User login
JAK inhibitors may increase risk of herpes zoster
For patients with inflammatory bowel disease or other immune-mediated inflammatory diseases, Janus kinase (JAK) inhibitors appear generally safe, though they may increase the risk of herpes zoster infection, according to a large-scale systematic review and meta-analysis.
Data from more than 66,000 patients revealed no significant links between JAK inhibitors and risks of serious infections, malignancy, or major adverse cardiovascular events, reported lead author Pablo Olivera, MD, of Centro de Educación Médica e Investigación Clínica (CEMIC) in Buenos Aires and colleagues.
“To the best of our knowledge, this is the first systematic review evaluating the risk profile of JAK inhibitors in a wide spectrum of immune-mediated inflammatory diseases,” they wrote in Gastroenterology.
The investigators drew studies from the Cochrane Central Register of Controlled Trials, MEDLINE, and EMBASE from 1990 to 2019 and from conference databases from 2012 to 2018. Out of 973 studies identified, 82 were included in the final analysis, of which two-thirds were randomized clinical trials. In total, 101,925 subjects were included, of whom a majority had rheumatoid arthritis (n = 86,308), followed by psoriasis (n = 9,311), inflammatory bowel disease (n = 5,987), and ankylosing spondylitis (n = 319).
Meta-analysis of JAK inhibitor usage involved 66,159 patients. Four JAK inhibitors were included: tofacitinib, filgotinib, baricitinib, and upadacitinib. The primary outcomes were the incidence rates of adverse events and serious adverse events. The investigators also estimated incidence rates of herpes zoster infection, serious infections, mortality, malignancy, and major adverse cardiovascular events. These rates were compared with those of patients who received placebo or an active comparator in clinical trials.
Analysis showed that almost 9 out of 10 patients (87.16%) who were exposed to a JAK inhibitor received tofacitinib. The investigators described high variability in treatment duration and baseline characteristics of participants. Rates of adverse events and serious adverse events also fell across a broad spectrum, from 10% to 82% and from 0% to 29%, respectively.
“Most [adverse events] were mild, and included worsening of the underlying condition, probably showing lack of efficacy,” the investigators wrote.
Rates of mortality and most adverse events were not significantly associated with JAK inhibitor exposure. In contrast, relative risk of herpes zoster infection was 57% higher in patients who received a JAK inhibitor than in those who received a placebo or comparator (RR, 1.57; 95% confidence interval, 1.01-2.37).
“Regarding the risk of herpes zoster with JAK inhibitors, the largest evidence comes from the use of tofacitinib, but it appears to be a class effect, with a clear dose-dependent effect,” the investigators wrote.
Although risks of herpes zoster may be carried across the drug class, they may not be evenly distributed given that a subgroup analysis revealed that some JAK inhibitors may bring higher risks than others; specifically, tofacitinib and baricitinib were associated with higher relative risks of herpes zoster than were upadacitinib and filgotinib.
“Although this is merely a qualitative comparison, this difference could be related to the fact that both filgotinib and upadacitinib are selective JAK1 inhibitors, whereas tofacitinib is a JAK1/JAK3 inhibitor and baricitinib a JAK1/JAK2 inhibitor,” the investigators wrote. “Further studies are needed to determine if JAK isoform selectivity affects the risk of herpes zoster.”
The investigators emphasized this need for more research. While the present findings help illuminate the safety profile of JAK inhibitors, they are clouded by various other factors, such as disease-specific considerations, a lack of real-world data, and studies that are likely too short to accurately determine risk of malignancy, the investigators wrote.
“More studies with long follow-up and in the real world setting, in different conditions, will be needed to fully elucidate the safety profile of the different JAK inhibitors,” the investigators concluded.
The investigators disclosed relationships with AbbVie, Takeda, Pfizer, and others.
SOURCE: Olivera P et al. Gastroenterology. 2020 Jan 8. doi: 10.1053/j.gastro.2020.01.001.
The multiple different cytokines contributing to intestinal inflammation in IBD patients have been a major challenge in the design of therapies. Because the JAK signaling pathway (comprised of JAK1, JAK2, JAK3, and TYK2) is required for responses to a broad range of cytokines, therapies that inhibit JAK signaling have been an active area of interest. A simultaneous and important concern, however, has been the potential for adverse consequences when inhibiting the breadth of immune and hematopoietic molecules that depend on JAK family members for their functions. This meta-analysis by Olivera et al. examined adverse outcomes of four different JAK inhibitors in clinical trials across four immune-mediated diseases (rheumatoid arthritis, IBD, psoriasis, and ankylosing spondylitis), finding that herpes zoster infection was significantly increased (relative risk, 1.57). In contrast, patients treated with JAK inhibitors were not at a significantly increased risk for various other adverse events.
Reduced dosing of JAK inhibitors has been implemented as a means of improving safety profiles in select immune-mediated diseases. Another approach is more selective JAK inhibition, although it is unclear whether this will eliminate the risk of herpes zoster infection. In the current meta-analysis, about 87% of the studies had evaluated tofacitinib treatment, which inhibits both JAK1 and JAK3; more selective JAK inhibitors could not be evaluated in an equivalent manner. Of note, JAK1 is required for signaling by various cytokines that participate in the response to viruses, including type I IFNs and gamma c family members (such as IL-2 and IL-15); therefore, even the more selective JAK1 inhibitors do not leave this immune function fully intact. However, simply reducing the number of JAK family members inhibited simultaneously may be sufficient to reduce risk.
JAK inhibitors warrant further evaluation as additional infectious challenges arise, particularly with respect to viruses. In addition, more selective targeting of JAK inhibition of intestinal tissues may ultimately reduce systemic effects, including the risk of herpes zoster.
Clara Abraham, MD, professor of medicine, section of digestive diseases, Yale University, New Haven, Conn.
The multiple different cytokines contributing to intestinal inflammation in IBD patients have been a major challenge in the design of therapies. Because the JAK signaling pathway (comprised of JAK1, JAK2, JAK3, and TYK2) is required for responses to a broad range of cytokines, therapies that inhibit JAK signaling have been an active area of interest. A simultaneous and important concern, however, has been the potential for adverse consequences when inhibiting the breadth of immune and hematopoietic molecules that depend on JAK family members for their functions. This meta-analysis by Olivera et al. examined adverse outcomes of four different JAK inhibitors in clinical trials across four immune-mediated diseases (rheumatoid arthritis, IBD, psoriasis, and ankylosing spondylitis), finding that herpes zoster infection was significantly increased (relative risk, 1.57). In contrast, patients treated with JAK inhibitors were not at a significantly increased risk for various other adverse events.
Reduced dosing of JAK inhibitors has been implemented as a means of improving safety profiles in select immune-mediated diseases. Another approach is more selective JAK inhibition, although it is unclear whether this will eliminate the risk of herpes zoster infection. In the current meta-analysis, about 87% of the studies had evaluated tofacitinib treatment, which inhibits both JAK1 and JAK3; more selective JAK inhibitors could not be evaluated in an equivalent manner. Of note, JAK1 is required for signaling by various cytokines that participate in the response to viruses, including type I IFNs and gamma c family members (such as IL-2 and IL-15); therefore, even the more selective JAK1 inhibitors do not leave this immune function fully intact. However, simply reducing the number of JAK family members inhibited simultaneously may be sufficient to reduce risk.
JAK inhibitors warrant further evaluation as additional infectious challenges arise, particularly with respect to viruses. In addition, more selective targeting of JAK inhibition of intestinal tissues may ultimately reduce systemic effects, including the risk of herpes zoster.
Clara Abraham, MD, professor of medicine, section of digestive diseases, Yale University, New Haven, Conn.
The multiple different cytokines contributing to intestinal inflammation in IBD patients have been a major challenge in the design of therapies. Because the JAK signaling pathway (comprised of JAK1, JAK2, JAK3, and TYK2) is required for responses to a broad range of cytokines, therapies that inhibit JAK signaling have been an active area of interest. A simultaneous and important concern, however, has been the potential for adverse consequences when inhibiting the breadth of immune and hematopoietic molecules that depend on JAK family members for their functions. This meta-analysis by Olivera et al. examined adverse outcomes of four different JAK inhibitors in clinical trials across four immune-mediated diseases (rheumatoid arthritis, IBD, psoriasis, and ankylosing spondylitis), finding that herpes zoster infection was significantly increased (relative risk, 1.57). In contrast, patients treated with JAK inhibitors were not at a significantly increased risk for various other adverse events.
Reduced dosing of JAK inhibitors has been implemented as a means of improving safety profiles in select immune-mediated diseases. Another approach is more selective JAK inhibition, although it is unclear whether this will eliminate the risk of herpes zoster infection. In the current meta-analysis, about 87% of the studies had evaluated tofacitinib treatment, which inhibits both JAK1 and JAK3; more selective JAK inhibitors could not be evaluated in an equivalent manner. Of note, JAK1 is required for signaling by various cytokines that participate in the response to viruses, including type I IFNs and gamma c family members (such as IL-2 and IL-15); therefore, even the more selective JAK1 inhibitors do not leave this immune function fully intact. However, simply reducing the number of JAK family members inhibited simultaneously may be sufficient to reduce risk.
JAK inhibitors warrant further evaluation as additional infectious challenges arise, particularly with respect to viruses. In addition, more selective targeting of JAK inhibition of intestinal tissues may ultimately reduce systemic effects, including the risk of herpes zoster.
Clara Abraham, MD, professor of medicine, section of digestive diseases, Yale University, New Haven, Conn.
For patients with inflammatory bowel disease or other immune-mediated inflammatory diseases, Janus kinase (JAK) inhibitors appear generally safe, though they may increase the risk of herpes zoster infection, according to a large-scale systematic review and meta-analysis.
Data from more than 66,000 patients revealed no significant links between JAK inhibitors and risks of serious infections, malignancy, or major adverse cardiovascular events, reported lead author Pablo Olivera, MD, of Centro de Educación Médica e Investigación Clínica (CEMIC) in Buenos Aires and colleagues.
“To the best of our knowledge, this is the first systematic review evaluating the risk profile of JAK inhibitors in a wide spectrum of immune-mediated inflammatory diseases,” they wrote in Gastroenterology.
The investigators drew studies from the Cochrane Central Register of Controlled Trials, MEDLINE, and EMBASE from 1990 to 2019 and from conference databases from 2012 to 2018. Out of 973 studies identified, 82 were included in the final analysis, of which two-thirds were randomized clinical trials. In total, 101,925 subjects were included, of whom a majority had rheumatoid arthritis (n = 86,308), followed by psoriasis (n = 9,311), inflammatory bowel disease (n = 5,987), and ankylosing spondylitis (n = 319).
Meta-analysis of JAK inhibitor usage involved 66,159 patients. Four JAK inhibitors were included: tofacitinib, filgotinib, baricitinib, and upadacitinib. The primary outcomes were the incidence rates of adverse events and serious adverse events. The investigators also estimated incidence rates of herpes zoster infection, serious infections, mortality, malignancy, and major adverse cardiovascular events. These rates were compared with those of patients who received placebo or an active comparator in clinical trials.
Analysis showed that almost 9 out of 10 patients (87.16%) who were exposed to a JAK inhibitor received tofacitinib. The investigators described high variability in treatment duration and baseline characteristics of participants. Rates of adverse events and serious adverse events also fell across a broad spectrum, from 10% to 82% and from 0% to 29%, respectively.
“Most [adverse events] were mild, and included worsening of the underlying condition, probably showing lack of efficacy,” the investigators wrote.
Rates of mortality and most adverse events were not significantly associated with JAK inhibitor exposure. In contrast, relative risk of herpes zoster infection was 57% higher in patients who received a JAK inhibitor than in those who received a placebo or comparator (RR, 1.57; 95% confidence interval, 1.01-2.37).
“Regarding the risk of herpes zoster with JAK inhibitors, the largest evidence comes from the use of tofacitinib, but it appears to be a class effect, with a clear dose-dependent effect,” the investigators wrote.
Although risks of herpes zoster may be carried across the drug class, they may not be evenly distributed given that a subgroup analysis revealed that some JAK inhibitors may bring higher risks than others; specifically, tofacitinib and baricitinib were associated with higher relative risks of herpes zoster than were upadacitinib and filgotinib.
“Although this is merely a qualitative comparison, this difference could be related to the fact that both filgotinib and upadacitinib are selective JAK1 inhibitors, whereas tofacitinib is a JAK1/JAK3 inhibitor and baricitinib a JAK1/JAK2 inhibitor,” the investigators wrote. “Further studies are needed to determine if JAK isoform selectivity affects the risk of herpes zoster.”
The investigators emphasized this need for more research. While the present findings help illuminate the safety profile of JAK inhibitors, they are clouded by various other factors, such as disease-specific considerations, a lack of real-world data, and studies that are likely too short to accurately determine risk of malignancy, the investigators wrote.
“More studies with long follow-up and in the real world setting, in different conditions, will be needed to fully elucidate the safety profile of the different JAK inhibitors,” the investigators concluded.
The investigators disclosed relationships with AbbVie, Takeda, Pfizer, and others.
SOURCE: Olivera P et al. Gastroenterology. 2020 Jan 8. doi: 10.1053/j.gastro.2020.01.001.
For patients with inflammatory bowel disease or other immune-mediated inflammatory diseases, Janus kinase (JAK) inhibitors appear generally safe, though they may increase the risk of herpes zoster infection, according to a large-scale systematic review and meta-analysis.
Data from more than 66,000 patients revealed no significant links between JAK inhibitors and risks of serious infections, malignancy, or major adverse cardiovascular events, reported lead author Pablo Olivera, MD, of Centro de Educación Médica e Investigación Clínica (CEMIC) in Buenos Aires and colleagues.
“To the best of our knowledge, this is the first systematic review evaluating the risk profile of JAK inhibitors in a wide spectrum of immune-mediated inflammatory diseases,” they wrote in Gastroenterology.
The investigators drew studies from the Cochrane Central Register of Controlled Trials, MEDLINE, and EMBASE from 1990 to 2019 and from conference databases from 2012 to 2018. Out of 973 studies identified, 82 were included in the final analysis, of which two-thirds were randomized clinical trials. In total, 101,925 subjects were included, of whom a majority had rheumatoid arthritis (n = 86,308), followed by psoriasis (n = 9,311), inflammatory bowel disease (n = 5,987), and ankylosing spondylitis (n = 319).
Meta-analysis of JAK inhibitor usage involved 66,159 patients. Four JAK inhibitors were included: tofacitinib, filgotinib, baricitinib, and upadacitinib. The primary outcomes were the incidence rates of adverse events and serious adverse events. The investigators also estimated incidence rates of herpes zoster infection, serious infections, mortality, malignancy, and major adverse cardiovascular events. These rates were compared with those of patients who received placebo or an active comparator in clinical trials.
Analysis showed that almost 9 out of 10 patients (87.16%) who were exposed to a JAK inhibitor received tofacitinib. The investigators described high variability in treatment duration and baseline characteristics of participants. Rates of adverse events and serious adverse events also fell across a broad spectrum, from 10% to 82% and from 0% to 29%, respectively.
“Most [adverse events] were mild, and included worsening of the underlying condition, probably showing lack of efficacy,” the investigators wrote.
Rates of mortality and most adverse events were not significantly associated with JAK inhibitor exposure. In contrast, relative risk of herpes zoster infection was 57% higher in patients who received a JAK inhibitor than in those who received a placebo or comparator (RR, 1.57; 95% confidence interval, 1.01-2.37).
“Regarding the risk of herpes zoster with JAK inhibitors, the largest evidence comes from the use of tofacitinib, but it appears to be a class effect, with a clear dose-dependent effect,” the investigators wrote.
Although risks of herpes zoster may be carried across the drug class, they may not be evenly distributed given that a subgroup analysis revealed that some JAK inhibitors may bring higher risks than others; specifically, tofacitinib and baricitinib were associated with higher relative risks of herpes zoster than were upadacitinib and filgotinib.
“Although this is merely a qualitative comparison, this difference could be related to the fact that both filgotinib and upadacitinib are selective JAK1 inhibitors, whereas tofacitinib is a JAK1/JAK3 inhibitor and baricitinib a JAK1/JAK2 inhibitor,” the investigators wrote. “Further studies are needed to determine if JAK isoform selectivity affects the risk of herpes zoster.”
The investigators emphasized this need for more research. While the present findings help illuminate the safety profile of JAK inhibitors, they are clouded by various other factors, such as disease-specific considerations, a lack of real-world data, and studies that are likely too short to accurately determine risk of malignancy, the investigators wrote.
“More studies with long follow-up and in the real world setting, in different conditions, will be needed to fully elucidate the safety profile of the different JAK inhibitors,” the investigators concluded.
The investigators disclosed relationships with AbbVie, Takeda, Pfizer, and others.
SOURCE: Olivera P et al. Gastroenterology. 2020 Jan 8. doi: 10.1053/j.gastro.2020.01.001.
FROM GASTROENTEROLOGY
Rapid shift to adalimumab biosimilars in Denmark contrasts with U.S. experience
Adalimumab biosimilars are years away from entering the marketplace in the United States because of patent disputes, but they already have led to substantial discounts in Denmark, researchers wrote in JAMA Internal Medicine.
The Danish health care system switched almost entirely to adalimumab biosimilars after the patent on the original adalimumab product, Humira, expired there in October 2018. The switch to biosimilars led to an 82% decrease in costs for the medication, wrote Thomas Bo Jensen, MD, and colleagues in a research letter.
Denmark did not automatically substitute biosimilars, but the Danish Medicines Council recommended adalimumab biosimilars for all indications following Humira’s patent expiration. The recommendations “included switching patients to a biosimilar who were already well treated with the originator,” the researchers wrote.
To study the shift to adalimumab biosimilars across all indications in Denmark and calculate cost reductions, Dr. Jensen, of the department of clinical pharmacology at Copenhagen University Hospital Bispebjerg, and coinvestigators examined monthly data on drug sales from Amgros, which purchases all hospital drugs in the country.
“The proportion of adalimumab biosimilars increased from 71.6% (7,040 of 9,829 pens) in November 2018 to 95.1% (8,974 of 9,438 pens) in December 2018,” the researchers wrote. “Costs of adalimumab decreased by 82.8% from September 2018 to December 2018 (September: 8,197 pens at $5.13 million; December: 9,438 pens at $1.01 million).” The results were similar in rheumatology, dermatology, and gastroenterology.
The Food and Drug Administration has approved five adalimumab biosimilars in the United States, but “they will not enter the market until 2023 owing to patent disputes with AbbVie, the manufacturer of Humira,” wrote Jennifer D. Claytor, MD, of the department of internal medicine at University of California, San Francisco, and Walid Gellad, MD, of the division of general internal medicine at University of Pittsburgh, in an accompanying editorial.
The annual postrebate price of Humira doubled between 2013 and 2018, from $19,000 to $38,000, and these price increases may influence the price of biosimilars, “which will be priced using Humira’s price as an anchor,” Dr. Claytor and Dr. Gellad wrote.
A rapid shift to adalimumab biosimilars across the United States when they become available is “unlikely,” they wrote. Nonetheless, “some health care systems of comparable size to Denmark (e.g., the Veterans Affairs system) and others that are larger (e.g., Kaiser Permanente) ... have the ability to switch products quickly through use of formularies and a prescriber workforce. For example, Kaiser Permanente has successfully replaced Remicade (infliximab) with biosimilars in 80% of patients.”
Given the many biologics in development and increasing health care spending, “we need to take seriously the substantial savings offered by biosimilars and the feasibility, as evidenced by Denmark, of switching to biosimilars quickly once they are available on the market,” Dr. Claytor and Dr. Gellad concluded.
The research was supported by an unrestricted grant from Helsefonden. One author disclosed receiving grants from Pfizer, AbbVie, Roche, and Bristol-Myers Squibb outside the current study. The editorial authors had no disclosures.
SOURCE: Jensen TB et al. JAMA Intern Med. 2020 Mar 30. doi: 10.1001/jamainternmed.2020.0338.
Adalimumab biosimilars are years away from entering the marketplace in the United States because of patent disputes, but they already have led to substantial discounts in Denmark, researchers wrote in JAMA Internal Medicine.
The Danish health care system switched almost entirely to adalimumab biosimilars after the patent on the original adalimumab product, Humira, expired there in October 2018. The switch to biosimilars led to an 82% decrease in costs for the medication, wrote Thomas Bo Jensen, MD, and colleagues in a research letter.
Denmark did not automatically substitute biosimilars, but the Danish Medicines Council recommended adalimumab biosimilars for all indications following Humira’s patent expiration. The recommendations “included switching patients to a biosimilar who were already well treated with the originator,” the researchers wrote.
To study the shift to adalimumab biosimilars across all indications in Denmark and calculate cost reductions, Dr. Jensen, of the department of clinical pharmacology at Copenhagen University Hospital Bispebjerg, and coinvestigators examined monthly data on drug sales from Amgros, which purchases all hospital drugs in the country.
“The proportion of adalimumab biosimilars increased from 71.6% (7,040 of 9,829 pens) in November 2018 to 95.1% (8,974 of 9,438 pens) in December 2018,” the researchers wrote. “Costs of adalimumab decreased by 82.8% from September 2018 to December 2018 (September: 8,197 pens at $5.13 million; December: 9,438 pens at $1.01 million).” The results were similar in rheumatology, dermatology, and gastroenterology.
The Food and Drug Administration has approved five adalimumab biosimilars in the United States, but “they will not enter the market until 2023 owing to patent disputes with AbbVie, the manufacturer of Humira,” wrote Jennifer D. Claytor, MD, of the department of internal medicine at University of California, San Francisco, and Walid Gellad, MD, of the division of general internal medicine at University of Pittsburgh, in an accompanying editorial.
The annual postrebate price of Humira doubled between 2013 and 2018, from $19,000 to $38,000, and these price increases may influence the price of biosimilars, “which will be priced using Humira’s price as an anchor,” Dr. Claytor and Dr. Gellad wrote.
A rapid shift to adalimumab biosimilars across the United States when they become available is “unlikely,” they wrote. Nonetheless, “some health care systems of comparable size to Denmark (e.g., the Veterans Affairs system) and others that are larger (e.g., Kaiser Permanente) ... have the ability to switch products quickly through use of formularies and a prescriber workforce. For example, Kaiser Permanente has successfully replaced Remicade (infliximab) with biosimilars in 80% of patients.”
Given the many biologics in development and increasing health care spending, “we need to take seriously the substantial savings offered by biosimilars and the feasibility, as evidenced by Denmark, of switching to biosimilars quickly once they are available on the market,” Dr. Claytor and Dr. Gellad concluded.
The research was supported by an unrestricted grant from Helsefonden. One author disclosed receiving grants from Pfizer, AbbVie, Roche, and Bristol-Myers Squibb outside the current study. The editorial authors had no disclosures.
SOURCE: Jensen TB et al. JAMA Intern Med. 2020 Mar 30. doi: 10.1001/jamainternmed.2020.0338.
Adalimumab biosimilars are years away from entering the marketplace in the United States because of patent disputes, but they already have led to substantial discounts in Denmark, researchers wrote in JAMA Internal Medicine.
The Danish health care system switched almost entirely to adalimumab biosimilars after the patent on the original adalimumab product, Humira, expired there in October 2018. The switch to biosimilars led to an 82% decrease in costs for the medication, wrote Thomas Bo Jensen, MD, and colleagues in a research letter.
Denmark did not automatically substitute biosimilars, but the Danish Medicines Council recommended adalimumab biosimilars for all indications following Humira’s patent expiration. The recommendations “included switching patients to a biosimilar who were already well treated with the originator,” the researchers wrote.
To study the shift to adalimumab biosimilars across all indications in Denmark and calculate cost reductions, Dr. Jensen, of the department of clinical pharmacology at Copenhagen University Hospital Bispebjerg, and coinvestigators examined monthly data on drug sales from Amgros, which purchases all hospital drugs in the country.
“The proportion of adalimumab biosimilars increased from 71.6% (7,040 of 9,829 pens) in November 2018 to 95.1% (8,974 of 9,438 pens) in December 2018,” the researchers wrote. “Costs of adalimumab decreased by 82.8% from September 2018 to December 2018 (September: 8,197 pens at $5.13 million; December: 9,438 pens at $1.01 million).” The results were similar in rheumatology, dermatology, and gastroenterology.
The Food and Drug Administration has approved five adalimumab biosimilars in the United States, but “they will not enter the market until 2023 owing to patent disputes with AbbVie, the manufacturer of Humira,” wrote Jennifer D. Claytor, MD, of the department of internal medicine at University of California, San Francisco, and Walid Gellad, MD, of the division of general internal medicine at University of Pittsburgh, in an accompanying editorial.
The annual postrebate price of Humira doubled between 2013 and 2018, from $19,000 to $38,000, and these price increases may influence the price of biosimilars, “which will be priced using Humira’s price as an anchor,” Dr. Claytor and Dr. Gellad wrote.
A rapid shift to adalimumab biosimilars across the United States when they become available is “unlikely,” they wrote. Nonetheless, “some health care systems of comparable size to Denmark (e.g., the Veterans Affairs system) and others that are larger (e.g., Kaiser Permanente) ... have the ability to switch products quickly through use of formularies and a prescriber workforce. For example, Kaiser Permanente has successfully replaced Remicade (infliximab) with biosimilars in 80% of patients.”
Given the many biologics in development and increasing health care spending, “we need to take seriously the substantial savings offered by biosimilars and the feasibility, as evidenced by Denmark, of switching to biosimilars quickly once they are available on the market,” Dr. Claytor and Dr. Gellad concluded.
The research was supported by an unrestricted grant from Helsefonden. One author disclosed receiving grants from Pfizer, AbbVie, Roche, and Bristol-Myers Squibb outside the current study. The editorial authors had no disclosures.
SOURCE: Jensen TB et al. JAMA Intern Med. 2020 Mar 30. doi: 10.1001/jamainternmed.2020.0338.
FROM JAMA INTERNAL MEDICINE
What is seronegative rheumatoid arthritis, anyway?
MAUI, HAWAII – Viewing seronegative rheumatoid arthritis as something akin to RA-lite would be a big mistake, John J. Cush, MD, asserted at the 2020 Rheumatology Winter Clinical Symposium.
“It’s not a benign subtype of RA. And then again, it may not be RA,” Dr. Cush observed,
“Seronegative RA means that either you need to get serious about what is probably badass disease or you need to reevaluate whether this really is RA and your need for DMARDs [disease-modifying antirheumatic drugs] in an ongoing fashion,” the rheumatologist said. “Always reconsider whether they need less therapy or maybe no therapy at all. Maybe they had inflammation at one point and now they’re left with degenerative and mechanical changes that don’t require a DMARD or biologic.”
He highlighted a Finnish 10-year, prospective, observational study that sheds light on the subject. The study demonstrated that seronegative RA is seldom what it at first seems. The Finnish rheumatologists followed 435 consecutive patients initially diagnosed as having seronegative early RA. The structured follow-up entailed four or five interdisciplinary clinic visits within the first 2 years after diagnosis and again at 5 and 10 years.
By the 10-year mark only 4 of the 435 initially seronegative RA patients had been reclassified as having seropositive RA, while another 9 were reclassified as having erosive RA based upon the development of pathognomonic joint lesions. That’s a paltry 3% reclassification rate to classic RA.
Nearly two-thirds of patients were ultimately reclassified within 10 years as they evolved into diagnoses other than their original seronegative RA. The most common included nonerosive polymyalgia rheumatica in 16% of participants, psoriatic arthritis in 11%, osteoarthritis in 10%, spondyloarthritis in 8.7%, gout in 2.3%, and pseudogout in 3.9%.
“I think that’s sobering for you if you’re taking care of these patients, that maybe you need to rethink the diagnosis at every visit or at periodic intervals, especially if you’re going to change therapy,” advised Dr. Cush, who is professor of medicine and rheumatology at Baylor University Medical Center, Dallas, and director of clinical rheumatology at the Baylor Research Institute.
The Finnish rheumatologists observed that their findings have important implications both for clinical practice and for research, since RA clinical trials typically include a substantial proportion of seronegative patients.
“If seronegative patients are treated according to the treatment guidelines for progressive RA, a substantial proportion of patients is exposed to unnecessary long-term medication,” the investigators wrote, adding that their “results suggest that it may not be reasonable to study seronegative arthritis patients as a homogeneous entity in RA studies.”
The best recent data suggests about 15% of RA patients are seronegative, Dr. Cush said.
Delay in diagnosis is common in seronegative RA, as highlighted in a recent population-based study by Mayo Clinic rheumatologists. They reported that the median time from first joint swelling to diagnosis of seronegative RA using the 2010 American College of Rheumatology/European League Against Rheumatism criteria was 187 days, compared with a mere 11 days for seropositive RA. The median time to DMARD initiation was longer, too. Half of seropositive RA patients achieved remission within 5 years, as did 28% of seronegative patients, prompting the investigators to conclude “the window of opportunity for intervention may be more frequently missed in this group.”
Choosing the best treatment
Several medications appear to have greater efficacy in seropositive than seronegative RA patients. For example, a meta-analysis of four randomized trials including a collective 2,177 RA patients assigned 2:1 to rituximab (Rituxan) or placebo concluded that 75% of seropositive RA patients had a EULAR moderate or good response at week 24 on the biologic, compared with 44% of seronegative patients.
“Would you not use rituximab in someone who’s seronegative? No, I actually would use it. I may not rush to use it as much, maybe give it earlier in someone who’s seropositive, but I’ve used rituximab in seronegative patients who’ve done just fine,” according to Dr. Cush.
The published experience with abatacept (Orencia) is mixed, most of it coming from European observational datasets. On balance though, 80% of the articles addressing the issue have concluded that response rates to the biologic are better in seropositive RA, he continued.
Australian investigators who pooled data from five phase 3 randomized clinical trials of tofacitinib (Xeljanz) in RA concluded that double-positive patients – that is, those who were seropositive for both rheumatoid factor and anti–citrullinated protein antibody (ACPA) – were roughly twice as likely to achieve ACR20 and ACR50 responses to the oral Janus kinase inhibitor at either 5 or 10 mg twice daily than patients who were double negative.
“Double positivity is very important in prognosis and severity, compared to single positivity,” the rheumatologist observed. “I think you should worry most about the patients who have the highest titers of rheumatoid factor and ACPA.”
Asked about the merits of supplemental laboratory testing for serum 14-3-3 eta, a proposed novel biomarker in RA, as well as for anti–carbamylated protein antibodies (anti-CarP), Dr. Cush replied that it’s unclear that the additional testing is really worthwhile.
“Ordering more tests doesn’t make us smarter,” he commented. “Quite simply, with rheumatoid factor and ACPA, adding one on top of the other, you just gain maybe 10% more certainty in the diagnosis. Adding anti-CarP antibodies or serum 14-3-3 eta doesn’t add more than a few percentage points, but now you’ve quadrupled the cost of testing.”
Dr. Cush reported receiving research funding from and/or serving as a consultant to numerous pharmaceutical companies.
MAUI, HAWAII – Viewing seronegative rheumatoid arthritis as something akin to RA-lite would be a big mistake, John J. Cush, MD, asserted at the 2020 Rheumatology Winter Clinical Symposium.
“It’s not a benign subtype of RA. And then again, it may not be RA,” Dr. Cush observed,
“Seronegative RA means that either you need to get serious about what is probably badass disease or you need to reevaluate whether this really is RA and your need for DMARDs [disease-modifying antirheumatic drugs] in an ongoing fashion,” the rheumatologist said. “Always reconsider whether they need less therapy or maybe no therapy at all. Maybe they had inflammation at one point and now they’re left with degenerative and mechanical changes that don’t require a DMARD or biologic.”
He highlighted a Finnish 10-year, prospective, observational study that sheds light on the subject. The study demonstrated that seronegative RA is seldom what it at first seems. The Finnish rheumatologists followed 435 consecutive patients initially diagnosed as having seronegative early RA. The structured follow-up entailed four or five interdisciplinary clinic visits within the first 2 years after diagnosis and again at 5 and 10 years.
By the 10-year mark only 4 of the 435 initially seronegative RA patients had been reclassified as having seropositive RA, while another 9 were reclassified as having erosive RA based upon the development of pathognomonic joint lesions. That’s a paltry 3% reclassification rate to classic RA.
Nearly two-thirds of patients were ultimately reclassified within 10 years as they evolved into diagnoses other than their original seronegative RA. The most common included nonerosive polymyalgia rheumatica in 16% of participants, psoriatic arthritis in 11%, osteoarthritis in 10%, spondyloarthritis in 8.7%, gout in 2.3%, and pseudogout in 3.9%.
“I think that’s sobering for you if you’re taking care of these patients, that maybe you need to rethink the diagnosis at every visit or at periodic intervals, especially if you’re going to change therapy,” advised Dr. Cush, who is professor of medicine and rheumatology at Baylor University Medical Center, Dallas, and director of clinical rheumatology at the Baylor Research Institute.
The Finnish rheumatologists observed that their findings have important implications both for clinical practice and for research, since RA clinical trials typically include a substantial proportion of seronegative patients.
“If seronegative patients are treated according to the treatment guidelines for progressive RA, a substantial proportion of patients is exposed to unnecessary long-term medication,” the investigators wrote, adding that their “results suggest that it may not be reasonable to study seronegative arthritis patients as a homogeneous entity in RA studies.”
The best recent data suggests about 15% of RA patients are seronegative, Dr. Cush said.
Delay in diagnosis is common in seronegative RA, as highlighted in a recent population-based study by Mayo Clinic rheumatologists. They reported that the median time from first joint swelling to diagnosis of seronegative RA using the 2010 American College of Rheumatology/European League Against Rheumatism criteria was 187 days, compared with a mere 11 days for seropositive RA. The median time to DMARD initiation was longer, too. Half of seropositive RA patients achieved remission within 5 years, as did 28% of seronegative patients, prompting the investigators to conclude “the window of opportunity for intervention may be more frequently missed in this group.”
Choosing the best treatment
Several medications appear to have greater efficacy in seropositive than seronegative RA patients. For example, a meta-analysis of four randomized trials including a collective 2,177 RA patients assigned 2:1 to rituximab (Rituxan) or placebo concluded that 75% of seropositive RA patients had a EULAR moderate or good response at week 24 on the biologic, compared with 44% of seronegative patients.
“Would you not use rituximab in someone who’s seronegative? No, I actually would use it. I may not rush to use it as much, maybe give it earlier in someone who’s seropositive, but I’ve used rituximab in seronegative patients who’ve done just fine,” according to Dr. Cush.
The published experience with abatacept (Orencia) is mixed, most of it coming from European observational datasets. On balance though, 80% of the articles addressing the issue have concluded that response rates to the biologic are better in seropositive RA, he continued.
Australian investigators who pooled data from five phase 3 randomized clinical trials of tofacitinib (Xeljanz) in RA concluded that double-positive patients – that is, those who were seropositive for both rheumatoid factor and anti–citrullinated protein antibody (ACPA) – were roughly twice as likely to achieve ACR20 and ACR50 responses to the oral Janus kinase inhibitor at either 5 or 10 mg twice daily than patients who were double negative.
“Double positivity is very important in prognosis and severity, compared to single positivity,” the rheumatologist observed. “I think you should worry most about the patients who have the highest titers of rheumatoid factor and ACPA.”
Asked about the merits of supplemental laboratory testing for serum 14-3-3 eta, a proposed novel biomarker in RA, as well as for anti–carbamylated protein antibodies (anti-CarP), Dr. Cush replied that it’s unclear that the additional testing is really worthwhile.
“Ordering more tests doesn’t make us smarter,” he commented. “Quite simply, with rheumatoid factor and ACPA, adding one on top of the other, you just gain maybe 10% more certainty in the diagnosis. Adding anti-CarP antibodies or serum 14-3-3 eta doesn’t add more than a few percentage points, but now you’ve quadrupled the cost of testing.”
Dr. Cush reported receiving research funding from and/or serving as a consultant to numerous pharmaceutical companies.
MAUI, HAWAII – Viewing seronegative rheumatoid arthritis as something akin to RA-lite would be a big mistake, John J. Cush, MD, asserted at the 2020 Rheumatology Winter Clinical Symposium.
“It’s not a benign subtype of RA. And then again, it may not be RA,” Dr. Cush observed,
“Seronegative RA means that either you need to get serious about what is probably badass disease or you need to reevaluate whether this really is RA and your need for DMARDs [disease-modifying antirheumatic drugs] in an ongoing fashion,” the rheumatologist said. “Always reconsider whether they need less therapy or maybe no therapy at all. Maybe they had inflammation at one point and now they’re left with degenerative and mechanical changes that don’t require a DMARD or biologic.”
He highlighted a Finnish 10-year, prospective, observational study that sheds light on the subject. The study demonstrated that seronegative RA is seldom what it at first seems. The Finnish rheumatologists followed 435 consecutive patients initially diagnosed as having seronegative early RA. The structured follow-up entailed four or five interdisciplinary clinic visits within the first 2 years after diagnosis and again at 5 and 10 years.
By the 10-year mark only 4 of the 435 initially seronegative RA patients had been reclassified as having seropositive RA, while another 9 were reclassified as having erosive RA based upon the development of pathognomonic joint lesions. That’s a paltry 3% reclassification rate to classic RA.
Nearly two-thirds of patients were ultimately reclassified within 10 years as they evolved into diagnoses other than their original seronegative RA. The most common included nonerosive polymyalgia rheumatica in 16% of participants, psoriatic arthritis in 11%, osteoarthritis in 10%, spondyloarthritis in 8.7%, gout in 2.3%, and pseudogout in 3.9%.
“I think that’s sobering for you if you’re taking care of these patients, that maybe you need to rethink the diagnosis at every visit or at periodic intervals, especially if you’re going to change therapy,” advised Dr. Cush, who is professor of medicine and rheumatology at Baylor University Medical Center, Dallas, and director of clinical rheumatology at the Baylor Research Institute.
The Finnish rheumatologists observed that their findings have important implications both for clinical practice and for research, since RA clinical trials typically include a substantial proportion of seronegative patients.
“If seronegative patients are treated according to the treatment guidelines for progressive RA, a substantial proportion of patients is exposed to unnecessary long-term medication,” the investigators wrote, adding that their “results suggest that it may not be reasonable to study seronegative arthritis patients as a homogeneous entity in RA studies.”
The best recent data suggests about 15% of RA patients are seronegative, Dr. Cush said.
Delay in diagnosis is common in seronegative RA, as highlighted in a recent population-based study by Mayo Clinic rheumatologists. They reported that the median time from first joint swelling to diagnosis of seronegative RA using the 2010 American College of Rheumatology/European League Against Rheumatism criteria was 187 days, compared with a mere 11 days for seropositive RA. The median time to DMARD initiation was longer, too. Half of seropositive RA patients achieved remission within 5 years, as did 28% of seronegative patients, prompting the investigators to conclude “the window of opportunity for intervention may be more frequently missed in this group.”
Choosing the best treatment
Several medications appear to have greater efficacy in seropositive than seronegative RA patients. For example, a meta-analysis of four randomized trials including a collective 2,177 RA patients assigned 2:1 to rituximab (Rituxan) or placebo concluded that 75% of seropositive RA patients had a EULAR moderate or good response at week 24 on the biologic, compared with 44% of seronegative patients.
“Would you not use rituximab in someone who’s seronegative? No, I actually would use it. I may not rush to use it as much, maybe give it earlier in someone who’s seropositive, but I’ve used rituximab in seronegative patients who’ve done just fine,” according to Dr. Cush.
The published experience with abatacept (Orencia) is mixed, most of it coming from European observational datasets. On balance though, 80% of the articles addressing the issue have concluded that response rates to the biologic are better in seropositive RA, he continued.
Australian investigators who pooled data from five phase 3 randomized clinical trials of tofacitinib (Xeljanz) in RA concluded that double-positive patients – that is, those who were seropositive for both rheumatoid factor and anti–citrullinated protein antibody (ACPA) – were roughly twice as likely to achieve ACR20 and ACR50 responses to the oral Janus kinase inhibitor at either 5 or 10 mg twice daily than patients who were double negative.
“Double positivity is very important in prognosis and severity, compared to single positivity,” the rheumatologist observed. “I think you should worry most about the patients who have the highest titers of rheumatoid factor and ACPA.”
Asked about the merits of supplemental laboratory testing for serum 14-3-3 eta, a proposed novel biomarker in RA, as well as for anti–carbamylated protein antibodies (anti-CarP), Dr. Cush replied that it’s unclear that the additional testing is really worthwhile.
“Ordering more tests doesn’t make us smarter,” he commented. “Quite simply, with rheumatoid factor and ACPA, adding one on top of the other, you just gain maybe 10% more certainty in the diagnosis. Adding anti-CarP antibodies or serum 14-3-3 eta doesn’t add more than a few percentage points, but now you’ve quadrupled the cost of testing.”
Dr. Cush reported receiving research funding from and/or serving as a consultant to numerous pharmaceutical companies.
EXPERT ANALYSIS FROM RWCS 2020
Live zoster vaccine confers limited protection during tofacitinib therapy
The live attenuated zoster vaccine (Zostavax) does not provide adequate long-term protection in patients with rheumatoid arthritis (RA) starting tofacitinib, suggests the ORAL Sequel extension study.
The incidence of herpes zoster in patients with RA taking tofacitinib (Xeljanz), an oral Janus kinase inhibitor, is about double the rate seen with biologic disease-modifying antirheumatic drugs, noted the investigators, who were led by Kevin L. Winthrop, MD, professor of infectious diseases, ophthalmology, public health, and preventive medicine at Oregon Health & Science University, Portland. The American College of Rheumatology’s guideline for the treatment of RA recommends herpes zoster vaccination before patients aged 50 years or older initiate any of these agents.
The investigators studied 100 patients with RA from an index randomized, placebo-controlled trial of tofacitinib who started the long-term extension study 14 weeks after receiving the live attenuated zoster vaccine. All were given open-label tofacitinib, at 5 or 10 mg two times per day, along with background RA therapy as needed.
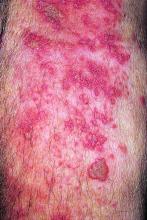
With a follow-up of 27 months, five patients (5%) developed herpes zoster, including two treated with the 5-mg dose and three treated with the 10-mg dose, according to results reported in Annals of the Rheumatic Diseases. Cases occurred between 218 and 741 days after vaccination.
Four of the patients had herpes zoster involving a single dermatome, while one had involvement of five dermatomes. All episodes were mild or moderate, and resolved with antiviral therapy.
Humoral and cell-mediated immunity to the varicella zoster virus were assessed with immunoglobulin G titer and an interferon-gamma enzyme-linked immunosorbent spot assay, respectively. Results showed that three of the patients developing herpes zoster had undetectable cell-mediated immunity to the virus at baseline and week 6 after vaccination. The other two patients had an adequate humoral and cell-mediated immune response to the vaccine, as assessed from changes from baseline, but had below-average immunoglobulin G titer at baseline and week 6.
“These results suggest that [live attenuated zoster vaccine] may not provide adequate long-term protection, as previously demonstrated in healthy individuals aged ≥60 years 3 years post-vaccination, in which [herpes zoster] risk was reduced by 51%,” Dr. Winthrop and colleagues wrote.
“While it is possible that [live attenuated zoster vaccine] booster vaccinations may improve vaccine efficacy, to date there is a lack of data on the use and timing of booster vaccinations, and no recommendations on the use of [live attenuated zoster vaccine] booster vaccinations currently exist,” they concluded. “This highlights the importance of evaluating the newly approved subunit non-live vaccine (Shingrix) in patients with RA receiving tofacitinib.”
The study was sponsored by Pfizer. Dr. Winthrop disclosed consulting for AbbVie, Bristol-Myers Squibb, Eli Lilly, Galapagos, Gilead, Pfizer, and UCB and receiving grant/research support from Bristol-Myers Squibb. Two coauthors disclosed financial relationships with Pfizer and other pharmaceutical companies, and the other seven coauthors were employees and shareholders of Pfizer.
SOURCE: Winthrop KL et al. Ann Rheum Dis. 2020 Mar 11. doi: 10.1136/annrheumdis-2019-216566.
The live attenuated zoster vaccine (Zostavax) does not provide adequate long-term protection in patients with rheumatoid arthritis (RA) starting tofacitinib, suggests the ORAL Sequel extension study.
The incidence of herpes zoster in patients with RA taking tofacitinib (Xeljanz), an oral Janus kinase inhibitor, is about double the rate seen with biologic disease-modifying antirheumatic drugs, noted the investigators, who were led by Kevin L. Winthrop, MD, professor of infectious diseases, ophthalmology, public health, and preventive medicine at Oregon Health & Science University, Portland. The American College of Rheumatology’s guideline for the treatment of RA recommends herpes zoster vaccination before patients aged 50 years or older initiate any of these agents.
The investigators studied 100 patients with RA from an index randomized, placebo-controlled trial of tofacitinib who started the long-term extension study 14 weeks after receiving the live attenuated zoster vaccine. All were given open-label tofacitinib, at 5 or 10 mg two times per day, along with background RA therapy as needed.
With a follow-up of 27 months, five patients (5%) developed herpes zoster, including two treated with the 5-mg dose and three treated with the 10-mg dose, according to results reported in Annals of the Rheumatic Diseases. Cases occurred between 218 and 741 days after vaccination.
Four of the patients had herpes zoster involving a single dermatome, while one had involvement of five dermatomes. All episodes were mild or moderate, and resolved with antiviral therapy.
Humoral and cell-mediated immunity to the varicella zoster virus were assessed with immunoglobulin G titer and an interferon-gamma enzyme-linked immunosorbent spot assay, respectively. Results showed that three of the patients developing herpes zoster had undetectable cell-mediated immunity to the virus at baseline and week 6 after vaccination. The other two patients had an adequate humoral and cell-mediated immune response to the vaccine, as assessed from changes from baseline, but had below-average immunoglobulin G titer at baseline and week 6.
“These results suggest that [live attenuated zoster vaccine] may not provide adequate long-term protection, as previously demonstrated in healthy individuals aged ≥60 years 3 years post-vaccination, in which [herpes zoster] risk was reduced by 51%,” Dr. Winthrop and colleagues wrote.
“While it is possible that [live attenuated zoster vaccine] booster vaccinations may improve vaccine efficacy, to date there is a lack of data on the use and timing of booster vaccinations, and no recommendations on the use of [live attenuated zoster vaccine] booster vaccinations currently exist,” they concluded. “This highlights the importance of evaluating the newly approved subunit non-live vaccine (Shingrix) in patients with RA receiving tofacitinib.”
The study was sponsored by Pfizer. Dr. Winthrop disclosed consulting for AbbVie, Bristol-Myers Squibb, Eli Lilly, Galapagos, Gilead, Pfizer, and UCB and receiving grant/research support from Bristol-Myers Squibb. Two coauthors disclosed financial relationships with Pfizer and other pharmaceutical companies, and the other seven coauthors were employees and shareholders of Pfizer.
SOURCE: Winthrop KL et al. Ann Rheum Dis. 2020 Mar 11. doi: 10.1136/annrheumdis-2019-216566.
The live attenuated zoster vaccine (Zostavax) does not provide adequate long-term protection in patients with rheumatoid arthritis (RA) starting tofacitinib, suggests the ORAL Sequel extension study.
The incidence of herpes zoster in patients with RA taking tofacitinib (Xeljanz), an oral Janus kinase inhibitor, is about double the rate seen with biologic disease-modifying antirheumatic drugs, noted the investigators, who were led by Kevin L. Winthrop, MD, professor of infectious diseases, ophthalmology, public health, and preventive medicine at Oregon Health & Science University, Portland. The American College of Rheumatology’s guideline for the treatment of RA recommends herpes zoster vaccination before patients aged 50 years or older initiate any of these agents.
The investigators studied 100 patients with RA from an index randomized, placebo-controlled trial of tofacitinib who started the long-term extension study 14 weeks after receiving the live attenuated zoster vaccine. All were given open-label tofacitinib, at 5 or 10 mg two times per day, along with background RA therapy as needed.
With a follow-up of 27 months, five patients (5%) developed herpes zoster, including two treated with the 5-mg dose and three treated with the 10-mg dose, according to results reported in Annals of the Rheumatic Diseases. Cases occurred between 218 and 741 days after vaccination.
Four of the patients had herpes zoster involving a single dermatome, while one had involvement of five dermatomes. All episodes were mild or moderate, and resolved with antiviral therapy.
Humoral and cell-mediated immunity to the varicella zoster virus were assessed with immunoglobulin G titer and an interferon-gamma enzyme-linked immunosorbent spot assay, respectively. Results showed that three of the patients developing herpes zoster had undetectable cell-mediated immunity to the virus at baseline and week 6 after vaccination. The other two patients had an adequate humoral and cell-mediated immune response to the vaccine, as assessed from changes from baseline, but had below-average immunoglobulin G titer at baseline and week 6.
“These results suggest that [live attenuated zoster vaccine] may not provide adequate long-term protection, as previously demonstrated in healthy individuals aged ≥60 years 3 years post-vaccination, in which [herpes zoster] risk was reduced by 51%,” Dr. Winthrop and colleagues wrote.
“While it is possible that [live attenuated zoster vaccine] booster vaccinations may improve vaccine efficacy, to date there is a lack of data on the use and timing of booster vaccinations, and no recommendations on the use of [live attenuated zoster vaccine] booster vaccinations currently exist,” they concluded. “This highlights the importance of evaluating the newly approved subunit non-live vaccine (Shingrix) in patients with RA receiving tofacitinib.”
The study was sponsored by Pfizer. Dr. Winthrop disclosed consulting for AbbVie, Bristol-Myers Squibb, Eli Lilly, Galapagos, Gilead, Pfizer, and UCB and receiving grant/research support from Bristol-Myers Squibb. Two coauthors disclosed financial relationships with Pfizer and other pharmaceutical companies, and the other seven coauthors were employees and shareholders of Pfizer.
SOURCE: Winthrop KL et al. Ann Rheum Dis. 2020 Mar 11. doi: 10.1136/annrheumdis-2019-216566.
FROM ANNALS OF THE RHEUMATIC DISEASES
Stronger links forged between RA and asthma, COPD
Asthma and chronic obstructive pulmonary disease were both linked to an increased risk of rheumatoid arthritis in a recent large, prospective cohort study, researchers have reported, which adds to a growing body of evidence that airway inflammation is implicated in the development of this disease.
RA risk was increased by about 50% among asthma patients, even when excluding those who had ever smoked, according to the study’s results, which were based on more than 200,000 women in the Nurses’ Health Study I and II.
Risk of RA nearly doubled among those with chronic obstructive pulmonary disease (COPD), with an even stronger association seen in older ever-smokers, according to authors of the study.
The findings not only strengthen the case for the potential role of obstructive lung diseases in RA development, according to the study’s authors, but also suggest that health care providers need to lower the bar for evaluation of patients with lung diseases and inflammatory joint symptoms.
“If these patients develop arthralgias, then the clinicians taking care of them should have a low threshold to consider RA, and perhaps refer, or check these patients with a diagnostic test for RA,” said researcher Jeffrey A. Sparks, MD, of Brigham and Women’s Hospital and Harvard Medical School in Boston.
What’s perhaps not as clear now is whether screening obstructive lung disease patients in the absence of early RA signs would be warranted: “I don’t know if we’re quite at the point where we would need to screen these patients if they’re not symptomatic,” Dr. Sparks said in an interview.
The study by Dr. Sparks and colleagues is, by far, not the first study to implicate asthma or other lung conditions in RA development. However, most previous studies are retrospective, and interpretation of the findings has been subject to limitations such as inadequate power to detect an increased risk or lack of adjustment for important confounding factors, such as smoking.
As such, the study by Dr. Sparks and colleagues is believed to be the first-ever prospective study to evaluate asthma and COPD as risk factors for RA, study authors reported in Arthritis & Rheumatology.
Researchers were able to identify 1,060 incident RA cases that developed in 15,148 women with asthma and 3,573 with COPD in the study with more than 4 million person-years of follow-up.
The association between asthma and increased RA risk was seen not only for the asthma population as a whole (hazard ratio, 1.53; 95% confidence interval, 1.24-1.88), but also for the subset of women who had never smoked, to a similar degree (HR, 1.53; 95% CI, 1.14-2.05), the report shows.
COPD’s association with RA risk was apparent overall (HR, 1.89; 95% CI, 1.31-2.75) and even more so in the subgroup of ever-smokers 55 years of age and older (HR, 2.20; 95% CI, 1.38-3.51), the data further show.
Findings of studies looking at the inflammation of airways and other mucosal sites are “critically important to understand” when it comes to trying to prevent RA, said Kevin Deane, MD, of the University of Colorado at Denver at Aurora.
“If we indeed are trying to prevent rheumatoid arthritis in terms of the joint disease, we may need to look at these mucosal sites in individuals who don’t yet have joint disease as potential sites to target for prevention, or at least areas to study to understand how prevention may work,” said Dr. Deane, principal investigator on the National Institutes of Health–funded Strategy for the Prevention of RA (StopRA) trial.
With that in mind, it’s conceivable targeting a lung process might prevent joint disease in a patient with asthma or airway inflammation and blood markers that indicate a risk of RA, Dr. Deane said in an interview.
Blood markers of RA have been evaluated in some recent studies, with findings that provide further evidence of a link between lung diseases and RA, and vice versa.
In particular, anti–citrullinated protein antibodies (ACPA) are clearly central to RA pathogenesis. And while asthma is increasingly linked to RA risk, there have been relatively little data on any potential links between ACPA and asthma.
That research gap led to a case-control study of the Nurses’ Health Study I and II (on which Dr. Sparks was senior author) showing that asthma was strongly linked to elevated ACPA in blood drawn from patients prior to a diagnosis of RA.
Results, published last year in Arthritis Research & Therapy, showed a significant association between asthma and pre-RA ACPA elevation (odds ratio, 3.57; 95% CI, 1.58-8.04), after adjustment for smoking and other potentially confounding factors. Investigators said the findings provided evidence that chronic mucosal airway inflammation is a factor in the development of ACPA and in the pathogenesis of RA.
In a follow-up study published more recently in Arthritis Care & Research, investigators showed that, among women in the Nurses Health Study I and II, pre-RA ACPA elevation was linked to increased risk of COPD, compared with controls (HR, 3.04; 95% CI, 1.33-7.00), while the risk for development of asthma was similar in women with or without elevated pre-RA ACPA.
That study was in part an attempt to establish a “timeline” related to antibodies, lung diseases, and RA onset, Dr. Sparks said in the interview.
“We think that probably the asthma is more important in developing the antibody, but that once you have the antibody, if you didn’t have asthma by then, you’re unlikely to develop it,” he said. “So asthma seems to be something that could happen before the antibody production, whereas COPD seems to happen after – but ACPA seems to be the common link in both of these obstructive lung diseases.”
The study in Arthritis & Rheumatology linking asthma and COPD to risk of incident RA was supported by the National Institutes of Health. Dr. Sparks reported grant support from Amgen and Bristol Myers Squibb and consulting fees from Inova and Optum. Coauthors provided disclosures related to GlaxoSmithKline, AstraZeneca, Merck, Neutrolis, and Genentech.
SOURCE: Ford JA et al. Arthritis Rheumatol. 2020 Mar 4. doi: 10.1002/art.41194.
Asthma and chronic obstructive pulmonary disease were both linked to an increased risk of rheumatoid arthritis in a recent large, prospective cohort study, researchers have reported, which adds to a growing body of evidence that airway inflammation is implicated in the development of this disease.
RA risk was increased by about 50% among asthma patients, even when excluding those who had ever smoked, according to the study’s results, which were based on more than 200,000 women in the Nurses’ Health Study I and II.
Risk of RA nearly doubled among those with chronic obstructive pulmonary disease (COPD), with an even stronger association seen in older ever-smokers, according to authors of the study.
The findings not only strengthen the case for the potential role of obstructive lung diseases in RA development, according to the study’s authors, but also suggest that health care providers need to lower the bar for evaluation of patients with lung diseases and inflammatory joint symptoms.
“If these patients develop arthralgias, then the clinicians taking care of them should have a low threshold to consider RA, and perhaps refer, or check these patients with a diagnostic test for RA,” said researcher Jeffrey A. Sparks, MD, of Brigham and Women’s Hospital and Harvard Medical School in Boston.
What’s perhaps not as clear now is whether screening obstructive lung disease patients in the absence of early RA signs would be warranted: “I don’t know if we’re quite at the point where we would need to screen these patients if they’re not symptomatic,” Dr. Sparks said in an interview.
The study by Dr. Sparks and colleagues is, by far, not the first study to implicate asthma or other lung conditions in RA development. However, most previous studies are retrospective, and interpretation of the findings has been subject to limitations such as inadequate power to detect an increased risk or lack of adjustment for important confounding factors, such as smoking.
As such, the study by Dr. Sparks and colleagues is believed to be the first-ever prospective study to evaluate asthma and COPD as risk factors for RA, study authors reported in Arthritis & Rheumatology.
Researchers were able to identify 1,060 incident RA cases that developed in 15,148 women with asthma and 3,573 with COPD in the study with more than 4 million person-years of follow-up.
The association between asthma and increased RA risk was seen not only for the asthma population as a whole (hazard ratio, 1.53; 95% confidence interval, 1.24-1.88), but also for the subset of women who had never smoked, to a similar degree (HR, 1.53; 95% CI, 1.14-2.05), the report shows.
COPD’s association with RA risk was apparent overall (HR, 1.89; 95% CI, 1.31-2.75) and even more so in the subgroup of ever-smokers 55 years of age and older (HR, 2.20; 95% CI, 1.38-3.51), the data further show.
Findings of studies looking at the inflammation of airways and other mucosal sites are “critically important to understand” when it comes to trying to prevent RA, said Kevin Deane, MD, of the University of Colorado at Denver at Aurora.
“If we indeed are trying to prevent rheumatoid arthritis in terms of the joint disease, we may need to look at these mucosal sites in individuals who don’t yet have joint disease as potential sites to target for prevention, or at least areas to study to understand how prevention may work,” said Dr. Deane, principal investigator on the National Institutes of Health–funded Strategy for the Prevention of RA (StopRA) trial.
With that in mind, it’s conceivable targeting a lung process might prevent joint disease in a patient with asthma or airway inflammation and blood markers that indicate a risk of RA, Dr. Deane said in an interview.
Blood markers of RA have been evaluated in some recent studies, with findings that provide further evidence of a link between lung diseases and RA, and vice versa.
In particular, anti–citrullinated protein antibodies (ACPA) are clearly central to RA pathogenesis. And while asthma is increasingly linked to RA risk, there have been relatively little data on any potential links between ACPA and asthma.
That research gap led to a case-control study of the Nurses’ Health Study I and II (on which Dr. Sparks was senior author) showing that asthma was strongly linked to elevated ACPA in blood drawn from patients prior to a diagnosis of RA.
Results, published last year in Arthritis Research & Therapy, showed a significant association between asthma and pre-RA ACPA elevation (odds ratio, 3.57; 95% CI, 1.58-8.04), after adjustment for smoking and other potentially confounding factors. Investigators said the findings provided evidence that chronic mucosal airway inflammation is a factor in the development of ACPA and in the pathogenesis of RA.
In a follow-up study published more recently in Arthritis Care & Research, investigators showed that, among women in the Nurses Health Study I and II, pre-RA ACPA elevation was linked to increased risk of COPD, compared with controls (HR, 3.04; 95% CI, 1.33-7.00), while the risk for development of asthma was similar in women with or without elevated pre-RA ACPA.
That study was in part an attempt to establish a “timeline” related to antibodies, lung diseases, and RA onset, Dr. Sparks said in the interview.
“We think that probably the asthma is more important in developing the antibody, but that once you have the antibody, if you didn’t have asthma by then, you’re unlikely to develop it,” he said. “So asthma seems to be something that could happen before the antibody production, whereas COPD seems to happen after – but ACPA seems to be the common link in both of these obstructive lung diseases.”
The study in Arthritis & Rheumatology linking asthma and COPD to risk of incident RA was supported by the National Institutes of Health. Dr. Sparks reported grant support from Amgen and Bristol Myers Squibb and consulting fees from Inova and Optum. Coauthors provided disclosures related to GlaxoSmithKline, AstraZeneca, Merck, Neutrolis, and Genentech.
SOURCE: Ford JA et al. Arthritis Rheumatol. 2020 Mar 4. doi: 10.1002/art.41194.
Asthma and chronic obstructive pulmonary disease were both linked to an increased risk of rheumatoid arthritis in a recent large, prospective cohort study, researchers have reported, which adds to a growing body of evidence that airway inflammation is implicated in the development of this disease.
RA risk was increased by about 50% among asthma patients, even when excluding those who had ever smoked, according to the study’s results, which were based on more than 200,000 women in the Nurses’ Health Study I and II.
Risk of RA nearly doubled among those with chronic obstructive pulmonary disease (COPD), with an even stronger association seen in older ever-smokers, according to authors of the study.
The findings not only strengthen the case for the potential role of obstructive lung diseases in RA development, according to the study’s authors, but also suggest that health care providers need to lower the bar for evaluation of patients with lung diseases and inflammatory joint symptoms.
“If these patients develop arthralgias, then the clinicians taking care of them should have a low threshold to consider RA, and perhaps refer, or check these patients with a diagnostic test for RA,” said researcher Jeffrey A. Sparks, MD, of Brigham and Women’s Hospital and Harvard Medical School in Boston.
What’s perhaps not as clear now is whether screening obstructive lung disease patients in the absence of early RA signs would be warranted: “I don’t know if we’re quite at the point where we would need to screen these patients if they’re not symptomatic,” Dr. Sparks said in an interview.
The study by Dr. Sparks and colleagues is, by far, not the first study to implicate asthma or other lung conditions in RA development. However, most previous studies are retrospective, and interpretation of the findings has been subject to limitations such as inadequate power to detect an increased risk or lack of adjustment for important confounding factors, such as smoking.
As such, the study by Dr. Sparks and colleagues is believed to be the first-ever prospective study to evaluate asthma and COPD as risk factors for RA, study authors reported in Arthritis & Rheumatology.
Researchers were able to identify 1,060 incident RA cases that developed in 15,148 women with asthma and 3,573 with COPD in the study with more than 4 million person-years of follow-up.
The association between asthma and increased RA risk was seen not only for the asthma population as a whole (hazard ratio, 1.53; 95% confidence interval, 1.24-1.88), but also for the subset of women who had never smoked, to a similar degree (HR, 1.53; 95% CI, 1.14-2.05), the report shows.
COPD’s association with RA risk was apparent overall (HR, 1.89; 95% CI, 1.31-2.75) and even more so in the subgroup of ever-smokers 55 years of age and older (HR, 2.20; 95% CI, 1.38-3.51), the data further show.
Findings of studies looking at the inflammation of airways and other mucosal sites are “critically important to understand” when it comes to trying to prevent RA, said Kevin Deane, MD, of the University of Colorado at Denver at Aurora.
“If we indeed are trying to prevent rheumatoid arthritis in terms of the joint disease, we may need to look at these mucosal sites in individuals who don’t yet have joint disease as potential sites to target for prevention, or at least areas to study to understand how prevention may work,” said Dr. Deane, principal investigator on the National Institutes of Health–funded Strategy for the Prevention of RA (StopRA) trial.
With that in mind, it’s conceivable targeting a lung process might prevent joint disease in a patient with asthma or airway inflammation and blood markers that indicate a risk of RA, Dr. Deane said in an interview.
Blood markers of RA have been evaluated in some recent studies, with findings that provide further evidence of a link between lung diseases and RA, and vice versa.
In particular, anti–citrullinated protein antibodies (ACPA) are clearly central to RA pathogenesis. And while asthma is increasingly linked to RA risk, there have been relatively little data on any potential links between ACPA and asthma.
That research gap led to a case-control study of the Nurses’ Health Study I and II (on which Dr. Sparks was senior author) showing that asthma was strongly linked to elevated ACPA in blood drawn from patients prior to a diagnosis of RA.
Results, published last year in Arthritis Research & Therapy, showed a significant association between asthma and pre-RA ACPA elevation (odds ratio, 3.57; 95% CI, 1.58-8.04), after adjustment for smoking and other potentially confounding factors. Investigators said the findings provided evidence that chronic mucosal airway inflammation is a factor in the development of ACPA and in the pathogenesis of RA.
In a follow-up study published more recently in Arthritis Care & Research, investigators showed that, among women in the Nurses Health Study I and II, pre-RA ACPA elevation was linked to increased risk of COPD, compared with controls (HR, 3.04; 95% CI, 1.33-7.00), while the risk for development of asthma was similar in women with or without elevated pre-RA ACPA.
That study was in part an attempt to establish a “timeline” related to antibodies, lung diseases, and RA onset, Dr. Sparks said in the interview.
“We think that probably the asthma is more important in developing the antibody, but that once you have the antibody, if you didn’t have asthma by then, you’re unlikely to develop it,” he said. “So asthma seems to be something that could happen before the antibody production, whereas COPD seems to happen after – but ACPA seems to be the common link in both of these obstructive lung diseases.”
The study in Arthritis & Rheumatology linking asthma and COPD to risk of incident RA was supported by the National Institutes of Health. Dr. Sparks reported grant support from Amgen and Bristol Myers Squibb and consulting fees from Inova and Optum. Coauthors provided disclosures related to GlaxoSmithKline, AstraZeneca, Merck, Neutrolis, and Genentech.
SOURCE: Ford JA et al. Arthritis Rheumatol. 2020 Mar 4. doi: 10.1002/art.41194.
FROM ARTHRITIS & RHEUMATOLOGY
Inactivated flu vaccine succeeds among autoimmune rheumatic disease patients
Use of the inactivated influenza vaccine by adults with autoimmune rheumatic diseases significantly reduced their risk of influenza-like illness, hospitalization for pneumonia and chronic obstructive pulmonary disease, and death from pneumonia, according to findings from an observational study of more than 30,000 patients in the U.K. Clinical Practice Research Datalink.
Although the inactivated vaccine has been recommended for patients with autoimmune rheumatic diseases (AIRDs), including rheumatoid arthritis and spondyloarthritis, the vaccine’s impact on patient outcomes including pneumonia, hospitalization, and death has not been well studied, wrote Georgina Nakafero, PhD, of the University of Nottingham, England, and colleagues.
In a study published in Rheumatology, the researchers identified 30,788 adults with AIRDs from the longitudinal Clinical Practice Research Datalink database in the United Kingdom. Of these, 66% were women, 76% had rheumatoid arthritis, and 61% had been prescribed methotrexate. The study included a total of 125,034 flu cycles between 2006 and 2009 and between 2010 and 2015.
Overall, vaccination with the inactivated influenza vaccine (IIV) reduced the risk of primary care consultation for influenza-like illness (adjusted odds ratio, 0.70), hospitalization for pneumonia (aOR, 0.61), exacerbation of chronic obstructive pulmonary disease (aOR, 0.67), and death caused by pneumonia (aOR, 0.48) in the study population. In a propensity score–adjusted analysis, only protection from influenza-like illness lost statistical significance.
In addition, vaccination was associated with a reduction in all-cause mortality among AIRDs patients, but restricting the outcomes to the active influenza periods may have confounded this result, the researchers said.
The study findings were limited by several factors including observational design, the use of a single vaccine efficacy estimate for each outcome, potential missed vaccination cycles, and potential confounding by indication and healthy user bias that could inflate the vaccine effectiveness, the researchers noted. However, the results were strengthened by the large sample size, including a range of AIRDs, and the use of both diagnostic and prescription codes, they said.
“The findings of this study, together with the results of our previous study demonstrating the safety of IIV in people with AIRDs, provides evidence to promote seasonal flu vaccination in this population,” they concluded. They still emphasized that randomized, controlled trials are needed for an assessment of vaccine efficacy.
The study was supported by Versus Arthritis and the National Institute of Health Research. Lead author Dr. Nakafero had no financial conflicts to disclose. Several coauthors disclosed relationships with companies, including AstraZeneca, Roche, and Pfizer.
SOURCE: Nakafero G et al. Rheumatology. 2020 Mar 11. doi: 10.1093/rheumatology/keaa078.
Use of the inactivated influenza vaccine by adults with autoimmune rheumatic diseases significantly reduced their risk of influenza-like illness, hospitalization for pneumonia and chronic obstructive pulmonary disease, and death from pneumonia, according to findings from an observational study of more than 30,000 patients in the U.K. Clinical Practice Research Datalink.
Although the inactivated vaccine has been recommended for patients with autoimmune rheumatic diseases (AIRDs), including rheumatoid arthritis and spondyloarthritis, the vaccine’s impact on patient outcomes including pneumonia, hospitalization, and death has not been well studied, wrote Georgina Nakafero, PhD, of the University of Nottingham, England, and colleagues.
In a study published in Rheumatology, the researchers identified 30,788 adults with AIRDs from the longitudinal Clinical Practice Research Datalink database in the United Kingdom. Of these, 66% were women, 76% had rheumatoid arthritis, and 61% had been prescribed methotrexate. The study included a total of 125,034 flu cycles between 2006 and 2009 and between 2010 and 2015.
Overall, vaccination with the inactivated influenza vaccine (IIV) reduced the risk of primary care consultation for influenza-like illness (adjusted odds ratio, 0.70), hospitalization for pneumonia (aOR, 0.61), exacerbation of chronic obstructive pulmonary disease (aOR, 0.67), and death caused by pneumonia (aOR, 0.48) in the study population. In a propensity score–adjusted analysis, only protection from influenza-like illness lost statistical significance.
In addition, vaccination was associated with a reduction in all-cause mortality among AIRDs patients, but restricting the outcomes to the active influenza periods may have confounded this result, the researchers said.
The study findings were limited by several factors including observational design, the use of a single vaccine efficacy estimate for each outcome, potential missed vaccination cycles, and potential confounding by indication and healthy user bias that could inflate the vaccine effectiveness, the researchers noted. However, the results were strengthened by the large sample size, including a range of AIRDs, and the use of both diagnostic and prescription codes, they said.
“The findings of this study, together with the results of our previous study demonstrating the safety of IIV in people with AIRDs, provides evidence to promote seasonal flu vaccination in this population,” they concluded. They still emphasized that randomized, controlled trials are needed for an assessment of vaccine efficacy.
The study was supported by Versus Arthritis and the National Institute of Health Research. Lead author Dr. Nakafero had no financial conflicts to disclose. Several coauthors disclosed relationships with companies, including AstraZeneca, Roche, and Pfizer.
SOURCE: Nakafero G et al. Rheumatology. 2020 Mar 11. doi: 10.1093/rheumatology/keaa078.
Use of the inactivated influenza vaccine by adults with autoimmune rheumatic diseases significantly reduced their risk of influenza-like illness, hospitalization for pneumonia and chronic obstructive pulmonary disease, and death from pneumonia, according to findings from an observational study of more than 30,000 patients in the U.K. Clinical Practice Research Datalink.
Although the inactivated vaccine has been recommended for patients with autoimmune rheumatic diseases (AIRDs), including rheumatoid arthritis and spondyloarthritis, the vaccine’s impact on patient outcomes including pneumonia, hospitalization, and death has not been well studied, wrote Georgina Nakafero, PhD, of the University of Nottingham, England, and colleagues.
In a study published in Rheumatology, the researchers identified 30,788 adults with AIRDs from the longitudinal Clinical Practice Research Datalink database in the United Kingdom. Of these, 66% were women, 76% had rheumatoid arthritis, and 61% had been prescribed methotrexate. The study included a total of 125,034 flu cycles between 2006 and 2009 and between 2010 and 2015.
Overall, vaccination with the inactivated influenza vaccine (IIV) reduced the risk of primary care consultation for influenza-like illness (adjusted odds ratio, 0.70), hospitalization for pneumonia (aOR, 0.61), exacerbation of chronic obstructive pulmonary disease (aOR, 0.67), and death caused by pneumonia (aOR, 0.48) in the study population. In a propensity score–adjusted analysis, only protection from influenza-like illness lost statistical significance.
In addition, vaccination was associated with a reduction in all-cause mortality among AIRDs patients, but restricting the outcomes to the active influenza periods may have confounded this result, the researchers said.
The study findings were limited by several factors including observational design, the use of a single vaccine efficacy estimate for each outcome, potential missed vaccination cycles, and potential confounding by indication and healthy user bias that could inflate the vaccine effectiveness, the researchers noted. However, the results were strengthened by the large sample size, including a range of AIRDs, and the use of both diagnostic and prescription codes, they said.
“The findings of this study, together with the results of our previous study demonstrating the safety of IIV in people with AIRDs, provides evidence to promote seasonal flu vaccination in this population,” they concluded. They still emphasized that randomized, controlled trials are needed for an assessment of vaccine efficacy.
The study was supported by Versus Arthritis and the National Institute of Health Research. Lead author Dr. Nakafero had no financial conflicts to disclose. Several coauthors disclosed relationships with companies, including AstraZeneca, Roche, and Pfizer.
SOURCE: Nakafero G et al. Rheumatology. 2020 Mar 11. doi: 10.1093/rheumatology/keaa078.
FROM RHEUMATOLOGY
Key clinical point: Adults with autoimmune rheumatic diseases who received the inactivated flu vaccine had lower rates of flu-like illness, hospitalization, and death than did those not vaccinated.
Major finding: Vaccination significantly reduced the risk of flu-like illness, hospitalization for pneumonia or COPD exacerbation, and death from pneumonia by 30%, 39%, 33%, and 52%, respectively.
Study details: The data come from 30,788 adults with AIRD and included 125,034 influenza cycles.
Disclosures: The study was supported by Versus Arthritis and the National Institute of Health Research. Lead author Dr. Nakafero had no financial conflicts to disclose. Several coauthors disclosed relationships with companies, including AstraZeneca, Roche, and Pfizer.
Source: Nakafero G et al. Rheumatology. 2020 Mar 11. doi: 10.1093/rheumatology/keaa078.
New RA diagnosis prompts patients to quit smoking
Health care factors, such as type of health system and being newly diagnosed, rather than patient socioeconomic factors, were significant predictors of smoking cessation in patients with RA, according to findings published in Arthritis Care & Research.
Smoking is a known risk factor for poor outcomes in RA, but data on factors that predict smoking cessation in these patients are limited, wrote Maria Schletzbaum, a student in the MD-PhD program at the University of Wisconsin–Madison, and colleagues. “Further, most patients with RA are not aware of the associations between smoking and RA development and complications, although such knowledge could influence cessation attempts.”
To identify predictors of smoking cessation, the investigators reviewed the 2005-2016 electronic medical records for 3,577 adults aged 18 years or older and with at least two RA diagnosis codes. The records were from two health systems, one urban and one rural.
Overall, patients who were baseline smokers and who were new to rheumatology care were 60% more likely to quit smoking (adjusted odds ratio, 1.60). However, patients who were seropositive and baseline smokers were 43% less likely to quit (aOR, 0.57). Demographic factors, including age, race, and sex, were not significantly associated with smoking cessation.
“The observed increased likelihood of quitting in patients new to rheumatology care may partially be due to cessation following a new RA diagnosis, a phenomenon previously reported in RA and other chronic diseases,” the researchers noted. Notably, the significance of being new to rheumatology disappeared in an analysis controlling for health system, “potentially suggesting greater importance of system-level factors versus individual patient factors.”
In addition, patients in the rural community health system were 66% more likely to quit smoking, possibly because about half of the patients in that system were covered by the systems’ insurance, and therefore qualified for various smoking cessation interventions, they wrote.
The study population included 915 former smokers and 507 current smokers. Seropositivity was most common in current smokers (71%), followed by former smokers (64%) and never smokers (59%). The disinclination of seropositive patients to quit smoking may reflect greater smoking intensity, and these patients may need greater support, the researchers wrote.
The study findings were limited by several factors; for example, some differences in patient populations, such as education and income levels, were not measured, and there was a lack of complete information on cumulative smoking exposure. However, the results were strengthened by the large sample size and use of data from two centers and support national guidelines for health system interventions in smoking cessation for RA patients, they noted.
The study was funded in part by the Rheumatology Research Foundation, the University of Wisconsin Medical Scientist Training Program, and the University of Wisconsin Clinical and Translational Science Award. The authors did not report any financial conflicts of interest.
SOURCE: Schletzbaum M et al. Arthritis Car Res. 2020 Mar 3. doi: 10.1002/ACR.24154.
Health care factors, such as type of health system and being newly diagnosed, rather than patient socioeconomic factors, were significant predictors of smoking cessation in patients with RA, according to findings published in Arthritis Care & Research.
Smoking is a known risk factor for poor outcomes in RA, but data on factors that predict smoking cessation in these patients are limited, wrote Maria Schletzbaum, a student in the MD-PhD program at the University of Wisconsin–Madison, and colleagues. “Further, most patients with RA are not aware of the associations between smoking and RA development and complications, although such knowledge could influence cessation attempts.”
To identify predictors of smoking cessation, the investigators reviewed the 2005-2016 electronic medical records for 3,577 adults aged 18 years or older and with at least two RA diagnosis codes. The records were from two health systems, one urban and one rural.
Overall, patients who were baseline smokers and who were new to rheumatology care were 60% more likely to quit smoking (adjusted odds ratio, 1.60). However, patients who were seropositive and baseline smokers were 43% less likely to quit (aOR, 0.57). Demographic factors, including age, race, and sex, were not significantly associated with smoking cessation.
“The observed increased likelihood of quitting in patients new to rheumatology care may partially be due to cessation following a new RA diagnosis, a phenomenon previously reported in RA and other chronic diseases,” the researchers noted. Notably, the significance of being new to rheumatology disappeared in an analysis controlling for health system, “potentially suggesting greater importance of system-level factors versus individual patient factors.”
In addition, patients in the rural community health system were 66% more likely to quit smoking, possibly because about half of the patients in that system were covered by the systems’ insurance, and therefore qualified for various smoking cessation interventions, they wrote.
The study population included 915 former smokers and 507 current smokers. Seropositivity was most common in current smokers (71%), followed by former smokers (64%) and never smokers (59%). The disinclination of seropositive patients to quit smoking may reflect greater smoking intensity, and these patients may need greater support, the researchers wrote.
The study findings were limited by several factors; for example, some differences in patient populations, such as education and income levels, were not measured, and there was a lack of complete information on cumulative smoking exposure. However, the results were strengthened by the large sample size and use of data from two centers and support national guidelines for health system interventions in smoking cessation for RA patients, they noted.
The study was funded in part by the Rheumatology Research Foundation, the University of Wisconsin Medical Scientist Training Program, and the University of Wisconsin Clinical and Translational Science Award. The authors did not report any financial conflicts of interest.
SOURCE: Schletzbaum M et al. Arthritis Car Res. 2020 Mar 3. doi: 10.1002/ACR.24154.
Health care factors, such as type of health system and being newly diagnosed, rather than patient socioeconomic factors, were significant predictors of smoking cessation in patients with RA, according to findings published in Arthritis Care & Research.
Smoking is a known risk factor for poor outcomes in RA, but data on factors that predict smoking cessation in these patients are limited, wrote Maria Schletzbaum, a student in the MD-PhD program at the University of Wisconsin–Madison, and colleagues. “Further, most patients with RA are not aware of the associations between smoking and RA development and complications, although such knowledge could influence cessation attempts.”
To identify predictors of smoking cessation, the investigators reviewed the 2005-2016 electronic medical records for 3,577 adults aged 18 years or older and with at least two RA diagnosis codes. The records were from two health systems, one urban and one rural.
Overall, patients who were baseline smokers and who were new to rheumatology care were 60% more likely to quit smoking (adjusted odds ratio, 1.60). However, patients who were seropositive and baseline smokers were 43% less likely to quit (aOR, 0.57). Demographic factors, including age, race, and sex, were not significantly associated with smoking cessation.
“The observed increased likelihood of quitting in patients new to rheumatology care may partially be due to cessation following a new RA diagnosis, a phenomenon previously reported in RA and other chronic diseases,” the researchers noted. Notably, the significance of being new to rheumatology disappeared in an analysis controlling for health system, “potentially suggesting greater importance of system-level factors versus individual patient factors.”
In addition, patients in the rural community health system were 66% more likely to quit smoking, possibly because about half of the patients in that system were covered by the systems’ insurance, and therefore qualified for various smoking cessation interventions, they wrote.
The study population included 915 former smokers and 507 current smokers. Seropositivity was most common in current smokers (71%), followed by former smokers (64%) and never smokers (59%). The disinclination of seropositive patients to quit smoking may reflect greater smoking intensity, and these patients may need greater support, the researchers wrote.
The study findings were limited by several factors; for example, some differences in patient populations, such as education and income levels, were not measured, and there was a lack of complete information on cumulative smoking exposure. However, the results were strengthened by the large sample size and use of data from two centers and support national guidelines for health system interventions in smoking cessation for RA patients, they noted.
The study was funded in part by the Rheumatology Research Foundation, the University of Wisconsin Medical Scientist Training Program, and the University of Wisconsin Clinical and Translational Science Award. The authors did not report any financial conflicts of interest.
SOURCE: Schletzbaum M et al. Arthritis Car Res. 2020 Mar 3. doi: 10.1002/ACR.24154.
FROM ARTHRITIS CARE & RESEARCH
Genetic variants in nudix hydrolase linked to thiopurine myelosuppression
MAUI, HAWAII – There’s a new kid on the block to worry about when it comes to thiopurine pharmacogenetics: Genetic variants in the thiopurine-metabolizing enzyme nudix hydrolase 15 have been linked to a markedly increased risk of thiopurine myelosuppression among inflammatory bowel disease patients.
The Food and Drug Administration and others already recommend screening for genetic variants in thiopurine methyltransferase (TPMT), another enzyme that metabolizes thiopurine. Polymorphisms lead to TMPT dysfunction, accumulation of cytotoxic metabolites, and increased risk of thiopurine-induced myelosuppression (TIM). Carriers are advised to use reduced doses with careful drug monitoring, or to skip thiopurines altogether.
A similar picture is emerging for nudix hydrolase 15 (NUDT15). It’s been known for several years that genetic variants are not uncommon among East Asian people and lead to TIM, but their prevalence and impact among people of European decent wasn’t clearly understood until now.
Investigators led by Gareth Walker, MBBS, of the Royal Devon and Exeter Hospital in Exeter, England, compared rates of problematic TMPT and NUDT15 variants among European inflammatory bowel disease patients who had developed TIM and those who had not, about 1,000 patients in all. The majority were on azathioprine and had Crohn’s disease. Finnish people were excluded because “their unique genetic background ... has led to the enrichment of some disease-causing gene variants and losses of others,” according to the study, which was published in JAMA.
Carriage of any of three coding NUDT15 variants greatly increased the risk of TIM (odds ratio, 27.3; 95% confidence interval, 9.3-116.7), independent of TPMT genotype and thiopurine dose. A particular variant – an in-frame deletion in NUDT15 – increased the risk 38-fold (95% CI, 5.1-286.1), and was carried by 5.8% of TIM patients.
The analysis also confirmed the importance of TPMT variants, which were found in 30.5% of TIM patients (95/311) versus 16.4% (100/608) of patients who did not develop TIM.
“Patients with variants of either NUDT15 or TPMT, or among those with variants of both genes, had a faster onset of TIM, more severe TIM, and had a greater need for granulocyte colony–stimulating factor rescue therapy. ... Our data suggest that pretreatment sequencing of the NUDT15 gene ... may be considered prior to initiation of thiopurine therapy,” the team concluded.
The prevalence of problematic NUDT15 variants among non-Finnish Europeans is about 1.6%, 6.9% among people from Finland, and almost 30% among East Asians. The team estimated NUDT15 would have to be genotyped in 95 non-Finnish Europeans to prevent one case of TIM; the number is 123 for TPMT. “Given the widespread use of thiopurines” – primarily in rheumatology and transplant medicine, in addition to gastroenterology – “these findings may have ramifications beyond the management of IBD,” the investigators wrote.
“I do think it’s worthwhile” to screen for NUDT15, said Edward Loftus, MD, a professor and consultant in the division of gastroenterology and hepatology at the Mayo Clinic in Rochester, Minn., who reviewed the study at the Gastroenterology Updates, IBD, Liver Disease Conference. “If you are a homozygote for this, your chance of getting profound leukopenia is very high, so I would probably not use a thiopurine.”
“If you are going to start low dose on everyone” with careful blood monitoring, “I suppose you could just do that, but I would say if you can get” the test and “are reassured the patient is not” carrying problematic NUDT15 or TMPT variants, “then I think you just go ahead and do full dose,” he said.
Testing for the relevant variants is available through the Mayo Clinic and several commercial labs.
“Even though the thiopurine dose is adjusted based on genotype, patients still need to be monitored closely for the development of thiopurine-induced myelosuppression as” TMPT and NUDT15 variants “do not explain all cases of” TIM, Marieke Coenen, PhD, an associate professor of pharmacogenetics at Radboud University Medical Center in Nijmegen, the Netherlands, cautioned in an editorial (Transl Gastroenterol Hepatol. 2019 Dec 12;4:81).
The prevalence of problematic NUDT15 variants is 0.7% among African and 20.7% among Hispanic people.
The work was funded by the National Institutes of Health, Crohn’s & Colitis UK, the Wellcome Trust, and others. Dr. Walker and other investigators reported numerous industry ties. Dr. Loftus is a consultant and/or has research funding from Abbott, Pfizer, and other companies. Dr. Coenen had no disclosures.
SOURCE: Walker GJ et al. JAMA. 2019 Feb 26;321(8):773-85.
MAUI, HAWAII – There’s a new kid on the block to worry about when it comes to thiopurine pharmacogenetics: Genetic variants in the thiopurine-metabolizing enzyme nudix hydrolase 15 have been linked to a markedly increased risk of thiopurine myelosuppression among inflammatory bowel disease patients.
The Food and Drug Administration and others already recommend screening for genetic variants in thiopurine methyltransferase (TPMT), another enzyme that metabolizes thiopurine. Polymorphisms lead to TMPT dysfunction, accumulation of cytotoxic metabolites, and increased risk of thiopurine-induced myelosuppression (TIM). Carriers are advised to use reduced doses with careful drug monitoring, or to skip thiopurines altogether.
A similar picture is emerging for nudix hydrolase 15 (NUDT15). It’s been known for several years that genetic variants are not uncommon among East Asian people and lead to TIM, but their prevalence and impact among people of European decent wasn’t clearly understood until now.
Investigators led by Gareth Walker, MBBS, of the Royal Devon and Exeter Hospital in Exeter, England, compared rates of problematic TMPT and NUDT15 variants among European inflammatory bowel disease patients who had developed TIM and those who had not, about 1,000 patients in all. The majority were on azathioprine and had Crohn’s disease. Finnish people were excluded because “their unique genetic background ... has led to the enrichment of some disease-causing gene variants and losses of others,” according to the study, which was published in JAMA.
Carriage of any of three coding NUDT15 variants greatly increased the risk of TIM (odds ratio, 27.3; 95% confidence interval, 9.3-116.7), independent of TPMT genotype and thiopurine dose. A particular variant – an in-frame deletion in NUDT15 – increased the risk 38-fold (95% CI, 5.1-286.1), and was carried by 5.8% of TIM patients.
The analysis also confirmed the importance of TPMT variants, which were found in 30.5% of TIM patients (95/311) versus 16.4% (100/608) of patients who did not develop TIM.
“Patients with variants of either NUDT15 or TPMT, or among those with variants of both genes, had a faster onset of TIM, more severe TIM, and had a greater need for granulocyte colony–stimulating factor rescue therapy. ... Our data suggest that pretreatment sequencing of the NUDT15 gene ... may be considered prior to initiation of thiopurine therapy,” the team concluded.
The prevalence of problematic NUDT15 variants among non-Finnish Europeans is about 1.6%, 6.9% among people from Finland, and almost 30% among East Asians. The team estimated NUDT15 would have to be genotyped in 95 non-Finnish Europeans to prevent one case of TIM; the number is 123 for TPMT. “Given the widespread use of thiopurines” – primarily in rheumatology and transplant medicine, in addition to gastroenterology – “these findings may have ramifications beyond the management of IBD,” the investigators wrote.
“I do think it’s worthwhile” to screen for NUDT15, said Edward Loftus, MD, a professor and consultant in the division of gastroenterology and hepatology at the Mayo Clinic in Rochester, Minn., who reviewed the study at the Gastroenterology Updates, IBD, Liver Disease Conference. “If you are a homozygote for this, your chance of getting profound leukopenia is very high, so I would probably not use a thiopurine.”
“If you are going to start low dose on everyone” with careful blood monitoring, “I suppose you could just do that, but I would say if you can get” the test and “are reassured the patient is not” carrying problematic NUDT15 or TMPT variants, “then I think you just go ahead and do full dose,” he said.
Testing for the relevant variants is available through the Mayo Clinic and several commercial labs.
“Even though the thiopurine dose is adjusted based on genotype, patients still need to be monitored closely for the development of thiopurine-induced myelosuppression as” TMPT and NUDT15 variants “do not explain all cases of” TIM, Marieke Coenen, PhD, an associate professor of pharmacogenetics at Radboud University Medical Center in Nijmegen, the Netherlands, cautioned in an editorial (Transl Gastroenterol Hepatol. 2019 Dec 12;4:81).
The prevalence of problematic NUDT15 variants is 0.7% among African and 20.7% among Hispanic people.
The work was funded by the National Institutes of Health, Crohn’s & Colitis UK, the Wellcome Trust, and others. Dr. Walker and other investigators reported numerous industry ties. Dr. Loftus is a consultant and/or has research funding from Abbott, Pfizer, and other companies. Dr. Coenen had no disclosures.
SOURCE: Walker GJ et al. JAMA. 2019 Feb 26;321(8):773-85.
MAUI, HAWAII – There’s a new kid on the block to worry about when it comes to thiopurine pharmacogenetics: Genetic variants in the thiopurine-metabolizing enzyme nudix hydrolase 15 have been linked to a markedly increased risk of thiopurine myelosuppression among inflammatory bowel disease patients.
The Food and Drug Administration and others already recommend screening for genetic variants in thiopurine methyltransferase (TPMT), another enzyme that metabolizes thiopurine. Polymorphisms lead to TMPT dysfunction, accumulation of cytotoxic metabolites, and increased risk of thiopurine-induced myelosuppression (TIM). Carriers are advised to use reduced doses with careful drug monitoring, or to skip thiopurines altogether.
A similar picture is emerging for nudix hydrolase 15 (NUDT15). It’s been known for several years that genetic variants are not uncommon among East Asian people and lead to TIM, but their prevalence and impact among people of European decent wasn’t clearly understood until now.
Investigators led by Gareth Walker, MBBS, of the Royal Devon and Exeter Hospital in Exeter, England, compared rates of problematic TMPT and NUDT15 variants among European inflammatory bowel disease patients who had developed TIM and those who had not, about 1,000 patients in all. The majority were on azathioprine and had Crohn’s disease. Finnish people were excluded because “their unique genetic background ... has led to the enrichment of some disease-causing gene variants and losses of others,” according to the study, which was published in JAMA.
Carriage of any of three coding NUDT15 variants greatly increased the risk of TIM (odds ratio, 27.3; 95% confidence interval, 9.3-116.7), independent of TPMT genotype and thiopurine dose. A particular variant – an in-frame deletion in NUDT15 – increased the risk 38-fold (95% CI, 5.1-286.1), and was carried by 5.8% of TIM patients.
The analysis also confirmed the importance of TPMT variants, which were found in 30.5% of TIM patients (95/311) versus 16.4% (100/608) of patients who did not develop TIM.
“Patients with variants of either NUDT15 or TPMT, or among those with variants of both genes, had a faster onset of TIM, more severe TIM, and had a greater need for granulocyte colony–stimulating factor rescue therapy. ... Our data suggest that pretreatment sequencing of the NUDT15 gene ... may be considered prior to initiation of thiopurine therapy,” the team concluded.
The prevalence of problematic NUDT15 variants among non-Finnish Europeans is about 1.6%, 6.9% among people from Finland, and almost 30% among East Asians. The team estimated NUDT15 would have to be genotyped in 95 non-Finnish Europeans to prevent one case of TIM; the number is 123 for TPMT. “Given the widespread use of thiopurines” – primarily in rheumatology and transplant medicine, in addition to gastroenterology – “these findings may have ramifications beyond the management of IBD,” the investigators wrote.
“I do think it’s worthwhile” to screen for NUDT15, said Edward Loftus, MD, a professor and consultant in the division of gastroenterology and hepatology at the Mayo Clinic in Rochester, Minn., who reviewed the study at the Gastroenterology Updates, IBD, Liver Disease Conference. “If you are a homozygote for this, your chance of getting profound leukopenia is very high, so I would probably not use a thiopurine.”
“If you are going to start low dose on everyone” with careful blood monitoring, “I suppose you could just do that, but I would say if you can get” the test and “are reassured the patient is not” carrying problematic NUDT15 or TMPT variants, “then I think you just go ahead and do full dose,” he said.
Testing for the relevant variants is available through the Mayo Clinic and several commercial labs.
“Even though the thiopurine dose is adjusted based on genotype, patients still need to be monitored closely for the development of thiopurine-induced myelosuppression as” TMPT and NUDT15 variants “do not explain all cases of” TIM, Marieke Coenen, PhD, an associate professor of pharmacogenetics at Radboud University Medical Center in Nijmegen, the Netherlands, cautioned in an editorial (Transl Gastroenterol Hepatol. 2019 Dec 12;4:81).
The prevalence of problematic NUDT15 variants is 0.7% among African and 20.7% among Hispanic people.
The work was funded by the National Institutes of Health, Crohn’s & Colitis UK, the Wellcome Trust, and others. Dr. Walker and other investigators reported numerous industry ties. Dr. Loftus is a consultant and/or has research funding from Abbott, Pfizer, and other companies. Dr. Coenen had no disclosures.
SOURCE: Walker GJ et al. JAMA. 2019 Feb 26;321(8):773-85.
REPORTING FROM GUILD 2020
RA magnifies fragility fracture risk in ESRD
MAUI, HAWAII – Comorbid rheumatoid arthritis is a force multiplier for fragility fracture risk in patients with end-stage renal disease, Renée Peterkin-McCalman, MD, reported at the 2020 Rheumatology Winter Clinical Symposium.
“Patients with RA and ESRD are at substantially increased risk of osteoporotic fragility fractures compared to the overall population of ESRD patients. So fracture prevention prior to initiation of dialysis should be a focus of care in patients with RA,” said Dr. Peterkin-McCalman, a rheumatology fellow at the Medical College of Georgia, Augusta.
She presented a retrospective cohort study of 10,706 adults who initiated hemodialysis or peritoneal dialysis for ESRD during 2005-2008, including 1,040 who also had RA. All subjects were drawn from the United States Renal Data System. The impetus for the study, Dr. Peterkin-McCalman explained in an interview, was that although prior studies have established that RA and ESRD are independent risk factors for osteoporotic fractures, the interplay between the two was previously unknown.
The risk of incident osteoporotic fractures during the first 3 years after going on renal dialysis was 14.7% in patients with ESRD only, vaulting to 25.6% in those with comorbid RA. Individuals with both RA and ESRD were at an adjusted 1.83-fold increased overall risk for new fragility fractures and at 1.85-fold increased risk for hip fracture, compared to those without RA.
Far and away the strongest risk factor for incident osteoporotic fractures in the group with RA plus ESRD was a history of a fracture sustained within 5 years prior to initiation of dialysis, with an associated 11.5-fold increased fracture risk overall and an 8.2-fold increased risk of hip fracture.
“The reason that’s important is we don’t really have any medications to reduce fracture risk once you get to ESRD. Of course, we have bisphosphonates and Prolia (denosumab) and things like that, but that’s in patients with milder CKD [chronic kidney disease] or no renal disease at all. So the goal is to identify the patients early who are at higher risk so that we can protect those bones before they get to ESRD and we have nothing left to treat them with,” she said.
In addition to a history of prevalent fracture prior to starting ESRD, the other risk factors for fracture in patients with ESRD and comorbid RA Dr. Peterkin-McCalman identified in her study included age greater than 50 years at the start of dialysis and female gender, which was associated with a twofold greater fracture risk than in men. Black patients with ESRD and RA were 64% less likely than whites to experience an incident fragility fracture. And the fracture risk was higher in patients on hemodialysis than with peritoneal dialysis.
Her study was supported by the Medical College of Georgia and a research grant from Dialysis Clinic Inc.
SOURCE: Peterkin-McCalman R et al. RWCS 2020.
MAUI, HAWAII – Comorbid rheumatoid arthritis is a force multiplier for fragility fracture risk in patients with end-stage renal disease, Renée Peterkin-McCalman, MD, reported at the 2020 Rheumatology Winter Clinical Symposium.
“Patients with RA and ESRD are at substantially increased risk of osteoporotic fragility fractures compared to the overall population of ESRD patients. So fracture prevention prior to initiation of dialysis should be a focus of care in patients with RA,” said Dr. Peterkin-McCalman, a rheumatology fellow at the Medical College of Georgia, Augusta.
She presented a retrospective cohort study of 10,706 adults who initiated hemodialysis or peritoneal dialysis for ESRD during 2005-2008, including 1,040 who also had RA. All subjects were drawn from the United States Renal Data System. The impetus for the study, Dr. Peterkin-McCalman explained in an interview, was that although prior studies have established that RA and ESRD are independent risk factors for osteoporotic fractures, the interplay between the two was previously unknown.
The risk of incident osteoporotic fractures during the first 3 years after going on renal dialysis was 14.7% in patients with ESRD only, vaulting to 25.6% in those with comorbid RA. Individuals with both RA and ESRD were at an adjusted 1.83-fold increased overall risk for new fragility fractures and at 1.85-fold increased risk for hip fracture, compared to those without RA.
Far and away the strongest risk factor for incident osteoporotic fractures in the group with RA plus ESRD was a history of a fracture sustained within 5 years prior to initiation of dialysis, with an associated 11.5-fold increased fracture risk overall and an 8.2-fold increased risk of hip fracture.
“The reason that’s important is we don’t really have any medications to reduce fracture risk once you get to ESRD. Of course, we have bisphosphonates and Prolia (denosumab) and things like that, but that’s in patients with milder CKD [chronic kidney disease] or no renal disease at all. So the goal is to identify the patients early who are at higher risk so that we can protect those bones before they get to ESRD and we have nothing left to treat them with,” she said.
In addition to a history of prevalent fracture prior to starting ESRD, the other risk factors for fracture in patients with ESRD and comorbid RA Dr. Peterkin-McCalman identified in her study included age greater than 50 years at the start of dialysis and female gender, which was associated with a twofold greater fracture risk than in men. Black patients with ESRD and RA were 64% less likely than whites to experience an incident fragility fracture. And the fracture risk was higher in patients on hemodialysis than with peritoneal dialysis.
Her study was supported by the Medical College of Georgia and a research grant from Dialysis Clinic Inc.
SOURCE: Peterkin-McCalman R et al. RWCS 2020.
MAUI, HAWAII – Comorbid rheumatoid arthritis is a force multiplier for fragility fracture risk in patients with end-stage renal disease, Renée Peterkin-McCalman, MD, reported at the 2020 Rheumatology Winter Clinical Symposium.
“Patients with RA and ESRD are at substantially increased risk of osteoporotic fragility fractures compared to the overall population of ESRD patients. So fracture prevention prior to initiation of dialysis should be a focus of care in patients with RA,” said Dr. Peterkin-McCalman, a rheumatology fellow at the Medical College of Georgia, Augusta.
She presented a retrospective cohort study of 10,706 adults who initiated hemodialysis or peritoneal dialysis for ESRD during 2005-2008, including 1,040 who also had RA. All subjects were drawn from the United States Renal Data System. The impetus for the study, Dr. Peterkin-McCalman explained in an interview, was that although prior studies have established that RA and ESRD are independent risk factors for osteoporotic fractures, the interplay between the two was previously unknown.
The risk of incident osteoporotic fractures during the first 3 years after going on renal dialysis was 14.7% in patients with ESRD only, vaulting to 25.6% in those with comorbid RA. Individuals with both RA and ESRD were at an adjusted 1.83-fold increased overall risk for new fragility fractures and at 1.85-fold increased risk for hip fracture, compared to those without RA.
Far and away the strongest risk factor for incident osteoporotic fractures in the group with RA plus ESRD was a history of a fracture sustained within 5 years prior to initiation of dialysis, with an associated 11.5-fold increased fracture risk overall and an 8.2-fold increased risk of hip fracture.
“The reason that’s important is we don’t really have any medications to reduce fracture risk once you get to ESRD. Of course, we have bisphosphonates and Prolia (denosumab) and things like that, but that’s in patients with milder CKD [chronic kidney disease] or no renal disease at all. So the goal is to identify the patients early who are at higher risk so that we can protect those bones before they get to ESRD and we have nothing left to treat them with,” she said.
In addition to a history of prevalent fracture prior to starting ESRD, the other risk factors for fracture in patients with ESRD and comorbid RA Dr. Peterkin-McCalman identified in her study included age greater than 50 years at the start of dialysis and female gender, which was associated with a twofold greater fracture risk than in men. Black patients with ESRD and RA were 64% less likely than whites to experience an incident fragility fracture. And the fracture risk was higher in patients on hemodialysis than with peritoneal dialysis.
Her study was supported by the Medical College of Georgia and a research grant from Dialysis Clinic Inc.
SOURCE: Peterkin-McCalman R et al. RWCS 2020.
REPORTING FROM RWCS 2020
Meta-analysis highlights safety concerns with interleukin inhibition
MAUI, HAWAII – The use of interleukin inhibitors for treatment of rheumatologic diseases doubles a patient’s risk of serious infections, according to a comprehensive systematic review and meta-analysis of 74 randomized, placebo-controlled trials presented by Jawad Bilal, MBBS, at the 2020 Rheumatology Winter Clinical Symposium.
The meta-analysis, which incorporated 29,214 patients with a variety of rheumatic diseases, demonstrated that patients receiving interleukin (IL) inhibitors had a 1.97-fold increased risk of serious infections, a finding accompanied by a high degree of statistical certainty. The number-needed-to-harm was 67 patients treated for a median of 24 weeks in order to generate one additional serious infection.
“That number-needed-to-harm is a significant finding because having a serious infection means by definition you’re getting admitted to the hospital and receiving IV antibiotics,” Dr. Bilal observed in an interview.
The meta-analysis also found that IL inhibition was associated with a 2.35-fold increased risk of opportunistic infections and a 1.52-fold higher risk of developing cancer, both findings with statistical significance (P =.03) but only moderate certainty because fewer of those events were captured in the trials compared to the numbers of serious infections, explained Dr. Bilal of the University of Arizona, Tucson.
For opportunistic infections, the number-needed-to-harm was 250 patients treated with an IL inhibitor for a median of 54 weeks in order to result in one additional opportunistic infection. For cancer, the number-needed-to-harm was 250 for a median of 24 weeks.
Dr. Bilal noted that while the IL inhibitors are drugs of established efficacy in rheumatologic diseases, their safety has not previously undergone anything approaching the comprehensive scrutiny carried out in this meta-analysis. The meta-analysis, which included all published placebo-controlled randomized trials and their extension studies, employed rigorous methodology in accord with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) statement and the GRADE approach to data analysis. Studies of IL inhibitors in patients with dermatologic and GI diseases were excluded from the meta-analysis.
He offered a caveat regarding the cancer risk findings: “Our analysis showed that the cancer risk is increased, but the results are not conclusive because we only had a few years of data. With cancer, you really need at least 8-10 years of data. So the real-world experience with the interleukin inhibitors in the large registries is what’s going to tell if the cancer risk is really increased or not. In the meantime, we all have to be cautious.”
The number of serious infections collected in the meta-analysis afforded sufficient statistical power for the investigators to break down differential risks based on individual drugs and indications. Among the drugs associated with significantly increased risk of serious infections were anakinra, with an odds ratio of 2.67, compared with placebo; secukinumab with an OR of 2.43; and tocilizumab with an OR or 1.76. Ustekinumab and ixekizumab were associated with 2.57- and 3.89-fold increased risks, respectively, but the number of rheumatology patients treated with those two biologics wasn’t large enough for those findings to achieve statistical significance.
Rheumatoid arthritis patients who received an IL inhibitor rather than placebo had a 1.98-fold increased risk of serious infection, while those with psoriatic arthritis had a 2.21-fold increased risk. Patients treated for SLE had a 6.44-fold increased risk, and those with juvenile idiopathic arthritis had a 5.37-fold higher risk, but the margins for error were such that those results weren’t statistically significant.
“I think this study is going to help clinicians and patients when they’re trying to weigh the risks and benefits of IL inhibitors, especially if they already have risk factors, like a recent history of serious infection or a history of cancer or of opportunistic infection,” Dr. Bilal commented.
A study limitation was that he and his coinvestigators had to lump together the various IL inhibitors in order to gain statistical power, even though the drugs work differently, he noted.
Dr. Bilal reported having no financial conflicts regarding his study, the full details of which have been published (JAMA Netw Open. 2019 Oct 2. doi: 10.1001/jamanetworkopen.2019.13102).
MAUI, HAWAII – The use of interleukin inhibitors for treatment of rheumatologic diseases doubles a patient’s risk of serious infections, according to a comprehensive systematic review and meta-analysis of 74 randomized, placebo-controlled trials presented by Jawad Bilal, MBBS, at the 2020 Rheumatology Winter Clinical Symposium.
The meta-analysis, which incorporated 29,214 patients with a variety of rheumatic diseases, demonstrated that patients receiving interleukin (IL) inhibitors had a 1.97-fold increased risk of serious infections, a finding accompanied by a high degree of statistical certainty. The number-needed-to-harm was 67 patients treated for a median of 24 weeks in order to generate one additional serious infection.
“That number-needed-to-harm is a significant finding because having a serious infection means by definition you’re getting admitted to the hospital and receiving IV antibiotics,” Dr. Bilal observed in an interview.
The meta-analysis also found that IL inhibition was associated with a 2.35-fold increased risk of opportunistic infections and a 1.52-fold higher risk of developing cancer, both findings with statistical significance (P =.03) but only moderate certainty because fewer of those events were captured in the trials compared to the numbers of serious infections, explained Dr. Bilal of the University of Arizona, Tucson.
For opportunistic infections, the number-needed-to-harm was 250 patients treated with an IL inhibitor for a median of 54 weeks in order to result in one additional opportunistic infection. For cancer, the number-needed-to-harm was 250 for a median of 24 weeks.
Dr. Bilal noted that while the IL inhibitors are drugs of established efficacy in rheumatologic diseases, their safety has not previously undergone anything approaching the comprehensive scrutiny carried out in this meta-analysis. The meta-analysis, which included all published placebo-controlled randomized trials and their extension studies, employed rigorous methodology in accord with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) statement and the GRADE approach to data analysis. Studies of IL inhibitors in patients with dermatologic and GI diseases were excluded from the meta-analysis.
He offered a caveat regarding the cancer risk findings: “Our analysis showed that the cancer risk is increased, but the results are not conclusive because we only had a few years of data. With cancer, you really need at least 8-10 years of data. So the real-world experience with the interleukin inhibitors in the large registries is what’s going to tell if the cancer risk is really increased or not. In the meantime, we all have to be cautious.”
The number of serious infections collected in the meta-analysis afforded sufficient statistical power for the investigators to break down differential risks based on individual drugs and indications. Among the drugs associated with significantly increased risk of serious infections were anakinra, with an odds ratio of 2.67, compared with placebo; secukinumab with an OR of 2.43; and tocilizumab with an OR or 1.76. Ustekinumab and ixekizumab were associated with 2.57- and 3.89-fold increased risks, respectively, but the number of rheumatology patients treated with those two biologics wasn’t large enough for those findings to achieve statistical significance.
Rheumatoid arthritis patients who received an IL inhibitor rather than placebo had a 1.98-fold increased risk of serious infection, while those with psoriatic arthritis had a 2.21-fold increased risk. Patients treated for SLE had a 6.44-fold increased risk, and those with juvenile idiopathic arthritis had a 5.37-fold higher risk, but the margins for error were such that those results weren’t statistically significant.
“I think this study is going to help clinicians and patients when they’re trying to weigh the risks and benefits of IL inhibitors, especially if they already have risk factors, like a recent history of serious infection or a history of cancer or of opportunistic infection,” Dr. Bilal commented.
A study limitation was that he and his coinvestigators had to lump together the various IL inhibitors in order to gain statistical power, even though the drugs work differently, he noted.
Dr. Bilal reported having no financial conflicts regarding his study, the full details of which have been published (JAMA Netw Open. 2019 Oct 2. doi: 10.1001/jamanetworkopen.2019.13102).
MAUI, HAWAII – The use of interleukin inhibitors for treatment of rheumatologic diseases doubles a patient’s risk of serious infections, according to a comprehensive systematic review and meta-analysis of 74 randomized, placebo-controlled trials presented by Jawad Bilal, MBBS, at the 2020 Rheumatology Winter Clinical Symposium.
The meta-analysis, which incorporated 29,214 patients with a variety of rheumatic diseases, demonstrated that patients receiving interleukin (IL) inhibitors had a 1.97-fold increased risk of serious infections, a finding accompanied by a high degree of statistical certainty. The number-needed-to-harm was 67 patients treated for a median of 24 weeks in order to generate one additional serious infection.
“That number-needed-to-harm is a significant finding because having a serious infection means by definition you’re getting admitted to the hospital and receiving IV antibiotics,” Dr. Bilal observed in an interview.
The meta-analysis also found that IL inhibition was associated with a 2.35-fold increased risk of opportunistic infections and a 1.52-fold higher risk of developing cancer, both findings with statistical significance (P =.03) but only moderate certainty because fewer of those events were captured in the trials compared to the numbers of serious infections, explained Dr. Bilal of the University of Arizona, Tucson.
For opportunistic infections, the number-needed-to-harm was 250 patients treated with an IL inhibitor for a median of 54 weeks in order to result in one additional opportunistic infection. For cancer, the number-needed-to-harm was 250 for a median of 24 weeks.
Dr. Bilal noted that while the IL inhibitors are drugs of established efficacy in rheumatologic diseases, their safety has not previously undergone anything approaching the comprehensive scrutiny carried out in this meta-analysis. The meta-analysis, which included all published placebo-controlled randomized trials and their extension studies, employed rigorous methodology in accord with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) statement and the GRADE approach to data analysis. Studies of IL inhibitors in patients with dermatologic and GI diseases were excluded from the meta-analysis.
He offered a caveat regarding the cancer risk findings: “Our analysis showed that the cancer risk is increased, but the results are not conclusive because we only had a few years of data. With cancer, you really need at least 8-10 years of data. So the real-world experience with the interleukin inhibitors in the large registries is what’s going to tell if the cancer risk is really increased or not. In the meantime, we all have to be cautious.”
The number of serious infections collected in the meta-analysis afforded sufficient statistical power for the investigators to break down differential risks based on individual drugs and indications. Among the drugs associated with significantly increased risk of serious infections were anakinra, with an odds ratio of 2.67, compared with placebo; secukinumab with an OR of 2.43; and tocilizumab with an OR or 1.76. Ustekinumab and ixekizumab were associated with 2.57- and 3.89-fold increased risks, respectively, but the number of rheumatology patients treated with those two biologics wasn’t large enough for those findings to achieve statistical significance.
Rheumatoid arthritis patients who received an IL inhibitor rather than placebo had a 1.98-fold increased risk of serious infection, while those with psoriatic arthritis had a 2.21-fold increased risk. Patients treated for SLE had a 6.44-fold increased risk, and those with juvenile idiopathic arthritis had a 5.37-fold higher risk, but the margins for error were such that those results weren’t statistically significant.
“I think this study is going to help clinicians and patients when they’re trying to weigh the risks and benefits of IL inhibitors, especially if they already have risk factors, like a recent history of serious infection or a history of cancer or of opportunistic infection,” Dr. Bilal commented.
A study limitation was that he and his coinvestigators had to lump together the various IL inhibitors in order to gain statistical power, even though the drugs work differently, he noted.
Dr. Bilal reported having no financial conflicts regarding his study, the full details of which have been published (JAMA Netw Open. 2019 Oct 2. doi: 10.1001/jamanetworkopen.2019.13102).
REPORTING FROM RWCS 2020