User login
Obese teens heading for bariatric surgery already show kidney damage
LAS VEGAS – Seventeen percent of severely obese adolescents slated for bariatric surgery in the Teen-Longitudinal Assessment of Bariatric Surgery study already had micro- or macroalbuminuria.
At a meeting sponsored by the National Kidney Foundation, Dr. Nianzhou Xiao said that future reports from the ongoing Teen-LABS study will provide the answer to a critical question: Is this worrisome loss of kidney function so early in life reversible via surgical weight loss?
Dr. Xiao presented a cross-sectional baseline report on 242 severely obese adolescents with a median body mass index of 50.5 kg/m2. Fourteen percent had microalbuminuria and another 3.1% had macroalbuminuria. Although the group’s mean estimated glomerular filtration rate was 107.6 mL/min per 1.73 m2, 3% of the teens had an eGFR below 60 mL/min 1.73 m2, which is the definition of stage 3 chronic kidney disease.
In addition, 45% of the teens were hypertensive before surgery, 74% were dyslipidemic, and 13.6% had diabetes. The group’s median serum ferritin was 37 mcg/L, noted Dr. Xiao of Cincinnati Children’s Hospital Medical Center.
Multivariate analysis identified two independent risk factors for an elevated albumin to creatinine ratio: female gender, with an associated 2.34-fold increased risk, and elevated serum ferritin. For every 10 mcg/L of ferritin, the likelihood of an elevated albumin-to-creatinine ratio rose by 7%.
An estimated 4%-6% of U.S. children and adolescents are severely obese, defined as a body mass index of 35 kg/m2 or more or a body weight above the 120th percentile. The ongoing Teen-LABS study is the most comprehensive examination of kidney status in severely obese adolescents undergoing bariatric surgery.
Teen-LABS is funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Xiao reported having no financial conflicts.
LAS VEGAS – Seventeen percent of severely obese adolescents slated for bariatric surgery in the Teen-Longitudinal Assessment of Bariatric Surgery study already had micro- or macroalbuminuria.
At a meeting sponsored by the National Kidney Foundation, Dr. Nianzhou Xiao said that future reports from the ongoing Teen-LABS study will provide the answer to a critical question: Is this worrisome loss of kidney function so early in life reversible via surgical weight loss?
Dr. Xiao presented a cross-sectional baseline report on 242 severely obese adolescents with a median body mass index of 50.5 kg/m2. Fourteen percent had microalbuminuria and another 3.1% had macroalbuminuria. Although the group’s mean estimated glomerular filtration rate was 107.6 mL/min per 1.73 m2, 3% of the teens had an eGFR below 60 mL/min 1.73 m2, which is the definition of stage 3 chronic kidney disease.
In addition, 45% of the teens were hypertensive before surgery, 74% were dyslipidemic, and 13.6% had diabetes. The group’s median serum ferritin was 37 mcg/L, noted Dr. Xiao of Cincinnati Children’s Hospital Medical Center.
Multivariate analysis identified two independent risk factors for an elevated albumin to creatinine ratio: female gender, with an associated 2.34-fold increased risk, and elevated serum ferritin. For every 10 mcg/L of ferritin, the likelihood of an elevated albumin-to-creatinine ratio rose by 7%.
An estimated 4%-6% of U.S. children and adolescents are severely obese, defined as a body mass index of 35 kg/m2 or more or a body weight above the 120th percentile. The ongoing Teen-LABS study is the most comprehensive examination of kidney status in severely obese adolescents undergoing bariatric surgery.
Teen-LABS is funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Xiao reported having no financial conflicts.
LAS VEGAS – Seventeen percent of severely obese adolescents slated for bariatric surgery in the Teen-Longitudinal Assessment of Bariatric Surgery study already had micro- or macroalbuminuria.
At a meeting sponsored by the National Kidney Foundation, Dr. Nianzhou Xiao said that future reports from the ongoing Teen-LABS study will provide the answer to a critical question: Is this worrisome loss of kidney function so early in life reversible via surgical weight loss?
Dr. Xiao presented a cross-sectional baseline report on 242 severely obese adolescents with a median body mass index of 50.5 kg/m2. Fourteen percent had microalbuminuria and another 3.1% had macroalbuminuria. Although the group’s mean estimated glomerular filtration rate was 107.6 mL/min per 1.73 m2, 3% of the teens had an eGFR below 60 mL/min 1.73 m2, which is the definition of stage 3 chronic kidney disease.
In addition, 45% of the teens were hypertensive before surgery, 74% were dyslipidemic, and 13.6% had diabetes. The group’s median serum ferritin was 37 mcg/L, noted Dr. Xiao of Cincinnati Children’s Hospital Medical Center.
Multivariate analysis identified two independent risk factors for an elevated albumin to creatinine ratio: female gender, with an associated 2.34-fold increased risk, and elevated serum ferritin. For every 10 mcg/L of ferritin, the likelihood of an elevated albumin-to-creatinine ratio rose by 7%.
An estimated 4%-6% of U.S. children and adolescents are severely obese, defined as a body mass index of 35 kg/m2 or more or a body weight above the 120th percentile. The ongoing Teen-LABS study is the most comprehensive examination of kidney status in severely obese adolescents undergoing bariatric surgery.
Teen-LABS is funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Xiao reported having no financial conflicts.
AT SCM 14
Key clinical point: Follow-up will determine if baseline abnormalities in obese teens’ kidney function and other areas are reversible through weight-loss surgery.
Major finding: 14% of a large group of severely obese adolescents scheduled for bariatric surgery already had microalbuminuria and another 3.1% had macroalbuminuria. Three percent had stage 3 chronic kidney disease.
Data source: A cross-sectional analysis involving 242 severely obese adolescents in the ongoing Teen-Longitudinal Assessment of Bariatric Surgery (Teen-LABS) study.
Disclosures: The ongoing study is funded by the National Institute of Diabetes and Digestive and Kidney Diseases.
Eccentric exercise tamed elevated liver enzymes
WASHINGTON – Eccentric exercise in the form of downhill walking is a promising new means of reducing elevated liver enzyme levels in overweight and obese sedentary individuals.
This finding suggests this form of exercise may have a role as a therapeutic intervention in patients with nonalcoholic fatty liver disease, a condition which is rapidly growing in prevalence and which is associated with increased risks of diabetes and cardiovascular events, Dr. Christoph Saely observed at the annual meeting of the American College of Cardiology.
It’s known that physical exercise can be effective in lowering elevated hepatic enzymes, but it’s equally clear that conventional forms of exercise such as stair-climbing, jogging, and brisk walking are too vigorous and demanding for many obese sedentary patients. They’re unable to get with the program.
That’s where eccentric training holds particular value, according to Dr. Saely of the Academic Teaching Hospital of Feldkirch, Austria.
Eccentric exercise occurs when active muscle contraction occurs simultaneously with the muscle’s lengthening. Thus, it involves active resistance to stretching. During a biceps curl, for example, the eccentric muscle work occurs as the weight is being lowered, while concentric muscle training happens as the weight is being raised. Similarly, walking uphill involves concentric muscle contraction, while walking downhill emphasizes eccentric muscle action.
Eccentric exercise training creates more force yet uses less energy. It protects joints from damage. Physical therapists and fitness trainers find eccentric exercise regimens are particularly useful in the elderly, infirm, and sedentary because such individuals find it less exhausting and they are more willing to participate.
Dr. Saely and his coinvestigators have an ideal outdoor laboratory in which to conduct a pilot study of the effects of eccentric exercise because their hospital is located in the Austrian Alps. Subjects can hike downhill, then take a cable car back up, with adherence to the program monitored electronically at the vehicle entrance.
He reported on 42 overweight or obese sedentary patients with elevated liver enzymes and 12 matched controls who were assigned to hike down a predefined route in the Alps three to five times per week for 8 weeks. The course featured a 540-meter drop in altitude.
Mean serum alanine aminotransferase levels in the overweight group dropped from 36 IU/L at baseline to 31 IU/L at 8 weeks. Serum gamma-glutamyl transferase fell from 56 IU/L to 44 IU/L. And the alanine aminotransferase/aspartate aminotransferase ratio improved from 1.22 IU/L to 1.02 IU/L. Meanwhile, liver enzyme levels remained unchanged over time in the controls.
The eccentric exercise regimen was well tolerated, participation was high, and there were no serious adverse events, according to Dr. Saely.
While not everyone is fortunate enough to have a mountain in the backyard with a gondola for the return trip, knowledgeable fitness instructors can demonstrate eccentric exercises using weights and machines that target large muscle groups.
Dr. Saely reported having no conflicts of interest regarding this study.
WASHINGTON – Eccentric exercise in the form of downhill walking is a promising new means of reducing elevated liver enzyme levels in overweight and obese sedentary individuals.
This finding suggests this form of exercise may have a role as a therapeutic intervention in patients with nonalcoholic fatty liver disease, a condition which is rapidly growing in prevalence and which is associated with increased risks of diabetes and cardiovascular events, Dr. Christoph Saely observed at the annual meeting of the American College of Cardiology.
It’s known that physical exercise can be effective in lowering elevated hepatic enzymes, but it’s equally clear that conventional forms of exercise such as stair-climbing, jogging, and brisk walking are too vigorous and demanding for many obese sedentary patients. They’re unable to get with the program.
That’s where eccentric training holds particular value, according to Dr. Saely of the Academic Teaching Hospital of Feldkirch, Austria.
Eccentric exercise occurs when active muscle contraction occurs simultaneously with the muscle’s lengthening. Thus, it involves active resistance to stretching. During a biceps curl, for example, the eccentric muscle work occurs as the weight is being lowered, while concentric muscle training happens as the weight is being raised. Similarly, walking uphill involves concentric muscle contraction, while walking downhill emphasizes eccentric muscle action.
Eccentric exercise training creates more force yet uses less energy. It protects joints from damage. Physical therapists and fitness trainers find eccentric exercise regimens are particularly useful in the elderly, infirm, and sedentary because such individuals find it less exhausting and they are more willing to participate.
Dr. Saely and his coinvestigators have an ideal outdoor laboratory in which to conduct a pilot study of the effects of eccentric exercise because their hospital is located in the Austrian Alps. Subjects can hike downhill, then take a cable car back up, with adherence to the program monitored electronically at the vehicle entrance.
He reported on 42 overweight or obese sedentary patients with elevated liver enzymes and 12 matched controls who were assigned to hike down a predefined route in the Alps three to five times per week for 8 weeks. The course featured a 540-meter drop in altitude.
Mean serum alanine aminotransferase levels in the overweight group dropped from 36 IU/L at baseline to 31 IU/L at 8 weeks. Serum gamma-glutamyl transferase fell from 56 IU/L to 44 IU/L. And the alanine aminotransferase/aspartate aminotransferase ratio improved from 1.22 IU/L to 1.02 IU/L. Meanwhile, liver enzyme levels remained unchanged over time in the controls.
The eccentric exercise regimen was well tolerated, participation was high, and there were no serious adverse events, according to Dr. Saely.
While not everyone is fortunate enough to have a mountain in the backyard with a gondola for the return trip, knowledgeable fitness instructors can demonstrate eccentric exercises using weights and machines that target large muscle groups.
Dr. Saely reported having no conflicts of interest regarding this study.
WASHINGTON – Eccentric exercise in the form of downhill walking is a promising new means of reducing elevated liver enzyme levels in overweight and obese sedentary individuals.
This finding suggests this form of exercise may have a role as a therapeutic intervention in patients with nonalcoholic fatty liver disease, a condition which is rapidly growing in prevalence and which is associated with increased risks of diabetes and cardiovascular events, Dr. Christoph Saely observed at the annual meeting of the American College of Cardiology.
It’s known that physical exercise can be effective in lowering elevated hepatic enzymes, but it’s equally clear that conventional forms of exercise such as stair-climbing, jogging, and brisk walking are too vigorous and demanding for many obese sedentary patients. They’re unable to get with the program.
That’s where eccentric training holds particular value, according to Dr. Saely of the Academic Teaching Hospital of Feldkirch, Austria.
Eccentric exercise occurs when active muscle contraction occurs simultaneously with the muscle’s lengthening. Thus, it involves active resistance to stretching. During a biceps curl, for example, the eccentric muscle work occurs as the weight is being lowered, while concentric muscle training happens as the weight is being raised. Similarly, walking uphill involves concentric muscle contraction, while walking downhill emphasizes eccentric muscle action.
Eccentric exercise training creates more force yet uses less energy. It protects joints from damage. Physical therapists and fitness trainers find eccentric exercise regimens are particularly useful in the elderly, infirm, and sedentary because such individuals find it less exhausting and they are more willing to participate.
Dr. Saely and his coinvestigators have an ideal outdoor laboratory in which to conduct a pilot study of the effects of eccentric exercise because their hospital is located in the Austrian Alps. Subjects can hike downhill, then take a cable car back up, with adherence to the program monitored electronically at the vehicle entrance.
He reported on 42 overweight or obese sedentary patients with elevated liver enzymes and 12 matched controls who were assigned to hike down a predefined route in the Alps three to five times per week for 8 weeks. The course featured a 540-meter drop in altitude.
Mean serum alanine aminotransferase levels in the overweight group dropped from 36 IU/L at baseline to 31 IU/L at 8 weeks. Serum gamma-glutamyl transferase fell from 56 IU/L to 44 IU/L. And the alanine aminotransferase/aspartate aminotransferase ratio improved from 1.22 IU/L to 1.02 IU/L. Meanwhile, liver enzyme levels remained unchanged over time in the controls.
The eccentric exercise regimen was well tolerated, participation was high, and there were no serious adverse events, according to Dr. Saely.
While not everyone is fortunate enough to have a mountain in the backyard with a gondola for the return trip, knowledgeable fitness instructors can demonstrate eccentric exercises using weights and machines that target large muscle groups.
Dr. Saely reported having no conflicts of interest regarding this study.
AT ACC 14
Major finding: An exercise regimen of regular downhill walking for 8 weeks lowered serum alanine aminotransferase levels in overweight and obese sedentary patients from a mean baseline of 36 IU/L to 31 IU/L.
Data source: Pilot study of 42 overweight or obese sedentary patients with elevated liver enzyme levels and 12 matched controls who participated in an eccentric muscle contraction regimen involving downhill hiking 3-5 times per week for 8 weeks.
Disclosures: The presenter reported having no financial conflicts. The study was conducted free of commercial support.
PEARL-II: Oral ‘3D’ regimen effective in hepatitis C with prior treatment failure
CHICAGO – The investigational "3D" combination of oral antiviral drugs achieved a cure rate that was nothing short of sensational in the phase III PEARL-II trial involving noncirrhotic patients with hepatitis C genotype 1b who had previously failed pegylated interferon–based therapy.
Moreover, these results were achieved without the use of daily oral ribavirin, formerly a standard part of hepatitis C therapy, and one responsible for a substantial burden of side effects.
Genotype 1b is the most prevalent form of hepatitis C disease worldwide. While it is especially common in Europe and Japan, genotype 1b also accounts for about one-third of cases of HCV in the United States.
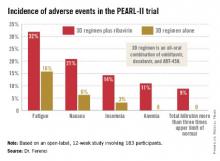
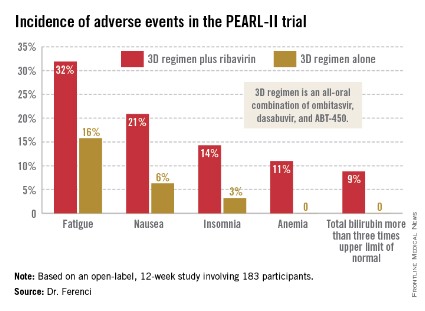
The so-called 3D regimen is an all-oral combination of three direct-acting antiviral agents: ombitasvir, dasabuvir, and ABT-450. Each of these agents inhibits the viral life cycle at a different point, Dr. Peter Ferenci explained in presenting the PEARL-II results at the annual Digestive Disease Week.
PEARL-II was an open-label, 12-week trial which asked the question, what added benefit does daily oral ribavirin provide in conjunction with 3D therapy in noncirrhotic HCV genotype 1b patients previously treated unsuccessfully with a pegylated interferon/ribavirin regimen? The answer, as it turns out, is none.
The 186 participants were randomized to the 3D regimen with or without oral ribavirin. The oral 3D regimen consists of ABT-450 (150 mg)/ritonavir (100 mg)/ombitasvir (25 mg), formulated as a single once-daily pill plus dasabuvir at 250 mg twice daily.
The primary endpoint – a sustained virologic response 12 weeks after completion of the 12-week course of treatment – was achieved in 96.6% of the ribavirin group and 100% who didn’t receive ribavirin. Both rates are superior to the historical sustained virologic response rate seen in studies of the former state-of-the-art regimen of telaprevir plus pegylated interferon and ribavirin, noted Dr. Ferenci of the Medical University of Vienna.
There were no virologic failures in PEARL-II. Two patients stopped the study drugs: one who developed pancreatitis deemed unrelated to treatment and another with anxiety, tachycardia, shortness of breath, and fever considered possibly treatment related.
Results of 3D therapy were equally good in men and women and in former relapsers, nonresponders, and partial responders to pegylated interferon-based regimens.
Treatment-related adverse events were more frequent in the ribavirin study arm (see graphic), but were generally mild and manageable.
Asked if in light of the impressive study results he thought physicians should hold off on treating PEARL-II–type patients until the 3D regimen receives marketing approval, Dr. Ferenci replied, "I think if you really need to treat a patient now, you have to use what exists. But I think in half a year your options will change."
PEARL-II was a companion trial to PEARL-III, conducted in previously untreated patients with HCV 1b infection (N. Engl. J. Med. 2014 [doi:10.1056/NEJMoa1402338]). Both studies were sponsored by AbbVie. Dr. Ferenci reported serving on advisory panels for roughly a dozen pharmaceutical companies.
daily oral ribavirin, formerly a standard part of hepatitis C therapy, Europe and Japan, HCV, ombitasvir, dasabuvir, ABT-450, Dr. Peter Ferenci, annual Digestive Disease Week,
CHICAGO – The investigational "3D" combination of oral antiviral drugs achieved a cure rate that was nothing short of sensational in the phase III PEARL-II trial involving noncirrhotic patients with hepatitis C genotype 1b who had previously failed pegylated interferon–based therapy.
Moreover, these results were achieved without the use of daily oral ribavirin, formerly a standard part of hepatitis C therapy, and one responsible for a substantial burden of side effects.
Genotype 1b is the most prevalent form of hepatitis C disease worldwide. While it is especially common in Europe and Japan, genotype 1b also accounts for about one-third of cases of HCV in the United States.
The so-called 3D regimen is an all-oral combination of three direct-acting antiviral agents: ombitasvir, dasabuvir, and ABT-450. Each of these agents inhibits the viral life cycle at a different point, Dr. Peter Ferenci explained in presenting the PEARL-II results at the annual Digestive Disease Week.
PEARL-II was an open-label, 12-week trial which asked the question, what added benefit does daily oral ribavirin provide in conjunction with 3D therapy in noncirrhotic HCV genotype 1b patients previously treated unsuccessfully with a pegylated interferon/ribavirin regimen? The answer, as it turns out, is none.
The 186 participants were randomized to the 3D regimen with or without oral ribavirin. The oral 3D regimen consists of ABT-450 (150 mg)/ritonavir (100 mg)/ombitasvir (25 mg), formulated as a single once-daily pill plus dasabuvir at 250 mg twice daily.
The primary endpoint – a sustained virologic response 12 weeks after completion of the 12-week course of treatment – was achieved in 96.6% of the ribavirin group and 100% who didn’t receive ribavirin. Both rates are superior to the historical sustained virologic response rate seen in studies of the former state-of-the-art regimen of telaprevir plus pegylated interferon and ribavirin, noted Dr. Ferenci of the Medical University of Vienna.
There were no virologic failures in PEARL-II. Two patients stopped the study drugs: one who developed pancreatitis deemed unrelated to treatment and another with anxiety, tachycardia, shortness of breath, and fever considered possibly treatment related.
Results of 3D therapy were equally good in men and women and in former relapsers, nonresponders, and partial responders to pegylated interferon-based regimens.
Treatment-related adverse events were more frequent in the ribavirin study arm (see graphic), but were generally mild and manageable.
Asked if in light of the impressive study results he thought physicians should hold off on treating PEARL-II–type patients until the 3D regimen receives marketing approval, Dr. Ferenci replied, "I think if you really need to treat a patient now, you have to use what exists. But I think in half a year your options will change."
PEARL-II was a companion trial to PEARL-III, conducted in previously untreated patients with HCV 1b infection (N. Engl. J. Med. 2014 [doi:10.1056/NEJMoa1402338]). Both studies were sponsored by AbbVie. Dr. Ferenci reported serving on advisory panels for roughly a dozen pharmaceutical companies.
CHICAGO – The investigational "3D" combination of oral antiviral drugs achieved a cure rate that was nothing short of sensational in the phase III PEARL-II trial involving noncirrhotic patients with hepatitis C genotype 1b who had previously failed pegylated interferon–based therapy.
Moreover, these results were achieved without the use of daily oral ribavirin, formerly a standard part of hepatitis C therapy, and one responsible for a substantial burden of side effects.
Genotype 1b is the most prevalent form of hepatitis C disease worldwide. While it is especially common in Europe and Japan, genotype 1b also accounts for about one-third of cases of HCV in the United States.
The so-called 3D regimen is an all-oral combination of three direct-acting antiviral agents: ombitasvir, dasabuvir, and ABT-450. Each of these agents inhibits the viral life cycle at a different point, Dr. Peter Ferenci explained in presenting the PEARL-II results at the annual Digestive Disease Week.
PEARL-II was an open-label, 12-week trial which asked the question, what added benefit does daily oral ribavirin provide in conjunction with 3D therapy in noncirrhotic HCV genotype 1b patients previously treated unsuccessfully with a pegylated interferon/ribavirin regimen? The answer, as it turns out, is none.
The 186 participants were randomized to the 3D regimen with or without oral ribavirin. The oral 3D regimen consists of ABT-450 (150 mg)/ritonavir (100 mg)/ombitasvir (25 mg), formulated as a single once-daily pill plus dasabuvir at 250 mg twice daily.
The primary endpoint – a sustained virologic response 12 weeks after completion of the 12-week course of treatment – was achieved in 96.6% of the ribavirin group and 100% who didn’t receive ribavirin. Both rates are superior to the historical sustained virologic response rate seen in studies of the former state-of-the-art regimen of telaprevir plus pegylated interferon and ribavirin, noted Dr. Ferenci of the Medical University of Vienna.
There were no virologic failures in PEARL-II. Two patients stopped the study drugs: one who developed pancreatitis deemed unrelated to treatment and another with anxiety, tachycardia, shortness of breath, and fever considered possibly treatment related.
Results of 3D therapy were equally good in men and women and in former relapsers, nonresponders, and partial responders to pegylated interferon-based regimens.
Treatment-related adverse events were more frequent in the ribavirin study arm (see graphic), but were generally mild and manageable.
Asked if in light of the impressive study results he thought physicians should hold off on treating PEARL-II–type patients until the 3D regimen receives marketing approval, Dr. Ferenci replied, "I think if you really need to treat a patient now, you have to use what exists. But I think in half a year your options will change."
PEARL-II was a companion trial to PEARL-III, conducted in previously untreated patients with HCV 1b infection (N. Engl. J. Med. 2014 [doi:10.1056/NEJMoa1402338]). Both studies were sponsored by AbbVie. Dr. Ferenci reported serving on advisory panels for roughly a dozen pharmaceutical companies.
daily oral ribavirin, formerly a standard part of hepatitis C therapy, Europe and Japan, HCV, ombitasvir, dasabuvir, ABT-450, Dr. Peter Ferenci, annual Digestive Disease Week,
daily oral ribavirin, formerly a standard part of hepatitis C therapy, Europe and Japan, HCV, ombitasvir, dasabuvir, ABT-450, Dr. Peter Ferenci, annual Digestive Disease Week,
AT DDW 2014
Major finding: Noncirrhotic patients with hepatitis C genotype 1b disease who had failed previous pegylated interferon–based therapy had a 100% sustained virologic response rate 12 weeks after the conclusion of a 12-week course of all-oral therapy with three direct-acting antiviral agents without daily ribavirin and a 96.6% rate if they did get ribavirin.
Data source: This was an open-label, 12-week, phase III trial including 186 patients.
Disclosures: The study was sponsored by AbbVie. The presenter reported serving on advisory panels for about a dozen pharmaceutical companies.
Expert: Choose your sinus surgeon carefully
KEYSTONE, COLO. – Surgical treatment of chronic rhinosinusitis has come a long way from the earlier "grab and tear" days, but referring physicians need to understand that not all otolaryngologists are providing state-of-the-art care.
"I am critical of some of my colleagues," Dr. Todd T. Kingdom said at a meeting on allergy and respiratory diseases sponsored by National Jewish Health.
"If I leave you with one message, it’s to set high expectations of your consultants in otolaryngology. You should find colleagues who are interested in sinus disease, who are committed to it, and who are excellent," added Dr. Kingdom, professor and vice chairman of the department of otolaryngology, head and neck surgery, at the University of Colorado, Denver, and immediate past president of the American Rhinologic Society.
One fine source is the pool of graduates of U.S. subspecialty surgical rhinology fellowship programs. Each year, 30 surgeons complete one of these fellowships, he said.
Technical innovations over the past 15 years have driven major advances in endoscopic sinus surgery. Powered microdebriders are used to precisely and efficiently remove hyperplastic mucosal disease and restore mucociliary clearance. Mucosal preservation is now a central tenet. Forward-thinking surgeons place a priority on creating exposure for delivery of topical medications. The procedures are routinely done on an outpatient basis, and they are less invasive than in former times. The outcomes are better, too, with this modern patient-centered, symptom-based approach.
"We have efficient ways now to take care of very severe disease atraumatically," Dr. Kingdom explained.
He emphasized that postoperative care is critical to successful sinus surgery outcomes. "My biggest criticism of my colleagues in otolaryngology is that many of them cut and go. There isn’t an emphasis on postoperative care," he said. "That’s a clear, clear deficiency in our approach.
"My postop schedule is to see patients at 1, 3, and 6 weeks and 3 and 6 months after surgery – and that’s if they’re doing perfectly. My point is you should have your otolaryngologist really fussing over these people. It’s not, ‘Well, it’s been a couple of weeks, you look fine, you can go back to your allergist now, I’ll see you later.’ It shouldn’t be that way," Dr. Kingdom said.
He reported having no financial conflicts of interest.
Dr. Todd T. Kingdom, allergy and respiratory diseases, National Jewish Health, sinus disease, American Rhinologic Society, U.S. subspecialty surgical rhinology fellowship programs, endoscopic sinus surgery, Powered microdebriders, hyperplastic mucosal disease, restore mucociliary clearance, Mucosal preservation,
KEYSTONE, COLO. – Surgical treatment of chronic rhinosinusitis has come a long way from the earlier "grab and tear" days, but referring physicians need to understand that not all otolaryngologists are providing state-of-the-art care.
"I am critical of some of my colleagues," Dr. Todd T. Kingdom said at a meeting on allergy and respiratory diseases sponsored by National Jewish Health.
"If I leave you with one message, it’s to set high expectations of your consultants in otolaryngology. You should find colleagues who are interested in sinus disease, who are committed to it, and who are excellent," added Dr. Kingdom, professor and vice chairman of the department of otolaryngology, head and neck surgery, at the University of Colorado, Denver, and immediate past president of the American Rhinologic Society.
One fine source is the pool of graduates of U.S. subspecialty surgical rhinology fellowship programs. Each year, 30 surgeons complete one of these fellowships, he said.
Technical innovations over the past 15 years have driven major advances in endoscopic sinus surgery. Powered microdebriders are used to precisely and efficiently remove hyperplastic mucosal disease and restore mucociliary clearance. Mucosal preservation is now a central tenet. Forward-thinking surgeons place a priority on creating exposure for delivery of topical medications. The procedures are routinely done on an outpatient basis, and they are less invasive than in former times. The outcomes are better, too, with this modern patient-centered, symptom-based approach.
"We have efficient ways now to take care of very severe disease atraumatically," Dr. Kingdom explained.
He emphasized that postoperative care is critical to successful sinus surgery outcomes. "My biggest criticism of my colleagues in otolaryngology is that many of them cut and go. There isn’t an emphasis on postoperative care," he said. "That’s a clear, clear deficiency in our approach.
"My postop schedule is to see patients at 1, 3, and 6 weeks and 3 and 6 months after surgery – and that’s if they’re doing perfectly. My point is you should have your otolaryngologist really fussing over these people. It’s not, ‘Well, it’s been a couple of weeks, you look fine, you can go back to your allergist now, I’ll see you later.’ It shouldn’t be that way," Dr. Kingdom said.
He reported having no financial conflicts of interest.
KEYSTONE, COLO. – Surgical treatment of chronic rhinosinusitis has come a long way from the earlier "grab and tear" days, but referring physicians need to understand that not all otolaryngologists are providing state-of-the-art care.
"I am critical of some of my colleagues," Dr. Todd T. Kingdom said at a meeting on allergy and respiratory diseases sponsored by National Jewish Health.
"If I leave you with one message, it’s to set high expectations of your consultants in otolaryngology. You should find colleagues who are interested in sinus disease, who are committed to it, and who are excellent," added Dr. Kingdom, professor and vice chairman of the department of otolaryngology, head and neck surgery, at the University of Colorado, Denver, and immediate past president of the American Rhinologic Society.
One fine source is the pool of graduates of U.S. subspecialty surgical rhinology fellowship programs. Each year, 30 surgeons complete one of these fellowships, he said.
Technical innovations over the past 15 years have driven major advances in endoscopic sinus surgery. Powered microdebriders are used to precisely and efficiently remove hyperplastic mucosal disease and restore mucociliary clearance. Mucosal preservation is now a central tenet. Forward-thinking surgeons place a priority on creating exposure for delivery of topical medications. The procedures are routinely done on an outpatient basis, and they are less invasive than in former times. The outcomes are better, too, with this modern patient-centered, symptom-based approach.
"We have efficient ways now to take care of very severe disease atraumatically," Dr. Kingdom explained.
He emphasized that postoperative care is critical to successful sinus surgery outcomes. "My biggest criticism of my colleagues in otolaryngology is that many of them cut and go. There isn’t an emphasis on postoperative care," he said. "That’s a clear, clear deficiency in our approach.
"My postop schedule is to see patients at 1, 3, and 6 weeks and 3 and 6 months after surgery – and that’s if they’re doing perfectly. My point is you should have your otolaryngologist really fussing over these people. It’s not, ‘Well, it’s been a couple of weeks, you look fine, you can go back to your allergist now, I’ll see you later.’ It shouldn’t be that way," Dr. Kingdom said.
He reported having no financial conflicts of interest.
Dr. Todd T. Kingdom, allergy and respiratory diseases, National Jewish Health, sinus disease, American Rhinologic Society, U.S. subspecialty surgical rhinology fellowship programs, endoscopic sinus surgery, Powered microdebriders, hyperplastic mucosal disease, restore mucociliary clearance, Mucosal preservation,
Dr. Todd T. Kingdom, allergy and respiratory diseases, National Jewish Health, sinus disease, American Rhinologic Society, U.S. subspecialty surgical rhinology fellowship programs, endoscopic sinus surgery, Powered microdebriders, hyperplastic mucosal disease, restore mucociliary clearance, Mucosal preservation,
EXPERT ANALYSIS FROM THE PULMONARY AND ALLERGY UPDATE
Misery, thy name is chronic rhinosinusitis
KEYSTONE, COLO. – Just how lousy do patients with medically refractory chronic rhinosinusitis feel in daily life? A lot worse than you might guess.
Patients who elected to undergo endoscopic sinus surgery after failing medical therapy for chronic rhinosinusitis (CRS) rated their own baseline health state on standardized measures as being well below U.S. population norms. Their degree of impairment was similar to the self-rated scores among age- and gender-matched individuals with end-stage renal disease or Parkinson’s disease, according to Dr. Todd T. Kingdom, professor and vice chairman of the department of otolaryngology, head and neck surgery, at the University of Colorado, Denver, and immediate past president of the American Rhinologic Society.
He cited a 5-year study that prospectively followed 232 adults with CRS who elected to undergo endoscopic sinus surgery (ESS) after failing to improve on medical therapy (Laryngoscope 2011;121:2672-8). Their mean presurgical health state utility value – derived using the Short Form 6D via methods routinely employed by health economists – was 0.65, on a scale in which 0 is death and 1.0 is perfect health.
That was worse than the self-rated scores among patients with heart failure or moderate COPD, as reported in other studies, and only slightly better than the self-rated health of patients awaiting hip replacement or liver transplantation. The U.S. population norm was a score of 0.81, Dr. Kingdom noted at a meeting on allergy and respiratory diseases sponsored by National Jewish Health.
When self-rated health status scores were determined again 6 months or longer after ESS, patients who underwent a revision procedure had a statistically and clinically significant 0.06-point improvement on the 0-1 scale, while those with no prior sinus surgery showed an even more robust 0.09-point gain.
Those are markedly larger improvements than documented in other studies following initiation of drug therapy for Parkinson’s disease, for example, or tumor necrosis factor–inhibitor therapy for psoriasis. Of the specific interventions assessed, only total hip replacement and bariatric surgery resulted in greater self-rated gains in health status than ESS.
In this and other studies, a patient’s baseline clinical phenotype didn’t predict the degree of improvement on quality of life measures following ESS, and gender, age, comorbid asthma, or aspirin-exacerbated respiratory disease did not influence how much benefit a patient would receive from ESS.
Patients with baseline self-reported depression, however, were slightly, albeit statistically significantly, less likely than nondepressed patients to experience significant improvement. And patients who presented without nasal polyps showed significantly more improvement in self-reported health status after ESS than did those with polyps.
Dr. Kingdom reported having no financial conflicts of interest.
KEYSTONE, COLO. – Just how lousy do patients with medically refractory chronic rhinosinusitis feel in daily life? A lot worse than you might guess.
Patients who elected to undergo endoscopic sinus surgery after failing medical therapy for chronic rhinosinusitis (CRS) rated their own baseline health state on standardized measures as being well below U.S. population norms. Their degree of impairment was similar to the self-rated scores among age- and gender-matched individuals with end-stage renal disease or Parkinson’s disease, according to Dr. Todd T. Kingdom, professor and vice chairman of the department of otolaryngology, head and neck surgery, at the University of Colorado, Denver, and immediate past president of the American Rhinologic Society.
He cited a 5-year study that prospectively followed 232 adults with CRS who elected to undergo endoscopic sinus surgery (ESS) after failing to improve on medical therapy (Laryngoscope 2011;121:2672-8). Their mean presurgical health state utility value – derived using the Short Form 6D via methods routinely employed by health economists – was 0.65, on a scale in which 0 is death and 1.0 is perfect health.
That was worse than the self-rated scores among patients with heart failure or moderate COPD, as reported in other studies, and only slightly better than the self-rated health of patients awaiting hip replacement or liver transplantation. The U.S. population norm was a score of 0.81, Dr. Kingdom noted at a meeting on allergy and respiratory diseases sponsored by National Jewish Health.
When self-rated health status scores were determined again 6 months or longer after ESS, patients who underwent a revision procedure had a statistically and clinically significant 0.06-point improvement on the 0-1 scale, while those with no prior sinus surgery showed an even more robust 0.09-point gain.
Those are markedly larger improvements than documented in other studies following initiation of drug therapy for Parkinson’s disease, for example, or tumor necrosis factor–inhibitor therapy for psoriasis. Of the specific interventions assessed, only total hip replacement and bariatric surgery resulted in greater self-rated gains in health status than ESS.
In this and other studies, a patient’s baseline clinical phenotype didn’t predict the degree of improvement on quality of life measures following ESS, and gender, age, comorbid asthma, or aspirin-exacerbated respiratory disease did not influence how much benefit a patient would receive from ESS.
Patients with baseline self-reported depression, however, were slightly, albeit statistically significantly, less likely than nondepressed patients to experience significant improvement. And patients who presented without nasal polyps showed significantly more improvement in self-reported health status after ESS than did those with polyps.
Dr. Kingdom reported having no financial conflicts of interest.
KEYSTONE, COLO. – Just how lousy do patients with medically refractory chronic rhinosinusitis feel in daily life? A lot worse than you might guess.
Patients who elected to undergo endoscopic sinus surgery after failing medical therapy for chronic rhinosinusitis (CRS) rated their own baseline health state on standardized measures as being well below U.S. population norms. Their degree of impairment was similar to the self-rated scores among age- and gender-matched individuals with end-stage renal disease or Parkinson’s disease, according to Dr. Todd T. Kingdom, professor and vice chairman of the department of otolaryngology, head and neck surgery, at the University of Colorado, Denver, and immediate past president of the American Rhinologic Society.
He cited a 5-year study that prospectively followed 232 adults with CRS who elected to undergo endoscopic sinus surgery (ESS) after failing to improve on medical therapy (Laryngoscope 2011;121:2672-8). Their mean presurgical health state utility value – derived using the Short Form 6D via methods routinely employed by health economists – was 0.65, on a scale in which 0 is death and 1.0 is perfect health.
That was worse than the self-rated scores among patients with heart failure or moderate COPD, as reported in other studies, and only slightly better than the self-rated health of patients awaiting hip replacement or liver transplantation. The U.S. population norm was a score of 0.81, Dr. Kingdom noted at a meeting on allergy and respiratory diseases sponsored by National Jewish Health.
When self-rated health status scores were determined again 6 months or longer after ESS, patients who underwent a revision procedure had a statistically and clinically significant 0.06-point improvement on the 0-1 scale, while those with no prior sinus surgery showed an even more robust 0.09-point gain.
Those are markedly larger improvements than documented in other studies following initiation of drug therapy for Parkinson’s disease, for example, or tumor necrosis factor–inhibitor therapy for psoriasis. Of the specific interventions assessed, only total hip replacement and bariatric surgery resulted in greater self-rated gains in health status than ESS.
In this and other studies, a patient’s baseline clinical phenotype didn’t predict the degree of improvement on quality of life measures following ESS, and gender, age, comorbid asthma, or aspirin-exacerbated respiratory disease did not influence how much benefit a patient would receive from ESS.
Patients with baseline self-reported depression, however, were slightly, albeit statistically significantly, less likely than nondepressed patients to experience significant improvement. And patients who presented without nasal polyps showed significantly more improvement in self-reported health status after ESS than did those with polyps.
Dr. Kingdom reported having no financial conflicts of interest.
EXPERT ANALYSIS FROM THE PULMONARY AND ALLERGY UPDATE
Oral PPI therapy gets nod following bleeding ulcer repair
CHICAGO – Oral proton pump inhibitor therapy on the medical ward following endoscopic treatment of patients with a bleeding ulcer is equally effective as the current guideline-recommended practice of administering a PPI bolus followed by a 72-hour continuous intravenous infusion – and a lot less resource intensive.
That’s the highly practical message from a meta-analysis of randomized trials addressing the issue that Dr. Hamita Sachar presented at the annual Digestive Disease Week.
Current national and international guidelines recommend an intravenous 80-mg bolus of PPI followed by a 72-hour infusion at 8 mg/hr after successful endoscopic therapy of a high-risk bleeding ulcer. These guidelines need to be revisited in light of the new data, according to Dr. Sachar, a fellow in digestive diseases at Yale University, New Haven, Conn.
"This is practice changing. Intermittent oral therapy is less expensive. Also, it’s quite easy to administer. If you can get away with twice-daily oral dosing, it’s preferred over an IV infusion by patients, it requires less nursing time, and there’s less pharmacy personnel time. As long as patients don’t have nausea and vomiting, I think it’s reasonable for them to get oral therapy because equivalent doses of IV and oral drug have exactly the same pharmacokinetic profiles," she said in an interview.
The meta-analysis, which featured a noninferiority design, included a dozen randomized trials totaling more than 1,600 patients. The primary outcome was the rate of rebleeding within 7 days. It turned out to be virtually identical in the two groups.
Moreover, there was no significant difference between patients who received intermittent oral PPI therapy at standard doses and those who got the guideline-recommended bolus, continuous-infusion regimen in terms of any of the secondary endpoints in the meta-analysis, which included rebleeding within 30 days, rebleeding within 3 days, need for blood transfusion, urgent surgical or endoscopic interventions, and length of hospital stay.
Dr. Sachar and her coinvestigators performed the meta-analysis because their physiologic model suggested that intravenous therapy was safely avoidable. They noted that several randomized trials in the literature concluded that the two treatment strategies might yield comparable outcomes; however, the individual studies were too small to allow any firm conclusions to be drawn.
The study was supported by institutional funds. Dr. Sachar reported having no financial conflicts regarding this study.
Sachar and colleagues presented an interesting systematic review and meta-analysis at DDW 2014 (Gastroenterology 2014;146(5 Suppl 1):S-75). This presentation has fueled discussions on whether high-risk patients with peptic ulcer bleeding (those who had successful endoscopic hemostasis for high-risk stigmata) should receive PPIs intermittently (intravenously or even orally) as opposed to the current practice of high-dose continuous intravenous infusion.

|
| Dr. Grigorios I. Leontiadis |
In conclusion, until the quality of the evidence can be fully assessed, it would be prudent to adhere to the recommendations from the existing practice guidelines and treat high-risk patients with peptic ulcer bleeding with high-dose intravenous infusion PPI treatment, since this is the only dose that has been proven to reduce not only rebleeding but also mortality.
Dr. Grigorios I. Leontiadis is assistant professor of medicine, division of gastroenterology, McMaster University, Hamilton, Ont. He was a consultant to a PPI manufacturer more than 5 years ago.
Sachar and colleagues presented an interesting systematic review and meta-analysis at DDW 2014 (Gastroenterology 2014;146(5 Suppl 1):S-75). This presentation has fueled discussions on whether high-risk patients with peptic ulcer bleeding (those who had successful endoscopic hemostasis for high-risk stigmata) should receive PPIs intermittently (intravenously or even orally) as opposed to the current practice of high-dose continuous intravenous infusion.

|
| Dr. Grigorios I. Leontiadis |
In conclusion, until the quality of the evidence can be fully assessed, it would be prudent to adhere to the recommendations from the existing practice guidelines and treat high-risk patients with peptic ulcer bleeding with high-dose intravenous infusion PPI treatment, since this is the only dose that has been proven to reduce not only rebleeding but also mortality.
Dr. Grigorios I. Leontiadis is assistant professor of medicine, division of gastroenterology, McMaster University, Hamilton, Ont. He was a consultant to a PPI manufacturer more than 5 years ago.
Sachar and colleagues presented an interesting systematic review and meta-analysis at DDW 2014 (Gastroenterology 2014;146(5 Suppl 1):S-75). This presentation has fueled discussions on whether high-risk patients with peptic ulcer bleeding (those who had successful endoscopic hemostasis for high-risk stigmata) should receive PPIs intermittently (intravenously or even orally) as opposed to the current practice of high-dose continuous intravenous infusion.

|
| Dr. Grigorios I. Leontiadis |
In conclusion, until the quality of the evidence can be fully assessed, it would be prudent to adhere to the recommendations from the existing practice guidelines and treat high-risk patients with peptic ulcer bleeding with high-dose intravenous infusion PPI treatment, since this is the only dose that has been proven to reduce not only rebleeding but also mortality.
Dr. Grigorios I. Leontiadis is assistant professor of medicine, division of gastroenterology, McMaster University, Hamilton, Ont. He was a consultant to a PPI manufacturer more than 5 years ago.
CHICAGO – Oral proton pump inhibitor therapy on the medical ward following endoscopic treatment of patients with a bleeding ulcer is equally effective as the current guideline-recommended practice of administering a PPI bolus followed by a 72-hour continuous intravenous infusion – and a lot less resource intensive.
That’s the highly practical message from a meta-analysis of randomized trials addressing the issue that Dr. Hamita Sachar presented at the annual Digestive Disease Week.
Current national and international guidelines recommend an intravenous 80-mg bolus of PPI followed by a 72-hour infusion at 8 mg/hr after successful endoscopic therapy of a high-risk bleeding ulcer. These guidelines need to be revisited in light of the new data, according to Dr. Sachar, a fellow in digestive diseases at Yale University, New Haven, Conn.
"This is practice changing. Intermittent oral therapy is less expensive. Also, it’s quite easy to administer. If you can get away with twice-daily oral dosing, it’s preferred over an IV infusion by patients, it requires less nursing time, and there’s less pharmacy personnel time. As long as patients don’t have nausea and vomiting, I think it’s reasonable for them to get oral therapy because equivalent doses of IV and oral drug have exactly the same pharmacokinetic profiles," she said in an interview.
The meta-analysis, which featured a noninferiority design, included a dozen randomized trials totaling more than 1,600 patients. The primary outcome was the rate of rebleeding within 7 days. It turned out to be virtually identical in the two groups.
Moreover, there was no significant difference between patients who received intermittent oral PPI therapy at standard doses and those who got the guideline-recommended bolus, continuous-infusion regimen in terms of any of the secondary endpoints in the meta-analysis, which included rebleeding within 30 days, rebleeding within 3 days, need for blood transfusion, urgent surgical or endoscopic interventions, and length of hospital stay.
Dr. Sachar and her coinvestigators performed the meta-analysis because their physiologic model suggested that intravenous therapy was safely avoidable. They noted that several randomized trials in the literature concluded that the two treatment strategies might yield comparable outcomes; however, the individual studies were too small to allow any firm conclusions to be drawn.
The study was supported by institutional funds. Dr. Sachar reported having no financial conflicts regarding this study.
CHICAGO – Oral proton pump inhibitor therapy on the medical ward following endoscopic treatment of patients with a bleeding ulcer is equally effective as the current guideline-recommended practice of administering a PPI bolus followed by a 72-hour continuous intravenous infusion – and a lot less resource intensive.
That’s the highly practical message from a meta-analysis of randomized trials addressing the issue that Dr. Hamita Sachar presented at the annual Digestive Disease Week.
Current national and international guidelines recommend an intravenous 80-mg bolus of PPI followed by a 72-hour infusion at 8 mg/hr after successful endoscopic therapy of a high-risk bleeding ulcer. These guidelines need to be revisited in light of the new data, according to Dr. Sachar, a fellow in digestive diseases at Yale University, New Haven, Conn.
"This is practice changing. Intermittent oral therapy is less expensive. Also, it’s quite easy to administer. If you can get away with twice-daily oral dosing, it’s preferred over an IV infusion by patients, it requires less nursing time, and there’s less pharmacy personnel time. As long as patients don’t have nausea and vomiting, I think it’s reasonable for them to get oral therapy because equivalent doses of IV and oral drug have exactly the same pharmacokinetic profiles," she said in an interview.
The meta-analysis, which featured a noninferiority design, included a dozen randomized trials totaling more than 1,600 patients. The primary outcome was the rate of rebleeding within 7 days. It turned out to be virtually identical in the two groups.
Moreover, there was no significant difference between patients who received intermittent oral PPI therapy at standard doses and those who got the guideline-recommended bolus, continuous-infusion regimen in terms of any of the secondary endpoints in the meta-analysis, which included rebleeding within 30 days, rebleeding within 3 days, need for blood transfusion, urgent surgical or endoscopic interventions, and length of hospital stay.
Dr. Sachar and her coinvestigators performed the meta-analysis because their physiologic model suggested that intravenous therapy was safely avoidable. They noted that several randomized trials in the literature concluded that the two treatment strategies might yield comparable outcomes; however, the individual studies were too small to allow any firm conclusions to be drawn.
The study was supported by institutional funds. Dr. Sachar reported having no financial conflicts regarding this study.
AT DDW 2014
Major finding: Oral proton pump inhibitor therapy can safely and effectively replace the guideline-recommended standard regimen consisting of a PPI bolus followed by 72 hours of continuous intravenous infusion after endoscopic treatment of a bleeding ulcer.
Data source: This was a meta-analysis with a noninferiority design. It included 12 randomized trials totaling 1,653 patients who had undergone endoscopic hemostasis for a bleeding ulcer.
Disclosures: The study was supported by institutional funds. Dr. Sachar reported having no financial conflicts regarding this study.
Think ‘celiac disease’ in patients requiring high-dose levothyroxine
CHICAGO – Hypothyroid patients who need either at least 125 mcg or 1.5 mcg/kg of levothyroxine per day in order to remain euthyroid should routinely be tested for celiac disease, Dr. Richard S. Zubarik asserted at the annual Digestive Disease Week.
In his cross-sectional study of 400 consecutive patients with hypothyroidism who underwent testing for celiac disease, 5% of those on a levothyroxine dose at or above that threshold were found to have biopsy-confirmed celiac disease.
Mass screening for celiac disease in the broad U.S. population isn’t recommended at present because the prevalence – 0.75% is deemed too low to justify such a practice. However, current national and international guidelines do recommend routine testing for case finding in selected populations known to be at increased risk of celiac disease. These include, for example, patients with asymptomatic iron-deficiency anemia, who have a celiac disease prevalence of 2.3%-5%. Thus, the 5% prevalence of celiac disease in hypothyroid patients requiring high-dose levothyroxine in order to maintain a euthyroid state is at least as high as, and perhaps higher than, the prevalence in groups having a guideline-recommended indication for testing, noted Dr. Zubarik, professor of medicine and director of GI endoscopy at the University of Vermont, Burlington.
Testing involves a simple serologic test for tissue transglutaminase. An elevated level triggers endoscopy with duodenal biopsies. A positive biopsy confirms the diagnosis and warrants initiation of a gluten-free diet.
The 400 consecutive patients on treatment for hypothyroidism in Dr. Zubarik’s study averaged 59 years of age, and 82% were women. Thirty percent of participants required 125 mcg or more of levothyroxine, and 29% needed at least 1.5 mcg/kg daily in order to maintain euthyroid status. Dr. Zubarik and his coinvestigators selected those doses as their threshold in the cross-sectional study because their earlier retrospective study had suggested patients requiring that much levothyroxine might have an increased prevalence of concomitant celiac disease (Am. J. Med. 2012;125:278-82). The group requiring an elevated dose and those who were able to remain euthyroid on lower doses were similar in age, sex, weight, body mass index, and the prevalence of diabetes.
All subjects took a serologic tissue transglutaminase test, and those with an elevated level underwent endoscopy with biopsies.
Eight of the 400 patients had an elevated serum tissue transglutaminase level, and seven of the eight were subsequently confirmed as having biopsy-proven celiac disease. Six of the seven patients with celiac disease met or exceeded the levothyroxine dose threshold.
Gastrointestinal symptoms weren’t helpful in differentiating the hypothyroid patients with and without celiac disease. Scores on the Gastrointestinal Symptom Rating Scale weren’t significantly different between the two groups.
Malabsorption of vitamins, minerals, nutrients, and medications is not uncommon in patients with celiac disease, which accounts for the increased disease prevalence seen among patients with iron-deficiency anemia. Whether the increased prevalence of celiac disease among hypothyroid patients requiring elevated doses of levothyroxine is the result of levothyroxine malabsorption or perhaps a consequence of more severe hypothyroidism being present in patients with concomitant celiac disease is an unanswered question Dr. Zubarik plans to study.
The association between thyroid disease and celiac disease is independent of gluten exposure and most likely stems from a common genetic predisposition, the gastroenterologist said.
This study was carried out free of commercial support. Dr. Zubarik reported having no financial conflicts.
CHICAGO – Hypothyroid patients who need either at least 125 mcg or 1.5 mcg/kg of levothyroxine per day in order to remain euthyroid should routinely be tested for celiac disease, Dr. Richard S. Zubarik asserted at the annual Digestive Disease Week.
In his cross-sectional study of 400 consecutive patients with hypothyroidism who underwent testing for celiac disease, 5% of those on a levothyroxine dose at or above that threshold were found to have biopsy-confirmed celiac disease.
Mass screening for celiac disease in the broad U.S. population isn’t recommended at present because the prevalence – 0.75% is deemed too low to justify such a practice. However, current national and international guidelines do recommend routine testing for case finding in selected populations known to be at increased risk of celiac disease. These include, for example, patients with asymptomatic iron-deficiency anemia, who have a celiac disease prevalence of 2.3%-5%. Thus, the 5% prevalence of celiac disease in hypothyroid patients requiring high-dose levothyroxine in order to maintain a euthyroid state is at least as high as, and perhaps higher than, the prevalence in groups having a guideline-recommended indication for testing, noted Dr. Zubarik, professor of medicine and director of GI endoscopy at the University of Vermont, Burlington.
Testing involves a simple serologic test for tissue transglutaminase. An elevated level triggers endoscopy with duodenal biopsies. A positive biopsy confirms the diagnosis and warrants initiation of a gluten-free diet.
The 400 consecutive patients on treatment for hypothyroidism in Dr. Zubarik’s study averaged 59 years of age, and 82% were women. Thirty percent of participants required 125 mcg or more of levothyroxine, and 29% needed at least 1.5 mcg/kg daily in order to maintain euthyroid status. Dr. Zubarik and his coinvestigators selected those doses as their threshold in the cross-sectional study because their earlier retrospective study had suggested patients requiring that much levothyroxine might have an increased prevalence of concomitant celiac disease (Am. J. Med. 2012;125:278-82). The group requiring an elevated dose and those who were able to remain euthyroid on lower doses were similar in age, sex, weight, body mass index, and the prevalence of diabetes.
All subjects took a serologic tissue transglutaminase test, and those with an elevated level underwent endoscopy with biopsies.
Eight of the 400 patients had an elevated serum tissue transglutaminase level, and seven of the eight were subsequently confirmed as having biopsy-proven celiac disease. Six of the seven patients with celiac disease met or exceeded the levothyroxine dose threshold.
Gastrointestinal symptoms weren’t helpful in differentiating the hypothyroid patients with and without celiac disease. Scores on the Gastrointestinal Symptom Rating Scale weren’t significantly different between the two groups.
Malabsorption of vitamins, minerals, nutrients, and medications is not uncommon in patients with celiac disease, which accounts for the increased disease prevalence seen among patients with iron-deficiency anemia. Whether the increased prevalence of celiac disease among hypothyroid patients requiring elevated doses of levothyroxine is the result of levothyroxine malabsorption or perhaps a consequence of more severe hypothyroidism being present in patients with concomitant celiac disease is an unanswered question Dr. Zubarik plans to study.
The association between thyroid disease and celiac disease is independent of gluten exposure and most likely stems from a common genetic predisposition, the gastroenterologist said.
This study was carried out free of commercial support. Dr. Zubarik reported having no financial conflicts.
CHICAGO – Hypothyroid patients who need either at least 125 mcg or 1.5 mcg/kg of levothyroxine per day in order to remain euthyroid should routinely be tested for celiac disease, Dr. Richard S. Zubarik asserted at the annual Digestive Disease Week.
In his cross-sectional study of 400 consecutive patients with hypothyroidism who underwent testing for celiac disease, 5% of those on a levothyroxine dose at or above that threshold were found to have biopsy-confirmed celiac disease.
Mass screening for celiac disease in the broad U.S. population isn’t recommended at present because the prevalence – 0.75% is deemed too low to justify such a practice. However, current national and international guidelines do recommend routine testing for case finding in selected populations known to be at increased risk of celiac disease. These include, for example, patients with asymptomatic iron-deficiency anemia, who have a celiac disease prevalence of 2.3%-5%. Thus, the 5% prevalence of celiac disease in hypothyroid patients requiring high-dose levothyroxine in order to maintain a euthyroid state is at least as high as, and perhaps higher than, the prevalence in groups having a guideline-recommended indication for testing, noted Dr. Zubarik, professor of medicine and director of GI endoscopy at the University of Vermont, Burlington.
Testing involves a simple serologic test for tissue transglutaminase. An elevated level triggers endoscopy with duodenal biopsies. A positive biopsy confirms the diagnosis and warrants initiation of a gluten-free diet.
The 400 consecutive patients on treatment for hypothyroidism in Dr. Zubarik’s study averaged 59 years of age, and 82% were women. Thirty percent of participants required 125 mcg or more of levothyroxine, and 29% needed at least 1.5 mcg/kg daily in order to maintain euthyroid status. Dr. Zubarik and his coinvestigators selected those doses as their threshold in the cross-sectional study because their earlier retrospective study had suggested patients requiring that much levothyroxine might have an increased prevalence of concomitant celiac disease (Am. J. Med. 2012;125:278-82). The group requiring an elevated dose and those who were able to remain euthyroid on lower doses were similar in age, sex, weight, body mass index, and the prevalence of diabetes.
All subjects took a serologic tissue transglutaminase test, and those with an elevated level underwent endoscopy with biopsies.
Eight of the 400 patients had an elevated serum tissue transglutaminase level, and seven of the eight were subsequently confirmed as having biopsy-proven celiac disease. Six of the seven patients with celiac disease met or exceeded the levothyroxine dose threshold.
Gastrointestinal symptoms weren’t helpful in differentiating the hypothyroid patients with and without celiac disease. Scores on the Gastrointestinal Symptom Rating Scale weren’t significantly different between the two groups.
Malabsorption of vitamins, minerals, nutrients, and medications is not uncommon in patients with celiac disease, which accounts for the increased disease prevalence seen among patients with iron-deficiency anemia. Whether the increased prevalence of celiac disease among hypothyroid patients requiring elevated doses of levothyroxine is the result of levothyroxine malabsorption or perhaps a consequence of more severe hypothyroidism being present in patients with concomitant celiac disease is an unanswered question Dr. Zubarik plans to study.
The association between thyroid disease and celiac disease is independent of gluten exposure and most likely stems from a common genetic predisposition, the gastroenterologist said.
This study was carried out free of commercial support. Dr. Zubarik reported having no financial conflicts.
AT DDW 2014
Major finding: Five percent of hypothyroid patients who needed 125 mcg or more of levothyroxine daily in order to remain euthyroid proved to have previously undiagnosed celiac disease, a prevalence deemed high enough to warrant routine testing for the GI disease in that population.
Data source: This was a cross-sectional study involving 400 consecutive patients being treated for hypothyroidism, all of whom underwent serologic testing for celiac disease via the serum tissue transglutaminase test.
Disclosures: This study was carried out free of commercial support. Dr. Zubarik reported having no financial conflicts.
Eluxadoline scores in phase III for irritable bowel syndrome
CHICAGO – A first-in-class oral drug targeting diarrhea-predominant irritable bowel syndrome achieved its primary and secondary endpoints in a pair of large phase III trials presented at the annual Digestive Disease Week.
Eluxadoline is a locally active mixed mu-opioid receptor agonist and delta-opioid receptor antagonist. It was compared with placebo at doses of 75 mg and 100 mg b.i.d. in the two double-blind phase III trials, which totaled 2,427 patients with diarrhea-predominant irritable bowel syndrome (IBS-D) by Rome III criteria. The trials featured identical outcome measures; however, one study entailed 12 weeks of double-blind therapy in accord with a Food and Drug Administration request, while the other involved 26 weeks as stipulated by the European Medicines Agency, explained Dr. Anthony Lembo, director of the GI motility center at Beth Israel Deaconess Medical Center, Boston.
The primary endpoint was a composite requiring at least 30% improvement in daily abdominal pain, compared with baseline, and improved stool consistency as reflected in a Bristol Stool Score of less than 5. In the 12-week trial, this was achieved by 27% of patients on eluxadoline at 100 mg b.i.d. and by 26.2% on 75 mg b.i.d., both significantly better rates than the 16.7% in placebo-treated controls. In the 26-week study, the rates were 31%, 26.7%, and 19.5%, respectively. Improvement in the eluxadoline group was seen within the first several days of treatment.
The drug was equally effective in men and women.
Turning to secondary endpoints, Dr. Lembo highlighted the finding that 42% of the eluxadoline 100 mg b.i.d. group reported more than 50% of their days during weeks 1-12 as being urgency free, compared with 21% of controls. Twenty-nine percent of those on the higher dose reported more than 75% of days were urgency free, compared with 12% on placebo.
Bowel movement frequency improved from a baseline average of 4.9 per day to 2.9 per day at week 26 in the group on eluxadoline 100 mg b.i.d., significantly better than the 3.3 movements per day in controls. A 40% or greater improvement in abdominal pain score was reported at week 12 by 43.2% of patients on eluxadoline 100 mg b.i.d. compared with 35.8% of controls.
Safety concerns arose. Eight confirmed cases of hepatobiliary sphincter of Oddi spasm occurred, all in patients on eluxadoline, and all in patients with prior cholecystectomy. Seven of the eight patients were in the high-dose therapy arm. Six cases occurred during the first week of therapy. All eight cases were rapidly reversed upon prompt drug discontinuation; seven were managed on an outpatient basis.
In addition, there were five confirmed cases of pancreatitis, again all in the eluxadoline group. All five cases were mild, and all occurred in patients with pancreatitis risk factors: Three were chronic heavy alcohol abusers, one had biliary sludge, and one had a history of cholecystectomy with sphincter of Oddi spasm. These five patients had brief hospitalizations with no sequelae.
Several audience members greeted this news as a potential roadblock in the drug’s development. Dr. Lembo took a different view.
"These risks can be mitigated through appropriate patient selection and education," the gastroenterologist said. "There does seem to be a group of patients that seem to be particularly at risk: those with prior cholecystectomy or chronic alcohol use. If that’s true and the drug is relatively safe in the remainder of the population, then I think the risk/benefit ratio would be acceptable, especially since there are relatively few other treatment options for these patients."
Other audience members were critical of eluxadoline’s efficacy. "You have a drug that has at best extremely modest effects, with about a 10% absolute overall improvement over placebo, which is statistically significant primarily because you had so many patients in the studies," one gastroenterologist asserted.
Dr. Lembo replied that this magnitude of benefit is similar to what’s seen with drugs now approved for the common and vexing problem of IBS-D.
The phase III trials were sponsored by Furiex Pharmaceuticals. Dr. Lembo reported serving as an adviser or consultant to five pharmaceutical companies, not including Furiex.
CHICAGO – A first-in-class oral drug targeting diarrhea-predominant irritable bowel syndrome achieved its primary and secondary endpoints in a pair of large phase III trials presented at the annual Digestive Disease Week.
Eluxadoline is a locally active mixed mu-opioid receptor agonist and delta-opioid receptor antagonist. It was compared with placebo at doses of 75 mg and 100 mg b.i.d. in the two double-blind phase III trials, which totaled 2,427 patients with diarrhea-predominant irritable bowel syndrome (IBS-D) by Rome III criteria. The trials featured identical outcome measures; however, one study entailed 12 weeks of double-blind therapy in accord with a Food and Drug Administration request, while the other involved 26 weeks as stipulated by the European Medicines Agency, explained Dr. Anthony Lembo, director of the GI motility center at Beth Israel Deaconess Medical Center, Boston.
The primary endpoint was a composite requiring at least 30% improvement in daily abdominal pain, compared with baseline, and improved stool consistency as reflected in a Bristol Stool Score of less than 5. In the 12-week trial, this was achieved by 27% of patients on eluxadoline at 100 mg b.i.d. and by 26.2% on 75 mg b.i.d., both significantly better rates than the 16.7% in placebo-treated controls. In the 26-week study, the rates were 31%, 26.7%, and 19.5%, respectively. Improvement in the eluxadoline group was seen within the first several days of treatment.
The drug was equally effective in men and women.
Turning to secondary endpoints, Dr. Lembo highlighted the finding that 42% of the eluxadoline 100 mg b.i.d. group reported more than 50% of their days during weeks 1-12 as being urgency free, compared with 21% of controls. Twenty-nine percent of those on the higher dose reported more than 75% of days were urgency free, compared with 12% on placebo.
Bowel movement frequency improved from a baseline average of 4.9 per day to 2.9 per day at week 26 in the group on eluxadoline 100 mg b.i.d., significantly better than the 3.3 movements per day in controls. A 40% or greater improvement in abdominal pain score was reported at week 12 by 43.2% of patients on eluxadoline 100 mg b.i.d. compared with 35.8% of controls.
Safety concerns arose. Eight confirmed cases of hepatobiliary sphincter of Oddi spasm occurred, all in patients on eluxadoline, and all in patients with prior cholecystectomy. Seven of the eight patients were in the high-dose therapy arm. Six cases occurred during the first week of therapy. All eight cases were rapidly reversed upon prompt drug discontinuation; seven were managed on an outpatient basis.
In addition, there were five confirmed cases of pancreatitis, again all in the eluxadoline group. All five cases were mild, and all occurred in patients with pancreatitis risk factors: Three were chronic heavy alcohol abusers, one had biliary sludge, and one had a history of cholecystectomy with sphincter of Oddi spasm. These five patients had brief hospitalizations with no sequelae.
Several audience members greeted this news as a potential roadblock in the drug’s development. Dr. Lembo took a different view.
"These risks can be mitigated through appropriate patient selection and education," the gastroenterologist said. "There does seem to be a group of patients that seem to be particularly at risk: those with prior cholecystectomy or chronic alcohol use. If that’s true and the drug is relatively safe in the remainder of the population, then I think the risk/benefit ratio would be acceptable, especially since there are relatively few other treatment options for these patients."
Other audience members were critical of eluxadoline’s efficacy. "You have a drug that has at best extremely modest effects, with about a 10% absolute overall improvement over placebo, which is statistically significant primarily because you had so many patients in the studies," one gastroenterologist asserted.
Dr. Lembo replied that this magnitude of benefit is similar to what’s seen with drugs now approved for the common and vexing problem of IBS-D.
The phase III trials were sponsored by Furiex Pharmaceuticals. Dr. Lembo reported serving as an adviser or consultant to five pharmaceutical companies, not including Furiex.
CHICAGO – A first-in-class oral drug targeting diarrhea-predominant irritable bowel syndrome achieved its primary and secondary endpoints in a pair of large phase III trials presented at the annual Digestive Disease Week.
Eluxadoline is a locally active mixed mu-opioid receptor agonist and delta-opioid receptor antagonist. It was compared with placebo at doses of 75 mg and 100 mg b.i.d. in the two double-blind phase III trials, which totaled 2,427 patients with diarrhea-predominant irritable bowel syndrome (IBS-D) by Rome III criteria. The trials featured identical outcome measures; however, one study entailed 12 weeks of double-blind therapy in accord with a Food and Drug Administration request, while the other involved 26 weeks as stipulated by the European Medicines Agency, explained Dr. Anthony Lembo, director of the GI motility center at Beth Israel Deaconess Medical Center, Boston.
The primary endpoint was a composite requiring at least 30% improvement in daily abdominal pain, compared with baseline, and improved stool consistency as reflected in a Bristol Stool Score of less than 5. In the 12-week trial, this was achieved by 27% of patients on eluxadoline at 100 mg b.i.d. and by 26.2% on 75 mg b.i.d., both significantly better rates than the 16.7% in placebo-treated controls. In the 26-week study, the rates were 31%, 26.7%, and 19.5%, respectively. Improvement in the eluxadoline group was seen within the first several days of treatment.
The drug was equally effective in men and women.
Turning to secondary endpoints, Dr. Lembo highlighted the finding that 42% of the eluxadoline 100 mg b.i.d. group reported more than 50% of their days during weeks 1-12 as being urgency free, compared with 21% of controls. Twenty-nine percent of those on the higher dose reported more than 75% of days were urgency free, compared with 12% on placebo.
Bowel movement frequency improved from a baseline average of 4.9 per day to 2.9 per day at week 26 in the group on eluxadoline 100 mg b.i.d., significantly better than the 3.3 movements per day in controls. A 40% or greater improvement in abdominal pain score was reported at week 12 by 43.2% of patients on eluxadoline 100 mg b.i.d. compared with 35.8% of controls.
Safety concerns arose. Eight confirmed cases of hepatobiliary sphincter of Oddi spasm occurred, all in patients on eluxadoline, and all in patients with prior cholecystectomy. Seven of the eight patients were in the high-dose therapy arm. Six cases occurred during the first week of therapy. All eight cases were rapidly reversed upon prompt drug discontinuation; seven were managed on an outpatient basis.
In addition, there were five confirmed cases of pancreatitis, again all in the eluxadoline group. All five cases were mild, and all occurred in patients with pancreatitis risk factors: Three were chronic heavy alcohol abusers, one had biliary sludge, and one had a history of cholecystectomy with sphincter of Oddi spasm. These five patients had brief hospitalizations with no sequelae.
Several audience members greeted this news as a potential roadblock in the drug’s development. Dr. Lembo took a different view.
"These risks can be mitigated through appropriate patient selection and education," the gastroenterologist said. "There does seem to be a group of patients that seem to be particularly at risk: those with prior cholecystectomy or chronic alcohol use. If that’s true and the drug is relatively safe in the remainder of the population, then I think the risk/benefit ratio would be acceptable, especially since there are relatively few other treatment options for these patients."
Other audience members were critical of eluxadoline’s efficacy. "You have a drug that has at best extremely modest effects, with about a 10% absolute overall improvement over placebo, which is statistically significant primarily because you had so many patients in the studies," one gastroenterologist asserted.
Dr. Lembo replied that this magnitude of benefit is similar to what’s seen with drugs now approved for the common and vexing problem of IBS-D.
The phase III trials were sponsored by Furiex Pharmaceuticals. Dr. Lembo reported serving as an adviser or consultant to five pharmaceutical companies, not including Furiex.
AT DDW 2014
Major finding: Thirty-one percent of patients with diarrhea-predominant irritable bowel syndrome on 100 mg b.i.d. of the investigational oral agent eluxadoline for 26 weeks met the primary composite study efficacy endpoint, compared with 19.5% on placebo.
Data source: These two phase III, double-blind, randomized, placebo-controlled trials included 2,427 patients with diarrhea-predominant irritable bowel syndrome.
Disclosures: The phase III trials were sponsored by Furiex Pharmaceuticals. Dr. Lembo reported serving as an adviser or consultant to five pharmaceutical companies, not including Furiex.
Larazotide eased symptoms in phase II celiac trial
CHICAGO – Larazotide acetate, a first-in-class oral medication developed specifically to treat celiac disease, reduced both gastrointestinal and non-GI symptoms in patients who were symptomatic despite being on a gluten-free diet in a 74-site, randomized, double-blind phase II study.
"Safety and tolerability were comparable to placebo, setting this drug up, we believe, for phase III trials," Dr. Joseph A. Murray said at the annual Digestive Disease Week.
The study included 342 adult patients with celiac disease who had been on a gluten-free diet (GFD) for at least 12 months and continued on the GFD throughout the 12-week treatment period. Thus, this was a study of larazotide as an adjunct to the GFD, not as an alternative to it.
A GFD is of well-established efficacy as the sole proven therapy for celiac disease. However, the diet can be challenging. Surveys indicate 70% of celiac disease patients on a GFD experience recurrent symptoms due to inadvertent exposure or lapses in the strict adherence required. Thus, there is a significant unmet need for an effective pharmacologic therapy for this common disease, observed Dr. Murray, professor of medicine at the Mayo Clinic in Rochester, Minn.
Study participants were randomized to 12 weeks of double-blind treatment with larazotide at 0.5, 1, or 2 mg three times daily or to placebo. The study provided a successful opportunity to validate a new instrument designed for evaluation of celiac disease symptoms both in clinical trials and in daily practice. The instrument, known as the Celiac Disease Patient Reported Outcome (CeD PRO), entails daily assessment of symptoms in three key domains: abdominal symptoms such as cramping, bloating, and pain; diarrhea and loose stools; and the non-GI symptoms of headache and tiredness, which figure prominently in the disease but aren’t included in the older Celiac Disease Gastrointestinal Symptom Rating Scale.
Patients in the larazotide 0.5-mg t.i.d. group showed a significant 26% reduction in the average weekly number of CeD PRO symptomatic days, compared with placebo-treated controls. Twenty-nine percent of patients in the larazotide 0.5-mg group also achieved at least a 50% decrease from baseline in the weekly average CeD PRO abdominal symptom domain scores for at least 6 of the 12 weeks of treatment, significantly greater than the 14% rate with placebo. In addition, the larazotide 0.5-mg t.i.d. group had a 31% increase in the average number of CeD PRO days with minimal or no symptoms, compared with controls. Moreover, patients on the lowest dose of larazotide showed a significant reduction in CeD PRO non-GI symptoms, the only study arm to do so.
Of note, patients on the two higher doses of larazotide didn’t significantly outperform the control group in any of these study endpoints, although favorable trends were seen.
No changes in serially measured levels of anti–tissue transglutaminase antibodies or anti–deaminated gliadin antibodies were seen in any treatment arm.
Larazotide is an oral peptide that prevents the opening of epithelial tight junctions in the small bowel, an event that occurs in response to gluten exposure and inflammatory cytokines in patients with celiac disease. When these tight junctions are open inappropriately, the result is increased intestinal permeability and inflammation.
One audience member expressed reservations about the finding that only the lowest tested dose in a dose-ranging study proved effective.
"I’m troubled by drugs that don’t show a dose-response curve. This positive result could just be the play of chance," he cautioned.
Dr. Murray replied that he believes the consistency of the positive results with the 0.5-mg dose across multiple endpoints reduces that likelihood. Also, the lowest tested dose was the one that was effective in all the prior challenge studies. In any case, he added, the larger patient numbers planned for the phase III trials will sort out this issue.
Dr. Benjamin Lebwohl was enthusiastic about the larazotide results.
"It’s a very exciting time in the celiac disease community. Larazotide is one of several nondietary treatments currently in the pipeline," noted Dr. Lebwohl of the celiac disease center at Columbia University, New York.
The time is at hand, he added, to think big in terms of celiac disease research questions.
"Is it possible to prevent celiac disease in those individuals at increased risk? And among those individuals who come to us having been diagnosed with celiac disease, can we stop it in its tracks? Can we tolerize patients to gluten, allowing them to eat gluten either intermittently or even resume a full gluten-containing diet? Will there be a cure for celiac disease?" Dr. Lebwohl asked.
The study was supported by Alba Therapeutics. Dr. Murray reported serving as a consultant to or on the advisory boards of nearly a dozen pharmaceutical companies, Alba not among them. Dr. Lebwohl reported having no financial conflicts.
CHICAGO – Larazotide acetate, a first-in-class oral medication developed specifically to treat celiac disease, reduced both gastrointestinal and non-GI symptoms in patients who were symptomatic despite being on a gluten-free diet in a 74-site, randomized, double-blind phase II study.
"Safety and tolerability were comparable to placebo, setting this drug up, we believe, for phase III trials," Dr. Joseph A. Murray said at the annual Digestive Disease Week.
The study included 342 adult patients with celiac disease who had been on a gluten-free diet (GFD) for at least 12 months and continued on the GFD throughout the 12-week treatment period. Thus, this was a study of larazotide as an adjunct to the GFD, not as an alternative to it.
A GFD is of well-established efficacy as the sole proven therapy for celiac disease. However, the diet can be challenging. Surveys indicate 70% of celiac disease patients on a GFD experience recurrent symptoms due to inadvertent exposure or lapses in the strict adherence required. Thus, there is a significant unmet need for an effective pharmacologic therapy for this common disease, observed Dr. Murray, professor of medicine at the Mayo Clinic in Rochester, Minn.
Study participants were randomized to 12 weeks of double-blind treatment with larazotide at 0.5, 1, or 2 mg three times daily or to placebo. The study provided a successful opportunity to validate a new instrument designed for evaluation of celiac disease symptoms both in clinical trials and in daily practice. The instrument, known as the Celiac Disease Patient Reported Outcome (CeD PRO), entails daily assessment of symptoms in three key domains: abdominal symptoms such as cramping, bloating, and pain; diarrhea and loose stools; and the non-GI symptoms of headache and tiredness, which figure prominently in the disease but aren’t included in the older Celiac Disease Gastrointestinal Symptom Rating Scale.
Patients in the larazotide 0.5-mg t.i.d. group showed a significant 26% reduction in the average weekly number of CeD PRO symptomatic days, compared with placebo-treated controls. Twenty-nine percent of patients in the larazotide 0.5-mg group also achieved at least a 50% decrease from baseline in the weekly average CeD PRO abdominal symptom domain scores for at least 6 of the 12 weeks of treatment, significantly greater than the 14% rate with placebo. In addition, the larazotide 0.5-mg t.i.d. group had a 31% increase in the average number of CeD PRO days with minimal or no symptoms, compared with controls. Moreover, patients on the lowest dose of larazotide showed a significant reduction in CeD PRO non-GI symptoms, the only study arm to do so.
Of note, patients on the two higher doses of larazotide didn’t significantly outperform the control group in any of these study endpoints, although favorable trends were seen.
No changes in serially measured levels of anti–tissue transglutaminase antibodies or anti–deaminated gliadin antibodies were seen in any treatment arm.
Larazotide is an oral peptide that prevents the opening of epithelial tight junctions in the small bowel, an event that occurs in response to gluten exposure and inflammatory cytokines in patients with celiac disease. When these tight junctions are open inappropriately, the result is increased intestinal permeability and inflammation.
One audience member expressed reservations about the finding that only the lowest tested dose in a dose-ranging study proved effective.
"I’m troubled by drugs that don’t show a dose-response curve. This positive result could just be the play of chance," he cautioned.
Dr. Murray replied that he believes the consistency of the positive results with the 0.5-mg dose across multiple endpoints reduces that likelihood. Also, the lowest tested dose was the one that was effective in all the prior challenge studies. In any case, he added, the larger patient numbers planned for the phase III trials will sort out this issue.
Dr. Benjamin Lebwohl was enthusiastic about the larazotide results.
"It’s a very exciting time in the celiac disease community. Larazotide is one of several nondietary treatments currently in the pipeline," noted Dr. Lebwohl of the celiac disease center at Columbia University, New York.
The time is at hand, he added, to think big in terms of celiac disease research questions.
"Is it possible to prevent celiac disease in those individuals at increased risk? And among those individuals who come to us having been diagnosed with celiac disease, can we stop it in its tracks? Can we tolerize patients to gluten, allowing them to eat gluten either intermittently or even resume a full gluten-containing diet? Will there be a cure for celiac disease?" Dr. Lebwohl asked.
The study was supported by Alba Therapeutics. Dr. Murray reported serving as a consultant to or on the advisory boards of nearly a dozen pharmaceutical companies, Alba not among them. Dr. Lebwohl reported having no financial conflicts.
CHICAGO – Larazotide acetate, a first-in-class oral medication developed specifically to treat celiac disease, reduced both gastrointestinal and non-GI symptoms in patients who were symptomatic despite being on a gluten-free diet in a 74-site, randomized, double-blind phase II study.
"Safety and tolerability were comparable to placebo, setting this drug up, we believe, for phase III trials," Dr. Joseph A. Murray said at the annual Digestive Disease Week.
The study included 342 adult patients with celiac disease who had been on a gluten-free diet (GFD) for at least 12 months and continued on the GFD throughout the 12-week treatment period. Thus, this was a study of larazotide as an adjunct to the GFD, not as an alternative to it.
A GFD is of well-established efficacy as the sole proven therapy for celiac disease. However, the diet can be challenging. Surveys indicate 70% of celiac disease patients on a GFD experience recurrent symptoms due to inadvertent exposure or lapses in the strict adherence required. Thus, there is a significant unmet need for an effective pharmacologic therapy for this common disease, observed Dr. Murray, professor of medicine at the Mayo Clinic in Rochester, Minn.
Study participants were randomized to 12 weeks of double-blind treatment with larazotide at 0.5, 1, or 2 mg three times daily or to placebo. The study provided a successful opportunity to validate a new instrument designed for evaluation of celiac disease symptoms both in clinical trials and in daily practice. The instrument, known as the Celiac Disease Patient Reported Outcome (CeD PRO), entails daily assessment of symptoms in three key domains: abdominal symptoms such as cramping, bloating, and pain; diarrhea and loose stools; and the non-GI symptoms of headache and tiredness, which figure prominently in the disease but aren’t included in the older Celiac Disease Gastrointestinal Symptom Rating Scale.
Patients in the larazotide 0.5-mg t.i.d. group showed a significant 26% reduction in the average weekly number of CeD PRO symptomatic days, compared with placebo-treated controls. Twenty-nine percent of patients in the larazotide 0.5-mg group also achieved at least a 50% decrease from baseline in the weekly average CeD PRO abdominal symptom domain scores for at least 6 of the 12 weeks of treatment, significantly greater than the 14% rate with placebo. In addition, the larazotide 0.5-mg t.i.d. group had a 31% increase in the average number of CeD PRO days with minimal or no symptoms, compared with controls. Moreover, patients on the lowest dose of larazotide showed a significant reduction in CeD PRO non-GI symptoms, the only study arm to do so.
Of note, patients on the two higher doses of larazotide didn’t significantly outperform the control group in any of these study endpoints, although favorable trends were seen.
No changes in serially measured levels of anti–tissue transglutaminase antibodies or anti–deaminated gliadin antibodies were seen in any treatment arm.
Larazotide is an oral peptide that prevents the opening of epithelial tight junctions in the small bowel, an event that occurs in response to gluten exposure and inflammatory cytokines in patients with celiac disease. When these tight junctions are open inappropriately, the result is increased intestinal permeability and inflammation.
One audience member expressed reservations about the finding that only the lowest tested dose in a dose-ranging study proved effective.
"I’m troubled by drugs that don’t show a dose-response curve. This positive result could just be the play of chance," he cautioned.
Dr. Murray replied that he believes the consistency of the positive results with the 0.5-mg dose across multiple endpoints reduces that likelihood. Also, the lowest tested dose was the one that was effective in all the prior challenge studies. In any case, he added, the larger patient numbers planned for the phase III trials will sort out this issue.
Dr. Benjamin Lebwohl was enthusiastic about the larazotide results.
"It’s a very exciting time in the celiac disease community. Larazotide is one of several nondietary treatments currently in the pipeline," noted Dr. Lebwohl of the celiac disease center at Columbia University, New York.
The time is at hand, he added, to think big in terms of celiac disease research questions.
"Is it possible to prevent celiac disease in those individuals at increased risk? And among those individuals who come to us having been diagnosed with celiac disease, can we stop it in its tracks? Can we tolerize patients to gluten, allowing them to eat gluten either intermittently or even resume a full gluten-containing diet? Will there be a cure for celiac disease?" Dr. Lebwohl asked.
The study was supported by Alba Therapeutics. Dr. Murray reported serving as a consultant to or on the advisory boards of nearly a dozen pharmaceutical companies, Alba not among them. Dr. Lebwohl reported having no financial conflicts.
AT DDW 2014
Major finding: Patients with symptomatic celiac disease despite being on a gluten-free diet showed a 26% greater reduction in a composite symptom score combining GI and non-GI symptoms in response to oral larazotide acetate, compared with placebo-treated controls.
Data source: This was a randomized, double-blind, placebo-controlled, 74-site, phase II study including 342 adult celiac disease patients who were symptomatic despite being on a gluten-free diet.
Disclosures: The study was sponsored by Alba Therapeutics. Dr. Murray reported having no financial relationship with that company.
Phase III study: Prucalopride eases severe chronic constipation in men
CHICAGO – Prucalopride, a high-affinity serotonin 5-hydroxytryptamine4 receptor agonist, proved effective for the treatment of severe chronic constipation in men in a phase III clinical trial.
In this randomized, 66-site European trial involving 358 subjects on once-daily prucalopride at 2 mg or placebo, 37.9% of the prucalopride group achieved the primary endpoint of a mean of at least three spontaneous complete bowel movements per week over the course of the 12-week treatment period, significantly better than the 17.7% rate in controls, reported Dr. Kevin Etherson of Durham (England) University.
This was no small feat, he noted. Participants had a mean 9.2-year duration of chronic constipation. One-third of controls and 42% of the prucalopride group had experienced less than one spontaneous complete bowel movement per week during the 6 months prior to enrollment.
Prucalopride is already approved in the European Union for the treatment of chronic constipation in women for whom laxatives fail to provide adequate relief. The new phase III data pave the way for an expanded European indication including men. As for when U.S. physicians can expect to be able to prescribe the drug, a Shire spokesperson said the company is "working with the FDA [Food and Drug Administration] to create a regulatory pathway forward."
The new phase III results were consistent with the outcomes seen in the four published pivotal phase III trials conducted with the same endpoints in women, Dr. Etherson said at the annual Digestive Disease Week.
At baseline, 68% of the prucalopride group rated their constipation as "severe" or "very severe." After 12 weeks of active treatment, that was the case for only 22% of the men. At follow-up, 47% of the prucalopride group rated their global treatment efficacy as "quite a bit" or "extremely effective," compared with 30% of placebo-treated controls.
No significant ECG changes occurred during the study. Six men discontinued prucalopride, and seven stopped taking placebo. Gastrointestinal adverse events – mostly diarrhea, nausea, and abdominal pain – were reported by 20% of the men on prucalopride and 14% of the controls. Headaches and dizziness were also more common in the prucalopride group.
The phase III study was sponsored by Shire. Dr. Etherson reported having no financial conflicts of interest.
CHICAGO – Prucalopride, a high-affinity serotonin 5-hydroxytryptamine4 receptor agonist, proved effective for the treatment of severe chronic constipation in men in a phase III clinical trial.
In this randomized, 66-site European trial involving 358 subjects on once-daily prucalopride at 2 mg or placebo, 37.9% of the prucalopride group achieved the primary endpoint of a mean of at least three spontaneous complete bowel movements per week over the course of the 12-week treatment period, significantly better than the 17.7% rate in controls, reported Dr. Kevin Etherson of Durham (England) University.
This was no small feat, he noted. Participants had a mean 9.2-year duration of chronic constipation. One-third of controls and 42% of the prucalopride group had experienced less than one spontaneous complete bowel movement per week during the 6 months prior to enrollment.
Prucalopride is already approved in the European Union for the treatment of chronic constipation in women for whom laxatives fail to provide adequate relief. The new phase III data pave the way for an expanded European indication including men. As for when U.S. physicians can expect to be able to prescribe the drug, a Shire spokesperson said the company is "working with the FDA [Food and Drug Administration] to create a regulatory pathway forward."
The new phase III results were consistent with the outcomes seen in the four published pivotal phase III trials conducted with the same endpoints in women, Dr. Etherson said at the annual Digestive Disease Week.
At baseline, 68% of the prucalopride group rated their constipation as "severe" or "very severe." After 12 weeks of active treatment, that was the case for only 22% of the men. At follow-up, 47% of the prucalopride group rated their global treatment efficacy as "quite a bit" or "extremely effective," compared with 30% of placebo-treated controls.
No significant ECG changes occurred during the study. Six men discontinued prucalopride, and seven stopped taking placebo. Gastrointestinal adverse events – mostly diarrhea, nausea, and abdominal pain – were reported by 20% of the men on prucalopride and 14% of the controls. Headaches and dizziness were also more common in the prucalopride group.
The phase III study was sponsored by Shire. Dr. Etherson reported having no financial conflicts of interest.
CHICAGO – Prucalopride, a high-affinity serotonin 5-hydroxytryptamine4 receptor agonist, proved effective for the treatment of severe chronic constipation in men in a phase III clinical trial.
In this randomized, 66-site European trial involving 358 subjects on once-daily prucalopride at 2 mg or placebo, 37.9% of the prucalopride group achieved the primary endpoint of a mean of at least three spontaneous complete bowel movements per week over the course of the 12-week treatment period, significantly better than the 17.7% rate in controls, reported Dr. Kevin Etherson of Durham (England) University.
This was no small feat, he noted. Participants had a mean 9.2-year duration of chronic constipation. One-third of controls and 42% of the prucalopride group had experienced less than one spontaneous complete bowel movement per week during the 6 months prior to enrollment.
Prucalopride is already approved in the European Union for the treatment of chronic constipation in women for whom laxatives fail to provide adequate relief. The new phase III data pave the way for an expanded European indication including men. As for when U.S. physicians can expect to be able to prescribe the drug, a Shire spokesperson said the company is "working with the FDA [Food and Drug Administration] to create a regulatory pathway forward."
The new phase III results were consistent with the outcomes seen in the four published pivotal phase III trials conducted with the same endpoints in women, Dr. Etherson said at the annual Digestive Disease Week.
At baseline, 68% of the prucalopride group rated their constipation as "severe" or "very severe." After 12 weeks of active treatment, that was the case for only 22% of the men. At follow-up, 47% of the prucalopride group rated their global treatment efficacy as "quite a bit" or "extremely effective," compared with 30% of placebo-treated controls.
No significant ECG changes occurred during the study. Six men discontinued prucalopride, and seven stopped taking placebo. Gastrointestinal adverse events – mostly diarrhea, nausea, and abdominal pain – were reported by 20% of the men on prucalopride and 14% of the controls. Headaches and dizziness were also more common in the prucalopride group.
The phase III study was sponsored by Shire. Dr. Etherson reported having no financial conflicts of interest.
AT DDW 2014
Major finding: Thirty-eight percent of men with longstanding severe chronic constipation achieved a mean of three or more spontaneous complete bowel movements per week while being treated with prucalopride, compared with 17.7% on placebo.
Data source: This was a phase III, randomized, double-blind clinical trial in which 358 evaluable men with chronic constipation received prucalopride at 2 mg once daily or placebo for 12 weeks.
Disclosures: The phase III trial was sponsored by Shire. The presenter reported having no financial conflicts.