User login
Acellular pertussis vaccines don’t prevent transmission
Acellular pertussis vaccines prevent children from developing pertussis, but they do not prevent those children from becoming colonized with Bordetella pertussis or from transmitting the organism to others who are unvaccinated, according to a report published online Nov. 25 in Proceedings of the National Academy of Sciences.
This finding provides a plausible explanation for the resurgence of pertussis disease in the United States and in other industrialized countries, despite vaccine coverage in excess of 95%, said Dr. Jason M. Warfel of the division of bacterial, parasitic, and allergenic products, Center for Biologics Evaluation and Research, Food and Drug Administration, and his associates.
This resurgence in symptomatic pertussis was noted at approximately the same time that whole-cell vaccines were replaced with acellular vaccines in the 1990s, and it continues to the present day. There were 42,000 reported cases in the United States last year, which represents a 50-year high.
Until now, the cause of the resurgence has been uncertain. Researchers have proposed that perhaps the acellular vaccines are less effective than the whole-cell vaccines are or that the immunity they confer waned more quickly than anticipated. But it was nearly impossible to study pertussis pathogenesis in humans because of ethical considerations as well as logistical problems, and there is no animal model that adequately mimics human disease.
Dr. Warfel and his associates were able to develop a nonhuman primate model of pertussis using baboons, which closely corresponded to human pertussis disease. Using this model, they found that the acellular vaccine, whole-cell vaccine, and natural infection all induced comparably high antibody titers. This indicates that both vaccines are immunogenic and that the pertussis resurgence is not attributable to low antibody titers.
Both vaccines protected baboon subjects from developing severe symptomatic pertussis, but neither one prevented them from becoming colonized with the organism. However, baboons that received the acellular vaccine took twice as long to clear the organism from their systems as those that had received the whole-cell vaccine.
And most important, baboons that received the acellular vaccine readily transmitted B. pertussis to unvaccinated cage mates during this relatively long interval. "We hypothesized ... that [children who receive acellular pertussis vaccinations] can act as asymptomatic or mildly symptomatic carriers and contribute significantly to transmission in the [general] population," Dr. Warfel and his colleagues said (Proceed. Natl. Acad. Sci. 2013 [doi:10.1073/pnas.1314688110]).
This finding indicates that "cocooning" – vaccinating people who have contact with infants who have not completed the full series of the acellular vaccine, to prevent transmission to those infants – is unlikely to reduce the burden of pertussis in infants, they noted.
The investigators also found key differences in T-cell immune response to the two vaccines. Animals that received the whole-cell pertussis vaccine or had previously had severe pertussis disease possessed strong, pertussis-specific T helper 17 (Th17) memory and T helper 1 (Th1) memory, but those that received the acellular vaccine instead had strong, pertussis-specific Th1/Th2 memory. Th17 is a recently identified T cell that "specializes in controlling extracellular bacterial infections at mucosal surfaces through stimulating neutrophil recruitment."
"Although we have not definitively shown that Th17 cells are required for B. pertussis clearance, this correlation is consistent with the role these [Th17] cells play in fighting extracellular bacterial infections at mucosal surfaces by inducing neutrophil chemotaxis," they added.
No financial conflicts of interest were reported. This work was funded by the FDA and NIH/National Institute of Allergy and Infectious Diseases.
Acellular pertussis vaccines prevent children from developing pertussis, but they do not prevent those children from becoming colonized with Bordetella pertussis or from transmitting the organism to others who are unvaccinated, according to a report published online Nov. 25 in Proceedings of the National Academy of Sciences.
This finding provides a plausible explanation for the resurgence of pertussis disease in the United States and in other industrialized countries, despite vaccine coverage in excess of 95%, said Dr. Jason M. Warfel of the division of bacterial, parasitic, and allergenic products, Center for Biologics Evaluation and Research, Food and Drug Administration, and his associates.
This resurgence in symptomatic pertussis was noted at approximately the same time that whole-cell vaccines were replaced with acellular vaccines in the 1990s, and it continues to the present day. There were 42,000 reported cases in the United States last year, which represents a 50-year high.
Until now, the cause of the resurgence has been uncertain. Researchers have proposed that perhaps the acellular vaccines are less effective than the whole-cell vaccines are or that the immunity they confer waned more quickly than anticipated. But it was nearly impossible to study pertussis pathogenesis in humans because of ethical considerations as well as logistical problems, and there is no animal model that adequately mimics human disease.
Dr. Warfel and his associates were able to develop a nonhuman primate model of pertussis using baboons, which closely corresponded to human pertussis disease. Using this model, they found that the acellular vaccine, whole-cell vaccine, and natural infection all induced comparably high antibody titers. This indicates that both vaccines are immunogenic and that the pertussis resurgence is not attributable to low antibody titers.
Both vaccines protected baboon subjects from developing severe symptomatic pertussis, but neither one prevented them from becoming colonized with the organism. However, baboons that received the acellular vaccine took twice as long to clear the organism from their systems as those that had received the whole-cell vaccine.
And most important, baboons that received the acellular vaccine readily transmitted B. pertussis to unvaccinated cage mates during this relatively long interval. "We hypothesized ... that [children who receive acellular pertussis vaccinations] can act as asymptomatic or mildly symptomatic carriers and contribute significantly to transmission in the [general] population," Dr. Warfel and his colleagues said (Proceed. Natl. Acad. Sci. 2013 [doi:10.1073/pnas.1314688110]).
This finding indicates that "cocooning" – vaccinating people who have contact with infants who have not completed the full series of the acellular vaccine, to prevent transmission to those infants – is unlikely to reduce the burden of pertussis in infants, they noted.
The investigators also found key differences in T-cell immune response to the two vaccines. Animals that received the whole-cell pertussis vaccine or had previously had severe pertussis disease possessed strong, pertussis-specific T helper 17 (Th17) memory and T helper 1 (Th1) memory, but those that received the acellular vaccine instead had strong, pertussis-specific Th1/Th2 memory. Th17 is a recently identified T cell that "specializes in controlling extracellular bacterial infections at mucosal surfaces through stimulating neutrophil recruitment."
"Although we have not definitively shown that Th17 cells are required for B. pertussis clearance, this correlation is consistent with the role these [Th17] cells play in fighting extracellular bacterial infections at mucosal surfaces by inducing neutrophil chemotaxis," they added.
No financial conflicts of interest were reported. This work was funded by the FDA and NIH/National Institute of Allergy and Infectious Diseases.
Acellular pertussis vaccines prevent children from developing pertussis, but they do not prevent those children from becoming colonized with Bordetella pertussis or from transmitting the organism to others who are unvaccinated, according to a report published online Nov. 25 in Proceedings of the National Academy of Sciences.
This finding provides a plausible explanation for the resurgence of pertussis disease in the United States and in other industrialized countries, despite vaccine coverage in excess of 95%, said Dr. Jason M. Warfel of the division of bacterial, parasitic, and allergenic products, Center for Biologics Evaluation and Research, Food and Drug Administration, and his associates.
This resurgence in symptomatic pertussis was noted at approximately the same time that whole-cell vaccines were replaced with acellular vaccines in the 1990s, and it continues to the present day. There were 42,000 reported cases in the United States last year, which represents a 50-year high.
Until now, the cause of the resurgence has been uncertain. Researchers have proposed that perhaps the acellular vaccines are less effective than the whole-cell vaccines are or that the immunity they confer waned more quickly than anticipated. But it was nearly impossible to study pertussis pathogenesis in humans because of ethical considerations as well as logistical problems, and there is no animal model that adequately mimics human disease.
Dr. Warfel and his associates were able to develop a nonhuman primate model of pertussis using baboons, which closely corresponded to human pertussis disease. Using this model, they found that the acellular vaccine, whole-cell vaccine, and natural infection all induced comparably high antibody titers. This indicates that both vaccines are immunogenic and that the pertussis resurgence is not attributable to low antibody titers.
Both vaccines protected baboon subjects from developing severe symptomatic pertussis, but neither one prevented them from becoming colonized with the organism. However, baboons that received the acellular vaccine took twice as long to clear the organism from their systems as those that had received the whole-cell vaccine.
And most important, baboons that received the acellular vaccine readily transmitted B. pertussis to unvaccinated cage mates during this relatively long interval. "We hypothesized ... that [children who receive acellular pertussis vaccinations] can act as asymptomatic or mildly symptomatic carriers and contribute significantly to transmission in the [general] population," Dr. Warfel and his colleagues said (Proceed. Natl. Acad. Sci. 2013 [doi:10.1073/pnas.1314688110]).
This finding indicates that "cocooning" – vaccinating people who have contact with infants who have not completed the full series of the acellular vaccine, to prevent transmission to those infants – is unlikely to reduce the burden of pertussis in infants, they noted.
The investigators also found key differences in T-cell immune response to the two vaccines. Animals that received the whole-cell pertussis vaccine or had previously had severe pertussis disease possessed strong, pertussis-specific T helper 17 (Th17) memory and T helper 1 (Th1) memory, but those that received the acellular vaccine instead had strong, pertussis-specific Th1/Th2 memory. Th17 is a recently identified T cell that "specializes in controlling extracellular bacterial infections at mucosal surfaces through stimulating neutrophil recruitment."
"Although we have not definitively shown that Th17 cells are required for B. pertussis clearance, this correlation is consistent with the role these [Th17] cells play in fighting extracellular bacterial infections at mucosal surfaces by inducing neutrophil chemotaxis," they added.
No financial conflicts of interest were reported. This work was funded by the FDA and NIH/National Institute of Allergy and Infectious Diseases.
FROM PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES
Major finding: Baboons that received an acellular pertussis vaccine were twice as slow to clear the organism from their systems as were those that received whole-cell pertussis vaccine, and during that interval, they were colonized with B. pertussis and transmitted it to unvaccinated cage mates.
Data source: A series of laboratory and animal studies testing the hypothesis that acellular pertussis vaccines fail to prevent colonization and transmission of B. pertussis, even though they do prevent vaccinated subjects from developing severe symptomatic pertussis.
Disclosures: No financial conflicts of interest were reported. This work was funded by the FDA and NIH/National Institute of Allergy and Infectious Diseases.
Analysis: No such thing as ‘benign’ obesity
"Metabolically healthy" overweight and obese patients may carry the same short-term risk as normal-weight patients for all-cause mortality and cardiovascular events, but their weight puts them at increased risk over the long term, according to a report published online Dec. 2 in Annals of Internal Medicine.
Several studies have reported finding a subgroup of obese individuals who have normal metabolic features despite their high adiposity, and the researchers have called this condition "benign obesity" or "metabolically healthy obesity." Such patients reportedly have the same or even lower all-cause mortality and CV risk as normal-weight patients. But those studies tended to have short follow-up, making their conclusions somewhat questionable, said Dr. Caroline K. Kramer of the Leadership Sinai Centre for Diabetes, Toronto, and her associates.
The findings from their meta-analysis of eight observational studies indicate that excess weight confers increased risk after 10 years, even in "metabolically healthy" obese patients. "Our results do not support this concept of ‘benign obesity’ and demonstrate that there is no ‘healthy’ pattern of obesity," the researchers wrote (Ann. Intern. Med. 2013 Dec. 2;159 [doi=10.7326/0003-4819-159-11-201312030-00002]).
Dr. Kramer and her colleagues pooled the results of eight studies that examined participants’ metabolic status as well as weight, which had follow-up intervals ranging from 3 years to 30 years. That yielded a sample of 61,386 patients, with 3,988 deaths or CV events.
Metabolic status was categorized as either healthy or unhealthy, depending on factors such as lipid profile, glucose tolerance, blood pressure, insulin resistance, and waist circumference.
In an initial analysis, overweight and obese patients who were metabolically healthy had similar outcomes as metabolically healthy normal-weight patients. But when the analysis was confined to studies with at least 10 years of follow-up, the metabolically healthy obese group had increased mortality and CV risk, with a risk ratio of 1.24.
In addition, all patients who were metabolically unhealthy were at increased risk for death and CV events, regardless of their weight. In fact, normal-weight patients who were metabolically unhealthy carried the same risk of death or CV events as did overweight and obese patients who were metabolically unhealthy. That indicates that normal-weight patients who are metabolically unhealthy are a patient population that merits particular attention in clinical practice, the investigators said.
"Our findings highlight the need for comprehensive evaluation of not only BMI [body mass index] but also metabolic factors for the prediction of future morbidity and mortality," Dr. Kramer and her associates said.
The findings also show that duration of follow-up "is a critical element in evaluating low-risk populations for future events, the researchers noted.
The investigators proposed that in patients erroneously deemed to have "benign obesity" or "metabolically healthy obesity," excess weight is associated with subclinical metabolic and vascular dysfunction that ultimately leads to increased mortality and CV events over time.
Dr. Kramer had no relevant conflicts of interest. One of her coauthors reported receiving grants and fees from Merck and Novo Nordisk.
The meta-analysis provides strong evidence that "healthy obesity" is a myth. And it refutes the idea that, because society cannot afford to treat everyone with obesity, it should prioritize treatment only for those at metabolic risk, said James O. Hill, Ph.D., and Dr. Holly R. Wyatt.
"Accepting that no level of obesity is healthy is an important step toward deciding how best to use our resources and our political will to develop and implement strategies to combat the obesity epidemic," they said.
Dr. Hill and Dr. Wyatt are at the Anschutz Health and Wellness Center at the University of Colorado, Aurora. Dr. Hill reported financial ties to McDonald’s, the Coca-Cola Company, and other companies. Dr. Wyatt reported ties to Atkins, Retrofit, and other companies. These remarks were taken from their editorial accompanying Dr. Kramer’s report (Ann. Intern. Med. 2013 Dec. 2;159:789-90).
The meta-analysis provides strong evidence that "healthy obesity" is a myth. And it refutes the idea that, because society cannot afford to treat everyone with obesity, it should prioritize treatment only for those at metabolic risk, said James O. Hill, Ph.D., and Dr. Holly R. Wyatt.
"Accepting that no level of obesity is healthy is an important step toward deciding how best to use our resources and our political will to develop and implement strategies to combat the obesity epidemic," they said.
Dr. Hill and Dr. Wyatt are at the Anschutz Health and Wellness Center at the University of Colorado, Aurora. Dr. Hill reported financial ties to McDonald’s, the Coca-Cola Company, and other companies. Dr. Wyatt reported ties to Atkins, Retrofit, and other companies. These remarks were taken from their editorial accompanying Dr. Kramer’s report (Ann. Intern. Med. 2013 Dec. 2;159:789-90).
The meta-analysis provides strong evidence that "healthy obesity" is a myth. And it refutes the idea that, because society cannot afford to treat everyone with obesity, it should prioritize treatment only for those at metabolic risk, said James O. Hill, Ph.D., and Dr. Holly R. Wyatt.
"Accepting that no level of obesity is healthy is an important step toward deciding how best to use our resources and our political will to develop and implement strategies to combat the obesity epidemic," they said.
Dr. Hill and Dr. Wyatt are at the Anschutz Health and Wellness Center at the University of Colorado, Aurora. Dr. Hill reported financial ties to McDonald’s, the Coca-Cola Company, and other companies. Dr. Wyatt reported ties to Atkins, Retrofit, and other companies. These remarks were taken from their editorial accompanying Dr. Kramer’s report (Ann. Intern. Med. 2013 Dec. 2;159:789-90).
"Metabolically healthy" overweight and obese patients may carry the same short-term risk as normal-weight patients for all-cause mortality and cardiovascular events, but their weight puts them at increased risk over the long term, according to a report published online Dec. 2 in Annals of Internal Medicine.
Several studies have reported finding a subgroup of obese individuals who have normal metabolic features despite their high adiposity, and the researchers have called this condition "benign obesity" or "metabolically healthy obesity." Such patients reportedly have the same or even lower all-cause mortality and CV risk as normal-weight patients. But those studies tended to have short follow-up, making their conclusions somewhat questionable, said Dr. Caroline K. Kramer of the Leadership Sinai Centre for Diabetes, Toronto, and her associates.
The findings from their meta-analysis of eight observational studies indicate that excess weight confers increased risk after 10 years, even in "metabolically healthy" obese patients. "Our results do not support this concept of ‘benign obesity’ and demonstrate that there is no ‘healthy’ pattern of obesity," the researchers wrote (Ann. Intern. Med. 2013 Dec. 2;159 [doi=10.7326/0003-4819-159-11-201312030-00002]).
Dr. Kramer and her colleagues pooled the results of eight studies that examined participants’ metabolic status as well as weight, which had follow-up intervals ranging from 3 years to 30 years. That yielded a sample of 61,386 patients, with 3,988 deaths or CV events.
Metabolic status was categorized as either healthy or unhealthy, depending on factors such as lipid profile, glucose tolerance, blood pressure, insulin resistance, and waist circumference.
In an initial analysis, overweight and obese patients who were metabolically healthy had similar outcomes as metabolically healthy normal-weight patients. But when the analysis was confined to studies with at least 10 years of follow-up, the metabolically healthy obese group had increased mortality and CV risk, with a risk ratio of 1.24.
In addition, all patients who were metabolically unhealthy were at increased risk for death and CV events, regardless of their weight. In fact, normal-weight patients who were metabolically unhealthy carried the same risk of death or CV events as did overweight and obese patients who were metabolically unhealthy. That indicates that normal-weight patients who are metabolically unhealthy are a patient population that merits particular attention in clinical practice, the investigators said.
"Our findings highlight the need for comprehensive evaluation of not only BMI [body mass index] but also metabolic factors for the prediction of future morbidity and mortality," Dr. Kramer and her associates said.
The findings also show that duration of follow-up "is a critical element in evaluating low-risk populations for future events, the researchers noted.
The investigators proposed that in patients erroneously deemed to have "benign obesity" or "metabolically healthy obesity," excess weight is associated with subclinical metabolic and vascular dysfunction that ultimately leads to increased mortality and CV events over time.
Dr. Kramer had no relevant conflicts of interest. One of her coauthors reported receiving grants and fees from Merck and Novo Nordisk.
"Metabolically healthy" overweight and obese patients may carry the same short-term risk as normal-weight patients for all-cause mortality and cardiovascular events, but their weight puts them at increased risk over the long term, according to a report published online Dec. 2 in Annals of Internal Medicine.
Several studies have reported finding a subgroup of obese individuals who have normal metabolic features despite their high adiposity, and the researchers have called this condition "benign obesity" or "metabolically healthy obesity." Such patients reportedly have the same or even lower all-cause mortality and CV risk as normal-weight patients. But those studies tended to have short follow-up, making their conclusions somewhat questionable, said Dr. Caroline K. Kramer of the Leadership Sinai Centre for Diabetes, Toronto, and her associates.
The findings from their meta-analysis of eight observational studies indicate that excess weight confers increased risk after 10 years, even in "metabolically healthy" obese patients. "Our results do not support this concept of ‘benign obesity’ and demonstrate that there is no ‘healthy’ pattern of obesity," the researchers wrote (Ann. Intern. Med. 2013 Dec. 2;159 [doi=10.7326/0003-4819-159-11-201312030-00002]).
Dr. Kramer and her colleagues pooled the results of eight studies that examined participants’ metabolic status as well as weight, which had follow-up intervals ranging from 3 years to 30 years. That yielded a sample of 61,386 patients, with 3,988 deaths or CV events.
Metabolic status was categorized as either healthy or unhealthy, depending on factors such as lipid profile, glucose tolerance, blood pressure, insulin resistance, and waist circumference.
In an initial analysis, overweight and obese patients who were metabolically healthy had similar outcomes as metabolically healthy normal-weight patients. But when the analysis was confined to studies with at least 10 years of follow-up, the metabolically healthy obese group had increased mortality and CV risk, with a risk ratio of 1.24.
In addition, all patients who were metabolically unhealthy were at increased risk for death and CV events, regardless of their weight. In fact, normal-weight patients who were metabolically unhealthy carried the same risk of death or CV events as did overweight and obese patients who were metabolically unhealthy. That indicates that normal-weight patients who are metabolically unhealthy are a patient population that merits particular attention in clinical practice, the investigators said.
"Our findings highlight the need for comprehensive evaluation of not only BMI [body mass index] but also metabolic factors for the prediction of future morbidity and mortality," Dr. Kramer and her associates said.
The findings also show that duration of follow-up "is a critical element in evaluating low-risk populations for future events, the researchers noted.
The investigators proposed that in patients erroneously deemed to have "benign obesity" or "metabolically healthy obesity," excess weight is associated with subclinical metabolic and vascular dysfunction that ultimately leads to increased mortality and CV events over time.
Dr. Kramer had no relevant conflicts of interest. One of her coauthors reported receiving grants and fees from Merck and Novo Nordisk.
FROM ANNALS OF INTERNAL MEDICINE
Major finding: Overweight and obese patients who were metabolically healthy had similar mortality and cardiovascular outcomes as metabolically healthy normal-weight patients in an initial analysis; but when the analysis was confined to studies with at least 10 years of follow-up, the metabolically healthy obese group had increased mortality and CV risk, with a risk ratio of 1.24.
Data source: A systematic review of the literature and meta-analysis of eight large studies (61,386 participants) examining the effects of weight and metabolic status on mortality and CV outcomes.
Disclosures: Dr. Kramer had no relevant conflicts of interest. One of her coauthors reported receiving grants and fees from Merck and Novo Nordisk.
Abrupt increase noted in LVAD thrombosis
An abrupt and unexpected increase in pump thrombosis has occurred since March 2011 in patients with advanced heart failure who received the HeartMate II left ventricular assist device, according to a report published online Nov. 27 in the New England Journal of Medicine.
Those who develop LVAD thrombosis are at substantial risk of death and morbidity unless the device is replaced or cardiac transplantation is performed, said Dr. Randall C. Starling of the Cleveland Clinic and his associates.
The reason for the sudden rise in pump thrombosis is not yet known, but it may be related to excess "deposition of material (fibrin and denatured protein) in proximity to the inflow bearing" of the device, which has been observed by clinicians in at least three cardiac centers.
"Our data suggest that the HeartMate II may have a more narrow tolerance with respect to thrombus formation than originally understood, and may be vulnerable to the timing and intensity of anticoagulation," Dr. Starling and his colleagues noted.
The HeartMate II, made by Thoratec, is a small axial-flow LVAD that "rapidly became integral in the treatment of patients with advanced heart failure" after it was introduced. Early trials and postmarketing approval studies reported a 2%-4% incidence of thrombosis. But a quality review at the Cleveland Clinic showed a sudden, marked increase in that rate beginning in March 2011.
Researchers at the Cleveland Clinic then pooled their data on the HeartMate II (from October 2004 to February 2013) with data on the device from two other high-volume cardiology centers: Washington University Barnes-Jewish Hospital, St. Louis (January 2004 to April 2013) and Duke University Medical Center, Durham, N.C. (May 2005 to May 2013). They now report on their findings from 895 HeartMate II LVADs implanted in 837 patients during that time.
"The HeartMate II may have a more narrow tolerance with respect to thrombus formation than originally understood, and may be vulnerable to the timing and intensity of anticoagulation..."
The mean patient age was 55 years, and 21% of patients were women.
The primary endpoint of the study was confirmed LVAD thrombosis, defined as a finding at pump replacement, urgent transplantation, or autopsy of a thrombus on the blood-contacting surface of the device, its inflow cannula, or its outflow conduit. A total of 72 such thromboses were found in 66 patients; in addition, 36 cases of suspected thromboses occurred.
The rate of confirmed pump thrombosis was initially stable at roughly 2.2% for several years, then rose steeply after March 2011 to 8.4%. This pattern was seen at all three medical centers and across all the cardiac surgeons at those centers.
The median time interval from implantation to pump thrombosis had been 18.6 months before March 2011, but that rate plummeted to 2.7 months during and after March 2011, Dr. Starling and his associates reported (N. Engl. J. Med. 2013 Nov. 27 [doi: 10.1056/NEJMoa1313385]).
The investigators also reviewed serial measurements of lactate dehydrogenase (LDH) that had been taken in recipients of 568 of the devices. Elevated LDH indicates hemolysis that may herald impending thrombosis.
"The graph of the occurrence of LDH levels above 1000 IU per liter ... was nearly superimposable on the graph of the occurrence of confirmed pump thrombosis. The LDH data showed a parallel tendency for elevations to occur early after implantation beginning in approximately March 2011," they said.
The LDH level typically rose from an average of 540 IU/L to an average of 1,490 IU/L during the weeks leading up to LVAD thrombosis at all three medical centers. This confirms that LDH level is a useful clinical biomarker of hemolysis associated with LVAD thrombosis, the researchers said.
Of the 72 pump thromboses, 11 were managed by heart transplantation and 21 by pump replacement. Of the remaining 40 thromboses, 2 were managed by LVAD removal because left ventricular function had improved, and 38 were managed medically, with augmentation of anticoagulation therapy and thrombolytic agents. This stabilized the clinical course in some patients, but others elected withdrawal of care, citing "futility."
Mortality was 48% among the patients who could not be managed by heart transplantation or pump replacement, and this elevated rate was consistent across the three medical centers.
The reasons for this abrupt, dramatic rise in thromboses associated with the HeartMate II are not known. There were no changes in LVAD implantation techniques at any of the three medical centers during the course of this study, "and we could find no association between pump thrombosis and the surgeon performing the implantation." The design of the HeartMate II was changed during this interval – "modification of the outflow graft and bend relief, inflow conduit, and software" was introduced – but these changes have not been linked to pump thrombosis as yet.
Dr. Starling and his associates did note the deposition of fibrin and denatured protein around the inflow bearing, which could narrow the inflow pathway, "increasing shear stress on the red cells and, if the deposition is large enough, decreasing the ability of the pump to unload the left ventricle." But it remains to be seen whether this or other factors played a role.
"Further investigation of predisposing patient and device factors and preventive and therapeutic strategies are urgently needed to resolve this important safety issue," the investigators said.
They added that a fourth cardiology center, the University of Pennsylvania, Philadelphia, found a similar pattern in a preliminary analysis that was reported as the article went to press. In that report on 150 HeartMate II LVADs implanted in 148 patients between November 2005 and September 2013, there were 15 thrombotic events. "Similar to the findings in our study, the event rate has increased abruptly and unexpectedly, with a rate of confirmed thrombosis that continues to rise," Dr. Starling and his associates said.
Dr. Starling reported ties to Thoratec and HeartWare, and his associates reported ties to Thoratec, HeartWare, Abiomed, and Syncardia.
An abrupt and unexpected increase in pump thrombosis has occurred since March 2011 in patients with advanced heart failure who received the HeartMate II left ventricular assist device, according to a report published online Nov. 27 in the New England Journal of Medicine.
Those who develop LVAD thrombosis are at substantial risk of death and morbidity unless the device is replaced or cardiac transplantation is performed, said Dr. Randall C. Starling of the Cleveland Clinic and his associates.
The reason for the sudden rise in pump thrombosis is not yet known, but it may be related to excess "deposition of material (fibrin and denatured protein) in proximity to the inflow bearing" of the device, which has been observed by clinicians in at least three cardiac centers.
"Our data suggest that the HeartMate II may have a more narrow tolerance with respect to thrombus formation than originally understood, and may be vulnerable to the timing and intensity of anticoagulation," Dr. Starling and his colleagues noted.
The HeartMate II, made by Thoratec, is a small axial-flow LVAD that "rapidly became integral in the treatment of patients with advanced heart failure" after it was introduced. Early trials and postmarketing approval studies reported a 2%-4% incidence of thrombosis. But a quality review at the Cleveland Clinic showed a sudden, marked increase in that rate beginning in March 2011.
Researchers at the Cleveland Clinic then pooled their data on the HeartMate II (from October 2004 to February 2013) with data on the device from two other high-volume cardiology centers: Washington University Barnes-Jewish Hospital, St. Louis (January 2004 to April 2013) and Duke University Medical Center, Durham, N.C. (May 2005 to May 2013). They now report on their findings from 895 HeartMate II LVADs implanted in 837 patients during that time.
"The HeartMate II may have a more narrow tolerance with respect to thrombus formation than originally understood, and may be vulnerable to the timing and intensity of anticoagulation..."
The mean patient age was 55 years, and 21% of patients were women.
The primary endpoint of the study was confirmed LVAD thrombosis, defined as a finding at pump replacement, urgent transplantation, or autopsy of a thrombus on the blood-contacting surface of the device, its inflow cannula, or its outflow conduit. A total of 72 such thromboses were found in 66 patients; in addition, 36 cases of suspected thromboses occurred.
The rate of confirmed pump thrombosis was initially stable at roughly 2.2% for several years, then rose steeply after March 2011 to 8.4%. This pattern was seen at all three medical centers and across all the cardiac surgeons at those centers.
The median time interval from implantation to pump thrombosis had been 18.6 months before March 2011, but that rate plummeted to 2.7 months during and after March 2011, Dr. Starling and his associates reported (N. Engl. J. Med. 2013 Nov. 27 [doi: 10.1056/NEJMoa1313385]).
The investigators also reviewed serial measurements of lactate dehydrogenase (LDH) that had been taken in recipients of 568 of the devices. Elevated LDH indicates hemolysis that may herald impending thrombosis.
"The graph of the occurrence of LDH levels above 1000 IU per liter ... was nearly superimposable on the graph of the occurrence of confirmed pump thrombosis. The LDH data showed a parallel tendency for elevations to occur early after implantation beginning in approximately March 2011," they said.
The LDH level typically rose from an average of 540 IU/L to an average of 1,490 IU/L during the weeks leading up to LVAD thrombosis at all three medical centers. This confirms that LDH level is a useful clinical biomarker of hemolysis associated with LVAD thrombosis, the researchers said.
Of the 72 pump thromboses, 11 were managed by heart transplantation and 21 by pump replacement. Of the remaining 40 thromboses, 2 were managed by LVAD removal because left ventricular function had improved, and 38 were managed medically, with augmentation of anticoagulation therapy and thrombolytic agents. This stabilized the clinical course in some patients, but others elected withdrawal of care, citing "futility."
Mortality was 48% among the patients who could not be managed by heart transplantation or pump replacement, and this elevated rate was consistent across the three medical centers.
The reasons for this abrupt, dramatic rise in thromboses associated with the HeartMate II are not known. There were no changes in LVAD implantation techniques at any of the three medical centers during the course of this study, "and we could find no association between pump thrombosis and the surgeon performing the implantation." The design of the HeartMate II was changed during this interval – "modification of the outflow graft and bend relief, inflow conduit, and software" was introduced – but these changes have not been linked to pump thrombosis as yet.
Dr. Starling and his associates did note the deposition of fibrin and denatured protein around the inflow bearing, which could narrow the inflow pathway, "increasing shear stress on the red cells and, if the deposition is large enough, decreasing the ability of the pump to unload the left ventricle." But it remains to be seen whether this or other factors played a role.
"Further investigation of predisposing patient and device factors and preventive and therapeutic strategies are urgently needed to resolve this important safety issue," the investigators said.
They added that a fourth cardiology center, the University of Pennsylvania, Philadelphia, found a similar pattern in a preliminary analysis that was reported as the article went to press. In that report on 150 HeartMate II LVADs implanted in 148 patients between November 2005 and September 2013, there were 15 thrombotic events. "Similar to the findings in our study, the event rate has increased abruptly and unexpectedly, with a rate of confirmed thrombosis that continues to rise," Dr. Starling and his associates said.
Dr. Starling reported ties to Thoratec and HeartWare, and his associates reported ties to Thoratec, HeartWare, Abiomed, and Syncardia.
An abrupt and unexpected increase in pump thrombosis has occurred since March 2011 in patients with advanced heart failure who received the HeartMate II left ventricular assist device, according to a report published online Nov. 27 in the New England Journal of Medicine.
Those who develop LVAD thrombosis are at substantial risk of death and morbidity unless the device is replaced or cardiac transplantation is performed, said Dr. Randall C. Starling of the Cleveland Clinic and his associates.
The reason for the sudden rise in pump thrombosis is not yet known, but it may be related to excess "deposition of material (fibrin and denatured protein) in proximity to the inflow bearing" of the device, which has been observed by clinicians in at least three cardiac centers.
"Our data suggest that the HeartMate II may have a more narrow tolerance with respect to thrombus formation than originally understood, and may be vulnerable to the timing and intensity of anticoagulation," Dr. Starling and his colleagues noted.
The HeartMate II, made by Thoratec, is a small axial-flow LVAD that "rapidly became integral in the treatment of patients with advanced heart failure" after it was introduced. Early trials and postmarketing approval studies reported a 2%-4% incidence of thrombosis. But a quality review at the Cleveland Clinic showed a sudden, marked increase in that rate beginning in March 2011.
Researchers at the Cleveland Clinic then pooled their data on the HeartMate II (from October 2004 to February 2013) with data on the device from two other high-volume cardiology centers: Washington University Barnes-Jewish Hospital, St. Louis (January 2004 to April 2013) and Duke University Medical Center, Durham, N.C. (May 2005 to May 2013). They now report on their findings from 895 HeartMate II LVADs implanted in 837 patients during that time.
"The HeartMate II may have a more narrow tolerance with respect to thrombus formation than originally understood, and may be vulnerable to the timing and intensity of anticoagulation..."
The mean patient age was 55 years, and 21% of patients were women.
The primary endpoint of the study was confirmed LVAD thrombosis, defined as a finding at pump replacement, urgent transplantation, or autopsy of a thrombus on the blood-contacting surface of the device, its inflow cannula, or its outflow conduit. A total of 72 such thromboses were found in 66 patients; in addition, 36 cases of suspected thromboses occurred.
The rate of confirmed pump thrombosis was initially stable at roughly 2.2% for several years, then rose steeply after March 2011 to 8.4%. This pattern was seen at all three medical centers and across all the cardiac surgeons at those centers.
The median time interval from implantation to pump thrombosis had been 18.6 months before March 2011, but that rate plummeted to 2.7 months during and after March 2011, Dr. Starling and his associates reported (N. Engl. J. Med. 2013 Nov. 27 [doi: 10.1056/NEJMoa1313385]).
The investigators also reviewed serial measurements of lactate dehydrogenase (LDH) that had been taken in recipients of 568 of the devices. Elevated LDH indicates hemolysis that may herald impending thrombosis.
"The graph of the occurrence of LDH levels above 1000 IU per liter ... was nearly superimposable on the graph of the occurrence of confirmed pump thrombosis. The LDH data showed a parallel tendency for elevations to occur early after implantation beginning in approximately March 2011," they said.
The LDH level typically rose from an average of 540 IU/L to an average of 1,490 IU/L during the weeks leading up to LVAD thrombosis at all three medical centers. This confirms that LDH level is a useful clinical biomarker of hemolysis associated with LVAD thrombosis, the researchers said.
Of the 72 pump thromboses, 11 were managed by heart transplantation and 21 by pump replacement. Of the remaining 40 thromboses, 2 were managed by LVAD removal because left ventricular function had improved, and 38 were managed medically, with augmentation of anticoagulation therapy and thrombolytic agents. This stabilized the clinical course in some patients, but others elected withdrawal of care, citing "futility."
Mortality was 48% among the patients who could not be managed by heart transplantation or pump replacement, and this elevated rate was consistent across the three medical centers.
The reasons for this abrupt, dramatic rise in thromboses associated with the HeartMate II are not known. There were no changes in LVAD implantation techniques at any of the three medical centers during the course of this study, "and we could find no association between pump thrombosis and the surgeon performing the implantation." The design of the HeartMate II was changed during this interval – "modification of the outflow graft and bend relief, inflow conduit, and software" was introduced – but these changes have not been linked to pump thrombosis as yet.
Dr. Starling and his associates did note the deposition of fibrin and denatured protein around the inflow bearing, which could narrow the inflow pathway, "increasing shear stress on the red cells and, if the deposition is large enough, decreasing the ability of the pump to unload the left ventricle." But it remains to be seen whether this or other factors played a role.
"Further investigation of predisposing patient and device factors and preventive and therapeutic strategies are urgently needed to resolve this important safety issue," the investigators said.
They added that a fourth cardiology center, the University of Pennsylvania, Philadelphia, found a similar pattern in a preliminary analysis that was reported as the article went to press. In that report on 150 HeartMate II LVADs implanted in 148 patients between November 2005 and September 2013, there were 15 thrombotic events. "Similar to the findings in our study, the event rate has increased abruptly and unexpectedly, with a rate of confirmed thrombosis that continues to rise," Dr. Starling and his associates said.
Dr. Starling reported ties to Thoratec and HeartWare, and his associates reported ties to Thoratec, HeartWare, Abiomed, and Syncardia.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: The rate of pump thrombosis was stable at approximately 2.2% for several years, then rose steeply after March 2011 to 8.4%; the median interval from implantation to pump thrombosis, which had been stable at 18.6 months for several years, plummeted to 2.7 months after March 2011.
Data source: A retrospective study involving 837 recipients of 895 HeartMate II LVADs at three U.S. medical centers during 2004-2013, in which 72 LVAD thromboses were confirmed in 66 patients and an additional 36 thromboses were suspected.
Disclosures: Dr. Starling reported ties to Thoratec and HeartWare, and his associates reported ties to Thoratec, HeartWare, Abiomed, and Syncardia.
Delay in diagnosing eosinophilic esophagitis raises stricture risk
The longer the interval between symptom onset and the diagnosis of eosinophilic esophagitis, the greater the chance that the patient will have developed esophageal strictures, according to a report in the December issue of Gastroenterology (doi:10.1053/j.gastro.2013.08.015).
In a retrospective study of 523 cases of eosinophilic esophagitis (EoE) recorded in a national Swiss database, patients "were likely to present with purely inflammatory endoscopic EoE features early in the disease course and then progress to develop fibrotic endoscopic features, in addition to inflammatory features," said Dr. Alain M. Schoepfer of the division of gastroenerology and hepatology, Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland, and his associates.
Moreover, an additional analysis of numerous potential disease-, environmental-, and patient-related risk factors demonstrated that the length of diagnostic delay is the single strongest risk factor for stricture formation, the investigators noted.
The natural history of untreated EoE has not been investigated extensively, and data regarding stricture formation are particularly lacking. In addition, eosinophilic esophagitis has long been associated with a substantial delay in diagnosis, with a median of 5 years elapsing between symptom onset and correct identification of the disorder.
Dr. Schoepfer and his colleagues examined the relationship between the duration of untreated disease (before EoE was diagnosed) and the prevalence of esophageal stricture using a database with detailed medical records of 323 patients they personally diagnosed and treated plus 200 others who were diagnosed and treated by other gastroenterologists throughout Switzerland. The database included the results from numerous biopsies of the proximal and distal esophagus for every patient, as well as information on 98 clinical factors that might influence stricture formation.
The median diagnostic delay was 6 years (range, 0 to more than 20 years).
Strictures were present at diagnosis in 75 patients (37.5%).
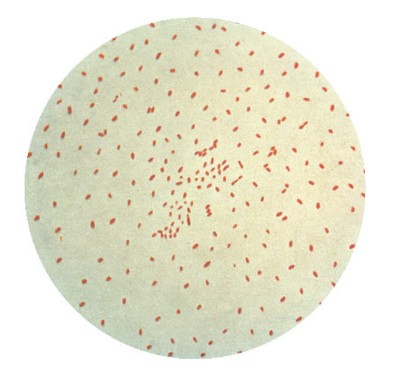
Features of active inflammation, such as edema, furrows, and whitish exudates, were present in 79.5% of patients while features of fibrotic activity, such as strictures, rings, and crepe-paper esophagus, were seen in 63.0%.
The prevalence of fibrotic features including strictures increased with increasing duration of diagnostic delay. This prevalence was 46.5% among patients who were diagnosed as having EoE within 0-2 years of symptom onset, rising to 87.5% among those diagnosed 20 years or more after symptom onset.
In contrast, the prevalence of inflammatory features alone decreased with increasing duration of diagnostic delay.
The prevalence of esophageal strictures likewise correlated with diagnostic delay. It was 17.2% among patients diagnosed within 0-2 years of symptom onset, compared with 70.8% among those diagnosed 20 years or more after symptom onset, the investigators said.
The prevalence of strictures did not differ by patient age at diagnosis. It was comparable between patients who were diagnosed before they reached 20 years of age and those diagnosed after age 20. The prevalence of strictures also did not differ between the 323 patients diagnosed and treated by Dr. Schoepfer and his associates, who were managed according to a strict standardized protocol, and the 200 other patients who were managed by numerous other gastroenterologists according to their own individual practice preferences.
In an analysis of nearly 100 potential risk factors for stricture formation, only the length of diagnostic delay was found to be significantly associated with the presence of strictures at diagnosis.
These findings demonstrate that a patient’s disease course "is a continuum – a march from a disease predominantly inflammatory in nature to a disease with endoscopic fibrotic features, including strictures, in addition to existing inflammation," the researchers said.
Clinicians should make every effort to reduce the delay in diagnosis of EoE, they added.
This study was supported by the Swiss National Science Foundation. No relevant financial conflicts of interest were reported.
The longer the interval between symptom onset and the diagnosis of eosinophilic esophagitis, the greater the chance that the patient will have developed esophageal strictures, according to a report in the December issue of Gastroenterology (doi:10.1053/j.gastro.2013.08.015).
In a retrospective study of 523 cases of eosinophilic esophagitis (EoE) recorded in a national Swiss database, patients "were likely to present with purely inflammatory endoscopic EoE features early in the disease course and then progress to develop fibrotic endoscopic features, in addition to inflammatory features," said Dr. Alain M. Schoepfer of the division of gastroenerology and hepatology, Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland, and his associates.
Moreover, an additional analysis of numerous potential disease-, environmental-, and patient-related risk factors demonstrated that the length of diagnostic delay is the single strongest risk factor for stricture formation, the investigators noted.
The natural history of untreated EoE has not been investigated extensively, and data regarding stricture formation are particularly lacking. In addition, eosinophilic esophagitis has long been associated with a substantial delay in diagnosis, with a median of 5 years elapsing between symptom onset and correct identification of the disorder.
Dr. Schoepfer and his colleagues examined the relationship between the duration of untreated disease (before EoE was diagnosed) and the prevalence of esophageal stricture using a database with detailed medical records of 323 patients they personally diagnosed and treated plus 200 others who were diagnosed and treated by other gastroenterologists throughout Switzerland. The database included the results from numerous biopsies of the proximal and distal esophagus for every patient, as well as information on 98 clinical factors that might influence stricture formation.
The median diagnostic delay was 6 years (range, 0 to more than 20 years).
Strictures were present at diagnosis in 75 patients (37.5%).
Features of active inflammation, such as edema, furrows, and whitish exudates, were present in 79.5% of patients while features of fibrotic activity, such as strictures, rings, and crepe-paper esophagus, were seen in 63.0%.
The prevalence of fibrotic features including strictures increased with increasing duration of diagnostic delay. This prevalence was 46.5% among patients who were diagnosed as having EoE within 0-2 years of symptom onset, rising to 87.5% among those diagnosed 20 years or more after symptom onset.
In contrast, the prevalence of inflammatory features alone decreased with increasing duration of diagnostic delay.
The prevalence of esophageal strictures likewise correlated with diagnostic delay. It was 17.2% among patients diagnosed within 0-2 years of symptom onset, compared with 70.8% among those diagnosed 20 years or more after symptom onset, the investigators said.
The prevalence of strictures did not differ by patient age at diagnosis. It was comparable between patients who were diagnosed before they reached 20 years of age and those diagnosed after age 20. The prevalence of strictures also did not differ between the 323 patients diagnosed and treated by Dr. Schoepfer and his associates, who were managed according to a strict standardized protocol, and the 200 other patients who were managed by numerous other gastroenterologists according to their own individual practice preferences.
In an analysis of nearly 100 potential risk factors for stricture formation, only the length of diagnostic delay was found to be significantly associated with the presence of strictures at diagnosis.
These findings demonstrate that a patient’s disease course "is a continuum – a march from a disease predominantly inflammatory in nature to a disease with endoscopic fibrotic features, including strictures, in addition to existing inflammation," the researchers said.
Clinicians should make every effort to reduce the delay in diagnosis of EoE, they added.
This study was supported by the Swiss National Science Foundation. No relevant financial conflicts of interest were reported.
The longer the interval between symptom onset and the diagnosis of eosinophilic esophagitis, the greater the chance that the patient will have developed esophageal strictures, according to a report in the December issue of Gastroenterology (doi:10.1053/j.gastro.2013.08.015).
In a retrospective study of 523 cases of eosinophilic esophagitis (EoE) recorded in a national Swiss database, patients "were likely to present with purely inflammatory endoscopic EoE features early in the disease course and then progress to develop fibrotic endoscopic features, in addition to inflammatory features," said Dr. Alain M. Schoepfer of the division of gastroenerology and hepatology, Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland, and his associates.
Moreover, an additional analysis of numerous potential disease-, environmental-, and patient-related risk factors demonstrated that the length of diagnostic delay is the single strongest risk factor for stricture formation, the investigators noted.
The natural history of untreated EoE has not been investigated extensively, and data regarding stricture formation are particularly lacking. In addition, eosinophilic esophagitis has long been associated with a substantial delay in diagnosis, with a median of 5 years elapsing between symptom onset and correct identification of the disorder.
Dr. Schoepfer and his colleagues examined the relationship between the duration of untreated disease (before EoE was diagnosed) and the prevalence of esophageal stricture using a database with detailed medical records of 323 patients they personally diagnosed and treated plus 200 others who were diagnosed and treated by other gastroenterologists throughout Switzerland. The database included the results from numerous biopsies of the proximal and distal esophagus for every patient, as well as information on 98 clinical factors that might influence stricture formation.
The median diagnostic delay was 6 years (range, 0 to more than 20 years).
Strictures were present at diagnosis in 75 patients (37.5%).
Features of active inflammation, such as edema, furrows, and whitish exudates, were present in 79.5% of patients while features of fibrotic activity, such as strictures, rings, and crepe-paper esophagus, were seen in 63.0%.
The prevalence of fibrotic features including strictures increased with increasing duration of diagnostic delay. This prevalence was 46.5% among patients who were diagnosed as having EoE within 0-2 years of symptom onset, rising to 87.5% among those diagnosed 20 years or more after symptom onset.
In contrast, the prevalence of inflammatory features alone decreased with increasing duration of diagnostic delay.
The prevalence of esophageal strictures likewise correlated with diagnostic delay. It was 17.2% among patients diagnosed within 0-2 years of symptom onset, compared with 70.8% among those diagnosed 20 years or more after symptom onset, the investigators said.
The prevalence of strictures did not differ by patient age at diagnosis. It was comparable between patients who were diagnosed before they reached 20 years of age and those diagnosed after age 20. The prevalence of strictures also did not differ between the 323 patients diagnosed and treated by Dr. Schoepfer and his associates, who were managed according to a strict standardized protocol, and the 200 other patients who were managed by numerous other gastroenterologists according to their own individual practice preferences.
In an analysis of nearly 100 potential risk factors for stricture formation, only the length of diagnostic delay was found to be significantly associated with the presence of strictures at diagnosis.
These findings demonstrate that a patient’s disease course "is a continuum – a march from a disease predominantly inflammatory in nature to a disease with endoscopic fibrotic features, including strictures, in addition to existing inflammation," the researchers said.
Clinicians should make every effort to reduce the delay in diagnosis of EoE, they added.
This study was supported by the Swiss National Science Foundation. No relevant financial conflicts of interest were reported.
FROM GASTROENTEROLOGY
Major finding: The prevalence of esophageal strictures rose as diagnostic delay increased: 17.2% among patients diagnosed within 0-2 years of symptom onset, compared with 70.8% among those diagnosed 20 years or more after symptom onset.
Data source: A retrospective cohort study involving 523 cases of EoE, including 75 patients (37.5%) who had esophageal strictures at diagnosis, in a national Swiss database; the median delay in diagnosis was 6 years.
Disclosures: This study was supported by the Swiss National Science Foundation. No relevant financial conflicts of interest were reported.
New definition of kidney injury is more predictive of mortality
The newly proposed consensus definition of acute kidney injury in patients with cirrhosis accurately predicts 30-day mortality and other adverse outcomes in this patient population much better than the current, more rigid definition would, according to a report in the December issue of Gastroenterology (doi:10.1053/j.gastro.2013.08.051).
In what they described as the largest prospective study of this topic to date, researchers found that the recently proposed, broader redefinition of acute kidney injury (AKI) correctly identified which patients were likely to die, develop severe complications such as organ failure, or require longer hospitalization, even when the AKI was transient and resolved completely after treatment.
Courtesy American Gastroenterological Association
More than half of the patients in this study who had episodes of AKI according to the new definition did not meet the criteria of the old definition. So using the new definition will help identify these high-risk patients at an earlier stage of renal dysfunction, "well before the stringent diagnostic criteria of [the old definition] are reached," when they will have a better treatment response, said Dr. Florence Wong of the division of gastroenterology, University of Toronto, and her associates.
The old definition of AKI required the presence of hepatorenal syndrome, with a serum creatinine level of greater than 2.5 mg/dL. This meant that patients with less severe renal dysfunction didn’t qualify and weren’t treated. But emerging evidence indicates that even mild degrees of renal dysfunction signal a poor prognosis, and that serum creatinine alone doesn’t accurately reflect renal dysfunction in advanced cirrhosis.
So the International Ascites Club and the Acute Dialysis Quality Initiative (ADQI) group proposed that acute kidney injury in cirrhosis should be redefined as an increase in serum creatinine level of 0.3 mg/dL or greater within 48 hours, or a 50% increase in serum creatinine level from a stable baseline reading within the previous 6 months, regardless of final serum creatinine level.
Dr. Wong and her colleagues assessed the new definition in a cohort of 337 cirrhotic patients treated during a 2-year period at 12 North American medical centers who were admitted with a bacterial infection (287 subjects) or who developed a bacterial infection during hospitalization (50 subjects). The most common infections were urinary tract infection (27% of patients), spontaneous bacterial peritonitis (21%), skin infection (14%), pneumonia (10%), and spontaneous bacteremia with no clear source of infection (9%).
Approximately half of these patients (49%) developed at least one episode of AKI during hospitalization. The 30-day mortality was significantly higher for those who developed AKI according to the new definition (34% mortality) than in those who did not (7% mortality), the investigators said.
Most patients who developed AKI had only a transient case, and their renal function completely recovered. Yet their subsequent mortality within 30 days was twice as high as that for patients who didn’t have any AKI.
The negative predictive value of the new definition of AKI was 93%, and the positive predictive value was 34%.
This study was supported in part by the National Institutes of Health, the National Institute of Diabetes and Digestive and Kidney Diseases, and the National Center for Research Resources. No financial conflicts of interest were reported.
Renal dysfunction in patients with cirrhosis is often associated with sepsis. This combination involves a very high probability of death. Recently, the concept of acute kidney injury has been proposed to be extended also to renal failure occurring in patients with cirrhosis. AKI should overcome limitations associated with a fixed creatinine threshold, ensure rapid identification of renal dysfunction, and allow timely treatment in patients with hepatorenal syndrome. However, AKI should also overcome the skepticism of those who wish not to abandon previous definitions.
The recent paper of Dr. Wong and her colleagues explored the impact of AKI in 337 hospitalized patients with cirrhosis. Two-hundred eighty-seven patients had bacterial infection at admission, and 93 developed it during hospitalization. Overall, 68 patients died from multiorgan failure, whereas only 7% of patients without AKI died. Mortality ranged from 15% in patients who recovered from AKI to 80% in those who did not. Moreover, 76 patients (23%) developed a second infection, often associated with invasive procedures! An elevated Model for End-Stage Liver Disease score and a second infection were factors independently associated with AKI. Accordingly, the development of AKI in cirrhosis, even if reversible, was shown to be a strong predictor of short survival.
These findings show that, in cirrhosis, even small creatinine changes (0.3 mg) are clinically relevant, and that AKI is probably a hallmark of hemodynamic instability with a risk of multiorgan failure and death. The altered hemodynamics in patients with cirrhosis cause central hypovolemia. Aiming at protecting our patients from infection and AKI, we should also pay more attention to clinical procedures that raise serum creatinine level.
Dr. Francesco Salerno is in the department of internal medicine, at the Policlinico IRCCS San Donato, University of Milan (Italy); Dr. Vincenzo La Mura is with the Fondazione IRCCS Ca'Granda, in the department of gastroenterologia-1 of the Hospital Maggiore Policlinico, Milan. They reported no relevant financial conflicts.
Renal dysfunction in patients with cirrhosis is often associated with sepsis. This combination involves a very high probability of death. Recently, the concept of acute kidney injury has been proposed to be extended also to renal failure occurring in patients with cirrhosis. AKI should overcome limitations associated with a fixed creatinine threshold, ensure rapid identification of renal dysfunction, and allow timely treatment in patients with hepatorenal syndrome. However, AKI should also overcome the skepticism of those who wish not to abandon previous definitions.
The recent paper of Dr. Wong and her colleagues explored the impact of AKI in 337 hospitalized patients with cirrhosis. Two-hundred eighty-seven patients had bacterial infection at admission, and 93 developed it during hospitalization. Overall, 68 patients died from multiorgan failure, whereas only 7% of patients without AKI died. Mortality ranged from 15% in patients who recovered from AKI to 80% in those who did not. Moreover, 76 patients (23%) developed a second infection, often associated with invasive procedures! An elevated Model for End-Stage Liver Disease score and a second infection were factors independently associated with AKI. Accordingly, the development of AKI in cirrhosis, even if reversible, was shown to be a strong predictor of short survival.
These findings show that, in cirrhosis, even small creatinine changes (0.3 mg) are clinically relevant, and that AKI is probably a hallmark of hemodynamic instability with a risk of multiorgan failure and death. The altered hemodynamics in patients with cirrhosis cause central hypovolemia. Aiming at protecting our patients from infection and AKI, we should also pay more attention to clinical procedures that raise serum creatinine level.
Dr. Francesco Salerno is in the department of internal medicine, at the Policlinico IRCCS San Donato, University of Milan (Italy); Dr. Vincenzo La Mura is with the Fondazione IRCCS Ca'Granda, in the department of gastroenterologia-1 of the Hospital Maggiore Policlinico, Milan. They reported no relevant financial conflicts.
Renal dysfunction in patients with cirrhosis is often associated with sepsis. This combination involves a very high probability of death. Recently, the concept of acute kidney injury has been proposed to be extended also to renal failure occurring in patients with cirrhosis. AKI should overcome limitations associated with a fixed creatinine threshold, ensure rapid identification of renal dysfunction, and allow timely treatment in patients with hepatorenal syndrome. However, AKI should also overcome the skepticism of those who wish not to abandon previous definitions.
The recent paper of Dr. Wong and her colleagues explored the impact of AKI in 337 hospitalized patients with cirrhosis. Two-hundred eighty-seven patients had bacterial infection at admission, and 93 developed it during hospitalization. Overall, 68 patients died from multiorgan failure, whereas only 7% of patients without AKI died. Mortality ranged from 15% in patients who recovered from AKI to 80% in those who did not. Moreover, 76 patients (23%) developed a second infection, often associated with invasive procedures! An elevated Model for End-Stage Liver Disease score and a second infection were factors independently associated with AKI. Accordingly, the development of AKI in cirrhosis, even if reversible, was shown to be a strong predictor of short survival.
These findings show that, in cirrhosis, even small creatinine changes (0.3 mg) are clinically relevant, and that AKI is probably a hallmark of hemodynamic instability with a risk of multiorgan failure and death. The altered hemodynamics in patients with cirrhosis cause central hypovolemia. Aiming at protecting our patients from infection and AKI, we should also pay more attention to clinical procedures that raise serum creatinine level.
Dr. Francesco Salerno is in the department of internal medicine, at the Policlinico IRCCS San Donato, University of Milan (Italy); Dr. Vincenzo La Mura is with the Fondazione IRCCS Ca'Granda, in the department of gastroenterologia-1 of the Hospital Maggiore Policlinico, Milan. They reported no relevant financial conflicts.
The newly proposed consensus definition of acute kidney injury in patients with cirrhosis accurately predicts 30-day mortality and other adverse outcomes in this patient population much better than the current, more rigid definition would, according to a report in the December issue of Gastroenterology (doi:10.1053/j.gastro.2013.08.051).
In what they described as the largest prospective study of this topic to date, researchers found that the recently proposed, broader redefinition of acute kidney injury (AKI) correctly identified which patients were likely to die, develop severe complications such as organ failure, or require longer hospitalization, even when the AKI was transient and resolved completely after treatment.
Courtesy American Gastroenterological Association
More than half of the patients in this study who had episodes of AKI according to the new definition did not meet the criteria of the old definition. So using the new definition will help identify these high-risk patients at an earlier stage of renal dysfunction, "well before the stringent diagnostic criteria of [the old definition] are reached," when they will have a better treatment response, said Dr. Florence Wong of the division of gastroenterology, University of Toronto, and her associates.
The old definition of AKI required the presence of hepatorenal syndrome, with a serum creatinine level of greater than 2.5 mg/dL. This meant that patients with less severe renal dysfunction didn’t qualify and weren’t treated. But emerging evidence indicates that even mild degrees of renal dysfunction signal a poor prognosis, and that serum creatinine alone doesn’t accurately reflect renal dysfunction in advanced cirrhosis.
So the International Ascites Club and the Acute Dialysis Quality Initiative (ADQI) group proposed that acute kidney injury in cirrhosis should be redefined as an increase in serum creatinine level of 0.3 mg/dL or greater within 48 hours, or a 50% increase in serum creatinine level from a stable baseline reading within the previous 6 months, regardless of final serum creatinine level.
Dr. Wong and her colleagues assessed the new definition in a cohort of 337 cirrhotic patients treated during a 2-year period at 12 North American medical centers who were admitted with a bacterial infection (287 subjects) or who developed a bacterial infection during hospitalization (50 subjects). The most common infections were urinary tract infection (27% of patients), spontaneous bacterial peritonitis (21%), skin infection (14%), pneumonia (10%), and spontaneous bacteremia with no clear source of infection (9%).
Approximately half of these patients (49%) developed at least one episode of AKI during hospitalization. The 30-day mortality was significantly higher for those who developed AKI according to the new definition (34% mortality) than in those who did not (7% mortality), the investigators said.
Most patients who developed AKI had only a transient case, and their renal function completely recovered. Yet their subsequent mortality within 30 days was twice as high as that for patients who didn’t have any AKI.
The negative predictive value of the new definition of AKI was 93%, and the positive predictive value was 34%.
This study was supported in part by the National Institutes of Health, the National Institute of Diabetes and Digestive and Kidney Diseases, and the National Center for Research Resources. No financial conflicts of interest were reported.
The newly proposed consensus definition of acute kidney injury in patients with cirrhosis accurately predicts 30-day mortality and other adverse outcomes in this patient population much better than the current, more rigid definition would, according to a report in the December issue of Gastroenterology (doi:10.1053/j.gastro.2013.08.051).
In what they described as the largest prospective study of this topic to date, researchers found that the recently proposed, broader redefinition of acute kidney injury (AKI) correctly identified which patients were likely to die, develop severe complications such as organ failure, or require longer hospitalization, even when the AKI was transient and resolved completely after treatment.
Courtesy American Gastroenterological Association
More than half of the patients in this study who had episodes of AKI according to the new definition did not meet the criteria of the old definition. So using the new definition will help identify these high-risk patients at an earlier stage of renal dysfunction, "well before the stringent diagnostic criteria of [the old definition] are reached," when they will have a better treatment response, said Dr. Florence Wong of the division of gastroenterology, University of Toronto, and her associates.
The old definition of AKI required the presence of hepatorenal syndrome, with a serum creatinine level of greater than 2.5 mg/dL. This meant that patients with less severe renal dysfunction didn’t qualify and weren’t treated. But emerging evidence indicates that even mild degrees of renal dysfunction signal a poor prognosis, and that serum creatinine alone doesn’t accurately reflect renal dysfunction in advanced cirrhosis.
So the International Ascites Club and the Acute Dialysis Quality Initiative (ADQI) group proposed that acute kidney injury in cirrhosis should be redefined as an increase in serum creatinine level of 0.3 mg/dL or greater within 48 hours, or a 50% increase in serum creatinine level from a stable baseline reading within the previous 6 months, regardless of final serum creatinine level.
Dr. Wong and her colleagues assessed the new definition in a cohort of 337 cirrhotic patients treated during a 2-year period at 12 North American medical centers who were admitted with a bacterial infection (287 subjects) or who developed a bacterial infection during hospitalization (50 subjects). The most common infections were urinary tract infection (27% of patients), spontaneous bacterial peritonitis (21%), skin infection (14%), pneumonia (10%), and spontaneous bacteremia with no clear source of infection (9%).
Approximately half of these patients (49%) developed at least one episode of AKI during hospitalization. The 30-day mortality was significantly higher for those who developed AKI according to the new definition (34% mortality) than in those who did not (7% mortality), the investigators said.
Most patients who developed AKI had only a transient case, and their renal function completely recovered. Yet their subsequent mortality within 30 days was twice as high as that for patients who didn’t have any AKI.
The negative predictive value of the new definition of AKI was 93%, and the positive predictive value was 34%.
This study was supported in part by the National Institutes of Health, the National Institute of Diabetes and Digestive and Kidney Diseases, and the National Center for Research Resources. No financial conflicts of interest were reported.
FROM GASTROENTEROLOGY
Major finding: 30-day mortality was significantly higher for those who developed acute kidney injury according to a new definition (34% mortality) than in those who did not (7% mortality).
Data source: A cohort study of 337 inpatients at 12 North American medical centers who had cirrhosis and a bacterial infection, half of whom developed AKI.
Disclosures: This study was supported in part by the National Institutes of Health, the National Institute of Diabetes and Digestive and Kidney Diseases, and the National Center for Research Resources. No financial conflicts of interest were reported.
Afatinib superior to chemotherapy for EGFR-mutated NSCLC
The oral EGFR blocker afatinib was superior to standard chemotherapy at prolonging progression-free survival in patients with advanced non–small-cell lung cancers that harbor EGFR mutations, according to a report in the Journal of Clinical Oncology.
Patients who received afatinib in this international, industry-sponsored phase-III clinical trial (NCT00949650) also showed statistically significant and clinically meaningful improvements in lung cancer symptoms and a higher treatment response rate than did those who received a regimen of cisplatin plus pemetrexed, which is widely considered to be the optimal combination chemotherapy, said Dr. Lecia V. Sequist of Massachusetts General Hospital and Harvard Medical School, Boston, and her associates.
Afatinib should now be considered a standard therapeutic option for such patients, they noted.
Mutations in the EGFR (epidermal growth factor receptor) gene, found in a subset of lung adenocarcinomas, make the tumors uniquely susceptible to selective EGFR blockers. Previous trials have demonstrated tumor shrinkage and durable responses to treatment with the EGFR tyrosine kinase inhibitors gefitinib and erlotinib, and afatinib has also shown promise.
In what they described as the largest prospective, randomized trial of afatinib to date, Dr. Sequist and her colleagues compared first-line afatinib against combination cisplatin plus pemetrexed in 340 patients treated at 133 medical centers in Asia, Europe, North America, South America, and Australia during a 2-year period. All the patients had advanced treatment-naive disease with proven EGFR mutations.
The patients were randomly assigned to receive either daily oral afatinib (229 patients) or monthly IV chemotherapy (111 patients) until the cancer progressed, and underwent periodic CT or MRI imaging to track progression.
After a median follow-up of 16 months, the primary endpoint of progression-free survival was significantly longer with afatinib (11.1 months) than with cisplatin plus pemetrexed (6.9 months) in an analysis conducted by independent reviewers. This endpoint also was significantly longer with afatinib (11.1 months) than with cisplatin plus pemetrexed (6.7 months) in an analysis conducted by the trial investigators, Dr. Sequist and her associates said (J. Clin. Oncol. 2013;31:3327-34).
This benefit in progression-free survival was robust across most clinically relevant subgroups of patients, regardless of age, sex, race, or Eastern Cooperative Oncology Group status.
The treatment response rate also was significantly higher with afatinib (56%) than with chemotherapy (23%) in the independent analysis, as well as in the trial investigators’ analysis (69% and 44%, respectively).
In addition, a higher proportion of patients achieved disease control with afatinib (90%) than with chemotherapy (81%). And the median duration of disease control was 13.6 months for afatinib, compared with 8.1 months for chemotherapy.
Patients reported that the time to meaningful worsening of cough and dyspnea was significantly longer with afatinib than with chemotherapy, and the time to meaningful worsening of pain was longer but not to a significant degree.
At the time of data analysis, only 28% of the study population had died, and median survival had not yet been reached for either group. The data on overall survival therefore are considered preliminary. Overall survival was not significantly different between the afatinib group (16.6 months) and the chemotherapy group (14.8 months).
"Both treatments were well tolerated, and adverse effects were manageable with dose reductions and delays. Treatment-related adverse effects of grade 3 or greater occurred in 112 patients (49%) receiving afatinib and 53 patients (48%) receiving chemotherapy," the investigators said.
Treatment was discontinued because of adverse effects in 8% of patients receiving afatinib and 12% of those receiving chemotherapy.
Pharmacokinetic studies showed that dose modification of afatinib according to individual tolerability "optimized the exposure to afatinib and maintained efficacious plasma levels," they added.
This study was supported by Boehringer Ingelheim. Dr. Sequist reported ties to Boehringer Ingelheim, Clovis Oncology, Merrimack Pharmaceuticals, and Daiichi Sankyo, and her associates reported ties to numerous industry sources.
This is by far the largest study of its kind and the first to include American rather than exclusively Asian participants, but its conclusions are scarcely surprising, according to Dr. Corey J. Langer.
Eight separate clinical trials have now consistently shown "a major, statistically significant and clinically relevant improvement in response rates, quality of life, and progression-free survival that is unheralded in the history of thoracic oncology," he noted.
Now it is time to turn to determining which, if any, EGFR tyrosine kinase inhibitor is the best, and how to prevent or overcome resistance to these agents. Ultimately, enhancing cure rates is our chief objective; testing these agents properly in the adjuvant and locally advanced setting must be a priority, he wrote.
Dr. Langer is at the Abramson Cancer Center at the University of Pennsylvania, Philadelphia. He reported ties to Boehringer Ingelheim, OSI-Astellas, Bristol-Myers Squibb, and Eli Lilly. These remarks were taken from his editorial accompanying Dr. Sequist’s report (J. Clin. Oncol. 2013;31:3303-06).
This is by far the largest study of its kind and the first to include American rather than exclusively Asian participants, but its conclusions are scarcely surprising, according to Dr. Corey J. Langer.
Eight separate clinical trials have now consistently shown "a major, statistically significant and clinically relevant improvement in response rates, quality of life, and progression-free survival that is unheralded in the history of thoracic oncology," he noted.
Now it is time to turn to determining which, if any, EGFR tyrosine kinase inhibitor is the best, and how to prevent or overcome resistance to these agents. Ultimately, enhancing cure rates is our chief objective; testing these agents properly in the adjuvant and locally advanced setting must be a priority, he wrote.
Dr. Langer is at the Abramson Cancer Center at the University of Pennsylvania, Philadelphia. He reported ties to Boehringer Ingelheim, OSI-Astellas, Bristol-Myers Squibb, and Eli Lilly. These remarks were taken from his editorial accompanying Dr. Sequist’s report (J. Clin. Oncol. 2013;31:3303-06).
This is by far the largest study of its kind and the first to include American rather than exclusively Asian participants, but its conclusions are scarcely surprising, according to Dr. Corey J. Langer.
Eight separate clinical trials have now consistently shown "a major, statistically significant and clinically relevant improvement in response rates, quality of life, and progression-free survival that is unheralded in the history of thoracic oncology," he noted.
Now it is time to turn to determining which, if any, EGFR tyrosine kinase inhibitor is the best, and how to prevent or overcome resistance to these agents. Ultimately, enhancing cure rates is our chief objective; testing these agents properly in the adjuvant and locally advanced setting must be a priority, he wrote.
Dr. Langer is at the Abramson Cancer Center at the University of Pennsylvania, Philadelphia. He reported ties to Boehringer Ingelheim, OSI-Astellas, Bristol-Myers Squibb, and Eli Lilly. These remarks were taken from his editorial accompanying Dr. Sequist’s report (J. Clin. Oncol. 2013;31:3303-06).
The oral EGFR blocker afatinib was superior to standard chemotherapy at prolonging progression-free survival in patients with advanced non–small-cell lung cancers that harbor EGFR mutations, according to a report in the Journal of Clinical Oncology.
Patients who received afatinib in this international, industry-sponsored phase-III clinical trial (NCT00949650) also showed statistically significant and clinically meaningful improvements in lung cancer symptoms and a higher treatment response rate than did those who received a regimen of cisplatin plus pemetrexed, which is widely considered to be the optimal combination chemotherapy, said Dr. Lecia V. Sequist of Massachusetts General Hospital and Harvard Medical School, Boston, and her associates.
Afatinib should now be considered a standard therapeutic option for such patients, they noted.
Mutations in the EGFR (epidermal growth factor receptor) gene, found in a subset of lung adenocarcinomas, make the tumors uniquely susceptible to selective EGFR blockers. Previous trials have demonstrated tumor shrinkage and durable responses to treatment with the EGFR tyrosine kinase inhibitors gefitinib and erlotinib, and afatinib has also shown promise.
In what they described as the largest prospective, randomized trial of afatinib to date, Dr. Sequist and her colleagues compared first-line afatinib against combination cisplatin plus pemetrexed in 340 patients treated at 133 medical centers in Asia, Europe, North America, South America, and Australia during a 2-year period. All the patients had advanced treatment-naive disease with proven EGFR mutations.
The patients were randomly assigned to receive either daily oral afatinib (229 patients) or monthly IV chemotherapy (111 patients) until the cancer progressed, and underwent periodic CT or MRI imaging to track progression.
After a median follow-up of 16 months, the primary endpoint of progression-free survival was significantly longer with afatinib (11.1 months) than with cisplatin plus pemetrexed (6.9 months) in an analysis conducted by independent reviewers. This endpoint also was significantly longer with afatinib (11.1 months) than with cisplatin plus pemetrexed (6.7 months) in an analysis conducted by the trial investigators, Dr. Sequist and her associates said (J. Clin. Oncol. 2013;31:3327-34).
This benefit in progression-free survival was robust across most clinically relevant subgroups of patients, regardless of age, sex, race, or Eastern Cooperative Oncology Group status.
The treatment response rate also was significantly higher with afatinib (56%) than with chemotherapy (23%) in the independent analysis, as well as in the trial investigators’ analysis (69% and 44%, respectively).
In addition, a higher proportion of patients achieved disease control with afatinib (90%) than with chemotherapy (81%). And the median duration of disease control was 13.6 months for afatinib, compared with 8.1 months for chemotherapy.
Patients reported that the time to meaningful worsening of cough and dyspnea was significantly longer with afatinib than with chemotherapy, and the time to meaningful worsening of pain was longer but not to a significant degree.
At the time of data analysis, only 28% of the study population had died, and median survival had not yet been reached for either group. The data on overall survival therefore are considered preliminary. Overall survival was not significantly different between the afatinib group (16.6 months) and the chemotherapy group (14.8 months).
"Both treatments were well tolerated, and adverse effects were manageable with dose reductions and delays. Treatment-related adverse effects of grade 3 or greater occurred in 112 patients (49%) receiving afatinib and 53 patients (48%) receiving chemotherapy," the investigators said.
Treatment was discontinued because of adverse effects in 8% of patients receiving afatinib and 12% of those receiving chemotherapy.
Pharmacokinetic studies showed that dose modification of afatinib according to individual tolerability "optimized the exposure to afatinib and maintained efficacious plasma levels," they added.
This study was supported by Boehringer Ingelheim. Dr. Sequist reported ties to Boehringer Ingelheim, Clovis Oncology, Merrimack Pharmaceuticals, and Daiichi Sankyo, and her associates reported ties to numerous industry sources.
The oral EGFR blocker afatinib was superior to standard chemotherapy at prolonging progression-free survival in patients with advanced non–small-cell lung cancers that harbor EGFR mutations, according to a report in the Journal of Clinical Oncology.
Patients who received afatinib in this international, industry-sponsored phase-III clinical trial (NCT00949650) also showed statistically significant and clinically meaningful improvements in lung cancer symptoms and a higher treatment response rate than did those who received a regimen of cisplatin plus pemetrexed, which is widely considered to be the optimal combination chemotherapy, said Dr. Lecia V. Sequist of Massachusetts General Hospital and Harvard Medical School, Boston, and her associates.
Afatinib should now be considered a standard therapeutic option for such patients, they noted.
Mutations in the EGFR (epidermal growth factor receptor) gene, found in a subset of lung adenocarcinomas, make the tumors uniquely susceptible to selective EGFR blockers. Previous trials have demonstrated tumor shrinkage and durable responses to treatment with the EGFR tyrosine kinase inhibitors gefitinib and erlotinib, and afatinib has also shown promise.
In what they described as the largest prospective, randomized trial of afatinib to date, Dr. Sequist and her colleagues compared first-line afatinib against combination cisplatin plus pemetrexed in 340 patients treated at 133 medical centers in Asia, Europe, North America, South America, and Australia during a 2-year period. All the patients had advanced treatment-naive disease with proven EGFR mutations.
The patients were randomly assigned to receive either daily oral afatinib (229 patients) or monthly IV chemotherapy (111 patients) until the cancer progressed, and underwent periodic CT or MRI imaging to track progression.
After a median follow-up of 16 months, the primary endpoint of progression-free survival was significantly longer with afatinib (11.1 months) than with cisplatin plus pemetrexed (6.9 months) in an analysis conducted by independent reviewers. This endpoint also was significantly longer with afatinib (11.1 months) than with cisplatin plus pemetrexed (6.7 months) in an analysis conducted by the trial investigators, Dr. Sequist and her associates said (J. Clin. Oncol. 2013;31:3327-34).
This benefit in progression-free survival was robust across most clinically relevant subgroups of patients, regardless of age, sex, race, or Eastern Cooperative Oncology Group status.
The treatment response rate also was significantly higher with afatinib (56%) than with chemotherapy (23%) in the independent analysis, as well as in the trial investigators’ analysis (69% and 44%, respectively).
In addition, a higher proportion of patients achieved disease control with afatinib (90%) than with chemotherapy (81%). And the median duration of disease control was 13.6 months for afatinib, compared with 8.1 months for chemotherapy.
Patients reported that the time to meaningful worsening of cough and dyspnea was significantly longer with afatinib than with chemotherapy, and the time to meaningful worsening of pain was longer but not to a significant degree.
At the time of data analysis, only 28% of the study population had died, and median survival had not yet been reached for either group. The data on overall survival therefore are considered preliminary. Overall survival was not significantly different between the afatinib group (16.6 months) and the chemotherapy group (14.8 months).
"Both treatments were well tolerated, and adverse effects were manageable with dose reductions and delays. Treatment-related adverse effects of grade 3 or greater occurred in 112 patients (49%) receiving afatinib and 53 patients (48%) receiving chemotherapy," the investigators said.
Treatment was discontinued because of adverse effects in 8% of patients receiving afatinib and 12% of those receiving chemotherapy.
Pharmacokinetic studies showed that dose modification of afatinib according to individual tolerability "optimized the exposure to afatinib and maintained efficacious plasma levels," they added.
This study was supported by Boehringer Ingelheim. Dr. Sequist reported ties to Boehringer Ingelheim, Clovis Oncology, Merrimack Pharmaceuticals, and Daiichi Sankyo, and her associates reported ties to numerous industry sources.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Major finding: Progression-free survival was significantly longer with afatinib (11.1 months) than with combination chemotherapy (6.9 months), treatment response rate was significantly higher (56% vs 23%), and median duration of disease control was longer (13.6 months vs 8.1 months).
Data source: An international phase-III open-label randomized clinical trial involving 340 patients with advanced treatment-naive NSCLC and EGFR mutations.
Disclosures: This study was supported by Boehringer Ingelheim. Dr. Sequist reported ties to Boehringer Ingelheim, Clovis Oncology, Merrimack Pharmaceuticals, and Daiichi Sankyo, and her associates reported ties to numerous industry sources.
Thalidomide induced remission in pediatric Crohn’s
An 8-week course of daily oral thalidomide induced clinical remission in 13 of 28 children and adolescents with refractory Crohn’s disease who participated in the first multicenter, double-blind, placebo-controlled clinical trial for this indication, which was reported online Nov. 26 in JAMA.
Thalidomide also consistently reduced scores on the Pediatric Crohn Disease Activity Index (PCDAI), decreased erythrocyte sedimentation rate, improved weight-for-age measures, and increased physicians’ global assessment scores, said Marzia Lazzerini, Ph.D., of the Institute for Maternal and Child Health, Trieste (Italy), and her associates.
The drug also induced longer maintenance of remission than did placebo in an extended follow-up phase of the trial, they noted.
"These findings require replication to definitively determine the utility of this treatment," the investigators said.
Previous observational studies have reported that thalidomide yielded "encouraging results" in patients with Crohn’s disease, with remission rates of 40%-70%. The drug also has proved effective against inflammatory diseases of the skin and mucous membranes.
Dr. Lazzerini and her colleagues performed their clinical trial in 54 patients aged 2-18 years, who were treated at six pediatric tertiary care centers in Italy over a 4-year period.
All the study participants had active Crohn’s disease despite receiving immunosuppressant therapy, or had been forced to discontinue such therapy because of adverse events or increasing resistance to its beneficial effects. Previous treatments included prednisone, azathioprine, mercaptopurine, methotrexate, infliximab, and oral or intravenous cyclosporine.
A total of 28 patients were randomly assigned to receive thalidomide at a dose appropriate for their weight, and 26 were randomized to receive a matching placebo for 8 weeks. Patients, parents, and health caregivers were blinded to treatment assignment. In an open-label extension phase of the study, patients in the placebo group who were not in clinical remission were given thalidomide and followed for an additional 8 weeks.
All patients who responded to thalidomide were then followed for a minimum of 1 year to monitor long-term efficacy and adverse events.
The primary efficacy endpoints were clinical remission at week 8, a 25% reduction in PCDAI score at week 4, or a 75% or greater reduction in PCDAI score at week 4 or week 8.
Thirteen of the 28 patients receiving thalidomide (46.4%) achieved clinical remission at week 8, compared with 3 of 26 patients receiving placebo (11.5%), for a risk ratio of 4. The number of patients who needed to be treated to achieve one clinical remission was 2.86.
The other efficacy endpoints also strongly favored thalidomide: 64.2% of the thalidomide group had a 25% response at week 8, compared with only 30.8% of the placebo group, and 46.4% of the thalidomide group had a 75% response at week 8, compared with only 11.5% of the placebo group.
A total of 21 patients who failed to respond to placebo received thalidomide in the extended phase of the study. Eleven (52.4%) of them achieved clinical remission at week 8, for a risk ratio of 4.5. The number needed to treat was 2.45.
The overall remission rate for both phases of the trial was 63.3%, and the mean time to reach remission was 10 weeks, the investigators reported (JAMA 2013 Nov. 26 [doi: 10.1001/jama.2013.280777]).
The mean duration of clinical remission was 181.1 weeks among the patients who received thalidomide, compared with 6.3 weeks among those who received placebo.
There were nine severe adverse events requiring the suspension of treatment, with peripheral neuropathy, a known adverse effect of thalidomide, being the most frequent. Other severe adverse events included amenorrhea and bradycardia, which also were reported previously in association with thalidomide. "Patients treated with thalidomide should be monitored for bradycardia, and dose reduction or discontinuation may be required," Dr. Lazzerini and her associates said.
One patient developed an acute neurologic event while taking thalidomide, which was thought to be possibly a migraine or transient ischemic attack. Another child had a seizure and was diagnosed as having idiopathic epilepsy unrelated to treatment. Thalidomide was discontinued in both patients.
"All immunosuppressants used for treating Crohn’s disease present a risk of severe adverse events," the researchers noted. "Overall, this study suggests that safety of thalidomide in children with Crohn disease may be acceptable compared with that of other drugs. However, the study was clearly underpowered to detect rare adverse events."
This study was supported by the Italian Medicines Agency. Pharmion/Celgene, the manufacturer of thalidomide, provided the study drug and matching placebo. Dr. Lazzerini and her associates reported no financial conflicts of interest.
An 8-week course of daily oral thalidomide induced clinical remission in 13 of 28 children and adolescents with refractory Crohn’s disease who participated in the first multicenter, double-blind, placebo-controlled clinical trial for this indication, which was reported online Nov. 26 in JAMA.
Thalidomide also consistently reduced scores on the Pediatric Crohn Disease Activity Index (PCDAI), decreased erythrocyte sedimentation rate, improved weight-for-age measures, and increased physicians’ global assessment scores, said Marzia Lazzerini, Ph.D., of the Institute for Maternal and Child Health, Trieste (Italy), and her associates.
The drug also induced longer maintenance of remission than did placebo in an extended follow-up phase of the trial, they noted.
"These findings require replication to definitively determine the utility of this treatment," the investigators said.
Previous observational studies have reported that thalidomide yielded "encouraging results" in patients with Crohn’s disease, with remission rates of 40%-70%. The drug also has proved effective against inflammatory diseases of the skin and mucous membranes.
Dr. Lazzerini and her colleagues performed their clinical trial in 54 patients aged 2-18 years, who were treated at six pediatric tertiary care centers in Italy over a 4-year period.
All the study participants had active Crohn’s disease despite receiving immunosuppressant therapy, or had been forced to discontinue such therapy because of adverse events or increasing resistance to its beneficial effects. Previous treatments included prednisone, azathioprine, mercaptopurine, methotrexate, infliximab, and oral or intravenous cyclosporine.
A total of 28 patients were randomly assigned to receive thalidomide at a dose appropriate for their weight, and 26 were randomized to receive a matching placebo for 8 weeks. Patients, parents, and health caregivers were blinded to treatment assignment. In an open-label extension phase of the study, patients in the placebo group who were not in clinical remission were given thalidomide and followed for an additional 8 weeks.
All patients who responded to thalidomide were then followed for a minimum of 1 year to monitor long-term efficacy and adverse events.
The primary efficacy endpoints were clinical remission at week 8, a 25% reduction in PCDAI score at week 4, or a 75% or greater reduction in PCDAI score at week 4 or week 8.
Thirteen of the 28 patients receiving thalidomide (46.4%) achieved clinical remission at week 8, compared with 3 of 26 patients receiving placebo (11.5%), for a risk ratio of 4. The number of patients who needed to be treated to achieve one clinical remission was 2.86.
The other efficacy endpoints also strongly favored thalidomide: 64.2% of the thalidomide group had a 25% response at week 8, compared with only 30.8% of the placebo group, and 46.4% of the thalidomide group had a 75% response at week 8, compared with only 11.5% of the placebo group.
A total of 21 patients who failed to respond to placebo received thalidomide in the extended phase of the study. Eleven (52.4%) of them achieved clinical remission at week 8, for a risk ratio of 4.5. The number needed to treat was 2.45.
The overall remission rate for both phases of the trial was 63.3%, and the mean time to reach remission was 10 weeks, the investigators reported (JAMA 2013 Nov. 26 [doi: 10.1001/jama.2013.280777]).
The mean duration of clinical remission was 181.1 weeks among the patients who received thalidomide, compared with 6.3 weeks among those who received placebo.
There were nine severe adverse events requiring the suspension of treatment, with peripheral neuropathy, a known adverse effect of thalidomide, being the most frequent. Other severe adverse events included amenorrhea and bradycardia, which also were reported previously in association with thalidomide. "Patients treated with thalidomide should be monitored for bradycardia, and dose reduction or discontinuation may be required," Dr. Lazzerini and her associates said.
One patient developed an acute neurologic event while taking thalidomide, which was thought to be possibly a migraine or transient ischemic attack. Another child had a seizure and was diagnosed as having idiopathic epilepsy unrelated to treatment. Thalidomide was discontinued in both patients.
"All immunosuppressants used for treating Crohn’s disease present a risk of severe adverse events," the researchers noted. "Overall, this study suggests that safety of thalidomide in children with Crohn disease may be acceptable compared with that of other drugs. However, the study was clearly underpowered to detect rare adverse events."
This study was supported by the Italian Medicines Agency. Pharmion/Celgene, the manufacturer of thalidomide, provided the study drug and matching placebo. Dr. Lazzerini and her associates reported no financial conflicts of interest.
An 8-week course of daily oral thalidomide induced clinical remission in 13 of 28 children and adolescents with refractory Crohn’s disease who participated in the first multicenter, double-blind, placebo-controlled clinical trial for this indication, which was reported online Nov. 26 in JAMA.
Thalidomide also consistently reduced scores on the Pediatric Crohn Disease Activity Index (PCDAI), decreased erythrocyte sedimentation rate, improved weight-for-age measures, and increased physicians’ global assessment scores, said Marzia Lazzerini, Ph.D., of the Institute for Maternal and Child Health, Trieste (Italy), and her associates.
The drug also induced longer maintenance of remission than did placebo in an extended follow-up phase of the trial, they noted.
"These findings require replication to definitively determine the utility of this treatment," the investigators said.
Previous observational studies have reported that thalidomide yielded "encouraging results" in patients with Crohn’s disease, with remission rates of 40%-70%. The drug also has proved effective against inflammatory diseases of the skin and mucous membranes.
Dr. Lazzerini and her colleagues performed their clinical trial in 54 patients aged 2-18 years, who were treated at six pediatric tertiary care centers in Italy over a 4-year period.
All the study participants had active Crohn’s disease despite receiving immunosuppressant therapy, or had been forced to discontinue such therapy because of adverse events or increasing resistance to its beneficial effects. Previous treatments included prednisone, azathioprine, mercaptopurine, methotrexate, infliximab, and oral or intravenous cyclosporine.
A total of 28 patients were randomly assigned to receive thalidomide at a dose appropriate for their weight, and 26 were randomized to receive a matching placebo for 8 weeks. Patients, parents, and health caregivers were blinded to treatment assignment. In an open-label extension phase of the study, patients in the placebo group who were not in clinical remission were given thalidomide and followed for an additional 8 weeks.
All patients who responded to thalidomide were then followed for a minimum of 1 year to monitor long-term efficacy and adverse events.
The primary efficacy endpoints were clinical remission at week 8, a 25% reduction in PCDAI score at week 4, or a 75% or greater reduction in PCDAI score at week 4 or week 8.
Thirteen of the 28 patients receiving thalidomide (46.4%) achieved clinical remission at week 8, compared with 3 of 26 patients receiving placebo (11.5%), for a risk ratio of 4. The number of patients who needed to be treated to achieve one clinical remission was 2.86.
The other efficacy endpoints also strongly favored thalidomide: 64.2% of the thalidomide group had a 25% response at week 8, compared with only 30.8% of the placebo group, and 46.4% of the thalidomide group had a 75% response at week 8, compared with only 11.5% of the placebo group.
A total of 21 patients who failed to respond to placebo received thalidomide in the extended phase of the study. Eleven (52.4%) of them achieved clinical remission at week 8, for a risk ratio of 4.5. The number needed to treat was 2.45.
The overall remission rate for both phases of the trial was 63.3%, and the mean time to reach remission was 10 weeks, the investigators reported (JAMA 2013 Nov. 26 [doi: 10.1001/jama.2013.280777]).
The mean duration of clinical remission was 181.1 weeks among the patients who received thalidomide, compared with 6.3 weeks among those who received placebo.
There were nine severe adverse events requiring the suspension of treatment, with peripheral neuropathy, a known adverse effect of thalidomide, being the most frequent. Other severe adverse events included amenorrhea and bradycardia, which also were reported previously in association with thalidomide. "Patients treated with thalidomide should be monitored for bradycardia, and dose reduction or discontinuation may be required," Dr. Lazzerini and her associates said.
One patient developed an acute neurologic event while taking thalidomide, which was thought to be possibly a migraine or transient ischemic attack. Another child had a seizure and was diagnosed as having idiopathic epilepsy unrelated to treatment. Thalidomide was discontinued in both patients.
"All immunosuppressants used for treating Crohn’s disease present a risk of severe adverse events," the researchers noted. "Overall, this study suggests that safety of thalidomide in children with Crohn disease may be acceptable compared with that of other drugs. However, the study was clearly underpowered to detect rare adverse events."
This study was supported by the Italian Medicines Agency. Pharmion/Celgene, the manufacturer of thalidomide, provided the study drug and matching placebo. Dr. Lazzerini and her associates reported no financial conflicts of interest.
FROM JAMA
Major finding: Of 28 patients receiving thalidomide, 13 (46.4%) achieved clinical remission at week 8, compared with 3 of 26 patients receiving placebo (11.5%), for a risk ratio of 4.
Data source: A multicenter, double-blind, placebo-controlled, randomized trial involving 54 patients aged 2-18 years with refractory, active Crohn’s disease who were treated for 8 weeks and followed for at least 1 year.
Disclosures: This study was supported by the Italian Medicines Agency. Pharmion/Celgene, the manufacturer of thalidomide, provided the study drug and matching placebo. Dr. Lazzerini and her associates reported no financial conflicts of interest.
Chest pain traits didn’t identify MI in women
The telltale traits of acute myocardial infarction–associated chest pain may differ in women and men, but they aren’t diagnostically telling enough to determine which women with chest pain are having a heart attack, according to a report published online Nov. 25 in JAMA Internal Medicine.
In a large international study, 11 of 34 possible traits related to chest pain were found to be significantly different in women compared with men who presented to an emergency department with acute chest pain and possible MI. But none of these pain characteristics was clinically useful in distinguishing MI from other, non-MI causes of chest pain in women, said Dr. Maria Rubini Gimenez of the cardiology department, University Hospital Basel (Switzerland), and her associates (JAMA Intern. Med. 2013 Nov. 25 [doi: 10.1001/jamainternmed.2013.12199]).
The investigators performed the ongoing APACE (Advantageous Predictors of Acute Coronary Syndrome Evaluation) study to determine whether sex-specific chest pain characteristics could be used to differentiate women who had acute MI from women who had other causes of chest pain, including unstable angina, cardiac but noncoronary disease such as tachyarrhythmia or perimyocarditis, or noncardiac chest pain.
They assessed 2,475 consecutive patients – 796 women and 1,679 men – who presented with symptoms suggestive of acute MI to nine medical centers in Switzerland, Spain, and Italy during a 6-year period.
In addition to receiving a routine clinical assessment, all of the study’s patients were assessed for 34 characteristics of chest pain, including severity, location (midchest, left side, right side, inframammillary, or supramammillary), size of the pain area (more than or less than 3 cm in diameter), pain quality (pressure, stabbing, burning, aching, and related or unrelated to dyspnea), radiation pattern, onset, duration, dynamics (whether it was currently increasing, decreasing, or stable), aggravating or inducing factors (whether pain was worsened by exertion, change in position, coughing, palpation, or emotional stress), and relieving factors (such as nitrate therapy).
A total of 143 women (18%) and 369 men (22%) were found to have acute MI.
Most of the 34 chest pain characteristics were reported with similar frequency in women and men. Only 11 traits differed significantly between the sexes. Women were significantly more likely than men to report greater pain severity, pressurelike pain, accompanying dyspnea, pain aggravated by palpation, pain radiating to the throat or back, sudden onset of pain, and pain duration of longer than 30 minutes. And women were significantly less likely to report no pain radiation, radiation to the right side, or shorter pain duration.
When the analysis was restricted to patients who had only acute MI, only 5 of the 34 chest pain characteristics differed significantly between women and men. Women with acute MI were less likely than men with acute MI to report pain radiation to the right arm/shoulder, short duration of pain, and pain that decreased with time. They were more likely than men to report pain radiation to the back and pain duration of 30 minutes or more, the investigators reported.
When the analysis turned to chest pain characteristics that could distinguish acute MI from other causes, only 3 of the 34 traits (8%) contributed to sex-specific diagnosis. Chest pain duration of only 2-30 minutes weakly decreased the likelihood of an MI diagnosis in women but weakly increased it in men. Pain duration of more than 30 minutes weakly increased the likelihood of an MI diagnosis in women but was neutral in men. And decreasing pain intensity weakly decreased the likelihood of an MI diagnosis in women but weakly increased the likelihood in men.
However, none of those three chest pain characteristics was considered to be clinically helpful in establishing an MI diagnosis, Dr. Gimenez and her associates said.
"Our data confirm that [chest pain characteristics] are not powerful enough to be used as a single tool in the diagnosis of AMI [acute myocardial infarction] and need to be used always in conjunction with the ECG and [cardiac troponin] test results in the diagnosis of AMI," the researchers noted. Given that the differences were small in those characteristics’ sex-specific diagnostic performance, "our findings do not seem to support the use of [chest pain characteristics] specific to women in the early diagnosis of AMI in women," they concluded.
This study was supported by the Swiss National Science Foundation, the Swiss Heart Foundation, the Cardiovascular Research Foundation Basel, the University of Basel, and University Hospital Basel. Dr. Gimenez reported no financial conflicts of interest, and her associates reported ties to 8sense, Abbott, Alere, and other companies.
The telltale traits of acute myocardial infarction–associated chest pain may differ in women and men, but they aren’t diagnostically telling enough to determine which women with chest pain are having a heart attack, according to a report published online Nov. 25 in JAMA Internal Medicine.
In a large international study, 11 of 34 possible traits related to chest pain were found to be significantly different in women compared with men who presented to an emergency department with acute chest pain and possible MI. But none of these pain characteristics was clinically useful in distinguishing MI from other, non-MI causes of chest pain in women, said Dr. Maria Rubini Gimenez of the cardiology department, University Hospital Basel (Switzerland), and her associates (JAMA Intern. Med. 2013 Nov. 25 [doi: 10.1001/jamainternmed.2013.12199]).
The investigators performed the ongoing APACE (Advantageous Predictors of Acute Coronary Syndrome Evaluation) study to determine whether sex-specific chest pain characteristics could be used to differentiate women who had acute MI from women who had other causes of chest pain, including unstable angina, cardiac but noncoronary disease such as tachyarrhythmia or perimyocarditis, or noncardiac chest pain.
They assessed 2,475 consecutive patients – 796 women and 1,679 men – who presented with symptoms suggestive of acute MI to nine medical centers in Switzerland, Spain, and Italy during a 6-year period.
In addition to receiving a routine clinical assessment, all of the study’s patients were assessed for 34 characteristics of chest pain, including severity, location (midchest, left side, right side, inframammillary, or supramammillary), size of the pain area (more than or less than 3 cm in diameter), pain quality (pressure, stabbing, burning, aching, and related or unrelated to dyspnea), radiation pattern, onset, duration, dynamics (whether it was currently increasing, decreasing, or stable), aggravating or inducing factors (whether pain was worsened by exertion, change in position, coughing, palpation, or emotional stress), and relieving factors (such as nitrate therapy).
A total of 143 women (18%) and 369 men (22%) were found to have acute MI.
Most of the 34 chest pain characteristics were reported with similar frequency in women and men. Only 11 traits differed significantly between the sexes. Women were significantly more likely than men to report greater pain severity, pressurelike pain, accompanying dyspnea, pain aggravated by palpation, pain radiating to the throat or back, sudden onset of pain, and pain duration of longer than 30 minutes. And women were significantly less likely to report no pain radiation, radiation to the right side, or shorter pain duration.
When the analysis was restricted to patients who had only acute MI, only 5 of the 34 chest pain characteristics differed significantly between women and men. Women with acute MI were less likely than men with acute MI to report pain radiation to the right arm/shoulder, short duration of pain, and pain that decreased with time. They were more likely than men to report pain radiation to the back and pain duration of 30 minutes or more, the investigators reported.
When the analysis turned to chest pain characteristics that could distinguish acute MI from other causes, only 3 of the 34 traits (8%) contributed to sex-specific diagnosis. Chest pain duration of only 2-30 minutes weakly decreased the likelihood of an MI diagnosis in women but weakly increased it in men. Pain duration of more than 30 minutes weakly increased the likelihood of an MI diagnosis in women but was neutral in men. And decreasing pain intensity weakly decreased the likelihood of an MI diagnosis in women but weakly increased the likelihood in men.
However, none of those three chest pain characteristics was considered to be clinically helpful in establishing an MI diagnosis, Dr. Gimenez and her associates said.
"Our data confirm that [chest pain characteristics] are not powerful enough to be used as a single tool in the diagnosis of AMI [acute myocardial infarction] and need to be used always in conjunction with the ECG and [cardiac troponin] test results in the diagnosis of AMI," the researchers noted. Given that the differences were small in those characteristics’ sex-specific diagnostic performance, "our findings do not seem to support the use of [chest pain characteristics] specific to women in the early diagnosis of AMI in women," they concluded.
This study was supported by the Swiss National Science Foundation, the Swiss Heart Foundation, the Cardiovascular Research Foundation Basel, the University of Basel, and University Hospital Basel. Dr. Gimenez reported no financial conflicts of interest, and her associates reported ties to 8sense, Abbott, Alere, and other companies.
The telltale traits of acute myocardial infarction–associated chest pain may differ in women and men, but they aren’t diagnostically telling enough to determine which women with chest pain are having a heart attack, according to a report published online Nov. 25 in JAMA Internal Medicine.
In a large international study, 11 of 34 possible traits related to chest pain were found to be significantly different in women compared with men who presented to an emergency department with acute chest pain and possible MI. But none of these pain characteristics was clinically useful in distinguishing MI from other, non-MI causes of chest pain in women, said Dr. Maria Rubini Gimenez of the cardiology department, University Hospital Basel (Switzerland), and her associates (JAMA Intern. Med. 2013 Nov. 25 [doi: 10.1001/jamainternmed.2013.12199]).
The investigators performed the ongoing APACE (Advantageous Predictors of Acute Coronary Syndrome Evaluation) study to determine whether sex-specific chest pain characteristics could be used to differentiate women who had acute MI from women who had other causes of chest pain, including unstable angina, cardiac but noncoronary disease such as tachyarrhythmia or perimyocarditis, or noncardiac chest pain.
They assessed 2,475 consecutive patients – 796 women and 1,679 men – who presented with symptoms suggestive of acute MI to nine medical centers in Switzerland, Spain, and Italy during a 6-year period.
In addition to receiving a routine clinical assessment, all of the study’s patients were assessed for 34 characteristics of chest pain, including severity, location (midchest, left side, right side, inframammillary, or supramammillary), size of the pain area (more than or less than 3 cm in diameter), pain quality (pressure, stabbing, burning, aching, and related or unrelated to dyspnea), radiation pattern, onset, duration, dynamics (whether it was currently increasing, decreasing, or stable), aggravating or inducing factors (whether pain was worsened by exertion, change in position, coughing, palpation, or emotional stress), and relieving factors (such as nitrate therapy).
A total of 143 women (18%) and 369 men (22%) were found to have acute MI.
Most of the 34 chest pain characteristics were reported with similar frequency in women and men. Only 11 traits differed significantly between the sexes. Women were significantly more likely than men to report greater pain severity, pressurelike pain, accompanying dyspnea, pain aggravated by palpation, pain radiating to the throat or back, sudden onset of pain, and pain duration of longer than 30 minutes. And women were significantly less likely to report no pain radiation, radiation to the right side, or shorter pain duration.
When the analysis was restricted to patients who had only acute MI, only 5 of the 34 chest pain characteristics differed significantly between women and men. Women with acute MI were less likely than men with acute MI to report pain radiation to the right arm/shoulder, short duration of pain, and pain that decreased with time. They were more likely than men to report pain radiation to the back and pain duration of 30 minutes or more, the investigators reported.
When the analysis turned to chest pain characteristics that could distinguish acute MI from other causes, only 3 of the 34 traits (8%) contributed to sex-specific diagnosis. Chest pain duration of only 2-30 minutes weakly decreased the likelihood of an MI diagnosis in women but weakly increased it in men. Pain duration of more than 30 minutes weakly increased the likelihood of an MI diagnosis in women but was neutral in men. And decreasing pain intensity weakly decreased the likelihood of an MI diagnosis in women but weakly increased the likelihood in men.
However, none of those three chest pain characteristics was considered to be clinically helpful in establishing an MI diagnosis, Dr. Gimenez and her associates said.
"Our data confirm that [chest pain characteristics] are not powerful enough to be used as a single tool in the diagnosis of AMI [acute myocardial infarction] and need to be used always in conjunction with the ECG and [cardiac troponin] test results in the diagnosis of AMI," the researchers noted. Given that the differences were small in those characteristics’ sex-specific diagnostic performance, "our findings do not seem to support the use of [chest pain characteristics] specific to women in the early diagnosis of AMI in women," they concluded.
This study was supported by the Swiss National Science Foundation, the Swiss Heart Foundation, the Cardiovascular Research Foundation Basel, the University of Basel, and University Hospital Basel. Dr. Gimenez reported no financial conflicts of interest, and her associates reported ties to 8sense, Abbott, Alere, and other companies.
FROM JAMA INTERNAL MEDICINE
Major finding: Only 3 of the 34 characteristics of chest pain differed significantly enough between men and women to possibly enhance diagnosis of myocardial infarction in the women, but those 3 were so weakly significant that they were deemed to be "not helpful" at distinguishing MI-related chest pain from non–MI-related chest pain in the clinical setting.
Data source: An international prospective study of 2,475 consecutive patients presenting to emergency departments with chest pain and possible MI during a 6-year period, who gave detailed reports on 34 characteristics of the chest pain.
Disclosures: This study was supported by the Swiss National Science Foundation, the Swiss Heart Foundation, the Cardiovascular Research Foundation Basel, the University of Basel, and University Hospital Basel. Dr. Gimenez reported no financial conflicts of interest, and her associates reported ties to 8sense, Abbott, Alere, and other companies.
Clinician Recommendation Key to HPV Vaccine Coverage
Better vaccine coverage of adolescents against HPV infection hinges on clinicians and other health care professionals both recommending the vaccine and educating patients and parents about its importance, according to a report published online Nov. 25 in JAMA Pediatrics.
In a review of the extensive literature concerning barriers to HPV vaccination among patients aged 11-17 years, health care professionals’ failure to recommend the vaccine was found to be a leading obstacle to more widespread coverage, said Dawn M. Holman of the division of cancer prevention and control, Centers for Disease Control and Prevention, Atlanta, and her associates.
The most recent data from the National Immunization Survey indicates that only 53% of adolescent girls in the United States have received at least one dose of the HPV vaccine series, only 35% have completed all three doses in the series, and only 8% of adolescent boys in the United States have initiated the HPV vaccine series. By comparison, coverage for the meningococcal conjugate vaccine is approximately 70% and coverage for the tetanus, diphtheria, and acellular pertussis vaccine is 78% in the same age group.
Ms. Holman and her associates performed a systematic review of the literature and found 55 studies published in 2009-2012 that addressed obstacles to HPV vaccination in this age group.
In ten articles that addressed barriers to HPV vaccination among health care professionals, researchers found that while most physicians support HPV vaccination and offer it to their patients, many tend not to offer it to the youngest adolescents or to boys. Many also offer HPV vaccination only according to their own perception of the patient’s risk of contracting the virus. And many do not appear to understand the importance of giving the HPV vaccine well before adolescents become sexually active.
Health care professionals also tend not to provide detailed information to either patients or their parents regarding the risks and benefits of HPV vaccination, or they emphasize the fact that it is optional – both of which come across to patients and parents as a less-than-ringing endorsement. Some health care professionals reported that HPV vaccination is under the parents’ control rather than theirs, and many cited the cost of the HPV vaccine, and the lack or lateness of insurance reimbursement, as a significant obstacle.
In contrast, concerns about the safety and efficacy of the HPV vaccine were rarely cited as contributing to poor coverage (JAMA Pediatrics 2013 Nov. 25 [doi:10.1001/jamapediatrics.2013.2752]).
A total of 64% of the reviewed articles involved barriers to vaccination among parents. Not having a physician recommendation and not getting enough information about the HPV vaccine from health care providers were the chief obstacles to greater coverage in this group. Certain religious affiliations also were associated with opposition to the HPV vaccine.
In contrast, costs and lack of access were cited infrequently. Few parents reported concerns about vaccine safety or efficacy, and few reported concern that the vaccination might affect their child’s sexual behavior.
"Providing adequate, clear, and accessible information to parents about HPV infection, vaccine safety, adverse effects, and the appropriate age for vaccination may be one way health care professionals can reduce concerns and misconceptions about the vaccine," Ms. Holman and her colleagues noted.
Other strategies to improve HPV vaccination coverage include educating health care professionals and the public in general about the importance of vaccinating adolescent males, and ensuring that underserved populations are informed about and given access to the vaccine, they added.
Better vaccine coverage of adolescents against HPV infection hinges on clinicians and other health care professionals both recommending the vaccine and educating patients and parents about its importance, according to a report published online Nov. 25 in JAMA Pediatrics.
In a review of the extensive literature concerning barriers to HPV vaccination among patients aged 11-17 years, health care professionals’ failure to recommend the vaccine was found to be a leading obstacle to more widespread coverage, said Dawn M. Holman of the division of cancer prevention and control, Centers for Disease Control and Prevention, Atlanta, and her associates.
The most recent data from the National Immunization Survey indicates that only 53% of adolescent girls in the United States have received at least one dose of the HPV vaccine series, only 35% have completed all three doses in the series, and only 8% of adolescent boys in the United States have initiated the HPV vaccine series. By comparison, coverage for the meningococcal conjugate vaccine is approximately 70% and coverage for the tetanus, diphtheria, and acellular pertussis vaccine is 78% in the same age group.
Ms. Holman and her associates performed a systematic review of the literature and found 55 studies published in 2009-2012 that addressed obstacles to HPV vaccination in this age group.
In ten articles that addressed barriers to HPV vaccination among health care professionals, researchers found that while most physicians support HPV vaccination and offer it to their patients, many tend not to offer it to the youngest adolescents or to boys. Many also offer HPV vaccination only according to their own perception of the patient’s risk of contracting the virus. And many do not appear to understand the importance of giving the HPV vaccine well before adolescents become sexually active.
Health care professionals also tend not to provide detailed information to either patients or their parents regarding the risks and benefits of HPV vaccination, or they emphasize the fact that it is optional – both of which come across to patients and parents as a less-than-ringing endorsement. Some health care professionals reported that HPV vaccination is under the parents’ control rather than theirs, and many cited the cost of the HPV vaccine, and the lack or lateness of insurance reimbursement, as a significant obstacle.
In contrast, concerns about the safety and efficacy of the HPV vaccine were rarely cited as contributing to poor coverage (JAMA Pediatrics 2013 Nov. 25 [doi:10.1001/jamapediatrics.2013.2752]).
A total of 64% of the reviewed articles involved barriers to vaccination among parents. Not having a physician recommendation and not getting enough information about the HPV vaccine from health care providers were the chief obstacles to greater coverage in this group. Certain religious affiliations also were associated with opposition to the HPV vaccine.
In contrast, costs and lack of access were cited infrequently. Few parents reported concerns about vaccine safety or efficacy, and few reported concern that the vaccination might affect their child’s sexual behavior.
"Providing adequate, clear, and accessible information to parents about HPV infection, vaccine safety, adverse effects, and the appropriate age for vaccination may be one way health care professionals can reduce concerns and misconceptions about the vaccine," Ms. Holman and her colleagues noted.
Other strategies to improve HPV vaccination coverage include educating health care professionals and the public in general about the importance of vaccinating adolescent males, and ensuring that underserved populations are informed about and given access to the vaccine, they added.
Better vaccine coverage of adolescents against HPV infection hinges on clinicians and other health care professionals both recommending the vaccine and educating patients and parents about its importance, according to a report published online Nov. 25 in JAMA Pediatrics.
In a review of the extensive literature concerning barriers to HPV vaccination among patients aged 11-17 years, health care professionals’ failure to recommend the vaccine was found to be a leading obstacle to more widespread coverage, said Dawn M. Holman of the division of cancer prevention and control, Centers for Disease Control and Prevention, Atlanta, and her associates.
The most recent data from the National Immunization Survey indicates that only 53% of adolescent girls in the United States have received at least one dose of the HPV vaccine series, only 35% have completed all three doses in the series, and only 8% of adolescent boys in the United States have initiated the HPV vaccine series. By comparison, coverage for the meningococcal conjugate vaccine is approximately 70% and coverage for the tetanus, diphtheria, and acellular pertussis vaccine is 78% in the same age group.
Ms. Holman and her associates performed a systematic review of the literature and found 55 studies published in 2009-2012 that addressed obstacles to HPV vaccination in this age group.
In ten articles that addressed barriers to HPV vaccination among health care professionals, researchers found that while most physicians support HPV vaccination and offer it to their patients, many tend not to offer it to the youngest adolescents or to boys. Many also offer HPV vaccination only according to their own perception of the patient’s risk of contracting the virus. And many do not appear to understand the importance of giving the HPV vaccine well before adolescents become sexually active.
Health care professionals also tend not to provide detailed information to either patients or their parents regarding the risks and benefits of HPV vaccination, or they emphasize the fact that it is optional – both of which come across to patients and parents as a less-than-ringing endorsement. Some health care professionals reported that HPV vaccination is under the parents’ control rather than theirs, and many cited the cost of the HPV vaccine, and the lack or lateness of insurance reimbursement, as a significant obstacle.
In contrast, concerns about the safety and efficacy of the HPV vaccine were rarely cited as contributing to poor coverage (JAMA Pediatrics 2013 Nov. 25 [doi:10.1001/jamapediatrics.2013.2752]).
A total of 64% of the reviewed articles involved barriers to vaccination among parents. Not having a physician recommendation and not getting enough information about the HPV vaccine from health care providers were the chief obstacles to greater coverage in this group. Certain religious affiliations also were associated with opposition to the HPV vaccine.
In contrast, costs and lack of access were cited infrequently. Few parents reported concerns about vaccine safety or efficacy, and few reported concern that the vaccination might affect their child’s sexual behavior.
"Providing adequate, clear, and accessible information to parents about HPV infection, vaccine safety, adverse effects, and the appropriate age for vaccination may be one way health care professionals can reduce concerns and misconceptions about the vaccine," Ms. Holman and her colleagues noted.
Other strategies to improve HPV vaccination coverage include educating health care professionals and the public in general about the importance of vaccinating adolescent males, and ensuring that underserved populations are informed about and given access to the vaccine, they added.
FROM JAMA PEDIATRICS
Physician recommendation key to HPV vaccine coverage
Better vaccine coverage of adolescents against HPV infection hinges on physicians and other health care professionals both recommending the vaccine and educating patients and parents about its importance, according to a report published online Nov. 25 in JAMA Pediatrics.
In a review of the extensive literature concerning barriers to HPV vaccination among patients aged 11-17 years, health care professionals’ failure to recommend the vaccine was found to be a leading obstacle to more widespread coverage, said Dawn M. Holman of the division of cancer prevention and control, Centers for Disease Control and Prevention, Atlanta, and her associates.
The most recent data from the National Immunization Survey indicates that only 53% of adolescent girls in the United States have received at least one dose of the HPV vaccine series, only 35% have completed all three doses in the series, and only 8% of adolescent boys in the United States have initiated the HPV vaccine series. By comparison, coverage for the meningococcal conjugate vaccine is approximately 70% and coverage for the tetanus, diphtheria, and acellular pertussis vaccine is 78% in the same age group.
Ms. Holman and her associates performed a systematic review of the literature and found 55 studies published in 2009-2012 that addressed obstacles to HPV vaccination in this age group.
In ten articles that addressed barriers to HPV vaccination among health care professionals, researchers found that while most physicians support HPV vaccination and offer it to their patients, many tend not to offer it to the youngest adolescents or to boys. Many also offer HPV vaccination only according to their own perception of the patient’s risk of contracting the virus. And many do not appear to understand the importance of giving the HPV vaccine well before adolescents become sexually active.
Health care professionals also tend not to provide detailed information to either patients or their parents regarding the risks and benefits of HPV vaccination, or they emphasize the fact that it is optional – both of which come across to patients and parents as a less-than-ringing endorsement. Some health care professionals reported that HPV vaccination is under the parents’ control rather than theirs, and many cited the cost of the HPV vaccine, and the lack or lateness of insurance reimbursement, as a significant obstacle.
In contrast, concerns about the safety and efficacy of the HPV vaccine were rarely cited as contributing to poor coverage (JAMA Pediatrics 2013 Nov. 25 [doi:10.1001/jamapediatrics.2013.2752]).
A total of 64% of the reviewed articles involved barriers to vaccination among parents. Not having a physician recommendation and not getting enough information about the HPV vaccine from health care providers were the chief obstacles to greater coverage in this group. Certain religious affiliations also were associated with opposition to the HPV vaccine.
In contrast, costs and lack of access were cited infrequently. Few parents reported concerns about vaccine safety or efficacy, and few reported concern that the vaccination might affect their child’s sexual behavior.
"Providing adequate, clear, and accessible information to parents about HPV infection, vaccine safety, adverse effects, and the appropriate age for vaccination may be one way health care professionals can reduce concerns and misconceptions about the vaccine," Ms. Holman and her colleagues noted.
Other strategies to improve HPV vaccination coverage include educating health care professionals and the public in general about the importance of vaccinating adolescent males, and ensuring that underserved populations are informed about and given access to the vaccine, they added.
Better vaccine coverage of adolescents against HPV infection hinges on physicians and other health care professionals both recommending the vaccine and educating patients and parents about its importance, according to a report published online Nov. 25 in JAMA Pediatrics.
In a review of the extensive literature concerning barriers to HPV vaccination among patients aged 11-17 years, health care professionals’ failure to recommend the vaccine was found to be a leading obstacle to more widespread coverage, said Dawn M. Holman of the division of cancer prevention and control, Centers for Disease Control and Prevention, Atlanta, and her associates.
The most recent data from the National Immunization Survey indicates that only 53% of adolescent girls in the United States have received at least one dose of the HPV vaccine series, only 35% have completed all three doses in the series, and only 8% of adolescent boys in the United States have initiated the HPV vaccine series. By comparison, coverage for the meningococcal conjugate vaccine is approximately 70% and coverage for the tetanus, diphtheria, and acellular pertussis vaccine is 78% in the same age group.
Ms. Holman and her associates performed a systematic review of the literature and found 55 studies published in 2009-2012 that addressed obstacles to HPV vaccination in this age group.
In ten articles that addressed barriers to HPV vaccination among health care professionals, researchers found that while most physicians support HPV vaccination and offer it to their patients, many tend not to offer it to the youngest adolescents or to boys. Many also offer HPV vaccination only according to their own perception of the patient’s risk of contracting the virus. And many do not appear to understand the importance of giving the HPV vaccine well before adolescents become sexually active.
Health care professionals also tend not to provide detailed information to either patients or their parents regarding the risks and benefits of HPV vaccination, or they emphasize the fact that it is optional – both of which come across to patients and parents as a less-than-ringing endorsement. Some health care professionals reported that HPV vaccination is under the parents’ control rather than theirs, and many cited the cost of the HPV vaccine, and the lack or lateness of insurance reimbursement, as a significant obstacle.
In contrast, concerns about the safety and efficacy of the HPV vaccine were rarely cited as contributing to poor coverage (JAMA Pediatrics 2013 Nov. 25 [doi:10.1001/jamapediatrics.2013.2752]).
A total of 64% of the reviewed articles involved barriers to vaccination among parents. Not having a physician recommendation and not getting enough information about the HPV vaccine from health care providers were the chief obstacles to greater coverage in this group. Certain religious affiliations also were associated with opposition to the HPV vaccine.
In contrast, costs and lack of access were cited infrequently. Few parents reported concerns about vaccine safety or efficacy, and few reported concern that the vaccination might affect their child’s sexual behavior.
"Providing adequate, clear, and accessible information to parents about HPV infection, vaccine safety, adverse effects, and the appropriate age for vaccination may be one way health care professionals can reduce concerns and misconceptions about the vaccine," Ms. Holman and her colleagues noted.
Other strategies to improve HPV vaccination coverage include educating health care professionals and the public in general about the importance of vaccinating adolescent males, and ensuring that underserved populations are informed about and given access to the vaccine, they added.
Better vaccine coverage of adolescents against HPV infection hinges on physicians and other health care professionals both recommending the vaccine and educating patients and parents about its importance, according to a report published online Nov. 25 in JAMA Pediatrics.
In a review of the extensive literature concerning barriers to HPV vaccination among patients aged 11-17 years, health care professionals’ failure to recommend the vaccine was found to be a leading obstacle to more widespread coverage, said Dawn M. Holman of the division of cancer prevention and control, Centers for Disease Control and Prevention, Atlanta, and her associates.
The most recent data from the National Immunization Survey indicates that only 53% of adolescent girls in the United States have received at least one dose of the HPV vaccine series, only 35% have completed all three doses in the series, and only 8% of adolescent boys in the United States have initiated the HPV vaccine series. By comparison, coverage for the meningococcal conjugate vaccine is approximately 70% and coverage for the tetanus, diphtheria, and acellular pertussis vaccine is 78% in the same age group.
Ms. Holman and her associates performed a systematic review of the literature and found 55 studies published in 2009-2012 that addressed obstacles to HPV vaccination in this age group.
In ten articles that addressed barriers to HPV vaccination among health care professionals, researchers found that while most physicians support HPV vaccination and offer it to their patients, many tend not to offer it to the youngest adolescents or to boys. Many also offer HPV vaccination only according to their own perception of the patient’s risk of contracting the virus. And many do not appear to understand the importance of giving the HPV vaccine well before adolescents become sexually active.
Health care professionals also tend not to provide detailed information to either patients or their parents regarding the risks and benefits of HPV vaccination, or they emphasize the fact that it is optional – both of which come across to patients and parents as a less-than-ringing endorsement. Some health care professionals reported that HPV vaccination is under the parents’ control rather than theirs, and many cited the cost of the HPV vaccine, and the lack or lateness of insurance reimbursement, as a significant obstacle.
In contrast, concerns about the safety and efficacy of the HPV vaccine were rarely cited as contributing to poor coverage (JAMA Pediatrics 2013 Nov. 25 [doi:10.1001/jamapediatrics.2013.2752]).
A total of 64% of the reviewed articles involved barriers to vaccination among parents. Not having a physician recommendation and not getting enough information about the HPV vaccine from health care providers were the chief obstacles to greater coverage in this group. Certain religious affiliations also were associated with opposition to the HPV vaccine.
In contrast, costs and lack of access were cited infrequently. Few parents reported concerns about vaccine safety or efficacy, and few reported concern that the vaccination might affect their child’s sexual behavior.
"Providing adequate, clear, and accessible information to parents about HPV infection, vaccine safety, adverse effects, and the appropriate age for vaccination may be one way health care professionals can reduce concerns and misconceptions about the vaccine," Ms. Holman and her colleagues noted.
Other strategies to improve HPV vaccination coverage include educating health care professionals and the public in general about the importance of vaccinating adolescent males, and ensuring that underserved populations are informed about and given access to the vaccine, they added.
FROM JAMA PEDIATRICS
Major Finding: Studies examining barriers to HPV vaccination among health care professionals and parents both cited the lack of a physician’s recommendation to get the vaccine and the lack of information provided to patients and parents as crucial obstacles.
Data Source: A systematic review of the literature of 2009-2012 concerning barriers to wider HPV vaccination coverage among patients aged 11-17 years.
Disclosures: No relevant financial conflicts of interest were reported.