User login
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in Patient With Melanoma
Immune checkpoint inhibitors (ICIs) have become important in oncology and represent an evolving area of therapeutics. Since their approval by the US Food and Drug Administration (FDA) in 2011, ICIs have been increasingly used as modalities in neoadjuvant and adjuvant treatment for resectable solid malignancies and in unresectable disease, such as advanced melanoma, and are associated with improved survival.1
Immune checkpoints are present on the cell surface of activated T cells as well as other immune cells like B cells and natural killer cells. By regulating the length and amplitude of the body’s innate immune response, they maintain immune homeostasis and prevent its overactivation. Immune checkpoints are often thought of as the brakes on the immune system.2
Two glycoproteins that act as immune checkpoints and are targeted by ICIs are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1). CTLA-4 is upregulated on activated T cells. PD-1 is also expressed on activated T cells, as well as macrophages, B cells, and dendritic cells. Cancer cells can evade immune surveillance by exploiting immune checkpoint pathways. Inhibition of these checkpoints with ICIs reactivates T cells and enables the immune system to recognize and attack cancer cells more effectively. Ipilimumab blocks the activity of CTLA-4 on T cells. Nivolumab and pembrolizumab block the interaction between PD-1 on T cells and its ligand PD-L1 on cancer cells.3,4
Inhibition of these checkpoints is often effective in cancer treatment but can result in the loss of immunologic tolerance with resultant immune-related adverse events (irAEs) and potentially permanent autoimmune disorders. Autoreactive T cells can damage host cell tissues including the colon, lungs, liver, pituitary gland, thyroid, and skin. Severe irAEs include type 1 diabetes mellitus, myositis, nephritis, colitis, pneumonitis, hepatitis, uveitis, hypophysitis, and adrenalitis.4
Hypophysitis is inflammation of the pituitary gland, often with thickening of the pituitary stalk, resulting in dysfunction and hormone deficiencies. While primary hypophysitis is idiopathic, secondary hypophysitis is the result of an underlying condition such as exposure to an ICI. Immune-mediated inflammation of the pituitary gland in hypophysitis may disrupt corticotroph function, leading to adrenocorticotropic hormone (ACTH) deficiency. Early warning features are often vague and nonspecific, such as headache, fatigue, and weakness, which makes diagnosis challenging.3,5
CASE PRESENTATION
A 73-year-old male veteran with a history of metastatic melanoma on ipilimumab 3 mg/kg and nivolumab 1 mg/kg every 3 weeks (a standard combination regimen for advanced melanoma) presented to the emergency department (ED) with 2 weeks of cough, nausea, and severe headache 3 weeks after cycle 2 of combination ICI therapy. The patient had undergone excision of multiple sites of melanoma in situ with recurrence and disease progression after 5 cycles of pembrolizumab. He was subsequently started on combination ICI therapy.
On ED arrival, the patient was afebrile and saturating well on room air. He was normotensive but found to have orthostatic blood pressure. Physical examination was remarkable for dry oral mucosa and decreased skin turgor. Initial laboratory results were significant for hyponatremia of 123 mmol/L (reference range, 136-145 mmol/L), low-normal free thyroxine (T4) level of 0.5 ng/dL (reference range, 0.6-1.2 ng/dL), a low total triiodothyronine level of 32.14 ng/dL (reference range, 85-178 ng/dL), and a low thyrotropin level of 0.19 mIU/L (reference range, 0.35-5.50 mIU/L). Serum osmolarity was low at 259 mOsm/kg (reference range, 285-315 mOsm/kg), urine sodium was high at 168 mEq/L (reference, 20 mEq/L), and urine osmolarity was inappropriately concentrated at 726 mOsm/kg (reference range, 250-1000 mOsm/kg). The patient was admitted for additional testing. His morning cortisol level was within normal limits at 15 mcg/dL (reference range, 6.7-22.5 mcg/dL).
Computed tomography (CT) of the patient’s head revealed no acute findings. Chest CT revealed posterior right lower lobe mild ground-glass opacities, with possible ICI-induced pneumonitis. The patient received fluid resuscitation. Given concern for syndrome of inappropriate antidiuretic hormone secretion, the patient was started on 3 g salt tablets 3 times a day and urea 30 g powder daily. The etiology of the abnormal thyroid levels was unclear to endocrinology at that time. The differential diagnosis included a nonthyroidal illness or central hypothyroidism.
The patient started levothyroxine 75 mcg due to abnormal thyroid levels and persistent fatigue and fludrocortisone 0.1 mg daily to manage orthostatic hypotension. His sodium levels improved to 132 mmol/L over 6 days and he was discharged with levothyroxine 75 mcg daily, fludrocortisone 0.1 mg daily, 3 g salt tabs 3 times a day, urea 30 g powder daily, as well as oral cefpodoxime 500 mg twice daily for 3 days and azithromycin 500 mg once daily for 2 days (for a total of 10 days of antibiotic therapy) to treat potential occult pneumonia.
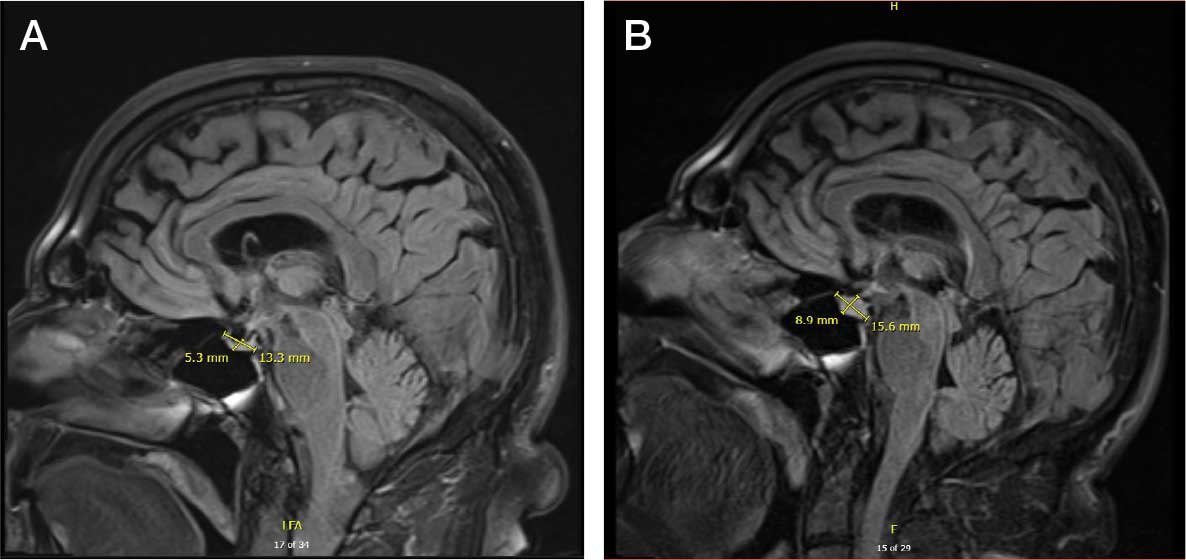
The patient returned to the ED 3 days after discharge following an outpatient oncology appointment with ongoing severe headaches and persistent nausea. There was concern for recurrent hyponatremia. His sodium level was within normal limits at 133 mmol/L. Repeat morning cortisol was low-normal at 9 mcg/dL. Magnetic resonance imaging (MRI) of the brain was negative for metastatic disease, but showed a slight interval increase in size of the pituitary gland compared with an MRI from 6 months prior, with mild fullness and a slightly convex superior margin near homogeneous enhancement, raising concern for infection or hypophysitis (Figure 1).
The patient was readmitted to the general medicine service and was given intravenous hydrocortisone 100 mg every 8 hours because of concern for central adrenal insufficiency due to grade 3 hypophysitis in the setting of MRI imaging and severe headaches (Table 1). He was not hypotensive at the time of hydrocortisone initiation and other vital signs were stable. A cosyntropin stimulation test—a standard diagnostic test for central adrenal insufficiency—was not performed because the patient had already started high-dose hydrocortisone. The patient’s free T4 on this admission remained low at 0.6 ng/dL.

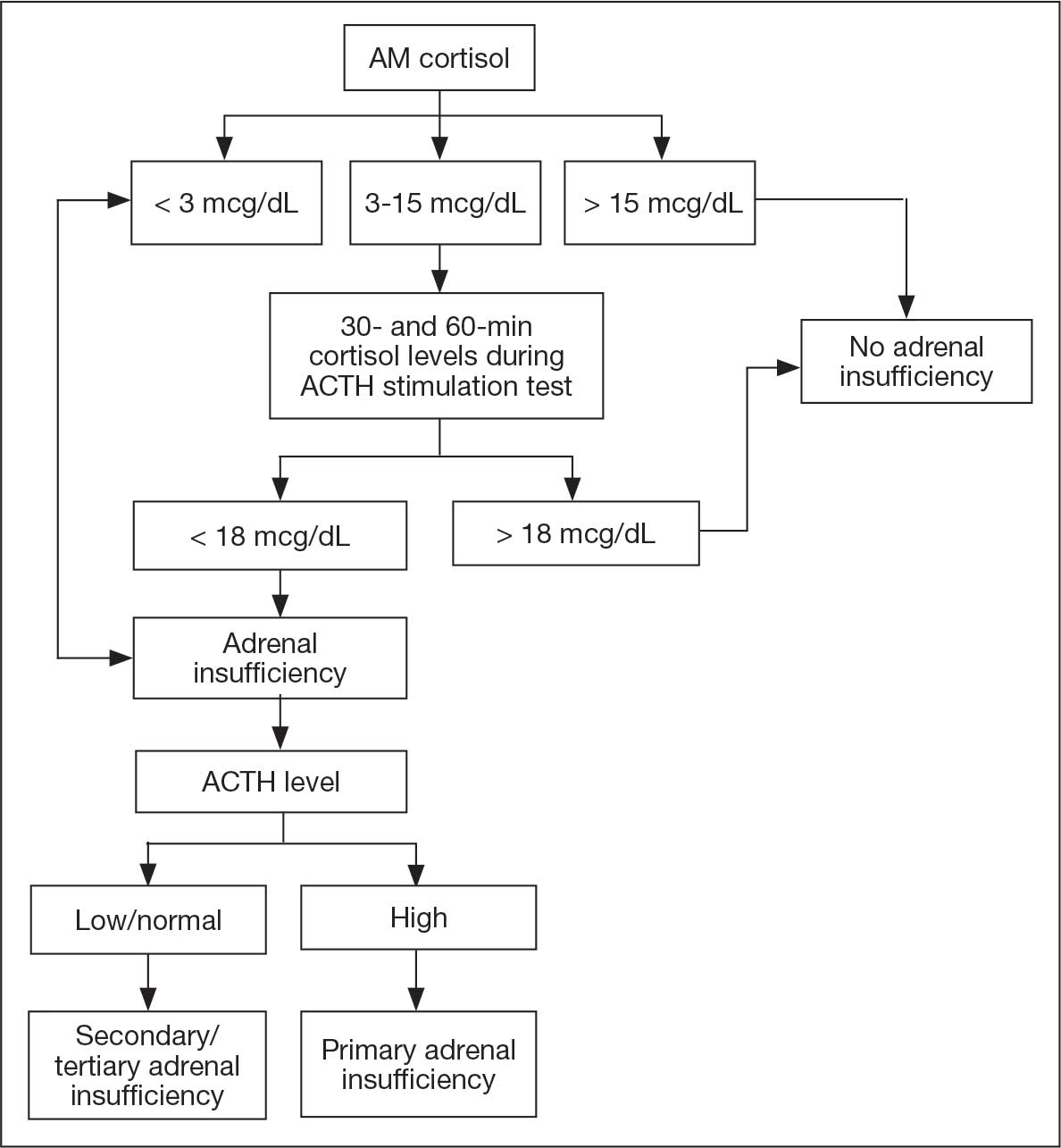
No adjustments were made to his levothyroxine dose given that he recently began the medication and levels may lag after initiation. After a 4-day hospitalization, the decision was made to continue with the steroid taper and follow up with outpatient endocrinology to obtain a cosyntropin stimulation test to complete a full assessment of his pituitary axis (Figure 2). Repeat thyroid function testing for levothyroxine titration was arranged. The levothyroxine dosage was later increased to 88 mcg daily, but the patient discontinued the medication and remained euthyroid. Endocrinology attributed a nonthyroidal illness as the etiology of his hypothyroidism, likely euthyroid sick syndrome in the setting of illness. His hydrocortisone was tapered during outpatient care and fludrocortisone was discontinued due to hypertension.
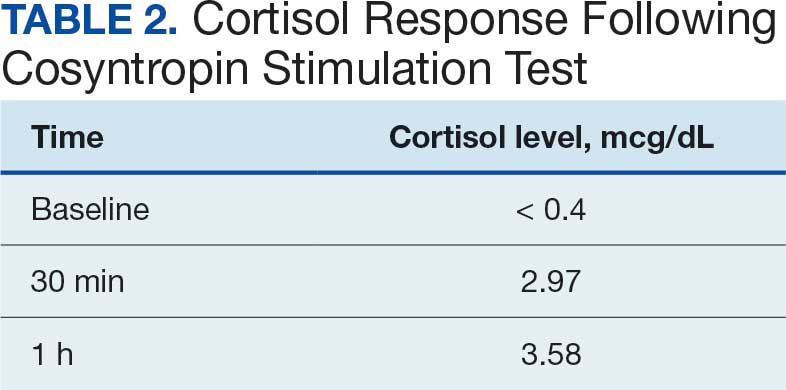
One month after his second discharge, the patient presented to the ED with 2 weeks of dizziness, associated lightheadedness, and blurred vision when standing from a sitting position. Upon assessment, symptoms were attributed to poor oral intake. The patient’s vital signs were again positive for orthostatic hypotension, though refractory to adequate fluid replacement. Laboratory testing was significant for a low ACTH level of 3.0 pg/mL (reference range, 7.2-63.3 pg/mL). Given that the patient had not received steroids for 1 week, he underwent a cosyntropin stimulation test, which revealed a blunted response supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis (Table 2).
The patient was again readmitted to the general medicine service. A brain MRI showed interval shrinkage of the pituitary gland compared to imaging one month prior, which was attributed to hydrocortisone treatment during this month. CT of the patient’s abdomen demonstrated normal-sized adrenal glands. Positron emission tomography (PET)/CT showed no evidence of pituitary or adrenal metastases. Endocrinology recommended reinitiating oral hydrocortisone 50 mg in the morning and 50 mg around 3 pm daily with fludrocortisone 0.2 mg once daily, which resulted in near resolution of the patient’s symptoms. He was discharged after a 14-day hospitalization with home physical therapy services and endocrinology, nephrology, and oncology follow-up appointments.
The patient was readmitted twice to the general medicine service over the next 6 months for complications from hydrocortisone and fludrocortisone treatment including hypokalemia. He followed up with outpatient clinicians until his death 14 months later. He did not restart ICI therapy, and eventually joined a clinical trial for other advanced melanoma treatments at another institution. The patient’s family consented to the publication of this case report with the accompanying images.
DISCUSSION
The combination of ipilimumab (anti-CTLA-4 monoclonal antibody) and nivolumab (anti-PD-1 monoclonal antibody) is FDA-approved for treatment of advanced melanoma with the goal of harnessing complementary and synergistic mechanisms of dual therapy.6-8 Combination therapy, however, can increase the incidence of irAEs, which are often endocrine-related and more common in patients treated with dual immunotherapy than with monotherapy.9 Hypophysitis has the lowest reported fatality rate among ICI-related irAEs (< 1%), compared with higher mortality rates seen in myocarditis (25%-50%) and pneumonitis (10%-20%).4,10
The patient initially presented with ICI-related hypothyroidism, later identified as secondary (central) hypothyroidism. He was treated with levothyroxine until central hypothyroidism was confirmed. Subsequently, the patient developed headache, poor appetite, and lightheadedness, with MRI findings suggestive of hypophysitis, for which he was started on hydrocortisone. A component of primary adrenal insufficiency was initially considered, given the low ACTH level and blunted response to cosyntropin stimulation following prior high-dose steroid therapy. However, CT imaging demonstrated normal adrenal morphology without atrophy, supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis.
The estimated incidence of ICI-induced hypophysitis is 1.5% to 13.3% with anti-CTLA-4 agents, 0.3% to 3.0% with anti-PD-1 agents, and can be as high as 12.8% with combination therapy.1 ICI-induced hypophysitis is believed to arise from the direct binding of ICI antibodies to their targets on anterior pituitary cells, such as corticotrophs, thyrotrophs, and gonadotrophs, triggering an immune response. One theory for targeting these cells is high CTLA-4 expression in the anterior pituitary gland.11 PD-1 therapies tend to manifest as either hypothyroidism, hyperthyroidism, Graves’ disease, diabetes, or adrenal insufficiency.10
A concern in patients with advanced melanoma is metastasis. Melanoma has a high propensity for brain metastasis.12 There was moderate suspicion for pituitary gland metastasis in this case, though pituitary metastasis more often manifests with symptoms of posterior pituitary gland deficiency, such as polyuria and polydipsia.13 The adrenal gland is the fourth-most common site for melanoma metastases, after the lung, liver, and bone.14 This patient had no evidence of pituitary or adrenal metastases on PET/CT. Therefore, his symptoms were most likely due to ICI therapy. Cases of ≥ 1 endocrine dysfunction have been reported as an ICI therapy irAE.15 In these situations, diagnosing primary and central adrenal insufficiency in the same patient is complex because hormone profiles are intertwined.
Many patients who develop hypophysitis from ICI therapy will require permanent replacement therapy. It is unclear whether low-dose replacement steroids have a significant effect on the efficacy of ICIs. Given that ICI treatment works by enhancing the immune system, medications that suppress the body’s immune system, such as steroids, could interfere with treatment efficacy. However, there are speculations that the development of irAEs is an indicator of effective treatment. In a phase 1 trial of a CTLA-4 blocker in patients with metastatic melanoma, there was a correlation between reduced CTLA-4 expression as well as low rates of melanoma recurrence and a higher incidence of irAEs.16
When assessing patients on ICI treatment, clinicians must remain vigilant for all potential irAEs, especially in patients receiving combination therapy. ICI-induced irAEs can present with vague and nonspecific symptoms. Concurrent endocrine irAEs, such as hypophysitis with thyroiditis or adrenalitis, are not uncommon in combination therapy and can complicate interpretation of hormone profiles. It is prudent for clinicians to review known risk factors. Hypophysitis is typically associated with older adult male patients.17,18
The irAEs of ICI therapy deeply affected the quality of life of the patient in this case, as he was often experiencing many of the clinical symptoms of his hormone insufficiencies as well as the treatment modalities, thus requiring repeated hospital admissions. The risks and benefits of continuing ICI therapy should be an ongoing discussion between the physician and patient and should take into account the acuity and severity of irAEs and oncological disease burden, among other variables. Given the severity of his AEs, the patient stopped ICI therapy and instead opted to enroll in a clinical trial at another institution for continued alternative treatments.
CONCLUSIONS
This case offers a lesson in the diagnostic challenges of vague symptoms in patients with cancer who are receiving ICI therapy. ICI therapy is widely used in the treatment of solid malignancies, and as its use increases, it is expected that clinicians will likely see more cases of irAEs in hospitalized patients. The vague presentation of irAEs can often lead to treatment delays, especially when > 1 irAE presents concurrently. There are ongoing studies researching potential ways to predict the likelihood of developing these irAEs. It is imperative that clinicians are aware of these ICI-related complications and that more research be conducted to understand patient quality of life and treatment guidance based on irAE severity and disease burden.
- Villani A, Potestio L, Fabbrocini G, et al. The treatment of advanced melanoma: therapeutic update. Int J Mol Sci. 2022;23:6388. doi:10.3390/ijms23126388
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. doi:10.1038/nrc3239
- Chang LS, Barroso-Sousa R, Tolaney SM, et al. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40:17-65. doi:10.1210/er.2018-00006
- June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23:540-547. doi:10.1038/nm.4321
- Jessel S, Weiss SA, Austin M, et al. Immune checkpoint inhibitor-induced hypophysitis and patterns of loss of pituitary function. Front Oncol. 2022;12:836859. doi:10.3389/fonc.2022.836859
- Betof AS, Nipp RD, Giobbie-Hurder A, et al. Impact of age on outcomes with immunotherapy for patients with melanoma. Oncologist. 2017;22:963-971. doi:10.1634/theoncologist.2016-0450
- Wolchok JD, Kluger H, Callahan MK, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. doi:10.1056/NEJMoa1302369
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. doi:10.1056/NEJMoa1003466
- Benhima N, Belbaraka R, Langouo Fontsa MD. Single agent vs combination immunotherapy in advanced melanoma: a review of the evidence. Curr Opin Oncol. 2024;36:69-73. doi:10.1097/CCO.0000000000001014
- Tong J, Kartolo A, Yeung C, et al. Long-term toxicities of immune checkpoint inhibitor (ICI) in melanoma patients. Curr Oncol. 2022;29:7953-7963. doi:10.3390/curroncol29100629
- Grouthier V, Lebrun-Vignes B, Moey M, et al. Immune checkpoint inhibitor-associated primary adrenal insufficiency: WHO VigiBase report analysis. Oncologist. 2020;25:696-701. doi:10.1634/theoncologist.2019-0555
- Park BC, Jung S, Wright JJ, et al. Recurrence of hypophysitis after immune checkpoint inhibitor rechallenge. Oncologist. 2022;27:e967-e969. doi:10.1093/oncolo/oyac220
- Zhang D, Wang Z, Shang D, et al. Incidence and prognosis of brain metastases in cutaneous melanoma patients: a population-based study. Melanoma Res. 2019;29:77-84. doi:10.1097/CMR.0000000000000538
- Barnabei A, Carpano S, Chiefari A, et al. Case report: ipilimumab-induced panhypophysitis: an infrequent occurrence and literature review. Front Oncol. 2020;10:582394. doi:10.3389/fonc.2020.582394
- Shortreed H, Burute N, Aseyev O. Management of undifferentiated adrenal gland metastases from malignant melanoma: case report. Front Oncol. 2024;14:1419827. doi:10.3389/fonc.2024.1419827
- Rossi S, Silvetti F, Bordoni M, et al. Pembrolizumab-induced thyroiditis, hypophysitis and adrenalitis: a case of triple endocrine dysfunction. JCEM Case Rep. 2024;2:luae200. doi:10.1210/jcemcr/luae200
- Sanderson K, Scotland R, Lee P, et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J Clin Oncol. 2005;23:741-750. doi:10.1200/JCO.2005.01.128
- de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51:145-156. doi:10.1055/a-0843-3366
Immune checkpoint inhibitors (ICIs) have become important in oncology and represent an evolving area of therapeutics. Since their approval by the US Food and Drug Administration (FDA) in 2011, ICIs have been increasingly used as modalities in neoadjuvant and adjuvant treatment for resectable solid malignancies and in unresectable disease, such as advanced melanoma, and are associated with improved survival.1
Immune checkpoints are present on the cell surface of activated T cells as well as other immune cells like B cells and natural killer cells. By regulating the length and amplitude of the body’s innate immune response, they maintain immune homeostasis and prevent its overactivation. Immune checkpoints are often thought of as the brakes on the immune system.2
Two glycoproteins that act as immune checkpoints and are targeted by ICIs are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1). CTLA-4 is upregulated on activated T cells. PD-1 is also expressed on activated T cells, as well as macrophages, B cells, and dendritic cells. Cancer cells can evade immune surveillance by exploiting immune checkpoint pathways. Inhibition of these checkpoints with ICIs reactivates T cells and enables the immune system to recognize and attack cancer cells more effectively. Ipilimumab blocks the activity of CTLA-4 on T cells. Nivolumab and pembrolizumab block the interaction between PD-1 on T cells and its ligand PD-L1 on cancer cells.3,4
Inhibition of these checkpoints is often effective in cancer treatment but can result in the loss of immunologic tolerance with resultant immune-related adverse events (irAEs) and potentially permanent autoimmune disorders. Autoreactive T cells can damage host cell tissues including the colon, lungs, liver, pituitary gland, thyroid, and skin. Severe irAEs include type 1 diabetes mellitus, myositis, nephritis, colitis, pneumonitis, hepatitis, uveitis, hypophysitis, and adrenalitis.4
Hypophysitis is inflammation of the pituitary gland, often with thickening of the pituitary stalk, resulting in dysfunction and hormone deficiencies. While primary hypophysitis is idiopathic, secondary hypophysitis is the result of an underlying condition such as exposure to an ICI. Immune-mediated inflammation of the pituitary gland in hypophysitis may disrupt corticotroph function, leading to adrenocorticotropic hormone (ACTH) deficiency. Early warning features are often vague and nonspecific, such as headache, fatigue, and weakness, which makes diagnosis challenging.3,5
CASE PRESENTATION
A 73-year-old male veteran with a history of metastatic melanoma on ipilimumab 3 mg/kg and nivolumab 1 mg/kg every 3 weeks (a standard combination regimen for advanced melanoma) presented to the emergency department (ED) with 2 weeks of cough, nausea, and severe headache 3 weeks after cycle 2 of combination ICI therapy. The patient had undergone excision of multiple sites of melanoma in situ with recurrence and disease progression after 5 cycles of pembrolizumab. He was subsequently started on combination ICI therapy.
On ED arrival, the patient was afebrile and saturating well on room air. He was normotensive but found to have orthostatic blood pressure. Physical examination was remarkable for dry oral mucosa and decreased skin turgor. Initial laboratory results were significant for hyponatremia of 123 mmol/L (reference range, 136-145 mmol/L), low-normal free thyroxine (T4) level of 0.5 ng/dL (reference range, 0.6-1.2 ng/dL), a low total triiodothyronine level of 32.14 ng/dL (reference range, 85-178 ng/dL), and a low thyrotropin level of 0.19 mIU/L (reference range, 0.35-5.50 mIU/L). Serum osmolarity was low at 259 mOsm/kg (reference range, 285-315 mOsm/kg), urine sodium was high at 168 mEq/L (reference, 20 mEq/L), and urine osmolarity was inappropriately concentrated at 726 mOsm/kg (reference range, 250-1000 mOsm/kg). The patient was admitted for additional testing. His morning cortisol level was within normal limits at 15 mcg/dL (reference range, 6.7-22.5 mcg/dL).
Computed tomography (CT) of the patient’s head revealed no acute findings. Chest CT revealed posterior right lower lobe mild ground-glass opacities, with possible ICI-induced pneumonitis. The patient received fluid resuscitation. Given concern for syndrome of inappropriate antidiuretic hormone secretion, the patient was started on 3 g salt tablets 3 times a day and urea 30 g powder daily. The etiology of the abnormal thyroid levels was unclear to endocrinology at that time. The differential diagnosis included a nonthyroidal illness or central hypothyroidism.
The patient started levothyroxine 75 mcg due to abnormal thyroid levels and persistent fatigue and fludrocortisone 0.1 mg daily to manage orthostatic hypotension. His sodium levels improved to 132 mmol/L over 6 days and he was discharged with levothyroxine 75 mcg daily, fludrocortisone 0.1 mg daily, 3 g salt tabs 3 times a day, urea 30 g powder daily, as well as oral cefpodoxime 500 mg twice daily for 3 days and azithromycin 500 mg once daily for 2 days (for a total of 10 days of antibiotic therapy) to treat potential occult pneumonia.
The patient returned to the ED 3 days after discharge following an outpatient oncology appointment with ongoing severe headaches and persistent nausea. There was concern for recurrent hyponatremia. His sodium level was within normal limits at 133 mmol/L. Repeat morning cortisol was low-normal at 9 mcg/dL. Magnetic resonance imaging (MRI) of the brain was negative for metastatic disease, but showed a slight interval increase in size of the pituitary gland compared with an MRI from 6 months prior, with mild fullness and a slightly convex superior margin near homogeneous enhancement, raising concern for infection or hypophysitis (Figure 1).

The patient was readmitted to the general medicine service and was given intravenous hydrocortisone 100 mg every 8 hours because of concern for central adrenal insufficiency due to grade 3 hypophysitis in the setting of MRI imaging and severe headaches (Table 1). He was not hypotensive at the time of hydrocortisone initiation and other vital signs were stable. A cosyntropin stimulation test—a standard diagnostic test for central adrenal insufficiency—was not performed because the patient had already started high-dose hydrocortisone. The patient’s free T4 on this admission remained low at 0.6 ng/dL.

No adjustments were made to his levothyroxine dose given that he recently began the medication and levels may lag after initiation. After a 4-day hospitalization, the decision was made to continue with the steroid taper and follow up with outpatient endocrinology to obtain a cosyntropin stimulation test to complete a full assessment of his pituitary axis (Figure 2). Repeat thyroid function testing for levothyroxine titration was arranged. The levothyroxine dosage was later increased to 88 mcg daily, but the patient discontinued the medication and remained euthyroid. Endocrinology attributed a nonthyroidal illness as the etiology of his hypothyroidism, likely euthyroid sick syndrome in the setting of illness. His hydrocortisone was tapered during outpatient care and fludrocortisone was discontinued due to hypertension.

One month after his second discharge, the patient presented to the ED with 2 weeks of dizziness, associated lightheadedness, and blurred vision when standing from a sitting position. Upon assessment, symptoms were attributed to poor oral intake. The patient’s vital signs were again positive for orthostatic hypotension, though refractory to adequate fluid replacement. Laboratory testing was significant for a low ACTH level of 3.0 pg/mL (reference range, 7.2-63.3 pg/mL). Given that the patient had not received steroids for 1 week, he underwent a cosyntropin stimulation test, which revealed a blunted response supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis (Table 2).

The patient was again readmitted to the general medicine service. A brain MRI showed interval shrinkage of the pituitary gland compared to imaging one month prior, which was attributed to hydrocortisone treatment during this month. CT of the patient’s abdomen demonstrated normal-sized adrenal glands. Positron emission tomography (PET)/CT showed no evidence of pituitary or adrenal metastases. Endocrinology recommended reinitiating oral hydrocortisone 50 mg in the morning and 50 mg around 3 pm daily with fludrocortisone 0.2 mg once daily, which resulted in near resolution of the patient’s symptoms. He was discharged after a 14-day hospitalization with home physical therapy services and endocrinology, nephrology, and oncology follow-up appointments.
The patient was readmitted twice to the general medicine service over the next 6 months for complications from hydrocortisone and fludrocortisone treatment including hypokalemia. He followed up with outpatient clinicians until his death 14 months later. He did not restart ICI therapy, and eventually joined a clinical trial for other advanced melanoma treatments at another institution. The patient’s family consented to the publication of this case report with the accompanying images.
DISCUSSION
The combination of ipilimumab (anti-CTLA-4 monoclonal antibody) and nivolumab (anti-PD-1 monoclonal antibody) is FDA-approved for treatment of advanced melanoma with the goal of harnessing complementary and synergistic mechanisms of dual therapy.6-8 Combination therapy, however, can increase the incidence of irAEs, which are often endocrine-related and more common in patients treated with dual immunotherapy than with monotherapy.9 Hypophysitis has the lowest reported fatality rate among ICI-related irAEs (< 1%), compared with higher mortality rates seen in myocarditis (25%-50%) and pneumonitis (10%-20%).4,10
The patient initially presented with ICI-related hypothyroidism, later identified as secondary (central) hypothyroidism. He was treated with levothyroxine until central hypothyroidism was confirmed. Subsequently, the patient developed headache, poor appetite, and lightheadedness, with MRI findings suggestive of hypophysitis, for which he was started on hydrocortisone. A component of primary adrenal insufficiency was initially considered, given the low ACTH level and blunted response to cosyntropin stimulation following prior high-dose steroid therapy. However, CT imaging demonstrated normal adrenal morphology without atrophy, supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis.
The estimated incidence of ICI-induced hypophysitis is 1.5% to 13.3% with anti-CTLA-4 agents, 0.3% to 3.0% with anti-PD-1 agents, and can be as high as 12.8% with combination therapy.1 ICI-induced hypophysitis is believed to arise from the direct binding of ICI antibodies to their targets on anterior pituitary cells, such as corticotrophs, thyrotrophs, and gonadotrophs, triggering an immune response. One theory for targeting these cells is high CTLA-4 expression in the anterior pituitary gland.11 PD-1 therapies tend to manifest as either hypothyroidism, hyperthyroidism, Graves’ disease, diabetes, or adrenal insufficiency.10
A concern in patients with advanced melanoma is metastasis. Melanoma has a high propensity for brain metastasis.12 There was moderate suspicion for pituitary gland metastasis in this case, though pituitary metastasis more often manifests with symptoms of posterior pituitary gland deficiency, such as polyuria and polydipsia.13 The adrenal gland is the fourth-most common site for melanoma metastases, after the lung, liver, and bone.14 This patient had no evidence of pituitary or adrenal metastases on PET/CT. Therefore, his symptoms were most likely due to ICI therapy. Cases of ≥ 1 endocrine dysfunction have been reported as an ICI therapy irAE.15 In these situations, diagnosing primary and central adrenal insufficiency in the same patient is complex because hormone profiles are intertwined.
Many patients who develop hypophysitis from ICI therapy will require permanent replacement therapy. It is unclear whether low-dose replacement steroids have a significant effect on the efficacy of ICIs. Given that ICI treatment works by enhancing the immune system, medications that suppress the body’s immune system, such as steroids, could interfere with treatment efficacy. However, there are speculations that the development of irAEs is an indicator of effective treatment. In a phase 1 trial of a CTLA-4 blocker in patients with metastatic melanoma, there was a correlation between reduced CTLA-4 expression as well as low rates of melanoma recurrence and a higher incidence of irAEs.16
When assessing patients on ICI treatment, clinicians must remain vigilant for all potential irAEs, especially in patients receiving combination therapy. ICI-induced irAEs can present with vague and nonspecific symptoms. Concurrent endocrine irAEs, such as hypophysitis with thyroiditis or adrenalitis, are not uncommon in combination therapy and can complicate interpretation of hormone profiles. It is prudent for clinicians to review known risk factors. Hypophysitis is typically associated with older adult male patients.17,18
The irAEs of ICI therapy deeply affected the quality of life of the patient in this case, as he was often experiencing many of the clinical symptoms of his hormone insufficiencies as well as the treatment modalities, thus requiring repeated hospital admissions. The risks and benefits of continuing ICI therapy should be an ongoing discussion between the physician and patient and should take into account the acuity and severity of irAEs and oncological disease burden, among other variables. Given the severity of his AEs, the patient stopped ICI therapy and instead opted to enroll in a clinical trial at another institution for continued alternative treatments.
CONCLUSIONS
This case offers a lesson in the diagnostic challenges of vague symptoms in patients with cancer who are receiving ICI therapy. ICI therapy is widely used in the treatment of solid malignancies, and as its use increases, it is expected that clinicians will likely see more cases of irAEs in hospitalized patients. The vague presentation of irAEs can often lead to treatment delays, especially when > 1 irAE presents concurrently. There are ongoing studies researching potential ways to predict the likelihood of developing these irAEs. It is imperative that clinicians are aware of these ICI-related complications and that more research be conducted to understand patient quality of life and treatment guidance based on irAE severity and disease burden.
Immune checkpoint inhibitors (ICIs) have become important in oncology and represent an evolving area of therapeutics. Since their approval by the US Food and Drug Administration (FDA) in 2011, ICIs have been increasingly used as modalities in neoadjuvant and adjuvant treatment for resectable solid malignancies and in unresectable disease, such as advanced melanoma, and are associated with improved survival.1
Immune checkpoints are present on the cell surface of activated T cells as well as other immune cells like B cells and natural killer cells. By regulating the length and amplitude of the body’s innate immune response, they maintain immune homeostasis and prevent its overactivation. Immune checkpoints are often thought of as the brakes on the immune system.2
Two glycoproteins that act as immune checkpoints and are targeted by ICIs are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1). CTLA-4 is upregulated on activated T cells. PD-1 is also expressed on activated T cells, as well as macrophages, B cells, and dendritic cells. Cancer cells can evade immune surveillance by exploiting immune checkpoint pathways. Inhibition of these checkpoints with ICIs reactivates T cells and enables the immune system to recognize and attack cancer cells more effectively. Ipilimumab blocks the activity of CTLA-4 on T cells. Nivolumab and pembrolizumab block the interaction between PD-1 on T cells and its ligand PD-L1 on cancer cells.3,4
Inhibition of these checkpoints is often effective in cancer treatment but can result in the loss of immunologic tolerance with resultant immune-related adverse events (irAEs) and potentially permanent autoimmune disorders. Autoreactive T cells can damage host cell tissues including the colon, lungs, liver, pituitary gland, thyroid, and skin. Severe irAEs include type 1 diabetes mellitus, myositis, nephritis, colitis, pneumonitis, hepatitis, uveitis, hypophysitis, and adrenalitis.4
Hypophysitis is inflammation of the pituitary gland, often with thickening of the pituitary stalk, resulting in dysfunction and hormone deficiencies. While primary hypophysitis is idiopathic, secondary hypophysitis is the result of an underlying condition such as exposure to an ICI. Immune-mediated inflammation of the pituitary gland in hypophysitis may disrupt corticotroph function, leading to adrenocorticotropic hormone (ACTH) deficiency. Early warning features are often vague and nonspecific, such as headache, fatigue, and weakness, which makes diagnosis challenging.3,5
CASE PRESENTATION
A 73-year-old male veteran with a history of metastatic melanoma on ipilimumab 3 mg/kg and nivolumab 1 mg/kg every 3 weeks (a standard combination regimen for advanced melanoma) presented to the emergency department (ED) with 2 weeks of cough, nausea, and severe headache 3 weeks after cycle 2 of combination ICI therapy. The patient had undergone excision of multiple sites of melanoma in situ with recurrence and disease progression after 5 cycles of pembrolizumab. He was subsequently started on combination ICI therapy.
On ED arrival, the patient was afebrile and saturating well on room air. He was normotensive but found to have orthostatic blood pressure. Physical examination was remarkable for dry oral mucosa and decreased skin turgor. Initial laboratory results were significant for hyponatremia of 123 mmol/L (reference range, 136-145 mmol/L), low-normal free thyroxine (T4) level of 0.5 ng/dL (reference range, 0.6-1.2 ng/dL), a low total triiodothyronine level of 32.14 ng/dL (reference range, 85-178 ng/dL), and a low thyrotropin level of 0.19 mIU/L (reference range, 0.35-5.50 mIU/L). Serum osmolarity was low at 259 mOsm/kg (reference range, 285-315 mOsm/kg), urine sodium was high at 168 mEq/L (reference, 20 mEq/L), and urine osmolarity was inappropriately concentrated at 726 mOsm/kg (reference range, 250-1000 mOsm/kg). The patient was admitted for additional testing. His morning cortisol level was within normal limits at 15 mcg/dL (reference range, 6.7-22.5 mcg/dL).
Computed tomography (CT) of the patient’s head revealed no acute findings. Chest CT revealed posterior right lower lobe mild ground-glass opacities, with possible ICI-induced pneumonitis. The patient received fluid resuscitation. Given concern for syndrome of inappropriate antidiuretic hormone secretion, the patient was started on 3 g salt tablets 3 times a day and urea 30 g powder daily. The etiology of the abnormal thyroid levels was unclear to endocrinology at that time. The differential diagnosis included a nonthyroidal illness or central hypothyroidism.
The patient started levothyroxine 75 mcg due to abnormal thyroid levels and persistent fatigue and fludrocortisone 0.1 mg daily to manage orthostatic hypotension. His sodium levels improved to 132 mmol/L over 6 days and he was discharged with levothyroxine 75 mcg daily, fludrocortisone 0.1 mg daily, 3 g salt tabs 3 times a day, urea 30 g powder daily, as well as oral cefpodoxime 500 mg twice daily for 3 days and azithromycin 500 mg once daily for 2 days (for a total of 10 days of antibiotic therapy) to treat potential occult pneumonia.
The patient returned to the ED 3 days after discharge following an outpatient oncology appointment with ongoing severe headaches and persistent nausea. There was concern for recurrent hyponatremia. His sodium level was within normal limits at 133 mmol/L. Repeat morning cortisol was low-normal at 9 mcg/dL. Magnetic resonance imaging (MRI) of the brain was negative for metastatic disease, but showed a slight interval increase in size of the pituitary gland compared with an MRI from 6 months prior, with mild fullness and a slightly convex superior margin near homogeneous enhancement, raising concern for infection or hypophysitis (Figure 1).

The patient was readmitted to the general medicine service and was given intravenous hydrocortisone 100 mg every 8 hours because of concern for central adrenal insufficiency due to grade 3 hypophysitis in the setting of MRI imaging and severe headaches (Table 1). He was not hypotensive at the time of hydrocortisone initiation and other vital signs were stable. A cosyntropin stimulation test—a standard diagnostic test for central adrenal insufficiency—was not performed because the patient had already started high-dose hydrocortisone. The patient’s free T4 on this admission remained low at 0.6 ng/dL.

No adjustments were made to his levothyroxine dose given that he recently began the medication and levels may lag after initiation. After a 4-day hospitalization, the decision was made to continue with the steroid taper and follow up with outpatient endocrinology to obtain a cosyntropin stimulation test to complete a full assessment of his pituitary axis (Figure 2). Repeat thyroid function testing for levothyroxine titration was arranged. The levothyroxine dosage was later increased to 88 mcg daily, but the patient discontinued the medication and remained euthyroid. Endocrinology attributed a nonthyroidal illness as the etiology of his hypothyroidism, likely euthyroid sick syndrome in the setting of illness. His hydrocortisone was tapered during outpatient care and fludrocortisone was discontinued due to hypertension.

One month after his second discharge, the patient presented to the ED with 2 weeks of dizziness, associated lightheadedness, and blurred vision when standing from a sitting position. Upon assessment, symptoms were attributed to poor oral intake. The patient’s vital signs were again positive for orthostatic hypotension, though refractory to adequate fluid replacement. Laboratory testing was significant for a low ACTH level of 3.0 pg/mL (reference range, 7.2-63.3 pg/mL). Given that the patient had not received steroids for 1 week, he underwent a cosyntropin stimulation test, which revealed a blunted response supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis (Table 2).

The patient was again readmitted to the general medicine service. A brain MRI showed interval shrinkage of the pituitary gland compared to imaging one month prior, which was attributed to hydrocortisone treatment during this month. CT of the patient’s abdomen demonstrated normal-sized adrenal glands. Positron emission tomography (PET)/CT showed no evidence of pituitary or adrenal metastases. Endocrinology recommended reinitiating oral hydrocortisone 50 mg in the morning and 50 mg around 3 pm daily with fludrocortisone 0.2 mg once daily, which resulted in near resolution of the patient’s symptoms. He was discharged after a 14-day hospitalization with home physical therapy services and endocrinology, nephrology, and oncology follow-up appointments.
The patient was readmitted twice to the general medicine service over the next 6 months for complications from hydrocortisone and fludrocortisone treatment including hypokalemia. He followed up with outpatient clinicians until his death 14 months later. He did not restart ICI therapy, and eventually joined a clinical trial for other advanced melanoma treatments at another institution. The patient’s family consented to the publication of this case report with the accompanying images.
DISCUSSION
The combination of ipilimumab (anti-CTLA-4 monoclonal antibody) and nivolumab (anti-PD-1 monoclonal antibody) is FDA-approved for treatment of advanced melanoma with the goal of harnessing complementary and synergistic mechanisms of dual therapy.6-8 Combination therapy, however, can increase the incidence of irAEs, which are often endocrine-related and more common in patients treated with dual immunotherapy than with monotherapy.9 Hypophysitis has the lowest reported fatality rate among ICI-related irAEs (< 1%), compared with higher mortality rates seen in myocarditis (25%-50%) and pneumonitis (10%-20%).4,10
The patient initially presented with ICI-related hypothyroidism, later identified as secondary (central) hypothyroidism. He was treated with levothyroxine until central hypothyroidism was confirmed. Subsequently, the patient developed headache, poor appetite, and lightheadedness, with MRI findings suggestive of hypophysitis, for which he was started on hydrocortisone. A component of primary adrenal insufficiency was initially considered, given the low ACTH level and blunted response to cosyntropin stimulation following prior high-dose steroid therapy. However, CT imaging demonstrated normal adrenal morphology without atrophy, supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis.
The estimated incidence of ICI-induced hypophysitis is 1.5% to 13.3% with anti-CTLA-4 agents, 0.3% to 3.0% with anti-PD-1 agents, and can be as high as 12.8% with combination therapy.1 ICI-induced hypophysitis is believed to arise from the direct binding of ICI antibodies to their targets on anterior pituitary cells, such as corticotrophs, thyrotrophs, and gonadotrophs, triggering an immune response. One theory for targeting these cells is high CTLA-4 expression in the anterior pituitary gland.11 PD-1 therapies tend to manifest as either hypothyroidism, hyperthyroidism, Graves’ disease, diabetes, or adrenal insufficiency.10
A concern in patients with advanced melanoma is metastasis. Melanoma has a high propensity for brain metastasis.12 There was moderate suspicion for pituitary gland metastasis in this case, though pituitary metastasis more often manifests with symptoms of posterior pituitary gland deficiency, such as polyuria and polydipsia.13 The adrenal gland is the fourth-most common site for melanoma metastases, after the lung, liver, and bone.14 This patient had no evidence of pituitary or adrenal metastases on PET/CT. Therefore, his symptoms were most likely due to ICI therapy. Cases of ≥ 1 endocrine dysfunction have been reported as an ICI therapy irAE.15 In these situations, diagnosing primary and central adrenal insufficiency in the same patient is complex because hormone profiles are intertwined.
Many patients who develop hypophysitis from ICI therapy will require permanent replacement therapy. It is unclear whether low-dose replacement steroids have a significant effect on the efficacy of ICIs. Given that ICI treatment works by enhancing the immune system, medications that suppress the body’s immune system, such as steroids, could interfere with treatment efficacy. However, there are speculations that the development of irAEs is an indicator of effective treatment. In a phase 1 trial of a CTLA-4 blocker in patients with metastatic melanoma, there was a correlation between reduced CTLA-4 expression as well as low rates of melanoma recurrence and a higher incidence of irAEs.16
When assessing patients on ICI treatment, clinicians must remain vigilant for all potential irAEs, especially in patients receiving combination therapy. ICI-induced irAEs can present with vague and nonspecific symptoms. Concurrent endocrine irAEs, such as hypophysitis with thyroiditis or adrenalitis, are not uncommon in combination therapy and can complicate interpretation of hormone profiles. It is prudent for clinicians to review known risk factors. Hypophysitis is typically associated with older adult male patients.17,18
The irAEs of ICI therapy deeply affected the quality of life of the patient in this case, as he was often experiencing many of the clinical symptoms of his hormone insufficiencies as well as the treatment modalities, thus requiring repeated hospital admissions. The risks and benefits of continuing ICI therapy should be an ongoing discussion between the physician and patient and should take into account the acuity and severity of irAEs and oncological disease burden, among other variables. Given the severity of his AEs, the patient stopped ICI therapy and instead opted to enroll in a clinical trial at another institution for continued alternative treatments.
CONCLUSIONS
This case offers a lesson in the diagnostic challenges of vague symptoms in patients with cancer who are receiving ICI therapy. ICI therapy is widely used in the treatment of solid malignancies, and as its use increases, it is expected that clinicians will likely see more cases of irAEs in hospitalized patients. The vague presentation of irAEs can often lead to treatment delays, especially when > 1 irAE presents concurrently. There are ongoing studies researching potential ways to predict the likelihood of developing these irAEs. It is imperative that clinicians are aware of these ICI-related complications and that more research be conducted to understand patient quality of life and treatment guidance based on irAE severity and disease burden.
- Villani A, Potestio L, Fabbrocini G, et al. The treatment of advanced melanoma: therapeutic update. Int J Mol Sci. 2022;23:6388. doi:10.3390/ijms23126388
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. doi:10.1038/nrc3239
- Chang LS, Barroso-Sousa R, Tolaney SM, et al. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40:17-65. doi:10.1210/er.2018-00006
- June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23:540-547. doi:10.1038/nm.4321
- Jessel S, Weiss SA, Austin M, et al. Immune checkpoint inhibitor-induced hypophysitis and patterns of loss of pituitary function. Front Oncol. 2022;12:836859. doi:10.3389/fonc.2022.836859
- Betof AS, Nipp RD, Giobbie-Hurder A, et al. Impact of age on outcomes with immunotherapy for patients with melanoma. Oncologist. 2017;22:963-971. doi:10.1634/theoncologist.2016-0450
- Wolchok JD, Kluger H, Callahan MK, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. doi:10.1056/NEJMoa1302369
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. doi:10.1056/NEJMoa1003466
- Benhima N, Belbaraka R, Langouo Fontsa MD. Single agent vs combination immunotherapy in advanced melanoma: a review of the evidence. Curr Opin Oncol. 2024;36:69-73. doi:10.1097/CCO.0000000000001014
- Tong J, Kartolo A, Yeung C, et al. Long-term toxicities of immune checkpoint inhibitor (ICI) in melanoma patients. Curr Oncol. 2022;29:7953-7963. doi:10.3390/curroncol29100629
- Grouthier V, Lebrun-Vignes B, Moey M, et al. Immune checkpoint inhibitor-associated primary adrenal insufficiency: WHO VigiBase report analysis. Oncologist. 2020;25:696-701. doi:10.1634/theoncologist.2019-0555
- Park BC, Jung S, Wright JJ, et al. Recurrence of hypophysitis after immune checkpoint inhibitor rechallenge. Oncologist. 2022;27:e967-e969. doi:10.1093/oncolo/oyac220
- Zhang D, Wang Z, Shang D, et al. Incidence and prognosis of brain metastases in cutaneous melanoma patients: a population-based study. Melanoma Res. 2019;29:77-84. doi:10.1097/CMR.0000000000000538
- Barnabei A, Carpano S, Chiefari A, et al. Case report: ipilimumab-induced panhypophysitis: an infrequent occurrence and literature review. Front Oncol. 2020;10:582394. doi:10.3389/fonc.2020.582394
- Shortreed H, Burute N, Aseyev O. Management of undifferentiated adrenal gland metastases from malignant melanoma: case report. Front Oncol. 2024;14:1419827. doi:10.3389/fonc.2024.1419827
- Rossi S, Silvetti F, Bordoni M, et al. Pembrolizumab-induced thyroiditis, hypophysitis and adrenalitis: a case of triple endocrine dysfunction. JCEM Case Rep. 2024;2:luae200. doi:10.1210/jcemcr/luae200
- Sanderson K, Scotland R, Lee P, et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J Clin Oncol. 2005;23:741-750. doi:10.1200/JCO.2005.01.128
- de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51:145-156. doi:10.1055/a-0843-3366
- Villani A, Potestio L, Fabbrocini G, et al. The treatment of advanced melanoma: therapeutic update. Int J Mol Sci. 2022;23:6388. doi:10.3390/ijms23126388
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. doi:10.1038/nrc3239
- Chang LS, Barroso-Sousa R, Tolaney SM, et al. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40:17-65. doi:10.1210/er.2018-00006
- June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23:540-547. doi:10.1038/nm.4321
- Jessel S, Weiss SA, Austin M, et al. Immune checkpoint inhibitor-induced hypophysitis and patterns of loss of pituitary function. Front Oncol. 2022;12:836859. doi:10.3389/fonc.2022.836859
- Betof AS, Nipp RD, Giobbie-Hurder A, et al. Impact of age on outcomes with immunotherapy for patients with melanoma. Oncologist. 2017;22:963-971. doi:10.1634/theoncologist.2016-0450
- Wolchok JD, Kluger H, Callahan MK, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. doi:10.1056/NEJMoa1302369
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. doi:10.1056/NEJMoa1003466
- Benhima N, Belbaraka R, Langouo Fontsa MD. Single agent vs combination immunotherapy in advanced melanoma: a review of the evidence. Curr Opin Oncol. 2024;36:69-73. doi:10.1097/CCO.0000000000001014
- Tong J, Kartolo A, Yeung C, et al. Long-term toxicities of immune checkpoint inhibitor (ICI) in melanoma patients. Curr Oncol. 2022;29:7953-7963. doi:10.3390/curroncol29100629
- Grouthier V, Lebrun-Vignes B, Moey M, et al. Immune checkpoint inhibitor-associated primary adrenal insufficiency: WHO VigiBase report analysis. Oncologist. 2020;25:696-701. doi:10.1634/theoncologist.2019-0555
- Park BC, Jung S, Wright JJ, et al. Recurrence of hypophysitis after immune checkpoint inhibitor rechallenge. Oncologist. 2022;27:e967-e969. doi:10.1093/oncolo/oyac220
- Zhang D, Wang Z, Shang D, et al. Incidence and prognosis of brain metastases in cutaneous melanoma patients: a population-based study. Melanoma Res. 2019;29:77-84. doi:10.1097/CMR.0000000000000538
- Barnabei A, Carpano S, Chiefari A, et al. Case report: ipilimumab-induced panhypophysitis: an infrequent occurrence and literature review. Front Oncol. 2020;10:582394. doi:10.3389/fonc.2020.582394
- Shortreed H, Burute N, Aseyev O. Management of undifferentiated adrenal gland metastases from malignant melanoma: case report. Front Oncol. 2024;14:1419827. doi:10.3389/fonc.2024.1419827
- Rossi S, Silvetti F, Bordoni M, et al. Pembrolizumab-induced thyroiditis, hypophysitis and adrenalitis: a case of triple endocrine dysfunction. JCEM Case Rep. 2024;2:luae200. doi:10.1210/jcemcr/luae200
- Sanderson K, Scotland R, Lee P, et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J Clin Oncol. 2005;23:741-750. doi:10.1200/JCO.2005.01.128
- de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51:145-156. doi:10.1055/a-0843-3366
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in Patient With Melanoma
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in Patient With Melanoma