User login
Algorithm proposes approach for managing TNF inhibitor–induced psoriasis
Patients with tumor necrosis factor inhibitor–induced psoriasis could potentially be switched to a different drug class if they have moderate to severe skin eruption or mild skin eruption with an uncontrolled underlying disease such as inflammatory bowel disease, psoriasis, psoriatic arthritis, or rheumatoid arthritis, according to a new treatment algorithm proposed by researchers from Brigham and Women’s Hospital and Harvard Medical School in Boston.
The researchers outlined the prevalence of tumor necrosis factor–alpha inhibitor (TNFi)-induced psoriasis in a literature review of inflammatory bowel disease (IBD), psoriasis, psoriatic arthritis (PsA), and rheumatoid arthritis (RA) and identified an estimated rate of between 2.3% and 5% in patients with RA and between 1.6% and 2.7% in patients with IBD. Although there have been reports of TNFi-induced psoriasis in patients with psoriasis and PsA, the prevalence is unclear, they wrote in the Journal of Psoriasis and Psoriatic Arthritis.
The authors then created an algorithm to manage and treat TNFi-induced psoriasiform skin eruptions with decisions to continue therapy and “treat through” symptoms, switch to a different anti-TNF therapy, or switch to a different drug class based on severity of symptoms, whether the underlying disease is well controlled, and how patients with those underlying diseases have fared with those specific therapies or agents.
“We’ve shifted gears over the past decade, and we’ve gone from having very few agents and trying to keep patients desperately on one or two agents because we didn’t want to have to give up on them for their other comorbid disease, whether it was Crohn’s, colitis, RA, or whatever it may be,” senior author Joseph Merola, MD, director of the Center for Skin and Related Musculoskeletal Diseases at Brigham and Women’s Hospital, Boston, said in an interview. “We’re now in an area where we can have an algorithm like this, and we have so many more mechanistic options to move to.”
Dr. Merola, who is board certified in dermatology and rheumatology, said the algorithm is meant to “open a dialogue” with other specialists in different areas and raise awareness of treatments in related but separate fields. For diseases not often seen by more than one specialty, with the exception of psoriasis and PsA, he said that “the idea is to start a dialogue and increase communication between specialists.”
Dr. Merola noted that while the algorithm in many respects is meant to guide a physician in a specialty in appropriate medication decisions, at the same time he hopes that “it opens a dialogue and communication with the other specialty who tends to oversee this particular disease state or class of medicine to really work together to try to find the right drug for the right person.”
For patients with a mild skin eruption and a controlled underlying disease, the algorithm recommends a “treat through” approach by continuing anti-TNF therapy and treating psoriasis symptoms with topical steroids, ultraviolet therapy, methotrexate, cyclosporine, or acitretin, and to consider dapsone in cases of pustular psoriasis. However, the researchers noted that “treat through” studies have reported complete symptom resolution in 26%-41% of patients.
For patients with recalcitrant or worsening TNFi-induced psoriasis or patients with mild skin eruptions with an uncontrolled underlying disease, the researchers proposed considering switching to a different anti-TNF therapy, although studies have shown complete resolution of symptoms in only 5%-37% of patients.
If patients worsen from there, or if they have moderate to-severe skin eruption with uncontrolled underlying disease, they could be considered for switching to a different drug class and treated based on their underlying disease, along with treatment for psoriasis symptoms. This approach has been shown to completely resolve lesions in up to 64% of cases, they said. IBD patients could benefit from ustekinumab, vedolizumab, 6-mercaptopurine, or azathioprine as an alternative to anti-TNF therapy. Those patients with psoriasis should be considered for guselkumab, while ustekinumab, ixekizumab, secukinumab, and apremilast are effective treatments for patients with psoriasis and PsA. Patients with RA could receive treatment with tocilizumab, rituximab, abatacept, and tofacitinib, the authors wrote.
Dr. Merola reported serving as a consultant and/or investigator for Merck Research Laboratories, AbbVie, Dermavant, Eli Lilly, Novartis, Janssen, UCB, Samumed, Celgene, Sanofi Regeneron, GlaxoSmithKline, Almirall, Sun Pharma, Biogen, Pfizer, Incyte, Aclaris, and Leo Pharma.
SOURCE: Li SJ et al. J Psoriasis Psoriatic Arthritis. 2018 Nov 21. doi: 10.1177/2475530318810851.
Patients with tumor necrosis factor inhibitor–induced psoriasis could potentially be switched to a different drug class if they have moderate to severe skin eruption or mild skin eruption with an uncontrolled underlying disease such as inflammatory bowel disease, psoriasis, psoriatic arthritis, or rheumatoid arthritis, according to a new treatment algorithm proposed by researchers from Brigham and Women’s Hospital and Harvard Medical School in Boston.
The researchers outlined the prevalence of tumor necrosis factor–alpha inhibitor (TNFi)-induced psoriasis in a literature review of inflammatory bowel disease (IBD), psoriasis, psoriatic arthritis (PsA), and rheumatoid arthritis (RA) and identified an estimated rate of between 2.3% and 5% in patients with RA and between 1.6% and 2.7% in patients with IBD. Although there have been reports of TNFi-induced psoriasis in patients with psoriasis and PsA, the prevalence is unclear, they wrote in the Journal of Psoriasis and Psoriatic Arthritis.
The authors then created an algorithm to manage and treat TNFi-induced psoriasiform skin eruptions with decisions to continue therapy and “treat through” symptoms, switch to a different anti-TNF therapy, or switch to a different drug class based on severity of symptoms, whether the underlying disease is well controlled, and how patients with those underlying diseases have fared with those specific therapies or agents.
“We’ve shifted gears over the past decade, and we’ve gone from having very few agents and trying to keep patients desperately on one or two agents because we didn’t want to have to give up on them for their other comorbid disease, whether it was Crohn’s, colitis, RA, or whatever it may be,” senior author Joseph Merola, MD, director of the Center for Skin and Related Musculoskeletal Diseases at Brigham and Women’s Hospital, Boston, said in an interview. “We’re now in an area where we can have an algorithm like this, and we have so many more mechanistic options to move to.”
Dr. Merola, who is board certified in dermatology and rheumatology, said the algorithm is meant to “open a dialogue” with other specialists in different areas and raise awareness of treatments in related but separate fields. For diseases not often seen by more than one specialty, with the exception of psoriasis and PsA, he said that “the idea is to start a dialogue and increase communication between specialists.”
Dr. Merola noted that while the algorithm in many respects is meant to guide a physician in a specialty in appropriate medication decisions, at the same time he hopes that “it opens a dialogue and communication with the other specialty who tends to oversee this particular disease state or class of medicine to really work together to try to find the right drug for the right person.”
For patients with a mild skin eruption and a controlled underlying disease, the algorithm recommends a “treat through” approach by continuing anti-TNF therapy and treating psoriasis symptoms with topical steroids, ultraviolet therapy, methotrexate, cyclosporine, or acitretin, and to consider dapsone in cases of pustular psoriasis. However, the researchers noted that “treat through” studies have reported complete symptom resolution in 26%-41% of patients.
For patients with recalcitrant or worsening TNFi-induced psoriasis or patients with mild skin eruptions with an uncontrolled underlying disease, the researchers proposed considering switching to a different anti-TNF therapy, although studies have shown complete resolution of symptoms in only 5%-37% of patients.
If patients worsen from there, or if they have moderate to-severe skin eruption with uncontrolled underlying disease, they could be considered for switching to a different drug class and treated based on their underlying disease, along with treatment for psoriasis symptoms. This approach has been shown to completely resolve lesions in up to 64% of cases, they said. IBD patients could benefit from ustekinumab, vedolizumab, 6-mercaptopurine, or azathioprine as an alternative to anti-TNF therapy. Those patients with psoriasis should be considered for guselkumab, while ustekinumab, ixekizumab, secukinumab, and apremilast are effective treatments for patients with psoriasis and PsA. Patients with RA could receive treatment with tocilizumab, rituximab, abatacept, and tofacitinib, the authors wrote.
Dr. Merola reported serving as a consultant and/or investigator for Merck Research Laboratories, AbbVie, Dermavant, Eli Lilly, Novartis, Janssen, UCB, Samumed, Celgene, Sanofi Regeneron, GlaxoSmithKline, Almirall, Sun Pharma, Biogen, Pfizer, Incyte, Aclaris, and Leo Pharma.
SOURCE: Li SJ et al. J Psoriasis Psoriatic Arthritis. 2018 Nov 21. doi: 10.1177/2475530318810851.
Patients with tumor necrosis factor inhibitor–induced psoriasis could potentially be switched to a different drug class if they have moderate to severe skin eruption or mild skin eruption with an uncontrolled underlying disease such as inflammatory bowel disease, psoriasis, psoriatic arthritis, or rheumatoid arthritis, according to a new treatment algorithm proposed by researchers from Brigham and Women’s Hospital and Harvard Medical School in Boston.
The researchers outlined the prevalence of tumor necrosis factor–alpha inhibitor (TNFi)-induced psoriasis in a literature review of inflammatory bowel disease (IBD), psoriasis, psoriatic arthritis (PsA), and rheumatoid arthritis (RA) and identified an estimated rate of between 2.3% and 5% in patients with RA and between 1.6% and 2.7% in patients with IBD. Although there have been reports of TNFi-induced psoriasis in patients with psoriasis and PsA, the prevalence is unclear, they wrote in the Journal of Psoriasis and Psoriatic Arthritis.
The authors then created an algorithm to manage and treat TNFi-induced psoriasiform skin eruptions with decisions to continue therapy and “treat through” symptoms, switch to a different anti-TNF therapy, or switch to a different drug class based on severity of symptoms, whether the underlying disease is well controlled, and how patients with those underlying diseases have fared with those specific therapies or agents.
“We’ve shifted gears over the past decade, and we’ve gone from having very few agents and trying to keep patients desperately on one or two agents because we didn’t want to have to give up on them for their other comorbid disease, whether it was Crohn’s, colitis, RA, or whatever it may be,” senior author Joseph Merola, MD, director of the Center for Skin and Related Musculoskeletal Diseases at Brigham and Women’s Hospital, Boston, said in an interview. “We’re now in an area where we can have an algorithm like this, and we have so many more mechanistic options to move to.”
Dr. Merola, who is board certified in dermatology and rheumatology, said the algorithm is meant to “open a dialogue” with other specialists in different areas and raise awareness of treatments in related but separate fields. For diseases not often seen by more than one specialty, with the exception of psoriasis and PsA, he said that “the idea is to start a dialogue and increase communication between specialists.”
Dr. Merola noted that while the algorithm in many respects is meant to guide a physician in a specialty in appropriate medication decisions, at the same time he hopes that “it opens a dialogue and communication with the other specialty who tends to oversee this particular disease state or class of medicine to really work together to try to find the right drug for the right person.”
For patients with a mild skin eruption and a controlled underlying disease, the algorithm recommends a “treat through” approach by continuing anti-TNF therapy and treating psoriasis symptoms with topical steroids, ultraviolet therapy, methotrexate, cyclosporine, or acitretin, and to consider dapsone in cases of pustular psoriasis. However, the researchers noted that “treat through” studies have reported complete symptom resolution in 26%-41% of patients.
For patients with recalcitrant or worsening TNFi-induced psoriasis or patients with mild skin eruptions with an uncontrolled underlying disease, the researchers proposed considering switching to a different anti-TNF therapy, although studies have shown complete resolution of symptoms in only 5%-37% of patients.
If patients worsen from there, or if they have moderate to-severe skin eruption with uncontrolled underlying disease, they could be considered for switching to a different drug class and treated based on their underlying disease, along with treatment for psoriasis symptoms. This approach has been shown to completely resolve lesions in up to 64% of cases, they said. IBD patients could benefit from ustekinumab, vedolizumab, 6-mercaptopurine, or azathioprine as an alternative to anti-TNF therapy. Those patients with psoriasis should be considered for guselkumab, while ustekinumab, ixekizumab, secukinumab, and apremilast are effective treatments for patients with psoriasis and PsA. Patients with RA could receive treatment with tocilizumab, rituximab, abatacept, and tofacitinib, the authors wrote.
Dr. Merola reported serving as a consultant and/or investigator for Merck Research Laboratories, AbbVie, Dermavant, Eli Lilly, Novartis, Janssen, UCB, Samumed, Celgene, Sanofi Regeneron, GlaxoSmithKline, Almirall, Sun Pharma, Biogen, Pfizer, Incyte, Aclaris, and Leo Pharma.
SOURCE: Li SJ et al. J Psoriasis Psoriatic Arthritis. 2018 Nov 21. doi: 10.1177/2475530318810851.
FROM JOURNAL OF PSORIASIS AND PSORIATIC ARTHRITIS
Debunking Psoriasis Myths: Psoriasis Is More Than Skin Deep
Myth: Psoriasis Is Only a Skin Problem
Psoriasis is predominantly regarded as a skin disease because of the outward clinical presentation of the condition. However, psoriasis is a disorder of the immune system and its damage may be more than skin deep.
Psoriasis commonly presents on the skin and nails, but a growing body of evidence has suggested that psoriasis is associated with systemic comorbidities. Up to 25% of psoriasis patients develop joint inflammation, and psoriatic arthritis (PsA) may precede skin involvement. There also is a risk for cardiovascular complications. Because of the emotional distress caused by psoriasis, patients may develop psychosocial disorders. Other conditions in patients with psoriasis include diabetes mellitus, high blood pressure, Crohn disease, and the metabolic syndrome.
Results from surveys conducted by the National Psoriasis Foundation from 2003 to 2011 found that the diagnosis of psoriasis preceded PsA in the majority of patients by a mean period of 14.6 years. Patients with moderate to severe psoriasis were more likely to develop PsA than patients with mild psoriasis. Furthermore, patients with severe psoriasis were more likely to develop diabetes mellitus and cardiovascular disease.
In a Cutis editorial, Dr. Jeffrey Weinberg emphasizes that the role of the dermatologist “is to identify and educate patients with psoriasis who are at risk of systemic complications and ensure appropriate follow-up for their treatment and overall health.” An infographic created by the American Academy of Dermatology illustrates areas of the body that may be impacted by psoriasis beyond the skin; for example, patients may develop eye problems, weight gain, or mood changes. Consider distributing this infographic to patients to show how psoriasis can affect more than their skin.
More Cutis content is available on psoriasis comorbidities:
- Armstrong AW, Schupp C, Bebo B. Psoriasis comorbidities: results from the National Psoriasis Foundation surveys 2003 to 2011. Dermatology. 2012;225:121-126.
- Can psoriasis affect more than my skin? American Academy of Dermatology website. https://www.aad.org/public/diseases/scaly-skin/psoriasis/psoriasis-signs-and-symptoms/can-psoriasis-affect-more-than-my-skin. Accessed December 10, 2018.
- Psoriasis: more than skin deep. Harv Mens Health Watch. 2010;14:4-5. https://www.health.harvard.edu/newsletter_article/psoriasis-more-than-skin-deep. Accessed December 10, 2018.
- Weinberg JM. More than skin deep. Cutis. 2008;82:175.
Myth: Psoriasis Is Only a Skin Problem
Psoriasis is predominantly regarded as a skin disease because of the outward clinical presentation of the condition. However, psoriasis is a disorder of the immune system and its damage may be more than skin deep.
Psoriasis commonly presents on the skin and nails, but a growing body of evidence has suggested that psoriasis is associated with systemic comorbidities. Up to 25% of psoriasis patients develop joint inflammation, and psoriatic arthritis (PsA) may precede skin involvement. There also is a risk for cardiovascular complications. Because of the emotional distress caused by psoriasis, patients may develop psychosocial disorders. Other conditions in patients with psoriasis include diabetes mellitus, high blood pressure, Crohn disease, and the metabolic syndrome.
Results from surveys conducted by the National Psoriasis Foundation from 2003 to 2011 found that the diagnosis of psoriasis preceded PsA in the majority of patients by a mean period of 14.6 years. Patients with moderate to severe psoriasis were more likely to develop PsA than patients with mild psoriasis. Furthermore, patients with severe psoriasis were more likely to develop diabetes mellitus and cardiovascular disease.
In a Cutis editorial, Dr. Jeffrey Weinberg emphasizes that the role of the dermatologist “is to identify and educate patients with psoriasis who are at risk of systemic complications and ensure appropriate follow-up for their treatment and overall health.” An infographic created by the American Academy of Dermatology illustrates areas of the body that may be impacted by psoriasis beyond the skin; for example, patients may develop eye problems, weight gain, or mood changes. Consider distributing this infographic to patients to show how psoriasis can affect more than their skin.
More Cutis content is available on psoriasis comorbidities:
Myth: Psoriasis Is Only a Skin Problem
Psoriasis is predominantly regarded as a skin disease because of the outward clinical presentation of the condition. However, psoriasis is a disorder of the immune system and its damage may be more than skin deep.
Psoriasis commonly presents on the skin and nails, but a growing body of evidence has suggested that psoriasis is associated with systemic comorbidities. Up to 25% of psoriasis patients develop joint inflammation, and psoriatic arthritis (PsA) may precede skin involvement. There also is a risk for cardiovascular complications. Because of the emotional distress caused by psoriasis, patients may develop psychosocial disorders. Other conditions in patients with psoriasis include diabetes mellitus, high blood pressure, Crohn disease, and the metabolic syndrome.
Results from surveys conducted by the National Psoriasis Foundation from 2003 to 2011 found that the diagnosis of psoriasis preceded PsA in the majority of patients by a mean period of 14.6 years. Patients with moderate to severe psoriasis were more likely to develop PsA than patients with mild psoriasis. Furthermore, patients with severe psoriasis were more likely to develop diabetes mellitus and cardiovascular disease.
In a Cutis editorial, Dr. Jeffrey Weinberg emphasizes that the role of the dermatologist “is to identify and educate patients with psoriasis who are at risk of systemic complications and ensure appropriate follow-up for their treatment and overall health.” An infographic created by the American Academy of Dermatology illustrates areas of the body that may be impacted by psoriasis beyond the skin; for example, patients may develop eye problems, weight gain, or mood changes. Consider distributing this infographic to patients to show how psoriasis can affect more than their skin.
More Cutis content is available on psoriasis comorbidities:
- Armstrong AW, Schupp C, Bebo B. Psoriasis comorbidities: results from the National Psoriasis Foundation surveys 2003 to 2011. Dermatology. 2012;225:121-126.
- Can psoriasis affect more than my skin? American Academy of Dermatology website. https://www.aad.org/public/diseases/scaly-skin/psoriasis/psoriasis-signs-and-symptoms/can-psoriasis-affect-more-than-my-skin. Accessed December 10, 2018.
- Psoriasis: more than skin deep. Harv Mens Health Watch. 2010;14:4-5. https://www.health.harvard.edu/newsletter_article/psoriasis-more-than-skin-deep. Accessed December 10, 2018.
- Weinberg JM. More than skin deep. Cutis. 2008;82:175.
- Armstrong AW, Schupp C, Bebo B. Psoriasis comorbidities: results from the National Psoriasis Foundation surveys 2003 to 2011. Dermatology. 2012;225:121-126.
- Can psoriasis affect more than my skin? American Academy of Dermatology website. https://www.aad.org/public/diseases/scaly-skin/psoriasis/psoriasis-signs-and-symptoms/can-psoriasis-affect-more-than-my-skin. Accessed December 10, 2018.
- Psoriasis: more than skin deep. Harv Mens Health Watch. 2010;14:4-5. https://www.health.harvard.edu/newsletter_article/psoriasis-more-than-skin-deep. Accessed December 10, 2018.
- Weinberg JM. More than skin deep. Cutis. 2008;82:175.
Weight loss cuts risk of psoriatic arthritis
CHICAGO – Overweight and obese psoriasis patients have it within their power to reduce their risk of developing psoriatic arthritis through weight loss, according to a large British longitudinal study.
Of the three modifiable lifestyle factors evaluated in the study as potential risk factors for the development of psoriatic arthritis in psoriasis patients – body mass index, smoking, and alcohol intake – reduction in BMI over time was clearly the winning strategy, Neil McHugh, MD, said at the annual meeting of the American College of Rheumatology.
The message from this study of 90,189 incident cases of psoriasis identified in the U.K. Clinical Practice Research Datalink was unequivocal: “If you’re overweight and have psoriasis and you lose weight, you reduce your chance of developing a nasty form of arthritis,” said Dr. McHugh, professor of pharmacoepidemiology and a rheumatologist at the University of Bath, England.
“As psoriatic arthritis affects around 20% of people with psoriasis, weight reduction amongst those who are obese may have the potential to greatly reduce their risk of psoriatic arthritis in addition to providing additional health benefits,” he added.
Among the more than 90,000 patients diagnosed with psoriasis, 1,409 subsequently developed psoriatic arthritis, with an overall incidence rate of 2.72 cases per 1,000 person-years. Baseline BMI was strongly associated in stepwise fashion with subsequent psoriatic arthritis. Psoriasis patients with a baseline BMI of 25-29.9 kg/m2 were at an adjusted 1.76-fold increased risk of later developing psoriatic arthritis, compared with psoriasis patients having a BMI of less than 25. For those with a BMI of 30-34.9 kg/m2, the risk of subsequent psoriatic arthritis was increased 2.04-fold. And for those with a baseline BMI of 35 kg/m2 or more, the risk was increased 2.42-fold in analyses adjusted for age, sex, psoriasis duration and severity, history of trauma, and diabetes.
In contrast, the risk of developing psoriatic arthritis wasn’t significantly different between psoriasis patients who were nonsmokers, ex-smokers, or current smokers. And while there was a significantly increased risk of developing psoriatic arthritis in psoriasis patients who were current drinkers, compared with nondrinkers, the risk in ex-drinkers and heavy drinkers was similar to that in nondrinkers, a counterintuitive finding Dr. McHugh suspects was a distortion due to small numbers.
While the observed relationship between baseline BMI and subsequent risk of psoriatic arthritis was informative, it only tells part of the story, since body weight so often changes over time. Dr. McHugh and his coinvestigators had data on change in BMI over the course of 10 years of follow-up in 15,627 psoriasis patients free of psoriatic arthritis at the time their psoriasis was diagnosed. The researchers developed a BMI risk calculator that expressed the effect of change in BMI over time on the cumulative risk of developing psoriatic arthritis.
“We were able to show that if, for instance, you started with a BMI of 25 at baseline and ended up with a BMI of 30, your risk of psoriatic arthritis goes up by 13%, whereas if you start at 30 and come down to 25, your risk decreases by 13%. And the more weight you lose, the greater you reduce your risk of developing psoriatic arthritis,” the rheumatologist explained in an interview.
Indeed, with more extreme changes in BMI over the course of a decade following diagnosis of psoriasis – for example, dropping from a baseline BMI of 36 kg/m2 to 23 kg/m2 – the risk of developing psoriatic arthritis fell by close to 30%.
Dr. McHugh reported having no financial conflicts regarding this study, funded by the U.K. National Institute for Health Research.
SOURCE: Green A et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 2134.
CHICAGO – Overweight and obese psoriasis patients have it within their power to reduce their risk of developing psoriatic arthritis through weight loss, according to a large British longitudinal study.
Of the three modifiable lifestyle factors evaluated in the study as potential risk factors for the development of psoriatic arthritis in psoriasis patients – body mass index, smoking, and alcohol intake – reduction in BMI over time was clearly the winning strategy, Neil McHugh, MD, said at the annual meeting of the American College of Rheumatology.
The message from this study of 90,189 incident cases of psoriasis identified in the U.K. Clinical Practice Research Datalink was unequivocal: “If you’re overweight and have psoriasis and you lose weight, you reduce your chance of developing a nasty form of arthritis,” said Dr. McHugh, professor of pharmacoepidemiology and a rheumatologist at the University of Bath, England.
“As psoriatic arthritis affects around 20% of people with psoriasis, weight reduction amongst those who are obese may have the potential to greatly reduce their risk of psoriatic arthritis in addition to providing additional health benefits,” he added.
Among the more than 90,000 patients diagnosed with psoriasis, 1,409 subsequently developed psoriatic arthritis, with an overall incidence rate of 2.72 cases per 1,000 person-years. Baseline BMI was strongly associated in stepwise fashion with subsequent psoriatic arthritis. Psoriasis patients with a baseline BMI of 25-29.9 kg/m2 were at an adjusted 1.76-fold increased risk of later developing psoriatic arthritis, compared with psoriasis patients having a BMI of less than 25. For those with a BMI of 30-34.9 kg/m2, the risk of subsequent psoriatic arthritis was increased 2.04-fold. And for those with a baseline BMI of 35 kg/m2 or more, the risk was increased 2.42-fold in analyses adjusted for age, sex, psoriasis duration and severity, history of trauma, and diabetes.
In contrast, the risk of developing psoriatic arthritis wasn’t significantly different between psoriasis patients who were nonsmokers, ex-smokers, or current smokers. And while there was a significantly increased risk of developing psoriatic arthritis in psoriasis patients who were current drinkers, compared with nondrinkers, the risk in ex-drinkers and heavy drinkers was similar to that in nondrinkers, a counterintuitive finding Dr. McHugh suspects was a distortion due to small numbers.
While the observed relationship between baseline BMI and subsequent risk of psoriatic arthritis was informative, it only tells part of the story, since body weight so often changes over time. Dr. McHugh and his coinvestigators had data on change in BMI over the course of 10 years of follow-up in 15,627 psoriasis patients free of psoriatic arthritis at the time their psoriasis was diagnosed. The researchers developed a BMI risk calculator that expressed the effect of change in BMI over time on the cumulative risk of developing psoriatic arthritis.
“We were able to show that if, for instance, you started with a BMI of 25 at baseline and ended up with a BMI of 30, your risk of psoriatic arthritis goes up by 13%, whereas if you start at 30 and come down to 25, your risk decreases by 13%. And the more weight you lose, the greater you reduce your risk of developing psoriatic arthritis,” the rheumatologist explained in an interview.
Indeed, with more extreme changes in BMI over the course of a decade following diagnosis of psoriasis – for example, dropping from a baseline BMI of 36 kg/m2 to 23 kg/m2 – the risk of developing psoriatic arthritis fell by close to 30%.
Dr. McHugh reported having no financial conflicts regarding this study, funded by the U.K. National Institute for Health Research.
SOURCE: Green A et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 2134.
CHICAGO – Overweight and obese psoriasis patients have it within their power to reduce their risk of developing psoriatic arthritis through weight loss, according to a large British longitudinal study.
Of the three modifiable lifestyle factors evaluated in the study as potential risk factors for the development of psoriatic arthritis in psoriasis patients – body mass index, smoking, and alcohol intake – reduction in BMI over time was clearly the winning strategy, Neil McHugh, MD, said at the annual meeting of the American College of Rheumatology.
The message from this study of 90,189 incident cases of psoriasis identified in the U.K. Clinical Practice Research Datalink was unequivocal: “If you’re overweight and have psoriasis and you lose weight, you reduce your chance of developing a nasty form of arthritis,” said Dr. McHugh, professor of pharmacoepidemiology and a rheumatologist at the University of Bath, England.
“As psoriatic arthritis affects around 20% of people with psoriasis, weight reduction amongst those who are obese may have the potential to greatly reduce their risk of psoriatic arthritis in addition to providing additional health benefits,” he added.
Among the more than 90,000 patients diagnosed with psoriasis, 1,409 subsequently developed psoriatic arthritis, with an overall incidence rate of 2.72 cases per 1,000 person-years. Baseline BMI was strongly associated in stepwise fashion with subsequent psoriatic arthritis. Psoriasis patients with a baseline BMI of 25-29.9 kg/m2 were at an adjusted 1.76-fold increased risk of later developing psoriatic arthritis, compared with psoriasis patients having a BMI of less than 25. For those with a BMI of 30-34.9 kg/m2, the risk of subsequent psoriatic arthritis was increased 2.04-fold. And for those with a baseline BMI of 35 kg/m2 or more, the risk was increased 2.42-fold in analyses adjusted for age, sex, psoriasis duration and severity, history of trauma, and diabetes.
In contrast, the risk of developing psoriatic arthritis wasn’t significantly different between psoriasis patients who were nonsmokers, ex-smokers, or current smokers. And while there was a significantly increased risk of developing psoriatic arthritis in psoriasis patients who were current drinkers, compared with nondrinkers, the risk in ex-drinkers and heavy drinkers was similar to that in nondrinkers, a counterintuitive finding Dr. McHugh suspects was a distortion due to small numbers.
While the observed relationship between baseline BMI and subsequent risk of psoriatic arthritis was informative, it only tells part of the story, since body weight so often changes over time. Dr. McHugh and his coinvestigators had data on change in BMI over the course of 10 years of follow-up in 15,627 psoriasis patients free of psoriatic arthritis at the time their psoriasis was diagnosed. The researchers developed a BMI risk calculator that expressed the effect of change in BMI over time on the cumulative risk of developing psoriatic arthritis.
“We were able to show that if, for instance, you started with a BMI of 25 at baseline and ended up with a BMI of 30, your risk of psoriatic arthritis goes up by 13%, whereas if you start at 30 and come down to 25, your risk decreases by 13%. And the more weight you lose, the greater you reduce your risk of developing psoriatic arthritis,” the rheumatologist explained in an interview.
Indeed, with more extreme changes in BMI over the course of a decade following diagnosis of psoriasis – for example, dropping from a baseline BMI of 36 kg/m2 to 23 kg/m2 – the risk of developing psoriatic arthritis fell by close to 30%.
Dr. McHugh reported having no financial conflicts regarding this study, funded by the U.K. National Institute for Health Research.
SOURCE: Green A et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 2134.
REPORTING FROM THE ACR ANNUAL MEETING
Key clinical point:
Major finding: A psoriasis patient’s risk of developing psoriatic arthritis increases stepwise with greater body mass index, and the converse is true as well.
Study details: This study included more than 90,000 patients with a diagnosis of psoriasis in the U.K. Clinical Practice Research Datalink.
Disclosures: The presenter reported having no financial conflicts regarding this study, funded by the U.K. National Institute for Health Research.
Source: Green A et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 2134.
Treating psoriasis with biologics: Recommendations from an expert
LAS VEGAS – If you’re considering adding biologics for psoriasis to your clinical practice, dermatologist Kristina C. Duffin, MD, has some advice: Don’t expect to just use one drug, focus on comorbidities, and embrace strategies to bypass the potential obstacle of prior-authorization approvals.
Here are some tips from Dr. Duffin, who spoke at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar:
- Don’t expect a one-size-fits-all medication. “There is no one, single go-to drug,” said Dr. Duffin, who is cochair of the department of dermatology at the University of Utah, Salt Lake City. “Maybe someday, we will have a biological personalized medicine marker to say this is the right drug, but for now we don’t.” More than 10 biologics are available to treat psoriasis, she said, and more are in the pipeline.
- Pay close attention to comorbidities. It’s important to “have a good grasp” of a patient’s comorbidities, which can help focus the choice of a biologic, Dr. Duffin said. She recommends starting with an anti–tumor necrosis factor (TNF) agents for patients with psoriatic arthritis. For patients with Crohn’s disease, she recommends anti-TNF (adalimumab, infliximab) and anti-interleukin–12/23 or anti-IL-23 agents (ustekinumab). Anti-TNF agents should be avoided in patients with multiple sclerosis, and anti-IL-17 agents shouldn’t be given to patients with recurrent candidiasis, she noted.
- Encourage patients to make prompt decisions. Dr. Duffin sits down with patients to discuss various biologic options, and she goes over information in handouts. She also focuses on their needs: “Are they interested in getting better fast? Do they want to be clear for their wedding in a month?” She prefers to not let patients go home to think about what they’d like to do. Instead, she advises patients to make choices while at the office visit.
- Order lab tests and be careful about vaccines. Dr. Duffin orders the following tests for all patients who are starting on biologics: CBC, comprehensive metabolic panel, hepatitis B and C, and tuberculosis. She orders HIV, Hba1c and lipid tests, if appropriate. She prefers that patients treated with biologics avoid live vaccines. She suggests other vaccines, if indicated, such as seasonal influenza and pneumonia vaccines, and for those aged 50 years and older, herpes zoster vaccine. She urges patients to call the office if they have an infection or need surgery because they may need to discuss putting a temporary hold on the biologics.
- Understand how to navigate formularies.“Getting drugs approved for patients with Medicare is a challenge,” Dr. Duffin said. It’s helpful to understand how insurers handle specific psoriasis drugs so you can choose one that’s likely to be covered if you’re unsure which one is best. The website www.covermymeds.com allows physicians to easily check insurer formularies, free of charge, she said.
- Documentation is crucial when you’re dealing with an insurer. Document body surface area, Psoriasis Area Severity Index scores, or physician global assessment measures, she advised. An app provided by the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis, is a helpful in determining these measurements, she said. Also include information about failed treatments and the rationale behind why you chose a specific treatment, she said. “If denial happens, get the details,” she said. This may turn up a clerical error on the insurer’s part that incorrectly led to a denial.
- Escalate challenges to drug denials. If the preferred treatment is denied, one option is to appeal the denial. As a resource, Dr. Duffin pointed to sample letters for appealing denials for physicians and patients on the websites for the American Academy of Dermatology and the National Psoriasis Foundation. Ask for a limited 6-month approval, she said, or have the patient write a letter to the insurer using one of the sample letter templates. Another option is to ask the insurer for a “peer-to-peer” review, she said. “Sometimes it’s really hard for insurance company folks to say no to you if you have a really good story,” she commented.
- Help your patients get financial assistance. Almost every biologic manufacturer has a patient assistance plan, which can also help with deductibles and copays, Dr. Duffin said.
Dr. Duffin discloses consulting for AbbVie, Amgen, Celgene, Janssen, Lilly, Novartis, Pfizer, and Sienna. She has received grant/contracted research support from AbbVie, Bristol-Myers Squibb, Celgene, Eli Lilly, Janssen, Novartis, Pfizer, Sienna, Stiefel, and UCB.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – If you’re considering adding biologics for psoriasis to your clinical practice, dermatologist Kristina C. Duffin, MD, has some advice: Don’t expect to just use one drug, focus on comorbidities, and embrace strategies to bypass the potential obstacle of prior-authorization approvals.
Here are some tips from Dr. Duffin, who spoke at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar:
- Don’t expect a one-size-fits-all medication. “There is no one, single go-to drug,” said Dr. Duffin, who is cochair of the department of dermatology at the University of Utah, Salt Lake City. “Maybe someday, we will have a biological personalized medicine marker to say this is the right drug, but for now we don’t.” More than 10 biologics are available to treat psoriasis, she said, and more are in the pipeline.
- Pay close attention to comorbidities. It’s important to “have a good grasp” of a patient’s comorbidities, which can help focus the choice of a biologic, Dr. Duffin said. She recommends starting with an anti–tumor necrosis factor (TNF) agents for patients with psoriatic arthritis. For patients with Crohn’s disease, she recommends anti-TNF (adalimumab, infliximab) and anti-interleukin–12/23 or anti-IL-23 agents (ustekinumab). Anti-TNF agents should be avoided in patients with multiple sclerosis, and anti-IL-17 agents shouldn’t be given to patients with recurrent candidiasis, she noted.
- Encourage patients to make prompt decisions. Dr. Duffin sits down with patients to discuss various biologic options, and she goes over information in handouts. She also focuses on their needs: “Are they interested in getting better fast? Do they want to be clear for their wedding in a month?” She prefers to not let patients go home to think about what they’d like to do. Instead, she advises patients to make choices while at the office visit.
- Order lab tests and be careful about vaccines. Dr. Duffin orders the following tests for all patients who are starting on biologics: CBC, comprehensive metabolic panel, hepatitis B and C, and tuberculosis. She orders HIV, Hba1c and lipid tests, if appropriate. She prefers that patients treated with biologics avoid live vaccines. She suggests other vaccines, if indicated, such as seasonal influenza and pneumonia vaccines, and for those aged 50 years and older, herpes zoster vaccine. She urges patients to call the office if they have an infection or need surgery because they may need to discuss putting a temporary hold on the biologics.
- Understand how to navigate formularies.“Getting drugs approved for patients with Medicare is a challenge,” Dr. Duffin said. It’s helpful to understand how insurers handle specific psoriasis drugs so you can choose one that’s likely to be covered if you’re unsure which one is best. The website www.covermymeds.com allows physicians to easily check insurer formularies, free of charge, she said.
- Documentation is crucial when you’re dealing with an insurer. Document body surface area, Psoriasis Area Severity Index scores, or physician global assessment measures, she advised. An app provided by the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis, is a helpful in determining these measurements, she said. Also include information about failed treatments and the rationale behind why you chose a specific treatment, she said. “If denial happens, get the details,” she said. This may turn up a clerical error on the insurer’s part that incorrectly led to a denial.
- Escalate challenges to drug denials. If the preferred treatment is denied, one option is to appeal the denial. As a resource, Dr. Duffin pointed to sample letters for appealing denials for physicians and patients on the websites for the American Academy of Dermatology and the National Psoriasis Foundation. Ask for a limited 6-month approval, she said, or have the patient write a letter to the insurer using one of the sample letter templates. Another option is to ask the insurer for a “peer-to-peer” review, she said. “Sometimes it’s really hard for insurance company folks to say no to you if you have a really good story,” she commented.
- Help your patients get financial assistance. Almost every biologic manufacturer has a patient assistance plan, which can also help with deductibles and copays, Dr. Duffin said.
Dr. Duffin discloses consulting for AbbVie, Amgen, Celgene, Janssen, Lilly, Novartis, Pfizer, and Sienna. She has received grant/contracted research support from AbbVie, Bristol-Myers Squibb, Celgene, Eli Lilly, Janssen, Novartis, Pfizer, Sienna, Stiefel, and UCB.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – If you’re considering adding biologics for psoriasis to your clinical practice, dermatologist Kristina C. Duffin, MD, has some advice: Don’t expect to just use one drug, focus on comorbidities, and embrace strategies to bypass the potential obstacle of prior-authorization approvals.
Here are some tips from Dr. Duffin, who spoke at the Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar:
- Don’t expect a one-size-fits-all medication. “There is no one, single go-to drug,” said Dr. Duffin, who is cochair of the department of dermatology at the University of Utah, Salt Lake City. “Maybe someday, we will have a biological personalized medicine marker to say this is the right drug, but for now we don’t.” More than 10 biologics are available to treat psoriasis, she said, and more are in the pipeline.
- Pay close attention to comorbidities. It’s important to “have a good grasp” of a patient’s comorbidities, which can help focus the choice of a biologic, Dr. Duffin said. She recommends starting with an anti–tumor necrosis factor (TNF) agents for patients with psoriatic arthritis. For patients with Crohn’s disease, she recommends anti-TNF (adalimumab, infliximab) and anti-interleukin–12/23 or anti-IL-23 agents (ustekinumab). Anti-TNF agents should be avoided in patients with multiple sclerosis, and anti-IL-17 agents shouldn’t be given to patients with recurrent candidiasis, she noted.
- Encourage patients to make prompt decisions. Dr. Duffin sits down with patients to discuss various biologic options, and she goes over information in handouts. She also focuses on their needs: “Are they interested in getting better fast? Do they want to be clear for their wedding in a month?” She prefers to not let patients go home to think about what they’d like to do. Instead, she advises patients to make choices while at the office visit.
- Order lab tests and be careful about vaccines. Dr. Duffin orders the following tests for all patients who are starting on biologics: CBC, comprehensive metabolic panel, hepatitis B and C, and tuberculosis. She orders HIV, Hba1c and lipid tests, if appropriate. She prefers that patients treated with biologics avoid live vaccines. She suggests other vaccines, if indicated, such as seasonal influenza and pneumonia vaccines, and for those aged 50 years and older, herpes zoster vaccine. She urges patients to call the office if they have an infection or need surgery because they may need to discuss putting a temporary hold on the biologics.
- Understand how to navigate formularies.“Getting drugs approved for patients with Medicare is a challenge,” Dr. Duffin said. It’s helpful to understand how insurers handle specific psoriasis drugs so you can choose one that’s likely to be covered if you’re unsure which one is best. The website www.covermymeds.com allows physicians to easily check insurer formularies, free of charge, she said.
- Documentation is crucial when you’re dealing with an insurer. Document body surface area, Psoriasis Area Severity Index scores, or physician global assessment measures, she advised. An app provided by the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis, is a helpful in determining these measurements, she said. Also include information about failed treatments and the rationale behind why you chose a specific treatment, she said. “If denial happens, get the details,” she said. This may turn up a clerical error on the insurer’s part that incorrectly led to a denial.
- Escalate challenges to drug denials. If the preferred treatment is denied, one option is to appeal the denial. As a resource, Dr. Duffin pointed to sample letters for appealing denials for physicians and patients on the websites for the American Academy of Dermatology and the National Psoriasis Foundation. Ask for a limited 6-month approval, she said, or have the patient write a letter to the insurer using one of the sample letter templates. Another option is to ask the insurer for a “peer-to-peer” review, she said. “Sometimes it’s really hard for insurance company folks to say no to you if you have a really good story,” she commented.
- Help your patients get financial assistance. Almost every biologic manufacturer has a patient assistance plan, which can also help with deductibles and copays, Dr. Duffin said.
Dr. Duffin discloses consulting for AbbVie, Amgen, Celgene, Janssen, Lilly, Novartis, Pfizer, and Sienna. She has received grant/contracted research support from AbbVie, Bristol-Myers Squibb, Celgene, Eli Lilly, Janssen, Novartis, Pfizer, Sienna, Stiefel, and UCB.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Comorbidities are important in psoriasis care
LAS VEGAS – Pay attention to comorbidities in your psoriasis patients because there may not be anyone else doing so.
“Many of our patients don’t have primary care physicians; many are untreated for psoriasis. They come to a clinical trial to get treated – some of them may not have insurance – so it is important for us to watch for these comorbidities,” Kristina C. Duffin, MD, said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Yet, that does not seem to be happening consistently, according to Dr. Duffin, of the department of dermatology at the University of Utah, Salt Lake City. One in five dermatologists admitted to never screening or referring their psoriasis patients for management of cardiovascular risks in a 2015 survey (J Am Acad Dermatol. 2015 doi: 10.1016/j.jaad.2015.07.029).
Often patients at the start of biologic therapy are counseled about the risk for developing tuberculosis, yet the lifetime risk for doing so in the United States is 0.3%. Similarly, patients are often counseled on the risk for developing lymphoma, even though the excess risk for developing lymphoma that can be attributed to psoriasis treatment is 7.9 per 100,000 psoriasis patients per year. That screening seems to be driven by warnings issued in direct-to-consumer advertising, Dr. Duffin suggested.
“Although psoriasis patients have an increased relative risk of lymphoma, the absolute risk attributable to psoriasis is low,” Dr. Duffin pointed out.
Some of the comorbidities she advised dermatologists to watch for are described below.
Psoriatic arthritis
Psoriatic arthritis is the most important psoriasis comorbidity, Dr. Duffin said. Between 20% and 30% of psoriasis patients will develop psoriatic arthritis.
In a study of 1,511 patients in 48 centers in Germany, 21% of psoriasis patients were diagnosed with psoriatic arthritis and of those, more than 95% had active arthritis and 53% had five or more affected joints (Br J Dermatol. 2009;160[5]:1040-7).
The GRAPPA app is an easy, free screening tool for psoriatic arthritis; patients who score 3 or more out of 5 items on the psoriasis epidemiology screening tool (PEST) are deemed positive for psoriatic arthritis, Dr. Duffin noted.
Cardiovascular disease
Psoriasis patients are at increased risk of myocardial infarction, stroke, cardiovascular death, diabetes, and chronic kidney disease, Dr. Duffin said. In fact, CV risk from severe psoriasis is similar to the risk conferred by diabetes.
She added that there is epidemiologic evidence for CV risk modification with several of the biologics approved for psoriasis.
Hypertension
Hypertension is prevalent and more severe in psoriasis patients, Dr. Duffin said, citing a 2011 case-control study of electronic medical records at the University of California, Davis. Psoriasis patients with hypertension were 5 times more likely than patients without psoriasis to be on one antihypertensive medication, 9.5 times more likely to be on two, and almost 20 times more likely to be on four antihypertensive medications (PLoS One. 2011 Mar 29;6[3]:e18227. doi: 10.1371/journal.pone.0018227).
Importantly, few primary care physicians and cardiologists are aware of the increased risk for hypertension in psoriasis patients.
Less than half (45%) of primary care physicians and 57% of cardiologists reported they were aware that psoriasis was associated with worse cardiovascular outcome, and only 43% of physicians reported screening psoriasis patients for hypertension starting at age 20 years, according to a 2012 survey of 251 physicians (J Am Acad Dermatol. 2012 Sep;67[3]:357-62).
Dr. Duffin called on dermatologists to ensure that the primary care physicians they work with understand these increased risks.
“Commit to including a comment in consultation letters or letters back to primary care physicians that talks about the cardiovascular risks of the disease,” she said.
Dr. Duffin reported that she is a consultant and has received grant or contracted research support for many companies that manufacture dermatologic therapies.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Pay attention to comorbidities in your psoriasis patients because there may not be anyone else doing so.
“Many of our patients don’t have primary care physicians; many are untreated for psoriasis. They come to a clinical trial to get treated – some of them may not have insurance – so it is important for us to watch for these comorbidities,” Kristina C. Duffin, MD, said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Yet, that does not seem to be happening consistently, according to Dr. Duffin, of the department of dermatology at the University of Utah, Salt Lake City. One in five dermatologists admitted to never screening or referring their psoriasis patients for management of cardiovascular risks in a 2015 survey (J Am Acad Dermatol. 2015 doi: 10.1016/j.jaad.2015.07.029).
Often patients at the start of biologic therapy are counseled about the risk for developing tuberculosis, yet the lifetime risk for doing so in the United States is 0.3%. Similarly, patients are often counseled on the risk for developing lymphoma, even though the excess risk for developing lymphoma that can be attributed to psoriasis treatment is 7.9 per 100,000 psoriasis patients per year. That screening seems to be driven by warnings issued in direct-to-consumer advertising, Dr. Duffin suggested.
“Although psoriasis patients have an increased relative risk of lymphoma, the absolute risk attributable to psoriasis is low,” Dr. Duffin pointed out.
Some of the comorbidities she advised dermatologists to watch for are described below.
Psoriatic arthritis
Psoriatic arthritis is the most important psoriasis comorbidity, Dr. Duffin said. Between 20% and 30% of psoriasis patients will develop psoriatic arthritis.
In a study of 1,511 patients in 48 centers in Germany, 21% of psoriasis patients were diagnosed with psoriatic arthritis and of those, more than 95% had active arthritis and 53% had five or more affected joints (Br J Dermatol. 2009;160[5]:1040-7).
The GRAPPA app is an easy, free screening tool for psoriatic arthritis; patients who score 3 or more out of 5 items on the psoriasis epidemiology screening tool (PEST) are deemed positive for psoriatic arthritis, Dr. Duffin noted.
Cardiovascular disease
Psoriasis patients are at increased risk of myocardial infarction, stroke, cardiovascular death, diabetes, and chronic kidney disease, Dr. Duffin said. In fact, CV risk from severe psoriasis is similar to the risk conferred by diabetes.
She added that there is epidemiologic evidence for CV risk modification with several of the biologics approved for psoriasis.
Hypertension
Hypertension is prevalent and more severe in psoriasis patients, Dr. Duffin said, citing a 2011 case-control study of electronic medical records at the University of California, Davis. Psoriasis patients with hypertension were 5 times more likely than patients without psoriasis to be on one antihypertensive medication, 9.5 times more likely to be on two, and almost 20 times more likely to be on four antihypertensive medications (PLoS One. 2011 Mar 29;6[3]:e18227. doi: 10.1371/journal.pone.0018227).
Importantly, few primary care physicians and cardiologists are aware of the increased risk for hypertension in psoriasis patients.
Less than half (45%) of primary care physicians and 57% of cardiologists reported they were aware that psoriasis was associated with worse cardiovascular outcome, and only 43% of physicians reported screening psoriasis patients for hypertension starting at age 20 years, according to a 2012 survey of 251 physicians (J Am Acad Dermatol. 2012 Sep;67[3]:357-62).
Dr. Duffin called on dermatologists to ensure that the primary care physicians they work with understand these increased risks.
“Commit to including a comment in consultation letters or letters back to primary care physicians that talks about the cardiovascular risks of the disease,” she said.
Dr. Duffin reported that she is a consultant and has received grant or contracted research support for many companies that manufacture dermatologic therapies.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Pay attention to comorbidities in your psoriasis patients because there may not be anyone else doing so.
“Many of our patients don’t have primary care physicians; many are untreated for psoriasis. They come to a clinical trial to get treated – some of them may not have insurance – so it is important for us to watch for these comorbidities,” Kristina C. Duffin, MD, said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
Yet, that does not seem to be happening consistently, according to Dr. Duffin, of the department of dermatology at the University of Utah, Salt Lake City. One in five dermatologists admitted to never screening or referring their psoriasis patients for management of cardiovascular risks in a 2015 survey (J Am Acad Dermatol. 2015 doi: 10.1016/j.jaad.2015.07.029).
Often patients at the start of biologic therapy are counseled about the risk for developing tuberculosis, yet the lifetime risk for doing so in the United States is 0.3%. Similarly, patients are often counseled on the risk for developing lymphoma, even though the excess risk for developing lymphoma that can be attributed to psoriasis treatment is 7.9 per 100,000 psoriasis patients per year. That screening seems to be driven by warnings issued in direct-to-consumer advertising, Dr. Duffin suggested.
“Although psoriasis patients have an increased relative risk of lymphoma, the absolute risk attributable to psoriasis is low,” Dr. Duffin pointed out.
Some of the comorbidities she advised dermatologists to watch for are described below.
Psoriatic arthritis
Psoriatic arthritis is the most important psoriasis comorbidity, Dr. Duffin said. Between 20% and 30% of psoriasis patients will develop psoriatic arthritis.
In a study of 1,511 patients in 48 centers in Germany, 21% of psoriasis patients were diagnosed with psoriatic arthritis and of those, more than 95% had active arthritis and 53% had five or more affected joints (Br J Dermatol. 2009;160[5]:1040-7).
The GRAPPA app is an easy, free screening tool for psoriatic arthritis; patients who score 3 or more out of 5 items on the psoriasis epidemiology screening tool (PEST) are deemed positive for psoriatic arthritis, Dr. Duffin noted.
Cardiovascular disease
Psoriasis patients are at increased risk of myocardial infarction, stroke, cardiovascular death, diabetes, and chronic kidney disease, Dr. Duffin said. In fact, CV risk from severe psoriasis is similar to the risk conferred by diabetes.
She added that there is epidemiologic evidence for CV risk modification with several of the biologics approved for psoriasis.
Hypertension
Hypertension is prevalent and more severe in psoriasis patients, Dr. Duffin said, citing a 2011 case-control study of electronic medical records at the University of California, Davis. Psoriasis patients with hypertension were 5 times more likely than patients without psoriasis to be on one antihypertensive medication, 9.5 times more likely to be on two, and almost 20 times more likely to be on four antihypertensive medications (PLoS One. 2011 Mar 29;6[3]:e18227. doi: 10.1371/journal.pone.0018227).
Importantly, few primary care physicians and cardiologists are aware of the increased risk for hypertension in psoriasis patients.
Less than half (45%) of primary care physicians and 57% of cardiologists reported they were aware that psoriasis was associated with worse cardiovascular outcome, and only 43% of physicians reported screening psoriasis patients for hypertension starting at age 20 years, according to a 2012 survey of 251 physicians (J Am Acad Dermatol. 2012 Sep;67[3]:357-62).
Dr. Duffin called on dermatologists to ensure that the primary care physicians they work with understand these increased risks.
“Commit to including a comment in consultation letters or letters back to primary care physicians that talks about the cardiovascular risks of the disease,” she said.
Dr. Duffin reported that she is a consultant and has received grant or contracted research support for many companies that manufacture dermatologic therapies.
SDEF and this news organization are owned by the same parent company.
REPORTING FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Debunking Psoriasis Myths: Remove Psoriasis Scales Gently
Myth: Pick Psoriasis Scales to Remove Them
Patients may be inclined to pick psoriasis scales that appear in noticeable areas or on the scalp. However, they should be counseled to avoid this practice, which could cause an infection. Instead, Dr. Steven Feldman (Winston-Salem, North Carolina) suggests putting on an ointment or oil-like medication to soften the scale. “Almost any kind of moisturizer will change the reflective properties of the scale so that you don’t see the scale,” he advised. He also suggested descaling agents such as topical salicylic acid or lactic acid. His patient education video is available on the American Academy of Dermatology website should you wish to direct your patients to it.
Because salicylic acid is a keratolytic (or peeling agent), it works by causing the outer layer of skin to shed. When applied topically, it helps to soften and lift psoriasis scales. Coal tar over-the-counter products also can be used for the same purpose. The over-the-counter product guide from the National Psoriasis Foundation is a valuable resource to share with patients.
Expert Commentary
I agree that it is very important to treat scale very gently. In addition to risk for infection, picking and traumatizing scale can lead to worsening of the psoriasis. This is known as the Koebner phenomenon. The phenomenon was first described by Heinrich Koebner in 1876 as the formation of psoriatic lesions in uninvolved skin of patients with psoriasis after cutaneous trauma. This isomorphic phenomenon is now known to involve numerous diseases, among them vitiligo, lichen planus, and Darier disease.
—Jeffrey M. Weinberg, MD (New York, New York)
Feldman S. How should I remove psoriasis scale? American Academy of Dermatology website. https://www.aad.org/public/diseases/scaly-skin/psoriasis/tips-for-managing-psoriasis/how-should-i-remove-psoriasis-scale. Accessed October 31, 2018.
National Psoriasis Foundation. Over-the-counter products. https://www.aad.org/public/diseases/scaly-skin/psoriasis/tips-for-managing-psoriasis/how-should-i-remove-psoriasis-scale. Published June 2017. Accessed October 31, 2018.
Sagi L, Trau H. The Koebner phenomenon. Clin Dermatol. 2011;29:231-236.
Myth: Pick Psoriasis Scales to Remove Them
Patients may be inclined to pick psoriasis scales that appear in noticeable areas or on the scalp. However, they should be counseled to avoid this practice, which could cause an infection. Instead, Dr. Steven Feldman (Winston-Salem, North Carolina) suggests putting on an ointment or oil-like medication to soften the scale. “Almost any kind of moisturizer will change the reflective properties of the scale so that you don’t see the scale,” he advised. He also suggested descaling agents such as topical salicylic acid or lactic acid. His patient education video is available on the American Academy of Dermatology website should you wish to direct your patients to it.
Because salicylic acid is a keratolytic (or peeling agent), it works by causing the outer layer of skin to shed. When applied topically, it helps to soften and lift psoriasis scales. Coal tar over-the-counter products also can be used for the same purpose. The over-the-counter product guide from the National Psoriasis Foundation is a valuable resource to share with patients.
Expert Commentary
I agree that it is very important to treat scale very gently. In addition to risk for infection, picking and traumatizing scale can lead to worsening of the psoriasis. This is known as the Koebner phenomenon. The phenomenon was first described by Heinrich Koebner in 1876 as the formation of psoriatic lesions in uninvolved skin of patients with psoriasis after cutaneous trauma. This isomorphic phenomenon is now known to involve numerous diseases, among them vitiligo, lichen planus, and Darier disease.
—Jeffrey M. Weinberg, MD (New York, New York)
Myth: Pick Psoriasis Scales to Remove Them
Patients may be inclined to pick psoriasis scales that appear in noticeable areas or on the scalp. However, they should be counseled to avoid this practice, which could cause an infection. Instead, Dr. Steven Feldman (Winston-Salem, North Carolina) suggests putting on an ointment or oil-like medication to soften the scale. “Almost any kind of moisturizer will change the reflective properties of the scale so that you don’t see the scale,” he advised. He also suggested descaling agents such as topical salicylic acid or lactic acid. His patient education video is available on the American Academy of Dermatology website should you wish to direct your patients to it.
Because salicylic acid is a keratolytic (or peeling agent), it works by causing the outer layer of skin to shed. When applied topically, it helps to soften and lift psoriasis scales. Coal tar over-the-counter products also can be used for the same purpose. The over-the-counter product guide from the National Psoriasis Foundation is a valuable resource to share with patients.
Expert Commentary
I agree that it is very important to treat scale very gently. In addition to risk for infection, picking and traumatizing scale can lead to worsening of the psoriasis. This is known as the Koebner phenomenon. The phenomenon was first described by Heinrich Koebner in 1876 as the formation of psoriatic lesions in uninvolved skin of patients with psoriasis after cutaneous trauma. This isomorphic phenomenon is now known to involve numerous diseases, among them vitiligo, lichen planus, and Darier disease.
—Jeffrey M. Weinberg, MD (New York, New York)
Feldman S. How should I remove psoriasis scale? American Academy of Dermatology website. https://www.aad.org/public/diseases/scaly-skin/psoriasis/tips-for-managing-psoriasis/how-should-i-remove-psoriasis-scale. Accessed October 31, 2018.
National Psoriasis Foundation. Over-the-counter products. https://www.aad.org/public/diseases/scaly-skin/psoriasis/tips-for-managing-psoriasis/how-should-i-remove-psoriasis-scale. Published June 2017. Accessed October 31, 2018.
Sagi L, Trau H. The Koebner phenomenon. Clin Dermatol. 2011;29:231-236.
Feldman S. How should I remove psoriasis scale? American Academy of Dermatology website. https://www.aad.org/public/diseases/scaly-skin/psoriasis/tips-for-managing-psoriasis/how-should-i-remove-psoriasis-scale. Accessed October 31, 2018.
National Psoriasis Foundation. Over-the-counter products. https://www.aad.org/public/diseases/scaly-skin/psoriasis/tips-for-managing-psoriasis/how-should-i-remove-psoriasis-scale. Published June 2017. Accessed October 31, 2018.
Sagi L, Trau H. The Koebner phenomenon. Clin Dermatol. 2011;29:231-236.
Over one-third of psoriasis patients have PsA
Over one-third of psoriasis patients have PsA
About two-thirds of patients with psoriasis in a national registry also had psoriatic arthritis (PsA) and/or psoriasis in at least one challenging-to-treat (CTT) area, and one-quarter had both, according to Kristina Callis Duffin, MD, of the University of Utah, Salt Lake City, and her associates.
Their analysis included 2,042 psoriasis patients who were enrolled in the Corrona Psoriasis Registry between April 2015 and May 2018 and initiated biologic treatment during that time. The mean age was 49.6 years, 80% of the patients were white, and 51% were obese. Mean disease duration was 19.9 years and 89.2% of the patients had moderate to severe disease. CTT areas include the scalp, nails, and palmoplantar areas.
A total of 784 people in the cohort (38.4%) had PsA, 778 (38.1%) had scalp psoriasis, 326 (16.0%) had nail psoriasis, 223 (10.9%) had palmoplantar psoriasis, and 535 (26.2%) had both PsA and psoriasis in at least two CTT areas. The most common combinations were PsA plus scalp psoriasis and PsA plus nail and scalp psoriasis.
“These results indicate a need to further characterize patients with psoriasis who have PsA and CTT areas and evaluate the impact of these factors to better understand their treatment needs,” the investigators noted.
The Corrona registry has been supported by numerous pharmaceutical companies, and the study authors reported numerous financial relationships with industry; two authors are Novartis employees.
Secukinumab effective for slowing radiographic progression in active PsA
Treatment with secukinumab significantly reduced radiographic progression in patients with active PsA, according to Désirée van der Heijde, MD, PhD, professor of rheumatology at Leiden University Medical Center, and her associates.
The results come from an analysis of the FUTURE 5 trial, a study of 996 patients with active PsA despite previous NSAID treatment, disease-modifying antirheumatic drug treatment, or anti–tumor necrosis factor (TNF) therapy. Patients were randomized to receive 300 mg subcutaneous secukinumab with loading dose, 150 mg secukinumab with loading dose, 150 mg secukinumab without loading dose, or placebo, at baseline; weeks 1, 2, 3, and 4; then every 4 weeks.
After 24 weeks, the mean change in van der Heijde–modified Total Sharp Score for PsA was 0.08 for the 300-mg secukinumab group (P less than .01), 0.17 for the 150-mg secukinumab with loading dose group (P less than .05), a reduction of 0.09 for the 150-mg secukinumab without loading dose group (P less than .01), and 0.50 for the placebo group. Lower radiographic progression was seen regardless of prior anti-TNF or concomitant methotrexate treatment.
The study was funded by Novartis. The study authors reported financial disclosures with numerous companies; five authors are Novartis employees.
Tildrakizumab sustains efficacy in plaque psoriasis treatment after 1 year
Nearly all patients receiving the interleukin-23 inhibitor tildrakizumab for the treatment of moderate to severe plaque psoriasis maintained or improved their Psoriasis Area and Severity Index (PASI) response rate after 52 weeks of treatment, compared with their response after 28 weeks.
The analysis, conducted by Boni E. Elewski, MD, of the University of Alabama at Birmingham, and her associates, included 352 patients who received 100 mg tildrakizumab and 313 who received 200 mg tildrakizumab. Treatment was received at baseline, at 4 weeks, and then every 12 weeks afterward.
At week 28, the proportions of patients achieving PASI 100, PASI 90-99, PASI 75-89, and PASI 50-74 at week 28 were 25.9%, 38.4%, 25.3%, and 10.5%, respectively, among those treated with the 100-mg dose. The proportions were 24.6%, 24.3%, 19.5%, and 31.6%, respectively, among those treated with the 200-mg dose.
In patients who achieved at least PASI 90 on either dose at week 28, 88.9%-89.4% maintained that response at week 52. For patients with PASI 75-89, 39.3%-40.4% maintained that response and 33.7%-41.0% achieved a PASI 90 response. At week 52, in patients with PASI 50-74, 20.2%-29.7% achieved at least a PASI 90, 52.5%-64.9% achieved PASI 75, and only 2.6% of patients on either dose had fallen below PASI 50.
Four study authors reported being clinical investigators on studies sponsored by Merck and Sun Pharmaceuticals; five authors are employees of Sun Pharmaceuticals.
Halobetasol/tazarotene combination most effective for plaque psoriasis treatment
A fixed combination of halobetasol propionate 0.01% and tazarotene 0.045% lotion provided a synergistic effect over either component on its own for the treatment of plaque psoriasis, according to Leon H. Kircik, MD, of Indiana University, Indianapolis, and his associates.
The investigators performed a post hoc analysis of 212 patients with moderate to severe plaque psoriasis randomized to receive either the halobetasol/tazarotene combination, halobetasol only, tazarotene only, or vehicle only for 8 weeks, with follow-up at 12 weeks. Treatment success was based on the proportion of patients who achieved at least a 2-grade improvement in the Investigator Global Assessment (IGA) score, IGA scores of “clear” or “almost clear,” and percent change from baseline in IGA multiplied by Body Surface Area (BSA) composite score (IGAxBSA). “Synergy was calculated by summing up the contribution of the individual active ingredients (HP and TAZ) to overall efficacy and comparing to the efficacy achieved with HP/TAZ lotion relative to vehicle,” the authors explained.
Relative to vehicle, treatment success for halobetasol/tazarotene after 8 weeks was 42.8%, 23.6% for halobetasol alone, and 9.0% for tazarotene alone. After 12 weeks, the difference was 31.3%, 14.1%, and 5.9%, respectively. The percent change in IGAxBSA scores from baseline after 8 weeks, relative to vehicle, were 51.6%, 37.3%, and 3.3%, respectively. After 12 weeks, the change was 47.3%, 25.7%, and 8.6%, respectively.
After 8 weeks, the synergy ratio for treatment success and IGAxBSA scores for the halobetasol/tazarotene combination was 1.3. After 12 weeks, the synergy ratio for treatment success was 1.6 and the ratio for IGAxBSA scores was 1.4.
“By combining two agents into one once-daily formulation, this novel formulation reduces the number of product applications and may help patient adherence,” the study authors noted.
Dr. Kircik reported serving as a consultant and investigator for Valeant Pharmaceuticals. One study author is an employee of Bausch Health and Ortho Dermatologics, and another is an employee of Dow Pharmaceutical Sciences (a division of Valeant).
Brodalumab demonstrates low immunogenicity in moderate to severe psoriasis
The immunogenicity of brodalumab in patients with moderate to severe plaque psoriasis was low and did not compromise the efficacy or safety profile of the drug, according to Kristian Reich, MD, of Dermatologikum Berlin and SCIderm Research Institute in Hamburg, Germany, and his associates.
Data from a 12-week, phase 2 trial with a 352-week, open-label extension and three 52-week phase 3 trials were included in the analysis. Antidrug antibodies (ADAs) were tested, and positive samples were further analyzed for neutralizing ADAs by a cell-based assay.
Out of the 4,461 patients who received brodalumab, 122 (2.7%) were positive for ADAs after starting brodalumab. The incidence rate ranged from 1.9% to 3.4% between all dosing groups (140 mg, 210 mg, variable dosing, and 210 mg of brodalumab after ustekinumab). In 58 (1.4%) of patients, ADAs were transient. No patients had neutralizing ADAs, and no evidence of altered pharmacokinetics, loss of efficacy, or changes in the safety profile of brodalumab in subjects positive for ADAs was seen.
No significant difference was seen in the incidence rate of hypersensitivity or injection site reactions in brodalumab, compared with placebo or ustekinumab. The most common injection site reactions were injection site pain, erythema, and bruising.
The study was supported by Amgen. The study authors reported numerous disclosures. Two authors are employees of Leo Pharma, one author is a former employee of the company.
Secukinumab improves patient-reported outcomes in CTT psoriasis
Treatment with secukinumab significantly improved patient-reported outcomes such as fatigue, itch, pain, and quality of life measures in patients with CTT psoriasis after 6 months, according to Jerry Bagel, MD, of the Psoriasis Treatment Center of Central New Jersey, East Windsor, and his associates.
A total of 68 patients with psoriasis localized to at least one CTT area who were enrolled in the Corrona Psoriasis Registry from April 15, 2015, through May 10, 2018, and were receiving secukinumab for the entirety of the 6-month study period were included in the analysis. Patient-reported outcomes included in the analysis were fatigue, itch, pain, Dermatology Quality of Life Index (DLQI) score, and Work Productivity and Activity Impairment (WPAI) scale.
The mean age at enrollment was 51.2 years and almost 80% of patients were white. Mean psoriasis duration was 21.8 years and nearly half had PsA.
Visual analog scale scores improved over baseline for fatigue (mean, 23.2 vs. 33.2; P = .01), itch (20.9 vs. 49.6; P less than .0001), and pain (12.1 vs. 33.8; P less than .0001). DLQI scores also improved (2.9 vs. 8.1; P less than .0001), and the proportion of patients who reported that psoriasis had at least a moderate effect on their life was reduced after 6 months (22.1% vs. 59.7%; P less than .0001).
Based on WPAI results, patients experienced significant improvements in the percentage of daily activities impaired (mean, 9.5% vs 17.5%; P = .0075); of the 42 patients who were employed, both impairment percentage (3.7% vs. 11.2%; P = .0148) and percentage of work hours affected (4.9% vs. 11.9%; P = .0486) were reduced from baseline.
“These results are consistent with previous reports from secukinumab clinical trials; however, additional real-world studies are needed to evaluate the long-term effectiveness of secukinumab for improving [patient-reported outcomes] in patients with psoriasis in CTT areas,” the authors noted.
The Corrona registry has been supported by numerous pharmaceutical companies, and several study authors reported various disclosures with industry. Two authors are Novartis employees. The study was supported by Novartis; the company participated in the interpretation of data and review and approval of the abstract.
These posters were presented at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. SDEF and this news organization are owned by the same parent company.
Over one-third of psoriasis patients have PsA
About two-thirds of patients with psoriasis in a national registry also had psoriatic arthritis (PsA) and/or psoriasis in at least one challenging-to-treat (CTT) area, and one-quarter had both, according to Kristina Callis Duffin, MD, of the University of Utah, Salt Lake City, and her associates.
Their analysis included 2,042 psoriasis patients who were enrolled in the Corrona Psoriasis Registry between April 2015 and May 2018 and initiated biologic treatment during that time. The mean age was 49.6 years, 80% of the patients were white, and 51% were obese. Mean disease duration was 19.9 years and 89.2% of the patients had moderate to severe disease. CTT areas include the scalp, nails, and palmoplantar areas.
A total of 784 people in the cohort (38.4%) had PsA, 778 (38.1%) had scalp psoriasis, 326 (16.0%) had nail psoriasis, 223 (10.9%) had palmoplantar psoriasis, and 535 (26.2%) had both PsA and psoriasis in at least two CTT areas. The most common combinations were PsA plus scalp psoriasis and PsA plus nail and scalp psoriasis.
“These results indicate a need to further characterize patients with psoriasis who have PsA and CTT areas and evaluate the impact of these factors to better understand their treatment needs,” the investigators noted.
The Corrona registry has been supported by numerous pharmaceutical companies, and the study authors reported numerous financial relationships with industry; two authors are Novartis employees.
Secukinumab effective for slowing radiographic progression in active PsA
Treatment with secukinumab significantly reduced radiographic progression in patients with active PsA, according to Désirée van der Heijde, MD, PhD, professor of rheumatology at Leiden University Medical Center, and her associates.
The results come from an analysis of the FUTURE 5 trial, a study of 996 patients with active PsA despite previous NSAID treatment, disease-modifying antirheumatic drug treatment, or anti–tumor necrosis factor (TNF) therapy. Patients were randomized to receive 300 mg subcutaneous secukinumab with loading dose, 150 mg secukinumab with loading dose, 150 mg secukinumab without loading dose, or placebo, at baseline; weeks 1, 2, 3, and 4; then every 4 weeks.
After 24 weeks, the mean change in van der Heijde–modified Total Sharp Score for PsA was 0.08 for the 300-mg secukinumab group (P less than .01), 0.17 for the 150-mg secukinumab with loading dose group (P less than .05), a reduction of 0.09 for the 150-mg secukinumab without loading dose group (P less than .01), and 0.50 for the placebo group. Lower radiographic progression was seen regardless of prior anti-TNF or concomitant methotrexate treatment.
The study was funded by Novartis. The study authors reported financial disclosures with numerous companies; five authors are Novartis employees.
Tildrakizumab sustains efficacy in plaque psoriasis treatment after 1 year
Nearly all patients receiving the interleukin-23 inhibitor tildrakizumab for the treatment of moderate to severe plaque psoriasis maintained or improved their Psoriasis Area and Severity Index (PASI) response rate after 52 weeks of treatment, compared with their response after 28 weeks.
The analysis, conducted by Boni E. Elewski, MD, of the University of Alabama at Birmingham, and her associates, included 352 patients who received 100 mg tildrakizumab and 313 who received 200 mg tildrakizumab. Treatment was received at baseline, at 4 weeks, and then every 12 weeks afterward.
At week 28, the proportions of patients achieving PASI 100, PASI 90-99, PASI 75-89, and PASI 50-74 at week 28 were 25.9%, 38.4%, 25.3%, and 10.5%, respectively, among those treated with the 100-mg dose. The proportions were 24.6%, 24.3%, 19.5%, and 31.6%, respectively, among those treated with the 200-mg dose.
In patients who achieved at least PASI 90 on either dose at week 28, 88.9%-89.4% maintained that response at week 52. For patients with PASI 75-89, 39.3%-40.4% maintained that response and 33.7%-41.0% achieved a PASI 90 response. At week 52, in patients with PASI 50-74, 20.2%-29.7% achieved at least a PASI 90, 52.5%-64.9% achieved PASI 75, and only 2.6% of patients on either dose had fallen below PASI 50.
Four study authors reported being clinical investigators on studies sponsored by Merck and Sun Pharmaceuticals; five authors are employees of Sun Pharmaceuticals.
Halobetasol/tazarotene combination most effective for plaque psoriasis treatment
A fixed combination of halobetasol propionate 0.01% and tazarotene 0.045% lotion provided a synergistic effect over either component on its own for the treatment of plaque psoriasis, according to Leon H. Kircik, MD, of Indiana University, Indianapolis, and his associates.
The investigators performed a post hoc analysis of 212 patients with moderate to severe plaque psoriasis randomized to receive either the halobetasol/tazarotene combination, halobetasol only, tazarotene only, or vehicle only for 8 weeks, with follow-up at 12 weeks. Treatment success was based on the proportion of patients who achieved at least a 2-grade improvement in the Investigator Global Assessment (IGA) score, IGA scores of “clear” or “almost clear,” and percent change from baseline in IGA multiplied by Body Surface Area (BSA) composite score (IGAxBSA). “Synergy was calculated by summing up the contribution of the individual active ingredients (HP and TAZ) to overall efficacy and comparing to the efficacy achieved with HP/TAZ lotion relative to vehicle,” the authors explained.
Relative to vehicle, treatment success for halobetasol/tazarotene after 8 weeks was 42.8%, 23.6% for halobetasol alone, and 9.0% for tazarotene alone. After 12 weeks, the difference was 31.3%, 14.1%, and 5.9%, respectively. The percent change in IGAxBSA scores from baseline after 8 weeks, relative to vehicle, were 51.6%, 37.3%, and 3.3%, respectively. After 12 weeks, the change was 47.3%, 25.7%, and 8.6%, respectively.
After 8 weeks, the synergy ratio for treatment success and IGAxBSA scores for the halobetasol/tazarotene combination was 1.3. After 12 weeks, the synergy ratio for treatment success was 1.6 and the ratio for IGAxBSA scores was 1.4.
“By combining two agents into one once-daily formulation, this novel formulation reduces the number of product applications and may help patient adherence,” the study authors noted.
Dr. Kircik reported serving as a consultant and investigator for Valeant Pharmaceuticals. One study author is an employee of Bausch Health and Ortho Dermatologics, and another is an employee of Dow Pharmaceutical Sciences (a division of Valeant).
Brodalumab demonstrates low immunogenicity in moderate to severe psoriasis
The immunogenicity of brodalumab in patients with moderate to severe plaque psoriasis was low and did not compromise the efficacy or safety profile of the drug, according to Kristian Reich, MD, of Dermatologikum Berlin and SCIderm Research Institute in Hamburg, Germany, and his associates.
Data from a 12-week, phase 2 trial with a 352-week, open-label extension and three 52-week phase 3 trials were included in the analysis. Antidrug antibodies (ADAs) were tested, and positive samples were further analyzed for neutralizing ADAs by a cell-based assay.
Out of the 4,461 patients who received brodalumab, 122 (2.7%) were positive for ADAs after starting brodalumab. The incidence rate ranged from 1.9% to 3.4% between all dosing groups (140 mg, 210 mg, variable dosing, and 210 mg of brodalumab after ustekinumab). In 58 (1.4%) of patients, ADAs were transient. No patients had neutralizing ADAs, and no evidence of altered pharmacokinetics, loss of efficacy, or changes in the safety profile of brodalumab in subjects positive for ADAs was seen.
No significant difference was seen in the incidence rate of hypersensitivity or injection site reactions in brodalumab, compared with placebo or ustekinumab. The most common injection site reactions were injection site pain, erythema, and bruising.
The study was supported by Amgen. The study authors reported numerous disclosures. Two authors are employees of Leo Pharma, one author is a former employee of the company.
Secukinumab improves patient-reported outcomes in CTT psoriasis
Treatment with secukinumab significantly improved patient-reported outcomes such as fatigue, itch, pain, and quality of life measures in patients with CTT psoriasis after 6 months, according to Jerry Bagel, MD, of the Psoriasis Treatment Center of Central New Jersey, East Windsor, and his associates.
A total of 68 patients with psoriasis localized to at least one CTT area who were enrolled in the Corrona Psoriasis Registry from April 15, 2015, through May 10, 2018, and were receiving secukinumab for the entirety of the 6-month study period were included in the analysis. Patient-reported outcomes included in the analysis were fatigue, itch, pain, Dermatology Quality of Life Index (DLQI) score, and Work Productivity and Activity Impairment (WPAI) scale.
The mean age at enrollment was 51.2 years and almost 80% of patients were white. Mean psoriasis duration was 21.8 years and nearly half had PsA.
Visual analog scale scores improved over baseline for fatigue (mean, 23.2 vs. 33.2; P = .01), itch (20.9 vs. 49.6; P less than .0001), and pain (12.1 vs. 33.8; P less than .0001). DLQI scores also improved (2.9 vs. 8.1; P less than .0001), and the proportion of patients who reported that psoriasis had at least a moderate effect on their life was reduced after 6 months (22.1% vs. 59.7%; P less than .0001).
Based on WPAI results, patients experienced significant improvements in the percentage of daily activities impaired (mean, 9.5% vs 17.5%; P = .0075); of the 42 patients who were employed, both impairment percentage (3.7% vs. 11.2%; P = .0148) and percentage of work hours affected (4.9% vs. 11.9%; P = .0486) were reduced from baseline.
“These results are consistent with previous reports from secukinumab clinical trials; however, additional real-world studies are needed to evaluate the long-term effectiveness of secukinumab for improving [patient-reported outcomes] in patients with psoriasis in CTT areas,” the authors noted.
The Corrona registry has been supported by numerous pharmaceutical companies, and several study authors reported various disclosures with industry. Two authors are Novartis employees. The study was supported by Novartis; the company participated in the interpretation of data and review and approval of the abstract.
These posters were presented at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. SDEF and this news organization are owned by the same parent company.
Over one-third of psoriasis patients have PsA
About two-thirds of patients with psoriasis in a national registry also had psoriatic arthritis (PsA) and/or psoriasis in at least one challenging-to-treat (CTT) area, and one-quarter had both, according to Kristina Callis Duffin, MD, of the University of Utah, Salt Lake City, and her associates.
Their analysis included 2,042 psoriasis patients who were enrolled in the Corrona Psoriasis Registry between April 2015 and May 2018 and initiated biologic treatment during that time. The mean age was 49.6 years, 80% of the patients were white, and 51% were obese. Mean disease duration was 19.9 years and 89.2% of the patients had moderate to severe disease. CTT areas include the scalp, nails, and palmoplantar areas.
A total of 784 people in the cohort (38.4%) had PsA, 778 (38.1%) had scalp psoriasis, 326 (16.0%) had nail psoriasis, 223 (10.9%) had palmoplantar psoriasis, and 535 (26.2%) had both PsA and psoriasis in at least two CTT areas. The most common combinations were PsA plus scalp psoriasis and PsA plus nail and scalp psoriasis.
“These results indicate a need to further characterize patients with psoriasis who have PsA and CTT areas and evaluate the impact of these factors to better understand their treatment needs,” the investigators noted.
The Corrona registry has been supported by numerous pharmaceutical companies, and the study authors reported numerous financial relationships with industry; two authors are Novartis employees.
Secukinumab effective for slowing radiographic progression in active PsA
Treatment with secukinumab significantly reduced radiographic progression in patients with active PsA, according to Désirée van der Heijde, MD, PhD, professor of rheumatology at Leiden University Medical Center, and her associates.
The results come from an analysis of the FUTURE 5 trial, a study of 996 patients with active PsA despite previous NSAID treatment, disease-modifying antirheumatic drug treatment, or anti–tumor necrosis factor (TNF) therapy. Patients were randomized to receive 300 mg subcutaneous secukinumab with loading dose, 150 mg secukinumab with loading dose, 150 mg secukinumab without loading dose, or placebo, at baseline; weeks 1, 2, 3, and 4; then every 4 weeks.
After 24 weeks, the mean change in van der Heijde–modified Total Sharp Score for PsA was 0.08 for the 300-mg secukinumab group (P less than .01), 0.17 for the 150-mg secukinumab with loading dose group (P less than .05), a reduction of 0.09 for the 150-mg secukinumab without loading dose group (P less than .01), and 0.50 for the placebo group. Lower radiographic progression was seen regardless of prior anti-TNF or concomitant methotrexate treatment.
The study was funded by Novartis. The study authors reported financial disclosures with numerous companies; five authors are Novartis employees.
Tildrakizumab sustains efficacy in plaque psoriasis treatment after 1 year
Nearly all patients receiving the interleukin-23 inhibitor tildrakizumab for the treatment of moderate to severe plaque psoriasis maintained or improved their Psoriasis Area and Severity Index (PASI) response rate after 52 weeks of treatment, compared with their response after 28 weeks.
The analysis, conducted by Boni E. Elewski, MD, of the University of Alabama at Birmingham, and her associates, included 352 patients who received 100 mg tildrakizumab and 313 who received 200 mg tildrakizumab. Treatment was received at baseline, at 4 weeks, and then every 12 weeks afterward.
At week 28, the proportions of patients achieving PASI 100, PASI 90-99, PASI 75-89, and PASI 50-74 at week 28 were 25.9%, 38.4%, 25.3%, and 10.5%, respectively, among those treated with the 100-mg dose. The proportions were 24.6%, 24.3%, 19.5%, and 31.6%, respectively, among those treated with the 200-mg dose.
In patients who achieved at least PASI 90 on either dose at week 28, 88.9%-89.4% maintained that response at week 52. For patients with PASI 75-89, 39.3%-40.4% maintained that response and 33.7%-41.0% achieved a PASI 90 response. At week 52, in patients with PASI 50-74, 20.2%-29.7% achieved at least a PASI 90, 52.5%-64.9% achieved PASI 75, and only 2.6% of patients on either dose had fallen below PASI 50.
Four study authors reported being clinical investigators on studies sponsored by Merck and Sun Pharmaceuticals; five authors are employees of Sun Pharmaceuticals.
Halobetasol/tazarotene combination most effective for plaque psoriasis treatment
A fixed combination of halobetasol propionate 0.01% and tazarotene 0.045% lotion provided a synergistic effect over either component on its own for the treatment of plaque psoriasis, according to Leon H. Kircik, MD, of Indiana University, Indianapolis, and his associates.
The investigators performed a post hoc analysis of 212 patients with moderate to severe plaque psoriasis randomized to receive either the halobetasol/tazarotene combination, halobetasol only, tazarotene only, or vehicle only for 8 weeks, with follow-up at 12 weeks. Treatment success was based on the proportion of patients who achieved at least a 2-grade improvement in the Investigator Global Assessment (IGA) score, IGA scores of “clear” or “almost clear,” and percent change from baseline in IGA multiplied by Body Surface Area (BSA) composite score (IGAxBSA). “Synergy was calculated by summing up the contribution of the individual active ingredients (HP and TAZ) to overall efficacy and comparing to the efficacy achieved with HP/TAZ lotion relative to vehicle,” the authors explained.
Relative to vehicle, treatment success for halobetasol/tazarotene after 8 weeks was 42.8%, 23.6% for halobetasol alone, and 9.0% for tazarotene alone. After 12 weeks, the difference was 31.3%, 14.1%, and 5.9%, respectively. The percent change in IGAxBSA scores from baseline after 8 weeks, relative to vehicle, were 51.6%, 37.3%, and 3.3%, respectively. After 12 weeks, the change was 47.3%, 25.7%, and 8.6%, respectively.
After 8 weeks, the synergy ratio for treatment success and IGAxBSA scores for the halobetasol/tazarotene combination was 1.3. After 12 weeks, the synergy ratio for treatment success was 1.6 and the ratio for IGAxBSA scores was 1.4.
“By combining two agents into one once-daily formulation, this novel formulation reduces the number of product applications and may help patient adherence,” the study authors noted.
Dr. Kircik reported serving as a consultant and investigator for Valeant Pharmaceuticals. One study author is an employee of Bausch Health and Ortho Dermatologics, and another is an employee of Dow Pharmaceutical Sciences (a division of Valeant).
Brodalumab demonstrates low immunogenicity in moderate to severe psoriasis
The immunogenicity of brodalumab in patients with moderate to severe plaque psoriasis was low and did not compromise the efficacy or safety profile of the drug, according to Kristian Reich, MD, of Dermatologikum Berlin and SCIderm Research Institute in Hamburg, Germany, and his associates.
Data from a 12-week, phase 2 trial with a 352-week, open-label extension and three 52-week phase 3 trials were included in the analysis. Antidrug antibodies (ADAs) were tested, and positive samples were further analyzed for neutralizing ADAs by a cell-based assay.
Out of the 4,461 patients who received brodalumab, 122 (2.7%) were positive for ADAs after starting brodalumab. The incidence rate ranged from 1.9% to 3.4% between all dosing groups (140 mg, 210 mg, variable dosing, and 210 mg of brodalumab after ustekinumab). In 58 (1.4%) of patients, ADAs were transient. No patients had neutralizing ADAs, and no evidence of altered pharmacokinetics, loss of efficacy, or changes in the safety profile of brodalumab in subjects positive for ADAs was seen.
No significant difference was seen in the incidence rate of hypersensitivity or injection site reactions in brodalumab, compared with placebo or ustekinumab. The most common injection site reactions were injection site pain, erythema, and bruising.
The study was supported by Amgen. The study authors reported numerous disclosures. Two authors are employees of Leo Pharma, one author is a former employee of the company.
Secukinumab improves patient-reported outcomes in CTT psoriasis
Treatment with secukinumab significantly improved patient-reported outcomes such as fatigue, itch, pain, and quality of life measures in patients with CTT psoriasis after 6 months, according to Jerry Bagel, MD, of the Psoriasis Treatment Center of Central New Jersey, East Windsor, and his associates.
A total of 68 patients with psoriasis localized to at least one CTT area who were enrolled in the Corrona Psoriasis Registry from April 15, 2015, through May 10, 2018, and were receiving secukinumab for the entirety of the 6-month study period were included in the analysis. Patient-reported outcomes included in the analysis were fatigue, itch, pain, Dermatology Quality of Life Index (DLQI) score, and Work Productivity and Activity Impairment (WPAI) scale.
The mean age at enrollment was 51.2 years and almost 80% of patients were white. Mean psoriasis duration was 21.8 years and nearly half had PsA.
Visual analog scale scores improved over baseline for fatigue (mean, 23.2 vs. 33.2; P = .01), itch (20.9 vs. 49.6; P less than .0001), and pain (12.1 vs. 33.8; P less than .0001). DLQI scores also improved (2.9 vs. 8.1; P less than .0001), and the proportion of patients who reported that psoriasis had at least a moderate effect on their life was reduced after 6 months (22.1% vs. 59.7%; P less than .0001).
Based on WPAI results, patients experienced significant improvements in the percentage of daily activities impaired (mean, 9.5% vs 17.5%; P = .0075); of the 42 patients who were employed, both impairment percentage (3.7% vs. 11.2%; P = .0148) and percentage of work hours affected (4.9% vs. 11.9%; P = .0486) were reduced from baseline.
“These results are consistent with previous reports from secukinumab clinical trials; however, additional real-world studies are needed to evaluate the long-term effectiveness of secukinumab for improving [patient-reported outcomes] in patients with psoriasis in CTT areas,” the authors noted.
The Corrona registry has been supported by numerous pharmaceutical companies, and several study authors reported various disclosures with industry. Two authors are Novartis employees. The study was supported by Novartis; the company participated in the interpretation of data and review and approval of the abstract.
These posters were presented at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar. SDEF and this news organization are owned by the same parent company.
FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
FDA approves adalimumab biosimilar Hyrimoz
The Food and Drug Administration has approved the adalimumab biosimilar Hyrimoz (adalimumab-adaz) for a variety of conditions, according to Sandoz, the drug’s manufacturer and a division of Novartis.
FDA approval for Hyrimoz is based on a randomized, double-blind, three-arm, parallel biosimilarity study that demonstrated equivalence for all primary pharmacokinetic parameters, according to the press release. A second study confirmed these results in patients with moderate to severe plaque psoriasis, with Hyrimoz having a safety profile similar to that of adalimumab. Hyrimoz was approved in Europe in July 2018.
Hyrimoz has been approved to treat rheumatoid arthritis, juvenile idiopathic arthritis in patients aged 4 years and older, psoriatic arthritis, ankylosing spondylitis, adult Crohn’s disease, ulcerative colitis, and plaque psoriasis. The most common adverse events associated with the drug, according to the label, are infections, injection site reactions, headache, and rash.
Hyrimoz is the third adalimumab biosimilar approved by the FDA.
“Biosimilars can help people suffering from chronic, debilitating conditions gain expanded access to important medicines that may change the outcome of their disease. With the FDA approval of Hyrimoz, Sandoz is one step closer to offering U.S. patients with autoimmune diseases the same critical access already available in Europe,” Stefan Hendriks, global head of biopharmaceuticals at Sandoz, said in the press release.
Find the full press release on the Novartis website.
The Food and Drug Administration has approved the adalimumab biosimilar Hyrimoz (adalimumab-adaz) for a variety of conditions, according to Sandoz, the drug’s manufacturer and a division of Novartis.
FDA approval for Hyrimoz is based on a randomized, double-blind, three-arm, parallel biosimilarity study that demonstrated equivalence for all primary pharmacokinetic parameters, according to the press release. A second study confirmed these results in patients with moderate to severe plaque psoriasis, with Hyrimoz having a safety profile similar to that of adalimumab. Hyrimoz was approved in Europe in July 2018.
Hyrimoz has been approved to treat rheumatoid arthritis, juvenile idiopathic arthritis in patients aged 4 years and older, psoriatic arthritis, ankylosing spondylitis, adult Crohn’s disease, ulcerative colitis, and plaque psoriasis. The most common adverse events associated with the drug, according to the label, are infections, injection site reactions, headache, and rash.
Hyrimoz is the third adalimumab biosimilar approved by the FDA.
“Biosimilars can help people suffering from chronic, debilitating conditions gain expanded access to important medicines that may change the outcome of their disease. With the FDA approval of Hyrimoz, Sandoz is one step closer to offering U.S. patients with autoimmune diseases the same critical access already available in Europe,” Stefan Hendriks, global head of biopharmaceuticals at Sandoz, said in the press release.
Find the full press release on the Novartis website.
The Food and Drug Administration has approved the adalimumab biosimilar Hyrimoz (adalimumab-adaz) for a variety of conditions, according to Sandoz, the drug’s manufacturer and a division of Novartis.
FDA approval for Hyrimoz is based on a randomized, double-blind, three-arm, parallel biosimilarity study that demonstrated equivalence for all primary pharmacokinetic parameters, according to the press release. A second study confirmed these results in patients with moderate to severe plaque psoriasis, with Hyrimoz having a safety profile similar to that of adalimumab. Hyrimoz was approved in Europe in July 2018.
Hyrimoz has been approved to treat rheumatoid arthritis, juvenile idiopathic arthritis in patients aged 4 years and older, psoriatic arthritis, ankylosing spondylitis, adult Crohn’s disease, ulcerative colitis, and plaque psoriasis. The most common adverse events associated with the drug, according to the label, are infections, injection site reactions, headache, and rash.
Hyrimoz is the third adalimumab biosimilar approved by the FDA.
“Biosimilars can help people suffering from chronic, debilitating conditions gain expanded access to important medicines that may change the outcome of their disease. With the FDA approval of Hyrimoz, Sandoz is one step closer to offering U.S. patients with autoimmune diseases the same critical access already available in Europe,” Stefan Hendriks, global head of biopharmaceuticals at Sandoz, said in the press release.
Find the full press release on the Novartis website.
Psoriasis adds to increased risk of cardiovascular procedures, surgery in patients with hypertension
compared with patients with hypertension alone.
“The results suggested that hypertensive patients with concurrent psoriasis experienced an earlier and more aggressive disease progression of hypertension, compared with general hypertensive patients,” Hsien-Yi Chiu, MD, PhD, from the department of dermatology at the National Taiwan University Hospital in Hsinchu, Taiwan, and his colleagues wrote in the Journal of Dermatology. “Thus, patients with hypertension and psoriasis should be considered for more aggressive strategies for prevention of primary [cardiovascular disease] and more intense assessments for cardiovascular interventions needed to improve [cardiovascular disease] outcome in these patients.”
They performed a nationwide cohort study of patients in the Taiwan National Health Insurance Research Database with new onset hypertension from 2005 to 2006. Those with psoriasis (4,039 patients) were matched by age and sex to patients in the database who were diagnosed with hypertension but not psoriasis; the mean follow-up was 5.62 years. Their mean age was 58 years and about 31% of the psoriasis cohort were female. They were divided into groups based on psoriasis severity (mild and severe psoriasis) and type (psoriasis with and without arthritis). Researchers noted patients with both psoriasis and hypertension also had higher rates of cerebrovascular disease, coronary heart disease, hyperlipidemia, and diabetes mellitus during the year prior to the study.
The outcome measured was having a cardiovascular procedure (percutaneous coronary intervention with/without stenting or percutaneous transluminal coronary angioplasty and transcatheter radiofrequency ablation for arrhythmia) and cardiovascular surgery (coronary artery bypass grafting and other surgery for heart valves, arrhythmia, cerebrovascular disease, peripheral vessels, and the aorta).
Patients with both psoriasis and hypertension were at an increased risk for having a cardiovascular procedure and surgery (adjusted hazard ratio, 1.28; 95% confidence interval, 1.07-1.53), compared with patients with only hypertension. The risk of this outcome was also increased among patients with severe psoriasis or psoriatic arthritis, compared with patients who had mild psoriasis (aHR, 1.22; 95% CI, 0.98-1.51) and with patients with psoriasis but not arthritis (aHR, 1.15; 95% CI, 0.84-1.58); however, the results did not reach statistical significance after adjustment, which the researchers attributed to the small subgroup size.
“Another possible explanation was that the observed increased requirement for cardiovascular procedure and surgery in patients with severe psoriasis was mediated by a complex interplay among inflammation, traditional risk factors for [cardiovascular disease], and antirheumatic drugs, which probably attenuate the effects conferred by psoriasis,” the authors wrote.
Limitations in the study included reliance on administrative claims data for psoriasis diagnosis, unavailability of some details of the cardiovascular procedures and surgery, lack of blood pressure data to identify hypertension severity, as well as unmeasured factors and confounders. Further, “comparative occurrence of a requirement for cardiovascular procedure and surgery in the two groups may be influenced by a competing risk for death,” the researchers noted.
This study was supported in part through grants by the National Taiwan University Hospital, Asia-Pacific La Roche–Posay Foundation 2014, and the Ministry of Science and Technology. Dr. Chiu is on the speaker’s bureau for AbbVie, Janssen Pharmaceuticals, Novartis, Eli Lilly and Pfizer. Another author has conducted clinical trials for or received fees for being a consultant or speaker for companies that include Abbvie, Boehringer Ingelheim, and Celgene. The remaining authors reported no relevant conflicts of interest.
SOURCE: Chiu H-Y et al. J Dermatol. 2018 Oct 16. doi: 10.1111/1346-8138.14654.
compared with patients with hypertension alone.
“The results suggested that hypertensive patients with concurrent psoriasis experienced an earlier and more aggressive disease progression of hypertension, compared with general hypertensive patients,” Hsien-Yi Chiu, MD, PhD, from the department of dermatology at the National Taiwan University Hospital in Hsinchu, Taiwan, and his colleagues wrote in the Journal of Dermatology. “Thus, patients with hypertension and psoriasis should be considered for more aggressive strategies for prevention of primary [cardiovascular disease] and more intense assessments for cardiovascular interventions needed to improve [cardiovascular disease] outcome in these patients.”
They performed a nationwide cohort study of patients in the Taiwan National Health Insurance Research Database with new onset hypertension from 2005 to 2006. Those with psoriasis (4,039 patients) were matched by age and sex to patients in the database who were diagnosed with hypertension but not psoriasis; the mean follow-up was 5.62 years. Their mean age was 58 years and about 31% of the psoriasis cohort were female. They were divided into groups based on psoriasis severity (mild and severe psoriasis) and type (psoriasis with and without arthritis). Researchers noted patients with both psoriasis and hypertension also had higher rates of cerebrovascular disease, coronary heart disease, hyperlipidemia, and diabetes mellitus during the year prior to the study.
The outcome measured was having a cardiovascular procedure (percutaneous coronary intervention with/without stenting or percutaneous transluminal coronary angioplasty and transcatheter radiofrequency ablation for arrhythmia) and cardiovascular surgery (coronary artery bypass grafting and other surgery for heart valves, arrhythmia, cerebrovascular disease, peripheral vessels, and the aorta).
Patients with both psoriasis and hypertension were at an increased risk for having a cardiovascular procedure and surgery (adjusted hazard ratio, 1.28; 95% confidence interval, 1.07-1.53), compared with patients with only hypertension. The risk of this outcome was also increased among patients with severe psoriasis or psoriatic arthritis, compared with patients who had mild psoriasis (aHR, 1.22; 95% CI, 0.98-1.51) and with patients with psoriasis but not arthritis (aHR, 1.15; 95% CI, 0.84-1.58); however, the results did not reach statistical significance after adjustment, which the researchers attributed to the small subgroup size.
“Another possible explanation was that the observed increased requirement for cardiovascular procedure and surgery in patients with severe psoriasis was mediated by a complex interplay among inflammation, traditional risk factors for [cardiovascular disease], and antirheumatic drugs, which probably attenuate the effects conferred by psoriasis,” the authors wrote.
Limitations in the study included reliance on administrative claims data for psoriasis diagnosis, unavailability of some details of the cardiovascular procedures and surgery, lack of blood pressure data to identify hypertension severity, as well as unmeasured factors and confounders. Further, “comparative occurrence of a requirement for cardiovascular procedure and surgery in the two groups may be influenced by a competing risk for death,” the researchers noted.
This study was supported in part through grants by the National Taiwan University Hospital, Asia-Pacific La Roche–Posay Foundation 2014, and the Ministry of Science and Technology. Dr. Chiu is on the speaker’s bureau for AbbVie, Janssen Pharmaceuticals, Novartis, Eli Lilly and Pfizer. Another author has conducted clinical trials for or received fees for being a consultant or speaker for companies that include Abbvie, Boehringer Ingelheim, and Celgene. The remaining authors reported no relevant conflicts of interest.
SOURCE: Chiu H-Y et al. J Dermatol. 2018 Oct 16. doi: 10.1111/1346-8138.14654.
compared with patients with hypertension alone.
“The results suggested that hypertensive patients with concurrent psoriasis experienced an earlier and more aggressive disease progression of hypertension, compared with general hypertensive patients,” Hsien-Yi Chiu, MD, PhD, from the department of dermatology at the National Taiwan University Hospital in Hsinchu, Taiwan, and his colleagues wrote in the Journal of Dermatology. “Thus, patients with hypertension and psoriasis should be considered for more aggressive strategies for prevention of primary [cardiovascular disease] and more intense assessments for cardiovascular interventions needed to improve [cardiovascular disease] outcome in these patients.”
They performed a nationwide cohort study of patients in the Taiwan National Health Insurance Research Database with new onset hypertension from 2005 to 2006. Those with psoriasis (4,039 patients) were matched by age and sex to patients in the database who were diagnosed with hypertension but not psoriasis; the mean follow-up was 5.62 years. Their mean age was 58 years and about 31% of the psoriasis cohort were female. They were divided into groups based on psoriasis severity (mild and severe psoriasis) and type (psoriasis with and without arthritis). Researchers noted patients with both psoriasis and hypertension also had higher rates of cerebrovascular disease, coronary heart disease, hyperlipidemia, and diabetes mellitus during the year prior to the study.
The outcome measured was having a cardiovascular procedure (percutaneous coronary intervention with/without stenting or percutaneous transluminal coronary angioplasty and transcatheter radiofrequency ablation for arrhythmia) and cardiovascular surgery (coronary artery bypass grafting and other surgery for heart valves, arrhythmia, cerebrovascular disease, peripheral vessels, and the aorta).
Patients with both psoriasis and hypertension were at an increased risk for having a cardiovascular procedure and surgery (adjusted hazard ratio, 1.28; 95% confidence interval, 1.07-1.53), compared with patients with only hypertension. The risk of this outcome was also increased among patients with severe psoriasis or psoriatic arthritis, compared with patients who had mild psoriasis (aHR, 1.22; 95% CI, 0.98-1.51) and with patients with psoriasis but not arthritis (aHR, 1.15; 95% CI, 0.84-1.58); however, the results did not reach statistical significance after adjustment, which the researchers attributed to the small subgroup size.
“Another possible explanation was that the observed increased requirement for cardiovascular procedure and surgery in patients with severe psoriasis was mediated by a complex interplay among inflammation, traditional risk factors for [cardiovascular disease], and antirheumatic drugs, which probably attenuate the effects conferred by psoriasis,” the authors wrote.
Limitations in the study included reliance on administrative claims data for psoriasis diagnosis, unavailability of some details of the cardiovascular procedures and surgery, lack of blood pressure data to identify hypertension severity, as well as unmeasured factors and confounders. Further, “comparative occurrence of a requirement for cardiovascular procedure and surgery in the two groups may be influenced by a competing risk for death,” the researchers noted.
This study was supported in part through grants by the National Taiwan University Hospital, Asia-Pacific La Roche–Posay Foundation 2014, and the Ministry of Science and Technology. Dr. Chiu is on the speaker’s bureau for AbbVie, Janssen Pharmaceuticals, Novartis, Eli Lilly and Pfizer. Another author has conducted clinical trials for or received fees for being a consultant or speaker for companies that include Abbvie, Boehringer Ingelheim, and Celgene. The remaining authors reported no relevant conflicts of interest.
SOURCE: Chiu H-Y et al. J Dermatol. 2018 Oct 16. doi: 10.1111/1346-8138.14654.
FROM THE JOURNAL OF DERMATOLOGY
Key clinical point: More aggressive cardiovascular disease preventive strategies should be considered in patients with hypertension who also have psoriasis.
Major finding: Patients with both psoriasis and hypertension were at an increased risk for requiring a cardiovascular procedure and surgery (adjusted hazard ratio, 1.28), compared with patients with hypertension alone.
Study details: A retrospective cohort study evaluated risk of this outcome in 4,039 patients with psoriasis and hypertension, compared with patients who had hypertension, matched for age and sex.
Disclosures: This study was supported in part through grants by the National Taiwan University Hospital Hsin-Chu Branch, Asia-Pacific La Roche–Posay Foundation 2014, and the Ministry of Science and Technology. Dr. Chiu is on the speaker’s bureau for companies including AbbVie, Novartis, and Eli Lilly. Another author has conducted clinical trials for or received fees for being a consultant or speaker for Abbvie, Boehringer Ingelheim, Celgene, Janssen Pharmaceuticals, Eli Lilly, Galderma, Novartis, and Pfizer. The other authors reported no relevant conflicts of interest.
Source: Chiu H-Y et al. J Dermatol. 2018 Oct 16. doi:10.1111/1346-8138.14654.
Psychosocial Impact of Psoriasis: A Review for Dermatology Residents
The psychosocial impact of psoriasis is a critical component of disease burden. Psoriatic patients have high rates of depression and anxiety, problems at work, and difficulties with interpersonal relationships and intimacy.1 A National Psoriasis Foundation (NPF) survey from 2003 to 2011 reported that psoriasis affects overall emotional well-being in 88% of patients and enjoyment of life in 82% of patients.2
The reasons for psychosocial burden stem from public misconceptions and disease stigma. A survey of 1005 individuals (age range, 16–64 years) about their perceptions of psoriasis revealed that 16.5% believed that psoriasis is contagious and 6.8% believed that psoriasis is related to personal hygiene.3 Fifty percent practiced discriminatory behavior toward psoriatic patients, including reluctance to shake hands (28.8%) and engage in sexual relations/intercourse (44.1%). Sixty-five percent of psoriatic patients felt their appearance is unsightly, and 73% felt self-conscious about having psoriasis.2
The psychosocial burden exists despite medical treatment of the disease. In a cross-sectional study of 1184 psoriatic patients, 70.2% had impaired quality of life (QOL) as measured by the dermatology life quality index (DLQI), even after receiving a 4-week treatment for psoriasis.4 Medical treatment of psoriasis is not enough; providers need to assess overall QOL and provide treatment and resources for these patients in addition to symptomatic management.
There have been many studies on the psychosocial burden of psoriasis, but few have focused on a dermatology resident’s role in addressing this issue. This article will review psychosocial domains—psychiatric comorbidities and social functioning including occupational functioning, interpersonal relationships, and sexual functioning— and discuss a dermatology resident’s role in assessing and addressing each of these areas.
Methods
A PubMed search of articles indexed for MEDLINE was conducted using the following terms: psoriasis, depression, anxiety, work productivity, sexual functioning, and interpersonal relationships. Selected articles covered prevalence, assessment, and management of each psychosocial domain.
Results
Psychiatric Comorbidities
Prevalence
A high prevalence of psychiatric comorbidities exists in psoriatic patients. In a study of 469,097 patients with psoriasis, depression was the third most prevalent comorbidity (17.91%), following hyperlipidemia (45.64%) and hypertension (42.19%).5 In a 10-year longitudinal, population-based, prospective cohort study, antidepressant prescriptions were twice as frequent in psoriatic patients (17.8%) compared to control (7.9%)(P<.001).6 In a meta-analysis of 98 studies investigating psoriatic patients and psychiatric comorbidities, patients with psoriasis were 1.5 times more likely to experience depression (odds ratio [OR]: 1.57; 95% CI, 1.40-1.76) and use antidepressants (OR: 4.24; 95% CI, 1.53-11.76) compared to control.7 Patients with psoriasis were more likely to attempt suicide (OR: 1.32; 95% CI, 1.14-1.54) and complete suicide (OR: 1.20; 95% CI, 1.04-1.39) compared to people without psoriasis.8 A 1-year cross-sectional study of 90 psoriatic patients reported 78.7% were diagnosed with depression and 76.7% were diagnosed with anxiety. Seventy-two percent reported both anxiety and depression, correlating with worse QOL (χ2=26.7; P<.05).9
Assessment
Psychiatric comorbidities are assessed using clinical judgment and formal screening questionnaires in research studies. Signs of depression in patients with psoriasis can manifest as poor treatment adherence and recurrent flares of psoriasis.10,11 Psoriatic patients with psychiatric comorbidities were less likely to be adherent to treatment (risk ratio: 0.35; P<.003).10 The patient health questionnaire (PHQ) 9 and generalized anxiety disorder scale (GAD) 7 are validated and reliable questionnaires. The first 2 questions in PHQ-9 and GAD-7 screen for depression and anxiety, respectively.12-14 These 2-question screens are practical in a fast-paced dermatology outpatient setting. Systematic questionnaires specifically targeting mood disorders may be more beneficial than the widely used DLQI, which may not adequately capture mood disorders. Over the course of 10 months, 607 patients with psoriasis were asked to fill out the PHQ-9, GAD-7, and DLQI. Thirty-eight percent of patients with major depressive disorder had a DLQI score lower than 10, while 46% of patients with generalized anxiety disorder had a DLQI score lower than 10.15 Other questionnaires, including the hospital anxiety and depression scale and Beck depression inventory, are valid instruments with high sensitivity but are commonly used for research purposes and may not be clinically feasible.16
Management
Dermatologists should refer patients with depression and/or anxiety to psychiatry. Interventions include pharmacologic and nonpharmacologic management. First-line therapy for depression and anxiety is a combination of selective serotonin reuptake inhibitors and cognitive behavioral therapy.17 In addition, providers can direct patients to online resources such as the NPF website, where patients with psoriasis can access information about the signs and symptoms of mood disorders and contact the patient navigation center for further help.18
Social Functioning
Occupational Prevalence
The NPF found that 92% of patients with psoriasis or psoriatic arthritis (PsA) surveyed between 2003 and 2011 cited their psoriasis as reason for unemployment.2 In a survey of 43 patients asked about social and occupational functioning using the social and occupational assessment scale, 62.5% of psoriatic patients reported distress at work and 51.1% reported decreased efficiency at work.19 A national online survey that was conducted in France and issued to patients with and without psoriasis assessed overall QOL and work productivity using the work productivity and activity impairment questionnaire for psoriasis (WPAI-PSO). Of 714 patients with psoriasis and PsA, the latter had a 57.6% decrease in work productivity over 7 days compared to 27.9% in controls (P<.05).20 Occupational impairment leads to lost wages and hinders advancement, further exacerbating the psychosocial burden of psoriasis.21
Occupational Assessment
Formal assessment of occupational function can be done with the WPAI-PSO, a 6-question valid instrument.22 Providers may look for risk factors associated with greater loss in work productivity to help identify and offer support for patients. Patients with increased severity of itching, pain, and scaling experienced a greater decrease in work productivity.21,23 Patients with PsA warrant early detection and treatment because they experience greater physical restraints that can interfere with work activities. Of the 459 psoriatic patients without a prior diagnosis of PsA who filled out the PsA screening and evaluation questionnaire, 144 (31.4%) received a score of 44 or higher and were referred to rheumatology for further evaluation with the classification criteria for PsA. Nine percent of patients failed to be screened and remained undiagnosed with PsA.24 In a study using the health assessment questionnaire to assess 400 patients with PsA, those with worse physical function due to joint pain and stiffness were less likely to remain employed (OR: 0.56; P=.02).25
Occupational Management
Identifying and coordinating symptoms of PsA between dermatology and rheumatology is beneficial for patients who experience debilitating symptoms. There are a variety of treatments available for PsA. According to the European League Against Rheumatism 2015 guidelines developed from expert opinion and systematic reviews for PsA management, there are 4 phases of treatment, with reassessment every 3 to 6 months for effectiveness of therapy.26,27 Phase I involves initiating nonsteroidal anti-inflammatory drugs with or without glucocorticoid injections. Phase II involves synthetic disease-modifying drugs, including methotrexate, leflunomide, sulfasalazine, or cyclosporine. Phase III involves adding a second synthetic disease-modifying drug or starting a biologic, such as an anti–tumor necrosis factor, IL-12/IL-23, or IL-17 inhibitor. Phase IV involves switching to a different drug in either aforementioned class.26,27 Treatment with biologics improves work productivity as assessed by WPAI-PSO for psoriasis and PsA.28-30 Encouraging patients to speak up in the workplace and request small accommodations such as timely breaks or ergonomic chairs can help patients feel more comfortable and supported in the work environment.18 Patients who felt supported at work were more likely to remain employed.25
Interpersonal Relationships Prevalence
Misinformation about psoriasis, fear of rejection, and feelings of isolation may contribute to interpersonal conflict. Patients have feelings of shame and self-consciousness that hinder them from engaging in social activities and seeking out relationships.31 Twenty-nine percent of patients feel that psoriasis has interfered with establishing relationships because of negative self-esteem associated with the disease,32 and 26.3% have experienced people avoiding physical contact.33 Family and spouses of patients with psoriasis may be secondarily affected due to economic and emotional distress. Ninety-eight percent of family members of psoriatic patients experienced emotional distress and 54% experienced the burden of care.34 In a survey of 63 relatives and partners of patients with psoriasis, 57% experienced psychological distress, including anxiety and worry over a psoriatic patient’s future.35
Interpersonal Relationships Assessment
Current available tools, including the DLQI and short form health survey, measure overall QOL, including social functioning, but may not be practical in a clinic setting. Although no quick-screening test to assess for this domain exists, providers are encouraged to ask patients about disease impact on interpersonal relationships. The family DLQI questionnaire, adapted from the DLQI, may help physicians and social workers evaluate the burden on a patient’s family members.34
Interpersonal Relationships Management
It may be difficult for providers to address problems with interpersonal relationships without accessible tools. Patients may not be accompanied by family or friends during appointments, and it is difficult to screen for these issues during visits. Providers may offer resources such as the NPF website, which provides information about support groups. It also provides tips on dating and connecting to others in the community who share similar experiences.18 Encouraging patients to seek family or couples therapy also may be beneficial. Increased social support can lead to better QOL and fewer depressive symptoms.36
Sexual Functioning Prevalence
Psoriasis affects both physical and psychological components of sexual function. Among 3485 patients with skin conditions who were surveyed about sexual function, 34% of psoriatic patients reported that psoriasis interfered with sexual functioning at least to a certain degree.37 Sexual impairment was strongly associated with depression, anxiety, and suicidal ideation; 24% of depressed patients and 20% of anxious patients experienced sexual problems a lot or very much, based on the DLQI.37 Depending on the questionnaire used, the prevalence of sexual dysfunction due to psoriasis ranged from 35.5% to 71.3%.38 In an observational cohort study of 158 participants (n=79 psoriasis patients and n=79 controls), 34.2% of patients with psoriasis experienced erectile dysfunction compared to 17.7% of controls.39 Forty-two percent of psoriatic patients with genital involvement reported dyspareunia, 32% reported worsening of genital psoriasis after intercourse, and 43% reported decreased frequency of intercourse.40
Sexual Functioning Assessment
The Skindex-29, DLQI, and psoriasis disability index are available QOL tools that include one question evaluating difficulties with sexual function. The
Sexual Functioning Management
Better disease control leads to improved sexual function, as patients experience fewer feelings of shame, anxiety, and depression, as well as improvement of physical symptoms that can interfere with sexual functioning.38,43,44 Reducing friction, warmth, and moisture, as well as avoiding tight clothing, can help those with genital psoriasis. Patients are advised to reapply topical medications after sexual intercourse. Patients also can apply makeup to disguise psoriasis and help reduce feelings of self-consciousness that can impede sexual intimacy.18
Comment
The psychosocial burden of psoriasis penetrates many facets of patient lives. Psoriasis can invoke feelings of shame and embarrassment that are worsened by the public’s misconceptions about psoriasis, resulting in serious mental health issues that can cause even greater disability. Depression and anxiety are prevalent in patients with psoriasis. The characteristic symptoms of pain and pruritus along with psychiatric comorbidities can have an underestimated impact on daily activities, including employment, interpersonal relationships, and sexual function. Such dysfunctions have serious implications toward wages, professional advancement, social support, and overall QOL.
Dermatology providers play an important role in screening for these problems through validated questionnaires and identifying risks. Simple screening questions such as the PHQ-9 can be beneficial and feasible during dermatology visits. Screening for PsA can help patients avoid problems at work. Sexual dysfunction is a sensitive topic; however, providers can use a 1-question screen from valid questionnaires and inquire about the location of lesions as opportunities to address this issue.
Interventions lead to better disease control, which concurrently improves overall QOL. These interventions depend on both patient adherence and a physician’s commitment to finding an optimal treatment regimen for each individual. Medical management; coordinating care; developing treatment plans with psychiatry, rheumatology, and primary care providers; and psychological counseling and services may be necessary and beneficial (Table). Offering accessible resources such as the NPF website helps patients access information outside the clinic when it is not feasible to address all these concerns in a single visit. Psoriasis requires more than just medical management; it requires dermatology providers to use a multidisciplinary approach to address the psychosocial aspects of the disease.
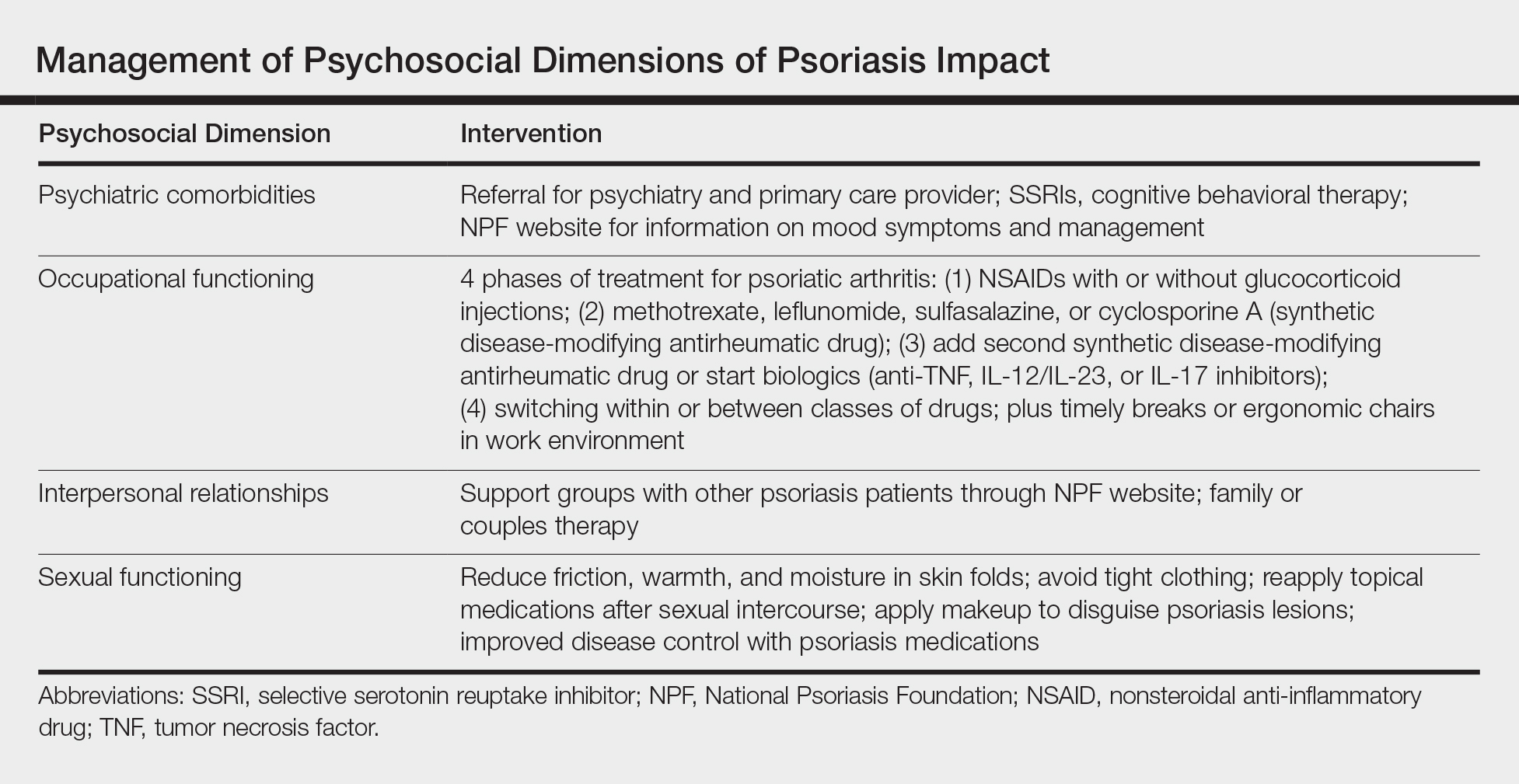
Conclusion
The psychosocial burden of psoriasis is immense. Stigma, public misconception, mental health concerns, and occupational and interpersonal difficulty are the basis of disease burden. Providers play a vital role in assessing the effect psoriasis has on different areas of patients’ lives and providing appropriate interventions and resources to reduce disease burden.
- Kimball AB, Jacobson C, Weiss S, et al. The psychosocial burden of psoriasis. Am J Clin Dermatol. 2005;6:383-392.
- Armstrong AW, Schupp C, Wu J, et al. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003-2011. PloS One. 2012;7:e52935.
- Halioua B, Sid-Mohand D, Roussel ME, et al. Extent of misconceptions, negative prejudices and discriminatory behaviour to psoriasis patients in France. J Eur Acad Dermatol Venereol. 2016;30:650-654.
- Wolf P, Weger W, Legat F, et al. Quality of life and treatment goals in psoriasis from the patient perspective: results of an Austrian cross-sectional survey. J Dtsch Dermatol Ges. 2018;16:981-990.
- Shah K, Mellars L, Changolkar A, et al. Real-world burden of comorbidities in US patients with psoriasis. J Am Acad Dermatol. 2017;77:287-292.e4.
- Dowlatshahi EA, Wakkee M, Herings RM, et al. Increased antidepressant drug exposure in psoriasis patients: a longitudinal population-based cohort study. Acta Derm Venereol. 2013;93:544-550.
- Dowlatshahi EA, Wakkee M, Arends LR, et al. The prevalence and odds of depressive symptoms and clinical depression in psoriasis patients: a systematic review and meta-analysis. J Invest Dermatol. 2014;134:1542-1551.
- Singh S, Taylor C, Kornmehl H, et al. Psoriasis and suicidality: a systematic review and meta-analysis. J Am Acad Dermatol. 2017;77:425.e2-440.e2.
- Lakshmy S, Balasundaram S, Sarkar S, et al. A cross-sectional study of prevalence and implications of depression and anxiety in psoriasis. Indian J Psychol Med. 2015;37:434-440.
- Renzi C, Picardi A, Abeni D, et al. Association of dissatisfaction with care and psychiatric morbidity with poor treatment compliance. Arch Dermatol. 2002;138:337-342.
- Kulkarni AS, Balkrishnan R, Camacho FT, et al. Medication and health care service utilization related to depressive symptoms in older adults with psoriasis. J Drugs Dermatol. 2004;3:661-666.
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613.
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097.
- Kroenke K, Spitzer RL, Williams JB. The Patient Health Questionnaire-2: validity of a two-item depression screener. Med Care. 2003;41:1284-1292.
- Lamb RC, Matcham F, Turner MA, et al. Screening for anxiety and depression in people with psoriasis: a cross-sectional study in a tertiary referral setting. Br J Dermatol. 2017;176:1028-1034.
- Law M, Naughton MT, Dhar A, et al. Validation of two depression screening instruments in a sleep disorders clinic. J Clin Sleep Med. 2014;10:683-688.
- Cuijpers P, Dekker J, Hollon SD, et al. Adding psychotherapy to pharmacotherapy in the treatment of depressive disorders in adults: a meta-analysis. J Clin Psychiatry. 2009;70:1219-1229.
- National Psoriasis Foundation. Living with psoriatic arthritis. https://www.psoriasis.org/life-with-psoriatic-arthritis. Accessed September 23, 2018.
- Gaikwad R, Deshpande S, Raje S, et al. Evaluation of functional impairment in psoriasis. Indian J Dermatol Venereol Leprol. 2006;72:37-40.
- Claudepierre P, Lahfa M, Levy P, et al. The impact of psoriasis on professional life: PsoPRO, a French national survey [published online April 6, 2018]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.14986.
- Korman NJ, Zhao Y, Pike J, et al. Relationship between psoriasis severity, clinical symptoms, quality of life and work productivity among patients in the USA. Clin Exp Dermatol. 2016;41:514-521.
- Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. PharmacoEconomics. 1993;4:353-365.
- Korman NJ, Zhao Y, Pike J, et al. Increased severity of itching, pain, and scaling in psoriasis patients is associated with increased disease severity, reduced quality of life, and reduced work productivity. Dermatol Online J. 2015;21.
- Spelman L, Su JC, Fernandez-Penas P, et al. Frequency of undiagnosed psoriatic arthritis among psoriasis patients in Australian dermatology practice. J Eur Acad Dermatol Venereol. 2015;29:2184-2191.
- Tillett W, Shaddick G, Askari A, et al. Factors influencing work disability in psoriatic arthritis: first results from a large UK multicentre study. Rheumatology (Oxford). 2015;54:157-162.
- Raychaudhuri SP, Wilken R, Sukhov AC, et al. Management of psoriatic arthritis: early diagnosis, monitoring of disease severity and cutting edge therapies. J Autoimmun. 2017;76:21-37.
- Gossec L, Smolen JS, Ramiro S, et al. European League Against Rheumatism (EULAR) recommendations for the manegement of psoriatic arthritis with pharmacological therapies: 2015 update. Ann Rheum Dis. 2016;75:499-510.
- Beroukhim K, Danesh M, Nguyen C, et al. A prospective, interventional assessment of the impact of ustekinumab treatment on psoriasis-related work productivity and activity impairment. J Dermatol Treat. 2016;27:552-555.
- Armstrong AW, Lynde CW, McBride SR, et al. Effect of ixekizumab treatment on work productivity for patients with moderate-to-severe plaque psoriasis: analysis of results from 3 randomized phase 3 clinical trials. JAMA Dermatol. 2016;152:661-669.
- Kimball AB, Yu AP, Signorovitch J, et al. The effects of adalimumab treatment and psoriasis severity on self-reported work productivity and activity impairment for patients with moderate to severe psoriasis. J Am Acad Dermatol. 2012;66:e67-76.
- Feldman SR, Malakouti M, Koo JY. Social impact of the burden of psoriasis: effects on patients and practice. Dermatol Online J. 2014;20.
- Reich A, Welz-Kubiak K, Rams Ł. Apprehension of the disease by patients suffering from psoriasis. Postepy Dermatol Alergol. 2014;31:289-293.
- Gupta MA, Gupta AK, Watteel GN. Perceived deprivation of social touch in psoriasis is associated with greater psychologic morbidity: an index of the stigma experience in dermatologic disorders. Cutis. 1998;61:339-342.
- Basra MK, Finlay AY. The family impact of skin diseases: the Greater Patient concept. Br J Dermatol. 2007;156:929-937.
- Eghlileb AM, Davies EE, Finlay AY. Psoriasis has a major secondary impact on the lives of family members and partners. Br J Dermatol. 2007;156:1245-1250.
- Janowski K, Steuden S, Pietrzak A, et al. Social support and adaptation to the disease in men and women with psoriasis. Arch Dermatol Res. 2012;304:421-432.
- Sampogna F, Abeni D, Gieler U, et al. Impairment of sexual life in 3,485 dermatological outpatients from a multicentre study in 13 European countries. Acta Derm Venereol. 2017;97:478-482.
- Sampogna F, Gisondi P, Tabolli S, et al. Impairment of sexual life in patients with psoriasis. Dermatology. 2007;214:144-150.
- Molina-Leyva A, Molina-Leyva I, Almodovar-Real A, et al. Prevalence and associated factors of erectile dysfunction in patients with moderate to severe psoriasis and healthy population: a comparative study considering physical and psychological factors. Arch Sex Behav. 2016;45:2047-2055.
- Ryan C, Sadlier M, De Vol E, et al. Genital psoriasis is associated with significant impairment in quality of life and sexual functioning. J Am Acad Dermatol. 2015;72:978-983.
- Labbate LA, Lare SB. Sexual dysfunction in male psychiatric outpatients: validity of the Massachusetts General Hospital Sexual Functioning Questionnaire. Psychother Psychosom. 2001;70:221-225.
- Molina-Leyva A, Almodovar-Real A, Ruiz-Carrascosa JC, et al. Distribution pattern of psoriasis affects sexual function in moderate to severe psoriasis: a prospective case series study. J Sex Med. 2014;11:2882-2889.
- Guenther L, Han C, Szapary P, et al. Impact of ustekinumab on health-related quality of life and sexual difficulties associated with psoriasis: results from two phase III clinical trials. J Eur Acad Dermatol Venereol. 2011;25:851-857.
- Guenther L, Warren RB, Cather JC, et al. Impact of ixekizumab treatment on skin-related personal relationship difficulties in moderate-to-severe psoriasis patients: 12-week results from two Phase 3 trials. J Eur Acad Dermatol Venereol. 2017;31:1867-1875.
The psychosocial impact of psoriasis is a critical component of disease burden. Psoriatic patients have high rates of depression and anxiety, problems at work, and difficulties with interpersonal relationships and intimacy.1 A National Psoriasis Foundation (NPF) survey from 2003 to 2011 reported that psoriasis affects overall emotional well-being in 88% of patients and enjoyment of life in 82% of patients.2
The reasons for psychosocial burden stem from public misconceptions and disease stigma. A survey of 1005 individuals (age range, 16–64 years) about their perceptions of psoriasis revealed that 16.5% believed that psoriasis is contagious and 6.8% believed that psoriasis is related to personal hygiene.3 Fifty percent practiced discriminatory behavior toward psoriatic patients, including reluctance to shake hands (28.8%) and engage in sexual relations/intercourse (44.1%). Sixty-five percent of psoriatic patients felt their appearance is unsightly, and 73% felt self-conscious about having psoriasis.2
The psychosocial burden exists despite medical treatment of the disease. In a cross-sectional study of 1184 psoriatic patients, 70.2% had impaired quality of life (QOL) as measured by the dermatology life quality index (DLQI), even after receiving a 4-week treatment for psoriasis.4 Medical treatment of psoriasis is not enough; providers need to assess overall QOL and provide treatment and resources for these patients in addition to symptomatic management.
There have been many studies on the psychosocial burden of psoriasis, but few have focused on a dermatology resident’s role in addressing this issue. This article will review psychosocial domains—psychiatric comorbidities and social functioning including occupational functioning, interpersonal relationships, and sexual functioning— and discuss a dermatology resident’s role in assessing and addressing each of these areas.
Methods
A PubMed search of articles indexed for MEDLINE was conducted using the following terms: psoriasis, depression, anxiety, work productivity, sexual functioning, and interpersonal relationships. Selected articles covered prevalence, assessment, and management of each psychosocial domain.
Results
Psychiatric Comorbidities
Prevalence
A high prevalence of psychiatric comorbidities exists in psoriatic patients. In a study of 469,097 patients with psoriasis, depression was the third most prevalent comorbidity (17.91%), following hyperlipidemia (45.64%) and hypertension (42.19%).5 In a 10-year longitudinal, population-based, prospective cohort study, antidepressant prescriptions were twice as frequent in psoriatic patients (17.8%) compared to control (7.9%)(P<.001).6 In a meta-analysis of 98 studies investigating psoriatic patients and psychiatric comorbidities, patients with psoriasis were 1.5 times more likely to experience depression (odds ratio [OR]: 1.57; 95% CI, 1.40-1.76) and use antidepressants (OR: 4.24; 95% CI, 1.53-11.76) compared to control.7 Patients with psoriasis were more likely to attempt suicide (OR: 1.32; 95% CI, 1.14-1.54) and complete suicide (OR: 1.20; 95% CI, 1.04-1.39) compared to people without psoriasis.8 A 1-year cross-sectional study of 90 psoriatic patients reported 78.7% were diagnosed with depression and 76.7% were diagnosed with anxiety. Seventy-two percent reported both anxiety and depression, correlating with worse QOL (χ2=26.7; P<.05).9
Assessment
Psychiatric comorbidities are assessed using clinical judgment and formal screening questionnaires in research studies. Signs of depression in patients with psoriasis can manifest as poor treatment adherence and recurrent flares of psoriasis.10,11 Psoriatic patients with psychiatric comorbidities were less likely to be adherent to treatment (risk ratio: 0.35; P<.003).10 The patient health questionnaire (PHQ) 9 and generalized anxiety disorder scale (GAD) 7 are validated and reliable questionnaires. The first 2 questions in PHQ-9 and GAD-7 screen for depression and anxiety, respectively.12-14 These 2-question screens are practical in a fast-paced dermatology outpatient setting. Systematic questionnaires specifically targeting mood disorders may be more beneficial than the widely used DLQI, which may not adequately capture mood disorders. Over the course of 10 months, 607 patients with psoriasis were asked to fill out the PHQ-9, GAD-7, and DLQI. Thirty-eight percent of patients with major depressive disorder had a DLQI score lower than 10, while 46% of patients with generalized anxiety disorder had a DLQI score lower than 10.15 Other questionnaires, including the hospital anxiety and depression scale and Beck depression inventory, are valid instruments with high sensitivity but are commonly used for research purposes and may not be clinically feasible.16
Management
Dermatologists should refer patients with depression and/or anxiety to psychiatry. Interventions include pharmacologic and nonpharmacologic management. First-line therapy for depression and anxiety is a combination of selective serotonin reuptake inhibitors and cognitive behavioral therapy.17 In addition, providers can direct patients to online resources such as the NPF website, where patients with psoriasis can access information about the signs and symptoms of mood disorders and contact the patient navigation center for further help.18
Social Functioning
Occupational Prevalence
The NPF found that 92% of patients with psoriasis or psoriatic arthritis (PsA) surveyed between 2003 and 2011 cited their psoriasis as reason for unemployment.2 In a survey of 43 patients asked about social and occupational functioning using the social and occupational assessment scale, 62.5% of psoriatic patients reported distress at work and 51.1% reported decreased efficiency at work.19 A national online survey that was conducted in France and issued to patients with and without psoriasis assessed overall QOL and work productivity using the work productivity and activity impairment questionnaire for psoriasis (WPAI-PSO). Of 714 patients with psoriasis and PsA, the latter had a 57.6% decrease in work productivity over 7 days compared to 27.9% in controls (P<.05).20 Occupational impairment leads to lost wages and hinders advancement, further exacerbating the psychosocial burden of psoriasis.21
Occupational Assessment
Formal assessment of occupational function can be done with the WPAI-PSO, a 6-question valid instrument.22 Providers may look for risk factors associated with greater loss in work productivity to help identify and offer support for patients. Patients with increased severity of itching, pain, and scaling experienced a greater decrease in work productivity.21,23 Patients with PsA warrant early detection and treatment because they experience greater physical restraints that can interfere with work activities. Of the 459 psoriatic patients without a prior diagnosis of PsA who filled out the PsA screening and evaluation questionnaire, 144 (31.4%) received a score of 44 or higher and were referred to rheumatology for further evaluation with the classification criteria for PsA. Nine percent of patients failed to be screened and remained undiagnosed with PsA.24 In a study using the health assessment questionnaire to assess 400 patients with PsA, those with worse physical function due to joint pain and stiffness were less likely to remain employed (OR: 0.56; P=.02).25
Occupational Management
Identifying and coordinating symptoms of PsA between dermatology and rheumatology is beneficial for patients who experience debilitating symptoms. There are a variety of treatments available for PsA. According to the European League Against Rheumatism 2015 guidelines developed from expert opinion and systematic reviews for PsA management, there are 4 phases of treatment, with reassessment every 3 to 6 months for effectiveness of therapy.26,27 Phase I involves initiating nonsteroidal anti-inflammatory drugs with or without glucocorticoid injections. Phase II involves synthetic disease-modifying drugs, including methotrexate, leflunomide, sulfasalazine, or cyclosporine. Phase III involves adding a second synthetic disease-modifying drug or starting a biologic, such as an anti–tumor necrosis factor, IL-12/IL-23, or IL-17 inhibitor. Phase IV involves switching to a different drug in either aforementioned class.26,27 Treatment with biologics improves work productivity as assessed by WPAI-PSO for psoriasis and PsA.28-30 Encouraging patients to speak up in the workplace and request small accommodations such as timely breaks or ergonomic chairs can help patients feel more comfortable and supported in the work environment.18 Patients who felt supported at work were more likely to remain employed.25
Interpersonal Relationships Prevalence
Misinformation about psoriasis, fear of rejection, and feelings of isolation may contribute to interpersonal conflict. Patients have feelings of shame and self-consciousness that hinder them from engaging in social activities and seeking out relationships.31 Twenty-nine percent of patients feel that psoriasis has interfered with establishing relationships because of negative self-esteem associated with the disease,32 and 26.3% have experienced people avoiding physical contact.33 Family and spouses of patients with psoriasis may be secondarily affected due to economic and emotional distress. Ninety-eight percent of family members of psoriatic patients experienced emotional distress and 54% experienced the burden of care.34 In a survey of 63 relatives and partners of patients with psoriasis, 57% experienced psychological distress, including anxiety and worry over a psoriatic patient’s future.35
Interpersonal Relationships Assessment
Current available tools, including the DLQI and short form health survey, measure overall QOL, including social functioning, but may not be practical in a clinic setting. Although no quick-screening test to assess for this domain exists, providers are encouraged to ask patients about disease impact on interpersonal relationships. The family DLQI questionnaire, adapted from the DLQI, may help physicians and social workers evaluate the burden on a patient’s family members.34
Interpersonal Relationships Management
It may be difficult for providers to address problems with interpersonal relationships without accessible tools. Patients may not be accompanied by family or friends during appointments, and it is difficult to screen for these issues during visits. Providers may offer resources such as the NPF website, which provides information about support groups. It also provides tips on dating and connecting to others in the community who share similar experiences.18 Encouraging patients to seek family or couples therapy also may be beneficial. Increased social support can lead to better QOL and fewer depressive symptoms.36
Sexual Functioning Prevalence
Psoriasis affects both physical and psychological components of sexual function. Among 3485 patients with skin conditions who were surveyed about sexual function, 34% of psoriatic patients reported that psoriasis interfered with sexual functioning at least to a certain degree.37 Sexual impairment was strongly associated with depression, anxiety, and suicidal ideation; 24% of depressed patients and 20% of anxious patients experienced sexual problems a lot or very much, based on the DLQI.37 Depending on the questionnaire used, the prevalence of sexual dysfunction due to psoriasis ranged from 35.5% to 71.3%.38 In an observational cohort study of 158 participants (n=79 psoriasis patients and n=79 controls), 34.2% of patients with psoriasis experienced erectile dysfunction compared to 17.7% of controls.39 Forty-two percent of psoriatic patients with genital involvement reported dyspareunia, 32% reported worsening of genital psoriasis after intercourse, and 43% reported decreased frequency of intercourse.40
Sexual Functioning Assessment
The Skindex-29, DLQI, and psoriasis disability index are available QOL tools that include one question evaluating difficulties with sexual function. The
Sexual Functioning Management
Better disease control leads to improved sexual function, as patients experience fewer feelings of shame, anxiety, and depression, as well as improvement of physical symptoms that can interfere with sexual functioning.38,43,44 Reducing friction, warmth, and moisture, as well as avoiding tight clothing, can help those with genital psoriasis. Patients are advised to reapply topical medications after sexual intercourse. Patients also can apply makeup to disguise psoriasis and help reduce feelings of self-consciousness that can impede sexual intimacy.18
Comment
The psychosocial burden of psoriasis penetrates many facets of patient lives. Psoriasis can invoke feelings of shame and embarrassment that are worsened by the public’s misconceptions about psoriasis, resulting in serious mental health issues that can cause even greater disability. Depression and anxiety are prevalent in patients with psoriasis. The characteristic symptoms of pain and pruritus along with psychiatric comorbidities can have an underestimated impact on daily activities, including employment, interpersonal relationships, and sexual function. Such dysfunctions have serious implications toward wages, professional advancement, social support, and overall QOL.
Dermatology providers play an important role in screening for these problems through validated questionnaires and identifying risks. Simple screening questions such as the PHQ-9 can be beneficial and feasible during dermatology visits. Screening for PsA can help patients avoid problems at work. Sexual dysfunction is a sensitive topic; however, providers can use a 1-question screen from valid questionnaires and inquire about the location of lesions as opportunities to address this issue.
Interventions lead to better disease control, which concurrently improves overall QOL. These interventions depend on both patient adherence and a physician’s commitment to finding an optimal treatment regimen for each individual. Medical management; coordinating care; developing treatment plans with psychiatry, rheumatology, and primary care providers; and psychological counseling and services may be necessary and beneficial (Table). Offering accessible resources such as the NPF website helps patients access information outside the clinic when it is not feasible to address all these concerns in a single visit. Psoriasis requires more than just medical management; it requires dermatology providers to use a multidisciplinary approach to address the psychosocial aspects of the disease.

Conclusion
The psychosocial burden of psoriasis is immense. Stigma, public misconception, mental health concerns, and occupational and interpersonal difficulty are the basis of disease burden. Providers play a vital role in assessing the effect psoriasis has on different areas of patients’ lives and providing appropriate interventions and resources to reduce disease burden.
The psychosocial impact of psoriasis is a critical component of disease burden. Psoriatic patients have high rates of depression and anxiety, problems at work, and difficulties with interpersonal relationships and intimacy.1 A National Psoriasis Foundation (NPF) survey from 2003 to 2011 reported that psoriasis affects overall emotional well-being in 88% of patients and enjoyment of life in 82% of patients.2
The reasons for psychosocial burden stem from public misconceptions and disease stigma. A survey of 1005 individuals (age range, 16–64 years) about their perceptions of psoriasis revealed that 16.5% believed that psoriasis is contagious and 6.8% believed that psoriasis is related to personal hygiene.3 Fifty percent practiced discriminatory behavior toward psoriatic patients, including reluctance to shake hands (28.8%) and engage in sexual relations/intercourse (44.1%). Sixty-five percent of psoriatic patients felt their appearance is unsightly, and 73% felt self-conscious about having psoriasis.2
The psychosocial burden exists despite medical treatment of the disease. In a cross-sectional study of 1184 psoriatic patients, 70.2% had impaired quality of life (QOL) as measured by the dermatology life quality index (DLQI), even after receiving a 4-week treatment for psoriasis.4 Medical treatment of psoriasis is not enough; providers need to assess overall QOL and provide treatment and resources for these patients in addition to symptomatic management.
There have been many studies on the psychosocial burden of psoriasis, but few have focused on a dermatology resident’s role in addressing this issue. This article will review psychosocial domains—psychiatric comorbidities and social functioning including occupational functioning, interpersonal relationships, and sexual functioning— and discuss a dermatology resident’s role in assessing and addressing each of these areas.
Methods
A PubMed search of articles indexed for MEDLINE was conducted using the following terms: psoriasis, depression, anxiety, work productivity, sexual functioning, and interpersonal relationships. Selected articles covered prevalence, assessment, and management of each psychosocial domain.
Results
Psychiatric Comorbidities
Prevalence
A high prevalence of psychiatric comorbidities exists in psoriatic patients. In a study of 469,097 patients with psoriasis, depression was the third most prevalent comorbidity (17.91%), following hyperlipidemia (45.64%) and hypertension (42.19%).5 In a 10-year longitudinal, population-based, prospective cohort study, antidepressant prescriptions were twice as frequent in psoriatic patients (17.8%) compared to control (7.9%)(P<.001).6 In a meta-analysis of 98 studies investigating psoriatic patients and psychiatric comorbidities, patients with psoriasis were 1.5 times more likely to experience depression (odds ratio [OR]: 1.57; 95% CI, 1.40-1.76) and use antidepressants (OR: 4.24; 95% CI, 1.53-11.76) compared to control.7 Patients with psoriasis were more likely to attempt suicide (OR: 1.32; 95% CI, 1.14-1.54) and complete suicide (OR: 1.20; 95% CI, 1.04-1.39) compared to people without psoriasis.8 A 1-year cross-sectional study of 90 psoriatic patients reported 78.7% were diagnosed with depression and 76.7% were diagnosed with anxiety. Seventy-two percent reported both anxiety and depression, correlating with worse QOL (χ2=26.7; P<.05).9
Assessment
Psychiatric comorbidities are assessed using clinical judgment and formal screening questionnaires in research studies. Signs of depression in patients with psoriasis can manifest as poor treatment adherence and recurrent flares of psoriasis.10,11 Psoriatic patients with psychiatric comorbidities were less likely to be adherent to treatment (risk ratio: 0.35; P<.003).10 The patient health questionnaire (PHQ) 9 and generalized anxiety disorder scale (GAD) 7 are validated and reliable questionnaires. The first 2 questions in PHQ-9 and GAD-7 screen for depression and anxiety, respectively.12-14 These 2-question screens are practical in a fast-paced dermatology outpatient setting. Systematic questionnaires specifically targeting mood disorders may be more beneficial than the widely used DLQI, which may not adequately capture mood disorders. Over the course of 10 months, 607 patients with psoriasis were asked to fill out the PHQ-9, GAD-7, and DLQI. Thirty-eight percent of patients with major depressive disorder had a DLQI score lower than 10, while 46% of patients with generalized anxiety disorder had a DLQI score lower than 10.15 Other questionnaires, including the hospital anxiety and depression scale and Beck depression inventory, are valid instruments with high sensitivity but are commonly used for research purposes and may not be clinically feasible.16
Management
Dermatologists should refer patients with depression and/or anxiety to psychiatry. Interventions include pharmacologic and nonpharmacologic management. First-line therapy for depression and anxiety is a combination of selective serotonin reuptake inhibitors and cognitive behavioral therapy.17 In addition, providers can direct patients to online resources such as the NPF website, where patients with psoriasis can access information about the signs and symptoms of mood disorders and contact the patient navigation center for further help.18
Social Functioning
Occupational Prevalence
The NPF found that 92% of patients with psoriasis or psoriatic arthritis (PsA) surveyed between 2003 and 2011 cited their psoriasis as reason for unemployment.2 In a survey of 43 patients asked about social and occupational functioning using the social and occupational assessment scale, 62.5% of psoriatic patients reported distress at work and 51.1% reported decreased efficiency at work.19 A national online survey that was conducted in France and issued to patients with and without psoriasis assessed overall QOL and work productivity using the work productivity and activity impairment questionnaire for psoriasis (WPAI-PSO). Of 714 patients with psoriasis and PsA, the latter had a 57.6% decrease in work productivity over 7 days compared to 27.9% in controls (P<.05).20 Occupational impairment leads to lost wages and hinders advancement, further exacerbating the psychosocial burden of psoriasis.21
Occupational Assessment
Formal assessment of occupational function can be done with the WPAI-PSO, a 6-question valid instrument.22 Providers may look for risk factors associated with greater loss in work productivity to help identify and offer support for patients. Patients with increased severity of itching, pain, and scaling experienced a greater decrease in work productivity.21,23 Patients with PsA warrant early detection and treatment because they experience greater physical restraints that can interfere with work activities. Of the 459 psoriatic patients without a prior diagnosis of PsA who filled out the PsA screening and evaluation questionnaire, 144 (31.4%) received a score of 44 or higher and were referred to rheumatology for further evaluation with the classification criteria for PsA. Nine percent of patients failed to be screened and remained undiagnosed with PsA.24 In a study using the health assessment questionnaire to assess 400 patients with PsA, those with worse physical function due to joint pain and stiffness were less likely to remain employed (OR: 0.56; P=.02).25
Occupational Management
Identifying and coordinating symptoms of PsA between dermatology and rheumatology is beneficial for patients who experience debilitating symptoms. There are a variety of treatments available for PsA. According to the European League Against Rheumatism 2015 guidelines developed from expert opinion and systematic reviews for PsA management, there are 4 phases of treatment, with reassessment every 3 to 6 months for effectiveness of therapy.26,27 Phase I involves initiating nonsteroidal anti-inflammatory drugs with or without glucocorticoid injections. Phase II involves synthetic disease-modifying drugs, including methotrexate, leflunomide, sulfasalazine, or cyclosporine. Phase III involves adding a second synthetic disease-modifying drug or starting a biologic, such as an anti–tumor necrosis factor, IL-12/IL-23, or IL-17 inhibitor. Phase IV involves switching to a different drug in either aforementioned class.26,27 Treatment with biologics improves work productivity as assessed by WPAI-PSO for psoriasis and PsA.28-30 Encouraging patients to speak up in the workplace and request small accommodations such as timely breaks or ergonomic chairs can help patients feel more comfortable and supported in the work environment.18 Patients who felt supported at work were more likely to remain employed.25
Interpersonal Relationships Prevalence
Misinformation about psoriasis, fear of rejection, and feelings of isolation may contribute to interpersonal conflict. Patients have feelings of shame and self-consciousness that hinder them from engaging in social activities and seeking out relationships.31 Twenty-nine percent of patients feel that psoriasis has interfered with establishing relationships because of negative self-esteem associated with the disease,32 and 26.3% have experienced people avoiding physical contact.33 Family and spouses of patients with psoriasis may be secondarily affected due to economic and emotional distress. Ninety-eight percent of family members of psoriatic patients experienced emotional distress and 54% experienced the burden of care.34 In a survey of 63 relatives and partners of patients with psoriasis, 57% experienced psychological distress, including anxiety and worry over a psoriatic patient’s future.35
Interpersonal Relationships Assessment
Current available tools, including the DLQI and short form health survey, measure overall QOL, including social functioning, but may not be practical in a clinic setting. Although no quick-screening test to assess for this domain exists, providers are encouraged to ask patients about disease impact on interpersonal relationships. The family DLQI questionnaire, adapted from the DLQI, may help physicians and social workers evaluate the burden on a patient’s family members.34
Interpersonal Relationships Management
It may be difficult for providers to address problems with interpersonal relationships without accessible tools. Patients may not be accompanied by family or friends during appointments, and it is difficult to screen for these issues during visits. Providers may offer resources such as the NPF website, which provides information about support groups. It also provides tips on dating and connecting to others in the community who share similar experiences.18 Encouraging patients to seek family or couples therapy also may be beneficial. Increased social support can lead to better QOL and fewer depressive symptoms.36
Sexual Functioning Prevalence
Psoriasis affects both physical and psychological components of sexual function. Among 3485 patients with skin conditions who were surveyed about sexual function, 34% of psoriatic patients reported that psoriasis interfered with sexual functioning at least to a certain degree.37 Sexual impairment was strongly associated with depression, anxiety, and suicidal ideation; 24% of depressed patients and 20% of anxious patients experienced sexual problems a lot or very much, based on the DLQI.37 Depending on the questionnaire used, the prevalence of sexual dysfunction due to psoriasis ranged from 35.5% to 71.3%.38 In an observational cohort study of 158 participants (n=79 psoriasis patients and n=79 controls), 34.2% of patients with psoriasis experienced erectile dysfunction compared to 17.7% of controls.39 Forty-two percent of psoriatic patients with genital involvement reported dyspareunia, 32% reported worsening of genital psoriasis after intercourse, and 43% reported decreased frequency of intercourse.40
Sexual Functioning Assessment
The Skindex-29, DLQI, and psoriasis disability index are available QOL tools that include one question evaluating difficulties with sexual function. The
Sexual Functioning Management
Better disease control leads to improved sexual function, as patients experience fewer feelings of shame, anxiety, and depression, as well as improvement of physical symptoms that can interfere with sexual functioning.38,43,44 Reducing friction, warmth, and moisture, as well as avoiding tight clothing, can help those with genital psoriasis. Patients are advised to reapply topical medications after sexual intercourse. Patients also can apply makeup to disguise psoriasis and help reduce feelings of self-consciousness that can impede sexual intimacy.18
Comment
The psychosocial burden of psoriasis penetrates many facets of patient lives. Psoriasis can invoke feelings of shame and embarrassment that are worsened by the public’s misconceptions about psoriasis, resulting in serious mental health issues that can cause even greater disability. Depression and anxiety are prevalent in patients with psoriasis. The characteristic symptoms of pain and pruritus along with psychiatric comorbidities can have an underestimated impact on daily activities, including employment, interpersonal relationships, and sexual function. Such dysfunctions have serious implications toward wages, professional advancement, social support, and overall QOL.
Dermatology providers play an important role in screening for these problems through validated questionnaires and identifying risks. Simple screening questions such as the PHQ-9 can be beneficial and feasible during dermatology visits. Screening for PsA can help patients avoid problems at work. Sexual dysfunction is a sensitive topic; however, providers can use a 1-question screen from valid questionnaires and inquire about the location of lesions as opportunities to address this issue.
Interventions lead to better disease control, which concurrently improves overall QOL. These interventions depend on both patient adherence and a physician’s commitment to finding an optimal treatment regimen for each individual. Medical management; coordinating care; developing treatment plans with psychiatry, rheumatology, and primary care providers; and psychological counseling and services may be necessary and beneficial (Table). Offering accessible resources such as the NPF website helps patients access information outside the clinic when it is not feasible to address all these concerns in a single visit. Psoriasis requires more than just medical management; it requires dermatology providers to use a multidisciplinary approach to address the psychosocial aspects of the disease.

Conclusion
The psychosocial burden of psoriasis is immense. Stigma, public misconception, mental health concerns, and occupational and interpersonal difficulty are the basis of disease burden. Providers play a vital role in assessing the effect psoriasis has on different areas of patients’ lives and providing appropriate interventions and resources to reduce disease burden.
- Kimball AB, Jacobson C, Weiss S, et al. The psychosocial burden of psoriasis. Am J Clin Dermatol. 2005;6:383-392.
- Armstrong AW, Schupp C, Wu J, et al. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003-2011. PloS One. 2012;7:e52935.
- Halioua B, Sid-Mohand D, Roussel ME, et al. Extent of misconceptions, negative prejudices and discriminatory behaviour to psoriasis patients in France. J Eur Acad Dermatol Venereol. 2016;30:650-654.
- Wolf P, Weger W, Legat F, et al. Quality of life and treatment goals in psoriasis from the patient perspective: results of an Austrian cross-sectional survey. J Dtsch Dermatol Ges. 2018;16:981-990.
- Shah K, Mellars L, Changolkar A, et al. Real-world burden of comorbidities in US patients with psoriasis. J Am Acad Dermatol. 2017;77:287-292.e4.
- Dowlatshahi EA, Wakkee M, Herings RM, et al. Increased antidepressant drug exposure in psoriasis patients: a longitudinal population-based cohort study. Acta Derm Venereol. 2013;93:544-550.
- Dowlatshahi EA, Wakkee M, Arends LR, et al. The prevalence and odds of depressive symptoms and clinical depression in psoriasis patients: a systematic review and meta-analysis. J Invest Dermatol. 2014;134:1542-1551.
- Singh S, Taylor C, Kornmehl H, et al. Psoriasis and suicidality: a systematic review and meta-analysis. J Am Acad Dermatol. 2017;77:425.e2-440.e2.
- Lakshmy S, Balasundaram S, Sarkar S, et al. A cross-sectional study of prevalence and implications of depression and anxiety in psoriasis. Indian J Psychol Med. 2015;37:434-440.
- Renzi C, Picardi A, Abeni D, et al. Association of dissatisfaction with care and psychiatric morbidity with poor treatment compliance. Arch Dermatol. 2002;138:337-342.
- Kulkarni AS, Balkrishnan R, Camacho FT, et al. Medication and health care service utilization related to depressive symptoms in older adults with psoriasis. J Drugs Dermatol. 2004;3:661-666.
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613.
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097.
- Kroenke K, Spitzer RL, Williams JB. The Patient Health Questionnaire-2: validity of a two-item depression screener. Med Care. 2003;41:1284-1292.
- Lamb RC, Matcham F, Turner MA, et al. Screening for anxiety and depression in people with psoriasis: a cross-sectional study in a tertiary referral setting. Br J Dermatol. 2017;176:1028-1034.
- Law M, Naughton MT, Dhar A, et al. Validation of two depression screening instruments in a sleep disorders clinic. J Clin Sleep Med. 2014;10:683-688.
- Cuijpers P, Dekker J, Hollon SD, et al. Adding psychotherapy to pharmacotherapy in the treatment of depressive disorders in adults: a meta-analysis. J Clin Psychiatry. 2009;70:1219-1229.
- National Psoriasis Foundation. Living with psoriatic arthritis. https://www.psoriasis.org/life-with-psoriatic-arthritis. Accessed September 23, 2018.
- Gaikwad R, Deshpande S, Raje S, et al. Evaluation of functional impairment in psoriasis. Indian J Dermatol Venereol Leprol. 2006;72:37-40.
- Claudepierre P, Lahfa M, Levy P, et al. The impact of psoriasis on professional life: PsoPRO, a French national survey [published online April 6, 2018]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.14986.
- Korman NJ, Zhao Y, Pike J, et al. Relationship between psoriasis severity, clinical symptoms, quality of life and work productivity among patients in the USA. Clin Exp Dermatol. 2016;41:514-521.
- Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. PharmacoEconomics. 1993;4:353-365.
- Korman NJ, Zhao Y, Pike J, et al. Increased severity of itching, pain, and scaling in psoriasis patients is associated with increased disease severity, reduced quality of life, and reduced work productivity. Dermatol Online J. 2015;21.
- Spelman L, Su JC, Fernandez-Penas P, et al. Frequency of undiagnosed psoriatic arthritis among psoriasis patients in Australian dermatology practice. J Eur Acad Dermatol Venereol. 2015;29:2184-2191.
- Tillett W, Shaddick G, Askari A, et al. Factors influencing work disability in psoriatic arthritis: first results from a large UK multicentre study. Rheumatology (Oxford). 2015;54:157-162.
- Raychaudhuri SP, Wilken R, Sukhov AC, et al. Management of psoriatic arthritis: early diagnosis, monitoring of disease severity and cutting edge therapies. J Autoimmun. 2017;76:21-37.
- Gossec L, Smolen JS, Ramiro S, et al. European League Against Rheumatism (EULAR) recommendations for the manegement of psoriatic arthritis with pharmacological therapies: 2015 update. Ann Rheum Dis. 2016;75:499-510.
- Beroukhim K, Danesh M, Nguyen C, et al. A prospective, interventional assessment of the impact of ustekinumab treatment on psoriasis-related work productivity and activity impairment. J Dermatol Treat. 2016;27:552-555.
- Armstrong AW, Lynde CW, McBride SR, et al. Effect of ixekizumab treatment on work productivity for patients with moderate-to-severe plaque psoriasis: analysis of results from 3 randomized phase 3 clinical trials. JAMA Dermatol. 2016;152:661-669.
- Kimball AB, Yu AP, Signorovitch J, et al. The effects of adalimumab treatment and psoriasis severity on self-reported work productivity and activity impairment for patients with moderate to severe psoriasis. J Am Acad Dermatol. 2012;66:e67-76.
- Feldman SR, Malakouti M, Koo JY. Social impact of the burden of psoriasis: effects on patients and practice. Dermatol Online J. 2014;20.
- Reich A, Welz-Kubiak K, Rams Ł. Apprehension of the disease by patients suffering from psoriasis. Postepy Dermatol Alergol. 2014;31:289-293.
- Gupta MA, Gupta AK, Watteel GN. Perceived deprivation of social touch in psoriasis is associated with greater psychologic morbidity: an index of the stigma experience in dermatologic disorders. Cutis. 1998;61:339-342.
- Basra MK, Finlay AY. The family impact of skin diseases: the Greater Patient concept. Br J Dermatol. 2007;156:929-937.
- Eghlileb AM, Davies EE, Finlay AY. Psoriasis has a major secondary impact on the lives of family members and partners. Br J Dermatol. 2007;156:1245-1250.
- Janowski K, Steuden S, Pietrzak A, et al. Social support and adaptation to the disease in men and women with psoriasis. Arch Dermatol Res. 2012;304:421-432.
- Sampogna F, Abeni D, Gieler U, et al. Impairment of sexual life in 3,485 dermatological outpatients from a multicentre study in 13 European countries. Acta Derm Venereol. 2017;97:478-482.
- Sampogna F, Gisondi P, Tabolli S, et al. Impairment of sexual life in patients with psoriasis. Dermatology. 2007;214:144-150.
- Molina-Leyva A, Molina-Leyva I, Almodovar-Real A, et al. Prevalence and associated factors of erectile dysfunction in patients with moderate to severe psoriasis and healthy population: a comparative study considering physical and psychological factors. Arch Sex Behav. 2016;45:2047-2055.
- Ryan C, Sadlier M, De Vol E, et al. Genital psoriasis is associated with significant impairment in quality of life and sexual functioning. J Am Acad Dermatol. 2015;72:978-983.
- Labbate LA, Lare SB. Sexual dysfunction in male psychiatric outpatients: validity of the Massachusetts General Hospital Sexual Functioning Questionnaire. Psychother Psychosom. 2001;70:221-225.
- Molina-Leyva A, Almodovar-Real A, Ruiz-Carrascosa JC, et al. Distribution pattern of psoriasis affects sexual function in moderate to severe psoriasis: a prospective case series study. J Sex Med. 2014;11:2882-2889.
- Guenther L, Han C, Szapary P, et al. Impact of ustekinumab on health-related quality of life and sexual difficulties associated with psoriasis: results from two phase III clinical trials. J Eur Acad Dermatol Venereol. 2011;25:851-857.
- Guenther L, Warren RB, Cather JC, et al. Impact of ixekizumab treatment on skin-related personal relationship difficulties in moderate-to-severe psoriasis patients: 12-week results from two Phase 3 trials. J Eur Acad Dermatol Venereol. 2017;31:1867-1875.
- Kimball AB, Jacobson C, Weiss S, et al. The psychosocial burden of psoriasis. Am J Clin Dermatol. 2005;6:383-392.
- Armstrong AW, Schupp C, Wu J, et al. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003-2011. PloS One. 2012;7:e52935.
- Halioua B, Sid-Mohand D, Roussel ME, et al. Extent of misconceptions, negative prejudices and discriminatory behaviour to psoriasis patients in France. J Eur Acad Dermatol Venereol. 2016;30:650-654.
- Wolf P, Weger W, Legat F, et al. Quality of life and treatment goals in psoriasis from the patient perspective: results of an Austrian cross-sectional survey. J Dtsch Dermatol Ges. 2018;16:981-990.
- Shah K, Mellars L, Changolkar A, et al. Real-world burden of comorbidities in US patients with psoriasis. J Am Acad Dermatol. 2017;77:287-292.e4.
- Dowlatshahi EA, Wakkee M, Herings RM, et al. Increased antidepressant drug exposure in psoriasis patients: a longitudinal population-based cohort study. Acta Derm Venereol. 2013;93:544-550.
- Dowlatshahi EA, Wakkee M, Arends LR, et al. The prevalence and odds of depressive symptoms and clinical depression in psoriasis patients: a systematic review and meta-analysis. J Invest Dermatol. 2014;134:1542-1551.
- Singh S, Taylor C, Kornmehl H, et al. Psoriasis and suicidality: a systematic review and meta-analysis. J Am Acad Dermatol. 2017;77:425.e2-440.e2.
- Lakshmy S, Balasundaram S, Sarkar S, et al. A cross-sectional study of prevalence and implications of depression and anxiety in psoriasis. Indian J Psychol Med. 2015;37:434-440.
- Renzi C, Picardi A, Abeni D, et al. Association of dissatisfaction with care and psychiatric morbidity with poor treatment compliance. Arch Dermatol. 2002;138:337-342.
- Kulkarni AS, Balkrishnan R, Camacho FT, et al. Medication and health care service utilization related to depressive symptoms in older adults with psoriasis. J Drugs Dermatol. 2004;3:661-666.
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613.
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097.
- Kroenke K, Spitzer RL, Williams JB. The Patient Health Questionnaire-2: validity of a two-item depression screener. Med Care. 2003;41:1284-1292.
- Lamb RC, Matcham F, Turner MA, et al. Screening for anxiety and depression in people with psoriasis: a cross-sectional study in a tertiary referral setting. Br J Dermatol. 2017;176:1028-1034.
- Law M, Naughton MT, Dhar A, et al. Validation of two depression screening instruments in a sleep disorders clinic. J Clin Sleep Med. 2014;10:683-688.
- Cuijpers P, Dekker J, Hollon SD, et al. Adding psychotherapy to pharmacotherapy in the treatment of depressive disorders in adults: a meta-analysis. J Clin Psychiatry. 2009;70:1219-1229.
- National Psoriasis Foundation. Living with psoriatic arthritis. https://www.psoriasis.org/life-with-psoriatic-arthritis. Accessed September 23, 2018.
- Gaikwad R, Deshpande S, Raje S, et al. Evaluation of functional impairment in psoriasis. Indian J Dermatol Venereol Leprol. 2006;72:37-40.
- Claudepierre P, Lahfa M, Levy P, et al. The impact of psoriasis on professional life: PsoPRO, a French national survey [published online April 6, 2018]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.14986.
- Korman NJ, Zhao Y, Pike J, et al. Relationship between psoriasis severity, clinical symptoms, quality of life and work productivity among patients in the USA. Clin Exp Dermatol. 2016;41:514-521.
- Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. PharmacoEconomics. 1993;4:353-365.
- Korman NJ, Zhao Y, Pike J, et al. Increased severity of itching, pain, and scaling in psoriasis patients is associated with increased disease severity, reduced quality of life, and reduced work productivity. Dermatol Online J. 2015;21.
- Spelman L, Su JC, Fernandez-Penas P, et al. Frequency of undiagnosed psoriatic arthritis among psoriasis patients in Australian dermatology practice. J Eur Acad Dermatol Venereol. 2015;29:2184-2191.
- Tillett W, Shaddick G, Askari A, et al. Factors influencing work disability in psoriatic arthritis: first results from a large UK multicentre study. Rheumatology (Oxford). 2015;54:157-162.
- Raychaudhuri SP, Wilken R, Sukhov AC, et al. Management of psoriatic arthritis: early diagnosis, monitoring of disease severity and cutting edge therapies. J Autoimmun. 2017;76:21-37.
- Gossec L, Smolen JS, Ramiro S, et al. European League Against Rheumatism (EULAR) recommendations for the manegement of psoriatic arthritis with pharmacological therapies: 2015 update. Ann Rheum Dis. 2016;75:499-510.
- Beroukhim K, Danesh M, Nguyen C, et al. A prospective, interventional assessment of the impact of ustekinumab treatment on psoriasis-related work productivity and activity impairment. J Dermatol Treat. 2016;27:552-555.
- Armstrong AW, Lynde CW, McBride SR, et al. Effect of ixekizumab treatment on work productivity for patients with moderate-to-severe plaque psoriasis: analysis of results from 3 randomized phase 3 clinical trials. JAMA Dermatol. 2016;152:661-669.
- Kimball AB, Yu AP, Signorovitch J, et al. The effects of adalimumab treatment and psoriasis severity on self-reported work productivity and activity impairment for patients with moderate to severe psoriasis. J Am Acad Dermatol. 2012;66:e67-76.
- Feldman SR, Malakouti M, Koo JY. Social impact of the burden of psoriasis: effects on patients and practice. Dermatol Online J. 2014;20.
- Reich A, Welz-Kubiak K, Rams Ł. Apprehension of the disease by patients suffering from psoriasis. Postepy Dermatol Alergol. 2014;31:289-293.
- Gupta MA, Gupta AK, Watteel GN. Perceived deprivation of social touch in psoriasis is associated with greater psychologic morbidity: an index of the stigma experience in dermatologic disorders. Cutis. 1998;61:339-342.
- Basra MK, Finlay AY. The family impact of skin diseases: the Greater Patient concept. Br J Dermatol. 2007;156:929-937.
- Eghlileb AM, Davies EE, Finlay AY. Psoriasis has a major secondary impact on the lives of family members and partners. Br J Dermatol. 2007;156:1245-1250.
- Janowski K, Steuden S, Pietrzak A, et al. Social support and adaptation to the disease in men and women with psoriasis. Arch Dermatol Res. 2012;304:421-432.
- Sampogna F, Abeni D, Gieler U, et al. Impairment of sexual life in 3,485 dermatological outpatients from a multicentre study in 13 European countries. Acta Derm Venereol. 2017;97:478-482.
- Sampogna F, Gisondi P, Tabolli S, et al. Impairment of sexual life in patients with psoriasis. Dermatology. 2007;214:144-150.
- Molina-Leyva A, Molina-Leyva I, Almodovar-Real A, et al. Prevalence and associated factors of erectile dysfunction in patients with moderate to severe psoriasis and healthy population: a comparative study considering physical and psychological factors. Arch Sex Behav. 2016;45:2047-2055.
- Ryan C, Sadlier M, De Vol E, et al. Genital psoriasis is associated with significant impairment in quality of life and sexual functioning. J Am Acad Dermatol. 2015;72:978-983.
- Labbate LA, Lare SB. Sexual dysfunction in male psychiatric outpatients: validity of the Massachusetts General Hospital Sexual Functioning Questionnaire. Psychother Psychosom. 2001;70:221-225.
- Molina-Leyva A, Almodovar-Real A, Ruiz-Carrascosa JC, et al. Distribution pattern of psoriasis affects sexual function in moderate to severe psoriasis: a prospective case series study. J Sex Med. 2014;11:2882-2889.
- Guenther L, Han C, Szapary P, et al. Impact of ustekinumab on health-related quality of life and sexual difficulties associated with psoriasis: results from two phase III clinical trials. J Eur Acad Dermatol Venereol. 2011;25:851-857.
- Guenther L, Warren RB, Cather JC, et al. Impact of ixekizumab treatment on skin-related personal relationship difficulties in moderate-to-severe psoriasis patients: 12-week results from two Phase 3 trials. J Eur Acad Dermatol Venereol. 2017;31:1867-1875.
Practice Points
- The psychosocial impact of psoriasis is an important component of the disease burden leading to reduced quality of life.
- Assessment of psychosocial dysfunction can be done through short questionnaires, asking patients directly about these issues and anticipating these problems in patients who are most vulnerable.
- Management of psychosocial impact ranges from pharmacological interventions to helpful resources such as the National Psoriasis Foundation website.






