User login
General Applications of Ultrasound in Rheumatology Practice
Over the past 2 decades, an increasing number of rheumatologists have progressively incorporated ultrasound (US) as an invaluable diagnostic and monitoring tool into their clinical and research practice.1,2 This imaging modality has become an established aid incorporated into the clinical evaluation of periarticular and articular structures involved in the diagnosis of several rheumatic disorders.
Ultrasound is a safe, noninvasive, patient-friendly imaging modality with a lack of contraindications and free of ionizing radiation. It allows real-time evaluation with dynamic assessment in a multiplanar view, assessment of multiple targets, and lower cost compared with magnetic resonance imaging (MRI) or computerized tomography scan. Above all, for the rheumatologist, US provides real-time scanning of all peripheral joints as many times as is required at the time of consultation. It is of great advantage in the assessment of a wide spectrum of abnormalities in rheumatic diseases with the potential of point-of-care imaging modality in the clinical evaluation and management of the patient. It facilitates a direct correlation between imaging findings and clinical data that improves the approach to a wide range of rheumatic diseases, from acute to chronic inflammatory arthritis, crystalline arthropathies, osteoarthritis (OA), spondyloarthropathies (SpA), vasculitis, and soft tissue syndromes. In addition, US is a bedside tool for performing accurate and safe diagnostic arthrocentesis, injections, and synovial biopsies.3,4
Recently, a gradual attempt has been made to incorporate US into rheumatology disease classification or diagnostic criteria for rheumatoid arthritis (RA), polymyalgia rheumatica, gout, calcium pyrophosphate deposition disease (CPPD), and Sjögren’s syndrome.5-10 Furthermore, the American College of Rheumatology (ACR) and the European League Against Rheumatism (EULAR) have produced evidence and expert opinion-based recommendations on the use of US in the clinical management of rheumatic diseases.10-12 This article highlights the most common applications of US for assessment and management of different rheumatic diseases frequently encountered at the VAMC rheumatology inpatient and outpatient clinical service.
Evaluation of Inflammatory Arthritis
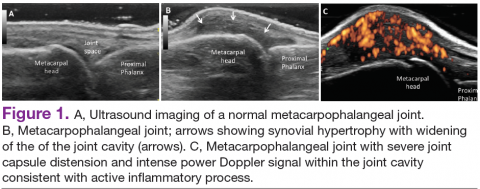
In RA and any other inflammatory arthritis, US has been used for the detection of joint effusions, synovitis, bone erosions, and tendon and enthesis involvement.11,12 Ultrasound B-mode and power Doppler (PD) techniques have demonstrated a consistent and relevant role in optimizing the diagnosis, assessing the inflammatory activity, monitoring response to therapy, and predicting the inflammatory arthritis outcomes (Figures 1-3).10-12 Ultrasound provides real-time information about the status of the synovial membrane, tendons, cartilage, bursae, and cortical bones, allowing an accurate assessment of the degree of inflammatory process in periarticular and articular tissues. Also, US can provide details about the characteristics of the collected fluid (ie, effusion or synovial hypertrophy), which is fundamental for the correct interpretation of the pathologic joint and/or soft tissue processes. The inflammatory process can be assessed by using PD mode, which detects and quantifies the vascular changes in the pannus due to vasodilation and the increased blood flow characteristic of active inflammation.13,14
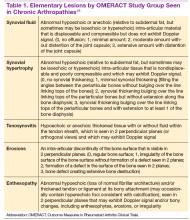
The Outcomes Measures in Rheumatoid Arthritis Clinical Trials (OMERACT) study group developed standardized sonopathologic definitions and scanning methods to be used in the daily rheumatologic practice and clinical trials (Table 1).15 Furthermore, it developed a semiquantitative scale to assess the degree of synovitis in US B-mode and PD mode (Table 2).15
The use of US to find subclinical synovitis in patients with RA considered to be in clinical remission is a new issue.16 Some reports have demonstrated progressive joint damage in these patients with evidence of active inflammation on PDUS despite clinical remission.17,18 More prospective studies are required to provide a better understanding of the long-term effects of residual inflammation and the proper long-term treatment of these patients. Furthermore, the PD signal has been shown to be superior to the Disease Activity Score 28 (DAS-28) in evaluating disease activity, particularly in predicting joint damage.18
Ultrasound may be considered the gold standard imaging tool for the assessment of tendons in inflammatory arthritis and includes the detection of tenosynovitis and anatomical damage represented by the loss of the normal fibrillar echotexture and loss of definition of the tendon margins, which may occur in early disease.19,20 Tenosynovitis of the extensor carpi ulnaris (ECU) detected by US has been shown to be an independent predictive factor of erosive joint damage, suggesting that ECU tenosynovitis represents a useful ultrasonographic landmark in the diagnosis of early RA.21
The availability of new nonbiologic and biologic therapies for inflammatory arthritis has raised the importance of identifying early changes, such as the detection of early erosions, which portend a poor long-term prognosis. The capability of US in identifying this lesion at an earlier stage compared with conventional radiography (CR) has allowed the early diagnosis and treatment of these patients before irreversible joint destruction occurs.22 In spite of all the supportive evidence of US utility in RA, it is not considered among the mandatory diagnostic criteria in the ACR/EULAR classification criteria for RA.5 Still, the addition of US findings to these criteria has increased the number of patients who fulfilled the 1987 ACR classification criteria for RA after 18 months of follow-up.23 Despite extensive evidence of its utility in the diagnosis and monitoring of RA, further studies are still needed.
Spondyloarthritis
Similar to RA, SpA discloses sonographic findings of inflammatory arthritis; however, with more entheseal and tenosynovium involvement. Ultrasound has also been used in the early identification of characteristic changes of the skin and nail tissues, which can aid the global assessment of this heterogeneous disease, especially in psoriatic arthritis (PsA). The most common locations of enthesitis in SpA are the quadriceps and the Achilles enthesis.24,25
Although US offers detailed imaging for the assessment of both tendons and enthesis, there is a lack of literature evaluating dactylitis. The OMERACT group recently released a composite measure of activity and severity of US dactylitis, which included newly defined elementary US lesions that may discern dactylitis of a digit.26,27 Ultrasound has been compared with MRI in the detection of SpA-related synovitis of the hands and feet and has demonstrated competitive diagnostic sensitivity.28 Ultrasound also shows higher sensitivity in detecting synovitis of the hands and feet compared with clinical examination and CR in PsA.28,29 Unfortunately, there are no strongly validated US findings that can aid in the differential diagnosis of PsA against other chronic inflammatory arthritides. The presence of peritendinous extensor tendon inflammation was a highly specific sonographic feature of PsA, because it was present in 66% of metacarpophalangeal (MCP) joints as the only US sign of inflammation compared with patients with RA.30
Another application of US is in the evaluation of subclinical inflammation at the enthesis in patients with a history of psoriasis without prior history of PsA.27,31 In those patients with psoriatic nail changes, more subclinical enthesitis was found compared with patients with psoriasis without nail involvement.32 Furthermore, subclinical joint inflammation has also been described.33 These findings suggest a possible predictive value in patients with psoriasis who should be monitored on a regular basis, because they are at risk of developing PsA.
Subclinical enthesitis by US imaging has been described in patients with recurrent anterior uveitis and inflammatory bowel disease.34,35 In cases where SpA is suspected but diagnostic criteria are not fulfilled, the presence of one enthesis with increased PD signal highly predicts the eventual development of SpA.36 Therefore, B-mode and PD evaluations of the entheses are critical in the identification of patients who are at an increased risk of developing SpA.37 Treatment monitoring is performed by using a US scoring system in a follow-up evaluation of patients with PsA. Some of the scoring systems have evaluated changes in B-mode US lesions (enthesis and soft tissues, such as skin and nails), whereas others focus on changes in the PD signal.37,38
The Five Targets Power Doppler for Psoriatic Disease PD scoring system comprises the assessment of PD signal in the joint, tendon with synovial sheath, enthesis, skin, and nails. Each of the targets is scored from 0 to 3 points, with a maximum of 15 total points. Some studies have shown that PDUS can provide valuable information in the evaluation of psoriatic plaques and onychopathy in patients with psoriasis and PsA.39 The detection of a PD signal within the dermis and nail bed is equivalent to active inflammation in these sites.39-41 However, further studies with larger cohorts proving inter- and intra-observer reliability are necessary to consolidate these findings and comfortably apply them in clinical practice.
Osteoarthritis
Increasingly US is studied for its validity and reliability in evaluating periarticular soft tissue and cartilage changes in knee OA. The associated US findings include a high prevalence of synovitis with a low prevalence of a PD signal, the presence of osteophytes, and joint space narrowing.42,43 Increased PD signal, synovial hypertrophy, and joint effusion were observed in patients with radiographically erosive OA compared with those with radiographically nonerosive OA.44
Bone erosions and inflammatory changes are also frequently detected by US in both erosive and nodal hand OA.45 Compared with MRI, US has shown a good to excellent correlation in the assessment of osteophytes, bone erosions, synovitis, and tenosynovitis in erosive and hand nodal OA.46 In comparison with CR, US has shown to have a higher sensitivity in the assessment of bony erosions, osteophytes, and space narrowing.47 Ultrasound is able to detect changes in the earlier stages of cartilage erosion in OA, characterized by loss of the sharp contour and variations in the echogenicity of the cartilage matrix, asymmetric shrinkage, and ultimately the disappearance of the cartilaginous band, which is more evident in the later stages of OA.45
Similar to RA management, US has been used to monitor disease activity and response to OA treatment. Patients who received intra-articular hyaluronic acid or intramuscular methylprednisolone for OA treatment were found to have a decrease of PD signal intensity and synovial effusion posttreatment.48 One could extrapolate these findings and conclude that US could be an additional tool for monitoring disease activity and assessing response to local and systemic treatments in OA.
Crystalline Arthropathies
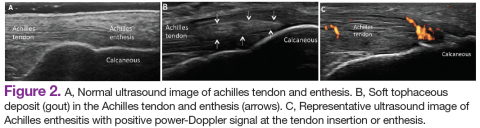
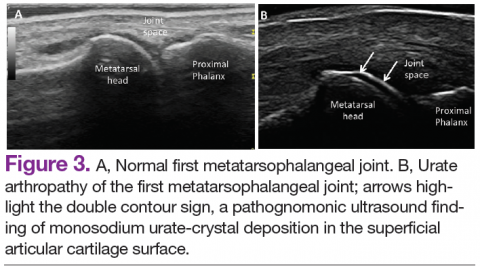
Ultrasound application to crystal diseases facilitates the identification of microcrystalline deposits within the synovial membrane (joints), cartilage (both hyaline cartilage and fibrocartilage), and periarticular tissues (tendons, bursae, and soft tissues). Crystals appear as hyperechogenic spots of different sizes and shapes that can be seen in both articular and periarticular tissues.49,50 The crystal deposition pattern on hyaline cartilage allows the differentiation between monosodium urate (MSU) and calcium pyrophosphate dehydrate (CPP) crystals. The MSU crystals are deposited at the chondrosynovial (or superficial) margin of the hyaline cartilage and described sonographically as the double contour sign in gout, whereas CPP crystals are deposited within the intermediate layer of the hyaline cartilage and are seen as hyperechoic spots frequently described as rosary beads on US.6,49,50
Other important sites that can be evaluated to determine the presence of CPP crystals include the menisci, symphysis pubis, and triangular fibrocartilage at the wrists, hips, and shoulders. Recent EULAR recommendations have incorporated US as part of the diagnostic imaging modality for the diagnosis of CPPD and more recently for gout.6,51 Tophi are seen as MSU precipitates deposited in the joint cavity, tendons, and/or periarticular tissues such as bursae. They can show different echogenic signal. Soft tophi can demonstrate high PD signal due to high vascularization. On the other hand, hard tophi are hyperechoic on B-mode due to the presence of calcification, which does not allow passage of US waves, creating postacoustic shadowing.8 Studies have evaluated the predictive role of US in evaluating patients with asymptomatic hyperuricemia without any prior history of crystal-related joint disease and found tophaceous deposits in the triceps and patellar and quadriceps tendons.52-55 Studies have also looked at using US in the assessment of treatment response to serum uratelowering therapy in patients with gout.56,57 These studies have noted an improvement in the double contour sign, hyperechoic spots, cloudy areas in the synovial fluid, and tophus diameter and size in those patients who achieved a treat-to-target with a serum uric acid level ≤ 6 mg/dL. Patients who did not reach this target had no changes in the gout US features.56-57 Larger cohort studies are needed to confirm these findings.
An active inflammatory process can be determined by using a PD signal in the acute gout setting with increased vascularization; however, an increased PD signal can also be seen in septic arthritis or tenosynovitis, which sometimes can coexist with crystal-induced arthritis. Therefore, diagnostic arthrocentesis, Gram stain, and culture, as well as evaluation of crystals under polarized microscopy, are still recommended.
Therapeutic Interventions
Real-time visualization of the injection needle by US allows reliable placement of the needle tip in the tissue or cavity of interest. Multiple studies have shown the low accuracy of palpation-guided injection for reaching the site of interest.58,59 Some studies have shown a higher response rate to US-guided injections compared with palpation-guided as well as a higher rate of successful aspirations and clinical outcomes. Meta-analyses have demonstrated improved treatment response with the use of US-guided procedures compared with blinded injections.60,61 Ultrasound-guided interventions are performed in both peripheral and axial joints.62 The most common US-guided procedures at the VA rheumatology clinic include arthrocentesis and intra-articular corticosteroid injections of small and medium-sized joints, such as MCP joints, elbows, wrists, and ankles.
Conclusions
Ultrasound is becoming a relevant part of rheumatology practice and research and can be regarded as a feasible and effective imaging technique that can allow real-time recognition of early anatomical changes, provide careful guidance for aspiration, and monitor local and/or systemic treatment response at the joint, tendon, enthesis, nail, and skin levels. Ultrasound is a user-friendly imaging modality readily applied at the bedside and considered an extension of the rheumatologist's physical examination.
The success of US depends on the individual operator. For this reason, structured educational programs during fellowship training programs and an efficient competency assessment system would facilitate proper implementation of US in rheumatology practice as performed by some but not all institutions.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Naredo E, D’Agostino MA, Conaghan PG, et al. Current state of musculoskeletal ultrasound training and implementation in Europe: results of a survey of experts and scientific societies. Rheumatology (Oxford). 2010;49(12):2438-2943.
2. Micu MC, Alcalde M, Sáenz JI, et al. Impact of musculoskeletal ultrasound in an outpatient rheumatology clinic. Arthritis Care Res (Hoboken). 2013;65(4):615-621.
3. Koski JM. Ultrasound guided injections in rheumatology. J Rheumatol. 2000;27(9):2131-2138.
4. Kelly S, Humby F, Filer A, et al. Ultrasound-guided synovial biopsy: a safe, well-tolerated and reliable technique for obtaining high-quality synovial tissue from both large and small joints in early arthritis patients. Ann Rheum Dis. 2015;74(3):611-617.
5. Aletaha D, Neogi T, Silman AJ, et al. 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum Dis. 2010;69(9):1580-1588.
6. Zhang W, Doherty M, Bardin T, et al. European League Against Rheumatism recommendations for calcium pyrophosphate deposition. Part I: terminology and diagnosis. Ann Rheum Dis. 2011;70(4):563-570.
7. Dasgupta B, Cimmino MA, Kremers HM, et al. 2012 provisional classification criteria for polymyalgia rheumatica: a European League Against Rheumatism/ American College of Rheumatology collaborative initiative. Arthritis Rheum. 2012;64(4):943-954.
8. Fodor D, Nestorova R, Vlad V, Micu M. The place of musculoskeletal ultrasonography in gout diagnosis. Med Ultrason. 2014;16(4):336-344.
9. Takagi Y, Sumi M, Nakamura H, et al. Ultrasonography as an additional item in the American College of Rheumatology classification of Sjögren’s syndrome. Rheumatology (Oxford). 2014;53(11):1977-1983.
10. Colebatch AN, Edwards CJ, Østergaard M, et al. EULAR recommendations for the use of imaging of the joints in the clinical management of rheumatoid arthritis. Ann Rheum Dis. 2013;72(6):804-814.
11. American College of Rheumatology Musculoskeletal Ultrasound Task Force. Ultrasound in American rheumatology practice: report of the American College of Rheumatology musculoskeletal ultrasound task force. Arthritis Care Res (Hoboken). 2010;62(9):1206-1219.
12. McAlindon T, Kissin E, Nazarian L, et al. American College of Rheumatology report on reasonable use of musculoskeletal ultrasonography in rheumatology clinical practice. Arthritis Care Res (Hoboken). 2012;64(11):1625-1640.
13. Naredo E, Möller I, Cruz A, Carmona L, Garrido J. Power Doppler ultrasonographic monitoring of response to anti-tumor necrosis factor therapy in patients with rheumatoid arthritis. Arthritis Rheum. 2008;58(8):2248-2256.
14. Newman JS, Laing TJ, McCarthy CJ, Adler RS. Power Doppler sonography of synovitis: assessment of therapeutic response—preliminary observations. Radiology. 1996;198(2):582-584.
15. Wakefield RJ, Balint PV, Szkudlarek M, et al; OMERACT 7 Special Interest Group. Musculoskeletal ultrasound including definitions for ultrasonographic pathology. J Rheumatol. 2005;32(12):2485-2487.
16. Wakefield RJ, Green MJ, Marzo-Ortega H, et al. Should oligoarthritis be reclassified? Ultrasound reveals a high prevalence of subclinical disease. Ann Rheum Dis. 2004;63(4):382-385.
17. Brown AK, Quinn MA, Karim Z, et al. Presence of significant synovitis in rheumatoid arthritis patients with disease-modifying antirheumatic drug-induced clinical remission: evidence from an imaging study may explain structural progression. Arthritis Rheum. 2006;54(12):3761-3773.
18. Brown AK, Conaghan PG, Karim Z, et al. An explanation for the apparent dissociation between clinical remission and continued structural deterioration in rheumatoid arthritis. Arthritis Rheum. 2008;58(10):2958-2967.
19. Bruyn GA, Hanova P, Iagnocco A, et al; OMERACT Ultrasound Task Force. Ultrasound definition of tendon damage in patients with rheumatoid arthritis. Results of a OMERACT consensus-based ultrasound score focusing on the diagnostic reliability. Ann Rheum Dis. 2014;73(11):1929-1934.
20. Filippucci E, Gabba A, Di Geso L, Girolimetti R, Salaffi F, Grassi W. Hand tendon involvement in rheumatoid arthritis: an ultrasound study. Semin Arthritis Rheum. 2012;41(6):752-760.
21. Lillegraven S, Bøyesen P, Hammer HB, et al. Tenosynovitis of the extensor carpi ulnaris tendon predicts erosive progression in early rheumatoid arthritis. Ann Rheum Dis. 2011;70(11):2049-2050.
22. Baillet A, Gaujoux-Viala C, Mouterde G, et al. Comparison of the efficacy of sonography, magnetic resonance imaging and conventional radiography for the detection of bone erosions in rheumatoid arthritis patients: a systematic review and meta-analysis. Rheumatology (Oxford). 2011;50(6):1137-1147.
23. Filer A, de Pablo P, Allen G, et al. Utility of ultrasound joint counts in the prediction of rheumatoid arthritis in patients with very early synovitis. Ann Rheum Dis. 2011;70(3):500-507.
24. Frediani B, Falsetti P, Storri L, et al. Quadricepital tendon enthesitis in psoriatic arthritis and rheumatoid arthritis: ultrasound examinations and clinical correlations. J Rheumatol. 2001;28(11):2566-2568.
25. D’Agostino MA, Said-Nahal R, Hacquard-Bouder C, Brasseur JL, Dougados M, Breban M. Assessment of peripheral enthesitis in the spondylarthropathies by ultrasonography combined with power Doppler: a cross-sectional study. Arthritis Rheum. 2003;48(2):523-533.
26. Gisondi P, Tinazzi I, El-Dalati G, et al. Lower limb enthesopathy in patients with psoriasis without clinical signs of arthropathy: a hospital-based case-control study. Ann Rheum Dis. 2008;67(1):26-30.
27. Gutierrez M, Filippucci E, De Angelis R, et al. Subclinical entheseal involvement
in patients with psoriasis: an ultrasound study. Semin Arthritis Rheum. 2011;40(5):407-412.
28. Weiner SM, Jurenz S, Uhl M, et al. Ultrasonography in the assessment of peripheral joint involvement in psoriatic arthritis: a comparison with radiography, MRI and scintigraphy. Clin Rheumatol. 2008;27(8):983-989.
29. Balint PV, Kane D, Wilson H, McInnes IB, Sturrock RD. Ultrasonography of entheseal insertions in the lower limb in spondyloarthropathy. Ann Rheum Dis. 2002;61(10):905-910.
30. Gutierrez M, Filippucci E, Salaffi F, Di Geso L, Grassi W. Differential diagnosis
between rheumatoid arthritis and psoriatic arthritis: the value of ultrasound findings at metacarpophalangeal joints level. Ann Rheum Dis. 2011;70(6):1111-1114.
31. De Miguel E, Cobo T, Muñoz-Fernández S, et al. Validity of enthesis ultrasound assessment in spondyloarthropathy. Ann Rheum Dis. 2009;68(2):169-174.
32. Ash ZR, Tinazzi I, Gallego CC, et al. Psoriasis patients with nail disease have a greater magnitude of underlying systemic subclinical enthesopathy than those with normal nails. Ann Rheum Dis. 2012;71(4):553-556.
33. Naredo E, Möller I, de Miguel E, et al; Ultrasound School of the Spanish Society of Rheumatology and Spanish ECO-APs Group. High prevalence of ultrasonographic synovitis and enthesopathy in patients with psoriasis without psoriatic arthritis: a prospective case-control study. Rheumatology (Oxford). 2011;50(10):1838-1848.
34. Muñoz-Fernández S, de Miguel E, Cobo-Ibáñez T, et al. Enthesis inflammation in recurrent acute anterior uveitis without spondylarthritis. Arthritis Rheum. 2009;60(7):1985-1990.
35. Bandinelli F, Milla M, Genise S, et al. Ultrasound discloses entheseal involvement
in inactive and low active inflammatory bowel disease without clinical signs and symptoms of spondyloarthropathy. Rheumatology (Oxford). 2011;50(7):1275-1279.
36. D’Agostino MA, Aegerter P, Bechara K, et al. How to diagnose spondyloarthritis early? Accuracy of peripheral enthesitis detection by power Doppler ultrasonography. Ann Rheum Dis. 2011;70(8):1433-1440.
37. Aydin SZ, Karadag O, Filippucci E, et al. Monitoring Achilles enthesitis in ankylosing spondylitis during TNF-alpha antagonist therapy: an ultrasound study. Rheumatology (Oxford). 2010;49(3):578-582.
38. Naredo E, Batlle-Gualda E, Garcia-Vivar ML, et al; Ultrasound Group of the Spanish Society of Rheumatology. Power Doppler ultrasonography assessment of entheses in spondyloarthropathies: response to therapy of entheseal abnormalities. J Rheumatol. 2010;37(10):2110-2117.
39. Gutierrez M, Di Geso L, Salaffi F, et al. Development of a preliminary US power Doppler composite score for monitoring treatment in PsA. Rheumatology (Oxford). 2012;51(7):1261-1268.
40. Gutierrez M, De Angelis R, Bernardini ML, et al. Clinical, power Doppler sonography and histological assessment of the psoriatic plaque: short-term monitoring in patients treated with etanercept. Br J Dermatol. 2011;164(1):33-37.
41. Gutierrez M, Filippucci E, Bertolazzi C, Grassi W. Sonographic monitoring of psoriatic plaque. J Rheumatol. 2009;36(4):850-851.
42. Keen HI, Wakefield RJ, Grainger AJ, Hensor EM, Emery P, Conaghan PG. An ultrasonographic study of osteoarthritis of the hand: synovitis and its relationship to structural pathology and symptoms. Arthritis Rheum. 2008;59(12):1756-1763.
43. Kortekaas MC, Kwok WY, Reijnierse M, Watt I, Huizinga TW, Kloppenburg M. Pain in hand osteoarthritis is associated with inflammation: the value of ultrasound. Ann Rheum Dis. 2010;69(7):1367-1369.
44. Mancarella L, Magnani M, Addimanda O, Pignotti E, Galletti S, Meliconi R. Ultrasound-detected synovitis with power Doppler signal is associated with severe radiographic damage and reduced cartilage thickness in hand osteoarthritis. Osteoarthritis Cartilage. 2010;18(10):1263-1268.
45. Möller I, Bong D, Naredo E, et al. Ultrasound in the study and monitoring of osteoarthritis. Osteoarthritis Cartilage. 2008;16(suppl 3):S4-S7.
46. Vlychou M, Koutroumpas A, Alexiou I, Fezoulidis I, Sakkas LI. High-resolution ultrasonography and 3.0 T magnetic resonance imaging in erosive and nodal hand osteoarthritis: high frequency of erosions in nodal osteoarthritis. Clin Rheumatol. 2013;32(6):755-762.
47. Keen HI, Wakefield RJ, Grainger AJ, Hensor EM, Emery P, Conaghan PG. Can ultrasonography improve on radiographic assessment in osteoarthritis of the hands? A comparison between radiographic and ultrasonographic detected pathology. Ann Rheum Dis. 2008;67(8):1116-1120.
48. Keen HI, Wakefield RJ, Hensor EM, Emery P, Conaghan PG. Response of symptoms
and synovitis to intra-muscular methylprednisolone in osteoarthritis of the hand: an ultrasonographic study. Rheumatology (Oxford). 2010;49(6):1093-1100.
49. Grassi W, Meenagh G, Pascual E, Filippucci E. “Crystal clear”-sonographic assessment of gout and calcium pyrophosphate deposition disease. Semin Arthritis Rheum. 2006;36(3):197-202.
50. Ciapetti A, Filippucci E, Gutierrez M, Grassi W. Calcium pyrophosphate dihydrate crystal deposition disease: sonographic findings. Clin Rheumatol. 2009;28(3):271-276.
51. Taylor WJ, Fransen J, Jansen TL, et al. Study for Updated Gout Classification Criteria (SUGAR): identification of features to classify gout. Arthritis Care Res (Hoboken). [Published online ahead of print March 16, 2015.]
52. Puig JG, de Miguel E, Castillo MC, Rocha AL, Martinez MA, Torres RJ. Asymptomatic hyperuricemia: impact of ultrasonography. Nucleosides Nucleotides Nucleic Acids. 2008;27(6):592-595.
53. Pineda C, Amezcua-Guerra LM, Solano C, et al. Joint and tendon subclinical involvement suggestive of gouty arthritis in asymptomatic hyperuricemia: an ultrasound controlled study. Arthritis Res Ther. 2011;13(1):R4.
54. Naredo E, Uson J, Jiménez-Palop M, et al. Ultrasound-detected musculoskeletal urate crystal deposition: which joints and what findings should be assessed for diagnosing gout? Ann Rheum Dis. 2014;73(8):1522-1528.
55. De Miguel E, Puig JG, Castillo C, Peiteado D, Torres RJ, Martín-Mola E. Diagnosis of gout in patients with asymptomatic hyperuricaemia: a pilot ultrasound study. Ann Rheum Dis. 2012;71(1):157-158.
56. Perez-Ruiz F, Martin I, Canteli B. Ultrasonographic measurement of tophi as an outcome measure for chronic gout. J Rheumatol. 2007;34(9):1888-1893.
57. Thiele RG, Schlesinger N. Ultrasonography shows disappearance of monosodium urate crystal deposition on hyaline cartilage after sustained normouricemia is achieved. Rheumatol Int. 2010;30(4):495-503.
58. Balint PV, Kane D, Hunter J, McInnes IB, Field M, Sturrock RD. Ultrasound guided versus conventional joint and soft tissue fluid aspiration in rheumatology practice: a pilot study. J Rheumatol. 2002;29(10):2209-2213.
59. Raza K, Lee CY, Pilling D, et al. Ultrasound guidance allows accurate needle placement and aspiration from small joints in patients with early inflammatory arthritis. Rheumatology (Oxford). 2003;42(8):976-979.
60. Dubreuil M, Greger S, LaValley M, Cunnington J, Sibbitt WL Jr, Kissin EY. Improvement in wrist pain with ultrasound-guided glucocorticoid injections: a metaanalysis of individual patient data. Semin Arthritis Rheum. 2013;42(5):492-497.
61. Sage W, Pickup L, Smith TO, Denton ER, Toms AP. The clinical and functional outcomes of ultrasound-guided vs landmark-guided injections for adults with shoulder pathology—a systematic review and meta-analysis. Rheumatology (Oxford). 2013;52(4):743-751.
62. Darrieutort-Laffite C, Hamel O, Glémarec J, Maugars Y, Le Goff B. Ultrasonography of the lumbar spine: sonoanatomy and practical applications. Joint Bone Spine. 2014;81(2):130-136.
Over the past 2 decades, an increasing number of rheumatologists have progressively incorporated ultrasound (US) as an invaluable diagnostic and monitoring tool into their clinical and research practice.1,2 This imaging modality has become an established aid incorporated into the clinical evaluation of periarticular and articular structures involved in the diagnosis of several rheumatic disorders.
Ultrasound is a safe, noninvasive, patient-friendly imaging modality with a lack of contraindications and free of ionizing radiation. It allows real-time evaluation with dynamic assessment in a multiplanar view, assessment of multiple targets, and lower cost compared with magnetic resonance imaging (MRI) or computerized tomography scan. Above all, for the rheumatologist, US provides real-time scanning of all peripheral joints as many times as is required at the time of consultation. It is of great advantage in the assessment of a wide spectrum of abnormalities in rheumatic diseases with the potential of point-of-care imaging modality in the clinical evaluation and management of the patient. It facilitates a direct correlation between imaging findings and clinical data that improves the approach to a wide range of rheumatic diseases, from acute to chronic inflammatory arthritis, crystalline arthropathies, osteoarthritis (OA), spondyloarthropathies (SpA), vasculitis, and soft tissue syndromes. In addition, US is a bedside tool for performing accurate and safe diagnostic arthrocentesis, injections, and synovial biopsies.3,4
Recently, a gradual attempt has been made to incorporate US into rheumatology disease classification or diagnostic criteria for rheumatoid arthritis (RA), polymyalgia rheumatica, gout, calcium pyrophosphate deposition disease (CPPD), and Sjögren’s syndrome.5-10 Furthermore, the American College of Rheumatology (ACR) and the European League Against Rheumatism (EULAR) have produced evidence and expert opinion-based recommendations on the use of US in the clinical management of rheumatic diseases.10-12 This article highlights the most common applications of US for assessment and management of different rheumatic diseases frequently encountered at the VAMC rheumatology inpatient and outpatient clinical service.
Evaluation of Inflammatory Arthritis
In RA and any other inflammatory arthritis, US has been used for the detection of joint effusions, synovitis, bone erosions, and tendon and enthesis involvement.11,12 Ultrasound B-mode and power Doppler (PD) techniques have demonstrated a consistent and relevant role in optimizing the diagnosis, assessing the inflammatory activity, monitoring response to therapy, and predicting the inflammatory arthritis outcomes (Figures 1-3).10-12 Ultrasound provides real-time information about the status of the synovial membrane, tendons, cartilage, bursae, and cortical bones, allowing an accurate assessment of the degree of inflammatory process in periarticular and articular tissues. Also, US can provide details about the characteristics of the collected fluid (ie, effusion or synovial hypertrophy), which is fundamental for the correct interpretation of the pathologic joint and/or soft tissue processes. The inflammatory process can be assessed by using PD mode, which detects and quantifies the vascular changes in the pannus due to vasodilation and the increased blood flow characteristic of active inflammation.13,14
The Outcomes Measures in Rheumatoid Arthritis Clinical Trials (OMERACT) study group developed standardized sonopathologic definitions and scanning methods to be used in the daily rheumatologic practice and clinical trials (Table 1).15 Furthermore, it developed a semiquantitative scale to assess the degree of synovitis in US B-mode and PD mode (Table 2).15
The use of US to find subclinical synovitis in patients with RA considered to be in clinical remission is a new issue.16 Some reports have demonstrated progressive joint damage in these patients with evidence of active inflammation on PDUS despite clinical remission.17,18 More prospective studies are required to provide a better understanding of the long-term effects of residual inflammation and the proper long-term treatment of these patients. Furthermore, the PD signal has been shown to be superior to the Disease Activity Score 28 (DAS-28) in evaluating disease activity, particularly in predicting joint damage.18
Ultrasound may be considered the gold standard imaging tool for the assessment of tendons in inflammatory arthritis and includes the detection of tenosynovitis and anatomical damage represented by the loss of the normal fibrillar echotexture and loss of definition of the tendon margins, which may occur in early disease.19,20 Tenosynovitis of the extensor carpi ulnaris (ECU) detected by US has been shown to be an independent predictive factor of erosive joint damage, suggesting that ECU tenosynovitis represents a useful ultrasonographic landmark in the diagnosis of early RA.21
The availability of new nonbiologic and biologic therapies for inflammatory arthritis has raised the importance of identifying early changes, such as the detection of early erosions, which portend a poor long-term prognosis. The capability of US in identifying this lesion at an earlier stage compared with conventional radiography (CR) has allowed the early diagnosis and treatment of these patients before irreversible joint destruction occurs.22 In spite of all the supportive evidence of US utility in RA, it is not considered among the mandatory diagnostic criteria in the ACR/EULAR classification criteria for RA.5 Still, the addition of US findings to these criteria has increased the number of patients who fulfilled the 1987 ACR classification criteria for RA after 18 months of follow-up.23 Despite extensive evidence of its utility in the diagnosis and monitoring of RA, further studies are still needed.
Spondyloarthritis
Similar to RA, SpA discloses sonographic findings of inflammatory arthritis; however, with more entheseal and tenosynovium involvement. Ultrasound has also been used in the early identification of characteristic changes of the skin and nail tissues, which can aid the global assessment of this heterogeneous disease, especially in psoriatic arthritis (PsA). The most common locations of enthesitis in SpA are the quadriceps and the Achilles enthesis.24,25
Although US offers detailed imaging for the assessment of both tendons and enthesis, there is a lack of literature evaluating dactylitis. The OMERACT group recently released a composite measure of activity and severity of US dactylitis, which included newly defined elementary US lesions that may discern dactylitis of a digit.26,27 Ultrasound has been compared with MRI in the detection of SpA-related synovitis of the hands and feet and has demonstrated competitive diagnostic sensitivity.28 Ultrasound also shows higher sensitivity in detecting synovitis of the hands and feet compared with clinical examination and CR in PsA.28,29 Unfortunately, there are no strongly validated US findings that can aid in the differential diagnosis of PsA against other chronic inflammatory arthritides. The presence of peritendinous extensor tendon inflammation was a highly specific sonographic feature of PsA, because it was present in 66% of metacarpophalangeal (MCP) joints as the only US sign of inflammation compared with patients with RA.30
Another application of US is in the evaluation of subclinical inflammation at the enthesis in patients with a history of psoriasis without prior history of PsA.27,31 In those patients with psoriatic nail changes, more subclinical enthesitis was found compared with patients with psoriasis without nail involvement.32 Furthermore, subclinical joint inflammation has also been described.33 These findings suggest a possible predictive value in patients with psoriasis who should be monitored on a regular basis, because they are at risk of developing PsA.
Subclinical enthesitis by US imaging has been described in patients with recurrent anterior uveitis and inflammatory bowel disease.34,35 In cases where SpA is suspected but diagnostic criteria are not fulfilled, the presence of one enthesis with increased PD signal highly predicts the eventual development of SpA.36 Therefore, B-mode and PD evaluations of the entheses are critical in the identification of patients who are at an increased risk of developing SpA.37 Treatment monitoring is performed by using a US scoring system in a follow-up evaluation of patients with PsA. Some of the scoring systems have evaluated changes in B-mode US lesions (enthesis and soft tissues, such as skin and nails), whereas others focus on changes in the PD signal.37,38
The Five Targets Power Doppler for Psoriatic Disease PD scoring system comprises the assessment of PD signal in the joint, tendon with synovial sheath, enthesis, skin, and nails. Each of the targets is scored from 0 to 3 points, with a maximum of 15 total points. Some studies have shown that PDUS can provide valuable information in the evaluation of psoriatic plaques and onychopathy in patients with psoriasis and PsA.39 The detection of a PD signal within the dermis and nail bed is equivalent to active inflammation in these sites.39-41 However, further studies with larger cohorts proving inter- and intra-observer reliability are necessary to consolidate these findings and comfortably apply them in clinical practice.
Osteoarthritis
Increasingly US is studied for its validity and reliability in evaluating periarticular soft tissue and cartilage changes in knee OA. The associated US findings include a high prevalence of synovitis with a low prevalence of a PD signal, the presence of osteophytes, and joint space narrowing.42,43 Increased PD signal, synovial hypertrophy, and joint effusion were observed in patients with radiographically erosive OA compared with those with radiographically nonerosive OA.44
Bone erosions and inflammatory changes are also frequently detected by US in both erosive and nodal hand OA.45 Compared with MRI, US has shown a good to excellent correlation in the assessment of osteophytes, bone erosions, synovitis, and tenosynovitis in erosive and hand nodal OA.46 In comparison with CR, US has shown to have a higher sensitivity in the assessment of bony erosions, osteophytes, and space narrowing.47 Ultrasound is able to detect changes in the earlier stages of cartilage erosion in OA, characterized by loss of the sharp contour and variations in the echogenicity of the cartilage matrix, asymmetric shrinkage, and ultimately the disappearance of the cartilaginous band, which is more evident in the later stages of OA.45
Similar to RA management, US has been used to monitor disease activity and response to OA treatment. Patients who received intra-articular hyaluronic acid or intramuscular methylprednisolone for OA treatment were found to have a decrease of PD signal intensity and synovial effusion posttreatment.48 One could extrapolate these findings and conclude that US could be an additional tool for monitoring disease activity and assessing response to local and systemic treatments in OA.
Crystalline Arthropathies
Ultrasound application to crystal diseases facilitates the identification of microcrystalline deposits within the synovial membrane (joints), cartilage (both hyaline cartilage and fibrocartilage), and periarticular tissues (tendons, bursae, and soft tissues). Crystals appear as hyperechogenic spots of different sizes and shapes that can be seen in both articular and periarticular tissues.49,50 The crystal deposition pattern on hyaline cartilage allows the differentiation between monosodium urate (MSU) and calcium pyrophosphate dehydrate (CPP) crystals. The MSU crystals are deposited at the chondrosynovial (or superficial) margin of the hyaline cartilage and described sonographically as the double contour sign in gout, whereas CPP crystals are deposited within the intermediate layer of the hyaline cartilage and are seen as hyperechoic spots frequently described as rosary beads on US.6,49,50
Other important sites that can be evaluated to determine the presence of CPP crystals include the menisci, symphysis pubis, and triangular fibrocartilage at the wrists, hips, and shoulders. Recent EULAR recommendations have incorporated US as part of the diagnostic imaging modality for the diagnosis of CPPD and more recently for gout.6,51 Tophi are seen as MSU precipitates deposited in the joint cavity, tendons, and/or periarticular tissues such as bursae. They can show different echogenic signal. Soft tophi can demonstrate high PD signal due to high vascularization. On the other hand, hard tophi are hyperechoic on B-mode due to the presence of calcification, which does not allow passage of US waves, creating postacoustic shadowing.8 Studies have evaluated the predictive role of US in evaluating patients with asymptomatic hyperuricemia without any prior history of crystal-related joint disease and found tophaceous deposits in the triceps and patellar and quadriceps tendons.52-55 Studies have also looked at using US in the assessment of treatment response to serum uratelowering therapy in patients with gout.56,57 These studies have noted an improvement in the double contour sign, hyperechoic spots, cloudy areas in the synovial fluid, and tophus diameter and size in those patients who achieved a treat-to-target with a serum uric acid level ≤ 6 mg/dL. Patients who did not reach this target had no changes in the gout US features.56-57 Larger cohort studies are needed to confirm these findings.
An active inflammatory process can be determined by using a PD signal in the acute gout setting with increased vascularization; however, an increased PD signal can also be seen in septic arthritis or tenosynovitis, which sometimes can coexist with crystal-induced arthritis. Therefore, diagnostic arthrocentesis, Gram stain, and culture, as well as evaluation of crystals under polarized microscopy, are still recommended.
Therapeutic Interventions
Real-time visualization of the injection needle by US allows reliable placement of the needle tip in the tissue or cavity of interest. Multiple studies have shown the low accuracy of palpation-guided injection for reaching the site of interest.58,59 Some studies have shown a higher response rate to US-guided injections compared with palpation-guided as well as a higher rate of successful aspirations and clinical outcomes. Meta-analyses have demonstrated improved treatment response with the use of US-guided procedures compared with blinded injections.60,61 Ultrasound-guided interventions are performed in both peripheral and axial joints.62 The most common US-guided procedures at the VA rheumatology clinic include arthrocentesis and intra-articular corticosteroid injections of small and medium-sized joints, such as MCP joints, elbows, wrists, and ankles.
Conclusions
Ultrasound is becoming a relevant part of rheumatology practice and research and can be regarded as a feasible and effective imaging technique that can allow real-time recognition of early anatomical changes, provide careful guidance for aspiration, and monitor local and/or systemic treatment response at the joint, tendon, enthesis, nail, and skin levels. Ultrasound is a user-friendly imaging modality readily applied at the bedside and considered an extension of the rheumatologist's physical examination.
The success of US depends on the individual operator. For this reason, structured educational programs during fellowship training programs and an efficient competency assessment system would facilitate proper implementation of US in rheumatology practice as performed by some but not all institutions.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Over the past 2 decades, an increasing number of rheumatologists have progressively incorporated ultrasound (US) as an invaluable diagnostic and monitoring tool into their clinical and research practice.1,2 This imaging modality has become an established aid incorporated into the clinical evaluation of periarticular and articular structures involved in the diagnosis of several rheumatic disorders.
Ultrasound is a safe, noninvasive, patient-friendly imaging modality with a lack of contraindications and free of ionizing radiation. It allows real-time evaluation with dynamic assessment in a multiplanar view, assessment of multiple targets, and lower cost compared with magnetic resonance imaging (MRI) or computerized tomography scan. Above all, for the rheumatologist, US provides real-time scanning of all peripheral joints as many times as is required at the time of consultation. It is of great advantage in the assessment of a wide spectrum of abnormalities in rheumatic diseases with the potential of point-of-care imaging modality in the clinical evaluation and management of the patient. It facilitates a direct correlation between imaging findings and clinical data that improves the approach to a wide range of rheumatic diseases, from acute to chronic inflammatory arthritis, crystalline arthropathies, osteoarthritis (OA), spondyloarthropathies (SpA), vasculitis, and soft tissue syndromes. In addition, US is a bedside tool for performing accurate and safe diagnostic arthrocentesis, injections, and synovial biopsies.3,4
Recently, a gradual attempt has been made to incorporate US into rheumatology disease classification or diagnostic criteria for rheumatoid arthritis (RA), polymyalgia rheumatica, gout, calcium pyrophosphate deposition disease (CPPD), and Sjögren’s syndrome.5-10 Furthermore, the American College of Rheumatology (ACR) and the European League Against Rheumatism (EULAR) have produced evidence and expert opinion-based recommendations on the use of US in the clinical management of rheumatic diseases.10-12 This article highlights the most common applications of US for assessment and management of different rheumatic diseases frequently encountered at the VAMC rheumatology inpatient and outpatient clinical service.
Evaluation of Inflammatory Arthritis
In RA and any other inflammatory arthritis, US has been used for the detection of joint effusions, synovitis, bone erosions, and tendon and enthesis involvement.11,12 Ultrasound B-mode and power Doppler (PD) techniques have demonstrated a consistent and relevant role in optimizing the diagnosis, assessing the inflammatory activity, monitoring response to therapy, and predicting the inflammatory arthritis outcomes (Figures 1-3).10-12 Ultrasound provides real-time information about the status of the synovial membrane, tendons, cartilage, bursae, and cortical bones, allowing an accurate assessment of the degree of inflammatory process in periarticular and articular tissues. Also, US can provide details about the characteristics of the collected fluid (ie, effusion or synovial hypertrophy), which is fundamental for the correct interpretation of the pathologic joint and/or soft tissue processes. The inflammatory process can be assessed by using PD mode, which detects and quantifies the vascular changes in the pannus due to vasodilation and the increased blood flow characteristic of active inflammation.13,14
The Outcomes Measures in Rheumatoid Arthritis Clinical Trials (OMERACT) study group developed standardized sonopathologic definitions and scanning methods to be used in the daily rheumatologic practice and clinical trials (Table 1).15 Furthermore, it developed a semiquantitative scale to assess the degree of synovitis in US B-mode and PD mode (Table 2).15
The use of US to find subclinical synovitis in patients with RA considered to be in clinical remission is a new issue.16 Some reports have demonstrated progressive joint damage in these patients with evidence of active inflammation on PDUS despite clinical remission.17,18 More prospective studies are required to provide a better understanding of the long-term effects of residual inflammation and the proper long-term treatment of these patients. Furthermore, the PD signal has been shown to be superior to the Disease Activity Score 28 (DAS-28) in evaluating disease activity, particularly in predicting joint damage.18
Ultrasound may be considered the gold standard imaging tool for the assessment of tendons in inflammatory arthritis and includes the detection of tenosynovitis and anatomical damage represented by the loss of the normal fibrillar echotexture and loss of definition of the tendon margins, which may occur in early disease.19,20 Tenosynovitis of the extensor carpi ulnaris (ECU) detected by US has been shown to be an independent predictive factor of erosive joint damage, suggesting that ECU tenosynovitis represents a useful ultrasonographic landmark in the diagnosis of early RA.21
The availability of new nonbiologic and biologic therapies for inflammatory arthritis has raised the importance of identifying early changes, such as the detection of early erosions, which portend a poor long-term prognosis. The capability of US in identifying this lesion at an earlier stage compared with conventional radiography (CR) has allowed the early diagnosis and treatment of these patients before irreversible joint destruction occurs.22 In spite of all the supportive evidence of US utility in RA, it is not considered among the mandatory diagnostic criteria in the ACR/EULAR classification criteria for RA.5 Still, the addition of US findings to these criteria has increased the number of patients who fulfilled the 1987 ACR classification criteria for RA after 18 months of follow-up.23 Despite extensive evidence of its utility in the diagnosis and monitoring of RA, further studies are still needed.
Spondyloarthritis
Similar to RA, SpA discloses sonographic findings of inflammatory arthritis; however, with more entheseal and tenosynovium involvement. Ultrasound has also been used in the early identification of characteristic changes of the skin and nail tissues, which can aid the global assessment of this heterogeneous disease, especially in psoriatic arthritis (PsA). The most common locations of enthesitis in SpA are the quadriceps and the Achilles enthesis.24,25
Although US offers detailed imaging for the assessment of both tendons and enthesis, there is a lack of literature evaluating dactylitis. The OMERACT group recently released a composite measure of activity and severity of US dactylitis, which included newly defined elementary US lesions that may discern dactylitis of a digit.26,27 Ultrasound has been compared with MRI in the detection of SpA-related synovitis of the hands and feet and has demonstrated competitive diagnostic sensitivity.28 Ultrasound also shows higher sensitivity in detecting synovitis of the hands and feet compared with clinical examination and CR in PsA.28,29 Unfortunately, there are no strongly validated US findings that can aid in the differential diagnosis of PsA against other chronic inflammatory arthritides. The presence of peritendinous extensor tendon inflammation was a highly specific sonographic feature of PsA, because it was present in 66% of metacarpophalangeal (MCP) joints as the only US sign of inflammation compared with patients with RA.30
Another application of US is in the evaluation of subclinical inflammation at the enthesis in patients with a history of psoriasis without prior history of PsA.27,31 In those patients with psoriatic nail changes, more subclinical enthesitis was found compared with patients with psoriasis without nail involvement.32 Furthermore, subclinical joint inflammation has also been described.33 These findings suggest a possible predictive value in patients with psoriasis who should be monitored on a regular basis, because they are at risk of developing PsA.
Subclinical enthesitis by US imaging has been described in patients with recurrent anterior uveitis and inflammatory bowel disease.34,35 In cases where SpA is suspected but diagnostic criteria are not fulfilled, the presence of one enthesis with increased PD signal highly predicts the eventual development of SpA.36 Therefore, B-mode and PD evaluations of the entheses are critical in the identification of patients who are at an increased risk of developing SpA.37 Treatment monitoring is performed by using a US scoring system in a follow-up evaluation of patients with PsA. Some of the scoring systems have evaluated changes in B-mode US lesions (enthesis and soft tissues, such as skin and nails), whereas others focus on changes in the PD signal.37,38
The Five Targets Power Doppler for Psoriatic Disease PD scoring system comprises the assessment of PD signal in the joint, tendon with synovial sheath, enthesis, skin, and nails. Each of the targets is scored from 0 to 3 points, with a maximum of 15 total points. Some studies have shown that PDUS can provide valuable information in the evaluation of psoriatic plaques and onychopathy in patients with psoriasis and PsA.39 The detection of a PD signal within the dermis and nail bed is equivalent to active inflammation in these sites.39-41 However, further studies with larger cohorts proving inter- and intra-observer reliability are necessary to consolidate these findings and comfortably apply them in clinical practice.
Osteoarthritis
Increasingly US is studied for its validity and reliability in evaluating periarticular soft tissue and cartilage changes in knee OA. The associated US findings include a high prevalence of synovitis with a low prevalence of a PD signal, the presence of osteophytes, and joint space narrowing.42,43 Increased PD signal, synovial hypertrophy, and joint effusion were observed in patients with radiographically erosive OA compared with those with radiographically nonerosive OA.44
Bone erosions and inflammatory changes are also frequently detected by US in both erosive and nodal hand OA.45 Compared with MRI, US has shown a good to excellent correlation in the assessment of osteophytes, bone erosions, synovitis, and tenosynovitis in erosive and hand nodal OA.46 In comparison with CR, US has shown to have a higher sensitivity in the assessment of bony erosions, osteophytes, and space narrowing.47 Ultrasound is able to detect changes in the earlier stages of cartilage erosion in OA, characterized by loss of the sharp contour and variations in the echogenicity of the cartilage matrix, asymmetric shrinkage, and ultimately the disappearance of the cartilaginous band, which is more evident in the later stages of OA.45
Similar to RA management, US has been used to monitor disease activity and response to OA treatment. Patients who received intra-articular hyaluronic acid or intramuscular methylprednisolone for OA treatment were found to have a decrease of PD signal intensity and synovial effusion posttreatment.48 One could extrapolate these findings and conclude that US could be an additional tool for monitoring disease activity and assessing response to local and systemic treatments in OA.
Crystalline Arthropathies
Ultrasound application to crystal diseases facilitates the identification of microcrystalline deposits within the synovial membrane (joints), cartilage (both hyaline cartilage and fibrocartilage), and periarticular tissues (tendons, bursae, and soft tissues). Crystals appear as hyperechogenic spots of different sizes and shapes that can be seen in both articular and periarticular tissues.49,50 The crystal deposition pattern on hyaline cartilage allows the differentiation between monosodium urate (MSU) and calcium pyrophosphate dehydrate (CPP) crystals. The MSU crystals are deposited at the chondrosynovial (or superficial) margin of the hyaline cartilage and described sonographically as the double contour sign in gout, whereas CPP crystals are deposited within the intermediate layer of the hyaline cartilage and are seen as hyperechoic spots frequently described as rosary beads on US.6,49,50
Other important sites that can be evaluated to determine the presence of CPP crystals include the menisci, symphysis pubis, and triangular fibrocartilage at the wrists, hips, and shoulders. Recent EULAR recommendations have incorporated US as part of the diagnostic imaging modality for the diagnosis of CPPD and more recently for gout.6,51 Tophi are seen as MSU precipitates deposited in the joint cavity, tendons, and/or periarticular tissues such as bursae. They can show different echogenic signal. Soft tophi can demonstrate high PD signal due to high vascularization. On the other hand, hard tophi are hyperechoic on B-mode due to the presence of calcification, which does not allow passage of US waves, creating postacoustic shadowing.8 Studies have evaluated the predictive role of US in evaluating patients with asymptomatic hyperuricemia without any prior history of crystal-related joint disease and found tophaceous deposits in the triceps and patellar and quadriceps tendons.52-55 Studies have also looked at using US in the assessment of treatment response to serum uratelowering therapy in patients with gout.56,57 These studies have noted an improvement in the double contour sign, hyperechoic spots, cloudy areas in the synovial fluid, and tophus diameter and size in those patients who achieved a treat-to-target with a serum uric acid level ≤ 6 mg/dL. Patients who did not reach this target had no changes in the gout US features.56-57 Larger cohort studies are needed to confirm these findings.
An active inflammatory process can be determined by using a PD signal in the acute gout setting with increased vascularization; however, an increased PD signal can also be seen in septic arthritis or tenosynovitis, which sometimes can coexist with crystal-induced arthritis. Therefore, diagnostic arthrocentesis, Gram stain, and culture, as well as evaluation of crystals under polarized microscopy, are still recommended.
Therapeutic Interventions
Real-time visualization of the injection needle by US allows reliable placement of the needle tip in the tissue or cavity of interest. Multiple studies have shown the low accuracy of palpation-guided injection for reaching the site of interest.58,59 Some studies have shown a higher response rate to US-guided injections compared with palpation-guided as well as a higher rate of successful aspirations and clinical outcomes. Meta-analyses have demonstrated improved treatment response with the use of US-guided procedures compared with blinded injections.60,61 Ultrasound-guided interventions are performed in both peripheral and axial joints.62 The most common US-guided procedures at the VA rheumatology clinic include arthrocentesis and intra-articular corticosteroid injections of small and medium-sized joints, such as MCP joints, elbows, wrists, and ankles.
Conclusions
Ultrasound is becoming a relevant part of rheumatology practice and research and can be regarded as a feasible and effective imaging technique that can allow real-time recognition of early anatomical changes, provide careful guidance for aspiration, and monitor local and/or systemic treatment response at the joint, tendon, enthesis, nail, and skin levels. Ultrasound is a user-friendly imaging modality readily applied at the bedside and considered an extension of the rheumatologist's physical examination.
The success of US depends on the individual operator. For this reason, structured educational programs during fellowship training programs and an efficient competency assessment system would facilitate proper implementation of US in rheumatology practice as performed by some but not all institutions.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Naredo E, D’Agostino MA, Conaghan PG, et al. Current state of musculoskeletal ultrasound training and implementation in Europe: results of a survey of experts and scientific societies. Rheumatology (Oxford). 2010;49(12):2438-2943.
2. Micu MC, Alcalde M, Sáenz JI, et al. Impact of musculoskeletal ultrasound in an outpatient rheumatology clinic. Arthritis Care Res (Hoboken). 2013;65(4):615-621.
3. Koski JM. Ultrasound guided injections in rheumatology. J Rheumatol. 2000;27(9):2131-2138.
4. Kelly S, Humby F, Filer A, et al. Ultrasound-guided synovial biopsy: a safe, well-tolerated and reliable technique for obtaining high-quality synovial tissue from both large and small joints in early arthritis patients. Ann Rheum Dis. 2015;74(3):611-617.
5. Aletaha D, Neogi T, Silman AJ, et al. 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum Dis. 2010;69(9):1580-1588.
6. Zhang W, Doherty M, Bardin T, et al. European League Against Rheumatism recommendations for calcium pyrophosphate deposition. Part I: terminology and diagnosis. Ann Rheum Dis. 2011;70(4):563-570.
7. Dasgupta B, Cimmino MA, Kremers HM, et al. 2012 provisional classification criteria for polymyalgia rheumatica: a European League Against Rheumatism/ American College of Rheumatology collaborative initiative. Arthritis Rheum. 2012;64(4):943-954.
8. Fodor D, Nestorova R, Vlad V, Micu M. The place of musculoskeletal ultrasonography in gout diagnosis. Med Ultrason. 2014;16(4):336-344.
9. Takagi Y, Sumi M, Nakamura H, et al. Ultrasonography as an additional item in the American College of Rheumatology classification of Sjögren’s syndrome. Rheumatology (Oxford). 2014;53(11):1977-1983.
10. Colebatch AN, Edwards CJ, Østergaard M, et al. EULAR recommendations for the use of imaging of the joints in the clinical management of rheumatoid arthritis. Ann Rheum Dis. 2013;72(6):804-814.
11. American College of Rheumatology Musculoskeletal Ultrasound Task Force. Ultrasound in American rheumatology practice: report of the American College of Rheumatology musculoskeletal ultrasound task force. Arthritis Care Res (Hoboken). 2010;62(9):1206-1219.
12. McAlindon T, Kissin E, Nazarian L, et al. American College of Rheumatology report on reasonable use of musculoskeletal ultrasonography in rheumatology clinical practice. Arthritis Care Res (Hoboken). 2012;64(11):1625-1640.
13. Naredo E, Möller I, Cruz A, Carmona L, Garrido J. Power Doppler ultrasonographic monitoring of response to anti-tumor necrosis factor therapy in patients with rheumatoid arthritis. Arthritis Rheum. 2008;58(8):2248-2256.
14. Newman JS, Laing TJ, McCarthy CJ, Adler RS. Power Doppler sonography of synovitis: assessment of therapeutic response—preliminary observations. Radiology. 1996;198(2):582-584.
15. Wakefield RJ, Balint PV, Szkudlarek M, et al; OMERACT 7 Special Interest Group. Musculoskeletal ultrasound including definitions for ultrasonographic pathology. J Rheumatol. 2005;32(12):2485-2487.
16. Wakefield RJ, Green MJ, Marzo-Ortega H, et al. Should oligoarthritis be reclassified? Ultrasound reveals a high prevalence of subclinical disease. Ann Rheum Dis. 2004;63(4):382-385.
17. Brown AK, Quinn MA, Karim Z, et al. Presence of significant synovitis in rheumatoid arthritis patients with disease-modifying antirheumatic drug-induced clinical remission: evidence from an imaging study may explain structural progression. Arthritis Rheum. 2006;54(12):3761-3773.
18. Brown AK, Conaghan PG, Karim Z, et al. An explanation for the apparent dissociation between clinical remission and continued structural deterioration in rheumatoid arthritis. Arthritis Rheum. 2008;58(10):2958-2967.
19. Bruyn GA, Hanova P, Iagnocco A, et al; OMERACT Ultrasound Task Force. Ultrasound definition of tendon damage in patients with rheumatoid arthritis. Results of a OMERACT consensus-based ultrasound score focusing on the diagnostic reliability. Ann Rheum Dis. 2014;73(11):1929-1934.
20. Filippucci E, Gabba A, Di Geso L, Girolimetti R, Salaffi F, Grassi W. Hand tendon involvement in rheumatoid arthritis: an ultrasound study. Semin Arthritis Rheum. 2012;41(6):752-760.
21. Lillegraven S, Bøyesen P, Hammer HB, et al. Tenosynovitis of the extensor carpi ulnaris tendon predicts erosive progression in early rheumatoid arthritis. Ann Rheum Dis. 2011;70(11):2049-2050.
22. Baillet A, Gaujoux-Viala C, Mouterde G, et al. Comparison of the efficacy of sonography, magnetic resonance imaging and conventional radiography for the detection of bone erosions in rheumatoid arthritis patients: a systematic review and meta-analysis. Rheumatology (Oxford). 2011;50(6):1137-1147.
23. Filer A, de Pablo P, Allen G, et al. Utility of ultrasound joint counts in the prediction of rheumatoid arthritis in patients with very early synovitis. Ann Rheum Dis. 2011;70(3):500-507.
24. Frediani B, Falsetti P, Storri L, et al. Quadricepital tendon enthesitis in psoriatic arthritis and rheumatoid arthritis: ultrasound examinations and clinical correlations. J Rheumatol. 2001;28(11):2566-2568.
25. D’Agostino MA, Said-Nahal R, Hacquard-Bouder C, Brasseur JL, Dougados M, Breban M. Assessment of peripheral enthesitis in the spondylarthropathies by ultrasonography combined with power Doppler: a cross-sectional study. Arthritis Rheum. 2003;48(2):523-533.
26. Gisondi P, Tinazzi I, El-Dalati G, et al. Lower limb enthesopathy in patients with psoriasis without clinical signs of arthropathy: a hospital-based case-control study. Ann Rheum Dis. 2008;67(1):26-30.
27. Gutierrez M, Filippucci E, De Angelis R, et al. Subclinical entheseal involvement
in patients with psoriasis: an ultrasound study. Semin Arthritis Rheum. 2011;40(5):407-412.
28. Weiner SM, Jurenz S, Uhl M, et al. Ultrasonography in the assessment of peripheral joint involvement in psoriatic arthritis: a comparison with radiography, MRI and scintigraphy. Clin Rheumatol. 2008;27(8):983-989.
29. Balint PV, Kane D, Wilson H, McInnes IB, Sturrock RD. Ultrasonography of entheseal insertions in the lower limb in spondyloarthropathy. Ann Rheum Dis. 2002;61(10):905-910.
30. Gutierrez M, Filippucci E, Salaffi F, Di Geso L, Grassi W. Differential diagnosis
between rheumatoid arthritis and psoriatic arthritis: the value of ultrasound findings at metacarpophalangeal joints level. Ann Rheum Dis. 2011;70(6):1111-1114.
31. De Miguel E, Cobo T, Muñoz-Fernández S, et al. Validity of enthesis ultrasound assessment in spondyloarthropathy. Ann Rheum Dis. 2009;68(2):169-174.
32. Ash ZR, Tinazzi I, Gallego CC, et al. Psoriasis patients with nail disease have a greater magnitude of underlying systemic subclinical enthesopathy than those with normal nails. Ann Rheum Dis. 2012;71(4):553-556.
33. Naredo E, Möller I, de Miguel E, et al; Ultrasound School of the Spanish Society of Rheumatology and Spanish ECO-APs Group. High prevalence of ultrasonographic synovitis and enthesopathy in patients with psoriasis without psoriatic arthritis: a prospective case-control study. Rheumatology (Oxford). 2011;50(10):1838-1848.
34. Muñoz-Fernández S, de Miguel E, Cobo-Ibáñez T, et al. Enthesis inflammation in recurrent acute anterior uveitis without spondylarthritis. Arthritis Rheum. 2009;60(7):1985-1990.
35. Bandinelli F, Milla M, Genise S, et al. Ultrasound discloses entheseal involvement
in inactive and low active inflammatory bowel disease without clinical signs and symptoms of spondyloarthropathy. Rheumatology (Oxford). 2011;50(7):1275-1279.
36. D’Agostino MA, Aegerter P, Bechara K, et al. How to diagnose spondyloarthritis early? Accuracy of peripheral enthesitis detection by power Doppler ultrasonography. Ann Rheum Dis. 2011;70(8):1433-1440.
37. Aydin SZ, Karadag O, Filippucci E, et al. Monitoring Achilles enthesitis in ankylosing spondylitis during TNF-alpha antagonist therapy: an ultrasound study. Rheumatology (Oxford). 2010;49(3):578-582.
38. Naredo E, Batlle-Gualda E, Garcia-Vivar ML, et al; Ultrasound Group of the Spanish Society of Rheumatology. Power Doppler ultrasonography assessment of entheses in spondyloarthropathies: response to therapy of entheseal abnormalities. J Rheumatol. 2010;37(10):2110-2117.
39. Gutierrez M, Di Geso L, Salaffi F, et al. Development of a preliminary US power Doppler composite score for monitoring treatment in PsA. Rheumatology (Oxford). 2012;51(7):1261-1268.
40. Gutierrez M, De Angelis R, Bernardini ML, et al. Clinical, power Doppler sonography and histological assessment of the psoriatic plaque: short-term monitoring in patients treated with etanercept. Br J Dermatol. 2011;164(1):33-37.
41. Gutierrez M, Filippucci E, Bertolazzi C, Grassi W. Sonographic monitoring of psoriatic plaque. J Rheumatol. 2009;36(4):850-851.
42. Keen HI, Wakefield RJ, Grainger AJ, Hensor EM, Emery P, Conaghan PG. An ultrasonographic study of osteoarthritis of the hand: synovitis and its relationship to structural pathology and symptoms. Arthritis Rheum. 2008;59(12):1756-1763.
43. Kortekaas MC, Kwok WY, Reijnierse M, Watt I, Huizinga TW, Kloppenburg M. Pain in hand osteoarthritis is associated with inflammation: the value of ultrasound. Ann Rheum Dis. 2010;69(7):1367-1369.
44. Mancarella L, Magnani M, Addimanda O, Pignotti E, Galletti S, Meliconi R. Ultrasound-detected synovitis with power Doppler signal is associated with severe radiographic damage and reduced cartilage thickness in hand osteoarthritis. Osteoarthritis Cartilage. 2010;18(10):1263-1268.
45. Möller I, Bong D, Naredo E, et al. Ultrasound in the study and monitoring of osteoarthritis. Osteoarthritis Cartilage. 2008;16(suppl 3):S4-S7.
46. Vlychou M, Koutroumpas A, Alexiou I, Fezoulidis I, Sakkas LI. High-resolution ultrasonography and 3.0 T magnetic resonance imaging in erosive and nodal hand osteoarthritis: high frequency of erosions in nodal osteoarthritis. Clin Rheumatol. 2013;32(6):755-762.
47. Keen HI, Wakefield RJ, Grainger AJ, Hensor EM, Emery P, Conaghan PG. Can ultrasonography improve on radiographic assessment in osteoarthritis of the hands? A comparison between radiographic and ultrasonographic detected pathology. Ann Rheum Dis. 2008;67(8):1116-1120.
48. Keen HI, Wakefield RJ, Hensor EM, Emery P, Conaghan PG. Response of symptoms
and synovitis to intra-muscular methylprednisolone in osteoarthritis of the hand: an ultrasonographic study. Rheumatology (Oxford). 2010;49(6):1093-1100.
49. Grassi W, Meenagh G, Pascual E, Filippucci E. “Crystal clear”-sonographic assessment of gout and calcium pyrophosphate deposition disease. Semin Arthritis Rheum. 2006;36(3):197-202.
50. Ciapetti A, Filippucci E, Gutierrez M, Grassi W. Calcium pyrophosphate dihydrate crystal deposition disease: sonographic findings. Clin Rheumatol. 2009;28(3):271-276.
51. Taylor WJ, Fransen J, Jansen TL, et al. Study for Updated Gout Classification Criteria (SUGAR): identification of features to classify gout. Arthritis Care Res (Hoboken). [Published online ahead of print March 16, 2015.]
52. Puig JG, de Miguel E, Castillo MC, Rocha AL, Martinez MA, Torres RJ. Asymptomatic hyperuricemia: impact of ultrasonography. Nucleosides Nucleotides Nucleic Acids. 2008;27(6):592-595.
53. Pineda C, Amezcua-Guerra LM, Solano C, et al. Joint and tendon subclinical involvement suggestive of gouty arthritis in asymptomatic hyperuricemia: an ultrasound controlled study. Arthritis Res Ther. 2011;13(1):R4.
54. Naredo E, Uson J, Jiménez-Palop M, et al. Ultrasound-detected musculoskeletal urate crystal deposition: which joints and what findings should be assessed for diagnosing gout? Ann Rheum Dis. 2014;73(8):1522-1528.
55. De Miguel E, Puig JG, Castillo C, Peiteado D, Torres RJ, Martín-Mola E. Diagnosis of gout in patients with asymptomatic hyperuricaemia: a pilot ultrasound study. Ann Rheum Dis. 2012;71(1):157-158.
56. Perez-Ruiz F, Martin I, Canteli B. Ultrasonographic measurement of tophi as an outcome measure for chronic gout. J Rheumatol. 2007;34(9):1888-1893.
57. Thiele RG, Schlesinger N. Ultrasonography shows disappearance of monosodium urate crystal deposition on hyaline cartilage after sustained normouricemia is achieved. Rheumatol Int. 2010;30(4):495-503.
58. Balint PV, Kane D, Hunter J, McInnes IB, Field M, Sturrock RD. Ultrasound guided versus conventional joint and soft tissue fluid aspiration in rheumatology practice: a pilot study. J Rheumatol. 2002;29(10):2209-2213.
59. Raza K, Lee CY, Pilling D, et al. Ultrasound guidance allows accurate needle placement and aspiration from small joints in patients with early inflammatory arthritis. Rheumatology (Oxford). 2003;42(8):976-979.
60. Dubreuil M, Greger S, LaValley M, Cunnington J, Sibbitt WL Jr, Kissin EY. Improvement in wrist pain with ultrasound-guided glucocorticoid injections: a metaanalysis of individual patient data. Semin Arthritis Rheum. 2013;42(5):492-497.
61. Sage W, Pickup L, Smith TO, Denton ER, Toms AP. The clinical and functional outcomes of ultrasound-guided vs landmark-guided injections for adults with shoulder pathology—a systematic review and meta-analysis. Rheumatology (Oxford). 2013;52(4):743-751.
62. Darrieutort-Laffite C, Hamel O, Glémarec J, Maugars Y, Le Goff B. Ultrasonography of the lumbar spine: sonoanatomy and practical applications. Joint Bone Spine. 2014;81(2):130-136.
1. Naredo E, D’Agostino MA, Conaghan PG, et al. Current state of musculoskeletal ultrasound training and implementation in Europe: results of a survey of experts and scientific societies. Rheumatology (Oxford). 2010;49(12):2438-2943.
2. Micu MC, Alcalde M, Sáenz JI, et al. Impact of musculoskeletal ultrasound in an outpatient rheumatology clinic. Arthritis Care Res (Hoboken). 2013;65(4):615-621.
3. Koski JM. Ultrasound guided injections in rheumatology. J Rheumatol. 2000;27(9):2131-2138.
4. Kelly S, Humby F, Filer A, et al. Ultrasound-guided synovial biopsy: a safe, well-tolerated and reliable technique for obtaining high-quality synovial tissue from both large and small joints in early arthritis patients. Ann Rheum Dis. 2015;74(3):611-617.
5. Aletaha D, Neogi T, Silman AJ, et al. 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum Dis. 2010;69(9):1580-1588.
6. Zhang W, Doherty M, Bardin T, et al. European League Against Rheumatism recommendations for calcium pyrophosphate deposition. Part I: terminology and diagnosis. Ann Rheum Dis. 2011;70(4):563-570.
7. Dasgupta B, Cimmino MA, Kremers HM, et al. 2012 provisional classification criteria for polymyalgia rheumatica: a European League Against Rheumatism/ American College of Rheumatology collaborative initiative. Arthritis Rheum. 2012;64(4):943-954.
8. Fodor D, Nestorova R, Vlad V, Micu M. The place of musculoskeletal ultrasonography in gout diagnosis. Med Ultrason. 2014;16(4):336-344.
9. Takagi Y, Sumi M, Nakamura H, et al. Ultrasonography as an additional item in the American College of Rheumatology classification of Sjögren’s syndrome. Rheumatology (Oxford). 2014;53(11):1977-1983.
10. Colebatch AN, Edwards CJ, Østergaard M, et al. EULAR recommendations for the use of imaging of the joints in the clinical management of rheumatoid arthritis. Ann Rheum Dis. 2013;72(6):804-814.
11. American College of Rheumatology Musculoskeletal Ultrasound Task Force. Ultrasound in American rheumatology practice: report of the American College of Rheumatology musculoskeletal ultrasound task force. Arthritis Care Res (Hoboken). 2010;62(9):1206-1219.
12. McAlindon T, Kissin E, Nazarian L, et al. American College of Rheumatology report on reasonable use of musculoskeletal ultrasonography in rheumatology clinical practice. Arthritis Care Res (Hoboken). 2012;64(11):1625-1640.
13. Naredo E, Möller I, Cruz A, Carmona L, Garrido J. Power Doppler ultrasonographic monitoring of response to anti-tumor necrosis factor therapy in patients with rheumatoid arthritis. Arthritis Rheum. 2008;58(8):2248-2256.
14. Newman JS, Laing TJ, McCarthy CJ, Adler RS. Power Doppler sonography of synovitis: assessment of therapeutic response—preliminary observations. Radiology. 1996;198(2):582-584.
15. Wakefield RJ, Balint PV, Szkudlarek M, et al; OMERACT 7 Special Interest Group. Musculoskeletal ultrasound including definitions for ultrasonographic pathology. J Rheumatol. 2005;32(12):2485-2487.
16. Wakefield RJ, Green MJ, Marzo-Ortega H, et al. Should oligoarthritis be reclassified? Ultrasound reveals a high prevalence of subclinical disease. Ann Rheum Dis. 2004;63(4):382-385.
17. Brown AK, Quinn MA, Karim Z, et al. Presence of significant synovitis in rheumatoid arthritis patients with disease-modifying antirheumatic drug-induced clinical remission: evidence from an imaging study may explain structural progression. Arthritis Rheum. 2006;54(12):3761-3773.
18. Brown AK, Conaghan PG, Karim Z, et al. An explanation for the apparent dissociation between clinical remission and continued structural deterioration in rheumatoid arthritis. Arthritis Rheum. 2008;58(10):2958-2967.
19. Bruyn GA, Hanova P, Iagnocco A, et al; OMERACT Ultrasound Task Force. Ultrasound definition of tendon damage in patients with rheumatoid arthritis. Results of a OMERACT consensus-based ultrasound score focusing on the diagnostic reliability. Ann Rheum Dis. 2014;73(11):1929-1934.
20. Filippucci E, Gabba A, Di Geso L, Girolimetti R, Salaffi F, Grassi W. Hand tendon involvement in rheumatoid arthritis: an ultrasound study. Semin Arthritis Rheum. 2012;41(6):752-760.
21. Lillegraven S, Bøyesen P, Hammer HB, et al. Tenosynovitis of the extensor carpi ulnaris tendon predicts erosive progression in early rheumatoid arthritis. Ann Rheum Dis. 2011;70(11):2049-2050.
22. Baillet A, Gaujoux-Viala C, Mouterde G, et al. Comparison of the efficacy of sonography, magnetic resonance imaging and conventional radiography for the detection of bone erosions in rheumatoid arthritis patients: a systematic review and meta-analysis. Rheumatology (Oxford). 2011;50(6):1137-1147.
23. Filer A, de Pablo P, Allen G, et al. Utility of ultrasound joint counts in the prediction of rheumatoid arthritis in patients with very early synovitis. Ann Rheum Dis. 2011;70(3):500-507.
24. Frediani B, Falsetti P, Storri L, et al. Quadricepital tendon enthesitis in psoriatic arthritis and rheumatoid arthritis: ultrasound examinations and clinical correlations. J Rheumatol. 2001;28(11):2566-2568.
25. D’Agostino MA, Said-Nahal R, Hacquard-Bouder C, Brasseur JL, Dougados M, Breban M. Assessment of peripheral enthesitis in the spondylarthropathies by ultrasonography combined with power Doppler: a cross-sectional study. Arthritis Rheum. 2003;48(2):523-533.
26. Gisondi P, Tinazzi I, El-Dalati G, et al. Lower limb enthesopathy in patients with psoriasis without clinical signs of arthropathy: a hospital-based case-control study. Ann Rheum Dis. 2008;67(1):26-30.
27. Gutierrez M, Filippucci E, De Angelis R, et al. Subclinical entheseal involvement
in patients with psoriasis: an ultrasound study. Semin Arthritis Rheum. 2011;40(5):407-412.
28. Weiner SM, Jurenz S, Uhl M, et al. Ultrasonography in the assessment of peripheral joint involvement in psoriatic arthritis: a comparison with radiography, MRI and scintigraphy. Clin Rheumatol. 2008;27(8):983-989.
29. Balint PV, Kane D, Wilson H, McInnes IB, Sturrock RD. Ultrasonography of entheseal insertions in the lower limb in spondyloarthropathy. Ann Rheum Dis. 2002;61(10):905-910.
30. Gutierrez M, Filippucci E, Salaffi F, Di Geso L, Grassi W. Differential diagnosis
between rheumatoid arthritis and psoriatic arthritis: the value of ultrasound findings at metacarpophalangeal joints level. Ann Rheum Dis. 2011;70(6):1111-1114.
31. De Miguel E, Cobo T, Muñoz-Fernández S, et al. Validity of enthesis ultrasound assessment in spondyloarthropathy. Ann Rheum Dis. 2009;68(2):169-174.
32. Ash ZR, Tinazzi I, Gallego CC, et al. Psoriasis patients with nail disease have a greater magnitude of underlying systemic subclinical enthesopathy than those with normal nails. Ann Rheum Dis. 2012;71(4):553-556.
33. Naredo E, Möller I, de Miguel E, et al; Ultrasound School of the Spanish Society of Rheumatology and Spanish ECO-APs Group. High prevalence of ultrasonographic synovitis and enthesopathy in patients with psoriasis without psoriatic arthritis: a prospective case-control study. Rheumatology (Oxford). 2011;50(10):1838-1848.
34. Muñoz-Fernández S, de Miguel E, Cobo-Ibáñez T, et al. Enthesis inflammation in recurrent acute anterior uveitis without spondylarthritis. Arthritis Rheum. 2009;60(7):1985-1990.
35. Bandinelli F, Milla M, Genise S, et al. Ultrasound discloses entheseal involvement
in inactive and low active inflammatory bowel disease without clinical signs and symptoms of spondyloarthropathy. Rheumatology (Oxford). 2011;50(7):1275-1279.
36. D’Agostino MA, Aegerter P, Bechara K, et al. How to diagnose spondyloarthritis early? Accuracy of peripheral enthesitis detection by power Doppler ultrasonography. Ann Rheum Dis. 2011;70(8):1433-1440.
37. Aydin SZ, Karadag O, Filippucci E, et al. Monitoring Achilles enthesitis in ankylosing spondylitis during TNF-alpha antagonist therapy: an ultrasound study. Rheumatology (Oxford). 2010;49(3):578-582.
38. Naredo E, Batlle-Gualda E, Garcia-Vivar ML, et al; Ultrasound Group of the Spanish Society of Rheumatology. Power Doppler ultrasonography assessment of entheses in spondyloarthropathies: response to therapy of entheseal abnormalities. J Rheumatol. 2010;37(10):2110-2117.
39. Gutierrez M, Di Geso L, Salaffi F, et al. Development of a preliminary US power Doppler composite score for monitoring treatment in PsA. Rheumatology (Oxford). 2012;51(7):1261-1268.
40. Gutierrez M, De Angelis R, Bernardini ML, et al. Clinical, power Doppler sonography and histological assessment of the psoriatic plaque: short-term monitoring in patients treated with etanercept. Br J Dermatol. 2011;164(1):33-37.
41. Gutierrez M, Filippucci E, Bertolazzi C, Grassi W. Sonographic monitoring of psoriatic plaque. J Rheumatol. 2009;36(4):850-851.
42. Keen HI, Wakefield RJ, Grainger AJ, Hensor EM, Emery P, Conaghan PG. An ultrasonographic study of osteoarthritis of the hand: synovitis and its relationship to structural pathology and symptoms. Arthritis Rheum. 2008;59(12):1756-1763.
43. Kortekaas MC, Kwok WY, Reijnierse M, Watt I, Huizinga TW, Kloppenburg M. Pain in hand osteoarthritis is associated with inflammation: the value of ultrasound. Ann Rheum Dis. 2010;69(7):1367-1369.
44. Mancarella L, Magnani M, Addimanda O, Pignotti E, Galletti S, Meliconi R. Ultrasound-detected synovitis with power Doppler signal is associated with severe radiographic damage and reduced cartilage thickness in hand osteoarthritis. Osteoarthritis Cartilage. 2010;18(10):1263-1268.
45. Möller I, Bong D, Naredo E, et al. Ultrasound in the study and monitoring of osteoarthritis. Osteoarthritis Cartilage. 2008;16(suppl 3):S4-S7.
46. Vlychou M, Koutroumpas A, Alexiou I, Fezoulidis I, Sakkas LI. High-resolution ultrasonography and 3.0 T magnetic resonance imaging in erosive and nodal hand osteoarthritis: high frequency of erosions in nodal osteoarthritis. Clin Rheumatol. 2013;32(6):755-762.
47. Keen HI, Wakefield RJ, Grainger AJ, Hensor EM, Emery P, Conaghan PG. Can ultrasonography improve on radiographic assessment in osteoarthritis of the hands? A comparison between radiographic and ultrasonographic detected pathology. Ann Rheum Dis. 2008;67(8):1116-1120.
48. Keen HI, Wakefield RJ, Hensor EM, Emery P, Conaghan PG. Response of symptoms
and synovitis to intra-muscular methylprednisolone in osteoarthritis of the hand: an ultrasonographic study. Rheumatology (Oxford). 2010;49(6):1093-1100.
49. Grassi W, Meenagh G, Pascual E, Filippucci E. “Crystal clear”-sonographic assessment of gout and calcium pyrophosphate deposition disease. Semin Arthritis Rheum. 2006;36(3):197-202.
50. Ciapetti A, Filippucci E, Gutierrez M, Grassi W. Calcium pyrophosphate dihydrate crystal deposition disease: sonographic findings. Clin Rheumatol. 2009;28(3):271-276.
51. Taylor WJ, Fransen J, Jansen TL, et al. Study for Updated Gout Classification Criteria (SUGAR): identification of features to classify gout. Arthritis Care Res (Hoboken). [Published online ahead of print March 16, 2015.]
52. Puig JG, de Miguel E, Castillo MC, Rocha AL, Martinez MA, Torres RJ. Asymptomatic hyperuricemia: impact of ultrasonography. Nucleosides Nucleotides Nucleic Acids. 2008;27(6):592-595.
53. Pineda C, Amezcua-Guerra LM, Solano C, et al. Joint and tendon subclinical involvement suggestive of gouty arthritis in asymptomatic hyperuricemia: an ultrasound controlled study. Arthritis Res Ther. 2011;13(1):R4.
54. Naredo E, Uson J, Jiménez-Palop M, et al. Ultrasound-detected musculoskeletal urate crystal deposition: which joints and what findings should be assessed for diagnosing gout? Ann Rheum Dis. 2014;73(8):1522-1528.
55. De Miguel E, Puig JG, Castillo C, Peiteado D, Torres RJ, Martín-Mola E. Diagnosis of gout in patients with asymptomatic hyperuricaemia: a pilot ultrasound study. Ann Rheum Dis. 2012;71(1):157-158.
56. Perez-Ruiz F, Martin I, Canteli B. Ultrasonographic measurement of tophi as an outcome measure for chronic gout. J Rheumatol. 2007;34(9):1888-1893.
57. Thiele RG, Schlesinger N. Ultrasonography shows disappearance of monosodium urate crystal deposition on hyaline cartilage after sustained normouricemia is achieved. Rheumatol Int. 2010;30(4):495-503.
58. Balint PV, Kane D, Hunter J, McInnes IB, Field M, Sturrock RD. Ultrasound guided versus conventional joint and soft tissue fluid aspiration in rheumatology practice: a pilot study. J Rheumatol. 2002;29(10):2209-2213.
59. Raza K, Lee CY, Pilling D, et al. Ultrasound guidance allows accurate needle placement and aspiration from small joints in patients with early inflammatory arthritis. Rheumatology (Oxford). 2003;42(8):976-979.
60. Dubreuil M, Greger S, LaValley M, Cunnington J, Sibbitt WL Jr, Kissin EY. Improvement in wrist pain with ultrasound-guided glucocorticoid injections: a metaanalysis of individual patient data. Semin Arthritis Rheum. 2013;42(5):492-497.
61. Sage W, Pickup L, Smith TO, Denton ER, Toms AP. The clinical and functional outcomes of ultrasound-guided vs landmark-guided injections for adults with shoulder pathology—a systematic review and meta-analysis. Rheumatology (Oxford). 2013;52(4):743-751.
62. Darrieutort-Laffite C, Hamel O, Glémarec J, Maugars Y, Le Goff B. Ultrasonography of the lumbar spine: sonoanatomy and practical applications. Joint Bone Spine. 2014;81(2):130-136.