User login
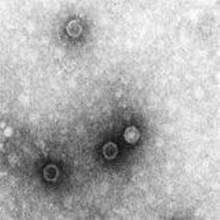
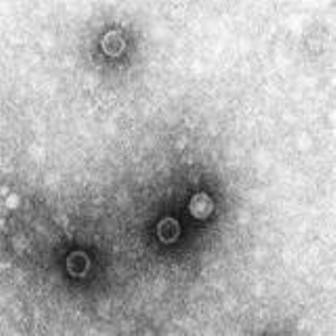
Poliomyelitis outbreak in China controlled rapidly
An outbreak of imported wild-type poliovirus in China was interrupted within 6 weeks of laboratory confirmation of the index case, thanks to an immediate, potent public health response that accorded with World Health Organization recommendations, according to a report published online Nov. 21 in the New England Journal of Medicine.
The last indigenous wild-type poliovirus in China was isolated in 1994, and the country was certified as poliomyelitis free in 2000. This strain of the virus was traced back to Pakistan, which, along with Afghanistan and Nigeria, are the only countries in the world that have failed to control transmission of wild-type poliovirus, said Dr. Hui-Ming Luo of the Chinese Center for Disease Control and Prevention, Beijing, and associates.
"Until wild-type poliovirus transmission is interrupted globally, poliomyelitis-free countries will continue to be at risk for viral importation," they wrote.
In this outbreak, the index case was a 16-month-old girl whose family had no history of travel beyond southern Xinjiang province. She was hospitalized for flaccid paralysis, and an epidemiologic investigation began immediately. Three more cases of flaccid paralysis were identified in the same county, and the Chinese CDC confirmed that the same strain of wild-type poliovirus was present in all cases and that it had been imported from Pakistan.
All together, 21 cases of infection were confirmed and 23 clinically compatible cases were reported in southern Xinjiang. A single sewage sample from the index case’s hometown and another from a sample of river water there were found to be positive for wild-type poliovirus type 1.
"The incidence rates of wild-type poliovirus infection and clinically compatible poliomyelitis were highest among children younger than 1 year of age (3.46/100,000 and 1.15 cases/100,000 population, respectively)", they reported. Just under half of the 21 infections were in children under age 14 years.
The virus was isolated from 14 of 673 (2.1%) healthy people who were in contact with the affected patients and from 13 of 491 (2.6%) healthy people who were not in contact with any patients.
The national and local governments responded according to the 2006 World Health Assembly resolution on such outbreaks: they launched an investigation within 72 hours; conducted five rounds of supplementary immunization, beginning within 4 weeks of the start of the outbreak and continuing after the last cases were reported; surveyed and found that the coverage rates for all five rounds were greater than 95%; and enhanced the surveillance for acute flaccid paralysis throughout the affected province.
"Key response measures were the declaration of a public health emergency, enhancement of the sensitivity of surveillance for acute flaccid paralysis, the conduct of epidemiologically targeted vaccination campaigns, the use of monovalent OPV1 [oral poliovirus vaccine] for two rounds of supplementary immunization, close collaboration with international agencies such as the WHO and the U.S. Centers for Disease Control and Prevention, and provision of adequate resources to mount the response," the investigators said (N. Engl. J. Med. 2013 Nov. 21 [doi:10.1056/NEJMoa1303368]).
This effort was estimated to have cost the equivalent of $26 million U.S. dollars and required more than 500 public health experts to assist the local health workers, government officials, and approximately 500,000 volunteers who were involved.
"The global eradication of poliomyelitis will benefit all countries, including those that have been certified as poliomyelitis-free," the researchers wrote.
No financial conflicts of interest were reported.
Public health authorities in China should be commended for containing this outbreak of poliomyelitis so rapidly, said Dr. Trevor Mundel and Dr. Walter A. Orenstein.
Containment would be much more difficult if a similar outbreak occurred in a country that wasn’t capable of responding so quickly. And as this case shows, all countries are at risk, even if they are currently certified as polio free, as long as polioviruses circulate anywhere in the world. Six such incidents have been reported so far this year, in Somalia, Kenya, Ethiopia, Syria, Cameroon, and Israel.
"Without question, the best defense against poliovirus is a good offense that eliminates the virus from the remaining reservoirs and truly eradicates the disease," they said.
Dr. Mundel is at the Bill and Melinda Gates Foundation, Seattle. Dr. Orenstein is at the Emory University Vaccine Center, Atlanta. They reported no financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Luo’s report (N. Engl. J. Med. 2013 Nov. 21 [doi:10.1056/NEJMe1311591]).
Public health authorities in China should be commended for containing this outbreak of poliomyelitis so rapidly, said Dr. Trevor Mundel and Dr. Walter A. Orenstein.
Containment would be much more difficult if a similar outbreak occurred in a country that wasn’t capable of responding so quickly. And as this case shows, all countries are at risk, even if they are currently certified as polio free, as long as polioviruses circulate anywhere in the world. Six such incidents have been reported so far this year, in Somalia, Kenya, Ethiopia, Syria, Cameroon, and Israel.
"Without question, the best defense against poliovirus is a good offense that eliminates the virus from the remaining reservoirs and truly eradicates the disease," they said.
Dr. Mundel is at the Bill and Melinda Gates Foundation, Seattle. Dr. Orenstein is at the Emory University Vaccine Center, Atlanta. They reported no financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Luo’s report (N. Engl. J. Med. 2013 Nov. 21 [doi:10.1056/NEJMe1311591]).
Public health authorities in China should be commended for containing this outbreak of poliomyelitis so rapidly, said Dr. Trevor Mundel and Dr. Walter A. Orenstein.
Containment would be much more difficult if a similar outbreak occurred in a country that wasn’t capable of responding so quickly. And as this case shows, all countries are at risk, even if they are currently certified as polio free, as long as polioviruses circulate anywhere in the world. Six such incidents have been reported so far this year, in Somalia, Kenya, Ethiopia, Syria, Cameroon, and Israel.
"Without question, the best defense against poliovirus is a good offense that eliminates the virus from the remaining reservoirs and truly eradicates the disease," they said.
Dr. Mundel is at the Bill and Melinda Gates Foundation, Seattle. Dr. Orenstein is at the Emory University Vaccine Center, Atlanta. They reported no financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Luo’s report (N. Engl. J. Med. 2013 Nov. 21 [doi:10.1056/NEJMe1311591]).
An outbreak of imported wild-type poliovirus in China was interrupted within 6 weeks of laboratory confirmation of the index case, thanks to an immediate, potent public health response that accorded with World Health Organization recommendations, according to a report published online Nov. 21 in the New England Journal of Medicine.
The last indigenous wild-type poliovirus in China was isolated in 1994, and the country was certified as poliomyelitis free in 2000. This strain of the virus was traced back to Pakistan, which, along with Afghanistan and Nigeria, are the only countries in the world that have failed to control transmission of wild-type poliovirus, said Dr. Hui-Ming Luo of the Chinese Center for Disease Control and Prevention, Beijing, and associates.
"Until wild-type poliovirus transmission is interrupted globally, poliomyelitis-free countries will continue to be at risk for viral importation," they wrote.
In this outbreak, the index case was a 16-month-old girl whose family had no history of travel beyond southern Xinjiang province. She was hospitalized for flaccid paralysis, and an epidemiologic investigation began immediately. Three more cases of flaccid paralysis were identified in the same county, and the Chinese CDC confirmed that the same strain of wild-type poliovirus was present in all cases and that it had been imported from Pakistan.
All together, 21 cases of infection were confirmed and 23 clinically compatible cases were reported in southern Xinjiang. A single sewage sample from the index case’s hometown and another from a sample of river water there were found to be positive for wild-type poliovirus type 1.
"The incidence rates of wild-type poliovirus infection and clinically compatible poliomyelitis were highest among children younger than 1 year of age (3.46/100,000 and 1.15 cases/100,000 population, respectively)", they reported. Just under half of the 21 infections were in children under age 14 years.
The virus was isolated from 14 of 673 (2.1%) healthy people who were in contact with the affected patients and from 13 of 491 (2.6%) healthy people who were not in contact with any patients.
The national and local governments responded according to the 2006 World Health Assembly resolution on such outbreaks: they launched an investigation within 72 hours; conducted five rounds of supplementary immunization, beginning within 4 weeks of the start of the outbreak and continuing after the last cases were reported; surveyed and found that the coverage rates for all five rounds were greater than 95%; and enhanced the surveillance for acute flaccid paralysis throughout the affected province.
"Key response measures were the declaration of a public health emergency, enhancement of the sensitivity of surveillance for acute flaccid paralysis, the conduct of epidemiologically targeted vaccination campaigns, the use of monovalent OPV1 [oral poliovirus vaccine] for two rounds of supplementary immunization, close collaboration with international agencies such as the WHO and the U.S. Centers for Disease Control and Prevention, and provision of adequate resources to mount the response," the investigators said (N. Engl. J. Med. 2013 Nov. 21 [doi:10.1056/NEJMoa1303368]).
This effort was estimated to have cost the equivalent of $26 million U.S. dollars and required more than 500 public health experts to assist the local health workers, government officials, and approximately 500,000 volunteers who were involved.
"The global eradication of poliomyelitis will benefit all countries, including those that have been certified as poliomyelitis-free," the researchers wrote.
No financial conflicts of interest were reported.
An outbreak of imported wild-type poliovirus in China was interrupted within 6 weeks of laboratory confirmation of the index case, thanks to an immediate, potent public health response that accorded with World Health Organization recommendations, according to a report published online Nov. 21 in the New England Journal of Medicine.
The last indigenous wild-type poliovirus in China was isolated in 1994, and the country was certified as poliomyelitis free in 2000. This strain of the virus was traced back to Pakistan, which, along with Afghanistan and Nigeria, are the only countries in the world that have failed to control transmission of wild-type poliovirus, said Dr. Hui-Ming Luo of the Chinese Center for Disease Control and Prevention, Beijing, and associates.
"Until wild-type poliovirus transmission is interrupted globally, poliomyelitis-free countries will continue to be at risk for viral importation," they wrote.
In this outbreak, the index case was a 16-month-old girl whose family had no history of travel beyond southern Xinjiang province. She was hospitalized for flaccid paralysis, and an epidemiologic investigation began immediately. Three more cases of flaccid paralysis were identified in the same county, and the Chinese CDC confirmed that the same strain of wild-type poliovirus was present in all cases and that it had been imported from Pakistan.
All together, 21 cases of infection were confirmed and 23 clinically compatible cases were reported in southern Xinjiang. A single sewage sample from the index case’s hometown and another from a sample of river water there were found to be positive for wild-type poliovirus type 1.
"The incidence rates of wild-type poliovirus infection and clinically compatible poliomyelitis were highest among children younger than 1 year of age (3.46/100,000 and 1.15 cases/100,000 population, respectively)", they reported. Just under half of the 21 infections were in children under age 14 years.
The virus was isolated from 14 of 673 (2.1%) healthy people who were in contact with the affected patients and from 13 of 491 (2.6%) healthy people who were not in contact with any patients.
The national and local governments responded according to the 2006 World Health Assembly resolution on such outbreaks: they launched an investigation within 72 hours; conducted five rounds of supplementary immunization, beginning within 4 weeks of the start of the outbreak and continuing after the last cases were reported; surveyed and found that the coverage rates for all five rounds were greater than 95%; and enhanced the surveillance for acute flaccid paralysis throughout the affected province.
"Key response measures were the declaration of a public health emergency, enhancement of the sensitivity of surveillance for acute flaccid paralysis, the conduct of epidemiologically targeted vaccination campaigns, the use of monovalent OPV1 [oral poliovirus vaccine] for two rounds of supplementary immunization, close collaboration with international agencies such as the WHO and the U.S. Centers for Disease Control and Prevention, and provision of adequate resources to mount the response," the investigators said (N. Engl. J. Med. 2013 Nov. 21 [doi:10.1056/NEJMoa1303368]).
This effort was estimated to have cost the equivalent of $26 million U.S. dollars and required more than 500 public health experts to assist the local health workers, government officials, and approximately 500,000 volunteers who were involved.
"The global eradication of poliomyelitis will benefit all countries, including those that have been certified as poliomyelitis-free," the researchers wrote.
No financial conflicts of interest were reported.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: Twenty-one cases of infection were confirmed and 23 clinically compatible cases were reported in southern Xinjiang; polio virus was isolated from 14 of 673 (2.1%) healthy people who were in contact with the affected patients and from 13 of 491 (2.6%) healthy people who were not in contact with any patients.
Data source: A descriptive epidemiological study of a poliomyelitis outbreak in southern China that affected 21 people and was contained within 6 weeks.
Disclosures: No financial conflicts of interest were reported.
Breast MRI both overused and underused
In real-world practice, breast MRI is being overused in women who won’t benefit and might even be harmed by it, but at the same time underused by the women who could gain the most from it, according to two separate studies reported online Nov. 18 in JAMA Internal Medicine.
The two large cohort studies, which involved different patient populations and different methodologies, had remarkably similar findings. In addition to the widespread overuse and underuse of breast MRI, both studies found that overall use of the technology has soared since the year 2000 for a variety of indications, even though the evidence only supports its use for a few particular indications.
Thankfully, that rapid increase appears to have plateaued in the most recent years for which data are available, both research groups noted.
Both studies were performed simply to gather data on national patterns of breast MRI use in community practice, because of the dearth of information on this topic.
National guidelines support breast MRI to screen asymptomatic women only if they are at high risk for breast cancer because they carry BRCA gene mutations, are first-degree relatives of BRCA carriers but haven’t been tested themselves, or are at more than 20% lifetime risk of breast cancer according to risk assessment tools based on family history.
In the first study, investigators assessed breast MRI use using data from five regional registries participating in the Breast Cancer Surveillance Consortium. The study population comprised women aged 18-79 years who had breast MRI (8,931 exams in 6,777 subjects) and/or screening mammography (1,288,924 subjects) during 2005-2009, reported Karen J. Wernli, Ph.D., of Group Health Research Institute, Seattle, and her associates.
During the relatively brief study period, the overall use of breast MRI nearly tripled, from 4.2 to 11.5 exams per 1,000 women. The total number of exams rose steeply during the first 2 years, from 863 in 2005 to 2,264 in 2007; it then remained stable at about 2,150 per year through 2009.
Overall, only 25% of women who had screening breast MRI were considered at high lifetime risk for breast cancer and thus fit the recommended criteria for the procedure. That proportion was only 9% in 2005, and it rose to 29% in 2009.
It appears that most of these women and their clinicians overestimated their breast cancer risk. They may obtain more accurate assessments by using approved risk calculators rather than by relying on family history alone, Dr. Wernli and her colleagues said (JAMA Intern. Med. 2013 Nov. 18 [doi: 10.1001/jamainternmed.2013.11963]).
Conversely, fewer than 5% of the approximately 25,200 study participants deemed to be at high lifetime risk for breast cancer underwent screening breast MRI. Previous studies have reported that the most common reasons that high-risk women cite for forgoing breast MRI were claustrophobia, time constraints, financial concerns, a physician who didn’t endorse the procedure, lack of patient interest, and lack of access to the technology.
In the second study, investigators assessed the use of breast MRI from 2000 through 2011 in a large, not-for-profit health plan covering more than 1 million patients throughout New England. The study population involved 10,518 women aged 20-89 years (mean age, 49 years) who had 18,215 breast MRI exams, reported Natasha K. Stout, Ph.D., of the department of population medicine, Harvard Medical School and Harvard Pilgrim Health Care Institute, Boston, and her associates.
Overall use of the procedure increased 14-fold during the study period, from 198 per 10,000 women in 2000 to 2,744 per 10,000 in 2011. As in the previous study, there was a steep rise in breast MRI use until approximately 2008, followed by a leveling off. This coincides with the release of American Cancer Society guidelines in 2007 recommending breast MRI as a screening tool only for women at high risk of breast cancer, Dr. Stout and her colleagues said.
Only 21% of the women who underwent screening breast MRI were at high risk and thus met the recommended criteria for the procedure. And fewer than half of the women who were at high risk by virtue of their BRCA status or family history underwent breast MRI (JAMA Intern. Med. 2013 Nov. 18 [doi: 10.1001/jamainternmed.2013.11958]).
"Understanding who is receiving breast MRI and the downstream consequences of this use should be a high research priority, to ensure that the limited health care funds available are used wisely to maximize population health," Dr. Stout and her colleagues said.
Dr. Wernli’s study was supported in part by the National Cancer Institute and the Agency for Healthcare Research and Quality. Dr. Wernli reported no financial conflicts of interest; her associates reported ties to GE Medical Systems, Phillips Medical Systems, and other companies. Dr. Stout’s study was supported in part by the American Cancer Society and the National Center for Research Resources. Dr. Stout and her associates reported no financial conflicts of interest.
The "striking" overuse of breast MRI in women who didn’t meet guideline criteria and simultaneous underuse in women who could derive the greatest benefit "clearly indicate the need for better patient selection," said Dr. E. Shelley Hwang and Dr. Isabelle Bedrosian.
Both studies also showed that the procedure continues to be used for nonscreening purposes – such as for staging newly diagnosed breast cancers and post-treatment surveillance – for which there is insufficient data supporting that use.
"A thoughtful, data-driven allocation of technology is necessary for clinicians and patients to make the best choices. As a medical community, we bear a collective responsibility to ensure that breast MRI provides sufficient clinical benefit to warrant the additional biopsies, increased patient anxiety, and cost that accrue with its use," they said.
Dr. Hwang is at Duke Cancer Institute, Durham, N.C. Dr. Bedrosian is at M.D. Anderson Cancer Center, Houston. They reported no potential financial conflicts of interest. These remarks were taken from their invited commentary accompanying Dr. Wernli’s and Dr. Stout’s reports (JAMA Intern. Med. 2013 Nov. 18 [doi: 10.1001/jamainternmed.2013.10502]).
The "striking" overuse of breast MRI in women who didn’t meet guideline criteria and simultaneous underuse in women who could derive the greatest benefit "clearly indicate the need for better patient selection," said Dr. E. Shelley Hwang and Dr. Isabelle Bedrosian.
Both studies also showed that the procedure continues to be used for nonscreening purposes – such as for staging newly diagnosed breast cancers and post-treatment surveillance – for which there is insufficient data supporting that use.
"A thoughtful, data-driven allocation of technology is necessary for clinicians and patients to make the best choices. As a medical community, we bear a collective responsibility to ensure that breast MRI provides sufficient clinical benefit to warrant the additional biopsies, increased patient anxiety, and cost that accrue with its use," they said.
Dr. Hwang is at Duke Cancer Institute, Durham, N.C. Dr. Bedrosian is at M.D. Anderson Cancer Center, Houston. They reported no potential financial conflicts of interest. These remarks were taken from their invited commentary accompanying Dr. Wernli’s and Dr. Stout’s reports (JAMA Intern. Med. 2013 Nov. 18 [doi: 10.1001/jamainternmed.2013.10502]).
The "striking" overuse of breast MRI in women who didn’t meet guideline criteria and simultaneous underuse in women who could derive the greatest benefit "clearly indicate the need for better patient selection," said Dr. E. Shelley Hwang and Dr. Isabelle Bedrosian.
Both studies also showed that the procedure continues to be used for nonscreening purposes – such as for staging newly diagnosed breast cancers and post-treatment surveillance – for which there is insufficient data supporting that use.
"A thoughtful, data-driven allocation of technology is necessary for clinicians and patients to make the best choices. As a medical community, we bear a collective responsibility to ensure that breast MRI provides sufficient clinical benefit to warrant the additional biopsies, increased patient anxiety, and cost that accrue with its use," they said.
Dr. Hwang is at Duke Cancer Institute, Durham, N.C. Dr. Bedrosian is at M.D. Anderson Cancer Center, Houston. They reported no potential financial conflicts of interest. These remarks were taken from their invited commentary accompanying Dr. Wernli’s and Dr. Stout’s reports (JAMA Intern. Med. 2013 Nov. 18 [doi: 10.1001/jamainternmed.2013.10502]).
In real-world practice, breast MRI is being overused in women who won’t benefit and might even be harmed by it, but at the same time underused by the women who could gain the most from it, according to two separate studies reported online Nov. 18 in JAMA Internal Medicine.
The two large cohort studies, which involved different patient populations and different methodologies, had remarkably similar findings. In addition to the widespread overuse and underuse of breast MRI, both studies found that overall use of the technology has soared since the year 2000 for a variety of indications, even though the evidence only supports its use for a few particular indications.
Thankfully, that rapid increase appears to have plateaued in the most recent years for which data are available, both research groups noted.
Both studies were performed simply to gather data on national patterns of breast MRI use in community practice, because of the dearth of information on this topic.
National guidelines support breast MRI to screen asymptomatic women only if they are at high risk for breast cancer because they carry BRCA gene mutations, are first-degree relatives of BRCA carriers but haven’t been tested themselves, or are at more than 20% lifetime risk of breast cancer according to risk assessment tools based on family history.
In the first study, investigators assessed breast MRI use using data from five regional registries participating in the Breast Cancer Surveillance Consortium. The study population comprised women aged 18-79 years who had breast MRI (8,931 exams in 6,777 subjects) and/or screening mammography (1,288,924 subjects) during 2005-2009, reported Karen J. Wernli, Ph.D., of Group Health Research Institute, Seattle, and her associates.
During the relatively brief study period, the overall use of breast MRI nearly tripled, from 4.2 to 11.5 exams per 1,000 women. The total number of exams rose steeply during the first 2 years, from 863 in 2005 to 2,264 in 2007; it then remained stable at about 2,150 per year through 2009.
Overall, only 25% of women who had screening breast MRI were considered at high lifetime risk for breast cancer and thus fit the recommended criteria for the procedure. That proportion was only 9% in 2005, and it rose to 29% in 2009.
It appears that most of these women and their clinicians overestimated their breast cancer risk. They may obtain more accurate assessments by using approved risk calculators rather than by relying on family history alone, Dr. Wernli and her colleagues said (JAMA Intern. Med. 2013 Nov. 18 [doi: 10.1001/jamainternmed.2013.11963]).
Conversely, fewer than 5% of the approximately 25,200 study participants deemed to be at high lifetime risk for breast cancer underwent screening breast MRI. Previous studies have reported that the most common reasons that high-risk women cite for forgoing breast MRI were claustrophobia, time constraints, financial concerns, a physician who didn’t endorse the procedure, lack of patient interest, and lack of access to the technology.
In the second study, investigators assessed the use of breast MRI from 2000 through 2011 in a large, not-for-profit health plan covering more than 1 million patients throughout New England. The study population involved 10,518 women aged 20-89 years (mean age, 49 years) who had 18,215 breast MRI exams, reported Natasha K. Stout, Ph.D., of the department of population medicine, Harvard Medical School and Harvard Pilgrim Health Care Institute, Boston, and her associates.
Overall use of the procedure increased 14-fold during the study period, from 198 per 10,000 women in 2000 to 2,744 per 10,000 in 2011. As in the previous study, there was a steep rise in breast MRI use until approximately 2008, followed by a leveling off. This coincides with the release of American Cancer Society guidelines in 2007 recommending breast MRI as a screening tool only for women at high risk of breast cancer, Dr. Stout and her colleagues said.
Only 21% of the women who underwent screening breast MRI were at high risk and thus met the recommended criteria for the procedure. And fewer than half of the women who were at high risk by virtue of their BRCA status or family history underwent breast MRI (JAMA Intern. Med. 2013 Nov. 18 [doi: 10.1001/jamainternmed.2013.11958]).
"Understanding who is receiving breast MRI and the downstream consequences of this use should be a high research priority, to ensure that the limited health care funds available are used wisely to maximize population health," Dr. Stout and her colleagues said.
Dr. Wernli’s study was supported in part by the National Cancer Institute and the Agency for Healthcare Research and Quality. Dr. Wernli reported no financial conflicts of interest; her associates reported ties to GE Medical Systems, Phillips Medical Systems, and other companies. Dr. Stout’s study was supported in part by the American Cancer Society and the National Center for Research Resources. Dr. Stout and her associates reported no financial conflicts of interest.
In real-world practice, breast MRI is being overused in women who won’t benefit and might even be harmed by it, but at the same time underused by the women who could gain the most from it, according to two separate studies reported online Nov. 18 in JAMA Internal Medicine.
The two large cohort studies, which involved different patient populations and different methodologies, had remarkably similar findings. In addition to the widespread overuse and underuse of breast MRI, both studies found that overall use of the technology has soared since the year 2000 for a variety of indications, even though the evidence only supports its use for a few particular indications.
Thankfully, that rapid increase appears to have plateaued in the most recent years for which data are available, both research groups noted.
Both studies were performed simply to gather data on national patterns of breast MRI use in community practice, because of the dearth of information on this topic.
National guidelines support breast MRI to screen asymptomatic women only if they are at high risk for breast cancer because they carry BRCA gene mutations, are first-degree relatives of BRCA carriers but haven’t been tested themselves, or are at more than 20% lifetime risk of breast cancer according to risk assessment tools based on family history.
In the first study, investigators assessed breast MRI use using data from five regional registries participating in the Breast Cancer Surveillance Consortium. The study population comprised women aged 18-79 years who had breast MRI (8,931 exams in 6,777 subjects) and/or screening mammography (1,288,924 subjects) during 2005-2009, reported Karen J. Wernli, Ph.D., of Group Health Research Institute, Seattle, and her associates.
During the relatively brief study period, the overall use of breast MRI nearly tripled, from 4.2 to 11.5 exams per 1,000 women. The total number of exams rose steeply during the first 2 years, from 863 in 2005 to 2,264 in 2007; it then remained stable at about 2,150 per year through 2009.
Overall, only 25% of women who had screening breast MRI were considered at high lifetime risk for breast cancer and thus fit the recommended criteria for the procedure. That proportion was only 9% in 2005, and it rose to 29% in 2009.
It appears that most of these women and their clinicians overestimated their breast cancer risk. They may obtain more accurate assessments by using approved risk calculators rather than by relying on family history alone, Dr. Wernli and her colleagues said (JAMA Intern. Med. 2013 Nov. 18 [doi: 10.1001/jamainternmed.2013.11963]).
Conversely, fewer than 5% of the approximately 25,200 study participants deemed to be at high lifetime risk for breast cancer underwent screening breast MRI. Previous studies have reported that the most common reasons that high-risk women cite for forgoing breast MRI were claustrophobia, time constraints, financial concerns, a physician who didn’t endorse the procedure, lack of patient interest, and lack of access to the technology.
In the second study, investigators assessed the use of breast MRI from 2000 through 2011 in a large, not-for-profit health plan covering more than 1 million patients throughout New England. The study population involved 10,518 women aged 20-89 years (mean age, 49 years) who had 18,215 breast MRI exams, reported Natasha K. Stout, Ph.D., of the department of population medicine, Harvard Medical School and Harvard Pilgrim Health Care Institute, Boston, and her associates.
Overall use of the procedure increased 14-fold during the study period, from 198 per 10,000 women in 2000 to 2,744 per 10,000 in 2011. As in the previous study, there was a steep rise in breast MRI use until approximately 2008, followed by a leveling off. This coincides with the release of American Cancer Society guidelines in 2007 recommending breast MRI as a screening tool only for women at high risk of breast cancer, Dr. Stout and her colleagues said.
Only 21% of the women who underwent screening breast MRI were at high risk and thus met the recommended criteria for the procedure. And fewer than half of the women who were at high risk by virtue of their BRCA status or family history underwent breast MRI (JAMA Intern. Med. 2013 Nov. 18 [doi: 10.1001/jamainternmed.2013.11958]).
"Understanding who is receiving breast MRI and the downstream consequences of this use should be a high research priority, to ensure that the limited health care funds available are used wisely to maximize population health," Dr. Stout and her colleagues said.
Dr. Wernli’s study was supported in part by the National Cancer Institute and the Agency for Healthcare Research and Quality. Dr. Wernli reported no financial conflicts of interest; her associates reported ties to GE Medical Systems, Phillips Medical Systems, and other companies. Dr. Stout’s study was supported in part by the American Cancer Society and the National Center for Research Resources. Dr. Stout and her associates reported no financial conflicts of interest.
FROM JAMA INTERNAL MEDICINE
Major finding: Only 25% of the women in one study and 21% in the other who underwent breast MRI met the high-risk criteria recommended for the procedure.
Data source: A cohort study involving women across the country who underwent 8,931 breast MRI exams and more than 1.2 million mammograms during 2005-2009, and a separate cohort study involving 10,518 women in New England who underwent these exams during 2000-2011.
Disclosures: Dr. Wernli’s study was supported in part by the National Cancer Institute and the Agency for Healthcare Research and Quality. Dr. Wernli reported no financial conflicts of interest; her associates reported ties to GE Medical Systems, Phillips Medical Systems, and other companies. Dr. Stout’s study was supported in part by the American Cancer Society and the National Center for Research Resources. Dr. Stout and her associates reported no financial conflicts of interest.
Varespladib raises MI risk after recent acute coronary syndrome
Far from reducing the risk of cardiovascular events in patients with recent acute coronary syndrome, the sPLA2 inhibitor varespladib actually raised the risk of MI in an international randomized clinical trial.
The drug did exert beneficial effects on inflammatory and lipid biomarkers as expected, "which theoretically should have translated to a lower propensity to plaque rupture" in the 16 weeks following ACS. But the study was terminated early "for futility" after an interim analysis of the outcomes for only 212 of the 5,012 randomized patients, said Dr. Stephen J. Nicholls and his associates in the VISTA-16 clinical trial, which was reported simultaneously at the American Heart Association scientific sessions and published online in JAMA (2013 Nov. 18 [doi:10.1001/jama.2013.282836]).
Compared with placebo, varespladib caused an excess of MIs and of the composite outcome of cardiovascular mortality, MI, and stroke. "These findings suggest that short-term sPLA2 inhibition with varespladib is harmful following ACS," said Dr. Nicholls of the South Australian Health and Medical Research Institute, Adelaide, and his colleagues.
"Our findings ... reemphasize that identification of a circulating marker of cardiovascular risk does not necessarily imply that pharmacologic suppression or inhibition of the marker will reduce risk," they noted.
Varespladib is a nonspecific inhibitor of the family of secretory phospholipase A2 (sPLA2) enzymes that are involved in inflammation, some of which are present in atherosclerotic lesions and myocardial areas injured by ischemia. In initial studies, the drug reduced circulating levels of sPLA2 by more than 90%, lowered LDL cholesterol, and decreased C-reactive protein in ACS patients.
The VISTA-16 (Vascular Inflammation Suppression to Treat Acute coronary syndrome for 16 weeks) trial, sponsored by Anthera Pharmaceuticals, the maker of varespladib, was designed to assess the drug’s efficacy at preventing CV events during the high-risk period of the 4 months immediately following ACS.
A total of 5,145 patients were enrolled at 362 medical centers in 17 countries over the course of 3 years, within hours of presenting with ACS. They had a high prevalence of CV risk factors and established atherosclerotic disease, and most were already receiving appropriate therapy with antiplatelet agents, statins, beta-blockers, ACE inhibitors, or receptor blockers. Eighty percent underwent coronary revascularization after the index event.
The primary endpoint was a composite of CV mortality, nonfatal MI, nonfatal stroke, or unstable angina requiring hospitalization. When the trial was terminated, outcomes in 212 patients had been documented after a mean of 13.4 weeks of treatment. The primary endpoint occurred in 6.1% of patients who took varespladib, compared with 5.1% of those who took placebo (hazard ratio, 1.25), the investigators said.
A secondary outcome, a composite of CV mortality, MI, and stroke, occurred in 4.6% of patients who took varespladib and 3.8% of those who took placebo (HR, 1.36).
"This was due primarily to a greater incidence of MI in the varespladib group compared with the placebo group (3.4% vs 2.2%; HR, 1.66)," the researchers said.
Six months after patients had discontinued study treatment, all-cause mortality was 2.7% in the varespladib group and 2.0% in the placebo group.
Twice as many patients taking the active drug (2.8%) as taking placebo (1.4%) discontinued treatment because of adverse or serious adverse events.
The reasons for varespladib’s adverse effect on MI are not yet known. "Whether this represents an adverse effect of the varespladib molecule or a consequence of pan-sPLA2 inhibition remains to be determined," Dr. Nicholls and his associates said.
The VISTA-16 trial was funded by Anthera Pharmaceuticals. Dr. Nicholls and his associates reported numerous ties to industry sources.
Far from reducing the risk of cardiovascular events in patients with recent acute coronary syndrome, the sPLA2 inhibitor varespladib actually raised the risk of MI in an international randomized clinical trial.
The drug did exert beneficial effects on inflammatory and lipid biomarkers as expected, "which theoretically should have translated to a lower propensity to plaque rupture" in the 16 weeks following ACS. But the study was terminated early "for futility" after an interim analysis of the outcomes for only 212 of the 5,012 randomized patients, said Dr. Stephen J. Nicholls and his associates in the VISTA-16 clinical trial, which was reported simultaneously at the American Heart Association scientific sessions and published online in JAMA (2013 Nov. 18 [doi:10.1001/jama.2013.282836]).
Compared with placebo, varespladib caused an excess of MIs and of the composite outcome of cardiovascular mortality, MI, and stroke. "These findings suggest that short-term sPLA2 inhibition with varespladib is harmful following ACS," said Dr. Nicholls of the South Australian Health and Medical Research Institute, Adelaide, and his colleagues.
"Our findings ... reemphasize that identification of a circulating marker of cardiovascular risk does not necessarily imply that pharmacologic suppression or inhibition of the marker will reduce risk," they noted.
Varespladib is a nonspecific inhibitor of the family of secretory phospholipase A2 (sPLA2) enzymes that are involved in inflammation, some of which are present in atherosclerotic lesions and myocardial areas injured by ischemia. In initial studies, the drug reduced circulating levels of sPLA2 by more than 90%, lowered LDL cholesterol, and decreased C-reactive protein in ACS patients.
The VISTA-16 (Vascular Inflammation Suppression to Treat Acute coronary syndrome for 16 weeks) trial, sponsored by Anthera Pharmaceuticals, the maker of varespladib, was designed to assess the drug’s efficacy at preventing CV events during the high-risk period of the 4 months immediately following ACS.
A total of 5,145 patients were enrolled at 362 medical centers in 17 countries over the course of 3 years, within hours of presenting with ACS. They had a high prevalence of CV risk factors and established atherosclerotic disease, and most were already receiving appropriate therapy with antiplatelet agents, statins, beta-blockers, ACE inhibitors, or receptor blockers. Eighty percent underwent coronary revascularization after the index event.
The primary endpoint was a composite of CV mortality, nonfatal MI, nonfatal stroke, or unstable angina requiring hospitalization. When the trial was terminated, outcomes in 212 patients had been documented after a mean of 13.4 weeks of treatment. The primary endpoint occurred in 6.1% of patients who took varespladib, compared with 5.1% of those who took placebo (hazard ratio, 1.25), the investigators said.
A secondary outcome, a composite of CV mortality, MI, and stroke, occurred in 4.6% of patients who took varespladib and 3.8% of those who took placebo (HR, 1.36).
"This was due primarily to a greater incidence of MI in the varespladib group compared with the placebo group (3.4% vs 2.2%; HR, 1.66)," the researchers said.
Six months after patients had discontinued study treatment, all-cause mortality was 2.7% in the varespladib group and 2.0% in the placebo group.
Twice as many patients taking the active drug (2.8%) as taking placebo (1.4%) discontinued treatment because of adverse or serious adverse events.
The reasons for varespladib’s adverse effect on MI are not yet known. "Whether this represents an adverse effect of the varespladib molecule or a consequence of pan-sPLA2 inhibition remains to be determined," Dr. Nicholls and his associates said.
The VISTA-16 trial was funded by Anthera Pharmaceuticals. Dr. Nicholls and his associates reported numerous ties to industry sources.
Far from reducing the risk of cardiovascular events in patients with recent acute coronary syndrome, the sPLA2 inhibitor varespladib actually raised the risk of MI in an international randomized clinical trial.
The drug did exert beneficial effects on inflammatory and lipid biomarkers as expected, "which theoretically should have translated to a lower propensity to plaque rupture" in the 16 weeks following ACS. But the study was terminated early "for futility" after an interim analysis of the outcomes for only 212 of the 5,012 randomized patients, said Dr. Stephen J. Nicholls and his associates in the VISTA-16 clinical trial, which was reported simultaneously at the American Heart Association scientific sessions and published online in JAMA (2013 Nov. 18 [doi:10.1001/jama.2013.282836]).
Compared with placebo, varespladib caused an excess of MIs and of the composite outcome of cardiovascular mortality, MI, and stroke. "These findings suggest that short-term sPLA2 inhibition with varespladib is harmful following ACS," said Dr. Nicholls of the South Australian Health and Medical Research Institute, Adelaide, and his colleagues.
"Our findings ... reemphasize that identification of a circulating marker of cardiovascular risk does not necessarily imply that pharmacologic suppression or inhibition of the marker will reduce risk," they noted.
Varespladib is a nonspecific inhibitor of the family of secretory phospholipase A2 (sPLA2) enzymes that are involved in inflammation, some of which are present in atherosclerotic lesions and myocardial areas injured by ischemia. In initial studies, the drug reduced circulating levels of sPLA2 by more than 90%, lowered LDL cholesterol, and decreased C-reactive protein in ACS patients.
The VISTA-16 (Vascular Inflammation Suppression to Treat Acute coronary syndrome for 16 weeks) trial, sponsored by Anthera Pharmaceuticals, the maker of varespladib, was designed to assess the drug’s efficacy at preventing CV events during the high-risk period of the 4 months immediately following ACS.
A total of 5,145 patients were enrolled at 362 medical centers in 17 countries over the course of 3 years, within hours of presenting with ACS. They had a high prevalence of CV risk factors and established atherosclerotic disease, and most were already receiving appropriate therapy with antiplatelet agents, statins, beta-blockers, ACE inhibitors, or receptor blockers. Eighty percent underwent coronary revascularization after the index event.
The primary endpoint was a composite of CV mortality, nonfatal MI, nonfatal stroke, or unstable angina requiring hospitalization. When the trial was terminated, outcomes in 212 patients had been documented after a mean of 13.4 weeks of treatment. The primary endpoint occurred in 6.1% of patients who took varespladib, compared with 5.1% of those who took placebo (hazard ratio, 1.25), the investigators said.
A secondary outcome, a composite of CV mortality, MI, and stroke, occurred in 4.6% of patients who took varespladib and 3.8% of those who took placebo (HR, 1.36).
"This was due primarily to a greater incidence of MI in the varespladib group compared with the placebo group (3.4% vs 2.2%; HR, 1.66)," the researchers said.
Six months after patients had discontinued study treatment, all-cause mortality was 2.7% in the varespladib group and 2.0% in the placebo group.
Twice as many patients taking the active drug (2.8%) as taking placebo (1.4%) discontinued treatment because of adverse or serious adverse events.
The reasons for varespladib’s adverse effect on MI are not yet known. "Whether this represents an adverse effect of the varespladib molecule or a consequence of pan-sPLA2 inhibition remains to be determined," Dr. Nicholls and his associates said.
The VISTA-16 trial was funded by Anthera Pharmaceuticals. Dr. Nicholls and his associates reported numerous ties to industry sources.
FROM THE AHA SCIENTIFIC SESSIONS
Major Finding: Patients who took varespladib had a significantly higher rate of MI (3.4%) than those who took placebo (2.2%), with a hazard ratio of 1.66.
Data Source: A double-blind, randomized clinical trial comparing varespladib against placebo in 5,012 ACS patients, which was terminated early when final outcomes were available for just 212 patients.
Disclosures: The VISTA-16 trial was funded by Anthera Pharmaceuticals. Dr. Nicholls and his associates reported numerous ties to industry sources.
Mitral valve repair no better than replacement
Mitral valve repair was no better than chordal-sparing mitral valve replacement in the first randomized clinical trial attempting to settle the controversy over which procedure is superior for treating functional ischemic mitral regurgitation, which was simultaneously reported at the annual scientific sessions of the American Heart Association and online Nov. 18 in the New England Journal of Medicine.
In the past few years, the use of mitral valve repair has far exceeded that of mitral valve replacement for this indication, largely on the basis of reports that the repair procedure yields lower operative mortality, improved left ventricular function, and higher long-term survival rates. In particular, a 2011 meta-analysis found a 35% lower relative risk of death in the long term with mitral valve repair, compared with replacement, said Dr. Michael A. Acker and his associates in the Cardiothoracic Surgical Trials Network (CTSN).
But in their multicenter study directly comparing the two procedures in 251 patients with severe functional ischemic mitral regurgitation, there was no significant difference between the surgeries in left ventricular end-systolic volume index at 1 year, nor in mortality at either 1 month or 1 year.
Moreover, study participants who underwent mitral valve repair showed a disturbing excess in the rate of recurrence of mitral regurgitation at 1 year, with a rate that was 30 percentage points higher than that among patients who underwent mitral valve replacement. "This lack of durability in correction of mitral regurgitation is disconcerting, given its reported association with further progression and long-term negative outcomes," said Dr. Acker of the division of cardiovascular surgery, University of Pennsylvania, Philadelphia, and his associates.
Functional ischemic mitral regurgitation, a "high-prevalence" condition affecting an estimated 2-3 million Americans, differs from primary degenerative mitral regurgitation in that the valve leaflets themselves remain normal while the defect occurs in the myocardium. "Ischemic mitral regurgitation is a consequence of adverse left ventricular remodeling after myocardial injury, with enlargement of the left ventricular chamber and mitral annulus, apical and lateral migration of the papillary muscles, leaflet retethering, and reduced closing forces.
"These processes lead to malcoaptation of the leaflets and variable degrees of mitral regurgitation that can fluctuate dynamically as a function of volume status, afterload, heart rhythm, and residual ischemia," the researchers said.
Current practice guidelines recommend mitral valve repair or chordal-sparing mitral valve replacement for severe regurgitation unresponsive to medical therapy, but do not specify which procedure is preferred because there is no conclusive evidence demonstrating the superiority of one over the other. "Recently, the field has embraced mitral valve repair over replacement," even without such evidence, Dr. Acker and his colleagues said.
The CTSN performed this study at 22 medical centers to assess the relative benefits of the two surgeries, with 126 patients randomized to undergo mitral valve repair and 125 to undergo replacement that included complete preservation of the subvalvular apparatus.
The primary endpoint was the degree of LV reverse remodeling, as measured by the left ventricular end-systolic volume index (LVESVI) on transthoracic echocardiography, at 1 year. The mean LVESVI was not significantly different between the repair group (54.6 mL per square meter) and the replacement group (60.7 mL per square meter), reflecting decreases of 6.6 mL per square meter and 6.8 mL per square meter, respectively, the investigators said (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1312808]).
The median between group difference in the change in LVESVI score after surgery also was not clinically significant.
However, 32.6% of patients who underwent mitral valve repair had a recurrence of regurgitation within 1 year, compared with only 2.3% of those who had mitral valve replacement. Three patients in the repair group required reoperation, compared with none in the replacement group.
There were no significant differences in cumulative mortality, 30-day postoperative mortality, or 1-year mortality between the two study groups, and no significant difference in a composite endpoint of major adverse cardiac or cerebrovascular events.
Rates of serious adverse events were similar, and the durations of hospitalization were similar between the two study groups, as were rates of readmission. All measures of quality of life and functional status on two different assessment tools also were similar.
"Our findings contradict much of the published literature on this topic, which reports several advantages to mitral valve repair over replacement, including lower operative mortality, improved left ventricular function, and higher rates of long-term survival," Dr. Acker and his associates noted.
The evolution of the valve replacement procedure, which now includes chordal sparing, "may account for the improved results we observed, as compared with previous studies, since the retention of the internal architectural support of the left ventricle may preserve contractile efficiency and reduce left ventricular dilatation and dysfunction," they said.
This study was funded by the National Heart, Lung, and Blood Institute, the National Institute of Neurological Diseases and Stroke, and the Canadian Institutes of Health Research. Dr. Acker and his associates reported no financial conflicts of interest.
Mitral valve repair was no better than chordal-sparing mitral valve replacement in the first randomized clinical trial attempting to settle the controversy over which procedure is superior for treating functional ischemic mitral regurgitation, which was simultaneously reported at the annual scientific sessions of the American Heart Association and online Nov. 18 in the New England Journal of Medicine.
In the past few years, the use of mitral valve repair has far exceeded that of mitral valve replacement for this indication, largely on the basis of reports that the repair procedure yields lower operative mortality, improved left ventricular function, and higher long-term survival rates. In particular, a 2011 meta-analysis found a 35% lower relative risk of death in the long term with mitral valve repair, compared with replacement, said Dr. Michael A. Acker and his associates in the Cardiothoracic Surgical Trials Network (CTSN).
But in their multicenter study directly comparing the two procedures in 251 patients with severe functional ischemic mitral regurgitation, there was no significant difference between the surgeries in left ventricular end-systolic volume index at 1 year, nor in mortality at either 1 month or 1 year.
Moreover, study participants who underwent mitral valve repair showed a disturbing excess in the rate of recurrence of mitral regurgitation at 1 year, with a rate that was 30 percentage points higher than that among patients who underwent mitral valve replacement. "This lack of durability in correction of mitral regurgitation is disconcerting, given its reported association with further progression and long-term negative outcomes," said Dr. Acker of the division of cardiovascular surgery, University of Pennsylvania, Philadelphia, and his associates.
Functional ischemic mitral regurgitation, a "high-prevalence" condition affecting an estimated 2-3 million Americans, differs from primary degenerative mitral regurgitation in that the valve leaflets themselves remain normal while the defect occurs in the myocardium. "Ischemic mitral regurgitation is a consequence of adverse left ventricular remodeling after myocardial injury, with enlargement of the left ventricular chamber and mitral annulus, apical and lateral migration of the papillary muscles, leaflet retethering, and reduced closing forces.
"These processes lead to malcoaptation of the leaflets and variable degrees of mitral regurgitation that can fluctuate dynamically as a function of volume status, afterload, heart rhythm, and residual ischemia," the researchers said.
Current practice guidelines recommend mitral valve repair or chordal-sparing mitral valve replacement for severe regurgitation unresponsive to medical therapy, but do not specify which procedure is preferred because there is no conclusive evidence demonstrating the superiority of one over the other. "Recently, the field has embraced mitral valve repair over replacement," even without such evidence, Dr. Acker and his colleagues said.
The CTSN performed this study at 22 medical centers to assess the relative benefits of the two surgeries, with 126 patients randomized to undergo mitral valve repair and 125 to undergo replacement that included complete preservation of the subvalvular apparatus.
The primary endpoint was the degree of LV reverse remodeling, as measured by the left ventricular end-systolic volume index (LVESVI) on transthoracic echocardiography, at 1 year. The mean LVESVI was not significantly different between the repair group (54.6 mL per square meter) and the replacement group (60.7 mL per square meter), reflecting decreases of 6.6 mL per square meter and 6.8 mL per square meter, respectively, the investigators said (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1312808]).
The median between group difference in the change in LVESVI score after surgery also was not clinically significant.
However, 32.6% of patients who underwent mitral valve repair had a recurrence of regurgitation within 1 year, compared with only 2.3% of those who had mitral valve replacement. Three patients in the repair group required reoperation, compared with none in the replacement group.
There were no significant differences in cumulative mortality, 30-day postoperative mortality, or 1-year mortality between the two study groups, and no significant difference in a composite endpoint of major adverse cardiac or cerebrovascular events.
Rates of serious adverse events were similar, and the durations of hospitalization were similar between the two study groups, as were rates of readmission. All measures of quality of life and functional status on two different assessment tools also were similar.
"Our findings contradict much of the published literature on this topic, which reports several advantages to mitral valve repair over replacement, including lower operative mortality, improved left ventricular function, and higher rates of long-term survival," Dr. Acker and his associates noted.
The evolution of the valve replacement procedure, which now includes chordal sparing, "may account for the improved results we observed, as compared with previous studies, since the retention of the internal architectural support of the left ventricle may preserve contractile efficiency and reduce left ventricular dilatation and dysfunction," they said.
This study was funded by the National Heart, Lung, and Blood Institute, the National Institute of Neurological Diseases and Stroke, and the Canadian Institutes of Health Research. Dr. Acker and his associates reported no financial conflicts of interest.
Mitral valve repair was no better than chordal-sparing mitral valve replacement in the first randomized clinical trial attempting to settle the controversy over which procedure is superior for treating functional ischemic mitral regurgitation, which was simultaneously reported at the annual scientific sessions of the American Heart Association and online Nov. 18 in the New England Journal of Medicine.
In the past few years, the use of mitral valve repair has far exceeded that of mitral valve replacement for this indication, largely on the basis of reports that the repair procedure yields lower operative mortality, improved left ventricular function, and higher long-term survival rates. In particular, a 2011 meta-analysis found a 35% lower relative risk of death in the long term with mitral valve repair, compared with replacement, said Dr. Michael A. Acker and his associates in the Cardiothoracic Surgical Trials Network (CTSN).
But in their multicenter study directly comparing the two procedures in 251 patients with severe functional ischemic mitral regurgitation, there was no significant difference between the surgeries in left ventricular end-systolic volume index at 1 year, nor in mortality at either 1 month or 1 year.
Moreover, study participants who underwent mitral valve repair showed a disturbing excess in the rate of recurrence of mitral regurgitation at 1 year, with a rate that was 30 percentage points higher than that among patients who underwent mitral valve replacement. "This lack of durability in correction of mitral regurgitation is disconcerting, given its reported association with further progression and long-term negative outcomes," said Dr. Acker of the division of cardiovascular surgery, University of Pennsylvania, Philadelphia, and his associates.
Functional ischemic mitral regurgitation, a "high-prevalence" condition affecting an estimated 2-3 million Americans, differs from primary degenerative mitral regurgitation in that the valve leaflets themselves remain normal while the defect occurs in the myocardium. "Ischemic mitral regurgitation is a consequence of adverse left ventricular remodeling after myocardial injury, with enlargement of the left ventricular chamber and mitral annulus, apical and lateral migration of the papillary muscles, leaflet retethering, and reduced closing forces.
"These processes lead to malcoaptation of the leaflets and variable degrees of mitral regurgitation that can fluctuate dynamically as a function of volume status, afterload, heart rhythm, and residual ischemia," the researchers said.
Current practice guidelines recommend mitral valve repair or chordal-sparing mitral valve replacement for severe regurgitation unresponsive to medical therapy, but do not specify which procedure is preferred because there is no conclusive evidence demonstrating the superiority of one over the other. "Recently, the field has embraced mitral valve repair over replacement," even without such evidence, Dr. Acker and his colleagues said.
The CTSN performed this study at 22 medical centers to assess the relative benefits of the two surgeries, with 126 patients randomized to undergo mitral valve repair and 125 to undergo replacement that included complete preservation of the subvalvular apparatus.
The primary endpoint was the degree of LV reverse remodeling, as measured by the left ventricular end-systolic volume index (LVESVI) on transthoracic echocardiography, at 1 year. The mean LVESVI was not significantly different between the repair group (54.6 mL per square meter) and the replacement group (60.7 mL per square meter), reflecting decreases of 6.6 mL per square meter and 6.8 mL per square meter, respectively, the investigators said (N. Engl. J. Med. 2013 [doi:10.1056/NEJMoa1312808]).
The median between group difference in the change in LVESVI score after surgery also was not clinically significant.
However, 32.6% of patients who underwent mitral valve repair had a recurrence of regurgitation within 1 year, compared with only 2.3% of those who had mitral valve replacement. Three patients in the repair group required reoperation, compared with none in the replacement group.
There were no significant differences in cumulative mortality, 30-day postoperative mortality, or 1-year mortality between the two study groups, and no significant difference in a composite endpoint of major adverse cardiac or cerebrovascular events.
Rates of serious adverse events were similar, and the durations of hospitalization were similar between the two study groups, as were rates of readmission. All measures of quality of life and functional status on two different assessment tools also were similar.
"Our findings contradict much of the published literature on this topic, which reports several advantages to mitral valve repair over replacement, including lower operative mortality, improved left ventricular function, and higher rates of long-term survival," Dr. Acker and his associates noted.
The evolution of the valve replacement procedure, which now includes chordal sparing, "may account for the improved results we observed, as compared with previous studies, since the retention of the internal architectural support of the left ventricle may preserve contractile efficiency and reduce left ventricular dilatation and dysfunction," they said.
This study was funded by the National Heart, Lung, and Blood Institute, the National Institute of Neurological Diseases and Stroke, and the Canadian Institutes of Health Research. Dr. Acker and his associates reported no financial conflicts of interest.
FROM THE AHA SCIENTIFIC SESSIONS
Major Finding: The primary end point – the degree of LV reverse remodeling, as measured by the left ventricular end-systolic volume index on transthoracic echocardiography at 1 year – was not significantly different between the repair group (54.6 mL per square meter) and the replacement group (60.7 mL per square meter).
Data Source: A randomized clinical trial involving 126 patients with severe functional ischemic mitral regurgitation who underwent mitral valve repair and 125 who underwent chordal-sparing mitral valve replacement, and who were followed for 1 year.
Disclosures: This study was funded by the National Heart, Lung, and Blood Institute, the National Institute of Neurological Diseases and Stroke, and the Canadian Institutes of Health Research. Dr. Acker and his associates reported no financial conflicts of interest.
Low-intensity chemo works for Burkitt’s lymphoma
Two low-intensity chemotherapy regimens proved effective against Burkitt’s lymphoma and markedly less toxic than existing treatments, according to a report published online Nov. 13 in the New England Journal of Medicine.
In a prospective study, 30 consecutive patients with untreated Burkitt’s lymphoma were enrolled over a 10-year period in one of two low-intensity regimens; both included etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab (EPOCH-R). A complete remission was achieved in every patient and was sustained for a median of 73-86 months of follow-up, said Dr. Kieron M. Dunleavy of the National Cancer Institute and his associates.
"Low-intensity EPOCH-R–based therapy appears to obviate the need for high-intensity treatment and markedly reduces treatment toxicity while achieving high rates of durable response. Two confirmatory trials [of these regimens] are [now] under way in adults (NCT01092182) and children (NCT01760226)," they noted.
Until now, high-dose methotrexate and cytarabine were considered essential for control of this rare and typically aggressive cancer. "It is accepted that the high growth fraction and short doubling time of Burkitt’s lymphoma make intensive short-course chemotherapy a therapeutic necessity," albeit one that causes severe adverse effects and long-term morbidity. These study findings demonstrate that other less toxic approaches can be at least as effective, Dr. Dunleavy and his colleagues said.
The researchers undertook this study (NCT00001337 and NCT00006436) after hypothesizing that "the exquisite sensitivity of Burkitt’s lymphoma cells to genotoxic stress makes prolonged exposure time, not increased dose, the important therapeutic strategy for maximizing the killing of tumor cells." To test the idea, they studied 17 patients who had the sporadic variant of the malignancy and 13 who had the immunodeficiency-associated variant. Patients were aged 15-88 years, with a median age of 33.
Ten percent of the study population had high-risk disease, 17% had low-risk disease, and 73% had intermediate-risk disease.
The 19 patients who were HIV negative received a standard dose-adjusted EPOCH-R regimen. The 11 HIV-positive patients received a short-course regimen to reduce toxicity even further and got a double-dose of rituximab. The median cumulative dose of doxorubicin-etoposide was 47% lower and that of cyclophosphamide was 57% lower in the latter group.
All patients also were given filgrastim, beginning after the final dose of chemotherapy and continuing until absolute neutrophil recovery.
The HIV-negative group was followed for a median of 86 months and the HIV-positive group for a median of 73 months. At median follow-up, the HIV-negative group had a 95% rate of freedom from disease progression and an overall survival of 100%; the HIV-positive group had a 100% rate of freedom from disease progression and an overall survival of 90%.
Even though the latter group had more advanced Burkitt’s lymphoma as well as immunodeficiency, "they all had complete remissions that have been sustained without additional therapy," Dr. Dunleavy and his associates wrote (N. Engl. J. Med. 2013 Nov. 13 [doi:10.1056/NEJMoa1308392]).
At the most recent follow-up, none of the patients in either group had a disease recurrence or had died from Burkitt’s lymphoma. One HIV-positive patient died of acute myeloid leukemia nearly 3 years after completing the EPOCH-R regimen; it is not yet known whether the chemotherapy may have contributed to the later development of the leukemia, but HIV is a known risk factor for AML, the researchers noted.
There were no treatment-related deaths, and the toxic effects of both low-dose regimens were generally mild. Thrombocytopenia occurred in only 2% of the chemotherapy cycles; fever and neutropenia occurred in 22%. Almost all the chemotherapy infusions were outpatient, with hospital admission from fever and neutropenia occurring in 10% of cycles and only in HIV-positive patients.
This study was funded by the National Cancer Institute. Amgen provides filgrastim to NCI but had no other role in the study. No financial conflicts of interest were reported.
Two low-intensity chemotherapy regimens proved effective against Burkitt’s lymphoma and markedly less toxic than existing treatments, according to a report published online Nov. 13 in the New England Journal of Medicine.
In a prospective study, 30 consecutive patients with untreated Burkitt’s lymphoma were enrolled over a 10-year period in one of two low-intensity regimens; both included etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab (EPOCH-R). A complete remission was achieved in every patient and was sustained for a median of 73-86 months of follow-up, said Dr. Kieron M. Dunleavy of the National Cancer Institute and his associates.
"Low-intensity EPOCH-R–based therapy appears to obviate the need for high-intensity treatment and markedly reduces treatment toxicity while achieving high rates of durable response. Two confirmatory trials [of these regimens] are [now] under way in adults (NCT01092182) and children (NCT01760226)," they noted.
Until now, high-dose methotrexate and cytarabine were considered essential for control of this rare and typically aggressive cancer. "It is accepted that the high growth fraction and short doubling time of Burkitt’s lymphoma make intensive short-course chemotherapy a therapeutic necessity," albeit one that causes severe adverse effects and long-term morbidity. These study findings demonstrate that other less toxic approaches can be at least as effective, Dr. Dunleavy and his colleagues said.
The researchers undertook this study (NCT00001337 and NCT00006436) after hypothesizing that "the exquisite sensitivity of Burkitt’s lymphoma cells to genotoxic stress makes prolonged exposure time, not increased dose, the important therapeutic strategy for maximizing the killing of tumor cells." To test the idea, they studied 17 patients who had the sporadic variant of the malignancy and 13 who had the immunodeficiency-associated variant. Patients were aged 15-88 years, with a median age of 33.
Ten percent of the study population had high-risk disease, 17% had low-risk disease, and 73% had intermediate-risk disease.
The 19 patients who were HIV negative received a standard dose-adjusted EPOCH-R regimen. The 11 HIV-positive patients received a short-course regimen to reduce toxicity even further and got a double-dose of rituximab. The median cumulative dose of doxorubicin-etoposide was 47% lower and that of cyclophosphamide was 57% lower in the latter group.
All patients also were given filgrastim, beginning after the final dose of chemotherapy and continuing until absolute neutrophil recovery.
The HIV-negative group was followed for a median of 86 months and the HIV-positive group for a median of 73 months. At median follow-up, the HIV-negative group had a 95% rate of freedom from disease progression and an overall survival of 100%; the HIV-positive group had a 100% rate of freedom from disease progression and an overall survival of 90%.
Even though the latter group had more advanced Burkitt’s lymphoma as well as immunodeficiency, "they all had complete remissions that have been sustained without additional therapy," Dr. Dunleavy and his associates wrote (N. Engl. J. Med. 2013 Nov. 13 [doi:10.1056/NEJMoa1308392]).
At the most recent follow-up, none of the patients in either group had a disease recurrence or had died from Burkitt’s lymphoma. One HIV-positive patient died of acute myeloid leukemia nearly 3 years after completing the EPOCH-R regimen; it is not yet known whether the chemotherapy may have contributed to the later development of the leukemia, but HIV is a known risk factor for AML, the researchers noted.
There were no treatment-related deaths, and the toxic effects of both low-dose regimens were generally mild. Thrombocytopenia occurred in only 2% of the chemotherapy cycles; fever and neutropenia occurred in 22%. Almost all the chemotherapy infusions were outpatient, with hospital admission from fever and neutropenia occurring in 10% of cycles and only in HIV-positive patients.
This study was funded by the National Cancer Institute. Amgen provides filgrastim to NCI but had no other role in the study. No financial conflicts of interest were reported.
Two low-intensity chemotherapy regimens proved effective against Burkitt’s lymphoma and markedly less toxic than existing treatments, according to a report published online Nov. 13 in the New England Journal of Medicine.
In a prospective study, 30 consecutive patients with untreated Burkitt’s lymphoma were enrolled over a 10-year period in one of two low-intensity regimens; both included etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab (EPOCH-R). A complete remission was achieved in every patient and was sustained for a median of 73-86 months of follow-up, said Dr. Kieron M. Dunleavy of the National Cancer Institute and his associates.
"Low-intensity EPOCH-R–based therapy appears to obviate the need for high-intensity treatment and markedly reduces treatment toxicity while achieving high rates of durable response. Two confirmatory trials [of these regimens] are [now] under way in adults (NCT01092182) and children (NCT01760226)," they noted.
Until now, high-dose methotrexate and cytarabine were considered essential for control of this rare and typically aggressive cancer. "It is accepted that the high growth fraction and short doubling time of Burkitt’s lymphoma make intensive short-course chemotherapy a therapeutic necessity," albeit one that causes severe adverse effects and long-term morbidity. These study findings demonstrate that other less toxic approaches can be at least as effective, Dr. Dunleavy and his colleagues said.
The researchers undertook this study (NCT00001337 and NCT00006436) after hypothesizing that "the exquisite sensitivity of Burkitt’s lymphoma cells to genotoxic stress makes prolonged exposure time, not increased dose, the important therapeutic strategy for maximizing the killing of tumor cells." To test the idea, they studied 17 patients who had the sporadic variant of the malignancy and 13 who had the immunodeficiency-associated variant. Patients were aged 15-88 years, with a median age of 33.
Ten percent of the study population had high-risk disease, 17% had low-risk disease, and 73% had intermediate-risk disease.
The 19 patients who were HIV negative received a standard dose-adjusted EPOCH-R regimen. The 11 HIV-positive patients received a short-course regimen to reduce toxicity even further and got a double-dose of rituximab. The median cumulative dose of doxorubicin-etoposide was 47% lower and that of cyclophosphamide was 57% lower in the latter group.
All patients also were given filgrastim, beginning after the final dose of chemotherapy and continuing until absolute neutrophil recovery.
The HIV-negative group was followed for a median of 86 months and the HIV-positive group for a median of 73 months. At median follow-up, the HIV-negative group had a 95% rate of freedom from disease progression and an overall survival of 100%; the HIV-positive group had a 100% rate of freedom from disease progression and an overall survival of 90%.
Even though the latter group had more advanced Burkitt’s lymphoma as well as immunodeficiency, "they all had complete remissions that have been sustained without additional therapy," Dr. Dunleavy and his associates wrote (N. Engl. J. Med. 2013 Nov. 13 [doi:10.1056/NEJMoa1308392]).
At the most recent follow-up, none of the patients in either group had a disease recurrence or had died from Burkitt’s lymphoma. One HIV-positive patient died of acute myeloid leukemia nearly 3 years after completing the EPOCH-R regimen; it is not yet known whether the chemotherapy may have contributed to the later development of the leukemia, but HIV is a known risk factor for AML, the researchers noted.
There were no treatment-related deaths, and the toxic effects of both low-dose regimens were generally mild. Thrombocytopenia occurred in only 2% of the chemotherapy cycles; fever and neutropenia occurred in 22%. Almost all the chemotherapy infusions were outpatient, with hospital admission from fever and neutropenia occurring in 10% of cycles and only in HIV-positive patients.
This study was funded by the National Cancer Institute. Amgen provides filgrastim to NCI but had no other role in the study. No financial conflicts of interest were reported.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: At median follow-ups of 86 and 73 months, respectively, the HIV-negative group had a 95% rate of freedom from disease progression and an overall survival of 100%; the HIV-positive group had a 100% rate of freedom from disease progression and an overall survival of 90%.
Data Source: A prospective study involving 30 consecutive patients who had untreated Burkitt’s lymphoma and received 2 low-intensity EPOCH-R regimens.
Disclosures: This study was funded by the National Cancer Institute. Amgen provides filgrastim to NCI but had no other role in the study. No financial conflicts of interest were reported.
CV events scuttle bardoxolone for diabetic kidney disease
Bardoxolone methyl may have reduced the risk of end-stage renal disease in patients with type 2 diabetes and stage 4 chronic kidney disease in a phase III clinical trial, but researchers stopped the study early after patients taking the drug had an increased rate of cardiovascular deaths, heart failure events, nonfatal myocardial infarction, and nonfatal stroke.
The BEACON trial had only an estimated 40% statistical power to determine bardoxolone methyl’s true effects, given its termination because of safety concerns, according to data reported at the annual Kidney Week meeting sponsored by the American Society of Nephrology.
The findings were simultaneously reported online Nov. 9 in the New England Journal of Medicine (doi:10.1056/NEJMoa1306033).
Bardoxolone methyl, the most potent known activator of a transcription factor that regulates antioxidant genes, was previously shown to raise the estimated glomerular filtration rate (eGFR) in patients with diabetes-related kidney disease. However, it also increased the incidence of albuminuria and induced unintended weight loss.
To determine whether longer-term treatment with bardoxolone methyl might translate that eGFR benefit into a slower progression to end-stage renal disease (ESRD), Dr. Dick de Zeeuw of the University of Groningen (the Netherlands) and his associates performed the double-blind BEACON trial (Bardoxolone Methyl Evaluation in Patients With Chronic Kidney Disease and Type 2 Diabetes Mellitus: The Occurrence of Renal Events).
BEACON was sponsored by Reata Pharmaceuticals and included patients from the United States, Europe, Australia, Canada, Israel, and Mexico, resulting in a diverse patient population with regard to age, race/ethnicity, and area of residence. Both diabetic retinopathy and neuropathy were common comorbidities, as was cardiovascular disease.
The 2,185 study participants had type 2 diabetes and moderate to severe chronic kidney diseases, with a baseline eGFR of 15 to less than 30 mL/min per 1.73 m2 of body surface area. They were randomly assigned to receive either once-daily bardoxolone methyl 20 mg (1,088 patients) or a matching placebo (1,097 patients), along with conventional background therapies given at the discretion of their treating physicians. Those included inhibitors of the renin-angiotensin-aldosterone system, insulin or other hypoglycemic agents, and appropriate cardiovascular medications.
The median duration of exposure was 7 months for bardoxolone methyl and 8 months for placebo, and the median follow-up for both was 9 months.
Bardoxolone significantly improved eGFR, compared with placebo, and fewer patients who took the drug progressed to ESRD. But BEACON was terminated early because of an excess of CV events among patients receiving bardoxolone methyl. That "truncated" study duration limited the trial’s statistical power.
The primary endpoint was a composite of progression to ESRD or cardiovascular death, and it occurred in 6% of both study groups. However, deaths from CV causes were significantly more frequent in the active treatment group (27 patients) than in the placebo group (19 patients), with a hazard ratio of 1.44, Dr. de Zeeuw reported.
In particular, 96 patients in the bardoxolone group had heart failure (HF) events, compared with only 55 patients in the placebo group. Moreover, significantly more of the HF events in the active treatment group were severe enough to require hospitalization or cause death.
Similarly, significantly more patients taking bardoxolone methyl had a composite outcome of nonfatal myocardial infarction, nonfatal stroke, HF hospitalization, or CV death. And the number of deaths from any cause was greater – though not significantly – with bardoxolone methyl (44 deaths) than with placebo (31 deaths) (HR, 1.47; P = .10).
Compared with placebo, bardoxolone methyl also raised blood pressure and heart rate, increased levels of B-type natriuretic peptide, raised the rate of albuminuria, and caused substantial unintended weight loss. The investigators were unable to determine whether there was a loss of body fat, intracellular (that is, skeletal muscle) water, or extracellular (interstitial) water. There was a concomitant fall in serum albumin and hemoglobin levels, which may reflect hemodilution caused by fluid retention, the investigators found.
The increases in blood pressure and heart rate "constitute a potentially potent combination of factors that are likely to precipitate HF in an at-risk population." and the increase in B-type natriuretic peptide "is consistent with an increase in left ventricular wall stress," Dr. de Zeeuw said.
The investigators attempted to identify patient characteristics that might be associated with the development of HF in the bardoxolone methyl recipients, but were unable to do so.
Reata Pharmaceuticals funded the BEACON trial. Dr. de Zeeuw reported ties to AbbVie, Astellas, Chemocentryx, Johnson & Johnson, and Reata, and his associates reported ties to numerous industry sources.
*This article was updated November 11, 2013.
The adverse events linked to bardoxolone methyl in this study included excess HF and cardiovascular events, as well as increased rates of high blood pressure, high heart rate, albuminuria, GI symptoms, and muscle-related symptoms, said Dr. Jonathan Himmelfarb and Dr. Katherine R. Tuttle.
"The authors speculate that fluid retention, increased afterload, and higher heart rate contributed to heart failure," but it also is possible that bardoxolone methyl may exert direct toxic effects on the heart, Dr. Himmelfarb and Dr. Tuttle said.
In any case, "caution should be exercised whenever any drug for diabetic kidney disease increases, rather than decreases, albuminuria," they noted.
Dr. Himmelfarb and Dr. Tuttle are at the Kidney Research Institute and the division of nephrology at the University of Washington, Seattle. Dr. Tuttle also is at Providence Sacred Heart Medical Center and Children’s Hospital, Spokane. Dr. Himmelfarb reported ties to Abbott Laboratories, and Dr. Tuttle reported ties to Eli Lilly. These remarks were taken from their editorial accompanying Dr. de Zeeuw’s report (N. Engl. J. Med. 2013 Nov. 9 [doi:10.1056/NEJMe1313104]).
The adverse events linked to bardoxolone methyl in this study included excess HF and cardiovascular events, as well as increased rates of high blood pressure, high heart rate, albuminuria, GI symptoms, and muscle-related symptoms, said Dr. Jonathan Himmelfarb and Dr. Katherine R. Tuttle.
"The authors speculate that fluid retention, increased afterload, and higher heart rate contributed to heart failure," but it also is possible that bardoxolone methyl may exert direct toxic effects on the heart, Dr. Himmelfarb and Dr. Tuttle said.
In any case, "caution should be exercised whenever any drug for diabetic kidney disease increases, rather than decreases, albuminuria," they noted.
Dr. Himmelfarb and Dr. Tuttle are at the Kidney Research Institute and the division of nephrology at the University of Washington, Seattle. Dr. Tuttle also is at Providence Sacred Heart Medical Center and Children’s Hospital, Spokane. Dr. Himmelfarb reported ties to Abbott Laboratories, and Dr. Tuttle reported ties to Eli Lilly. These remarks were taken from their editorial accompanying Dr. de Zeeuw’s report (N. Engl. J. Med. 2013 Nov. 9 [doi:10.1056/NEJMe1313104]).
The adverse events linked to bardoxolone methyl in this study included excess HF and cardiovascular events, as well as increased rates of high blood pressure, high heart rate, albuminuria, GI symptoms, and muscle-related symptoms, said Dr. Jonathan Himmelfarb and Dr. Katherine R. Tuttle.
"The authors speculate that fluid retention, increased afterload, and higher heart rate contributed to heart failure," but it also is possible that bardoxolone methyl may exert direct toxic effects on the heart, Dr. Himmelfarb and Dr. Tuttle said.
In any case, "caution should be exercised whenever any drug for diabetic kidney disease increases, rather than decreases, albuminuria," they noted.
Dr. Himmelfarb and Dr. Tuttle are at the Kidney Research Institute and the division of nephrology at the University of Washington, Seattle. Dr. Tuttle also is at Providence Sacred Heart Medical Center and Children’s Hospital, Spokane. Dr. Himmelfarb reported ties to Abbott Laboratories, and Dr. Tuttle reported ties to Eli Lilly. These remarks were taken from their editorial accompanying Dr. de Zeeuw’s report (N. Engl. J. Med. 2013 Nov. 9 [doi:10.1056/NEJMe1313104]).
Bardoxolone methyl may have reduced the risk of end-stage renal disease in patients with type 2 diabetes and stage 4 chronic kidney disease in a phase III clinical trial, but researchers stopped the study early after patients taking the drug had an increased rate of cardiovascular deaths, heart failure events, nonfatal myocardial infarction, and nonfatal stroke.
The BEACON trial had only an estimated 40% statistical power to determine bardoxolone methyl’s true effects, given its termination because of safety concerns, according to data reported at the annual Kidney Week meeting sponsored by the American Society of Nephrology.
The findings were simultaneously reported online Nov. 9 in the New England Journal of Medicine (doi:10.1056/NEJMoa1306033).
Bardoxolone methyl, the most potent known activator of a transcription factor that regulates antioxidant genes, was previously shown to raise the estimated glomerular filtration rate (eGFR) in patients with diabetes-related kidney disease. However, it also increased the incidence of albuminuria and induced unintended weight loss.
To determine whether longer-term treatment with bardoxolone methyl might translate that eGFR benefit into a slower progression to end-stage renal disease (ESRD), Dr. Dick de Zeeuw of the University of Groningen (the Netherlands) and his associates performed the double-blind BEACON trial (Bardoxolone Methyl Evaluation in Patients With Chronic Kidney Disease and Type 2 Diabetes Mellitus: The Occurrence of Renal Events).
BEACON was sponsored by Reata Pharmaceuticals and included patients from the United States, Europe, Australia, Canada, Israel, and Mexico, resulting in a diverse patient population with regard to age, race/ethnicity, and area of residence. Both diabetic retinopathy and neuropathy were common comorbidities, as was cardiovascular disease.
The 2,185 study participants had type 2 diabetes and moderate to severe chronic kidney diseases, with a baseline eGFR of 15 to less than 30 mL/min per 1.73 m2 of body surface area. They were randomly assigned to receive either once-daily bardoxolone methyl 20 mg (1,088 patients) or a matching placebo (1,097 patients), along with conventional background therapies given at the discretion of their treating physicians. Those included inhibitors of the renin-angiotensin-aldosterone system, insulin or other hypoglycemic agents, and appropriate cardiovascular medications.
The median duration of exposure was 7 months for bardoxolone methyl and 8 months for placebo, and the median follow-up for both was 9 months.
Bardoxolone significantly improved eGFR, compared with placebo, and fewer patients who took the drug progressed to ESRD. But BEACON was terminated early because of an excess of CV events among patients receiving bardoxolone methyl. That "truncated" study duration limited the trial’s statistical power.
The primary endpoint was a composite of progression to ESRD or cardiovascular death, and it occurred in 6% of both study groups. However, deaths from CV causes were significantly more frequent in the active treatment group (27 patients) than in the placebo group (19 patients), with a hazard ratio of 1.44, Dr. de Zeeuw reported.
In particular, 96 patients in the bardoxolone group had heart failure (HF) events, compared with only 55 patients in the placebo group. Moreover, significantly more of the HF events in the active treatment group were severe enough to require hospitalization or cause death.
Similarly, significantly more patients taking bardoxolone methyl had a composite outcome of nonfatal myocardial infarction, nonfatal stroke, HF hospitalization, or CV death. And the number of deaths from any cause was greater – though not significantly – with bardoxolone methyl (44 deaths) than with placebo (31 deaths) (HR, 1.47; P = .10).
Compared with placebo, bardoxolone methyl also raised blood pressure and heart rate, increased levels of B-type natriuretic peptide, raised the rate of albuminuria, and caused substantial unintended weight loss. The investigators were unable to determine whether there was a loss of body fat, intracellular (that is, skeletal muscle) water, or extracellular (interstitial) water. There was a concomitant fall in serum albumin and hemoglobin levels, which may reflect hemodilution caused by fluid retention, the investigators found.
The increases in blood pressure and heart rate "constitute a potentially potent combination of factors that are likely to precipitate HF in an at-risk population." and the increase in B-type natriuretic peptide "is consistent with an increase in left ventricular wall stress," Dr. de Zeeuw said.
The investigators attempted to identify patient characteristics that might be associated with the development of HF in the bardoxolone methyl recipients, but were unable to do so.
Reata Pharmaceuticals funded the BEACON trial. Dr. de Zeeuw reported ties to AbbVie, Astellas, Chemocentryx, Johnson & Johnson, and Reata, and his associates reported ties to numerous industry sources.
*This article was updated November 11, 2013.
Bardoxolone methyl may have reduced the risk of end-stage renal disease in patients with type 2 diabetes and stage 4 chronic kidney disease in a phase III clinical trial, but researchers stopped the study early after patients taking the drug had an increased rate of cardiovascular deaths, heart failure events, nonfatal myocardial infarction, and nonfatal stroke.
The BEACON trial had only an estimated 40% statistical power to determine bardoxolone methyl’s true effects, given its termination because of safety concerns, according to data reported at the annual Kidney Week meeting sponsored by the American Society of Nephrology.
The findings were simultaneously reported online Nov. 9 in the New England Journal of Medicine (doi:10.1056/NEJMoa1306033).
Bardoxolone methyl, the most potent known activator of a transcription factor that regulates antioxidant genes, was previously shown to raise the estimated glomerular filtration rate (eGFR) in patients with diabetes-related kidney disease. However, it also increased the incidence of albuminuria and induced unintended weight loss.
To determine whether longer-term treatment with bardoxolone methyl might translate that eGFR benefit into a slower progression to end-stage renal disease (ESRD), Dr. Dick de Zeeuw of the University of Groningen (the Netherlands) and his associates performed the double-blind BEACON trial (Bardoxolone Methyl Evaluation in Patients With Chronic Kidney Disease and Type 2 Diabetes Mellitus: The Occurrence of Renal Events).
BEACON was sponsored by Reata Pharmaceuticals and included patients from the United States, Europe, Australia, Canada, Israel, and Mexico, resulting in a diverse patient population with regard to age, race/ethnicity, and area of residence. Both diabetic retinopathy and neuropathy were common comorbidities, as was cardiovascular disease.
The 2,185 study participants had type 2 diabetes and moderate to severe chronic kidney diseases, with a baseline eGFR of 15 to less than 30 mL/min per 1.73 m2 of body surface area. They were randomly assigned to receive either once-daily bardoxolone methyl 20 mg (1,088 patients) or a matching placebo (1,097 patients), along with conventional background therapies given at the discretion of their treating physicians. Those included inhibitors of the renin-angiotensin-aldosterone system, insulin or other hypoglycemic agents, and appropriate cardiovascular medications.
The median duration of exposure was 7 months for bardoxolone methyl and 8 months for placebo, and the median follow-up for both was 9 months.
Bardoxolone significantly improved eGFR, compared with placebo, and fewer patients who took the drug progressed to ESRD. But BEACON was terminated early because of an excess of CV events among patients receiving bardoxolone methyl. That "truncated" study duration limited the trial’s statistical power.
The primary endpoint was a composite of progression to ESRD or cardiovascular death, and it occurred in 6% of both study groups. However, deaths from CV causes were significantly more frequent in the active treatment group (27 patients) than in the placebo group (19 patients), with a hazard ratio of 1.44, Dr. de Zeeuw reported.
In particular, 96 patients in the bardoxolone group had heart failure (HF) events, compared with only 55 patients in the placebo group. Moreover, significantly more of the HF events in the active treatment group were severe enough to require hospitalization or cause death.
Similarly, significantly more patients taking bardoxolone methyl had a composite outcome of nonfatal myocardial infarction, nonfatal stroke, HF hospitalization, or CV death. And the number of deaths from any cause was greater – though not significantly – with bardoxolone methyl (44 deaths) than with placebo (31 deaths) (HR, 1.47; P = .10).
Compared with placebo, bardoxolone methyl also raised blood pressure and heart rate, increased levels of B-type natriuretic peptide, raised the rate of albuminuria, and caused substantial unintended weight loss. The investigators were unable to determine whether there was a loss of body fat, intracellular (that is, skeletal muscle) water, or extracellular (interstitial) water. There was a concomitant fall in serum albumin and hemoglobin levels, which may reflect hemodilution caused by fluid retention, the investigators found.
The increases in blood pressure and heart rate "constitute a potentially potent combination of factors that are likely to precipitate HF in an at-risk population." and the increase in B-type natriuretic peptide "is consistent with an increase in left ventricular wall stress," Dr. de Zeeuw said.
The investigators attempted to identify patient characteristics that might be associated with the development of HF in the bardoxolone methyl recipients, but were unable to do so.
Reata Pharmaceuticals funded the BEACON trial. Dr. de Zeeuw reported ties to AbbVie, Astellas, Chemocentryx, Johnson & Johnson, and Reata, and his associates reported ties to numerous industry sources.
*This article was updated November 11, 2013.
FROM KIDNEY WEEK
Major Finding: Bardoxolone methyl improved eGFR and may have delayed progression to ESRD, but that result was inconclusive because the trial was terminated early as a result of an excess of cardiovascular deaths, heart failure events, nonfatal myocardial infarction, and nonfatal stroke in patients given the drug.
Data Source: An international phase III double-blind trial involving 2,185 patients with type 2 diabetes and stage 4 chronic kidney disease who were randomly assigned to receive daily oral bardoxolone methyl or placebo and were followed for a median of 9 months.
Disclosures: Reata Pharmaceuticals funded the BEACON trial. Dr. de Zeeuw reported ties to AbbVie, Astellas, Chemocentryx, Johnson & Johnson, and Reata, and his associates reported ties to numerous industry sources.
Flu Vaccine Linked to Lower Cardiovascular Risk
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
Use of the flu vaccine was consistently associated with a lower risk of adverse cardiovascular events in a meta-analysis of the worldwide medical literature, according to a report published online Oct. 23 in JAMA.
The risk reduction was greatest among people at highest cardiovascular risk, said Dr. Jacob A. Udell of Women’s College Hospital, University of Toronto, and his associates.
"Our findings provide some support for current guideline recommendations for influenza vaccination of patients with acute coronary syndromes," they noted.
In particular, the finding that a simple, once-yearly injection may prevent scores of cardiovascular deaths, hospitalizations, MIs, strokes, and cases of heart failure, urgent coronary revascularization, and unstable angina also is of "considerable clinical and health policy importance, given the profound underuse of vaccination among the general public and the potential impact this preventive strategy may have on high-risk patients," the investigators added.
Several observational studies have suggested an association between acute respiratory infection and increased risk of cardiac and cerebrovascular events, and some have suggested that such infection may trigger rupturing of atherosclerotic plaque, fluid-overload heart failure, myocarditis, or arrhythmia. But whether or not influenza immunization could actually prevent such events remains controversial.
Dr. Udell and his colleagues performed a systematic review of the literature from the inception of database entry (1946) to the present, identifying 71 potentially relevant studies to review. They then performed a meta-analysis of the 12 randomized clinical trials in which influenza vaccination was compared against either placebo or standard care, and in which cardiovascular outcomes during the year following vaccination were reported.
Five of the 12 randomized clinical trials were considered to be of high quality and the remainder were of low or uncertain quality. The meta-analysis included 6,469 participants (mean age, 67 years) who had varying degrees of cardiovascular risk.
"Despite differences in trial designs, risk of bias, sample size, cardiovascular risk of participants, circulating influenza activity, vaccination strategy, duration of follow-up, and number of observed events, our meta-analysis demonstrated a consistent association between influenza vaccination and a lower risk of CV events," the investigators said.
The overall rate of the primary end point, a composite of all major adverse cardiovascular events, was 2.9% among recipients of the influenza vaccine (95 of 3,238 patients). This was significantly lower than the 4.7% rate (151 of 3,231 patients) among control subjects.
The number needed to treat to prevent a single major adverse CV event was 58, Dr. Udell and his associates said (JAMA 2013 Oct. 23 [doi:10.1001/jama.2013.279206]).
In a subgroup analysis involving patients known to have coronary artery disease, influenza vaccination was even more protective. For example, the rate of major adverse CV events was 10.25% among vaccinated patients with a history of recent acute coronary syndrome, compared with a rate of 23.1% among control subjects. The number needed to treat to prevent a single CV event in this subset of patients was only 8.
Less than one-third of the general population in North America and less than half of those at high cardiovascular risk currently receive the annual flu vaccine. Therefore, the flu vaccine "could address a sizable component of residual CV risk not addressed by current therapy, and provide yearlong coverage through a simple inoculation," the researchers said.
"Future research with an adequately powered multicenter trial to confirm the efficacy of this low-cost, annual, safe, easily administered, and well-tolerated therapy to reduce CV risk beyond current therapies is warranted."
The Canadian Institutes for Health Research and the Canadian Foundation for Women’s Health supported the study. Dr. Udell reported no financial conflicts of interest; his associates reported numerous ties to industry sources.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
Use of the flu vaccine was consistently associated with a lower risk of adverse cardiovascular events in a meta-analysis of the worldwide medical literature, according to a report published online Oct. 23 in JAMA.
The risk reduction was greatest among people at highest cardiovascular risk, said Dr. Jacob A. Udell of Women’s College Hospital, University of Toronto, and his associates.
"Our findings provide some support for current guideline recommendations for influenza vaccination of patients with acute coronary syndromes," they noted.
In particular, the finding that a simple, once-yearly injection may prevent scores of cardiovascular deaths, hospitalizations, MIs, strokes, and cases of heart failure, urgent coronary revascularization, and unstable angina also is of "considerable clinical and health policy importance, given the profound underuse of vaccination among the general public and the potential impact this preventive strategy may have on high-risk patients," the investigators added.
Several observational studies have suggested an association between acute respiratory infection and increased risk of cardiac and cerebrovascular events, and some have suggested that such infection may trigger rupturing of atherosclerotic plaque, fluid-overload heart failure, myocarditis, or arrhythmia. But whether or not influenza immunization could actually prevent such events remains controversial.
Dr. Udell and his colleagues performed a systematic review of the literature from the inception of database entry (1946) to the present, identifying 71 potentially relevant studies to review. They then performed a meta-analysis of the 12 randomized clinical trials in which influenza vaccination was compared against either placebo or standard care, and in which cardiovascular outcomes during the year following vaccination were reported.
Five of the 12 randomized clinical trials were considered to be of high quality and the remainder were of low or uncertain quality. The meta-analysis included 6,469 participants (mean age, 67 years) who had varying degrees of cardiovascular risk.
"Despite differences in trial designs, risk of bias, sample size, cardiovascular risk of participants, circulating influenza activity, vaccination strategy, duration of follow-up, and number of observed events, our meta-analysis demonstrated a consistent association between influenza vaccination and a lower risk of CV events," the investigators said.
The overall rate of the primary end point, a composite of all major adverse cardiovascular events, was 2.9% among recipients of the influenza vaccine (95 of 3,238 patients). This was significantly lower than the 4.7% rate (151 of 3,231 patients) among control subjects.
The number needed to treat to prevent a single major adverse CV event was 58, Dr. Udell and his associates said (JAMA 2013 Oct. 23 [doi:10.1001/jama.2013.279206]).
In a subgroup analysis involving patients known to have coronary artery disease, influenza vaccination was even more protective. For example, the rate of major adverse CV events was 10.25% among vaccinated patients with a history of recent acute coronary syndrome, compared with a rate of 23.1% among control subjects. The number needed to treat to prevent a single CV event in this subset of patients was only 8.
Less than one-third of the general population in North America and less than half of those at high cardiovascular risk currently receive the annual flu vaccine. Therefore, the flu vaccine "could address a sizable component of residual CV risk not addressed by current therapy, and provide yearlong coverage through a simple inoculation," the researchers said.
"Future research with an adequately powered multicenter trial to confirm the efficacy of this low-cost, annual, safe, easily administered, and well-tolerated therapy to reduce CV risk beyond current therapies is warranted."
The Canadian Institutes for Health Research and the Canadian Foundation for Women’s Health supported the study. Dr. Udell reported no financial conflicts of interest; his associates reported numerous ties to industry sources.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
Use of the flu vaccine was consistently associated with a lower risk of adverse cardiovascular events in a meta-analysis of the worldwide medical literature, according to a report published online Oct. 23 in JAMA.
The risk reduction was greatest among people at highest cardiovascular risk, said Dr. Jacob A. Udell of Women’s College Hospital, University of Toronto, and his associates.
"Our findings provide some support for current guideline recommendations for influenza vaccination of patients with acute coronary syndromes," they noted.
In particular, the finding that a simple, once-yearly injection may prevent scores of cardiovascular deaths, hospitalizations, MIs, strokes, and cases of heart failure, urgent coronary revascularization, and unstable angina also is of "considerable clinical and health policy importance, given the profound underuse of vaccination among the general public and the potential impact this preventive strategy may have on high-risk patients," the investigators added.
Several observational studies have suggested an association between acute respiratory infection and increased risk of cardiac and cerebrovascular events, and some have suggested that such infection may trigger rupturing of atherosclerotic plaque, fluid-overload heart failure, myocarditis, or arrhythmia. But whether or not influenza immunization could actually prevent such events remains controversial.
Dr. Udell and his colleagues performed a systematic review of the literature from the inception of database entry (1946) to the present, identifying 71 potentially relevant studies to review. They then performed a meta-analysis of the 12 randomized clinical trials in which influenza vaccination was compared against either placebo or standard care, and in which cardiovascular outcomes during the year following vaccination were reported.
Five of the 12 randomized clinical trials were considered to be of high quality and the remainder were of low or uncertain quality. The meta-analysis included 6,469 participants (mean age, 67 years) who had varying degrees of cardiovascular risk.
"Despite differences in trial designs, risk of bias, sample size, cardiovascular risk of participants, circulating influenza activity, vaccination strategy, duration of follow-up, and number of observed events, our meta-analysis demonstrated a consistent association between influenza vaccination and a lower risk of CV events," the investigators said.
The overall rate of the primary end point, a composite of all major adverse cardiovascular events, was 2.9% among recipients of the influenza vaccine (95 of 3,238 patients). This was significantly lower than the 4.7% rate (151 of 3,231 patients) among control subjects.
The number needed to treat to prevent a single major adverse CV event was 58, Dr. Udell and his associates said (JAMA 2013 Oct. 23 [doi:10.1001/jama.2013.279206]).
In a subgroup analysis involving patients known to have coronary artery disease, influenza vaccination was even more protective. For example, the rate of major adverse CV events was 10.25% among vaccinated patients with a history of recent acute coronary syndrome, compared with a rate of 23.1% among control subjects. The number needed to treat to prevent a single CV event in this subset of patients was only 8.
Less than one-third of the general population in North America and less than half of those at high cardiovascular risk currently receive the annual flu vaccine. Therefore, the flu vaccine "could address a sizable component of residual CV risk not addressed by current therapy, and provide yearlong coverage through a simple inoculation," the researchers said.
"Future research with an adequately powered multicenter trial to confirm the efficacy of this low-cost, annual, safe, easily administered, and well-tolerated therapy to reduce CV risk beyond current therapies is warranted."
The Canadian Institutes for Health Research and the Canadian Foundation for Women’s Health supported the study. Dr. Udell reported no financial conflicts of interest; his associates reported numerous ties to industry sources.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Study shows antireflux procedures are overused in infants
Clinicians appear to be too quick to perform antireflux procedures in infants, compared with older children, according to a report published online Nov. 6 in JAMA Surgery.
In a retrospective cohort study involving 141,190 pediatric and adolescent hospitalizations for gastroesophageal reflux disease (GERD) across the country over an 8-year period, the proportional hazard ratio of undergoing antireflux surgery was markedly decreased for those aged 7 months to 4 years (0.63) or 5-17 years (0.43), compared with those aged 0-2 months or 2-6 months.
The reasons for this strong difference are not yet known for certain, but the data showed a lack of objective diagnostic studies preceding the surgery in all pediatric age groups, and most strikingly in the youngest patients. It may well be that clinicians are confusing physiologic regurgitation – which is common, benign, and self-resolving in infancy – with a more pathologic process, said Dr. Jarod McAteer of the division of pediatric general and thoracic surgery, Seattle Children’s Hospital, and his associates.
At the very least, it appears that many infants aren’t given an adequate trial of medical management, since most cases of gastroesophageal reflux in infancy will resolve with that alone within 3-6 months, they noted.
"Referral for surgical treatment of GERD is generally presumed to be a last resort after failure of medical management, with optimal candidates having undergone specific preoperative evaluations," the authors wrote. Diagnostic and treatment guidelines are well delineated for adults, but not so for children.
For example, "upper GI fluoroscopy is frequently used in the preoperative workup among children with GERD," even though it has been clearly demonstrated to be a poor predictor of pathologic reflux, the investigators said.
In what they described as the first study to examine the influence of patient age on progression to antireflux procedures, Dr. McAteer and his colleagues analyzed data from the Pediatric Health Information System database, which includes demographic and clinical information from 41 children’s hospitals that cover 85% of major metropolitan areas in the U.S.
Out of 141,190 patients aged 0-7 years who were hospitalized with gastroesophageal reflux or GERD, 64% were younger than 1 year of age, and 53% were younger than 6 months. These numbers highlight how common the diagnosis is in babies, they said.
They also "suggest that physicians may be more likely to apply the diagnosis in this patient group because of diagnostic uncertainty or because other characteristics of these hospitalized infants make it more likely that any regurgitation is perceived as pathologic and indicative of GERD."
Examples of such "other characteristics" include comorbidities such as neurodevelopmental delay, cardiopulmonary disorders, seizures, asthma, and cerebral palsy.
A total of 11,621 of the study population underwent antireflux procedures, of which 52.7% were aged 6 months or younger. Only 14% of these patients had first undergone upper GI endoscopy, 0.2% esophageal manometry, 1.3% a 24-hour esophageal pH study, 65% upper GI fluoroscopy, and 17.1% a gastric emptying study, the investigators said (JAMA Surg. 2013 Nov. 6 [doi: 10.1001/jamasurg.2013.2685]).
The study findings show that the threshold for performing antireflux procedures is lower in infants than in older children. And "despite the fact that expert guidelines urge the use of objective studies in the diagnosis of GERD and despite evidence that supports the use of objective studies before performing antireflux procedures, such a standardized evaluation is not common practice.
"A greater effort is needed to develop and disseminate best-practice standards for the diagnosis and treatment of children, especially infants, with possible GERD. We must clarify the indications for antireflux procedures," Dr. McAteer and his associates said.
No financial conflicts of interest were reported.
McAteer et al. report a retrospective study of a large population-based database in the United States to identify factors associated with antireflux surgery (i.e., Nissen fundoplication) in infants, children, and adolescents hospitalized with gastroesophageal reflux disease. Perhaps the most critical question this study raises is, should the surgery be done at all, and if so, in which patients? Published guidelines (J. Pediatr. Gastroenterol. Nutr. 2009;49:498-547) suggest that other disorders that could mimic GERD should be ruled out with as much objectivity as possible prior to surgery.
One of the striking findings was that the majority of children, particularly young infants, did not undergo evaluation with diagnostic testing. Of the little diagnostic testing performed prior to surgery, upper gastrointestinal contrast fluoroscopy (UGI) was the most frequent test used. UGI, reasonable for characterizing upper GI anatomy, has no diagnostic value for GERD.
Cases were identified using ICD-9 codes, and with documented variability between clinicians in the diagnosis of GERD (Am. J. Gastroenterol. 2009;104:1278-95; quiz 96), it is concerning that not only were a great number of hospitalized children undergoing fundoplication, but over half of the cohort was 6 months or less in age.
Even more disturbing was the apparent lack of input from a consultant pediatric gastroenterologist in the decision-making process that led 11,621 (8.2%) of the 141,190 patients to antireflux surgery. Why more than half of the study cohort (52.7%) undergoing surgery to "correct" reflux at these 41 premier U.S. children's hospitals was 6 months of age or less is critical for both clinical decision making and health care utilization implications.
Numerous studies show that more than 85% of infants less than 6 months of age with reflux outgrow their reflux with little to no intervention, and, outcome studies of antireflux surgery show complications from fundoplication ranging from 8% to 28%, including death (Aliment. Pharmacol. Ther. 2007;25:1365-72; Arch. Dis. Child. 2005;90:1047-52).
In the NASPGHAN-ESPGHAN pediatric GERD guidelines, the expert panels reported that in operated children, those with neurologic impairment have more than twice the complication rate, three times the morbidity, and four times the reoperation rate of children without neurologic impairment (Am. J. Gastroenterol. 2005;100:1844-52). These data are particularly relevant to McAteer's study cohort in which antireflux surgery was performed significantly more often in those children with comorbid diagnoses of failure to thrive, neurodevelopmental delay, cardiopulmonary anomalies, cerebral palsy, aspiration pneumonia, tracheoesophageal fistula, and diaphragmatic hernia.
Thus, McAteer et al. provide a unique opportunity to not only reevaluate current clinical practice guidelines, but also implement multicenter prospective studies, using pediatric subspecialists to establish evidence-based criteria for the selection of the appropriate pediatric cases with GERD who would benefit from undergoing antireflux surgery.
Dr. Jose Garza and Dr. Benjamin D. Gold, FACG, are both in the division of pediatric gastroenterology, hepatology, and nutrition at the Children's Center for Digestive Healthcare, Atlanta. They had no relevant financial conflicts of interest.
McAteer et al. report a retrospective study of a large population-based database in the United States to identify factors associated with antireflux surgery (i.e., Nissen fundoplication) in infants, children, and adolescents hospitalized with gastroesophageal reflux disease. Perhaps the most critical question this study raises is, should the surgery be done at all, and if so, in which patients? Published guidelines (J. Pediatr. Gastroenterol. Nutr. 2009;49:498-547) suggest that other disorders that could mimic GERD should be ruled out with as much objectivity as possible prior to surgery.
One of the striking findings was that the majority of children, particularly young infants, did not undergo evaluation with diagnostic testing. Of the little diagnostic testing performed prior to surgery, upper gastrointestinal contrast fluoroscopy (UGI) was the most frequent test used. UGI, reasonable for characterizing upper GI anatomy, has no diagnostic value for GERD.
Cases were identified using ICD-9 codes, and with documented variability between clinicians in the diagnosis of GERD (Am. J. Gastroenterol. 2009;104:1278-95; quiz 96), it is concerning that not only were a great number of hospitalized children undergoing fundoplication, but over half of the cohort was 6 months or less in age.
Even more disturbing was the apparent lack of input from a consultant pediatric gastroenterologist in the decision-making process that led 11,621 (8.2%) of the 141,190 patients to antireflux surgery. Why more than half of the study cohort (52.7%) undergoing surgery to "correct" reflux at these 41 premier U.S. children's hospitals was 6 months of age or less is critical for both clinical decision making and health care utilization implications.
Numerous studies show that more than 85% of infants less than 6 months of age with reflux outgrow their reflux with little to no intervention, and, outcome studies of antireflux surgery show complications from fundoplication ranging from 8% to 28%, including death (Aliment. Pharmacol. Ther. 2007;25:1365-72; Arch. Dis. Child. 2005;90:1047-52).
In the NASPGHAN-ESPGHAN pediatric GERD guidelines, the expert panels reported that in operated children, those with neurologic impairment have more than twice the complication rate, three times the morbidity, and four times the reoperation rate of children without neurologic impairment (Am. J. Gastroenterol. 2005;100:1844-52). These data are particularly relevant to McAteer's study cohort in which antireflux surgery was performed significantly more often in those children with comorbid diagnoses of failure to thrive, neurodevelopmental delay, cardiopulmonary anomalies, cerebral palsy, aspiration pneumonia, tracheoesophageal fistula, and diaphragmatic hernia.
Thus, McAteer et al. provide a unique opportunity to not only reevaluate current clinical practice guidelines, but also implement multicenter prospective studies, using pediatric subspecialists to establish evidence-based criteria for the selection of the appropriate pediatric cases with GERD who would benefit from undergoing antireflux surgery.
Dr. Jose Garza and Dr. Benjamin D. Gold, FACG, are both in the division of pediatric gastroenterology, hepatology, and nutrition at the Children's Center for Digestive Healthcare, Atlanta. They had no relevant financial conflicts of interest.
McAteer et al. report a retrospective study of a large population-based database in the United States to identify factors associated with antireflux surgery (i.e., Nissen fundoplication) in infants, children, and adolescents hospitalized with gastroesophageal reflux disease. Perhaps the most critical question this study raises is, should the surgery be done at all, and if so, in which patients? Published guidelines (J. Pediatr. Gastroenterol. Nutr. 2009;49:498-547) suggest that other disorders that could mimic GERD should be ruled out with as much objectivity as possible prior to surgery.
One of the striking findings was that the majority of children, particularly young infants, did not undergo evaluation with diagnostic testing. Of the little diagnostic testing performed prior to surgery, upper gastrointestinal contrast fluoroscopy (UGI) was the most frequent test used. UGI, reasonable for characterizing upper GI anatomy, has no diagnostic value for GERD.
Cases were identified using ICD-9 codes, and with documented variability between clinicians in the diagnosis of GERD (Am. J. Gastroenterol. 2009;104:1278-95; quiz 96), it is concerning that not only were a great number of hospitalized children undergoing fundoplication, but over half of the cohort was 6 months or less in age.
Even more disturbing was the apparent lack of input from a consultant pediatric gastroenterologist in the decision-making process that led 11,621 (8.2%) of the 141,190 patients to antireflux surgery. Why more than half of the study cohort (52.7%) undergoing surgery to "correct" reflux at these 41 premier U.S. children's hospitals was 6 months of age or less is critical for both clinical decision making and health care utilization implications.
Numerous studies show that more than 85% of infants less than 6 months of age with reflux outgrow their reflux with little to no intervention, and, outcome studies of antireflux surgery show complications from fundoplication ranging from 8% to 28%, including death (Aliment. Pharmacol. Ther. 2007;25:1365-72; Arch. Dis. Child. 2005;90:1047-52).
In the NASPGHAN-ESPGHAN pediatric GERD guidelines, the expert panels reported that in operated children, those with neurologic impairment have more than twice the complication rate, three times the morbidity, and four times the reoperation rate of children without neurologic impairment (Am. J. Gastroenterol. 2005;100:1844-52). These data are particularly relevant to McAteer's study cohort in which antireflux surgery was performed significantly more often in those children with comorbid diagnoses of failure to thrive, neurodevelopmental delay, cardiopulmonary anomalies, cerebral palsy, aspiration pneumonia, tracheoesophageal fistula, and diaphragmatic hernia.
Thus, McAteer et al. provide a unique opportunity to not only reevaluate current clinical practice guidelines, but also implement multicenter prospective studies, using pediatric subspecialists to establish evidence-based criteria for the selection of the appropriate pediatric cases with GERD who would benefit from undergoing antireflux surgery.
Dr. Jose Garza and Dr. Benjamin D. Gold, FACG, are both in the division of pediatric gastroenterology, hepatology, and nutrition at the Children's Center for Digestive Healthcare, Atlanta. They had no relevant financial conflicts of interest.
Clinicians appear to be too quick to perform antireflux procedures in infants, compared with older children, according to a report published online Nov. 6 in JAMA Surgery.
In a retrospective cohort study involving 141,190 pediatric and adolescent hospitalizations for gastroesophageal reflux disease (GERD) across the country over an 8-year period, the proportional hazard ratio of undergoing antireflux surgery was markedly decreased for those aged 7 months to 4 years (0.63) or 5-17 years (0.43), compared with those aged 0-2 months or 2-6 months.
The reasons for this strong difference are not yet known for certain, but the data showed a lack of objective diagnostic studies preceding the surgery in all pediatric age groups, and most strikingly in the youngest patients. It may well be that clinicians are confusing physiologic regurgitation – which is common, benign, and self-resolving in infancy – with a more pathologic process, said Dr. Jarod McAteer of the division of pediatric general and thoracic surgery, Seattle Children’s Hospital, and his associates.
At the very least, it appears that many infants aren’t given an adequate trial of medical management, since most cases of gastroesophageal reflux in infancy will resolve with that alone within 3-6 months, they noted.
"Referral for surgical treatment of GERD is generally presumed to be a last resort after failure of medical management, with optimal candidates having undergone specific preoperative evaluations," the authors wrote. Diagnostic and treatment guidelines are well delineated for adults, but not so for children.
For example, "upper GI fluoroscopy is frequently used in the preoperative workup among children with GERD," even though it has been clearly demonstrated to be a poor predictor of pathologic reflux, the investigators said.
In what they described as the first study to examine the influence of patient age on progression to antireflux procedures, Dr. McAteer and his colleagues analyzed data from the Pediatric Health Information System database, which includes demographic and clinical information from 41 children’s hospitals that cover 85% of major metropolitan areas in the U.S.
Out of 141,190 patients aged 0-7 years who were hospitalized with gastroesophageal reflux or GERD, 64% were younger than 1 year of age, and 53% were younger than 6 months. These numbers highlight how common the diagnosis is in babies, they said.
They also "suggest that physicians may be more likely to apply the diagnosis in this patient group because of diagnostic uncertainty or because other characteristics of these hospitalized infants make it more likely that any regurgitation is perceived as pathologic and indicative of GERD."
Examples of such "other characteristics" include comorbidities such as neurodevelopmental delay, cardiopulmonary disorders, seizures, asthma, and cerebral palsy.
A total of 11,621 of the study population underwent antireflux procedures, of which 52.7% were aged 6 months or younger. Only 14% of these patients had first undergone upper GI endoscopy, 0.2% esophageal manometry, 1.3% a 24-hour esophageal pH study, 65% upper GI fluoroscopy, and 17.1% a gastric emptying study, the investigators said (JAMA Surg. 2013 Nov. 6 [doi: 10.1001/jamasurg.2013.2685]).
The study findings show that the threshold for performing antireflux procedures is lower in infants than in older children. And "despite the fact that expert guidelines urge the use of objective studies in the diagnosis of GERD and despite evidence that supports the use of objective studies before performing antireflux procedures, such a standardized evaluation is not common practice.
"A greater effort is needed to develop and disseminate best-practice standards for the diagnosis and treatment of children, especially infants, with possible GERD. We must clarify the indications for antireflux procedures," Dr. McAteer and his associates said.
No financial conflicts of interest were reported.
Clinicians appear to be too quick to perform antireflux procedures in infants, compared with older children, according to a report published online Nov. 6 in JAMA Surgery.
In a retrospective cohort study involving 141,190 pediatric and adolescent hospitalizations for gastroesophageal reflux disease (GERD) across the country over an 8-year period, the proportional hazard ratio of undergoing antireflux surgery was markedly decreased for those aged 7 months to 4 years (0.63) or 5-17 years (0.43), compared with those aged 0-2 months or 2-6 months.
The reasons for this strong difference are not yet known for certain, but the data showed a lack of objective diagnostic studies preceding the surgery in all pediatric age groups, and most strikingly in the youngest patients. It may well be that clinicians are confusing physiologic regurgitation – which is common, benign, and self-resolving in infancy – with a more pathologic process, said Dr. Jarod McAteer of the division of pediatric general and thoracic surgery, Seattle Children’s Hospital, and his associates.
At the very least, it appears that many infants aren’t given an adequate trial of medical management, since most cases of gastroesophageal reflux in infancy will resolve with that alone within 3-6 months, they noted.
"Referral for surgical treatment of GERD is generally presumed to be a last resort after failure of medical management, with optimal candidates having undergone specific preoperative evaluations," the authors wrote. Diagnostic and treatment guidelines are well delineated for adults, but not so for children.
For example, "upper GI fluoroscopy is frequently used in the preoperative workup among children with GERD," even though it has been clearly demonstrated to be a poor predictor of pathologic reflux, the investigators said.
In what they described as the first study to examine the influence of patient age on progression to antireflux procedures, Dr. McAteer and his colleagues analyzed data from the Pediatric Health Information System database, which includes demographic and clinical information from 41 children’s hospitals that cover 85% of major metropolitan areas in the U.S.
Out of 141,190 patients aged 0-7 years who were hospitalized with gastroesophageal reflux or GERD, 64% were younger than 1 year of age, and 53% were younger than 6 months. These numbers highlight how common the diagnosis is in babies, they said.
They also "suggest that physicians may be more likely to apply the diagnosis in this patient group because of diagnostic uncertainty or because other characteristics of these hospitalized infants make it more likely that any regurgitation is perceived as pathologic and indicative of GERD."
Examples of such "other characteristics" include comorbidities such as neurodevelopmental delay, cardiopulmonary disorders, seizures, asthma, and cerebral palsy.
A total of 11,621 of the study population underwent antireflux procedures, of which 52.7% were aged 6 months or younger. Only 14% of these patients had first undergone upper GI endoscopy, 0.2% esophageal manometry, 1.3% a 24-hour esophageal pH study, 65% upper GI fluoroscopy, and 17.1% a gastric emptying study, the investigators said (JAMA Surg. 2013 Nov. 6 [doi: 10.1001/jamasurg.2013.2685]).
The study findings show that the threshold for performing antireflux procedures is lower in infants than in older children. And "despite the fact that expert guidelines urge the use of objective studies in the diagnosis of GERD and despite evidence that supports the use of objective studies before performing antireflux procedures, such a standardized evaluation is not common practice.
"A greater effort is needed to develop and disseminate best-practice standards for the diagnosis and treatment of children, especially infants, with possible GERD. We must clarify the indications for antireflux procedures," Dr. McAteer and his associates said.
No financial conflicts of interest were reported.
FROM JAMA SURGERY
Major finding: The proportional hazard ratio of undergoing antireflux surgery was markedly decreased for patients aged 7 months to 4 years (0.63) or 5-17 years (0.43), compared with those aged 0-2 months or 2-6 months.
Data source: A retrospective cohort study involving 141,190 inpatients aged 0-17 years diagnosed as having GERD during an 8-year period, of whom 11,621 underwent antireflux procedures.
Disclosures: No financial conflicts of interest were reported.
Onartuzumab plus erlotinib extended overall survival in MET-positive lung cancer
Adding onartuzumab to erlotinib therapy improved progression-free survival and overall survival in patients whose advanced non–small cell lung cancers showed elevated expression of the MET tyrosine kinase receptor.
In contrast, compared with control subjects given erlotinib alone, patients with MET-negative tumors had earlier disease progression and poorer overall survival with onartuzumab and erlotinib in this randomized phase II trial, reported Dr. David R. Spigel of Sarah Cannon Research Institute, Nashville, Tenn., and his associates.
The findings of this industry-sponsored, preliminary study must be confirmed, and a randomized phase III trial is now underway. Meanwhile, the results indicate that adding onartuzumab to erlotinib may eventually benefit a large proportion of patients with non–small cell lung cancer (NSCLC), the investigators noted in an article published in the Journal of Clinical Oncology (2013 Oct. 7, published online before print [doi: 10.1200/JCO.2012.47.4189]).
MET is a transmembrane tyrosine kinase receptor involved in cellular proliferation, survival, motility, and invasion. Elevated MET expression is common in NSCLCs and previously has been associated with a worse prognosis. In addition, elevated MET has been implicated as a mechanism of the resistance to erlotinib that eventually develops in most NSCLC patients who take that drug.
Onartuzumab is a monoclonal antibody thought to block some MET activity. Dr. Spigel and his colleagues performed their double-blind phase II study in 137 patients with recurrent NSCLC who were randomly assigned to receive daily oral erlotinib plus either IV-infused onartuzumab (69 patients) or placebo (68 patients) every 3 weeks until the disease progressed, toxicity became unacceptable, or the patient died.
The median follow-up was 10.4 months (range, 0.1-18.4 months).
Tumor tissue from 66 patients in the active-treatment group and from 62 patients in the placebo group was available for immunohistochemical assessment of MET status.
The coprimary end points were progression-free survival in the intention-to-treat population and progression-free survival in the subgroup of 66 patients with MET-positive tumors.
In the intention-to-treat analysis, progression-free survival was not significantly different between patients who received erlotinib plus onartuzumab (2.2 months) and those who received erlotinib plus placebo (2.6 months).
However, when the data were categorized by tumor MET status, the risk of disease progression was nearly halved in patients with MET-positive tumors by the addition of onartuzumab (median progression-free survival, 2.9 months) compared with placebo (median progression-free survival, 1.5 months).
The opposite trend was noted in patients with MET-negative tumors, in whom the addition of onartuzumab markedly shortened the median progression-free survival (1.4 months), compared with placebo (2.7 months).
A similar pattern occurred with overall survival. In the intention-to-treat analysis, overall survival was not significantly different between patients given erlotinib plus onartuzumab (8.9 months) and those given erlotinib plus placebo (7.4 months).
But overall survival was nearly three times longer in patients with MET-positive tumors given erlotinib plus onartuzumab (12.6 months) than in those given erlotinib plus placebo (3.8 months). In contrast, among patients with MET-negative tumors overall survival was markedly better for those given erlotinib plus placebo (15.3 months) than for those given erlotinib plus onartuzumab (8.1 months).
The proportion of patients who discontinued treatment because of adverse events was higher with onartuzumab (11.6%) than with placebo (4.4%). The adverse events that developed more often with onartuzumab than with placebo were peripheral edema, pyrexia, asthenia, insomnia, and pneumonia.
Serious adverse events also were more frequent with the addition of onartuzumab (42%) than with the addition of placebo (33%). However, serious adverse events were impossible to distinguish from the underlying disease (pneumonia, dyspnea, hemoptysis, pulmonary embolism, and respiratory distress) or from the concomitant erlotinib therapy (interstitial lung disease and rash).
The reason for the differential response between MET-positive and MET-negative NSCLCs is not yet fully understood, Dr. Spigel and his associates said.
This study was sponsored by Genentech, which also provided the study drugs. Dr. Spigel and his associates reported ties to Genentech and numerous other industry sources.
Adding onartuzumab to erlotinib therapy improved progression-free survival and overall survival in patients whose advanced non–small cell lung cancers showed elevated expression of the MET tyrosine kinase receptor.
In contrast, compared with control subjects given erlotinib alone, patients with MET-negative tumors had earlier disease progression and poorer overall survival with onartuzumab and erlotinib in this randomized phase II trial, reported Dr. David R. Spigel of Sarah Cannon Research Institute, Nashville, Tenn., and his associates.
The findings of this industry-sponsored, preliminary study must be confirmed, and a randomized phase III trial is now underway. Meanwhile, the results indicate that adding onartuzumab to erlotinib may eventually benefit a large proportion of patients with non–small cell lung cancer (NSCLC), the investigators noted in an article published in the Journal of Clinical Oncology (2013 Oct. 7, published online before print [doi: 10.1200/JCO.2012.47.4189]).
MET is a transmembrane tyrosine kinase receptor involved in cellular proliferation, survival, motility, and invasion. Elevated MET expression is common in NSCLCs and previously has been associated with a worse prognosis. In addition, elevated MET has been implicated as a mechanism of the resistance to erlotinib that eventually develops in most NSCLC patients who take that drug.
Onartuzumab is a monoclonal antibody thought to block some MET activity. Dr. Spigel and his colleagues performed their double-blind phase II study in 137 patients with recurrent NSCLC who were randomly assigned to receive daily oral erlotinib plus either IV-infused onartuzumab (69 patients) or placebo (68 patients) every 3 weeks until the disease progressed, toxicity became unacceptable, or the patient died.
The median follow-up was 10.4 months (range, 0.1-18.4 months).
Tumor tissue from 66 patients in the active-treatment group and from 62 patients in the placebo group was available for immunohistochemical assessment of MET status.
The coprimary end points were progression-free survival in the intention-to-treat population and progression-free survival in the subgroup of 66 patients with MET-positive tumors.
In the intention-to-treat analysis, progression-free survival was not significantly different between patients who received erlotinib plus onartuzumab (2.2 months) and those who received erlotinib plus placebo (2.6 months).
However, when the data were categorized by tumor MET status, the risk of disease progression was nearly halved in patients with MET-positive tumors by the addition of onartuzumab (median progression-free survival, 2.9 months) compared with placebo (median progression-free survival, 1.5 months).
The opposite trend was noted in patients with MET-negative tumors, in whom the addition of onartuzumab markedly shortened the median progression-free survival (1.4 months), compared with placebo (2.7 months).
A similar pattern occurred with overall survival. In the intention-to-treat analysis, overall survival was not significantly different between patients given erlotinib plus onartuzumab (8.9 months) and those given erlotinib plus placebo (7.4 months).
But overall survival was nearly three times longer in patients with MET-positive tumors given erlotinib plus onartuzumab (12.6 months) than in those given erlotinib plus placebo (3.8 months). In contrast, among patients with MET-negative tumors overall survival was markedly better for those given erlotinib plus placebo (15.3 months) than for those given erlotinib plus onartuzumab (8.1 months).
The proportion of patients who discontinued treatment because of adverse events was higher with onartuzumab (11.6%) than with placebo (4.4%). The adverse events that developed more often with onartuzumab than with placebo were peripheral edema, pyrexia, asthenia, insomnia, and pneumonia.
Serious adverse events also were more frequent with the addition of onartuzumab (42%) than with the addition of placebo (33%). However, serious adverse events were impossible to distinguish from the underlying disease (pneumonia, dyspnea, hemoptysis, pulmonary embolism, and respiratory distress) or from the concomitant erlotinib therapy (interstitial lung disease and rash).
The reason for the differential response between MET-positive and MET-negative NSCLCs is not yet fully understood, Dr. Spigel and his associates said.
This study was sponsored by Genentech, which also provided the study drugs. Dr. Spigel and his associates reported ties to Genentech and numerous other industry sources.
Adding onartuzumab to erlotinib therapy improved progression-free survival and overall survival in patients whose advanced non–small cell lung cancers showed elevated expression of the MET tyrosine kinase receptor.
In contrast, compared with control subjects given erlotinib alone, patients with MET-negative tumors had earlier disease progression and poorer overall survival with onartuzumab and erlotinib in this randomized phase II trial, reported Dr. David R. Spigel of Sarah Cannon Research Institute, Nashville, Tenn., and his associates.
The findings of this industry-sponsored, preliminary study must be confirmed, and a randomized phase III trial is now underway. Meanwhile, the results indicate that adding onartuzumab to erlotinib may eventually benefit a large proportion of patients with non–small cell lung cancer (NSCLC), the investigators noted in an article published in the Journal of Clinical Oncology (2013 Oct. 7, published online before print [doi: 10.1200/JCO.2012.47.4189]).
MET is a transmembrane tyrosine kinase receptor involved in cellular proliferation, survival, motility, and invasion. Elevated MET expression is common in NSCLCs and previously has been associated with a worse prognosis. In addition, elevated MET has been implicated as a mechanism of the resistance to erlotinib that eventually develops in most NSCLC patients who take that drug.
Onartuzumab is a monoclonal antibody thought to block some MET activity. Dr. Spigel and his colleagues performed their double-blind phase II study in 137 patients with recurrent NSCLC who were randomly assigned to receive daily oral erlotinib plus either IV-infused onartuzumab (69 patients) or placebo (68 patients) every 3 weeks until the disease progressed, toxicity became unacceptable, or the patient died.
The median follow-up was 10.4 months (range, 0.1-18.4 months).
Tumor tissue from 66 patients in the active-treatment group and from 62 patients in the placebo group was available for immunohistochemical assessment of MET status.
The coprimary end points were progression-free survival in the intention-to-treat population and progression-free survival in the subgroup of 66 patients with MET-positive tumors.
In the intention-to-treat analysis, progression-free survival was not significantly different between patients who received erlotinib plus onartuzumab (2.2 months) and those who received erlotinib plus placebo (2.6 months).
However, when the data were categorized by tumor MET status, the risk of disease progression was nearly halved in patients with MET-positive tumors by the addition of onartuzumab (median progression-free survival, 2.9 months) compared with placebo (median progression-free survival, 1.5 months).
The opposite trend was noted in patients with MET-negative tumors, in whom the addition of onartuzumab markedly shortened the median progression-free survival (1.4 months), compared with placebo (2.7 months).
A similar pattern occurred with overall survival. In the intention-to-treat analysis, overall survival was not significantly different between patients given erlotinib plus onartuzumab (8.9 months) and those given erlotinib plus placebo (7.4 months).
But overall survival was nearly three times longer in patients with MET-positive tumors given erlotinib plus onartuzumab (12.6 months) than in those given erlotinib plus placebo (3.8 months). In contrast, among patients with MET-negative tumors overall survival was markedly better for those given erlotinib plus placebo (15.3 months) than for those given erlotinib plus onartuzumab (8.1 months).
The proportion of patients who discontinued treatment because of adverse events was higher with onartuzumab (11.6%) than with placebo (4.4%). The adverse events that developed more often with onartuzumab than with placebo were peripheral edema, pyrexia, asthenia, insomnia, and pneumonia.
Serious adverse events also were more frequent with the addition of onartuzumab (42%) than with the addition of placebo (33%). However, serious adverse events were impossible to distinguish from the underlying disease (pneumonia, dyspnea, hemoptysis, pulmonary embolism, and respiratory distress) or from the concomitant erlotinib therapy (interstitial lung disease and rash).
The reason for the differential response between MET-positive and MET-negative NSCLCs is not yet fully understood, Dr. Spigel and his associates said.
This study was sponsored by Genentech, which also provided the study drugs. Dr. Spigel and his associates reported ties to Genentech and numerous other industry sources.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Major finding: In patients with MET-positive tumors, overall survival was nearly three times longer with erlotinib plus onartuzumab (12.6 months) than with erlotinib plus placebo (3.8 months).
Data source: A randomized, double-blind phase II clinical trial involving 137 adults with advanced NSCLC who were treated with either erlotinib plus onartuzumab or erlotinib plus placebo and were followed for a median of 10.4 months.
Disclosures: This study was sponsored by Genentech, which also provided the study drugs. Dr. Spigel and his associates reported ties to Genentech and numerous other industry sources.
Testosterone for ‘low T’ Raises Death, MI, and Stroke Risk
Testosterone therapy taken for "low T" raises the risk of mortality, myocardial infarction, and ischemic stroke, according to a report published online in JAMA.
The risk elevation is consistent in both men who have existing coronary artery disease and men who do not, and is not affected by differences in cardiovascular risk factors such as blood pressure or by the use of preventive medications, said Dr. Rebecca Vigen of the University of Texas at Southwestern Medical Center, Dallas, and her associates.
These findings, from what the investigators describe as the first observational study to suggest a causal link between testosterone therapy and adverse cardiovascular outcomes, "raise concerns about the potential safety of testosterone therapy." Randomized clinical trials are warranted to clarify this issue, they noted.
"Although physicians should continue to discuss the symptomatic benefits of testosterone therapy with patients, it is also important to inform patients that long-term risks are unknown and there is a possibility that testosterone therapy might be harmful," Dr. Vigen and her colleagues added.
Previous studies have shown that testosterone therapy improves sexual function and strength, as well as some cardiovascular risk factors such as lipid profiles and insulin resistance. They have not detected adverse CV effects, "but these trials were generally focused on intermediate endpoints, of short duration, and not powered for clinical endpoints," the investigators wrote.
One clinical trial that included many men with CV disorders was halted early in 2010 when the group receiving testosterone was found to have an excess of CV events, compared with the placebo group.
Dr. Vigen and her associates examined the potential CV risks of testosterone therapy in a retrospective cohort study using data from the Veterans Administration’s Clinical Assessment Reporting and Tracking (CART) program, which collects information on all procedures performed in the 76 VA cardiac catheterization laboratories across the country. They identified all men who underwent coronary angiography at these facilities in 2005-2011 (who therefore had well-characterized cardiovascular profiles) and whose medical records also showed a total testosterone level of less than 300 ng/dL.
This cohort of 8,709 men had a high burden of comorbidities: 20% had a history of MI, half had diabetes, and more than 80% had coronary artery disease. Rates of these comorbidities were approximately equal between men taking testosterone and men not taking testosterone.
A total of 1,223 of these men were taking testosterone therapy to address their low testosterone levels. Roughly 1% of them were using testosterone gel, 36% were using injections, and 63% were using patches. Men who used testosterone tended to be younger and healthier than those who did not.
During an average of 28 months of follow-up, there were 1,710 adverse events of interest: 748 men died, 443 had MIs, and 519 had strokes. The absolute risk of these events was significantly greater in testosterone users than in nonusers: 1.3% higher at 1 year, 3.1% higher at 2 years, and 5.8% higher at 3 years, the investigators said (JAMA 2013 [doi:10.1001/jama2013.280386]).
In a further, adjusted analysis of the data, testosterone use remained strongly associated with increased risk of death, MI, or stroke, with a hazard ratio of 1.29. This indicates that testosterone users were 29% more likely than were nonusers to experience these adverse outcomes.
These findings were unchanged after the data were further adjusted to account for the presence or absence of CAD and the use or nonuse of coronary revascularization procedures.
There was no significant difference in risk of adverse outcomes among the three formulations of testosterone.
The researchers noted that there are several potential mechanisms by which testosterone therapy might increase cardiovascular risk. It increases platelet aggregation, which contributes to arterial plaque formation. It enhances monocyte activation in the endothelium, which promotes atherosclerosis and is implicated in the pathogenesis of acute coronary syndromes. And it aggravates sleep-disordered breathing, another CV risk factor, they said.
This study was supported by the U.S. Department of Veterans Affairs. No financial conflicts of interest were reported.
The findings by Vigen et al. should prompt wariness among clinicians and patients alike, especially in light of the high volume of prescriptions and "aggressive marketing by testosterone manufacturers," said Dr. Anne R. Cappola.
The most important question raised by the study results may be how they can be generalized to the broader population of men taking testosterone: men of this age group who are taking testosterone for "low-T syndrome" or for antiaging purposes, and younger men taking it for physical enhancement. "Does the 29% increased risk of myocardial infarction, ischemic stroke, or mortality apply to these groups? Are the benefits – real or perceived – for these groups of men worth any increase in risk? These populations represent a sizable group of testosterone users, and there is only anecdotal evidence that testosterone is safe for these men."
Testosterone treatment is prescribed for an estimated 3% of U.S. men over age 40 years, even as evidence indicating that it exerts adverse cardiovascular effects is accumulating. The study results warrant "both cautious testosterone prescribing and additional investigation," she said.
Dr. Anne R. Cappola is in the division of endocrinology, diabetes, and metabolism at the University of Pennsylvania, Philadelphia. She is also an associate editor of JAMA. She reported ties to Abbott Laboratories and BioSante Pharmaceuticals. These remarks were taken from her editorial accompanying Dr. Vigen’s report (JAMA 2013;310:1805-6).
The findings by Vigen et al. should prompt wariness among clinicians and patients alike, especially in light of the high volume of prescriptions and "aggressive marketing by testosterone manufacturers," said Dr. Anne R. Cappola.
The most important question raised by the study results may be how they can be generalized to the broader population of men taking testosterone: men of this age group who are taking testosterone for "low-T syndrome" or for antiaging purposes, and younger men taking it for physical enhancement. "Does the 29% increased risk of myocardial infarction, ischemic stroke, or mortality apply to these groups? Are the benefits – real or perceived – for these groups of men worth any increase in risk? These populations represent a sizable group of testosterone users, and there is only anecdotal evidence that testosterone is safe for these men."
Testosterone treatment is prescribed for an estimated 3% of U.S. men over age 40 years, even as evidence indicating that it exerts adverse cardiovascular effects is accumulating. The study results warrant "both cautious testosterone prescribing and additional investigation," she said.
Dr. Anne R. Cappola is in the division of endocrinology, diabetes, and metabolism at the University of Pennsylvania, Philadelphia. She is also an associate editor of JAMA. She reported ties to Abbott Laboratories and BioSante Pharmaceuticals. These remarks were taken from her editorial accompanying Dr. Vigen’s report (JAMA 2013;310:1805-6).
The findings by Vigen et al. should prompt wariness among clinicians and patients alike, especially in light of the high volume of prescriptions and "aggressive marketing by testosterone manufacturers," said Dr. Anne R. Cappola.
The most important question raised by the study results may be how they can be generalized to the broader population of men taking testosterone: men of this age group who are taking testosterone for "low-T syndrome" or for antiaging purposes, and younger men taking it for physical enhancement. "Does the 29% increased risk of myocardial infarction, ischemic stroke, or mortality apply to these groups? Are the benefits – real or perceived – for these groups of men worth any increase in risk? These populations represent a sizable group of testosterone users, and there is only anecdotal evidence that testosterone is safe for these men."
Testosterone treatment is prescribed for an estimated 3% of U.S. men over age 40 years, even as evidence indicating that it exerts adverse cardiovascular effects is accumulating. The study results warrant "both cautious testosterone prescribing and additional investigation," she said.
Dr. Anne R. Cappola is in the division of endocrinology, diabetes, and metabolism at the University of Pennsylvania, Philadelphia. She is also an associate editor of JAMA. She reported ties to Abbott Laboratories and BioSante Pharmaceuticals. These remarks were taken from her editorial accompanying Dr. Vigen’s report (JAMA 2013;310:1805-6).
Testosterone therapy taken for "low T" raises the risk of mortality, myocardial infarction, and ischemic stroke, according to a report published online in JAMA.
The risk elevation is consistent in both men who have existing coronary artery disease and men who do not, and is not affected by differences in cardiovascular risk factors such as blood pressure or by the use of preventive medications, said Dr. Rebecca Vigen of the University of Texas at Southwestern Medical Center, Dallas, and her associates.
These findings, from what the investigators describe as the first observational study to suggest a causal link between testosterone therapy and adverse cardiovascular outcomes, "raise concerns about the potential safety of testosterone therapy." Randomized clinical trials are warranted to clarify this issue, they noted.
"Although physicians should continue to discuss the symptomatic benefits of testosterone therapy with patients, it is also important to inform patients that long-term risks are unknown and there is a possibility that testosterone therapy might be harmful," Dr. Vigen and her colleagues added.
Previous studies have shown that testosterone therapy improves sexual function and strength, as well as some cardiovascular risk factors such as lipid profiles and insulin resistance. They have not detected adverse CV effects, "but these trials were generally focused on intermediate endpoints, of short duration, and not powered for clinical endpoints," the investigators wrote.
One clinical trial that included many men with CV disorders was halted early in 2010 when the group receiving testosterone was found to have an excess of CV events, compared with the placebo group.
Dr. Vigen and her associates examined the potential CV risks of testosterone therapy in a retrospective cohort study using data from the Veterans Administration’s Clinical Assessment Reporting and Tracking (CART) program, which collects information on all procedures performed in the 76 VA cardiac catheterization laboratories across the country. They identified all men who underwent coronary angiography at these facilities in 2005-2011 (who therefore had well-characterized cardiovascular profiles) and whose medical records also showed a total testosterone level of less than 300 ng/dL.
This cohort of 8,709 men had a high burden of comorbidities: 20% had a history of MI, half had diabetes, and more than 80% had coronary artery disease. Rates of these comorbidities were approximately equal between men taking testosterone and men not taking testosterone.
A total of 1,223 of these men were taking testosterone therapy to address their low testosterone levels. Roughly 1% of them were using testosterone gel, 36% were using injections, and 63% were using patches. Men who used testosterone tended to be younger and healthier than those who did not.
During an average of 28 months of follow-up, there were 1,710 adverse events of interest: 748 men died, 443 had MIs, and 519 had strokes. The absolute risk of these events was significantly greater in testosterone users than in nonusers: 1.3% higher at 1 year, 3.1% higher at 2 years, and 5.8% higher at 3 years, the investigators said (JAMA 2013 [doi:10.1001/jama2013.280386]).
In a further, adjusted analysis of the data, testosterone use remained strongly associated with increased risk of death, MI, or stroke, with a hazard ratio of 1.29. This indicates that testosterone users were 29% more likely than were nonusers to experience these adverse outcomes.
These findings were unchanged after the data were further adjusted to account for the presence or absence of CAD and the use or nonuse of coronary revascularization procedures.
There was no significant difference in risk of adverse outcomes among the three formulations of testosterone.
The researchers noted that there are several potential mechanisms by which testosterone therapy might increase cardiovascular risk. It increases platelet aggregation, which contributes to arterial plaque formation. It enhances monocyte activation in the endothelium, which promotes atherosclerosis and is implicated in the pathogenesis of acute coronary syndromes. And it aggravates sleep-disordered breathing, another CV risk factor, they said.
This study was supported by the U.S. Department of Veterans Affairs. No financial conflicts of interest were reported.
Testosterone therapy taken for "low T" raises the risk of mortality, myocardial infarction, and ischemic stroke, according to a report published online in JAMA.
The risk elevation is consistent in both men who have existing coronary artery disease and men who do not, and is not affected by differences in cardiovascular risk factors such as blood pressure or by the use of preventive medications, said Dr. Rebecca Vigen of the University of Texas at Southwestern Medical Center, Dallas, and her associates.
These findings, from what the investigators describe as the first observational study to suggest a causal link between testosterone therapy and adverse cardiovascular outcomes, "raise concerns about the potential safety of testosterone therapy." Randomized clinical trials are warranted to clarify this issue, they noted.
"Although physicians should continue to discuss the symptomatic benefits of testosterone therapy with patients, it is also important to inform patients that long-term risks are unknown and there is a possibility that testosterone therapy might be harmful," Dr. Vigen and her colleagues added.
Previous studies have shown that testosterone therapy improves sexual function and strength, as well as some cardiovascular risk factors such as lipid profiles and insulin resistance. They have not detected adverse CV effects, "but these trials were generally focused on intermediate endpoints, of short duration, and not powered for clinical endpoints," the investigators wrote.
One clinical trial that included many men with CV disorders was halted early in 2010 when the group receiving testosterone was found to have an excess of CV events, compared with the placebo group.
Dr. Vigen and her associates examined the potential CV risks of testosterone therapy in a retrospective cohort study using data from the Veterans Administration’s Clinical Assessment Reporting and Tracking (CART) program, which collects information on all procedures performed in the 76 VA cardiac catheterization laboratories across the country. They identified all men who underwent coronary angiography at these facilities in 2005-2011 (who therefore had well-characterized cardiovascular profiles) and whose medical records also showed a total testosterone level of less than 300 ng/dL.
This cohort of 8,709 men had a high burden of comorbidities: 20% had a history of MI, half had diabetes, and more than 80% had coronary artery disease. Rates of these comorbidities were approximately equal between men taking testosterone and men not taking testosterone.
A total of 1,223 of these men were taking testosterone therapy to address their low testosterone levels. Roughly 1% of them were using testosterone gel, 36% were using injections, and 63% were using patches. Men who used testosterone tended to be younger and healthier than those who did not.
During an average of 28 months of follow-up, there were 1,710 adverse events of interest: 748 men died, 443 had MIs, and 519 had strokes. The absolute risk of these events was significantly greater in testosterone users than in nonusers: 1.3% higher at 1 year, 3.1% higher at 2 years, and 5.8% higher at 3 years, the investigators said (JAMA 2013 [doi:10.1001/jama2013.280386]).
In a further, adjusted analysis of the data, testosterone use remained strongly associated with increased risk of death, MI, or stroke, with a hazard ratio of 1.29. This indicates that testosterone users were 29% more likely than were nonusers to experience these adverse outcomes.
These findings were unchanged after the data were further adjusted to account for the presence or absence of CAD and the use or nonuse of coronary revascularization procedures.
There was no significant difference in risk of adverse outcomes among the three formulations of testosterone.
The researchers noted that there are several potential mechanisms by which testosterone therapy might increase cardiovascular risk. It increases platelet aggregation, which contributes to arterial plaque formation. It enhances monocyte activation in the endothelium, which promotes atherosclerosis and is implicated in the pathogenesis of acute coronary syndromes. And it aggravates sleep-disordered breathing, another CV risk factor, they said.
This study was supported by the U.S. Department of Veterans Affairs. No financial conflicts of interest were reported.
FROM JAMA