User login
Brodalumab, Ixekizumab Rapidly Improve Psoriasis
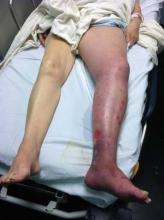
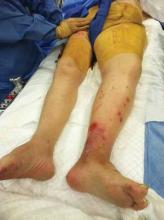
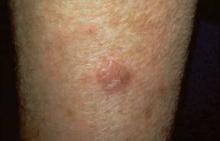
Two monoclonal antibodies targeting interleukin-17 proved effective and safe for moderate to severe plaque psoriasis, according to the results of two phase II clinical trials reported in the March 29 issue of the New England Journal of Medicine.
Both agents had a rapid onset of action and improved scores on the psoriasis area and severity (PASI) index, as well as on measures of dermatology-related quality of life and overall well-being, compared with placebo. Both agents also showed increasing efficacy at higher doses, indicating a dose-response effect, according to the industry-sponsored studies.
Brodalumab
In the first study, researchers assessed brodalumab (AMG 827), a human anti-interleukin-17RA monoclonal antibody that antagonizes the interleukin-17 pathway, in 198 patients at 23 international sites.
The study patients were randomly assigned to receive 70 mg, 140 mg, or 210 mg of brodalumab (at weeks 1, 2, 4, 6, 8, and 10), 280 mg of brodalumab monthly, or placebo injections. A subgroup of 20 patients also underwent skin biopsy at baseline and at week 12, reported Dr. Kim A. Papp of Probity Medical Research, Waterloo, Ont., and his associates.
The mean patient age was 43 years, and the mean duration of psoriasis was 19 years. Most patients had previously received topical and systemic treatments, including phototherapy, and 35% had previously received biologic therapy.
The primary efficacy outcome – percentage improvement in PASI score at week 12 – was significantly greater in every brodalumab group than in the placebo group. Mean improvements were 45% with 70 mg, 85.9% with 140 mg, 86.3% with 210 mg, and 76% with 280 mg, and 16% with placebo.
"Similarly, a significantly higher percentage of patients in the brodalumab groups than in the placebo group [was] assessed as being clear of psoriasis (score of 0 on the physician’s global assessment) or as having minimal disease (score of 1) at week 12," Dr. Papp and his colleagues wrote (N. Engl. J. Med. 2012;366:1181-9)
In addition, the extent of the affected body-surface area was significantly smaller with active treatment than with placebo. Scores on the Dermatology Life Quality Index (DLQI) were significantly lower in the brodalumab groups, indicating that psoriasis had less of an impact on health-related quality of life. And scores on both physical and mental components of the SF-36 were significantly higher, indicating improved well-being, with brodalumab.
Among study patients who underwent skin biopsy, keratin 16 staining of the upper epidermis was reduced, and epidermal thickness and CD3 counts were decreased, at all doses of brodalumab except 70 mg. "This finding indicates a reversal of regenerative epidermal maturation, which is the essential pathological characteristic of psoriasis that causes epidermal hyperplasia and abnormal differentiation, with scaling and thickness as the clinical correlates," Dr. Papp and his associates noted.
At 16-week follow-up, which was 6-8 weeks after the final dose of brodalumab, PASI scores showed a diminished drug effect but were still better than those at baseline.
There were three serious adverse events, only one of which was related to the study drug. This was an asymptomatic case of grade 3 neutropenia, which resolved when the study drug was discontinued. A less serious case of neutropenia developed in another patient.
Ixekizumab
The second study assessed ixekizumab (LY2439821), a humanized IgG4 monoclonal antibody that neutralizes interleukin-17, in 142 patients with moderate to severe plaque psoriasis. The study patients were randomly assigned to receive subcutaneous injections of placebo or 10 mg, 25 mg, 75 mg, or 150 mg of ixekizumab at baseline and at 2, 4, 8, 12, and 16 weeks, reported Dr. Craig Leonardi of the department of dermatology, St. Louis University, and his associates.
The primary efficacy end point was the proportion of patients who achieved at least a 75% improvement in PASI score at 12 weeks. Significantly more patients at all ixekizumab doses except 10 mg achieved this benchmark (76.7%-82.8%), compared with placebo (7.7%).
In addition, significantly more patients (approximately 40%) in the higher-dose groups achieved a 100% improvement in PASI scores, indicating complete clearance of psoriasis, than in the placebo group. And significantly more patients receiving active treatment received physician’s global assessment scores of 0, indicating complete clearance, or 1, indicating minimal disease.
As with brodalumab, the beneficial effect of ixekizumab was evident as early as 1 week into treatment. The drug also significantly improved DLQI scores and decreased the severity of itching, Dr. Leonardi and his colleagues reported (New Engl. J. Med. 2012;366:1190-9).
Of note, ixekizumab was significantly more effective than placebo in patients who had psoriasis in difficult to treat areas such as the scalp and nails.
There were no serious adverse events. Two patients developed grade 2 neutropenia. Two patients showed elevations in liver enzymes that subsided when the drug was discontinued.
Both groups of investigators said their findings demonstrate that interleukin-17 is an appropriate target for psoriasis treatment. They also noted that the phase-II studies were too small and too brief to assess these agents’ effects on the risk of infection and cardiovascular events, and noted that further, longer-term studies of safety and efficacy are needed.
Even though there were few adverse effects in these two clinical trials and few patients withdrew from the studies, "a 12-week follow-up period is too short to assess the safety of treatments targeting interleukin-17," said Ari Waisman, Ph.D., in remarks taken from his editorial accompanying the reports on brodalumab and ixekizumab (N. Engl. J. Med. 2012;366:1251-2).
Despite the patients’ marked improvements with both brodalumab and ixekizumab, "future trials involving larger numbers of patients treated and followed for a much longer period of time will be needed," Dr. Waisman of the Institute for Molecular Medicine at Johannes-Gutenberg University of Mainz (Germany) said.
Dr. Papp’s study was funded by Amgen; Dr. Papp and his associates reported ties to numerous industry sources. Dr. Leonardi’s study was funded by Eli Lilly; Dr. Leonardi reported ties to Abbott, Amgen, Celgene, Centocor, Eli Lilly, Pfizer, Galderma, Incyte, Maruho, Schering-Plough, Sirtris, Stiefel, Novartis, Novo Nordisk, Vascular Biogenics, and Wyeth. Dr. Waisman reported ties to Teva, Phenex, and GlaxoSmithKline.
Even though there were few adverse effects in these two clinical trials and few patients withdrew from the studies, "a 12-week follow-up period is too short to assess the safety of treatments targeting interleukin-17," said Dr. Ari Waisman.
Despite the patients’ marked improvements with both brodalumab and ixekizumab, "future trials involving larger numbers of patients treated and followed for a much longer period of time will be needed," he said.
Ari Waisman, Ph.D., is at the Institute for Molecular Medicine at Johannes-Gutenberg University of Mainz (Germany). He reported ties to Teva, Phenex, and GlaxoSmithKline. These remarks were taken from his editorial accompanying the reports on brodalumab and ixekizumab (N. Engl. J. Med. 2012;366:1251-2).
Even though there were few adverse effects in these two clinical trials and few patients withdrew from the studies, "a 12-week follow-up period is too short to assess the safety of treatments targeting interleukin-17," said Dr. Ari Waisman.
Despite the patients’ marked improvements with both brodalumab and ixekizumab, "future trials involving larger numbers of patients treated and followed for a much longer period of time will be needed," he said.
Ari Waisman, Ph.D., is at the Institute for Molecular Medicine at Johannes-Gutenberg University of Mainz (Germany). He reported ties to Teva, Phenex, and GlaxoSmithKline. These remarks were taken from his editorial accompanying the reports on brodalumab and ixekizumab (N. Engl. J. Med. 2012;366:1251-2).
Even though there were few adverse effects in these two clinical trials and few patients withdrew from the studies, "a 12-week follow-up period is too short to assess the safety of treatments targeting interleukin-17," said Dr. Ari Waisman.
Despite the patients’ marked improvements with both brodalumab and ixekizumab, "future trials involving larger numbers of patients treated and followed for a much longer period of time will be needed," he said.
Ari Waisman, Ph.D., is at the Institute for Molecular Medicine at Johannes-Gutenberg University of Mainz (Germany). He reported ties to Teva, Phenex, and GlaxoSmithKline. These remarks were taken from his editorial accompanying the reports on brodalumab and ixekizumab (N. Engl. J. Med. 2012;366:1251-2).
Two monoclonal antibodies targeting interleukin-17 proved effective and safe for moderate to severe plaque psoriasis, according to the results of two phase II clinical trials reported in the March 29 issue of the New England Journal of Medicine.
Both agents had a rapid onset of action and improved scores on the psoriasis area and severity (PASI) index, as well as on measures of dermatology-related quality of life and overall well-being, compared with placebo. Both agents also showed increasing efficacy at higher doses, indicating a dose-response effect, according to the industry-sponsored studies.
Brodalumab
In the first study, researchers assessed brodalumab (AMG 827), a human anti-interleukin-17RA monoclonal antibody that antagonizes the interleukin-17 pathway, in 198 patients at 23 international sites.
The study patients were randomly assigned to receive 70 mg, 140 mg, or 210 mg of brodalumab (at weeks 1, 2, 4, 6, 8, and 10), 280 mg of brodalumab monthly, or placebo injections. A subgroup of 20 patients also underwent skin biopsy at baseline and at week 12, reported Dr. Kim A. Papp of Probity Medical Research, Waterloo, Ont., and his associates.
The mean patient age was 43 years, and the mean duration of psoriasis was 19 years. Most patients had previously received topical and systemic treatments, including phototherapy, and 35% had previously received biologic therapy.
The primary efficacy outcome – percentage improvement in PASI score at week 12 – was significantly greater in every brodalumab group than in the placebo group. Mean improvements were 45% with 70 mg, 85.9% with 140 mg, 86.3% with 210 mg, and 76% with 280 mg, and 16% with placebo.
"Similarly, a significantly higher percentage of patients in the brodalumab groups than in the placebo group [was] assessed as being clear of psoriasis (score of 0 on the physician’s global assessment) or as having minimal disease (score of 1) at week 12," Dr. Papp and his colleagues wrote (N. Engl. J. Med. 2012;366:1181-9)
In addition, the extent of the affected body-surface area was significantly smaller with active treatment than with placebo. Scores on the Dermatology Life Quality Index (DLQI) were significantly lower in the brodalumab groups, indicating that psoriasis had less of an impact on health-related quality of life. And scores on both physical and mental components of the SF-36 were significantly higher, indicating improved well-being, with brodalumab.
Among study patients who underwent skin biopsy, keratin 16 staining of the upper epidermis was reduced, and epidermal thickness and CD3 counts were decreased, at all doses of brodalumab except 70 mg. "This finding indicates a reversal of regenerative epidermal maturation, which is the essential pathological characteristic of psoriasis that causes epidermal hyperplasia and abnormal differentiation, with scaling and thickness as the clinical correlates," Dr. Papp and his associates noted.
At 16-week follow-up, which was 6-8 weeks after the final dose of brodalumab, PASI scores showed a diminished drug effect but were still better than those at baseline.
There were three serious adverse events, only one of which was related to the study drug. This was an asymptomatic case of grade 3 neutropenia, which resolved when the study drug was discontinued. A less serious case of neutropenia developed in another patient.
Ixekizumab
The second study assessed ixekizumab (LY2439821), a humanized IgG4 monoclonal antibody that neutralizes interleukin-17, in 142 patients with moderate to severe plaque psoriasis. The study patients were randomly assigned to receive subcutaneous injections of placebo or 10 mg, 25 mg, 75 mg, or 150 mg of ixekizumab at baseline and at 2, 4, 8, 12, and 16 weeks, reported Dr. Craig Leonardi of the department of dermatology, St. Louis University, and his associates.
The primary efficacy end point was the proportion of patients who achieved at least a 75% improvement in PASI score at 12 weeks. Significantly more patients at all ixekizumab doses except 10 mg achieved this benchmark (76.7%-82.8%), compared with placebo (7.7%).
In addition, significantly more patients (approximately 40%) in the higher-dose groups achieved a 100% improvement in PASI scores, indicating complete clearance of psoriasis, than in the placebo group. And significantly more patients receiving active treatment received physician’s global assessment scores of 0, indicating complete clearance, or 1, indicating minimal disease.
As with brodalumab, the beneficial effect of ixekizumab was evident as early as 1 week into treatment. The drug also significantly improved DLQI scores and decreased the severity of itching, Dr. Leonardi and his colleagues reported (New Engl. J. Med. 2012;366:1190-9).
Of note, ixekizumab was significantly more effective than placebo in patients who had psoriasis in difficult to treat areas such as the scalp and nails.
There were no serious adverse events. Two patients developed grade 2 neutropenia. Two patients showed elevations in liver enzymes that subsided when the drug was discontinued.
Both groups of investigators said their findings demonstrate that interleukin-17 is an appropriate target for psoriasis treatment. They also noted that the phase-II studies were too small and too brief to assess these agents’ effects on the risk of infection and cardiovascular events, and noted that further, longer-term studies of safety and efficacy are needed.
Even though there were few adverse effects in these two clinical trials and few patients withdrew from the studies, "a 12-week follow-up period is too short to assess the safety of treatments targeting interleukin-17," said Ari Waisman, Ph.D., in remarks taken from his editorial accompanying the reports on brodalumab and ixekizumab (N. Engl. J. Med. 2012;366:1251-2).
Despite the patients’ marked improvements with both brodalumab and ixekizumab, "future trials involving larger numbers of patients treated and followed for a much longer period of time will be needed," Dr. Waisman of the Institute for Molecular Medicine at Johannes-Gutenberg University of Mainz (Germany) said.
Dr. Papp’s study was funded by Amgen; Dr. Papp and his associates reported ties to numerous industry sources. Dr. Leonardi’s study was funded by Eli Lilly; Dr. Leonardi reported ties to Abbott, Amgen, Celgene, Centocor, Eli Lilly, Pfizer, Galderma, Incyte, Maruho, Schering-Plough, Sirtris, Stiefel, Novartis, Novo Nordisk, Vascular Biogenics, and Wyeth. Dr. Waisman reported ties to Teva, Phenex, and GlaxoSmithKline.
Two monoclonal antibodies targeting interleukin-17 proved effective and safe for moderate to severe plaque psoriasis, according to the results of two phase II clinical trials reported in the March 29 issue of the New England Journal of Medicine.
Both agents had a rapid onset of action and improved scores on the psoriasis area and severity (PASI) index, as well as on measures of dermatology-related quality of life and overall well-being, compared with placebo. Both agents also showed increasing efficacy at higher doses, indicating a dose-response effect, according to the industry-sponsored studies.
Brodalumab
In the first study, researchers assessed brodalumab (AMG 827), a human anti-interleukin-17RA monoclonal antibody that antagonizes the interleukin-17 pathway, in 198 patients at 23 international sites.
The study patients were randomly assigned to receive 70 mg, 140 mg, or 210 mg of brodalumab (at weeks 1, 2, 4, 6, 8, and 10), 280 mg of brodalumab monthly, or placebo injections. A subgroup of 20 patients also underwent skin biopsy at baseline and at week 12, reported Dr. Kim A. Papp of Probity Medical Research, Waterloo, Ont., and his associates.
The mean patient age was 43 years, and the mean duration of psoriasis was 19 years. Most patients had previously received topical and systemic treatments, including phototherapy, and 35% had previously received biologic therapy.
The primary efficacy outcome – percentage improvement in PASI score at week 12 – was significantly greater in every brodalumab group than in the placebo group. Mean improvements were 45% with 70 mg, 85.9% with 140 mg, 86.3% with 210 mg, and 76% with 280 mg, and 16% with placebo.
"Similarly, a significantly higher percentage of patients in the brodalumab groups than in the placebo group [was] assessed as being clear of psoriasis (score of 0 on the physician’s global assessment) or as having minimal disease (score of 1) at week 12," Dr. Papp and his colleagues wrote (N. Engl. J. Med. 2012;366:1181-9)
In addition, the extent of the affected body-surface area was significantly smaller with active treatment than with placebo. Scores on the Dermatology Life Quality Index (DLQI) were significantly lower in the brodalumab groups, indicating that psoriasis had less of an impact on health-related quality of life. And scores on both physical and mental components of the SF-36 were significantly higher, indicating improved well-being, with brodalumab.
Among study patients who underwent skin biopsy, keratin 16 staining of the upper epidermis was reduced, and epidermal thickness and CD3 counts were decreased, at all doses of brodalumab except 70 mg. "This finding indicates a reversal of regenerative epidermal maturation, which is the essential pathological characteristic of psoriasis that causes epidermal hyperplasia and abnormal differentiation, with scaling and thickness as the clinical correlates," Dr. Papp and his associates noted.
At 16-week follow-up, which was 6-8 weeks after the final dose of brodalumab, PASI scores showed a diminished drug effect but were still better than those at baseline.
There were three serious adverse events, only one of which was related to the study drug. This was an asymptomatic case of grade 3 neutropenia, which resolved when the study drug was discontinued. A less serious case of neutropenia developed in another patient.
Ixekizumab
The second study assessed ixekizumab (LY2439821), a humanized IgG4 monoclonal antibody that neutralizes interleukin-17, in 142 patients with moderate to severe plaque psoriasis. The study patients were randomly assigned to receive subcutaneous injections of placebo or 10 mg, 25 mg, 75 mg, or 150 mg of ixekizumab at baseline and at 2, 4, 8, 12, and 16 weeks, reported Dr. Craig Leonardi of the department of dermatology, St. Louis University, and his associates.
The primary efficacy end point was the proportion of patients who achieved at least a 75% improvement in PASI score at 12 weeks. Significantly more patients at all ixekizumab doses except 10 mg achieved this benchmark (76.7%-82.8%), compared with placebo (7.7%).
In addition, significantly more patients (approximately 40%) in the higher-dose groups achieved a 100% improvement in PASI scores, indicating complete clearance of psoriasis, than in the placebo group. And significantly more patients receiving active treatment received physician’s global assessment scores of 0, indicating complete clearance, or 1, indicating minimal disease.
As with brodalumab, the beneficial effect of ixekizumab was evident as early as 1 week into treatment. The drug also significantly improved DLQI scores and decreased the severity of itching, Dr. Leonardi and his colleagues reported (New Engl. J. Med. 2012;366:1190-9).
Of note, ixekizumab was significantly more effective than placebo in patients who had psoriasis in difficult to treat areas such as the scalp and nails.
There were no serious adverse events. Two patients developed grade 2 neutropenia. Two patients showed elevations in liver enzymes that subsided when the drug was discontinued.
Both groups of investigators said their findings demonstrate that interleukin-17 is an appropriate target for psoriasis treatment. They also noted that the phase-II studies were too small and too brief to assess these agents’ effects on the risk of infection and cardiovascular events, and noted that further, longer-term studies of safety and efficacy are needed.
Even though there were few adverse effects in these two clinical trials and few patients withdrew from the studies, "a 12-week follow-up period is too short to assess the safety of treatments targeting interleukin-17," said Ari Waisman, Ph.D., in remarks taken from his editorial accompanying the reports on brodalumab and ixekizumab (N. Engl. J. Med. 2012;366:1251-2).
Despite the patients’ marked improvements with both brodalumab and ixekizumab, "future trials involving larger numbers of patients treated and followed for a much longer period of time will be needed," Dr. Waisman of the Institute for Molecular Medicine at Johannes-Gutenberg University of Mainz (Germany) said.
Dr. Papp’s study was funded by Amgen; Dr. Papp and his associates reported ties to numerous industry sources. Dr. Leonardi’s study was funded by Eli Lilly; Dr. Leonardi reported ties to Abbott, Amgen, Celgene, Centocor, Eli Lilly, Pfizer, Galderma, Incyte, Maruho, Schering-Plough, Sirtris, Stiefel, Novartis, Novo Nordisk, Vascular Biogenics, and Wyeth. Dr. Waisman reported ties to Teva, Phenex, and GlaxoSmithKline.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: Mean improvements in PASI scores, with brodalumab were 45% with 70 mg, 86% with 140 mg, 86% with 210 mg, and 76% with 280 mg. Those given placebo had a 16% mean improvement in PASI.
Data Source: The brodalumab data were taken from a 12-week phase II randomized trial with 198 subjects.
Disclosures: Dr. Papp’s study was funded by Amgen; Dr. Papp and his associates reported ties to numerous industry sources. Dr. Leonardi’s study was funded by Eli Lilly; Dr. Leonardi reported ties to Abbott, Amgen, Celgene, Centocor, Eli Lilly, Pfizer, Galderma, Incyte, Maruho, Schering-Plough, Sirtris, Stiefel, Novartis, Novo Nordisk, Vascular Biogenics, and Wyeth.
Cost Sharing Linked to Rise in Asthma Hospitalizations
Private health plans’ use of cost sharing for prescription drugs – the practice of shifting more of the expense of medications onto the patient – apparently has led parents of children with asthma to reduce their use of control therapy, according to a report in the March 28 issue of JAMA.
Higher out-of-pocket medication costs correlated with a significant reduction in the percentage of days covered by an asthma medication among privately insured children aged 5-18 years, and a simultaneous rise in the number of asthma-related hospitalizations, said Pinar Karaca-Mandic, Ph.D., of the division of health policy and management at the University of Minnesota School of Public Health, Minneapolis, and her associates.
Numerous studies have examined the effect of medication cost sharing on adults, consistently linking it to reduced medication use and increases in hospitalizations and emergency department visits. However, the effects on children "have been overlooked."
Dr. Karaca-Mandic and her colleagues used data on pharmacy and medical claims from a benefits consulting firm to assess how out-of-pocket drug costs affected these factors in children newly diagnosed with persistent asthma requiring long-acting control therapy.
They analyzed data on 8,834 de-identified patients covered by private health insurance provided by 37 geographically diverse U.S. employers from 1997 through 2008. The children were prescribed inhaled corticosteroids, long-acting beta-agonists, leukotriene receptor antagonists, methylxanthines, cromolyn sodium, and immunomodulators to be taken every day year-round.
The investigators followed the patients for 1 year, determining the percentage of days in which these asthma control drugs were supplied. They also calculated parents’ mean out-of-pocket costs for the drugs.
Since asthma is managed somewhat differently as children age, the researchers divided their study subjects into younger (2,921 subjects younger than 5 years) or older (5,913 subjects aged 5-18 years) groups. Approximately 60% of both age groups were prescribed more than one asthma medication.
The mean use of asthma control medications was low in both age groups, at 40%-46% of the year, Dr. Karaca-Mandic and her associates said (JAMA 2012;307:1284-91).
In the older age group, higher out-of-pocket drug costs were associated with a significant reduction in percentage of days covered by an asthma control medication, from approximately 42% to 40%. This association was not seen in younger children.
Similarly, in the older age group, higher out-of-pocket drug costs were associated with a significant increase in asthma-related hospitalizations but not other hospitalizations, from 1.7 to 2.4 per year. This association also was not seen in younger children.
Out-of-pocket drug costs did not significantly affect rates of ED visits in either age group.
The lack of an association between drug costs and drug usage in younger children "suggests that parents may be less sensitive to medication costs for younger children, who traditionally have more severe disease. In addition, parents may play a more active role in disease management for younger children with asthma compared with that for adolescents," the researchers said.
Overall, the study findings indicate that "greater prescription medication cost sharing among children with asthma may lead to small reductions in use of important medications, with unintended consequences of more frequent asthma-related hospitalizations," they said.
This study was limited in that it did not include information on family income or on asthma severity, the researchers added.
This study was funded by the National Institute of Child Health and Human Development, the National Institute on Aging, and the Roybal Center for Health Policy Simulation. Dr. Karaca-Mandic reported no financial conflicts. Her associates reported ties to Precision Health Economics, Pfizer, Novartis, and Bristol-Myers Squibb.
Since this study included only data on long-acting controller medications, it could not examine the possibility that parents were substituting the less expensive short-acting beta-agonist bronchodilators for these more costly agents, said Wendy J. Ungar, Ph.D.
There are many generic bronchodilators, but the longer-acting inhaled corticosteroids and dual agents are usually brand-name products and are the most expensive asthma treatments, "often costing upward of $100 per month," she noted.
"When faced with a fixed household spending budget, a parent may choose the wrong drug to refill," Dr. Ungar said.
Dr. Ungar is in the Child Health Evaluative Sciences program at the Hospital for Sick Children, Toronto, and in the Institute of Health Policy, Management, and Evaluation at the University of Toronto. She reported no relevant financial conflicts. These remarks were taken from her editorial accompanying Dr. Karaca-Mandic’s report (JAMA 2012;307:1316-18).
Since this study included only data on long-acting controller medications, it could not examine the possibility that parents were substituting the less expensive short-acting beta-agonist bronchodilators for these more costly agents, said Wendy J. Ungar, Ph.D.
There are many generic bronchodilators, but the longer-acting inhaled corticosteroids and dual agents are usually brand-name products and are the most expensive asthma treatments, "often costing upward of $100 per month," she noted.
"When faced with a fixed household spending budget, a parent may choose the wrong drug to refill," Dr. Ungar said.
Dr. Ungar is in the Child Health Evaluative Sciences program at the Hospital for Sick Children, Toronto, and in the Institute of Health Policy, Management, and Evaluation at the University of Toronto. She reported no relevant financial conflicts. These remarks were taken from her editorial accompanying Dr. Karaca-Mandic’s report (JAMA 2012;307:1316-18).
Since this study included only data on long-acting controller medications, it could not examine the possibility that parents were substituting the less expensive short-acting beta-agonist bronchodilators for these more costly agents, said Wendy J. Ungar, Ph.D.
There are many generic bronchodilators, but the longer-acting inhaled corticosteroids and dual agents are usually brand-name products and are the most expensive asthma treatments, "often costing upward of $100 per month," she noted.
"When faced with a fixed household spending budget, a parent may choose the wrong drug to refill," Dr. Ungar said.
Dr. Ungar is in the Child Health Evaluative Sciences program at the Hospital for Sick Children, Toronto, and in the Institute of Health Policy, Management, and Evaluation at the University of Toronto. She reported no relevant financial conflicts. These remarks were taken from her editorial accompanying Dr. Karaca-Mandic’s report (JAMA 2012;307:1316-18).
Private health plans’ use of cost sharing for prescription drugs – the practice of shifting more of the expense of medications onto the patient – apparently has led parents of children with asthma to reduce their use of control therapy, according to a report in the March 28 issue of JAMA.
Higher out-of-pocket medication costs correlated with a significant reduction in the percentage of days covered by an asthma medication among privately insured children aged 5-18 years, and a simultaneous rise in the number of asthma-related hospitalizations, said Pinar Karaca-Mandic, Ph.D., of the division of health policy and management at the University of Minnesota School of Public Health, Minneapolis, and her associates.
Numerous studies have examined the effect of medication cost sharing on adults, consistently linking it to reduced medication use and increases in hospitalizations and emergency department visits. However, the effects on children "have been overlooked."
Dr. Karaca-Mandic and her colleagues used data on pharmacy and medical claims from a benefits consulting firm to assess how out-of-pocket drug costs affected these factors in children newly diagnosed with persistent asthma requiring long-acting control therapy.
They analyzed data on 8,834 de-identified patients covered by private health insurance provided by 37 geographically diverse U.S. employers from 1997 through 2008. The children were prescribed inhaled corticosteroids, long-acting beta-agonists, leukotriene receptor antagonists, methylxanthines, cromolyn sodium, and immunomodulators to be taken every day year-round.
The investigators followed the patients for 1 year, determining the percentage of days in which these asthma control drugs were supplied. They also calculated parents’ mean out-of-pocket costs for the drugs.
Since asthma is managed somewhat differently as children age, the researchers divided their study subjects into younger (2,921 subjects younger than 5 years) or older (5,913 subjects aged 5-18 years) groups. Approximately 60% of both age groups were prescribed more than one asthma medication.
The mean use of asthma control medications was low in both age groups, at 40%-46% of the year, Dr. Karaca-Mandic and her associates said (JAMA 2012;307:1284-91).
In the older age group, higher out-of-pocket drug costs were associated with a significant reduction in percentage of days covered by an asthma control medication, from approximately 42% to 40%. This association was not seen in younger children.
Similarly, in the older age group, higher out-of-pocket drug costs were associated with a significant increase in asthma-related hospitalizations but not other hospitalizations, from 1.7 to 2.4 per year. This association also was not seen in younger children.
Out-of-pocket drug costs did not significantly affect rates of ED visits in either age group.
The lack of an association between drug costs and drug usage in younger children "suggests that parents may be less sensitive to medication costs for younger children, who traditionally have more severe disease. In addition, parents may play a more active role in disease management for younger children with asthma compared with that for adolescents," the researchers said.
Overall, the study findings indicate that "greater prescription medication cost sharing among children with asthma may lead to small reductions in use of important medications, with unintended consequences of more frequent asthma-related hospitalizations," they said.
This study was limited in that it did not include information on family income or on asthma severity, the researchers added.
This study was funded by the National Institute of Child Health and Human Development, the National Institute on Aging, and the Roybal Center for Health Policy Simulation. Dr. Karaca-Mandic reported no financial conflicts. Her associates reported ties to Precision Health Economics, Pfizer, Novartis, and Bristol-Myers Squibb.
Private health plans’ use of cost sharing for prescription drugs – the practice of shifting more of the expense of medications onto the patient – apparently has led parents of children with asthma to reduce their use of control therapy, according to a report in the March 28 issue of JAMA.
Higher out-of-pocket medication costs correlated with a significant reduction in the percentage of days covered by an asthma medication among privately insured children aged 5-18 years, and a simultaneous rise in the number of asthma-related hospitalizations, said Pinar Karaca-Mandic, Ph.D., of the division of health policy and management at the University of Minnesota School of Public Health, Minneapolis, and her associates.
Numerous studies have examined the effect of medication cost sharing on adults, consistently linking it to reduced medication use and increases in hospitalizations and emergency department visits. However, the effects on children "have been overlooked."
Dr. Karaca-Mandic and her colleagues used data on pharmacy and medical claims from a benefits consulting firm to assess how out-of-pocket drug costs affected these factors in children newly diagnosed with persistent asthma requiring long-acting control therapy.
They analyzed data on 8,834 de-identified patients covered by private health insurance provided by 37 geographically diverse U.S. employers from 1997 through 2008. The children were prescribed inhaled corticosteroids, long-acting beta-agonists, leukotriene receptor antagonists, methylxanthines, cromolyn sodium, and immunomodulators to be taken every day year-round.
The investigators followed the patients for 1 year, determining the percentage of days in which these asthma control drugs were supplied. They also calculated parents’ mean out-of-pocket costs for the drugs.
Since asthma is managed somewhat differently as children age, the researchers divided their study subjects into younger (2,921 subjects younger than 5 years) or older (5,913 subjects aged 5-18 years) groups. Approximately 60% of both age groups were prescribed more than one asthma medication.
The mean use of asthma control medications was low in both age groups, at 40%-46% of the year, Dr. Karaca-Mandic and her associates said (JAMA 2012;307:1284-91).
In the older age group, higher out-of-pocket drug costs were associated with a significant reduction in percentage of days covered by an asthma control medication, from approximately 42% to 40%. This association was not seen in younger children.
Similarly, in the older age group, higher out-of-pocket drug costs were associated with a significant increase in asthma-related hospitalizations but not other hospitalizations, from 1.7 to 2.4 per year. This association also was not seen in younger children.
Out-of-pocket drug costs did not significantly affect rates of ED visits in either age group.
The lack of an association between drug costs and drug usage in younger children "suggests that parents may be less sensitive to medication costs for younger children, who traditionally have more severe disease. In addition, parents may play a more active role in disease management for younger children with asthma compared with that for adolescents," the researchers said.
Overall, the study findings indicate that "greater prescription medication cost sharing among children with asthma may lead to small reductions in use of important medications, with unintended consequences of more frequent asthma-related hospitalizations," they said.
This study was limited in that it did not include information on family income or on asthma severity, the researchers added.
This study was funded by the National Institute of Child Health and Human Development, the National Institute on Aging, and the Roybal Center for Health Policy Simulation. Dr. Karaca-Mandic reported no financial conflicts. Her associates reported ties to Precision Health Economics, Pfizer, Novartis, and Bristol-Myers Squibb.
FROM JAMA
Major Finding: Higher out-of-pocket costs for prescription drugs correlated with a significant 2% reduction in the use of asthma control medications and a significant increase in the number of asthma-related hospitalizations per year.
Data Source: Data were taken from an analysis of pharmacy and medical claims data for 8,834 privately insured U.S. children newly diagnosed as having persistent asthma requiring long-acting control therapy during 1997-2008.
Disclosures: This study was funded by the National Institute of Child Health and Human Development, the National Institute on Aging, and the Roybal Center for Health Policy Simulation. Dr. Karaca-Mandic reported no financial conflicts. Her associates reported ties to Precision Health Economics, Pfizer, Novartis, and Bristol-Myers Squibb.
Cardiac Pacing Improves Survival in Myotonic Dystrophy Type 1
A strategy of implanting permanent cardiac pacing markedly improved survival for patients with myotonic dystrophy type 1 who presented with surface electrocardiogram manifestations of cardiac conduction abnormalities, compared with a noninvasive strategy of regular surveillance, according to a retrospective cohort study published March 28 in JAMA.
The likelihood of dying during a median 7.4 years of follow-up was nearly 40% lower among patients who underwent electrophysiological testing and, potentially, prophylactic pacing, than among those who did not undergo electrophysiological testing or pacing. The difference between the two groups was largely attributed to a decrease in sudden death in those who received a pacemaker or implantable cardioverter-defibrillator (ICD), said Dr. Karim Wahbi of the departments of cardiology and neurology at Cochin Hospital and the Myology Institute at Pitié-Salpêtrière Hospital, both in Paris, and his associates.
The study findings suggest that conduction system disease "is a major cause of sudden death that appears to be preventable" in this patient population, they noted.
Myotonic dystrophy type 1, also known as Steinert disease, is the most common inherited neuromuscular disease in adults and is characterized by muscle weakness, myotonia, multiple endocrine disorders, respiratory insufficiency, and cardiac abnormalities. As many as one-third of patients succumb to sudden death.
Both the American College of Cardiology and the American Heart Association recommend permanent pacing in patients with myotonic dystrophy type 1 when complete atrioventricular block or advanced high-degree atrioventricular block are detected, or prophylactically when first-degree atrioventricular or fascicular block are detected on ECG. However, whether this invasive approach actually prevents sudden death has never been assessed in a clinical study until now, Dr. Wahbi and his colleagues said.
Of 914 consecutive adults in the DM1 Heart Registry at the Myology Institute who were admitted during 2000-2009 for management of myotonic dystrophy type 1, 486 had ECGs showing minor conduction defects.
Of these subjects, 341 were managed invasively on the advice of their cardiologists. Electrophysiological testing in these patients found an HV interval greater than 70 ms in 164 patients and sustained ventricular tachyarrhythmias that were induced by programmed ventricular stimulation in 70 patients. The patients with an HV interval greater than 70 ms underwent placement of a pacemaker (150) or received an ICD (14). The remaining patients in the invasive-strategy group did not undergo a device implantation.
The remaining 145 subjects were managed noninvasively on the advice of their cardiologists. They had no electrophysiological study and no implantation of pacing devices, but were followed yearly by a neurologist and a cardiologist, both of whom specialized in muscular diseases.
Overall, 49 patients received antiarrhythmic drugs, including amiodarone and beta-blockers.
During a median follow-up of 7.4 years (range, 0-9.9 years), 80 patients died (50 in the invasive-strategy group and 30 in the noninvasive group), giving a 9-year overall survival of 74.4%. Survival at 9 years was 76.7% in the invasive strategy group and 69.2% in the noninvasive group.
After the data were adjusted to account for patient age, sex, history of supraventricular tachyarrhythmia, left ventricular ejection fraction, PR interval, QRS duration, and heart rate, the invasive strategy group had a hazard ratio of 0.61 for dying when compared with the noninvasive-strategy group (JAMA 2012;307:1292-301).
This difference was attributable mostly to the lower rate of sudden death in the invasive-strategy group (10 sudden deaths: 8 pacemaker recipients, 1 ICD recipient, and 1 patient without a pacing device), compared with those in the noninvasive strategy group (16 sudden deaths). "The 9-year cumulative incidence of sudden death was 4.5% in the invasive-strategy group and 18.0% in the noninvasive-strategy group," they added.
There were no significant differences between the two study groups in deaths due to respiratory failure or to other causes.
"While other studies are needed to confirm these findings, consideration of [the invasive] strategy may be prudent in this population, [who are] at higher than average risk for sudden death," Dr. Wahbi and his associates wrote.
This study was funded by the Association Française Contre les Myopathies. Dr. Wahbi reported no financial conflicts of interest, and two of his coauthors reported ties to Boston Scientific, Sorin Group France, Biotronik France, and Genzyme.
A strategy of implanting permanent cardiac pacing markedly improved survival for patients with myotonic dystrophy type 1 who presented with surface electrocardiogram manifestations of cardiac conduction abnormalities, compared with a noninvasive strategy of regular surveillance, according to a retrospective cohort study published March 28 in JAMA.
The likelihood of dying during a median 7.4 years of follow-up was nearly 40% lower among patients who underwent electrophysiological testing and, potentially, prophylactic pacing, than among those who did not undergo electrophysiological testing or pacing. The difference between the two groups was largely attributed to a decrease in sudden death in those who received a pacemaker or implantable cardioverter-defibrillator (ICD), said Dr. Karim Wahbi of the departments of cardiology and neurology at Cochin Hospital and the Myology Institute at Pitié-Salpêtrière Hospital, both in Paris, and his associates.
The study findings suggest that conduction system disease "is a major cause of sudden death that appears to be preventable" in this patient population, they noted.
Myotonic dystrophy type 1, also known as Steinert disease, is the most common inherited neuromuscular disease in adults and is characterized by muscle weakness, myotonia, multiple endocrine disorders, respiratory insufficiency, and cardiac abnormalities. As many as one-third of patients succumb to sudden death.
Both the American College of Cardiology and the American Heart Association recommend permanent pacing in patients with myotonic dystrophy type 1 when complete atrioventricular block or advanced high-degree atrioventricular block are detected, or prophylactically when first-degree atrioventricular or fascicular block are detected on ECG. However, whether this invasive approach actually prevents sudden death has never been assessed in a clinical study until now, Dr. Wahbi and his colleagues said.
Of 914 consecutive adults in the DM1 Heart Registry at the Myology Institute who were admitted during 2000-2009 for management of myotonic dystrophy type 1, 486 had ECGs showing minor conduction defects.
Of these subjects, 341 were managed invasively on the advice of their cardiologists. Electrophysiological testing in these patients found an HV interval greater than 70 ms in 164 patients and sustained ventricular tachyarrhythmias that were induced by programmed ventricular stimulation in 70 patients. The patients with an HV interval greater than 70 ms underwent placement of a pacemaker (150) or received an ICD (14). The remaining patients in the invasive-strategy group did not undergo a device implantation.
The remaining 145 subjects were managed noninvasively on the advice of their cardiologists. They had no electrophysiological study and no implantation of pacing devices, but were followed yearly by a neurologist and a cardiologist, both of whom specialized in muscular diseases.
Overall, 49 patients received antiarrhythmic drugs, including amiodarone and beta-blockers.
During a median follow-up of 7.4 years (range, 0-9.9 years), 80 patients died (50 in the invasive-strategy group and 30 in the noninvasive group), giving a 9-year overall survival of 74.4%. Survival at 9 years was 76.7% in the invasive strategy group and 69.2% in the noninvasive group.
After the data were adjusted to account for patient age, sex, history of supraventricular tachyarrhythmia, left ventricular ejection fraction, PR interval, QRS duration, and heart rate, the invasive strategy group had a hazard ratio of 0.61 for dying when compared with the noninvasive-strategy group (JAMA 2012;307:1292-301).
This difference was attributable mostly to the lower rate of sudden death in the invasive-strategy group (10 sudden deaths: 8 pacemaker recipients, 1 ICD recipient, and 1 patient without a pacing device), compared with those in the noninvasive strategy group (16 sudden deaths). "The 9-year cumulative incidence of sudden death was 4.5% in the invasive-strategy group and 18.0% in the noninvasive-strategy group," they added.
There were no significant differences between the two study groups in deaths due to respiratory failure or to other causes.
"While other studies are needed to confirm these findings, consideration of [the invasive] strategy may be prudent in this population, [who are] at higher than average risk for sudden death," Dr. Wahbi and his associates wrote.
This study was funded by the Association Française Contre les Myopathies. Dr. Wahbi reported no financial conflicts of interest, and two of his coauthors reported ties to Boston Scientific, Sorin Group France, Biotronik France, and Genzyme.
A strategy of implanting permanent cardiac pacing markedly improved survival for patients with myotonic dystrophy type 1 who presented with surface electrocardiogram manifestations of cardiac conduction abnormalities, compared with a noninvasive strategy of regular surveillance, according to a retrospective cohort study published March 28 in JAMA.
The likelihood of dying during a median 7.4 years of follow-up was nearly 40% lower among patients who underwent electrophysiological testing and, potentially, prophylactic pacing, than among those who did not undergo electrophysiological testing or pacing. The difference between the two groups was largely attributed to a decrease in sudden death in those who received a pacemaker or implantable cardioverter-defibrillator (ICD), said Dr. Karim Wahbi of the departments of cardiology and neurology at Cochin Hospital and the Myology Institute at Pitié-Salpêtrière Hospital, both in Paris, and his associates.
The study findings suggest that conduction system disease "is a major cause of sudden death that appears to be preventable" in this patient population, they noted.
Myotonic dystrophy type 1, also known as Steinert disease, is the most common inherited neuromuscular disease in adults and is characterized by muscle weakness, myotonia, multiple endocrine disorders, respiratory insufficiency, and cardiac abnormalities. As many as one-third of patients succumb to sudden death.
Both the American College of Cardiology and the American Heart Association recommend permanent pacing in patients with myotonic dystrophy type 1 when complete atrioventricular block or advanced high-degree atrioventricular block are detected, or prophylactically when first-degree atrioventricular or fascicular block are detected on ECG. However, whether this invasive approach actually prevents sudden death has never been assessed in a clinical study until now, Dr. Wahbi and his colleagues said.
Of 914 consecutive adults in the DM1 Heart Registry at the Myology Institute who were admitted during 2000-2009 for management of myotonic dystrophy type 1, 486 had ECGs showing minor conduction defects.
Of these subjects, 341 were managed invasively on the advice of their cardiologists. Electrophysiological testing in these patients found an HV interval greater than 70 ms in 164 patients and sustained ventricular tachyarrhythmias that were induced by programmed ventricular stimulation in 70 patients. The patients with an HV interval greater than 70 ms underwent placement of a pacemaker (150) or received an ICD (14). The remaining patients in the invasive-strategy group did not undergo a device implantation.
The remaining 145 subjects were managed noninvasively on the advice of their cardiologists. They had no electrophysiological study and no implantation of pacing devices, but were followed yearly by a neurologist and a cardiologist, both of whom specialized in muscular diseases.
Overall, 49 patients received antiarrhythmic drugs, including amiodarone and beta-blockers.
During a median follow-up of 7.4 years (range, 0-9.9 years), 80 patients died (50 in the invasive-strategy group and 30 in the noninvasive group), giving a 9-year overall survival of 74.4%. Survival at 9 years was 76.7% in the invasive strategy group and 69.2% in the noninvasive group.
After the data were adjusted to account for patient age, sex, history of supraventricular tachyarrhythmia, left ventricular ejection fraction, PR interval, QRS duration, and heart rate, the invasive strategy group had a hazard ratio of 0.61 for dying when compared with the noninvasive-strategy group (JAMA 2012;307:1292-301).
This difference was attributable mostly to the lower rate of sudden death in the invasive-strategy group (10 sudden deaths: 8 pacemaker recipients, 1 ICD recipient, and 1 patient without a pacing device), compared with those in the noninvasive strategy group (16 sudden deaths). "The 9-year cumulative incidence of sudden death was 4.5% in the invasive-strategy group and 18.0% in the noninvasive-strategy group," they added.
There were no significant differences between the two study groups in deaths due to respiratory failure or to other causes.
"While other studies are needed to confirm these findings, consideration of [the invasive] strategy may be prudent in this population, [who are] at higher than average risk for sudden death," Dr. Wahbi and his associates wrote.
This study was funded by the Association Française Contre les Myopathies. Dr. Wahbi reported no financial conflicts of interest, and two of his coauthors reported ties to Boston Scientific, Sorin Group France, Biotronik France, and Genzyme.
FROM JAMA
Major Finding: In patients with myotonic dystrophy type 1 and conduction abnormalities on ECG, the likelihood of dying was nearly 40% lower in those who had prophylactic cardiac pacing using a pacemaker or ICD than in those who were managed with regular surveillance by a cardiologist and a neurologist.
Data Source: This was a retrospective cohort study of 486 patients who were followed for a median of 7.4 years.
Disclosures: This study was funded by the Association Française Contre les Myopathies. Dr. Wahbi reported no financial conflicts of interest, and two of his coauthors reported ties to Boston Scientific, Sorin Group France, Biotronik France, and Genzyme.
Benchmarks Cited for In-Hospital VTE Post Arthroplasty
For the first time, researchers say they have established benchmarks for the rates of in-hospital venous thromboembolism that occur after total or partial hip arthroplasty and after total or partial knee arthroplasty.
In a meta-analysis of 47 studies published in JAMA that documented venous thromboembolism (VTE) event rates in nearly 45,000 patients who received recommended prophylaxis during hospitalization for knee or hip arthroplasty, investigators estimated that approximately 1 in every 100 patients undergoing knee arthroplasty and 1 in every 200 undergoing hip arthroplasty will develop symptomatic VTE before discharge.
"These estimates are of value to individual patients and clinicians in the consideration of risks and benefits" of the two procedures. They also are important because rates of in-hospital VTE are increasingly used as indicators of patient safety at individual medical centers, even though the expected background rates haven’t been established until now, said Jean-Marie Januel, a registered nurse with the Institute of Social and Preventive Medicine, Lausanne (Switzerland) University Hospital, and his associates.
Mr. Januel and his colleagues analyzed 41 randomized clinical trials and 6 observational studies performed between 1996 and 2011 in which subjects having hip or knee arthroplasty received VTE prophylaxis according to published guidelines, including either low-molecular-weight heparin or inhibitors of factor Xa or IIa.
A total of 22 of the studies were performed in Europe, 14 were in North America, and 11 were in other regions. The mean duration of follow-up after either surgery was 13 days. This included 21 studies of partial or total hip arthroplasty, 20 of partial or total knee arthroplasty, and 6 studies of both procedures, with a total of 44,844 subjects.
There were 443 cases of symptomatic postoperative VTE that developed before hospital discharge: 288 in the 23,475 knee patients and 155 in the 23,475 hip patients. The pooled incidence rates of VTE were approximately 1% after knee arthroplasty and approximately 0.5% after hip arthroplasty. (JAMA 2012;307:294-303). When the data were broken down by type of VTE, the pooled incidence rates were 0.26% for deep vein thrombosis and 0.14% for pulmonary embolism after knee arthroplasty. The corresponding rates were 0.63% for deep vein thrombosis and 0.27% for pulmonary embolism after hip arthroplasty.
The pooled incidence rates of deep vein thrombosis were lower for both knee patients and hip patients when factor Xa or IIa inhibitors, rather than low-molecular-weight heparin, were given for prophylaxis. "However, we cannot make assertions regarding comparative efficacy among treatments, because our meta-analysis did not directly compare [these agents]," they said.
They reported no financial conflicts.
This is an important meta-analysis in that it provides what may be the lowest expected incidence of in-hospital development of VTE complications following orthopedic surgery. However, as the authors point out, real world incidence may be higher since it is generally accepted that adequate prophylaxis is not being implemented in many institutions. More importantly, this report may instill a false sense of security about the actual postoperative incidence of VTE after total knee or hip reconstructions since many VTE occur after discharge. It is suggested that as many as 70% may actually occur at home. Accordingly, the recent guidelines on antithrombotic therapy as published in Chest (2012;141:7S-47S) recommend extending VTE prophylaxis well into the outpatient setting.
Dr. Russell H. Samson is a clinical associate professor of surgery (vascular) at Florida State University, and an attending surgeon at Sarasota Vascular Specialists, and associate medical editor of Vascular Specialist.
This is an important meta-analysis in that it provides what may be the lowest expected incidence of in-hospital development of VTE complications following orthopedic surgery. However, as the authors point out, real world incidence may be higher since it is generally accepted that adequate prophylaxis is not being implemented in many institutions. More importantly, this report may instill a false sense of security about the actual postoperative incidence of VTE after total knee or hip reconstructions since many VTE occur after discharge. It is suggested that as many as 70% may actually occur at home. Accordingly, the recent guidelines on antithrombotic therapy as published in Chest (2012;141:7S-47S) recommend extending VTE prophylaxis well into the outpatient setting.
Dr. Russell H. Samson is a clinical associate professor of surgery (vascular) at Florida State University, and an attending surgeon at Sarasota Vascular Specialists, and associate medical editor of Vascular Specialist.
This is an important meta-analysis in that it provides what may be the lowest expected incidence of in-hospital development of VTE complications following orthopedic surgery. However, as the authors point out, real world incidence may be higher since it is generally accepted that adequate prophylaxis is not being implemented in many institutions. More importantly, this report may instill a false sense of security about the actual postoperative incidence of VTE after total knee or hip reconstructions since many VTE occur after discharge. It is suggested that as many as 70% may actually occur at home. Accordingly, the recent guidelines on antithrombotic therapy as published in Chest (2012;141:7S-47S) recommend extending VTE prophylaxis well into the outpatient setting.
Dr. Russell H. Samson is a clinical associate professor of surgery (vascular) at Florida State University, and an attending surgeon at Sarasota Vascular Specialists, and associate medical editor of Vascular Specialist.
For the first time, researchers say they have established benchmarks for the rates of in-hospital venous thromboembolism that occur after total or partial hip arthroplasty and after total or partial knee arthroplasty.
In a meta-analysis of 47 studies published in JAMA that documented venous thromboembolism (VTE) event rates in nearly 45,000 patients who received recommended prophylaxis during hospitalization for knee or hip arthroplasty, investigators estimated that approximately 1 in every 100 patients undergoing knee arthroplasty and 1 in every 200 undergoing hip arthroplasty will develop symptomatic VTE before discharge.
"These estimates are of value to individual patients and clinicians in the consideration of risks and benefits" of the two procedures. They also are important because rates of in-hospital VTE are increasingly used as indicators of patient safety at individual medical centers, even though the expected background rates haven’t been established until now, said Jean-Marie Januel, a registered nurse with the Institute of Social and Preventive Medicine, Lausanne (Switzerland) University Hospital, and his associates.
Mr. Januel and his colleagues analyzed 41 randomized clinical trials and 6 observational studies performed between 1996 and 2011 in which subjects having hip or knee arthroplasty received VTE prophylaxis according to published guidelines, including either low-molecular-weight heparin or inhibitors of factor Xa or IIa.
A total of 22 of the studies were performed in Europe, 14 were in North America, and 11 were in other regions. The mean duration of follow-up after either surgery was 13 days. This included 21 studies of partial or total hip arthroplasty, 20 of partial or total knee arthroplasty, and 6 studies of both procedures, with a total of 44,844 subjects.
There were 443 cases of symptomatic postoperative VTE that developed before hospital discharge: 288 in the 23,475 knee patients and 155 in the 23,475 hip patients. The pooled incidence rates of VTE were approximately 1% after knee arthroplasty and approximately 0.5% after hip arthroplasty. (JAMA 2012;307:294-303). When the data were broken down by type of VTE, the pooled incidence rates were 0.26% for deep vein thrombosis and 0.14% for pulmonary embolism after knee arthroplasty. The corresponding rates were 0.63% for deep vein thrombosis and 0.27% for pulmonary embolism after hip arthroplasty.
The pooled incidence rates of deep vein thrombosis were lower for both knee patients and hip patients when factor Xa or IIa inhibitors, rather than low-molecular-weight heparin, were given for prophylaxis. "However, we cannot make assertions regarding comparative efficacy among treatments, because our meta-analysis did not directly compare [these agents]," they said.
They reported no financial conflicts.
For the first time, researchers say they have established benchmarks for the rates of in-hospital venous thromboembolism that occur after total or partial hip arthroplasty and after total or partial knee arthroplasty.
In a meta-analysis of 47 studies published in JAMA that documented venous thromboembolism (VTE) event rates in nearly 45,000 patients who received recommended prophylaxis during hospitalization for knee or hip arthroplasty, investigators estimated that approximately 1 in every 100 patients undergoing knee arthroplasty and 1 in every 200 undergoing hip arthroplasty will develop symptomatic VTE before discharge.
"These estimates are of value to individual patients and clinicians in the consideration of risks and benefits" of the two procedures. They also are important because rates of in-hospital VTE are increasingly used as indicators of patient safety at individual medical centers, even though the expected background rates haven’t been established until now, said Jean-Marie Januel, a registered nurse with the Institute of Social and Preventive Medicine, Lausanne (Switzerland) University Hospital, and his associates.
Mr. Januel and his colleagues analyzed 41 randomized clinical trials and 6 observational studies performed between 1996 and 2011 in which subjects having hip or knee arthroplasty received VTE prophylaxis according to published guidelines, including either low-molecular-weight heparin or inhibitors of factor Xa or IIa.
A total of 22 of the studies were performed in Europe, 14 were in North America, and 11 were in other regions. The mean duration of follow-up after either surgery was 13 days. This included 21 studies of partial or total hip arthroplasty, 20 of partial or total knee arthroplasty, and 6 studies of both procedures, with a total of 44,844 subjects.
There were 443 cases of symptomatic postoperative VTE that developed before hospital discharge: 288 in the 23,475 knee patients and 155 in the 23,475 hip patients. The pooled incidence rates of VTE were approximately 1% after knee arthroplasty and approximately 0.5% after hip arthroplasty. (JAMA 2012;307:294-303). When the data were broken down by type of VTE, the pooled incidence rates were 0.26% for deep vein thrombosis and 0.14% for pulmonary embolism after knee arthroplasty. The corresponding rates were 0.63% for deep vein thrombosis and 0.27% for pulmonary embolism after hip arthroplasty.
The pooled incidence rates of deep vein thrombosis were lower for both knee patients and hip patients when factor Xa or IIa inhibitors, rather than low-molecular-weight heparin, were given for prophylaxis. "However, we cannot make assertions regarding comparative efficacy among treatments, because our meta-analysis did not directly compare [these agents]," they said.
They reported no financial conflicts.
Major Finding: 443 cases of VTE developed during hospitalization, for an incidence of approximately 1 in every 100 patients undergoing knee arthroplasty and 1 in every 200 patients undergoing hip arthroplasty.
Data Source: A meta-analysis of 47 randomized clinical trials and observational studies from 1996 to 2011 that documented VTE rates in 44,844 patients undergoing total or partial hip or knee arthroplasty.
Disclosures: This study was supported by Alberta Innovates Health Solutions, a government research funding agency, and the International Methodology Consortium for Coded Health Information, a collaboration of health sciences researchers to promote quality of care. No financial conflicts of interest were reported.
Rivaroxaban Found Noninferior to Standard Therapy for Pulmonary Embolism
Rivaroxaban was noninferior to standard treatment at preventing a recurrence of pulmonary embolism in an international open-label trial reported online in the New England Journal of Medicine and presented simultaneously at the annual meeting of the American College of Cardiology.
Rates of adverse bleeding events with rivaroxaban were similar to those with the more complex standard therapy of enoxaparin plus a vitamin K antagonist, a regimen that requires INR (international normalized ratio) monitoring, said Dr. Harry R. Buller of the department of vascular medicine at the University of Amsterdam and his associated in the EINSTEIN-PE clinical trial.
The study, sponsored by Bayer HealthCare and Janssen Pharmaceuticals, involved 4,832 adults with acute symptomatic pulmonary embolism (PE), with or without deep vein thrombosis (DVT), who were treated at 263 sites in 38 countries in 2007-2011. In all, 2,419 study subjects were randomly assigned to receive oral rivaroxaban (15 mg twice daily for 3 weeks, followed by 20 mg once daily) and 2,413 to receive enoxaparin (1 mg/kg twice daily, which was discontinued when the INR reached 2.0 or greater for 2 consecutive days after at least 5 days of therapy) plus either warfarin or acenocoumarol at a dosage adjusted to maintain an INR of 2.0-3.0.
All study subjects were treated for durations of 3, 6, or 12 months, according to the wishes of their treating physicians and in keeping with current practice. The mean duration of treatment was approximately 9 months.
In the standard treatment group, the INR was in the therapeutic range 62.7% of the time. The INR was not measured in the rivaroxaban group, but adherence to therapy was high in 94.2% of patients.
The primary efficacy outcome was symptomatic recurrent venous thromboembolism, a composite of fatal and nonfatal PE or DVT. This occurred in 50 patients (2.1%) receiving rivaroxaban and 44 (1.8%) receiving standard therapy, which met the criterion for noninferiority, the investigators said (N. Engl. J. Med. 2012 March 26 [doi:10.1056/NEJMoa1113572]).
During the initial 3-week period of intensive rivaroxaban therapy, the primary efficacy outcome occurred in 18 patients (0.7%) of the rivaroxaban group and in 21 patients (0.9%) in the standard therapy group.
The efficacy results were similar in a per-protocol analysis and in an intention-to-treat analysis.
The primary safety outcome was clinically relevant bleeding, and it occurred in 249 subjects (10.3%) in the rivaroxaban group and in 274 (11.4%) in the standard treatment group. In particular, major bleeding events occurred in 26 patients (1.1%) taking rivaroxaban and 52 (2.2%) taking standard treatment.
During the initial 3-week period of intensive rivaroxaban therapy, bleeding rates were similar between the two study groups. Over the full course of treatment, "there were fewer episodes of intracranial bleeding or bleeding in critical areas in the rivaroxaban group than in the standard therapy group," Dr. Buller and his colleagues said.
With regard to other safety outcomes, the rates of acute coronary events were similar between the two study groups, at 0.6% with rivaroxaban and 0.9% with standard therapy. And abnormal findings on liver function tests were seen in 0.2% of both groups.
"The suggestion that rivaroxaban can be administered at the same dose in all patients without laboratory monitoring has raised concern," so the researchers performed multiple subgroup analyses to examine whether any particular patient group was at increased risk while taking the drug. They found that the rates of recurrent venous thromboembolism and of adverse bleeding events were similar between patients taking rivaroxaban and those on standard therapy regardless of age, sex, obesity status, level of renal function, and the extent of initial PE.
"We believe that our population is representative of the spectrum of patients who present with symptomatic PE," the investigators said, noting that nearly 25% of the study population had extensive PE and 13% had limited PE. Moreover, almost 25% also had concomitant DVT.
This study was supported by Bayer HealthCare and Janssen Pharmaceuticals. Dr. Buller reported ties to ICTOM/Bayer HealthCare, Sanofi-Aventis, Boehringer Ingelheim, Pfizer, GlaxoSmithKline, and Daiichi Sankyo, and his associates reported ties to numerous industry sources.
As soon as the FDA gives its approval, "rivaroxaban is going to be used by hospitalists and other physicians to manage patients with pulmonary embolism and deep vein thrombosis," said Dr. Amir K. Jaffer, the division chief of hospital medicine at the University of Miami. He is also a member of the Hospitalist News advisory board.
Rivaroxaban appears to be just as safe as low-molecular-weight heparin followed by coumadin, which is considered conventional therapy for pulmonary embolism (PE), which involves heparin for 5 days followed by coumadin that requires monitoring.
Compared to conventional therapy, it appears that this drug may be safer and just as effective. Now, instead of injections, you have an oral drug. In this study, the researchers used 15 mg rivaroxaban given orally twice daily for 3 weeks, followed by 20 mg once daily. I think this may allow us to send patients home early from the hospital.
"With standard therapy, patients have to be taught how to perform self injections. You have to make sure that they're doing it correctly before you send them home. …Potentially, this new drug will cut the length of hospital stay," given that patients don't need to be taught about self-injections.
"Of course, it will still be necessary to teach patients signs to look out for that could mean PE or DVT [deep vein thrombosis]," he said. Patients still will need continued care from their primary care physician or specialist.
Rivaroxaban may be safer as well. There was a 1.1% risk reduction with the use of rivaroxaban compared with standard therapy in the current study.
Of note, about a quarter of the patients in this study also had DVT. In similar trial, that came out in 2010, researchers looked at the efficacy and safety of rivaroxaban for DVT (N. Engl. J. Med. 2010;363:2499-510). Rivaroxaban was found to be safe and efficacious for DVT as well, said Dr. Jaffer.
"I haven't seen any cost-effectiveness analysis just yet. The drug may be more expensive to purchase but it may be that cost-effective analyses suggest that it will be more cost effective overall to use this newer drug."
Dr. Jaffer reported that he is a consultant for Janssen Pharmaceuticals.
As soon as the FDA gives its approval, "rivaroxaban is going to be used by hospitalists and other physicians to manage patients with pulmonary embolism and deep vein thrombosis," said Dr. Amir K. Jaffer, the division chief of hospital medicine at the University of Miami. He is also a member of the Hospitalist News advisory board.
Rivaroxaban appears to be just as safe as low-molecular-weight heparin followed by coumadin, which is considered conventional therapy for pulmonary embolism (PE), which involves heparin for 5 days followed by coumadin that requires monitoring.
Compared to conventional therapy, it appears that this drug may be safer and just as effective. Now, instead of injections, you have an oral drug. In this study, the researchers used 15 mg rivaroxaban given orally twice daily for 3 weeks, followed by 20 mg once daily. I think this may allow us to send patients home early from the hospital.
"With standard therapy, patients have to be taught how to perform self injections. You have to make sure that they're doing it correctly before you send them home. …Potentially, this new drug will cut the length of hospital stay," given that patients don't need to be taught about self-injections.
"Of course, it will still be necessary to teach patients signs to look out for that could mean PE or DVT [deep vein thrombosis]," he said. Patients still will need continued care from their primary care physician or specialist.
Rivaroxaban may be safer as well. There was a 1.1% risk reduction with the use of rivaroxaban compared with standard therapy in the current study.
Of note, about a quarter of the patients in this study also had DVT. In similar trial, that came out in 2010, researchers looked at the efficacy and safety of rivaroxaban for DVT (N. Engl. J. Med. 2010;363:2499-510). Rivaroxaban was found to be safe and efficacious for DVT as well, said Dr. Jaffer.
"I haven't seen any cost-effectiveness analysis just yet. The drug may be more expensive to purchase but it may be that cost-effective analyses suggest that it will be more cost effective overall to use this newer drug."
Dr. Jaffer reported that he is a consultant for Janssen Pharmaceuticals.
As soon as the FDA gives its approval, "rivaroxaban is going to be used by hospitalists and other physicians to manage patients with pulmonary embolism and deep vein thrombosis," said Dr. Amir K. Jaffer, the division chief of hospital medicine at the University of Miami. He is also a member of the Hospitalist News advisory board.
Rivaroxaban appears to be just as safe as low-molecular-weight heparin followed by coumadin, which is considered conventional therapy for pulmonary embolism (PE), which involves heparin for 5 days followed by coumadin that requires monitoring.
Compared to conventional therapy, it appears that this drug may be safer and just as effective. Now, instead of injections, you have an oral drug. In this study, the researchers used 15 mg rivaroxaban given orally twice daily for 3 weeks, followed by 20 mg once daily. I think this may allow us to send patients home early from the hospital.
"With standard therapy, patients have to be taught how to perform self injections. You have to make sure that they're doing it correctly before you send them home. …Potentially, this new drug will cut the length of hospital stay," given that patients don't need to be taught about self-injections.
"Of course, it will still be necessary to teach patients signs to look out for that could mean PE or DVT [deep vein thrombosis]," he said. Patients still will need continued care from their primary care physician or specialist.
Rivaroxaban may be safer as well. There was a 1.1% risk reduction with the use of rivaroxaban compared with standard therapy in the current study.
Of note, about a quarter of the patients in this study also had DVT. In similar trial, that came out in 2010, researchers looked at the efficacy and safety of rivaroxaban for DVT (N. Engl. J. Med. 2010;363:2499-510). Rivaroxaban was found to be safe and efficacious for DVT as well, said Dr. Jaffer.
"I haven't seen any cost-effectiveness analysis just yet. The drug may be more expensive to purchase but it may be that cost-effective analyses suggest that it will be more cost effective overall to use this newer drug."
Dr. Jaffer reported that he is a consultant for Janssen Pharmaceuticals.
Rivaroxaban was noninferior to standard treatment at preventing a recurrence of pulmonary embolism in an international open-label trial reported online in the New England Journal of Medicine and presented simultaneously at the annual meeting of the American College of Cardiology.
Rates of adverse bleeding events with rivaroxaban were similar to those with the more complex standard therapy of enoxaparin plus a vitamin K antagonist, a regimen that requires INR (international normalized ratio) monitoring, said Dr. Harry R. Buller of the department of vascular medicine at the University of Amsterdam and his associated in the EINSTEIN-PE clinical trial.
The study, sponsored by Bayer HealthCare and Janssen Pharmaceuticals, involved 4,832 adults with acute symptomatic pulmonary embolism (PE), with or without deep vein thrombosis (DVT), who were treated at 263 sites in 38 countries in 2007-2011. In all, 2,419 study subjects were randomly assigned to receive oral rivaroxaban (15 mg twice daily for 3 weeks, followed by 20 mg once daily) and 2,413 to receive enoxaparin (1 mg/kg twice daily, which was discontinued when the INR reached 2.0 or greater for 2 consecutive days after at least 5 days of therapy) plus either warfarin or acenocoumarol at a dosage adjusted to maintain an INR of 2.0-3.0.
All study subjects were treated for durations of 3, 6, or 12 months, according to the wishes of their treating physicians and in keeping with current practice. The mean duration of treatment was approximately 9 months.
In the standard treatment group, the INR was in the therapeutic range 62.7% of the time. The INR was not measured in the rivaroxaban group, but adherence to therapy was high in 94.2% of patients.
The primary efficacy outcome was symptomatic recurrent venous thromboembolism, a composite of fatal and nonfatal PE or DVT. This occurred in 50 patients (2.1%) receiving rivaroxaban and 44 (1.8%) receiving standard therapy, which met the criterion for noninferiority, the investigators said (N. Engl. J. Med. 2012 March 26 [doi:10.1056/NEJMoa1113572]).
During the initial 3-week period of intensive rivaroxaban therapy, the primary efficacy outcome occurred in 18 patients (0.7%) of the rivaroxaban group and in 21 patients (0.9%) in the standard therapy group.
The efficacy results were similar in a per-protocol analysis and in an intention-to-treat analysis.
The primary safety outcome was clinically relevant bleeding, and it occurred in 249 subjects (10.3%) in the rivaroxaban group and in 274 (11.4%) in the standard treatment group. In particular, major bleeding events occurred in 26 patients (1.1%) taking rivaroxaban and 52 (2.2%) taking standard treatment.
During the initial 3-week period of intensive rivaroxaban therapy, bleeding rates were similar between the two study groups. Over the full course of treatment, "there were fewer episodes of intracranial bleeding or bleeding in critical areas in the rivaroxaban group than in the standard therapy group," Dr. Buller and his colleagues said.
With regard to other safety outcomes, the rates of acute coronary events were similar between the two study groups, at 0.6% with rivaroxaban and 0.9% with standard therapy. And abnormal findings on liver function tests were seen in 0.2% of both groups.
"The suggestion that rivaroxaban can be administered at the same dose in all patients without laboratory monitoring has raised concern," so the researchers performed multiple subgroup analyses to examine whether any particular patient group was at increased risk while taking the drug. They found that the rates of recurrent venous thromboembolism and of adverse bleeding events were similar between patients taking rivaroxaban and those on standard therapy regardless of age, sex, obesity status, level of renal function, and the extent of initial PE.
"We believe that our population is representative of the spectrum of patients who present with symptomatic PE," the investigators said, noting that nearly 25% of the study population had extensive PE and 13% had limited PE. Moreover, almost 25% also had concomitant DVT.
This study was supported by Bayer HealthCare and Janssen Pharmaceuticals. Dr. Buller reported ties to ICTOM/Bayer HealthCare, Sanofi-Aventis, Boehringer Ingelheim, Pfizer, GlaxoSmithKline, and Daiichi Sankyo, and his associates reported ties to numerous industry sources.
Rivaroxaban was noninferior to standard treatment at preventing a recurrence of pulmonary embolism in an international open-label trial reported online in the New England Journal of Medicine and presented simultaneously at the annual meeting of the American College of Cardiology.
Rates of adverse bleeding events with rivaroxaban were similar to those with the more complex standard therapy of enoxaparin plus a vitamin K antagonist, a regimen that requires INR (international normalized ratio) monitoring, said Dr. Harry R. Buller of the department of vascular medicine at the University of Amsterdam and his associated in the EINSTEIN-PE clinical trial.
The study, sponsored by Bayer HealthCare and Janssen Pharmaceuticals, involved 4,832 adults with acute symptomatic pulmonary embolism (PE), with or without deep vein thrombosis (DVT), who were treated at 263 sites in 38 countries in 2007-2011. In all, 2,419 study subjects were randomly assigned to receive oral rivaroxaban (15 mg twice daily for 3 weeks, followed by 20 mg once daily) and 2,413 to receive enoxaparin (1 mg/kg twice daily, which was discontinued when the INR reached 2.0 or greater for 2 consecutive days after at least 5 days of therapy) plus either warfarin or acenocoumarol at a dosage adjusted to maintain an INR of 2.0-3.0.
All study subjects were treated for durations of 3, 6, or 12 months, according to the wishes of their treating physicians and in keeping with current practice. The mean duration of treatment was approximately 9 months.
In the standard treatment group, the INR was in the therapeutic range 62.7% of the time. The INR was not measured in the rivaroxaban group, but adherence to therapy was high in 94.2% of patients.
The primary efficacy outcome was symptomatic recurrent venous thromboembolism, a composite of fatal and nonfatal PE or DVT. This occurred in 50 patients (2.1%) receiving rivaroxaban and 44 (1.8%) receiving standard therapy, which met the criterion for noninferiority, the investigators said (N. Engl. J. Med. 2012 March 26 [doi:10.1056/NEJMoa1113572]).
During the initial 3-week period of intensive rivaroxaban therapy, the primary efficacy outcome occurred in 18 patients (0.7%) of the rivaroxaban group and in 21 patients (0.9%) in the standard therapy group.
The efficacy results were similar in a per-protocol analysis and in an intention-to-treat analysis.
The primary safety outcome was clinically relevant bleeding, and it occurred in 249 subjects (10.3%) in the rivaroxaban group and in 274 (11.4%) in the standard treatment group. In particular, major bleeding events occurred in 26 patients (1.1%) taking rivaroxaban and 52 (2.2%) taking standard treatment.
During the initial 3-week period of intensive rivaroxaban therapy, bleeding rates were similar between the two study groups. Over the full course of treatment, "there were fewer episodes of intracranial bleeding or bleeding in critical areas in the rivaroxaban group than in the standard therapy group," Dr. Buller and his colleagues said.
With regard to other safety outcomes, the rates of acute coronary events were similar between the two study groups, at 0.6% with rivaroxaban and 0.9% with standard therapy. And abnormal findings on liver function tests were seen in 0.2% of both groups.
"The suggestion that rivaroxaban can be administered at the same dose in all patients without laboratory monitoring has raised concern," so the researchers performed multiple subgroup analyses to examine whether any particular patient group was at increased risk while taking the drug. They found that the rates of recurrent venous thromboembolism and of adverse bleeding events were similar between patients taking rivaroxaban and those on standard therapy regardless of age, sex, obesity status, level of renal function, and the extent of initial PE.
"We believe that our population is representative of the spectrum of patients who present with symptomatic PE," the investigators said, noting that nearly 25% of the study population had extensive PE and 13% had limited PE. Moreover, almost 25% also had concomitant DVT.
This study was supported by Bayer HealthCare and Janssen Pharmaceuticals. Dr. Buller reported ties to ICTOM/Bayer HealthCare, Sanofi-Aventis, Boehringer Ingelheim, Pfizer, GlaxoSmithKline, and Daiichi Sankyo, and his associates reported ties to numerous industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: PE, with or without DVT, recurred in 2.1% of patients receiving rivaroxaban and in 1.8% of those taking standard preventive therapy, meeting the prespecified criterion for noninferiority.
Data Source: This was a 4-year, randomized, open-label, noninferiority trial comparing rivaroxaban (2,419 subjects) and standard treatment (2,413 subjects) in 38 countries.
Disclosures: This study was supported by Bayer HealthCare and Janssen Pharmaceuticals. Dr. Buller reported ties to ICTOM/Bayer HealthCare, Sanofi-Aventis, Boehringer Ingelheim, Pfizer, GlaxoSmithKline, and Daiichi Sankyo, and his associates reported ties to numerous industry sources.
Anesthesia Services for GI Procedures Have Doubled
The proportion of outpatient endoscopies and colonoscopies in which the services of an anesthesiologist or nurse anesthetist were used doubled in a recent 6-year period, according to a report in the March 21 issue of JAMA.
Anesthesia services include deep sedation or general anesthesia and must be administered by an anesthesiologist or nurse anesthetist, in contrast to lighter sedation that is typically provided by nurses under the endoscopist’s supervision. Current payment guidelines hold that low-risk patients can be adequately managed with light sedation and that anesthesia services are only justified in high-risk patients, according to Hangsheng Liu, Ph.D., and his associates at the RAND Corporation.
In this study of a large, nationally representative sample of patients covered by both Medicare and commercial medical insurance, the rate at which anesthesia services were used during GI procedures rose from approximately 14% in 2003 to more than 30% in 2009, they noted.
The researchers performed a retrospective analysis of insurance claims data to track the use of anesthesia services for endoscopies and colonoscopies over time because this has been identified as an area in which "excess" health care costs can be trimmed. They analyzed a Medicare fee-for-service sample of 6.6 million patients and a sample of 5.5 million patients covered by commercial insurance companies.
A medical need for anesthesia services during GI procedures in these cohorts could not be determined directly from the medical data, so it was "approximated" by classifying patients according to American Society of Anesthesiologists guidelines, which factor in age, sex, comorbidities, and medical history.
Patients who met criteria for ASA status level 1 or 2 were considered low-risk; the use of deep sedation or general anesthesia in such patients was not required and was judged to be "discretionary." In contrast, patients with an ASA status level of 3 or higher were considered high-risk and were judged to be appropriate candidates for deep sedation or general anesthesia.
Overall, there were 2.2 million GI procedures performed in Medicare patients and 7.0 million in commercially insured patients, for a total of 9.2 million during the study period.
The proportion of procedures using anesthesia services more than doubled during the 6-year study period in both groups: from 13.5% to 30.2% for Medicare patients and from 13.6% to 35.5% for commercially insured patients.
Among low-risk Medicare patients, the proportion of GI procedures using anesthesia services rose from 18,989 per million enrollees in 2003 to 25,069 per million enrollees in 2009. Annual payments per million enrollees rose by 8% per year.
Among low-risk commercially insured patients, the proportion of GI procedures using anesthesia services rose from 3,938 to 15,108 per million enrollees, and annual payments rose more than fourfold, from $1.69 million to $7.05 million per million enrollees.
Applying these figures to national statistics, the annual spending for anesthesia services on low-risk patients who probably don’t need them was estimated to be $1.1 billion in 2009 – $129 million for Medicare patients and $945 million for commercially insured patients.
Thus, "the majority of gastroenterology-related anesthesia services are provided to low-risk patients and can be considered potentially discretionary based on current payment policies," Dr. Liu and his colleagues concluded (JAMA 2012;307:1178-84).
The use of anesthesia services varied substantially by geographic region, another indication that clinical necessity is not the chief factor driving the use of heavy sedation or general anesthesia during these procedures. In the most recent year for which data were available, the use of anesthesia services during endoscopy or colonoscopy was lowest in the western United States (14% of Medicare patients and 13% of commercially insured patients) and highest in the northeast (48% and 59%, respectively). This is a nearly fourfold difference.
The rise in the use of anesthesia services "has been partly attributed to the adoption of propofol, which by virtue of a short half-life and rapid onset of action is thought to be more convenient and to offer a more consistent level of sedation than regimens previously used, but in the United States ‘should be administered only by persons trained in the administration of general anesthesia.’ Insurers’ payment policies and marketing by the anesthesiology community have also been cited as drivers of increased use," the investigators noted.
They added that since the number of endoscopies and colonoscopies is increasing, the "discretionary" use of anesthesia in low-risk patients "represents a sizable target for cost savings."
This study was supported by Ethicon Endo-Surgery Inc. No financial conflicts of interest were reported.
Endoscopists may prefer to use anesthesia services in their low-risk patients for several reasons, including the fact that deep sedation or general anesthesia allows for more rapid completion of the exam and the belief that it may improve disease detection, said Dr. Lee A. Fleisher.
Many patients may be unwilling to undergo the procedure without it. And transferring the responsibility for managing sedation from the endoscopist to the anesthesiologist or nurse anesthetist may reduce the endoscopist’s medicolegal liability.
In some cases, the choice to use anesthesia services may be about efficiency, such as in patients who fail moderate sedation and are forced "to either wait for the availability of an anesthesiologist or reschedule the procedure for another day."
Moreover, the use of propofol in particular may be preferred because its faster onset/offset profile allows the endoscopist to perform more procedures per day.
Dr. Fleisher is in the department of anesthesiology and critical care and at the Leonard Davis Institute of Health Economics at the University of Pennsylvania, Philadelphia. He reported no financial conflicts of interest. These remarks were taken from his editorial accompanying Dr. Liu’s report (JAMA 2012;307:1200-01).
Endoscopists may prefer to use anesthesia services in their low-risk patients for several reasons, including the fact that deep sedation or general anesthesia allows for more rapid completion of the exam and the belief that it may improve disease detection, said Dr. Lee A. Fleisher.
Many patients may be unwilling to undergo the procedure without it. And transferring the responsibility for managing sedation from the endoscopist to the anesthesiologist or nurse anesthetist may reduce the endoscopist’s medicolegal liability.
In some cases, the choice to use anesthesia services may be about efficiency, such as in patients who fail moderate sedation and are forced "to either wait for the availability of an anesthesiologist or reschedule the procedure for another day."
Moreover, the use of propofol in particular may be preferred because its faster onset/offset profile allows the endoscopist to perform more procedures per day.
Dr. Fleisher is in the department of anesthesiology and critical care and at the Leonard Davis Institute of Health Economics at the University of Pennsylvania, Philadelphia. He reported no financial conflicts of interest. These remarks were taken from his editorial accompanying Dr. Liu’s report (JAMA 2012;307:1200-01).
Endoscopists may prefer to use anesthesia services in their low-risk patients for several reasons, including the fact that deep sedation or general anesthesia allows for more rapid completion of the exam and the belief that it may improve disease detection, said Dr. Lee A. Fleisher.
Many patients may be unwilling to undergo the procedure without it. And transferring the responsibility for managing sedation from the endoscopist to the anesthesiologist or nurse anesthetist may reduce the endoscopist’s medicolegal liability.
In some cases, the choice to use anesthesia services may be about efficiency, such as in patients who fail moderate sedation and are forced "to either wait for the availability of an anesthesiologist or reschedule the procedure for another day."
Moreover, the use of propofol in particular may be preferred because its faster onset/offset profile allows the endoscopist to perform more procedures per day.
Dr. Fleisher is in the department of anesthesiology and critical care and at the Leonard Davis Institute of Health Economics at the University of Pennsylvania, Philadelphia. He reported no financial conflicts of interest. These remarks were taken from his editorial accompanying Dr. Liu’s report (JAMA 2012;307:1200-01).
The proportion of outpatient endoscopies and colonoscopies in which the services of an anesthesiologist or nurse anesthetist were used doubled in a recent 6-year period, according to a report in the March 21 issue of JAMA.
Anesthesia services include deep sedation or general anesthesia and must be administered by an anesthesiologist or nurse anesthetist, in contrast to lighter sedation that is typically provided by nurses under the endoscopist’s supervision. Current payment guidelines hold that low-risk patients can be adequately managed with light sedation and that anesthesia services are only justified in high-risk patients, according to Hangsheng Liu, Ph.D., and his associates at the RAND Corporation.
In this study of a large, nationally representative sample of patients covered by both Medicare and commercial medical insurance, the rate at which anesthesia services were used during GI procedures rose from approximately 14% in 2003 to more than 30% in 2009, they noted.
The researchers performed a retrospective analysis of insurance claims data to track the use of anesthesia services for endoscopies and colonoscopies over time because this has been identified as an area in which "excess" health care costs can be trimmed. They analyzed a Medicare fee-for-service sample of 6.6 million patients and a sample of 5.5 million patients covered by commercial insurance companies.
A medical need for anesthesia services during GI procedures in these cohorts could not be determined directly from the medical data, so it was "approximated" by classifying patients according to American Society of Anesthesiologists guidelines, which factor in age, sex, comorbidities, and medical history.
Patients who met criteria for ASA status level 1 or 2 were considered low-risk; the use of deep sedation or general anesthesia in such patients was not required and was judged to be "discretionary." In contrast, patients with an ASA status level of 3 or higher were considered high-risk and were judged to be appropriate candidates for deep sedation or general anesthesia.
Overall, there were 2.2 million GI procedures performed in Medicare patients and 7.0 million in commercially insured patients, for a total of 9.2 million during the study period.
The proportion of procedures using anesthesia services more than doubled during the 6-year study period in both groups: from 13.5% to 30.2% for Medicare patients and from 13.6% to 35.5% for commercially insured patients.
Among low-risk Medicare patients, the proportion of GI procedures using anesthesia services rose from 18,989 per million enrollees in 2003 to 25,069 per million enrollees in 2009. Annual payments per million enrollees rose by 8% per year.
Among low-risk commercially insured patients, the proportion of GI procedures using anesthesia services rose from 3,938 to 15,108 per million enrollees, and annual payments rose more than fourfold, from $1.69 million to $7.05 million per million enrollees.
Applying these figures to national statistics, the annual spending for anesthesia services on low-risk patients who probably don’t need them was estimated to be $1.1 billion in 2009 – $129 million for Medicare patients and $945 million for commercially insured patients.
Thus, "the majority of gastroenterology-related anesthesia services are provided to low-risk patients and can be considered potentially discretionary based on current payment policies," Dr. Liu and his colleagues concluded (JAMA 2012;307:1178-84).
The use of anesthesia services varied substantially by geographic region, another indication that clinical necessity is not the chief factor driving the use of heavy sedation or general anesthesia during these procedures. In the most recent year for which data were available, the use of anesthesia services during endoscopy or colonoscopy was lowest in the western United States (14% of Medicare patients and 13% of commercially insured patients) and highest in the northeast (48% and 59%, respectively). This is a nearly fourfold difference.
The rise in the use of anesthesia services "has been partly attributed to the adoption of propofol, which by virtue of a short half-life and rapid onset of action is thought to be more convenient and to offer a more consistent level of sedation than regimens previously used, but in the United States ‘should be administered only by persons trained in the administration of general anesthesia.’ Insurers’ payment policies and marketing by the anesthesiology community have also been cited as drivers of increased use," the investigators noted.
They added that since the number of endoscopies and colonoscopies is increasing, the "discretionary" use of anesthesia in low-risk patients "represents a sizable target for cost savings."
This study was supported by Ethicon Endo-Surgery Inc. No financial conflicts of interest were reported.
The proportion of outpatient endoscopies and colonoscopies in which the services of an anesthesiologist or nurse anesthetist were used doubled in a recent 6-year period, according to a report in the March 21 issue of JAMA.
Anesthesia services include deep sedation or general anesthesia and must be administered by an anesthesiologist or nurse anesthetist, in contrast to lighter sedation that is typically provided by nurses under the endoscopist’s supervision. Current payment guidelines hold that low-risk patients can be adequately managed with light sedation and that anesthesia services are only justified in high-risk patients, according to Hangsheng Liu, Ph.D., and his associates at the RAND Corporation.
In this study of a large, nationally representative sample of patients covered by both Medicare and commercial medical insurance, the rate at which anesthesia services were used during GI procedures rose from approximately 14% in 2003 to more than 30% in 2009, they noted.
The researchers performed a retrospective analysis of insurance claims data to track the use of anesthesia services for endoscopies and colonoscopies over time because this has been identified as an area in which "excess" health care costs can be trimmed. They analyzed a Medicare fee-for-service sample of 6.6 million patients and a sample of 5.5 million patients covered by commercial insurance companies.
A medical need for anesthesia services during GI procedures in these cohorts could not be determined directly from the medical data, so it was "approximated" by classifying patients according to American Society of Anesthesiologists guidelines, which factor in age, sex, comorbidities, and medical history.
Patients who met criteria for ASA status level 1 or 2 were considered low-risk; the use of deep sedation or general anesthesia in such patients was not required and was judged to be "discretionary." In contrast, patients with an ASA status level of 3 or higher were considered high-risk and were judged to be appropriate candidates for deep sedation or general anesthesia.
Overall, there were 2.2 million GI procedures performed in Medicare patients and 7.0 million in commercially insured patients, for a total of 9.2 million during the study period.
The proportion of procedures using anesthesia services more than doubled during the 6-year study period in both groups: from 13.5% to 30.2% for Medicare patients and from 13.6% to 35.5% for commercially insured patients.
Among low-risk Medicare patients, the proportion of GI procedures using anesthesia services rose from 18,989 per million enrollees in 2003 to 25,069 per million enrollees in 2009. Annual payments per million enrollees rose by 8% per year.
Among low-risk commercially insured patients, the proportion of GI procedures using anesthesia services rose from 3,938 to 15,108 per million enrollees, and annual payments rose more than fourfold, from $1.69 million to $7.05 million per million enrollees.
Applying these figures to national statistics, the annual spending for anesthesia services on low-risk patients who probably don’t need them was estimated to be $1.1 billion in 2009 – $129 million for Medicare patients and $945 million for commercially insured patients.
Thus, "the majority of gastroenterology-related anesthesia services are provided to low-risk patients and can be considered potentially discretionary based on current payment policies," Dr. Liu and his colleagues concluded (JAMA 2012;307:1178-84).
The use of anesthesia services varied substantially by geographic region, another indication that clinical necessity is not the chief factor driving the use of heavy sedation or general anesthesia during these procedures. In the most recent year for which data were available, the use of anesthesia services during endoscopy or colonoscopy was lowest in the western United States (14% of Medicare patients and 13% of commercially insured patients) and highest in the northeast (48% and 59%, respectively). This is a nearly fourfold difference.
The rise in the use of anesthesia services "has been partly attributed to the adoption of propofol, which by virtue of a short half-life and rapid onset of action is thought to be more convenient and to offer a more consistent level of sedation than regimens previously used, but in the United States ‘should be administered only by persons trained in the administration of general anesthesia.’ Insurers’ payment policies and marketing by the anesthesiology community have also been cited as drivers of increased use," the investigators noted.
They added that since the number of endoscopies and colonoscopies is increasing, the "discretionary" use of anesthesia in low-risk patients "represents a sizable target for cost savings."
This study was supported by Ethicon Endo-Surgery Inc. No financial conflicts of interest were reported.
FROM JAMA
Major Finding: The proportion of endoscopies and colonoscopies in which anesthesia services were used increased from 14% in 2003 to more than 30% in 2009.
Data Source: This was a retrospective analysis of claims data on 1.1 million Medicare fee-for-service patients and 5.5 million commercially insured patients who underwent outpatient upper GI endoscopy or colonoscopy in 2003-2009.
Disclosures: This study was supported by Ethicon Endo-Surgery Inc. No financial conflicts of interest were reported.
More Extensive Gene Profiling Urged in AML, MDS
More detailed genetic profiling of patients with acute myeloid leukemia and of those with precursor myelodysplastic syndromes is likely to improve prognostic and therapeutic decision making, according to two separate studies published online March 14 in the New England Journal of Medicine.
In one study, investigators found that the presence of DNMT3A and NPM1 mutations and MLL translocations predicted an improved outcome when patients received high-dose daunorubicin instead of the standard dose in induction chemotherapy for acute myeloid leukemia (AML).
The results suggest that "mutational profiling can be used to determine which patients will benefit from dose-intensive induction therapy," wrote Jay P. Patel of the human oncology and pathogenesis program at Memorial Sloan-Kettering Cancer Center, New York, and his associates.
In the other study, researchers reported that "nearly all" of the bone marrow cells were clonally derived in paired samples of skin and bone marrow from seven patients with myelodysplastic syndromes (MDS) and secondary AML. Founding clones and daughter subclones in all seven paired samples had recurrent gene mutations, including at least one mutation in a coding gene.
"Although clonality is not sufficient to define malignant transformation, it is a cardinal manifestation of most human cancers, and our findings suggest that the myelodysplastic syndromes and secondary AML are both highly clonal hematologic cancers," said Dr. Matthew J. Walter of the departments of internal medicine and genetics at the Siteman Cancer Center, Washington University, St. Louis, and his associates.
Mutational Analysis of Trial Results
In the first study, researchers performed a more-extensive mutational analysis than is typically done to better discriminate among patients with different prognoses.
"Previous studies have suggested that mutational analysis of [the genes] CEBPA, NPM1, and FLT3-ITD can be used to stratify risk among patients with intermediate-risk AML," wrote Mr. Patel and his colleagues.
"We hypothesized that integrated mutational analysis of all known molecular alterations occurring in more than 5% of patients with AML would allow us to identify novel molecular markers of outcome ... and to identify molecularly defined subgroups of patients who would benefit from dose-intensified induction therapy."
For DNA extraction and profiling, the investigators used diagnostic samples of bone marrow and peripheral blood from 398 patients who were participating in the phase III ECOG (Eastern Cooperative Oncology Group) E1900 clinical trial in which two doses of induction therapy were tested. They found that 97.3% of the study subjects had mutations in 18 genes, and performed extensive mutational analysis of these 18 candidate genes.
The results led them to identify three distinct risk groups. Patients with favorable genetic profiles had a 3-year overall survival of 64% and had not yet reached a median survival; those with intermediate-risk genetic profiles had a 3-year survival of 42% and a median survival of 25 months; and those with unfavorable genetic profiles had a 3-year overall survival of 12% and a median survival of 10 months.
These findings were then validated in a separate group of 104 patients from the same clinical trial. The value of the genetic risk profiles was confirmed, with the favorable, intermediate, and unfavorable profiles accurately predicting patient outcomes independently of patient age, white cell count, induction dose, transplantation status, and type of postremission therapy, Mr. Patel and his colleagues said (N. Engl. J. Med. 2012 March 14 [doi:10.1056/NEJMoa1112304]).
Moreover, the 3-year overall survival rate in patients with a mutation in the DNMT3A or NPM1 genes or a MLL translocation was 44% with high-dose chemotherapy vs. 25% with the standard dose. In patients with other genotypes, it was 35% with the high-dose regimen and 39% with the standard dose.
"These data indicate that more detailed genetic analysis may lead to improved risk stratification and identification of patients who can benefit from more intensive induction chemotherapy. The challenge is to provide genetic information in a timely and affordable way and show that this information could prospectively influence treatment decisions," they noted.
Founding MDS Clones Persist in AML
In the second study, Dr. Walter and his associates used bone marrow biopsy specimens from seven patients who progressed from MDS to AML to define changes in the proportion of clonal cells and the genetic architecture that took place during that progression.
Several genes have already been identified that show recurrent mutations during this process, "but our understanding of the total number and clonal distribution of mutations in this disease is limited," they noted.
For each subject, DNA sequences were obtained from samples of normal skin, bone marrow obtained during the MDS stage, and bone marrow obtained during the secondary AML stage, to analyze mutations. In all seven samples, the founding clones (containing 182-660 mutations) persisted in the secondary samples, while acquiring at least one new mutation predicting translational consequences.
"We have found that the proportion of neoplastic bone marrow cells is indistinguishable [between] myelodysplastic-syndrome and secondary-AML samples, suggesting that the myelodysplastic syndromes are as clonal as secondary AML," Dr. Walter and his colleagues said (N. Engl. J. Med. 2012 March 14 [doi:10.1056/NEJMoa1106968]).
There are three major clinical implications, according to the authors.
First, MDS is currently distinguished from secondary AML based on hand counting of bone marrow myeloblasts – a method prone to inaccuracy but nonetheless relied upon to drive major treatment decisions. "Ultimately, identifying the patterns of pathogenic mutations and their clonality in bone marrow samples ... should lead to greater diagnostic certainty and improved prognostic algorithms," the investigators said.
Second, the dominant AML clone was derived from a founding MDS clone in every case, suggesting that "therapies targeted to these early mutations might be the most effective strategy for eliminating disease-propagating cells and improving the rate of response to traditional chemotherapy."
Third, it is possible that progression from MDS to AML "is driven not only by the presence of recurrent mutations ... but also by the clone ([that is], founding vs. daughter) in which they arise." Combining genotyping of samples with analysis of the clonal architecture "may yield more informative biomarkers and a better understanding of the pathogenesis of the myelodysplastic syndrome," Dr. Walter and his associates said.
Dr. Patel’s study was supported by the National Cancer Institute Physical Sciences Oncology Center, Gabrielle’s Angel Fund, the Starr Cancer Consortium, the Peter Solomon Fund, the American Society of Hematology, the Leukemia and Lymphoma Society, the Fund for Scientific Research Flanders, Burroughs Wellcome, the Sackler Center for Biomedical and Research Sciences, and the Howard Hughes Medical Institute. One of Dr. Patel’s associates reported ties to Agios, Incyte, and Novartis. Dr. Walter’s study was supported by the National Institutes of Health, the Howard Hughes Medical Institute, and the National Center for Research Resources. He and his associates reported no financial conflicts of interest.
The findings of Dr. Patel and colleagues "are sufficient to justify the expansion of the number of genetic mutations being examined in patients with AML at presentation, beyond the current analysis of [the] FLT3, NPM1, and CEBPA [genes]." These results also "challenge the field to address at what point data are compelling enough to change routine practice," said Dr. Lucy A. Godley.
The findings of Dr. Walter and colleagues also are challenging, since "it may be overwhelming for clinicians to receive a report with hundreds of gene mutations and expect them to make rational clinical decisions. An approach in which a fixed panel of genes was examined for mutations of particular clinical significance might be more affordable and the results easier to understand," she said.
Dr. Godley is in the section of hematology-oncology in the department of medicine at the University of Chicago. She reported ties to Celgene. These remarks were adapted from her editorial accompanying the two reports (N. Engl. J. Med. 2012 March 14 [doi:10.1056/NEJMe1200409]).
The findings of Dr. Patel and colleagues "are sufficient to justify the expansion of the number of genetic mutations being examined in patients with AML at presentation, beyond the current analysis of [the] FLT3, NPM1, and CEBPA [genes]." These results also "challenge the field to address at what point data are compelling enough to change routine practice," said Dr. Lucy A. Godley.
The findings of Dr. Walter and colleagues also are challenging, since "it may be overwhelming for clinicians to receive a report with hundreds of gene mutations and expect them to make rational clinical decisions. An approach in which a fixed panel of genes was examined for mutations of particular clinical significance might be more affordable and the results easier to understand," she said.
Dr. Godley is in the section of hematology-oncology in the department of medicine at the University of Chicago. She reported ties to Celgene. These remarks were adapted from her editorial accompanying the two reports (N. Engl. J. Med. 2012 March 14 [doi:10.1056/NEJMe1200409]).
The findings of Dr. Patel and colleagues "are sufficient to justify the expansion of the number of genetic mutations being examined in patients with AML at presentation, beyond the current analysis of [the] FLT3, NPM1, and CEBPA [genes]." These results also "challenge the field to address at what point data are compelling enough to change routine practice," said Dr. Lucy A. Godley.
The findings of Dr. Walter and colleagues also are challenging, since "it may be overwhelming for clinicians to receive a report with hundreds of gene mutations and expect them to make rational clinical decisions. An approach in which a fixed panel of genes was examined for mutations of particular clinical significance might be more affordable and the results easier to understand," she said.
Dr. Godley is in the section of hematology-oncology in the department of medicine at the University of Chicago. She reported ties to Celgene. These remarks were adapted from her editorial accompanying the two reports (N. Engl. J. Med. 2012 March 14 [doi:10.1056/NEJMe1200409]).
More detailed genetic profiling of patients with acute myeloid leukemia and of those with precursor myelodysplastic syndromes is likely to improve prognostic and therapeutic decision making, according to two separate studies published online March 14 in the New England Journal of Medicine.
In one study, investigators found that the presence of DNMT3A and NPM1 mutations and MLL translocations predicted an improved outcome when patients received high-dose daunorubicin instead of the standard dose in induction chemotherapy for acute myeloid leukemia (AML).
The results suggest that "mutational profiling can be used to determine which patients will benefit from dose-intensive induction therapy," wrote Jay P. Patel of the human oncology and pathogenesis program at Memorial Sloan-Kettering Cancer Center, New York, and his associates.
In the other study, researchers reported that "nearly all" of the bone marrow cells were clonally derived in paired samples of skin and bone marrow from seven patients with myelodysplastic syndromes (MDS) and secondary AML. Founding clones and daughter subclones in all seven paired samples had recurrent gene mutations, including at least one mutation in a coding gene.
"Although clonality is not sufficient to define malignant transformation, it is a cardinal manifestation of most human cancers, and our findings suggest that the myelodysplastic syndromes and secondary AML are both highly clonal hematologic cancers," said Dr. Matthew J. Walter of the departments of internal medicine and genetics at the Siteman Cancer Center, Washington University, St. Louis, and his associates.
Mutational Analysis of Trial Results
In the first study, researchers performed a more-extensive mutational analysis than is typically done to better discriminate among patients with different prognoses.
"Previous studies have suggested that mutational analysis of [the genes] CEBPA, NPM1, and FLT3-ITD can be used to stratify risk among patients with intermediate-risk AML," wrote Mr. Patel and his colleagues.
"We hypothesized that integrated mutational analysis of all known molecular alterations occurring in more than 5% of patients with AML would allow us to identify novel molecular markers of outcome ... and to identify molecularly defined subgroups of patients who would benefit from dose-intensified induction therapy."
For DNA extraction and profiling, the investigators used diagnostic samples of bone marrow and peripheral blood from 398 patients who were participating in the phase III ECOG (Eastern Cooperative Oncology Group) E1900 clinical trial in which two doses of induction therapy were tested. They found that 97.3% of the study subjects had mutations in 18 genes, and performed extensive mutational analysis of these 18 candidate genes.
The results led them to identify three distinct risk groups. Patients with favorable genetic profiles had a 3-year overall survival of 64% and had not yet reached a median survival; those with intermediate-risk genetic profiles had a 3-year survival of 42% and a median survival of 25 months; and those with unfavorable genetic profiles had a 3-year overall survival of 12% and a median survival of 10 months.
These findings were then validated in a separate group of 104 patients from the same clinical trial. The value of the genetic risk profiles was confirmed, with the favorable, intermediate, and unfavorable profiles accurately predicting patient outcomes independently of patient age, white cell count, induction dose, transplantation status, and type of postremission therapy, Mr. Patel and his colleagues said (N. Engl. J. Med. 2012 March 14 [doi:10.1056/NEJMoa1112304]).
Moreover, the 3-year overall survival rate in patients with a mutation in the DNMT3A or NPM1 genes or a MLL translocation was 44% with high-dose chemotherapy vs. 25% with the standard dose. In patients with other genotypes, it was 35% with the high-dose regimen and 39% with the standard dose.
"These data indicate that more detailed genetic analysis may lead to improved risk stratification and identification of patients who can benefit from more intensive induction chemotherapy. The challenge is to provide genetic information in a timely and affordable way and show that this information could prospectively influence treatment decisions," they noted.
Founding MDS Clones Persist in AML
In the second study, Dr. Walter and his associates used bone marrow biopsy specimens from seven patients who progressed from MDS to AML to define changes in the proportion of clonal cells and the genetic architecture that took place during that progression.
Several genes have already been identified that show recurrent mutations during this process, "but our understanding of the total number and clonal distribution of mutations in this disease is limited," they noted.
For each subject, DNA sequences were obtained from samples of normal skin, bone marrow obtained during the MDS stage, and bone marrow obtained during the secondary AML stage, to analyze mutations. In all seven samples, the founding clones (containing 182-660 mutations) persisted in the secondary samples, while acquiring at least one new mutation predicting translational consequences.
"We have found that the proportion of neoplastic bone marrow cells is indistinguishable [between] myelodysplastic-syndrome and secondary-AML samples, suggesting that the myelodysplastic syndromes are as clonal as secondary AML," Dr. Walter and his colleagues said (N. Engl. J. Med. 2012 March 14 [doi:10.1056/NEJMoa1106968]).
There are three major clinical implications, according to the authors.
First, MDS is currently distinguished from secondary AML based on hand counting of bone marrow myeloblasts – a method prone to inaccuracy but nonetheless relied upon to drive major treatment decisions. "Ultimately, identifying the patterns of pathogenic mutations and their clonality in bone marrow samples ... should lead to greater diagnostic certainty and improved prognostic algorithms," the investigators said.
Second, the dominant AML clone was derived from a founding MDS clone in every case, suggesting that "therapies targeted to these early mutations might be the most effective strategy for eliminating disease-propagating cells and improving the rate of response to traditional chemotherapy."
Third, it is possible that progression from MDS to AML "is driven not only by the presence of recurrent mutations ... but also by the clone ([that is], founding vs. daughter) in which they arise." Combining genotyping of samples with analysis of the clonal architecture "may yield more informative biomarkers and a better understanding of the pathogenesis of the myelodysplastic syndrome," Dr. Walter and his associates said.
Dr. Patel’s study was supported by the National Cancer Institute Physical Sciences Oncology Center, Gabrielle’s Angel Fund, the Starr Cancer Consortium, the Peter Solomon Fund, the American Society of Hematology, the Leukemia and Lymphoma Society, the Fund for Scientific Research Flanders, Burroughs Wellcome, the Sackler Center for Biomedical and Research Sciences, and the Howard Hughes Medical Institute. One of Dr. Patel’s associates reported ties to Agios, Incyte, and Novartis. Dr. Walter’s study was supported by the National Institutes of Health, the Howard Hughes Medical Institute, and the National Center for Research Resources. He and his associates reported no financial conflicts of interest.
More detailed genetic profiling of patients with acute myeloid leukemia and of those with precursor myelodysplastic syndromes is likely to improve prognostic and therapeutic decision making, according to two separate studies published online March 14 in the New England Journal of Medicine.
In one study, investigators found that the presence of DNMT3A and NPM1 mutations and MLL translocations predicted an improved outcome when patients received high-dose daunorubicin instead of the standard dose in induction chemotherapy for acute myeloid leukemia (AML).
The results suggest that "mutational profiling can be used to determine which patients will benefit from dose-intensive induction therapy," wrote Jay P. Patel of the human oncology and pathogenesis program at Memorial Sloan-Kettering Cancer Center, New York, and his associates.
In the other study, researchers reported that "nearly all" of the bone marrow cells were clonally derived in paired samples of skin and bone marrow from seven patients with myelodysplastic syndromes (MDS) and secondary AML. Founding clones and daughter subclones in all seven paired samples had recurrent gene mutations, including at least one mutation in a coding gene.
"Although clonality is not sufficient to define malignant transformation, it is a cardinal manifestation of most human cancers, and our findings suggest that the myelodysplastic syndromes and secondary AML are both highly clonal hematologic cancers," said Dr. Matthew J. Walter of the departments of internal medicine and genetics at the Siteman Cancer Center, Washington University, St. Louis, and his associates.
Mutational Analysis of Trial Results
In the first study, researchers performed a more-extensive mutational analysis than is typically done to better discriminate among patients with different prognoses.
"Previous studies have suggested that mutational analysis of [the genes] CEBPA, NPM1, and FLT3-ITD can be used to stratify risk among patients with intermediate-risk AML," wrote Mr. Patel and his colleagues.
"We hypothesized that integrated mutational analysis of all known molecular alterations occurring in more than 5% of patients with AML would allow us to identify novel molecular markers of outcome ... and to identify molecularly defined subgroups of patients who would benefit from dose-intensified induction therapy."
For DNA extraction and profiling, the investigators used diagnostic samples of bone marrow and peripheral blood from 398 patients who were participating in the phase III ECOG (Eastern Cooperative Oncology Group) E1900 clinical trial in which two doses of induction therapy were tested. They found that 97.3% of the study subjects had mutations in 18 genes, and performed extensive mutational analysis of these 18 candidate genes.
The results led them to identify three distinct risk groups. Patients with favorable genetic profiles had a 3-year overall survival of 64% and had not yet reached a median survival; those with intermediate-risk genetic profiles had a 3-year survival of 42% and a median survival of 25 months; and those with unfavorable genetic profiles had a 3-year overall survival of 12% and a median survival of 10 months.
These findings were then validated in a separate group of 104 patients from the same clinical trial. The value of the genetic risk profiles was confirmed, with the favorable, intermediate, and unfavorable profiles accurately predicting patient outcomes independently of patient age, white cell count, induction dose, transplantation status, and type of postremission therapy, Mr. Patel and his colleagues said (N. Engl. J. Med. 2012 March 14 [doi:10.1056/NEJMoa1112304]).
Moreover, the 3-year overall survival rate in patients with a mutation in the DNMT3A or NPM1 genes or a MLL translocation was 44% with high-dose chemotherapy vs. 25% with the standard dose. In patients with other genotypes, it was 35% with the high-dose regimen and 39% with the standard dose.
"These data indicate that more detailed genetic analysis may lead to improved risk stratification and identification of patients who can benefit from more intensive induction chemotherapy. The challenge is to provide genetic information in a timely and affordable way and show that this information could prospectively influence treatment decisions," they noted.
Founding MDS Clones Persist in AML
In the second study, Dr. Walter and his associates used bone marrow biopsy specimens from seven patients who progressed from MDS to AML to define changes in the proportion of clonal cells and the genetic architecture that took place during that progression.
Several genes have already been identified that show recurrent mutations during this process, "but our understanding of the total number and clonal distribution of mutations in this disease is limited," they noted.
For each subject, DNA sequences were obtained from samples of normal skin, bone marrow obtained during the MDS stage, and bone marrow obtained during the secondary AML stage, to analyze mutations. In all seven samples, the founding clones (containing 182-660 mutations) persisted in the secondary samples, while acquiring at least one new mutation predicting translational consequences.
"We have found that the proportion of neoplastic bone marrow cells is indistinguishable [between] myelodysplastic-syndrome and secondary-AML samples, suggesting that the myelodysplastic syndromes are as clonal as secondary AML," Dr. Walter and his colleagues said (N. Engl. J. Med. 2012 March 14 [doi:10.1056/NEJMoa1106968]).
There are three major clinical implications, according to the authors.
First, MDS is currently distinguished from secondary AML based on hand counting of bone marrow myeloblasts – a method prone to inaccuracy but nonetheless relied upon to drive major treatment decisions. "Ultimately, identifying the patterns of pathogenic mutations and their clonality in bone marrow samples ... should lead to greater diagnostic certainty and improved prognostic algorithms," the investigators said.
Second, the dominant AML clone was derived from a founding MDS clone in every case, suggesting that "therapies targeted to these early mutations might be the most effective strategy for eliminating disease-propagating cells and improving the rate of response to traditional chemotherapy."
Third, it is possible that progression from MDS to AML "is driven not only by the presence of recurrent mutations ... but also by the clone ([that is], founding vs. daughter) in which they arise." Combining genotyping of samples with analysis of the clonal architecture "may yield more informative biomarkers and a better understanding of the pathogenesis of the myelodysplastic syndrome," Dr. Walter and his associates said.
Dr. Patel’s study was supported by the National Cancer Institute Physical Sciences Oncology Center, Gabrielle’s Angel Fund, the Starr Cancer Consortium, the Peter Solomon Fund, the American Society of Hematology, the Leukemia and Lymphoma Society, the Fund for Scientific Research Flanders, Burroughs Wellcome, the Sackler Center for Biomedical and Research Sciences, and the Howard Hughes Medical Institute. One of Dr. Patel’s associates reported ties to Agios, Incyte, and Novartis. Dr. Walter’s study was supported by the National Institutes of Health, the Howard Hughes Medical Institute, and the National Center for Research Resources. He and his associates reported no financial conflicts of interest.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: In one study 97.3% of samples from patients with AML had mutations in 18 genes. In the other, the founding clone in seven MDS samples persisted in all seven samples of secondary AML from the same patients.
Data Source: The first study was an extensive mutational analysis of 398 patients with AML, with findings confirmed in a validation cohort of 104 patients. The second was a whole-genome sequencing of paired samples of skin and bone marrow from seven patients who had MDS that progressed to secondary AML.
Disclosures: Dr. Patel’s study was supported by the National Cancer Institute Physical Sciences Oncology Center, Gabrielle’s Angel Fund, the Starr Cancer Consortium, the Peter Solomon Fund, the American Society of Hematology, the Leukemia and Lymphoma Society, the Fund for Scientific Research Flanders, Burroughs Wellcome, the Sackler Center for Biomedical and Research Sciences, and the Howard Hughes Medical Institute. One of Dr. Patel’s associates reported ties to Agios, Incyte, and Novartis. Dr. Walter’s study was supported by the National Institutes of Health, the Howard Hughes Medical Institute, and the National Center for Research Resources. He and his associates reported no financial conflicts of interest.
Study Results Support Ingenol Mebutate's AK Effectiveness
Topical ingenol mebutate gel effectively treated actinic keratoses when applied to the face, scalp, trunk, or extremities for 2-3 days, according to a report in the March 15 issue of the New England Journal of Medicine.
Compared with existing therapies for actinic keratoses, the chief advantage of ingenol mebutate gel is the short exposure time, reported Dr. Mark Lebwohl, professor and chairman of the department of dermatology at Mount Sinai School of Medicine, New York, and his associates. This allows for relatively rapid resolution of local reactions, and it will likely improve adherence to treatment, which in turn should improve the therapy’s effectiveness, they noted.
"Many patients find it difficult to adhere to the currently available regimens of topical treatment that last for periods of 1-4 months, which may result in ‘real-world’ effectiveness lower than that achieved in supervised and patient-compensated clinical trials," Dr. Lebwohl and his colleagues wrote.
The researchers evaluated the safety and effectiveness of ingenol mebutate gel compared with a placebo in a manufacturer-sponsored, randomized, double-blind trial of 1,005 patients treated at four medical centers. The majority of the patients had Fitzpatrick type I or II skin, and their mean age was 65 years.
Approximately half of the study patients had a history of skin cancer, and 75% had already had their keratosis treated with cryotherapy, imiquimod, or topical fluorouracil.
In each patient, a 25-cm contiguous field containing at least four to eight clinically typical and discrete actinic keratoses was selected for treatment. The study patients were divided into two groups according to the location of the treated area: on the head (face or scalp), and on the body (trunk or extremities).
Those with facial or scalp lesions were randomly assigned to self-apply either 0.015% active gel (277 patients) or placebo gel (270 patients) to the area once daily for 3 consecutive days. Those with trunk or extremity lesions were randomly assigned to self-apply 0.05% active gel (226 patients) or placebo gel (232 patients) to the area once daily for 2 consecutive days.
The primary end point was complete clearance of all clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 42.2% of those who used active gel reached this end point, compared with only 3.7% of those who used placebo gel. Among patients with trunk or extremity lesions, 34.1% who used active gel reached this end point, compared with only 4.7% of those who used placebo gel.
In addition, patients with face or scalp lesions that were treated with ingenol mebutate showed a median reduction of 83% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who needed to be treated with ingenol mebutate to obtain complete clearance in one patient was 2.6," the investigators reported (N. Engl. J. Med. 2012;366:1010-19).
Patients with trunk or extremity lesions treated with ingenol mebutate showed a median reduction of 75% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who would need to be treated with ingenol mebutate to obtain complete clearance in one patient was 3.4," the researchers noted.
A secondary end point was partial (75% or more) clearance in the number of clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 63.9% of those who used active gel reached this end point, compared with only 7.4% of those who used placebo gel. Among patients with trunk or extremity lesions, 49.1% of those who used active gel reached this end point, compared with only 6.9% of those who used placebo gel.
Patients who used ingenol mebutate showed minimal scarring or change in pigmentation. Local reactions such as erythema, crusting, swelling, vesiculation, or pustulation were common, but were mild to moderate in intensity; they peaked within a few days of treatment and resolved rapidly afterward, with no sequelae. One patient developed eye pain, burning, and periorbital edema related to ingenol mebutate and dropped out of the study, Dr. Lebwohl and his associates noted.
"Future studies are needed to assess the risks and benefits of treating larger areas of skin, using multiple treatments in the same area, and using combination therapies," they added.
This study was funded by LEO Pharma, maker of ingenol mebutate. Dr. Lebwohl reported ties to LEO Pharma, Graceway, PharmaDerm, and Peplin LTD.
Topical ingenol mebutate gel effectively treated actinic keratoses when applied to the face, scalp, trunk, or extremities for 2-3 days, according to a report in the March 15 issue of the New England Journal of Medicine.
Compared with existing therapies for actinic keratoses, the chief advantage of ingenol mebutate gel is the short exposure time, reported Dr. Mark Lebwohl, professor and chairman of the department of dermatology at Mount Sinai School of Medicine, New York, and his associates. This allows for relatively rapid resolution of local reactions, and it will likely improve adherence to treatment, which in turn should improve the therapy’s effectiveness, they noted.
"Many patients find it difficult to adhere to the currently available regimens of topical treatment that last for periods of 1-4 months, which may result in ‘real-world’ effectiveness lower than that achieved in supervised and patient-compensated clinical trials," Dr. Lebwohl and his colleagues wrote.
The researchers evaluated the safety and effectiveness of ingenol mebutate gel compared with a placebo in a manufacturer-sponsored, randomized, double-blind trial of 1,005 patients treated at four medical centers. The majority of the patients had Fitzpatrick type I or II skin, and their mean age was 65 years.
Approximately half of the study patients had a history of skin cancer, and 75% had already had their keratosis treated with cryotherapy, imiquimod, or topical fluorouracil.
In each patient, a 25-cm contiguous field containing at least four to eight clinically typical and discrete actinic keratoses was selected for treatment. The study patients were divided into two groups according to the location of the treated area: on the head (face or scalp), and on the body (trunk or extremities).
Those with facial or scalp lesions were randomly assigned to self-apply either 0.015% active gel (277 patients) or placebo gel (270 patients) to the area once daily for 3 consecutive days. Those with trunk or extremity lesions were randomly assigned to self-apply 0.05% active gel (226 patients) or placebo gel (232 patients) to the area once daily for 2 consecutive days.
The primary end point was complete clearance of all clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 42.2% of those who used active gel reached this end point, compared with only 3.7% of those who used placebo gel. Among patients with trunk or extremity lesions, 34.1% who used active gel reached this end point, compared with only 4.7% of those who used placebo gel.
In addition, patients with face or scalp lesions that were treated with ingenol mebutate showed a median reduction of 83% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who needed to be treated with ingenol mebutate to obtain complete clearance in one patient was 2.6," the investigators reported (N. Engl. J. Med. 2012;366:1010-19).
Patients with trunk or extremity lesions treated with ingenol mebutate showed a median reduction of 75% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who would need to be treated with ingenol mebutate to obtain complete clearance in one patient was 3.4," the researchers noted.
A secondary end point was partial (75% or more) clearance in the number of clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 63.9% of those who used active gel reached this end point, compared with only 7.4% of those who used placebo gel. Among patients with trunk or extremity lesions, 49.1% of those who used active gel reached this end point, compared with only 6.9% of those who used placebo gel.
Patients who used ingenol mebutate showed minimal scarring or change in pigmentation. Local reactions such as erythema, crusting, swelling, vesiculation, or pustulation were common, but were mild to moderate in intensity; they peaked within a few days of treatment and resolved rapidly afterward, with no sequelae. One patient developed eye pain, burning, and periorbital edema related to ingenol mebutate and dropped out of the study, Dr. Lebwohl and his associates noted.
"Future studies are needed to assess the risks and benefits of treating larger areas of skin, using multiple treatments in the same area, and using combination therapies," they added.
This study was funded by LEO Pharma, maker of ingenol mebutate. Dr. Lebwohl reported ties to LEO Pharma, Graceway, PharmaDerm, and Peplin LTD.
Topical ingenol mebutate gel effectively treated actinic keratoses when applied to the face, scalp, trunk, or extremities for 2-3 days, according to a report in the March 15 issue of the New England Journal of Medicine.
Compared with existing therapies for actinic keratoses, the chief advantage of ingenol mebutate gel is the short exposure time, reported Dr. Mark Lebwohl, professor and chairman of the department of dermatology at Mount Sinai School of Medicine, New York, and his associates. This allows for relatively rapid resolution of local reactions, and it will likely improve adherence to treatment, which in turn should improve the therapy’s effectiveness, they noted.
"Many patients find it difficult to adhere to the currently available regimens of topical treatment that last for periods of 1-4 months, which may result in ‘real-world’ effectiveness lower than that achieved in supervised and patient-compensated clinical trials," Dr. Lebwohl and his colleagues wrote.
The researchers evaluated the safety and effectiveness of ingenol mebutate gel compared with a placebo in a manufacturer-sponsored, randomized, double-blind trial of 1,005 patients treated at four medical centers. The majority of the patients had Fitzpatrick type I or II skin, and their mean age was 65 years.
Approximately half of the study patients had a history of skin cancer, and 75% had already had their keratosis treated with cryotherapy, imiquimod, or topical fluorouracil.
In each patient, a 25-cm contiguous field containing at least four to eight clinically typical and discrete actinic keratoses was selected for treatment. The study patients were divided into two groups according to the location of the treated area: on the head (face or scalp), and on the body (trunk or extremities).
Those with facial or scalp lesions were randomly assigned to self-apply either 0.015% active gel (277 patients) or placebo gel (270 patients) to the area once daily for 3 consecutive days. Those with trunk or extremity lesions were randomly assigned to self-apply 0.05% active gel (226 patients) or placebo gel (232 patients) to the area once daily for 2 consecutive days.
The primary end point was complete clearance of all clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 42.2% of those who used active gel reached this end point, compared with only 3.7% of those who used placebo gel. Among patients with trunk or extremity lesions, 34.1% who used active gel reached this end point, compared with only 4.7% of those who used placebo gel.
In addition, patients with face or scalp lesions that were treated with ingenol mebutate showed a median reduction of 83% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who needed to be treated with ingenol mebutate to obtain complete clearance in one patient was 2.6," the investigators reported (N. Engl. J. Med. 2012;366:1010-19).
Patients with trunk or extremity lesions treated with ingenol mebutate showed a median reduction of 75% in the number of actinic keratoses, compared with a 0% reduction with placebo gel. "We calculated that the number of patients who would need to be treated with ingenol mebutate to obtain complete clearance in one patient was 3.4," the researchers noted.
A secondary end point was partial (75% or more) clearance in the number of clinically visible actinic keratoses in the treatment area on day 57.
Among patients with facial or scalp lesions, 63.9% of those who used active gel reached this end point, compared with only 7.4% of those who used placebo gel. Among patients with trunk or extremity lesions, 49.1% of those who used active gel reached this end point, compared with only 6.9% of those who used placebo gel.
Patients who used ingenol mebutate showed minimal scarring or change in pigmentation. Local reactions such as erythema, crusting, swelling, vesiculation, or pustulation were common, but were mild to moderate in intensity; they peaked within a few days of treatment and resolved rapidly afterward, with no sequelae. One patient developed eye pain, burning, and periorbital edema related to ingenol mebutate and dropped out of the study, Dr. Lebwohl and his associates noted.
"Future studies are needed to assess the risks and benefits of treating larger areas of skin, using multiple treatments in the same area, and using combination therapies," they added.
This study was funded by LEO Pharma, maker of ingenol mebutate. Dr. Lebwohl reported ties to LEO Pharma, Graceway, PharmaDerm, and Peplin LTD.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: In patients with facial or scalp lesions, 42.2% who used ingenol mebutate gel achieved complete clearance at 2 months, compared with only 3.7% of those who used placebo gel; in patients with trunk or extremity lesions, 34.1% who used active gel achieved complete clearance, compared with only 4.7% of those who used placebo gel.
Data Source: Data are from a manufacturer-sponsored, randomized, double-blind trial of 1,005 patients treated at four medical centers.
Disclosures: This study was funded by LEO Pharma, maker of ingenol mebutate. Dr. Lebwohl reported ties to LEO Pharma, Graceway, PharmaDerm, and Peplin LTD.
Higher Spending Linked to Better Hospital Care
Higher spending intensity was associated with substantially better hospital care in Ontario, which has a universal health care system, according to a report in the March 14 issue of JAMA.
Patients with acute illness treated at hospitals with higher spending intensity had lower mortality, lower readmission rates, and fewer adverse events than did those treated at hospitals with lower spending intensity, said Therese A. Stukel, Ph.D., of the Institute for Clinical Evaluative Sciences, Toronto, and her associates.
However, it would be "facile" to conclude that more spending necessarily leads to better patient outcomes, and that providing more money to lower-spending facilities would necessarily improve their patient outcomes. "Higher-spending hospitals differed in many ways, such as greater use of evidence-based care, skilled nursing and critical care staff, more intensive inpatient specialist services, and high technology, all of which are more expensive," the investigators noted.
Many studies have examined whether higher health care spending produces better patient outcomes, but until now, none have assessed the issue in an area with universal access to health care but a lower supply of specialists and of medical technology than in the United States. "Our objective was to assess whether acute care patients admitted to Canadian hospitals that treat patients more intensively (and at higher cost) have lower mortality and readmissions and higher quality of care," Dr. Stukel and her colleagues said.
They analyzed the medical records of adults with an index admission to one of 129 acute care hospitals in Ontario in 1998-2008 for any of four common conditions for which treatment follows relatively standard protocols: acute myocardial infarction (179,139 patients), heart failure (92,377 patients), hip fracture (90,046 patients), or surgical resection of colon cancer (26,195 patients). The study subjects were followed for 1 year after the index admission.
Several quality-of-care measures were assessed: whether patients received preoperative visits from a surgeon and an anesthetist, whether surgery took place within 2 days of admission, whether patients received in-hospital rehabilitation, whether MI patients underwent same-day percutaneous coronary intervention, and the number of visits from medical specialists during the hospital stay.
Higher-spending hospitals tended to be high-volume centers in urban areas. They tended to be affiliated with regional cancer centers and to have on-site CT, MRI, cardiac catheterization, and cardiac surgery capabilities. They also tended to employ critical care response teams, as well as attending physicians who were specialists. And they provided 30% more inpatient nursing hours per patient-day and per bed than did lower-spending hospitals.
At higher-spending hospitals, 30-day mortality was 12.7% (vs 12.8%) for acute MI, 10.2% (vs. 12.4%) for CHF, 7.7% (vs. 9.7%) for hip fracture, and 3.3% (vs 3.9%) for colon cancer. The 30-day major cardiac event rate was 17.4% (vs.18.7%) for acute MI and 15.0% (vs. 17.6%) for CHF. And the 30-day readmission rate was 23.1% (vs. 25.8%) for hip fracture and 10.3% (vs 13.1%) for colon cancer.
After the data were adjusted fully to account for patient age, sex, illness severity, and other variables, mortality and readmission rates remained significantly lower in high-spending hospitals for every study subgroup, Dr. Stukel and her colleagues said (JAMA 2012;307:1037-45).
Compared with patients at lower-spending hospitals, those at higher-spending hospitals were more likely to see a medical specialist during their stay. Cardiac patients at higher-spending hospitals were more likely to receive the indicated cardiac interventions and evidence-based medications, to attend ambulatory care within 1 month, and to visit a cardiologist within 1 year.
CHF patients at higher-spending hospitals were less likely than were those at lower-spending hospitals to receive contraindicated medications. Those with hip fracture were more likely to begin rehabilitation during their inpatient stay. And those with colon cancer were more likely to have a preoperative consultation with a surgeon and an anesthetist, and to undergo CT for preoperative staging, compared with colon cancer patients at lower-spending hospitals.
The study findings "suggest that it is critical to understand not simply how much money is spent but whether it is spent on effective procedures and services," the researchers noted.
This study was supported by the Canadian Institute of Health Research, the U.S. National Institute on Aging, the Institute for Clinical Evaluative Sciences, and the Ontario Ministry of Health and Long-Term Care. No relevant financial conflicts of interest were reported.
"The notion that payments to hospitals can be reduced while maintaining or improving the quality of care delivered at these hospitals has become so ingrained in policy circles as to be a given," said Dr. Karen E. Joynt and Dr. Ashish K. Jha.
Dr. Karen E. Joynt |
Dr. Ashish K. Jha |
"Although paying hospitals less may appear to be a good strategy to save money, the findings reported by Stukel et al. serve as a timely reminder that this approach is likely to have negative consequences for patients," they noted.
So-called "expensive" hospitals are likely spending that money directly on nurses, specialists, and technology – in other words, on care that improves the outcomes of acutely ill patients, Dr. Joynt and Dr. Jha said.
Dr. Joynt and Dr. Jha are in the department of health policy and management at Harvard School of Public Health, Boston. Dr. Joynt is also in the cardiovascular division and Dr. Jha is in the division of general internal medicine at Brigham and Women’s Hospital, Boston. They reported no relevant financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Stukel’s report (JAMA 2012;307:1082-3).
"The notion that payments to hospitals can be reduced while maintaining or improving the quality of care delivered at these hospitals has become so ingrained in policy circles as to be a given," said Dr. Karen E. Joynt and Dr. Ashish K. Jha.
Dr. Karen E. Joynt |
Dr. Ashish K. Jha |
"Although paying hospitals less may appear to be a good strategy to save money, the findings reported by Stukel et al. serve as a timely reminder that this approach is likely to have negative consequences for patients," they noted.
So-called "expensive" hospitals are likely spending that money directly on nurses, specialists, and technology – in other words, on care that improves the outcomes of acutely ill patients, Dr. Joynt and Dr. Jha said.
Dr. Joynt and Dr. Jha are in the department of health policy and management at Harvard School of Public Health, Boston. Dr. Joynt is also in the cardiovascular division and Dr. Jha is in the division of general internal medicine at Brigham and Women’s Hospital, Boston. They reported no relevant financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Stukel’s report (JAMA 2012;307:1082-3).
"The notion that payments to hospitals can be reduced while maintaining or improving the quality of care delivered at these hospitals has become so ingrained in policy circles as to be a given," said Dr. Karen E. Joynt and Dr. Ashish K. Jha.
Dr. Karen E. Joynt |
Dr. Ashish K. Jha |
"Although paying hospitals less may appear to be a good strategy to save money, the findings reported by Stukel et al. serve as a timely reminder that this approach is likely to have negative consequences for patients," they noted.
So-called "expensive" hospitals are likely spending that money directly on nurses, specialists, and technology – in other words, on care that improves the outcomes of acutely ill patients, Dr. Joynt and Dr. Jha said.
Dr. Joynt and Dr. Jha are in the department of health policy and management at Harvard School of Public Health, Boston. Dr. Joynt is also in the cardiovascular division and Dr. Jha is in the division of general internal medicine at Brigham and Women’s Hospital, Boston. They reported no relevant financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Stukel’s report (JAMA 2012;307:1082-3).
Higher spending intensity was associated with substantially better hospital care in Ontario, which has a universal health care system, according to a report in the March 14 issue of JAMA.
Patients with acute illness treated at hospitals with higher spending intensity had lower mortality, lower readmission rates, and fewer adverse events than did those treated at hospitals with lower spending intensity, said Therese A. Stukel, Ph.D., of the Institute for Clinical Evaluative Sciences, Toronto, and her associates.
However, it would be "facile" to conclude that more spending necessarily leads to better patient outcomes, and that providing more money to lower-spending facilities would necessarily improve their patient outcomes. "Higher-spending hospitals differed in many ways, such as greater use of evidence-based care, skilled nursing and critical care staff, more intensive inpatient specialist services, and high technology, all of which are more expensive," the investigators noted.
Many studies have examined whether higher health care spending produces better patient outcomes, but until now, none have assessed the issue in an area with universal access to health care but a lower supply of specialists and of medical technology than in the United States. "Our objective was to assess whether acute care patients admitted to Canadian hospitals that treat patients more intensively (and at higher cost) have lower mortality and readmissions and higher quality of care," Dr. Stukel and her colleagues said.
They analyzed the medical records of adults with an index admission to one of 129 acute care hospitals in Ontario in 1998-2008 for any of four common conditions for which treatment follows relatively standard protocols: acute myocardial infarction (179,139 patients), heart failure (92,377 patients), hip fracture (90,046 patients), or surgical resection of colon cancer (26,195 patients). The study subjects were followed for 1 year after the index admission.
Several quality-of-care measures were assessed: whether patients received preoperative visits from a surgeon and an anesthetist, whether surgery took place within 2 days of admission, whether patients received in-hospital rehabilitation, whether MI patients underwent same-day percutaneous coronary intervention, and the number of visits from medical specialists during the hospital stay.
Higher-spending hospitals tended to be high-volume centers in urban areas. They tended to be affiliated with regional cancer centers and to have on-site CT, MRI, cardiac catheterization, and cardiac surgery capabilities. They also tended to employ critical care response teams, as well as attending physicians who were specialists. And they provided 30% more inpatient nursing hours per patient-day and per bed than did lower-spending hospitals.
At higher-spending hospitals, 30-day mortality was 12.7% (vs 12.8%) for acute MI, 10.2% (vs. 12.4%) for CHF, 7.7% (vs. 9.7%) for hip fracture, and 3.3% (vs 3.9%) for colon cancer. The 30-day major cardiac event rate was 17.4% (vs.18.7%) for acute MI and 15.0% (vs. 17.6%) for CHF. And the 30-day readmission rate was 23.1% (vs. 25.8%) for hip fracture and 10.3% (vs 13.1%) for colon cancer.
After the data were adjusted fully to account for patient age, sex, illness severity, and other variables, mortality and readmission rates remained significantly lower in high-spending hospitals for every study subgroup, Dr. Stukel and her colleagues said (JAMA 2012;307:1037-45).
Compared with patients at lower-spending hospitals, those at higher-spending hospitals were more likely to see a medical specialist during their stay. Cardiac patients at higher-spending hospitals were more likely to receive the indicated cardiac interventions and evidence-based medications, to attend ambulatory care within 1 month, and to visit a cardiologist within 1 year.
CHF patients at higher-spending hospitals were less likely than were those at lower-spending hospitals to receive contraindicated medications. Those with hip fracture were more likely to begin rehabilitation during their inpatient stay. And those with colon cancer were more likely to have a preoperative consultation with a surgeon and an anesthetist, and to undergo CT for preoperative staging, compared with colon cancer patients at lower-spending hospitals.
The study findings "suggest that it is critical to understand not simply how much money is spent but whether it is spent on effective procedures and services," the researchers noted.
This study was supported by the Canadian Institute of Health Research, the U.S. National Institute on Aging, the Institute for Clinical Evaluative Sciences, and the Ontario Ministry of Health and Long-Term Care. No relevant financial conflicts of interest were reported.
Higher spending intensity was associated with substantially better hospital care in Ontario, which has a universal health care system, according to a report in the March 14 issue of JAMA.
Patients with acute illness treated at hospitals with higher spending intensity had lower mortality, lower readmission rates, and fewer adverse events than did those treated at hospitals with lower spending intensity, said Therese A. Stukel, Ph.D., of the Institute for Clinical Evaluative Sciences, Toronto, and her associates.
However, it would be "facile" to conclude that more spending necessarily leads to better patient outcomes, and that providing more money to lower-spending facilities would necessarily improve their patient outcomes. "Higher-spending hospitals differed in many ways, such as greater use of evidence-based care, skilled nursing and critical care staff, more intensive inpatient specialist services, and high technology, all of which are more expensive," the investigators noted.
Many studies have examined whether higher health care spending produces better patient outcomes, but until now, none have assessed the issue in an area with universal access to health care but a lower supply of specialists and of medical technology than in the United States. "Our objective was to assess whether acute care patients admitted to Canadian hospitals that treat patients more intensively (and at higher cost) have lower mortality and readmissions and higher quality of care," Dr. Stukel and her colleagues said.
They analyzed the medical records of adults with an index admission to one of 129 acute care hospitals in Ontario in 1998-2008 for any of four common conditions for which treatment follows relatively standard protocols: acute myocardial infarction (179,139 patients), heart failure (92,377 patients), hip fracture (90,046 patients), or surgical resection of colon cancer (26,195 patients). The study subjects were followed for 1 year after the index admission.
Several quality-of-care measures were assessed: whether patients received preoperative visits from a surgeon and an anesthetist, whether surgery took place within 2 days of admission, whether patients received in-hospital rehabilitation, whether MI patients underwent same-day percutaneous coronary intervention, and the number of visits from medical specialists during the hospital stay.
Higher-spending hospitals tended to be high-volume centers in urban areas. They tended to be affiliated with regional cancer centers and to have on-site CT, MRI, cardiac catheterization, and cardiac surgery capabilities. They also tended to employ critical care response teams, as well as attending physicians who were specialists. And they provided 30% more inpatient nursing hours per patient-day and per bed than did lower-spending hospitals.
At higher-spending hospitals, 30-day mortality was 12.7% (vs 12.8%) for acute MI, 10.2% (vs. 12.4%) for CHF, 7.7% (vs. 9.7%) for hip fracture, and 3.3% (vs 3.9%) for colon cancer. The 30-day major cardiac event rate was 17.4% (vs.18.7%) for acute MI and 15.0% (vs. 17.6%) for CHF. And the 30-day readmission rate was 23.1% (vs. 25.8%) for hip fracture and 10.3% (vs 13.1%) for colon cancer.
After the data were adjusted fully to account for patient age, sex, illness severity, and other variables, mortality and readmission rates remained significantly lower in high-spending hospitals for every study subgroup, Dr. Stukel and her colleagues said (JAMA 2012;307:1037-45).
Compared with patients at lower-spending hospitals, those at higher-spending hospitals were more likely to see a medical specialist during their stay. Cardiac patients at higher-spending hospitals were more likely to receive the indicated cardiac interventions and evidence-based medications, to attend ambulatory care within 1 month, and to visit a cardiologist within 1 year.
CHF patients at higher-spending hospitals were less likely than were those at lower-spending hospitals to receive contraindicated medications. Those with hip fracture were more likely to begin rehabilitation during their inpatient stay. And those with colon cancer were more likely to have a preoperative consultation with a surgeon and an anesthetist, and to undergo CT for preoperative staging, compared with colon cancer patients at lower-spending hospitals.
The study findings "suggest that it is critical to understand not simply how much money is spent but whether it is spent on effective procedures and services," the researchers noted.
This study was supported by the Canadian Institute of Health Research, the U.S. National Institute on Aging, the Institute for Clinical Evaluative Sciences, and the Ontario Ministry of Health and Long-Term Care. No relevant financial conflicts of interest were reported.
FROM JAMA
Major Finding: Thirty-day and 1-year rates of mortality and readmission were consistently lower in patients treated at hospitals with higher spending intensity than in those treated at hospitals with lower spending intensity.
Data Source: A longitudinal cohort study involving 179,139 men and women with acute MI, 92,377 with CHF, 90,046 with hip fracture, and 26,195 with colon cancer requiring surgical resection, all of whom were hospitalized at 129 Ontario medical centers in 1998-2008 and followed for 1 year.
Disclosures: This study was supported by the Canadian Institute of Health Research, the U.S. National Institute on Aging, the Institute for Clinical Evaluative Sciences, and the Ontario Ministry of Health and Long-Term Care. No relevant financial conflicts of interest were reported.
Azithromycin Cuts Carriage of Shiga Toxin-Producing E. coli
Among patients infected with Shiga toxin–producing Escherichia coli during the 2011 outbreak in Germany, azithromycin significantly decreased colonization with the organism, allowing patients to return to normal work and social activities instead of being quarantined, according to a report in the March 14 JAMA.
Most recommendations discourage the use of antibiotics in cases of STEC infection because this therapy is believed to increase the risk of hemolytic uremia syndrome (HUS), said Dr. Martin Nitschke of the University Hospital of Schleswig-Holstein, Lübeck, Germany, and his associates.
However, in the context of severe neurologic involvement or persistent HUS despite plasmapheresis therapy, treatment with eculizumab is advised. And because eculizumab enhances susceptibility to meningococcal infection, concomitant preventive therapy with azithromycin also is recommended.
Dr. Nitschke and his colleagues compared the rates of persistent STEC colonization in 22 patients who received azithromycin in this fashion vs. 43 control subjects who did not receive azithromycin. All 65 study subjects were hospitalized at their medical center and were followed for up to 45 days.
At 3 weeks, the rate of STEC carriage was 32% in the patients who received azithromycin, compared with 84% in the control group. At 4 weeks, the rates were 5% and 81%, respectively, the investigators said (JAMA 2012;307:1046-52).
By 5 weeks, all patients who received azithromycin were clear of STEC organisms and remained STEC negative thereafter. In contrast, 25 of 43 control subjects (58%) still carried STEC and continued to do so for more than 50 days; some control subjects remained STEC carriers for more than 160 days.
Patients who are colonized with STEC are considered infective, and their social and work activities are legally restricted by German health authorities, "posing a high psychological and socioeconmic burden," Dr. Nitschke and his associates said.
No cases of HUS developed in association with azithromycin therapy.
When the researchers noted the rapid clearance of STEC colonization with azithromycin and the lack of toxicity, they offered the antibiotic to an additional 15 patients who had persistent colonization. Seven of these patients achieved rapid decolonization without any clinical sequelae; four achieved later decolonization at 43-68 days, also without clinical sequelae; and four were lost to follow-up.
No patients in any of the study groups developed HUS or required rehospitalization during 6 more months of follow-up.
These findings indicate that azithromycin "might be considered a potentially effective and safe antibiotic for the treatment of enteroaggregative STEC infection," Dr. Nitschke and his colleagues said.
Their study was limited in that it was a single-center assessment with a "relatively small" sample size, and patients were not randomly assigned to treatments. The results, therefore, warrant confirmation in larger studies, they added.
This study received no industry funding. Dr. Nitschke reported ties to Novartis, Genzyme, and Roche, and his associates reported ties to numerous industry sources.
Among patients infected with Shiga toxin–producing Escherichia coli during the 2011 outbreak in Germany, azithromycin significantly decreased colonization with the organism, allowing patients to return to normal work and social activities instead of being quarantined, according to a report in the March 14 JAMA.
Most recommendations discourage the use of antibiotics in cases of STEC infection because this therapy is believed to increase the risk of hemolytic uremia syndrome (HUS), said Dr. Martin Nitschke of the University Hospital of Schleswig-Holstein, Lübeck, Germany, and his associates.
However, in the context of severe neurologic involvement or persistent HUS despite plasmapheresis therapy, treatment with eculizumab is advised. And because eculizumab enhances susceptibility to meningococcal infection, concomitant preventive therapy with azithromycin also is recommended.
Dr. Nitschke and his colleagues compared the rates of persistent STEC colonization in 22 patients who received azithromycin in this fashion vs. 43 control subjects who did not receive azithromycin. All 65 study subjects were hospitalized at their medical center and were followed for up to 45 days.
At 3 weeks, the rate of STEC carriage was 32% in the patients who received azithromycin, compared with 84% in the control group. At 4 weeks, the rates were 5% and 81%, respectively, the investigators said (JAMA 2012;307:1046-52).
By 5 weeks, all patients who received azithromycin were clear of STEC organisms and remained STEC negative thereafter. In contrast, 25 of 43 control subjects (58%) still carried STEC and continued to do so for more than 50 days; some control subjects remained STEC carriers for more than 160 days.
Patients who are colonized with STEC are considered infective, and their social and work activities are legally restricted by German health authorities, "posing a high psychological and socioeconmic burden," Dr. Nitschke and his associates said.
No cases of HUS developed in association with azithromycin therapy.
When the researchers noted the rapid clearance of STEC colonization with azithromycin and the lack of toxicity, they offered the antibiotic to an additional 15 patients who had persistent colonization. Seven of these patients achieved rapid decolonization without any clinical sequelae; four achieved later decolonization at 43-68 days, also without clinical sequelae; and four were lost to follow-up.
No patients in any of the study groups developed HUS or required rehospitalization during 6 more months of follow-up.
These findings indicate that azithromycin "might be considered a potentially effective and safe antibiotic for the treatment of enteroaggregative STEC infection," Dr. Nitschke and his colleagues said.
Their study was limited in that it was a single-center assessment with a "relatively small" sample size, and patients were not randomly assigned to treatments. The results, therefore, warrant confirmation in larger studies, they added.
This study received no industry funding. Dr. Nitschke reported ties to Novartis, Genzyme, and Roche, and his associates reported ties to numerous industry sources.
Among patients infected with Shiga toxin–producing Escherichia coli during the 2011 outbreak in Germany, azithromycin significantly decreased colonization with the organism, allowing patients to return to normal work and social activities instead of being quarantined, according to a report in the March 14 JAMA.
Most recommendations discourage the use of antibiotics in cases of STEC infection because this therapy is believed to increase the risk of hemolytic uremia syndrome (HUS), said Dr. Martin Nitschke of the University Hospital of Schleswig-Holstein, Lübeck, Germany, and his associates.
However, in the context of severe neurologic involvement or persistent HUS despite plasmapheresis therapy, treatment with eculizumab is advised. And because eculizumab enhances susceptibility to meningococcal infection, concomitant preventive therapy with azithromycin also is recommended.
Dr. Nitschke and his colleagues compared the rates of persistent STEC colonization in 22 patients who received azithromycin in this fashion vs. 43 control subjects who did not receive azithromycin. All 65 study subjects were hospitalized at their medical center and were followed for up to 45 days.
At 3 weeks, the rate of STEC carriage was 32% in the patients who received azithromycin, compared with 84% in the control group. At 4 weeks, the rates were 5% and 81%, respectively, the investigators said (JAMA 2012;307:1046-52).
By 5 weeks, all patients who received azithromycin were clear of STEC organisms and remained STEC negative thereafter. In contrast, 25 of 43 control subjects (58%) still carried STEC and continued to do so for more than 50 days; some control subjects remained STEC carriers for more than 160 days.
Patients who are colonized with STEC are considered infective, and their social and work activities are legally restricted by German health authorities, "posing a high psychological and socioeconmic burden," Dr. Nitschke and his associates said.
No cases of HUS developed in association with azithromycin therapy.
When the researchers noted the rapid clearance of STEC colonization with azithromycin and the lack of toxicity, they offered the antibiotic to an additional 15 patients who had persistent colonization. Seven of these patients achieved rapid decolonization without any clinical sequelae; four achieved later decolonization at 43-68 days, also without clinical sequelae; and four were lost to follow-up.
No patients in any of the study groups developed HUS or required rehospitalization during 6 more months of follow-up.
These findings indicate that azithromycin "might be considered a potentially effective and safe antibiotic for the treatment of enteroaggregative STEC infection," Dr. Nitschke and his colleagues said.
Their study was limited in that it was a single-center assessment with a "relatively small" sample size, and patients were not randomly assigned to treatments. The results, therefore, warrant confirmation in larger studies, they added.
This study received no industry funding. Dr. Nitschke reported ties to Novartis, Genzyme, and Roche, and his associates reported ties to numerous industry sources.
FROM JAMA
Major Finding: At 4 weeks, the rate of carriage of Shiga toxin–producing E. coli was 5% in patients who received azithromycin, compared with 81% in those who had not.
Data Source: Data are from a single-center study of the duration of bacterial shedding in 65 patients infected with STEC in a 2011 outbreak in Germany.
Disclosures: This study received no industry funding. Dr. Nitschke reported ties to Novartis, Genzyme, and Roche, and his associates reported ties to numerous industry sources.