User login
Baltimore’s pilot project in outpatient civil commitment
Novel program has gotten off to a rough start
No topic is quite so controversial in psychiatry as outpatient civil commitment.
We all value the right to make our own medical decisions, perhaps even more so when it comes to issues of mental health. The concern, however, is that some patients lack the ability to see that they are ill, and their refusal to participate in treatment leads them to repeated involuntary hospitalizations or to behaviors that result in their incarceration. Mandated care for forensic patients – those who have ended up in the criminal justice system – is not as controversial, but for those who have committed no crime, the legislative struggles can get very heated. Add to that the cost of implementing outpatient civil commitment (OCC), the difficulties in enforcing the orders, the call to include an array of services for participants, the difficulties in ascertaining if these mandates are successful in helping patients live better lives, and the fact that treatment doesn’t always work, then what seems like a simple solution to the problem of noncompliance is actually quite complex.
Maryland is one of four states that does not have OCC. In 2017, it was announced that the city of Baltimore would begin an OCC pilot program. The program, administered by Behavioral Health System Baltimore (BHSB) in partnership with numerous other mental health organizations, has had several fits and starts. The program initially was funded by the Substance Abuse and Mental Health Services Administration (SAMHSA), and the grant required the program to serve a minimum of 75 patients. However, it was soon discovered that there were no written regulations, and that began a 1-year delay while regulations were written, including two periods for open public comment. Once set, BHSB needed to let local hospitals know the program existed and solicit referrals.
When it became clear that they would not be able to serve 75 individuals, BHSB elected to give its funding back to SAMHSA, and a second 3-month delay ensued until the state of Maryland picked up the cost. The Baltimore pilot project in OCC is different from other civil commitment programs. The criteria for inclusion are brief: The patient must be aged over 18 years, be a resident of Baltimore, and must have been retained by a judge at a hospital inpatient civil commitment hearing at least two times in the past year because of noncompliance with outpatient treatment. The referral needs to come from the inpatient unit before the patient is discharged, and entails a “settlement agreement” with a judge, typically the same administrative law judge that visits the hospital weekly for the inpatient commitment hearings. A connection is made with the patient prior to discharge from the inpatient facility.
The Baltimore project also differs in many ways from traditional outpatient commitment programs in other states. In this program, the patient is not mandated to treatment. Instead, the patient is required to meet with the program’s peer-recovery specialist. The recovery specialist provides support for the patient after discharge, facilitates engagement in both psychiatric and somatic medical care, and helps with transportation to appointments. Services for each participant continue for 6 months; the initial SAMHSA grant was for $2.8 million for 4 years.
Nicole McGill, the program’s monitor notes: “When people are repeatedly hospitalized, it signals to us that maybe the system hasn’t been doing what it needs to do. For those people, the OCC pilot aims to be the connection to existing services in the community.”
Of the 19 people who have been referred to the program since October 2017, 9 have been accepted. Six of those patients enrolled voluntarily; three were ordered into the program against their will. Of those three, one patient has refused to meet with the peer-recovery specialist. That patient, however, is enrolled with an Assertive Community Treatment and is not someone who is refusing psychiatric treatment. That patient was enlisted with the hope that more engagement might prevent future hospitalizations.
“We didn’t want to create a program that forced people into treatment,” said Adrienne Breidenstine, vice president for policy and communications for BHSB. “Those programs don’t work. If a program participant chooses not to engage in services, the OCC peers will step up their engagement. There are no punitive or legal consequences; they aren’t brought back to the hospital or taken to jail.”
So far, the program has been well received by its clients. Feedback is solicited through the Mental Health Association of Maryland. One client provided a testimonial saying: “I didn’t know there were so many resources out there. They took me to the food co-op and gave me free sheets. I got a fan. They didn’t waste time getting me that. It helped a lot with the heat. They get me to appointments. They helped so much.”
This fall, BHSB is expanding its services; as of Sept. 9, 2019, any Baltimore resident who is currently hospitalized and has been hospitalized at least one other time in the past 12 months will be eligible for the outpatient “commitment” program; there will no longer be a requirement that these inpatient stays involved retention at a civil commitment hearing or that the patient ever had an involuntary status. Patients who have not been committed to the hospital, however, will only be able to enter the program on a voluntary track.
This model of OCC is a bit of a misnomer: Most of the patients are in the program voluntarily, and they are not being required to go to treatment or take medications. It is a way to get a higher level of service to the sickest of patients who have needed repeated hospitalizations without the objections that get raised when patients are mandated to care. Given the distress that involuntary hospitalization causes, it may well be helpful to expand a voluntary offer of services to all patients who are retained at a hearing, even if only once; perhaps the added support could prevent future decompensations.
For now, the program has had a rough beginning, and the expense is hard to justify for so few patients. For advocates who support outpatient commitment, this program does not go far enough, as there is no requirement to take medications and no consequence for refusal to participate. As the criteria for inclusion are loosened to include those who are repeatedly hospitalized on a voluntary basis, it will be interesting to see if hospitalizations can be prevented and if the quality of life can be improved for these patients.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University, 2016). She has a private practice, and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
Novel program has gotten off to a rough start
Novel program has gotten off to a rough start
No topic is quite so controversial in psychiatry as outpatient civil commitment.
We all value the right to make our own medical decisions, perhaps even more so when it comes to issues of mental health. The concern, however, is that some patients lack the ability to see that they are ill, and their refusal to participate in treatment leads them to repeated involuntary hospitalizations or to behaviors that result in their incarceration. Mandated care for forensic patients – those who have ended up in the criminal justice system – is not as controversial, but for those who have committed no crime, the legislative struggles can get very heated. Add to that the cost of implementing outpatient civil commitment (OCC), the difficulties in enforcing the orders, the call to include an array of services for participants, the difficulties in ascertaining if these mandates are successful in helping patients live better lives, and the fact that treatment doesn’t always work, then what seems like a simple solution to the problem of noncompliance is actually quite complex.
Maryland is one of four states that does not have OCC. In 2017, it was announced that the city of Baltimore would begin an OCC pilot program. The program, administered by Behavioral Health System Baltimore (BHSB) in partnership with numerous other mental health organizations, has had several fits and starts. The program initially was funded by the Substance Abuse and Mental Health Services Administration (SAMHSA), and the grant required the program to serve a minimum of 75 patients. However, it was soon discovered that there were no written regulations, and that began a 1-year delay while regulations were written, including two periods for open public comment. Once set, BHSB needed to let local hospitals know the program existed and solicit referrals.
When it became clear that they would not be able to serve 75 individuals, BHSB elected to give its funding back to SAMHSA, and a second 3-month delay ensued until the state of Maryland picked up the cost. The Baltimore pilot project in OCC is different from other civil commitment programs. The criteria for inclusion are brief: The patient must be aged over 18 years, be a resident of Baltimore, and must have been retained by a judge at a hospital inpatient civil commitment hearing at least two times in the past year because of noncompliance with outpatient treatment. The referral needs to come from the inpatient unit before the patient is discharged, and entails a “settlement agreement” with a judge, typically the same administrative law judge that visits the hospital weekly for the inpatient commitment hearings. A connection is made with the patient prior to discharge from the inpatient facility.
The Baltimore project also differs in many ways from traditional outpatient commitment programs in other states. In this program, the patient is not mandated to treatment. Instead, the patient is required to meet with the program’s peer-recovery specialist. The recovery specialist provides support for the patient after discharge, facilitates engagement in both psychiatric and somatic medical care, and helps with transportation to appointments. Services for each participant continue for 6 months; the initial SAMHSA grant was for $2.8 million for 4 years.
Nicole McGill, the program’s monitor notes: “When people are repeatedly hospitalized, it signals to us that maybe the system hasn’t been doing what it needs to do. For those people, the OCC pilot aims to be the connection to existing services in the community.”
Of the 19 people who have been referred to the program since October 2017, 9 have been accepted. Six of those patients enrolled voluntarily; three were ordered into the program against their will. Of those three, one patient has refused to meet with the peer-recovery specialist. That patient, however, is enrolled with an Assertive Community Treatment and is not someone who is refusing psychiatric treatment. That patient was enlisted with the hope that more engagement might prevent future hospitalizations.
“We didn’t want to create a program that forced people into treatment,” said Adrienne Breidenstine, vice president for policy and communications for BHSB. “Those programs don’t work. If a program participant chooses not to engage in services, the OCC peers will step up their engagement. There are no punitive or legal consequences; they aren’t brought back to the hospital or taken to jail.”
So far, the program has been well received by its clients. Feedback is solicited through the Mental Health Association of Maryland. One client provided a testimonial saying: “I didn’t know there were so many resources out there. They took me to the food co-op and gave me free sheets. I got a fan. They didn’t waste time getting me that. It helped a lot with the heat. They get me to appointments. They helped so much.”
This fall, BHSB is expanding its services; as of Sept. 9, 2019, any Baltimore resident who is currently hospitalized and has been hospitalized at least one other time in the past 12 months will be eligible for the outpatient “commitment” program; there will no longer be a requirement that these inpatient stays involved retention at a civil commitment hearing or that the patient ever had an involuntary status. Patients who have not been committed to the hospital, however, will only be able to enter the program on a voluntary track.
This model of OCC is a bit of a misnomer: Most of the patients are in the program voluntarily, and they are not being required to go to treatment or take medications. It is a way to get a higher level of service to the sickest of patients who have needed repeated hospitalizations without the objections that get raised when patients are mandated to care. Given the distress that involuntary hospitalization causes, it may well be helpful to expand a voluntary offer of services to all patients who are retained at a hearing, even if only once; perhaps the added support could prevent future decompensations.
For now, the program has had a rough beginning, and the expense is hard to justify for so few patients. For advocates who support outpatient commitment, this program does not go far enough, as there is no requirement to take medications and no consequence for refusal to participate. As the criteria for inclusion are loosened to include those who are repeatedly hospitalized on a voluntary basis, it will be interesting to see if hospitalizations can be prevented and if the quality of life can be improved for these patients.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University, 2016). She has a private practice, and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
No topic is quite so controversial in psychiatry as outpatient civil commitment.
We all value the right to make our own medical decisions, perhaps even more so when it comes to issues of mental health. The concern, however, is that some patients lack the ability to see that they are ill, and their refusal to participate in treatment leads them to repeated involuntary hospitalizations or to behaviors that result in their incarceration. Mandated care for forensic patients – those who have ended up in the criminal justice system – is not as controversial, but for those who have committed no crime, the legislative struggles can get very heated. Add to that the cost of implementing outpatient civil commitment (OCC), the difficulties in enforcing the orders, the call to include an array of services for participants, the difficulties in ascertaining if these mandates are successful in helping patients live better lives, and the fact that treatment doesn’t always work, then what seems like a simple solution to the problem of noncompliance is actually quite complex.
Maryland is one of four states that does not have OCC. In 2017, it was announced that the city of Baltimore would begin an OCC pilot program. The program, administered by Behavioral Health System Baltimore (BHSB) in partnership with numerous other mental health organizations, has had several fits and starts. The program initially was funded by the Substance Abuse and Mental Health Services Administration (SAMHSA), and the grant required the program to serve a minimum of 75 patients. However, it was soon discovered that there were no written regulations, and that began a 1-year delay while regulations were written, including two periods for open public comment. Once set, BHSB needed to let local hospitals know the program existed and solicit referrals.
When it became clear that they would not be able to serve 75 individuals, BHSB elected to give its funding back to SAMHSA, and a second 3-month delay ensued until the state of Maryland picked up the cost. The Baltimore pilot project in OCC is different from other civil commitment programs. The criteria for inclusion are brief: The patient must be aged over 18 years, be a resident of Baltimore, and must have been retained by a judge at a hospital inpatient civil commitment hearing at least two times in the past year because of noncompliance with outpatient treatment. The referral needs to come from the inpatient unit before the patient is discharged, and entails a “settlement agreement” with a judge, typically the same administrative law judge that visits the hospital weekly for the inpatient commitment hearings. A connection is made with the patient prior to discharge from the inpatient facility.
The Baltimore project also differs in many ways from traditional outpatient commitment programs in other states. In this program, the patient is not mandated to treatment. Instead, the patient is required to meet with the program’s peer-recovery specialist. The recovery specialist provides support for the patient after discharge, facilitates engagement in both psychiatric and somatic medical care, and helps with transportation to appointments. Services for each participant continue for 6 months; the initial SAMHSA grant was for $2.8 million for 4 years.
Nicole McGill, the program’s monitor notes: “When people are repeatedly hospitalized, it signals to us that maybe the system hasn’t been doing what it needs to do. For those people, the OCC pilot aims to be the connection to existing services in the community.”
Of the 19 people who have been referred to the program since October 2017, 9 have been accepted. Six of those patients enrolled voluntarily; three were ordered into the program against their will. Of those three, one patient has refused to meet with the peer-recovery specialist. That patient, however, is enrolled with an Assertive Community Treatment and is not someone who is refusing psychiatric treatment. That patient was enlisted with the hope that more engagement might prevent future hospitalizations.
“We didn’t want to create a program that forced people into treatment,” said Adrienne Breidenstine, vice president for policy and communications for BHSB. “Those programs don’t work. If a program participant chooses not to engage in services, the OCC peers will step up their engagement. There are no punitive or legal consequences; they aren’t brought back to the hospital or taken to jail.”
So far, the program has been well received by its clients. Feedback is solicited through the Mental Health Association of Maryland. One client provided a testimonial saying: “I didn’t know there were so many resources out there. They took me to the food co-op and gave me free sheets. I got a fan. They didn’t waste time getting me that. It helped a lot with the heat. They get me to appointments. They helped so much.”
This fall, BHSB is expanding its services; as of Sept. 9, 2019, any Baltimore resident who is currently hospitalized and has been hospitalized at least one other time in the past 12 months will be eligible for the outpatient “commitment” program; there will no longer be a requirement that these inpatient stays involved retention at a civil commitment hearing or that the patient ever had an involuntary status. Patients who have not been committed to the hospital, however, will only be able to enter the program on a voluntary track.
This model of OCC is a bit of a misnomer: Most of the patients are in the program voluntarily, and they are not being required to go to treatment or take medications. It is a way to get a higher level of service to the sickest of patients who have needed repeated hospitalizations without the objections that get raised when patients are mandated to care. Given the distress that involuntary hospitalization causes, it may well be helpful to expand a voluntary offer of services to all patients who are retained at a hearing, even if only once; perhaps the added support could prevent future decompensations.
For now, the program has had a rough beginning, and the expense is hard to justify for so few patients. For advocates who support outpatient commitment, this program does not go far enough, as there is no requirement to take medications and no consequence for refusal to participate. As the criteria for inclusion are loosened to include those who are repeatedly hospitalized on a voluntary basis, it will be interesting to see if hospitalizations can be prevented and if the quality of life can be improved for these patients.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University, 2016). She has a private practice, and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
The Naming Project: Renaming Schizophrenia
One in 100 people worldwide experience a condition called schizophrenia. There is widespread disagreement regarding what should be the appropriate name for this condition. Raquelle Mesholam-Gately, PhD, Matcheri Keshavan, MD, the Consumer Advisory Board, and associated members of the research team at Beth Israel Deaconess Medical Center are conducting a study about renaming schizophrenia.
They are requesting your opinion on this matter. If you choose to be in the study, you will complete this survey, which will take 5 to 10 minutes. The survey is anonymous, and no one will be able to link your answers back to you. Please do not include your name or other information that could be used to identify you in the survey responses. Participation in this study is voluntary.
Take the survey: https://forms.gle/fBVvrkGbVSU3H7Cz8
One in 100 people worldwide experience a condition called schizophrenia. There is widespread disagreement regarding what should be the appropriate name for this condition. Raquelle Mesholam-Gately, PhD, Matcheri Keshavan, MD, the Consumer Advisory Board, and associated members of the research team at Beth Israel Deaconess Medical Center are conducting a study about renaming schizophrenia.
They are requesting your opinion on this matter. If you choose to be in the study, you will complete this survey, which will take 5 to 10 minutes. The survey is anonymous, and no one will be able to link your answers back to you. Please do not include your name or other information that could be used to identify you in the survey responses. Participation in this study is voluntary.
Take the survey: https://forms.gle/fBVvrkGbVSU3H7Cz8
One in 100 people worldwide experience a condition called schizophrenia. There is widespread disagreement regarding what should be the appropriate name for this condition. Raquelle Mesholam-Gately, PhD, Matcheri Keshavan, MD, the Consumer Advisory Board, and associated members of the research team at Beth Israel Deaconess Medical Center are conducting a study about renaming schizophrenia.
They are requesting your opinion on this matter. If you choose to be in the study, you will complete this survey, which will take 5 to 10 minutes. The survey is anonymous, and no one will be able to link your answers back to you. Please do not include your name or other information that could be used to identify you in the survey responses. Participation in this study is voluntary.
Take the survey: https://forms.gle/fBVvrkGbVSU3H7Cz8
Artificial intelligence in psychiatry
For many people, artificial intelligence (AI) brings to mind some form of humanoid robot that speaks and acts like a human. However, AI is much more than merely robotics and machines. Professor John McCarthy of Stanford University, who first coined the term “artificial intelligence” in the early 1950s, defined it as “the science and engineering of making intelligent machines, especially intelligent computer programs”; he defined intelligence as “the computational part of the ability to achieve goals.”1 Artificial intelligence also is commonly defined as the development of computer systems able to perform tasks that normally require human intelligence.2 English Mathematician Alan Turing is considered one of the forefathers of AI research, and devised the first test to determine if a computer program was intelligent (Box 13). Today, AI has established itself as an integral part of medicine and psychiatry.
Box 1
During World War II, the English Mathematician Alan Turing helped the British government crack the Enigma machine, a coding device used by the Nazi army. He went on to pioneer many research projects in the field of artificial intelligence, including developing the Turing Test, which can determine if a computer program is intelligent.3 In this test, a human questioner uses a computer interface to pose questions to 2 respondents in different rooms; one of the respondents is a human and the other a computer program. If the questioner cannot tell the difference between the 2 respondents’ answers, then the computer program is deemed to be “artificially intelligent” because it can pass
The semantics of AI
Two subsets of AI are machine learning and deep learning.4,5 Machine learning is defined as a set of methods that can automatically detect patterns in data and then use the uncovered pattern to predict future data.4 Deep learning is a form of machine learning that enables computers to learn from experience and understand the world in terms of a hierarchy of concepts.5
Machine learning can be supervised, semi-supervised, or unsupervised. The majority of practical machine learning uses supervised learning, where all data are labeled and an algorithm is used to learn the mapping function from the input to the output. In unsupervised learning, all data are unlabeled and the algorithm models the underlying structure of the data by itself. Semi-supervised learning is a mixture of both.6
Many researchers also categorize AI into 2 types: general or “strong” AI, and narrow or “weak” AI. Strong AI is defined as computers that can think on a level at least equal to humans and are able to experience emotions and even consciousness.7 Weak AI includes adding “thinking-like” features to computers to make them more useful tools. Almost all AI technologies available today are considered to be weak AI.
AI in medicine
AI is being developed for a broad range of applications in medicine. This includes informatics approaches, including learning in health management systems such as electronic health records, and actively guiding physicians in their treatment decisions.8
AI has been applied to assist administrative workflows that reach beyond automated non-patient care activities such as chart documentation and placing orders. One example is the Judy Reitz Capacity Command Center, which was designed and built with GE Healthcare Partners.9 It combines AI technology in the form of systems engineering and predictive analytics to better manage multiple workflows in different administrative settings, including patient safety, volume, flow, and access to care.9
In April 2018, Intel Corporation surveyed 200 health-care decision makers in the United States regarding their use of AI in practice and their attitudes toward it.10 Overall, 37% of respondents reported using AI and 54% expected to increase their use of AI in the next 5 years. Clinical use of AI (77%) was more common than administrative use (41%) or financial use (26 %).10
Continue to: Box 2
Box 211-19 describes studies that evaluated the clinical use of AI in specialties other than psychiatry.
Box 2
Ophthalmology. Multiple studies have evaluated using artificial intelligence (AI) to screen for diabetic retinopathy, which is one of the fastest growing causes of blindness worldwide.11 In a recent study, researchers used a deep learning algorithm to automatically detect diabetic retinopathy and diabetic macular edema by analyzing retinal images. It was trained over a dataset of 128,000 images that were evaluated by 3 to 7 ophthalmologists. The algorithm showed high sensitivity and specificity for detecting referable diabetic retinopathy.11
Cardiology. One study looked at training a deep learning algorithm to predict cardiovascular risk based on analysis of retinal fundus images from 284,335 patients. In this study, the algorithm was able to predict a cardiovascular event in the next 5 years with 70% accuracy.12 The results were based on risk factors not previously thought to be quantifiable in retinal images, such as age, gender, smoking status, systolic blood pressure, and major adverse cardiac events.12 Similarly, researchers in the United Kingdom wanted to assess AI’s ability to predict a first cardiovascular event over 10 years by comparing a machine-learning algorithm to current guidelines from the American College of Cardiology, which include age, smoking history, cholesterol levels, and diabetes history.13 The algorithm was applied to data from approximately 82,000 patients known to have a future cardiac event. It was able to significantly improve the accuracy of cardiovascular risk prediction.13
Radiology. Researchers in the Department of Radiology at Thomas Jefferson University Hospital trained 2 convolutional neural networks (CNNs), AlexNet and GoogleNet, on 150 chest X-ray images to diagnose the presence or absence of tuberculosis (TB).14 They found that the CNNs could accurately classify TB on chest X-ray, with an area under the curve of 0.99.14 The best-performing AI model was a combination of the 2 networks, which had an accuracy of 96%.14
Stroke. The ALADIN trial compared an AI algorithm vs 2 trained neuroradiologists for detecting large artery occlusions on 300 CT scans.15 The algorithm had a sensitivity of 97%, a specificity of 52%, and an accuracy of 78%.15
Surgery. AI in the form of surgical robots has been around for many decades. Probably the best-known surgical robot is the da Vinci Surgical System, which was FDA-approved in 2000 for laparoscopic procedures.16 The da Vinci Surgical System functions as an extension of the human surgeon, who controls the device from a nearby console. Researchers at McGill University developed an anesthesia robot called “McSleepy” that can analyze biological information and recognize malfunctions while constantly adapting its own behavior.17
Dermatology. One study compared the use of deep CNNs vs 21 board-certified dermatologists to identify skin cancer on 2,000 biopsy-proven clinical images.18 The CNNs were capable of classifying skin cancer with a level of competence comparable to that of the dermatologists.18
Pathology. One study compared the efficacy of a CNN to that of human pathologists in detecting breast cancer metastasis to lymph nodes on microscopy images.19 The CNN detected 92.4% of the tumors, whereas the pathologists had a sensitivity of 73.2%.19
How can AI be used in psychiatry?
Artificially intelligent technologies have been used in psychiatry for several decades. One of the earliest examples is ELIZA, a computer program published by Professor Joseph Weizenbaum of the Massachusetts Institute of Technology in 1966.20 ELIZA consisted of a language analyzer and a script or a set of rules to improvise around a certain theme; the script DOCTOR was used to simulate a Rogerian psychotherapist.20
The application of AI in psychiatry has come a long way since the pioneering work of Weizenbaum. A recent study examined AI’s ability to distinguish between an individual who had suicidal ideation vs a control group. Machine-learning algorithms were used to evaluate functional MRI scans of 34 participants (17 who had suicidal ideation and 17 controls) to identify certain neural signatures of concepts related to life and death.21 The machine-learning algorithms were able to distinguish between these 2 groups with 91% accuracy. They also were able to distinguish between individuals who attempted suicide and those who did not with 94% accuracy.21
A study from the University of Cincinnati looked at using machine learning and natural language processing to distinguish genuine suicide notes from “fake” suicide notes that had been written by a healthy control group.22 Sixty-six notes were evaluated and categorized by 11 mental health professionals (psychiatrists, social workers, and emergency medicine physicians) and 31 PGY-3 residents. The accuracy of their results was compared with that of 9 machine-learning algorithms.22 The best machine-learning algorithm accurately classified the notes 78% of the time, compared with 63% of the time for the mental health professionals and 49% of the time for the residents.22
Researchers at Vanderbilt University examined using machine learning to predict suicide risk.23 They developed algorithms to scan electronic health records of 5,167 adults, 3,250 of whom had attempted suicide. In a review of the patients’ data from 1 week to 2 years before the attempt, the algorithms looked for certain predictors of suicide attempts, including recurrent depression, psychotic disorder, and substance use. The algorithm was 80% accurate at predicting whether a patient would attempt suicide within the next 2 years, and 84% accurate at predicting an attempt within the next week.23
Continue to: In a prospective study...
In a prospective study, researchers at Cincinnati Children’s Hospital used a machine-learning algorithm to evaluate 379 patients who were categorized into 3 groups: suicidal, mentally ill but not suicidal, or controls.24 All participants completed a standardized behavioral rating scale and participated in a semi-structured interview. Based on the participants’ linguistic and acoustic characteristics, the algorithm was able to classify them into the 3 groups with 85% accuracy.24
Many studies have looked at using language analysis to predicting the risk of psychosis in at-risk individuals. In one study, researchers evaluated individuals known to be at high risk for developing psychosis, some of whom eventually did develop psychosis.25 Participants were asked to retell a story and to answer questions about that story. Researchers fed the transcripts of these interviews into a language analysis program that looked at semantic coherence, syntactic complexity, and other factors. The algorithm was able to predict the future occurrence of psychosis with 82% accuracy. Participants who converted to psychosis had decreased semantic coherence and reduced syntactic complexity.25
A similar study looked at 34 at-risk youth in an attempt to predict who would develop psychosis based on speech pattern analysis.26 The participants underwent baseline interviews and were assessed quarterly for 2.5 years. The algorithm was able to predict who would develop psychosis with 100% accuracy.26
Challenges and limitations
The amount of research about applying machine learning to various fields of psychiatry continues to grow. With this increased interest, there have been reports of bias and human influence in the various stages of machine learning. Therefore, being aware of these challenges and engaging in practices to minimize their effects are necessary. Such practices include providing more details on data collection and processing, and constantly evaluating machine learning models for their relevance and utility to the research question proposed.27
As is the case with most innovative, fast-growing technologies, AI has its fair share of criticisms and concerns. Critics have focused on the potential threat of privacy issues, medical errors, and ethical concerns. Researchers at the Stanford Center for Biomedical Ethics emphasize the importance of being aware of the different types of bias that humans and algorithm designs can introduce into health data.28
Continue to: The Nuffield Council on Bioethics...
The Nuffield Council on Bioethics also emphasizes the importance of identifying the ethical issues raised by using AI in health care. Concerns include erroneous decisions made by AI and determining who is responsible for such errors, difficulty in validating the outputs of AI systems, and the potential for AI to be used for malicious purposes.29
For clinicians who are considering implementing AI into their practice, it is vital to recognize where this technology belongs in a workflow and in the decision-making process. Jeffery Axt, a researcher on the clinical applications of AI, encourages clinicians to view using AI as a consulting tool to eliminate the element of fear associated with not having control over diagnostics and management.30
What’s on the horizon
Research into using AI in psychiatry has drawn the attention of large companies. IBM is building an automated speech analysis application that uses machine learning to provide a real-time overview of a patient’s mental health.31 Social media platforms are also starting to incorporate AI technologies to scan posts for language and image patterns suggestive of suicidal thoughts or behavior.32
“Chat bots”—AI that can conduct a conversation in natural language—are becoming popular as well. Woebot is a cognitive-behavioral therapy–based chat bot designed by a Stanford psychologist that can be accessed through Facebook Messenger. In a 2-week study, 70 young adults (age 18 to 28) with depression were randomly assigned to use Woebot or to read mental health e-books.33 Participants who used Woebot experienced a significant reduction in depressive symptoms as measured by change in score on the Patient Health Questionnaire-9, while those assigned to the reading group did not.33
Other researchers have focused on identifying patterns of inattention, hyperactivity, and impulsivity in children using AI technologies such as computer vision, machine learning, and data mining. For example, researchers at the University of Texas at Arlington and Yale University are analyzing data from watching children perform certain tasks involving attention, decision making, and emotion management.34 There have been several advances in using AI to note abnormalities in a child’s gaze pattern that might suggest autism.35
Continue to: A project at...
A project at the University of Southern California called SimSensei/Multisense uses software to track real-time behavior descriptors such as facial expressions, body postures, and acoustic features that can help identify psychological distress.36 This software is combined with a virtual human platform that communicates with the patient as a therapist would.36
The future of AI in health care appears to have great possibilities. Putting aside irrational fears of being replaced by computers one day, AI may someday be highly transformative, leading to vast improvements in patient care.
Bottom Line
Artificial intelligence (AI) —the development of computer systems able to perform tasks that normally require human intelligence—is being developed for use in a wide range of medical specialties. Potential uses in psychiatry include predicting a patient’s risk for suicide or psychosis. Privacy concerns, ethical issues, and the potential for medical errors are among the challenges of AI use in psychiatry.
Related Resources
- Durstewitz D, Koppe G, Meyer-Lindenberg A. Deep neural networks in psychiatry. Mol Psychiatry. 2019. doi:10.1038/s41380-019-0365-9.
- Kretzschmar K, Tyroll H, Pavarini G, et al; NeurOx Young People’s Advisory Group. Can your phone be your therapist? Young people’s ethical perspectives on the use of fully automated conversational agents (chatbots) in mental health support. Biomed Inform Insights. 2019;11:1178222619829083. doi: 10.1177/1178222619829083.
1. McCarthy J. What is AI? Basic questions. http://jmc.stanford.edu/artificial-intelligence/what-is-ai/index.html. Accessed July 19, 2019.
2. Oxford Reference. Artificial intelligence. http://www.oxfordreference.com/view/10.1093/oi/authority.20110803095426960. Accessed July 19, 2019.
3. Turing AM. Computing machinery and intelligence. Mind. 1950;49:433-460.
4. Robert C. Book review: machine learning, a probabilistic perspective. CHANCE. 2014;27:2:62-63.
5. Goodfellow I, Bengio Y, Courville A. Deep learning. Cambridge, MA: The MIT Press; 2016.
6. Brownlee J. Supervised and unsupervised machine learning algorithms. https://machinelearningmastery.com/supervised-and-unsupervised-machine-learning-algorithms/. Published March 16, 2016. Accessed July 19, 2019.
7. Russell S, Norvig P. Artificial intelligence: a modern approach. Upper Saddle River, NJ: Pearson; 1995.
8. Hamet P, Tremblay J. Artificial intelligence in medicine. Metabolism. 2017;69S:S36-S40.
9. The Johns Hopkins hospital launches capacity command center to enhance hospital operations. Johns Hopkins Medicine. https://www.hopkinsmedicine.org/news/media/releases/the_johns_hopkins_hospital_launches_capacity_command_center_to_enhance_hospital_operations. Published October 26, 2016. Accessed July, 19 2019.
10. U.S. healthcare leaders expect widespread adoption of artificial intelligence by 2023. Intel. https://newsroom.intel.com/news-releases/u-s-healthcare-leaders-expect-widespread-adoption-artificial-intelligence-2023/#gs.7j7yjk. Published July 2, 2018. Accessed July, 19 2019.
11. Gulshan V, Peng L, Coram M, et al. Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA. 2016;316(22):2402-2410.
12. Poplin R, Varadarajan AV, Blumer K, et al. Prediction of cardiovascular risk factors from retinal fundus photographs via deep learning. Nature Biomedical Engineering. 2018;2:158-164.
13. Weng SF, Reps J, Kai J, et al. Can machine-learning improve cardiovascular risk prediction using routine clinical data? PLoS One. 2017;12(4):e0174944. doi: 10.1371/journal.pone. 0174944.
14. Lakhani P, Sundaram B. Deep learning at chest radiography: Automated classification of pulmonary tuberculosis by using convolutional neural networks. Radiology. 2017;284(2):574-582.
15. Bluemke DA. Radiology in 2018: Are you working with ai or being replaced by AI? Radiology. 2018;287(2):365-366.
16. Kakar PN, Das J, Roy PM, et al. Robotic invasion of operation theatre and associated anaesthetic issues: A review. Indian J Anaesth. 2011;55(1):18-25.
17. World first: researchers develop completely automated anesthesia system. McGill University. https://www.mcgill.ca/newsroom/channels/news/world-first-researchers-develop-completely-automated-anesthesia-system-100263. Published May 1, 2008. Accessed July 19, 2019.
18. Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542(7639):115-118.
19. Liu Y, Gadepalli K, Norouzi M, et al. Detecting cancer metastases on gigapixel pathology images. https://arxiv.org/abs/1703.02442. Published March 8, 2017. Accessed July 19, 2019.
20. Bassett C. The computational therapeutic: exploring Weizenbaum’s ELIZA as a history of the present. AI & Soc. 2018. https://doi.org/10.1007/s00146-018-0825-9.
21. Just MA, Pan L, Cherkassky VL, et al. Machine learning of neural representations of suicide and emotion concepts identifies suicidal youth. Nat Hum Behav. 2017;1:911-919.
22. Pestian J, Nasrallah H, Matykiewicz P, et al. Suicide note classification using natural language processing: a content analysis. Biomed Inform Insights. 2010;2010(3):19-28.
23. Walsh CG, Ribeiro JD, Franklin JC. Predicting risk of suicide attempts over time through machine learning. Clinical Psychological Science. 2017;5(3):457-469.
24. Pestian JP, Sorter M, Connolly B, et al; STM Research Group. A machine learning approach to identifying the thought markers of suicidal subjects: a prospective multicenter trial. Suicide Life Threat Behav. 2017;47(1):112-121.
25. Corcoran CM, Carrillo F, Fernández-Slezak D, et al. Prediction of psychosis across protocols and risk cohorts using automated language analysis. World Psychiatry. 2018;17(1):67-75.
26. Bedi G, Carrillo F, Cecchi GA, et al. Automated analysis of free speech predicts psychosis onset in high-risk youths. NPJ Schizophr. 2015;1:15030. doi:10.1038/npjschz.2015.30.
27. Tandon N, Tandon R. Will machine learning enable us to finally cut the Gordian Knot of schizophrenia. Schizophr Bull. 2018;44(5):939-941.
28. Char DS, Shah NH, Magnus D. Implementing machine learning in health care - addressing ethical challenges. N Engl J Med. 2018;378(11):981-983.
29. Nuffield Council on Bioethics. The big ethical questions for artificial intelligence (AI) in healthcare. http://nuffieldbioethics.org/news/2018/big-ethical-questions-artificial-intelligence-ai-healthcare. Published May 15, 2018. Accessed July 19, 2019.
30. Axt J. Artificial neural networks: a systematic review of their efficacy as an innovative resource for health care practice managers. https://www.researchgate.net/publication/322101587_Running_head_ANN_EFFICACY_IN_HEALTHCARE-A_SYSTEMATIC_REVIEW_1_Artificial_Neural_Networks_A_systematic_review_of_their_efficacy_as_an_innovative_resource_for_healthcare_practice_managers. Published October 2017. Accessed July 19, 2019.
31. Cecchi G. IBM 5 in 5: with AI, our words will be a window into our mental health. IBM Research Blog. https://www.ibm.com/blogs/research/2017/1/ibm-5-in-5-our-words-will-be-the-windows-to-our-mental-health/. Published January 5, 2017. Accessed July 19, 2019.
32. Constine J. Facebook rolls out AI to detect suicidal posts before they’re reported. TechCrunch. http://tcrn.ch/2hUBi3B. Published November 27, 2017. Accessed July 19, 2019.
33. Fitzpatrick KK, Darcy A, Vierhile M. Delivering cognitive behavior therapy to young adults with symptoms of depression and anxiety using a fully automated conversational agent (Woebot): a randomized controlled trial. JMIR Ment Health. 2017;4(2):e19. doi:10.2196/mental.7785.
34. UTA researchers use artificial intelligence to assess, enhance cognitive abilities in school-aged children. University of Texas at Arlington. https://www.uta.edu/news/releases/2016/10/makedon-children-learning-difficulties.php. Published October 13, 2016. Accessed July 19, 2019.
35. Nealon C. App for early autism detection launched on World Autism Awareness Day, April 2. University at Buffalo. http://www.buffalo.edu/news/releases/2018/04/001.html. Published April 2, 2018. Accessed July 19, 2019.
36. SimSensei. University of Southern California Institute for Creative Technologies. http://ict.usc.edu/prototypes/simsensei/. Accessed July 19, 2019.
For many people, artificial intelligence (AI) brings to mind some form of humanoid robot that speaks and acts like a human. However, AI is much more than merely robotics and machines. Professor John McCarthy of Stanford University, who first coined the term “artificial intelligence” in the early 1950s, defined it as “the science and engineering of making intelligent machines, especially intelligent computer programs”; he defined intelligence as “the computational part of the ability to achieve goals.”1 Artificial intelligence also is commonly defined as the development of computer systems able to perform tasks that normally require human intelligence.2 English Mathematician Alan Turing is considered one of the forefathers of AI research, and devised the first test to determine if a computer program was intelligent (Box 13). Today, AI has established itself as an integral part of medicine and psychiatry.
Box 1
During World War II, the English Mathematician Alan Turing helped the British government crack the Enigma machine, a coding device used by the Nazi army. He went on to pioneer many research projects in the field of artificial intelligence, including developing the Turing Test, which can determine if a computer program is intelligent.3 In this test, a human questioner uses a computer interface to pose questions to 2 respondents in different rooms; one of the respondents is a human and the other a computer program. If the questioner cannot tell the difference between the 2 respondents’ answers, then the computer program is deemed to be “artificially intelligent” because it can pass
The semantics of AI
Two subsets of AI are machine learning and deep learning.4,5 Machine learning is defined as a set of methods that can automatically detect patterns in data and then use the uncovered pattern to predict future data.4 Deep learning is a form of machine learning that enables computers to learn from experience and understand the world in terms of a hierarchy of concepts.5
Machine learning can be supervised, semi-supervised, or unsupervised. The majority of practical machine learning uses supervised learning, where all data are labeled and an algorithm is used to learn the mapping function from the input to the output. In unsupervised learning, all data are unlabeled and the algorithm models the underlying structure of the data by itself. Semi-supervised learning is a mixture of both.6
Many researchers also categorize AI into 2 types: general or “strong” AI, and narrow or “weak” AI. Strong AI is defined as computers that can think on a level at least equal to humans and are able to experience emotions and even consciousness.7 Weak AI includes adding “thinking-like” features to computers to make them more useful tools. Almost all AI technologies available today are considered to be weak AI.
AI in medicine
AI is being developed for a broad range of applications in medicine. This includes informatics approaches, including learning in health management systems such as electronic health records, and actively guiding physicians in their treatment decisions.8
AI has been applied to assist administrative workflows that reach beyond automated non-patient care activities such as chart documentation and placing orders. One example is the Judy Reitz Capacity Command Center, which was designed and built with GE Healthcare Partners.9 It combines AI technology in the form of systems engineering and predictive analytics to better manage multiple workflows in different administrative settings, including patient safety, volume, flow, and access to care.9
In April 2018, Intel Corporation surveyed 200 health-care decision makers in the United States regarding their use of AI in practice and their attitudes toward it.10 Overall, 37% of respondents reported using AI and 54% expected to increase their use of AI in the next 5 years. Clinical use of AI (77%) was more common than administrative use (41%) or financial use (26 %).10
Continue to: Box 2
Box 211-19 describes studies that evaluated the clinical use of AI in specialties other than psychiatry.
Box 2
Ophthalmology. Multiple studies have evaluated using artificial intelligence (AI) to screen for diabetic retinopathy, which is one of the fastest growing causes of blindness worldwide.11 In a recent study, researchers used a deep learning algorithm to automatically detect diabetic retinopathy and diabetic macular edema by analyzing retinal images. It was trained over a dataset of 128,000 images that were evaluated by 3 to 7 ophthalmologists. The algorithm showed high sensitivity and specificity for detecting referable diabetic retinopathy.11
Cardiology. One study looked at training a deep learning algorithm to predict cardiovascular risk based on analysis of retinal fundus images from 284,335 patients. In this study, the algorithm was able to predict a cardiovascular event in the next 5 years with 70% accuracy.12 The results were based on risk factors not previously thought to be quantifiable in retinal images, such as age, gender, smoking status, systolic blood pressure, and major adverse cardiac events.12 Similarly, researchers in the United Kingdom wanted to assess AI’s ability to predict a first cardiovascular event over 10 years by comparing a machine-learning algorithm to current guidelines from the American College of Cardiology, which include age, smoking history, cholesterol levels, and diabetes history.13 The algorithm was applied to data from approximately 82,000 patients known to have a future cardiac event. It was able to significantly improve the accuracy of cardiovascular risk prediction.13
Radiology. Researchers in the Department of Radiology at Thomas Jefferson University Hospital trained 2 convolutional neural networks (CNNs), AlexNet and GoogleNet, on 150 chest X-ray images to diagnose the presence or absence of tuberculosis (TB).14 They found that the CNNs could accurately classify TB on chest X-ray, with an area under the curve of 0.99.14 The best-performing AI model was a combination of the 2 networks, which had an accuracy of 96%.14
Stroke. The ALADIN trial compared an AI algorithm vs 2 trained neuroradiologists for detecting large artery occlusions on 300 CT scans.15 The algorithm had a sensitivity of 97%, a specificity of 52%, and an accuracy of 78%.15
Surgery. AI in the form of surgical robots has been around for many decades. Probably the best-known surgical robot is the da Vinci Surgical System, which was FDA-approved in 2000 for laparoscopic procedures.16 The da Vinci Surgical System functions as an extension of the human surgeon, who controls the device from a nearby console. Researchers at McGill University developed an anesthesia robot called “McSleepy” that can analyze biological information and recognize malfunctions while constantly adapting its own behavior.17
Dermatology. One study compared the use of deep CNNs vs 21 board-certified dermatologists to identify skin cancer on 2,000 biopsy-proven clinical images.18 The CNNs were capable of classifying skin cancer with a level of competence comparable to that of the dermatologists.18
Pathology. One study compared the efficacy of a CNN to that of human pathologists in detecting breast cancer metastasis to lymph nodes on microscopy images.19 The CNN detected 92.4% of the tumors, whereas the pathologists had a sensitivity of 73.2%.19
How can AI be used in psychiatry?
Artificially intelligent technologies have been used in psychiatry for several decades. One of the earliest examples is ELIZA, a computer program published by Professor Joseph Weizenbaum of the Massachusetts Institute of Technology in 1966.20 ELIZA consisted of a language analyzer and a script or a set of rules to improvise around a certain theme; the script DOCTOR was used to simulate a Rogerian psychotherapist.20
The application of AI in psychiatry has come a long way since the pioneering work of Weizenbaum. A recent study examined AI’s ability to distinguish between an individual who had suicidal ideation vs a control group. Machine-learning algorithms were used to evaluate functional MRI scans of 34 participants (17 who had suicidal ideation and 17 controls) to identify certain neural signatures of concepts related to life and death.21 The machine-learning algorithms were able to distinguish between these 2 groups with 91% accuracy. They also were able to distinguish between individuals who attempted suicide and those who did not with 94% accuracy.21
A study from the University of Cincinnati looked at using machine learning and natural language processing to distinguish genuine suicide notes from “fake” suicide notes that had been written by a healthy control group.22 Sixty-six notes were evaluated and categorized by 11 mental health professionals (psychiatrists, social workers, and emergency medicine physicians) and 31 PGY-3 residents. The accuracy of their results was compared with that of 9 machine-learning algorithms.22 The best machine-learning algorithm accurately classified the notes 78% of the time, compared with 63% of the time for the mental health professionals and 49% of the time for the residents.22
Researchers at Vanderbilt University examined using machine learning to predict suicide risk.23 They developed algorithms to scan electronic health records of 5,167 adults, 3,250 of whom had attempted suicide. In a review of the patients’ data from 1 week to 2 years before the attempt, the algorithms looked for certain predictors of suicide attempts, including recurrent depression, psychotic disorder, and substance use. The algorithm was 80% accurate at predicting whether a patient would attempt suicide within the next 2 years, and 84% accurate at predicting an attempt within the next week.23
Continue to: In a prospective study...
In a prospective study, researchers at Cincinnati Children’s Hospital used a machine-learning algorithm to evaluate 379 patients who were categorized into 3 groups: suicidal, mentally ill but not suicidal, or controls.24 All participants completed a standardized behavioral rating scale and participated in a semi-structured interview. Based on the participants’ linguistic and acoustic characteristics, the algorithm was able to classify them into the 3 groups with 85% accuracy.24
Many studies have looked at using language analysis to predicting the risk of psychosis in at-risk individuals. In one study, researchers evaluated individuals known to be at high risk for developing psychosis, some of whom eventually did develop psychosis.25 Participants were asked to retell a story and to answer questions about that story. Researchers fed the transcripts of these interviews into a language analysis program that looked at semantic coherence, syntactic complexity, and other factors. The algorithm was able to predict the future occurrence of psychosis with 82% accuracy. Participants who converted to psychosis had decreased semantic coherence and reduced syntactic complexity.25
A similar study looked at 34 at-risk youth in an attempt to predict who would develop psychosis based on speech pattern analysis.26 The participants underwent baseline interviews and were assessed quarterly for 2.5 years. The algorithm was able to predict who would develop psychosis with 100% accuracy.26
Challenges and limitations
The amount of research about applying machine learning to various fields of psychiatry continues to grow. With this increased interest, there have been reports of bias and human influence in the various stages of machine learning. Therefore, being aware of these challenges and engaging in practices to minimize their effects are necessary. Such practices include providing more details on data collection and processing, and constantly evaluating machine learning models for their relevance and utility to the research question proposed.27
As is the case with most innovative, fast-growing technologies, AI has its fair share of criticisms and concerns. Critics have focused on the potential threat of privacy issues, medical errors, and ethical concerns. Researchers at the Stanford Center for Biomedical Ethics emphasize the importance of being aware of the different types of bias that humans and algorithm designs can introduce into health data.28
Continue to: The Nuffield Council on Bioethics...
The Nuffield Council on Bioethics also emphasizes the importance of identifying the ethical issues raised by using AI in health care. Concerns include erroneous decisions made by AI and determining who is responsible for such errors, difficulty in validating the outputs of AI systems, and the potential for AI to be used for malicious purposes.29
For clinicians who are considering implementing AI into their practice, it is vital to recognize where this technology belongs in a workflow and in the decision-making process. Jeffery Axt, a researcher on the clinical applications of AI, encourages clinicians to view using AI as a consulting tool to eliminate the element of fear associated with not having control over diagnostics and management.30
What’s on the horizon
Research into using AI in psychiatry has drawn the attention of large companies. IBM is building an automated speech analysis application that uses machine learning to provide a real-time overview of a patient’s mental health.31 Social media platforms are also starting to incorporate AI technologies to scan posts for language and image patterns suggestive of suicidal thoughts or behavior.32
“Chat bots”—AI that can conduct a conversation in natural language—are becoming popular as well. Woebot is a cognitive-behavioral therapy–based chat bot designed by a Stanford psychologist that can be accessed through Facebook Messenger. In a 2-week study, 70 young adults (age 18 to 28) with depression were randomly assigned to use Woebot or to read mental health e-books.33 Participants who used Woebot experienced a significant reduction in depressive symptoms as measured by change in score on the Patient Health Questionnaire-9, while those assigned to the reading group did not.33
Other researchers have focused on identifying patterns of inattention, hyperactivity, and impulsivity in children using AI technologies such as computer vision, machine learning, and data mining. For example, researchers at the University of Texas at Arlington and Yale University are analyzing data from watching children perform certain tasks involving attention, decision making, and emotion management.34 There have been several advances in using AI to note abnormalities in a child’s gaze pattern that might suggest autism.35
Continue to: A project at...
A project at the University of Southern California called SimSensei/Multisense uses software to track real-time behavior descriptors such as facial expressions, body postures, and acoustic features that can help identify psychological distress.36 This software is combined with a virtual human platform that communicates with the patient as a therapist would.36
The future of AI in health care appears to have great possibilities. Putting aside irrational fears of being replaced by computers one day, AI may someday be highly transformative, leading to vast improvements in patient care.
Bottom Line
Artificial intelligence (AI) —the development of computer systems able to perform tasks that normally require human intelligence—is being developed for use in a wide range of medical specialties. Potential uses in psychiatry include predicting a patient’s risk for suicide or psychosis. Privacy concerns, ethical issues, and the potential for medical errors are among the challenges of AI use in psychiatry.
Related Resources
- Durstewitz D, Koppe G, Meyer-Lindenberg A. Deep neural networks in psychiatry. Mol Psychiatry. 2019. doi:10.1038/s41380-019-0365-9.
- Kretzschmar K, Tyroll H, Pavarini G, et al; NeurOx Young People’s Advisory Group. Can your phone be your therapist? Young people’s ethical perspectives on the use of fully automated conversational agents (chatbots) in mental health support. Biomed Inform Insights. 2019;11:1178222619829083. doi: 10.1177/1178222619829083.
For many people, artificial intelligence (AI) brings to mind some form of humanoid robot that speaks and acts like a human. However, AI is much more than merely robotics and machines. Professor John McCarthy of Stanford University, who first coined the term “artificial intelligence” in the early 1950s, defined it as “the science and engineering of making intelligent machines, especially intelligent computer programs”; he defined intelligence as “the computational part of the ability to achieve goals.”1 Artificial intelligence also is commonly defined as the development of computer systems able to perform tasks that normally require human intelligence.2 English Mathematician Alan Turing is considered one of the forefathers of AI research, and devised the first test to determine if a computer program was intelligent (Box 13). Today, AI has established itself as an integral part of medicine and psychiatry.
Box 1
During World War II, the English Mathematician Alan Turing helped the British government crack the Enigma machine, a coding device used by the Nazi army. He went on to pioneer many research projects in the field of artificial intelligence, including developing the Turing Test, which can determine if a computer program is intelligent.3 In this test, a human questioner uses a computer interface to pose questions to 2 respondents in different rooms; one of the respondents is a human and the other a computer program. If the questioner cannot tell the difference between the 2 respondents’ answers, then the computer program is deemed to be “artificially intelligent” because it can pass
The semantics of AI
Two subsets of AI are machine learning and deep learning.4,5 Machine learning is defined as a set of methods that can automatically detect patterns in data and then use the uncovered pattern to predict future data.4 Deep learning is a form of machine learning that enables computers to learn from experience and understand the world in terms of a hierarchy of concepts.5
Machine learning can be supervised, semi-supervised, or unsupervised. The majority of practical machine learning uses supervised learning, where all data are labeled and an algorithm is used to learn the mapping function from the input to the output. In unsupervised learning, all data are unlabeled and the algorithm models the underlying structure of the data by itself. Semi-supervised learning is a mixture of both.6
Many researchers also categorize AI into 2 types: general or “strong” AI, and narrow or “weak” AI. Strong AI is defined as computers that can think on a level at least equal to humans and are able to experience emotions and even consciousness.7 Weak AI includes adding “thinking-like” features to computers to make them more useful tools. Almost all AI technologies available today are considered to be weak AI.
AI in medicine
AI is being developed for a broad range of applications in medicine. This includes informatics approaches, including learning in health management systems such as electronic health records, and actively guiding physicians in their treatment decisions.8
AI has been applied to assist administrative workflows that reach beyond automated non-patient care activities such as chart documentation and placing orders. One example is the Judy Reitz Capacity Command Center, which was designed and built with GE Healthcare Partners.9 It combines AI technology in the form of systems engineering and predictive analytics to better manage multiple workflows in different administrative settings, including patient safety, volume, flow, and access to care.9
In April 2018, Intel Corporation surveyed 200 health-care decision makers in the United States regarding their use of AI in practice and their attitudes toward it.10 Overall, 37% of respondents reported using AI and 54% expected to increase their use of AI in the next 5 years. Clinical use of AI (77%) was more common than administrative use (41%) or financial use (26 %).10
Continue to: Box 2
Box 211-19 describes studies that evaluated the clinical use of AI in specialties other than psychiatry.
Box 2
Ophthalmology. Multiple studies have evaluated using artificial intelligence (AI) to screen for diabetic retinopathy, which is one of the fastest growing causes of blindness worldwide.11 In a recent study, researchers used a deep learning algorithm to automatically detect diabetic retinopathy and diabetic macular edema by analyzing retinal images. It was trained over a dataset of 128,000 images that were evaluated by 3 to 7 ophthalmologists. The algorithm showed high sensitivity and specificity for detecting referable diabetic retinopathy.11
Cardiology. One study looked at training a deep learning algorithm to predict cardiovascular risk based on analysis of retinal fundus images from 284,335 patients. In this study, the algorithm was able to predict a cardiovascular event in the next 5 years with 70% accuracy.12 The results were based on risk factors not previously thought to be quantifiable in retinal images, such as age, gender, smoking status, systolic blood pressure, and major adverse cardiac events.12 Similarly, researchers in the United Kingdom wanted to assess AI’s ability to predict a first cardiovascular event over 10 years by comparing a machine-learning algorithm to current guidelines from the American College of Cardiology, which include age, smoking history, cholesterol levels, and diabetes history.13 The algorithm was applied to data from approximately 82,000 patients known to have a future cardiac event. It was able to significantly improve the accuracy of cardiovascular risk prediction.13
Radiology. Researchers in the Department of Radiology at Thomas Jefferson University Hospital trained 2 convolutional neural networks (CNNs), AlexNet and GoogleNet, on 150 chest X-ray images to diagnose the presence or absence of tuberculosis (TB).14 They found that the CNNs could accurately classify TB on chest X-ray, with an area under the curve of 0.99.14 The best-performing AI model was a combination of the 2 networks, which had an accuracy of 96%.14
Stroke. The ALADIN trial compared an AI algorithm vs 2 trained neuroradiologists for detecting large artery occlusions on 300 CT scans.15 The algorithm had a sensitivity of 97%, a specificity of 52%, and an accuracy of 78%.15
Surgery. AI in the form of surgical robots has been around for many decades. Probably the best-known surgical robot is the da Vinci Surgical System, which was FDA-approved in 2000 for laparoscopic procedures.16 The da Vinci Surgical System functions as an extension of the human surgeon, who controls the device from a nearby console. Researchers at McGill University developed an anesthesia robot called “McSleepy” that can analyze biological information and recognize malfunctions while constantly adapting its own behavior.17
Dermatology. One study compared the use of deep CNNs vs 21 board-certified dermatologists to identify skin cancer on 2,000 biopsy-proven clinical images.18 The CNNs were capable of classifying skin cancer with a level of competence comparable to that of the dermatologists.18
Pathology. One study compared the efficacy of a CNN to that of human pathologists in detecting breast cancer metastasis to lymph nodes on microscopy images.19 The CNN detected 92.4% of the tumors, whereas the pathologists had a sensitivity of 73.2%.19
How can AI be used in psychiatry?
Artificially intelligent technologies have been used in psychiatry for several decades. One of the earliest examples is ELIZA, a computer program published by Professor Joseph Weizenbaum of the Massachusetts Institute of Technology in 1966.20 ELIZA consisted of a language analyzer and a script or a set of rules to improvise around a certain theme; the script DOCTOR was used to simulate a Rogerian psychotherapist.20
The application of AI in psychiatry has come a long way since the pioneering work of Weizenbaum. A recent study examined AI’s ability to distinguish between an individual who had suicidal ideation vs a control group. Machine-learning algorithms were used to evaluate functional MRI scans of 34 participants (17 who had suicidal ideation and 17 controls) to identify certain neural signatures of concepts related to life and death.21 The machine-learning algorithms were able to distinguish between these 2 groups with 91% accuracy. They also were able to distinguish between individuals who attempted suicide and those who did not with 94% accuracy.21
A study from the University of Cincinnati looked at using machine learning and natural language processing to distinguish genuine suicide notes from “fake” suicide notes that had been written by a healthy control group.22 Sixty-six notes were evaluated and categorized by 11 mental health professionals (psychiatrists, social workers, and emergency medicine physicians) and 31 PGY-3 residents. The accuracy of their results was compared with that of 9 machine-learning algorithms.22 The best machine-learning algorithm accurately classified the notes 78% of the time, compared with 63% of the time for the mental health professionals and 49% of the time for the residents.22
Researchers at Vanderbilt University examined using machine learning to predict suicide risk.23 They developed algorithms to scan electronic health records of 5,167 adults, 3,250 of whom had attempted suicide. In a review of the patients’ data from 1 week to 2 years before the attempt, the algorithms looked for certain predictors of suicide attempts, including recurrent depression, psychotic disorder, and substance use. The algorithm was 80% accurate at predicting whether a patient would attempt suicide within the next 2 years, and 84% accurate at predicting an attempt within the next week.23
Continue to: In a prospective study...
In a prospective study, researchers at Cincinnati Children’s Hospital used a machine-learning algorithm to evaluate 379 patients who were categorized into 3 groups: suicidal, mentally ill but not suicidal, or controls.24 All participants completed a standardized behavioral rating scale and participated in a semi-structured interview. Based on the participants’ linguistic and acoustic characteristics, the algorithm was able to classify them into the 3 groups with 85% accuracy.24
Many studies have looked at using language analysis to predicting the risk of psychosis in at-risk individuals. In one study, researchers evaluated individuals known to be at high risk for developing psychosis, some of whom eventually did develop psychosis.25 Participants were asked to retell a story and to answer questions about that story. Researchers fed the transcripts of these interviews into a language analysis program that looked at semantic coherence, syntactic complexity, and other factors. The algorithm was able to predict the future occurrence of psychosis with 82% accuracy. Participants who converted to psychosis had decreased semantic coherence and reduced syntactic complexity.25
A similar study looked at 34 at-risk youth in an attempt to predict who would develop psychosis based on speech pattern analysis.26 The participants underwent baseline interviews and were assessed quarterly for 2.5 years. The algorithm was able to predict who would develop psychosis with 100% accuracy.26
Challenges and limitations
The amount of research about applying machine learning to various fields of psychiatry continues to grow. With this increased interest, there have been reports of bias and human influence in the various stages of machine learning. Therefore, being aware of these challenges and engaging in practices to minimize their effects are necessary. Such practices include providing more details on data collection and processing, and constantly evaluating machine learning models for their relevance and utility to the research question proposed.27
As is the case with most innovative, fast-growing technologies, AI has its fair share of criticisms and concerns. Critics have focused on the potential threat of privacy issues, medical errors, and ethical concerns. Researchers at the Stanford Center for Biomedical Ethics emphasize the importance of being aware of the different types of bias that humans and algorithm designs can introduce into health data.28
Continue to: The Nuffield Council on Bioethics...
The Nuffield Council on Bioethics also emphasizes the importance of identifying the ethical issues raised by using AI in health care. Concerns include erroneous decisions made by AI and determining who is responsible for such errors, difficulty in validating the outputs of AI systems, and the potential for AI to be used for malicious purposes.29
For clinicians who are considering implementing AI into their practice, it is vital to recognize where this technology belongs in a workflow and in the decision-making process. Jeffery Axt, a researcher on the clinical applications of AI, encourages clinicians to view using AI as a consulting tool to eliminate the element of fear associated with not having control over diagnostics and management.30
What’s on the horizon
Research into using AI in psychiatry has drawn the attention of large companies. IBM is building an automated speech analysis application that uses machine learning to provide a real-time overview of a patient’s mental health.31 Social media platforms are also starting to incorporate AI technologies to scan posts for language and image patterns suggestive of suicidal thoughts or behavior.32
“Chat bots”—AI that can conduct a conversation in natural language—are becoming popular as well. Woebot is a cognitive-behavioral therapy–based chat bot designed by a Stanford psychologist that can be accessed through Facebook Messenger. In a 2-week study, 70 young adults (age 18 to 28) with depression were randomly assigned to use Woebot or to read mental health e-books.33 Participants who used Woebot experienced a significant reduction in depressive symptoms as measured by change in score on the Patient Health Questionnaire-9, while those assigned to the reading group did not.33
Other researchers have focused on identifying patterns of inattention, hyperactivity, and impulsivity in children using AI technologies such as computer vision, machine learning, and data mining. For example, researchers at the University of Texas at Arlington and Yale University are analyzing data from watching children perform certain tasks involving attention, decision making, and emotion management.34 There have been several advances in using AI to note abnormalities in a child’s gaze pattern that might suggest autism.35
Continue to: A project at...
A project at the University of Southern California called SimSensei/Multisense uses software to track real-time behavior descriptors such as facial expressions, body postures, and acoustic features that can help identify psychological distress.36 This software is combined with a virtual human platform that communicates with the patient as a therapist would.36
The future of AI in health care appears to have great possibilities. Putting aside irrational fears of being replaced by computers one day, AI may someday be highly transformative, leading to vast improvements in patient care.
Bottom Line
Artificial intelligence (AI) —the development of computer systems able to perform tasks that normally require human intelligence—is being developed for use in a wide range of medical specialties. Potential uses in psychiatry include predicting a patient’s risk for suicide or psychosis. Privacy concerns, ethical issues, and the potential for medical errors are among the challenges of AI use in psychiatry.
Related Resources
- Durstewitz D, Koppe G, Meyer-Lindenberg A. Deep neural networks in psychiatry. Mol Psychiatry. 2019. doi:10.1038/s41380-019-0365-9.
- Kretzschmar K, Tyroll H, Pavarini G, et al; NeurOx Young People’s Advisory Group. Can your phone be your therapist? Young people’s ethical perspectives on the use of fully automated conversational agents (chatbots) in mental health support. Biomed Inform Insights. 2019;11:1178222619829083. doi: 10.1177/1178222619829083.
1. McCarthy J. What is AI? Basic questions. http://jmc.stanford.edu/artificial-intelligence/what-is-ai/index.html. Accessed July 19, 2019.
2. Oxford Reference. Artificial intelligence. http://www.oxfordreference.com/view/10.1093/oi/authority.20110803095426960. Accessed July 19, 2019.
3. Turing AM. Computing machinery and intelligence. Mind. 1950;49:433-460.
4. Robert C. Book review: machine learning, a probabilistic perspective. CHANCE. 2014;27:2:62-63.
5. Goodfellow I, Bengio Y, Courville A. Deep learning. Cambridge, MA: The MIT Press; 2016.
6. Brownlee J. Supervised and unsupervised machine learning algorithms. https://machinelearningmastery.com/supervised-and-unsupervised-machine-learning-algorithms/. Published March 16, 2016. Accessed July 19, 2019.
7. Russell S, Norvig P. Artificial intelligence: a modern approach. Upper Saddle River, NJ: Pearson; 1995.
8. Hamet P, Tremblay J. Artificial intelligence in medicine. Metabolism. 2017;69S:S36-S40.
9. The Johns Hopkins hospital launches capacity command center to enhance hospital operations. Johns Hopkins Medicine. https://www.hopkinsmedicine.org/news/media/releases/the_johns_hopkins_hospital_launches_capacity_command_center_to_enhance_hospital_operations. Published October 26, 2016. Accessed July, 19 2019.
10. U.S. healthcare leaders expect widespread adoption of artificial intelligence by 2023. Intel. https://newsroom.intel.com/news-releases/u-s-healthcare-leaders-expect-widespread-adoption-artificial-intelligence-2023/#gs.7j7yjk. Published July 2, 2018. Accessed July, 19 2019.
11. Gulshan V, Peng L, Coram M, et al. Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA. 2016;316(22):2402-2410.
12. Poplin R, Varadarajan AV, Blumer K, et al. Prediction of cardiovascular risk factors from retinal fundus photographs via deep learning. Nature Biomedical Engineering. 2018;2:158-164.
13. Weng SF, Reps J, Kai J, et al. Can machine-learning improve cardiovascular risk prediction using routine clinical data? PLoS One. 2017;12(4):e0174944. doi: 10.1371/journal.pone. 0174944.
14. Lakhani P, Sundaram B. Deep learning at chest radiography: Automated classification of pulmonary tuberculosis by using convolutional neural networks. Radiology. 2017;284(2):574-582.
15. Bluemke DA. Radiology in 2018: Are you working with ai or being replaced by AI? Radiology. 2018;287(2):365-366.
16. Kakar PN, Das J, Roy PM, et al. Robotic invasion of operation theatre and associated anaesthetic issues: A review. Indian J Anaesth. 2011;55(1):18-25.
17. World first: researchers develop completely automated anesthesia system. McGill University. https://www.mcgill.ca/newsroom/channels/news/world-first-researchers-develop-completely-automated-anesthesia-system-100263. Published May 1, 2008. Accessed July 19, 2019.
18. Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542(7639):115-118.
19. Liu Y, Gadepalli K, Norouzi M, et al. Detecting cancer metastases on gigapixel pathology images. https://arxiv.org/abs/1703.02442. Published March 8, 2017. Accessed July 19, 2019.
20. Bassett C. The computational therapeutic: exploring Weizenbaum’s ELIZA as a history of the present. AI & Soc. 2018. https://doi.org/10.1007/s00146-018-0825-9.
21. Just MA, Pan L, Cherkassky VL, et al. Machine learning of neural representations of suicide and emotion concepts identifies suicidal youth. Nat Hum Behav. 2017;1:911-919.
22. Pestian J, Nasrallah H, Matykiewicz P, et al. Suicide note classification using natural language processing: a content analysis. Biomed Inform Insights. 2010;2010(3):19-28.
23. Walsh CG, Ribeiro JD, Franklin JC. Predicting risk of suicide attempts over time through machine learning. Clinical Psychological Science. 2017;5(3):457-469.
24. Pestian JP, Sorter M, Connolly B, et al; STM Research Group. A machine learning approach to identifying the thought markers of suicidal subjects: a prospective multicenter trial. Suicide Life Threat Behav. 2017;47(1):112-121.
25. Corcoran CM, Carrillo F, Fernández-Slezak D, et al. Prediction of psychosis across protocols and risk cohorts using automated language analysis. World Psychiatry. 2018;17(1):67-75.
26. Bedi G, Carrillo F, Cecchi GA, et al. Automated analysis of free speech predicts psychosis onset in high-risk youths. NPJ Schizophr. 2015;1:15030. doi:10.1038/npjschz.2015.30.
27. Tandon N, Tandon R. Will machine learning enable us to finally cut the Gordian Knot of schizophrenia. Schizophr Bull. 2018;44(5):939-941.
28. Char DS, Shah NH, Magnus D. Implementing machine learning in health care - addressing ethical challenges. N Engl J Med. 2018;378(11):981-983.
29. Nuffield Council on Bioethics. The big ethical questions for artificial intelligence (AI) in healthcare. http://nuffieldbioethics.org/news/2018/big-ethical-questions-artificial-intelligence-ai-healthcare. Published May 15, 2018. Accessed July 19, 2019.
30. Axt J. Artificial neural networks: a systematic review of their efficacy as an innovative resource for health care practice managers. https://www.researchgate.net/publication/322101587_Running_head_ANN_EFFICACY_IN_HEALTHCARE-A_SYSTEMATIC_REVIEW_1_Artificial_Neural_Networks_A_systematic_review_of_their_efficacy_as_an_innovative_resource_for_healthcare_practice_managers. Published October 2017. Accessed July 19, 2019.
31. Cecchi G. IBM 5 in 5: with AI, our words will be a window into our mental health. IBM Research Blog. https://www.ibm.com/blogs/research/2017/1/ibm-5-in-5-our-words-will-be-the-windows-to-our-mental-health/. Published January 5, 2017. Accessed July 19, 2019.
32. Constine J. Facebook rolls out AI to detect suicidal posts before they’re reported. TechCrunch. http://tcrn.ch/2hUBi3B. Published November 27, 2017. Accessed July 19, 2019.
33. Fitzpatrick KK, Darcy A, Vierhile M. Delivering cognitive behavior therapy to young adults with symptoms of depression and anxiety using a fully automated conversational agent (Woebot): a randomized controlled trial. JMIR Ment Health. 2017;4(2):e19. doi:10.2196/mental.7785.
34. UTA researchers use artificial intelligence to assess, enhance cognitive abilities in school-aged children. University of Texas at Arlington. https://www.uta.edu/news/releases/2016/10/makedon-children-learning-difficulties.php. Published October 13, 2016. Accessed July 19, 2019.
35. Nealon C. App for early autism detection launched on World Autism Awareness Day, April 2. University at Buffalo. http://www.buffalo.edu/news/releases/2018/04/001.html. Published April 2, 2018. Accessed July 19, 2019.
36. SimSensei. University of Southern California Institute for Creative Technologies. http://ict.usc.edu/prototypes/simsensei/. Accessed July 19, 2019.
1. McCarthy J. What is AI? Basic questions. http://jmc.stanford.edu/artificial-intelligence/what-is-ai/index.html. Accessed July 19, 2019.
2. Oxford Reference. Artificial intelligence. http://www.oxfordreference.com/view/10.1093/oi/authority.20110803095426960. Accessed July 19, 2019.
3. Turing AM. Computing machinery and intelligence. Mind. 1950;49:433-460.
4. Robert C. Book review: machine learning, a probabilistic perspective. CHANCE. 2014;27:2:62-63.
5. Goodfellow I, Bengio Y, Courville A. Deep learning. Cambridge, MA: The MIT Press; 2016.
6. Brownlee J. Supervised and unsupervised machine learning algorithms. https://machinelearningmastery.com/supervised-and-unsupervised-machine-learning-algorithms/. Published March 16, 2016. Accessed July 19, 2019.
7. Russell S, Norvig P. Artificial intelligence: a modern approach. Upper Saddle River, NJ: Pearson; 1995.
8. Hamet P, Tremblay J. Artificial intelligence in medicine. Metabolism. 2017;69S:S36-S40.
9. The Johns Hopkins hospital launches capacity command center to enhance hospital operations. Johns Hopkins Medicine. https://www.hopkinsmedicine.org/news/media/releases/the_johns_hopkins_hospital_launches_capacity_command_center_to_enhance_hospital_operations. Published October 26, 2016. Accessed July, 19 2019.
10. U.S. healthcare leaders expect widespread adoption of artificial intelligence by 2023. Intel. https://newsroom.intel.com/news-releases/u-s-healthcare-leaders-expect-widespread-adoption-artificial-intelligence-2023/#gs.7j7yjk. Published July 2, 2018. Accessed July, 19 2019.
11. Gulshan V, Peng L, Coram M, et al. Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA. 2016;316(22):2402-2410.
12. Poplin R, Varadarajan AV, Blumer K, et al. Prediction of cardiovascular risk factors from retinal fundus photographs via deep learning. Nature Biomedical Engineering. 2018;2:158-164.
13. Weng SF, Reps J, Kai J, et al. Can machine-learning improve cardiovascular risk prediction using routine clinical data? PLoS One. 2017;12(4):e0174944. doi: 10.1371/journal.pone. 0174944.
14. Lakhani P, Sundaram B. Deep learning at chest radiography: Automated classification of pulmonary tuberculosis by using convolutional neural networks. Radiology. 2017;284(2):574-582.
15. Bluemke DA. Radiology in 2018: Are you working with ai or being replaced by AI? Radiology. 2018;287(2):365-366.
16. Kakar PN, Das J, Roy PM, et al. Robotic invasion of operation theatre and associated anaesthetic issues: A review. Indian J Anaesth. 2011;55(1):18-25.
17. World first: researchers develop completely automated anesthesia system. McGill University. https://www.mcgill.ca/newsroom/channels/news/world-first-researchers-develop-completely-automated-anesthesia-system-100263. Published May 1, 2008. Accessed July 19, 2019.
18. Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542(7639):115-118.
19. Liu Y, Gadepalli K, Norouzi M, et al. Detecting cancer metastases on gigapixel pathology images. https://arxiv.org/abs/1703.02442. Published March 8, 2017. Accessed July 19, 2019.
20. Bassett C. The computational therapeutic: exploring Weizenbaum’s ELIZA as a history of the present. AI & Soc. 2018. https://doi.org/10.1007/s00146-018-0825-9.
21. Just MA, Pan L, Cherkassky VL, et al. Machine learning of neural representations of suicide and emotion concepts identifies suicidal youth. Nat Hum Behav. 2017;1:911-919.
22. Pestian J, Nasrallah H, Matykiewicz P, et al. Suicide note classification using natural language processing: a content analysis. Biomed Inform Insights. 2010;2010(3):19-28.
23. Walsh CG, Ribeiro JD, Franklin JC. Predicting risk of suicide attempts over time through machine learning. Clinical Psychological Science. 2017;5(3):457-469.
24. Pestian JP, Sorter M, Connolly B, et al; STM Research Group. A machine learning approach to identifying the thought markers of suicidal subjects: a prospective multicenter trial. Suicide Life Threat Behav. 2017;47(1):112-121.
25. Corcoran CM, Carrillo F, Fernández-Slezak D, et al. Prediction of psychosis across protocols and risk cohorts using automated language analysis. World Psychiatry. 2018;17(1):67-75.
26. Bedi G, Carrillo F, Cecchi GA, et al. Automated analysis of free speech predicts psychosis onset in high-risk youths. NPJ Schizophr. 2015;1:15030. doi:10.1038/npjschz.2015.30.
27. Tandon N, Tandon R. Will machine learning enable us to finally cut the Gordian Knot of schizophrenia. Schizophr Bull. 2018;44(5):939-941.
28. Char DS, Shah NH, Magnus D. Implementing machine learning in health care - addressing ethical challenges. N Engl J Med. 2018;378(11):981-983.
29. Nuffield Council on Bioethics. The big ethical questions for artificial intelligence (AI) in healthcare. http://nuffieldbioethics.org/news/2018/big-ethical-questions-artificial-intelligence-ai-healthcare. Published May 15, 2018. Accessed July 19, 2019.
30. Axt J. Artificial neural networks: a systematic review of their efficacy as an innovative resource for health care practice managers. https://www.researchgate.net/publication/322101587_Running_head_ANN_EFFICACY_IN_HEALTHCARE-A_SYSTEMATIC_REVIEW_1_Artificial_Neural_Networks_A_systematic_review_of_their_efficacy_as_an_innovative_resource_for_healthcare_practice_managers. Published October 2017. Accessed July 19, 2019.
31. Cecchi G. IBM 5 in 5: with AI, our words will be a window into our mental health. IBM Research Blog. https://www.ibm.com/blogs/research/2017/1/ibm-5-in-5-our-words-will-be-the-windows-to-our-mental-health/. Published January 5, 2017. Accessed July 19, 2019.
32. Constine J. Facebook rolls out AI to detect suicidal posts before they’re reported. TechCrunch. http://tcrn.ch/2hUBi3B. Published November 27, 2017. Accessed July 19, 2019.
33. Fitzpatrick KK, Darcy A, Vierhile M. Delivering cognitive behavior therapy to young adults with symptoms of depression and anxiety using a fully automated conversational agent (Woebot): a randomized controlled trial. JMIR Ment Health. 2017;4(2):e19. doi:10.2196/mental.7785.
34. UTA researchers use artificial intelligence to assess, enhance cognitive abilities in school-aged children. University of Texas at Arlington. https://www.uta.edu/news/releases/2016/10/makedon-children-learning-difficulties.php. Published October 13, 2016. Accessed July 19, 2019.
35. Nealon C. App for early autism detection launched on World Autism Awareness Day, April 2. University at Buffalo. http://www.buffalo.edu/news/releases/2018/04/001.html. Published April 2, 2018. Accessed July 19, 2019.
36. SimSensei. University of Southern California Institute for Creative Technologies. http://ict.usc.edu/prototypes/simsensei/. Accessed July 19, 2019.
Suicidal, violent, and treatment-resistant
CASE Violent, then catatonic
Mr. T, age 52, has a long history of schizoaffective disorder, depressed type; several suicide attempts; and violent episodes. He is admitted to a mental health rehabilitation center under a forensic commitment.
Several years earlier, Mr. T had been charged with first-degree attempted murder, assault with a deadly weapon, and abuse of a dependent/geriatric adult after allegedly stabbing his mother in the upper chest and neck. At that time, Mr. T was not in psychiatric treatment and was drinking heavily. He had become obsessed with John F. Kennedy’s assassination and believed the Central Intelligence Agency (CIA), not Lee Harvey Oswald, was responsible. He feared the CIA wanted to kill him because of his knowledge, and he heard voices from his television he believed were threatening him. He acquired knives for self-protection. When his mother arrived at his apartment to take him to a psychiatric appointment, he believed she was conspiring with the CIA and attacked her. Mr. T’s mother survived her injuries. He was taken to the county jail, where psychiatric staff noted that Mr. T was psychotic.
The court found Mr. T incompetent to stand trial and sent him to a state hospital for psychiatric treatment and competency restoration. After 3 years, he was declared unable to be restored because of repeated decompensations, placed on a conservatorship, and sent back to county jail.
In the jail, Mr. T began to show signs of catatonia. He refused medications, food, and water, and became mute. He was admitted to a medical center after a 45-minute episode that appeared similar to a seizure; however, all laboratory evaluations were within normal limits, head CT was negative, and an EEG was unremarkable.
Mr. T’s catatonic state gradually resolved with increasing dosages of lorazepam, as well as clozapine. He showed improved mobility and oral intake. A month later, his train of thought was rambling and difficult to follow, circumstantial, and perseverating. However, at times he could be directed and respond to questions in a linear and logical fashion. Lorazepam was tapered, discontinued, and replaced with gabapentin because Mr. T viewed taking lorazepam as a threat to his sobriety.
Recently, Mr. T was transferred to our mental health rehabilitation center, where he expresses that he is grateful to be in a therapeutic environment. Upon admission, his medication regimen consists of clozapine, 300 mg by mouth at bedtime, duloxetine, 60 mg/d by mouth, gabapentin 600 mg by mouth 3 times a day, and docusate sodium, 250 mg/d by mouth. Our team has a discussion about the growing recognition of the pro-inflammatory state present in many patients who experience serious mental illness and the importance of augmenting standard evidence-based psychopharmacotherapy with agents that have neuroprotective properties.1,2 We offer Mr. T
[polldaddy:10375843]
The authors’ observations
Several studies have found that acute psychosis is associated with an inflammatory state, and interleukin-6 (IL-6) is a crucial biomarker. A recent meta-analysis of serum cytokines in patients with schizophrenia found that IL-6 levels were significantly increased among acutely ill patients compared with controls.3 IL-6 levels significantly decreased after treating acute episodes of schizophrenia.3 Further, levels of peripheral IL-6 mRNA levels in individuals with schizophrenia are directly correlated with severity of positive symptoms.4
Continue to: A meta-analyis reported...
A meta-analysis reported that tumor necrosis factor-alpha and IL-6 are elevated during acute psychosis3; however, IL-6 normalized with treatment, whereas tumor necrosis factor-alpha did not. This means that IL-6 is a more clinically meaningful biomarker to help gauge treatment response.
EVALUATION Elevated markers of inflammation
Laboratory testing reveals that Mr. T’s IL-6 level is 56.64 pg/mL, which is significantly elevated (reference range: 0.31 to 5.00 pg/mL). After reviewing the IL-6 results with Mr. T and explaining that there is “too much inflammation” in his brain, he agrees to take minocycline and complete follow-up IL-6 level tests to monitor his progress during treatment.
HISTORY Alcohol abuse, treatment resistance
According to Mr. T’s mother, he had met all developmental milestones and graduated from high school with plans to enter culinary school. At age 20, Mr. T began to experience psychotic symptoms, telling family members that he was being followed by FBI agents and was receiving messages from televisions. He began drinking heavily and was arrested twice for driving under the influence. In his mid-20s, he attempted suicide by overdose after his father died. Mr. T required inpatient hospitalization nearly every year thereafter. His mother, a registered nurse, was significantly involved in his care and carefully documented his treatment history.
Mr. T has had numerous medication trials, including oral and long-acting injectable risperidone, olanzapine, aripiprazole, ziprasidone, lithium, gabapentin, buspirone, quetiapine, trazodone, bupropion, and paroxetine. None of these medications were effective.
In his mid-40s, Mr. T attempted suicide by wandering into traffic and being struck by a motor vehicle. A year later, he attempted suicide by driving his car at high speed into a concrete highway median. Mr. T told first responders that he was “possessed,” and a demonic entity “forced” him to crash his car. He begged law enforcement officers at the scene to give him a gun so he could shoot himself.
Continue to: Mr. T entered an intensive outpatient treatment program...
Mr. T entered an intensive outpatient treatment program and was switched from long-acting injectable risperidone to oral aripiprazole. After taking aripiprazole for several weeks, he began to gamble compulsively at a nearby casino. Frustrated by the lack of response to psychotropic medications and his idiosyncratic response to aripiprazole, he stopped psychiatric treatment, relapsed to alcohol use, and isolated himself in his apartment shortly before stabbing his mother.
EVALUATION Pharmacogenomics testing
At the mental health rehabilitation center, Mr. T agrees to undergo pharmacogenomics testing, which suggests that he will have a normal response to selective serotonin reuptake inhibitors and is unlikely to experience adverse reactions. He does not carry the 2 alleles that place him at higher risk of serious dermatologic reactions when taking certain mood stabilizers. He is heterozygous for the C677T allele polymorphism in the MTHFR gene that is associated with reduced folic acid metabolism, moderately decreased serum folate levels, and moderately increased homocysteine levels. On the pharmacokinetic genes tested, Mr. T has the normal metabolism genotype on 5 of 6 cytochrome P450 (CYP) enzymes; he has the ultrarapid metabolizer genotype on CYP1A2. He also has normal activity and intermediate metabolizer phenotype on the 2 UGT enzymes tested, which are responsible for the glucuronidation process, a major part of phase II metabolism.
Based on these results, Mr. T’s clozapine dosage is decreased by 50% (from 300 to 150 mg/d) and he is started on fluvoxamine, 50 mg/d, because it is a strong inhibitor of CYP1A2. The reduced conversion of clozapine to norclozapine results in an average serum clozapine level of 527 ng/mL (a level of 350 ng/mL is usually therapeutic in patients with schizophrenia) and norclozapine level of 140 ng/mL (clozapine:norclozapine ratio = 3.8), which is to be expected because fluvoxamine can increase serum clozapine levels.
Due to accumulating evidence in the literature suggesting that latent infections in the CNS play a role in serious mental illnesses such as schizophrenia, Mr. T undergoes further laboratory testing.
[polldaddy:10375845]
The authors’ observations
Mr. T tested positive for TG and CMV and negative for HSV-1. We were aware of accumulating evidence that latent infections in the CNS play a role in serious mental illnesses such as schizophrenia, specifically TG5—a parasite transmitted by cats—and CMV and HSV-1,6 which are transmitted by humans. The theory that TG infection could be a factor in schizophrenia emerged in the 1990s but only in recent years received mainstream scientific attention. Toxoplasma gondii, the infectious parasite that causes toxoplasmosis, infects more than 30 million people in the United States; however, most individuals are asymptomatic because of the body’s immune response to the parasite.7
Continue to: A study of 162 individuals...
A study of 162 individuals with schizophrenia, bipolar disorder, or major depressive disorder found that this immunologic profile is associated with suicide attempts,8 which is consistent with Mr. T’s history. Research suggests that individuals with schizophrenia who have latent TG infection have a more severe form of the illness compared with patients without the infection.9-12 Many of these factors were present in Mr. T’s case (Table 18-12).
TREATMENT Improvement, then setback
Mr. T’s medication regimen at the rehabilitation center includes clozapine, 100 mg/d; minocycline, 200 mg/d; fluvoxamine, 200 mg/d; and N-acetylcysteine, 1,200 mg/d. N-acetylcysteine is an antioxidant that could ease negative symptoms of schizophrenia by reducing oxidative stress caused by free radicals.13 Mr. T makes slow but steady improvement, and his IL-6 levels drop steadily (Figure 1).
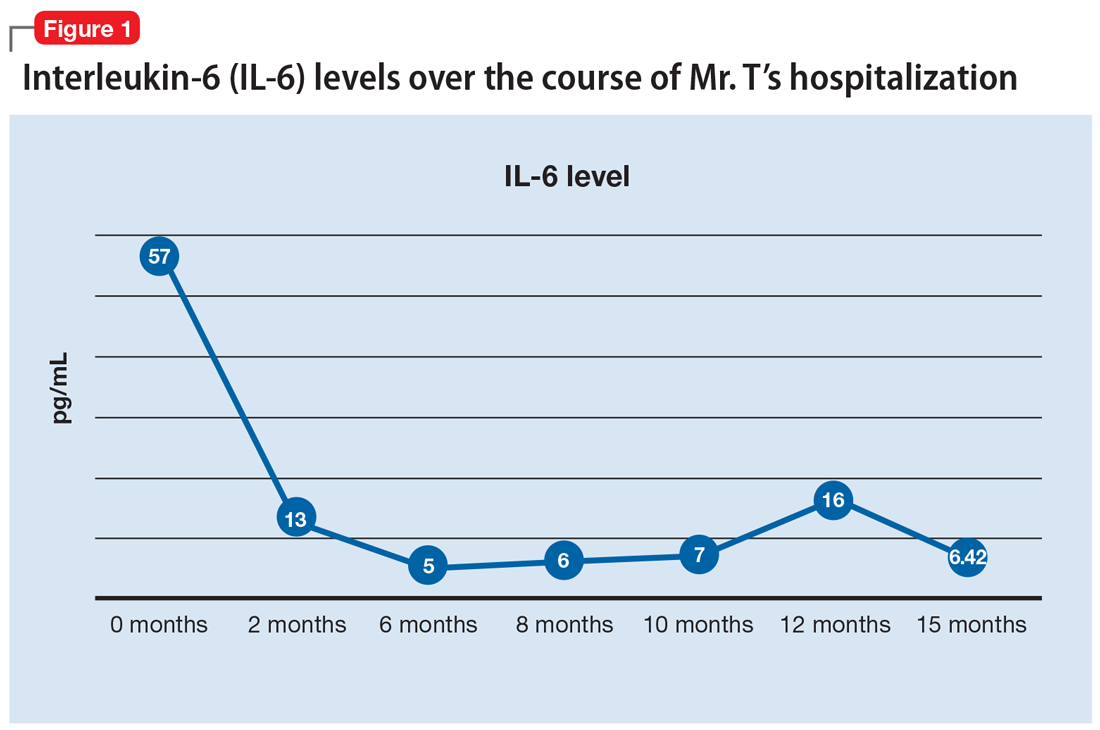
After 6 months in the rehabilitation center, Mr. T no longer experiences catatonic symptoms and is able to participate in the therapeutic program. He is permitted to leave the facility on day passes with family members. However, approximately every 8 weeks, he continues to cycle through periods of intense anxiety, perseverates on topics, and exhibits fragmented thinking and speech. During these episodes, he has difficulty receiving and processing information.
During one of these periods, Mr. T eats 4 oleander leaves he gathered while on day pass outside of the facility. After he experiences stomach pain, nausea, and vomiting, he informs nursing staff that he ate oleander. He is brought to the emergency department, receives activated charcoal and a digoxin antidote, and is placed on continuous electrocardiogram monitoring. When asked why he made the suicide attempt, he said “I realized things will never be the same because of what happened. I felt trapped.” He later expresses regret and wants to return to the mental health rehabilitation center.
At the facility, Mr. T agrees to take 2 more agents—valproic acid and ginger root extract—that specifically target latent toxoplasmosis infection before pursuing electroconvulsive therapy. We offer valproic acid because it inhibits replication of TG in an in vitro model.14 Mr. T is started on extended-release valproic acid, 1,500 mg/d, which results in a therapeutic serum level of 74.8 µg/mL.
Continue to: Additionally, Mr. T expresses interest...
Additionally, Mr. T expresses interest in taking “natural” agents in addition to psychotropics. After reviewing the quality of available ginger root extract products, Mr. T is started on a supplement that contains 22.4 mg of gingerols and 6.7 mg of shogaols, titrated to 4 capsules twice daily.
The authors’ observations
A retrospective cross-sectional analysis reported that patients with bipolar disorder who received medications with anti-toxoplasmic activity (Table 215), specifically valproic acid, had significantly fewer lifetime depressive episodes compared with patients who received medications without anti-toxoplasmic activity.15
Alternative medicine options
Research has demonstrated the beneficial effects of Chinese herbal plants for toxoplasmosis16,17 and ginger root extract has potent anti-toxoplasmic activity. A mouse model found that ginger root extract (Zingiber officinale) reduced the number of TG-infected cells by suppressing activation of apoptotic proteins the parasite induces, which prevents programmed cell death.18
Table 3 presents a stepwise approach to identifying and treating inflammation in patients with treatment-resistant psychosis.

OUTCOME Immune response, improvement
One month after the valproic acid and ginger root extract therapy is initiated, Mr. T’s toxoplasma antibody immunoglobulin G increases by 15.2 IU/mL, indicating that his immune system is mounting an enhanced response against the parasite (Figure 2). Mr. T continues to make progress while receiving the new regimen of clozapine, minocycline, valproic acid, and ginger root extract. He no longer cycles into periods of intense anxiety, perseverative thought, and fragmented thought and speech. He participates meaningfully in weekly psychotherapy and hopes to live independently and obtain gainful employment.
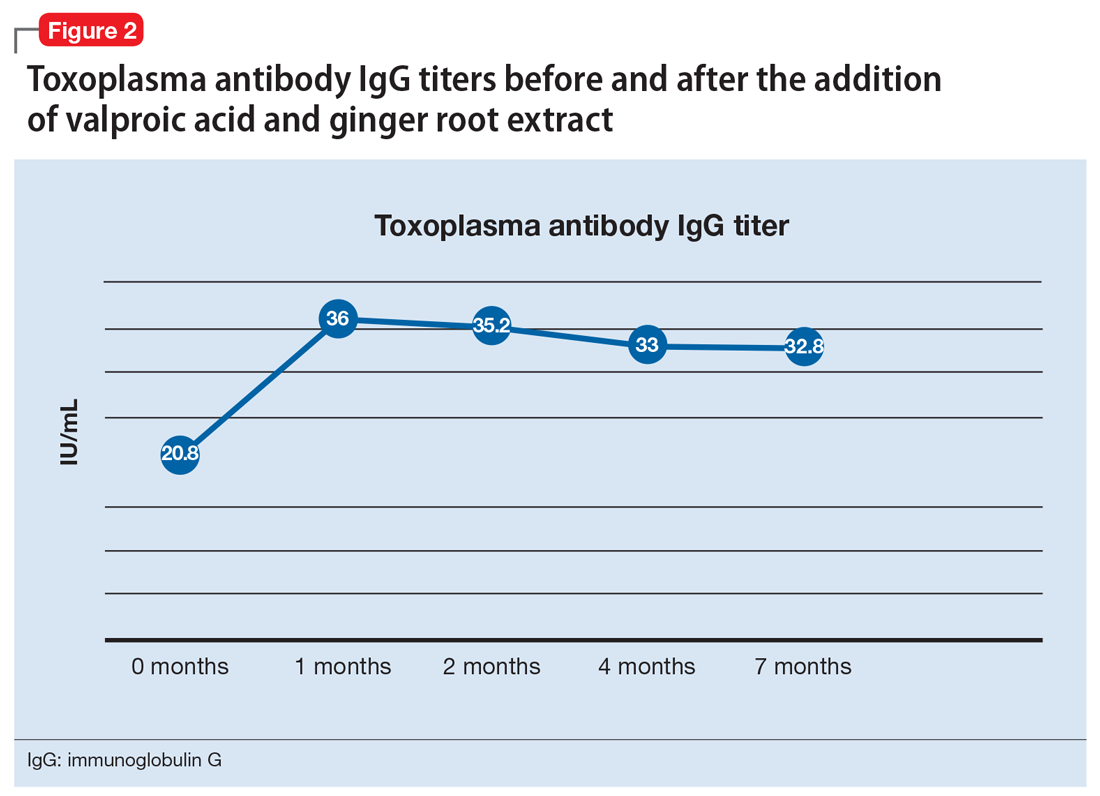
The District Attorney’s office dismisses his criminal charges, and Mr. T is discharged to a less restrictive level of care.
Continue to: Bottom Line
Bottom Line
Several studies have shown that neuroinflammation increases the severity of mental illness. Consider adjunct anti-inflammatory agents for patients who have elevated levels of inflammatory biomarkers and for whom standard treatment approaches do not adequately control psychiatric symptoms. Also consider testing for the presence of latent infections in the CNS, which could reveal the underlying cause of treatment resistance or the genesis of disabling psychiatric symptoms.
Related Resources
- Fond G, Macgregor A, Tamouza R, et al. Comparative analysis of anti-toxoplasmic activity of antipsychotic drugs and valproate. Eur Arch Psychiatry Clin Neurosci. 2014;264(2):179-183.
- Hamdani N, Daban-Huard C, Lajnef M, et al. Cognitive deterioration among bipolar disorder patients infected by Toxoplasma gondii is correlated to interleukin 6 levels. J Affect Disord. 2015;179:161-166.
- Monroe JM, Buckley PF, Miller BJ. Meta-analysis of antitoxoplasma gondii IgM antibodies in acute psychosis. Schizophr Bull. 2015;41(4):989-998.
Drug Brand Names
Acyclovir • Zovirax
Aripiprazole • Abilify
Bupropion • Wellbutrin
Buspirone • Buspar
Clozapine • Clozaril
Duloxetine • Cymbalta
Fluphenazine • Prolixin
Fluvoxamine • Luvox
Gabapentin • Neurontin
Haloperidol • Haldol
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Loxapine • Loxitane
Minocycline • Minocin
Olanzapine • Zyprexa
Paliperidone • Invega
Paroxetine • Paxil
Quetiapine • Seroquel
Risperidone • Risperdal, Risperdal Consta
Thioridazine • Mellaril
Trifluoperazine • Stelazine
Trazodone • Desyrel
Valproic acid • Depakote
Ziprasidone • Geodon
1. Koola MM, Raines JK, Hamilton RG, et al. Can anti-inflammatory medications improve symptoms and reduce mortality in schizophrenia? Current Psychiatry. 2016;15(5):52-57.
2. Nasrallah HA. Are you neuroprotecting your patients? 10 Adjunctive therapies to consider. Current Psychiatry. 2016;15(12):12-14.
3. Goldsmith DR, Rapaport MH, Miller BJ. A meta-analysis of blood cytokine network alterations in psychiatric patients: comparisons between schizophrenia, bipolar disorder and depression. Mol Psychiatry. 2016;21(12):1696-1709.
4. Chase KA, Cone JJ, Rosen C, et al. The value of interleukin 6 as a peripheral diagnostic marker in schizophrenia. BMC Psychiatry. 2016;16:152.
5. Torrey EF, Bartko JJ, Lun ZR, et al. Antibodies to Toxoplasma gondii in patients with schizophrenia: a meta-analysis. Schizophr Bull. 2007;33(3):729-736.
6. Shirts BH, Prasad KM, Pogue-Geile MF, et al. Antibodies to cytomegalovirus and herpes simplex virus 1 associated with cognitive function in schizophrenia. Schizophr Res. 2008;106(2-3):268-274.
7. Centers for Disease Control and Prevention. Parasites - Toxoplasmosis (Toxoplasma infection). https://www.cdc.gov/parasites/toxoplasmosis/index.html. Accessed February 26, 2019.
8. Dickerson F, Wilcox HC, Adamos M, et al. Suicide attempts and markers of immune response in individuals with serious mental illness. J Psychiatr Res. 2017;87:37-43.
9. Celik T, Kartalci S, Aytas O, et al. Association between latent toxoplasmosis and clinical course of schizophrenia - continuous course of the disease is characteristic for Toxoplasma gondii-infected patients. Folia Parasitol (Praha). 2015;62. doi: 10.14411/fp.2015.015.
10. Dickerson F, Boronow J, Stallings C, et al. Toxoplasma gondii in individuals with schizophrenia: association with clinical and demographic factors and with mortality. Schizophr Bull. 2007;33(3):737-740.
11. Esshili A, Thabet S, Jemli A, et al. Toxoplasma gondii infection in schizophrenia and associated clinical features. Psychiatry Res. 2016;245:327-332.
12. Holub D, Flegr J, Dragomirecka E, et al. Differences in onset of disease and severity of psychopathology between toxoplasmosis-related and toxoplasmosis-unrelated schizophrenia. Acta Psychiatr Scand. 2013;127(3):227-238.
13. Chen AT, Chibnall JT, Nasrallah HA. Placebo-controlled augmentation trials of the antioxidant NAC in schizophrenia: a review. Ann Clin Psychiatry. 2016;28(3):190-196.
14. Jones-Brando L, Torrey EF, Yolken R. Drugs used in the treatment of schizophrenia and bipolar disorder inhibit the replication of Toxoplasma gondii. Schizophr Res. 2003;62(3):237-244.
15. Fond G, Boyer L, Gaman A, et al. Treatment with anti-toxoplasmic activity (TATA) for toxoplasma positive patients with bipolar disorders or schizophrenia: a cross-sectional study. J Psychiatr Res. 2015;63:58-64.
16. Wei HX, Wei SS, Lindsay DS, et al. A systematic review and meta-analysis of the efficacy of anti-Toxoplasma gondii medicines in humans. PLoS One. 2015;10(9):e0138204.
17. Zhuo XH, Sun HC, Huang B, et al. Evaluation of potential anti-toxoplasmosis efficiency of combined traditional herbs in a mouse model. J Zhejiang Univ Sci B. 2017;18(6):453-461.
18. Choi WH, Jiang MH, Chu JP. Antiparasitic effects of Zingiber officinale (Ginger) extract against Toxoplasma gondii. Journal of Applied Biomedicine. 2013;11:15-26.
CASE Violent, then catatonic
Mr. T, age 52, has a long history of schizoaffective disorder, depressed type; several suicide attempts; and violent episodes. He is admitted to a mental health rehabilitation center under a forensic commitment.
Several years earlier, Mr. T had been charged with first-degree attempted murder, assault with a deadly weapon, and abuse of a dependent/geriatric adult after allegedly stabbing his mother in the upper chest and neck. At that time, Mr. T was not in psychiatric treatment and was drinking heavily. He had become obsessed with John F. Kennedy’s assassination and believed the Central Intelligence Agency (CIA), not Lee Harvey Oswald, was responsible. He feared the CIA wanted to kill him because of his knowledge, and he heard voices from his television he believed were threatening him. He acquired knives for self-protection. When his mother arrived at his apartment to take him to a psychiatric appointment, he believed she was conspiring with the CIA and attacked her. Mr. T’s mother survived her injuries. He was taken to the county jail, where psychiatric staff noted that Mr. T was psychotic.
The court found Mr. T incompetent to stand trial and sent him to a state hospital for psychiatric treatment and competency restoration. After 3 years, he was declared unable to be restored because of repeated decompensations, placed on a conservatorship, and sent back to county jail.
In the jail, Mr. T began to show signs of catatonia. He refused medications, food, and water, and became mute. He was admitted to a medical center after a 45-minute episode that appeared similar to a seizure; however, all laboratory evaluations were within normal limits, head CT was negative, and an EEG was unremarkable.
Mr. T’s catatonic state gradually resolved with increasing dosages of lorazepam, as well as clozapine. He showed improved mobility and oral intake. A month later, his train of thought was rambling and difficult to follow, circumstantial, and perseverating. However, at times he could be directed and respond to questions in a linear and logical fashion. Lorazepam was tapered, discontinued, and replaced with gabapentin because Mr. T viewed taking lorazepam as a threat to his sobriety.
Recently, Mr. T was transferred to our mental health rehabilitation center, where he expresses that he is grateful to be in a therapeutic environment. Upon admission, his medication regimen consists of clozapine, 300 mg by mouth at bedtime, duloxetine, 60 mg/d by mouth, gabapentin 600 mg by mouth 3 times a day, and docusate sodium, 250 mg/d by mouth. Our team has a discussion about the growing recognition of the pro-inflammatory state present in many patients who experience serious mental illness and the importance of augmenting standard evidence-based psychopharmacotherapy with agents that have neuroprotective properties.1,2 We offer Mr. T
[polldaddy:10375843]
The authors’ observations
Several studies have found that acute psychosis is associated with an inflammatory state, and interleukin-6 (IL-6) is a crucial biomarker. A recent meta-analysis of serum cytokines in patients with schizophrenia found that IL-6 levels were significantly increased among acutely ill patients compared with controls.3 IL-6 levels significantly decreased after treating acute episodes of schizophrenia.3 Further, levels of peripheral IL-6 mRNA levels in individuals with schizophrenia are directly correlated with severity of positive symptoms.4
Continue to: A meta-analyis reported...
A meta-analysis reported that tumor necrosis factor-alpha and IL-6 are elevated during acute psychosis3; however, IL-6 normalized with treatment, whereas tumor necrosis factor-alpha did not. This means that IL-6 is a more clinically meaningful biomarker to help gauge treatment response.
EVALUATION Elevated markers of inflammation
Laboratory testing reveals that Mr. T’s IL-6 level is 56.64 pg/mL, which is significantly elevated (reference range: 0.31 to 5.00 pg/mL). After reviewing the IL-6 results with Mr. T and explaining that there is “too much inflammation” in his brain, he agrees to take minocycline and complete follow-up IL-6 level tests to monitor his progress during treatment.
HISTORY Alcohol abuse, treatment resistance
According to Mr. T’s mother, he had met all developmental milestones and graduated from high school with plans to enter culinary school. At age 20, Mr. T began to experience psychotic symptoms, telling family members that he was being followed by FBI agents and was receiving messages from televisions. He began drinking heavily and was arrested twice for driving under the influence. In his mid-20s, he attempted suicide by overdose after his father died. Mr. T required inpatient hospitalization nearly every year thereafter. His mother, a registered nurse, was significantly involved in his care and carefully documented his treatment history.
Mr. T has had numerous medication trials, including oral and long-acting injectable risperidone, olanzapine, aripiprazole, ziprasidone, lithium, gabapentin, buspirone, quetiapine, trazodone, bupropion, and paroxetine. None of these medications were effective.
In his mid-40s, Mr. T attempted suicide by wandering into traffic and being struck by a motor vehicle. A year later, he attempted suicide by driving his car at high speed into a concrete highway median. Mr. T told first responders that he was “possessed,” and a demonic entity “forced” him to crash his car. He begged law enforcement officers at the scene to give him a gun so he could shoot himself.
Continue to: Mr. T entered an intensive outpatient treatment program...
Mr. T entered an intensive outpatient treatment program and was switched from long-acting injectable risperidone to oral aripiprazole. After taking aripiprazole for several weeks, he began to gamble compulsively at a nearby casino. Frustrated by the lack of response to psychotropic medications and his idiosyncratic response to aripiprazole, he stopped psychiatric treatment, relapsed to alcohol use, and isolated himself in his apartment shortly before stabbing his mother.
EVALUATION Pharmacogenomics testing
At the mental health rehabilitation center, Mr. T agrees to undergo pharmacogenomics testing, which suggests that he will have a normal response to selective serotonin reuptake inhibitors and is unlikely to experience adverse reactions. He does not carry the 2 alleles that place him at higher risk of serious dermatologic reactions when taking certain mood stabilizers. He is heterozygous for the C677T allele polymorphism in the MTHFR gene that is associated with reduced folic acid metabolism, moderately decreased serum folate levels, and moderately increased homocysteine levels. On the pharmacokinetic genes tested, Mr. T has the normal metabolism genotype on 5 of 6 cytochrome P450 (CYP) enzymes; he has the ultrarapid metabolizer genotype on CYP1A2. He also has normal activity and intermediate metabolizer phenotype on the 2 UGT enzymes tested, which are responsible for the glucuronidation process, a major part of phase II metabolism.
Based on these results, Mr. T’s clozapine dosage is decreased by 50% (from 300 to 150 mg/d) and he is started on fluvoxamine, 50 mg/d, because it is a strong inhibitor of CYP1A2. The reduced conversion of clozapine to norclozapine results in an average serum clozapine level of 527 ng/mL (a level of 350 ng/mL is usually therapeutic in patients with schizophrenia) and norclozapine level of 140 ng/mL (clozapine:norclozapine ratio = 3.8), which is to be expected because fluvoxamine can increase serum clozapine levels.
Due to accumulating evidence in the literature suggesting that latent infections in the CNS play a role in serious mental illnesses such as schizophrenia, Mr. T undergoes further laboratory testing.
[polldaddy:10375845]
The authors’ observations
Mr. T tested positive for TG and CMV and negative for HSV-1. We were aware of accumulating evidence that latent infections in the CNS play a role in serious mental illnesses such as schizophrenia, specifically TG5—a parasite transmitted by cats—and CMV and HSV-1,6 which are transmitted by humans. The theory that TG infection could be a factor in schizophrenia emerged in the 1990s but only in recent years received mainstream scientific attention. Toxoplasma gondii, the infectious parasite that causes toxoplasmosis, infects more than 30 million people in the United States; however, most individuals are asymptomatic because of the body’s immune response to the parasite.7
Continue to: A study of 162 individuals...
A study of 162 individuals with schizophrenia, bipolar disorder, or major depressive disorder found that this immunologic profile is associated with suicide attempts,8 which is consistent with Mr. T’s history. Research suggests that individuals with schizophrenia who have latent TG infection have a more severe form of the illness compared with patients without the infection.9-12 Many of these factors were present in Mr. T’s case (Table 18-12).
TREATMENT Improvement, then setback
Mr. T’s medication regimen at the rehabilitation center includes clozapine, 100 mg/d; minocycline, 200 mg/d; fluvoxamine, 200 mg/d; and N-acetylcysteine, 1,200 mg/d. N-acetylcysteine is an antioxidant that could ease negative symptoms of schizophrenia by reducing oxidative stress caused by free radicals.13 Mr. T makes slow but steady improvement, and his IL-6 levels drop steadily (Figure 1).

After 6 months in the rehabilitation center, Mr. T no longer experiences catatonic symptoms and is able to participate in the therapeutic program. He is permitted to leave the facility on day passes with family members. However, approximately every 8 weeks, he continues to cycle through periods of intense anxiety, perseverates on topics, and exhibits fragmented thinking and speech. During these episodes, he has difficulty receiving and processing information.
During one of these periods, Mr. T eats 4 oleander leaves he gathered while on day pass outside of the facility. After he experiences stomach pain, nausea, and vomiting, he informs nursing staff that he ate oleander. He is brought to the emergency department, receives activated charcoal and a digoxin antidote, and is placed on continuous electrocardiogram monitoring. When asked why he made the suicide attempt, he said “I realized things will never be the same because of what happened. I felt trapped.” He later expresses regret and wants to return to the mental health rehabilitation center.
At the facility, Mr. T agrees to take 2 more agents—valproic acid and ginger root extract—that specifically target latent toxoplasmosis infection before pursuing electroconvulsive therapy. We offer valproic acid because it inhibits replication of TG in an in vitro model.14 Mr. T is started on extended-release valproic acid, 1,500 mg/d, which results in a therapeutic serum level of 74.8 µg/mL.
Continue to: Additionally, Mr. T expresses interest...
Additionally, Mr. T expresses interest in taking “natural” agents in addition to psychotropics. After reviewing the quality of available ginger root extract products, Mr. T is started on a supplement that contains 22.4 mg of gingerols and 6.7 mg of shogaols, titrated to 4 capsules twice daily.
The authors’ observations
A retrospective cross-sectional analysis reported that patients with bipolar disorder who received medications with anti-toxoplasmic activity (Table 215), specifically valproic acid, had significantly fewer lifetime depressive episodes compared with patients who received medications without anti-toxoplasmic activity.15
Alternative medicine options
Research has demonstrated the beneficial effects of Chinese herbal plants for toxoplasmosis16,17 and ginger root extract has potent anti-toxoplasmic activity. A mouse model found that ginger root extract (Zingiber officinale) reduced the number of TG-infected cells by suppressing activation of apoptotic proteins the parasite induces, which prevents programmed cell death.18
Table 3 presents a stepwise approach to identifying and treating inflammation in patients with treatment-resistant psychosis.

OUTCOME Immune response, improvement
One month after the valproic acid and ginger root extract therapy is initiated, Mr. T’s toxoplasma antibody immunoglobulin G increases by 15.2 IU/mL, indicating that his immune system is mounting an enhanced response against the parasite (Figure 2). Mr. T continues to make progress while receiving the new regimen of clozapine, minocycline, valproic acid, and ginger root extract. He no longer cycles into periods of intense anxiety, perseverative thought, and fragmented thought and speech. He participates meaningfully in weekly psychotherapy and hopes to live independently and obtain gainful employment.

The District Attorney’s office dismisses his criminal charges, and Mr. T is discharged to a less restrictive level of care.
Continue to: Bottom Line
Bottom Line
Several studies have shown that neuroinflammation increases the severity of mental illness. Consider adjunct anti-inflammatory agents for patients who have elevated levels of inflammatory biomarkers and for whom standard treatment approaches do not adequately control psychiatric symptoms. Also consider testing for the presence of latent infections in the CNS, which could reveal the underlying cause of treatment resistance or the genesis of disabling psychiatric symptoms.
Related Resources
- Fond G, Macgregor A, Tamouza R, et al. Comparative analysis of anti-toxoplasmic activity of antipsychotic drugs and valproate. Eur Arch Psychiatry Clin Neurosci. 2014;264(2):179-183.
- Hamdani N, Daban-Huard C, Lajnef M, et al. Cognitive deterioration among bipolar disorder patients infected by Toxoplasma gondii is correlated to interleukin 6 levels. J Affect Disord. 2015;179:161-166.
- Monroe JM, Buckley PF, Miller BJ. Meta-analysis of antitoxoplasma gondii IgM antibodies in acute psychosis. Schizophr Bull. 2015;41(4):989-998.
Drug Brand Names
Acyclovir • Zovirax
Aripiprazole • Abilify
Bupropion • Wellbutrin
Buspirone • Buspar
Clozapine • Clozaril
Duloxetine • Cymbalta
Fluphenazine • Prolixin
Fluvoxamine • Luvox
Gabapentin • Neurontin
Haloperidol • Haldol
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Loxapine • Loxitane
Minocycline • Minocin
Olanzapine • Zyprexa
Paliperidone • Invega
Paroxetine • Paxil
Quetiapine • Seroquel
Risperidone • Risperdal, Risperdal Consta
Thioridazine • Mellaril
Trifluoperazine • Stelazine
Trazodone • Desyrel
Valproic acid • Depakote
Ziprasidone • Geodon
CASE Violent, then catatonic
Mr. T, age 52, has a long history of schizoaffective disorder, depressed type; several suicide attempts; and violent episodes. He is admitted to a mental health rehabilitation center under a forensic commitment.
Several years earlier, Mr. T had been charged with first-degree attempted murder, assault with a deadly weapon, and abuse of a dependent/geriatric adult after allegedly stabbing his mother in the upper chest and neck. At that time, Mr. T was not in psychiatric treatment and was drinking heavily. He had become obsessed with John F. Kennedy’s assassination and believed the Central Intelligence Agency (CIA), not Lee Harvey Oswald, was responsible. He feared the CIA wanted to kill him because of his knowledge, and he heard voices from his television he believed were threatening him. He acquired knives for self-protection. When his mother arrived at his apartment to take him to a psychiatric appointment, he believed she was conspiring with the CIA and attacked her. Mr. T’s mother survived her injuries. He was taken to the county jail, where psychiatric staff noted that Mr. T was psychotic.
The court found Mr. T incompetent to stand trial and sent him to a state hospital for psychiatric treatment and competency restoration. After 3 years, he was declared unable to be restored because of repeated decompensations, placed on a conservatorship, and sent back to county jail.
In the jail, Mr. T began to show signs of catatonia. He refused medications, food, and water, and became mute. He was admitted to a medical center after a 45-minute episode that appeared similar to a seizure; however, all laboratory evaluations were within normal limits, head CT was negative, and an EEG was unremarkable.
Mr. T’s catatonic state gradually resolved with increasing dosages of lorazepam, as well as clozapine. He showed improved mobility and oral intake. A month later, his train of thought was rambling and difficult to follow, circumstantial, and perseverating. However, at times he could be directed and respond to questions in a linear and logical fashion. Lorazepam was tapered, discontinued, and replaced with gabapentin because Mr. T viewed taking lorazepam as a threat to his sobriety.
Recently, Mr. T was transferred to our mental health rehabilitation center, where he expresses that he is grateful to be in a therapeutic environment. Upon admission, his medication regimen consists of clozapine, 300 mg by mouth at bedtime, duloxetine, 60 mg/d by mouth, gabapentin 600 mg by mouth 3 times a day, and docusate sodium, 250 mg/d by mouth. Our team has a discussion about the growing recognition of the pro-inflammatory state present in many patients who experience serious mental illness and the importance of augmenting standard evidence-based psychopharmacotherapy with agents that have neuroprotective properties.1,2 We offer Mr. T
[polldaddy:10375843]
The authors’ observations
Several studies have found that acute psychosis is associated with an inflammatory state, and interleukin-6 (IL-6) is a crucial biomarker. A recent meta-analysis of serum cytokines in patients with schizophrenia found that IL-6 levels were significantly increased among acutely ill patients compared with controls.3 IL-6 levels significantly decreased after treating acute episodes of schizophrenia.3 Further, levels of peripheral IL-6 mRNA levels in individuals with schizophrenia are directly correlated with severity of positive symptoms.4
Continue to: A meta-analyis reported...
A meta-analysis reported that tumor necrosis factor-alpha and IL-6 are elevated during acute psychosis3; however, IL-6 normalized with treatment, whereas tumor necrosis factor-alpha did not. This means that IL-6 is a more clinically meaningful biomarker to help gauge treatment response.
EVALUATION Elevated markers of inflammation
Laboratory testing reveals that Mr. T’s IL-6 level is 56.64 pg/mL, which is significantly elevated (reference range: 0.31 to 5.00 pg/mL). After reviewing the IL-6 results with Mr. T and explaining that there is “too much inflammation” in his brain, he agrees to take minocycline and complete follow-up IL-6 level tests to monitor his progress during treatment.
HISTORY Alcohol abuse, treatment resistance
According to Mr. T’s mother, he had met all developmental milestones and graduated from high school with plans to enter culinary school. At age 20, Mr. T began to experience psychotic symptoms, telling family members that he was being followed by FBI agents and was receiving messages from televisions. He began drinking heavily and was arrested twice for driving under the influence. In his mid-20s, he attempted suicide by overdose after his father died. Mr. T required inpatient hospitalization nearly every year thereafter. His mother, a registered nurse, was significantly involved in his care and carefully documented his treatment history.
Mr. T has had numerous medication trials, including oral and long-acting injectable risperidone, olanzapine, aripiprazole, ziprasidone, lithium, gabapentin, buspirone, quetiapine, trazodone, bupropion, and paroxetine. None of these medications were effective.
In his mid-40s, Mr. T attempted suicide by wandering into traffic and being struck by a motor vehicle. A year later, he attempted suicide by driving his car at high speed into a concrete highway median. Mr. T told first responders that he was “possessed,” and a demonic entity “forced” him to crash his car. He begged law enforcement officers at the scene to give him a gun so he could shoot himself.
Continue to: Mr. T entered an intensive outpatient treatment program...
Mr. T entered an intensive outpatient treatment program and was switched from long-acting injectable risperidone to oral aripiprazole. After taking aripiprazole for several weeks, he began to gamble compulsively at a nearby casino. Frustrated by the lack of response to psychotropic medications and his idiosyncratic response to aripiprazole, he stopped psychiatric treatment, relapsed to alcohol use, and isolated himself in his apartment shortly before stabbing his mother.
EVALUATION Pharmacogenomics testing
At the mental health rehabilitation center, Mr. T agrees to undergo pharmacogenomics testing, which suggests that he will have a normal response to selective serotonin reuptake inhibitors and is unlikely to experience adverse reactions. He does not carry the 2 alleles that place him at higher risk of serious dermatologic reactions when taking certain mood stabilizers. He is heterozygous for the C677T allele polymorphism in the MTHFR gene that is associated with reduced folic acid metabolism, moderately decreased serum folate levels, and moderately increased homocysteine levels. On the pharmacokinetic genes tested, Mr. T has the normal metabolism genotype on 5 of 6 cytochrome P450 (CYP) enzymes; he has the ultrarapid metabolizer genotype on CYP1A2. He also has normal activity and intermediate metabolizer phenotype on the 2 UGT enzymes tested, which are responsible for the glucuronidation process, a major part of phase II metabolism.
Based on these results, Mr. T’s clozapine dosage is decreased by 50% (from 300 to 150 mg/d) and he is started on fluvoxamine, 50 mg/d, because it is a strong inhibitor of CYP1A2. The reduced conversion of clozapine to norclozapine results in an average serum clozapine level of 527 ng/mL (a level of 350 ng/mL is usually therapeutic in patients with schizophrenia) and norclozapine level of 140 ng/mL (clozapine:norclozapine ratio = 3.8), which is to be expected because fluvoxamine can increase serum clozapine levels.
Due to accumulating evidence in the literature suggesting that latent infections in the CNS play a role in serious mental illnesses such as schizophrenia, Mr. T undergoes further laboratory testing.
[polldaddy:10375845]
The authors’ observations
Mr. T tested positive for TG and CMV and negative for HSV-1. We were aware of accumulating evidence that latent infections in the CNS play a role in serious mental illnesses such as schizophrenia, specifically TG5—a parasite transmitted by cats—and CMV and HSV-1,6 which are transmitted by humans. The theory that TG infection could be a factor in schizophrenia emerged in the 1990s but only in recent years received mainstream scientific attention. Toxoplasma gondii, the infectious parasite that causes toxoplasmosis, infects more than 30 million people in the United States; however, most individuals are asymptomatic because of the body’s immune response to the parasite.7
Continue to: A study of 162 individuals...
A study of 162 individuals with schizophrenia, bipolar disorder, or major depressive disorder found that this immunologic profile is associated with suicide attempts,8 which is consistent with Mr. T’s history. Research suggests that individuals with schizophrenia who have latent TG infection have a more severe form of the illness compared with patients without the infection.9-12 Many of these factors were present in Mr. T’s case (Table 18-12).
TREATMENT Improvement, then setback
Mr. T’s medication regimen at the rehabilitation center includes clozapine, 100 mg/d; minocycline, 200 mg/d; fluvoxamine, 200 mg/d; and N-acetylcysteine, 1,200 mg/d. N-acetylcysteine is an antioxidant that could ease negative symptoms of schizophrenia by reducing oxidative stress caused by free radicals.13 Mr. T makes slow but steady improvement, and his IL-6 levels drop steadily (Figure 1).

After 6 months in the rehabilitation center, Mr. T no longer experiences catatonic symptoms and is able to participate in the therapeutic program. He is permitted to leave the facility on day passes with family members. However, approximately every 8 weeks, he continues to cycle through periods of intense anxiety, perseverates on topics, and exhibits fragmented thinking and speech. During these episodes, he has difficulty receiving and processing information.
During one of these periods, Mr. T eats 4 oleander leaves he gathered while on day pass outside of the facility. After he experiences stomach pain, nausea, and vomiting, he informs nursing staff that he ate oleander. He is brought to the emergency department, receives activated charcoal and a digoxin antidote, and is placed on continuous electrocardiogram monitoring. When asked why he made the suicide attempt, he said “I realized things will never be the same because of what happened. I felt trapped.” He later expresses regret and wants to return to the mental health rehabilitation center.
At the facility, Mr. T agrees to take 2 more agents—valproic acid and ginger root extract—that specifically target latent toxoplasmosis infection before pursuing electroconvulsive therapy. We offer valproic acid because it inhibits replication of TG in an in vitro model.14 Mr. T is started on extended-release valproic acid, 1,500 mg/d, which results in a therapeutic serum level of 74.8 µg/mL.
Continue to: Additionally, Mr. T expresses interest...
Additionally, Mr. T expresses interest in taking “natural” agents in addition to psychotropics. After reviewing the quality of available ginger root extract products, Mr. T is started on a supplement that contains 22.4 mg of gingerols and 6.7 mg of shogaols, titrated to 4 capsules twice daily.
The authors’ observations
A retrospective cross-sectional analysis reported that patients with bipolar disorder who received medications with anti-toxoplasmic activity (Table 215), specifically valproic acid, had significantly fewer lifetime depressive episodes compared with patients who received medications without anti-toxoplasmic activity.15
Alternative medicine options
Research has demonstrated the beneficial effects of Chinese herbal plants for toxoplasmosis16,17 and ginger root extract has potent anti-toxoplasmic activity. A mouse model found that ginger root extract (Zingiber officinale) reduced the number of TG-infected cells by suppressing activation of apoptotic proteins the parasite induces, which prevents programmed cell death.18
Table 3 presents a stepwise approach to identifying and treating inflammation in patients with treatment-resistant psychosis.

OUTCOME Immune response, improvement
One month after the valproic acid and ginger root extract therapy is initiated, Mr. T’s toxoplasma antibody immunoglobulin G increases by 15.2 IU/mL, indicating that his immune system is mounting an enhanced response against the parasite (Figure 2). Mr. T continues to make progress while receiving the new regimen of clozapine, minocycline, valproic acid, and ginger root extract. He no longer cycles into periods of intense anxiety, perseverative thought, and fragmented thought and speech. He participates meaningfully in weekly psychotherapy and hopes to live independently and obtain gainful employment.

The District Attorney’s office dismisses his criminal charges, and Mr. T is discharged to a less restrictive level of care.
Continue to: Bottom Line
Bottom Line
Several studies have shown that neuroinflammation increases the severity of mental illness. Consider adjunct anti-inflammatory agents for patients who have elevated levels of inflammatory biomarkers and for whom standard treatment approaches do not adequately control psychiatric symptoms. Also consider testing for the presence of latent infections in the CNS, which could reveal the underlying cause of treatment resistance or the genesis of disabling psychiatric symptoms.
Related Resources
- Fond G, Macgregor A, Tamouza R, et al. Comparative analysis of anti-toxoplasmic activity of antipsychotic drugs and valproate. Eur Arch Psychiatry Clin Neurosci. 2014;264(2):179-183.
- Hamdani N, Daban-Huard C, Lajnef M, et al. Cognitive deterioration among bipolar disorder patients infected by Toxoplasma gondii is correlated to interleukin 6 levels. J Affect Disord. 2015;179:161-166.
- Monroe JM, Buckley PF, Miller BJ. Meta-analysis of antitoxoplasma gondii IgM antibodies in acute psychosis. Schizophr Bull. 2015;41(4):989-998.
Drug Brand Names
Acyclovir • Zovirax
Aripiprazole • Abilify
Bupropion • Wellbutrin
Buspirone • Buspar
Clozapine • Clozaril
Duloxetine • Cymbalta
Fluphenazine • Prolixin
Fluvoxamine • Luvox
Gabapentin • Neurontin
Haloperidol • Haldol
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Loxapine • Loxitane
Minocycline • Minocin
Olanzapine • Zyprexa
Paliperidone • Invega
Paroxetine • Paxil
Quetiapine • Seroquel
Risperidone • Risperdal, Risperdal Consta
Thioridazine • Mellaril
Trifluoperazine • Stelazine
Trazodone • Desyrel
Valproic acid • Depakote
Ziprasidone • Geodon
1. Koola MM, Raines JK, Hamilton RG, et al. Can anti-inflammatory medications improve symptoms and reduce mortality in schizophrenia? Current Psychiatry. 2016;15(5):52-57.
2. Nasrallah HA. Are you neuroprotecting your patients? 10 Adjunctive therapies to consider. Current Psychiatry. 2016;15(12):12-14.
3. Goldsmith DR, Rapaport MH, Miller BJ. A meta-analysis of blood cytokine network alterations in psychiatric patients: comparisons between schizophrenia, bipolar disorder and depression. Mol Psychiatry. 2016;21(12):1696-1709.
4. Chase KA, Cone JJ, Rosen C, et al. The value of interleukin 6 as a peripheral diagnostic marker in schizophrenia. BMC Psychiatry. 2016;16:152.
5. Torrey EF, Bartko JJ, Lun ZR, et al. Antibodies to Toxoplasma gondii in patients with schizophrenia: a meta-analysis. Schizophr Bull. 2007;33(3):729-736.
6. Shirts BH, Prasad KM, Pogue-Geile MF, et al. Antibodies to cytomegalovirus and herpes simplex virus 1 associated with cognitive function in schizophrenia. Schizophr Res. 2008;106(2-3):268-274.
7. Centers for Disease Control and Prevention. Parasites - Toxoplasmosis (Toxoplasma infection). https://www.cdc.gov/parasites/toxoplasmosis/index.html. Accessed February 26, 2019.
8. Dickerson F, Wilcox HC, Adamos M, et al. Suicide attempts and markers of immune response in individuals with serious mental illness. J Psychiatr Res. 2017;87:37-43.
9. Celik T, Kartalci S, Aytas O, et al. Association between latent toxoplasmosis and clinical course of schizophrenia - continuous course of the disease is characteristic for Toxoplasma gondii-infected patients. Folia Parasitol (Praha). 2015;62. doi: 10.14411/fp.2015.015.
10. Dickerson F, Boronow J, Stallings C, et al. Toxoplasma gondii in individuals with schizophrenia: association with clinical and demographic factors and with mortality. Schizophr Bull. 2007;33(3):737-740.
11. Esshili A, Thabet S, Jemli A, et al. Toxoplasma gondii infection in schizophrenia and associated clinical features. Psychiatry Res. 2016;245:327-332.
12. Holub D, Flegr J, Dragomirecka E, et al. Differences in onset of disease and severity of psychopathology between toxoplasmosis-related and toxoplasmosis-unrelated schizophrenia. Acta Psychiatr Scand. 2013;127(3):227-238.
13. Chen AT, Chibnall JT, Nasrallah HA. Placebo-controlled augmentation trials of the antioxidant NAC in schizophrenia: a review. Ann Clin Psychiatry. 2016;28(3):190-196.
14. Jones-Brando L, Torrey EF, Yolken R. Drugs used in the treatment of schizophrenia and bipolar disorder inhibit the replication of Toxoplasma gondii. Schizophr Res. 2003;62(3):237-244.
15. Fond G, Boyer L, Gaman A, et al. Treatment with anti-toxoplasmic activity (TATA) for toxoplasma positive patients with bipolar disorders or schizophrenia: a cross-sectional study. J Psychiatr Res. 2015;63:58-64.
16. Wei HX, Wei SS, Lindsay DS, et al. A systematic review and meta-analysis of the efficacy of anti-Toxoplasma gondii medicines in humans. PLoS One. 2015;10(9):e0138204.
17. Zhuo XH, Sun HC, Huang B, et al. Evaluation of potential anti-toxoplasmosis efficiency of combined traditional herbs in a mouse model. J Zhejiang Univ Sci B. 2017;18(6):453-461.
18. Choi WH, Jiang MH, Chu JP. Antiparasitic effects of Zingiber officinale (Ginger) extract against Toxoplasma gondii. Journal of Applied Biomedicine. 2013;11:15-26.
1. Koola MM, Raines JK, Hamilton RG, et al. Can anti-inflammatory medications improve symptoms and reduce mortality in schizophrenia? Current Psychiatry. 2016;15(5):52-57.
2. Nasrallah HA. Are you neuroprotecting your patients? 10 Adjunctive therapies to consider. Current Psychiatry. 2016;15(12):12-14.
3. Goldsmith DR, Rapaport MH, Miller BJ. A meta-analysis of blood cytokine network alterations in psychiatric patients: comparisons between schizophrenia, bipolar disorder and depression. Mol Psychiatry. 2016;21(12):1696-1709.
4. Chase KA, Cone JJ, Rosen C, et al. The value of interleukin 6 as a peripheral diagnostic marker in schizophrenia. BMC Psychiatry. 2016;16:152.
5. Torrey EF, Bartko JJ, Lun ZR, et al. Antibodies to Toxoplasma gondii in patients with schizophrenia: a meta-analysis. Schizophr Bull. 2007;33(3):729-736.
6. Shirts BH, Prasad KM, Pogue-Geile MF, et al. Antibodies to cytomegalovirus and herpes simplex virus 1 associated with cognitive function in schizophrenia. Schizophr Res. 2008;106(2-3):268-274.
7. Centers for Disease Control and Prevention. Parasites - Toxoplasmosis (Toxoplasma infection). https://www.cdc.gov/parasites/toxoplasmosis/index.html. Accessed February 26, 2019.
8. Dickerson F, Wilcox HC, Adamos M, et al. Suicide attempts and markers of immune response in individuals with serious mental illness. J Psychiatr Res. 2017;87:37-43.
9. Celik T, Kartalci S, Aytas O, et al. Association between latent toxoplasmosis and clinical course of schizophrenia - continuous course of the disease is characteristic for Toxoplasma gondii-infected patients. Folia Parasitol (Praha). 2015;62. doi: 10.14411/fp.2015.015.
10. Dickerson F, Boronow J, Stallings C, et al. Toxoplasma gondii in individuals with schizophrenia: association with clinical and demographic factors and with mortality. Schizophr Bull. 2007;33(3):737-740.
11. Esshili A, Thabet S, Jemli A, et al. Toxoplasma gondii infection in schizophrenia and associated clinical features. Psychiatry Res. 2016;245:327-332.
12. Holub D, Flegr J, Dragomirecka E, et al. Differences in onset of disease and severity of psychopathology between toxoplasmosis-related and toxoplasmosis-unrelated schizophrenia. Acta Psychiatr Scand. 2013;127(3):227-238.
13. Chen AT, Chibnall JT, Nasrallah HA. Placebo-controlled augmentation trials of the antioxidant NAC in schizophrenia: a review. Ann Clin Psychiatry. 2016;28(3):190-196.
14. Jones-Brando L, Torrey EF, Yolken R. Drugs used in the treatment of schizophrenia and bipolar disorder inhibit the replication of Toxoplasma gondii. Schizophr Res. 2003;62(3):237-244.
15. Fond G, Boyer L, Gaman A, et al. Treatment with anti-toxoplasmic activity (TATA) for toxoplasma positive patients with bipolar disorders or schizophrenia: a cross-sectional study. J Psychiatr Res. 2015;63:58-64.
16. Wei HX, Wei SS, Lindsay DS, et al. A systematic review and meta-analysis of the efficacy of anti-Toxoplasma gondii medicines in humans. PLoS One. 2015;10(9):e0138204.
17. Zhuo XH, Sun HC, Huang B, et al. Evaluation of potential anti-toxoplasmosis efficiency of combined traditional herbs in a mouse model. J Zhejiang Univ Sci B. 2017;18(6):453-461.
18. Choi WH, Jiang MH, Chu JP. Antiparasitic effects of Zingiber officinale (Ginger) extract against Toxoplasma gondii. Journal of Applied Biomedicine. 2013;11:15-26.
Lumateperone schizophrenia drug seems to hit snag
FDA cancels lumateperone advisory panel
U.S. regulators canceled a July 31, 2019, advisory committee about lumateperone, an experimental schizophrenia drug that has had some mixed results in testing.
On July 23, the Food and Drug Administration announced the cancellation of the Psychopharmacologic Drugs Advisory Committee meeting it had previously called for to review the new drug application for lumateperone. The agency said the meeting was canceled because of “new information regarding the application.” The FDA said it was continuing to evaluate the application and would, as needed, announce a future meeting on it.
The developer of lumateperone, Intra-Cellular Therapies, issued its own statement on July 23, noting that a meeting had been scheduled with the FDA “shortly” and that an update would be provided after the meeting. The New York–based firm also said it recently had provided additional information to the FDA to meet agency requests. This information was related to “nonclinical studies.”
“The FDA canceled the advisory committee meeting to allow sufficient time to review this new and any forthcoming information as they continue” to review the new drug application for lumateperone, Intra-Cellular said in the July 23 statement. The company also said there may be an extension of the FDA’s Sept. 27, 2019, target action date on the lumateperone application.
Investors viewed this as bad news. Shares of Intra-Cellular on July 23 dropped from an opening price of $11.90 to a closing one of $8.19. On July 29, they closed at $8.12.
Still, it is unclear how the FDA will decide on the lumateperone application and whether the agency will call another advisory committee meeting on it.
Last year, the FDA accepted the application for lumateperone, a once-daily treatment, Intra-Cellular said. The agency had in 2017 given a fast-track designation to lumateperone for the treatment of schizophrenia.
Lumateperone is the lead product for the company.
On the company’s website, Intra-Cellular says three large randomized, double-blind, placebo-controlled trials have been done for lumateperone as a schizophrenia drug. In two of these studies, results for lumateperone at a 60-mg dose showed a “statistically significant separation from placebo on the primary endpoint, the Positive and Negative Syndrome Scale or PANSS total score.”
In a recent routine filing with the Securities and Exchange Commission, Intra-Cellular said it was having an “ongoing dialogue” with the FDA about lumateperone. The company in 2016 had announced that, in a phase 3 study known as ITI-007-302, lumateperone had not separated from placebo on the primary endpoint, change from baseline on the PANSS total score, in the predefined patient population. The active control for ITI-007-302, risperidone, did separate from placebo.
In the recent SEC filing, Intra-Cellular said the FDA already has confirmed that the results of ITI-007-302 did not preclude the submission of a new drug application.
Intra-Cellular also said “lumateperone was statistically significantly better than risperidone on key safety and tolerability parameters, and exhibited a safety profile similar to placebo” in the 302 study. Lumateperone’s failure to best placebo in the 302 test was “in part due to an unusually high placebo response at certain sites.”
FDA cancels lumateperone advisory panel
FDA cancels lumateperone advisory panel
U.S. regulators canceled a July 31, 2019, advisory committee about lumateperone, an experimental schizophrenia drug that has had some mixed results in testing.
On July 23, the Food and Drug Administration announced the cancellation of the Psychopharmacologic Drugs Advisory Committee meeting it had previously called for to review the new drug application for lumateperone. The agency said the meeting was canceled because of “new information regarding the application.” The FDA said it was continuing to evaluate the application and would, as needed, announce a future meeting on it.
The developer of lumateperone, Intra-Cellular Therapies, issued its own statement on July 23, noting that a meeting had been scheduled with the FDA “shortly” and that an update would be provided after the meeting. The New York–based firm also said it recently had provided additional information to the FDA to meet agency requests. This information was related to “nonclinical studies.”
“The FDA canceled the advisory committee meeting to allow sufficient time to review this new and any forthcoming information as they continue” to review the new drug application for lumateperone, Intra-Cellular said in the July 23 statement. The company also said there may be an extension of the FDA’s Sept. 27, 2019, target action date on the lumateperone application.
Investors viewed this as bad news. Shares of Intra-Cellular on July 23 dropped from an opening price of $11.90 to a closing one of $8.19. On July 29, they closed at $8.12.
Still, it is unclear how the FDA will decide on the lumateperone application and whether the agency will call another advisory committee meeting on it.
Last year, the FDA accepted the application for lumateperone, a once-daily treatment, Intra-Cellular said. The agency had in 2017 given a fast-track designation to lumateperone for the treatment of schizophrenia.
Lumateperone is the lead product for the company.
On the company’s website, Intra-Cellular says three large randomized, double-blind, placebo-controlled trials have been done for lumateperone as a schizophrenia drug. In two of these studies, results for lumateperone at a 60-mg dose showed a “statistically significant separation from placebo on the primary endpoint, the Positive and Negative Syndrome Scale or PANSS total score.”
In a recent routine filing with the Securities and Exchange Commission, Intra-Cellular said it was having an “ongoing dialogue” with the FDA about lumateperone. The company in 2016 had announced that, in a phase 3 study known as ITI-007-302, lumateperone had not separated from placebo on the primary endpoint, change from baseline on the PANSS total score, in the predefined patient population. The active control for ITI-007-302, risperidone, did separate from placebo.
In the recent SEC filing, Intra-Cellular said the FDA already has confirmed that the results of ITI-007-302 did not preclude the submission of a new drug application.
Intra-Cellular also said “lumateperone was statistically significantly better than risperidone on key safety and tolerability parameters, and exhibited a safety profile similar to placebo” in the 302 study. Lumateperone’s failure to best placebo in the 302 test was “in part due to an unusually high placebo response at certain sites.”
U.S. regulators canceled a July 31, 2019, advisory committee about lumateperone, an experimental schizophrenia drug that has had some mixed results in testing.
On July 23, the Food and Drug Administration announced the cancellation of the Psychopharmacologic Drugs Advisory Committee meeting it had previously called for to review the new drug application for lumateperone. The agency said the meeting was canceled because of “new information regarding the application.” The FDA said it was continuing to evaluate the application and would, as needed, announce a future meeting on it.
The developer of lumateperone, Intra-Cellular Therapies, issued its own statement on July 23, noting that a meeting had been scheduled with the FDA “shortly” and that an update would be provided after the meeting. The New York–based firm also said it recently had provided additional information to the FDA to meet agency requests. This information was related to “nonclinical studies.”
“The FDA canceled the advisory committee meeting to allow sufficient time to review this new and any forthcoming information as they continue” to review the new drug application for lumateperone, Intra-Cellular said in the July 23 statement. The company also said there may be an extension of the FDA’s Sept. 27, 2019, target action date on the lumateperone application.
Investors viewed this as bad news. Shares of Intra-Cellular on July 23 dropped from an opening price of $11.90 to a closing one of $8.19. On July 29, they closed at $8.12.
Still, it is unclear how the FDA will decide on the lumateperone application and whether the agency will call another advisory committee meeting on it.
Last year, the FDA accepted the application for lumateperone, a once-daily treatment, Intra-Cellular said. The agency had in 2017 given a fast-track designation to lumateperone for the treatment of schizophrenia.
Lumateperone is the lead product for the company.
On the company’s website, Intra-Cellular says three large randomized, double-blind, placebo-controlled trials have been done for lumateperone as a schizophrenia drug. In two of these studies, results for lumateperone at a 60-mg dose showed a “statistically significant separation from placebo on the primary endpoint, the Positive and Negative Syndrome Scale or PANSS total score.”
In a recent routine filing with the Securities and Exchange Commission, Intra-Cellular said it was having an “ongoing dialogue” with the FDA about lumateperone. The company in 2016 had announced that, in a phase 3 study known as ITI-007-302, lumateperone had not separated from placebo on the primary endpoint, change from baseline on the PANSS total score, in the predefined patient population. The active control for ITI-007-302, risperidone, did separate from placebo.
In the recent SEC filing, Intra-Cellular said the FDA already has confirmed that the results of ITI-007-302 did not preclude the submission of a new drug application.
Intra-Cellular also said “lumateperone was statistically significantly better than risperidone on key safety and tolerability parameters, and exhibited a safety profile similar to placebo” in the 302 study. Lumateperone’s failure to best placebo in the 302 test was “in part due to an unusually high placebo response at certain sites.”
LAIs still underused for patients with psychosis
Long-acting injectables (LAIs) continue to be underused for patients with chronic diseases such as schizophrenia and bipolar disorder. However, in my practice, I have found the use of those medications to be useful for promoting adherence, and I wonder why they are not used more often – in light of their effectiveness. Specifically, among individuals with schizophrenia, LAIs can lead to significant improvements in symptom control, quality of life, and overall functioning.1
The following three cases illustrate the power of LAIs:
Case 1: A male patient with schizoaffective diagnosis had been admitted several times to the inpatient psychiatric unit and had poor compliance to medications by mouth. He had multiple emergency department visits besides having community health behavioral support. After various medication trials by mouth, he responded to LAIs. He was able to function in the community for longer periods of time and required far fewer ED visits. He followed up with his outpatient psychiatric support regularly.
Case 2: A female patient with schizoaffective disorder had psychosis of a persecutory nature and paranoia. She was unable to function in the community and struggled with delusional thoughts leading to anger outbursts in the community. She continually refused medicines by mouth in the outpatient unit. Upon involuntary inpatient management as per court order, the patient responded to LAIs. Her insight improved, and she displayed better judgment in the future.
Case 3: A female patient with bipolar I was impulsive and promiscuous, and routinely entered into high-risk situations. She was not able to negotiate safely in the community, and was shuttling from shelter to shelter. She was losing her medications time and again during her transition in the community. She responded well to LAIs, however, and was able to keep herself out of the inpatient hospital for longer periods of time. She said she felt relieved about not depending on daily oral medications. She also reported not self medicating with street substances.
A recent retrospective study of more than 3,600 patients showed that those who initiate LAIs versus oral antipsychotics have greater reductions in the number of hospitalizations.2 Furthermore, treatment with and comorbid substance use disorders.3,4
Introduction of LAI intervention within the treatment plan also might provide additional benefits and potentially reduce the burden on health care resources.5 Psychiatrists seem to use LAIs conservatively and tend to be too slow to introduce this intervention even after patients experience several acute episodes. Psychiatrists should inform patients about different forms of treatment, including LAIs, during the early stages of the illness.6
Improving medication adherence in physical and mental health care is of paramount importance for the effective care of patients. Psychiatrists and primary care physicians should be made aware of the anticipated benefits of this intervention.
References
1. Kaplan G et al. Impact of long-acting injectable antipsychotics on medication adherence and clinical, functional, and economic outcomes of schizophrenia. Patient Prefer Adherence. 2013;13:1171-80.
2. Brissos S et al. The role of long-acting injectable antipsychotics in schizophrenia: a critical appraisal. Therapeutic advances in psychopharmacology. 2014 Oct;4(5):198-219.
3. Ravasio R et al. Analisi di costo-efficacia dello switch da un antipsicotico orale a risperidone a rilascio prolungato nel trattamento dei pazienti affetti da schizofrenia. Giorn Ital Health Technol Ass. 2019;2:1-8.
4. Reichhart T and W Kissling. Societal costs of nonadherence in schizophrenia: homicide/suicide. Mind & Brain, J Psychiatry. 2010 Aug 1(2):29-32.
5. Offord S et al. Health care resource usage of schizophrenia patients initiating long-acting injectable antipsychotics vs oral. J Med Econ. 2013;16:231-9.
6. Matthias J and W Rossler. Attitudes toward long-acting depot antipsychotics: a survey of patients, relatives and psychiatrists. Psychiatry Res. 2010 Jan 30;175(1-2):58-62.
Dr. Lamba, a psychiatrist and medical director at BayRidge Hospital in Lynn, Mass., has no disclosures. He changed key facts about the patients discussed to protect their confidentiality.
Long-acting injectables (LAIs) continue to be underused for patients with chronic diseases such as schizophrenia and bipolar disorder. However, in my practice, I have found the use of those medications to be useful for promoting adherence, and I wonder why they are not used more often – in light of their effectiveness. Specifically, among individuals with schizophrenia, LAIs can lead to significant improvements in symptom control, quality of life, and overall functioning.1
The following three cases illustrate the power of LAIs:
Case 1: A male patient with schizoaffective diagnosis had been admitted several times to the inpatient psychiatric unit and had poor compliance to medications by mouth. He had multiple emergency department visits besides having community health behavioral support. After various medication trials by mouth, he responded to LAIs. He was able to function in the community for longer periods of time and required far fewer ED visits. He followed up with his outpatient psychiatric support regularly.
Case 2: A female patient with schizoaffective disorder had psychosis of a persecutory nature and paranoia. She was unable to function in the community and struggled with delusional thoughts leading to anger outbursts in the community. She continually refused medicines by mouth in the outpatient unit. Upon involuntary inpatient management as per court order, the patient responded to LAIs. Her insight improved, and she displayed better judgment in the future.
Case 3: A female patient with bipolar I was impulsive and promiscuous, and routinely entered into high-risk situations. She was not able to negotiate safely in the community, and was shuttling from shelter to shelter. She was losing her medications time and again during her transition in the community. She responded well to LAIs, however, and was able to keep herself out of the inpatient hospital for longer periods of time. She said she felt relieved about not depending on daily oral medications. She also reported not self medicating with street substances.
A recent retrospective study of more than 3,600 patients showed that those who initiate LAIs versus oral antipsychotics have greater reductions in the number of hospitalizations.2 Furthermore, treatment with and comorbid substance use disorders.3,4
Introduction of LAI intervention within the treatment plan also might provide additional benefits and potentially reduce the burden on health care resources.5 Psychiatrists seem to use LAIs conservatively and tend to be too slow to introduce this intervention even after patients experience several acute episodes. Psychiatrists should inform patients about different forms of treatment, including LAIs, during the early stages of the illness.6
Improving medication adherence in physical and mental health care is of paramount importance for the effective care of patients. Psychiatrists and primary care physicians should be made aware of the anticipated benefits of this intervention.
References
1. Kaplan G et al. Impact of long-acting injectable antipsychotics on medication adherence and clinical, functional, and economic outcomes of schizophrenia. Patient Prefer Adherence. 2013;13:1171-80.
2. Brissos S et al. The role of long-acting injectable antipsychotics in schizophrenia: a critical appraisal. Therapeutic advances in psychopharmacology. 2014 Oct;4(5):198-219.
3. Ravasio R et al. Analisi di costo-efficacia dello switch da un antipsicotico orale a risperidone a rilascio prolungato nel trattamento dei pazienti affetti da schizofrenia. Giorn Ital Health Technol Ass. 2019;2:1-8.
4. Reichhart T and W Kissling. Societal costs of nonadherence in schizophrenia: homicide/suicide. Mind & Brain, J Psychiatry. 2010 Aug 1(2):29-32.
5. Offord S et al. Health care resource usage of schizophrenia patients initiating long-acting injectable antipsychotics vs oral. J Med Econ. 2013;16:231-9.
6. Matthias J and W Rossler. Attitudes toward long-acting depot antipsychotics: a survey of patients, relatives and psychiatrists. Psychiatry Res. 2010 Jan 30;175(1-2):58-62.
Dr. Lamba, a psychiatrist and medical director at BayRidge Hospital in Lynn, Mass., has no disclosures. He changed key facts about the patients discussed to protect their confidentiality.
Long-acting injectables (LAIs) continue to be underused for patients with chronic diseases such as schizophrenia and bipolar disorder. However, in my practice, I have found the use of those medications to be useful for promoting adherence, and I wonder why they are not used more often – in light of their effectiveness. Specifically, among individuals with schizophrenia, LAIs can lead to significant improvements in symptom control, quality of life, and overall functioning.1
The following three cases illustrate the power of LAIs:
Case 1: A male patient with schizoaffective diagnosis had been admitted several times to the inpatient psychiatric unit and had poor compliance to medications by mouth. He had multiple emergency department visits besides having community health behavioral support. After various medication trials by mouth, he responded to LAIs. He was able to function in the community for longer periods of time and required far fewer ED visits. He followed up with his outpatient psychiatric support regularly.
Case 2: A female patient with schizoaffective disorder had psychosis of a persecutory nature and paranoia. She was unable to function in the community and struggled with delusional thoughts leading to anger outbursts in the community. She continually refused medicines by mouth in the outpatient unit. Upon involuntary inpatient management as per court order, the patient responded to LAIs. Her insight improved, and she displayed better judgment in the future.
Case 3: A female patient with bipolar I was impulsive and promiscuous, and routinely entered into high-risk situations. She was not able to negotiate safely in the community, and was shuttling from shelter to shelter. She was losing her medications time and again during her transition in the community. She responded well to LAIs, however, and was able to keep herself out of the inpatient hospital for longer periods of time. She said she felt relieved about not depending on daily oral medications. She also reported not self medicating with street substances.
A recent retrospective study of more than 3,600 patients showed that those who initiate LAIs versus oral antipsychotics have greater reductions in the number of hospitalizations.2 Furthermore, treatment with and comorbid substance use disorders.3,4
Introduction of LAI intervention within the treatment plan also might provide additional benefits and potentially reduce the burden on health care resources.5 Psychiatrists seem to use LAIs conservatively and tend to be too slow to introduce this intervention even after patients experience several acute episodes. Psychiatrists should inform patients about different forms of treatment, including LAIs, during the early stages of the illness.6
Improving medication adherence in physical and mental health care is of paramount importance for the effective care of patients. Psychiatrists and primary care physicians should be made aware of the anticipated benefits of this intervention.
References
1. Kaplan G et al. Impact of long-acting injectable antipsychotics on medication adherence and clinical, functional, and economic outcomes of schizophrenia. Patient Prefer Adherence. 2013;13:1171-80.
2. Brissos S et al. The role of long-acting injectable antipsychotics in schizophrenia: a critical appraisal. Therapeutic advances in psychopharmacology. 2014 Oct;4(5):198-219.
3. Ravasio R et al. Analisi di costo-efficacia dello switch da un antipsicotico orale a risperidone a rilascio prolungato nel trattamento dei pazienti affetti da schizofrenia. Giorn Ital Health Technol Ass. 2019;2:1-8.
4. Reichhart T and W Kissling. Societal costs of nonadherence in schizophrenia: homicide/suicide. Mind & Brain, J Psychiatry. 2010 Aug 1(2):29-32.
5. Offord S et al. Health care resource usage of schizophrenia patients initiating long-acting injectable antipsychotics vs oral. J Med Econ. 2013;16:231-9.
6. Matthias J and W Rossler. Attitudes toward long-acting depot antipsychotics: a survey of patients, relatives and psychiatrists. Psychiatry Res. 2010 Jan 30;175(1-2):58-62.
Dr. Lamba, a psychiatrist and medical director at BayRidge Hospital in Lynn, Mass., has no disclosures. He changed key facts about the patients discussed to protect their confidentiality.
The jealous insomniac
CASE Anxious and jealous
Mrs. H, age 28, presents to the emergency department (ED) with pressured speech, emotional lability, loose associations, and echolalia. On physical examination, Mrs. H is noted to have hand tremors. Mrs. H says she has not slept for the past 5 days and is experiencing anxiety and heart palpitations.
She also says that for the past 2 years she has believed that her husband is having an affair with her best friend. However, her current presentation—which she attributes to the alleged affair—began a week before she came to the ED. According to her husband, Mrs. H was “perfectly fine until a week ago” and her symptoms “appeared out of nowhere.” He reports that this has never happened before.
Mrs. H is admitted to the psychiatry unit. The nursing team reports that on the first night, Mrs. H was “running and screaming on the unit, out of control,” and was “tearful, manicky, and dysphoric.”
Mrs. H has no significant medical or psychiatric history. Her family history is significant for hyperthyroidism in her mother and maternal grandmother. Mrs. H says she smokes cigarettes (1 pack/d) but denies alcohol or illicit drug use.
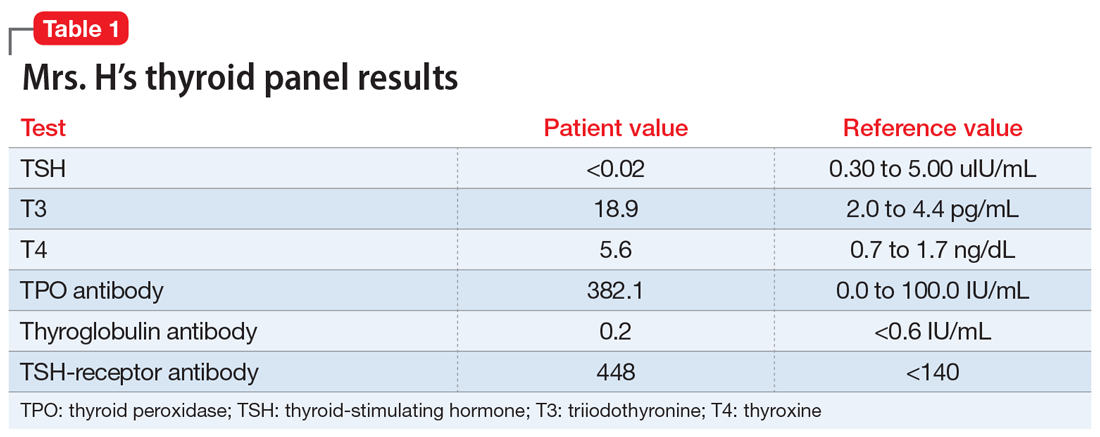
EVALUATION A telling thyroid panel
Mrs. H undergoes laboratory testing, including a complete blood count, comprehensive metabolic panel, and thyroid panel due to her family history of thyroid-related disorders. The thyroid panel shows the presence of the thyroid-stimulating hormone (TSH) receptor antibody; a low TSH level; elevated triiodothyronine (T3) and thyroxine (T4) levels, with T3 > T4; elevated thyroid peroxidase (TPO) antibody; and elevated thyroglobulin antibody (Table 1). A scan shows the thyroid gland to be normal/top-normal size and is read by radiology to be indicative of a resolving thyroiditis vs Graves’ disease. An electrocardiogram indicates a heart rate of 139 beats per minute.
[polldaddy:10352133]
The authors’ observations
Mrs. H fits the presentation of psychosis secondary to Graves’ disease. However, our differential consisted of thyroiditis, brief psychotic disorder, delusional disorder (jealous type), and bipolar mania.
Brief psychotic disorder, bipolar mania, and delusional disorder were better explained by Graves’ disease, and Mrs. H’s jealous delusion resulted in functional impairment, which eliminated delusional disorder. Her family history of hyperthyroidism, as well as her sex and history of tobacco use, supported the diagnosis of Graves’ disease. Although Mrs. H did not experience goiter, ophthalmopathy, or dermopathy, which are common signs and symptoms of Graves’ disease (Table 2), she did present with irritability, insomnia, tachycardia, and a hand tremor. Her psychiatric symptoms included anxiety, emotional lability and, most importantly, psychosis. Her laboratory results included the presence of the TSH-receptor antibody, a low TSH level, and elevated T3 and T4 levels (T3>T4), confirming the diagnosis of early-onset Graves’ disease.
Continue to: Graves' disease
Graves’ disease
Graves’ disease is the most common cause of hyperthyroidism, representing approximately 50% to 80% of cases.1 Graves’ disease occurs most often in women, smokers, and those with a personal or family history of autoimmune disease; although patients of any age may be affected, the peak incidence occurs between age 40 and 60.1
Graves’ disease results from the production of immunoglobulin G (IgG) antibodies that activate the TSH receptor on the surface of thyroid follicular cells.1 The presence of the TSH-receptor antibody, in addition to a low TSH and elevated T3 and T4 levels (T3>T4), are common laboratory findings in patients with this disease. A thyroid scan will also show increased radiotracer accumulation.
Patients with Graves’ disease, as well as those with hyperthyroidism, tend to report weight loss, increased appetite, heat intolerance, irritability, insomnia, and palpitations. In addition to the above symptoms, the identifying signs and symptoms of Graves’ disease include a goiter, ophthalmopathy, and dermopathy (Table 2). Rarely, patients with Graves’ disease can present with psychosis, which is often complicated by thyrotoxicosis.2
[polldaddy:10352135]
TREATMENT Antipsychotic and a beta blocker
Based on her signs, symptoms, and laboratory findings, Mrs. H receives risperidone, 1 mg twice daily, for psychosis, and atenolol, 25 mg twice daily, for heart palpitations. Over 4 days, her symptoms decrease; she experiences more linear thought and decreased flight-of-ideas, and becomes unsure about the truth of her husband’s alleged affair. Her impulsive behaviors and severe mood lability cease. Her tachycardia remains controlled with atenolol.
The authors’ observations
Rapid initiation of treatment is important when managing patients with Graves’ disease, because untreated patients have a higher risk of psychiatric illness, cardiac disease, arrhythmia, and sudden cardiac death.1 Patients with Graves’ disease typically are treated with thionamides, radioactive iodine, and/or surgery. When a patient presents with psychosis as a result of thyrotoxicosis, treatment focuses on improving the thyrotoxicosis through anti-thyroid medications and beta blockers (Table 33). Psychotropic medications, such as antipsychotics, are not indicated for primary treatment, but are given to patients who have severe psychosis until symptoms have resolved.3 For Mrs. H, the severity of her psychosis necessitated risperidone in addition to atenolol.
OUTCOME Continuous medical management; no ablation
Mrs. H is discharged with immediate outpatient follow-up with an endocrinology team to discuss the best long-term management of her thyroiditis. Mrs. H opts for continuous medical management (as opposed to ablation) and is administered methimazole, 15 mg/d, to treat Graves’ disease.
The authors’ observations
This case provides useful information regarding recognizing psychosis as the initial sign of Graves’ disease. Although Graves’ disease represents 50% to 80% of cases of hyperthyroidism,1 psychosis as the first clinical presentation of this disease is extremely rare. Several case reports, however, have described this phenomenon,2,3 and further studies would be helpful to determine its true prevalence.
Continue to: Bottom Line
Bottom Line
Although extremely rare, psychosis as the initial clinical presentation of Graves’ disease can occur. The early diagnosis of Graves’ disease is critical to prevent cardiovascular implications and death.
Related Resources
- Abraham P, Acharya S. Current and emerging treatment options for Graves’ hyperthyroidism. Ther Clin Risk Manag. 2010;6:29-40.
- Bunevicius R, Prange AJ Jr. Psychiatric manifestations of Graves’ hyperthyroidism: pathophysiology and treatment options. CNS Drugs. 2006;20(11):897-909.
- Ginsberg J. Diagnosis and management of Graves’ disease. CMAJ. 2003;168(5):575-585.
Drug Brand Names
Atenolol • Tenormin
Methimazole • Tapazole
Risperidone • Risperdal
1. Girgis C, Champion B, Wall J. Current concepts in Graves’ disease. Ther Adv Endocrinol Metab. 2011;2(3):135-144.
2. Urias-Uribe L, Valdez-Solis E, González-Milán C, et al. Psychosis crisis associated with thyrotoxicosis due to Graves’ disease. Case Rep Psychiatry. 2017;2017:6803682. doi: 10.1155/2017/6803682.
3. Ugwu ET, Maluze J, Onyebueke GC. Graves’ thyrotoxicosis presenting as schizophreniform psychosis: a case report and literature review. Int J Endocrinol Metab. 2017;15(1):e41977. doi: 10.5812/ijem.41977.
CASE Anxious and jealous
Mrs. H, age 28, presents to the emergency department (ED) with pressured speech, emotional lability, loose associations, and echolalia. On physical examination, Mrs. H is noted to have hand tremors. Mrs. H says she has not slept for the past 5 days and is experiencing anxiety and heart palpitations.
She also says that for the past 2 years she has believed that her husband is having an affair with her best friend. However, her current presentation—which she attributes to the alleged affair—began a week before she came to the ED. According to her husband, Mrs. H was “perfectly fine until a week ago” and her symptoms “appeared out of nowhere.” He reports that this has never happened before.
Mrs. H is admitted to the psychiatry unit. The nursing team reports that on the first night, Mrs. H was “running and screaming on the unit, out of control,” and was “tearful, manicky, and dysphoric.”
Mrs. H has no significant medical or psychiatric history. Her family history is significant for hyperthyroidism in her mother and maternal grandmother. Mrs. H says she smokes cigarettes (1 pack/d) but denies alcohol or illicit drug use.

EVALUATION A telling thyroid panel
Mrs. H undergoes laboratory testing, including a complete blood count, comprehensive metabolic panel, and thyroid panel due to her family history of thyroid-related disorders. The thyroid panel shows the presence of the thyroid-stimulating hormone (TSH) receptor antibody; a low TSH level; elevated triiodothyronine (T3) and thyroxine (T4) levels, with T3 > T4; elevated thyroid peroxidase (TPO) antibody; and elevated thyroglobulin antibody (Table 1). A scan shows the thyroid gland to be normal/top-normal size and is read by radiology to be indicative of a resolving thyroiditis vs Graves’ disease. An electrocardiogram indicates a heart rate of 139 beats per minute.
[polldaddy:10352133]
The authors’ observations
Mrs. H fits the presentation of psychosis secondary to Graves’ disease. However, our differential consisted of thyroiditis, brief psychotic disorder, delusional disorder (jealous type), and bipolar mania.
Brief psychotic disorder, bipolar mania, and delusional disorder were better explained by Graves’ disease, and Mrs. H’s jealous delusion resulted in functional impairment, which eliminated delusional disorder. Her family history of hyperthyroidism, as well as her sex and history of tobacco use, supported the diagnosis of Graves’ disease. Although Mrs. H did not experience goiter, ophthalmopathy, or dermopathy, which are common signs and symptoms of Graves’ disease (Table 2), she did present with irritability, insomnia, tachycardia, and a hand tremor. Her psychiatric symptoms included anxiety, emotional lability and, most importantly, psychosis. Her laboratory results included the presence of the TSH-receptor antibody, a low TSH level, and elevated T3 and T4 levels (T3>T4), confirming the diagnosis of early-onset Graves’ disease.
Continue to: Graves' disease
Graves’ disease
Graves’ disease is the most common cause of hyperthyroidism, representing approximately 50% to 80% of cases.1 Graves’ disease occurs most often in women, smokers, and those with a personal or family history of autoimmune disease; although patients of any age may be affected, the peak incidence occurs between age 40 and 60.1
Graves’ disease results from the production of immunoglobulin G (IgG) antibodies that activate the TSH receptor on the surface of thyroid follicular cells.1 The presence of the TSH-receptor antibody, in addition to a low TSH and elevated T3 and T4 levels (T3>T4), are common laboratory findings in patients with this disease. A thyroid scan will also show increased radiotracer accumulation.
Patients with Graves’ disease, as well as those with hyperthyroidism, tend to report weight loss, increased appetite, heat intolerance, irritability, insomnia, and palpitations. In addition to the above symptoms, the identifying signs and symptoms of Graves’ disease include a goiter, ophthalmopathy, and dermopathy (Table 2). Rarely, patients with Graves’ disease can present with psychosis, which is often complicated by thyrotoxicosis.2
[polldaddy:10352135]
TREATMENT Antipsychotic and a beta blocker
Based on her signs, symptoms, and laboratory findings, Mrs. H receives risperidone, 1 mg twice daily, for psychosis, and atenolol, 25 mg twice daily, for heart palpitations. Over 4 days, her symptoms decrease; she experiences more linear thought and decreased flight-of-ideas, and becomes unsure about the truth of her husband’s alleged affair. Her impulsive behaviors and severe mood lability cease. Her tachycardia remains controlled with atenolol.
The authors’ observations
Rapid initiation of treatment is important when managing patients with Graves’ disease, because untreated patients have a higher risk of psychiatric illness, cardiac disease, arrhythmia, and sudden cardiac death.1 Patients with Graves’ disease typically are treated with thionamides, radioactive iodine, and/or surgery. When a patient presents with psychosis as a result of thyrotoxicosis, treatment focuses on improving the thyrotoxicosis through anti-thyroid medications and beta blockers (Table 33). Psychotropic medications, such as antipsychotics, are not indicated for primary treatment, but are given to patients who have severe psychosis until symptoms have resolved.3 For Mrs. H, the severity of her psychosis necessitated risperidone in addition to atenolol.
OUTCOME Continuous medical management; no ablation
Mrs. H is discharged with immediate outpatient follow-up with an endocrinology team to discuss the best long-term management of her thyroiditis. Mrs. H opts for continuous medical management (as opposed to ablation) and is administered methimazole, 15 mg/d, to treat Graves’ disease.
The authors’ observations
This case provides useful information regarding recognizing psychosis as the initial sign of Graves’ disease. Although Graves’ disease represents 50% to 80% of cases of hyperthyroidism,1 psychosis as the first clinical presentation of this disease is extremely rare. Several case reports, however, have described this phenomenon,2,3 and further studies would be helpful to determine its true prevalence.
Continue to: Bottom Line
Bottom Line
Although extremely rare, psychosis as the initial clinical presentation of Graves’ disease can occur. The early diagnosis of Graves’ disease is critical to prevent cardiovascular implications and death.
Related Resources
- Abraham P, Acharya S. Current and emerging treatment options for Graves’ hyperthyroidism. Ther Clin Risk Manag. 2010;6:29-40.
- Bunevicius R, Prange AJ Jr. Psychiatric manifestations of Graves’ hyperthyroidism: pathophysiology and treatment options. CNS Drugs. 2006;20(11):897-909.
- Ginsberg J. Diagnosis and management of Graves’ disease. CMAJ. 2003;168(5):575-585.
Drug Brand Names
Atenolol • Tenormin
Methimazole • Tapazole
Risperidone • Risperdal
CASE Anxious and jealous
Mrs. H, age 28, presents to the emergency department (ED) with pressured speech, emotional lability, loose associations, and echolalia. On physical examination, Mrs. H is noted to have hand tremors. Mrs. H says she has not slept for the past 5 days and is experiencing anxiety and heart palpitations.
She also says that for the past 2 years she has believed that her husband is having an affair with her best friend. However, her current presentation—which she attributes to the alleged affair—began a week before she came to the ED. According to her husband, Mrs. H was “perfectly fine until a week ago” and her symptoms “appeared out of nowhere.” He reports that this has never happened before.
Mrs. H is admitted to the psychiatry unit. The nursing team reports that on the first night, Mrs. H was “running and screaming on the unit, out of control,” and was “tearful, manicky, and dysphoric.”
Mrs. H has no significant medical or psychiatric history. Her family history is significant for hyperthyroidism in her mother and maternal grandmother. Mrs. H says she smokes cigarettes (1 pack/d) but denies alcohol or illicit drug use.

EVALUATION A telling thyroid panel
Mrs. H undergoes laboratory testing, including a complete blood count, comprehensive metabolic panel, and thyroid panel due to her family history of thyroid-related disorders. The thyroid panel shows the presence of the thyroid-stimulating hormone (TSH) receptor antibody; a low TSH level; elevated triiodothyronine (T3) and thyroxine (T4) levels, with T3 > T4; elevated thyroid peroxidase (TPO) antibody; and elevated thyroglobulin antibody (Table 1). A scan shows the thyroid gland to be normal/top-normal size and is read by radiology to be indicative of a resolving thyroiditis vs Graves’ disease. An electrocardiogram indicates a heart rate of 139 beats per minute.
[polldaddy:10352133]
The authors’ observations
Mrs. H fits the presentation of psychosis secondary to Graves’ disease. However, our differential consisted of thyroiditis, brief psychotic disorder, delusional disorder (jealous type), and bipolar mania.
Brief psychotic disorder, bipolar mania, and delusional disorder were better explained by Graves’ disease, and Mrs. H’s jealous delusion resulted in functional impairment, which eliminated delusional disorder. Her family history of hyperthyroidism, as well as her sex and history of tobacco use, supported the diagnosis of Graves’ disease. Although Mrs. H did not experience goiter, ophthalmopathy, or dermopathy, which are common signs and symptoms of Graves’ disease (Table 2), she did present with irritability, insomnia, tachycardia, and a hand tremor. Her psychiatric symptoms included anxiety, emotional lability and, most importantly, psychosis. Her laboratory results included the presence of the TSH-receptor antibody, a low TSH level, and elevated T3 and T4 levels (T3>T4), confirming the diagnosis of early-onset Graves’ disease.
Continue to: Graves' disease
Graves’ disease
Graves’ disease is the most common cause of hyperthyroidism, representing approximately 50% to 80% of cases.1 Graves’ disease occurs most often in women, smokers, and those with a personal or family history of autoimmune disease; although patients of any age may be affected, the peak incidence occurs between age 40 and 60.1
Graves’ disease results from the production of immunoglobulin G (IgG) antibodies that activate the TSH receptor on the surface of thyroid follicular cells.1 The presence of the TSH-receptor antibody, in addition to a low TSH and elevated T3 and T4 levels (T3>T4), are common laboratory findings in patients with this disease. A thyroid scan will also show increased radiotracer accumulation.
Patients with Graves’ disease, as well as those with hyperthyroidism, tend to report weight loss, increased appetite, heat intolerance, irritability, insomnia, and palpitations. In addition to the above symptoms, the identifying signs and symptoms of Graves’ disease include a goiter, ophthalmopathy, and dermopathy (Table 2). Rarely, patients with Graves’ disease can present with psychosis, which is often complicated by thyrotoxicosis.2
[polldaddy:10352135]
TREATMENT Antipsychotic and a beta blocker
Based on her signs, symptoms, and laboratory findings, Mrs. H receives risperidone, 1 mg twice daily, for psychosis, and atenolol, 25 mg twice daily, for heart palpitations. Over 4 days, her symptoms decrease; she experiences more linear thought and decreased flight-of-ideas, and becomes unsure about the truth of her husband’s alleged affair. Her impulsive behaviors and severe mood lability cease. Her tachycardia remains controlled with atenolol.
The authors’ observations
Rapid initiation of treatment is important when managing patients with Graves’ disease, because untreated patients have a higher risk of psychiatric illness, cardiac disease, arrhythmia, and sudden cardiac death.1 Patients with Graves’ disease typically are treated with thionamides, radioactive iodine, and/or surgery. When a patient presents with psychosis as a result of thyrotoxicosis, treatment focuses on improving the thyrotoxicosis through anti-thyroid medications and beta blockers (Table 33). Psychotropic medications, such as antipsychotics, are not indicated for primary treatment, but are given to patients who have severe psychosis until symptoms have resolved.3 For Mrs. H, the severity of her psychosis necessitated risperidone in addition to atenolol.
OUTCOME Continuous medical management; no ablation
Mrs. H is discharged with immediate outpatient follow-up with an endocrinology team to discuss the best long-term management of her thyroiditis. Mrs. H opts for continuous medical management (as opposed to ablation) and is administered methimazole, 15 mg/d, to treat Graves’ disease.
The authors’ observations
This case provides useful information regarding recognizing psychosis as the initial sign of Graves’ disease. Although Graves’ disease represents 50% to 80% of cases of hyperthyroidism,1 psychosis as the first clinical presentation of this disease is extremely rare. Several case reports, however, have described this phenomenon,2,3 and further studies would be helpful to determine its true prevalence.
Continue to: Bottom Line
Bottom Line
Although extremely rare, psychosis as the initial clinical presentation of Graves’ disease can occur. The early diagnosis of Graves’ disease is critical to prevent cardiovascular implications and death.
Related Resources
- Abraham P, Acharya S. Current and emerging treatment options for Graves’ hyperthyroidism. Ther Clin Risk Manag. 2010;6:29-40.
- Bunevicius R, Prange AJ Jr. Psychiatric manifestations of Graves’ hyperthyroidism: pathophysiology and treatment options. CNS Drugs. 2006;20(11):897-909.
- Ginsberg J. Diagnosis and management of Graves’ disease. CMAJ. 2003;168(5):575-585.
Drug Brand Names
Atenolol • Tenormin
Methimazole • Tapazole
Risperidone • Risperdal
1. Girgis C, Champion B, Wall J. Current concepts in Graves’ disease. Ther Adv Endocrinol Metab. 2011;2(3):135-144.
2. Urias-Uribe L, Valdez-Solis E, González-Milán C, et al. Psychosis crisis associated with thyrotoxicosis due to Graves’ disease. Case Rep Psychiatry. 2017;2017:6803682. doi: 10.1155/2017/6803682.
3. Ugwu ET, Maluze J, Onyebueke GC. Graves’ thyrotoxicosis presenting as schizophreniform psychosis: a case report and literature review. Int J Endocrinol Metab. 2017;15(1):e41977. doi: 10.5812/ijem.41977.
1. Girgis C, Champion B, Wall J. Current concepts in Graves’ disease. Ther Adv Endocrinol Metab. 2011;2(3):135-144.
2. Urias-Uribe L, Valdez-Solis E, González-Milán C, et al. Psychosis crisis associated with thyrotoxicosis due to Graves’ disease. Case Rep Psychiatry. 2017;2017:6803682. doi: 10.1155/2017/6803682.
3. Ugwu ET, Maluze J, Onyebueke GC. Graves’ thyrotoxicosis presenting as schizophreniform psychosis: a case report and literature review. Int J Endocrinol Metab. 2017;15(1):e41977. doi: 10.5812/ijem.41977.
Psychosis as a common thread across psychiatric disorders
Ask a psychiatrist to name a psychotic disorder, and the answer will most likely be “schizophrenia.” But if you closely examine the symptom structure of DSM-5 psychiatric disorders, you will note the presence of psychosis in almost all of them.
Fixed false beliefs and impaired reality testing are core features of psychosis. Those are certainly prominent in severe psychoses such as schizophrenia, schizoaffective disorder, or delusional disorder. But psychosis is actually a continuum of varying severity across most psychiatric disorders, although they carry different diagnostic labels. Irrational false beliefs and impaired functioning due to poor reality testing are embedded among many DSM-5 disorders. Hallucinations are less common; they are perceptual aberrations, not thought abnormalities, although they can trigger delusional explanations as to their causation.
Consider the following:
- Bipolar disorder. A large proportion of patients with bipolar disorder manifest delusions, usually grandiose, but often paranoid or referential.
- Major depressive disorder (MDD). Although regarded as a “pure mood disorder,” the core symptoms of MDD—self-deprecation and sense of worthlessness—as well as the poor reality testing of suicidal thoughts (that death is a better option than living) are psychotic false beliefs.
- Anxiety and panic disorder. The central symptom in anxiety and panic attacks is a belief in impending doom and/or death. The fear in anxiety disorders is actually based on a false belief (eg, if I get on the plane, it will crash, and I will die). Thus, technically an irrational/psychotic thought process underpins the terror and fear of anxiety disorders.
- Borderline personality disorder. Frank psychotic symptoms, such as paranoid beliefs, are known to be a component of borderline personality disorder symptoms. Although these symptoms tend to be brief and episodic, they can have a deleterious effect on the person’s coping and relationships.
- Other personality disorders. While many individuals with narcissistic personality disorder are functional, their exaggerated sense of self-importance, entitlement, and self-aggrandizement certainly qualifies as a fixed false belief. Patients with other personality disorders, such as schizotypal and paranoid, are known to harbor false beliefs or magical thinking.
- Body dysmorphic disorder. False beliefs about one’s appearance (such as blemishes or asymmetry) are at the center of this disorder, and it meets the litmus test of a psychosis.
- Anorexia nervosa. This disorder is well known to be characterized by a fixed false belief that one is “fat,” even when the patient’s body borders on being cachectic in appearance according to objective observers.
- Autism. This spectrum of diseases includes false beliefs that drive the ritualistic or odd behaviors.
- Obsessive-compulsive disorder. Although obsessions are usually ego-dystonic, in severe cases, they become ego-syntonic, similar to delusions. On the other hand, compulsions are often driven by a false belief, such as believing that one’s hands are dirty and must be washed incessantly, or that the locks on the door must be rechecked repeatedly because an intruder may break into the house and harm the inhabitants.
- Neurodegenerative syndromes. Neurodegenerative syndromes are neuropsychiatric disorders that very frequently include psychotic symptoms, such as paranoid delusions, delusions of marital infidelity, Capgras syndrome, or folie à deux. These disorders include Alzheimer’s disease, Parkinson’s disease, Lewy body dementia, frontal temporal dementia, metachromatic leukodystrophy, Huntington’s chorea, temporal lobe epilepsy, stroke, xenomelia, reduplicative phenomena, etc. This reflects the common emergence of faulty thinking with disintegration of neural tissue, both gray and white matter.
Continue to: So it should not be...
So it should not be surprising that antipsychotic medications, especially second-generation agents, have been shown to be helpful as monotherapy or adjunctive therapy in practically all the above psychiatric disorders, whether on-label or off-label.
Finally, it should also be noted that a case has been made for the existence of one dimension in all mental disorders manifesting in multiple psychopathologies.1 It is possible that a continuum of delusional thinking is a common thread across many psychiatric disorders due to this putative shared dimension. The milder form of this dimension may also explain the presence of pre-psychotic thinking in a significant proportion of the general population who do not seek psychiatric help.2 Just think of how many people you befriend, socialize with, and regard as perfectly “normal” endorse wild superstitions and astrological predictions, or believe in various conspiracy theories that have no basis in reality.
To comment on this editorial or other topics of interest: henry.nasrallah@currentpsychiatry.com.
1. Caspi A, Moffitt TE. All for one and one for all: mental disorders in one dimension. Am J Psychiatry. 2018;175(9):831-844.
2. van Os J, Linscott RJ, Myin-Germeys I, et al. A systematic review and meta-analysis of the psychosis continuum: evidence for a psychosis proneness-persistence-impairment model of psychotic disorder. Psychol Med. 2009;39(2):179-195.
Ask a psychiatrist to name a psychotic disorder, and the answer will most likely be “schizophrenia.” But if you closely examine the symptom structure of DSM-5 psychiatric disorders, you will note the presence of psychosis in almost all of them.
Fixed false beliefs and impaired reality testing are core features of psychosis. Those are certainly prominent in severe psychoses such as schizophrenia, schizoaffective disorder, or delusional disorder. But psychosis is actually a continuum of varying severity across most psychiatric disorders, although they carry different diagnostic labels. Irrational false beliefs and impaired functioning due to poor reality testing are embedded among many DSM-5 disorders. Hallucinations are less common; they are perceptual aberrations, not thought abnormalities, although they can trigger delusional explanations as to their causation.
Consider the following:
- Bipolar disorder. A large proportion of patients with bipolar disorder manifest delusions, usually grandiose, but often paranoid or referential.
- Major depressive disorder (MDD). Although regarded as a “pure mood disorder,” the core symptoms of MDD—self-deprecation and sense of worthlessness—as well as the poor reality testing of suicidal thoughts (that death is a better option than living) are psychotic false beliefs.
- Anxiety and panic disorder. The central symptom in anxiety and panic attacks is a belief in impending doom and/or death. The fear in anxiety disorders is actually based on a false belief (eg, if I get on the plane, it will crash, and I will die). Thus, technically an irrational/psychotic thought process underpins the terror and fear of anxiety disorders.
- Borderline personality disorder. Frank psychotic symptoms, such as paranoid beliefs, are known to be a component of borderline personality disorder symptoms. Although these symptoms tend to be brief and episodic, they can have a deleterious effect on the person’s coping and relationships.
- Other personality disorders. While many individuals with narcissistic personality disorder are functional, their exaggerated sense of self-importance, entitlement, and self-aggrandizement certainly qualifies as a fixed false belief. Patients with other personality disorders, such as schizotypal and paranoid, are known to harbor false beliefs or magical thinking.
- Body dysmorphic disorder. False beliefs about one’s appearance (such as blemishes or asymmetry) are at the center of this disorder, and it meets the litmus test of a psychosis.
- Anorexia nervosa. This disorder is well known to be characterized by a fixed false belief that one is “fat,” even when the patient’s body borders on being cachectic in appearance according to objective observers.
- Autism. This spectrum of diseases includes false beliefs that drive the ritualistic or odd behaviors.
- Obsessive-compulsive disorder. Although obsessions are usually ego-dystonic, in severe cases, they become ego-syntonic, similar to delusions. On the other hand, compulsions are often driven by a false belief, such as believing that one’s hands are dirty and must be washed incessantly, or that the locks on the door must be rechecked repeatedly because an intruder may break into the house and harm the inhabitants.
- Neurodegenerative syndromes. Neurodegenerative syndromes are neuropsychiatric disorders that very frequently include psychotic symptoms, such as paranoid delusions, delusions of marital infidelity, Capgras syndrome, or folie à deux. These disorders include Alzheimer’s disease, Parkinson’s disease, Lewy body dementia, frontal temporal dementia, metachromatic leukodystrophy, Huntington’s chorea, temporal lobe epilepsy, stroke, xenomelia, reduplicative phenomena, etc. This reflects the common emergence of faulty thinking with disintegration of neural tissue, both gray and white matter.
Continue to: So it should not be...
So it should not be surprising that antipsychotic medications, especially second-generation agents, have been shown to be helpful as monotherapy or adjunctive therapy in practically all the above psychiatric disorders, whether on-label or off-label.
Finally, it should also be noted that a case has been made for the existence of one dimension in all mental disorders manifesting in multiple psychopathologies.1 It is possible that a continuum of delusional thinking is a common thread across many psychiatric disorders due to this putative shared dimension. The milder form of this dimension may also explain the presence of pre-psychotic thinking in a significant proportion of the general population who do not seek psychiatric help.2 Just think of how many people you befriend, socialize with, and regard as perfectly “normal” endorse wild superstitions and astrological predictions, or believe in various conspiracy theories that have no basis in reality.
To comment on this editorial or other topics of interest: henry.nasrallah@currentpsychiatry.com.
Ask a psychiatrist to name a psychotic disorder, and the answer will most likely be “schizophrenia.” But if you closely examine the symptom structure of DSM-5 psychiatric disorders, you will note the presence of psychosis in almost all of them.
Fixed false beliefs and impaired reality testing are core features of psychosis. Those are certainly prominent in severe psychoses such as schizophrenia, schizoaffective disorder, or delusional disorder. But psychosis is actually a continuum of varying severity across most psychiatric disorders, although they carry different diagnostic labels. Irrational false beliefs and impaired functioning due to poor reality testing are embedded among many DSM-5 disorders. Hallucinations are less common; they are perceptual aberrations, not thought abnormalities, although they can trigger delusional explanations as to their causation.
Consider the following:
- Bipolar disorder. A large proportion of patients with bipolar disorder manifest delusions, usually grandiose, but often paranoid or referential.
- Major depressive disorder (MDD). Although regarded as a “pure mood disorder,” the core symptoms of MDD—self-deprecation and sense of worthlessness—as well as the poor reality testing of suicidal thoughts (that death is a better option than living) are psychotic false beliefs.
- Anxiety and panic disorder. The central symptom in anxiety and panic attacks is a belief in impending doom and/or death. The fear in anxiety disorders is actually based on a false belief (eg, if I get on the plane, it will crash, and I will die). Thus, technically an irrational/psychotic thought process underpins the terror and fear of anxiety disorders.
- Borderline personality disorder. Frank psychotic symptoms, such as paranoid beliefs, are known to be a component of borderline personality disorder symptoms. Although these symptoms tend to be brief and episodic, they can have a deleterious effect on the person’s coping and relationships.
- Other personality disorders. While many individuals with narcissistic personality disorder are functional, their exaggerated sense of self-importance, entitlement, and self-aggrandizement certainly qualifies as a fixed false belief. Patients with other personality disorders, such as schizotypal and paranoid, are known to harbor false beliefs or magical thinking.
- Body dysmorphic disorder. False beliefs about one’s appearance (such as blemishes or asymmetry) are at the center of this disorder, and it meets the litmus test of a psychosis.
- Anorexia nervosa. This disorder is well known to be characterized by a fixed false belief that one is “fat,” even when the patient’s body borders on being cachectic in appearance according to objective observers.
- Autism. This spectrum of diseases includes false beliefs that drive the ritualistic or odd behaviors.
- Obsessive-compulsive disorder. Although obsessions are usually ego-dystonic, in severe cases, they become ego-syntonic, similar to delusions. On the other hand, compulsions are often driven by a false belief, such as believing that one’s hands are dirty and must be washed incessantly, or that the locks on the door must be rechecked repeatedly because an intruder may break into the house and harm the inhabitants.
- Neurodegenerative syndromes. Neurodegenerative syndromes are neuropsychiatric disorders that very frequently include psychotic symptoms, such as paranoid delusions, delusions of marital infidelity, Capgras syndrome, or folie à deux. These disorders include Alzheimer’s disease, Parkinson’s disease, Lewy body dementia, frontal temporal dementia, metachromatic leukodystrophy, Huntington’s chorea, temporal lobe epilepsy, stroke, xenomelia, reduplicative phenomena, etc. This reflects the common emergence of faulty thinking with disintegration of neural tissue, both gray and white matter.
Continue to: So it should not be...
So it should not be surprising that antipsychotic medications, especially second-generation agents, have been shown to be helpful as monotherapy or adjunctive therapy in practically all the above psychiatric disorders, whether on-label or off-label.
Finally, it should also be noted that a case has been made for the existence of one dimension in all mental disorders manifesting in multiple psychopathologies.1 It is possible that a continuum of delusional thinking is a common thread across many psychiatric disorders due to this putative shared dimension. The milder form of this dimension may also explain the presence of pre-psychotic thinking in a significant proportion of the general population who do not seek psychiatric help.2 Just think of how many people you befriend, socialize with, and regard as perfectly “normal” endorse wild superstitions and astrological predictions, or believe in various conspiracy theories that have no basis in reality.
To comment on this editorial or other topics of interest: henry.nasrallah@currentpsychiatry.com.
1. Caspi A, Moffitt TE. All for one and one for all: mental disorders in one dimension. Am J Psychiatry. 2018;175(9):831-844.
2. van Os J, Linscott RJ, Myin-Germeys I, et al. A systematic review and meta-analysis of the psychosis continuum: evidence for a psychosis proneness-persistence-impairment model of psychotic disorder. Psychol Med. 2009;39(2):179-195.
1. Caspi A, Moffitt TE. All for one and one for all: mental disorders in one dimension. Am J Psychiatry. 2018;175(9):831-844.
2. van Os J, Linscott RJ, Myin-Germeys I, et al. A systematic review and meta-analysis of the psychosis continuum: evidence for a psychosis proneness-persistence-impairment model of psychotic disorder. Psychol Med. 2009;39(2):179-195.
Anticholinergic drugs linked to dementia in older populations
Exposures to various types of anticholinergic medications were associated with a significantly increased risk of dementia in people aged 55 years or older in a large pharmacoepidemiologic study.
“This study was designed to assess the association between cumulative anticholinergic drug use and risk of dementia in a large, representative British population,” wrote Carol A. C. Coupland, PhD, of the division of primary care at the University of Nottingham (England), and colleagues. The findings were published in JAMA Internal Medicine.
The researchers conducted a large nested case-control study that included 58,769 patients with dementia and 225,574 matched controls from the QResearch database in England. Each study participant was matched to five controls based on various characteristics, including sex, age, and calendar time, among others.
Prescription data related to 56 different drugs with strong anticholinergic properties, including antipsychotics, bladder antimuscarinics, antiepileptics, antiparkinson agents, and antidepressants were used to measure drug exposure. The study data were analyzed from 2016 to 2018.
“The primary exposure was the total standardized daily doses (TSDDs) of anticholinergic drugs prescribed in the 1 to 11 years prior to the date of diagnosis of dementia or equivalent date in matched controls,” Dr. Coupland and colleagues wrote.
After analysis, the researchers found that exposure to antipsychotics (adjusted odds ratio, 1.70), bladder antimuscarinics (aOR, 1.65), antiepileptics (aOR, 1.39), antiparkinson agents (aOR, 1.52), and anticholinergic antidepressants (aOR, 1.29) was associated with an increased risk of dementia after adjustment for confounding factors.
“Associations were stronger in [dementia] cases diagnosed before the age of 80 years,” the researchers noted.
However, antihistamine, antivertigo/antiemetic, skeletal muscle relaxant, gastrointestinal antispasmodic, antiarrhythmic, and antimuscarinic bronchodilator anticholinergic agents were not associated with any increased risk of dementia.
One key limitation of the study was the absence of medication compliance assessment, which could result in exposure misclassification. Dr. Coupland and colleagues acknowledged this could underestimate some associations with medication exposure.
The stronger risk of dementia found among people who had dementia before age 80 “indicates that anticholinergic drugs should be prescribed with caution in middle-aged and older people,” they concluded.
One question that remains from the current study is whether anticholinergic drugs are a definite modifiable risk factor for Alzheimer’s disease and related dementias, Noll L. Campbell, PharmD, of Purdue University, West Lafayette, Ind., and colleagues wrote in an editorial accompanying the study by Dr. Coupland and associates (JAMA Intern Med. 2019 Jun 24. doi: 10.1001/jamainternmed.2019.0676).
While a pharmacologic basis for this association has been proposed, causation has yet to be established by means of prospective randomized studies. The current supposition is that deprescribing anticholinergic medications has the potential to positively effect cholinergic neurotransmission in certain regions of the brain, which could lead to improved cognitive functioning, and lower the likelihood of developing Alzheimer’s disease and related dementias, they wrote in the editorial.
However, the discontinuation of some anticholinergic agents may pose other risks, such as worsening pain or depressive symptoms, in addition to increasing the utilization of acute care facilities. As a result, high-quality, well-designed, randomized trials are needed to better understand the long-term effects of deprescribing anticholinergic medications. These trials would help inform clinicians, patients, and policymakers about the risks and benefits of deprescribing interventions, Dr. Campbell and coauthors said.
The study was supported by the National Institute for Health Research and the University of Nottingham. The authors reported financial affiliations with ClinRisk Ltd. The authors of the editorial reported receiving support from the National Institute on Aging and the Agency for Healthcare Research and Quality. Dr. Campbell reported receiving personal fees from Astellas Pharma US.
SOURCE: Coupland C et al. JAMA Intern Med. 2019 Jun 24. doi: 10.1001/jamainternmed.2019.0677
Exposures to various types of anticholinergic medications were associated with a significantly increased risk of dementia in people aged 55 years or older in a large pharmacoepidemiologic study.
“This study was designed to assess the association between cumulative anticholinergic drug use and risk of dementia in a large, representative British population,” wrote Carol A. C. Coupland, PhD, of the division of primary care at the University of Nottingham (England), and colleagues. The findings were published in JAMA Internal Medicine.
The researchers conducted a large nested case-control study that included 58,769 patients with dementia and 225,574 matched controls from the QResearch database in England. Each study participant was matched to five controls based on various characteristics, including sex, age, and calendar time, among others.
Prescription data related to 56 different drugs with strong anticholinergic properties, including antipsychotics, bladder antimuscarinics, antiepileptics, antiparkinson agents, and antidepressants were used to measure drug exposure. The study data were analyzed from 2016 to 2018.
“The primary exposure was the total standardized daily doses (TSDDs) of anticholinergic drugs prescribed in the 1 to 11 years prior to the date of diagnosis of dementia or equivalent date in matched controls,” Dr. Coupland and colleagues wrote.
After analysis, the researchers found that exposure to antipsychotics (adjusted odds ratio, 1.70), bladder antimuscarinics (aOR, 1.65), antiepileptics (aOR, 1.39), antiparkinson agents (aOR, 1.52), and anticholinergic antidepressants (aOR, 1.29) was associated with an increased risk of dementia after adjustment for confounding factors.
“Associations were stronger in [dementia] cases diagnosed before the age of 80 years,” the researchers noted.
However, antihistamine, antivertigo/antiemetic, skeletal muscle relaxant, gastrointestinal antispasmodic, antiarrhythmic, and antimuscarinic bronchodilator anticholinergic agents were not associated with any increased risk of dementia.
One key limitation of the study was the absence of medication compliance assessment, which could result in exposure misclassification. Dr. Coupland and colleagues acknowledged this could underestimate some associations with medication exposure.
The stronger risk of dementia found among people who had dementia before age 80 “indicates that anticholinergic drugs should be prescribed with caution in middle-aged and older people,” they concluded.
One question that remains from the current study is whether anticholinergic drugs are a definite modifiable risk factor for Alzheimer’s disease and related dementias, Noll L. Campbell, PharmD, of Purdue University, West Lafayette, Ind., and colleagues wrote in an editorial accompanying the study by Dr. Coupland and associates (JAMA Intern Med. 2019 Jun 24. doi: 10.1001/jamainternmed.2019.0676).
While a pharmacologic basis for this association has been proposed, causation has yet to be established by means of prospective randomized studies. The current supposition is that deprescribing anticholinergic medications has the potential to positively effect cholinergic neurotransmission in certain regions of the brain, which could lead to improved cognitive functioning, and lower the likelihood of developing Alzheimer’s disease and related dementias, they wrote in the editorial.
However, the discontinuation of some anticholinergic agents may pose other risks, such as worsening pain or depressive symptoms, in addition to increasing the utilization of acute care facilities. As a result, high-quality, well-designed, randomized trials are needed to better understand the long-term effects of deprescribing anticholinergic medications. These trials would help inform clinicians, patients, and policymakers about the risks and benefits of deprescribing interventions, Dr. Campbell and coauthors said.
The study was supported by the National Institute for Health Research and the University of Nottingham. The authors reported financial affiliations with ClinRisk Ltd. The authors of the editorial reported receiving support from the National Institute on Aging and the Agency for Healthcare Research and Quality. Dr. Campbell reported receiving personal fees from Astellas Pharma US.
SOURCE: Coupland C et al. JAMA Intern Med. 2019 Jun 24. doi: 10.1001/jamainternmed.2019.0677
Exposures to various types of anticholinergic medications were associated with a significantly increased risk of dementia in people aged 55 years or older in a large pharmacoepidemiologic study.
“This study was designed to assess the association between cumulative anticholinergic drug use and risk of dementia in a large, representative British population,” wrote Carol A. C. Coupland, PhD, of the division of primary care at the University of Nottingham (England), and colleagues. The findings were published in JAMA Internal Medicine.
The researchers conducted a large nested case-control study that included 58,769 patients with dementia and 225,574 matched controls from the QResearch database in England. Each study participant was matched to five controls based on various characteristics, including sex, age, and calendar time, among others.
Prescription data related to 56 different drugs with strong anticholinergic properties, including antipsychotics, bladder antimuscarinics, antiepileptics, antiparkinson agents, and antidepressants were used to measure drug exposure. The study data were analyzed from 2016 to 2018.
“The primary exposure was the total standardized daily doses (TSDDs) of anticholinergic drugs prescribed in the 1 to 11 years prior to the date of diagnosis of dementia or equivalent date in matched controls,” Dr. Coupland and colleagues wrote.
After analysis, the researchers found that exposure to antipsychotics (adjusted odds ratio, 1.70), bladder antimuscarinics (aOR, 1.65), antiepileptics (aOR, 1.39), antiparkinson agents (aOR, 1.52), and anticholinergic antidepressants (aOR, 1.29) was associated with an increased risk of dementia after adjustment for confounding factors.
“Associations were stronger in [dementia] cases diagnosed before the age of 80 years,” the researchers noted.
However, antihistamine, antivertigo/antiemetic, skeletal muscle relaxant, gastrointestinal antispasmodic, antiarrhythmic, and antimuscarinic bronchodilator anticholinergic agents were not associated with any increased risk of dementia.
One key limitation of the study was the absence of medication compliance assessment, which could result in exposure misclassification. Dr. Coupland and colleagues acknowledged this could underestimate some associations with medication exposure.
The stronger risk of dementia found among people who had dementia before age 80 “indicates that anticholinergic drugs should be prescribed with caution in middle-aged and older people,” they concluded.
One question that remains from the current study is whether anticholinergic drugs are a definite modifiable risk factor for Alzheimer’s disease and related dementias, Noll L. Campbell, PharmD, of Purdue University, West Lafayette, Ind., and colleagues wrote in an editorial accompanying the study by Dr. Coupland and associates (JAMA Intern Med. 2019 Jun 24. doi: 10.1001/jamainternmed.2019.0676).
While a pharmacologic basis for this association has been proposed, causation has yet to be established by means of prospective randomized studies. The current supposition is that deprescribing anticholinergic medications has the potential to positively effect cholinergic neurotransmission in certain regions of the brain, which could lead to improved cognitive functioning, and lower the likelihood of developing Alzheimer’s disease and related dementias, they wrote in the editorial.
However, the discontinuation of some anticholinergic agents may pose other risks, such as worsening pain or depressive symptoms, in addition to increasing the utilization of acute care facilities. As a result, high-quality, well-designed, randomized trials are needed to better understand the long-term effects of deprescribing anticholinergic medications. These trials would help inform clinicians, patients, and policymakers about the risks and benefits of deprescribing interventions, Dr. Campbell and coauthors said.
The study was supported by the National Institute for Health Research and the University of Nottingham. The authors reported financial affiliations with ClinRisk Ltd. The authors of the editorial reported receiving support from the National Institute on Aging and the Agency for Healthcare Research and Quality. Dr. Campbell reported receiving personal fees from Astellas Pharma US.
SOURCE: Coupland C et al. JAMA Intern Med. 2019 Jun 24. doi: 10.1001/jamainternmed.2019.0677
FROM JAMA INTERNAL MEDICINE
What makes a mass murderer?
‘A Dark Night in Aurora’ gives view into one killer’s mind
It was an unthinkable crime; on July 20, 2012, a 24-year-old neuroscience graduate student* walked into a movie theatre in Aurora, Colo., and began shooting people he had never before seen. Twelve people died and 70 were wounded in this calculated and intricately planned massacre that challenged everything we know about the intersection of mental illness and evil.
So much has been written about mass shooters as we’ve struggled to understand what motivates someone to kill strangers in a public place. The hope is that if we can understand it, then we can prevent it. The topic has been one of great interest to me because it gets quickly linked to advocacy for involuntary psychiatric care: Half of mass murderers suffer from psychiatric disorders and if we could force them to get treatment, then we could prevent these horrific events.

Many mass shooters die at the scene. Not only did the Aurora shooter survive, but his case went to trial – also a rarity – providing insights we have not had with other similar cases. With information gained through this court case comes “A Dark Night In Aurora” (Skyhorse Press, 2018), a careful dissection of the Aurora shooter by William H. Reid, MD, MPH. There are many books about crimes and criminals, but what caught my attention about this book was the author. Dr. Reid is one of five expert/forensic psychiatrists retained for the case and he was brought on by the judge as a second nonpartisan psychiatrist. Dr. Reid interviewed the defendant for more than 20 hours, he video-recorded the interviews, and he had access to, “... the existing seventy-five thousand pages of evidence, hundreds of audio and video files, and access to the defendants and scores of witnesses, experts, and others associated with the case ...” Dr. Reid notes that when the defendant placed his sanity in question, all confidentiality was waived.
In this case, the perpetrator had been treated by a therapist and two psychiatrists at his university counseling center in the semester before the shooting. There were video recordings of the defendant in his cell, a correctional psychiatrist in the local jail; psychiatric records from an admission to Denver Health Medical Center and two admissions to the Colorado Health Institute at Pueblo (a secure, forensic setting); and neuropsychological testing done by psychologists hired for the case. In all, there was a lot of mental health data, and I’m not aware of any other book that has been written by a psychiatrist about a criminal case where the author testified in the case.
Let me start by saying that the book is well written, very readable, and fascinating. I’d followed parts of the case in the news and had even watched some of the trial being live-streamed, so not all of the information was new, but Dr. Reid has put together the many facts of a perplexing case together in a cohesive way. There’s no literary descriptions or flowery writing; Dr. Reid is a focused writer. He also does a good job of explaining the specifics of Colorado state law to the forensic aspects of the case. In the end, there were nuances of how the shooter came to be found both competent and sane but was spared the death penalty by a single vote. Overall, I found the book haunting, and it would have been a more comfortable read if it were fiction. There was something terribly disconcerting about following along this miserable journey, all the while knowing that the killer ultimately would destroy so many lives and then watching his disconnected existence after the massacre.
As a general adult psychiatrist, I wanted to learn something very specific from this book: What is the recipe for creating a mass murderer and what are the warning signs a psychiatrist should catch? I would like my career to be free of patients who kill. The Aurora shooter’s family life sounded fairly pedestrian. His parents are professionals; there was a boy and a girl in an intact family with attentive parents, and no findings of abuse, torture, neglect, bullying, or anything out of the ordinary.
As a child, the shooter was noted for his kindness, and he was particularly caring toward his younger sister. As he grew older, reports about his personality were conflicting: Some saw him as awkward, and later even as odd, while others described him as normal, especially within a cohort of studious kids. He played on sports teams, was an excellent student, liked nonviolent video games, and while he was not outgoing, he always had some friends, though he shunned them during the semester before the shooting. He was not comfortable with women, but he had one girlfriend in graduate school, and when that relationship broke up, he texted with another classmate about how “hot” she was and that they went hiking together. The breakup may have contributed to the shooter’s unraveling, but if it did, Dr. Reid did not present this as the precipitant, and many young men are awkward with women – and endure breakups and rejection.
While the media said the shooter failed out of school, this was not the case. He did well in his classes but chose not to study for an important oral exam while he devoted his time to gathering weapons for his mission to kill, planning out the details, learning to use firearms, and rigging up a complex explosive system around his apartment. When he failed the oral exam, he was told he could study and retake it in a few weeks. He chose, instead, to withdraw from graduate school.
What stood out for me was that this young man talked about thoughts of killing people. He did so to the point of worrying his psychiatrist: She called in a second psychiatrist for a consultation, alerted the university’s threat assessment team, contacted an out-of-state parent, and considered admitting him on an involuntary hold. While many patients say they might hurt someone if provoked or threatened, few discuss thoughts of killing indiscriminately. But when people do talk about killing, we do our best to flush out their intentions, whether it is a fantasy or a plan, if they’ve been violent before. This shooter had no history of violence, and he hid from the psychiatrist the fact that he was acquiring weapons and actually planning a massacre. His psychiatrist diagnosed him with social anxiety, obsessive-compulsive disorder, and possible schizotypal personality disorder. He was prescribed an antidepressant, and later offered an antipsychotic, which he refused.
Three of the four psychiatrists who evaluated the defendant for the legal proceedings made a diagnosis of schizoaffective disorder. (The fifth psychiatrist for the prosecution never examined the defendant.) Dr. Reid made a diagnosis of schizotypal personality disorder. Everyone agreed that the shooter had a mental illness that influenced his actions. When it came to ascertaining the defendant’s mental state at the time of the crime, there was a glitch: The first psychiatrist for the defense examined the defendant only once, days after the shooting, and not again until 3 years later, right before the trial. His exam was limited: The defense attorney was present in the room, and he had instructed the psychiatrist not to ask about the shooting. The attorney interrupted the interview twice, and it’s not the usual practice to place these conditions on a forensic evaluation. After that exam, there was some misunderstanding about who was in charge of the treatment, and the defendant refused the services of the jail psychiatrist. It was not until 4 months later when the inmate became dehydrated, delirious, and psychotic that he finally was evaluated and treated. Despite the abundance of psychiatric information available, no direct and complete assessment was made by a psychiatrist immediately before the shooting, or in the months right after.
When we try to understand what motivates someone to commit such a heinous act, we look for psychosis. If the person suffers from paranoid delusions and believes his behavior is in self-defense, the behavior becomes understandable and justifiable. Even if it’s less organized, if it is the clear result of a psychotic thought process, we often attribute the behavior to illness. Usually, people with psychosis are too disorganized to enact complex plans, to acquire and learn to use ammunition, to plot out when and where there will be potential victims, and to plot out this degree of planning.
This shooter had an odd belief that if he killed people, he would attain “human capital.” Their deaths might fortify him, increase his value and decrease his suffering, and thereby prevent him from dying by suicide. Dr. Reid talks about whether it’s a delusion, an overvalued idea, or just an unusual belief. The shooter was aware that others don’t agree with this, that they would see it as wrong and criminal, and he thought there was only a 50% chance that it would work. If this were a delusion, it was not one we typically see, nor was it accompanied by more usual perceptual phenomena. Ultimately, there was no consensus on whether the patient was psychotic at the time of the shooting. On antipsychotic medication, he continued to believe that if he killed people that he would attain their human capital. In his book, Dr. Reid concluded that he remained dangerous.
Because the shooter told the university health center psychiatrist that he did not want to be locked up, she considered involuntary hospitalization, but she did not believe he would meet criteria for commitment. I saw nothing that indicated whether he was offered voluntary inpatient care with an explanation that hospital treatment is not the same as being locked up and might help alleviate his suffering. We don’t know if he could have been persuaded to enter the hospital willingly, but I suspect that this would have been difficult to justify to an insurer. There’s nothing to indicate that a hospitalization would have prevented this massacre.
From my perspective, I concluded that if individuals say they are thinking about killing strangers, they may be at risk of violence. This is a much smaller group of people to target for intervention than everyone with mental illness or everyone who is odd. This particular shooter appears to suffer from some type of mental derangement that does not fit neatly into our current psychiatric nomenclature or respond to our current treatments, and thoughtful psychiatric intervention – which he had – could not prevent his actions. It seems the only thing that would have changed this outcome is if someone had discovered his arsenal before July 20, 2012.
*Please note: The shooter’s name is intentionally omitted from my review.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016).
‘A Dark Night in Aurora’ gives view into one killer’s mind
‘A Dark Night in Aurora’ gives view into one killer’s mind
It was an unthinkable crime; on July 20, 2012, a 24-year-old neuroscience graduate student* walked into a movie theatre in Aurora, Colo., and began shooting people he had never before seen. Twelve people died and 70 were wounded in this calculated and intricately planned massacre that challenged everything we know about the intersection of mental illness and evil.
So much has been written about mass shooters as we’ve struggled to understand what motivates someone to kill strangers in a public place. The hope is that if we can understand it, then we can prevent it. The topic has been one of great interest to me because it gets quickly linked to advocacy for involuntary psychiatric care: Half of mass murderers suffer from psychiatric disorders and if we could force them to get treatment, then we could prevent these horrific events.

Many mass shooters die at the scene. Not only did the Aurora shooter survive, but his case went to trial – also a rarity – providing insights we have not had with other similar cases. With information gained through this court case comes “A Dark Night In Aurora” (Skyhorse Press, 2018), a careful dissection of the Aurora shooter by William H. Reid, MD, MPH. There are many books about crimes and criminals, but what caught my attention about this book was the author. Dr. Reid is one of five expert/forensic psychiatrists retained for the case and he was brought on by the judge as a second nonpartisan psychiatrist. Dr. Reid interviewed the defendant for more than 20 hours, he video-recorded the interviews, and he had access to, “... the existing seventy-five thousand pages of evidence, hundreds of audio and video files, and access to the defendants and scores of witnesses, experts, and others associated with the case ...” Dr. Reid notes that when the defendant placed his sanity in question, all confidentiality was waived.
In this case, the perpetrator had been treated by a therapist and two psychiatrists at his university counseling center in the semester before the shooting. There were video recordings of the defendant in his cell, a correctional psychiatrist in the local jail; psychiatric records from an admission to Denver Health Medical Center and two admissions to the Colorado Health Institute at Pueblo (a secure, forensic setting); and neuropsychological testing done by psychologists hired for the case. In all, there was a lot of mental health data, and I’m not aware of any other book that has been written by a psychiatrist about a criminal case where the author testified in the case.
Let me start by saying that the book is well written, very readable, and fascinating. I’d followed parts of the case in the news and had even watched some of the trial being live-streamed, so not all of the information was new, but Dr. Reid has put together the many facts of a perplexing case together in a cohesive way. There’s no literary descriptions or flowery writing; Dr. Reid is a focused writer. He also does a good job of explaining the specifics of Colorado state law to the forensic aspects of the case. In the end, there were nuances of how the shooter came to be found both competent and sane but was spared the death penalty by a single vote. Overall, I found the book haunting, and it would have been a more comfortable read if it were fiction. There was something terribly disconcerting about following along this miserable journey, all the while knowing that the killer ultimately would destroy so many lives and then watching his disconnected existence after the massacre.
As a general adult psychiatrist, I wanted to learn something very specific from this book: What is the recipe for creating a mass murderer and what are the warning signs a psychiatrist should catch? I would like my career to be free of patients who kill. The Aurora shooter’s family life sounded fairly pedestrian. His parents are professionals; there was a boy and a girl in an intact family with attentive parents, and no findings of abuse, torture, neglect, bullying, or anything out of the ordinary.
As a child, the shooter was noted for his kindness, and he was particularly caring toward his younger sister. As he grew older, reports about his personality were conflicting: Some saw him as awkward, and later even as odd, while others described him as normal, especially within a cohort of studious kids. He played on sports teams, was an excellent student, liked nonviolent video games, and while he was not outgoing, he always had some friends, though he shunned them during the semester before the shooting. He was not comfortable with women, but he had one girlfriend in graduate school, and when that relationship broke up, he texted with another classmate about how “hot” she was and that they went hiking together. The breakup may have contributed to the shooter’s unraveling, but if it did, Dr. Reid did not present this as the precipitant, and many young men are awkward with women – and endure breakups and rejection.
While the media said the shooter failed out of school, this was not the case. He did well in his classes but chose not to study for an important oral exam while he devoted his time to gathering weapons for his mission to kill, planning out the details, learning to use firearms, and rigging up a complex explosive system around his apartment. When he failed the oral exam, he was told he could study and retake it in a few weeks. He chose, instead, to withdraw from graduate school.
What stood out for me was that this young man talked about thoughts of killing people. He did so to the point of worrying his psychiatrist: She called in a second psychiatrist for a consultation, alerted the university’s threat assessment team, contacted an out-of-state parent, and considered admitting him on an involuntary hold. While many patients say they might hurt someone if provoked or threatened, few discuss thoughts of killing indiscriminately. But when people do talk about killing, we do our best to flush out their intentions, whether it is a fantasy or a plan, if they’ve been violent before. This shooter had no history of violence, and he hid from the psychiatrist the fact that he was acquiring weapons and actually planning a massacre. His psychiatrist diagnosed him with social anxiety, obsessive-compulsive disorder, and possible schizotypal personality disorder. He was prescribed an antidepressant, and later offered an antipsychotic, which he refused.
Three of the four psychiatrists who evaluated the defendant for the legal proceedings made a diagnosis of schizoaffective disorder. (The fifth psychiatrist for the prosecution never examined the defendant.) Dr. Reid made a diagnosis of schizotypal personality disorder. Everyone agreed that the shooter had a mental illness that influenced his actions. When it came to ascertaining the defendant’s mental state at the time of the crime, there was a glitch: The first psychiatrist for the defense examined the defendant only once, days after the shooting, and not again until 3 years later, right before the trial. His exam was limited: The defense attorney was present in the room, and he had instructed the psychiatrist not to ask about the shooting. The attorney interrupted the interview twice, and it’s not the usual practice to place these conditions on a forensic evaluation. After that exam, there was some misunderstanding about who was in charge of the treatment, and the defendant refused the services of the jail psychiatrist. It was not until 4 months later when the inmate became dehydrated, delirious, and psychotic that he finally was evaluated and treated. Despite the abundance of psychiatric information available, no direct and complete assessment was made by a psychiatrist immediately before the shooting, or in the months right after.
When we try to understand what motivates someone to commit such a heinous act, we look for psychosis. If the person suffers from paranoid delusions and believes his behavior is in self-defense, the behavior becomes understandable and justifiable. Even if it’s less organized, if it is the clear result of a psychotic thought process, we often attribute the behavior to illness. Usually, people with psychosis are too disorganized to enact complex plans, to acquire and learn to use ammunition, to plot out when and where there will be potential victims, and to plot out this degree of planning.
This shooter had an odd belief that if he killed people, he would attain “human capital.” Their deaths might fortify him, increase his value and decrease his suffering, and thereby prevent him from dying by suicide. Dr. Reid talks about whether it’s a delusion, an overvalued idea, or just an unusual belief. The shooter was aware that others don’t agree with this, that they would see it as wrong and criminal, and he thought there was only a 50% chance that it would work. If this were a delusion, it was not one we typically see, nor was it accompanied by more usual perceptual phenomena. Ultimately, there was no consensus on whether the patient was psychotic at the time of the shooting. On antipsychotic medication, he continued to believe that if he killed people that he would attain their human capital. In his book, Dr. Reid concluded that he remained dangerous.
Because the shooter told the university health center psychiatrist that he did not want to be locked up, she considered involuntary hospitalization, but she did not believe he would meet criteria for commitment. I saw nothing that indicated whether he was offered voluntary inpatient care with an explanation that hospital treatment is not the same as being locked up and might help alleviate his suffering. We don’t know if he could have been persuaded to enter the hospital willingly, but I suspect that this would have been difficult to justify to an insurer. There’s nothing to indicate that a hospitalization would have prevented this massacre.
From my perspective, I concluded that if individuals say they are thinking about killing strangers, they may be at risk of violence. This is a much smaller group of people to target for intervention than everyone with mental illness or everyone who is odd. This particular shooter appears to suffer from some type of mental derangement that does not fit neatly into our current psychiatric nomenclature or respond to our current treatments, and thoughtful psychiatric intervention – which he had – could not prevent his actions. It seems the only thing that would have changed this outcome is if someone had discovered his arsenal before July 20, 2012.
*Please note: The shooter’s name is intentionally omitted from my review.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016).
It was an unthinkable crime; on July 20, 2012, a 24-year-old neuroscience graduate student* walked into a movie theatre in Aurora, Colo., and began shooting people he had never before seen. Twelve people died and 70 were wounded in this calculated and intricately planned massacre that challenged everything we know about the intersection of mental illness and evil.
So much has been written about mass shooters as we’ve struggled to understand what motivates someone to kill strangers in a public place. The hope is that if we can understand it, then we can prevent it. The topic has been one of great interest to me because it gets quickly linked to advocacy for involuntary psychiatric care: Half of mass murderers suffer from psychiatric disorders and if we could force them to get treatment, then we could prevent these horrific events.

Many mass shooters die at the scene. Not only did the Aurora shooter survive, but his case went to trial – also a rarity – providing insights we have not had with other similar cases. With information gained through this court case comes “A Dark Night In Aurora” (Skyhorse Press, 2018), a careful dissection of the Aurora shooter by William H. Reid, MD, MPH. There are many books about crimes and criminals, but what caught my attention about this book was the author. Dr. Reid is one of five expert/forensic psychiatrists retained for the case and he was brought on by the judge as a second nonpartisan psychiatrist. Dr. Reid interviewed the defendant for more than 20 hours, he video-recorded the interviews, and he had access to, “... the existing seventy-five thousand pages of evidence, hundreds of audio and video files, and access to the defendants and scores of witnesses, experts, and others associated with the case ...” Dr. Reid notes that when the defendant placed his sanity in question, all confidentiality was waived.
In this case, the perpetrator had been treated by a therapist and two psychiatrists at his university counseling center in the semester before the shooting. There were video recordings of the defendant in his cell, a correctional psychiatrist in the local jail; psychiatric records from an admission to Denver Health Medical Center and two admissions to the Colorado Health Institute at Pueblo (a secure, forensic setting); and neuropsychological testing done by psychologists hired for the case. In all, there was a lot of mental health data, and I’m not aware of any other book that has been written by a psychiatrist about a criminal case where the author testified in the case.
Let me start by saying that the book is well written, very readable, and fascinating. I’d followed parts of the case in the news and had even watched some of the trial being live-streamed, so not all of the information was new, but Dr. Reid has put together the many facts of a perplexing case together in a cohesive way. There’s no literary descriptions or flowery writing; Dr. Reid is a focused writer. He also does a good job of explaining the specifics of Colorado state law to the forensic aspects of the case. In the end, there were nuances of how the shooter came to be found both competent and sane but was spared the death penalty by a single vote. Overall, I found the book haunting, and it would have been a more comfortable read if it were fiction. There was something terribly disconcerting about following along this miserable journey, all the while knowing that the killer ultimately would destroy so many lives and then watching his disconnected existence after the massacre.
As a general adult psychiatrist, I wanted to learn something very specific from this book: What is the recipe for creating a mass murderer and what are the warning signs a psychiatrist should catch? I would like my career to be free of patients who kill. The Aurora shooter’s family life sounded fairly pedestrian. His parents are professionals; there was a boy and a girl in an intact family with attentive parents, and no findings of abuse, torture, neglect, bullying, or anything out of the ordinary.
As a child, the shooter was noted for his kindness, and he was particularly caring toward his younger sister. As he grew older, reports about his personality were conflicting: Some saw him as awkward, and later even as odd, while others described him as normal, especially within a cohort of studious kids. He played on sports teams, was an excellent student, liked nonviolent video games, and while he was not outgoing, he always had some friends, though he shunned them during the semester before the shooting. He was not comfortable with women, but he had one girlfriend in graduate school, and when that relationship broke up, he texted with another classmate about how “hot” she was and that they went hiking together. The breakup may have contributed to the shooter’s unraveling, but if it did, Dr. Reid did not present this as the precipitant, and many young men are awkward with women – and endure breakups and rejection.
While the media said the shooter failed out of school, this was not the case. He did well in his classes but chose not to study for an important oral exam while he devoted his time to gathering weapons for his mission to kill, planning out the details, learning to use firearms, and rigging up a complex explosive system around his apartment. When he failed the oral exam, he was told he could study and retake it in a few weeks. He chose, instead, to withdraw from graduate school.
What stood out for me was that this young man talked about thoughts of killing people. He did so to the point of worrying his psychiatrist: She called in a second psychiatrist for a consultation, alerted the university’s threat assessment team, contacted an out-of-state parent, and considered admitting him on an involuntary hold. While many patients say they might hurt someone if provoked or threatened, few discuss thoughts of killing indiscriminately. But when people do talk about killing, we do our best to flush out their intentions, whether it is a fantasy or a plan, if they’ve been violent before. This shooter had no history of violence, and he hid from the psychiatrist the fact that he was acquiring weapons and actually planning a massacre. His psychiatrist diagnosed him with social anxiety, obsessive-compulsive disorder, and possible schizotypal personality disorder. He was prescribed an antidepressant, and later offered an antipsychotic, which he refused.
Three of the four psychiatrists who evaluated the defendant for the legal proceedings made a diagnosis of schizoaffective disorder. (The fifth psychiatrist for the prosecution never examined the defendant.) Dr. Reid made a diagnosis of schizotypal personality disorder. Everyone agreed that the shooter had a mental illness that influenced his actions. When it came to ascertaining the defendant’s mental state at the time of the crime, there was a glitch: The first psychiatrist for the defense examined the defendant only once, days after the shooting, and not again until 3 years later, right before the trial. His exam was limited: The defense attorney was present in the room, and he had instructed the psychiatrist not to ask about the shooting. The attorney interrupted the interview twice, and it’s not the usual practice to place these conditions on a forensic evaluation. After that exam, there was some misunderstanding about who was in charge of the treatment, and the defendant refused the services of the jail psychiatrist. It was not until 4 months later when the inmate became dehydrated, delirious, and psychotic that he finally was evaluated and treated. Despite the abundance of psychiatric information available, no direct and complete assessment was made by a psychiatrist immediately before the shooting, or in the months right after.
When we try to understand what motivates someone to commit such a heinous act, we look for psychosis. If the person suffers from paranoid delusions and believes his behavior is in self-defense, the behavior becomes understandable and justifiable. Even if it’s less organized, if it is the clear result of a psychotic thought process, we often attribute the behavior to illness. Usually, people with psychosis are too disorganized to enact complex plans, to acquire and learn to use ammunition, to plot out when and where there will be potential victims, and to plot out this degree of planning.
This shooter had an odd belief that if he killed people, he would attain “human capital.” Their deaths might fortify him, increase his value and decrease his suffering, and thereby prevent him from dying by suicide. Dr. Reid talks about whether it’s a delusion, an overvalued idea, or just an unusual belief. The shooter was aware that others don’t agree with this, that they would see it as wrong and criminal, and he thought there was only a 50% chance that it would work. If this were a delusion, it was not one we typically see, nor was it accompanied by more usual perceptual phenomena. Ultimately, there was no consensus on whether the patient was psychotic at the time of the shooting. On antipsychotic medication, he continued to believe that if he killed people that he would attain their human capital. In his book, Dr. Reid concluded that he remained dangerous.
Because the shooter told the university health center psychiatrist that he did not want to be locked up, she considered involuntary hospitalization, but she did not believe he would meet criteria for commitment. I saw nothing that indicated whether he was offered voluntary inpatient care with an explanation that hospital treatment is not the same as being locked up and might help alleviate his suffering. We don’t know if he could have been persuaded to enter the hospital willingly, but I suspect that this would have been difficult to justify to an insurer. There’s nothing to indicate that a hospitalization would have prevented this massacre.
From my perspective, I concluded that if individuals say they are thinking about killing strangers, they may be at risk of violence. This is a much smaller group of people to target for intervention than everyone with mental illness or everyone who is odd. This particular shooter appears to suffer from some type of mental derangement that does not fit neatly into our current psychiatric nomenclature or respond to our current treatments, and thoughtful psychiatric intervention – which he had – could not prevent his actions. It seems the only thing that would have changed this outcome is if someone had discovered his arsenal before July 20, 2012.
*Please note: The shooter’s name is intentionally omitted from my review.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016).