User login
Necrotizing pancreatitis: Diagnose, treat, consult
Acute pancreatitis accounted for more than 300,000 admissions and $2.6 billion in associated healthcare costs in the United States in 2012.1 First-line management is early aggressive fluid resuscitation and analgesics for pain control. Guidelines recommend estimating the clinical severity of each attack using a validated scoring system such as the Bedside Index of Severity in Acute Pancreatitis.2 Clinically severe pancreatitis is associated with necrosis.
Acute pancreatitis results from inappropriate activation of zymogens and subsequent autodigestion of the pancreas by its own enzymes. Though necrotizing pancreatitis is thought to be an ischemic complication, its pathogenesis is not completely understood. Necrosis increases the morbidity and mortality risk of acute pancreatitis because of its association with organ failure and infectious complications. As such, patients with necrotizing pancreatitis may need admission to the intensive care unit, nutritional support, antibiotics, and radiologic, endoscopic, or surgical interventions.
Here, we review current evidence regarding the diagnosis and management of necrotizing pancreatitis.
PROPER TERMINOLOGY HELPS COLLABORATION
Managing necrotizing pancreatitis requires the combined efforts of internists, gastroenterologists, radiologists, and surgeons. This collaboration is aided by proper terminology.
A classification system was devised in Atlanta, GA, in 1992 to facilitate communication and interdisciplinary collaboration.3 Severe pancreatitis was differentiated from mild by the presence of organ failure or the complications of pseudocyst, necrosis, or abscess.
The original Atlanta classification had several limitations. First, the terminology for fluid collections was ambiguous and frequently misused. Second, the assessment of clinical severity required either the Ranson score or the Acute Physiology and Chronic Health Evaluation II score, both of which are complex and have other limitations. Finally, advances in imaging and treatment have rendered the original Atlanta nomenclature obsolete.
In 2012, the Acute Pancreatitis Classification Working Group issued a revised Atlanta classification that modernized the terminology pertaining to natural history, severity, imaging features, and complications. It divides the natural course of acute pancreatitis into early and late phases.4
Early vs late phase
In the early phase, findings on computed tomography (CT) neither correlate with clinical severity nor alter clinical management.6 Thus, early imaging is not indicated unless there is diagnostic uncertainty, lack of response to appropriate treatment, or sudden deterioration.
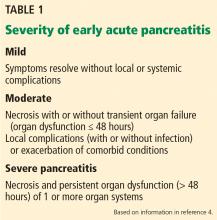
Moderate pancreatitis describes patients with pancreatic necrosis with or without transient organ failure (organ dysfunction for ≤ 48 hours).
Severe pancreatitis is defined by pancreatic necrosis and persistent organ dysfunction.4 It may be accompanied by pancreatic and peripancreatic fluid collections; bacteremia and sepsis can occur in association with infection of necrotic collections.
Interstitial edematous pancreatitis vs necrotizing pancreatitis
The revised Atlanta classification maintains the original classification of acute pancreatitis into 2 main categories: interstitial edematous pancreatitis and necrotizing pancreatitis.
Necrotizing pancreatitis is further divided into 3 subtypes based on extent and location of necrosis:
- Parenchymal necrosis alone (5% of cases)
- Necrosis of peripancreatic fat alone (20%)
- Necrosis of both parenchyma and peripancreatic fat (75%).
Peripancreatic involvement is commonly found in the mesentery, peripancreatic and distant retroperitoneum, and lesser sac.
Of the three subtypes, peripancreatic necrosis has the best prognosis. However, all of the subtypes of necrotizing pancreatitis are associated with poorer outcomes than interstitial edematous pancreatitis.
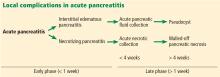
Fluid collections
Acute pancreatic fluid collections contain exclusively nonsolid components without an inflammatory wall and are typically found in the peripancreatic fat. These collections often resolve without intervention as the patient recovers. If they persist beyond 4 weeks and develop a nonepithelialized, fibrous wall, they become pseudocysts. Intervention is generally not recommended for pseudocysts unless they are symptomatic.
ROLE OF IMAGING
Radiographic imaging is not usually necessary to diagnose acute pancreatitis. However, it can be a valuable tool to clarify an ambiguous presentation, determine severity, and identify complications.
The timing and appropriate type of imaging are integral to obtaining useful data. Any imaging obtained in acute pancreatitis to evaluate necrosis should be performed at least 3 to 5 days from the initial symptom onset; if imaging is obtained before 72 hours, necrosis cannot be confidently excluded.8
COMPUTED TOMOGRAPHY
CT is the imaging test of choice when evaluating acute pancreatitis. In addition, almost all percutaneous interventions are performed with CT guidance. The Balthazar score is the most well-known CT severity index. It is calculated based on the degree of inflammation, acute fluid collections, and parenchymal necrosis.9 However, a modified severity index incorporates extrapancreatic complications such as ascites and vascular compromise and was found to more strongly correlate with outcomes than the standard Balthazar score.10
Contrast-enhanced CT is performed in 2 phases:
The pancreatic parenchymal phase
The pancreatic parenchymal or late arterial phase is obtained approximately 40 to 45 seconds after the start of the contrast bolus. It is used to detect necrosis in the early phase of acute pancreatitis and to assess the peripancreatic arteries for pseudoaneurysms in the late phase of acute pancreatitis.11
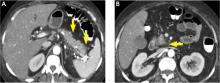
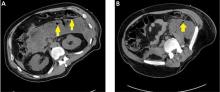
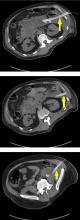
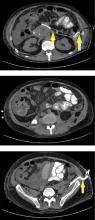
Pancreatic necrosis appears as an area of decreased parenchymal enhancement, either well-defined or heterogeneous. The normal pancreatic parenchyma has a postcontrast enhancement pattern similar to that of the spleen. Parenchyma that does not enhance to the same degree is considered necrotic. The severity of necrosis is graded based on the percentage of the pancreas involved (< 30%, 30%–50%, or > 50%), and a higher percentage correlates with a worse outcome.12,13
Peripancreatic necrosis is harder to detect, as there is no method to assess fat enhancement as there is with pancreatic parenchymal enhancement. In general, radiologists assume that heterogeneous peripancreatic changes, including areas of fat, fluid, and soft tissue attenuation, are consistent with peripancreatic necrosis. After 7 to 10 days, if these changes become more homogeneous and confluent with a more mass-like process, peripancreatic necrosis can be more confidently identified.12,13
The portal venous phase
The later, portal venous phase of the scan is obtained approximately 70 seconds after the start of the contrast bolus. It is used to detect and characterize fluid collections and venous complications of the disease.
Drawbacks of CT
A drawback of CT is the need for iodinated intravenous contrast media, which in severely ill patients may precipitate or worsen pre-existing acute kidney injury.
Further, several studies have shown that findings on CT rarely alter the management of patients in the early phase of acute pancreatitis and in fact may be an overuse of medical resources.14 Unless there are confounding clinical signs or symptoms, CT should be delayed for at least 72 hours.9,10,14,15
MAGNETIC RESONANCE IMAGING
Magnetic resonance imaging (MRI) is not a first-line imaging test in this disease because it is not as available as CT and takes longer to perform—20 to 30 minutes. The patient must be evaluated for candidacy, as it is difficult for acutely ill patients to tolerate an examination that takes this long and requires them to hold their breath multiple times.
MRI is an appropriate alternative in patients who are pregnant or who have severe iodinated-contrast allergy. While contrast is necessary to detect pancreatic necrosis with CT, MRI can detect necrosis without the need for contrast in patients with acute kidney injury or severe chronic kidney disease. Also, MRI may be better in complicated cases requiring repeated imaging because it does not expose the patient to radiation.
On MRI, pancreatic necrosis appears as a heterogeneous area, owing to its liquid and solid components. Liquid components appear hyperintense, and solid components hypointense, on T2 fluid-weighted imaging. This ability to differentiate the components of a walled-off pancreatic necrosis can be useful in determining whether a collection requires drainage or debridement. MRI is also more sensitive for hemorrhagic complications, best seen on T1 fat-weighted images.12,16
Magnetic resonance cholangiopancreatography is an excellent method for ductal evaluation through heavily T2-weighted imaging. It is more sensitive than CT for detecting common bile duct stones and can also detect pancreatic duct strictures or extravasation into fluid collections.16
SUPPORTIVE MANAGEMENT OF EARLY NECROTIZING PANCREATITIS
In the early phase of necrotizing pancreatitis, management is supportive with the primary aim of preventing intravascular volume depletion. Aggressive fluid resuscitation in the first 48 to 72 hours, pain control, and bowel rest are the mainstays of supportive therapy. Intensive care may be necessary if organ failure and hemodynamic instability accompany necrotizing pancreatitis.
Prophylactic antibiotic and antifungal therapy to prevent infected necrosis has been controversial. Recent studies of its utility have not yielded supportive results, and the American College of Gastroenterology and the Infectious Diseases Society of America no longer recommend it.9,17 These medications should not be given unless concomitant cholangitis or extrapancreatic infection is clinically suspected.
Early enteral nutrition is recommended in patients in whom pancreatitis is predicted to be severe and in those not expected to resume oral intake within 5 to 7 days. Enteral nutrition most commonly involves bedside or endoscopic placement of a nasojejunal feeding tube and collaboration with a nutritionist to determine protein-caloric requirements.
Compared with enteral nutrition, total parenteral nutrition is associated with higher rates of infection, multiorgan dysfunction and failure, and death.18
MANAGING COMPLICATIONS OF PANCREATIC NECROSIS
Necrotizing pancreatitis is a defining complication of acute pancreatitis, and its presence alone indicates greater severity. However, superimposed complications may further worsen outcomes.
Infected pancreatic necrosis
Infection occurs in approximately 20% of patients with necrotizing pancreatitis and confers a mortality rate of 20% to 50%.19 Infected pancreatic necrosis occurs when gut organisms translocate into the nearby necrotic pancreatic and peripancreatic tissue. The most commonly identified organisms include Escherichia coli and Enterococcus species.20
This complication usually manifests 2 to 4 weeks after symptom onset; earlier onset is uncommon to rare. It should be considered when the systemic inflammatory response syndrome persists or recurs after 10 days to 2 weeks. Systemic inflammatory response syndrome is also common in sterile necrotizing pancreatitis and sometimes in interstitial pancreatitis, particularly during the first week. However, its sudden appearance or resurgence, high spiking fevers, or worsening organ failure in the later phase (2–4 weeks) of pancreatitis should heighten suspicion of infected pancreatic necrosis.
Imaging may also help diagnose infection, and the presence of gas within a collection or region of necrosis is highly specific. However, the presence of gas is not completely sensitive for infection, as it is seen in only 12% to 22% of infected cases.
Before minimally invasive techniques became available, the diagnosis of infected pancreatic necrosis was confirmed by percutaneous CT-guided aspiration of the necrotic mass or collection for Gram stain and culture.
Antibiotic therapy is indicated in confirmed or suspected cases of infected pancreatic necrosis. Antibiotics with gram-negative coverage and appropriate penetration such as carbapenems, metronidazole, fluoroquinolones, and selected cephalosporins are most commonly used. Meropenem is the antibiotic of choice at our institution.
CT-guided fine-needle aspiration is often done if suspected infected pancreatic necrosis fails to respond to empiric antibiotic therapy.
Debridement or drainage. Generally, the diagnosis or suspicion of infected pancreatic necrosis (suggestive signs are high fever, elevated white blood cell count, and sepsis) warrants an intervention to debride or drain infected pancreatic tissue and control sepsis.21
While source control is integral to the successful treatment of infected pancreatic necrosis, antibiotic therapy may provide a bridge to intervention for critically ill patients by suppressing bacteremia and subsequent sepsis. A 2013 meta-analysis found that 324 of 409 patients with suspected infected pancreatic necrosis were successfully stabilized with antibiotic treatment.21,22 The trend toward conservative management and promising outcomes with antibiotic therapy alone or with minimally invasive techniques has lessened the need for diagnostic CT-guided fine-needle aspiration.
Hemorrhage
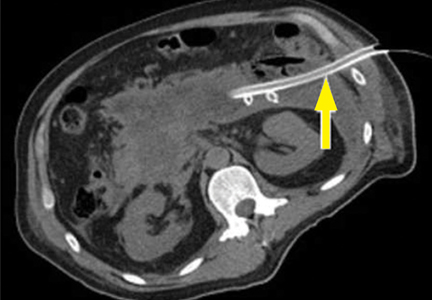
Spontaneous hemorrhage into pancreatic necrosis is a rare but life-threatening complication. Because CT is almost always performed with contrast enhancement, this complication is rarely identified with imaging. The diagnosis is made by noting a drop in hemoglobin and hematocrit.
Hemorrhage into the retroperitoneum or the peritoneal cavity, or both, can occur when an inflammatory process erodes into a nearby artery. Luminal gastrointestinal bleeding can occur from gastric varices arising from splenic vein thrombosis and resulting left-sided portal hypertension, or from pseudoaneurysms. These can also bleed into the pancreatic duct (hemosuccus pancreaticus). Pseudoaneurysm is a later complication that occurs when an arterial wall (most commonly the splenic or gastroduodenal artery) is weakened by pancreatic enzymes.23
Prompt recognition of hemorrhagic events and consultation with an interventional radiologist or surgeon are required to prevent death.
Inflammation and abdominal compartment syndrome
Inflammation from necrotizing pancreatitis can cause further complications by blocking nearby structures. Reported complications include jaundice from biliary compression, hydronephrosis from ureteral compression, bowel obstruction, and gastric outlet obstruction.
Abdominal compartment syndrome is an increasingly recognized complication of acute pancreatitis. Abdominal pressure can rise due to a number of factors, including fluid collections, ascites, ileus, and overly aggressive fluid resuscitation.24 Elevated abdominal pressure is associated with complications such as decreased respiratory compliance, increased peak airway pressure, decreased cardiac preload, hypotension, mesenteric and intestinal ischemia, feeding intolerance, and lower-extremity ischemia and thrombosis.
Patients with necrotizing pancreatitis who have abdominal compartment syndrome have a mortality rate 5 times higher than patients without abdominal compartment syndrome.25
Abdominal pressures should be monitored using a bladder pressure sensor in critically ill or ventilated patients with acute pancreatitis. If the abdominal pressure rises above 20 mm Hg, medical and surgical interventions should be offered in a stepwise fashion to decrease it. Interventions include decompression by nasogastric and rectal tube, sedation or paralysis to relax abdominal wall tension, minimization of intravenous fluids, percutaneous drainage of ascites, and (rarely) surgical midline or subcostal laparotomy.
ROLE OF INTERVENTION
The treatment of necrotizing pancreatitis has changed rapidly, thanks to a growing experience with minimally invasive techniques.
Indications for intervention
Infected pancreatic necrosis is the primary indication for surgical, percutaneous, or endoscopic intervention.
In sterile necrosis, the threshold for intervention is less clear, and intervention is often reserved for patients who fail to clinically improve or who have intractable abdominal pain, gastric outlet obstruction, or fistulating disease.26
In asymptomatic cases, intervention is almost never indicated regardless of the location or size of the necrotic area.
In walled-off pancreatic necrosis, less-invasive and less-morbid interventions such as endoscopic or percutaneous drainage or video-assisted retroperitoneal debridement can be done.
Timing of intervention
In the past, delaying intervention was thought to increase the risk of death. However, multiple studies have found that outcomes are often worse if intervention is done early, likely due to the lack of a fully formed fibrous wall or demarcation of the necrotic area.27
If the patient remains clinically stable, it is best to delay intervention until at least 4 weeks after the index event to achieve optimal outcomes. Delay can often be achieved by antibiotic treatment to suppress bacteremia and endoscopic or percutaneous drainage of infected collections to control sepsis.
Open surgery
The gold-standard intervention for infected pancreatic necrosis or symptomatic sterile walled-off pancreatic necrosis is open necrosectomy. This involves exploratory laparotomy with blunt debridement of all visible necrotic pancreatic tissue.
Methods to facilitate later evacuation of residual infected fluid and debris vary widely. Multiple large-caliber drains can be placed to facilitate irrigation and drainage before closure of the abdominal fascia. As infected pancreatic necrosis carries the risk of contaminating the peritoneal cavity, the skin is often left open to heal by secondary intention. An interventional radiologist is frequently enlisted to place, exchange, or downsize drainage catheters.
Infected pancreatic necrosis or symptomatic sterile walled-off pancreatic necrosis often requires more than one operation to achieve satisfactory debridement.
The goals of open necrosectomy are to remove nonviable tissue and infection, preserve viable pancreatic tissue, eliminate fistulous connections, and minimize damage to local organs and vasculature.
Minimally invasive techniques
Video-assisted retroperitoneal debridement has been described as a hybrid between endoscopic and open retroperitoneal debridement.28 This technique requires first placing a percutaneous catheter into the necrotic area through the left flank to create a retroperitoneal tract. A 5-cm incision is made and the necrotic space is entered using the drain for guidance. Necrotic tissue is carefully debrided under direct vision using a combination of forceps, irrigation, and suction. A laparoscopic port can also be introduced into the incision when the procedure can no longer be continued under direct vision.29,30
Although not all patients are candidates for minimal-access surgery, it remains an evolving surgical option.
Endoscopic transmural debridement is another option for infected pancreatic necrosis and symptomatic walled-off pancreatic necrosis. Depending on the location of the necrotic area, an echoendoscope is passed to either the stomach or duodenum. Guided by endoscopic ultrasonography, a needle is passed into the collection, allowing subsequent fistula creation and stenting for internal drainage or debridement. In the past, this process required several steps, multiple devices, fluoroscopic guidance, and considerable time. But newer endoscopic lumen-apposing metal stents have been developed that can be placed in a single step without fluoroscopy. A slimmer endoscope can then be introduced into the necrotic cavity via the stent, and the necrotic debris can be debrided with endoscopic baskets, snares, forceps, and irrigation.9,31
Similar to surgical necrosectomy, satisfactory debridement is not often obtained with a single procedure; 2 to 5 endoscopic procedures may be needed to achieve resolution. However, the luminal approach in endoscopic necrosectomy avoids the significant morbidity of major abdominal surgery and the potential for pancreaticocutaneous fistulae that may occur with drains.
In a randomized trial comparing endoscopic necrosectomy vs surgical necrosectomy (video-assisted retroperitoneal debridement and exploratory laparotomy),32 endoscopic necrosectomy showed less inflammatory response than surgical necrosectomy and had a lower risk of new-onset organ failure, bleeding, fistula formation, and death.32
Selecting the best intervention for the individual patient
Given the multiple available techniques, selecting the best intervention for individual patients can be challenging. A team approach with input from a gastroenterologist, surgeon, and interventional radiologist is best when determining which technique would best suit each patient.
Surgical necrosectomy is still the treatment of choice for unstable patients with infected pancreatic necrosis or multiple, inaccessible collections, but current evidence suggests a different approach in stable infected pancreatic necrosis and symptomatic sterile walled-off pancreatic necrosis.
The Dutch Pancreatitis Group28 randomized 88 patients with infected pancreatic necrosis or symptomatic walled-off pancreatic necrosis to open necrosectomy or a minimally invasive “step-up” approach consisting of up to 2 percutaneous drainage or endoscopic debridement procedures before escalation to video-assisted retroperitoneal debridement. The step-up approach resulted in lower rates of morbidity and death than surgical necrosectomy as first-line treatment. Furthermore, some patients in the step-up group avoided the need for surgery entirely.30
SUMMING UP
Necrosis significantly increases rates of morbidity and mortality in acute pancreatitis. Hospitalists, general internists, and general surgeons are all on the front lines in identifying severe cases and consulting the appropriate specialists for optimal multidisciplinary care. Selective and appropriate timing of radiologic imaging is key, and a vital tool in the management of necrotizing pancreatitis.
While the primary indication for intervention is infected pancreatic necrosis, additional indications are symptomatic walled-off pancreatic necrosis secondary to intractable abdominal pain, bowel obstruction, and failure to thrive. As a result of improving technology and inpatient care, these patients may present with intractable symptoms in the outpatient setting rather than the inpatient setting. The onus is on the primary care physician to maintain a high level of suspicion and refer these patients to subspecialists as appropriate.
Open surgical necrosectomy remains an important approach for care of infected pancreatic necrosis or patients with intractable symptoms. A step-up approach starting with a minimally invasive procedure and escalating if the initial intervention is unsuccessful is gradually becoming the standard of care.
- Peery AF, Crockett SD, Barritt AS, et al. Burden of gastrointestinal, liver, and pancreatic disease in the United States. Gastroenterology 2015; 149:1731–1741e3.
- Tenner S, Baillie J, DeWitt J, Vege SS; American College of Gastroenterology. American College of Gastroenterology guideline: management of acute pancreatitis. Am J Gastroenterol 2013; 108:1400–1416.
- Bradley EL 3rd. A clinically based classification system for acute pancreatitis. Summary of the International Symposium on Acute Pancreatitis, Atlanta, GA, September 11 through 13, 1992. Arch Surg 1993; 128:586–590.
- Banks PA, Bollen TL, Dervenis C, et al; Acute Pancreatitis Classification Working Group. Classification of acute pancreatitis—2012: revision of the Atlanta classification and definitions by international consensus. Gut 2013; 62:102–111.
- Marshall JC, Cook DJ, Christou NV, Bernard GR, Sprung CL, Sibbald WJ. Multiple organ dysfunction score: a reliable descriptor of a complex clinical outcome. Crit Care Med 1995; 23:1638–1652.
- Kadiyala V, Suleiman SL, McNabb-Baltar J, Wu BU, Banks PA, Singh VK. The Atlanta classification, revised Atlanta classification, and determinant-based classification of acute pancreatitis: which is best at stratifying outcomes? Pancreas 2016; 45:510–515.
- Singh VK, Bollen TL, Wu BU, et al. An assessment of the severity of interstitial pancreatitis. Clin Gastroenterol Hepatol 2011; 9:1098–1103.
- Kotwal V, Talukdar R, Levy M, Vege SS. Role of endoscopic ultrasound during hospitalization for acute pancreatitis. World J Gastroenterol 2010; 16:4888–4891.
- Balthazar EJ. Acute pancreatitis: assessment of severity with clinical and CT evaluation. Radiology 2002; 223:603–613.
- Mortele KJ, Wiesner W, Intriere L, et al. A modified CT severity index for evaluating acute pancreatitis: improved correlation with patient outcome. AJR Am J Roentgenol 2004; 183:1261–1265.
- Verde F, Fishman EK, Johnson PT. Arterial pseudoaneurysms complicating pancreatitis: literature review. J Comput Assist Tomogr 2015; 39:7–12.
- Shyu JY, Sainani NI, Sahni VA, et al. Necrotizing pancreatitis: diagnosis, imaging, and intervention. Radiographics 2014; 34:1218–1239.
- Thoeni RF. The revised Atlanta classification of acute pancreatitis: its importance for the radiologist and its effect on treatment. Radiology 2012; 262:751–764.
- Morgan DE, Ragheb CM, Lockhart ME, Cary B, Fineberg NS, Berland LL. Acute pancreatitis: computed tomography utilization and radiation exposure are related to severity but not patient age. Clin Gastroenterol Hepatol 2010; 8:303–308.
- Vitellas KM, Paulson EK, Enns RA, Keogan MT, Pappas TN. Pancreatitis complicated by gland necrosis: evolution of findings on contrast-enhanced CT. J Comput Assist Tomogr 1999; 23:898–905.
- Stimac D, Miletic D, Radic M, et al. The role of nonenhanced magnetic resonance imaging in the early assessment of acute pancreatitis. Am J Gastroenterol 2007; 102:997–1004.
- Solomkin JS, Mazuski JE, Bradley JS, et al. Diagnosis and management of complicated intra-abdominal infection in adults and children: guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Surg Infect (Larchmt) 2010; 11:79–109.
- Petrov MS, Kukosh MV, Emelyanov NV. A randomized controlled trial of enteral versus parenteral feeding in patients with predicted severe acute pancreatitis shows a significant reduction in mortality and in infected pancreatic complications with total enteral nutrition. Dig Surg 2006; 23:336–345.
- Petrov MS, Shanbhag S, Chakraborty M, Phillips AR, Windsor JA. Organ failure and infection of pancreatic necrosis as determinants of mortality in patients with acute pancreatitis. Gastroenterology 2010; 139:813–820.
- Villatoro E, Bassi C, Larvin M. Antibiotic therapy for prophylaxis against infection of pancreatic necrosis in acute pancreatitis. Cochrane Database Syst Rev 2006; 4:CD002941.
- Baril NB, Ralls PW, Wren SM, et al. Does an infected peripancreatic fluid collection or abscess mandate operation? Ann Surg 2000; 231:361–367.
- Mouli VP, Sreenivas V, Garg PK. Efficacy of conservative treatment, without necrosectomy, for infected pancreatic necrosis: a systematic review and meta-analysis. Gastroenterology 2013; 144:333–340.e2.
- Kirby JM, Vora P, Midia M, Rawlinson J. Vascular complications of pancreatitis: imaging and intervention. Cardiovasc Intervent Radiol 2008; 31:957–970.
- De Waele JJ, Hoste E, Blot SI, Decruyenaere J, Colardyn F. Intra-abdominal hypertension in patients with severe acute pancreatitis. Crit Care 2005; 9:R452–R457.
- van Brunschot S, Schut AJ, Bouwense SA, et al; Dutch Pancreatitis Study Group. Abdominal compartment syndrome in acute pancreatitis: a systematic review. Pancreas 2014; 43:665–674.
- Bugiantella W, Rondelli F, Boni M, et al. Necrotizing pancreatitis: a review of the interventions. Int J Surg 2016; 28(suppl 1):S163–S171.
- Besselink MG, Verwer TJ, Schoenmaeckers EJ, et al. Timing of surgical intervention in necrotizing pancreatitis. Arch Surg 2007; 142:1194–1201.
- van Santvoort HC, Besselink MG, Horvath KD, et al; Dutch Acute Pancreatis Study Group. Videoscopic assisted retroperitoneal debridement in infected necrotizing pancreatitis. HPB (Oxford) 2007; 9:156–159.
- van Santvoort HC, Besselink MG, Bollen TL, Buskens E, van Ramshorst B, Gooszen HG; Dutch Acute Pancreatitis Study Group. Case-matched comparison of the retroperitoneal approach with laparotomy for necrotizing pancreatitis. World J Surg 2007; 31:1635–1642.
- van Santvoort HC, Besselink MG, Bakker OJ, et al; Dutch Pancreatitis Study Group. A step-up approach or open necrosectomy for necrotizing pancreatitis. N Engl J Med 2010; 362:1491–1502.
- Thompson CC, Kumar N, Slattery J, et al. A standardized method for endoscopic necrosectomy improves complication and mortality rates. Pancreatology 2016; 16:66–72.
- Bakker OJ, van Santvoort HC, van Brunschot S, et al; Dutch Pancreatitis Study Group. Endoscopic transgastric vs surgical necrosectomy for infected necrotizing pancreatitis: a randomized trial. JAMA 2012; 307:1053–1061.
Acute pancreatitis accounted for more than 300,000 admissions and $2.6 billion in associated healthcare costs in the United States in 2012.1 First-line management is early aggressive fluid resuscitation and analgesics for pain control. Guidelines recommend estimating the clinical severity of each attack using a validated scoring system such as the Bedside Index of Severity in Acute Pancreatitis.2 Clinically severe pancreatitis is associated with necrosis.
Acute pancreatitis results from inappropriate activation of zymogens and subsequent autodigestion of the pancreas by its own enzymes. Though necrotizing pancreatitis is thought to be an ischemic complication, its pathogenesis is not completely understood. Necrosis increases the morbidity and mortality risk of acute pancreatitis because of its association with organ failure and infectious complications. As such, patients with necrotizing pancreatitis may need admission to the intensive care unit, nutritional support, antibiotics, and radiologic, endoscopic, or surgical interventions.
Here, we review current evidence regarding the diagnosis and management of necrotizing pancreatitis.
PROPER TERMINOLOGY HELPS COLLABORATION
Managing necrotizing pancreatitis requires the combined efforts of internists, gastroenterologists, radiologists, and surgeons. This collaboration is aided by proper terminology.
A classification system was devised in Atlanta, GA, in 1992 to facilitate communication and interdisciplinary collaboration.3 Severe pancreatitis was differentiated from mild by the presence of organ failure or the complications of pseudocyst, necrosis, or abscess.
The original Atlanta classification had several limitations. First, the terminology for fluid collections was ambiguous and frequently misused. Second, the assessment of clinical severity required either the Ranson score or the Acute Physiology and Chronic Health Evaluation II score, both of which are complex and have other limitations. Finally, advances in imaging and treatment have rendered the original Atlanta nomenclature obsolete.
In 2012, the Acute Pancreatitis Classification Working Group issued a revised Atlanta classification that modernized the terminology pertaining to natural history, severity, imaging features, and complications. It divides the natural course of acute pancreatitis into early and late phases.4
Early vs late phase
In the early phase, findings on computed tomography (CT) neither correlate with clinical severity nor alter clinical management.6 Thus, early imaging is not indicated unless there is diagnostic uncertainty, lack of response to appropriate treatment, or sudden deterioration.
Moderate pancreatitis describes patients with pancreatic necrosis with or without transient organ failure (organ dysfunction for ≤ 48 hours).
Severe pancreatitis is defined by pancreatic necrosis and persistent organ dysfunction.4 It may be accompanied by pancreatic and peripancreatic fluid collections; bacteremia and sepsis can occur in association with infection of necrotic collections.
Interstitial edematous pancreatitis vs necrotizing pancreatitis
The revised Atlanta classification maintains the original classification of acute pancreatitis into 2 main categories: interstitial edematous pancreatitis and necrotizing pancreatitis.
Necrotizing pancreatitis is further divided into 3 subtypes based on extent and location of necrosis:
- Parenchymal necrosis alone (5% of cases)
- Necrosis of peripancreatic fat alone (20%)
- Necrosis of both parenchyma and peripancreatic fat (75%).
Peripancreatic involvement is commonly found in the mesentery, peripancreatic and distant retroperitoneum, and lesser sac.
Of the three subtypes, peripancreatic necrosis has the best prognosis. However, all of the subtypes of necrotizing pancreatitis are associated with poorer outcomes than interstitial edematous pancreatitis.
Fluid collections
Acute pancreatic fluid collections contain exclusively nonsolid components without an inflammatory wall and are typically found in the peripancreatic fat. These collections often resolve without intervention as the patient recovers. If they persist beyond 4 weeks and develop a nonepithelialized, fibrous wall, they become pseudocysts. Intervention is generally not recommended for pseudocysts unless they are symptomatic.
ROLE OF IMAGING
Radiographic imaging is not usually necessary to diagnose acute pancreatitis. However, it can be a valuable tool to clarify an ambiguous presentation, determine severity, and identify complications.
The timing and appropriate type of imaging are integral to obtaining useful data. Any imaging obtained in acute pancreatitis to evaluate necrosis should be performed at least 3 to 5 days from the initial symptom onset; if imaging is obtained before 72 hours, necrosis cannot be confidently excluded.8
COMPUTED TOMOGRAPHY
CT is the imaging test of choice when evaluating acute pancreatitis. In addition, almost all percutaneous interventions are performed with CT guidance. The Balthazar score is the most well-known CT severity index. It is calculated based on the degree of inflammation, acute fluid collections, and parenchymal necrosis.9 However, a modified severity index incorporates extrapancreatic complications such as ascites and vascular compromise and was found to more strongly correlate with outcomes than the standard Balthazar score.10
Contrast-enhanced CT is performed in 2 phases:
The pancreatic parenchymal phase
The pancreatic parenchymal or late arterial phase is obtained approximately 40 to 45 seconds after the start of the contrast bolus. It is used to detect necrosis in the early phase of acute pancreatitis and to assess the peripancreatic arteries for pseudoaneurysms in the late phase of acute pancreatitis.11
Pancreatic necrosis appears as an area of decreased parenchymal enhancement, either well-defined or heterogeneous. The normal pancreatic parenchyma has a postcontrast enhancement pattern similar to that of the spleen. Parenchyma that does not enhance to the same degree is considered necrotic. The severity of necrosis is graded based on the percentage of the pancreas involved (< 30%, 30%–50%, or > 50%), and a higher percentage correlates with a worse outcome.12,13
Peripancreatic necrosis is harder to detect, as there is no method to assess fat enhancement as there is with pancreatic parenchymal enhancement. In general, radiologists assume that heterogeneous peripancreatic changes, including areas of fat, fluid, and soft tissue attenuation, are consistent with peripancreatic necrosis. After 7 to 10 days, if these changes become more homogeneous and confluent with a more mass-like process, peripancreatic necrosis can be more confidently identified.12,13
The portal venous phase
The later, portal venous phase of the scan is obtained approximately 70 seconds after the start of the contrast bolus. It is used to detect and characterize fluid collections and venous complications of the disease.
Drawbacks of CT
A drawback of CT is the need for iodinated intravenous contrast media, which in severely ill patients may precipitate or worsen pre-existing acute kidney injury.
Further, several studies have shown that findings on CT rarely alter the management of patients in the early phase of acute pancreatitis and in fact may be an overuse of medical resources.14 Unless there are confounding clinical signs or symptoms, CT should be delayed for at least 72 hours.9,10,14,15
MAGNETIC RESONANCE IMAGING
Magnetic resonance imaging (MRI) is not a first-line imaging test in this disease because it is not as available as CT and takes longer to perform—20 to 30 minutes. The patient must be evaluated for candidacy, as it is difficult for acutely ill patients to tolerate an examination that takes this long and requires them to hold their breath multiple times.
MRI is an appropriate alternative in patients who are pregnant or who have severe iodinated-contrast allergy. While contrast is necessary to detect pancreatic necrosis with CT, MRI can detect necrosis without the need for contrast in patients with acute kidney injury or severe chronic kidney disease. Also, MRI may be better in complicated cases requiring repeated imaging because it does not expose the patient to radiation.
On MRI, pancreatic necrosis appears as a heterogeneous area, owing to its liquid and solid components. Liquid components appear hyperintense, and solid components hypointense, on T2 fluid-weighted imaging. This ability to differentiate the components of a walled-off pancreatic necrosis can be useful in determining whether a collection requires drainage or debridement. MRI is also more sensitive for hemorrhagic complications, best seen on T1 fat-weighted images.12,16
Magnetic resonance cholangiopancreatography is an excellent method for ductal evaluation through heavily T2-weighted imaging. It is more sensitive than CT for detecting common bile duct stones and can also detect pancreatic duct strictures or extravasation into fluid collections.16
SUPPORTIVE MANAGEMENT OF EARLY NECROTIZING PANCREATITIS
In the early phase of necrotizing pancreatitis, management is supportive with the primary aim of preventing intravascular volume depletion. Aggressive fluid resuscitation in the first 48 to 72 hours, pain control, and bowel rest are the mainstays of supportive therapy. Intensive care may be necessary if organ failure and hemodynamic instability accompany necrotizing pancreatitis.
Prophylactic antibiotic and antifungal therapy to prevent infected necrosis has been controversial. Recent studies of its utility have not yielded supportive results, and the American College of Gastroenterology and the Infectious Diseases Society of America no longer recommend it.9,17 These medications should not be given unless concomitant cholangitis or extrapancreatic infection is clinically suspected.
Early enteral nutrition is recommended in patients in whom pancreatitis is predicted to be severe and in those not expected to resume oral intake within 5 to 7 days. Enteral nutrition most commonly involves bedside or endoscopic placement of a nasojejunal feeding tube and collaboration with a nutritionist to determine protein-caloric requirements.
Compared with enteral nutrition, total parenteral nutrition is associated with higher rates of infection, multiorgan dysfunction and failure, and death.18
MANAGING COMPLICATIONS OF PANCREATIC NECROSIS
Necrotizing pancreatitis is a defining complication of acute pancreatitis, and its presence alone indicates greater severity. However, superimposed complications may further worsen outcomes.
Infected pancreatic necrosis
Infection occurs in approximately 20% of patients with necrotizing pancreatitis and confers a mortality rate of 20% to 50%.19 Infected pancreatic necrosis occurs when gut organisms translocate into the nearby necrotic pancreatic and peripancreatic tissue. The most commonly identified organisms include Escherichia coli and Enterococcus species.20
This complication usually manifests 2 to 4 weeks after symptom onset; earlier onset is uncommon to rare. It should be considered when the systemic inflammatory response syndrome persists or recurs after 10 days to 2 weeks. Systemic inflammatory response syndrome is also common in sterile necrotizing pancreatitis and sometimes in interstitial pancreatitis, particularly during the first week. However, its sudden appearance or resurgence, high spiking fevers, or worsening organ failure in the later phase (2–4 weeks) of pancreatitis should heighten suspicion of infected pancreatic necrosis.
Imaging may also help diagnose infection, and the presence of gas within a collection or region of necrosis is highly specific. However, the presence of gas is not completely sensitive for infection, as it is seen in only 12% to 22% of infected cases.
Before minimally invasive techniques became available, the diagnosis of infected pancreatic necrosis was confirmed by percutaneous CT-guided aspiration of the necrotic mass or collection for Gram stain and culture.
Antibiotic therapy is indicated in confirmed or suspected cases of infected pancreatic necrosis. Antibiotics with gram-negative coverage and appropriate penetration such as carbapenems, metronidazole, fluoroquinolones, and selected cephalosporins are most commonly used. Meropenem is the antibiotic of choice at our institution.
CT-guided fine-needle aspiration is often done if suspected infected pancreatic necrosis fails to respond to empiric antibiotic therapy.
Debridement or drainage. Generally, the diagnosis or suspicion of infected pancreatic necrosis (suggestive signs are high fever, elevated white blood cell count, and sepsis) warrants an intervention to debride or drain infected pancreatic tissue and control sepsis.21
While source control is integral to the successful treatment of infected pancreatic necrosis, antibiotic therapy may provide a bridge to intervention for critically ill patients by suppressing bacteremia and subsequent sepsis. A 2013 meta-analysis found that 324 of 409 patients with suspected infected pancreatic necrosis were successfully stabilized with antibiotic treatment.21,22 The trend toward conservative management and promising outcomes with antibiotic therapy alone or with minimally invasive techniques has lessened the need for diagnostic CT-guided fine-needle aspiration.
Hemorrhage
Spontaneous hemorrhage into pancreatic necrosis is a rare but life-threatening complication. Because CT is almost always performed with contrast enhancement, this complication is rarely identified with imaging. The diagnosis is made by noting a drop in hemoglobin and hematocrit.
Hemorrhage into the retroperitoneum or the peritoneal cavity, or both, can occur when an inflammatory process erodes into a nearby artery. Luminal gastrointestinal bleeding can occur from gastric varices arising from splenic vein thrombosis and resulting left-sided portal hypertension, or from pseudoaneurysms. These can also bleed into the pancreatic duct (hemosuccus pancreaticus). Pseudoaneurysm is a later complication that occurs when an arterial wall (most commonly the splenic or gastroduodenal artery) is weakened by pancreatic enzymes.23
Prompt recognition of hemorrhagic events and consultation with an interventional radiologist or surgeon are required to prevent death.
Inflammation and abdominal compartment syndrome
Inflammation from necrotizing pancreatitis can cause further complications by blocking nearby structures. Reported complications include jaundice from biliary compression, hydronephrosis from ureteral compression, bowel obstruction, and gastric outlet obstruction.
Abdominal compartment syndrome is an increasingly recognized complication of acute pancreatitis. Abdominal pressure can rise due to a number of factors, including fluid collections, ascites, ileus, and overly aggressive fluid resuscitation.24 Elevated abdominal pressure is associated with complications such as decreased respiratory compliance, increased peak airway pressure, decreased cardiac preload, hypotension, mesenteric and intestinal ischemia, feeding intolerance, and lower-extremity ischemia and thrombosis.
Patients with necrotizing pancreatitis who have abdominal compartment syndrome have a mortality rate 5 times higher than patients without abdominal compartment syndrome.25
Abdominal pressures should be monitored using a bladder pressure sensor in critically ill or ventilated patients with acute pancreatitis. If the abdominal pressure rises above 20 mm Hg, medical and surgical interventions should be offered in a stepwise fashion to decrease it. Interventions include decompression by nasogastric and rectal tube, sedation or paralysis to relax abdominal wall tension, minimization of intravenous fluids, percutaneous drainage of ascites, and (rarely) surgical midline or subcostal laparotomy.
ROLE OF INTERVENTION
The treatment of necrotizing pancreatitis has changed rapidly, thanks to a growing experience with minimally invasive techniques.
Indications for intervention
Infected pancreatic necrosis is the primary indication for surgical, percutaneous, or endoscopic intervention.
In sterile necrosis, the threshold for intervention is less clear, and intervention is often reserved for patients who fail to clinically improve or who have intractable abdominal pain, gastric outlet obstruction, or fistulating disease.26
In asymptomatic cases, intervention is almost never indicated regardless of the location or size of the necrotic area.
In walled-off pancreatic necrosis, less-invasive and less-morbid interventions such as endoscopic or percutaneous drainage or video-assisted retroperitoneal debridement can be done.
Timing of intervention
In the past, delaying intervention was thought to increase the risk of death. However, multiple studies have found that outcomes are often worse if intervention is done early, likely due to the lack of a fully formed fibrous wall or demarcation of the necrotic area.27
If the patient remains clinically stable, it is best to delay intervention until at least 4 weeks after the index event to achieve optimal outcomes. Delay can often be achieved by antibiotic treatment to suppress bacteremia and endoscopic or percutaneous drainage of infected collections to control sepsis.
Open surgery
The gold-standard intervention for infected pancreatic necrosis or symptomatic sterile walled-off pancreatic necrosis is open necrosectomy. This involves exploratory laparotomy with blunt debridement of all visible necrotic pancreatic tissue.
Methods to facilitate later evacuation of residual infected fluid and debris vary widely. Multiple large-caliber drains can be placed to facilitate irrigation and drainage before closure of the abdominal fascia. As infected pancreatic necrosis carries the risk of contaminating the peritoneal cavity, the skin is often left open to heal by secondary intention. An interventional radiologist is frequently enlisted to place, exchange, or downsize drainage catheters.
Infected pancreatic necrosis or symptomatic sterile walled-off pancreatic necrosis often requires more than one operation to achieve satisfactory debridement.
The goals of open necrosectomy are to remove nonviable tissue and infection, preserve viable pancreatic tissue, eliminate fistulous connections, and minimize damage to local organs and vasculature.
Minimally invasive techniques
Video-assisted retroperitoneal debridement has been described as a hybrid between endoscopic and open retroperitoneal debridement.28 This technique requires first placing a percutaneous catheter into the necrotic area through the left flank to create a retroperitoneal tract. A 5-cm incision is made and the necrotic space is entered using the drain for guidance. Necrotic tissue is carefully debrided under direct vision using a combination of forceps, irrigation, and suction. A laparoscopic port can also be introduced into the incision when the procedure can no longer be continued under direct vision.29,30
Although not all patients are candidates for minimal-access surgery, it remains an evolving surgical option.
Endoscopic transmural debridement is another option for infected pancreatic necrosis and symptomatic walled-off pancreatic necrosis. Depending on the location of the necrotic area, an echoendoscope is passed to either the stomach or duodenum. Guided by endoscopic ultrasonography, a needle is passed into the collection, allowing subsequent fistula creation and stenting for internal drainage or debridement. In the past, this process required several steps, multiple devices, fluoroscopic guidance, and considerable time. But newer endoscopic lumen-apposing metal stents have been developed that can be placed in a single step without fluoroscopy. A slimmer endoscope can then be introduced into the necrotic cavity via the stent, and the necrotic debris can be debrided with endoscopic baskets, snares, forceps, and irrigation.9,31
Similar to surgical necrosectomy, satisfactory debridement is not often obtained with a single procedure; 2 to 5 endoscopic procedures may be needed to achieve resolution. However, the luminal approach in endoscopic necrosectomy avoids the significant morbidity of major abdominal surgery and the potential for pancreaticocutaneous fistulae that may occur with drains.
In a randomized trial comparing endoscopic necrosectomy vs surgical necrosectomy (video-assisted retroperitoneal debridement and exploratory laparotomy),32 endoscopic necrosectomy showed less inflammatory response than surgical necrosectomy and had a lower risk of new-onset organ failure, bleeding, fistula formation, and death.32
Selecting the best intervention for the individual patient
Given the multiple available techniques, selecting the best intervention for individual patients can be challenging. A team approach with input from a gastroenterologist, surgeon, and interventional radiologist is best when determining which technique would best suit each patient.
Surgical necrosectomy is still the treatment of choice for unstable patients with infected pancreatic necrosis or multiple, inaccessible collections, but current evidence suggests a different approach in stable infected pancreatic necrosis and symptomatic sterile walled-off pancreatic necrosis.
The Dutch Pancreatitis Group28 randomized 88 patients with infected pancreatic necrosis or symptomatic walled-off pancreatic necrosis to open necrosectomy or a minimally invasive “step-up” approach consisting of up to 2 percutaneous drainage or endoscopic debridement procedures before escalation to video-assisted retroperitoneal debridement. The step-up approach resulted in lower rates of morbidity and death than surgical necrosectomy as first-line treatment. Furthermore, some patients in the step-up group avoided the need for surgery entirely.30
SUMMING UP
Necrosis significantly increases rates of morbidity and mortality in acute pancreatitis. Hospitalists, general internists, and general surgeons are all on the front lines in identifying severe cases and consulting the appropriate specialists for optimal multidisciplinary care. Selective and appropriate timing of radiologic imaging is key, and a vital tool in the management of necrotizing pancreatitis.
While the primary indication for intervention is infected pancreatic necrosis, additional indications are symptomatic walled-off pancreatic necrosis secondary to intractable abdominal pain, bowel obstruction, and failure to thrive. As a result of improving technology and inpatient care, these patients may present with intractable symptoms in the outpatient setting rather than the inpatient setting. The onus is on the primary care physician to maintain a high level of suspicion and refer these patients to subspecialists as appropriate.
Open surgical necrosectomy remains an important approach for care of infected pancreatic necrosis or patients with intractable symptoms. A step-up approach starting with a minimally invasive procedure and escalating if the initial intervention is unsuccessful is gradually becoming the standard of care.
Acute pancreatitis accounted for more than 300,000 admissions and $2.6 billion in associated healthcare costs in the United States in 2012.1 First-line management is early aggressive fluid resuscitation and analgesics for pain control. Guidelines recommend estimating the clinical severity of each attack using a validated scoring system such as the Bedside Index of Severity in Acute Pancreatitis.2 Clinically severe pancreatitis is associated with necrosis.
Acute pancreatitis results from inappropriate activation of zymogens and subsequent autodigestion of the pancreas by its own enzymes. Though necrotizing pancreatitis is thought to be an ischemic complication, its pathogenesis is not completely understood. Necrosis increases the morbidity and mortality risk of acute pancreatitis because of its association with organ failure and infectious complications. As such, patients with necrotizing pancreatitis may need admission to the intensive care unit, nutritional support, antibiotics, and radiologic, endoscopic, or surgical interventions.
Here, we review current evidence regarding the diagnosis and management of necrotizing pancreatitis.
PROPER TERMINOLOGY HELPS COLLABORATION
Managing necrotizing pancreatitis requires the combined efforts of internists, gastroenterologists, radiologists, and surgeons. This collaboration is aided by proper terminology.
A classification system was devised in Atlanta, GA, in 1992 to facilitate communication and interdisciplinary collaboration.3 Severe pancreatitis was differentiated from mild by the presence of organ failure or the complications of pseudocyst, necrosis, or abscess.
The original Atlanta classification had several limitations. First, the terminology for fluid collections was ambiguous and frequently misused. Second, the assessment of clinical severity required either the Ranson score or the Acute Physiology and Chronic Health Evaluation II score, both of which are complex and have other limitations. Finally, advances in imaging and treatment have rendered the original Atlanta nomenclature obsolete.
In 2012, the Acute Pancreatitis Classification Working Group issued a revised Atlanta classification that modernized the terminology pertaining to natural history, severity, imaging features, and complications. It divides the natural course of acute pancreatitis into early and late phases.4
Early vs late phase
In the early phase, findings on computed tomography (CT) neither correlate with clinical severity nor alter clinical management.6 Thus, early imaging is not indicated unless there is diagnostic uncertainty, lack of response to appropriate treatment, or sudden deterioration.
Moderate pancreatitis describes patients with pancreatic necrosis with or without transient organ failure (organ dysfunction for ≤ 48 hours).
Severe pancreatitis is defined by pancreatic necrosis and persistent organ dysfunction.4 It may be accompanied by pancreatic and peripancreatic fluid collections; bacteremia and sepsis can occur in association with infection of necrotic collections.
Interstitial edematous pancreatitis vs necrotizing pancreatitis
The revised Atlanta classification maintains the original classification of acute pancreatitis into 2 main categories: interstitial edematous pancreatitis and necrotizing pancreatitis.
Necrotizing pancreatitis is further divided into 3 subtypes based on extent and location of necrosis:
- Parenchymal necrosis alone (5% of cases)
- Necrosis of peripancreatic fat alone (20%)
- Necrosis of both parenchyma and peripancreatic fat (75%).
Peripancreatic involvement is commonly found in the mesentery, peripancreatic and distant retroperitoneum, and lesser sac.
Of the three subtypes, peripancreatic necrosis has the best prognosis. However, all of the subtypes of necrotizing pancreatitis are associated with poorer outcomes than interstitial edematous pancreatitis.
Fluid collections
Acute pancreatic fluid collections contain exclusively nonsolid components without an inflammatory wall and are typically found in the peripancreatic fat. These collections often resolve without intervention as the patient recovers. If they persist beyond 4 weeks and develop a nonepithelialized, fibrous wall, they become pseudocysts. Intervention is generally not recommended for pseudocysts unless they are symptomatic.
ROLE OF IMAGING
Radiographic imaging is not usually necessary to diagnose acute pancreatitis. However, it can be a valuable tool to clarify an ambiguous presentation, determine severity, and identify complications.
The timing and appropriate type of imaging are integral to obtaining useful data. Any imaging obtained in acute pancreatitis to evaluate necrosis should be performed at least 3 to 5 days from the initial symptom onset; if imaging is obtained before 72 hours, necrosis cannot be confidently excluded.8
COMPUTED TOMOGRAPHY
CT is the imaging test of choice when evaluating acute pancreatitis. In addition, almost all percutaneous interventions are performed with CT guidance. The Balthazar score is the most well-known CT severity index. It is calculated based on the degree of inflammation, acute fluid collections, and parenchymal necrosis.9 However, a modified severity index incorporates extrapancreatic complications such as ascites and vascular compromise and was found to more strongly correlate with outcomes than the standard Balthazar score.10
Contrast-enhanced CT is performed in 2 phases:
The pancreatic parenchymal phase
The pancreatic parenchymal or late arterial phase is obtained approximately 40 to 45 seconds after the start of the contrast bolus. It is used to detect necrosis in the early phase of acute pancreatitis and to assess the peripancreatic arteries for pseudoaneurysms in the late phase of acute pancreatitis.11
Pancreatic necrosis appears as an area of decreased parenchymal enhancement, either well-defined or heterogeneous. The normal pancreatic parenchyma has a postcontrast enhancement pattern similar to that of the spleen. Parenchyma that does not enhance to the same degree is considered necrotic. The severity of necrosis is graded based on the percentage of the pancreas involved (< 30%, 30%–50%, or > 50%), and a higher percentage correlates with a worse outcome.12,13
Peripancreatic necrosis is harder to detect, as there is no method to assess fat enhancement as there is with pancreatic parenchymal enhancement. In general, radiologists assume that heterogeneous peripancreatic changes, including areas of fat, fluid, and soft tissue attenuation, are consistent with peripancreatic necrosis. After 7 to 10 days, if these changes become more homogeneous and confluent with a more mass-like process, peripancreatic necrosis can be more confidently identified.12,13
The portal venous phase
The later, portal venous phase of the scan is obtained approximately 70 seconds after the start of the contrast bolus. It is used to detect and characterize fluid collections and venous complications of the disease.
Drawbacks of CT
A drawback of CT is the need for iodinated intravenous contrast media, which in severely ill patients may precipitate or worsen pre-existing acute kidney injury.
Further, several studies have shown that findings on CT rarely alter the management of patients in the early phase of acute pancreatitis and in fact may be an overuse of medical resources.14 Unless there are confounding clinical signs or symptoms, CT should be delayed for at least 72 hours.9,10,14,15
MAGNETIC RESONANCE IMAGING
Magnetic resonance imaging (MRI) is not a first-line imaging test in this disease because it is not as available as CT and takes longer to perform—20 to 30 minutes. The patient must be evaluated for candidacy, as it is difficult for acutely ill patients to tolerate an examination that takes this long and requires them to hold their breath multiple times.
MRI is an appropriate alternative in patients who are pregnant or who have severe iodinated-contrast allergy. While contrast is necessary to detect pancreatic necrosis with CT, MRI can detect necrosis without the need for contrast in patients with acute kidney injury or severe chronic kidney disease. Also, MRI may be better in complicated cases requiring repeated imaging because it does not expose the patient to radiation.
On MRI, pancreatic necrosis appears as a heterogeneous area, owing to its liquid and solid components. Liquid components appear hyperintense, and solid components hypointense, on T2 fluid-weighted imaging. This ability to differentiate the components of a walled-off pancreatic necrosis can be useful in determining whether a collection requires drainage or debridement. MRI is also more sensitive for hemorrhagic complications, best seen on T1 fat-weighted images.12,16
Magnetic resonance cholangiopancreatography is an excellent method for ductal evaluation through heavily T2-weighted imaging. It is more sensitive than CT for detecting common bile duct stones and can also detect pancreatic duct strictures or extravasation into fluid collections.16
SUPPORTIVE MANAGEMENT OF EARLY NECROTIZING PANCREATITIS
In the early phase of necrotizing pancreatitis, management is supportive with the primary aim of preventing intravascular volume depletion. Aggressive fluid resuscitation in the first 48 to 72 hours, pain control, and bowel rest are the mainstays of supportive therapy. Intensive care may be necessary if organ failure and hemodynamic instability accompany necrotizing pancreatitis.
Prophylactic antibiotic and antifungal therapy to prevent infected necrosis has been controversial. Recent studies of its utility have not yielded supportive results, and the American College of Gastroenterology and the Infectious Diseases Society of America no longer recommend it.9,17 These medications should not be given unless concomitant cholangitis or extrapancreatic infection is clinically suspected.
Early enteral nutrition is recommended in patients in whom pancreatitis is predicted to be severe and in those not expected to resume oral intake within 5 to 7 days. Enteral nutrition most commonly involves bedside or endoscopic placement of a nasojejunal feeding tube and collaboration with a nutritionist to determine protein-caloric requirements.
Compared with enteral nutrition, total parenteral nutrition is associated with higher rates of infection, multiorgan dysfunction and failure, and death.18
MANAGING COMPLICATIONS OF PANCREATIC NECROSIS
Necrotizing pancreatitis is a defining complication of acute pancreatitis, and its presence alone indicates greater severity. However, superimposed complications may further worsen outcomes.
Infected pancreatic necrosis
Infection occurs in approximately 20% of patients with necrotizing pancreatitis and confers a mortality rate of 20% to 50%.19 Infected pancreatic necrosis occurs when gut organisms translocate into the nearby necrotic pancreatic and peripancreatic tissue. The most commonly identified organisms include Escherichia coli and Enterococcus species.20
This complication usually manifests 2 to 4 weeks after symptom onset; earlier onset is uncommon to rare. It should be considered when the systemic inflammatory response syndrome persists or recurs after 10 days to 2 weeks. Systemic inflammatory response syndrome is also common in sterile necrotizing pancreatitis and sometimes in interstitial pancreatitis, particularly during the first week. However, its sudden appearance or resurgence, high spiking fevers, or worsening organ failure in the later phase (2–4 weeks) of pancreatitis should heighten suspicion of infected pancreatic necrosis.
Imaging may also help diagnose infection, and the presence of gas within a collection or region of necrosis is highly specific. However, the presence of gas is not completely sensitive for infection, as it is seen in only 12% to 22% of infected cases.
Before minimally invasive techniques became available, the diagnosis of infected pancreatic necrosis was confirmed by percutaneous CT-guided aspiration of the necrotic mass or collection for Gram stain and culture.
Antibiotic therapy is indicated in confirmed or suspected cases of infected pancreatic necrosis. Antibiotics with gram-negative coverage and appropriate penetration such as carbapenems, metronidazole, fluoroquinolones, and selected cephalosporins are most commonly used. Meropenem is the antibiotic of choice at our institution.
CT-guided fine-needle aspiration is often done if suspected infected pancreatic necrosis fails to respond to empiric antibiotic therapy.
Debridement or drainage. Generally, the diagnosis or suspicion of infected pancreatic necrosis (suggestive signs are high fever, elevated white blood cell count, and sepsis) warrants an intervention to debride or drain infected pancreatic tissue and control sepsis.21
While source control is integral to the successful treatment of infected pancreatic necrosis, antibiotic therapy may provide a bridge to intervention for critically ill patients by suppressing bacteremia and subsequent sepsis. A 2013 meta-analysis found that 324 of 409 patients with suspected infected pancreatic necrosis were successfully stabilized with antibiotic treatment.21,22 The trend toward conservative management and promising outcomes with antibiotic therapy alone or with minimally invasive techniques has lessened the need for diagnostic CT-guided fine-needle aspiration.
Hemorrhage
Spontaneous hemorrhage into pancreatic necrosis is a rare but life-threatening complication. Because CT is almost always performed with contrast enhancement, this complication is rarely identified with imaging. The diagnosis is made by noting a drop in hemoglobin and hematocrit.
Hemorrhage into the retroperitoneum or the peritoneal cavity, or both, can occur when an inflammatory process erodes into a nearby artery. Luminal gastrointestinal bleeding can occur from gastric varices arising from splenic vein thrombosis and resulting left-sided portal hypertension, or from pseudoaneurysms. These can also bleed into the pancreatic duct (hemosuccus pancreaticus). Pseudoaneurysm is a later complication that occurs when an arterial wall (most commonly the splenic or gastroduodenal artery) is weakened by pancreatic enzymes.23
Prompt recognition of hemorrhagic events and consultation with an interventional radiologist or surgeon are required to prevent death.
Inflammation and abdominal compartment syndrome
Inflammation from necrotizing pancreatitis can cause further complications by blocking nearby structures. Reported complications include jaundice from biliary compression, hydronephrosis from ureteral compression, bowel obstruction, and gastric outlet obstruction.
Abdominal compartment syndrome is an increasingly recognized complication of acute pancreatitis. Abdominal pressure can rise due to a number of factors, including fluid collections, ascites, ileus, and overly aggressive fluid resuscitation.24 Elevated abdominal pressure is associated with complications such as decreased respiratory compliance, increased peak airway pressure, decreased cardiac preload, hypotension, mesenteric and intestinal ischemia, feeding intolerance, and lower-extremity ischemia and thrombosis.
Patients with necrotizing pancreatitis who have abdominal compartment syndrome have a mortality rate 5 times higher than patients without abdominal compartment syndrome.25
Abdominal pressures should be monitored using a bladder pressure sensor in critically ill or ventilated patients with acute pancreatitis. If the abdominal pressure rises above 20 mm Hg, medical and surgical interventions should be offered in a stepwise fashion to decrease it. Interventions include decompression by nasogastric and rectal tube, sedation or paralysis to relax abdominal wall tension, minimization of intravenous fluids, percutaneous drainage of ascites, and (rarely) surgical midline or subcostal laparotomy.
ROLE OF INTERVENTION
The treatment of necrotizing pancreatitis has changed rapidly, thanks to a growing experience with minimally invasive techniques.
Indications for intervention
Infected pancreatic necrosis is the primary indication for surgical, percutaneous, or endoscopic intervention.
In sterile necrosis, the threshold for intervention is less clear, and intervention is often reserved for patients who fail to clinically improve or who have intractable abdominal pain, gastric outlet obstruction, or fistulating disease.26
In asymptomatic cases, intervention is almost never indicated regardless of the location or size of the necrotic area.
In walled-off pancreatic necrosis, less-invasive and less-morbid interventions such as endoscopic or percutaneous drainage or video-assisted retroperitoneal debridement can be done.
Timing of intervention
In the past, delaying intervention was thought to increase the risk of death. However, multiple studies have found that outcomes are often worse if intervention is done early, likely due to the lack of a fully formed fibrous wall or demarcation of the necrotic area.27
If the patient remains clinically stable, it is best to delay intervention until at least 4 weeks after the index event to achieve optimal outcomes. Delay can often be achieved by antibiotic treatment to suppress bacteremia and endoscopic or percutaneous drainage of infected collections to control sepsis.
Open surgery
The gold-standard intervention for infected pancreatic necrosis or symptomatic sterile walled-off pancreatic necrosis is open necrosectomy. This involves exploratory laparotomy with blunt debridement of all visible necrotic pancreatic tissue.
Methods to facilitate later evacuation of residual infected fluid and debris vary widely. Multiple large-caliber drains can be placed to facilitate irrigation and drainage before closure of the abdominal fascia. As infected pancreatic necrosis carries the risk of contaminating the peritoneal cavity, the skin is often left open to heal by secondary intention. An interventional radiologist is frequently enlisted to place, exchange, or downsize drainage catheters.
Infected pancreatic necrosis or symptomatic sterile walled-off pancreatic necrosis often requires more than one operation to achieve satisfactory debridement.
The goals of open necrosectomy are to remove nonviable tissue and infection, preserve viable pancreatic tissue, eliminate fistulous connections, and minimize damage to local organs and vasculature.
Minimally invasive techniques
Video-assisted retroperitoneal debridement has been described as a hybrid between endoscopic and open retroperitoneal debridement.28 This technique requires first placing a percutaneous catheter into the necrotic area through the left flank to create a retroperitoneal tract. A 5-cm incision is made and the necrotic space is entered using the drain for guidance. Necrotic tissue is carefully debrided under direct vision using a combination of forceps, irrigation, and suction. A laparoscopic port can also be introduced into the incision when the procedure can no longer be continued under direct vision.29,30
Although not all patients are candidates for minimal-access surgery, it remains an evolving surgical option.
Endoscopic transmural debridement is another option for infected pancreatic necrosis and symptomatic walled-off pancreatic necrosis. Depending on the location of the necrotic area, an echoendoscope is passed to either the stomach or duodenum. Guided by endoscopic ultrasonography, a needle is passed into the collection, allowing subsequent fistula creation and stenting for internal drainage or debridement. In the past, this process required several steps, multiple devices, fluoroscopic guidance, and considerable time. But newer endoscopic lumen-apposing metal stents have been developed that can be placed in a single step without fluoroscopy. A slimmer endoscope can then be introduced into the necrotic cavity via the stent, and the necrotic debris can be debrided with endoscopic baskets, snares, forceps, and irrigation.9,31
Similar to surgical necrosectomy, satisfactory debridement is not often obtained with a single procedure; 2 to 5 endoscopic procedures may be needed to achieve resolution. However, the luminal approach in endoscopic necrosectomy avoids the significant morbidity of major abdominal surgery and the potential for pancreaticocutaneous fistulae that may occur with drains.
In a randomized trial comparing endoscopic necrosectomy vs surgical necrosectomy (video-assisted retroperitoneal debridement and exploratory laparotomy),32 endoscopic necrosectomy showed less inflammatory response than surgical necrosectomy and had a lower risk of new-onset organ failure, bleeding, fistula formation, and death.32
Selecting the best intervention for the individual patient
Given the multiple available techniques, selecting the best intervention for individual patients can be challenging. A team approach with input from a gastroenterologist, surgeon, and interventional radiologist is best when determining which technique would best suit each patient.
Surgical necrosectomy is still the treatment of choice for unstable patients with infected pancreatic necrosis or multiple, inaccessible collections, but current evidence suggests a different approach in stable infected pancreatic necrosis and symptomatic sterile walled-off pancreatic necrosis.
The Dutch Pancreatitis Group28 randomized 88 patients with infected pancreatic necrosis or symptomatic walled-off pancreatic necrosis to open necrosectomy or a minimally invasive “step-up” approach consisting of up to 2 percutaneous drainage or endoscopic debridement procedures before escalation to video-assisted retroperitoneal debridement. The step-up approach resulted in lower rates of morbidity and death than surgical necrosectomy as first-line treatment. Furthermore, some patients in the step-up group avoided the need for surgery entirely.30
SUMMING UP
Necrosis significantly increases rates of morbidity and mortality in acute pancreatitis. Hospitalists, general internists, and general surgeons are all on the front lines in identifying severe cases and consulting the appropriate specialists for optimal multidisciplinary care. Selective and appropriate timing of radiologic imaging is key, and a vital tool in the management of necrotizing pancreatitis.
While the primary indication for intervention is infected pancreatic necrosis, additional indications are symptomatic walled-off pancreatic necrosis secondary to intractable abdominal pain, bowel obstruction, and failure to thrive. As a result of improving technology and inpatient care, these patients may present with intractable symptoms in the outpatient setting rather than the inpatient setting. The onus is on the primary care physician to maintain a high level of suspicion and refer these patients to subspecialists as appropriate.
Open surgical necrosectomy remains an important approach for care of infected pancreatic necrosis or patients with intractable symptoms. A step-up approach starting with a minimally invasive procedure and escalating if the initial intervention is unsuccessful is gradually becoming the standard of care.
- Peery AF, Crockett SD, Barritt AS, et al. Burden of gastrointestinal, liver, and pancreatic disease in the United States. Gastroenterology 2015; 149:1731–1741e3.
- Tenner S, Baillie J, DeWitt J, Vege SS; American College of Gastroenterology. American College of Gastroenterology guideline: management of acute pancreatitis. Am J Gastroenterol 2013; 108:1400–1416.
- Bradley EL 3rd. A clinically based classification system for acute pancreatitis. Summary of the International Symposium on Acute Pancreatitis, Atlanta, GA, September 11 through 13, 1992. Arch Surg 1993; 128:586–590.
- Banks PA, Bollen TL, Dervenis C, et al; Acute Pancreatitis Classification Working Group. Classification of acute pancreatitis—2012: revision of the Atlanta classification and definitions by international consensus. Gut 2013; 62:102–111.
- Marshall JC, Cook DJ, Christou NV, Bernard GR, Sprung CL, Sibbald WJ. Multiple organ dysfunction score: a reliable descriptor of a complex clinical outcome. Crit Care Med 1995; 23:1638–1652.
- Kadiyala V, Suleiman SL, McNabb-Baltar J, Wu BU, Banks PA, Singh VK. The Atlanta classification, revised Atlanta classification, and determinant-based classification of acute pancreatitis: which is best at stratifying outcomes? Pancreas 2016; 45:510–515.
- Singh VK, Bollen TL, Wu BU, et al. An assessment of the severity of interstitial pancreatitis. Clin Gastroenterol Hepatol 2011; 9:1098–1103.
- Kotwal V, Talukdar R, Levy M, Vege SS. Role of endoscopic ultrasound during hospitalization for acute pancreatitis. World J Gastroenterol 2010; 16:4888–4891.
- Balthazar EJ. Acute pancreatitis: assessment of severity with clinical and CT evaluation. Radiology 2002; 223:603–613.
- Mortele KJ, Wiesner W, Intriere L, et al. A modified CT severity index for evaluating acute pancreatitis: improved correlation with patient outcome. AJR Am J Roentgenol 2004; 183:1261–1265.
- Verde F, Fishman EK, Johnson PT. Arterial pseudoaneurysms complicating pancreatitis: literature review. J Comput Assist Tomogr 2015; 39:7–12.
- Shyu JY, Sainani NI, Sahni VA, et al. Necrotizing pancreatitis: diagnosis, imaging, and intervention. Radiographics 2014; 34:1218–1239.
- Thoeni RF. The revised Atlanta classification of acute pancreatitis: its importance for the radiologist and its effect on treatment. Radiology 2012; 262:751–764.
- Morgan DE, Ragheb CM, Lockhart ME, Cary B, Fineberg NS, Berland LL. Acute pancreatitis: computed tomography utilization and radiation exposure are related to severity but not patient age. Clin Gastroenterol Hepatol 2010; 8:303–308.
- Vitellas KM, Paulson EK, Enns RA, Keogan MT, Pappas TN. Pancreatitis complicated by gland necrosis: evolution of findings on contrast-enhanced CT. J Comput Assist Tomogr 1999; 23:898–905.
- Stimac D, Miletic D, Radic M, et al. The role of nonenhanced magnetic resonance imaging in the early assessment of acute pancreatitis. Am J Gastroenterol 2007; 102:997–1004.
- Solomkin JS, Mazuski JE, Bradley JS, et al. Diagnosis and management of complicated intra-abdominal infection in adults and children: guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Surg Infect (Larchmt) 2010; 11:79–109.
- Petrov MS, Kukosh MV, Emelyanov NV. A randomized controlled trial of enteral versus parenteral feeding in patients with predicted severe acute pancreatitis shows a significant reduction in mortality and in infected pancreatic complications with total enteral nutrition. Dig Surg 2006; 23:336–345.
- Petrov MS, Shanbhag S, Chakraborty M, Phillips AR, Windsor JA. Organ failure and infection of pancreatic necrosis as determinants of mortality in patients with acute pancreatitis. Gastroenterology 2010; 139:813–820.
- Villatoro E, Bassi C, Larvin M. Antibiotic therapy for prophylaxis against infection of pancreatic necrosis in acute pancreatitis. Cochrane Database Syst Rev 2006; 4:CD002941.
- Baril NB, Ralls PW, Wren SM, et al. Does an infected peripancreatic fluid collection or abscess mandate operation? Ann Surg 2000; 231:361–367.
- Mouli VP, Sreenivas V, Garg PK. Efficacy of conservative treatment, without necrosectomy, for infected pancreatic necrosis: a systematic review and meta-analysis. Gastroenterology 2013; 144:333–340.e2.
- Kirby JM, Vora P, Midia M, Rawlinson J. Vascular complications of pancreatitis: imaging and intervention. Cardiovasc Intervent Radiol 2008; 31:957–970.
- De Waele JJ, Hoste E, Blot SI, Decruyenaere J, Colardyn F. Intra-abdominal hypertension in patients with severe acute pancreatitis. Crit Care 2005; 9:R452–R457.
- van Brunschot S, Schut AJ, Bouwense SA, et al; Dutch Pancreatitis Study Group. Abdominal compartment syndrome in acute pancreatitis: a systematic review. Pancreas 2014; 43:665–674.
- Bugiantella W, Rondelli F, Boni M, et al. Necrotizing pancreatitis: a review of the interventions. Int J Surg 2016; 28(suppl 1):S163–S171.
- Besselink MG, Verwer TJ, Schoenmaeckers EJ, et al. Timing of surgical intervention in necrotizing pancreatitis. Arch Surg 2007; 142:1194–1201.
- van Santvoort HC, Besselink MG, Horvath KD, et al; Dutch Acute Pancreatis Study Group. Videoscopic assisted retroperitoneal debridement in infected necrotizing pancreatitis. HPB (Oxford) 2007; 9:156–159.
- van Santvoort HC, Besselink MG, Bollen TL, Buskens E, van Ramshorst B, Gooszen HG; Dutch Acute Pancreatitis Study Group. Case-matched comparison of the retroperitoneal approach with laparotomy for necrotizing pancreatitis. World J Surg 2007; 31:1635–1642.
- van Santvoort HC, Besselink MG, Bakker OJ, et al; Dutch Pancreatitis Study Group. A step-up approach or open necrosectomy for necrotizing pancreatitis. N Engl J Med 2010; 362:1491–1502.
- Thompson CC, Kumar N, Slattery J, et al. A standardized method for endoscopic necrosectomy improves complication and mortality rates. Pancreatology 2016; 16:66–72.
- Bakker OJ, van Santvoort HC, van Brunschot S, et al; Dutch Pancreatitis Study Group. Endoscopic transgastric vs surgical necrosectomy for infected necrotizing pancreatitis: a randomized trial. JAMA 2012; 307:1053–1061.
- Peery AF, Crockett SD, Barritt AS, et al. Burden of gastrointestinal, liver, and pancreatic disease in the United States. Gastroenterology 2015; 149:1731–1741e3.
- Tenner S, Baillie J, DeWitt J, Vege SS; American College of Gastroenterology. American College of Gastroenterology guideline: management of acute pancreatitis. Am J Gastroenterol 2013; 108:1400–1416.
- Bradley EL 3rd. A clinically based classification system for acute pancreatitis. Summary of the International Symposium on Acute Pancreatitis, Atlanta, GA, September 11 through 13, 1992. Arch Surg 1993; 128:586–590.
- Banks PA, Bollen TL, Dervenis C, et al; Acute Pancreatitis Classification Working Group. Classification of acute pancreatitis—2012: revision of the Atlanta classification and definitions by international consensus. Gut 2013; 62:102–111.
- Marshall JC, Cook DJ, Christou NV, Bernard GR, Sprung CL, Sibbald WJ. Multiple organ dysfunction score: a reliable descriptor of a complex clinical outcome. Crit Care Med 1995; 23:1638–1652.
- Kadiyala V, Suleiman SL, McNabb-Baltar J, Wu BU, Banks PA, Singh VK. The Atlanta classification, revised Atlanta classification, and determinant-based classification of acute pancreatitis: which is best at stratifying outcomes? Pancreas 2016; 45:510–515.
- Singh VK, Bollen TL, Wu BU, et al. An assessment of the severity of interstitial pancreatitis. Clin Gastroenterol Hepatol 2011; 9:1098–1103.
- Kotwal V, Talukdar R, Levy M, Vege SS. Role of endoscopic ultrasound during hospitalization for acute pancreatitis. World J Gastroenterol 2010; 16:4888–4891.
- Balthazar EJ. Acute pancreatitis: assessment of severity with clinical and CT evaluation. Radiology 2002; 223:603–613.
- Mortele KJ, Wiesner W, Intriere L, et al. A modified CT severity index for evaluating acute pancreatitis: improved correlation with patient outcome. AJR Am J Roentgenol 2004; 183:1261–1265.
- Verde F, Fishman EK, Johnson PT. Arterial pseudoaneurysms complicating pancreatitis: literature review. J Comput Assist Tomogr 2015; 39:7–12.
- Shyu JY, Sainani NI, Sahni VA, et al. Necrotizing pancreatitis: diagnosis, imaging, and intervention. Radiographics 2014; 34:1218–1239.
- Thoeni RF. The revised Atlanta classification of acute pancreatitis: its importance for the radiologist and its effect on treatment. Radiology 2012; 262:751–764.
- Morgan DE, Ragheb CM, Lockhart ME, Cary B, Fineberg NS, Berland LL. Acute pancreatitis: computed tomography utilization and radiation exposure are related to severity but not patient age. Clin Gastroenterol Hepatol 2010; 8:303–308.
- Vitellas KM, Paulson EK, Enns RA, Keogan MT, Pappas TN. Pancreatitis complicated by gland necrosis: evolution of findings on contrast-enhanced CT. J Comput Assist Tomogr 1999; 23:898–905.
- Stimac D, Miletic D, Radic M, et al. The role of nonenhanced magnetic resonance imaging in the early assessment of acute pancreatitis. Am J Gastroenterol 2007; 102:997–1004.
- Solomkin JS, Mazuski JE, Bradley JS, et al. Diagnosis and management of complicated intra-abdominal infection in adults and children: guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Surg Infect (Larchmt) 2010; 11:79–109.
- Petrov MS, Kukosh MV, Emelyanov NV. A randomized controlled trial of enteral versus parenteral feeding in patients with predicted severe acute pancreatitis shows a significant reduction in mortality and in infected pancreatic complications with total enteral nutrition. Dig Surg 2006; 23:336–345.
- Petrov MS, Shanbhag S, Chakraborty M, Phillips AR, Windsor JA. Organ failure and infection of pancreatic necrosis as determinants of mortality in patients with acute pancreatitis. Gastroenterology 2010; 139:813–820.
- Villatoro E, Bassi C, Larvin M. Antibiotic therapy for prophylaxis against infection of pancreatic necrosis in acute pancreatitis. Cochrane Database Syst Rev 2006; 4:CD002941.
- Baril NB, Ralls PW, Wren SM, et al. Does an infected peripancreatic fluid collection or abscess mandate operation? Ann Surg 2000; 231:361–367.
- Mouli VP, Sreenivas V, Garg PK. Efficacy of conservative treatment, without necrosectomy, for infected pancreatic necrosis: a systematic review and meta-analysis. Gastroenterology 2013; 144:333–340.e2.
- Kirby JM, Vora P, Midia M, Rawlinson J. Vascular complications of pancreatitis: imaging and intervention. Cardiovasc Intervent Radiol 2008; 31:957–970.
- De Waele JJ, Hoste E, Blot SI, Decruyenaere J, Colardyn F. Intra-abdominal hypertension in patients with severe acute pancreatitis. Crit Care 2005; 9:R452–R457.
- van Brunschot S, Schut AJ, Bouwense SA, et al; Dutch Pancreatitis Study Group. Abdominal compartment syndrome in acute pancreatitis: a systematic review. Pancreas 2014; 43:665–674.
- Bugiantella W, Rondelli F, Boni M, et al. Necrotizing pancreatitis: a review of the interventions. Int J Surg 2016; 28(suppl 1):S163–S171.
- Besselink MG, Verwer TJ, Schoenmaeckers EJ, et al. Timing of surgical intervention in necrotizing pancreatitis. Arch Surg 2007; 142:1194–1201.
- van Santvoort HC, Besselink MG, Horvath KD, et al; Dutch Acute Pancreatis Study Group. Videoscopic assisted retroperitoneal debridement in infected necrotizing pancreatitis. HPB (Oxford) 2007; 9:156–159.
- van Santvoort HC, Besselink MG, Bollen TL, Buskens E, van Ramshorst B, Gooszen HG; Dutch Acute Pancreatitis Study Group. Case-matched comparison of the retroperitoneal approach with laparotomy for necrotizing pancreatitis. World J Surg 2007; 31:1635–1642.
- van Santvoort HC, Besselink MG, Bakker OJ, et al; Dutch Pancreatitis Study Group. A step-up approach or open necrosectomy for necrotizing pancreatitis. N Engl J Med 2010; 362:1491–1502.
- Thompson CC, Kumar N, Slattery J, et al. A standardized method for endoscopic necrosectomy improves complication and mortality rates. Pancreatology 2016; 16:66–72.
- Bakker OJ, van Santvoort HC, van Brunschot S, et al; Dutch Pancreatitis Study Group. Endoscopic transgastric vs surgical necrosectomy for infected necrotizing pancreatitis: a randomized trial. JAMA 2012; 307:1053–1061.
KEY POINTS
- Selective and appropriate timing of radiologic imaging is vital in managing necrotizing pancreatitis. Protocols are valuable tools.
- While the primary indication for debridement and drainage in necrotizing pancreatitis is infection, other indications are symptomatic walled-off pancreatic necrosis, intractable abdominal pain, bowel obstruction, and failure to thrive.
- Open surgical necrosectomy remains an important treatment for infected pancreatic necrosis or intractable symptoms.
- A “step-up” approach starting with a minimally invasive procedure and escalating if the initial intervention is unsuccessful is gradually becoming the standard of care.
Acute pancreatitis: Problems in adherence to guidelines
Several major gastroenterological and surgical societies have issued guidelines on how to manage acute pancreatitis, based on evidence from high-quality randomized trials and nonrandomized studies as well as on expert opinion.1–3 Information is limited on how well physicians in the United States comply with these guidelines, but compliance is suboptimal in other developed countries, according to several studies,4–8 and we suspect that many US physicians are not following the guidelines either.
Acute pancreatitis is a frequent inpatient diagnosis that internists, gastroenterologists, and surgeons all confront. The most common causes are gallstones and heavy alcohol intake. Its management is typically straightforward: intravenous fluids, analgesia, and nothing by mouth. However, treatment of severe cases can be quite complex, particularly if multiple organ systems are involved or if there are local complications.
The primary aim of this article is to raise awareness of recognized deviations from established recommendations that may lead to adverse patient outcomes.
MEASURING ENZYME LEVELS DAILY ADDS COST BUT LITTLE BENEFIT
Problem: Serum amylase and lipase levels are often needlessly measured every day.
Measuring the serum amylase and lipase levels is useful in diagnosing acute pancreatitis, which requires two of the following three features1:
- Characteristic abdominal pain
- Levels of serum amylase or serum lipase, or both, that are three or more times the upper limit of normal
- Findings of acute pancreatitis on computed tomography (CT).
However, the magnitude or duration of the serum enzyme elevation does not correlate with the severity of the attack. Further, we have noticed that physicians at our hospital often order daily serum amylase and lipase levels in patients admitted with acute pancreatitis.
The American College of Gastroenterology (ACG) guidelines1 state that daily monitoring of amylase and lipase has limited value in managing acute pancreatitis. Rechecking these concentrations may be reasonable if pain fails to resolve or worsens during a prolonged hospitalization, as this may suggest a recurrent attack of acute pancreatitis or a developing pseudocyst. But in most cases of acute pancreatitis, daily serum enzyme measurements add cost but little benefit.
REGULAR ASSESSMENT IS IMPORTANT
Problem: Often, severity assessments are not performed regularly or acted on.
Most cases of acute pancreatitis are mild, with rapid recovery and excellent prognosis. However, 15% to 20% are severe and may result in a prolonged hospitalization, systemic inflammatory response syndrome (SIRS), multiorgan system failure, and death.
In severe acute pancreatitis, as pancreatic enzymes and inflammatory cytokines damage the blood vessels, a vast amount of fluid leaks out into the interstitial (“third”) space. This fluid extravasation leads to decreased effective circulating volume, local pancreatic necrosis, hemodynamic instability, and end-organ failure.
It is important to recognize severe acute pancreatitis early because the patient needs to be transferred to a step-down unit or intensive care unit to receive optimal fluid resuscitation and supportive care for organ dysfunction. After 48 to 72 hours, a prediction of severe acute pancreatitis should also prompt the physician to order CT to detect pancreatic necrosis, and also to initiate nutritional support.
Assessment of severity begins in the emergency room or on admission to the hospital. Older age, obesity, organ failure, and pulmonary infiltrates or pleural effusions are initial indicators of poor prognosis. Signs of SIRS (high or low core body temperature, tachycardia, tachypnea, low or high peripheral white blood cell count) or organ failure (eg, elevated serum creatinine) are present on admission in 21% of patients with acute pancreatitis.9
Hemoconcentration is a marker of decreased effective circulating volume in severe acute pancreatitis. A hematocrit higher than 44% at admission or that rises in the first 24 to 48 hours of admission predicts necrosis.10,11 However, a more robust marker of organ failure may be the blood urea nitrogen level.12
Clinical scoring systems
Several clinical scoring systems have been studied for assessing severity.
Limitations of the Ranson score are that it can only be completed after 48 hours, all the data points are not always obtained, and it cannot be repeated on a daily basis. Owing to these limitations and its less-than-optimal predictive value, the Ranson score has fallen into disuse.
The APACHE II (Acute Physiology and Chronic Health Evaluation II) score is more versatile. It is based on multiple clinical and laboratory values, and it correlates very well with the risk of death in acute pancreatitis. Death rates are less than 4% when the APACHE II score is less than 8, and 11% to 18% when it is 8 or higher.1 The trajectory of the APACHE II score in the first 48 hours is also an accurate prognostic indicator.
Previous limitations of the APACHE II score were that it was complicated and timeconsuming to calculate and required arterial blood gas measurements. Easy-to-use online calculators are now available (eg, www.globalrph.com/apacheii.htm), and the venous bicarbonate level and the oxygen saturation can be substituted for the arterial pH and oxygen partial pressure.
BISAP, a new five-point scoring system,15 was recently prospectively validated.12 “BISAP” is an acronym for the five markers it is based on, each of which has been shown to predict severe illness in acute pancreatitis:
- Blood urea nitrogen level > 25 mg/dL
- Impaired mental status
- SIRS
- Age > 60 years
- Pleural effusion.
The presence of three or more of these factors correlates with higher risk of death, organ failure, and pancreatic necrosis.12
Compared with APACHE II, BISAP has similar accuracy and is easier to calculate. Also, BISAP was specifically developed for acute pancreatitis, whereas APACHE II is a generic score for all critically ill patients.
The Atlanta criteria16 define severe acute pancreatitis as one or more of the following:
- A Ranson score of 3 or higher during the first 48 hours
- An APACHE II score of 8 or higher at any time
- Failure of one or more organs
- One or more local complications (eg, necrosis, pseudocysts, abscesses).
Recommendation: Assess severity at least daily
A severity assessment should be performed at admission and at least every day thereafter. Clinical guidelines recognize the importance of severity assessment but vary in their specific recommendations.
The ACG advises calculating the APACHE II score within 3 days of admission and measuring the hematocrit at admission, at 12 hours, and at 24 hours. The level of evidence is III, ie, “from published well-designed trials without randomization, single group prepost, cohort, time series, or matched case controlled studies”.1
The American Gastroenterological Association (AGA) provides a more generalized recommendation, that “clinical judgment” should take into account the presence of risk factors (eg, age, obesity), presence or absence of SIRS, routine laboratory values (eg, hematocrit, serum creatinine), and APACHE II score when assessing severity and making decisions.2
In a German survey, only 32% of gastroenterologists used the APACHE II score for assessing risk in acute pancreatitis, in spite of national guidelines emphasizing its importance.7 Also, not all patients with severe acute pancreatitis are transferred to a step-down unit or intensive care unit as recommended. In a British study,4 only 8 (17%) of 46 patients with predicted severe acute pancreatitis were transferred, and 8 of the 38 patients who were not transferred died.
FLUID MUST BE AGGRESSIVELY REPLACED AND MONITORED
Problem: Often, not enough fluid is replaced, or fluid status is not adequately monitored.
Fluid must be aggressively replaced to balance the massive third-space fluid losses that occur in the early inflammatory phase of acute pancreatitis. Intravascular volume depletion can develop rapidly and result in tachycardia, hypotension, and renal failure. It may also impair the blood flow to the pancreas and worsen necrosis.
Animal studies show that aggressive fluid replacement supports the pancreatic microcirculation and prevents necrosis.17 It may also support the intestinal microcirculation and gut barrier, preventing bacterial translocation.
In humans, no controlled trials have been done to test the efficacy of aggressive fluid resuscitation in acute pancreatitis. However, the notion that intravascular fluid loss contributes to poor outcomes is inferred from human studies showing more necrosis and deaths in patients with hemoconcentration. In one study, patients who received inadequate fluid replacement (evidenced by a rise in hematocrit at 24 hours) were more likely to develop necrotizing pancreatitis.18
Recommendation: Early, aggressive fluid replacement
Experts have suggested initially infusing 500 to 1,000 mL of fluid per hour in those who are volume-depleted, initially infusing 250 to 350 mL per hour in those who are not volumedepleted, and adjusting the fluid rate every 1 to 4 hours on the basis of clinical variables.19 The sufficiency of fluid replacement should be carefully monitored by vital signs, urine output, and serum hematocrit.
On the other hand, overly aggressive fluid resuscitation can be detrimental in patients at risk of volume overload or pulmonary edema. Fluid replacement should be tempered in elderly patients and those with cardiac or renal comorbidities, and may require monitoring of central venous pressure.
The ACG and AGA guidelines both recognize the need for early aggressive volume replacement in acute pancreatitis (level of evidence III), but they do not specify the exact amounts and rates. Young and healthy patients should receive a rapid bolus of isotonic saline or Ringer’s lactate solution followed by an infusion at a high initial maintenance rate.
Few studies have been done to assess physicians’ compliance with recommendations for aggressive volume replacement. In an Italian multicenter study, patients with mild or severe acute pancreatitis received an average of only 2.5 L of fluid per day (about 100 mL/hour).20 Gardner et al21 recently summarized the available evidence for fluid support in acute pancreatitis.
NUTRITIONAL SUPPORT
Problem: In many severe cases, enteral or parenteral feeding is not started soon enough.
Nutritional support entails enteral or parenteral feeding when an oral diet is contraindicated. Enteral feeding is usually via a nasojejunal tube, which may need to be placed under endoscopic or radiographic guidance. Neither parenteral nor nasojejunal feeding stimulates pancreatic secretion, and both are safe in acute pancreatitis.
Severe acute pancreatitis is an intensely catabolic state characterized by increased energy expenditure, protein breakdown, and substrate utilization. Patients may not be able to resume an oral diet for weeks or even months, particularly if local complications develop. Early nutritional support has been shown to improve outcomes in severe acute pancreatitis.22 Therefore, nutritional support should be started as soon as possible in severe acute pancreatitis based on initial clinical and radiographic indicators of severity, optimally within the first 2 or 3 days.
Enteral nutrition is preferred to parenteral nutrition in pancreatitis: it is less expensive and does not pose a risk of catheter-related infection or thrombosis or hepatic complications. Also, there is experimental evidence that enteral nutrition may preserve the gut barrier, decreasing mucosal permeability and bacterial translocation.
A number of small randomized trials compared enteral and parenteral nutrition in acute pancreatitis, but they yielded mixed results. A meta-analysis of six trials showed a lower rate of infectious complications with enteral than with parenteral nutrition. 23 However, no significant difference was found in the rates of death or noninfectious complications.
Recommendation: Enteral feeding, when possible
Nutritional support is unnecessary in most cases of mild acute pancreatitis. Pancreatic inflammation typically resolves within a few days, allowing patients to resume eating. Occasionally, patients in whom pain resolves slowly and who fast for more than 5 to 7 days need nutritional support to prevent proteincalorie malnutrition.
The ACG guidelines1 and most others suggest that, whenever possible, enteral rather than parenteral feeding should be given to those who require nutritional support. The level of evidence is II (“strong evidence from at least one published properly designed randomized controlled trial of appropriate size and in an appropriate clinical setting”).
However, not all physicians recognize the benefit of enteral feeding. In a cohort of German gastroenterologists, only 73% favored enteral over parenteral feeding in acute pancreatitis.7
COMPUTED TOMOGRAPHY
Problem: CT is not done in many patients with severe acute pancreatitis, or it is done too soon during the admission.
Dual-phase, contrast-enhanced, pancreatic-protocol CT provides a sensitive structural evaluation of the pancreas and is useful to diagnose necrotizing pancreatitis. Pancreatic necrosis is correlated with a severe clinical course, the development of single or multiorgan dysfunction, and death.
Recommendation: CT in severe cases
Not every patient with acute pancreatitis needs to undergo CT. Most mild cases do not require routine CT, since necrosis and other local complications are infrequent in this group.
Also, CT is often ordered too soon during the hospitalization. Indicators of severity on CT are not usually evident until 2 to 3 days after admission.25 CT should be considered about 3 days after the onset of symptoms rather than immediately upon admission.
On the other hand, CT at the time of admission may be warranted to rule out other life-threatening causes of abdominal pain and hyperamylasemia (eg, bowel obstruction, viscus perforation). CT may also be useful in the late phase of acute pancreatitis (weeks after admission) to diagnose or monitor complications (eg, pseudocysts, abscesses, splenic vein thrombosis, splenic artery pseudoaneurysms). Magnetic resonance imaging with gadolinium contrast is a reasonable alternative to CT for detecting pancreatic necrosis and other local complications.
In patients who have severe acute pancreatitis and compromised renal function (serum creatinine > 1.5 mg/dL), CT can be performed without contrast to assess severity based on a limited Balthazar score (ie, without a necrosis score). Studies in rats suggest that iodinated contrast may decrease pancreatic microcirculation and worsen or precipitate necrosis,26 although published human studies do not support this contention.27,28
Guidelines uniformly recommend CT for patients with severe acute pancreatitis (the ACG guideline gives it a level of evidence of III), but this recommendation is not always followed. A study from Australia showed that CT was done in only 27% to 67% of patients with severe acute pancreatitis.5 In a British study, only 8 of 46 patients with clinically predicted severe pancreatitis underwent CT within the first 10 days of admission.4
SUSPECTED INFECTED NECROSIS
Problem: Fine-needle aspiration is not done in many cases of suspected infected necrosis.
Approximately one-third of patients with necrotizing pancreatitis develop infected necrosis. The death rate for patients with infected pancreatic necrosis is high—30%, compared with 12% in those with sterile necrosis.1 Differentiating sterile and infected necrosis is therefore essential.
Clinical signs such as fever are poor predictors of infection. Signs of SIRS can be present in both sterile and infected necrotizing pancreatitis.
Recommendation: Fine-needle aspiration of necrosis
For the reasons given above, the findings of necrosis on CT and persistent SIRS should prompt consideration of fine-needle aspiration with Gram stain and culture to differentiate sterile and infected necrosis (ACG guideline, level of evidence III).1 If infection is confirmed, surgical debridement should be strongly considered. Other less-invasive approaches such as endoscopic debridement can be used in selected cases.
Fine-needle aspiration of necrosis is too often neglected. In a cohort of German surgeons, only 55% complied with International Association of Pancreatology recommendations to perform biopsy to differentiate sterile from infected necrosis in patients with signs of sepsis.29
BROAD-SPECTRUM ANTIBIOTICS
Problem: Broad-spectrum antibiotics are often used inappropriately in patients with mild acute pancreatitis and in patients with sterile necrotizing pancreatitis who are clinically stable and have no signs of sepsis.
Antibiotics are not indicated in mild acute pancreatitis. A limited course of antibiotics is typically indicated in severe cases with suspected or proven infected necrosis (in conjunction with surgical necrosectomy). However, the use of antibiotics in sterile necrosis has been very controversial.
At least six small, nonblinded, randomized trials have evaluated the benefit of giving antibiotics prophylactically for presumed sterile necrosis. A recent Cochrane analysis of five of these trials (294 patients) suggested that patients who got antibiotics had a lower risk of death (odds ratio 0.37, 95% confidence interval [CI] 0.17–0.83) but no difference in the rates of pancreatic infection or surgery.30 These paradoxical results suggest that antibiotics may prevent death by preventing nonpancreatic infections (eg, pneumonia, line infections) rather than by preventing infection of necrotic pancreatic tissue. The five trials in the meta-analysis are limited by significant methodologic heterogeneity and by lack of double-blinding.
In spite of the overall lower death rate observed in the meta-analysis, the prophylactic use of antibiotics in sterile necrosis remains controversial. One concern is that patients given long prophylactic courses of antibiotics may develop resistant bacterial or fungal infections. However, the Cochrane and other meta-analyses have not shown a higher rate of fungal infections in those given antibiotics.31
Recommendation: No routine antibiotics for mild cases
The AGA guidelines recommend against routinely giving antibiotics in mild acute pancreatitis and do not provide strict recommendations for prophylactic antibiotic use in necrotizing acute pancreatitis.2 The guidelines state that antibiotics can be used “on demand” based on clinical signs of infection (eg, high fevers, rising leukocytosis, hypotension) or worsening organ failure.
If a purely prophylactic strategy is used, only patients at high risk of developing infection (eg, those with necrosis in more than 30% of the pancreas) should receive antibiotics. Antibiotics with high tissue-penetration should be used, such as imipenem-cilastin (Primaxin IV) or ciprofloxacin (Cipro) plus metronidazole (Flagyl).
Adherence to these guidelines is not optimal. For example, in an Italian multicenter study, 9% of patients with mild acute pancreatitis were treated with antibiotics.19 Moreover, many patients with proven infected necrosis received antibiotics that do not penetrate the pancreatic tissue very well.
ERCP IN SEVERE BILIARY ACUTE PANCREATITIS
Problem: Endoscopic retrograde cholangiopancreatography (ERCP) often is performed inappropriately in mild biliary acute pancreatitis or is not performed urgently in severe cases.
In most cases of mild biliary pancreatitis, the stones pass spontaneously, as verified by cholangiography done during laparoscopic cholecystectomy. Ongoing ampullary obstruction by impacted biliary stones can perpetuate pancreatic inflammation and delay recovery.
Two early randomized trials showed a benefit from early ERCP (within 72 hours) with sphincterotomy and stone extraction, primarily in those with severe biliary acute pancreatitis or ascending cholangitis,32,33 but a third trial failed to reveal a benefit.34 A Cochrane metaanalysis of these three trials failed to show a lower death rate with ERCP in mild or severe biliary pancreatitis.35 However, early ERCP did prevent complications in severe biliary pancreatitis (odds ratio 0.27, 95% CI 0.14–0.53).
Later, a fourth randomized trial was restricted to patients with suspected biliary pancreatitis, evidence of biliary obstruction, and no signs of cholangitis36: 103 patients were randomized to undergo either ERCP within 72 hours or conservative management. No difference was observed in rates of death or organ failure or in the CT severity index.
Recommendation: ER CP for suspected retained stones
ERCP has a limited role in patients with biliary pancreatitis, being used to clear retained bile duct stones or to relieve ongoing biliary obstruction.
The decision to perform ERCP before surgery should be based on how strongly one suspects retained stones. ERCP is most appropriate if the suspicion of retained stones and the likelihood of therapeutic intervention are high (eg, if the serum bilirubin and alkaline phosphatase levels are rising and ultrasonography shows a dilated bile duct). If there is moderate suspicion, a safer and less-invasive imaging study such as magnetic resonance cholangiopancreatography (MRCP) or endoscopic ultrasonography can be done to screen for bile duct stones before proceeding to ERCP.
The ACG guidelines suggest urgent ERCP (preferably within 24 hours) for those with severe biliary pancreatitis complicated by organ failure or those with suspicion of cholangitis. The level of evidence is I, ie, “strong evidence from at least one published systematic review of multiple well-designed randomized controlled trials.”1
Elective ERCP is recommended for those who are poor surgical candidates. ERCP is also recommended for those with rising liver enzyme values or imaging findings suggesting a retained common bile duct stone (including intraoperative cholangiography). Endoscopic ultrasonography or MRCP is recommended for those with slow clinical resolution, who are pregnant, or in whom uncertainty exists regarding the biliary etiology of pancreatitis.
Compliance rates with these and similar guidelines are not adequate. In an audit of adherence to the British Society of Gastroenterology guidelines, early ERCP was performed in only 25% of patients with severe biliary acute pancreatitis.6
LAPAROSCOPIC CHOLECYSTECTOMY FOR MILD BILIARY PANCREATITIS
Problem: Laparoscopic cholecystectomy is not done at admission or within 2 weeks in many patients with mild biliary pancreatitis.
If the gallbladder is not removed, biliary pancreatitis may recur in up to 61% of patients within 6 weeks of hospital discharge.37 This is the basis for guideline recommendations for surgery (or a confirmation of a surgery date) prior to hospital discharge.
The International Association of Pancreatology recommends early cholecystectomy (preferably during the same hospitalization) for patients with mild gallstone-associated acute pancreatitis.38 In severe gallstone-associated acute pancreatitis, cholecystectomy should be delayed until there is sufficient resolution of the inflammatory response and clinical recovery. The AGA guidelines advocate cholecystectomy as soon as possible and in no case later than 4 weeks after discharge to prevent relapse. ERCP with biliary sphinc-terotomy may also protect against relapse in those who are not fit to undergo surgery.
Recommendations for definitive management of gallstones (laparoscopic cholecystectomy or ERCP, or both) are not always followed. For example, a British study showed 70% compliance with this recommendation.4 A similar compliance audit in Germany revealed that cholecystectomy was performed during the initial hospital stay in only 23% of cases.7 In a New Zealand study, a regular compliance audit with feedback to surgeons resulted in an increase in the early cholecystectomy rate from 54% to 80%.8
- Banks PA, Freeman ML; Practice Parameters Committee of the American College of Gastroenterology. Practice guidelines in acute pancreatitis. Am J Gastroenterol 2006; 101:2379–2400.
- Forsmark CE, Baillie J; AGA Institute Clinical Practice and Economics Committee. AGA Institute technical review on acute pancreatitis. Gastroenterology 2007; 132:2022–2044.
- United Kingdom guidelines for the management of acute pancreatitis. British Society of Gastroenterology. Gut 1998; 42(suppl 2):S1–S13.
- Norton SA, Cheruvu CV, Collins J, Dix FP, Eyre-Brook IA. An assessment of clinical guidelines for the management of acute pancreatitis. Ann R Coll Surg Engl 2001; 83:399–405.
- Chiang DT, Anozie A, Fleming WR, Kiroff GK. Comparative study on acute pancreatitis management. ANZ J Surg 2004; 74:218–221.
- Barnard J, Siriwardena AK. Variations in implementation of current national guidelines for the treatment of acute pancreatitis: implications for acute surgical service provision. Ann R Coll Surg Engl 2002; 84:79–81.
- Lankisch PG, Weber-Dany B, Lerch MM. Clinical perspectives in pancreatology: compliance with acute pancreatitis in Germany [letter]. Pancreatology 2005; 5:591–593.
- Connor SJ, Lienert AR, Brown LA, Bagshaw PF. Closing the audit loop is necessary to achieve compliance with evidence-based guidelines in the management of acute pancreatitis. N Z Med J 2008; 121:19–25.
- Mofidi R, Duff MD, Wigmore SJ, Madhavan KK, Garden OJ, Parks RW. Association between early systemic inflammatory response, severity of multiorgan dysfunction, and death in acute pancreatitis. Br J Surg 2006; 93:738–744.
- Brown A, Orav J, Banks PA. Hemoconcentration is an early marker for organ failure and necrotizing pancreatitis. Pancreas 2000; 20:367–372.
- Lankisch PG, Mahlke R, Blum T, et al. Hemoconcentration: an early marker of severe and/or necrotizing pancreatitis? A critical appraisal. Am J Gastroenterol 2001; 96:2081–2085.
- Singh VK, Wu BU, Bollen TL, et al. A prospective evaluation of the bedside index for severity in acute pancreatitis score in assessing mortality and intermediate markers of severity in acute pancreatitis. Am J Gastroenterol 2009; 104:966–971.
- Ranson JH, Rifkind KM, Roses DF, Fink SD, Eng K, Spencer FC. Prognostic signs and the role of operative management in acute pancreatitis. Surg Gynecol Obstet 1974; 139:69–81.
- Larvin M. Assessment of clinical severity and prognosis. In:Beger HG, Warshaw AL, Buchler MW, et al, editors. The Pancreas. Blackwell Science: New York, 1998:489–502.
- Wu BU, Johannes RS, Sun X, Tabak Y, Conwell DL, Banks PA. The early prediction of mortality in acute pancreatitis: a large population-based study. Gut 2008; 57:1698–1703.
- Bradley EL. A clinically based classification system for acute pancreatitis. Summary of the International Symposium on Acute Pancreatitis, Atlanta, GA, September 11 through 13, 1992. Arch Surg 1993, 128:586–590.
- Forgacs B, Eible G, Faulhaber J, Kahrau S, Buhr H, Foitzik T. Effect of fluid resuscitation with and without endothelin A receptor blockade on hemoconcentration and organ function in experimental pancreatitis. Eur Surg Res 2000; 32:162–168.
- Brown A, Baillargeon JD, Hughes MD, Banks PA. Can fluid resuscitation prevent pancreatic necrosis in severe acute pancreatitis? Pancreatology 2002; 2:104–107.
- Pandol SJ, Saluja AK, Imrie CW, Banks PA. Acute pancreatitis: bench to the bedside. Gastroenterology 2007; 132:1127–1151.
- Pezzilli R, Uomo G, Gabbrielli A, et al; ProInf-AISP Study Group. A prospective multicenter survey on the treatment of acute pancreatitis in Italy. Dig Liver Dis 2007; 39:838–846.
- Gardner TB, Vege SS, Pearson RK, Chari ST. Fluid resuscitation in acute pancreatitis. Clin Gastroenterol Hepatol 2008; 6:1070–1076.
- Petrov MS, Pylypchuk RD, Emelyanov NV. Systematic review: nutritional support in acute pancreatitis. Aliment Pharmacol Ther 2008; 28:704–712.
- Marik PE, Zaloga GP. Meta-analysis of parenteral nutrition versus enteral nutrition in patients with acute pancreatitis. BMJ 2004; 328:1407.
- Balthazar EJ, Robinson DL, Megibow AJ, Ranson JH. Acute pancreatitis: value of CT in establishing prognosis. Radiology 1990; 174:331–336.
- Balthazar EJ. Acute pancreatitis: assessment of severity with clinical and CT evaluation. Radiology 2002; 223:603–613.
- Foitzik T, Bassi DG, Schmidt J, et al. Intravenous contrast medium accentuates the severity of acute necrotizing pancreatitis in the rat. Gastroenterology 1994; 106:207–214.
- Carmona-Sanchez R, Uscanga L, Bezaury-Rivas P, Robles-Díaz G, Suazo-Barahona J, Vargas-Vorácková F. Potential harmful effect of iodinated intravenous contrast medium on the clinical course of mild acute pancreatitis. Arch Surg 2000; 135:1280–1284.
- Uhl W, Roggo A, Kirschstein T, et al. Influence of contrast-enhanced computed tomography on couse and outcome in patients with acute pancreatitis. Pancreas 2002; 24:191–197.
- Foitzik T, Klar E. Non-compliance with guidelines for the management of severe acute pancreatitis among German surgeons. Pancreatology 2007; 7:80–85.
- Villatoro E, Bassi C, Larvin M. Antibiotic therapy for prophylaxis against infection of pancreatic necrosis in acute pancreatitis. Cochrane Database Syst Rev 2006;CD002941.
- Heinrich S, Schafer M, Rousson V, Clavien PA. Evidence-based treatment of acute pancreatitis: a look at established paradigms. Ann Surg 2006; 243:154–168.
- Neoptolemos JP, Carr-Locke DL, London NJ, Bailey IA, James D, Fossard DP. Controlled trial of urgent endoscopic retrograde cholangiopancreatography and endoscopic sphincterotomy versus conservative treatment for acute pancreatitis due to gallstones. Lancet 1988; 2:979–983.
- Fan ST, Lai EC, Mok FP, Lo CM, Zheng SS, Wong J. Early treatment of acute biliary pancreatitis by endoscopic papillotomy. N Engl J Med 1993; 328:228–232.
- Folsch UR, Nitsche R, Ludtke R, Hilgers RA, Creutzfeldt W. Early ERCP and papillotomy compared with conservative treatment for acute biliary pancreatitis. The German Study Group on Acute Biliary Pancreatitis. N Engl J Med 1997; 336:237–242.
- Ayub K, Imada R, Slavin J. Endoscopic retrograde cholangiopancreatography in gallstone associated pancreatitis. Cochrane Database Syst Rev 2004;CD003630
- Oria A, Cimmino D, Ocampo C, et al. Early endoscopic intervention versus early conservative management in patients with acute gallstone pancreatitis and biliopancreatic obstruction. A randomized clinical trial. Ann Surg 2007; 245:10–17.
- Frei GJ, Frei VT, Thirlby RC, McClelland RN. Biliary pancreatitis: clinical presentation and surgical management. Am J Surg 1986; 151:170–175.
- Uhl W, Warshaw A, Imrie C, et al; International Association of Pancreatology. IAP guidelines on the surgical management of acute pancreatitis. Pancreatology 2002; 2:565–573.
Several major gastroenterological and surgical societies have issued guidelines on how to manage acute pancreatitis, based on evidence from high-quality randomized trials and nonrandomized studies as well as on expert opinion.1–3 Information is limited on how well physicians in the United States comply with these guidelines, but compliance is suboptimal in other developed countries, according to several studies,4–8 and we suspect that many US physicians are not following the guidelines either.
Acute pancreatitis is a frequent inpatient diagnosis that internists, gastroenterologists, and surgeons all confront. The most common causes are gallstones and heavy alcohol intake. Its management is typically straightforward: intravenous fluids, analgesia, and nothing by mouth. However, treatment of severe cases can be quite complex, particularly if multiple organ systems are involved or if there are local complications.
The primary aim of this article is to raise awareness of recognized deviations from established recommendations that may lead to adverse patient outcomes.
MEASURING ENZYME LEVELS DAILY ADDS COST BUT LITTLE BENEFIT
Problem: Serum amylase and lipase levels are often needlessly measured every day.
Measuring the serum amylase and lipase levels is useful in diagnosing acute pancreatitis, which requires two of the following three features1:
- Characteristic abdominal pain
- Levels of serum amylase or serum lipase, or both, that are three or more times the upper limit of normal
- Findings of acute pancreatitis on computed tomography (CT).
However, the magnitude or duration of the serum enzyme elevation does not correlate with the severity of the attack. Further, we have noticed that physicians at our hospital often order daily serum amylase and lipase levels in patients admitted with acute pancreatitis.
The American College of Gastroenterology (ACG) guidelines1 state that daily monitoring of amylase and lipase has limited value in managing acute pancreatitis. Rechecking these concentrations may be reasonable if pain fails to resolve or worsens during a prolonged hospitalization, as this may suggest a recurrent attack of acute pancreatitis or a developing pseudocyst. But in most cases of acute pancreatitis, daily serum enzyme measurements add cost but little benefit.
REGULAR ASSESSMENT IS IMPORTANT
Problem: Often, severity assessments are not performed regularly or acted on.
Most cases of acute pancreatitis are mild, with rapid recovery and excellent prognosis. However, 15% to 20% are severe and may result in a prolonged hospitalization, systemic inflammatory response syndrome (SIRS), multiorgan system failure, and death.
In severe acute pancreatitis, as pancreatic enzymes and inflammatory cytokines damage the blood vessels, a vast amount of fluid leaks out into the interstitial (“third”) space. This fluid extravasation leads to decreased effective circulating volume, local pancreatic necrosis, hemodynamic instability, and end-organ failure.
It is important to recognize severe acute pancreatitis early because the patient needs to be transferred to a step-down unit or intensive care unit to receive optimal fluid resuscitation and supportive care for organ dysfunction. After 48 to 72 hours, a prediction of severe acute pancreatitis should also prompt the physician to order CT to detect pancreatic necrosis, and also to initiate nutritional support.
Assessment of severity begins in the emergency room or on admission to the hospital. Older age, obesity, organ failure, and pulmonary infiltrates or pleural effusions are initial indicators of poor prognosis. Signs of SIRS (high or low core body temperature, tachycardia, tachypnea, low or high peripheral white blood cell count) or organ failure (eg, elevated serum creatinine) are present on admission in 21% of patients with acute pancreatitis.9
Hemoconcentration is a marker of decreased effective circulating volume in severe acute pancreatitis. A hematocrit higher than 44% at admission or that rises in the first 24 to 48 hours of admission predicts necrosis.10,11 However, a more robust marker of organ failure may be the blood urea nitrogen level.12
Clinical scoring systems
Several clinical scoring systems have been studied for assessing severity.
Limitations of the Ranson score are that it can only be completed after 48 hours, all the data points are not always obtained, and it cannot be repeated on a daily basis. Owing to these limitations and its less-than-optimal predictive value, the Ranson score has fallen into disuse.
The APACHE II (Acute Physiology and Chronic Health Evaluation II) score is more versatile. It is based on multiple clinical and laboratory values, and it correlates very well with the risk of death in acute pancreatitis. Death rates are less than 4% when the APACHE II score is less than 8, and 11% to 18% when it is 8 or higher.1 The trajectory of the APACHE II score in the first 48 hours is also an accurate prognostic indicator.
Previous limitations of the APACHE II score were that it was complicated and timeconsuming to calculate and required arterial blood gas measurements. Easy-to-use online calculators are now available (eg, www.globalrph.com/apacheii.htm), and the venous bicarbonate level and the oxygen saturation can be substituted for the arterial pH and oxygen partial pressure.
BISAP, a new five-point scoring system,15 was recently prospectively validated.12 “BISAP” is an acronym for the five markers it is based on, each of which has been shown to predict severe illness in acute pancreatitis:
- Blood urea nitrogen level > 25 mg/dL
- Impaired mental status
- SIRS
- Age > 60 years
- Pleural effusion.
The presence of three or more of these factors correlates with higher risk of death, organ failure, and pancreatic necrosis.12
Compared with APACHE II, BISAP has similar accuracy and is easier to calculate. Also, BISAP was specifically developed for acute pancreatitis, whereas APACHE II is a generic score for all critically ill patients.
The Atlanta criteria16 define severe acute pancreatitis as one or more of the following:
- A Ranson score of 3 or higher during the first 48 hours
- An APACHE II score of 8 or higher at any time
- Failure of one or more organs
- One or more local complications (eg, necrosis, pseudocysts, abscesses).
Recommendation: Assess severity at least daily
A severity assessment should be performed at admission and at least every day thereafter. Clinical guidelines recognize the importance of severity assessment but vary in their specific recommendations.
The ACG advises calculating the APACHE II score within 3 days of admission and measuring the hematocrit at admission, at 12 hours, and at 24 hours. The level of evidence is III, ie, “from published well-designed trials without randomization, single group prepost, cohort, time series, or matched case controlled studies”.1
The American Gastroenterological Association (AGA) provides a more generalized recommendation, that “clinical judgment” should take into account the presence of risk factors (eg, age, obesity), presence or absence of SIRS, routine laboratory values (eg, hematocrit, serum creatinine), and APACHE II score when assessing severity and making decisions.2
In a German survey, only 32% of gastroenterologists used the APACHE II score for assessing risk in acute pancreatitis, in spite of national guidelines emphasizing its importance.7 Also, not all patients with severe acute pancreatitis are transferred to a step-down unit or intensive care unit as recommended. In a British study,4 only 8 (17%) of 46 patients with predicted severe acute pancreatitis were transferred, and 8 of the 38 patients who were not transferred died.
FLUID MUST BE AGGRESSIVELY REPLACED AND MONITORED
Problem: Often, not enough fluid is replaced, or fluid status is not adequately monitored.
Fluid must be aggressively replaced to balance the massive third-space fluid losses that occur in the early inflammatory phase of acute pancreatitis. Intravascular volume depletion can develop rapidly and result in tachycardia, hypotension, and renal failure. It may also impair the blood flow to the pancreas and worsen necrosis.
Animal studies show that aggressive fluid replacement supports the pancreatic microcirculation and prevents necrosis.17 It may also support the intestinal microcirculation and gut barrier, preventing bacterial translocation.
In humans, no controlled trials have been done to test the efficacy of aggressive fluid resuscitation in acute pancreatitis. However, the notion that intravascular fluid loss contributes to poor outcomes is inferred from human studies showing more necrosis and deaths in patients with hemoconcentration. In one study, patients who received inadequate fluid replacement (evidenced by a rise in hematocrit at 24 hours) were more likely to develop necrotizing pancreatitis.18
Recommendation: Early, aggressive fluid replacement
Experts have suggested initially infusing 500 to 1,000 mL of fluid per hour in those who are volume-depleted, initially infusing 250 to 350 mL per hour in those who are not volumedepleted, and adjusting the fluid rate every 1 to 4 hours on the basis of clinical variables.19 The sufficiency of fluid replacement should be carefully monitored by vital signs, urine output, and serum hematocrit.
On the other hand, overly aggressive fluid resuscitation can be detrimental in patients at risk of volume overload or pulmonary edema. Fluid replacement should be tempered in elderly patients and those with cardiac or renal comorbidities, and may require monitoring of central venous pressure.
The ACG and AGA guidelines both recognize the need for early aggressive volume replacement in acute pancreatitis (level of evidence III), but they do not specify the exact amounts and rates. Young and healthy patients should receive a rapid bolus of isotonic saline or Ringer’s lactate solution followed by an infusion at a high initial maintenance rate.
Few studies have been done to assess physicians’ compliance with recommendations for aggressive volume replacement. In an Italian multicenter study, patients with mild or severe acute pancreatitis received an average of only 2.5 L of fluid per day (about 100 mL/hour).20 Gardner et al21 recently summarized the available evidence for fluid support in acute pancreatitis.
NUTRITIONAL SUPPORT
Problem: In many severe cases, enteral or parenteral feeding is not started soon enough.
Nutritional support entails enteral or parenteral feeding when an oral diet is contraindicated. Enteral feeding is usually via a nasojejunal tube, which may need to be placed under endoscopic or radiographic guidance. Neither parenteral nor nasojejunal feeding stimulates pancreatic secretion, and both are safe in acute pancreatitis.
Severe acute pancreatitis is an intensely catabolic state characterized by increased energy expenditure, protein breakdown, and substrate utilization. Patients may not be able to resume an oral diet for weeks or even months, particularly if local complications develop. Early nutritional support has been shown to improve outcomes in severe acute pancreatitis.22 Therefore, nutritional support should be started as soon as possible in severe acute pancreatitis based on initial clinical and radiographic indicators of severity, optimally within the first 2 or 3 days.
Enteral nutrition is preferred to parenteral nutrition in pancreatitis: it is less expensive and does not pose a risk of catheter-related infection or thrombosis or hepatic complications. Also, there is experimental evidence that enteral nutrition may preserve the gut barrier, decreasing mucosal permeability and bacterial translocation.
A number of small randomized trials compared enteral and parenteral nutrition in acute pancreatitis, but they yielded mixed results. A meta-analysis of six trials showed a lower rate of infectious complications with enteral than with parenteral nutrition. 23 However, no significant difference was found in the rates of death or noninfectious complications.
Recommendation: Enteral feeding, when possible
Nutritional support is unnecessary in most cases of mild acute pancreatitis. Pancreatic inflammation typically resolves within a few days, allowing patients to resume eating. Occasionally, patients in whom pain resolves slowly and who fast for more than 5 to 7 days need nutritional support to prevent proteincalorie malnutrition.
The ACG guidelines1 and most others suggest that, whenever possible, enteral rather than parenteral feeding should be given to those who require nutritional support. The level of evidence is II (“strong evidence from at least one published properly designed randomized controlled trial of appropriate size and in an appropriate clinical setting”).
However, not all physicians recognize the benefit of enteral feeding. In a cohort of German gastroenterologists, only 73% favored enteral over parenteral feeding in acute pancreatitis.7
COMPUTED TOMOGRAPHY
Problem: CT is not done in many patients with severe acute pancreatitis, or it is done too soon during the admission.
Dual-phase, contrast-enhanced, pancreatic-protocol CT provides a sensitive structural evaluation of the pancreas and is useful to diagnose necrotizing pancreatitis. Pancreatic necrosis is correlated with a severe clinical course, the development of single or multiorgan dysfunction, and death.
Recommendation: CT in severe cases
Not every patient with acute pancreatitis needs to undergo CT. Most mild cases do not require routine CT, since necrosis and other local complications are infrequent in this group.
Also, CT is often ordered too soon during the hospitalization. Indicators of severity on CT are not usually evident until 2 to 3 days after admission.25 CT should be considered about 3 days after the onset of symptoms rather than immediately upon admission.
On the other hand, CT at the time of admission may be warranted to rule out other life-threatening causes of abdominal pain and hyperamylasemia (eg, bowel obstruction, viscus perforation). CT may also be useful in the late phase of acute pancreatitis (weeks after admission) to diagnose or monitor complications (eg, pseudocysts, abscesses, splenic vein thrombosis, splenic artery pseudoaneurysms). Magnetic resonance imaging with gadolinium contrast is a reasonable alternative to CT for detecting pancreatic necrosis and other local complications.
In patients who have severe acute pancreatitis and compromised renal function (serum creatinine > 1.5 mg/dL), CT can be performed without contrast to assess severity based on a limited Balthazar score (ie, without a necrosis score). Studies in rats suggest that iodinated contrast may decrease pancreatic microcirculation and worsen or precipitate necrosis,26 although published human studies do not support this contention.27,28
Guidelines uniformly recommend CT for patients with severe acute pancreatitis (the ACG guideline gives it a level of evidence of III), but this recommendation is not always followed. A study from Australia showed that CT was done in only 27% to 67% of patients with severe acute pancreatitis.5 In a British study, only 8 of 46 patients with clinically predicted severe pancreatitis underwent CT within the first 10 days of admission.4
SUSPECTED INFECTED NECROSIS
Problem: Fine-needle aspiration is not done in many cases of suspected infected necrosis.
Approximately one-third of patients with necrotizing pancreatitis develop infected necrosis. The death rate for patients with infected pancreatic necrosis is high—30%, compared with 12% in those with sterile necrosis.1 Differentiating sterile and infected necrosis is therefore essential.
Clinical signs such as fever are poor predictors of infection. Signs of SIRS can be present in both sterile and infected necrotizing pancreatitis.
Recommendation: Fine-needle aspiration of necrosis
For the reasons given above, the findings of necrosis on CT and persistent SIRS should prompt consideration of fine-needle aspiration with Gram stain and culture to differentiate sterile and infected necrosis (ACG guideline, level of evidence III).1 If infection is confirmed, surgical debridement should be strongly considered. Other less-invasive approaches such as endoscopic debridement can be used in selected cases.
Fine-needle aspiration of necrosis is too often neglected. In a cohort of German surgeons, only 55% complied with International Association of Pancreatology recommendations to perform biopsy to differentiate sterile from infected necrosis in patients with signs of sepsis.29
BROAD-SPECTRUM ANTIBIOTICS
Problem: Broad-spectrum antibiotics are often used inappropriately in patients with mild acute pancreatitis and in patients with sterile necrotizing pancreatitis who are clinically stable and have no signs of sepsis.
Antibiotics are not indicated in mild acute pancreatitis. A limited course of antibiotics is typically indicated in severe cases with suspected or proven infected necrosis (in conjunction with surgical necrosectomy). However, the use of antibiotics in sterile necrosis has been very controversial.
At least six small, nonblinded, randomized trials have evaluated the benefit of giving antibiotics prophylactically for presumed sterile necrosis. A recent Cochrane analysis of five of these trials (294 patients) suggested that patients who got antibiotics had a lower risk of death (odds ratio 0.37, 95% confidence interval [CI] 0.17–0.83) but no difference in the rates of pancreatic infection or surgery.30 These paradoxical results suggest that antibiotics may prevent death by preventing nonpancreatic infections (eg, pneumonia, line infections) rather than by preventing infection of necrotic pancreatic tissue. The five trials in the meta-analysis are limited by significant methodologic heterogeneity and by lack of double-blinding.
In spite of the overall lower death rate observed in the meta-analysis, the prophylactic use of antibiotics in sterile necrosis remains controversial. One concern is that patients given long prophylactic courses of antibiotics may develop resistant bacterial or fungal infections. However, the Cochrane and other meta-analyses have not shown a higher rate of fungal infections in those given antibiotics.31
Recommendation: No routine antibiotics for mild cases
The AGA guidelines recommend against routinely giving antibiotics in mild acute pancreatitis and do not provide strict recommendations for prophylactic antibiotic use in necrotizing acute pancreatitis.2 The guidelines state that antibiotics can be used “on demand” based on clinical signs of infection (eg, high fevers, rising leukocytosis, hypotension) or worsening organ failure.
If a purely prophylactic strategy is used, only patients at high risk of developing infection (eg, those with necrosis in more than 30% of the pancreas) should receive antibiotics. Antibiotics with high tissue-penetration should be used, such as imipenem-cilastin (Primaxin IV) or ciprofloxacin (Cipro) plus metronidazole (Flagyl).
Adherence to these guidelines is not optimal. For example, in an Italian multicenter study, 9% of patients with mild acute pancreatitis were treated with antibiotics.19 Moreover, many patients with proven infected necrosis received antibiotics that do not penetrate the pancreatic tissue very well.
ERCP IN SEVERE BILIARY ACUTE PANCREATITIS
Problem: Endoscopic retrograde cholangiopancreatography (ERCP) often is performed inappropriately in mild biliary acute pancreatitis or is not performed urgently in severe cases.
In most cases of mild biliary pancreatitis, the stones pass spontaneously, as verified by cholangiography done during laparoscopic cholecystectomy. Ongoing ampullary obstruction by impacted biliary stones can perpetuate pancreatic inflammation and delay recovery.
Two early randomized trials showed a benefit from early ERCP (within 72 hours) with sphincterotomy and stone extraction, primarily in those with severe biliary acute pancreatitis or ascending cholangitis,32,33 but a third trial failed to reveal a benefit.34 A Cochrane metaanalysis of these three trials failed to show a lower death rate with ERCP in mild or severe biliary pancreatitis.35 However, early ERCP did prevent complications in severe biliary pancreatitis (odds ratio 0.27, 95% CI 0.14–0.53).
Later, a fourth randomized trial was restricted to patients with suspected biliary pancreatitis, evidence of biliary obstruction, and no signs of cholangitis36: 103 patients were randomized to undergo either ERCP within 72 hours or conservative management. No difference was observed in rates of death or organ failure or in the CT severity index.
Recommendation: ER CP for suspected retained stones
ERCP has a limited role in patients with biliary pancreatitis, being used to clear retained bile duct stones or to relieve ongoing biliary obstruction.
The decision to perform ERCP before surgery should be based on how strongly one suspects retained stones. ERCP is most appropriate if the suspicion of retained stones and the likelihood of therapeutic intervention are high (eg, if the serum bilirubin and alkaline phosphatase levels are rising and ultrasonography shows a dilated bile duct). If there is moderate suspicion, a safer and less-invasive imaging study such as magnetic resonance cholangiopancreatography (MRCP) or endoscopic ultrasonography can be done to screen for bile duct stones before proceeding to ERCP.
The ACG guidelines suggest urgent ERCP (preferably within 24 hours) for those with severe biliary pancreatitis complicated by organ failure or those with suspicion of cholangitis. The level of evidence is I, ie, “strong evidence from at least one published systematic review of multiple well-designed randomized controlled trials.”1
Elective ERCP is recommended for those who are poor surgical candidates. ERCP is also recommended for those with rising liver enzyme values or imaging findings suggesting a retained common bile duct stone (including intraoperative cholangiography). Endoscopic ultrasonography or MRCP is recommended for those with slow clinical resolution, who are pregnant, or in whom uncertainty exists regarding the biliary etiology of pancreatitis.
Compliance rates with these and similar guidelines are not adequate. In an audit of adherence to the British Society of Gastroenterology guidelines, early ERCP was performed in only 25% of patients with severe biliary acute pancreatitis.6
LAPAROSCOPIC CHOLECYSTECTOMY FOR MILD BILIARY PANCREATITIS
Problem: Laparoscopic cholecystectomy is not done at admission or within 2 weeks in many patients with mild biliary pancreatitis.
If the gallbladder is not removed, biliary pancreatitis may recur in up to 61% of patients within 6 weeks of hospital discharge.37 This is the basis for guideline recommendations for surgery (or a confirmation of a surgery date) prior to hospital discharge.
The International Association of Pancreatology recommends early cholecystectomy (preferably during the same hospitalization) for patients with mild gallstone-associated acute pancreatitis.38 In severe gallstone-associated acute pancreatitis, cholecystectomy should be delayed until there is sufficient resolution of the inflammatory response and clinical recovery. The AGA guidelines advocate cholecystectomy as soon as possible and in no case later than 4 weeks after discharge to prevent relapse. ERCP with biliary sphinc-terotomy may also protect against relapse in those who are not fit to undergo surgery.
Recommendations for definitive management of gallstones (laparoscopic cholecystectomy or ERCP, or both) are not always followed. For example, a British study showed 70% compliance with this recommendation.4 A similar compliance audit in Germany revealed that cholecystectomy was performed during the initial hospital stay in only 23% of cases.7 In a New Zealand study, a regular compliance audit with feedback to surgeons resulted in an increase in the early cholecystectomy rate from 54% to 80%.8
Several major gastroenterological and surgical societies have issued guidelines on how to manage acute pancreatitis, based on evidence from high-quality randomized trials and nonrandomized studies as well as on expert opinion.1–3 Information is limited on how well physicians in the United States comply with these guidelines, but compliance is suboptimal in other developed countries, according to several studies,4–8 and we suspect that many US physicians are not following the guidelines either.
Acute pancreatitis is a frequent inpatient diagnosis that internists, gastroenterologists, and surgeons all confront. The most common causes are gallstones and heavy alcohol intake. Its management is typically straightforward: intravenous fluids, analgesia, and nothing by mouth. However, treatment of severe cases can be quite complex, particularly if multiple organ systems are involved or if there are local complications.
The primary aim of this article is to raise awareness of recognized deviations from established recommendations that may lead to adverse patient outcomes.
MEASURING ENZYME LEVELS DAILY ADDS COST BUT LITTLE BENEFIT
Problem: Serum amylase and lipase levels are often needlessly measured every day.
Measuring the serum amylase and lipase levels is useful in diagnosing acute pancreatitis, which requires two of the following three features1:
- Characteristic abdominal pain
- Levels of serum amylase or serum lipase, or both, that are three or more times the upper limit of normal
- Findings of acute pancreatitis on computed tomography (CT).
However, the magnitude or duration of the serum enzyme elevation does not correlate with the severity of the attack. Further, we have noticed that physicians at our hospital often order daily serum amylase and lipase levels in patients admitted with acute pancreatitis.
The American College of Gastroenterology (ACG) guidelines1 state that daily monitoring of amylase and lipase has limited value in managing acute pancreatitis. Rechecking these concentrations may be reasonable if pain fails to resolve or worsens during a prolonged hospitalization, as this may suggest a recurrent attack of acute pancreatitis or a developing pseudocyst. But in most cases of acute pancreatitis, daily serum enzyme measurements add cost but little benefit.
REGULAR ASSESSMENT IS IMPORTANT
Problem: Often, severity assessments are not performed regularly or acted on.
Most cases of acute pancreatitis are mild, with rapid recovery and excellent prognosis. However, 15% to 20% are severe and may result in a prolonged hospitalization, systemic inflammatory response syndrome (SIRS), multiorgan system failure, and death.
In severe acute pancreatitis, as pancreatic enzymes and inflammatory cytokines damage the blood vessels, a vast amount of fluid leaks out into the interstitial (“third”) space. This fluid extravasation leads to decreased effective circulating volume, local pancreatic necrosis, hemodynamic instability, and end-organ failure.
It is important to recognize severe acute pancreatitis early because the patient needs to be transferred to a step-down unit or intensive care unit to receive optimal fluid resuscitation and supportive care for organ dysfunction. After 48 to 72 hours, a prediction of severe acute pancreatitis should also prompt the physician to order CT to detect pancreatic necrosis, and also to initiate nutritional support.
Assessment of severity begins in the emergency room or on admission to the hospital. Older age, obesity, organ failure, and pulmonary infiltrates or pleural effusions are initial indicators of poor prognosis. Signs of SIRS (high or low core body temperature, tachycardia, tachypnea, low or high peripheral white blood cell count) or organ failure (eg, elevated serum creatinine) are present on admission in 21% of patients with acute pancreatitis.9
Hemoconcentration is a marker of decreased effective circulating volume in severe acute pancreatitis. A hematocrit higher than 44% at admission or that rises in the first 24 to 48 hours of admission predicts necrosis.10,11 However, a more robust marker of organ failure may be the blood urea nitrogen level.12
Clinical scoring systems
Several clinical scoring systems have been studied for assessing severity.
Limitations of the Ranson score are that it can only be completed after 48 hours, all the data points are not always obtained, and it cannot be repeated on a daily basis. Owing to these limitations and its less-than-optimal predictive value, the Ranson score has fallen into disuse.
The APACHE II (Acute Physiology and Chronic Health Evaluation II) score is more versatile. It is based on multiple clinical and laboratory values, and it correlates very well with the risk of death in acute pancreatitis. Death rates are less than 4% when the APACHE II score is less than 8, and 11% to 18% when it is 8 or higher.1 The trajectory of the APACHE II score in the first 48 hours is also an accurate prognostic indicator.
Previous limitations of the APACHE II score were that it was complicated and timeconsuming to calculate and required arterial blood gas measurements. Easy-to-use online calculators are now available (eg, www.globalrph.com/apacheii.htm), and the venous bicarbonate level and the oxygen saturation can be substituted for the arterial pH and oxygen partial pressure.
BISAP, a new five-point scoring system,15 was recently prospectively validated.12 “BISAP” is an acronym for the five markers it is based on, each of which has been shown to predict severe illness in acute pancreatitis:
- Blood urea nitrogen level > 25 mg/dL
- Impaired mental status
- SIRS
- Age > 60 years
- Pleural effusion.
The presence of three or more of these factors correlates with higher risk of death, organ failure, and pancreatic necrosis.12
Compared with APACHE II, BISAP has similar accuracy and is easier to calculate. Also, BISAP was specifically developed for acute pancreatitis, whereas APACHE II is a generic score for all critically ill patients.
The Atlanta criteria16 define severe acute pancreatitis as one or more of the following:
- A Ranson score of 3 or higher during the first 48 hours
- An APACHE II score of 8 or higher at any time
- Failure of one or more organs
- One or more local complications (eg, necrosis, pseudocysts, abscesses).
Recommendation: Assess severity at least daily
A severity assessment should be performed at admission and at least every day thereafter. Clinical guidelines recognize the importance of severity assessment but vary in their specific recommendations.
The ACG advises calculating the APACHE II score within 3 days of admission and measuring the hematocrit at admission, at 12 hours, and at 24 hours. The level of evidence is III, ie, “from published well-designed trials without randomization, single group prepost, cohort, time series, or matched case controlled studies”.1
The American Gastroenterological Association (AGA) provides a more generalized recommendation, that “clinical judgment” should take into account the presence of risk factors (eg, age, obesity), presence or absence of SIRS, routine laboratory values (eg, hematocrit, serum creatinine), and APACHE II score when assessing severity and making decisions.2
In a German survey, only 32% of gastroenterologists used the APACHE II score for assessing risk in acute pancreatitis, in spite of national guidelines emphasizing its importance.7 Also, not all patients with severe acute pancreatitis are transferred to a step-down unit or intensive care unit as recommended. In a British study,4 only 8 (17%) of 46 patients with predicted severe acute pancreatitis were transferred, and 8 of the 38 patients who were not transferred died.
FLUID MUST BE AGGRESSIVELY REPLACED AND MONITORED
Problem: Often, not enough fluid is replaced, or fluid status is not adequately monitored.
Fluid must be aggressively replaced to balance the massive third-space fluid losses that occur in the early inflammatory phase of acute pancreatitis. Intravascular volume depletion can develop rapidly and result in tachycardia, hypotension, and renal failure. It may also impair the blood flow to the pancreas and worsen necrosis.
Animal studies show that aggressive fluid replacement supports the pancreatic microcirculation and prevents necrosis.17 It may also support the intestinal microcirculation and gut barrier, preventing bacterial translocation.
In humans, no controlled trials have been done to test the efficacy of aggressive fluid resuscitation in acute pancreatitis. However, the notion that intravascular fluid loss contributes to poor outcomes is inferred from human studies showing more necrosis and deaths in patients with hemoconcentration. In one study, patients who received inadequate fluid replacement (evidenced by a rise in hematocrit at 24 hours) were more likely to develop necrotizing pancreatitis.18
Recommendation: Early, aggressive fluid replacement
Experts have suggested initially infusing 500 to 1,000 mL of fluid per hour in those who are volume-depleted, initially infusing 250 to 350 mL per hour in those who are not volumedepleted, and adjusting the fluid rate every 1 to 4 hours on the basis of clinical variables.19 The sufficiency of fluid replacement should be carefully monitored by vital signs, urine output, and serum hematocrit.
On the other hand, overly aggressive fluid resuscitation can be detrimental in patients at risk of volume overload or pulmonary edema. Fluid replacement should be tempered in elderly patients and those with cardiac or renal comorbidities, and may require monitoring of central venous pressure.
The ACG and AGA guidelines both recognize the need for early aggressive volume replacement in acute pancreatitis (level of evidence III), but they do not specify the exact amounts and rates. Young and healthy patients should receive a rapid bolus of isotonic saline or Ringer’s lactate solution followed by an infusion at a high initial maintenance rate.
Few studies have been done to assess physicians’ compliance with recommendations for aggressive volume replacement. In an Italian multicenter study, patients with mild or severe acute pancreatitis received an average of only 2.5 L of fluid per day (about 100 mL/hour).20 Gardner et al21 recently summarized the available evidence for fluid support in acute pancreatitis.
NUTRITIONAL SUPPORT
Problem: In many severe cases, enteral or parenteral feeding is not started soon enough.
Nutritional support entails enteral or parenteral feeding when an oral diet is contraindicated. Enteral feeding is usually via a nasojejunal tube, which may need to be placed under endoscopic or radiographic guidance. Neither parenteral nor nasojejunal feeding stimulates pancreatic secretion, and both are safe in acute pancreatitis.
Severe acute pancreatitis is an intensely catabolic state characterized by increased energy expenditure, protein breakdown, and substrate utilization. Patients may not be able to resume an oral diet for weeks or even months, particularly if local complications develop. Early nutritional support has been shown to improve outcomes in severe acute pancreatitis.22 Therefore, nutritional support should be started as soon as possible in severe acute pancreatitis based on initial clinical and radiographic indicators of severity, optimally within the first 2 or 3 days.
Enteral nutrition is preferred to parenteral nutrition in pancreatitis: it is less expensive and does not pose a risk of catheter-related infection or thrombosis or hepatic complications. Also, there is experimental evidence that enteral nutrition may preserve the gut barrier, decreasing mucosal permeability and bacterial translocation.
A number of small randomized trials compared enteral and parenteral nutrition in acute pancreatitis, but they yielded mixed results. A meta-analysis of six trials showed a lower rate of infectious complications with enteral than with parenteral nutrition. 23 However, no significant difference was found in the rates of death or noninfectious complications.
Recommendation: Enteral feeding, when possible
Nutritional support is unnecessary in most cases of mild acute pancreatitis. Pancreatic inflammation typically resolves within a few days, allowing patients to resume eating. Occasionally, patients in whom pain resolves slowly and who fast for more than 5 to 7 days need nutritional support to prevent proteincalorie malnutrition.
The ACG guidelines1 and most others suggest that, whenever possible, enteral rather than parenteral feeding should be given to those who require nutritional support. The level of evidence is II (“strong evidence from at least one published properly designed randomized controlled trial of appropriate size and in an appropriate clinical setting”).
However, not all physicians recognize the benefit of enteral feeding. In a cohort of German gastroenterologists, only 73% favored enteral over parenteral feeding in acute pancreatitis.7
COMPUTED TOMOGRAPHY
Problem: CT is not done in many patients with severe acute pancreatitis, or it is done too soon during the admission.
Dual-phase, contrast-enhanced, pancreatic-protocol CT provides a sensitive structural evaluation of the pancreas and is useful to diagnose necrotizing pancreatitis. Pancreatic necrosis is correlated with a severe clinical course, the development of single or multiorgan dysfunction, and death.
Recommendation: CT in severe cases
Not every patient with acute pancreatitis needs to undergo CT. Most mild cases do not require routine CT, since necrosis and other local complications are infrequent in this group.
Also, CT is often ordered too soon during the hospitalization. Indicators of severity on CT are not usually evident until 2 to 3 days after admission.25 CT should be considered about 3 days after the onset of symptoms rather than immediately upon admission.
On the other hand, CT at the time of admission may be warranted to rule out other life-threatening causes of abdominal pain and hyperamylasemia (eg, bowel obstruction, viscus perforation). CT may also be useful in the late phase of acute pancreatitis (weeks after admission) to diagnose or monitor complications (eg, pseudocysts, abscesses, splenic vein thrombosis, splenic artery pseudoaneurysms). Magnetic resonance imaging with gadolinium contrast is a reasonable alternative to CT for detecting pancreatic necrosis and other local complications.
In patients who have severe acute pancreatitis and compromised renal function (serum creatinine > 1.5 mg/dL), CT can be performed without contrast to assess severity based on a limited Balthazar score (ie, without a necrosis score). Studies in rats suggest that iodinated contrast may decrease pancreatic microcirculation and worsen or precipitate necrosis,26 although published human studies do not support this contention.27,28
Guidelines uniformly recommend CT for patients with severe acute pancreatitis (the ACG guideline gives it a level of evidence of III), but this recommendation is not always followed. A study from Australia showed that CT was done in only 27% to 67% of patients with severe acute pancreatitis.5 In a British study, only 8 of 46 patients with clinically predicted severe pancreatitis underwent CT within the first 10 days of admission.4
SUSPECTED INFECTED NECROSIS
Problem: Fine-needle aspiration is not done in many cases of suspected infected necrosis.
Approximately one-third of patients with necrotizing pancreatitis develop infected necrosis. The death rate for patients with infected pancreatic necrosis is high—30%, compared with 12% in those with sterile necrosis.1 Differentiating sterile and infected necrosis is therefore essential.
Clinical signs such as fever are poor predictors of infection. Signs of SIRS can be present in both sterile and infected necrotizing pancreatitis.
Recommendation: Fine-needle aspiration of necrosis
For the reasons given above, the findings of necrosis on CT and persistent SIRS should prompt consideration of fine-needle aspiration with Gram stain and culture to differentiate sterile and infected necrosis (ACG guideline, level of evidence III).1 If infection is confirmed, surgical debridement should be strongly considered. Other less-invasive approaches such as endoscopic debridement can be used in selected cases.
Fine-needle aspiration of necrosis is too often neglected. In a cohort of German surgeons, only 55% complied with International Association of Pancreatology recommendations to perform biopsy to differentiate sterile from infected necrosis in patients with signs of sepsis.29
BROAD-SPECTRUM ANTIBIOTICS
Problem: Broad-spectrum antibiotics are often used inappropriately in patients with mild acute pancreatitis and in patients with sterile necrotizing pancreatitis who are clinically stable and have no signs of sepsis.
Antibiotics are not indicated in mild acute pancreatitis. A limited course of antibiotics is typically indicated in severe cases with suspected or proven infected necrosis (in conjunction with surgical necrosectomy). However, the use of antibiotics in sterile necrosis has been very controversial.
At least six small, nonblinded, randomized trials have evaluated the benefit of giving antibiotics prophylactically for presumed sterile necrosis. A recent Cochrane analysis of five of these trials (294 patients) suggested that patients who got antibiotics had a lower risk of death (odds ratio 0.37, 95% confidence interval [CI] 0.17–0.83) but no difference in the rates of pancreatic infection or surgery.30 These paradoxical results suggest that antibiotics may prevent death by preventing nonpancreatic infections (eg, pneumonia, line infections) rather than by preventing infection of necrotic pancreatic tissue. The five trials in the meta-analysis are limited by significant methodologic heterogeneity and by lack of double-blinding.
In spite of the overall lower death rate observed in the meta-analysis, the prophylactic use of antibiotics in sterile necrosis remains controversial. One concern is that patients given long prophylactic courses of antibiotics may develop resistant bacterial or fungal infections. However, the Cochrane and other meta-analyses have not shown a higher rate of fungal infections in those given antibiotics.31
Recommendation: No routine antibiotics for mild cases
The AGA guidelines recommend against routinely giving antibiotics in mild acute pancreatitis and do not provide strict recommendations for prophylactic antibiotic use in necrotizing acute pancreatitis.2 The guidelines state that antibiotics can be used “on demand” based on clinical signs of infection (eg, high fevers, rising leukocytosis, hypotension) or worsening organ failure.
If a purely prophylactic strategy is used, only patients at high risk of developing infection (eg, those with necrosis in more than 30% of the pancreas) should receive antibiotics. Antibiotics with high tissue-penetration should be used, such as imipenem-cilastin (Primaxin IV) or ciprofloxacin (Cipro) plus metronidazole (Flagyl).
Adherence to these guidelines is not optimal. For example, in an Italian multicenter study, 9% of patients with mild acute pancreatitis were treated with antibiotics.19 Moreover, many patients with proven infected necrosis received antibiotics that do not penetrate the pancreatic tissue very well.
ERCP IN SEVERE BILIARY ACUTE PANCREATITIS
Problem: Endoscopic retrograde cholangiopancreatography (ERCP) often is performed inappropriately in mild biliary acute pancreatitis or is not performed urgently in severe cases.
In most cases of mild biliary pancreatitis, the stones pass spontaneously, as verified by cholangiography done during laparoscopic cholecystectomy. Ongoing ampullary obstruction by impacted biliary stones can perpetuate pancreatic inflammation and delay recovery.
Two early randomized trials showed a benefit from early ERCP (within 72 hours) with sphincterotomy and stone extraction, primarily in those with severe biliary acute pancreatitis or ascending cholangitis,32,33 but a third trial failed to reveal a benefit.34 A Cochrane metaanalysis of these three trials failed to show a lower death rate with ERCP in mild or severe biliary pancreatitis.35 However, early ERCP did prevent complications in severe biliary pancreatitis (odds ratio 0.27, 95% CI 0.14–0.53).
Later, a fourth randomized trial was restricted to patients with suspected biliary pancreatitis, evidence of biliary obstruction, and no signs of cholangitis36: 103 patients were randomized to undergo either ERCP within 72 hours or conservative management. No difference was observed in rates of death or organ failure or in the CT severity index.
Recommendation: ER CP for suspected retained stones
ERCP has a limited role in patients with biliary pancreatitis, being used to clear retained bile duct stones or to relieve ongoing biliary obstruction.
The decision to perform ERCP before surgery should be based on how strongly one suspects retained stones. ERCP is most appropriate if the suspicion of retained stones and the likelihood of therapeutic intervention are high (eg, if the serum bilirubin and alkaline phosphatase levels are rising and ultrasonography shows a dilated bile duct). If there is moderate suspicion, a safer and less-invasive imaging study such as magnetic resonance cholangiopancreatography (MRCP) or endoscopic ultrasonography can be done to screen for bile duct stones before proceeding to ERCP.
The ACG guidelines suggest urgent ERCP (preferably within 24 hours) for those with severe biliary pancreatitis complicated by organ failure or those with suspicion of cholangitis. The level of evidence is I, ie, “strong evidence from at least one published systematic review of multiple well-designed randomized controlled trials.”1
Elective ERCP is recommended for those who are poor surgical candidates. ERCP is also recommended for those with rising liver enzyme values or imaging findings suggesting a retained common bile duct stone (including intraoperative cholangiography). Endoscopic ultrasonography or MRCP is recommended for those with slow clinical resolution, who are pregnant, or in whom uncertainty exists regarding the biliary etiology of pancreatitis.
Compliance rates with these and similar guidelines are not adequate. In an audit of adherence to the British Society of Gastroenterology guidelines, early ERCP was performed in only 25% of patients with severe biliary acute pancreatitis.6
LAPAROSCOPIC CHOLECYSTECTOMY FOR MILD BILIARY PANCREATITIS
Problem: Laparoscopic cholecystectomy is not done at admission or within 2 weeks in many patients with mild biliary pancreatitis.
If the gallbladder is not removed, biliary pancreatitis may recur in up to 61% of patients within 6 weeks of hospital discharge.37 This is the basis for guideline recommendations for surgery (or a confirmation of a surgery date) prior to hospital discharge.
The International Association of Pancreatology recommends early cholecystectomy (preferably during the same hospitalization) for patients with mild gallstone-associated acute pancreatitis.38 In severe gallstone-associated acute pancreatitis, cholecystectomy should be delayed until there is sufficient resolution of the inflammatory response and clinical recovery. The AGA guidelines advocate cholecystectomy as soon as possible and in no case later than 4 weeks after discharge to prevent relapse. ERCP with biliary sphinc-terotomy may also protect against relapse in those who are not fit to undergo surgery.
Recommendations for definitive management of gallstones (laparoscopic cholecystectomy or ERCP, or both) are not always followed. For example, a British study showed 70% compliance with this recommendation.4 A similar compliance audit in Germany revealed that cholecystectomy was performed during the initial hospital stay in only 23% of cases.7 In a New Zealand study, a regular compliance audit with feedback to surgeons resulted in an increase in the early cholecystectomy rate from 54% to 80%.8
- Banks PA, Freeman ML; Practice Parameters Committee of the American College of Gastroenterology. Practice guidelines in acute pancreatitis. Am J Gastroenterol 2006; 101:2379–2400.
- Forsmark CE, Baillie J; AGA Institute Clinical Practice and Economics Committee. AGA Institute technical review on acute pancreatitis. Gastroenterology 2007; 132:2022–2044.
- United Kingdom guidelines for the management of acute pancreatitis. British Society of Gastroenterology. Gut 1998; 42(suppl 2):S1–S13.
- Norton SA, Cheruvu CV, Collins J, Dix FP, Eyre-Brook IA. An assessment of clinical guidelines for the management of acute pancreatitis. Ann R Coll Surg Engl 2001; 83:399–405.
- Chiang DT, Anozie A, Fleming WR, Kiroff GK. Comparative study on acute pancreatitis management. ANZ J Surg 2004; 74:218–221.
- Barnard J, Siriwardena AK. Variations in implementation of current national guidelines for the treatment of acute pancreatitis: implications for acute surgical service provision. Ann R Coll Surg Engl 2002; 84:79–81.
- Lankisch PG, Weber-Dany B, Lerch MM. Clinical perspectives in pancreatology: compliance with acute pancreatitis in Germany [letter]. Pancreatology 2005; 5:591–593.
- Connor SJ, Lienert AR, Brown LA, Bagshaw PF. Closing the audit loop is necessary to achieve compliance with evidence-based guidelines in the management of acute pancreatitis. N Z Med J 2008; 121:19–25.
- Mofidi R, Duff MD, Wigmore SJ, Madhavan KK, Garden OJ, Parks RW. Association between early systemic inflammatory response, severity of multiorgan dysfunction, and death in acute pancreatitis. Br J Surg 2006; 93:738–744.
- Brown A, Orav J, Banks PA. Hemoconcentration is an early marker for organ failure and necrotizing pancreatitis. Pancreas 2000; 20:367–372.
- Lankisch PG, Mahlke R, Blum T, et al. Hemoconcentration: an early marker of severe and/or necrotizing pancreatitis? A critical appraisal. Am J Gastroenterol 2001; 96:2081–2085.
- Singh VK, Wu BU, Bollen TL, et al. A prospective evaluation of the bedside index for severity in acute pancreatitis score in assessing mortality and intermediate markers of severity in acute pancreatitis. Am J Gastroenterol 2009; 104:966–971.
- Ranson JH, Rifkind KM, Roses DF, Fink SD, Eng K, Spencer FC. Prognostic signs and the role of operative management in acute pancreatitis. Surg Gynecol Obstet 1974; 139:69–81.
- Larvin M. Assessment of clinical severity and prognosis. In:Beger HG, Warshaw AL, Buchler MW, et al, editors. The Pancreas. Blackwell Science: New York, 1998:489–502.
- Wu BU, Johannes RS, Sun X, Tabak Y, Conwell DL, Banks PA. The early prediction of mortality in acute pancreatitis: a large population-based study. Gut 2008; 57:1698–1703.
- Bradley EL. A clinically based classification system for acute pancreatitis. Summary of the International Symposium on Acute Pancreatitis, Atlanta, GA, September 11 through 13, 1992. Arch Surg 1993, 128:586–590.
- Forgacs B, Eible G, Faulhaber J, Kahrau S, Buhr H, Foitzik T. Effect of fluid resuscitation with and without endothelin A receptor blockade on hemoconcentration and organ function in experimental pancreatitis. Eur Surg Res 2000; 32:162–168.
- Brown A, Baillargeon JD, Hughes MD, Banks PA. Can fluid resuscitation prevent pancreatic necrosis in severe acute pancreatitis? Pancreatology 2002; 2:104–107.
- Pandol SJ, Saluja AK, Imrie CW, Banks PA. Acute pancreatitis: bench to the bedside. Gastroenterology 2007; 132:1127–1151.
- Pezzilli R, Uomo G, Gabbrielli A, et al; ProInf-AISP Study Group. A prospective multicenter survey on the treatment of acute pancreatitis in Italy. Dig Liver Dis 2007; 39:838–846.
- Gardner TB, Vege SS, Pearson RK, Chari ST. Fluid resuscitation in acute pancreatitis. Clin Gastroenterol Hepatol 2008; 6:1070–1076.
- Petrov MS, Pylypchuk RD, Emelyanov NV. Systematic review: nutritional support in acute pancreatitis. Aliment Pharmacol Ther 2008; 28:704–712.
- Marik PE, Zaloga GP. Meta-analysis of parenteral nutrition versus enteral nutrition in patients with acute pancreatitis. BMJ 2004; 328:1407.
- Balthazar EJ, Robinson DL, Megibow AJ, Ranson JH. Acute pancreatitis: value of CT in establishing prognosis. Radiology 1990; 174:331–336.
- Balthazar EJ. Acute pancreatitis: assessment of severity with clinical and CT evaluation. Radiology 2002; 223:603–613.
- Foitzik T, Bassi DG, Schmidt J, et al. Intravenous contrast medium accentuates the severity of acute necrotizing pancreatitis in the rat. Gastroenterology 1994; 106:207–214.
- Carmona-Sanchez R, Uscanga L, Bezaury-Rivas P, Robles-Díaz G, Suazo-Barahona J, Vargas-Vorácková F. Potential harmful effect of iodinated intravenous contrast medium on the clinical course of mild acute pancreatitis. Arch Surg 2000; 135:1280–1284.
- Uhl W, Roggo A, Kirschstein T, et al. Influence of contrast-enhanced computed tomography on couse and outcome in patients with acute pancreatitis. Pancreas 2002; 24:191–197.
- Foitzik T, Klar E. Non-compliance with guidelines for the management of severe acute pancreatitis among German surgeons. Pancreatology 2007; 7:80–85.
- Villatoro E, Bassi C, Larvin M. Antibiotic therapy for prophylaxis against infection of pancreatic necrosis in acute pancreatitis. Cochrane Database Syst Rev 2006;CD002941.
- Heinrich S, Schafer M, Rousson V, Clavien PA. Evidence-based treatment of acute pancreatitis: a look at established paradigms. Ann Surg 2006; 243:154–168.
- Neoptolemos JP, Carr-Locke DL, London NJ, Bailey IA, James D, Fossard DP. Controlled trial of urgent endoscopic retrograde cholangiopancreatography and endoscopic sphincterotomy versus conservative treatment for acute pancreatitis due to gallstones. Lancet 1988; 2:979–983.
- Fan ST, Lai EC, Mok FP, Lo CM, Zheng SS, Wong J. Early treatment of acute biliary pancreatitis by endoscopic papillotomy. N Engl J Med 1993; 328:228–232.
- Folsch UR, Nitsche R, Ludtke R, Hilgers RA, Creutzfeldt W. Early ERCP and papillotomy compared with conservative treatment for acute biliary pancreatitis. The German Study Group on Acute Biliary Pancreatitis. N Engl J Med 1997; 336:237–242.
- Ayub K, Imada R, Slavin J. Endoscopic retrograde cholangiopancreatography in gallstone associated pancreatitis. Cochrane Database Syst Rev 2004;CD003630
- Oria A, Cimmino D, Ocampo C, et al. Early endoscopic intervention versus early conservative management in patients with acute gallstone pancreatitis and biliopancreatic obstruction. A randomized clinical trial. Ann Surg 2007; 245:10–17.
- Frei GJ, Frei VT, Thirlby RC, McClelland RN. Biliary pancreatitis: clinical presentation and surgical management. Am J Surg 1986; 151:170–175.
- Uhl W, Warshaw A, Imrie C, et al; International Association of Pancreatology. IAP guidelines on the surgical management of acute pancreatitis. Pancreatology 2002; 2:565–573.
- Banks PA, Freeman ML; Practice Parameters Committee of the American College of Gastroenterology. Practice guidelines in acute pancreatitis. Am J Gastroenterol 2006; 101:2379–2400.
- Forsmark CE, Baillie J; AGA Institute Clinical Practice and Economics Committee. AGA Institute technical review on acute pancreatitis. Gastroenterology 2007; 132:2022–2044.
- United Kingdom guidelines for the management of acute pancreatitis. British Society of Gastroenterology. Gut 1998; 42(suppl 2):S1–S13.
- Norton SA, Cheruvu CV, Collins J, Dix FP, Eyre-Brook IA. An assessment of clinical guidelines for the management of acute pancreatitis. Ann R Coll Surg Engl 2001; 83:399–405.
- Chiang DT, Anozie A, Fleming WR, Kiroff GK. Comparative study on acute pancreatitis management. ANZ J Surg 2004; 74:218–221.
- Barnard J, Siriwardena AK. Variations in implementation of current national guidelines for the treatment of acute pancreatitis: implications for acute surgical service provision. Ann R Coll Surg Engl 2002; 84:79–81.
- Lankisch PG, Weber-Dany B, Lerch MM. Clinical perspectives in pancreatology: compliance with acute pancreatitis in Germany [letter]. Pancreatology 2005; 5:591–593.
- Connor SJ, Lienert AR, Brown LA, Bagshaw PF. Closing the audit loop is necessary to achieve compliance with evidence-based guidelines in the management of acute pancreatitis. N Z Med J 2008; 121:19–25.
- Mofidi R, Duff MD, Wigmore SJ, Madhavan KK, Garden OJ, Parks RW. Association between early systemic inflammatory response, severity of multiorgan dysfunction, and death in acute pancreatitis. Br J Surg 2006; 93:738–744.
- Brown A, Orav J, Banks PA. Hemoconcentration is an early marker for organ failure and necrotizing pancreatitis. Pancreas 2000; 20:367–372.
- Lankisch PG, Mahlke R, Blum T, et al. Hemoconcentration: an early marker of severe and/or necrotizing pancreatitis? A critical appraisal. Am J Gastroenterol 2001; 96:2081–2085.
- Singh VK, Wu BU, Bollen TL, et al. A prospective evaluation of the bedside index for severity in acute pancreatitis score in assessing mortality and intermediate markers of severity in acute pancreatitis. Am J Gastroenterol 2009; 104:966–971.
- Ranson JH, Rifkind KM, Roses DF, Fink SD, Eng K, Spencer FC. Prognostic signs and the role of operative management in acute pancreatitis. Surg Gynecol Obstet 1974; 139:69–81.
- Larvin M. Assessment of clinical severity and prognosis. In:Beger HG, Warshaw AL, Buchler MW, et al, editors. The Pancreas. Blackwell Science: New York, 1998:489–502.
- Wu BU, Johannes RS, Sun X, Tabak Y, Conwell DL, Banks PA. The early prediction of mortality in acute pancreatitis: a large population-based study. Gut 2008; 57:1698–1703.
- Bradley EL. A clinically based classification system for acute pancreatitis. Summary of the International Symposium on Acute Pancreatitis, Atlanta, GA, September 11 through 13, 1992. Arch Surg 1993, 128:586–590.
- Forgacs B, Eible G, Faulhaber J, Kahrau S, Buhr H, Foitzik T. Effect of fluid resuscitation with and without endothelin A receptor blockade on hemoconcentration and organ function in experimental pancreatitis. Eur Surg Res 2000; 32:162–168.
- Brown A, Baillargeon JD, Hughes MD, Banks PA. Can fluid resuscitation prevent pancreatic necrosis in severe acute pancreatitis? Pancreatology 2002; 2:104–107.
- Pandol SJ, Saluja AK, Imrie CW, Banks PA. Acute pancreatitis: bench to the bedside. Gastroenterology 2007; 132:1127–1151.
- Pezzilli R, Uomo G, Gabbrielli A, et al; ProInf-AISP Study Group. A prospective multicenter survey on the treatment of acute pancreatitis in Italy. Dig Liver Dis 2007; 39:838–846.
- Gardner TB, Vege SS, Pearson RK, Chari ST. Fluid resuscitation in acute pancreatitis. Clin Gastroenterol Hepatol 2008; 6:1070–1076.
- Petrov MS, Pylypchuk RD, Emelyanov NV. Systematic review: nutritional support in acute pancreatitis. Aliment Pharmacol Ther 2008; 28:704–712.
- Marik PE, Zaloga GP. Meta-analysis of parenteral nutrition versus enteral nutrition in patients with acute pancreatitis. BMJ 2004; 328:1407.
- Balthazar EJ, Robinson DL, Megibow AJ, Ranson JH. Acute pancreatitis: value of CT in establishing prognosis. Radiology 1990; 174:331–336.
- Balthazar EJ. Acute pancreatitis: assessment of severity with clinical and CT evaluation. Radiology 2002; 223:603–613.
- Foitzik T, Bassi DG, Schmidt J, et al. Intravenous contrast medium accentuates the severity of acute necrotizing pancreatitis in the rat. Gastroenterology 1994; 106:207–214.
- Carmona-Sanchez R, Uscanga L, Bezaury-Rivas P, Robles-Díaz G, Suazo-Barahona J, Vargas-Vorácková F. Potential harmful effect of iodinated intravenous contrast medium on the clinical course of mild acute pancreatitis. Arch Surg 2000; 135:1280–1284.
- Uhl W, Roggo A, Kirschstein T, et al. Influence of contrast-enhanced computed tomography on couse and outcome in patients with acute pancreatitis. Pancreas 2002; 24:191–197.
- Foitzik T, Klar E. Non-compliance with guidelines for the management of severe acute pancreatitis among German surgeons. Pancreatology 2007; 7:80–85.
- Villatoro E, Bassi C, Larvin M. Antibiotic therapy for prophylaxis against infection of pancreatic necrosis in acute pancreatitis. Cochrane Database Syst Rev 2006;CD002941.
- Heinrich S, Schafer M, Rousson V, Clavien PA. Evidence-based treatment of acute pancreatitis: a look at established paradigms. Ann Surg 2006; 243:154–168.
- Neoptolemos JP, Carr-Locke DL, London NJ, Bailey IA, James D, Fossard DP. Controlled trial of urgent endoscopic retrograde cholangiopancreatography and endoscopic sphincterotomy versus conservative treatment for acute pancreatitis due to gallstones. Lancet 1988; 2:979–983.
- Fan ST, Lai EC, Mok FP, Lo CM, Zheng SS, Wong J. Early treatment of acute biliary pancreatitis by endoscopic papillotomy. N Engl J Med 1993; 328:228–232.
- Folsch UR, Nitsche R, Ludtke R, Hilgers RA, Creutzfeldt W. Early ERCP and papillotomy compared with conservative treatment for acute biliary pancreatitis. The German Study Group on Acute Biliary Pancreatitis. N Engl J Med 1997; 336:237–242.
- Ayub K, Imada R, Slavin J. Endoscopic retrograde cholangiopancreatography in gallstone associated pancreatitis. Cochrane Database Syst Rev 2004;CD003630
- Oria A, Cimmino D, Ocampo C, et al. Early endoscopic intervention versus early conservative management in patients with acute gallstone pancreatitis and biliopancreatic obstruction. A randomized clinical trial. Ann Surg 2007; 245:10–17.
- Frei GJ, Frei VT, Thirlby RC, McClelland RN. Biliary pancreatitis: clinical presentation and surgical management. Am J Surg 1986; 151:170–175.
- Uhl W, Warshaw A, Imrie C, et al; International Association of Pancreatology. IAP guidelines on the surgical management of acute pancreatitis. Pancreatology 2002; 2:565–573.
KEY POINTS
- Serum amylase and lipase levels are often needlessly measured every day.
- Often, severity assessments are not performed regularly or acted on.
- Often, not enough fluid is replaced, or fluid status is not adequately monitored.
- In many severe cases, enteral or parenteral feeding is not started soon enough.
- Computed tomography is not done in many patients with severe acute pancreatitis, or it is performed too soon.
- In many cases of suspected infected necrosis, fine-needle aspiration is not done.
- Broad-spectrum antibiotics are often used inappropriately in patients with mild acute pancreatitis and in patients with sterile necrotizing pancreatitis who are clinically stable and have no signs of sepsis.
Current therapies to shorten postoperative ileus
Rather than meerely wait for bowel sounds to return after patients undergo surgery, we can try to get the gut working again sooner. An active approach might shorten the duration of postoperative ileus, allow patients to go home from the hospital sooner, and improve their outcomes.
In the pages that follow, we review the pathophysiology, diagnosis, and current therapies to alter the course of postoperative ileus.
ILEUS CAN AFFECT THE STOMACH OR SMALL OR LARGE INTESTINES
Ileus is the absence of intestinal peristalsis without mechanical obstruction; postoperative ileus refers to the time after surgery before coordinated electromotor bowel function resumes.
Gastroparesis refers to abnormal gastric motility leading to impaired gastric emptying. This disabling, potentially chronic condition is associated with certain medical conditions such as diabetes, but can also occur after some surgical procedures, as we will discuss. It has been estimated to affect approximately 4% of the adult population, with a strong female predilection.1 Postoperative gastroparesis is probably most common after pancreaticoduodenectomy, in which it occurs in up to 57% of patients.2 Consensus guidelines for grading the severity of gastroparesis have been devised to help standardize the reporting of outcomes.2
Acute colonic pseudo-obstruction (colonic ileus) is often seen in elderly hospitalized patients with multiple medical comorbidities. Of note, it often occurs after surgery to parts of the body other than the abdomen, such as after orthopedic procedures. One study documented an incidence of 1.3% after hip replacement surgery and 1.2% after spine procedures.3
The small bowel normally resumes activity several hours after surgery, the stomach 24 to 48 hours after surgery, and the colon 3 to 5 days after surgery.4 When postoperative ileus persists longer than this, it can be considered pathologic and is sometimes called paralytic ileus.4,5
ILEUS AFFECTS OUTCOMES AND COSTS
Although not usually considered life-threatening, postoperative ileus is harmful for the patient and costly for society.
For the patient, ileus is uncomfortable, leads to nausea and vomiting, delays return to enteral nutrition, and prolongs the stay in the hospital. For many if not most patients undergoing gastrointestinal surgery, return of bowel function is the factor that delays going home. A prolonged hospital stay increases the risk of hospital-acquired infections, deep vein thrombosis, and other conditions.
The economic burden is also considerable. A retrospective review of more than 800,000 patients who underwent surgery in the United States in 2002 found a rate of postoperative ileus of 4.25% according to International Classification of Diseases–Ninth Revision (ICD-9) codes.6 The mean hospital length of stay was 9.3 days in patients with postoperative ileus vs 5.3 days in those without it. The difference in mean total hospital costs was US $6,300 per patient. The costs certainly add up when you consider the number of surgical procedures performed every year.
NEURAL AND CHEMICAL FACTORS
While observing exteriorized bowel in 1872, Goltz7 first noted enhanced spontaneous contractions when the spinal cord was severed at the level of the medulla. Not long after, Bayliss and Starling8 used a device called an “enterograph” to monitor small-bowel activity in vivo in dogs and found that cutting the splanchnic nerves led to vigorous bowel contraction after laparotomy. These early observations formed the foundation of our understanding of postoperative ileus and some of its possible causes.
Normal bowel contractility is influenced by a host of neural and chemical factors, the relative contributions of which vary depending on the segment of bowel.
The migrating motor complex is the basal level of activity in the bowel in the fasting state, serving a “housekeeping” function.9 It has four phases, consisting of escalating electrical and contractile activity punctuated by periods of quiescence. The resumption of this motor complex after surgery is responsible for recovery from postoperative ileus.
Sympathetic-parasympathetic imbalance
The sympathetic nervous system inhibits the small bowel; the parasympathetic nervous system stimulates it. Although vagal (parasympathetic) stimulation appears to have little actual impact on small-bowel activity, if sympathetic activity is blocked, contractility increases, indicating that tonic sympathetic inhibition normally predominates. The balance of these two competing influences determines the amount of acetylcholine released by excitatory nerve fibers in the myenteric plexi of the bowel.
These neural pathways can be manipulated clinically. Epidural catheters can block sympathetic output, thus allowing small-bowel function to return faster.
Vagus nerve activity appears to be more important in the stomach, where it promotes receptive relaxation of the fundus and contraction of the antrum, facilitating gastric emptying.10 After vagotomy, emptying of liquids may be normal or accelerated, but emptying of solids is impaired. This can occur after peptic ulcer surgery but is more likely after gastric resection for malignancy or after inadvertent vagal nerve injury during antireflux surgery.
The enteric nervous system is a complex, intrinsic network of neurons consisting of two distinct plexi within the bowel wall: the submucosal (Meissner) plexus, and the myenteric (Auerbach) plexus.11 The enteric nervous system in the small bowel is fundamentally different than the one in the colon in that the former contains gap junctions, allowing for coordinated electrical activity. Lacking these gap junctions, the colon depends more on input from the autonomic nervous system, perhaps explaining the longer recovery from postoperative ileus and the susceptibility to isolated colonic ileus due to a variety stressors and traumatic insults.12
Chemical mediators of bowel activity
A host of chemical mediators influence bowel motility. Perhaps the most important nonadrenergic inhibitor of gastrointestinal motility is nitric oxide.13 Animal studies have firmly established nitric oxide as an important factor in postoperative ileus, but its exact role in humans is not clear.14,15
Other mediators with possible roles include vasoactive intestinal peptide, substance P, calcitonin gene-related peptide, and endogenous opioids.13 Lack of duodenal-derived motilin is thought to be one cause of delayed gastric emptying after pancreaticoduodenectomy.2
Inflammation
The inflammatory response after surgery has also been an attractive target of study of the factors promoting postoperative ileus. In rat studies, Kalff et al16 found that surgical manipulation of the bowel induced an inflammatory cellular infiltrate in the bowel wall and diminished the response of smooth muscle to cholinergic stimulation. Cyclooxygenase-2, the enzymatic precursor to prostaglandins, has also been shown to be induced in enteric neurons after laparotomy.17
Narcotic analgesics
One of the greatest hurdles in preventing postoperative ileus is the use of narcotic analgesics to treat postoperative pain. It is also one of the most important modifiable risk factors.
Opiates delay colonic transit in postoperative patients, an effect that can be reversed by the narcotic antagonist naloxone (Narcan).18 This inhibitory effect is mediated by peripheral mu-opioid receptors. In a study of patients undergoing colectomy, the more morphine given, the longer the time to the return of bowel sounds and flatus and the first bowel movement.19
These observations have led to a search for selective opiate antagonists that allow narcotics to continue relieving pain while counteracting their effect on bowel motility, a topic discussed later in this review.
Nonsteroidal anti-inflammatory drugs such as ketorolac (Toradol) are attractive alternatives to opiate analgesics, both for their anti-inflammatory effect and for their opiatesparing properties. However, they can cause bleeding, renal insufficiency, and gastritis, drawbacks that limit their applicability and duration of use.
DIAGNOSIS BY CLINICAL SUSPICION AND IMAGING
The diagnosis of postoperative ileus is driven by a combination of clinical suspicion and imaging tests.
Regardless of the segment of bowel involved, it is imperative to exclude an obstructive cause. The diagnosis of ileus is presumed once obstruction has been excluded.
Diagnosing gastroparesis
Postoperative gastroparesis is usually suspected by its symptoms of early satiety, nausea, vomiting, eructation, and gastroesophageal reflux. Abdominal distention is usually not a prominent sign, but a succussion splash may be detected, indicating retention of food and liquid in the stomach.
Plain radiographs may reveal a large gastric air bubble in the left upper quadrant but may underestimate the degree of gastric distention. Computed tomography (CT) may show a large, fluid-filled stomach, often containing high-density food debris.
The gold standard for diagnosis is gastric emptying scintigraphy after a radiolabelled solid meal. The patient consumes a meal of egg white labelled with technetium 99m sulfur colloid, and scanning is performed at specified intervals to measure the percent retention of the isotope. Retention of more than 10% at 4 hours is considered abnormal.1 Severity can be graded on the basis of percent retention after 4 hours.20
This test is rarely indicated in the acute postoperative setting, however, and patients should be treated presumptively to prevent aspiration once mechanical obstruction is excluded.
Diagnosing small-bowel ileus
Small-bowel ileus often presents like gastroparesis, except that it more often causes abdominal distention. Plain radiographs reveal air-fluid levels and dilated loops of bowel.
Small-bowel ileus must then be differentiated from small-bowel obstruction by clinical and radiographic features. The presence of crampy abdominal pain, bowel sounds, and some bowel function implies a degree of mechanical obstruction. Plain radiographs showing “step-ladder” air-fluid levels also suggest obstruction. CT is more definitive in diagnosing obstruction by the presence of distended and decompressed bowel loops and may also reveal a source of obstruction (eg, postoperative interloop abscess).
Diagnosing colonic ileus
Colonic ileus is also characterized by abdominal distention, sometimes marked. Although it is the colon that is primarily involved, upstream small-bowel dilatation can also be seen if the ileocecal valve is incompetent. The cecum often shows the greatest degree of dilatation on plain radiographs and is at the greatest risk of perforation. CT, contrast enema studies, and endoscopy help rule out mechanical obstruction due to volvulus or a mass lesion.
STRATEGIES TO PREVENT AND TREAT ILEUS
Many strategies have been applied to prevent and manage postoperative ileus, ranging from changes in surgical technique, supportive care, and patient-initiated activities, to pharmacologic intervention.
Epidural anesthesia shortens ileus, reduces the need for narcotics
Epidural anesthesia has shown promise not only in improving pain control, but also in shortening the period of postoperative ileus. Most surgical patients either receive an epidural catheter before surgery, which is left in place for postoperative pain control, or are given patient-controlled analgesia with a narcotic. Generally, the surgeon chooses the pain control method.
Thoracic epidural analgesia has been shown to hasten the return of bowel function by 1 to 2 days and to reduce the need for opiates compared with systemic opioids alone.21–26 A likely explanation is that epidural anesthesia interferes with the afferent and efferent sympathetic reflex arcs. The level of the epidural placement is important: a thoracic epidural is needed to effectively block these sympathetic pathways.
Laparoscopic surgery is less traumatic
Laparoscopy has changed the landscape of surgery over the past few decades. Some of the most common surgical procedures (appendectomy, cholecystectomy) are now done mainly via the laparoscope, as are many procedures that are more complex.
Laparoscopic surgery has several advantages over open surgery. With smaller incisions, it is less traumatic to the body. The systemic inflammatory response appears to be less vigorous after laparoscopic surgery than after open surgery, as measured by circulating levels of interleukin 1, interleukin 6, and C-reactive protein.27
The length of stay after a laparoscopic procedure is shorter than after an open procedure for several reasons, not the least of which is a shorter duration of postoperative ileus. Animal studies show that intestinal recovery is faster after laparoscopic procedures than after open procedures.28–30 In a study in which their other care was comparable, significantly fewer patients undergoing laparoscopic colectomy had emesis or needed their nasogastric tube to be reinserted than patients who underwent an open operation, and their length of stay was shorter.31
As technology continues to advance in minimally invasive surgery, it is reasonable to expect these trends to continue.
Nasogastric tubes in selected cases
Patients are often allowed nothing by mouth or only minimal oral intake immediately after abdominal surgery, with or without nasogastric decompression. The role of nasogastric decompression has long been a topic of controversy. In a meta-analysis of 26 trials with 3,964 patients, the groups in which all patients routinely received a nasogastric tube had higher rates of pneumonia, fever, and atelectasis and longer duration to resumption of oral feeding than the groups in which nasogastric tubes were used selectively.32
Most clinicians agree that nasogastric tubes are uncomfortable and do little to prevent postoperative ileus. However, in selected cases they are useful for managing intractable vomiting and for preventing aspiration of gastric contents.
Early enteral feeding
Evidence is mounting that early postoperative enteral feeding may be advantageous for recovery.
In 1,173 patients undergoing both upper and lower gastrointestinal surgery in 13 trials, fewer patients died who were randomized to receive enteral feeding within 24 hours.33 There were also fewer infectious complications and anastomotic problems and a shorter length of stay, but these differences were not statistically significant. Vomiting was more common in the early-feeding groups but did not lead to higher rates of morbidity. Enteral feeding was by the oral, nasoduodenal, or nasojejunal routes, depending on the type of surgery performed.
Whether the number of calories given affects the outcome remains to be clarified, but at least for now it seems that feeding patients early in the course of their recovery is not detrimental and may in fact be beneficial.
Gum-chewing
Gum-chewing has been studied over the last decade as a form of sham feeding to stimulate bowel recovery after surgery. The presumed mechanism of action is vagal cholinergic (parasympathetic) stimulation of the gastrointestinal tract, similar to oral intake but with theoretically less risk of vomiting and aspiration.
In five such trials in patients undergoing colon resection, gum-chewing shortened the time until first flatus and bowel movement, but made no significant difference in length of stay.34
At the very least, gum-chewing immediately after surgery is a cheap and harmless strategy for reducing postoperative ileus, and it might make the patient more comfortable.
DRUGS THAT COAX THE GUT BACK TO WORK
Drugs that coax the gastrointestinal tract back to work have been tried for many years and have recently gained renewed enthusiasm. Their efficacy varies according to their target organ, with greater success in the stomach and colon than in the small bowel.
Cisapride (Propulsid) was an effective gastric prokinetic agent, as shown in several controlled trials. However, it was withdrawn from the US market in 2000 because of its propensity to cause cardiac arrhythmias.
Erythromycin is a macrolide antibiotic that is also a motilin receptor agonist. In patients who underwent antrectomy and vagotomy, it was shown to accelerate gastric emptying by roughly 40% as measured by solid-phase gastric emptying scintigraphy.35,36 In a randomized controlled trial in 118 patients who underwent pancreaticoduodenectomy, intravenous erythromycin reduced gastroparesis by 37% (measured by solid-phase gastric emptying study) and also reduced the need for nasogastric tube reinsertion.37 A major shortcoming is the development of tachyphylaxis, thought to be mediated by down-regulation of motilin receptors.
Metoclopramide (Reglan) is an antiemetic and prokinetic that acts as a dopamine D2 receptor antagonist and mixed serotonin 5-HT3 antagonist/5-HT4 agonist. Metoclopramide also stimulates gastric emptying, as shown in controlled trials in patients in intensive care units.38,39 The drug should not be used in patients with parkinsonism, in view of its antidopamine properties.
In 2009, the US Food and Drug Administration required that a black box warning be added to metoclopramide because of the risk of tardive dyskinesia with long-term use, and recommended that its use be limited to 3 weeks in the acute setting.40 Prescribers and patients need to decide if this risk is worth the potential benefit on a case-by-case basis.
Although erythromycin and metoclopramide are effective in managing gastroparesis, neither has been shown to be effective for small-bowel ileus.41,42 However, colonic ileus is highly responsive to drug therapy.
Neostigmine (Prostigmin) is a reversible acetylcholinesterase inhibitor that enhances the activity of the neurotransmitter acetylcholine at muscarinic receptors. It is the first-line treatment for colonic ileus.43 In three randomized, placebo-controlled trials,44–46 the success rates were 85% to 94% after the first dose.
Neostigmine is generally given either as an intravenous bolus dose of 2 to 2.5 mg or as an intravenous infusion over 24 hours. It must be given in a monitored setting, as both bradycardia and bronchospasm can occur. Patients should continue to be monitored clinically and with plain abdominal radiography after the drug is given, and they sometimes require a second or third dose.
In cases in which neostigmine fails, decompressive colonoscopy can be done as a second-line measure.
Alvimopan (Entereg), a peripherally acting, mu-opioid receptor antagonist, has come on the scene most recently. This agent first showed promise when it precipitated diarrhea in morphine-dependent mice.47 Early studies in humans focused on its ability to reverse the effect of opiates on gastrointestinal transit without interfering with their analgesic properties.48–50 Later investigations concentrated on its ability to reduce the duration of postoperative ileus after a variety of major abdominal surgical procedures.51,52
A pooled analysis of phase III studies of alvimopan focused on the subset of 1,212 patients who underwent bowel resections; it found a significant reduction in the time to gastrointestinal tract recovery and hospital discharge.53 A 12-mg dose was more beneficial than a 6-mg dose, especially in females and in older patients (over age 65).
Most recently, a multicenter, double-blind, placebo-controlled trial evaluated alvimopan as part of a standardized postoperative care plan in 654 patients undergoing partial small-bowel and large-bowel resection.54 The alvimopan group took less time to have their first bowel movements, pass flatus, and tolerate solid food. Patients randomized to alvimopan also had their discharge orders written an average of 1 day sooner than the placebo group. Importantly, opioid use was the same in both groups.
Alvimopan is given as a single oral dose of 12 mg 30 to 90 minutes before surgery and twice daily after surgery for up to 7 days, for a total of 15 doses. It is contraindicated in patients receiving therapeutic doses of opiates for more than 7 consecutive days immediately before surgery. Its use is currently limited to hospitals enrolled in the EASE (Entereg Access Support and Education) program.
Common adverse effects include constipation, dyspepsia, flatulence, and urinary retention. In a placebo-controlled 12-month study in patients treated with opiates for chronic pain, there were more reports of myocardial infarction in the alvimopan group.55 This finding has not been replicated in any other study. The need to give the drug preoperatively obviously necessitates identifying patients most at risk of postoperative ileus.
FUTURE DIRECTIONS
A multimodal approach to managing postoperative ileus seems likely to be the most effective model in the long run. This should involve using minimally invasive surgery when possible, pharmacotherapy, and accelerated standardized postoperative care.
Standardized postoperative care has been implemented for a variety of procedures and generally involves minimal (if any) use of nasogastric tubes, early enteral intake and ambulation, and specific discharge criteria such as passage of flatus or stool, adequate pain control, and tolerance of solid food.56–58 Compared with a “traditional” (nonstandardized) approach, standardized care has led to shorter hospital stays and lower costs with no impact on rates of morbidity or readmission.59,60 (However, one clearly cannot underestimate the role of patient expectations in the success of such postoperative care pathways.)
There are plenty of incentives for patients, physicians, health care organizations, and third-party payers to support this push. For patients, it means less time in the hospital and a quicker return to eating normally. Surgeons can expect more-satisfied patients and lower rates of hospital-acquired conditions. For hospitals and insurers, it means less use of resources for some patients, making resources available to those who need them more.
- Waseem S, Moshiree B, Draganov P. Gastroparesis: current diagnostic challenges and management considerations. World J Gastroenterol 2009; 15:25–37.
- Wente MN, Bassi C, Dervenis C, et al. Delayed gastric emptying (DGE) after pancreatic surgery: a suggested definition by the International Study Group of Pancreatic Surgery (ISGPS). Surgery 2007; 142:761–768.
- Norwood MG, Lykostratis H, Garcea G, Berry DP. Acute colonic pseudo-obstruction following major orthopaedic surgery. Colorectal Dis 2005; 7:496–499.
- Livingston EH, Passaro EP. Postoperative ileus. Dig Dis Sci 1990; 35:121–132.
- Catchpole BN. Ileus: use of sympathetic blocking agents in its treatment. Surgery 1969; 66:811–820.
- Saunders WB, Bowers B, Moss B, et al. Recorded rate and economic burden associated with postoperative ileus [abstract]. Presented at the ASHP Midyear Clinical Meeting, Orlando, FL, December 7, 2004. Abstract 30346.
- Neely J, Catchpole BN. The restoration of alimentary-tract motility by pharmacological means. Br J Surg 1971; 58:21–28.
- Bayliss WM, Starling EH. The movements and innervations of the small intestine. J Physiol 1899; 24:99–143.
- Szurszewksi JH. A migrating electrical complex of the canine small intestine. Am J Physiol 1969; 217:1757–1763.
- Livingston EH. Stomach and duodenum. In:Norton JA, Bollinger RR, Chang AE, et al (editors). Surgery: Basic Science and Clinical Evidence. New York, Springer-Verlag, 2001:489–516.
- Simeone DM. Anatomy and physiology of the small intestine. In:Mulholland MW, Lillemoe KD, Doherty GM, Maier RV, Upchurch GR, editors. Greenfield’s Surgery: Scientific Principles and Practice, 4th ed. Philadelphia: Lippincott Williams and Wilkins, 2006:756–766.
- Baig MK, Wexner SD. Postoperative ileus: a review. Dis Colon Rectum 2004; 47:516–526. Retraction in Dis Colon Rectum 2005; 48:1983.
- Luckey A, Livingston E, Taché Y. Mechanisms and treatment of postoperative ileus. Arch Surg 2003; 138:206–214.
- De Winter BY, Boeckxstaens GE, De Man JG, Moreels TG, Herman AG, Pelckmans PA. Effect of adrenergic and nitrergic blockade on experimental ileus in rats. Br J Pharmacol 1997; 120:464–468.
- Kalff JC, Schraut WH, Billiar TR, Simmons RL, Bauer AJ. Role of inducible nitric oxide synthase in postoperative intestinal smooth muscle dysfunction in rodents. Gastroenterology 2000; 118:316–327.
- Kalff JC, Schraut WH, Simmons RL, Bauer AJ. Surgical manipulation of the gut elicits an intestinal muscularis inflammatory response resulting in postsurgical ileus. Ann Surg 1998; 228:652–663.
- Schwarz NT, Kalff JC, Türler A, et al. Prostanoid production via COX-2 as a causative mechanism of rodent postoperative ileus. Gastroenterology 2001; 121:1354–1371.
- Kaufman PN, Krevsky B, Malmud LS, et al. Role of opiate receptors in the regulation of colonic transit. Gastroenterology 1988; 94:1351–1356.
- Cali RL, Meade PG, Swanson MS, Freeman C. Effect of morphine and incision length on bowel function after colectomy. Dis Colon Rectum 2000; 43:163–168.
- Abell TL, Camilleri M, Donahoe K, et al; American Neurogastroenterology and Motility Society and the Society of Nuclear Medicine. Consensus recommendations for gastric emptying scintigraphy: a joint report of the American Neurogastroenterology and Motility Society and the Society of Nuclear Medicine. Am J Gastroenterol 2008; 103:753–763.
- Zingg U, Miskovic D, Hamel CT, Erni L, Oertli D, Metzger U. Influence of thoracic epidural analgesia on postoperative pain relief and ileus after laparoscopic colorectal resection: benefit with epidural analgesia. Surg Endosc 2009; 23:276–282.
- Taqi A, Hong X, Mistraletti G, Stein B, Charlebois P, Carli F. Thoracic epidural analgesia facilitates the restoration of bowel function and dietary intake in patients undergoing laparoscopic colon resection using a traditional, nonaccelerated, perioperative care program. Surg Endosc 2007; 21:247–252.
- Kuo CP, Jao SW, Chen KM, et al. Comparison of the effects of thoracic epidural analgesia and i.v. infusion with lidocaine on cytokine response, postoperative pain, and bowel function in patients undergoing colonic surgery. Br J Anaesth 2006; 97:640–646.
- Carli F, Trudel JL, Belliveau P. The effect of intraoperative thoracic epidural anesthesia and postoperative analgesia on bowel function after colorectal surgery: a prospective, randomized trial. Dis Colon Rectum 2001; 44:1083–1089.
- Jørgensen H, Wetterslev J, Møiniche S, Dahl JB. Epidural local anaesthetics versus opioid-based analgesic regimens on postoperative gastrointestinal paralysis, PONV and pain after abdominal surgery. Cochrane Database Syst Rev 2000; 4:CD001893.
- de Leon-Casasola OA, Karabella D, Lema MJ. Bowel function recovery after radical hysterectomies: thoracic epidural bupivacainemorphine versus intravenous patient-controlled analgesia with morphine: a pilot study. J Clin Anesth 1996; 8:87–92.
- Leung KL, Lai PB, Ho RL, et al. Systemic cytokine response after laparoscopic-assisted resection of rectosigmoid carcinoma: a prospective randomized trial. Ann Surg 2000; 231:506–511.
- Böhm B, Milsom JW, Fazio VW. Postoperative intestinal motility following conventional and laparoscopic intestinal surgery. Arch Surg 1995; 130:415–419.
- Davies W, Kollmorgen CF, Tu QM, et al. Laparoscopic colectomy shortens postoperative ileus in a canine model. Surgery 1997; 121:550–555.
- Hotokezaka M, Combs MJ, Mentis EP, Schirmer BD. Recovery of fasted and fed gastrointestinal motility after open versus laparoscopic cholecystectomy in dogs. Ann Surg 1996; 223:413–419.
- Chen HH, Wexner SD, Iroatulam AJ, et al. Laparoscopic colectomy compares favorably with colectomy by laparotomy for reduction of postoperative ileus. Dis Colon Rectum 2000; 43:61–65.
- Cheatham ML, Chapman WC, Key SP, Sawyers JL. A meta-analysis of selective versus routine nasogastric decompression after elective laparotomy. Ann Surg 1995; 221:469–478.
- Lewis SJ, Andersen HK, Thomas S. Early enteral nutrition within 24 h of intestinal surgery versus later commencement of feeding: a systematic review and meta-analysis. J Gastrointest Surg 2009; 13:569–575.
- Purkayastha S, Tilney HS, Darzy AW, Tekkis PP. Meta-analysis of studies evaluating chewing gum to enhance postoperative recovery following colectomy. Arch Surg 2008; 143:788–793.
- Ramirez B, Eaker EY, Drane WE, Hocking MP, Sninsky CA. Erythromycin enhances gastric emptying in patients with gastroparesis after vagotomy and antrectomy. Dig Dis Sci 1994; 39:2295–2300.
- Kendall BJ, Chakravarti A, Kendall E, Soykan I, McCallum RW. The effect of intravenous erythromycin on solid meal gastric emptying in patients with chronic symptomatic post-vagotomy-antrectomy gastroparesis. Aliment Pharmacol Ther 1997; 11:381–385.
- Yeo CJ, Barry MK, Sauter PK, et al. Erythromycin accelerates gastric emptying after pancreaticoduodenectomy. A prospective, randomized, placebo-controlled trial. Ann Surg 1993; 218:229–237.
- Jooste CA, Mustoe J, Collee G. Metoclopramide improves gastric motility in critically ill patients. Intensive Care Med 1999; 25:464–468.
- Sustic A, Zelic M, Protic A, et al. Metoclopramide improves gastric but not gallbladder emptying in cardiac surgery patients with early intragastric enteral feeding: randomized controlled trial. Croat Med J 2005; 46:239–244.
- US Food and Drug Administration. FDA requires boxed warning and risk mitigation strategy for metoclopramide-containing drugs. Agency warns against chronic use of these products to treat gastrointestinal disorders. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm149533.htm. Accessed 8/24/2009.
- Smith AJ, Nissan A, Lanouette NM, et al. Prokinetic affect of erythromycin after colorectal surgery: a randomized, placebo-controlled, double-blind study. Dis Colon Rectum 2000; 43:333–337.
- Cheape JD, Wexner SD, Jagelman JK. Does metoclopramide reduce the length of ileus after colorectal surgery? A prospective randomized trial. Dis Colon Rectum 1991; 34:437–441.
- De Giorgio R, Knowles CH. Acute colonic pseudo-obstruction. Br J Surg 2009; 96:229–239.
- Ponec RJ, Saunders MD, Kimmey MB. Neostigmine for the treatment of acute colonic pseudo-obstruction. N Engl J Med 1999; 341:137–141.
- Amaro R, Rogers AI. Neostigmine infusions: new standard of care for acute colonic pseudo-obstruction? Am J Gastroenterol 2000; 95:304–305.
- van der Spoel JI, Oudemans-van Straaten HM, Stroutenbeek CP, Bosman RJ, Zandstra DF. Neostigmine resolves critical illness-related colonic ileus in intensive care patients with multiple organ failure: a prospective, double-blind, placebo-controlled trial. Intensive Care Med 2001; 27:822–827.
- Zimmerman DM, Gidda JS, Cantrell BE, et al. LY246736 dihydrate μ-opioid receptor antagonist. Drugs Future 1994; 19:1078–1083.
- Callaghan JT, Cerimele B, Nowak TV, et al. Effect of the opioid antagonist LY246736 on gastrointestinal transit in human subjects [abstract]. Gastroenterology 1998; 114:G3015.
- Hodgson PS, Liu SS, Carpenter RI. ADL 8-2698 prevents morphine inhibition of GI transit [abstract]. Clin Pharmacol Ther 2000; 67:93.
- Liu SS, Hodgson PS, Carpenter RL, Fricke JR. ADL 8-2698, a trans-3,4-dimethyl-4-(3-hydroxyphenyl) piperidine, prevents gastrointestinal effects of intravenous morphine without affecting analgesia. Clin Pharmacol Ther 2001; 68:66–71.
- Wolff BG, Michelassi F, Gerkin TM, et al. Alvimopan, a novel, peripherally acting mu opioid antagonist: results of a multicenter, randomized, double-blind, placebo-controlled, phase III trial of major abdominal surgery and postoperative ileus. Ann Surg 2004; 240:728–735.
- Delaney CP, Weese JL, Hyman NH, et al; Alvimopan Postoperative Ileus Study Group. Phase III trial of alvimopan, a novel, peripherally acting, mu opioid antagonist, for postoperative ileus after major abdominal surgery. Dis Colon Rectum 2005; 48:1114–1129.
- Delaney CP, Wolff BG, Viscusi ER, et al. Alvimopan, for postoperative ileus following bowel resection: a pooled analysis of phase III studies. Ann Surg 2007; 245:355–363.
- Ludwig K, Enker WE, Delaney CP, et al. Gastrointestinal tract recovery in patients undergoing bowel resection: results of a randomized trial of alvimopan and placebo with a standardized accelerated postoperative care pathway. Arch Surg 2008; 143:1098–1105.
- Adolor GlaxoSmithKline. Entereg (alvimopan). www.entereg.com/Accessed 8/24/2009.
- Pritts TA, Nussbaum MS, Flesch LV, Fegelman EJ, Parikh AA, Fischer JE. Implementation of a clinical pathway decreases length of stay and cost for bowel resection. Ann Surg 1999; 230:728–733.
- Delaney CP, Zutshi M, Senagore AJ, Remzi FH, Hammel J, Fazio VW. Prospective, randomized, controlled trial between a pathway of controlled rehabilitation with early ambulation and diet and traditional postoperative care after laparotomy and intestinal resection. Dis Colon Rectum 2003; 46:851–859.
- Joh YG, Lindsetmo RO, Stulberg J, Obias V, Champagne B, Delaney CP. Standardized postoperative pathway: accelerating recovery after ileostomy closure. Dis Colon Rectum 2008; 51:1786–1789.
- Kennedy EP, Rosato EL, Sauter PK, et al. Initiation of a critical pathway for pancreaticoduodenectomy at an academic institution—the first step in multidisciplinary team building. J Am Coll Surg 2007; 204:917–924.
- Kariv Y, Delaney CP, Senagore AJ, et al. Clinical outcomes and cost analysis of a “fast track” postoperative care pathway for ileal pouch-anal anastomosis: a case control study. Dis Colon Rectum 2007; 50:137–146.
Rather than meerely wait for bowel sounds to return after patients undergo surgery, we can try to get the gut working again sooner. An active approach might shorten the duration of postoperative ileus, allow patients to go home from the hospital sooner, and improve their outcomes.
In the pages that follow, we review the pathophysiology, diagnosis, and current therapies to alter the course of postoperative ileus.
ILEUS CAN AFFECT THE STOMACH OR SMALL OR LARGE INTESTINES
Ileus is the absence of intestinal peristalsis without mechanical obstruction; postoperative ileus refers to the time after surgery before coordinated electromotor bowel function resumes.
Gastroparesis refers to abnormal gastric motility leading to impaired gastric emptying. This disabling, potentially chronic condition is associated with certain medical conditions such as diabetes, but can also occur after some surgical procedures, as we will discuss. It has been estimated to affect approximately 4% of the adult population, with a strong female predilection.1 Postoperative gastroparesis is probably most common after pancreaticoduodenectomy, in which it occurs in up to 57% of patients.2 Consensus guidelines for grading the severity of gastroparesis have been devised to help standardize the reporting of outcomes.2
Acute colonic pseudo-obstruction (colonic ileus) is often seen in elderly hospitalized patients with multiple medical comorbidities. Of note, it often occurs after surgery to parts of the body other than the abdomen, such as after orthopedic procedures. One study documented an incidence of 1.3% after hip replacement surgery and 1.2% after spine procedures.3
The small bowel normally resumes activity several hours after surgery, the stomach 24 to 48 hours after surgery, and the colon 3 to 5 days after surgery.4 When postoperative ileus persists longer than this, it can be considered pathologic and is sometimes called paralytic ileus.4,5
ILEUS AFFECTS OUTCOMES AND COSTS
Although not usually considered life-threatening, postoperative ileus is harmful for the patient and costly for society.
For the patient, ileus is uncomfortable, leads to nausea and vomiting, delays return to enteral nutrition, and prolongs the stay in the hospital. For many if not most patients undergoing gastrointestinal surgery, return of bowel function is the factor that delays going home. A prolonged hospital stay increases the risk of hospital-acquired infections, deep vein thrombosis, and other conditions.
The economic burden is also considerable. A retrospective review of more than 800,000 patients who underwent surgery in the United States in 2002 found a rate of postoperative ileus of 4.25% according to International Classification of Diseases–Ninth Revision (ICD-9) codes.6 The mean hospital length of stay was 9.3 days in patients with postoperative ileus vs 5.3 days in those without it. The difference in mean total hospital costs was US $6,300 per patient. The costs certainly add up when you consider the number of surgical procedures performed every year.
NEURAL AND CHEMICAL FACTORS
While observing exteriorized bowel in 1872, Goltz7 first noted enhanced spontaneous contractions when the spinal cord was severed at the level of the medulla. Not long after, Bayliss and Starling8 used a device called an “enterograph” to monitor small-bowel activity in vivo in dogs and found that cutting the splanchnic nerves led to vigorous bowel contraction after laparotomy. These early observations formed the foundation of our understanding of postoperative ileus and some of its possible causes.
Normal bowel contractility is influenced by a host of neural and chemical factors, the relative contributions of which vary depending on the segment of bowel.
The migrating motor complex is the basal level of activity in the bowel in the fasting state, serving a “housekeeping” function.9 It has four phases, consisting of escalating electrical and contractile activity punctuated by periods of quiescence. The resumption of this motor complex after surgery is responsible for recovery from postoperative ileus.
Sympathetic-parasympathetic imbalance
The sympathetic nervous system inhibits the small bowel; the parasympathetic nervous system stimulates it. Although vagal (parasympathetic) stimulation appears to have little actual impact on small-bowel activity, if sympathetic activity is blocked, contractility increases, indicating that tonic sympathetic inhibition normally predominates. The balance of these two competing influences determines the amount of acetylcholine released by excitatory nerve fibers in the myenteric plexi of the bowel.
These neural pathways can be manipulated clinically. Epidural catheters can block sympathetic output, thus allowing small-bowel function to return faster.
Vagus nerve activity appears to be more important in the stomach, where it promotes receptive relaxation of the fundus and contraction of the antrum, facilitating gastric emptying.10 After vagotomy, emptying of liquids may be normal or accelerated, but emptying of solids is impaired. This can occur after peptic ulcer surgery but is more likely after gastric resection for malignancy or after inadvertent vagal nerve injury during antireflux surgery.
The enteric nervous system is a complex, intrinsic network of neurons consisting of two distinct plexi within the bowel wall: the submucosal (Meissner) plexus, and the myenteric (Auerbach) plexus.11 The enteric nervous system in the small bowel is fundamentally different than the one in the colon in that the former contains gap junctions, allowing for coordinated electrical activity. Lacking these gap junctions, the colon depends more on input from the autonomic nervous system, perhaps explaining the longer recovery from postoperative ileus and the susceptibility to isolated colonic ileus due to a variety stressors and traumatic insults.12
Chemical mediators of bowel activity
A host of chemical mediators influence bowel motility. Perhaps the most important nonadrenergic inhibitor of gastrointestinal motility is nitric oxide.13 Animal studies have firmly established nitric oxide as an important factor in postoperative ileus, but its exact role in humans is not clear.14,15
Other mediators with possible roles include vasoactive intestinal peptide, substance P, calcitonin gene-related peptide, and endogenous opioids.13 Lack of duodenal-derived motilin is thought to be one cause of delayed gastric emptying after pancreaticoduodenectomy.2
Inflammation
The inflammatory response after surgery has also been an attractive target of study of the factors promoting postoperative ileus. In rat studies, Kalff et al16 found that surgical manipulation of the bowel induced an inflammatory cellular infiltrate in the bowel wall and diminished the response of smooth muscle to cholinergic stimulation. Cyclooxygenase-2, the enzymatic precursor to prostaglandins, has also been shown to be induced in enteric neurons after laparotomy.17
Narcotic analgesics
One of the greatest hurdles in preventing postoperative ileus is the use of narcotic analgesics to treat postoperative pain. It is also one of the most important modifiable risk factors.
Opiates delay colonic transit in postoperative patients, an effect that can be reversed by the narcotic antagonist naloxone (Narcan).18 This inhibitory effect is mediated by peripheral mu-opioid receptors. In a study of patients undergoing colectomy, the more morphine given, the longer the time to the return of bowel sounds and flatus and the first bowel movement.19
These observations have led to a search for selective opiate antagonists that allow narcotics to continue relieving pain while counteracting their effect on bowel motility, a topic discussed later in this review.
Nonsteroidal anti-inflammatory drugs such as ketorolac (Toradol) are attractive alternatives to opiate analgesics, both for their anti-inflammatory effect and for their opiatesparing properties. However, they can cause bleeding, renal insufficiency, and gastritis, drawbacks that limit their applicability and duration of use.
DIAGNOSIS BY CLINICAL SUSPICION AND IMAGING
The diagnosis of postoperative ileus is driven by a combination of clinical suspicion and imaging tests.
Regardless of the segment of bowel involved, it is imperative to exclude an obstructive cause. The diagnosis of ileus is presumed once obstruction has been excluded.
Diagnosing gastroparesis
Postoperative gastroparesis is usually suspected by its symptoms of early satiety, nausea, vomiting, eructation, and gastroesophageal reflux. Abdominal distention is usually not a prominent sign, but a succussion splash may be detected, indicating retention of food and liquid in the stomach.
Plain radiographs may reveal a large gastric air bubble in the left upper quadrant but may underestimate the degree of gastric distention. Computed tomography (CT) may show a large, fluid-filled stomach, often containing high-density food debris.
The gold standard for diagnosis is gastric emptying scintigraphy after a radiolabelled solid meal. The patient consumes a meal of egg white labelled with technetium 99m sulfur colloid, and scanning is performed at specified intervals to measure the percent retention of the isotope. Retention of more than 10% at 4 hours is considered abnormal.1 Severity can be graded on the basis of percent retention after 4 hours.20
This test is rarely indicated in the acute postoperative setting, however, and patients should be treated presumptively to prevent aspiration once mechanical obstruction is excluded.
Diagnosing small-bowel ileus
Small-bowel ileus often presents like gastroparesis, except that it more often causes abdominal distention. Plain radiographs reveal air-fluid levels and dilated loops of bowel.
Small-bowel ileus must then be differentiated from small-bowel obstruction by clinical and radiographic features. The presence of crampy abdominal pain, bowel sounds, and some bowel function implies a degree of mechanical obstruction. Plain radiographs showing “step-ladder” air-fluid levels also suggest obstruction. CT is more definitive in diagnosing obstruction by the presence of distended and decompressed bowel loops and may also reveal a source of obstruction (eg, postoperative interloop abscess).
Diagnosing colonic ileus
Colonic ileus is also characterized by abdominal distention, sometimes marked. Although it is the colon that is primarily involved, upstream small-bowel dilatation can also be seen if the ileocecal valve is incompetent. The cecum often shows the greatest degree of dilatation on plain radiographs and is at the greatest risk of perforation. CT, contrast enema studies, and endoscopy help rule out mechanical obstruction due to volvulus or a mass lesion.
STRATEGIES TO PREVENT AND TREAT ILEUS
Many strategies have been applied to prevent and manage postoperative ileus, ranging from changes in surgical technique, supportive care, and patient-initiated activities, to pharmacologic intervention.
Epidural anesthesia shortens ileus, reduces the need for narcotics
Epidural anesthesia has shown promise not only in improving pain control, but also in shortening the period of postoperative ileus. Most surgical patients either receive an epidural catheter before surgery, which is left in place for postoperative pain control, or are given patient-controlled analgesia with a narcotic. Generally, the surgeon chooses the pain control method.
Thoracic epidural analgesia has been shown to hasten the return of bowel function by 1 to 2 days and to reduce the need for opiates compared with systemic opioids alone.21–26 A likely explanation is that epidural anesthesia interferes with the afferent and efferent sympathetic reflex arcs. The level of the epidural placement is important: a thoracic epidural is needed to effectively block these sympathetic pathways.
Laparoscopic surgery is less traumatic
Laparoscopy has changed the landscape of surgery over the past few decades. Some of the most common surgical procedures (appendectomy, cholecystectomy) are now done mainly via the laparoscope, as are many procedures that are more complex.
Laparoscopic surgery has several advantages over open surgery. With smaller incisions, it is less traumatic to the body. The systemic inflammatory response appears to be less vigorous after laparoscopic surgery than after open surgery, as measured by circulating levels of interleukin 1, interleukin 6, and C-reactive protein.27
The length of stay after a laparoscopic procedure is shorter than after an open procedure for several reasons, not the least of which is a shorter duration of postoperative ileus. Animal studies show that intestinal recovery is faster after laparoscopic procedures than after open procedures.28–30 In a study in which their other care was comparable, significantly fewer patients undergoing laparoscopic colectomy had emesis or needed their nasogastric tube to be reinserted than patients who underwent an open operation, and their length of stay was shorter.31
As technology continues to advance in minimally invasive surgery, it is reasonable to expect these trends to continue.
Nasogastric tubes in selected cases
Patients are often allowed nothing by mouth or only minimal oral intake immediately after abdominal surgery, with or without nasogastric decompression. The role of nasogastric decompression has long been a topic of controversy. In a meta-analysis of 26 trials with 3,964 patients, the groups in which all patients routinely received a nasogastric tube had higher rates of pneumonia, fever, and atelectasis and longer duration to resumption of oral feeding than the groups in which nasogastric tubes were used selectively.32
Most clinicians agree that nasogastric tubes are uncomfortable and do little to prevent postoperative ileus. However, in selected cases they are useful for managing intractable vomiting and for preventing aspiration of gastric contents.
Early enteral feeding
Evidence is mounting that early postoperative enteral feeding may be advantageous for recovery.
In 1,173 patients undergoing both upper and lower gastrointestinal surgery in 13 trials, fewer patients died who were randomized to receive enteral feeding within 24 hours.33 There were also fewer infectious complications and anastomotic problems and a shorter length of stay, but these differences were not statistically significant. Vomiting was more common in the early-feeding groups but did not lead to higher rates of morbidity. Enteral feeding was by the oral, nasoduodenal, or nasojejunal routes, depending on the type of surgery performed.
Whether the number of calories given affects the outcome remains to be clarified, but at least for now it seems that feeding patients early in the course of their recovery is not detrimental and may in fact be beneficial.
Gum-chewing
Gum-chewing has been studied over the last decade as a form of sham feeding to stimulate bowel recovery after surgery. The presumed mechanism of action is vagal cholinergic (parasympathetic) stimulation of the gastrointestinal tract, similar to oral intake but with theoretically less risk of vomiting and aspiration.
In five such trials in patients undergoing colon resection, gum-chewing shortened the time until first flatus and bowel movement, but made no significant difference in length of stay.34
At the very least, gum-chewing immediately after surgery is a cheap and harmless strategy for reducing postoperative ileus, and it might make the patient more comfortable.
DRUGS THAT COAX THE GUT BACK TO WORK
Drugs that coax the gastrointestinal tract back to work have been tried for many years and have recently gained renewed enthusiasm. Their efficacy varies according to their target organ, with greater success in the stomach and colon than in the small bowel.
Cisapride (Propulsid) was an effective gastric prokinetic agent, as shown in several controlled trials. However, it was withdrawn from the US market in 2000 because of its propensity to cause cardiac arrhythmias.
Erythromycin is a macrolide antibiotic that is also a motilin receptor agonist. In patients who underwent antrectomy and vagotomy, it was shown to accelerate gastric emptying by roughly 40% as measured by solid-phase gastric emptying scintigraphy.35,36 In a randomized controlled trial in 118 patients who underwent pancreaticoduodenectomy, intravenous erythromycin reduced gastroparesis by 37% (measured by solid-phase gastric emptying study) and also reduced the need for nasogastric tube reinsertion.37 A major shortcoming is the development of tachyphylaxis, thought to be mediated by down-regulation of motilin receptors.
Metoclopramide (Reglan) is an antiemetic and prokinetic that acts as a dopamine D2 receptor antagonist and mixed serotonin 5-HT3 antagonist/5-HT4 agonist. Metoclopramide also stimulates gastric emptying, as shown in controlled trials in patients in intensive care units.38,39 The drug should not be used in patients with parkinsonism, in view of its antidopamine properties.
In 2009, the US Food and Drug Administration required that a black box warning be added to metoclopramide because of the risk of tardive dyskinesia with long-term use, and recommended that its use be limited to 3 weeks in the acute setting.40 Prescribers and patients need to decide if this risk is worth the potential benefit on a case-by-case basis.
Although erythromycin and metoclopramide are effective in managing gastroparesis, neither has been shown to be effective for small-bowel ileus.41,42 However, colonic ileus is highly responsive to drug therapy.
Neostigmine (Prostigmin) is a reversible acetylcholinesterase inhibitor that enhances the activity of the neurotransmitter acetylcholine at muscarinic receptors. It is the first-line treatment for colonic ileus.43 In three randomized, placebo-controlled trials,44–46 the success rates were 85% to 94% after the first dose.
Neostigmine is generally given either as an intravenous bolus dose of 2 to 2.5 mg or as an intravenous infusion over 24 hours. It must be given in a monitored setting, as both bradycardia and bronchospasm can occur. Patients should continue to be monitored clinically and with plain abdominal radiography after the drug is given, and they sometimes require a second or third dose.
In cases in which neostigmine fails, decompressive colonoscopy can be done as a second-line measure.
Alvimopan (Entereg), a peripherally acting, mu-opioid receptor antagonist, has come on the scene most recently. This agent first showed promise when it precipitated diarrhea in morphine-dependent mice.47 Early studies in humans focused on its ability to reverse the effect of opiates on gastrointestinal transit without interfering with their analgesic properties.48–50 Later investigations concentrated on its ability to reduce the duration of postoperative ileus after a variety of major abdominal surgical procedures.51,52
A pooled analysis of phase III studies of alvimopan focused on the subset of 1,212 patients who underwent bowel resections; it found a significant reduction in the time to gastrointestinal tract recovery and hospital discharge.53 A 12-mg dose was more beneficial than a 6-mg dose, especially in females and in older patients (over age 65).
Most recently, a multicenter, double-blind, placebo-controlled trial evaluated alvimopan as part of a standardized postoperative care plan in 654 patients undergoing partial small-bowel and large-bowel resection.54 The alvimopan group took less time to have their first bowel movements, pass flatus, and tolerate solid food. Patients randomized to alvimopan also had their discharge orders written an average of 1 day sooner than the placebo group. Importantly, opioid use was the same in both groups.
Alvimopan is given as a single oral dose of 12 mg 30 to 90 minutes before surgery and twice daily after surgery for up to 7 days, for a total of 15 doses. It is contraindicated in patients receiving therapeutic doses of opiates for more than 7 consecutive days immediately before surgery. Its use is currently limited to hospitals enrolled in the EASE (Entereg Access Support and Education) program.
Common adverse effects include constipation, dyspepsia, flatulence, and urinary retention. In a placebo-controlled 12-month study in patients treated with opiates for chronic pain, there were more reports of myocardial infarction in the alvimopan group.55 This finding has not been replicated in any other study. The need to give the drug preoperatively obviously necessitates identifying patients most at risk of postoperative ileus.
FUTURE DIRECTIONS
A multimodal approach to managing postoperative ileus seems likely to be the most effective model in the long run. This should involve using minimally invasive surgery when possible, pharmacotherapy, and accelerated standardized postoperative care.
Standardized postoperative care has been implemented for a variety of procedures and generally involves minimal (if any) use of nasogastric tubes, early enteral intake and ambulation, and specific discharge criteria such as passage of flatus or stool, adequate pain control, and tolerance of solid food.56–58 Compared with a “traditional” (nonstandardized) approach, standardized care has led to shorter hospital stays and lower costs with no impact on rates of morbidity or readmission.59,60 (However, one clearly cannot underestimate the role of patient expectations in the success of such postoperative care pathways.)
There are plenty of incentives for patients, physicians, health care organizations, and third-party payers to support this push. For patients, it means less time in the hospital and a quicker return to eating normally. Surgeons can expect more-satisfied patients and lower rates of hospital-acquired conditions. For hospitals and insurers, it means less use of resources for some patients, making resources available to those who need them more.
Rather than meerely wait for bowel sounds to return after patients undergo surgery, we can try to get the gut working again sooner. An active approach might shorten the duration of postoperative ileus, allow patients to go home from the hospital sooner, and improve their outcomes.
In the pages that follow, we review the pathophysiology, diagnosis, and current therapies to alter the course of postoperative ileus.
ILEUS CAN AFFECT THE STOMACH OR SMALL OR LARGE INTESTINES
Ileus is the absence of intestinal peristalsis without mechanical obstruction; postoperative ileus refers to the time after surgery before coordinated electromotor bowel function resumes.
Gastroparesis refers to abnormal gastric motility leading to impaired gastric emptying. This disabling, potentially chronic condition is associated with certain medical conditions such as diabetes, but can also occur after some surgical procedures, as we will discuss. It has been estimated to affect approximately 4% of the adult population, with a strong female predilection.1 Postoperative gastroparesis is probably most common after pancreaticoduodenectomy, in which it occurs in up to 57% of patients.2 Consensus guidelines for grading the severity of gastroparesis have been devised to help standardize the reporting of outcomes.2
Acute colonic pseudo-obstruction (colonic ileus) is often seen in elderly hospitalized patients with multiple medical comorbidities. Of note, it often occurs after surgery to parts of the body other than the abdomen, such as after orthopedic procedures. One study documented an incidence of 1.3% after hip replacement surgery and 1.2% after spine procedures.3
The small bowel normally resumes activity several hours after surgery, the stomach 24 to 48 hours after surgery, and the colon 3 to 5 days after surgery.4 When postoperative ileus persists longer than this, it can be considered pathologic and is sometimes called paralytic ileus.4,5
ILEUS AFFECTS OUTCOMES AND COSTS
Although not usually considered life-threatening, postoperative ileus is harmful for the patient and costly for society.
For the patient, ileus is uncomfortable, leads to nausea and vomiting, delays return to enteral nutrition, and prolongs the stay in the hospital. For many if not most patients undergoing gastrointestinal surgery, return of bowel function is the factor that delays going home. A prolonged hospital stay increases the risk of hospital-acquired infections, deep vein thrombosis, and other conditions.
The economic burden is also considerable. A retrospective review of more than 800,000 patients who underwent surgery in the United States in 2002 found a rate of postoperative ileus of 4.25% according to International Classification of Diseases–Ninth Revision (ICD-9) codes.6 The mean hospital length of stay was 9.3 days in patients with postoperative ileus vs 5.3 days in those without it. The difference in mean total hospital costs was US $6,300 per patient. The costs certainly add up when you consider the number of surgical procedures performed every year.
NEURAL AND CHEMICAL FACTORS
While observing exteriorized bowel in 1872, Goltz7 first noted enhanced spontaneous contractions when the spinal cord was severed at the level of the medulla. Not long after, Bayliss and Starling8 used a device called an “enterograph” to monitor small-bowel activity in vivo in dogs and found that cutting the splanchnic nerves led to vigorous bowel contraction after laparotomy. These early observations formed the foundation of our understanding of postoperative ileus and some of its possible causes.
Normal bowel contractility is influenced by a host of neural and chemical factors, the relative contributions of which vary depending on the segment of bowel.
The migrating motor complex is the basal level of activity in the bowel in the fasting state, serving a “housekeeping” function.9 It has four phases, consisting of escalating electrical and contractile activity punctuated by periods of quiescence. The resumption of this motor complex after surgery is responsible for recovery from postoperative ileus.
Sympathetic-parasympathetic imbalance
The sympathetic nervous system inhibits the small bowel; the parasympathetic nervous system stimulates it. Although vagal (parasympathetic) stimulation appears to have little actual impact on small-bowel activity, if sympathetic activity is blocked, contractility increases, indicating that tonic sympathetic inhibition normally predominates. The balance of these two competing influences determines the amount of acetylcholine released by excitatory nerve fibers in the myenteric plexi of the bowel.
These neural pathways can be manipulated clinically. Epidural catheters can block sympathetic output, thus allowing small-bowel function to return faster.
Vagus nerve activity appears to be more important in the stomach, where it promotes receptive relaxation of the fundus and contraction of the antrum, facilitating gastric emptying.10 After vagotomy, emptying of liquids may be normal or accelerated, but emptying of solids is impaired. This can occur after peptic ulcer surgery but is more likely after gastric resection for malignancy or after inadvertent vagal nerve injury during antireflux surgery.
The enteric nervous system is a complex, intrinsic network of neurons consisting of two distinct plexi within the bowel wall: the submucosal (Meissner) plexus, and the myenteric (Auerbach) plexus.11 The enteric nervous system in the small bowel is fundamentally different than the one in the colon in that the former contains gap junctions, allowing for coordinated electrical activity. Lacking these gap junctions, the colon depends more on input from the autonomic nervous system, perhaps explaining the longer recovery from postoperative ileus and the susceptibility to isolated colonic ileus due to a variety stressors and traumatic insults.12
Chemical mediators of bowel activity
A host of chemical mediators influence bowel motility. Perhaps the most important nonadrenergic inhibitor of gastrointestinal motility is nitric oxide.13 Animal studies have firmly established nitric oxide as an important factor in postoperative ileus, but its exact role in humans is not clear.14,15
Other mediators with possible roles include vasoactive intestinal peptide, substance P, calcitonin gene-related peptide, and endogenous opioids.13 Lack of duodenal-derived motilin is thought to be one cause of delayed gastric emptying after pancreaticoduodenectomy.2
Inflammation
The inflammatory response after surgery has also been an attractive target of study of the factors promoting postoperative ileus. In rat studies, Kalff et al16 found that surgical manipulation of the bowel induced an inflammatory cellular infiltrate in the bowel wall and diminished the response of smooth muscle to cholinergic stimulation. Cyclooxygenase-2, the enzymatic precursor to prostaglandins, has also been shown to be induced in enteric neurons after laparotomy.17
Narcotic analgesics
One of the greatest hurdles in preventing postoperative ileus is the use of narcotic analgesics to treat postoperative pain. It is also one of the most important modifiable risk factors.
Opiates delay colonic transit in postoperative patients, an effect that can be reversed by the narcotic antagonist naloxone (Narcan).18 This inhibitory effect is mediated by peripheral mu-opioid receptors. In a study of patients undergoing colectomy, the more morphine given, the longer the time to the return of bowel sounds and flatus and the first bowel movement.19
These observations have led to a search for selective opiate antagonists that allow narcotics to continue relieving pain while counteracting their effect on bowel motility, a topic discussed later in this review.
Nonsteroidal anti-inflammatory drugs such as ketorolac (Toradol) are attractive alternatives to opiate analgesics, both for their anti-inflammatory effect and for their opiatesparing properties. However, they can cause bleeding, renal insufficiency, and gastritis, drawbacks that limit their applicability and duration of use.
DIAGNOSIS BY CLINICAL SUSPICION AND IMAGING
The diagnosis of postoperative ileus is driven by a combination of clinical suspicion and imaging tests.
Regardless of the segment of bowel involved, it is imperative to exclude an obstructive cause. The diagnosis of ileus is presumed once obstruction has been excluded.
Diagnosing gastroparesis
Postoperative gastroparesis is usually suspected by its symptoms of early satiety, nausea, vomiting, eructation, and gastroesophageal reflux. Abdominal distention is usually not a prominent sign, but a succussion splash may be detected, indicating retention of food and liquid in the stomach.
Plain radiographs may reveal a large gastric air bubble in the left upper quadrant but may underestimate the degree of gastric distention. Computed tomography (CT) may show a large, fluid-filled stomach, often containing high-density food debris.
The gold standard for diagnosis is gastric emptying scintigraphy after a radiolabelled solid meal. The patient consumes a meal of egg white labelled with technetium 99m sulfur colloid, and scanning is performed at specified intervals to measure the percent retention of the isotope. Retention of more than 10% at 4 hours is considered abnormal.1 Severity can be graded on the basis of percent retention after 4 hours.20
This test is rarely indicated in the acute postoperative setting, however, and patients should be treated presumptively to prevent aspiration once mechanical obstruction is excluded.
Diagnosing small-bowel ileus
Small-bowel ileus often presents like gastroparesis, except that it more often causes abdominal distention. Plain radiographs reveal air-fluid levels and dilated loops of bowel.
Small-bowel ileus must then be differentiated from small-bowel obstruction by clinical and radiographic features. The presence of crampy abdominal pain, bowel sounds, and some bowel function implies a degree of mechanical obstruction. Plain radiographs showing “step-ladder” air-fluid levels also suggest obstruction. CT is more definitive in diagnosing obstruction by the presence of distended and decompressed bowel loops and may also reveal a source of obstruction (eg, postoperative interloop abscess).
Diagnosing colonic ileus
Colonic ileus is also characterized by abdominal distention, sometimes marked. Although it is the colon that is primarily involved, upstream small-bowel dilatation can also be seen if the ileocecal valve is incompetent. The cecum often shows the greatest degree of dilatation on plain radiographs and is at the greatest risk of perforation. CT, contrast enema studies, and endoscopy help rule out mechanical obstruction due to volvulus or a mass lesion.
STRATEGIES TO PREVENT AND TREAT ILEUS
Many strategies have been applied to prevent and manage postoperative ileus, ranging from changes in surgical technique, supportive care, and patient-initiated activities, to pharmacologic intervention.
Epidural anesthesia shortens ileus, reduces the need for narcotics
Epidural anesthesia has shown promise not only in improving pain control, but also in shortening the period of postoperative ileus. Most surgical patients either receive an epidural catheter before surgery, which is left in place for postoperative pain control, or are given patient-controlled analgesia with a narcotic. Generally, the surgeon chooses the pain control method.
Thoracic epidural analgesia has been shown to hasten the return of bowel function by 1 to 2 days and to reduce the need for opiates compared with systemic opioids alone.21–26 A likely explanation is that epidural anesthesia interferes with the afferent and efferent sympathetic reflex arcs. The level of the epidural placement is important: a thoracic epidural is needed to effectively block these sympathetic pathways.
Laparoscopic surgery is less traumatic
Laparoscopy has changed the landscape of surgery over the past few decades. Some of the most common surgical procedures (appendectomy, cholecystectomy) are now done mainly via the laparoscope, as are many procedures that are more complex.
Laparoscopic surgery has several advantages over open surgery. With smaller incisions, it is less traumatic to the body. The systemic inflammatory response appears to be less vigorous after laparoscopic surgery than after open surgery, as measured by circulating levels of interleukin 1, interleukin 6, and C-reactive protein.27
The length of stay after a laparoscopic procedure is shorter than after an open procedure for several reasons, not the least of which is a shorter duration of postoperative ileus. Animal studies show that intestinal recovery is faster after laparoscopic procedures than after open procedures.28–30 In a study in which their other care was comparable, significantly fewer patients undergoing laparoscopic colectomy had emesis or needed their nasogastric tube to be reinserted than patients who underwent an open operation, and their length of stay was shorter.31
As technology continues to advance in minimally invasive surgery, it is reasonable to expect these trends to continue.
Nasogastric tubes in selected cases
Patients are often allowed nothing by mouth or only minimal oral intake immediately after abdominal surgery, with or without nasogastric decompression. The role of nasogastric decompression has long been a topic of controversy. In a meta-analysis of 26 trials with 3,964 patients, the groups in which all patients routinely received a nasogastric tube had higher rates of pneumonia, fever, and atelectasis and longer duration to resumption of oral feeding than the groups in which nasogastric tubes were used selectively.32
Most clinicians agree that nasogastric tubes are uncomfortable and do little to prevent postoperative ileus. However, in selected cases they are useful for managing intractable vomiting and for preventing aspiration of gastric contents.
Early enteral feeding
Evidence is mounting that early postoperative enteral feeding may be advantageous for recovery.
In 1,173 patients undergoing both upper and lower gastrointestinal surgery in 13 trials, fewer patients died who were randomized to receive enteral feeding within 24 hours.33 There were also fewer infectious complications and anastomotic problems and a shorter length of stay, but these differences were not statistically significant. Vomiting was more common in the early-feeding groups but did not lead to higher rates of morbidity. Enteral feeding was by the oral, nasoduodenal, or nasojejunal routes, depending on the type of surgery performed.
Whether the number of calories given affects the outcome remains to be clarified, but at least for now it seems that feeding patients early in the course of their recovery is not detrimental and may in fact be beneficial.
Gum-chewing
Gum-chewing has been studied over the last decade as a form of sham feeding to stimulate bowel recovery after surgery. The presumed mechanism of action is vagal cholinergic (parasympathetic) stimulation of the gastrointestinal tract, similar to oral intake but with theoretically less risk of vomiting and aspiration.
In five such trials in patients undergoing colon resection, gum-chewing shortened the time until first flatus and bowel movement, but made no significant difference in length of stay.34
At the very least, gum-chewing immediately after surgery is a cheap and harmless strategy for reducing postoperative ileus, and it might make the patient more comfortable.
DRUGS THAT COAX THE GUT BACK TO WORK
Drugs that coax the gastrointestinal tract back to work have been tried for many years and have recently gained renewed enthusiasm. Their efficacy varies according to their target organ, with greater success in the stomach and colon than in the small bowel.
Cisapride (Propulsid) was an effective gastric prokinetic agent, as shown in several controlled trials. However, it was withdrawn from the US market in 2000 because of its propensity to cause cardiac arrhythmias.
Erythromycin is a macrolide antibiotic that is also a motilin receptor agonist. In patients who underwent antrectomy and vagotomy, it was shown to accelerate gastric emptying by roughly 40% as measured by solid-phase gastric emptying scintigraphy.35,36 In a randomized controlled trial in 118 patients who underwent pancreaticoduodenectomy, intravenous erythromycin reduced gastroparesis by 37% (measured by solid-phase gastric emptying study) and also reduced the need for nasogastric tube reinsertion.37 A major shortcoming is the development of tachyphylaxis, thought to be mediated by down-regulation of motilin receptors.
Metoclopramide (Reglan) is an antiemetic and prokinetic that acts as a dopamine D2 receptor antagonist and mixed serotonin 5-HT3 antagonist/5-HT4 agonist. Metoclopramide also stimulates gastric emptying, as shown in controlled trials in patients in intensive care units.38,39 The drug should not be used in patients with parkinsonism, in view of its antidopamine properties.
In 2009, the US Food and Drug Administration required that a black box warning be added to metoclopramide because of the risk of tardive dyskinesia with long-term use, and recommended that its use be limited to 3 weeks in the acute setting.40 Prescribers and patients need to decide if this risk is worth the potential benefit on a case-by-case basis.
Although erythromycin and metoclopramide are effective in managing gastroparesis, neither has been shown to be effective for small-bowel ileus.41,42 However, colonic ileus is highly responsive to drug therapy.
Neostigmine (Prostigmin) is a reversible acetylcholinesterase inhibitor that enhances the activity of the neurotransmitter acetylcholine at muscarinic receptors. It is the first-line treatment for colonic ileus.43 In three randomized, placebo-controlled trials,44–46 the success rates were 85% to 94% after the first dose.
Neostigmine is generally given either as an intravenous bolus dose of 2 to 2.5 mg or as an intravenous infusion over 24 hours. It must be given in a monitored setting, as both bradycardia and bronchospasm can occur. Patients should continue to be monitored clinically and with plain abdominal radiography after the drug is given, and they sometimes require a second or third dose.
In cases in which neostigmine fails, decompressive colonoscopy can be done as a second-line measure.
Alvimopan (Entereg), a peripherally acting, mu-opioid receptor antagonist, has come on the scene most recently. This agent first showed promise when it precipitated diarrhea in morphine-dependent mice.47 Early studies in humans focused on its ability to reverse the effect of opiates on gastrointestinal transit without interfering with their analgesic properties.48–50 Later investigations concentrated on its ability to reduce the duration of postoperative ileus after a variety of major abdominal surgical procedures.51,52
A pooled analysis of phase III studies of alvimopan focused on the subset of 1,212 patients who underwent bowel resections; it found a significant reduction in the time to gastrointestinal tract recovery and hospital discharge.53 A 12-mg dose was more beneficial than a 6-mg dose, especially in females and in older patients (over age 65).
Most recently, a multicenter, double-blind, placebo-controlled trial evaluated alvimopan as part of a standardized postoperative care plan in 654 patients undergoing partial small-bowel and large-bowel resection.54 The alvimopan group took less time to have their first bowel movements, pass flatus, and tolerate solid food. Patients randomized to alvimopan also had their discharge orders written an average of 1 day sooner than the placebo group. Importantly, opioid use was the same in both groups.
Alvimopan is given as a single oral dose of 12 mg 30 to 90 minutes before surgery and twice daily after surgery for up to 7 days, for a total of 15 doses. It is contraindicated in patients receiving therapeutic doses of opiates for more than 7 consecutive days immediately before surgery. Its use is currently limited to hospitals enrolled in the EASE (Entereg Access Support and Education) program.
Common adverse effects include constipation, dyspepsia, flatulence, and urinary retention. In a placebo-controlled 12-month study in patients treated with opiates for chronic pain, there were more reports of myocardial infarction in the alvimopan group.55 This finding has not been replicated in any other study. The need to give the drug preoperatively obviously necessitates identifying patients most at risk of postoperative ileus.
FUTURE DIRECTIONS
A multimodal approach to managing postoperative ileus seems likely to be the most effective model in the long run. This should involve using minimally invasive surgery when possible, pharmacotherapy, and accelerated standardized postoperative care.
Standardized postoperative care has been implemented for a variety of procedures and generally involves minimal (if any) use of nasogastric tubes, early enteral intake and ambulation, and specific discharge criteria such as passage of flatus or stool, adequate pain control, and tolerance of solid food.56–58 Compared with a “traditional” (nonstandardized) approach, standardized care has led to shorter hospital stays and lower costs with no impact on rates of morbidity or readmission.59,60 (However, one clearly cannot underestimate the role of patient expectations in the success of such postoperative care pathways.)
There are plenty of incentives for patients, physicians, health care organizations, and third-party payers to support this push. For patients, it means less time in the hospital and a quicker return to eating normally. Surgeons can expect more-satisfied patients and lower rates of hospital-acquired conditions. For hospitals and insurers, it means less use of resources for some patients, making resources available to those who need them more.
- Waseem S, Moshiree B, Draganov P. Gastroparesis: current diagnostic challenges and management considerations. World J Gastroenterol 2009; 15:25–37.
- Wente MN, Bassi C, Dervenis C, et al. Delayed gastric emptying (DGE) after pancreatic surgery: a suggested definition by the International Study Group of Pancreatic Surgery (ISGPS). Surgery 2007; 142:761–768.
- Norwood MG, Lykostratis H, Garcea G, Berry DP. Acute colonic pseudo-obstruction following major orthopaedic surgery. Colorectal Dis 2005; 7:496–499.
- Livingston EH, Passaro EP. Postoperative ileus. Dig Dis Sci 1990; 35:121–132.
- Catchpole BN. Ileus: use of sympathetic blocking agents in its treatment. Surgery 1969; 66:811–820.
- Saunders WB, Bowers B, Moss B, et al. Recorded rate and economic burden associated with postoperative ileus [abstract]. Presented at the ASHP Midyear Clinical Meeting, Orlando, FL, December 7, 2004. Abstract 30346.
- Neely J, Catchpole BN. The restoration of alimentary-tract motility by pharmacological means. Br J Surg 1971; 58:21–28.
- Bayliss WM, Starling EH. The movements and innervations of the small intestine. J Physiol 1899; 24:99–143.
- Szurszewksi JH. A migrating electrical complex of the canine small intestine. Am J Physiol 1969; 217:1757–1763.
- Livingston EH. Stomach and duodenum. In:Norton JA, Bollinger RR, Chang AE, et al (editors). Surgery: Basic Science and Clinical Evidence. New York, Springer-Verlag, 2001:489–516.
- Simeone DM. Anatomy and physiology of the small intestine. In:Mulholland MW, Lillemoe KD, Doherty GM, Maier RV, Upchurch GR, editors. Greenfield’s Surgery: Scientific Principles and Practice, 4th ed. Philadelphia: Lippincott Williams and Wilkins, 2006:756–766.
- Baig MK, Wexner SD. Postoperative ileus: a review. Dis Colon Rectum 2004; 47:516–526. Retraction in Dis Colon Rectum 2005; 48:1983.
- Luckey A, Livingston E, Taché Y. Mechanisms and treatment of postoperative ileus. Arch Surg 2003; 138:206–214.
- De Winter BY, Boeckxstaens GE, De Man JG, Moreels TG, Herman AG, Pelckmans PA. Effect of adrenergic and nitrergic blockade on experimental ileus in rats. Br J Pharmacol 1997; 120:464–468.
- Kalff JC, Schraut WH, Billiar TR, Simmons RL, Bauer AJ. Role of inducible nitric oxide synthase in postoperative intestinal smooth muscle dysfunction in rodents. Gastroenterology 2000; 118:316–327.
- Kalff JC, Schraut WH, Simmons RL, Bauer AJ. Surgical manipulation of the gut elicits an intestinal muscularis inflammatory response resulting in postsurgical ileus. Ann Surg 1998; 228:652–663.
- Schwarz NT, Kalff JC, Türler A, et al. Prostanoid production via COX-2 as a causative mechanism of rodent postoperative ileus. Gastroenterology 2001; 121:1354–1371.
- Kaufman PN, Krevsky B, Malmud LS, et al. Role of opiate receptors in the regulation of colonic transit. Gastroenterology 1988; 94:1351–1356.
- Cali RL, Meade PG, Swanson MS, Freeman C. Effect of morphine and incision length on bowel function after colectomy. Dis Colon Rectum 2000; 43:163–168.
- Abell TL, Camilleri M, Donahoe K, et al; American Neurogastroenterology and Motility Society and the Society of Nuclear Medicine. Consensus recommendations for gastric emptying scintigraphy: a joint report of the American Neurogastroenterology and Motility Society and the Society of Nuclear Medicine. Am J Gastroenterol 2008; 103:753–763.
- Zingg U, Miskovic D, Hamel CT, Erni L, Oertli D, Metzger U. Influence of thoracic epidural analgesia on postoperative pain relief and ileus after laparoscopic colorectal resection: benefit with epidural analgesia. Surg Endosc 2009; 23:276–282.
- Taqi A, Hong X, Mistraletti G, Stein B, Charlebois P, Carli F. Thoracic epidural analgesia facilitates the restoration of bowel function and dietary intake in patients undergoing laparoscopic colon resection using a traditional, nonaccelerated, perioperative care program. Surg Endosc 2007; 21:247–252.
- Kuo CP, Jao SW, Chen KM, et al. Comparison of the effects of thoracic epidural analgesia and i.v. infusion with lidocaine on cytokine response, postoperative pain, and bowel function in patients undergoing colonic surgery. Br J Anaesth 2006; 97:640–646.
- Carli F, Trudel JL, Belliveau P. The effect of intraoperative thoracic epidural anesthesia and postoperative analgesia on bowel function after colorectal surgery: a prospective, randomized trial. Dis Colon Rectum 2001; 44:1083–1089.
- Jørgensen H, Wetterslev J, Møiniche S, Dahl JB. Epidural local anaesthetics versus opioid-based analgesic regimens on postoperative gastrointestinal paralysis, PONV and pain after abdominal surgery. Cochrane Database Syst Rev 2000; 4:CD001893.
- de Leon-Casasola OA, Karabella D, Lema MJ. Bowel function recovery after radical hysterectomies: thoracic epidural bupivacainemorphine versus intravenous patient-controlled analgesia with morphine: a pilot study. J Clin Anesth 1996; 8:87–92.
- Leung KL, Lai PB, Ho RL, et al. Systemic cytokine response after laparoscopic-assisted resection of rectosigmoid carcinoma: a prospective randomized trial. Ann Surg 2000; 231:506–511.
- Böhm B, Milsom JW, Fazio VW. Postoperative intestinal motility following conventional and laparoscopic intestinal surgery. Arch Surg 1995; 130:415–419.
- Davies W, Kollmorgen CF, Tu QM, et al. Laparoscopic colectomy shortens postoperative ileus in a canine model. Surgery 1997; 121:550–555.
- Hotokezaka M, Combs MJ, Mentis EP, Schirmer BD. Recovery of fasted and fed gastrointestinal motility after open versus laparoscopic cholecystectomy in dogs. Ann Surg 1996; 223:413–419.
- Chen HH, Wexner SD, Iroatulam AJ, et al. Laparoscopic colectomy compares favorably with colectomy by laparotomy for reduction of postoperative ileus. Dis Colon Rectum 2000; 43:61–65.
- Cheatham ML, Chapman WC, Key SP, Sawyers JL. A meta-analysis of selective versus routine nasogastric decompression after elective laparotomy. Ann Surg 1995; 221:469–478.
- Lewis SJ, Andersen HK, Thomas S. Early enteral nutrition within 24 h of intestinal surgery versus later commencement of feeding: a systematic review and meta-analysis. J Gastrointest Surg 2009; 13:569–575.
- Purkayastha S, Tilney HS, Darzy AW, Tekkis PP. Meta-analysis of studies evaluating chewing gum to enhance postoperative recovery following colectomy. Arch Surg 2008; 143:788–793.
- Ramirez B, Eaker EY, Drane WE, Hocking MP, Sninsky CA. Erythromycin enhances gastric emptying in patients with gastroparesis after vagotomy and antrectomy. Dig Dis Sci 1994; 39:2295–2300.
- Kendall BJ, Chakravarti A, Kendall E, Soykan I, McCallum RW. The effect of intravenous erythromycin on solid meal gastric emptying in patients with chronic symptomatic post-vagotomy-antrectomy gastroparesis. Aliment Pharmacol Ther 1997; 11:381–385.
- Yeo CJ, Barry MK, Sauter PK, et al. Erythromycin accelerates gastric emptying after pancreaticoduodenectomy. A prospective, randomized, placebo-controlled trial. Ann Surg 1993; 218:229–237.
- Jooste CA, Mustoe J, Collee G. Metoclopramide improves gastric motility in critically ill patients. Intensive Care Med 1999; 25:464–468.
- Sustic A, Zelic M, Protic A, et al. Metoclopramide improves gastric but not gallbladder emptying in cardiac surgery patients with early intragastric enteral feeding: randomized controlled trial. Croat Med J 2005; 46:239–244.
- US Food and Drug Administration. FDA requires boxed warning and risk mitigation strategy for metoclopramide-containing drugs. Agency warns against chronic use of these products to treat gastrointestinal disorders. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm149533.htm. Accessed 8/24/2009.
- Smith AJ, Nissan A, Lanouette NM, et al. Prokinetic affect of erythromycin after colorectal surgery: a randomized, placebo-controlled, double-blind study. Dis Colon Rectum 2000; 43:333–337.
- Cheape JD, Wexner SD, Jagelman JK. Does metoclopramide reduce the length of ileus after colorectal surgery? A prospective randomized trial. Dis Colon Rectum 1991; 34:437–441.
- De Giorgio R, Knowles CH. Acute colonic pseudo-obstruction. Br J Surg 2009; 96:229–239.
- Ponec RJ, Saunders MD, Kimmey MB. Neostigmine for the treatment of acute colonic pseudo-obstruction. N Engl J Med 1999; 341:137–141.
- Amaro R, Rogers AI. Neostigmine infusions: new standard of care for acute colonic pseudo-obstruction? Am J Gastroenterol 2000; 95:304–305.
- van der Spoel JI, Oudemans-van Straaten HM, Stroutenbeek CP, Bosman RJ, Zandstra DF. Neostigmine resolves critical illness-related colonic ileus in intensive care patients with multiple organ failure: a prospective, double-blind, placebo-controlled trial. Intensive Care Med 2001; 27:822–827.
- Zimmerman DM, Gidda JS, Cantrell BE, et al. LY246736 dihydrate μ-opioid receptor antagonist. Drugs Future 1994; 19:1078–1083.
- Callaghan JT, Cerimele B, Nowak TV, et al. Effect of the opioid antagonist LY246736 on gastrointestinal transit in human subjects [abstract]. Gastroenterology 1998; 114:G3015.
- Hodgson PS, Liu SS, Carpenter RI. ADL 8-2698 prevents morphine inhibition of GI transit [abstract]. Clin Pharmacol Ther 2000; 67:93.
- Liu SS, Hodgson PS, Carpenter RL, Fricke JR. ADL 8-2698, a trans-3,4-dimethyl-4-(3-hydroxyphenyl) piperidine, prevents gastrointestinal effects of intravenous morphine without affecting analgesia. Clin Pharmacol Ther 2001; 68:66–71.
- Wolff BG, Michelassi F, Gerkin TM, et al. Alvimopan, a novel, peripherally acting mu opioid antagonist: results of a multicenter, randomized, double-blind, placebo-controlled, phase III trial of major abdominal surgery and postoperative ileus. Ann Surg 2004; 240:728–735.
- Delaney CP, Weese JL, Hyman NH, et al; Alvimopan Postoperative Ileus Study Group. Phase III trial of alvimopan, a novel, peripherally acting, mu opioid antagonist, for postoperative ileus after major abdominal surgery. Dis Colon Rectum 2005; 48:1114–1129.
- Delaney CP, Wolff BG, Viscusi ER, et al. Alvimopan, for postoperative ileus following bowel resection: a pooled analysis of phase III studies. Ann Surg 2007; 245:355–363.
- Ludwig K, Enker WE, Delaney CP, et al. Gastrointestinal tract recovery in patients undergoing bowel resection: results of a randomized trial of alvimopan and placebo with a standardized accelerated postoperative care pathway. Arch Surg 2008; 143:1098–1105.
- Adolor GlaxoSmithKline. Entereg (alvimopan). www.entereg.com/Accessed 8/24/2009.
- Pritts TA, Nussbaum MS, Flesch LV, Fegelman EJ, Parikh AA, Fischer JE. Implementation of a clinical pathway decreases length of stay and cost for bowel resection. Ann Surg 1999; 230:728–733.
- Delaney CP, Zutshi M, Senagore AJ, Remzi FH, Hammel J, Fazio VW. Prospective, randomized, controlled trial between a pathway of controlled rehabilitation with early ambulation and diet and traditional postoperative care after laparotomy and intestinal resection. Dis Colon Rectum 2003; 46:851–859.
- Joh YG, Lindsetmo RO, Stulberg J, Obias V, Champagne B, Delaney CP. Standardized postoperative pathway: accelerating recovery after ileostomy closure. Dis Colon Rectum 2008; 51:1786–1789.
- Kennedy EP, Rosato EL, Sauter PK, et al. Initiation of a critical pathway for pancreaticoduodenectomy at an academic institution—the first step in multidisciplinary team building. J Am Coll Surg 2007; 204:917–924.
- Kariv Y, Delaney CP, Senagore AJ, et al. Clinical outcomes and cost analysis of a “fast track” postoperative care pathway for ileal pouch-anal anastomosis: a case control study. Dis Colon Rectum 2007; 50:137–146.
- Waseem S, Moshiree B, Draganov P. Gastroparesis: current diagnostic challenges and management considerations. World J Gastroenterol 2009; 15:25–37.
- Wente MN, Bassi C, Dervenis C, et al. Delayed gastric emptying (DGE) after pancreatic surgery: a suggested definition by the International Study Group of Pancreatic Surgery (ISGPS). Surgery 2007; 142:761–768.
- Norwood MG, Lykostratis H, Garcea G, Berry DP. Acute colonic pseudo-obstruction following major orthopaedic surgery. Colorectal Dis 2005; 7:496–499.
- Livingston EH, Passaro EP. Postoperative ileus. Dig Dis Sci 1990; 35:121–132.
- Catchpole BN. Ileus: use of sympathetic blocking agents in its treatment. Surgery 1969; 66:811–820.
- Saunders WB, Bowers B, Moss B, et al. Recorded rate and economic burden associated with postoperative ileus [abstract]. Presented at the ASHP Midyear Clinical Meeting, Orlando, FL, December 7, 2004. Abstract 30346.
- Neely J, Catchpole BN. The restoration of alimentary-tract motility by pharmacological means. Br J Surg 1971; 58:21–28.
- Bayliss WM, Starling EH. The movements and innervations of the small intestine. J Physiol 1899; 24:99–143.
- Szurszewksi JH. A migrating electrical complex of the canine small intestine. Am J Physiol 1969; 217:1757–1763.
- Livingston EH. Stomach and duodenum. In:Norton JA, Bollinger RR, Chang AE, et al (editors). Surgery: Basic Science and Clinical Evidence. New York, Springer-Verlag, 2001:489–516.
- Simeone DM. Anatomy and physiology of the small intestine. In:Mulholland MW, Lillemoe KD, Doherty GM, Maier RV, Upchurch GR, editors. Greenfield’s Surgery: Scientific Principles and Practice, 4th ed. Philadelphia: Lippincott Williams and Wilkins, 2006:756–766.
- Baig MK, Wexner SD. Postoperative ileus: a review. Dis Colon Rectum 2004; 47:516–526. Retraction in Dis Colon Rectum 2005; 48:1983.
- Luckey A, Livingston E, Taché Y. Mechanisms and treatment of postoperative ileus. Arch Surg 2003; 138:206–214.
- De Winter BY, Boeckxstaens GE, De Man JG, Moreels TG, Herman AG, Pelckmans PA. Effect of adrenergic and nitrergic blockade on experimental ileus in rats. Br J Pharmacol 1997; 120:464–468.
- Kalff JC, Schraut WH, Billiar TR, Simmons RL, Bauer AJ. Role of inducible nitric oxide synthase in postoperative intestinal smooth muscle dysfunction in rodents. Gastroenterology 2000; 118:316–327.
- Kalff JC, Schraut WH, Simmons RL, Bauer AJ. Surgical manipulation of the gut elicits an intestinal muscularis inflammatory response resulting in postsurgical ileus. Ann Surg 1998; 228:652–663.
- Schwarz NT, Kalff JC, Türler A, et al. Prostanoid production via COX-2 as a causative mechanism of rodent postoperative ileus. Gastroenterology 2001; 121:1354–1371.
- Kaufman PN, Krevsky B, Malmud LS, et al. Role of opiate receptors in the regulation of colonic transit. Gastroenterology 1988; 94:1351–1356.
- Cali RL, Meade PG, Swanson MS, Freeman C. Effect of morphine and incision length on bowel function after colectomy. Dis Colon Rectum 2000; 43:163–168.
- Abell TL, Camilleri M, Donahoe K, et al; American Neurogastroenterology and Motility Society and the Society of Nuclear Medicine. Consensus recommendations for gastric emptying scintigraphy: a joint report of the American Neurogastroenterology and Motility Society and the Society of Nuclear Medicine. Am J Gastroenterol 2008; 103:753–763.
- Zingg U, Miskovic D, Hamel CT, Erni L, Oertli D, Metzger U. Influence of thoracic epidural analgesia on postoperative pain relief and ileus after laparoscopic colorectal resection: benefit with epidural analgesia. Surg Endosc 2009; 23:276–282.
- Taqi A, Hong X, Mistraletti G, Stein B, Charlebois P, Carli F. Thoracic epidural analgesia facilitates the restoration of bowel function and dietary intake in patients undergoing laparoscopic colon resection using a traditional, nonaccelerated, perioperative care program. Surg Endosc 2007; 21:247–252.
- Kuo CP, Jao SW, Chen KM, et al. Comparison of the effects of thoracic epidural analgesia and i.v. infusion with lidocaine on cytokine response, postoperative pain, and bowel function in patients undergoing colonic surgery. Br J Anaesth 2006; 97:640–646.
- Carli F, Trudel JL, Belliveau P. The effect of intraoperative thoracic epidural anesthesia and postoperative analgesia on bowel function after colorectal surgery: a prospective, randomized trial. Dis Colon Rectum 2001; 44:1083–1089.
- Jørgensen H, Wetterslev J, Møiniche S, Dahl JB. Epidural local anaesthetics versus opioid-based analgesic regimens on postoperative gastrointestinal paralysis, PONV and pain after abdominal surgery. Cochrane Database Syst Rev 2000; 4:CD001893.
- de Leon-Casasola OA, Karabella D, Lema MJ. Bowel function recovery after radical hysterectomies: thoracic epidural bupivacainemorphine versus intravenous patient-controlled analgesia with morphine: a pilot study. J Clin Anesth 1996; 8:87–92.
- Leung KL, Lai PB, Ho RL, et al. Systemic cytokine response after laparoscopic-assisted resection of rectosigmoid carcinoma: a prospective randomized trial. Ann Surg 2000; 231:506–511.
- Böhm B, Milsom JW, Fazio VW. Postoperative intestinal motility following conventional and laparoscopic intestinal surgery. Arch Surg 1995; 130:415–419.
- Davies W, Kollmorgen CF, Tu QM, et al. Laparoscopic colectomy shortens postoperative ileus in a canine model. Surgery 1997; 121:550–555.
- Hotokezaka M, Combs MJ, Mentis EP, Schirmer BD. Recovery of fasted and fed gastrointestinal motility after open versus laparoscopic cholecystectomy in dogs. Ann Surg 1996; 223:413–419.
- Chen HH, Wexner SD, Iroatulam AJ, et al. Laparoscopic colectomy compares favorably with colectomy by laparotomy for reduction of postoperative ileus. Dis Colon Rectum 2000; 43:61–65.
- Cheatham ML, Chapman WC, Key SP, Sawyers JL. A meta-analysis of selective versus routine nasogastric decompression after elective laparotomy. Ann Surg 1995; 221:469–478.
- Lewis SJ, Andersen HK, Thomas S. Early enteral nutrition within 24 h of intestinal surgery versus later commencement of feeding: a systematic review and meta-analysis. J Gastrointest Surg 2009; 13:569–575.
- Purkayastha S, Tilney HS, Darzy AW, Tekkis PP. Meta-analysis of studies evaluating chewing gum to enhance postoperative recovery following colectomy. Arch Surg 2008; 143:788–793.
- Ramirez B, Eaker EY, Drane WE, Hocking MP, Sninsky CA. Erythromycin enhances gastric emptying in patients with gastroparesis after vagotomy and antrectomy. Dig Dis Sci 1994; 39:2295–2300.
- Kendall BJ, Chakravarti A, Kendall E, Soykan I, McCallum RW. The effect of intravenous erythromycin on solid meal gastric emptying in patients with chronic symptomatic post-vagotomy-antrectomy gastroparesis. Aliment Pharmacol Ther 1997; 11:381–385.
- Yeo CJ, Barry MK, Sauter PK, et al. Erythromycin accelerates gastric emptying after pancreaticoduodenectomy. A prospective, randomized, placebo-controlled trial. Ann Surg 1993; 218:229–237.
- Jooste CA, Mustoe J, Collee G. Metoclopramide improves gastric motility in critically ill patients. Intensive Care Med 1999; 25:464–468.
- Sustic A, Zelic M, Protic A, et al. Metoclopramide improves gastric but not gallbladder emptying in cardiac surgery patients with early intragastric enteral feeding: randomized controlled trial. Croat Med J 2005; 46:239–244.
- US Food and Drug Administration. FDA requires boxed warning and risk mitigation strategy for metoclopramide-containing drugs. Agency warns against chronic use of these products to treat gastrointestinal disorders. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm149533.htm. Accessed 8/24/2009.
- Smith AJ, Nissan A, Lanouette NM, et al. Prokinetic affect of erythromycin after colorectal surgery: a randomized, placebo-controlled, double-blind study. Dis Colon Rectum 2000; 43:333–337.
- Cheape JD, Wexner SD, Jagelman JK. Does metoclopramide reduce the length of ileus after colorectal surgery? A prospective randomized trial. Dis Colon Rectum 1991; 34:437–441.
- De Giorgio R, Knowles CH. Acute colonic pseudo-obstruction. Br J Surg 2009; 96:229–239.
- Ponec RJ, Saunders MD, Kimmey MB. Neostigmine for the treatment of acute colonic pseudo-obstruction. N Engl J Med 1999; 341:137–141.
- Amaro R, Rogers AI. Neostigmine infusions: new standard of care for acute colonic pseudo-obstruction? Am J Gastroenterol 2000; 95:304–305.
- van der Spoel JI, Oudemans-van Straaten HM, Stroutenbeek CP, Bosman RJ, Zandstra DF. Neostigmine resolves critical illness-related colonic ileus in intensive care patients with multiple organ failure: a prospective, double-blind, placebo-controlled trial. Intensive Care Med 2001; 27:822–827.
- Zimmerman DM, Gidda JS, Cantrell BE, et al. LY246736 dihydrate μ-opioid receptor antagonist. Drugs Future 1994; 19:1078–1083.
- Callaghan JT, Cerimele B, Nowak TV, et al. Effect of the opioid antagonist LY246736 on gastrointestinal transit in human subjects [abstract]. Gastroenterology 1998; 114:G3015.
- Hodgson PS, Liu SS, Carpenter RI. ADL 8-2698 prevents morphine inhibition of GI transit [abstract]. Clin Pharmacol Ther 2000; 67:93.
- Liu SS, Hodgson PS, Carpenter RL, Fricke JR. ADL 8-2698, a trans-3,4-dimethyl-4-(3-hydroxyphenyl) piperidine, prevents gastrointestinal effects of intravenous morphine without affecting analgesia. Clin Pharmacol Ther 2001; 68:66–71.
- Wolff BG, Michelassi F, Gerkin TM, et al. Alvimopan, a novel, peripherally acting mu opioid antagonist: results of a multicenter, randomized, double-blind, placebo-controlled, phase III trial of major abdominal surgery and postoperative ileus. Ann Surg 2004; 240:728–735.
- Delaney CP, Weese JL, Hyman NH, et al; Alvimopan Postoperative Ileus Study Group. Phase III trial of alvimopan, a novel, peripherally acting, mu opioid antagonist, for postoperative ileus after major abdominal surgery. Dis Colon Rectum 2005; 48:1114–1129.
- Delaney CP, Wolff BG, Viscusi ER, et al. Alvimopan, for postoperative ileus following bowel resection: a pooled analysis of phase III studies. Ann Surg 2007; 245:355–363.
- Ludwig K, Enker WE, Delaney CP, et al. Gastrointestinal tract recovery in patients undergoing bowel resection: results of a randomized trial of alvimopan and placebo with a standardized accelerated postoperative care pathway. Arch Surg 2008; 143:1098–1105.
- Adolor GlaxoSmithKline. Entereg (alvimopan). www.entereg.com/Accessed 8/24/2009.
- Pritts TA, Nussbaum MS, Flesch LV, Fegelman EJ, Parikh AA, Fischer JE. Implementation of a clinical pathway decreases length of stay and cost for bowel resection. Ann Surg 1999; 230:728–733.
- Delaney CP, Zutshi M, Senagore AJ, Remzi FH, Hammel J, Fazio VW. Prospective, randomized, controlled trial between a pathway of controlled rehabilitation with early ambulation and diet and traditional postoperative care after laparotomy and intestinal resection. Dis Colon Rectum 2003; 46:851–859.
- Joh YG, Lindsetmo RO, Stulberg J, Obias V, Champagne B, Delaney CP. Standardized postoperative pathway: accelerating recovery after ileostomy closure. Dis Colon Rectum 2008; 51:1786–1789.
- Kennedy EP, Rosato EL, Sauter PK, et al. Initiation of a critical pathway for pancreaticoduodenectomy at an academic institution—the first step in multidisciplinary team building. J Am Coll Surg 2007; 204:917–924.
- Kariv Y, Delaney CP, Senagore AJ, et al. Clinical outcomes and cost analysis of a “fast track” postoperative care pathway for ileal pouch-anal anastomosis: a case control study. Dis Colon Rectum 2007; 50:137–146.
KEY POINTS
- Postoperative ileus can selectively affect the stomach, small intestine, or large intestine, each with different causes and clinical presentation and each managed differently.
- Laparoscopic surgery is associated with a shorter duration of postoperative ileus compared with open surgery.
- Epidural anesthesia reduces the need for opiate analgesia after surgery and thus shortens the duration of postoperative ileus.
- Drugs are being developed that block the effects of opiates on the gut while preserving their pain-relieving properties.