User login
Metformin shows superior glycemic durability
Metformin showed superior glycemic durability compared with sulfonylurea and meglitinide in a nationwide observational study in Sweden, according to a report published online in BMJ Open Diabetes Research & Care.
To study glycemic durability in real-world patients, researchers analyzed data from five Swedish national registries of diabetes care, prescribed drugs, causes of death, hospital discharges, and health insurance. They examined the medical records of 69,667 adults who had heretofore untreated type 2 diabetes and were initially prescribed metformin, sulfonylurea, or meglitinide during a 4-year period. They also analyzed medication use in a subset of propensity-matched patients who were well balanced for numerous covariates that can impact diabetes progression, such as age, gender, hemoglobin A1c level, eGFR, and CVD history.
Each medication’s glycemic durability was judged according to whether these patients continued using their initial diabetes drug, switched to a different drug, or added a second drug during 5.5 years of follow-up, said Dr. Nils Ekstrom of the University of Gothenburg (Sweden) and his associates.
Mean patient age was 71 years, mean duration of diabetes was 5 years, and mean body mass index was 28 kg/m2. Overall, almost half of these patients discontinued their initial medication, switched to a different medication, or required a second diabetes drug be added to their regimen during follow-up.
Compared with metformin, sulfonylurea and meglitinide showed a significantly increased risk of monotherapy failure (hazard ratio, 1.74). Sulfonylurea and meglitinide had an even greater risk of requiring a switch to a new agent (HR, 2.81) and of requiring an add-on agent (HR, 3.14). These results from the main analyses were confirmed in two sensitivity analyses involving approximately 60,000 participants, the investigators said (BMJ Open Diab. Res. Care 2015 [doi:10.1136/bmjdrc-2014-000059]).
Patients who continued on their initial monotherapy throughout follow-up showed improved HbA1c levels of approximately 10%, regardless of which medication they used. This group represents treatment responders. In contrast, patients who discontinued, switched, or added a new agent to their initial medication showed stable or slightly increasing HbA1c levels over time, which represented deteriorating glycemic control.
“These results strengthen the current evidence of a superior durability with metformin compared with sulfonylureas” in the real-world setting, and suggest that the same is true regarding meglitinide, Dr. Ekstrom and his associates said.
Metformin showed superior glycemic durability compared with sulfonylurea and meglitinide in a nationwide observational study in Sweden, according to a report published online in BMJ Open Diabetes Research & Care.
To study glycemic durability in real-world patients, researchers analyzed data from five Swedish national registries of diabetes care, prescribed drugs, causes of death, hospital discharges, and health insurance. They examined the medical records of 69,667 adults who had heretofore untreated type 2 diabetes and were initially prescribed metformin, sulfonylurea, or meglitinide during a 4-year period. They also analyzed medication use in a subset of propensity-matched patients who were well balanced for numerous covariates that can impact diabetes progression, such as age, gender, hemoglobin A1c level, eGFR, and CVD history.
Each medication’s glycemic durability was judged according to whether these patients continued using their initial diabetes drug, switched to a different drug, or added a second drug during 5.5 years of follow-up, said Dr. Nils Ekstrom of the University of Gothenburg (Sweden) and his associates.
Mean patient age was 71 years, mean duration of diabetes was 5 years, and mean body mass index was 28 kg/m2. Overall, almost half of these patients discontinued their initial medication, switched to a different medication, or required a second diabetes drug be added to their regimen during follow-up.
Compared with metformin, sulfonylurea and meglitinide showed a significantly increased risk of monotherapy failure (hazard ratio, 1.74). Sulfonylurea and meglitinide had an even greater risk of requiring a switch to a new agent (HR, 2.81) and of requiring an add-on agent (HR, 3.14). These results from the main analyses were confirmed in two sensitivity analyses involving approximately 60,000 participants, the investigators said (BMJ Open Diab. Res. Care 2015 [doi:10.1136/bmjdrc-2014-000059]).
Patients who continued on their initial monotherapy throughout follow-up showed improved HbA1c levels of approximately 10%, regardless of which medication they used. This group represents treatment responders. In contrast, patients who discontinued, switched, or added a new agent to their initial medication showed stable or slightly increasing HbA1c levels over time, which represented deteriorating glycemic control.
“These results strengthen the current evidence of a superior durability with metformin compared with sulfonylureas” in the real-world setting, and suggest that the same is true regarding meglitinide, Dr. Ekstrom and his associates said.
Metformin showed superior glycemic durability compared with sulfonylurea and meglitinide in a nationwide observational study in Sweden, according to a report published online in BMJ Open Diabetes Research & Care.
To study glycemic durability in real-world patients, researchers analyzed data from five Swedish national registries of diabetes care, prescribed drugs, causes of death, hospital discharges, and health insurance. They examined the medical records of 69,667 adults who had heretofore untreated type 2 diabetes and were initially prescribed metformin, sulfonylurea, or meglitinide during a 4-year period. They also analyzed medication use in a subset of propensity-matched patients who were well balanced for numerous covariates that can impact diabetes progression, such as age, gender, hemoglobin A1c level, eGFR, and CVD history.
Each medication’s glycemic durability was judged according to whether these patients continued using their initial diabetes drug, switched to a different drug, or added a second drug during 5.5 years of follow-up, said Dr. Nils Ekstrom of the University of Gothenburg (Sweden) and his associates.
Mean patient age was 71 years, mean duration of diabetes was 5 years, and mean body mass index was 28 kg/m2. Overall, almost half of these patients discontinued their initial medication, switched to a different medication, or required a second diabetes drug be added to their regimen during follow-up.
Compared with metformin, sulfonylurea and meglitinide showed a significantly increased risk of monotherapy failure (hazard ratio, 1.74). Sulfonylurea and meglitinide had an even greater risk of requiring a switch to a new agent (HR, 2.81) and of requiring an add-on agent (HR, 3.14). These results from the main analyses were confirmed in two sensitivity analyses involving approximately 60,000 participants, the investigators said (BMJ Open Diab. Res. Care 2015 [doi:10.1136/bmjdrc-2014-000059]).
Patients who continued on their initial monotherapy throughout follow-up showed improved HbA1c levels of approximately 10%, regardless of which medication they used. This group represents treatment responders. In contrast, patients who discontinued, switched, or added a new agent to their initial medication showed stable or slightly increasing HbA1c levels over time, which represented deteriorating glycemic control.
“These results strengthen the current evidence of a superior durability with metformin compared with sulfonylureas” in the real-world setting, and suggest that the same is true regarding meglitinide, Dr. Ekstrom and his associates said.
FROM BMJ OPEN DIABETES RESEARCH & CARE
Key clinical point: Metformin’s glycemic durability was superior to that of sulfonylurea and meglitinide in a nationwide observational study in Sweden.
Major finding: Compared with metformin, sulfonylurea and meglitinide showed a significantly increased risk of monotherapy failure, with an hazard ratio of 1.74.
Data source: An observational cohort study of initial use of diabetes medication during a 5-year period in 69,667 adults across Sweden.
Disclosures: The Swedish Association of Local Authorities and Regions supported the study. Dr. Ekstrom and his associates reported having no financial disclosures.
Glyburide in pregnancy linked to respiratory distress, NICU admission
Newborns of women who have gestational diabetes treated with glyburide appear to be at increased risk for admission to the neonatal intensive care unit, respiratory distress, hypoglycemia, birth injury, and large for gestational age status, compared with newborns of women treated with insulin, according to a report published March 30 in JAMA Pediatrics.
In recent years, off-label use of glyburide for gestational diabetes has risen dramatically in the United States. A few small clinical trials and observational studies have suggested that the agent is associated with poorer neonatal outcomes than insulin. Currently, insulin is the only pharmacologic treatment approved for the treatment of gestational diabetes mellitus by the Food and Drug Administration and endorsed by the American Diabetes Association.
To examine the issue in a study population large enough to detect small, but clinically important, differences between the two treatments, researchers performed a retrospective cohort study involving 9,173 mother/infant pairs enrolled in a health plan database that covered approximately 30 million patients per year across the United States.
Compared with insulin, glyburide was associated with an increased risk in the newborns of NICU admission (adjusted relative risk, 1.41), respiratory distress (aRR, 1.63), neonatal hypoglycemia (aRR, 1.40), birth injury (aRR, 1.35), and large for gestation age status (aRR, 1.43).
Glyburide was not associated with an increased risk of neonatal jaundice, obstetric trauma, or preterm birth, the investigators wrote (JAMA Pediatr. 2015 March 30 [doi:10.1001/jamapediatrics.2015.74]).
The study population included women (average age, 33.5 years) who delivered singleton live-born infants in 2000-2011 and who filed pharmacy claims for either glyburide (4,982 patients) or insulin (4,191 patients) during the 150 days preceding delivery.
The mean duration of treatment was 50.4 days with glyburide and 54.1 days with insulin.
The proportion of women treated with glyburide rose from 8.5% at the beginning of the study period to 64.4% at the end, wrote Wendy Camelo Castillo, Ph.D., of the department of pharmaceutical health services research, University of Maryland, Baltimore, and her associates.
Glyburide’s link to adverse outcomes “demands further attention” and suggests that women treated with the drug are not achieving adequate glucose control, the researchers wrote.
The Agency for Healthcare Research and Quality and the University of North Carolina at Chapel Hill supported the study. Dr. Camelo Castillo reported having no financial disclosures; her associates reported financial ties to varous pharmaceutical companies.
These findings, together with those of previous studies, are cause for concern and indicate that the time has come to reconsider the place of glyburide in pregnancy.
Infants of women treated with glyburide had a 41% higher risk for NICU admission, a 63% higher risk of respiratory distress, a 40% higher risk of hypoglycemia, and a 35% higher risk of birth injury, compared with infants born to women treated with insulin. The large study sample provides much more statistical power than prior randomized clinical trials to detect any differences in outcomes and to characterize those differences with greater precision.

|
Dr. Richard I.G. Holt |
Richard I.G. Holt, Ph.D., is in the human development and health academic unit at the University of Southampton (U.K.). He was a member of the 2008 National Institute for Health and Care Excellence diabetes and pregnancy guideline development group. He reported having no financial disclosures. These comments are based on Dr. Holt’s accompanying editorial (JAMA Pediatr. 2015 March 30 [doi:10.1001/jamapediatrics.2015.144]).
These findings, together with those of previous studies, are cause for concern and indicate that the time has come to reconsider the place of glyburide in pregnancy.
Infants of women treated with glyburide had a 41% higher risk for NICU admission, a 63% higher risk of respiratory distress, a 40% higher risk of hypoglycemia, and a 35% higher risk of birth injury, compared with infants born to women treated with insulin. The large study sample provides much more statistical power than prior randomized clinical trials to detect any differences in outcomes and to characterize those differences with greater precision.

|
Dr. Richard I.G. Holt |
Richard I.G. Holt, Ph.D., is in the human development and health academic unit at the University of Southampton (U.K.). He was a member of the 2008 National Institute for Health and Care Excellence diabetes and pregnancy guideline development group. He reported having no financial disclosures. These comments are based on Dr. Holt’s accompanying editorial (JAMA Pediatr. 2015 March 30 [doi:10.1001/jamapediatrics.2015.144]).
These findings, together with those of previous studies, are cause for concern and indicate that the time has come to reconsider the place of glyburide in pregnancy.
Infants of women treated with glyburide had a 41% higher risk for NICU admission, a 63% higher risk of respiratory distress, a 40% higher risk of hypoglycemia, and a 35% higher risk of birth injury, compared with infants born to women treated with insulin. The large study sample provides much more statistical power than prior randomized clinical trials to detect any differences in outcomes and to characterize those differences with greater precision.

|
Dr. Richard I.G. Holt |
Richard I.G. Holt, Ph.D., is in the human development and health academic unit at the University of Southampton (U.K.). He was a member of the 2008 National Institute for Health and Care Excellence diabetes and pregnancy guideline development group. He reported having no financial disclosures. These comments are based on Dr. Holt’s accompanying editorial (JAMA Pediatr. 2015 March 30 [doi:10.1001/jamapediatrics.2015.144]).
Newborns of women who have gestational diabetes treated with glyburide appear to be at increased risk for admission to the neonatal intensive care unit, respiratory distress, hypoglycemia, birth injury, and large for gestational age status, compared with newborns of women treated with insulin, according to a report published March 30 in JAMA Pediatrics.
In recent years, off-label use of glyburide for gestational diabetes has risen dramatically in the United States. A few small clinical trials and observational studies have suggested that the agent is associated with poorer neonatal outcomes than insulin. Currently, insulin is the only pharmacologic treatment approved for the treatment of gestational diabetes mellitus by the Food and Drug Administration and endorsed by the American Diabetes Association.
To examine the issue in a study population large enough to detect small, but clinically important, differences between the two treatments, researchers performed a retrospective cohort study involving 9,173 mother/infant pairs enrolled in a health plan database that covered approximately 30 million patients per year across the United States.
Compared with insulin, glyburide was associated with an increased risk in the newborns of NICU admission (adjusted relative risk, 1.41), respiratory distress (aRR, 1.63), neonatal hypoglycemia (aRR, 1.40), birth injury (aRR, 1.35), and large for gestation age status (aRR, 1.43).
Glyburide was not associated with an increased risk of neonatal jaundice, obstetric trauma, or preterm birth, the investigators wrote (JAMA Pediatr. 2015 March 30 [doi:10.1001/jamapediatrics.2015.74]).
The study population included women (average age, 33.5 years) who delivered singleton live-born infants in 2000-2011 and who filed pharmacy claims for either glyburide (4,982 patients) or insulin (4,191 patients) during the 150 days preceding delivery.
The mean duration of treatment was 50.4 days with glyburide and 54.1 days with insulin.
The proportion of women treated with glyburide rose from 8.5% at the beginning of the study period to 64.4% at the end, wrote Wendy Camelo Castillo, Ph.D., of the department of pharmaceutical health services research, University of Maryland, Baltimore, and her associates.
Glyburide’s link to adverse outcomes “demands further attention” and suggests that women treated with the drug are not achieving adequate glucose control, the researchers wrote.
The Agency for Healthcare Research and Quality and the University of North Carolina at Chapel Hill supported the study. Dr. Camelo Castillo reported having no financial disclosures; her associates reported financial ties to varous pharmaceutical companies.
Newborns of women who have gestational diabetes treated with glyburide appear to be at increased risk for admission to the neonatal intensive care unit, respiratory distress, hypoglycemia, birth injury, and large for gestational age status, compared with newborns of women treated with insulin, according to a report published March 30 in JAMA Pediatrics.
In recent years, off-label use of glyburide for gestational diabetes has risen dramatically in the United States. A few small clinical trials and observational studies have suggested that the agent is associated with poorer neonatal outcomes than insulin. Currently, insulin is the only pharmacologic treatment approved for the treatment of gestational diabetes mellitus by the Food and Drug Administration and endorsed by the American Diabetes Association.
To examine the issue in a study population large enough to detect small, but clinically important, differences between the two treatments, researchers performed a retrospective cohort study involving 9,173 mother/infant pairs enrolled in a health plan database that covered approximately 30 million patients per year across the United States.
Compared with insulin, glyburide was associated with an increased risk in the newborns of NICU admission (adjusted relative risk, 1.41), respiratory distress (aRR, 1.63), neonatal hypoglycemia (aRR, 1.40), birth injury (aRR, 1.35), and large for gestation age status (aRR, 1.43).
Glyburide was not associated with an increased risk of neonatal jaundice, obstetric trauma, or preterm birth, the investigators wrote (JAMA Pediatr. 2015 March 30 [doi:10.1001/jamapediatrics.2015.74]).
The study population included women (average age, 33.5 years) who delivered singleton live-born infants in 2000-2011 and who filed pharmacy claims for either glyburide (4,982 patients) or insulin (4,191 patients) during the 150 days preceding delivery.
The mean duration of treatment was 50.4 days with glyburide and 54.1 days with insulin.
The proportion of women treated with glyburide rose from 8.5% at the beginning of the study period to 64.4% at the end, wrote Wendy Camelo Castillo, Ph.D., of the department of pharmaceutical health services research, University of Maryland, Baltimore, and her associates.
Glyburide’s link to adverse outcomes “demands further attention” and suggests that women treated with the drug are not achieving adequate glucose control, the researchers wrote.
The Agency for Healthcare Research and Quality and the University of North Carolina at Chapel Hill supported the study. Dr. Camelo Castillo reported having no financial disclosures; her associates reported financial ties to varous pharmaceutical companies.
Key clinical point: The newborns of women with gestational diabetes treated with glyburide are at increased risk for adverse outcomes, compared with newborns of women treated with insulin.
Major finding: Compared with insulin, glyburide was associated with an increased risk in newborns of NICU admission (adjusted relative risk, 1.41), respiratory distress (aRR, 1.63), neonatal hypoglycemia (aRR, 1.40), birth injury (aRR, 1.35), and LGA status (aRR, 1.43).
Data source: A retrospective cohort study of pregnancy and neonatal outcomes in 9,173 mothers across the United States who received pharmacotherapy for gestational diabetes during a 12-year period.
Disclosures: The Agency for Healthcare Research and Quality and the University of North Carolina at Chapel Hill supported the study. Dr. Camelo Castillo reported having no financial disclosures; her associates reported financial ties to various pharmaceutical companies.
Clonidine maintenance therapy extends opioid abstinence
Adding clonidine to buprenorphine as maintenance therapy extends the duration of abstinence in heroin- or opioid-dependent adults participating in outpatient drug treatment, according to a report published in American Journal of Psychiatry.
“Although a new standard of care cannot be derived from the findings of just one clinical trial, we think our results provide support for a potential new off-label use for an old medication, expanding the addiction treatment armamentarium of opioid-agonist clinics, and perhaps also of psychiatrists and internists who are not prescribing opioid agonists,” said William J. Kowalczyk, Ph.D., of the Clinical Pharmacology and Therapeutics Research Branch, National Institute on Drug Abuse, Baltimore, and his associates.
Animal and laboratory studies have suggested that alpha-2 adrenoceptor agonists like clonidine might block stress-related drug-seeking behavior, independently of their action as adjunctive medications during opioid detoxification. To investigate this possibility, the researchers performed a double-blind randomized trial at a single Baltimore clinic in 118 patients aged 18-60 years.
After abstaining from opioids for 6 weeks, the study participants were randomly assigned to receive daily clonidine (61 patients) or matching placebo capsules (57 patients) in addition to sublingual buprenorphine as maintenance therapy for 8 weeks. They provided urine and breath samples under observation three times per week to test for opioids, cocaine, marijuana, amphetamines, barbiturates, benzodiazepines, and alcohol. They also used electronic diaries to report on stress, drug cravings, mood, and drug-related environmental cues at four randomly chosen times each day.
Compared with placebo, clonidine significantly increased the time to initial opioid lapse, lengthening the duration of consecutive days of abstinence to 34.8 days, compared with 25.5 days for placebo. Other studies have reported that the longer the duration of abstinence, the greater the likelihood that it will be sustained, the investigators said (Am. J. Psychiatry 2015;AiA:1-8 [doi:10.1176/appi.ajp.2014.14081014]).
Moreover, clonidine blunted the increased drug cravings that accompanied daily life stressors. “Participants in the clonidine group were less likely than those in the placebo group to report heroin craving at moderately high levels of stress, reaching the same likelihood of heroin craving only at the highest levels of stress. We saw no such buffering effect in self-reported exposure to drug-associated cues, suggesting that clonidine’s protection was specific,” Dr. Kowalczyk and his associates said.
“Clonidine is especially appealing because it carries no special prescribing requirements ... and is known to be well-tolerated in opioid users,” they wrote. In this study, the only symptom that occurred more frequently with clonidine than with placebo was dry mouth. Symptoms that were expected to cause more problems – sedation, hypotension, and dizziness – did not differ significantly between clonidine and placebo.
Among the limitations is that it was not possible to draw conclusions about the impact of clonidine maintenance “outside the context of buprenorphine maintenance,” the investigators said.
Still, for clinicians, the study findings identify a subpopulation of patients who would benefit most from clonidine maintenance therapy, described as “those who are susceptible to stress-induced lapses and those who have persistent though moderate stress.
“This is the sort of individualized approach to treatment that could greatly improve outcomes,” Dr. Kowalczyk and his associates added.
This study was supported by the NIDA and the National Institute of Mental Health. Dr. Kowalczyk and his associates reported having no financial relationships with commercial interests.
Adding clonidine to buprenorphine as maintenance therapy extends the duration of abstinence in heroin- or opioid-dependent adults participating in outpatient drug treatment, according to a report published in American Journal of Psychiatry.
“Although a new standard of care cannot be derived from the findings of just one clinical trial, we think our results provide support for a potential new off-label use for an old medication, expanding the addiction treatment armamentarium of opioid-agonist clinics, and perhaps also of psychiatrists and internists who are not prescribing opioid agonists,” said William J. Kowalczyk, Ph.D., of the Clinical Pharmacology and Therapeutics Research Branch, National Institute on Drug Abuse, Baltimore, and his associates.
Animal and laboratory studies have suggested that alpha-2 adrenoceptor agonists like clonidine might block stress-related drug-seeking behavior, independently of their action as adjunctive medications during opioid detoxification. To investigate this possibility, the researchers performed a double-blind randomized trial at a single Baltimore clinic in 118 patients aged 18-60 years.
After abstaining from opioids for 6 weeks, the study participants were randomly assigned to receive daily clonidine (61 patients) or matching placebo capsules (57 patients) in addition to sublingual buprenorphine as maintenance therapy for 8 weeks. They provided urine and breath samples under observation three times per week to test for opioids, cocaine, marijuana, amphetamines, barbiturates, benzodiazepines, and alcohol. They also used electronic diaries to report on stress, drug cravings, mood, and drug-related environmental cues at four randomly chosen times each day.
Compared with placebo, clonidine significantly increased the time to initial opioid lapse, lengthening the duration of consecutive days of abstinence to 34.8 days, compared with 25.5 days for placebo. Other studies have reported that the longer the duration of abstinence, the greater the likelihood that it will be sustained, the investigators said (Am. J. Psychiatry 2015;AiA:1-8 [doi:10.1176/appi.ajp.2014.14081014]).
Moreover, clonidine blunted the increased drug cravings that accompanied daily life stressors. “Participants in the clonidine group were less likely than those in the placebo group to report heroin craving at moderately high levels of stress, reaching the same likelihood of heroin craving only at the highest levels of stress. We saw no such buffering effect in self-reported exposure to drug-associated cues, suggesting that clonidine’s protection was specific,” Dr. Kowalczyk and his associates said.
“Clonidine is especially appealing because it carries no special prescribing requirements ... and is known to be well-tolerated in opioid users,” they wrote. In this study, the only symptom that occurred more frequently with clonidine than with placebo was dry mouth. Symptoms that were expected to cause more problems – sedation, hypotension, and dizziness – did not differ significantly between clonidine and placebo.
Among the limitations is that it was not possible to draw conclusions about the impact of clonidine maintenance “outside the context of buprenorphine maintenance,” the investigators said.
Still, for clinicians, the study findings identify a subpopulation of patients who would benefit most from clonidine maintenance therapy, described as “those who are susceptible to stress-induced lapses and those who have persistent though moderate stress.
“This is the sort of individualized approach to treatment that could greatly improve outcomes,” Dr. Kowalczyk and his associates added.
This study was supported by the NIDA and the National Institute of Mental Health. Dr. Kowalczyk and his associates reported having no financial relationships with commercial interests.
Adding clonidine to buprenorphine as maintenance therapy extends the duration of abstinence in heroin- or opioid-dependent adults participating in outpatient drug treatment, according to a report published in American Journal of Psychiatry.
“Although a new standard of care cannot be derived from the findings of just one clinical trial, we think our results provide support for a potential new off-label use for an old medication, expanding the addiction treatment armamentarium of opioid-agonist clinics, and perhaps also of psychiatrists and internists who are not prescribing opioid agonists,” said William J. Kowalczyk, Ph.D., of the Clinical Pharmacology and Therapeutics Research Branch, National Institute on Drug Abuse, Baltimore, and his associates.
Animal and laboratory studies have suggested that alpha-2 adrenoceptor agonists like clonidine might block stress-related drug-seeking behavior, independently of their action as adjunctive medications during opioid detoxification. To investigate this possibility, the researchers performed a double-blind randomized trial at a single Baltimore clinic in 118 patients aged 18-60 years.
After abstaining from opioids for 6 weeks, the study participants were randomly assigned to receive daily clonidine (61 patients) or matching placebo capsules (57 patients) in addition to sublingual buprenorphine as maintenance therapy for 8 weeks. They provided urine and breath samples under observation three times per week to test for opioids, cocaine, marijuana, amphetamines, barbiturates, benzodiazepines, and alcohol. They also used electronic diaries to report on stress, drug cravings, mood, and drug-related environmental cues at four randomly chosen times each day.
Compared with placebo, clonidine significantly increased the time to initial opioid lapse, lengthening the duration of consecutive days of abstinence to 34.8 days, compared with 25.5 days for placebo. Other studies have reported that the longer the duration of abstinence, the greater the likelihood that it will be sustained, the investigators said (Am. J. Psychiatry 2015;AiA:1-8 [doi:10.1176/appi.ajp.2014.14081014]).
Moreover, clonidine blunted the increased drug cravings that accompanied daily life stressors. “Participants in the clonidine group were less likely than those in the placebo group to report heroin craving at moderately high levels of stress, reaching the same likelihood of heroin craving only at the highest levels of stress. We saw no such buffering effect in self-reported exposure to drug-associated cues, suggesting that clonidine’s protection was specific,” Dr. Kowalczyk and his associates said.
“Clonidine is especially appealing because it carries no special prescribing requirements ... and is known to be well-tolerated in opioid users,” they wrote. In this study, the only symptom that occurred more frequently with clonidine than with placebo was dry mouth. Symptoms that were expected to cause more problems – sedation, hypotension, and dizziness – did not differ significantly between clonidine and placebo.
Among the limitations is that it was not possible to draw conclusions about the impact of clonidine maintenance “outside the context of buprenorphine maintenance,” the investigators said.
Still, for clinicians, the study findings identify a subpopulation of patients who would benefit most from clonidine maintenance therapy, described as “those who are susceptible to stress-induced lapses and those who have persistent though moderate stress.
“This is the sort of individualized approach to treatment that could greatly improve outcomes,” Dr. Kowalczyk and his associates added.
This study was supported by the NIDA and the National Institute of Mental Health. Dr. Kowalczyk and his associates reported having no financial relationships with commercial interests.
Key clinical point: Adding clonidine to buprenorphine as maintenance therapy extends the duration of opioid abstinence.
Major finding: Clonidine lengthened the duration of consecutive days of abstinence to 34.8 days, compared with 25.5 days for placebo.
Data source: A double-blind, placebo-controlled, randomized clinical trial involving 118 heroin- or opioid-dependent outpatients at a Baltimore drug treatment clinic.
Disclosures: This study was supported by the National Institute on Drug Abuse and the National Institute of Mental Health. Dr. Kowalczyk and his associates reported having no financial relationships with any commercial interests.
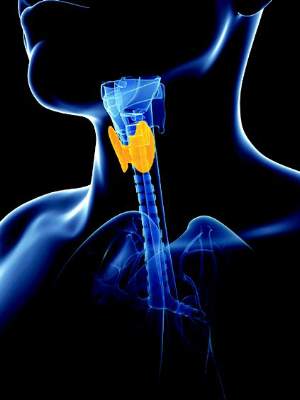
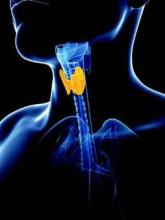
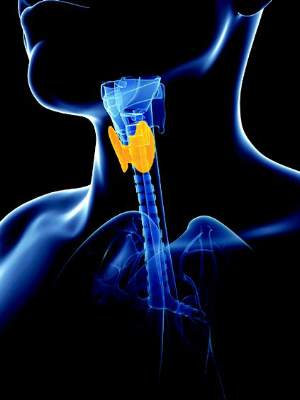
Evidence Inadequate to Support Routine Thyroid Screening
Current evidence is insufficient to assess the balance of benefits and harms of routinely screening nonpregnant, asymptomatic adults for thyroid dysfunction, according to a U.S. Preventive Services Task Force recommendation statement published online March 24 in the Annals of Internal Medicine.
In particular, there is not enough evidence to either support or refute the idea that such screening leads to clinically important benefits, such as reducing the incidence of cardiovascular disease and its related morbidity and mortality. Nor is there evidence that screening leads to early detection and treatment, which, in turn, improves quality of life or is beneficial for blood pressure, body mass index, bone mineral density, lipid levels, or cognitive function, said Dr. Michael L. LeFevre, chair of the task force and professor of family medicine at the University of Missouri–Columbia, and his associates.
On the other hand, the evidence concerning the harms of screening asymptomatic adults is also inadequate. It seems likely, but has not yet been proved, that screening large numbers of asymptomatic patients would result in frequent false-positive results and a “large degree” of overdiagnosis and overtreatment, given that only 3%-5% of adults have subclinical hypothyroidism and only 0.7% have subclinical hyperthyroidism. And the negative psychological effects of being labeled as chronically ill haven’t been well studied, either, the task force noted.
Regardless of this lack of evidence, it appears that many primary care providers already screen for thyroid dysfunction. Exact estimates are not available, but one brand of thyroid hormone is the most commonly prescribed drug in the country, and the number of filled prescriptions for levothyroxine rose 42% in a recent 5-year period. Given the high number of prescriptions and the low prevalence of overt thyroid dysfunction, “it is reasonable to conclude that many asymptomatic persons receive treatment. Clinicians seem to be treating more persons for thyroid dysfunction, at earlier times after initial diagnosis, and at TSH [thyroid-stimulating hormone] levels closer to normal,” Dr. LeFevre and his associates wrote (Ann. Intern. Med. 2015 March 24 [doi:10.7326/M15-0483]).
Continue for USPSTF recommendation statement >>
The USPSTF recommendation statement, an update of the one issued in 2004, uses a more limited definition of thyroid disease, restricting the term to denote only symptomatic “overt” hypothyroidism and hyperthyroidism (defined as persistently abnormal serum TSH and T4 levels with clearly associated clinical signs and symptoms that cannot be explained by another condition). The task force made this change to emphasize that screening can detect biochemical abnormalities as well as clinically important disease. However, “despite this change the USPSTF’s ultimate assessment is the same as in the previous recommendation,” the investigators added.
The USPSTF is an independent voluntary group of experts funded through congressional mandate by the Agency for Healthcare Research and Quality to make recommendations about the effectiveness of specific preventive care services for asymptomatic patients. Dr. LeFevre’s and his associates’ financial disclosures are available at www.uspreventiveservicestaskforce.org.
To obtain a copy of the recommendation statement, contact www.uspreventiveservicestaskforce.org.
Current evidence is insufficient to assess the balance of benefits and harms of routinely screening nonpregnant, asymptomatic adults for thyroid dysfunction, according to a U.S. Preventive Services Task Force recommendation statement published online March 24 in the Annals of Internal Medicine.
In particular, there is not enough evidence to either support or refute the idea that such screening leads to clinically important benefits, such as reducing the incidence of cardiovascular disease and its related morbidity and mortality. Nor is there evidence that screening leads to early detection and treatment, which, in turn, improves quality of life or is beneficial for blood pressure, body mass index, bone mineral density, lipid levels, or cognitive function, said Dr. Michael L. LeFevre, chair of the task force and professor of family medicine at the University of Missouri–Columbia, and his associates.
On the other hand, the evidence concerning the harms of screening asymptomatic adults is also inadequate. It seems likely, but has not yet been proved, that screening large numbers of asymptomatic patients would result in frequent false-positive results and a “large degree” of overdiagnosis and overtreatment, given that only 3%-5% of adults have subclinical hypothyroidism and only 0.7% have subclinical hyperthyroidism. And the negative psychological effects of being labeled as chronically ill haven’t been well studied, either, the task force noted.
Regardless of this lack of evidence, it appears that many primary care providers already screen for thyroid dysfunction. Exact estimates are not available, but one brand of thyroid hormone is the most commonly prescribed drug in the country, and the number of filled prescriptions for levothyroxine rose 42% in a recent 5-year period. Given the high number of prescriptions and the low prevalence of overt thyroid dysfunction, “it is reasonable to conclude that many asymptomatic persons receive treatment. Clinicians seem to be treating more persons for thyroid dysfunction, at earlier times after initial diagnosis, and at TSH [thyroid-stimulating hormone] levels closer to normal,” Dr. LeFevre and his associates wrote (Ann. Intern. Med. 2015 March 24 [doi:10.7326/M15-0483]).
Continue for USPSTF recommendation statement >>
The USPSTF recommendation statement, an update of the one issued in 2004, uses a more limited definition of thyroid disease, restricting the term to denote only symptomatic “overt” hypothyroidism and hyperthyroidism (defined as persistently abnormal serum TSH and T4 levels with clearly associated clinical signs and symptoms that cannot be explained by another condition). The task force made this change to emphasize that screening can detect biochemical abnormalities as well as clinically important disease. However, “despite this change the USPSTF’s ultimate assessment is the same as in the previous recommendation,” the investigators added.
The USPSTF is an independent voluntary group of experts funded through congressional mandate by the Agency for Healthcare Research and Quality to make recommendations about the effectiveness of specific preventive care services for asymptomatic patients. Dr. LeFevre’s and his associates’ financial disclosures are available at www.uspreventiveservicestaskforce.org.
To obtain a copy of the recommendation statement, contact www.uspreventiveservicestaskforce.org.
Current evidence is insufficient to assess the balance of benefits and harms of routinely screening nonpregnant, asymptomatic adults for thyroid dysfunction, according to a U.S. Preventive Services Task Force recommendation statement published online March 24 in the Annals of Internal Medicine.
In particular, there is not enough evidence to either support or refute the idea that such screening leads to clinically important benefits, such as reducing the incidence of cardiovascular disease and its related morbidity and mortality. Nor is there evidence that screening leads to early detection and treatment, which, in turn, improves quality of life or is beneficial for blood pressure, body mass index, bone mineral density, lipid levels, or cognitive function, said Dr. Michael L. LeFevre, chair of the task force and professor of family medicine at the University of Missouri–Columbia, and his associates.
On the other hand, the evidence concerning the harms of screening asymptomatic adults is also inadequate. It seems likely, but has not yet been proved, that screening large numbers of asymptomatic patients would result in frequent false-positive results and a “large degree” of overdiagnosis and overtreatment, given that only 3%-5% of adults have subclinical hypothyroidism and only 0.7% have subclinical hyperthyroidism. And the negative psychological effects of being labeled as chronically ill haven’t been well studied, either, the task force noted.
Regardless of this lack of evidence, it appears that many primary care providers already screen for thyroid dysfunction. Exact estimates are not available, but one brand of thyroid hormone is the most commonly prescribed drug in the country, and the number of filled prescriptions for levothyroxine rose 42% in a recent 5-year period. Given the high number of prescriptions and the low prevalence of overt thyroid dysfunction, “it is reasonable to conclude that many asymptomatic persons receive treatment. Clinicians seem to be treating more persons for thyroid dysfunction, at earlier times after initial diagnosis, and at TSH [thyroid-stimulating hormone] levels closer to normal,” Dr. LeFevre and his associates wrote (Ann. Intern. Med. 2015 March 24 [doi:10.7326/M15-0483]).
Continue for USPSTF recommendation statement >>
The USPSTF recommendation statement, an update of the one issued in 2004, uses a more limited definition of thyroid disease, restricting the term to denote only symptomatic “overt” hypothyroidism and hyperthyroidism (defined as persistently abnormal serum TSH and T4 levels with clearly associated clinical signs and symptoms that cannot be explained by another condition). The task force made this change to emphasize that screening can detect biochemical abnormalities as well as clinically important disease. However, “despite this change the USPSTF’s ultimate assessment is the same as in the previous recommendation,” the investigators added.
The USPSTF is an independent voluntary group of experts funded through congressional mandate by the Agency for Healthcare Research and Quality to make recommendations about the effectiveness of specific preventive care services for asymptomatic patients. Dr. LeFevre’s and his associates’ financial disclosures are available at www.uspreventiveservicestaskforce.org.
To obtain a copy of the recommendation statement, contact www.uspreventiveservicestaskforce.org.
Evidence inadequate to support routine thyroid screening
Current evidence is insufficient to assess the balance of benefits and harms of routinely screening nonpregnant, asymptomatic adults for thyroid dysfunction, according to a U.S. Preventive Services Task Force recommendation statement published online March 24 in the Annals of Internal Medicine.
In particular, there is not enough evidence to either support or refute the idea that such screening leads to clinically important benefits, such as reducing the incidence of cardiovascular disease and its related morbidity and mortality. Nor is there evidence that screening leads to early detection and treatment, which, in turn, improves quality of life or is beneficial for blood pressure, body mass index, bone mineral density, lipid levels, or cognitive function, said Dr. Michael L. LeFevre, chair of the task force and professor of family medicine at the University of Missouri–Columbia, and his associates.
On the other hand, the evidence concerning the harms of screening asymptomatic adults is also inadequate. It seems likely, but has not yet been proved, that screening large numbers of asymptomatic patients would result in frequent false-positive results and a “large degree” of overdiagnosis and overtreatment, given that only 3%-5% of adults have subclinical hypothyroidism and only 0.7% have subclinical hyperthyroidism. And the negative psychological effects of being labeled as chronically ill haven’t been well studied, either, the task force noted.
Regardless of this lack of evidence, it appears that many primary care providers already screen for thyroid dysfunction. Exact estimates are not available, but one brand of thyroid hormone is the most commonly prescribed drug in the country, and the number of filled prescriptions for levothyroxine rose 42% in a recent 5-year period. Given the high number of prescriptions and the low prevalence of overt thyroid dysfunction, “it is reasonable to conclude that many asymptomatic persons receive treatment. Clinicians seem to be treating more persons for thyroid dysfunction, at earlier times after initial diagnosis, and at TSH [thyroid-stimulating hormone] levels closer to normal,” Dr. LeFevre and his associates wrote (Ann. Intern. Med. 2015 March 24 [doi:10.7326/M15-0483]).
The USPSTF recommendation statement, an update of the one issued in 2004, uses a more limited definition of thyroid disease, restricting the term to denote only symptomatic “overt” hypothyroidism and hyperthyroidism (defined as persistently abnormal serum TSH and T4 levels with clearly associated clinical signs and symptoms that cannot be explained by another condition). The task force made this change to emphasize that screening can detect biochemical abnormalities as well as clinically important disease. However, “despite this change the USPSTF’s ultimate assessment is the same as in the previous recommendation,” the investigators added.
The USPSTF is an independent voluntary group of experts funded through congressional mandate by the Agency for Healthcare Research and Quality to make recommendations about the effectiveness of specific preventive care services for asymptomatic patients. Dr. LeFevre’s and his associates’ financial disclosures are available at www.uspreventiveservicestaskforce.org.
To obtain a copy of the recommendation statement, contact www.uspreventiveservicestaskforce.org.
Current evidence is insufficient to assess the balance of benefits and harms of routinely screening nonpregnant, asymptomatic adults for thyroid dysfunction, according to a U.S. Preventive Services Task Force recommendation statement published online March 24 in the Annals of Internal Medicine.
In particular, there is not enough evidence to either support or refute the idea that such screening leads to clinically important benefits, such as reducing the incidence of cardiovascular disease and its related morbidity and mortality. Nor is there evidence that screening leads to early detection and treatment, which, in turn, improves quality of life or is beneficial for blood pressure, body mass index, bone mineral density, lipid levels, or cognitive function, said Dr. Michael L. LeFevre, chair of the task force and professor of family medicine at the University of Missouri–Columbia, and his associates.
On the other hand, the evidence concerning the harms of screening asymptomatic adults is also inadequate. It seems likely, but has not yet been proved, that screening large numbers of asymptomatic patients would result in frequent false-positive results and a “large degree” of overdiagnosis and overtreatment, given that only 3%-5% of adults have subclinical hypothyroidism and only 0.7% have subclinical hyperthyroidism. And the negative psychological effects of being labeled as chronically ill haven’t been well studied, either, the task force noted.
Regardless of this lack of evidence, it appears that many primary care providers already screen for thyroid dysfunction. Exact estimates are not available, but one brand of thyroid hormone is the most commonly prescribed drug in the country, and the number of filled prescriptions for levothyroxine rose 42% in a recent 5-year period. Given the high number of prescriptions and the low prevalence of overt thyroid dysfunction, “it is reasonable to conclude that many asymptomatic persons receive treatment. Clinicians seem to be treating more persons for thyroid dysfunction, at earlier times after initial diagnosis, and at TSH [thyroid-stimulating hormone] levels closer to normal,” Dr. LeFevre and his associates wrote (Ann. Intern. Med. 2015 March 24 [doi:10.7326/M15-0483]).
The USPSTF recommendation statement, an update of the one issued in 2004, uses a more limited definition of thyroid disease, restricting the term to denote only symptomatic “overt” hypothyroidism and hyperthyroidism (defined as persistently abnormal serum TSH and T4 levels with clearly associated clinical signs and symptoms that cannot be explained by another condition). The task force made this change to emphasize that screening can detect biochemical abnormalities as well as clinically important disease. However, “despite this change the USPSTF’s ultimate assessment is the same as in the previous recommendation,” the investigators added.
The USPSTF is an independent voluntary group of experts funded through congressional mandate by the Agency for Healthcare Research and Quality to make recommendations about the effectiveness of specific preventive care services for asymptomatic patients. Dr. LeFevre’s and his associates’ financial disclosures are available at www.uspreventiveservicestaskforce.org.
To obtain a copy of the recommendation statement, contact www.uspreventiveservicestaskforce.org.
Current evidence is insufficient to assess the balance of benefits and harms of routinely screening nonpregnant, asymptomatic adults for thyroid dysfunction, according to a U.S. Preventive Services Task Force recommendation statement published online March 24 in the Annals of Internal Medicine.
In particular, there is not enough evidence to either support or refute the idea that such screening leads to clinically important benefits, such as reducing the incidence of cardiovascular disease and its related morbidity and mortality. Nor is there evidence that screening leads to early detection and treatment, which, in turn, improves quality of life or is beneficial for blood pressure, body mass index, bone mineral density, lipid levels, or cognitive function, said Dr. Michael L. LeFevre, chair of the task force and professor of family medicine at the University of Missouri–Columbia, and his associates.
On the other hand, the evidence concerning the harms of screening asymptomatic adults is also inadequate. It seems likely, but has not yet been proved, that screening large numbers of asymptomatic patients would result in frequent false-positive results and a “large degree” of overdiagnosis and overtreatment, given that only 3%-5% of adults have subclinical hypothyroidism and only 0.7% have subclinical hyperthyroidism. And the negative psychological effects of being labeled as chronically ill haven’t been well studied, either, the task force noted.
Regardless of this lack of evidence, it appears that many primary care providers already screen for thyroid dysfunction. Exact estimates are not available, but one brand of thyroid hormone is the most commonly prescribed drug in the country, and the number of filled prescriptions for levothyroxine rose 42% in a recent 5-year period. Given the high number of prescriptions and the low prevalence of overt thyroid dysfunction, “it is reasonable to conclude that many asymptomatic persons receive treatment. Clinicians seem to be treating more persons for thyroid dysfunction, at earlier times after initial diagnosis, and at TSH [thyroid-stimulating hormone] levels closer to normal,” Dr. LeFevre and his associates wrote (Ann. Intern. Med. 2015 March 24 [doi:10.7326/M15-0483]).
The USPSTF recommendation statement, an update of the one issued in 2004, uses a more limited definition of thyroid disease, restricting the term to denote only symptomatic “overt” hypothyroidism and hyperthyroidism (defined as persistently abnormal serum TSH and T4 levels with clearly associated clinical signs and symptoms that cannot be explained by another condition). The task force made this change to emphasize that screening can detect biochemical abnormalities as well as clinically important disease. However, “despite this change the USPSTF’s ultimate assessment is the same as in the previous recommendation,” the investigators added.
The USPSTF is an independent voluntary group of experts funded through congressional mandate by the Agency for Healthcare Research and Quality to make recommendations about the effectiveness of specific preventive care services for asymptomatic patients. Dr. LeFevre’s and his associates’ financial disclosures are available at www.uspreventiveservicestaskforce.org.
To obtain a copy of the recommendation statement, contact www.uspreventiveservicestaskforce.org.
Key clinical point: Current evidence is insufficient to assess the balance of benefits and harms of screening nonpregnant, asymptomatic adults for thyroid dysfunction.
Major finding: Only 3%-5% of adults in the United States have subclinical hypothyroidism, and only 0.7% have subclinical hyperthyroidism.
Data source: A comprehensive review of the current evidence and a compilation of recommendations regarding the benefits and harms of screening asymptomatic adults for thyroid dysfunction.
Disclosures: The USPSTF is an independent voluntary group of experts funded through congressional mandate by the Agency for Healthcare Research and Quality to make recommendations about the effectiveness of specific preventive care services for asymptomatic patients. Dr. LeFevre’s and his associates’ financial disclosures are available at www.uspreventiveservicestaskforce.org.
Survival advantage of intraperitoneal chemo persists beyond a decade
The survival advantage conferred by intraperitoneal chemotherapy over intravenous chemotherapy in advanced ovarian cancer persists beyond 10 years, according to a report published online March 23 in the Journal of Clinical Oncology.
Several clinical trials have demonstrated that compared with standard intravenous chemotherapy, intraperitoneal chemotherapy yields longer short-term survival. This benefit has been attributed to the much higher concentrations of active drug delivered into the peritoneal cavity with the latter approach, although this purported mechanism of action has never been proven. One previous study showed a 20-fold higher level of cisplatin in the peritoneal space with intraperitoneal than with intravenous administration, and another demonstrated a 1,000-fold higher concentration of paclitaxel in the peritoneal space with intraperitoneal than with intravenous administration, said Dr. Devansu Tewari of Kaiser Permanente Irvine (Calif.) Medical Center and his associates.
Nevertheless, intraperitoneal chemotherapy has not been widely adopted in the United States and is used only rarely in Europe. Clinicians’ hesitancy to use this approach has been attributed to its higher toxicity, inconvenience, and catheter-related complications, as well as to a lack of evidence regarding its long-term benefits.
To assess any long-term survival benefit, Dr. Tewari and his colleagues performed an exploratory, post hoc analysis of data pooled from two clinical trials first reported in 1996 and 2001 (Gynecologic Oncology Group protocols 114 and 172), which involved 876 women who had stage III epithelial ovarian or peritoneal carcinoma with no residual disease greater than 1 cm in diameter after surgery. The study participants were randomly assigned to receive either six cycles of intravenous paclitaxel plus intravenous cisplatin, or intravenous carboplatin plus intravenous paclitaxel plus intraperitoneal cisplatin in trial 114; those in trial 172 were randomly assigned to receive either intravenous paclitaxel plus intravenous cisplatin, or intraperitoneal cisplatin plus intraperitoneal paclitaxel for six cycles.
After a median follow-up of 10.7 years, the overall median survival was 61.8 months with intraperitoneal chemotherapy and 51.4 months with intravenous chemotherapy, a significant difference. Thus, intraperitoneal delivery was associated with a 23% decreased risk of death, with a hazard ratio of 0.77 after adjustment for patient age and performance status, tumor cell type and grade, and the presence or absence of residual disease, the investigators reported (J. Clin. Oncol. 2015 March 23 [doi:10.1200/JCO.2014.55.9898]).
Patients who completed all six cycles of intraperitoneal chemotherapy had longer survival than did those who completed only three. The risk of death decreased by 12% for each cycle of intraperitoneal chemotherapy completed, with an adjusted HR of 0.88. This indicates that clinicians should support patients through the entire intraperitoneal regimen, particularly if toxicities are not excessive, Dr. Tewari and his associates said.
In addition, the intraperitoneal approach conveyed a survival advantage in patients who had residual disease of 1 cm or less, as well as in those who had no visible residual disease.
If these findings are confirmed in future studies, more patients might be candidates for intraperitoneal chemotherapy, and more clinicians might adopt this approach in their practices, the researchers said.
This analysis, like the two Gynecologic Oncology Group trials on which it was based, was supported by the National Cancer Institute. Dr. Tewari reported having no financial disclosures; one of his associates reported receiving honoraria from Johnson & Johnson, Genentech, and AstraZeneca.
The role of intraperitoneal chemotherapy is one of the longest-running controversies in gynecologic oncology. One reason is that the chief rationale for intraperitoneal chemotherapy – that direct contact between tumor implants and peritoneal fluid containing high concentrations of cytotoxic agents is more effective than systemic intravenous therapy – has never been established. One might question why understanding the mechanism is important when the overall survival (OS) data are positive. However, IP therapy adds complexity, toxicity, and cost, particularly with the original GOG-0172 regimen, which includes cisplatin at 100 mg/m2 and paclitaxel over a 24-hour period. This has triggered several ad hoc modifications. None of these reasonable alternatives has been prospectively compared with the original regimen, and without a clear understanding of the underlying mechanism, it is difficult to predict which changes might be detrimental to OS. Our cumulative experience with intraperitoneal (IP) therapy has demonstrated sustained improvement in median OS (HR, 0.76) without an impact on long-term disease-specific mortality at the expense of increased complexity, toxicity, and cost. Similar gains in OS have been observed with other approaches, including intravenous therapy that incorporates dose-dense weekly paclitaxel, especially in high-risk patients with larger-volume residual disease. Important questions remain regarding patient selection (in terms of the extent of residual disease), mechanism of action, treatment modifications, timing, optimal number of IP cycles, and integration with targeted agents. Some of these questions will be addressed in the context of GOG-0252, and it remains to be seen whether better intravenous chemotherapy might match the outcomes previously associated with IP chemotherapy.
Dr. Michael A. Bookman is at U.S. Oncology Research in the Woodlands, Tex., and at Arizona Oncology in Tucson. He reported serving as a consultant or adviser to AstraZeneca, AbbVie, Sanofi, Boehringer Ingelheim, Genentech, Roche, Novartis, Immunogen, Endocyte, Gradalis, and NRG Oncology. Dr. Mark F. Brady is at the NRG Oncology Statistical and Data Center, Buffalo, N.Y. He reported ties to Endocyte, Genentech/Roche, and Advaxis. Dr. Bookman and Dr. Brady made these remarks in an editorial accompanying Dr. Tewari’s report (J. Clin. Oncol. 2015 March 23 [doi:10.1200/JCO.2014.60.2797]).
The role of intraperitoneal chemotherapy is one of the longest-running controversies in gynecologic oncology. One reason is that the chief rationale for intraperitoneal chemotherapy – that direct contact between tumor implants and peritoneal fluid containing high concentrations of cytotoxic agents is more effective than systemic intravenous therapy – has never been established. One might question why understanding the mechanism is important when the overall survival (OS) data are positive. However, IP therapy adds complexity, toxicity, and cost, particularly with the original GOG-0172 regimen, which includes cisplatin at 100 mg/m2 and paclitaxel over a 24-hour period. This has triggered several ad hoc modifications. None of these reasonable alternatives has been prospectively compared with the original regimen, and without a clear understanding of the underlying mechanism, it is difficult to predict which changes might be detrimental to OS. Our cumulative experience with intraperitoneal (IP) therapy has demonstrated sustained improvement in median OS (HR, 0.76) without an impact on long-term disease-specific mortality at the expense of increased complexity, toxicity, and cost. Similar gains in OS have been observed with other approaches, including intravenous therapy that incorporates dose-dense weekly paclitaxel, especially in high-risk patients with larger-volume residual disease. Important questions remain regarding patient selection (in terms of the extent of residual disease), mechanism of action, treatment modifications, timing, optimal number of IP cycles, and integration with targeted agents. Some of these questions will be addressed in the context of GOG-0252, and it remains to be seen whether better intravenous chemotherapy might match the outcomes previously associated with IP chemotherapy.
Dr. Michael A. Bookman is at U.S. Oncology Research in the Woodlands, Tex., and at Arizona Oncology in Tucson. He reported serving as a consultant or adviser to AstraZeneca, AbbVie, Sanofi, Boehringer Ingelheim, Genentech, Roche, Novartis, Immunogen, Endocyte, Gradalis, and NRG Oncology. Dr. Mark F. Brady is at the NRG Oncology Statistical and Data Center, Buffalo, N.Y. He reported ties to Endocyte, Genentech/Roche, and Advaxis. Dr. Bookman and Dr. Brady made these remarks in an editorial accompanying Dr. Tewari’s report (J. Clin. Oncol. 2015 March 23 [doi:10.1200/JCO.2014.60.2797]).
The role of intraperitoneal chemotherapy is one of the longest-running controversies in gynecologic oncology. One reason is that the chief rationale for intraperitoneal chemotherapy – that direct contact between tumor implants and peritoneal fluid containing high concentrations of cytotoxic agents is more effective than systemic intravenous therapy – has never been established. One might question why understanding the mechanism is important when the overall survival (OS) data are positive. However, IP therapy adds complexity, toxicity, and cost, particularly with the original GOG-0172 regimen, which includes cisplatin at 100 mg/m2 and paclitaxel over a 24-hour period. This has triggered several ad hoc modifications. None of these reasonable alternatives has been prospectively compared with the original regimen, and without a clear understanding of the underlying mechanism, it is difficult to predict which changes might be detrimental to OS. Our cumulative experience with intraperitoneal (IP) therapy has demonstrated sustained improvement in median OS (HR, 0.76) without an impact on long-term disease-specific mortality at the expense of increased complexity, toxicity, and cost. Similar gains in OS have been observed with other approaches, including intravenous therapy that incorporates dose-dense weekly paclitaxel, especially in high-risk patients with larger-volume residual disease. Important questions remain regarding patient selection (in terms of the extent of residual disease), mechanism of action, treatment modifications, timing, optimal number of IP cycles, and integration with targeted agents. Some of these questions will be addressed in the context of GOG-0252, and it remains to be seen whether better intravenous chemotherapy might match the outcomes previously associated with IP chemotherapy.
Dr. Michael A. Bookman is at U.S. Oncology Research in the Woodlands, Tex., and at Arizona Oncology in Tucson. He reported serving as a consultant or adviser to AstraZeneca, AbbVie, Sanofi, Boehringer Ingelheim, Genentech, Roche, Novartis, Immunogen, Endocyte, Gradalis, and NRG Oncology. Dr. Mark F. Brady is at the NRG Oncology Statistical and Data Center, Buffalo, N.Y. He reported ties to Endocyte, Genentech/Roche, and Advaxis. Dr. Bookman and Dr. Brady made these remarks in an editorial accompanying Dr. Tewari’s report (J. Clin. Oncol. 2015 March 23 [doi:10.1200/JCO.2014.60.2797]).
The survival advantage conferred by intraperitoneal chemotherapy over intravenous chemotherapy in advanced ovarian cancer persists beyond 10 years, according to a report published online March 23 in the Journal of Clinical Oncology.
Several clinical trials have demonstrated that compared with standard intravenous chemotherapy, intraperitoneal chemotherapy yields longer short-term survival. This benefit has been attributed to the much higher concentrations of active drug delivered into the peritoneal cavity with the latter approach, although this purported mechanism of action has never been proven. One previous study showed a 20-fold higher level of cisplatin in the peritoneal space with intraperitoneal than with intravenous administration, and another demonstrated a 1,000-fold higher concentration of paclitaxel in the peritoneal space with intraperitoneal than with intravenous administration, said Dr. Devansu Tewari of Kaiser Permanente Irvine (Calif.) Medical Center and his associates.
Nevertheless, intraperitoneal chemotherapy has not been widely adopted in the United States and is used only rarely in Europe. Clinicians’ hesitancy to use this approach has been attributed to its higher toxicity, inconvenience, and catheter-related complications, as well as to a lack of evidence regarding its long-term benefits.
To assess any long-term survival benefit, Dr. Tewari and his colleagues performed an exploratory, post hoc analysis of data pooled from two clinical trials first reported in 1996 and 2001 (Gynecologic Oncology Group protocols 114 and 172), which involved 876 women who had stage III epithelial ovarian or peritoneal carcinoma with no residual disease greater than 1 cm in diameter after surgery. The study participants were randomly assigned to receive either six cycles of intravenous paclitaxel plus intravenous cisplatin, or intravenous carboplatin plus intravenous paclitaxel plus intraperitoneal cisplatin in trial 114; those in trial 172 were randomly assigned to receive either intravenous paclitaxel plus intravenous cisplatin, or intraperitoneal cisplatin plus intraperitoneal paclitaxel for six cycles.
After a median follow-up of 10.7 years, the overall median survival was 61.8 months with intraperitoneal chemotherapy and 51.4 months with intravenous chemotherapy, a significant difference. Thus, intraperitoneal delivery was associated with a 23% decreased risk of death, with a hazard ratio of 0.77 after adjustment for patient age and performance status, tumor cell type and grade, and the presence or absence of residual disease, the investigators reported (J. Clin. Oncol. 2015 March 23 [doi:10.1200/JCO.2014.55.9898]).
Patients who completed all six cycles of intraperitoneal chemotherapy had longer survival than did those who completed only three. The risk of death decreased by 12% for each cycle of intraperitoneal chemotherapy completed, with an adjusted HR of 0.88. This indicates that clinicians should support patients through the entire intraperitoneal regimen, particularly if toxicities are not excessive, Dr. Tewari and his associates said.
In addition, the intraperitoneal approach conveyed a survival advantage in patients who had residual disease of 1 cm or less, as well as in those who had no visible residual disease.
If these findings are confirmed in future studies, more patients might be candidates for intraperitoneal chemotherapy, and more clinicians might adopt this approach in their practices, the researchers said.
This analysis, like the two Gynecologic Oncology Group trials on which it was based, was supported by the National Cancer Institute. Dr. Tewari reported having no financial disclosures; one of his associates reported receiving honoraria from Johnson & Johnson, Genentech, and AstraZeneca.
The survival advantage conferred by intraperitoneal chemotherapy over intravenous chemotherapy in advanced ovarian cancer persists beyond 10 years, according to a report published online March 23 in the Journal of Clinical Oncology.
Several clinical trials have demonstrated that compared with standard intravenous chemotherapy, intraperitoneal chemotherapy yields longer short-term survival. This benefit has been attributed to the much higher concentrations of active drug delivered into the peritoneal cavity with the latter approach, although this purported mechanism of action has never been proven. One previous study showed a 20-fold higher level of cisplatin in the peritoneal space with intraperitoneal than with intravenous administration, and another demonstrated a 1,000-fold higher concentration of paclitaxel in the peritoneal space with intraperitoneal than with intravenous administration, said Dr. Devansu Tewari of Kaiser Permanente Irvine (Calif.) Medical Center and his associates.
Nevertheless, intraperitoneal chemotherapy has not been widely adopted in the United States and is used only rarely in Europe. Clinicians’ hesitancy to use this approach has been attributed to its higher toxicity, inconvenience, and catheter-related complications, as well as to a lack of evidence regarding its long-term benefits.
To assess any long-term survival benefit, Dr. Tewari and his colleagues performed an exploratory, post hoc analysis of data pooled from two clinical trials first reported in 1996 and 2001 (Gynecologic Oncology Group protocols 114 and 172), which involved 876 women who had stage III epithelial ovarian or peritoneal carcinoma with no residual disease greater than 1 cm in diameter after surgery. The study participants were randomly assigned to receive either six cycles of intravenous paclitaxel plus intravenous cisplatin, or intravenous carboplatin plus intravenous paclitaxel plus intraperitoneal cisplatin in trial 114; those in trial 172 were randomly assigned to receive either intravenous paclitaxel plus intravenous cisplatin, or intraperitoneal cisplatin plus intraperitoneal paclitaxel for six cycles.
After a median follow-up of 10.7 years, the overall median survival was 61.8 months with intraperitoneal chemotherapy and 51.4 months with intravenous chemotherapy, a significant difference. Thus, intraperitoneal delivery was associated with a 23% decreased risk of death, with a hazard ratio of 0.77 after adjustment for patient age and performance status, tumor cell type and grade, and the presence or absence of residual disease, the investigators reported (J. Clin. Oncol. 2015 March 23 [doi:10.1200/JCO.2014.55.9898]).
Patients who completed all six cycles of intraperitoneal chemotherapy had longer survival than did those who completed only three. The risk of death decreased by 12% for each cycle of intraperitoneal chemotherapy completed, with an adjusted HR of 0.88. This indicates that clinicians should support patients through the entire intraperitoneal regimen, particularly if toxicities are not excessive, Dr. Tewari and his associates said.
In addition, the intraperitoneal approach conveyed a survival advantage in patients who had residual disease of 1 cm or less, as well as in those who had no visible residual disease.
If these findings are confirmed in future studies, more patients might be candidates for intraperitoneal chemotherapy, and more clinicians might adopt this approach in their practices, the researchers said.
This analysis, like the two Gynecologic Oncology Group trials on which it was based, was supported by the National Cancer Institute. Dr. Tewari reported having no financial disclosures; one of his associates reported receiving honoraria from Johnson & Johnson, Genentech, and AstraZeneca.
Key clinical point: The survival advantage of intraperitoneal chemotherapy over intravenous chemotherapy for advanced ovarian cancer persists beyond 10 years.
Major finding: After a median follow-up of 10.7 years, the overall median survival was 61.8 months with intraperitoneal chemotherapy and 51.4 months with intravenous chemotherapy.
Data source: An exploratory, post hoc analysis of data pooled from two phase III clinical trials comparing intraperitoneal vs. intravenous chemotherapy in 876 women with stage 3 epithelial ovarian cancer.
Disclosures: This analysis, like the two Gynecologic Oncology Group trials on which it was based, was supported by the National Cancer Institute. Dr. Tewari reported having no financial disclosures; one of his associates reported receiving honoraria from Johnson & Johnson, Genentech, and AstraZeneca.
Mongersen induces 55%-65% remission rates in Crohn’s
Mongersen, an oral SMAD7 antisense oligonucleotide formulated to deliver its active ingredient primarily into the lumen of the terminal ileum and right colon, induced remission rates as high as 55%-65% in a small, brief, manufacturer-sponsored, phase II clinical trial, according to a report published online March 19 in the New England Journal of Medicine.
In Crohn’s disease, gut inflammation is characterized by abnormal reductions in a particular immunosuppressive cytokine caused by increased levels of SMAD7. Mongersen (formerly GED0301) downregulates SMAD7 using a classic antisense mechanism, which in turn restores the proper cytokine function and suppresses inflammation, said Dr. Giovanni Monteleone of the department of systems medicine, University of Tor Vergata, Rome, and his associates.
They assessed a 2-week course of mongersen in 166 adults with active, moderate to severe Crohn’s disease who were treated and followed for approximately 3 months at 17 medical centers in Italy and Germany. The study participants were randomly assigned to receive one of three doses of the agent or a matching placebo in a double-blind fashion. The study’s primary endpoint was the percentage of patients in remission at day 15 who remained in remission for at least 2 more weeks. Remission was defined as a Crohn’s Disease Activity Index (CDAI) score of < 150.
Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo. Thus, remission rates at the two highest doses of mongersen exceeded those achieved in other phase II trials for Crohn’s therapies, which ranged from 16% to 48%, the investigators said (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMoa1407250]). Rates of attaining the secondary endpoint of “clinical response,” defined as a decrease of 100 or more points in the CDAI score at day 28, also were significantly higher at the two highest doses of mongersen – 72% and 58% – than with the lowest dose (37%) or with placebo (17%).
No safety issues related to mongersen were identified in this study, but a 2-week course of treatment in such a small group of patients likely is not adequate to determine safety. Adverse events occurred in 65% of the active-treatment groups and 64% of the placebo group and were mostly mild. The nine serious adverse events that occurred were unrelated to study treatment, Dr. Monteleone and his associates said.
Further study is needed to assess longer durations of treatment, and to judge the effectiveness of the drug on the basis of endoscopic analyses of mucosal healing rather than on CDAI score. It also will be important to determine whether higher doses or longer treatment courses of mongersen raise the risk of fibrosis, given that the targeted cytokine plays a profibrogenic role in many organs, they added.
AGA Note
Review the AGA IBD Clinical Service Line to learn more about managing Crohn’s Disease.
The clinical response reported by Monteleone et al. is impressive, but it was not confirmed by endoscopic evidence of mucosal healing and it did not correlate with normalization of biomarkers such has fecal calprotectin or C-reactive protein. In short, there is a lack of congruence between clinical remission and biologic remission, an issue that must be addressed in future studies of this agent.
Also intriguing was the finding that clinical response was maintained for the duration of follow-up even though mongersen was only administered for 2 weeks and is thought not to linger in tissues. This is a stark contrast to the rapid recurrence of symptoms that characterizes withdrawal of existing anti-inflammatory drugs.
Severine Vermeire, M.D., Ph.D., is in the department of gastroenterology at Leuven (Belgium) University Hospital. She reported receiving grant support and personal fees from AbbVie, Merck Sharp & Dohme, Pfizer, Genentech/Roche, Takeda, and Mundipharma. Dr. Vermeire made these remarks in an editorial accompanying Dr. Monteleone’s report (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMe1415053]).
The clinical response reported by Monteleone et al. is impressive, but it was not confirmed by endoscopic evidence of mucosal healing and it did not correlate with normalization of biomarkers such has fecal calprotectin or C-reactive protein. In short, there is a lack of congruence between clinical remission and biologic remission, an issue that must be addressed in future studies of this agent.
Also intriguing was the finding that clinical response was maintained for the duration of follow-up even though mongersen was only administered for 2 weeks and is thought not to linger in tissues. This is a stark contrast to the rapid recurrence of symptoms that characterizes withdrawal of existing anti-inflammatory drugs.
Severine Vermeire, M.D., Ph.D., is in the department of gastroenterology at Leuven (Belgium) University Hospital. She reported receiving grant support and personal fees from AbbVie, Merck Sharp & Dohme, Pfizer, Genentech/Roche, Takeda, and Mundipharma. Dr. Vermeire made these remarks in an editorial accompanying Dr. Monteleone’s report (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMe1415053]).
The clinical response reported by Monteleone et al. is impressive, but it was not confirmed by endoscopic evidence of mucosal healing and it did not correlate with normalization of biomarkers such has fecal calprotectin or C-reactive protein. In short, there is a lack of congruence between clinical remission and biologic remission, an issue that must be addressed in future studies of this agent.
Also intriguing was the finding that clinical response was maintained for the duration of follow-up even though mongersen was only administered for 2 weeks and is thought not to linger in tissues. This is a stark contrast to the rapid recurrence of symptoms that characterizes withdrawal of existing anti-inflammatory drugs.
Severine Vermeire, M.D., Ph.D., is in the department of gastroenterology at Leuven (Belgium) University Hospital. She reported receiving grant support and personal fees from AbbVie, Merck Sharp & Dohme, Pfizer, Genentech/Roche, Takeda, and Mundipharma. Dr. Vermeire made these remarks in an editorial accompanying Dr. Monteleone’s report (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMe1415053]).
Mongersen, an oral SMAD7 antisense oligonucleotide formulated to deliver its active ingredient primarily into the lumen of the terminal ileum and right colon, induced remission rates as high as 55%-65% in a small, brief, manufacturer-sponsored, phase II clinical trial, according to a report published online March 19 in the New England Journal of Medicine.
In Crohn’s disease, gut inflammation is characterized by abnormal reductions in a particular immunosuppressive cytokine caused by increased levels of SMAD7. Mongersen (formerly GED0301) downregulates SMAD7 using a classic antisense mechanism, which in turn restores the proper cytokine function and suppresses inflammation, said Dr. Giovanni Monteleone of the department of systems medicine, University of Tor Vergata, Rome, and his associates.
They assessed a 2-week course of mongersen in 166 adults with active, moderate to severe Crohn’s disease who were treated and followed for approximately 3 months at 17 medical centers in Italy and Germany. The study participants were randomly assigned to receive one of three doses of the agent or a matching placebo in a double-blind fashion. The study’s primary endpoint was the percentage of patients in remission at day 15 who remained in remission for at least 2 more weeks. Remission was defined as a Crohn’s Disease Activity Index (CDAI) score of < 150.
Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo. Thus, remission rates at the two highest doses of mongersen exceeded those achieved in other phase II trials for Crohn’s therapies, which ranged from 16% to 48%, the investigators said (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMoa1407250]). Rates of attaining the secondary endpoint of “clinical response,” defined as a decrease of 100 or more points in the CDAI score at day 28, also were significantly higher at the two highest doses of mongersen – 72% and 58% – than with the lowest dose (37%) or with placebo (17%).
No safety issues related to mongersen were identified in this study, but a 2-week course of treatment in such a small group of patients likely is not adequate to determine safety. Adverse events occurred in 65% of the active-treatment groups and 64% of the placebo group and were mostly mild. The nine serious adverse events that occurred were unrelated to study treatment, Dr. Monteleone and his associates said.
Further study is needed to assess longer durations of treatment, and to judge the effectiveness of the drug on the basis of endoscopic analyses of mucosal healing rather than on CDAI score. It also will be important to determine whether higher doses or longer treatment courses of mongersen raise the risk of fibrosis, given that the targeted cytokine plays a profibrogenic role in many organs, they added.
AGA Note
Review the AGA IBD Clinical Service Line to learn more about managing Crohn’s Disease.
Mongersen, an oral SMAD7 antisense oligonucleotide formulated to deliver its active ingredient primarily into the lumen of the terminal ileum and right colon, induced remission rates as high as 55%-65% in a small, brief, manufacturer-sponsored, phase II clinical trial, according to a report published online March 19 in the New England Journal of Medicine.
In Crohn’s disease, gut inflammation is characterized by abnormal reductions in a particular immunosuppressive cytokine caused by increased levels of SMAD7. Mongersen (formerly GED0301) downregulates SMAD7 using a classic antisense mechanism, which in turn restores the proper cytokine function and suppresses inflammation, said Dr. Giovanni Monteleone of the department of systems medicine, University of Tor Vergata, Rome, and his associates.
They assessed a 2-week course of mongersen in 166 adults with active, moderate to severe Crohn’s disease who were treated and followed for approximately 3 months at 17 medical centers in Italy and Germany. The study participants were randomly assigned to receive one of three doses of the agent or a matching placebo in a double-blind fashion. The study’s primary endpoint was the percentage of patients in remission at day 15 who remained in remission for at least 2 more weeks. Remission was defined as a Crohn’s Disease Activity Index (CDAI) score of < 150.
Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo. Thus, remission rates at the two highest doses of mongersen exceeded those achieved in other phase II trials for Crohn’s therapies, which ranged from 16% to 48%, the investigators said (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMoa1407250]). Rates of attaining the secondary endpoint of “clinical response,” defined as a decrease of 100 or more points in the CDAI score at day 28, also were significantly higher at the two highest doses of mongersen – 72% and 58% – than with the lowest dose (37%) or with placebo (17%).
No safety issues related to mongersen were identified in this study, but a 2-week course of treatment in such a small group of patients likely is not adequate to determine safety. Adverse events occurred in 65% of the active-treatment groups and 64% of the placebo group and were mostly mild. The nine serious adverse events that occurred were unrelated to study treatment, Dr. Monteleone and his associates said.
Further study is needed to assess longer durations of treatment, and to judge the effectiveness of the drug on the basis of endoscopic analyses of mucosal healing rather than on CDAI score. It also will be important to determine whether higher doses or longer treatment courses of mongersen raise the risk of fibrosis, given that the targeted cytokine plays a profibrogenic role in many organs, they added.
AGA Note
Review the AGA IBD Clinical Service Line to learn more about managing Crohn’s Disease.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Mongersen, an oral SMAD7 antisense oligonucleotide, induced remission rates as high as 55%-65% in a small 2-week phase II clinical trial.
Major finding: Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo.
Data source: A randomized placebo-controlled double-blind phase II clinical trial involving 166 adults at 17 medical centers in Italy and Germany.
Disclosures: This study was sponsored by Giuliani, acting under contract to Nogra Pharma. Dr. Monteleone reported ties to Giuliani, Novo Nordisk, Teva, Sitris, Lycera, Sofar, and Zambon, and holds a patent related to the use of SMAD7 antisense oligonucleotides in Crohn’s disease. His associates reported financial ties to numerous industry sources.
Mongersen induces 55%-65% remission rates in Crohn’s
Mongersen, an oral SMAD7 antisense oligonucleotide formulated to deliver its active ingredient primarily into the lumen of the terminal ileum and right colon, induced remission rates as high as 55%-65% in a small, brief, manufacturer-sponsored, phase II clinical trial, according to a report published online March 19 in the New England Journal of Medicine.
In Crohn’s disease, gut inflammation is characterized by abnormal reductions in a particular immunosuppressive cytokine caused by increased levels of SMAD7. Mongersen (formerly GED0301) downregulates SMAD7 using a classic antisense mechanism, which in turn restores the proper cytokine function and suppresses inflammation, said Dr. Giovanni Monteleone of the department of systems medicine, University of Tor Vergata, Rome, and his associates.
They assessed a 2-week course of mongersen in 166 adults with active, moderate to severe Crohn’s disease who were treated and followed for approximately 3 months at 17 medical centers in Italy and Germany. The study participants were randomly assigned to receive one of three doses of the agent or a matching placebo in a double-blind fashion. The study’s primary endpoint was the percentage of patients in remission at day 15 who remained in remission for at least 2 more weeks. Remission was defined as a Crohn’s Disease Activity Index (CDAI) score of < 150.
Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo. Thus, remission rates at the two highest doses of mongersen exceeded those achieved in other phase II trials for Crohn’s therapies, which ranged from 16% to 48%, the investigators said (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMoa1407250]). Rates of attaining the secondary endpoint of “clinical response,” defined as a decrease of 100 or more points in the CDAI score at day 28, also were significantly higher at the two highest doses of mongersen – 72% and 58% – than with the lowest dose (37%) or with placebo (17%).
No safety issues related to mongersen were identified in this study, but a 2-week course of treatment in such a small group of patients likely is not adequate to determine safety. Adverse events occurred in 65% of the active-treatment groups and 64% of the placebo group and were mostly mild. The nine serious adverse events that occurred were unrelated to study treatment, Dr. Monteleone and his associates said.
Further study is needed to assess longer durations of treatment, and to judge the effectiveness of the drug on the basis of endoscopic analyses of mucosal healing rather than on CDAI score. It also will be important to determine whether higher doses or longer treatment courses of mongersen raise the risk of fibrosis, given that the targeted cytokine plays a profibrogenic role in many organs, they added.
The clinical response reported by Monteleone et al. is impressive, but it was not confirmed by endoscopic evidence of mucosal healing and it did not correlate with normalization of biomarkers such has fecal calprotectin or C-reactive protein. In short, there is a lack of congruence between clinical remission and biologic remission, an issue that must be addressed in future studies of this agent.
Also intriguing was the finding that clinical response was maintained for the duration of follow-up even though mongersen was only administered for 2 weeks and is thought not to linger in tissues. This is a stark contrast to the rapid recurrence of symptoms that characterizes withdrawal of existing anti-inflammatory drugs.
Severine Vermeire, M.D., Ph.D., is in the department of gastroenterology at Leuven (Belgium) University Hospital. She reported receiving grant support and personal fees from AbbVie, Merck Sharp & Dohme, Pfizer, Genentech/Roche, Takeda, and Mundipharma. Dr. Vermeire made these remarks in an editorial accompanying Dr. Monteleone’s report (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMe1415053]).
The clinical response reported by Monteleone et al. is impressive, but it was not confirmed by endoscopic evidence of mucosal healing and it did not correlate with normalization of biomarkers such has fecal calprotectin or C-reactive protein. In short, there is a lack of congruence between clinical remission and biologic remission, an issue that must be addressed in future studies of this agent.
Also intriguing was the finding that clinical response was maintained for the duration of follow-up even though mongersen was only administered for 2 weeks and is thought not to linger in tissues. This is a stark contrast to the rapid recurrence of symptoms that characterizes withdrawal of existing anti-inflammatory drugs.
Severine Vermeire, M.D., Ph.D., is in the department of gastroenterology at Leuven (Belgium) University Hospital. She reported receiving grant support and personal fees from AbbVie, Merck Sharp & Dohme, Pfizer, Genentech/Roche, Takeda, and Mundipharma. Dr. Vermeire made these remarks in an editorial accompanying Dr. Monteleone’s report (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMe1415053]).
The clinical response reported by Monteleone et al. is impressive, but it was not confirmed by endoscopic evidence of mucosal healing and it did not correlate with normalization of biomarkers such has fecal calprotectin or C-reactive protein. In short, there is a lack of congruence between clinical remission and biologic remission, an issue that must be addressed in future studies of this agent.
Also intriguing was the finding that clinical response was maintained for the duration of follow-up even though mongersen was only administered for 2 weeks and is thought not to linger in tissues. This is a stark contrast to the rapid recurrence of symptoms that characterizes withdrawal of existing anti-inflammatory drugs.
Severine Vermeire, M.D., Ph.D., is in the department of gastroenterology at Leuven (Belgium) University Hospital. She reported receiving grant support and personal fees from AbbVie, Merck Sharp & Dohme, Pfizer, Genentech/Roche, Takeda, and Mundipharma. Dr. Vermeire made these remarks in an editorial accompanying Dr. Monteleone’s report (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMe1415053]).
Mongersen, an oral SMAD7 antisense oligonucleotide formulated to deliver its active ingredient primarily into the lumen of the terminal ileum and right colon, induced remission rates as high as 55%-65% in a small, brief, manufacturer-sponsored, phase II clinical trial, according to a report published online March 19 in the New England Journal of Medicine.
In Crohn’s disease, gut inflammation is characterized by abnormal reductions in a particular immunosuppressive cytokine caused by increased levels of SMAD7. Mongersen (formerly GED0301) downregulates SMAD7 using a classic antisense mechanism, which in turn restores the proper cytokine function and suppresses inflammation, said Dr. Giovanni Monteleone of the department of systems medicine, University of Tor Vergata, Rome, and his associates.
They assessed a 2-week course of mongersen in 166 adults with active, moderate to severe Crohn’s disease who were treated and followed for approximately 3 months at 17 medical centers in Italy and Germany. The study participants were randomly assigned to receive one of three doses of the agent or a matching placebo in a double-blind fashion. The study’s primary endpoint was the percentage of patients in remission at day 15 who remained in remission for at least 2 more weeks. Remission was defined as a Crohn’s Disease Activity Index (CDAI) score of < 150.
Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo. Thus, remission rates at the two highest doses of mongersen exceeded those achieved in other phase II trials for Crohn’s therapies, which ranged from 16% to 48%, the investigators said (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMoa1407250]). Rates of attaining the secondary endpoint of “clinical response,” defined as a decrease of 100 or more points in the CDAI score at day 28, also were significantly higher at the two highest doses of mongersen – 72% and 58% – than with the lowest dose (37%) or with placebo (17%).
No safety issues related to mongersen were identified in this study, but a 2-week course of treatment in such a small group of patients likely is not adequate to determine safety. Adverse events occurred in 65% of the active-treatment groups and 64% of the placebo group and were mostly mild. The nine serious adverse events that occurred were unrelated to study treatment, Dr. Monteleone and his associates said.
Further study is needed to assess longer durations of treatment, and to judge the effectiveness of the drug on the basis of endoscopic analyses of mucosal healing rather than on CDAI score. It also will be important to determine whether higher doses or longer treatment courses of mongersen raise the risk of fibrosis, given that the targeted cytokine plays a profibrogenic role in many organs, they added.
Mongersen, an oral SMAD7 antisense oligonucleotide formulated to deliver its active ingredient primarily into the lumen of the terminal ileum and right colon, induced remission rates as high as 55%-65% in a small, brief, manufacturer-sponsored, phase II clinical trial, according to a report published online March 19 in the New England Journal of Medicine.
In Crohn’s disease, gut inflammation is characterized by abnormal reductions in a particular immunosuppressive cytokine caused by increased levels of SMAD7. Mongersen (formerly GED0301) downregulates SMAD7 using a classic antisense mechanism, which in turn restores the proper cytokine function and suppresses inflammation, said Dr. Giovanni Monteleone of the department of systems medicine, University of Tor Vergata, Rome, and his associates.
They assessed a 2-week course of mongersen in 166 adults with active, moderate to severe Crohn’s disease who were treated and followed for approximately 3 months at 17 medical centers in Italy and Germany. The study participants were randomly assigned to receive one of three doses of the agent or a matching placebo in a double-blind fashion. The study’s primary endpoint was the percentage of patients in remission at day 15 who remained in remission for at least 2 more weeks. Remission was defined as a Crohn’s Disease Activity Index (CDAI) score of < 150.
Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo. Thus, remission rates at the two highest doses of mongersen exceeded those achieved in other phase II trials for Crohn’s therapies, which ranged from 16% to 48%, the investigators said (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMoa1407250]). Rates of attaining the secondary endpoint of “clinical response,” defined as a decrease of 100 or more points in the CDAI score at day 28, also were significantly higher at the two highest doses of mongersen – 72% and 58% – than with the lowest dose (37%) or with placebo (17%).
No safety issues related to mongersen were identified in this study, but a 2-week course of treatment in such a small group of patients likely is not adequate to determine safety. Adverse events occurred in 65% of the active-treatment groups and 64% of the placebo group and were mostly mild. The nine serious adverse events that occurred were unrelated to study treatment, Dr. Monteleone and his associates said.
Further study is needed to assess longer durations of treatment, and to judge the effectiveness of the drug on the basis of endoscopic analyses of mucosal healing rather than on CDAI score. It also will be important to determine whether higher doses or longer treatment courses of mongersen raise the risk of fibrosis, given that the targeted cytokine plays a profibrogenic role in many organs, they added.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Mongersen, an oral SMAD7 antisense oligonucleotide, induced remission rates as high as 55%-65% in a small 2-week phase II clinical trial.
Major finding: Rates of remission were 65% in the 43 participants who received 160 mg of mongersen, 55% in the 40 who received 40 mg, 12% in the 41 who received 10 mg, and 10% in the 42 who received placebo.
Data source: A randomized placebo-controlled double-blind phase II clinical trial involving 166 adults at 17 medical centers in Italy and Germany.
Disclosures: This study was sponsored by Giuliani, acting under contract to Nogra Pharma. Dr. Monteleone reported ties to Giuliani, Novo Nordisk, Teva, Sitris, Lycera, Sofar, and Zambon, and holds a patent related to the use of SMAD7 antisense oligonucleotides in Crohn’s disease. His associates reported financial ties to numerous industry sources.
PCV13 prevents pneumococcal pneumonia in elderly
The 13-valent polysaccharide conjugate vaccine showed “significant efficacy” against vaccine-type strains of community-acquired pneumonia and vaccine-type invasive pneumococcal disease among the elderly in a large clinical trial in the Netherlands, according to a report published online March 19 in the New England Journal of Medicine.
The PCV13 vaccine’s efficacy in adults aged 65 years and older has never been determined until now. It became possible to assess efficacy in this age group when a serotype-specific urinary antigen detection assay was developed, which can identify Streptococcus pneumonia polysaccharides in the urine of patients suspected of having pneumonia rather than requiring isolation of the organism in culture, said Dr. Marc J. M. Bonten of Julius Center for Health Sciences and Primary Care, University Medical Center Utrecht (the Netherlands), and his associates.
They used the assay to assess PCV13 efficacy in 84,496 older adults who were randomly assigned to receive active vaccine (42,240 participants) or placebo (42,256) and followed for a mean of 4 years in the trial, which was sponsored by Pfizer. In the per-protocol analysis, vaccine efficacy was 46% for preventing a first episode of vaccine-type community-acquired pneumonia, 45% for preventing nonbacteremic and noninvasive vaccine-type community-acquired pneumonia, and 75% for preventing vaccine-type invasive pneumococcal disease.
Evidence of the vaccine’s efficacy became apparent shortly after vaccination occurred and persisted throughout the duration of the study. There was no evidence of any safety concerns in the patients who received the active vaccine, wrote Dr. Bonten and his associates (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMoa1408544]).
The PCV13 vaccine did not show efficacy in preventing death from any cause, but the number of deaths associated with pneumococcal disease in this study was too small to allow a meaningful analysis of this outcome, the investigators noted.
The 13-valent polysaccharide conjugate vaccine showed “significant efficacy” against vaccine-type strains of community-acquired pneumonia and vaccine-type invasive pneumococcal disease among the elderly in a large clinical trial in the Netherlands, according to a report published online March 19 in the New England Journal of Medicine.
The PCV13 vaccine’s efficacy in adults aged 65 years and older has never been determined until now. It became possible to assess efficacy in this age group when a serotype-specific urinary antigen detection assay was developed, which can identify Streptococcus pneumonia polysaccharides in the urine of patients suspected of having pneumonia rather than requiring isolation of the organism in culture, said Dr. Marc J. M. Bonten of Julius Center for Health Sciences and Primary Care, University Medical Center Utrecht (the Netherlands), and his associates.
They used the assay to assess PCV13 efficacy in 84,496 older adults who were randomly assigned to receive active vaccine (42,240 participants) or placebo (42,256) and followed for a mean of 4 years in the trial, which was sponsored by Pfizer. In the per-protocol analysis, vaccine efficacy was 46% for preventing a first episode of vaccine-type community-acquired pneumonia, 45% for preventing nonbacteremic and noninvasive vaccine-type community-acquired pneumonia, and 75% for preventing vaccine-type invasive pneumococcal disease.
Evidence of the vaccine’s efficacy became apparent shortly after vaccination occurred and persisted throughout the duration of the study. There was no evidence of any safety concerns in the patients who received the active vaccine, wrote Dr. Bonten and his associates (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMoa1408544]).
The PCV13 vaccine did not show efficacy in preventing death from any cause, but the number of deaths associated with pneumococcal disease in this study was too small to allow a meaningful analysis of this outcome, the investigators noted.
The 13-valent polysaccharide conjugate vaccine showed “significant efficacy” against vaccine-type strains of community-acquired pneumonia and vaccine-type invasive pneumococcal disease among the elderly in a large clinical trial in the Netherlands, according to a report published online March 19 in the New England Journal of Medicine.
The PCV13 vaccine’s efficacy in adults aged 65 years and older has never been determined until now. It became possible to assess efficacy in this age group when a serotype-specific urinary antigen detection assay was developed, which can identify Streptococcus pneumonia polysaccharides in the urine of patients suspected of having pneumonia rather than requiring isolation of the organism in culture, said Dr. Marc J. M. Bonten of Julius Center for Health Sciences and Primary Care, University Medical Center Utrecht (the Netherlands), and his associates.
They used the assay to assess PCV13 efficacy in 84,496 older adults who were randomly assigned to receive active vaccine (42,240 participants) or placebo (42,256) and followed for a mean of 4 years in the trial, which was sponsored by Pfizer. In the per-protocol analysis, vaccine efficacy was 46% for preventing a first episode of vaccine-type community-acquired pneumonia, 45% for preventing nonbacteremic and noninvasive vaccine-type community-acquired pneumonia, and 75% for preventing vaccine-type invasive pneumococcal disease.
Evidence of the vaccine’s efficacy became apparent shortly after vaccination occurred and persisted throughout the duration of the study. There was no evidence of any safety concerns in the patients who received the active vaccine, wrote Dr. Bonten and his associates (N. Engl. J. Med. 2015 March 19 [doi:10.1056/NEJMoa1408544]).
The PCV13 vaccine did not show efficacy in preventing death from any cause, but the number of deaths associated with pneumococcal disease in this study was too small to allow a meaningful analysis of this outcome, the investigators noted.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: The 13-valent pneumococcal polysaccharide conjugate vaccine is effective against community-acquired pneumococcal pneumonia in adults aged 65 years and older.
Major finding: Vaccine efficacy was 46% for preventing a first episode of vaccine-type strains of community-acquired pneumonia, 45% for preventing nonbacteremic and noninvasive vaccine-type strains of community-acquired pneumonia, and 75% for preventing vaccine-type strains of invasive pneumococcal disease.
Data source: An industry-sponsored randomized placebo-controlled double-blind trial involving 84,496 Dutch adults aged 65 years and older followed for a mean of 4 years after vaccination.
Disclosures: Pfizer sponsored the study. Dr. Bonten and several associates reported ties to Pfizer, and his associates also reported ties to GlaxoSmithKline, Roche, and Novartis.
Clindamycin, TMP-SMX are equally effective for skin infections
Clindamycin and trimethoprim-sulfamethoxazole are similarly safe and effective for treating uncomplicated skin infections, including both cellulitis and abscesses, in ambulatory settings in regions where MRSA is endemic, according to a report published online March 19 in the New England Journal of Medicine.
The data comparing these two agents in an ambulatory setting are limited, though both are commonly recommended as empirical therapy for skin infections in patients who present to clinics and emergency departments and have only minor or no coexisting conditions, said Dr. Loren G. Miller of the Los Angeles Biomedical Research Institute and the division of infectious diseases at Harbor-UCLA Medical Center, and his associates.
They performed a prospective double-blind randomized trial comparing clindamycin against TMP-SMX in 524 ethnically diverse adults and children who presented as outpatients with uncomplicated skin infections during a 2-year period in Chicago, San Francisco, Los Angeles, and Nashville – areas in which community-associated MRSA is endemic. The mean patient age was 27 years, and approximately 30% were pediatric patients. All the participants had cellulitis without abscesses (including erysipelas), one or more abscesses larger than 5 cm in diameter, or both conditions. A total of 264 were randomized to clindamycin and 260 to TMP-SMX daily for 10 days.
Cure rates did not differ significantly between the two study groups. At 7-10 days after completing therapy, the rates of cure in the intention-to-treat population were 80.3% for clindamycin and 77.7% for TMP-SMX, and in the evaluable population the rates were 89.5% and 88.2%, respectively.
At 1 month follow-up, the cure rates in the evaluable population were 83.9% for clindamycin and 78.2% for TMP-SMX, the investigators said (N. Engl. J. Med. 2015;372:1093-103 [doi:10.1056/NEJMoa1403789]). Rates of adverse events were nearly identical between the two study groups (18.9% vs. 18.6%), and most were mild and resolved without sequelae. There were no treatment-associated serious adverse events, and the rates of treatment discontinuation were very similar between patients receiving clindamycin (8.3%) and those receiving TMP-SMX (8.8%).
The study was supported by grants from the National Institutes of Allergy and Infectious Diseases. Dr. Miller reported receiving consulting fees from Cubist, Durata, and Pfizer; his associates reported ties to Cubist, Pfizer, EMMES, Theravance, AstraZeneca, Trius, Merck, and Cerexa.
“Given the immense importance of MRSA infections during the past 20 years, prospective clinical trial data to inform the choice of outpatient treatment of skin infections are surprisingly sparse,” according to Dr. Michael R. Wessels. The findings of Miller et al. reassure that outcomes are good for most such patients when they are treated with either of the two most popular agents, he noted in an accompanying editorial.
But carefully designed clinical trials like this one are still needed to determine which therapies are most effective and safe for cellulitis and skin abscesses in patients who are more severely ill, such as those who have high fever or lymphangitis, as well as for patients who have coexisting conditions such as diabetes, cancer, or obesity, Dr. Wessels wrote.
Dr. Michael R. Wessels is a member of the division of infectious diseases at Boston Children’s Hospital and in the department of pediatrics at Harvard Medical School, Boston. He reported having no financial disclosures. Dr. Wessels made these remarks in an editorial accompanying Dr. Miller’s report (N. Engl. J. Med. 2015;372:1164-65 [doi:10.1056/NEJMe1500331]).
“Given the immense importance of MRSA infections during the past 20 years, prospective clinical trial data to inform the choice of outpatient treatment of skin infections are surprisingly sparse,” according to Dr. Michael R. Wessels. The findings of Miller et al. reassure that outcomes are good for most such patients when they are treated with either of the two most popular agents, he noted in an accompanying editorial.
But carefully designed clinical trials like this one are still needed to determine which therapies are most effective and safe for cellulitis and skin abscesses in patients who are more severely ill, such as those who have high fever or lymphangitis, as well as for patients who have coexisting conditions such as diabetes, cancer, or obesity, Dr. Wessels wrote.
Dr. Michael R. Wessels is a member of the division of infectious diseases at Boston Children’s Hospital and in the department of pediatrics at Harvard Medical School, Boston. He reported having no financial disclosures. Dr. Wessels made these remarks in an editorial accompanying Dr. Miller’s report (N. Engl. J. Med. 2015;372:1164-65 [doi:10.1056/NEJMe1500331]).
“Given the immense importance of MRSA infections during the past 20 years, prospective clinical trial data to inform the choice of outpatient treatment of skin infections are surprisingly sparse,” according to Dr. Michael R. Wessels. The findings of Miller et al. reassure that outcomes are good for most such patients when they are treated with either of the two most popular agents, he noted in an accompanying editorial.
But carefully designed clinical trials like this one are still needed to determine which therapies are most effective and safe for cellulitis and skin abscesses in patients who are more severely ill, such as those who have high fever or lymphangitis, as well as for patients who have coexisting conditions such as diabetes, cancer, or obesity, Dr. Wessels wrote.
Dr. Michael R. Wessels is a member of the division of infectious diseases at Boston Children’s Hospital and in the department of pediatrics at Harvard Medical School, Boston. He reported having no financial disclosures. Dr. Wessels made these remarks in an editorial accompanying Dr. Miller’s report (N. Engl. J. Med. 2015;372:1164-65 [doi:10.1056/NEJMe1500331]).
Clindamycin and trimethoprim-sulfamethoxazole are similarly safe and effective for treating uncomplicated skin infections, including both cellulitis and abscesses, in ambulatory settings in regions where MRSA is endemic, according to a report published online March 19 in the New England Journal of Medicine.
The data comparing these two agents in an ambulatory setting are limited, though both are commonly recommended as empirical therapy for skin infections in patients who present to clinics and emergency departments and have only minor or no coexisting conditions, said Dr. Loren G. Miller of the Los Angeles Biomedical Research Institute and the division of infectious diseases at Harbor-UCLA Medical Center, and his associates.
They performed a prospective double-blind randomized trial comparing clindamycin against TMP-SMX in 524 ethnically diverse adults and children who presented as outpatients with uncomplicated skin infections during a 2-year period in Chicago, San Francisco, Los Angeles, and Nashville – areas in which community-associated MRSA is endemic. The mean patient age was 27 years, and approximately 30% were pediatric patients. All the participants had cellulitis without abscesses (including erysipelas), one or more abscesses larger than 5 cm in diameter, or both conditions. A total of 264 were randomized to clindamycin and 260 to TMP-SMX daily for 10 days.
Cure rates did not differ significantly between the two study groups. At 7-10 days after completing therapy, the rates of cure in the intention-to-treat population were 80.3% for clindamycin and 77.7% for TMP-SMX, and in the evaluable population the rates were 89.5% and 88.2%, respectively.
At 1 month follow-up, the cure rates in the evaluable population were 83.9% for clindamycin and 78.2% for TMP-SMX, the investigators said (N. Engl. J. Med. 2015;372:1093-103 [doi:10.1056/NEJMoa1403789]). Rates of adverse events were nearly identical between the two study groups (18.9% vs. 18.6%), and most were mild and resolved without sequelae. There were no treatment-associated serious adverse events, and the rates of treatment discontinuation were very similar between patients receiving clindamycin (8.3%) and those receiving TMP-SMX (8.8%).
The study was supported by grants from the National Institutes of Allergy and Infectious Diseases. Dr. Miller reported receiving consulting fees from Cubist, Durata, and Pfizer; his associates reported ties to Cubist, Pfizer, EMMES, Theravance, AstraZeneca, Trius, Merck, and Cerexa.
Clindamycin and trimethoprim-sulfamethoxazole are similarly safe and effective for treating uncomplicated skin infections, including both cellulitis and abscesses, in ambulatory settings in regions where MRSA is endemic, according to a report published online March 19 in the New England Journal of Medicine.
The data comparing these two agents in an ambulatory setting are limited, though both are commonly recommended as empirical therapy for skin infections in patients who present to clinics and emergency departments and have only minor or no coexisting conditions, said Dr. Loren G. Miller of the Los Angeles Biomedical Research Institute and the division of infectious diseases at Harbor-UCLA Medical Center, and his associates.
They performed a prospective double-blind randomized trial comparing clindamycin against TMP-SMX in 524 ethnically diverse adults and children who presented as outpatients with uncomplicated skin infections during a 2-year period in Chicago, San Francisco, Los Angeles, and Nashville – areas in which community-associated MRSA is endemic. The mean patient age was 27 years, and approximately 30% were pediatric patients. All the participants had cellulitis without abscesses (including erysipelas), one or more abscesses larger than 5 cm in diameter, or both conditions. A total of 264 were randomized to clindamycin and 260 to TMP-SMX daily for 10 days.
Cure rates did not differ significantly between the two study groups. At 7-10 days after completing therapy, the rates of cure in the intention-to-treat population were 80.3% for clindamycin and 77.7% for TMP-SMX, and in the evaluable population the rates were 89.5% and 88.2%, respectively.
At 1 month follow-up, the cure rates in the evaluable population were 83.9% for clindamycin and 78.2% for TMP-SMX, the investigators said (N. Engl. J. Med. 2015;372:1093-103 [doi:10.1056/NEJMoa1403789]). Rates of adverse events were nearly identical between the two study groups (18.9% vs. 18.6%), and most were mild and resolved without sequelae. There were no treatment-associated serious adverse events, and the rates of treatment discontinuation were very similar between patients receiving clindamycin (8.3%) and those receiving TMP-SMX (8.8%).
The study was supported by grants from the National Institutes of Allergy and Infectious Diseases. Dr. Miller reported receiving consulting fees from Cubist, Durata, and Pfizer; his associates reported ties to Cubist, Pfizer, EMMES, Theravance, AstraZeneca, Trius, Merck, and Cerexa.
Key clinical point: Clindamycin and TMP-SMX had similar efficacy and side-effect profiles for treating uncomplicated skin infections, including both abscesses and cellulitis.
Major finding: At 7-10 days after completing therapy, the rates of cure in the evaluable population were 89.5% with clindamycin and 88.2% with TMP-SMX.
Data source: A prospective, multicenter, randomized, double-blind clinical trial involving 524 adults and children followed for 1 month after treatment.
Disclosures: This trial was supported by the National Institutes of Allergy and Infectious Diseases and the National Center for Advancing Translational Sciences (NCT00730028). Dr. Miller reported receiving consulting fees from Cubist, Durata, and Pfizer; his associates reported ties to Cubist, Pfizer, EMMES, Theravance, AstraZeneca, Trius, Merck, and Cerexa.