present, Dr. Starmer said.
All of the aforementioned items, except for chief complaint (99%) and patient summary (95%), were present in 100% of reports postintervention, likely because the data were being automatically imported, she said. Improvements also were observed for more complex items such as to-do lists (29% pre vs. 82% post) and contingency plans (18% vs. 47%), likely because of computer prompting. All comparisons, except chief complaint, were statistically significant (P less than .05).
In Unit 2, significant gains were observed in the inclusion of contingency plans (25% pre vs. 40% post), lab results with date (7% vs. 17%), and vital signs with date (2% vs. 9%) (P less than .05 for all), Dr. Starmer said.
Upward trends were noted for the routinely included items of chief complaint (96% vs. 100%), weight (99% vs. 100%), and patient summary (96% vs. 100%), with slight declines in allergies (94% vs. 91%) and code status (3% vs. 2%). Name and medical record number were included in 100% of records pre- and postintervention.
Limitations of the study include the inability to control for resident learning over time and seasonal variations, its single-site design, and the inability to sustain the use of the verbal mnemonic following the research period, said Dr. Starmer at the meeting, sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
She went on to say that these limitations will be addressed in I-PASS (IIPE-PRIS Accelerating Safe Sign-Outs), a multicenter study supported by the Initiative for Innovation in Pediatric Education (IIPE) and the Pediatric Research in Inpatient Settings (PRIS) network.
An attendee asked how the team approach to resident-intern verbal signoffs has been impacted by new resident work hours. Dr. Starmer said there was some concern that the new work rules would affect the amount of time spent with each patient, but that this has not changed.
Dr. Starmer and her coauthors reported no relevant financial relationships.
FROM THE PEDIATRIC HOSPITAL MEDICINE 2011 MEETING
Age, Glycopyrrolate Spotted as Pediatric Propofol Risks
KANSAS CITY, MO. – Improved patient selection and reduced use of oral midazolam are being credited with a fivefold decrease in adverse events associated with pediatric propofol sedation.
The rate of respiratory events, airway interventions, and any other event dropped from 11% in 2005 to 2.3% in 2009 among patients sedated with propofol (Diprivan) by hospitalists in an ambulatory procedure center at St. Louis Children’s Hospital, according to an analysis of 1,649 patients.
Multivariate analysis showed that age more than 12 years (odds ratio 4.0), American Society of Anesthesiologists (ASA) 3 status (OR 2.3), premedication with oral midazolam (Versed) (OR 1.9), use of glycopyrrolate (Robinul) during sedation (OR 4.7), and history of snoring (OR 2.4) increased the likelihood of respiratory events.
All of these, except for ASA 3 status, also significantly increased the likelihood for airway interventions, Dr. Mythili Srinivasan and her colleagues reported in a poster at Pediatric Hospital Medicine 2011.
Although snoring and midazolam were the two most prevalent risk factors, the use of adjuvant glycopyrrolate was found to be the single greatest risk factor for both respiratory events (OR 4.7) and airway interventions (OR 5.8).
"However, a history of recent or ongoing upper respiratory infection was not a significant risk factor, raising the question of whether glycopyrrolate itself may be a risk factor for propofol sedation similar to that reported for ketamine sedations," said Dr. Srinivasan, a pediatric hospitalist at Washington University in St. Louis, Missouri.
A previous analysis of risk factors for ketamine sedation also showed that use of glycopyrrolate was associated with significantly more airway and respiratory complications in the emergency department than either atropine or no anticholinergic in an individual patient-data meta-analysis of 8,282 children (Ann. Emerg. Med. 2009;54:158-68).
In the current analysis, propofol dose, procedure time, and the diagnostic category of the patients were not significant risk factors for adverse events.
The higher likelihood of respiratory events and airway interventions in patients 12 years or older was described by the authors as an unexpected finding. The increase was not related to higher doses of propofol normalized to body weight, nor did older patients have a higher incidence of risk factors such as history of snoring, ASA 3 status, premedication with midazolam, or use of glycopyrrolate compared with patients younger than 12 years. Further studies are needed to explain the surprise finding, the authors suggested.
Declines during the study period in three of the five risk factors – age more than 12 years, ASA 3 status, and premedication with oral midazolam – were significantly correlated with the overall decrease in rates of adverse events, Dr. Srinivasan reported at the meeting, which was sponsored by the Society of Hospital Medicine, American Academy of Pediatrics, and Academic Pediatric Association.
The percentage of patients older than 12 years dropped twofold from 8% in 2005 to 3.8% in 2009, while patients in the ASA 3 category decreased from 10% to 2%, and the use of oral midazolam fell from 41% to 3.3%.
Finally, the increased expertise of hospitalists in the use of propofol also likely contributed to the decrease in adverse events, she noted.
Dr. Srinivasan and her coauthors reported no relevant financial disclosures.
KANSAS CITY, MO. – Improved patient selection and reduced use of oral midazolam are being credited with a fivefold decrease in adverse events associated with pediatric propofol sedation.
The rate of respiratory events, airway interventions, and any other event dropped from 11% in 2005 to 2.3% in 2009 among patients sedated with propofol (Diprivan) by hospitalists in an ambulatory procedure center at St. Louis Children’s Hospital, according to an analysis of 1,649 patients.
Multivariate analysis showed that age more than 12 years (odds ratio 4.0), American Society of Anesthesiologists (ASA) 3 status (OR 2.3), premedication with oral midazolam (Versed) (OR 1.9), use of glycopyrrolate (Robinul) during sedation (OR 4.7), and history of snoring (OR 2.4) increased the likelihood of respiratory events.
All of these, except for ASA 3 status, also significantly increased the likelihood for airway interventions, Dr. Mythili Srinivasan and her colleagues reported in a poster at Pediatric Hospital Medicine 2011.
Although snoring and midazolam were the two most prevalent risk factors, the use of adjuvant glycopyrrolate was found to be the single greatest risk factor for both respiratory events (OR 4.7) and airway interventions (OR 5.8).
"However, a history of recent or ongoing upper respiratory infection was not a significant risk factor, raising the question of whether glycopyrrolate itself may be a risk factor for propofol sedation similar to that reported for ketamine sedations," said Dr. Srinivasan, a pediatric hospitalist at Washington University in St. Louis, Missouri.
A previous analysis of risk factors for ketamine sedation also showed that use of glycopyrrolate was associated with significantly more airway and respiratory complications in the emergency department than either atropine or no anticholinergic in an individual patient-data meta-analysis of 8,282 children (Ann. Emerg. Med. 2009;54:158-68).
In the current analysis, propofol dose, procedure time, and the diagnostic category of the patients were not significant risk factors for adverse events.
The higher likelihood of respiratory events and airway interventions in patients 12 years or older was described by the authors as an unexpected finding. The increase was not related to higher doses of propofol normalized to body weight, nor did older patients have a higher incidence of risk factors such as history of snoring, ASA 3 status, premedication with midazolam, or use of glycopyrrolate compared with patients younger than 12 years. Further studies are needed to explain the surprise finding, the authors suggested.
Declines during the study period in three of the five risk factors – age more than 12 years, ASA 3 status, and premedication with oral midazolam – were significantly correlated with the overall decrease in rates of adverse events, Dr. Srinivasan reported at the meeting, which was sponsored by the Society of Hospital Medicine, American Academy of Pediatrics, and Academic Pediatric Association.
The percentage of patients older than 12 years dropped twofold from 8% in 2005 to 3.8% in 2009, while patients in the ASA 3 category decreased from 10% to 2%, and the use of oral midazolam fell from 41% to 3.3%.
Finally, the increased expertise of hospitalists in the use of propofol also likely contributed to the decrease in adverse events, she noted.
Dr. Srinivasan and her coauthors reported no relevant financial disclosures.
KANSAS CITY, MO. – Improved patient selection and reduced use of oral midazolam are being credited with a fivefold decrease in adverse events associated with pediatric propofol sedation.
The rate of respiratory events, airway interventions, and any other event dropped from 11% in 2005 to 2.3% in 2009 among patients sedated with propofol (Diprivan) by hospitalists in an ambulatory procedure center at St. Louis Children’s Hospital, according to an analysis of 1,649 patients.
Multivariate analysis showed that age more than 12 years (odds ratio 4.0), American Society of Anesthesiologists (ASA) 3 status (OR 2.3), premedication with oral midazolam (Versed) (OR 1.9), use of glycopyrrolate (Robinul) during sedation (OR 4.7), and history of snoring (OR 2.4) increased the likelihood of respiratory events.
All of these, except for ASA 3 status, also significantly increased the likelihood for airway interventions, Dr. Mythili Srinivasan and her colleagues reported in a poster at Pediatric Hospital Medicine 2011.
Although snoring and midazolam were the two most prevalent risk factors, the use of adjuvant glycopyrrolate was found to be the single greatest risk factor for both respiratory events (OR 4.7) and airway interventions (OR 5.8).
"However, a history of recent or ongoing upper respiratory infection was not a significant risk factor, raising the question of whether glycopyrrolate itself may be a risk factor for propofol sedation similar to that reported for ketamine sedations," said Dr. Srinivasan, a pediatric hospitalist at Washington University in St. Louis, Missouri.
A previous analysis of risk factors for ketamine sedation also showed that use of glycopyrrolate was associated with significantly more airway and respiratory complications in the emergency department than either atropine or no anticholinergic in an individual patient-data meta-analysis of 8,282 children (Ann. Emerg. Med. 2009;54:158-68).
In the current analysis, propofol dose, procedure time, and the diagnostic category of the patients were not significant risk factors for adverse events.
The higher likelihood of respiratory events and airway interventions in patients 12 years or older was described by the authors as an unexpected finding. The increase was not related to higher doses of propofol normalized to body weight, nor did older patients have a higher incidence of risk factors such as history of snoring, ASA 3 status, premedication with midazolam, or use of glycopyrrolate compared with patients younger than 12 years. Further studies are needed to explain the surprise finding, the authors suggested.
Declines during the study period in three of the five risk factors – age more than 12 years, ASA 3 status, and premedication with oral midazolam – were significantly correlated with the overall decrease in rates of adverse events, Dr. Srinivasan reported at the meeting, which was sponsored by the Society of Hospital Medicine, American Academy of Pediatrics, and Academic Pediatric Association.
The percentage of patients older than 12 years dropped twofold from 8% in 2005 to 3.8% in 2009, while patients in the ASA 3 category decreased from 10% to 2%, and the use of oral midazolam fell from 41% to 3.3%.
Finally, the increased expertise of hospitalists in the use of propofol also likely contributed to the decrease in adverse events, she noted.
Dr. Srinivasan and her coauthors reported no relevant financial disclosures.
FROM THE PEDIATRIC HOSPITAL MEDICINE 2011 MEETING
Dual HER2 Blockade Strategies Advance in Breast Cancer Trials
Efforts to build on the effectiveness of trastuzumab with a dual blockade of the HER2 pathway continue to suggest such a strategy has the potential to enhance outcomes for HER2-positive breast cancer patients.
Genentech announced this summer that the combination of trastuzumab (Herceptin) and docetaxel (Taxotere) with the investigational agent pertuzumab (Omnitarg) met the primary end point of improved progression-free survival in the randomized, double-blind, placebo-controlled phase III CLEOPATRA trial.
A monoclonal antibody, pertuzumab prevents the HER2 receptor from pairing with other HER receptors, including HER1/EGFR, HER3, and HER4, according to Genentech. The Roche-owned company released no data, but said it expects to submit the trial results for "global regulatory approval" this year. A spokesperson said the results will be submitted at "an upcoming medical meeting,"
Trastuzumab emtansine (formerly known as trastuzumab-DM1) is also in ongoing trials, assessing it in various combinations involving trastuzumab, pertuzumab, and lapatinib (Tykerb), the last of which is a dual inhibitor of HER2 and the epidermal growth factor receptor (EGFR). Genentech is developing this experimental antibody-drug conjugate, with Immunogen. It sought accelerated approval based on a positive phase II study last summer but was rebuffed by the Food and Drug Administration on grounds that patients in the trial had not exhausted other therapies.
Since then, the combination of trastuzumab and lapatinib – both approved for HER2-positive breast cancer – was shown to boost complete response rates to 74% when combined with preoperative chemotherapy in a phase II trial reported in June at the annual meeting of the American Society of Clinical Oncology (ASCO).
The study was one of a trio of neoadjuvant trials that evaluated whether complete responses rates could be bettered by adding lapatinib to chemotherapy plus trastuzumab or by combining lapatinib and trastuzumab without chemotherapy, but with the option of endocrine therapy.
Trastuzumab, Lapatinib, and Chemotherapy
Investigators in the phase II CHER-LOB study defined pathological complete response (pCR) as the complete disappearance of invasive residual disease in the breast and axillary nodes. Among 115 evaluable women, the pCR rate increased from 25.7% with chemotherapy plus trastuzumab (arm A) and 27.8% with chemotherapy plus lapatinib (arm B) to 43.1% with the addition of lapatinib to chemotherapy plus trastuzumab (arm C).
The cardiac safety data were reassuring, with no reports of heart failure or significant loss of left ventricular function. Diarrhea, however, was a significant toxicity that caused dose reductions and treatment interruptions in the majority of patients receiving chemotherapy plus lapatinib, acknowledged Dr. Valentina Guarneri of the Modena (Italy) University Hospital. Grade 3 or higher diarrhea was reported in 36% of 39 patients in arm B and 33% of 46 patients in arm C. Permanent lapatinib discontinuation occurred in 25.6% and 13%, respectively.
Because of the high incidence of diarrhea, the protocol was amended to reduce continuous daily dosing of lapatinib from 1,500 mg to 1,250 mg in arm B and from 1,000 mg to 750 mg in arm C. After the amendment, dose reductions fell from 80% to 54% in arm B and from 55% to 34% in arm C, she said.
Chemotherapy consisted of weekly paclitaxel 80 mg/m2 for 12 weeks followed by four cycles of fluorouracil 600 mg/m2 plus epirubicin 75 mg/m2 plus cyclophosphamide 600 mg/m2 every 3 weeks (FE75C). Trastuzumab 4 mg/m2 was given as a loading dose, followed by 2 mg/m2 every week.
A Similar Protocol but With Modifications
The second trial extended the 24-week preoperative FE75C chemotherapy sequence to 26 weeks after a 2-week run-in period of anti-HER2 therapy and used weekly paclitaxel after, rather than before, FE75C. Also, the daily lapatinib dose was set at 1,250 mg in arm B and increased from 750 mg-1,000 mg in arm C. Trastuzumab dosing was the same as above.
Using this protocol, pCR rates among 78 evaluable women reached 54% with chemotherapy plus trastuzumab (arm A), 45% with chemotherapy plus lapatinib (arm B), and 74% with double HER2 blockade plus chemotherapy (arm C), reported Dr. Frankie Ann Holmes with Texas Oncology in Houston.
Patients had stage II-III biopsy proven invasive HER2-positive breast cancer, whereas CHER-LOB limited enrollment to stage II-IIIA disease.
Headed in the Right Direction
Invited discussant Dr. Gunther von Minckwitz, with the German Breast Group, said that despite the small patient numbers, the pCR rates were quite impressive, especially for the study presented by Dr. Holmes, and are in range with previous trials using longer chemotherapy regimens.
The difference in pCR rates between the two studies could be explained by the small sample sizes and differences in patient characteristics. "Still the tendency is going in the same direction," he observed.
Dr. von Minckwitz urged caution in using pCR as an end point, however, particularly among patients with HER2-positive and estrogen receptor (ER)-positive disease. His own meta-analysis presented at ASCO of seven German neoadjuvant breast trials using anthracycline/taxane regimens with or without trastuzumab, showed that pCR strongly correlates with disease-free survival in higher risk groups such as ductal, HER2-positive, and ER-negative disease, but not in luminal A-like and ER-positive/HER2-positive tumors (Abstract 1028).
Lapatinib vs. Trastuzumab Sans Chemo
The third study used neoadjuvant lapatinib 1,000 mg daily plus trastuzumab at a loading dose of 4 mg/kg followed by 2 mg/kg weekly, without chemotherapy, in women with large HER2-positive tumors (median 6 cm; range 1.5-30 cm). In an effort to overcome estrogen receptor crosstalk, ER-positive patients also received letrozole (Femara), plus goserelin, if premenopausal.
A pCR, defined as no invasive cancer in the breast only, was achieved by 28% of the 64 evaluable patients – 21% of ER-positive and 40% of ER-negative patients, Dr. Jenny C. Chang reported on behalf of the Translational Breast Cancer Research Consortium (TBCRC) 006 investigators. No patient progressed during the 12 weeks of targeted therapy alone.
The secondary end point of residual disease of less than 1 cm (near pCR) was seen in 56% of ER-positive and 48% of ER-negative patients, suggesting that these patients may benefit from longer therapy, she said. In all, 62% of patients were ER positive, 62% had a tumor size greater than 5 cm, and 54% were premenopausal.
The regimen in the phase II TBCRC 006 study was very well tolerated, with only five patients discontinuing therapy and 3 reports of grade 3/4 abnormal liver function tests that completely resolved.
Further study is needed before suggesting that select patients with HER2-positive tumors may not need neoadjuvant chemotherapy, said Dr. Chang, director of the Methodist Cancer Center in Houston.
The data compare favorably with those reported at the 2010 San Antonio Breast Cancer Symposium from the phase II NEOSPHERE trial, in which 17% of patients overall, 6% of ER-positive and 29% of ER-negative patients experienced a pCR after 12 weeks of neoadjuvant trastuzumab plus the monoclonal antibody, pertuzumab, Dr. von Minckwitz observed.
New insights are expected from three upcoming neoadjuvant chemotherapy trials in HER2-positive breast cancer: Cancer and Leukemia Group B 40601, European Organization for Research and Treatment of Cancer 10054 and a National Surgical Adjuvant Breast and Bowel Project B-41.
Molecular Profiles Begin to Emerge
Dr. Holmes and her colleagues conducted a variety of molecular analyses in tumor samples from 49 patients before and after anti-HER2 therapy. She reported that their findings indicate the following:
• Baseline phosphorylated EGFR tyrosine1068 is a marker of nonresponse to lapatinib.
• Baseline activation of autophagy correlates with no pCR to all therapies.
• Baseline ratios of phosphorylated Forkhead box O to PTEN and PI3K correlate with response.
• Phosphorylated STAT5 after trastuzumab correlates with pCR.
The molecular profiles suggest that pCR tumors rely on a truncated, rudimentary social network, while resistant tumors use a far more extensive network of autophagy and stem cell–regulated pathways to evade therapy and are thus harder to kill, Dr. Holmes said.
"Despite the limitations, this exploratory analysis shows that the pathway to accelerating the cure of breast cancer is available in every oncologist’s office if she or he can network to tissue-based research trials," she said.
The CHER-LOB investigators tested tumor samples for p95HER2, which is expressed in approximately 30% of HER2-positive breast cancer patients and thought to confer resistance to trastuzumab. No significant differences were observed in all treatment arms in pCR rates based on p95HER2 expression, Dr. Guarneri said.
The p95HER2 results contradict earlier research, observed Dr. von Minckwitz. "We really have to design these biomarker studies very cautiously and the first [issue] we have to address is that we have to have sufficient numbers to study," he added.
CHER-LOB was supported by GlaxoSmithKline. Dr. Guarneri reports no conflicts. Dr. Holmes reports a consultant-advisory role with Phillips Home Monitoring and honoraria from Genentech and Novartis. Dr. Chang reports no relevant conflicts. Dr. Von Minckwitz reports honoraria from Amgen, Roche, and Sanofi-Aventis and research funding from Abraxis BioScience, Amgen, Bayer, GSK, Novartis, Pfizer, Roche, Sanofi-Aventis, and bioMérieux.
Efforts to build on the effectiveness of trastuzumab with a dual blockade of the HER2 pathway continue to suggest such a strategy has the potential to enhance outcomes for HER2-positive breast cancer patients.
Genentech announced this summer that the combination of trastuzumab (Herceptin) and docetaxel (Taxotere) with the investigational agent pertuzumab (Omnitarg) met the primary end point of improved progression-free survival in the randomized, double-blind, placebo-controlled phase III CLEOPATRA trial.
A monoclonal antibody, pertuzumab prevents the HER2 receptor from pairing with other HER receptors, including HER1/EGFR, HER3, and HER4, according to Genentech. The Roche-owned company released no data, but said it expects to submit the trial results for "global regulatory approval" this year. A spokesperson said the results will be submitted at "an upcoming medical meeting,"
Trastuzumab emtansine (formerly known as trastuzumab-DM1) is also in ongoing trials, assessing it in various combinations involving trastuzumab, pertuzumab, and lapatinib (Tykerb), the last of which is a dual inhibitor of HER2 and the epidermal growth factor receptor (EGFR). Genentech is developing this experimental antibody-drug conjugate, with Immunogen. It sought accelerated approval based on a positive phase II study last summer but was rebuffed by the Food and Drug Administration on grounds that patients in the trial had not exhausted other therapies.
Since then, the combination of trastuzumab and lapatinib – both approved for HER2-positive breast cancer – was shown to boost complete response rates to 74% when combined with preoperative chemotherapy in a phase II trial reported in June at the annual meeting of the American Society of Clinical Oncology (ASCO).
The study was one of a trio of neoadjuvant trials that evaluated whether complete responses rates could be bettered by adding lapatinib to chemotherapy plus trastuzumab or by combining lapatinib and trastuzumab without chemotherapy, but with the option of endocrine therapy.
Trastuzumab, Lapatinib, and Chemotherapy
Investigators in the phase II CHER-LOB study defined pathological complete response (pCR) as the complete disappearance of invasive residual disease in the breast and axillary nodes. Among 115 evaluable women, the pCR rate increased from 25.7% with chemotherapy plus trastuzumab (arm A) and 27.8% with chemotherapy plus lapatinib (arm B) to 43.1% with the addition of lapatinib to chemotherapy plus trastuzumab (arm C).
The cardiac safety data were reassuring, with no reports of heart failure or significant loss of left ventricular function. Diarrhea, however, was a significant toxicity that caused dose reductions and treatment interruptions in the majority of patients receiving chemotherapy plus lapatinib, acknowledged Dr. Valentina Guarneri of the Modena (Italy) University Hospital. Grade 3 or higher diarrhea was reported in 36% of 39 patients in arm B and 33% of 46 patients in arm C. Permanent lapatinib discontinuation occurred in 25.6% and 13%, respectively.
Because of the high incidence of diarrhea, the protocol was amended to reduce continuous daily dosing of lapatinib from 1,500 mg to 1,250 mg in arm B and from 1,000 mg to 750 mg in arm C. After the amendment, dose reductions fell from 80% to 54% in arm B and from 55% to 34% in arm C, she said.
Chemotherapy consisted of weekly paclitaxel 80 mg/m2 for 12 weeks followed by four cycles of fluorouracil 600 mg/m2 plus epirubicin 75 mg/m2 plus cyclophosphamide 600 mg/m2 every 3 weeks (FE75C). Trastuzumab 4 mg/m2 was given as a loading dose, followed by 2 mg/m2 every week.
A Similar Protocol but With Modifications
The second trial extended the 24-week preoperative FE75C chemotherapy sequence to 26 weeks after a 2-week run-in period of anti-HER2 therapy and used weekly paclitaxel after, rather than before, FE75C. Also, the daily lapatinib dose was set at 1,250 mg in arm B and increased from 750 mg-1,000 mg in arm C. Trastuzumab dosing was the same as above.
Using this protocol, pCR rates among 78 evaluable women reached 54% with chemotherapy plus trastuzumab (arm A), 45% with chemotherapy plus lapatinib (arm B), and 74% with double HER2 blockade plus chemotherapy (arm C), reported Dr. Frankie Ann Holmes with Texas Oncology in Houston.
Patients had stage II-III biopsy proven invasive HER2-positive breast cancer, whereas CHER-LOB limited enrollment to stage II-IIIA disease.
Headed in the Right Direction
Invited discussant Dr. Gunther von Minckwitz, with the German Breast Group, said that despite the small patient numbers, the pCR rates were quite impressive, especially for the study presented by Dr. Holmes, and are in range with previous trials using longer chemotherapy regimens.
The difference in pCR rates between the two studies could be explained by the small sample sizes and differences in patient characteristics. "Still the tendency is going in the same direction," he observed.
Dr. von Minckwitz urged caution in using pCR as an end point, however, particularly among patients with HER2-positive and estrogen receptor (ER)-positive disease. His own meta-analysis presented at ASCO of seven German neoadjuvant breast trials using anthracycline/taxane regimens with or without trastuzumab, showed that pCR strongly correlates with disease-free survival in higher risk groups such as ductal, HER2-positive, and ER-negative disease, but not in luminal A-like and ER-positive/HER2-positive tumors (Abstract 1028).
Lapatinib vs. Trastuzumab Sans Chemo
The third study used neoadjuvant lapatinib 1,000 mg daily plus trastuzumab at a loading dose of 4 mg/kg followed by 2 mg/kg weekly, without chemotherapy, in women with large HER2-positive tumors (median 6 cm; range 1.5-30 cm). In an effort to overcome estrogen receptor crosstalk, ER-positive patients also received letrozole (Femara), plus goserelin, if premenopausal.
A pCR, defined as no invasive cancer in the breast only, was achieved by 28% of the 64 evaluable patients – 21% of ER-positive and 40% of ER-negative patients, Dr. Jenny C. Chang reported on behalf of the Translational Breast Cancer Research Consortium (TBCRC) 006 investigators. No patient progressed during the 12 weeks of targeted therapy alone.
The secondary end point of residual disease of less than 1 cm (near pCR) was seen in 56% of ER-positive and 48% of ER-negative patients, suggesting that these patients may benefit from longer therapy, she said. In all, 62% of patients were ER positive, 62% had a tumor size greater than 5 cm, and 54% were premenopausal.
The regimen in the phase II TBCRC 006 study was very well tolerated, with only five patients discontinuing therapy and 3 reports of grade 3/4 abnormal liver function tests that completely resolved.
Further study is needed before suggesting that select patients with HER2-positive tumors may not need neoadjuvant chemotherapy, said Dr. Chang, director of the Methodist Cancer Center in Houston.
The data compare favorably with those reported at the 2010 San Antonio Breast Cancer Symposium from the phase II NEOSPHERE trial, in which 17% of patients overall, 6% of ER-positive and 29% of ER-negative patients experienced a pCR after 12 weeks of neoadjuvant trastuzumab plus the monoclonal antibody, pertuzumab, Dr. von Minckwitz observed.
New insights are expected from three upcoming neoadjuvant chemotherapy trials in HER2-positive breast cancer: Cancer and Leukemia Group B 40601, European Organization for Research and Treatment of Cancer 10054 and a National Surgical Adjuvant Breast and Bowel Project B-41.
Molecular Profiles Begin to Emerge
Dr. Holmes and her colleagues conducted a variety of molecular analyses in tumor samples from 49 patients before and after anti-HER2 therapy. She reported that their findings indicate the following:
• Baseline phosphorylated EGFR tyrosine1068 is a marker of nonresponse to lapatinib.
• Baseline activation of autophagy correlates with no pCR to all therapies.
• Baseline ratios of phosphorylated Forkhead box O to PTEN and PI3K correlate with response.
• Phosphorylated STAT5 after trastuzumab correlates with pCR.
The molecular profiles suggest that pCR tumors rely on a truncated, rudimentary social network, while resistant tumors use a far more extensive network of autophagy and stem cell–regulated pathways to evade therapy and are thus harder to kill, Dr. Holmes said.
"Despite the limitations, this exploratory analysis shows that the pathway to accelerating the cure of breast cancer is available in every oncologist’s office if she or he can network to tissue-based research trials," she said.
The CHER-LOB investigators tested tumor samples for p95HER2, which is expressed in approximately 30% of HER2-positive breast cancer patients and thought to confer resistance to trastuzumab. No significant differences were observed in all treatment arms in pCR rates based on p95HER2 expression, Dr. Guarneri said.
The p95HER2 results contradict earlier research, observed Dr. von Minckwitz. "We really have to design these biomarker studies very cautiously and the first [issue] we have to address is that we have to have sufficient numbers to study," he added.
CHER-LOB was supported by GlaxoSmithKline. Dr. Guarneri reports no conflicts. Dr. Holmes reports a consultant-advisory role with Phillips Home Monitoring and honoraria from Genentech and Novartis. Dr. Chang reports no relevant conflicts. Dr. Von Minckwitz reports honoraria from Amgen, Roche, and Sanofi-Aventis and research funding from Abraxis BioScience, Amgen, Bayer, GSK, Novartis, Pfizer, Roche, Sanofi-Aventis, and bioMérieux.
Efforts to build on the effectiveness of trastuzumab with a dual blockade of the HER2 pathway continue to suggest such a strategy has the potential to enhance outcomes for HER2-positive breast cancer patients.
Genentech announced this summer that the combination of trastuzumab (Herceptin) and docetaxel (Taxotere) with the investigational agent pertuzumab (Omnitarg) met the primary end point of improved progression-free survival in the randomized, double-blind, placebo-controlled phase III CLEOPATRA trial.
A monoclonal antibody, pertuzumab prevents the HER2 receptor from pairing with other HER receptors, including HER1/EGFR, HER3, and HER4, according to Genentech. The Roche-owned company released no data, but said it expects to submit the trial results for "global regulatory approval" this year. A spokesperson said the results will be submitted at "an upcoming medical meeting,"
Trastuzumab emtansine (formerly known as trastuzumab-DM1) is also in ongoing trials, assessing it in various combinations involving trastuzumab, pertuzumab, and lapatinib (Tykerb), the last of which is a dual inhibitor of HER2 and the epidermal growth factor receptor (EGFR). Genentech is developing this experimental antibody-drug conjugate, with Immunogen. It sought accelerated approval based on a positive phase II study last summer but was rebuffed by the Food and Drug Administration on grounds that patients in the trial had not exhausted other therapies.
Since then, the combination of trastuzumab and lapatinib – both approved for HER2-positive breast cancer – was shown to boost complete response rates to 74% when combined with preoperative chemotherapy in a phase II trial reported in June at the annual meeting of the American Society of Clinical Oncology (ASCO).
The study was one of a trio of neoadjuvant trials that evaluated whether complete responses rates could be bettered by adding lapatinib to chemotherapy plus trastuzumab or by combining lapatinib and trastuzumab without chemotherapy, but with the option of endocrine therapy.
Trastuzumab, Lapatinib, and Chemotherapy
Investigators in the phase II CHER-LOB study defined pathological complete response (pCR) as the complete disappearance of invasive residual disease in the breast and axillary nodes. Among 115 evaluable women, the pCR rate increased from 25.7% with chemotherapy plus trastuzumab (arm A) and 27.8% with chemotherapy plus lapatinib (arm B) to 43.1% with the addition of lapatinib to chemotherapy plus trastuzumab (arm C).
The cardiac safety data were reassuring, with no reports of heart failure or significant loss of left ventricular function. Diarrhea, however, was a significant toxicity that caused dose reductions and treatment interruptions in the majority of patients receiving chemotherapy plus lapatinib, acknowledged Dr. Valentina Guarneri of the Modena (Italy) University Hospital. Grade 3 or higher diarrhea was reported in 36% of 39 patients in arm B and 33% of 46 patients in arm C. Permanent lapatinib discontinuation occurred in 25.6% and 13%, respectively.
Because of the high incidence of diarrhea, the protocol was amended to reduce continuous daily dosing of lapatinib from 1,500 mg to 1,250 mg in arm B and from 1,000 mg to 750 mg in arm C. After the amendment, dose reductions fell from 80% to 54% in arm B and from 55% to 34% in arm C, she said.
Chemotherapy consisted of weekly paclitaxel 80 mg/m2 for 12 weeks followed by four cycles of fluorouracil 600 mg/m2 plus epirubicin 75 mg/m2 plus cyclophosphamide 600 mg/m2 every 3 weeks (FE75C). Trastuzumab 4 mg/m2 was given as a loading dose, followed by 2 mg/m2 every week.
A Similar Protocol but With Modifications
The second trial extended the 24-week preoperative FE75C chemotherapy sequence to 26 weeks after a 2-week run-in period of anti-HER2 therapy and used weekly paclitaxel after, rather than before, FE75C. Also, the daily lapatinib dose was set at 1,250 mg in arm B and increased from 750 mg-1,000 mg in arm C. Trastuzumab dosing was the same as above.
Using this protocol, pCR rates among 78 evaluable women reached 54% with chemotherapy plus trastuzumab (arm A), 45% with chemotherapy plus lapatinib (arm B), and 74% with double HER2 blockade plus chemotherapy (arm C), reported Dr. Frankie Ann Holmes with Texas Oncology in Houston.
Patients had stage II-III biopsy proven invasive HER2-positive breast cancer, whereas CHER-LOB limited enrollment to stage II-IIIA disease.
Headed in the Right Direction
Invited discussant Dr. Gunther von Minckwitz, with the German Breast Group, said that despite the small patient numbers, the pCR rates were quite impressive, especially for the study presented by Dr. Holmes, and are in range with previous trials using longer chemotherapy regimens.
The difference in pCR rates between the two studies could be explained by the small sample sizes and differences in patient characteristics. "Still the tendency is going in the same direction," he observed.
Dr. von Minckwitz urged caution in using pCR as an end point, however, particularly among patients with HER2-positive and estrogen receptor (ER)-positive disease. His own meta-analysis presented at ASCO of seven German neoadjuvant breast trials using anthracycline/taxane regimens with or without trastuzumab, showed that pCR strongly correlates with disease-free survival in higher risk groups such as ductal, HER2-positive, and ER-negative disease, but not in luminal A-like and ER-positive/HER2-positive tumors (Abstract 1028).
Lapatinib vs. Trastuzumab Sans Chemo
The third study used neoadjuvant lapatinib 1,000 mg daily plus trastuzumab at a loading dose of 4 mg/kg followed by 2 mg/kg weekly, without chemotherapy, in women with large HER2-positive tumors (median 6 cm; range 1.5-30 cm). In an effort to overcome estrogen receptor crosstalk, ER-positive patients also received letrozole (Femara), plus goserelin, if premenopausal.
A pCR, defined as no invasive cancer in the breast only, was achieved by 28% of the 64 evaluable patients – 21% of ER-positive and 40% of ER-negative patients, Dr. Jenny C. Chang reported on behalf of the Translational Breast Cancer Research Consortium (TBCRC) 006 investigators. No patient progressed during the 12 weeks of targeted therapy alone.
The secondary end point of residual disease of less than 1 cm (near pCR) was seen in 56% of ER-positive and 48% of ER-negative patients, suggesting that these patients may benefit from longer therapy, she said. In all, 62% of patients were ER positive, 62% had a tumor size greater than 5 cm, and 54% were premenopausal.
The regimen in the phase II TBCRC 006 study was very well tolerated, with only five patients discontinuing therapy and 3 reports of grade 3/4 abnormal liver function tests that completely resolved.
Further study is needed before suggesting that select patients with HER2-positive tumors may not need neoadjuvant chemotherapy, said Dr. Chang, director of the Methodist Cancer Center in Houston.
The data compare favorably with those reported at the 2010 San Antonio Breast Cancer Symposium from the phase II NEOSPHERE trial, in which 17% of patients overall, 6% of ER-positive and 29% of ER-negative patients experienced a pCR after 12 weeks of neoadjuvant trastuzumab plus the monoclonal antibody, pertuzumab, Dr. von Minckwitz observed.
New insights are expected from three upcoming neoadjuvant chemotherapy trials in HER2-positive breast cancer: Cancer and Leukemia Group B 40601, European Organization for Research and Treatment of Cancer 10054 and a National Surgical Adjuvant Breast and Bowel Project B-41.
Molecular Profiles Begin to Emerge
Dr. Holmes and her colleagues conducted a variety of molecular analyses in tumor samples from 49 patients before and after anti-HER2 therapy. She reported that their findings indicate the following:
• Baseline phosphorylated EGFR tyrosine1068 is a marker of nonresponse to lapatinib.
• Baseline activation of autophagy correlates with no pCR to all therapies.
• Baseline ratios of phosphorylated Forkhead box O to PTEN and PI3K correlate with response.
• Phosphorylated STAT5 after trastuzumab correlates with pCR.
The molecular profiles suggest that pCR tumors rely on a truncated, rudimentary social network, while resistant tumors use a far more extensive network of autophagy and stem cell–regulated pathways to evade therapy and are thus harder to kill, Dr. Holmes said.
"Despite the limitations, this exploratory analysis shows that the pathway to accelerating the cure of breast cancer is available in every oncologist’s office if she or he can network to tissue-based research trials," she said.
The CHER-LOB investigators tested tumor samples for p95HER2, which is expressed in approximately 30% of HER2-positive breast cancer patients and thought to confer resistance to trastuzumab. No significant differences were observed in all treatment arms in pCR rates based on p95HER2 expression, Dr. Guarneri said.
The p95HER2 results contradict earlier research, observed Dr. von Minckwitz. "We really have to design these biomarker studies very cautiously and the first [issue] we have to address is that we have to have sufficient numbers to study," he added.
CHER-LOB was supported by GlaxoSmithKline. Dr. Guarneri reports no conflicts. Dr. Holmes reports a consultant-advisory role with Phillips Home Monitoring and honoraria from Genentech and Novartis. Dr. Chang reports no relevant conflicts. Dr. Von Minckwitz reports honoraria from Amgen, Roche, and Sanofi-Aventis and research funding from Abraxis BioScience, Amgen, Bayer, GSK, Novartis, Pfizer, Roche, Sanofi-Aventis, and bioMérieux.