User login
FDA grants approval to weekly growth hormone for adults
The human growth hormone formulation somapacitan for adults with growth hormone deficiency was approved by the Food and Drug Administration on Sept. 1. .
Somapacitan contains an albumin-binding element attached to the growth hormone, causing the reversible binding to albumin proteins in the body. This reduces clearance and increases the half-life of the hormone. The formulation has previous demonstrated safety and efficacy in children with growth hormone deficiency (J Clin Endocrinol Metab. 2020 Apr 1. doi: 10.1210/clinem/dgz310).
Growth hormone treatment can counter abdominal obesity, reduced lean body mass, fatigue, osteopenia, cardiovascular risks, and other manifestations of growth hormone deficiency in adults, but daily injections can be burdensome for patients. That makes long-acting versions attractive, but the lifelong nature of the treatment makes it important to characterize safety and tolerability.
The approval comes on the strength of a randomized, placebo-controlled phase 3 trial (REAL 1) of 300 adult patients in 17 countries with growth hormone deficiency (J Clin Endocrinol Metab. 2020 Apr 1. doi: 10.1210/clinem/dgaa049). Participants had either never received growth hormone treatment, or had stopped taking one at least 6 months before starting the trial. Subjects received once-weekly somapacitan, once-weekly placebo, or daily somatropin, which is FDA approved.
The primary endpoint was percentage change of truncal fat, which is regulated by growth hormone, and can lead to medical problems. After 34 weeks, subjects in the somapacitan group experienced a 1.06% decrease in truncal fat, compared with a 0.47% increase in the placebo group (P = .009) and a 2.23% decrease in the daily somatropin group.
After 34 weeks, a 52-week extension trial began. The somapacitan group continued on the drug and the placebo group was offered somapacitan. Patients on daily somatropin were randomized to continue daily treatment with somatropin or to switch to somapacitan.
At the end of the extension trial, those taking somapacitan for the full 86-week duration had an average reduction of 1.52% in truncal fat. After 86 weeks, the somapacitan and daily somatropin groups had similar values for percentage change in visceral fat, lean body mass, or appendicular skeletal muscle mass.
Common side effects of somapacitan were back pain, joint paint, indigestion, a sleep disorder, dizziness, tonsillitis, swelling in the arms or lower legs, vomiting, adrenal insufficiency, hypertension, increase in blood creatine phosphokinase, weight increase, and anemia.
Somapacitan, marketed as Sogroya by Novo Nordisk, is contraindicated in patients with an allergy to the drug, as well as those with an active malignancy, diabetic eye disease where increases in blood sugars could lead to retinal damage, acute critical illness, or acute respiratory failure.
The FDA recommends that providers perform an eye examination before drug initiation, as well as periodically while the patient is taking the drug, to rule out preexisting papilledema. This could be a sign of intracranial hypertension, which could be caused or worsened by growth hormones.
The human growth hormone formulation somapacitan for adults with growth hormone deficiency was approved by the Food and Drug Administration on Sept. 1. .
Somapacitan contains an albumin-binding element attached to the growth hormone, causing the reversible binding to albumin proteins in the body. This reduces clearance and increases the half-life of the hormone. The formulation has previous demonstrated safety and efficacy in children with growth hormone deficiency (J Clin Endocrinol Metab. 2020 Apr 1. doi: 10.1210/clinem/dgz310).
Growth hormone treatment can counter abdominal obesity, reduced lean body mass, fatigue, osteopenia, cardiovascular risks, and other manifestations of growth hormone deficiency in adults, but daily injections can be burdensome for patients. That makes long-acting versions attractive, but the lifelong nature of the treatment makes it important to characterize safety and tolerability.
The approval comes on the strength of a randomized, placebo-controlled phase 3 trial (REAL 1) of 300 adult patients in 17 countries with growth hormone deficiency (J Clin Endocrinol Metab. 2020 Apr 1. doi: 10.1210/clinem/dgaa049). Participants had either never received growth hormone treatment, or had stopped taking one at least 6 months before starting the trial. Subjects received once-weekly somapacitan, once-weekly placebo, or daily somatropin, which is FDA approved.
The primary endpoint was percentage change of truncal fat, which is regulated by growth hormone, and can lead to medical problems. After 34 weeks, subjects in the somapacitan group experienced a 1.06% decrease in truncal fat, compared with a 0.47% increase in the placebo group (P = .009) and a 2.23% decrease in the daily somatropin group.
After 34 weeks, a 52-week extension trial began. The somapacitan group continued on the drug and the placebo group was offered somapacitan. Patients on daily somatropin were randomized to continue daily treatment with somatropin or to switch to somapacitan.
At the end of the extension trial, those taking somapacitan for the full 86-week duration had an average reduction of 1.52% in truncal fat. After 86 weeks, the somapacitan and daily somatropin groups had similar values for percentage change in visceral fat, lean body mass, or appendicular skeletal muscle mass.
Common side effects of somapacitan were back pain, joint paint, indigestion, a sleep disorder, dizziness, tonsillitis, swelling in the arms or lower legs, vomiting, adrenal insufficiency, hypertension, increase in blood creatine phosphokinase, weight increase, and anemia.
Somapacitan, marketed as Sogroya by Novo Nordisk, is contraindicated in patients with an allergy to the drug, as well as those with an active malignancy, diabetic eye disease where increases in blood sugars could lead to retinal damage, acute critical illness, or acute respiratory failure.
The FDA recommends that providers perform an eye examination before drug initiation, as well as periodically while the patient is taking the drug, to rule out preexisting papilledema. This could be a sign of intracranial hypertension, which could be caused or worsened by growth hormones.
The human growth hormone formulation somapacitan for adults with growth hormone deficiency was approved by the Food and Drug Administration on Sept. 1. .
Somapacitan contains an albumin-binding element attached to the growth hormone, causing the reversible binding to albumin proteins in the body. This reduces clearance and increases the half-life of the hormone. The formulation has previous demonstrated safety and efficacy in children with growth hormone deficiency (J Clin Endocrinol Metab. 2020 Apr 1. doi: 10.1210/clinem/dgz310).
Growth hormone treatment can counter abdominal obesity, reduced lean body mass, fatigue, osteopenia, cardiovascular risks, and other manifestations of growth hormone deficiency in adults, but daily injections can be burdensome for patients. That makes long-acting versions attractive, but the lifelong nature of the treatment makes it important to characterize safety and tolerability.
The approval comes on the strength of a randomized, placebo-controlled phase 3 trial (REAL 1) of 300 adult patients in 17 countries with growth hormone deficiency (J Clin Endocrinol Metab. 2020 Apr 1. doi: 10.1210/clinem/dgaa049). Participants had either never received growth hormone treatment, or had stopped taking one at least 6 months before starting the trial. Subjects received once-weekly somapacitan, once-weekly placebo, or daily somatropin, which is FDA approved.
The primary endpoint was percentage change of truncal fat, which is regulated by growth hormone, and can lead to medical problems. After 34 weeks, subjects in the somapacitan group experienced a 1.06% decrease in truncal fat, compared with a 0.47% increase in the placebo group (P = .009) and a 2.23% decrease in the daily somatropin group.
After 34 weeks, a 52-week extension trial began. The somapacitan group continued on the drug and the placebo group was offered somapacitan. Patients on daily somatropin were randomized to continue daily treatment with somatropin or to switch to somapacitan.
At the end of the extension trial, those taking somapacitan for the full 86-week duration had an average reduction of 1.52% in truncal fat. After 86 weeks, the somapacitan and daily somatropin groups had similar values for percentage change in visceral fat, lean body mass, or appendicular skeletal muscle mass.
Common side effects of somapacitan were back pain, joint paint, indigestion, a sleep disorder, dizziness, tonsillitis, swelling in the arms or lower legs, vomiting, adrenal insufficiency, hypertension, increase in blood creatine phosphokinase, weight increase, and anemia.
Somapacitan, marketed as Sogroya by Novo Nordisk, is contraindicated in patients with an allergy to the drug, as well as those with an active malignancy, diabetic eye disease where increases in blood sugars could lead to retinal damage, acute critical illness, or acute respiratory failure.
The FDA recommends that providers perform an eye examination before drug initiation, as well as periodically while the patient is taking the drug, to rule out preexisting papilledema. This could be a sign of intracranial hypertension, which could be caused or worsened by growth hormones.
Humira topped drug-revenue list for 2019
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.
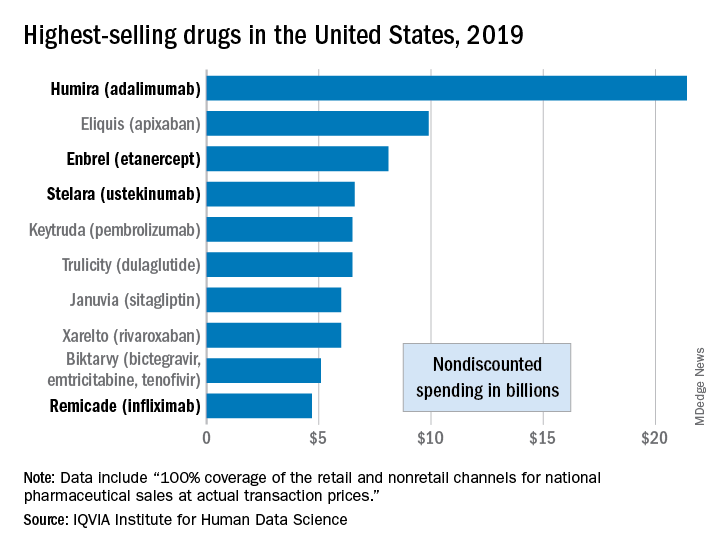
Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.

Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.

Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
Evidence mounts for COVID-19 effects on thyroid gland
Rates of thyrotoxicosis are significantly higher among patients who are critically ill with COVID-19 than among patients who are critically ill but who do not not have COVID-19, suggesting an atypical form of thyroiditis related to the novel coronavirus infection, according to new research.
“We suggest routine assessment of thyroid function in patients with COVID-19 requiring high-intensity care because they frequently present with thyrotoxicosis due to a form of subacute thyroiditis related to SARS-CoV-2,” the authors wrote in correspondence published online in The Lancet Diabetes and Endocrinology.
However, notably, the study – which compared critically ill ICU patients who had COVID-19 with those who did not have COVID-19 or who had milder cases of COVID-19 – indicates that thyroid disorders do not appear to increase the risk of developing COVID-19, first author Ilaria Muller, MD, PhD, of the department of endocrinology, IRCCS Fondazione Ca’ Granda Ospedale Maggiore Policlinico, Milan, said in an interview.
“It is important to highlight that we did not find an increased prevalence of preexisting thyroid disorders in COVID-19 patients (contrary to early media reports),” she said. “So far, clinical observations do not support this fear, and we need to reassure people with thyroid disorders, since such disorders are very common among the general population.”
Yet the findings add to emerging evidence of a COVID-19/thyroid relationship, Angela M. Leung, MD, said in an interview.
“Given the health care impacts of the current COVID-19 pandemic worldwide, this study provides some insight on the potential systemic inflammation, as well as thyroid-specific inflammation, of the SARS-Cov-2 virus that is described in some emerging reports,” she said.
“This study joins at least six others that have reported a clinical presentation resembling subacute thyroiditis in critically ill patients with COVID-19,” noted Dr. Leung, of the division of endocrinology, diabetes, and metabolism in the department of medicine at the University of California, Los Angeles.
Thyroid function analysis in those with severe COVID-19
Dr. Muller explained that preliminary data from her institution showed thyroid abnormalities in patients who were severely ill with COVID-19. She and her team extended the evaluation to include thyroid data and other data on 93 patients with COVID-19 who were admitted to high-intensity care units (HICUs) in Italy during the 2020 pandemic.
Those data were compared with data on 101 critically ill patients admitted to the same HICUs in 2019 who did not have COVID-19. A third group of 52 patients with COVID-19 who were admitted to low-intensity care units (LICUs) in Italy in 2020 were also included in the analysis.
The mean age of the patients in the HICU 2020 group was 65.3 years; in the HICU 2019 group, it was 73 years; and in the LICU group, it was 70 years (P = .001). In addition, the HICU 2020 group included more men than the other two groups (69% vs. 56% and 48%; P = .03).
Of note, only 9% of patients in the HICU 2020 group had preexisting thyroid disorders, compared with 21% in the LICU group and 23% in the HICU 2019 group (P = .017).
These findings suggest that “such conditions are not a risk factor for SARS-CoV-2 infection or severity of COVID-19,” the authors wrote.
The patients with the preexisting thyroid conditions were excluded from the thyroid function analysis.
A significantly higher proportion of patients in the HICU 2020 group (13; 15%) were thyrotoxic upon admission, compared with just 1 (1%) of 78 patients in the HICU 2019 group (P = .002) and one (2%) of 41 patients in the LICU group (P = .025).
Among the 14 patients in the two COVID-19 groups who had thyrotoxicosis, the majority were male (9; 64%)
Among those in the HICU 2020 group, serum thyroid-stimulating hormone concentrations were lower than in either of the other two groups (P = .018), and serum free thyroxine (free T4) concentrations were higher than in the LICU group (P = .016) but not the HICU 2019 group.
Differences compared with other infection-related thyroiditis
Although thyrotoxicosis relating to subacute viral thyroiditis can result from a wide variety of viral infections, there are some key differences with COVID-19, Dr. Muller said.
“Thyroid dysfunction related to SARS-CoV-2 seems to be milder than that of classic subacute thyroiditis due to other viruses,” she explained. Furthermore, thyroid dysfunction associated with other viral infections is more common in women, whereas there were more male patients with the COVID-19–related atypical thyroiditis.
In addition, the thyroid effects developed early with COVID-19, whereas they usually emerge after the infections by other viruses.
Patients did not demonstrate the neck pain that is common with classic viral thyroiditis, and the thyroid abnormalities appear to correlate with the severity of COVID-19, whereas they are seen even in patients with mild symptoms when other viral infections are the cause.
In addition to the risk for subacute viral thyroiditis, critically ill patients in general are at risk of developing nonthyroidal illness syndrome, with alterations in thyroid function. However, thyroid hormone measures in the patients severely ill with COVID-19 were not consistent with that syndrome.
A subanalysis of eight HICU 2020 patients with thyroid dysfunction who were followed for 55 days after discharge showed that two experienced hyperthyroidism but likely not from COVID-19; in the remaining six, thyroid function normalized.
Muller speculated that, when ill with COVID-19, the patients likely had a combination of SARS-CoV-2–related atypical thyroiditis and nonthyroidal illness syndrome, known as T4 toxicosis.
Will there be any long-term effects?
Importantly, it remains unknown whether the novel coronavirus has longer-term effects on the thyroid, Dr. Muller said.
“We cannot predict what will be the long-lasting thyroid effects after COVID-19,” she said.
With classic subacute viral thyroiditis, “After a few years ... 5%-20% of patients develop permanent hypothyroidism, [and] the same might happen in COVID-19 patients,” she hypothesized. “We will follow our patients long term to answer this question – this study is already ongoing.”
In the meantime, diagnosis of thyroid dysfunction in patients with COVID-19 is important, inasmuch as it could worsen the already critical conditions of patients, Muller stressed.
“The gold-standard treatment for thyroiditis is steroids, so the presence of thyroid dysfunction might represent an additional indication to such treatment in COVID-19 patients, to be verified in properly designed clinical trials,” she advised.
ACE2 cell receptors highly expressed in thyroid
Dr. Muller and colleagues also noted recent research showing that ACE2 – demonstrated to be a key host-cell entry receptor for both SARS-CoV and SARS-CoV-2 – is expressed in even higher levels in the thyroid than the lungs, where it causes COVID-19’s notorious pulmonary effects.
Dr. Muller said the implications of ACE2 expression in the thyroid remain to be elucidated.
“If ACE2 is confirmed to be expressed at higher levels, compared with the lungs in the thyroid gland and other tissues, i.e., small intestine, testis, kidney, heart, etc, dedicated studies will be needed to correlate ACE2 expression with the organs’ susceptibility to SARS-CoV-2 reflected by clinical presentation,” she said.
Dr. Leung added that, as a take-home message from these and the other thyroid/COVID-19 studies, “data are starting to show us that COVID-19 infection may cause thyrotoxicosis that is possibly related to thyroid and systemic inflammation. However, the serum thyroid function test abnormalities seen in COVID-19 patients with subacute thyroiditis are also likely exacerbated to a substantial extent by nonthyroidal illness physiology.”
The authors have disclosed no relevant financial relationships. Dr. Leung is on the advisory board of Medscape Diabetes and Endocrinology.
A version of this article originally appeared on Medscape.com.
Rates of thyrotoxicosis are significantly higher among patients who are critically ill with COVID-19 than among patients who are critically ill but who do not not have COVID-19, suggesting an atypical form of thyroiditis related to the novel coronavirus infection, according to new research.
“We suggest routine assessment of thyroid function in patients with COVID-19 requiring high-intensity care because they frequently present with thyrotoxicosis due to a form of subacute thyroiditis related to SARS-CoV-2,” the authors wrote in correspondence published online in The Lancet Diabetes and Endocrinology.
However, notably, the study – which compared critically ill ICU patients who had COVID-19 with those who did not have COVID-19 or who had milder cases of COVID-19 – indicates that thyroid disorders do not appear to increase the risk of developing COVID-19, first author Ilaria Muller, MD, PhD, of the department of endocrinology, IRCCS Fondazione Ca’ Granda Ospedale Maggiore Policlinico, Milan, said in an interview.
“It is important to highlight that we did not find an increased prevalence of preexisting thyroid disorders in COVID-19 patients (contrary to early media reports),” she said. “So far, clinical observations do not support this fear, and we need to reassure people with thyroid disorders, since such disorders are very common among the general population.”
Yet the findings add to emerging evidence of a COVID-19/thyroid relationship, Angela M. Leung, MD, said in an interview.
“Given the health care impacts of the current COVID-19 pandemic worldwide, this study provides some insight on the potential systemic inflammation, as well as thyroid-specific inflammation, of the SARS-Cov-2 virus that is described in some emerging reports,” she said.
“This study joins at least six others that have reported a clinical presentation resembling subacute thyroiditis in critically ill patients with COVID-19,” noted Dr. Leung, of the division of endocrinology, diabetes, and metabolism in the department of medicine at the University of California, Los Angeles.
Thyroid function analysis in those with severe COVID-19
Dr. Muller explained that preliminary data from her institution showed thyroid abnormalities in patients who were severely ill with COVID-19. She and her team extended the evaluation to include thyroid data and other data on 93 patients with COVID-19 who were admitted to high-intensity care units (HICUs) in Italy during the 2020 pandemic.
Those data were compared with data on 101 critically ill patients admitted to the same HICUs in 2019 who did not have COVID-19. A third group of 52 patients with COVID-19 who were admitted to low-intensity care units (LICUs) in Italy in 2020 were also included in the analysis.
The mean age of the patients in the HICU 2020 group was 65.3 years; in the HICU 2019 group, it was 73 years; and in the LICU group, it was 70 years (P = .001). In addition, the HICU 2020 group included more men than the other two groups (69% vs. 56% and 48%; P = .03).
Of note, only 9% of patients in the HICU 2020 group had preexisting thyroid disorders, compared with 21% in the LICU group and 23% in the HICU 2019 group (P = .017).
These findings suggest that “such conditions are not a risk factor for SARS-CoV-2 infection or severity of COVID-19,” the authors wrote.
The patients with the preexisting thyroid conditions were excluded from the thyroid function analysis.
A significantly higher proportion of patients in the HICU 2020 group (13; 15%) were thyrotoxic upon admission, compared with just 1 (1%) of 78 patients in the HICU 2019 group (P = .002) and one (2%) of 41 patients in the LICU group (P = .025).
Among the 14 patients in the two COVID-19 groups who had thyrotoxicosis, the majority were male (9; 64%)
Among those in the HICU 2020 group, serum thyroid-stimulating hormone concentrations were lower than in either of the other two groups (P = .018), and serum free thyroxine (free T4) concentrations were higher than in the LICU group (P = .016) but not the HICU 2019 group.
Differences compared with other infection-related thyroiditis
Although thyrotoxicosis relating to subacute viral thyroiditis can result from a wide variety of viral infections, there are some key differences with COVID-19, Dr. Muller said.
“Thyroid dysfunction related to SARS-CoV-2 seems to be milder than that of classic subacute thyroiditis due to other viruses,” she explained. Furthermore, thyroid dysfunction associated with other viral infections is more common in women, whereas there were more male patients with the COVID-19–related atypical thyroiditis.
In addition, the thyroid effects developed early with COVID-19, whereas they usually emerge after the infections by other viruses.
Patients did not demonstrate the neck pain that is common with classic viral thyroiditis, and the thyroid abnormalities appear to correlate with the severity of COVID-19, whereas they are seen even in patients with mild symptoms when other viral infections are the cause.
In addition to the risk for subacute viral thyroiditis, critically ill patients in general are at risk of developing nonthyroidal illness syndrome, with alterations in thyroid function. However, thyroid hormone measures in the patients severely ill with COVID-19 were not consistent with that syndrome.
A subanalysis of eight HICU 2020 patients with thyroid dysfunction who were followed for 55 days after discharge showed that two experienced hyperthyroidism but likely not from COVID-19; in the remaining six, thyroid function normalized.
Muller speculated that, when ill with COVID-19, the patients likely had a combination of SARS-CoV-2–related atypical thyroiditis and nonthyroidal illness syndrome, known as T4 toxicosis.
Will there be any long-term effects?
Importantly, it remains unknown whether the novel coronavirus has longer-term effects on the thyroid, Dr. Muller said.
“We cannot predict what will be the long-lasting thyroid effects after COVID-19,” she said.
With classic subacute viral thyroiditis, “After a few years ... 5%-20% of patients develop permanent hypothyroidism, [and] the same might happen in COVID-19 patients,” she hypothesized. “We will follow our patients long term to answer this question – this study is already ongoing.”
In the meantime, diagnosis of thyroid dysfunction in patients with COVID-19 is important, inasmuch as it could worsen the already critical conditions of patients, Muller stressed.
“The gold-standard treatment for thyroiditis is steroids, so the presence of thyroid dysfunction might represent an additional indication to such treatment in COVID-19 patients, to be verified in properly designed clinical trials,” she advised.
ACE2 cell receptors highly expressed in thyroid
Dr. Muller and colleagues also noted recent research showing that ACE2 – demonstrated to be a key host-cell entry receptor for both SARS-CoV and SARS-CoV-2 – is expressed in even higher levels in the thyroid than the lungs, where it causes COVID-19’s notorious pulmonary effects.
Dr. Muller said the implications of ACE2 expression in the thyroid remain to be elucidated.
“If ACE2 is confirmed to be expressed at higher levels, compared with the lungs in the thyroid gland and other tissues, i.e., small intestine, testis, kidney, heart, etc, dedicated studies will be needed to correlate ACE2 expression with the organs’ susceptibility to SARS-CoV-2 reflected by clinical presentation,” she said.
Dr. Leung added that, as a take-home message from these and the other thyroid/COVID-19 studies, “data are starting to show us that COVID-19 infection may cause thyrotoxicosis that is possibly related to thyroid and systemic inflammation. However, the serum thyroid function test abnormalities seen in COVID-19 patients with subacute thyroiditis are also likely exacerbated to a substantial extent by nonthyroidal illness physiology.”
The authors have disclosed no relevant financial relationships. Dr. Leung is on the advisory board of Medscape Diabetes and Endocrinology.
A version of this article originally appeared on Medscape.com.
Rates of thyrotoxicosis are significantly higher among patients who are critically ill with COVID-19 than among patients who are critically ill but who do not not have COVID-19, suggesting an atypical form of thyroiditis related to the novel coronavirus infection, according to new research.
“We suggest routine assessment of thyroid function in patients with COVID-19 requiring high-intensity care because they frequently present with thyrotoxicosis due to a form of subacute thyroiditis related to SARS-CoV-2,” the authors wrote in correspondence published online in The Lancet Diabetes and Endocrinology.
However, notably, the study – which compared critically ill ICU patients who had COVID-19 with those who did not have COVID-19 or who had milder cases of COVID-19 – indicates that thyroid disorders do not appear to increase the risk of developing COVID-19, first author Ilaria Muller, MD, PhD, of the department of endocrinology, IRCCS Fondazione Ca’ Granda Ospedale Maggiore Policlinico, Milan, said in an interview.
“It is important to highlight that we did not find an increased prevalence of preexisting thyroid disorders in COVID-19 patients (contrary to early media reports),” she said. “So far, clinical observations do not support this fear, and we need to reassure people with thyroid disorders, since such disorders are very common among the general population.”
Yet the findings add to emerging evidence of a COVID-19/thyroid relationship, Angela M. Leung, MD, said in an interview.
“Given the health care impacts of the current COVID-19 pandemic worldwide, this study provides some insight on the potential systemic inflammation, as well as thyroid-specific inflammation, of the SARS-Cov-2 virus that is described in some emerging reports,” she said.
“This study joins at least six others that have reported a clinical presentation resembling subacute thyroiditis in critically ill patients with COVID-19,” noted Dr. Leung, of the division of endocrinology, diabetes, and metabolism in the department of medicine at the University of California, Los Angeles.
Thyroid function analysis in those with severe COVID-19
Dr. Muller explained that preliminary data from her institution showed thyroid abnormalities in patients who were severely ill with COVID-19. She and her team extended the evaluation to include thyroid data and other data on 93 patients with COVID-19 who were admitted to high-intensity care units (HICUs) in Italy during the 2020 pandemic.
Those data were compared with data on 101 critically ill patients admitted to the same HICUs in 2019 who did not have COVID-19. A third group of 52 patients with COVID-19 who were admitted to low-intensity care units (LICUs) in Italy in 2020 were also included in the analysis.
The mean age of the patients in the HICU 2020 group was 65.3 years; in the HICU 2019 group, it was 73 years; and in the LICU group, it was 70 years (P = .001). In addition, the HICU 2020 group included more men than the other two groups (69% vs. 56% and 48%; P = .03).
Of note, only 9% of patients in the HICU 2020 group had preexisting thyroid disorders, compared with 21% in the LICU group and 23% in the HICU 2019 group (P = .017).
These findings suggest that “such conditions are not a risk factor for SARS-CoV-2 infection or severity of COVID-19,” the authors wrote.
The patients with the preexisting thyroid conditions were excluded from the thyroid function analysis.
A significantly higher proportion of patients in the HICU 2020 group (13; 15%) were thyrotoxic upon admission, compared with just 1 (1%) of 78 patients in the HICU 2019 group (P = .002) and one (2%) of 41 patients in the LICU group (P = .025).
Among the 14 patients in the two COVID-19 groups who had thyrotoxicosis, the majority were male (9; 64%)
Among those in the HICU 2020 group, serum thyroid-stimulating hormone concentrations were lower than in either of the other two groups (P = .018), and serum free thyroxine (free T4) concentrations were higher than in the LICU group (P = .016) but not the HICU 2019 group.
Differences compared with other infection-related thyroiditis
Although thyrotoxicosis relating to subacute viral thyroiditis can result from a wide variety of viral infections, there are some key differences with COVID-19, Dr. Muller said.
“Thyroid dysfunction related to SARS-CoV-2 seems to be milder than that of classic subacute thyroiditis due to other viruses,” she explained. Furthermore, thyroid dysfunction associated with other viral infections is more common in women, whereas there were more male patients with the COVID-19–related atypical thyroiditis.
In addition, the thyroid effects developed early with COVID-19, whereas they usually emerge after the infections by other viruses.
Patients did not demonstrate the neck pain that is common with classic viral thyroiditis, and the thyroid abnormalities appear to correlate with the severity of COVID-19, whereas they are seen even in patients with mild symptoms when other viral infections are the cause.
In addition to the risk for subacute viral thyroiditis, critically ill patients in general are at risk of developing nonthyroidal illness syndrome, with alterations in thyroid function. However, thyroid hormone measures in the patients severely ill with COVID-19 were not consistent with that syndrome.
A subanalysis of eight HICU 2020 patients with thyroid dysfunction who were followed for 55 days after discharge showed that two experienced hyperthyroidism but likely not from COVID-19; in the remaining six, thyroid function normalized.
Muller speculated that, when ill with COVID-19, the patients likely had a combination of SARS-CoV-2–related atypical thyroiditis and nonthyroidal illness syndrome, known as T4 toxicosis.
Will there be any long-term effects?
Importantly, it remains unknown whether the novel coronavirus has longer-term effects on the thyroid, Dr. Muller said.
“We cannot predict what will be the long-lasting thyroid effects after COVID-19,” she said.
With classic subacute viral thyroiditis, “After a few years ... 5%-20% of patients develop permanent hypothyroidism, [and] the same might happen in COVID-19 patients,” she hypothesized. “We will follow our patients long term to answer this question – this study is already ongoing.”
In the meantime, diagnosis of thyroid dysfunction in patients with COVID-19 is important, inasmuch as it could worsen the already critical conditions of patients, Muller stressed.
“The gold-standard treatment for thyroiditis is steroids, so the presence of thyroid dysfunction might represent an additional indication to such treatment in COVID-19 patients, to be verified in properly designed clinical trials,” she advised.
ACE2 cell receptors highly expressed in thyroid
Dr. Muller and colleagues also noted recent research showing that ACE2 – demonstrated to be a key host-cell entry receptor for both SARS-CoV and SARS-CoV-2 – is expressed in even higher levels in the thyroid than the lungs, where it causes COVID-19’s notorious pulmonary effects.
Dr. Muller said the implications of ACE2 expression in the thyroid remain to be elucidated.
“If ACE2 is confirmed to be expressed at higher levels, compared with the lungs in the thyroid gland and other tissues, i.e., small intestine, testis, kidney, heart, etc, dedicated studies will be needed to correlate ACE2 expression with the organs’ susceptibility to SARS-CoV-2 reflected by clinical presentation,” she said.
Dr. Leung added that, as a take-home message from these and the other thyroid/COVID-19 studies, “data are starting to show us that COVID-19 infection may cause thyrotoxicosis that is possibly related to thyroid and systemic inflammation. However, the serum thyroid function test abnormalities seen in COVID-19 patients with subacute thyroiditis are also likely exacerbated to a substantial extent by nonthyroidal illness physiology.”
The authors have disclosed no relevant financial relationships. Dr. Leung is on the advisory board of Medscape Diabetes and Endocrinology.
A version of this article originally appeared on Medscape.com.
Urine screen as part of triple test improves ID of adrenal cancer
A strategy that includes a urine steroid test along with imaging characteristics and tumor size criteria can significantly improve the challenging diagnosis of adrenocortical cancer, helping to avoid unnecessary, and often unsuccessful, further imaging and even surgery, new research shows.
“A triple-test strategy of tumor diameter, imaging characteristics, and urine steroid metabolomics improves detection of adrenocortical carcinoma, which could shorten time to surgery for patients with ... carcinoma and help to avoid unnecessary surgery in patients with benign tumors,” the authors say in research published online July 23 in The Lancet Diabetes & Endocrinology.
The triple-test strategy can be expected to make its way into international guidelines, notes joint lead author Irina Bancos, MD, an associate professor of endocrinology at the Mayo Clinic, Rochester, Minn., in a press statement issued by the University of Birmingham (England), which also had a number of researchers involved in the study.
“The findings of this study will feed into the next international guidelines on the management of adrenal tumors and the implementation of the new test will hopefully improve the overall outlook for patients diagnosed with adrenal tumors,” Dr. Bancos emphasized.
More imaging has led to detection of more adrenal tumors
Advances in CT and MRI imaging have increased the ability to detect adrenal incidentalomas, which are now picked up on about 5% of scans, and the widespread use of imaging has compounded the prevalence of such findings, particularly in older people.
Adrenocortical carcinomas represent only about 2%-12% of adrenal incidentalomas, but the prognosis is very poor, and early detection and surgery can improve outcomes, so findings of any adrenal tumor typically trigger additional multimodal imaging to rule out malignancy.
Evidence is lacking on the accuracy of imaging in determining whether such masses are truly cancerous, or benign, and such procedures add costs, as well as expose patients to radiation that may ultimately have no benefit. However, a previous proof-of-concept study from the same authors did show that the presence of excess adrenal steroid hormones in the urine is a key indicator of adrenal tumors, and other research has supported the findings.
All three tests together give best predictive value: EURINE-ACT
To further validate this work, the authors conducted the EURINE-ACT trial, a prospective 14-center study that is the first of its kind to evaluate the efficacy of a screening strategy for adrenocortical carcinoma that combines urine steroid profiling with tumor size and imaging characteristics.
The study of 2,017 participants with newly diagnosed adrenal masses, recruited from January 2011 to July 2016 from specialist centers in 11 different countries, assessed the diagnostic accuracy of three components: maximum tumor diameter (≥4 cm vs. <4 cm), imaging characteristics (positive vs. negative), and urine steroid metabolomics (low, medium, or high risk of adrenocortical carcinoma), separately and in combination.
Of the patients, 98 (4.9%) had adrenocortical carcinoma confirmed clinically, histopathologically, or biochemically.
Tumors with diameters of 4 cm or larger were identified in 488 patients (24.2%) and were observed in the vast majority of patients with adrenocortical carcinoma (96 of 98), for a positive predictive value (PPV) of 19.7%.
Likewise, the PPV for imaging characteristics was 19.7%. However, increasing the unenhanced CT tumor attenuation threshold to 20 Hounsfield units (HU) from the recommended 10 HU increased specificity for adrenocortical carcinoma (80.0% vs. 64.0%) while maintaining sensitivity (99.0% vs. 100.0%).
Comparatively, a urine steroid metabolomics result suggesting a high risk of adrenocortical carcinoma had a PPV of 34.6%.
A total of 106 patients (5.3%) met the criteria for all three measures, and the PPV for all three was 76.4%.
Using the criteria, 70 patients (3.5%) were classified as being at moderate risk of adrenocortical carcinoma and 1,841 (91.3%) at low risk, for a negative predictive value (NPV) of 99.7%.
“Use of radiation-free, noninvasive urine steroid metabolomics has a higher PPV than two standard imaging tests, and best performance was seen with the combination of all three tests,” the authors state.
Limit urine test to patients with larger tumors
They note that the use of the combined diagnostic strategy would have led to additional imaging in only 488 (24.2%) of the study’s 2,017 patients, compared with the 2,737 scans that were actually conducted before reaching a diagnostic decision.
“Implementation of urine steroid metabolomics in the routine diagnostic assessment of newly discovered adrenal masses could reduce the number of imaging procedures required to diagnose adrenocortical carcinoma and avoid unnecessary surgery of benign adrenal tumors, potentially yielding beneficial effects with respect to patient burden and health care costs,” they stress.
And regarding imaging parameters, “we also showed that using a cutoff of 20 HU for unenhanced CT tumor attenuation increases the accuracy of imaging characteristic assessment for exclusion of adrenocortical carcinoma, compared with the currently recommended cutoff of 10 HU, which has immediate implications for clinical practice,” they emphasize.
In an accompanying editorial, Adina F. Turcu, MD, of the division of metabolism, endocrinology, and diabetes, University of Michigan, Ann Arbor, and Axel K. Walch, MD, of the Helmholtz Zentrum München–German Research Centre for Environmental Health, agree. “The introduction of urine steroid metabolomics into routine clinical practice would provide major advantages,” they state.
However, they point out that, although the overall negative predictive value of the test was excellent, the specificity was weak.
“Thus, urine steroid metabolomics should be limited to patients who have adrenal nodules larger than 4 cm and have qualitative imaging characteristics suggestive of malignancy,” say Dr. Turcu and Dr. Walch.
The EURINE-ACT study results suggest this subgroup would represent roughly only 12% of all patients with adrenal incidentalomas, they add.
Issues that remain to be addressed with regard to the implementation of the screening strategy include how to best respond to patients who are classified as having intermediate or moderate risk of malignancy, and whether the diagnostic value of steroid metabolomics could be refined by adding analytes or parameters, the editorialists conclude.
The study was funded by the European Commission, U.K. Medical Research Council, Wellcome Trust, U.K. National Institute for Health Research, U.S. National Institutes of Health, the Claire Khan Trust Fund at University Hospitals Birmingham Charities, and the Mayo Clinic Foundation for Medical Education and Research.
A version of this article originally appeared on Medscape.com.
A strategy that includes a urine steroid test along with imaging characteristics and tumor size criteria can significantly improve the challenging diagnosis of adrenocortical cancer, helping to avoid unnecessary, and often unsuccessful, further imaging and even surgery, new research shows.
“A triple-test strategy of tumor diameter, imaging characteristics, and urine steroid metabolomics improves detection of adrenocortical carcinoma, which could shorten time to surgery for patients with ... carcinoma and help to avoid unnecessary surgery in patients with benign tumors,” the authors say in research published online July 23 in The Lancet Diabetes & Endocrinology.
The triple-test strategy can be expected to make its way into international guidelines, notes joint lead author Irina Bancos, MD, an associate professor of endocrinology at the Mayo Clinic, Rochester, Minn., in a press statement issued by the University of Birmingham (England), which also had a number of researchers involved in the study.
“The findings of this study will feed into the next international guidelines on the management of adrenal tumors and the implementation of the new test will hopefully improve the overall outlook for patients diagnosed with adrenal tumors,” Dr. Bancos emphasized.
More imaging has led to detection of more adrenal tumors
Advances in CT and MRI imaging have increased the ability to detect adrenal incidentalomas, which are now picked up on about 5% of scans, and the widespread use of imaging has compounded the prevalence of such findings, particularly in older people.
Adrenocortical carcinomas represent only about 2%-12% of adrenal incidentalomas, but the prognosis is very poor, and early detection and surgery can improve outcomes, so findings of any adrenal tumor typically trigger additional multimodal imaging to rule out malignancy.
Evidence is lacking on the accuracy of imaging in determining whether such masses are truly cancerous, or benign, and such procedures add costs, as well as expose patients to radiation that may ultimately have no benefit. However, a previous proof-of-concept study from the same authors did show that the presence of excess adrenal steroid hormones in the urine is a key indicator of adrenal tumors, and other research has supported the findings.
All three tests together give best predictive value: EURINE-ACT
To further validate this work, the authors conducted the EURINE-ACT trial, a prospective 14-center study that is the first of its kind to evaluate the efficacy of a screening strategy for adrenocortical carcinoma that combines urine steroid profiling with tumor size and imaging characteristics.
The study of 2,017 participants with newly diagnosed adrenal masses, recruited from January 2011 to July 2016 from specialist centers in 11 different countries, assessed the diagnostic accuracy of three components: maximum tumor diameter (≥4 cm vs. <4 cm), imaging characteristics (positive vs. negative), and urine steroid metabolomics (low, medium, or high risk of adrenocortical carcinoma), separately and in combination.
Of the patients, 98 (4.9%) had adrenocortical carcinoma confirmed clinically, histopathologically, or biochemically.
Tumors with diameters of 4 cm or larger were identified in 488 patients (24.2%) and were observed in the vast majority of patients with adrenocortical carcinoma (96 of 98), for a positive predictive value (PPV) of 19.7%.
Likewise, the PPV for imaging characteristics was 19.7%. However, increasing the unenhanced CT tumor attenuation threshold to 20 Hounsfield units (HU) from the recommended 10 HU increased specificity for adrenocortical carcinoma (80.0% vs. 64.0%) while maintaining sensitivity (99.0% vs. 100.0%).
Comparatively, a urine steroid metabolomics result suggesting a high risk of adrenocortical carcinoma had a PPV of 34.6%.
A total of 106 patients (5.3%) met the criteria for all three measures, and the PPV for all three was 76.4%.
Using the criteria, 70 patients (3.5%) were classified as being at moderate risk of adrenocortical carcinoma and 1,841 (91.3%) at low risk, for a negative predictive value (NPV) of 99.7%.
“Use of radiation-free, noninvasive urine steroid metabolomics has a higher PPV than two standard imaging tests, and best performance was seen with the combination of all three tests,” the authors state.
Limit urine test to patients with larger tumors
They note that the use of the combined diagnostic strategy would have led to additional imaging in only 488 (24.2%) of the study’s 2,017 patients, compared with the 2,737 scans that were actually conducted before reaching a diagnostic decision.
“Implementation of urine steroid metabolomics in the routine diagnostic assessment of newly discovered adrenal masses could reduce the number of imaging procedures required to diagnose adrenocortical carcinoma and avoid unnecessary surgery of benign adrenal tumors, potentially yielding beneficial effects with respect to patient burden and health care costs,” they stress.
And regarding imaging parameters, “we also showed that using a cutoff of 20 HU for unenhanced CT tumor attenuation increases the accuracy of imaging characteristic assessment for exclusion of adrenocortical carcinoma, compared with the currently recommended cutoff of 10 HU, which has immediate implications for clinical practice,” they emphasize.
In an accompanying editorial, Adina F. Turcu, MD, of the division of metabolism, endocrinology, and diabetes, University of Michigan, Ann Arbor, and Axel K. Walch, MD, of the Helmholtz Zentrum München–German Research Centre for Environmental Health, agree. “The introduction of urine steroid metabolomics into routine clinical practice would provide major advantages,” they state.
However, they point out that, although the overall negative predictive value of the test was excellent, the specificity was weak.
“Thus, urine steroid metabolomics should be limited to patients who have adrenal nodules larger than 4 cm and have qualitative imaging characteristics suggestive of malignancy,” say Dr. Turcu and Dr. Walch.
The EURINE-ACT study results suggest this subgroup would represent roughly only 12% of all patients with adrenal incidentalomas, they add.
Issues that remain to be addressed with regard to the implementation of the screening strategy include how to best respond to patients who are classified as having intermediate or moderate risk of malignancy, and whether the diagnostic value of steroid metabolomics could be refined by adding analytes or parameters, the editorialists conclude.
The study was funded by the European Commission, U.K. Medical Research Council, Wellcome Trust, U.K. National Institute for Health Research, U.S. National Institutes of Health, the Claire Khan Trust Fund at University Hospitals Birmingham Charities, and the Mayo Clinic Foundation for Medical Education and Research.
A version of this article originally appeared on Medscape.com.
A strategy that includes a urine steroid test along with imaging characteristics and tumor size criteria can significantly improve the challenging diagnosis of adrenocortical cancer, helping to avoid unnecessary, and often unsuccessful, further imaging and even surgery, new research shows.
“A triple-test strategy of tumor diameter, imaging characteristics, and urine steroid metabolomics improves detection of adrenocortical carcinoma, which could shorten time to surgery for patients with ... carcinoma and help to avoid unnecessary surgery in patients with benign tumors,” the authors say in research published online July 23 in The Lancet Diabetes & Endocrinology.
The triple-test strategy can be expected to make its way into international guidelines, notes joint lead author Irina Bancos, MD, an associate professor of endocrinology at the Mayo Clinic, Rochester, Minn., in a press statement issued by the University of Birmingham (England), which also had a number of researchers involved in the study.
“The findings of this study will feed into the next international guidelines on the management of adrenal tumors and the implementation of the new test will hopefully improve the overall outlook for patients diagnosed with adrenal tumors,” Dr. Bancos emphasized.
More imaging has led to detection of more adrenal tumors
Advances in CT and MRI imaging have increased the ability to detect adrenal incidentalomas, which are now picked up on about 5% of scans, and the widespread use of imaging has compounded the prevalence of such findings, particularly in older people.
Adrenocortical carcinomas represent only about 2%-12% of adrenal incidentalomas, but the prognosis is very poor, and early detection and surgery can improve outcomes, so findings of any adrenal tumor typically trigger additional multimodal imaging to rule out malignancy.
Evidence is lacking on the accuracy of imaging in determining whether such masses are truly cancerous, or benign, and such procedures add costs, as well as expose patients to radiation that may ultimately have no benefit. However, a previous proof-of-concept study from the same authors did show that the presence of excess adrenal steroid hormones in the urine is a key indicator of adrenal tumors, and other research has supported the findings.
All three tests together give best predictive value: EURINE-ACT
To further validate this work, the authors conducted the EURINE-ACT trial, a prospective 14-center study that is the first of its kind to evaluate the efficacy of a screening strategy for adrenocortical carcinoma that combines urine steroid profiling with tumor size and imaging characteristics.
The study of 2,017 participants with newly diagnosed adrenal masses, recruited from January 2011 to July 2016 from specialist centers in 11 different countries, assessed the diagnostic accuracy of three components: maximum tumor diameter (≥4 cm vs. <4 cm), imaging characteristics (positive vs. negative), and urine steroid metabolomics (low, medium, or high risk of adrenocortical carcinoma), separately and in combination.
Of the patients, 98 (4.9%) had adrenocortical carcinoma confirmed clinically, histopathologically, or biochemically.
Tumors with diameters of 4 cm or larger were identified in 488 patients (24.2%) and were observed in the vast majority of patients with adrenocortical carcinoma (96 of 98), for a positive predictive value (PPV) of 19.7%.
Likewise, the PPV for imaging characteristics was 19.7%. However, increasing the unenhanced CT tumor attenuation threshold to 20 Hounsfield units (HU) from the recommended 10 HU increased specificity for adrenocortical carcinoma (80.0% vs. 64.0%) while maintaining sensitivity (99.0% vs. 100.0%).
Comparatively, a urine steroid metabolomics result suggesting a high risk of adrenocortical carcinoma had a PPV of 34.6%.
A total of 106 patients (5.3%) met the criteria for all three measures, and the PPV for all three was 76.4%.
Using the criteria, 70 patients (3.5%) were classified as being at moderate risk of adrenocortical carcinoma and 1,841 (91.3%) at low risk, for a negative predictive value (NPV) of 99.7%.
“Use of radiation-free, noninvasive urine steroid metabolomics has a higher PPV than two standard imaging tests, and best performance was seen with the combination of all three tests,” the authors state.
Limit urine test to patients with larger tumors
They note that the use of the combined diagnostic strategy would have led to additional imaging in only 488 (24.2%) of the study’s 2,017 patients, compared with the 2,737 scans that were actually conducted before reaching a diagnostic decision.
“Implementation of urine steroid metabolomics in the routine diagnostic assessment of newly discovered adrenal masses could reduce the number of imaging procedures required to diagnose adrenocortical carcinoma and avoid unnecessary surgery of benign adrenal tumors, potentially yielding beneficial effects with respect to patient burden and health care costs,” they stress.
And regarding imaging parameters, “we also showed that using a cutoff of 20 HU for unenhanced CT tumor attenuation increases the accuracy of imaging characteristic assessment for exclusion of adrenocortical carcinoma, compared with the currently recommended cutoff of 10 HU, which has immediate implications for clinical practice,” they emphasize.
In an accompanying editorial, Adina F. Turcu, MD, of the division of metabolism, endocrinology, and diabetes, University of Michigan, Ann Arbor, and Axel K. Walch, MD, of the Helmholtz Zentrum München–German Research Centre for Environmental Health, agree. “The introduction of urine steroid metabolomics into routine clinical practice would provide major advantages,” they state.
However, they point out that, although the overall negative predictive value of the test was excellent, the specificity was weak.
“Thus, urine steroid metabolomics should be limited to patients who have adrenal nodules larger than 4 cm and have qualitative imaging characteristics suggestive of malignancy,” say Dr. Turcu and Dr. Walch.
The EURINE-ACT study results suggest this subgroup would represent roughly only 12% of all patients with adrenal incidentalomas, they add.
Issues that remain to be addressed with regard to the implementation of the screening strategy include how to best respond to patients who are classified as having intermediate or moderate risk of malignancy, and whether the diagnostic value of steroid metabolomics could be refined by adding analytes or parameters, the editorialists conclude.
The study was funded by the European Commission, U.K. Medical Research Council, Wellcome Trust, U.K. National Institute for Health Research, U.S. National Institutes of Health, the Claire Khan Trust Fund at University Hospitals Birmingham Charities, and the Mayo Clinic Foundation for Medical Education and Research.
A version of this article originally appeared on Medscape.com.
Levothyroxine: No LV benefit in subclinical hypothyroidism with MI
For patients with acute myocardial infarction (MI) and mild subclinical hypothyroidism (SCH), treatment with levothyroxine does not improve left ventricular function, according to results of the Thyroid in Acute Myocardial Infarction (ThyrAMI-2) trial.
“SCH is common, affecting approximately 10% of the adult population, and has been associated with worse outcomes in patients with cardiovascular disease in observational studies,” Salman Razvi, MD, Translational and Clinical Research Institute, Newcastle University, Newcastle upon Tyne, England, said in an interview.
This study shows that levothyroxine treatment for patients with SCH and acute MI is “unlikely to be of benefit,” he said.
“This study says that treating the thyroid failure does not help nor harm such patients,” Terry F. Davies, MD, director, division of endocrinology, diabetes, and bone diseases, Mount Sinai Beth Israel Medical Center, New York, said in an interview. He was not involved in the study, which was published online July 21 in JAMA.
Participants included 95 adults (mean age, 63.5 years; 72 men) with persistent mild SCH who presented with acute MI at six hospitals in the United Kingdom. Most (69%) had ST-segment elevation MI.
Inclusion criteria were age older than 18 years and serum thyrotropin level >4.0 mU/L with a normal free thyroxine level on two occasions 7-10 days apart and with one thyrotropin value <10 mU/L.
Forty-six participants were randomly allocated to receive levothyroxine starting at 25 mcg titrated to aim for serum thyrotropin levels between 0.4 and 2.5 mU/L and 49 to matching placebo capsules taken once daily for 52 weeks.
The primary outcome was left ventricular ejection fraction (LVEF) at 52 weeks, assessed via MRI, with adjustment for age, sex, acute MI type, affected coronary artery territory, and baseline LVEF.
Secondary outcomes were LV volume, infarct size, adverse events, and patient-reported outcome measures of health status, health-related quality of life, and depression.
The median daily dose of levothyroxine at the end of the study was 50 mcg. Adherence to study medication was 94% during the course of the study.
At week 52, mean LVEF improved from 51.3% at baseline to 53.8% in the levothyroxine group and from 54.0% to 56.1% in the placebo group.
The difference was not significant between groups, with an adjusted between-group difference of 0.76% (95% confidence interval, –0.93% to 2.46%; P = .37).
There were also no significant differences in any of the secondary outcomes. There were 15 (33.3%) cardiovascular adverse events in the levothyroxine group and 18 (36.7%) in the placebo group.
Recent clinical practice guidelines have highlighted a lack of high-quality data to make recommendations regarding the management of mild SCH, particularly for patients with cardiovascular disease, Dr. Razvi and colleagues noted in their article.
“On the basis of these findings, screening for and subsequent treatment of subclinical hypothyroidism in patients with acute myocardial infarction to preserve LV function is not justified,” they concluded.
Important caveats
The investigators noted several important caveats and limitations. The trial recruited patients with mild SCH because this group constitutes the majority of patients with SCH and for whom there is the “greatest uncertainty” regarding treatment efficacy. It’s not known whether targeting treatment for individuals with more severe disease may be beneficial.
The therapeutic benefit of levothyroxine may have been blunted, owing to the delay between coronary occlusion and the start of levothyroxine (median delay, 17 days). It’s unclear whether earlier treatment or treatment for a longer period may be beneficial.
But Dr. Davies noted that “treatment is usually avoided in the emergency situation,” and therefore he doesn’t think the treatment delay is a limitation; rather, “it would appear prudent,” he said in the interview.
“The real issues with an otherwise very careful study is the small size of the population despite the statistical assessment that this was all that was needed and, secondly, the small dose of thyroxine used,” Dr. Davies said.
The authors agree that the low dose of levothyroxine is a limitation. The median dose at the end of the study – 50 mcg daily – is “lower than that used in other trials that have demonstrated a benefit of treatment on endothelial function and lipid profiles,” they pointed out.
Dr. Davies noted that thyroid tests are “usually routine” for patients with MI. “Mild subclinical thyroid failure has been associated with worse cardiac outcomes, [but] treating such patients with thyroid hormone is very controversial since thyroid hormone can induce arrhythmias,” he said.
The study was supported in part by the National Institute for Health Research (NIHR) at the University of Leeds. Dr. Razvi received grants from the NIHR and nonfinancial support from Amdipharm Pharmaceuticals UK during the conduct of the study and personal fees from Merck and Abbott Pharmaceuticals outside the submitted work. Dr. Davies has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
For patients with acute myocardial infarction (MI) and mild subclinical hypothyroidism (SCH), treatment with levothyroxine does not improve left ventricular function, according to results of the Thyroid in Acute Myocardial Infarction (ThyrAMI-2) trial.
“SCH is common, affecting approximately 10% of the adult population, and has been associated with worse outcomes in patients with cardiovascular disease in observational studies,” Salman Razvi, MD, Translational and Clinical Research Institute, Newcastle University, Newcastle upon Tyne, England, said in an interview.
This study shows that levothyroxine treatment for patients with SCH and acute MI is “unlikely to be of benefit,” he said.
“This study says that treating the thyroid failure does not help nor harm such patients,” Terry F. Davies, MD, director, division of endocrinology, diabetes, and bone diseases, Mount Sinai Beth Israel Medical Center, New York, said in an interview. He was not involved in the study, which was published online July 21 in JAMA.
Participants included 95 adults (mean age, 63.5 years; 72 men) with persistent mild SCH who presented with acute MI at six hospitals in the United Kingdom. Most (69%) had ST-segment elevation MI.
Inclusion criteria were age older than 18 years and serum thyrotropin level >4.0 mU/L with a normal free thyroxine level on two occasions 7-10 days apart and with one thyrotropin value <10 mU/L.
Forty-six participants were randomly allocated to receive levothyroxine starting at 25 mcg titrated to aim for serum thyrotropin levels between 0.4 and 2.5 mU/L and 49 to matching placebo capsules taken once daily for 52 weeks.
The primary outcome was left ventricular ejection fraction (LVEF) at 52 weeks, assessed via MRI, with adjustment for age, sex, acute MI type, affected coronary artery territory, and baseline LVEF.
Secondary outcomes were LV volume, infarct size, adverse events, and patient-reported outcome measures of health status, health-related quality of life, and depression.
The median daily dose of levothyroxine at the end of the study was 50 mcg. Adherence to study medication was 94% during the course of the study.
At week 52, mean LVEF improved from 51.3% at baseline to 53.8% in the levothyroxine group and from 54.0% to 56.1% in the placebo group.
The difference was not significant between groups, with an adjusted between-group difference of 0.76% (95% confidence interval, –0.93% to 2.46%; P = .37).
There were also no significant differences in any of the secondary outcomes. There were 15 (33.3%) cardiovascular adverse events in the levothyroxine group and 18 (36.7%) in the placebo group.
Recent clinical practice guidelines have highlighted a lack of high-quality data to make recommendations regarding the management of mild SCH, particularly for patients with cardiovascular disease, Dr. Razvi and colleagues noted in their article.
“On the basis of these findings, screening for and subsequent treatment of subclinical hypothyroidism in patients with acute myocardial infarction to preserve LV function is not justified,” they concluded.
Important caveats
The investigators noted several important caveats and limitations. The trial recruited patients with mild SCH because this group constitutes the majority of patients with SCH and for whom there is the “greatest uncertainty” regarding treatment efficacy. It’s not known whether targeting treatment for individuals with more severe disease may be beneficial.
The therapeutic benefit of levothyroxine may have been blunted, owing to the delay between coronary occlusion and the start of levothyroxine (median delay, 17 days). It’s unclear whether earlier treatment or treatment for a longer period may be beneficial.
But Dr. Davies noted that “treatment is usually avoided in the emergency situation,” and therefore he doesn’t think the treatment delay is a limitation; rather, “it would appear prudent,” he said in the interview.
“The real issues with an otherwise very careful study is the small size of the population despite the statistical assessment that this was all that was needed and, secondly, the small dose of thyroxine used,” Dr. Davies said.
The authors agree that the low dose of levothyroxine is a limitation. The median dose at the end of the study – 50 mcg daily – is “lower than that used in other trials that have demonstrated a benefit of treatment on endothelial function and lipid profiles,” they pointed out.
Dr. Davies noted that thyroid tests are “usually routine” for patients with MI. “Mild subclinical thyroid failure has been associated with worse cardiac outcomes, [but] treating such patients with thyroid hormone is very controversial since thyroid hormone can induce arrhythmias,” he said.
The study was supported in part by the National Institute for Health Research (NIHR) at the University of Leeds. Dr. Razvi received grants from the NIHR and nonfinancial support from Amdipharm Pharmaceuticals UK during the conduct of the study and personal fees from Merck and Abbott Pharmaceuticals outside the submitted work. Dr. Davies has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
For patients with acute myocardial infarction (MI) and mild subclinical hypothyroidism (SCH), treatment with levothyroxine does not improve left ventricular function, according to results of the Thyroid in Acute Myocardial Infarction (ThyrAMI-2) trial.
“SCH is common, affecting approximately 10% of the adult population, and has been associated with worse outcomes in patients with cardiovascular disease in observational studies,” Salman Razvi, MD, Translational and Clinical Research Institute, Newcastle University, Newcastle upon Tyne, England, said in an interview.
This study shows that levothyroxine treatment for patients with SCH and acute MI is “unlikely to be of benefit,” he said.
“This study says that treating the thyroid failure does not help nor harm such patients,” Terry F. Davies, MD, director, division of endocrinology, diabetes, and bone diseases, Mount Sinai Beth Israel Medical Center, New York, said in an interview. He was not involved in the study, which was published online July 21 in JAMA.
Participants included 95 adults (mean age, 63.5 years; 72 men) with persistent mild SCH who presented with acute MI at six hospitals in the United Kingdom. Most (69%) had ST-segment elevation MI.
Inclusion criteria were age older than 18 years and serum thyrotropin level >4.0 mU/L with a normal free thyroxine level on two occasions 7-10 days apart and with one thyrotropin value <10 mU/L.
Forty-six participants were randomly allocated to receive levothyroxine starting at 25 mcg titrated to aim for serum thyrotropin levels between 0.4 and 2.5 mU/L and 49 to matching placebo capsules taken once daily for 52 weeks.
The primary outcome was left ventricular ejection fraction (LVEF) at 52 weeks, assessed via MRI, with adjustment for age, sex, acute MI type, affected coronary artery territory, and baseline LVEF.
Secondary outcomes were LV volume, infarct size, adverse events, and patient-reported outcome measures of health status, health-related quality of life, and depression.
The median daily dose of levothyroxine at the end of the study was 50 mcg. Adherence to study medication was 94% during the course of the study.
At week 52, mean LVEF improved from 51.3% at baseline to 53.8% in the levothyroxine group and from 54.0% to 56.1% in the placebo group.
The difference was not significant between groups, with an adjusted between-group difference of 0.76% (95% confidence interval, –0.93% to 2.46%; P = .37).
There were also no significant differences in any of the secondary outcomes. There were 15 (33.3%) cardiovascular adverse events in the levothyroxine group and 18 (36.7%) in the placebo group.
Recent clinical practice guidelines have highlighted a lack of high-quality data to make recommendations regarding the management of mild SCH, particularly for patients with cardiovascular disease, Dr. Razvi and colleagues noted in their article.
“On the basis of these findings, screening for and subsequent treatment of subclinical hypothyroidism in patients with acute myocardial infarction to preserve LV function is not justified,” they concluded.
Important caveats
The investigators noted several important caveats and limitations. The trial recruited patients with mild SCH because this group constitutes the majority of patients with SCH and for whom there is the “greatest uncertainty” regarding treatment efficacy. It’s not known whether targeting treatment for individuals with more severe disease may be beneficial.
The therapeutic benefit of levothyroxine may have been blunted, owing to the delay between coronary occlusion and the start of levothyroxine (median delay, 17 days). It’s unclear whether earlier treatment or treatment for a longer period may be beneficial.
But Dr. Davies noted that “treatment is usually avoided in the emergency situation,” and therefore he doesn’t think the treatment delay is a limitation; rather, “it would appear prudent,” he said in the interview.
“The real issues with an otherwise very careful study is the small size of the population despite the statistical assessment that this was all that was needed and, secondly, the small dose of thyroxine used,” Dr. Davies said.
The authors agree that the low dose of levothyroxine is a limitation. The median dose at the end of the study – 50 mcg daily – is “lower than that used in other trials that have demonstrated a benefit of treatment on endothelial function and lipid profiles,” they pointed out.
Dr. Davies noted that thyroid tests are “usually routine” for patients with MI. “Mild subclinical thyroid failure has been associated with worse cardiac outcomes, [but] treating such patients with thyroid hormone is very controversial since thyroid hormone can induce arrhythmias,” he said.
The study was supported in part by the National Institute for Health Research (NIHR) at the University of Leeds. Dr. Razvi received grants from the NIHR and nonfinancial support from Amdipharm Pharmaceuticals UK during the conduct of the study and personal fees from Merck and Abbott Pharmaceuticals outside the submitted work. Dr. Davies has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Cushing’s and COVID-19: Nontraditional symptoms keys to assessment, treatments
Do not rely on more traditional signs and symptoms of COVID-19 like fever and dyspnea when assessing patients with Cushing’s syndrome for the novel coronavirus, Rosario Pivonello, MD, PhD, and colleagues urged.
Physicians evaluating patients with Cushing’s syndrome for COVID-19 “should be suspicious of any change in health status of their patients with Cushing’s syndrome, rather than relying on fever and [dyspnea] as typical features,” Dr. Pivonello, an endocrinologist with the University of Naples (Italy) Federico II, and colleagues wrote in a commentary published in The Lancet Diabetes & Endocrinology.
COVID-19 symptoms are a unique concern among patients with Cushing’s syndrome because many of the cardiometabolic and immune impairments that place someone at higher risk of more severe disease or mortality for the novel coronavirus – such as obesity, hypertension, diabetes, and immunodeficiency syndromes – are also shared with Cushing’s syndrome.
Increased cardiovascular risk factors and susceptibility to severe infection are “two leading causes of death” for patients with Cushing’s syndrome, Dr. Pivonello and colleagues noted.
The immunocompromised state of patients with Cushing’s syndrome may make detection of COVID-19 infection difficult, the authors say. For example, fever is a common symptom of patients with COVID-19, but in patients with active Cushing’s syndrome, “low-grade chronic inflammation and the poor immune response might limit febrile response in the early phase of infection,” Dr. Pivonello and colleagues wrote.
In other cases, because Cushing’s syndrome and COVID-19 have overlapping symptoms, it may be difficult to attribute a particular symptom to either disease. Dyspnea is a common symptom of COVID-19, but may present in Cushing’s syndrome because of “cardiac insufficiency or weakness of respiratory muscles,” the authors wrote. Instead, physicians should look to other COVID-19 symptoms, such as cough, dysgeusia, anosmia, and diarrhea, for signs of the disease.
Patients with Cushing’s syndrome may also be predisposed to a more severe course of COVID-19 because of the prevalence of obesity, hypertension, or diabetes in these patients, which have been identified as comorbidities that increase the likelihood of severe COVID-19 and progression to acute respiratory distress syndrome (ARDS). “However, a key element in the development of ARDS during COVID-19 is the exaggerated cellular response induced by the cytokine increase, leading to massive alveolar–capillary wall damage and a decline in gas exchange,” Dr. Pivonello and colleagues wrote. “Because patients with Cushing’s syndrome might not mount a normal cytokine response, these patients might [paradoxically] be less prone to develop severe ARDS with COVID-19.”
As both Cushing’s syndrome and COVID-19 are associated with hypercoagulability, the authors “strongly advise” using low-molecular-weight heparin in hospitalized patients with active Cushing’s syndrome who develop COVID-19. In both diseases, there is also a risk of longer duration of viral infections and opportunistic infections such as atypical bacterial and invasive fungal infections. For this reason, the authors also recommended patients with Cushing’s syndrome who have COVID-19 be placed on prolonged antiviral and broad-spectrum antibiotic treatment as a prophylactic measure.
During the pandemic, avoiding surgery for Cushing’s syndrome should be considered to reduce the likelihood of acquiring COVID-19 in a hospital setting, the authors wrote. Medical therapy can be temporarily used where appropriate, such as using ketoconazole, metyrapone, osilodrostat, and etomidate to lower cortisol levels. They acknowledge that some cases of malignant Cushing’s syndrome may require “expeditious definitive diagnosis and proper surgical resolution.”
After remission, while infection risk should be significantly lowered, other comorbidities like obesity, hypertension, diabetes, and thromboembolic diathesis may remain. “Because these are features associated with an increased death risk in patients with COVID-19, patients with Cushing’s syndrome in remission should be considered a high-risk population and consequently adopt adequate self-protection strategies to [minimize] contagion risk,” the authors wrote.
Dr. Pivonello reported relationships with Novartis, Strongbridge Biopharma, HRA Pharma, Ipsen, Shire, and Pfizer, Corcept Therapeutics, IBSA Farmaceutici, Ferring, and Italfarmaco in the form of receiving grants and/or personal fees. One coauthor reported receiving grants and/or nonfinancial support from Takeda, Ipsen, Shire, Pfizer, and Corcept Therapeutics. One coauthor reported receiving grants and personal fees from Novartis and Strongbridge, and grants from Millendo Therapeutics. Another coauthor reported receiving grants and/or personal fees from Novartis, Ipsen, Shire, Pfizer, Italfarmaco, Lilly, Merck, and Novo Nordisk. The other authors reported no relevant conflicts of interest.
SOURCE: Pivonello R et al. Lancet Diabetes Endocrinol. 2020 Jun 9. doi: 10.1016/S2213-8587(20)30215-1.
Do not rely on more traditional signs and symptoms of COVID-19 like fever and dyspnea when assessing patients with Cushing’s syndrome for the novel coronavirus, Rosario Pivonello, MD, PhD, and colleagues urged.
Physicians evaluating patients with Cushing’s syndrome for COVID-19 “should be suspicious of any change in health status of their patients with Cushing’s syndrome, rather than relying on fever and [dyspnea] as typical features,” Dr. Pivonello, an endocrinologist with the University of Naples (Italy) Federico II, and colleagues wrote in a commentary published in The Lancet Diabetes & Endocrinology.
COVID-19 symptoms are a unique concern among patients with Cushing’s syndrome because many of the cardiometabolic and immune impairments that place someone at higher risk of more severe disease or mortality for the novel coronavirus – such as obesity, hypertension, diabetes, and immunodeficiency syndromes – are also shared with Cushing’s syndrome.
Increased cardiovascular risk factors and susceptibility to severe infection are “two leading causes of death” for patients with Cushing’s syndrome, Dr. Pivonello and colleagues noted.
The immunocompromised state of patients with Cushing’s syndrome may make detection of COVID-19 infection difficult, the authors say. For example, fever is a common symptom of patients with COVID-19, but in patients with active Cushing’s syndrome, “low-grade chronic inflammation and the poor immune response might limit febrile response in the early phase of infection,” Dr. Pivonello and colleagues wrote.
In other cases, because Cushing’s syndrome and COVID-19 have overlapping symptoms, it may be difficult to attribute a particular symptom to either disease. Dyspnea is a common symptom of COVID-19, but may present in Cushing’s syndrome because of “cardiac insufficiency or weakness of respiratory muscles,” the authors wrote. Instead, physicians should look to other COVID-19 symptoms, such as cough, dysgeusia, anosmia, and diarrhea, for signs of the disease.
Patients with Cushing’s syndrome may also be predisposed to a more severe course of COVID-19 because of the prevalence of obesity, hypertension, or diabetes in these patients, which have been identified as comorbidities that increase the likelihood of severe COVID-19 and progression to acute respiratory distress syndrome (ARDS). “However, a key element in the development of ARDS during COVID-19 is the exaggerated cellular response induced by the cytokine increase, leading to massive alveolar–capillary wall damage and a decline in gas exchange,” Dr. Pivonello and colleagues wrote. “Because patients with Cushing’s syndrome might not mount a normal cytokine response, these patients might [paradoxically] be less prone to develop severe ARDS with COVID-19.”
As both Cushing’s syndrome and COVID-19 are associated with hypercoagulability, the authors “strongly advise” using low-molecular-weight heparin in hospitalized patients with active Cushing’s syndrome who develop COVID-19. In both diseases, there is also a risk of longer duration of viral infections and opportunistic infections such as atypical bacterial and invasive fungal infections. For this reason, the authors also recommended patients with Cushing’s syndrome who have COVID-19 be placed on prolonged antiviral and broad-spectrum antibiotic treatment as a prophylactic measure.
During the pandemic, avoiding surgery for Cushing’s syndrome should be considered to reduce the likelihood of acquiring COVID-19 in a hospital setting, the authors wrote. Medical therapy can be temporarily used where appropriate, such as using ketoconazole, metyrapone, osilodrostat, and etomidate to lower cortisol levels. They acknowledge that some cases of malignant Cushing’s syndrome may require “expeditious definitive diagnosis and proper surgical resolution.”
After remission, while infection risk should be significantly lowered, other comorbidities like obesity, hypertension, diabetes, and thromboembolic diathesis may remain. “Because these are features associated with an increased death risk in patients with COVID-19, patients with Cushing’s syndrome in remission should be considered a high-risk population and consequently adopt adequate self-protection strategies to [minimize] contagion risk,” the authors wrote.
Dr. Pivonello reported relationships with Novartis, Strongbridge Biopharma, HRA Pharma, Ipsen, Shire, and Pfizer, Corcept Therapeutics, IBSA Farmaceutici, Ferring, and Italfarmaco in the form of receiving grants and/or personal fees. One coauthor reported receiving grants and/or nonfinancial support from Takeda, Ipsen, Shire, Pfizer, and Corcept Therapeutics. One coauthor reported receiving grants and personal fees from Novartis and Strongbridge, and grants from Millendo Therapeutics. Another coauthor reported receiving grants and/or personal fees from Novartis, Ipsen, Shire, Pfizer, Italfarmaco, Lilly, Merck, and Novo Nordisk. The other authors reported no relevant conflicts of interest.
SOURCE: Pivonello R et al. Lancet Diabetes Endocrinol. 2020 Jun 9. doi: 10.1016/S2213-8587(20)30215-1.
Do not rely on more traditional signs and symptoms of COVID-19 like fever and dyspnea when assessing patients with Cushing’s syndrome for the novel coronavirus, Rosario Pivonello, MD, PhD, and colleagues urged.
Physicians evaluating patients with Cushing’s syndrome for COVID-19 “should be suspicious of any change in health status of their patients with Cushing’s syndrome, rather than relying on fever and [dyspnea] as typical features,” Dr. Pivonello, an endocrinologist with the University of Naples (Italy) Federico II, and colleagues wrote in a commentary published in The Lancet Diabetes & Endocrinology.
COVID-19 symptoms are a unique concern among patients with Cushing’s syndrome because many of the cardiometabolic and immune impairments that place someone at higher risk of more severe disease or mortality for the novel coronavirus – such as obesity, hypertension, diabetes, and immunodeficiency syndromes – are also shared with Cushing’s syndrome.
Increased cardiovascular risk factors and susceptibility to severe infection are “two leading causes of death” for patients with Cushing’s syndrome, Dr. Pivonello and colleagues noted.
The immunocompromised state of patients with Cushing’s syndrome may make detection of COVID-19 infection difficult, the authors say. For example, fever is a common symptom of patients with COVID-19, but in patients with active Cushing’s syndrome, “low-grade chronic inflammation and the poor immune response might limit febrile response in the early phase of infection,” Dr. Pivonello and colleagues wrote.
In other cases, because Cushing’s syndrome and COVID-19 have overlapping symptoms, it may be difficult to attribute a particular symptom to either disease. Dyspnea is a common symptom of COVID-19, but may present in Cushing’s syndrome because of “cardiac insufficiency or weakness of respiratory muscles,” the authors wrote. Instead, physicians should look to other COVID-19 symptoms, such as cough, dysgeusia, anosmia, and diarrhea, for signs of the disease.
Patients with Cushing’s syndrome may also be predisposed to a more severe course of COVID-19 because of the prevalence of obesity, hypertension, or diabetes in these patients, which have been identified as comorbidities that increase the likelihood of severe COVID-19 and progression to acute respiratory distress syndrome (ARDS). “However, a key element in the development of ARDS during COVID-19 is the exaggerated cellular response induced by the cytokine increase, leading to massive alveolar–capillary wall damage and a decline in gas exchange,” Dr. Pivonello and colleagues wrote. “Because patients with Cushing’s syndrome might not mount a normal cytokine response, these patients might [paradoxically] be less prone to develop severe ARDS with COVID-19.”
As both Cushing’s syndrome and COVID-19 are associated with hypercoagulability, the authors “strongly advise” using low-molecular-weight heparin in hospitalized patients with active Cushing’s syndrome who develop COVID-19. In both diseases, there is also a risk of longer duration of viral infections and opportunistic infections such as atypical bacterial and invasive fungal infections. For this reason, the authors also recommended patients with Cushing’s syndrome who have COVID-19 be placed on prolonged antiviral and broad-spectrum antibiotic treatment as a prophylactic measure.
During the pandemic, avoiding surgery for Cushing’s syndrome should be considered to reduce the likelihood of acquiring COVID-19 in a hospital setting, the authors wrote. Medical therapy can be temporarily used where appropriate, such as using ketoconazole, metyrapone, osilodrostat, and etomidate to lower cortisol levels. They acknowledge that some cases of malignant Cushing’s syndrome may require “expeditious definitive diagnosis and proper surgical resolution.”
After remission, while infection risk should be significantly lowered, other comorbidities like obesity, hypertension, diabetes, and thromboembolic diathesis may remain. “Because these are features associated with an increased death risk in patients with COVID-19, patients with Cushing’s syndrome in remission should be considered a high-risk population and consequently adopt adequate self-protection strategies to [minimize] contagion risk,” the authors wrote.
Dr. Pivonello reported relationships with Novartis, Strongbridge Biopharma, HRA Pharma, Ipsen, Shire, and Pfizer, Corcept Therapeutics, IBSA Farmaceutici, Ferring, and Italfarmaco in the form of receiving grants and/or personal fees. One coauthor reported receiving grants and/or nonfinancial support from Takeda, Ipsen, Shire, Pfizer, and Corcept Therapeutics. One coauthor reported receiving grants and personal fees from Novartis and Strongbridge, and grants from Millendo Therapeutics. Another coauthor reported receiving grants and/or personal fees from Novartis, Ipsen, Shire, Pfizer, Italfarmaco, Lilly, Merck, and Novo Nordisk. The other authors reported no relevant conflicts of interest.
SOURCE: Pivonello R et al. Lancet Diabetes Endocrinol. 2020 Jun 9. doi: 10.1016/S2213-8587(20)30215-1.
FROM THE LANCET DIABETES & ENDOCRINOLOGY
FDA approves first oral somatostatin analog for acromegaly
who previously responded to and tolerated octreotide or lanreotide injections.
“People living with acromegaly experience many challenges associated with injectable therapies and are in need of new treatment options,” Jill Sisco, president of Acromegaly Community, a patient support group, said in a Chiasma press release.
“The entire acromegaly community has long awaited oral therapeutic options and it is gratifying to see that the FDA has now approved the first oral somatostatin analog (SSA) therapy with the potential to make a significant impact in the lives of people with acromegaly and their caregivers,” she added.
Acromegaly, a rare, chronic disease usually caused by a benign pituitary tumor that leads to excess production of growth hormone and insulin-like growth factor-1 (IGF-1) hormone, can be cured through the successful surgical removal of the pituitary tumor. However, management of the disease remains a lifelong challenge for many who must rely on chronic injections.
The new oral formulation of octreotide is the first and only oral somatostatin analog approved by the FDA.
The approval was based on the results of the 9-month, phase 3 pivotal CHIASMA OPTIMAL clinical trial, involving 56 adults with acromegaly controlled by injectable SSAs.
The patients, who were randomized 1:1 to octreotide capsules or placebo, were dose-titrated from 40 mg/day up to a maximum of 80 mg/day, equaling two capsules in the morning and two in the evening.
The study met its primary endpoint. Overall, 58% of patients taking octreotide maintained IGF-1 response compared with 19% of those on placebo at the end of 9 months (P = .008), according to the average of the last two IGF-1 levels that were 1 times or less the upper limit of normal, assessed at weeks 34 and 36.
The trial also met its secondary endpoints, which included the proportion of patients who maintain growth hormone response at week 36 compared with screening; time to loss of response; and proportion of patients requiring reversion to prior treatment.
Safety data were favorable. Adverse reactions to the drug, detailed in the prescribing information, include cholelithiasis and associated complications; hyperglycemia and hypoglycemia; thyroid function abnormalities; cardiac function abnormalities; decreased vitamin B12 levels, and abnormal Schilling’s test results.
Results from the clinical trial “are encouraging for patients with acromegaly,” the study’s principal investigator, Susan Samson, MD, PhD, of Baylor College of Medicine, Houston, said in the Chiasma statement.
“Based on data from the CHIASMA OPTIMAL trial showing patients on therapy being able to maintain mean IGF-1 levels within the normal range at the end of treatment, I believe oral octreotide capsules hold meaningful promise for patients with this disease and will address a long-standing unmet treatment need,” she added.
Chiasma reports that it expects Mycapssa to be available in the fourth quarter of 2020, pending FDA approval of a planned manufacturing supplement to the approved new drug application.
The company further plans to provide patient support services including assistance with insurance providers and specialty pharmacies and support in incorporating treatment into patients’ daily routines.
Despite effective biochemical control of growth hormone, many patients with acromegaly continue to suffer symptoms, mainly because of comorbidities, so it is important that these are also adequately treated, a consensus group concluded earlier this year.
The CHIASMA OPTIMAL trial was funded by Chiasma.
A version of this article originally appeared on Medscape.com.
who previously responded to and tolerated octreotide or lanreotide injections.
“People living with acromegaly experience many challenges associated with injectable therapies and are in need of new treatment options,” Jill Sisco, president of Acromegaly Community, a patient support group, said in a Chiasma press release.
“The entire acromegaly community has long awaited oral therapeutic options and it is gratifying to see that the FDA has now approved the first oral somatostatin analog (SSA) therapy with the potential to make a significant impact in the lives of people with acromegaly and their caregivers,” she added.
Acromegaly, a rare, chronic disease usually caused by a benign pituitary tumor that leads to excess production of growth hormone and insulin-like growth factor-1 (IGF-1) hormone, can be cured through the successful surgical removal of the pituitary tumor. However, management of the disease remains a lifelong challenge for many who must rely on chronic injections.
The new oral formulation of octreotide is the first and only oral somatostatin analog approved by the FDA.
The approval was based on the results of the 9-month, phase 3 pivotal CHIASMA OPTIMAL clinical trial, involving 56 adults with acromegaly controlled by injectable SSAs.
The patients, who were randomized 1:1 to octreotide capsules or placebo, were dose-titrated from 40 mg/day up to a maximum of 80 mg/day, equaling two capsules in the morning and two in the evening.
The study met its primary endpoint. Overall, 58% of patients taking octreotide maintained IGF-1 response compared with 19% of those on placebo at the end of 9 months (P = .008), according to the average of the last two IGF-1 levels that were 1 times or less the upper limit of normal, assessed at weeks 34 and 36.
The trial also met its secondary endpoints, which included the proportion of patients who maintain growth hormone response at week 36 compared with screening; time to loss of response; and proportion of patients requiring reversion to prior treatment.
Safety data were favorable. Adverse reactions to the drug, detailed in the prescribing information, include cholelithiasis and associated complications; hyperglycemia and hypoglycemia; thyroid function abnormalities; cardiac function abnormalities; decreased vitamin B12 levels, and abnormal Schilling’s test results.
Results from the clinical trial “are encouraging for patients with acromegaly,” the study’s principal investigator, Susan Samson, MD, PhD, of Baylor College of Medicine, Houston, said in the Chiasma statement.
“Based on data from the CHIASMA OPTIMAL trial showing patients on therapy being able to maintain mean IGF-1 levels within the normal range at the end of treatment, I believe oral octreotide capsules hold meaningful promise for patients with this disease and will address a long-standing unmet treatment need,” she added.
Chiasma reports that it expects Mycapssa to be available in the fourth quarter of 2020, pending FDA approval of a planned manufacturing supplement to the approved new drug application.
The company further plans to provide patient support services including assistance with insurance providers and specialty pharmacies and support in incorporating treatment into patients’ daily routines.
Despite effective biochemical control of growth hormone, many patients with acromegaly continue to suffer symptoms, mainly because of comorbidities, so it is important that these are also adequately treated, a consensus group concluded earlier this year.
The CHIASMA OPTIMAL trial was funded by Chiasma.
A version of this article originally appeared on Medscape.com.
who previously responded to and tolerated octreotide or lanreotide injections.
“People living with acromegaly experience many challenges associated with injectable therapies and are in need of new treatment options,” Jill Sisco, president of Acromegaly Community, a patient support group, said in a Chiasma press release.
“The entire acromegaly community has long awaited oral therapeutic options and it is gratifying to see that the FDA has now approved the first oral somatostatin analog (SSA) therapy with the potential to make a significant impact in the lives of people with acromegaly and their caregivers,” she added.
Acromegaly, a rare, chronic disease usually caused by a benign pituitary tumor that leads to excess production of growth hormone and insulin-like growth factor-1 (IGF-1) hormone, can be cured through the successful surgical removal of the pituitary tumor. However, management of the disease remains a lifelong challenge for many who must rely on chronic injections.
The new oral formulation of octreotide is the first and only oral somatostatin analog approved by the FDA.
The approval was based on the results of the 9-month, phase 3 pivotal CHIASMA OPTIMAL clinical trial, involving 56 adults with acromegaly controlled by injectable SSAs.
The patients, who were randomized 1:1 to octreotide capsules or placebo, were dose-titrated from 40 mg/day up to a maximum of 80 mg/day, equaling two capsules in the morning and two in the evening.
The study met its primary endpoint. Overall, 58% of patients taking octreotide maintained IGF-1 response compared with 19% of those on placebo at the end of 9 months (P = .008), according to the average of the last two IGF-1 levels that were 1 times or less the upper limit of normal, assessed at weeks 34 and 36.
The trial also met its secondary endpoints, which included the proportion of patients who maintain growth hormone response at week 36 compared with screening; time to loss of response; and proportion of patients requiring reversion to prior treatment.
Safety data were favorable. Adverse reactions to the drug, detailed in the prescribing information, include cholelithiasis and associated complications; hyperglycemia and hypoglycemia; thyroid function abnormalities; cardiac function abnormalities; decreased vitamin B12 levels, and abnormal Schilling’s test results.
Results from the clinical trial “are encouraging for patients with acromegaly,” the study’s principal investigator, Susan Samson, MD, PhD, of Baylor College of Medicine, Houston, said in the Chiasma statement.
“Based on data from the CHIASMA OPTIMAL trial showing patients on therapy being able to maintain mean IGF-1 levels within the normal range at the end of treatment, I believe oral octreotide capsules hold meaningful promise for patients with this disease and will address a long-standing unmet treatment need,” she added.
Chiasma reports that it expects Mycapssa to be available in the fourth quarter of 2020, pending FDA approval of a planned manufacturing supplement to the approved new drug application.
The company further plans to provide patient support services including assistance with insurance providers and specialty pharmacies and support in incorporating treatment into patients’ daily routines.
Despite effective biochemical control of growth hormone, many patients with acromegaly continue to suffer symptoms, mainly because of comorbidities, so it is important that these are also adequately treated, a consensus group concluded earlier this year.
The CHIASMA OPTIMAL trial was funded by Chiasma.
A version of this article originally appeared on Medscape.com.
Increased hypothyroidism risk seen in young men with HS
Anna Figueiredo, MD, declared at the virtual annual meeting of the American Academy of Dermatology.
The surprise about this finding from a large retrospective case-control study stems from the fact that the elevated risk for hypothyroidism didn’t also extend to younger women with hidradenitis suppurativa (HS) nor to patients older than 40 years of either gender, explained Dr. Figueiredo of the department of dermatology at Northwestern University, Chicago.
She presented a retrospective case-control study based on information extracted from a medical records database of more than 8 million Midwestern adults. Among nearly 141,000 dermatology patients with follow-up in the database for at least 1 year, there were 405 HS patients aged 18-40 years and 327 aged 41-89.
In an age-matched comparison with the dermatology patients without HS, the younger HS cohort was at a significant 1.52-fold increased risk for comorbid hypothyroidism. Upon further stratification by sex, only the younger men with HS were at increased risk. Those patients were at 3.95-fold greater risk for having a diagnosis of hypothyroidism than were age-matched younger male dermatology patients.
Both younger and older HS patients were at numerically increased risk for being diagnosed with hyperthyroidism; however, this difference didn’t approach statistical significance because there were so few cases: a total of just eight in the HS population across the full age spectrum.
Hidradenitis suppurativa is a chronic inflammatory disease with an estimated prevalence of up to 4% in the United States. Growing evidence suggests it is an immune-mediated disorder because the tumor necrosis factor inhibitor adalimumab (Humira) has been approved for treatment of HS.
Thyroid disease is also often autoimmune-mediated, but its relationship with HS hasn’t been extensively examined. A recent meta-analysis of five case-control studies concluded that HS was associated with a 1.36-fold increased risk of thyroid disease; however, the Nepalese investigators didn’t distinguish between hypo- and hyperthyroidism (J Am Acad Dermatol. 2020 Feb;82[2]:491-3).
Dr. Figueiredo reported having no financial conflicts regarding her study, which was without commercial support.
Anna Figueiredo, MD, declared at the virtual annual meeting of the American Academy of Dermatology.
The surprise about this finding from a large retrospective case-control study stems from the fact that the elevated risk for hypothyroidism didn’t also extend to younger women with hidradenitis suppurativa (HS) nor to patients older than 40 years of either gender, explained Dr. Figueiredo of the department of dermatology at Northwestern University, Chicago.
She presented a retrospective case-control study based on information extracted from a medical records database of more than 8 million Midwestern adults. Among nearly 141,000 dermatology patients with follow-up in the database for at least 1 year, there were 405 HS patients aged 18-40 years and 327 aged 41-89.
In an age-matched comparison with the dermatology patients without HS, the younger HS cohort was at a significant 1.52-fold increased risk for comorbid hypothyroidism. Upon further stratification by sex, only the younger men with HS were at increased risk. Those patients were at 3.95-fold greater risk for having a diagnosis of hypothyroidism than were age-matched younger male dermatology patients.
Both younger and older HS patients were at numerically increased risk for being diagnosed with hyperthyroidism; however, this difference didn’t approach statistical significance because there were so few cases: a total of just eight in the HS population across the full age spectrum.
Hidradenitis suppurativa is a chronic inflammatory disease with an estimated prevalence of up to 4% in the United States. Growing evidence suggests it is an immune-mediated disorder because the tumor necrosis factor inhibitor adalimumab (Humira) has been approved for treatment of HS.
Thyroid disease is also often autoimmune-mediated, but its relationship with HS hasn’t been extensively examined. A recent meta-analysis of five case-control studies concluded that HS was associated with a 1.36-fold increased risk of thyroid disease; however, the Nepalese investigators didn’t distinguish between hypo- and hyperthyroidism (J Am Acad Dermatol. 2020 Feb;82[2]:491-3).
Dr. Figueiredo reported having no financial conflicts regarding her study, which was without commercial support.
Anna Figueiredo, MD, declared at the virtual annual meeting of the American Academy of Dermatology.
The surprise about this finding from a large retrospective case-control study stems from the fact that the elevated risk for hypothyroidism didn’t also extend to younger women with hidradenitis suppurativa (HS) nor to patients older than 40 years of either gender, explained Dr. Figueiredo of the department of dermatology at Northwestern University, Chicago.
She presented a retrospective case-control study based on information extracted from a medical records database of more than 8 million Midwestern adults. Among nearly 141,000 dermatology patients with follow-up in the database for at least 1 year, there were 405 HS patients aged 18-40 years and 327 aged 41-89.
In an age-matched comparison with the dermatology patients without HS, the younger HS cohort was at a significant 1.52-fold increased risk for comorbid hypothyroidism. Upon further stratification by sex, only the younger men with HS were at increased risk. Those patients were at 3.95-fold greater risk for having a diagnosis of hypothyroidism than were age-matched younger male dermatology patients.
Both younger and older HS patients were at numerically increased risk for being diagnosed with hyperthyroidism; however, this difference didn’t approach statistical significance because there were so few cases: a total of just eight in the HS population across the full age spectrum.
Hidradenitis suppurativa is a chronic inflammatory disease with an estimated prevalence of up to 4% in the United States. Growing evidence suggests it is an immune-mediated disorder because the tumor necrosis factor inhibitor adalimumab (Humira) has been approved for treatment of HS.
Thyroid disease is also often autoimmune-mediated, but its relationship with HS hasn’t been extensively examined. A recent meta-analysis of five case-control studies concluded that HS was associated with a 1.36-fold increased risk of thyroid disease; however, the Nepalese investigators didn’t distinguish between hypo- and hyperthyroidism (J Am Acad Dermatol. 2020 Feb;82[2]:491-3).
Dr. Figueiredo reported having no financial conflicts regarding her study, which was without commercial support.
FROM AAD 20
Thyroid cancer overdiagnosis reaches beyond affluent regions
The rapid increase in thyroid cancer incidence that has occurred since the 1990s – considered an “epidemic of overdiagnosis,” has extended beyond high-income countries to less affluent settings, where unnecessary – and sometimes opportunistic – screening could continue to thrive.
“The impact of overdiagnosis on the increasing incidence of thyroid cancer highlighted in our report is a warning sign for countries with growing economies, where diagnostic technologies are increasingly and routinely offered, usually in exchange for payment, despite evidence that the harms far outweigh benefits,” the authors say.
“Overdiagnosis could turn healthy people into patients, and expose them to unnecessary harms and lifelong treatments,” say Mengmeng Li, PhD, of the International Agency for Research on Cancer, Lyon, France, and colleagues in their article published in Lancet Diabetes & Endocrinology.
With their previous research showing high rates of overdiagnosis in high-income countries, for this new analysis, they sought to evaluate whether similar patterns were occurring in less affluent settings.
They examined data from population-based cancer registries in 26 countries on four continents, looking at all cases of thyroid cancer reported between 1998 and 2012 in men and women aged 15 to 84 years.
A global public health problem
The results showed that while the incidence of thyroid cancer steadily increased from 1998 to 2002 and from 2008 to 2012 in all high-income countries, similar trends were also seen in less affluent nations, particularly in Belarus, China, Colombia, and Lithuania.
The increases were consistently greater among middle-aged women aged 35-64 years in all countries.
To determine what proportion of the higher incidence was overdiagnosis, the authors turned to historic age-specific thyroid cancer incidence data prior to the introduction of ultrasound and then looked at the progressive departure from that pattern, likely the result of the increased detection by ultrasound of thyroid nodules in middle-aged adults.
The results showed the proportion of thyroid cancer cases in women estimated to be attributable to overdiagnosis between 2008 and 2012 was as much as 93% in South Korea, 91% in Belarus, 87% in China, 84% in Italy and Croatia, and 83% in Slovakia and France.
Proportions attributable to overdiagnosis were lower in Denmark (66%), Norway (65%), Ireland (63%), United Kingdom (58%), Japan (55%), and Thailand (44%).
Women were much more likely to be overdiagnosed than men, with an approximate ratio of 3:1 in all countries; however, mortality and prevalence of thyroid cancer in autopsies were similar between genders.
Although researchers only looked at data up until 2012, Dr. Li said that, even in that year, “the amplitude of the phenomenon” was “already large and is increasing rapidly over time.”
Figures for periods subsequent to those assessed in the study “are likely to be higher.”
And the overdiagnosis is particularly remarkable in the context of the true risk of thyroid cancer, senior author Salvatore Vaccarella, PhD, told this news organization.
“What is surprising is the magnitude of this. Without overdiagnosis, thyroid cancer would probably still be a relatively rare cancer,” he said.
“Currently, it is the fifth most commonly diagnosed cancer in women of all ages and is third in women under 50 years of age. And the rates are still rising fast.”
“Overdiagnosis of thyroid cancer is still rapidly expanding in many high-income countries, and for the first time, we document and quantify the phenomenon also for several middle-income socioeconomically transitioning countries,” he observed. “In short, it is a global public health problem.”
Guidelines, physicians: No symptoms should mean no screening
With the implications of overdiagnosis ranging from physical, psychological in terms of the patient, and significant personal as well as societal costs, most international guidelines explicitly recommend against screening asymptomatic individuals and call for active surveillance of microcarcinomas that are detected.
The messaging appears to be making a difference.
As reported in research by American authors discussing the thyroid cancer epidemic from a 2017 perspective, the overdiagnosis situation in South Korea prompted a group of physicians there to make a high-profile public appeal in print and on television recommending against thyroid screening with ultrasound.
The result was a 35% reduction in the number of thyroidectomies performed in the subsequent year.
“This seems to be a striking example that the issue of overdiagnosis and overtreatment resonates with patients, and that public awareness can lead to changes in behavior,” say the U.S. authors of that article.
Senior author Louise Davies, MD, of the VA Outcomes Group, in White River Junction, Vt., said in an interview that the new study sheds more light on this issue.
“Even though the data only go through 2012, I think they give a nice snapshot of what’s happening across the globe with thyroid cancer incidence in countries of different levels of development,” she said.
The findings underscore that “it’s important that people are educated about the limits of medical testing and that sometimes when we see abnormalities we are truly catching a cancer early, but sometimes we’re seeing things that have been there a long time and may not change or become a problem in the future,” Dr. Davies remarked.
Important to know what size of cancers are being detected
One particular concern about overdiagnosis in middle-income countries is that the common approach of active monitoring may be more difficult in these settings, Dr. Davies added.
“In order to manage overdiagnosis, the health care systems in those countries have to think about whether they have the infrastructure for active monitoring and whether the patients will show up for the monitoring – so that’s a challenge.”
Also, she noted that the new study does not detail the size of cancers detected.
“We don’t know much about the size of the cancers being detected and whether these are truly the small asymptomatic cancers that we are worried about being overdiagnosed.”
“Probably, at least some of what we’re seeing is appropriate detection of cancers that, before there was economic development, were in fact being missed and people were dying of,” she said.
“So while overdiagnosis can be occurring, some of this represents better detection of disease overall, and that’s a good thing actually.”
The authors and Dr. Davies have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The rapid increase in thyroid cancer incidence that has occurred since the 1990s – considered an “epidemic of overdiagnosis,” has extended beyond high-income countries to less affluent settings, where unnecessary – and sometimes opportunistic – screening could continue to thrive.
“The impact of overdiagnosis on the increasing incidence of thyroid cancer highlighted in our report is a warning sign for countries with growing economies, where diagnostic technologies are increasingly and routinely offered, usually in exchange for payment, despite evidence that the harms far outweigh benefits,” the authors say.
“Overdiagnosis could turn healthy people into patients, and expose them to unnecessary harms and lifelong treatments,” say Mengmeng Li, PhD, of the International Agency for Research on Cancer, Lyon, France, and colleagues in their article published in Lancet Diabetes & Endocrinology.
With their previous research showing high rates of overdiagnosis in high-income countries, for this new analysis, they sought to evaluate whether similar patterns were occurring in less affluent settings.
They examined data from population-based cancer registries in 26 countries on four continents, looking at all cases of thyroid cancer reported between 1998 and 2012 in men and women aged 15 to 84 years.
A global public health problem
The results showed that while the incidence of thyroid cancer steadily increased from 1998 to 2002 and from 2008 to 2012 in all high-income countries, similar trends were also seen in less affluent nations, particularly in Belarus, China, Colombia, and Lithuania.
The increases were consistently greater among middle-aged women aged 35-64 years in all countries.
To determine what proportion of the higher incidence was overdiagnosis, the authors turned to historic age-specific thyroid cancer incidence data prior to the introduction of ultrasound and then looked at the progressive departure from that pattern, likely the result of the increased detection by ultrasound of thyroid nodules in middle-aged adults.
The results showed the proportion of thyroid cancer cases in women estimated to be attributable to overdiagnosis between 2008 and 2012 was as much as 93% in South Korea, 91% in Belarus, 87% in China, 84% in Italy and Croatia, and 83% in Slovakia and France.
Proportions attributable to overdiagnosis were lower in Denmark (66%), Norway (65%), Ireland (63%), United Kingdom (58%), Japan (55%), and Thailand (44%).
Women were much more likely to be overdiagnosed than men, with an approximate ratio of 3:1 in all countries; however, mortality and prevalence of thyroid cancer in autopsies were similar between genders.
Although researchers only looked at data up until 2012, Dr. Li said that, even in that year, “the amplitude of the phenomenon” was “already large and is increasing rapidly over time.”
Figures for periods subsequent to those assessed in the study “are likely to be higher.”
And the overdiagnosis is particularly remarkable in the context of the true risk of thyroid cancer, senior author Salvatore Vaccarella, PhD, told this news organization.
“What is surprising is the magnitude of this. Without overdiagnosis, thyroid cancer would probably still be a relatively rare cancer,” he said.
“Currently, it is the fifth most commonly diagnosed cancer in women of all ages and is third in women under 50 years of age. And the rates are still rising fast.”
“Overdiagnosis of thyroid cancer is still rapidly expanding in many high-income countries, and for the first time, we document and quantify the phenomenon also for several middle-income socioeconomically transitioning countries,” he observed. “In short, it is a global public health problem.”
Guidelines, physicians: No symptoms should mean no screening
With the implications of overdiagnosis ranging from physical, psychological in terms of the patient, and significant personal as well as societal costs, most international guidelines explicitly recommend against screening asymptomatic individuals and call for active surveillance of microcarcinomas that are detected.
The messaging appears to be making a difference.
As reported in research by American authors discussing the thyroid cancer epidemic from a 2017 perspective, the overdiagnosis situation in South Korea prompted a group of physicians there to make a high-profile public appeal in print and on television recommending against thyroid screening with ultrasound.
The result was a 35% reduction in the number of thyroidectomies performed in the subsequent year.
“This seems to be a striking example that the issue of overdiagnosis and overtreatment resonates with patients, and that public awareness can lead to changes in behavior,” say the U.S. authors of that article.
Senior author Louise Davies, MD, of the VA Outcomes Group, in White River Junction, Vt., said in an interview that the new study sheds more light on this issue.
“Even though the data only go through 2012, I think they give a nice snapshot of what’s happening across the globe with thyroid cancer incidence in countries of different levels of development,” she said.
The findings underscore that “it’s important that people are educated about the limits of medical testing and that sometimes when we see abnormalities we are truly catching a cancer early, but sometimes we’re seeing things that have been there a long time and may not change or become a problem in the future,” Dr. Davies remarked.
Important to know what size of cancers are being detected
One particular concern about overdiagnosis in middle-income countries is that the common approach of active monitoring may be more difficult in these settings, Dr. Davies added.
“In order to manage overdiagnosis, the health care systems in those countries have to think about whether they have the infrastructure for active monitoring and whether the patients will show up for the monitoring – so that’s a challenge.”
Also, she noted that the new study does not detail the size of cancers detected.
“We don’t know much about the size of the cancers being detected and whether these are truly the small asymptomatic cancers that we are worried about being overdiagnosed.”
“Probably, at least some of what we’re seeing is appropriate detection of cancers that, before there was economic development, were in fact being missed and people were dying of,” she said.
“So while overdiagnosis can be occurring, some of this represents better detection of disease overall, and that’s a good thing actually.”
The authors and Dr. Davies have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The rapid increase in thyroid cancer incidence that has occurred since the 1990s – considered an “epidemic of overdiagnosis,” has extended beyond high-income countries to less affluent settings, where unnecessary – and sometimes opportunistic – screening could continue to thrive.
“The impact of overdiagnosis on the increasing incidence of thyroid cancer highlighted in our report is a warning sign for countries with growing economies, where diagnostic technologies are increasingly and routinely offered, usually in exchange for payment, despite evidence that the harms far outweigh benefits,” the authors say.
“Overdiagnosis could turn healthy people into patients, and expose them to unnecessary harms and lifelong treatments,” say Mengmeng Li, PhD, of the International Agency for Research on Cancer, Lyon, France, and colleagues in their article published in Lancet Diabetes & Endocrinology.
With their previous research showing high rates of overdiagnosis in high-income countries, for this new analysis, they sought to evaluate whether similar patterns were occurring in less affluent settings.
They examined data from population-based cancer registries in 26 countries on four continents, looking at all cases of thyroid cancer reported between 1998 and 2012 in men and women aged 15 to 84 years.
A global public health problem
The results showed that while the incidence of thyroid cancer steadily increased from 1998 to 2002 and from 2008 to 2012 in all high-income countries, similar trends were also seen in less affluent nations, particularly in Belarus, China, Colombia, and Lithuania.
The increases were consistently greater among middle-aged women aged 35-64 years in all countries.
To determine what proportion of the higher incidence was overdiagnosis, the authors turned to historic age-specific thyroid cancer incidence data prior to the introduction of ultrasound and then looked at the progressive departure from that pattern, likely the result of the increased detection by ultrasound of thyroid nodules in middle-aged adults.
The results showed the proportion of thyroid cancer cases in women estimated to be attributable to overdiagnosis between 2008 and 2012 was as much as 93% in South Korea, 91% in Belarus, 87% in China, 84% in Italy and Croatia, and 83% in Slovakia and France.
Proportions attributable to overdiagnosis were lower in Denmark (66%), Norway (65%), Ireland (63%), United Kingdom (58%), Japan (55%), and Thailand (44%).
Women were much more likely to be overdiagnosed than men, with an approximate ratio of 3:1 in all countries; however, mortality and prevalence of thyroid cancer in autopsies were similar between genders.
Although researchers only looked at data up until 2012, Dr. Li said that, even in that year, “the amplitude of the phenomenon” was “already large and is increasing rapidly over time.”
Figures for periods subsequent to those assessed in the study “are likely to be higher.”
And the overdiagnosis is particularly remarkable in the context of the true risk of thyroid cancer, senior author Salvatore Vaccarella, PhD, told this news organization.
“What is surprising is the magnitude of this. Without overdiagnosis, thyroid cancer would probably still be a relatively rare cancer,” he said.
“Currently, it is the fifth most commonly diagnosed cancer in women of all ages and is third in women under 50 years of age. And the rates are still rising fast.”
“Overdiagnosis of thyroid cancer is still rapidly expanding in many high-income countries, and for the first time, we document and quantify the phenomenon also for several middle-income socioeconomically transitioning countries,” he observed. “In short, it is a global public health problem.”
Guidelines, physicians: No symptoms should mean no screening
With the implications of overdiagnosis ranging from physical, psychological in terms of the patient, and significant personal as well as societal costs, most international guidelines explicitly recommend against screening asymptomatic individuals and call for active surveillance of microcarcinomas that are detected.
The messaging appears to be making a difference.
As reported in research by American authors discussing the thyroid cancer epidemic from a 2017 perspective, the overdiagnosis situation in South Korea prompted a group of physicians there to make a high-profile public appeal in print and on television recommending against thyroid screening with ultrasound.
The result was a 35% reduction in the number of thyroidectomies performed in the subsequent year.
“This seems to be a striking example that the issue of overdiagnosis and overtreatment resonates with patients, and that public awareness can lead to changes in behavior,” say the U.S. authors of that article.
Senior author Louise Davies, MD, of the VA Outcomes Group, in White River Junction, Vt., said in an interview that the new study sheds more light on this issue.
“Even though the data only go through 2012, I think they give a nice snapshot of what’s happening across the globe with thyroid cancer incidence in countries of different levels of development,” she said.
The findings underscore that “it’s important that people are educated about the limits of medical testing and that sometimes when we see abnormalities we are truly catching a cancer early, but sometimes we’re seeing things that have been there a long time and may not change or become a problem in the future,” Dr. Davies remarked.
Important to know what size of cancers are being detected
One particular concern about overdiagnosis in middle-income countries is that the common approach of active monitoring may be more difficult in these settings, Dr. Davies added.
“In order to manage overdiagnosis, the health care systems in those countries have to think about whether they have the infrastructure for active monitoring and whether the patients will show up for the monitoring – so that’s a challenge.”
Also, she noted that the new study does not detail the size of cancers detected.
“We don’t know much about the size of the cancers being detected and whether these are truly the small asymptomatic cancers that we are worried about being overdiagnosed.”
“Probably, at least some of what we’re seeing is appropriate detection of cancers that, before there was economic development, were in fact being missed and people were dying of,” she said.
“So while overdiagnosis can be occurring, some of this represents better detection of disease overall, and that’s a good thing actually.”
The authors and Dr. Davies have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Aldosterone-driven hypertension found with unexpected frequency
Roughly 16%-22% of patients with hypertension appeared to have primary aldosteronism as the likely major cause of their elevated blood pressure, in an analysis of about 1,000 Americans, which is a much higher prevalence than previously appreciated and a finding that could potentially reorient both screening for aldosteronism and management for this subset of patients.
“Our findings show a high prevalence of unrecognized yet biochemically overt primary aldosteronism [PA] using current confirmatory diagnostic thresholds. They highlight the inadequacy of the current diagnostic approach that heavily relies on the ARR [aldosterone renin ratio] and, most important, show the existence of a pathologic continuum of nonsuppressible renin-independent aldosterone production that parallels the severity of hypertension,” wrote Jennifer M. Brown, MD, and coinvestigators in a report published in Annals of Internal Medicine on May 25. “These findings support the need to redefine primary aldosteronism from a rare and categorical disease to, instead, a common syndrome that manifests across a broad severity spectrum and may be a primary contributor to hypertension pathogenesis,” they wrote in the report.
The results, showing an underappreciated prevalence of both overt and subtler forms of aldosteronism that link with hypertension, won praise from several experts for the potential of these findings to boost the profile of excess aldosterone as a common and treatable cause of high blood pressure, but opinions on the role for the ARR as a screen to identify affected patients were more mixed.
“ARR is still the best screening approach we have” for identifying people who likely have PA, especially when the ratio threshold for finding patients who need further investigation is reduced from the traditional level of 30 ng/dL to 20 ng/dL, commented Michael Stowasser, MBBS, professor of medicine at the University of Queensland in Brisbane, Australia, and director of the Endocrine Hypertension Research Centre at Greenslopes and Princess Alexandra Hospitals in Brisbane. “I strongly recommend ARR testing in all newly diagnosed hypertensives.”
The study results “showed that PA is much more common than previously perceived, and suggest that perhaps PA in milder forms than we typically recognize contributes more to ‘essential’ hypertension than we previously thought,” said Anand Vaidya, MD, senior author of the report and director of the Center for Adrenal Disorders at Brigham and Women’s Hospital in Boston. The researchers found adjusted PA prevalence rates of 16% among 115 untreated patients with stage 1 hypertension (130-139/80-89 mm Hg), 22% among 203 patients with untreated stage 2 hypertension (at least 140/90 mm Hg), and 22% among 408 patients with treatment-resistant hypertension. All three prevalence rates were based on relatively conservative criteria that included all 726 patients with hypertension in the analysis (which also included 289 normotensive subjects) regardless of whether or not they also had low levels of serum renin. These PA prevalence rates were also based on a “conservative” definition of PA, a level of at least 12 mcg excreted in a 24-hour urine specimen.
When the researchers applied less stringent diagnostic criteria for PA or focused on the types of patients usually at highest risk for PA because of a suppressed renin level, the prevalence rates rose substantially and, in some subgroups, more than doubled. Of the 726 people with hypertension included in the analysis, 452 (62%) had suppressed renin (seated plasma renin activity < 1.0 mcg/L per hour or supine plasma renin activity < 0.6 mcg/L per hour). Within this subgroup of patients with suppressed renin, the adjusted prevalence of PA by the threshold of 24-hour urine aldosterone secretion of at least 12 mcg was 52% in those with treatment-resistant hypertension; among patients with stage 1 or 2 hypertension the adjusted prevalence rates were just slightly above the rates in the entire study group. But among patients with suppressed renin who were judged to have PA by a more liberal definition of at least 10 mcg in a 24-hour urine sample, the adjusted prevalence rates were 27% among untreated stage 1 hypertensives, 40% among untreated stage 2 patients, and 58% among treatment-resistant patients, the report showed.
A role for subtler forms of aldosteronism
Defining PA as at least 12 mcg secreted in a 24-hour urine collection “is relatively arbitrary, and our findings show that it bisects a continuous distribution. How we should redefine PA is also arbitrary, but step one is to recognize that many people have milder forms of PA” that could have an important effect on blood pressure, Dr. Vaidya said in an interview.
“This is the very first study to show that aldosterone may be contributing to the hypertensive process even though it is not severe enough to be diagnosed as PA according to current criteria,” said Robert M. Carey, MD, a cardiovascular endocrinologist and professor of medicine at the University of Virginia in Charlottesville and a coauthor on the new report. “More patients than we have ever known have an aldosterone component to their hypertension,” Dr. Carey said in an interview.
The new report on the prevalence of unrecognized PA in hypertensive patients “is a game changer,” wrote John W. Funder, MD, professor of medicine at Monash University in Clayton, Australia, in an editorial published along with the new report. In the editorial, he synthesized the new findings with results from prior reports to estimate that excess aldosteronism could play a clinically meaningful role in close to half of patients with hypertension, although Dr. Stowasser called this an “overestimate.” The new results also showed that “the single spot measurement of plasma aldosterone concentration, which clinicians have used for decades to screen for primary aldosteronism, is not merely useless but actually misleading. The authors cautioned readers about the uncertain representativeness of the study population to the U.S. population, but I believe that the findings are generalizable to the United States and elsewhere,” Dr. Funder wrote. “The central problem is that plasma aldosterone concentration is a very poor index of total daily aldosterone secretion. A single morning spot measurement of plasma aldosterone cannot take into account ultradian variation in aldosterone secretion.”
The importance of finding excess aldosterone
Identifying patients with hypertension and PA, as well as hypertensives with excess aldosterone production that may not meet the traditional definition of PA, is especially important because they are excellent candidates for two forms of targeted and very effective treatments that have a reliable and substantial impact on lowering blood pressure in these patients. One treatment is unilateral adrenal gland removal in patients who produce excess aldosterone because of benign adenomas in one adrenal gland, which accounts for “approximately 30%” of patients with PA. “Patients with suspected PA should have an opportunity to find out whether they have a unilateral variety and chance for surgical cure,” said Dr. Stowasser in an interview. “Patients with PA do far better in terms of blood pressure control, prevention of cardiovascular complications, and quality of life if they are treated specifically, either medically or particularly by surgery.”
The specific medical treatment he cited refers to one of the mineralocorticoid receptor antagonist (MRA) drugs, spironolactone and eplerenone (Inspra), because mineralocorticoid receptor blockade directly short-circuits the path by which aldosterone increases blood pressure. “We’re advocating earlier use of MRAs” for hypertensive patients identified with excess aldosterone production, said Dr. Carey. He noted that alternative, nonsteroidal MRAs, such as finerenone, have shown promise for efficacy levels similar to what spironolactone provides but without as many adverse effects because of greater receptor specificity. Finerenone and other nonsteroidal MRAs are all currently investigational. Spironolactone and eplerenone both cause hyperkalemia, although treatment with potassium binding agents can blunt the risk this poses. Spironolactone also causes bothersome adverse effects in men, including impotence and gynecomastia because of its action on androgen receptors, effects that diminished with eplerenone, but eplerenone is not as effective as spironolactone, Dr. Carey said.
Study details
The new study ran a post hoc analysis on data collected in five independent studies run at centers in four U.S. locations: Birmingham, Ala.; Boston; Charlottesville, Va.; and Salt Lake City. The studies included a total of 1,846 adults, mostly patients with hypertension of varying severity but also several hundred normotensive people. Data on 24-hour sodium excretion during an oral sodium suppression test were available for all participants, and the researchers excluded 831 people with an “inadequate” sodium balance of less than 190 mmol based on this metric, leaving a study population of 1,015. The researchers acknowledged the limitation that the study participants were not representative of the U.S. population.
The analysis included 289 normotensive people not on any blood pressure–lowering medications, and 239 fit the definition of having suppressed renin. The adjusted prevalence of aldosteronism at the level of at least 12 mcg excreted in a 24-hour urine specimen was 11% among all 289 normotensive subjects and 12% among the 239 with suppressed renin. When the definition of aldosteronism loosened to at least 10 mcg excreted during 24 hours the adjusted prevalence of excess aldosterone among normotensives increased to 19% among the entire group and 20% among those with suppressed renin. This finding may have identified a primordial phase of nascent hypertension that needs further study but may eventually provide a new scenario for intervention. “If a normotensive person has compliant arteries and healthy kidneys they can handle the excess salt and volume load of PA,” but when compensatory mechanisms start falling short through aging or other deteriorations, then blood pressure starts to rise, suggested Dr. Vaidya.
Whom to screen for aldosteronism and how
While several experts agreed these findings added to an existing and growing literature showing that PA is common and needs greater diagnostic attention, they differed on what this may mean for the specifics of screening and diagnosis, especially at the primary care level.
“Our results showed more explicitly that excess aldosterone exists on a broad severity spectrum and can’t be regarded as a categorical diagnosis that a patient either has or does not have. The hard part is figuring out where we should begin interventions,” said Dr. Vaidya.
“This publication will hopefully increase clinician awareness of this common and treatable form of hypertension. All people with high blood pressure should be tested at least once for PA,” commented William F. Young Jr., MD, professor and chair of endocrinology at the Mayo Clinic in Rochester, Minn. “Diagnosis of PA provides clinicians with a unique opportunity in medicine, to provide either surgical cure or targeted pharmacotherapy. It’s been frustrating to me to see patients not tested for PA when first diagnosed with hypertension, but only after they developed irreversible chronic kidney disease,” he said in an interview. Dr. Young cited statistics that only about 2% of patients diagnosed with treatment-resistant hypertension are assessed for PA, and only about 3% of patients with hypertension and concomitant hyperkalemia. “Primary care physicians don’t think about PA and don’t test for PA,” he lamented.
The new study “is very convincing, and confirms and extends the findings of several other groups that previously reported the high prevalence of PA among patients with hypertension,” commented Dr. Stowasser. Despite this accumulating evidence, uptake of testing for PA, usually starting with spot measurement of renin and aldosterone to obtain an ARR, has “remained dismally low” among primary care and specialist physicians in Australia, the United States, Europe, and elsewhere, he added.
One stumbling block may be the complexity, or at least perceived complexity, of screening by an ARR and follow-up steps as recommended in a 2016 guideline issued by the Endocrine Society and endorsed by several international medical societies including the American Heart Association, Dr. Carey said. Dr. Funder chaired the task force that wrote the 2016 Endocrine Society PA guideline, and the eight-member task force included Dr. Carey, Dr. Stowasser, and Dr. Young.
The new study highlights what its authors cited as a limitation of the ARR for screening. When set at the frequently used ratio threshold of 30 ng/dL/ng/mL per hour to identify likely cases of PA, the crude PA prevalence rates corresponding to this threshold were 4% in treated stage 1 hypertensives, 10% in treated stage 2 patients, and 7% in those with resistant hypertension, substantially below the adjusted PA prevalence rates calculated by applying different criteria for excess aldosterone. In addition to missing clinically meaningful cases, the ARR may also underachieve at a functional level, Dr. Carey suggested.
“We note the difficulty with point assessment of ARR, but that’s what we have at the moment. We’ll look for other ways to identify patients with excessive aldosterone production,” he said. “We need to design a [diagnostic] pathway that’s easily doable by primary care physicians. Right now it’s pretty complicated. Part of the reason why primary care physicians often don’t screen for PA is the pathway is too complicated. We need to simplify it.”
In his editorial, Dr. Funder wrote that “much of the present guideline needs to be jettisoned, and radically reconstructed recommendations should be developed.”
One answer may be to apply a less stringent ARR threshold for further work-up. Dr. Stowasser’s program in Brisbane, as well as some other groups worldwide, use an ARR of at least 20 ng/dL as an indication of possible PA. “If you lower the cutoff to 20 [ng/dL], and ignore the plasma aldosterone level, then the ARR should pick up the great majority of patients with PA,” he said.
Another controversial aspect is whether aldosterone detection should be screened by 24-hour urine collection or by spot testing. In his editorial, Dr. Funder called spot testing “useless” and “misleading,” but Dr. Vaidya acknowledged that the 24-hour collection used in his current study is “not practical” for widespread use. Despite that, the Mayo Clinic in Rochester has focused on 24-hour urine collected “for more than 4 decades,” said Dr. Young, even though “a morning blood sample remains a simple screening test” that will catch “more than 95% of patients with PA” when combined with a plasma aldosterone threshold of 10 ng/dL. Dr. Stowasser noted that “patients don’t like” 24-hour collection, and not infrequently muck up collection” by forgetting to collect their entire 1-day output. Regardless of its shortcomings, 24-hour urine has the advantage of greater precision and accuracy than spot measurement, and using it on newly diagnosed hypertensive patients who also show renin suppression may be a viable approach, Dr. Carey suggested.
Regardless of exactly how guidelines for assessing aldosterone in hypertensive patients change, prospects seem ripe for some sort of revision and for greater participation and buy-in by primary care physicians than in the past. Dr. Carey, who also served as vice-chair of the American College of Cardiology and American Heart Association Task Force that wrote the most current U.S. guideline for managing hypertension, said it was too soon to revise that document, but the time had come to revise the Endocrine Society’s 2016 guideline for diagnosing and treating PA and to hash out the revision “in partnership” with one or more primary care societies. He also highlighted that publishing the current study in a high-profile primary care journal was an intentional effort to reach a large segment of the primary care community.
The new report “has the potential to change the current state of inertia” over wider PA diagnosis and targeted treatment “by being published in a widely read, major international journal,” commented Dr. Stowasser.
Dr. Vaidya has been a consultant to Catalys Pacific, Corcept Therapeutics, HRA Pharma, Orphagen, and Selenity Therapeutics. None of the other report coauthors had commercial disclosures, including Dr. Carey. Dr. Funder, Dr. Stowasser, and Dr. Young had no disclosures.
SOURCE: Brown JM et al. Ann Int Med. 2020 May 25. doi: 10.7326/M20-0065.
Roughly 16%-22% of patients with hypertension appeared to have primary aldosteronism as the likely major cause of their elevated blood pressure, in an analysis of about 1,000 Americans, which is a much higher prevalence than previously appreciated and a finding that could potentially reorient both screening for aldosteronism and management for this subset of patients.
“Our findings show a high prevalence of unrecognized yet biochemically overt primary aldosteronism [PA] using current confirmatory diagnostic thresholds. They highlight the inadequacy of the current diagnostic approach that heavily relies on the ARR [aldosterone renin ratio] and, most important, show the existence of a pathologic continuum of nonsuppressible renin-independent aldosterone production that parallels the severity of hypertension,” wrote Jennifer M. Brown, MD, and coinvestigators in a report published in Annals of Internal Medicine on May 25. “These findings support the need to redefine primary aldosteronism from a rare and categorical disease to, instead, a common syndrome that manifests across a broad severity spectrum and may be a primary contributor to hypertension pathogenesis,” they wrote in the report.
The results, showing an underappreciated prevalence of both overt and subtler forms of aldosteronism that link with hypertension, won praise from several experts for the potential of these findings to boost the profile of excess aldosterone as a common and treatable cause of high blood pressure, but opinions on the role for the ARR as a screen to identify affected patients were more mixed.
“ARR is still the best screening approach we have” for identifying people who likely have PA, especially when the ratio threshold for finding patients who need further investigation is reduced from the traditional level of 30 ng/dL to 20 ng/dL, commented Michael Stowasser, MBBS, professor of medicine at the University of Queensland in Brisbane, Australia, and director of the Endocrine Hypertension Research Centre at Greenslopes and Princess Alexandra Hospitals in Brisbane. “I strongly recommend ARR testing in all newly diagnosed hypertensives.”
The study results “showed that PA is much more common than previously perceived, and suggest that perhaps PA in milder forms than we typically recognize contributes more to ‘essential’ hypertension than we previously thought,” said Anand Vaidya, MD, senior author of the report and director of the Center for Adrenal Disorders at Brigham and Women’s Hospital in Boston. The researchers found adjusted PA prevalence rates of 16% among 115 untreated patients with stage 1 hypertension (130-139/80-89 mm Hg), 22% among 203 patients with untreated stage 2 hypertension (at least 140/90 mm Hg), and 22% among 408 patients with treatment-resistant hypertension. All three prevalence rates were based on relatively conservative criteria that included all 726 patients with hypertension in the analysis (which also included 289 normotensive subjects) regardless of whether or not they also had low levels of serum renin. These PA prevalence rates were also based on a “conservative” definition of PA, a level of at least 12 mcg excreted in a 24-hour urine specimen.
When the researchers applied less stringent diagnostic criteria for PA or focused on the types of patients usually at highest risk for PA because of a suppressed renin level, the prevalence rates rose substantially and, in some subgroups, more than doubled. Of the 726 people with hypertension included in the analysis, 452 (62%) had suppressed renin (seated plasma renin activity < 1.0 mcg/L per hour or supine plasma renin activity < 0.6 mcg/L per hour). Within this subgroup of patients with suppressed renin, the adjusted prevalence of PA by the threshold of 24-hour urine aldosterone secretion of at least 12 mcg was 52% in those with treatment-resistant hypertension; among patients with stage 1 or 2 hypertension the adjusted prevalence rates were just slightly above the rates in the entire study group. But among patients with suppressed renin who were judged to have PA by a more liberal definition of at least 10 mcg in a 24-hour urine sample, the adjusted prevalence rates were 27% among untreated stage 1 hypertensives, 40% among untreated stage 2 patients, and 58% among treatment-resistant patients, the report showed.
A role for subtler forms of aldosteronism
Defining PA as at least 12 mcg secreted in a 24-hour urine collection “is relatively arbitrary, and our findings show that it bisects a continuous distribution. How we should redefine PA is also arbitrary, but step one is to recognize that many people have milder forms of PA” that could have an important effect on blood pressure, Dr. Vaidya said in an interview.
“This is the very first study to show that aldosterone may be contributing to the hypertensive process even though it is not severe enough to be diagnosed as PA according to current criteria,” said Robert M. Carey, MD, a cardiovascular endocrinologist and professor of medicine at the University of Virginia in Charlottesville and a coauthor on the new report. “More patients than we have ever known have an aldosterone component to their hypertension,” Dr. Carey said in an interview.
The new report on the prevalence of unrecognized PA in hypertensive patients “is a game changer,” wrote John W. Funder, MD, professor of medicine at Monash University in Clayton, Australia, in an editorial published along with the new report. In the editorial, he synthesized the new findings with results from prior reports to estimate that excess aldosteronism could play a clinically meaningful role in close to half of patients with hypertension, although Dr. Stowasser called this an “overestimate.” The new results also showed that “the single spot measurement of plasma aldosterone concentration, which clinicians have used for decades to screen for primary aldosteronism, is not merely useless but actually misleading. The authors cautioned readers about the uncertain representativeness of the study population to the U.S. population, but I believe that the findings are generalizable to the United States and elsewhere,” Dr. Funder wrote. “The central problem is that plasma aldosterone concentration is a very poor index of total daily aldosterone secretion. A single morning spot measurement of plasma aldosterone cannot take into account ultradian variation in aldosterone secretion.”
The importance of finding excess aldosterone
Identifying patients with hypertension and PA, as well as hypertensives with excess aldosterone production that may not meet the traditional definition of PA, is especially important because they are excellent candidates for two forms of targeted and very effective treatments that have a reliable and substantial impact on lowering blood pressure in these patients. One treatment is unilateral adrenal gland removal in patients who produce excess aldosterone because of benign adenomas in one adrenal gland, which accounts for “approximately 30%” of patients with PA. “Patients with suspected PA should have an opportunity to find out whether they have a unilateral variety and chance for surgical cure,” said Dr. Stowasser in an interview. “Patients with PA do far better in terms of blood pressure control, prevention of cardiovascular complications, and quality of life if they are treated specifically, either medically or particularly by surgery.”
The specific medical treatment he cited refers to one of the mineralocorticoid receptor antagonist (MRA) drugs, spironolactone and eplerenone (Inspra), because mineralocorticoid receptor blockade directly short-circuits the path by which aldosterone increases blood pressure. “We’re advocating earlier use of MRAs” for hypertensive patients identified with excess aldosterone production, said Dr. Carey. He noted that alternative, nonsteroidal MRAs, such as finerenone, have shown promise for efficacy levels similar to what spironolactone provides but without as many adverse effects because of greater receptor specificity. Finerenone and other nonsteroidal MRAs are all currently investigational. Spironolactone and eplerenone both cause hyperkalemia, although treatment with potassium binding agents can blunt the risk this poses. Spironolactone also causes bothersome adverse effects in men, including impotence and gynecomastia because of its action on androgen receptors, effects that diminished with eplerenone, but eplerenone is not as effective as spironolactone, Dr. Carey said.
Study details
The new study ran a post hoc analysis on data collected in five independent studies run at centers in four U.S. locations: Birmingham, Ala.; Boston; Charlottesville, Va.; and Salt Lake City. The studies included a total of 1,846 adults, mostly patients with hypertension of varying severity but also several hundred normotensive people. Data on 24-hour sodium excretion during an oral sodium suppression test were available for all participants, and the researchers excluded 831 people with an “inadequate” sodium balance of less than 190 mmol based on this metric, leaving a study population of 1,015. The researchers acknowledged the limitation that the study participants were not representative of the U.S. population.
The analysis included 289 normotensive people not on any blood pressure–lowering medications, and 239 fit the definition of having suppressed renin. The adjusted prevalence of aldosteronism at the level of at least 12 mcg excreted in a 24-hour urine specimen was 11% among all 289 normotensive subjects and 12% among the 239 with suppressed renin. When the definition of aldosteronism loosened to at least 10 mcg excreted during 24 hours the adjusted prevalence of excess aldosterone among normotensives increased to 19% among the entire group and 20% among those with suppressed renin. This finding may have identified a primordial phase of nascent hypertension that needs further study but may eventually provide a new scenario for intervention. “If a normotensive person has compliant arteries and healthy kidneys they can handle the excess salt and volume load of PA,” but when compensatory mechanisms start falling short through aging or other deteriorations, then blood pressure starts to rise, suggested Dr. Vaidya.
Whom to screen for aldosteronism and how
While several experts agreed these findings added to an existing and growing literature showing that PA is common and needs greater diagnostic attention, they differed on what this may mean for the specifics of screening and diagnosis, especially at the primary care level.
“Our results showed more explicitly that excess aldosterone exists on a broad severity spectrum and can’t be regarded as a categorical diagnosis that a patient either has or does not have. The hard part is figuring out where we should begin interventions,” said Dr. Vaidya.
“This publication will hopefully increase clinician awareness of this common and treatable form of hypertension. All people with high blood pressure should be tested at least once for PA,” commented William F. Young Jr., MD, professor and chair of endocrinology at the Mayo Clinic in Rochester, Minn. “Diagnosis of PA provides clinicians with a unique opportunity in medicine, to provide either surgical cure or targeted pharmacotherapy. It’s been frustrating to me to see patients not tested for PA when first diagnosed with hypertension, but only after they developed irreversible chronic kidney disease,” he said in an interview. Dr. Young cited statistics that only about 2% of patients diagnosed with treatment-resistant hypertension are assessed for PA, and only about 3% of patients with hypertension and concomitant hyperkalemia. “Primary care physicians don’t think about PA and don’t test for PA,” he lamented.
The new study “is very convincing, and confirms and extends the findings of several other groups that previously reported the high prevalence of PA among patients with hypertension,” commented Dr. Stowasser. Despite this accumulating evidence, uptake of testing for PA, usually starting with spot measurement of renin and aldosterone to obtain an ARR, has “remained dismally low” among primary care and specialist physicians in Australia, the United States, Europe, and elsewhere, he added.
One stumbling block may be the complexity, or at least perceived complexity, of screening by an ARR and follow-up steps as recommended in a 2016 guideline issued by the Endocrine Society and endorsed by several international medical societies including the American Heart Association, Dr. Carey said. Dr. Funder chaired the task force that wrote the 2016 Endocrine Society PA guideline, and the eight-member task force included Dr. Carey, Dr. Stowasser, and Dr. Young.
The new study highlights what its authors cited as a limitation of the ARR for screening. When set at the frequently used ratio threshold of 30 ng/dL/ng/mL per hour to identify likely cases of PA, the crude PA prevalence rates corresponding to this threshold were 4% in treated stage 1 hypertensives, 10% in treated stage 2 patients, and 7% in those with resistant hypertension, substantially below the adjusted PA prevalence rates calculated by applying different criteria for excess aldosterone. In addition to missing clinically meaningful cases, the ARR may also underachieve at a functional level, Dr. Carey suggested.
“We note the difficulty with point assessment of ARR, but that’s what we have at the moment. We’ll look for other ways to identify patients with excessive aldosterone production,” he said. “We need to design a [diagnostic] pathway that’s easily doable by primary care physicians. Right now it’s pretty complicated. Part of the reason why primary care physicians often don’t screen for PA is the pathway is too complicated. We need to simplify it.”
In his editorial, Dr. Funder wrote that “much of the present guideline needs to be jettisoned, and radically reconstructed recommendations should be developed.”
One answer may be to apply a less stringent ARR threshold for further work-up. Dr. Stowasser’s program in Brisbane, as well as some other groups worldwide, use an ARR of at least 20 ng/dL as an indication of possible PA. “If you lower the cutoff to 20 [ng/dL], and ignore the plasma aldosterone level, then the ARR should pick up the great majority of patients with PA,” he said.
Another controversial aspect is whether aldosterone detection should be screened by 24-hour urine collection or by spot testing. In his editorial, Dr. Funder called spot testing “useless” and “misleading,” but Dr. Vaidya acknowledged that the 24-hour collection used in his current study is “not practical” for widespread use. Despite that, the Mayo Clinic in Rochester has focused on 24-hour urine collected “for more than 4 decades,” said Dr. Young, even though “a morning blood sample remains a simple screening test” that will catch “more than 95% of patients with PA” when combined with a plasma aldosterone threshold of 10 ng/dL. Dr. Stowasser noted that “patients don’t like” 24-hour collection, and not infrequently muck up collection” by forgetting to collect their entire 1-day output. Regardless of its shortcomings, 24-hour urine has the advantage of greater precision and accuracy than spot measurement, and using it on newly diagnosed hypertensive patients who also show renin suppression may be a viable approach, Dr. Carey suggested.
Regardless of exactly how guidelines for assessing aldosterone in hypertensive patients change, prospects seem ripe for some sort of revision and for greater participation and buy-in by primary care physicians than in the past. Dr. Carey, who also served as vice-chair of the American College of Cardiology and American Heart Association Task Force that wrote the most current U.S. guideline for managing hypertension, said it was too soon to revise that document, but the time had come to revise the Endocrine Society’s 2016 guideline for diagnosing and treating PA and to hash out the revision “in partnership” with one or more primary care societies. He also highlighted that publishing the current study in a high-profile primary care journal was an intentional effort to reach a large segment of the primary care community.
The new report “has the potential to change the current state of inertia” over wider PA diagnosis and targeted treatment “by being published in a widely read, major international journal,” commented Dr. Stowasser.
Dr. Vaidya has been a consultant to Catalys Pacific, Corcept Therapeutics, HRA Pharma, Orphagen, and Selenity Therapeutics. None of the other report coauthors had commercial disclosures, including Dr. Carey. Dr. Funder, Dr. Stowasser, and Dr. Young had no disclosures.
SOURCE: Brown JM et al. Ann Int Med. 2020 May 25. doi: 10.7326/M20-0065.
Roughly 16%-22% of patients with hypertension appeared to have primary aldosteronism as the likely major cause of their elevated blood pressure, in an analysis of about 1,000 Americans, which is a much higher prevalence than previously appreciated and a finding that could potentially reorient both screening for aldosteronism and management for this subset of patients.
“Our findings show a high prevalence of unrecognized yet biochemically overt primary aldosteronism [PA] using current confirmatory diagnostic thresholds. They highlight the inadequacy of the current diagnostic approach that heavily relies on the ARR [aldosterone renin ratio] and, most important, show the existence of a pathologic continuum of nonsuppressible renin-independent aldosterone production that parallels the severity of hypertension,” wrote Jennifer M. Brown, MD, and coinvestigators in a report published in Annals of Internal Medicine on May 25. “These findings support the need to redefine primary aldosteronism from a rare and categorical disease to, instead, a common syndrome that manifests across a broad severity spectrum and may be a primary contributor to hypertension pathogenesis,” they wrote in the report.
The results, showing an underappreciated prevalence of both overt and subtler forms of aldosteronism that link with hypertension, won praise from several experts for the potential of these findings to boost the profile of excess aldosterone as a common and treatable cause of high blood pressure, but opinions on the role for the ARR as a screen to identify affected patients were more mixed.
“ARR is still the best screening approach we have” for identifying people who likely have PA, especially when the ratio threshold for finding patients who need further investigation is reduced from the traditional level of 30 ng/dL to 20 ng/dL, commented Michael Stowasser, MBBS, professor of medicine at the University of Queensland in Brisbane, Australia, and director of the Endocrine Hypertension Research Centre at Greenslopes and Princess Alexandra Hospitals in Brisbane. “I strongly recommend ARR testing in all newly diagnosed hypertensives.”
The study results “showed that PA is much more common than previously perceived, and suggest that perhaps PA in milder forms than we typically recognize contributes more to ‘essential’ hypertension than we previously thought,” said Anand Vaidya, MD, senior author of the report and director of the Center for Adrenal Disorders at Brigham and Women’s Hospital in Boston. The researchers found adjusted PA prevalence rates of 16% among 115 untreated patients with stage 1 hypertension (130-139/80-89 mm Hg), 22% among 203 patients with untreated stage 2 hypertension (at least 140/90 mm Hg), and 22% among 408 patients with treatment-resistant hypertension. All three prevalence rates were based on relatively conservative criteria that included all 726 patients with hypertension in the analysis (which also included 289 normotensive subjects) regardless of whether or not they also had low levels of serum renin. These PA prevalence rates were also based on a “conservative” definition of PA, a level of at least 12 mcg excreted in a 24-hour urine specimen.
When the researchers applied less stringent diagnostic criteria for PA or focused on the types of patients usually at highest risk for PA because of a suppressed renin level, the prevalence rates rose substantially and, in some subgroups, more than doubled. Of the 726 people with hypertension included in the analysis, 452 (62%) had suppressed renin (seated plasma renin activity < 1.0 mcg/L per hour or supine plasma renin activity < 0.6 mcg/L per hour). Within this subgroup of patients with suppressed renin, the adjusted prevalence of PA by the threshold of 24-hour urine aldosterone secretion of at least 12 mcg was 52% in those with treatment-resistant hypertension; among patients with stage 1 or 2 hypertension the adjusted prevalence rates were just slightly above the rates in the entire study group. But among patients with suppressed renin who were judged to have PA by a more liberal definition of at least 10 mcg in a 24-hour urine sample, the adjusted prevalence rates were 27% among untreated stage 1 hypertensives, 40% among untreated stage 2 patients, and 58% among treatment-resistant patients, the report showed.
A role for subtler forms of aldosteronism
Defining PA as at least 12 mcg secreted in a 24-hour urine collection “is relatively arbitrary, and our findings show that it bisects a continuous distribution. How we should redefine PA is also arbitrary, but step one is to recognize that many people have milder forms of PA” that could have an important effect on blood pressure, Dr. Vaidya said in an interview.
“This is the very first study to show that aldosterone may be contributing to the hypertensive process even though it is not severe enough to be diagnosed as PA according to current criteria,” said Robert M. Carey, MD, a cardiovascular endocrinologist and professor of medicine at the University of Virginia in Charlottesville and a coauthor on the new report. “More patients than we have ever known have an aldosterone component to their hypertension,” Dr. Carey said in an interview.
The new report on the prevalence of unrecognized PA in hypertensive patients “is a game changer,” wrote John W. Funder, MD, professor of medicine at Monash University in Clayton, Australia, in an editorial published along with the new report. In the editorial, he synthesized the new findings with results from prior reports to estimate that excess aldosteronism could play a clinically meaningful role in close to half of patients with hypertension, although Dr. Stowasser called this an “overestimate.” The new results also showed that “the single spot measurement of plasma aldosterone concentration, which clinicians have used for decades to screen for primary aldosteronism, is not merely useless but actually misleading. The authors cautioned readers about the uncertain representativeness of the study population to the U.S. population, but I believe that the findings are generalizable to the United States and elsewhere,” Dr. Funder wrote. “The central problem is that plasma aldosterone concentration is a very poor index of total daily aldosterone secretion. A single morning spot measurement of plasma aldosterone cannot take into account ultradian variation in aldosterone secretion.”
The importance of finding excess aldosterone
Identifying patients with hypertension and PA, as well as hypertensives with excess aldosterone production that may not meet the traditional definition of PA, is especially important because they are excellent candidates for two forms of targeted and very effective treatments that have a reliable and substantial impact on lowering blood pressure in these patients. One treatment is unilateral adrenal gland removal in patients who produce excess aldosterone because of benign adenomas in one adrenal gland, which accounts for “approximately 30%” of patients with PA. “Patients with suspected PA should have an opportunity to find out whether they have a unilateral variety and chance for surgical cure,” said Dr. Stowasser in an interview. “Patients with PA do far better in terms of blood pressure control, prevention of cardiovascular complications, and quality of life if they are treated specifically, either medically or particularly by surgery.”
The specific medical treatment he cited refers to one of the mineralocorticoid receptor antagonist (MRA) drugs, spironolactone and eplerenone (Inspra), because mineralocorticoid receptor blockade directly short-circuits the path by which aldosterone increases blood pressure. “We’re advocating earlier use of MRAs” for hypertensive patients identified with excess aldosterone production, said Dr. Carey. He noted that alternative, nonsteroidal MRAs, such as finerenone, have shown promise for efficacy levels similar to what spironolactone provides but without as many adverse effects because of greater receptor specificity. Finerenone and other nonsteroidal MRAs are all currently investigational. Spironolactone and eplerenone both cause hyperkalemia, although treatment with potassium binding agents can blunt the risk this poses. Spironolactone also causes bothersome adverse effects in men, including impotence and gynecomastia because of its action on androgen receptors, effects that diminished with eplerenone, but eplerenone is not as effective as spironolactone, Dr. Carey said.
Study details
The new study ran a post hoc analysis on data collected in five independent studies run at centers in four U.S. locations: Birmingham, Ala.; Boston; Charlottesville, Va.; and Salt Lake City. The studies included a total of 1,846 adults, mostly patients with hypertension of varying severity but also several hundred normotensive people. Data on 24-hour sodium excretion during an oral sodium suppression test were available for all participants, and the researchers excluded 831 people with an “inadequate” sodium balance of less than 190 mmol based on this metric, leaving a study population of 1,015. The researchers acknowledged the limitation that the study participants were not representative of the U.S. population.
The analysis included 289 normotensive people not on any blood pressure–lowering medications, and 239 fit the definition of having suppressed renin. The adjusted prevalence of aldosteronism at the level of at least 12 mcg excreted in a 24-hour urine specimen was 11% among all 289 normotensive subjects and 12% among the 239 with suppressed renin. When the definition of aldosteronism loosened to at least 10 mcg excreted during 24 hours the adjusted prevalence of excess aldosterone among normotensives increased to 19% among the entire group and 20% among those with suppressed renin. This finding may have identified a primordial phase of nascent hypertension that needs further study but may eventually provide a new scenario for intervention. “If a normotensive person has compliant arteries and healthy kidneys they can handle the excess salt and volume load of PA,” but when compensatory mechanisms start falling short through aging or other deteriorations, then blood pressure starts to rise, suggested Dr. Vaidya.
Whom to screen for aldosteronism and how
While several experts agreed these findings added to an existing and growing literature showing that PA is common and needs greater diagnostic attention, they differed on what this may mean for the specifics of screening and diagnosis, especially at the primary care level.
“Our results showed more explicitly that excess aldosterone exists on a broad severity spectrum and can’t be regarded as a categorical diagnosis that a patient either has or does not have. The hard part is figuring out where we should begin interventions,” said Dr. Vaidya.
“This publication will hopefully increase clinician awareness of this common and treatable form of hypertension. All people with high blood pressure should be tested at least once for PA,” commented William F. Young Jr., MD, professor and chair of endocrinology at the Mayo Clinic in Rochester, Minn. “Diagnosis of PA provides clinicians with a unique opportunity in medicine, to provide either surgical cure or targeted pharmacotherapy. It’s been frustrating to me to see patients not tested for PA when first diagnosed with hypertension, but only after they developed irreversible chronic kidney disease,” he said in an interview. Dr. Young cited statistics that only about 2% of patients diagnosed with treatment-resistant hypertension are assessed for PA, and only about 3% of patients with hypertension and concomitant hyperkalemia. “Primary care physicians don’t think about PA and don’t test for PA,” he lamented.
The new study “is very convincing, and confirms and extends the findings of several other groups that previously reported the high prevalence of PA among patients with hypertension,” commented Dr. Stowasser. Despite this accumulating evidence, uptake of testing for PA, usually starting with spot measurement of renin and aldosterone to obtain an ARR, has “remained dismally low” among primary care and specialist physicians in Australia, the United States, Europe, and elsewhere, he added.
One stumbling block may be the complexity, or at least perceived complexity, of screening by an ARR and follow-up steps as recommended in a 2016 guideline issued by the Endocrine Society and endorsed by several international medical societies including the American Heart Association, Dr. Carey said. Dr. Funder chaired the task force that wrote the 2016 Endocrine Society PA guideline, and the eight-member task force included Dr. Carey, Dr. Stowasser, and Dr. Young.
The new study highlights what its authors cited as a limitation of the ARR for screening. When set at the frequently used ratio threshold of 30 ng/dL/ng/mL per hour to identify likely cases of PA, the crude PA prevalence rates corresponding to this threshold were 4% in treated stage 1 hypertensives, 10% in treated stage 2 patients, and 7% in those with resistant hypertension, substantially below the adjusted PA prevalence rates calculated by applying different criteria for excess aldosterone. In addition to missing clinically meaningful cases, the ARR may also underachieve at a functional level, Dr. Carey suggested.
“We note the difficulty with point assessment of ARR, but that’s what we have at the moment. We’ll look for other ways to identify patients with excessive aldosterone production,” he said. “We need to design a [diagnostic] pathway that’s easily doable by primary care physicians. Right now it’s pretty complicated. Part of the reason why primary care physicians often don’t screen for PA is the pathway is too complicated. We need to simplify it.”
In his editorial, Dr. Funder wrote that “much of the present guideline needs to be jettisoned, and radically reconstructed recommendations should be developed.”
One answer may be to apply a less stringent ARR threshold for further work-up. Dr. Stowasser’s program in Brisbane, as well as some other groups worldwide, use an ARR of at least 20 ng/dL as an indication of possible PA. “If you lower the cutoff to 20 [ng/dL], and ignore the plasma aldosterone level, then the ARR should pick up the great majority of patients with PA,” he said.
Another controversial aspect is whether aldosterone detection should be screened by 24-hour urine collection or by spot testing. In his editorial, Dr. Funder called spot testing “useless” and “misleading,” but Dr. Vaidya acknowledged that the 24-hour collection used in his current study is “not practical” for widespread use. Despite that, the Mayo Clinic in Rochester has focused on 24-hour urine collected “for more than 4 decades,” said Dr. Young, even though “a morning blood sample remains a simple screening test” that will catch “more than 95% of patients with PA” when combined with a plasma aldosterone threshold of 10 ng/dL. Dr. Stowasser noted that “patients don’t like” 24-hour collection, and not infrequently muck up collection” by forgetting to collect their entire 1-day output. Regardless of its shortcomings, 24-hour urine has the advantage of greater precision and accuracy than spot measurement, and using it on newly diagnosed hypertensive patients who also show renin suppression may be a viable approach, Dr. Carey suggested.
Regardless of exactly how guidelines for assessing aldosterone in hypertensive patients change, prospects seem ripe for some sort of revision and for greater participation and buy-in by primary care physicians than in the past. Dr. Carey, who also served as vice-chair of the American College of Cardiology and American Heart Association Task Force that wrote the most current U.S. guideline for managing hypertension, said it was too soon to revise that document, but the time had come to revise the Endocrine Society’s 2016 guideline for diagnosing and treating PA and to hash out the revision “in partnership” with one or more primary care societies. He also highlighted that publishing the current study in a high-profile primary care journal was an intentional effort to reach a large segment of the primary care community.
The new report “has the potential to change the current state of inertia” over wider PA diagnosis and targeted treatment “by being published in a widely read, major international journal,” commented Dr. Stowasser.
Dr. Vaidya has been a consultant to Catalys Pacific, Corcept Therapeutics, HRA Pharma, Orphagen, and Selenity Therapeutics. None of the other report coauthors had commercial disclosures, including Dr. Carey. Dr. Funder, Dr. Stowasser, and Dr. Young had no disclosures.
SOURCE: Brown JM et al. Ann Int Med. 2020 May 25. doi: 10.7326/M20-0065.
REPORTING FROM ANNALS OF INTERNAL MEDICINE



