User login
Citicoline Failed to Improve Function or Cognition in TBI Patients
Citicoline proved to be no better than placebo at improving function or cognition after traumatic brain injury in the first large randomized clinical trial to test it in this patient population.
Citicoline, an endogenous compound that is an intermediate in the biosynthesis of phosphatidylcholine from choline, is thought to have a range of neuroprotective properties and has been approved as a treatment for traumatic brain injury (TBI) in 59 countries other than the United States. But it is widely available in the United States as a nutraceutical used for a range of neurological disorders, Dr. Ross D. Zafonte of the department of physical medicine and rehabilitation at Harvard Medical School, Boston, and his associates said in the November 21 issue of JAMA.
Given these new findings, "the worldwide use of citicoline for TBI should now be questioned," the investigators wrote.
It’s likely that no single therapeutic agent will ever be sufficient to improve functional outcomes in TBI because there is such a variety of pathological mechanisms at work, including hematoma, edema, infarction, contusions, and inflammation, Dr. Robert L. Ruff and Dr. Ronald G. Riechers II of the neurology and polytrauma services and the department of neurology at the Cleveland (Ohio) Veterans Affairs Medical Center said in an editorial (JAMA 2012;308:2032-3). "The diverse and complex nature of the pathological mechanisms activated by TBI suggests that multimodal treatment interventions may be needed to improve recovery."
Dr. Zafonte and his colleagues conducted the Citicoline Brain Injury Treatment Trial (COBRIT), the first large, phase III, double-blind study to compare citicoline with placebo in the acute and postacute phases after TBI. They enrolled patients with a broad range of severity of injury who had presented to eight level I trauma centers across the country (JAMA 2012;308:1993-2000).
The 1,213 study subjects were aged 18-70 years, and the study population was ethnically and demographically diverse. As is typical for TBI patients, three-quarters of the study subjects were male, and more than half were younger than age 45 years.
These patients were randomly assigned to receive 90 days of either citicoline (607 patients) or identical-looking placebo (606) through an enteral route; those who could not swallow the regular oral tablets received the compound as crushed tablets mixed with water or saline and administered via a nasogastric or percutaneous endoscopic gastrostomy tube. The dosage was 2,000 mg per day.
Treatment commenced within 24 hours of sustaining the injury.
The primary outcome of the study was functional status and cognitive performance at 90 days, as measured by all nine components of the TBI Clinical Trials Network Core Battery. This included the Glasgow Outcome Scale-Extended (GOS-E) instrument.
COBRIT was halted early when an interim analysis indicated that further accrual would not change the main outcome: Patients given citicoline did not differ from those given placebo when tested using the combined Battery (odds ratio, 0.98 [95% confidence interval, 0.83-1.15]) or when tested on any of the individual elements of the Battery.
In particular, rates of improvement on the GOS-E were almost exactly the same: 35.4% among subjects given citicoline and 35.6% among those given placebo, the researchers reported.
These findings didn’t change when the data were adjusted to account for patients’ results on the Abbreviated Injury Score at baseline or when the subjects were categorized by the severity of their TBI. The results also did not change when the analysis was restricted to only subjects who took at least 75% of their assigned study medication.
There also were no significant differences between the groups in any measure at 180 days post injury.
A total of 73 subjects died during the study, with no significant difference in survival between the groups.
Similarly, there were no differences between the groups in the overall rates of adverse events or in the rates of serious adverse events.
This study was supported by the National Institute of Child Health and Human Development. Ferrer Grupo provided the citicoline and identical placebo used in the study. No financial conflicts of interest were reported.
Citicoline proved to be no better than placebo at improving function or cognition after traumatic brain injury in the first large randomized clinical trial to test it in this patient population.
Citicoline, an endogenous compound that is an intermediate in the biosynthesis of phosphatidylcholine from choline, is thought to have a range of neuroprotective properties and has been approved as a treatment for traumatic brain injury (TBI) in 59 countries other than the United States. But it is widely available in the United States as a nutraceutical used for a range of neurological disorders, Dr. Ross D. Zafonte of the department of physical medicine and rehabilitation at Harvard Medical School, Boston, and his associates said in the November 21 issue of JAMA.
Given these new findings, "the worldwide use of citicoline for TBI should now be questioned," the investigators wrote.
It’s likely that no single therapeutic agent will ever be sufficient to improve functional outcomes in TBI because there is such a variety of pathological mechanisms at work, including hematoma, edema, infarction, contusions, and inflammation, Dr. Robert L. Ruff and Dr. Ronald G. Riechers II of the neurology and polytrauma services and the department of neurology at the Cleveland (Ohio) Veterans Affairs Medical Center said in an editorial (JAMA 2012;308:2032-3). "The diverse and complex nature of the pathological mechanisms activated by TBI suggests that multimodal treatment interventions may be needed to improve recovery."
Dr. Zafonte and his colleagues conducted the Citicoline Brain Injury Treatment Trial (COBRIT), the first large, phase III, double-blind study to compare citicoline with placebo in the acute and postacute phases after TBI. They enrolled patients with a broad range of severity of injury who had presented to eight level I trauma centers across the country (JAMA 2012;308:1993-2000).
The 1,213 study subjects were aged 18-70 years, and the study population was ethnically and demographically diverse. As is typical for TBI patients, three-quarters of the study subjects were male, and more than half were younger than age 45 years.
These patients were randomly assigned to receive 90 days of either citicoline (607 patients) or identical-looking placebo (606) through an enteral route; those who could not swallow the regular oral tablets received the compound as crushed tablets mixed with water or saline and administered via a nasogastric or percutaneous endoscopic gastrostomy tube. The dosage was 2,000 mg per day.
Treatment commenced within 24 hours of sustaining the injury.
The primary outcome of the study was functional status and cognitive performance at 90 days, as measured by all nine components of the TBI Clinical Trials Network Core Battery. This included the Glasgow Outcome Scale-Extended (GOS-E) instrument.
COBRIT was halted early when an interim analysis indicated that further accrual would not change the main outcome: Patients given citicoline did not differ from those given placebo when tested using the combined Battery (odds ratio, 0.98 [95% confidence interval, 0.83-1.15]) or when tested on any of the individual elements of the Battery.
In particular, rates of improvement on the GOS-E were almost exactly the same: 35.4% among subjects given citicoline and 35.6% among those given placebo, the researchers reported.
These findings didn’t change when the data were adjusted to account for patients’ results on the Abbreviated Injury Score at baseline or when the subjects were categorized by the severity of their TBI. The results also did not change when the analysis was restricted to only subjects who took at least 75% of their assigned study medication.
There also were no significant differences between the groups in any measure at 180 days post injury.
A total of 73 subjects died during the study, with no significant difference in survival between the groups.
Similarly, there were no differences between the groups in the overall rates of adverse events or in the rates of serious adverse events.
This study was supported by the National Institute of Child Health and Human Development. Ferrer Grupo provided the citicoline and identical placebo used in the study. No financial conflicts of interest were reported.
Citicoline proved to be no better than placebo at improving function or cognition after traumatic brain injury in the first large randomized clinical trial to test it in this patient population.
Citicoline, an endogenous compound that is an intermediate in the biosynthesis of phosphatidylcholine from choline, is thought to have a range of neuroprotective properties and has been approved as a treatment for traumatic brain injury (TBI) in 59 countries other than the United States. But it is widely available in the United States as a nutraceutical used for a range of neurological disorders, Dr. Ross D. Zafonte of the department of physical medicine and rehabilitation at Harvard Medical School, Boston, and his associates said in the November 21 issue of JAMA.
Given these new findings, "the worldwide use of citicoline for TBI should now be questioned," the investigators wrote.
It’s likely that no single therapeutic agent will ever be sufficient to improve functional outcomes in TBI because there is such a variety of pathological mechanisms at work, including hematoma, edema, infarction, contusions, and inflammation, Dr. Robert L. Ruff and Dr. Ronald G. Riechers II of the neurology and polytrauma services and the department of neurology at the Cleveland (Ohio) Veterans Affairs Medical Center said in an editorial (JAMA 2012;308:2032-3). "The diverse and complex nature of the pathological mechanisms activated by TBI suggests that multimodal treatment interventions may be needed to improve recovery."
Dr. Zafonte and his colleagues conducted the Citicoline Brain Injury Treatment Trial (COBRIT), the first large, phase III, double-blind study to compare citicoline with placebo in the acute and postacute phases after TBI. They enrolled patients with a broad range of severity of injury who had presented to eight level I trauma centers across the country (JAMA 2012;308:1993-2000).
The 1,213 study subjects were aged 18-70 years, and the study population was ethnically and demographically diverse. As is typical for TBI patients, three-quarters of the study subjects were male, and more than half were younger than age 45 years.
These patients were randomly assigned to receive 90 days of either citicoline (607 patients) or identical-looking placebo (606) through an enteral route; those who could not swallow the regular oral tablets received the compound as crushed tablets mixed with water or saline and administered via a nasogastric or percutaneous endoscopic gastrostomy tube. The dosage was 2,000 mg per day.
Treatment commenced within 24 hours of sustaining the injury.
The primary outcome of the study was functional status and cognitive performance at 90 days, as measured by all nine components of the TBI Clinical Trials Network Core Battery. This included the Glasgow Outcome Scale-Extended (GOS-E) instrument.
COBRIT was halted early when an interim analysis indicated that further accrual would not change the main outcome: Patients given citicoline did not differ from those given placebo when tested using the combined Battery (odds ratio, 0.98 [95% confidence interval, 0.83-1.15]) or when tested on any of the individual elements of the Battery.
In particular, rates of improvement on the GOS-E were almost exactly the same: 35.4% among subjects given citicoline and 35.6% among those given placebo, the researchers reported.
These findings didn’t change when the data were adjusted to account for patients’ results on the Abbreviated Injury Score at baseline or when the subjects were categorized by the severity of their TBI. The results also did not change when the analysis was restricted to only subjects who took at least 75% of their assigned study medication.
There also were no significant differences between the groups in any measure at 180 days post injury.
A total of 73 subjects died during the study, with no significant difference in survival between the groups.
Similarly, there were no differences between the groups in the overall rates of adverse events or in the rates of serious adverse events.
This study was supported by the National Institute of Child Health and Human Development. Ferrer Grupo provided the citicoline and identical placebo used in the study. No financial conflicts of interest were reported.
FROM JAMA
Major Finding: Patients given citicoline did not differ from those given placebo when tested using the TBI Clinical Trials Network Core Battery (odds ratio, 0.98 [95% confidence interval, 0.83-1.15])
Data Source: This was a multicenter, randomized, double-blind, phase III clinical trial comparing enteral citicoline (2,000 mg/day) with matching placebo in 1,213 patients who had TBI and were assessed for cognitive and functional improvement at 90 days.
Disclosures: This study was supported by the National Institute of Child Health and Human Development. Ferrer Grupo provided the citicoline and identical placebo used in the study. No financial conflicts of interest were reported.
Build a Portal? They'll Still Come
Online access to medical records and clinicians prompted the patients in a large health maintenance organization to increase rather than decrease their in-person and telephone contacts for clinical services, according to a report in the Nov. 21 issue of JAMA.
Of particular concern was the finding that those who used electronic patient portals also showed increased utilization of hospitals and emergency services, not just office contacts, according to Dr. Ted E. Palen of the Institute for Health Research, Kaiser Permanente Colorado, and his associates.
Proponents of online patient access have argued that if patients could "look up ... their test results, request prescription refills, schedule appointments, and send secure emails to clinicians, then their use of clinical in-person and telephone calls [would] decrease." But the results of this retrospective study of more than 88,000 adult patients argue the opposite, the investigators reported.
Dr. Palen and his colleagues assessed the use of clinical services among 44,321 patients who signed up for online access and a matched control group of 44,321 patients who did not during a 5-year period before and after the introduction of the patient portals.
Contrary to the researchers’ expectations, portal users had significantly more in-person and telephone contacts with their clinicians in the year after the patient portal program was introduced than they had in the preceding year. The rate of office visits rose by 3.2 per HMO member per year, and the rate of telephone contacts rose by 4.2 per member per year.
Patients who used online access also had significantly more in-person and telephone contacts with their clinicians than did those in the control group, the researchers said (JAMA 2012;308:2012-9).
Even more surprising, the rates of after-hours clinic visits, emergency department visits, and hospitalizations also increased significantly for portal users compared with nonusers. Rates of after-hours visits were higher by 18.7/1,000 HMO members per year, rates of ED visits were higher by 11.2/1,000, and hospitalizations were higher by 19.9/1,000.
This pattern of health care utilization persisted when the data were analyzed according to patient age, with both younger patients (under age 50) and older patients (over age 50) increasing their rate of in-person and telephone contact with clinicians after beginning online access.
Patient portals are thought to be especially beneficial for patients with chronic illnesses; the assumption has been that such patients could monitor their condition and communicate with clinicians electronically, reducing their need for in-person visits.
However, in this study, patients with asthma, diabetes, and congestive heart failure who used the patient portal all increased their in-person and telephone contacts with their clinicians, compared with nonusers who had these chronic illnesses.
Dr. Palen and his associates noted that any large study can identify differences between groups that are statistically but not clinically significant. "However, the magnitude of differences in utilization that we identified appears to be clinically significant.
"For example, in a health system with 100,000 adult members with online access, if the rate of office visits increases by 0.5 visits per member per year, concomitant with an increase in telephone encounters by 0.3 per member per year, over the course of a year clinicians . . . would need to provide 50,000 more clinic visits and respond to 30,000 more telephone calls.
"If this also holds true for the small group practice, a primary care physician with 1,000 adult patients who has online access would need to provide for almost 10 more clinic visits per week and over 5.5 more telephone calls per week," they noted.
This study was not designed to examine why patients might change their patterns of contact with clinicians, but the investigators proposed a possible explanation. Perhaps patients who are already more likely to use health care services may selectively sign up for online access, "and then use this technology to gain even more frequent access rather than view it as a substitute for contact with the health care system," they said.
This study was supported by the Kaiser Permanente Colorado Regional Initiative Committee Fund. No financial conflicts of interest were reported.
These findings "will be sobering for patient portal enthusiasts," said Dr. David W. Bates and Dr. Susan Wells.
Because of its design, this study couldn’t answer a crucial question: Was the extra utilization needed or beneficial? It’s possible that patients who are more anxious or who somatize are more likely to both sign up for online access and to interact more often with the health care system. But it’s also possible that patients who adopt online access differ in other important ways from patients who do not, they said.
David W. Bates, M.D., and Susan Wells, M.D., are in the division of general internal medicine, Brigham and Women’s Hospital, Boston. Dr. Bates is also in the department of health care policy and management at the Harvard School of Public Health, Boston. They reported no financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Palen’s report (JAMA 2012;308:2034-6).
These findings "will be sobering for patient portal enthusiasts," said Dr. David W. Bates and Dr. Susan Wells.
Because of its design, this study couldn’t answer a crucial question: Was the extra utilization needed or beneficial? It’s possible that patients who are more anxious or who somatize are more likely to both sign up for online access and to interact more often with the health care system. But it’s also possible that patients who adopt online access differ in other important ways from patients who do not, they said.
David W. Bates, M.D., and Susan Wells, M.D., are in the division of general internal medicine, Brigham and Women’s Hospital, Boston. Dr. Bates is also in the department of health care policy and management at the Harvard School of Public Health, Boston. They reported no financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Palen’s report (JAMA 2012;308:2034-6).
These findings "will be sobering for patient portal enthusiasts," said Dr. David W. Bates and Dr. Susan Wells.
Because of its design, this study couldn’t answer a crucial question: Was the extra utilization needed or beneficial? It’s possible that patients who are more anxious or who somatize are more likely to both sign up for online access and to interact more often with the health care system. But it’s also possible that patients who adopt online access differ in other important ways from patients who do not, they said.
David W. Bates, M.D., and Susan Wells, M.D., are in the division of general internal medicine, Brigham and Women’s Hospital, Boston. Dr. Bates is also in the department of health care policy and management at the Harvard School of Public Health, Boston. They reported no financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Palen’s report (JAMA 2012;308:2034-6).
Online access to medical records and clinicians prompted the patients in a large health maintenance organization to increase rather than decrease their in-person and telephone contacts for clinical services, according to a report in the Nov. 21 issue of JAMA.
Of particular concern was the finding that those who used electronic patient portals also showed increased utilization of hospitals and emergency services, not just office contacts, according to Dr. Ted E. Palen of the Institute for Health Research, Kaiser Permanente Colorado, and his associates.
Proponents of online patient access have argued that if patients could "look up ... their test results, request prescription refills, schedule appointments, and send secure emails to clinicians, then their use of clinical in-person and telephone calls [would] decrease." But the results of this retrospective study of more than 88,000 adult patients argue the opposite, the investigators reported.
Dr. Palen and his colleagues assessed the use of clinical services among 44,321 patients who signed up for online access and a matched control group of 44,321 patients who did not during a 5-year period before and after the introduction of the patient portals.
Contrary to the researchers’ expectations, portal users had significantly more in-person and telephone contacts with their clinicians in the year after the patient portal program was introduced than they had in the preceding year. The rate of office visits rose by 3.2 per HMO member per year, and the rate of telephone contacts rose by 4.2 per member per year.
Patients who used online access also had significantly more in-person and telephone contacts with their clinicians than did those in the control group, the researchers said (JAMA 2012;308:2012-9).
Even more surprising, the rates of after-hours clinic visits, emergency department visits, and hospitalizations also increased significantly for portal users compared with nonusers. Rates of after-hours visits were higher by 18.7/1,000 HMO members per year, rates of ED visits were higher by 11.2/1,000, and hospitalizations were higher by 19.9/1,000.
This pattern of health care utilization persisted when the data were analyzed according to patient age, with both younger patients (under age 50) and older patients (over age 50) increasing their rate of in-person and telephone contact with clinicians after beginning online access.
Patient portals are thought to be especially beneficial for patients with chronic illnesses; the assumption has been that such patients could monitor their condition and communicate with clinicians electronically, reducing their need for in-person visits.
However, in this study, patients with asthma, diabetes, and congestive heart failure who used the patient portal all increased their in-person and telephone contacts with their clinicians, compared with nonusers who had these chronic illnesses.
Dr. Palen and his associates noted that any large study can identify differences between groups that are statistically but not clinically significant. "However, the magnitude of differences in utilization that we identified appears to be clinically significant.
"For example, in a health system with 100,000 adult members with online access, if the rate of office visits increases by 0.5 visits per member per year, concomitant with an increase in telephone encounters by 0.3 per member per year, over the course of a year clinicians . . . would need to provide 50,000 more clinic visits and respond to 30,000 more telephone calls.
"If this also holds true for the small group practice, a primary care physician with 1,000 adult patients who has online access would need to provide for almost 10 more clinic visits per week and over 5.5 more telephone calls per week," they noted.
This study was not designed to examine why patients might change their patterns of contact with clinicians, but the investigators proposed a possible explanation. Perhaps patients who are already more likely to use health care services may selectively sign up for online access, "and then use this technology to gain even more frequent access rather than view it as a substitute for contact with the health care system," they said.
This study was supported by the Kaiser Permanente Colorado Regional Initiative Committee Fund. No financial conflicts of interest were reported.
Online access to medical records and clinicians prompted the patients in a large health maintenance organization to increase rather than decrease their in-person and telephone contacts for clinical services, according to a report in the Nov. 21 issue of JAMA.
Of particular concern was the finding that those who used electronic patient portals also showed increased utilization of hospitals and emergency services, not just office contacts, according to Dr. Ted E. Palen of the Institute for Health Research, Kaiser Permanente Colorado, and his associates.
Proponents of online patient access have argued that if patients could "look up ... their test results, request prescription refills, schedule appointments, and send secure emails to clinicians, then their use of clinical in-person and telephone calls [would] decrease." But the results of this retrospective study of more than 88,000 adult patients argue the opposite, the investigators reported.
Dr. Palen and his colleagues assessed the use of clinical services among 44,321 patients who signed up for online access and a matched control group of 44,321 patients who did not during a 5-year period before and after the introduction of the patient portals.
Contrary to the researchers’ expectations, portal users had significantly more in-person and telephone contacts with their clinicians in the year after the patient portal program was introduced than they had in the preceding year. The rate of office visits rose by 3.2 per HMO member per year, and the rate of telephone contacts rose by 4.2 per member per year.
Patients who used online access also had significantly more in-person and telephone contacts with their clinicians than did those in the control group, the researchers said (JAMA 2012;308:2012-9).
Even more surprising, the rates of after-hours clinic visits, emergency department visits, and hospitalizations also increased significantly for portal users compared with nonusers. Rates of after-hours visits were higher by 18.7/1,000 HMO members per year, rates of ED visits were higher by 11.2/1,000, and hospitalizations were higher by 19.9/1,000.
This pattern of health care utilization persisted when the data were analyzed according to patient age, with both younger patients (under age 50) and older patients (over age 50) increasing their rate of in-person and telephone contact with clinicians after beginning online access.
Patient portals are thought to be especially beneficial for patients with chronic illnesses; the assumption has been that such patients could monitor their condition and communicate with clinicians electronically, reducing their need for in-person visits.
However, in this study, patients with asthma, diabetes, and congestive heart failure who used the patient portal all increased their in-person and telephone contacts with their clinicians, compared with nonusers who had these chronic illnesses.
Dr. Palen and his associates noted that any large study can identify differences between groups that are statistically but not clinically significant. "However, the magnitude of differences in utilization that we identified appears to be clinically significant.
"For example, in a health system with 100,000 adult members with online access, if the rate of office visits increases by 0.5 visits per member per year, concomitant with an increase in telephone encounters by 0.3 per member per year, over the course of a year clinicians . . . would need to provide 50,000 more clinic visits and respond to 30,000 more telephone calls.
"If this also holds true for the small group practice, a primary care physician with 1,000 adult patients who has online access would need to provide for almost 10 more clinic visits per week and over 5.5 more telephone calls per week," they noted.
This study was not designed to examine why patients might change their patterns of contact with clinicians, but the investigators proposed a possible explanation. Perhaps patients who are already more likely to use health care services may selectively sign up for online access, "and then use this technology to gain even more frequent access rather than view it as a substitute for contact with the health care system," they said.
This study was supported by the Kaiser Permanente Colorado Regional Initiative Committee Fund. No financial conflicts of interest were reported.
FROM JAMA
Major Finding: Patients with online access to clinicians and medical records had more in-person and telephone contacts with their clinicians than did matched control subjects with no online access.
Data Source: This was a retrospective cohort study of utilization by 88,642 adults over a 5-year period, half of whom used online access to medical records and clinicians.
Disclosures: This study was supported by the Kaiser Permanente Colorado Regional Initiative Committee Fund. No financial conflicts of interest were reported.
Guidelines Update Approaches to Stable Ischemic Heart Disease
Updated clinical guidelines on stable ischemic heart disease should help primary care physicians navigate the complex and growing array of options to diagnose and treat the condition formerly known as chronic stable angina.
Revised by six medical organizations and published online Nov. 19 in Annals of Internal Medicine, the evaluation and management guidelines incorporate the best available evidence concerning known and suspected stable ischemic heart disease (SIHD) in adults.
The target audience for the guidelines is primary care clinicians who treat these patients, said Dr. Amir Qaseem and his associates on the clinical guidelines committee of the American College of Physicians, Philadelphia.
In the clinical practice guideline for evaluating patients with known or suspected SIHD, there are 28 recommendations that address initial cardiac testing to establish the diagnosis; cardiac stress testing in diagnosed patients to assess their risk of myocardial infarction or death; and coronary angiography for risk assessment.
Cardiac stress testing "is indicated in most symptomatic patients suspected of having SIHD to establish the diagnosis, and it is indicated in most patients with both suspected and established SIHD to identify [those] at very high risk for death or MI who might have lesions where anatomic intervention could be beneficial," stated the evaluation guideline’s executive summary.
Also helpful in establishing the diagnosis is resting ECG. And in patients with clinically evident ventricular dysfunction, rest echocardiography and radionuclide imaging are helpful.
Overall, "Physicians should integrate the information from their clinical evaluation with patient preferences when making decisions about further testing. Two patients with the same pretest probability of SIHD may prefer different approaches because of variations in personal beliefs, economic considerations, or stage of life," said Dr. Qaseem, who is also at Jefferson Medical College, Philadelphia, and his associates.
This clinical practice guideline includes detailed algorithms for diagnosing patients who are suspected of having SIHD, and for assessing risk in those who are known to have SIHD, which may be particularly helpful for clinicians in primary care practice.
The second guideline, for management of SIHD, includes 48 specific recommendations that address patient education, management of known risk factors (dyslipidemia, hypertension, diabetes, low levels of physical activity, overweight/obesity, and smoking), medical therapy to prevent MI and death, medical therapy to relieve symptoms, revascularization to improve symptoms, revascularization to improve survival, and patient follow-up.
"The goals of managing patients with SIHD include reducing premature cardiovascular death and nonfatal MI while maintaining a level of activity, functional capacity, and quality of life that is satisfactory to the patient," according to the management guideline’s executive summary.
Of particular interest is the finding that there is not sufficient high-quality evidence to support using estrogen therapy in postmenopausal women as a means to limit their cardiovascular risk. Similarly, supplementation with vitamin C, vitamin E, or beta-carotene is of unproven benefit for SIHD and is not recommended. And elevated homocysteine levels should not be treated with folate and/or vitamins B6 and B12.
Patients who have SIHD should receive 75-162 mg per day of aspirin indefinitely, as long as there are no contradindications. They also should be followed at least annually so that symptoms and clinical function can be assessed, complications of SIHD such as heart failure and arrhythmias can be tracked, cardiac risk factors can be monitored, and adherence to lifestyle changes and medical therapy can be assessed.
This guideline also includes detailed algorithms for tailoring medical therapy to patients’ individual needs, for choosing revascularization to improve survival, and for choosing revascularization to improve symptoms.
Besides the ACP, the other organizations that jointly developed the guidelines are the American College of Cardiology Foundation, the American Heart Association, the American Association for Thoracic Surgery, the Preventive Cardiovascular Nurses Association, the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons.
The full clinical practice guidelines are available at www.annals.org and are being published simultaneously in the Journal of the American College of Cardiology.
The American College of Physicians financed the development of these clinical practice guidelines. Potential conflicts of interest can be accessed at acponlilne.org.
The revised guidelines provide some surprises, noted Dr. William G. Kussmaul III. Although the recommendation for treadmill ECG stress testing without imaging in women who have intermediate-probability symptoms "may not match common clinical practice," Dr. Kussmaul noted, "it probably should."
In addition, the recommendation for initial testing with stress imaging for all patients who’ve had prior revascularization "makes good clinical sense, because it tells us not only if the chest pain is ‘real,’ but also how large a region of myocardium is at risk," he explained.
Dr. Kussmaul found the absence of CT coronary calcium scoring from the diagnostic algorithm surprising, given that it involves less radiation than coronary computed tomography angiography and requires no contrast. In addition, "a coronary calcium score of zero drastically reduces the probability of having significant coronary disease," he noted.
While the evaluation and management guidelines provide sound guidance, Dr. Kussmaul said, "digesting the 28 recommendations for diagnosis and 48 for management is daunting. Fortunately, the guidelines also contain algorithm figures that are clear, usable, and comprehensive.
"My advice: Go straight to the algorithms," he said.
Dr. Kussmaul is at Hahnemann University Hospital, Philadelphia. These remarks were taken from his editorial accompanying the publication of the two clinical practice guidelines (Ann. Intern. Med. 2012;157:749-51).
The revised guidelines provide some surprises, noted Dr. William G. Kussmaul III. Although the recommendation for treadmill ECG stress testing without imaging in women who have intermediate-probability symptoms "may not match common clinical practice," Dr. Kussmaul noted, "it probably should."
In addition, the recommendation for initial testing with stress imaging for all patients who’ve had prior revascularization "makes good clinical sense, because it tells us not only if the chest pain is ‘real,’ but also how large a region of myocardium is at risk," he explained.
Dr. Kussmaul found the absence of CT coronary calcium scoring from the diagnostic algorithm surprising, given that it involves less radiation than coronary computed tomography angiography and requires no contrast. In addition, "a coronary calcium score of zero drastically reduces the probability of having significant coronary disease," he noted.
While the evaluation and management guidelines provide sound guidance, Dr. Kussmaul said, "digesting the 28 recommendations for diagnosis and 48 for management is daunting. Fortunately, the guidelines also contain algorithm figures that are clear, usable, and comprehensive.
"My advice: Go straight to the algorithms," he said.
Dr. Kussmaul is at Hahnemann University Hospital, Philadelphia. These remarks were taken from his editorial accompanying the publication of the two clinical practice guidelines (Ann. Intern. Med. 2012;157:749-51).
The revised guidelines provide some surprises, noted Dr. William G. Kussmaul III. Although the recommendation for treadmill ECG stress testing without imaging in women who have intermediate-probability symptoms "may not match common clinical practice," Dr. Kussmaul noted, "it probably should."
In addition, the recommendation for initial testing with stress imaging for all patients who’ve had prior revascularization "makes good clinical sense, because it tells us not only if the chest pain is ‘real,’ but also how large a region of myocardium is at risk," he explained.
Dr. Kussmaul found the absence of CT coronary calcium scoring from the diagnostic algorithm surprising, given that it involves less radiation than coronary computed tomography angiography and requires no contrast. In addition, "a coronary calcium score of zero drastically reduces the probability of having significant coronary disease," he noted.
While the evaluation and management guidelines provide sound guidance, Dr. Kussmaul said, "digesting the 28 recommendations for diagnosis and 48 for management is daunting. Fortunately, the guidelines also contain algorithm figures that are clear, usable, and comprehensive.
"My advice: Go straight to the algorithms," he said.
Dr. Kussmaul is at Hahnemann University Hospital, Philadelphia. These remarks were taken from his editorial accompanying the publication of the two clinical practice guidelines (Ann. Intern. Med. 2012;157:749-51).
Updated clinical guidelines on stable ischemic heart disease should help primary care physicians navigate the complex and growing array of options to diagnose and treat the condition formerly known as chronic stable angina.
Revised by six medical organizations and published online Nov. 19 in Annals of Internal Medicine, the evaluation and management guidelines incorporate the best available evidence concerning known and suspected stable ischemic heart disease (SIHD) in adults.
The target audience for the guidelines is primary care clinicians who treat these patients, said Dr. Amir Qaseem and his associates on the clinical guidelines committee of the American College of Physicians, Philadelphia.
In the clinical practice guideline for evaluating patients with known or suspected SIHD, there are 28 recommendations that address initial cardiac testing to establish the diagnosis; cardiac stress testing in diagnosed patients to assess their risk of myocardial infarction or death; and coronary angiography for risk assessment.
Cardiac stress testing "is indicated in most symptomatic patients suspected of having SIHD to establish the diagnosis, and it is indicated in most patients with both suspected and established SIHD to identify [those] at very high risk for death or MI who might have lesions where anatomic intervention could be beneficial," stated the evaluation guideline’s executive summary.
Also helpful in establishing the diagnosis is resting ECG. And in patients with clinically evident ventricular dysfunction, rest echocardiography and radionuclide imaging are helpful.
Overall, "Physicians should integrate the information from their clinical evaluation with patient preferences when making decisions about further testing. Two patients with the same pretest probability of SIHD may prefer different approaches because of variations in personal beliefs, economic considerations, or stage of life," said Dr. Qaseem, who is also at Jefferson Medical College, Philadelphia, and his associates.
This clinical practice guideline includes detailed algorithms for diagnosing patients who are suspected of having SIHD, and for assessing risk in those who are known to have SIHD, which may be particularly helpful for clinicians in primary care practice.
The second guideline, for management of SIHD, includes 48 specific recommendations that address patient education, management of known risk factors (dyslipidemia, hypertension, diabetes, low levels of physical activity, overweight/obesity, and smoking), medical therapy to prevent MI and death, medical therapy to relieve symptoms, revascularization to improve symptoms, revascularization to improve survival, and patient follow-up.
"The goals of managing patients with SIHD include reducing premature cardiovascular death and nonfatal MI while maintaining a level of activity, functional capacity, and quality of life that is satisfactory to the patient," according to the management guideline’s executive summary.
Of particular interest is the finding that there is not sufficient high-quality evidence to support using estrogen therapy in postmenopausal women as a means to limit their cardiovascular risk. Similarly, supplementation with vitamin C, vitamin E, or beta-carotene is of unproven benefit for SIHD and is not recommended. And elevated homocysteine levels should not be treated with folate and/or vitamins B6 and B12.
Patients who have SIHD should receive 75-162 mg per day of aspirin indefinitely, as long as there are no contradindications. They also should be followed at least annually so that symptoms and clinical function can be assessed, complications of SIHD such as heart failure and arrhythmias can be tracked, cardiac risk factors can be monitored, and adherence to lifestyle changes and medical therapy can be assessed.
This guideline also includes detailed algorithms for tailoring medical therapy to patients’ individual needs, for choosing revascularization to improve survival, and for choosing revascularization to improve symptoms.
Besides the ACP, the other organizations that jointly developed the guidelines are the American College of Cardiology Foundation, the American Heart Association, the American Association for Thoracic Surgery, the Preventive Cardiovascular Nurses Association, the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons.
The full clinical practice guidelines are available at www.annals.org and are being published simultaneously in the Journal of the American College of Cardiology.
The American College of Physicians financed the development of these clinical practice guidelines. Potential conflicts of interest can be accessed at acponlilne.org.
Updated clinical guidelines on stable ischemic heart disease should help primary care physicians navigate the complex and growing array of options to diagnose and treat the condition formerly known as chronic stable angina.
Revised by six medical organizations and published online Nov. 19 in Annals of Internal Medicine, the evaluation and management guidelines incorporate the best available evidence concerning known and suspected stable ischemic heart disease (SIHD) in adults.
The target audience for the guidelines is primary care clinicians who treat these patients, said Dr. Amir Qaseem and his associates on the clinical guidelines committee of the American College of Physicians, Philadelphia.
In the clinical practice guideline for evaluating patients with known or suspected SIHD, there are 28 recommendations that address initial cardiac testing to establish the diagnosis; cardiac stress testing in diagnosed patients to assess their risk of myocardial infarction or death; and coronary angiography for risk assessment.
Cardiac stress testing "is indicated in most symptomatic patients suspected of having SIHD to establish the diagnosis, and it is indicated in most patients with both suspected and established SIHD to identify [those] at very high risk for death or MI who might have lesions where anatomic intervention could be beneficial," stated the evaluation guideline’s executive summary.
Also helpful in establishing the diagnosis is resting ECG. And in patients with clinically evident ventricular dysfunction, rest echocardiography and radionuclide imaging are helpful.
Overall, "Physicians should integrate the information from their clinical evaluation with patient preferences when making decisions about further testing. Two patients with the same pretest probability of SIHD may prefer different approaches because of variations in personal beliefs, economic considerations, or stage of life," said Dr. Qaseem, who is also at Jefferson Medical College, Philadelphia, and his associates.
This clinical practice guideline includes detailed algorithms for diagnosing patients who are suspected of having SIHD, and for assessing risk in those who are known to have SIHD, which may be particularly helpful for clinicians in primary care practice.
The second guideline, for management of SIHD, includes 48 specific recommendations that address patient education, management of known risk factors (dyslipidemia, hypertension, diabetes, low levels of physical activity, overweight/obesity, and smoking), medical therapy to prevent MI and death, medical therapy to relieve symptoms, revascularization to improve symptoms, revascularization to improve survival, and patient follow-up.
"The goals of managing patients with SIHD include reducing premature cardiovascular death and nonfatal MI while maintaining a level of activity, functional capacity, and quality of life that is satisfactory to the patient," according to the management guideline’s executive summary.
Of particular interest is the finding that there is not sufficient high-quality evidence to support using estrogen therapy in postmenopausal women as a means to limit their cardiovascular risk. Similarly, supplementation with vitamin C, vitamin E, or beta-carotene is of unproven benefit for SIHD and is not recommended. And elevated homocysteine levels should not be treated with folate and/or vitamins B6 and B12.
Patients who have SIHD should receive 75-162 mg per day of aspirin indefinitely, as long as there are no contradindications. They also should be followed at least annually so that symptoms and clinical function can be assessed, complications of SIHD such as heart failure and arrhythmias can be tracked, cardiac risk factors can be monitored, and adherence to lifestyle changes and medical therapy can be assessed.
This guideline also includes detailed algorithms for tailoring medical therapy to patients’ individual needs, for choosing revascularization to improve survival, and for choosing revascularization to improve symptoms.
Besides the ACP, the other organizations that jointly developed the guidelines are the American College of Cardiology Foundation, the American Heart Association, the American Association for Thoracic Surgery, the Preventive Cardiovascular Nurses Association, the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons.
The full clinical practice guidelines are available at www.annals.org and are being published simultaneously in the Journal of the American College of Cardiology.
The American College of Physicians financed the development of these clinical practice guidelines. Potential conflicts of interest can be accessed at acponlilne.org.
FROM ANNALS OF INTERNAL MEDICINE
Major Finding: Clinical practice guidelines for the evaluation and management of stable ischemic heart disease have been updated and now include 28 recommendations for diagnosing the disorder and 48 for treating it.
Data Source: A comprehensive review and summary of the current medical literature concerning diagnosis and treatment of SIHD by clinicians in primary care practice.
Disclosures: The American College of Physicians financed the development of these clinical practice guidelines. Potential conflicts of interest can be accessed at acponlilne.org.
Survival After In-Hospital Cardiac Arrest Has Improved Substantially
Patient survival after in-hospital cardiac arrest improved substantially in recent years, according to a report from a large registry published online Nov. 15 in the New England Journal of Medicine.
Moreover, the rate of clinically significant neurologic disability among survivors has not risen correspondingly; in fact, it also decreased during the same time period, said Dr. Saket Girotra of the University of Iowa Hospitals and Clinics, Iowa City, and his associates.
These gains were found in a study of nearly 85,000 patients treated at 374 hospitals participating in the Get With the Guidelines (GWTG)-Resuscitation registry, a nationwide, hospital-based, quality-improvement registry documenting all cases of confirmed cardiac arrest at member hospitals in which patients received cardiopulmonary resuscitation. If the findings are extrapolated to the roughly 200,000 such events that occur annually in the United States, "we estimate that an additional 17,200 patients survived to hospital discharge in 2009 as compared with 2000. ... We also estimate that more than 13,000 cases of clinically significant neurologic disability were avoided," the investigators said.
They performed the study because numerous quality-improvement efforts to advance resuscitation care have been adopted in recent years, but no study has yet assessed whether they affected survival.
Dr. Girotra and his colleagues restricted their analysis to patients who had cardiac arrests during 2000-2009 while in an intensive care unit or inpatient ward, excluding cases that occurred in operating rooms, procedural suites, or emergency departments, because events in those settings "have distinct clinical circumstances and outcomes." The study sample comprised 84,625 patients.
The initial cardiac-arrest rhythm was asystole or pulseless electrical activity in 79% and ventricular fibrillation or pulseless ventricular tachycardia in 21%.
The primary outcome of the study was survival to hospital discharge. This was achieved by over 14,000 patients, or 17%.
"There was a significant trend toward increased survival during the study period for all study patients as well as for both rhythm groups. ... After adjustment for temporal trends in patient and hospital characteristics, overall survival increased from 13.7% in 2000 to 22.3% in 2009," the researchers said (N. Engl. J. Med. 2012;367:1012-20 [doi: 10.1056/NEJMoa1109148]).
This gain was seen across all subgroups of patients, regardless of the type of initial heart rhythm, age (younger or older than 65 years), race, or gender. It also remained robust in an analysis restricted to the 85 hospitals that had participated in the GWTG-Resuscitation registry for at least 8 years. This analysis was intended to adjust for the inclusion of centers that were particularly motivated to improve, as compared with other centers.
At the same time, rates of clinically significant neurologic disability among survivors decreased from 32.9% in 2000 to 28.1% in 2009. Rates of severe neurologic disability among survivors did not change significantly over time.
This study was not designed to ascertain the reasons that survival improved over time. However, it was notable that among patients whose initial cardiac rhythm was treatable by defibrillation, improved survival over time was not accompanied by shorter defibrillation times. "This observation suggests that factors other than rapid defibrillation may have accounted for the improvement in survival," Dr. Girotra and his associates said.
Such factors might include earlier recognition of cardiac arrest, leading to shorter response times; greater availability of trained personnel delivering better (i.e., uninterrupted) chest compressions; and improvements in postresuscitation care, such as therapeutic hypothermia and early cardiac catheterization.
More studies are needed "to better understand which specific factors are responsible for improvements in survival after cardiac arrest, so that survival gains can be consolidated and expanded to all hospitals," the investigators said.
This study was funded by the American Heart Association. Dr. Girotra’s associates reported ties to Prescription Solutions, United Health Care, Medtronic, Lumen, and St. Jude Medical.
Patient survival after in-hospital cardiac arrest improved substantially in recent years, according to a report from a large registry published online Nov. 15 in the New England Journal of Medicine.
Moreover, the rate of clinically significant neurologic disability among survivors has not risen correspondingly; in fact, it also decreased during the same time period, said Dr. Saket Girotra of the University of Iowa Hospitals and Clinics, Iowa City, and his associates.
These gains were found in a study of nearly 85,000 patients treated at 374 hospitals participating in the Get With the Guidelines (GWTG)-Resuscitation registry, a nationwide, hospital-based, quality-improvement registry documenting all cases of confirmed cardiac arrest at member hospitals in which patients received cardiopulmonary resuscitation. If the findings are extrapolated to the roughly 200,000 such events that occur annually in the United States, "we estimate that an additional 17,200 patients survived to hospital discharge in 2009 as compared with 2000. ... We also estimate that more than 13,000 cases of clinically significant neurologic disability were avoided," the investigators said.
They performed the study because numerous quality-improvement efforts to advance resuscitation care have been adopted in recent years, but no study has yet assessed whether they affected survival.
Dr. Girotra and his colleagues restricted their analysis to patients who had cardiac arrests during 2000-2009 while in an intensive care unit or inpatient ward, excluding cases that occurred in operating rooms, procedural suites, or emergency departments, because events in those settings "have distinct clinical circumstances and outcomes." The study sample comprised 84,625 patients.
The initial cardiac-arrest rhythm was asystole or pulseless electrical activity in 79% and ventricular fibrillation or pulseless ventricular tachycardia in 21%.
The primary outcome of the study was survival to hospital discharge. This was achieved by over 14,000 patients, or 17%.
"There was a significant trend toward increased survival during the study period for all study patients as well as for both rhythm groups. ... After adjustment for temporal trends in patient and hospital characteristics, overall survival increased from 13.7% in 2000 to 22.3% in 2009," the researchers said (N. Engl. J. Med. 2012;367:1012-20 [doi: 10.1056/NEJMoa1109148]).
This gain was seen across all subgroups of patients, regardless of the type of initial heart rhythm, age (younger or older than 65 years), race, or gender. It also remained robust in an analysis restricted to the 85 hospitals that had participated in the GWTG-Resuscitation registry for at least 8 years. This analysis was intended to adjust for the inclusion of centers that were particularly motivated to improve, as compared with other centers.
At the same time, rates of clinically significant neurologic disability among survivors decreased from 32.9% in 2000 to 28.1% in 2009. Rates of severe neurologic disability among survivors did not change significantly over time.
This study was not designed to ascertain the reasons that survival improved over time. However, it was notable that among patients whose initial cardiac rhythm was treatable by defibrillation, improved survival over time was not accompanied by shorter defibrillation times. "This observation suggests that factors other than rapid defibrillation may have accounted for the improvement in survival," Dr. Girotra and his associates said.
Such factors might include earlier recognition of cardiac arrest, leading to shorter response times; greater availability of trained personnel delivering better (i.e., uninterrupted) chest compressions; and improvements in postresuscitation care, such as therapeutic hypothermia and early cardiac catheterization.
More studies are needed "to better understand which specific factors are responsible for improvements in survival after cardiac arrest, so that survival gains can be consolidated and expanded to all hospitals," the investigators said.
This study was funded by the American Heart Association. Dr. Girotra’s associates reported ties to Prescription Solutions, United Health Care, Medtronic, Lumen, and St. Jude Medical.
Patient survival after in-hospital cardiac arrest improved substantially in recent years, according to a report from a large registry published online Nov. 15 in the New England Journal of Medicine.
Moreover, the rate of clinically significant neurologic disability among survivors has not risen correspondingly; in fact, it also decreased during the same time period, said Dr. Saket Girotra of the University of Iowa Hospitals and Clinics, Iowa City, and his associates.
These gains were found in a study of nearly 85,000 patients treated at 374 hospitals participating in the Get With the Guidelines (GWTG)-Resuscitation registry, a nationwide, hospital-based, quality-improvement registry documenting all cases of confirmed cardiac arrest at member hospitals in which patients received cardiopulmonary resuscitation. If the findings are extrapolated to the roughly 200,000 such events that occur annually in the United States, "we estimate that an additional 17,200 patients survived to hospital discharge in 2009 as compared with 2000. ... We also estimate that more than 13,000 cases of clinically significant neurologic disability were avoided," the investigators said.
They performed the study because numerous quality-improvement efforts to advance resuscitation care have been adopted in recent years, but no study has yet assessed whether they affected survival.
Dr. Girotra and his colleagues restricted their analysis to patients who had cardiac arrests during 2000-2009 while in an intensive care unit or inpatient ward, excluding cases that occurred in operating rooms, procedural suites, or emergency departments, because events in those settings "have distinct clinical circumstances and outcomes." The study sample comprised 84,625 patients.
The initial cardiac-arrest rhythm was asystole or pulseless electrical activity in 79% and ventricular fibrillation or pulseless ventricular tachycardia in 21%.
The primary outcome of the study was survival to hospital discharge. This was achieved by over 14,000 patients, or 17%.
"There was a significant trend toward increased survival during the study period for all study patients as well as for both rhythm groups. ... After adjustment for temporal trends in patient and hospital characteristics, overall survival increased from 13.7% in 2000 to 22.3% in 2009," the researchers said (N. Engl. J. Med. 2012;367:1012-20 [doi: 10.1056/NEJMoa1109148]).
This gain was seen across all subgroups of patients, regardless of the type of initial heart rhythm, age (younger or older than 65 years), race, or gender. It also remained robust in an analysis restricted to the 85 hospitals that had participated in the GWTG-Resuscitation registry for at least 8 years. This analysis was intended to adjust for the inclusion of centers that were particularly motivated to improve, as compared with other centers.
At the same time, rates of clinically significant neurologic disability among survivors decreased from 32.9% in 2000 to 28.1% in 2009. Rates of severe neurologic disability among survivors did not change significantly over time.
This study was not designed to ascertain the reasons that survival improved over time. However, it was notable that among patients whose initial cardiac rhythm was treatable by defibrillation, improved survival over time was not accompanied by shorter defibrillation times. "This observation suggests that factors other than rapid defibrillation may have accounted for the improvement in survival," Dr. Girotra and his associates said.
Such factors might include earlier recognition of cardiac arrest, leading to shorter response times; greater availability of trained personnel delivering better (i.e., uninterrupted) chest compressions; and improvements in postresuscitation care, such as therapeutic hypothermia and early cardiac catheterization.
More studies are needed "to better understand which specific factors are responsible for improvements in survival after cardiac arrest, so that survival gains can be consolidated and expanded to all hospitals," the investigators said.
This study was funded by the American Heart Association. Dr. Girotra’s associates reported ties to Prescription Solutions, United Health Care, Medtronic, Lumen, and St. Jude Medical.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: Overall survival after in-hospital cardiac arrest rose from 13.7% in 2000 to 22.3% in 2009, while the rate of clinically significant neurologic disability among survivors dropped from 32.9% to 28.1%.
Data Source: This was an analysis of data on CPR for in-hospital cardiac arrests occurring in 2000-2009 among 84,625 patients at 374 U.S. hospitals participating in the Get With the Guidelines–Resuscitation registry.
Disclosures: This study was funded by the American Heart Association. Dr. Girotra’s associates reported ties to Prescription Solutions, United Health Care, Medtronic, Lumen, and St. Jude Medical.
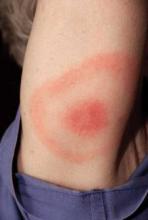
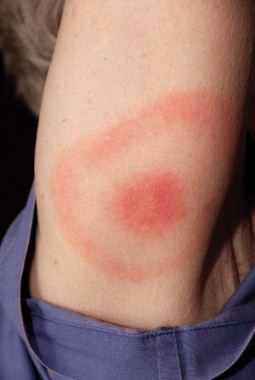
Reinfection, Not Relapse, Likely Spurred Lyme Recurrence
When erythema migrans recurs a year or more after standard courses of antibiotic therapy for Lyme disease, it most likely represents a reinfection from another tick bite, not a relapse of the first infection, according to a report published online Nov. 15 in the New England Journal of Medicine.
The limited clinical and epidemiologic evidence has suggested that such cases probably represent new infections – but some have argued that they could instead be relapses, citing animal experiments in which Borrelia burgdorferi persisted despite antibiotic treatment, said Dr. Robert B. Nadelman of the division of infectious diseases, New York Medical College, Valhalla, and his associates.
To settle the question, Dr. Nadelman and his colleagues analyzed the genotypes of the infecting organisms in a study of 17 adults who had a total of 22 paired episodes of erythema migrans between 1991 and 2011. The study subjects all were enrolled in prospective studies of Lyme disease at New York Medical College, and had provided skin and blood specimens for culture of B. burgdorferi before being treated with standard antibiotics. All skin lesions resolved with therapy.
All of the 9 men and 8 women were believed to have acquired the infections in the Lower Hudson Valley region of New York state. Their median age was 47 years (range, 27-80 years at the time of the first episode of erythema migrans).
In every case, the second episode of erythema migrans occurred from 1 year to 15 years after the first episode. The median duration of the interval between episodes was 4 years.
The investigators did not identify the same genotype in any of the 22 paired episodes in any patient. All the second episodes of erythema migrans were caused by a strain that was genotypically distinct from the organism that had caused the first episode.
Moreover, a second genotyping method confirmed infection by a different genotype of B. burgdorferi in all 22 paired episodes, the investigators said (N. Engl. J. Med. 2012;367:1883-90 [doi: 10.1056/NEJMoa1114362]).
These findings "provide further evidence of the success of antibiotics in eradication of B. burgdorferi from the skin of patients with erythema migrans in the United States," Dr. Nadelman and his associates said.
Other evidence also suggested that these patients experienced reinfections rather than relapses. In 20 (91%) of the cases, the recurrence developed during the summer months, paralleling the activity of the vector that likely was responsible.
"It is unlikely that a relapsed infection would have such a seasonal distribution when there is a delay of 1 year or more between episodes," the investigators noted.
The study was supported in part by the National Institutes of Health and the William and Sylvia Silberstein Foundation. Dr. Nadelman reported serving as a consultant to Decision Resources and Guidepoint Global, and his associates reported ties to Abbott, Baxter, and other companies.
"The issue of relapse versus reinfection has a broader context because of patient-advocacy groups that promote months or years of antibiotic therapy for ‘chronic Lyme disease,’ " said Dr. Allen C. Steere.
This has become a common designation for medically unexplained pain, neurocognitive symptoms, or fatigue, even when there is little or no evidence of previous B. burgdorferi infection. "Although B. burgdorferi infection may persist for years in untreated patients, the weight of evidence is strongly against persistent infection as the explanation for persistent symptoms in antibiotic-treated patients with Lyme disease," he said.
Dr. Steere is at Massachusetts General Hospital and Harvard Medical School, Boston. He reported ties to Merck. These remarks were taken from his editorial accompanying Dr. Nadelman’s report (N. Engl. J. Med. 2012 [doi: 10.1056/NEJMe1211361]).
"The issue of relapse versus reinfection has a broader context because of patient-advocacy groups that promote months or years of antibiotic therapy for ‘chronic Lyme disease,’ " said Dr. Allen C. Steere.
This has become a common designation for medically unexplained pain, neurocognitive symptoms, or fatigue, even when there is little or no evidence of previous B. burgdorferi infection. "Although B. burgdorferi infection may persist for years in untreated patients, the weight of evidence is strongly against persistent infection as the explanation for persistent symptoms in antibiotic-treated patients with Lyme disease," he said.
Dr. Steere is at Massachusetts General Hospital and Harvard Medical School, Boston. He reported ties to Merck. These remarks were taken from his editorial accompanying Dr. Nadelman’s report (N. Engl. J. Med. 2012 [doi: 10.1056/NEJMe1211361]).
"The issue of relapse versus reinfection has a broader context because of patient-advocacy groups that promote months or years of antibiotic therapy for ‘chronic Lyme disease,’ " said Dr. Allen C. Steere.
This has become a common designation for medically unexplained pain, neurocognitive symptoms, or fatigue, even when there is little or no evidence of previous B. burgdorferi infection. "Although B. burgdorferi infection may persist for years in untreated patients, the weight of evidence is strongly against persistent infection as the explanation for persistent symptoms in antibiotic-treated patients with Lyme disease," he said.
Dr. Steere is at Massachusetts General Hospital and Harvard Medical School, Boston. He reported ties to Merck. These remarks were taken from his editorial accompanying Dr. Nadelman’s report (N. Engl. J. Med. 2012 [doi: 10.1056/NEJMe1211361]).
When erythema migrans recurs a year or more after standard courses of antibiotic therapy for Lyme disease, it most likely represents a reinfection from another tick bite, not a relapse of the first infection, according to a report published online Nov. 15 in the New England Journal of Medicine.
The limited clinical and epidemiologic evidence has suggested that such cases probably represent new infections – but some have argued that they could instead be relapses, citing animal experiments in which Borrelia burgdorferi persisted despite antibiotic treatment, said Dr. Robert B. Nadelman of the division of infectious diseases, New York Medical College, Valhalla, and his associates.
To settle the question, Dr. Nadelman and his colleagues analyzed the genotypes of the infecting organisms in a study of 17 adults who had a total of 22 paired episodes of erythema migrans between 1991 and 2011. The study subjects all were enrolled in prospective studies of Lyme disease at New York Medical College, and had provided skin and blood specimens for culture of B. burgdorferi before being treated with standard antibiotics. All skin lesions resolved with therapy.
All of the 9 men and 8 women were believed to have acquired the infections in the Lower Hudson Valley region of New York state. Their median age was 47 years (range, 27-80 years at the time of the first episode of erythema migrans).
In every case, the second episode of erythema migrans occurred from 1 year to 15 years after the first episode. The median duration of the interval between episodes was 4 years.
The investigators did not identify the same genotype in any of the 22 paired episodes in any patient. All the second episodes of erythema migrans were caused by a strain that was genotypically distinct from the organism that had caused the first episode.
Moreover, a second genotyping method confirmed infection by a different genotype of B. burgdorferi in all 22 paired episodes, the investigators said (N. Engl. J. Med. 2012;367:1883-90 [doi: 10.1056/NEJMoa1114362]).
These findings "provide further evidence of the success of antibiotics in eradication of B. burgdorferi from the skin of patients with erythema migrans in the United States," Dr. Nadelman and his associates said.
Other evidence also suggested that these patients experienced reinfections rather than relapses. In 20 (91%) of the cases, the recurrence developed during the summer months, paralleling the activity of the vector that likely was responsible.
"It is unlikely that a relapsed infection would have such a seasonal distribution when there is a delay of 1 year or more between episodes," the investigators noted.
The study was supported in part by the National Institutes of Health and the William and Sylvia Silberstein Foundation. Dr. Nadelman reported serving as a consultant to Decision Resources and Guidepoint Global, and his associates reported ties to Abbott, Baxter, and other companies.
When erythema migrans recurs a year or more after standard courses of antibiotic therapy for Lyme disease, it most likely represents a reinfection from another tick bite, not a relapse of the first infection, according to a report published online Nov. 15 in the New England Journal of Medicine.
The limited clinical and epidemiologic evidence has suggested that such cases probably represent new infections – but some have argued that they could instead be relapses, citing animal experiments in which Borrelia burgdorferi persisted despite antibiotic treatment, said Dr. Robert B. Nadelman of the division of infectious diseases, New York Medical College, Valhalla, and his associates.
To settle the question, Dr. Nadelman and his colleagues analyzed the genotypes of the infecting organisms in a study of 17 adults who had a total of 22 paired episodes of erythema migrans between 1991 and 2011. The study subjects all were enrolled in prospective studies of Lyme disease at New York Medical College, and had provided skin and blood specimens for culture of B. burgdorferi before being treated with standard antibiotics. All skin lesions resolved with therapy.
All of the 9 men and 8 women were believed to have acquired the infections in the Lower Hudson Valley region of New York state. Their median age was 47 years (range, 27-80 years at the time of the first episode of erythema migrans).
In every case, the second episode of erythema migrans occurred from 1 year to 15 years after the first episode. The median duration of the interval between episodes was 4 years.
The investigators did not identify the same genotype in any of the 22 paired episodes in any patient. All the second episodes of erythema migrans were caused by a strain that was genotypically distinct from the organism that had caused the first episode.
Moreover, a second genotyping method confirmed infection by a different genotype of B. burgdorferi in all 22 paired episodes, the investigators said (N. Engl. J. Med. 2012;367:1883-90 [doi: 10.1056/NEJMoa1114362]).
These findings "provide further evidence of the success of antibiotics in eradication of B. burgdorferi from the skin of patients with erythema migrans in the United States," Dr. Nadelman and his associates said.
Other evidence also suggested that these patients experienced reinfections rather than relapses. In 20 (91%) of the cases, the recurrence developed during the summer months, paralleling the activity of the vector that likely was responsible.
"It is unlikely that a relapsed infection would have such a seasonal distribution when there is a delay of 1 year or more between episodes," the investigators noted.
The study was supported in part by the National Institutes of Health and the William and Sylvia Silberstein Foundation. Dr. Nadelman reported serving as a consultant to Decision Resources and Guidepoint Global, and his associates reported ties to Abbott, Baxter, and other companies.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: In all 22 paired episodes of erythema migrans, the second episode was caused by a strain of Borrelia burgdorferi that was genotypically different from the one that had caused the first episode.
Data Source: This was an analysis of the genotypes of B. burgdorferi strains obtained from skin and blood cultures of 17 adults who had experienced serial episodes of erythema migrans.
Disclosures: The study was supported in part by the National Institutes of Health and the William and Sylvia Silberstein Foundation. Dr. Nadelman reported serving as a consultant to Decision Resources and Guidepoint Global, and his associates reported ties to Abbott, Baxter, and other companies.
Antivirals Linked to Lower Risk of Liver Cancer Return
Using antiviral drugs after "curative" surgery for hepatitis B–related liver cancer may lower the risk of cancer recurrence, according to a Taiwanese study presented at the annual meeting of the American Association for the Study of Liver Diseases and simultaneously published online in JAMA.
The cumulative 6-year incidence of hepatocellular carcinoma (HCC) recurrence was significantly lower for patients who were treated with the nucleoside analogues lamivudine, entecavir, or telbivudine than it was for patients who weren’t given these agents.
Accordingly, overall 6-year mortality and disease-related mortality also were significantly lower, said Dr. Chun-Ying Wu of National Yang-Ming University, Taipei, and his associates (JAMA 2012;308:1906-13 [doi: 10.1001/2012.jama.11975]).
The investigators noted that their study could not definitively establish that these antiviral drugs caused the reductions in cancer recurrence and mortality, because no retrospective observational study can prove causality. But nucleoside analogues are known to be effective at suppressing hepatitis B virus (HBV) replication, ameliorating other HBV-related liver diseases, and reducing the risk of HCC in patients with chronic hepatitis or cirrhosis.
"Although it is generally accepted that HBV viral load plays an important role in HCC recurrence, studies regarding the effectiveness of nucleoside analogues in HCC recurrence have been very limited and have produced conflicting results," they said.
The investigators examined the issue using data from Taiwan’s National Health Insurance Research Database, which covers more than 99% of the country’s population of approximately 25 million. They identified all 4,569 inpatients with newly diagnosed HBV-related HCC who underwent "curative" liver resection during a 6-year period.
A total of 518 of these patients received lamivudine (159 patients), entecavir (292 patients), telbivudine (36 patients), or a combination of these antivirals (31 patients) for at least 90 days. The remaining 4,051 patients never received any nucleoside analogues.
The mean duration of antiviral therapy with these nucleoside analogues was 1.5 years. The patients were followed up until HCC recurrence, death, or the end of the 6-year study period.
After the data were adjusted to account for competing mortality, the cumulative rate of HCC recurrence was significantly lower for patients who received the nucleoside analogues (45.6%) than for those who did not (54.6%), the researchers said.
The unadjusted number needed to treat to achieve 1 less HCC recurrence within 6 years was 12. "This implies that use of nucleoside analogues in 12 HCC patients after liver resection is associated with 1 less HCC recurrence within 6 years," the investigators explained.
The overall cumulative 6-year mortality rate also was significantly lower (29.0%) in patients who received the nucleoside analogues than in those who did not (42.4%).
The unadjusted number needed to treat associated with 1 less death within 6 years was 8. "This implies that use of nucleoside analogues in 8 HCC patients after liver resection is associated with 1 less death within 6 years," Dr. Wu and his colleagues said.
Treatment with nucleoside analogues also was associated with improved disease-free survival.
These results were unchanged when the data were analyzed according to the use or nonuse of NSAIDs and the presence or absence of comorbid diabetes.
However, the use of nucleoside analogues was associated with a lower risk of HCC recurrence in patients who didn’t have concomitant cirrhosis but not in those who did have cirrhosis, they noted.
This study was supported by Taiwan’s National Health Research Institute. No financial conflicts of interest were reported.
The researchers show that nucleoside analogues may reduce short-term mortality after liver resection, but "given the long interval between cell damage, malignant transformation, and tumor development, it is unrealistic to expect that administration of antiviral therapy for 1-2 years can prevent HCC recurrence," said Dr. Anna S. F. Lok.
"Further studies with longer duration of treatment and better characterization of HBV replication status and liver disease are needed to determine the magnitude of benefit" from taking the drugs, she said.
Dr. Lok is in the division of gastroenterology and hepatology at the University of Michigan, Ann Arbor. She reported serving as an adviser for Arrowhead, Bayer, and other companies, and receiving research grants from Bristol-Myers Squibb, Gilead, and GlaxoSmithKline. These remarks were taken from her editorial accompanying Dr. Wu’s report (JAMA 2012;308:1922-24 [doi: 10.1001/jama.2012.12971]).
cancer recurrence,
The researchers show that nucleoside analogues may reduce short-term mortality after liver resection, but "given the long interval between cell damage, malignant transformation, and tumor development, it is unrealistic to expect that administration of antiviral therapy for 1-2 years can prevent HCC recurrence," said Dr. Anna S. F. Lok.
"Further studies with longer duration of treatment and better characterization of HBV replication status and liver disease are needed to determine the magnitude of benefit" from taking the drugs, she said.
Dr. Lok is in the division of gastroenterology and hepatology at the University of Michigan, Ann Arbor. She reported serving as an adviser for Arrowhead, Bayer, and other companies, and receiving research grants from Bristol-Myers Squibb, Gilead, and GlaxoSmithKline. These remarks were taken from her editorial accompanying Dr. Wu’s report (JAMA 2012;308:1922-24 [doi: 10.1001/jama.2012.12971]).
The researchers show that nucleoside analogues may reduce short-term mortality after liver resection, but "given the long interval between cell damage, malignant transformation, and tumor development, it is unrealistic to expect that administration of antiviral therapy for 1-2 years can prevent HCC recurrence," said Dr. Anna S. F. Lok.
"Further studies with longer duration of treatment and better characterization of HBV replication status and liver disease are needed to determine the magnitude of benefit" from taking the drugs, she said.
Dr. Lok is in the division of gastroenterology and hepatology at the University of Michigan, Ann Arbor. She reported serving as an adviser for Arrowhead, Bayer, and other companies, and receiving research grants from Bristol-Myers Squibb, Gilead, and GlaxoSmithKline. These remarks were taken from her editorial accompanying Dr. Wu’s report (JAMA 2012;308:1922-24 [doi: 10.1001/jama.2012.12971]).
Using antiviral drugs after "curative" surgery for hepatitis B–related liver cancer may lower the risk of cancer recurrence, according to a Taiwanese study presented at the annual meeting of the American Association for the Study of Liver Diseases and simultaneously published online in JAMA.
The cumulative 6-year incidence of hepatocellular carcinoma (HCC) recurrence was significantly lower for patients who were treated with the nucleoside analogues lamivudine, entecavir, or telbivudine than it was for patients who weren’t given these agents.
Accordingly, overall 6-year mortality and disease-related mortality also were significantly lower, said Dr. Chun-Ying Wu of National Yang-Ming University, Taipei, and his associates (JAMA 2012;308:1906-13 [doi: 10.1001/2012.jama.11975]).
The investigators noted that their study could not definitively establish that these antiviral drugs caused the reductions in cancer recurrence and mortality, because no retrospective observational study can prove causality. But nucleoside analogues are known to be effective at suppressing hepatitis B virus (HBV) replication, ameliorating other HBV-related liver diseases, and reducing the risk of HCC in patients with chronic hepatitis or cirrhosis.
"Although it is generally accepted that HBV viral load plays an important role in HCC recurrence, studies regarding the effectiveness of nucleoside analogues in HCC recurrence have been very limited and have produced conflicting results," they said.
The investigators examined the issue using data from Taiwan’s National Health Insurance Research Database, which covers more than 99% of the country’s population of approximately 25 million. They identified all 4,569 inpatients with newly diagnosed HBV-related HCC who underwent "curative" liver resection during a 6-year period.
A total of 518 of these patients received lamivudine (159 patients), entecavir (292 patients), telbivudine (36 patients), or a combination of these antivirals (31 patients) for at least 90 days. The remaining 4,051 patients never received any nucleoside analogues.
The mean duration of antiviral therapy with these nucleoside analogues was 1.5 years. The patients were followed up until HCC recurrence, death, or the end of the 6-year study period.
After the data were adjusted to account for competing mortality, the cumulative rate of HCC recurrence was significantly lower for patients who received the nucleoside analogues (45.6%) than for those who did not (54.6%), the researchers said.
The unadjusted number needed to treat to achieve 1 less HCC recurrence within 6 years was 12. "This implies that use of nucleoside analogues in 12 HCC patients after liver resection is associated with 1 less HCC recurrence within 6 years," the investigators explained.
The overall cumulative 6-year mortality rate also was significantly lower (29.0%) in patients who received the nucleoside analogues than in those who did not (42.4%).
The unadjusted number needed to treat associated with 1 less death within 6 years was 8. "This implies that use of nucleoside analogues in 8 HCC patients after liver resection is associated with 1 less death within 6 years," Dr. Wu and his colleagues said.
Treatment with nucleoside analogues also was associated with improved disease-free survival.
These results were unchanged when the data were analyzed according to the use or nonuse of NSAIDs and the presence or absence of comorbid diabetes.
However, the use of nucleoside analogues was associated with a lower risk of HCC recurrence in patients who didn’t have concomitant cirrhosis but not in those who did have cirrhosis, they noted.
This study was supported by Taiwan’s National Health Research Institute. No financial conflicts of interest were reported.
Using antiviral drugs after "curative" surgery for hepatitis B–related liver cancer may lower the risk of cancer recurrence, according to a Taiwanese study presented at the annual meeting of the American Association for the Study of Liver Diseases and simultaneously published online in JAMA.
The cumulative 6-year incidence of hepatocellular carcinoma (HCC) recurrence was significantly lower for patients who were treated with the nucleoside analogues lamivudine, entecavir, or telbivudine than it was for patients who weren’t given these agents.
Accordingly, overall 6-year mortality and disease-related mortality also were significantly lower, said Dr. Chun-Ying Wu of National Yang-Ming University, Taipei, and his associates (JAMA 2012;308:1906-13 [doi: 10.1001/2012.jama.11975]).
The investigators noted that their study could not definitively establish that these antiviral drugs caused the reductions in cancer recurrence and mortality, because no retrospective observational study can prove causality. But nucleoside analogues are known to be effective at suppressing hepatitis B virus (HBV) replication, ameliorating other HBV-related liver diseases, and reducing the risk of HCC in patients with chronic hepatitis or cirrhosis.
"Although it is generally accepted that HBV viral load plays an important role in HCC recurrence, studies regarding the effectiveness of nucleoside analogues in HCC recurrence have been very limited and have produced conflicting results," they said.
The investigators examined the issue using data from Taiwan’s National Health Insurance Research Database, which covers more than 99% of the country’s population of approximately 25 million. They identified all 4,569 inpatients with newly diagnosed HBV-related HCC who underwent "curative" liver resection during a 6-year period.
A total of 518 of these patients received lamivudine (159 patients), entecavir (292 patients), telbivudine (36 patients), or a combination of these antivirals (31 patients) for at least 90 days. The remaining 4,051 patients never received any nucleoside analogues.
The mean duration of antiviral therapy with these nucleoside analogues was 1.5 years. The patients were followed up until HCC recurrence, death, or the end of the 6-year study period.
After the data were adjusted to account for competing mortality, the cumulative rate of HCC recurrence was significantly lower for patients who received the nucleoside analogues (45.6%) than for those who did not (54.6%), the researchers said.
The unadjusted number needed to treat to achieve 1 less HCC recurrence within 6 years was 12. "This implies that use of nucleoside analogues in 12 HCC patients after liver resection is associated with 1 less HCC recurrence within 6 years," the investigators explained.
The overall cumulative 6-year mortality rate also was significantly lower (29.0%) in patients who received the nucleoside analogues than in those who did not (42.4%).
The unadjusted number needed to treat associated with 1 less death within 6 years was 8. "This implies that use of nucleoside analogues in 8 HCC patients after liver resection is associated with 1 less death within 6 years," Dr. Wu and his colleagues said.
Treatment with nucleoside analogues also was associated with improved disease-free survival.
These results were unchanged when the data were analyzed according to the use or nonuse of NSAIDs and the presence or absence of comorbid diabetes.
However, the use of nucleoside analogues was associated with a lower risk of HCC recurrence in patients who didn’t have concomitant cirrhosis but not in those who did have cirrhosis, they noted.
This study was supported by Taiwan’s National Health Research Institute. No financial conflicts of interest were reported.
cancer recurrence,
cancer recurrence,
FROM THE ANNUAL MEETING OF THE AMERICAN ASSOCIATION FOR THE STUDY OF LIVER DISEASES
Major Finding: The cumulative 6-year rate of hepatocellular carcinoma recurrence was 45.6% for patients taking nucleoside analogues and 54.6% for those not taking the drugs, and overall cumulative mortality was 29.0% and 42.4%, respectively.
Data Source: This was a retrospective observational study of patients with newly diagnosed hepatitis B virus–associated HCC who underwent liver resection and were followed for 6 years for the development of HCC recurrence, of whom 518 received antiviral therapy with nucleoside analogues and 4,051 did not.
Disclosures: This study was supported by Taiwan’s National Health Research Institute. No financial conflicts of interest were reported.
Excessive Acetaminophen Dosing Seen Among Inpatients
Four percent of hospitalized adolescents and adults at two academic tertiary care medical centers received supratherapeutic doses of acetaminophen during their stays, according to a report published online Nov. 12 in Archives of Internal Medicine.
"These were not isolated events but often were successive and overlapping." Nearly half of the exposed patients received 5 g or more of acetaminophen per day, and 40% received excessive dosing for 3 or more days, said Dr. Li Zhou of Partners HealthCare System, Wellesley, Mass., and her associates.
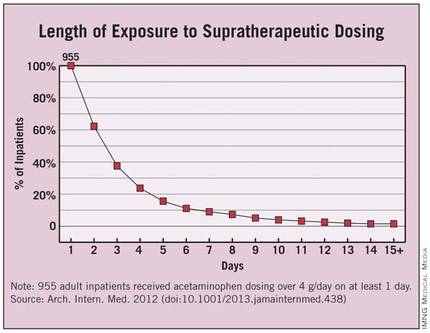
Such overmedicating puts patients at unnecessary risk for hepatotoxicity, acute liver failure, and even death. In this study, even inpatients at specific risk for liver damage didn’t escape excessive dosing with acetaminophen: 22% of the elderly and 18% of patients with existing chronic liver disease were given acetaminophen in amounts exceeding their recommended limit of 3 g/day, said Dr. Zhou, who is also at Brigham and Women’s Hospital and Harvard Medical School, Boston, and her colleagues.
The risk factors most strongly associated with supratherapeutic dosing were receiving recurring scheduled dosing (that is, standing orders) as opposed to as-needed or one-time-only administration, which carried a hazard ratio of 16.6; receiving multiple products containing acetaminophen, which carried a hazard ratio of 2.4 for each additional product; and receiving products containing 500 mg of acetaminophen, which carried an HR of 1.9.
The investigators used the hospitals’ electronic medication administration record systems to assess acetaminophen exposure in the inpatient setting – a topic that has received little attention from researchers before now, they wrote. The study population comprised the 14,411 inpatients aged 12 and older who received any acetaminophen during their stays in a 3-month period. That accounts for about 61% of the entire patient population hospitalized at the two centers during the study period.
The average age of the study subjects was 55 years (range, 12-110 years), and a little more than one-third were older than 65. Approximately 75 had chronic liver disease.
Overall, 955 patients were given acetaminophen in doses exceeding the 4-g/day limit. This represents 4% of the entire hospitalized population and 6.6% of the cohort of patients given any acetaminophen, the researchers said (Arch. Intern. Med. 2012 [doi:10.1001/2013.jamainternmed.438]).
Each patient who received supratherapeutic dosing had a mean of five such incidents during the course of a mean of 3 days. Instances of such overmedicating ranged as high as 48 separate occasions over the course of 30 days.
Approximately 40% of the 955 patients received supratherapeutic dosing for 3 or more days, and 4% received it for 10 or more days.
"Our findings show that despite policies and procedures to monitor and control patients’ acetaminophen exposure, the incidence of supratherapeutic acetaminophen dosing in hospitalized patients remains high," Dr. Zhou and her associates said.
Previous studies have shown that many physicians and nurses aren’t aware of the maximal recommended daily dosing of acetaminophen, and that many have some difficulty identifying which products contain the agent. Therefore, increased training is warranted "to help clinicians identify acetaminophen-containing products and monitor closely the daily dose of acetaminophen," the investigators said.
A reassessment of long-accepted dosing regimens may also be warranted, "especially the scheduled use of products containing 500 mg of acetaminophen," they added.
The study was funded by the Partners-Siemens Research Council. The investigators reported no relevant financial conflicts.
Dr. Li Zhou and her colleagues have identified a serious threat to patient safety and a cause for great concern, said Dr. Walter H. Ettinger.
"On an annualized basis, more than 3,800 patients at these two hospitals alone were put at unnecessary risk. If the estimates in this manuscript are generalizable, hundreds of thousands of patients nationwide may be receiving toxic doses of acetaminophen while in the hospital," he noted.
The study is important, "but it is incomplete and illustrates the obstacles we must overcome in using [health information technology] to drive performance. The authors identified a major flaw in the way in which a common drug is used in the hospital setting, and in a rapid-learning, high-performance health system, the results would be rapidly disseminated, together with a definitive solution for preventing excessive dosing in the future. The authors suggested educating health care providers about the risks of acetaminophen use and changing hospital policies to reduce risk factors for excessive dosing as a means of prevention. But excessive dosing of acetaminophen should be a ‘never event.’ The best way to prevent excessive dosing is to engineer a process and imbed it in the [electronic health record] such that it creates a hard stop that prevents ordering and administering more than 4 g/day of acetaminophen."
Dr. Ettinger is at the University of Massachusetts, Worcester, and at Accretive Health, Chicago, a health care technology and services firm. He reported no other relevant financial conflicts. These remarks were taken from his invited commentary accompanying Dr. Zhou’s report (Arch. Intern. Med. 2012 [doi:10.1001/2013.jamainternmed.607]).
Dr. Li Zhou and her colleagues have identified a serious threat to patient safety and a cause for great concern, said Dr. Walter H. Ettinger.
"On an annualized basis, more than 3,800 patients at these two hospitals alone were put at unnecessary risk. If the estimates in this manuscript are generalizable, hundreds of thousands of patients nationwide may be receiving toxic doses of acetaminophen while in the hospital," he noted.
The study is important, "but it is incomplete and illustrates the obstacles we must overcome in using [health information technology] to drive performance. The authors identified a major flaw in the way in which a common drug is used in the hospital setting, and in a rapid-learning, high-performance health system, the results would be rapidly disseminated, together with a definitive solution for preventing excessive dosing in the future. The authors suggested educating health care providers about the risks of acetaminophen use and changing hospital policies to reduce risk factors for excessive dosing as a means of prevention. But excessive dosing of acetaminophen should be a ‘never event.’ The best way to prevent excessive dosing is to engineer a process and imbed it in the [electronic health record] such that it creates a hard stop that prevents ordering and administering more than 4 g/day of acetaminophen."
Dr. Ettinger is at the University of Massachusetts, Worcester, and at Accretive Health, Chicago, a health care technology and services firm. He reported no other relevant financial conflicts. These remarks were taken from his invited commentary accompanying Dr. Zhou’s report (Arch. Intern. Med. 2012 [doi:10.1001/2013.jamainternmed.607]).
Dr. Li Zhou and her colleagues have identified a serious threat to patient safety and a cause for great concern, said Dr. Walter H. Ettinger.
"On an annualized basis, more than 3,800 patients at these two hospitals alone were put at unnecessary risk. If the estimates in this manuscript are generalizable, hundreds of thousands of patients nationwide may be receiving toxic doses of acetaminophen while in the hospital," he noted.
The study is important, "but it is incomplete and illustrates the obstacles we must overcome in using [health information technology] to drive performance. The authors identified a major flaw in the way in which a common drug is used in the hospital setting, and in a rapid-learning, high-performance health system, the results would be rapidly disseminated, together with a definitive solution for preventing excessive dosing in the future. The authors suggested educating health care providers about the risks of acetaminophen use and changing hospital policies to reduce risk factors for excessive dosing as a means of prevention. But excessive dosing of acetaminophen should be a ‘never event.’ The best way to prevent excessive dosing is to engineer a process and imbed it in the [electronic health record] such that it creates a hard stop that prevents ordering and administering more than 4 g/day of acetaminophen."
Dr. Ettinger is at the University of Massachusetts, Worcester, and at Accretive Health, Chicago, a health care technology and services firm. He reported no other relevant financial conflicts. These remarks were taken from his invited commentary accompanying Dr. Zhou’s report (Arch. Intern. Med. 2012 [doi:10.1001/2013.jamainternmed.607]).
Four percent of hospitalized adolescents and adults at two academic tertiary care medical centers received supratherapeutic doses of acetaminophen during their stays, according to a report published online Nov. 12 in Archives of Internal Medicine.
"These were not isolated events but often were successive and overlapping." Nearly half of the exposed patients received 5 g or more of acetaminophen per day, and 40% received excessive dosing for 3 or more days, said Dr. Li Zhou of Partners HealthCare System, Wellesley, Mass., and her associates.

Such overmedicating puts patients at unnecessary risk for hepatotoxicity, acute liver failure, and even death. In this study, even inpatients at specific risk for liver damage didn’t escape excessive dosing with acetaminophen: 22% of the elderly and 18% of patients with existing chronic liver disease were given acetaminophen in amounts exceeding their recommended limit of 3 g/day, said Dr. Zhou, who is also at Brigham and Women’s Hospital and Harvard Medical School, Boston, and her colleagues.
The risk factors most strongly associated with supratherapeutic dosing were receiving recurring scheduled dosing (that is, standing orders) as opposed to as-needed or one-time-only administration, which carried a hazard ratio of 16.6; receiving multiple products containing acetaminophen, which carried a hazard ratio of 2.4 for each additional product; and receiving products containing 500 mg of acetaminophen, which carried an HR of 1.9.
The investigators used the hospitals’ electronic medication administration record systems to assess acetaminophen exposure in the inpatient setting – a topic that has received little attention from researchers before now, they wrote. The study population comprised the 14,411 inpatients aged 12 and older who received any acetaminophen during their stays in a 3-month period. That accounts for about 61% of the entire patient population hospitalized at the two centers during the study period.
The average age of the study subjects was 55 years (range, 12-110 years), and a little more than one-third were older than 65. Approximately 75 had chronic liver disease.
Overall, 955 patients were given acetaminophen in doses exceeding the 4-g/day limit. This represents 4% of the entire hospitalized population and 6.6% of the cohort of patients given any acetaminophen, the researchers said (Arch. Intern. Med. 2012 [doi:10.1001/2013.jamainternmed.438]).
Each patient who received supratherapeutic dosing had a mean of five such incidents during the course of a mean of 3 days. Instances of such overmedicating ranged as high as 48 separate occasions over the course of 30 days.
Approximately 40% of the 955 patients received supratherapeutic dosing for 3 or more days, and 4% received it for 10 or more days.
"Our findings show that despite policies and procedures to monitor and control patients’ acetaminophen exposure, the incidence of supratherapeutic acetaminophen dosing in hospitalized patients remains high," Dr. Zhou and her associates said.
Previous studies have shown that many physicians and nurses aren’t aware of the maximal recommended daily dosing of acetaminophen, and that many have some difficulty identifying which products contain the agent. Therefore, increased training is warranted "to help clinicians identify acetaminophen-containing products and monitor closely the daily dose of acetaminophen," the investigators said.
A reassessment of long-accepted dosing regimens may also be warranted, "especially the scheduled use of products containing 500 mg of acetaminophen," they added.
The study was funded by the Partners-Siemens Research Council. The investigators reported no relevant financial conflicts.
Four percent of hospitalized adolescents and adults at two academic tertiary care medical centers received supratherapeutic doses of acetaminophen during their stays, according to a report published online Nov. 12 in Archives of Internal Medicine.
"These were not isolated events but often were successive and overlapping." Nearly half of the exposed patients received 5 g or more of acetaminophen per day, and 40% received excessive dosing for 3 or more days, said Dr. Li Zhou of Partners HealthCare System, Wellesley, Mass., and her associates.

Such overmedicating puts patients at unnecessary risk for hepatotoxicity, acute liver failure, and even death. In this study, even inpatients at specific risk for liver damage didn’t escape excessive dosing with acetaminophen: 22% of the elderly and 18% of patients with existing chronic liver disease were given acetaminophen in amounts exceeding their recommended limit of 3 g/day, said Dr. Zhou, who is also at Brigham and Women’s Hospital and Harvard Medical School, Boston, and her colleagues.
The risk factors most strongly associated with supratherapeutic dosing were receiving recurring scheduled dosing (that is, standing orders) as opposed to as-needed or one-time-only administration, which carried a hazard ratio of 16.6; receiving multiple products containing acetaminophen, which carried a hazard ratio of 2.4 for each additional product; and receiving products containing 500 mg of acetaminophen, which carried an HR of 1.9.
The investigators used the hospitals’ electronic medication administration record systems to assess acetaminophen exposure in the inpatient setting – a topic that has received little attention from researchers before now, they wrote. The study population comprised the 14,411 inpatients aged 12 and older who received any acetaminophen during their stays in a 3-month period. That accounts for about 61% of the entire patient population hospitalized at the two centers during the study period.
The average age of the study subjects was 55 years (range, 12-110 years), and a little more than one-third were older than 65. Approximately 75 had chronic liver disease.
Overall, 955 patients were given acetaminophen in doses exceeding the 4-g/day limit. This represents 4% of the entire hospitalized population and 6.6% of the cohort of patients given any acetaminophen, the researchers said (Arch. Intern. Med. 2012 [doi:10.1001/2013.jamainternmed.438]).
Each patient who received supratherapeutic dosing had a mean of five such incidents during the course of a mean of 3 days. Instances of such overmedicating ranged as high as 48 separate occasions over the course of 30 days.
Approximately 40% of the 955 patients received supratherapeutic dosing for 3 or more days, and 4% received it for 10 or more days.
"Our findings show that despite policies and procedures to monitor and control patients’ acetaminophen exposure, the incidence of supratherapeutic acetaminophen dosing in hospitalized patients remains high," Dr. Zhou and her associates said.
Previous studies have shown that many physicians and nurses aren’t aware of the maximal recommended daily dosing of acetaminophen, and that many have some difficulty identifying which products contain the agent. Therefore, increased training is warranted "to help clinicians identify acetaminophen-containing products and monitor closely the daily dose of acetaminophen," the investigators said.
A reassessment of long-accepted dosing regimens may also be warranted, "especially the scheduled use of products containing 500 mg of acetaminophen," they added.
The study was funded by the Partners-Siemens Research Council. The investigators reported no relevant financial conflicts.
FROM ARCHIVES OF INTERNAL MEDICINE
Major Finding: Four percent of all adolescent and adult inpatients at two hospitals received supratherapeutic doses of acetaminophen during their stays, including 22% of the elderly and 18% of patients with chronic liver disease.
Data Source: Data are based on acetaminophen exposure determined from electronic medication administration records for 14,411 inpatients aged 12 and older at two urban academic tertiary care hospitals during a 3-month period.
Disclosures: The study was funded by the Partners-Siemens Research Council. The authors reported no relevant financial conflicts.
Nonpayment Fails to Help Infection Rates
The 2008 Medicare policy to withhold payment for treating certain hospital-acquired infections failed to decrease infection rates in U.S. hospitals, according to a report published online in the New England Journal of Medicine.
In a study involving a total of 398 hospitals or medical systems across the country, implementing a Centers for Medicare and Medicaid Services policy of nonpayment for the treatment of preventable catheter-associated bloodstream infections and catheter-associated urinary tract infections appeared to have no impact at all on the acquisition of those infections, according to Dr. Ashish K. Jha of the department of health policy and management, Harvard School of Public Health, Boston, and his associates.
"As CMS continues to expand this policy to cover Medicaid through the Affordable Care Act, require public reporting of National Healthcare Safety Network [NHSN] data through the Hospital Compare website, and impose greater financial penalties on hospitals that perform poorly on these measures, careful evaluation is needed to determine when these programs work, when they have unintended consequences, and what might be done to improve patient outcomes," Dr. Jha noted.
Dr. Jha and his colleagues assessed data from the NHSN, a public health surveillance program for monitoring health care-associated infections across the country. A total of 1,166 nonfederal acute-care hospitals report their infection rates to this Centers for Disease Control and Prevention's sponsored network every month.
Dr. Jha and his colleagues assessed NHSN data on three different types of infection at 398 of those hospitals in 41 states. They examined central catheter-associated bloodstream and catheter-associated urinary tract infections because these are the two hospital-acquired infections for which CMS currently does not pay. They also looked at ventilator-associated pneumonia, which is not targeted by the CMS policy, as a control.
Rates of central catheter-associated bloodstream infections were already decreasing at the time the CMS policy was implemented, likely because the federal government, national organizations, and accrediting agencies had already focused attention on preventing these nosocomial infections. The rate of these infections was 4.8% per quarter before the policy was implemented and 4.7% afterward, a nonsignificant difference, the investigators said (N. Engl. J. Med. 2012 [doi:10.1056/NEJMsa1202419]).
This pattern also was seen with catheter-associated UTIs, in which there was a small, nonsignificant increase in the infection rate after implementation of the CMS policy. For the control condition of ventilator-associated pneumonia, the infection rate was 7.3% before implementation and 8.2% after implementation of the policy, also showing no significant impact on infection rates.
These findings were consistent across all hospital types, regardless of size, regional location, type of ownership, or teaching status.
To assess whether any benefit of the nonpayment policy may have been offset by strategies to lower infection rates, such as mandatory reporting, the researchers performed a separate analysis involving only the hospital units located in states that didn't have mandatory reporting. Again, no demonstrable effect on infection rates was seen.
To allow more time for hospitals to adapt to the policy change, the investigators performed a sensitivity analysis comparing infection rates 2 years after implementation with those before implementation. Again, they found no further decreases in the rates of any infections.
A possible explanation for these findings is that the amount of this financial disincentive was quite small. "Reductions in payment may have been equivalent to as little as 0.6% of Medicare revenue for the average hospital," Dr. Jha and his associates said.
"Greater financial penalties might induce a greater change in hospital responsiveness to the CMS policy."
The study results are particularly important given the increasing use of financial disincentives to improve the quality of health care. There is very little evidence that this strategy, or other pay-for-performance strategies, actually improves patient outcomes, the authors noted.
This study was supported by the Agency for Healthcare Research and Quality.
None of the authors reported having any financial conflicts of interest regarding this study.
The 2008 Medicare policy to withhold payment for treating certain hospital-acquired infections failed to decrease infection rates in U.S. hospitals, according to a report published online in the New England Journal of Medicine.
In a study involving a total of 398 hospitals or medical systems across the country, implementing a Centers for Medicare and Medicaid Services policy of nonpayment for the treatment of preventable catheter-associated bloodstream infections and catheter-associated urinary tract infections appeared to have no impact at all on the acquisition of those infections, according to Dr. Ashish K. Jha of the department of health policy and management, Harvard School of Public Health, Boston, and his associates.
"As CMS continues to expand this policy to cover Medicaid through the Affordable Care Act, require public reporting of National Healthcare Safety Network [NHSN] data through the Hospital Compare website, and impose greater financial penalties on hospitals that perform poorly on these measures, careful evaluation is needed to determine when these programs work, when they have unintended consequences, and what might be done to improve patient outcomes," Dr. Jha noted.
Dr. Jha and his colleagues assessed data from the NHSN, a public health surveillance program for monitoring health care-associated infections across the country. A total of 1,166 nonfederal acute-care hospitals report their infection rates to this Centers for Disease Control and Prevention's sponsored network every month.
Dr. Jha and his colleagues assessed NHSN data on three different types of infection at 398 of those hospitals in 41 states. They examined central catheter-associated bloodstream and catheter-associated urinary tract infections because these are the two hospital-acquired infections for which CMS currently does not pay. They also looked at ventilator-associated pneumonia, which is not targeted by the CMS policy, as a control.
Rates of central catheter-associated bloodstream infections were already decreasing at the time the CMS policy was implemented, likely because the federal government, national organizations, and accrediting agencies had already focused attention on preventing these nosocomial infections. The rate of these infections was 4.8% per quarter before the policy was implemented and 4.7% afterward, a nonsignificant difference, the investigators said (N. Engl. J. Med. 2012 [doi:10.1056/NEJMsa1202419]).
This pattern also was seen with catheter-associated UTIs, in which there was a small, nonsignificant increase in the infection rate after implementation of the CMS policy. For the control condition of ventilator-associated pneumonia, the infection rate was 7.3% before implementation and 8.2% after implementation of the policy, also showing no significant impact on infection rates.
These findings were consistent across all hospital types, regardless of size, regional location, type of ownership, or teaching status.
To assess whether any benefit of the nonpayment policy may have been offset by strategies to lower infection rates, such as mandatory reporting, the researchers performed a separate analysis involving only the hospital units located in states that didn't have mandatory reporting. Again, no demonstrable effect on infection rates was seen.
To allow more time for hospitals to adapt to the policy change, the investigators performed a sensitivity analysis comparing infection rates 2 years after implementation with those before implementation. Again, they found no further decreases in the rates of any infections.
A possible explanation for these findings is that the amount of this financial disincentive was quite small. "Reductions in payment may have been equivalent to as little as 0.6% of Medicare revenue for the average hospital," Dr. Jha and his associates said.
"Greater financial penalties might induce a greater change in hospital responsiveness to the CMS policy."
The study results are particularly important given the increasing use of financial disincentives to improve the quality of health care. There is very little evidence that this strategy, or other pay-for-performance strategies, actually improves patient outcomes, the authors noted.
This study was supported by the Agency for Healthcare Research and Quality.
None of the authors reported having any financial conflicts of interest regarding this study.
The 2008 Medicare policy to withhold payment for treating certain hospital-acquired infections failed to decrease infection rates in U.S. hospitals, according to a report published online in the New England Journal of Medicine.
In a study involving a total of 398 hospitals or medical systems across the country, implementing a Centers for Medicare and Medicaid Services policy of nonpayment for the treatment of preventable catheter-associated bloodstream infections and catheter-associated urinary tract infections appeared to have no impact at all on the acquisition of those infections, according to Dr. Ashish K. Jha of the department of health policy and management, Harvard School of Public Health, Boston, and his associates.
"As CMS continues to expand this policy to cover Medicaid through the Affordable Care Act, require public reporting of National Healthcare Safety Network [NHSN] data through the Hospital Compare website, and impose greater financial penalties on hospitals that perform poorly on these measures, careful evaluation is needed to determine when these programs work, when they have unintended consequences, and what might be done to improve patient outcomes," Dr. Jha noted.
Dr. Jha and his colleagues assessed data from the NHSN, a public health surveillance program for monitoring health care-associated infections across the country. A total of 1,166 nonfederal acute-care hospitals report their infection rates to this Centers for Disease Control and Prevention's sponsored network every month.
Dr. Jha and his colleagues assessed NHSN data on three different types of infection at 398 of those hospitals in 41 states. They examined central catheter-associated bloodstream and catheter-associated urinary tract infections because these are the two hospital-acquired infections for which CMS currently does not pay. They also looked at ventilator-associated pneumonia, which is not targeted by the CMS policy, as a control.
Rates of central catheter-associated bloodstream infections were already decreasing at the time the CMS policy was implemented, likely because the federal government, national organizations, and accrediting agencies had already focused attention on preventing these nosocomial infections. The rate of these infections was 4.8% per quarter before the policy was implemented and 4.7% afterward, a nonsignificant difference, the investigators said (N. Engl. J. Med. 2012 [doi:10.1056/NEJMsa1202419]).
This pattern also was seen with catheter-associated UTIs, in which there was a small, nonsignificant increase in the infection rate after implementation of the CMS policy. For the control condition of ventilator-associated pneumonia, the infection rate was 7.3% before implementation and 8.2% after implementation of the policy, also showing no significant impact on infection rates.
These findings were consistent across all hospital types, regardless of size, regional location, type of ownership, or teaching status.
To assess whether any benefit of the nonpayment policy may have been offset by strategies to lower infection rates, such as mandatory reporting, the researchers performed a separate analysis involving only the hospital units located in states that didn't have mandatory reporting. Again, no demonstrable effect on infection rates was seen.
To allow more time for hospitals to adapt to the policy change, the investigators performed a sensitivity analysis comparing infection rates 2 years after implementation with those before implementation. Again, they found no further decreases in the rates of any infections.
A possible explanation for these findings is that the amount of this financial disincentive was quite small. "Reductions in payment may have been equivalent to as little as 0.6% of Medicare revenue for the average hospital," Dr. Jha and his associates said.
"Greater financial penalties might induce a greater change in hospital responsiveness to the CMS policy."
The study results are particularly important given the increasing use of financial disincentives to improve the quality of health care. There is very little evidence that this strategy, or other pay-for-performance strategies, actually improves patient outcomes, the authors noted.
This study was supported by the Agency for Healthcare Research and Quality.
None of the authors reported having any financial conflicts of interest regarding this study.
Major Finding: The rate of central catheter–associated bloodstream infections was 4.8% before the nonpayment policy was implemented and 4.7% afterward, showing that the policy failed to decrease the infection rate.
Data Source: The data come from an analysis of trends in hospital-acquired infection rates before and after implementation of a federal policy to withhold payment for treating those infections, involving 398 hospitals in 41 states.
Disclosures: This study was supported by the Agency for Healthcare Research and Quality. No financial conflicts of interest were reported.
Use of PCI for MI Drops With Public Reporting
The use of percutaneous coronary intervention for acute myocardial infarction was found to be lower in three states that implemented public reporting of PCI outcomes than in seven nearby states without public reporting, according to a study of nearly 98,000 cases.
In addition, the use of PCI for acute MI declined in one state after public reporting of PCI outcomes was implemented there, said Dr. Karen E. Joynt of the departments of health policy and management at Harvard School of Public Health, Boston, and her associates.
Nevertheless, public reporting was not associated with any change in mortality for patients with acute MI in this study, they noted.
Collecting and publicly reporting patient outcomes is a tool intended to improve health care by motivating clinicians to improve their performance and allowing patients to choose the highest-quality hospitals. Critics of this strategy, however, say that it creates disincentives for physicians and hospitals to care for the sickest patients and may lead them to avoid offering lifesaving procedures such as PCI to the sickest or highest-risk patients.
To date, no national studies have examined whether public reporting of PCI outcomes has affected either the rates of PCI or the outcomes in patients with acute MI. Dr. Joynt and her colleagues did so using data from Medicare files.
They first performed a cross-sectional analysis of PCI rates in 30,745 patients who had a discharge diagnosis of acute MI in a single year, 2010. They compared the rates in three states that mandated public reporting -- Massachusetts, Pennsylvania, and New York -- against rates in seven nearby states that did not (Maine, Vermont, New Hampshire, Connecticut, Rhode Island, Maryland, and Delaware).
Patients in the public-reporting states were significantly less likely to receive PCI (37.7%) than were those in nonreporting states (42.7%). This trend was most pronounced in MI patients who had ST-elevation MI, cardiogenic shock, or cardiac arrest and was not seen in patients with non- ST-elevation MI, the investigators said (JAMA 2012;308:1460-8).
When the patients were categorized by age -- 65-74 years vs. 75 years and older -- the results were the same: Regardless of their age, patients in publicly reporting states were less likely to receive PCI than were those in nonreporting states.
The researchers then performed a longitudinal analysis of trends in PCI rates for 49,660 acute MI patients in reporting states and 48,142 in nonreporting states who were treated in 2002-2010. They focused on the experience in Massachusetts, tracking the rates before public reporting of PCI was implemented there (2002-2004) with the rates after it was implemented (2006-2010).
Before public reporting of PCI was implemented, the PCI rate in Massachusetts (40.6%) was comparable with that in nonreporting states (41.8%), but PCI rates in Massachusetts began to decline when reporting was implemented and by 2010 patients in Massachusetts were significantly less likely to receive PCI than were those in nonreporting states.
As in the cross-sectional study, PCI rates in Massachusetts declined the most among MI patients who had cardiogenic shock or cardiac arrest, and these findings did not change when patients were categorized by younger vs. older age.
Despite these declines in PCI rates, there was no significant difference in 30-day mortality between acute MI patients in reporting states (12.8%) and those in nonreporting states (12.1%). Some may find it reassuring that mandating public reporting did not increase patient mortality, but, conversely, it also did not reduce mortality, the researchers said.
The authors proposed two explanations why mortality was not affected by reductions in PCI rates. First, it may be that public reporting had its intended effect of focusing clinicians on performing PCI in only the most appropriate patients. Alternatively, it's possible that public reporting had an effect of pressuring physicians to avoid PCI in eligible but high-risk patients.
This study was funded by the NHLBI. No financial conflicts were reported.
There are three explanations for the observed lack of difference in mortality between reporting and nonreporting states, said Dr. Mauro Moscucci.
First, "futility assessments" in reporting states may have led to avoidance of PCI in patients who were less likely to benefit. Second, public reporting might have resulted in a drive toward improved quality of care and improved outcomes in patients receiving PCI, offsetting the adverse effect of not performing PCI in high-risk patients. Third, the optimal coding of comorbid condition required in risk adjustment may have been gamed through upcoding, thus leading to observed outcomes that are better than predicted. Better coding in public reporting states might have mitigated the adverse effect of denial of care after risk adjustment, he suggested.
This study highlights the possible unintended consequences of public reporting. The findings "may help spearhead a new focus on procedures that, while perceived [to be] appropriate based on current use criteria, might not result in added benefit in selected patients," he added.
Mauro Moscucci, M.D., is chief of the cardiovascular division at the University of Miami. He reported no relevant conflicts of interest. These remarks were taken from his editorial accompanying Dr. Joynt's report (JAMA 2012;308:148-9).
There are three explanations for the observed lack of difference in mortality between reporting and nonreporting states, said Dr. Mauro Moscucci.
First, "futility assessments" in reporting states may have led to avoidance of PCI in patients who were less likely to benefit. Second, public reporting might have resulted in a drive toward improved quality of care and improved outcomes in patients receiving PCI, offsetting the adverse effect of not performing PCI in high-risk patients. Third, the optimal coding of comorbid condition required in risk adjustment may have been gamed through upcoding, thus leading to observed outcomes that are better than predicted. Better coding in public reporting states might have mitigated the adverse effect of denial of care after risk adjustment, he suggested.
This study highlights the possible unintended consequences of public reporting. The findings "may help spearhead a new focus on procedures that, while perceived [to be] appropriate based on current use criteria, might not result in added benefit in selected patients," he added.
Mauro Moscucci, M.D., is chief of the cardiovascular division at the University of Miami. He reported no relevant conflicts of interest. These remarks were taken from his editorial accompanying Dr. Joynt's report (JAMA 2012;308:148-9).
There are three explanations for the observed lack of difference in mortality between reporting and nonreporting states, said Dr. Mauro Moscucci.
First, "futility assessments" in reporting states may have led to avoidance of PCI in patients who were less likely to benefit. Second, public reporting might have resulted in a drive toward improved quality of care and improved outcomes in patients receiving PCI, offsetting the adverse effect of not performing PCI in high-risk patients. Third, the optimal coding of comorbid condition required in risk adjustment may have been gamed through upcoding, thus leading to observed outcomes that are better than predicted. Better coding in public reporting states might have mitigated the adverse effect of denial of care after risk adjustment, he suggested.
This study highlights the possible unintended consequences of public reporting. The findings "may help spearhead a new focus on procedures that, while perceived [to be] appropriate based on current use criteria, might not result in added benefit in selected patients," he added.
Mauro Moscucci, M.D., is chief of the cardiovascular division at the University of Miami. He reported no relevant conflicts of interest. These remarks were taken from his editorial accompanying Dr. Joynt's report (JAMA 2012;308:148-9).
The use of percutaneous coronary intervention for acute myocardial infarction was found to be lower in three states that implemented public reporting of PCI outcomes than in seven nearby states without public reporting, according to a study of nearly 98,000 cases.
In addition, the use of PCI for acute MI declined in one state after public reporting of PCI outcomes was implemented there, said Dr. Karen E. Joynt of the departments of health policy and management at Harvard School of Public Health, Boston, and her associates.
Nevertheless, public reporting was not associated with any change in mortality for patients with acute MI in this study, they noted.
Collecting and publicly reporting patient outcomes is a tool intended to improve health care by motivating clinicians to improve their performance and allowing patients to choose the highest-quality hospitals. Critics of this strategy, however, say that it creates disincentives for physicians and hospitals to care for the sickest patients and may lead them to avoid offering lifesaving procedures such as PCI to the sickest or highest-risk patients.
To date, no national studies have examined whether public reporting of PCI outcomes has affected either the rates of PCI or the outcomes in patients with acute MI. Dr. Joynt and her colleagues did so using data from Medicare files.
They first performed a cross-sectional analysis of PCI rates in 30,745 patients who had a discharge diagnosis of acute MI in a single year, 2010. They compared the rates in three states that mandated public reporting -- Massachusetts, Pennsylvania, and New York -- against rates in seven nearby states that did not (Maine, Vermont, New Hampshire, Connecticut, Rhode Island, Maryland, and Delaware).
Patients in the public-reporting states were significantly less likely to receive PCI (37.7%) than were those in nonreporting states (42.7%). This trend was most pronounced in MI patients who had ST-elevation MI, cardiogenic shock, or cardiac arrest and was not seen in patients with non- ST-elevation MI, the investigators said (JAMA 2012;308:1460-8).
When the patients were categorized by age -- 65-74 years vs. 75 years and older -- the results were the same: Regardless of their age, patients in publicly reporting states were less likely to receive PCI than were those in nonreporting states.
The researchers then performed a longitudinal analysis of trends in PCI rates for 49,660 acute MI patients in reporting states and 48,142 in nonreporting states who were treated in 2002-2010. They focused on the experience in Massachusetts, tracking the rates before public reporting of PCI was implemented there (2002-2004) with the rates after it was implemented (2006-2010).
Before public reporting of PCI was implemented, the PCI rate in Massachusetts (40.6%) was comparable with that in nonreporting states (41.8%), but PCI rates in Massachusetts began to decline when reporting was implemented and by 2010 patients in Massachusetts were significantly less likely to receive PCI than were those in nonreporting states.
As in the cross-sectional study, PCI rates in Massachusetts declined the most among MI patients who had cardiogenic shock or cardiac arrest, and these findings did not change when patients were categorized by younger vs. older age.
Despite these declines in PCI rates, there was no significant difference in 30-day mortality between acute MI patients in reporting states (12.8%) and those in nonreporting states (12.1%). Some may find it reassuring that mandating public reporting did not increase patient mortality, but, conversely, it also did not reduce mortality, the researchers said.
The authors proposed two explanations why mortality was not affected by reductions in PCI rates. First, it may be that public reporting had its intended effect of focusing clinicians on performing PCI in only the most appropriate patients. Alternatively, it's possible that public reporting had an effect of pressuring physicians to avoid PCI in eligible but high-risk patients.
This study was funded by the NHLBI. No financial conflicts were reported.
The use of percutaneous coronary intervention for acute myocardial infarction was found to be lower in three states that implemented public reporting of PCI outcomes than in seven nearby states without public reporting, according to a study of nearly 98,000 cases.
In addition, the use of PCI for acute MI declined in one state after public reporting of PCI outcomes was implemented there, said Dr. Karen E. Joynt of the departments of health policy and management at Harvard School of Public Health, Boston, and her associates.
Nevertheless, public reporting was not associated with any change in mortality for patients with acute MI in this study, they noted.
Collecting and publicly reporting patient outcomes is a tool intended to improve health care by motivating clinicians to improve their performance and allowing patients to choose the highest-quality hospitals. Critics of this strategy, however, say that it creates disincentives for physicians and hospitals to care for the sickest patients and may lead them to avoid offering lifesaving procedures such as PCI to the sickest or highest-risk patients.
To date, no national studies have examined whether public reporting of PCI outcomes has affected either the rates of PCI or the outcomes in patients with acute MI. Dr. Joynt and her colleagues did so using data from Medicare files.
They first performed a cross-sectional analysis of PCI rates in 30,745 patients who had a discharge diagnosis of acute MI in a single year, 2010. They compared the rates in three states that mandated public reporting -- Massachusetts, Pennsylvania, and New York -- against rates in seven nearby states that did not (Maine, Vermont, New Hampshire, Connecticut, Rhode Island, Maryland, and Delaware).
Patients in the public-reporting states were significantly less likely to receive PCI (37.7%) than were those in nonreporting states (42.7%). This trend was most pronounced in MI patients who had ST-elevation MI, cardiogenic shock, or cardiac arrest and was not seen in patients with non- ST-elevation MI, the investigators said (JAMA 2012;308:1460-8).
When the patients were categorized by age -- 65-74 years vs. 75 years and older -- the results were the same: Regardless of their age, patients in publicly reporting states were less likely to receive PCI than were those in nonreporting states.
The researchers then performed a longitudinal analysis of trends in PCI rates for 49,660 acute MI patients in reporting states and 48,142 in nonreporting states who were treated in 2002-2010. They focused on the experience in Massachusetts, tracking the rates before public reporting of PCI was implemented there (2002-2004) with the rates after it was implemented (2006-2010).
Before public reporting of PCI was implemented, the PCI rate in Massachusetts (40.6%) was comparable with that in nonreporting states (41.8%), but PCI rates in Massachusetts began to decline when reporting was implemented and by 2010 patients in Massachusetts were significantly less likely to receive PCI than were those in nonreporting states.
As in the cross-sectional study, PCI rates in Massachusetts declined the most among MI patients who had cardiogenic shock or cardiac arrest, and these findings did not change when patients were categorized by younger vs. older age.
Despite these declines in PCI rates, there was no significant difference in 30-day mortality between acute MI patients in reporting states (12.8%) and those in nonreporting states (12.1%). Some may find it reassuring that mandating public reporting did not increase patient mortality, but, conversely, it also did not reduce mortality, the researchers said.
The authors proposed two explanations why mortality was not affected by reductions in PCI rates. First, it may be that public reporting had its intended effect of focusing clinicians on performing PCI in only the most appropriate patients. Alternatively, it's possible that public reporting had an effect of pressuring physicians to avoid PCI in eligible but high-risk patients.
This study was funded by the NHLBI. No financial conflicts were reported.
Major Finding: Patients with acute MI were less likely to receive PCI in states with mandatory public reporting of PCI outcomes (37.7%) than in those without public reporting (42.7%).
Data Source: A longitudinal analysis included PCI rates and outcomes for 49,660 acute MI patients treated in reporting states and 48,142 treated in nonreporting states in 2002-2010; there also was a cross-sectional analysis of 30,745 patients treated in 2010 only.
Disclosures: This study was supported by the National Heart, Lung, and Blood Institute. No financial conflicts of interest were reported.
Use of PCI for MI Drops With Public Reporting
The use of percutaneous coronary intervention for acute myocardial infarction was found to be lower in three states that implemented public reporting of PCI outcomes than in seven nearby states without public reporting, according to a study of nearly 98,000 cases.
In addition, the use of PCI for acute MI declined in one state after public reporting of PCI outcomes was implemented there, said Dr. Karen E. Joynt of the departments of health policy and management at Harvard School of Public Health, Boston, and her associates.
Nevertheless, public reporting was not associated with any change in mortality for patients with acute MI in this study, they noted.
Collecting and publicly reporting patient outcomes is a tool intended to improve health care by motivating clinicians to improve their performance and allowing patients to choose the highest-quality hospitals. Critics of this strategy, however, say that it creates disincentives for physicians and hospitals to care for the sickest patients and may lead them to avoid offering lifesaving procedures such as PCI to the sickest or highest-risk patients.
To date, no national studies have examined whether public reporting of PCI outcomes has affected either the rates of PCI or the outcomes in patients with acute MI. Dr. Joynt and her colleagues did so using data from Medicare files.
They first performed a cross-sectional analysis of PCI rates in 30,745 patients who had a discharge diagnosis of acute MI in a single year, 2010. They compared the rates in three states that mandated public reporting -- Massachusetts, Pennsylvania, and New York -- against rates in seven nearby states that did not (Maine, Vermont, New Hampshire, Connecticut, Rhode Island, Maryland, and Delaware).
Patients in the public-reporting states were significantly less likely to receive PCI (37.7%) than were those in nonreporting states (42.7%). This trend was most pronounced in MI patients who had ST-elevation MI, cardiogenic shock, or cardiac arrest and was not seen in patients with non- ST-elevation MI, the investigators said (JAMA 2012;308:1460-8).
When the patients were categorized by age -- 65-74 years vs. 75 years and older -- the results were the same: Regardless of their age, patients in publicly reporting states were less likely to receive PCI than were those in nonreporting states.
The researchers then performed a longitudinal analysis of trends in PCI rates for 49,660 acute MI patients in reporting states and 48,142 in nonreporting states who were treated in 2002-2010. They focused on the experience in Massachusetts, tracking the rates before public reporting of PCI was implemented there (2002-2004) with the rates after it was implemented (2006-2010).
Before public reporting of PCI was implemented, the PCI rate in Massachusetts (40.6%) was comparable with that in nonreporting states (41.8%), but PCI rates in Massachusetts began to decline when reporting was implemented and by 2010 patients in Massachusetts were significantly less likely to receive PCI than were those in nonreporting states.
As in the cross-sectional study, PCI rates in Massachusetts declined the most among MI patients who had cardiogenic shock or cardiac arrest, and these findings did not change when patients were categorized by younger vs. older age.
Despite these declines in PCI rates, there was no significant difference in 30-day mortality between acute MI patients in reporting states (12.8%) and those in nonreporting states (12.1%). Some may find it reassuring that mandating public reporting did not increase patient mortality, but, conversely, it also did not reduce mortality, the researchers said.
The authors proposed two explanations why mortality was not affected by reductions in PCI rates. First, it may be that public reporting had its intended effect of focusing clinicians on performing PCI in only the most appropriate patients. Alternatively, it's possible that public reporting had an effect of pressuring physicians to avoid PCI in eligible but high-risk patients.
This study was funded by the NHLBI. No financial conflicts were reported.
There are three explanations for the observed lack of difference in mortality between reporting and nonreporting states, said Dr. Mauro Moscucci.
First, "futility assessments" in reporting states may have led to avoidance of PCI in patients who were less likely to benefit. Second, public reporting might have resulted in a drive toward improved quality of care and improved outcomes in patients receiving PCI, offsetting the adverse effect of not performing PCI in high-risk patients. Third, the optimal coding of comorbid condition required in risk adjustment may have been gamed through upcoding, thus leading to observed outcomes that are better than predicted. Better coding in public reporting states might have mitigated the adverse effect of denial of care after risk adjustment, he suggested.
This study highlights the possible unintended consequences of public reporting. The findings "may help spearhead a new focus on procedures that, while perceived [to be] appropriate based on current use criteria, might not result in added benefit in selected patients," he added.
Mauro Moscucci, M.D., is chief of the cardiovascular division at the University of Miami. He reported no relevant conflicts of interest. These remarks were taken from his editorial accompanying Dr. Joynt's report (JAMA 2012;308:148-9).
There are three explanations for the observed lack of difference in mortality between reporting and nonreporting states, said Dr. Mauro Moscucci.
First, "futility assessments" in reporting states may have led to avoidance of PCI in patients who were less likely to benefit. Second, public reporting might have resulted in a drive toward improved quality of care and improved outcomes in patients receiving PCI, offsetting the adverse effect of not performing PCI in high-risk patients. Third, the optimal coding of comorbid condition required in risk adjustment may have been gamed through upcoding, thus leading to observed outcomes that are better than predicted. Better coding in public reporting states might have mitigated the adverse effect of denial of care after risk adjustment, he suggested.
This study highlights the possible unintended consequences of public reporting. The findings "may help spearhead a new focus on procedures that, while perceived [to be] appropriate based on current use criteria, might not result in added benefit in selected patients," he added.
Mauro Moscucci, M.D., is chief of the cardiovascular division at the University of Miami. He reported no relevant conflicts of interest. These remarks were taken from his editorial accompanying Dr. Joynt's report (JAMA 2012;308:148-9).
There are three explanations for the observed lack of difference in mortality between reporting and nonreporting states, said Dr. Mauro Moscucci.
First, "futility assessments" in reporting states may have led to avoidance of PCI in patients who were less likely to benefit. Second, public reporting might have resulted in a drive toward improved quality of care and improved outcomes in patients receiving PCI, offsetting the adverse effect of not performing PCI in high-risk patients. Third, the optimal coding of comorbid condition required in risk adjustment may have been gamed through upcoding, thus leading to observed outcomes that are better than predicted. Better coding in public reporting states might have mitigated the adverse effect of denial of care after risk adjustment, he suggested.
This study highlights the possible unintended consequences of public reporting. The findings "may help spearhead a new focus on procedures that, while perceived [to be] appropriate based on current use criteria, might not result in added benefit in selected patients," he added.
Mauro Moscucci, M.D., is chief of the cardiovascular division at the University of Miami. He reported no relevant conflicts of interest. These remarks were taken from his editorial accompanying Dr. Joynt's report (JAMA 2012;308:148-9).
The use of percutaneous coronary intervention for acute myocardial infarction was found to be lower in three states that implemented public reporting of PCI outcomes than in seven nearby states without public reporting, according to a study of nearly 98,000 cases.
In addition, the use of PCI for acute MI declined in one state after public reporting of PCI outcomes was implemented there, said Dr. Karen E. Joynt of the departments of health policy and management at Harvard School of Public Health, Boston, and her associates.
Nevertheless, public reporting was not associated with any change in mortality for patients with acute MI in this study, they noted.
Collecting and publicly reporting patient outcomes is a tool intended to improve health care by motivating clinicians to improve their performance and allowing patients to choose the highest-quality hospitals. Critics of this strategy, however, say that it creates disincentives for physicians and hospitals to care for the sickest patients and may lead them to avoid offering lifesaving procedures such as PCI to the sickest or highest-risk patients.
To date, no national studies have examined whether public reporting of PCI outcomes has affected either the rates of PCI or the outcomes in patients with acute MI. Dr. Joynt and her colleagues did so using data from Medicare files.
They first performed a cross-sectional analysis of PCI rates in 30,745 patients who had a discharge diagnosis of acute MI in a single year, 2010. They compared the rates in three states that mandated public reporting -- Massachusetts, Pennsylvania, and New York -- against rates in seven nearby states that did not (Maine, Vermont, New Hampshire, Connecticut, Rhode Island, Maryland, and Delaware).
Patients in the public-reporting states were significantly less likely to receive PCI (37.7%) than were those in nonreporting states (42.7%). This trend was most pronounced in MI patients who had ST-elevation MI, cardiogenic shock, or cardiac arrest and was not seen in patients with non- ST-elevation MI, the investigators said (JAMA 2012;308:1460-8).
When the patients were categorized by age -- 65-74 years vs. 75 years and older -- the results were the same: Regardless of their age, patients in publicly reporting states were less likely to receive PCI than were those in nonreporting states.
The researchers then performed a longitudinal analysis of trends in PCI rates for 49,660 acute MI patients in reporting states and 48,142 in nonreporting states who were treated in 2002-2010. They focused on the experience in Massachusetts, tracking the rates before public reporting of PCI was implemented there (2002-2004) with the rates after it was implemented (2006-2010).
Before public reporting of PCI was implemented, the PCI rate in Massachusetts (40.6%) was comparable with that in nonreporting states (41.8%), but PCI rates in Massachusetts began to decline when reporting was implemented and by 2010 patients in Massachusetts were significantly less likely to receive PCI than were those in nonreporting states.
As in the cross-sectional study, PCI rates in Massachusetts declined the most among MI patients who had cardiogenic shock or cardiac arrest, and these findings did not change when patients were categorized by younger vs. older age.
Despite these declines in PCI rates, there was no significant difference in 30-day mortality between acute MI patients in reporting states (12.8%) and those in nonreporting states (12.1%). Some may find it reassuring that mandating public reporting did not increase patient mortality, but, conversely, it also did not reduce mortality, the researchers said.
The authors proposed two explanations why mortality was not affected by reductions in PCI rates. First, it may be that public reporting had its intended effect of focusing clinicians on performing PCI in only the most appropriate patients. Alternatively, it's possible that public reporting had an effect of pressuring physicians to avoid PCI in eligible but high-risk patients.
This study was funded by the NHLBI. No financial conflicts were reported.
The use of percutaneous coronary intervention for acute myocardial infarction was found to be lower in three states that implemented public reporting of PCI outcomes than in seven nearby states without public reporting, according to a study of nearly 98,000 cases.
In addition, the use of PCI for acute MI declined in one state after public reporting of PCI outcomes was implemented there, said Dr. Karen E. Joynt of the departments of health policy and management at Harvard School of Public Health, Boston, and her associates.
Nevertheless, public reporting was not associated with any change in mortality for patients with acute MI in this study, they noted.
Collecting and publicly reporting patient outcomes is a tool intended to improve health care by motivating clinicians to improve their performance and allowing patients to choose the highest-quality hospitals. Critics of this strategy, however, say that it creates disincentives for physicians and hospitals to care for the sickest patients and may lead them to avoid offering lifesaving procedures such as PCI to the sickest or highest-risk patients.
To date, no national studies have examined whether public reporting of PCI outcomes has affected either the rates of PCI or the outcomes in patients with acute MI. Dr. Joynt and her colleagues did so using data from Medicare files.
They first performed a cross-sectional analysis of PCI rates in 30,745 patients who had a discharge diagnosis of acute MI in a single year, 2010. They compared the rates in three states that mandated public reporting -- Massachusetts, Pennsylvania, and New York -- against rates in seven nearby states that did not (Maine, Vermont, New Hampshire, Connecticut, Rhode Island, Maryland, and Delaware).
Patients in the public-reporting states were significantly less likely to receive PCI (37.7%) than were those in nonreporting states (42.7%). This trend was most pronounced in MI patients who had ST-elevation MI, cardiogenic shock, or cardiac arrest and was not seen in patients with non- ST-elevation MI, the investigators said (JAMA 2012;308:1460-8).
When the patients were categorized by age -- 65-74 years vs. 75 years and older -- the results were the same: Regardless of their age, patients in publicly reporting states were less likely to receive PCI than were those in nonreporting states.
The researchers then performed a longitudinal analysis of trends in PCI rates for 49,660 acute MI patients in reporting states and 48,142 in nonreporting states who were treated in 2002-2010. They focused on the experience in Massachusetts, tracking the rates before public reporting of PCI was implemented there (2002-2004) with the rates after it was implemented (2006-2010).
Before public reporting of PCI was implemented, the PCI rate in Massachusetts (40.6%) was comparable with that in nonreporting states (41.8%), but PCI rates in Massachusetts began to decline when reporting was implemented and by 2010 patients in Massachusetts were significantly less likely to receive PCI than were those in nonreporting states.
As in the cross-sectional study, PCI rates in Massachusetts declined the most among MI patients who had cardiogenic shock or cardiac arrest, and these findings did not change when patients were categorized by younger vs. older age.
Despite these declines in PCI rates, there was no significant difference in 30-day mortality between acute MI patients in reporting states (12.8%) and those in nonreporting states (12.1%). Some may find it reassuring that mandating public reporting did not increase patient mortality, but, conversely, it also did not reduce mortality, the researchers said.
The authors proposed two explanations why mortality was not affected by reductions in PCI rates. First, it may be that public reporting had its intended effect of focusing clinicians on performing PCI in only the most appropriate patients. Alternatively, it's possible that public reporting had an effect of pressuring physicians to avoid PCI in eligible but high-risk patients.
This study was funded by the NHLBI. No financial conflicts were reported.
Major Finding: Patients with acute MI were less likely to receive PCI in states with mandatory public reporting of PCI outcomes (37.7%) than in those without public reporting (42.7%).
Data Source: A longitudinal analysis included PCI rates and outcomes for 49,660 acute MI patients treated in reporting states and 48,142 treated in nonreporting states in 2002-2010; there also was a cross-sectional analysis of 30,745 patients treated in 2010 only.
Disclosures: This study was supported by the National Heart, Lung, and Blood Institute. No financial conflicts of interest were reported.