User login
Steroid-sparing therapy is promising for allergic asthma
KEYSTONE, COLO. – A novel toll-like receptor-9 agonist showed impressive clinical efficacy in persistent allergic asthma in a double-blind, placebo-controlled clinical trial.
"This is really interesting," Dr. Harold S. Nelson commented in highlighting the European study as one of the most promising new developments in immunology during the past year in his talk at a meeting on allergy and respiratory diseases sponsored by National Jewish Health.
The toll-like receptor-9 agonist under study is known as QbG10. It consists of a protein shell derived from the Q-beta bacteriophage which is wrapped around a core of DNA oligomer G10 rich in A-type CpG motifs. The protein shell protects the G10 CpG so that when the antigen-presenting cell digests that outer coat, the CpG sits like a Trojan horse inside the T cell or macrophage. The G10 CpG induces interferon-alpha production, which in turn causes degradation of transcription factor GATA-3, explained Dr. Nelson, professor of medicine and of pediatrics at the University of Colorado, Denver, and National Jewish Health.
Allergic asthma is a disease in which allergen-specific Th2 responses figure prominently. QbG10 therapy is designed to nudge the immune system toward a Th1-mediated protective response.
What’s particularly intriguing about this novel immunotherapy, he added, is that the clinical efficacy is achieved without administering any allergen. The results are achieved simply by showing the CpG to asthma patients. CpGs are DNA oligonucleotides containing unmethylated deoxycytidylyl-deoxyguanosine dinucleotides.
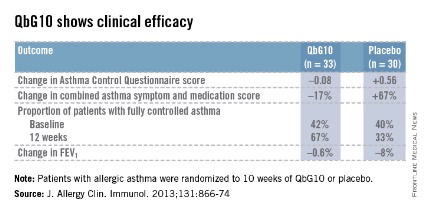
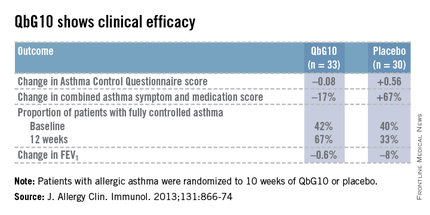
The European proof-of-concept study included 63 patients with allergic asthma being treated with moderate- or high-dose inhaled corticosteroids. They were randomized to receive seven injections of QbG10 or placebo over the course of 10 weeks, during which a controlled steroid withdrawal was carried out. After 4 weeks, the patients’ inhaled steroid dose was cut by 50%. After 8 weeks, the inhaled steroid was discontinued altogether. At evaluation at 12 weeks – 4 weeks after inhaled steroids were halted – two-thirds of QbG10-treated patients had well-controlled asthma as defined by an Asthma Control Questionnaire score of 0.75 or less, compared with one-third of controls. The QbG10 group showed similar advantages on other measures of asthma control, in spite of the steroid taper (see chart).
Local injection site reactions were common in the QbG10 group. The reactions were generally mild to moderate and were most pronounced 1-2 days post injection. The biggest reactions were noted after the third of the seven injections. No systemic reactions occurred (J. Allergy Clin. Immunol. 2013;131:866-74).
Dr. Nelson reported having no financial conflicts of interest regarding this work.
KEYSTONE, COLO. – A novel toll-like receptor-9 agonist showed impressive clinical efficacy in persistent allergic asthma in a double-blind, placebo-controlled clinical trial.
"This is really interesting," Dr. Harold S. Nelson commented in highlighting the European study as one of the most promising new developments in immunology during the past year in his talk at a meeting on allergy and respiratory diseases sponsored by National Jewish Health.

The toll-like receptor-9 agonist under study is known as QbG10. It consists of a protein shell derived from the Q-beta bacteriophage which is wrapped around a core of DNA oligomer G10 rich in A-type CpG motifs. The protein shell protects the G10 CpG so that when the antigen-presenting cell digests that outer coat, the CpG sits like a Trojan horse inside the T cell or macrophage. The G10 CpG induces interferon-alpha production, which in turn causes degradation of transcription factor GATA-3, explained Dr. Nelson, professor of medicine and of pediatrics at the University of Colorado, Denver, and National Jewish Health.
Allergic asthma is a disease in which allergen-specific Th2 responses figure prominently. QbG10 therapy is designed to nudge the immune system toward a Th1-mediated protective response.
What’s particularly intriguing about this novel immunotherapy, he added, is that the clinical efficacy is achieved without administering any allergen. The results are achieved simply by showing the CpG to asthma patients. CpGs are DNA oligonucleotides containing unmethylated deoxycytidylyl-deoxyguanosine dinucleotides.
The European proof-of-concept study included 63 patients with allergic asthma being treated with moderate- or high-dose inhaled corticosteroids. They were randomized to receive seven injections of QbG10 or placebo over the course of 10 weeks, during which a controlled steroid withdrawal was carried out. After 4 weeks, the patients’ inhaled steroid dose was cut by 50%. After 8 weeks, the inhaled steroid was discontinued altogether. At evaluation at 12 weeks – 4 weeks after inhaled steroids were halted – two-thirds of QbG10-treated patients had well-controlled asthma as defined by an Asthma Control Questionnaire score of 0.75 or less, compared with one-third of controls. The QbG10 group showed similar advantages on other measures of asthma control, in spite of the steroid taper (see chart).
Local injection site reactions were common in the QbG10 group. The reactions were generally mild to moderate and were most pronounced 1-2 days post injection. The biggest reactions were noted after the third of the seven injections. No systemic reactions occurred (J. Allergy Clin. Immunol. 2013;131:866-74).
Dr. Nelson reported having no financial conflicts of interest regarding this work.
KEYSTONE, COLO. – A novel toll-like receptor-9 agonist showed impressive clinical efficacy in persistent allergic asthma in a double-blind, placebo-controlled clinical trial.
"This is really interesting," Dr. Harold S. Nelson commented in highlighting the European study as one of the most promising new developments in immunology during the past year in his talk at a meeting on allergy and respiratory diseases sponsored by National Jewish Health.

The toll-like receptor-9 agonist under study is known as QbG10. It consists of a protein shell derived from the Q-beta bacteriophage which is wrapped around a core of DNA oligomer G10 rich in A-type CpG motifs. The protein shell protects the G10 CpG so that when the antigen-presenting cell digests that outer coat, the CpG sits like a Trojan horse inside the T cell or macrophage. The G10 CpG induces interferon-alpha production, which in turn causes degradation of transcription factor GATA-3, explained Dr. Nelson, professor of medicine and of pediatrics at the University of Colorado, Denver, and National Jewish Health.
Allergic asthma is a disease in which allergen-specific Th2 responses figure prominently. QbG10 therapy is designed to nudge the immune system toward a Th1-mediated protective response.
What’s particularly intriguing about this novel immunotherapy, he added, is that the clinical efficacy is achieved without administering any allergen. The results are achieved simply by showing the CpG to asthma patients. CpGs are DNA oligonucleotides containing unmethylated deoxycytidylyl-deoxyguanosine dinucleotides.
The European proof-of-concept study included 63 patients with allergic asthma being treated with moderate- or high-dose inhaled corticosteroids. They were randomized to receive seven injections of QbG10 or placebo over the course of 10 weeks, during which a controlled steroid withdrawal was carried out. After 4 weeks, the patients’ inhaled steroid dose was cut by 50%. After 8 weeks, the inhaled steroid was discontinued altogether. At evaluation at 12 weeks – 4 weeks after inhaled steroids were halted – two-thirds of QbG10-treated patients had well-controlled asthma as defined by an Asthma Control Questionnaire score of 0.75 or less, compared with one-third of controls. The QbG10 group showed similar advantages on other measures of asthma control, in spite of the steroid taper (see chart).
Local injection site reactions were common in the QbG10 group. The reactions were generally mild to moderate and were most pronounced 1-2 days post injection. The biggest reactions were noted after the third of the seven injections. No systemic reactions occurred (J. Allergy Clin. Immunol. 2013;131:866-74).
Dr. Nelson reported having no financial conflicts of interest regarding this work.
EXPERT ANALYSIS FROM THE PULMONARY AND ALLERGY UPDATE
Apixaban for VTE reduced subsequent hospitalizations
WASHINGTON – Treating acute venous thromboembolism with the fixed-dose oral factor Xa inhibitor apixaban significantly reduces subsequent all-cause hospitalizations, compared with conventional therapy with enoxaparin followed by warfarin, according to a secondary analysis of the landmark AMPLIFY trial.
The 21% reduction in the risk of hospitalization in the apixaban group during the 6 months following the initial VTE was driven mainly by fewer hospitalizations for recurrent VTE or major bleeding. There were also significantly fewer physician office visits by patients on apixaban than for those on enoxaparin/warfarin, Dr. Margot Johnson reported at the annual meeting of the American College of Cardiology.
An analysis of the cost savings associated with this reduction in hospitalizations seen in AMPLIFY is underway and will be reported later this year, added Dr. Johnson of King’s College Hospital in London.
AMPLIFY (Apixaban for the Initial Management of Pulmonary Embolism and Deep Vein Thrombosis) was an industry-sponsored randomized double-blind study of 5,365 patients with acute symptomatic VTE who were assigned to 6 months of treatment with apixaban (Eliquis) at 10 mg b.i.d. for 7 days followed by 5 mg b.i.d. or to enoxaparin followed by warfarin. In the previously reported primary outcomes (N. Engl. J. Med. 2013;369:799-808, apixaban showed noninferiority to the conventional regimen in terms of the rate of recurrent VTE or VTE-related death, and a highly significant superiority in terms of major bleeding, with a 69% risk reduction.
Dr. Johnson reported that during the 6-month study period, 5.72% of the apixaban group had one or more hospitalizations after the initial event, compared with 7.07% of the control group. This translated to a highly significant 21% relative risk reduction. For every 74 patients treated with apixaban instead of enoxaparin/warfarin, one hospitalization was avoided. Moreover, when a hospitalization occurred in apixaban-treated patients, the mean length of stay was shorter: 10.2 vs. 11.7 days in the enoxaparin-warfarin group.
The median time to a first hospitalization was 63 days in the apixaban group, compared with 34.5 days in controls. The apixaban group’s advantage in terms of hospitalization risk was consistent across subgroups on the basis of age, body weight, sex, and renal function.
The number of emergency department visits during the 6-month follow-up period was similar in the two study arms. However, only 5.8% of the apixaban group visited a physician’s office, compared with 7.3% of controls. The reasons for these office visits were basically the same as for the hospitalizations: mostly recurrent VTEs and bleeding episodes. In the apixaban group, 35 patients had an office visit for recurrent VTE, compared with 61 controls. And 71 apixaban-treated patients made an office visit for bleeding episodes, compared with 130 controls.
Session cochair Dr. Emile R. Mohler commented on the finding that 37 patients in the apixaban group and 48 on enoxaparin/warfarin required hospitalization for recurrent VTE.
"It seems like we’re not doing a good enough job there. Either both of these anticoagulants don’t work well, or the patients aren’t taking the medication, or we’re not following up with them enough. I can’t remember the last time in my own clinical practice that somebody who took their medication came back within 6 months of having a VTE. It seems strange. I think there’s a lot of room for improvement," commented Dr. Mohler, professor of medicine and director of vascular medicine at the University of Pennsylvania, Philadelphia.
Dr. Johnson agreed about the room for improvement. But she added that, although the data she presented were based upon an intention-to-treat analysis, the results were the same – significantly fewer hospitalizations in the apixaban group – in a per-protocol analysis that excluded patients with less than 80% adherence to their study medication.
"One of the big things we saw in AMPLIFY is that whenever you stop an anticoagulant, you see the recurrence rate go up by about 10% per year. So it’s very important that these people continue their anticoagulation – and the more we can reduce their bleeding events, the more likely they are to comply with that therapy and be protected from having another VTE," she added.
Session cochair Dr. John P. Cooke commented that one underutilized aspect of treatment for acute VTE is compressive support, which he said has been given short shrift in the major practice guidelines.
Often as physicians, we give patients a pill and we think that we’ve treated them. Compressive support is important in VTE," stressed Dr. Cooke, chair of the department of cardiovascular sciences at the Houston Methodist Research Institute and director of the Center for Cardiovascular Regeneration at the Houston Methodist DeBakey Heart and Vascular Center.
The AMPLIFY trial was sponsored by Bristol-Myers Squibb and Pfizer. Dr. Johnson reported having no financial conflicts.
Dr. Vera DePalo, FCCP, comments: This large, industry-sponsored trial provides an alternative to the standard treatments for VTE. Patients had a less likelihood of hospitalization for recurrence or major bleeding. If hospitalization did occur, there was a much longer median time to hospitalization and a shorter hospital length of stay.
Dr. Vera DePalo, FCCP, comments: This large, industry-sponsored trial provides an alternative to the standard treatments for VTE. Patients had a less likelihood of hospitalization for recurrence or major bleeding. If hospitalization did occur, there was a much longer median time to hospitalization and a shorter hospital length of stay.
Dr. Vera DePalo, FCCP, comments: This large, industry-sponsored trial provides an alternative to the standard treatments for VTE. Patients had a less likelihood of hospitalization for recurrence or major bleeding. If hospitalization did occur, there was a much longer median time to hospitalization and a shorter hospital length of stay.
WASHINGTON – Treating acute venous thromboembolism with the fixed-dose oral factor Xa inhibitor apixaban significantly reduces subsequent all-cause hospitalizations, compared with conventional therapy with enoxaparin followed by warfarin, according to a secondary analysis of the landmark AMPLIFY trial.
The 21% reduction in the risk of hospitalization in the apixaban group during the 6 months following the initial VTE was driven mainly by fewer hospitalizations for recurrent VTE or major bleeding. There were also significantly fewer physician office visits by patients on apixaban than for those on enoxaparin/warfarin, Dr. Margot Johnson reported at the annual meeting of the American College of Cardiology.
An analysis of the cost savings associated with this reduction in hospitalizations seen in AMPLIFY is underway and will be reported later this year, added Dr. Johnson of King’s College Hospital in London.
AMPLIFY (Apixaban for the Initial Management of Pulmonary Embolism and Deep Vein Thrombosis) was an industry-sponsored randomized double-blind study of 5,365 patients with acute symptomatic VTE who were assigned to 6 months of treatment with apixaban (Eliquis) at 10 mg b.i.d. for 7 days followed by 5 mg b.i.d. or to enoxaparin followed by warfarin. In the previously reported primary outcomes (N. Engl. J. Med. 2013;369:799-808, apixaban showed noninferiority to the conventional regimen in terms of the rate of recurrent VTE or VTE-related death, and a highly significant superiority in terms of major bleeding, with a 69% risk reduction.
Dr. Johnson reported that during the 6-month study period, 5.72% of the apixaban group had one or more hospitalizations after the initial event, compared with 7.07% of the control group. This translated to a highly significant 21% relative risk reduction. For every 74 patients treated with apixaban instead of enoxaparin/warfarin, one hospitalization was avoided. Moreover, when a hospitalization occurred in apixaban-treated patients, the mean length of stay was shorter: 10.2 vs. 11.7 days in the enoxaparin-warfarin group.
The median time to a first hospitalization was 63 days in the apixaban group, compared with 34.5 days in controls. The apixaban group’s advantage in terms of hospitalization risk was consistent across subgroups on the basis of age, body weight, sex, and renal function.
The number of emergency department visits during the 6-month follow-up period was similar in the two study arms. However, only 5.8% of the apixaban group visited a physician’s office, compared with 7.3% of controls. The reasons for these office visits were basically the same as for the hospitalizations: mostly recurrent VTEs and bleeding episodes. In the apixaban group, 35 patients had an office visit for recurrent VTE, compared with 61 controls. And 71 apixaban-treated patients made an office visit for bleeding episodes, compared with 130 controls.
Session cochair Dr. Emile R. Mohler commented on the finding that 37 patients in the apixaban group and 48 on enoxaparin/warfarin required hospitalization for recurrent VTE.
"It seems like we’re not doing a good enough job there. Either both of these anticoagulants don’t work well, or the patients aren’t taking the medication, or we’re not following up with them enough. I can’t remember the last time in my own clinical practice that somebody who took their medication came back within 6 months of having a VTE. It seems strange. I think there’s a lot of room for improvement," commented Dr. Mohler, professor of medicine and director of vascular medicine at the University of Pennsylvania, Philadelphia.
Dr. Johnson agreed about the room for improvement. But she added that, although the data she presented were based upon an intention-to-treat analysis, the results were the same – significantly fewer hospitalizations in the apixaban group – in a per-protocol analysis that excluded patients with less than 80% adherence to their study medication.
"One of the big things we saw in AMPLIFY is that whenever you stop an anticoagulant, you see the recurrence rate go up by about 10% per year. So it’s very important that these people continue their anticoagulation – and the more we can reduce their bleeding events, the more likely they are to comply with that therapy and be protected from having another VTE," she added.
Session cochair Dr. John P. Cooke commented that one underutilized aspect of treatment for acute VTE is compressive support, which he said has been given short shrift in the major practice guidelines.
Often as physicians, we give patients a pill and we think that we’ve treated them. Compressive support is important in VTE," stressed Dr. Cooke, chair of the department of cardiovascular sciences at the Houston Methodist Research Institute and director of the Center for Cardiovascular Regeneration at the Houston Methodist DeBakey Heart and Vascular Center.
The AMPLIFY trial was sponsored by Bristol-Myers Squibb and Pfizer. Dr. Johnson reported having no financial conflicts.
WASHINGTON – Treating acute venous thromboembolism with the fixed-dose oral factor Xa inhibitor apixaban significantly reduces subsequent all-cause hospitalizations, compared with conventional therapy with enoxaparin followed by warfarin, according to a secondary analysis of the landmark AMPLIFY trial.
The 21% reduction in the risk of hospitalization in the apixaban group during the 6 months following the initial VTE was driven mainly by fewer hospitalizations for recurrent VTE or major bleeding. There were also significantly fewer physician office visits by patients on apixaban than for those on enoxaparin/warfarin, Dr. Margot Johnson reported at the annual meeting of the American College of Cardiology.
An analysis of the cost savings associated with this reduction in hospitalizations seen in AMPLIFY is underway and will be reported later this year, added Dr. Johnson of King’s College Hospital in London.
AMPLIFY (Apixaban for the Initial Management of Pulmonary Embolism and Deep Vein Thrombosis) was an industry-sponsored randomized double-blind study of 5,365 patients with acute symptomatic VTE who were assigned to 6 months of treatment with apixaban (Eliquis) at 10 mg b.i.d. for 7 days followed by 5 mg b.i.d. or to enoxaparin followed by warfarin. In the previously reported primary outcomes (N. Engl. J. Med. 2013;369:799-808, apixaban showed noninferiority to the conventional regimen in terms of the rate of recurrent VTE or VTE-related death, and a highly significant superiority in terms of major bleeding, with a 69% risk reduction.
Dr. Johnson reported that during the 6-month study period, 5.72% of the apixaban group had one or more hospitalizations after the initial event, compared with 7.07% of the control group. This translated to a highly significant 21% relative risk reduction. For every 74 patients treated with apixaban instead of enoxaparin/warfarin, one hospitalization was avoided. Moreover, when a hospitalization occurred in apixaban-treated patients, the mean length of stay was shorter: 10.2 vs. 11.7 days in the enoxaparin-warfarin group.
The median time to a first hospitalization was 63 days in the apixaban group, compared with 34.5 days in controls. The apixaban group’s advantage in terms of hospitalization risk was consistent across subgroups on the basis of age, body weight, sex, and renal function.
The number of emergency department visits during the 6-month follow-up period was similar in the two study arms. However, only 5.8% of the apixaban group visited a physician’s office, compared with 7.3% of controls. The reasons for these office visits were basically the same as for the hospitalizations: mostly recurrent VTEs and bleeding episodes. In the apixaban group, 35 patients had an office visit for recurrent VTE, compared with 61 controls. And 71 apixaban-treated patients made an office visit for bleeding episodes, compared with 130 controls.
Session cochair Dr. Emile R. Mohler commented on the finding that 37 patients in the apixaban group and 48 on enoxaparin/warfarin required hospitalization for recurrent VTE.
"It seems like we’re not doing a good enough job there. Either both of these anticoagulants don’t work well, or the patients aren’t taking the medication, or we’re not following up with them enough. I can’t remember the last time in my own clinical practice that somebody who took their medication came back within 6 months of having a VTE. It seems strange. I think there’s a lot of room for improvement," commented Dr. Mohler, professor of medicine and director of vascular medicine at the University of Pennsylvania, Philadelphia.
Dr. Johnson agreed about the room for improvement. But she added that, although the data she presented were based upon an intention-to-treat analysis, the results were the same – significantly fewer hospitalizations in the apixaban group – in a per-protocol analysis that excluded patients with less than 80% adherence to their study medication.
"One of the big things we saw in AMPLIFY is that whenever you stop an anticoagulant, you see the recurrence rate go up by about 10% per year. So it’s very important that these people continue their anticoagulation – and the more we can reduce their bleeding events, the more likely they are to comply with that therapy and be protected from having another VTE," she added.
Session cochair Dr. John P. Cooke commented that one underutilized aspect of treatment for acute VTE is compressive support, which he said has been given short shrift in the major practice guidelines.
Often as physicians, we give patients a pill and we think that we’ve treated them. Compressive support is important in VTE," stressed Dr. Cooke, chair of the department of cardiovascular sciences at the Houston Methodist Research Institute and director of the Center for Cardiovascular Regeneration at the Houston Methodist DeBakey Heart and Vascular Center.
The AMPLIFY trial was sponsored by Bristol-Myers Squibb and Pfizer. Dr. Johnson reported having no financial conflicts.
Major finding: Patients whose acute venous thromboembolism was treated with 6 months of apixaban were 21% less likely to have another hospitalization than were those who received enoxaparin followed by warfarin.
Data source: The double-blind AMPLIFY trial, which randomized 5,365 patients with acute VTE to 6 months of fixed-dose apixaban or conventional therapy with enoxaparin followed by warfarin.
Disclosures: The study was sponsored by Bristol-Myers Squibb and Pfizer. The presenter reported having no financial conflicts.
Apixaban for VTE reduced subsequent hospitalizations
WASHINGTON – Treating acute venous thromboembolism with the fixed-dose oral factor Xa inhibitor apixaban significantly reduces subsequent all-cause hospitalizations, compared with conventional therapy with enoxaparin followed by warfarin, according to a secondary analysis of the landmark AMPLIFY trial.
The 21% reduction in the risk of hospitalization in the apixaban group during the 6 months following the initial VTE was driven mainly by fewer hospitalizations for recurrent VTE or major bleeding. There were also significantly fewer physician office visits by patients on apixaban than for those on enoxaparin/warfarin, Dr. Margot Johnson reported at the annual meeting of the American College of Cardiology.
An analysis of the cost savings associated with this reduction in hospitalizations seen in AMPLIFY is underway and will be reported later this year, added Dr. Johnson of King’s College Hospital in London.
AMPLIFY (Apixaban for the Initial Management of Pulmonary Embolism and Deep Vein Thrombosis) was an industry-sponsored randomized double-blind study of 5,365 patients with acute symptomatic VTE who were assigned to 6 months of treatment with apixaban (Eliquis) at 10 mg b.i.d. for 7 days followed by 5 mg b.i.d. or to enoxaparin followed by warfarin. In the previously reported primary outcomes (N. Engl. J. Med. 2013;369:799-808, apixaban showed noninferiority to the conventional regimen in terms of the rate of recurrent VTE or VTE-related death, and a highly significant superiority in terms of major bleeding, with a 69% risk reduction.
Dr. Johnson reported that during the 6-month study period, 5.72% of the apixaban group had one or more hospitalizations after the initial event, compared with 7.07% of the control group. This translated to a highly significant 21% relative risk reduction. For every 74 patients treated with apixaban instead of enoxaparin/warfarin, one hospitalization was avoided. Moreover, when a hospitalization occurred in apixaban-treated patients, the mean length of stay was shorter: 10.2 vs. 11.7 days in the enoxaparin-warfarin group.
The median time to a first hospitalization was 63 days in the apixaban group, compared with 34.5 days in controls. The apixaban group’s advantage in terms of hospitalization risk was consistent across subgroups on the basis of age, body weight, sex, and renal function.
The number of emergency department visits during the 6-month follow-up period was similar in the two study arms. However, only 5.8% of the apixaban group visited a physician’s office, compared with 7.3% of controls. The reasons for these office visits were basically the same as for the hospitalizations: mostly recurrent VTEs and bleeding episodes. In the apixaban group, 35 patients had an office visit for recurrent VTE, compared with 61 controls. And 71 apixaban-treated patients made an office visit for bleeding episodes, compared with 130 controls.
Session cochair Dr. Emile R. Mohler commented on the finding that 37 patients in the apixaban group and 48 on enoxaparin/warfarin required hospitalization for recurrent VTE.
"It seems like we’re not doing a good enough job there. Either both of these anticoagulants don’t work well, or the patients aren’t taking the medication, or we’re not following up with them enough. I can’t remember the last time in my own clinical practice that somebody who took their medication came back within 6 months of having a VTE. It seems strange. I think there’s a lot of room for improvement," commented Dr. Mohler, professor of medicine and director of vascular medicine at the University of Pennsylvania, Philadelphia.
Dr. Johnson agreed about the room for improvement. But she added that, although the data she presented were based upon an intention-to-treat analysis, the results were the same – significantly fewer hospitalizations in the apixaban group – in a per-protocol analysis that excluded patients with less than 80% adherence to their study medication.
"One of the big things we saw in AMPLIFY is that whenever you stop an anticoagulant, you see the recurrence rate go up by about 10% per year. So it’s very important that these people continue their anticoagulation – and the more we can reduce their bleeding events, the more likely they are to comply with that therapy and be protected from having another VTE," she added.
Session cochair Dr. John P. Cooke commented that one underutilized aspect of treatment for acute VTE is compressive support, which he said has been given short shrift in the major practice guidelines.
Often as physicians, we give patients a pill and we think that we’ve treated them. Compressive support is important in VTE," stressed Dr. Cooke, chair of the department of cardiovascular sciences at the Houston Methodist Research Institute and director of the Center for Cardiovascular Regeneration at the Houston Methodist DeBakey Heart and Vascular Center.
The AMPLIFY trial was sponsored by Bristol-Myers Squibb and Pfizer. Dr. Johnson reported having no financial conflicts.
Dr. Vera DePalo, FCCP, comments: This large, industry-sponsored trial provides an alternative to the standard treatments for VTE. Patients had a less likelihood of hospitalization for recurrence or major bleeding. If hospitalization did occur, there was a much longer median time to hospitalization and a shorter hospital length of stay.
Dr. Vera DePalo, FCCP, comments: This large, industry-sponsored trial provides an alternative to the standard treatments for VTE. Patients had a less likelihood of hospitalization for recurrence or major bleeding. If hospitalization did occur, there was a much longer median time to hospitalization and a shorter hospital length of stay.
Dr. Vera DePalo, FCCP, comments: This large, industry-sponsored trial provides an alternative to the standard treatments for VTE. Patients had a less likelihood of hospitalization for recurrence or major bleeding. If hospitalization did occur, there was a much longer median time to hospitalization and a shorter hospital length of stay.
WASHINGTON – Treating acute venous thromboembolism with the fixed-dose oral factor Xa inhibitor apixaban significantly reduces subsequent all-cause hospitalizations, compared with conventional therapy with enoxaparin followed by warfarin, according to a secondary analysis of the landmark AMPLIFY trial.
The 21% reduction in the risk of hospitalization in the apixaban group during the 6 months following the initial VTE was driven mainly by fewer hospitalizations for recurrent VTE or major bleeding. There were also significantly fewer physician office visits by patients on apixaban than for those on enoxaparin/warfarin, Dr. Margot Johnson reported at the annual meeting of the American College of Cardiology.
An analysis of the cost savings associated with this reduction in hospitalizations seen in AMPLIFY is underway and will be reported later this year, added Dr. Johnson of King’s College Hospital in London.
AMPLIFY (Apixaban for the Initial Management of Pulmonary Embolism and Deep Vein Thrombosis) was an industry-sponsored randomized double-blind study of 5,365 patients with acute symptomatic VTE who were assigned to 6 months of treatment with apixaban (Eliquis) at 10 mg b.i.d. for 7 days followed by 5 mg b.i.d. or to enoxaparin followed by warfarin. In the previously reported primary outcomes (N. Engl. J. Med. 2013;369:799-808, apixaban showed noninferiority to the conventional regimen in terms of the rate of recurrent VTE or VTE-related death, and a highly significant superiority in terms of major bleeding, with a 69% risk reduction.
Dr. Johnson reported that during the 6-month study period, 5.72% of the apixaban group had one or more hospitalizations after the initial event, compared with 7.07% of the control group. This translated to a highly significant 21% relative risk reduction. For every 74 patients treated with apixaban instead of enoxaparin/warfarin, one hospitalization was avoided. Moreover, when a hospitalization occurred in apixaban-treated patients, the mean length of stay was shorter: 10.2 vs. 11.7 days in the enoxaparin-warfarin group.
The median time to a first hospitalization was 63 days in the apixaban group, compared with 34.5 days in controls. The apixaban group’s advantage in terms of hospitalization risk was consistent across subgroups on the basis of age, body weight, sex, and renal function.
The number of emergency department visits during the 6-month follow-up period was similar in the two study arms. However, only 5.8% of the apixaban group visited a physician’s office, compared with 7.3% of controls. The reasons for these office visits were basically the same as for the hospitalizations: mostly recurrent VTEs and bleeding episodes. In the apixaban group, 35 patients had an office visit for recurrent VTE, compared with 61 controls. And 71 apixaban-treated patients made an office visit for bleeding episodes, compared with 130 controls.
Session cochair Dr. Emile R. Mohler commented on the finding that 37 patients in the apixaban group and 48 on enoxaparin/warfarin required hospitalization for recurrent VTE.
"It seems like we’re not doing a good enough job there. Either both of these anticoagulants don’t work well, or the patients aren’t taking the medication, or we’re not following up with them enough. I can’t remember the last time in my own clinical practice that somebody who took their medication came back within 6 months of having a VTE. It seems strange. I think there’s a lot of room for improvement," commented Dr. Mohler, professor of medicine and director of vascular medicine at the University of Pennsylvania, Philadelphia.
Dr. Johnson agreed about the room for improvement. But she added that, although the data she presented were based upon an intention-to-treat analysis, the results were the same – significantly fewer hospitalizations in the apixaban group – in a per-protocol analysis that excluded patients with less than 80% adherence to their study medication.
"One of the big things we saw in AMPLIFY is that whenever you stop an anticoagulant, you see the recurrence rate go up by about 10% per year. So it’s very important that these people continue their anticoagulation – and the more we can reduce their bleeding events, the more likely they are to comply with that therapy and be protected from having another VTE," she added.
Session cochair Dr. John P. Cooke commented that one underutilized aspect of treatment for acute VTE is compressive support, which he said has been given short shrift in the major practice guidelines.
Often as physicians, we give patients a pill and we think that we’ve treated them. Compressive support is important in VTE," stressed Dr. Cooke, chair of the department of cardiovascular sciences at the Houston Methodist Research Institute and director of the Center for Cardiovascular Regeneration at the Houston Methodist DeBakey Heart and Vascular Center.
The AMPLIFY trial was sponsored by Bristol-Myers Squibb and Pfizer. Dr. Johnson reported having no financial conflicts.
WASHINGTON – Treating acute venous thromboembolism with the fixed-dose oral factor Xa inhibitor apixaban significantly reduces subsequent all-cause hospitalizations, compared with conventional therapy with enoxaparin followed by warfarin, according to a secondary analysis of the landmark AMPLIFY trial.
The 21% reduction in the risk of hospitalization in the apixaban group during the 6 months following the initial VTE was driven mainly by fewer hospitalizations for recurrent VTE or major bleeding. There were also significantly fewer physician office visits by patients on apixaban than for those on enoxaparin/warfarin, Dr. Margot Johnson reported at the annual meeting of the American College of Cardiology.
An analysis of the cost savings associated with this reduction in hospitalizations seen in AMPLIFY is underway and will be reported later this year, added Dr. Johnson of King’s College Hospital in London.
AMPLIFY (Apixaban for the Initial Management of Pulmonary Embolism and Deep Vein Thrombosis) was an industry-sponsored randomized double-blind study of 5,365 patients with acute symptomatic VTE who were assigned to 6 months of treatment with apixaban (Eliquis) at 10 mg b.i.d. for 7 days followed by 5 mg b.i.d. or to enoxaparin followed by warfarin. In the previously reported primary outcomes (N. Engl. J. Med. 2013;369:799-808, apixaban showed noninferiority to the conventional regimen in terms of the rate of recurrent VTE or VTE-related death, and a highly significant superiority in terms of major bleeding, with a 69% risk reduction.
Dr. Johnson reported that during the 6-month study period, 5.72% of the apixaban group had one or more hospitalizations after the initial event, compared with 7.07% of the control group. This translated to a highly significant 21% relative risk reduction. For every 74 patients treated with apixaban instead of enoxaparin/warfarin, one hospitalization was avoided. Moreover, when a hospitalization occurred in apixaban-treated patients, the mean length of stay was shorter: 10.2 vs. 11.7 days in the enoxaparin-warfarin group.
The median time to a first hospitalization was 63 days in the apixaban group, compared with 34.5 days in controls. The apixaban group’s advantage in terms of hospitalization risk was consistent across subgroups on the basis of age, body weight, sex, and renal function.
The number of emergency department visits during the 6-month follow-up period was similar in the two study arms. However, only 5.8% of the apixaban group visited a physician’s office, compared with 7.3% of controls. The reasons for these office visits were basically the same as for the hospitalizations: mostly recurrent VTEs and bleeding episodes. In the apixaban group, 35 patients had an office visit for recurrent VTE, compared with 61 controls. And 71 apixaban-treated patients made an office visit for bleeding episodes, compared with 130 controls.
Session cochair Dr. Emile R. Mohler commented on the finding that 37 patients in the apixaban group and 48 on enoxaparin/warfarin required hospitalization for recurrent VTE.
"It seems like we’re not doing a good enough job there. Either both of these anticoagulants don’t work well, or the patients aren’t taking the medication, or we’re not following up with them enough. I can’t remember the last time in my own clinical practice that somebody who took their medication came back within 6 months of having a VTE. It seems strange. I think there’s a lot of room for improvement," commented Dr. Mohler, professor of medicine and director of vascular medicine at the University of Pennsylvania, Philadelphia.
Dr. Johnson agreed about the room for improvement. But she added that, although the data she presented were based upon an intention-to-treat analysis, the results were the same – significantly fewer hospitalizations in the apixaban group – in a per-protocol analysis that excluded patients with less than 80% adherence to their study medication.
"One of the big things we saw in AMPLIFY is that whenever you stop an anticoagulant, you see the recurrence rate go up by about 10% per year. So it’s very important that these people continue their anticoagulation – and the more we can reduce their bleeding events, the more likely they are to comply with that therapy and be protected from having another VTE," she added.
Session cochair Dr. John P. Cooke commented that one underutilized aspect of treatment for acute VTE is compressive support, which he said has been given short shrift in the major practice guidelines.
Often as physicians, we give patients a pill and we think that we’ve treated them. Compressive support is important in VTE," stressed Dr. Cooke, chair of the department of cardiovascular sciences at the Houston Methodist Research Institute and director of the Center for Cardiovascular Regeneration at the Houston Methodist DeBakey Heart and Vascular Center.
The AMPLIFY trial was sponsored by Bristol-Myers Squibb and Pfizer. Dr. Johnson reported having no financial conflicts.
Major finding: Patients whose acute venous thromboembolism was treated with 6 months of apixaban were 21% less likely to have another hospitalization than were those who received enoxaparin followed by warfarin.
Data source: The double-blind AMPLIFY trial, which randomized 5,365 patients with acute VTE to 6 months of fixed-dose apixaban or conventional therapy with enoxaparin followed by warfarin.
Disclosures: The study was sponsored by Bristol-Myers Squibb and Pfizer. The presenter reported having no financial conflicts.
Off-label use of novel anticoagulants accelerates
WASHINGTON – The off-label use of novel oral anticoagulants for stroke prevention in patients with valvular atrial fibrillation has climbed steeply since the drugs reached the marketplace, mirroring the medications’ rapid adoption for the approved indication of preventing strokes in nonvalvular AF, according to Dr. Sandeep Mahrendra Jani.
An analysis of 190,227 nonvalvular atrial fibrillation (NVAF) patients in 95 practices participating in the American College of Cardiology’s National Cardiovascular Data Registry – PINNACLE Registry – showed that during the first quarter of 2011, just 4.8% were on dabigatran, the sole novel oral anticoagulant then available. By the fourth quarter of 2012, however, 14.9% of NVAF patients were on a novel oral anticoagulant, either dabigatran or the subsequently approved rivaroxiban, he reported at the annual meeting of the ACC.
Similarly, among 2,142 registry participants with valvular atrial fibrillation (AF), the use of any novel oral anticoagulant shot up from 2.7% in the first quarter of 2011 to 13.8% in the fourth quarter of 2012, noted Dr. Jani of Medstar Washington (D.C.) Hospital Center.
During this time – prior to the arrival of apixiban on the market – the use of warfarin for stroke prevention in patients with NVAF declined from 47.9% to 44.3%. Among patients with valvular atrial fibrillation, the prevalence of warfarin therapy fell from 65.8% in the first quarter of 2011 to 60.1% in fourth quarter 2012.
During the first quarter of 2011, 51.2% of all patients with NVAF and 66.4% with valvular AF were on any oral anticoagulant. By fourth quarter 2012, these rates had increased to 56.9% and 66.8%, respectively.
The use of dabigatran in patients with valvular AF took a hit in late 2012 in response to the premature halt of the RE-ALIGN (Dabigatran Etexilate in Patients With Mechanical Heart Valves) trial, followed by the Food and Drug Administration’s warning against using dabigatran in patients with mechanical heart valves. Dabigatran was used by 2.7% of valvular AF patients in the first quarter of 2011, rising steadily to 12.1% by the third quarter of 2012, then plunging to just 1.4% in the year’s final quarter.
In light of the rapidly accelerating use of novel oral anticoagulants in patients with valvular AF, despite a lack of evidence of efficacy for stroke prevention in this setting, Further studies are a priority, Dr. Jani said.
The PINNACLE registry is funded by the ACC, with founding sponsorship provided by Bristol-Myers Squibb and Pfizer. Dr. Jani reported having no relevant financial conflicts.
Dr. Jun Chiong, FCCP, comments: Direct thrombin inhibitor (DTI) use has definitely gained wide acceptance in a short period of time, mainly because of its efficacy and its stable drug level, which eliminated regular prothrombin time testing requirements that are inherent with warfarin.
Marketing including print advertisements played a significant role in its widespread use. However, it is important for prescribers to remember that it's our role to read package inserts before prescribing any drugs as well as manufacturers role to educate the medical providers about its use, contraindications, and misuse of these agents.
To date, studies on DTI for valvular atrial fibrillation failed to demonstrate benefit over warfarin.
Dr. Jun Chiong, FCCP, comments: Direct thrombin inhibitor (DTI) use has definitely gained wide acceptance in a short period of time, mainly because of its efficacy and its stable drug level, which eliminated regular prothrombin time testing requirements that are inherent with warfarin.
Marketing including print advertisements played a significant role in its widespread use. However, it is important for prescribers to remember that it's our role to read package inserts before prescribing any drugs as well as manufacturers role to educate the medical providers about its use, contraindications, and misuse of these agents.
To date, studies on DTI for valvular atrial fibrillation failed to demonstrate benefit over warfarin.
Dr. Jun Chiong, FCCP, comments: Direct thrombin inhibitor (DTI) use has definitely gained wide acceptance in a short period of time, mainly because of its efficacy and its stable drug level, which eliminated regular prothrombin time testing requirements that are inherent with warfarin.
Marketing including print advertisements played a significant role in its widespread use. However, it is important for prescribers to remember that it's our role to read package inserts before prescribing any drugs as well as manufacturers role to educate the medical providers about its use, contraindications, and misuse of these agents.
To date, studies on DTI for valvular atrial fibrillation failed to demonstrate benefit over warfarin.
WASHINGTON – The off-label use of novel oral anticoagulants for stroke prevention in patients with valvular atrial fibrillation has climbed steeply since the drugs reached the marketplace, mirroring the medications’ rapid adoption for the approved indication of preventing strokes in nonvalvular AF, according to Dr. Sandeep Mahrendra Jani.
An analysis of 190,227 nonvalvular atrial fibrillation (NVAF) patients in 95 practices participating in the American College of Cardiology’s National Cardiovascular Data Registry – PINNACLE Registry – showed that during the first quarter of 2011, just 4.8% were on dabigatran, the sole novel oral anticoagulant then available. By the fourth quarter of 2012, however, 14.9% of NVAF patients were on a novel oral anticoagulant, either dabigatran or the subsequently approved rivaroxiban, he reported at the annual meeting of the ACC.
Similarly, among 2,142 registry participants with valvular atrial fibrillation (AF), the use of any novel oral anticoagulant shot up from 2.7% in the first quarter of 2011 to 13.8% in the fourth quarter of 2012, noted Dr. Jani of Medstar Washington (D.C.) Hospital Center.
During this time – prior to the arrival of apixiban on the market – the use of warfarin for stroke prevention in patients with NVAF declined from 47.9% to 44.3%. Among patients with valvular atrial fibrillation, the prevalence of warfarin therapy fell from 65.8% in the first quarter of 2011 to 60.1% in fourth quarter 2012.
During the first quarter of 2011, 51.2% of all patients with NVAF and 66.4% with valvular AF were on any oral anticoagulant. By fourth quarter 2012, these rates had increased to 56.9% and 66.8%, respectively.
The use of dabigatran in patients with valvular AF took a hit in late 2012 in response to the premature halt of the RE-ALIGN (Dabigatran Etexilate in Patients With Mechanical Heart Valves) trial, followed by the Food and Drug Administration’s warning against using dabigatran in patients with mechanical heart valves. Dabigatran was used by 2.7% of valvular AF patients in the first quarter of 2011, rising steadily to 12.1% by the third quarter of 2012, then plunging to just 1.4% in the year’s final quarter.
In light of the rapidly accelerating use of novel oral anticoagulants in patients with valvular AF, despite a lack of evidence of efficacy for stroke prevention in this setting, Further studies are a priority, Dr. Jani said.
The PINNACLE registry is funded by the ACC, with founding sponsorship provided by Bristol-Myers Squibb and Pfizer. Dr. Jani reported having no relevant financial conflicts.
WASHINGTON – The off-label use of novel oral anticoagulants for stroke prevention in patients with valvular atrial fibrillation has climbed steeply since the drugs reached the marketplace, mirroring the medications’ rapid adoption for the approved indication of preventing strokes in nonvalvular AF, according to Dr. Sandeep Mahrendra Jani.
An analysis of 190,227 nonvalvular atrial fibrillation (NVAF) patients in 95 practices participating in the American College of Cardiology’s National Cardiovascular Data Registry – PINNACLE Registry – showed that during the first quarter of 2011, just 4.8% were on dabigatran, the sole novel oral anticoagulant then available. By the fourth quarter of 2012, however, 14.9% of NVAF patients were on a novel oral anticoagulant, either dabigatran or the subsequently approved rivaroxiban, he reported at the annual meeting of the ACC.
Similarly, among 2,142 registry participants with valvular atrial fibrillation (AF), the use of any novel oral anticoagulant shot up from 2.7% in the first quarter of 2011 to 13.8% in the fourth quarter of 2012, noted Dr. Jani of Medstar Washington (D.C.) Hospital Center.
During this time – prior to the arrival of apixiban on the market – the use of warfarin for stroke prevention in patients with NVAF declined from 47.9% to 44.3%. Among patients with valvular atrial fibrillation, the prevalence of warfarin therapy fell from 65.8% in the first quarter of 2011 to 60.1% in fourth quarter 2012.
During the first quarter of 2011, 51.2% of all patients with NVAF and 66.4% with valvular AF were on any oral anticoagulant. By fourth quarter 2012, these rates had increased to 56.9% and 66.8%, respectively.
The use of dabigatran in patients with valvular AF took a hit in late 2012 in response to the premature halt of the RE-ALIGN (Dabigatran Etexilate in Patients With Mechanical Heart Valves) trial, followed by the Food and Drug Administration’s warning against using dabigatran in patients with mechanical heart valves. Dabigatran was used by 2.7% of valvular AF patients in the first quarter of 2011, rising steadily to 12.1% by the third quarter of 2012, then plunging to just 1.4% in the year’s final quarter.
In light of the rapidly accelerating use of novel oral anticoagulants in patients with valvular AF, despite a lack of evidence of efficacy for stroke prevention in this setting, Further studies are a priority, Dr. Jani said.
The PINNACLE registry is funded by the ACC, with founding sponsorship provided by Bristol-Myers Squibb and Pfizer. Dr. Jani reported having no relevant financial conflicts.
Key clinical point: Off-label use of novel anticoagulants is growing.
Major finding: By the fourth quarter of 2012, about 15% of patients with nonvalvular AF and about 14% of those with valvular AF were on a novel anticoagulant.
Data source: A study of more than 190,000 patients participating in the PINNACLE Registry.
Disclosures: The PINNACLE Registry is funded by the ACC’s National Cardiovascular Data Registry. The presenter reported having no financial conflicts.
Even mild preop sepsis boosts postop clot risk
WASHINGTON – Preoperative sepsis proved to be an important independent risk factor for both arterial and venous thrombosis during or after surgery in an analysis of nearly 1.75 million U.S. surgical procedures.
The take-home message here is that the risk-benefit assessment of surgical procedures should take into account the presence of sepsis. And if the surgery can’t be delayed, prophylaxis against arterial as well as venous thrombosis should be employed, Dr. Jacques Donzé said at the annual meeting of the American College of Cardiology.
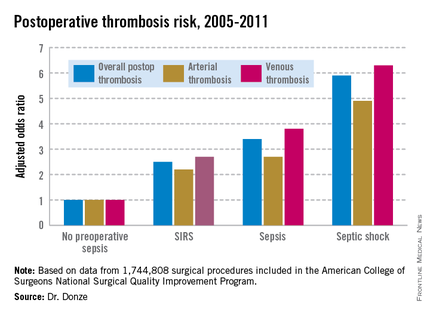
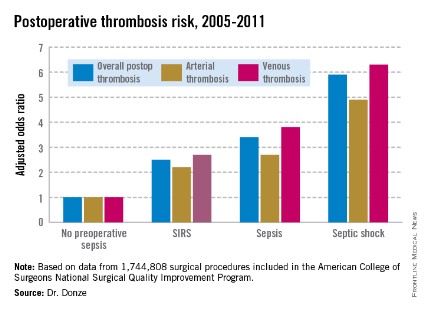
Another key finding in this study was that the risk of postoperative thrombosis varied according to the severity of preoperative sepsis. Even the early form of sepsis known as systemic inflammatory response syndrome, or SIRS, was associated with a 2.5-fold increased risk.
"Include even early signs of sepsis as a risk factor," urged Dr. Donzé of Brigham and Women’s Hospital, Boston.
Also, preoperative sepsis was a risk factor for postoperative thrombosis in connection with outpatient elective surgery as well as inpatient operations, he added.
Dr. Donzé presented an analysis of 1,744,808 surgical procedures performed during 2005-2011 at 314 U.S. hospitals participating in the American College of Surgeons National Surgical Quality Improvement Program. This large, prospective, observational registry is known for its high-quality data.
Within 48 hours prior to surgery, 7.8% of patients – totaling more than 136,000 – had SIRS, sepsis, or septic shock. Their postoperative thrombosis rate was 4.2%, compared with a 1.2% rate in patients without sepsis. In a multivariate regression analysis adjusted for potential confounding factors, the postoperative thrombosis risk climbed with increasing severity of preoperative sepsis (see graphic).
SIRS was defined on the basis of temperature, heart rate, respiratory rate, WBC count, and/or the presence of anion gap acidosis. "Sepsis" was defined as SIRS plus infection. And septic shock required the presence of sepsis plus documented organ dysfunction, such as hypotension.
The importance of recognizing this newly spotlighted sepsis/postoperative thrombosis connection is that most of the other known risk factors for thrombosis in surgical patients, including age, cancer, renal failure, and immobilization, are nonmodifiable, Dr. Donze observed.
Among the factors known to contribute to thrombosis are a hypercoagulable state, a proinflammatory state, hypoxemia, hypotension, and endothelial dysfunction. "All of these factors can be triggered by sepsis," Dr. Donze noted.
He reported having no financial conflicts regarding this study.
Dr. Marcos I. Restrepo, FCCP, comments: This interesting observation links the associated risk of preoperative sepsis and any degree of sepsis severity with a higher risk of developing postoperative arterial and venous thrombosis. Therefore, it is critical to recognize and identify preoperative patients with systemic inflammatory response syndrome with a suspected or confirmed source of infection ("sepsis") in order to initiate early and appropriate thromboprophylactic measures.
Dr. Marcos I. Restrepo, FCCP, comments: This interesting observation links the associated risk of preoperative sepsis and any degree of sepsis severity with a higher risk of developing postoperative arterial and venous thrombosis. Therefore, it is critical to recognize and identify preoperative patients with systemic inflammatory response syndrome with a suspected or confirmed source of infection ("sepsis") in order to initiate early and appropriate thromboprophylactic measures.
Dr. Marcos I. Restrepo, FCCP, comments: This interesting observation links the associated risk of preoperative sepsis and any degree of sepsis severity with a higher risk of developing postoperative arterial and venous thrombosis. Therefore, it is critical to recognize and identify preoperative patients with systemic inflammatory response syndrome with a suspected or confirmed source of infection ("sepsis") in order to initiate early and appropriate thromboprophylactic measures.
WASHINGTON – Preoperative sepsis proved to be an important independent risk factor for both arterial and venous thrombosis during or after surgery in an analysis of nearly 1.75 million U.S. surgical procedures.
The take-home message here is that the risk-benefit assessment of surgical procedures should take into account the presence of sepsis. And if the surgery can’t be delayed, prophylaxis against arterial as well as venous thrombosis should be employed, Dr. Jacques Donzé said at the annual meeting of the American College of Cardiology.

Another key finding in this study was that the risk of postoperative thrombosis varied according to the severity of preoperative sepsis. Even the early form of sepsis known as systemic inflammatory response syndrome, or SIRS, was associated with a 2.5-fold increased risk.
"Include even early signs of sepsis as a risk factor," urged Dr. Donzé of Brigham and Women’s Hospital, Boston.
Also, preoperative sepsis was a risk factor for postoperative thrombosis in connection with outpatient elective surgery as well as inpatient operations, he added.
Dr. Donzé presented an analysis of 1,744,808 surgical procedures performed during 2005-2011 at 314 U.S. hospitals participating in the American College of Surgeons National Surgical Quality Improvement Program. This large, prospective, observational registry is known for its high-quality data.
Within 48 hours prior to surgery, 7.8% of patients – totaling more than 136,000 – had SIRS, sepsis, or septic shock. Their postoperative thrombosis rate was 4.2%, compared with a 1.2% rate in patients without sepsis. In a multivariate regression analysis adjusted for potential confounding factors, the postoperative thrombosis risk climbed with increasing severity of preoperative sepsis (see graphic).
SIRS was defined on the basis of temperature, heart rate, respiratory rate, WBC count, and/or the presence of anion gap acidosis. "Sepsis" was defined as SIRS plus infection. And septic shock required the presence of sepsis plus documented organ dysfunction, such as hypotension.
The importance of recognizing this newly spotlighted sepsis/postoperative thrombosis connection is that most of the other known risk factors for thrombosis in surgical patients, including age, cancer, renal failure, and immobilization, are nonmodifiable, Dr. Donze observed.
Among the factors known to contribute to thrombosis are a hypercoagulable state, a proinflammatory state, hypoxemia, hypotension, and endothelial dysfunction. "All of these factors can be triggered by sepsis," Dr. Donze noted.
He reported having no financial conflicts regarding this study.
WASHINGTON – Preoperative sepsis proved to be an important independent risk factor for both arterial and venous thrombosis during or after surgery in an analysis of nearly 1.75 million U.S. surgical procedures.
The take-home message here is that the risk-benefit assessment of surgical procedures should take into account the presence of sepsis. And if the surgery can’t be delayed, prophylaxis against arterial as well as venous thrombosis should be employed, Dr. Jacques Donzé said at the annual meeting of the American College of Cardiology.

Another key finding in this study was that the risk of postoperative thrombosis varied according to the severity of preoperative sepsis. Even the early form of sepsis known as systemic inflammatory response syndrome, or SIRS, was associated with a 2.5-fold increased risk.
"Include even early signs of sepsis as a risk factor," urged Dr. Donzé of Brigham and Women’s Hospital, Boston.
Also, preoperative sepsis was a risk factor for postoperative thrombosis in connection with outpatient elective surgery as well as inpatient operations, he added.
Dr. Donzé presented an analysis of 1,744,808 surgical procedures performed during 2005-2011 at 314 U.S. hospitals participating in the American College of Surgeons National Surgical Quality Improvement Program. This large, prospective, observational registry is known for its high-quality data.
Within 48 hours prior to surgery, 7.8% of patients – totaling more than 136,000 – had SIRS, sepsis, or septic shock. Their postoperative thrombosis rate was 4.2%, compared with a 1.2% rate in patients without sepsis. In a multivariate regression analysis adjusted for potential confounding factors, the postoperative thrombosis risk climbed with increasing severity of preoperative sepsis (see graphic).
SIRS was defined on the basis of temperature, heart rate, respiratory rate, WBC count, and/or the presence of anion gap acidosis. "Sepsis" was defined as SIRS plus infection. And septic shock required the presence of sepsis plus documented organ dysfunction, such as hypotension.
The importance of recognizing this newly spotlighted sepsis/postoperative thrombosis connection is that most of the other known risk factors for thrombosis in surgical patients, including age, cancer, renal failure, and immobilization, are nonmodifiable, Dr. Donze observed.
Among the factors known to contribute to thrombosis are a hypercoagulable state, a proinflammatory state, hypoxemia, hypotension, and endothelial dysfunction. "All of these factors can be triggered by sepsis," Dr. Donze noted.
He reported having no financial conflicts regarding this study.
Major finding: Preoperative sepsis is a strong independent risk factor for postoperative arterial and venous thrombosis; the more severe the sepsis, the greater the thrombosis risk.
Data source: This was an analysis of nearly 1.75 million surgical procedures at 314 U.S. hospitals detailed in the American College of Surgeons National Quality Improvement Program registry.
Disclosures: The presenter reported having no financial conflicts.
High uric acid boosts fracture risk
LAS VEGAS – A serum uric acid level of 7 mg/dL or more was independently associated with a 62% increased risk of hip fracture among older men participating in the Cardiovascular Health Study.
This finding confirms the clinical relevance of prior translational work pointing to a link between uric acid and bone health, Dr. Tapan Mehta observed at a meeting sponsored by the National Kidney Foundation.
He and his coinvestigators had suspected the existence of such a link, based upon their prior observations that uric acid inhibits expression of 1-alpha hydroxylase protein in vivo and that elevated uric acid levels are associated with higher parathyroid hormone levels in humans.
A key question now under study in clinical trials is whether lowering uric acid levels in the absence of symptomatic gout provides ancillary benefits to the kidneys.
The Cardiovascular Health Study was a National Heart, Lung, and Blood Institute–funded, population-based, observational study conducted to identify cardiovascular risk factors in men and women aged 65 years and older. In this analysis, Dr. Mehta reported on 1,963 male and 2,729 female participants with baseline serum uric acid levels available.
During a median 11 years of follow-up, 156 first-incident hip fractures occurred. The association between hyperuricemia and increased fracture risk was confined to men, 430 of whom had a baseline serum uric acid of 7 mg/dL or more.
Participants with an elevated uric acid were disproportionately black, obese, in less than good health, and had significantly higher levels of insulin, C-reactive protein, and cystatin C. In a multivariate regression analysis adjusted for demographics, body mass index, alcohol, smoking, physical activity, diabetes, C-reactive protein, hypertension, and known cardiovascular disease, men with a serum uric acid of 7 mg/dL or higher remained at 62% greater risk of hip fracture, compared with those with a uric acid below 7 mg/dL, according to Dr. Mehta of the University of Colorado, Denver.
High uric acid is known to increase the risk of gout, kidney stones, and more rapid progression of chronic kidney disease. It has also been linked to hypertension.
Possible mechanisms that might explain the link between increased serum uric acid and hip fractures include the fact that uric acid suppresses vitamin D activation. Also, uric acid inhibits expression of 1-alpha hydroxylase protein, and higher uric acid levels are associated with higher levels of parathyroid hormone. In addition, uric acid may cause inflammation in bone, which would be expected to increase fragility. But all of these thoughts on mechanism remain speculative for now, according to the nephrologist.
The absence of a relationship between hyperuricemia and hip fractures in older women in the Cardiovascular Health Study suggests other factors are more important than uric acid levels in women.
Dr. Mehta reported having no financial conflicts regarding this study.
LAS VEGAS – A serum uric acid level of 7 mg/dL or more was independently associated with a 62% increased risk of hip fracture among older men participating in the Cardiovascular Health Study.
This finding confirms the clinical relevance of prior translational work pointing to a link between uric acid and bone health, Dr. Tapan Mehta observed at a meeting sponsored by the National Kidney Foundation.
He and his coinvestigators had suspected the existence of such a link, based upon their prior observations that uric acid inhibits expression of 1-alpha hydroxylase protein in vivo and that elevated uric acid levels are associated with higher parathyroid hormone levels in humans.
A key question now under study in clinical trials is whether lowering uric acid levels in the absence of symptomatic gout provides ancillary benefits to the kidneys.
The Cardiovascular Health Study was a National Heart, Lung, and Blood Institute–funded, population-based, observational study conducted to identify cardiovascular risk factors in men and women aged 65 years and older. In this analysis, Dr. Mehta reported on 1,963 male and 2,729 female participants with baseline serum uric acid levels available.
During a median 11 years of follow-up, 156 first-incident hip fractures occurred. The association between hyperuricemia and increased fracture risk was confined to men, 430 of whom had a baseline serum uric acid of 7 mg/dL or more.
Participants with an elevated uric acid were disproportionately black, obese, in less than good health, and had significantly higher levels of insulin, C-reactive protein, and cystatin C. In a multivariate regression analysis adjusted for demographics, body mass index, alcohol, smoking, physical activity, diabetes, C-reactive protein, hypertension, and known cardiovascular disease, men with a serum uric acid of 7 mg/dL or higher remained at 62% greater risk of hip fracture, compared with those with a uric acid below 7 mg/dL, according to Dr. Mehta of the University of Colorado, Denver.
High uric acid is known to increase the risk of gout, kidney stones, and more rapid progression of chronic kidney disease. It has also been linked to hypertension.
Possible mechanisms that might explain the link between increased serum uric acid and hip fractures include the fact that uric acid suppresses vitamin D activation. Also, uric acid inhibits expression of 1-alpha hydroxylase protein, and higher uric acid levels are associated with higher levels of parathyroid hormone. In addition, uric acid may cause inflammation in bone, which would be expected to increase fragility. But all of these thoughts on mechanism remain speculative for now, according to the nephrologist.
The absence of a relationship between hyperuricemia and hip fractures in older women in the Cardiovascular Health Study suggests other factors are more important than uric acid levels in women.
Dr. Mehta reported having no financial conflicts regarding this study.
LAS VEGAS – A serum uric acid level of 7 mg/dL or more was independently associated with a 62% increased risk of hip fracture among older men participating in the Cardiovascular Health Study.
This finding confirms the clinical relevance of prior translational work pointing to a link between uric acid and bone health, Dr. Tapan Mehta observed at a meeting sponsored by the National Kidney Foundation.
He and his coinvestigators had suspected the existence of such a link, based upon their prior observations that uric acid inhibits expression of 1-alpha hydroxylase protein in vivo and that elevated uric acid levels are associated with higher parathyroid hormone levels in humans.
A key question now under study in clinical trials is whether lowering uric acid levels in the absence of symptomatic gout provides ancillary benefits to the kidneys.
The Cardiovascular Health Study was a National Heart, Lung, and Blood Institute–funded, population-based, observational study conducted to identify cardiovascular risk factors in men and women aged 65 years and older. In this analysis, Dr. Mehta reported on 1,963 male and 2,729 female participants with baseline serum uric acid levels available.
During a median 11 years of follow-up, 156 first-incident hip fractures occurred. The association between hyperuricemia and increased fracture risk was confined to men, 430 of whom had a baseline serum uric acid of 7 mg/dL or more.
Participants with an elevated uric acid were disproportionately black, obese, in less than good health, and had significantly higher levels of insulin, C-reactive protein, and cystatin C. In a multivariate regression analysis adjusted for demographics, body mass index, alcohol, smoking, physical activity, diabetes, C-reactive protein, hypertension, and known cardiovascular disease, men with a serum uric acid of 7 mg/dL or higher remained at 62% greater risk of hip fracture, compared with those with a uric acid below 7 mg/dL, according to Dr. Mehta of the University of Colorado, Denver.
High uric acid is known to increase the risk of gout, kidney stones, and more rapid progression of chronic kidney disease. It has also been linked to hypertension.
Possible mechanisms that might explain the link between increased serum uric acid and hip fractures include the fact that uric acid suppresses vitamin D activation. Also, uric acid inhibits expression of 1-alpha hydroxylase protein, and higher uric acid levels are associated with higher levels of parathyroid hormone. In addition, uric acid may cause inflammation in bone, which would be expected to increase fragility. But all of these thoughts on mechanism remain speculative for now, according to the nephrologist.
The absence of a relationship between hyperuricemia and hip fractures in older women in the Cardiovascular Health Study suggests other factors are more important than uric acid levels in women.
Dr. Mehta reported having no financial conflicts regarding this study.
AT SCM 14
Major finding: Men aged 65 years or older with a baseline serum uric acid level of 7.0 mg/dL or higher had an adjusted 62% increased risk of a first hip fracture during a median 11 years of follow-up.
Data source: This was an analysis of 1,963 men and 2,729 women, all aged 65 years or older at entry into the population-based, observational, prospective Cardiovascular Health Study.
Disclosures: The Cardiovascular Health Study was funded by the National Heart, Lung, and Blood Institute. The presenter reported having no financial conflicts.
VIDEO: Vibrating pill may shake up constipation treatment
CHICAGO – A newly developed oral capsule may offer chronic idiopathic constipation patients a novel, nonpharmacologic approach to treatment: It vibrates as it moves through the intestines.
In a video interview at the annual Digestive Disease Week, the study’s lead researcher, Dr. Yishai Ron of Tel Aviv Sourasky Medical Center, explained how the mechanical pill works, what effects it had on users’ spontaneous bowel movements and constipation symptoms, and why such an approach may have promise in treating chronic idiopathic constipation.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – A newly developed oral capsule may offer chronic idiopathic constipation patients a novel, nonpharmacologic approach to treatment: It vibrates as it moves through the intestines.
In a video interview at the annual Digestive Disease Week, the study’s lead researcher, Dr. Yishai Ron of Tel Aviv Sourasky Medical Center, explained how the mechanical pill works, what effects it had on users’ spontaneous bowel movements and constipation symptoms, and why such an approach may have promise in treating chronic idiopathic constipation.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – A newly developed oral capsule may offer chronic idiopathic constipation patients a novel, nonpharmacologic approach to treatment: It vibrates as it moves through the intestines.
In a video interview at the annual Digestive Disease Week, the study’s lead researcher, Dr. Yishai Ron of Tel Aviv Sourasky Medical Center, explained how the mechanical pill works, what effects it had on users’ spontaneous bowel movements and constipation symptoms, and why such an approach may have promise in treating chronic idiopathic constipation.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT DDW 2014
GERD may boost risk of MI
CHICAGO – Gastroesophageal reflux disease may constitute a heretofore unrecognized risk factor for coronary heart disease.
In a nationwide case-control study of prodigious proportions, endoscopically confirmed GERD in patients without known coronary or peripheral artery disease at baseline was independently associated with a 57% increased risk of having a first acute MI within the next 5 years, Dr. Ravi K. Prakash reported at the annual Digestive Disease Week.
If this novel finding is confirmed in other databases, the clinical implications would be profound. GERD is after all an extremely common problem, affecting 30%-40% of the U.S. population, noted Dr. Prakash, a gastroenterology fellow at MetroHealth Medical Center and Case Western Reserve University, Cleveland.
The good news: When Dr. Prakash and his coinvestigators divided the 316,390 study participants with GERD into those who were on proton pump inhibitor therapy and those who weren’t, they found that the PPI users weren’t at increased MI risk, compared with the more than 13.6 million similarly aged control subjects who didn’t have GERD, had never undergone an upper endoscopy, and were free of known atherosclerotic disease at baseline.
"Effective treatment of GERD appears to protect against MI," he concluded.
The study population was drawn from Explorys, a private, cloud-based health database containing the electronic health records of 35 million U.S. patients as provided by more than 200,000 physicians in 300-plus health care systems.
A first MI occurred during follow-up in 18,860 GERD patients, or 5.96%, compared with 144,140 controls, or 1.05%. This translates into an unadjusted sixfold increased risk of acute MI in patients with rigorously diagnosed GERD, compared with GERD-free controls.
The investigators then performed a multivariate logistic regression analysis adjusted for six major cardiovascular risk factors: obesity, hypertension, diabetes, tobacco use, hyperlipidemia, and sex. Many of these risk factors were substantially more prevalent in the GERD population. As a result, the unadjusted sixfold increase in MI risk associated with GERD was attenuated in this adjusted risk model; however, the adjusted 57% increased risk remained highly significant.
Moreover, the increased relative risk of GERD seen in the multivariate analysis was comparable with the risks posed by many of the established risk factors. Obesity, for example, showed a 49% increase in MI risk, smoking a 90% increase, and hypertension a 10% increased relative risk, compared with normotension. Only hyperlipidemia and diabetes were in another league, with relative risks of 9.5- and 2.2-fold, respectively, Dr. Prakash continued.
He observed that during the past decade, the research focus has shifted away from GERD as a caustic disease process causing local injury to GERD as a systemic inflammatory disease. Among the proinflammatory cytokines shown to be elevated both in the esophagus and circulation of GERD patients are interleukins-6, -8, and -1B as well as platelet-activating factor. These cytokines are well established as important players in the formation of atherosclerotic plaque in arteries. It was the shared elevations in proinflammatory cytokines that led Dr. Prakash and coworkers to hypothesize that patients with GERD might have an increased incidence of MI.
It’s only relatively recently that several other common chronic diseases have been reconceptualized as systemic inflammatory diseases associated with increased cardiovascular risk. Diabetes, for example, has been repositioned from a straightforward endocrine disease to new status as a coronary heart disease equivalent. And strong bodies of evidence link psoriasis and periodontitis to increased cardiovascular risk, Dr. Prakash noted.
Audience member Dr. Nimish B. Vakil, a Milwaukee gastroenterologist, rose to caution about the possibility of residual confounding by other factors not accounted for in the multivariate analysis, and thus the need for confirmatory studies. That being said, he added that he finds the notion of a GERD/acute MI link mechanistically quite plausible. Acid exposure from GERD might very well trigger exaggerated firing of the esophagocardiac reflex, with resultant episodes of coronary hypoperfusion. Effective PPI therapy for GERD would be expected to reduce such events.
Dr. Prakash said his literature search suggested another possible mechanism for the apparent protective effect of PPIs against MI: effective treatment of GERD, whether using PPIs or via a fundoplication procedure, has been reported to reduce levels of the inflammatory cytokines present in GERD.
Dr. Prakash reported having no financial conflicts with regard to his study, which was supported by institutional funds.
CHICAGO – Gastroesophageal reflux disease may constitute a heretofore unrecognized risk factor for coronary heart disease.
In a nationwide case-control study of prodigious proportions, endoscopically confirmed GERD in patients without known coronary or peripheral artery disease at baseline was independently associated with a 57% increased risk of having a first acute MI within the next 5 years, Dr. Ravi K. Prakash reported at the annual Digestive Disease Week.
If this novel finding is confirmed in other databases, the clinical implications would be profound. GERD is after all an extremely common problem, affecting 30%-40% of the U.S. population, noted Dr. Prakash, a gastroenterology fellow at MetroHealth Medical Center and Case Western Reserve University, Cleveland.
The good news: When Dr. Prakash and his coinvestigators divided the 316,390 study participants with GERD into those who were on proton pump inhibitor therapy and those who weren’t, they found that the PPI users weren’t at increased MI risk, compared with the more than 13.6 million similarly aged control subjects who didn’t have GERD, had never undergone an upper endoscopy, and were free of known atherosclerotic disease at baseline.
"Effective treatment of GERD appears to protect against MI," he concluded.
The study population was drawn from Explorys, a private, cloud-based health database containing the electronic health records of 35 million U.S. patients as provided by more than 200,000 physicians in 300-plus health care systems.
A first MI occurred during follow-up in 18,860 GERD patients, or 5.96%, compared with 144,140 controls, or 1.05%. This translates into an unadjusted sixfold increased risk of acute MI in patients with rigorously diagnosed GERD, compared with GERD-free controls.
The investigators then performed a multivariate logistic regression analysis adjusted for six major cardiovascular risk factors: obesity, hypertension, diabetes, tobacco use, hyperlipidemia, and sex. Many of these risk factors were substantially more prevalent in the GERD population. As a result, the unadjusted sixfold increase in MI risk associated with GERD was attenuated in this adjusted risk model; however, the adjusted 57% increased risk remained highly significant.
Moreover, the increased relative risk of GERD seen in the multivariate analysis was comparable with the risks posed by many of the established risk factors. Obesity, for example, showed a 49% increase in MI risk, smoking a 90% increase, and hypertension a 10% increased relative risk, compared with normotension. Only hyperlipidemia and diabetes were in another league, with relative risks of 9.5- and 2.2-fold, respectively, Dr. Prakash continued.
He observed that during the past decade, the research focus has shifted away from GERD as a caustic disease process causing local injury to GERD as a systemic inflammatory disease. Among the proinflammatory cytokines shown to be elevated both in the esophagus and circulation of GERD patients are interleukins-6, -8, and -1B as well as platelet-activating factor. These cytokines are well established as important players in the formation of atherosclerotic plaque in arteries. It was the shared elevations in proinflammatory cytokines that led Dr. Prakash and coworkers to hypothesize that patients with GERD might have an increased incidence of MI.
It’s only relatively recently that several other common chronic diseases have been reconceptualized as systemic inflammatory diseases associated with increased cardiovascular risk. Diabetes, for example, has been repositioned from a straightforward endocrine disease to new status as a coronary heart disease equivalent. And strong bodies of evidence link psoriasis and periodontitis to increased cardiovascular risk, Dr. Prakash noted.
Audience member Dr. Nimish B. Vakil, a Milwaukee gastroenterologist, rose to caution about the possibility of residual confounding by other factors not accounted for in the multivariate analysis, and thus the need for confirmatory studies. That being said, he added that he finds the notion of a GERD/acute MI link mechanistically quite plausible. Acid exposure from GERD might very well trigger exaggerated firing of the esophagocardiac reflex, with resultant episodes of coronary hypoperfusion. Effective PPI therapy for GERD would be expected to reduce such events.
Dr. Prakash said his literature search suggested another possible mechanism for the apparent protective effect of PPIs against MI: effective treatment of GERD, whether using PPIs or via a fundoplication procedure, has been reported to reduce levels of the inflammatory cytokines present in GERD.
Dr. Prakash reported having no financial conflicts with regard to his study, which was supported by institutional funds.
CHICAGO – Gastroesophageal reflux disease may constitute a heretofore unrecognized risk factor for coronary heart disease.
In a nationwide case-control study of prodigious proportions, endoscopically confirmed GERD in patients without known coronary or peripheral artery disease at baseline was independently associated with a 57% increased risk of having a first acute MI within the next 5 years, Dr. Ravi K. Prakash reported at the annual Digestive Disease Week.
If this novel finding is confirmed in other databases, the clinical implications would be profound. GERD is after all an extremely common problem, affecting 30%-40% of the U.S. population, noted Dr. Prakash, a gastroenterology fellow at MetroHealth Medical Center and Case Western Reserve University, Cleveland.
The good news: When Dr. Prakash and his coinvestigators divided the 316,390 study participants with GERD into those who were on proton pump inhibitor therapy and those who weren’t, they found that the PPI users weren’t at increased MI risk, compared with the more than 13.6 million similarly aged control subjects who didn’t have GERD, had never undergone an upper endoscopy, and were free of known atherosclerotic disease at baseline.
"Effective treatment of GERD appears to protect against MI," he concluded.
The study population was drawn from Explorys, a private, cloud-based health database containing the electronic health records of 35 million U.S. patients as provided by more than 200,000 physicians in 300-plus health care systems.
A first MI occurred during follow-up in 18,860 GERD patients, or 5.96%, compared with 144,140 controls, or 1.05%. This translates into an unadjusted sixfold increased risk of acute MI in patients with rigorously diagnosed GERD, compared with GERD-free controls.
The investigators then performed a multivariate logistic regression analysis adjusted for six major cardiovascular risk factors: obesity, hypertension, diabetes, tobacco use, hyperlipidemia, and sex. Many of these risk factors were substantially more prevalent in the GERD population. As a result, the unadjusted sixfold increase in MI risk associated with GERD was attenuated in this adjusted risk model; however, the adjusted 57% increased risk remained highly significant.
Moreover, the increased relative risk of GERD seen in the multivariate analysis was comparable with the risks posed by many of the established risk factors. Obesity, for example, showed a 49% increase in MI risk, smoking a 90% increase, and hypertension a 10% increased relative risk, compared with normotension. Only hyperlipidemia and diabetes were in another league, with relative risks of 9.5- and 2.2-fold, respectively, Dr. Prakash continued.
He observed that during the past decade, the research focus has shifted away from GERD as a caustic disease process causing local injury to GERD as a systemic inflammatory disease. Among the proinflammatory cytokines shown to be elevated both in the esophagus and circulation of GERD patients are interleukins-6, -8, and -1B as well as platelet-activating factor. These cytokines are well established as important players in the formation of atherosclerotic plaque in arteries. It was the shared elevations in proinflammatory cytokines that led Dr. Prakash and coworkers to hypothesize that patients with GERD might have an increased incidence of MI.
It’s only relatively recently that several other common chronic diseases have been reconceptualized as systemic inflammatory diseases associated with increased cardiovascular risk. Diabetes, for example, has been repositioned from a straightforward endocrine disease to new status as a coronary heart disease equivalent. And strong bodies of evidence link psoriasis and periodontitis to increased cardiovascular risk, Dr. Prakash noted.
Audience member Dr. Nimish B. Vakil, a Milwaukee gastroenterologist, rose to caution about the possibility of residual confounding by other factors not accounted for in the multivariate analysis, and thus the need for confirmatory studies. That being said, he added that he finds the notion of a GERD/acute MI link mechanistically quite plausible. Acid exposure from GERD might very well trigger exaggerated firing of the esophagocardiac reflex, with resultant episodes of coronary hypoperfusion. Effective PPI therapy for GERD would be expected to reduce such events.
Dr. Prakash said his literature search suggested another possible mechanism for the apparent protective effect of PPIs against MI: effective treatment of GERD, whether using PPIs or via a fundoplication procedure, has been reported to reduce levels of the inflammatory cytokines present in GERD.
Dr. Prakash reported having no financial conflicts with regard to his study, which was supported by institutional funds.
AT DDW 2014
Major finding: GERD was found to be independently associated with a 57% increased risk of acute MI.
Data source: This was a case-control study that harnessed the huge national Explorys database to compare first MI rates in 316,390 subjects with confirmed GERD to more than 13.6 million controls without GERD who’d never had an upper endoscopy.
Disclosures: This study was supported by institutional funds. The presenter reported having no financial conflicts.
VIDEO: GERD may increase heart attack risk
CHICAGO – Gastroesophageal reflux disease may constitute a heretofore unrecognized risk factor for coronary heart disease.
In a nationwide case-control study of prodigious proportions, endoscopically confirmed GERD in patients without known coronary or peripheral artery disease at baseline was independently associated with a 57% increased risk of having a first acute MI within the next 5 years, Dr. Ravi K. Prakash reported.
In a video interview at the annual Digestive Disease Week, Dr. Prakash of MetroHealth Medical Center, Cleveland, discusses the clinical implications of the study findings.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Gastroesophageal reflux disease may constitute a heretofore unrecognized risk factor for coronary heart disease.
In a nationwide case-control study of prodigious proportions, endoscopically confirmed GERD in patients without known coronary or peripheral artery disease at baseline was independently associated with a 57% increased risk of having a first acute MI within the next 5 years, Dr. Ravi K. Prakash reported.
In a video interview at the annual Digestive Disease Week, Dr. Prakash of MetroHealth Medical Center, Cleveland, discusses the clinical implications of the study findings.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Gastroesophageal reflux disease may constitute a heretofore unrecognized risk factor for coronary heart disease.
In a nationwide case-control study of prodigious proportions, endoscopically confirmed GERD in patients without known coronary or peripheral artery disease at baseline was independently associated with a 57% increased risk of having a first acute MI within the next 5 years, Dr. Ravi K. Prakash reported.
In a video interview at the annual Digestive Disease Week, Dr. Prakash of MetroHealth Medical Center, Cleveland, discusses the clinical implications of the study findings.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT DDW 2014
Halve Acne Lesion Counts in One Month
WAIKOLOA, HAWAII – The fixed-dose combination of adapalene/benzoyl peroxide gel 0.1%/2.5% typically results in reductions of 40%-50% in inflammatory and 30%-40% in noninflammatory acne lesion counts during the first 4 weeks of therapy.
In a pooled analysis of 14 studies totaling 2,358 acne patients aged 9-61 years, the proportion with an Investigator’s Global Assessment (IGA) of moderate or severe acne dropped from 92% at baseline to 51% at 4 weeks, according to Dr. Linda Stein Gold.
The pooled analysis was carried out to provide physicians and patients with information as to what to realistically expect in the first 4 weeks of therapy. The analysis is particularly timely in light of the Food and Drug Administration’s recent expansion of the indication for adapalene/benzoyl peroxide gel 0.1%/2.5% (Epiduo) to include acne patients as young as 9 years of age, noted Dr. Stein Gold, director of clinical research in the department of dermatology at Henry Ford Hospital, Detroit.
Mild skin irritation was common, especially in the first 2 weeks of use.
"It’s a fiction that retinoids are too harsh for younger patients to use," Dr. Stein Gold said. "I tell all my acne patients, especially the younger ones, no matter what topical retinoid they’re using, to use it every other night for the first 2 weeks, make sure their skin is completely dry, and use a gentle cleanser and a good moisturizer," she said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation. "If they get through those first 2 weeks, they’ll see that the tolerability really improves."
Dr. Stein Gold said she likes the fixed combination of adapalene/benzoyl peroxide gel 0.1%/2.5% because it combines several key elements of cutting-edge acne treatment. Topical retinoids are not mere comedone busters, as formerly thought, but are also effective agents for papules and pustular lesions. While they do not kill Propionibacterium acnes, they down-regulate Toll-like receptor 2, which is produced by the bacterium and induces proinflammatory cytokines. Topical retinoids are a key part of maintenance-of-remission strategies. And adapalene is unique among topical retinoids in that it is inherently stable with benzoyl peroxide (BPO) and is stable in daylight.
BPO is unique in that it has potent antibacterial activity, but never causes P. acnes resistance, even after many years of treatment. BPO is quite effective for inflammatory lesions and moderately effective for comedonal acne lesions, Dr. Stein Gold noted. Also, it is well established that BPO at a concentration of 2.5% is as efficacious as 5% or 10%, and much better tolerated than at the higher concentrations. The molecule size and the use of a vehicle with good penetration into the hair follicle are much more important factors in treatment effectiveness than is the BPO concentration, she added.
In one study of acne patients with antibiotic-resistant P. acnes, including clindamycin-, doxycycline-, erythromycin-, and minocycline-resistant microorganisms, 88% of the antibiotic-resistant P. acnes bacteria were killed after 2 weeks of treatment with adapalene/BPO gel 0.1%/2.5%. After 4 weeks of treatment, 97% of the antibiotic-resistant organisms were dead. That’s testimony to the P. acnes–killing power of BPO, said Dr. Stein Gold.
"There’s a sense among many that with all the newer medications we have for acne, benzoyl peroxide is really your grandfather’s treatment, with no place in today’s modern world. This is totally false. I really feel that benzoyl peroxide should play a central role in all of our acne patients’ treatment regimens, unless of course they’re allergic to it, which occurs in only a small percentage of our patients," she said.
The pooled analysis was funded by Galderma. Dr. Stein Gold is a consultant to Galderma, Stiefel, Medicis, and Warner Chilcott.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The fixed-dose combination of adapalene/benzoyl peroxide gel 0.1%/2.5% typically results in reductions of 40%-50% in inflammatory and 30%-40% in noninflammatory acne lesion counts during the first 4 weeks of therapy.
In a pooled analysis of 14 studies totaling 2,358 acne patients aged 9-61 years, the proportion with an Investigator’s Global Assessment (IGA) of moderate or severe acne dropped from 92% at baseline to 51% at 4 weeks, according to Dr. Linda Stein Gold.
The pooled analysis was carried out to provide physicians and patients with information as to what to realistically expect in the first 4 weeks of therapy. The analysis is particularly timely in light of the Food and Drug Administration’s recent expansion of the indication for adapalene/benzoyl peroxide gel 0.1%/2.5% (Epiduo) to include acne patients as young as 9 years of age, noted Dr. Stein Gold, director of clinical research in the department of dermatology at Henry Ford Hospital, Detroit.
Mild skin irritation was common, especially in the first 2 weeks of use.
"It’s a fiction that retinoids are too harsh for younger patients to use," Dr. Stein Gold said. "I tell all my acne patients, especially the younger ones, no matter what topical retinoid they’re using, to use it every other night for the first 2 weeks, make sure their skin is completely dry, and use a gentle cleanser and a good moisturizer," she said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation. "If they get through those first 2 weeks, they’ll see that the tolerability really improves."
Dr. Stein Gold said she likes the fixed combination of adapalene/benzoyl peroxide gel 0.1%/2.5% because it combines several key elements of cutting-edge acne treatment. Topical retinoids are not mere comedone busters, as formerly thought, but are also effective agents for papules and pustular lesions. While they do not kill Propionibacterium acnes, they down-regulate Toll-like receptor 2, which is produced by the bacterium and induces proinflammatory cytokines. Topical retinoids are a key part of maintenance-of-remission strategies. And adapalene is unique among topical retinoids in that it is inherently stable with benzoyl peroxide (BPO) and is stable in daylight.
BPO is unique in that it has potent antibacterial activity, but never causes P. acnes resistance, even after many years of treatment. BPO is quite effective for inflammatory lesions and moderately effective for comedonal acne lesions, Dr. Stein Gold noted. Also, it is well established that BPO at a concentration of 2.5% is as efficacious as 5% or 10%, and much better tolerated than at the higher concentrations. The molecule size and the use of a vehicle with good penetration into the hair follicle are much more important factors in treatment effectiveness than is the BPO concentration, she added.
In one study of acne patients with antibiotic-resistant P. acnes, including clindamycin-, doxycycline-, erythromycin-, and minocycline-resistant microorganisms, 88% of the antibiotic-resistant P. acnes bacteria were killed after 2 weeks of treatment with adapalene/BPO gel 0.1%/2.5%. After 4 weeks of treatment, 97% of the antibiotic-resistant organisms were dead. That’s testimony to the P. acnes–killing power of BPO, said Dr. Stein Gold.
"There’s a sense among many that with all the newer medications we have for acne, benzoyl peroxide is really your grandfather’s treatment, with no place in today’s modern world. This is totally false. I really feel that benzoyl peroxide should play a central role in all of our acne patients’ treatment regimens, unless of course they’re allergic to it, which occurs in only a small percentage of our patients," she said.
The pooled analysis was funded by Galderma. Dr. Stein Gold is a consultant to Galderma, Stiefel, Medicis, and Warner Chilcott.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The fixed-dose combination of adapalene/benzoyl peroxide gel 0.1%/2.5% typically results in reductions of 40%-50% in inflammatory and 30%-40% in noninflammatory acne lesion counts during the first 4 weeks of therapy.
In a pooled analysis of 14 studies totaling 2,358 acne patients aged 9-61 years, the proportion with an Investigator’s Global Assessment (IGA) of moderate or severe acne dropped from 92% at baseline to 51% at 4 weeks, according to Dr. Linda Stein Gold.
The pooled analysis was carried out to provide physicians and patients with information as to what to realistically expect in the first 4 weeks of therapy. The analysis is particularly timely in light of the Food and Drug Administration’s recent expansion of the indication for adapalene/benzoyl peroxide gel 0.1%/2.5% (Epiduo) to include acne patients as young as 9 years of age, noted Dr. Stein Gold, director of clinical research in the department of dermatology at Henry Ford Hospital, Detroit.
Mild skin irritation was common, especially in the first 2 weeks of use.
"It’s a fiction that retinoids are too harsh for younger patients to use," Dr. Stein Gold said. "I tell all my acne patients, especially the younger ones, no matter what topical retinoid they’re using, to use it every other night for the first 2 weeks, make sure their skin is completely dry, and use a gentle cleanser and a good moisturizer," she said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation. "If they get through those first 2 weeks, they’ll see that the tolerability really improves."
Dr. Stein Gold said she likes the fixed combination of adapalene/benzoyl peroxide gel 0.1%/2.5% because it combines several key elements of cutting-edge acne treatment. Topical retinoids are not mere comedone busters, as formerly thought, but are also effective agents for papules and pustular lesions. While they do not kill Propionibacterium acnes, they down-regulate Toll-like receptor 2, which is produced by the bacterium and induces proinflammatory cytokines. Topical retinoids are a key part of maintenance-of-remission strategies. And adapalene is unique among topical retinoids in that it is inherently stable with benzoyl peroxide (BPO) and is stable in daylight.
BPO is unique in that it has potent antibacterial activity, but never causes P. acnes resistance, even after many years of treatment. BPO is quite effective for inflammatory lesions and moderately effective for comedonal acne lesions, Dr. Stein Gold noted. Also, it is well established that BPO at a concentration of 2.5% is as efficacious as 5% or 10%, and much better tolerated than at the higher concentrations. The molecule size and the use of a vehicle with good penetration into the hair follicle are much more important factors in treatment effectiveness than is the BPO concentration, she added.
In one study of acne patients with antibiotic-resistant P. acnes, including clindamycin-, doxycycline-, erythromycin-, and minocycline-resistant microorganisms, 88% of the antibiotic-resistant P. acnes bacteria were killed after 2 weeks of treatment with adapalene/BPO gel 0.1%/2.5%. After 4 weeks of treatment, 97% of the antibiotic-resistant organisms were dead. That’s testimony to the P. acnes–killing power of BPO, said Dr. Stein Gold.
"There’s a sense among many that with all the newer medications we have for acne, benzoyl peroxide is really your grandfather’s treatment, with no place in today’s modern world. This is totally false. I really feel that benzoyl peroxide should play a central role in all of our acne patients’ treatment regimens, unless of course they’re allergic to it, which occurs in only a small percentage of our patients," she said.
The pooled analysis was funded by Galderma. Dr. Stein Gold is a consultant to Galderma, Stiefel, Medicis, and Warner Chilcott.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR