User login
Breast cancer drug could treat AML
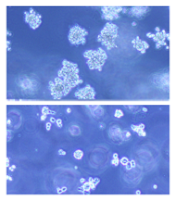
(below) palbociclib treatment
Images courtesy of Iris Uras
and Vetmeduni Vienna
Palbociclib, a CDK4/6 kinase inhibitor approved to treat breast cancer, could also be used to treat acute myeloid leukemia (AML), according to research published in Blood.
The drug induced apoptosis in FLT3-mutant AML cells and inhibited tumor growth in mouse models of FLT3-ITD+ AML.
Palbociclib also demonstrated synergy with a range of FLT3 inhibitors.
The researchers said these results can be explained by the fact that CDK6 is “absolutely required” for the viability of FLT3-dependent leukemic cells and FLT3-ITD-induced leukemogenesis.
“We found a novel therapeutic window that attacks the dependency of a cancer cell on its growth regulator,” said study author Iris Uras, PhD, of Vetmeduni Vienna in Austria.
Dr Uras and her colleagues first found that palbociclib acts specifically on FLT3-ITD+ AML cells, inhibiting their viability in a dose-dependent manner. Palbociclib induced cell-cycle arrest and apoptosis in these cells.
Palbociclib also arrested tumor growth in mouse models of FLT3-ITD+ AML, significantly decreasing tumor size when compared to untreated controls (P<0.05).
Further investigation revealed that CDK6—but not CDK4—directly regulates FLT3 expression.
CDK6 acts as a transcriptional regulator of FLT3 and the serine threonine kinase PIM1, which also plays a role in leukemogenesis. As palbociclib inhibits CDK6, it downregulates FLT3 and reduces PIM1 transcription.
To build upon these findings, the researchers tested palbociclib in combination with FLT3 inhibitors.
They observed “pronounced in vitro synergy” between palbociclib and TCS-359, tandutinib, and quizartinib in FLT3-ITD+ AML cell lines and samples from patients with FLT3-ITD+ AML.
“We are attacking FLT3 from two sides there—blocking its expression and inhibiting its activity,” Dr Uras said. “A combination therapy could be a breakthrough for many patients suffering from leukemia.” ![]()

(below) palbociclib treatment
Images courtesy of Iris Uras
and Vetmeduni Vienna
Palbociclib, a CDK4/6 kinase inhibitor approved to treat breast cancer, could also be used to treat acute myeloid leukemia (AML), according to research published in Blood.
The drug induced apoptosis in FLT3-mutant AML cells and inhibited tumor growth in mouse models of FLT3-ITD+ AML.
Palbociclib also demonstrated synergy with a range of FLT3 inhibitors.
The researchers said these results can be explained by the fact that CDK6 is “absolutely required” for the viability of FLT3-dependent leukemic cells and FLT3-ITD-induced leukemogenesis.
“We found a novel therapeutic window that attacks the dependency of a cancer cell on its growth regulator,” said study author Iris Uras, PhD, of Vetmeduni Vienna in Austria.
Dr Uras and her colleagues first found that palbociclib acts specifically on FLT3-ITD+ AML cells, inhibiting their viability in a dose-dependent manner. Palbociclib induced cell-cycle arrest and apoptosis in these cells.
Palbociclib also arrested tumor growth in mouse models of FLT3-ITD+ AML, significantly decreasing tumor size when compared to untreated controls (P<0.05).
Further investigation revealed that CDK6—but not CDK4—directly regulates FLT3 expression.
CDK6 acts as a transcriptional regulator of FLT3 and the serine threonine kinase PIM1, which also plays a role in leukemogenesis. As palbociclib inhibits CDK6, it downregulates FLT3 and reduces PIM1 transcription.
To build upon these findings, the researchers tested palbociclib in combination with FLT3 inhibitors.
They observed “pronounced in vitro synergy” between palbociclib and TCS-359, tandutinib, and quizartinib in FLT3-ITD+ AML cell lines and samples from patients with FLT3-ITD+ AML.
“We are attacking FLT3 from two sides there—blocking its expression and inhibiting its activity,” Dr Uras said. “A combination therapy could be a breakthrough for many patients suffering from leukemia.” ![]()

(below) palbociclib treatment
Images courtesy of Iris Uras
and Vetmeduni Vienna
Palbociclib, a CDK4/6 kinase inhibitor approved to treat breast cancer, could also be used to treat acute myeloid leukemia (AML), according to research published in Blood.
The drug induced apoptosis in FLT3-mutant AML cells and inhibited tumor growth in mouse models of FLT3-ITD+ AML.
Palbociclib also demonstrated synergy with a range of FLT3 inhibitors.
The researchers said these results can be explained by the fact that CDK6 is “absolutely required” for the viability of FLT3-dependent leukemic cells and FLT3-ITD-induced leukemogenesis.
“We found a novel therapeutic window that attacks the dependency of a cancer cell on its growth regulator,” said study author Iris Uras, PhD, of Vetmeduni Vienna in Austria.
Dr Uras and her colleagues first found that palbociclib acts specifically on FLT3-ITD+ AML cells, inhibiting their viability in a dose-dependent manner. Palbociclib induced cell-cycle arrest and apoptosis in these cells.
Palbociclib also arrested tumor growth in mouse models of FLT3-ITD+ AML, significantly decreasing tumor size when compared to untreated controls (P<0.05).
Further investigation revealed that CDK6—but not CDK4—directly regulates FLT3 expression.
CDK6 acts as a transcriptional regulator of FLT3 and the serine threonine kinase PIM1, which also plays a role in leukemogenesis. As palbociclib inhibits CDK6, it downregulates FLT3 and reduces PIM1 transcription.
To build upon these findings, the researchers tested palbociclib in combination with FLT3 inhibitors.
They observed “pronounced in vitro synergy” between palbociclib and TCS-359, tandutinib, and quizartinib in FLT3-ITD+ AML cell lines and samples from patients with FLT3-ITD+ AML.
“We are attacking FLT3 from two sides there—blocking its expression and inhibiting its activity,” Dr Uras said. “A combination therapy could be a breakthrough for many patients suffering from leukemia.” ![]()
SMAC mimetics could treat relapsed/refractory ALL

Patients with high-risk, relapsed/refractory acute lymphoblastic leukemia (ALL) may be sensitive to treatment with SMAC mimetics, according to researchers.
One SMAC mimetic in particular, birinapant, demonstrated varied activity in samples from ALL patients, but samples from patients with resistant disease were the most sensitive to the drug.
Birinapant also had “marked antileukemic effects” in some mice with ALL.
The researchers found this antileukemic activity was dependent on simultaneous activation of apoptosis and necroptosis.
The team reported these findings in Science Translational Medicine.
“Our research reveals that an alternative cell-death program, necroptosis, can be activated in human ALL cells,” said study author Beat Bornhauser, PhD, of the Children’s Hospital Zurich in Switzerland.
“This enables leukemia cells that barely respond to existing chemotherapeutic drugs to be killed off.”
In vitro and in vivo activity
The researchers tested the efficacy of SMAC mimetics in a set of 51 patient-derived B-cell precursor ALL xenografts, which was enriched for samples from relapsed and drug-resistant disease.
The response to birinapant varied greatly, but samples from high-risk or relapsed patients tended to be highly sensitive to the drug.
The researchers observed similar response profiles with the SMAC mimetic LCL161, although this drug proved less potent than birinapant.
The team also evaluated the antileukemic activity of SMAC mimetics in mouse models of ALL.
Birinapant delayed disease progression and induced complete responses in sensitive ALL cases. LCL161, on the other hand, did not display any in vivo activity.
Determining the mechanism of activity
The researchers used CRISPR-Cas9 to determine how SMAC mimetics fight ALL, and they discovered that the drugs trigger both apoptosis and necroptosis.
If the genes responsible for apoptosis were disabled via genome editing, leukemia cells died due to necroptosis after SMAC mimetics had been administered. If necroptotic genes were disabled, apoptosis led to cell death.
Only the simultaneous deactivation of apoptotic and necroptotic genes resulted in the complete resistance of leukemic cells to SMAC mimetics.
Therefore, the researchers concluded that simultaneous activation of apoptosis and necroptosis is responsible for the strong anti-leukemic effect of SMAC mimetics.
“SMAC mimetics have great potential to eliminate leukemia cells in patients that aren’t sensitive to established chemotherapeutic drugs,” Dr Bornhauser said. “They are effectively a double-edged sword. They kill cells that block apoptosis through necroptosis.”
The researchers are now looking for suitable biomarkers to identify patients who might benefit from treatment with SMAC mimetics in clinical trials. ![]()

Patients with high-risk, relapsed/refractory acute lymphoblastic leukemia (ALL) may be sensitive to treatment with SMAC mimetics, according to researchers.
One SMAC mimetic in particular, birinapant, demonstrated varied activity in samples from ALL patients, but samples from patients with resistant disease were the most sensitive to the drug.
Birinapant also had “marked antileukemic effects” in some mice with ALL.
The researchers found this antileukemic activity was dependent on simultaneous activation of apoptosis and necroptosis.
The team reported these findings in Science Translational Medicine.
“Our research reveals that an alternative cell-death program, necroptosis, can be activated in human ALL cells,” said study author Beat Bornhauser, PhD, of the Children’s Hospital Zurich in Switzerland.
“This enables leukemia cells that barely respond to existing chemotherapeutic drugs to be killed off.”
In vitro and in vivo activity
The researchers tested the efficacy of SMAC mimetics in a set of 51 patient-derived B-cell precursor ALL xenografts, which was enriched for samples from relapsed and drug-resistant disease.
The response to birinapant varied greatly, but samples from high-risk or relapsed patients tended to be highly sensitive to the drug.
The researchers observed similar response profiles with the SMAC mimetic LCL161, although this drug proved less potent than birinapant.
The team also evaluated the antileukemic activity of SMAC mimetics in mouse models of ALL.
Birinapant delayed disease progression and induced complete responses in sensitive ALL cases. LCL161, on the other hand, did not display any in vivo activity.
Determining the mechanism of activity
The researchers used CRISPR-Cas9 to determine how SMAC mimetics fight ALL, and they discovered that the drugs trigger both apoptosis and necroptosis.
If the genes responsible for apoptosis were disabled via genome editing, leukemia cells died due to necroptosis after SMAC mimetics had been administered. If necroptotic genes were disabled, apoptosis led to cell death.
Only the simultaneous deactivation of apoptotic and necroptotic genes resulted in the complete resistance of leukemic cells to SMAC mimetics.
Therefore, the researchers concluded that simultaneous activation of apoptosis and necroptosis is responsible for the strong anti-leukemic effect of SMAC mimetics.
“SMAC mimetics have great potential to eliminate leukemia cells in patients that aren’t sensitive to established chemotherapeutic drugs,” Dr Bornhauser said. “They are effectively a double-edged sword. They kill cells that block apoptosis through necroptosis.”
The researchers are now looking for suitable biomarkers to identify patients who might benefit from treatment with SMAC mimetics in clinical trials. ![]()

Patients with high-risk, relapsed/refractory acute lymphoblastic leukemia (ALL) may be sensitive to treatment with SMAC mimetics, according to researchers.
One SMAC mimetic in particular, birinapant, demonstrated varied activity in samples from ALL patients, but samples from patients with resistant disease were the most sensitive to the drug.
Birinapant also had “marked antileukemic effects” in some mice with ALL.
The researchers found this antileukemic activity was dependent on simultaneous activation of apoptosis and necroptosis.
The team reported these findings in Science Translational Medicine.
“Our research reveals that an alternative cell-death program, necroptosis, can be activated in human ALL cells,” said study author Beat Bornhauser, PhD, of the Children’s Hospital Zurich in Switzerland.
“This enables leukemia cells that barely respond to existing chemotherapeutic drugs to be killed off.”
In vitro and in vivo activity
The researchers tested the efficacy of SMAC mimetics in a set of 51 patient-derived B-cell precursor ALL xenografts, which was enriched for samples from relapsed and drug-resistant disease.
The response to birinapant varied greatly, but samples from high-risk or relapsed patients tended to be highly sensitive to the drug.
The researchers observed similar response profiles with the SMAC mimetic LCL161, although this drug proved less potent than birinapant.
The team also evaluated the antileukemic activity of SMAC mimetics in mouse models of ALL.
Birinapant delayed disease progression and induced complete responses in sensitive ALL cases. LCL161, on the other hand, did not display any in vivo activity.
Determining the mechanism of activity
The researchers used CRISPR-Cas9 to determine how SMAC mimetics fight ALL, and they discovered that the drugs trigger both apoptosis and necroptosis.
If the genes responsible for apoptosis were disabled via genome editing, leukemia cells died due to necroptosis after SMAC mimetics had been administered. If necroptotic genes were disabled, apoptosis led to cell death.
Only the simultaneous deactivation of apoptotic and necroptotic genes resulted in the complete resistance of leukemic cells to SMAC mimetics.
Therefore, the researchers concluded that simultaneous activation of apoptosis and necroptosis is responsible for the strong anti-leukemic effect of SMAC mimetics.
“SMAC mimetics have great potential to eliminate leukemia cells in patients that aren’t sensitive to established chemotherapeutic drugs,” Dr Bornhauser said. “They are effectively a double-edged sword. They kill cells that block apoptosis through necroptosis.”
The researchers are now looking for suitable biomarkers to identify patients who might benefit from treatment with SMAC mimetics in clinical trials. ![]()
Physical activity may lower risk of some cancers

Photo by K. Johansson
Being physically active during leisure time may lower a person’s risk of certain cancers, according to a new study.
A high level of physical activity was associated with a 20% lower risk of myeloid leukemia, a 17% lower risk of myeloma, a 9% lower risk of non-Hodgkin lymphoma, and a 7% lower risk of cancer in general.
On the other hand, a high level of physical activity was also associated with a higher risk of malignant melanoma and prostate cancer.
Steven C. Moore, PhD, of the National Cancer Institute in Bethesda, Maryland, and his colleagues reported these findings in JAMA Internal Medicine.
The researchers pooled data from 12 US and European study cohorts with self-reported physical activity (1987-2004). And they analyzed associations between physical activity and 26 types of cancer.
The study included 1.4 million participants, and 186,932 cancers were identified during a median of 11 years of follow-up.
Compared with the lowest level of leisure-time physical activity (10th percentile), the highest level of activity (90th percentile) had strong inverse associations (a 20% or greater reduction in risk) for 7 cancer types:
- Myeloid leukemia (hazard ratio [HR]=0.80 [95% CI, 0.70-0.92])
- Esophageal adenocarcinoma (HR=0.58 [95% CI, 0.37-0.89])
- Liver cancer (HR=0.73 [95% CI, 0.55-0.98])
- Lung cancer (HR=0.74 [95% CI, 0.71-0.77])
- Kidney cancer (HR=0.77 [95% CI, 0.70-0.85])
- Gastric cardia (HR=0.78 [95% CI, 0.64-0.95])
- Endometrial cancer (HR=0.79 [95% CI, 0.68-0.92]).
There were moderate inverse associations (a 10% to 20% reduction in risk) between the highest level of activity and 6 cancers:
- Myeloma (HR=0.83 [95% CI, 0.72-0.95])
- Colon cancer (HR=0.84 [95% CI, 0.77-0.91])
- Head and neck cancer (HR=0.85 [95% CI, 0.78-0.93])
- Rectal cancer (HR=0.87 [95% CI, 0.80-0.95])
- Bladder cancer (HR=0.87 [95% CI, 0.82-0.92])
- Breast cancer (HR=0.90 [95% CI, 0.87-0.93]).
And there were suggestive inverse associations between the highest level of activity and 3 cancers:
- Non-Hodgkin lymphoma (HR=0.91 [95% CI, 0.83-1.00])
- Gallbladder cancer (HR=0.72 [95% CI, 0.51-1.01])
- Small intestine cancer (HR=0.78 [95% CI, 0.60-1.00]).
However, the highest level of activity was also associated with an increased risk of prostate cancer (HR=1.05 [95% CI, 1.03-1.08]) and malignant melanoma (HR=1.27 [95% CI, 1.16-1.40]).
The researchers said the main limitation of this study is that they cannot fully exclude the possibility that diet, smoking, and other factors may have affected these results. Also, the study used self-reported physical activity, which can mean errors in recall.
Still, the team said these findings support promoting physical activity as a key component of population-wide cancer prevention and control efforts. ![]()

Photo by K. Johansson
Being physically active during leisure time may lower a person’s risk of certain cancers, according to a new study.
A high level of physical activity was associated with a 20% lower risk of myeloid leukemia, a 17% lower risk of myeloma, a 9% lower risk of non-Hodgkin lymphoma, and a 7% lower risk of cancer in general.
On the other hand, a high level of physical activity was also associated with a higher risk of malignant melanoma and prostate cancer.
Steven C. Moore, PhD, of the National Cancer Institute in Bethesda, Maryland, and his colleagues reported these findings in JAMA Internal Medicine.
The researchers pooled data from 12 US and European study cohorts with self-reported physical activity (1987-2004). And they analyzed associations between physical activity and 26 types of cancer.
The study included 1.4 million participants, and 186,932 cancers were identified during a median of 11 years of follow-up.
Compared with the lowest level of leisure-time physical activity (10th percentile), the highest level of activity (90th percentile) had strong inverse associations (a 20% or greater reduction in risk) for 7 cancer types:
- Myeloid leukemia (hazard ratio [HR]=0.80 [95% CI, 0.70-0.92])
- Esophageal adenocarcinoma (HR=0.58 [95% CI, 0.37-0.89])
- Liver cancer (HR=0.73 [95% CI, 0.55-0.98])
- Lung cancer (HR=0.74 [95% CI, 0.71-0.77])
- Kidney cancer (HR=0.77 [95% CI, 0.70-0.85])
- Gastric cardia (HR=0.78 [95% CI, 0.64-0.95])
- Endometrial cancer (HR=0.79 [95% CI, 0.68-0.92]).
There were moderate inverse associations (a 10% to 20% reduction in risk) between the highest level of activity and 6 cancers:
- Myeloma (HR=0.83 [95% CI, 0.72-0.95])
- Colon cancer (HR=0.84 [95% CI, 0.77-0.91])
- Head and neck cancer (HR=0.85 [95% CI, 0.78-0.93])
- Rectal cancer (HR=0.87 [95% CI, 0.80-0.95])
- Bladder cancer (HR=0.87 [95% CI, 0.82-0.92])
- Breast cancer (HR=0.90 [95% CI, 0.87-0.93]).
And there were suggestive inverse associations between the highest level of activity and 3 cancers:
- Non-Hodgkin lymphoma (HR=0.91 [95% CI, 0.83-1.00])
- Gallbladder cancer (HR=0.72 [95% CI, 0.51-1.01])
- Small intestine cancer (HR=0.78 [95% CI, 0.60-1.00]).
However, the highest level of activity was also associated with an increased risk of prostate cancer (HR=1.05 [95% CI, 1.03-1.08]) and malignant melanoma (HR=1.27 [95% CI, 1.16-1.40]).
The researchers said the main limitation of this study is that they cannot fully exclude the possibility that diet, smoking, and other factors may have affected these results. Also, the study used self-reported physical activity, which can mean errors in recall.
Still, the team said these findings support promoting physical activity as a key component of population-wide cancer prevention and control efforts. ![]()

Photo by K. Johansson
Being physically active during leisure time may lower a person’s risk of certain cancers, according to a new study.
A high level of physical activity was associated with a 20% lower risk of myeloid leukemia, a 17% lower risk of myeloma, a 9% lower risk of non-Hodgkin lymphoma, and a 7% lower risk of cancer in general.
On the other hand, a high level of physical activity was also associated with a higher risk of malignant melanoma and prostate cancer.
Steven C. Moore, PhD, of the National Cancer Institute in Bethesda, Maryland, and his colleagues reported these findings in JAMA Internal Medicine.
The researchers pooled data from 12 US and European study cohorts with self-reported physical activity (1987-2004). And they analyzed associations between physical activity and 26 types of cancer.
The study included 1.4 million participants, and 186,932 cancers were identified during a median of 11 years of follow-up.
Compared with the lowest level of leisure-time physical activity (10th percentile), the highest level of activity (90th percentile) had strong inverse associations (a 20% or greater reduction in risk) for 7 cancer types:
- Myeloid leukemia (hazard ratio [HR]=0.80 [95% CI, 0.70-0.92])
- Esophageal adenocarcinoma (HR=0.58 [95% CI, 0.37-0.89])
- Liver cancer (HR=0.73 [95% CI, 0.55-0.98])
- Lung cancer (HR=0.74 [95% CI, 0.71-0.77])
- Kidney cancer (HR=0.77 [95% CI, 0.70-0.85])
- Gastric cardia (HR=0.78 [95% CI, 0.64-0.95])
- Endometrial cancer (HR=0.79 [95% CI, 0.68-0.92]).
There were moderate inverse associations (a 10% to 20% reduction in risk) between the highest level of activity and 6 cancers:
- Myeloma (HR=0.83 [95% CI, 0.72-0.95])
- Colon cancer (HR=0.84 [95% CI, 0.77-0.91])
- Head and neck cancer (HR=0.85 [95% CI, 0.78-0.93])
- Rectal cancer (HR=0.87 [95% CI, 0.80-0.95])
- Bladder cancer (HR=0.87 [95% CI, 0.82-0.92])
- Breast cancer (HR=0.90 [95% CI, 0.87-0.93]).
And there were suggestive inverse associations between the highest level of activity and 3 cancers:
- Non-Hodgkin lymphoma (HR=0.91 [95% CI, 0.83-1.00])
- Gallbladder cancer (HR=0.72 [95% CI, 0.51-1.01])
- Small intestine cancer (HR=0.78 [95% CI, 0.60-1.00]).
However, the highest level of activity was also associated with an increased risk of prostate cancer (HR=1.05 [95% CI, 1.03-1.08]) and malignant melanoma (HR=1.27 [95% CI, 1.16-1.40]).
The researchers said the main limitation of this study is that they cannot fully exclude the possibility that diet, smoking, and other factors may have affected these results. Also, the study used self-reported physical activity, which can mean errors in recall.
Still, the team said these findings support promoting physical activity as a key component of population-wide cancer prevention and control efforts. ![]()
Improving NK cell therapy

Image by Joshua Stokes
New findings published in PNAS may help scientists improve the efficacy of natural killer (NK) cell therapy for patients with leukemia.
The preclinical research revealed a tolerance mechanism that restrains the activity of NK cells, as well as a potential way to overcome this problem.
Investigators found that a transcription factor, Kruppel-like factor 2 (KFL2), is critical for NK cell expansion and survival.
Specifically, KLF2 limits immature NK cell proliferation and instructs mature NK cells to home to niches rich in interleukin 15 (IL-15), which is necessary for their continued survival.
“This is the same process likely used by cancer cells to avoid destruction by NK cells,” said study author Eric Sebzda, PhD, of Vanderbilt University Medical Center in Nashville, Tennessee.
In particular, tumors may avoid immune clearance by promoting KLF2 destruction within the NK cell population, thereby starving these cells of IL-15.
Dr Sebzda and his colleagues noted that increased expression of IL-15 can improve immune responses against tumors. Unfortunately, it’s not easy to introduce the cytokine only within a tumor microenvironment, and high systemic levels of IL-15 can be toxic.
Recruiting cells that transpresent IL-15 to the tumor microenvironment may overcome this barrier and therefore improve NK cell-mediated cancer therapy, the investigators said. However, the methodology hasn’t been worked out yet.
“Our paper should encourage this line of inquiry,” Dr Sebzda concluded. ![]()

Image by Joshua Stokes
New findings published in PNAS may help scientists improve the efficacy of natural killer (NK) cell therapy for patients with leukemia.
The preclinical research revealed a tolerance mechanism that restrains the activity of NK cells, as well as a potential way to overcome this problem.
Investigators found that a transcription factor, Kruppel-like factor 2 (KFL2), is critical for NK cell expansion and survival.
Specifically, KLF2 limits immature NK cell proliferation and instructs mature NK cells to home to niches rich in interleukin 15 (IL-15), which is necessary for their continued survival.
“This is the same process likely used by cancer cells to avoid destruction by NK cells,” said study author Eric Sebzda, PhD, of Vanderbilt University Medical Center in Nashville, Tennessee.
In particular, tumors may avoid immune clearance by promoting KLF2 destruction within the NK cell population, thereby starving these cells of IL-15.
Dr Sebzda and his colleagues noted that increased expression of IL-15 can improve immune responses against tumors. Unfortunately, it’s not easy to introduce the cytokine only within a tumor microenvironment, and high systemic levels of IL-15 can be toxic.
Recruiting cells that transpresent IL-15 to the tumor microenvironment may overcome this barrier and therefore improve NK cell-mediated cancer therapy, the investigators said. However, the methodology hasn’t been worked out yet.
“Our paper should encourage this line of inquiry,” Dr Sebzda concluded. ![]()

Image by Joshua Stokes
New findings published in PNAS may help scientists improve the efficacy of natural killer (NK) cell therapy for patients with leukemia.
The preclinical research revealed a tolerance mechanism that restrains the activity of NK cells, as well as a potential way to overcome this problem.
Investigators found that a transcription factor, Kruppel-like factor 2 (KFL2), is critical for NK cell expansion and survival.
Specifically, KLF2 limits immature NK cell proliferation and instructs mature NK cells to home to niches rich in interleukin 15 (IL-15), which is necessary for their continued survival.
“This is the same process likely used by cancer cells to avoid destruction by NK cells,” said study author Eric Sebzda, PhD, of Vanderbilt University Medical Center in Nashville, Tennessee.
In particular, tumors may avoid immune clearance by promoting KLF2 destruction within the NK cell population, thereby starving these cells of IL-15.
Dr Sebzda and his colleagues noted that increased expression of IL-15 can improve immune responses against tumors. Unfortunately, it’s not easy to introduce the cytokine only within a tumor microenvironment, and high systemic levels of IL-15 can be toxic.
Recruiting cells that transpresent IL-15 to the tumor microenvironment may overcome this barrier and therefore improve NK cell-mediated cancer therapy, the investigators said. However, the methodology hasn’t been worked out yet.
“Our paper should encourage this line of inquiry,” Dr Sebzda concluded. ![]()
Drug may reduce severity of AEs from dexamethasone

Photo by Bill Branson
Adding a physiologic dose of hydrocortisone to treatment with dexamethasone can reduce the severity of certain adverse effects (AEs) in pediatric patients with acute lymphoblastic leukemia (ALL), according to researchers.
Hydrocortisone did not decrease the incidence of psychosocial problems or sleep-related issues in these patients, but the drug did reduce the severity of these problems among patients who experienced them.
Lidewij T. Warris, MD, of Erasmus MC-Sophia Children’s Hospital in Rotterdam, the Netherlands, and her colleagues reported these results in the Journal of Clinical Oncology.
The team conducted this study to determine whether a physiologic dose of hydrocortisone could reduce neuropsychologic and metabolic AEs in children with ALL who were receiving dexamethasone.
The study enrolled 50 patients (ages 3 to 16) who were set to receive 2 consecutive courses of dexamethasone in accordance with Dutch Childhood Oncology Group ALL protocols.
The patients were randomized to receive either hydrocortisone or placebo in a circadian rhythm (10 mg/m2/d) during their first dexamethasone course. During their second course, the patients were assigned to the opposite arm.
The treatment groups were similar with regard to age, type of leukemia, treatment protocol, and CNS status at diagnosis.
Psychosocial problems
The researchers assessed psychosocial problems by having parents complete the Strength and Difficulties Questionnaire (SDQ). Forty-six parents completed the questionnaire at all 4 time points tested.
The results showed that 4 days of dexamethasone treatment significantly increased patient problems, as reported by all SDQ scales and subscales. However, one-third of the population did not have any increase in SDQ total difficulties with dexamethasone.
The addition of hydrocortisone did not affect patients’ total difficulties score (mean difference, -0.8 ± 5.5; P=0.33), emotional symptoms (mean difference, -0.6 ± 2.3; P=0.08), conduct problems (mean difference, 0.0 ± 1.5; P=1.00), or other SDQ subscales.
However, hydrocortisone did have a clinically significant effect in the subset of 16 patients who had clinically relevant dexamethasone-related AEs. This was defined as an increase of ≥5 in their SDQ total difficulties score.
In these patients, hydrocortisone improved the total difficulties delta-score (median difference, -5.0; IQR, -7.8 to -3.0), emotional symptoms score (median difference, -1.5; IQR, -4.0 to -1.0), conduct problems score (median difference, -1.0; IQR, -2.0 to 0.0), and impact of stress score (median difference, -1.0; IQR, -2.0 to 0.0).
Sleep issues
The researchers used the Sleep Disturbance Scale for Children (SDSC) to assess sleep quality and sleep disturbances. Forty-seven parents completed the questionnaire at all 4 time points tested.
Results showed that dexamethasone significantly increased disorders of arousal (P=0.04), sleep-wake transition disorders (P=0.01), and disorders of excessive somnolence (P=0.01).
The addition of hydrocortisone had no significant effect on patients’ total SDSC score (P=0.84), disorders of initiating and maintaining sleep (P=0.74), disorders of excessive somnolence (P=0.29), or sleep-wake transition disorder (P=0.29).
However, hydrocortisone did have a clinically significant effect in the subset of 9 children who had clinically relevant dexamethasone-induced sleeping problems, which were defined as a change of ≥7 in SDSC total score.
Hydrocortisone reduced SDSC total scores (median difference, -11.0; IQR, -16.0 to 0.0) and disorders of initiating and maintaining sleep scores (median difference, -3.0; IQR, -7.0 to –0.5).
Other outcomes
The researchers also found that dexamethasone treatment alone did not affect patients’ attention, visual-spatial functions, memory, or processing speed.
However, the addition of hydrocortisone significantly improved patients’ long-term visual memory (P=0.01).
Hydrocortisone did not have any effect on other neuropsychological tests or on metabolic parameters. ![]()

Photo by Bill Branson
Adding a physiologic dose of hydrocortisone to treatment with dexamethasone can reduce the severity of certain adverse effects (AEs) in pediatric patients with acute lymphoblastic leukemia (ALL), according to researchers.
Hydrocortisone did not decrease the incidence of psychosocial problems or sleep-related issues in these patients, but the drug did reduce the severity of these problems among patients who experienced them.
Lidewij T. Warris, MD, of Erasmus MC-Sophia Children’s Hospital in Rotterdam, the Netherlands, and her colleagues reported these results in the Journal of Clinical Oncology.
The team conducted this study to determine whether a physiologic dose of hydrocortisone could reduce neuropsychologic and metabolic AEs in children with ALL who were receiving dexamethasone.
The study enrolled 50 patients (ages 3 to 16) who were set to receive 2 consecutive courses of dexamethasone in accordance with Dutch Childhood Oncology Group ALL protocols.
The patients were randomized to receive either hydrocortisone or placebo in a circadian rhythm (10 mg/m2/d) during their first dexamethasone course. During their second course, the patients were assigned to the opposite arm.
The treatment groups were similar with regard to age, type of leukemia, treatment protocol, and CNS status at diagnosis.
Psychosocial problems
The researchers assessed psychosocial problems by having parents complete the Strength and Difficulties Questionnaire (SDQ). Forty-six parents completed the questionnaire at all 4 time points tested.
The results showed that 4 days of dexamethasone treatment significantly increased patient problems, as reported by all SDQ scales and subscales. However, one-third of the population did not have any increase in SDQ total difficulties with dexamethasone.
The addition of hydrocortisone did not affect patients’ total difficulties score (mean difference, -0.8 ± 5.5; P=0.33), emotional symptoms (mean difference, -0.6 ± 2.3; P=0.08), conduct problems (mean difference, 0.0 ± 1.5; P=1.00), or other SDQ subscales.
However, hydrocortisone did have a clinically significant effect in the subset of 16 patients who had clinically relevant dexamethasone-related AEs. This was defined as an increase of ≥5 in their SDQ total difficulties score.
In these patients, hydrocortisone improved the total difficulties delta-score (median difference, -5.0; IQR, -7.8 to -3.0), emotional symptoms score (median difference, -1.5; IQR, -4.0 to -1.0), conduct problems score (median difference, -1.0; IQR, -2.0 to 0.0), and impact of stress score (median difference, -1.0; IQR, -2.0 to 0.0).
Sleep issues
The researchers used the Sleep Disturbance Scale for Children (SDSC) to assess sleep quality and sleep disturbances. Forty-seven parents completed the questionnaire at all 4 time points tested.
Results showed that dexamethasone significantly increased disorders of arousal (P=0.04), sleep-wake transition disorders (P=0.01), and disorders of excessive somnolence (P=0.01).
The addition of hydrocortisone had no significant effect on patients’ total SDSC score (P=0.84), disorders of initiating and maintaining sleep (P=0.74), disorders of excessive somnolence (P=0.29), or sleep-wake transition disorder (P=0.29).
However, hydrocortisone did have a clinically significant effect in the subset of 9 children who had clinically relevant dexamethasone-induced sleeping problems, which were defined as a change of ≥7 in SDSC total score.
Hydrocortisone reduced SDSC total scores (median difference, -11.0; IQR, -16.0 to 0.0) and disorders of initiating and maintaining sleep scores (median difference, -3.0; IQR, -7.0 to –0.5).
Other outcomes
The researchers also found that dexamethasone treatment alone did not affect patients’ attention, visual-spatial functions, memory, or processing speed.
However, the addition of hydrocortisone significantly improved patients’ long-term visual memory (P=0.01).
Hydrocortisone did not have any effect on other neuropsychological tests or on metabolic parameters. ![]()

Photo by Bill Branson
Adding a physiologic dose of hydrocortisone to treatment with dexamethasone can reduce the severity of certain adverse effects (AEs) in pediatric patients with acute lymphoblastic leukemia (ALL), according to researchers.
Hydrocortisone did not decrease the incidence of psychosocial problems or sleep-related issues in these patients, but the drug did reduce the severity of these problems among patients who experienced them.
Lidewij T. Warris, MD, of Erasmus MC-Sophia Children’s Hospital in Rotterdam, the Netherlands, and her colleagues reported these results in the Journal of Clinical Oncology.
The team conducted this study to determine whether a physiologic dose of hydrocortisone could reduce neuropsychologic and metabolic AEs in children with ALL who were receiving dexamethasone.
The study enrolled 50 patients (ages 3 to 16) who were set to receive 2 consecutive courses of dexamethasone in accordance with Dutch Childhood Oncology Group ALL protocols.
The patients were randomized to receive either hydrocortisone or placebo in a circadian rhythm (10 mg/m2/d) during their first dexamethasone course. During their second course, the patients were assigned to the opposite arm.
The treatment groups were similar with regard to age, type of leukemia, treatment protocol, and CNS status at diagnosis.
Psychosocial problems
The researchers assessed psychosocial problems by having parents complete the Strength and Difficulties Questionnaire (SDQ). Forty-six parents completed the questionnaire at all 4 time points tested.
The results showed that 4 days of dexamethasone treatment significantly increased patient problems, as reported by all SDQ scales and subscales. However, one-third of the population did not have any increase in SDQ total difficulties with dexamethasone.
The addition of hydrocortisone did not affect patients’ total difficulties score (mean difference, -0.8 ± 5.5; P=0.33), emotional symptoms (mean difference, -0.6 ± 2.3; P=0.08), conduct problems (mean difference, 0.0 ± 1.5; P=1.00), or other SDQ subscales.
However, hydrocortisone did have a clinically significant effect in the subset of 16 patients who had clinically relevant dexamethasone-related AEs. This was defined as an increase of ≥5 in their SDQ total difficulties score.
In these patients, hydrocortisone improved the total difficulties delta-score (median difference, -5.0; IQR, -7.8 to -3.0), emotional symptoms score (median difference, -1.5; IQR, -4.0 to -1.0), conduct problems score (median difference, -1.0; IQR, -2.0 to 0.0), and impact of stress score (median difference, -1.0; IQR, -2.0 to 0.0).
Sleep issues
The researchers used the Sleep Disturbance Scale for Children (SDSC) to assess sleep quality and sleep disturbances. Forty-seven parents completed the questionnaire at all 4 time points tested.
Results showed that dexamethasone significantly increased disorders of arousal (P=0.04), sleep-wake transition disorders (P=0.01), and disorders of excessive somnolence (P=0.01).
The addition of hydrocortisone had no significant effect on patients’ total SDSC score (P=0.84), disorders of initiating and maintaining sleep (P=0.74), disorders of excessive somnolence (P=0.29), or sleep-wake transition disorder (P=0.29).
However, hydrocortisone did have a clinically significant effect in the subset of 9 children who had clinically relevant dexamethasone-induced sleeping problems, which were defined as a change of ≥7 in SDSC total score.
Hydrocortisone reduced SDSC total scores (median difference, -11.0; IQR, -16.0 to 0.0) and disorders of initiating and maintaining sleep scores (median difference, -3.0; IQR, -7.0 to –0.5).
Other outcomes
The researchers also found that dexamethasone treatment alone did not affect patients’ attention, visual-spatial functions, memory, or processing speed.
However, the addition of hydrocortisone significantly improved patients’ long-term visual memory (P=0.01).
Hydrocortisone did not have any effect on other neuropsychological tests or on metabolic parameters. ![]()
Study reveals potential treatment avenue for DBA, MDS

The production of two components of hemoglobin may be out of sync in Diamond Blackfan anemia (DBA) and myelodysplastic syndromes (MDS), according to a new study.
Researchers found that, in samples from patients with DBA or MDS, ribosome dysfunction delayed globin production, while heme synthesis proceeded normally.
This disruption in heme-globin coordination led to a buildup of toxic heme that killed red blood cell (RBC) precursors.
However, treating patient samples with a compound that blocks heme synthesis increased RBC production in both DBA and MDS.
Zhantao Yang, MD, of the University of Washington in Seattle, and his colleagues reported these findings in Science Translational Medicine.
Both DBA and MDS have been linked to defects in ribosome assembly, which is critical to protein production, but how this leads to anemia remains unknown.
To find out, Dr Yang and his colleagues analyzed bone marrow cells from patients with DBA (n=3) or MDS with del(5q) (n=6).
The researchers found that globin translation proceeded slowly in these samples, but heme synthesis proceeded normally.
This resulted in insufficient globin, excess heme, and excess reactive oxygen species in early erythroid precursors and, ultimately, the death of colony-forming unit–erythroid/proerythroblast cells.
The cells that were able to rapidly export heme or slow its synthesis survived and matured into RBCs, but the other colony-forming unit–erythroid cells/early proerythroblasts died.
The researchers noted that it is not clear how excess heme induces cell death in RBC precursors, but they said it likely involves both ferroptosis and apoptosis.
Regardless of the mechanism of cell death, the team found that treating the patients’ cells with succinylacetone (10 mM), a compound that blocks heme synthesis, improved RBC production.
The treatment improved RBC production in DBA and del(5q) MDS marrow cultures by 68% to 95% (P=0.03 to 0.05). In comparison, RBC production in control marrow cultures decreased by 4% to 13%.
The researchers said their experiments revealed additional important findings. First, they found that erythroid differentiation in the marrow cultures “excellently” phenocopied erythroid differentiation in vivo. This suggests these cultures can serve as a reliable platform in preclinical studies.
Second, the team said the fact that epigenetic differences between RBC precursors can lead to their preferential death or survival has broad implications. And querying the cells that preferentially survive could provide important insights. ![]()

The production of two components of hemoglobin may be out of sync in Diamond Blackfan anemia (DBA) and myelodysplastic syndromes (MDS), according to a new study.
Researchers found that, in samples from patients with DBA or MDS, ribosome dysfunction delayed globin production, while heme synthesis proceeded normally.
This disruption in heme-globin coordination led to a buildup of toxic heme that killed red blood cell (RBC) precursors.
However, treating patient samples with a compound that blocks heme synthesis increased RBC production in both DBA and MDS.
Zhantao Yang, MD, of the University of Washington in Seattle, and his colleagues reported these findings in Science Translational Medicine.
Both DBA and MDS have been linked to defects in ribosome assembly, which is critical to protein production, but how this leads to anemia remains unknown.
To find out, Dr Yang and his colleagues analyzed bone marrow cells from patients with DBA (n=3) or MDS with del(5q) (n=6).
The researchers found that globin translation proceeded slowly in these samples, but heme synthesis proceeded normally.
This resulted in insufficient globin, excess heme, and excess reactive oxygen species in early erythroid precursors and, ultimately, the death of colony-forming unit–erythroid/proerythroblast cells.
The cells that were able to rapidly export heme or slow its synthesis survived and matured into RBCs, but the other colony-forming unit–erythroid cells/early proerythroblasts died.
The researchers noted that it is not clear how excess heme induces cell death in RBC precursors, but they said it likely involves both ferroptosis and apoptosis.
Regardless of the mechanism of cell death, the team found that treating the patients’ cells with succinylacetone (10 mM), a compound that blocks heme synthesis, improved RBC production.
The treatment improved RBC production in DBA and del(5q) MDS marrow cultures by 68% to 95% (P=0.03 to 0.05). In comparison, RBC production in control marrow cultures decreased by 4% to 13%.
The researchers said their experiments revealed additional important findings. First, they found that erythroid differentiation in the marrow cultures “excellently” phenocopied erythroid differentiation in vivo. This suggests these cultures can serve as a reliable platform in preclinical studies.
Second, the team said the fact that epigenetic differences between RBC precursors can lead to their preferential death or survival has broad implications. And querying the cells that preferentially survive could provide important insights. ![]()

The production of two components of hemoglobin may be out of sync in Diamond Blackfan anemia (DBA) and myelodysplastic syndromes (MDS), according to a new study.
Researchers found that, in samples from patients with DBA or MDS, ribosome dysfunction delayed globin production, while heme synthesis proceeded normally.
This disruption in heme-globin coordination led to a buildup of toxic heme that killed red blood cell (RBC) precursors.
However, treating patient samples with a compound that blocks heme synthesis increased RBC production in both DBA and MDS.
Zhantao Yang, MD, of the University of Washington in Seattle, and his colleagues reported these findings in Science Translational Medicine.
Both DBA and MDS have been linked to defects in ribosome assembly, which is critical to protein production, but how this leads to anemia remains unknown.
To find out, Dr Yang and his colleagues analyzed bone marrow cells from patients with DBA (n=3) or MDS with del(5q) (n=6).
The researchers found that globin translation proceeded slowly in these samples, but heme synthesis proceeded normally.
This resulted in insufficient globin, excess heme, and excess reactive oxygen species in early erythroid precursors and, ultimately, the death of colony-forming unit–erythroid/proerythroblast cells.
The cells that were able to rapidly export heme or slow its synthesis survived and matured into RBCs, but the other colony-forming unit–erythroid cells/early proerythroblasts died.
The researchers noted that it is not clear how excess heme induces cell death in RBC precursors, but they said it likely involves both ferroptosis and apoptosis.
Regardless of the mechanism of cell death, the team found that treating the patients’ cells with succinylacetone (10 mM), a compound that blocks heme synthesis, improved RBC production.
The treatment improved RBC production in DBA and del(5q) MDS marrow cultures by 68% to 95% (P=0.03 to 0.05). In comparison, RBC production in control marrow cultures decreased by 4% to 13%.
The researchers said their experiments revealed additional important findings. First, they found that erythroid differentiation in the marrow cultures “excellently” phenocopied erythroid differentiation in vivo. This suggests these cultures can serve as a reliable platform in preclinical studies.
Second, the team said the fact that epigenetic differences between RBC precursors can lead to their preferential death or survival has broad implications. And querying the cells that preferentially survive could provide important insights. ![]()
CAR T-cell start-up launched
Dr. Siddhartha Mukherjee has partnered with Puretech Health to launch a new biotechnology and immuno-oncology company to broaden the use of chimeric antigen receptor (CAR) T-cell therapy. Dr. Mukherjee, a Columbia University researcher, hematologist, oncologist, and Pulitzer Prize–winning author of “The Emperor of All Maladies: A Biography of Cancer,” (New York: Scribner, a division of Simon & Schuster, 2011) is licensing his CAR T-cell technology to the joint venture, called Vor BioPharma.
Vor BioPharma will focus on advancing and expanding CAR T-cell therapy, a relatively new cancer treatment where T cells are first collected from a patient’s blood and then genetically engineered to produce CAR proteins on their surface. The CAR proteins are designed to bind specific antigens found on the patient’s cancer cells. These genetically engineered T cells are grown in a laboratory and then infused into the patient. As of now, CAR T-cell therapy is primarily used to treat B-cell leukemias and other chronic lymphocytic leukemia.
“We continue to make great strides in developing new ways to treat cancer using the body’s immune system,” said Dr. Mukherjee in a written statement announcing the partnership. “The positive clinical response researchers have achieved with CAR T-cell therapies in B-cell leukemias has led to great interest within the oncology community and is something we hope to achieve in other cancers over time,” he said.
“CAR T-cell therapies have shown remarkable progress in the clinic, yet their applicability beyond a small subset of cancers is currently very limited,” said Dr. Sanjiv Sam Gambhir of Stanford University and a member of the Vor Scientific Advisory Board. “This technology seeks to address bottlenecks that prevent CAR T-cell therapy from becoming more broadly useful in treating cancers outside of B-cell cancers.”
Other Vor BioPharma employees and Scientific Advisory Board members include Dr. Joseph Bolen, former President and Chief Scientific Officer of Moderna Therapeutics; Dr. Dan Littman of the Howard Hughes Medical Institute; and Dr. Derrick Rossi of Harvard University.
On Twitter @jess_craig94
Dr. Siddhartha Mukherjee has partnered with Puretech Health to launch a new biotechnology and immuno-oncology company to broaden the use of chimeric antigen receptor (CAR) T-cell therapy. Dr. Mukherjee, a Columbia University researcher, hematologist, oncologist, and Pulitzer Prize–winning author of “The Emperor of All Maladies: A Biography of Cancer,” (New York: Scribner, a division of Simon & Schuster, 2011) is licensing his CAR T-cell technology to the joint venture, called Vor BioPharma.
Vor BioPharma will focus on advancing and expanding CAR T-cell therapy, a relatively new cancer treatment where T cells are first collected from a patient’s blood and then genetically engineered to produce CAR proteins on their surface. The CAR proteins are designed to bind specific antigens found on the patient’s cancer cells. These genetically engineered T cells are grown in a laboratory and then infused into the patient. As of now, CAR T-cell therapy is primarily used to treat B-cell leukemias and other chronic lymphocytic leukemia.
“We continue to make great strides in developing new ways to treat cancer using the body’s immune system,” said Dr. Mukherjee in a written statement announcing the partnership. “The positive clinical response researchers have achieved with CAR T-cell therapies in B-cell leukemias has led to great interest within the oncology community and is something we hope to achieve in other cancers over time,” he said.
“CAR T-cell therapies have shown remarkable progress in the clinic, yet their applicability beyond a small subset of cancers is currently very limited,” said Dr. Sanjiv Sam Gambhir of Stanford University and a member of the Vor Scientific Advisory Board. “This technology seeks to address bottlenecks that prevent CAR T-cell therapy from becoming more broadly useful in treating cancers outside of B-cell cancers.”
Other Vor BioPharma employees and Scientific Advisory Board members include Dr. Joseph Bolen, former President and Chief Scientific Officer of Moderna Therapeutics; Dr. Dan Littman of the Howard Hughes Medical Institute; and Dr. Derrick Rossi of Harvard University.
On Twitter @jess_craig94
Dr. Siddhartha Mukherjee has partnered with Puretech Health to launch a new biotechnology and immuno-oncology company to broaden the use of chimeric antigen receptor (CAR) T-cell therapy. Dr. Mukherjee, a Columbia University researcher, hematologist, oncologist, and Pulitzer Prize–winning author of “The Emperor of All Maladies: A Biography of Cancer,” (New York: Scribner, a division of Simon & Schuster, 2011) is licensing his CAR T-cell technology to the joint venture, called Vor BioPharma.
Vor BioPharma will focus on advancing and expanding CAR T-cell therapy, a relatively new cancer treatment where T cells are first collected from a patient’s blood and then genetically engineered to produce CAR proteins on their surface. The CAR proteins are designed to bind specific antigens found on the patient’s cancer cells. These genetically engineered T cells are grown in a laboratory and then infused into the patient. As of now, CAR T-cell therapy is primarily used to treat B-cell leukemias and other chronic lymphocytic leukemia.
“We continue to make great strides in developing new ways to treat cancer using the body’s immune system,” said Dr. Mukherjee in a written statement announcing the partnership. “The positive clinical response researchers have achieved with CAR T-cell therapies in B-cell leukemias has led to great interest within the oncology community and is something we hope to achieve in other cancers over time,” he said.
“CAR T-cell therapies have shown remarkable progress in the clinic, yet their applicability beyond a small subset of cancers is currently very limited,” said Dr. Sanjiv Sam Gambhir of Stanford University and a member of the Vor Scientific Advisory Board. “This technology seeks to address bottlenecks that prevent CAR T-cell therapy from becoming more broadly useful in treating cancers outside of B-cell cancers.”
Other Vor BioPharma employees and Scientific Advisory Board members include Dr. Joseph Bolen, former President and Chief Scientific Officer of Moderna Therapeutics; Dr. Dan Littman of the Howard Hughes Medical Institute; and Dr. Derrick Rossi of Harvard University.
On Twitter @jess_craig94
Company warns of counterfeit drug

Photo by Bill Branson
Heritage Pharmaceuticals Inc., has announced the existence of a counterfeit drug product labeled as BiCNU® (carmustine for injection) 100 mg.
The company said that, to the best of its knowledge, the counterfeit product has only been distributed in India, Ireland, and Israel.
However, Heritage is consulting with the US Food and Drug Administration (FDA) to aid the agency’s evaluations of this product, assist with determining the source of the counterfeit drug, and prevent the further distribution of this product or its introduction into the US.
BiCNU® is primarily used for chemotherapy in the treatment of lymphomas, multiple myeloma, and brain cancers. But the drug is also used for immunosuppression before organ transplant or hematopoietic stem cell transplant.
Heritage said it has directly notified all customers and provided detailed information that will help them identify a counterfeit BiCNU® product. Customers have been instructed to examine their inventory immediately and to quarantine, discontinue distribution of, and return any suspected counterfeit product.
Any customers who may have recently distributed the BiCNU® products to their own customers have been asked to convey this information to their customers so they will be able to carefully examine all BiCNU® products before use and identify the characteristics of a suspected counterfeit product.
Any end users who believe they may have received a counterfeit drug should return the product to the pharmacy that dispensed the medicine.
Any US health practitioners who determine they are in possession of a counterfeit product should contact the FDA through MedWatch. Instructions for such reporting are available on the FDA website.
Anyone with questions about the counterfeit product should contact the Heritage customer call center directly at (866) 901-3784, which is open Monday through Friday, from 9 am to 5 pm EST. ![]()

Photo by Bill Branson
Heritage Pharmaceuticals Inc., has announced the existence of a counterfeit drug product labeled as BiCNU® (carmustine for injection) 100 mg.
The company said that, to the best of its knowledge, the counterfeit product has only been distributed in India, Ireland, and Israel.
However, Heritage is consulting with the US Food and Drug Administration (FDA) to aid the agency’s evaluations of this product, assist with determining the source of the counterfeit drug, and prevent the further distribution of this product or its introduction into the US.
BiCNU® is primarily used for chemotherapy in the treatment of lymphomas, multiple myeloma, and brain cancers. But the drug is also used for immunosuppression before organ transplant or hematopoietic stem cell transplant.
Heritage said it has directly notified all customers and provided detailed information that will help them identify a counterfeit BiCNU® product. Customers have been instructed to examine their inventory immediately and to quarantine, discontinue distribution of, and return any suspected counterfeit product.
Any customers who may have recently distributed the BiCNU® products to their own customers have been asked to convey this information to their customers so they will be able to carefully examine all BiCNU® products before use and identify the characteristics of a suspected counterfeit product.
Any end users who believe they may have received a counterfeit drug should return the product to the pharmacy that dispensed the medicine.
Any US health practitioners who determine they are in possession of a counterfeit product should contact the FDA through MedWatch. Instructions for such reporting are available on the FDA website.
Anyone with questions about the counterfeit product should contact the Heritage customer call center directly at (866) 901-3784, which is open Monday through Friday, from 9 am to 5 pm EST. ![]()

Photo by Bill Branson
Heritage Pharmaceuticals Inc., has announced the existence of a counterfeit drug product labeled as BiCNU® (carmustine for injection) 100 mg.
The company said that, to the best of its knowledge, the counterfeit product has only been distributed in India, Ireland, and Israel.
However, Heritage is consulting with the US Food and Drug Administration (FDA) to aid the agency’s evaluations of this product, assist with determining the source of the counterfeit drug, and prevent the further distribution of this product or its introduction into the US.
BiCNU® is primarily used for chemotherapy in the treatment of lymphomas, multiple myeloma, and brain cancers. But the drug is also used for immunosuppression before organ transplant or hematopoietic stem cell transplant.
Heritage said it has directly notified all customers and provided detailed information that will help them identify a counterfeit BiCNU® product. Customers have been instructed to examine their inventory immediately and to quarantine, discontinue distribution of, and return any suspected counterfeit product.
Any customers who may have recently distributed the BiCNU® products to their own customers have been asked to convey this information to their customers so they will be able to carefully examine all BiCNU® products before use and identify the characteristics of a suspected counterfeit product.
Any end users who believe they may have received a counterfeit drug should return the product to the pharmacy that dispensed the medicine.
Any US health practitioners who determine they are in possession of a counterfeit product should contact the FDA through MedWatch. Instructions for such reporting are available on the FDA website.
Anyone with questions about the counterfeit product should contact the Heritage customer call center directly at (866) 901-3784, which is open Monday through Friday, from 9 am to 5 pm EST.
Tools may aid transition from pediatric to adult care

Photo courtesy of the CDC
WASHINGTON, DC—The American Society of Hematology (ASH) has created a toolkit to help hematologists aid patients who are transitioning from pediatric to adult practices.
The toolkit contains general resources for all hematologic conditions, as well as specific resources for patients with hemophilia and sickle cell disease.
It includes 2 types of forms—a transition-readiness assessment and a clinical summary.
The toolkit was presented at the American College of Physicians (ACP) Internal Medicine Meeting 2016.
“Transitioning from pediatric to adult healthcare practices is often a challenge for patients with chronic medical issues because it can be difficult to adhere to a treatment regimen or attend regular appointments without the assistance of a parent or guardian,” said ASH President Charles S. Abrams, MD, of the University of Pennsylvania in Philadelphia.
“ASH recognizes that understanding a patient’s preparedness to take control of his or her medical condition in adulthood can make a huge difference in quality of care, which is why we are pleased to join the American College of Physicians and partner societies in this important initiative.”
ASH joined more than 2 dozen groups to participate in the ACP’s Pediatric to Adult Care Transition Initiative. The goal of this initiative was to develop guidance and tools that both primary care internal medicine and subspecialty practices can use for patients who are transitioning from pediatric/adolescent practices to adult care.
An ASH Transitions Work Group, made up of society members from pediatric and adult practices, developed 3 segments of the hematology-specific toolkit:
- generic forms for patients with any hematologic condition, with an addendum that includes links to additional condition-specific guidelines and resources
- specific forms for hemophilia
- specific forms for sickle cell disease.
For each segment, there are 2 types of forms— a transition-readiness assessment and a clinical summary.
The transition-readiness assessment should be completed by the patient. It assesses the patient’s readiness for the transition to adult care by evaluating the patient’s understanding of his or her condition and ability to manage medications, appointments, insurance, and medical privacy issues.
This assessment should be used by the adult care team to assess any remaining gaps in the patient’s self-care knowledge or additional issues that should be addressed to ensure optimal care.
The clinical summary is a medical record summary to be completed by the referring provider and the patient. The summary contains essential clinical information regarding the patient’s condition that is to be included in the patient’s medical record upon transfer to the adult practice.
More information on the ACP Pediatric to Adult Care Transitions Initiative is available on the ACP website. The forms for the ASH transitions toolkit are available in the “Hematology” section of the Condition-Specific Tools page.

Photo courtesy of the CDC
WASHINGTON, DC—The American Society of Hematology (ASH) has created a toolkit to help hematologists aid patients who are transitioning from pediatric to adult practices.
The toolkit contains general resources for all hematologic conditions, as well as specific resources for patients with hemophilia and sickle cell disease.
It includes 2 types of forms—a transition-readiness assessment and a clinical summary.
The toolkit was presented at the American College of Physicians (ACP) Internal Medicine Meeting 2016.
“Transitioning from pediatric to adult healthcare practices is often a challenge for patients with chronic medical issues because it can be difficult to adhere to a treatment regimen or attend regular appointments without the assistance of a parent or guardian,” said ASH President Charles S. Abrams, MD, of the University of Pennsylvania in Philadelphia.
“ASH recognizes that understanding a patient’s preparedness to take control of his or her medical condition in adulthood can make a huge difference in quality of care, which is why we are pleased to join the American College of Physicians and partner societies in this important initiative.”
ASH joined more than 2 dozen groups to participate in the ACP’s Pediatric to Adult Care Transition Initiative. The goal of this initiative was to develop guidance and tools that both primary care internal medicine and subspecialty practices can use for patients who are transitioning from pediatric/adolescent practices to adult care.
An ASH Transitions Work Group, made up of society members from pediatric and adult practices, developed 3 segments of the hematology-specific toolkit:
- generic forms for patients with any hematologic condition, with an addendum that includes links to additional condition-specific guidelines and resources
- specific forms for hemophilia
- specific forms for sickle cell disease.
For each segment, there are 2 types of forms— a transition-readiness assessment and a clinical summary.
The transition-readiness assessment should be completed by the patient. It assesses the patient’s readiness for the transition to adult care by evaluating the patient’s understanding of his or her condition and ability to manage medications, appointments, insurance, and medical privacy issues.
This assessment should be used by the adult care team to assess any remaining gaps in the patient’s self-care knowledge or additional issues that should be addressed to ensure optimal care.
The clinical summary is a medical record summary to be completed by the referring provider and the patient. The summary contains essential clinical information regarding the patient’s condition that is to be included in the patient’s medical record upon transfer to the adult practice.
More information on the ACP Pediatric to Adult Care Transitions Initiative is available on the ACP website. The forms for the ASH transitions toolkit are available in the “Hematology” section of the Condition-Specific Tools page.

Photo courtesy of the CDC
WASHINGTON, DC—The American Society of Hematology (ASH) has created a toolkit to help hematologists aid patients who are transitioning from pediatric to adult practices.
The toolkit contains general resources for all hematologic conditions, as well as specific resources for patients with hemophilia and sickle cell disease.
It includes 2 types of forms—a transition-readiness assessment and a clinical summary.
The toolkit was presented at the American College of Physicians (ACP) Internal Medicine Meeting 2016.
“Transitioning from pediatric to adult healthcare practices is often a challenge for patients with chronic medical issues because it can be difficult to adhere to a treatment regimen or attend regular appointments without the assistance of a parent or guardian,” said ASH President Charles S. Abrams, MD, of the University of Pennsylvania in Philadelphia.
“ASH recognizes that understanding a patient’s preparedness to take control of his or her medical condition in adulthood can make a huge difference in quality of care, which is why we are pleased to join the American College of Physicians and partner societies in this important initiative.”
ASH joined more than 2 dozen groups to participate in the ACP’s Pediatric to Adult Care Transition Initiative. The goal of this initiative was to develop guidance and tools that both primary care internal medicine and subspecialty practices can use for patients who are transitioning from pediatric/adolescent practices to adult care.
An ASH Transitions Work Group, made up of society members from pediatric and adult practices, developed 3 segments of the hematology-specific toolkit:
- generic forms for patients with any hematologic condition, with an addendum that includes links to additional condition-specific guidelines and resources
- specific forms for hemophilia
- specific forms for sickle cell disease.
For each segment, there are 2 types of forms— a transition-readiness assessment and a clinical summary.
The transition-readiness assessment should be completed by the patient. It assesses the patient’s readiness for the transition to adult care by evaluating the patient’s understanding of his or her condition and ability to manage medications, appointments, insurance, and medical privacy issues.
This assessment should be used by the adult care team to assess any remaining gaps in the patient’s self-care knowledge or additional issues that should be addressed to ensure optimal care.
The clinical summary is a medical record summary to be completed by the referring provider and the patient. The summary contains essential clinical information regarding the patient’s condition that is to be included in the patient’s medical record upon transfer to the adult practice.
More information on the ACP Pediatric to Adult Care Transitions Initiative is available on the ACP website. The forms for the ASH transitions toolkit are available in the “Hematology” section of the Condition-Specific Tools page.
FDA grants priority review for blinatumomab

and solution for infusion
Photo courtesy of Amgen
The US Food and Drug Administration (FDA) has accepted for priority review the supplemental biologics license application for blinatumomab (Blincyto) as a treatment for pediatric and adolescent patients with Philadelphia chromosome negative (Ph-) relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL).
To grant an application priority review, the FDA must believe the drug would provide a significant improvement in the treatment, diagnosis, or prevention of a serious condition.
The priority review designation means the FDA’s goal is to take action on an application within 6 months, rather than the 10 months typically taken for a standard review.
The Prescription Drug User Fee Act target action date for the supplemental biologics license application for blinatumomab is September 1, 2016.
About blinatumomab
Blinatumomab is a bispecific, CD19-directed, CD3 T-cell engager (BiTE®) antibody construct that binds specifically to CD19 expressed on the surface of cells of B-lineage origin and CD3 expressed on the surface of T cells.
Blinatumomab was previously granted breakthrough therapy and priority review designations by the FDA and is now approved in the US for the treatment of adults with Ph- relapsed or refractory B-cell precursor ALL.
This indication is approved under accelerated approval. Continued approval for this indication may be contingent upon verification of clinical benefit in subsequent trials.
Blinatumomab is marketed by Amgen as Blincyto. The full US prescribing information is available at www.BLINCYTO.com.
‘205 trial
The supplemental biologics license application for blinatumomab in pediatric and adolescent patients is based on data from the phase 1/2 '205 trial.
In this study, researchers evaluated blinatumomab in patients younger than 18 years of age. The patients had B-cell precursor ALL that was refractory, had relapsed at least twice, or relapsed after an allogeneic hematopoietic stem cell transplant.
Treatment in this study has been completed, and subjects are being monitored for long-term efficacy. The data is being submitted for publication.
Preliminary data were presented at the 2014 ASH Annual Meeting (abstract 3703).

and solution for infusion
Photo courtesy of Amgen
The US Food and Drug Administration (FDA) has accepted for priority review the supplemental biologics license application for blinatumomab (Blincyto) as a treatment for pediatric and adolescent patients with Philadelphia chromosome negative (Ph-) relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL).
To grant an application priority review, the FDA must believe the drug would provide a significant improvement in the treatment, diagnosis, or prevention of a serious condition.
The priority review designation means the FDA’s goal is to take action on an application within 6 months, rather than the 10 months typically taken for a standard review.
The Prescription Drug User Fee Act target action date for the supplemental biologics license application for blinatumomab is September 1, 2016.
About blinatumomab
Blinatumomab is a bispecific, CD19-directed, CD3 T-cell engager (BiTE®) antibody construct that binds specifically to CD19 expressed on the surface of cells of B-lineage origin and CD3 expressed on the surface of T cells.
Blinatumomab was previously granted breakthrough therapy and priority review designations by the FDA and is now approved in the US for the treatment of adults with Ph- relapsed or refractory B-cell precursor ALL.
This indication is approved under accelerated approval. Continued approval for this indication may be contingent upon verification of clinical benefit in subsequent trials.
Blinatumomab is marketed by Amgen as Blincyto. The full US prescribing information is available at www.BLINCYTO.com.
‘205 trial
The supplemental biologics license application for blinatumomab in pediatric and adolescent patients is based on data from the phase 1/2 '205 trial.
In this study, researchers evaluated blinatumomab in patients younger than 18 years of age. The patients had B-cell precursor ALL that was refractory, had relapsed at least twice, or relapsed after an allogeneic hematopoietic stem cell transplant.
Treatment in this study has been completed, and subjects are being monitored for long-term efficacy. The data is being submitted for publication.
Preliminary data were presented at the 2014 ASH Annual Meeting (abstract 3703).

and solution for infusion
Photo courtesy of Amgen
The US Food and Drug Administration (FDA) has accepted for priority review the supplemental biologics license application for blinatumomab (Blincyto) as a treatment for pediatric and adolescent patients with Philadelphia chromosome negative (Ph-) relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL).
To grant an application priority review, the FDA must believe the drug would provide a significant improvement in the treatment, diagnosis, or prevention of a serious condition.
The priority review designation means the FDA’s goal is to take action on an application within 6 months, rather than the 10 months typically taken for a standard review.
The Prescription Drug User Fee Act target action date for the supplemental biologics license application for blinatumomab is September 1, 2016.
About blinatumomab
Blinatumomab is a bispecific, CD19-directed, CD3 T-cell engager (BiTE®) antibody construct that binds specifically to CD19 expressed on the surface of cells of B-lineage origin and CD3 expressed on the surface of T cells.
Blinatumomab was previously granted breakthrough therapy and priority review designations by the FDA and is now approved in the US for the treatment of adults with Ph- relapsed or refractory B-cell precursor ALL.
This indication is approved under accelerated approval. Continued approval for this indication may be contingent upon verification of clinical benefit in subsequent trials.
Blinatumomab is marketed by Amgen as Blincyto. The full US prescribing information is available at www.BLINCYTO.com.
‘205 trial
The supplemental biologics license application for blinatumomab in pediatric and adolescent patients is based on data from the phase 1/2 '205 trial.
In this study, researchers evaluated blinatumomab in patients younger than 18 years of age. The patients had B-cell precursor ALL that was refractory, had relapsed at least twice, or relapsed after an allogeneic hematopoietic stem cell transplant.
Treatment in this study has been completed, and subjects are being monitored for long-term efficacy. The data is being submitted for publication.
Preliminary data were presented at the 2014 ASH Annual Meeting (abstract 3703).