User login
Lucas Franki is an associate editor for MDedge News, and has been with the company since 2014. He has a BA in English from Penn State University and is an Eagle Scout.
Cancer mortality lowest in western United States
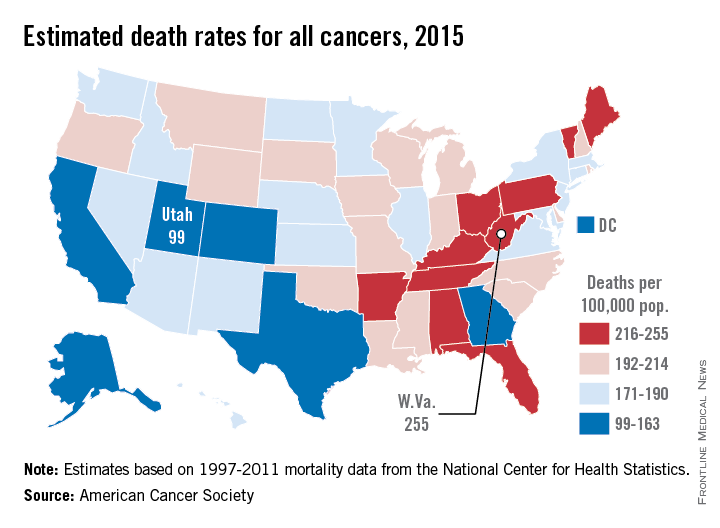
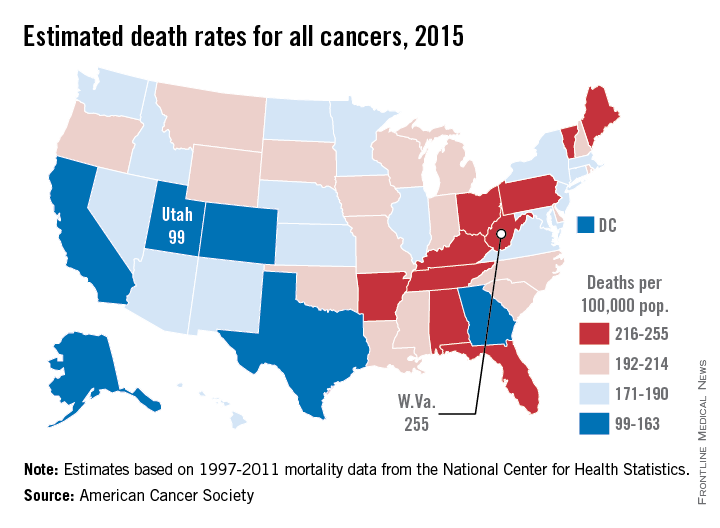
Utah will have the lowest cancer mortality rate in the United States in 2015, according to a report from the American Cancer Society.
While 11 states are predicted to have fewer cancer-related deaths in 2015 than 2014, the mortality rate will be lowest in Utah, with just 99 deaths per 100,000 people. Alaska and Colorado will have the next lowest mortality rates at 141 and 142 per 100,000 people, respectively. West Virginia is estimated to have the highest cancer mortality rate at 255 per 100,000 people, followed by Kentucky and Arkansas at 231 and 228, respectively. The national cancer mortality rate will be 185 per 100,000.
There will be about 1.66 million new cases of cancer in 2015 and about 590,000 deaths. Female breast cancer will probably be the most common with 231,000 new cases, but with an estimated 221,000 new cases each, lung/bronchus and prostate cancer also will rank high. Lung and bronchus cancer will be the most common cause of death, with 158,000 deaths predicted in 2015, more than a quarter of overall cancer deaths, according to the ACS.
Socioeconomic status makes a big difference in both cancer incidence and mortality. Cancer can stem from a higher likelihood to engage in risky behavior such as smoking, an unhealthy diet, or a sedentary lifestyle. Demographics matter more than ethnicity for cancer mortality, as “cancer mortality rates among both black and non-Hispanic white men with 12 or fewer years of education are almost 3 times higher than those of college graduates for all cancers combined and 4-5 times higher for lung cancer,” the ACS said.
Estimated data were based on 1995-2011 cancer incidence rates collected by the National Center for Health Statistics and the ACS.
Utah will have the lowest cancer mortality rate in the United States in 2015, according to a report from the American Cancer Society.
While 11 states are predicted to have fewer cancer-related deaths in 2015 than 2014, the mortality rate will be lowest in Utah, with just 99 deaths per 100,000 people. Alaska and Colorado will have the next lowest mortality rates at 141 and 142 per 100,000 people, respectively. West Virginia is estimated to have the highest cancer mortality rate at 255 per 100,000 people, followed by Kentucky and Arkansas at 231 and 228, respectively. The national cancer mortality rate will be 185 per 100,000.

There will be about 1.66 million new cases of cancer in 2015 and about 590,000 deaths. Female breast cancer will probably be the most common with 231,000 new cases, but with an estimated 221,000 new cases each, lung/bronchus and prostate cancer also will rank high. Lung and bronchus cancer will be the most common cause of death, with 158,000 deaths predicted in 2015, more than a quarter of overall cancer deaths, according to the ACS.
Socioeconomic status makes a big difference in both cancer incidence and mortality. Cancer can stem from a higher likelihood to engage in risky behavior such as smoking, an unhealthy diet, or a sedentary lifestyle. Demographics matter more than ethnicity for cancer mortality, as “cancer mortality rates among both black and non-Hispanic white men with 12 or fewer years of education are almost 3 times higher than those of college graduates for all cancers combined and 4-5 times higher for lung cancer,” the ACS said.
Estimated data were based on 1995-2011 cancer incidence rates collected by the National Center for Health Statistics and the ACS.
Utah will have the lowest cancer mortality rate in the United States in 2015, according to a report from the American Cancer Society.
While 11 states are predicted to have fewer cancer-related deaths in 2015 than 2014, the mortality rate will be lowest in Utah, with just 99 deaths per 100,000 people. Alaska and Colorado will have the next lowest mortality rates at 141 and 142 per 100,000 people, respectively. West Virginia is estimated to have the highest cancer mortality rate at 255 per 100,000 people, followed by Kentucky and Arkansas at 231 and 228, respectively. The national cancer mortality rate will be 185 per 100,000.

There will be about 1.66 million new cases of cancer in 2015 and about 590,000 deaths. Female breast cancer will probably be the most common with 231,000 new cases, but with an estimated 221,000 new cases each, lung/bronchus and prostate cancer also will rank high. Lung and bronchus cancer will be the most common cause of death, with 158,000 deaths predicted in 2015, more than a quarter of overall cancer deaths, according to the ACS.
Socioeconomic status makes a big difference in both cancer incidence and mortality. Cancer can stem from a higher likelihood to engage in risky behavior such as smoking, an unhealthy diet, or a sedentary lifestyle. Demographics matter more than ethnicity for cancer mortality, as “cancer mortality rates among both black and non-Hispanic white men with 12 or fewer years of education are almost 3 times higher than those of college graduates for all cancers combined and 4-5 times higher for lung cancer,” the ACS said.
Estimated data were based on 1995-2011 cancer incidence rates collected by the National Center for Health Statistics and the ACS.
ACIP Recommends Pneumococcal Vaccine for 65+ in 2015 Schedule
The 2015 adult immunization schedule is similar to last year’s schedule, with one major change, according to the Centers for Disease Control and Prevention's Advisory Committee on Immunization Practices.
The 13-valent pneumococcal conjugate vaccine in series with the 23-valent pneumococcal polysaccharide vaccine is now recommended for all adults over 65, rather than being recommended only if another risk was present.
While incidence of invasive pneumococcal diseases caused by PCV13 serotypes has dropped 50% since 2010 because of indirect effects from pediatric vaccination efforts, 20%-25% of invasive pneumococcal diseases in people aged older than 65 years are still caused by PCV13 serotypes, the ACIP noted (Ann. Intern. Med. 2015;162:214-223 [doi:10.7326/M14-2755]).
The committee recommends that adults over 65 who have not already had PCV13 should receive the vaccine, followed by a dose of PPSV23 6-12 months later. Additional doses of PPSV23 are recommended if other risk factors are present. The footnotes for pneumococcal vaccinations also have been revised to provide guidance for physicians seeking appropriate vaccination decisions for individual patients.
In the influenza vaccination footnotes, all adults can now receive recombinant influenza vaccine, previously only recommended for adults aged 18-49. Several contraindications for the live attenuated influenza vaccine have been reclassified as precautions, including influenza antiviral use within the last 48 hours; asthma and chronic lung diseases; diabetes; and cardiovascular, renal, and hepatic diseases.
While the Affordable Care Act requires that all vaccines recommended by ACIP be covered with no copayment for the patient as preventive care services, the same is not true for Medicare. Medicare currently covers only one dose of pneumococcal vaccine. A rule change will be necessary before Medicare can cover the recommended vaccinations; such a change could take more than a year to go into effect, Dr. Sandra Fryhofer, the American College of Physicians’ liaison to the ACIP, said in a related editorial (Ann. Intern. Med. 2015;162:235-6 [doi:10.7326/M14-2547]).
The ACIP schedule can be found on the CDC website.
The 2015 adult immunization schedule is similar to last year’s schedule, with one major change, according to the Centers for Disease Control and Prevention's Advisory Committee on Immunization Practices.
The 13-valent pneumococcal conjugate vaccine in series with the 23-valent pneumococcal polysaccharide vaccine is now recommended for all adults over 65, rather than being recommended only if another risk was present.
While incidence of invasive pneumococcal diseases caused by PCV13 serotypes has dropped 50% since 2010 because of indirect effects from pediatric vaccination efforts, 20%-25% of invasive pneumococcal diseases in people aged older than 65 years are still caused by PCV13 serotypes, the ACIP noted (Ann. Intern. Med. 2015;162:214-223 [doi:10.7326/M14-2755]).
The committee recommends that adults over 65 who have not already had PCV13 should receive the vaccine, followed by a dose of PPSV23 6-12 months later. Additional doses of PPSV23 are recommended if other risk factors are present. The footnotes for pneumococcal vaccinations also have been revised to provide guidance for physicians seeking appropriate vaccination decisions for individual patients.
In the influenza vaccination footnotes, all adults can now receive recombinant influenza vaccine, previously only recommended for adults aged 18-49. Several contraindications for the live attenuated influenza vaccine have been reclassified as precautions, including influenza antiviral use within the last 48 hours; asthma and chronic lung diseases; diabetes; and cardiovascular, renal, and hepatic diseases.
While the Affordable Care Act requires that all vaccines recommended by ACIP be covered with no copayment for the patient as preventive care services, the same is not true for Medicare. Medicare currently covers only one dose of pneumococcal vaccine. A rule change will be necessary before Medicare can cover the recommended vaccinations; such a change could take more than a year to go into effect, Dr. Sandra Fryhofer, the American College of Physicians’ liaison to the ACIP, said in a related editorial (Ann. Intern. Med. 2015;162:235-6 [doi:10.7326/M14-2547]).
The ACIP schedule can be found on the CDC website.
The 2015 adult immunization schedule is similar to last year’s schedule, with one major change, according to the Centers for Disease Control and Prevention's Advisory Committee on Immunization Practices.
The 13-valent pneumococcal conjugate vaccine in series with the 23-valent pneumococcal polysaccharide vaccine is now recommended for all adults over 65, rather than being recommended only if another risk was present.
While incidence of invasive pneumococcal diseases caused by PCV13 serotypes has dropped 50% since 2010 because of indirect effects from pediatric vaccination efforts, 20%-25% of invasive pneumococcal diseases in people aged older than 65 years are still caused by PCV13 serotypes, the ACIP noted (Ann. Intern. Med. 2015;162:214-223 [doi:10.7326/M14-2755]).
The committee recommends that adults over 65 who have not already had PCV13 should receive the vaccine, followed by a dose of PPSV23 6-12 months later. Additional doses of PPSV23 are recommended if other risk factors are present. The footnotes for pneumococcal vaccinations also have been revised to provide guidance for physicians seeking appropriate vaccination decisions for individual patients.
In the influenza vaccination footnotes, all adults can now receive recombinant influenza vaccine, previously only recommended for adults aged 18-49. Several contraindications for the live attenuated influenza vaccine have been reclassified as precautions, including influenza antiviral use within the last 48 hours; asthma and chronic lung diseases; diabetes; and cardiovascular, renal, and hepatic diseases.
While the Affordable Care Act requires that all vaccines recommended by ACIP be covered with no copayment for the patient as preventive care services, the same is not true for Medicare. Medicare currently covers only one dose of pneumococcal vaccine. A rule change will be necessary before Medicare can cover the recommended vaccinations; such a change could take more than a year to go into effect, Dr. Sandra Fryhofer, the American College of Physicians’ liaison to the ACIP, said in a related editorial (Ann. Intern. Med. 2015;162:235-6 [doi:10.7326/M14-2547]).
The ACIP schedule can be found on the CDC website.
FROM ANNALS OF INTERNAL MEDICINE
ACIP recommends pneumococcal vaccine for 65+ in 2015 schedule
The 2015 adult immunization schedule is similar to last year’s schedule, with one major change, according to the Centers for Disease Control and Prevention's Advisory Committee on Immunization Practices.
The 13-valent pneumococcal conjugate vaccine in series with the 23-valent pneumococcal polysaccharide vaccine is now recommended for all adults over 65, rather than being recommended only if another risk was presen
While incidence of invasive pneumococcal diseases caused by PCV13 serotypes has dropped 50% since 2010 because of indirect effects from pediatric vaccination efforts, 20%-25% of invasive pneumococcal diseases in people aged older than 65 years are still caused by PCV13 serotypes, the ACIP noted (Ann. Intern. Med. 2015;162:214-223 [doi:10.7326/M14-2755]).
The committee recommends that adults over 65 who have not already had PCV13 should receive the vaccine, followed by a dose of PPSV23 6-12 months later. Additional doses of PPSV23 are recommended if other risk factors are present. The footnotes for pneumococcal vaccinations also have been revised to provide guidance for physicians seeking appropriate vaccination decisions for individual patients.
In the influenza vaccination footnotes, all adults can now receive recombinant influenza vaccine, previously only recommended for adults aged 18-49. Several contraindications for the live attenuated influenza vaccine have been reclassified as precautions, including influenza antiviral use within the last 48 hours; asthma and chronic lung diseases; diabetes; and cardiovascular, renal, and hepatic diseases.
While the Affordable Care Act requires that all vaccines recommended by ACIP be covered with no copayment for the patient as preventive care services, the same is not true for Medicare. Medicare currently covers only one dose of pneumococcal vaccine. A rule change will be necessary before Medicare can cover the recommended vaccinations; such a change could take more than a year to go into effect, Dr. Sandra Fryhofer, the American College of Physicians’ liaison to the ACIP, said in a related editorial (Ann. Intern. Med. 2015;162:235-6 [doi:10.7326/M14-2547]).
The ACIP schedule can be found on the CDC website.
The 2015 adult immunization schedule is similar to last year’s schedule, with one major change, according to the Centers for Disease Control and Prevention's Advisory Committee on Immunization Practices.
The 13-valent pneumococcal conjugate vaccine in series with the 23-valent pneumococcal polysaccharide vaccine is now recommended for all adults over 65, rather than being recommended only if another risk was presen
While incidence of invasive pneumococcal diseases caused by PCV13 serotypes has dropped 50% since 2010 because of indirect effects from pediatric vaccination efforts, 20%-25% of invasive pneumococcal diseases in people aged older than 65 years are still caused by PCV13 serotypes, the ACIP noted (Ann. Intern. Med. 2015;162:214-223 [doi:10.7326/M14-2755]).
The committee recommends that adults over 65 who have not already had PCV13 should receive the vaccine, followed by a dose of PPSV23 6-12 months later. Additional doses of PPSV23 are recommended if other risk factors are present. The footnotes for pneumococcal vaccinations also have been revised to provide guidance for physicians seeking appropriate vaccination decisions for individual patients.
In the influenza vaccination footnotes, all adults can now receive recombinant influenza vaccine, previously only recommended for adults aged 18-49. Several contraindications for the live attenuated influenza vaccine have been reclassified as precautions, including influenza antiviral use within the last 48 hours; asthma and chronic lung diseases; diabetes; and cardiovascular, renal, and hepatic diseases.
While the Affordable Care Act requires that all vaccines recommended by ACIP be covered with no copayment for the patient as preventive care services, the same is not true for Medicare. Medicare currently covers only one dose of pneumococcal vaccine. A rule change will be necessary before Medicare can cover the recommended vaccinations; such a change could take more than a year to go into effect, Dr. Sandra Fryhofer, the American College of Physicians’ liaison to the ACIP, said in a related editorial (Ann. Intern. Med. 2015;162:235-6 [doi:10.7326/M14-2547]).
The ACIP schedule can be found on the CDC website.
The 2015 adult immunization schedule is similar to last year’s schedule, with one major change, according to the Centers for Disease Control and Prevention's Advisory Committee on Immunization Practices.
The 13-valent pneumococcal conjugate vaccine in series with the 23-valent pneumococcal polysaccharide vaccine is now recommended for all adults over 65, rather than being recommended only if another risk was presen
While incidence of invasive pneumococcal diseases caused by PCV13 serotypes has dropped 50% since 2010 because of indirect effects from pediatric vaccination efforts, 20%-25% of invasive pneumococcal diseases in people aged older than 65 years are still caused by PCV13 serotypes, the ACIP noted (Ann. Intern. Med. 2015;162:214-223 [doi:10.7326/M14-2755]).
The committee recommends that adults over 65 who have not already had PCV13 should receive the vaccine, followed by a dose of PPSV23 6-12 months later. Additional doses of PPSV23 are recommended if other risk factors are present. The footnotes for pneumococcal vaccinations also have been revised to provide guidance for physicians seeking appropriate vaccination decisions for individual patients.
In the influenza vaccination footnotes, all adults can now receive recombinant influenza vaccine, previously only recommended for adults aged 18-49. Several contraindications for the live attenuated influenza vaccine have been reclassified as precautions, including influenza antiviral use within the last 48 hours; asthma and chronic lung diseases; diabetes; and cardiovascular, renal, and hepatic diseases.
While the Affordable Care Act requires that all vaccines recommended by ACIP be covered with no copayment for the patient as preventive care services, the same is not true for Medicare. Medicare currently covers only one dose of pneumococcal vaccine. A rule change will be necessary before Medicare can cover the recommended vaccinations; such a change could take more than a year to go into effect, Dr. Sandra Fryhofer, the American College of Physicians’ liaison to the ACIP, said in a related editorial (Ann. Intern. Med. 2015;162:235-6 [doi:10.7326/M14-2547]).
The ACIP schedule can be found on the CDC website.
FROM ANNALS OF INTERNAL MEDICINE
New immunization schedule moving online, but changes are minor
The 2015 childhood and adolescent immunization schedule will be published online only for the first time, according to the American Academy of Pediatrics and the Centers for Disease Control and Prevention.
Moving the schedule online will ensure that providers have the most current recommendations, according to the AAP’s Committee on Infectious Diseases (Pediatrics 2015 [doi:10.1542/peds.2014-3955]). The schedule will be updated when new vaccines are released, new recommendations are published, or when a current recommendation is changed. The website also will include tables to clarify recommended use of certain vaccines, such as vaccines containing pertussis.
Changes to the 2015 schedule are relatively minor. A new column highlights availability of the inactivated and live-attenuated influenza vaccine for children 2-8 years old, along with a recommendation for two doses for some children; a second column indicates when two doses are no longer needed. In addition, a purple bar has been added for young children aged 6 months to less than 12 months traveling outside the United States and who will need the MMR vaccine, the committee said.
Small clarifying changes were added to many vaccinations in the catch-up schedule, and minor changes were made in the footnotes to the DTaP and pneumococcal conjugate vaccines. The influenza footnote now includes new contraindications and precautions for the live-attenuated vaccine. Extensive changes were made to the meningococcal footnote, which clarify dosing schedules for children and infants at high risk, the committee reported.
The 2015 schedule can be found on the CDC website and on the AAP website.
The 2015 childhood and adolescent immunization schedule will be published online only for the first time, according to the American Academy of Pediatrics and the Centers for Disease Control and Prevention.
Moving the schedule online will ensure that providers have the most current recommendations, according to the AAP’s Committee on Infectious Diseases (Pediatrics 2015 [doi:10.1542/peds.2014-3955]). The schedule will be updated when new vaccines are released, new recommendations are published, or when a current recommendation is changed. The website also will include tables to clarify recommended use of certain vaccines, such as vaccines containing pertussis.
Changes to the 2015 schedule are relatively minor. A new column highlights availability of the inactivated and live-attenuated influenza vaccine for children 2-8 years old, along with a recommendation for two doses for some children; a second column indicates when two doses are no longer needed. In addition, a purple bar has been added for young children aged 6 months to less than 12 months traveling outside the United States and who will need the MMR vaccine, the committee said.
Small clarifying changes were added to many vaccinations in the catch-up schedule, and minor changes were made in the footnotes to the DTaP and pneumococcal conjugate vaccines. The influenza footnote now includes new contraindications and precautions for the live-attenuated vaccine. Extensive changes were made to the meningococcal footnote, which clarify dosing schedules for children and infants at high risk, the committee reported.
The 2015 schedule can be found on the CDC website and on the AAP website.
The 2015 childhood and adolescent immunization schedule will be published online only for the first time, according to the American Academy of Pediatrics and the Centers for Disease Control and Prevention.
Moving the schedule online will ensure that providers have the most current recommendations, according to the AAP’s Committee on Infectious Diseases (Pediatrics 2015 [doi:10.1542/peds.2014-3955]). The schedule will be updated when new vaccines are released, new recommendations are published, or when a current recommendation is changed. The website also will include tables to clarify recommended use of certain vaccines, such as vaccines containing pertussis.
Changes to the 2015 schedule are relatively minor. A new column highlights availability of the inactivated and live-attenuated influenza vaccine for children 2-8 years old, along with a recommendation for two doses for some children; a second column indicates when two doses are no longer needed. In addition, a purple bar has been added for young children aged 6 months to less than 12 months traveling outside the United States and who will need the MMR vaccine, the committee said.
Small clarifying changes were added to many vaccinations in the catch-up schedule, and minor changes were made in the footnotes to the DTaP and pneumococcal conjugate vaccines. The influenza footnote now includes new contraindications and precautions for the live-attenuated vaccine. Extensive changes were made to the meningococcal footnote, which clarify dosing schedules for children and infants at high risk, the committee reported.
The 2015 schedule can be found on the CDC website and on the AAP website.
FROM PEDIATRICS
FDA Approves Use of Parathyroid Hormone to Manage Hypocalcemia
The parathyroid hormone Natpara has been approved to manage hypocalcemia in patients with hypoparathyroidism, according to a press release from the Food and Drug Administration.
In the phase III REPLACE study, 124 participants were randomly assigned Natpara or a placebo to determine whether Natpara can be used as a substitute for or help reduce the amount of active forms of vitamin D or oral calcium taken by participants. Of patients treated with Natpara, 42% achieved normal blood calcium levels, while 3% of patients achieved normal blood calcium levels on the placebo, the FDA said.
“People with hypoparathyroidism have limited treatment options and face challenging symptoms that can severely impact their quality of life. This product offers an alternative to patients whose calcium levels cannot be controlled on calcium supplementation and active forms of vitamin D,” Dr. Jean-Marc Guettier, director of the Division of Metabolism and Endocrinology Products in the FDA’s Center for Drug Evaluation and Research, said in the press release.
Because of instances of bone cancer in rat studies, Natpara carries a warning and is only recommended for patients whose hypocalcemia cannot be controlled by other means. Common side effects include sensations of tingling, low blood calcium, headache, high blood calcium, and nausea.
Hypoparathyroidism is a rare disease, only affecting about 60,000 people in the United States. It occurs when loss of function of the parathyroid glands causes the body to secrete abnormally low levels of parathyroid hormone, which helps regulate calcium and phosphorus levels in the body; surgical removal of the parathyroid glands is another cause as is, and more rarely, autoimmune or congenital diseases, according to the FDA.
The parathyroid hormone Natpara has been approved to manage hypocalcemia in patients with hypoparathyroidism, according to a press release from the Food and Drug Administration.
In the phase III REPLACE study, 124 participants were randomly assigned Natpara or a placebo to determine whether Natpara can be used as a substitute for or help reduce the amount of active forms of vitamin D or oral calcium taken by participants. Of patients treated with Natpara, 42% achieved normal blood calcium levels, while 3% of patients achieved normal blood calcium levels on the placebo, the FDA said.
“People with hypoparathyroidism have limited treatment options and face challenging symptoms that can severely impact their quality of life. This product offers an alternative to patients whose calcium levels cannot be controlled on calcium supplementation and active forms of vitamin D,” Dr. Jean-Marc Guettier, director of the Division of Metabolism and Endocrinology Products in the FDA’s Center for Drug Evaluation and Research, said in the press release.
Because of instances of bone cancer in rat studies, Natpara carries a warning and is only recommended for patients whose hypocalcemia cannot be controlled by other means. Common side effects include sensations of tingling, low blood calcium, headache, high blood calcium, and nausea.
Hypoparathyroidism is a rare disease, only affecting about 60,000 people in the United States. It occurs when loss of function of the parathyroid glands causes the body to secrete abnormally low levels of parathyroid hormone, which helps regulate calcium and phosphorus levels in the body; surgical removal of the parathyroid glands is another cause as is, and more rarely, autoimmune or congenital diseases, according to the FDA.
The parathyroid hormone Natpara has been approved to manage hypocalcemia in patients with hypoparathyroidism, according to a press release from the Food and Drug Administration.
In the phase III REPLACE study, 124 participants were randomly assigned Natpara or a placebo to determine whether Natpara can be used as a substitute for or help reduce the amount of active forms of vitamin D or oral calcium taken by participants. Of patients treated with Natpara, 42% achieved normal blood calcium levels, while 3% of patients achieved normal blood calcium levels on the placebo, the FDA said.
“People with hypoparathyroidism have limited treatment options and face challenging symptoms that can severely impact their quality of life. This product offers an alternative to patients whose calcium levels cannot be controlled on calcium supplementation and active forms of vitamin D,” Dr. Jean-Marc Guettier, director of the Division of Metabolism and Endocrinology Products in the FDA’s Center for Drug Evaluation and Research, said in the press release.
Because of instances of bone cancer in rat studies, Natpara carries a warning and is only recommended for patients whose hypocalcemia cannot be controlled by other means. Common side effects include sensations of tingling, low blood calcium, headache, high blood calcium, and nausea.
Hypoparathyroidism is a rare disease, only affecting about 60,000 people in the United States. It occurs when loss of function of the parathyroid glands causes the body to secrete abnormally low levels of parathyroid hormone, which helps regulate calcium and phosphorus levels in the body; surgical removal of the parathyroid glands is another cause as is, and more rarely, autoimmune or congenital diseases, according to the FDA.
FROM THE FDA
FDA approves use of parathyroid hormone to manage hypocalcemia
The parathyroid hormone Natpara has been approved to manage hypocalcemia in patients with hypoparathyroidism, according to a press release from the Food and Drug Administration.
In the phase III REPLACE study, 124 participants were randomly assigned Natpara or a placebo to determine whether Natpara can be used as a substitute for or help reduce the amount of active forms of vitamin D or oral calcium taken by participants. Of patients treated with Natpara, 42% achieved normal blood calcium levels, while 3% of patients achieved normal blood calcium levels on the placebo, the FDA said.
“People with hypoparathyroidism have limited treatment options and face challenging symptoms that can severely impact their quality of life. This product offers an alternative to patients whose calcium levels cannot be controlled on calcium supplementation and active forms of vitamin D,” Dr. Jean-Marc Guettier, director of the Division of Metabolism and Endocrinology Products in the FDA’s Center for Drug Evaluation and Research, said in the press release.
Because of instances of bone cancer in rat studies, Natpara carries a warning and is only recommended for patients whose hypocalcemia cannot be controlled by other means. Common side effects include sensations of tingling, low blood calcium, headache, high blood calcium, and nausea.
Hypoparathyroidism is a rare disease, only affecting about 60,000 people in the United States. It occurs when loss of function of the parathyroid glands causes the body to secrete abnormally low levels of parathyroid hormone, which helps regulate calcium and phosphorus levels in the body; surgical removal of the parathyroid glands is another cause as is, and more rarely, autoimmune or congenital diseases, according to the FDA.
Natpara is manufactured by NPS Pharmaceuticals, Inc.
The parathyroid hormone Natpara has been approved to manage hypocalcemia in patients with hypoparathyroidism, according to a press release from the Food and Drug Administration.
In the phase III REPLACE study, 124 participants were randomly assigned Natpara or a placebo to determine whether Natpara can be used as a substitute for or help reduce the amount of active forms of vitamin D or oral calcium taken by participants. Of patients treated with Natpara, 42% achieved normal blood calcium levels, while 3% of patients achieved normal blood calcium levels on the placebo, the FDA said.
“People with hypoparathyroidism have limited treatment options and face challenging symptoms that can severely impact their quality of life. This product offers an alternative to patients whose calcium levels cannot be controlled on calcium supplementation and active forms of vitamin D,” Dr. Jean-Marc Guettier, director of the Division of Metabolism and Endocrinology Products in the FDA’s Center for Drug Evaluation and Research, said in the press release.
Because of instances of bone cancer in rat studies, Natpara carries a warning and is only recommended for patients whose hypocalcemia cannot be controlled by other means. Common side effects include sensations of tingling, low blood calcium, headache, high blood calcium, and nausea.
Hypoparathyroidism is a rare disease, only affecting about 60,000 people in the United States. It occurs when loss of function of the parathyroid glands causes the body to secrete abnormally low levels of parathyroid hormone, which helps regulate calcium and phosphorus levels in the body; surgical removal of the parathyroid glands is another cause as is, and more rarely, autoimmune or congenital diseases, according to the FDA.
Natpara is manufactured by NPS Pharmaceuticals, Inc.
The parathyroid hormone Natpara has been approved to manage hypocalcemia in patients with hypoparathyroidism, according to a press release from the Food and Drug Administration.
In the phase III REPLACE study, 124 participants were randomly assigned Natpara or a placebo to determine whether Natpara can be used as a substitute for or help reduce the amount of active forms of vitamin D or oral calcium taken by participants. Of patients treated with Natpara, 42% achieved normal blood calcium levels, while 3% of patients achieved normal blood calcium levels on the placebo, the FDA said.
“People with hypoparathyroidism have limited treatment options and face challenging symptoms that can severely impact their quality of life. This product offers an alternative to patients whose calcium levels cannot be controlled on calcium supplementation and active forms of vitamin D,” Dr. Jean-Marc Guettier, director of the Division of Metabolism and Endocrinology Products in the FDA’s Center for Drug Evaluation and Research, said in the press release.
Because of instances of bone cancer in rat studies, Natpara carries a warning and is only recommended for patients whose hypocalcemia cannot be controlled by other means. Common side effects include sensations of tingling, low blood calcium, headache, high blood calcium, and nausea.
Hypoparathyroidism is a rare disease, only affecting about 60,000 people in the United States. It occurs when loss of function of the parathyroid glands causes the body to secrete abnormally low levels of parathyroid hormone, which helps regulate calcium and phosphorus levels in the body; surgical removal of the parathyroid glands is another cause as is, and more rarely, autoimmune or congenital diseases, according to the FDA.
Natpara is manufactured by NPS Pharmaceuticals, Inc.
FROM THE FDA
Psychiatric diagnoses rank first and third among Medicaid admissions
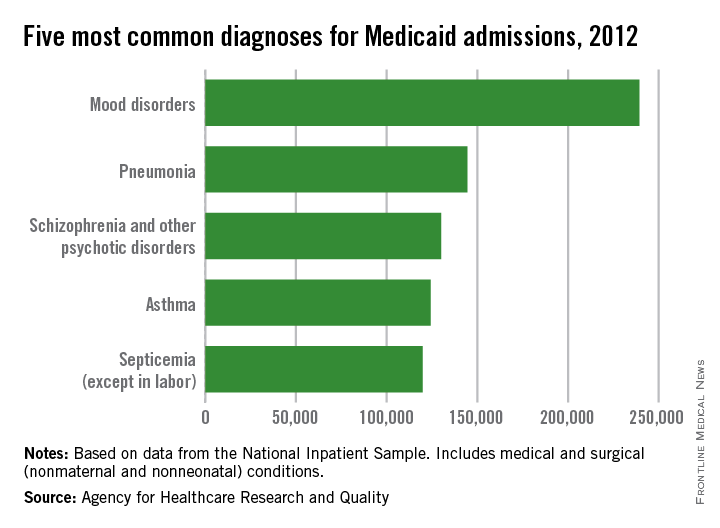
Mood disorders were the most common diagnosis for Medicaid-insured hospitalizations not involving neonatal or maternal stays in 2012, according to a report from the Agency for Healthcare Research and Quality.
Of the nearly 4 million Medicaid-insured hospitalizations, mood disorders were the primary diagnosis in about 239,000 cases, or just over 6%. Pneumonia was the second most common diagnosis, accounting for 145,000 cases. Schizophrenia and other psychoses were next with 130,000 cases, followed by asthma and septicemia, with 124,000 and 120,000 cases, respectively, according to the AHRQ.
Of the 10 most common diagnoses for Medicaid hospitalizations, 6 diseases are classified as ambulatory care sensitive conditions (ACSCs); that is, conditions that should not require hospitalization if properly treated outside a hospital, such as pneumonia, asthma, diabetes mellitus, epilepsy, acute bronchitis, and chronic obstructive pulmonary disease. Only pneumonia was in the 10 most common diagnoses for hospitalization of both uninsured and privately insured patients, and diabetes mellitus was the third most common cause of hospitalization among the uninsured. Although ACSCs accounted for almost 16% of Medicaid hospitalizations, they accounted for 6% of uninsured hospitalizations and less than 3% of privately insured hospitalizations, the report noted.
The remaining four common diagnoses for Medicaid hospitalizations include mood disorders, schizophrenia, septicemia, and skin infections. Of these, mood disorders, septicemia, and skin infections are common diagnoses for uninsured and privately insured hospitalizations. Schizophrenia and other psychotic disorders were a very common diagnosis for Medicaid-insured hospitalizations but not for uninsured patients or those who were privately insured, perhaps indicating a strength of Medicaid at getting schizophrenic patients into hospitals.
The AHRQ report used data collected by the National Inpatient Sample.
Mood disorders were the most common diagnosis for Medicaid-insured hospitalizations not involving neonatal or maternal stays in 2012, according to a report from the Agency for Healthcare Research and Quality.
Of the nearly 4 million Medicaid-insured hospitalizations, mood disorders were the primary diagnosis in about 239,000 cases, or just over 6%. Pneumonia was the second most common diagnosis, accounting for 145,000 cases. Schizophrenia and other psychoses were next with 130,000 cases, followed by asthma and septicemia, with 124,000 and 120,000 cases, respectively, according to the AHRQ.
Of the 10 most common diagnoses for Medicaid hospitalizations, 6 diseases are classified as ambulatory care sensitive conditions (ACSCs); that is, conditions that should not require hospitalization if properly treated outside a hospital, such as pneumonia, asthma, diabetes mellitus, epilepsy, acute bronchitis, and chronic obstructive pulmonary disease. Only pneumonia was in the 10 most common diagnoses for hospitalization of both uninsured and privately insured patients, and diabetes mellitus was the third most common cause of hospitalization among the uninsured. Although ACSCs accounted for almost 16% of Medicaid hospitalizations, they accounted for 6% of uninsured hospitalizations and less than 3% of privately insured hospitalizations, the report noted.
The remaining four common diagnoses for Medicaid hospitalizations include mood disorders, schizophrenia, septicemia, and skin infections. Of these, mood disorders, septicemia, and skin infections are common diagnoses for uninsured and privately insured hospitalizations. Schizophrenia and other psychotic disorders were a very common diagnosis for Medicaid-insured hospitalizations but not for uninsured patients or those who were privately insured, perhaps indicating a strength of Medicaid at getting schizophrenic patients into hospitals.
The AHRQ report used data collected by the National Inpatient Sample.
Mood disorders were the most common diagnosis for Medicaid-insured hospitalizations not involving neonatal or maternal stays in 2012, according to a report from the Agency for Healthcare Research and Quality.
Of the nearly 4 million Medicaid-insured hospitalizations, mood disorders were the primary diagnosis in about 239,000 cases, or just over 6%. Pneumonia was the second most common diagnosis, accounting for 145,000 cases. Schizophrenia and other psychoses were next with 130,000 cases, followed by asthma and septicemia, with 124,000 and 120,000 cases, respectively, according to the AHRQ.
Of the 10 most common diagnoses for Medicaid hospitalizations, 6 diseases are classified as ambulatory care sensitive conditions (ACSCs); that is, conditions that should not require hospitalization if properly treated outside a hospital, such as pneumonia, asthma, diabetes mellitus, epilepsy, acute bronchitis, and chronic obstructive pulmonary disease. Only pneumonia was in the 10 most common diagnoses for hospitalization of both uninsured and privately insured patients, and diabetes mellitus was the third most common cause of hospitalization among the uninsured. Although ACSCs accounted for almost 16% of Medicaid hospitalizations, they accounted for 6% of uninsured hospitalizations and less than 3% of privately insured hospitalizations, the report noted.
The remaining four common diagnoses for Medicaid hospitalizations include mood disorders, schizophrenia, septicemia, and skin infections. Of these, mood disorders, septicemia, and skin infections are common diagnoses for uninsured and privately insured hospitalizations. Schizophrenia and other psychotic disorders were a very common diagnosis for Medicaid-insured hospitalizations but not for uninsured patients or those who were privately insured, perhaps indicating a strength of Medicaid at getting schizophrenic patients into hospitals.
The AHRQ report used data collected by the National Inpatient Sample.
Increased heroin use may not be linked to rise in prescription opioid use
The increase in nonmedical prescription opioid use in the United States does not appear to be strongly related to the concurrent increase in heroin use, according to a review by Dr. Wilson Compton of the National Institute on Drug Abuse, Bethesda, Md., and his associates.
While heroin users are 3.9 times more likely to have used nonmedical prescription opioids than are those who haven’t used heroin, heroin use only occurs in a small number of nonmedical prescription opioid users. The researchers cited studies showing that 3.6% of opioid users began using heroin within 5 years of beginning opioid use, and 4.2% of opioid users reported also using heroin in the past year.
A more likely driver for the increased use of heroin and heroin death rate is decreased cost and increased availability, the investigators wrote. For every $100 decrease in price per gram of heroin, hospitalizations for heroin overdose increase by 2.9%. In addition, heroin use has grown significantly in areas of the United States that were not typically centers for heroin distribution, the researchers reported.
“Fundamentally, prescription opioids and heroin are each elements of a larger epidemic of opioid-related disorders and death. Viewing them from a unified perspective is essential to improving public health. The perniciousness of this epidemic requires a multipronged interventional approach that engages all sectors of society,” the investigators wrote.
Dr. Compton has ties with General Electric, 3M, and Pfizer. No other conflicts were reported. Find the study in the New England Journal of Medicine (doi: 10.1056/NEJMra1508490).
The increase in nonmedical prescription opioid use in the United States does not appear to be strongly related to the concurrent increase in heroin use, according to a review by Dr. Wilson Compton of the National Institute on Drug Abuse, Bethesda, Md., and his associates.
While heroin users are 3.9 times more likely to have used nonmedical prescription opioids than are those who haven’t used heroin, heroin use only occurs in a small number of nonmedical prescription opioid users. The researchers cited studies showing that 3.6% of opioid users began using heroin within 5 years of beginning opioid use, and 4.2% of opioid users reported also using heroin in the past year.
A more likely driver for the increased use of heroin and heroin death rate is decreased cost and increased availability, the investigators wrote. For every $100 decrease in price per gram of heroin, hospitalizations for heroin overdose increase by 2.9%. In addition, heroin use has grown significantly in areas of the United States that were not typically centers for heroin distribution, the researchers reported.
“Fundamentally, prescription opioids and heroin are each elements of a larger epidemic of opioid-related disorders and death. Viewing them from a unified perspective is essential to improving public health. The perniciousness of this epidemic requires a multipronged interventional approach that engages all sectors of society,” the investigators wrote.
Dr. Compton has ties with General Electric, 3M, and Pfizer. No other conflicts were reported. Find the study in the New England Journal of Medicine (doi: 10.1056/NEJMra1508490).
The increase in nonmedical prescription opioid use in the United States does not appear to be strongly related to the concurrent increase in heroin use, according to a review by Dr. Wilson Compton of the National Institute on Drug Abuse, Bethesda, Md., and his associates.
While heroin users are 3.9 times more likely to have used nonmedical prescription opioids than are those who haven’t used heroin, heroin use only occurs in a small number of nonmedical prescription opioid users. The researchers cited studies showing that 3.6% of opioid users began using heroin within 5 years of beginning opioid use, and 4.2% of opioid users reported also using heroin in the past year.
A more likely driver for the increased use of heroin and heroin death rate is decreased cost and increased availability, the investigators wrote. For every $100 decrease in price per gram of heroin, hospitalizations for heroin overdose increase by 2.9%. In addition, heroin use has grown significantly in areas of the United States that were not typically centers for heroin distribution, the researchers reported.
“Fundamentally, prescription opioids and heroin are each elements of a larger epidemic of opioid-related disorders and death. Viewing them from a unified perspective is essential to improving public health. The perniciousness of this epidemic requires a multipronged interventional approach that engages all sectors of society,” the investigators wrote.
Dr. Compton has ties with General Electric, 3M, and Pfizer. No other conflicts were reported. Find the study in the New England Journal of Medicine (doi: 10.1056/NEJMra1508490).
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Increased heroin use may not be linked to rise in prescription opioid use
The increase in nonmedical prescription opioid use in the United States does not appear to be strongly related to the concurrent increase in heroin use, according to a review by Dr. Wilson Compton of the National Institute on Drug Abuse, Bethesda, Md., and his associates.
While heroin users are 3.9 times more likely to have used nonmedical prescription opioids than are those who haven’t used heroin, heroin use does occur in a small number of nonmedical prescription opioid users. The researchers cited studies showing that 3.6% of opioid users began using heroin within 5 years of beginning opioid use, and 4.2% of opioid users reported also using heroin in the past year.
A more likely driver for the increased use of heroin and heroin death rate is decreased cost and increased availability, the investigators wrote. For every $100 decrease in price per gram of heroin, hospitalizations for heroin overdose increase by 2.9%. In addition, heroin use has grown significantly in areas of the United States that were not typically centers for heroin distribution, the researchers reported.
“Fundamentally, prescription opioids and heroin are each elements of a larger epidemic of opioid-related disorders and death. Viewing them from a unified perspective is essential to improving public health. The perniciousness of this epidemic requires a multipronged interventional approach that engages all sectors of society,” the investigators wrote.
Dr. Compton has ties with General Electric, 3M, and Pfizer. No other conflicts were reported. Find the study in the New England Journal of Medicine (doi: 10.1056/NEJMra1508490).
The increase in nonmedical prescription opioid use in the United States does not appear to be strongly related to the concurrent increase in heroin use, according to a review by Dr. Wilson Compton of the National Institute on Drug Abuse, Bethesda, Md., and his associates.
While heroin users are 3.9 times more likely to have used nonmedical prescription opioids than are those who haven’t used heroin, heroin use does occur in a small number of nonmedical prescription opioid users. The researchers cited studies showing that 3.6% of opioid users began using heroin within 5 years of beginning opioid use, and 4.2% of opioid users reported also using heroin in the past year.
A more likely driver for the increased use of heroin and heroin death rate is decreased cost and increased availability, the investigators wrote. For every $100 decrease in price per gram of heroin, hospitalizations for heroin overdose increase by 2.9%. In addition, heroin use has grown significantly in areas of the United States that were not typically centers for heroin distribution, the researchers reported.
“Fundamentally, prescription opioids and heroin are each elements of a larger epidemic of opioid-related disorders and death. Viewing them from a unified perspective is essential to improving public health. The perniciousness of this epidemic requires a multipronged interventional approach that engages all sectors of society,” the investigators wrote.
Dr. Compton has ties with General Electric, 3M, and Pfizer. No other conflicts were reported. Find the study in the New England Journal of Medicine (doi: 10.1056/NEJMra1508490).
The increase in nonmedical prescription opioid use in the United States does not appear to be strongly related to the concurrent increase in heroin use, according to a review by Dr. Wilson Compton of the National Institute on Drug Abuse, Bethesda, Md., and his associates.
While heroin users are 3.9 times more likely to have used nonmedical prescription opioids than are those who haven’t used heroin, heroin use does occur in a small number of nonmedical prescription opioid users. The researchers cited studies showing that 3.6% of opioid users began using heroin within 5 years of beginning opioid use, and 4.2% of opioid users reported also using heroin in the past year.
A more likely driver for the increased use of heroin and heroin death rate is decreased cost and increased availability, the investigators wrote. For every $100 decrease in price per gram of heroin, hospitalizations for heroin overdose increase by 2.9%. In addition, heroin use has grown significantly in areas of the United States that were not typically centers for heroin distribution, the researchers reported.
“Fundamentally, prescription opioids and heroin are each elements of a larger epidemic of opioid-related disorders and death. Viewing them from a unified perspective is essential to improving public health. The perniciousness of this epidemic requires a multipronged interventional approach that engages all sectors of society,” the investigators wrote.
Dr. Compton has ties with General Electric, 3M, and Pfizer. No other conflicts were reported. Find the study in the New England Journal of Medicine (doi: 10.1056/NEJMra1508490).
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Reviewing the new topical treatments for onychomycosis
Dr. Lorraine L. Rosamilia of the department of dermatology at Geisinger Health System in State College, Pa., shared her views on new data regarding topical treatments for onychomycosis with Dermatology News’ sister publication, Cutis, remarking, “Do you agree that nary a day goes by without an e-mailed article or continuing medical education opportunity tasked at ‘getting to know’ new topical onychomycosis therapies? That being said, how often have you summarily deleted them, assuming that topicals just don’t work? I know I have.” But these treatments deserve some attention, she said, based on her review of an article published in the Journal of Clinical and Aesthetic Dermatology (2014;7:10-8). Based on those data, she said, “perhaps these medications, particularly efinaconazole, at least deserve perusal compared to our current meager topical and systemic options.”
Dr. Rosamilia had no financial conflicts to disclose.
Dr. Lorraine L. Rosamilia of the department of dermatology at Geisinger Health System in State College, Pa., shared her views on new data regarding topical treatments for onychomycosis with Dermatology News’ sister publication, Cutis, remarking, “Do you agree that nary a day goes by without an e-mailed article or continuing medical education opportunity tasked at ‘getting to know’ new topical onychomycosis therapies? That being said, how often have you summarily deleted them, assuming that topicals just don’t work? I know I have.” But these treatments deserve some attention, she said, based on her review of an article published in the Journal of Clinical and Aesthetic Dermatology (2014;7:10-8). Based on those data, she said, “perhaps these medications, particularly efinaconazole, at least deserve perusal compared to our current meager topical and systemic options.”
Dr. Rosamilia had no financial conflicts to disclose.
Dr. Lorraine L. Rosamilia of the department of dermatology at Geisinger Health System in State College, Pa., shared her views on new data regarding topical treatments for onychomycosis with Dermatology News’ sister publication, Cutis, remarking, “Do you agree that nary a day goes by without an e-mailed article or continuing medical education opportunity tasked at ‘getting to know’ new topical onychomycosis therapies? That being said, how often have you summarily deleted them, assuming that topicals just don’t work? I know I have.” But these treatments deserve some attention, she said, based on her review of an article published in the Journal of Clinical and Aesthetic Dermatology (2014;7:10-8). Based on those data, she said, “perhaps these medications, particularly efinaconazole, at least deserve perusal compared to our current meager topical and systemic options.”
Dr. Rosamilia had no financial conflicts to disclose.