User login
Implementing shared decision making in labor and delivery: TeamBirth is a model for person-centered birthing care
CASE The TeamBirth experience: Making a difference
“At a community hospital in Washington where we had implemented TeamBirth (a labor and delivery shared decision making model), a patient, her partner, a labor and delivery nurse, and myself (an ObGyn) were making a plan for the patient’s induction of labor admission. I asked the patient, a 29-year-old (G2P1001), how we could improve her care in relation to her first birth. Her answer was simple: I want to be treated with respect. Her partner went on to describe their past experience in which the provider was inappropriately texting while in between the patient’s knees during delivery. Our team had the opportunity to undo some of the trauma from her first birth. That’s what I like about TeamBirth. It gives every patient the opportunity, regardless of their background, to define safety and participate in their care experience.”
–Angela Chien, MD, Obstetrician and Quality Improvement leader, Washington
Unfortunately, disrespect and mistreatment are far from an anomaly in the obstetrics setting. In a systematic review of respectful maternity care, the World Health Organization delineated 7 dimensions of maternal mistreatment: physical abuse, sexual abuse, verbal abuse, stigma and discrimination, failure to meet professional standards of care, poor rapport between women and providers, and poor conditions and constraints presented by the health system.1 In 2019, the Giving Voice to Mothers study showed that 17% of birthing people in the United States reported experiencing 1 or more types of maternal mistreatment.2 Rates of mistreatment were disproportionately greater in populations of color, hospital-based births, and among those with social, economic, or health challenges.2 It is well known that Black and African American and American Indian and Alaska Native populations experience the rare events of severe maternal morbidity and mortality more frequently than their White counterparts; the disproportionate burden of mistreatment is lesser known and far more common.
Overlooking the longitudinal harm of a negative birth experience has cascading impact. While an empowering perinatal experience can foster preventive screening and management of chronic disease, a poor experience conversely can seed mistrust at an individual, generational, and community level.
The patient quality enterprise is beginning to shift attention toward maternal experience with the development of PREMs (patient-reported experience measures), PROMs (patient-reported outcome measures), and novel validated scales that assess autonomy and trust.3 Development of a maternal Consumer Assessment of Healthcare Providers and Systems (CAHPS) survey on childbirth is forthcoming.4 Of course, continuing to prioritize physical safety through initiatives on blood pressure monitoring and severe maternal morbidity and mortality remains paramount. Yet emotional and psychological safety also must be recognized as essential pillars of patient safety. Transgressions related to autonomy and dignity, as well as racism, sexism, classicism, and ableism, should be treated as “adverse and never events.”5
How the TeamBirth model works
Shared decision making (SDM) is cited in medical pedagogy as the solution to respectfullyrecognizing social context, integrating subjective experience, and honoring patient autonomy.6 The onus has always been on individual clinicians to exercise SDM. A new practice model, TeamBirth, embeds SDM into the culture and workflow. It offers a behavioral framework to mitigate implicit bias and operationalizes SDM tools, such that every patient is an empowered participant in their care.
TeamBirth was created through Ariadne Labs’ Delivery Decisions Initiative, a research and social impact program that designs, tests, and scales transformative, systems-level solutions that promote quality, equity, and dignity in childbirth. By the end of 2023, TeamBirth will be implemented in more than 100 hospitals across the United States, cumulatively touching over 200,000 lives. (For more information on the TeamBirth model, view the “Why TeamBirth” video at: https://www.youtube.com/watch?v=EoVrSaGk7gc.)
The tenets of TeamBirth are enacted through a patient-facing, shared whiteboard or dry-erase planning board in the labor room (FIGURE 1). Research has demonstrated how dry-erase boards in clinical settings can support safety and dignity in care, especially to improve patient-provider communication, teamwork, and patient satisfaction.7,8 The planning board is initially filled out by a clinical team member and is updated during team “huddles” throughout labor.

Huddles are care plan discussions with the full care team (the patient, nurse, doula and/or other support person(s), delivering provider, and interpreter or social worker as needed). At a minimum, huddles occur on admission, with changes to the clinical course and care plan, and at the request of any team member. Huddles can transpire through in-person, virtual, or phone communication.9 The concept builds on interdisciplinary and patient-centered rounding and establishes a communication system that is suited to the dynamic environment and amplified patient autonomy unique to labor and delivery. Dr. Bob Barbieri, a steadfast leader and champion of TeamBirth implementation at Brigham and Women’s Hospital in Boston (and the Editor in Chief of OBG M
Continue to: Patient response to TeamBirth is positive...
Patient response to TeamBirth is positive
Patients and providers alike have endorsed TeamBirth. In initial pilot testing across 4 sites, 99% of all patients surveyed “definitely” or “somewhat” had the role they wanted in making decisions about their labor.9
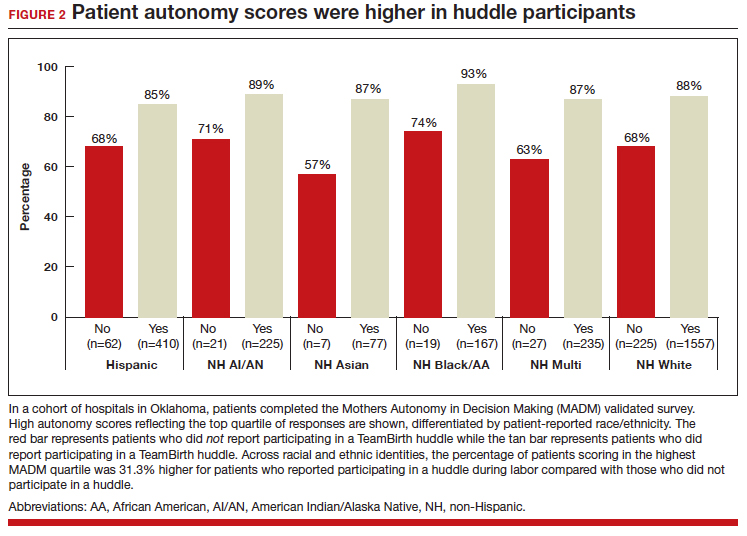
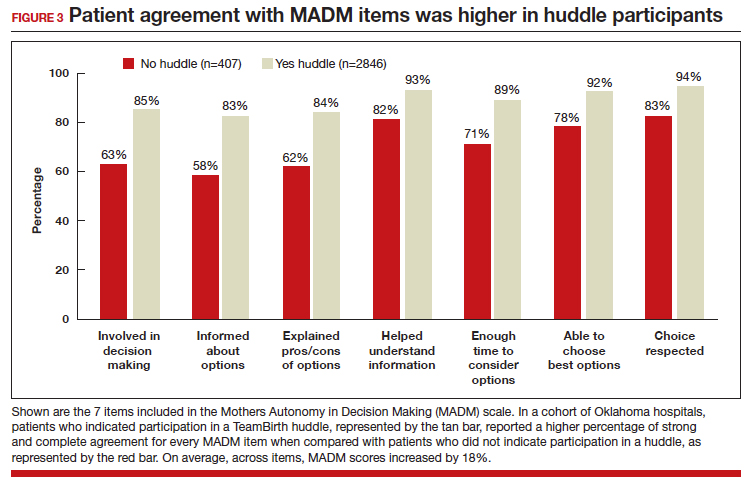
In partnership with the Oklahoma Perinatal Quality Improvement Collaborative (OPQIC), the impact of TeamBirth was assessed in a statewide patient cohort (n = 3,121) using the validated Mothers Autonomy in Decision Making (MADM) scale created by the Birth Place Lab at the University of British Columbia. The percentage of patients who scored in the highest MADM quartile was 31.3% higher for patients who indicated participation in a huddle during labor compared with those who did not participate in a huddle. This trend held across all racial and ethnic groups: For example, 93% of non-Hispanic Black/African American patients who had a TeamBirth huddle reported high autonomy, a nearly 20 percentage point increase from those without a huddle (FIGURE 2). Similarly, a higher percentage of agreement was observed across all 7 items in the MADM scale for patients who reported a TeamBirth huddle (FIGURE 3). TeamBirth’s effect has been observed across surveys and multiple validated metrics.
Data collection related to TeamBirth continues to be ongoing, with reported values retrieved on July 14, 2023. Rigorous review of patient-reported outcomes is forthcoming, and assessing impact on clinical outcomes, such as NTSV (nulliparous, term, singleton vertex) cesarean delivery rates and severe maternal morbidity, is on the horizon.
Qualitative survey responses reinforce how patients value TeamBirth and appreciate huddles and whiteboards.
Continue to: Patient testimonials...
Patient testimonials
The following testimonials were obtained from a TeamBirth survey that patients in participating Massachusetts hospitals completed in the postpartum unit prior to discharge.
According to one patient, “TeamBirth is great, feels like all obstacles are covered by multiple people with many talents, expertise. Feels like mom is part of the process, much different than my delivery 2 years ago when I felt like things were decided for me/I was ‘told’ what we were doing and questioned if I felt uneasy about it…. We felt safe and like all things were covered no matter what may happen.”
Another patient, also at a Massachusetts hospital, offered these comments about TeamBirth: “The entire staff was very genuine and my experience the best it could be. They deserve updated whiteboards in every room. I found them to be very useful.”
The clinician perspective
To be certain, clinician workflow must be a consideration for any practice change. The feasibility, acceptability, and safety of the TeamBirth model to clinicians was validated through a study at 4 community hospitals across the United States in which TeamBirth had been implemented in the 8 months prior.9
The clinician response rate was an impressive 78%. Ninety percent of clinicians, including physicians, midwives, and nurses, indicated that they would “definitely” (68%) or “probably” (22%) recommend TeamBirth for use in other labor and delivery units. None of the clinicians surveyed (n = 375) reported that TeamBirth negatively impacted care delivery.9
Obstetricians also provided qualitative commentary, noting that, while at times huddling infringed on efficiency, it also enhanced staff fulfillment. An obstetrician at a Massachusetts hospital observed, “Overall I think [TeamBirth is] helpful in slowing us down a little bit to really make sure that we’re providing the human part of the care, like the communication, and not just the medical care. And I think most providers value the human part and the communication. You know, we all think most providers value good communication with the patients, but when you’re in the middle of running around doing a bunch of stuff, you don’t always remember to prioritize it. And I think that at the end of the day…when you know you’ve communicated well with your patients, you end up feeling better about what you’re doing.”
As with most cross-sectional survey studies, selection bias remains an important caveat; patients and providers may decide to complete or not complete voluntary surveys based on particularly positive or negative experiences.
Metrics aside, obstetricians have an ethical duty to provide dignified and safe care, both physically and psychologically. Collectively, as a specialty, we share the responsibility to mitigate maternal mistreatment. As individuals, we can prevent perpetuation of birth trauma and foster healing and empowerment, one patient at a time, by employing tenets of TeamBirth.
To connect with Delivery Decisions Initiative, visit our website: https://www.ariadnelabs.org/deliverydecisions-initiative/ or contact: deliverydecisions@ ariadnelabs.org
Steps for implementing the TeamBirth model
To incorporate TeamBirth into your practice:
- Make patients the “team captain” and center them as the primary decision maker.
- Elicit patient preferences and subjective experiences to develop a collaborative plan on admission and when changes occur in clinical status.
- Round with and utilize the expertise of the full care team—nurse and midwife or obstetrician, as well as support person(s) and/or doula, learners, interpreter, and social worker as applicable.
- Ensure that the patient knows the names and roles of the care team members and provide updates at shift change.
- If your birthing rooms have a whiteboard, use it to keep the patient and team informed of the plan.
- Delineate status updates by maternal condition, fetal condition, and labor progress.
- Provide explicit permission for patients to call for a team huddle at any time and encourage support from their support people and/or doula. ●
This project is supported by:
- The Oklahoma Department of Health as part of the State Maternal Health Innovation Program Grant, Maternal and Child Health Bureau, Health Resources and Services Administration, Department of Health and Human Services.
- The Health Resources and Services Administration (HRSA) of the U.S. Department of Health and Human Services (HHS) as part of an award to the Oklahoma State Department of Health. The contents are those of the author(s) and do not necessarily represent the official views of, nor an endorsement, by HRSA, HHS, or the U.S. Government. For more information, please visit HRSA.gov.
- The Health Resources and Services Administration (HRSA) of the U.S. Department of Health and Human Services (HHS) under grant T76MC00001 and entitled Training Grant in Maternal and Child Health.
- Point32 Health’s Clinical Innovation Fund.
Data included in this article was collected and analyzed in partnership with the Oklahoma Perinatal Quality Improvement Collaborative, Department of OB/GYN, University of Oklahoma Health Sciences Center, Oklahoma City.
- Bohren MA, Vogel JP, Hunter EC, et al. The mistreatment of women during childbirth in health facilities globally: a mixedmethods systematic review. PLoS Med. 2015;12:e100184. doi:10.1371/journal.pmed.1001847
- Vedam S, Stoll K, Taiwo TK, et al. The Giving Voice to Mothers study: inequity and mistreatment during pregnancy and childbirth in the United States. Reprod Health. 2019;16. doi:10.1186/s12978-019-0729-2
- Kemmerer A, Alteras T. Evolving the maternal health quality measurement enterprise to support the communitybased maternity model. Maternal Health Hub. April 25, 2023. Accessed September 13, 2023. https:/www .maternalhealthhub.org
- Potential CAHPS survey to assess patients’ prenatal and childbirth care experiences. Agency for Healthcare Research and Quality. March 2023. Accessed September 13, 2023. https://www.ahrq.gov/news/cahps-comments-sought.html
- Lyndon A, Davis DA, Sharma AE, et al. Emotional safety is patient safety. BMJ Qual Saf. 2023;32:369-372. doi:10.1136 /bmjqs-2022-015573
- American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 819. Informed consent and shared decision making in obstetrics and gynecology. Obstet Gynecol. 2021;137:e34-e41. Accessed September 13, 2023. https://www.acog.org/clinical/clinical-guidance /committee-opinion/articles/2021/02/informed -consent-and-shared-decision-making-in-obstetrics-and -gynecology
- Goyal AA, Tur K, Mann J, et al. Do bedside visual tools improve patient and caregiver satisfaction? A systematic review of the literature. J Hosp Med. 2017;12:930-936. doi:10.12788 /jhm.2871
- Sehgal NL, Green A, Vidyarthi AR, et al. Patient whiteboards as a communication tool in the hospital setting: a survey of practices and recommendations. J Hosp Med. 2010;5:234-239. doi:10.1002/jhm.638
- Weiseth A, Plough A, Aggarwal R, et al. Improving communication and teamwork during labor: a feasibility, acceptability, and safety study. Birth. 2022:49:637-647. doi:10.1111/birt.12630
CASE The TeamBirth experience: Making a difference
“At a community hospital in Washington where we had implemented TeamBirth (a labor and delivery shared decision making model), a patient, her partner, a labor and delivery nurse, and myself (an ObGyn) were making a plan for the patient’s induction of labor admission. I asked the patient, a 29-year-old (G2P1001), how we could improve her care in relation to her first birth. Her answer was simple: I want to be treated with respect. Her partner went on to describe their past experience in which the provider was inappropriately texting while in between the patient’s knees during delivery. Our team had the opportunity to undo some of the trauma from her first birth. That’s what I like about TeamBirth. It gives every patient the opportunity, regardless of their background, to define safety and participate in their care experience.”
–Angela Chien, MD, Obstetrician and Quality Improvement leader, Washington
Unfortunately, disrespect and mistreatment are far from an anomaly in the obstetrics setting. In a systematic review of respectful maternity care, the World Health Organization delineated 7 dimensions of maternal mistreatment: physical abuse, sexual abuse, verbal abuse, stigma and discrimination, failure to meet professional standards of care, poor rapport between women and providers, and poor conditions and constraints presented by the health system.1 In 2019, the Giving Voice to Mothers study showed that 17% of birthing people in the United States reported experiencing 1 or more types of maternal mistreatment.2 Rates of mistreatment were disproportionately greater in populations of color, hospital-based births, and among those with social, economic, or health challenges.2 It is well known that Black and African American and American Indian and Alaska Native populations experience the rare events of severe maternal morbidity and mortality more frequently than their White counterparts; the disproportionate burden of mistreatment is lesser known and far more common.
Overlooking the longitudinal harm of a negative birth experience has cascading impact. While an empowering perinatal experience can foster preventive screening and management of chronic disease, a poor experience conversely can seed mistrust at an individual, generational, and community level.
The patient quality enterprise is beginning to shift attention toward maternal experience with the development of PREMs (patient-reported experience measures), PROMs (patient-reported outcome measures), and novel validated scales that assess autonomy and trust.3 Development of a maternal Consumer Assessment of Healthcare Providers and Systems (CAHPS) survey on childbirth is forthcoming.4 Of course, continuing to prioritize physical safety through initiatives on blood pressure monitoring and severe maternal morbidity and mortality remains paramount. Yet emotional and psychological safety also must be recognized as essential pillars of patient safety. Transgressions related to autonomy and dignity, as well as racism, sexism, classicism, and ableism, should be treated as “adverse and never events.”5
How the TeamBirth model works
Shared decision making (SDM) is cited in medical pedagogy as the solution to respectfullyrecognizing social context, integrating subjective experience, and honoring patient autonomy.6 The onus has always been on individual clinicians to exercise SDM. A new practice model, TeamBirth, embeds SDM into the culture and workflow. It offers a behavioral framework to mitigate implicit bias and operationalizes SDM tools, such that every patient is an empowered participant in their care.
TeamBirth was created through Ariadne Labs’ Delivery Decisions Initiative, a research and social impact program that designs, tests, and scales transformative, systems-level solutions that promote quality, equity, and dignity in childbirth. By the end of 2023, TeamBirth will be implemented in more than 100 hospitals across the United States, cumulatively touching over 200,000 lives. (For more information on the TeamBirth model, view the “Why TeamBirth” video at: https://www.youtube.com/watch?v=EoVrSaGk7gc.)
The tenets of TeamBirth are enacted through a patient-facing, shared whiteboard or dry-erase planning board in the labor room (FIGURE 1). Research has demonstrated how dry-erase boards in clinical settings can support safety and dignity in care, especially to improve patient-provider communication, teamwork, and patient satisfaction.7,8 The planning board is initially filled out by a clinical team member and is updated during team “huddles” throughout labor.

Huddles are care plan discussions with the full care team (the patient, nurse, doula and/or other support person(s), delivering provider, and interpreter or social worker as needed). At a minimum, huddles occur on admission, with changes to the clinical course and care plan, and at the request of any team member. Huddles can transpire through in-person, virtual, or phone communication.9 The concept builds on interdisciplinary and patient-centered rounding and establishes a communication system that is suited to the dynamic environment and amplified patient autonomy unique to labor and delivery. Dr. Bob Barbieri, a steadfast leader and champion of TeamBirth implementation at Brigham and Women’s Hospital in Boston (and the Editor in Chief of OBG M
Continue to: Patient response to TeamBirth is positive...
Patient response to TeamBirth is positive
Patients and providers alike have endorsed TeamBirth. In initial pilot testing across 4 sites, 99% of all patients surveyed “definitely” or “somewhat” had the role they wanted in making decisions about their labor.9
In partnership with the Oklahoma Perinatal Quality Improvement Collaborative (OPQIC), the impact of TeamBirth was assessed in a statewide patient cohort (n = 3,121) using the validated Mothers Autonomy in Decision Making (MADM) scale created by the Birth Place Lab at the University of British Columbia. The percentage of patients who scored in the highest MADM quartile was 31.3% higher for patients who indicated participation in a huddle during labor compared with those who did not participate in a huddle. This trend held across all racial and ethnic groups: For example, 93% of non-Hispanic Black/African American patients who had a TeamBirth huddle reported high autonomy, a nearly 20 percentage point increase from those without a huddle (FIGURE 2). Similarly, a higher percentage of agreement was observed across all 7 items in the MADM scale for patients who reported a TeamBirth huddle (FIGURE 3). TeamBirth’s effect has been observed across surveys and multiple validated metrics.


Data collection related to TeamBirth continues to be ongoing, with reported values retrieved on July 14, 2023. Rigorous review of patient-reported outcomes is forthcoming, and assessing impact on clinical outcomes, such as NTSV (nulliparous, term, singleton vertex) cesarean delivery rates and severe maternal morbidity, is on the horizon.
Qualitative survey responses reinforce how patients value TeamBirth and appreciate huddles and whiteboards.
Continue to: Patient testimonials...
Patient testimonials
The following testimonials were obtained from a TeamBirth survey that patients in participating Massachusetts hospitals completed in the postpartum unit prior to discharge.
According to one patient, “TeamBirth is great, feels like all obstacles are covered by multiple people with many talents, expertise. Feels like mom is part of the process, much different than my delivery 2 years ago when I felt like things were decided for me/I was ‘told’ what we were doing and questioned if I felt uneasy about it…. We felt safe and like all things were covered no matter what may happen.”
Another patient, also at a Massachusetts hospital, offered these comments about TeamBirth: “The entire staff was very genuine and my experience the best it could be. They deserve updated whiteboards in every room. I found them to be very useful.”
The clinician perspective
To be certain, clinician workflow must be a consideration for any practice change. The feasibility, acceptability, and safety of the TeamBirth model to clinicians was validated through a study at 4 community hospitals across the United States in which TeamBirth had been implemented in the 8 months prior.9
The clinician response rate was an impressive 78%. Ninety percent of clinicians, including physicians, midwives, and nurses, indicated that they would “definitely” (68%) or “probably” (22%) recommend TeamBirth for use in other labor and delivery units. None of the clinicians surveyed (n = 375) reported that TeamBirth negatively impacted care delivery.9
Obstetricians also provided qualitative commentary, noting that, while at times huddling infringed on efficiency, it also enhanced staff fulfillment. An obstetrician at a Massachusetts hospital observed, “Overall I think [TeamBirth is] helpful in slowing us down a little bit to really make sure that we’re providing the human part of the care, like the communication, and not just the medical care. And I think most providers value the human part and the communication. You know, we all think most providers value good communication with the patients, but when you’re in the middle of running around doing a bunch of stuff, you don’t always remember to prioritize it. And I think that at the end of the day…when you know you’ve communicated well with your patients, you end up feeling better about what you’re doing.”
As with most cross-sectional survey studies, selection bias remains an important caveat; patients and providers may decide to complete or not complete voluntary surveys based on particularly positive or negative experiences.
Metrics aside, obstetricians have an ethical duty to provide dignified and safe care, both physically and psychologically. Collectively, as a specialty, we share the responsibility to mitigate maternal mistreatment. As individuals, we can prevent perpetuation of birth trauma and foster healing and empowerment, one patient at a time, by employing tenets of TeamBirth.
To connect with Delivery Decisions Initiative, visit our website: https://www.ariadnelabs.org/deliverydecisions-initiative/ or contact: deliverydecisions@ ariadnelabs.org
Steps for implementing the TeamBirth model
To incorporate TeamBirth into your practice:
- Make patients the “team captain” and center them as the primary decision maker.
- Elicit patient preferences and subjective experiences to develop a collaborative plan on admission and when changes occur in clinical status.
- Round with and utilize the expertise of the full care team—nurse and midwife or obstetrician, as well as support person(s) and/or doula, learners, interpreter, and social worker as applicable.
- Ensure that the patient knows the names and roles of the care team members and provide updates at shift change.
- If your birthing rooms have a whiteboard, use it to keep the patient and team informed of the plan.
- Delineate status updates by maternal condition, fetal condition, and labor progress.
- Provide explicit permission for patients to call for a team huddle at any time and encourage support from their support people and/or doula. ●
This project is supported by:
- The Oklahoma Department of Health as part of the State Maternal Health Innovation Program Grant, Maternal and Child Health Bureau, Health Resources and Services Administration, Department of Health and Human Services.
- The Health Resources and Services Administration (HRSA) of the U.S. Department of Health and Human Services (HHS) as part of an award to the Oklahoma State Department of Health. The contents are those of the author(s) and do not necessarily represent the official views of, nor an endorsement, by HRSA, HHS, or the U.S. Government. For more information, please visit HRSA.gov.
- The Health Resources and Services Administration (HRSA) of the U.S. Department of Health and Human Services (HHS) under grant T76MC00001 and entitled Training Grant in Maternal and Child Health.
- Point32 Health’s Clinical Innovation Fund.
Data included in this article was collected and analyzed in partnership with the Oklahoma Perinatal Quality Improvement Collaborative, Department of OB/GYN, University of Oklahoma Health Sciences Center, Oklahoma City.
CASE The TeamBirth experience: Making a difference
“At a community hospital in Washington where we had implemented TeamBirth (a labor and delivery shared decision making model), a patient, her partner, a labor and delivery nurse, and myself (an ObGyn) were making a plan for the patient’s induction of labor admission. I asked the patient, a 29-year-old (G2P1001), how we could improve her care in relation to her first birth. Her answer was simple: I want to be treated with respect. Her partner went on to describe their past experience in which the provider was inappropriately texting while in between the patient’s knees during delivery. Our team had the opportunity to undo some of the trauma from her first birth. That’s what I like about TeamBirth. It gives every patient the opportunity, regardless of their background, to define safety and participate in their care experience.”
–Angela Chien, MD, Obstetrician and Quality Improvement leader, Washington
Unfortunately, disrespect and mistreatment are far from an anomaly in the obstetrics setting. In a systematic review of respectful maternity care, the World Health Organization delineated 7 dimensions of maternal mistreatment: physical abuse, sexual abuse, verbal abuse, stigma and discrimination, failure to meet professional standards of care, poor rapport between women and providers, and poor conditions and constraints presented by the health system.1 In 2019, the Giving Voice to Mothers study showed that 17% of birthing people in the United States reported experiencing 1 or more types of maternal mistreatment.2 Rates of mistreatment were disproportionately greater in populations of color, hospital-based births, and among those with social, economic, or health challenges.2 It is well known that Black and African American and American Indian and Alaska Native populations experience the rare events of severe maternal morbidity and mortality more frequently than their White counterparts; the disproportionate burden of mistreatment is lesser known and far more common.
Overlooking the longitudinal harm of a negative birth experience has cascading impact. While an empowering perinatal experience can foster preventive screening and management of chronic disease, a poor experience conversely can seed mistrust at an individual, generational, and community level.
The patient quality enterprise is beginning to shift attention toward maternal experience with the development of PREMs (patient-reported experience measures), PROMs (patient-reported outcome measures), and novel validated scales that assess autonomy and trust.3 Development of a maternal Consumer Assessment of Healthcare Providers and Systems (CAHPS) survey on childbirth is forthcoming.4 Of course, continuing to prioritize physical safety through initiatives on blood pressure monitoring and severe maternal morbidity and mortality remains paramount. Yet emotional and psychological safety also must be recognized as essential pillars of patient safety. Transgressions related to autonomy and dignity, as well as racism, sexism, classicism, and ableism, should be treated as “adverse and never events.”5
How the TeamBirth model works
Shared decision making (SDM) is cited in medical pedagogy as the solution to respectfullyrecognizing social context, integrating subjective experience, and honoring patient autonomy.6 The onus has always been on individual clinicians to exercise SDM. A new practice model, TeamBirth, embeds SDM into the culture and workflow. It offers a behavioral framework to mitigate implicit bias and operationalizes SDM tools, such that every patient is an empowered participant in their care.
TeamBirth was created through Ariadne Labs’ Delivery Decisions Initiative, a research and social impact program that designs, tests, and scales transformative, systems-level solutions that promote quality, equity, and dignity in childbirth. By the end of 2023, TeamBirth will be implemented in more than 100 hospitals across the United States, cumulatively touching over 200,000 lives. (For more information on the TeamBirth model, view the “Why TeamBirth” video at: https://www.youtube.com/watch?v=EoVrSaGk7gc.)
The tenets of TeamBirth are enacted through a patient-facing, shared whiteboard or dry-erase planning board in the labor room (FIGURE 1). Research has demonstrated how dry-erase boards in clinical settings can support safety and dignity in care, especially to improve patient-provider communication, teamwork, and patient satisfaction.7,8 The planning board is initially filled out by a clinical team member and is updated during team “huddles” throughout labor.

Huddles are care plan discussions with the full care team (the patient, nurse, doula and/or other support person(s), delivering provider, and interpreter or social worker as needed). At a minimum, huddles occur on admission, with changes to the clinical course and care plan, and at the request of any team member. Huddles can transpire through in-person, virtual, or phone communication.9 The concept builds on interdisciplinary and patient-centered rounding and establishes a communication system that is suited to the dynamic environment and amplified patient autonomy unique to labor and delivery. Dr. Bob Barbieri, a steadfast leader and champion of TeamBirth implementation at Brigham and Women’s Hospital in Boston (and the Editor in Chief of OBG M
Continue to: Patient response to TeamBirth is positive...
Patient response to TeamBirth is positive
Patients and providers alike have endorsed TeamBirth. In initial pilot testing across 4 sites, 99% of all patients surveyed “definitely” or “somewhat” had the role they wanted in making decisions about their labor.9
In partnership with the Oklahoma Perinatal Quality Improvement Collaborative (OPQIC), the impact of TeamBirth was assessed in a statewide patient cohort (n = 3,121) using the validated Mothers Autonomy in Decision Making (MADM) scale created by the Birth Place Lab at the University of British Columbia. The percentage of patients who scored in the highest MADM quartile was 31.3% higher for patients who indicated participation in a huddle during labor compared with those who did not participate in a huddle. This trend held across all racial and ethnic groups: For example, 93% of non-Hispanic Black/African American patients who had a TeamBirth huddle reported high autonomy, a nearly 20 percentage point increase from those without a huddle (FIGURE 2). Similarly, a higher percentage of agreement was observed across all 7 items in the MADM scale for patients who reported a TeamBirth huddle (FIGURE 3). TeamBirth’s effect has been observed across surveys and multiple validated metrics.


Data collection related to TeamBirth continues to be ongoing, with reported values retrieved on July 14, 2023. Rigorous review of patient-reported outcomes is forthcoming, and assessing impact on clinical outcomes, such as NTSV (nulliparous, term, singleton vertex) cesarean delivery rates and severe maternal morbidity, is on the horizon.
Qualitative survey responses reinforce how patients value TeamBirth and appreciate huddles and whiteboards.
Continue to: Patient testimonials...
Patient testimonials
The following testimonials were obtained from a TeamBirth survey that patients in participating Massachusetts hospitals completed in the postpartum unit prior to discharge.
According to one patient, “TeamBirth is great, feels like all obstacles are covered by multiple people with many talents, expertise. Feels like mom is part of the process, much different than my delivery 2 years ago when I felt like things were decided for me/I was ‘told’ what we were doing and questioned if I felt uneasy about it…. We felt safe and like all things were covered no matter what may happen.”
Another patient, also at a Massachusetts hospital, offered these comments about TeamBirth: “The entire staff was very genuine and my experience the best it could be. They deserve updated whiteboards in every room. I found them to be very useful.”
The clinician perspective
To be certain, clinician workflow must be a consideration for any practice change. The feasibility, acceptability, and safety of the TeamBirth model to clinicians was validated through a study at 4 community hospitals across the United States in which TeamBirth had been implemented in the 8 months prior.9
The clinician response rate was an impressive 78%. Ninety percent of clinicians, including physicians, midwives, and nurses, indicated that they would “definitely” (68%) or “probably” (22%) recommend TeamBirth for use in other labor and delivery units. None of the clinicians surveyed (n = 375) reported that TeamBirth negatively impacted care delivery.9
Obstetricians also provided qualitative commentary, noting that, while at times huddling infringed on efficiency, it also enhanced staff fulfillment. An obstetrician at a Massachusetts hospital observed, “Overall I think [TeamBirth is] helpful in slowing us down a little bit to really make sure that we’re providing the human part of the care, like the communication, and not just the medical care. And I think most providers value the human part and the communication. You know, we all think most providers value good communication with the patients, but when you’re in the middle of running around doing a bunch of stuff, you don’t always remember to prioritize it. And I think that at the end of the day…when you know you’ve communicated well with your patients, you end up feeling better about what you’re doing.”
As with most cross-sectional survey studies, selection bias remains an important caveat; patients and providers may decide to complete or not complete voluntary surveys based on particularly positive or negative experiences.
Metrics aside, obstetricians have an ethical duty to provide dignified and safe care, both physically and psychologically. Collectively, as a specialty, we share the responsibility to mitigate maternal mistreatment. As individuals, we can prevent perpetuation of birth trauma and foster healing and empowerment, one patient at a time, by employing tenets of TeamBirth.
To connect with Delivery Decisions Initiative, visit our website: https://www.ariadnelabs.org/deliverydecisions-initiative/ or contact: deliverydecisions@ ariadnelabs.org
Steps for implementing the TeamBirth model
To incorporate TeamBirth into your practice:
- Make patients the “team captain” and center them as the primary decision maker.
- Elicit patient preferences and subjective experiences to develop a collaborative plan on admission and when changes occur in clinical status.
- Round with and utilize the expertise of the full care team—nurse and midwife or obstetrician, as well as support person(s) and/or doula, learners, interpreter, and social worker as applicable.
- Ensure that the patient knows the names and roles of the care team members and provide updates at shift change.
- If your birthing rooms have a whiteboard, use it to keep the patient and team informed of the plan.
- Delineate status updates by maternal condition, fetal condition, and labor progress.
- Provide explicit permission for patients to call for a team huddle at any time and encourage support from their support people and/or doula. ●
This project is supported by:
- The Oklahoma Department of Health as part of the State Maternal Health Innovation Program Grant, Maternal and Child Health Bureau, Health Resources and Services Administration, Department of Health and Human Services.
- The Health Resources and Services Administration (HRSA) of the U.S. Department of Health and Human Services (HHS) as part of an award to the Oklahoma State Department of Health. The contents are those of the author(s) and do not necessarily represent the official views of, nor an endorsement, by HRSA, HHS, or the U.S. Government. For more information, please visit HRSA.gov.
- The Health Resources and Services Administration (HRSA) of the U.S. Department of Health and Human Services (HHS) under grant T76MC00001 and entitled Training Grant in Maternal and Child Health.
- Point32 Health’s Clinical Innovation Fund.
Data included in this article was collected and analyzed in partnership with the Oklahoma Perinatal Quality Improvement Collaborative, Department of OB/GYN, University of Oklahoma Health Sciences Center, Oklahoma City.
- Bohren MA, Vogel JP, Hunter EC, et al. The mistreatment of women during childbirth in health facilities globally: a mixedmethods systematic review. PLoS Med. 2015;12:e100184. doi:10.1371/journal.pmed.1001847
- Vedam S, Stoll K, Taiwo TK, et al. The Giving Voice to Mothers study: inequity and mistreatment during pregnancy and childbirth in the United States. Reprod Health. 2019;16. doi:10.1186/s12978-019-0729-2
- Kemmerer A, Alteras T. Evolving the maternal health quality measurement enterprise to support the communitybased maternity model. Maternal Health Hub. April 25, 2023. Accessed September 13, 2023. https:/www .maternalhealthhub.org
- Potential CAHPS survey to assess patients’ prenatal and childbirth care experiences. Agency for Healthcare Research and Quality. March 2023. Accessed September 13, 2023. https://www.ahrq.gov/news/cahps-comments-sought.html
- Lyndon A, Davis DA, Sharma AE, et al. Emotional safety is patient safety. BMJ Qual Saf. 2023;32:369-372. doi:10.1136 /bmjqs-2022-015573
- American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 819. Informed consent and shared decision making in obstetrics and gynecology. Obstet Gynecol. 2021;137:e34-e41. Accessed September 13, 2023. https://www.acog.org/clinical/clinical-guidance /committee-opinion/articles/2021/02/informed -consent-and-shared-decision-making-in-obstetrics-and -gynecology
- Goyal AA, Tur K, Mann J, et al. Do bedside visual tools improve patient and caregiver satisfaction? A systematic review of the literature. J Hosp Med. 2017;12:930-936. doi:10.12788 /jhm.2871
- Sehgal NL, Green A, Vidyarthi AR, et al. Patient whiteboards as a communication tool in the hospital setting: a survey of practices and recommendations. J Hosp Med. 2010;5:234-239. doi:10.1002/jhm.638
- Weiseth A, Plough A, Aggarwal R, et al. Improving communication and teamwork during labor: a feasibility, acceptability, and safety study. Birth. 2022:49:637-647. doi:10.1111/birt.12630
- Bohren MA, Vogel JP, Hunter EC, et al. The mistreatment of women during childbirth in health facilities globally: a mixedmethods systematic review. PLoS Med. 2015;12:e100184. doi:10.1371/journal.pmed.1001847
- Vedam S, Stoll K, Taiwo TK, et al. The Giving Voice to Mothers study: inequity and mistreatment during pregnancy and childbirth in the United States. Reprod Health. 2019;16. doi:10.1186/s12978-019-0729-2
- Kemmerer A, Alteras T. Evolving the maternal health quality measurement enterprise to support the communitybased maternity model. Maternal Health Hub. April 25, 2023. Accessed September 13, 2023. https:/www .maternalhealthhub.org
- Potential CAHPS survey to assess patients’ prenatal and childbirth care experiences. Agency for Healthcare Research and Quality. March 2023. Accessed September 13, 2023. https://www.ahrq.gov/news/cahps-comments-sought.html
- Lyndon A, Davis DA, Sharma AE, et al. Emotional safety is patient safety. BMJ Qual Saf. 2023;32:369-372. doi:10.1136 /bmjqs-2022-015573
- American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 819. Informed consent and shared decision making in obstetrics and gynecology. Obstet Gynecol. 2021;137:e34-e41. Accessed September 13, 2023. https://www.acog.org/clinical/clinical-guidance /committee-opinion/articles/2021/02/informed -consent-and-shared-decision-making-in-obstetrics-and -gynecology
- Goyal AA, Tur K, Mann J, et al. Do bedside visual tools improve patient and caregiver satisfaction? A systematic review of the literature. J Hosp Med. 2017;12:930-936. doi:10.12788 /jhm.2871
- Sehgal NL, Green A, Vidyarthi AR, et al. Patient whiteboards as a communication tool in the hospital setting: a survey of practices and recommendations. J Hosp Med. 2010;5:234-239. doi:10.1002/jhm.638
- Weiseth A, Plough A, Aggarwal R, et al. Improving communication and teamwork during labor: a feasibility, acceptability, and safety study. Birth. 2022:49:637-647. doi:10.1111/birt.12630
New RSV vaccine will cut hospitalizations, study shows
, according to research presented at an annual scientific meeting on infectious diseases.
“With RSV maternal vaccination that is associated with clinical efficacy of 69% against severe RSV disease at 6 months, we estimated that up to 200,000 cases can be averted, and that is associated with almost $800 million in total,” presenting author Amy W. Law, PharmD, director of global value and evidence at Pfizer, pointed out during a news briefing.
“RSV is associated with a significant burden in the U.S. and this newly approved and recommended maternal RSV vaccine can have substantial impact in easing some of that burden,” Dr. Law explained.
This study is “particularly timely as we head into RSV peak season,” said briefing moderator Natasha Halasa, MD, MPH, professor of pediatrics, division of pediatric infectious diseases at Vanderbilt University, Nashville, Tenn.
The challenge, said Dr. Halasa, is that uptake of maternal vaccines and vaccines in general is “not optimal,” making increased awareness of this new maternal RSV vaccine important.
Strong efficacy data
Most children are infected with RSV at least once by the time they reach age 2 years. Very young children are at particular risk of severe complications, such as pneumonia or bronchitis.
As reported previously by this news organization, in the randomized, double-blind, placebo-controlled phase 3 study, Pfizer’s maternal RSV vaccine had an almost 82% efficacy against severe RSV infection in infants from birth through the first 90 days of life.
The vaccine also had a 69% efficacy against severe disease through the first 6 months of life. As part of the trial, a total of 7,400 women received a single dose of the vaccine in the late second or third trimester of their pregnancy. There were no signs of safety issues for the mothers or infants.
Based on the results, the U.S. Food and Drug Administration approved the vaccine, known as Abrysvo, in August, to be given between weeks 32 and 36 of pregnancy.
New modeling study
Dr. Law and colleagues modeled the potential public health impact – both clinical and economic – of the maternal RSV vaccine among the population of all pregnant women and their infants born during a 12-month period in the United States. The model focused on severe RSV disease in babies that required medical attention.
According to their model, without widespread use of the maternal RSV vaccine, 48,246 hospitalizations, 144,495 emergency department encounters, and 399,313 outpatient clinic visits related to RSV are projected to occur annually among the U.S. birth cohort of 3.7 million infants younger than 12 months.
With widespread use of the vaccine, annual hospitalizations resulting from infant RSV would fall by 51%, emergency department encounters would decline by 32%, and outpatient clinic visits by 32% – corresponding to a decrease in direct medical costs of about $692 million and indirect nonmedical costs of roughly $110 million.
Dr. Law highlighted two important caveats to the data. “The protections are based on the year-round administration of the vaccine to pregnant women at 32 to 36 weeks’ gestational age, and this is also assuming 100% uptake. Of course, in reality, that most likely is not the case,” she told the briefing.
Dr. Halasa noted that the peak age for severe RSV illness is 3 months and it’s tough to identify infants at highest risk for severe RSV.
Nearly 80% of infants with RSV who are hospitalized do not have an underlying medical condition, “so we don’t even know who those high-risk infants are. That’s why having this vaccine is so exciting,” she told the briefing.
Dr. Halasa said it’s also important to note that infants with severe RSV typically make not just one but multiple visits to the clinic or emergency department, leading to missed days of work for the parent, not to mention the “emotional burden of having your otherwise healthy newborn or young infant in the hospital.”
In addition to Pfizer’s maternal RSV vaccine, the FDA in July approved AstraZeneca’s monoclonal antibody nirsevimab (Beyfortus) for the prevention of RSV in neonates and infants entering their first RSV season, and in children up to 24 months who remain vulnerable to severe RSV disease through their second RSV season.
The study was funded by Pfizer. Dr. Law is employed by Pfizer. Dr. Halasa has received grant and research support from Merck.
A version of this article first appeared on Medscape.com.
, according to research presented at an annual scientific meeting on infectious diseases.
“With RSV maternal vaccination that is associated with clinical efficacy of 69% against severe RSV disease at 6 months, we estimated that up to 200,000 cases can be averted, and that is associated with almost $800 million in total,” presenting author Amy W. Law, PharmD, director of global value and evidence at Pfizer, pointed out during a news briefing.
“RSV is associated with a significant burden in the U.S. and this newly approved and recommended maternal RSV vaccine can have substantial impact in easing some of that burden,” Dr. Law explained.
This study is “particularly timely as we head into RSV peak season,” said briefing moderator Natasha Halasa, MD, MPH, professor of pediatrics, division of pediatric infectious diseases at Vanderbilt University, Nashville, Tenn.
The challenge, said Dr. Halasa, is that uptake of maternal vaccines and vaccines in general is “not optimal,” making increased awareness of this new maternal RSV vaccine important.
Strong efficacy data
Most children are infected with RSV at least once by the time they reach age 2 years. Very young children are at particular risk of severe complications, such as pneumonia or bronchitis.
As reported previously by this news organization, in the randomized, double-blind, placebo-controlled phase 3 study, Pfizer’s maternal RSV vaccine had an almost 82% efficacy against severe RSV infection in infants from birth through the first 90 days of life.
The vaccine also had a 69% efficacy against severe disease through the first 6 months of life. As part of the trial, a total of 7,400 women received a single dose of the vaccine in the late second or third trimester of their pregnancy. There were no signs of safety issues for the mothers or infants.
Based on the results, the U.S. Food and Drug Administration approved the vaccine, known as Abrysvo, in August, to be given between weeks 32 and 36 of pregnancy.
New modeling study
Dr. Law and colleagues modeled the potential public health impact – both clinical and economic – of the maternal RSV vaccine among the population of all pregnant women and their infants born during a 12-month period in the United States. The model focused on severe RSV disease in babies that required medical attention.
According to their model, without widespread use of the maternal RSV vaccine, 48,246 hospitalizations, 144,495 emergency department encounters, and 399,313 outpatient clinic visits related to RSV are projected to occur annually among the U.S. birth cohort of 3.7 million infants younger than 12 months.
With widespread use of the vaccine, annual hospitalizations resulting from infant RSV would fall by 51%, emergency department encounters would decline by 32%, and outpatient clinic visits by 32% – corresponding to a decrease in direct medical costs of about $692 million and indirect nonmedical costs of roughly $110 million.
Dr. Law highlighted two important caveats to the data. “The protections are based on the year-round administration of the vaccine to pregnant women at 32 to 36 weeks’ gestational age, and this is also assuming 100% uptake. Of course, in reality, that most likely is not the case,” she told the briefing.
Dr. Halasa noted that the peak age for severe RSV illness is 3 months and it’s tough to identify infants at highest risk for severe RSV.
Nearly 80% of infants with RSV who are hospitalized do not have an underlying medical condition, “so we don’t even know who those high-risk infants are. That’s why having this vaccine is so exciting,” she told the briefing.
Dr. Halasa said it’s also important to note that infants with severe RSV typically make not just one but multiple visits to the clinic or emergency department, leading to missed days of work for the parent, not to mention the “emotional burden of having your otherwise healthy newborn or young infant in the hospital.”
In addition to Pfizer’s maternal RSV vaccine, the FDA in July approved AstraZeneca’s monoclonal antibody nirsevimab (Beyfortus) for the prevention of RSV in neonates and infants entering their first RSV season, and in children up to 24 months who remain vulnerable to severe RSV disease through their second RSV season.
The study was funded by Pfizer. Dr. Law is employed by Pfizer. Dr. Halasa has received grant and research support from Merck.
A version of this article first appeared on Medscape.com.
, according to research presented at an annual scientific meeting on infectious diseases.
“With RSV maternal vaccination that is associated with clinical efficacy of 69% against severe RSV disease at 6 months, we estimated that up to 200,000 cases can be averted, and that is associated with almost $800 million in total,” presenting author Amy W. Law, PharmD, director of global value and evidence at Pfizer, pointed out during a news briefing.
“RSV is associated with a significant burden in the U.S. and this newly approved and recommended maternal RSV vaccine can have substantial impact in easing some of that burden,” Dr. Law explained.
This study is “particularly timely as we head into RSV peak season,” said briefing moderator Natasha Halasa, MD, MPH, professor of pediatrics, division of pediatric infectious diseases at Vanderbilt University, Nashville, Tenn.
The challenge, said Dr. Halasa, is that uptake of maternal vaccines and vaccines in general is “not optimal,” making increased awareness of this new maternal RSV vaccine important.
Strong efficacy data
Most children are infected with RSV at least once by the time they reach age 2 years. Very young children are at particular risk of severe complications, such as pneumonia or bronchitis.
As reported previously by this news organization, in the randomized, double-blind, placebo-controlled phase 3 study, Pfizer’s maternal RSV vaccine had an almost 82% efficacy against severe RSV infection in infants from birth through the first 90 days of life.
The vaccine also had a 69% efficacy against severe disease through the first 6 months of life. As part of the trial, a total of 7,400 women received a single dose of the vaccine in the late second or third trimester of their pregnancy. There were no signs of safety issues for the mothers or infants.
Based on the results, the U.S. Food and Drug Administration approved the vaccine, known as Abrysvo, in August, to be given between weeks 32 and 36 of pregnancy.
New modeling study
Dr. Law and colleagues modeled the potential public health impact – both clinical and economic – of the maternal RSV vaccine among the population of all pregnant women and their infants born during a 12-month period in the United States. The model focused on severe RSV disease in babies that required medical attention.
According to their model, without widespread use of the maternal RSV vaccine, 48,246 hospitalizations, 144,495 emergency department encounters, and 399,313 outpatient clinic visits related to RSV are projected to occur annually among the U.S. birth cohort of 3.7 million infants younger than 12 months.
With widespread use of the vaccine, annual hospitalizations resulting from infant RSV would fall by 51%, emergency department encounters would decline by 32%, and outpatient clinic visits by 32% – corresponding to a decrease in direct medical costs of about $692 million and indirect nonmedical costs of roughly $110 million.
Dr. Law highlighted two important caveats to the data. “The protections are based on the year-round administration of the vaccine to pregnant women at 32 to 36 weeks’ gestational age, and this is also assuming 100% uptake. Of course, in reality, that most likely is not the case,” she told the briefing.
Dr. Halasa noted that the peak age for severe RSV illness is 3 months and it’s tough to identify infants at highest risk for severe RSV.
Nearly 80% of infants with RSV who are hospitalized do not have an underlying medical condition, “so we don’t even know who those high-risk infants are. That’s why having this vaccine is so exciting,” she told the briefing.
Dr. Halasa said it’s also important to note that infants with severe RSV typically make not just one but multiple visits to the clinic or emergency department, leading to missed days of work for the parent, not to mention the “emotional burden of having your otherwise healthy newborn or young infant in the hospital.”
In addition to Pfizer’s maternal RSV vaccine, the FDA in July approved AstraZeneca’s monoclonal antibody nirsevimab (Beyfortus) for the prevention of RSV in neonates and infants entering their first RSV season, and in children up to 24 months who remain vulnerable to severe RSV disease through their second RSV season.
The study was funded by Pfizer. Dr. Law is employed by Pfizer. Dr. Halasa has received grant and research support from Merck.
A version of this article first appeared on Medscape.com.
FROM IDWEEK 2023
Can a novel, rapid-acting oral treatment effectively manage PPD?
Deligiannidis KM, Meltzer-Brody S, Maximos B, et al. Zuranolone for the treatment of postpartum depression. Am J Psychiatry. 2023;180:668-675. doi:10.1176/appi.ajp.20220785.
EXPERT COMMENTARY
Postpartum depression affects approximately 17.2% of patients in the peripartum period.1 Typical pharmacologic treatment of PPD includes selective serotonin reuptake inhibitors (SSRIs), which may take up to 12 weeks to take effect. Postpartum depression is thought to be secondary to maladaptation to hormonal fluctuations in the peripartum period, including allopregnanolone, a positive allosteric modulator of GABAA (γ-aminobutyric acid type A)receptors and a metabolite of progesterone, levels of which increase in pregnancy and abruptly decrease following delivery.1 In 2019, the GABAA receptor modulator brexanalone was approved by the US Food and Drug Administration (FDA) to treat PPD through continuous intravenous infusion over 60 hours in the hospital setting.
Zuranolone, an allosteric modulator of GABAA receptors, also has been studied as an investigational medication for rapid treatment of PPD. Prior studies demonstrated the efficacy of oral zuranolone 30 mg daily for the treatment of PPD2 and 50 mg for the treatment of major depression in nonpregnant patients.3 Deligiannidis and colleagues conducted a trial to investigate the 50-mg dose of zuranolone for the treatment of PPD. (Notably, in August 2023, the FDA approved oral zuranolone once daily for 14 days for the treatment of PPD.) Following the FDA approval, the American College of Obstetricians and Gynecologists (ACOG) released a Practice Advisory recommending consideration of zuranolone for PPD that takes into account balancing the benefits and risks, including known sedative effects, potential need for decreasing the dose due to adverse effects, lack of safety data in lactation, and unknown long-term efficacy.4
Details of the study
This randomized, double-blind, placebo-controlled study included 196 patients with an episode of major depression, characterized as a baseline score of 26 or greater on the Hamilton Depression Rating Scale (HAM-D) beginning in the third trimester or within the first 4 weeks postpartum. Patients were randomly assigned in a 1:1 ratio to receive zuranolone 50 mg daily or placebo, with stratification by stable concurrent antidepressant use. Treatment duration was for 14 days, with follow-up through day 45.
The study’s primary outcome was a change in the baseline HAM-D score at day 15. Changes in HAM-D score also were recorded at days 3, 28, and 45.
The 2 study groups were well balanced by demographic and baseline characteristics. In both groups, the majority of patients experienced the onset of their major depressive episodes within the first 4 weeks postpartum. Completion rates of the 14-day treatment course and 45-day follow-up were high and similar in both groups; 170 patients completed the study. The rate of concurrent psychiatric medications taken, most of which were SSRIs, was similar between the 2 groups at approximately 15% of patients.
Results. A statistically significant improvement in the primary outcome (the change in HAM-D score) at day 15 occurred in patients who received zuranolone versus placebo (P = .001). Additionally, there were statistically significant improvements in the secondary outcomes HAM-D scores at days 3, 28, and 45. Initial response, as measured by changes in HAM-D scores, occurred at a median duration of 9 days in the zuranolone group and 43 days in the placebo group. More patients in the zuranolone group achieved a reduction in HAM-D score at 15 days (57.0% vs 38.9%; P = .02). Zuranolone was associated with a higher rate of HAM-D remission at day 45 (44.0% vs 29.4%; P = .02).
With regard to safety, 16.3% of patients (17) in the zuranolone group (vs 1% in the placebo group) experienced an adverse event, most commonly somnolence, dizziness, and sedation, which led to a dose reduction. However, 15 of these 17 patients still completed the study, and there were no serious adverse events.
Study strengths and limitations
This study’s strengths include the double-blinded design that was continued throughout the duration of the follow-up. Additionally, the study population was heterogeneous andreflective of patients from diverse racial and ethnic backgrounds. Lastly, only minor and moderate adverse events were reported and, despite this, nearly all patients who experienced adverse events completed the study.
Limitations of the study include the lack of generalizability, as patients with bipolar disorder and mild or moderate PPD were excluded. Additionally, the majority of patients had depressive episodes within the first 4 weeks postpartum, thereby excluding patients with depressive episodes at other time points in the peripartum period. Further, as breastfeeding was prohibited, safety in lactating patients using zuranolone is unknown. Lastly, the study follow-up period was 45 days; therefore, the long-term efficacy of zuranolone treatment is unclear. ●
Zuranolone, a GABAA allosteric modulator, shows promise as an alternative to existing pharmacologic treatments for severe PPD that is orally administered and rapidly acting. While it is reasonable to consider its use in the specific patient population that benefited in this study, further studies are needed to determine its efficacy in other populations, the lowest effective dose for clinical improvement, and its interaction with other medications and breastfeeding. Additionally, the long-term remission rates of depressive symptoms in patients treated with zuranolone are unknown and warrant further study.
JAIMEY M. PAULI, MD; KENDALL CUNNINGHAM, MD
- Deligiannidis KM, Meltzer-Brody S, Maximos B, et al. Zuranolone for the treatment of postpartum depression. Am J Psychiatry. 2023;180:668-675. doi:10.1176/appi.ajp .20220785
- Deligiannidis KM, Meltzer-Brody S, Gunduz-Bruce H, et al. Effect of zuranolone vs placebo in postpartum depression: a randomized clinical trial. JAMA Psychiatry. 2021;78:951-959. doi:10.1001/jamapsychiatry.2021.1559
- Clayton AH, Lasser R, Parikh SV, et al. Zuranolone for the treatment of adults with major depressive disorder: a randomized, placebo-controlled phase 3 trial. Am J Psychiatry. 2023;180:676-684. doi:10.1176/appi.ajp.20220459
- Zuranolone for the treatment of postpartum depression. Practice Advisory. American College of Obstetricians and Gynecologists. August 2023. Accessed September 18, 2023. https://www.acog.org/clinical/clinical-guidance/practice -advisory/articles/2023/08/zuranolone-for-the-treatment-of -postpartum-depression
Deligiannidis KM, Meltzer-Brody S, Maximos B, et al. Zuranolone for the treatment of postpartum depression. Am J Psychiatry. 2023;180:668-675. doi:10.1176/appi.ajp.20220785.
EXPERT COMMENTARY
Postpartum depression affects approximately 17.2% of patients in the peripartum period.1 Typical pharmacologic treatment of PPD includes selective serotonin reuptake inhibitors (SSRIs), which may take up to 12 weeks to take effect. Postpartum depression is thought to be secondary to maladaptation to hormonal fluctuations in the peripartum period, including allopregnanolone, a positive allosteric modulator of GABAA (γ-aminobutyric acid type A)receptors and a metabolite of progesterone, levels of which increase in pregnancy and abruptly decrease following delivery.1 In 2019, the GABAA receptor modulator brexanalone was approved by the US Food and Drug Administration (FDA) to treat PPD through continuous intravenous infusion over 60 hours in the hospital setting.
Zuranolone, an allosteric modulator of GABAA receptors, also has been studied as an investigational medication for rapid treatment of PPD. Prior studies demonstrated the efficacy of oral zuranolone 30 mg daily for the treatment of PPD2 and 50 mg for the treatment of major depression in nonpregnant patients.3 Deligiannidis and colleagues conducted a trial to investigate the 50-mg dose of zuranolone for the treatment of PPD. (Notably, in August 2023, the FDA approved oral zuranolone once daily for 14 days for the treatment of PPD.) Following the FDA approval, the American College of Obstetricians and Gynecologists (ACOG) released a Practice Advisory recommending consideration of zuranolone for PPD that takes into account balancing the benefits and risks, including known sedative effects, potential need for decreasing the dose due to adverse effects, lack of safety data in lactation, and unknown long-term efficacy.4
Details of the study
This randomized, double-blind, placebo-controlled study included 196 patients with an episode of major depression, characterized as a baseline score of 26 or greater on the Hamilton Depression Rating Scale (HAM-D) beginning in the third trimester or within the first 4 weeks postpartum. Patients were randomly assigned in a 1:1 ratio to receive zuranolone 50 mg daily or placebo, with stratification by stable concurrent antidepressant use. Treatment duration was for 14 days, with follow-up through day 45.
The study’s primary outcome was a change in the baseline HAM-D score at day 15. Changes in HAM-D score also were recorded at days 3, 28, and 45.
The 2 study groups were well balanced by demographic and baseline characteristics. In both groups, the majority of patients experienced the onset of their major depressive episodes within the first 4 weeks postpartum. Completion rates of the 14-day treatment course and 45-day follow-up were high and similar in both groups; 170 patients completed the study. The rate of concurrent psychiatric medications taken, most of which were SSRIs, was similar between the 2 groups at approximately 15% of patients.
Results. A statistically significant improvement in the primary outcome (the change in HAM-D score) at day 15 occurred in patients who received zuranolone versus placebo (P = .001). Additionally, there were statistically significant improvements in the secondary outcomes HAM-D scores at days 3, 28, and 45. Initial response, as measured by changes in HAM-D scores, occurred at a median duration of 9 days in the zuranolone group and 43 days in the placebo group. More patients in the zuranolone group achieved a reduction in HAM-D score at 15 days (57.0% vs 38.9%; P = .02). Zuranolone was associated with a higher rate of HAM-D remission at day 45 (44.0% vs 29.4%; P = .02).
With regard to safety, 16.3% of patients (17) in the zuranolone group (vs 1% in the placebo group) experienced an adverse event, most commonly somnolence, dizziness, and sedation, which led to a dose reduction. However, 15 of these 17 patients still completed the study, and there were no serious adverse events.
Study strengths and limitations
This study’s strengths include the double-blinded design that was continued throughout the duration of the follow-up. Additionally, the study population was heterogeneous andreflective of patients from diverse racial and ethnic backgrounds. Lastly, only minor and moderate adverse events were reported and, despite this, nearly all patients who experienced adverse events completed the study.
Limitations of the study include the lack of generalizability, as patients with bipolar disorder and mild or moderate PPD were excluded. Additionally, the majority of patients had depressive episodes within the first 4 weeks postpartum, thereby excluding patients with depressive episodes at other time points in the peripartum period. Further, as breastfeeding was prohibited, safety in lactating patients using zuranolone is unknown. Lastly, the study follow-up period was 45 days; therefore, the long-term efficacy of zuranolone treatment is unclear. ●
Zuranolone, a GABAA allosteric modulator, shows promise as an alternative to existing pharmacologic treatments for severe PPD that is orally administered and rapidly acting. While it is reasonable to consider its use in the specific patient population that benefited in this study, further studies are needed to determine its efficacy in other populations, the lowest effective dose for clinical improvement, and its interaction with other medications and breastfeeding. Additionally, the long-term remission rates of depressive symptoms in patients treated with zuranolone are unknown and warrant further study.
JAIMEY M. PAULI, MD; KENDALL CUNNINGHAM, MD
Deligiannidis KM, Meltzer-Brody S, Maximos B, et al. Zuranolone for the treatment of postpartum depression. Am J Psychiatry. 2023;180:668-675. doi:10.1176/appi.ajp.20220785.
EXPERT COMMENTARY
Postpartum depression affects approximately 17.2% of patients in the peripartum period.1 Typical pharmacologic treatment of PPD includes selective serotonin reuptake inhibitors (SSRIs), which may take up to 12 weeks to take effect. Postpartum depression is thought to be secondary to maladaptation to hormonal fluctuations in the peripartum period, including allopregnanolone, a positive allosteric modulator of GABAA (γ-aminobutyric acid type A)receptors and a metabolite of progesterone, levels of which increase in pregnancy and abruptly decrease following delivery.1 In 2019, the GABAA receptor modulator brexanalone was approved by the US Food and Drug Administration (FDA) to treat PPD through continuous intravenous infusion over 60 hours in the hospital setting.
Zuranolone, an allosteric modulator of GABAA receptors, also has been studied as an investigational medication for rapid treatment of PPD. Prior studies demonstrated the efficacy of oral zuranolone 30 mg daily for the treatment of PPD2 and 50 mg for the treatment of major depression in nonpregnant patients.3 Deligiannidis and colleagues conducted a trial to investigate the 50-mg dose of zuranolone for the treatment of PPD. (Notably, in August 2023, the FDA approved oral zuranolone once daily for 14 days for the treatment of PPD.) Following the FDA approval, the American College of Obstetricians and Gynecologists (ACOG) released a Practice Advisory recommending consideration of zuranolone for PPD that takes into account balancing the benefits and risks, including known sedative effects, potential need for decreasing the dose due to adverse effects, lack of safety data in lactation, and unknown long-term efficacy.4
Details of the study
This randomized, double-blind, placebo-controlled study included 196 patients with an episode of major depression, characterized as a baseline score of 26 or greater on the Hamilton Depression Rating Scale (HAM-D) beginning in the third trimester or within the first 4 weeks postpartum. Patients were randomly assigned in a 1:1 ratio to receive zuranolone 50 mg daily or placebo, with stratification by stable concurrent antidepressant use. Treatment duration was for 14 days, with follow-up through day 45.
The study’s primary outcome was a change in the baseline HAM-D score at day 15. Changes in HAM-D score also were recorded at days 3, 28, and 45.
The 2 study groups were well balanced by demographic and baseline characteristics. In both groups, the majority of patients experienced the onset of their major depressive episodes within the first 4 weeks postpartum. Completion rates of the 14-day treatment course and 45-day follow-up were high and similar in both groups; 170 patients completed the study. The rate of concurrent psychiatric medications taken, most of which were SSRIs, was similar between the 2 groups at approximately 15% of patients.
Results. A statistically significant improvement in the primary outcome (the change in HAM-D score) at day 15 occurred in patients who received zuranolone versus placebo (P = .001). Additionally, there were statistically significant improvements in the secondary outcomes HAM-D scores at days 3, 28, and 45. Initial response, as measured by changes in HAM-D scores, occurred at a median duration of 9 days in the zuranolone group and 43 days in the placebo group. More patients in the zuranolone group achieved a reduction in HAM-D score at 15 days (57.0% vs 38.9%; P = .02). Zuranolone was associated with a higher rate of HAM-D remission at day 45 (44.0% vs 29.4%; P = .02).
With regard to safety, 16.3% of patients (17) in the zuranolone group (vs 1% in the placebo group) experienced an adverse event, most commonly somnolence, dizziness, and sedation, which led to a dose reduction. However, 15 of these 17 patients still completed the study, and there were no serious adverse events.
Study strengths and limitations
This study’s strengths include the double-blinded design that was continued throughout the duration of the follow-up. Additionally, the study population was heterogeneous andreflective of patients from diverse racial and ethnic backgrounds. Lastly, only minor and moderate adverse events were reported and, despite this, nearly all patients who experienced adverse events completed the study.
Limitations of the study include the lack of generalizability, as patients with bipolar disorder and mild or moderate PPD were excluded. Additionally, the majority of patients had depressive episodes within the first 4 weeks postpartum, thereby excluding patients with depressive episodes at other time points in the peripartum period. Further, as breastfeeding was prohibited, safety in lactating patients using zuranolone is unknown. Lastly, the study follow-up period was 45 days; therefore, the long-term efficacy of zuranolone treatment is unclear. ●
Zuranolone, a GABAA allosteric modulator, shows promise as an alternative to existing pharmacologic treatments for severe PPD that is orally administered and rapidly acting. While it is reasonable to consider its use in the specific patient population that benefited in this study, further studies are needed to determine its efficacy in other populations, the lowest effective dose for clinical improvement, and its interaction with other medications and breastfeeding. Additionally, the long-term remission rates of depressive symptoms in patients treated with zuranolone are unknown and warrant further study.
JAIMEY M. PAULI, MD; KENDALL CUNNINGHAM, MD
- Deligiannidis KM, Meltzer-Brody S, Maximos B, et al. Zuranolone for the treatment of postpartum depression. Am J Psychiatry. 2023;180:668-675. doi:10.1176/appi.ajp .20220785
- Deligiannidis KM, Meltzer-Brody S, Gunduz-Bruce H, et al. Effect of zuranolone vs placebo in postpartum depression: a randomized clinical trial. JAMA Psychiatry. 2021;78:951-959. doi:10.1001/jamapsychiatry.2021.1559
- Clayton AH, Lasser R, Parikh SV, et al. Zuranolone for the treatment of adults with major depressive disorder: a randomized, placebo-controlled phase 3 trial. Am J Psychiatry. 2023;180:676-684. doi:10.1176/appi.ajp.20220459
- Zuranolone for the treatment of postpartum depression. Practice Advisory. American College of Obstetricians and Gynecologists. August 2023. Accessed September 18, 2023. https://www.acog.org/clinical/clinical-guidance/practice -advisory/articles/2023/08/zuranolone-for-the-treatment-of -postpartum-depression
- Deligiannidis KM, Meltzer-Brody S, Maximos B, et al. Zuranolone for the treatment of postpartum depression. Am J Psychiatry. 2023;180:668-675. doi:10.1176/appi.ajp .20220785
- Deligiannidis KM, Meltzer-Brody S, Gunduz-Bruce H, et al. Effect of zuranolone vs placebo in postpartum depression: a randomized clinical trial. JAMA Psychiatry. 2021;78:951-959. doi:10.1001/jamapsychiatry.2021.1559
- Clayton AH, Lasser R, Parikh SV, et al. Zuranolone for the treatment of adults with major depressive disorder: a randomized, placebo-controlled phase 3 trial. Am J Psychiatry. 2023;180:676-684. doi:10.1176/appi.ajp.20220459
- Zuranolone for the treatment of postpartum depression. Practice Advisory. American College of Obstetricians and Gynecologists. August 2023. Accessed September 18, 2023. https://www.acog.org/clinical/clinical-guidance/practice -advisory/articles/2023/08/zuranolone-for-the-treatment-of -postpartum-depression
Zuranolone: FAQs for clinicians and patients
The Food and Drug Administration approval of zuranolone for postpartum depression in August 2023 has raised many important questions (and opinions) about its future use in clinical practice.
At the UNC-Chapel Hill Center for Women’s Mood Disorders, we treat women and pregnant people throughout hormonal transitions, including pregnancy and the postpartum, and have been part of development, research, and now delivery of both brexanolone and zuranolone. While we are excited about new tools in the arsenal for alleviating maternal mental health, we also want to be clear that our work is far from complete and continued efforts to care for pregnant people and their families are imperative.
What is zuranolone?
Zuranolone (brand name Zurzuvae) is an oral medication developed by Sage Therapeutics and Biogen. It is a positive allosteric modulator of the GABAA receptor, the brain’s major inhibitory system. As a positive allosteric modulator, it increases the sensitivity of the GABAA receptor to GABA.
Zuranolone is very similar to brexanolone, a synthetic form of allopregnanolone, a neurosteroid byproduct of progesterone (see below). However, zuranolone is not an oral form of brexanolone – it was slightly modified to ensure good oral stability and bioavailability. It is metabolized by the hepatic enzyme CYP3A4 and has a half-life of 16-23 hours. Zurzuvae is currently produced in capsule form.
What does zuranolone treat?
Zuranolone is the first FDA-approved oral drug for postpartum depression (PPD). It follows brexanolone, an intravenous drug, which was the first FDA-approved medication for PPD. Though these are the first medications with specific approval for PDD, many other treatment options are currently available including therapy, SSRIs, serotonin norepinephrine reuptake inhibitors (SNRIs), and other treatments used in major depression.
How does zuranolone work?
Zuranolone is a neuroactive steroid, which means that it is a steroid that goes into and acts on the brain. Zuranolone binds to different GABA receptor subunits from those bound by other positive modulators, such as benzodiazepines (for example, lorazepam). As a synthetic form of allopregnanolone, a metabolite of progesterone which rises dramatically in pregnancy then drops during labor and delivery, zuranolone was originally thought to mitigate the response to this drop in patients that are vulnerable to it during the postpartum. An alternative proposed mechanism is that the increased GABAergic, inhibitory signaling with zuranolone may act directly to decrease depression irrespective of the exact mechanism by which the depression occurred.
How was it studied?
Zuranolone was studied in women with severe postpartum depression and had to meet criteria for major depressive disorder (MDD) no earlier than the third trimester of pregnancy (about 28 weeks’ gestation) and no later than 4 weeks post partum. Patients were excluded from these studies if they had a history of bipolar disorder, psychotic disorders, attempted suicide, or if they were at risk for suicide.
The two phase 3 clinical trials that led to FDA approval are ROBIN and SKYLARK. These studies measured the efficacy and safety of zuranolone at 30 mg and 50 mg, respectively, and met their end points of rapid improvement in depressive and anxiety symptoms in postpartum depression.
When will we be able to start using it?
It is anticipated that zuranolone will become commercially available in early 2024.
Who can prescribe it?
Those with medical licenses. Most people will likely receive treatment from their obstetric, family medicine, or psychiatric clinicians.
How much will it cost?
The manufacturers have not released this information as of August 2023.
What sort of doses and duration is recommended?
The current FDA recommended dose is 50 mg for 14 days, taken once per evening with a fatty meal. The dose can be reduced to 40 mg if there are central nervous system (CNS) depressant effects, and to 30 mg if the patient has severe hepatic or moderate-severe renal impairment. There are currently no studies on longer courses of treatment.
What happens if the patient relapses after a 14-day trial?
While there is no clear guidance, an open-label trial (The SHORELINE Study) demonstrated that a repeated 14-day administration can restore clinical response.
What are the side effects?
Common side effects include drowsiness, dizziness, lower energy, diarrhea, and symptoms similar to the common cold. Zuranolone can act like a CNS depressant and can lead to sedation and somnolence.
Are there any boxed warnings?
Because of the CNS depressant effects, zuranolone was given a boxed warning that patients should not drive or operate heavy machinery within 12 hours of taking the medication as it may lead to impairment. Similar to other antidepressants, there is also a warning that zuranolone may increase risk for suicidal thoughts in patients under 24 years old.
Can it be used with other medications?
Yes. In the original trials, women were allowed to remain on medications treating their depressive symptoms (such as SSRIs and SNRIs). According to the FDA, zuranolone can be used alone or with other antidepressants.
Are there any medicines to avoid?
We recommend caution with other medications which may increase sedation, such as benzodiazepines.
Can it be used with birth control?
Yes. In fact, because the outcomes on a fetus are not yet studied, it is recommended that patients be on concurrent birth control during treatment and for a week after cessation. This does not mean that zuranolone is known to cause issues with fetal development, but rather that we do not know at this time.
Can it be used in pregnancy?
As above, the outcomes on fetal development are not known at this time, nor are the effects of zuranolone on labor and delivery. More research will need to be done to understand if there is risk with taking zuranolone during pregnancy. It should be noted that allopregnanolone levels ordinarily reach quite high levels during pregnancy.
Long-term side effects?
Long-term side effects are unknown. The study duration of ROBIN and SKYLARK was 45 days.
Breastfeeding?
Use in lactation has not yet been studied. Continued research is needed.
Can it be used in mood changes related to other reproductive changes or diagnoses like premenstrual dysphoric disorder and perimenopause?
The mechanism by which zuranolone is thought to work – that is, during changes in reproductive hormones – is implicated in other reproductive transitions such as premenstrual dysphoric disorder and perimenopause when reproductive hormones are fluctuating, though at lower levels than in pregnancy. Research will be required to assess efficacy and safety; however, the mechanistic reasons is worth pursuing. Additionally, zuranolone has not been studied in postpartum psychosis.
Can zuranolone be used to treat other affective conditions besides postpartum depression? Bipolar disorder?
Whether it may be beneficial for patients with a depressive episode that is part of an underlying bipolar disorder or other psychiatric illness is not yet known.
Anxiety?
Along with depressive symptoms, women who received zuranolone in the clinical trials also had improvements in anxiety symptoms. These findings provide some hope that zuranolone may eventually be beneficial in patients with anxiety.
However, to date zuranolone has not been directly studied as a treatment for anxiety disorders (such as generalized anxiety disorder, panic disorder, etc.), so its efficacy for these illnesses is currently unknown.
Insomnia?
In a study of 153 postpartum women, randomized to placebo or zuranolone, scale questions for insomnia were improved in the group receiving zuranolone. This provides some hope that, if zuranolone is appropriate, concurrent polypharmacy with a sleep aid can be avoided. Additionally, future evaluation of use in insomnia outside of PPD may be warranted.
How is it different from brexanolone?
The two are slightly different molecules. Brexanolone is synthetically identical to allopregnanolone and zuranolone has been altered to be active and orally bioavailable.
Brexanolone is a 60-hour infusion that requires hospital admission at an approved health care site. Zuranolone is an oral at-home once-daily dosing treatment for 14 days. Zuranolone does not require enrollment in a risk evaluation and mitigation strategy for risk of excessive sedation and sudden loss of consciousness.
When would you consider zuranolone vs. brexanolone vs. other antidepressants?
Zuranolone and brexanolone are rapid-acting antidepressants with a response within 14 days or 60 hours, respectively. Antidepressants such as SSRIs/SNRIs are still available, well studied, and work, although take longer to reach clinical efficacy and are accompanied by potentially troubling side effects (for example, weight gain, sexual dysfunction).
Time to treatment effect should be considered when assessing severity of symptoms and functional impairment of the mother and the overall family unit. Brexanolone requires continuous monitoring which may be beneficial for women who are severely impaired and may benefit from frequent clinical monitoring. Brexanolone does not require a dose reduction with hepatic impairment, however, should be avoided in end-stage renal disease because of the potential accumulation of the solubilizing agent.
Where can I find more information?
Many states have maternal mental health consultation lines (examples include NCMATTERS here in North Carolina and MCPAP for Moms in Massachusetts) for clinicians (mental health, primary care, and obstetricians) that can be utilized for questions about prescribing. Postpartum Support International also has a clinician line for those without state services.
We plan to update this entry upon market release and access to new information.
Dr. Riddle and Dr. Nathan are assistant professors in the department of psychiatry at the University of North Carolina at Chapel Hill. Dr. Richardson is a perinatal psychiatry fellow, department of psychiatry, UNC-Chapel Hill. Dr. Rubinow is Distinguished Professor in the department of psychiatry, UNC-Chapel Hill. Dr. Meltzer-Brody is Assad Meymandi Distinguished Professor and Chair, department of psychiatry, UNC-Chapel Hill.
References
Deligiannidis KM et al. J Clin Psychiatry. 2023 Jan 30;84(1):22m14475. doi: 10.4088/JCP.22m14475.
Deligiannidis KM et al. . Obstetrics & Gynecology. 2023 May;141(5S):64S-65S. doi: 10.1097/01.AOG.0000930588.16136.3f.
Deligiannidis KM et al. Am J Psychiatry. 2023 Sep 1;180(9):668-75. doi: 10.1176/appi.ajp.20220785.
Deligiannidis KM et al. JAMA Psychiatry. 2021 Sep 1;78(9):951-59. doi: 10.1001/jamapsychiatry.2021.1559.
FDA Approves First Oral Treatment for Postpartum Depression. 2023 Aug 4. https://www.fda.gov/news-events/press-announcements/fda-approves-first-oral-treatment-postpartum-depression
ZURZUVAE – HIGHLIGHTS OF PRESCRIBING INFORMATION. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/217369s000lbl.pdf
The Food and Drug Administration approval of zuranolone for postpartum depression in August 2023 has raised many important questions (and opinions) about its future use in clinical practice.
At the UNC-Chapel Hill Center for Women’s Mood Disorders, we treat women and pregnant people throughout hormonal transitions, including pregnancy and the postpartum, and have been part of development, research, and now delivery of both brexanolone and zuranolone. While we are excited about new tools in the arsenal for alleviating maternal mental health, we also want to be clear that our work is far from complete and continued efforts to care for pregnant people and their families are imperative.
What is zuranolone?
Zuranolone (brand name Zurzuvae) is an oral medication developed by Sage Therapeutics and Biogen. It is a positive allosteric modulator of the GABAA receptor, the brain’s major inhibitory system. As a positive allosteric modulator, it increases the sensitivity of the GABAA receptor to GABA.
Zuranolone is very similar to brexanolone, a synthetic form of allopregnanolone, a neurosteroid byproduct of progesterone (see below). However, zuranolone is not an oral form of brexanolone – it was slightly modified to ensure good oral stability and bioavailability. It is metabolized by the hepatic enzyme CYP3A4 and has a half-life of 16-23 hours. Zurzuvae is currently produced in capsule form.
What does zuranolone treat?
Zuranolone is the first FDA-approved oral drug for postpartum depression (PPD). It follows brexanolone, an intravenous drug, which was the first FDA-approved medication for PPD. Though these are the first medications with specific approval for PDD, many other treatment options are currently available including therapy, SSRIs, serotonin norepinephrine reuptake inhibitors (SNRIs), and other treatments used in major depression.
How does zuranolone work?
Zuranolone is a neuroactive steroid, which means that it is a steroid that goes into and acts on the brain. Zuranolone binds to different GABA receptor subunits from those bound by other positive modulators, such as benzodiazepines (for example, lorazepam). As a synthetic form of allopregnanolone, a metabolite of progesterone which rises dramatically in pregnancy then drops during labor and delivery, zuranolone was originally thought to mitigate the response to this drop in patients that are vulnerable to it during the postpartum. An alternative proposed mechanism is that the increased GABAergic, inhibitory signaling with zuranolone may act directly to decrease depression irrespective of the exact mechanism by which the depression occurred.
How was it studied?
Zuranolone was studied in women with severe postpartum depression and had to meet criteria for major depressive disorder (MDD) no earlier than the third trimester of pregnancy (about 28 weeks’ gestation) and no later than 4 weeks post partum. Patients were excluded from these studies if they had a history of bipolar disorder, psychotic disorders, attempted suicide, or if they were at risk for suicide.
The two phase 3 clinical trials that led to FDA approval are ROBIN and SKYLARK. These studies measured the efficacy and safety of zuranolone at 30 mg and 50 mg, respectively, and met their end points of rapid improvement in depressive and anxiety symptoms in postpartum depression.
When will we be able to start using it?
It is anticipated that zuranolone will become commercially available in early 2024.
Who can prescribe it?
Those with medical licenses. Most people will likely receive treatment from their obstetric, family medicine, or psychiatric clinicians.
How much will it cost?
The manufacturers have not released this information as of August 2023.
What sort of doses and duration is recommended?
The current FDA recommended dose is 50 mg for 14 days, taken once per evening with a fatty meal. The dose can be reduced to 40 mg if there are central nervous system (CNS) depressant effects, and to 30 mg if the patient has severe hepatic or moderate-severe renal impairment. There are currently no studies on longer courses of treatment.
What happens if the patient relapses after a 14-day trial?
While there is no clear guidance, an open-label trial (The SHORELINE Study) demonstrated that a repeated 14-day administration can restore clinical response.
What are the side effects?
Common side effects include drowsiness, dizziness, lower energy, diarrhea, and symptoms similar to the common cold. Zuranolone can act like a CNS depressant and can lead to sedation and somnolence.
Are there any boxed warnings?
Because of the CNS depressant effects, zuranolone was given a boxed warning that patients should not drive or operate heavy machinery within 12 hours of taking the medication as it may lead to impairment. Similar to other antidepressants, there is also a warning that zuranolone may increase risk for suicidal thoughts in patients under 24 years old.
Can it be used with other medications?
Yes. In the original trials, women were allowed to remain on medications treating their depressive symptoms (such as SSRIs and SNRIs). According to the FDA, zuranolone can be used alone or with other antidepressants.
Are there any medicines to avoid?
We recommend caution with other medications which may increase sedation, such as benzodiazepines.
Can it be used with birth control?
Yes. In fact, because the outcomes on a fetus are not yet studied, it is recommended that patients be on concurrent birth control during treatment and for a week after cessation. This does not mean that zuranolone is known to cause issues with fetal development, but rather that we do not know at this time.
Can it be used in pregnancy?
As above, the outcomes on fetal development are not known at this time, nor are the effects of zuranolone on labor and delivery. More research will need to be done to understand if there is risk with taking zuranolone during pregnancy. It should be noted that allopregnanolone levels ordinarily reach quite high levels during pregnancy.
Long-term side effects?
Long-term side effects are unknown. The study duration of ROBIN and SKYLARK was 45 days.
Breastfeeding?
Use in lactation has not yet been studied. Continued research is needed.
Can it be used in mood changes related to other reproductive changes or diagnoses like premenstrual dysphoric disorder and perimenopause?
The mechanism by which zuranolone is thought to work – that is, during changes in reproductive hormones – is implicated in other reproductive transitions such as premenstrual dysphoric disorder and perimenopause when reproductive hormones are fluctuating, though at lower levels than in pregnancy. Research will be required to assess efficacy and safety; however, the mechanistic reasons is worth pursuing. Additionally, zuranolone has not been studied in postpartum psychosis.
Can zuranolone be used to treat other affective conditions besides postpartum depression? Bipolar disorder?
Whether it may be beneficial for patients with a depressive episode that is part of an underlying bipolar disorder or other psychiatric illness is not yet known.
Anxiety?
Along with depressive symptoms, women who received zuranolone in the clinical trials also had improvements in anxiety symptoms. These findings provide some hope that zuranolone may eventually be beneficial in patients with anxiety.
However, to date zuranolone has not been directly studied as a treatment for anxiety disorders (such as generalized anxiety disorder, panic disorder, etc.), so its efficacy for these illnesses is currently unknown.
Insomnia?
In a study of 153 postpartum women, randomized to placebo or zuranolone, scale questions for insomnia were improved in the group receiving zuranolone. This provides some hope that, if zuranolone is appropriate, concurrent polypharmacy with a sleep aid can be avoided. Additionally, future evaluation of use in insomnia outside of PPD may be warranted.
How is it different from brexanolone?
The two are slightly different molecules. Brexanolone is synthetically identical to allopregnanolone and zuranolone has been altered to be active and orally bioavailable.
Brexanolone is a 60-hour infusion that requires hospital admission at an approved health care site. Zuranolone is an oral at-home once-daily dosing treatment for 14 days. Zuranolone does not require enrollment in a risk evaluation and mitigation strategy for risk of excessive sedation and sudden loss of consciousness.
When would you consider zuranolone vs. brexanolone vs. other antidepressants?
Zuranolone and brexanolone are rapid-acting antidepressants with a response within 14 days or 60 hours, respectively. Antidepressants such as SSRIs/SNRIs are still available, well studied, and work, although take longer to reach clinical efficacy and are accompanied by potentially troubling side effects (for example, weight gain, sexual dysfunction).
Time to treatment effect should be considered when assessing severity of symptoms and functional impairment of the mother and the overall family unit. Brexanolone requires continuous monitoring which may be beneficial for women who are severely impaired and may benefit from frequent clinical monitoring. Brexanolone does not require a dose reduction with hepatic impairment, however, should be avoided in end-stage renal disease because of the potential accumulation of the solubilizing agent.
Where can I find more information?
Many states have maternal mental health consultation lines (examples include NCMATTERS here in North Carolina and MCPAP for Moms in Massachusetts) for clinicians (mental health, primary care, and obstetricians) that can be utilized for questions about prescribing. Postpartum Support International also has a clinician line for those without state services.
We plan to update this entry upon market release and access to new information.
Dr. Riddle and Dr. Nathan are assistant professors in the department of psychiatry at the University of North Carolina at Chapel Hill. Dr. Richardson is a perinatal psychiatry fellow, department of psychiatry, UNC-Chapel Hill. Dr. Rubinow is Distinguished Professor in the department of psychiatry, UNC-Chapel Hill. Dr. Meltzer-Brody is Assad Meymandi Distinguished Professor and Chair, department of psychiatry, UNC-Chapel Hill.
References
Deligiannidis KM et al. J Clin Psychiatry. 2023 Jan 30;84(1):22m14475. doi: 10.4088/JCP.22m14475.
Deligiannidis KM et al. . Obstetrics & Gynecology. 2023 May;141(5S):64S-65S. doi: 10.1097/01.AOG.0000930588.16136.3f.
Deligiannidis KM et al. Am J Psychiatry. 2023 Sep 1;180(9):668-75. doi: 10.1176/appi.ajp.20220785.
Deligiannidis KM et al. JAMA Psychiatry. 2021 Sep 1;78(9):951-59. doi: 10.1001/jamapsychiatry.2021.1559.
FDA Approves First Oral Treatment for Postpartum Depression. 2023 Aug 4. https://www.fda.gov/news-events/press-announcements/fda-approves-first-oral-treatment-postpartum-depression
ZURZUVAE – HIGHLIGHTS OF PRESCRIBING INFORMATION. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/217369s000lbl.pdf
The Food and Drug Administration approval of zuranolone for postpartum depression in August 2023 has raised many important questions (and opinions) about its future use in clinical practice.
At the UNC-Chapel Hill Center for Women’s Mood Disorders, we treat women and pregnant people throughout hormonal transitions, including pregnancy and the postpartum, and have been part of development, research, and now delivery of both brexanolone and zuranolone. While we are excited about new tools in the arsenal for alleviating maternal mental health, we also want to be clear that our work is far from complete and continued efforts to care for pregnant people and their families are imperative.
What is zuranolone?
Zuranolone (brand name Zurzuvae) is an oral medication developed by Sage Therapeutics and Biogen. It is a positive allosteric modulator of the GABAA receptor, the brain’s major inhibitory system. As a positive allosteric modulator, it increases the sensitivity of the GABAA receptor to GABA.
Zuranolone is very similar to brexanolone, a synthetic form of allopregnanolone, a neurosteroid byproduct of progesterone (see below). However, zuranolone is not an oral form of brexanolone – it was slightly modified to ensure good oral stability and bioavailability. It is metabolized by the hepatic enzyme CYP3A4 and has a half-life of 16-23 hours. Zurzuvae is currently produced in capsule form.
What does zuranolone treat?
Zuranolone is the first FDA-approved oral drug for postpartum depression (PPD). It follows brexanolone, an intravenous drug, which was the first FDA-approved medication for PPD. Though these are the first medications with specific approval for PDD, many other treatment options are currently available including therapy, SSRIs, serotonin norepinephrine reuptake inhibitors (SNRIs), and other treatments used in major depression.
How does zuranolone work?
Zuranolone is a neuroactive steroid, which means that it is a steroid that goes into and acts on the brain. Zuranolone binds to different GABA receptor subunits from those bound by other positive modulators, such as benzodiazepines (for example, lorazepam). As a synthetic form of allopregnanolone, a metabolite of progesterone which rises dramatically in pregnancy then drops during labor and delivery, zuranolone was originally thought to mitigate the response to this drop in patients that are vulnerable to it during the postpartum. An alternative proposed mechanism is that the increased GABAergic, inhibitory signaling with zuranolone may act directly to decrease depression irrespective of the exact mechanism by which the depression occurred.
How was it studied?
Zuranolone was studied in women with severe postpartum depression and had to meet criteria for major depressive disorder (MDD) no earlier than the third trimester of pregnancy (about 28 weeks’ gestation) and no later than 4 weeks post partum. Patients were excluded from these studies if they had a history of bipolar disorder, psychotic disorders, attempted suicide, or if they were at risk for suicide.
The two phase 3 clinical trials that led to FDA approval are ROBIN and SKYLARK. These studies measured the efficacy and safety of zuranolone at 30 mg and 50 mg, respectively, and met their end points of rapid improvement in depressive and anxiety symptoms in postpartum depression.
When will we be able to start using it?
It is anticipated that zuranolone will become commercially available in early 2024.
Who can prescribe it?
Those with medical licenses. Most people will likely receive treatment from their obstetric, family medicine, or psychiatric clinicians.
How much will it cost?
The manufacturers have not released this information as of August 2023.
What sort of doses and duration is recommended?
The current FDA recommended dose is 50 mg for 14 days, taken once per evening with a fatty meal. The dose can be reduced to 40 mg if there are central nervous system (CNS) depressant effects, and to 30 mg if the patient has severe hepatic or moderate-severe renal impairment. There are currently no studies on longer courses of treatment.
What happens if the patient relapses after a 14-day trial?
While there is no clear guidance, an open-label trial (The SHORELINE Study) demonstrated that a repeated 14-day administration can restore clinical response.
What are the side effects?
Common side effects include drowsiness, dizziness, lower energy, diarrhea, and symptoms similar to the common cold. Zuranolone can act like a CNS depressant and can lead to sedation and somnolence.
Are there any boxed warnings?
Because of the CNS depressant effects, zuranolone was given a boxed warning that patients should not drive or operate heavy machinery within 12 hours of taking the medication as it may lead to impairment. Similar to other antidepressants, there is also a warning that zuranolone may increase risk for suicidal thoughts in patients under 24 years old.
Can it be used with other medications?
Yes. In the original trials, women were allowed to remain on medications treating their depressive symptoms (such as SSRIs and SNRIs). According to the FDA, zuranolone can be used alone or with other antidepressants.
Are there any medicines to avoid?
We recommend caution with other medications which may increase sedation, such as benzodiazepines.
Can it be used with birth control?
Yes. In fact, because the outcomes on a fetus are not yet studied, it is recommended that patients be on concurrent birth control during treatment and for a week after cessation. This does not mean that zuranolone is known to cause issues with fetal development, but rather that we do not know at this time.
Can it be used in pregnancy?
As above, the outcomes on fetal development are not known at this time, nor are the effects of zuranolone on labor and delivery. More research will need to be done to understand if there is risk with taking zuranolone during pregnancy. It should be noted that allopregnanolone levels ordinarily reach quite high levels during pregnancy.
Long-term side effects?
Long-term side effects are unknown. The study duration of ROBIN and SKYLARK was 45 days.
Breastfeeding?
Use in lactation has not yet been studied. Continued research is needed.
Can it be used in mood changes related to other reproductive changes or diagnoses like premenstrual dysphoric disorder and perimenopause?
The mechanism by which zuranolone is thought to work – that is, during changes in reproductive hormones – is implicated in other reproductive transitions such as premenstrual dysphoric disorder and perimenopause when reproductive hormones are fluctuating, though at lower levels than in pregnancy. Research will be required to assess efficacy and safety; however, the mechanistic reasons is worth pursuing. Additionally, zuranolone has not been studied in postpartum psychosis.
Can zuranolone be used to treat other affective conditions besides postpartum depression? Bipolar disorder?
Whether it may be beneficial for patients with a depressive episode that is part of an underlying bipolar disorder or other psychiatric illness is not yet known.
Anxiety?
Along with depressive symptoms, women who received zuranolone in the clinical trials also had improvements in anxiety symptoms. These findings provide some hope that zuranolone may eventually be beneficial in patients with anxiety.
However, to date zuranolone has not been directly studied as a treatment for anxiety disorders (such as generalized anxiety disorder, panic disorder, etc.), so its efficacy for these illnesses is currently unknown.
Insomnia?
In a study of 153 postpartum women, randomized to placebo or zuranolone, scale questions for insomnia were improved in the group receiving zuranolone. This provides some hope that, if zuranolone is appropriate, concurrent polypharmacy with a sleep aid can be avoided. Additionally, future evaluation of use in insomnia outside of PPD may be warranted.
How is it different from brexanolone?
The two are slightly different molecules. Brexanolone is synthetically identical to allopregnanolone and zuranolone has been altered to be active and orally bioavailable.
Brexanolone is a 60-hour infusion that requires hospital admission at an approved health care site. Zuranolone is an oral at-home once-daily dosing treatment for 14 days. Zuranolone does not require enrollment in a risk evaluation and mitigation strategy for risk of excessive sedation and sudden loss of consciousness.
When would you consider zuranolone vs. brexanolone vs. other antidepressants?
Zuranolone and brexanolone are rapid-acting antidepressants with a response within 14 days or 60 hours, respectively. Antidepressants such as SSRIs/SNRIs are still available, well studied, and work, although take longer to reach clinical efficacy and are accompanied by potentially troubling side effects (for example, weight gain, sexual dysfunction).
Time to treatment effect should be considered when assessing severity of symptoms and functional impairment of the mother and the overall family unit. Brexanolone requires continuous monitoring which may be beneficial for women who are severely impaired and may benefit from frequent clinical monitoring. Brexanolone does not require a dose reduction with hepatic impairment, however, should be avoided in end-stage renal disease because of the potential accumulation of the solubilizing agent.
Where can I find more information?
Many states have maternal mental health consultation lines (examples include NCMATTERS here in North Carolina and MCPAP for Moms in Massachusetts) for clinicians (mental health, primary care, and obstetricians) that can be utilized for questions about prescribing. Postpartum Support International also has a clinician line for those without state services.
We plan to update this entry upon market release and access to new information.
Dr. Riddle and Dr. Nathan are assistant professors in the department of psychiatry at the University of North Carolina at Chapel Hill. Dr. Richardson is a perinatal psychiatry fellow, department of psychiatry, UNC-Chapel Hill. Dr. Rubinow is Distinguished Professor in the department of psychiatry, UNC-Chapel Hill. Dr. Meltzer-Brody is Assad Meymandi Distinguished Professor and Chair, department of psychiatry, UNC-Chapel Hill.
References
Deligiannidis KM et al. J Clin Psychiatry. 2023 Jan 30;84(1):22m14475. doi: 10.4088/JCP.22m14475.
Deligiannidis KM et al. . Obstetrics & Gynecology. 2023 May;141(5S):64S-65S. doi: 10.1097/01.AOG.0000930588.16136.3f.
Deligiannidis KM et al. Am J Psychiatry. 2023 Sep 1;180(9):668-75. doi: 10.1176/appi.ajp.20220785.
Deligiannidis KM et al. JAMA Psychiatry. 2021 Sep 1;78(9):951-59. doi: 10.1001/jamapsychiatry.2021.1559.
FDA Approves First Oral Treatment for Postpartum Depression. 2023 Aug 4. https://www.fda.gov/news-events/press-announcements/fda-approves-first-oral-treatment-postpartum-depression
ZURZUVAE – HIGHLIGHTS OF PRESCRIBING INFORMATION. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/217369s000lbl.pdf
Anemia, iron deficit common in rheumatic disease pregnancy
TOPLINE:
, according to findings from a longitudinal cohort study.
METHODOLOGY:
- Researchers analyzed data from 368 pregnancies in women with rheumatic diseases during the period 2014-2022; nearly two-thirds (62%) had a connective tissue disease, 16% had rheumatoid arthritis or juvenile idiopathic arthritis, 14% had spondyloarthritis, 3% had vasculitis, and 7% had other diseases.
- Patients were aged 17-44 years, with a median age of 32 years at the time of birth.
- Researchers examined the frequency of anemia and iron deficiency and the impact of anemia on adverse maternal and child outcomes.
TAKEAWAY:
- The prevalence of iron deficiency was 28%, 51%, and 62% in the first, second, and third trimesters, respectively.
- The prevalence of anemia was 18%, 27%, and 33% in the first, second, and third trimesters, respectively.
- There was an increased risk for fetal complications such as malformation, infections, small for gestational age, neonatal lupus, preterm birth, and abortion or stillbirth in association with maternal connective tissue disease (odds ratio, 2.14) and also with low maternal hemoglobin levels and maternal iron deficiency (ORs, 0.52 and 0.86, respectively).
- Lower maternal hemoglobin levels were associated with an increased risk for maternal complications (OR, 1.47) such as flare with adaption of rheumatic medication and pregnancy-related adverse events (preeclampsia, gestational diabetes, bleeding complications, and thromboembolism), but patients with connective tissue disease had a lower risk for maternal complications (OR, 0.51); mean serum ferritin had no significant impact on maternal complications (OR, 1.02).
IN PRACTICE:
“Patients with rheumatic diseases suffer more often and already in early pregnancy from iron deficiency,” the researchers write. Therefore, early identification of anemia and iron deficiency in this population could inform prepregnancy counseling.
SOURCE:
The lead author on the study was Ann-Christin Pecher, MD, of University Hospital Tübingen, Germany. The study was published online in Joint Bone Spine.
LIMITATIONS:
The findings were limited by the use of a single dataset that might not be representative of all pregnant patients with rheumatic diseases. Other limitations included the lack of a standardized approach to iron supplementation.
DISCLOSURES:
The study was supported by a grant from the Medical Faculty of Tübingen Clinician-Scientist to the lead author. The researchers report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE:
, according to findings from a longitudinal cohort study.
METHODOLOGY:
- Researchers analyzed data from 368 pregnancies in women with rheumatic diseases during the period 2014-2022; nearly two-thirds (62%) had a connective tissue disease, 16% had rheumatoid arthritis or juvenile idiopathic arthritis, 14% had spondyloarthritis, 3% had vasculitis, and 7% had other diseases.
- Patients were aged 17-44 years, with a median age of 32 years at the time of birth.
- Researchers examined the frequency of anemia and iron deficiency and the impact of anemia on adverse maternal and child outcomes.
TAKEAWAY:
- The prevalence of iron deficiency was 28%, 51%, and 62% in the first, second, and third trimesters, respectively.
- The prevalence of anemia was 18%, 27%, and 33% in the first, second, and third trimesters, respectively.
- There was an increased risk for fetal complications such as malformation, infections, small for gestational age, neonatal lupus, preterm birth, and abortion or stillbirth in association with maternal connective tissue disease (odds ratio, 2.14) and also with low maternal hemoglobin levels and maternal iron deficiency (ORs, 0.52 and 0.86, respectively).
- Lower maternal hemoglobin levels were associated with an increased risk for maternal complications (OR, 1.47) such as flare with adaption of rheumatic medication and pregnancy-related adverse events (preeclampsia, gestational diabetes, bleeding complications, and thromboembolism), but patients with connective tissue disease had a lower risk for maternal complications (OR, 0.51); mean serum ferritin had no significant impact on maternal complications (OR, 1.02).
IN PRACTICE:
“Patients with rheumatic diseases suffer more often and already in early pregnancy from iron deficiency,” the researchers write. Therefore, early identification of anemia and iron deficiency in this population could inform prepregnancy counseling.
SOURCE:
The lead author on the study was Ann-Christin Pecher, MD, of University Hospital Tübingen, Germany. The study was published online in Joint Bone Spine.
LIMITATIONS:
The findings were limited by the use of a single dataset that might not be representative of all pregnant patients with rheumatic diseases. Other limitations included the lack of a standardized approach to iron supplementation.
DISCLOSURES:
The study was supported by a grant from the Medical Faculty of Tübingen Clinician-Scientist to the lead author. The researchers report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE:
, according to findings from a longitudinal cohort study.
METHODOLOGY:
- Researchers analyzed data from 368 pregnancies in women with rheumatic diseases during the period 2014-2022; nearly two-thirds (62%) had a connective tissue disease, 16% had rheumatoid arthritis or juvenile idiopathic arthritis, 14% had spondyloarthritis, 3% had vasculitis, and 7% had other diseases.
- Patients were aged 17-44 years, with a median age of 32 years at the time of birth.
- Researchers examined the frequency of anemia and iron deficiency and the impact of anemia on adverse maternal and child outcomes.
TAKEAWAY:
- The prevalence of iron deficiency was 28%, 51%, and 62% in the first, second, and third trimesters, respectively.
- The prevalence of anemia was 18%, 27%, and 33% in the first, second, and third trimesters, respectively.
- There was an increased risk for fetal complications such as malformation, infections, small for gestational age, neonatal lupus, preterm birth, and abortion or stillbirth in association with maternal connective tissue disease (odds ratio, 2.14) and also with low maternal hemoglobin levels and maternal iron deficiency (ORs, 0.52 and 0.86, respectively).
- Lower maternal hemoglobin levels were associated with an increased risk for maternal complications (OR, 1.47) such as flare with adaption of rheumatic medication and pregnancy-related adverse events (preeclampsia, gestational diabetes, bleeding complications, and thromboembolism), but patients with connective tissue disease had a lower risk for maternal complications (OR, 0.51); mean serum ferritin had no significant impact on maternal complications (OR, 1.02).
IN PRACTICE:
“Patients with rheumatic diseases suffer more often and already in early pregnancy from iron deficiency,” the researchers write. Therefore, early identification of anemia and iron deficiency in this population could inform prepregnancy counseling.
SOURCE:
The lead author on the study was Ann-Christin Pecher, MD, of University Hospital Tübingen, Germany. The study was published online in Joint Bone Spine.
LIMITATIONS:
The findings were limited by the use of a single dataset that might not be representative of all pregnant patients with rheumatic diseases. Other limitations included the lack of a standardized approach to iron supplementation.
DISCLOSURES:
The study was supported by a grant from the Medical Faculty of Tübingen Clinician-Scientist to the lead author. The researchers report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Maternal pertussis vax effective for infants in most vulnerable months
Maternal pertussis vaccinations, given during pregnancy, prevent an estimated 65% of pertussis infections in infants, new research indicates.
The study, led by Annette K. Regan, PhD, MPH, a perinatal and pediatric infectious disease epidemiologist at Curtin University, Perth, Australia, was published online in Pediatrics.
Dr. Regan – who is also with the University of San Francisco and the University of California, Los Angeles – and colleagues reviewed data on 279,418 infants born to 252,444 mothers in Australia.
There, about 52% of the women in this study received the Tdap vaccine through a maternal pertussis vaccination program.
Duration of effectiveness in infants was one of the main questions the study sought to answer.
The authors wrote that they assessed vaccine effectiveness through 18 months of age. “We observed significant protection against disease until at least 8 months of age, 2 months longer than reported in previous studies.” From 70% to 90% of all pertussis-attributable hospitalizations and death occur in infancy.
Answering the ‘blunting’ question
This study also set out to clarify an important clinical question regarding a potential “blunting” effect in infants. Previous work had suggested that maternal antibodies from the vaccination could interfere with the effectiveness of infants’ DtaP (the version of Tdap for infants) and other vaccines.
Dr. Regan and colleagues found that, “although we observed slightly lower VE [vaccine effectiveness] point estimates for the third dose of infant pertussis vaccine among maternally vaccinated compared with unvaccinated infants (76.5% vs. 92.9%, P = .002), we did not observe higher rates of pertussis infection (hazard ratio, 0.70; 95% confidence interval, 0.61-3.39).
Best time to give mothers the vaccine
Another clinical debate has centered on when to give the mother the vaccine during pregnancy. The authors concluded: “Our findings support the infant health benefits of recommendations to administer a booster dose of pertussis vaccine near 28 weeks of gestational age.”
That 28-week mark was associated with lower risk of infection in infants through 8 months of age, they wrote.
Positive results in the United States
In an invited commentary, Kathryn M. Edwards, MD, with the division of infectious diseases, department of pediatrics, at Vanderbilt University Medical Center, Nashville, Tenn., highlighted similar positive findings for maternal pertussis vaccination in the United States.
The Centers for Disease Control and Prevention did an ecologic study of infant pertussis cases reported between Jan. 1, 2000, and Dec. 31, 2019. Rates were compared for the years before maternal Tdap vaccinations were recommended against the 7-year period after they were implemented.
That study found that in the period before maternal Tdap vaccination, annual pertussis incidence did not change among infants younger than 2 months and increased slightly in infants 6-12 months.
However, during the period after maternal Tdap vaccination had started (2012-2019), pertussis incidence significantly decreased in infants younger than 2 months and was unchanged in infants 6-12 months.
“As with the Australian data, the U.S. data support the overall benefit of the maternal Tdap program and, as with the Australian data, do not suggest that blunting has led to an increase in cases within the first year of life,” Dr. Edwards wrote.
The CDC notes that pertussis cases are rising and outbreaks are happening across the United States.
“On average, about 1,000 infants are hospitalized and typically between 5 and 15 infants die each year in the United States due to pertussis,” the CDC states.
Uptake low despite positive data
Dr. Edwards noted that, despite positive data supporting maternal vaccination to reduce pertussis, uptake rates are low – between 50% and 60% in Australia, the United Kingdom, and the United States. “Active engagement to increase these rates should be implemented.”
Maternal vaccination might also be implemented soon to protect against other diseases including respiratory syncytial virus and group B streptococcal disease after promising study data, she said.
As with pertussis, the potential “blunting” effect will need to be carefully monitored, she said, “as was done in the carefully conducted study of pertussis reported in this issue of Pediatrics.”
One coauthor has received institutional honoraria for participation in advisory groups for Merck Sharpe & Dohme and Pfizer unrelated to this work. Another coauthor was supported by scholarships provided by the Wesfarmers Centre of Vaccines and Infectious Disease at the Telethon Kids Institute. Dr. Edwards reported receiving grants from the CDC and consulting for Bionet, Dynavax, and IBM. She is a member of the data safety and monitoring board for Sanofi, X-4 Pharma, Seqirus, Moderna, Pfizer, Merck, Roche, Novavax, and Brighton Collaboration.
Maternal pertussis vaccinations, given during pregnancy, prevent an estimated 65% of pertussis infections in infants, new research indicates.
The study, led by Annette K. Regan, PhD, MPH, a perinatal and pediatric infectious disease epidemiologist at Curtin University, Perth, Australia, was published online in Pediatrics.
Dr. Regan – who is also with the University of San Francisco and the University of California, Los Angeles – and colleagues reviewed data on 279,418 infants born to 252,444 mothers in Australia.
There, about 52% of the women in this study received the Tdap vaccine through a maternal pertussis vaccination program.
Duration of effectiveness in infants was one of the main questions the study sought to answer.
The authors wrote that they assessed vaccine effectiveness through 18 months of age. “We observed significant protection against disease until at least 8 months of age, 2 months longer than reported in previous studies.” From 70% to 90% of all pertussis-attributable hospitalizations and death occur in infancy.
Answering the ‘blunting’ question
This study also set out to clarify an important clinical question regarding a potential “blunting” effect in infants. Previous work had suggested that maternal antibodies from the vaccination could interfere with the effectiveness of infants’ DtaP (the version of Tdap for infants) and other vaccines.
Dr. Regan and colleagues found that, “although we observed slightly lower VE [vaccine effectiveness] point estimates for the third dose of infant pertussis vaccine among maternally vaccinated compared with unvaccinated infants (76.5% vs. 92.9%, P = .002), we did not observe higher rates of pertussis infection (hazard ratio, 0.70; 95% confidence interval, 0.61-3.39).
Best time to give mothers the vaccine
Another clinical debate has centered on when to give the mother the vaccine during pregnancy. The authors concluded: “Our findings support the infant health benefits of recommendations to administer a booster dose of pertussis vaccine near 28 weeks of gestational age.”
That 28-week mark was associated with lower risk of infection in infants through 8 months of age, they wrote.
Positive results in the United States
In an invited commentary, Kathryn M. Edwards, MD, with the division of infectious diseases, department of pediatrics, at Vanderbilt University Medical Center, Nashville, Tenn., highlighted similar positive findings for maternal pertussis vaccination in the United States.
The Centers for Disease Control and Prevention did an ecologic study of infant pertussis cases reported between Jan. 1, 2000, and Dec. 31, 2019. Rates were compared for the years before maternal Tdap vaccinations were recommended against the 7-year period after they were implemented.
That study found that in the period before maternal Tdap vaccination, annual pertussis incidence did not change among infants younger than 2 months and increased slightly in infants 6-12 months.
However, during the period after maternal Tdap vaccination had started (2012-2019), pertussis incidence significantly decreased in infants younger than 2 months and was unchanged in infants 6-12 months.
“As with the Australian data, the U.S. data support the overall benefit of the maternal Tdap program and, as with the Australian data, do not suggest that blunting has led to an increase in cases within the first year of life,” Dr. Edwards wrote.
The CDC notes that pertussis cases are rising and outbreaks are happening across the United States.
“On average, about 1,000 infants are hospitalized and typically between 5 and 15 infants die each year in the United States due to pertussis,” the CDC states.
Uptake low despite positive data
Dr. Edwards noted that, despite positive data supporting maternal vaccination to reduce pertussis, uptake rates are low – between 50% and 60% in Australia, the United Kingdom, and the United States. “Active engagement to increase these rates should be implemented.”
Maternal vaccination might also be implemented soon to protect against other diseases including respiratory syncytial virus and group B streptococcal disease after promising study data, she said.
As with pertussis, the potential “blunting” effect will need to be carefully monitored, she said, “as was done in the carefully conducted study of pertussis reported in this issue of Pediatrics.”
One coauthor has received institutional honoraria for participation in advisory groups for Merck Sharpe & Dohme and Pfizer unrelated to this work. Another coauthor was supported by scholarships provided by the Wesfarmers Centre of Vaccines and Infectious Disease at the Telethon Kids Institute. Dr. Edwards reported receiving grants from the CDC and consulting for Bionet, Dynavax, and IBM. She is a member of the data safety and monitoring board for Sanofi, X-4 Pharma, Seqirus, Moderna, Pfizer, Merck, Roche, Novavax, and Brighton Collaboration.
Maternal pertussis vaccinations, given during pregnancy, prevent an estimated 65% of pertussis infections in infants, new research indicates.
The study, led by Annette K. Regan, PhD, MPH, a perinatal and pediatric infectious disease epidemiologist at Curtin University, Perth, Australia, was published online in Pediatrics.
Dr. Regan – who is also with the University of San Francisco and the University of California, Los Angeles – and colleagues reviewed data on 279,418 infants born to 252,444 mothers in Australia.
There, about 52% of the women in this study received the Tdap vaccine through a maternal pertussis vaccination program.
Duration of effectiveness in infants was one of the main questions the study sought to answer.
The authors wrote that they assessed vaccine effectiveness through 18 months of age. “We observed significant protection against disease until at least 8 months of age, 2 months longer than reported in previous studies.” From 70% to 90% of all pertussis-attributable hospitalizations and death occur in infancy.
Answering the ‘blunting’ question
This study also set out to clarify an important clinical question regarding a potential “blunting” effect in infants. Previous work had suggested that maternal antibodies from the vaccination could interfere with the effectiveness of infants’ DtaP (the version of Tdap for infants) and other vaccines.
Dr. Regan and colleagues found that, “although we observed slightly lower VE [vaccine effectiveness] point estimates for the third dose of infant pertussis vaccine among maternally vaccinated compared with unvaccinated infants (76.5% vs. 92.9%, P = .002), we did not observe higher rates of pertussis infection (hazard ratio, 0.70; 95% confidence interval, 0.61-3.39).
Best time to give mothers the vaccine
Another clinical debate has centered on when to give the mother the vaccine during pregnancy. The authors concluded: “Our findings support the infant health benefits of recommendations to administer a booster dose of pertussis vaccine near 28 weeks of gestational age.”
That 28-week mark was associated with lower risk of infection in infants through 8 months of age, they wrote.
Positive results in the United States
In an invited commentary, Kathryn M. Edwards, MD, with the division of infectious diseases, department of pediatrics, at Vanderbilt University Medical Center, Nashville, Tenn., highlighted similar positive findings for maternal pertussis vaccination in the United States.
The Centers for Disease Control and Prevention did an ecologic study of infant pertussis cases reported between Jan. 1, 2000, and Dec. 31, 2019. Rates were compared for the years before maternal Tdap vaccinations were recommended against the 7-year period after they were implemented.
That study found that in the period before maternal Tdap vaccination, annual pertussis incidence did not change among infants younger than 2 months and increased slightly in infants 6-12 months.
However, during the period after maternal Tdap vaccination had started (2012-2019), pertussis incidence significantly decreased in infants younger than 2 months and was unchanged in infants 6-12 months.
“As with the Australian data, the U.S. data support the overall benefit of the maternal Tdap program and, as with the Australian data, do not suggest that blunting has led to an increase in cases within the first year of life,” Dr. Edwards wrote.
The CDC notes that pertussis cases are rising and outbreaks are happening across the United States.
“On average, about 1,000 infants are hospitalized and typically between 5 and 15 infants die each year in the United States due to pertussis,” the CDC states.
Uptake low despite positive data
Dr. Edwards noted that, despite positive data supporting maternal vaccination to reduce pertussis, uptake rates are low – between 50% and 60% in Australia, the United Kingdom, and the United States. “Active engagement to increase these rates should be implemented.”
Maternal vaccination might also be implemented soon to protect against other diseases including respiratory syncytial virus and group B streptococcal disease after promising study data, she said.
As with pertussis, the potential “blunting” effect will need to be carefully monitored, she said, “as was done in the carefully conducted study of pertussis reported in this issue of Pediatrics.”
One coauthor has received institutional honoraria for participation in advisory groups for Merck Sharpe & Dohme and Pfizer unrelated to this work. Another coauthor was supported by scholarships provided by the Wesfarmers Centre of Vaccines and Infectious Disease at the Telethon Kids Institute. Dr. Edwards reported receiving grants from the CDC and consulting for Bionet, Dynavax, and IBM. She is a member of the data safety and monitoring board for Sanofi, X-4 Pharma, Seqirus, Moderna, Pfizer, Merck, Roche, Novavax, and Brighton Collaboration.
FROM PEDIATRICS
History of heart transplant tied to worse pregnancy outcome
TOPLINE:
than do other pregnant women, results of a large study with a nationwide sample suggest.
METHODOLOGY:
- The retrospective cohort study included 2010-2020 information from the Nationwide Readmissions Database (NRD), a large, all-payer administrative dataset that allows for tracking of patient hospital readmissions in the same U.S. state within the same calendar year and includes patient demographics, hospital characteristics, diagnosis and procedure codes (including for cardiac transplants), length of stay, and discharge disposition.
- The primary outcome was nontransfusion SMM which, among other conditions, included acute myocardial infarction, aortic aneurysm, acute renal failure, adult respiratory distress syndrome, amniotic fluid embolism, cardiac arrest/ventricular fibrillation, and heart failure/arrest, during the delivery hospitalization.
- Additional outcomes included rates of all SMMs (including transfusion), a composite cardiovascular SMM (cSMM) outcome that included acute myocardial infarction, aortic aneurysm, cardiac arrest/ventricular fibrillation, cardioversion, and acute heart failure, preterm birth, and readmission rates.
TAKEAWAY:
- From 2010 to 2020, there were 19,399,521 hospital deliveries, of which, 105 were in HT recipients.
- In unadjusted comparisons, rates of all outcomes were higher in HT, compared with non-HT delivery hospitalizations, and after adjusting for age, demographic and facility characteristics, comorbid conditions, and calendar year, HT recipients continued to have higher odds of adverse maternal outcomes. For example, HT recipients had higher rates of nontransfusion SMM (adjusted odds ratio, 28.12; 95% confidence interval, 15.65-50.53), all SMM (aOR, 15.73; 95% CI, 9.17-27.00), cSMM (aOR, 37.7; 95% CI, 17.39-82.01), and preterm birth (aOR, 7.15; 95%, CI 4.75-10.77).
- HT recipients also had longer hospital stays and higher rates of cesarean delivery, although the authors noted that it’s unclear whether this increase was caused by the HT or complications of pregnancy because data were unavailable regarding indication for cesareans.
- Patients with HT were also at increased risk for hospital readmission within the first year after delivery, particularly within the first 6 months, including for HT-related complications, a finding that supports guidelines recommending an initial postpartum visit within 7-14 days of discharge for patients with cardiac conditions, write the authors.
IN PRACTICE:
The findings demonstrate the importance of counseling HT patients at early gestational ages “to provide information about anticipated risks in pregnancy and the postpartum period to allow patients the opportunity to make informed choices regarding their reproductive options,” the authors conclude.
SOURCE:
The study was conducted by Amanda M. Craig, MD, division of maternal fetal medicine, department of obstetrics and gynecology, Duke University Medical Center, Durham, N.C., and colleagues. It was published online in JACC Heart Failure.
LIMITATIONS:
Relying on diagnosis and procedure codes in administrative datasets like NRD may result in underestimation of outcomes. In this study, outcomes were limited to delivery hospitalizations, which may underestimate the true incidence of complications or fail to include pregnancies that didn’t end in a delivery, including pregnancy terminations or spontaneous abortions. Information related to race, ethnicity, hospital regions, and cause of death are not captured in the NRD dataset.
DISCLOSURES:
The authors have no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
than do other pregnant women, results of a large study with a nationwide sample suggest.
METHODOLOGY:
- The retrospective cohort study included 2010-2020 information from the Nationwide Readmissions Database (NRD), a large, all-payer administrative dataset that allows for tracking of patient hospital readmissions in the same U.S. state within the same calendar year and includes patient demographics, hospital characteristics, diagnosis and procedure codes (including for cardiac transplants), length of stay, and discharge disposition.
- The primary outcome was nontransfusion SMM which, among other conditions, included acute myocardial infarction, aortic aneurysm, acute renal failure, adult respiratory distress syndrome, amniotic fluid embolism, cardiac arrest/ventricular fibrillation, and heart failure/arrest, during the delivery hospitalization.
- Additional outcomes included rates of all SMMs (including transfusion), a composite cardiovascular SMM (cSMM) outcome that included acute myocardial infarction, aortic aneurysm, cardiac arrest/ventricular fibrillation, cardioversion, and acute heart failure, preterm birth, and readmission rates.
TAKEAWAY:
- From 2010 to 2020, there were 19,399,521 hospital deliveries, of which, 105 were in HT recipients.
- In unadjusted comparisons, rates of all outcomes were higher in HT, compared with non-HT delivery hospitalizations, and after adjusting for age, demographic and facility characteristics, comorbid conditions, and calendar year, HT recipients continued to have higher odds of adverse maternal outcomes. For example, HT recipients had higher rates of nontransfusion SMM (adjusted odds ratio, 28.12; 95% confidence interval, 15.65-50.53), all SMM (aOR, 15.73; 95% CI, 9.17-27.00), cSMM (aOR, 37.7; 95% CI, 17.39-82.01), and preterm birth (aOR, 7.15; 95%, CI 4.75-10.77).
- HT recipients also had longer hospital stays and higher rates of cesarean delivery, although the authors noted that it’s unclear whether this increase was caused by the HT or complications of pregnancy because data were unavailable regarding indication for cesareans.
- Patients with HT were also at increased risk for hospital readmission within the first year after delivery, particularly within the first 6 months, including for HT-related complications, a finding that supports guidelines recommending an initial postpartum visit within 7-14 days of discharge for patients with cardiac conditions, write the authors.
IN PRACTICE:
The findings demonstrate the importance of counseling HT patients at early gestational ages “to provide information about anticipated risks in pregnancy and the postpartum period to allow patients the opportunity to make informed choices regarding their reproductive options,” the authors conclude.
SOURCE:
The study was conducted by Amanda M. Craig, MD, division of maternal fetal medicine, department of obstetrics and gynecology, Duke University Medical Center, Durham, N.C., and colleagues. It was published online in JACC Heart Failure.
LIMITATIONS:
Relying on diagnosis and procedure codes in administrative datasets like NRD may result in underestimation of outcomes. In this study, outcomes were limited to delivery hospitalizations, which may underestimate the true incidence of complications or fail to include pregnancies that didn’t end in a delivery, including pregnancy terminations or spontaneous abortions. Information related to race, ethnicity, hospital regions, and cause of death are not captured in the NRD dataset.
DISCLOSURES:
The authors have no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
than do other pregnant women, results of a large study with a nationwide sample suggest.
METHODOLOGY:
- The retrospective cohort study included 2010-2020 information from the Nationwide Readmissions Database (NRD), a large, all-payer administrative dataset that allows for tracking of patient hospital readmissions in the same U.S. state within the same calendar year and includes patient demographics, hospital characteristics, diagnosis and procedure codes (including for cardiac transplants), length of stay, and discharge disposition.
- The primary outcome was nontransfusion SMM which, among other conditions, included acute myocardial infarction, aortic aneurysm, acute renal failure, adult respiratory distress syndrome, amniotic fluid embolism, cardiac arrest/ventricular fibrillation, and heart failure/arrest, during the delivery hospitalization.
- Additional outcomes included rates of all SMMs (including transfusion), a composite cardiovascular SMM (cSMM) outcome that included acute myocardial infarction, aortic aneurysm, cardiac arrest/ventricular fibrillation, cardioversion, and acute heart failure, preterm birth, and readmission rates.
TAKEAWAY:
- From 2010 to 2020, there were 19,399,521 hospital deliveries, of which, 105 were in HT recipients.
- In unadjusted comparisons, rates of all outcomes were higher in HT, compared with non-HT delivery hospitalizations, and after adjusting for age, demographic and facility characteristics, comorbid conditions, and calendar year, HT recipients continued to have higher odds of adverse maternal outcomes. For example, HT recipients had higher rates of nontransfusion SMM (adjusted odds ratio, 28.12; 95% confidence interval, 15.65-50.53), all SMM (aOR, 15.73; 95% CI, 9.17-27.00), cSMM (aOR, 37.7; 95% CI, 17.39-82.01), and preterm birth (aOR, 7.15; 95%, CI 4.75-10.77).
- HT recipients also had longer hospital stays and higher rates of cesarean delivery, although the authors noted that it’s unclear whether this increase was caused by the HT or complications of pregnancy because data were unavailable regarding indication for cesareans.
- Patients with HT were also at increased risk for hospital readmission within the first year after delivery, particularly within the first 6 months, including for HT-related complications, a finding that supports guidelines recommending an initial postpartum visit within 7-14 days of discharge for patients with cardiac conditions, write the authors.
IN PRACTICE:
The findings demonstrate the importance of counseling HT patients at early gestational ages “to provide information about anticipated risks in pregnancy and the postpartum period to allow patients the opportunity to make informed choices regarding their reproductive options,” the authors conclude.
SOURCE:
The study was conducted by Amanda M. Craig, MD, division of maternal fetal medicine, department of obstetrics and gynecology, Duke University Medical Center, Durham, N.C., and colleagues. It was published online in JACC Heart Failure.
LIMITATIONS:
Relying on diagnosis and procedure codes in administrative datasets like NRD may result in underestimation of outcomes. In this study, outcomes were limited to delivery hospitalizations, which may underestimate the true incidence of complications or fail to include pregnancies that didn’t end in a delivery, including pregnancy terminations or spontaneous abortions. Information related to race, ethnicity, hospital regions, and cause of death are not captured in the NRD dataset.
DISCLOSURES:
The authors have no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Metformin treatment shows benefit in gestational diabetes
HAMBURG –
Overall, the trial’s primary outcome, a composite of insulin initiation or a fasting glucose level ≥ 5.1 mmol/L (92 mg/dL) at gestation weeks 32 or 38, did not differ between women with gestational diabetes randomly assigned to either placebo or metformin. However, women taking metformin were significantly less likely to require insulin and had significantly lower fasting blood glucose levels at weeks 32 and 38.
“With a composite outcome it’s more difficult to find a positive result ... So, although the primary composite outcome was not positive, the components of the primary outcome that are clinically meaningful were positive,” lead study author Fidelma Dunne, PhD, professor and endocrine consultant at the University of Galway, Ireland, said in an interview.
There were no differences in maternal or neonatal morbidities, but there was a nonsignificant increase in small for gestational age (SGA), a finding that has been seen in some but not all previous studies of metformin use in gestational diabetes.
Dr. Dunne presented the findings on Oct. 3 at the annual meeting of the European Association for the Study of Diabetes. The results were simultaneously published in JAMA.
Current recommendations from the United Kingdom’s National Institute for Health and Care Excellence say metformin is a suitable first-line therapy for gestational diabetes. However, both the American Diabetes Association and the Society of Maternal-Fetal Medicine do not, particularly for pregnancies with hypertension or preeclampsia or in those who are at risk for intrauterine growth restriction.
“Gestational diabetes is now reaching epidemic proportions. And of course, the vast majority of these women are in low- and middle-income countries where insulin might not be available, or the storage may not allow it to be used effectively. If you have a medication that in the majority of women is safe and effective it may actually help a lot of women in [those regions],” Dr. Dunne said.
Moreover, she noted, “women with gestational diabetes are testing their sugar with finger pricks four to seven times per day and we ask them to take insulin one to four times a day. So if you can relieve any of that pain related to treatment of their condition than that is a benefit for the women as well.”
Asked to comment, Katrien Benhalima, MD, PhD, of University Hospital Gasthuisberg, KU Leuven, Belgium, said, “I think it’s an interesting study because they investigated something novel, to initiate immediately metformin or placebo. Normally what we do with gestational diabetes is once we get the diagnosis, we treat them with lifestyle, and if that’s insufficient then we start with medical therapy. So this is a novel approach.”
She also agreed with Dr. Dunne that the lack of significance for the primary outcome “isn’t an issue of power but it is a composite outcome. If you look at the individual outcomes, as can be expected, the women taking metformin had less need for insulin treatment.”
But, Dr. Benhalima said, the study still leaves open the SGA issue. “It wasn’t significant, but it’s still something we are worried about in the sense that we feel we need more data, especially in the long-term for the offspring health ... You really need to follow them for 10 years or longer to see an effect.”
So for now, Dr. Benhalima said that she wouldn’t use metformin as a first-line treatment for gestational diabetes. “Normally if lifestyle isn’t enough we will still start insulin ... Another issue is why would you offer everybody medical treatment when pregnancy outcomes can be met with lifestyle alone?”
Then again, she added, “of course metformin is easier than an injection. Treatment satisfaction is improved, and the cost is less.”
Primary outcome didn’t differ, but study findings point toward metformin benefit
The double-blind, placebo-controlled trial was conducted at two sites in Ireland, with 510 individuals (535 gestational diabetes pregnancies) enrolled between June 2017 and September 2022. In addition to usual care, they were randomly assigned 1:1 to either placebo or metformin (maximum 2,500 mg) at the time of gestational diabetes diagnosis and continued until delivery.
The primary outcome, a composite of insulin initiation or a fasting glucose ≥ 5.1 mmol/L at gestation weeks 32 or 38, did not differ significantly between the two groups, with risk ratio 0.89 (P = 0.13).
Insulin initiation occurred in 38.4% of the metformin and 51.1% of the placebo groups (relative risk, 0.75, P = .004). The amount of insulin required at the last assessment prior to delivery did not differ between the two groups (P = .17).
Mean fasting glucose was significantly lower with metformin vs. placebo at gestational week 32 (4.9 vs. 5.0 mmol/L; P = .03) and at gestational week 38 (4.5 vs 4.7 mmol/L; P < .001).
On average, those in the metformin group gained less weight between randomization and delivery (0.8 kg vs. 2.0 kg; P = .003).
Gestational week at delivery didn’t differ between the groups, both 39.1 weeks, nor did preterm births prior to 37 weeks’ gestation (9.2% metformin vs. 6.5% placebo; P = .33) or any other pregnancy-related complications.
More participants in the metformin group said that they would choose the drug compared with placebo (76.2% vs. 67.1%, P = .04).
Mean birth weight was lower in the metformin group compared with placebo, 3,393 g vs. 3,506 g (P = .005), with fewer weighing > 4,000 g (7.6% vs. 14.8%; P = .02) or being large for gestational age, i.e., above the 90th percentile (6.5% vs. 14.9%; P = .003).
Proportions of offspring that were SGA (less than 10th percentile) were 5.7% in the metformin group vs. 2.7% with placebo (P = .13).
There were no other significant differences in neonatal variables.
Dr. Dunne told this news organization that her group has recently received funding for long-term follow-up of the SGA offspring. “As other papers have pointed out, if there’s any hint of SGA that’s really important to follow up. So we’re now beginning our longitudinal follow up of the mother and infants to see if the small number that were SGA will in fact turn out to have an increase in body mass index and weight in their childhood and adolescent years.”
The trial was funded by the Health Review Board (HRB) of Ireland, coordinated by the HRB-Clinical Research Facility Galway, and sponsored by the University of Galway, Ireland. Metformin and matched placebo were provided by Merck Healthcare KGaA, Darmstadt, Germany (operating as EMD Serono in the United States), and blood glucose monitoring strips were provided by Ascensia.
Dr. Dunne reported nonfinancial support from Merck and matched placebo and nonfinancial support from Ascensia during the conduct of the study. Dr. Benhalima receives research funds from Flemish Research Fund, study medication from Novo Nordisk, and devices and unrestricted grants from Medtronic and Dexcom.
A version of this article appeared on Medscape.com.
HAMBURG –
Overall, the trial’s primary outcome, a composite of insulin initiation or a fasting glucose level ≥ 5.1 mmol/L (92 mg/dL) at gestation weeks 32 or 38, did not differ between women with gestational diabetes randomly assigned to either placebo or metformin. However, women taking metformin were significantly less likely to require insulin and had significantly lower fasting blood glucose levels at weeks 32 and 38.
“With a composite outcome it’s more difficult to find a positive result ... So, although the primary composite outcome was not positive, the components of the primary outcome that are clinically meaningful were positive,” lead study author Fidelma Dunne, PhD, professor and endocrine consultant at the University of Galway, Ireland, said in an interview.
There were no differences in maternal or neonatal morbidities, but there was a nonsignificant increase in small for gestational age (SGA), a finding that has been seen in some but not all previous studies of metformin use in gestational diabetes.
Dr. Dunne presented the findings on Oct. 3 at the annual meeting of the European Association for the Study of Diabetes. The results were simultaneously published in JAMA.
Current recommendations from the United Kingdom’s National Institute for Health and Care Excellence say metformin is a suitable first-line therapy for gestational diabetes. However, both the American Diabetes Association and the Society of Maternal-Fetal Medicine do not, particularly for pregnancies with hypertension or preeclampsia or in those who are at risk for intrauterine growth restriction.
“Gestational diabetes is now reaching epidemic proportions. And of course, the vast majority of these women are in low- and middle-income countries where insulin might not be available, or the storage may not allow it to be used effectively. If you have a medication that in the majority of women is safe and effective it may actually help a lot of women in [those regions],” Dr. Dunne said.
Moreover, she noted, “women with gestational diabetes are testing their sugar with finger pricks four to seven times per day and we ask them to take insulin one to four times a day. So if you can relieve any of that pain related to treatment of their condition than that is a benefit for the women as well.”
Asked to comment, Katrien Benhalima, MD, PhD, of University Hospital Gasthuisberg, KU Leuven, Belgium, said, “I think it’s an interesting study because they investigated something novel, to initiate immediately metformin or placebo. Normally what we do with gestational diabetes is once we get the diagnosis, we treat them with lifestyle, and if that’s insufficient then we start with medical therapy. So this is a novel approach.”
She also agreed with Dr. Dunne that the lack of significance for the primary outcome “isn’t an issue of power but it is a composite outcome. If you look at the individual outcomes, as can be expected, the women taking metformin had less need for insulin treatment.”
But, Dr. Benhalima said, the study still leaves open the SGA issue. “It wasn’t significant, but it’s still something we are worried about in the sense that we feel we need more data, especially in the long-term for the offspring health ... You really need to follow them for 10 years or longer to see an effect.”
So for now, Dr. Benhalima said that she wouldn’t use metformin as a first-line treatment for gestational diabetes. “Normally if lifestyle isn’t enough we will still start insulin ... Another issue is why would you offer everybody medical treatment when pregnancy outcomes can be met with lifestyle alone?”
Then again, she added, “of course metformin is easier than an injection. Treatment satisfaction is improved, and the cost is less.”
Primary outcome didn’t differ, but study findings point toward metformin benefit
The double-blind, placebo-controlled trial was conducted at two sites in Ireland, with 510 individuals (535 gestational diabetes pregnancies) enrolled between June 2017 and September 2022. In addition to usual care, they were randomly assigned 1:1 to either placebo or metformin (maximum 2,500 mg) at the time of gestational diabetes diagnosis and continued until delivery.
The primary outcome, a composite of insulin initiation or a fasting glucose ≥ 5.1 mmol/L at gestation weeks 32 or 38, did not differ significantly between the two groups, with risk ratio 0.89 (P = 0.13).
Insulin initiation occurred in 38.4% of the metformin and 51.1% of the placebo groups (relative risk, 0.75, P = .004). The amount of insulin required at the last assessment prior to delivery did not differ between the two groups (P = .17).
Mean fasting glucose was significantly lower with metformin vs. placebo at gestational week 32 (4.9 vs. 5.0 mmol/L; P = .03) and at gestational week 38 (4.5 vs 4.7 mmol/L; P < .001).
On average, those in the metformin group gained less weight between randomization and delivery (0.8 kg vs. 2.0 kg; P = .003).
Gestational week at delivery didn’t differ between the groups, both 39.1 weeks, nor did preterm births prior to 37 weeks’ gestation (9.2% metformin vs. 6.5% placebo; P = .33) or any other pregnancy-related complications.
More participants in the metformin group said that they would choose the drug compared with placebo (76.2% vs. 67.1%, P = .04).
Mean birth weight was lower in the metformin group compared with placebo, 3,393 g vs. 3,506 g (P = .005), with fewer weighing > 4,000 g (7.6% vs. 14.8%; P = .02) or being large for gestational age, i.e., above the 90th percentile (6.5% vs. 14.9%; P = .003).
Proportions of offspring that were SGA (less than 10th percentile) were 5.7% in the metformin group vs. 2.7% with placebo (P = .13).
There were no other significant differences in neonatal variables.
Dr. Dunne told this news organization that her group has recently received funding for long-term follow-up of the SGA offspring. “As other papers have pointed out, if there’s any hint of SGA that’s really important to follow up. So we’re now beginning our longitudinal follow up of the mother and infants to see if the small number that were SGA will in fact turn out to have an increase in body mass index and weight in their childhood and adolescent years.”
The trial was funded by the Health Review Board (HRB) of Ireland, coordinated by the HRB-Clinical Research Facility Galway, and sponsored by the University of Galway, Ireland. Metformin and matched placebo were provided by Merck Healthcare KGaA, Darmstadt, Germany (operating as EMD Serono in the United States), and blood glucose monitoring strips were provided by Ascensia.
Dr. Dunne reported nonfinancial support from Merck and matched placebo and nonfinancial support from Ascensia during the conduct of the study. Dr. Benhalima receives research funds from Flemish Research Fund, study medication from Novo Nordisk, and devices and unrestricted grants from Medtronic and Dexcom.
A version of this article appeared on Medscape.com.
HAMBURG –
Overall, the trial’s primary outcome, a composite of insulin initiation or a fasting glucose level ≥ 5.1 mmol/L (92 mg/dL) at gestation weeks 32 or 38, did not differ between women with gestational diabetes randomly assigned to either placebo or metformin. However, women taking metformin were significantly less likely to require insulin and had significantly lower fasting blood glucose levels at weeks 32 and 38.
“With a composite outcome it’s more difficult to find a positive result ... So, although the primary composite outcome was not positive, the components of the primary outcome that are clinically meaningful were positive,” lead study author Fidelma Dunne, PhD, professor and endocrine consultant at the University of Galway, Ireland, said in an interview.
There were no differences in maternal or neonatal morbidities, but there was a nonsignificant increase in small for gestational age (SGA), a finding that has been seen in some but not all previous studies of metformin use in gestational diabetes.
Dr. Dunne presented the findings on Oct. 3 at the annual meeting of the European Association for the Study of Diabetes. The results were simultaneously published in JAMA.
Current recommendations from the United Kingdom’s National Institute for Health and Care Excellence say metformin is a suitable first-line therapy for gestational diabetes. However, both the American Diabetes Association and the Society of Maternal-Fetal Medicine do not, particularly for pregnancies with hypertension or preeclampsia or in those who are at risk for intrauterine growth restriction.
“Gestational diabetes is now reaching epidemic proportions. And of course, the vast majority of these women are in low- and middle-income countries where insulin might not be available, or the storage may not allow it to be used effectively. If you have a medication that in the majority of women is safe and effective it may actually help a lot of women in [those regions],” Dr. Dunne said.
Moreover, she noted, “women with gestational diabetes are testing their sugar with finger pricks four to seven times per day and we ask them to take insulin one to four times a day. So if you can relieve any of that pain related to treatment of their condition than that is a benefit for the women as well.”
Asked to comment, Katrien Benhalima, MD, PhD, of University Hospital Gasthuisberg, KU Leuven, Belgium, said, “I think it’s an interesting study because they investigated something novel, to initiate immediately metformin or placebo. Normally what we do with gestational diabetes is once we get the diagnosis, we treat them with lifestyle, and if that’s insufficient then we start with medical therapy. So this is a novel approach.”
She also agreed with Dr. Dunne that the lack of significance for the primary outcome “isn’t an issue of power but it is a composite outcome. If you look at the individual outcomes, as can be expected, the women taking metformin had less need for insulin treatment.”
But, Dr. Benhalima said, the study still leaves open the SGA issue. “It wasn’t significant, but it’s still something we are worried about in the sense that we feel we need more data, especially in the long-term for the offspring health ... You really need to follow them for 10 years or longer to see an effect.”
So for now, Dr. Benhalima said that she wouldn’t use metformin as a first-line treatment for gestational diabetes. “Normally if lifestyle isn’t enough we will still start insulin ... Another issue is why would you offer everybody medical treatment when pregnancy outcomes can be met with lifestyle alone?”
Then again, she added, “of course metformin is easier than an injection. Treatment satisfaction is improved, and the cost is less.”
Primary outcome didn’t differ, but study findings point toward metformin benefit
The double-blind, placebo-controlled trial was conducted at two sites in Ireland, with 510 individuals (535 gestational diabetes pregnancies) enrolled between June 2017 and September 2022. In addition to usual care, they were randomly assigned 1:1 to either placebo or metformin (maximum 2,500 mg) at the time of gestational diabetes diagnosis and continued until delivery.
The primary outcome, a composite of insulin initiation or a fasting glucose ≥ 5.1 mmol/L at gestation weeks 32 or 38, did not differ significantly between the two groups, with risk ratio 0.89 (P = 0.13).
Insulin initiation occurred in 38.4% of the metformin and 51.1% of the placebo groups (relative risk, 0.75, P = .004). The amount of insulin required at the last assessment prior to delivery did not differ between the two groups (P = .17).
Mean fasting glucose was significantly lower with metformin vs. placebo at gestational week 32 (4.9 vs. 5.0 mmol/L; P = .03) and at gestational week 38 (4.5 vs 4.7 mmol/L; P < .001).
On average, those in the metformin group gained less weight between randomization and delivery (0.8 kg vs. 2.0 kg; P = .003).
Gestational week at delivery didn’t differ between the groups, both 39.1 weeks, nor did preterm births prior to 37 weeks’ gestation (9.2% metformin vs. 6.5% placebo; P = .33) or any other pregnancy-related complications.
More participants in the metformin group said that they would choose the drug compared with placebo (76.2% vs. 67.1%, P = .04).
Mean birth weight was lower in the metformin group compared with placebo, 3,393 g vs. 3,506 g (P = .005), with fewer weighing > 4,000 g (7.6% vs. 14.8%; P = .02) or being large for gestational age, i.e., above the 90th percentile (6.5% vs. 14.9%; P = .003).
Proportions of offspring that were SGA (less than 10th percentile) were 5.7% in the metformin group vs. 2.7% with placebo (P = .13).
There were no other significant differences in neonatal variables.
Dr. Dunne told this news organization that her group has recently received funding for long-term follow-up of the SGA offspring. “As other papers have pointed out, if there’s any hint of SGA that’s really important to follow up. So we’re now beginning our longitudinal follow up of the mother and infants to see if the small number that were SGA will in fact turn out to have an increase in body mass index and weight in their childhood and adolescent years.”
The trial was funded by the Health Review Board (HRB) of Ireland, coordinated by the HRB-Clinical Research Facility Galway, and sponsored by the University of Galway, Ireland. Metformin and matched placebo were provided by Merck Healthcare KGaA, Darmstadt, Germany (operating as EMD Serono in the United States), and blood glucose monitoring strips were provided by Ascensia.
Dr. Dunne reported nonfinancial support from Merck and matched placebo and nonfinancial support from Ascensia during the conduct of the study. Dr. Benhalima receives research funds from Flemish Research Fund, study medication from Novo Nordisk, and devices and unrestricted grants from Medtronic and Dexcom.
A version of this article appeared on Medscape.com.
AT EASD 2023
Substance use in pregnancy linked to adverse CVD outcomes
TOPLINE:
, including more than double the risk of maternal mortality, a new study shows.
METHODOLOGY:
- Using the National Inpatient Sample database to identify hospital deliveries between 2004 and 2018 and diagnostic codes to identify maternal substance use, researchers compared 955,531 pregnancies with accompanying substance use – the most common substances being cannabis and opioids, followed by stimulants – to over 60 million pregnancies in which there was no substance use.
- The primary outcome was any CV event, including acute myocardial infarction, stroke, arrhythmia, endocarditis, any acute cardiomyopathy or heart failure, or cardiac arrest; other outcomes included maternal mortality and major adverse cardiac events (MACE).
TAKEAWAY:
- Deliveries complicated by substance use increased from 1,126 per 100,000 deliveries in 2004 to 1,547 per 100,000 in 2018, peaking at 2,187 per 100,000 in 2014.
- After the researchers controlled for patient demographics and CVD risk factors, results showed that pregnant women who used any substance (cannabis, opioids, methamphetamine, alcohol, tobacco, or cocaine) were more likely to experience a CVD event (adjusted odds ratio [aOR], 1.61; 95% confidence interval [CI], 1.53-1.70; P < .001), MACE (aOR, 1.53; 95% CI, 1.46-1.61; P < .001), or maternal mortality (aOR, 2.65; 95% CI, 2.15-3.25; P < .001) during hospitalization for delivery.
- Those using amphetamine/methamphetamine had ninefold higher odds of cardiomyopathy or heart failure and more than sevenfold higher odds of cardiac arrest.
IN PRACTICE:
“For the wellbeing of pregnant women and their children, substance use needs to be considered an independent risk factor for CV events in pregnancy,” the authors wrote. They called for prenatal assessments by a multidisciplinary cardio-obstetrics team to try to decrease cardiac complications.
In an accompanying editorial by Abha Khandelwal, MD, department of medicine, Stanford (Calif.) University, and others, the authors said the findings “highlight the critical support required during pregnancy and postpartum” for substance users, which should include comprehensive medical care and social services as well as access to addiction medicine and treatment of co-occurring mental health disorders.
SOURCE:
The study was carried out by Kari Evans, MD, division of maternal fetal medicine, department of obstetrics and gynecology, University of Arizona, Phoenix. It was published online in the Journal of the American College of Cardiology: Advances.
LIMITATIONS:
Use of administrative databases may have resulted in underreporting of diagnoses. The researchers could not assess the association of dose, duration, method, or timing of use for any substance with CV events. They also could not examine the effect of vaping on maternal CV events or differentiate hospitalizations for delivery that were complicated by CV events from hospitalizations for CV events that prompted delivery. The data did not reflect the postpartum period, during which a high rate of adverse CV events occurs.
DISCLOSURES:
The authors and editorial writers have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
, including more than double the risk of maternal mortality, a new study shows.
METHODOLOGY:
- Using the National Inpatient Sample database to identify hospital deliveries between 2004 and 2018 and diagnostic codes to identify maternal substance use, researchers compared 955,531 pregnancies with accompanying substance use – the most common substances being cannabis and opioids, followed by stimulants – to over 60 million pregnancies in which there was no substance use.
- The primary outcome was any CV event, including acute myocardial infarction, stroke, arrhythmia, endocarditis, any acute cardiomyopathy or heart failure, or cardiac arrest; other outcomes included maternal mortality and major adverse cardiac events (MACE).
TAKEAWAY:
- Deliveries complicated by substance use increased from 1,126 per 100,000 deliveries in 2004 to 1,547 per 100,000 in 2018, peaking at 2,187 per 100,000 in 2014.
- After the researchers controlled for patient demographics and CVD risk factors, results showed that pregnant women who used any substance (cannabis, opioids, methamphetamine, alcohol, tobacco, or cocaine) were more likely to experience a CVD event (adjusted odds ratio [aOR], 1.61; 95% confidence interval [CI], 1.53-1.70; P < .001), MACE (aOR, 1.53; 95% CI, 1.46-1.61; P < .001), or maternal mortality (aOR, 2.65; 95% CI, 2.15-3.25; P < .001) during hospitalization for delivery.
- Those using amphetamine/methamphetamine had ninefold higher odds of cardiomyopathy or heart failure and more than sevenfold higher odds of cardiac arrest.
IN PRACTICE:
“For the wellbeing of pregnant women and their children, substance use needs to be considered an independent risk factor for CV events in pregnancy,” the authors wrote. They called for prenatal assessments by a multidisciplinary cardio-obstetrics team to try to decrease cardiac complications.
In an accompanying editorial by Abha Khandelwal, MD, department of medicine, Stanford (Calif.) University, and others, the authors said the findings “highlight the critical support required during pregnancy and postpartum” for substance users, which should include comprehensive medical care and social services as well as access to addiction medicine and treatment of co-occurring mental health disorders.
SOURCE:
The study was carried out by Kari Evans, MD, division of maternal fetal medicine, department of obstetrics and gynecology, University of Arizona, Phoenix. It was published online in the Journal of the American College of Cardiology: Advances.
LIMITATIONS:
Use of administrative databases may have resulted in underreporting of diagnoses. The researchers could not assess the association of dose, duration, method, or timing of use for any substance with CV events. They also could not examine the effect of vaping on maternal CV events or differentiate hospitalizations for delivery that were complicated by CV events from hospitalizations for CV events that prompted delivery. The data did not reflect the postpartum period, during which a high rate of adverse CV events occurs.
DISCLOSURES:
The authors and editorial writers have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
, including more than double the risk of maternal mortality, a new study shows.
METHODOLOGY:
- Using the National Inpatient Sample database to identify hospital deliveries between 2004 and 2018 and diagnostic codes to identify maternal substance use, researchers compared 955,531 pregnancies with accompanying substance use – the most common substances being cannabis and opioids, followed by stimulants – to over 60 million pregnancies in which there was no substance use.
- The primary outcome was any CV event, including acute myocardial infarction, stroke, arrhythmia, endocarditis, any acute cardiomyopathy or heart failure, or cardiac arrest; other outcomes included maternal mortality and major adverse cardiac events (MACE).
TAKEAWAY:
- Deliveries complicated by substance use increased from 1,126 per 100,000 deliveries in 2004 to 1,547 per 100,000 in 2018, peaking at 2,187 per 100,000 in 2014.
- After the researchers controlled for patient demographics and CVD risk factors, results showed that pregnant women who used any substance (cannabis, opioids, methamphetamine, alcohol, tobacco, or cocaine) were more likely to experience a CVD event (adjusted odds ratio [aOR], 1.61; 95% confidence interval [CI], 1.53-1.70; P < .001), MACE (aOR, 1.53; 95% CI, 1.46-1.61; P < .001), or maternal mortality (aOR, 2.65; 95% CI, 2.15-3.25; P < .001) during hospitalization for delivery.
- Those using amphetamine/methamphetamine had ninefold higher odds of cardiomyopathy or heart failure and more than sevenfold higher odds of cardiac arrest.
IN PRACTICE:
“For the wellbeing of pregnant women and their children, substance use needs to be considered an independent risk factor for CV events in pregnancy,” the authors wrote. They called for prenatal assessments by a multidisciplinary cardio-obstetrics team to try to decrease cardiac complications.
In an accompanying editorial by Abha Khandelwal, MD, department of medicine, Stanford (Calif.) University, and others, the authors said the findings “highlight the critical support required during pregnancy and postpartum” for substance users, which should include comprehensive medical care and social services as well as access to addiction medicine and treatment of co-occurring mental health disorders.
SOURCE:
The study was carried out by Kari Evans, MD, division of maternal fetal medicine, department of obstetrics and gynecology, University of Arizona, Phoenix. It was published online in the Journal of the American College of Cardiology: Advances.
LIMITATIONS:
Use of administrative databases may have resulted in underreporting of diagnoses. The researchers could not assess the association of dose, duration, method, or timing of use for any substance with CV events. They also could not examine the effect of vaping on maternal CV events or differentiate hospitalizations for delivery that were complicated by CV events from hospitalizations for CV events that prompted delivery. The data did not reflect the postpartum period, during which a high rate of adverse CV events occurs.
DISCLOSURES:
The authors and editorial writers have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
FROM JACC: ADVANCES
Babies conceived during winter/spring may be at higher risk for cerebral palsy
TOPLINE:
Cerebral palsy (CP) affects 1-4 per 1,000 live births in the United States. A new cohort study found Fall months carried about the same or only slightly higher risk of CP than summer months.
METHODOLOGY:
- Researchers examined data from nearly 4.5 million live births registered in California between 2007 and 2015, exploring if the season of conception could serve as an indicator of exposure to environmental risk factors.
- For instance, infants conceived in winter months may have higher exposure to viruses like influenza. In California, agricultural pesticides are most often applied in summer months, when pregnant people would be in their first or second trimester and receive their most exposure to the fine particulates, the authors hypothesize.
- Almost 4,700 babies in the study population were diagnosed with CP. Researchers also considered the role of preterm birth as a potential mediating factor, and adjusted for sociodemographic characteristics such as maternal age, race, education, smoking during pregnancy, and body mass index.
TAKEAWAY:
- The study found that children conceived in winter and spring had a 9% (95% confidence interval, 1.01-1.19) to 10% (95% CI, 1.02-1.20) higher risk of developing CP than those conceived in the summer.
- Children conceived in January, February, or May carried a 15% higher risk, compared with babies conceived in July.
- The risk was more pronounced among mothers with low education levels or living in neighborhoods where residents have high rates of unemployment, single-parent households, multiunit households, and lower rates of high school graduates.
IN PRACTICE:
The researchers noted that possible explanations for the seasonal link to CP risk may include the prevalence of maternal infections during pregnancy, variations in exposure to pesticides, and seasonal patterns for air pollution. “Investigating seasonal variations in disease occurrence can provide clues about etiologically relevant factors.”
SOURCE:
Lead author Haoran Zhou, MPH, Yale University, New Haven, Conn., and colleagues published their findings online in JAMA Network Open. The study was partly supported by a grant from the American Academy for Cerebral Palsy and Developmental Medicine.
LIMITATIONS:
The study may not have fully captured all children with CP in the cohort due to the possibility of misclassification. The findings may not be generalizable beyond California. The overall increased risk associated with the season of conception was relatively small, suggesting family planning strategies may not need to change based on these findings. The exact mechanisms involving potential environmental factors need further investigation.
DISCLOSURES:
The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE:
Cerebral palsy (CP) affects 1-4 per 1,000 live births in the United States. A new cohort study found Fall months carried about the same or only slightly higher risk of CP than summer months.
METHODOLOGY:
- Researchers examined data from nearly 4.5 million live births registered in California between 2007 and 2015, exploring if the season of conception could serve as an indicator of exposure to environmental risk factors.
- For instance, infants conceived in winter months may have higher exposure to viruses like influenza. In California, agricultural pesticides are most often applied in summer months, when pregnant people would be in their first or second trimester and receive their most exposure to the fine particulates, the authors hypothesize.
- Almost 4,700 babies in the study population were diagnosed with CP. Researchers also considered the role of preterm birth as a potential mediating factor, and adjusted for sociodemographic characteristics such as maternal age, race, education, smoking during pregnancy, and body mass index.
TAKEAWAY:
- The study found that children conceived in winter and spring had a 9% (95% confidence interval, 1.01-1.19) to 10% (95% CI, 1.02-1.20) higher risk of developing CP than those conceived in the summer.
- Children conceived in January, February, or May carried a 15% higher risk, compared with babies conceived in July.
- The risk was more pronounced among mothers with low education levels or living in neighborhoods where residents have high rates of unemployment, single-parent households, multiunit households, and lower rates of high school graduates.
IN PRACTICE:
The researchers noted that possible explanations for the seasonal link to CP risk may include the prevalence of maternal infections during pregnancy, variations in exposure to pesticides, and seasonal patterns for air pollution. “Investigating seasonal variations in disease occurrence can provide clues about etiologically relevant factors.”
SOURCE:
Lead author Haoran Zhou, MPH, Yale University, New Haven, Conn., and colleagues published their findings online in JAMA Network Open. The study was partly supported by a grant from the American Academy for Cerebral Palsy and Developmental Medicine.
LIMITATIONS:
The study may not have fully captured all children with CP in the cohort due to the possibility of misclassification. The findings may not be generalizable beyond California. The overall increased risk associated with the season of conception was relatively small, suggesting family planning strategies may not need to change based on these findings. The exact mechanisms involving potential environmental factors need further investigation.
DISCLOSURES:
The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE:
Cerebral palsy (CP) affects 1-4 per 1,000 live births in the United States. A new cohort study found Fall months carried about the same or only slightly higher risk of CP than summer months.
METHODOLOGY:
- Researchers examined data from nearly 4.5 million live births registered in California between 2007 and 2015, exploring if the season of conception could serve as an indicator of exposure to environmental risk factors.
- For instance, infants conceived in winter months may have higher exposure to viruses like influenza. In California, agricultural pesticides are most often applied in summer months, when pregnant people would be in their first or second trimester and receive their most exposure to the fine particulates, the authors hypothesize.
- Almost 4,700 babies in the study population were diagnosed with CP. Researchers also considered the role of preterm birth as a potential mediating factor, and adjusted for sociodemographic characteristics such as maternal age, race, education, smoking during pregnancy, and body mass index.
TAKEAWAY:
- The study found that children conceived in winter and spring had a 9% (95% confidence interval, 1.01-1.19) to 10% (95% CI, 1.02-1.20) higher risk of developing CP than those conceived in the summer.
- Children conceived in January, February, or May carried a 15% higher risk, compared with babies conceived in July.
- The risk was more pronounced among mothers with low education levels or living in neighborhoods where residents have high rates of unemployment, single-parent households, multiunit households, and lower rates of high school graduates.
IN PRACTICE:
The researchers noted that possible explanations for the seasonal link to CP risk may include the prevalence of maternal infections during pregnancy, variations in exposure to pesticides, and seasonal patterns for air pollution. “Investigating seasonal variations in disease occurrence can provide clues about etiologically relevant factors.”
SOURCE:
Lead author Haoran Zhou, MPH, Yale University, New Haven, Conn., and colleagues published their findings online in JAMA Network Open. The study was partly supported by a grant from the American Academy for Cerebral Palsy and Developmental Medicine.
LIMITATIONS:
The study may not have fully captured all children with CP in the cohort due to the possibility of misclassification. The findings may not be generalizable beyond California. The overall increased risk associated with the season of conception was relatively small, suggesting family planning strategies may not need to change based on these findings. The exact mechanisms involving potential environmental factors need further investigation.
DISCLOSURES:
The authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN