User login
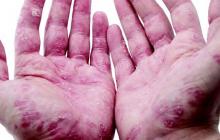
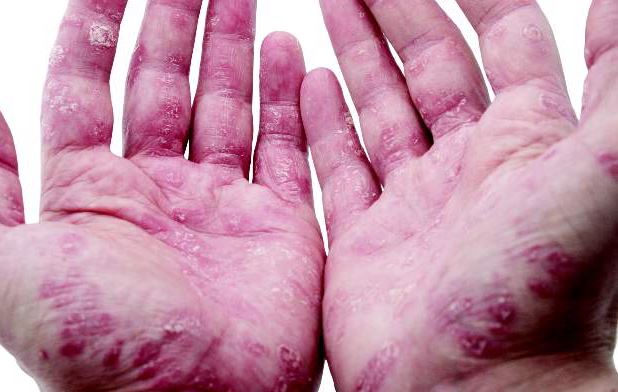
Debunking Psoriasis Myths: Can Diet Clear Psoriasis?
Myth: Psoriasis Can Be Treated By Eating or Avoiding Certain Foods
Patients who Google “diet and psoriasis” are flooded with search results of diets claiming to cure psoriasis. This misinformation is dangerous for patients, as there is no scientific evidence that any specific psoriasis diet can treat the condition. Patients may wish to improve their diet to prevent comorbidities such as cardiovascular disease and metabolic syndrome. Even though it may not be a cure, encouraging patients to eat healthy is never a bad thing.
In a 2014 analysis of psoriasis, obesity, body mass index (BMI), and diet literature, an increased risk for psoriasis development in the setting of obesity was discussed. There is evidence suggesting that a BMI greater than 30 kg/m2 may potentially play a role in the ability to achieve a full therapeutic effect of psoriasis therapy. “This could be for two possible reasons,” Debbaneh et al reported. “It may be a consequence of decreased drug distribution into the body due to increased body mass, or it may be a consequence of increased pro-inflammatory cytokine release as a result of increased adipocyte count.” However, this finding may be treatment specific. For example, higher body weight was an independent predictor of response to ustekinumab, providing the rationale for offering 2 weight-based dosing regimens of the drug. Overweight and obese patients also were less likely to experience clearance with adalimumab. However, studies have found no association between BMI and biologic treatment.
Weight loss through a low-calorie diet has been reported to achieve a greater reduction in psoriasis severity and a slower rebound of disease. “Interestingly, studies have shown that caloric restriction in obese subjects lowers the level of circulating inflammatory cytokines,” reported Debbaneh et al. “This may contribute to the observed beneficial effect in psoriatic disease.”
In patients whose disease has had a significant impact on quality of life, it is important that they are consulting resources online that will help them maintain a healthy lifestyle. The National Psoriasis Foundation provides useful information on diet and psoriasis, emphasizing that diet is not going to cure psoriatic disease but eating healthier can only help.
Expert Commentary
Many of my psoriasis patients ask me what should they avoid eating to prevent the psoriasis from worsening, or what did they eat to cause psoriasis to occur in the first place. I stress to patients that what they eat is not likely the cause of their psoriasis nor will avoiding certain foods prevent a flare. However, alcohol use may induce a psoriasis flare.
—Jashin J. Wu, MD (Los Angeles, California)
Debbaneh M, Millsop JW, Bhatia BK, et al. Diet and psoriasis: part I. impact of weight loss interventions. J Am Acad Dermatol. 2014;71:133-140.
National Psoriasis Foundation. Diet and psoriasis. https://www.psoriasis.org/about-psoriasis/treatments/alternative/diet-supplements. Accessed June 20, 2016.
Myth: Psoriasis Can Be Treated By Eating or Avoiding Certain Foods
Patients who Google “diet and psoriasis” are flooded with search results of diets claiming to cure psoriasis. This misinformation is dangerous for patients, as there is no scientific evidence that any specific psoriasis diet can treat the condition. Patients may wish to improve their diet to prevent comorbidities such as cardiovascular disease and metabolic syndrome. Even though it may not be a cure, encouraging patients to eat healthy is never a bad thing.
In a 2014 analysis of psoriasis, obesity, body mass index (BMI), and diet literature, an increased risk for psoriasis development in the setting of obesity was discussed. There is evidence suggesting that a BMI greater than 30 kg/m2 may potentially play a role in the ability to achieve a full therapeutic effect of psoriasis therapy. “This could be for two possible reasons,” Debbaneh et al reported. “It may be a consequence of decreased drug distribution into the body due to increased body mass, or it may be a consequence of increased pro-inflammatory cytokine release as a result of increased adipocyte count.” However, this finding may be treatment specific. For example, higher body weight was an independent predictor of response to ustekinumab, providing the rationale for offering 2 weight-based dosing regimens of the drug. Overweight and obese patients also were less likely to experience clearance with adalimumab. However, studies have found no association between BMI and biologic treatment.
Weight loss through a low-calorie diet has been reported to achieve a greater reduction in psoriasis severity and a slower rebound of disease. “Interestingly, studies have shown that caloric restriction in obese subjects lowers the level of circulating inflammatory cytokines,” reported Debbaneh et al. “This may contribute to the observed beneficial effect in psoriatic disease.”
In patients whose disease has had a significant impact on quality of life, it is important that they are consulting resources online that will help them maintain a healthy lifestyle. The National Psoriasis Foundation provides useful information on diet and psoriasis, emphasizing that diet is not going to cure psoriatic disease but eating healthier can only help.
Expert Commentary
Many of my psoriasis patients ask me what should they avoid eating to prevent the psoriasis from worsening, or what did they eat to cause psoriasis to occur in the first place. I stress to patients that what they eat is not likely the cause of their psoriasis nor will avoiding certain foods prevent a flare. However, alcohol use may induce a psoriasis flare.
—Jashin J. Wu, MD (Los Angeles, California)
Myth: Psoriasis Can Be Treated By Eating or Avoiding Certain Foods
Patients who Google “diet and psoriasis” are flooded with search results of diets claiming to cure psoriasis. This misinformation is dangerous for patients, as there is no scientific evidence that any specific psoriasis diet can treat the condition. Patients may wish to improve their diet to prevent comorbidities such as cardiovascular disease and metabolic syndrome. Even though it may not be a cure, encouraging patients to eat healthy is never a bad thing.
In a 2014 analysis of psoriasis, obesity, body mass index (BMI), and diet literature, an increased risk for psoriasis development in the setting of obesity was discussed. There is evidence suggesting that a BMI greater than 30 kg/m2 may potentially play a role in the ability to achieve a full therapeutic effect of psoriasis therapy. “This could be for two possible reasons,” Debbaneh et al reported. “It may be a consequence of decreased drug distribution into the body due to increased body mass, or it may be a consequence of increased pro-inflammatory cytokine release as a result of increased adipocyte count.” However, this finding may be treatment specific. For example, higher body weight was an independent predictor of response to ustekinumab, providing the rationale for offering 2 weight-based dosing regimens of the drug. Overweight and obese patients also were less likely to experience clearance with adalimumab. However, studies have found no association between BMI and biologic treatment.
Weight loss through a low-calorie diet has been reported to achieve a greater reduction in psoriasis severity and a slower rebound of disease. “Interestingly, studies have shown that caloric restriction in obese subjects lowers the level of circulating inflammatory cytokines,” reported Debbaneh et al. “This may contribute to the observed beneficial effect in psoriatic disease.”
In patients whose disease has had a significant impact on quality of life, it is important that they are consulting resources online that will help them maintain a healthy lifestyle. The National Psoriasis Foundation provides useful information on diet and psoriasis, emphasizing that diet is not going to cure psoriatic disease but eating healthier can only help.
Expert Commentary
Many of my psoriasis patients ask me what should they avoid eating to prevent the psoriasis from worsening, or what did they eat to cause psoriasis to occur in the first place. I stress to patients that what they eat is not likely the cause of their psoriasis nor will avoiding certain foods prevent a flare. However, alcohol use may induce a psoriasis flare.
—Jashin J. Wu, MD (Los Angeles, California)
Debbaneh M, Millsop JW, Bhatia BK, et al. Diet and psoriasis: part I. impact of weight loss interventions. J Am Acad Dermatol. 2014;71:133-140.
National Psoriasis Foundation. Diet and psoriasis. https://www.psoriasis.org/about-psoriasis/treatments/alternative/diet-supplements. Accessed June 20, 2016.
Debbaneh M, Millsop JW, Bhatia BK, et al. Diet and psoriasis: part I. impact of weight loss interventions. J Am Acad Dermatol. 2014;71:133-140.
National Psoriasis Foundation. Diet and psoriasis. https://www.psoriasis.org/about-psoriasis/treatments/alternative/diet-supplements. Accessed June 20, 2016.
Debunking Psoriasis Myths: Do Biologics Cause Cancer?
Myth: Biologics Cause Cancer
Biologics generally are safe and well-tolerated therapies; however, due to their immunosuppressive properties, the risk for lymphoma has been of potential concern, leading patients to believe that biologics cause cancer. The risk of some cancers, including some solid cancers, hematologic cancers, and skin cancers, appears to be increased in patients with psoriasis, possibly associated with chronic inflammation. Some psoriasis therapies may increase the risk for malignancy, including phototherapy with psoralen plus UVA, cyclosporine, and methotrexate.
Many studies have supported a favorable safety profile for biologics in terms of the risk for developing malignancy. In a 2015 analysis of 12,093 patients enrolled in PSOLAR (Psoriasis Longitudinal Assessment and Registry), none of the biologics were found to be associated with increased risk for malignancy.
In psoriasis patients with existing or prior malignancies, the benefits of biologic therapy to improve quality of life often outweigh the negligible risks for malignancy. However, coordinated care with oncology is recommended for psoriasis patients with a history of prior malignancy.
General recommendations from the American Academy of Dermatology indicate one should carefully consider the decision to use a tumor necrosis factor (TNF) antagonist in patients with a history of malignancy, particularly lymphoma. Short-term treatment with biologics (up to 4 years) appears to be safe with respect to lymphoma risk, especially with TNF-α inhibitors. The potential risk for melanoma, cutaneous T-cell lymphoma, and nonmelanoma skin cancer in patients treated with TNF inhibitors also has been raised.
Expert Commentary
OBSERVE-5 was a 5-year phase 4, prospective, multicenter surveillance registry of 2510 psoriasis patients with at least a baseline dose of etanercept (Kimball et al, 2015). There was no increased risk for cancer when compared to the Truven Health MarketScan database, which is a proxy for the general population.
ESPRIT is an ongoing, 10-year, international, prospective, observational registry of 6059 psoriasis patients with at least a baseline dose of adalimumab (Menter et al). There are no signals of increased risk for cancer.
We do not have enough numbers of psoriasis patients on secukinumab or ixekizumab yet, but their phase 3 trials also do not seem to indicate an increased risk for cancer.
—Jashin J. Wu, MD (Los Angeles, California)
American Academy of Dermatology. Psoriasis: TNF inhibitors general recommendations. https://www.aad.org/practice-tools/quality-care/clinical-guidelines/psoriasis/biologics/tnf-inhibitors-recommendations. Accessed June 14, 2016.
Dommasch E, Gelfand JM. Is there truly a risk of lymphoma from biologic therapies? Dermatol Ther. 2009;22:418-430.
Kimball AB, Rothman KJ, Kricorian G, et al. OBSERVE-5: Observational postmarketing safety surveillance registry of etanercept for the treatment of psoriasis final 5-year results. J Am Acad Dermatol. 2015;72:115-122.
Kimball AB, Schenfeld J, Accortt NA, et al. 5-year incidence rates of malignancies in psoriasis patients compared with the general US population. Poster presented at: Summer Meeting of the American Academy of Dermatology; August 6-10, 2014; Chicago, IL.
Menter A, Thaçi D, Papp KA, et al. Five-year analysis from the ESPRIT 10-year postmarketing surveillance registry of adalimumab treatment for moderate to severe psoriasis. J Am Acad Dermatol. 2015;73:410-419.e6.
Papp K, Gottlieb AB, Naldi L, et al. Safety surveillance for ustekinumab and other psoriasis treatments from the Psoriasis Longitudinal Assessment and Registry (PSOLAR). J Drugs Dermatol. 2015;14:706-714.
Patel S, Patel T, Kerdel FA. The risk of malignancy or progression of existing malignancy in patients with psoriasis treated with biologics: case report and review of the literature. Int J Dermatol. 2016;55:487-493.
Myth: Biologics Cause Cancer
Biologics generally are safe and well-tolerated therapies; however, due to their immunosuppressive properties, the risk for lymphoma has been of potential concern, leading patients to believe that biologics cause cancer. The risk of some cancers, including some solid cancers, hematologic cancers, and skin cancers, appears to be increased in patients with psoriasis, possibly associated with chronic inflammation. Some psoriasis therapies may increase the risk for malignancy, including phototherapy with psoralen plus UVA, cyclosporine, and methotrexate.
Many studies have supported a favorable safety profile for biologics in terms of the risk for developing malignancy. In a 2015 analysis of 12,093 patients enrolled in PSOLAR (Psoriasis Longitudinal Assessment and Registry), none of the biologics were found to be associated with increased risk for malignancy.
In psoriasis patients with existing or prior malignancies, the benefits of biologic therapy to improve quality of life often outweigh the negligible risks for malignancy. However, coordinated care with oncology is recommended for psoriasis patients with a history of prior malignancy.
General recommendations from the American Academy of Dermatology indicate one should carefully consider the decision to use a tumor necrosis factor (TNF) antagonist in patients with a history of malignancy, particularly lymphoma. Short-term treatment with biologics (up to 4 years) appears to be safe with respect to lymphoma risk, especially with TNF-α inhibitors. The potential risk for melanoma, cutaneous T-cell lymphoma, and nonmelanoma skin cancer in patients treated with TNF inhibitors also has been raised.
Expert Commentary
OBSERVE-5 was a 5-year phase 4, prospective, multicenter surveillance registry of 2510 psoriasis patients with at least a baseline dose of etanercept (Kimball et al, 2015). There was no increased risk for cancer when compared to the Truven Health MarketScan database, which is a proxy for the general population.
ESPRIT is an ongoing, 10-year, international, prospective, observational registry of 6059 psoriasis patients with at least a baseline dose of adalimumab (Menter et al). There are no signals of increased risk for cancer.
We do not have enough numbers of psoriasis patients on secukinumab or ixekizumab yet, but their phase 3 trials also do not seem to indicate an increased risk for cancer.
—Jashin J. Wu, MD (Los Angeles, California)
Myth: Biologics Cause Cancer
Biologics generally are safe and well-tolerated therapies; however, due to their immunosuppressive properties, the risk for lymphoma has been of potential concern, leading patients to believe that biologics cause cancer. The risk of some cancers, including some solid cancers, hematologic cancers, and skin cancers, appears to be increased in patients with psoriasis, possibly associated with chronic inflammation. Some psoriasis therapies may increase the risk for malignancy, including phototherapy with psoralen plus UVA, cyclosporine, and methotrexate.
Many studies have supported a favorable safety profile for biologics in terms of the risk for developing malignancy. In a 2015 analysis of 12,093 patients enrolled in PSOLAR (Psoriasis Longitudinal Assessment and Registry), none of the biologics were found to be associated with increased risk for malignancy.
In psoriasis patients with existing or prior malignancies, the benefits of biologic therapy to improve quality of life often outweigh the negligible risks for malignancy. However, coordinated care with oncology is recommended for psoriasis patients with a history of prior malignancy.
General recommendations from the American Academy of Dermatology indicate one should carefully consider the decision to use a tumor necrosis factor (TNF) antagonist in patients with a history of malignancy, particularly lymphoma. Short-term treatment with biologics (up to 4 years) appears to be safe with respect to lymphoma risk, especially with TNF-α inhibitors. The potential risk for melanoma, cutaneous T-cell lymphoma, and nonmelanoma skin cancer in patients treated with TNF inhibitors also has been raised.
Expert Commentary
OBSERVE-5 was a 5-year phase 4, prospective, multicenter surveillance registry of 2510 psoriasis patients with at least a baseline dose of etanercept (Kimball et al, 2015). There was no increased risk for cancer when compared to the Truven Health MarketScan database, which is a proxy for the general population.
ESPRIT is an ongoing, 10-year, international, prospective, observational registry of 6059 psoriasis patients with at least a baseline dose of adalimumab (Menter et al). There are no signals of increased risk for cancer.
We do not have enough numbers of psoriasis patients on secukinumab or ixekizumab yet, but their phase 3 trials also do not seem to indicate an increased risk for cancer.
—Jashin J. Wu, MD (Los Angeles, California)
American Academy of Dermatology. Psoriasis: TNF inhibitors general recommendations. https://www.aad.org/practice-tools/quality-care/clinical-guidelines/psoriasis/biologics/tnf-inhibitors-recommendations. Accessed June 14, 2016.
Dommasch E, Gelfand JM. Is there truly a risk of lymphoma from biologic therapies? Dermatol Ther. 2009;22:418-430.
Kimball AB, Rothman KJ, Kricorian G, et al. OBSERVE-5: Observational postmarketing safety surveillance registry of etanercept for the treatment of psoriasis final 5-year results. J Am Acad Dermatol. 2015;72:115-122.
Kimball AB, Schenfeld J, Accortt NA, et al. 5-year incidence rates of malignancies in psoriasis patients compared with the general US population. Poster presented at: Summer Meeting of the American Academy of Dermatology; August 6-10, 2014; Chicago, IL.
Menter A, Thaçi D, Papp KA, et al. Five-year analysis from the ESPRIT 10-year postmarketing surveillance registry of adalimumab treatment for moderate to severe psoriasis. J Am Acad Dermatol. 2015;73:410-419.e6.
Papp K, Gottlieb AB, Naldi L, et al. Safety surveillance for ustekinumab and other psoriasis treatments from the Psoriasis Longitudinal Assessment and Registry (PSOLAR). J Drugs Dermatol. 2015;14:706-714.
Patel S, Patel T, Kerdel FA. The risk of malignancy or progression of existing malignancy in patients with psoriasis treated with biologics: case report and review of the literature. Int J Dermatol. 2016;55:487-493.
American Academy of Dermatology. Psoriasis: TNF inhibitors general recommendations. https://www.aad.org/practice-tools/quality-care/clinical-guidelines/psoriasis/biologics/tnf-inhibitors-recommendations. Accessed June 14, 2016.
Dommasch E, Gelfand JM. Is there truly a risk of lymphoma from biologic therapies? Dermatol Ther. 2009;22:418-430.
Kimball AB, Rothman KJ, Kricorian G, et al. OBSERVE-5: Observational postmarketing safety surveillance registry of etanercept for the treatment of psoriasis final 5-year results. J Am Acad Dermatol. 2015;72:115-122.
Kimball AB, Schenfeld J, Accortt NA, et al. 5-year incidence rates of malignancies in psoriasis patients compared with the general US population. Poster presented at: Summer Meeting of the American Academy of Dermatology; August 6-10, 2014; Chicago, IL.
Menter A, Thaçi D, Papp KA, et al. Five-year analysis from the ESPRIT 10-year postmarketing surveillance registry of adalimumab treatment for moderate to severe psoriasis. J Am Acad Dermatol. 2015;73:410-419.e6.
Papp K, Gottlieb AB, Naldi L, et al. Safety surveillance for ustekinumab and other psoriasis treatments from the Psoriasis Longitudinal Assessment and Registry (PSOLAR). J Drugs Dermatol. 2015;14:706-714.
Patel S, Patel T, Kerdel FA. The risk of malignancy or progression of existing malignancy in patients with psoriasis treated with biologics: case report and review of the literature. Int J Dermatol. 2016;55:487-493.
Study: TNF inhibitors improve extraintestinal IBD manifestations
SAN DIEGO – Tumor necrosis factor inhibitors improved the extraintestinal manifestations of inflammatory bowel disease (IBD) among more than half of affected patients, according to a national cohort study.
“The best response rates were for psoriasis, aphthous stomatitis, uveitis, and peripheral arthritis,” said Dr. Thomas Greuter of University Hospital in Zürich. Patients responded similarly whether they received oral infliximab or subcutaneous adalimumab or certolizumab, he noted.
IBD often is associated with debilitating disorders of the skin, joints, eyes, and hepatobiliary tract, but “due to the lack of randomized, controlled trials, the therapy of extraintestinal manifestations remains rather empirical,” Dr. Greuter said at the annual Digestive Disease Week.
To study the role of anti-TNF agents in treating these disorders, he and his associates analyzed data for 1,249 patients from the national Swiss IBD Cohort Study between 2006 and 2010. Patients were typically in their mid-30s and had lived with IBD for about 9 years, he said.
A total of 366 patients (29%) had at least one extraintestinal manifestation of IBD – most commonly peripheral arthritis (75%), followed by aphthous stomatitis (24%), and ankylosing spondylitis (22%). In all, 213 (58%) patients received at least one anti-TNF agent, and 40% received the prescription specifically for extraintestinal manifestations. Nearly two-thirds of the patients received infliximab, while 22% received adalimumab and 15% received certolizumab.
About 55% of patients improved on anti-TNF therapy over an average of 7 years of follow-up, Dr. Greuter and his associates reported. Among all three anti-TNF agents, response rates ranged from 100% for psoriasis, to 80% for erythema nodosum and stomatitis, to 73% for arthritis and uveitis, to 50% for pyoderma granulosum. Overall rates of improvement were slightly higher for infliximab than for the other two drugs, but “adalimumab and certolizumab were used mostly as a second or a third-line anti-TNF agent, and the response rate to a second or third-line treatment was lower than for the first one,” Dr. Greuter said. Some patients also received corticosteroids and immunomodulators, but excluding this subgroup had little effect on rates of response to anti-TNF therapy, he added.
Dr. Greuter also reported that 11 patients (about 5% of the cohort) developed 14 new extraintestinal manifestations after starting anti-TNF agents – usually peripheral arthritis, but also pyoderma granulosum, aphthous stomatitis, psoriasis, and uveitis. “We cannot say if this was primary, or a side effect of treatment,” he said. These disorders usually improved if patients stayed on their anti-TNF agent, he added.
About two-thirds of patients in the cohort were female, more than three-quarters had Crohn’s disease, 19% had ulcerative colitis, and 3% had indeterminate colitis, he noted.
A research grant from the Swiss National Science Foundation funded the study. Dr. Greuter had no disclosures.
SAN DIEGO – Tumor necrosis factor inhibitors improved the extraintestinal manifestations of inflammatory bowel disease (IBD) among more than half of affected patients, according to a national cohort study.
“The best response rates were for psoriasis, aphthous stomatitis, uveitis, and peripheral arthritis,” said Dr. Thomas Greuter of University Hospital in Zürich. Patients responded similarly whether they received oral infliximab or subcutaneous adalimumab or certolizumab, he noted.
IBD often is associated with debilitating disorders of the skin, joints, eyes, and hepatobiliary tract, but “due to the lack of randomized, controlled trials, the therapy of extraintestinal manifestations remains rather empirical,” Dr. Greuter said at the annual Digestive Disease Week.
To study the role of anti-TNF agents in treating these disorders, he and his associates analyzed data for 1,249 patients from the national Swiss IBD Cohort Study between 2006 and 2010. Patients were typically in their mid-30s and had lived with IBD for about 9 years, he said.
A total of 366 patients (29%) had at least one extraintestinal manifestation of IBD – most commonly peripheral arthritis (75%), followed by aphthous stomatitis (24%), and ankylosing spondylitis (22%). In all, 213 (58%) patients received at least one anti-TNF agent, and 40% received the prescription specifically for extraintestinal manifestations. Nearly two-thirds of the patients received infliximab, while 22% received adalimumab and 15% received certolizumab.
About 55% of patients improved on anti-TNF therapy over an average of 7 years of follow-up, Dr. Greuter and his associates reported. Among all three anti-TNF agents, response rates ranged from 100% for psoriasis, to 80% for erythema nodosum and stomatitis, to 73% for arthritis and uveitis, to 50% for pyoderma granulosum. Overall rates of improvement were slightly higher for infliximab than for the other two drugs, but “adalimumab and certolizumab were used mostly as a second or a third-line anti-TNF agent, and the response rate to a second or third-line treatment was lower than for the first one,” Dr. Greuter said. Some patients also received corticosteroids and immunomodulators, but excluding this subgroup had little effect on rates of response to anti-TNF therapy, he added.
Dr. Greuter also reported that 11 patients (about 5% of the cohort) developed 14 new extraintestinal manifestations after starting anti-TNF agents – usually peripheral arthritis, but also pyoderma granulosum, aphthous stomatitis, psoriasis, and uveitis. “We cannot say if this was primary, or a side effect of treatment,” he said. These disorders usually improved if patients stayed on their anti-TNF agent, he added.
About two-thirds of patients in the cohort were female, more than three-quarters had Crohn’s disease, 19% had ulcerative colitis, and 3% had indeterminate colitis, he noted.
A research grant from the Swiss National Science Foundation funded the study. Dr. Greuter had no disclosures.
SAN DIEGO – Tumor necrosis factor inhibitors improved the extraintestinal manifestations of inflammatory bowel disease (IBD) among more than half of affected patients, according to a national cohort study.
“The best response rates were for psoriasis, aphthous stomatitis, uveitis, and peripheral arthritis,” said Dr. Thomas Greuter of University Hospital in Zürich. Patients responded similarly whether they received oral infliximab or subcutaneous adalimumab or certolizumab, he noted.
IBD often is associated with debilitating disorders of the skin, joints, eyes, and hepatobiliary tract, but “due to the lack of randomized, controlled trials, the therapy of extraintestinal manifestations remains rather empirical,” Dr. Greuter said at the annual Digestive Disease Week.
To study the role of anti-TNF agents in treating these disorders, he and his associates analyzed data for 1,249 patients from the national Swiss IBD Cohort Study between 2006 and 2010. Patients were typically in their mid-30s and had lived with IBD for about 9 years, he said.
A total of 366 patients (29%) had at least one extraintestinal manifestation of IBD – most commonly peripheral arthritis (75%), followed by aphthous stomatitis (24%), and ankylosing spondylitis (22%). In all, 213 (58%) patients received at least one anti-TNF agent, and 40% received the prescription specifically for extraintestinal manifestations. Nearly two-thirds of the patients received infliximab, while 22% received adalimumab and 15% received certolizumab.
About 55% of patients improved on anti-TNF therapy over an average of 7 years of follow-up, Dr. Greuter and his associates reported. Among all three anti-TNF agents, response rates ranged from 100% for psoriasis, to 80% for erythema nodosum and stomatitis, to 73% for arthritis and uveitis, to 50% for pyoderma granulosum. Overall rates of improvement were slightly higher for infliximab than for the other two drugs, but “adalimumab and certolizumab were used mostly as a second or a third-line anti-TNF agent, and the response rate to a second or third-line treatment was lower than for the first one,” Dr. Greuter said. Some patients also received corticosteroids and immunomodulators, but excluding this subgroup had little effect on rates of response to anti-TNF therapy, he added.
Dr. Greuter also reported that 11 patients (about 5% of the cohort) developed 14 new extraintestinal manifestations after starting anti-TNF agents – usually peripheral arthritis, but also pyoderma granulosum, aphthous stomatitis, psoriasis, and uveitis. “We cannot say if this was primary, or a side effect of treatment,” he said. These disorders usually improved if patients stayed on their anti-TNF agent, he added.
About two-thirds of patients in the cohort were female, more than three-quarters had Crohn’s disease, 19% had ulcerative colitis, and 3% had indeterminate colitis, he noted.
A research grant from the Swiss National Science Foundation funded the study. Dr. Greuter had no disclosures.
AT DDW® 2016
Key clinical point: Tumor necrosis factor inhibitors improved the extraintestinal manifestations of inflammatory bowel disease among more than half of affected patients.
Major finding: About 55% of patients who received infliximab, adalimumab, or certolizumab had a clinical response.
Data source: A study of 1,249 patients with IBD from a national cohort.
Disclosures: A research grant from the Swiss National Science Foundation funded the study. Dr. Greuter had no disclosures.
TB still a risk in psoriasis patients taking TNF blockers
Despite following guidelines aimed at reducing the likelihood of tuberculosis infection in patients with psoriasis treated with tumor necrosis factor antagonists, a French study has identified a number of active TB cases in this patient group.
The nationwide retrospective study identified 12 cases of tuberculosis in individuals with psoriasis during treatment with a TNF-antagonist between 2006 and 2014 – 9 men and 3 women, whose mean age was 49 years – according to Dr. E. Guinard of the dermatology service, CHU Toulouse Larrey at Paul Sabatier University, Toulouse, France, and associates (J Eur Acad Dermatol Venereol. 2016 Jun 3. doi: 10.1111/jdv.13633).
The investigators identified the cases from a national pharmacosurveillance database and by contacting members of the psoriasis research group in France’s national dermatology society and the national college of French dermatology professors.
Before starting anti-TNF therapy, all 12 patients had been screened for latent TB based on French guidelines: three patients were tested with the tuberculosis skin test alone, six were tested with QuantiFERON-TB Gold test, and three patients had both the skin test and QuantiFERON-TB. Each had a pretreatment chest x-ray.
Based on this testing, three were diagnosed with latent TB and received chemoprophylaxis; the other nine patients tested negative.
Information on TB risk factors was not available for all 12 cases, but three people had had contact with someone with TB during treatment. Three of the individuals had traveled to or were born in areas where TB was endemic, and one patient had had a case of TB in the family in the year before starting anti-TNF therapy. Nine patients had a known history of BCG vaccination, the investigators said.
The time between starting anti-TNF therapy and the appearance of TB symptoms ranged from 2 to 176 weeks (mean, 23 weeks), and diagnosis was based on clinical signs – such as fever, night sweats, and cough – and on imaging and laboratory investigations. Most (10) of the cases were extrapulmonary presentations, and two were pleuropulmonary. When diagnosed with tuberculosis, seven of the patients were being treated with infliximab, four were being treated with adalimumab, and one was being treated with certolizumab. All stopped anti-TNF therapy once they were diagnosed with tuberculosis.
Dr. Guinard and associates noted that the low sensitivity of Mycobacterium tuberculosis diagnostic tests was “a striking feature” of these cases. Only one-third of the patients showed a positive direct Ziehl-Neelsen stain for acid-fast bacilli and half had a positive culture.
“According to this study and published experience, in a patient with suggestive symptoms, a negative Mycobacterium tuberculosis test should not delay the decision to initiate anti-tuberculosis treatment,” they wrote. “This is particularly important considering the high mortality of tuberculosis in patients treated with TNF antagonists,” which ranges from 6%-17%, they added.
Two of the twelve patients – aged 77 and 32 years – died of disseminated tuberculosis disease during treatment for TB.
“This is the largest study of tuberculosis in psoriasis patients treated with TNF antagonists,” according to the authors.
No conflicts of interest were declared, and the study had no funding source.
Despite following guidelines aimed at reducing the likelihood of tuberculosis infection in patients with psoriasis treated with tumor necrosis factor antagonists, a French study has identified a number of active TB cases in this patient group.
The nationwide retrospective study identified 12 cases of tuberculosis in individuals with psoriasis during treatment with a TNF-antagonist between 2006 and 2014 – 9 men and 3 women, whose mean age was 49 years – according to Dr. E. Guinard of the dermatology service, CHU Toulouse Larrey at Paul Sabatier University, Toulouse, France, and associates (J Eur Acad Dermatol Venereol. 2016 Jun 3. doi: 10.1111/jdv.13633).
The investigators identified the cases from a national pharmacosurveillance database and by contacting members of the psoriasis research group in France’s national dermatology society and the national college of French dermatology professors.
Before starting anti-TNF therapy, all 12 patients had been screened for latent TB based on French guidelines: three patients were tested with the tuberculosis skin test alone, six were tested with QuantiFERON-TB Gold test, and three patients had both the skin test and QuantiFERON-TB. Each had a pretreatment chest x-ray.
Based on this testing, three were diagnosed with latent TB and received chemoprophylaxis; the other nine patients tested negative.
Information on TB risk factors was not available for all 12 cases, but three people had had contact with someone with TB during treatment. Three of the individuals had traveled to or were born in areas where TB was endemic, and one patient had had a case of TB in the family in the year before starting anti-TNF therapy. Nine patients had a known history of BCG vaccination, the investigators said.
The time between starting anti-TNF therapy and the appearance of TB symptoms ranged from 2 to 176 weeks (mean, 23 weeks), and diagnosis was based on clinical signs – such as fever, night sweats, and cough – and on imaging and laboratory investigations. Most (10) of the cases were extrapulmonary presentations, and two were pleuropulmonary. When diagnosed with tuberculosis, seven of the patients were being treated with infliximab, four were being treated with adalimumab, and one was being treated with certolizumab. All stopped anti-TNF therapy once they were diagnosed with tuberculosis.
Dr. Guinard and associates noted that the low sensitivity of Mycobacterium tuberculosis diagnostic tests was “a striking feature” of these cases. Only one-third of the patients showed a positive direct Ziehl-Neelsen stain for acid-fast bacilli and half had a positive culture.
“According to this study and published experience, in a patient with suggestive symptoms, a negative Mycobacterium tuberculosis test should not delay the decision to initiate anti-tuberculosis treatment,” they wrote. “This is particularly important considering the high mortality of tuberculosis in patients treated with TNF antagonists,” which ranges from 6%-17%, they added.
Two of the twelve patients – aged 77 and 32 years – died of disseminated tuberculosis disease during treatment for TB.
“This is the largest study of tuberculosis in psoriasis patients treated with TNF antagonists,” according to the authors.
No conflicts of interest were declared, and the study had no funding source.
Despite following guidelines aimed at reducing the likelihood of tuberculosis infection in patients with psoriasis treated with tumor necrosis factor antagonists, a French study has identified a number of active TB cases in this patient group.
The nationwide retrospective study identified 12 cases of tuberculosis in individuals with psoriasis during treatment with a TNF-antagonist between 2006 and 2014 – 9 men and 3 women, whose mean age was 49 years – according to Dr. E. Guinard of the dermatology service, CHU Toulouse Larrey at Paul Sabatier University, Toulouse, France, and associates (J Eur Acad Dermatol Venereol. 2016 Jun 3. doi: 10.1111/jdv.13633).
The investigators identified the cases from a national pharmacosurveillance database and by contacting members of the psoriasis research group in France’s national dermatology society and the national college of French dermatology professors.
Before starting anti-TNF therapy, all 12 patients had been screened for latent TB based on French guidelines: three patients were tested with the tuberculosis skin test alone, six were tested with QuantiFERON-TB Gold test, and three patients had both the skin test and QuantiFERON-TB. Each had a pretreatment chest x-ray.
Based on this testing, three were diagnosed with latent TB and received chemoprophylaxis; the other nine patients tested negative.
Information on TB risk factors was not available for all 12 cases, but three people had had contact with someone with TB during treatment. Three of the individuals had traveled to or were born in areas where TB was endemic, and one patient had had a case of TB in the family in the year before starting anti-TNF therapy. Nine patients had a known history of BCG vaccination, the investigators said.
The time between starting anti-TNF therapy and the appearance of TB symptoms ranged from 2 to 176 weeks (mean, 23 weeks), and diagnosis was based on clinical signs – such as fever, night sweats, and cough – and on imaging and laboratory investigations. Most (10) of the cases were extrapulmonary presentations, and two were pleuropulmonary. When diagnosed with tuberculosis, seven of the patients were being treated with infliximab, four were being treated with adalimumab, and one was being treated with certolizumab. All stopped anti-TNF therapy once they were diagnosed with tuberculosis.
Dr. Guinard and associates noted that the low sensitivity of Mycobacterium tuberculosis diagnostic tests was “a striking feature” of these cases. Only one-third of the patients showed a positive direct Ziehl-Neelsen stain for acid-fast bacilli and half had a positive culture.
“According to this study and published experience, in a patient with suggestive symptoms, a negative Mycobacterium tuberculosis test should not delay the decision to initiate anti-tuberculosis treatment,” they wrote. “This is particularly important considering the high mortality of tuberculosis in patients treated with TNF antagonists,” which ranges from 6%-17%, they added.
Two of the twelve patients – aged 77 and 32 years – died of disseminated tuberculosis disease during treatment for TB.
“This is the largest study of tuberculosis in psoriasis patients treated with TNF antagonists,” according to the authors.
No conflicts of interest were declared, and the study had no funding source.
FROM THE JOURNAL OF THE EADV
Key clinical point: Individuals with psoriasis treated with tumor necrosis factor inhibitors may still present with tuberculosis, despite guidelines to reduce the likelihood of developing the disease.
Major finding: Twelve individuals with psoriasis were diagnosed with TB while on anti-TNF therapy, although TB prevention guidelines had been employed.
Data source: The cases were identified from a national pharmacosurveillance database in France and from members of the psoriasis research group in France’s national dermatology society and the national college of French dermatology professors.
Disclosures: No funding or conflicts of interest were declared.
TNF blocker safety may differ in RA and psoriasis patients
Rheumatoid arthritis patients on anti–tumor necrosis factor medications had approximately twice the rate of serious adverse events as did psoriasis patients on the same medications, based on data from a pair of prospective studies of about 4,000 adults.
The findings were published online in the British Journal of Dermatology.
“Current trends are to extrapolate the abundant literature existing on the safety of TNF antagonists in RA to define safety management for psoriasis,” wrote Dr. Ignacio García-Doval of the Fundación Academia Española de Dermatología y Venereología, Madrid, and colleagues. However, data on the similarity of risk associated with anti-TNF medications in RA and psoriasis are limited, the investigators said (BJD. 2016. doi: 10.1111/bjd.14776).
The researchers compared data from two similarly designed, overlapping prospective safety registry studies of anti-TNF medications in RA patients (the BIOBADASER study) and psoriasis patients (the BIOBADADERM study).
In the cohort of 3,171 RA patients, the researchers identified 1,248 serious or fatal adverse events during 16,230 person-years of follow-up; in the cohort of 946 psoriasis patients, they identified 124 serious or fatal adverse events during 2,760 person-years of follow-up. The resulting incidence rate ratio of psoriasis to RA was 0.6. The increased risk of serious adverse events for RA patients compared with psoriasis patients remained after the investigators controlled for confounding factors including age, sex, treatments, and comorbid conditions including hypertension, diabetes, hypercholesterolemia, and methotrexate therapy, for a hazard ratio of 0.54.
Moreover, the types of serious adverse events were different between the RA and psoriasis groups. Among those with RA, the rates of serious infections, cardiac disorders, respiratory disorders, and infusion reactions were significantly greater among those with RA, while psoriatic patients “had more skin and subcutaneous tissue disorders and hepatobiliary disorders,” the researchers noted.
By contrast, “rates of nonserious adverse events cannot be compared between the two cohorts,” because of differences in definitions, they pointed out. These differences resulted in a nonserious adverse event rate that was more than twice as high in the psoriasis group as in the RA group (582.2 events per 1,000 patient-years vs. 242.8 events per 1,000 patient-years).
Based on the findings, “published safety results of TNF-antagonists in RA cannot be fully extrapolated to psoriasis, as patients with RA have a higher risk and a different pattern of serious adverse events,” they concluded.
The BIOBADADERM project is promoted by the Fundación Academia Española de Dermatología y Venereología, which is supported by the Spanish Medicines and Health Products Agency and by multiple pharmaceutical companies. BIOBADASER received funding from Fundacion Española de Reumatología and the Spanish Medicines and Health Products Agency and grants from numerous pharmaceutical companies. Lead author Dr. Garcia-Doval disclosed travel grants for congresses from Merck/Schering-Plough Pharmaceuticals, Pfizer, and Janssen; the remaining two authors disclosed being a speaker and/or a consultant for several companies, including AbbVie.
Rheumatoid arthritis patients on anti–tumor necrosis factor medications had approximately twice the rate of serious adverse events as did psoriasis patients on the same medications, based on data from a pair of prospective studies of about 4,000 adults.
The findings were published online in the British Journal of Dermatology.
“Current trends are to extrapolate the abundant literature existing on the safety of TNF antagonists in RA to define safety management for psoriasis,” wrote Dr. Ignacio García-Doval of the Fundación Academia Española de Dermatología y Venereología, Madrid, and colleagues. However, data on the similarity of risk associated with anti-TNF medications in RA and psoriasis are limited, the investigators said (BJD. 2016. doi: 10.1111/bjd.14776).
The researchers compared data from two similarly designed, overlapping prospective safety registry studies of anti-TNF medications in RA patients (the BIOBADASER study) and psoriasis patients (the BIOBADADERM study).
In the cohort of 3,171 RA patients, the researchers identified 1,248 serious or fatal adverse events during 16,230 person-years of follow-up; in the cohort of 946 psoriasis patients, they identified 124 serious or fatal adverse events during 2,760 person-years of follow-up. The resulting incidence rate ratio of psoriasis to RA was 0.6. The increased risk of serious adverse events for RA patients compared with psoriasis patients remained after the investigators controlled for confounding factors including age, sex, treatments, and comorbid conditions including hypertension, diabetes, hypercholesterolemia, and methotrexate therapy, for a hazard ratio of 0.54.
Moreover, the types of serious adverse events were different between the RA and psoriasis groups. Among those with RA, the rates of serious infections, cardiac disorders, respiratory disorders, and infusion reactions were significantly greater among those with RA, while psoriatic patients “had more skin and subcutaneous tissue disorders and hepatobiliary disorders,” the researchers noted.
By contrast, “rates of nonserious adverse events cannot be compared between the two cohorts,” because of differences in definitions, they pointed out. These differences resulted in a nonserious adverse event rate that was more than twice as high in the psoriasis group as in the RA group (582.2 events per 1,000 patient-years vs. 242.8 events per 1,000 patient-years).
Based on the findings, “published safety results of TNF-antagonists in RA cannot be fully extrapolated to psoriasis, as patients with RA have a higher risk and a different pattern of serious adverse events,” they concluded.
The BIOBADADERM project is promoted by the Fundación Academia Española de Dermatología y Venereología, which is supported by the Spanish Medicines and Health Products Agency and by multiple pharmaceutical companies. BIOBADASER received funding from Fundacion Española de Reumatología and the Spanish Medicines and Health Products Agency and grants from numerous pharmaceutical companies. Lead author Dr. Garcia-Doval disclosed travel grants for congresses from Merck/Schering-Plough Pharmaceuticals, Pfizer, and Janssen; the remaining two authors disclosed being a speaker and/or a consultant for several companies, including AbbVie.
Rheumatoid arthritis patients on anti–tumor necrosis factor medications had approximately twice the rate of serious adverse events as did psoriasis patients on the same medications, based on data from a pair of prospective studies of about 4,000 adults.
The findings were published online in the British Journal of Dermatology.
“Current trends are to extrapolate the abundant literature existing on the safety of TNF antagonists in RA to define safety management for psoriasis,” wrote Dr. Ignacio García-Doval of the Fundación Academia Española de Dermatología y Venereología, Madrid, and colleagues. However, data on the similarity of risk associated with anti-TNF medications in RA and psoriasis are limited, the investigators said (BJD. 2016. doi: 10.1111/bjd.14776).
The researchers compared data from two similarly designed, overlapping prospective safety registry studies of anti-TNF medications in RA patients (the BIOBADASER study) and psoriasis patients (the BIOBADADERM study).
In the cohort of 3,171 RA patients, the researchers identified 1,248 serious or fatal adverse events during 16,230 person-years of follow-up; in the cohort of 946 psoriasis patients, they identified 124 serious or fatal adverse events during 2,760 person-years of follow-up. The resulting incidence rate ratio of psoriasis to RA was 0.6. The increased risk of serious adverse events for RA patients compared with psoriasis patients remained after the investigators controlled for confounding factors including age, sex, treatments, and comorbid conditions including hypertension, diabetes, hypercholesterolemia, and methotrexate therapy, for a hazard ratio of 0.54.
Moreover, the types of serious adverse events were different between the RA and psoriasis groups. Among those with RA, the rates of serious infections, cardiac disorders, respiratory disorders, and infusion reactions were significantly greater among those with RA, while psoriatic patients “had more skin and subcutaneous tissue disorders and hepatobiliary disorders,” the researchers noted.
By contrast, “rates of nonserious adverse events cannot be compared between the two cohorts,” because of differences in definitions, they pointed out. These differences resulted in a nonserious adverse event rate that was more than twice as high in the psoriasis group as in the RA group (582.2 events per 1,000 patient-years vs. 242.8 events per 1,000 patient-years).
Based on the findings, “published safety results of TNF-antagonists in RA cannot be fully extrapolated to psoriasis, as patients with RA have a higher risk and a different pattern of serious adverse events,” they concluded.
The BIOBADADERM project is promoted by the Fundación Academia Española de Dermatología y Venereología, which is supported by the Spanish Medicines and Health Products Agency and by multiple pharmaceutical companies. BIOBADASER received funding from Fundacion Española de Reumatología and the Spanish Medicines and Health Products Agency and grants from numerous pharmaceutical companies. Lead author Dr. Garcia-Doval disclosed travel grants for congresses from Merck/Schering-Plough Pharmaceuticals, Pfizer, and Janssen; the remaining two authors disclosed being a speaker and/or a consultant for several companies, including AbbVie.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Key clinical point: TNF-antagonists provoke different adverse events in patients with RA than in those with psoriasis, and safety data should be extrapolated with caution.
Major finding: The risk of serious adverse events associated with anti-TNF therapy was approximately twice as high in RA patients as in psoriasis patients (hazard ratio, 0.54).
Data source: A pair of prospective studies including 4,117 adults with RA or psoriasis who received anti-TNF agents.
Disclosures: The BIOBADADERM project is promoted by the Fundación Academia Española de Dermatología y Venereología, which is supported by the Spanish Medicines and Health Products Agency and by multiple pharmaceutical companies. BIOBADASER received funding from Fundacion Española de Reumatología and the Spanish Medicines and Health Products Agency and grants from numerous pharmaceutical companies. Lead author Dr. Garcia-Doval disclosed travel grants for congresses from Merck/Schering-Plough Pharmaceuticals, Pfizer, and Janssen; the remaining two authors disclosed being a speaker and/or a consultant for several companies, including AbbVie.
Full resolution of psoriasis in half of ixekizumab patients
More than half of the patients treated with the interleukin-17A antagonist, ixekizumab, achieved full resolution of skin plaques after a year of 2- to 4-week dosing, according to the latest analysis of data from the three UNCOVER trials of almost 4,000 patients with moderate to severe psoriasis.
The results of the three randomized, double-blind placebo-controlled pivotal trials were published online in the New England Journal of Medicine (doi: 10.1056/NEJMoa1512711).
Ixekizumab was approved in March for the treatment of adults with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy; it is marketed as Taltz by Eli Lilly. Some of the UNCOVER data have been previously reported. The UNCOVER studies were conducted at more than 100 sites in 21 countries.
Patients in the UNCOVER trials were randomized to 80 mg of ixekizumab every 2 weeks after a starting dose of 160 mg, 80 mg every 4 weeks after the same starting dose, or subcutaneous placebo injections, for 12 weeks.
In the UNCOVER-2 and UNCOVER-3 trials, additional cohorts were also randomized to twice-weekly etanercept (50 mg). All three trials also included a long-term extension period to 60 weeks, which was randomly assigned in UNCOVER-3 (where patients received 80 mg of ixekizumab every 4 weeks), or was implemented for all patients who responded to ixekizumab in the other two trials (where patients were randomized to 80 mg every 4 or 12 weeks, or placebo).
In the UNCOVER-1 trial in 1,296 patients, 89.1% of patients in the 2-week dosing group and 82.6% of the 4-week dosing group had achieved a 75% reduction in the Psoriasis Area and Severity Index score (PASI 75) by week 12, compared with 3.9% in the placebo group (P less than .0001 for all). Similarly, 35.3% of patients in the 2-week dosing group and 33.6% of those in the 4-week dosing group achieved a PASI score of 100 by week 12.
The high response rates seen during the induction period “were generally maintained during the long-term extension period in UNCOVER-3,” wrote Dr. Kenneth B. Gordon, professor of dermatology, Northwestern University, Chicago, and his coauthors. In the analysis of the long-term data from UNCOVER-3, the researchers reported that by week 60, 55% of patients receiving 80 mg of ixekizumab every 2 weeks during the induction period had achieved a PASI 100, and 52% of patients receiving ixekizumab every 4 weeks during the induction period had achieved a PASI 100.
Patients randomized to 80 mg of ixekizumab every 12 weeks after the first 12 weeks of treatment also showed significant long-term responses in the UNCOVER-1 and -2 trials. Nearly half (49.1%) of those who were initiated on 2-week dosing and 44.9% of those initiated on 4-week dosing achieved a PASI score of 75 on the 12-week dosing schedule, compared with 8%-9% of those randomized to placebo from week 12 through week 60.
The authors concluded that ixekizumab “provided high levels of clinical response at week 12 and through week 60,” adding, however, that “as with any treatment, the benefits need to be weighed against the adverse events, and the safety profile of longer-term treatment with ixekizumab should be examined.”
Although low serum IL-17 levels have previously been linked with cardiovascular disease, the study found no significant differences between the treatment and placebo groups in adverse cardiovascular events. Candidal infections were more common among those treated with ixekizumab, “a finding that is consistent with the role of IL-17A in the mucocutaneous defense against fungal infections,” they wrote.
There were 11 cases of inflammatory bowel disease reported among patients during treatment with ixekizumab and another three cases in patients receiving placebo during a randomized withdrawal period, who had received ixekizumab during the induction* period. These results “suggest that further evaluation is needed to understand the relationship between IL-17A inhibitors and inflammatory bowel disease,” the investigators said.
The authors of the study include Eli Lilly employees, several of whom also have stock options; the other authors declared a range of funding, advisory board positions, and speakers fees from pharmaceutical companies, including Eli Lilly. The UNCOVER studies were sponsored by Eli Lilly.
*A previous version of this article misstated the period during which three placebo patients received ixekizumab.
More than half of the patients treated with the interleukin-17A antagonist, ixekizumab, achieved full resolution of skin plaques after a year of 2- to 4-week dosing, according to the latest analysis of data from the three UNCOVER trials of almost 4,000 patients with moderate to severe psoriasis.
The results of the three randomized, double-blind placebo-controlled pivotal trials were published online in the New England Journal of Medicine (doi: 10.1056/NEJMoa1512711).
Ixekizumab was approved in March for the treatment of adults with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy; it is marketed as Taltz by Eli Lilly. Some of the UNCOVER data have been previously reported. The UNCOVER studies were conducted at more than 100 sites in 21 countries.
Patients in the UNCOVER trials were randomized to 80 mg of ixekizumab every 2 weeks after a starting dose of 160 mg, 80 mg every 4 weeks after the same starting dose, or subcutaneous placebo injections, for 12 weeks.
In the UNCOVER-2 and UNCOVER-3 trials, additional cohorts were also randomized to twice-weekly etanercept (50 mg). All three trials also included a long-term extension period to 60 weeks, which was randomly assigned in UNCOVER-3 (where patients received 80 mg of ixekizumab every 4 weeks), or was implemented for all patients who responded to ixekizumab in the other two trials (where patients were randomized to 80 mg every 4 or 12 weeks, or placebo).
In the UNCOVER-1 trial in 1,296 patients, 89.1% of patients in the 2-week dosing group and 82.6% of the 4-week dosing group had achieved a 75% reduction in the Psoriasis Area and Severity Index score (PASI 75) by week 12, compared with 3.9% in the placebo group (P less than .0001 for all). Similarly, 35.3% of patients in the 2-week dosing group and 33.6% of those in the 4-week dosing group achieved a PASI score of 100 by week 12.
The high response rates seen during the induction period “were generally maintained during the long-term extension period in UNCOVER-3,” wrote Dr. Kenneth B. Gordon, professor of dermatology, Northwestern University, Chicago, and his coauthors. In the analysis of the long-term data from UNCOVER-3, the researchers reported that by week 60, 55% of patients receiving 80 mg of ixekizumab every 2 weeks during the induction period had achieved a PASI 100, and 52% of patients receiving ixekizumab every 4 weeks during the induction period had achieved a PASI 100.
Patients randomized to 80 mg of ixekizumab every 12 weeks after the first 12 weeks of treatment also showed significant long-term responses in the UNCOVER-1 and -2 trials. Nearly half (49.1%) of those who were initiated on 2-week dosing and 44.9% of those initiated on 4-week dosing achieved a PASI score of 75 on the 12-week dosing schedule, compared with 8%-9% of those randomized to placebo from week 12 through week 60.
The authors concluded that ixekizumab “provided high levels of clinical response at week 12 and through week 60,” adding, however, that “as with any treatment, the benefits need to be weighed against the adverse events, and the safety profile of longer-term treatment with ixekizumab should be examined.”
Although low serum IL-17 levels have previously been linked with cardiovascular disease, the study found no significant differences between the treatment and placebo groups in adverse cardiovascular events. Candidal infections were more common among those treated with ixekizumab, “a finding that is consistent with the role of IL-17A in the mucocutaneous defense against fungal infections,” they wrote.
There were 11 cases of inflammatory bowel disease reported among patients during treatment with ixekizumab and another three cases in patients receiving placebo during a randomized withdrawal period, who had received ixekizumab during the induction* period. These results “suggest that further evaluation is needed to understand the relationship between IL-17A inhibitors and inflammatory bowel disease,” the investigators said.
The authors of the study include Eli Lilly employees, several of whom also have stock options; the other authors declared a range of funding, advisory board positions, and speakers fees from pharmaceutical companies, including Eli Lilly. The UNCOVER studies were sponsored by Eli Lilly.
*A previous version of this article misstated the period during which three placebo patients received ixekizumab.
More than half of the patients treated with the interleukin-17A antagonist, ixekizumab, achieved full resolution of skin plaques after a year of 2- to 4-week dosing, according to the latest analysis of data from the three UNCOVER trials of almost 4,000 patients with moderate to severe psoriasis.
The results of the three randomized, double-blind placebo-controlled pivotal trials were published online in the New England Journal of Medicine (doi: 10.1056/NEJMoa1512711).
Ixekizumab was approved in March for the treatment of adults with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy; it is marketed as Taltz by Eli Lilly. Some of the UNCOVER data have been previously reported. The UNCOVER studies were conducted at more than 100 sites in 21 countries.
Patients in the UNCOVER trials were randomized to 80 mg of ixekizumab every 2 weeks after a starting dose of 160 mg, 80 mg every 4 weeks after the same starting dose, or subcutaneous placebo injections, for 12 weeks.
In the UNCOVER-2 and UNCOVER-3 trials, additional cohorts were also randomized to twice-weekly etanercept (50 mg). All three trials also included a long-term extension period to 60 weeks, which was randomly assigned in UNCOVER-3 (where patients received 80 mg of ixekizumab every 4 weeks), or was implemented for all patients who responded to ixekizumab in the other two trials (where patients were randomized to 80 mg every 4 or 12 weeks, or placebo).
In the UNCOVER-1 trial in 1,296 patients, 89.1% of patients in the 2-week dosing group and 82.6% of the 4-week dosing group had achieved a 75% reduction in the Psoriasis Area and Severity Index score (PASI 75) by week 12, compared with 3.9% in the placebo group (P less than .0001 for all). Similarly, 35.3% of patients in the 2-week dosing group and 33.6% of those in the 4-week dosing group achieved a PASI score of 100 by week 12.
The high response rates seen during the induction period “were generally maintained during the long-term extension period in UNCOVER-3,” wrote Dr. Kenneth B. Gordon, professor of dermatology, Northwestern University, Chicago, and his coauthors. In the analysis of the long-term data from UNCOVER-3, the researchers reported that by week 60, 55% of patients receiving 80 mg of ixekizumab every 2 weeks during the induction period had achieved a PASI 100, and 52% of patients receiving ixekizumab every 4 weeks during the induction period had achieved a PASI 100.
Patients randomized to 80 mg of ixekizumab every 12 weeks after the first 12 weeks of treatment also showed significant long-term responses in the UNCOVER-1 and -2 trials. Nearly half (49.1%) of those who were initiated on 2-week dosing and 44.9% of those initiated on 4-week dosing achieved a PASI score of 75 on the 12-week dosing schedule, compared with 8%-9% of those randomized to placebo from week 12 through week 60.
The authors concluded that ixekizumab “provided high levels of clinical response at week 12 and through week 60,” adding, however, that “as with any treatment, the benefits need to be weighed against the adverse events, and the safety profile of longer-term treatment with ixekizumab should be examined.”
Although low serum IL-17 levels have previously been linked with cardiovascular disease, the study found no significant differences between the treatment and placebo groups in adverse cardiovascular events. Candidal infections were more common among those treated with ixekizumab, “a finding that is consistent with the role of IL-17A in the mucocutaneous defense against fungal infections,” they wrote.
There were 11 cases of inflammatory bowel disease reported among patients during treatment with ixekizumab and another three cases in patients receiving placebo during a randomized withdrawal period, who had received ixekizumab during the induction* period. These results “suggest that further evaluation is needed to understand the relationship between IL-17A inhibitors and inflammatory bowel disease,” the investigators said.
The authors of the study include Eli Lilly employees, several of whom also have stock options; the other authors declared a range of funding, advisory board positions, and speakers fees from pharmaceutical companies, including Eli Lilly. The UNCOVER studies were sponsored by Eli Lilly.
*A previous version of this article misstated the period during which three placebo patients received ixekizumab.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Half of patients treated with the IL-17A–neutralizing ixekizumab achieve full resolution of psoriasis plaques after 1 year.
Major finding: Among psoriasis patients treated with two weekly or four weekly doses of ixekizumab, 55% and 52%, respectively, achieve full resolution of plaques at 60 weeks.
Data source: Randomized, double-blind placebo-controlled trials (UNCOVER-1, -2, and -3) in 3,866 patients with psoriasis.
Disclosures: The studies were supported by Eli Lilly. The authors included employees of Eli Lilly, some of whom have stock options, while the other authors all declared a range of funding, advisory board positions, and speakers fees from pharmaceutical companies, including Eli Lilly.
Abdominal Aortic Aneurysm in Psoriasis Patients
In a study published online on April 14 in Arteriosclerosis, Thrombosis, and Vascular Biology, Khalid et al evaluated the risk for AAA in patients with psoriasis in a nationwide cohort study in Denmark. The study participants were Danish residents 18 years and older who were observed from January 1, 1997 until diagnosis of AAA; December 31, 2011; migration; or death. Incidence rates for AAA were calculated, and incidence rate ratios were adjusted for age, sex, comorbidity, medications, socioeconomic status, and smoking.
A total of 5,495,203 individuals were eligible for this study. Of them, Khalid et al identified 59,423 patients with mild psoriasis and 11,566 patients with severe psoriasis. The overall incidence rates of AAA were 3.72, 7.30, and 9.87 per 10,000 person-years for the reference population (23,696 cases), mild psoriasis (240 cases), and severe psoriasis (50 cases), respectively. The corresponding adjusted incidence rate ratios for AAA were increased in patients with psoriasis with incidence rate ratios of 1.20 (95% CI, 1.03-1.39) and 1.67 (95% CI, 1.21-2.32) for individuals with mild and severe disease, respectively.
Khalid et al concluded that psoriasis was associated with a disease severity–dependent increased risk for AAA; however, the mechanisms and consequences of this novel finding require further investigation.
What’s the issue?
Another example of an association of a comorbidity with psoriasis, this finding emphasizes the need for cardiovascular referral in psoriasis patients with risk factors such as hypertension and diabetes mellitus. How will these data influence your evaluation of psoriasis patients?
In a study published online on April 14 in Arteriosclerosis, Thrombosis, and Vascular Biology, Khalid et al evaluated the risk for AAA in patients with psoriasis in a nationwide cohort study in Denmark. The study participants were Danish residents 18 years and older who were observed from January 1, 1997 until diagnosis of AAA; December 31, 2011; migration; or death. Incidence rates for AAA were calculated, and incidence rate ratios were adjusted for age, sex, comorbidity, medications, socioeconomic status, and smoking.
A total of 5,495,203 individuals were eligible for this study. Of them, Khalid et al identified 59,423 patients with mild psoriasis and 11,566 patients with severe psoriasis. The overall incidence rates of AAA were 3.72, 7.30, and 9.87 per 10,000 person-years for the reference population (23,696 cases), mild psoriasis (240 cases), and severe psoriasis (50 cases), respectively. The corresponding adjusted incidence rate ratios for AAA were increased in patients with psoriasis with incidence rate ratios of 1.20 (95% CI, 1.03-1.39) and 1.67 (95% CI, 1.21-2.32) for individuals with mild and severe disease, respectively.
Khalid et al concluded that psoriasis was associated with a disease severity–dependent increased risk for AAA; however, the mechanisms and consequences of this novel finding require further investigation.
What’s the issue?
Another example of an association of a comorbidity with psoriasis, this finding emphasizes the need for cardiovascular referral in psoriasis patients with risk factors such as hypertension and diabetes mellitus. How will these data influence your evaluation of psoriasis patients?
In a study published online on April 14 in Arteriosclerosis, Thrombosis, and Vascular Biology, Khalid et al evaluated the risk for AAA in patients with psoriasis in a nationwide cohort study in Denmark. The study participants were Danish residents 18 years and older who were observed from January 1, 1997 until diagnosis of AAA; December 31, 2011; migration; or death. Incidence rates for AAA were calculated, and incidence rate ratios were adjusted for age, sex, comorbidity, medications, socioeconomic status, and smoking.
A total of 5,495,203 individuals were eligible for this study. Of them, Khalid et al identified 59,423 patients with mild psoriasis and 11,566 patients with severe psoriasis. The overall incidence rates of AAA were 3.72, 7.30, and 9.87 per 10,000 person-years for the reference population (23,696 cases), mild psoriasis (240 cases), and severe psoriasis (50 cases), respectively. The corresponding adjusted incidence rate ratios for AAA were increased in patients with psoriasis with incidence rate ratios of 1.20 (95% CI, 1.03-1.39) and 1.67 (95% CI, 1.21-2.32) for individuals with mild and severe disease, respectively.
Khalid et al concluded that psoriasis was associated with a disease severity–dependent increased risk for AAA; however, the mechanisms and consequences of this novel finding require further investigation.
What’s the issue?
Another example of an association of a comorbidity with psoriasis, this finding emphasizes the need for cardiovascular referral in psoriasis patients with risk factors such as hypertension and diabetes mellitus. How will these data influence your evaluation of psoriasis patients?
Serious infections are increasing among psoriasis inpatients
SCOTTSDALE, ARIZ. – From enterocolitis to MRSA, serious infections are on the rise among inpatients with psoriasis, and psoriasis is an independent risk factor for serious infections, according to findings from large retrospective studies from the United States and the United Kingdom.
Inpatients with psoriasis in the United States also were at greater risk of serious infections, compared with nonpsoriatic inpatients at every time point studied, and serious infections were associated with increased hospital costs, length of stay, and risk of mortality, reported Derek Hsu, a medical student at Northwestern University, Chicago, and his associates. “Research is needed to determine how to reduce the risk of serious infections in patients with psoriasis,” the investigators emphasized.
Psoriasis affects some 7 million adults in the United States. Biologics, which are transforming the treatment landscape for moderate-to-severe psoriasis, “should reduce inherent infectious risk by controlling the inflammatory process and reducing disease severity, [but] these effects may be immunosuppressing and increase the risk of infection in other ways,” according to Mr. Hsu and his associates. For their study, they analyzed data for 2002-2012 from the Nationwide Inpatient Sample, which covers 20% of hospitalizations in the United States. They extracted validated ICD-9 codes for psoriasis and serious infections, and calculated costs of care after adjusting for 2014 inflation, based on the United States Consumer Price Index.
Overall rates of serious infection and rates of pneumonia, MRSA, septicemia, diverticulitis, enterocolitis, encephalitis, and any viral or fungal infection rose significantly among inpatients with psoriasis between 2002 and 2012 (all P-values less than .05). Predictors of serious infections among inpatients with psoriasis included diabetes mellitus, obesity, and being of non-Caucasian race or ethnicity, female, older than 60 years, and on Medicare or Medicaid, the researchers reported at the annual meeting of the Society for Investigative Dermatology.
Furthermore, after controlling for age, sex, and race, psoriasis was a significant risk factor for many different types of serious infections. Among these were cellulitis, herpes simplex virus, infectious arthritis, osteomyelitis, meningitis, influenza, encephalitis, septicemia, enterocolitis, MRSA, methicillin-sensitive Staphylococcus aureus infections, and Clostridium difficile. Further, inpatients with psoriasis were more prone to urinary tract infection, peritonitis or intestinal abscess, appendicitis, tuberculosis, and viral and fungal infections (all P-values less than .05). The average cost of hospital stay for inpatients with psoriasis was more than $2,200 greater when they were diagnosed with one or more serious infections than otherwise, and their average length of hospital stay was 2 days longer.
The study in the United Kingdom included nearly 200,000 patients with psoriasis and almost 1 million patients without psoriasis from The Health Improvement Network electronic medical record database. Between 2002 and 2013, patients without psoriasis developed an estimated 78.5 serious infections per 100,000 person-years, compared with 88.9, 85.7, and 145.7 serious infections per 100,000 person-years, respectively, for all psoriasis patients, patients with mild disease, and patients with severe disease requiring systemic or phototherapy, said Dr. Junko Takeshita and her colleagues at the University of Pennsylvania in Philadelphia. After controlling for many potential demographic and clinical confounders, psoriasis increased the risk of serious infection by about 21% (hazard ratio, 1.21; 95% confidence interval, 1.18-1.23). Patients with severe psoriasis had a 63% greater risk of infection than patients without psoriasis, compared with an 18% increase for patients with mild psoriasis.
The findings show “serious infection, particularly respiratory and skin or soft tissue infections, to be an important and common cause of morbidity among patients with psoriasis, especially those with more severe disease,” Dr. Takeshita and her associates said. Notably, the link between psoriasis and risk of serious infection persisted after excluding patients on immunosuppressive therapies, suggesting “that the greater infection risk is at least partially attributable to more severe psoriasis, itself,” they added.
The analysis of Nationwide Inpatient Sample data was funded by the Agency for Healthcare Research and Quality and by the Dermatology Foundation. The analysis of Health Improvement Network data was funded by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, which is part of the National Institutes of Health, and by the Dermatology Foundation. None of the investigators reported conflicts of interest.
SCOTTSDALE, ARIZ. – From enterocolitis to MRSA, serious infections are on the rise among inpatients with psoriasis, and psoriasis is an independent risk factor for serious infections, according to findings from large retrospective studies from the United States and the United Kingdom.
Inpatients with psoriasis in the United States also were at greater risk of serious infections, compared with nonpsoriatic inpatients at every time point studied, and serious infections were associated with increased hospital costs, length of stay, and risk of mortality, reported Derek Hsu, a medical student at Northwestern University, Chicago, and his associates. “Research is needed to determine how to reduce the risk of serious infections in patients with psoriasis,” the investigators emphasized.
Psoriasis affects some 7 million adults in the United States. Biologics, which are transforming the treatment landscape for moderate-to-severe psoriasis, “should reduce inherent infectious risk by controlling the inflammatory process and reducing disease severity, [but] these effects may be immunosuppressing and increase the risk of infection in other ways,” according to Mr. Hsu and his associates. For their study, they analyzed data for 2002-2012 from the Nationwide Inpatient Sample, which covers 20% of hospitalizations in the United States. They extracted validated ICD-9 codes for psoriasis and serious infections, and calculated costs of care after adjusting for 2014 inflation, based on the United States Consumer Price Index.
Overall rates of serious infection and rates of pneumonia, MRSA, septicemia, diverticulitis, enterocolitis, encephalitis, and any viral or fungal infection rose significantly among inpatients with psoriasis between 2002 and 2012 (all P-values less than .05). Predictors of serious infections among inpatients with psoriasis included diabetes mellitus, obesity, and being of non-Caucasian race or ethnicity, female, older than 60 years, and on Medicare or Medicaid, the researchers reported at the annual meeting of the Society for Investigative Dermatology.
Furthermore, after controlling for age, sex, and race, psoriasis was a significant risk factor for many different types of serious infections. Among these were cellulitis, herpes simplex virus, infectious arthritis, osteomyelitis, meningitis, influenza, encephalitis, septicemia, enterocolitis, MRSA, methicillin-sensitive Staphylococcus aureus infections, and Clostridium difficile. Further, inpatients with psoriasis were more prone to urinary tract infection, peritonitis or intestinal abscess, appendicitis, tuberculosis, and viral and fungal infections (all P-values less than .05). The average cost of hospital stay for inpatients with psoriasis was more than $2,200 greater when they were diagnosed with one or more serious infections than otherwise, and their average length of hospital stay was 2 days longer.
The study in the United Kingdom included nearly 200,000 patients with psoriasis and almost 1 million patients without psoriasis from The Health Improvement Network electronic medical record database. Between 2002 and 2013, patients without psoriasis developed an estimated 78.5 serious infections per 100,000 person-years, compared with 88.9, 85.7, and 145.7 serious infections per 100,000 person-years, respectively, for all psoriasis patients, patients with mild disease, and patients with severe disease requiring systemic or phototherapy, said Dr. Junko Takeshita and her colleagues at the University of Pennsylvania in Philadelphia. After controlling for many potential demographic and clinical confounders, psoriasis increased the risk of serious infection by about 21% (hazard ratio, 1.21; 95% confidence interval, 1.18-1.23). Patients with severe psoriasis had a 63% greater risk of infection than patients without psoriasis, compared with an 18% increase for patients with mild psoriasis.
The findings show “serious infection, particularly respiratory and skin or soft tissue infections, to be an important and common cause of morbidity among patients with psoriasis, especially those with more severe disease,” Dr. Takeshita and her associates said. Notably, the link between psoriasis and risk of serious infection persisted after excluding patients on immunosuppressive therapies, suggesting “that the greater infection risk is at least partially attributable to more severe psoriasis, itself,” they added.
The analysis of Nationwide Inpatient Sample data was funded by the Agency for Healthcare Research and Quality and by the Dermatology Foundation. The analysis of Health Improvement Network data was funded by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, which is part of the National Institutes of Health, and by the Dermatology Foundation. None of the investigators reported conflicts of interest.
SCOTTSDALE, ARIZ. – From enterocolitis to MRSA, serious infections are on the rise among inpatients with psoriasis, and psoriasis is an independent risk factor for serious infections, according to findings from large retrospective studies from the United States and the United Kingdom.
Inpatients with psoriasis in the United States also were at greater risk of serious infections, compared with nonpsoriatic inpatients at every time point studied, and serious infections were associated with increased hospital costs, length of stay, and risk of mortality, reported Derek Hsu, a medical student at Northwestern University, Chicago, and his associates. “Research is needed to determine how to reduce the risk of serious infections in patients with psoriasis,” the investigators emphasized.
Psoriasis affects some 7 million adults in the United States. Biologics, which are transforming the treatment landscape for moderate-to-severe psoriasis, “should reduce inherent infectious risk by controlling the inflammatory process and reducing disease severity, [but] these effects may be immunosuppressing and increase the risk of infection in other ways,” according to Mr. Hsu and his associates. For their study, they analyzed data for 2002-2012 from the Nationwide Inpatient Sample, which covers 20% of hospitalizations in the United States. They extracted validated ICD-9 codes for psoriasis and serious infections, and calculated costs of care after adjusting for 2014 inflation, based on the United States Consumer Price Index.
Overall rates of serious infection and rates of pneumonia, MRSA, septicemia, diverticulitis, enterocolitis, encephalitis, and any viral or fungal infection rose significantly among inpatients with psoriasis between 2002 and 2012 (all P-values less than .05). Predictors of serious infections among inpatients with psoriasis included diabetes mellitus, obesity, and being of non-Caucasian race or ethnicity, female, older than 60 years, and on Medicare or Medicaid, the researchers reported at the annual meeting of the Society for Investigative Dermatology.
Furthermore, after controlling for age, sex, and race, psoriasis was a significant risk factor for many different types of serious infections. Among these were cellulitis, herpes simplex virus, infectious arthritis, osteomyelitis, meningitis, influenza, encephalitis, septicemia, enterocolitis, MRSA, methicillin-sensitive Staphylococcus aureus infections, and Clostridium difficile. Further, inpatients with psoriasis were more prone to urinary tract infection, peritonitis or intestinal abscess, appendicitis, tuberculosis, and viral and fungal infections (all P-values less than .05). The average cost of hospital stay for inpatients with psoriasis was more than $2,200 greater when they were diagnosed with one or more serious infections than otherwise, and their average length of hospital stay was 2 days longer.
The study in the United Kingdom included nearly 200,000 patients with psoriasis and almost 1 million patients without psoriasis from The Health Improvement Network electronic medical record database. Between 2002 and 2013, patients without psoriasis developed an estimated 78.5 serious infections per 100,000 person-years, compared with 88.9, 85.7, and 145.7 serious infections per 100,000 person-years, respectively, for all psoriasis patients, patients with mild disease, and patients with severe disease requiring systemic or phototherapy, said Dr. Junko Takeshita and her colleagues at the University of Pennsylvania in Philadelphia. After controlling for many potential demographic and clinical confounders, psoriasis increased the risk of serious infection by about 21% (hazard ratio, 1.21; 95% confidence interval, 1.18-1.23). Patients with severe psoriasis had a 63% greater risk of infection than patients without psoriasis, compared with an 18% increase for patients with mild psoriasis.
The findings show “serious infection, particularly respiratory and skin or soft tissue infections, to be an important and common cause of morbidity among patients with psoriasis, especially those with more severe disease,” Dr. Takeshita and her associates said. Notably, the link between psoriasis and risk of serious infection persisted after excluding patients on immunosuppressive therapies, suggesting “that the greater infection risk is at least partially attributable to more severe psoriasis, itself,” they added.
The analysis of Nationwide Inpatient Sample data was funded by the Agency for Healthcare Research and Quality and by the Dermatology Foundation. The analysis of Health Improvement Network data was funded by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, which is part of the National Institutes of Health, and by the Dermatology Foundation. None of the investigators reported conflicts of interest.
AT THE 2016 SID ANNUAL MEETING
Key clinical point: Psoriasis is an independent risk factor for serious infections, and serious infections are increasing among inpatients with psoriasis.
Major finding: Overall rates of serious infection and rates of pneumonia, MRSA, septicemia, diverticulitis, enterocolitis, encephalitis, and any viral or fungal infection rose significantly among inpatients in the United States with psoriasis between 2002 and 2012 (all P-values less than .05). In the United Kingdom during the same time period, patients with severe psoriasis had a 63% greater risk of serious infection than patients without psoriasis.
Data source: Analyses of data from the Nationwide Inpatient Sample for 2002 through 2012, and from The Health Improvement Network for 2003 through 2012.
Disclosures: The Nationwide Inpatient Sample analysis was funded by the Agency for Healthcare Research and Quality and the Dermatology Foundation. The analysis of The Health Improvement Network was funded by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, which is part of the National Institutes of Health, and by the Dermatology Foundation. None of the investigators reported conflicts of interest.
Make informed treatment decisions about biosimilars
In recent years federal laws have been enacted to help provide more treatment options and possibly lower costs to patients for therapeutic biological products. You have likely heard or read about “biosimilars” on the U.S. drug market. But are you ready to make informed treatment decisions and answer patients’ questions about these new drugs?
Biosimilars are not “generic biologics.” Although the shared goal of making biosimilars and generic drugs available is to provide more treatment options, there are important differences between the two approval pathways. For instance, unlike generic drugs, which are “bioequivalent” to their reference listed drug, biosimilar products can be either “biosimilar” to or “interchangeable” with their reference product. The differences may seem subtle based on the terminology, but the differences are important, and health care professionals who prescribe, dispense, or administer these products will need to understand them – as well as other aspects of these new therapies.
To help busy health care professionals better understand biosimilar and interchangeable products, the FDA has created a free 1.5-hour continuing education program titled, FDA Overview of Biosimilar Products. The course describes the important characteristics of biological products, particularly their complexity. For instance, a single molecule of a biological product, such as a monoclonal antibody, can easily be many hundreds of times larger and much more complex than that of a “small molecule” drug such as aspirin. Their increased complexity is one reason they are regulated differently than generics – and why, in the case of biosimilars, the law allows some differences from the reference product in clinically inactive components. The CE course discusses the “inherent variability” in the production of all biological products – both reference and biosimilar – and how the FDA accounts for these differences in assessing safety and efficacy of the products. The course also provides detailed definitions of important terms – such as “biosimilar” and “interchangeable” – and an overview of the standards the FDA has established for reviewing and approving biosimilar and interchangeable products. It also outlines the FDA review and approval standards for biosimilars and interchangeable products to help practitioners be assured that these products have been demonstrated to be safe and effective treatment options for their patients.
Some of our most important and expensive drugs are biological products used to treat patients who have serious medical conditions that are often life threatening and usually life altering. In April 2016, the Food and Drug Administration approved Inflectra (infliximab-dyyb) to help treat certain patients with certain gastrointestinal disorders, such as Crohn’s disease, and for other indications such as treatment of psoriasis and rheumatoid arthritis. The product is approved but not yet marketed in the United States. However, last year, Zarxio (filgrastim-sndz) became the first biosimilar actually available in the U.S. marketplace – approved to help boost white blood cell production for patients with severe neutropenia as well as for patients receiving various cancer therapies. These products are “biosimilar” to already-approved biological products called “reference products.” Inflectra is biosimilar to the reference product, Remicade (infliximab), and Zarxio is biosimilar to Neupogen (filgrastim).
Use of safe and effective biosimilar and interchangeable biological products can potentially make treatment more affordable for patients in need and improve public health. As patients begin to ask about the use of biosimilar and interchangeable products, the FDA hopes this course will help prepare health care practitioners to make informed decisions on behalf of their patients.
Dr. Christl is Associate Director for Therapeutic Biologics at the Office of New Drugs, Center for Drug Evaluation and Research, FDA.
The Food and Drug Administration’s recent approval of Inflectra (infliximab-dyyb) signals the advent of new therapeutic options in the treatment of patients with digestive diseases. However, there is still much to learn about biosimilars, their safety, and their efficacy. In the absence of sufficient clinical data, the American Gastroenterological Association has deferred taking a position on biosimilars. However, we recognize a critical need to educate gastroenterologists and their patients on this issue based on the information we have to date.

|
Dr. Gary R. Lichtenstein |
In April 2016, the AGA Biosimilars Advisory Panel was created to determine key knowledge gaps regarding biosimilars, anticipate emerging issues around which to prepare AGA members, and recommend educational activities that address these gaps and issues. Upon review and approval by the AGA Institute Governing Board, the panel’s recommendations will be implemented by the AGA Center for Diagnostics and Therapeutics (CDT) in conjunction with other AGA committees. I am honored to chair this panel and look forward to working with my esteemed colleagues, all of whom are thought leaders in the field:
• Jean-Frederic Colombel, M.D., Mount Sinai Hospital
• Stephen B. Hanauer, M.D., AGAF, Northwestern University
• Sunanda V. Kane, M.D., MSPH, AGAF, Mayo Clinic
• Garrett Lawlor, M.D., Columbia University Medical Center
• James D. Lewis, M.D., MSCE, AGAF, University of Pennsylvania
• Loren A. Laine, M.D., AGAF, Yale University (CDT scientific advisory board liaison)
We hope to develop our initial recommendations in the next few months, recognizing that the field will continue to evolve quickly. AGA will also continue to work with the FDA and industry to ensure that the concerns of gastroenterologists are appropriately communicated and ensure that all of us are working together to improve the lives of patients with digestive diseases.
Dr. Gary R. Lichtenstein, AGAF, is chair, AGA Biosimilars Advisory Panel, and professor of medicine, Hospital of the University of Pennsylvania, Philadelphia.
The Food and Drug Administration’s recent approval of Inflectra (infliximab-dyyb) signals the advent of new therapeutic options in the treatment of patients with digestive diseases. However, there is still much to learn about biosimilars, their safety, and their efficacy. In the absence of sufficient clinical data, the American Gastroenterological Association has deferred taking a position on biosimilars. However, we recognize a critical need to educate gastroenterologists and their patients on this issue based on the information we have to date.

|
Dr. Gary R. Lichtenstein |
In April 2016, the AGA Biosimilars Advisory Panel was created to determine key knowledge gaps regarding biosimilars, anticipate emerging issues around which to prepare AGA members, and recommend educational activities that address these gaps and issues. Upon review and approval by the AGA Institute Governing Board, the panel’s recommendations will be implemented by the AGA Center for Diagnostics and Therapeutics (CDT) in conjunction with other AGA committees. I am honored to chair this panel and look forward to working with my esteemed colleagues, all of whom are thought leaders in the field:
• Jean-Frederic Colombel, M.D., Mount Sinai Hospital
• Stephen B. Hanauer, M.D., AGAF, Northwestern University
• Sunanda V. Kane, M.D., MSPH, AGAF, Mayo Clinic
• Garrett Lawlor, M.D., Columbia University Medical Center
• James D. Lewis, M.D., MSCE, AGAF, University of Pennsylvania
• Loren A. Laine, M.D., AGAF, Yale University (CDT scientific advisory board liaison)
We hope to develop our initial recommendations in the next few months, recognizing that the field will continue to evolve quickly. AGA will also continue to work with the FDA and industry to ensure that the concerns of gastroenterologists are appropriately communicated and ensure that all of us are working together to improve the lives of patients with digestive diseases.
Dr. Gary R. Lichtenstein, AGAF, is chair, AGA Biosimilars Advisory Panel, and professor of medicine, Hospital of the University of Pennsylvania, Philadelphia.
The Food and Drug Administration’s recent approval of Inflectra (infliximab-dyyb) signals the advent of new therapeutic options in the treatment of patients with digestive diseases. However, there is still much to learn about biosimilars, their safety, and their efficacy. In the absence of sufficient clinical data, the American Gastroenterological Association has deferred taking a position on biosimilars. However, we recognize a critical need to educate gastroenterologists and their patients on this issue based on the information we have to date.

|
Dr. Gary R. Lichtenstein |
In April 2016, the AGA Biosimilars Advisory Panel was created to determine key knowledge gaps regarding biosimilars, anticipate emerging issues around which to prepare AGA members, and recommend educational activities that address these gaps and issues. Upon review and approval by the AGA Institute Governing Board, the panel’s recommendations will be implemented by the AGA Center for Diagnostics and Therapeutics (CDT) in conjunction with other AGA committees. I am honored to chair this panel and look forward to working with my esteemed colleagues, all of whom are thought leaders in the field:
• Jean-Frederic Colombel, M.D., Mount Sinai Hospital
• Stephen B. Hanauer, M.D., AGAF, Northwestern University
• Sunanda V. Kane, M.D., MSPH, AGAF, Mayo Clinic
• Garrett Lawlor, M.D., Columbia University Medical Center
• James D. Lewis, M.D., MSCE, AGAF, University of Pennsylvania
• Loren A. Laine, M.D., AGAF, Yale University (CDT scientific advisory board liaison)
We hope to develop our initial recommendations in the next few months, recognizing that the field will continue to evolve quickly. AGA will also continue to work with the FDA and industry to ensure that the concerns of gastroenterologists are appropriately communicated and ensure that all of us are working together to improve the lives of patients with digestive diseases.
Dr. Gary R. Lichtenstein, AGAF, is chair, AGA Biosimilars Advisory Panel, and professor of medicine, Hospital of the University of Pennsylvania, Philadelphia.
In recent years federal laws have been enacted to help provide more treatment options and possibly lower costs to patients for therapeutic biological products. You have likely heard or read about “biosimilars” on the U.S. drug market. But are you ready to make informed treatment decisions and answer patients’ questions about these new drugs?
Biosimilars are not “generic biologics.” Although the shared goal of making biosimilars and generic drugs available is to provide more treatment options, there are important differences between the two approval pathways. For instance, unlike generic drugs, which are “bioequivalent” to their reference listed drug, biosimilar products can be either “biosimilar” to or “interchangeable” with their reference product. The differences may seem subtle based on the terminology, but the differences are important, and health care professionals who prescribe, dispense, or administer these products will need to understand them – as well as other aspects of these new therapies.
To help busy health care professionals better understand biosimilar and interchangeable products, the FDA has created a free 1.5-hour continuing education program titled, FDA Overview of Biosimilar Products. The course describes the important characteristics of biological products, particularly their complexity. For instance, a single molecule of a biological product, such as a monoclonal antibody, can easily be many hundreds of times larger and much more complex than that of a “small molecule” drug such as aspirin. Their increased complexity is one reason they are regulated differently than generics – and why, in the case of biosimilars, the law allows some differences from the reference product in clinically inactive components. The CE course discusses the “inherent variability” in the production of all biological products – both reference and biosimilar – and how the FDA accounts for these differences in assessing safety and efficacy of the products. The course also provides detailed definitions of important terms – such as “biosimilar” and “interchangeable” – and an overview of the standards the FDA has established for reviewing and approving biosimilar and interchangeable products. It also outlines the FDA review and approval standards for biosimilars and interchangeable products to help practitioners be assured that these products have been demonstrated to be safe and effective treatment options for their patients.
Some of our most important and expensive drugs are biological products used to treat patients who have serious medical conditions that are often life threatening and usually life altering. In April 2016, the Food and Drug Administration approved Inflectra (infliximab-dyyb) to help treat certain patients with certain gastrointestinal disorders, such as Crohn’s disease, and for other indications such as treatment of psoriasis and rheumatoid arthritis. The product is approved but not yet marketed in the United States. However, last year, Zarxio (filgrastim-sndz) became the first biosimilar actually available in the U.S. marketplace – approved to help boost white blood cell production for patients with severe neutropenia as well as for patients receiving various cancer therapies. These products are “biosimilar” to already-approved biological products called “reference products.” Inflectra is biosimilar to the reference product, Remicade (infliximab), and Zarxio is biosimilar to Neupogen (filgrastim).
Use of safe and effective biosimilar and interchangeable biological products can potentially make treatment more affordable for patients in need and improve public health. As patients begin to ask about the use of biosimilar and interchangeable products, the FDA hopes this course will help prepare health care practitioners to make informed decisions on behalf of their patients.
Dr. Christl is Associate Director for Therapeutic Biologics at the Office of New Drugs, Center for Drug Evaluation and Research, FDA.
In recent years federal laws have been enacted to help provide more treatment options and possibly lower costs to patients for therapeutic biological products. You have likely heard or read about “biosimilars” on the U.S. drug market. But are you ready to make informed treatment decisions and answer patients’ questions about these new drugs?
Biosimilars are not “generic biologics.” Although the shared goal of making biosimilars and generic drugs available is to provide more treatment options, there are important differences between the two approval pathways. For instance, unlike generic drugs, which are “bioequivalent” to their reference listed drug, biosimilar products can be either “biosimilar” to or “interchangeable” with their reference product. The differences may seem subtle based on the terminology, but the differences are important, and health care professionals who prescribe, dispense, or administer these products will need to understand them – as well as other aspects of these new therapies.
To help busy health care professionals better understand biosimilar and interchangeable products, the FDA has created a free 1.5-hour continuing education program titled, FDA Overview of Biosimilar Products. The course describes the important characteristics of biological products, particularly their complexity. For instance, a single molecule of a biological product, such as a monoclonal antibody, can easily be many hundreds of times larger and much more complex than that of a “small molecule” drug such as aspirin. Their increased complexity is one reason they are regulated differently than generics – and why, in the case of biosimilars, the law allows some differences from the reference product in clinically inactive components. The CE course discusses the “inherent variability” in the production of all biological products – both reference and biosimilar – and how the FDA accounts for these differences in assessing safety and efficacy of the products. The course also provides detailed definitions of important terms – such as “biosimilar” and “interchangeable” – and an overview of the standards the FDA has established for reviewing and approving biosimilar and interchangeable products. It also outlines the FDA review and approval standards for biosimilars and interchangeable products to help practitioners be assured that these products have been demonstrated to be safe and effective treatment options for their patients.
Some of our most important and expensive drugs are biological products used to treat patients who have serious medical conditions that are often life threatening and usually life altering. In April 2016, the Food and Drug Administration approved Inflectra (infliximab-dyyb) to help treat certain patients with certain gastrointestinal disorders, such as Crohn’s disease, and for other indications such as treatment of psoriasis and rheumatoid arthritis. The product is approved but not yet marketed in the United States. However, last year, Zarxio (filgrastim-sndz) became the first biosimilar actually available in the U.S. marketplace – approved to help boost white blood cell production for patients with severe neutropenia as well as for patients receiving various cancer therapies. These products are “biosimilar” to already-approved biological products called “reference products.” Inflectra is biosimilar to the reference product, Remicade (infliximab), and Zarxio is biosimilar to Neupogen (filgrastim).
Use of safe and effective biosimilar and interchangeable biological products can potentially make treatment more affordable for patients in need and improve public health. As patients begin to ask about the use of biosimilar and interchangeable products, the FDA hopes this course will help prepare health care practitioners to make informed decisions on behalf of their patients.
Dr. Christl is Associate Director for Therapeutic Biologics at the Office of New Drugs, Center for Drug Evaluation and Research, FDA.
What Patients Need From Their Psoriasis Therapy
Psoriasis patients consider reduced scaling/flaking (64%), reduced itching (62%), and reduced redness and inflammation (52%) to be the top 3 most meaningful benefits when considering a new treatment, according to a patient poll during the US Food and Drug Administration (FDA) public meeting on patient-focused drug development for psoriasis. When asked to choose 1 benefit, 22% of patients indicated reduced scaling/flaking and reduced itching would be the most meaningful, with 18% of patients choosing reduction in the number of plaques, 16% reduced burning/stinging, and 11% reduced redness and inflammation.
The FDA held the meeting on March 17. According to Theresa Mullin, PhD, Director of the Office of Strategic Programs for the Center for Drug Evaluation and Research of the FDA, “Patient perspective helps inform [the FDA’s] understanding of the context for the assessment of benefit-risk and decision making for new drugs.” In addition to patient testimonials, the FDA posed a number of polls to learn about patient preferences. Their responses offer insight for dermatologists on the symptoms that are most bothersome to patients and the results patients want to see with treatment.
Of all the nonjoint symptoms of psoriasis, patients indicated that flaking/scaling (67%), itching (67%), dry cracked skin that may bleed (39%), pain or soreness (37%), and burning/stinging (25%) were the top 5 symptoms having the most significant impact on their daily life. The top 5 most bothersome impacts of psoriasis symptoms included emotional impacts (eg, self-esteem)(59%), limitations on activities (eg, work, school, sports, hobbies)(42%), stigma or embarrassment (41%), ability to fall asleep or stay asleep (39%), and impact on sexual intimacy (34%).
Of the drug therapies or medical devices used by patients to treat psoriasis, an overwhelming 96% of patients reported use of topical treatments, followed by oral or injected medications (76%), phototherapy (64%), and oral prescription medications (62%). Aside from drug therapies, patients indicated that they are managing their symptoms with over-the-counter products (eg, coal tar, salicylic acid, Epsom salt)(35%), complementary or alternative therapies (27%), other therapies not mentioned (18%), diet modifications (10%), no therapy (8%), or dietary and herbal supplements (2%).
The top ranking factors affecting the patients’ decisions about using treatments to help reduce or control the spread of psoriasis included whether the drug showed effectiveness for the specific benefit that is most meaningful for you (64%), access to treatment (eg, insurance coverage)(44%), and the possibility of rare but serious side effects (eg, blood disorders, certain cancers)(31%). The lowest ranking factor was how the medication is administered (4%).
In March 2015, Jeffrey M. Weinberg, MD, explored the topic of treatment refusal in his editorial, “First Refusal.” He reviewed the results from a study that sought to investigate refusal of topical treatments by patients living with psoriasis in France as well as the factors that influence such refusal (J Dermatolog Treat. 2015;2:1-5). “The findings of this study indicate possible strategies to reduce patient refusal,” Dr. Weinberg wrote. “For example, enhanced education about the therapeutic options for psoriasis and their benefits could counter negative perceptions about these therapies. It also appears that increased focus on the physician-patient relationship may have a positive impact in this area.” Focusing on the treatment of the patient’s most bothersome symptoms with therapies that will target these symptoms may help to strengthen the patient-physician relationship and ensure patient compliance to therapy.
Psoriasis patients consider reduced scaling/flaking (64%), reduced itching (62%), and reduced redness and inflammation (52%) to be the top 3 most meaningful benefits when considering a new treatment, according to a patient poll during the US Food and Drug Administration (FDA) public meeting on patient-focused drug development for psoriasis. When asked to choose 1 benefit, 22% of patients indicated reduced scaling/flaking and reduced itching would be the most meaningful, with 18% of patients choosing reduction in the number of plaques, 16% reduced burning/stinging, and 11% reduced redness and inflammation.
The FDA held the meeting on March 17. According to Theresa Mullin, PhD, Director of the Office of Strategic Programs for the Center for Drug Evaluation and Research of the FDA, “Patient perspective helps inform [the FDA’s] understanding of the context for the assessment of benefit-risk and decision making for new drugs.” In addition to patient testimonials, the FDA posed a number of polls to learn about patient preferences. Their responses offer insight for dermatologists on the symptoms that are most bothersome to patients and the results patients want to see with treatment.
Of all the nonjoint symptoms of psoriasis, patients indicated that flaking/scaling (67%), itching (67%), dry cracked skin that may bleed (39%), pain or soreness (37%), and burning/stinging (25%) were the top 5 symptoms having the most significant impact on their daily life. The top 5 most bothersome impacts of psoriasis symptoms included emotional impacts (eg, self-esteem)(59%), limitations on activities (eg, work, school, sports, hobbies)(42%), stigma or embarrassment (41%), ability to fall asleep or stay asleep (39%), and impact on sexual intimacy (34%).
Of the drug therapies or medical devices used by patients to treat psoriasis, an overwhelming 96% of patients reported use of topical treatments, followed by oral or injected medications (76%), phototherapy (64%), and oral prescription medications (62%). Aside from drug therapies, patients indicated that they are managing their symptoms with over-the-counter products (eg, coal tar, salicylic acid, Epsom salt)(35%), complementary or alternative therapies (27%), other therapies not mentioned (18%), diet modifications (10%), no therapy (8%), or dietary and herbal supplements (2%).
The top ranking factors affecting the patients’ decisions about using treatments to help reduce or control the spread of psoriasis included whether the drug showed effectiveness for the specific benefit that is most meaningful for you (64%), access to treatment (eg, insurance coverage)(44%), and the possibility of rare but serious side effects (eg, blood disorders, certain cancers)(31%). The lowest ranking factor was how the medication is administered (4%).
In March 2015, Jeffrey M. Weinberg, MD, explored the topic of treatment refusal in his editorial, “First Refusal.” He reviewed the results from a study that sought to investigate refusal of topical treatments by patients living with psoriasis in France as well as the factors that influence such refusal (J Dermatolog Treat. 2015;2:1-5). “The findings of this study indicate possible strategies to reduce patient refusal,” Dr. Weinberg wrote. “For example, enhanced education about the therapeutic options for psoriasis and their benefits could counter negative perceptions about these therapies. It also appears that increased focus on the physician-patient relationship may have a positive impact in this area.” Focusing on the treatment of the patient’s most bothersome symptoms with therapies that will target these symptoms may help to strengthen the patient-physician relationship and ensure patient compliance to therapy.
Psoriasis patients consider reduced scaling/flaking (64%), reduced itching (62%), and reduced redness and inflammation (52%) to be the top 3 most meaningful benefits when considering a new treatment, according to a patient poll during the US Food and Drug Administration (FDA) public meeting on patient-focused drug development for psoriasis. When asked to choose 1 benefit, 22% of patients indicated reduced scaling/flaking and reduced itching would be the most meaningful, with 18% of patients choosing reduction in the number of plaques, 16% reduced burning/stinging, and 11% reduced redness and inflammation.
The FDA held the meeting on March 17. According to Theresa Mullin, PhD, Director of the Office of Strategic Programs for the Center for Drug Evaluation and Research of the FDA, “Patient perspective helps inform [the FDA’s] understanding of the context for the assessment of benefit-risk and decision making for new drugs.” In addition to patient testimonials, the FDA posed a number of polls to learn about patient preferences. Their responses offer insight for dermatologists on the symptoms that are most bothersome to patients and the results patients want to see with treatment.
Of all the nonjoint symptoms of psoriasis, patients indicated that flaking/scaling (67%), itching (67%), dry cracked skin that may bleed (39%), pain or soreness (37%), and burning/stinging (25%) were the top 5 symptoms having the most significant impact on their daily life. The top 5 most bothersome impacts of psoriasis symptoms included emotional impacts (eg, self-esteem)(59%), limitations on activities (eg, work, school, sports, hobbies)(42%), stigma or embarrassment (41%), ability to fall asleep or stay asleep (39%), and impact on sexual intimacy (34%).
Of the drug therapies or medical devices used by patients to treat psoriasis, an overwhelming 96% of patients reported use of topical treatments, followed by oral or injected medications (76%), phototherapy (64%), and oral prescription medications (62%). Aside from drug therapies, patients indicated that they are managing their symptoms with over-the-counter products (eg, coal tar, salicylic acid, Epsom salt)(35%), complementary or alternative therapies (27%), other therapies not mentioned (18%), diet modifications (10%), no therapy (8%), or dietary and herbal supplements (2%).
The top ranking factors affecting the patients’ decisions about using treatments to help reduce or control the spread of psoriasis included whether the drug showed effectiveness for the specific benefit that is most meaningful for you (64%), access to treatment (eg, insurance coverage)(44%), and the possibility of rare but serious side effects (eg, blood disorders, certain cancers)(31%). The lowest ranking factor was how the medication is administered (4%).
In March 2015, Jeffrey M. Weinberg, MD, explored the topic of treatment refusal in his editorial, “First Refusal.” He reviewed the results from a study that sought to investigate refusal of topical treatments by patients living with psoriasis in France as well as the factors that influence such refusal (J Dermatolog Treat. 2015;2:1-5). “The findings of this study indicate possible strategies to reduce patient refusal,” Dr. Weinberg wrote. “For example, enhanced education about the therapeutic options for psoriasis and their benefits could counter negative perceptions about these therapies. It also appears that increased focus on the physician-patient relationship may have a positive impact in this area.” Focusing on the treatment of the patient’s most bothersome symptoms with therapies that will target these symptoms may help to strengthen the patient-physician relationship and ensure patient compliance to therapy.