User login
Adjuvant Scalp Rolling for Patients With Refractory Alopecia Areata
To the Editor:
Alopecia areata (AA) is an autoimmune nonscarring hair loss disorder that can present at any age. Patients with AA have a disproportionately high comorbidity burden and low quality of life, often grappling with anxiety, depression, and psychosocial sequelae involving identity, such as reduced self-esteem.1,2 Although conventional therapies aim to reduce hair loss, none are curative.3 Response to treatment is highly unpredictable, with current data suggesting that up to 50% of patients recover within 1 year while 14% to 25% progress to either alopecia totalis (total scalp hair loss) or alopecia universalis (total body hair loss).4 Options for therapeutic intervention remain limited and vary in safety and effectiveness, warranting further research to identify optimal modalities and minimize side effects. Interestingly, scalp rolling has been used as an adjuvant to topical triamcinolone acetonide.3,5 However, the extent of its effect in combination with other therapies remains unclear. We report 3 pediatric patients with confirmed AA refractory to conventional topical treatment who experienced remarkable scalp hair regrowth after adding biweekly scalp rolling as an adjuvant therapy.
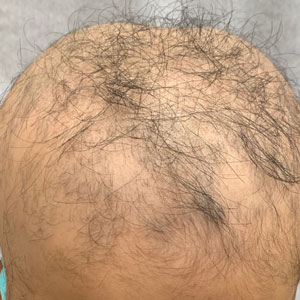
A 7-year-old boy with AA presented with 95% scalp hair loss of 7 months’ duration (Figure 1A)(patient 1). Prior treatments included mometasone solution and clobetasol solution 0.05%. After 3 months of conventional topical therapy, twice-weekly scalp rolling with a 0.25-mm scalp roller of their choosing was added to the regimen, with clobetasol solution 0.05% and minoxidil foam 5% applied immediately after each scalp rolling session. The patient experienced 95% scalp hair regrowth after 13 months of treatment (Figure 1B). No pain, bleeding, or other side effects were reported.

An 11-year-old girl with AA presented with 100% hair loss of 7 months’ duration (Figure 2A)(patient 2). Prior treatments included fluocinonide solution and intralesional Kenalog injections. After 4 months of conventional topical therapy, twice-weekly scalp rolling with a 0.25-mm scalp roller of their choosing was added to the regimen, with clobetasol solution 0.05% and minoxidil foam 5% applied immediately after each scalp rolling session. The patient experienced 95% scalp hair regrowth after 13 months of treatment (Figure 2B). No pain, bleeding, or other side effects were reported.

A 16-year-old boy with AA presented with 30% hair loss of 4 years’ duration (Figure 3A)(patient 3). Prior treatments included squaric acid and intralesional Kenalog injections. After 2 years of conventional topical therapy, twice-weekly scalp rolling with a 0.25-mm scalp roller of their choosing was added to the regimen, with clobetasol solution 0.05% and minoxidil foam 5% applied immediately after each scalp rolling session. The patient experienced 95% scalp hair regrowth at 17 months (Figure 3B). No pain, bleeding, or other side effects were reported.

Scalp rolling—also known as microneedling—provides a multifactorial approach to hair regrowth in patients with AA. The mechanism of action involves both the hair cycle and wound repair pathways by stimulation of the dermal papillae and stem cells.6 Scalp rolling has been observed to induce the expression of several hair growth pathway mediators, such as WNT3A, β-catenin, vascular endothelial growth factor, and WNT10B.7 Wnt/β-catenin pathway signaling is integral to multiple aspects of the hair regrowth process, including hair morphogenesis, follicle regeneration, and growth of the shaft itself.8,9 Scalp rolling causes microinjuries to the skin, thereby diverting blood supply to the follicles and stimulating wound regeneration, a process suggested to induce follicle regeneration. This effect is due to increased expression of vascular endothelial growth factor after cutaneous injury, a mediator of both hair growth and cycling as well as wound repair.7 Adjuvant scalp rolling creates a synergistic effect by facilitating absorption of topical and intralesional therapies. The physical breakdown of dermal capillary barriers creates microchannels that traverse the stratum corneum, improving the permeability of small-molecule substances and allowing for relatively painless and uniform delivery of combination therapies. A secondary benefit is hypertrophy, which counteracts the atrophy caused by topical steroids via collagen induction.7
Additionally, scalp rolling confers minimal risk to the patient, making it safer than conventional pharmacologic therapies such as corticosteroids or Janus kinase (JAK) inhibitors. Although intralesional steroid injections are first-line treatments for limited disease, they can cause pain and skin atrophy.10 In one cohort of 54 patients, topical steroids were inferior to both oral and intralesional treatment, and oral steroids carried a systemic side-effect profile and worsening of comorbidities including hyperglycemia and hypertension as well as negative effects on bone density.11 Baricitinib, a JAK inhibitor, was the first systemic treatment to gain US Food and Drug Administration approval for severe AA.12 However, this novel therapeutic confers adverse effects including infection, acne, and hypercholesterolemia, as reported in the BRAVE-AA trials.13 More broadly, the US Food and Drug Administration warns of serious long-term risks such as cardiovascular events and malignancy.14 Given the tremendous potential of JAK inhibitors, further research is warranted to understand both the efficacy of topical formulations as well as the possible role of scalp rolling as its adjuvant.
Finally, scalp rolling is easily accessible and affordable to patients. Scalp rolling devices are readily available and affordable online, and they can be used autonomously at home. This pragmatic option allows patients to take control of their own treatment course and offers a financially feasible alternative to navigating insurance coverage as well as the need for extra office visits for medication refills and monitoring.
We report 3 cases of the use of scalp rolling as an adjuvant to conventional therapy for refractory AA in young patients. Although prospective research is required to establish causality and characterize age-related trends in treatment response, consideration of scalp rolling as an adjuvant to conventional therapy may help to optimize treatment regimens. Given its low risk for side effects and potential benefits, we recommend scalp rolling for patients with refractory AA.
1. Senna M, Ko J, Tosti A, et al. Alopecia areata treatment patterns, healthcare resource utilization, and comorbidities in the US population using insurance claims. Adv Ther. 2021;38:4646-4658.
2. Huang CH, Fu Y, Chi CC. Health-related quality of life, depression, and self-esteem in patients with androgenetic alopecia: a systematic review and meta-analysis. JAMA Dermatol. 2021;157:963-970.
3. Deepak SH, Shwetha S. Scalp roller therapy in resistant alopecia areata. J Cutan Aesthet Surg. 2014;7:61-62.
4. Darwin E, Hirt PA, Fertig R, et al. Alopecia areata: review of epidemiology, clinical features, pathogenesis, and new treatment options.Int J Trichology. 2018;10:51-60.
5. Ito T, Yoshimasu T, Furukawa F, et al. Three-microneedle device as an effective option for intralesional corticosteroid administration for the treatment of alopecia areata. J Dermatol. 2017;44:304-305.
6. Dhurat R, Sukesh M, Avhad G, et al. A randomized evaluator blinded study of effect of microneedling in androgenetic alopecia: a pilot study. Int J Trichology. 2013;5:6-11.
7. Kim YS, Jeong KH, Kim JE, et al. Repeated microneedle stimulation induces enhanced hair growth in a murine model. Ann Dermatol. 2016;28:586-592.
8. Leirós GJ, Attorresi AI, Balañá ME. Hair follicle stem cell differentiation is inhibited through cross-talk between Wnt/β-catenin and androgen signalling in dermal papilla cells from patients with androgenetic alopecia. Br J Dermatol. 2012;166:1035-1042.
9. Myung PS, Takeo M, Ito M, et al. Epithelial Wnt ligand secretion is required for adult hair follicle growth and regeneration. J Invest Dermatol. 2013;133:31-41.
10. Strazzulla LC, Wang EHC, Avila L, et al. Alopecia areata: disease characteristics, clinical evaluation, and new perspectives on pathogenesis. J Am Acad Dermatol. 2018;78:1-12.
11. Charuwichitratana S, Wattanakrai P, Tanrattanakorn S. Randomized double-blind placebo-controlled trial in the treatment of alopecia areata with 0.25% desoximetasone cream. Arch Dermatol. 2000;136:1276
12.
13. King B, Ohyama M, Kwon O, et al. Two phase 3 trials of baricitinib for alopecia areata. N Engl J Med. 2022;386:1687-1699.
14. US Food and Drug Administration. FDA requires warnings about increased risk of serious heart-related events, cancer, blood clots, and death for JAK inhibitors that treat certain chronic inflammatory conditions. September 1, 2021.
To the Editor:
Alopecia areata (AA) is an autoimmune nonscarring hair loss disorder that can present at any age. Patients with AA have a disproportionately high comorbidity burden and low quality of life, often grappling with anxiety, depression, and psychosocial sequelae involving identity, such as reduced self-esteem.1,2 Although conventional therapies aim to reduce hair loss, none are curative.3 Response to treatment is highly unpredictable, with current data suggesting that up to 50% of patients recover within 1 year while 14% to 25% progress to either alopecia totalis (total scalp hair loss) or alopecia universalis (total body hair loss).4 Options for therapeutic intervention remain limited and vary in safety and effectiveness, warranting further research to identify optimal modalities and minimize side effects. Interestingly, scalp rolling has been used as an adjuvant to topical triamcinolone acetonide.3,5 However, the extent of its effect in combination with other therapies remains unclear. We report 3 pediatric patients with confirmed AA refractory to conventional topical treatment who experienced remarkable scalp hair regrowth after adding biweekly scalp rolling as an adjuvant therapy.
A 7-year-old boy with AA presented with 95% scalp hair loss of 7 months’ duration (Figure 1A)(patient 1). Prior treatments included mometasone solution and clobetasol solution 0.05%. After 3 months of conventional topical therapy, twice-weekly scalp rolling with a 0.25-mm scalp roller of their choosing was added to the regimen, with clobetasol solution 0.05% and minoxidil foam 5% applied immediately after each scalp rolling session. The patient experienced 95% scalp hair regrowth after 13 months of treatment (Figure 1B). No pain, bleeding, or other side effects were reported.

An 11-year-old girl with AA presented with 100% hair loss of 7 months’ duration (Figure 2A)(patient 2). Prior treatments included fluocinonide solution and intralesional Kenalog injections. After 4 months of conventional topical therapy, twice-weekly scalp rolling with a 0.25-mm scalp roller of their choosing was added to the regimen, with clobetasol solution 0.05% and minoxidil foam 5% applied immediately after each scalp rolling session. The patient experienced 95% scalp hair regrowth after 13 months of treatment (Figure 2B). No pain, bleeding, or other side effects were reported.

A 16-year-old boy with AA presented with 30% hair loss of 4 years’ duration (Figure 3A)(patient 3). Prior treatments included squaric acid and intralesional Kenalog injections. After 2 years of conventional topical therapy, twice-weekly scalp rolling with a 0.25-mm scalp roller of their choosing was added to the regimen, with clobetasol solution 0.05% and minoxidil foam 5% applied immediately after each scalp rolling session. The patient experienced 95% scalp hair regrowth at 17 months (Figure 3B). No pain, bleeding, or other side effects were reported.

Scalp rolling—also known as microneedling—provides a multifactorial approach to hair regrowth in patients with AA. The mechanism of action involves both the hair cycle and wound repair pathways by stimulation of the dermal papillae and stem cells.6 Scalp rolling has been observed to induce the expression of several hair growth pathway mediators, such as WNT3A, β-catenin, vascular endothelial growth factor, and WNT10B.7 Wnt/β-catenin pathway signaling is integral to multiple aspects of the hair regrowth process, including hair morphogenesis, follicle regeneration, and growth of the shaft itself.8,9 Scalp rolling causes microinjuries to the skin, thereby diverting blood supply to the follicles and stimulating wound regeneration, a process suggested to induce follicle regeneration. This effect is due to increased expression of vascular endothelial growth factor after cutaneous injury, a mediator of both hair growth and cycling as well as wound repair.7 Adjuvant scalp rolling creates a synergistic effect by facilitating absorption of topical and intralesional therapies. The physical breakdown of dermal capillary barriers creates microchannels that traverse the stratum corneum, improving the permeability of small-molecule substances and allowing for relatively painless and uniform delivery of combination therapies. A secondary benefit is hypertrophy, which counteracts the atrophy caused by topical steroids via collagen induction.7
Additionally, scalp rolling confers minimal risk to the patient, making it safer than conventional pharmacologic therapies such as corticosteroids or Janus kinase (JAK) inhibitors. Although intralesional steroid injections are first-line treatments for limited disease, they can cause pain and skin atrophy.10 In one cohort of 54 patients, topical steroids were inferior to both oral and intralesional treatment, and oral steroids carried a systemic side-effect profile and worsening of comorbidities including hyperglycemia and hypertension as well as negative effects on bone density.11 Baricitinib, a JAK inhibitor, was the first systemic treatment to gain US Food and Drug Administration approval for severe AA.12 However, this novel therapeutic confers adverse effects including infection, acne, and hypercholesterolemia, as reported in the BRAVE-AA trials.13 More broadly, the US Food and Drug Administration warns of serious long-term risks such as cardiovascular events and malignancy.14 Given the tremendous potential of JAK inhibitors, further research is warranted to understand both the efficacy of topical formulations as well as the possible role of scalp rolling as its adjuvant.
Finally, scalp rolling is easily accessible and affordable to patients. Scalp rolling devices are readily available and affordable online, and they can be used autonomously at home. This pragmatic option allows patients to take control of their own treatment course and offers a financially feasible alternative to navigating insurance coverage as well as the need for extra office visits for medication refills and monitoring.
We report 3 cases of the use of scalp rolling as an adjuvant to conventional therapy for refractory AA in young patients. Although prospective research is required to establish causality and characterize age-related trends in treatment response, consideration of scalp rolling as an adjuvant to conventional therapy may help to optimize treatment regimens. Given its low risk for side effects and potential benefits, we recommend scalp rolling for patients with refractory AA.
To the Editor:
Alopecia areata (AA) is an autoimmune nonscarring hair loss disorder that can present at any age. Patients with AA have a disproportionately high comorbidity burden and low quality of life, often grappling with anxiety, depression, and psychosocial sequelae involving identity, such as reduced self-esteem.1,2 Although conventional therapies aim to reduce hair loss, none are curative.3 Response to treatment is highly unpredictable, with current data suggesting that up to 50% of patients recover within 1 year while 14% to 25% progress to either alopecia totalis (total scalp hair loss) or alopecia universalis (total body hair loss).4 Options for therapeutic intervention remain limited and vary in safety and effectiveness, warranting further research to identify optimal modalities and minimize side effects. Interestingly, scalp rolling has been used as an adjuvant to topical triamcinolone acetonide.3,5 However, the extent of its effect in combination with other therapies remains unclear. We report 3 pediatric patients with confirmed AA refractory to conventional topical treatment who experienced remarkable scalp hair regrowth after adding biweekly scalp rolling as an adjuvant therapy.
A 7-year-old boy with AA presented with 95% scalp hair loss of 7 months’ duration (Figure 1A)(patient 1). Prior treatments included mometasone solution and clobetasol solution 0.05%. After 3 months of conventional topical therapy, twice-weekly scalp rolling with a 0.25-mm scalp roller of their choosing was added to the regimen, with clobetasol solution 0.05% and minoxidil foam 5% applied immediately after each scalp rolling session. The patient experienced 95% scalp hair regrowth after 13 months of treatment (Figure 1B). No pain, bleeding, or other side effects were reported.

An 11-year-old girl with AA presented with 100% hair loss of 7 months’ duration (Figure 2A)(patient 2). Prior treatments included fluocinonide solution and intralesional Kenalog injections. After 4 months of conventional topical therapy, twice-weekly scalp rolling with a 0.25-mm scalp roller of their choosing was added to the regimen, with clobetasol solution 0.05% and minoxidil foam 5% applied immediately after each scalp rolling session. The patient experienced 95% scalp hair regrowth after 13 months of treatment (Figure 2B). No pain, bleeding, or other side effects were reported.

A 16-year-old boy with AA presented with 30% hair loss of 4 years’ duration (Figure 3A)(patient 3). Prior treatments included squaric acid and intralesional Kenalog injections. After 2 years of conventional topical therapy, twice-weekly scalp rolling with a 0.25-mm scalp roller of their choosing was added to the regimen, with clobetasol solution 0.05% and minoxidil foam 5% applied immediately after each scalp rolling session. The patient experienced 95% scalp hair regrowth at 17 months (Figure 3B). No pain, bleeding, or other side effects were reported.

Scalp rolling—also known as microneedling—provides a multifactorial approach to hair regrowth in patients with AA. The mechanism of action involves both the hair cycle and wound repair pathways by stimulation of the dermal papillae and stem cells.6 Scalp rolling has been observed to induce the expression of several hair growth pathway mediators, such as WNT3A, β-catenin, vascular endothelial growth factor, and WNT10B.7 Wnt/β-catenin pathway signaling is integral to multiple aspects of the hair regrowth process, including hair morphogenesis, follicle regeneration, and growth of the shaft itself.8,9 Scalp rolling causes microinjuries to the skin, thereby diverting blood supply to the follicles and stimulating wound regeneration, a process suggested to induce follicle regeneration. This effect is due to increased expression of vascular endothelial growth factor after cutaneous injury, a mediator of both hair growth and cycling as well as wound repair.7 Adjuvant scalp rolling creates a synergistic effect by facilitating absorption of topical and intralesional therapies. The physical breakdown of dermal capillary barriers creates microchannels that traverse the stratum corneum, improving the permeability of small-molecule substances and allowing for relatively painless and uniform delivery of combination therapies. A secondary benefit is hypertrophy, which counteracts the atrophy caused by topical steroids via collagen induction.7
Additionally, scalp rolling confers minimal risk to the patient, making it safer than conventional pharmacologic therapies such as corticosteroids or Janus kinase (JAK) inhibitors. Although intralesional steroid injections are first-line treatments for limited disease, they can cause pain and skin atrophy.10 In one cohort of 54 patients, topical steroids were inferior to both oral and intralesional treatment, and oral steroids carried a systemic side-effect profile and worsening of comorbidities including hyperglycemia and hypertension as well as negative effects on bone density.11 Baricitinib, a JAK inhibitor, was the first systemic treatment to gain US Food and Drug Administration approval for severe AA.12 However, this novel therapeutic confers adverse effects including infection, acne, and hypercholesterolemia, as reported in the BRAVE-AA trials.13 More broadly, the US Food and Drug Administration warns of serious long-term risks such as cardiovascular events and malignancy.14 Given the tremendous potential of JAK inhibitors, further research is warranted to understand both the efficacy of topical formulations as well as the possible role of scalp rolling as its adjuvant.
Finally, scalp rolling is easily accessible and affordable to patients. Scalp rolling devices are readily available and affordable online, and they can be used autonomously at home. This pragmatic option allows patients to take control of their own treatment course and offers a financially feasible alternative to navigating insurance coverage as well as the need for extra office visits for medication refills and monitoring.
We report 3 cases of the use of scalp rolling as an adjuvant to conventional therapy for refractory AA in young patients. Although prospective research is required to establish causality and characterize age-related trends in treatment response, consideration of scalp rolling as an adjuvant to conventional therapy may help to optimize treatment regimens. Given its low risk for side effects and potential benefits, we recommend scalp rolling for patients with refractory AA.
1. Senna M, Ko J, Tosti A, et al. Alopecia areata treatment patterns, healthcare resource utilization, and comorbidities in the US population using insurance claims. Adv Ther. 2021;38:4646-4658.
2. Huang CH, Fu Y, Chi CC. Health-related quality of life, depression, and self-esteem in patients with androgenetic alopecia: a systematic review and meta-analysis. JAMA Dermatol. 2021;157:963-970.
3. Deepak SH, Shwetha S. Scalp roller therapy in resistant alopecia areata. J Cutan Aesthet Surg. 2014;7:61-62.
4. Darwin E, Hirt PA, Fertig R, et al. Alopecia areata: review of epidemiology, clinical features, pathogenesis, and new treatment options.Int J Trichology. 2018;10:51-60.
5. Ito T, Yoshimasu T, Furukawa F, et al. Three-microneedle device as an effective option for intralesional corticosteroid administration for the treatment of alopecia areata. J Dermatol. 2017;44:304-305.
6. Dhurat R, Sukesh M, Avhad G, et al. A randomized evaluator blinded study of effect of microneedling in androgenetic alopecia: a pilot study. Int J Trichology. 2013;5:6-11.
7. Kim YS, Jeong KH, Kim JE, et al. Repeated microneedle stimulation induces enhanced hair growth in a murine model. Ann Dermatol. 2016;28:586-592.
8. Leirós GJ, Attorresi AI, Balañá ME. Hair follicle stem cell differentiation is inhibited through cross-talk between Wnt/β-catenin and androgen signalling in dermal papilla cells from patients with androgenetic alopecia. Br J Dermatol. 2012;166:1035-1042.
9. Myung PS, Takeo M, Ito M, et al. Epithelial Wnt ligand secretion is required for adult hair follicle growth and regeneration. J Invest Dermatol. 2013;133:31-41.
10. Strazzulla LC, Wang EHC, Avila L, et al. Alopecia areata: disease characteristics, clinical evaluation, and new perspectives on pathogenesis. J Am Acad Dermatol. 2018;78:1-12.
11. Charuwichitratana S, Wattanakrai P, Tanrattanakorn S. Randomized double-blind placebo-controlled trial in the treatment of alopecia areata with 0.25% desoximetasone cream. Arch Dermatol. 2000;136:1276
12.
13. King B, Ohyama M, Kwon O, et al. Two phase 3 trials of baricitinib for alopecia areata. N Engl J Med. 2022;386:1687-1699.
14. US Food and Drug Administration. FDA requires warnings about increased risk of serious heart-related events, cancer, blood clots, and death for JAK inhibitors that treat certain chronic inflammatory conditions. September 1, 2021.
1. Senna M, Ko J, Tosti A, et al. Alopecia areata treatment patterns, healthcare resource utilization, and comorbidities in the US population using insurance claims. Adv Ther. 2021;38:4646-4658.
2. Huang CH, Fu Y, Chi CC. Health-related quality of life, depression, and self-esteem in patients with androgenetic alopecia: a systematic review and meta-analysis. JAMA Dermatol. 2021;157:963-970.
3. Deepak SH, Shwetha S. Scalp roller therapy in resistant alopecia areata. J Cutan Aesthet Surg. 2014;7:61-62.
4. Darwin E, Hirt PA, Fertig R, et al. Alopecia areata: review of epidemiology, clinical features, pathogenesis, and new treatment options.Int J Trichology. 2018;10:51-60.
5. Ito T, Yoshimasu T, Furukawa F, et al. Three-microneedle device as an effective option for intralesional corticosteroid administration for the treatment of alopecia areata. J Dermatol. 2017;44:304-305.
6. Dhurat R, Sukesh M, Avhad G, et al. A randomized evaluator blinded study of effect of microneedling in androgenetic alopecia: a pilot study. Int J Trichology. 2013;5:6-11.
7. Kim YS, Jeong KH, Kim JE, et al. Repeated microneedle stimulation induces enhanced hair growth in a murine model. Ann Dermatol. 2016;28:586-592.
8. Leirós GJ, Attorresi AI, Balañá ME. Hair follicle stem cell differentiation is inhibited through cross-talk between Wnt/β-catenin and androgen signalling in dermal papilla cells from patients with androgenetic alopecia. Br J Dermatol. 2012;166:1035-1042.
9. Myung PS, Takeo M, Ito M, et al. Epithelial Wnt ligand secretion is required for adult hair follicle growth and regeneration. J Invest Dermatol. 2013;133:31-41.
10. Strazzulla LC, Wang EHC, Avila L, et al. Alopecia areata: disease characteristics, clinical evaluation, and new perspectives on pathogenesis. J Am Acad Dermatol. 2018;78:1-12.
11. Charuwichitratana S, Wattanakrai P, Tanrattanakorn S. Randomized double-blind placebo-controlled trial in the treatment of alopecia areata with 0.25% desoximetasone cream. Arch Dermatol. 2000;136:1276
12.
13. King B, Ohyama M, Kwon O, et al. Two phase 3 trials of baricitinib for alopecia areata. N Engl J Med. 2022;386:1687-1699.
14. US Food and Drug Administration. FDA requires warnings about increased risk of serious heart-related events, cancer, blood clots, and death for JAK inhibitors that treat certain chronic inflammatory conditions. September 1, 2021.
Practice Points
- Alopecia areata (AA) is an autoimmune hair loss disorder with few effective treatments and no cure.
- Scalp rolling is a promising new treatment option that may stimulate hair regrowth by both direct collagen induction and indirect synergy with the use of topical medications.
- Dermatologists should be aware of scalp rolling as a safe, affordable, and potentially effective adjuvant to conventional therapy for AA.
Complex Regional Pain Syndrome Type II After a Brachial Plexus and C6 Nerve Root Injury
To the Editor:
A 62-year-old man presented with an atrophied painful left arm of 17 years’ duration that began when he was hit by a car as a pedestrian. He sustained severe multisystem injuries from the accident, including left brachial plexus and C6 nerve root avulsion injury. When he regained consciousness after 6 weeks in the intensive care unit, he immediately noted diffuse pain throughout the body, especially in the left arm. Since the accident, the patient continued to have diminished sensation to touch and temperature in the left arm. He also had burning, throbbing, and electrical pain in the left arm with light touch as well as spontaneously. He was thoroughly evaluated by a neurologist and was diagnosed with complex regional pain syndrome (CRPS) type II. For the treatment of pain, dorsal column stimulation and hemilaminectomy with exploration of the avulsed nerve root were attempted, both of which had minimal effect. He was maintained on hydromorphone, methadone, and oxazepam. He reported that for many years he was unable move out of bed due to the unbearable pain. With pain medications, he was able to regain most of his independence in his daily life, though the pain and other clinical aspects of CRPS still completely limited his use of the left arm.
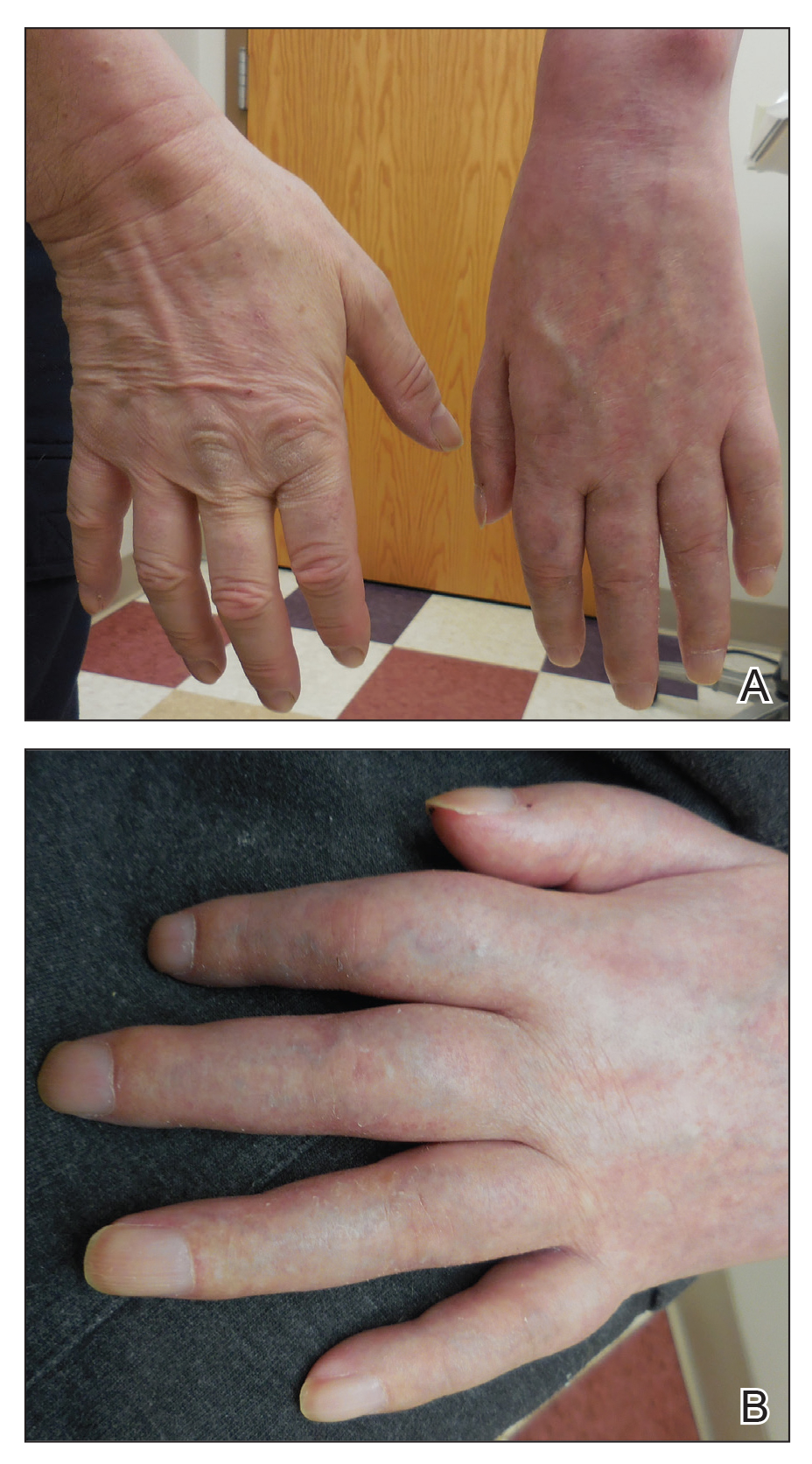
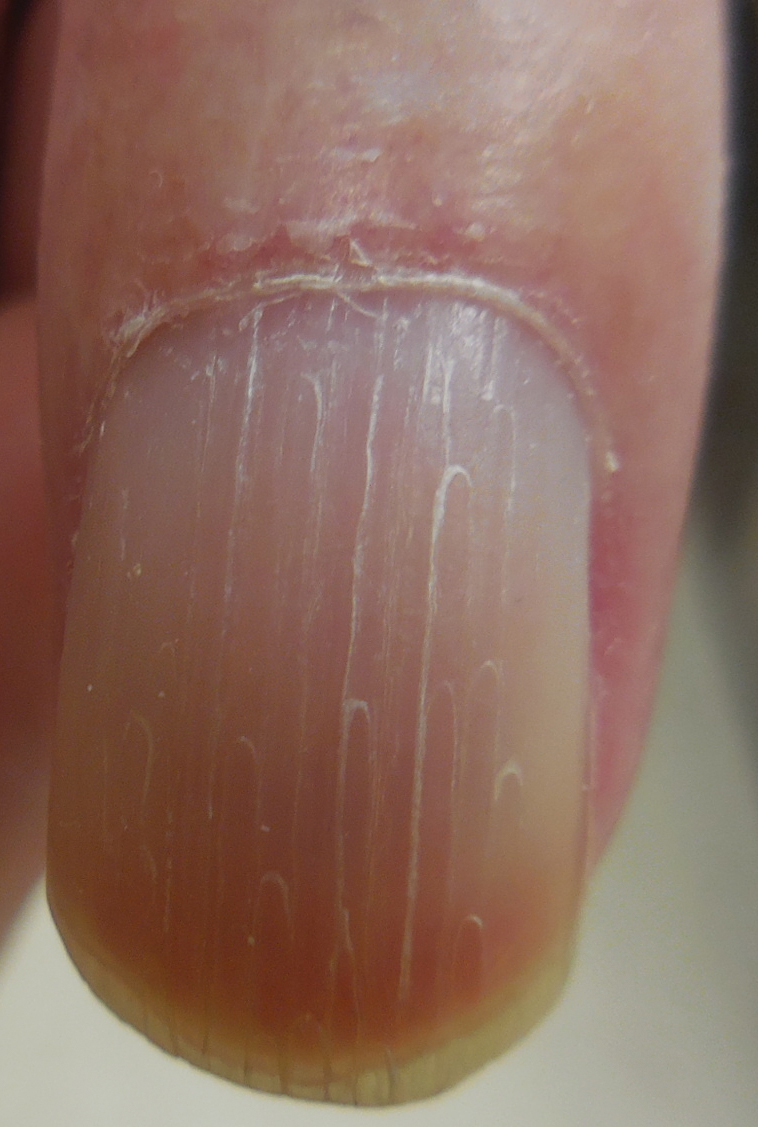
Physical examination revealed glossy, cold, hairless skin with hypohidrosis of the left arm, forearm, and hand (Figures 1 and 2A). The left arm was conspicuously atrophied, with the forearm and hand erythematous. The fingers were taut, contracted, and edematous (Figure 2B), and the skin was unable to be pinched. The fingernails on the left hand had dystrophic changes including yellow color and brittleness with longitudinal ridges (Figure 3). The patient could activate the left bicep and tricep muscles against gravity but had minimal function of the deltoid muscle. He also had minimal movement of the left index finger and was unable to move any other digits of the left hand. The patient was continued on pain management treatments and physical therapy for his condition.

Complex regional pain syndrome is a neuropathic disorder of the extremities characterized by pain and a variety of autonomic and motor disturbances such as local edema, limited active range of motion, and vasomotor and trophic skin changes. There are 2 types of CRPS: type II is marked by explicit nerve injury and type I is not. The pathophysiology of CRPS is unknown.1-3
There is no definite set of diagnostic criteria for CRPS. The lack of any gold-standard diagnostic test for CRPS has made arriving at one valid, widely accepted set of diagnostic criteria impossible.1 There are 4 widely used sets of diagnostic criteria. One is the International Association for the Study of Pain diagnostic criteria defined in 1994.4 However, the criteria rely entirely on subjective symptoms and have been under great scrutiny due to their questionable validity.2 Veldman et al5 presented other widely used CRPS diagnostic criteria in their prospective study of 829 reflex sympathetic dystrophy patients, which paid particular attention to the early clinical manifestations of CRPS. In 1999, Bruehl et al2 proposed their own modified diagnostic criteria, which required physician-assessed signs in 2 of 4 categories to avoid the practice of exclusively relying on subjective symptoms. In addition, during a consensus meeting in Budapest, Hungary, a modified version of the Bruehl criteria was proposed.6 All 4 criteria rely solely on detailed history and physical examination, and the choice of diagnostic criteria remains subjective.
The pathophysiology of CRPS also remains unclear. There are several proposed mechanisms such as sympathetic nervous system dysfunction, abnormal inflammatory response, and central nervous system involvement.1 Psychologic factors, sequelae of nerve injury, and genetic predisposition also have been implicated in the pathophysiology of CRPS.1 It is likely that several mechanisms variably contribute to each presentation of CRPS.
Many dermatologic findings, in addition to neuromuscular symptoms, accompany CRPS and serve as important clues to making the clinical diagnosis. Complex regional pain syndrome has been thought to have 3 distinct sequential stages of CRPS.1,3,7 Stage 1—the acute stage—is marked by hyperalgesia, allodynia, sudomotor disturbances, and prominent edema. Stage 2—the dystrophic stage—is characterized by more marked pain and sensory dysfunction, vasomotor dysfunction, development of motor dysfunction, soft tissue edema, skin and articular soft tissue thickening, and development of dystrophic nail changes. Stage 3—the atrophic stage—is marked by decreased pain and sensory disturbances, markedly increased motor dysfunction, waxy atrophic skin changes, progression of dystrophic nail changes, and skeletal cystic and subchondral erosions with diffuse osteoporosis.1,3,7
The staging model, however, has been called into question.3 In a cluster analysis, Bruehl et al3 arrived at 3 relatively consistent CRPS patient subgroups that did not have notably different pain duration, suggesting the existence of 3 CRPS subtypes, not stages. Their study found that one of the subgroups best represented the clinical presentation of CRPS type II. This subgroup had the greatest pain and sensory abnormalities and the least vasomotor dysfunction of all 3 subgroups. Nonetheless, this study has not settled the discussion, as it only included 113 patients.3 Thus, with future studies, our understanding of CRPS in stages may change, which likely will impact how the clinical diagnosis is made.
There is a lack of high-quality evidence for most treatment interventions for CRPS8; however, the current practice is to use an interdisciplinary approach.1,9,10 The main therapeutic arm of this approach is rehabilitation; physical and occupational therapy can help improve range of motion, contracture, and atrophy. The other 2 arms of the approach are psychologic therapy to improve quality of life and pain management with pharmacologic therapy and/or invasive interventions. The choice of therapy remains empirical; trial and error should be expected in developing an adequate treatment plan for each individual patient.
Many aspects of CRPS remain unclear, and even our current understanding of the disease will inevitably change over time. The syndrome can cause life-changing morbidities in patients, and late diagnosis and treatment are associated with poor prognosis. Because there are many dermatologic findings associated with the disorder, it is crucial for dermatologists to clinically recognize the disorder and to refer patients to appropriate channels so that treatment can be started as soon as possible.
- Borchers A, Gershwin M. Complex regional pain syndrome: a comprehensive and critical review. Autoimmun Rev. 2014;13:242-265.
- Bruehl S, Harden RN, Galer BS, et al. External validation of IASP diagnostic criteria for complex regional pain syndrome and proposed research diagnostic criteria. International Association for the Study of Pain. Pain. 1999;81:147-154.
- Bruehl S, Harden RN, Gaker BS, et al. Complex regional pain syndrome: are there distinct subtypes and sequential stages of the syndrome? Pain. 2002;95:119-124.
- Merskey H, Bogduk N. Classification of Chronic Pain: Descriptions of Chronic Pain Syndromes and Definitions of Pain Terms. 2nd ed. Seattle, WA: IASP Press; 1994.
- Veldman PH, Reynen HM, Arntz IE, et al. Signs and symptoms of reflex sympathetic dystrophy: prospective study of 829 patients. Lancet. 1993;342:1012-1016.
- Harden RN, Bruehl S, Perez RS, et al. Validation of proposed diagnostic criteria (the “Budapest Criteria”) for complex regional pain syndrome. Pain. 2010;150:268-274.
- Sebastin SJ. Complex regional pain syndrome. Indian J Plast Surg. 2011;44:298-307.
- O’Connell NE, Wand BM, McAuley J, et al. Interventions for treating pain and disability in adults with complex regional pain syndrome. Cochrane Database Syst Rev. 2013;4:CD009416.
- Hsu ES. Practical management of complex regional pain syndrome. Am J Ther. 2009;16:147-154.
- Stanton-Hicks MD, Burton AW, Bruehl SP, et al. An updated interdisciplinary clinical pathway for CRPS: report of an expert panel. Pain Pract. 2002;2:1-16.
To the Editor:
A 62-year-old man presented with an atrophied painful left arm of 17 years’ duration that began when he was hit by a car as a pedestrian. He sustained severe multisystem injuries from the accident, including left brachial plexus and C6 nerve root avulsion injury. When he regained consciousness after 6 weeks in the intensive care unit, he immediately noted diffuse pain throughout the body, especially in the left arm. Since the accident, the patient continued to have diminished sensation to touch and temperature in the left arm. He also had burning, throbbing, and electrical pain in the left arm with light touch as well as spontaneously. He was thoroughly evaluated by a neurologist and was diagnosed with complex regional pain syndrome (CRPS) type II. For the treatment of pain, dorsal column stimulation and hemilaminectomy with exploration of the avulsed nerve root were attempted, both of which had minimal effect. He was maintained on hydromorphone, methadone, and oxazepam. He reported that for many years he was unable move out of bed due to the unbearable pain. With pain medications, he was able to regain most of his independence in his daily life, though the pain and other clinical aspects of CRPS still completely limited his use of the left arm.
Physical examination revealed glossy, cold, hairless skin with hypohidrosis of the left arm, forearm, and hand (Figures 1 and 2A). The left arm was conspicuously atrophied, with the forearm and hand erythematous. The fingers were taut, contracted, and edematous (Figure 2B), and the skin was unable to be pinched. The fingernails on the left hand had dystrophic changes including yellow color and brittleness with longitudinal ridges (Figure 3). The patient could activate the left bicep and tricep muscles against gravity but had minimal function of the deltoid muscle. He also had minimal movement of the left index finger and was unable to move any other digits of the left hand. The patient was continued on pain management treatments and physical therapy for his condition.



Complex regional pain syndrome is a neuropathic disorder of the extremities characterized by pain and a variety of autonomic and motor disturbances such as local edema, limited active range of motion, and vasomotor and trophic skin changes. There are 2 types of CRPS: type II is marked by explicit nerve injury and type I is not. The pathophysiology of CRPS is unknown.1-3
There is no definite set of diagnostic criteria for CRPS. The lack of any gold-standard diagnostic test for CRPS has made arriving at one valid, widely accepted set of diagnostic criteria impossible.1 There are 4 widely used sets of diagnostic criteria. One is the International Association for the Study of Pain diagnostic criteria defined in 1994.4 However, the criteria rely entirely on subjective symptoms and have been under great scrutiny due to their questionable validity.2 Veldman et al5 presented other widely used CRPS diagnostic criteria in their prospective study of 829 reflex sympathetic dystrophy patients, which paid particular attention to the early clinical manifestations of CRPS. In 1999, Bruehl et al2 proposed their own modified diagnostic criteria, which required physician-assessed signs in 2 of 4 categories to avoid the practice of exclusively relying on subjective symptoms. In addition, during a consensus meeting in Budapest, Hungary, a modified version of the Bruehl criteria was proposed.6 All 4 criteria rely solely on detailed history and physical examination, and the choice of diagnostic criteria remains subjective.
The pathophysiology of CRPS also remains unclear. There are several proposed mechanisms such as sympathetic nervous system dysfunction, abnormal inflammatory response, and central nervous system involvement.1 Psychologic factors, sequelae of nerve injury, and genetic predisposition also have been implicated in the pathophysiology of CRPS.1 It is likely that several mechanisms variably contribute to each presentation of CRPS.
Many dermatologic findings, in addition to neuromuscular symptoms, accompany CRPS and serve as important clues to making the clinical diagnosis. Complex regional pain syndrome has been thought to have 3 distinct sequential stages of CRPS.1,3,7 Stage 1—the acute stage—is marked by hyperalgesia, allodynia, sudomotor disturbances, and prominent edema. Stage 2—the dystrophic stage—is characterized by more marked pain and sensory dysfunction, vasomotor dysfunction, development of motor dysfunction, soft tissue edema, skin and articular soft tissue thickening, and development of dystrophic nail changes. Stage 3—the atrophic stage—is marked by decreased pain and sensory disturbances, markedly increased motor dysfunction, waxy atrophic skin changes, progression of dystrophic nail changes, and skeletal cystic and subchondral erosions with diffuse osteoporosis.1,3,7
The staging model, however, has been called into question.3 In a cluster analysis, Bruehl et al3 arrived at 3 relatively consistent CRPS patient subgroups that did not have notably different pain duration, suggesting the existence of 3 CRPS subtypes, not stages. Their study found that one of the subgroups best represented the clinical presentation of CRPS type II. This subgroup had the greatest pain and sensory abnormalities and the least vasomotor dysfunction of all 3 subgroups. Nonetheless, this study has not settled the discussion, as it only included 113 patients.3 Thus, with future studies, our understanding of CRPS in stages may change, which likely will impact how the clinical diagnosis is made.
There is a lack of high-quality evidence for most treatment interventions for CRPS8; however, the current practice is to use an interdisciplinary approach.1,9,10 The main therapeutic arm of this approach is rehabilitation; physical and occupational therapy can help improve range of motion, contracture, and atrophy. The other 2 arms of the approach are psychologic therapy to improve quality of life and pain management with pharmacologic therapy and/or invasive interventions. The choice of therapy remains empirical; trial and error should be expected in developing an adequate treatment plan for each individual patient.
Many aspects of CRPS remain unclear, and even our current understanding of the disease will inevitably change over time. The syndrome can cause life-changing morbidities in patients, and late diagnosis and treatment are associated with poor prognosis. Because there are many dermatologic findings associated with the disorder, it is crucial for dermatologists to clinically recognize the disorder and to refer patients to appropriate channels so that treatment can be started as soon as possible.
To the Editor:
A 62-year-old man presented with an atrophied painful left arm of 17 years’ duration that began when he was hit by a car as a pedestrian. He sustained severe multisystem injuries from the accident, including left brachial plexus and C6 nerve root avulsion injury. When he regained consciousness after 6 weeks in the intensive care unit, he immediately noted diffuse pain throughout the body, especially in the left arm. Since the accident, the patient continued to have diminished sensation to touch and temperature in the left arm. He also had burning, throbbing, and electrical pain in the left arm with light touch as well as spontaneously. He was thoroughly evaluated by a neurologist and was diagnosed with complex regional pain syndrome (CRPS) type II. For the treatment of pain, dorsal column stimulation and hemilaminectomy with exploration of the avulsed nerve root were attempted, both of which had minimal effect. He was maintained on hydromorphone, methadone, and oxazepam. He reported that for many years he was unable move out of bed due to the unbearable pain. With pain medications, he was able to regain most of his independence in his daily life, though the pain and other clinical aspects of CRPS still completely limited his use of the left arm.
Physical examination revealed glossy, cold, hairless skin with hypohidrosis of the left arm, forearm, and hand (Figures 1 and 2A). The left arm was conspicuously atrophied, with the forearm and hand erythematous. The fingers were taut, contracted, and edematous (Figure 2B), and the skin was unable to be pinched. The fingernails on the left hand had dystrophic changes including yellow color and brittleness with longitudinal ridges (Figure 3). The patient could activate the left bicep and tricep muscles against gravity but had minimal function of the deltoid muscle. He also had minimal movement of the left index finger and was unable to move any other digits of the left hand. The patient was continued on pain management treatments and physical therapy for his condition.



Complex regional pain syndrome is a neuropathic disorder of the extremities characterized by pain and a variety of autonomic and motor disturbances such as local edema, limited active range of motion, and vasomotor and trophic skin changes. There are 2 types of CRPS: type II is marked by explicit nerve injury and type I is not. The pathophysiology of CRPS is unknown.1-3
There is no definite set of diagnostic criteria for CRPS. The lack of any gold-standard diagnostic test for CRPS has made arriving at one valid, widely accepted set of diagnostic criteria impossible.1 There are 4 widely used sets of diagnostic criteria. One is the International Association for the Study of Pain diagnostic criteria defined in 1994.4 However, the criteria rely entirely on subjective symptoms and have been under great scrutiny due to their questionable validity.2 Veldman et al5 presented other widely used CRPS diagnostic criteria in their prospective study of 829 reflex sympathetic dystrophy patients, which paid particular attention to the early clinical manifestations of CRPS. In 1999, Bruehl et al2 proposed their own modified diagnostic criteria, which required physician-assessed signs in 2 of 4 categories to avoid the practice of exclusively relying on subjective symptoms. In addition, during a consensus meeting in Budapest, Hungary, a modified version of the Bruehl criteria was proposed.6 All 4 criteria rely solely on detailed history and physical examination, and the choice of diagnostic criteria remains subjective.
The pathophysiology of CRPS also remains unclear. There are several proposed mechanisms such as sympathetic nervous system dysfunction, abnormal inflammatory response, and central nervous system involvement.1 Psychologic factors, sequelae of nerve injury, and genetic predisposition also have been implicated in the pathophysiology of CRPS.1 It is likely that several mechanisms variably contribute to each presentation of CRPS.
Many dermatologic findings, in addition to neuromuscular symptoms, accompany CRPS and serve as important clues to making the clinical diagnosis. Complex regional pain syndrome has been thought to have 3 distinct sequential stages of CRPS.1,3,7 Stage 1—the acute stage—is marked by hyperalgesia, allodynia, sudomotor disturbances, and prominent edema. Stage 2—the dystrophic stage—is characterized by more marked pain and sensory dysfunction, vasomotor dysfunction, development of motor dysfunction, soft tissue edema, skin and articular soft tissue thickening, and development of dystrophic nail changes. Stage 3—the atrophic stage—is marked by decreased pain and sensory disturbances, markedly increased motor dysfunction, waxy atrophic skin changes, progression of dystrophic nail changes, and skeletal cystic and subchondral erosions with diffuse osteoporosis.1,3,7
The staging model, however, has been called into question.3 In a cluster analysis, Bruehl et al3 arrived at 3 relatively consistent CRPS patient subgroups that did not have notably different pain duration, suggesting the existence of 3 CRPS subtypes, not stages. Their study found that one of the subgroups best represented the clinical presentation of CRPS type II. This subgroup had the greatest pain and sensory abnormalities and the least vasomotor dysfunction of all 3 subgroups. Nonetheless, this study has not settled the discussion, as it only included 113 patients.3 Thus, with future studies, our understanding of CRPS in stages may change, which likely will impact how the clinical diagnosis is made.
There is a lack of high-quality evidence for most treatment interventions for CRPS8; however, the current practice is to use an interdisciplinary approach.1,9,10 The main therapeutic arm of this approach is rehabilitation; physical and occupational therapy can help improve range of motion, contracture, and atrophy. The other 2 arms of the approach are psychologic therapy to improve quality of life and pain management with pharmacologic therapy and/or invasive interventions. The choice of therapy remains empirical; trial and error should be expected in developing an adequate treatment plan for each individual patient.
Many aspects of CRPS remain unclear, and even our current understanding of the disease will inevitably change over time. The syndrome can cause life-changing morbidities in patients, and late diagnosis and treatment are associated with poor prognosis. Because there are many dermatologic findings associated with the disorder, it is crucial for dermatologists to clinically recognize the disorder and to refer patients to appropriate channels so that treatment can be started as soon as possible.
- Borchers A, Gershwin M. Complex regional pain syndrome: a comprehensive and critical review. Autoimmun Rev. 2014;13:242-265.
- Bruehl S, Harden RN, Galer BS, et al. External validation of IASP diagnostic criteria for complex regional pain syndrome and proposed research diagnostic criteria. International Association for the Study of Pain. Pain. 1999;81:147-154.
- Bruehl S, Harden RN, Gaker BS, et al. Complex regional pain syndrome: are there distinct subtypes and sequential stages of the syndrome? Pain. 2002;95:119-124.
- Merskey H, Bogduk N. Classification of Chronic Pain: Descriptions of Chronic Pain Syndromes and Definitions of Pain Terms. 2nd ed. Seattle, WA: IASP Press; 1994.
- Veldman PH, Reynen HM, Arntz IE, et al. Signs and symptoms of reflex sympathetic dystrophy: prospective study of 829 patients. Lancet. 1993;342:1012-1016.
- Harden RN, Bruehl S, Perez RS, et al. Validation of proposed diagnostic criteria (the “Budapest Criteria”) for complex regional pain syndrome. Pain. 2010;150:268-274.
- Sebastin SJ. Complex regional pain syndrome. Indian J Plast Surg. 2011;44:298-307.
- O’Connell NE, Wand BM, McAuley J, et al. Interventions for treating pain and disability in adults with complex regional pain syndrome. Cochrane Database Syst Rev. 2013;4:CD009416.
- Hsu ES. Practical management of complex regional pain syndrome. Am J Ther. 2009;16:147-154.
- Stanton-Hicks MD, Burton AW, Bruehl SP, et al. An updated interdisciplinary clinical pathway for CRPS: report of an expert panel. Pain Pract. 2002;2:1-16.
- Borchers A, Gershwin M. Complex regional pain syndrome: a comprehensive and critical review. Autoimmun Rev. 2014;13:242-265.
- Bruehl S, Harden RN, Galer BS, et al. External validation of IASP diagnostic criteria for complex regional pain syndrome and proposed research diagnostic criteria. International Association for the Study of Pain. Pain. 1999;81:147-154.
- Bruehl S, Harden RN, Gaker BS, et al. Complex regional pain syndrome: are there distinct subtypes and sequential stages of the syndrome? Pain. 2002;95:119-124.
- Merskey H, Bogduk N. Classification of Chronic Pain: Descriptions of Chronic Pain Syndromes and Definitions of Pain Terms. 2nd ed. Seattle, WA: IASP Press; 1994.
- Veldman PH, Reynen HM, Arntz IE, et al. Signs and symptoms of reflex sympathetic dystrophy: prospective study of 829 patients. Lancet. 1993;342:1012-1016.
- Harden RN, Bruehl S, Perez RS, et al. Validation of proposed diagnostic criteria (the “Budapest Criteria”) for complex regional pain syndrome. Pain. 2010;150:268-274.
- Sebastin SJ. Complex regional pain syndrome. Indian J Plast Surg. 2011;44:298-307.
- O’Connell NE, Wand BM, McAuley J, et al. Interventions for treating pain and disability in adults with complex regional pain syndrome. Cochrane Database Syst Rev. 2013;4:CD009416.
- Hsu ES. Practical management of complex regional pain syndrome. Am J Ther. 2009;16:147-154.
- Stanton-Hicks MD, Burton AW, Bruehl SP, et al. An updated interdisciplinary clinical pathway for CRPS: report of an expert panel. Pain Pract. 2002;2:1-16.
Practice Points
- Complex regional pain syndrome (CRPS) is a neuropathic disorder of the extremities characterized by pain, a variety of autonomic and motor disturbances, and dermatologic findings.
- Early recognition of CRPS is critical, as it presents life-changing morbidities to patients.
- A multidisciplinary treatment approach with physical therapy, occupational therapy, psychological support, and pain control is needed for the management of CRPS.