User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
Stop using radiation therapy in older women, expert says
MIAMI BEACH – The use of radiation therapy should be decreased in women at low risk of local recurrence of breast cancer, Dr. Kevin S. Hughes said at the annual Miami Breast Cancer Conference.
This is particularly true for women aged 70 years and older with clinical stage 1, estrogen receptor–positive cancers, but may also apply to younger women with low recurrence risk, Dr. Hughes, codirector of the Avon Comprehensive Breast Cancer Evaluation Center at Massachusetts General Hospital, Boston, said at the conference, held by Physicians’ Education Resource.
Older women have lower-risk breast cancers that are more responsive to hormonal treatments, he explained, noting that Cancer and Leukemia Group B protocol 9343 showed that radiation therapy provides little benefit among older women; the 10-year recurrence of 9% in those treated with conservation and radiation therapy was similar to the recurrence rate among women under age 40 years, and to the unilateral cancer risk of a patient with lobular carcinoma in situ.
No difference was seen in ultimate breast preservation, distant disease-free survival, or survival in those with vs. without radiation therapy, he said.
In fact, 94% of patients who died at the time of 12-year follow-up died of something other than breast cancer, he noted.
Despite efforts over the years to publicize these and similar findings, the use of radiation therapy in this population has not declined substantially.
“It is important to stop using radiation in older women who don’t benefit from this therapy and don’t really need it,” he stressed.
As for younger women, the PRIME II trial showed similar results in 658 women aged 65 years and older, and the PRECISION (Profiling Early Breast Cancer for Radiotherapy Omission) trial will look at women aged 50-75 years with stage 1, ER+ cancer who are at low risk of recurrence, Dr. Hughes said.
In PRIME II, postoperative whole-breast radiotherapy after breast-conserving surgery and adjuvant endocrine treatment provided some reduction in the local recurrence at 5-year follow-up, but the investigators concluded that the rate of ipsilateral breast tumor recurrence “is probably low enough for omission of radiotherapy to be considered for some patients (Lancet. 2015;16[3]:266-73).
The findings in older women are clear with the respect to the need for avoiding radiation therapy in those with low local recurrence risk, and the findings in younger women are promising, he concluded.
Dr. Hughes is on the speakers bureau for Myriad Genetics, is a shareholder of the healthcare IT company 5AM Solutions, and is the founder (with a financial interest) of Hughes Risk Apps, LLC.
MIAMI BEACH – The use of radiation therapy should be decreased in women at low risk of local recurrence of breast cancer, Dr. Kevin S. Hughes said at the annual Miami Breast Cancer Conference.
This is particularly true for women aged 70 years and older with clinical stage 1, estrogen receptor–positive cancers, but may also apply to younger women with low recurrence risk, Dr. Hughes, codirector of the Avon Comprehensive Breast Cancer Evaluation Center at Massachusetts General Hospital, Boston, said at the conference, held by Physicians’ Education Resource.
Older women have lower-risk breast cancers that are more responsive to hormonal treatments, he explained, noting that Cancer and Leukemia Group B protocol 9343 showed that radiation therapy provides little benefit among older women; the 10-year recurrence of 9% in those treated with conservation and radiation therapy was similar to the recurrence rate among women under age 40 years, and to the unilateral cancer risk of a patient with lobular carcinoma in situ.
No difference was seen in ultimate breast preservation, distant disease-free survival, or survival in those with vs. without radiation therapy, he said.
In fact, 94% of patients who died at the time of 12-year follow-up died of something other than breast cancer, he noted.
Despite efforts over the years to publicize these and similar findings, the use of radiation therapy in this population has not declined substantially.
“It is important to stop using radiation in older women who don’t benefit from this therapy and don’t really need it,” he stressed.
As for younger women, the PRIME II trial showed similar results in 658 women aged 65 years and older, and the PRECISION (Profiling Early Breast Cancer for Radiotherapy Omission) trial will look at women aged 50-75 years with stage 1, ER+ cancer who are at low risk of recurrence, Dr. Hughes said.
In PRIME II, postoperative whole-breast radiotherapy after breast-conserving surgery and adjuvant endocrine treatment provided some reduction in the local recurrence at 5-year follow-up, but the investigators concluded that the rate of ipsilateral breast tumor recurrence “is probably low enough for omission of radiotherapy to be considered for some patients (Lancet. 2015;16[3]:266-73).
The findings in older women are clear with the respect to the need for avoiding radiation therapy in those with low local recurrence risk, and the findings in younger women are promising, he concluded.
Dr. Hughes is on the speakers bureau for Myriad Genetics, is a shareholder of the healthcare IT company 5AM Solutions, and is the founder (with a financial interest) of Hughes Risk Apps, LLC.
MIAMI BEACH – The use of radiation therapy should be decreased in women at low risk of local recurrence of breast cancer, Dr. Kevin S. Hughes said at the annual Miami Breast Cancer Conference.
This is particularly true for women aged 70 years and older with clinical stage 1, estrogen receptor–positive cancers, but may also apply to younger women with low recurrence risk, Dr. Hughes, codirector of the Avon Comprehensive Breast Cancer Evaluation Center at Massachusetts General Hospital, Boston, said at the conference, held by Physicians’ Education Resource.
Older women have lower-risk breast cancers that are more responsive to hormonal treatments, he explained, noting that Cancer and Leukemia Group B protocol 9343 showed that radiation therapy provides little benefit among older women; the 10-year recurrence of 9% in those treated with conservation and radiation therapy was similar to the recurrence rate among women under age 40 years, and to the unilateral cancer risk of a patient with lobular carcinoma in situ.
No difference was seen in ultimate breast preservation, distant disease-free survival, or survival in those with vs. without radiation therapy, he said.
In fact, 94% of patients who died at the time of 12-year follow-up died of something other than breast cancer, he noted.
Despite efforts over the years to publicize these and similar findings, the use of radiation therapy in this population has not declined substantially.
“It is important to stop using radiation in older women who don’t benefit from this therapy and don’t really need it,” he stressed.
As for younger women, the PRIME II trial showed similar results in 658 women aged 65 years and older, and the PRECISION (Profiling Early Breast Cancer for Radiotherapy Omission) trial will look at women aged 50-75 years with stage 1, ER+ cancer who are at low risk of recurrence, Dr. Hughes said.
In PRIME II, postoperative whole-breast radiotherapy after breast-conserving surgery and adjuvant endocrine treatment provided some reduction in the local recurrence at 5-year follow-up, but the investigators concluded that the rate of ipsilateral breast tumor recurrence “is probably low enough for omission of radiotherapy to be considered for some patients (Lancet. 2015;16[3]:266-73).
The findings in older women are clear with the respect to the need for avoiding radiation therapy in those with low local recurrence risk, and the findings in younger women are promising, he concluded.
Dr. Hughes is on the speakers bureau for Myriad Genetics, is a shareholder of the healthcare IT company 5AM Solutions, and is the founder (with a financial interest) of Hughes Risk Apps, LLC.
EXPERT ANALYSIS FROM MBCC
First U.S. uterus transplant raises questions about ethics, cost
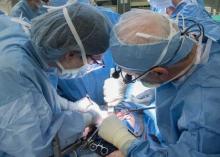
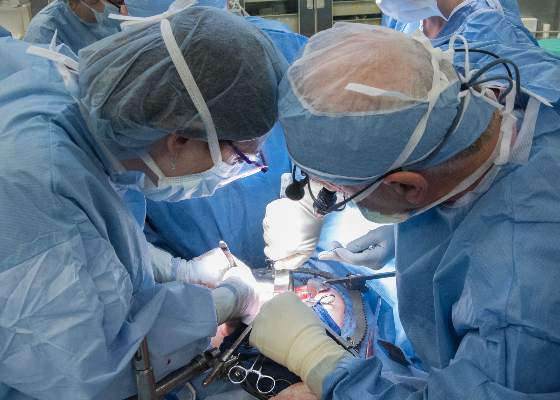
The recipient of the first uterus transplant performed in the United States is recovering well and looking forward to the next phase of the research project: pregnancy, according to the team of Cleveland Clinic surgeons who performed the groundbreaking Feb. 24 transplant.
“I’m pleased to report to you that our patient is doing very well,” lead surgeon Dr. Andreas G. Tzakis said during a press conference on March 7.
The transplant recipient, known only as “Lindsey” to protect her privacy, is a 26-year-old woman with uterine factor infertility (UFI). She was selected from among more than 250 applicants to undergo the 9-hour procedure involving transplantation of a uterus from a deceased organ donor of reproductive age. She is the first of 10 women set to undergo the procedure as part of the Cleveland Clinic study.
“Lindsey” made an appearance at the press conference and expressed, first and foremost, her “immense gratitude” to the donor’s family.
“They have provided me with a gift that I will never be able to repay,” she said in an emotional statement in which she shared about learning at age 16 that she would never be able to have children.
“From that moment on I prayed that God would allow me the opportunity to experience pregnancy, and here we are today at the beginning of that journey,” she said.
Lindsey will undergo a year of anti-rejection treatment prior to undergoing in vitro fertilization; her eggs were previously fertilized using her husband’s sperm, and the embryos are in cryogenic storage, according to Dr. Rebecca Flyckt, another member of the surgical team.
The embryos will be transferred one by one until the goal of a healthy pregnancy and healthy baby delivered by cesarean section is achieved, Dr. Flyckt said.
After one or two successful pregnancies and births, the uterus will be removed. Although substantial evidence suggests that anti-rejection medications are relatively safe in pregnancy, minimizing and ultimately eliminating the need for them is advisable, thus uterus transplants, at least under the Cleveland Clinic research protocol, are meant to be temporary.
Uterus transplants are performed to enable patients – who have either lost their uterus to disease or who were born without a uterus or a functioning uterus – to experience pregnancy and childbirth.
Prior to the Cleveland Clinic transplant, nine had been performed successfully with healthy outcomes (the first post-transplant baby was born healthy in 2014). All were performed at the University of Gothenburg in Sweden. Dr. Tzakis traveled there to work with surgeons prior to performing the procedure at the Cleveland Clinic, where he is the Transplant Center program director.
Ethical issues
Addressing the bioethical issues surrounding uterine transplant was an important part of this project, according to team member Dr. Ruth Farrell, a bioethicist and ob.gyn. surgeon at the Cleveland Clinic.
“Despite the name uterine transplant, the focus of this procedure is not on the uterus. It’s on women and children and families,” she said, adding that to understand the ethical issues of uterine transplant, it’s important to consider the perspectives of women with UFI.
“For instance, women born without a uterus have a medical condition that affects every aspect of their lives, from the time they are diagnosed in adolescence, to when they are adults looking for relationships and trying to decide if and how to have a family,” she said, adding that “these women face the real possibility of never having children.”
But this advance in reproductive medicine and science also requires a close look at how the potential risks and benefits of uterine transplant weigh against existing options of adoption and surrogacy. While there are many successful stories involving surrogacy and adoption, these options are not available to all women because of “legal, cultural, religious, and other very personal reasons,” Dr. Farrell said.
“Our research on uterine transplant, we hope, may give women another option which may work better for them and their families,” she said, also noting that while living donor transplants have been performed, these come with some risk, thus deceased donors are being used for the current study.
Insurance coverage
As for the feasibility of uterine transplant, many questions remain to be answered, according to Dr. Tommaso Falcone, chair of the department of obstetrics and gynecology at the Cleveland Clinic, who said that the procedures done as part of the current study will be paid for by an institutional grant.
While the American Society for Reproductive Medicine contends that infertility is a disease worthy of insurance coverage, that is “outside of our hands,” he said.
Indeed, while this first uterine transplant in the United States is a “huge and exciting breakthrough,” and while “the folks at Cleveland Clinic should be congratulated,” the possibility of this procedure ever being covered by insurance and being available to women outside of the research protocol is questionable, especially given the available alternatives, such as gestational carriers and surrogacy, Dr. David A. Forstein, a reproductive endocrinologist at the University of South Carolina, Greenville, said in an interview.
In a patient-centered model, this “tremendous, wonderful gift from science” would give patients – like the 1 in 5,000 women in the United States who are born without a uterus – a very viable alternative, but the question is whether having one’s own children is a right, and whether extensive financial resources should be committed to helping women achieve that, he said.
Dr. Charles E. Miller, a reproductive endocrinologist in Chicago, and head of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital in Park Ridge, Ill., agreed that this transplant is to be celebrated.
“It’s a big moment, to say the least,” he said in an interview.
But Dr. Miller also questioned the feasibility of the procedure and whether society will “look favorably upon donation,” given the availability of alternatives for which there is now great success.
“I salute the pioneering effort,” he said, “But at the end of the day, can society take on this burden of a procedure performed not to sustain life, but to help create life? That’s a tough one.”
The recipient of the first uterus transplant performed in the United States is recovering well and looking forward to the next phase of the research project: pregnancy, according to the team of Cleveland Clinic surgeons who performed the groundbreaking Feb. 24 transplant.
“I’m pleased to report to you that our patient is doing very well,” lead surgeon Dr. Andreas G. Tzakis said during a press conference on March 7.
The transplant recipient, known only as “Lindsey” to protect her privacy, is a 26-year-old woman with uterine factor infertility (UFI). She was selected from among more than 250 applicants to undergo the 9-hour procedure involving transplantation of a uterus from a deceased organ donor of reproductive age. She is the first of 10 women set to undergo the procedure as part of the Cleveland Clinic study.
“Lindsey” made an appearance at the press conference and expressed, first and foremost, her “immense gratitude” to the donor’s family.
“They have provided me with a gift that I will never be able to repay,” she said in an emotional statement in which she shared about learning at age 16 that she would never be able to have children.
“From that moment on I prayed that God would allow me the opportunity to experience pregnancy, and here we are today at the beginning of that journey,” she said.
Lindsey will undergo a year of anti-rejection treatment prior to undergoing in vitro fertilization; her eggs were previously fertilized using her husband’s sperm, and the embryos are in cryogenic storage, according to Dr. Rebecca Flyckt, another member of the surgical team.
The embryos will be transferred one by one until the goal of a healthy pregnancy and healthy baby delivered by cesarean section is achieved, Dr. Flyckt said.
After one or two successful pregnancies and births, the uterus will be removed. Although substantial evidence suggests that anti-rejection medications are relatively safe in pregnancy, minimizing and ultimately eliminating the need for them is advisable, thus uterus transplants, at least under the Cleveland Clinic research protocol, are meant to be temporary.
Uterus transplants are performed to enable patients – who have either lost their uterus to disease or who were born without a uterus or a functioning uterus – to experience pregnancy and childbirth.
Prior to the Cleveland Clinic transplant, nine had been performed successfully with healthy outcomes (the first post-transplant baby was born healthy in 2014). All were performed at the University of Gothenburg in Sweden. Dr. Tzakis traveled there to work with surgeons prior to performing the procedure at the Cleveland Clinic, where he is the Transplant Center program director.
Ethical issues
Addressing the bioethical issues surrounding uterine transplant was an important part of this project, according to team member Dr. Ruth Farrell, a bioethicist and ob.gyn. surgeon at the Cleveland Clinic.
“Despite the name uterine transplant, the focus of this procedure is not on the uterus. It’s on women and children and families,” she said, adding that to understand the ethical issues of uterine transplant, it’s important to consider the perspectives of women with UFI.
“For instance, women born without a uterus have a medical condition that affects every aspect of their lives, from the time they are diagnosed in adolescence, to when they are adults looking for relationships and trying to decide if and how to have a family,” she said, adding that “these women face the real possibility of never having children.”
But this advance in reproductive medicine and science also requires a close look at how the potential risks and benefits of uterine transplant weigh against existing options of adoption and surrogacy. While there are many successful stories involving surrogacy and adoption, these options are not available to all women because of “legal, cultural, religious, and other very personal reasons,” Dr. Farrell said.
“Our research on uterine transplant, we hope, may give women another option which may work better for them and their families,” she said, also noting that while living donor transplants have been performed, these come with some risk, thus deceased donors are being used for the current study.
Insurance coverage
As for the feasibility of uterine transplant, many questions remain to be answered, according to Dr. Tommaso Falcone, chair of the department of obstetrics and gynecology at the Cleveland Clinic, who said that the procedures done as part of the current study will be paid for by an institutional grant.
While the American Society for Reproductive Medicine contends that infertility is a disease worthy of insurance coverage, that is “outside of our hands,” he said.
Indeed, while this first uterine transplant in the United States is a “huge and exciting breakthrough,” and while “the folks at Cleveland Clinic should be congratulated,” the possibility of this procedure ever being covered by insurance and being available to women outside of the research protocol is questionable, especially given the available alternatives, such as gestational carriers and surrogacy, Dr. David A. Forstein, a reproductive endocrinologist at the University of South Carolina, Greenville, said in an interview.
In a patient-centered model, this “tremendous, wonderful gift from science” would give patients – like the 1 in 5,000 women in the United States who are born without a uterus – a very viable alternative, but the question is whether having one’s own children is a right, and whether extensive financial resources should be committed to helping women achieve that, he said.
Dr. Charles E. Miller, a reproductive endocrinologist in Chicago, and head of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital in Park Ridge, Ill., agreed that this transplant is to be celebrated.
“It’s a big moment, to say the least,” he said in an interview.
But Dr. Miller also questioned the feasibility of the procedure and whether society will “look favorably upon donation,” given the availability of alternatives for which there is now great success.
“I salute the pioneering effort,” he said, “But at the end of the day, can society take on this burden of a procedure performed not to sustain life, but to help create life? That’s a tough one.”
The recipient of the first uterus transplant performed in the United States is recovering well and looking forward to the next phase of the research project: pregnancy, according to the team of Cleveland Clinic surgeons who performed the groundbreaking Feb. 24 transplant.
“I’m pleased to report to you that our patient is doing very well,” lead surgeon Dr. Andreas G. Tzakis said during a press conference on March 7.
The transplant recipient, known only as “Lindsey” to protect her privacy, is a 26-year-old woman with uterine factor infertility (UFI). She was selected from among more than 250 applicants to undergo the 9-hour procedure involving transplantation of a uterus from a deceased organ donor of reproductive age. She is the first of 10 women set to undergo the procedure as part of the Cleveland Clinic study.
“Lindsey” made an appearance at the press conference and expressed, first and foremost, her “immense gratitude” to the donor’s family.
“They have provided me with a gift that I will never be able to repay,” she said in an emotional statement in which she shared about learning at age 16 that she would never be able to have children.
“From that moment on I prayed that God would allow me the opportunity to experience pregnancy, and here we are today at the beginning of that journey,” she said.
Lindsey will undergo a year of anti-rejection treatment prior to undergoing in vitro fertilization; her eggs were previously fertilized using her husband’s sperm, and the embryos are in cryogenic storage, according to Dr. Rebecca Flyckt, another member of the surgical team.
The embryos will be transferred one by one until the goal of a healthy pregnancy and healthy baby delivered by cesarean section is achieved, Dr. Flyckt said.
After one or two successful pregnancies and births, the uterus will be removed. Although substantial evidence suggests that anti-rejection medications are relatively safe in pregnancy, minimizing and ultimately eliminating the need for them is advisable, thus uterus transplants, at least under the Cleveland Clinic research protocol, are meant to be temporary.
Uterus transplants are performed to enable patients – who have either lost their uterus to disease or who were born without a uterus or a functioning uterus – to experience pregnancy and childbirth.
Prior to the Cleveland Clinic transplant, nine had been performed successfully with healthy outcomes (the first post-transplant baby was born healthy in 2014). All were performed at the University of Gothenburg in Sweden. Dr. Tzakis traveled there to work with surgeons prior to performing the procedure at the Cleveland Clinic, where he is the Transplant Center program director.
Ethical issues
Addressing the bioethical issues surrounding uterine transplant was an important part of this project, according to team member Dr. Ruth Farrell, a bioethicist and ob.gyn. surgeon at the Cleveland Clinic.
“Despite the name uterine transplant, the focus of this procedure is not on the uterus. It’s on women and children and families,” she said, adding that to understand the ethical issues of uterine transplant, it’s important to consider the perspectives of women with UFI.
“For instance, women born without a uterus have a medical condition that affects every aspect of their lives, from the time they are diagnosed in adolescence, to when they are adults looking for relationships and trying to decide if and how to have a family,” she said, adding that “these women face the real possibility of never having children.”
But this advance in reproductive medicine and science also requires a close look at how the potential risks and benefits of uterine transplant weigh against existing options of adoption and surrogacy. While there are many successful stories involving surrogacy and adoption, these options are not available to all women because of “legal, cultural, religious, and other very personal reasons,” Dr. Farrell said.
“Our research on uterine transplant, we hope, may give women another option which may work better for them and their families,” she said, also noting that while living donor transplants have been performed, these come with some risk, thus deceased donors are being used for the current study.
Insurance coverage
As for the feasibility of uterine transplant, many questions remain to be answered, according to Dr. Tommaso Falcone, chair of the department of obstetrics and gynecology at the Cleveland Clinic, who said that the procedures done as part of the current study will be paid for by an institutional grant.
While the American Society for Reproductive Medicine contends that infertility is a disease worthy of insurance coverage, that is “outside of our hands,” he said.
Indeed, while this first uterine transplant in the United States is a “huge and exciting breakthrough,” and while “the folks at Cleveland Clinic should be congratulated,” the possibility of this procedure ever being covered by insurance and being available to women outside of the research protocol is questionable, especially given the available alternatives, such as gestational carriers and surrogacy, Dr. David A. Forstein, a reproductive endocrinologist at the University of South Carolina, Greenville, said in an interview.
In a patient-centered model, this “tremendous, wonderful gift from science” would give patients – like the 1 in 5,000 women in the United States who are born without a uterus – a very viable alternative, but the question is whether having one’s own children is a right, and whether extensive financial resources should be committed to helping women achieve that, he said.
Dr. Charles E. Miller, a reproductive endocrinologist in Chicago, and head of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital in Park Ridge, Ill., agreed that this transplant is to be celebrated.
“It’s a big moment, to say the least,” he said in an interview.
But Dr. Miller also questioned the feasibility of the procedure and whether society will “look favorably upon donation,” given the availability of alternatives for which there is now great success.
“I salute the pioneering effort,” he said, “But at the end of the day, can society take on this burden of a procedure performed not to sustain life, but to help create life? That’s a tough one.”
Evidence builds for mesenchymal stem cell therapy in MS
NEW ORLEANS – Repeated intrathecal administration of autologous mesenchymal bone marrow-derived stromal stem cells for the treatment of multiple sclerosis was safe and induced accelerated beneficial effects in some patients in an uncontrolled, prospective study.
Of 28 patients with either secondary progressive or relapsing-progressive MS who were experiencing severe clinical deterioration and failure to respond to first- and second-line immunomodulatory treatments, 25 experienced either stable or improved Expanded Disability Status Scale (EDSS) scores following autologous mesenchymal stem cell (MSC) injections. The mean score decreased from 6.76 at study entry to 6.57 at a mean follow-up of 3.6 years, Dr. Panayiota Petrou of Hadassah-Hebrew University Medical Center, Jerusalem, Israel, and her colleagues reported in a poster at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
In addition, 17 patients experienced improvements in at least one functional system of the EDSS, including 14 who experienced improved motor function, 5 who experienced improved speech/bulbar functions, 4 who experienced improved urinary functions, and 6 who experienced improved cerebellar function. Eight patients remained stable during the entire follow-up period.
In a prior pilot trial, intrathecal administration of MSCs was shown to be safe and provided “some indications of potentially clinically meaningful beneficial effects on the progression of the disease,” the investigators said.
The current study provides further support for those findings. It included patients who experienced severe clinical deterioration (at least 0.5-1 points in the EDSS) during the year prior to study enrollment, or who had at least one major relapse without sufficient recovery following steroid treatment. Study subjects had a mean age of 56 years and mean disease duration of 15.4 years. They received at least 2 courses and up to 10 injections with 1 million cells/kg; most received 2 (8 patients) or 3 (9 patients) injections, and they were followed for up to 6 years.
No serious side effects were observed during long-term follow-up after repeated intrathecal injections. Eight patients experienced headaches and/or fever in the hours and days after injection, and two experienced symptoms of encephalopathy, which resolved within a few hours. Also, one patient experienced back pain and one had neck rigidity, but no long-term side effects were reported, the investigators said.
Immunological follow-up showed a transient up-regulation of regulatory T cells and down-regulation of the proliferative ability of lymphocytes and of several immune activation surface markers for up to 3 months, they noted.
The investigators reported having no disclosures.
NEW ORLEANS – Repeated intrathecal administration of autologous mesenchymal bone marrow-derived stromal stem cells for the treatment of multiple sclerosis was safe and induced accelerated beneficial effects in some patients in an uncontrolled, prospective study.
Of 28 patients with either secondary progressive or relapsing-progressive MS who were experiencing severe clinical deterioration and failure to respond to first- and second-line immunomodulatory treatments, 25 experienced either stable or improved Expanded Disability Status Scale (EDSS) scores following autologous mesenchymal stem cell (MSC) injections. The mean score decreased from 6.76 at study entry to 6.57 at a mean follow-up of 3.6 years, Dr. Panayiota Petrou of Hadassah-Hebrew University Medical Center, Jerusalem, Israel, and her colleagues reported in a poster at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
In addition, 17 patients experienced improvements in at least one functional system of the EDSS, including 14 who experienced improved motor function, 5 who experienced improved speech/bulbar functions, 4 who experienced improved urinary functions, and 6 who experienced improved cerebellar function. Eight patients remained stable during the entire follow-up period.
In a prior pilot trial, intrathecal administration of MSCs was shown to be safe and provided “some indications of potentially clinically meaningful beneficial effects on the progression of the disease,” the investigators said.
The current study provides further support for those findings. It included patients who experienced severe clinical deterioration (at least 0.5-1 points in the EDSS) during the year prior to study enrollment, or who had at least one major relapse without sufficient recovery following steroid treatment. Study subjects had a mean age of 56 years and mean disease duration of 15.4 years. They received at least 2 courses and up to 10 injections with 1 million cells/kg; most received 2 (8 patients) or 3 (9 patients) injections, and they were followed for up to 6 years.
No serious side effects were observed during long-term follow-up after repeated intrathecal injections. Eight patients experienced headaches and/or fever in the hours and days after injection, and two experienced symptoms of encephalopathy, which resolved within a few hours. Also, one patient experienced back pain and one had neck rigidity, but no long-term side effects were reported, the investigators said.
Immunological follow-up showed a transient up-regulation of regulatory T cells and down-regulation of the proliferative ability of lymphocytes and of several immune activation surface markers for up to 3 months, they noted.
The investigators reported having no disclosures.
NEW ORLEANS – Repeated intrathecal administration of autologous mesenchymal bone marrow-derived stromal stem cells for the treatment of multiple sclerosis was safe and induced accelerated beneficial effects in some patients in an uncontrolled, prospective study.
Of 28 patients with either secondary progressive or relapsing-progressive MS who were experiencing severe clinical deterioration and failure to respond to first- and second-line immunomodulatory treatments, 25 experienced either stable or improved Expanded Disability Status Scale (EDSS) scores following autologous mesenchymal stem cell (MSC) injections. The mean score decreased from 6.76 at study entry to 6.57 at a mean follow-up of 3.6 years, Dr. Panayiota Petrou of Hadassah-Hebrew University Medical Center, Jerusalem, Israel, and her colleagues reported in a poster at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
In addition, 17 patients experienced improvements in at least one functional system of the EDSS, including 14 who experienced improved motor function, 5 who experienced improved speech/bulbar functions, 4 who experienced improved urinary functions, and 6 who experienced improved cerebellar function. Eight patients remained stable during the entire follow-up period.
In a prior pilot trial, intrathecal administration of MSCs was shown to be safe and provided “some indications of potentially clinically meaningful beneficial effects on the progression of the disease,” the investigators said.
The current study provides further support for those findings. It included patients who experienced severe clinical deterioration (at least 0.5-1 points in the EDSS) during the year prior to study enrollment, or who had at least one major relapse without sufficient recovery following steroid treatment. Study subjects had a mean age of 56 years and mean disease duration of 15.4 years. They received at least 2 courses and up to 10 injections with 1 million cells/kg; most received 2 (8 patients) or 3 (9 patients) injections, and they were followed for up to 6 years.
No serious side effects were observed during long-term follow-up after repeated intrathecal injections. Eight patients experienced headaches and/or fever in the hours and days after injection, and two experienced symptoms of encephalopathy, which resolved within a few hours. Also, one patient experienced back pain and one had neck rigidity, but no long-term side effects were reported, the investigators said.
Immunological follow-up showed a transient up-regulation of regulatory T cells and down-regulation of the proliferative ability of lymphocytes and of several immune activation surface markers for up to 3 months, they noted.
The investigators reported having no disclosures.
AT ACTRIMS FORUM 2016
Key clinical point: Repeated intrathecal administration of autologous mesenchymal bone marrow–derived stromal stem cells stabilized or improved EDSS scores in most MS patients at up to 6 years of follow-up.
Major finding: The mean EDSS score decreased from 6.76 at study entry to 6.57 at a mean follow-up of 3.6 years.
Data source: An uncontrolled, prospective study involving 28 MS patients.
Disclosures: The investigators reported having no disclosures.
Prenatal, Postpartum Education Program Improves Breastfeeding Duration
ATLANTA – Targeted education to increase breastfeeding duration among women with gestational diabetes was feasible and effective in a randomized controlled lifestyle intervention trial.
Of 100 women with gestational diabetes mellitus (GDM) who were enrolled in the trial and randomized, 21 and 27 in the experimental and control groups, respectively, attended a study visit. Those who received the intervention initiated breastfeeding earlier after birth than those who did not receive the intervention (37 minutes vs. 112 minutes).
Mothers in the intervention group were also more likely to report any breastfeeding at 6 weeks (85% vs. 59%) and exclusive breastfeeding at 6 weeks (38% vs. 16%), Dr. Alison Stuebe of the University of North Carolina at Chapel Hill reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
“When we considered duration of any breastfeeding, we found that participation in the intervention was associated with a markedly longer duration of any breastfeeding,” Dr. Stuebe said.
After adjusting for baseline differences, clinic site, and breastfeeding intention score, the hazard ratio for breastfeeding cessation was 0.40, she noted. For exclusive breastfeeding, the adjusted hazard ratio for cessation was 0.49.
Breastfeeding intensity at 6 weeks also differed in the experimental intervention and control group, with 62% and 36%, respectively, reporting high-intensity breastfeeding (greater than 80% of feedings) at that time point.
Women in the study were enrolled and randomized in the third trimester. Just more than half (52%) were African American, 31% were non-Hispanic white, 9% were American Indian or Alaskan Native, and 8% were multiracial.
Prior to giving birth, each attended a class that addressed the benefits of breastfeeding for both mothers and infants, the importance of skin-to-skin contact, feeding cues, and positioning. The women also received breastfeeding pillows and weekly text messages with breastfeeding tips. At 6 weeks postpartum, they participated in a 12-week nutrition, exercise, and coping skills program that included monthly meetings, and they continued this program on their own for an additional 3 months, Dr. Stuebe said.
The control group received usual care for gestational diabetes, and usual postpartum care. Outcomes were adjusted for race, as a greater proportion of patients in the treatment group were African American.
Women with GDM, which affects up to 9% of pregnancies, are at increased lifetime risk of developing metabolic disease. However, data increasingly suggest that breastfeeding attenuates this risk, Dr. Stuebe said, noting that Gunderson, et al. recently demonstrated a halving of the risk of progressing to type 2 diabetes among women who breastfed for at least 10 months, compared with those with shorter breastfeeding duration (Ann Intern Med. 2015 Dec 15;163(12):889-898. doi: 10.7326/M15-0807).
Given these findings, efforts to promote breastfeeding among women with GDM are particularly important, she said, concluding that the current findings support tailored breastfeeding interventions among women with GDM.
Though limited by factors such as higher-than-expected–loss to follow-up, baseline differences in the two groups, and self-reported breastfeeding intensity outcomes, the study also has several strengths including that it was based on an approach used in an effective randomized controlled trial, it was pragmatic, and it used an intervention that could be incorporated into standard care.
“Our results suggest that targeted breastfeeding support for women with GDM may be feasible and efficacious,” Dr. Stuebe said. “Further studies are needed to test the effectiveness of this strategy in diverse clinical settings. However, if such interventions are found to be effective, breastfeeding support in women with GDM may improve health outcomes across two generations.”
The study was sponsored by the University of North Carolina at Chapel Hill, with support from the National Institute of Diabetes and Digestive and Kidney Diseases.
ATLANTA – Targeted education to increase breastfeeding duration among women with gestational diabetes was feasible and effective in a randomized controlled lifestyle intervention trial.
Of 100 women with gestational diabetes mellitus (GDM) who were enrolled in the trial and randomized, 21 and 27 in the experimental and control groups, respectively, attended a study visit. Those who received the intervention initiated breastfeeding earlier after birth than those who did not receive the intervention (37 minutes vs. 112 minutes).
Mothers in the intervention group were also more likely to report any breastfeeding at 6 weeks (85% vs. 59%) and exclusive breastfeeding at 6 weeks (38% vs. 16%), Dr. Alison Stuebe of the University of North Carolina at Chapel Hill reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
“When we considered duration of any breastfeeding, we found that participation in the intervention was associated with a markedly longer duration of any breastfeeding,” Dr. Stuebe said.
After adjusting for baseline differences, clinic site, and breastfeeding intention score, the hazard ratio for breastfeeding cessation was 0.40, she noted. For exclusive breastfeeding, the adjusted hazard ratio for cessation was 0.49.
Breastfeeding intensity at 6 weeks also differed in the experimental intervention and control group, with 62% and 36%, respectively, reporting high-intensity breastfeeding (greater than 80% of feedings) at that time point.
Women in the study were enrolled and randomized in the third trimester. Just more than half (52%) were African American, 31% were non-Hispanic white, 9% were American Indian or Alaskan Native, and 8% were multiracial.
Prior to giving birth, each attended a class that addressed the benefits of breastfeeding for both mothers and infants, the importance of skin-to-skin contact, feeding cues, and positioning. The women also received breastfeeding pillows and weekly text messages with breastfeeding tips. At 6 weeks postpartum, they participated in a 12-week nutrition, exercise, and coping skills program that included monthly meetings, and they continued this program on their own for an additional 3 months, Dr. Stuebe said.
The control group received usual care for gestational diabetes, and usual postpartum care. Outcomes were adjusted for race, as a greater proportion of patients in the treatment group were African American.
Women with GDM, which affects up to 9% of pregnancies, are at increased lifetime risk of developing metabolic disease. However, data increasingly suggest that breastfeeding attenuates this risk, Dr. Stuebe said, noting that Gunderson, et al. recently demonstrated a halving of the risk of progressing to type 2 diabetes among women who breastfed for at least 10 months, compared with those with shorter breastfeeding duration (Ann Intern Med. 2015 Dec 15;163(12):889-898. doi: 10.7326/M15-0807).
Given these findings, efforts to promote breastfeeding among women with GDM are particularly important, she said, concluding that the current findings support tailored breastfeeding interventions among women with GDM.
Though limited by factors such as higher-than-expected–loss to follow-up, baseline differences in the two groups, and self-reported breastfeeding intensity outcomes, the study also has several strengths including that it was based on an approach used in an effective randomized controlled trial, it was pragmatic, and it used an intervention that could be incorporated into standard care.
“Our results suggest that targeted breastfeeding support for women with GDM may be feasible and efficacious,” Dr. Stuebe said. “Further studies are needed to test the effectiveness of this strategy in diverse clinical settings. However, if such interventions are found to be effective, breastfeeding support in women with GDM may improve health outcomes across two generations.”
The study was sponsored by the University of North Carolina at Chapel Hill, with support from the National Institute of Diabetes and Digestive and Kidney Diseases.
ATLANTA – Targeted education to increase breastfeeding duration among women with gestational diabetes was feasible and effective in a randomized controlled lifestyle intervention trial.
Of 100 women with gestational diabetes mellitus (GDM) who were enrolled in the trial and randomized, 21 and 27 in the experimental and control groups, respectively, attended a study visit. Those who received the intervention initiated breastfeeding earlier after birth than those who did not receive the intervention (37 minutes vs. 112 minutes).
Mothers in the intervention group were also more likely to report any breastfeeding at 6 weeks (85% vs. 59%) and exclusive breastfeeding at 6 weeks (38% vs. 16%), Dr. Alison Stuebe of the University of North Carolina at Chapel Hill reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
“When we considered duration of any breastfeeding, we found that participation in the intervention was associated with a markedly longer duration of any breastfeeding,” Dr. Stuebe said.
After adjusting for baseline differences, clinic site, and breastfeeding intention score, the hazard ratio for breastfeeding cessation was 0.40, she noted. For exclusive breastfeeding, the adjusted hazard ratio for cessation was 0.49.
Breastfeeding intensity at 6 weeks also differed in the experimental intervention and control group, with 62% and 36%, respectively, reporting high-intensity breastfeeding (greater than 80% of feedings) at that time point.
Women in the study were enrolled and randomized in the third trimester. Just more than half (52%) were African American, 31% were non-Hispanic white, 9% were American Indian or Alaskan Native, and 8% were multiracial.
Prior to giving birth, each attended a class that addressed the benefits of breastfeeding for both mothers and infants, the importance of skin-to-skin contact, feeding cues, and positioning. The women also received breastfeeding pillows and weekly text messages with breastfeeding tips. At 6 weeks postpartum, they participated in a 12-week nutrition, exercise, and coping skills program that included monthly meetings, and they continued this program on their own for an additional 3 months, Dr. Stuebe said.
The control group received usual care for gestational diabetes, and usual postpartum care. Outcomes were adjusted for race, as a greater proportion of patients in the treatment group were African American.
Women with GDM, which affects up to 9% of pregnancies, are at increased lifetime risk of developing metabolic disease. However, data increasingly suggest that breastfeeding attenuates this risk, Dr. Stuebe said, noting that Gunderson, et al. recently demonstrated a halving of the risk of progressing to type 2 diabetes among women who breastfed for at least 10 months, compared with those with shorter breastfeeding duration (Ann Intern Med. 2015 Dec 15;163(12):889-898. doi: 10.7326/M15-0807).
Given these findings, efforts to promote breastfeeding among women with GDM are particularly important, she said, concluding that the current findings support tailored breastfeeding interventions among women with GDM.
Though limited by factors such as higher-than-expected–loss to follow-up, baseline differences in the two groups, and self-reported breastfeeding intensity outcomes, the study also has several strengths including that it was based on an approach used in an effective randomized controlled trial, it was pragmatic, and it used an intervention that could be incorporated into standard care.
“Our results suggest that targeted breastfeeding support for women with GDM may be feasible and efficacious,” Dr. Stuebe said. “Further studies are needed to test the effectiveness of this strategy in diverse clinical settings. However, if such interventions are found to be effective, breastfeeding support in women with GDM may improve health outcomes across two generations.”
The study was sponsored by the University of North Carolina at Chapel Hill, with support from the National Institute of Diabetes and Digestive and Kidney Diseases.
AT THE PREGNANCY MEETING
Prenatal, postpartum education program improves breastfeeding duration
ATLANTA – Targeted education to increase breastfeeding duration among women with gestational diabetes was feasible and effective in a randomized controlled lifestyle intervention trial.
Of 100 women with gestational diabetes mellitus (GDM) who were enrolled in the trial and randomized, 21 and 27 in the experimental and control groups, respectively, attended a study visit. Those who received the intervention initiated breastfeeding earlier after birth than those who did not receive the intervention (37 minutes vs. 112 minutes).
Mothers in the intervention group were also more likely to report any breastfeeding at 6 weeks (85% vs. 59%) and exclusive breastfeeding at 6 weeks (38% vs. 16%), Dr. Alison Stuebe of the University of North Carolina at Chapel Hill reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
“When we considered duration of any breastfeeding, we found that participation in the intervention was associated with a markedly longer duration of any breastfeeding,” Dr. Stuebe said.
After adjusting for baseline differences, clinic site, and breastfeeding intention score, the hazard ratio for breastfeeding cessation was 0.40, she noted. For exclusive breastfeeding, the adjusted hazard ratio for cessation was 0.49.
Breastfeeding intensity at 6 weeks also differed in the experimental intervention and control group, with 62% and 36%, respectively, reporting high-intensity breastfeeding (greater than 80% of feedings) at that time point.
Women in the study were enrolled and randomized in the third trimester. Just more than half (52%) were African American, 31% were non-Hispanic white, 9% were American Indian or Alaskan Native, and 8% were multiracial.
Prior to giving birth, each attended a class that addressed the benefits of breastfeeding for both mothers and infants, the importance of skin-to-skin contact, feeding cues, and positioning. The women also received breastfeeding pillows and weekly text messages with breastfeeding tips. At 6 weeks postpartum, they participated in a 12-week nutrition, exercise, and coping skills program that included monthly meetings, and they continued this program on their own for an additional 3 months, Dr. Stuebe said.
The control group received usual care for gestational diabetes, and usual postpartum care. Outcomes were adjusted for race, as a greater proportion of patients in the treatment group were African American.
Women with GDM, which affects up to 9% of pregnancies, are at increased lifetime risk of developing metabolic disease. However, data increasingly suggest that breastfeeding attenuates this risk, Dr. Stuebe said, noting that Gunderson, et al. recently demonstrated a halving of the risk of progressing to type 2 diabetes among women who breastfed for at least 10 months, compared with those with shorter breastfeeding duration (Ann Intern Med. 2015 Dec 15;163(12):889-898. doi: 10.7326/M15-0807).
Given these findings, efforts to promote breastfeeding among women with GDM are particularly important, she said, concluding that the current findings support tailored breastfeeding interventions among women with GDM.
Though limited by factors such as higher-than-expected–loss to follow-up, baseline differences in the two groups, and self-reported breastfeeding intensity outcomes, the study also has several strengths including that it was based on an approach used in an effective randomized controlled trial, it was pragmatic, and it used an intervention that could be incorporated into standard care.
“Our results suggest that targeted breastfeeding support for women with GDM may be feasible and efficacious,” Dr. Stuebe said. “Further studies are needed to test the effectiveness of this strategy in diverse clinical settings. However, if such interventions are found to be effective, breastfeeding support in women with GDM may improve health outcomes across two generations.”
The study was sponsored by the University of North Carolina at Chapel Hill, with support from the National Institute of Diabetes and Digestive and Kidney Diseases.
ATLANTA – Targeted education to increase breastfeeding duration among women with gestational diabetes was feasible and effective in a randomized controlled lifestyle intervention trial.
Of 100 women with gestational diabetes mellitus (GDM) who were enrolled in the trial and randomized, 21 and 27 in the experimental and control groups, respectively, attended a study visit. Those who received the intervention initiated breastfeeding earlier after birth than those who did not receive the intervention (37 minutes vs. 112 minutes).
Mothers in the intervention group were also more likely to report any breastfeeding at 6 weeks (85% vs. 59%) and exclusive breastfeeding at 6 weeks (38% vs. 16%), Dr. Alison Stuebe of the University of North Carolina at Chapel Hill reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
“When we considered duration of any breastfeeding, we found that participation in the intervention was associated with a markedly longer duration of any breastfeeding,” Dr. Stuebe said.
After adjusting for baseline differences, clinic site, and breastfeeding intention score, the hazard ratio for breastfeeding cessation was 0.40, she noted. For exclusive breastfeeding, the adjusted hazard ratio for cessation was 0.49.
Breastfeeding intensity at 6 weeks also differed in the experimental intervention and control group, with 62% and 36%, respectively, reporting high-intensity breastfeeding (greater than 80% of feedings) at that time point.
Women in the study were enrolled and randomized in the third trimester. Just more than half (52%) were African American, 31% were non-Hispanic white, 9% were American Indian or Alaskan Native, and 8% were multiracial.
Prior to giving birth, each attended a class that addressed the benefits of breastfeeding for both mothers and infants, the importance of skin-to-skin contact, feeding cues, and positioning. The women also received breastfeeding pillows and weekly text messages with breastfeeding tips. At 6 weeks postpartum, they participated in a 12-week nutrition, exercise, and coping skills program that included monthly meetings, and they continued this program on their own for an additional 3 months, Dr. Stuebe said.
The control group received usual care for gestational diabetes, and usual postpartum care. Outcomes were adjusted for race, as a greater proportion of patients in the treatment group were African American.
Women with GDM, which affects up to 9% of pregnancies, are at increased lifetime risk of developing metabolic disease. However, data increasingly suggest that breastfeeding attenuates this risk, Dr. Stuebe said, noting that Gunderson, et al. recently demonstrated a halving of the risk of progressing to type 2 diabetes among women who breastfed for at least 10 months, compared with those with shorter breastfeeding duration (Ann Intern Med. 2015 Dec 15;163(12):889-898. doi: 10.7326/M15-0807).
Given these findings, efforts to promote breastfeeding among women with GDM are particularly important, she said, concluding that the current findings support tailored breastfeeding interventions among women with GDM.
Though limited by factors such as higher-than-expected–loss to follow-up, baseline differences in the two groups, and self-reported breastfeeding intensity outcomes, the study also has several strengths including that it was based on an approach used in an effective randomized controlled trial, it was pragmatic, and it used an intervention that could be incorporated into standard care.
“Our results suggest that targeted breastfeeding support for women with GDM may be feasible and efficacious,” Dr. Stuebe said. “Further studies are needed to test the effectiveness of this strategy in diverse clinical settings. However, if such interventions are found to be effective, breastfeeding support in women with GDM may improve health outcomes across two generations.”
The study was sponsored by the University of North Carolina at Chapel Hill, with support from the National Institute of Diabetes and Digestive and Kidney Diseases.
ATLANTA – Targeted education to increase breastfeeding duration among women with gestational diabetes was feasible and effective in a randomized controlled lifestyle intervention trial.
Of 100 women with gestational diabetes mellitus (GDM) who were enrolled in the trial and randomized, 21 and 27 in the experimental and control groups, respectively, attended a study visit. Those who received the intervention initiated breastfeeding earlier after birth than those who did not receive the intervention (37 minutes vs. 112 minutes).
Mothers in the intervention group were also more likely to report any breastfeeding at 6 weeks (85% vs. 59%) and exclusive breastfeeding at 6 weeks (38% vs. 16%), Dr. Alison Stuebe of the University of North Carolina at Chapel Hill reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
“When we considered duration of any breastfeeding, we found that participation in the intervention was associated with a markedly longer duration of any breastfeeding,” Dr. Stuebe said.
After adjusting for baseline differences, clinic site, and breastfeeding intention score, the hazard ratio for breastfeeding cessation was 0.40, she noted. For exclusive breastfeeding, the adjusted hazard ratio for cessation was 0.49.
Breastfeeding intensity at 6 weeks also differed in the experimental intervention and control group, with 62% and 36%, respectively, reporting high-intensity breastfeeding (greater than 80% of feedings) at that time point.
Women in the study were enrolled and randomized in the third trimester. Just more than half (52%) were African American, 31% were non-Hispanic white, 9% were American Indian or Alaskan Native, and 8% were multiracial.
Prior to giving birth, each attended a class that addressed the benefits of breastfeeding for both mothers and infants, the importance of skin-to-skin contact, feeding cues, and positioning. The women also received breastfeeding pillows and weekly text messages with breastfeeding tips. At 6 weeks postpartum, they participated in a 12-week nutrition, exercise, and coping skills program that included monthly meetings, and they continued this program on their own for an additional 3 months, Dr. Stuebe said.
The control group received usual care for gestational diabetes, and usual postpartum care. Outcomes were adjusted for race, as a greater proportion of patients in the treatment group were African American.
Women with GDM, which affects up to 9% of pregnancies, are at increased lifetime risk of developing metabolic disease. However, data increasingly suggest that breastfeeding attenuates this risk, Dr. Stuebe said, noting that Gunderson, et al. recently demonstrated a halving of the risk of progressing to type 2 diabetes among women who breastfed for at least 10 months, compared with those with shorter breastfeeding duration (Ann Intern Med. 2015 Dec 15;163(12):889-898. doi: 10.7326/M15-0807).
Given these findings, efforts to promote breastfeeding among women with GDM are particularly important, she said, concluding that the current findings support tailored breastfeeding interventions among women with GDM.
Though limited by factors such as higher-than-expected–loss to follow-up, baseline differences in the two groups, and self-reported breastfeeding intensity outcomes, the study also has several strengths including that it was based on an approach used in an effective randomized controlled trial, it was pragmatic, and it used an intervention that could be incorporated into standard care.
“Our results suggest that targeted breastfeeding support for women with GDM may be feasible and efficacious,” Dr. Stuebe said. “Further studies are needed to test the effectiveness of this strategy in diverse clinical settings. However, if such interventions are found to be effective, breastfeeding support in women with GDM may improve health outcomes across two generations.”
The study was sponsored by the University of North Carolina at Chapel Hill, with support from the National Institute of Diabetes and Digestive and Kidney Diseases.
AT THE PREGNANCY MEETING
Key clinical point: Targeted education is effective at increasing breastfeeding duration among women with gestational diabetes.
Major finding: Women in the intervention group initiated breastfeeding earlier after birth (37 minutes vs. 112 minutes), and breastfed for longer (85% vs. 59% reported any breastfeeding at 6 weeks).
Data source: A randomized intervention trial involving 100 women.
Disclosures: This study was sponsored by the University of North Carolina at Chapel Hill with support from the National Institute of Diabetes and Digestive and Kidney Diseases.
BENEFIT 11: No new safety signals with interferon beta-1b
NEW ORLEANS – The long-term safety of interferon beta-1b for the treatment of multiple sclerosis is well established, and 11-year outcomes from the randomized, placebo-controlled BENEFIT trial (BENEFIT 11) provide further support for the existing data.
Eleven years after patients with clinically isolated syndrome were randomized to receive 250 mcg every other day or placebo (with an option at 2 years to switch to active treatment), no new safety signals were found, and the safety profile remained favorable, Dr. Mark S. Freedman of the University of Ottawa and his colleagues reported in a poster at the meeting sponsored by the Americas Committee for Treatment and Research in Multiple Sclerosis.
In the 278 of the 468 patients originally enrolled in BENEFIT who were evaluated at 11 years, 107 (38.5%) reported one or more safety events (total, 278). Interferon beta-1b was used continuously during the 2 years prior to the 11-year evaluation in 82 patients (29.5%), and 28 (34.1%) of those reported an adverse event or medical history event (total, 72 events). Of 196 patients not treated continuously with interferon beta-1b in the 2 years prior to BENEFIT 11, 79 (40.3%) reported an adverse event or medical history event (total, 206 events), the investigators said.
The events reported in BENEFIT 11 were consistent with those seen previously in interferon beta-1b–treated patients; back pain was the most commonly reported musculoskeletal/connective tissue adverse event (reported by 2.4% of patients in the continuous treatment group and 2.6% in the remaining patients, and headache was the most common neurologic adverse event (reported by 1.2% and 2.0% of patients in the groups, respectively).
Neoplasms occurred in 3.7% and 3.6%; most were benign.
“Overall, the safety results from BENEFIT 11 supported the already well-established long-term safety profile of interferon beta-1b,” the investigators wrote.
The higher rates of some events in patients without continuous interferon beta-1b treatment may be due to escalation to second-line therapy in some patients who required agents that may be associated with greater frequency of safety events, they explained.
This study was supported by Bayer. Dr. Freedman has received compensation from Actelion, Bayer, Biogen Idec, Chugai, EMD Canada, Genzyme, Merck Serono, Novartis, Roche Canada, Sanofi-Aventis, and Teva Canada Innovation for consulting services. He also participates in a Genzyme-sponsored speakers bureau.
NEW ORLEANS – The long-term safety of interferon beta-1b for the treatment of multiple sclerosis is well established, and 11-year outcomes from the randomized, placebo-controlled BENEFIT trial (BENEFIT 11) provide further support for the existing data.
Eleven years after patients with clinically isolated syndrome were randomized to receive 250 mcg every other day or placebo (with an option at 2 years to switch to active treatment), no new safety signals were found, and the safety profile remained favorable, Dr. Mark S. Freedman of the University of Ottawa and his colleagues reported in a poster at the meeting sponsored by the Americas Committee for Treatment and Research in Multiple Sclerosis.
In the 278 of the 468 patients originally enrolled in BENEFIT who were evaluated at 11 years, 107 (38.5%) reported one or more safety events (total, 278). Interferon beta-1b was used continuously during the 2 years prior to the 11-year evaluation in 82 patients (29.5%), and 28 (34.1%) of those reported an adverse event or medical history event (total, 72 events). Of 196 patients not treated continuously with interferon beta-1b in the 2 years prior to BENEFIT 11, 79 (40.3%) reported an adverse event or medical history event (total, 206 events), the investigators said.
The events reported in BENEFIT 11 were consistent with those seen previously in interferon beta-1b–treated patients; back pain was the most commonly reported musculoskeletal/connective tissue adverse event (reported by 2.4% of patients in the continuous treatment group and 2.6% in the remaining patients, and headache was the most common neurologic adverse event (reported by 1.2% and 2.0% of patients in the groups, respectively).
Neoplasms occurred in 3.7% and 3.6%; most were benign.
“Overall, the safety results from BENEFIT 11 supported the already well-established long-term safety profile of interferon beta-1b,” the investigators wrote.
The higher rates of some events in patients without continuous interferon beta-1b treatment may be due to escalation to second-line therapy in some patients who required agents that may be associated with greater frequency of safety events, they explained.
This study was supported by Bayer. Dr. Freedman has received compensation from Actelion, Bayer, Biogen Idec, Chugai, EMD Canada, Genzyme, Merck Serono, Novartis, Roche Canada, Sanofi-Aventis, and Teva Canada Innovation for consulting services. He also participates in a Genzyme-sponsored speakers bureau.
NEW ORLEANS – The long-term safety of interferon beta-1b for the treatment of multiple sclerosis is well established, and 11-year outcomes from the randomized, placebo-controlled BENEFIT trial (BENEFIT 11) provide further support for the existing data.
Eleven years after patients with clinically isolated syndrome were randomized to receive 250 mcg every other day or placebo (with an option at 2 years to switch to active treatment), no new safety signals were found, and the safety profile remained favorable, Dr. Mark S. Freedman of the University of Ottawa and his colleagues reported in a poster at the meeting sponsored by the Americas Committee for Treatment and Research in Multiple Sclerosis.
In the 278 of the 468 patients originally enrolled in BENEFIT who were evaluated at 11 years, 107 (38.5%) reported one or more safety events (total, 278). Interferon beta-1b was used continuously during the 2 years prior to the 11-year evaluation in 82 patients (29.5%), and 28 (34.1%) of those reported an adverse event or medical history event (total, 72 events). Of 196 patients not treated continuously with interferon beta-1b in the 2 years prior to BENEFIT 11, 79 (40.3%) reported an adverse event or medical history event (total, 206 events), the investigators said.
The events reported in BENEFIT 11 were consistent with those seen previously in interferon beta-1b–treated patients; back pain was the most commonly reported musculoskeletal/connective tissue adverse event (reported by 2.4% of patients in the continuous treatment group and 2.6% in the remaining patients, and headache was the most common neurologic adverse event (reported by 1.2% and 2.0% of patients in the groups, respectively).
Neoplasms occurred in 3.7% and 3.6%; most were benign.
“Overall, the safety results from BENEFIT 11 supported the already well-established long-term safety profile of interferon beta-1b,” the investigators wrote.
The higher rates of some events in patients without continuous interferon beta-1b treatment may be due to escalation to second-line therapy in some patients who required agents that may be associated with greater frequency of safety events, they explained.
This study was supported by Bayer. Dr. Freedman has received compensation from Actelion, Bayer, Biogen Idec, Chugai, EMD Canada, Genzyme, Merck Serono, Novartis, Roche Canada, Sanofi-Aventis, and Teva Canada Innovation for consulting services. He also participates in a Genzyme-sponsored speakers bureau.
AT ACTRIMS FORUM 2016
Key clinical point: The long-term safety of interferon beta-1b for the treatment of multiple sclerosis is well established, and 11-year outcomes from the randomized, placebo-controlled BENEFIT trial (BENEFIT 11) provide further support for the existing data.
Major finding: 107 of 278 patients reported at least one safety event; no new safety signals were found.
Data source: The 11-year follow-up of 278 patients from the randomized controlled BENEFIT Trial.
Disclosures: This study was supported by Bayer. Dr. Freedman has received compensation from Actelion, Bayer, Biogen Idec, Chugai, EMD Canada, Genzyme, Merck Serono, Novartis, Roche Canada, Sanofi-Aventis, and Teva Canada Innovation for consulting services. He also participates in a Genzyme-sponsored speakers bureau.
Twin Birth Study: No benefit with planned C-section
ATLANTA – A policy of planned cesarean delivery provides no significant benefit, compared with a policy of planned vaginal delivery in cases involving uncomplicated twin pregnancies between 32 and 38 weeks gestation where the first twin is in cephalic presentation, according to 2-year findings from the Twin Birth Study.
Prior findings from the randomized trial demonstrated that a policy of planned cesarean delivery benefited neither the mother nor the baby, compared with a policy of planned vaginal delivery; there was no difference between the groups with respect to the primary outcome of fetal or neonatal death or serious neonatal morbidity. The current analysis looked at the secondary outcome – a composite outcome of death or neurodevelopmental delay in offspring at age 2 years.
This composite outcome, corrected for gestational age at birth, occurred in 5.99% of 2,320 planned cesarean deliveries and in 5.83% of 2,283 planned vaginal deliveries (odds ratio, 1.04), Dr. Elizabeth Asztalos of the University of Toronto reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
There also was no significant difference between the groups in the individual components of the composite outcome. Death occurred in 1.51% and 1.01% of deliveries in the planned cesarean and planned vaginal delivery groups, respectively (OR, 1.48). In the surviving children, neurodevelopmental delay occurred in 4.55% and 4.87% (OR, 0.95), Dr. Asztalos said.
The Twin Birth study enrolled only women with uncomplicated twin pregnancies between 32 and 38 weeks’ gestation with the first twin in cephalic presentation. Subjects were randomized to either a planned cesarean or planned vaginal delivery group, and for the current analysis, children were assessed, using the Ages and Stages Questionnaire, at a mean age of 25-26 months in both groups.
Abnormal findings were validated by a clinical neurodevelopmental assessment.
The rate of cerebral palsy was extremely low in the study population and was not amenable to analysis, Dr. Asztalos said.
A post hoc analysis to determine if there were any differences between twin A and twin B with respect to the secondary outcome measure, showed no significant difference based on birth order, she added.
Notably, the incidence of the primary outcome in the study was almost threefold higher than the 2% originally anticipated in the planned vaginal delivery group. This may be due to the fact that for both approaches to delivery management, just under half of the infants were born preterm, she said.
The 2-year follow-up of the Twin Birth Study reinforces the initial findings of the international study, she said.
The Twin Birth Study was sponsored by Sunnybrook Health Sciences Centre in Toronto.
ATLANTA – A policy of planned cesarean delivery provides no significant benefit, compared with a policy of planned vaginal delivery in cases involving uncomplicated twin pregnancies between 32 and 38 weeks gestation where the first twin is in cephalic presentation, according to 2-year findings from the Twin Birth Study.
Prior findings from the randomized trial demonstrated that a policy of planned cesarean delivery benefited neither the mother nor the baby, compared with a policy of planned vaginal delivery; there was no difference between the groups with respect to the primary outcome of fetal or neonatal death or serious neonatal morbidity. The current analysis looked at the secondary outcome – a composite outcome of death or neurodevelopmental delay in offspring at age 2 years.
This composite outcome, corrected for gestational age at birth, occurred in 5.99% of 2,320 planned cesarean deliveries and in 5.83% of 2,283 planned vaginal deliveries (odds ratio, 1.04), Dr. Elizabeth Asztalos of the University of Toronto reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
There also was no significant difference between the groups in the individual components of the composite outcome. Death occurred in 1.51% and 1.01% of deliveries in the planned cesarean and planned vaginal delivery groups, respectively (OR, 1.48). In the surviving children, neurodevelopmental delay occurred in 4.55% and 4.87% (OR, 0.95), Dr. Asztalos said.
The Twin Birth study enrolled only women with uncomplicated twin pregnancies between 32 and 38 weeks’ gestation with the first twin in cephalic presentation. Subjects were randomized to either a planned cesarean or planned vaginal delivery group, and for the current analysis, children were assessed, using the Ages and Stages Questionnaire, at a mean age of 25-26 months in both groups.
Abnormal findings were validated by a clinical neurodevelopmental assessment.
The rate of cerebral palsy was extremely low in the study population and was not amenable to analysis, Dr. Asztalos said.
A post hoc analysis to determine if there were any differences between twin A and twin B with respect to the secondary outcome measure, showed no significant difference based on birth order, she added.
Notably, the incidence of the primary outcome in the study was almost threefold higher than the 2% originally anticipated in the planned vaginal delivery group. This may be due to the fact that for both approaches to delivery management, just under half of the infants were born preterm, she said.
The 2-year follow-up of the Twin Birth Study reinforces the initial findings of the international study, she said.
The Twin Birth Study was sponsored by Sunnybrook Health Sciences Centre in Toronto.
ATLANTA – A policy of planned cesarean delivery provides no significant benefit, compared with a policy of planned vaginal delivery in cases involving uncomplicated twin pregnancies between 32 and 38 weeks gestation where the first twin is in cephalic presentation, according to 2-year findings from the Twin Birth Study.
Prior findings from the randomized trial demonstrated that a policy of planned cesarean delivery benefited neither the mother nor the baby, compared with a policy of planned vaginal delivery; there was no difference between the groups with respect to the primary outcome of fetal or neonatal death or serious neonatal morbidity. The current analysis looked at the secondary outcome – a composite outcome of death or neurodevelopmental delay in offspring at age 2 years.
This composite outcome, corrected for gestational age at birth, occurred in 5.99% of 2,320 planned cesarean deliveries and in 5.83% of 2,283 planned vaginal deliveries (odds ratio, 1.04), Dr. Elizabeth Asztalos of the University of Toronto reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
There also was no significant difference between the groups in the individual components of the composite outcome. Death occurred in 1.51% and 1.01% of deliveries in the planned cesarean and planned vaginal delivery groups, respectively (OR, 1.48). In the surviving children, neurodevelopmental delay occurred in 4.55% and 4.87% (OR, 0.95), Dr. Asztalos said.
The Twin Birth study enrolled only women with uncomplicated twin pregnancies between 32 and 38 weeks’ gestation with the first twin in cephalic presentation. Subjects were randomized to either a planned cesarean or planned vaginal delivery group, and for the current analysis, children were assessed, using the Ages and Stages Questionnaire, at a mean age of 25-26 months in both groups.
Abnormal findings were validated by a clinical neurodevelopmental assessment.
The rate of cerebral palsy was extremely low in the study population and was not amenable to analysis, Dr. Asztalos said.
A post hoc analysis to determine if there were any differences between twin A and twin B with respect to the secondary outcome measure, showed no significant difference based on birth order, she added.
Notably, the incidence of the primary outcome in the study was almost threefold higher than the 2% originally anticipated in the planned vaginal delivery group. This may be due to the fact that for both approaches to delivery management, just under half of the infants were born preterm, she said.
The 2-year follow-up of the Twin Birth Study reinforces the initial findings of the international study, she said.
The Twin Birth Study was sponsored by Sunnybrook Health Sciences Centre in Toronto.
AT THE PREGNANCY MEETING
Key clinical point: Planned cesarean delivery provides no significant benefit in uncomplicated twin pregnancies, compared with planned vaginal delivery.
Major finding: The composite outcome occurred in 5.99% of 2,320 planned cesarean deliveries and in 5.83% of 2,283 planned vaginal deliveries (odds ratio, 1.04).
Data source: The randomized Twin Birth Study involving 4,603 children.
Disclosures: The Twin Birth Study was sponsored by Sunnybrook Health Sciences Centre in Toronto.
Study examines efficacy of small group programs for fatigue in MS
NEW ORLEANS – Fatigue: Take Control (FTC), a 6-week group education and behavioral program widely used to address fatigue in patients with multiple sclerosis, was comparable to a general MS education program for decreasing fatigue and improving self-efficacy in a randomized, controlled study.
In 218 subjects randomized to participate in either FTC or a general MS education program (MS: Take Control, or MSTC), Modified Fatigue Impact Scale scores improved significantly, compared with baseline and regardless of group assignment; there was no difference between the groups at visit 6 (mean change, –4.5 and –3.6 in the FTC and MSTC groups, respectively), Cinda L. Hugos of the VA Portland (Ore.) Health Care System and her colleagues reported at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
Multiple Sclerosis Self-Efficacy scale scores did, however, improve significantly in the FTC group vs. the MSTC group (mean increase of 45.5 vs. a decrease of 15.8), but the improvement was not maintained at 6 months, the investigators reported in a poster at the meeting.
The two groups had comparable baseline characteristics, with a mean age of nearly 54 years, as well as similar mean time since diagnosis (12.5 years), self-administered Expanded Disability Status Scale scores (5.2), and demographics.
Fatigue is common in MS patients, occurring in up to 95% of patients, and many report that it is the most disabling symptom. FTC, which addresses medical management of fatigue, exercise, environment, and changes and choices with respect to energy and fatigue control, is often used by chapters of the National MS Society, but its effectiveness for decreasing fatigue or improving self-efficacy has not been proven, the investigators noted.
To determine if FTC decreases fatigue and increases self-efficacy, compared with a general MS education program that addresses issues such as nutrition, emotional health, cognitive problems, and fitness, the investigators randomized subjects at four sites in groups of between 3 and 10 participants to either an FTC or MSTC group. All subjects had moderate to severe fatigue. Those with pregnancy, severe depression, uncontrolled problems that would limit participation, relapse in the prior month, or initiation of a new disease-modifying treatment within the prior 3 months were excluded.
“Both FTC and MSTC structured small group programs were associated with improved self-reported fatigue at program completion and at 3- and 6-month follow-up, but there were no significant differences in fatigue scores between FTC and MSTC participants at any time point,” the investigators wrote.
Self-efficacy was significantly better among FTC participants at program completion, but was not sustained, they noted, concluding that the findings suggest that “participating in structured small group programs is associated with prolonged reductions in fatigue in people with MS and that supporting goal setting provides short-term improvements in self-efficacy.”
Further research is needed to determine whether booster sessions would be beneficial for sustaining improvements in self-efficacy, they added.
This study was supported by the Rehabilitation, Research, & Development Service of the Veterans Affairs Office of Research & Development. The authors reported having no disclosures.
NEW ORLEANS – Fatigue: Take Control (FTC), a 6-week group education and behavioral program widely used to address fatigue in patients with multiple sclerosis, was comparable to a general MS education program for decreasing fatigue and improving self-efficacy in a randomized, controlled study.
In 218 subjects randomized to participate in either FTC or a general MS education program (MS: Take Control, or MSTC), Modified Fatigue Impact Scale scores improved significantly, compared with baseline and regardless of group assignment; there was no difference between the groups at visit 6 (mean change, –4.5 and –3.6 in the FTC and MSTC groups, respectively), Cinda L. Hugos of the VA Portland (Ore.) Health Care System and her colleagues reported at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
Multiple Sclerosis Self-Efficacy scale scores did, however, improve significantly in the FTC group vs. the MSTC group (mean increase of 45.5 vs. a decrease of 15.8), but the improvement was not maintained at 6 months, the investigators reported in a poster at the meeting.
The two groups had comparable baseline characteristics, with a mean age of nearly 54 years, as well as similar mean time since diagnosis (12.5 years), self-administered Expanded Disability Status Scale scores (5.2), and demographics.
Fatigue is common in MS patients, occurring in up to 95% of patients, and many report that it is the most disabling symptom. FTC, which addresses medical management of fatigue, exercise, environment, and changes and choices with respect to energy and fatigue control, is often used by chapters of the National MS Society, but its effectiveness for decreasing fatigue or improving self-efficacy has not been proven, the investigators noted.
To determine if FTC decreases fatigue and increases self-efficacy, compared with a general MS education program that addresses issues such as nutrition, emotional health, cognitive problems, and fitness, the investigators randomized subjects at four sites in groups of between 3 and 10 participants to either an FTC or MSTC group. All subjects had moderate to severe fatigue. Those with pregnancy, severe depression, uncontrolled problems that would limit participation, relapse in the prior month, or initiation of a new disease-modifying treatment within the prior 3 months were excluded.
“Both FTC and MSTC structured small group programs were associated with improved self-reported fatigue at program completion and at 3- and 6-month follow-up, but there were no significant differences in fatigue scores between FTC and MSTC participants at any time point,” the investigators wrote.
Self-efficacy was significantly better among FTC participants at program completion, but was not sustained, they noted, concluding that the findings suggest that “participating in structured small group programs is associated with prolonged reductions in fatigue in people with MS and that supporting goal setting provides short-term improvements in self-efficacy.”
Further research is needed to determine whether booster sessions would be beneficial for sustaining improvements in self-efficacy, they added.
This study was supported by the Rehabilitation, Research, & Development Service of the Veterans Affairs Office of Research & Development. The authors reported having no disclosures.
NEW ORLEANS – Fatigue: Take Control (FTC), a 6-week group education and behavioral program widely used to address fatigue in patients with multiple sclerosis, was comparable to a general MS education program for decreasing fatigue and improving self-efficacy in a randomized, controlled study.
In 218 subjects randomized to participate in either FTC or a general MS education program (MS: Take Control, or MSTC), Modified Fatigue Impact Scale scores improved significantly, compared with baseline and regardless of group assignment; there was no difference between the groups at visit 6 (mean change, –4.5 and –3.6 in the FTC and MSTC groups, respectively), Cinda L. Hugos of the VA Portland (Ore.) Health Care System and her colleagues reported at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
Multiple Sclerosis Self-Efficacy scale scores did, however, improve significantly in the FTC group vs. the MSTC group (mean increase of 45.5 vs. a decrease of 15.8), but the improvement was not maintained at 6 months, the investigators reported in a poster at the meeting.
The two groups had comparable baseline characteristics, with a mean age of nearly 54 years, as well as similar mean time since diagnosis (12.5 years), self-administered Expanded Disability Status Scale scores (5.2), and demographics.
Fatigue is common in MS patients, occurring in up to 95% of patients, and many report that it is the most disabling symptom. FTC, which addresses medical management of fatigue, exercise, environment, and changes and choices with respect to energy and fatigue control, is often used by chapters of the National MS Society, but its effectiveness for decreasing fatigue or improving self-efficacy has not been proven, the investigators noted.
To determine if FTC decreases fatigue and increases self-efficacy, compared with a general MS education program that addresses issues such as nutrition, emotional health, cognitive problems, and fitness, the investigators randomized subjects at four sites in groups of between 3 and 10 participants to either an FTC or MSTC group. All subjects had moderate to severe fatigue. Those with pregnancy, severe depression, uncontrolled problems that would limit participation, relapse in the prior month, or initiation of a new disease-modifying treatment within the prior 3 months were excluded.
“Both FTC and MSTC structured small group programs were associated with improved self-reported fatigue at program completion and at 3- and 6-month follow-up, but there were no significant differences in fatigue scores between FTC and MSTC participants at any time point,” the investigators wrote.
Self-efficacy was significantly better among FTC participants at program completion, but was not sustained, they noted, concluding that the findings suggest that “participating in structured small group programs is associated with prolonged reductions in fatigue in people with MS and that supporting goal setting provides short-term improvements in self-efficacy.”
Further research is needed to determine whether booster sessions would be beneficial for sustaining improvements in self-efficacy, they added.
This study was supported by the Rehabilitation, Research, & Development Service of the Veterans Affairs Office of Research & Development. The authors reported having no disclosures.
AT ACTRIMS FORUM 2016
Key clinical point: Fatigue: Take Control (FTC), a 6-week program widely used to address fatigue in patients with multiple sclerosis, was comparable to a general MS education program for decreasing fatigue and improving self-efficacy in a randomized, controlled study.
Major finding: FTC and a general MS education program improved Modified Fatigue Impact Scale scores significantly, with no difference between the groups at visit 6 (mean change, –4.5 and –3.6 in the groups, respectively).
Data source: A randomized, controlled trial of 218 MS patients.
Disclosures: This study was supported by the Rehabilitation, Research & Development Service of the VA Office of Research and Development. The authors reported having no disclosures.
Triage MS-related ED visits to reduce unnecessary treatment
NEW ORLEANS – The majority of multiple sclerosis–related emergency department visits in a recent chart review were related to pseudoflares or MS-related complications, rather than to true MS relapse.
The findings suggest that many diagnostic tests, treatments, and hospital admissions are unnecessary, Dr. Hesham Abboud of the Cleveland Clinic and his colleagues reported in a poster at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
Of 97 MS-related visits among 75 patients, 33 were for new neurologic symptoms, 29 were for worsening of preexisting symptoms, and 36 were for MS-related complications. New relapse was diagnosed in only 27 visits (27.8%), and urinary tract infections were found in about one-third of patients presenting with either urinary or neurologic symptoms, the investigators said.
New MRIs were ordered in 37 patients (38.1%); 89 emergency department visits (91.7%) resulted in hospital admissions and 40.4% were related to neurology; and steroid treatment was used in 24 visits (24.7%), 7 of which were for worsening of preexisting symptoms, the investigators said.
Of the visits involving new neurologic symptoms, one-third were not from relapse; 59% of the MRIs done in those situations were positive for enhancing or new lesions. Of the visits with worsening preexisting symptoms, only 16.6% were associated with a new relapse, and 28.5% of MRIs done in those situations were positive, the investigators said.
Although many ED visits among MS patients are driven by neurologic complaints, true relapse is rarely present, and not all those with true relapse require hospital admission and steroid treatment, they noted, concluding that developing a care path and triaging system for MS patients in the ED could prevent unnecessary MRIs, steroid treatment, and hospital admissions.
The investigators reported having no disclosures.
NEW ORLEANS – The majority of multiple sclerosis–related emergency department visits in a recent chart review were related to pseudoflares or MS-related complications, rather than to true MS relapse.
The findings suggest that many diagnostic tests, treatments, and hospital admissions are unnecessary, Dr. Hesham Abboud of the Cleveland Clinic and his colleagues reported in a poster at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
Of 97 MS-related visits among 75 patients, 33 were for new neurologic symptoms, 29 were for worsening of preexisting symptoms, and 36 were for MS-related complications. New relapse was diagnosed in only 27 visits (27.8%), and urinary tract infections were found in about one-third of patients presenting with either urinary or neurologic symptoms, the investigators said.
New MRIs were ordered in 37 patients (38.1%); 89 emergency department visits (91.7%) resulted in hospital admissions and 40.4% were related to neurology; and steroid treatment was used in 24 visits (24.7%), 7 of which were for worsening of preexisting symptoms, the investigators said.
Of the visits involving new neurologic symptoms, one-third were not from relapse; 59% of the MRIs done in those situations were positive for enhancing or new lesions. Of the visits with worsening preexisting symptoms, only 16.6% were associated with a new relapse, and 28.5% of MRIs done in those situations were positive, the investigators said.
Although many ED visits among MS patients are driven by neurologic complaints, true relapse is rarely present, and not all those with true relapse require hospital admission and steroid treatment, they noted, concluding that developing a care path and triaging system for MS patients in the ED could prevent unnecessary MRIs, steroid treatment, and hospital admissions.
The investigators reported having no disclosures.
NEW ORLEANS – The majority of multiple sclerosis–related emergency department visits in a recent chart review were related to pseudoflares or MS-related complications, rather than to true MS relapse.
The findings suggest that many diagnostic tests, treatments, and hospital admissions are unnecessary, Dr. Hesham Abboud of the Cleveland Clinic and his colleagues reported in a poster at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
Of 97 MS-related visits among 75 patients, 33 were for new neurologic symptoms, 29 were for worsening of preexisting symptoms, and 36 were for MS-related complications. New relapse was diagnosed in only 27 visits (27.8%), and urinary tract infections were found in about one-third of patients presenting with either urinary or neurologic symptoms, the investigators said.
New MRIs were ordered in 37 patients (38.1%); 89 emergency department visits (91.7%) resulted in hospital admissions and 40.4% were related to neurology; and steroid treatment was used in 24 visits (24.7%), 7 of which were for worsening of preexisting symptoms, the investigators said.
Of the visits involving new neurologic symptoms, one-third were not from relapse; 59% of the MRIs done in those situations were positive for enhancing or new lesions. Of the visits with worsening preexisting symptoms, only 16.6% were associated with a new relapse, and 28.5% of MRIs done in those situations were positive, the investigators said.
Although many ED visits among MS patients are driven by neurologic complaints, true relapse is rarely present, and not all those with true relapse require hospital admission and steroid treatment, they noted, concluding that developing a care path and triaging system for MS patients in the ED could prevent unnecessary MRIs, steroid treatment, and hospital admissions.
The investigators reported having no disclosures.
AT ACTRIMS FORUM 2016
Key clinical point: The majority of multiple sclerosis–related emergency department visits in a recent chart review were related to pseudoflares or MS-related complications, rather than true MS relapse.
Major finding: New relapse was diagnosed in only 27 visits (27.8%).
Data source: A retrospective chart review of 75 patients with 97 MS-related ED visits.
Disclosures: The investigators reported having no disclosures.
Risk Score Reliably Predicts Cesarean Delivery
ATLANTA – A risk score derived from five maternal and fetal parameters can help determine the overall risk of cesarean delivery in nulliparous women at term, according to findings from the prospective multicenter Genesis Study.
The score can be used to better inform patients of their individualized cesarean delivery risk in early pregnancy and late pregnancy, facilitating patient decision making about place and mode of delivery, Dr. Naomi Burke of the Royal College of Surgeons in Dublin, Ireland, reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
Of 2,336 nulliparous women with a vertex presentation after 39 weeks’ gestation who were recruited for the Genesis Study, 491 had an unplanned C-section. On multivariate analysis, several factors were found to be associated with risk for cesarean delivery, and the combination of maternal age, maternal height, body mass index, fetal abdominal circumference, and fetal head circumference were found to be the best combined predictors of cesarean delivery, Dr. Burke said.
At the initial visit, the odds ratios for the maternal parameters were 1.22 for maternal age, -1.59 for maternal height, and 1.32 for body mass index. After 39 weeks’ gestation the odds ratios were 1.21, -1.72, and 1.29 for those parameters, respectively. The odds ratios were 1.23 for fetal abdominal circumference and 1.27 for fetal head circumference.
Individual z scores were calculated for demographic and biometric data to determine the odds ratios and a risk score for cesarean delivery. The individual scores were added to give a total risk score, which was then converted into a probability of cesarean delivery, Dr. Burke explained. The prediction model performed well in a bootstrapped cross-validation study, she said.
Actual outcomes “were almost exactly as predicted” in different risk categories. “For example, in a cohort of 614 women who had a 30%-40% risk of cesarean delivery, the actual cesarean delivery rate was 26%,” she said.
Further, of 52 women with a risk score over 50% for cesarean delivery, the actual rate was 56%. Among 23 women who had a vaginal delivery, 15 required an operative vaginal delivery, 5 had an obstetric anal sphincter injury, and 1 had shoulder dystocia with a fractured clavicle.
“So you can see that although these women avoided intrapartum cesarean, there was still significant maternal and neonatal morbidity associated,” she said.
The findings are important because cesarean delivery rates continue to generate concern and prior efforts to predict risk for unplanned cesarean delivery have failed. Despite extensive research and costly interventions, it has been unclear which women would suffer the greatest difficulty in childbirth, she said.
The current findings could change that.
“We can now tell, using our prediction model, which women are at high risk of unplanned cesarean delivery,” she said, noting that a randomized, controlled trial is already underway to assess how the implementation of this model can be used to assess maternal and neonatal morbidity.
The Genesis study is sponsored by Perinatal Ireland. Dr. Burke reported having no disclosures.
ATLANTA – A risk score derived from five maternal and fetal parameters can help determine the overall risk of cesarean delivery in nulliparous women at term, according to findings from the prospective multicenter Genesis Study.
The score can be used to better inform patients of their individualized cesarean delivery risk in early pregnancy and late pregnancy, facilitating patient decision making about place and mode of delivery, Dr. Naomi Burke of the Royal College of Surgeons in Dublin, Ireland, reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
Of 2,336 nulliparous women with a vertex presentation after 39 weeks’ gestation who were recruited for the Genesis Study, 491 had an unplanned C-section. On multivariate analysis, several factors were found to be associated with risk for cesarean delivery, and the combination of maternal age, maternal height, body mass index, fetal abdominal circumference, and fetal head circumference were found to be the best combined predictors of cesarean delivery, Dr. Burke said.
At the initial visit, the odds ratios for the maternal parameters were 1.22 for maternal age, -1.59 for maternal height, and 1.32 for body mass index. After 39 weeks’ gestation the odds ratios were 1.21, -1.72, and 1.29 for those parameters, respectively. The odds ratios were 1.23 for fetal abdominal circumference and 1.27 for fetal head circumference.
Individual z scores were calculated for demographic and biometric data to determine the odds ratios and a risk score for cesarean delivery. The individual scores were added to give a total risk score, which was then converted into a probability of cesarean delivery, Dr. Burke explained. The prediction model performed well in a bootstrapped cross-validation study, she said.
Actual outcomes “were almost exactly as predicted” in different risk categories. “For example, in a cohort of 614 women who had a 30%-40% risk of cesarean delivery, the actual cesarean delivery rate was 26%,” she said.
Further, of 52 women with a risk score over 50% for cesarean delivery, the actual rate was 56%. Among 23 women who had a vaginal delivery, 15 required an operative vaginal delivery, 5 had an obstetric anal sphincter injury, and 1 had shoulder dystocia with a fractured clavicle.
“So you can see that although these women avoided intrapartum cesarean, there was still significant maternal and neonatal morbidity associated,” she said.
The findings are important because cesarean delivery rates continue to generate concern and prior efforts to predict risk for unplanned cesarean delivery have failed. Despite extensive research and costly interventions, it has been unclear which women would suffer the greatest difficulty in childbirth, she said.
The current findings could change that.
“We can now tell, using our prediction model, which women are at high risk of unplanned cesarean delivery,” she said, noting that a randomized, controlled trial is already underway to assess how the implementation of this model can be used to assess maternal and neonatal morbidity.
The Genesis study is sponsored by Perinatal Ireland. Dr. Burke reported having no disclosures.
ATLANTA – A risk score derived from five maternal and fetal parameters can help determine the overall risk of cesarean delivery in nulliparous women at term, according to findings from the prospective multicenter Genesis Study.
The score can be used to better inform patients of their individualized cesarean delivery risk in early pregnancy and late pregnancy, facilitating patient decision making about place and mode of delivery, Dr. Naomi Burke of the Royal College of Surgeons in Dublin, Ireland, reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
Of 2,336 nulliparous women with a vertex presentation after 39 weeks’ gestation who were recruited for the Genesis Study, 491 had an unplanned C-section. On multivariate analysis, several factors were found to be associated with risk for cesarean delivery, and the combination of maternal age, maternal height, body mass index, fetal abdominal circumference, and fetal head circumference were found to be the best combined predictors of cesarean delivery, Dr. Burke said.
At the initial visit, the odds ratios for the maternal parameters were 1.22 for maternal age, -1.59 for maternal height, and 1.32 for body mass index. After 39 weeks’ gestation the odds ratios were 1.21, -1.72, and 1.29 for those parameters, respectively. The odds ratios were 1.23 for fetal abdominal circumference and 1.27 for fetal head circumference.
Individual z scores were calculated for demographic and biometric data to determine the odds ratios and a risk score for cesarean delivery. The individual scores were added to give a total risk score, which was then converted into a probability of cesarean delivery, Dr. Burke explained. The prediction model performed well in a bootstrapped cross-validation study, she said.
Actual outcomes “were almost exactly as predicted” in different risk categories. “For example, in a cohort of 614 women who had a 30%-40% risk of cesarean delivery, the actual cesarean delivery rate was 26%,” she said.
Further, of 52 women with a risk score over 50% for cesarean delivery, the actual rate was 56%. Among 23 women who had a vaginal delivery, 15 required an operative vaginal delivery, 5 had an obstetric anal sphincter injury, and 1 had shoulder dystocia with a fractured clavicle.
“So you can see that although these women avoided intrapartum cesarean, there was still significant maternal and neonatal morbidity associated,” she said.
The findings are important because cesarean delivery rates continue to generate concern and prior efforts to predict risk for unplanned cesarean delivery have failed. Despite extensive research and costly interventions, it has been unclear which women would suffer the greatest difficulty in childbirth, she said.
The current findings could change that.
“We can now tell, using our prediction model, which women are at high risk of unplanned cesarean delivery,” she said, noting that a randomized, controlled trial is already underway to assess how the implementation of this model can be used to assess maternal and neonatal morbidity.
The Genesis study is sponsored by Perinatal Ireland. Dr. Burke reported having no disclosures.
AT THE PREGNANCY MEETING