User login
Lucas Franki is an associate editor for MDedge News, and has been with the company since 2014. He has a BA in English from Penn State University and is an Eagle Scout.
Interviews with psoriasis patients reveal disease hardships
Interviewing patients who live with psoriasis can reveal important information not included in psoriasis severity measures, according to Dr. David Pariser of Eastern Virginia Medical School, Norfolk, and his associates.
More than 90% of 101 patients with moderate to severe plaque psoriasis, with and without psoriatic arthritis, reported emotional and social impacts to their lives. Social impacts included avoiding/changing activities, relationships, or trying new things. Emotional impacts included a sense of anger, frustration, embarrassment, and self-consciousness; a lowered sense of self-esteem was common. Patients reported impacts in family, professional, social, and educational areas.
Patients with psoriasis and psoriatic arthritis tended to report both symptoms and well-being complications more often, with 77% reporting physical impacts and 34% reporting educational impacts, compared with 38% and 17%, respectively, for those with psoriasis only.
The study data “provide practitioners with deep insight into how patients with moderate to severe psoriasis are suffering, regardless of whether the concerns are elucidated during patient visits. The burdens in patients’ lives support the use of effective treatment; indeed, patients expressed how they value physicians who are knowledgeable about psoriasis and who will try various therapies,” the investigators concluded.
Find the full study in the Journal of Dermatological Treatment (doi: 10.3109/09546634.2015.1044492).
Interviewing patients who live with psoriasis can reveal important information not included in psoriasis severity measures, according to Dr. David Pariser of Eastern Virginia Medical School, Norfolk, and his associates.
More than 90% of 101 patients with moderate to severe plaque psoriasis, with and without psoriatic arthritis, reported emotional and social impacts to their lives. Social impacts included avoiding/changing activities, relationships, or trying new things. Emotional impacts included a sense of anger, frustration, embarrassment, and self-consciousness; a lowered sense of self-esteem was common. Patients reported impacts in family, professional, social, and educational areas.
Patients with psoriasis and psoriatic arthritis tended to report both symptoms and well-being complications more often, with 77% reporting physical impacts and 34% reporting educational impacts, compared with 38% and 17%, respectively, for those with psoriasis only.
The study data “provide practitioners with deep insight into how patients with moderate to severe psoriasis are suffering, regardless of whether the concerns are elucidated during patient visits. The burdens in patients’ lives support the use of effective treatment; indeed, patients expressed how they value physicians who are knowledgeable about psoriasis and who will try various therapies,” the investigators concluded.
Find the full study in the Journal of Dermatological Treatment (doi: 10.3109/09546634.2015.1044492).
Interviewing patients who live with psoriasis can reveal important information not included in psoriasis severity measures, according to Dr. David Pariser of Eastern Virginia Medical School, Norfolk, and his associates.
More than 90% of 101 patients with moderate to severe plaque psoriasis, with and without psoriatic arthritis, reported emotional and social impacts to their lives. Social impacts included avoiding/changing activities, relationships, or trying new things. Emotional impacts included a sense of anger, frustration, embarrassment, and self-consciousness; a lowered sense of self-esteem was common. Patients reported impacts in family, professional, social, and educational areas.
Patients with psoriasis and psoriatic arthritis tended to report both symptoms and well-being complications more often, with 77% reporting physical impacts and 34% reporting educational impacts, compared with 38% and 17%, respectively, for those with psoriasis only.
The study data “provide practitioners with deep insight into how patients with moderate to severe psoriasis are suffering, regardless of whether the concerns are elucidated during patient visits. The burdens in patients’ lives support the use of effective treatment; indeed, patients expressed how they value physicians who are knowledgeable about psoriasis and who will try various therapies,” the investigators concluded.
Find the full study in the Journal of Dermatological Treatment (doi: 10.3109/09546634.2015.1044492).
High-calorie American Diet Causes Insulin Resistance Within Days
A very-high-calorie version of the typical United States diet caused rapid weight gain, along with other adverse side effects, according to Dr. Guenther Boden and his associates.
The study group of six overweight but otherwise healthy men was fed a roughly 6,000-calorie diet representing the average U.S. diet, composed of 50% carbohydrates, 35% protein, and 15% fat. After consuming this high-calorie diet for 1 week, participants gained an average of 3.5 kg. It took only 2-3 days before subjects developed systemic and adipose insulin resistance and oxidative stress. No inflammatory or endoplasmic reticulum stress was found.
Oxidative stress was linked to extensive oxidation and carbonylation of various proteins within adipose tissue, including carbonylation and loss of glucose transporter type 4 (GLUT4) activity. “The observation of carbonylation in proximity to the GLUT4 glucose transport channel strongly suggested that GLUT4 had become dysfunctional, resulting in insulin resistance and providing a likely causal link between overnutrition and insulin resistance,” the investigators noted.
Find the full study in Science Translational Medicine (doi:10.1126/scitranslmed.aac4765).
A very-high-calorie version of the typical United States diet caused rapid weight gain, along with other adverse side effects, according to Dr. Guenther Boden and his associates.
The study group of six overweight but otherwise healthy men was fed a roughly 6,000-calorie diet representing the average U.S. diet, composed of 50% carbohydrates, 35% protein, and 15% fat. After consuming this high-calorie diet for 1 week, participants gained an average of 3.5 kg. It took only 2-3 days before subjects developed systemic and adipose insulin resistance and oxidative stress. No inflammatory or endoplasmic reticulum stress was found.
Oxidative stress was linked to extensive oxidation and carbonylation of various proteins within adipose tissue, including carbonylation and loss of glucose transporter type 4 (GLUT4) activity. “The observation of carbonylation in proximity to the GLUT4 glucose transport channel strongly suggested that GLUT4 had become dysfunctional, resulting in insulin resistance and providing a likely causal link between overnutrition and insulin resistance,” the investigators noted.
Find the full study in Science Translational Medicine (doi:10.1126/scitranslmed.aac4765).
A very-high-calorie version of the typical United States diet caused rapid weight gain, along with other adverse side effects, according to Dr. Guenther Boden and his associates.
The study group of six overweight but otherwise healthy men was fed a roughly 6,000-calorie diet representing the average U.S. diet, composed of 50% carbohydrates, 35% protein, and 15% fat. After consuming this high-calorie diet for 1 week, participants gained an average of 3.5 kg. It took only 2-3 days before subjects developed systemic and adipose insulin resistance and oxidative stress. No inflammatory or endoplasmic reticulum stress was found.
Oxidative stress was linked to extensive oxidation and carbonylation of various proteins within adipose tissue, including carbonylation and loss of glucose transporter type 4 (GLUT4) activity. “The observation of carbonylation in proximity to the GLUT4 glucose transport channel strongly suggested that GLUT4 had become dysfunctional, resulting in insulin resistance and providing a likely causal link between overnutrition and insulin resistance,” the investigators noted.
Find the full study in Science Translational Medicine (doi:10.1126/scitranslmed.aac4765).
High-calorie American diet causes insulin resistance within days
A very-high-calorie version of the typical United States diet caused rapid weight gain, along with other adverse side effects, according to Dr. Guenther Boden and his associates.
The study group of six overweight but otherwise healthy men was fed a roughly 6,000-calorie diet representing the average U.S. diet, composed of 50% carbohydrates, 35% protein, and 15% fat. After consuming this high-calorie diet for 1 week, participants gained an average of 3.5 kg. It took only 2-3 days before subjects developed systemic and adipose insulin resistance and oxidative stress. No inflammatory or endoplasmic reticulum stress was found.
Oxidative stress was linked to extensive oxidation and carbonylation of various proteins within adipose tissue, including carbonylation and loss of glucose transporter type 4 (GLUT4) activity. “The observation of carbonylation in proximity to the GLUT4 glucose transport channel strongly suggested that GLUT4 had become dysfunctional, resulting in insulin resistance and providing a likely causal link between overnutrition and insulin resistance,” the investigators noted.
Find the full study in Science Translational Medicine (doi:10.1126/scitranslmed.aac4765).
A very-high-calorie version of the typical United States diet caused rapid weight gain, along with other adverse side effects, according to Dr. Guenther Boden and his associates.
The study group of six overweight but otherwise healthy men was fed a roughly 6,000-calorie diet representing the average U.S. diet, composed of 50% carbohydrates, 35% protein, and 15% fat. After consuming this high-calorie diet for 1 week, participants gained an average of 3.5 kg. It took only 2-3 days before subjects developed systemic and adipose insulin resistance and oxidative stress. No inflammatory or endoplasmic reticulum stress was found.
Oxidative stress was linked to extensive oxidation and carbonylation of various proteins within adipose tissue, including carbonylation and loss of glucose transporter type 4 (GLUT4) activity. “The observation of carbonylation in proximity to the GLUT4 glucose transport channel strongly suggested that GLUT4 had become dysfunctional, resulting in insulin resistance and providing a likely causal link between overnutrition and insulin resistance,” the investigators noted.
Find the full study in Science Translational Medicine (doi:10.1126/scitranslmed.aac4765).
A very-high-calorie version of the typical United States diet caused rapid weight gain, along with other adverse side effects, according to Dr. Guenther Boden and his associates.
The study group of six overweight but otherwise healthy men was fed a roughly 6,000-calorie diet representing the average U.S. diet, composed of 50% carbohydrates, 35% protein, and 15% fat. After consuming this high-calorie diet for 1 week, participants gained an average of 3.5 kg. It took only 2-3 days before subjects developed systemic and adipose insulin resistance and oxidative stress. No inflammatory or endoplasmic reticulum stress was found.
Oxidative stress was linked to extensive oxidation and carbonylation of various proteins within adipose tissue, including carbonylation and loss of glucose transporter type 4 (GLUT4) activity. “The observation of carbonylation in proximity to the GLUT4 glucose transport channel strongly suggested that GLUT4 had become dysfunctional, resulting in insulin resistance and providing a likely causal link between overnutrition and insulin resistance,” the investigators noted.
Find the full study in Science Translational Medicine (doi:10.1126/scitranslmed.aac4765).
JCO publishes special issue on head and neck cancers
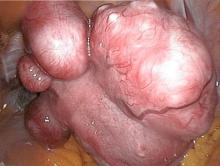
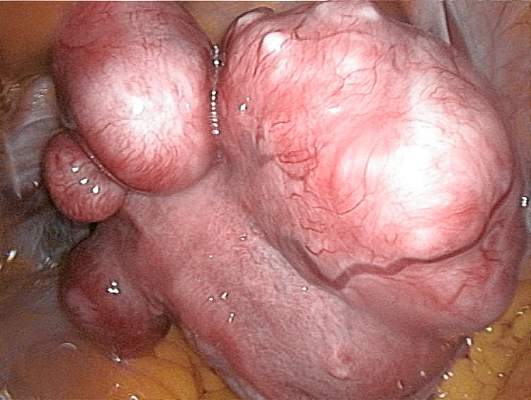
The Journal of Clinical Oncology has published a special series issue on head and neck cancers, with a major focus on squamous cell carcinomas arising from the upper aerodigestive tract mucosa.
“In addition, we also address nasopharyngeal cancer, nonmelanoma cutaneous head and neck cancer, and squamous carcinoma of the neck of unknown primary origin,” writes Dr. Danny Rischin of the Peter MacCallum Cancer Centre and University of Melbourne, Victoria, Australia, and associates in an overview for the issue.
The 16 articles in the issue provide the reader with an overview of current evidence and management, recent developments, and insights into future directions, they write.
It was only recently that human papillomavirus was identified as a cause of oropharyngeal cancer and that HPV-associated oropharyngeal cancer was found to have its own unique biologic signature. Three articles are included in the series that review the rapidly evolving understanding of HPV-related head and neck cancers.
Find the overview and series of 16 articles in the Sept. 7 issue of the Journal of Clinical Oncology.
The Journal of Clinical Oncology has published a special series issue on head and neck cancers, with a major focus on squamous cell carcinomas arising from the upper aerodigestive tract mucosa.
“In addition, we also address nasopharyngeal cancer, nonmelanoma cutaneous head and neck cancer, and squamous carcinoma of the neck of unknown primary origin,” writes Dr. Danny Rischin of the Peter MacCallum Cancer Centre and University of Melbourne, Victoria, Australia, and associates in an overview for the issue.
The 16 articles in the issue provide the reader with an overview of current evidence and management, recent developments, and insights into future directions, they write.
It was only recently that human papillomavirus was identified as a cause of oropharyngeal cancer and that HPV-associated oropharyngeal cancer was found to have its own unique biologic signature. Three articles are included in the series that review the rapidly evolving understanding of HPV-related head and neck cancers.
Find the overview and series of 16 articles in the Sept. 7 issue of the Journal of Clinical Oncology.
The Journal of Clinical Oncology has published a special series issue on head and neck cancers, with a major focus on squamous cell carcinomas arising from the upper aerodigestive tract mucosa.
“In addition, we also address nasopharyngeal cancer, nonmelanoma cutaneous head and neck cancer, and squamous carcinoma of the neck of unknown primary origin,” writes Dr. Danny Rischin of the Peter MacCallum Cancer Centre and University of Melbourne, Victoria, Australia, and associates in an overview for the issue.
The 16 articles in the issue provide the reader with an overview of current evidence and management, recent developments, and insights into future directions, they write.
It was only recently that human papillomavirus was identified as a cause of oropharyngeal cancer and that HPV-associated oropharyngeal cancer was found to have its own unique biologic signature. Three articles are included in the series that review the rapidly evolving understanding of HPV-related head and neck cancers.
Find the overview and series of 16 articles in the Sept. 7 issue of the Journal of Clinical Oncology.
Prescription Opioid Overdoses Targeted in New CDC Program
The Centers for Disease Control and Prevention has launched a program aimed at helping states combat and prevent opioid drug overdoses.
The Prescription Drug Overdose: Prevention for States program will be launching in 16 states chosen in a competitive application process. The CDC is committing $20 million in fiscal year 2015, and each state will receive $750,000 to $1 million each year for the next 4 years to advance prevention in several areas, such as enhancing prescription drug–monitoring programs, putting prevention into action in communities nationwide, and investigating the connection between prescription opioid abuse and heroin use, the CDC said in a press release.
In 2013, 16,000 people died from prescription opioid overdoses, four times more than in 1999, with prescription of opioids increasing at the same rate over the same time. Despite more opioids being prescribed, the amount of pain Americans report has not changed. In addition, heroin deaths also have spiked, with the 8,000 heroin overdose deaths nearly three times as many as in 2010.
“The prescription drug overdose epidemic requires a multifaceted approach, and states are key partners in our efforts on the front lines to prevent overdose deaths. With this funding, states can improve their ability to track the problem, work with insurers to help providers make informed prescribing decisions, and take action to combat this epidemic,” U.S. Department of Health & Human Services Secretary Sylvia M. Burwell said in the release.
Find the full CDC press release here.
The Centers for Disease Control and Prevention has launched a program aimed at helping states combat and prevent opioid drug overdoses.
The Prescription Drug Overdose: Prevention for States program will be launching in 16 states chosen in a competitive application process. The CDC is committing $20 million in fiscal year 2015, and each state will receive $750,000 to $1 million each year for the next 4 years to advance prevention in several areas, such as enhancing prescription drug–monitoring programs, putting prevention into action in communities nationwide, and investigating the connection between prescription opioid abuse and heroin use, the CDC said in a press release.
In 2013, 16,000 people died from prescription opioid overdoses, four times more than in 1999, with prescription of opioids increasing at the same rate over the same time. Despite more opioids being prescribed, the amount of pain Americans report has not changed. In addition, heroin deaths also have spiked, with the 8,000 heroin overdose deaths nearly three times as many as in 2010.
“The prescription drug overdose epidemic requires a multifaceted approach, and states are key partners in our efforts on the front lines to prevent overdose deaths. With this funding, states can improve their ability to track the problem, work with insurers to help providers make informed prescribing decisions, and take action to combat this epidemic,” U.S. Department of Health & Human Services Secretary Sylvia M. Burwell said in the release.
Find the full CDC press release here.
The Centers for Disease Control and Prevention has launched a program aimed at helping states combat and prevent opioid drug overdoses.
The Prescription Drug Overdose: Prevention for States program will be launching in 16 states chosen in a competitive application process. The CDC is committing $20 million in fiscal year 2015, and each state will receive $750,000 to $1 million each year for the next 4 years to advance prevention in several areas, such as enhancing prescription drug–monitoring programs, putting prevention into action in communities nationwide, and investigating the connection between prescription opioid abuse and heroin use, the CDC said in a press release.
In 2013, 16,000 people died from prescription opioid overdoses, four times more than in 1999, with prescription of opioids increasing at the same rate over the same time. Despite more opioids being prescribed, the amount of pain Americans report has not changed. In addition, heroin deaths also have spiked, with the 8,000 heroin overdose deaths nearly three times as many as in 2010.
“The prescription drug overdose epidemic requires a multifaceted approach, and states are key partners in our efforts on the front lines to prevent overdose deaths. With this funding, states can improve their ability to track the problem, work with insurers to help providers make informed prescribing decisions, and take action to combat this epidemic,” U.S. Department of Health & Human Services Secretary Sylvia M. Burwell said in the release.
Find the full CDC press release here.
Prescription opioid overdoses targeted in new CDC program
The Centers for Disease Control and Prevention has launched a program aimed at helping states combat and prevent opioid drug overdoses.
The Prescription Drug Overdose: Prevention for States program will be launching in 16 states chosen in a competitive application process. The CDC is committing $20 million in fiscal year 2015, and each state will receive $750,000 to $1 million each year for the next 4 years to advance prevention in several areas, such as enhancing prescription drug–monitoring programs, putting prevention into action in communities nationwide, and investigating the connection between prescription opioid abuse and heroin use, the CDC said in a press release.
In 2013, 16,000 people died from prescription opioid overdoses, four times more than in 1999, with prescription of opioids increasing at the same rate over the same time. Despite more opioids being prescribed, the amount of pain Americans report has not changed. In addition, heroin deaths also have spiked, with the 8,000 heroin overdose deaths nearly three times as many as in 2010.
“The prescription drug overdose epidemic requires a multifaceted approach, and states are key partners in our efforts on the front lines to prevent overdose deaths. With this funding, states can improve their ability to track the problem, work with insurers to help providers make informed prescribing decisions, and take action to combat this epidemic,” U.S. Department of Health & Human Services Secretary Sylvia M. Burwell said in the release.
Find the full CDC press release here.
The Centers for Disease Control and Prevention has launched a program aimed at helping states combat and prevent opioid drug overdoses.
The Prescription Drug Overdose: Prevention for States program will be launching in 16 states chosen in a competitive application process. The CDC is committing $20 million in fiscal year 2015, and each state will receive $750,000 to $1 million each year for the next 4 years to advance prevention in several areas, such as enhancing prescription drug–monitoring programs, putting prevention into action in communities nationwide, and investigating the connection between prescription opioid abuse and heroin use, the CDC said in a press release.
In 2013, 16,000 people died from prescription opioid overdoses, four times more than in 1999, with prescription of opioids increasing at the same rate over the same time. Despite more opioids being prescribed, the amount of pain Americans report has not changed. In addition, heroin deaths also have spiked, with the 8,000 heroin overdose deaths nearly three times as many as in 2010.
“The prescription drug overdose epidemic requires a multifaceted approach, and states are key partners in our efforts on the front lines to prevent overdose deaths. With this funding, states can improve their ability to track the problem, work with insurers to help providers make informed prescribing decisions, and take action to combat this epidemic,” U.S. Department of Health & Human Services Secretary Sylvia M. Burwell said in the release.
Find the full CDC press release here.
The Centers for Disease Control and Prevention has launched a program aimed at helping states combat and prevent opioid drug overdoses.
The Prescription Drug Overdose: Prevention for States program will be launching in 16 states chosen in a competitive application process. The CDC is committing $20 million in fiscal year 2015, and each state will receive $750,000 to $1 million each year for the next 4 years to advance prevention in several areas, such as enhancing prescription drug–monitoring programs, putting prevention into action in communities nationwide, and investigating the connection between prescription opioid abuse and heroin use, the CDC said in a press release.
In 2013, 16,000 people died from prescription opioid overdoses, four times more than in 1999, with prescription of opioids increasing at the same rate over the same time. Despite more opioids being prescribed, the amount of pain Americans report has not changed. In addition, heroin deaths also have spiked, with the 8,000 heroin overdose deaths nearly three times as many as in 2010.
“The prescription drug overdose epidemic requires a multifaceted approach, and states are key partners in our efforts on the front lines to prevent overdose deaths. With this funding, states can improve their ability to track the problem, work with insurers to help providers make informed prescribing decisions, and take action to combat this epidemic,” U.S. Department of Health & Human Services Secretary Sylvia M. Burwell said in the release.
Find the full CDC press release here.
Repeat uterine preservation procedures likely for women with fibroids
Women who undergo a uterus-preserving procedure to manage uterine fibroids often have to have further procedures, according to Dr. Elisa Martin-Merino of the Spanish Centre for Pharmacoepidemiologic Research in Madrid and her associates.
After 1 year, 23.6% of women aged 15-54 with uterine fibroids had a hysterectomy or uterus-preserving procedure (UPP), with the rate increasing to 40.9% at the end of the follow-up period, which was a median of 3.6 years. About one-third of all study participants underwent a hysterectomy during the study period. Myomectomy, endometrial ablation, and uterine artery embolization were much less common, performed in 3.9%, 6.4%, and 1.9% of study participants respectively.
The incidence of a repeat UPP after being initially treated with a UPP was 11.5% at 1 year, and 26.1% at the end of the study period. The cumulative incidences of women undergoing hysterectomy, myomectomy, endometrial ablation, and uterine artery embolization were 19.0%, 4.3%, 3.4%, and 1.4%, respectively.
“Women considering UPPs for the management of [uterine fibroids] should be made aware that the incidence of further treatments is high, with hysterectomy being the most frequent procedure undergone,” the investigators wrote.
Find the full study in the European Journal of Obstetrics & Gynecology and Reproductive Biology (doi: 10.1016/j.ejogrb.2015.08.034).
Women who undergo a uterus-preserving procedure to manage uterine fibroids often have to have further procedures, according to Dr. Elisa Martin-Merino of the Spanish Centre for Pharmacoepidemiologic Research in Madrid and her associates.
After 1 year, 23.6% of women aged 15-54 with uterine fibroids had a hysterectomy or uterus-preserving procedure (UPP), with the rate increasing to 40.9% at the end of the follow-up period, which was a median of 3.6 years. About one-third of all study participants underwent a hysterectomy during the study period. Myomectomy, endometrial ablation, and uterine artery embolization were much less common, performed in 3.9%, 6.4%, and 1.9% of study participants respectively.
The incidence of a repeat UPP after being initially treated with a UPP was 11.5% at 1 year, and 26.1% at the end of the study period. The cumulative incidences of women undergoing hysterectomy, myomectomy, endometrial ablation, and uterine artery embolization were 19.0%, 4.3%, 3.4%, and 1.4%, respectively.
“Women considering UPPs for the management of [uterine fibroids] should be made aware that the incidence of further treatments is high, with hysterectomy being the most frequent procedure undergone,” the investigators wrote.
Find the full study in the European Journal of Obstetrics & Gynecology and Reproductive Biology (doi: 10.1016/j.ejogrb.2015.08.034).
Women who undergo a uterus-preserving procedure to manage uterine fibroids often have to have further procedures, according to Dr. Elisa Martin-Merino of the Spanish Centre for Pharmacoepidemiologic Research in Madrid and her associates.
After 1 year, 23.6% of women aged 15-54 with uterine fibroids had a hysterectomy or uterus-preserving procedure (UPP), with the rate increasing to 40.9% at the end of the follow-up period, which was a median of 3.6 years. About one-third of all study participants underwent a hysterectomy during the study period. Myomectomy, endometrial ablation, and uterine artery embolization were much less common, performed in 3.9%, 6.4%, and 1.9% of study participants respectively.
The incidence of a repeat UPP after being initially treated with a UPP was 11.5% at 1 year, and 26.1% at the end of the study period. The cumulative incidences of women undergoing hysterectomy, myomectomy, endometrial ablation, and uterine artery embolization were 19.0%, 4.3%, 3.4%, and 1.4%, respectively.
“Women considering UPPs for the management of [uterine fibroids] should be made aware that the incidence of further treatments is high, with hysterectomy being the most frequent procedure undergone,” the investigators wrote.
Find the full study in the European Journal of Obstetrics & Gynecology and Reproductive Biology (doi: 10.1016/j.ejogrb.2015.08.034).
Liberia officially Ebola free for second time
Liberia was declared free of Ebola by the World Health Organization for the second time on Sept. 3, according to a report from the Centers for Disease Control and Prevention.
Ebola was first detected in Liberia in late March 2014, and since that time the country has reported 5,036 confirmed or probable cases of Ebola, and more than 4,800 deaths. Case incidence reached its highest point in September and October 2014 and began to fall dramatically by December. On May 9, 2015, the country was declared Ebola free.
The disease reentered Liberia on June 29, but through rapid response and active case tracking, the new outbreak was contained within a month. Only 6 confirmed and 2 probable cases were identified, and 143 contacts were identified and monitored. On July 23, the last patient with Ebola was discharged, and after the standard 42-day waiting period, Liberia is once again officially Ebola free.
“As Liberia transitions again from an emergency public health response to a phase of continued vigilance, many of the practices that have been put into place will, in addition to ensuring continued heightened surveillance for Ebola, facilitate the overall rebuilding of the country’s public health infrastructure,” the CDC investigators concluded.
Find the full report in the MMWR (Sept. 3, 2015/Vol. 64).
Liberia was declared free of Ebola by the World Health Organization for the second time on Sept. 3, according to a report from the Centers for Disease Control and Prevention.
Ebola was first detected in Liberia in late March 2014, and since that time the country has reported 5,036 confirmed or probable cases of Ebola, and more than 4,800 deaths. Case incidence reached its highest point in September and October 2014 and began to fall dramatically by December. On May 9, 2015, the country was declared Ebola free.
The disease reentered Liberia on June 29, but through rapid response and active case tracking, the new outbreak was contained within a month. Only 6 confirmed and 2 probable cases were identified, and 143 contacts were identified and monitored. On July 23, the last patient with Ebola was discharged, and after the standard 42-day waiting period, Liberia is once again officially Ebola free.
“As Liberia transitions again from an emergency public health response to a phase of continued vigilance, many of the practices that have been put into place will, in addition to ensuring continued heightened surveillance for Ebola, facilitate the overall rebuilding of the country’s public health infrastructure,” the CDC investigators concluded.
Find the full report in the MMWR (Sept. 3, 2015/Vol. 64).
Liberia was declared free of Ebola by the World Health Organization for the second time on Sept. 3, according to a report from the Centers for Disease Control and Prevention.
Ebola was first detected in Liberia in late March 2014, and since that time the country has reported 5,036 confirmed or probable cases of Ebola, and more than 4,800 deaths. Case incidence reached its highest point in September and October 2014 and began to fall dramatically by December. On May 9, 2015, the country was declared Ebola free.
The disease reentered Liberia on June 29, but through rapid response and active case tracking, the new outbreak was contained within a month. Only 6 confirmed and 2 probable cases were identified, and 143 contacts were identified and monitored. On July 23, the last patient with Ebola was discharged, and after the standard 42-day waiting period, Liberia is once again officially Ebola free.
“As Liberia transitions again from an emergency public health response to a phase of continued vigilance, many of the practices that have been put into place will, in addition to ensuring continued heightened surveillance for Ebola, facilitate the overall rebuilding of the country’s public health infrastructure,” the CDC investigators concluded.
Find the full report in the MMWR (Sept. 3, 2015/Vol. 64).
FROM THE MMWR
Skin-lightening Products May Not Be Safe, FDA Warns
Use of an injectable skin-lightening product poses a potential health risk to consumers, as no such product has been approved, according to a consumer alert from the Food and Drug Administration.
Injectable skin products – sold online or in some retail locations such as health spas – can contain a wide range of ingredients, such as glutathione, vitamin C, collagen, or human placenta. They are advertised as being able to lighten skin, correct uneven skin tone, and clear blemishes. More exaggerated claims include the ability to treat Parkinson’s disease and liver disorders.
Although makers and distributors of unlicensed skin-lightening products have been subject to legal action and recalls in the past, the products continue to be marketed. Also of concern are noninjectable, over-the-counter skin-bleaching products that contain ammoniated mercury.
“In general, consumers should be cautious of any product marketed online with exaggerated claims on safety and effectiveness. They also should consult their health care practitioner before deciding to use any new product,” In Kim, an FDA pharmacist, said in a written statement.
Find the full statement on the FDA website here.
Use of an injectable skin-lightening product poses a potential health risk to consumers, as no such product has been approved, according to a consumer alert from the Food and Drug Administration.
Injectable skin products – sold online or in some retail locations such as health spas – can contain a wide range of ingredients, such as glutathione, vitamin C, collagen, or human placenta. They are advertised as being able to lighten skin, correct uneven skin tone, and clear blemishes. More exaggerated claims include the ability to treat Parkinson’s disease and liver disorders.
Although makers and distributors of unlicensed skin-lightening products have been subject to legal action and recalls in the past, the products continue to be marketed. Also of concern are noninjectable, over-the-counter skin-bleaching products that contain ammoniated mercury.
“In general, consumers should be cautious of any product marketed online with exaggerated claims on safety and effectiveness. They also should consult their health care practitioner before deciding to use any new product,” In Kim, an FDA pharmacist, said in a written statement.
Find the full statement on the FDA website here.
Use of an injectable skin-lightening product poses a potential health risk to consumers, as no such product has been approved, according to a consumer alert from the Food and Drug Administration.
Injectable skin products – sold online or in some retail locations such as health spas – can contain a wide range of ingredients, such as glutathione, vitamin C, collagen, or human placenta. They are advertised as being able to lighten skin, correct uneven skin tone, and clear blemishes. More exaggerated claims include the ability to treat Parkinson’s disease and liver disorders.
Although makers and distributors of unlicensed skin-lightening products have been subject to legal action and recalls in the past, the products continue to be marketed. Also of concern are noninjectable, over-the-counter skin-bleaching products that contain ammoniated mercury.
“In general, consumers should be cautious of any product marketed online with exaggerated claims on safety and effectiveness. They also should consult their health care practitioner before deciding to use any new product,” In Kim, an FDA pharmacist, said in a written statement.
Find the full statement on the FDA website here.
Skin-lightening products may not be safe, FDA warns
Use of an injectable skin-lightening product poses a potential health risk to consumers, as no such product has been approved, according to a consumer alert from the Food and Drug Administration.
Injectable skin products – sold online or in some retail locations such as health spas – can contain a wide range of ingredients, such as glutathione, vitamin C, collagen, or human placenta. They are advertised as being able to lighten skin, correct uneven skin tone, and clear blemishes. More exaggerated claims include the ability to treat Parkinson’s disease and liver disorders.
Although makers and distributors of unlicensed skin-lightening products have been subject to legal action and recalls in the past, the products continue to be marketed. Also of concern are noninjectable, over-the-counter skin-bleaching products that contain ammoniated mercury.
“In general, consumers should be cautious of any product marketed online with exaggerated claims on safety and effectiveness. They also should consult their health care practitioner before deciding to use any new product,” In Kim, an FDA pharmacist, said in a written statement.
Find the full statement on the FDA website here.
Use of an injectable skin-lightening product poses a potential health risk to consumers, as no such product has been approved, according to a consumer alert from the Food and Drug Administration.
Injectable skin products – sold online or in some retail locations such as health spas – can contain a wide range of ingredients, such as glutathione, vitamin C, collagen, or human placenta. They are advertised as being able to lighten skin, correct uneven skin tone, and clear blemishes. More exaggerated claims include the ability to treat Parkinson’s disease and liver disorders.
Although makers and distributors of unlicensed skin-lightening products have been subject to legal action and recalls in the past, the products continue to be marketed. Also of concern are noninjectable, over-the-counter skin-bleaching products that contain ammoniated mercury.
“In general, consumers should be cautious of any product marketed online with exaggerated claims on safety and effectiveness. They also should consult their health care practitioner before deciding to use any new product,” In Kim, an FDA pharmacist, said in a written statement.
Find the full statement on the FDA website here.
Use of an injectable skin-lightening product poses a potential health risk to consumers, as no such product has been approved, according to a consumer alert from the Food and Drug Administration.
Injectable skin products – sold online or in some retail locations such as health spas – can contain a wide range of ingredients, such as glutathione, vitamin C, collagen, or human placenta. They are advertised as being able to lighten skin, correct uneven skin tone, and clear blemishes. More exaggerated claims include the ability to treat Parkinson’s disease and liver disorders.
Although makers and distributors of unlicensed skin-lightening products have been subject to legal action and recalls in the past, the products continue to be marketed. Also of concern are noninjectable, over-the-counter skin-bleaching products that contain ammoniated mercury.
“In general, consumers should be cautious of any product marketed online with exaggerated claims on safety and effectiveness. They also should consult their health care practitioner before deciding to use any new product,” In Kim, an FDA pharmacist, said in a written statement.
Find the full statement on the FDA website here.