User login
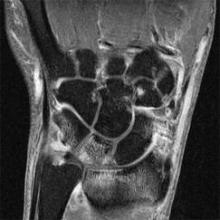
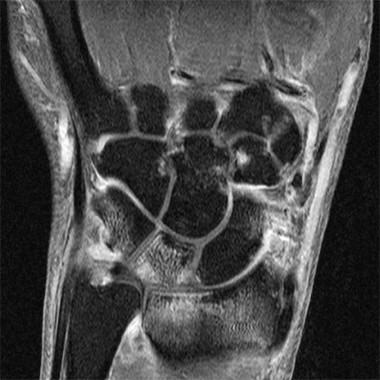
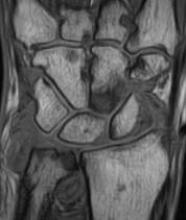
MRI detects high level of subclinical small joint inflammation
MADRID – A high percentage of patients with early arthritis have inflammation of the small joints that can be detected with MRI but not by physical examination.
Results of a cross-sectional study, presented by Dr. Annemarie Krabben at the annual European Congress of Rheumatology, found that 66% of wrist, 27% of metacarpophalangeal (MCP), and 13% of metatarsophalangeal (MTP) joints that were not clinically swollen showed signs of inflammation on MRI. However, inflammation on MRI was present in 92% of wrists, 86% of MCP, and 29% of MTP joints that were clinically swollen.
"You would expect that inflammation on MRI would be present in the clinically swollen joints, but we also saw inflammation in the non-swollen joints," explained Dr. Krabben of Leiden University Medical Center in the Netherlands.
Furthermore, "when you look at the joints with MRI-detected inflammation, a lot of these didn’t have clinical inflammation," she added.
Clinical joint swelling was absent but signs of bone marrow edema were detected on MRI in 60% of wrist, 53% of MCP, and 78% of MTP joints. If severe MRI-detected edema was considered, joint swelling was absent in 35%, 39%, and 58% of wrist, MCP, and MTP joints, respectively. Joints without clinical swelling showed signs of inflammation on MRI in 61% of wrist, 64% of MCP, and 77% of MTP joints.
The study involved patients with early arthritis who were part of the Leiden Early Arthritis Clinic cohort. This cohort was established in 1993 to detect and treat inflammatory disorders early in the disease state (Rheumatology [Oxford] 2011;50:93-100).
Upon entry into the cohort, patients underwent a physical examination that included 68 tender and 66 swollen joint counts and 1.5 Tesla MRI of the wrist, MCP, and MTP joints. The latter were used to determine the presence and extent of synovitis, bone marrow edema, and tenosynovitis.
In total, 1,790 small joints were examined in 179 patients who had a median duration of symptoms of 15 weeks. Overall, 30% of wrist, 15% of MCP, and 11% of MTP joints were swollen at physical examination and the majority also showed inflammation on MRI.
"There was a lot of subclinical inflammation, especially bone marrow edema, in the nonswollen joints," Dr. Krabben said. Bone marrow edema is linked to erosive disease progression, she observed and suggested that the next step is to see what happens to patients with subclinical inflammation at baseline, and whether this will eventually progress to erosive disease.
The study was supported by the Dutch Arthritis Foundation (Reumafonds), the Netherlands Organization for Health Research and Development, and the Center for Translational Molecular Medicine. Dr. Krabben has received research funding from Reumafonds.
MADRID – A high percentage of patients with early arthritis have inflammation of the small joints that can be detected with MRI but not by physical examination.
Results of a cross-sectional study, presented by Dr. Annemarie Krabben at the annual European Congress of Rheumatology, found that 66% of wrist, 27% of metacarpophalangeal (MCP), and 13% of metatarsophalangeal (MTP) joints that were not clinically swollen showed signs of inflammation on MRI. However, inflammation on MRI was present in 92% of wrists, 86% of MCP, and 29% of MTP joints that were clinically swollen.
"You would expect that inflammation on MRI would be present in the clinically swollen joints, but we also saw inflammation in the non-swollen joints," explained Dr. Krabben of Leiden University Medical Center in the Netherlands.
Furthermore, "when you look at the joints with MRI-detected inflammation, a lot of these didn’t have clinical inflammation," she added.
Clinical joint swelling was absent but signs of bone marrow edema were detected on MRI in 60% of wrist, 53% of MCP, and 78% of MTP joints. If severe MRI-detected edema was considered, joint swelling was absent in 35%, 39%, and 58% of wrist, MCP, and MTP joints, respectively. Joints without clinical swelling showed signs of inflammation on MRI in 61% of wrist, 64% of MCP, and 77% of MTP joints.
The study involved patients with early arthritis who were part of the Leiden Early Arthritis Clinic cohort. This cohort was established in 1993 to detect and treat inflammatory disorders early in the disease state (Rheumatology [Oxford] 2011;50:93-100).
Upon entry into the cohort, patients underwent a physical examination that included 68 tender and 66 swollen joint counts and 1.5 Tesla MRI of the wrist, MCP, and MTP joints. The latter were used to determine the presence and extent of synovitis, bone marrow edema, and tenosynovitis.
In total, 1,790 small joints were examined in 179 patients who had a median duration of symptoms of 15 weeks. Overall, 30% of wrist, 15% of MCP, and 11% of MTP joints were swollen at physical examination and the majority also showed inflammation on MRI.
"There was a lot of subclinical inflammation, especially bone marrow edema, in the nonswollen joints," Dr. Krabben said. Bone marrow edema is linked to erosive disease progression, she observed and suggested that the next step is to see what happens to patients with subclinical inflammation at baseline, and whether this will eventually progress to erosive disease.
The study was supported by the Dutch Arthritis Foundation (Reumafonds), the Netherlands Organization for Health Research and Development, and the Center for Translational Molecular Medicine. Dr. Krabben has received research funding from Reumafonds.
MADRID – A high percentage of patients with early arthritis have inflammation of the small joints that can be detected with MRI but not by physical examination.
Results of a cross-sectional study, presented by Dr. Annemarie Krabben at the annual European Congress of Rheumatology, found that 66% of wrist, 27% of metacarpophalangeal (MCP), and 13% of metatarsophalangeal (MTP) joints that were not clinically swollen showed signs of inflammation on MRI. However, inflammation on MRI was present in 92% of wrists, 86% of MCP, and 29% of MTP joints that were clinically swollen.
"You would expect that inflammation on MRI would be present in the clinically swollen joints, but we also saw inflammation in the non-swollen joints," explained Dr. Krabben of Leiden University Medical Center in the Netherlands.
Furthermore, "when you look at the joints with MRI-detected inflammation, a lot of these didn’t have clinical inflammation," she added.
Clinical joint swelling was absent but signs of bone marrow edema were detected on MRI in 60% of wrist, 53% of MCP, and 78% of MTP joints. If severe MRI-detected edema was considered, joint swelling was absent in 35%, 39%, and 58% of wrist, MCP, and MTP joints, respectively. Joints without clinical swelling showed signs of inflammation on MRI in 61% of wrist, 64% of MCP, and 77% of MTP joints.
The study involved patients with early arthritis who were part of the Leiden Early Arthritis Clinic cohort. This cohort was established in 1993 to detect and treat inflammatory disorders early in the disease state (Rheumatology [Oxford] 2011;50:93-100).
Upon entry into the cohort, patients underwent a physical examination that included 68 tender and 66 swollen joint counts and 1.5 Tesla MRI of the wrist, MCP, and MTP joints. The latter were used to determine the presence and extent of synovitis, bone marrow edema, and tenosynovitis.
In total, 1,790 small joints were examined in 179 patients who had a median duration of symptoms of 15 weeks. Overall, 30% of wrist, 15% of MCP, and 11% of MTP joints were swollen at physical examination and the majority also showed inflammation on MRI.
"There was a lot of subclinical inflammation, especially bone marrow edema, in the nonswollen joints," Dr. Krabben said. Bone marrow edema is linked to erosive disease progression, she observed and suggested that the next step is to see what happens to patients with subclinical inflammation at baseline, and whether this will eventually progress to erosive disease.
The study was supported by the Dutch Arthritis Foundation (Reumafonds), the Netherlands Organization for Health Research and Development, and the Center for Translational Molecular Medicine. Dr. Krabben has received research funding from Reumafonds.
AT THE EULAR CONGRESS 2013
Abatacept, adalimumab equivalent for RA in 2-year head-to-head trial
MADRID – A 2-year head-to-head comparison of abatacept and adalimumab in rheumatoid arthritis patients who were on background methotrexate has found equal improvement with both biologics, according to results from a study presented at the annual European Congress of Rheumatology.
The randomized, investigator-blinded AMPLE trial is the first 2-year comparator study of biologics done in biologic-naive rheumatoid arthritis patients.
"Through 2 years of treatment, in this first active comparator study between biologic agents in rheumatoid arthritis patients with an inadequate response to methotrexate, this robust data set demonstrates that subcutaneous abatacept and adalimumab were equally efficacious in clinical, functional, and radiographic outcomes," said Dr. Michael H. Schiff, a professor of medicine at the University of Colorado, Denver.
Researchers recruited 646 patients with active RA and an inadequate response to methotrexate, and randomized them to either 125 mg of abatacept weekly (without an IV load) or 40 mg of adalimumab biweekly, with a stable dose of methotrexate.
The data show that both agents have an excellent retention rate, with 79% of the abatacept and 65% of the adalimumab groups completing the 2-year follow-up.
The two medications showed similar efficacy for American College of Rheumatology (ACR) 20, 50, 70, and 90 responses and rates of remission on the Disease Activity Score-28 (DAS28), Dr. Schiff said. For ACR 20, the 2-year response rate was 60% in each group. The ACR 50 response rate was 47% for the adalimumab group and 45% for the abatacept group. For the ACR 70, the rates were 29% for adalimumab and 31% for abatacept, and for the ACR 90, the rates were 8% for adalimumab and 15% for abatacept.
The 2-year DAS28 rate was virtually identical in each group, with a mean decrease of about 2.2 from baseline. X-ray non-progression was seen in 84% of each group at 2 years, Dr. Schiff said.
The study found similar numbers of serious adverse events in both arms (14% of the abatacept group and 17% of the adalimumab group). However, serious adverse events leading to discontinuation of the study medication occurred in 5% of patients taking adalimumab and 2% of those taking abatacept.
There was one death in each group – neither of which was related to the study drugs. There were seven malignancies in each group; four patients in each group discontinued their study medication due to neoplasm.
Infections were the most common serious side effects (31 total), occurring in 4% of the abatacept and 6% of the adalimumab groups). There were eight opportunistic infections – four in each group. The adalimumab group had two cases of pulmonary tuberculosis, one case of disseminated tuberculosis, and one case of disseminated histoplasmosis. There were three cases of pneumonia in the abatacept group and four in the adalimumab group.
Autoimmune events were also relatively common – 18 in all, with 12 in the abatacept group (4%) and 6 in the adalimumab group (2%). Dr. Schiff said none of these were serious or clinically important.
During the question-and-answer period, Dr. Schiff said it’s not currently possible to predict which patients would respond to the drugs. "We looked at responders in both groups and were not able to differentiate them based on clinical characteristics," he said. "We are now analyzing the biomarkers and hope to have that information for EULAR 2014."
"EULAR/ACR guidelines recommend starting a patient on methotrexate and then optimizing the dose over 3-6 months, and if a patient has an incomplete response to methotrexate, then to add a biological agent," said Dr. Schiff in an interview.
He noted that anti-tumor necrosis factor (anti-TNF) agents have been the first choice of most rheumatologists, and adalimumab is the most widely chosen anti-TNF agent worldwide, which is why it was selected as one of the agents for the head-to-head trial. Abatacept employs another method of action: T-cell inhibition.
"This paper has important clinical significance because a patient and his or her rheumatologist want to have data to make an informed choice of a biologic agent to add when an incomplete response to methotrexate occurs," Dr. Schiff said.
Senior writer Michele Sullivan contributed to this report.
MADRID – A 2-year head-to-head comparison of abatacept and adalimumab in rheumatoid arthritis patients who were on background methotrexate has found equal improvement with both biologics, according to results from a study presented at the annual European Congress of Rheumatology.
The randomized, investigator-blinded AMPLE trial is the first 2-year comparator study of biologics done in biologic-naive rheumatoid arthritis patients.
"Through 2 years of treatment, in this first active comparator study between biologic agents in rheumatoid arthritis patients with an inadequate response to methotrexate, this robust data set demonstrates that subcutaneous abatacept and adalimumab were equally efficacious in clinical, functional, and radiographic outcomes," said Dr. Michael H. Schiff, a professor of medicine at the University of Colorado, Denver.
Researchers recruited 646 patients with active RA and an inadequate response to methotrexate, and randomized them to either 125 mg of abatacept weekly (without an IV load) or 40 mg of adalimumab biweekly, with a stable dose of methotrexate.
The data show that both agents have an excellent retention rate, with 79% of the abatacept and 65% of the adalimumab groups completing the 2-year follow-up.
The two medications showed similar efficacy for American College of Rheumatology (ACR) 20, 50, 70, and 90 responses and rates of remission on the Disease Activity Score-28 (DAS28), Dr. Schiff said. For ACR 20, the 2-year response rate was 60% in each group. The ACR 50 response rate was 47% for the adalimumab group and 45% for the abatacept group. For the ACR 70, the rates were 29% for adalimumab and 31% for abatacept, and for the ACR 90, the rates were 8% for adalimumab and 15% for abatacept.
The 2-year DAS28 rate was virtually identical in each group, with a mean decrease of about 2.2 from baseline. X-ray non-progression was seen in 84% of each group at 2 years, Dr. Schiff said.
The study found similar numbers of serious adverse events in both arms (14% of the abatacept group and 17% of the adalimumab group). However, serious adverse events leading to discontinuation of the study medication occurred in 5% of patients taking adalimumab and 2% of those taking abatacept.
There was one death in each group – neither of which was related to the study drugs. There were seven malignancies in each group; four patients in each group discontinued their study medication due to neoplasm.
Infections were the most common serious side effects (31 total), occurring in 4% of the abatacept and 6% of the adalimumab groups). There were eight opportunistic infections – four in each group. The adalimumab group had two cases of pulmonary tuberculosis, one case of disseminated tuberculosis, and one case of disseminated histoplasmosis. There were three cases of pneumonia in the abatacept group and four in the adalimumab group.
Autoimmune events were also relatively common – 18 in all, with 12 in the abatacept group (4%) and 6 in the adalimumab group (2%). Dr. Schiff said none of these were serious or clinically important.
During the question-and-answer period, Dr. Schiff said it’s not currently possible to predict which patients would respond to the drugs. "We looked at responders in both groups and were not able to differentiate them based on clinical characteristics," he said. "We are now analyzing the biomarkers and hope to have that information for EULAR 2014."
"EULAR/ACR guidelines recommend starting a patient on methotrexate and then optimizing the dose over 3-6 months, and if a patient has an incomplete response to methotrexate, then to add a biological agent," said Dr. Schiff in an interview.
He noted that anti-tumor necrosis factor (anti-TNF) agents have been the first choice of most rheumatologists, and adalimumab is the most widely chosen anti-TNF agent worldwide, which is why it was selected as one of the agents for the head-to-head trial. Abatacept employs another method of action: T-cell inhibition.
"This paper has important clinical significance because a patient and his or her rheumatologist want to have data to make an informed choice of a biologic agent to add when an incomplete response to methotrexate occurs," Dr. Schiff said.
Senior writer Michele Sullivan contributed to this report.
MADRID – A 2-year head-to-head comparison of abatacept and adalimumab in rheumatoid arthritis patients who were on background methotrexate has found equal improvement with both biologics, according to results from a study presented at the annual European Congress of Rheumatology.
The randomized, investigator-blinded AMPLE trial is the first 2-year comparator study of biologics done in biologic-naive rheumatoid arthritis patients.
"Through 2 years of treatment, in this first active comparator study between biologic agents in rheumatoid arthritis patients with an inadequate response to methotrexate, this robust data set demonstrates that subcutaneous abatacept and adalimumab were equally efficacious in clinical, functional, and radiographic outcomes," said Dr. Michael H. Schiff, a professor of medicine at the University of Colorado, Denver.
Researchers recruited 646 patients with active RA and an inadequate response to methotrexate, and randomized them to either 125 mg of abatacept weekly (without an IV load) or 40 mg of adalimumab biweekly, with a stable dose of methotrexate.
The data show that both agents have an excellent retention rate, with 79% of the abatacept and 65% of the adalimumab groups completing the 2-year follow-up.
The two medications showed similar efficacy for American College of Rheumatology (ACR) 20, 50, 70, and 90 responses and rates of remission on the Disease Activity Score-28 (DAS28), Dr. Schiff said. For ACR 20, the 2-year response rate was 60% in each group. The ACR 50 response rate was 47% for the adalimumab group and 45% for the abatacept group. For the ACR 70, the rates were 29% for adalimumab and 31% for abatacept, and for the ACR 90, the rates were 8% for adalimumab and 15% for abatacept.
The 2-year DAS28 rate was virtually identical in each group, with a mean decrease of about 2.2 from baseline. X-ray non-progression was seen in 84% of each group at 2 years, Dr. Schiff said.
The study found similar numbers of serious adverse events in both arms (14% of the abatacept group and 17% of the adalimumab group). However, serious adverse events leading to discontinuation of the study medication occurred in 5% of patients taking adalimumab and 2% of those taking abatacept.
There was one death in each group – neither of which was related to the study drugs. There were seven malignancies in each group; four patients in each group discontinued their study medication due to neoplasm.
Infections were the most common serious side effects (31 total), occurring in 4% of the abatacept and 6% of the adalimumab groups). There were eight opportunistic infections – four in each group. The adalimumab group had two cases of pulmonary tuberculosis, one case of disseminated tuberculosis, and one case of disseminated histoplasmosis. There were three cases of pneumonia in the abatacept group and four in the adalimumab group.
Autoimmune events were also relatively common – 18 in all, with 12 in the abatacept group (4%) and 6 in the adalimumab group (2%). Dr. Schiff said none of these were serious or clinically important.
During the question-and-answer period, Dr. Schiff said it’s not currently possible to predict which patients would respond to the drugs. "We looked at responders in both groups and were not able to differentiate them based on clinical characteristics," he said. "We are now analyzing the biomarkers and hope to have that information for EULAR 2014."
"EULAR/ACR guidelines recommend starting a patient on methotrexate and then optimizing the dose over 3-6 months, and if a patient has an incomplete response to methotrexate, then to add a biological agent," said Dr. Schiff in an interview.
He noted that anti-tumor necrosis factor (anti-TNF) agents have been the first choice of most rheumatologists, and adalimumab is the most widely chosen anti-TNF agent worldwide, which is why it was selected as one of the agents for the head-to-head trial. Abatacept employs another method of action: T-cell inhibition.
"This paper has important clinical significance because a patient and his or her rheumatologist want to have data to make an informed choice of a biologic agent to add when an incomplete response to methotrexate occurs," Dr. Schiff said.
Senior writer Michele Sullivan contributed to this report.
AT THE EULAR CONGRESS 2013
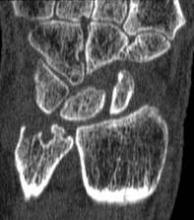
MRI score of joint narrowing has research promise
A magnetic resonance imaging scoring system of joint-space narrowing in rheumatoid arthritis showed "a very high" agreement with computed tomography scores and may become a useful tool in rheumatoid arthritis clinical trials after further validation, judging from data presented by Dr. Uffe Møller Døhn.
In a small study, which was conducted to validate the OMERACT-RAMRIS MRI JSN scoring system in the wrists and metacarpophalangeal (MCP) joints, there was a very high agreement between the joint-space narrowing scores on MRI and CT and moderate agreement between scores on MRI and x-ray, said Dr. Møller Døhn of Copenhagen University Hospital at Glostrup at the annual European Congress of Rheumatology. In addition, there was "high to very high" inter- and intrareader reliability, particularly for the wrist joints.
An OMERACT (Outcome Measures in Rheumatology) initiative, this scoring system is being developed to provide a more precise and sensitive method of measuring joint space damage in patients with rheumatoid arthritis (RA), but it needs to be validated through comparisons to other imaging methods.
To evaluate the degree of agreement with CT and x-ray scores, this study assessed MRI and CT images of the wrist and the second to fifth metacarpophalangeal (MCP 2-5) joints of 14 people with RA and one healthy control, who were from a clinical trial. Three readers assessed the images twice, and a single reader scored x-rays using the Sharp-Van der Heidje method, said Dr. Møller Døhn, who is in the center for rheumatology and spine diseases at the hospital.
The MRI scores of joint space narrowing "were very highly correlated" with CT scores, when comparing the wrist and MCP scores both separately and combined: Using intraclass correlation coefficients (ICCs) as a measure of agreement between scores and scorers, the MRI and CT scores for joint space narrowing were 0.94 for the MCP joints, 0.92 for the wrist, and 0.92 for the wrist and MCP joints combined. But the ICCs for the x-ray joint space narrowing scores were lower: With MRI scores, the ICCs were 0.49 for the MCP 2-5 joints and 0.55 for the wrist. With CT scores, the ICCs were 0.56 for the MCP 2-5 joints and 0.43 for the wrist.
"The most important next step is to test the scoring system in a longitudinal setting, in order to investigate the sensitivity to change," Dr. Møller Døhn said in an interview before the congress. "Before the system can be implemented as an outcome measure in clinical trials, we need to know if it is more sensitive than other methods that are already available. If it turns out that [joint space narrowing] assessment of several joints on x-ray is just as good as - or better than - MRI, then it does not add information to what we already use today."
Dr. Møller Døhn reported that he had no relevant financial disclosures.
A magnetic resonance imaging scoring system of joint-space narrowing in rheumatoid arthritis showed "a very high" agreement with computed tomography scores and may become a useful tool in rheumatoid arthritis clinical trials after further validation, judging from data presented by Dr. Uffe Møller Døhn.
In a small study, which was conducted to validate the OMERACT-RAMRIS MRI JSN scoring system in the wrists and metacarpophalangeal (MCP) joints, there was a very high agreement between the joint-space narrowing scores on MRI and CT and moderate agreement between scores on MRI and x-ray, said Dr. Møller Døhn of Copenhagen University Hospital at Glostrup at the annual European Congress of Rheumatology. In addition, there was "high to very high" inter- and intrareader reliability, particularly for the wrist joints.
An OMERACT (Outcome Measures in Rheumatology) initiative, this scoring system is being developed to provide a more precise and sensitive method of measuring joint space damage in patients with rheumatoid arthritis (RA), but it needs to be validated through comparisons to other imaging methods.
To evaluate the degree of agreement with CT and x-ray scores, this study assessed MRI and CT images of the wrist and the second to fifth metacarpophalangeal (MCP 2-5) joints of 14 people with RA and one healthy control, who were from a clinical trial. Three readers assessed the images twice, and a single reader scored x-rays using the Sharp-Van der Heidje method, said Dr. Møller Døhn, who is in the center for rheumatology and spine diseases at the hospital.
The MRI scores of joint space narrowing "were very highly correlated" with CT scores, when comparing the wrist and MCP scores both separately and combined: Using intraclass correlation coefficients (ICCs) as a measure of agreement between scores and scorers, the MRI and CT scores for joint space narrowing were 0.94 for the MCP joints, 0.92 for the wrist, and 0.92 for the wrist and MCP joints combined. But the ICCs for the x-ray joint space narrowing scores were lower: With MRI scores, the ICCs were 0.49 for the MCP 2-5 joints and 0.55 for the wrist. With CT scores, the ICCs were 0.56 for the MCP 2-5 joints and 0.43 for the wrist.
"The most important next step is to test the scoring system in a longitudinal setting, in order to investigate the sensitivity to change," Dr. Møller Døhn said in an interview before the congress. "Before the system can be implemented as an outcome measure in clinical trials, we need to know if it is more sensitive than other methods that are already available. If it turns out that [joint space narrowing] assessment of several joints on x-ray is just as good as - or better than - MRI, then it does not add information to what we already use today."
Dr. Møller Døhn reported that he had no relevant financial disclosures.
A magnetic resonance imaging scoring system of joint-space narrowing in rheumatoid arthritis showed "a very high" agreement with computed tomography scores and may become a useful tool in rheumatoid arthritis clinical trials after further validation, judging from data presented by Dr. Uffe Møller Døhn.
In a small study, which was conducted to validate the OMERACT-RAMRIS MRI JSN scoring system in the wrists and metacarpophalangeal (MCP) joints, there was a very high agreement between the joint-space narrowing scores on MRI and CT and moderate agreement between scores on MRI and x-ray, said Dr. Møller Døhn of Copenhagen University Hospital at Glostrup at the annual European Congress of Rheumatology. In addition, there was "high to very high" inter- and intrareader reliability, particularly for the wrist joints.
An OMERACT (Outcome Measures in Rheumatology) initiative, this scoring system is being developed to provide a more precise and sensitive method of measuring joint space damage in patients with rheumatoid arthritis (RA), but it needs to be validated through comparisons to other imaging methods.
To evaluate the degree of agreement with CT and x-ray scores, this study assessed MRI and CT images of the wrist and the second to fifth metacarpophalangeal (MCP 2-5) joints of 14 people with RA and one healthy control, who were from a clinical trial. Three readers assessed the images twice, and a single reader scored x-rays using the Sharp-Van der Heidje method, said Dr. Møller Døhn, who is in the center for rheumatology and spine diseases at the hospital.
The MRI scores of joint space narrowing "were very highly correlated" with CT scores, when comparing the wrist and MCP scores both separately and combined: Using intraclass correlation coefficients (ICCs) as a measure of agreement between scores and scorers, the MRI and CT scores for joint space narrowing were 0.94 for the MCP joints, 0.92 for the wrist, and 0.92 for the wrist and MCP joints combined. But the ICCs for the x-ray joint space narrowing scores were lower: With MRI scores, the ICCs were 0.49 for the MCP 2-5 joints and 0.55 for the wrist. With CT scores, the ICCs were 0.56 for the MCP 2-5 joints and 0.43 for the wrist.
"The most important next step is to test the scoring system in a longitudinal setting, in order to investigate the sensitivity to change," Dr. Møller Døhn said in an interview before the congress. "Before the system can be implemented as an outcome measure in clinical trials, we need to know if it is more sensitive than other methods that are already available. If it turns out that [joint space narrowing] assessment of several joints on x-ray is just as good as - or better than - MRI, then it does not add information to what we already use today."
Dr. Møller Døhn reported that he had no relevant financial disclosures.
FROM THE EULAR CONGRESS 2013
Biologics reduced sick leave in RA patients
Biologics and improved strategies for their use have significantly reduced the relatively high rate of sick leave among patients with rheumatoid arthritis, but more efficient, multiprofessional intervention strategies are still needed to reduce its incidence, Mathilda Bjork, Ph.D., reported at the annual European Congress of Rheumatism.
Dr. Bjork, of Jonkoping University, Sweden, conducted a subanalysis of the Swedish Early Rheumatoid Arthritis cohort study (Swedish acronym – TIRA). The study included patients with early rheumatoid arthritis, and was designed to calculate direct and indirect costs of the disease over a 3-year period. There have been two TIRA cohorts – one in 1996-1998 and one in 2005-2008. All had early disease; they were a mean of 62 years at baseline.
At all follow-up visits, the patients met with a multidisciplinary team including a physician, an occupational therapist and a physiotherapist, and were given individual treatment based on their needs.
Those in the more recent cohort were treated more aggressively with disease-modifying antirheumatic drugs (DMARDs), mainly methotrexate, starting at their first visit. They also received biologics when required.
Dr. Bjork’s study examined sick leave rates between the two TIRA cohorts: 1996-1998 and 2005-2008. The comparison found that sick leave rates in the newer cohort declined about 50% compared to those in the older cohort.
In the early cohort, sick leave rates were stable over the 3-year study period. At baseline, 60% of the patients were taking sick leave due to their RA; that number was unchanged at 3 years.
In the newer cohort, the baseline sick leave rate was similar, with 55% taking leave due to their disease. But at the 3-year follow-up, only 30% were on sick leave.
"I think it’s good news," Dr. Bjork said in an interview. Since both groups were making use of the multidisciplinary team treatment, DMARD treatment appeared to be the main driver behind the difference. "They are being used more frequently and in higher doses, and it’s working."
Dr. Bjork said the study came about not only because RA is associated with such high indirect costs for sick leave, but also because of the direct treatment costs of new medications such as biological agents.
"The rationale behind the study was to explore whether more effective disease control reduces sick leave in a postbiologic cohort compared to a prebiologic cohort, with the potential for compensating some of the increased treatment cost."
The researchers suggested that changes in political policies and the sickness insurance system may also have had some impact on the differences in sick leave between the two cohorts.
Despite the significant reductions in sick leave, she suggested that more could be done to address the persistently high rate of sick leave among individuals with RA.
"The impact of rheumatoid arthritis on an individual’s ability to work is a complex interaction of biological, psychological, social, and occupational factors," she said. "The interventions need to have a wider perspective than the rheumatoid arthritis per se and [should be] done in a close interaction between the patient, clinicians, employers, and policy makers early in the disease process."
Dr. Bjork had no conflicts of interest relevant to the study.
Biologics and improved strategies for their use have significantly reduced the relatively high rate of sick leave among patients with rheumatoid arthritis, but more efficient, multiprofessional intervention strategies are still needed to reduce its incidence, Mathilda Bjork, Ph.D., reported at the annual European Congress of Rheumatism.
Dr. Bjork, of Jonkoping University, Sweden, conducted a subanalysis of the Swedish Early Rheumatoid Arthritis cohort study (Swedish acronym – TIRA). The study included patients with early rheumatoid arthritis, and was designed to calculate direct and indirect costs of the disease over a 3-year period. There have been two TIRA cohorts – one in 1996-1998 and one in 2005-2008. All had early disease; they were a mean of 62 years at baseline.
At all follow-up visits, the patients met with a multidisciplinary team including a physician, an occupational therapist and a physiotherapist, and were given individual treatment based on their needs.
Those in the more recent cohort were treated more aggressively with disease-modifying antirheumatic drugs (DMARDs), mainly methotrexate, starting at their first visit. They also received biologics when required.
Dr. Bjork’s study examined sick leave rates between the two TIRA cohorts: 1996-1998 and 2005-2008. The comparison found that sick leave rates in the newer cohort declined about 50% compared to those in the older cohort.
In the early cohort, sick leave rates were stable over the 3-year study period. At baseline, 60% of the patients were taking sick leave due to their RA; that number was unchanged at 3 years.
In the newer cohort, the baseline sick leave rate was similar, with 55% taking leave due to their disease. But at the 3-year follow-up, only 30% were on sick leave.
"I think it’s good news," Dr. Bjork said in an interview. Since both groups were making use of the multidisciplinary team treatment, DMARD treatment appeared to be the main driver behind the difference. "They are being used more frequently and in higher doses, and it’s working."
Dr. Bjork said the study came about not only because RA is associated with such high indirect costs for sick leave, but also because of the direct treatment costs of new medications such as biological agents.
"The rationale behind the study was to explore whether more effective disease control reduces sick leave in a postbiologic cohort compared to a prebiologic cohort, with the potential for compensating some of the increased treatment cost."
The researchers suggested that changes in political policies and the sickness insurance system may also have had some impact on the differences in sick leave between the two cohorts.
Despite the significant reductions in sick leave, she suggested that more could be done to address the persistently high rate of sick leave among individuals with RA.
"The impact of rheumatoid arthritis on an individual’s ability to work is a complex interaction of biological, psychological, social, and occupational factors," she said. "The interventions need to have a wider perspective than the rheumatoid arthritis per se and [should be] done in a close interaction between the patient, clinicians, employers, and policy makers early in the disease process."
Dr. Bjork had no conflicts of interest relevant to the study.
Biologics and improved strategies for their use have significantly reduced the relatively high rate of sick leave among patients with rheumatoid arthritis, but more efficient, multiprofessional intervention strategies are still needed to reduce its incidence, Mathilda Bjork, Ph.D., reported at the annual European Congress of Rheumatism.
Dr. Bjork, of Jonkoping University, Sweden, conducted a subanalysis of the Swedish Early Rheumatoid Arthritis cohort study (Swedish acronym – TIRA). The study included patients with early rheumatoid arthritis, and was designed to calculate direct and indirect costs of the disease over a 3-year period. There have been two TIRA cohorts – one in 1996-1998 and one in 2005-2008. All had early disease; they were a mean of 62 years at baseline.
At all follow-up visits, the patients met with a multidisciplinary team including a physician, an occupational therapist and a physiotherapist, and were given individual treatment based on their needs.
Those in the more recent cohort were treated more aggressively with disease-modifying antirheumatic drugs (DMARDs), mainly methotrexate, starting at their first visit. They also received biologics when required.
Dr. Bjork’s study examined sick leave rates between the two TIRA cohorts: 1996-1998 and 2005-2008. The comparison found that sick leave rates in the newer cohort declined about 50% compared to those in the older cohort.
In the early cohort, sick leave rates were stable over the 3-year study period. At baseline, 60% of the patients were taking sick leave due to their RA; that number was unchanged at 3 years.
In the newer cohort, the baseline sick leave rate was similar, with 55% taking leave due to their disease. But at the 3-year follow-up, only 30% were on sick leave.
"I think it’s good news," Dr. Bjork said in an interview. Since both groups were making use of the multidisciplinary team treatment, DMARD treatment appeared to be the main driver behind the difference. "They are being used more frequently and in higher doses, and it’s working."
Dr. Bjork said the study came about not only because RA is associated with such high indirect costs for sick leave, but also because of the direct treatment costs of new medications such as biological agents.
"The rationale behind the study was to explore whether more effective disease control reduces sick leave in a postbiologic cohort compared to a prebiologic cohort, with the potential for compensating some of the increased treatment cost."
The researchers suggested that changes in political policies and the sickness insurance system may also have had some impact on the differences in sick leave between the two cohorts.
Despite the significant reductions in sick leave, she suggested that more could be done to address the persistently high rate of sick leave among individuals with RA.
"The impact of rheumatoid arthritis on an individual’s ability to work is a complex interaction of biological, psychological, social, and occupational factors," she said. "The interventions need to have a wider perspective than the rheumatoid arthritis per se and [should be] done in a close interaction between the patient, clinicians, employers, and policy makers early in the disease process."
Dr. Bjork had no conflicts of interest relevant to the study.
AT THE EULAR CONGRESS 2013
Ustekinumab benefits in PsA sustained through 1 year
MADRID – The lessening of the signs and symptoms of psoriatic arthritis that occurs during the first 6 months of ustekinumab treatment persisted and improved further at the end of 1 year, with a favorable safety profile, according to 52-week data from the PSUMMIT II trial.
The sustained benefits in American College of Rheumatology (ACR) 20 responses and other efficacy endpoints were evident even in patients who had been treated previously with anti-tumor necrosis factor (anti-TNF) agents and among those who were anti-TNF naïve, although the benefits were greater in the latter group patients, according to Dr. Christopher T. Ritchlin, a professor in the department of medicine, allergy/immunology, and rheumatology at the University of Rochester (N.Y.). This includes beneficial effects on skin and enthesitis, Dr. Ritchlin said at the annual European Congress of Rheumatology.
The PSUMMIT II study is a follow-up to the PSUMMIT I study, the findings of which showed that ustekinumab, a human interleukin (IL)-12 and IL-23 antagonist, showed significant effectiveness in patients with psoriatic arthritis (PsA) who had not been exposed to anti-TNF drugs.
Ustekinumab is currently approved for treating moderate to severe plaque psoriasis in adults who are candidates for phototherapy or systemic therapy in the United States, or those who have failed to respond to, have a contraindication to, or are intolerant of other systemic therapies in Europe. In December 2012, the manufacturer, Janssen, announced that it had filed for further approval for ustekinumab in both the United States and Europe for the treatment of active disease.
The PSUMMIT II study enrolled 312 patients with active PsA who had five or more tender and five or more swollen joints, and a C-reactive protein level of 0.3 mg/dL or higher. Patients who had been treated previously with anti-TNF therapy (n = 180) and those naive to anti-TNF therapy (n = 132) were included and randomized to one of two doses of ustekinumab (45 mg or 90 mg) or placebo administered at week 0, 4, and 12. At 16 weeks, patients with less than a 5% improvement in tender and swollen joint counts on placebo were switched to active treatment, those on 45 mg ustekinumab had their dose upped to 90 mg, and those on 90 mg remained on that dose.
At 6 months, significantly more patients treated with ustekinumab than placebo achieved the primary endpoint of an ACR 20, and more patients on active treatment had an ACR 50, and at least a 75% improvement in the Psoriasis Area and Severity Index (PASI 75).
These results were sustained at 1 year, with 47%-48% of those on 45 mg and 90 mg, and 56% of those who switched from placebo to the 45-mg dose, achieving an ACR 20. In addition, 26%-29% achieved an ACR 50 (29% for those switched from placebo), and 13%-18% achieved an ACR 70 (15% for placebo). There were also improvements associated with treatment in HAQ-DI (Health Assessment Questionnaire-Disability Index) scores at week 52, according to Dr. Ritchlin. The mean change in HAQ-DI scores from baseline to week 52 were -0.21 for placebo, -0.20 for the 45-mg dose of ustekinumab, and -0.28 for the 90-mg dose.
Among those who had not been treated before with an anti-TNF agent, 59%-60% of those on ustekinumab (73% for those switched from placebo) achieved an ACR 20 at week 52, compared with 37%-41% of those who had taken an anti-TNF agent previously before being treated with ustekinumab (30% for placebo). Although responses among anti-TNF naive patients were superior, the responses among those who had been treated with these agents previously were still significantly improved, an indication that ustekinumab "offers an alternative for patients who cannot take or fail anti-TNF agents," Dr. Ritchlin said in the interview.
Treatment was "very effective" for skin symptoms and for enthesitis, he noted. Compared with baseline, dactylitis was improved by a median of 95% among those on the 45-mg dose, 91% among those on the 90-mg dose, and 100% in those who switched from placebo to the 45-mg dose of ustekinumab. Similar improvements in enthesitis were seen, with the highest improvement (60%) seen with the highest dose of ustekinumab. PASI scores at baseline ranged from 11 to 13 and improved by 56%-64% by follow-up at week 52.
In general, ustekinumab was well tolerated, with no deaths or cases of tuberculosis reported and with similar rates of adverse events and serious adverse events between the two doses (just under 6%). There were two malignancies: one breast cancer and one squamous cell carcinoma in two patients taking the 90-mg dose of ustekinumab, who had both been treated with anti-TNFs previously. The rate of serious infections was less than 1% among those treated with ustekinumab. Through 60 weeks of treatment, there were three major adverse cardiovascular events, all myocardial infarctions, in patients treated with ustekinumab. These patients all had multiple cardiovascular risk factors, Dr. Ritchlin said. They had also been exposed previously to anti-TNF treatment.
Radiographic data from the trial are expected and likely to be available by the end of the year for presentation at the annual American College of Rheumatology meeting.
Dr. Ritchlin disclosed having received grant and research support from Janssen. Four of the nine remaining authors are Janssen employees and shareholders of Johnson & Johnson, Janssen’s parent company. Ustekinumab is marketed as Stelara in the United States.
Sara Freeman contributed to this report.
MADRID – The lessening of the signs and symptoms of psoriatic arthritis that occurs during the first 6 months of ustekinumab treatment persisted and improved further at the end of 1 year, with a favorable safety profile, according to 52-week data from the PSUMMIT II trial.
The sustained benefits in American College of Rheumatology (ACR) 20 responses and other efficacy endpoints were evident even in patients who had been treated previously with anti-tumor necrosis factor (anti-TNF) agents and among those who were anti-TNF naïve, although the benefits were greater in the latter group patients, according to Dr. Christopher T. Ritchlin, a professor in the department of medicine, allergy/immunology, and rheumatology at the University of Rochester (N.Y.). This includes beneficial effects on skin and enthesitis, Dr. Ritchlin said at the annual European Congress of Rheumatology.
The PSUMMIT II study is a follow-up to the PSUMMIT I study, the findings of which showed that ustekinumab, a human interleukin (IL)-12 and IL-23 antagonist, showed significant effectiveness in patients with psoriatic arthritis (PsA) who had not been exposed to anti-TNF drugs.
Ustekinumab is currently approved for treating moderate to severe plaque psoriasis in adults who are candidates for phototherapy or systemic therapy in the United States, or those who have failed to respond to, have a contraindication to, or are intolerant of other systemic therapies in Europe. In December 2012, the manufacturer, Janssen, announced that it had filed for further approval for ustekinumab in both the United States and Europe for the treatment of active disease.
The PSUMMIT II study enrolled 312 patients with active PsA who had five or more tender and five or more swollen joints, and a C-reactive protein level of 0.3 mg/dL or higher. Patients who had been treated previously with anti-TNF therapy (n = 180) and those naive to anti-TNF therapy (n = 132) were included and randomized to one of two doses of ustekinumab (45 mg or 90 mg) or placebo administered at week 0, 4, and 12. At 16 weeks, patients with less than a 5% improvement in tender and swollen joint counts on placebo were switched to active treatment, those on 45 mg ustekinumab had their dose upped to 90 mg, and those on 90 mg remained on that dose.
At 6 months, significantly more patients treated with ustekinumab than placebo achieved the primary endpoint of an ACR 20, and more patients on active treatment had an ACR 50, and at least a 75% improvement in the Psoriasis Area and Severity Index (PASI 75).
These results were sustained at 1 year, with 47%-48% of those on 45 mg and 90 mg, and 56% of those who switched from placebo to the 45-mg dose, achieving an ACR 20. In addition, 26%-29% achieved an ACR 50 (29% for those switched from placebo), and 13%-18% achieved an ACR 70 (15% for placebo). There were also improvements associated with treatment in HAQ-DI (Health Assessment Questionnaire-Disability Index) scores at week 52, according to Dr. Ritchlin. The mean change in HAQ-DI scores from baseline to week 52 were -0.21 for placebo, -0.20 for the 45-mg dose of ustekinumab, and -0.28 for the 90-mg dose.
Among those who had not been treated before with an anti-TNF agent, 59%-60% of those on ustekinumab (73% for those switched from placebo) achieved an ACR 20 at week 52, compared with 37%-41% of those who had taken an anti-TNF agent previously before being treated with ustekinumab (30% for placebo). Although responses among anti-TNF naive patients were superior, the responses among those who had been treated with these agents previously were still significantly improved, an indication that ustekinumab "offers an alternative for patients who cannot take or fail anti-TNF agents," Dr. Ritchlin said in the interview.
Treatment was "very effective" for skin symptoms and for enthesitis, he noted. Compared with baseline, dactylitis was improved by a median of 95% among those on the 45-mg dose, 91% among those on the 90-mg dose, and 100% in those who switched from placebo to the 45-mg dose of ustekinumab. Similar improvements in enthesitis were seen, with the highest improvement (60%) seen with the highest dose of ustekinumab. PASI scores at baseline ranged from 11 to 13 and improved by 56%-64% by follow-up at week 52.
In general, ustekinumab was well tolerated, with no deaths or cases of tuberculosis reported and with similar rates of adverse events and serious adverse events between the two doses (just under 6%). There were two malignancies: one breast cancer and one squamous cell carcinoma in two patients taking the 90-mg dose of ustekinumab, who had both been treated with anti-TNFs previously. The rate of serious infections was less than 1% among those treated with ustekinumab. Through 60 weeks of treatment, there were three major adverse cardiovascular events, all myocardial infarctions, in patients treated with ustekinumab. These patients all had multiple cardiovascular risk factors, Dr. Ritchlin said. They had also been exposed previously to anti-TNF treatment.
Radiographic data from the trial are expected and likely to be available by the end of the year for presentation at the annual American College of Rheumatology meeting.
Dr. Ritchlin disclosed having received grant and research support from Janssen. Four of the nine remaining authors are Janssen employees and shareholders of Johnson & Johnson, Janssen’s parent company. Ustekinumab is marketed as Stelara in the United States.
Sara Freeman contributed to this report.
MADRID – The lessening of the signs and symptoms of psoriatic arthritis that occurs during the first 6 months of ustekinumab treatment persisted and improved further at the end of 1 year, with a favorable safety profile, according to 52-week data from the PSUMMIT II trial.
The sustained benefits in American College of Rheumatology (ACR) 20 responses and other efficacy endpoints were evident even in patients who had been treated previously with anti-tumor necrosis factor (anti-TNF) agents and among those who were anti-TNF naïve, although the benefits were greater in the latter group patients, according to Dr. Christopher T. Ritchlin, a professor in the department of medicine, allergy/immunology, and rheumatology at the University of Rochester (N.Y.). This includes beneficial effects on skin and enthesitis, Dr. Ritchlin said at the annual European Congress of Rheumatology.
The PSUMMIT II study is a follow-up to the PSUMMIT I study, the findings of which showed that ustekinumab, a human interleukin (IL)-12 and IL-23 antagonist, showed significant effectiveness in patients with psoriatic arthritis (PsA) who had not been exposed to anti-TNF drugs.
Ustekinumab is currently approved for treating moderate to severe plaque psoriasis in adults who are candidates for phototherapy or systemic therapy in the United States, or those who have failed to respond to, have a contraindication to, or are intolerant of other systemic therapies in Europe. In December 2012, the manufacturer, Janssen, announced that it had filed for further approval for ustekinumab in both the United States and Europe for the treatment of active disease.
The PSUMMIT II study enrolled 312 patients with active PsA who had five or more tender and five or more swollen joints, and a C-reactive protein level of 0.3 mg/dL or higher. Patients who had been treated previously with anti-TNF therapy (n = 180) and those naive to anti-TNF therapy (n = 132) were included and randomized to one of two doses of ustekinumab (45 mg or 90 mg) or placebo administered at week 0, 4, and 12. At 16 weeks, patients with less than a 5% improvement in tender and swollen joint counts on placebo were switched to active treatment, those on 45 mg ustekinumab had their dose upped to 90 mg, and those on 90 mg remained on that dose.
At 6 months, significantly more patients treated with ustekinumab than placebo achieved the primary endpoint of an ACR 20, and more patients on active treatment had an ACR 50, and at least a 75% improvement in the Psoriasis Area and Severity Index (PASI 75).
These results were sustained at 1 year, with 47%-48% of those on 45 mg and 90 mg, and 56% of those who switched from placebo to the 45-mg dose, achieving an ACR 20. In addition, 26%-29% achieved an ACR 50 (29% for those switched from placebo), and 13%-18% achieved an ACR 70 (15% for placebo). There were also improvements associated with treatment in HAQ-DI (Health Assessment Questionnaire-Disability Index) scores at week 52, according to Dr. Ritchlin. The mean change in HAQ-DI scores from baseline to week 52 were -0.21 for placebo, -0.20 for the 45-mg dose of ustekinumab, and -0.28 for the 90-mg dose.
Among those who had not been treated before with an anti-TNF agent, 59%-60% of those on ustekinumab (73% for those switched from placebo) achieved an ACR 20 at week 52, compared with 37%-41% of those who had taken an anti-TNF agent previously before being treated with ustekinumab (30% for placebo). Although responses among anti-TNF naive patients were superior, the responses among those who had been treated with these agents previously were still significantly improved, an indication that ustekinumab "offers an alternative for patients who cannot take or fail anti-TNF agents," Dr. Ritchlin said in the interview.
Treatment was "very effective" for skin symptoms and for enthesitis, he noted. Compared with baseline, dactylitis was improved by a median of 95% among those on the 45-mg dose, 91% among those on the 90-mg dose, and 100% in those who switched from placebo to the 45-mg dose of ustekinumab. Similar improvements in enthesitis were seen, with the highest improvement (60%) seen with the highest dose of ustekinumab. PASI scores at baseline ranged from 11 to 13 and improved by 56%-64% by follow-up at week 52.
In general, ustekinumab was well tolerated, with no deaths or cases of tuberculosis reported and with similar rates of adverse events and serious adverse events between the two doses (just under 6%). There were two malignancies: one breast cancer and one squamous cell carcinoma in two patients taking the 90-mg dose of ustekinumab, who had both been treated with anti-TNFs previously. The rate of serious infections was less than 1% among those treated with ustekinumab. Through 60 weeks of treatment, there were three major adverse cardiovascular events, all myocardial infarctions, in patients treated with ustekinumab. These patients all had multiple cardiovascular risk factors, Dr. Ritchlin said. They had also been exposed previously to anti-TNF treatment.
Radiographic data from the trial are expected and likely to be available by the end of the year for presentation at the annual American College of Rheumatology meeting.
Dr. Ritchlin disclosed having received grant and research support from Janssen. Four of the nine remaining authors are Janssen employees and shareholders of Johnson & Johnson, Janssen’s parent company. Ustekinumab is marketed as Stelara in the United States.
Sara Freeman contributed to this report.
AT THE EULAR CONGRESS 2013
Exercise program improved rheumatoid arthritis of the hands
BIRMINGHAM, ENGLAND – Patients with hand or wrist problems from rheumatoid arthritis can significantly benefit from a hand-specific exercise program beyond what is achieved with usual care.
"There was a significant difference in the primary outcome measure [the Michigan Hand Outcomes Questionnaire] in favor of the exercise program and this was actually maintained over the 12-month follow period as well," study investigator Dr. Mark Williams said at the annual meeting of the British Society for Rheumatology. Dr. Williams is a research fellow in the Clinical Trials Unit at the University of Warwick, England, where the trial is being coordinated.
In the randomized, controlled trial called SARAH (Strengthening and Stretching for Rheumatoid Arthritis of the Hand), mean Michigan Hand Outcomes Questionnaire (MHQ) scores at 4 months’ assessment improved from 52.1 at baseline in both groups to 61.1 in the exercise group (n = 246) and 56.6 in the usual care group (n = 244), giving a mean difference in hand function improvement of 4.5 (P less than .0002) in favor of the exercise program.
The effects of the 12-week and home-based intervention were sustained at 12 months’ follow-up, with mean MHQ scores of 60.7 (n = 216) and 56.4 (n = 222) in the exercise and usual care groups, respectively, giving a mean difference of 4.3 (P less than .002).
Clinical guidelines in England and Wales state that patients with RA should have access to specialist hand therapy to help increase strength, movement, and function. Prior to the results of the SARAH trial, however, the evidence upon which this recommendation is based was "rather weak," Dr. Williams observed.
The aims of the study were therefore to determine the clinical effectiveness of an exercise program developed to specifically target the hands and upper limbs in patients with RA hand dysfunction, and then to examine its cost-effectiveness.
The study was performed within 17 National Health Service Trusts in England and included RA patients with pain or dysfunction of the hands or wrist joints who were not taking disease-modifying antirheumatic therapy, or if they were, had been stable on treatment for at least 3 months. The 490 patients randomized in the trial had a median age of 63 years and 10 years’ RA disease duration.
Usual care consisted of one-on-one sessions with a hand therapist and information about joint protection and general exercise advice, with functional splinting if there was a clinical need. In addition to this, the patients who were randomized to the SARAH exercise program received a further five sessions of supervised exercises over a 12-week period; sessions included 11 exercises designed to help with stretching and strengthening of the hand, guidance on a daily exercise program to be performed by patients at home, and strategies to encourage adherence (Physiotherapy 2012;98:121-30).
"In order to get patients to adhere to this exercise plan, which is fairly key, we used behavioral strategies, including an exercise diary and a behavioral action plan," Dr. Williams explained. "This was like a contract between the patient and [his or her] therapist." A total of 438 (89%) patients were followed up at 12 months.
Patient-reported self-efficacy was higher with the exercise program, compared with usual care. Indeed, in a letter recently sent to all the patients who participated in the trial, Dr. Williams noted that, "43% of the patients receiving the SARAH exercise programme and 20% of patients receiving usual care reported some or much improvement."
There was no difference in pain scores between groups, with trends for all participants to report some improvement in pain over time. There was also no difference in the number of adverse events between the groups.
Importantly, the SARAH exercise program was shown to be cost effective, incurring only an additional £100 per patient (approximately US$150) to provide. This means existing health care staff could potentially deliver the program, and with relative ease, the SARAH trial team believes.
Their next step is to see if the intervention’s benefits are extended beyond 12 months. The researchers are also looking at how to best to train healthcare professionals to deliver the program in routine practice in the United Kingdom.
The U.K. National Institute for Health Research Health Technology Assessment Program funded the trial. Dr. Williams had no conflicts of interest.
BIRMINGHAM, ENGLAND – Patients with hand or wrist problems from rheumatoid arthritis can significantly benefit from a hand-specific exercise program beyond what is achieved with usual care.
"There was a significant difference in the primary outcome measure [the Michigan Hand Outcomes Questionnaire] in favor of the exercise program and this was actually maintained over the 12-month follow period as well," study investigator Dr. Mark Williams said at the annual meeting of the British Society for Rheumatology. Dr. Williams is a research fellow in the Clinical Trials Unit at the University of Warwick, England, where the trial is being coordinated.
In the randomized, controlled trial called SARAH (Strengthening and Stretching for Rheumatoid Arthritis of the Hand), mean Michigan Hand Outcomes Questionnaire (MHQ) scores at 4 months’ assessment improved from 52.1 at baseline in both groups to 61.1 in the exercise group (n = 246) and 56.6 in the usual care group (n = 244), giving a mean difference in hand function improvement of 4.5 (P less than .0002) in favor of the exercise program.
The effects of the 12-week and home-based intervention were sustained at 12 months’ follow-up, with mean MHQ scores of 60.7 (n = 216) and 56.4 (n = 222) in the exercise and usual care groups, respectively, giving a mean difference of 4.3 (P less than .002).
Clinical guidelines in England and Wales state that patients with RA should have access to specialist hand therapy to help increase strength, movement, and function. Prior to the results of the SARAH trial, however, the evidence upon which this recommendation is based was "rather weak," Dr. Williams observed.
The aims of the study were therefore to determine the clinical effectiveness of an exercise program developed to specifically target the hands and upper limbs in patients with RA hand dysfunction, and then to examine its cost-effectiveness.
The study was performed within 17 National Health Service Trusts in England and included RA patients with pain or dysfunction of the hands or wrist joints who were not taking disease-modifying antirheumatic therapy, or if they were, had been stable on treatment for at least 3 months. The 490 patients randomized in the trial had a median age of 63 years and 10 years’ RA disease duration.
Usual care consisted of one-on-one sessions with a hand therapist and information about joint protection and general exercise advice, with functional splinting if there was a clinical need. In addition to this, the patients who were randomized to the SARAH exercise program received a further five sessions of supervised exercises over a 12-week period; sessions included 11 exercises designed to help with stretching and strengthening of the hand, guidance on a daily exercise program to be performed by patients at home, and strategies to encourage adherence (Physiotherapy 2012;98:121-30).
"In order to get patients to adhere to this exercise plan, which is fairly key, we used behavioral strategies, including an exercise diary and a behavioral action plan," Dr. Williams explained. "This was like a contract between the patient and [his or her] therapist." A total of 438 (89%) patients were followed up at 12 months.
Patient-reported self-efficacy was higher with the exercise program, compared with usual care. Indeed, in a letter recently sent to all the patients who participated in the trial, Dr. Williams noted that, "43% of the patients receiving the SARAH exercise programme and 20% of patients receiving usual care reported some or much improvement."
There was no difference in pain scores between groups, with trends for all participants to report some improvement in pain over time. There was also no difference in the number of adverse events between the groups.
Importantly, the SARAH exercise program was shown to be cost effective, incurring only an additional £100 per patient (approximately US$150) to provide. This means existing health care staff could potentially deliver the program, and with relative ease, the SARAH trial team believes.
Their next step is to see if the intervention’s benefits are extended beyond 12 months. The researchers are also looking at how to best to train healthcare professionals to deliver the program in routine practice in the United Kingdom.
The U.K. National Institute for Health Research Health Technology Assessment Program funded the trial. Dr. Williams had no conflicts of interest.
BIRMINGHAM, ENGLAND – Patients with hand or wrist problems from rheumatoid arthritis can significantly benefit from a hand-specific exercise program beyond what is achieved with usual care.
"There was a significant difference in the primary outcome measure [the Michigan Hand Outcomes Questionnaire] in favor of the exercise program and this was actually maintained over the 12-month follow period as well," study investigator Dr. Mark Williams said at the annual meeting of the British Society for Rheumatology. Dr. Williams is a research fellow in the Clinical Trials Unit at the University of Warwick, England, where the trial is being coordinated.
In the randomized, controlled trial called SARAH (Strengthening and Stretching for Rheumatoid Arthritis of the Hand), mean Michigan Hand Outcomes Questionnaire (MHQ) scores at 4 months’ assessment improved from 52.1 at baseline in both groups to 61.1 in the exercise group (n = 246) and 56.6 in the usual care group (n = 244), giving a mean difference in hand function improvement of 4.5 (P less than .0002) in favor of the exercise program.
The effects of the 12-week and home-based intervention were sustained at 12 months’ follow-up, with mean MHQ scores of 60.7 (n = 216) and 56.4 (n = 222) in the exercise and usual care groups, respectively, giving a mean difference of 4.3 (P less than .002).
Clinical guidelines in England and Wales state that patients with RA should have access to specialist hand therapy to help increase strength, movement, and function. Prior to the results of the SARAH trial, however, the evidence upon which this recommendation is based was "rather weak," Dr. Williams observed.
The aims of the study were therefore to determine the clinical effectiveness of an exercise program developed to specifically target the hands and upper limbs in patients with RA hand dysfunction, and then to examine its cost-effectiveness.
The study was performed within 17 National Health Service Trusts in England and included RA patients with pain or dysfunction of the hands or wrist joints who were not taking disease-modifying antirheumatic therapy, or if they were, had been stable on treatment for at least 3 months. The 490 patients randomized in the trial had a median age of 63 years and 10 years’ RA disease duration.
Usual care consisted of one-on-one sessions with a hand therapist and information about joint protection and general exercise advice, with functional splinting if there was a clinical need. In addition to this, the patients who were randomized to the SARAH exercise program received a further five sessions of supervised exercises over a 12-week period; sessions included 11 exercises designed to help with stretching and strengthening of the hand, guidance on a daily exercise program to be performed by patients at home, and strategies to encourage adherence (Physiotherapy 2012;98:121-30).
"In order to get patients to adhere to this exercise plan, which is fairly key, we used behavioral strategies, including an exercise diary and a behavioral action plan," Dr. Williams explained. "This was like a contract between the patient and [his or her] therapist." A total of 438 (89%) patients were followed up at 12 months.
Patient-reported self-efficacy was higher with the exercise program, compared with usual care. Indeed, in a letter recently sent to all the patients who participated in the trial, Dr. Williams noted that, "43% of the patients receiving the SARAH exercise programme and 20% of patients receiving usual care reported some or much improvement."
There was no difference in pain scores between groups, with trends for all participants to report some improvement in pain over time. There was also no difference in the number of adverse events between the groups.
Importantly, the SARAH exercise program was shown to be cost effective, incurring only an additional £100 per patient (approximately US$150) to provide. This means existing health care staff could potentially deliver the program, and with relative ease, the SARAH trial team believes.
Their next step is to see if the intervention’s benefits are extended beyond 12 months. The researchers are also looking at how to best to train healthcare professionals to deliver the program in routine practice in the United Kingdom.
The U.K. National Institute for Health Research Health Technology Assessment Program funded the trial. Dr. Williams had no conflicts of interest.
AT RHEUMATOLOGY 2013
Major finding: There was a mean difference in Michigan Hand Outcomes Questionnaire scores of 4.5 (P less than .002) at 4 months and 4.3 (P less than .002) at 12 months.
Data source: Multicenter, randomized, controlled trial of 490 patients with hand or wrist problems from RA who were treated with usual care or a hand-specific exercise program, with follow-up at 4 and 12 months.
Disclosures: The U.K. National Institute for Health Research Health Technology Assessment Program funded the trial. Dr. Williams had no conflicts of interest.
Golimumab indication expanded to include ulcerative colitis
The approval of the biologic drug golimumab has been expanded to include the treatment of adults with moderate to severe ulcerative colitis that is refractory to prior treatment or requires continuous steroid therapy, the Food and Drug Administration announced on May 15.
Golimumab (Simponi), a tumor necrosis factor–blocker, was approved in 2009 for treatment of moderate to severe active rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis, all in adults. Simponi is marketed by Janssen Ortho Biotech.
The approval for ulcerative colitis was based on two studies of patients with moderate to severe ulcerative colitis, according to the FDA statement announcing the approval. In one study, which enrolled 513 patients who could not tolerate or had not responded to other treatments, "a greater proportion of Simponi-treated patients achieved clinical response, clinical remission and, as seen during endoscopy, had improved appearance of the colon after 6 weeks," compared with those on placebo, the statement said.
In a study of 310 patients who had responded to golimumab and were then randomized to continue treatment with golimumab or were switched to placebo, "a greater proportion of Simponi-treated patients maintained clinical response through week 54, had clinical remission at both weeks 30 and 54 and, as seen during endoscopy, had improved appearance of the colon at both weeks 30 and 54 compared with the placebo group," the FDA said. The most common adverse effects associated with golimumab are upper respiratory infection and redness at the injection site.
The risks of serious infections, invasive fungal infections, reactivation of hepatitis B infection, lymphoma, heart failure, nervous system disorders, and allergic reactions are increased with treatment.
Serious adverse events associated with golimumab should be reported to the FDA at 800-332-1088 or www.fda.gov/medwatch/.
The approval of the biologic drug golimumab has been expanded to include the treatment of adults with moderate to severe ulcerative colitis that is refractory to prior treatment or requires continuous steroid therapy, the Food and Drug Administration announced on May 15.
Golimumab (Simponi), a tumor necrosis factor–blocker, was approved in 2009 for treatment of moderate to severe active rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis, all in adults. Simponi is marketed by Janssen Ortho Biotech.
The approval for ulcerative colitis was based on two studies of patients with moderate to severe ulcerative colitis, according to the FDA statement announcing the approval. In one study, which enrolled 513 patients who could not tolerate or had not responded to other treatments, "a greater proportion of Simponi-treated patients achieved clinical response, clinical remission and, as seen during endoscopy, had improved appearance of the colon after 6 weeks," compared with those on placebo, the statement said.
In a study of 310 patients who had responded to golimumab and were then randomized to continue treatment with golimumab or were switched to placebo, "a greater proportion of Simponi-treated patients maintained clinical response through week 54, had clinical remission at both weeks 30 and 54 and, as seen during endoscopy, had improved appearance of the colon at both weeks 30 and 54 compared with the placebo group," the FDA said. The most common adverse effects associated with golimumab are upper respiratory infection and redness at the injection site.
The risks of serious infections, invasive fungal infections, reactivation of hepatitis B infection, lymphoma, heart failure, nervous system disorders, and allergic reactions are increased with treatment.
Serious adverse events associated with golimumab should be reported to the FDA at 800-332-1088 or www.fda.gov/medwatch/.
The approval of the biologic drug golimumab has been expanded to include the treatment of adults with moderate to severe ulcerative colitis that is refractory to prior treatment or requires continuous steroid therapy, the Food and Drug Administration announced on May 15.
Golimumab (Simponi), a tumor necrosis factor–blocker, was approved in 2009 for treatment of moderate to severe active rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis, all in adults. Simponi is marketed by Janssen Ortho Biotech.
The approval for ulcerative colitis was based on two studies of patients with moderate to severe ulcerative colitis, according to the FDA statement announcing the approval. In one study, which enrolled 513 patients who could not tolerate or had not responded to other treatments, "a greater proportion of Simponi-treated patients achieved clinical response, clinical remission and, as seen during endoscopy, had improved appearance of the colon after 6 weeks," compared with those on placebo, the statement said.
In a study of 310 patients who had responded to golimumab and were then randomized to continue treatment with golimumab or were switched to placebo, "a greater proportion of Simponi-treated patients maintained clinical response through week 54, had clinical remission at both weeks 30 and 54 and, as seen during endoscopy, had improved appearance of the colon at both weeks 30 and 54 compared with the placebo group," the FDA said. The most common adverse effects associated with golimumab are upper respiratory infection and redness at the injection site.
The risks of serious infections, invasive fungal infections, reactivation of hepatitis B infection, lymphoma, heart failure, nervous system disorders, and allergic reactions are increased with treatment.
Serious adverse events associated with golimumab should be reported to the FDA at 800-332-1088 or www.fda.gov/medwatch/.
Registry links anti-TNF therapy to reduced heart attack risk
BIRMINGHAM, ENGLAND – The risk of heart attack appears to be lower in patients treated with anti–tumor necrosis factor therapy than with conventional disease-modifying antirheumatic drugs.
Patients treated with the biologic agents had a 30% lower risk for having a myocardial infarction, based on data from the British Society for Rheumatology Biologics Register (BSRBR) for rheumatoid arthritis. However, no differences were detected in 30-day or 1-year mortality rates (adjusted hazard ratios of 0.9 and 0.97, respectively).
"There seems to be a signal that subjects ever exposed to anti-TNF therapy were potentially at reduced risk for developing an MI," Dr. Audrey Low said in an interview at the British Society for Rheumatology annual conference.
Dr. Low, a clinical research fellow in the Arthritis Research UK Epidemiology Unit, University of Manchester, England, explained that traditional risk factors do not fully account for the well-known increased risk for cardiovascular disease in patients with rheumatoid arthritis. Underlying inflammation might play a role, and it was hypothesized that anti-TNF therapy might help to reduce this inflammation, with subsequent cardiovascular benefits.
The BSRBR-RA is one of the largest biologics registers in the world and has been running for more than 10 years. The register currently includes data on more than 20,000 participants treated with either anti-TNF agents or nonbiologic disease-modifying antirheumatic drugs (DMARDs), such as methotrexate.
The present study used data collated between 2001 and 2008 on 11,536 patients treated with anti-TNF drugs licensed at the time in the United Kingdom (etanercept, infliximab, and adalimumab), and 3,225 patients who received nonbiologic DMARDs. Patients with prior MI or angina were excluded, and patients were followed up through April 2010 or until death, incident MI, or date of last clinician assessment, whichever came first.
Patients treated with anti-TNF therapy were younger than those given nonbiologic DMARDs, with a mean age of 56 years versus 60 years at recruitment into the BSRBR-RA. They also tended to have higher disease activity and longer disease duration (11 years vs. 6 years). There were similar percentages of patients with hypertension (29% vs. 32%), diabetes (6% for both), and a history of ever having smoked (59% vs. 62%).
There were 224 incident MIs in the anti-TNF–treated group at a median follow-up of 6 years per person, and 52 heart attacks in the nonbiologic DMARD–treated group at a median follow-up of 4 years per person. "Even though the study has been running for over 10 years, the amount of follow-up per person is between 4 and 6 years," Dr. Low observed.
The unadjusted hazard ratio for MI risk was 0.9, and it increased to 1.2 when adjusted for age and gender. "The hazard ratio goes up as you’d expect," Dr. Low observed. "People in the anti-TNF cohort are younger, and there are more females [77% vs. 74% for nonbiologic DMARDs]."
After adjustment for risk factors, analysis gave a hazard ratio of 0.7 in favor of anti-TNFs reducing the risk for MI. The risk factors included disease-specific factors (age, gender, disease duration, disease activity, steroid exposure, number of previous nonbiologic DMARDs received, and year of entry into the study), as well as traditional cardiovascular risk factors (hypertension, diabetes, smoking, chronic obstructive pulmonary disease, antiplatelet therapy, and use of nonsteroidal anti-inflammatory drugs and COX-2 inhibitors). "Statins were also included in the adjustment," Dr. Low confirmed.
The possible reduction in the risk for heart attack is another potential benefit of anti-TNFs that can be discussed with patients, she suggested, although patients would of course not be put on these drugs in order to reduce their cardiovascular risk.
Why anti-TNFs have this effect is still not clear. Dr. Low said. "It’s going to be very difficult to tease out whether it’s the inflammation that has been reduced by the anti-TNF therapy or whether anti-TNF therapy affects other factors, such as hypertension and lipids, but I don’t think we can say that for certain from this analysis.
Further research will look at the effect of anti-TNFs on the severity of MI to see if there is any difference from nonbiologic DMARDs.
The BSRBR is funded by a grant from the British Society for Rheumatology (BSR), which receives funding from multiple drug companies. This income finances a wholly separate contract between the BSR and the University of Manchester to host the BSRBR. Dr. Low had no conflicts of interest.
BIRMINGHAM, ENGLAND – The risk of heart attack appears to be lower in patients treated with anti–tumor necrosis factor therapy than with conventional disease-modifying antirheumatic drugs.
Patients treated with the biologic agents had a 30% lower risk for having a myocardial infarction, based on data from the British Society for Rheumatology Biologics Register (BSRBR) for rheumatoid arthritis. However, no differences were detected in 30-day or 1-year mortality rates (adjusted hazard ratios of 0.9 and 0.97, respectively).
"There seems to be a signal that subjects ever exposed to anti-TNF therapy were potentially at reduced risk for developing an MI," Dr. Audrey Low said in an interview at the British Society for Rheumatology annual conference.
Dr. Low, a clinical research fellow in the Arthritis Research UK Epidemiology Unit, University of Manchester, England, explained that traditional risk factors do not fully account for the well-known increased risk for cardiovascular disease in patients with rheumatoid arthritis. Underlying inflammation might play a role, and it was hypothesized that anti-TNF therapy might help to reduce this inflammation, with subsequent cardiovascular benefits.
The BSRBR-RA is one of the largest biologics registers in the world and has been running for more than 10 years. The register currently includes data on more than 20,000 participants treated with either anti-TNF agents or nonbiologic disease-modifying antirheumatic drugs (DMARDs), such as methotrexate.
The present study used data collated between 2001 and 2008 on 11,536 patients treated with anti-TNF drugs licensed at the time in the United Kingdom (etanercept, infliximab, and adalimumab), and 3,225 patients who received nonbiologic DMARDs. Patients with prior MI or angina were excluded, and patients were followed up through April 2010 or until death, incident MI, or date of last clinician assessment, whichever came first.
Patients treated with anti-TNF therapy were younger than those given nonbiologic DMARDs, with a mean age of 56 years versus 60 years at recruitment into the BSRBR-RA. They also tended to have higher disease activity and longer disease duration (11 years vs. 6 years). There were similar percentages of patients with hypertension (29% vs. 32%), diabetes (6% for both), and a history of ever having smoked (59% vs. 62%).
There were 224 incident MIs in the anti-TNF–treated group at a median follow-up of 6 years per person, and 52 heart attacks in the nonbiologic DMARD–treated group at a median follow-up of 4 years per person. "Even though the study has been running for over 10 years, the amount of follow-up per person is between 4 and 6 years," Dr. Low observed.
The unadjusted hazard ratio for MI risk was 0.9, and it increased to 1.2 when adjusted for age and gender. "The hazard ratio goes up as you’d expect," Dr. Low observed. "People in the anti-TNF cohort are younger, and there are more females [77% vs. 74% for nonbiologic DMARDs]."
After adjustment for risk factors, analysis gave a hazard ratio of 0.7 in favor of anti-TNFs reducing the risk for MI. The risk factors included disease-specific factors (age, gender, disease duration, disease activity, steroid exposure, number of previous nonbiologic DMARDs received, and year of entry into the study), as well as traditional cardiovascular risk factors (hypertension, diabetes, smoking, chronic obstructive pulmonary disease, antiplatelet therapy, and use of nonsteroidal anti-inflammatory drugs and COX-2 inhibitors). "Statins were also included in the adjustment," Dr. Low confirmed.
The possible reduction in the risk for heart attack is another potential benefit of anti-TNFs that can be discussed with patients, she suggested, although patients would of course not be put on these drugs in order to reduce their cardiovascular risk.
Why anti-TNFs have this effect is still not clear. Dr. Low said. "It’s going to be very difficult to tease out whether it’s the inflammation that has been reduced by the anti-TNF therapy or whether anti-TNF therapy affects other factors, such as hypertension and lipids, but I don’t think we can say that for certain from this analysis.
Further research will look at the effect of anti-TNFs on the severity of MI to see if there is any difference from nonbiologic DMARDs.
The BSRBR is funded by a grant from the British Society for Rheumatology (BSR), which receives funding from multiple drug companies. This income finances a wholly separate contract between the BSR and the University of Manchester to host the BSRBR. Dr. Low had no conflicts of interest.
BIRMINGHAM, ENGLAND – The risk of heart attack appears to be lower in patients treated with anti–tumor necrosis factor therapy than with conventional disease-modifying antirheumatic drugs.
Patients treated with the biologic agents had a 30% lower risk for having a myocardial infarction, based on data from the British Society for Rheumatology Biologics Register (BSRBR) for rheumatoid arthritis. However, no differences were detected in 30-day or 1-year mortality rates (adjusted hazard ratios of 0.9 and 0.97, respectively).
"There seems to be a signal that subjects ever exposed to anti-TNF therapy were potentially at reduced risk for developing an MI," Dr. Audrey Low said in an interview at the British Society for Rheumatology annual conference.
Dr. Low, a clinical research fellow in the Arthritis Research UK Epidemiology Unit, University of Manchester, England, explained that traditional risk factors do not fully account for the well-known increased risk for cardiovascular disease in patients with rheumatoid arthritis. Underlying inflammation might play a role, and it was hypothesized that anti-TNF therapy might help to reduce this inflammation, with subsequent cardiovascular benefits.
The BSRBR-RA is one of the largest biologics registers in the world and has been running for more than 10 years. The register currently includes data on more than 20,000 participants treated with either anti-TNF agents or nonbiologic disease-modifying antirheumatic drugs (DMARDs), such as methotrexate.
The present study used data collated between 2001 and 2008 on 11,536 patients treated with anti-TNF drugs licensed at the time in the United Kingdom (etanercept, infliximab, and adalimumab), and 3,225 patients who received nonbiologic DMARDs. Patients with prior MI or angina were excluded, and patients were followed up through April 2010 or until death, incident MI, or date of last clinician assessment, whichever came first.
Patients treated with anti-TNF therapy were younger than those given nonbiologic DMARDs, with a mean age of 56 years versus 60 years at recruitment into the BSRBR-RA. They also tended to have higher disease activity and longer disease duration (11 years vs. 6 years). There were similar percentages of patients with hypertension (29% vs. 32%), diabetes (6% for both), and a history of ever having smoked (59% vs. 62%).
There were 224 incident MIs in the anti-TNF–treated group at a median follow-up of 6 years per person, and 52 heart attacks in the nonbiologic DMARD–treated group at a median follow-up of 4 years per person. "Even though the study has been running for over 10 years, the amount of follow-up per person is between 4 and 6 years," Dr. Low observed.
The unadjusted hazard ratio for MI risk was 0.9, and it increased to 1.2 when adjusted for age and gender. "The hazard ratio goes up as you’d expect," Dr. Low observed. "People in the anti-TNF cohort are younger, and there are more females [77% vs. 74% for nonbiologic DMARDs]."
After adjustment for risk factors, analysis gave a hazard ratio of 0.7 in favor of anti-TNFs reducing the risk for MI. The risk factors included disease-specific factors (age, gender, disease duration, disease activity, steroid exposure, number of previous nonbiologic DMARDs received, and year of entry into the study), as well as traditional cardiovascular risk factors (hypertension, diabetes, smoking, chronic obstructive pulmonary disease, antiplatelet therapy, and use of nonsteroidal anti-inflammatory drugs and COX-2 inhibitors). "Statins were also included in the adjustment," Dr. Low confirmed.
The possible reduction in the risk for heart attack is another potential benefit of anti-TNFs that can be discussed with patients, she suggested, although patients would of course not be put on these drugs in order to reduce their cardiovascular risk.
Why anti-TNFs have this effect is still not clear. Dr. Low said. "It’s going to be very difficult to tease out whether it’s the inflammation that has been reduced by the anti-TNF therapy or whether anti-TNF therapy affects other factors, such as hypertension and lipids, but I don’t think we can say that for certain from this analysis.
Further research will look at the effect of anti-TNFs on the severity of MI to see if there is any difference from nonbiologic DMARDs.
The BSRBR is funded by a grant from the British Society for Rheumatology (BSR), which receives funding from multiple drug companies. This income finances a wholly separate contract between the BSR and the University of Manchester to host the BSRBR. Dr. Low had no conflicts of interest.
AT RHEUMATOLOGY 2013
Major finding: The fully adjusted hazard ratio for myocardial infarction was 0.7 in favor of ever using anti-TNF therapy.
Data source: The British Society for Rheumatology Biologics Register (BSRBR) for rheumatoid arthritis, consisting of 11,536 patients treated with anti-TNF therapy and 3,225 patients treated with traditional, nonbiologic disease-modifying antirheumatic drugs.
Disclosures: The BSRBR is funded by a grant from the British Society for Rheumatology (BSR), which receives funding from multiple drug companies. This income finances a wholly separate contract between the BSR and the University of Manchester to host the BSRBR. Dr. Low had no conflicts of interest.
Preimmunosuppresive hepatitis B screening often goes by the wayside
NEW YORK – While nowadays there is less likelihood that the hepatitis B virus causes rheumatic disease, major changes in epidemiology and drug therapy, especially the use of immunosuppressives, have made reactivation of the virus during the course of rheumatic disease treatment a major public health concern, according to Dr. Leonard H. Calabrese.
"HBV reactivation has been well known in cancer and transplantation but is now really coming to the fore in autoimmune and autoinflammatory diseases. It may be mild (asymptomatic viremia) or severe (fulminant liver failure and death) and has been observed in patients with both chronic infection or resolved disease. While it is largely preventable, unfortunately many physicians who regularly prescribe immunosuppressive therapies – including rheumatologists, oncologists, gastroenterologists, and dermatologists – do not recognize this potential," Dr. Calabrese said at a meeting sponsored by New York University.
The major risk of HBV is not during but after immunosuppression, when reactivation leading to severe or fatal hepatitis can occur. Reactivations have been reported rarely with low-dose prednisone and methotrexate, as well as all tumor necrosis factor (TNF) inhibitors, rituximab, abatacept, and infliximab, prompting a black box warning for many biologics, said Dr. Calabrese, director of the Fasenmyer Center for Clinical Immunology at the Cleveland Clinic.
In a retrospective study of 257 patients with positive HBV markers who were treated with TNF-targeted therapies, 39% with chronic infections (as indicated by the presence of HB surface antigens) developed HBV reactivation. Five developed acute liver failure, and four died (Medicine 2011;90:359-71). "This is not a trivial risk," Dr. Calabrese said.
Yet many rheumatologists do not screen for HBV prior to prescribing immunosuppressive therapy, he said. Dr. Calabrese cited a 2010 survey of rheumatologists that found that only 42% routinely screen for HBV before a patient begins nonbiologic disease-modifying antirheumatic drug therapy and 69% screen before prescribing a biologic DMARD. Seven percent of rheumatologists reported witnessing a reactivation event (Arthritis Care Res. 2010;62:704-11).
Of the three serologic markers for HBV (the hepatitis B surface antigen [HBsAg], the total hepatitis B core antibody [anti-HBc]), and the hepatitis B surface antibody (anti-HBsAb), Dr. Calabrese said he prefers HBsAg. "If you remember anything from this talk, remember that all patients with hepatitis B surface antigen need to be tended to before beginning any rheumatic therapy," he said. All positive tests should be followed up with HBV DNA testing.
Although all patients with a history of HBV are at some risk for HBV reactivation, with or without the presence of serologic markers, Dr. Calabrese outlined the relative risks of HBV reactivation as indicated by serologic findings. Those at highest risk are patients with chronic HBV infection (HBsAg positive, high level of HBV DNA), followed by inactive carriers (HBsAg positive, some HBV DNA) and those with resolved HBV (HBsAg negative, anti-HBc positive, anti-HBsAb positive). Those at lowest risk have resolved HBV (anti-HBc alone).
Rheumatologists should also be aware of the increased chance of HBV reactivation in high-risk populations. While in urban areas HBV is known to be associated with drug abuse, there is also a heightened risk in immigrants, especially those from Asia. Asian Americans are 2.7 times more likely to develop hepatocellular carcinoma. According to Dr. Calabrese, in mainland China there are an estimated 1 million people with rheumatologic problems who manifest chronic HBsAg positivity. "This is a global health problem," he noted.
Dr. Calabrese recommended that preemptive therapeutic measures be taken for all HBsAg-positive and HBV DNA–positive patients, including possible lifelong antiviral medication, and then these patients can be treated for their rheumatologic issues. Those who test positive for anti-HBc should be carefully monitored. "There should not be a patient with spondylitis, rheumatoid arthritis, lupus, or vasculitis who is getting less than effective therapy because they have chronic HBV," he said.
Dr. Calabrese reported no relevant financial disclosures.
NEW YORK – While nowadays there is less likelihood that the hepatitis B virus causes rheumatic disease, major changes in epidemiology and drug therapy, especially the use of immunosuppressives, have made reactivation of the virus during the course of rheumatic disease treatment a major public health concern, according to Dr. Leonard H. Calabrese.
"HBV reactivation has been well known in cancer and transplantation but is now really coming to the fore in autoimmune and autoinflammatory diseases. It may be mild (asymptomatic viremia) or severe (fulminant liver failure and death) and has been observed in patients with both chronic infection or resolved disease. While it is largely preventable, unfortunately many physicians who regularly prescribe immunosuppressive therapies – including rheumatologists, oncologists, gastroenterologists, and dermatologists – do not recognize this potential," Dr. Calabrese said at a meeting sponsored by New York University.
The major risk of HBV is not during but after immunosuppression, when reactivation leading to severe or fatal hepatitis can occur. Reactivations have been reported rarely with low-dose prednisone and methotrexate, as well as all tumor necrosis factor (TNF) inhibitors, rituximab, abatacept, and infliximab, prompting a black box warning for many biologics, said Dr. Calabrese, director of the Fasenmyer Center for Clinical Immunology at the Cleveland Clinic.
In a retrospective study of 257 patients with positive HBV markers who were treated with TNF-targeted therapies, 39% with chronic infections (as indicated by the presence of HB surface antigens) developed HBV reactivation. Five developed acute liver failure, and four died (Medicine 2011;90:359-71). "This is not a trivial risk," Dr. Calabrese said.
Yet many rheumatologists do not screen for HBV prior to prescribing immunosuppressive therapy, he said. Dr. Calabrese cited a 2010 survey of rheumatologists that found that only 42% routinely screen for HBV before a patient begins nonbiologic disease-modifying antirheumatic drug therapy and 69% screen before prescribing a biologic DMARD. Seven percent of rheumatologists reported witnessing a reactivation event (Arthritis Care Res. 2010;62:704-11).
Of the three serologic markers for HBV (the hepatitis B surface antigen [HBsAg], the total hepatitis B core antibody [anti-HBc]), and the hepatitis B surface antibody (anti-HBsAb), Dr. Calabrese said he prefers HBsAg. "If you remember anything from this talk, remember that all patients with hepatitis B surface antigen need to be tended to before beginning any rheumatic therapy," he said. All positive tests should be followed up with HBV DNA testing.
Although all patients with a history of HBV are at some risk for HBV reactivation, with or without the presence of serologic markers, Dr. Calabrese outlined the relative risks of HBV reactivation as indicated by serologic findings. Those at highest risk are patients with chronic HBV infection (HBsAg positive, high level of HBV DNA), followed by inactive carriers (HBsAg positive, some HBV DNA) and those with resolved HBV (HBsAg negative, anti-HBc positive, anti-HBsAb positive). Those at lowest risk have resolved HBV (anti-HBc alone).
Rheumatologists should also be aware of the increased chance of HBV reactivation in high-risk populations. While in urban areas HBV is known to be associated with drug abuse, there is also a heightened risk in immigrants, especially those from Asia. Asian Americans are 2.7 times more likely to develop hepatocellular carcinoma. According to Dr. Calabrese, in mainland China there are an estimated 1 million people with rheumatologic problems who manifest chronic HBsAg positivity. "This is a global health problem," he noted.
Dr. Calabrese recommended that preemptive therapeutic measures be taken for all HBsAg-positive and HBV DNA–positive patients, including possible lifelong antiviral medication, and then these patients can be treated for their rheumatologic issues. Those who test positive for anti-HBc should be carefully monitored. "There should not be a patient with spondylitis, rheumatoid arthritis, lupus, or vasculitis who is getting less than effective therapy because they have chronic HBV," he said.
Dr. Calabrese reported no relevant financial disclosures.
NEW YORK – While nowadays there is less likelihood that the hepatitis B virus causes rheumatic disease, major changes in epidemiology and drug therapy, especially the use of immunosuppressives, have made reactivation of the virus during the course of rheumatic disease treatment a major public health concern, according to Dr. Leonard H. Calabrese.
"HBV reactivation has been well known in cancer and transplantation but is now really coming to the fore in autoimmune and autoinflammatory diseases. It may be mild (asymptomatic viremia) or severe (fulminant liver failure and death) and has been observed in patients with both chronic infection or resolved disease. While it is largely preventable, unfortunately many physicians who regularly prescribe immunosuppressive therapies – including rheumatologists, oncologists, gastroenterologists, and dermatologists – do not recognize this potential," Dr. Calabrese said at a meeting sponsored by New York University.
The major risk of HBV is not during but after immunosuppression, when reactivation leading to severe or fatal hepatitis can occur. Reactivations have been reported rarely with low-dose prednisone and methotrexate, as well as all tumor necrosis factor (TNF) inhibitors, rituximab, abatacept, and infliximab, prompting a black box warning for many biologics, said Dr. Calabrese, director of the Fasenmyer Center for Clinical Immunology at the Cleveland Clinic.
In a retrospective study of 257 patients with positive HBV markers who were treated with TNF-targeted therapies, 39% with chronic infections (as indicated by the presence of HB surface antigens) developed HBV reactivation. Five developed acute liver failure, and four died (Medicine 2011;90:359-71). "This is not a trivial risk," Dr. Calabrese said.
Yet many rheumatologists do not screen for HBV prior to prescribing immunosuppressive therapy, he said. Dr. Calabrese cited a 2010 survey of rheumatologists that found that only 42% routinely screen for HBV before a patient begins nonbiologic disease-modifying antirheumatic drug therapy and 69% screen before prescribing a biologic DMARD. Seven percent of rheumatologists reported witnessing a reactivation event (Arthritis Care Res. 2010;62:704-11).
Of the three serologic markers for HBV (the hepatitis B surface antigen [HBsAg], the total hepatitis B core antibody [anti-HBc]), and the hepatitis B surface antibody (anti-HBsAb), Dr. Calabrese said he prefers HBsAg. "If you remember anything from this talk, remember that all patients with hepatitis B surface antigen need to be tended to before beginning any rheumatic therapy," he said. All positive tests should be followed up with HBV DNA testing.
Although all patients with a history of HBV are at some risk for HBV reactivation, with or without the presence of serologic markers, Dr. Calabrese outlined the relative risks of HBV reactivation as indicated by serologic findings. Those at highest risk are patients with chronic HBV infection (HBsAg positive, high level of HBV DNA), followed by inactive carriers (HBsAg positive, some HBV DNA) and those with resolved HBV (HBsAg negative, anti-HBc positive, anti-HBsAb positive). Those at lowest risk have resolved HBV (anti-HBc alone).
Rheumatologists should also be aware of the increased chance of HBV reactivation in high-risk populations. While in urban areas HBV is known to be associated with drug abuse, there is also a heightened risk in immigrants, especially those from Asia. Asian Americans are 2.7 times more likely to develop hepatocellular carcinoma. According to Dr. Calabrese, in mainland China there are an estimated 1 million people with rheumatologic problems who manifest chronic HBsAg positivity. "This is a global health problem," he noted.
Dr. Calabrese recommended that preemptive therapeutic measures be taken for all HBsAg-positive and HBV DNA–positive patients, including possible lifelong antiviral medication, and then these patients can be treated for their rheumatologic issues. Those who test positive for anti-HBc should be carefully monitored. "There should not be a patient with spondylitis, rheumatoid arthritis, lupus, or vasculitis who is getting less than effective therapy because they have chronic HBV," he said.
Dr. Calabrese reported no relevant financial disclosures.
AT THE NYU SEMINAR IN ADVANCED RHEUMATOLOGY
Intensified therapy can delay anti-TNF use for active RA
BIRMINGHAM, ENGLAND – Combinations of conventional antirheumatic drugs are as clinically effective as more expensive biologic agents in managing patients with active, established rheumatoid arthritis, randomized trial data suggest.
Health Assessment Questionnaire (HAQ) scores at 12 months were lower in patients given conventional disease-modifying antirheumatic drugs (cDMARDs) as compared to a tumor necrosis factor inhibitor (TNFi) after failure of methotrexate and at least one other DMARD (1.35 vs. 1.60, P = .046).
Nevertheless, the two approaches were "clinically equivalent," said trial investigator Dr. David L. Scott at the annual meeting of the British Society for Rheumatology.
"In patients with methotrexate-resistant rheumatoid arthritis [RA], giving the cDMARD intensive therapy and starting off with a TNFi achieves similar outcomes, causes comparable harms, but the cDMARDs cost less," said Dr. Scott, who is professor of clinical rheumatology at King’s College London.
These data, from the TACIT (Tumor Necrosis Factor Inhibitors or Conventional Drugs in Rheumatoid Arthritis) trial, suggest that combination DMARD therapy could be an alternative approach for patients with established RA, reserving anti-TNF agents for those who fail intensive DMARD treatment.
Dr. Scott noted that the findings might warrant a change in practice in the United Kingdom, where anti-TNFs are currently recommended as the next choice in patients who do not respond to treatment with two individual DMARDs, one of which must be methotrexate. Such an approach is based on evidence from placebo-controlled, rather than head-to-head, trials, however, and such a "one size fits all" approach perhaps needs reevaluation.
The TACIT trial was conducted in 24 centers in the United Kingdom and involved 205 patients who had RA for a median duration of 4 years. The aim of the trial was to directly compare two different treatment strategies: a TNFi (infliximab, etanercept, or adalimumab) given to patients after failing methotrexate and a subsequent DMARD versus methotrexate followed by combination cDMARDs and then a TNFi.
Combinations of cDMARDs used in the trial included triple therapy with methotrexate, sulfasalazine, and hydroxychloroquine; other methotrexate combinations (methotrexate-cyclosporin, methotrexate-leflunomide, and methotrexate-gold); and one sulfasalazine combination (sulfasalazine-leflunomide).
At 12 months’ follow-up, there was no significant difference between the treatment strategies in terms of quality of life measured using the EuroQol instrument, radiographic progression assessed using Larsen scores, or disease activity assessed via monthly Disease Activity Score-28 (DAS28).
The mean change in DAS28 score was initially greater in TNFi-treated patients (–2.07 vs. –1.45, P = .007) at 6 months, Dr. Scott said, but this difference was lost with longer follow-up. "This simply shows that, as we’ve always known, TNFi’s act rapidly and DMARDs are slow acting," he observed.
Economic analysis showed a clear advantage for the use of cDMARDs over anti-TNF agents, with an annual estimated saving of health and social care costs of £5,552 (U.S.$8,400) per patient. Taking lost productivity and social security payments into account did not change the findings.
Safety is a concern of using multiple cDMARDs, particularly older combinations, but the TACIT data showed that there were a comparable number of adverse reactions. The total number of events was 635 for cDMARDs and 465 in the TNFi group. Of these, there were more digestive adverse events in the cDMARD arm than in the TNFi group (148 vs. 60), but more infections in the anti-TNF-treated patients than in the cDMARD group (54 vs. 30).
Serious adverse events were noted in 10 and 18 of cDMARD- and TNFi-treated patients, respectively, with 10 and 6 patients in each group withdrawing as a result of experiencing a side effect. There was one death, due to pneumonia and multiple organ failure, in the anti-TNF arm.
"We should think about the treatment strategy in terms of intensive DMARDs first of all and then moving onto biologics," Dr. Scott said. Remission needs to be a target of treatment, he added, although this is rarely achieved by either strategy. Remission rates at 12 months seen in TACIT are comparable to rates reported from several registries, including CORRONA (Consortium of Rheumatology Researchers of North America) in the United States, DANBIO in Denmark, RABBIT in Germany, and BSRBR-RA (British Society for Rheumatology Biologics Register-Rheumatoid Arthritis) in the United Kingdom.
Dr. Scott concluded by noting that there are three unresolved questions. First, it is not clear what the best combination of cDMARDs is; second, the optimum way to use biologics is currently unknown; and third, it is not clear how to convert patients with intermediate disease activity into patients who may achieve clinical remission.
The TACIT trial was supported by a Health Technology Assessment grant from the U.K. National Institute for Health Research. Dr. Scott and his coinvestigators reported having no conflicts of interest.
BIRMINGHAM, ENGLAND – Combinations of conventional antirheumatic drugs are as clinically effective as more expensive biologic agents in managing patients with active, established rheumatoid arthritis, randomized trial data suggest.
Health Assessment Questionnaire (HAQ) scores at 12 months were lower in patients given conventional disease-modifying antirheumatic drugs (cDMARDs) as compared to a tumor necrosis factor inhibitor (TNFi) after failure of methotrexate and at least one other DMARD (1.35 vs. 1.60, P = .046).
Nevertheless, the two approaches were "clinically equivalent," said trial investigator Dr. David L. Scott at the annual meeting of the British Society for Rheumatology.
"In patients with methotrexate-resistant rheumatoid arthritis [RA], giving the cDMARD intensive therapy and starting off with a TNFi achieves similar outcomes, causes comparable harms, but the cDMARDs cost less," said Dr. Scott, who is professor of clinical rheumatology at King’s College London.
These data, from the TACIT (Tumor Necrosis Factor Inhibitors or Conventional Drugs in Rheumatoid Arthritis) trial, suggest that combination DMARD therapy could be an alternative approach for patients with established RA, reserving anti-TNF agents for those who fail intensive DMARD treatment.
Dr. Scott noted that the findings might warrant a change in practice in the United Kingdom, where anti-TNFs are currently recommended as the next choice in patients who do not respond to treatment with two individual DMARDs, one of which must be methotrexate. Such an approach is based on evidence from placebo-controlled, rather than head-to-head, trials, however, and such a "one size fits all" approach perhaps needs reevaluation.
The TACIT trial was conducted in 24 centers in the United Kingdom and involved 205 patients who had RA for a median duration of 4 years. The aim of the trial was to directly compare two different treatment strategies: a TNFi (infliximab, etanercept, or adalimumab) given to patients after failing methotrexate and a subsequent DMARD versus methotrexate followed by combination cDMARDs and then a TNFi.
Combinations of cDMARDs used in the trial included triple therapy with methotrexate, sulfasalazine, and hydroxychloroquine; other methotrexate combinations (methotrexate-cyclosporin, methotrexate-leflunomide, and methotrexate-gold); and one sulfasalazine combination (sulfasalazine-leflunomide).
At 12 months’ follow-up, there was no significant difference between the treatment strategies in terms of quality of life measured using the EuroQol instrument, radiographic progression assessed using Larsen scores, or disease activity assessed via monthly Disease Activity Score-28 (DAS28).
The mean change in DAS28 score was initially greater in TNFi-treated patients (–2.07 vs. –1.45, P = .007) at 6 months, Dr. Scott said, but this difference was lost with longer follow-up. "This simply shows that, as we’ve always known, TNFi’s act rapidly and DMARDs are slow acting," he observed.
Economic analysis showed a clear advantage for the use of cDMARDs over anti-TNF agents, with an annual estimated saving of health and social care costs of £5,552 (U.S.$8,400) per patient. Taking lost productivity and social security payments into account did not change the findings.
Safety is a concern of using multiple cDMARDs, particularly older combinations, but the TACIT data showed that there were a comparable number of adverse reactions. The total number of events was 635 for cDMARDs and 465 in the TNFi group. Of these, there were more digestive adverse events in the cDMARD arm than in the TNFi group (148 vs. 60), but more infections in the anti-TNF-treated patients than in the cDMARD group (54 vs. 30).
Serious adverse events were noted in 10 and 18 of cDMARD- and TNFi-treated patients, respectively, with 10 and 6 patients in each group withdrawing as a result of experiencing a side effect. There was one death, due to pneumonia and multiple organ failure, in the anti-TNF arm.
"We should think about the treatment strategy in terms of intensive DMARDs first of all and then moving onto biologics," Dr. Scott said. Remission needs to be a target of treatment, he added, although this is rarely achieved by either strategy. Remission rates at 12 months seen in TACIT are comparable to rates reported from several registries, including CORRONA (Consortium of Rheumatology Researchers of North America) in the United States, DANBIO in Denmark, RABBIT in Germany, and BSRBR-RA (British Society for Rheumatology Biologics Register-Rheumatoid Arthritis) in the United Kingdom.
Dr. Scott concluded by noting that there are three unresolved questions. First, it is not clear what the best combination of cDMARDs is; second, the optimum way to use biologics is currently unknown; and third, it is not clear how to convert patients with intermediate disease activity into patients who may achieve clinical remission.
The TACIT trial was supported by a Health Technology Assessment grant from the U.K. National Institute for Health Research. Dr. Scott and his coinvestigators reported having no conflicts of interest.
BIRMINGHAM, ENGLAND – Combinations of conventional antirheumatic drugs are as clinically effective as more expensive biologic agents in managing patients with active, established rheumatoid arthritis, randomized trial data suggest.
Health Assessment Questionnaire (HAQ) scores at 12 months were lower in patients given conventional disease-modifying antirheumatic drugs (cDMARDs) as compared to a tumor necrosis factor inhibitor (TNFi) after failure of methotrexate and at least one other DMARD (1.35 vs. 1.60, P = .046).
Nevertheless, the two approaches were "clinically equivalent," said trial investigator Dr. David L. Scott at the annual meeting of the British Society for Rheumatology.
"In patients with methotrexate-resistant rheumatoid arthritis [RA], giving the cDMARD intensive therapy and starting off with a TNFi achieves similar outcomes, causes comparable harms, but the cDMARDs cost less," said Dr. Scott, who is professor of clinical rheumatology at King’s College London.
These data, from the TACIT (Tumor Necrosis Factor Inhibitors or Conventional Drugs in Rheumatoid Arthritis) trial, suggest that combination DMARD therapy could be an alternative approach for patients with established RA, reserving anti-TNF agents for those who fail intensive DMARD treatment.
Dr. Scott noted that the findings might warrant a change in practice in the United Kingdom, where anti-TNFs are currently recommended as the next choice in patients who do not respond to treatment with two individual DMARDs, one of which must be methotrexate. Such an approach is based on evidence from placebo-controlled, rather than head-to-head, trials, however, and such a "one size fits all" approach perhaps needs reevaluation.
The TACIT trial was conducted in 24 centers in the United Kingdom and involved 205 patients who had RA for a median duration of 4 years. The aim of the trial was to directly compare two different treatment strategies: a TNFi (infliximab, etanercept, or adalimumab) given to patients after failing methotrexate and a subsequent DMARD versus methotrexate followed by combination cDMARDs and then a TNFi.
Combinations of cDMARDs used in the trial included triple therapy with methotrexate, sulfasalazine, and hydroxychloroquine; other methotrexate combinations (methotrexate-cyclosporin, methotrexate-leflunomide, and methotrexate-gold); and one sulfasalazine combination (sulfasalazine-leflunomide).
At 12 months’ follow-up, there was no significant difference between the treatment strategies in terms of quality of life measured using the EuroQol instrument, radiographic progression assessed using Larsen scores, or disease activity assessed via monthly Disease Activity Score-28 (DAS28).
The mean change in DAS28 score was initially greater in TNFi-treated patients (–2.07 vs. –1.45, P = .007) at 6 months, Dr. Scott said, but this difference was lost with longer follow-up. "This simply shows that, as we’ve always known, TNFi’s act rapidly and DMARDs are slow acting," he observed.
Economic analysis showed a clear advantage for the use of cDMARDs over anti-TNF agents, with an annual estimated saving of health and social care costs of £5,552 (U.S.$8,400) per patient. Taking lost productivity and social security payments into account did not change the findings.
Safety is a concern of using multiple cDMARDs, particularly older combinations, but the TACIT data showed that there were a comparable number of adverse reactions. The total number of events was 635 for cDMARDs and 465 in the TNFi group. Of these, there were more digestive adverse events in the cDMARD arm than in the TNFi group (148 vs. 60), but more infections in the anti-TNF-treated patients than in the cDMARD group (54 vs. 30).
Serious adverse events were noted in 10 and 18 of cDMARD- and TNFi-treated patients, respectively, with 10 and 6 patients in each group withdrawing as a result of experiencing a side effect. There was one death, due to pneumonia and multiple organ failure, in the anti-TNF arm.
"We should think about the treatment strategy in terms of intensive DMARDs first of all and then moving onto biologics," Dr. Scott said. Remission needs to be a target of treatment, he added, although this is rarely achieved by either strategy. Remission rates at 12 months seen in TACIT are comparable to rates reported from several registries, including CORRONA (Consortium of Rheumatology Researchers of North America) in the United States, DANBIO in Denmark, RABBIT in Germany, and BSRBR-RA (British Society for Rheumatology Biologics Register-Rheumatoid Arthritis) in the United Kingdom.
Dr. Scott concluded by noting that there are three unresolved questions. First, it is not clear what the best combination of cDMARDs is; second, the optimum way to use biologics is currently unknown; and third, it is not clear how to convert patients with intermediate disease activity into patients who may achieve clinical remission.
The TACIT trial was supported by a Health Technology Assessment grant from the U.K. National Institute for Health Research. Dr. Scott and his coinvestigators reported having no conflicts of interest.
AT THE ANNUAL MEETING OF THE BRITISH SOCIETY FOR RHEUMATOLOGY
Major finding: At 12 months, HAQ scores were lower in patients treated with DMARDs than in patients treated with a TNF inhibitor (1.35 vs. 1.60; P = .046).
Data source: The TACIT multicenter, randomized, observer-blind trial of 205 patients with active, established RA eligible for anti-TNF therapy.
Disclosures: The TACIT trial was supported by a Health Technology Assessment grant from the U.K. National Institute for Health Research. Dr. Scott and his coinvestigators reported having no conflicts of interest.