User login
GERD and sleep disorders often go hand in glove
DENVER – Gastroesophageal reflux disease is fertile soil for medical and psychiatric comorbid conditions, with sleep disorders leading the way, Maurice M. Ohayon, MD, reported at the annual meeting of the Associated Professional Sleep Societies.
He presented a very large, longitudinal, population-based study of gastroesophageal reflux disease (GERD) and its fellow travelers. The study entailed telephone interviews with 12,218 nationally representative subjects in 8 states at time 1, and reinterviews with 10,830 of them 3 years later. The interviews were guided by Sleep-EVAL, a validated computerized diagnostic interview system.
At time 1, 10.6% of subjects reported that they were told by a physician they have GERD and/or they were taking a medication for it. At time 2 (3 years later), the prevalence had increased to 12.4%, according to Dr. Ohayon, professor of psychiatry and behavioral sciences, chief of the division of public mental health and population sciences, and director of the sleep epidemiology research center at Stanford (Calif.) University.
Chronic GERD – that is, GERD at both time points – was present in 3.9% of subjects; 6% of subjects were remitters, meaning that they had GERD at time 1 but not at time 2. Another 8.5% of the total sample had incident GERD, meaning they had GERD at time 2 but not time 1.
The prevalence of GERD rose stepwise with increasing age, from the low single digits in 18- to 34-year-olds to a peak in the 55- to 64-year-old age group, where the prevalence was 13.5% at time 1 and 17% at time 2.

GERD was significantly more common in women. At time 2, the prevalence was 10% in men and 14.4% in women.
Nocturnal awakening was the sleep disorder symptom most commonly associated with GERD. It was a complaint in 28% of subjects who never had GERD, 46% of those with GERD at time 1, and 40% of those with GERD at time 2.
Insomnia, obstructive sleep apnea, and restless legs syndrome were also significantly more common among subjects with GERD. So were musculoskeletal and connective tissue diseases, hypertension, hypercholesterolemia, and major depressive disorder.
The study was supported by the John Arrillaga Foundation and Takeda Pharmaceuticals. Dr. Ohayon reported no financial conflicts of interest.
DENVER – Gastroesophageal reflux disease is fertile soil for medical and psychiatric comorbid conditions, with sleep disorders leading the way, Maurice M. Ohayon, MD, reported at the annual meeting of the Associated Professional Sleep Societies.
He presented a very large, longitudinal, population-based study of gastroesophageal reflux disease (GERD) and its fellow travelers. The study entailed telephone interviews with 12,218 nationally representative subjects in 8 states at time 1, and reinterviews with 10,830 of them 3 years later. The interviews were guided by Sleep-EVAL, a validated computerized diagnostic interview system.
At time 1, 10.6% of subjects reported that they were told by a physician they have GERD and/or they were taking a medication for it. At time 2 (3 years later), the prevalence had increased to 12.4%, according to Dr. Ohayon, professor of psychiatry and behavioral sciences, chief of the division of public mental health and population sciences, and director of the sleep epidemiology research center at Stanford (Calif.) University.
Chronic GERD – that is, GERD at both time points – was present in 3.9% of subjects; 6% of subjects were remitters, meaning that they had GERD at time 1 but not at time 2. Another 8.5% of the total sample had incident GERD, meaning they had GERD at time 2 but not time 1.
The prevalence of GERD rose stepwise with increasing age, from the low single digits in 18- to 34-year-olds to a peak in the 55- to 64-year-old age group, where the prevalence was 13.5% at time 1 and 17% at time 2.

GERD was significantly more common in women. At time 2, the prevalence was 10% in men and 14.4% in women.
Nocturnal awakening was the sleep disorder symptom most commonly associated with GERD. It was a complaint in 28% of subjects who never had GERD, 46% of those with GERD at time 1, and 40% of those with GERD at time 2.
Insomnia, obstructive sleep apnea, and restless legs syndrome were also significantly more common among subjects with GERD. So were musculoskeletal and connective tissue diseases, hypertension, hypercholesterolemia, and major depressive disorder.
The study was supported by the John Arrillaga Foundation and Takeda Pharmaceuticals. Dr. Ohayon reported no financial conflicts of interest.
DENVER – Gastroesophageal reflux disease is fertile soil for medical and psychiatric comorbid conditions, with sleep disorders leading the way, Maurice M. Ohayon, MD, reported at the annual meeting of the Associated Professional Sleep Societies.
He presented a very large, longitudinal, population-based study of gastroesophageal reflux disease (GERD) and its fellow travelers. The study entailed telephone interviews with 12,218 nationally representative subjects in 8 states at time 1, and reinterviews with 10,830 of them 3 years later. The interviews were guided by Sleep-EVAL, a validated computerized diagnostic interview system.
At time 1, 10.6% of subjects reported that they were told by a physician they have GERD and/or they were taking a medication for it. At time 2 (3 years later), the prevalence had increased to 12.4%, according to Dr. Ohayon, professor of psychiatry and behavioral sciences, chief of the division of public mental health and population sciences, and director of the sleep epidemiology research center at Stanford (Calif.) University.
Chronic GERD – that is, GERD at both time points – was present in 3.9% of subjects; 6% of subjects were remitters, meaning that they had GERD at time 1 but not at time 2. Another 8.5% of the total sample had incident GERD, meaning they had GERD at time 2 but not time 1.
The prevalence of GERD rose stepwise with increasing age, from the low single digits in 18- to 34-year-olds to a peak in the 55- to 64-year-old age group, where the prevalence was 13.5% at time 1 and 17% at time 2.

GERD was significantly more common in women. At time 2, the prevalence was 10% in men and 14.4% in women.
Nocturnal awakening was the sleep disorder symptom most commonly associated with GERD. It was a complaint in 28% of subjects who never had GERD, 46% of those with GERD at time 1, and 40% of those with GERD at time 2.
Insomnia, obstructive sleep apnea, and restless legs syndrome were also significantly more common among subjects with GERD. So were musculoskeletal and connective tissue diseases, hypertension, hypercholesterolemia, and major depressive disorder.
The study was supported by the John Arrillaga Foundation and Takeda Pharmaceuticals. Dr. Ohayon reported no financial conflicts of interest.
AT SLEEP 2016
Key clinical point: Inquire about potentially treatable sleep disturbances in patients with GERD.
Major finding: Nocturnal awakenings were reported to be a problem in 28% of subjects with no history of gastroesophageal reflux disease, but in up to 46% of those with the digestive disease.
Data source: A longitudinal, population-based study of 12,218 nationally representative adults interviewed regarding the prevalence of GERD as well as comorbid sleep, medical, and psychiatric conditions.
Disclosures: The study was supported by the John Arrillaga Foundation and Takeda Pharmaceuticals. The presenter reported having no financial conflicts of interest.
Expert: Screen military spouses for sleep problems
DENVER – Sleep problems and their host of deleterious physical and psychosocial consequences are pervasive among the civilian female spouses of U.S. military service members, according to the first large study to examine the issue.
“Our findings suggest that if we’re trying to promote the resilience and adjustment of military spouses and their families – particularly in light of 14 years of protracted overseas combat, where many families are experiencing deployment – sleep might be a really important target,” Wendy M. Troxel, PhD, said while presenting the study results at the annual meeting of the Associated Professional Sleep Societies.
The study population consisted of a nationally representative group of 1,805 female civilian spouses of military service members. Forty-four percent reported short sleep duration, meaning 6 hours or less per night. Another 18% got 5 hours or less. The Centers for Disease Control and Prevention and the American Academy of Sleep Medicine recommend 7 hours or more, noted Dr. Troxel, senior behavioral and social scientist at RAND Corporation in Pittsburgh.
More than half (54%) of military spouses reported that their sleep problems contributed to daytime impairment, such as intolerance of their spouse or children, frequent crying, or suboptimal performance at work or chores. One-third of the spouses reported feeling daytime fatigue three or more times per week, and another 29% experienced daytime fatigue one or two days per week.
As has been observed in other studies, sleep problems in military spouses were associated with psychosocial impairment. In linear regression analyses, the spouses with poor sleep quality, daytime impairment, and/or fatigue had significantly more depressive symptoms on the Patient Health Questionnaire–8, lower marital satisfaction, and poor self-rated health. Women with shorter sleep duration had more depressive symptoms and poorer self-rated health, but not lower marital satisfaction.
The spouses of services members currently deployed in combat zones had significantly worse sleep quality as measured on the Pittsburgh Sleep Quality Index compared with spouses of previously or never-deployed service members. But for the other outcomes of interest – sleep duration, daytime impairment, and fatigue – there were no differences based upon deployment status.
“This shows that it’s not just about the stress of combat deployment, it’s also about the stresses of everyday military life. Military families experience a great deal of stress that could lead to sleep disturbances whether or not a service member has been deployed: frequent residential moves, very long and unpredictable work hours, demanding jobs, threatening training environments,” Dr. Troxel said. “I think we need to be thinking about sleep as an important health indicator of military families in general across the deployment cycle.”
The study results are a call to action, she added.
“These findings highlight the importance of targeted screening, prevention, and intervention methods for military spouses. And primary care is where most people present with sleep problems,” Dr. Troxel said.
There are formal screening instruments for sleep problems, but in her view they really need to be better validated for use in primary care before widespread adoption.
“Simply having providers ask three quick questions about their patients’ sleep and daytime function is a good, practical approach: How’s the quality of your sleep? How much do you sleep on average? Do you have enough energy during the daytime to get through your day-to-day functioning? That’s informative enough to indicate the utility of moving on to longer screening tools or to referral for evidence-based treatments,” she said.
Dr. Troxel noted successful intervention in sleep problems in civilian spouses is a priority for reasons beyond their personal well-being. “Civilian spouses are most often the primary caretakers for service members who return from war with either invisible mental health wounds or physical health wounds,” she said. “And we’re expecting to have need for a lot of caretakers.”
The RAND National Defense Research Institute, the Office of the Surgeon General, the U.S. Army, and the Defense Centers of Excellence for Psychological Health and Traumatic Brain Injury funded the study.
DENVER – Sleep problems and their host of deleterious physical and psychosocial consequences are pervasive among the civilian female spouses of U.S. military service members, according to the first large study to examine the issue.
“Our findings suggest that if we’re trying to promote the resilience and adjustment of military spouses and their families – particularly in light of 14 years of protracted overseas combat, where many families are experiencing deployment – sleep might be a really important target,” Wendy M. Troxel, PhD, said while presenting the study results at the annual meeting of the Associated Professional Sleep Societies.
The study population consisted of a nationally representative group of 1,805 female civilian spouses of military service members. Forty-four percent reported short sleep duration, meaning 6 hours or less per night. Another 18% got 5 hours or less. The Centers for Disease Control and Prevention and the American Academy of Sleep Medicine recommend 7 hours or more, noted Dr. Troxel, senior behavioral and social scientist at RAND Corporation in Pittsburgh.
More than half (54%) of military spouses reported that their sleep problems contributed to daytime impairment, such as intolerance of their spouse or children, frequent crying, or suboptimal performance at work or chores. One-third of the spouses reported feeling daytime fatigue three or more times per week, and another 29% experienced daytime fatigue one or two days per week.
As has been observed in other studies, sleep problems in military spouses were associated with psychosocial impairment. In linear regression analyses, the spouses with poor sleep quality, daytime impairment, and/or fatigue had significantly more depressive symptoms on the Patient Health Questionnaire–8, lower marital satisfaction, and poor self-rated health. Women with shorter sleep duration had more depressive symptoms and poorer self-rated health, but not lower marital satisfaction.
The spouses of services members currently deployed in combat zones had significantly worse sleep quality as measured on the Pittsburgh Sleep Quality Index compared with spouses of previously or never-deployed service members. But for the other outcomes of interest – sleep duration, daytime impairment, and fatigue – there were no differences based upon deployment status.
“This shows that it’s not just about the stress of combat deployment, it’s also about the stresses of everyday military life. Military families experience a great deal of stress that could lead to sleep disturbances whether or not a service member has been deployed: frequent residential moves, very long and unpredictable work hours, demanding jobs, threatening training environments,” Dr. Troxel said. “I think we need to be thinking about sleep as an important health indicator of military families in general across the deployment cycle.”
The study results are a call to action, she added.
“These findings highlight the importance of targeted screening, prevention, and intervention methods for military spouses. And primary care is where most people present with sleep problems,” Dr. Troxel said.
There are formal screening instruments for sleep problems, but in her view they really need to be better validated for use in primary care before widespread adoption.
“Simply having providers ask three quick questions about their patients’ sleep and daytime function is a good, practical approach: How’s the quality of your sleep? How much do you sleep on average? Do you have enough energy during the daytime to get through your day-to-day functioning? That’s informative enough to indicate the utility of moving on to longer screening tools or to referral for evidence-based treatments,” she said.
Dr. Troxel noted successful intervention in sleep problems in civilian spouses is a priority for reasons beyond their personal well-being. “Civilian spouses are most often the primary caretakers for service members who return from war with either invisible mental health wounds or physical health wounds,” she said. “And we’re expecting to have need for a lot of caretakers.”
The RAND National Defense Research Institute, the Office of the Surgeon General, the U.S. Army, and the Defense Centers of Excellence for Psychological Health and Traumatic Brain Injury funded the study.
DENVER – Sleep problems and their host of deleterious physical and psychosocial consequences are pervasive among the civilian female spouses of U.S. military service members, according to the first large study to examine the issue.
“Our findings suggest that if we’re trying to promote the resilience and adjustment of military spouses and their families – particularly in light of 14 years of protracted overseas combat, where many families are experiencing deployment – sleep might be a really important target,” Wendy M. Troxel, PhD, said while presenting the study results at the annual meeting of the Associated Professional Sleep Societies.
The study population consisted of a nationally representative group of 1,805 female civilian spouses of military service members. Forty-four percent reported short sleep duration, meaning 6 hours or less per night. Another 18% got 5 hours or less. The Centers for Disease Control and Prevention and the American Academy of Sleep Medicine recommend 7 hours or more, noted Dr. Troxel, senior behavioral and social scientist at RAND Corporation in Pittsburgh.
More than half (54%) of military spouses reported that their sleep problems contributed to daytime impairment, such as intolerance of their spouse or children, frequent crying, or suboptimal performance at work or chores. One-third of the spouses reported feeling daytime fatigue three or more times per week, and another 29% experienced daytime fatigue one or two days per week.
As has been observed in other studies, sleep problems in military spouses were associated with psychosocial impairment. In linear regression analyses, the spouses with poor sleep quality, daytime impairment, and/or fatigue had significantly more depressive symptoms on the Patient Health Questionnaire–8, lower marital satisfaction, and poor self-rated health. Women with shorter sleep duration had more depressive symptoms and poorer self-rated health, but not lower marital satisfaction.
The spouses of services members currently deployed in combat zones had significantly worse sleep quality as measured on the Pittsburgh Sleep Quality Index compared with spouses of previously or never-deployed service members. But for the other outcomes of interest – sleep duration, daytime impairment, and fatigue – there were no differences based upon deployment status.
“This shows that it’s not just about the stress of combat deployment, it’s also about the stresses of everyday military life. Military families experience a great deal of stress that could lead to sleep disturbances whether or not a service member has been deployed: frequent residential moves, very long and unpredictable work hours, demanding jobs, threatening training environments,” Dr. Troxel said. “I think we need to be thinking about sleep as an important health indicator of military families in general across the deployment cycle.”
The study results are a call to action, she added.
“These findings highlight the importance of targeted screening, prevention, and intervention methods for military spouses. And primary care is where most people present with sleep problems,” Dr. Troxel said.
There are formal screening instruments for sleep problems, but in her view they really need to be better validated for use in primary care before widespread adoption.
“Simply having providers ask three quick questions about their patients’ sleep and daytime function is a good, practical approach: How’s the quality of your sleep? How much do you sleep on average? Do you have enough energy during the daytime to get through your day-to-day functioning? That’s informative enough to indicate the utility of moving on to longer screening tools or to referral for evidence-based treatments,” she said.
Dr. Troxel noted successful intervention in sleep problems in civilian spouses is a priority for reasons beyond their personal well-being. “Civilian spouses are most often the primary caretakers for service members who return from war with either invisible mental health wounds or physical health wounds,” she said. “And we’re expecting to have need for a lot of caretakers.”
The RAND National Defense Research Institute, the Office of the Surgeon General, the U.S. Army, and the Defense Centers of Excellence for Psychological Health and Traumatic Brain Injury funded the study.
AT SLEEP 2016
Key clinical point: A significant portion of civilian female spouses of U.S. military members report short sleep duration and daytime impairment due to sleep problems.
Major finding: Fifty-four percent of a large group of civilian spouses of U.S. military service members reported daytime impairment due to sleep problems.
Data source: A cross-sectional study of the prevalence and consequences of sleep problems in 1,805 civilian female military spouses.
Disclosures: The RAND National Defense Research Institute, the Office of the Surgeon General, the U.S. Army, and the Defense Centers of Excellence for Psychological Health and Traumatic Brain Injury funded the study. Dr. Troxel is employed by RAND Corporation.
Bioabsorbable percutaneous device fully closes large femoral arteriotomies
PARIS – A new bioabsorbable sutureless device provides operators with a safe, simple, and dependable option for fully percutaneous closure of the large, 12-24 French femoral arteriotomies created for transcatheter aortic valve replacement or endovascular abdominal aortic aneurysm repair, Arne Schwindt, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
He presented the 12-month follow-up results of the FRONTIER II study of the Vivasure PerQseal device. The device recently received European marketing approval on the strength of FRONTIER II but remains investigational in the United States.
The PerQseal device is deployed in less than 1 minute at the end of the primary procedure and achieves immediate hemostasis. It’s a simple three-step deployment process with essentially no learning curve, as evidenced by the fact that in FRONTIER II, the technical success rate starting from no experience was 97%, explained Dr. Schwindt of St. Franziskus Hospital in Muenster, Germany.
The large femoral arteriotomies created for TAVR or endovascular abdominal aortic aneurysm repair have typically required surgical cut down and sutured repair with a 3- to 5-cm incision. Vascular complications have been common. Indeed, the literature shows this method entails on average a 14.7% rate of major vascular complications up to 3 months post procedure, so that was the bar set in FRONTIER II: In order for the PerQseal to be deemed noninferior to surgical cut down and sutured repair, the rate of major complications directly related to the novel device at 3 months follow-up could be no greater than 14.7%. The current Valve Academic Research Consortium (VARC) 2 definition of major complications was used.
In fact, the vascular complication rate proved to be zero. Moreover, at 12 months of follow-up, no cases of groin fibrosis or scarring had been observed, Dr. Schwindt reported.
FRONTIER II was a single-arm, prospective, multicenter European study of 58 patients who received the PerQseal device for 66 closures. They were evaluated at discharge and 1, 3, and 12 months post procedure by Doppler ultrasound, with uniformly unremarkable findings.
PerQseal features a synthetic low-profile implant with over-the-wire delivery. The implant is loaded into a sheath, released by pulling back on the sheath, then pulled up against the arteriotomy from the inside. Blood pressure molds the device to the arterial wall and seals it. The device is fully absorbed in 180 days.
Session co-chair Dr. Ted E. Feldman was favorably impressed.
“This is really terrific. It’s very nice to see we’re finally making progress with large-bore closure devices,” commented Dr. Feldman, professor of medicine at Northwestern University, Chicago.
Dr. Schwindt reported receiving a research grant from Vivasure, which sponsored the FRONTIER II study. In addition, he serves as a consultant to a half dozen medical device companies.
PARIS – A new bioabsorbable sutureless device provides operators with a safe, simple, and dependable option for fully percutaneous closure of the large, 12-24 French femoral arteriotomies created for transcatheter aortic valve replacement or endovascular abdominal aortic aneurysm repair, Arne Schwindt, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
He presented the 12-month follow-up results of the FRONTIER II study of the Vivasure PerQseal device. The device recently received European marketing approval on the strength of FRONTIER II but remains investigational in the United States.
The PerQseal device is deployed in less than 1 minute at the end of the primary procedure and achieves immediate hemostasis. It’s a simple three-step deployment process with essentially no learning curve, as evidenced by the fact that in FRONTIER II, the technical success rate starting from no experience was 97%, explained Dr. Schwindt of St. Franziskus Hospital in Muenster, Germany.
The large femoral arteriotomies created for TAVR or endovascular abdominal aortic aneurysm repair have typically required surgical cut down and sutured repair with a 3- to 5-cm incision. Vascular complications have been common. Indeed, the literature shows this method entails on average a 14.7% rate of major vascular complications up to 3 months post procedure, so that was the bar set in FRONTIER II: In order for the PerQseal to be deemed noninferior to surgical cut down and sutured repair, the rate of major complications directly related to the novel device at 3 months follow-up could be no greater than 14.7%. The current Valve Academic Research Consortium (VARC) 2 definition of major complications was used.
In fact, the vascular complication rate proved to be zero. Moreover, at 12 months of follow-up, no cases of groin fibrosis or scarring had been observed, Dr. Schwindt reported.
FRONTIER II was a single-arm, prospective, multicenter European study of 58 patients who received the PerQseal device for 66 closures. They were evaluated at discharge and 1, 3, and 12 months post procedure by Doppler ultrasound, with uniformly unremarkable findings.
PerQseal features a synthetic low-profile implant with over-the-wire delivery. The implant is loaded into a sheath, released by pulling back on the sheath, then pulled up against the arteriotomy from the inside. Blood pressure molds the device to the arterial wall and seals it. The device is fully absorbed in 180 days.
Session co-chair Dr. Ted E. Feldman was favorably impressed.
“This is really terrific. It’s very nice to see we’re finally making progress with large-bore closure devices,” commented Dr. Feldman, professor of medicine at Northwestern University, Chicago.
Dr. Schwindt reported receiving a research grant from Vivasure, which sponsored the FRONTIER II study. In addition, he serves as a consultant to a half dozen medical device companies.
PARIS – A new bioabsorbable sutureless device provides operators with a safe, simple, and dependable option for fully percutaneous closure of the large, 12-24 French femoral arteriotomies created for transcatheter aortic valve replacement or endovascular abdominal aortic aneurysm repair, Arne Schwindt, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
He presented the 12-month follow-up results of the FRONTIER II study of the Vivasure PerQseal device. The device recently received European marketing approval on the strength of FRONTIER II but remains investigational in the United States.
The PerQseal device is deployed in less than 1 minute at the end of the primary procedure and achieves immediate hemostasis. It’s a simple three-step deployment process with essentially no learning curve, as evidenced by the fact that in FRONTIER II, the technical success rate starting from no experience was 97%, explained Dr. Schwindt of St. Franziskus Hospital in Muenster, Germany.
The large femoral arteriotomies created for TAVR or endovascular abdominal aortic aneurysm repair have typically required surgical cut down and sutured repair with a 3- to 5-cm incision. Vascular complications have been common. Indeed, the literature shows this method entails on average a 14.7% rate of major vascular complications up to 3 months post procedure, so that was the bar set in FRONTIER II: In order for the PerQseal to be deemed noninferior to surgical cut down and sutured repair, the rate of major complications directly related to the novel device at 3 months follow-up could be no greater than 14.7%. The current Valve Academic Research Consortium (VARC) 2 definition of major complications was used.
In fact, the vascular complication rate proved to be zero. Moreover, at 12 months of follow-up, no cases of groin fibrosis or scarring had been observed, Dr. Schwindt reported.
FRONTIER II was a single-arm, prospective, multicenter European study of 58 patients who received the PerQseal device for 66 closures. They were evaluated at discharge and 1, 3, and 12 months post procedure by Doppler ultrasound, with uniformly unremarkable findings.
PerQseal features a synthetic low-profile implant with over-the-wire delivery. The implant is loaded into a sheath, released by pulling back on the sheath, then pulled up against the arteriotomy from the inside. Blood pressure molds the device to the arterial wall and seals it. The device is fully absorbed in 180 days.
Session co-chair Dr. Ted E. Feldman was favorably impressed.
“This is really terrific. It’s very nice to see we’re finally making progress with large-bore closure devices,” commented Dr. Feldman, professor of medicine at Northwestern University, Chicago.
Dr. Schwindt reported receiving a research grant from Vivasure, which sponsored the FRONTIER II study. In addition, he serves as a consultant to a half dozen medical device companies.
AT EUROPCR 2016
Key clinical point: The PerQseal device provides a simpler alternative to existing closure methods.
Major finding: No major vascular complications were seen during structured follow-up of recipients of the novel bioabsorbable fully percutaneous PerQseal device for closure of large-bore femoral arteriotomies.
Data source: FRONTIER II was a prospective, single-arm, 12-month multicenter study including 58 patients who underwent closure of 66 large femoral arteriotomies using the PerQseal device.
Disclosures: The presenter reported receiving a research grant from Vivasure, which sponsored the FRONTIER II study.
Most interventional cardiologists don’t fully grasp radiation risks
PARIS – What most interventional cardiologists and electrophysiologists do not know about their health risks due to occupational radiation exposure and how best to protect themselves could fill a book – or better still, make for an illuminating 2-hour expert panel discussion at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“There’s a problem of lack of awareness on the part of interventional cardiologists, and also of institutional insensitivity to the problem,” declared Emanuela Piccaluga, MD, an investigator in the eye-opening Healthy Cath Lab study. This Italian national study showed that cardiac catheterization laboratory staff had radiation exposure duration–dependent increased risks of cataracts, cancers, and skin lesions, as well as other radiogenic noncancer effects: anxiety and depression, hypertension, and hyperlipidemia.
Cardiologists at some European hospitals have to pay for their own lead aprons and other protective gear. And even if the hospital does pick up the bill, administrators often balk at authorizing replacement of a lead apron that has developed microfractures and cracks. They view these imperfections as cosmetic defects, unaware that the damage renders the apron less protective, according to Dr. Piccaluga of Niguarda Ca’ Granda Hospital in Milan.
Ariel Roguin, MD, head of interventional cardiology at Rambam Medical Center in Haifa, Israel, said every cardiologist working with radiation should understand the three principles of radiation reduction, which he refers to in shorthand as “TDS,” for Time, Distance, and Shielding. Radiation is here to stay in cardiology, he said, but interventionalists can maximize their safety by keeping the fluoroscopy time and number of acquired images down, standing as far away as possible from both the radiation source and patient while still getting the job done well, and using appropriate shielding routinely.
Dr. Roguin gained notoriety with his report that 26 of 30 interventional cardiologists with glioblastoma multiforme or other brain malignancies had left-hemisphere cancers and 1 had a midline malignancy; only 3 were right-sided (Eur Heart J. 2014 Mar;35[10]:599-600). That distribution is highly unlikely to be due to play of chance, given that an interventional cardiologist’s left side is the side that’s usually exposed to more radiation.
“We should form a wall against radiation. Apart from the leaded aprons, for every procedure we all should also use lead skirts going from the table to the floor to block backscatter, ceiling-mounted overhead radiation shields, special glasses to protect against cataracts, and thyroid collars. And it’s very important to wear a dosimeter with sound; it helps increase awareness of our exposure,” he said.
Dr. Roguin has been a pioneer in the use of a thin, 0.5-mm lead shield draped across the patient’s abdomen from the umbilicus down during radial-access angiography. In a 322-patient randomized trial, he and his coinvestigators showed that this practice results in a threefold reduction in radiation to the operator, albeit at the cost of doubling the patient’s radiation exposure (Catheter Cardiovasc Interv. 2015 Jun;85[7]:1164-70).
“We now routinely do our radials with the lead apron across the patient’s abdomen. We’ve reached the conclusion that we work with radiation in the cath lab every day for many years and the patient is there only once or twice in a lifetime, hopefully,” the cardiologist explained.
With the growing popularity of radial-access interventions, audience members wanted to know if there is an advantage in terms of radiation exposure to left versus right radial artery access. The answer is no, according to Dr. Roguin.
“There are several studies showing no difference in radiation exposure. Left radial artery access is faster, but you’re leaning on the patient and getting more radiation as a result, while with right radial access you have to do more catheter manipulation, which takes longer. Both approaches involve more radiation to the operator than the transfemoral approach,” he said.
Dr. Piccaluga presented highlights from the Healthy Cath Lab study, sponsored by the Italian Society of Invasive Cardiology and the Italian National Research Council’s Institute of Clinical Physiology. The study involved detailed self-administered questionnaires completed by 218 interventional cardiologists and electrophysiologists, 191 nurses, and 57 technicians regularly exposed to ionizing radiation in the cardiac cath lab for a median of 10 years, along with 280 unexposed controls.
A variety of health problems were more frequent in the cath lab personnel regularly exposed to radiation. Rates were consistently highest in the cardiologists, followed next by the cath lab nurses, and then the radiation technicians.
Rates of health problems were highest in the 227 individuals with at least 13 years of cath lab radiation exposure. For example, their adjusted risks of cataracts, hypertension, hypercholesterolemia, and cancers were respectively 9-, 1.7-, 2.9-, and 4.5-fold fold greater than in unexposed controls, as detailed in a recent report (Circ Cardiovasc Interv. 2016 April. doi: 10.1161/CIRCINTERVENTIONS.115.003273).
Dr. Piccaluga also shared data from several other pertinent recent studies in which she was a coinvestigator. In one, 83 cardiologists and nurses working in cardiac catheterization laboratories and 83 matched radiation-nonexposed controls completed a neuropsychological test battery. The radiation-exposed group scored significantly lower on measures of delayed recall, visual short-term memory, and verbal fluency, all of which are skills located in left hemisphere structures of the brain – the side with more exposure to ionizing radiation during interventional procedures (J Int Neuropsychol Soc. 2015 Oct;21[9]:670-6).
In another study, Dr. Piccaluga and her coinvestigators had participants perform an odor-sniffing test. Olfactory discrimination in the cardiac cath lab staffers was significantly diminished in a pattern that has been identified in other studies as an early signal of impending clinical onset of Alzheimer’s and Parkinson’s diseases (Int J Cardiol. 2014 Feb 15;171[3]:461-3).
And in yet another study, Dr. Piccaluga and her coworkers found that left and right carotid intima-media thickness as measured by ultrasound in cardiac cath lab personnel having high lifetime radiation exposure was significantly greater than in those with low exposure and in nonexposed controls. In the left carotid artery, but not the right, intimal-medial thickness was significantly correlated with a total occupational radiologic risk score.
Moreover, the Italian investigators found a significant reduction in leukocyte telomere length – a biomarker for accelerated vascular aging – in cardiac cath lab staff regularly exposed to ionizing radiation, compared with controls (JACC Cardiovasc Interv. 2015 Apr 20;8[4]:616-27).
All of these findings, she stressed, make a persuasive case for interventional cardiologists doing everything they can to protect themselves from unnecessary radiation exposure at all times.
How to best go about accomplishing this was the territory covered by Alaide Chieffo, MD, of San Raffaele Scientific Institute in Milan.
The patient-related factors germane to radiation dose – procedure complexity and body thickness – are outside physician control. But there are plenty of operator-dependent factors, including, for starters, procedural experience. In one classic study, Dr. Chieffo noted, Greek investigators showed that interventional cardiologists’ radiation exposure dose was 60% greater in their first year of practice than in their second year.
Distance from the patient is crucial, she observed, since the patient is the greatest source of radiation to the operator. If the operator is 35 cm from the patient, the radiation exposure is fourfold greater than at a distance of 70 cm. At a distance of 17.5 cm, the exposure intensity is 16-fold greater than at a 70-cm distance. And at 8.8 cm of distance, it’s 64 times greater.
Image acquisition is another key variable within the interventionalist’s control. Cine images entail 12- to 20-times greater radiation doses than those of fluoroscopy, so don’t resort to cine when fluoroscopy will do. Also, reducing the fluoroscopy frame rate from 15 to 7.5 frames per second significantly decreases the amount of radiation released while providing images of adequate quality for many procedures. Tight collimation, the use of manually inserted wedge filters, and thoughtful selection of tube angulations result in less radiation for both patient and physician. It has been shown that tube angulations that expose a patient to intense radiation levels increase the operator’s radiation exposure exponentially. The least-irradiating tube angulations are caudal posteroanterior 0°/30°– angulation for the left coronary main stem, cranial posteroanterior 0°/30°+ for the left anterior descending coronary artery bifurcation, and right anterior oblique views of 40° or more. Left anterior oblique projections are the most radiation intensive, according to a comprehensive study (J Am Coll Cardiol. 2004 Oct 6;44[7]:1420-8), Dr. Chieffo continued.
Panelist Ghada Mikhail, MD, of Imperial College London, said there is some relatively new operator protective gear available. She cited lightweight protective caps, for example, but an audience show of hands indicated almost no one uses them.
“Protectors for the breasts and gonads are available. You can wear them underneath the lead. The extra time to put them on is worthwhile,” she said.
“I think the risk of radiation is completely underestimated,” Dr. Mikhail added. “We have a responsibility to young trainees to teach them about radiation protection, which a lot of institutions and supervisors don’t do. That’s partly because a lot of them don’t know the details.”
All of the speakers indicated they had no financial conflicts regarding their presentations.
PARIS – What most interventional cardiologists and electrophysiologists do not know about their health risks due to occupational radiation exposure and how best to protect themselves could fill a book – or better still, make for an illuminating 2-hour expert panel discussion at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“There’s a problem of lack of awareness on the part of interventional cardiologists, and also of institutional insensitivity to the problem,” declared Emanuela Piccaluga, MD, an investigator in the eye-opening Healthy Cath Lab study. This Italian national study showed that cardiac catheterization laboratory staff had radiation exposure duration–dependent increased risks of cataracts, cancers, and skin lesions, as well as other radiogenic noncancer effects: anxiety and depression, hypertension, and hyperlipidemia.
Cardiologists at some European hospitals have to pay for their own lead aprons and other protective gear. And even if the hospital does pick up the bill, administrators often balk at authorizing replacement of a lead apron that has developed microfractures and cracks. They view these imperfections as cosmetic defects, unaware that the damage renders the apron less protective, according to Dr. Piccaluga of Niguarda Ca’ Granda Hospital in Milan.
Ariel Roguin, MD, head of interventional cardiology at Rambam Medical Center in Haifa, Israel, said every cardiologist working with radiation should understand the three principles of radiation reduction, which he refers to in shorthand as “TDS,” for Time, Distance, and Shielding. Radiation is here to stay in cardiology, he said, but interventionalists can maximize their safety by keeping the fluoroscopy time and number of acquired images down, standing as far away as possible from both the radiation source and patient while still getting the job done well, and using appropriate shielding routinely.
Dr. Roguin gained notoriety with his report that 26 of 30 interventional cardiologists with glioblastoma multiforme or other brain malignancies had left-hemisphere cancers and 1 had a midline malignancy; only 3 were right-sided (Eur Heart J. 2014 Mar;35[10]:599-600). That distribution is highly unlikely to be due to play of chance, given that an interventional cardiologist’s left side is the side that’s usually exposed to more radiation.
“We should form a wall against radiation. Apart from the leaded aprons, for every procedure we all should also use lead skirts going from the table to the floor to block backscatter, ceiling-mounted overhead radiation shields, special glasses to protect against cataracts, and thyroid collars. And it’s very important to wear a dosimeter with sound; it helps increase awareness of our exposure,” he said.
Dr. Roguin has been a pioneer in the use of a thin, 0.5-mm lead shield draped across the patient’s abdomen from the umbilicus down during radial-access angiography. In a 322-patient randomized trial, he and his coinvestigators showed that this practice results in a threefold reduction in radiation to the operator, albeit at the cost of doubling the patient’s radiation exposure (Catheter Cardiovasc Interv. 2015 Jun;85[7]:1164-70).
“We now routinely do our radials with the lead apron across the patient’s abdomen. We’ve reached the conclusion that we work with radiation in the cath lab every day for many years and the patient is there only once or twice in a lifetime, hopefully,” the cardiologist explained.
With the growing popularity of radial-access interventions, audience members wanted to know if there is an advantage in terms of radiation exposure to left versus right radial artery access. The answer is no, according to Dr. Roguin.
“There are several studies showing no difference in radiation exposure. Left radial artery access is faster, but you’re leaning on the patient and getting more radiation as a result, while with right radial access you have to do more catheter manipulation, which takes longer. Both approaches involve more radiation to the operator than the transfemoral approach,” he said.
Dr. Piccaluga presented highlights from the Healthy Cath Lab study, sponsored by the Italian Society of Invasive Cardiology and the Italian National Research Council’s Institute of Clinical Physiology. The study involved detailed self-administered questionnaires completed by 218 interventional cardiologists and electrophysiologists, 191 nurses, and 57 technicians regularly exposed to ionizing radiation in the cardiac cath lab for a median of 10 years, along with 280 unexposed controls.
A variety of health problems were more frequent in the cath lab personnel regularly exposed to radiation. Rates were consistently highest in the cardiologists, followed next by the cath lab nurses, and then the radiation technicians.
Rates of health problems were highest in the 227 individuals with at least 13 years of cath lab radiation exposure. For example, their adjusted risks of cataracts, hypertension, hypercholesterolemia, and cancers were respectively 9-, 1.7-, 2.9-, and 4.5-fold fold greater than in unexposed controls, as detailed in a recent report (Circ Cardiovasc Interv. 2016 April. doi: 10.1161/CIRCINTERVENTIONS.115.003273).
Dr. Piccaluga also shared data from several other pertinent recent studies in which she was a coinvestigator. In one, 83 cardiologists and nurses working in cardiac catheterization laboratories and 83 matched radiation-nonexposed controls completed a neuropsychological test battery. The radiation-exposed group scored significantly lower on measures of delayed recall, visual short-term memory, and verbal fluency, all of which are skills located in left hemisphere structures of the brain – the side with more exposure to ionizing radiation during interventional procedures (J Int Neuropsychol Soc. 2015 Oct;21[9]:670-6).
In another study, Dr. Piccaluga and her coinvestigators had participants perform an odor-sniffing test. Olfactory discrimination in the cardiac cath lab staffers was significantly diminished in a pattern that has been identified in other studies as an early signal of impending clinical onset of Alzheimer’s and Parkinson’s diseases (Int J Cardiol. 2014 Feb 15;171[3]:461-3).
And in yet another study, Dr. Piccaluga and her coworkers found that left and right carotid intima-media thickness as measured by ultrasound in cardiac cath lab personnel having high lifetime radiation exposure was significantly greater than in those with low exposure and in nonexposed controls. In the left carotid artery, but not the right, intimal-medial thickness was significantly correlated with a total occupational radiologic risk score.
Moreover, the Italian investigators found a significant reduction in leukocyte telomere length – a biomarker for accelerated vascular aging – in cardiac cath lab staff regularly exposed to ionizing radiation, compared with controls (JACC Cardiovasc Interv. 2015 Apr 20;8[4]:616-27).
All of these findings, she stressed, make a persuasive case for interventional cardiologists doing everything they can to protect themselves from unnecessary radiation exposure at all times.
How to best go about accomplishing this was the territory covered by Alaide Chieffo, MD, of San Raffaele Scientific Institute in Milan.
The patient-related factors germane to radiation dose – procedure complexity and body thickness – are outside physician control. But there are plenty of operator-dependent factors, including, for starters, procedural experience. In one classic study, Dr. Chieffo noted, Greek investigators showed that interventional cardiologists’ radiation exposure dose was 60% greater in their first year of practice than in their second year.
Distance from the patient is crucial, she observed, since the patient is the greatest source of radiation to the operator. If the operator is 35 cm from the patient, the radiation exposure is fourfold greater than at a distance of 70 cm. At a distance of 17.5 cm, the exposure intensity is 16-fold greater than at a 70-cm distance. And at 8.8 cm of distance, it’s 64 times greater.
Image acquisition is another key variable within the interventionalist’s control. Cine images entail 12- to 20-times greater radiation doses than those of fluoroscopy, so don’t resort to cine when fluoroscopy will do. Also, reducing the fluoroscopy frame rate from 15 to 7.5 frames per second significantly decreases the amount of radiation released while providing images of adequate quality for many procedures. Tight collimation, the use of manually inserted wedge filters, and thoughtful selection of tube angulations result in less radiation for both patient and physician. It has been shown that tube angulations that expose a patient to intense radiation levels increase the operator’s radiation exposure exponentially. The least-irradiating tube angulations are caudal posteroanterior 0°/30°– angulation for the left coronary main stem, cranial posteroanterior 0°/30°+ for the left anterior descending coronary artery bifurcation, and right anterior oblique views of 40° or more. Left anterior oblique projections are the most radiation intensive, according to a comprehensive study (J Am Coll Cardiol. 2004 Oct 6;44[7]:1420-8), Dr. Chieffo continued.
Panelist Ghada Mikhail, MD, of Imperial College London, said there is some relatively new operator protective gear available. She cited lightweight protective caps, for example, but an audience show of hands indicated almost no one uses them.
“Protectors for the breasts and gonads are available. You can wear them underneath the lead. The extra time to put them on is worthwhile,” she said.
“I think the risk of radiation is completely underestimated,” Dr. Mikhail added. “We have a responsibility to young trainees to teach them about radiation protection, which a lot of institutions and supervisors don’t do. That’s partly because a lot of them don’t know the details.”
All of the speakers indicated they had no financial conflicts regarding their presentations.
PARIS – What most interventional cardiologists and electrophysiologists do not know about their health risks due to occupational radiation exposure and how best to protect themselves could fill a book – or better still, make for an illuminating 2-hour expert panel discussion at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“There’s a problem of lack of awareness on the part of interventional cardiologists, and also of institutional insensitivity to the problem,” declared Emanuela Piccaluga, MD, an investigator in the eye-opening Healthy Cath Lab study. This Italian national study showed that cardiac catheterization laboratory staff had radiation exposure duration–dependent increased risks of cataracts, cancers, and skin lesions, as well as other radiogenic noncancer effects: anxiety and depression, hypertension, and hyperlipidemia.
Cardiologists at some European hospitals have to pay for their own lead aprons and other protective gear. And even if the hospital does pick up the bill, administrators often balk at authorizing replacement of a lead apron that has developed microfractures and cracks. They view these imperfections as cosmetic defects, unaware that the damage renders the apron less protective, according to Dr. Piccaluga of Niguarda Ca’ Granda Hospital in Milan.
Ariel Roguin, MD, head of interventional cardiology at Rambam Medical Center in Haifa, Israel, said every cardiologist working with radiation should understand the three principles of radiation reduction, which he refers to in shorthand as “TDS,” for Time, Distance, and Shielding. Radiation is here to stay in cardiology, he said, but interventionalists can maximize their safety by keeping the fluoroscopy time and number of acquired images down, standing as far away as possible from both the radiation source and patient while still getting the job done well, and using appropriate shielding routinely.
Dr. Roguin gained notoriety with his report that 26 of 30 interventional cardiologists with glioblastoma multiforme or other brain malignancies had left-hemisphere cancers and 1 had a midline malignancy; only 3 were right-sided (Eur Heart J. 2014 Mar;35[10]:599-600). That distribution is highly unlikely to be due to play of chance, given that an interventional cardiologist’s left side is the side that’s usually exposed to more radiation.
“We should form a wall against radiation. Apart from the leaded aprons, for every procedure we all should also use lead skirts going from the table to the floor to block backscatter, ceiling-mounted overhead radiation shields, special glasses to protect against cataracts, and thyroid collars. And it’s very important to wear a dosimeter with sound; it helps increase awareness of our exposure,” he said.
Dr. Roguin has been a pioneer in the use of a thin, 0.5-mm lead shield draped across the patient’s abdomen from the umbilicus down during radial-access angiography. In a 322-patient randomized trial, he and his coinvestigators showed that this practice results in a threefold reduction in radiation to the operator, albeit at the cost of doubling the patient’s radiation exposure (Catheter Cardiovasc Interv. 2015 Jun;85[7]:1164-70).
“We now routinely do our radials with the lead apron across the patient’s abdomen. We’ve reached the conclusion that we work with radiation in the cath lab every day for many years and the patient is there only once or twice in a lifetime, hopefully,” the cardiologist explained.
With the growing popularity of radial-access interventions, audience members wanted to know if there is an advantage in terms of radiation exposure to left versus right radial artery access. The answer is no, according to Dr. Roguin.
“There are several studies showing no difference in radiation exposure. Left radial artery access is faster, but you’re leaning on the patient and getting more radiation as a result, while with right radial access you have to do more catheter manipulation, which takes longer. Both approaches involve more radiation to the operator than the transfemoral approach,” he said.
Dr. Piccaluga presented highlights from the Healthy Cath Lab study, sponsored by the Italian Society of Invasive Cardiology and the Italian National Research Council’s Institute of Clinical Physiology. The study involved detailed self-administered questionnaires completed by 218 interventional cardiologists and electrophysiologists, 191 nurses, and 57 technicians regularly exposed to ionizing radiation in the cardiac cath lab for a median of 10 years, along with 280 unexposed controls.
A variety of health problems were more frequent in the cath lab personnel regularly exposed to radiation. Rates were consistently highest in the cardiologists, followed next by the cath lab nurses, and then the radiation technicians.
Rates of health problems were highest in the 227 individuals with at least 13 years of cath lab radiation exposure. For example, their adjusted risks of cataracts, hypertension, hypercholesterolemia, and cancers were respectively 9-, 1.7-, 2.9-, and 4.5-fold fold greater than in unexposed controls, as detailed in a recent report (Circ Cardiovasc Interv. 2016 April. doi: 10.1161/CIRCINTERVENTIONS.115.003273).
Dr. Piccaluga also shared data from several other pertinent recent studies in which she was a coinvestigator. In one, 83 cardiologists and nurses working in cardiac catheterization laboratories and 83 matched radiation-nonexposed controls completed a neuropsychological test battery. The radiation-exposed group scored significantly lower on measures of delayed recall, visual short-term memory, and verbal fluency, all of which are skills located in left hemisphere structures of the brain – the side with more exposure to ionizing radiation during interventional procedures (J Int Neuropsychol Soc. 2015 Oct;21[9]:670-6).
In another study, Dr. Piccaluga and her coinvestigators had participants perform an odor-sniffing test. Olfactory discrimination in the cardiac cath lab staffers was significantly diminished in a pattern that has been identified in other studies as an early signal of impending clinical onset of Alzheimer’s and Parkinson’s diseases (Int J Cardiol. 2014 Feb 15;171[3]:461-3).
And in yet another study, Dr. Piccaluga and her coworkers found that left and right carotid intima-media thickness as measured by ultrasound in cardiac cath lab personnel having high lifetime radiation exposure was significantly greater than in those with low exposure and in nonexposed controls. In the left carotid artery, but not the right, intimal-medial thickness was significantly correlated with a total occupational radiologic risk score.
Moreover, the Italian investigators found a significant reduction in leukocyte telomere length – a biomarker for accelerated vascular aging – in cardiac cath lab staff regularly exposed to ionizing radiation, compared with controls (JACC Cardiovasc Interv. 2015 Apr 20;8[4]:616-27).
All of these findings, she stressed, make a persuasive case for interventional cardiologists doing everything they can to protect themselves from unnecessary radiation exposure at all times.
How to best go about accomplishing this was the territory covered by Alaide Chieffo, MD, of San Raffaele Scientific Institute in Milan.
The patient-related factors germane to radiation dose – procedure complexity and body thickness – are outside physician control. But there are plenty of operator-dependent factors, including, for starters, procedural experience. In one classic study, Dr. Chieffo noted, Greek investigators showed that interventional cardiologists’ radiation exposure dose was 60% greater in their first year of practice than in their second year.
Distance from the patient is crucial, she observed, since the patient is the greatest source of radiation to the operator. If the operator is 35 cm from the patient, the radiation exposure is fourfold greater than at a distance of 70 cm. At a distance of 17.5 cm, the exposure intensity is 16-fold greater than at a 70-cm distance. And at 8.8 cm of distance, it’s 64 times greater.
Image acquisition is another key variable within the interventionalist’s control. Cine images entail 12- to 20-times greater radiation doses than those of fluoroscopy, so don’t resort to cine when fluoroscopy will do. Also, reducing the fluoroscopy frame rate from 15 to 7.5 frames per second significantly decreases the amount of radiation released while providing images of adequate quality for many procedures. Tight collimation, the use of manually inserted wedge filters, and thoughtful selection of tube angulations result in less radiation for both patient and physician. It has been shown that tube angulations that expose a patient to intense radiation levels increase the operator’s radiation exposure exponentially. The least-irradiating tube angulations are caudal posteroanterior 0°/30°– angulation for the left coronary main stem, cranial posteroanterior 0°/30°+ for the left anterior descending coronary artery bifurcation, and right anterior oblique views of 40° or more. Left anterior oblique projections are the most radiation intensive, according to a comprehensive study (J Am Coll Cardiol. 2004 Oct 6;44[7]:1420-8), Dr. Chieffo continued.
Panelist Ghada Mikhail, MD, of Imperial College London, said there is some relatively new operator protective gear available. She cited lightweight protective caps, for example, but an audience show of hands indicated almost no one uses them.
“Protectors for the breasts and gonads are available. You can wear them underneath the lead. The extra time to put them on is worthwhile,” she said.
“I think the risk of radiation is completely underestimated,” Dr. Mikhail added. “We have a responsibility to young trainees to teach them about radiation protection, which a lot of institutions and supervisors don’t do. That’s partly because a lot of them don’t know the details.”
All of the speakers indicated they had no financial conflicts regarding their presentations.
EXPERT ANALYSIS FROM EUROPCR 2016
New risk score predicts PCI outcomes in octogenarians
PARIS – A fast and simple clinically based scoring system enables physicians to determine the chance of a successful outcome for octogenarians undergoing percutaneous coronary intervention, James Cockburn, MD, said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
All six elements of the scoring system are readily available either at the time the very elderly patient presents or at diagnostic angiography, noted Dr. Cockburn of Brighton and Sussex University Hospital in Brighton, England.
He and his coworkers developed the risk score through analysis of a registry of 44,221 patients aged 80 or older who underwent percutaneous coronary intervention (PCI). The procedural success rate – defined as less than a 30% residual stenosis and TIMI (Thrombolysis in Myocardial Infarction) 3 antegrade blood flow – was 92.3%. The 30-day mortality rate was 3.9%. The investigators teased out a set of easily accessible clinical factors associated with 30-day mortality and came up with a novel risk scoring system using a 9-point scale.
The clinical factors and scoring system are as follows:
• Age. 1 point for being 80-89, and 2 points at age 90 or older.
• Indication for PCI. 1 point for unstable angina/non–ST segment elevation MI, 2 points for STEMI, 0 points for other indications.
• Ventilated preprocedure. 1 point if yes.
• Creatinine level above 200 umol/L. 1 point for yes.
• Preprocedural cardiogenic shock. 2 points for yes.
• Poor left ventricular ejection fraction. If less than 30%, 1 point.
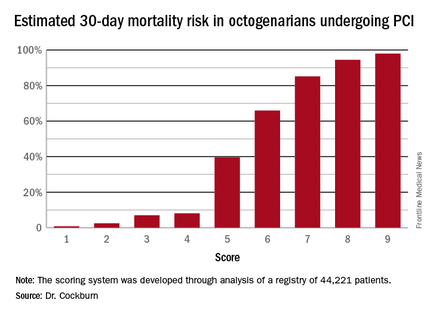
Thus, scores can range from 1 to 9. Dr. Cockburn and his coworkers calculated the risk of 30-day mortality for each possible score. They validated the score by performing a receiver operator curve analysis that showed an area under the curve of 0.83, suggestive of relatively high sensitivity and specificity.
A score of 4 or less suggests a very good chance of survival at 30 days. In contrast, a score of 6 was associated with a two in three chance of death by 30 days. And it’s not hard to reach a 6: A patient who is 90 years old (2 points), presents with STEMI (2 points), and is in cardiogenic shock (2 points) is already there. But if a 90-year-old patient presents with unstable angina and none of the other risk factors, that’s a score of 3 points, with an estimated probability of death at 30 days of only 7%, he noted.
Dr. Cockburn stressed that this risk score should not be used to base decisions on whether to take very elderly patients to the cardiac catheterization laboratory. “It enables you to have a useful conversation with relatives in which you can explain that this is a very high-risk intervention, or perhaps a low-risk intervention,” according to the cardiologist.
Discussants were emphatic in their agreement with Dr. Cockburn that this risk score shouldn’t be utilized to decide who does or doesn’t get PCI. One panelist said that what’s really lacking now in clinical practice – and where that huge British registry database could be helpful – is a scoring system that would predict which patients who don’t present in cardiogenic shock are going to develop it post PCI.
Dr. Cockburn reported having no relevant financial conflicts.
PARIS – A fast and simple clinically based scoring system enables physicians to determine the chance of a successful outcome for octogenarians undergoing percutaneous coronary intervention, James Cockburn, MD, said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
All six elements of the scoring system are readily available either at the time the very elderly patient presents or at diagnostic angiography, noted Dr. Cockburn of Brighton and Sussex University Hospital in Brighton, England.
He and his coworkers developed the risk score through analysis of a registry of 44,221 patients aged 80 or older who underwent percutaneous coronary intervention (PCI). The procedural success rate – defined as less than a 30% residual stenosis and TIMI (Thrombolysis in Myocardial Infarction) 3 antegrade blood flow – was 92.3%. The 30-day mortality rate was 3.9%. The investigators teased out a set of easily accessible clinical factors associated with 30-day mortality and came up with a novel risk scoring system using a 9-point scale.
The clinical factors and scoring system are as follows:
• Age. 1 point for being 80-89, and 2 points at age 90 or older.
• Indication for PCI. 1 point for unstable angina/non–ST segment elevation MI, 2 points for STEMI, 0 points for other indications.
• Ventilated preprocedure. 1 point if yes.
• Creatinine level above 200 umol/L. 1 point for yes.
• Preprocedural cardiogenic shock. 2 points for yes.
• Poor left ventricular ejection fraction. If less than 30%, 1 point.

Thus, scores can range from 1 to 9. Dr. Cockburn and his coworkers calculated the risk of 30-day mortality for each possible score. They validated the score by performing a receiver operator curve analysis that showed an area under the curve of 0.83, suggestive of relatively high sensitivity and specificity.
A score of 4 or less suggests a very good chance of survival at 30 days. In contrast, a score of 6 was associated with a two in three chance of death by 30 days. And it’s not hard to reach a 6: A patient who is 90 years old (2 points), presents with STEMI (2 points), and is in cardiogenic shock (2 points) is already there. But if a 90-year-old patient presents with unstable angina and none of the other risk factors, that’s a score of 3 points, with an estimated probability of death at 30 days of only 7%, he noted.
Dr. Cockburn stressed that this risk score should not be used to base decisions on whether to take very elderly patients to the cardiac catheterization laboratory. “It enables you to have a useful conversation with relatives in which you can explain that this is a very high-risk intervention, or perhaps a low-risk intervention,” according to the cardiologist.
Discussants were emphatic in their agreement with Dr. Cockburn that this risk score shouldn’t be utilized to decide who does or doesn’t get PCI. One panelist said that what’s really lacking now in clinical practice – and where that huge British registry database could be helpful – is a scoring system that would predict which patients who don’t present in cardiogenic shock are going to develop it post PCI.
Dr. Cockburn reported having no relevant financial conflicts.
PARIS – A fast and simple clinically based scoring system enables physicians to determine the chance of a successful outcome for octogenarians undergoing percutaneous coronary intervention, James Cockburn, MD, said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
All six elements of the scoring system are readily available either at the time the very elderly patient presents or at diagnostic angiography, noted Dr. Cockburn of Brighton and Sussex University Hospital in Brighton, England.
He and his coworkers developed the risk score through analysis of a registry of 44,221 patients aged 80 or older who underwent percutaneous coronary intervention (PCI). The procedural success rate – defined as less than a 30% residual stenosis and TIMI (Thrombolysis in Myocardial Infarction) 3 antegrade blood flow – was 92.3%. The 30-day mortality rate was 3.9%. The investigators teased out a set of easily accessible clinical factors associated with 30-day mortality and came up with a novel risk scoring system using a 9-point scale.
The clinical factors and scoring system are as follows:
• Age. 1 point for being 80-89, and 2 points at age 90 or older.
• Indication for PCI. 1 point for unstable angina/non–ST segment elevation MI, 2 points for STEMI, 0 points for other indications.
• Ventilated preprocedure. 1 point if yes.
• Creatinine level above 200 umol/L. 1 point for yes.
• Preprocedural cardiogenic shock. 2 points for yes.
• Poor left ventricular ejection fraction. If less than 30%, 1 point.

Thus, scores can range from 1 to 9. Dr. Cockburn and his coworkers calculated the risk of 30-day mortality for each possible score. They validated the score by performing a receiver operator curve analysis that showed an area under the curve of 0.83, suggestive of relatively high sensitivity and specificity.
A score of 4 or less suggests a very good chance of survival at 30 days. In contrast, a score of 6 was associated with a two in three chance of death by 30 days. And it’s not hard to reach a 6: A patient who is 90 years old (2 points), presents with STEMI (2 points), and is in cardiogenic shock (2 points) is already there. But if a 90-year-old patient presents with unstable angina and none of the other risk factors, that’s a score of 3 points, with an estimated probability of death at 30 days of only 7%, he noted.
Dr. Cockburn stressed that this risk score should not be used to base decisions on whether to take very elderly patients to the cardiac catheterization laboratory. “It enables you to have a useful conversation with relatives in which you can explain that this is a very high-risk intervention, or perhaps a low-risk intervention,” according to the cardiologist.
Discussants were emphatic in their agreement with Dr. Cockburn that this risk score shouldn’t be utilized to decide who does or doesn’t get PCI. One panelist said that what’s really lacking now in clinical practice – and where that huge British registry database could be helpful – is a scoring system that would predict which patients who don’t present in cardiogenic shock are going to develop it post PCI.
Dr. Cockburn reported having no relevant financial conflicts.
AT EUROPCR 2016
Key clinical point: Six readily obtainable clinical factors can be added up to allow accurate estimates of 30-day mortality risk after PCI.
Major finding: Very elderly patients with a score of 3 out of a possible 9 had an estimated 30-day mortality risk of 7%, while at a score of 5, the risk jumped to 40%, and at 6 to 66%.
Data source: This novel 30-day risk scoring system for octogenarians undergoing PCI was derived from a registry of 44,221 such patients.
Disclosures: The presenter reported having no relevant financial conflicts.
Sleep apnea in pregnancy linked to preterm birth
DENVER – Pregnant women with sleep apnea are more likely to have planned obstetric interventions, results of an Australian population-based cohort study suggest.
The study included all 636,227 in-hospital births during 2002-2012 in New South Wales, Australia’s most populous state. Maternal sleep apnea was also associated with increased rates of planned preterm birth, even though preterm birth is widely considered the greatest contributor to neonatal morbidity and mortality, Yu Sun Bin, PhD, said at the annual meeting of the Associated Professional Sleep Societies.
“Somewhere along the line, clinicians decided that the risks of preterm birth to the baby were outweighed by the risks to the mother of delivering at term,” said Dr. Bin of the University of Sydney.
She and her coinvestigators undertook this study because even though previous studies have linked maternal sleep apnea to increased risks of gestational diabetes and gestational hypertension, most of the prior studies were small, cross-sectional, and/or relied upon snoring as a proxy for sleep apnea, which many sleep specialists consider invalid.
The investigators compared maternal and infant outcomes for mothers with a documented diagnosis of sleep apnea – either central or obstructive – in the year before or during pregnancy with outcomes for mothers without that diagnosis.
There were 519 mothers with diagnosed sleep apnea, for a prevalence of 0.08%. That figure is low in light of other evidence, making it likely that the 635,708 women in the no-sleep-apnea group actually included a substantial number of mothers with undiagnosed sleep apnea. Thus, the investigators’ estimates of the adverse impacts of sleep apnea in pregnancy are “rather conservative,” according to Dr. Bin.
Australian women with sleep apnea were older and less healthy than mothers without sleep apnea were. They had higher baseline rates of obesity, preexisting diabetes, chronic hypertension, and were more likely to be smokers.
The incidence of pregnancy hypertension was 19.7% in the sleep apnea group and 8.7% in controls. In a multivariate regression analysis adjusted for potential confounders, the maternal sleep apnea group had a 40% greater risk of developing hypertension than did controls. However, contrary to previous smaller studies, they did not have a significantly increased rate of gestational diabetes.
Even after controlling for both pregnancy hypertension and gestational diabetes, the sleep apnea group still had a significant 15% increase in the relative likelihood of a planned delivery.
The rate of preterm birth at 36 weeks or earlier was 14.5% in the maternal sleep apnea group, compared with 6.9% in controls, for an adjusted 1.5-fold increased relative risk.
Perinatal death occurred in 1.9% of the sleep apnea group and 0.9% of controls; however, the resultant adjusted 1.73-fold increased risk didn’t attain statistical significance because of the small number of deaths in the study. Dr. Bin said she and her colleagues plan to further investigate this signal to learn whether it is real or an artifact of small numbers.
The incidence of 5-minute Apgar scores below 7 was 4.6% in the sleep apnea group, compared with 2.4% in controls, for an adjusted 1.6-fold increased risk. The rate of neonatal intensive care unit admission in the sleep apnea group was 27.9%, versus 16% in controls, for a 1.61-fold increased relative risk.
The NICU admission rate for preterm infants didn’t differ between the two groups. The difference occurred in term babies, whose NICU admission rate was 20.3% if they were in the sleep apnea group, but just 12.1% in the control group.
“This suggests that maternal sleep apnea is contributing to some condition in the baby that requires additional support,” Dr. Bin observed.
The nature of that condition, however, remains unclear, since all patient data available to the investigators was deidentified.
The incidence of small-for-gestational-age babies was similar in the sleep apnea and control groups. In contrast, the large-for-gestational-age rate was 15.2% in the sleep apnea group, compared with 9.1% in controls, for an adjusted 1.27-fold increased risk.
The two main limitations of the Australian study were the likely underdiagnosis of sleep apnea and the lack of any information on treatment of affected patients, according to Dr. Bin. A key unresolved question, she added, is whether interventions for maternal sleep apnea reduce the risks identified in the New South Wales study. She noted that one 16-patient randomized study of nasal continuous positive airway pressure suggests the answer is yes (Sleep Med. 2007 Dec;9:15-21).
The Australian National Health and Medical Research Council supported the study. Dr. Bin reported having no financial conflicts.
DENVER – Pregnant women with sleep apnea are more likely to have planned obstetric interventions, results of an Australian population-based cohort study suggest.
The study included all 636,227 in-hospital births during 2002-2012 in New South Wales, Australia’s most populous state. Maternal sleep apnea was also associated with increased rates of planned preterm birth, even though preterm birth is widely considered the greatest contributor to neonatal morbidity and mortality, Yu Sun Bin, PhD, said at the annual meeting of the Associated Professional Sleep Societies.
“Somewhere along the line, clinicians decided that the risks of preterm birth to the baby were outweighed by the risks to the mother of delivering at term,” said Dr. Bin of the University of Sydney.
She and her coinvestigators undertook this study because even though previous studies have linked maternal sleep apnea to increased risks of gestational diabetes and gestational hypertension, most of the prior studies were small, cross-sectional, and/or relied upon snoring as a proxy for sleep apnea, which many sleep specialists consider invalid.
The investigators compared maternal and infant outcomes for mothers with a documented diagnosis of sleep apnea – either central or obstructive – in the year before or during pregnancy with outcomes for mothers without that diagnosis.
There were 519 mothers with diagnosed sleep apnea, for a prevalence of 0.08%. That figure is low in light of other evidence, making it likely that the 635,708 women in the no-sleep-apnea group actually included a substantial number of mothers with undiagnosed sleep apnea. Thus, the investigators’ estimates of the adverse impacts of sleep apnea in pregnancy are “rather conservative,” according to Dr. Bin.
Australian women with sleep apnea were older and less healthy than mothers without sleep apnea were. They had higher baseline rates of obesity, preexisting diabetes, chronic hypertension, and were more likely to be smokers.
The incidence of pregnancy hypertension was 19.7% in the sleep apnea group and 8.7% in controls. In a multivariate regression analysis adjusted for potential confounders, the maternal sleep apnea group had a 40% greater risk of developing hypertension than did controls. However, contrary to previous smaller studies, they did not have a significantly increased rate of gestational diabetes.
Even after controlling for both pregnancy hypertension and gestational diabetes, the sleep apnea group still had a significant 15% increase in the relative likelihood of a planned delivery.
The rate of preterm birth at 36 weeks or earlier was 14.5% in the maternal sleep apnea group, compared with 6.9% in controls, for an adjusted 1.5-fold increased relative risk.
Perinatal death occurred in 1.9% of the sleep apnea group and 0.9% of controls; however, the resultant adjusted 1.73-fold increased risk didn’t attain statistical significance because of the small number of deaths in the study. Dr. Bin said she and her colleagues plan to further investigate this signal to learn whether it is real or an artifact of small numbers.
The incidence of 5-minute Apgar scores below 7 was 4.6% in the sleep apnea group, compared with 2.4% in controls, for an adjusted 1.6-fold increased risk. The rate of neonatal intensive care unit admission in the sleep apnea group was 27.9%, versus 16% in controls, for a 1.61-fold increased relative risk.
The NICU admission rate for preterm infants didn’t differ between the two groups. The difference occurred in term babies, whose NICU admission rate was 20.3% if they were in the sleep apnea group, but just 12.1% in the control group.
“This suggests that maternal sleep apnea is contributing to some condition in the baby that requires additional support,” Dr. Bin observed.
The nature of that condition, however, remains unclear, since all patient data available to the investigators was deidentified.
The incidence of small-for-gestational-age babies was similar in the sleep apnea and control groups. In contrast, the large-for-gestational-age rate was 15.2% in the sleep apnea group, compared with 9.1% in controls, for an adjusted 1.27-fold increased risk.
The two main limitations of the Australian study were the likely underdiagnosis of sleep apnea and the lack of any information on treatment of affected patients, according to Dr. Bin. A key unresolved question, she added, is whether interventions for maternal sleep apnea reduce the risks identified in the New South Wales study. She noted that one 16-patient randomized study of nasal continuous positive airway pressure suggests the answer is yes (Sleep Med. 2007 Dec;9:15-21).
The Australian National Health and Medical Research Council supported the study. Dr. Bin reported having no financial conflicts.
DENVER – Pregnant women with sleep apnea are more likely to have planned obstetric interventions, results of an Australian population-based cohort study suggest.
The study included all 636,227 in-hospital births during 2002-2012 in New South Wales, Australia’s most populous state. Maternal sleep apnea was also associated with increased rates of planned preterm birth, even though preterm birth is widely considered the greatest contributor to neonatal morbidity and mortality, Yu Sun Bin, PhD, said at the annual meeting of the Associated Professional Sleep Societies.
“Somewhere along the line, clinicians decided that the risks of preterm birth to the baby were outweighed by the risks to the mother of delivering at term,” said Dr. Bin of the University of Sydney.
She and her coinvestigators undertook this study because even though previous studies have linked maternal sleep apnea to increased risks of gestational diabetes and gestational hypertension, most of the prior studies were small, cross-sectional, and/or relied upon snoring as a proxy for sleep apnea, which many sleep specialists consider invalid.
The investigators compared maternal and infant outcomes for mothers with a documented diagnosis of sleep apnea – either central or obstructive – in the year before or during pregnancy with outcomes for mothers without that diagnosis.
There were 519 mothers with diagnosed sleep apnea, for a prevalence of 0.08%. That figure is low in light of other evidence, making it likely that the 635,708 women in the no-sleep-apnea group actually included a substantial number of mothers with undiagnosed sleep apnea. Thus, the investigators’ estimates of the adverse impacts of sleep apnea in pregnancy are “rather conservative,” according to Dr. Bin.
Australian women with sleep apnea were older and less healthy than mothers without sleep apnea were. They had higher baseline rates of obesity, preexisting diabetes, chronic hypertension, and were more likely to be smokers.
The incidence of pregnancy hypertension was 19.7% in the sleep apnea group and 8.7% in controls. In a multivariate regression analysis adjusted for potential confounders, the maternal sleep apnea group had a 40% greater risk of developing hypertension than did controls. However, contrary to previous smaller studies, they did not have a significantly increased rate of gestational diabetes.
Even after controlling for both pregnancy hypertension and gestational diabetes, the sleep apnea group still had a significant 15% increase in the relative likelihood of a planned delivery.
The rate of preterm birth at 36 weeks or earlier was 14.5% in the maternal sleep apnea group, compared with 6.9% in controls, for an adjusted 1.5-fold increased relative risk.
Perinatal death occurred in 1.9% of the sleep apnea group and 0.9% of controls; however, the resultant adjusted 1.73-fold increased risk didn’t attain statistical significance because of the small number of deaths in the study. Dr. Bin said she and her colleagues plan to further investigate this signal to learn whether it is real or an artifact of small numbers.
The incidence of 5-minute Apgar scores below 7 was 4.6% in the sleep apnea group, compared with 2.4% in controls, for an adjusted 1.6-fold increased risk. The rate of neonatal intensive care unit admission in the sleep apnea group was 27.9%, versus 16% in controls, for a 1.61-fold increased relative risk.
The NICU admission rate for preterm infants didn’t differ between the two groups. The difference occurred in term babies, whose NICU admission rate was 20.3% if they were in the sleep apnea group, but just 12.1% in the control group.
“This suggests that maternal sleep apnea is contributing to some condition in the baby that requires additional support,” Dr. Bin observed.
The nature of that condition, however, remains unclear, since all patient data available to the investigators was deidentified.
The incidence of small-for-gestational-age babies was similar in the sleep apnea and control groups. In contrast, the large-for-gestational-age rate was 15.2% in the sleep apnea group, compared with 9.1% in controls, for an adjusted 1.27-fold increased risk.
The two main limitations of the Australian study were the likely underdiagnosis of sleep apnea and the lack of any information on treatment of affected patients, according to Dr. Bin. A key unresolved question, she added, is whether interventions for maternal sleep apnea reduce the risks identified in the New South Wales study. She noted that one 16-patient randomized study of nasal continuous positive airway pressure suggests the answer is yes (Sleep Med. 2007 Dec;9:15-21).
The Australian National Health and Medical Research Council supported the study. Dr. Bin reported having no financial conflicts.
AT SLEEP 2016
Key clinical point: Maternal sleep apnea is associated with increased rates of obstetric intervention and preterm birth.
Major finding: The rate of preterm birth at 36 weeks or earlier was 14.5% in the group with maternal sleep apnea, compared with 6.9% in controls.
Data source: A population-based cohort study of 636,227 women who gave birth in a New South Wales, Australia, hospital during 2002-2012.
Disclosures: The Australian National Health and Medical Research Council supported the study. Dr. Bin reported having no financial conflicts.
Below-knee angioplasty for limb salvage: Keep it simple
PARIS – Below-the-knee plain balloon angioplasty is an effective strategy for limb-salvage in patients with critical limb ischemia who otherwise face the prospect of a major amputation, Ana P. Mollon, MD, said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
Dr. Mollon of Posadas National Hospital in Buenos Aires, presented a retrospective series of 82 consecutive patients who underwent below-the-knee percutaneous angioplasty for critical limb ischemia with multivessel involvement. The amputation-free survival rate at a mean of 15.1 months of follow-up was 88%.
Sixty of the 82 patients had triple-artery involvement below the knee. The other 22 had two involved arteries. As is typical in patients with critical limb ischemia, comorbid conditions were common: Seventy-five patients had diabetes, 58 were hypertensive, and 48 were current smokers.
Of the 124 arteries treated by Dr. Mollon and coworkers, the posterior tibial artery was addressed in 41% of cases, the anterior tibial artery in 39%, and the peroneal artery in 18%. Two percent of patients received dilatation of plantar arch lesions.
Seventy percent of treated lesions were total occlusions, nearly half of which were more than 5 cm in length.
The treatment was plain balloon angioplasty in 78% of cases, drug-coated balloons in 12%, bare metal stenting in 7%, and drug-eluting stents in 3%. An antegrade approach was used in 95% of cases, and the remainder received a dual antegrade/retrograde approach.
Roughly 80% of patients were Rutherford category 5 or 6 before treatment. At 12 months post angioplasty, most patients were category 1 or 2, and about one-quarter were category 5 or 6.
Angioplasty was unsuccessful in restoring straight line flow in six patients.
All 10 patients who underwent a major amputation had triple-vessel involvement below the knee; in 9 of the 10, interventionalists were able to treat one of the three severely diseased arteries. Five of the 10 amputees had osteomyelitis.
Session chair Flavio Ribichini, MD, applauded Dr. Mollon and her Argentine colleagues for their predominant use of plain balloon angioplasty in this setting.
“I absolutely share your view on this. It’s the simplest and cheapest approach. The point is that you’re saving the foot now. It’s not that important what’s going to happen in 1 year. I don’t think it makes sense to use drug-coated balloons in this setting. It’s much more sensible to use a simple procedure and see how it goes,” said Dr. Ribichini, professor of cardiovascular medicine at the University of Verona (Italy).
Dr. Mollon said that several years ago her group briefly turned to the use of drug-coated balloons for below-the-knee limb salvage, but they soon switched back to plain balloon angioplasty because they didn’t see any advantage in patient outcomes with the more elaborate technology.
Discussant Benjamin Honton, MD, of the Pasteur Clinic in Toulouse, France, said, “We, too, have been disappointed with the drug-coated balloon, especially in the posterior tibial artery.”
Dr. Mollon reported having no financial conflicts.
PARIS – Below-the-knee plain balloon angioplasty is an effective strategy for limb-salvage in patients with critical limb ischemia who otherwise face the prospect of a major amputation, Ana P. Mollon, MD, said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
Dr. Mollon of Posadas National Hospital in Buenos Aires, presented a retrospective series of 82 consecutive patients who underwent below-the-knee percutaneous angioplasty for critical limb ischemia with multivessel involvement. The amputation-free survival rate at a mean of 15.1 months of follow-up was 88%.
Sixty of the 82 patients had triple-artery involvement below the knee. The other 22 had two involved arteries. As is typical in patients with critical limb ischemia, comorbid conditions were common: Seventy-five patients had diabetes, 58 were hypertensive, and 48 were current smokers.
Of the 124 arteries treated by Dr. Mollon and coworkers, the posterior tibial artery was addressed in 41% of cases, the anterior tibial artery in 39%, and the peroneal artery in 18%. Two percent of patients received dilatation of plantar arch lesions.
Seventy percent of treated lesions were total occlusions, nearly half of which were more than 5 cm in length.
The treatment was plain balloon angioplasty in 78% of cases, drug-coated balloons in 12%, bare metal stenting in 7%, and drug-eluting stents in 3%. An antegrade approach was used in 95% of cases, and the remainder received a dual antegrade/retrograde approach.
Roughly 80% of patients were Rutherford category 5 or 6 before treatment. At 12 months post angioplasty, most patients were category 1 or 2, and about one-quarter were category 5 or 6.
Angioplasty was unsuccessful in restoring straight line flow in six patients.
All 10 patients who underwent a major amputation had triple-vessel involvement below the knee; in 9 of the 10, interventionalists were able to treat one of the three severely diseased arteries. Five of the 10 amputees had osteomyelitis.
Session chair Flavio Ribichini, MD, applauded Dr. Mollon and her Argentine colleagues for their predominant use of plain balloon angioplasty in this setting.
“I absolutely share your view on this. It’s the simplest and cheapest approach. The point is that you’re saving the foot now. It’s not that important what’s going to happen in 1 year. I don’t think it makes sense to use drug-coated balloons in this setting. It’s much more sensible to use a simple procedure and see how it goes,” said Dr. Ribichini, professor of cardiovascular medicine at the University of Verona (Italy).
Dr. Mollon said that several years ago her group briefly turned to the use of drug-coated balloons for below-the-knee limb salvage, but they soon switched back to plain balloon angioplasty because they didn’t see any advantage in patient outcomes with the more elaborate technology.
Discussant Benjamin Honton, MD, of the Pasteur Clinic in Toulouse, France, said, “We, too, have been disappointed with the drug-coated balloon, especially in the posterior tibial artery.”
Dr. Mollon reported having no financial conflicts.
PARIS – Below-the-knee plain balloon angioplasty is an effective strategy for limb-salvage in patients with critical limb ischemia who otherwise face the prospect of a major amputation, Ana P. Mollon, MD, said at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
Dr. Mollon of Posadas National Hospital in Buenos Aires, presented a retrospective series of 82 consecutive patients who underwent below-the-knee percutaneous angioplasty for critical limb ischemia with multivessel involvement. The amputation-free survival rate at a mean of 15.1 months of follow-up was 88%.
Sixty of the 82 patients had triple-artery involvement below the knee. The other 22 had two involved arteries. As is typical in patients with critical limb ischemia, comorbid conditions were common: Seventy-five patients had diabetes, 58 were hypertensive, and 48 were current smokers.
Of the 124 arteries treated by Dr. Mollon and coworkers, the posterior tibial artery was addressed in 41% of cases, the anterior tibial artery in 39%, and the peroneal artery in 18%. Two percent of patients received dilatation of plantar arch lesions.
Seventy percent of treated lesions were total occlusions, nearly half of which were more than 5 cm in length.
The treatment was plain balloon angioplasty in 78% of cases, drug-coated balloons in 12%, bare metal stenting in 7%, and drug-eluting stents in 3%. An antegrade approach was used in 95% of cases, and the remainder received a dual antegrade/retrograde approach.
Roughly 80% of patients were Rutherford category 5 or 6 before treatment. At 12 months post angioplasty, most patients were category 1 or 2, and about one-quarter were category 5 or 6.
Angioplasty was unsuccessful in restoring straight line flow in six patients.
All 10 patients who underwent a major amputation had triple-vessel involvement below the knee; in 9 of the 10, interventionalists were able to treat one of the three severely diseased arteries. Five of the 10 amputees had osteomyelitis.
Session chair Flavio Ribichini, MD, applauded Dr. Mollon and her Argentine colleagues for their predominant use of plain balloon angioplasty in this setting.
“I absolutely share your view on this. It’s the simplest and cheapest approach. The point is that you’re saving the foot now. It’s not that important what’s going to happen in 1 year. I don’t think it makes sense to use drug-coated balloons in this setting. It’s much more sensible to use a simple procedure and see how it goes,” said Dr. Ribichini, professor of cardiovascular medicine at the University of Verona (Italy).
Dr. Mollon said that several years ago her group briefly turned to the use of drug-coated balloons for below-the-knee limb salvage, but they soon switched back to plain balloon angioplasty because they didn’t see any advantage in patient outcomes with the more elaborate technology.
Discussant Benjamin Honton, MD, of the Pasteur Clinic in Toulouse, France, said, “We, too, have been disappointed with the drug-coated balloon, especially in the posterior tibial artery.”
Dr. Mollon reported having no financial conflicts.
AT EUROPCR 2016
Key clinical point: Experts agree that plain balloon angioplasty is the way to go for limb salvage in patients with critical limb ischemia.
Major finding: The amputation-free survival rate in a consecutive series of patients who underwent below-the-knee angioplasty for critical limb ischemia was 88% at a mean of 15.1 months of follow-up.
Data source: A retrospective case series comprising 82 consecutive patients.
Disclosures: The presenter reported having no financial conflicts.
Permanent pacemaker in TAVR: Earlier implantation costs much less
PARIS – When a patient undergoing transcatheter aortic valve replacement needs a permanent pacemaker, the additional hospital costs are significantly less if the device is implanted within 24 hours post TAVR rather than later, Seth Clancy reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“Not only the need for permanent pacemaker implantation but also the timing of the procedure as well as the management and monitoring of conduction disturbances have important resource use implications for TAVR,” observed Mr. Clancy of Edwards Lifesciences of Irvine, Calif.
He presented an economic analysis of all 12,148 TAVR hospitalizations included in the Medicare database for 2014. A key finding: The mean cost of TAVR hospitalizations with no permanent pacemaker implantation was $63,136, while for the 12% of TAVRs that did include permanent pacemaker implantation, the mean cost shot up to $80,441, for a difference of $17,305.
The additional cost of putting in a permanent pacemaker included nearly $8,000 for supplies, more than $2,600 for additional time in the operating room and/or catheterization laboratory, and in excess of $2,100 worth of extra ICU or cardiac care unit time.
Patients who received a permanent pacemaker during their TAVR hospitalization spent an average of 2.3 days longer in the hospital than the mean 6.6 days for patients who didn’t get a permanent pacemaker.
Drilling down further into the data, Mr. Clancy found that 41% of permanent pacemakers implanted during hospitalization for TAVR went in within 24 hours of the TAVR procedure. In a multivariate regression analysis adjusted for differences in patient demographics, comorbid conditions, and complications, those patients generated an average of $9,843 more in hospital costs than patients who didn’t get a permanent pacemaker during their TAVR hospitalization. However, patients who received a permanent pacemaker more than 24 hours after TAVR cost an average of $17,681 more and had a 2.72-day longer stay than patients who didn’t get a permanent pacemaker.
The need for a permanent pacemaker is a common complication following TAVR. This has been a sticking point for many cardiothoracic surgeons, who note that rates of permanent pacemaker implantation following surgical aortic valve replacement are far lower. Still, rates in TAVR patients have come down over time with advances in valve technology. Currently, permanent pacemaker implantation rates in TAVR patients are 5%-25%, depending upon the valve system, according to Mr. Clancy.
Advances in device design and techniques aimed at reducing the permanent pacemaker implantation rate substantially below the 12% figure seen in 2014 have the potential to generate substantial cost savings, he observed.
Session chair Mohammad Abdelghani, MD, of the Academic Medical Center at Amsterdam questioned whether the study results are relevant to European practice because of the large differences in health care costs.
Discussant Sonia Petronio, MD, expressed a more fundamental reservation.
“This is a very important subject – and a very dangerous one,” said Dr. Petronio of the University of Pisa (Italy). “It’s easier and less costly for a hospital to encourage increasing early permanent pacemaker implantation because the patient can go home earlier.”
“We don’t want to put in a pacemaker earlier to save money,” agreed Dr. Abdelghani. “This is not a cost-effectiveness analysis, it’s purely a cost analysis. Cost-effectiveness would take into account the long-term clinical outcomes and welfare of the patients. We would like to see that from you next year.”
Mr. Clancy is an employee of Edwards Lifesciences, which funded the study.
PARIS – When a patient undergoing transcatheter aortic valve replacement needs a permanent pacemaker, the additional hospital costs are significantly less if the device is implanted within 24 hours post TAVR rather than later, Seth Clancy reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“Not only the need for permanent pacemaker implantation but also the timing of the procedure as well as the management and monitoring of conduction disturbances have important resource use implications for TAVR,” observed Mr. Clancy of Edwards Lifesciences of Irvine, Calif.
He presented an economic analysis of all 12,148 TAVR hospitalizations included in the Medicare database for 2014. A key finding: The mean cost of TAVR hospitalizations with no permanent pacemaker implantation was $63,136, while for the 12% of TAVRs that did include permanent pacemaker implantation, the mean cost shot up to $80,441, for a difference of $17,305.
The additional cost of putting in a permanent pacemaker included nearly $8,000 for supplies, more than $2,600 for additional time in the operating room and/or catheterization laboratory, and in excess of $2,100 worth of extra ICU or cardiac care unit time.
Patients who received a permanent pacemaker during their TAVR hospitalization spent an average of 2.3 days longer in the hospital than the mean 6.6 days for patients who didn’t get a permanent pacemaker.
Drilling down further into the data, Mr. Clancy found that 41% of permanent pacemakers implanted during hospitalization for TAVR went in within 24 hours of the TAVR procedure. In a multivariate regression analysis adjusted for differences in patient demographics, comorbid conditions, and complications, those patients generated an average of $9,843 more in hospital costs than patients who didn’t get a permanent pacemaker during their TAVR hospitalization. However, patients who received a permanent pacemaker more than 24 hours after TAVR cost an average of $17,681 more and had a 2.72-day longer stay than patients who didn’t get a permanent pacemaker.
The need for a permanent pacemaker is a common complication following TAVR. This has been a sticking point for many cardiothoracic surgeons, who note that rates of permanent pacemaker implantation following surgical aortic valve replacement are far lower. Still, rates in TAVR patients have come down over time with advances in valve technology. Currently, permanent pacemaker implantation rates in TAVR patients are 5%-25%, depending upon the valve system, according to Mr. Clancy.
Advances in device design and techniques aimed at reducing the permanent pacemaker implantation rate substantially below the 12% figure seen in 2014 have the potential to generate substantial cost savings, he observed.
Session chair Mohammad Abdelghani, MD, of the Academic Medical Center at Amsterdam questioned whether the study results are relevant to European practice because of the large differences in health care costs.
Discussant Sonia Petronio, MD, expressed a more fundamental reservation.
“This is a very important subject – and a very dangerous one,” said Dr. Petronio of the University of Pisa (Italy). “It’s easier and less costly for a hospital to encourage increasing early permanent pacemaker implantation because the patient can go home earlier.”
“We don’t want to put in a pacemaker earlier to save money,” agreed Dr. Abdelghani. “This is not a cost-effectiveness analysis, it’s purely a cost analysis. Cost-effectiveness would take into account the long-term clinical outcomes and welfare of the patients. We would like to see that from you next year.”
Mr. Clancy is an employee of Edwards Lifesciences, which funded the study.
PARIS – When a patient undergoing transcatheter aortic valve replacement needs a permanent pacemaker, the additional hospital costs are significantly less if the device is implanted within 24 hours post TAVR rather than later, Seth Clancy reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“Not only the need for permanent pacemaker implantation but also the timing of the procedure as well as the management and monitoring of conduction disturbances have important resource use implications for TAVR,” observed Mr. Clancy of Edwards Lifesciences of Irvine, Calif.
He presented an economic analysis of all 12,148 TAVR hospitalizations included in the Medicare database for 2014. A key finding: The mean cost of TAVR hospitalizations with no permanent pacemaker implantation was $63,136, while for the 12% of TAVRs that did include permanent pacemaker implantation, the mean cost shot up to $80,441, for a difference of $17,305.
The additional cost of putting in a permanent pacemaker included nearly $8,000 for supplies, more than $2,600 for additional time in the operating room and/or catheterization laboratory, and in excess of $2,100 worth of extra ICU or cardiac care unit time.
Patients who received a permanent pacemaker during their TAVR hospitalization spent an average of 2.3 days longer in the hospital than the mean 6.6 days for patients who didn’t get a permanent pacemaker.
Drilling down further into the data, Mr. Clancy found that 41% of permanent pacemakers implanted during hospitalization for TAVR went in within 24 hours of the TAVR procedure. In a multivariate regression analysis adjusted for differences in patient demographics, comorbid conditions, and complications, those patients generated an average of $9,843 more in hospital costs than patients who didn’t get a permanent pacemaker during their TAVR hospitalization. However, patients who received a permanent pacemaker more than 24 hours after TAVR cost an average of $17,681 more and had a 2.72-day longer stay than patients who didn’t get a permanent pacemaker.
The need for a permanent pacemaker is a common complication following TAVR. This has been a sticking point for many cardiothoracic surgeons, who note that rates of permanent pacemaker implantation following surgical aortic valve replacement are far lower. Still, rates in TAVR patients have come down over time with advances in valve technology. Currently, permanent pacemaker implantation rates in TAVR patients are 5%-25%, depending upon the valve system, according to Mr. Clancy.
Advances in device design and techniques aimed at reducing the permanent pacemaker implantation rate substantially below the 12% figure seen in 2014 have the potential to generate substantial cost savings, he observed.
Session chair Mohammad Abdelghani, MD, of the Academic Medical Center at Amsterdam questioned whether the study results are relevant to European practice because of the large differences in health care costs.
Discussant Sonia Petronio, MD, expressed a more fundamental reservation.
“This is a very important subject – and a very dangerous one,” said Dr. Petronio of the University of Pisa (Italy). “It’s easier and less costly for a hospital to encourage increasing early permanent pacemaker implantation because the patient can go home earlier.”
“We don’t want to put in a pacemaker earlier to save money,” agreed Dr. Abdelghani. “This is not a cost-effectiveness analysis, it’s purely a cost analysis. Cost-effectiveness would take into account the long-term clinical outcomes and welfare of the patients. We would like to see that from you next year.”
Mr. Clancy is an employee of Edwards Lifesciences, which funded the study.
AT EUROPCR 2016
Key clinical point: The incremental cost of permanent pacemaker implantation more than 24 hours after transcatheter aortic valve replacement is almost twice as great as if the pacemaker goes in within 24 hours.
Major finding: The mean cost of hospitalization for transcatheter aortic valve replacement without permanent pacemaker implication in Medicare patients in 2014 was $63,136, compared with $80,441 if they needed a pacemaker.
Data source: This was a retrospective study of the health care costs and lengths of stay for all 12,148 hospitalizations for transcatheter aortic valve replacement in the Medicare inpatient database for 2014.
Disclosures: The presenter is an employee of Edwards Lifesciences, which funded the study.
Minimalist TAVR without on-site surgery under study
PARIS – The buzzword in transcatheter aortic valve replacement today is “minimalist.” The search is on for ways in which to safely simplify the procedure to reduce the current unsustainably high cost and improve the patient experience.
Among the elements typically involved in minimalist TAVR are performance of the procedure in the cardiac catheterization laboratory via transfemoral access rather than in the costlier operating room, use of conscious sedation rather than general anesthesia, transthoracic echocardiographic guidance, no Swan-Ganz catheter, and no ICU stay for most patients. But these are tepid measures compared with what’s under study in Germany.
The German health care system is engaged in a study of what has to be the ultimate in minimalist TAVR: doing it in hospitals without on-site cardiac surgery. And the short-term results in more than 1,300 German patients treated in such a setting look every bit as good as in patients whose procedure took place in hospitals more conventionally equipped with both cardiology and cardiothoracic surgery departments, Holger Eggebrecht, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“Lack of a cardiac surgery department on site should not be regarded as a contraindication for TAVR,” concluded Dr. Eggebrecht of Agaplesion Bethanien Hospital in Frankfurt, Germany.
Of course, it is deemed an absolute contraindication to TAVR both in the current European Society of Cardiology and U.S. guidelines. But that position was developed in an earlier era when procedural safety had not yet been established. It was based on the expert consensus opinion of physicians drawn mostly from large tertiary centers with both cardiology and cardiac surgery on site. And this absolute contraindication was never supported by any data, he said.
In the United States, arguably the most litigious nation in the world, it’s virtually unthinkable – at least for now – to perform TAVR without a cardiothoracic surgeon on site in the event bailout emergency cardiac surgery should become necessary. But Germany, which boasts universal health care coverage at an affordable cost, operates differently. Indeed, in Dr. Eggebrecht’s study of all 17,919 transfemoral TAVR procedures performed in Germany during 2013 and 2014, fully 22 of the 77 hospitals where the procedures took place had no on-site cardiac surgery department.
He presented a comparison of in-hospital outcomes in the 1,332 German patients (7.4%) whose TAVR took place in hospitals without a cardiac surgery department and the 16,587 patients treated in hospitals with both cardiology and cardiac surgery departments. The data came from the prospective German Quality Assurance Registry on Aortic Valve Replacement, which records in extensive detail all TAVR and surgical replacements in the country. Participation in the registry is mandatory.
The main study finding: Even though patients at no–cardiac surgery hospitals were older, had more comorbid conditions, and were at higher predicted perioperative risk of mortality, the rates of intraprocedural complications, in-hospital strokes, and mortality were similar in the two groups.
Moreover, when Dr. Eggebrecht and coinvestigators performed a case-control substudy involving 555 patient pairs extensively matched on more than a dozen variables, including a requirement for identical scores on validated risk prediction tools for estimating in-hospital mortality, the results were the same as in the full study population.
The key to the high-quality TAVR outcomes documented at hospitals without a cardiac surgery department, Dr. Eggebrecht emphasized, is that in Germany TAVR can be performed only at hospitals where a contractually obligated heart team has signed off on the procedure. At hospitals without a cardiac surgery department, this heart team is composed of in-house cardiologists and visiting cardiac surgeons from collaborating hospitals. In TAVRs at some of these hospitals, a collaborating cardiac surgeon is present for the procedure and brings along a heart-lung machine which is primed and ready to go, if needed, as was the case in just 0.4% of the 1,332 TAVRs. At others, the surgeon is off site.
“I would think our data show that close cooperation within the heart team is the key to successful outcomes, not having a cardiac surgeon on site,” he concluded.
Audience member Volkmar Falk, MD, strode briskly to a microphone and made no effort to hide his incredulity at this project.
“What is the real advantage in not having a surgeon present? I don’t get it,” declared Dr. Falk, professor and director of the department of cardiovascular and vascular surgery at Charité University Hospital in Berlin.
“For a surgeon, this is all quite difficult to understand,” he continued. “If a surgeon has to come to a TAVR rescue from 10 km away, I don’t know how well that works. And if surgeons have to travel with all their equipment in order to be on site, I think this is a logistical nightmare and shouldn’t be done at all.
“All of the studies, all the clinical trials we always discuss, have been done in the setting of hospitals where the procedure was done together with a surgeon on site. That’s why we have these excellent results,” Dr. Falk added.
Dr. Eggebrecht replied, “We’re having a scientific discussion here, and I think our data clearly show that you can construct a successful heart team even though you don’t have a cardiac surgery department on site.”
An Israeli cardiologist in the audience said a similar effort is underway in his country to open up TAVR to hospitals without an on-site cardiac surgeon. His objection is that, at least in Israel, a hospital with no on-site cardiac surgery is a marker for a TAVR center that is low volume, is late to embrace TAVR, and has cardiologists who are probably still early on the procedural learning curve.
Dr. Eggebrecht said that this is not the case in Germany, where some of the most experienced TAVR operators work at sites without a cardiac surgery department.
He reported that his study was partially funded by the German Cardiac Society, and he had no financial conflicts.
Simultaneously with his presentation, the study was published online (Eur Heart J. 2016 May 17. pii: ehw190).
PARIS – The buzzword in transcatheter aortic valve replacement today is “minimalist.” The search is on for ways in which to safely simplify the procedure to reduce the current unsustainably high cost and improve the patient experience.
Among the elements typically involved in minimalist TAVR are performance of the procedure in the cardiac catheterization laboratory via transfemoral access rather than in the costlier operating room, use of conscious sedation rather than general anesthesia, transthoracic echocardiographic guidance, no Swan-Ganz catheter, and no ICU stay for most patients. But these are tepid measures compared with what’s under study in Germany.
The German health care system is engaged in a study of what has to be the ultimate in minimalist TAVR: doing it in hospitals without on-site cardiac surgery. And the short-term results in more than 1,300 German patients treated in such a setting look every bit as good as in patients whose procedure took place in hospitals more conventionally equipped with both cardiology and cardiothoracic surgery departments, Holger Eggebrecht, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“Lack of a cardiac surgery department on site should not be regarded as a contraindication for TAVR,” concluded Dr. Eggebrecht of Agaplesion Bethanien Hospital in Frankfurt, Germany.
Of course, it is deemed an absolute contraindication to TAVR both in the current European Society of Cardiology and U.S. guidelines. But that position was developed in an earlier era when procedural safety had not yet been established. It was based on the expert consensus opinion of physicians drawn mostly from large tertiary centers with both cardiology and cardiac surgery on site. And this absolute contraindication was never supported by any data, he said.
In the United States, arguably the most litigious nation in the world, it’s virtually unthinkable – at least for now – to perform TAVR without a cardiothoracic surgeon on site in the event bailout emergency cardiac surgery should become necessary. But Germany, which boasts universal health care coverage at an affordable cost, operates differently. Indeed, in Dr. Eggebrecht’s study of all 17,919 transfemoral TAVR procedures performed in Germany during 2013 and 2014, fully 22 of the 77 hospitals where the procedures took place had no on-site cardiac surgery department.
He presented a comparison of in-hospital outcomes in the 1,332 German patients (7.4%) whose TAVR took place in hospitals without a cardiac surgery department and the 16,587 patients treated in hospitals with both cardiology and cardiac surgery departments. The data came from the prospective German Quality Assurance Registry on Aortic Valve Replacement, which records in extensive detail all TAVR and surgical replacements in the country. Participation in the registry is mandatory.
The main study finding: Even though patients at no–cardiac surgery hospitals were older, had more comorbid conditions, and were at higher predicted perioperative risk of mortality, the rates of intraprocedural complications, in-hospital strokes, and mortality were similar in the two groups.
Moreover, when Dr. Eggebrecht and coinvestigators performed a case-control substudy involving 555 patient pairs extensively matched on more than a dozen variables, including a requirement for identical scores on validated risk prediction tools for estimating in-hospital mortality, the results were the same as in the full study population.

The key to the high-quality TAVR outcomes documented at hospitals without a cardiac surgery department, Dr. Eggebrecht emphasized, is that in Germany TAVR can be performed only at hospitals where a contractually obligated heart team has signed off on the procedure. At hospitals without a cardiac surgery department, this heart team is composed of in-house cardiologists and visiting cardiac surgeons from collaborating hospitals. In TAVRs at some of these hospitals, a collaborating cardiac surgeon is present for the procedure and brings along a heart-lung machine which is primed and ready to go, if needed, as was the case in just 0.4% of the 1,332 TAVRs. At others, the surgeon is off site.
“I would think our data show that close cooperation within the heart team is the key to successful outcomes, not having a cardiac surgeon on site,” he concluded.
Audience member Volkmar Falk, MD, strode briskly to a microphone and made no effort to hide his incredulity at this project.
“What is the real advantage in not having a surgeon present? I don’t get it,” declared Dr. Falk, professor and director of the department of cardiovascular and vascular surgery at Charité University Hospital in Berlin.
“For a surgeon, this is all quite difficult to understand,” he continued. “If a surgeon has to come to a TAVR rescue from 10 km away, I don’t know how well that works. And if surgeons have to travel with all their equipment in order to be on site, I think this is a logistical nightmare and shouldn’t be done at all.
“All of the studies, all the clinical trials we always discuss, have been done in the setting of hospitals where the procedure was done together with a surgeon on site. That’s why we have these excellent results,” Dr. Falk added.
Dr. Eggebrecht replied, “We’re having a scientific discussion here, and I think our data clearly show that you can construct a successful heart team even though you don’t have a cardiac surgery department on site.”
An Israeli cardiologist in the audience said a similar effort is underway in his country to open up TAVR to hospitals without an on-site cardiac surgeon. His objection is that, at least in Israel, a hospital with no on-site cardiac surgery is a marker for a TAVR center that is low volume, is late to embrace TAVR, and has cardiologists who are probably still early on the procedural learning curve.
Dr. Eggebrecht said that this is not the case in Germany, where some of the most experienced TAVR operators work at sites without a cardiac surgery department.
He reported that his study was partially funded by the German Cardiac Society, and he had no financial conflicts.
Simultaneously with his presentation, the study was published online (Eur Heart J. 2016 May 17. pii: ehw190).
PARIS – The buzzword in transcatheter aortic valve replacement today is “minimalist.” The search is on for ways in which to safely simplify the procedure to reduce the current unsustainably high cost and improve the patient experience.
Among the elements typically involved in minimalist TAVR are performance of the procedure in the cardiac catheterization laboratory via transfemoral access rather than in the costlier operating room, use of conscious sedation rather than general anesthesia, transthoracic echocardiographic guidance, no Swan-Ganz catheter, and no ICU stay for most patients. But these are tepid measures compared with what’s under study in Germany.
The German health care system is engaged in a study of what has to be the ultimate in minimalist TAVR: doing it in hospitals without on-site cardiac surgery. And the short-term results in more than 1,300 German patients treated in such a setting look every bit as good as in patients whose procedure took place in hospitals more conventionally equipped with both cardiology and cardiothoracic surgery departments, Holger Eggebrecht, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“Lack of a cardiac surgery department on site should not be regarded as a contraindication for TAVR,” concluded Dr. Eggebrecht of Agaplesion Bethanien Hospital in Frankfurt, Germany.
Of course, it is deemed an absolute contraindication to TAVR both in the current European Society of Cardiology and U.S. guidelines. But that position was developed in an earlier era when procedural safety had not yet been established. It was based on the expert consensus opinion of physicians drawn mostly from large tertiary centers with both cardiology and cardiac surgery on site. And this absolute contraindication was never supported by any data, he said.
In the United States, arguably the most litigious nation in the world, it’s virtually unthinkable – at least for now – to perform TAVR without a cardiothoracic surgeon on site in the event bailout emergency cardiac surgery should become necessary. But Germany, which boasts universal health care coverage at an affordable cost, operates differently. Indeed, in Dr. Eggebrecht’s study of all 17,919 transfemoral TAVR procedures performed in Germany during 2013 and 2014, fully 22 of the 77 hospitals where the procedures took place had no on-site cardiac surgery department.
He presented a comparison of in-hospital outcomes in the 1,332 German patients (7.4%) whose TAVR took place in hospitals without a cardiac surgery department and the 16,587 patients treated in hospitals with both cardiology and cardiac surgery departments. The data came from the prospective German Quality Assurance Registry on Aortic Valve Replacement, which records in extensive detail all TAVR and surgical replacements in the country. Participation in the registry is mandatory.
The main study finding: Even though patients at no–cardiac surgery hospitals were older, had more comorbid conditions, and were at higher predicted perioperative risk of mortality, the rates of intraprocedural complications, in-hospital strokes, and mortality were similar in the two groups.
Moreover, when Dr. Eggebrecht and coinvestigators performed a case-control substudy involving 555 patient pairs extensively matched on more than a dozen variables, including a requirement for identical scores on validated risk prediction tools for estimating in-hospital mortality, the results were the same as in the full study population.

The key to the high-quality TAVR outcomes documented at hospitals without a cardiac surgery department, Dr. Eggebrecht emphasized, is that in Germany TAVR can be performed only at hospitals where a contractually obligated heart team has signed off on the procedure. At hospitals without a cardiac surgery department, this heart team is composed of in-house cardiologists and visiting cardiac surgeons from collaborating hospitals. In TAVRs at some of these hospitals, a collaborating cardiac surgeon is present for the procedure and brings along a heart-lung machine which is primed and ready to go, if needed, as was the case in just 0.4% of the 1,332 TAVRs. At others, the surgeon is off site.
“I would think our data show that close cooperation within the heart team is the key to successful outcomes, not having a cardiac surgeon on site,” he concluded.
Audience member Volkmar Falk, MD, strode briskly to a microphone and made no effort to hide his incredulity at this project.
“What is the real advantage in not having a surgeon present? I don’t get it,” declared Dr. Falk, professor and director of the department of cardiovascular and vascular surgery at Charité University Hospital in Berlin.
“For a surgeon, this is all quite difficult to understand,” he continued. “If a surgeon has to come to a TAVR rescue from 10 km away, I don’t know how well that works. And if surgeons have to travel with all their equipment in order to be on site, I think this is a logistical nightmare and shouldn’t be done at all.
“All of the studies, all the clinical trials we always discuss, have been done in the setting of hospitals where the procedure was done together with a surgeon on site. That’s why we have these excellent results,” Dr. Falk added.
Dr. Eggebrecht replied, “We’re having a scientific discussion here, and I think our data clearly show that you can construct a successful heart team even though you don’t have a cardiac surgery department on site.”
An Israeli cardiologist in the audience said a similar effort is underway in his country to open up TAVR to hospitals without an on-site cardiac surgeon. His objection is that, at least in Israel, a hospital with no on-site cardiac surgery is a marker for a TAVR center that is low volume, is late to embrace TAVR, and has cardiologists who are probably still early on the procedural learning curve.
Dr. Eggebrecht said that this is not the case in Germany, where some of the most experienced TAVR operators work at sites without a cardiac surgery department.
He reported that his study was partially funded by the German Cardiac Society, and he had no financial conflicts.
Simultaneously with his presentation, the study was published online (Eur Heart J. 2016 May 17. pii: ehw190).
AT EUROPCR 2016
Key clinical point: TAVR can be performed in hospitals safely without a cardiac surgery department.
Major finding: In-hospital mortality occurred in 3.8% of patients who underwent TAVR at hospitals without on-site cardiac surgery and in 4.2% of patients whose procedure was done at hospitals with a cardiac surgery department.
Data source: An analysis of in-hospital outcomes of all 17,919 patients who underwent TAVR in Germany during 2013 and 2014, including 1,332 whose procedures took place at one of the 22 hospitals with no on-site cardiac surgery department.
Disclosures: The study was partially funded by the German Cardiac Society. The presenter reported having no financial conflicts.
Insomnia in Young Men Boosts Cardiovascular and Cerebrovascular Risk
DENVER – Young to early middle-aged men with insomnia symptoms are at increased risk for cardiovascular and cerebrovascular events, according to an analysis from the landmark CARDIA study.
“We found that younger to mid-life men with difficulty initiating sleep or with more than one insomnia symptom were at greater risk for incident cardiovascular disease events. And despite the fact that women in general seemed to be more prone to report those sleep difficulties, those that did were not at increased risk,” Megan E. Petrov, PhD, reported at the annual meeting of the Associated Professional Sleep Societies.
It is well established that insomnia in older adults is associated with increased cardiovascular risk. For example, a meta-analysis of 13 prospective studies with more than 123,000 subjects concluded that insomnia was associated with a 45% increased risk of fatal and nonfatal cardiovascular events (Eur J Prev Cardiol. 2014 Jan;21[1]:57-64). But those studies typically involved older individuals, which makes cause and effect more difficult to determine because so many chronic conditions become more prevalent with advancing age, noted Dr. Petrov of Arizona State University in Phoenix.
“We wanted to see if insomnia is truly an early risk factor in the pathogenesis of cardiovascular disease and stroke,” she said.
To do so, she and her coinvestigators turned to the CARDIA database. CARDIA (the Coronary Artery Risk Development in Young Adults study), is a National Heart, Lung, and Blood Institute–sponsored prospective, epidemiologic study.
She reported on 2,950 non-Hispanic black or white participants aged 33-45 and free of any history of cardiovascular disease in 2000-2001, when they answered questions about insomnia symptoms. Difficulty in initiating sleep was reported by 16.3% of men and 20.7% of women. Difficulty maintaining sleep was a problem for 9.3% of men and 14.5% of women. And 20.6% of men and 20.1% of women reported frequent early morning awakening.
During a mean 11.5 years of prospective follow-up, 4.1% of men and 2.3% of women had a fatal or nonfatal MI, stroke, transient ischemic attack, heart failure, peripheral vascular disease, or hospitalization for an acute coronary syndrome.
In a multivariate logistic regression analysis fully adjusted for demographics, socioeconomic status, body mass index, blood pressure, diabetes, depression, health behaviors, medications, thyroid disease, and kidney problems, men who reported difficulty in getting to sleep had a 2.64-fold increased risk of one of these adverse outcomes. That was the only insomnia symptom associated with significantly increased risk. However, men but not women who reported having more than one insomnia symptom had a 39% increased risk of a cardiovascular event for each additional symptom.
Prior studies have shown that insomnia is associated with cardiac sympathetic hyperactivation. That observation suggests a plausible mechanism for increased cardiovascular and cerebrovascular risk, but it doesn’t explain why that risk was confined to young men in CARDIA. One possibility is that because of gender-related differences in perception, men tend to report having insomnia symptoms only when the insomnia is more severe, Dr. Petrov suggested.
The strengths of the CARDIA study are that all cardiovascular endpoints had to be physician certified, and the study includes a large black population. A study limitation is the relatively small number of cardiovascular events, as to be expected in a younger population. Thus, confirmation of the new findings in another large data set will be important, she noted.
As a next step in her research, Dr. Petrov said she plans to drill down in the CARDIA data to see if race modified the impact of insomnia symptoms on cardiovascular outcomes. Black participants reported all insomnia symptoms more frequently than did whites. For example, difficulty initiating sleep was reported by 25.7% of blacks, compared with just 13.5% of whites, and early morning awakening was twice as prevalent among the black participants.
Also, CARDIA participants provided self-reported sleep duration data. It will be illuminating to see if sleep duration had a modifying effect upon the insomnia/cardiovascular risk association observed in men, she said.
Dr. Petrov reported having no relevant financial conflicts.
DENVER – Young to early middle-aged men with insomnia symptoms are at increased risk for cardiovascular and cerebrovascular events, according to an analysis from the landmark CARDIA study.
“We found that younger to mid-life men with difficulty initiating sleep or with more than one insomnia symptom were at greater risk for incident cardiovascular disease events. And despite the fact that women in general seemed to be more prone to report those sleep difficulties, those that did were not at increased risk,” Megan E. Petrov, PhD, reported at the annual meeting of the Associated Professional Sleep Societies.
It is well established that insomnia in older adults is associated with increased cardiovascular risk. For example, a meta-analysis of 13 prospective studies with more than 123,000 subjects concluded that insomnia was associated with a 45% increased risk of fatal and nonfatal cardiovascular events (Eur J Prev Cardiol. 2014 Jan;21[1]:57-64). But those studies typically involved older individuals, which makes cause and effect more difficult to determine because so many chronic conditions become more prevalent with advancing age, noted Dr. Petrov of Arizona State University in Phoenix.
“We wanted to see if insomnia is truly an early risk factor in the pathogenesis of cardiovascular disease and stroke,” she said.
To do so, she and her coinvestigators turned to the CARDIA database. CARDIA (the Coronary Artery Risk Development in Young Adults study), is a National Heart, Lung, and Blood Institute–sponsored prospective, epidemiologic study.
She reported on 2,950 non-Hispanic black or white participants aged 33-45 and free of any history of cardiovascular disease in 2000-2001, when they answered questions about insomnia symptoms. Difficulty in initiating sleep was reported by 16.3% of men and 20.7% of women. Difficulty maintaining sleep was a problem for 9.3% of men and 14.5% of women. And 20.6% of men and 20.1% of women reported frequent early morning awakening.
During a mean 11.5 years of prospective follow-up, 4.1% of men and 2.3% of women had a fatal or nonfatal MI, stroke, transient ischemic attack, heart failure, peripheral vascular disease, or hospitalization for an acute coronary syndrome.
In a multivariate logistic regression analysis fully adjusted for demographics, socioeconomic status, body mass index, blood pressure, diabetes, depression, health behaviors, medications, thyroid disease, and kidney problems, men who reported difficulty in getting to sleep had a 2.64-fold increased risk of one of these adverse outcomes. That was the only insomnia symptom associated with significantly increased risk. However, men but not women who reported having more than one insomnia symptom had a 39% increased risk of a cardiovascular event for each additional symptom.
Prior studies have shown that insomnia is associated with cardiac sympathetic hyperactivation. That observation suggests a plausible mechanism for increased cardiovascular and cerebrovascular risk, but it doesn’t explain why that risk was confined to young men in CARDIA. One possibility is that because of gender-related differences in perception, men tend to report having insomnia symptoms only when the insomnia is more severe, Dr. Petrov suggested.
The strengths of the CARDIA study are that all cardiovascular endpoints had to be physician certified, and the study includes a large black population. A study limitation is the relatively small number of cardiovascular events, as to be expected in a younger population. Thus, confirmation of the new findings in another large data set will be important, she noted.
As a next step in her research, Dr. Petrov said she plans to drill down in the CARDIA data to see if race modified the impact of insomnia symptoms on cardiovascular outcomes. Black participants reported all insomnia symptoms more frequently than did whites. For example, difficulty initiating sleep was reported by 25.7% of blacks, compared with just 13.5% of whites, and early morning awakening was twice as prevalent among the black participants.
Also, CARDIA participants provided self-reported sleep duration data. It will be illuminating to see if sleep duration had a modifying effect upon the insomnia/cardiovascular risk association observed in men, she said.
Dr. Petrov reported having no relevant financial conflicts.
DENVER – Young to early middle-aged men with insomnia symptoms are at increased risk for cardiovascular and cerebrovascular events, according to an analysis from the landmark CARDIA study.
“We found that younger to mid-life men with difficulty initiating sleep or with more than one insomnia symptom were at greater risk for incident cardiovascular disease events. And despite the fact that women in general seemed to be more prone to report those sleep difficulties, those that did were not at increased risk,” Megan E. Petrov, PhD, reported at the annual meeting of the Associated Professional Sleep Societies.
It is well established that insomnia in older adults is associated with increased cardiovascular risk. For example, a meta-analysis of 13 prospective studies with more than 123,000 subjects concluded that insomnia was associated with a 45% increased risk of fatal and nonfatal cardiovascular events (Eur J Prev Cardiol. 2014 Jan;21[1]:57-64). But those studies typically involved older individuals, which makes cause and effect more difficult to determine because so many chronic conditions become more prevalent with advancing age, noted Dr. Petrov of Arizona State University in Phoenix.
“We wanted to see if insomnia is truly an early risk factor in the pathogenesis of cardiovascular disease and stroke,” she said.
To do so, she and her coinvestigators turned to the CARDIA database. CARDIA (the Coronary Artery Risk Development in Young Adults study), is a National Heart, Lung, and Blood Institute–sponsored prospective, epidemiologic study.
She reported on 2,950 non-Hispanic black or white participants aged 33-45 and free of any history of cardiovascular disease in 2000-2001, when they answered questions about insomnia symptoms. Difficulty in initiating sleep was reported by 16.3% of men and 20.7% of women. Difficulty maintaining sleep was a problem for 9.3% of men and 14.5% of women. And 20.6% of men and 20.1% of women reported frequent early morning awakening.
During a mean 11.5 years of prospective follow-up, 4.1% of men and 2.3% of women had a fatal or nonfatal MI, stroke, transient ischemic attack, heart failure, peripheral vascular disease, or hospitalization for an acute coronary syndrome.
In a multivariate logistic regression analysis fully adjusted for demographics, socioeconomic status, body mass index, blood pressure, diabetes, depression, health behaviors, medications, thyroid disease, and kidney problems, men who reported difficulty in getting to sleep had a 2.64-fold increased risk of one of these adverse outcomes. That was the only insomnia symptom associated with significantly increased risk. However, men but not women who reported having more than one insomnia symptom had a 39% increased risk of a cardiovascular event for each additional symptom.
Prior studies have shown that insomnia is associated with cardiac sympathetic hyperactivation. That observation suggests a plausible mechanism for increased cardiovascular and cerebrovascular risk, but it doesn’t explain why that risk was confined to young men in CARDIA. One possibility is that because of gender-related differences in perception, men tend to report having insomnia symptoms only when the insomnia is more severe, Dr. Petrov suggested.
The strengths of the CARDIA study are that all cardiovascular endpoints had to be physician certified, and the study includes a large black population. A study limitation is the relatively small number of cardiovascular events, as to be expected in a younger population. Thus, confirmation of the new findings in another large data set will be important, she noted.
As a next step in her research, Dr. Petrov said she plans to drill down in the CARDIA data to see if race modified the impact of insomnia symptoms on cardiovascular outcomes. Black participants reported all insomnia symptoms more frequently than did whites. For example, difficulty initiating sleep was reported by 25.7% of blacks, compared with just 13.5% of whites, and early morning awakening was twice as prevalent among the black participants.
Also, CARDIA participants provided self-reported sleep duration data. It will be illuminating to see if sleep duration had a modifying effect upon the insomnia/cardiovascular risk association observed in men, she said.
Dr. Petrov reported having no relevant financial conflicts.
AT SLEEP 2016





















