User login
NSAIDs’ colon cancer protection tied to three genetic variants
The well-documented protective effect that aspirin and nonsteroidal anti-inflammatory drugs exert against colon cancer has been linked to three genetic variations at chromosome 12 and 15, according to a report published online March 17 in JAMA.
In a genome-wide investigation of the interrelationship between genetic markers and the regular use of aspirin/NSAIDs, researchers found that the drugs were differentially associated with colorectal cancer risk according to variations at single-nucleotide polymorphisms (SNPs) on chromosome 12p12.3 (rs2965667 and rs10505806) and chromosome 15q25.2 (rs16973225).
These findings “can help to identify population subgroups defined by genetic background that may preferentially benefit from chemopreventive use of these agents and offer novel insights into underlying mechanisms of carcinogenesis,” said Dr. Hongmei Nan of the department of epidemiology at Indiana University and the Bren Simon Cancer Center, both in Indianapolis, and her associates.
To obtain a large population for the genome-wide study, the investigators pooled data from 10 observational studies conducted between 1976 and 2003 in Australia, Canada, Germany, and the United States, which enrolled 8,634 colon cancer cases and 8,553 control subjects matched for age, sex, and race/ethnicity.
Compared with nonuse of aspirin, NSAIDs, or both, regular use was associated with a lower risk of colorectal cancer: The overall prevalence of the disease was 28% among regular users, compared with 38% among nonusers, for an odds ratio of 0.69. The regular use of aspirin alone also was associated with a lower risk of colorectal cancer: The prevalence was 24% among regular users and 31% among nonusers, for an odds ratio of 0.71.
This protective effect, however, and thus an NSAID user’s risk for colorectal cancer, varied by whether patients carried the genetic variants and how many copies of the risk alleles they carried, Dr. Nan and her associates said (JAMA 2015;313:1133-42).All three genetic variants are located near genes known to be involved in the pathogenesis of colon cancer, including genes that regulate the production of proinflammatory prostaglandins and interleukins, particularly within the gut. This proximity supports the idea that aspirin and NSAIDs exert their gut-protective effects through these inflammatory mediators, the investigators said.
The National Cancer Institute and the National Institutes of Health supported the study. Dr. Nan reported having no financial disclosures. One of her associates reported holding a patent for aspirin as a colorectal chemopreventive agent and two others reported ties to Arctic Dx, Bayer Healthcare, Pfizer, and Pozen.
These findings add complexity to, but do not answer, the longstanding clinical question of whether healthy adults should take aspirin regularly to reduce their risk of colon cancer.
In the not-too-distant future, it will become affordable and practical to conduct genetic testing routinely in healthy people. At that time, primary care clinicians will need to understand genetic risks and to have informed, clear, literacy-adjusted, culturally competent discussions with their patients about how to use this information.
Dr. Richard C. Wender is with the American Cancer Society, Atlanta, and the department of family and community medicine at Thomas Jefferson University, Philadelphia. He reported having no financial disclosures. Dr. Wender made these remarks in an editorial accompanying Dr. Nan’s report (JAMA 2015;313:1111-2).
These findings add complexity to, but do not answer, the longstanding clinical question of whether healthy adults should take aspirin regularly to reduce their risk of colon cancer.
In the not-too-distant future, it will become affordable and practical to conduct genetic testing routinely in healthy people. At that time, primary care clinicians will need to understand genetic risks and to have informed, clear, literacy-adjusted, culturally competent discussions with their patients about how to use this information.
Dr. Richard C. Wender is with the American Cancer Society, Atlanta, and the department of family and community medicine at Thomas Jefferson University, Philadelphia. He reported having no financial disclosures. Dr. Wender made these remarks in an editorial accompanying Dr. Nan’s report (JAMA 2015;313:1111-2).
These findings add complexity to, but do not answer, the longstanding clinical question of whether healthy adults should take aspirin regularly to reduce their risk of colon cancer.
In the not-too-distant future, it will become affordable and practical to conduct genetic testing routinely in healthy people. At that time, primary care clinicians will need to understand genetic risks and to have informed, clear, literacy-adjusted, culturally competent discussions with their patients about how to use this information.
Dr. Richard C. Wender is with the American Cancer Society, Atlanta, and the department of family and community medicine at Thomas Jefferson University, Philadelphia. He reported having no financial disclosures. Dr. Wender made these remarks in an editorial accompanying Dr. Nan’s report (JAMA 2015;313:1111-2).
The well-documented protective effect that aspirin and nonsteroidal anti-inflammatory drugs exert against colon cancer has been linked to three genetic variations at chromosome 12 and 15, according to a report published online March 17 in JAMA.
In a genome-wide investigation of the interrelationship between genetic markers and the regular use of aspirin/NSAIDs, researchers found that the drugs were differentially associated with colorectal cancer risk according to variations at single-nucleotide polymorphisms (SNPs) on chromosome 12p12.3 (rs2965667 and rs10505806) and chromosome 15q25.2 (rs16973225).
These findings “can help to identify population subgroups defined by genetic background that may preferentially benefit from chemopreventive use of these agents and offer novel insights into underlying mechanisms of carcinogenesis,” said Dr. Hongmei Nan of the department of epidemiology at Indiana University and the Bren Simon Cancer Center, both in Indianapolis, and her associates.
To obtain a large population for the genome-wide study, the investigators pooled data from 10 observational studies conducted between 1976 and 2003 in Australia, Canada, Germany, and the United States, which enrolled 8,634 colon cancer cases and 8,553 control subjects matched for age, sex, and race/ethnicity.
Compared with nonuse of aspirin, NSAIDs, or both, regular use was associated with a lower risk of colorectal cancer: The overall prevalence of the disease was 28% among regular users, compared with 38% among nonusers, for an odds ratio of 0.69. The regular use of aspirin alone also was associated with a lower risk of colorectal cancer: The prevalence was 24% among regular users and 31% among nonusers, for an odds ratio of 0.71.
This protective effect, however, and thus an NSAID user’s risk for colorectal cancer, varied by whether patients carried the genetic variants and how many copies of the risk alleles they carried, Dr. Nan and her associates said (JAMA 2015;313:1133-42).All three genetic variants are located near genes known to be involved in the pathogenesis of colon cancer, including genes that regulate the production of proinflammatory prostaglandins and interleukins, particularly within the gut. This proximity supports the idea that aspirin and NSAIDs exert their gut-protective effects through these inflammatory mediators, the investigators said.
The National Cancer Institute and the National Institutes of Health supported the study. Dr. Nan reported having no financial disclosures. One of her associates reported holding a patent for aspirin as a colorectal chemopreventive agent and two others reported ties to Arctic Dx, Bayer Healthcare, Pfizer, and Pozen.
The well-documented protective effect that aspirin and nonsteroidal anti-inflammatory drugs exert against colon cancer has been linked to three genetic variations at chromosome 12 and 15, according to a report published online March 17 in JAMA.
In a genome-wide investigation of the interrelationship between genetic markers and the regular use of aspirin/NSAIDs, researchers found that the drugs were differentially associated with colorectal cancer risk according to variations at single-nucleotide polymorphisms (SNPs) on chromosome 12p12.3 (rs2965667 and rs10505806) and chromosome 15q25.2 (rs16973225).
These findings “can help to identify population subgroups defined by genetic background that may preferentially benefit from chemopreventive use of these agents and offer novel insights into underlying mechanisms of carcinogenesis,” said Dr. Hongmei Nan of the department of epidemiology at Indiana University and the Bren Simon Cancer Center, both in Indianapolis, and her associates.
To obtain a large population for the genome-wide study, the investigators pooled data from 10 observational studies conducted between 1976 and 2003 in Australia, Canada, Germany, and the United States, which enrolled 8,634 colon cancer cases and 8,553 control subjects matched for age, sex, and race/ethnicity.
Compared with nonuse of aspirin, NSAIDs, or both, regular use was associated with a lower risk of colorectal cancer: The overall prevalence of the disease was 28% among regular users, compared with 38% among nonusers, for an odds ratio of 0.69. The regular use of aspirin alone also was associated with a lower risk of colorectal cancer: The prevalence was 24% among regular users and 31% among nonusers, for an odds ratio of 0.71.
This protective effect, however, and thus an NSAID user’s risk for colorectal cancer, varied by whether patients carried the genetic variants and how many copies of the risk alleles they carried, Dr. Nan and her associates said (JAMA 2015;313:1133-42).All three genetic variants are located near genes known to be involved in the pathogenesis of colon cancer, including genes that regulate the production of proinflammatory prostaglandins and interleukins, particularly within the gut. This proximity supports the idea that aspirin and NSAIDs exert their gut-protective effects through these inflammatory mediators, the investigators said.
The National Cancer Institute and the National Institutes of Health supported the study. Dr. Nan reported having no financial disclosures. One of her associates reported holding a patent for aspirin as a colorectal chemopreventive agent and two others reported ties to Arctic Dx, Bayer Healthcare, Pfizer, and Pozen.
FROM JAMA
Key clinical point: The protective effect of aspirin and NSAIDs against colon cancer was linked to three genetic variants on chromosomes 12 and 15.
Major finding: The prevalence of colon cancer was 28% among regular users of aspirin/NSAIDs, compared with 38% among nonusers, for an odds ratio of 0.69.
Data source: A genome-wide analysis of gene and environment interactions using data from 17,187 participants in 10 case-control studies conducted over a 40-year period in Australia, Canada, Germany, and the United States.
Disclosures: The National Cancer Institute and the National Institutes of Health supported the study. Dr. Nan reported having no financial disclosures. One of her associates reported holding a patent for aspirin as a colorectal chemopreventive agent and two others reported ties to Arctic Dx, Bayer Healthcare, Pfizer, and Pozen.
Novel HCV Therapies Found Cost Effective, With Caveats
Two different statistical models found that novel therapies for chronic HCV infection, particularly the combination of sofosbuvir and ledipasvir, are cost effective in most patients, according to separate reports published online March 17 in Annals of Internal Medicine.
However, both groups of researchers cautioned that if these expensive agents are made available to the millions of eligible patients across the country, it would have an immense impact on health care costs for both public and private payers.
The novel therapies, which typically contain sofosbuvir in combination with ledipasvir, simeprevir, or daclatasvir, substantially reduce the length of treatment, achieve much higher rates of sustained viral response (SVR), and offer interferon-free alternatives for patients who can’t tolerate or don’t respond to standard interferon-based treatments. But it is unclear whether these benefits justify their profound expense, compared with current care. Both statistical models were developed to examine this issue, but from different perspectives.
In one study, funded primarily by the National Institutes of Health, investigators constructed a model that simulated 120 possible clinical courses of HCV-infected adults based on different ages and sexes, treatment histories, HCV genotypes, fibrosis scores, and interferon tolerances. For each of these patient profiles, they ran simulations in which patients received either “the old standard of care” (peginterferon and ribavirin, either with or without boceprevir and telaprevir) or sofosbuvir plus ledipasvir.
The average per-patient cost of standard care ranged from $15,000 to $71,000, depending on the patient profile, while that of sofosbuvir-ledipasvir ranged from $66,000 to $154,000, said Jagpreet Chhatwal, Ph.D., of the University of Texas MD Anderson Cancer Center, Houston, and his associates.
Compared with standard care, treating 10,000 patients with sofosbuvir-ledipasvir was projected to prevent 600 cases of decompensated cirrhosis, 310 cases of hepatocellular carcinoma (HCC), 60 liver transplantations, and 550 liver-related deaths, which would result in substantial cost savings. Also, compared with standard care, the incremental cost-effectiveness ratio of sofosbuvir-ledipasvir was $55,400 per additional quality-of-life-year (QALY) gained, which falls well within the accepted range for therapies for other medical conditions. Thus, the new therapy proved to be cost-effective for most HCV patients (Ann. Intern. Med. 2015 March 17 [doi:10.7326/M14-1336]). But there was an important caveat: Many more patients would be eligible for the novel therapies than for the standard care, because the novel therapies are much more easily tolerated. With the addition of so many eligible patients, the resources needed to treat them “could be immense and unsustainable.” Compared with standard care, giving these novel HCV therapies to all eligible patients “would cost an additional $65 billion in the next 5 years,” which would not be counterbalanced by the estimated $16 billion saved by preventing cirrhosis, HCC, and transplantations.
Therefore, “despite the cost-effectiveness of [novel] HCV treatments, our analysis shows that it is unaffordable at the current price,” Dr. Chhatwal and his associates said.
In the other study, funded primarily by CVS Health, researchers developed a discrete-event simulation model of the natural history and progression of liver disease in treatment-naive patients, categorized by whether they were infected with HCV genotype 1, 2, or 3. Several possible treatment regimens were considered for each genotype, and the SVR rates they were projected to attain were derived from those reported in clinical trials, said Mehdi Najafzadeh, Ph.D., of the division of pharmacoepidemiology and pharmacoeconomics, Brigham and Women’s Hospital and Harvard Medical School, Boston, and his associates.
“From a societal perspective, the newly approved PEG-free regimen of sofosbuvir-ledipasvir for 12 weeks could be very cost-effective relative to usual care (costing $12,825/QALY gained) for patients with HCV genotype 1.” This treatment proved to be the optimal strategy in the greatest number of simulations involving genotype 1.
Similarly, for genotype 3 the combination of sofosbuvir plus ledipasvir plus ribavirin for 12 weeks cost $73,000/QALY gained, compared with usual care. This also represents “relatively good value.” However, for genotype 2 the most cost-effective novel therapy, sofosbuvir-ribavirin, was $110,168/QALY gained, which is not considered cost-effective, Dr. Najafzadeh and his associates said (Ann. Intern. Med. 2015 March 17 [doi:10.7326;M14-1152]). Again, an important caveat to these findings was that, at their current prices, the cost of these drugs were not outweighed by the savings that accrued from preventing the complications of HCV. And “regardless of the cost-effectiveness of novel HCV treatments, there is considerable concern that their very high prices could substantially increase short-term overall drug spending for many public and private payers,” the investigators noted.
However, the fact that these regimens don’t reduce health care costs “is an exceptionally high bar” to hold them to – one that “is generally not expected when evaluating whether a new strategy represents good value for the money,” they added.
Two different statistical models found that novel therapies for chronic HCV infection, particularly the combination of sofosbuvir and ledipasvir, are cost effective in most patients, according to separate reports published online March 17 in Annals of Internal Medicine.
However, both groups of researchers cautioned that if these expensive agents are made available to the millions of eligible patients across the country, it would have an immense impact on health care costs for both public and private payers.
The novel therapies, which typically contain sofosbuvir in combination with ledipasvir, simeprevir, or daclatasvir, substantially reduce the length of treatment, achieve much higher rates of sustained viral response (SVR), and offer interferon-free alternatives for patients who can’t tolerate or don’t respond to standard interferon-based treatments. But it is unclear whether these benefits justify their profound expense, compared with current care. Both statistical models were developed to examine this issue, but from different perspectives.
In one study, funded primarily by the National Institutes of Health, investigators constructed a model that simulated 120 possible clinical courses of HCV-infected adults based on different ages and sexes, treatment histories, HCV genotypes, fibrosis scores, and interferon tolerances. For each of these patient profiles, they ran simulations in which patients received either “the old standard of care” (peginterferon and ribavirin, either with or without boceprevir and telaprevir) or sofosbuvir plus ledipasvir.
The average per-patient cost of standard care ranged from $15,000 to $71,000, depending on the patient profile, while that of sofosbuvir-ledipasvir ranged from $66,000 to $154,000, said Jagpreet Chhatwal, Ph.D., of the University of Texas MD Anderson Cancer Center, Houston, and his associates.
Compared with standard care, treating 10,000 patients with sofosbuvir-ledipasvir was projected to prevent 600 cases of decompensated cirrhosis, 310 cases of hepatocellular carcinoma (HCC), 60 liver transplantations, and 550 liver-related deaths, which would result in substantial cost savings. Also, compared with standard care, the incremental cost-effectiveness ratio of sofosbuvir-ledipasvir was $55,400 per additional quality-of-life-year (QALY) gained, which falls well within the accepted range for therapies for other medical conditions. Thus, the new therapy proved to be cost-effective for most HCV patients (Ann. Intern. Med. 2015 March 17 [doi:10.7326/M14-1336]). But there was an important caveat: Many more patients would be eligible for the novel therapies than for the standard care, because the novel therapies are much more easily tolerated. With the addition of so many eligible patients, the resources needed to treat them “could be immense and unsustainable.” Compared with standard care, giving these novel HCV therapies to all eligible patients “would cost an additional $65 billion in the next 5 years,” which would not be counterbalanced by the estimated $16 billion saved by preventing cirrhosis, HCC, and transplantations.
Therefore, “despite the cost-effectiveness of [novel] HCV treatments, our analysis shows that it is unaffordable at the current price,” Dr. Chhatwal and his associates said.
In the other study, funded primarily by CVS Health, researchers developed a discrete-event simulation model of the natural history and progression of liver disease in treatment-naive patients, categorized by whether they were infected with HCV genotype 1, 2, or 3. Several possible treatment regimens were considered for each genotype, and the SVR rates they were projected to attain were derived from those reported in clinical trials, said Mehdi Najafzadeh, Ph.D., of the division of pharmacoepidemiology and pharmacoeconomics, Brigham and Women’s Hospital and Harvard Medical School, Boston, and his associates.
“From a societal perspective, the newly approved PEG-free regimen of sofosbuvir-ledipasvir for 12 weeks could be very cost-effective relative to usual care (costing $12,825/QALY gained) for patients with HCV genotype 1.” This treatment proved to be the optimal strategy in the greatest number of simulations involving genotype 1.
Similarly, for genotype 3 the combination of sofosbuvir plus ledipasvir plus ribavirin for 12 weeks cost $73,000/QALY gained, compared with usual care. This also represents “relatively good value.” However, for genotype 2 the most cost-effective novel therapy, sofosbuvir-ribavirin, was $110,168/QALY gained, which is not considered cost-effective, Dr. Najafzadeh and his associates said (Ann. Intern. Med. 2015 March 17 [doi:10.7326;M14-1152]). Again, an important caveat to these findings was that, at their current prices, the cost of these drugs were not outweighed by the savings that accrued from preventing the complications of HCV. And “regardless of the cost-effectiveness of novel HCV treatments, there is considerable concern that their very high prices could substantially increase short-term overall drug spending for many public and private payers,” the investigators noted.
However, the fact that these regimens don’t reduce health care costs “is an exceptionally high bar” to hold them to – one that “is generally not expected when evaluating whether a new strategy represents good value for the money,” they added.
Two different statistical models found that novel therapies for chronic HCV infection, particularly the combination of sofosbuvir and ledipasvir, are cost effective in most patients, according to separate reports published online March 17 in Annals of Internal Medicine.
However, both groups of researchers cautioned that if these expensive agents are made available to the millions of eligible patients across the country, it would have an immense impact on health care costs for both public and private payers.
The novel therapies, which typically contain sofosbuvir in combination with ledipasvir, simeprevir, or daclatasvir, substantially reduce the length of treatment, achieve much higher rates of sustained viral response (SVR), and offer interferon-free alternatives for patients who can’t tolerate or don’t respond to standard interferon-based treatments. But it is unclear whether these benefits justify their profound expense, compared with current care. Both statistical models were developed to examine this issue, but from different perspectives.
In one study, funded primarily by the National Institutes of Health, investigators constructed a model that simulated 120 possible clinical courses of HCV-infected adults based on different ages and sexes, treatment histories, HCV genotypes, fibrosis scores, and interferon tolerances. For each of these patient profiles, they ran simulations in which patients received either “the old standard of care” (peginterferon and ribavirin, either with or without boceprevir and telaprevir) or sofosbuvir plus ledipasvir.
The average per-patient cost of standard care ranged from $15,000 to $71,000, depending on the patient profile, while that of sofosbuvir-ledipasvir ranged from $66,000 to $154,000, said Jagpreet Chhatwal, Ph.D., of the University of Texas MD Anderson Cancer Center, Houston, and his associates.
Compared with standard care, treating 10,000 patients with sofosbuvir-ledipasvir was projected to prevent 600 cases of decompensated cirrhosis, 310 cases of hepatocellular carcinoma (HCC), 60 liver transplantations, and 550 liver-related deaths, which would result in substantial cost savings. Also, compared with standard care, the incremental cost-effectiveness ratio of sofosbuvir-ledipasvir was $55,400 per additional quality-of-life-year (QALY) gained, which falls well within the accepted range for therapies for other medical conditions. Thus, the new therapy proved to be cost-effective for most HCV patients (Ann. Intern. Med. 2015 March 17 [doi:10.7326/M14-1336]). But there was an important caveat: Many more patients would be eligible for the novel therapies than for the standard care, because the novel therapies are much more easily tolerated. With the addition of so many eligible patients, the resources needed to treat them “could be immense and unsustainable.” Compared with standard care, giving these novel HCV therapies to all eligible patients “would cost an additional $65 billion in the next 5 years,” which would not be counterbalanced by the estimated $16 billion saved by preventing cirrhosis, HCC, and transplantations.
Therefore, “despite the cost-effectiveness of [novel] HCV treatments, our analysis shows that it is unaffordable at the current price,” Dr. Chhatwal and his associates said.
In the other study, funded primarily by CVS Health, researchers developed a discrete-event simulation model of the natural history and progression of liver disease in treatment-naive patients, categorized by whether they were infected with HCV genotype 1, 2, or 3. Several possible treatment regimens were considered for each genotype, and the SVR rates they were projected to attain were derived from those reported in clinical trials, said Mehdi Najafzadeh, Ph.D., of the division of pharmacoepidemiology and pharmacoeconomics, Brigham and Women’s Hospital and Harvard Medical School, Boston, and his associates.
“From a societal perspective, the newly approved PEG-free regimen of sofosbuvir-ledipasvir for 12 weeks could be very cost-effective relative to usual care (costing $12,825/QALY gained) for patients with HCV genotype 1.” This treatment proved to be the optimal strategy in the greatest number of simulations involving genotype 1.
Similarly, for genotype 3 the combination of sofosbuvir plus ledipasvir plus ribavirin for 12 weeks cost $73,000/QALY gained, compared with usual care. This also represents “relatively good value.” However, for genotype 2 the most cost-effective novel therapy, sofosbuvir-ribavirin, was $110,168/QALY gained, which is not considered cost-effective, Dr. Najafzadeh and his associates said (Ann. Intern. Med. 2015 March 17 [doi:10.7326;M14-1152]). Again, an important caveat to these findings was that, at their current prices, the cost of these drugs were not outweighed by the savings that accrued from preventing the complications of HCV. And “regardless of the cost-effectiveness of novel HCV treatments, there is considerable concern that their very high prices could substantially increase short-term overall drug spending for many public and private payers,” the investigators noted.
However, the fact that these regimens don’t reduce health care costs “is an exceptionally high bar” to hold them to – one that “is generally not expected when evaluating whether a new strategy represents good value for the money,” they added.
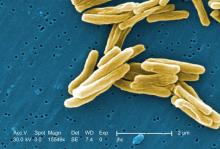
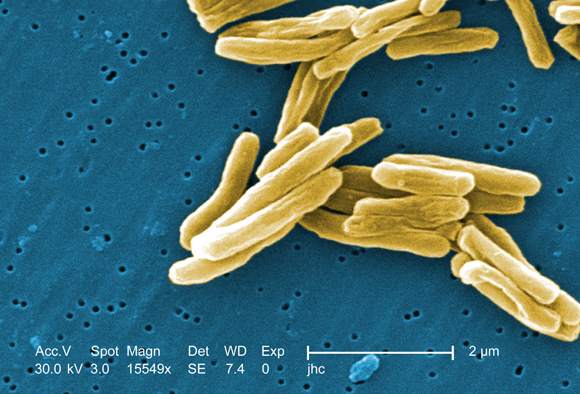
Screen cut latent TB rate among those immigrating to the U.S.
A new approach to screening has improved detection of tuberculosis among people applying to move to the United States, according to a report published online March 17 in Annals of Internal Medicine.
The previous preimmigration screening process involved chest radiography and, for those with abnormal radiographs, acid-fast bacilli smears of three consecutive sputum samples. If at least one smear was positive, the patient completed TB treatment overseas and was followed up after arrival in the United States. This approach failed, however, to detect some latent cases of TB that became reactivated soon after immigrants arrived in the United States.
The new screening process also includes Mycobacterium tuberculosis culture results. For those who have at least one positive sputum smear or culture result, drug-susceptibility testing is performed and directly observed therapy for TB is typically completed overseas before U.S. relocation is allowed, Yecai Liu, a mathematical statistician with the division of global migration and quarantine, Centers for Disease Control and Prevention, and his associates said.
To assess the effectiveness of the new screening algorithm, the researchers analyzed information in the CDC’s disease notification database and the Department of Homeland Security’s immigration records regarding 3,212,421 immigrants and refugees who were screened overseas and arrived in the U.S. during a 7-year period. A total of 51.4% were screened by the old method and 48.6% by the new method.
Before the new screen was implemented, the annual number of smear-negative TB cases among newly arrived immigrants and refugees was relatively constant, at approximately 1,500/year. That number dropped to 940 cases/year after the new screening method was implemented. At the same time, the number of smear-negative but culture-positive TB cases diagnosed overseas among people bound for the U.S. increased from 4 to 629 cases per year.
Thus, the cumulative decline in U.S. cases during the study period was similar to the cumulative total of cases detected before relocation, the investigators said (Ann. Intern. Med. 2015 March 17 [doi:10.7326/M14-2082]).
The findings indicate the new culture-based TB screening is superior to smear-based TB screening and is “a high-yield intervention for preventing TB in immigrants and refugees bound for the United States.” Expanding such screening to cover exchange students, other exchange visitors, and temporary workers from countries with a high incidence of TB might further reduce the rate of the disease in foreign-born populations, the researchers concluded.
A new approach to screening has improved detection of tuberculosis among people applying to move to the United States, according to a report published online March 17 in Annals of Internal Medicine.
The previous preimmigration screening process involved chest radiography and, for those with abnormal radiographs, acid-fast bacilli smears of three consecutive sputum samples. If at least one smear was positive, the patient completed TB treatment overseas and was followed up after arrival in the United States. This approach failed, however, to detect some latent cases of TB that became reactivated soon after immigrants arrived in the United States.
The new screening process also includes Mycobacterium tuberculosis culture results. For those who have at least one positive sputum smear or culture result, drug-susceptibility testing is performed and directly observed therapy for TB is typically completed overseas before U.S. relocation is allowed, Yecai Liu, a mathematical statistician with the division of global migration and quarantine, Centers for Disease Control and Prevention, and his associates said.
To assess the effectiveness of the new screening algorithm, the researchers analyzed information in the CDC’s disease notification database and the Department of Homeland Security’s immigration records regarding 3,212,421 immigrants and refugees who were screened overseas and arrived in the U.S. during a 7-year period. A total of 51.4% were screened by the old method and 48.6% by the new method.
Before the new screen was implemented, the annual number of smear-negative TB cases among newly arrived immigrants and refugees was relatively constant, at approximately 1,500/year. That number dropped to 940 cases/year after the new screening method was implemented. At the same time, the number of smear-negative but culture-positive TB cases diagnosed overseas among people bound for the U.S. increased from 4 to 629 cases per year.
Thus, the cumulative decline in U.S. cases during the study period was similar to the cumulative total of cases detected before relocation, the investigators said (Ann. Intern. Med. 2015 March 17 [doi:10.7326/M14-2082]).
The findings indicate the new culture-based TB screening is superior to smear-based TB screening and is “a high-yield intervention for preventing TB in immigrants and refugees bound for the United States.” Expanding such screening to cover exchange students, other exchange visitors, and temporary workers from countries with a high incidence of TB might further reduce the rate of the disease in foreign-born populations, the researchers concluded.
A new approach to screening has improved detection of tuberculosis among people applying to move to the United States, according to a report published online March 17 in Annals of Internal Medicine.
The previous preimmigration screening process involved chest radiography and, for those with abnormal radiographs, acid-fast bacilli smears of three consecutive sputum samples. If at least one smear was positive, the patient completed TB treatment overseas and was followed up after arrival in the United States. This approach failed, however, to detect some latent cases of TB that became reactivated soon after immigrants arrived in the United States.
The new screening process also includes Mycobacterium tuberculosis culture results. For those who have at least one positive sputum smear or culture result, drug-susceptibility testing is performed and directly observed therapy for TB is typically completed overseas before U.S. relocation is allowed, Yecai Liu, a mathematical statistician with the division of global migration and quarantine, Centers for Disease Control and Prevention, and his associates said.
To assess the effectiveness of the new screening algorithm, the researchers analyzed information in the CDC’s disease notification database and the Department of Homeland Security’s immigration records regarding 3,212,421 immigrants and refugees who were screened overseas and arrived in the U.S. during a 7-year period. A total of 51.4% were screened by the old method and 48.6% by the new method.
Before the new screen was implemented, the annual number of smear-negative TB cases among newly arrived immigrants and refugees was relatively constant, at approximately 1,500/year. That number dropped to 940 cases/year after the new screening method was implemented. At the same time, the number of smear-negative but culture-positive TB cases diagnosed overseas among people bound for the U.S. increased from 4 to 629 cases per year.
Thus, the cumulative decline in U.S. cases during the study period was similar to the cumulative total of cases detected before relocation, the investigators said (Ann. Intern. Med. 2015 March 17 [doi:10.7326/M14-2082]).
The findings indicate the new culture-based TB screening is superior to smear-based TB screening and is “a high-yield intervention for preventing TB in immigrants and refugees bound for the United States.” Expanding such screening to cover exchange students, other exchange visitors, and temporary workers from countries with a high incidence of TB might further reduce the rate of the disease in foreign-born populations, the researchers concluded.
FROM ANNALS OF INTERNAL MEDICINE
Key clinical point: A new screening program reduced the number of tuberculosis cases among people immigrating to the United States.
Major finding: The mean annual number of latent TB cases among newly arrived immigrants and refugees went from 1,500 before the screening program to 940 after the program was implemented.
Data source: A population-based cross-sectional analysis of TB cases among 3,212,421 immigrants and refugees.
Disclosures: This study was supported by the Centers for Disease Control and Prevention. Mr. Liu and his associates reported having no financial disclosures.
Novel HCV therapies found cost effective, with caveats
Two different statistical models found that novel therapies for chronic HCV infection, particularly the combination of sofosbuvir and ledipasvir, are cost effective in most patients, according to separate reports published online March 17 in Annals of Internal Medicine.
However, both groups of researchers cautioned that if these expensive agents are made available to the millions of eligible patients across the country, it would have an immense impact on health care costs for both public and private payers.
The novel therapies, which typically contain sofosbuvir in combination with ledipasvir, simeprevir, or daclatasvir, substantially reduce the length of treatment, achieve much higher rates of sustained viral response (SVR), and offer interferon-free alternatives for patients who can’t tolerate or don’t respond to standard interferon-based treatments. But it is unclear whether these benefits justify their profound expense, compared with current care. Both statistical models were developed to examine this issue, but from different perspectives.
In one study, funded primarily by the National Institutes of Health, investigators constructed a model that simulated 120 possible clinical courses of HCV-infected adults based on different ages and sexes, treatment histories, HCV genotypes, fibrosis scores, and interferon tolerances. For each of these patient profiles, they ran simulations in which patients received either “the old standard of care” (peginterferon and ribavirin, either with or without boceprevir and telaprevir) or sofosbuvir plus ledipasvir.
The average per-patient cost of standard care ranged from $15,000 to $71,000, depending on the patient profile, while that of sofosbuvir-ledipasvir ranged from $66,000 to $154,000, said Jagpreet Chhatwal, Ph.D., of the University of Texas MD Anderson Cancer Center, Houston, and his associates.
Compared with standard care, treating 10,000 patients with sofosbuvir-ledipasvir was projected to prevent 600 cases of decompensated cirrhosis, 310 cases of hepatocellular carcinoma (HCC), 60 liver transplantations, and 550 liver-related deaths, which would result in substantial cost savings. Also, compared with standard care, the incremental cost-effectiveness ratio of sofosbuvir-ledipasvir was $55,400 per additional quality-of-life-year (QALY) gained, which falls well within the accepted range for therapies for other medical conditions. Thus, the new therapy proved to be cost-effective for most HCV patients (Ann. Intern. Med. 2015 March 17 [doi:10.7326/M14-1336]). But there was an important caveat: Many more patients would be eligible for the novel therapies than for the standard care, because the novel therapies are much more easily tolerated. With the addition of so many eligible patients, the resources needed to treat them “could be immense and unsustainable.” Compared with standard care, giving these novel HCV therapies to all eligible patients “would cost an additional $65 billion in the next 5 years,” which would not be counterbalanced by the estimated $16 billion saved by preventing cirrhosis, HCC, and transplantations.
Therefore, “despite the cost-effectiveness of [novel] HCV treatments, our analysis shows that it is unaffordable at the current price,” Dr. Chhatwal and his associates said.
In the other study, funded primarily by CVS Health, researchers developed a discrete-event simulation model of the natural history and progression of liver disease in treatment-naive patients, categorized by whether they were infected with HCV genotype 1, 2, or 3. Several possible treatment regimens were considered for each genotype, and the SVR rates they were projected to attain were derived from those reported in clinical trials, said Mehdi Najafzadeh, Ph.D., of the division of pharmacoepidemiology and pharmacoeconomics, Brigham and Women’s Hospital and Harvard Medical School, Boston, and his associates.
“From a societal perspective, the newly approved PEG-free regimen of sofosbuvir-ledipasvir for 12 weeks could be very cost-effective relative to usual care (costing $12,825/QALY gained) for patients with HCV genotype 1.” This treatment proved to be the optimal strategy in the greatest number of simulations involving genotype 1.
Similarly, for genotype 3 the combination of sofosbuvir plus ledipasvir plus ribavirin for 12 weeks cost $73,000/QALY gained, compared with usual care. This also represents “relatively good value.” However, for genotype 2 the most cost-effective novel therapy, sofosbuvir-ribavirin, was $110,168/QALY gained, which is not considered cost-effective, Dr. Najafzadeh and his associates said (Ann. Intern. Med. 2015 March 17 [doi:10.7326;M14-1152]). Again, an important caveat to these findings was that, at their current prices, the cost of these drugs were not outweighed by the savings that accrued from preventing the complications of HCV. And “regardless of the cost-effectiveness of novel HCV treatments, there is considerable concern that their very high prices could substantially increase short-term overall drug spending for many public and private payers,” the investigators noted.
However, the fact that these regimens don’t reduce health care costs “is an exceptionally high bar” to hold them to – one that “is generally not expected when evaluating whether a new strategy represents good value for the money,” they added.
The recent development and widespread use of well-tolerated and highly efficacious direct-acting antiviral agents (DAAs) represent a paradigm shift in which the retail cost of treatment is now the most significant barrier to hepatitis C virus (HCV) eradication. While we have begun to learn the medical value of curing HCV in the context of the staggering burden of chronic liver disease, much less is known about the economic value of the cost of therapy. There has been swift public outcry over the $1,000 per pill price tag of sofosbuvir, and demand for the medications remains high as nearly all HCV-infected patients are now treatment eligible. Despite the high cost, these two studies collectively demonstrate a favorable incremental cost-effectiveness ratio per adjusted life-year relative to interferon-containing regimens in most patients with HCV: genotype 1, treatment-experienced, and cirrhotics.
These studies illustrate the paradox at the crux of the issue: How can the novel HCV therapies be both cost effective for most HCV patients but simultaneously unaffordable for payers? Although the price of achieving a sustained virologic response (SVR) is reduced with the DAA regimens, the cost of treating all infected patients in the United States could exceed $300 billion, which greatly outweighs the short-term cost of the annual HCV-related burden (approximately $6.5 billion [Hepatology 2013;57:2164-70]).
Treatment of other chronic illness such as HIV may incur greater costs but are distributed over a lifetime. Additionally, the current payers may not be the recipients of the downstream financial benefits of prevented liver-related outcomes. Ultimately, value depends on perspective; payers may balk at the price for the same cure that our patients consider invaluable.
Dr. J.P. Norvell is assistant professor of medicine, Emory University, Atlanta. He has been a consultant to Gilead Sciences.
The recent development and widespread use of well-tolerated and highly efficacious direct-acting antiviral agents (DAAs) represent a paradigm shift in which the retail cost of treatment is now the most significant barrier to hepatitis C virus (HCV) eradication. While we have begun to learn the medical value of curing HCV in the context of the staggering burden of chronic liver disease, much less is known about the economic value of the cost of therapy. There has been swift public outcry over the $1,000 per pill price tag of sofosbuvir, and demand for the medications remains high as nearly all HCV-infected patients are now treatment eligible. Despite the high cost, these two studies collectively demonstrate a favorable incremental cost-effectiveness ratio per adjusted life-year relative to interferon-containing regimens in most patients with HCV: genotype 1, treatment-experienced, and cirrhotics.
These studies illustrate the paradox at the crux of the issue: How can the novel HCV therapies be both cost effective for most HCV patients but simultaneously unaffordable for payers? Although the price of achieving a sustained virologic response (SVR) is reduced with the DAA regimens, the cost of treating all infected patients in the United States could exceed $300 billion, which greatly outweighs the short-term cost of the annual HCV-related burden (approximately $6.5 billion [Hepatology 2013;57:2164-70]).
Treatment of other chronic illness such as HIV may incur greater costs but are distributed over a lifetime. Additionally, the current payers may not be the recipients of the downstream financial benefits of prevented liver-related outcomes. Ultimately, value depends on perspective; payers may balk at the price for the same cure that our patients consider invaluable.
Dr. J.P. Norvell is assistant professor of medicine, Emory University, Atlanta. He has been a consultant to Gilead Sciences.
The recent development and widespread use of well-tolerated and highly efficacious direct-acting antiviral agents (DAAs) represent a paradigm shift in which the retail cost of treatment is now the most significant barrier to hepatitis C virus (HCV) eradication. While we have begun to learn the medical value of curing HCV in the context of the staggering burden of chronic liver disease, much less is known about the economic value of the cost of therapy. There has been swift public outcry over the $1,000 per pill price tag of sofosbuvir, and demand for the medications remains high as nearly all HCV-infected patients are now treatment eligible. Despite the high cost, these two studies collectively demonstrate a favorable incremental cost-effectiveness ratio per adjusted life-year relative to interferon-containing regimens in most patients with HCV: genotype 1, treatment-experienced, and cirrhotics.
These studies illustrate the paradox at the crux of the issue: How can the novel HCV therapies be both cost effective for most HCV patients but simultaneously unaffordable for payers? Although the price of achieving a sustained virologic response (SVR) is reduced with the DAA regimens, the cost of treating all infected patients in the United States could exceed $300 billion, which greatly outweighs the short-term cost of the annual HCV-related burden (approximately $6.5 billion [Hepatology 2013;57:2164-70]).
Treatment of other chronic illness such as HIV may incur greater costs but are distributed over a lifetime. Additionally, the current payers may not be the recipients of the downstream financial benefits of prevented liver-related outcomes. Ultimately, value depends on perspective; payers may balk at the price for the same cure that our patients consider invaluable.
Dr. J.P. Norvell is assistant professor of medicine, Emory University, Atlanta. He has been a consultant to Gilead Sciences.
Two different statistical models found that novel therapies for chronic HCV infection, particularly the combination of sofosbuvir and ledipasvir, are cost effective in most patients, according to separate reports published online March 17 in Annals of Internal Medicine.
However, both groups of researchers cautioned that if these expensive agents are made available to the millions of eligible patients across the country, it would have an immense impact on health care costs for both public and private payers.
The novel therapies, which typically contain sofosbuvir in combination with ledipasvir, simeprevir, or daclatasvir, substantially reduce the length of treatment, achieve much higher rates of sustained viral response (SVR), and offer interferon-free alternatives for patients who can’t tolerate or don’t respond to standard interferon-based treatments. But it is unclear whether these benefits justify their profound expense, compared with current care. Both statistical models were developed to examine this issue, but from different perspectives.
In one study, funded primarily by the National Institutes of Health, investigators constructed a model that simulated 120 possible clinical courses of HCV-infected adults based on different ages and sexes, treatment histories, HCV genotypes, fibrosis scores, and interferon tolerances. For each of these patient profiles, they ran simulations in which patients received either “the old standard of care” (peginterferon and ribavirin, either with or without boceprevir and telaprevir) or sofosbuvir plus ledipasvir.
The average per-patient cost of standard care ranged from $15,000 to $71,000, depending on the patient profile, while that of sofosbuvir-ledipasvir ranged from $66,000 to $154,000, said Jagpreet Chhatwal, Ph.D., of the University of Texas MD Anderson Cancer Center, Houston, and his associates.
Compared with standard care, treating 10,000 patients with sofosbuvir-ledipasvir was projected to prevent 600 cases of decompensated cirrhosis, 310 cases of hepatocellular carcinoma (HCC), 60 liver transplantations, and 550 liver-related deaths, which would result in substantial cost savings. Also, compared with standard care, the incremental cost-effectiveness ratio of sofosbuvir-ledipasvir was $55,400 per additional quality-of-life-year (QALY) gained, which falls well within the accepted range for therapies for other medical conditions. Thus, the new therapy proved to be cost-effective for most HCV patients (Ann. Intern. Med. 2015 March 17 [doi:10.7326/M14-1336]). But there was an important caveat: Many more patients would be eligible for the novel therapies than for the standard care, because the novel therapies are much more easily tolerated. With the addition of so many eligible patients, the resources needed to treat them “could be immense and unsustainable.” Compared with standard care, giving these novel HCV therapies to all eligible patients “would cost an additional $65 billion in the next 5 years,” which would not be counterbalanced by the estimated $16 billion saved by preventing cirrhosis, HCC, and transplantations.
Therefore, “despite the cost-effectiveness of [novel] HCV treatments, our analysis shows that it is unaffordable at the current price,” Dr. Chhatwal and his associates said.
In the other study, funded primarily by CVS Health, researchers developed a discrete-event simulation model of the natural history and progression of liver disease in treatment-naive patients, categorized by whether they were infected with HCV genotype 1, 2, or 3. Several possible treatment regimens were considered for each genotype, and the SVR rates they were projected to attain were derived from those reported in clinical trials, said Mehdi Najafzadeh, Ph.D., of the division of pharmacoepidemiology and pharmacoeconomics, Brigham and Women’s Hospital and Harvard Medical School, Boston, and his associates.
“From a societal perspective, the newly approved PEG-free regimen of sofosbuvir-ledipasvir for 12 weeks could be very cost-effective relative to usual care (costing $12,825/QALY gained) for patients with HCV genotype 1.” This treatment proved to be the optimal strategy in the greatest number of simulations involving genotype 1.
Similarly, for genotype 3 the combination of sofosbuvir plus ledipasvir plus ribavirin for 12 weeks cost $73,000/QALY gained, compared with usual care. This also represents “relatively good value.” However, for genotype 2 the most cost-effective novel therapy, sofosbuvir-ribavirin, was $110,168/QALY gained, which is not considered cost-effective, Dr. Najafzadeh and his associates said (Ann. Intern. Med. 2015 March 17 [doi:10.7326;M14-1152]). Again, an important caveat to these findings was that, at their current prices, the cost of these drugs were not outweighed by the savings that accrued from preventing the complications of HCV. And “regardless of the cost-effectiveness of novel HCV treatments, there is considerable concern that their very high prices could substantially increase short-term overall drug spending for many public and private payers,” the investigators noted.
However, the fact that these regimens don’t reduce health care costs “is an exceptionally high bar” to hold them to – one that “is generally not expected when evaluating whether a new strategy represents good value for the money,” they added.
Two different statistical models found that novel therapies for chronic HCV infection, particularly the combination of sofosbuvir and ledipasvir, are cost effective in most patients, according to separate reports published online March 17 in Annals of Internal Medicine.
However, both groups of researchers cautioned that if these expensive agents are made available to the millions of eligible patients across the country, it would have an immense impact on health care costs for both public and private payers.
The novel therapies, which typically contain sofosbuvir in combination with ledipasvir, simeprevir, or daclatasvir, substantially reduce the length of treatment, achieve much higher rates of sustained viral response (SVR), and offer interferon-free alternatives for patients who can’t tolerate or don’t respond to standard interferon-based treatments. But it is unclear whether these benefits justify their profound expense, compared with current care. Both statistical models were developed to examine this issue, but from different perspectives.
In one study, funded primarily by the National Institutes of Health, investigators constructed a model that simulated 120 possible clinical courses of HCV-infected adults based on different ages and sexes, treatment histories, HCV genotypes, fibrosis scores, and interferon tolerances. For each of these patient profiles, they ran simulations in which patients received either “the old standard of care” (peginterferon and ribavirin, either with or without boceprevir and telaprevir) or sofosbuvir plus ledipasvir.
The average per-patient cost of standard care ranged from $15,000 to $71,000, depending on the patient profile, while that of sofosbuvir-ledipasvir ranged from $66,000 to $154,000, said Jagpreet Chhatwal, Ph.D., of the University of Texas MD Anderson Cancer Center, Houston, and his associates.
Compared with standard care, treating 10,000 patients with sofosbuvir-ledipasvir was projected to prevent 600 cases of decompensated cirrhosis, 310 cases of hepatocellular carcinoma (HCC), 60 liver transplantations, and 550 liver-related deaths, which would result in substantial cost savings. Also, compared with standard care, the incremental cost-effectiveness ratio of sofosbuvir-ledipasvir was $55,400 per additional quality-of-life-year (QALY) gained, which falls well within the accepted range for therapies for other medical conditions. Thus, the new therapy proved to be cost-effective for most HCV patients (Ann. Intern. Med. 2015 March 17 [doi:10.7326/M14-1336]). But there was an important caveat: Many more patients would be eligible for the novel therapies than for the standard care, because the novel therapies are much more easily tolerated. With the addition of so many eligible patients, the resources needed to treat them “could be immense and unsustainable.” Compared with standard care, giving these novel HCV therapies to all eligible patients “would cost an additional $65 billion in the next 5 years,” which would not be counterbalanced by the estimated $16 billion saved by preventing cirrhosis, HCC, and transplantations.
Therefore, “despite the cost-effectiveness of [novel] HCV treatments, our analysis shows that it is unaffordable at the current price,” Dr. Chhatwal and his associates said.
In the other study, funded primarily by CVS Health, researchers developed a discrete-event simulation model of the natural history and progression of liver disease in treatment-naive patients, categorized by whether they were infected with HCV genotype 1, 2, or 3. Several possible treatment regimens were considered for each genotype, and the SVR rates they were projected to attain were derived from those reported in clinical trials, said Mehdi Najafzadeh, Ph.D., of the division of pharmacoepidemiology and pharmacoeconomics, Brigham and Women’s Hospital and Harvard Medical School, Boston, and his associates.
“From a societal perspective, the newly approved PEG-free regimen of sofosbuvir-ledipasvir for 12 weeks could be very cost-effective relative to usual care (costing $12,825/QALY gained) for patients with HCV genotype 1.” This treatment proved to be the optimal strategy in the greatest number of simulations involving genotype 1.
Similarly, for genotype 3 the combination of sofosbuvir plus ledipasvir plus ribavirin for 12 weeks cost $73,000/QALY gained, compared with usual care. This also represents “relatively good value.” However, for genotype 2 the most cost-effective novel therapy, sofosbuvir-ribavirin, was $110,168/QALY gained, which is not considered cost-effective, Dr. Najafzadeh and his associates said (Ann. Intern. Med. 2015 March 17 [doi:10.7326;M14-1152]). Again, an important caveat to these findings was that, at their current prices, the cost of these drugs were not outweighed by the savings that accrued from preventing the complications of HCV. And “regardless of the cost-effectiveness of novel HCV treatments, there is considerable concern that their very high prices could substantially increase short-term overall drug spending for many public and private payers,” the investigators noted.
However, the fact that these regimens don’t reduce health care costs “is an exceptionally high bar” to hold them to – one that “is generally not expected when evaluating whether a new strategy represents good value for the money,” they added.
FROM ANNALS OF INTERNAL MEDICINE
Key clinical point: Two separate computerized statistical models found that new HCV therapies are cost effective in most cases, with important caveats.
Major finding: Compared with standard care, treating 10,000 patients with sofosbuvir-ledipasvir was projected to prevent 600 cases of decompensated cirrhosis, 310 cases of hepatocellular carcinoma, 60 liver transplantations, and 550 liver-related deaths.
Data source: A microsimulation model of the cost-effectiveness of various HCV therapies, and a discrete-event simulation model of cost-effectiveness from a societal standpoint.
Disclosures: Dr. Chhatwal’s study was supported by the National Center for Advancing Translational Sciences and the Veterans Affairs Health Services Research and Development Center for Innovations in Quality, Effectiveness, and Safety. Dr. Najafzadeh’s study was supported by an unrestricted grant from CVS Health to Brigham and Women’s Hospital and by a Canadian Institutes of Health Research fellowship. Both research groups’ financial disclosures are available at www.annals.org.
Despite requirement, only 13% of clinical trials report results
Only 13% of the clinical trials legally required since 2007 to report their findings to ClinicalTrials.gov actually do so, according to a report published online in the New England Journal of Medicine.
The Food and Drug Administration Amendments Act requires sponsors of most clinical trials to register and report their basic summary results within 1 year of either completing data collection for the primary outcome or of terminating. Failure to report study findings is punishable by sanctions including civil penalties of up to $10,000 per day and loss of funding.
The reporting regulation “reflect[s] the ethical obligation of researchers and sponsors to respect human trial participants through fidelity to commitments made explicit in informed consent: namely, to make results of trials available to contribute to generalizable knowledge,” said Dr. Monique L. Anderson of the Duke Clinical Research Institute, Durham, N.C., and her associates.
The law was enacted because of public concern that sponsors and investigators were “selectively publishing trials that favored interests of the sponsors, and that journals were selectively reporting positive findings.” Among other detriments, the failure to report results could harm participants in similar studies by failing to warn them of possible risks, the investigators noted.
They assessed compliance with the law more than 5 years after it was enacted, using a National Library of Medicine algorithm to identify 13,327 eligible clinical trials that were either completed or terminated during 2008-2012.
Only 13.4% of them reported their results within 1 year, and only 38.3% reported their results at any time during the study period (N. Engl. J. Med. 2015;372:1031-9). Moreover, “despite ethical mandates, statutory obligations, and considerable societal pressure, most trials that were funded by the NIH or other government or academic institutions ... have yet to report results at ClinicalTrials.gov, whereas the medical-products industry has been more responsive to the legal mandate,” the researchers explained.
At 1 year, the rate of reporting was 17.0% for industry-sponsored trials, 8.1% for NIH-funded trials, and 5.7% for other government- or academically funded trials. The corresponding rates of reporting at 5 years were only slightly better, at 41.5%, 38.9%, and 27.7%, respectively.
Phase I, II, and III clinical trials were the less likely than phase IV trials were to report their results in a timely fashion. These earlier-phase studies “are primarily focused on proof-of-concept demonstrations and as such represent valuable, closely guarded intellectual property,” Dr. Anderson and her associates said.
Despite the regulation’s threat of penalties, no enforcement has yet occurred, the researchers noted, in part because this portion of the FDA Administration and Amendments Act is still under public discussion and hasn’t been finalized.
Only 13% of the clinical trials legally required since 2007 to report their findings to ClinicalTrials.gov actually do so, according to a report published online in the New England Journal of Medicine.
The Food and Drug Administration Amendments Act requires sponsors of most clinical trials to register and report their basic summary results within 1 year of either completing data collection for the primary outcome or of terminating. Failure to report study findings is punishable by sanctions including civil penalties of up to $10,000 per day and loss of funding.
The reporting regulation “reflect[s] the ethical obligation of researchers and sponsors to respect human trial participants through fidelity to commitments made explicit in informed consent: namely, to make results of trials available to contribute to generalizable knowledge,” said Dr. Monique L. Anderson of the Duke Clinical Research Institute, Durham, N.C., and her associates.
The law was enacted because of public concern that sponsors and investigators were “selectively publishing trials that favored interests of the sponsors, and that journals were selectively reporting positive findings.” Among other detriments, the failure to report results could harm participants in similar studies by failing to warn them of possible risks, the investigators noted.
They assessed compliance with the law more than 5 years after it was enacted, using a National Library of Medicine algorithm to identify 13,327 eligible clinical trials that were either completed or terminated during 2008-2012.
Only 13.4% of them reported their results within 1 year, and only 38.3% reported their results at any time during the study period (N. Engl. J. Med. 2015;372:1031-9). Moreover, “despite ethical mandates, statutory obligations, and considerable societal pressure, most trials that were funded by the NIH or other government or academic institutions ... have yet to report results at ClinicalTrials.gov, whereas the medical-products industry has been more responsive to the legal mandate,” the researchers explained.
At 1 year, the rate of reporting was 17.0% for industry-sponsored trials, 8.1% for NIH-funded trials, and 5.7% for other government- or academically funded trials. The corresponding rates of reporting at 5 years were only slightly better, at 41.5%, 38.9%, and 27.7%, respectively.
Phase I, II, and III clinical trials were the less likely than phase IV trials were to report their results in a timely fashion. These earlier-phase studies “are primarily focused on proof-of-concept demonstrations and as such represent valuable, closely guarded intellectual property,” Dr. Anderson and her associates said.
Despite the regulation’s threat of penalties, no enforcement has yet occurred, the researchers noted, in part because this portion of the FDA Administration and Amendments Act is still under public discussion and hasn’t been finalized.
Only 13% of the clinical trials legally required since 2007 to report their findings to ClinicalTrials.gov actually do so, according to a report published online in the New England Journal of Medicine.
The Food and Drug Administration Amendments Act requires sponsors of most clinical trials to register and report their basic summary results within 1 year of either completing data collection for the primary outcome or of terminating. Failure to report study findings is punishable by sanctions including civil penalties of up to $10,000 per day and loss of funding.
The reporting regulation “reflect[s] the ethical obligation of researchers and sponsors to respect human trial participants through fidelity to commitments made explicit in informed consent: namely, to make results of trials available to contribute to generalizable knowledge,” said Dr. Monique L. Anderson of the Duke Clinical Research Institute, Durham, N.C., and her associates.
The law was enacted because of public concern that sponsors and investigators were “selectively publishing trials that favored interests of the sponsors, and that journals were selectively reporting positive findings.” Among other detriments, the failure to report results could harm participants in similar studies by failing to warn them of possible risks, the investigators noted.
They assessed compliance with the law more than 5 years after it was enacted, using a National Library of Medicine algorithm to identify 13,327 eligible clinical trials that were either completed or terminated during 2008-2012.
Only 13.4% of them reported their results within 1 year, and only 38.3% reported their results at any time during the study period (N. Engl. J. Med. 2015;372:1031-9). Moreover, “despite ethical mandates, statutory obligations, and considerable societal pressure, most trials that were funded by the NIH or other government or academic institutions ... have yet to report results at ClinicalTrials.gov, whereas the medical-products industry has been more responsive to the legal mandate,” the researchers explained.
At 1 year, the rate of reporting was 17.0% for industry-sponsored trials, 8.1% for NIH-funded trials, and 5.7% for other government- or academically funded trials. The corresponding rates of reporting at 5 years were only slightly better, at 41.5%, 38.9%, and 27.7%, respectively.
Phase I, II, and III clinical trials were the less likely than phase IV trials were to report their results in a timely fashion. These earlier-phase studies “are primarily focused on proof-of-concept demonstrations and as such represent valuable, closely guarded intellectual property,” Dr. Anderson and her associates said.
Despite the regulation’s threat of penalties, no enforcement has yet occurred, the researchers noted, in part because this portion of the FDA Administration and Amendments Act is still under public discussion and hasn’t been finalized.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Only 13% of clinical trials legally required to report their findings to ClinicalTrials.gov actually do so.
Major finding: Only 13.4% of 13,327 clinical trials reported their results within 1 year, and only 38.3% of those trials reported results at any time during the 5-year study period.
Data source: An analysis of 13,327 clinical trials completed between 2008 and 2012 that were required to report their results to ClinicalTrials.gov within 1 year.
Disclosures: The study was supported by the Clinical Trials Transformation Initiative and the NIH Common Fund. Dr. Anderson reported having no financial disclosures; two of her associates reported ties to numerous industry sources.
Restrictive vs. liberal transfusion after cardiac surgery
After cardiac surgery, using a restrictive transfusion threshold – forgoing transfusion until hemoglobin level drops to 7.5 g/dL – does not decrease morbidity or costs of care, compared with using a liberal transfusion threshold of 9 g/dL, according to a report published online March 12 in the New England Journal of Medicine.
Several blood management guidelines and health policy statements recommend the restrictive approach in the hope that it will reduce the increasing demand on blood services and the high costs of storing, handling, and administering red-cell units, and also because transfusions following cardiac surgery have been linked to infection, low cardiac output, acute kidney injury, and increased mortality. Clinicians remain uncertain about a safe threshold for transfusions in this setting, which is evidenced by the striking variation in transfusion rates among cardiac centers in the United States (8%-93%) and the United Kingdom (25%-75%), said Dr. Gavin J. Murphy of the British Heart Foundation and department of cardiovascular sciences, University of Leicester (England), and his associates.
They performed the Transfusion Indication Threshold Reduction (TITRe2) study to test the hypothesis that the restrictive approach is superior to the liberal approach regarding both postoperative morbidity and health care costs. Adults undergoing nonemergency cardiac surgery at 17 specialty centers in the United Kingdom were randomly assigned to a restricted (1,000 patients) or a liberal (1,003 patients) transfusion threshold. The median patient age was 70 years, and 68% were men. Most of the procedures were CABG or valve surgeries.
Contrary to expectations, the primary outcome – a composite of serious infection or an ischemic event such as stroke, MI, gut infarction, or acute kidney injury within 3 months – occurred in 35.1% of patients in the restrictive-threshold group and 33.0% in the liberal-threshold group. Secondary outcomes, including length of ICU stay and rates of clinically significant pulmonary complications, also were similar between the two study groups. Rates of other serious postoperative complications were similar, at 35.7% and 34.2%, as was general health status as assessed via the EuroQol Group 5-Dimension Self-Report Questionnaire, further contradicting the study hypothesis.
Mean health care costs were similar between the two study groups: the equivalent of $17,762 U.S. dollars with restrictive-threshhold transfusions and $18,059 with liberal-threshold transfusions, Dr. Murphy and his associates noted (N. Engl. J. Med. 2015 March 12 [doi:10.1056/NEJMoa1403612]).
Unexpectedly, 3-month mortality was significantly higher with restrictive- than with liberal-threshold transfusions (4.2% vs 2.6%). This association persisted in sensitivity analyses and “is a cause for concern,” but it may be due to chance alone, the investigators added.
Findings like those of Murphy et al. provide a great opportunity for discussion and debate, which could lead to development of a consensus on the best postoperative care for these patients. Cardiac surgery departments should review the TITRe2 trial results and decide which threshold they deem to be the most appropriate for transfusion.
The extreme range in hospitals’ rates of transfusion in cardiac surgery – from less than 5% to more than 90% – is extraordinary. Having clinicians actively debate the evidence presented in TITRe2, create transparent interpretations, develop protocols, and hold themselves accountable for following those protocols would represent important steps for improving patient care.
John Spertus, M.D., is at the University of Missouri-Kansas City and Saint Luke’s Mid America Heart Institute, Kansas City. He reported receiving grant support from Lilly, Gilead, Amorcyte, Genentech, and Abbott Vascular; receiving personal fees from United Healthcare, Novartis, and Amgen; having an equity interest in Health Outcomes Sciences; and owning copyrights to the Seattle Angina Questionnaire, the Kansas City Cardiomyopathy Questionnaire, and the Peripheral Artery Questionnaire. Dr. Spertus made these remarks in an editorial accompanying Dr. Murphy’s report (N. Engl. J. Med. 2015 March 12 [doi:10.1056/NEJMe1415394]).
Findings like those of Murphy et al. provide a great opportunity for discussion and debate, which could lead to development of a consensus on the best postoperative care for these patients. Cardiac surgery departments should review the TITRe2 trial results and decide which threshold they deem to be the most appropriate for transfusion.
The extreme range in hospitals’ rates of transfusion in cardiac surgery – from less than 5% to more than 90% – is extraordinary. Having clinicians actively debate the evidence presented in TITRe2, create transparent interpretations, develop protocols, and hold themselves accountable for following those protocols would represent important steps for improving patient care.
John Spertus, M.D., is at the University of Missouri-Kansas City and Saint Luke’s Mid America Heart Institute, Kansas City. He reported receiving grant support from Lilly, Gilead, Amorcyte, Genentech, and Abbott Vascular; receiving personal fees from United Healthcare, Novartis, and Amgen; having an equity interest in Health Outcomes Sciences; and owning copyrights to the Seattle Angina Questionnaire, the Kansas City Cardiomyopathy Questionnaire, and the Peripheral Artery Questionnaire. Dr. Spertus made these remarks in an editorial accompanying Dr. Murphy’s report (N. Engl. J. Med. 2015 March 12 [doi:10.1056/NEJMe1415394]).
Findings like those of Murphy et al. provide a great opportunity for discussion and debate, which could lead to development of a consensus on the best postoperative care for these patients. Cardiac surgery departments should review the TITRe2 trial results and decide which threshold they deem to be the most appropriate for transfusion.
The extreme range in hospitals’ rates of transfusion in cardiac surgery – from less than 5% to more than 90% – is extraordinary. Having clinicians actively debate the evidence presented in TITRe2, create transparent interpretations, develop protocols, and hold themselves accountable for following those protocols would represent important steps for improving patient care.
John Spertus, M.D., is at the University of Missouri-Kansas City and Saint Luke’s Mid America Heart Institute, Kansas City. He reported receiving grant support from Lilly, Gilead, Amorcyte, Genentech, and Abbott Vascular; receiving personal fees from United Healthcare, Novartis, and Amgen; having an equity interest in Health Outcomes Sciences; and owning copyrights to the Seattle Angina Questionnaire, the Kansas City Cardiomyopathy Questionnaire, and the Peripheral Artery Questionnaire. Dr. Spertus made these remarks in an editorial accompanying Dr. Murphy’s report (N. Engl. J. Med. 2015 March 12 [doi:10.1056/NEJMe1415394]).
After cardiac surgery, using a restrictive transfusion threshold – forgoing transfusion until hemoglobin level drops to 7.5 g/dL – does not decrease morbidity or costs of care, compared with using a liberal transfusion threshold of 9 g/dL, according to a report published online March 12 in the New England Journal of Medicine.
Several blood management guidelines and health policy statements recommend the restrictive approach in the hope that it will reduce the increasing demand on blood services and the high costs of storing, handling, and administering red-cell units, and also because transfusions following cardiac surgery have been linked to infection, low cardiac output, acute kidney injury, and increased mortality. Clinicians remain uncertain about a safe threshold for transfusions in this setting, which is evidenced by the striking variation in transfusion rates among cardiac centers in the United States (8%-93%) and the United Kingdom (25%-75%), said Dr. Gavin J. Murphy of the British Heart Foundation and department of cardiovascular sciences, University of Leicester (England), and his associates.
They performed the Transfusion Indication Threshold Reduction (TITRe2) study to test the hypothesis that the restrictive approach is superior to the liberal approach regarding both postoperative morbidity and health care costs. Adults undergoing nonemergency cardiac surgery at 17 specialty centers in the United Kingdom were randomly assigned to a restricted (1,000 patients) or a liberal (1,003 patients) transfusion threshold. The median patient age was 70 years, and 68% were men. Most of the procedures were CABG or valve surgeries.
Contrary to expectations, the primary outcome – a composite of serious infection or an ischemic event such as stroke, MI, gut infarction, or acute kidney injury within 3 months – occurred in 35.1% of patients in the restrictive-threshold group and 33.0% in the liberal-threshold group. Secondary outcomes, including length of ICU stay and rates of clinically significant pulmonary complications, also were similar between the two study groups. Rates of other serious postoperative complications were similar, at 35.7% and 34.2%, as was general health status as assessed via the EuroQol Group 5-Dimension Self-Report Questionnaire, further contradicting the study hypothesis.
Mean health care costs were similar between the two study groups: the equivalent of $17,762 U.S. dollars with restrictive-threshhold transfusions and $18,059 with liberal-threshold transfusions, Dr. Murphy and his associates noted (N. Engl. J. Med. 2015 March 12 [doi:10.1056/NEJMoa1403612]).
Unexpectedly, 3-month mortality was significantly higher with restrictive- than with liberal-threshold transfusions (4.2% vs 2.6%). This association persisted in sensitivity analyses and “is a cause for concern,” but it may be due to chance alone, the investigators added.
After cardiac surgery, using a restrictive transfusion threshold – forgoing transfusion until hemoglobin level drops to 7.5 g/dL – does not decrease morbidity or costs of care, compared with using a liberal transfusion threshold of 9 g/dL, according to a report published online March 12 in the New England Journal of Medicine.
Several blood management guidelines and health policy statements recommend the restrictive approach in the hope that it will reduce the increasing demand on blood services and the high costs of storing, handling, and administering red-cell units, and also because transfusions following cardiac surgery have been linked to infection, low cardiac output, acute kidney injury, and increased mortality. Clinicians remain uncertain about a safe threshold for transfusions in this setting, which is evidenced by the striking variation in transfusion rates among cardiac centers in the United States (8%-93%) and the United Kingdom (25%-75%), said Dr. Gavin J. Murphy of the British Heart Foundation and department of cardiovascular sciences, University of Leicester (England), and his associates.
They performed the Transfusion Indication Threshold Reduction (TITRe2) study to test the hypothesis that the restrictive approach is superior to the liberal approach regarding both postoperative morbidity and health care costs. Adults undergoing nonemergency cardiac surgery at 17 specialty centers in the United Kingdom were randomly assigned to a restricted (1,000 patients) or a liberal (1,003 patients) transfusion threshold. The median patient age was 70 years, and 68% were men. Most of the procedures were CABG or valve surgeries.
Contrary to expectations, the primary outcome – a composite of serious infection or an ischemic event such as stroke, MI, gut infarction, or acute kidney injury within 3 months – occurred in 35.1% of patients in the restrictive-threshold group and 33.0% in the liberal-threshold group. Secondary outcomes, including length of ICU stay and rates of clinically significant pulmonary complications, also were similar between the two study groups. Rates of other serious postoperative complications were similar, at 35.7% and 34.2%, as was general health status as assessed via the EuroQol Group 5-Dimension Self-Report Questionnaire, further contradicting the study hypothesis.
Mean health care costs were similar between the two study groups: the equivalent of $17,762 U.S. dollars with restrictive-threshhold transfusions and $18,059 with liberal-threshold transfusions, Dr. Murphy and his associates noted (N. Engl. J. Med. 2015 March 12 [doi:10.1056/NEJMoa1403612]).
Unexpectedly, 3-month mortality was significantly higher with restrictive- than with liberal-threshold transfusions (4.2% vs 2.6%). This association persisted in sensitivity analyses and “is a cause for concern,” but it may be due to chance alone, the investigators added.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: After cardiac surgery, using a restrictive transfusion threshold – forgoing transfusion unless hemoglobin level drops to 7.5 g/dL – doesn’t decrease morbidity or costs, compared with using a liberal transfusion threshold (9 g/dL).
Major finding: Contrary to expectations, the primary outcome, a composite of serious infection or an ischemic event such as stroke, MI, gut infarction, or acute kidney injury within 3 months, occurred in 35% of patients in the restrictive-threshold group and 33% in the liberal-threshold group.
Data source: A multicenter randomized controlled trial comparing restrictive and liberal transfusion thresholds in 2,003 cardiac surgery patients in the United Kingdom who were followed for 3 months for the development of serious complications.
Disclosures: The National Institute for Health Research’s Health Technology Assessment Program, the NIHR Bristol Biomedical Research Unit in Cardiovascular Disease, and the British Heart Foundation supported the study. Dr. Murphy and his associates reported having no financial disclosures.
Type 2 Diabetes Lower in Familial Hypercholesterolemia
The prevalence of type 2 diabetes appears to be significantly lower in patients with familial hypercholesterolemia than in their unaffected relatives, according to a report published online March 10 in JAMA.
In an observational cross-sectional analysis of data from a nationwide Dutch registry of familial hypercholesterolemia, the prevalence of type 2 diabetes was 1.75% in 25,137 patients with the disorder, compared with 2.93% in 38,183 of their unaffected relatives. If this finding is confirmed in further research, it would support the hypothesis that cellular cholesterol metabolism plays a role in the development of type 2 diabetes, “perhaps because increased intracellular cholesterol levels are detrimental for pancreatic beta-cell function,” said Dr. Joost Besseling of the department of vascular medicine, Academic Medical Centre, Amsterdam, and his associates.
Patients with familial hypercholesterolemia require strict clinical follow-up, and it has been noticed – but, until now, not substantiated – that they are less prone to developing type 2 diabetes.
In what they described as the first study to examine this relationship, Dr. Besseling and his colleagues found that the strong inverse association (odds ratio, 0.62; 95% confidence interval, 0.55-0.69) between familial hypercholesterolemia and type 2 diabetes was consistent across every subgroup they evaluated, regardless of patient age, statin use, smoking status, and other possible confounding factors.
In addition, they found that the severity of the genetic mutation underlying the familial hypercholesterolemia also correlated, in an inverse dose-response manner, with the prevalence of type 2 diabetes (JAMA 2015;313:1029-36).
If these findings are confirmed, “they might provide support for development of new approaches to the prevention and treatment of type 2 diabetes by improving function and survival of pancreatic beta cells,” the investigators said.
The prevalence of type 2 diabetes appears to be significantly lower in patients with familial hypercholesterolemia than in their unaffected relatives, according to a report published online March 10 in JAMA.
In an observational cross-sectional analysis of data from a nationwide Dutch registry of familial hypercholesterolemia, the prevalence of type 2 diabetes was 1.75% in 25,137 patients with the disorder, compared with 2.93% in 38,183 of their unaffected relatives. If this finding is confirmed in further research, it would support the hypothesis that cellular cholesterol metabolism plays a role in the development of type 2 diabetes, “perhaps because increased intracellular cholesterol levels are detrimental for pancreatic beta-cell function,” said Dr. Joost Besseling of the department of vascular medicine, Academic Medical Centre, Amsterdam, and his associates.
Patients with familial hypercholesterolemia require strict clinical follow-up, and it has been noticed – but, until now, not substantiated – that they are less prone to developing type 2 diabetes.
In what they described as the first study to examine this relationship, Dr. Besseling and his colleagues found that the strong inverse association (odds ratio, 0.62; 95% confidence interval, 0.55-0.69) between familial hypercholesterolemia and type 2 diabetes was consistent across every subgroup they evaluated, regardless of patient age, statin use, smoking status, and other possible confounding factors.
In addition, they found that the severity of the genetic mutation underlying the familial hypercholesterolemia also correlated, in an inverse dose-response manner, with the prevalence of type 2 diabetes (JAMA 2015;313:1029-36).
If these findings are confirmed, “they might provide support for development of new approaches to the prevention and treatment of type 2 diabetes by improving function and survival of pancreatic beta cells,” the investigators said.
The prevalence of type 2 diabetes appears to be significantly lower in patients with familial hypercholesterolemia than in their unaffected relatives, according to a report published online March 10 in JAMA.
In an observational cross-sectional analysis of data from a nationwide Dutch registry of familial hypercholesterolemia, the prevalence of type 2 diabetes was 1.75% in 25,137 patients with the disorder, compared with 2.93% in 38,183 of their unaffected relatives. If this finding is confirmed in further research, it would support the hypothesis that cellular cholesterol metabolism plays a role in the development of type 2 diabetes, “perhaps because increased intracellular cholesterol levels are detrimental for pancreatic beta-cell function,” said Dr. Joost Besseling of the department of vascular medicine, Academic Medical Centre, Amsterdam, and his associates.
Patients with familial hypercholesterolemia require strict clinical follow-up, and it has been noticed – but, until now, not substantiated – that they are less prone to developing type 2 diabetes.
In what they described as the first study to examine this relationship, Dr. Besseling and his colleagues found that the strong inverse association (odds ratio, 0.62; 95% confidence interval, 0.55-0.69) between familial hypercholesterolemia and type 2 diabetes was consistent across every subgroup they evaluated, regardless of patient age, statin use, smoking status, and other possible confounding factors.
In addition, they found that the severity of the genetic mutation underlying the familial hypercholesterolemia also correlated, in an inverse dose-response manner, with the prevalence of type 2 diabetes (JAMA 2015;313:1029-36).
If these findings are confirmed, “they might provide support for development of new approaches to the prevention and treatment of type 2 diabetes by improving function and survival of pancreatic beta cells,” the investigators said.
Autopsy studies verify flutemetamol’s ability to identify brain beta-amyloid
PET scans using radio-labeled flutemetamol were safe to use in vivo and confirmed the presence or absence of neuritic beta-amyloid levels “with high sensitivity and specificity” in a phase III trial involving 68 terminally ill older adults.
“In-vivo detection of brain beta-amyloid may help increase diagnostic accuracy in cognitively impaired patients, compared with clinical diagnosis alone,” wrote Dr. Craig Curtis of Compass Research, Orlando, and his associates in their report (JAMA Neurol. 2015;72:287-94).
The PET scanning agent used in the study, [18F]flutemetamol (Vizamyl), binds with high affinity to beta-amyloid but rapidly clears from normal brain tissue; it was approved in October 2013 for PET imaging of the brain in adults being evaluated for Alzheimer’s disease and dementia.
The trial involved 203 consecutive patients attending 19 dementia clinics, memory centers, and hospices in the United States and England who were aged 55 years and older and who had a life expectancy of 1 year or less. A total of 180 who underwent [18F]flutemetamol PET scanning comprised the population for the safety analysis. A subset of 68 patients who died during the study period underwent autopsy so that brain pathology could be compared with the PET images; these patients comprised the sensitivity/specificity analysis.
Five specially trained readers who were blinded to patient information – four nuclear medicine physicians and one radiologist – independently interpreted the PET scans as being either positive or negative for neuritic beta-amyloid. All the images were of good technical quality, and none were considered unevaluable.
On postmortem examination, 25 of the 68 brains (37%) were found to be beta-amyloid negative and 43 (63%) to be beta-amyloid positive.
The sensitivity of [18F]flutemetamol PET scanning ranged from 81% to 93% (median, 88%) across the five scan readers, and the specificity ranged from 44% to 92% (median, 88%). One of the readers frequently overestimated the level of beta-amyloid in the scans and was considered an outlier regarding specificity. Between-reader agreement was 90% or higher when that reader’s interpretations were excluded from the analysis, the investigators said.
Overall, 6 (14%) of 43 brains found to be beta-amyloid positive at autopsy were read as false negatives, and 2 (8%) of 25 brains found to be beta-amyloid–negative at autopsy were read as false positives.
The intravenous [18F]flutemetamol injections were well tolerated, and no adverse events were attributed to the agent or the PET procedure. However, four participants were withdrawn from the study because they were unable to remain motionless in the scanner.
This study was funded by GE Healthcare, maker of [18F]flutemetamol, which also planned and conducted the data analysis. Dr. Curtis reported having no financial disclosures; his associates reported numerous ties to industry sources.
A scan that accurately detects beta-amyloid is not the same thing as a test for Alzheimer’s disease. Approximately 25% of cognitively normal older adults and nearly 50% of people older than 80 years also show beta-amyloid on PET scans, as do patients with conditions such as dementia with Lewy bodies and cerebral amyloid angiopathy.

|
| Courtesy Dr. Gil D. Rabinovici Dr. Gil D. Rabinovici |
In addition, it is concerning that one of the five rigorously trained PET scan readers frequently overestimated the amount of beta-amyloid and showed worryingly low specificity (44%) in interpreting these scans. At present, PET amyloid imaging should be considered only for patients in whom there is considerable diagnostic uncertainty after a comprehensive clinical evaluation and in whom knowledge of beta-amyloid status is likely to change diagnosis and management. This would include patients with unexplained mild cognitive impairment, with atypical or mixed presentations, and those who present with dementia at an unusually early age.
Dr. Gil D. Rabinovici is at the Memory and Aging Center in the department of neurology at the University of California, San Francisco. He reported receiving speaking honoraria from GE Healthcare, maker of [18F]flutemetamol and funder of Dr. Curtis’s study, and research support from Avid Radiopharmaceuticals, a subsidiary of Eli Lilly. Dr. Rabinovici made these remarks in an editorial accompanying Dr. Curtis’s report (JAMA Neurol. 2015;72:265-6).
A scan that accurately detects beta-amyloid is not the same thing as a test for Alzheimer’s disease. Approximately 25% of cognitively normal older adults and nearly 50% of people older than 80 years also show beta-amyloid on PET scans, as do patients with conditions such as dementia with Lewy bodies and cerebral amyloid angiopathy.

|
| Courtesy Dr. Gil D. Rabinovici Dr. Gil D. Rabinovici |
In addition, it is concerning that one of the five rigorously trained PET scan readers frequently overestimated the amount of beta-amyloid and showed worryingly low specificity (44%) in interpreting these scans. At present, PET amyloid imaging should be considered only for patients in whom there is considerable diagnostic uncertainty after a comprehensive clinical evaluation and in whom knowledge of beta-amyloid status is likely to change diagnosis and management. This would include patients with unexplained mild cognitive impairment, with atypical or mixed presentations, and those who present with dementia at an unusually early age.
Dr. Gil D. Rabinovici is at the Memory and Aging Center in the department of neurology at the University of California, San Francisco. He reported receiving speaking honoraria from GE Healthcare, maker of [18F]flutemetamol and funder of Dr. Curtis’s study, and research support from Avid Radiopharmaceuticals, a subsidiary of Eli Lilly. Dr. Rabinovici made these remarks in an editorial accompanying Dr. Curtis’s report (JAMA Neurol. 2015;72:265-6).
A scan that accurately detects beta-amyloid is not the same thing as a test for Alzheimer’s disease. Approximately 25% of cognitively normal older adults and nearly 50% of people older than 80 years also show beta-amyloid on PET scans, as do patients with conditions such as dementia with Lewy bodies and cerebral amyloid angiopathy.

|
| Courtesy Dr. Gil D. Rabinovici Dr. Gil D. Rabinovici |
In addition, it is concerning that one of the five rigorously trained PET scan readers frequently overestimated the amount of beta-amyloid and showed worryingly low specificity (44%) in interpreting these scans. At present, PET amyloid imaging should be considered only for patients in whom there is considerable diagnostic uncertainty after a comprehensive clinical evaluation and in whom knowledge of beta-amyloid status is likely to change diagnosis and management. This would include patients with unexplained mild cognitive impairment, with atypical or mixed presentations, and those who present with dementia at an unusually early age.
Dr. Gil D. Rabinovici is at the Memory and Aging Center in the department of neurology at the University of California, San Francisco. He reported receiving speaking honoraria from GE Healthcare, maker of [18F]flutemetamol and funder of Dr. Curtis’s study, and research support from Avid Radiopharmaceuticals, a subsidiary of Eli Lilly. Dr. Rabinovici made these remarks in an editorial accompanying Dr. Curtis’s report (JAMA Neurol. 2015;72:265-6).
PET scans using radio-labeled flutemetamol were safe to use in vivo and confirmed the presence or absence of neuritic beta-amyloid levels “with high sensitivity and specificity” in a phase III trial involving 68 terminally ill older adults.
“In-vivo detection of brain beta-amyloid may help increase diagnostic accuracy in cognitively impaired patients, compared with clinical diagnosis alone,” wrote Dr. Craig Curtis of Compass Research, Orlando, and his associates in their report (JAMA Neurol. 2015;72:287-94).
The PET scanning agent used in the study, [18F]flutemetamol (Vizamyl), binds with high affinity to beta-amyloid but rapidly clears from normal brain tissue; it was approved in October 2013 for PET imaging of the brain in adults being evaluated for Alzheimer’s disease and dementia.
The trial involved 203 consecutive patients attending 19 dementia clinics, memory centers, and hospices in the United States and England who were aged 55 years and older and who had a life expectancy of 1 year or less. A total of 180 who underwent [18F]flutemetamol PET scanning comprised the population for the safety analysis. A subset of 68 patients who died during the study period underwent autopsy so that brain pathology could be compared with the PET images; these patients comprised the sensitivity/specificity analysis.
Five specially trained readers who were blinded to patient information – four nuclear medicine physicians and one radiologist – independently interpreted the PET scans as being either positive or negative for neuritic beta-amyloid. All the images were of good technical quality, and none were considered unevaluable.
On postmortem examination, 25 of the 68 brains (37%) were found to be beta-amyloid negative and 43 (63%) to be beta-amyloid positive.
The sensitivity of [18F]flutemetamol PET scanning ranged from 81% to 93% (median, 88%) across the five scan readers, and the specificity ranged from 44% to 92% (median, 88%). One of the readers frequently overestimated the level of beta-amyloid in the scans and was considered an outlier regarding specificity. Between-reader agreement was 90% or higher when that reader’s interpretations were excluded from the analysis, the investigators said.
Overall, 6 (14%) of 43 brains found to be beta-amyloid positive at autopsy were read as false negatives, and 2 (8%) of 25 brains found to be beta-amyloid–negative at autopsy were read as false positives.
The intravenous [18F]flutemetamol injections were well tolerated, and no adverse events were attributed to the agent or the PET procedure. However, four participants were withdrawn from the study because they were unable to remain motionless in the scanner.
This study was funded by GE Healthcare, maker of [18F]flutemetamol, which also planned and conducted the data analysis. Dr. Curtis reported having no financial disclosures; his associates reported numerous ties to industry sources.
PET scans using radio-labeled flutemetamol were safe to use in vivo and confirmed the presence or absence of neuritic beta-amyloid levels “with high sensitivity and specificity” in a phase III trial involving 68 terminally ill older adults.
“In-vivo detection of brain beta-amyloid may help increase diagnostic accuracy in cognitively impaired patients, compared with clinical diagnosis alone,” wrote Dr. Craig Curtis of Compass Research, Orlando, and his associates in their report (JAMA Neurol. 2015;72:287-94).
The PET scanning agent used in the study, [18F]flutemetamol (Vizamyl), binds with high affinity to beta-amyloid but rapidly clears from normal brain tissue; it was approved in October 2013 for PET imaging of the brain in adults being evaluated for Alzheimer’s disease and dementia.
The trial involved 203 consecutive patients attending 19 dementia clinics, memory centers, and hospices in the United States and England who were aged 55 years and older and who had a life expectancy of 1 year or less. A total of 180 who underwent [18F]flutemetamol PET scanning comprised the population for the safety analysis. A subset of 68 patients who died during the study period underwent autopsy so that brain pathology could be compared with the PET images; these patients comprised the sensitivity/specificity analysis.
Five specially trained readers who were blinded to patient information – four nuclear medicine physicians and one radiologist – independently interpreted the PET scans as being either positive or negative for neuritic beta-amyloid. All the images were of good technical quality, and none were considered unevaluable.
On postmortem examination, 25 of the 68 brains (37%) were found to be beta-amyloid negative and 43 (63%) to be beta-amyloid positive.
The sensitivity of [18F]flutemetamol PET scanning ranged from 81% to 93% (median, 88%) across the five scan readers, and the specificity ranged from 44% to 92% (median, 88%). One of the readers frequently overestimated the level of beta-amyloid in the scans and was considered an outlier regarding specificity. Between-reader agreement was 90% or higher when that reader’s interpretations were excluded from the analysis, the investigators said.
Overall, 6 (14%) of 43 brains found to be beta-amyloid positive at autopsy were read as false negatives, and 2 (8%) of 25 brains found to be beta-amyloid–negative at autopsy were read as false positives.
The intravenous [18F]flutemetamol injections were well tolerated, and no adverse events were attributed to the agent or the PET procedure. However, four participants were withdrawn from the study because they were unable to remain motionless in the scanner.
This study was funded by GE Healthcare, maker of [18F]flutemetamol, which also planned and conducted the data analysis. Dr. Curtis reported having no financial disclosures; his associates reported numerous ties to industry sources.
FROM JAMA NEUROLOGY
Key clinical point: PET scans using radio-labeled flutemetamol were safe and showed neuritic beta-amyloid levels with high sensitivity and specificity in terminally ill older adults.
Major finding: The sensitivity of [18F]flutemetamol PET scanning ranged from 81% to 93% (median, 88%) across five scan readers, and the specificity ranged from 44% to 92% (median, 88%).
Data source: A multicenter, phase III clinical trial comparing in-vivo PET scans against later postmortem findings in the brains of 68 older adults.
Disclosures: This study was funded by GE Healthcare, maker of [18F]flutemetamol, which also planned and conducted the data analysis. Dr. Curtis reported having no financial disclosures; his associates reported numerous ties to industry sources.
Type 2 diabetes lower in familial hypercholesterolemia
The prevalence of type 2 diabetes appears to be significantly lower in patients with familial hypercholesterolemia than in their unaffected relatives, according to a report published online March 10 in JAMA.
In an observational cross-sectional analysis of data from a nationwide Dutch registry of familial hypercholesterolemia, the prevalence of type 2 diabetes was 1.75% in 25,137 patients with the disorder, compared with 2.93% in 38,183 of their unaffected relatives. If this finding is confirmed in further research, it would support the hypothesis that cellular cholesterol metabolism plays a role in the development of type 2 diabetes, “perhaps because increased intracellular cholesterol levels are detrimental for pancreatic beta-cell function,” said Dr. Joost Besseling of the department of vascular medicine, Academic Medical Centre, Amsterdam, and his associates.
Patients with familial hypercholesterolemia require strict clinical follow-up, and it has been noticed – but, until now, not substantiated – that they are less prone to developing type 2 diabetes.
In what they described as the first study to examine this relationship, Dr. Besseling and his colleagues found that the strong inverse association (odds ratio, 0.62; 95% confidence interval, 0.55-0.69) between familial hypercholesterolemia and type 2 diabetes was consistent across every subgroup they evaluated, regardless of patient age, statin use, smoking status, and other possible confounding factors.
In addition, they found that the severity of the genetic mutation underlying the familial hypercholesterolemia also correlated, in an inverse dose-response manner, with the prevalence of type 2 diabetes (JAMA 2015;313:1029-36).
If these findings are confirmed, “they might provide support for development of new approaches to the prevention and treatment of type 2 diabetes by improving function and survival of pancreatic beta cells,” the investigators said.
The findings by Besseling et al. should allay any concerns physicians may have about using statins in patients who have familial hypercholesterolemia. The drugs are now known to promote type 2 diabetes in the general population, but these patients appear to be at low risk for that metabolic disorder.
The study results do not, and should not, alter the use of these important medications in patients at elevated cardiovascular risk, who clearly benefit from statin therapy.
Dr. David Preiss and Dr. Naveed Sattar are at the BHF Glasgow Cardiovascular Research Centre at the University of Glasgow. Dr. Preiss reported serving as a consultant for Sanofi-Aventis; Dr. Sattar reported financial relationships with Amgen, Kowa Pharmaceuticals, and Sanofi-Aventis. They made these remarks in an editorial accompanying Dr. Besseling’s report (JAMA 2015;313:1016-7).
The findings by Besseling et al. should allay any concerns physicians may have about using statins in patients who have familial hypercholesterolemia. The drugs are now known to promote type 2 diabetes in the general population, but these patients appear to be at low risk for that metabolic disorder.
The study results do not, and should not, alter the use of these important medications in patients at elevated cardiovascular risk, who clearly benefit from statin therapy.
Dr. David Preiss and Dr. Naveed Sattar are at the BHF Glasgow Cardiovascular Research Centre at the University of Glasgow. Dr. Preiss reported serving as a consultant for Sanofi-Aventis; Dr. Sattar reported financial relationships with Amgen, Kowa Pharmaceuticals, and Sanofi-Aventis. They made these remarks in an editorial accompanying Dr. Besseling’s report (JAMA 2015;313:1016-7).
The findings by Besseling et al. should allay any concerns physicians may have about using statins in patients who have familial hypercholesterolemia. The drugs are now known to promote type 2 diabetes in the general population, but these patients appear to be at low risk for that metabolic disorder.
The study results do not, and should not, alter the use of these important medications in patients at elevated cardiovascular risk, who clearly benefit from statin therapy.
Dr. David Preiss and Dr. Naveed Sattar are at the BHF Glasgow Cardiovascular Research Centre at the University of Glasgow. Dr. Preiss reported serving as a consultant for Sanofi-Aventis; Dr. Sattar reported financial relationships with Amgen, Kowa Pharmaceuticals, and Sanofi-Aventis. They made these remarks in an editorial accompanying Dr. Besseling’s report (JAMA 2015;313:1016-7).
The prevalence of type 2 diabetes appears to be significantly lower in patients with familial hypercholesterolemia than in their unaffected relatives, according to a report published online March 10 in JAMA.
In an observational cross-sectional analysis of data from a nationwide Dutch registry of familial hypercholesterolemia, the prevalence of type 2 diabetes was 1.75% in 25,137 patients with the disorder, compared with 2.93% in 38,183 of their unaffected relatives. If this finding is confirmed in further research, it would support the hypothesis that cellular cholesterol metabolism plays a role in the development of type 2 diabetes, “perhaps because increased intracellular cholesterol levels are detrimental for pancreatic beta-cell function,” said Dr. Joost Besseling of the department of vascular medicine, Academic Medical Centre, Amsterdam, and his associates.
Patients with familial hypercholesterolemia require strict clinical follow-up, and it has been noticed – but, until now, not substantiated – that they are less prone to developing type 2 diabetes.
In what they described as the first study to examine this relationship, Dr. Besseling and his colleagues found that the strong inverse association (odds ratio, 0.62; 95% confidence interval, 0.55-0.69) between familial hypercholesterolemia and type 2 diabetes was consistent across every subgroup they evaluated, regardless of patient age, statin use, smoking status, and other possible confounding factors.
In addition, they found that the severity of the genetic mutation underlying the familial hypercholesterolemia also correlated, in an inverse dose-response manner, with the prevalence of type 2 diabetes (JAMA 2015;313:1029-36).
If these findings are confirmed, “they might provide support for development of new approaches to the prevention and treatment of type 2 diabetes by improving function and survival of pancreatic beta cells,” the investigators said.
The prevalence of type 2 diabetes appears to be significantly lower in patients with familial hypercholesterolemia than in their unaffected relatives, according to a report published online March 10 in JAMA.
In an observational cross-sectional analysis of data from a nationwide Dutch registry of familial hypercholesterolemia, the prevalence of type 2 diabetes was 1.75% in 25,137 patients with the disorder, compared with 2.93% in 38,183 of their unaffected relatives. If this finding is confirmed in further research, it would support the hypothesis that cellular cholesterol metabolism plays a role in the development of type 2 diabetes, “perhaps because increased intracellular cholesterol levels are detrimental for pancreatic beta-cell function,” said Dr. Joost Besseling of the department of vascular medicine, Academic Medical Centre, Amsterdam, and his associates.
Patients with familial hypercholesterolemia require strict clinical follow-up, and it has been noticed – but, until now, not substantiated – that they are less prone to developing type 2 diabetes.
In what they described as the first study to examine this relationship, Dr. Besseling and his colleagues found that the strong inverse association (odds ratio, 0.62; 95% confidence interval, 0.55-0.69) between familial hypercholesterolemia and type 2 diabetes was consistent across every subgroup they evaluated, regardless of patient age, statin use, smoking status, and other possible confounding factors.
In addition, they found that the severity of the genetic mutation underlying the familial hypercholesterolemia also correlated, in an inverse dose-response manner, with the prevalence of type 2 diabetes (JAMA 2015;313:1029-36).
If these findings are confirmed, “they might provide support for development of new approaches to the prevention and treatment of type 2 diabetes by improving function and survival of pancreatic beta cells,” the investigators said.
FROM JAMA
Key clinical point: The prevalence of type 2 diabetes appears to be significantly lower in patients with familial hypercholesterolemia than in their unaffected relatives.
Major finding: The prevalence of type 2 diabetes was 1.75% in 25,137 patients with the familial hypercholesterolemia, compared with 2.93% in 38,183 of their unaffected relatives.
Data source: An observational cross-sectional analysis of data for 63,320 people in the Dutch national registry of familial hypercholesterolemia.
Disclosures: The study sponsor was not specified; the familial hypercholesterolemia registry is subsidized by the Dutch government. Dr. Besseling reported having no financial disclosures; his associates reported ties to Aegerion, Amgen, AstraZeneca, Boehringer Ingelheim, Cerenis, Eli Lilly, Genzyme, JSiS, MSD, Novartis, Pfizer, Regeneron, Roche, and Sanofi.
26% 1-year death, stroke rate after TAVR
One year after transcatheter aortic valve replacement in the United States, the overall mortality was 23.7%, the stroke rate was 4.1%, and the composite outcome of death and stroke was 26.0%, according to a report published online March 10 in JAMA.
Long-term outcomes for TAVR haven’t been well studied until now, yet the procedure is being performed with increasing frequency for aortic stenosis in patients who are too high risk to undergo conventional surgical aortic valve replacement, said Dr. David R. Holmes Jr. of the Mayo Clinic, Rochester, Minn., and his associates.
They assessed 1-year outcomes by analyzing administrative data from the Centers for Medicare & Medicaid Services and clinical data from the Transcatheter Valve Therapies Registry, an initiative of the Society of Thoracic Surgeons and the American College of Cardiology. The study involved 12,182 patients who underwent TAVR at 299 medical centers across the country during a 19-month period. The patients’ median age was 84 years; 95% were white and 52% were women. The transfemoral approach was used in most patients, but alternative approaches were used in roughly 44%. As expected for an elderly, high-risk study population, baseline functional status was poor and comorbidities were common. They included reduced left ventricular ejection fraction (26% of patients), prior stroke (12%), moderate or severe lung disease (28%), renal failure (16%), peripheral vascular disease (32%), and atrial fibrillation (42%), Dr. Holmes and his associates reported (JAMA 2015 March 10 [doi:10.1001/jama.2015.1474]).
In addition to the mortality and stroke rates listed above, the 1-year rate of one rehospitalization was 24.4%, that of two rehospitalizations was 12.5%, and that of three or more rehospitalizations was 11.6%. The 1-year readmission rate specifically for stroke, heart failure, or repeat aortic valve intervention was 18.6%. These are important considerations for elderly, fragile patients because rehospitalizations indicate “an unacceptable quality-of-life outcome” and are very costly, the investigators noted.
Several baseline characteristics, including male sex, severe chronic obstructive pulmonary disease, dialysis-dependent end-stage renal disease, older age, higher STS Predicted Risk of Operative Mortality (PROM) score, a history of atrial fibrillation/flutter, and use of an access route (other than transfemoral), were found to be independently associated with higher 1-year mortality. Thus, “It may be possible to identify patients who may not benefit from this procedure and who should be counseled accordingly.” For example, in this study there was a small (77 patients) very high-risk subset of patients – aged 85-94 years, dependent on dialysis, and having an STS PROM score greater than 15% – whose 1-year mortality was 53.5%.
The STS and the ACC supported this study, and support the Transcatheter Valve Therapies Registry. Dr. Holmes reported having no relevant financial disclosures; his associates reported ties to Boston Scientific, Edwards Lifesciences, Janssen, Eli Lilly, Boehringer Ingelheim, Bayer, and AstraZeneca.
One year after transcatheter aortic valve replacement in the United States, the overall mortality was 23.7%, the stroke rate was 4.1%, and the composite outcome of death and stroke was 26.0%, according to a report published online March 10 in JAMA.
Long-term outcomes for TAVR haven’t been well studied until now, yet the procedure is being performed with increasing frequency for aortic stenosis in patients who are too high risk to undergo conventional surgical aortic valve replacement, said Dr. David R. Holmes Jr. of the Mayo Clinic, Rochester, Minn., and his associates.
They assessed 1-year outcomes by analyzing administrative data from the Centers for Medicare & Medicaid Services and clinical data from the Transcatheter Valve Therapies Registry, an initiative of the Society of Thoracic Surgeons and the American College of Cardiology. The study involved 12,182 patients who underwent TAVR at 299 medical centers across the country during a 19-month period. The patients’ median age was 84 years; 95% were white and 52% were women. The transfemoral approach was used in most patients, but alternative approaches were used in roughly 44%. As expected for an elderly, high-risk study population, baseline functional status was poor and comorbidities were common. They included reduced left ventricular ejection fraction (26% of patients), prior stroke (12%), moderate or severe lung disease (28%), renal failure (16%), peripheral vascular disease (32%), and atrial fibrillation (42%), Dr. Holmes and his associates reported (JAMA 2015 March 10 [doi:10.1001/jama.2015.1474]).
In addition to the mortality and stroke rates listed above, the 1-year rate of one rehospitalization was 24.4%, that of two rehospitalizations was 12.5%, and that of three or more rehospitalizations was 11.6%. The 1-year readmission rate specifically for stroke, heart failure, or repeat aortic valve intervention was 18.6%. These are important considerations for elderly, fragile patients because rehospitalizations indicate “an unacceptable quality-of-life outcome” and are very costly, the investigators noted.
Several baseline characteristics, including male sex, severe chronic obstructive pulmonary disease, dialysis-dependent end-stage renal disease, older age, higher STS Predicted Risk of Operative Mortality (PROM) score, a history of atrial fibrillation/flutter, and use of an access route (other than transfemoral), were found to be independently associated with higher 1-year mortality. Thus, “It may be possible to identify patients who may not benefit from this procedure and who should be counseled accordingly.” For example, in this study there was a small (77 patients) very high-risk subset of patients – aged 85-94 years, dependent on dialysis, and having an STS PROM score greater than 15% – whose 1-year mortality was 53.5%.
The STS and the ACC supported this study, and support the Transcatheter Valve Therapies Registry. Dr. Holmes reported having no relevant financial disclosures; his associates reported ties to Boston Scientific, Edwards Lifesciences, Janssen, Eli Lilly, Boehringer Ingelheim, Bayer, and AstraZeneca.
One year after transcatheter aortic valve replacement in the United States, the overall mortality was 23.7%, the stroke rate was 4.1%, and the composite outcome of death and stroke was 26.0%, according to a report published online March 10 in JAMA.
Long-term outcomes for TAVR haven’t been well studied until now, yet the procedure is being performed with increasing frequency for aortic stenosis in patients who are too high risk to undergo conventional surgical aortic valve replacement, said Dr. David R. Holmes Jr. of the Mayo Clinic, Rochester, Minn., and his associates.
They assessed 1-year outcomes by analyzing administrative data from the Centers for Medicare & Medicaid Services and clinical data from the Transcatheter Valve Therapies Registry, an initiative of the Society of Thoracic Surgeons and the American College of Cardiology. The study involved 12,182 patients who underwent TAVR at 299 medical centers across the country during a 19-month period. The patients’ median age was 84 years; 95% were white and 52% were women. The transfemoral approach was used in most patients, but alternative approaches were used in roughly 44%. As expected for an elderly, high-risk study population, baseline functional status was poor and comorbidities were common. They included reduced left ventricular ejection fraction (26% of patients), prior stroke (12%), moderate or severe lung disease (28%), renal failure (16%), peripheral vascular disease (32%), and atrial fibrillation (42%), Dr. Holmes and his associates reported (JAMA 2015 March 10 [doi:10.1001/jama.2015.1474]).
In addition to the mortality and stroke rates listed above, the 1-year rate of one rehospitalization was 24.4%, that of two rehospitalizations was 12.5%, and that of three or more rehospitalizations was 11.6%. The 1-year readmission rate specifically for stroke, heart failure, or repeat aortic valve intervention was 18.6%. These are important considerations for elderly, fragile patients because rehospitalizations indicate “an unacceptable quality-of-life outcome” and are very costly, the investigators noted.
Several baseline characteristics, including male sex, severe chronic obstructive pulmonary disease, dialysis-dependent end-stage renal disease, older age, higher STS Predicted Risk of Operative Mortality (PROM) score, a history of atrial fibrillation/flutter, and use of an access route (other than transfemoral), were found to be independently associated with higher 1-year mortality. Thus, “It may be possible to identify patients who may not benefit from this procedure and who should be counseled accordingly.” For example, in this study there was a small (77 patients) very high-risk subset of patients – aged 85-94 years, dependent on dialysis, and having an STS PROM score greater than 15% – whose 1-year mortality was 53.5%.
The STS and the ACC supported this study, and support the Transcatheter Valve Therapies Registry. Dr. Holmes reported having no relevant financial disclosures; his associates reported ties to Boston Scientific, Edwards Lifesciences, Janssen, Eli Lilly, Boehringer Ingelheim, Bayer, and AstraZeneca.
FROM JAMA
Key clinical point: Severe chronic obstructive pulmonary disease, end-stage renal disease, older age, higher STS PROM score, and use of an access route (other than transfemoral) are among the baseline characteristics linked with higher 1-year mortality after TAVR.
Major finding: The overall mortality was 23.7%, the stroke rate was 4.1%, and the composite rate of death and stroke was 26% 1 year after TAVR.
Data source: An analysis of data from the Transcatheter Valve Therapies Registry for 12,182 patients who underwent TAVR procedures at 299 U.S. hospitals during a 19-month period.
Disclosures: The Society of Thoracic Surgeons and the American College of Cardiology supported this study, and support the Transcatheter Valve Therapies Registry. Dr. Holmes reported having no relevant financial disclosures; his associates reported ties to Boston Scientific, Edwards Lifesciences, Janssen, Eli Lilly, Boehringer Ingelheim, Bayer, and AstraZeneca.