User login
Adapalene/benzoyl peroxide gel halves acne lesion counts in 1 month
WAIKOLOA, HAWAII – The fixed-dose combination of adapalene/benzoyl peroxide gel 0.1%/2.5% typically results in reductions of 40%-50% in inflammatory and 30%-40% in noninflammatory acne lesion counts during the first 4 weeks of therapy.
In a pooled analysis of 14 studies totaling 2,358 acne patients aged 9-61 years, the proportion with an Investigator’s Global Assessment (IGA) of moderate or severe acne dropped from 92% at baseline to 51% at 4 weeks, according to Dr. Linda Stein Gold.
The pooled analysis was carried out to provide physicians and patients with information as to what to realistically expect in the first 4 weeks of therapy. The analysis is particularly timely in light of the Food and Drug Administration’s recent expansion of the indication for adapalene/benzoyl peroxide gel 0.1%/2.5% (Epiduo) to include acne patients as young as 9 years of age, noted Dr. Stein Gold, director of clinical research in the department of dermatology at Henry Ford Hospital, Detroit.
Mild skin irritation was common, especially in the first 2 weeks of use.
"It’s a fiction that retinoids are too harsh for younger patients to use," Dr. Stein Gold said. "I tell all my acne patients, especially the younger ones, no matter what topical retinoid they’re using, to use it every other night for the first 2 weeks, make sure their skin is completely dry, and use a gentle cleanser and a good moisturizer," she said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation. "If they get through those first 2 weeks, they’ll see that the tolerability really improves."
Dr. Stein Gold said she likes the fixed combination of adapalene/benzoyl peroxide gel 0.1%/2.5% because it combines several key elements of cutting-edge acne treatment. Topical retinoids are not mere comedone busters, as formerly thought, but are also effective agents for papules and pustular lesions. While they do not kill Propionibacterium acnes, they down-regulate Toll-like receptor 2, which is produced by the bacterium and induces proinflammatory cytokines. Topical retinoids are a key part of maintenance-of-remission strategies. And adapalene is unique among topical retinoids in that it is inherently stable with benzoyl peroxide (BPO) and is stable in daylight.
BPO is unique in that it has potent antibacterial activity, but never causes P. acnes resistance, even after many years of treatment. BPO is quite effective for inflammatory lesions and moderately effective for comedonal acne lesions, Dr. Stein Gold noted. Also, it is well established that BPO at a concentration of 2.5% is as efficacious as 5% or 10%, and much better tolerated than at the higher concentrations. The molecule size and the use of a vehicle with good penetration into the hair follicle are much more important factors in treatment effectiveness than is the BPO concentration, she added.
In one study of acne patients with antibiotic-resistant P. acnes, including clindamycin-, doxycycline-, erythromycin-, and minocycline-resistant microorganisms, 88% of the antibiotic-resistant P. acnes bacteria were killed after 2 weeks of treatment with adapalene/BPO gel 0.1%/2.5%. After 4 weeks of treatment, 97% of the antibiotic-resistant organisms were dead. That’s testimony to the P. acnes–killing power of BPO, said Dr. Stein Gold.
"There’s a sense among many that with all the newer medications we have for acne, benzoyl peroxide is really your grandfather’s treatment, with no place in today’s modern world. This is totally false. I really feel that benzoyl peroxide should play a central role in all of our acne patients’ treatment regimens, unless of course they’re allergic to it, which occurs in only a small percentage of our patients," she said.
The pooled analysis was funded by Galderma. Dr. Stein Gold is a consultant to Galderma, Stiefel, Medicis, and Warner Chilcott.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The fixed-dose combination of adapalene/benzoyl peroxide gel 0.1%/2.5% typically results in reductions of 40%-50% in inflammatory and 30%-40% in noninflammatory acne lesion counts during the first 4 weeks of therapy.
In a pooled analysis of 14 studies totaling 2,358 acne patients aged 9-61 years, the proportion with an Investigator’s Global Assessment (IGA) of moderate or severe acne dropped from 92% at baseline to 51% at 4 weeks, according to Dr. Linda Stein Gold.
The pooled analysis was carried out to provide physicians and patients with information as to what to realistically expect in the first 4 weeks of therapy. The analysis is particularly timely in light of the Food and Drug Administration’s recent expansion of the indication for adapalene/benzoyl peroxide gel 0.1%/2.5% (Epiduo) to include acne patients as young as 9 years of age, noted Dr. Stein Gold, director of clinical research in the department of dermatology at Henry Ford Hospital, Detroit.
Mild skin irritation was common, especially in the first 2 weeks of use.
"It’s a fiction that retinoids are too harsh for younger patients to use," Dr. Stein Gold said. "I tell all my acne patients, especially the younger ones, no matter what topical retinoid they’re using, to use it every other night for the first 2 weeks, make sure their skin is completely dry, and use a gentle cleanser and a good moisturizer," she said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation. "If they get through those first 2 weeks, they’ll see that the tolerability really improves."
Dr. Stein Gold said she likes the fixed combination of adapalene/benzoyl peroxide gel 0.1%/2.5% because it combines several key elements of cutting-edge acne treatment. Topical retinoids are not mere comedone busters, as formerly thought, but are also effective agents for papules and pustular lesions. While they do not kill Propionibacterium acnes, they down-regulate Toll-like receptor 2, which is produced by the bacterium and induces proinflammatory cytokines. Topical retinoids are a key part of maintenance-of-remission strategies. And adapalene is unique among topical retinoids in that it is inherently stable with benzoyl peroxide (BPO) and is stable in daylight.
BPO is unique in that it has potent antibacterial activity, but never causes P. acnes resistance, even after many years of treatment. BPO is quite effective for inflammatory lesions and moderately effective for comedonal acne lesions, Dr. Stein Gold noted. Also, it is well established that BPO at a concentration of 2.5% is as efficacious as 5% or 10%, and much better tolerated than at the higher concentrations. The molecule size and the use of a vehicle with good penetration into the hair follicle are much more important factors in treatment effectiveness than is the BPO concentration, she added.
In one study of acne patients with antibiotic-resistant P. acnes, including clindamycin-, doxycycline-, erythromycin-, and minocycline-resistant microorganisms, 88% of the antibiotic-resistant P. acnes bacteria were killed after 2 weeks of treatment with adapalene/BPO gel 0.1%/2.5%. After 4 weeks of treatment, 97% of the antibiotic-resistant organisms were dead. That’s testimony to the P. acnes–killing power of BPO, said Dr. Stein Gold.
"There’s a sense among many that with all the newer medications we have for acne, benzoyl peroxide is really your grandfather’s treatment, with no place in today’s modern world. This is totally false. I really feel that benzoyl peroxide should play a central role in all of our acne patients’ treatment regimens, unless of course they’re allergic to it, which occurs in only a small percentage of our patients," she said.
The pooled analysis was funded by Galderma. Dr. Stein Gold is a consultant to Galderma, Stiefel, Medicis, and Warner Chilcott.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The fixed-dose combination of adapalene/benzoyl peroxide gel 0.1%/2.5% typically results in reductions of 40%-50% in inflammatory and 30%-40% in noninflammatory acne lesion counts during the first 4 weeks of therapy.
In a pooled analysis of 14 studies totaling 2,358 acne patients aged 9-61 years, the proportion with an Investigator’s Global Assessment (IGA) of moderate or severe acne dropped from 92% at baseline to 51% at 4 weeks, according to Dr. Linda Stein Gold.
The pooled analysis was carried out to provide physicians and patients with information as to what to realistically expect in the first 4 weeks of therapy. The analysis is particularly timely in light of the Food and Drug Administration’s recent expansion of the indication for adapalene/benzoyl peroxide gel 0.1%/2.5% (Epiduo) to include acne patients as young as 9 years of age, noted Dr. Stein Gold, director of clinical research in the department of dermatology at Henry Ford Hospital, Detroit.
Mild skin irritation was common, especially in the first 2 weeks of use.
"It’s a fiction that retinoids are too harsh for younger patients to use," Dr. Stein Gold said. "I tell all my acne patients, especially the younger ones, no matter what topical retinoid they’re using, to use it every other night for the first 2 weeks, make sure their skin is completely dry, and use a gentle cleanser and a good moisturizer," she said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation. "If they get through those first 2 weeks, they’ll see that the tolerability really improves."
Dr. Stein Gold said she likes the fixed combination of adapalene/benzoyl peroxide gel 0.1%/2.5% because it combines several key elements of cutting-edge acne treatment. Topical retinoids are not mere comedone busters, as formerly thought, but are also effective agents for papules and pustular lesions. While they do not kill Propionibacterium acnes, they down-regulate Toll-like receptor 2, which is produced by the bacterium and induces proinflammatory cytokines. Topical retinoids are a key part of maintenance-of-remission strategies. And adapalene is unique among topical retinoids in that it is inherently stable with benzoyl peroxide (BPO) and is stable in daylight.
BPO is unique in that it has potent antibacterial activity, but never causes P. acnes resistance, even after many years of treatment. BPO is quite effective for inflammatory lesions and moderately effective for comedonal acne lesions, Dr. Stein Gold noted. Also, it is well established that BPO at a concentration of 2.5% is as efficacious as 5% or 10%, and much better tolerated than at the higher concentrations. The molecule size and the use of a vehicle with good penetration into the hair follicle are much more important factors in treatment effectiveness than is the BPO concentration, she added.
In one study of acne patients with antibiotic-resistant P. acnes, including clindamycin-, doxycycline-, erythromycin-, and minocycline-resistant microorganisms, 88% of the antibiotic-resistant P. acnes bacteria were killed after 2 weeks of treatment with adapalene/BPO gel 0.1%/2.5%. After 4 weeks of treatment, 97% of the antibiotic-resistant organisms were dead. That’s testimony to the P. acnes–killing power of BPO, said Dr. Stein Gold.
"There’s a sense among many that with all the newer medications we have for acne, benzoyl peroxide is really your grandfather’s treatment, with no place in today’s modern world. This is totally false. I really feel that benzoyl peroxide should play a central role in all of our acne patients’ treatment regimens, unless of course they’re allergic to it, which occurs in only a small percentage of our patients," she said.
The pooled analysis was funded by Galderma. Dr. Stein Gold is a consultant to Galderma, Stiefel, Medicis, and Warner Chilcott.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
When to Screen for Celiac Disease in Hypothyroidism
CHICAGO – Patients with hypothyroidism who needed higher doses of levothyroxine were more likely to have celiac disease, according to a study released at the annual Digestive Disease Week.
In a study of 400 patients with hypothyroidism, about 5% of those who needed at least 125 mcg/day of levothyroxine also had celiac disease, according to Dr. Richard Zubarik of the University of Vermont, Burlington. That prevalence rate tops even the rate among patients with iron deficiency anemia, for whom celiac disease screening is recommended.
In a video interview, Dr. Zubarik explains which patients with hypothyroidism should be screened for celiac disease, and he discusses what mechanisms might link the need for higher levels of levothyroxine with an increased risk of celiac disease.

CHICAGO – Patients with hypothyroidism who needed higher doses of levothyroxine were more likely to have celiac disease, according to a study released at the annual Digestive Disease Week.
In a study of 400 patients with hypothyroidism, about 5% of those who needed at least 125 mcg/day of levothyroxine also had celiac disease, according to Dr. Richard Zubarik of the University of Vermont, Burlington. That prevalence rate tops even the rate among patients with iron deficiency anemia, for whom celiac disease screening is recommended.
In a video interview, Dr. Zubarik explains which patients with hypothyroidism should be screened for celiac disease, and he discusses what mechanisms might link the need for higher levels of levothyroxine with an increased risk of celiac disease.

CHICAGO – Patients with hypothyroidism who needed higher doses of levothyroxine were more likely to have celiac disease, according to a study released at the annual Digestive Disease Week.
In a study of 400 patients with hypothyroidism, about 5% of those who needed at least 125 mcg/day of levothyroxine also had celiac disease, according to Dr. Richard Zubarik of the University of Vermont, Burlington. That prevalence rate tops even the rate among patients with iron deficiency anemia, for whom celiac disease screening is recommended.
In a video interview, Dr. Zubarik explains which patients with hypothyroidism should be screened for celiac disease, and he discusses what mechanisms might link the need for higher levels of levothyroxine with an increased risk of celiac disease.

At DDW 2014
VIDEO: When to screen for celiac disease in hypothyroidism
CHICAGO – Patients with hypothyroidism who needed higher doses of levothyroxine were more likely to have celiac disease, according to a study released at the annual Digestive Disease Week.
In a study of 400 patients with hypothyroidism, about 5% of those who needed at least 125 mcg/day of levothyroxine also had celiac disease, according to Dr. Richard Zubarik of the University of Vermont, Burlington.That prevalence rate tops even the rate among patients with iron deficiency anemia, for whom celiac disease screening is recommended.
In a video interview, Dr. Zubarik explains which patients with hypothyroidism should be screened for celiac disease, and he discusses what mechanisms might link the need for higher levels of levothyroxine with an increased risk of celiac disease.
CHICAGO – Patients with hypothyroidism who needed higher doses of levothyroxine were more likely to have celiac disease, according to a study released at the annual Digestive Disease Week.
In a study of 400 patients with hypothyroidism, about 5% of those who needed at least 125 mcg/day of levothyroxine also had celiac disease, according to Dr. Richard Zubarik of the University of Vermont, Burlington.That prevalence rate tops even the rate among patients with iron deficiency anemia, for whom celiac disease screening is recommended.
In a video interview, Dr. Zubarik explains which patients with hypothyroidism should be screened for celiac disease, and he discusses what mechanisms might link the need for higher levels of levothyroxine with an increased risk of celiac disease.
CHICAGO – Patients with hypothyroidism who needed higher doses of levothyroxine were more likely to have celiac disease, according to a study released at the annual Digestive Disease Week.
In a study of 400 patients with hypothyroidism, about 5% of those who needed at least 125 mcg/day of levothyroxine also had celiac disease, according to Dr. Richard Zubarik of the University of Vermont, Burlington.That prevalence rate tops even the rate among patients with iron deficiency anemia, for whom celiac disease screening is recommended.
In a video interview, Dr. Zubarik explains which patients with hypothyroidism should be screened for celiac disease, and he discusses what mechanisms might link the need for higher levels of levothyroxine with an increased risk of celiac disease.
At DDW 2014
VIDEO: Low FODMAP diet reduced kids’ IBS pain
CHICAGO – A diet that restricts wheat, beans, dairy, and fruit juice in children with irritable bowel syndrome reduced the frequency and severity of abdominal pain, according to a study presented at the annual Digestive Disease Week.
In a double-blind, randomized, crossover trial of 55 children, those who followed a low FODMAP (fermentable oligo-di-monosaccharides and polyols) diet decreased abdominal pain frequency 20%-30%, explained Dr. Bruno Chumpitazi of Texas Children’s Hospital, Houston.
In a video interview, Dr. Chumpitazi discusses the study's implications for the nearly 20% of school-age children and adolescents who may have IBS, and whether the low FODMAP diet could be a first-line therapy for those children.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – A diet that restricts wheat, beans, dairy, and fruit juice in children with irritable bowel syndrome reduced the frequency and severity of abdominal pain, according to a study presented at the annual Digestive Disease Week.
In a double-blind, randomized, crossover trial of 55 children, those who followed a low FODMAP (fermentable oligo-di-monosaccharides and polyols) diet decreased abdominal pain frequency 20%-30%, explained Dr. Bruno Chumpitazi of Texas Children’s Hospital, Houston.
In a video interview, Dr. Chumpitazi discusses the study's implications for the nearly 20% of school-age children and adolescents who may have IBS, and whether the low FODMAP diet could be a first-line therapy for those children.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – A diet that restricts wheat, beans, dairy, and fruit juice in children with irritable bowel syndrome reduced the frequency and severity of abdominal pain, according to a study presented at the annual Digestive Disease Week.
In a double-blind, randomized, crossover trial of 55 children, those who followed a low FODMAP (fermentable oligo-di-monosaccharides and polyols) diet decreased abdominal pain frequency 20%-30%, explained Dr. Bruno Chumpitazi of Texas Children’s Hospital, Houston.
In a video interview, Dr. Chumpitazi discusses the study's implications for the nearly 20% of school-age children and adolescents who may have IBS, and whether the low FODMAP diet could be a first-line therapy for those children.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
At DDW 2014
Initial dilation no help in eosinophilic esophagitis
CHICAGO – Esophageal dilation combined with standard medical management of eosinophilic esophagitis doesn’t provide added benefit over medication alone in terms of dysphagia relief, according to a randomized, blinded clinical trial.
"In our group of patients with moderate endoscopic findings and without severe stricturing disease, esophageal dilation does not appear to be a necessary additional treatment strategy," Dr. Robert T. Kavitt stated at the annual Digestive Disease Week.
The study involved 31 patients newly diagnosed with eosinophilic esophagitis and baseline moderate to severe difficulty in swallowing. They were randomized to dilation or no dilation at initial endoscopy. Then all patients received standard medical management with 440 mcg of swallowed fluticasone b.i.d. and dexlansoprazole at 60 mg/day for 2 months. Patients were blinded as to their dilation status, as were the physicians who rated their change in dysphagia scores during follow-up.
Both groups experienced robust albeit equal reductions in overall dysphagia scores upon assessment at 30 and 60 days after endoscopy. At baseline, dysphagia scores averaged 6-6.5 on a 0-9 scale, indicative of moderate to severe dysphagia. At follow-up, scores in both groups had dropped to an average of 3 or less, reported Dr. Kavitt of the University of Chicago.
Complete resolution of dysphagia, defined as a dysphagia score of 0, occurred in only 23% of the dilation group and 57% of the no-dilation controls, which was not a statistically significant difference. The looser standard of "significant improvement" – meaning a dysphagia score of 3 or less – was met by 92% of the dilation group and 86% of controls.
Two patients in the dilation group and one control reported odynophagia.
Patients in the dilation group were dilated to the endpoint of mucosal tear. Three-quarters of the patients were dilated to a maximum size of 50 French or larger.
Baseline endoscopic scores assessing strictures, fissures, rings, and other abnormalities were in the moderate range on a 0-13 severity scale, so the study finding of a lack of benefit for dilation as part of an initial treatment strategy in eosinophilic esophagitis may not extend to the minority of patients having truly severe stricturing disease, according to Dr. Kavitt.
He noted that, before this study, the role of dilation in the treatment of eosinophilic esophagitis was a matter of divergent expert opinion unsupported by randomized trial evidence. The 2013 American College of Gastroenterology guidelines state that "the role of dilation as a primary monotherapy of eosinophilic esophagitis is still controversial and should be individualized."
Later during the meeting, in his state-of-the-art lecture on changing therapeutic concepts in eosinophilic esophagitis, Dr. Ikuo Hirano cited Dr. Kavitt’s randomized trial in support of his argument against dilation as primary therapy.
"Dilation does nothing to address the underlying inflammatory response that’s causing strictures to form," noted Dr. Hirano, professor of medicine at Northwestern University, Chicago. "I believe that dilation is inappropriate therapy for children and adults with a predominantly inflammatory phenotype of disease."
Medication and diet therapies not only relieve the symptoms of eosinophilic esophagitis, he continued, they also improve the histopathology.
Dilation entails considerable pain as well as a risk of perforation. Recent reassuring safety data regarding dilation for eosinophilic esophagitis come from specialized esophageal centers with an unusual amount of experience with the procedure, the gastroenterologist said.
Dr. Kavitt reported having no financial conflicts of interest with regard to the randomized trial, which was supported by institutional funds. Dr. Hirano serves as a consultant to Meritage Pharma, Receptos, and Aptalis.
This is the first prospective examination of the role of esophageal dilation in eosinophilic esophagitis (EoE). The symptom-based outcomes suggest that dilation is not necessary for the initial therapy of EoE. The improvement in dysphagia was equivalent in patients treated with dilation combined with medical therapy, compared with those treated with medical therapy alone.

|
| Dr. Ikuo Hirano |
Moreover, while all patients randomized to dilation had dysphagia, not all had endoscopically identified esophageal strictures. One would not expect therapeutic gain for esophageal dilation in the absence of an identifiable stricture. Furthermore, randomized controlled trials of medical therapy for EoE have identified a surprisingly high placebo-response rate for the outcome of dysphagia that may have made demonstration of the benefits of dilation difficult to detect, especially in patients with mild disease severity. Symptom improvement in dysphagia may occur in response to modifications in eating habits such as meticulous mastication or avoidance of highly textured foods.
Finally, the inability to discern a difference in treatment groups may reflect the use of a nonvalidated dysphagia severity assessment instrument.
Despite these shortcomings, the results support current guideline recommendations that dilation should target aspects of esophageal remodeling in EoE that are not amenable to currently available medical or diet therapies.
The data, however, do not exclude an important therapeutic benefit of esophageal dilation in EoE. The study excluded patients with significant strictures that could not be traversed with a standard adult endoscope. The findings, therefore, would not apply to EoE patients with substantial fibrostenosis.
In this study, medical therapy was highly effective at relieving symptoms, but prior studies have shown that symptoms recur in almost all patients following cessation of medications.
Dilation, on the other hand, is generally safe and provides long-lasting relief of dysphagia in adults, even in the absence of medical or diet therapy. Additional prospective studies are needed to define the most appropriate patient subgroups that would benefit from dilation.
Dr. Ikuo Hirano is professor of medicine in the division of gastroenterology at Northwestern University's Feinberg School of Medicine in Chicago. He serves as a consultant to Meritage Pharma, Receptos, and Aptalis.
This is the first prospective examination of the role of esophageal dilation in eosinophilic esophagitis (EoE). The symptom-based outcomes suggest that dilation is not necessary for the initial therapy of EoE. The improvement in dysphagia was equivalent in patients treated with dilation combined with medical therapy, compared with those treated with medical therapy alone.

|
| Dr. Ikuo Hirano |
Moreover, while all patients randomized to dilation had dysphagia, not all had endoscopically identified esophageal strictures. One would not expect therapeutic gain for esophageal dilation in the absence of an identifiable stricture. Furthermore, randomized controlled trials of medical therapy for EoE have identified a surprisingly high placebo-response rate for the outcome of dysphagia that may have made demonstration of the benefits of dilation difficult to detect, especially in patients with mild disease severity. Symptom improvement in dysphagia may occur in response to modifications in eating habits such as meticulous mastication or avoidance of highly textured foods.
Finally, the inability to discern a difference in treatment groups may reflect the use of a nonvalidated dysphagia severity assessment instrument.
Despite these shortcomings, the results support current guideline recommendations that dilation should target aspects of esophageal remodeling in EoE that are not amenable to currently available medical or diet therapies.
The data, however, do not exclude an important therapeutic benefit of esophageal dilation in EoE. The study excluded patients with significant strictures that could not be traversed with a standard adult endoscope. The findings, therefore, would not apply to EoE patients with substantial fibrostenosis.
In this study, medical therapy was highly effective at relieving symptoms, but prior studies have shown that symptoms recur in almost all patients following cessation of medications.
Dilation, on the other hand, is generally safe and provides long-lasting relief of dysphagia in adults, even in the absence of medical or diet therapy. Additional prospective studies are needed to define the most appropriate patient subgroups that would benefit from dilation.
Dr. Ikuo Hirano is professor of medicine in the division of gastroenterology at Northwestern University's Feinberg School of Medicine in Chicago. He serves as a consultant to Meritage Pharma, Receptos, and Aptalis.
This is the first prospective examination of the role of esophageal dilation in eosinophilic esophagitis (EoE). The symptom-based outcomes suggest that dilation is not necessary for the initial therapy of EoE. The improvement in dysphagia was equivalent in patients treated with dilation combined with medical therapy, compared with those treated with medical therapy alone.

|
| Dr. Ikuo Hirano |
Moreover, while all patients randomized to dilation had dysphagia, not all had endoscopically identified esophageal strictures. One would not expect therapeutic gain for esophageal dilation in the absence of an identifiable stricture. Furthermore, randomized controlled trials of medical therapy for EoE have identified a surprisingly high placebo-response rate for the outcome of dysphagia that may have made demonstration of the benefits of dilation difficult to detect, especially in patients with mild disease severity. Symptom improvement in dysphagia may occur in response to modifications in eating habits such as meticulous mastication or avoidance of highly textured foods.
Finally, the inability to discern a difference in treatment groups may reflect the use of a nonvalidated dysphagia severity assessment instrument.
Despite these shortcomings, the results support current guideline recommendations that dilation should target aspects of esophageal remodeling in EoE that are not amenable to currently available medical or diet therapies.
The data, however, do not exclude an important therapeutic benefit of esophageal dilation in EoE. The study excluded patients with significant strictures that could not be traversed with a standard adult endoscope. The findings, therefore, would not apply to EoE patients with substantial fibrostenosis.
In this study, medical therapy was highly effective at relieving symptoms, but prior studies have shown that symptoms recur in almost all patients following cessation of medications.
Dilation, on the other hand, is generally safe and provides long-lasting relief of dysphagia in adults, even in the absence of medical or diet therapy. Additional prospective studies are needed to define the most appropriate patient subgroups that would benefit from dilation.
Dr. Ikuo Hirano is professor of medicine in the division of gastroenterology at Northwestern University's Feinberg School of Medicine in Chicago. He serves as a consultant to Meritage Pharma, Receptos, and Aptalis.
CHICAGO – Esophageal dilation combined with standard medical management of eosinophilic esophagitis doesn’t provide added benefit over medication alone in terms of dysphagia relief, according to a randomized, blinded clinical trial.
"In our group of patients with moderate endoscopic findings and without severe stricturing disease, esophageal dilation does not appear to be a necessary additional treatment strategy," Dr. Robert T. Kavitt stated at the annual Digestive Disease Week.
The study involved 31 patients newly diagnosed with eosinophilic esophagitis and baseline moderate to severe difficulty in swallowing. They were randomized to dilation or no dilation at initial endoscopy. Then all patients received standard medical management with 440 mcg of swallowed fluticasone b.i.d. and dexlansoprazole at 60 mg/day for 2 months. Patients were blinded as to their dilation status, as were the physicians who rated their change in dysphagia scores during follow-up.
Both groups experienced robust albeit equal reductions in overall dysphagia scores upon assessment at 30 and 60 days after endoscopy. At baseline, dysphagia scores averaged 6-6.5 on a 0-9 scale, indicative of moderate to severe dysphagia. At follow-up, scores in both groups had dropped to an average of 3 or less, reported Dr. Kavitt of the University of Chicago.
Complete resolution of dysphagia, defined as a dysphagia score of 0, occurred in only 23% of the dilation group and 57% of the no-dilation controls, which was not a statistically significant difference. The looser standard of "significant improvement" – meaning a dysphagia score of 3 or less – was met by 92% of the dilation group and 86% of controls.
Two patients in the dilation group and one control reported odynophagia.
Patients in the dilation group were dilated to the endpoint of mucosal tear. Three-quarters of the patients were dilated to a maximum size of 50 French or larger.
Baseline endoscopic scores assessing strictures, fissures, rings, and other abnormalities were in the moderate range on a 0-13 severity scale, so the study finding of a lack of benefit for dilation as part of an initial treatment strategy in eosinophilic esophagitis may not extend to the minority of patients having truly severe stricturing disease, according to Dr. Kavitt.
He noted that, before this study, the role of dilation in the treatment of eosinophilic esophagitis was a matter of divergent expert opinion unsupported by randomized trial evidence. The 2013 American College of Gastroenterology guidelines state that "the role of dilation as a primary monotherapy of eosinophilic esophagitis is still controversial and should be individualized."
Later during the meeting, in his state-of-the-art lecture on changing therapeutic concepts in eosinophilic esophagitis, Dr. Ikuo Hirano cited Dr. Kavitt’s randomized trial in support of his argument against dilation as primary therapy.
"Dilation does nothing to address the underlying inflammatory response that’s causing strictures to form," noted Dr. Hirano, professor of medicine at Northwestern University, Chicago. "I believe that dilation is inappropriate therapy for children and adults with a predominantly inflammatory phenotype of disease."
Medication and diet therapies not only relieve the symptoms of eosinophilic esophagitis, he continued, they also improve the histopathology.
Dilation entails considerable pain as well as a risk of perforation. Recent reassuring safety data regarding dilation for eosinophilic esophagitis come from specialized esophageal centers with an unusual amount of experience with the procedure, the gastroenterologist said.
Dr. Kavitt reported having no financial conflicts of interest with regard to the randomized trial, which was supported by institutional funds. Dr. Hirano serves as a consultant to Meritage Pharma, Receptos, and Aptalis.
CHICAGO – Esophageal dilation combined with standard medical management of eosinophilic esophagitis doesn’t provide added benefit over medication alone in terms of dysphagia relief, according to a randomized, blinded clinical trial.
"In our group of patients with moderate endoscopic findings and without severe stricturing disease, esophageal dilation does not appear to be a necessary additional treatment strategy," Dr. Robert T. Kavitt stated at the annual Digestive Disease Week.
The study involved 31 patients newly diagnosed with eosinophilic esophagitis and baseline moderate to severe difficulty in swallowing. They were randomized to dilation or no dilation at initial endoscopy. Then all patients received standard medical management with 440 mcg of swallowed fluticasone b.i.d. and dexlansoprazole at 60 mg/day for 2 months. Patients were blinded as to their dilation status, as were the physicians who rated their change in dysphagia scores during follow-up.
Both groups experienced robust albeit equal reductions in overall dysphagia scores upon assessment at 30 and 60 days after endoscopy. At baseline, dysphagia scores averaged 6-6.5 on a 0-9 scale, indicative of moderate to severe dysphagia. At follow-up, scores in both groups had dropped to an average of 3 or less, reported Dr. Kavitt of the University of Chicago.
Complete resolution of dysphagia, defined as a dysphagia score of 0, occurred in only 23% of the dilation group and 57% of the no-dilation controls, which was not a statistically significant difference. The looser standard of "significant improvement" – meaning a dysphagia score of 3 or less – was met by 92% of the dilation group and 86% of controls.
Two patients in the dilation group and one control reported odynophagia.
Patients in the dilation group were dilated to the endpoint of mucosal tear. Three-quarters of the patients were dilated to a maximum size of 50 French or larger.
Baseline endoscopic scores assessing strictures, fissures, rings, and other abnormalities were in the moderate range on a 0-13 severity scale, so the study finding of a lack of benefit for dilation as part of an initial treatment strategy in eosinophilic esophagitis may not extend to the minority of patients having truly severe stricturing disease, according to Dr. Kavitt.
He noted that, before this study, the role of dilation in the treatment of eosinophilic esophagitis was a matter of divergent expert opinion unsupported by randomized trial evidence. The 2013 American College of Gastroenterology guidelines state that "the role of dilation as a primary monotherapy of eosinophilic esophagitis is still controversial and should be individualized."
Later during the meeting, in his state-of-the-art lecture on changing therapeutic concepts in eosinophilic esophagitis, Dr. Ikuo Hirano cited Dr. Kavitt’s randomized trial in support of his argument against dilation as primary therapy.
"Dilation does nothing to address the underlying inflammatory response that’s causing strictures to form," noted Dr. Hirano, professor of medicine at Northwestern University, Chicago. "I believe that dilation is inappropriate therapy for children and adults with a predominantly inflammatory phenotype of disease."
Medication and diet therapies not only relieve the symptoms of eosinophilic esophagitis, he continued, they also improve the histopathology.
Dilation entails considerable pain as well as a risk of perforation. Recent reassuring safety data regarding dilation for eosinophilic esophagitis come from specialized esophageal centers with an unusual amount of experience with the procedure, the gastroenterologist said.
Dr. Kavitt reported having no financial conflicts of interest with regard to the randomized trial, which was supported by institutional funds. Dr. Hirano serves as a consultant to Meritage Pharma, Receptos, and Aptalis.
AT DDW 2014
Key clinical point: Dilation offers no added benefit over medication alone in relieving dysphagia symptoms in eosinophilic esophagitis.
Major finding: Significant improvement in dysphagia symptoms was documented 30 and 60 days post endoscopy in 92% of eosinophilic esophagitis patients who underwent dilation plus medical therapy and in 86% who got medication alone.
Data source: This was a randomized trial involving 31 patients newly diagnosed with eosinophilic esophagitis who were assigned to dilation or no dilation at initial endoscopy, after which all participants received 2 months of medical therapy with a swallowed corticosteroid and a proton pump inhibitor. Patients as well as the physicians who rated their dysphagia symptoms 30 and 60 days post endoscopy were blinded as to dilation status.
Disclosures: Dr. Kavitt reported having no financial conflicts regarding this study, which was supported by institutional funds. Dr. Hirano serves as a consultant to Meritage Pharma, Receptos, and Aptalis.
Bariatric surgery improved liver histopathology in NAFLD
CHICAGO – Grade 2 or 3 hepatic fibrosis in the setting of nonalcoholic fatty liver disease resolved or improved in 56% of affected patients following their bariatric surgery for severe obesity, according to a large blinded, paired-biopsy study.
Bariatric surgery in patients with nonalcoholic fatty liver disease (NAFLD) also achieved a high rate of resolution of steatosis and steatohepatitis, but it was the improvement in advanced fibrosis, including stage 3 or bridging fibrosis, that was particularly impressive. Traditionally, liver fibrosis was thought to be an irreversible finding, Dr. Andrew A. Taitano observed at the annual Digestive Disease Week.
"Our data provide strong evidence that bariatric surgery improves liver histology in NAFLD," he said. "We conclude that bariatric surgery should be considered one of the treatments for NAFLD in patients with severe obesity."
Dr. Taitano presented a retrospective, single-center study of 160 patients with NAFLD at the time they underwent bariatric surgery for weight loss, all of whom had follow-up liver biopsies when they underwent abdominal operations for any reason an average of 31 months later. Their mean body-mass index was 52 kg/m2 at bariatric surgery, with a 62% excess body weight loss at the time of their subsequent abdominal surgery.
At bariatric surgery, 65% of patients had hepatic fibrosis. At follow-up, two blinded pathologists reported that only 36% of patients had liver fibrosis. Fibrosis was resolved or improved by at least one grade in 56% of patients, worse in 16%, and unchanged in the rest, reported Dr. Taitano, a bariatric surgery fellow at the University of South Florida, Tampa.
Of 56 patients without baseline liver fibrosis, only 12 had developed fibrosis at follow-up.
Steatosis was present in 77% of patients at bariatric surgery and in 21% at follow-up. Steatosis was resolved in 86% of patients at follow-up, the same in 8%, and worse in 6%, he said.
Steatohepatitis was found on liver biopsies at bariatric surgery in 26% of patients but was present in only 3% at follow-up. This form of histopathology was resolved or improved at follow-up in 93% of affected patients; fibrosis was worse in none.
NAFLD is the most common liver disorder in Western countries. The rate has doubled in the last 20 years. Risk factors include type 2 diabetes, central obesity, and dyslipidemia – all features of the metabolic syndrome. NAFLD is a progressive disease. Previous studies indicate that up to half of patients with NAFLD develop fibrosis within 13 years, and it’s estimated that one in five patients with NAFLD with steatohepatitis will develop cirrhosis within 20 years.
Discussant Dr. Guilherme M. Campos, a surgeon at the University of Wisconsin, Madison, commented, "It’s important to underscore that the magnitude of histologic improvements observed here is far greater than seen with maximum nonoperative therapy."
Asked if the study data pointed to any particular type of bypass surgery or any patient characteristics that correlate with change in NAFLD histopathology, Dr. Taitano replied that gastric bypass was the predominant operation. Although the limited patient numbers didn’t permit meaningful comparisons, he said, "as an overall recommendation, I would advise a weight-loss procedure that has a metabolic effect, such as gastric bypass, because I think both the amount of weight loss and the operation’s metabolic effect are improving this disease."
He reported having no financial conflicts regarding this study, which was conducted using institutional funds.
CHICAGO – Grade 2 or 3 hepatic fibrosis in the setting of nonalcoholic fatty liver disease resolved or improved in 56% of affected patients following their bariatric surgery for severe obesity, according to a large blinded, paired-biopsy study.
Bariatric surgery in patients with nonalcoholic fatty liver disease (NAFLD) also achieved a high rate of resolution of steatosis and steatohepatitis, but it was the improvement in advanced fibrosis, including stage 3 or bridging fibrosis, that was particularly impressive. Traditionally, liver fibrosis was thought to be an irreversible finding, Dr. Andrew A. Taitano observed at the annual Digestive Disease Week.
"Our data provide strong evidence that bariatric surgery improves liver histology in NAFLD," he said. "We conclude that bariatric surgery should be considered one of the treatments for NAFLD in patients with severe obesity."
Dr. Taitano presented a retrospective, single-center study of 160 patients with NAFLD at the time they underwent bariatric surgery for weight loss, all of whom had follow-up liver biopsies when they underwent abdominal operations for any reason an average of 31 months later. Their mean body-mass index was 52 kg/m2 at bariatric surgery, with a 62% excess body weight loss at the time of their subsequent abdominal surgery.
At bariatric surgery, 65% of patients had hepatic fibrosis. At follow-up, two blinded pathologists reported that only 36% of patients had liver fibrosis. Fibrosis was resolved or improved by at least one grade in 56% of patients, worse in 16%, and unchanged in the rest, reported Dr. Taitano, a bariatric surgery fellow at the University of South Florida, Tampa.
Of 56 patients without baseline liver fibrosis, only 12 had developed fibrosis at follow-up.
Steatosis was present in 77% of patients at bariatric surgery and in 21% at follow-up. Steatosis was resolved in 86% of patients at follow-up, the same in 8%, and worse in 6%, he said.
Steatohepatitis was found on liver biopsies at bariatric surgery in 26% of patients but was present in only 3% at follow-up. This form of histopathology was resolved or improved at follow-up in 93% of affected patients; fibrosis was worse in none.
NAFLD is the most common liver disorder in Western countries. The rate has doubled in the last 20 years. Risk factors include type 2 diabetes, central obesity, and dyslipidemia – all features of the metabolic syndrome. NAFLD is a progressive disease. Previous studies indicate that up to half of patients with NAFLD develop fibrosis within 13 years, and it’s estimated that one in five patients with NAFLD with steatohepatitis will develop cirrhosis within 20 years.
Discussant Dr. Guilherme M. Campos, a surgeon at the University of Wisconsin, Madison, commented, "It’s important to underscore that the magnitude of histologic improvements observed here is far greater than seen with maximum nonoperative therapy."
Asked if the study data pointed to any particular type of bypass surgery or any patient characteristics that correlate with change in NAFLD histopathology, Dr. Taitano replied that gastric bypass was the predominant operation. Although the limited patient numbers didn’t permit meaningful comparisons, he said, "as an overall recommendation, I would advise a weight-loss procedure that has a metabolic effect, such as gastric bypass, because I think both the amount of weight loss and the operation’s metabolic effect are improving this disease."
He reported having no financial conflicts regarding this study, which was conducted using institutional funds.
CHICAGO – Grade 2 or 3 hepatic fibrosis in the setting of nonalcoholic fatty liver disease resolved or improved in 56% of affected patients following their bariatric surgery for severe obesity, according to a large blinded, paired-biopsy study.
Bariatric surgery in patients with nonalcoholic fatty liver disease (NAFLD) also achieved a high rate of resolution of steatosis and steatohepatitis, but it was the improvement in advanced fibrosis, including stage 3 or bridging fibrosis, that was particularly impressive. Traditionally, liver fibrosis was thought to be an irreversible finding, Dr. Andrew A. Taitano observed at the annual Digestive Disease Week.
"Our data provide strong evidence that bariatric surgery improves liver histology in NAFLD," he said. "We conclude that bariatric surgery should be considered one of the treatments for NAFLD in patients with severe obesity."
Dr. Taitano presented a retrospective, single-center study of 160 patients with NAFLD at the time they underwent bariatric surgery for weight loss, all of whom had follow-up liver biopsies when they underwent abdominal operations for any reason an average of 31 months later. Their mean body-mass index was 52 kg/m2 at bariatric surgery, with a 62% excess body weight loss at the time of their subsequent abdominal surgery.
At bariatric surgery, 65% of patients had hepatic fibrosis. At follow-up, two blinded pathologists reported that only 36% of patients had liver fibrosis. Fibrosis was resolved or improved by at least one grade in 56% of patients, worse in 16%, and unchanged in the rest, reported Dr. Taitano, a bariatric surgery fellow at the University of South Florida, Tampa.
Of 56 patients without baseline liver fibrosis, only 12 had developed fibrosis at follow-up.
Steatosis was present in 77% of patients at bariatric surgery and in 21% at follow-up. Steatosis was resolved in 86% of patients at follow-up, the same in 8%, and worse in 6%, he said.
Steatohepatitis was found on liver biopsies at bariatric surgery in 26% of patients but was present in only 3% at follow-up. This form of histopathology was resolved or improved at follow-up in 93% of affected patients; fibrosis was worse in none.
NAFLD is the most common liver disorder in Western countries. The rate has doubled in the last 20 years. Risk factors include type 2 diabetes, central obesity, and dyslipidemia – all features of the metabolic syndrome. NAFLD is a progressive disease. Previous studies indicate that up to half of patients with NAFLD develop fibrosis within 13 years, and it’s estimated that one in five patients with NAFLD with steatohepatitis will develop cirrhosis within 20 years.
Discussant Dr. Guilherme M. Campos, a surgeon at the University of Wisconsin, Madison, commented, "It’s important to underscore that the magnitude of histologic improvements observed here is far greater than seen with maximum nonoperative therapy."
Asked if the study data pointed to any particular type of bypass surgery or any patient characteristics that correlate with change in NAFLD histopathology, Dr. Taitano replied that gastric bypass was the predominant operation. Although the limited patient numbers didn’t permit meaningful comparisons, he said, "as an overall recommendation, I would advise a weight-loss procedure that has a metabolic effect, such as gastric bypass, because I think both the amount of weight loss and the operation’s metabolic effect are improving this disease."
He reported having no financial conflicts regarding this study, which was conducted using institutional funds.
AT DDW 2014
Key clinical point: Bariatric surgery should be considered one of the treatments for NAFLD in patients with severe obesity.
Major finding: Advanced liver fibrosis in patients with nonalcoholic fatty liver disease at the time of bariatric surgery for severe obesity was resolved or improved by at least one grade in nearly 60% of cases at follow-up biopsy.
Data source: A retrospective, blinded, paired-biopsy study involving 160 patients with NAFLD at the time they underwent bariatric surgery and who had follow-up liver biopsies when they later underwent abdominal surgery.
Disclosures: The presenter reported having no financial conflicts regarding this study, which was conducted using institutional funds.
Novel complex shows unique benefits in chronic kidney disease
LAS VEGAS – Treatment with ferric citrate coordination complex conferred multiple benefits with cardioprotective implications in non–dialysis-dependent chronic kidney disease patients with elevated serum phosphate and iron-deficiency anemia in a randomized trial.
Ferric citrate coordination complex (FCCC), an investigational oral product also known as Zerenex, effectively lowered patients’ elevated serum phosphate into normal range while repleting iron stores, boosting hemoglobin, and reducing levels of the cardiotoxic protein fibroblast growth factor 23 (FGF23), Dr. Geoffrey A. Block reported at a meeting sponsored by the National Kidney Foundation.
"We are quite happy with these results and the implications they may have in trying to address cardiovascular risk at multiple levels in chronic kidney disease," declared Dr. Block of Denver Nephrology, who was principal investigator in the phase II study.
FCCC is currently under Food and Drug Administration review for potential marketing approval as a treatment in patients with end-stage renal disease complicated by iron-deficiency anemia and elevated serum phosphate. However, the phase II study led by Dr. Block focused on the much larger patient population with non–dialysis-dependent chronic kidney disease (CKD) with elevated serum phosphate and iron-deficiency anemia. Experience has shown it is far more difficult to lower serum phosphate in such patients than in those with end-stage renal disease, the nephrologist noted.
The 12-week, double-blind clinical trial included 141 subjects with an estimated glomerular filtration rate below 60 mL/min per 1.73 m2, a serum phosphate in excess of 4.0 mg/dL, a transferrin saturation below 30%, a hemoglobin level of 9-12 g/dL, and a serum ferritin below 300 ng/mL. Participants were not permitted to use intravenous iron or an erythropoietin-stimulating agent in the months prior to or during the trial. They were randomized to FCCC titrated to achieve a serum phosphate below 3.5 mg/dL or to placebo.
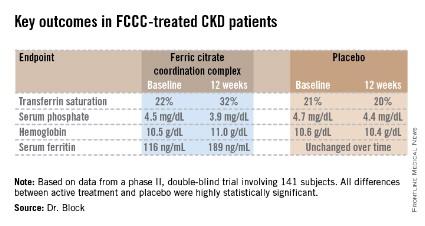
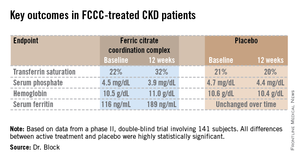
The coprimary endpoints were changes from baseline through 12 weeks in serum phosphate and transferrin saturation. The FCCC-treated patients fared significantly better than controls on those endpoints as well as the secondary outcomes (see chart).
• FGF23: The FCCC group’s 40% drop in C-terminal FGF23 over the course of the 12-week study, while levels remained static in the control group, was particularly noteworthy, according to Dr. Block. In several observational studies, including the Heart and Soul Study (Ann. Intern. Med. 2010;152:640-8), elevated FGF23 levels have been associated with significantly increased risk of cardiovascular events and all-cause mortality.
Elevated FGF23 levels appear to promote left-ventricular hypertrophy, while hyperphosphatemia promotes vascular calcification. Through these different mechanisms, both abnormalities increase the risk of cardiovascular events. Both abnormalities are common in patients with CKD. And FCCC resulted in significant reductions in both FGF23 and serum phosphate, he noted.
A prespecified safety feature of the phase II trial was that any participant whose hemoglobin fell below 9.0 g/dL or whose serum phosphate climbed above 6.0 mg/dL would have to be taken out of the study. One patient on FCCC and nine controls were withdrawn from the study for low hemoglobin. No patients in the FCCC group and two controls were removed due to an excessive phosphate level.
The safety profile of FCCC was essentially the same as for placebo with two exceptions: 20% of the FCCC group reported diarrhea, compared with 6% of controls; and one-third of the FCCC group reported discolored feces as a consequence of iron utilization.
• Serum phosphate: Dr. Block said that "reasonably solid evidence" from observational studies, clinical trials, and animal studies indicates that a serum phosphate level greater than 4.0 mg/dL leads to faster progression of kidney disease and an increase in cardiovascular events. For example, in a fully adjusted reanalysis of data from the randomized prospective REIN (Ramipril Efficacy in Nephropathy) trial, patients with a baseline serum phosphate above 4.0 mg/dL were at greatly increased risk for the combined endpoint of a doubling of serum creatinine or progression to end-stage renal disease (J. Am. Soc. Nephrol. 2011;22:1923-30).
Moreover, the renoprotective effect of ramipril decreased as baseline serum phosphate increased: While the ACE inhibitor reduced the risk of the combined endpoint by 85%, compared with placebo, in subjects with a serum phosphate less than 3.45 mg/dL and by 63% in those with a level of 3.45-4.0 mg/dL, ramipril wasn’t significantly more effective than placebo in those with a serum phosphate above 4.0 mg/dL.
Dietary phosphorus intake, which is high in the United States, has also come under scrutiny as a public health concern. A recent prospective cohort study in 9,686 healthy U.S. adults concluded that consumption of more than 1,400 mg daily – the median intake in this representative study population was 1,166 mg per day – was independently associated with higher all-cause mortality. Those with a phosphorus density greater than 0.35 mg/kcal, a statistic derived by dividing phosphorus intake by energy intake – had a significantly increased risk of cardiovascular mortality (Am. J. Clin. Nutr. 2014;99:320-7). And in an analysis of 4,494 participants in MESA (Multi-Ethnic Study of Atherosclerosis), dietary phosphorus intake was independently associated with greater left-ventricular mass such that subjects in the top dietary phosphorus quintile had a 6.1-g greater left-ventricular mass than those in the lowest quintile (Kidney Intl. 2013;83:707-14).
The three phosphate binders currently on the market for reduction of elevated serum phosphate in CKD patients are "really marginal treatments," according to Dr. Block. He was first author on a study in which 148 patients with moderate CKD were randomized to 9 months of calcium acetate, sevelamer carbonate, lanthanum carbonate, or placebo. Serum phosphorus inched lower over the 9 months from a baseline of 4.2 mg/dL to 3.9 mg/dL with active therapy, which was only 0.2 mg/dL better than with placebo. In contrast, serum phosphate fell by an average of 0.6 mg/dL during 3 months on FCCC in the phase II study. Moreover, active therapy with the commercially available phosphate binders had no effect upon FGF23 levels, and it significantly increased coronary artery and abdominal aorta calcification by a median of 18% and 15%, respectively (J. Am. Soc. Nephrol. 2012;23:1407-15).
• Iron-deficiency anemia: The Kidney Dialysis International Guideline Organization defines iron deficiency warranting iron supplementation in CKD patients as a transferrin saturation of 30% or less and a serum ferritin of 50 ng/mL or less. By those criteria, it is estimated that nearly 70% of CKD patients are iron deficient. So there is a large unmet need for iron repletion therapies that avoid the use of erythropoietin-stimulating agents and intravenous iron, Dr. Block noted.
The FCCC trial was sponsored by Keryx Biopharmaceuticals. Dr. Block serves as a consultant to the company and was principal investigator in the study.
LAS VEGAS – Treatment with ferric citrate coordination complex conferred multiple benefits with cardioprotective implications in non–dialysis-dependent chronic kidney disease patients with elevated serum phosphate and iron-deficiency anemia in a randomized trial.
Ferric citrate coordination complex (FCCC), an investigational oral product also known as Zerenex, effectively lowered patients’ elevated serum phosphate into normal range while repleting iron stores, boosting hemoglobin, and reducing levels of the cardiotoxic protein fibroblast growth factor 23 (FGF23), Dr. Geoffrey A. Block reported at a meeting sponsored by the National Kidney Foundation.

"We are quite happy with these results and the implications they may have in trying to address cardiovascular risk at multiple levels in chronic kidney disease," declared Dr. Block of Denver Nephrology, who was principal investigator in the phase II study.
FCCC is currently under Food and Drug Administration review for potential marketing approval as a treatment in patients with end-stage renal disease complicated by iron-deficiency anemia and elevated serum phosphate. However, the phase II study led by Dr. Block focused on the much larger patient population with non–dialysis-dependent chronic kidney disease (CKD) with elevated serum phosphate and iron-deficiency anemia. Experience has shown it is far more difficult to lower serum phosphate in such patients than in those with end-stage renal disease, the nephrologist noted.
The 12-week, double-blind clinical trial included 141 subjects with an estimated glomerular filtration rate below 60 mL/min per 1.73 m2, a serum phosphate in excess of 4.0 mg/dL, a transferrin saturation below 30%, a hemoglobin level of 9-12 g/dL, and a serum ferritin below 300 ng/mL. Participants were not permitted to use intravenous iron or an erythropoietin-stimulating agent in the months prior to or during the trial. They were randomized to FCCC titrated to achieve a serum phosphate below 3.5 mg/dL or to placebo.
The coprimary endpoints were changes from baseline through 12 weeks in serum phosphate and transferrin saturation. The FCCC-treated patients fared significantly better than controls on those endpoints as well as the secondary outcomes (see chart).
• FGF23: The FCCC group’s 40% drop in C-terminal FGF23 over the course of the 12-week study, while levels remained static in the control group, was particularly noteworthy, according to Dr. Block. In several observational studies, including the Heart and Soul Study (Ann. Intern. Med. 2010;152:640-8), elevated FGF23 levels have been associated with significantly increased risk of cardiovascular events and all-cause mortality.
Elevated FGF23 levels appear to promote left-ventricular hypertrophy, while hyperphosphatemia promotes vascular calcification. Through these different mechanisms, both abnormalities increase the risk of cardiovascular events. Both abnormalities are common in patients with CKD. And FCCC resulted in significant reductions in both FGF23 and serum phosphate, he noted.
A prespecified safety feature of the phase II trial was that any participant whose hemoglobin fell below 9.0 g/dL or whose serum phosphate climbed above 6.0 mg/dL would have to be taken out of the study. One patient on FCCC and nine controls were withdrawn from the study for low hemoglobin. No patients in the FCCC group and two controls were removed due to an excessive phosphate level.
The safety profile of FCCC was essentially the same as for placebo with two exceptions: 20% of the FCCC group reported diarrhea, compared with 6% of controls; and one-third of the FCCC group reported discolored feces as a consequence of iron utilization.
• Serum phosphate: Dr. Block said that "reasonably solid evidence" from observational studies, clinical trials, and animal studies indicates that a serum phosphate level greater than 4.0 mg/dL leads to faster progression of kidney disease and an increase in cardiovascular events. For example, in a fully adjusted reanalysis of data from the randomized prospective REIN (Ramipril Efficacy in Nephropathy) trial, patients with a baseline serum phosphate above 4.0 mg/dL were at greatly increased risk for the combined endpoint of a doubling of serum creatinine or progression to end-stage renal disease (J. Am. Soc. Nephrol. 2011;22:1923-30).
Moreover, the renoprotective effect of ramipril decreased as baseline serum phosphate increased: While the ACE inhibitor reduced the risk of the combined endpoint by 85%, compared with placebo, in subjects with a serum phosphate less than 3.45 mg/dL and by 63% in those with a level of 3.45-4.0 mg/dL, ramipril wasn’t significantly more effective than placebo in those with a serum phosphate above 4.0 mg/dL.
Dietary phosphorus intake, which is high in the United States, has also come under scrutiny as a public health concern. A recent prospective cohort study in 9,686 healthy U.S. adults concluded that consumption of more than 1,400 mg daily – the median intake in this representative study population was 1,166 mg per day – was independently associated with higher all-cause mortality. Those with a phosphorus density greater than 0.35 mg/kcal, a statistic derived by dividing phosphorus intake by energy intake – had a significantly increased risk of cardiovascular mortality (Am. J. Clin. Nutr. 2014;99:320-7). And in an analysis of 4,494 participants in MESA (Multi-Ethnic Study of Atherosclerosis), dietary phosphorus intake was independently associated with greater left-ventricular mass such that subjects in the top dietary phosphorus quintile had a 6.1-g greater left-ventricular mass than those in the lowest quintile (Kidney Intl. 2013;83:707-14).
The three phosphate binders currently on the market for reduction of elevated serum phosphate in CKD patients are "really marginal treatments," according to Dr. Block. He was first author on a study in which 148 patients with moderate CKD were randomized to 9 months of calcium acetate, sevelamer carbonate, lanthanum carbonate, or placebo. Serum phosphorus inched lower over the 9 months from a baseline of 4.2 mg/dL to 3.9 mg/dL with active therapy, which was only 0.2 mg/dL better than with placebo. In contrast, serum phosphate fell by an average of 0.6 mg/dL during 3 months on FCCC in the phase II study. Moreover, active therapy with the commercially available phosphate binders had no effect upon FGF23 levels, and it significantly increased coronary artery and abdominal aorta calcification by a median of 18% and 15%, respectively (J. Am. Soc. Nephrol. 2012;23:1407-15).
• Iron-deficiency anemia: The Kidney Dialysis International Guideline Organization defines iron deficiency warranting iron supplementation in CKD patients as a transferrin saturation of 30% or less and a serum ferritin of 50 ng/mL or less. By those criteria, it is estimated that nearly 70% of CKD patients are iron deficient. So there is a large unmet need for iron repletion therapies that avoid the use of erythropoietin-stimulating agents and intravenous iron, Dr. Block noted.
The FCCC trial was sponsored by Keryx Biopharmaceuticals. Dr. Block serves as a consultant to the company and was principal investigator in the study.
LAS VEGAS – Treatment with ferric citrate coordination complex conferred multiple benefits with cardioprotective implications in non–dialysis-dependent chronic kidney disease patients with elevated serum phosphate and iron-deficiency anemia in a randomized trial.
Ferric citrate coordination complex (FCCC), an investigational oral product also known as Zerenex, effectively lowered patients’ elevated serum phosphate into normal range while repleting iron stores, boosting hemoglobin, and reducing levels of the cardiotoxic protein fibroblast growth factor 23 (FGF23), Dr. Geoffrey A. Block reported at a meeting sponsored by the National Kidney Foundation.

"We are quite happy with these results and the implications they may have in trying to address cardiovascular risk at multiple levels in chronic kidney disease," declared Dr. Block of Denver Nephrology, who was principal investigator in the phase II study.
FCCC is currently under Food and Drug Administration review for potential marketing approval as a treatment in patients with end-stage renal disease complicated by iron-deficiency anemia and elevated serum phosphate. However, the phase II study led by Dr. Block focused on the much larger patient population with non–dialysis-dependent chronic kidney disease (CKD) with elevated serum phosphate and iron-deficiency anemia. Experience has shown it is far more difficult to lower serum phosphate in such patients than in those with end-stage renal disease, the nephrologist noted.
The 12-week, double-blind clinical trial included 141 subjects with an estimated glomerular filtration rate below 60 mL/min per 1.73 m2, a serum phosphate in excess of 4.0 mg/dL, a transferrin saturation below 30%, a hemoglobin level of 9-12 g/dL, and a serum ferritin below 300 ng/mL. Participants were not permitted to use intravenous iron or an erythropoietin-stimulating agent in the months prior to or during the trial. They were randomized to FCCC titrated to achieve a serum phosphate below 3.5 mg/dL or to placebo.
The coprimary endpoints were changes from baseline through 12 weeks in serum phosphate and transferrin saturation. The FCCC-treated patients fared significantly better than controls on those endpoints as well as the secondary outcomes (see chart).
• FGF23: The FCCC group’s 40% drop in C-terminal FGF23 over the course of the 12-week study, while levels remained static in the control group, was particularly noteworthy, according to Dr. Block. In several observational studies, including the Heart and Soul Study (Ann. Intern. Med. 2010;152:640-8), elevated FGF23 levels have been associated with significantly increased risk of cardiovascular events and all-cause mortality.
Elevated FGF23 levels appear to promote left-ventricular hypertrophy, while hyperphosphatemia promotes vascular calcification. Through these different mechanisms, both abnormalities increase the risk of cardiovascular events. Both abnormalities are common in patients with CKD. And FCCC resulted in significant reductions in both FGF23 and serum phosphate, he noted.
A prespecified safety feature of the phase II trial was that any participant whose hemoglobin fell below 9.0 g/dL or whose serum phosphate climbed above 6.0 mg/dL would have to be taken out of the study. One patient on FCCC and nine controls were withdrawn from the study for low hemoglobin. No patients in the FCCC group and two controls were removed due to an excessive phosphate level.
The safety profile of FCCC was essentially the same as for placebo with two exceptions: 20% of the FCCC group reported diarrhea, compared with 6% of controls; and one-third of the FCCC group reported discolored feces as a consequence of iron utilization.
• Serum phosphate: Dr. Block said that "reasonably solid evidence" from observational studies, clinical trials, and animal studies indicates that a serum phosphate level greater than 4.0 mg/dL leads to faster progression of kidney disease and an increase in cardiovascular events. For example, in a fully adjusted reanalysis of data from the randomized prospective REIN (Ramipril Efficacy in Nephropathy) trial, patients with a baseline serum phosphate above 4.0 mg/dL were at greatly increased risk for the combined endpoint of a doubling of serum creatinine or progression to end-stage renal disease (J. Am. Soc. Nephrol. 2011;22:1923-30).
Moreover, the renoprotective effect of ramipril decreased as baseline serum phosphate increased: While the ACE inhibitor reduced the risk of the combined endpoint by 85%, compared with placebo, in subjects with a serum phosphate less than 3.45 mg/dL and by 63% in those with a level of 3.45-4.0 mg/dL, ramipril wasn’t significantly more effective than placebo in those with a serum phosphate above 4.0 mg/dL.
Dietary phosphorus intake, which is high in the United States, has also come under scrutiny as a public health concern. A recent prospective cohort study in 9,686 healthy U.S. adults concluded that consumption of more than 1,400 mg daily – the median intake in this representative study population was 1,166 mg per day – was independently associated with higher all-cause mortality. Those with a phosphorus density greater than 0.35 mg/kcal, a statistic derived by dividing phosphorus intake by energy intake – had a significantly increased risk of cardiovascular mortality (Am. J. Clin. Nutr. 2014;99:320-7). And in an analysis of 4,494 participants in MESA (Multi-Ethnic Study of Atherosclerosis), dietary phosphorus intake was independently associated with greater left-ventricular mass such that subjects in the top dietary phosphorus quintile had a 6.1-g greater left-ventricular mass than those in the lowest quintile (Kidney Intl. 2013;83:707-14).
The three phosphate binders currently on the market for reduction of elevated serum phosphate in CKD patients are "really marginal treatments," according to Dr. Block. He was first author on a study in which 148 patients with moderate CKD were randomized to 9 months of calcium acetate, sevelamer carbonate, lanthanum carbonate, or placebo. Serum phosphorus inched lower over the 9 months from a baseline of 4.2 mg/dL to 3.9 mg/dL with active therapy, which was only 0.2 mg/dL better than with placebo. In contrast, serum phosphate fell by an average of 0.6 mg/dL during 3 months on FCCC in the phase II study. Moreover, active therapy with the commercially available phosphate binders had no effect upon FGF23 levels, and it significantly increased coronary artery and abdominal aorta calcification by a median of 18% and 15%, respectively (J. Am. Soc. Nephrol. 2012;23:1407-15).
• Iron-deficiency anemia: The Kidney Dialysis International Guideline Organization defines iron deficiency warranting iron supplementation in CKD patients as a transferrin saturation of 30% or less and a serum ferritin of 50 ng/mL or less. By those criteria, it is estimated that nearly 70% of CKD patients are iron deficient. So there is a large unmet need for iron repletion therapies that avoid the use of erythropoietin-stimulating agents and intravenous iron, Dr. Block noted.
The FCCC trial was sponsored by Keryx Biopharmaceuticals. Dr. Block serves as a consultant to the company and was principal investigator in the study.
AT SCM 14
Major finding: FCCC safely repleted low iron stores, reduced elevated serum phosphate, raised hemoglobin, and slashed FGF23 levels, compared with placebo.
Data source: A prospective, double-blind, placebo-controlled, 12-week trial in which 141 patients with non–dialysis-dependent CKD, iron-deficiency anemia, and elevated serum phosphate were assigned to FCCC or placebo.
Disclosures: The study was sponsored by Keryx Biopharmaceuticals. Dr. Block is a consultant to the company and was principal investigator in the trial.
Chronic kidney disease may raise cancer risk
LAS VEGAS – Patients with chronic kidney disease appear to be at increased risk for cancer, a study showed.
A retrospective analysis of data on 31,896 participants in ALLHAT (the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial) showed that during a mean 4.9 years of in-trial follow-up, 2,529 subjects were diagnosed with various cancers.
The 5-year rate of incident cancer was 7.24 cases per 100 person-years among subjects with a baseline normal estimated glomerular filtration rate (eGFR) greater than 90 mL/min per 1.73 m2. The cancer rate rose with decreasing renal function: 8.38 cases per 100 person-years in patients with a baseline eGFR of 60-89.9, 9.18 per 100 person-years in those with an eGFR of 45-59.9, and 11.58 per 100 person-years in patients with an eGFR below 45 mL/min per 1.73 m2, Dr. Dhruti P. Chen reported at a meeting sponsored by the National Kidney Foundation.
An additional 4 years of follow-up for mortality due to cancer was available through national databases after ALLHAT ended. During the mean total 8.9 years of follow-up, there were 2,338 cancer-related deaths. The 10-year rate of cancer mortality was 7.90 cases per 100 person-years in patients with an eGFR greater than 90; 7.71 in those with an eGFR of 60-89.9; 10.11 per 100 person-years with an eGFR of 45-59.9; and 13.19 per 100 person-years in patients with an eGFR below 45 mL/min per 1.73 m2, added Dr. Chen of the department of medicine at Case Western Reserve University, Cleveland.
In a multivariate analysis adjusted for demographics, body mass index, diabetes, cardiovascular risk factors, blood glucose level, and the antihypertensive agent to which a patient was randomized, the association between chronic kidney disease and incident cancer was attenuated. Nonetheless, having a baseline eGFR below 45 mL/min per 1.73 m2 was independently associated with an adjusted 28% increased risk of incident cancer during 4.9 years of follow-up, compared with those with an eGFR greater than 90, as well as with a 55% increased risk of cancer mortality during 8.9 years of follow-up.
Colon cancer was the only common type of malignancy whose incidence was significantly increased in patients with chronic kidney disease, although Dr. Chen said she didn’t attach much significance to this finding, given the limited number of cancers that accrued.
In an interview, she observed that a post hoc analysis such as this can’t establish causality or the mechanisms involved. She speculated that the most likely explanation for the findings is that patients with impaired kidney function have incomplete removal of various toxins, some of which are oncogenic.
ALLHAT was funded by the National Heart, Lung, and Blood Institute. Dr. Chen reported having no financial conflicts of interest.
LAS VEGAS – Patients with chronic kidney disease appear to be at increased risk for cancer, a study showed.
A retrospective analysis of data on 31,896 participants in ALLHAT (the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial) showed that during a mean 4.9 years of in-trial follow-up, 2,529 subjects were diagnosed with various cancers.
The 5-year rate of incident cancer was 7.24 cases per 100 person-years among subjects with a baseline normal estimated glomerular filtration rate (eGFR) greater than 90 mL/min per 1.73 m2. The cancer rate rose with decreasing renal function: 8.38 cases per 100 person-years in patients with a baseline eGFR of 60-89.9, 9.18 per 100 person-years in those with an eGFR of 45-59.9, and 11.58 per 100 person-years in patients with an eGFR below 45 mL/min per 1.73 m2, Dr. Dhruti P. Chen reported at a meeting sponsored by the National Kidney Foundation.
An additional 4 years of follow-up for mortality due to cancer was available through national databases after ALLHAT ended. During the mean total 8.9 years of follow-up, there were 2,338 cancer-related deaths. The 10-year rate of cancer mortality was 7.90 cases per 100 person-years in patients with an eGFR greater than 90; 7.71 in those with an eGFR of 60-89.9; 10.11 per 100 person-years with an eGFR of 45-59.9; and 13.19 per 100 person-years in patients with an eGFR below 45 mL/min per 1.73 m2, added Dr. Chen of the department of medicine at Case Western Reserve University, Cleveland.
In a multivariate analysis adjusted for demographics, body mass index, diabetes, cardiovascular risk factors, blood glucose level, and the antihypertensive agent to which a patient was randomized, the association between chronic kidney disease and incident cancer was attenuated. Nonetheless, having a baseline eGFR below 45 mL/min per 1.73 m2 was independently associated with an adjusted 28% increased risk of incident cancer during 4.9 years of follow-up, compared with those with an eGFR greater than 90, as well as with a 55% increased risk of cancer mortality during 8.9 years of follow-up.
Colon cancer was the only common type of malignancy whose incidence was significantly increased in patients with chronic kidney disease, although Dr. Chen said she didn’t attach much significance to this finding, given the limited number of cancers that accrued.
In an interview, she observed that a post hoc analysis such as this can’t establish causality or the mechanisms involved. She speculated that the most likely explanation for the findings is that patients with impaired kidney function have incomplete removal of various toxins, some of which are oncogenic.
ALLHAT was funded by the National Heart, Lung, and Blood Institute. Dr. Chen reported having no financial conflicts of interest.
LAS VEGAS – Patients with chronic kidney disease appear to be at increased risk for cancer, a study showed.
A retrospective analysis of data on 31,896 participants in ALLHAT (the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial) showed that during a mean 4.9 years of in-trial follow-up, 2,529 subjects were diagnosed with various cancers.
The 5-year rate of incident cancer was 7.24 cases per 100 person-years among subjects with a baseline normal estimated glomerular filtration rate (eGFR) greater than 90 mL/min per 1.73 m2. The cancer rate rose with decreasing renal function: 8.38 cases per 100 person-years in patients with a baseline eGFR of 60-89.9, 9.18 per 100 person-years in those with an eGFR of 45-59.9, and 11.58 per 100 person-years in patients with an eGFR below 45 mL/min per 1.73 m2, Dr. Dhruti P. Chen reported at a meeting sponsored by the National Kidney Foundation.
An additional 4 years of follow-up for mortality due to cancer was available through national databases after ALLHAT ended. During the mean total 8.9 years of follow-up, there were 2,338 cancer-related deaths. The 10-year rate of cancer mortality was 7.90 cases per 100 person-years in patients with an eGFR greater than 90; 7.71 in those with an eGFR of 60-89.9; 10.11 per 100 person-years with an eGFR of 45-59.9; and 13.19 per 100 person-years in patients with an eGFR below 45 mL/min per 1.73 m2, added Dr. Chen of the department of medicine at Case Western Reserve University, Cleveland.
In a multivariate analysis adjusted for demographics, body mass index, diabetes, cardiovascular risk factors, blood glucose level, and the antihypertensive agent to which a patient was randomized, the association between chronic kidney disease and incident cancer was attenuated. Nonetheless, having a baseline eGFR below 45 mL/min per 1.73 m2 was independently associated with an adjusted 28% increased risk of incident cancer during 4.9 years of follow-up, compared with those with an eGFR greater than 90, as well as with a 55% increased risk of cancer mortality during 8.9 years of follow-up.
Colon cancer was the only common type of malignancy whose incidence was significantly increased in patients with chronic kidney disease, although Dr. Chen said she didn’t attach much significance to this finding, given the limited number of cancers that accrued.
In an interview, she observed that a post hoc analysis such as this can’t establish causality or the mechanisms involved. She speculated that the most likely explanation for the findings is that patients with impaired kidney function have incomplete removal of various toxins, some of which are oncogenic.
ALLHAT was funded by the National Heart, Lung, and Blood Institute. Dr. Chen reported having no financial conflicts of interest.
AT SCM 14
Major finding: Patients with chronic kidney disease had an adjusted 55% increased risk of cancer-related mortality during a mean 8.9 years of prospective follow-up, compared with those with normal kidney function.
Data source: This was a post hoc analysis of 31,896 participants in the prospective ALLHAT study.
Disclosures: ALLHAT was funded by the National Heart, Lung, and Blood Institute. The presenter reported having no financial conflicts of interest.
Avoid these seven risk factors and slash Alzheimer’s risk
ORLANDO – Up to half of all cases of Alzheimer’s disease are attributable to seven modifiable risk factors – including depression, Dr. Kristine Yaffe asserted in her 2014 AAGP Distinguished Scientist Award Lecture.
In addition to depression, the modifiable risk factors for Alzheimer's disease backed by the best supporting evidence from observational studies are diabetes, smoking, midlife obesity, midlife hypertension, low educational attainment, and physical inactivity.
A public health campaign successful in achieving a rather modest 10% reduction across the board in these seven risk factors could prevent an estimated 184,000 cases of Alzheimer's disease in the United States alone. A more ambitious 25% reduction in the levels of these seven risk factors could prevent close to half a million cases in the United States and 3 million worldwide, she predicted at the annual meeting of the American Association for Geriatric Psychiatry.
"We’re all told by the Alzheimer’s Association and others that there’s an epidemic in dementia. And it’s true: There is an epidemic in dementia, because we’re living longer and the baby boomers are aging. We’re seeing things we didn’t see before. There wasn’t so much neurodegenerative disease when people died in their 60s and 70s. Now that we’re living into our 80s and 90s, we’re going to see a lot more neurodegenerative disease. But there are also a lot of interesting secular trends that hold promise. I think we have reason to be cautiously optimistic," declared Dr. Yaffe, professor of psychiatry, neurology, and epidemiology at the University of California, San Francisco. She is also Roy and Marie Scola Endowed Chair in Psychiatry at UCSF, and is chief of geriatric psychiatry and director of the memory disorders clinic at the San Francisco Veterans Affairs Medical Center
Dr. Yaffe is credited with having played a pivotal role in establishing many of these risk factors, including, for example, depression (Arch. Gen. Psychiatry 2012;69:493-8), limited educational attainment, or literacy (J. Gerontol. A. Biol. Sci. Med. Sci. 2013 [doi:10.1093/gerona/glt176]), and cardiorespiratory fitness as a protective factor (Neurology 2014;82:1339-46) and J. Gerontol. A. Biol. Sci. Med. Sci. 2014 [doi:10.1093/gerona/glt287]).
She has also been a leader in identifying posttraumatic stress disorder and traumatic brain injury as risk factors, although those didn’t crack her list of key risks. Her interest in developing a preventive approach targeting modifiable risk factors amenable to a public health campaign has been fueled by recognition that Alzheimer’s disease is irreversible, slow to develop, and that existing drugs are of only limited symptomatic benefit and don’t halt disease progression.
She and coinvestigator Deborah E. Barnes, Ph.D., estimated the impact of reducing levels of the risk factors both in the United States and globally by first calculating the population-attributable risk; that is, the percentage of Alzheimer’s disease cases attributable to a given risk factor. They found that, globally, lack of education and smoking are the biggest contributors to risk. In the United States, physical inactivity contributed to the biggest proportion of cases (Lancet Neurol. 2011;10:819-28).
The current favored hypothesis as to how higher educational attainment, mental stimulation, physical exercise, and other factors protect against development of Alzheimer’s disease and other forms of dementia involves what’s called "cognitive reserve." Cognitive reserve is conceptualized as a sort of buffer in the brain that protects against neuropathologic accumulation. It’s what enables many seniors to have died with the normal cognitive function despite meeting neuropathologic criteria for Alzheimer’s disease at autopsy.
Dr. Yaffe draws hope from the well-documented marked decline in deaths from heart disease and stroke in recent decades, about half of which is attributable to treatment advances and the other half to public health measures aimed at risk factor reduction.
Evidence from recent studies suggests that a parallel decline in the incidence of dementia has been underway during the past couple of decades. For example, a population-based U.S. survey found the prevalence of cognitive impairment among people aged 70 years and older fell from 12.2% in 1993 to 8.7% in 2002 (Alzheimer's Dement. 2008;4:134-44). Dutch investigators reported that in the Rotterdam Study, the incidence rate ratio of dementia fell from 6.56 cases/1,000 person-years in 1990 to 4.92 cases/1,000 person-years in 2000 (Neurology 2012;78:1456-63). In three regions of England under study, the prevalence of dementia declined from 8.3% in 1989-1994 to 6.5% in 2008-2011 (Lancet 2013;382:1405-12). And, in Stockholm, there is evidence to suggest a decreasing incidence of dementia during a recent 20-year period (Neurology 2013;80:1888-94).
The authors of these studies attributed their findings to higher educational level, a decline in stroke incidence, and adoption of healthier lifestyles.
Working against these favorable trends, at least in the United States, are the epidemics of obesity and type 2 diabetes as well as widespread sedentary behavior.
"A lot of cardiovascular risk factors are shifting earlier now, so the exposures are in the teens and 20s as opposed to the 50s and 60s. And that’s bad," Dr. Yaffe observed.
Nonetheless, she reported that she sees opportunities. Earlier this year, Dr. Yaffe together with Dr. A. David Smith, professor emeritus of pharmacology, of the University of Oxford (England) penned an appeal to the governments of the G8 countries to make prevention of dementia a major health priority. The open letter, entitled "Dementia (Including Alzheimer’s Disease) Can Be Prevented," was signed by 109 experts from 36 countries.
"There is already sufficient evidence to justify immediate action," the authors declared (J. Alzheimer's Dis. 2014;38:699-703). "Tell people that adopting a healthy lifestyle may help ward off dementia as it does for other diseases."
Dr. Yaffe reported serving as a consultant to Novartis and Pfizer. She receives grant support from the National Institute on Aging, the Alzheimer’s Association, the American Health Assistance Foundation, the California Department of Public Health, and the U.S. Department of Defense.
ORLANDO – Up to half of all cases of Alzheimer’s disease are attributable to seven modifiable risk factors – including depression, Dr. Kristine Yaffe asserted in her 2014 AAGP Distinguished Scientist Award Lecture.
In addition to depression, the modifiable risk factors for Alzheimer's disease backed by the best supporting evidence from observational studies are diabetes, smoking, midlife obesity, midlife hypertension, low educational attainment, and physical inactivity.
A public health campaign successful in achieving a rather modest 10% reduction across the board in these seven risk factors could prevent an estimated 184,000 cases of Alzheimer's disease in the United States alone. A more ambitious 25% reduction in the levels of these seven risk factors could prevent close to half a million cases in the United States and 3 million worldwide, she predicted at the annual meeting of the American Association for Geriatric Psychiatry.
"We’re all told by the Alzheimer’s Association and others that there’s an epidemic in dementia. And it’s true: There is an epidemic in dementia, because we’re living longer and the baby boomers are aging. We’re seeing things we didn’t see before. There wasn’t so much neurodegenerative disease when people died in their 60s and 70s. Now that we’re living into our 80s and 90s, we’re going to see a lot more neurodegenerative disease. But there are also a lot of interesting secular trends that hold promise. I think we have reason to be cautiously optimistic," declared Dr. Yaffe, professor of psychiatry, neurology, and epidemiology at the University of California, San Francisco. She is also Roy and Marie Scola Endowed Chair in Psychiatry at UCSF, and is chief of geriatric psychiatry and director of the memory disorders clinic at the San Francisco Veterans Affairs Medical Center
Dr. Yaffe is credited with having played a pivotal role in establishing many of these risk factors, including, for example, depression (Arch. Gen. Psychiatry 2012;69:493-8), limited educational attainment, or literacy (J. Gerontol. A. Biol. Sci. Med. Sci. 2013 [doi:10.1093/gerona/glt176]), and cardiorespiratory fitness as a protective factor (Neurology 2014;82:1339-46) and J. Gerontol. A. Biol. Sci. Med. Sci. 2014 [doi:10.1093/gerona/glt287]).
She has also been a leader in identifying posttraumatic stress disorder and traumatic brain injury as risk factors, although those didn’t crack her list of key risks. Her interest in developing a preventive approach targeting modifiable risk factors amenable to a public health campaign has been fueled by recognition that Alzheimer’s disease is irreversible, slow to develop, and that existing drugs are of only limited symptomatic benefit and don’t halt disease progression.
She and coinvestigator Deborah E. Barnes, Ph.D., estimated the impact of reducing levels of the risk factors both in the United States and globally by first calculating the population-attributable risk; that is, the percentage of Alzheimer’s disease cases attributable to a given risk factor. They found that, globally, lack of education and smoking are the biggest contributors to risk. In the United States, physical inactivity contributed to the biggest proportion of cases (Lancet Neurol. 2011;10:819-28).
The current favored hypothesis as to how higher educational attainment, mental stimulation, physical exercise, and other factors protect against development of Alzheimer’s disease and other forms of dementia involves what’s called "cognitive reserve." Cognitive reserve is conceptualized as a sort of buffer in the brain that protects against neuropathologic accumulation. It’s what enables many seniors to have died with the normal cognitive function despite meeting neuropathologic criteria for Alzheimer’s disease at autopsy.
Dr. Yaffe draws hope from the well-documented marked decline in deaths from heart disease and stroke in recent decades, about half of which is attributable to treatment advances and the other half to public health measures aimed at risk factor reduction.
Evidence from recent studies suggests that a parallel decline in the incidence of dementia has been underway during the past couple of decades. For example, a population-based U.S. survey found the prevalence of cognitive impairment among people aged 70 years and older fell from 12.2% in 1993 to 8.7% in 2002 (Alzheimer's Dement. 2008;4:134-44). Dutch investigators reported that in the Rotterdam Study, the incidence rate ratio of dementia fell from 6.56 cases/1,000 person-years in 1990 to 4.92 cases/1,000 person-years in 2000 (Neurology 2012;78:1456-63). In three regions of England under study, the prevalence of dementia declined from 8.3% in 1989-1994 to 6.5% in 2008-2011 (Lancet 2013;382:1405-12). And, in Stockholm, there is evidence to suggest a decreasing incidence of dementia during a recent 20-year period (Neurology 2013;80:1888-94).
The authors of these studies attributed their findings to higher educational level, a decline in stroke incidence, and adoption of healthier lifestyles.
Working against these favorable trends, at least in the United States, are the epidemics of obesity and type 2 diabetes as well as widespread sedentary behavior.
"A lot of cardiovascular risk factors are shifting earlier now, so the exposures are in the teens and 20s as opposed to the 50s and 60s. And that’s bad," Dr. Yaffe observed.
Nonetheless, she reported that she sees opportunities. Earlier this year, Dr. Yaffe together with Dr. A. David Smith, professor emeritus of pharmacology, of the University of Oxford (England) penned an appeal to the governments of the G8 countries to make prevention of dementia a major health priority. The open letter, entitled "Dementia (Including Alzheimer’s Disease) Can Be Prevented," was signed by 109 experts from 36 countries.
"There is already sufficient evidence to justify immediate action," the authors declared (J. Alzheimer's Dis. 2014;38:699-703). "Tell people that adopting a healthy lifestyle may help ward off dementia as it does for other diseases."
Dr. Yaffe reported serving as a consultant to Novartis and Pfizer. She receives grant support from the National Institute on Aging, the Alzheimer’s Association, the American Health Assistance Foundation, the California Department of Public Health, and the U.S. Department of Defense.
ORLANDO – Up to half of all cases of Alzheimer’s disease are attributable to seven modifiable risk factors – including depression, Dr. Kristine Yaffe asserted in her 2014 AAGP Distinguished Scientist Award Lecture.
In addition to depression, the modifiable risk factors for Alzheimer's disease backed by the best supporting evidence from observational studies are diabetes, smoking, midlife obesity, midlife hypertension, low educational attainment, and physical inactivity.
A public health campaign successful in achieving a rather modest 10% reduction across the board in these seven risk factors could prevent an estimated 184,000 cases of Alzheimer's disease in the United States alone. A more ambitious 25% reduction in the levels of these seven risk factors could prevent close to half a million cases in the United States and 3 million worldwide, she predicted at the annual meeting of the American Association for Geriatric Psychiatry.
"We’re all told by the Alzheimer’s Association and others that there’s an epidemic in dementia. And it’s true: There is an epidemic in dementia, because we’re living longer and the baby boomers are aging. We’re seeing things we didn’t see before. There wasn’t so much neurodegenerative disease when people died in their 60s and 70s. Now that we’re living into our 80s and 90s, we’re going to see a lot more neurodegenerative disease. But there are also a lot of interesting secular trends that hold promise. I think we have reason to be cautiously optimistic," declared Dr. Yaffe, professor of psychiatry, neurology, and epidemiology at the University of California, San Francisco. She is also Roy and Marie Scola Endowed Chair in Psychiatry at UCSF, and is chief of geriatric psychiatry and director of the memory disorders clinic at the San Francisco Veterans Affairs Medical Center
Dr. Yaffe is credited with having played a pivotal role in establishing many of these risk factors, including, for example, depression (Arch. Gen. Psychiatry 2012;69:493-8), limited educational attainment, or literacy (J. Gerontol. A. Biol. Sci. Med. Sci. 2013 [doi:10.1093/gerona/glt176]), and cardiorespiratory fitness as a protective factor (Neurology 2014;82:1339-46) and J. Gerontol. A. Biol. Sci. Med. Sci. 2014 [doi:10.1093/gerona/glt287]).
She has also been a leader in identifying posttraumatic stress disorder and traumatic brain injury as risk factors, although those didn’t crack her list of key risks. Her interest in developing a preventive approach targeting modifiable risk factors amenable to a public health campaign has been fueled by recognition that Alzheimer’s disease is irreversible, slow to develop, and that existing drugs are of only limited symptomatic benefit and don’t halt disease progression.
She and coinvestigator Deborah E. Barnes, Ph.D., estimated the impact of reducing levels of the risk factors both in the United States and globally by first calculating the population-attributable risk; that is, the percentage of Alzheimer’s disease cases attributable to a given risk factor. They found that, globally, lack of education and smoking are the biggest contributors to risk. In the United States, physical inactivity contributed to the biggest proportion of cases (Lancet Neurol. 2011;10:819-28).
The current favored hypothesis as to how higher educational attainment, mental stimulation, physical exercise, and other factors protect against development of Alzheimer’s disease and other forms of dementia involves what’s called "cognitive reserve." Cognitive reserve is conceptualized as a sort of buffer in the brain that protects against neuropathologic accumulation. It’s what enables many seniors to have died with the normal cognitive function despite meeting neuropathologic criteria for Alzheimer’s disease at autopsy.
Dr. Yaffe draws hope from the well-documented marked decline in deaths from heart disease and stroke in recent decades, about half of which is attributable to treatment advances and the other half to public health measures aimed at risk factor reduction.
Evidence from recent studies suggests that a parallel decline in the incidence of dementia has been underway during the past couple of decades. For example, a population-based U.S. survey found the prevalence of cognitive impairment among people aged 70 years and older fell from 12.2% in 1993 to 8.7% in 2002 (Alzheimer's Dement. 2008;4:134-44). Dutch investigators reported that in the Rotterdam Study, the incidence rate ratio of dementia fell from 6.56 cases/1,000 person-years in 1990 to 4.92 cases/1,000 person-years in 2000 (Neurology 2012;78:1456-63). In three regions of England under study, the prevalence of dementia declined from 8.3% in 1989-1994 to 6.5% in 2008-2011 (Lancet 2013;382:1405-12). And, in Stockholm, there is evidence to suggest a decreasing incidence of dementia during a recent 20-year period (Neurology 2013;80:1888-94).
The authors of these studies attributed their findings to higher educational level, a decline in stroke incidence, and adoption of healthier lifestyles.
Working against these favorable trends, at least in the United States, are the epidemics of obesity and type 2 diabetes as well as widespread sedentary behavior.
"A lot of cardiovascular risk factors are shifting earlier now, so the exposures are in the teens and 20s as opposed to the 50s and 60s. And that’s bad," Dr. Yaffe observed.
Nonetheless, she reported that she sees opportunities. Earlier this year, Dr. Yaffe together with Dr. A. David Smith, professor emeritus of pharmacology, of the University of Oxford (England) penned an appeal to the governments of the G8 countries to make prevention of dementia a major health priority. The open letter, entitled "Dementia (Including Alzheimer’s Disease) Can Be Prevented," was signed by 109 experts from 36 countries.
"There is already sufficient evidence to justify immediate action," the authors declared (J. Alzheimer's Dis. 2014;38:699-703). "Tell people that adopting a healthy lifestyle may help ward off dementia as it does for other diseases."
Dr. Yaffe reported serving as a consultant to Novartis and Pfizer. She receives grant support from the National Institute on Aging, the Alzheimer’s Association, the American Health Assistance Foundation, the California Department of Public Health, and the U.S. Department of Defense.
EXPERT ANALYSIS FROM THE AAGP ANNUAL MEETING
Geriatric depression, pain respond to various drugs
ORLANDO – Duloxetine for the treatment of late-life depression missed its primary end point in a recent large randomized trial, but it did result in consistently significant clinical improvement, compared with placebo across numerous secondary outcome measures.
The results overall provide a suggestion of antidepressant efficacy along with evidence of a meaningful reduction in pain in this elderly patient population, Dr. David C. Steffens observed at the annual meeting of the American Association for Geriatric Psychiatry.
This was a multicenter, double-blind, placebo-controlled 24-week study. It included 296 patients age 65 or older with major depressive disorder, a Montgomery-Asberg Depression Rating Scale score of 20 or above indicative of at least moderately severe depression, and a Mini-Mental Status Examination score of 20 or more. Participants were randomized 2-to-1 to duloxetine (Cymbalta) at either 60 mg/day, 120 mg/day, or placebo.
The primary end point was change in the Maier subscale of the 17-item Hamilton Depression Rating Scale (HAMD-17) at week 12, compared with placebo. The difference fell short of statistical significance at week 12, which was surprising given that the difference was significant at weeks 4, 8, 16, and 20. Moreover, significantly greater improvement in depressive symptoms as measured by the Geriatric Depression Scale was seen in the duloxetine group at weeks 2, 4, 8, 16, 20, and 24 – but once again, not at week 12, which was the point when the study allowed for dose adjustment from 60 mg/day to 120 mg/day, noted Dr. Steffens, professor and chairman of the department of psychiatry at the University of Connecticut, Farmington.
Participants had baseline mild to moderate pain as reflected in their scores on the Brief Pain Inventory and Pain Numeric Rating Scale. The duloxetine-treated group showed significant improvement in pain severity and interference, compared with controls. That’s an important finding given that close to two-thirds of patients with geriatric depression have comorbid pain that typically compounds their mood disorder.
Prior trials of duloxetine have been shorter. In this one, the remission rate, as defined by a HAMD-17 score of 7 or less among patients on duloxetine at 60 mg/day for the full 24 weeks, was 66% by week 12 and 65% at week 24. Thus, if a patient wasn’t in remission after 12 weeks at 60 mg/day, it was unlikely to happen with another 12 weeks on that regimen, and a dose increase to 120 mg/day may be warranted.
Five patients discontinued duloxetine for serious adverse events, compared with one placebo-treated control. The most prominent duloxetine-related side effects were diarrhea and dizziness. Duloxetine-treated patients showed a small increase in heart rate and a modest decrease in blood pressure, issues that need to be borne in mind in an elderly population at increased cardiovascular risk. One patient on duloxetine fell and sustained a hip fracture (Am. J. Geriatr. Psychiatry 2014;22:34-45).
Dr. Steffens, who didn’t participate in the duloxetine trial, highlighted another encouraging development in the treatment of late-life depression: a pooled subgroup analysis he and other investigators conducted using the data from three similar 14-week, prospective, randomized, double-blind trials of adjuvant aripiprazole (Abilify), compared with second-line antidepressant therapy in patients with major depressive disorder who had an incomplete response to standard antidepressant therapy. Their analysis included 679 patients under 50 years of age and 409 others aged 50-67 years. The older group was further subdivided by age: 50-55, 56-60, and 61-67 years.
"Our goal was to generate some preliminary data. Industry has been less and less interested in thinking about atypical antipsychotics in the elderly because of the black box warnings. Our reasoning was any data that we can get that might support further study in this population of older adults makes sense," the psychiatrist explained.
Older patients on aripiprazole showed significantly greater improvement on the Montgomery-Asberg Depression Rating Scale at week 14 than placebo-treated controls. Their remission rate was 33%, compared with 27% in younger aripiprazole-treated patients (Int. J. Geriatr. Psychiatry 2011;26:564-72).
These data are "as good as we have in terms of this particular research issue. We showed people are not keeling over from this therapy. We can at least say that among the few elderly patients we have here, we have preliminary data that this may be helpful and can be tolerated. There are some signals here. Subsequent studies looking at the older population are needed," according to Dr. Steffens.
In response to an audience question, Dr. Steffens said that in the 10 or 12 patients with late-life depression where he has tried using aripiprazole off-label at a starting dose of 1 or 2 mg/day he has had a fair degree of success.
Dr. Steffens reported having no financial conflicts.
ORLANDO – Duloxetine for the treatment of late-life depression missed its primary end point in a recent large randomized trial, but it did result in consistently significant clinical improvement, compared with placebo across numerous secondary outcome measures.
The results overall provide a suggestion of antidepressant efficacy along with evidence of a meaningful reduction in pain in this elderly patient population, Dr. David C. Steffens observed at the annual meeting of the American Association for Geriatric Psychiatry.
This was a multicenter, double-blind, placebo-controlled 24-week study. It included 296 patients age 65 or older with major depressive disorder, a Montgomery-Asberg Depression Rating Scale score of 20 or above indicative of at least moderately severe depression, and a Mini-Mental Status Examination score of 20 or more. Participants were randomized 2-to-1 to duloxetine (Cymbalta) at either 60 mg/day, 120 mg/day, or placebo.
The primary end point was change in the Maier subscale of the 17-item Hamilton Depression Rating Scale (HAMD-17) at week 12, compared with placebo. The difference fell short of statistical significance at week 12, which was surprising given that the difference was significant at weeks 4, 8, 16, and 20. Moreover, significantly greater improvement in depressive symptoms as measured by the Geriatric Depression Scale was seen in the duloxetine group at weeks 2, 4, 8, 16, 20, and 24 – but once again, not at week 12, which was the point when the study allowed for dose adjustment from 60 mg/day to 120 mg/day, noted Dr. Steffens, professor and chairman of the department of psychiatry at the University of Connecticut, Farmington.
Participants had baseline mild to moderate pain as reflected in their scores on the Brief Pain Inventory and Pain Numeric Rating Scale. The duloxetine-treated group showed significant improvement in pain severity and interference, compared with controls. That’s an important finding given that close to two-thirds of patients with geriatric depression have comorbid pain that typically compounds their mood disorder.
Prior trials of duloxetine have been shorter. In this one, the remission rate, as defined by a HAMD-17 score of 7 or less among patients on duloxetine at 60 mg/day for the full 24 weeks, was 66% by week 12 and 65% at week 24. Thus, if a patient wasn’t in remission after 12 weeks at 60 mg/day, it was unlikely to happen with another 12 weeks on that regimen, and a dose increase to 120 mg/day may be warranted.
Five patients discontinued duloxetine for serious adverse events, compared with one placebo-treated control. The most prominent duloxetine-related side effects were diarrhea and dizziness. Duloxetine-treated patients showed a small increase in heart rate and a modest decrease in blood pressure, issues that need to be borne in mind in an elderly population at increased cardiovascular risk. One patient on duloxetine fell and sustained a hip fracture (Am. J. Geriatr. Psychiatry 2014;22:34-45).
Dr. Steffens, who didn’t participate in the duloxetine trial, highlighted another encouraging development in the treatment of late-life depression: a pooled subgroup analysis he and other investigators conducted using the data from three similar 14-week, prospective, randomized, double-blind trials of adjuvant aripiprazole (Abilify), compared with second-line antidepressant therapy in patients with major depressive disorder who had an incomplete response to standard antidepressant therapy. Their analysis included 679 patients under 50 years of age and 409 others aged 50-67 years. The older group was further subdivided by age: 50-55, 56-60, and 61-67 years.
"Our goal was to generate some preliminary data. Industry has been less and less interested in thinking about atypical antipsychotics in the elderly because of the black box warnings. Our reasoning was any data that we can get that might support further study in this population of older adults makes sense," the psychiatrist explained.
Older patients on aripiprazole showed significantly greater improvement on the Montgomery-Asberg Depression Rating Scale at week 14 than placebo-treated controls. Their remission rate was 33%, compared with 27% in younger aripiprazole-treated patients (Int. J. Geriatr. Psychiatry 2011;26:564-72).
These data are "as good as we have in terms of this particular research issue. We showed people are not keeling over from this therapy. We can at least say that among the few elderly patients we have here, we have preliminary data that this may be helpful and can be tolerated. There are some signals here. Subsequent studies looking at the older population are needed," according to Dr. Steffens.
In response to an audience question, Dr. Steffens said that in the 10 or 12 patients with late-life depression where he has tried using aripiprazole off-label at a starting dose of 1 or 2 mg/day he has had a fair degree of success.
Dr. Steffens reported having no financial conflicts.
ORLANDO – Duloxetine for the treatment of late-life depression missed its primary end point in a recent large randomized trial, but it did result in consistently significant clinical improvement, compared with placebo across numerous secondary outcome measures.
The results overall provide a suggestion of antidepressant efficacy along with evidence of a meaningful reduction in pain in this elderly patient population, Dr. David C. Steffens observed at the annual meeting of the American Association for Geriatric Psychiatry.
This was a multicenter, double-blind, placebo-controlled 24-week study. It included 296 patients age 65 or older with major depressive disorder, a Montgomery-Asberg Depression Rating Scale score of 20 or above indicative of at least moderately severe depression, and a Mini-Mental Status Examination score of 20 or more. Participants were randomized 2-to-1 to duloxetine (Cymbalta) at either 60 mg/day, 120 mg/day, or placebo.
The primary end point was change in the Maier subscale of the 17-item Hamilton Depression Rating Scale (HAMD-17) at week 12, compared with placebo. The difference fell short of statistical significance at week 12, which was surprising given that the difference was significant at weeks 4, 8, 16, and 20. Moreover, significantly greater improvement in depressive symptoms as measured by the Geriatric Depression Scale was seen in the duloxetine group at weeks 2, 4, 8, 16, 20, and 24 – but once again, not at week 12, which was the point when the study allowed for dose adjustment from 60 mg/day to 120 mg/day, noted Dr. Steffens, professor and chairman of the department of psychiatry at the University of Connecticut, Farmington.
Participants had baseline mild to moderate pain as reflected in their scores on the Brief Pain Inventory and Pain Numeric Rating Scale. The duloxetine-treated group showed significant improvement in pain severity and interference, compared with controls. That’s an important finding given that close to two-thirds of patients with geriatric depression have comorbid pain that typically compounds their mood disorder.
Prior trials of duloxetine have been shorter. In this one, the remission rate, as defined by a HAMD-17 score of 7 or less among patients on duloxetine at 60 mg/day for the full 24 weeks, was 66% by week 12 and 65% at week 24. Thus, if a patient wasn’t in remission after 12 weeks at 60 mg/day, it was unlikely to happen with another 12 weeks on that regimen, and a dose increase to 120 mg/day may be warranted.
Five patients discontinued duloxetine for serious adverse events, compared with one placebo-treated control. The most prominent duloxetine-related side effects were diarrhea and dizziness. Duloxetine-treated patients showed a small increase in heart rate and a modest decrease in blood pressure, issues that need to be borne in mind in an elderly population at increased cardiovascular risk. One patient on duloxetine fell and sustained a hip fracture (Am. J. Geriatr. Psychiatry 2014;22:34-45).
Dr. Steffens, who didn’t participate in the duloxetine trial, highlighted another encouraging development in the treatment of late-life depression: a pooled subgroup analysis he and other investigators conducted using the data from three similar 14-week, prospective, randomized, double-blind trials of adjuvant aripiprazole (Abilify), compared with second-line antidepressant therapy in patients with major depressive disorder who had an incomplete response to standard antidepressant therapy. Their analysis included 679 patients under 50 years of age and 409 others aged 50-67 years. The older group was further subdivided by age: 50-55, 56-60, and 61-67 years.
"Our goal was to generate some preliminary data. Industry has been less and less interested in thinking about atypical antipsychotics in the elderly because of the black box warnings. Our reasoning was any data that we can get that might support further study in this population of older adults makes sense," the psychiatrist explained.
Older patients on aripiprazole showed significantly greater improvement on the Montgomery-Asberg Depression Rating Scale at week 14 than placebo-treated controls. Their remission rate was 33%, compared with 27% in younger aripiprazole-treated patients (Int. J. Geriatr. Psychiatry 2011;26:564-72).
These data are "as good as we have in terms of this particular research issue. We showed people are not keeling over from this therapy. We can at least say that among the few elderly patients we have here, we have preliminary data that this may be helpful and can be tolerated. There are some signals here. Subsequent studies looking at the older population are needed," according to Dr. Steffens.
In response to an audience question, Dr. Steffens said that in the 10 or 12 patients with late-life depression where he has tried using aripiprazole off-label at a starting dose of 1 or 2 mg/day he has had a fair degree of success.
Dr. Steffens reported having no financial conflicts.
EXPERT ANALYSIS FROM THE AAGP ANNUAL MEETING