User login
AHA Late-Breaking Clinical Trials preview
The emphasis on this year’s American Heart Association Scientific Sessions in New Orleans is bigness: “Big science, big technology, and big networking opportunities,” the AHA 16 website says.
And so the 19 abstracts out of thousands submitted that got the biggest score from program committee for AHA 2016, led by Frank Sellke, MD, were chosen for presentation at four Late-Breaking Clinical Trials session previewed the late-breaking science.
Big trials for big questions
The first late-breaker session, on Sunday, Nov. 13, at 3:45 p.m., CT, is titled will, as its title says present the long-awaited results of four trials with large enrollment and long-term outcomes.
EUCLID (A Study Comparing Cardiovascular Effects of Ticagrelor and Clopidogrel in Patients With Peripheral Artery Disease) randomized an estimated 16,000 patients with symptomatic PAD to long-term antiplatelet monotherapy with either ticagrelor or clopidogrel to see which one would be superior in preventing the composite of cardiovascular death, myocardial infarction and ischemic stroke up to 40 months. Secondarily, it looked at acute limb ischemia, need for revascularization, and disease progression. “This could have tremendous implications for patients treat for pad trying to prevent CV disease,” Dr. Sellke said.
PRECISION (Prospective Randomized Evaluation of Celecoxib Integrated Safety vs Ibuprofen or Naproxen) harks back to 2005, when the Food and Drug Administration, wrestling with the growing evidence that NSAIDs were linked with cardiovascular events, asked for a large, cardiovascular outcomes trial. PRECISION, sponsored by Pfizer but run by an academic-led steering committee led by Steven Nissen, MD, now chief of cardiovascular medicine at the Cleveland Clinic, randomized some 20,000 arthritis patients with or at risk for cardiovascular disease to long-term pain treatment with celecoxib, naproxen, or ibuprofen for a planned follow-up of 2 years. The primary endpoint is a composite of cardiovascular death, nonfatal MI, and nonfatal stroke. Dr. Sellke noted that the results will be important for many physicians and patients wanting to minimize the risks associated with NSAIDs.
HOPE 3 (Heart Outcomes Evaluation 3), presented in April this year at the American College of Cardiology meeting in Chicago, showed the combination of rosuvastatin plus candesartan and hydrochlorothiazide reduced cardiovascular events in intermediate-risk patients with hypertension, regardless of their baseline LDL cholesterol and inflammatory biomarker levels. The analysis to be presented at AHA will show whether the combination has any effect on cognitive function. As evidence builds of the cardiovascular benefit of aggressive treatment of hypertension, as in the SPRINT trial, the results could be tremendously important, Dr. Sellke said.
TRUE AHF (Efficacy and Safety of Ularitide for the Treatment of Acute Decompensated Heart Failure) randomized about 2,150 patients with acute decompensated heart failure to receive a 48-hour intravenous infusion of the natriuretic peptide ularitide or placebo. The primary outcome is a composite of 48-hour improved in-hospital worsening or unchanged clinical conditions, as well as long-term cardiovascular mortality with a median follow-up of 7 months. Because there are no effective treatments for acute systolic heart failure, the results of TRUE AHF could be of tremendous benefit, Dr. Sellke said.
Pioneering the Future of HeART Interventions
The trials with the greatest impact for practice to be presented at AHA 2015, according to the Dr. Sellke’s admitted bias as a cardiothoracic surgeon, will all be presented in this second of the late-breaker sessions, on Monday, Nov. 14, at 10:45 a.m., CT.
ART (Arterial Revascularization Trial) was a comparison of single vs. bilateral internal mammary artery grafting in more than 3,000 randomized patients undergoing coronary artery bypass surgery (CABG). The outcomes of mortality, stroke, MI, and repeat revascularization were published in 2010, showing no differences between groups. The 5-year results to be presented on Monday may resolve some of the controversy surrounding the two methods, as surgeons and cardiologists are strongly divided on the benefits and risks of single, compared with double, internal mammary artery grafting.
FUTURE (Functional Testing Underlying Coronary Revascularization) compared fractional flow reserve–guided management with conventional management in roughly 900 patients undergoing revascularization with multivessel coronary artery disease. The primary outcome is a composite of death, MI, coronary revascularization, and stroke. FFR has received a lot of attention recently, Dr. Sellke said, because it looks at the physiologic, rather than the anatomic, effects of lesion on catheterization. The results will show whether there’s clinical benefit to adding FFR to angiography that will offset the additional time it takes to perform before PCI or CABG.
PIONEER AF-PCI (An Open-label, Randomized, Controlled, Multicenter Study Exploring Two Treatment Strategies of Rivaroxaban and a Dose-Adjusted Oral Vitamin K Antagonist Treatment Strategy in Subjects With Atrial Fibrillation Who Undergo Percutaneous Coronary Intervention) addressed the conundrum of treating anticoagulated patients with atrial fibrillation who are undergoing PCI with adequate dual-antiplatelet therapy – and avoiding bleeding events. About 2,000 patients were randomized to varying combinations of rivaroxaban or warfarin plus aspirin, ticagrelor prasugrel, and/or clopidogrel for 1 year. The primary outcome is significant bleeding. Dr. Sellke said that because drug-eluting stents require at least a year of DAPT, the PIONEER AF-PCI results will add knowledge in an important and controversial area.
GERMANY is a report from the German Aortic Valve Registry (GARY) on the 1-year outcomes of patients with intermediate-risk severe aortic stenosis who underwent either transcatheter or surgical aortic replacement on the efficacy and outcomes of the two approaches. Dr. Sellke noted that these results will be important because the patients in this registry were not at high risk or ineligible for surgical aortic replacement.
Insights from New Therapeutic Trials for Lipids
Of the five trials presented in this session on Tuesday, Nov. 15, at 10:45 a.m., CT, only one is in an approved treatment for lowering lipids. That is GLAGOV (Global Assessment of Plaque Regression With a PCSK9 Antibody as Measured by Intravascular Ultrasound), is looking at whether LDL lowering with the PCSK9 inhibitor evolocumab reduces atheroma volume in almost 1,000 patients.
Guiding the Momentum to Effect HF Outcomes – Ironing Out the Wrinkles
Two of the six heart failure trials presented in this session on Wednesday, Nov. 16, at 10:45 a.m., CT, study cardiorespiratory effects of iron, thus the title, Dr. Sellke said.
REDUCE LAP HF (A Study to Evaluate the DC Devices, Inc. IASD System II to REDUCE Elevated Left Atrial Pressure in Patients With Heart Failure). The primary outcome is a composite of death, stroke, MI, or a systemic embolic event at 6 months. The trial evaluated a transcatheter interatrial shunt device to left atrial pressure in patients with heart failure with preserved ejection fraction (HFpEF). In this type of diastolic heart failure in which patients’ hearts cannot relax, there is really no treatment, Dr. Sellke said. So although this treatment seems “hokey,” a positive result could be important.
ATHENA HF (Aldosterone Targeted Neurohormonal Combined with Natriuresis Therapy in Heart Failure) tested the diuretic spironolactone in heart failure. The investigators randomized 360 patients to high-dose spironolactone or usual care to see whether they could provide greater reductions of n-terminal prohormone of brain natriuretic peptide (NT-proBNP) levels within 96 hours. There’s evidence that spironolactone can provide symptomatic relief for patients with heart failure, so these results could be important, Dr. Sellke said.
IRONOUT HF (Oral Iron Repletion Effects on Oxygen Up Take in Heart Failure) randomized heart failure patients with iron deficiency to oral iron supplementation or placebo and measured peak oxygen uptake at 16 weeks.
EFFECT-HF (Effect of Ferric Carboxymaltose on Exercise Capacity in Patients with Iron Deficiency and Chronic Heart Failure) also studied the effect of iron supplementation, intravenous in this case, on exercise capacity in heart failure patients at 24 weeks. Iron depletion is a hallmark of heart failure, Dr. Sellke pointed out, so iron repletion could be a simple way to improve functional capacity.
MOMENTUM 3 (Multicenter Study of MagLev Technology in Patients Undergoing Mechanical Circulatory Support Therapy with HM3) evaluated the safety and effectiveness of the Thoratec HeartMate 3 left-ventricular assist device employing MagLev technology, which is said to facilitate the free flow of blood through the device. Roughly 1,000 patients with advanced, refractory heart failure were randomized to receive either the investigational HeartMate 3 or the HeartMate 2. The primary outcomes included short- and long-term survival and freedom from debilitating stroke. Trials such as this are very important, Dr. Sellke said, because the need for donor hearts far exceeds demand and better, cheaper LVADs that last longer could extend the lives of many thousands of patients every year.
MultiSENSE (Evaluation of Multisensor Data in Heart Failure Patients With Implanted Devices) collected information taken from sensors in an implanted cardiac synchronization therapy device in 1,000 patients to develop algorithms that would detect worsening heart failure. Multiple readmissions for heart failure are frequent and ineffective, and detecting the onset of worsening heart failure has the potential to bring those admissions way down, Dr. Sellke said.
The emphasis on this year’s American Heart Association Scientific Sessions in New Orleans is bigness: “Big science, big technology, and big networking opportunities,” the AHA 16 website says.
And so the 19 abstracts out of thousands submitted that got the biggest score from program committee for AHA 2016, led by Frank Sellke, MD, were chosen for presentation at four Late-Breaking Clinical Trials session previewed the late-breaking science.
Big trials for big questions
The first late-breaker session, on Sunday, Nov. 13, at 3:45 p.m., CT, is titled will, as its title says present the long-awaited results of four trials with large enrollment and long-term outcomes.
EUCLID (A Study Comparing Cardiovascular Effects of Ticagrelor and Clopidogrel in Patients With Peripheral Artery Disease) randomized an estimated 16,000 patients with symptomatic PAD to long-term antiplatelet monotherapy with either ticagrelor or clopidogrel to see which one would be superior in preventing the composite of cardiovascular death, myocardial infarction and ischemic stroke up to 40 months. Secondarily, it looked at acute limb ischemia, need for revascularization, and disease progression. “This could have tremendous implications for patients treat for pad trying to prevent CV disease,” Dr. Sellke said.
PRECISION (Prospective Randomized Evaluation of Celecoxib Integrated Safety vs Ibuprofen or Naproxen) harks back to 2005, when the Food and Drug Administration, wrestling with the growing evidence that NSAIDs were linked with cardiovascular events, asked for a large, cardiovascular outcomes trial. PRECISION, sponsored by Pfizer but run by an academic-led steering committee led by Steven Nissen, MD, now chief of cardiovascular medicine at the Cleveland Clinic, randomized some 20,000 arthritis patients with or at risk for cardiovascular disease to long-term pain treatment with celecoxib, naproxen, or ibuprofen for a planned follow-up of 2 years. The primary endpoint is a composite of cardiovascular death, nonfatal MI, and nonfatal stroke. Dr. Sellke noted that the results will be important for many physicians and patients wanting to minimize the risks associated with NSAIDs.
HOPE 3 (Heart Outcomes Evaluation 3), presented in April this year at the American College of Cardiology meeting in Chicago, showed the combination of rosuvastatin plus candesartan and hydrochlorothiazide reduced cardiovascular events in intermediate-risk patients with hypertension, regardless of their baseline LDL cholesterol and inflammatory biomarker levels. The analysis to be presented at AHA will show whether the combination has any effect on cognitive function. As evidence builds of the cardiovascular benefit of aggressive treatment of hypertension, as in the SPRINT trial, the results could be tremendously important, Dr. Sellke said.
TRUE AHF (Efficacy and Safety of Ularitide for the Treatment of Acute Decompensated Heart Failure) randomized about 2,150 patients with acute decompensated heart failure to receive a 48-hour intravenous infusion of the natriuretic peptide ularitide or placebo. The primary outcome is a composite of 48-hour improved in-hospital worsening or unchanged clinical conditions, as well as long-term cardiovascular mortality with a median follow-up of 7 months. Because there are no effective treatments for acute systolic heart failure, the results of TRUE AHF could be of tremendous benefit, Dr. Sellke said.
Pioneering the Future of HeART Interventions
The trials with the greatest impact for practice to be presented at AHA 2015, according to the Dr. Sellke’s admitted bias as a cardiothoracic surgeon, will all be presented in this second of the late-breaker sessions, on Monday, Nov. 14, at 10:45 a.m., CT.
ART (Arterial Revascularization Trial) was a comparison of single vs. bilateral internal mammary artery grafting in more than 3,000 randomized patients undergoing coronary artery bypass surgery (CABG). The outcomes of mortality, stroke, MI, and repeat revascularization were published in 2010, showing no differences between groups. The 5-year results to be presented on Monday may resolve some of the controversy surrounding the two methods, as surgeons and cardiologists are strongly divided on the benefits and risks of single, compared with double, internal mammary artery grafting.
FUTURE (Functional Testing Underlying Coronary Revascularization) compared fractional flow reserve–guided management with conventional management in roughly 900 patients undergoing revascularization with multivessel coronary artery disease. The primary outcome is a composite of death, MI, coronary revascularization, and stroke. FFR has received a lot of attention recently, Dr. Sellke said, because it looks at the physiologic, rather than the anatomic, effects of lesion on catheterization. The results will show whether there’s clinical benefit to adding FFR to angiography that will offset the additional time it takes to perform before PCI or CABG.
PIONEER AF-PCI (An Open-label, Randomized, Controlled, Multicenter Study Exploring Two Treatment Strategies of Rivaroxaban and a Dose-Adjusted Oral Vitamin K Antagonist Treatment Strategy in Subjects With Atrial Fibrillation Who Undergo Percutaneous Coronary Intervention) addressed the conundrum of treating anticoagulated patients with atrial fibrillation who are undergoing PCI with adequate dual-antiplatelet therapy – and avoiding bleeding events. About 2,000 patients were randomized to varying combinations of rivaroxaban or warfarin plus aspirin, ticagrelor prasugrel, and/or clopidogrel for 1 year. The primary outcome is significant bleeding. Dr. Sellke said that because drug-eluting stents require at least a year of DAPT, the PIONEER AF-PCI results will add knowledge in an important and controversial area.
GERMANY is a report from the German Aortic Valve Registry (GARY) on the 1-year outcomes of patients with intermediate-risk severe aortic stenosis who underwent either transcatheter or surgical aortic replacement on the efficacy and outcomes of the two approaches. Dr. Sellke noted that these results will be important because the patients in this registry were not at high risk or ineligible for surgical aortic replacement.
Insights from New Therapeutic Trials for Lipids
Of the five trials presented in this session on Tuesday, Nov. 15, at 10:45 a.m., CT, only one is in an approved treatment for lowering lipids. That is GLAGOV (Global Assessment of Plaque Regression With a PCSK9 Antibody as Measured by Intravascular Ultrasound), is looking at whether LDL lowering with the PCSK9 inhibitor evolocumab reduces atheroma volume in almost 1,000 patients.
Guiding the Momentum to Effect HF Outcomes – Ironing Out the Wrinkles
Two of the six heart failure trials presented in this session on Wednesday, Nov. 16, at 10:45 a.m., CT, study cardiorespiratory effects of iron, thus the title, Dr. Sellke said.
REDUCE LAP HF (A Study to Evaluate the DC Devices, Inc. IASD System II to REDUCE Elevated Left Atrial Pressure in Patients With Heart Failure). The primary outcome is a composite of death, stroke, MI, or a systemic embolic event at 6 months. The trial evaluated a transcatheter interatrial shunt device to left atrial pressure in patients with heart failure with preserved ejection fraction (HFpEF). In this type of diastolic heart failure in which patients’ hearts cannot relax, there is really no treatment, Dr. Sellke said. So although this treatment seems “hokey,” a positive result could be important.
ATHENA HF (Aldosterone Targeted Neurohormonal Combined with Natriuresis Therapy in Heart Failure) tested the diuretic spironolactone in heart failure. The investigators randomized 360 patients to high-dose spironolactone or usual care to see whether they could provide greater reductions of n-terminal prohormone of brain natriuretic peptide (NT-proBNP) levels within 96 hours. There’s evidence that spironolactone can provide symptomatic relief for patients with heart failure, so these results could be important, Dr. Sellke said.
IRONOUT HF (Oral Iron Repletion Effects on Oxygen Up Take in Heart Failure) randomized heart failure patients with iron deficiency to oral iron supplementation or placebo and measured peak oxygen uptake at 16 weeks.
EFFECT-HF (Effect of Ferric Carboxymaltose on Exercise Capacity in Patients with Iron Deficiency and Chronic Heart Failure) also studied the effect of iron supplementation, intravenous in this case, on exercise capacity in heart failure patients at 24 weeks. Iron depletion is a hallmark of heart failure, Dr. Sellke pointed out, so iron repletion could be a simple way to improve functional capacity.
MOMENTUM 3 (Multicenter Study of MagLev Technology in Patients Undergoing Mechanical Circulatory Support Therapy with HM3) evaluated the safety and effectiveness of the Thoratec HeartMate 3 left-ventricular assist device employing MagLev technology, which is said to facilitate the free flow of blood through the device. Roughly 1,000 patients with advanced, refractory heart failure were randomized to receive either the investigational HeartMate 3 or the HeartMate 2. The primary outcomes included short- and long-term survival and freedom from debilitating stroke. Trials such as this are very important, Dr. Sellke said, because the need for donor hearts far exceeds demand and better, cheaper LVADs that last longer could extend the lives of many thousands of patients every year.
MultiSENSE (Evaluation of Multisensor Data in Heart Failure Patients With Implanted Devices) collected information taken from sensors in an implanted cardiac synchronization therapy device in 1,000 patients to develop algorithms that would detect worsening heart failure. Multiple readmissions for heart failure are frequent and ineffective, and detecting the onset of worsening heart failure has the potential to bring those admissions way down, Dr. Sellke said.
The emphasis on this year’s American Heart Association Scientific Sessions in New Orleans is bigness: “Big science, big technology, and big networking opportunities,” the AHA 16 website says.
And so the 19 abstracts out of thousands submitted that got the biggest score from program committee for AHA 2016, led by Frank Sellke, MD, were chosen for presentation at four Late-Breaking Clinical Trials session previewed the late-breaking science.
Big trials for big questions
The first late-breaker session, on Sunday, Nov. 13, at 3:45 p.m., CT, is titled will, as its title says present the long-awaited results of four trials with large enrollment and long-term outcomes.
EUCLID (A Study Comparing Cardiovascular Effects of Ticagrelor and Clopidogrel in Patients With Peripheral Artery Disease) randomized an estimated 16,000 patients with symptomatic PAD to long-term antiplatelet monotherapy with either ticagrelor or clopidogrel to see which one would be superior in preventing the composite of cardiovascular death, myocardial infarction and ischemic stroke up to 40 months. Secondarily, it looked at acute limb ischemia, need for revascularization, and disease progression. “This could have tremendous implications for patients treat for pad trying to prevent CV disease,” Dr. Sellke said.
PRECISION (Prospective Randomized Evaluation of Celecoxib Integrated Safety vs Ibuprofen or Naproxen) harks back to 2005, when the Food and Drug Administration, wrestling with the growing evidence that NSAIDs were linked with cardiovascular events, asked for a large, cardiovascular outcomes trial. PRECISION, sponsored by Pfizer but run by an academic-led steering committee led by Steven Nissen, MD, now chief of cardiovascular medicine at the Cleveland Clinic, randomized some 20,000 arthritis patients with or at risk for cardiovascular disease to long-term pain treatment with celecoxib, naproxen, or ibuprofen for a planned follow-up of 2 years. The primary endpoint is a composite of cardiovascular death, nonfatal MI, and nonfatal stroke. Dr. Sellke noted that the results will be important for many physicians and patients wanting to minimize the risks associated with NSAIDs.
HOPE 3 (Heart Outcomes Evaluation 3), presented in April this year at the American College of Cardiology meeting in Chicago, showed the combination of rosuvastatin plus candesartan and hydrochlorothiazide reduced cardiovascular events in intermediate-risk patients with hypertension, regardless of their baseline LDL cholesterol and inflammatory biomarker levels. The analysis to be presented at AHA will show whether the combination has any effect on cognitive function. As evidence builds of the cardiovascular benefit of aggressive treatment of hypertension, as in the SPRINT trial, the results could be tremendously important, Dr. Sellke said.
TRUE AHF (Efficacy and Safety of Ularitide for the Treatment of Acute Decompensated Heart Failure) randomized about 2,150 patients with acute decompensated heart failure to receive a 48-hour intravenous infusion of the natriuretic peptide ularitide or placebo. The primary outcome is a composite of 48-hour improved in-hospital worsening or unchanged clinical conditions, as well as long-term cardiovascular mortality with a median follow-up of 7 months. Because there are no effective treatments for acute systolic heart failure, the results of TRUE AHF could be of tremendous benefit, Dr. Sellke said.
Pioneering the Future of HeART Interventions
The trials with the greatest impact for practice to be presented at AHA 2015, according to the Dr. Sellke’s admitted bias as a cardiothoracic surgeon, will all be presented in this second of the late-breaker sessions, on Monday, Nov. 14, at 10:45 a.m., CT.
ART (Arterial Revascularization Trial) was a comparison of single vs. bilateral internal mammary artery grafting in more than 3,000 randomized patients undergoing coronary artery bypass surgery (CABG). The outcomes of mortality, stroke, MI, and repeat revascularization were published in 2010, showing no differences between groups. The 5-year results to be presented on Monday may resolve some of the controversy surrounding the two methods, as surgeons and cardiologists are strongly divided on the benefits and risks of single, compared with double, internal mammary artery grafting.
FUTURE (Functional Testing Underlying Coronary Revascularization) compared fractional flow reserve–guided management with conventional management in roughly 900 patients undergoing revascularization with multivessel coronary artery disease. The primary outcome is a composite of death, MI, coronary revascularization, and stroke. FFR has received a lot of attention recently, Dr. Sellke said, because it looks at the physiologic, rather than the anatomic, effects of lesion on catheterization. The results will show whether there’s clinical benefit to adding FFR to angiography that will offset the additional time it takes to perform before PCI or CABG.
PIONEER AF-PCI (An Open-label, Randomized, Controlled, Multicenter Study Exploring Two Treatment Strategies of Rivaroxaban and a Dose-Adjusted Oral Vitamin K Antagonist Treatment Strategy in Subjects With Atrial Fibrillation Who Undergo Percutaneous Coronary Intervention) addressed the conundrum of treating anticoagulated patients with atrial fibrillation who are undergoing PCI with adequate dual-antiplatelet therapy – and avoiding bleeding events. About 2,000 patients were randomized to varying combinations of rivaroxaban or warfarin plus aspirin, ticagrelor prasugrel, and/or clopidogrel for 1 year. The primary outcome is significant bleeding. Dr. Sellke said that because drug-eluting stents require at least a year of DAPT, the PIONEER AF-PCI results will add knowledge in an important and controversial area.
GERMANY is a report from the German Aortic Valve Registry (GARY) on the 1-year outcomes of patients with intermediate-risk severe aortic stenosis who underwent either transcatheter or surgical aortic replacement on the efficacy and outcomes of the two approaches. Dr. Sellke noted that these results will be important because the patients in this registry were not at high risk or ineligible for surgical aortic replacement.
Insights from New Therapeutic Trials for Lipids
Of the five trials presented in this session on Tuesday, Nov. 15, at 10:45 a.m., CT, only one is in an approved treatment for lowering lipids. That is GLAGOV (Global Assessment of Plaque Regression With a PCSK9 Antibody as Measured by Intravascular Ultrasound), is looking at whether LDL lowering with the PCSK9 inhibitor evolocumab reduces atheroma volume in almost 1,000 patients.
Guiding the Momentum to Effect HF Outcomes – Ironing Out the Wrinkles
Two of the six heart failure trials presented in this session on Wednesday, Nov. 16, at 10:45 a.m., CT, study cardiorespiratory effects of iron, thus the title, Dr. Sellke said.
REDUCE LAP HF (A Study to Evaluate the DC Devices, Inc. IASD System II to REDUCE Elevated Left Atrial Pressure in Patients With Heart Failure). The primary outcome is a composite of death, stroke, MI, or a systemic embolic event at 6 months. The trial evaluated a transcatheter interatrial shunt device to left atrial pressure in patients with heart failure with preserved ejection fraction (HFpEF). In this type of diastolic heart failure in which patients’ hearts cannot relax, there is really no treatment, Dr. Sellke said. So although this treatment seems “hokey,” a positive result could be important.
ATHENA HF (Aldosterone Targeted Neurohormonal Combined with Natriuresis Therapy in Heart Failure) tested the diuretic spironolactone in heart failure. The investigators randomized 360 patients to high-dose spironolactone or usual care to see whether they could provide greater reductions of n-terminal prohormone of brain natriuretic peptide (NT-proBNP) levels within 96 hours. There’s evidence that spironolactone can provide symptomatic relief for patients with heart failure, so these results could be important, Dr. Sellke said.
IRONOUT HF (Oral Iron Repletion Effects on Oxygen Up Take in Heart Failure) randomized heart failure patients with iron deficiency to oral iron supplementation or placebo and measured peak oxygen uptake at 16 weeks.
EFFECT-HF (Effect of Ferric Carboxymaltose on Exercise Capacity in Patients with Iron Deficiency and Chronic Heart Failure) also studied the effect of iron supplementation, intravenous in this case, on exercise capacity in heart failure patients at 24 weeks. Iron depletion is a hallmark of heart failure, Dr. Sellke pointed out, so iron repletion could be a simple way to improve functional capacity.
MOMENTUM 3 (Multicenter Study of MagLev Technology in Patients Undergoing Mechanical Circulatory Support Therapy with HM3) evaluated the safety and effectiveness of the Thoratec HeartMate 3 left-ventricular assist device employing MagLev technology, which is said to facilitate the free flow of blood through the device. Roughly 1,000 patients with advanced, refractory heart failure were randomized to receive either the investigational HeartMate 3 or the HeartMate 2. The primary outcomes included short- and long-term survival and freedom from debilitating stroke. Trials such as this are very important, Dr. Sellke said, because the need for donor hearts far exceeds demand and better, cheaper LVADs that last longer could extend the lives of many thousands of patients every year.
MultiSENSE (Evaluation of Multisensor Data in Heart Failure Patients With Implanted Devices) collected information taken from sensors in an implanted cardiac synchronization therapy device in 1,000 patients to develop algorithms that would detect worsening heart failure. Multiple readmissions for heart failure are frequent and ineffective, and detecting the onset of worsening heart failure has the potential to bring those admissions way down, Dr. Sellke said.
Diabetes drugs with cardiovascular benefits broaden cardiology’s turf
The dramatic reduction in cardiovascular death and heart failure hospitalization seen during treatment with empagliflozin (Jardiance) in the EMPA-REG OUTCOME (Empagliflozin Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients) trial, for example, has prompted some cardiologists in the year since the first EMPA-REG report to become active prescribers of the drug to their patients who have type 2 diabetes and cardiovascular disease. The same evidence has driven other cardiologists who may not feel fully comfortable prescribing an antidiabetic drug on their own to enter into active partnerships with endocrinologists to work as a team to put diabetes patients with cardiovascular disease on empagliflozin.
In Dr. Fitchett’s practice, “if a patient with type 2 diabetes has an endocrinologist, then I will send a letter to that physician saying I think the patient should be on one of these drugs,” empagliflozin or liraglutide, he said. “If the patient is being treated by a primary care physician, then I will prescribe empagliflozin myself because most primary care physicians are not willing to prescribe it. I think more and more cardiologists are doing this. The great thing about empagliflozin and liraglutide is that they do not cause hypoglycemia and the adverse effect profiles are relatively good. As long as drug cost is not an issue, then as cardiologists we need to adjust glycemia control with cardiovascular benefit as we did years ago with statin treatment,” explained Dr. Fitchett, a cardiologist at St. Michael’s Hospital in Toronto and a senior collaborator and coauthor on the EMPA-REG study.
When results from the 4S [Scandinavian Simvastatin Survival Study] came out in 1994, proving that long-term statin treatment was both safe and increased survival in patients with coronary heart disease, “cardiologists took over lipid management from endocrinologists,” he recalled. “We now have a safe and simple treatment for glucose lowering that also cuts cardiovascular disease events, so cardiologists have to also be involved, at least to some extent. Their degree of involvement depends on their practice and who provides a patient’s primary diabetes care,” he said.
Cardiologists vary on empagliflozin
Other cardiologists are mixed in their take on personally prescribing antidiabetic drugs to high-risk patients with type 2 diabetes. Greg C. Fonarow, MD, has also aggressively taken to empagliflozin over the past year, especially for his patients with heart failure or at high risk for developing heart failure. The EMPA-REG results showed that empagliflozin’s potent impact on reducing cardiovascular death in patients linked closely with a reduction in heart failure hospitalizations. In his recent experience, endocrinologists as well as other physicians who care for patients with type 2 diabetes “are often reluctant to make any changes [in a patient’s hypoglycemic regimen], and in general they have not gravitated toward the treatments that have been shown to improve cardiovascular outcomes and instead focus solely on a patient’s hemoglobin A1c,” Dr. Fonarow said in an interview at the recent annual meeting of the Heart Failure Society of America.
He said he prescribes empagliflozin to patients with type 2 diabetes if they are hospitalized for heart failure or as outpatients, and he targets it to patients diagnosed with heart failure – including heart failure with preserved ejection fraction – as well as to patients with other forms of cardiovascular disease, closely following the EMPA-REG enrollment criteria. It’s too early in the experience with empagliflozin to use it preferentially in diabetes patients without cardiovascular disease or patients who in any other way fall outside the enrollment criteria for EMPA-REG, he said.
“I am happy to consult with their endocrinologist, or I tell patients to discuss this treatment with their endocrinologist. If the endocrinologist prescribes empagliflozin, great; if not, I feel an obligation to provide the best care I can to my patients. This is not a hard medication to use. The safety profile is good. Treatment with empagliflozin obviously has renal-function considerations, but that’s true for many drugs. The biggest challenge is what is covered by the patient’s insurance. We often need preauthorization.
“So far I have seen excellent responses in patients for both metabolic control and clinical responses in patients with heart failure. Their symptoms seem to improve,” said Dr. Fonarow, professor of medicine and co-chief of cardiology at the University of Southern California , Los Angeles.
While Dr. Fonarow cautioned that he also would not start empagliflozin in a patient with a HbA1c below 7%, he would seriously consider swapping out a patient’s drug for empagliflozin if it were a sulfonylurea or a dipeptidyl peptidase-4 inhibitor. He stopped short of suggesting a substitution of empagliflozin for metformin. In Dr. Fonarow’s opinion, the evidence for empagliflozin is also “more robust” than it has been for liraglutide or semaglutide. With what’s now known about the clinical impact of these drugs, he foresees a time when a combination between a SGLT-2 inhibitor, with its effect on heart failure, and a GLP-1 analogue, with its effect on atherosclerotic disease, may seem an ideal initial drug pairing for patients with type 2 diabetes and significant cardiovascular disease risk, with metformin relegated to a second-line role.
Other cardiologists endorsed a more collaborative approach to prescribing empagliflozin and liraglutide.
Another team-approach advocate is Robert O. Bonow, MD, cardiologist and professor of medicine at Northwestern University in Chicago. “Cardiologists are comfortable prescribing metformin and telling patients about lifestyle, but when it comes to newer antidiabetic drugs, that’s a new field, and a team approach may be best,” he said in an interview. “If possible, a cardiologist should have a friendly partnership with a diabetologist or endocrinologist who is expert in treating diabetes.” Many cardiologists now work in and for hospitals, and easy access to an endocrinologist is probably available, he noted.
But new analyses of the EMPA-REG data reported by Dr. Fitchett at the ESC congress showed that empagliflozin treatment exerted a similar benefit of reduced cardiovascular death regardless of whether patients had prevalent heart failure at entry into the study, incident heart failure during follow-up, or no heart failure of any sort.
Impact of heart failure in EMPA-REG
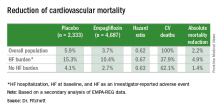
Roughly 10% of the 7,020 patients enrolled in EMPA-REG had heart failure at the time they entered the trial. During a median follow-up of just over 3 years, the incidence of new-onset heart failure – tallied as either a new heart failure hospitalization or a clinical episode deemed to be heart failure by an investigator – occurred in 4.6% of patients on empagliflozin and in 6.5% of patients in the placebo arm, a 1.9-percentage-point difference and a 30% relative risk reduction linked with empagliflozin use, Dr. Fitchett reported.
The main EMPA-REG outcome was a composite of cardiovascular death, nonfatal MI, and nonfatal stroke. This positive outcome in favor of empagliflozin treatment was primarily driven by a difference in the rate of cardiovascular death. In the new analysis, the relative reduction in cardiovascular deaths with empagliflozin compared with placebo was 29% among patients with prevalent heart failure at baseline, 35% among those who had an incident heart failure hospitalization during follow-up, 27% among patients with an incident heart failure episode diagnosed by an investigator during follow-up, 33% among the combined group of trial patients with any form of heart failure at trial entry or during the trial (those with prevalent heart failure at baseline plus those with an incident event), and 37% among the large number of patients in the trial who remained free from any indication of heart failure during follow-up.
In short, treatment with empagliflozin “reduced cardiovascular mortality by the same relative amount” regardless of whether patients did or did not have heart failure during the trial,” Dr. Fitchett concluded.
Additional secondary analyses from EMPA-REG reported at the ESC congress in August also documented that the benefit from empagliflozin treatment was roughly the same regardless of the age of patients enrolled in the trial and regardless of patients’ blood level of LDL cholesterol at entry into the study. These findings provide “confidence in the consistency of the effect” by empagliflozin, Dr. Fitchett said.
The endocrinologists’ view
“Most cardiologists are not thoroughly familiar with the full palette of medications for hyperglycemia. Selection of medication should not be made solely on the basis of results from a cardiovascular outcomes trial,” said Helena W. Rodbard, MD, a clinical endocrinologist in Rockville, Md.
“The EMPA-REG OUTCOMES and LEADER results are very exciting and encouraging. When all other factors are equal, the cardiovascular results could sway the decision about which medication to use. But an endocrinologist is in the best position to balance the many factors when choosing combination therapy and to set a target level for HbA1c, fasting blood glucose, and postprandial glucose, and to adjust therapy to minimize the risk of hypoglycemia,” Dr. Rodbard said in an interview.
He called empagliflozin a drug with “interesting promise,” especially for patients with incipient heart failure. The extra cardiovascular benefit from the GLP-1 analogues is “less settled,” although the liraglutide and semaglutide trial results are important and mean these drugs need more consideration and study. The EMPA-REG results were more clearly positive, he said.
“Metformin is still the initial drug” for most patients with type 2 diabetes, echoed Dr. Levy. Drugs like empagliflozin and liraglutide are usually used in combination with metformin.
“Like many endocrinologists, I have for some time used the oral SGLT-2 inhibitors and GLP-1 analogues in combination with metformin. It made sense before the recent cardiovascular data appeared, and it makes even more sense now,” said Dr. Jellinger, professor of clinical medicine and an endocrinologist at the University of Miami.
“Endocrinologists and diabetologists are aware that cardiologists have been taking a larger role in the care of patients with diabetes,” noted Dr. Rodbard. “I favor cardiologists and endocrinologists working in concert to improve the care of patients with diabetes.”
“Over the next few years, we will need to decide whether to treat patients with type 2 diabetes with an agent with proven benefits,” said Dr. Fitchett. “Until the results from EMPA-REG and the LEADER trial came out, there was no specific glucose-lowering agent that also reduced cardiovascular events. Some cardiologists might ask when they should get involved in managing patients with type 2 diabetes. What I would do for patients with a history of cardiovascular disease who develop new type 2 diabetes is start empagliflozin as their first drug,” Dr. Fitchett said, though he admitted that no evidence yet exists to back that approach.
The EMPA-REG trial was sponsored by Boehringer Ingelheim and by Eli Lilly, the companies that market empagliflozin. The LEADER trial was sponsored in part by Novo Nordisk, the company that markets liraglutide. Dr. Fitchett and Dr. Mentz were both researchers for EMPA-REG. Dr. Fitchett has been a consultant to AstraZeneca, Merck, and Amgen. Dr. Mentz has been an adviser to Boehringer Ingelheim. Dr. Fonarow has been an adviser to Amgen, Janssen, Novartis, and ZS Pharma. Dr. Bozkurt had no disclosures. Dr. Bonow has been a consultant to Gilead. Dr. Jellinger has been a speaker on behalf of Boehringer-Ingelheim, Novo Nordisk, Merck, and Janssen. Dr. Rodbard has been a consultant to or speaker for several drug companies including Boehringer-Ingelheim, Eli Lilly, and Novo Nordisk. Dr. Levy has been a speaker on behalf of Boehringer-Ingelheim, Eli Lilly, Novo Nordisk, and AstraZeneca. Dr. Hellman had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
The dramatic reduction in cardiovascular death and heart failure hospitalization seen during treatment with empagliflozin (Jardiance) in the EMPA-REG OUTCOME (Empagliflozin Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients) trial, for example, has prompted some cardiologists in the year since the first EMPA-REG report to become active prescribers of the drug to their patients who have type 2 diabetes and cardiovascular disease. The same evidence has driven other cardiologists who may not feel fully comfortable prescribing an antidiabetic drug on their own to enter into active partnerships with endocrinologists to work as a team to put diabetes patients with cardiovascular disease on empagliflozin.
In Dr. Fitchett’s practice, “if a patient with type 2 diabetes has an endocrinologist, then I will send a letter to that physician saying I think the patient should be on one of these drugs,” empagliflozin or liraglutide, he said. “If the patient is being treated by a primary care physician, then I will prescribe empagliflozin myself because most primary care physicians are not willing to prescribe it. I think more and more cardiologists are doing this. The great thing about empagliflozin and liraglutide is that they do not cause hypoglycemia and the adverse effect profiles are relatively good. As long as drug cost is not an issue, then as cardiologists we need to adjust glycemia control with cardiovascular benefit as we did years ago with statin treatment,” explained Dr. Fitchett, a cardiologist at St. Michael’s Hospital in Toronto and a senior collaborator and coauthor on the EMPA-REG study.
When results from the 4S [Scandinavian Simvastatin Survival Study] came out in 1994, proving that long-term statin treatment was both safe and increased survival in patients with coronary heart disease, “cardiologists took over lipid management from endocrinologists,” he recalled. “We now have a safe and simple treatment for glucose lowering that also cuts cardiovascular disease events, so cardiologists have to also be involved, at least to some extent. Their degree of involvement depends on their practice and who provides a patient’s primary diabetes care,” he said.
Cardiologists vary on empagliflozin
Other cardiologists are mixed in their take on personally prescribing antidiabetic drugs to high-risk patients with type 2 diabetes. Greg C. Fonarow, MD, has also aggressively taken to empagliflozin over the past year, especially for his patients with heart failure or at high risk for developing heart failure. The EMPA-REG results showed that empagliflozin’s potent impact on reducing cardiovascular death in patients linked closely with a reduction in heart failure hospitalizations. In his recent experience, endocrinologists as well as other physicians who care for patients with type 2 diabetes “are often reluctant to make any changes [in a patient’s hypoglycemic regimen], and in general they have not gravitated toward the treatments that have been shown to improve cardiovascular outcomes and instead focus solely on a patient’s hemoglobin A1c,” Dr. Fonarow said in an interview at the recent annual meeting of the Heart Failure Society of America.
He said he prescribes empagliflozin to patients with type 2 diabetes if they are hospitalized for heart failure or as outpatients, and he targets it to patients diagnosed with heart failure – including heart failure with preserved ejection fraction – as well as to patients with other forms of cardiovascular disease, closely following the EMPA-REG enrollment criteria. It’s too early in the experience with empagliflozin to use it preferentially in diabetes patients without cardiovascular disease or patients who in any other way fall outside the enrollment criteria for EMPA-REG, he said.
“I am happy to consult with their endocrinologist, or I tell patients to discuss this treatment with their endocrinologist. If the endocrinologist prescribes empagliflozin, great; if not, I feel an obligation to provide the best care I can to my patients. This is not a hard medication to use. The safety profile is good. Treatment with empagliflozin obviously has renal-function considerations, but that’s true for many drugs. The biggest challenge is what is covered by the patient’s insurance. We often need preauthorization.
“So far I have seen excellent responses in patients for both metabolic control and clinical responses in patients with heart failure. Their symptoms seem to improve,” said Dr. Fonarow, professor of medicine and co-chief of cardiology at the University of Southern California , Los Angeles.
While Dr. Fonarow cautioned that he also would not start empagliflozin in a patient with a HbA1c below 7%, he would seriously consider swapping out a patient’s drug for empagliflozin if it were a sulfonylurea or a dipeptidyl peptidase-4 inhibitor. He stopped short of suggesting a substitution of empagliflozin for metformin. In Dr. Fonarow’s opinion, the evidence for empagliflozin is also “more robust” than it has been for liraglutide or semaglutide. With what’s now known about the clinical impact of these drugs, he foresees a time when a combination between a SGLT-2 inhibitor, with its effect on heart failure, and a GLP-1 analogue, with its effect on atherosclerotic disease, may seem an ideal initial drug pairing for patients with type 2 diabetes and significant cardiovascular disease risk, with metformin relegated to a second-line role.
Other cardiologists endorsed a more collaborative approach to prescribing empagliflozin and liraglutide.
Another team-approach advocate is Robert O. Bonow, MD, cardiologist and professor of medicine at Northwestern University in Chicago. “Cardiologists are comfortable prescribing metformin and telling patients about lifestyle, but when it comes to newer antidiabetic drugs, that’s a new field, and a team approach may be best,” he said in an interview. “If possible, a cardiologist should have a friendly partnership with a diabetologist or endocrinologist who is expert in treating diabetes.” Many cardiologists now work in and for hospitals, and easy access to an endocrinologist is probably available, he noted.
But new analyses of the EMPA-REG data reported by Dr. Fitchett at the ESC congress showed that empagliflozin treatment exerted a similar benefit of reduced cardiovascular death regardless of whether patients had prevalent heart failure at entry into the study, incident heart failure during follow-up, or no heart failure of any sort.
Impact of heart failure in EMPA-REG
Roughly 10% of the 7,020 patients enrolled in EMPA-REG had heart failure at the time they entered the trial. During a median follow-up of just over 3 years, the incidence of new-onset heart failure – tallied as either a new heart failure hospitalization or a clinical episode deemed to be heart failure by an investigator – occurred in 4.6% of patients on empagliflozin and in 6.5% of patients in the placebo arm, a 1.9-percentage-point difference and a 30% relative risk reduction linked with empagliflozin use, Dr. Fitchett reported.
The main EMPA-REG outcome was a composite of cardiovascular death, nonfatal MI, and nonfatal stroke. This positive outcome in favor of empagliflozin treatment was primarily driven by a difference in the rate of cardiovascular death. In the new analysis, the relative reduction in cardiovascular deaths with empagliflozin compared with placebo was 29% among patients with prevalent heart failure at baseline, 35% among those who had an incident heart failure hospitalization during follow-up, 27% among patients with an incident heart failure episode diagnosed by an investigator during follow-up, 33% among the combined group of trial patients with any form of heart failure at trial entry or during the trial (those with prevalent heart failure at baseline plus those with an incident event), and 37% among the large number of patients in the trial who remained free from any indication of heart failure during follow-up.
In short, treatment with empagliflozin “reduced cardiovascular mortality by the same relative amount” regardless of whether patients did or did not have heart failure during the trial,” Dr. Fitchett concluded.
Additional secondary analyses from EMPA-REG reported at the ESC congress in August also documented that the benefit from empagliflozin treatment was roughly the same regardless of the age of patients enrolled in the trial and regardless of patients’ blood level of LDL cholesterol at entry into the study. These findings provide “confidence in the consistency of the effect” by empagliflozin, Dr. Fitchett said.
The endocrinologists’ view
“Most cardiologists are not thoroughly familiar with the full palette of medications for hyperglycemia. Selection of medication should not be made solely on the basis of results from a cardiovascular outcomes trial,” said Helena W. Rodbard, MD, a clinical endocrinologist in Rockville, Md.
“The EMPA-REG OUTCOMES and LEADER results are very exciting and encouraging. When all other factors are equal, the cardiovascular results could sway the decision about which medication to use. But an endocrinologist is in the best position to balance the many factors when choosing combination therapy and to set a target level for HbA1c, fasting blood glucose, and postprandial glucose, and to adjust therapy to minimize the risk of hypoglycemia,” Dr. Rodbard said in an interview.
He called empagliflozin a drug with “interesting promise,” especially for patients with incipient heart failure. The extra cardiovascular benefit from the GLP-1 analogues is “less settled,” although the liraglutide and semaglutide trial results are important and mean these drugs need more consideration and study. The EMPA-REG results were more clearly positive, he said.
“Metformin is still the initial drug” for most patients with type 2 diabetes, echoed Dr. Levy. Drugs like empagliflozin and liraglutide are usually used in combination with metformin.
“Like many endocrinologists, I have for some time used the oral SGLT-2 inhibitors and GLP-1 analogues in combination with metformin. It made sense before the recent cardiovascular data appeared, and it makes even more sense now,” said Dr. Jellinger, professor of clinical medicine and an endocrinologist at the University of Miami.
“Endocrinologists and diabetologists are aware that cardiologists have been taking a larger role in the care of patients with diabetes,” noted Dr. Rodbard. “I favor cardiologists and endocrinologists working in concert to improve the care of patients with diabetes.”
“Over the next few years, we will need to decide whether to treat patients with type 2 diabetes with an agent with proven benefits,” said Dr. Fitchett. “Until the results from EMPA-REG and the LEADER trial came out, there was no specific glucose-lowering agent that also reduced cardiovascular events. Some cardiologists might ask when they should get involved in managing patients with type 2 diabetes. What I would do for patients with a history of cardiovascular disease who develop new type 2 diabetes is start empagliflozin as their first drug,” Dr. Fitchett said, though he admitted that no evidence yet exists to back that approach.
The EMPA-REG trial was sponsored by Boehringer Ingelheim and by Eli Lilly, the companies that market empagliflozin. The LEADER trial was sponsored in part by Novo Nordisk, the company that markets liraglutide. Dr. Fitchett and Dr. Mentz were both researchers for EMPA-REG. Dr. Fitchett has been a consultant to AstraZeneca, Merck, and Amgen. Dr. Mentz has been an adviser to Boehringer Ingelheim. Dr. Fonarow has been an adviser to Amgen, Janssen, Novartis, and ZS Pharma. Dr. Bozkurt had no disclosures. Dr. Bonow has been a consultant to Gilead. Dr. Jellinger has been a speaker on behalf of Boehringer-Ingelheim, Novo Nordisk, Merck, and Janssen. Dr. Rodbard has been a consultant to or speaker for several drug companies including Boehringer-Ingelheim, Eli Lilly, and Novo Nordisk. Dr. Levy has been a speaker on behalf of Boehringer-Ingelheim, Eli Lilly, Novo Nordisk, and AstraZeneca. Dr. Hellman had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
The dramatic reduction in cardiovascular death and heart failure hospitalization seen during treatment with empagliflozin (Jardiance) in the EMPA-REG OUTCOME (Empagliflozin Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients) trial, for example, has prompted some cardiologists in the year since the first EMPA-REG report to become active prescribers of the drug to their patients who have type 2 diabetes and cardiovascular disease. The same evidence has driven other cardiologists who may not feel fully comfortable prescribing an antidiabetic drug on their own to enter into active partnerships with endocrinologists to work as a team to put diabetes patients with cardiovascular disease on empagliflozin.
In Dr. Fitchett’s practice, “if a patient with type 2 diabetes has an endocrinologist, then I will send a letter to that physician saying I think the patient should be on one of these drugs,” empagliflozin or liraglutide, he said. “If the patient is being treated by a primary care physician, then I will prescribe empagliflozin myself because most primary care physicians are not willing to prescribe it. I think more and more cardiologists are doing this. The great thing about empagliflozin and liraglutide is that they do not cause hypoglycemia and the adverse effect profiles are relatively good. As long as drug cost is not an issue, then as cardiologists we need to adjust glycemia control with cardiovascular benefit as we did years ago with statin treatment,” explained Dr. Fitchett, a cardiologist at St. Michael’s Hospital in Toronto and a senior collaborator and coauthor on the EMPA-REG study.
When results from the 4S [Scandinavian Simvastatin Survival Study] came out in 1994, proving that long-term statin treatment was both safe and increased survival in patients with coronary heart disease, “cardiologists took over lipid management from endocrinologists,” he recalled. “We now have a safe and simple treatment for glucose lowering that also cuts cardiovascular disease events, so cardiologists have to also be involved, at least to some extent. Their degree of involvement depends on their practice and who provides a patient’s primary diabetes care,” he said.
Cardiologists vary on empagliflozin
Other cardiologists are mixed in their take on personally prescribing antidiabetic drugs to high-risk patients with type 2 diabetes. Greg C. Fonarow, MD, has also aggressively taken to empagliflozin over the past year, especially for his patients with heart failure or at high risk for developing heart failure. The EMPA-REG results showed that empagliflozin’s potent impact on reducing cardiovascular death in patients linked closely with a reduction in heart failure hospitalizations. In his recent experience, endocrinologists as well as other physicians who care for patients with type 2 diabetes “are often reluctant to make any changes [in a patient’s hypoglycemic regimen], and in general they have not gravitated toward the treatments that have been shown to improve cardiovascular outcomes and instead focus solely on a patient’s hemoglobin A1c,” Dr. Fonarow said in an interview at the recent annual meeting of the Heart Failure Society of America.
He said he prescribes empagliflozin to patients with type 2 diabetes if they are hospitalized for heart failure or as outpatients, and he targets it to patients diagnosed with heart failure – including heart failure with preserved ejection fraction – as well as to patients with other forms of cardiovascular disease, closely following the EMPA-REG enrollment criteria. It’s too early in the experience with empagliflozin to use it preferentially in diabetes patients without cardiovascular disease or patients who in any other way fall outside the enrollment criteria for EMPA-REG, he said.
“I am happy to consult with their endocrinologist, or I tell patients to discuss this treatment with their endocrinologist. If the endocrinologist prescribes empagliflozin, great; if not, I feel an obligation to provide the best care I can to my patients. This is not a hard medication to use. The safety profile is good. Treatment with empagliflozin obviously has renal-function considerations, but that’s true for many drugs. The biggest challenge is what is covered by the patient’s insurance. We often need preauthorization.
“So far I have seen excellent responses in patients for both metabolic control and clinical responses in patients with heart failure. Their symptoms seem to improve,” said Dr. Fonarow, professor of medicine and co-chief of cardiology at the University of Southern California , Los Angeles.
While Dr. Fonarow cautioned that he also would not start empagliflozin in a patient with a HbA1c below 7%, he would seriously consider swapping out a patient’s drug for empagliflozin if it were a sulfonylurea or a dipeptidyl peptidase-4 inhibitor. He stopped short of suggesting a substitution of empagliflozin for metformin. In Dr. Fonarow’s opinion, the evidence for empagliflozin is also “more robust” than it has been for liraglutide or semaglutide. With what’s now known about the clinical impact of these drugs, he foresees a time when a combination between a SGLT-2 inhibitor, with its effect on heart failure, and a GLP-1 analogue, with its effect on atherosclerotic disease, may seem an ideal initial drug pairing for patients with type 2 diabetes and significant cardiovascular disease risk, with metformin relegated to a second-line role.
Other cardiologists endorsed a more collaborative approach to prescribing empagliflozin and liraglutide.
Another team-approach advocate is Robert O. Bonow, MD, cardiologist and professor of medicine at Northwestern University in Chicago. “Cardiologists are comfortable prescribing metformin and telling patients about lifestyle, but when it comes to newer antidiabetic drugs, that’s a new field, and a team approach may be best,” he said in an interview. “If possible, a cardiologist should have a friendly partnership with a diabetologist or endocrinologist who is expert in treating diabetes.” Many cardiologists now work in and for hospitals, and easy access to an endocrinologist is probably available, he noted.
But new analyses of the EMPA-REG data reported by Dr. Fitchett at the ESC congress showed that empagliflozin treatment exerted a similar benefit of reduced cardiovascular death regardless of whether patients had prevalent heart failure at entry into the study, incident heart failure during follow-up, or no heart failure of any sort.
Impact of heart failure in EMPA-REG
Roughly 10% of the 7,020 patients enrolled in EMPA-REG had heart failure at the time they entered the trial. During a median follow-up of just over 3 years, the incidence of new-onset heart failure – tallied as either a new heart failure hospitalization or a clinical episode deemed to be heart failure by an investigator – occurred in 4.6% of patients on empagliflozin and in 6.5% of patients in the placebo arm, a 1.9-percentage-point difference and a 30% relative risk reduction linked with empagliflozin use, Dr. Fitchett reported.
The main EMPA-REG outcome was a composite of cardiovascular death, nonfatal MI, and nonfatal stroke. This positive outcome in favor of empagliflozin treatment was primarily driven by a difference in the rate of cardiovascular death. In the new analysis, the relative reduction in cardiovascular deaths with empagliflozin compared with placebo was 29% among patients with prevalent heart failure at baseline, 35% among those who had an incident heart failure hospitalization during follow-up, 27% among patients with an incident heart failure episode diagnosed by an investigator during follow-up, 33% among the combined group of trial patients with any form of heart failure at trial entry or during the trial (those with prevalent heart failure at baseline plus those with an incident event), and 37% among the large number of patients in the trial who remained free from any indication of heart failure during follow-up.
In short, treatment with empagliflozin “reduced cardiovascular mortality by the same relative amount” regardless of whether patients did or did not have heart failure during the trial,” Dr. Fitchett concluded.
Additional secondary analyses from EMPA-REG reported at the ESC congress in August also documented that the benefit from empagliflozin treatment was roughly the same regardless of the age of patients enrolled in the trial and regardless of patients’ blood level of LDL cholesterol at entry into the study. These findings provide “confidence in the consistency of the effect” by empagliflozin, Dr. Fitchett said.
The endocrinologists’ view
“Most cardiologists are not thoroughly familiar with the full palette of medications for hyperglycemia. Selection of medication should not be made solely on the basis of results from a cardiovascular outcomes trial,” said Helena W. Rodbard, MD, a clinical endocrinologist in Rockville, Md.
“The EMPA-REG OUTCOMES and LEADER results are very exciting and encouraging. When all other factors are equal, the cardiovascular results could sway the decision about which medication to use. But an endocrinologist is in the best position to balance the many factors when choosing combination therapy and to set a target level for HbA1c, fasting blood glucose, and postprandial glucose, and to adjust therapy to minimize the risk of hypoglycemia,” Dr. Rodbard said in an interview.
He called empagliflozin a drug with “interesting promise,” especially for patients with incipient heart failure. The extra cardiovascular benefit from the GLP-1 analogues is “less settled,” although the liraglutide and semaglutide trial results are important and mean these drugs need more consideration and study. The EMPA-REG results were more clearly positive, he said.
“Metformin is still the initial drug” for most patients with type 2 diabetes, echoed Dr. Levy. Drugs like empagliflozin and liraglutide are usually used in combination with metformin.
“Like many endocrinologists, I have for some time used the oral SGLT-2 inhibitors and GLP-1 analogues in combination with metformin. It made sense before the recent cardiovascular data appeared, and it makes even more sense now,” said Dr. Jellinger, professor of clinical medicine and an endocrinologist at the University of Miami.
“Endocrinologists and diabetologists are aware that cardiologists have been taking a larger role in the care of patients with diabetes,” noted Dr. Rodbard. “I favor cardiologists and endocrinologists working in concert to improve the care of patients with diabetes.”
“Over the next few years, we will need to decide whether to treat patients with type 2 diabetes with an agent with proven benefits,” said Dr. Fitchett. “Until the results from EMPA-REG and the LEADER trial came out, there was no specific glucose-lowering agent that also reduced cardiovascular events. Some cardiologists might ask when they should get involved in managing patients with type 2 diabetes. What I would do for patients with a history of cardiovascular disease who develop new type 2 diabetes is start empagliflozin as their first drug,” Dr. Fitchett said, though he admitted that no evidence yet exists to back that approach.
The EMPA-REG trial was sponsored by Boehringer Ingelheim and by Eli Lilly, the companies that market empagliflozin. The LEADER trial was sponsored in part by Novo Nordisk, the company that markets liraglutide. Dr. Fitchett and Dr. Mentz were both researchers for EMPA-REG. Dr. Fitchett has been a consultant to AstraZeneca, Merck, and Amgen. Dr. Mentz has been an adviser to Boehringer Ingelheim. Dr. Fonarow has been an adviser to Amgen, Janssen, Novartis, and ZS Pharma. Dr. Bozkurt had no disclosures. Dr. Bonow has been a consultant to Gilead. Dr. Jellinger has been a speaker on behalf of Boehringer-Ingelheim, Novo Nordisk, Merck, and Janssen. Dr. Rodbard has been a consultant to or speaker for several drug companies including Boehringer-Ingelheim, Eli Lilly, and Novo Nordisk. Dr. Levy has been a speaker on behalf of Boehringer-Ingelheim, Eli Lilly, Novo Nordisk, and AstraZeneca. Dr. Hellman had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Rheumatoid arthritis increases heart failure risk
ROME – Rheumatoid arthritis is associated with a significantly increased risk of hospitalization for heart failure, according to a nationwide Danish study, Usman Khalid, MD, reported at the annual congress of the European Society of Cardiology.
As a chronic systemic inflammatory disease, rheumatoid arthritis (RA) has been associated with increased risk of a variety of comorbid conditions, including cardiovascular disease. But RA’s relationship specifically with heart failure hasn’t previously been looked at in the comprehensive way that’s possible in Denmark, where linked national registries enable researchers to follow health issues in the entire population from birth to death, noted Dr. Khalid of the University of Copenhagen.
In an analysis adjusted for age, sex, and calendar year, individuals with RA were 83% more likely to be hospitalized for heart failure than were the non-RA Danish population. In a fully adjusted analysis that controlled for those potential confounders as well as comorbid conditions, smoking, alcohol intake, socioeconomic status, and prescription medications, patients with RA remained at a statistically significant and clinically meaningful 38% increased risk of heart failure hospitalization.
Further studies are planned to determine the underlying mechanisms of this association, Dr. Khalid added.
He reported having no financial conflicts of interest regarding this study, which was supported by an unrestricted grant from Leo Pharma.
ROME – Rheumatoid arthritis is associated with a significantly increased risk of hospitalization for heart failure, according to a nationwide Danish study, Usman Khalid, MD, reported at the annual congress of the European Society of Cardiology.
As a chronic systemic inflammatory disease, rheumatoid arthritis (RA) has been associated with increased risk of a variety of comorbid conditions, including cardiovascular disease. But RA’s relationship specifically with heart failure hasn’t previously been looked at in the comprehensive way that’s possible in Denmark, where linked national registries enable researchers to follow health issues in the entire population from birth to death, noted Dr. Khalid of the University of Copenhagen.
In an analysis adjusted for age, sex, and calendar year, individuals with RA were 83% more likely to be hospitalized for heart failure than were the non-RA Danish population. In a fully adjusted analysis that controlled for those potential confounders as well as comorbid conditions, smoking, alcohol intake, socioeconomic status, and prescription medications, patients with RA remained at a statistically significant and clinically meaningful 38% increased risk of heart failure hospitalization.
Further studies are planned to determine the underlying mechanisms of this association, Dr. Khalid added.
He reported having no financial conflicts of interest regarding this study, which was supported by an unrestricted grant from Leo Pharma.
ROME – Rheumatoid arthritis is associated with a significantly increased risk of hospitalization for heart failure, according to a nationwide Danish study, Usman Khalid, MD, reported at the annual congress of the European Society of Cardiology.
As a chronic systemic inflammatory disease, rheumatoid arthritis (RA) has been associated with increased risk of a variety of comorbid conditions, including cardiovascular disease. But RA’s relationship specifically with heart failure hasn’t previously been looked at in the comprehensive way that’s possible in Denmark, where linked national registries enable researchers to follow health issues in the entire population from birth to death, noted Dr. Khalid of the University of Copenhagen.
In an analysis adjusted for age, sex, and calendar year, individuals with RA were 83% more likely to be hospitalized for heart failure than were the non-RA Danish population. In a fully adjusted analysis that controlled for those potential confounders as well as comorbid conditions, smoking, alcohol intake, socioeconomic status, and prescription medications, patients with RA remained at a statistically significant and clinically meaningful 38% increased risk of heart failure hospitalization.
Further studies are planned to determine the underlying mechanisms of this association, Dr. Khalid added.
He reported having no financial conflicts of interest regarding this study, which was supported by an unrestricted grant from Leo Pharma.
AT THE ESC CONGRESS 2016
Key clinical point:
Major finding: The incidence rate for heart failure hospitalization was 7.37 per 1,000 person-years in Danish adults with rheumatoid arthritis, compared with 2.45 per 1,000 in the general population.
Data source: This study utilized Danish comprehensive national registries to determine the rate at which 13,800 Danes with rheumatoid arthritis and no baseline history of heart failure were hospitalized for heart failure during 15 years of follow-up.
Disclosures: The study was supported by an unrestricted grant from Leo Pharma. The presenter reported having no financial conflicts of interest.
Heart failure targets African Americans
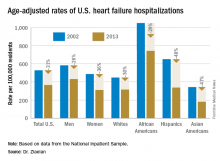
ORLANDO – The disparity in U.S. heart failure incidence continued undiminished during 2002-2013, with African Americans maintaining a steady 2.3-fold increased rate of heart failure, compared with whites, based on national levels of heart failure hospitalizations, a reasonable surrogate for incidence rates, Boback Ziaeian, MD, reported at the annual scientific meeting of the Heart Failure Society of America.
The same period also showed a substantial relative improvement in the heart failure hospitalization rates among U.S. Hispanics, compared with whites, so that, by 2013, the ethnic disparity seen in 2002 between Hispanics and whites largely disappeared, reported Dr. Ziaeian, a cardiologist at the University of California, Los Angeles. The data he analyzed also showed that Asian Americans had the lowest heart failure hospitalization rates of any racial or ethnic group throughout the 11-year period, and that the incidence of heart failure fell more sharply in women than in men during the period, based on the hospitalization numbers.
Age-adjusted heart failure hospitalizations among whites dropped by 30%, and among African Americans by a nearly identical 29%. But this maintained a greater than twofold disparity in rates between the two groups. Among whites, the rate per 100,000 fell from 448 to 315; among African Americans, it dropped from 1,048 to 741. In 2013, the rate of heart failure hospitalizations was 2.4-fold higher in African Americans, compared with whites.
Heart failure hospitalizations fell among Hispanics from 650 per 100,000 to 337 per 100,000 in 2013, a 48% drop that brought the rate among Hispanics to nearly the same as among whites. Asian Americans remained the group with the least heart failure throughout the period, falling from 343 hospitalizations per 100,000 in 2002 to 181 per 100,000 in 2013, a 47% drop.
Among women, the age-adjusted rate per 100,000 fell from 486 to 311, a 36% drop, compared with a decrease from 582 to 431 per 100,000 in men, a 26% reduction. Lower incidence in women may reflect better risk factor control during the study period, compared with men, such as a higher rate of quiting smoking and better treatment compliance, Dr. Ziaeian suggested.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ORLANDO – The disparity in U.S. heart failure incidence continued undiminished during 2002-2013, with African Americans maintaining a steady 2.3-fold increased rate of heart failure, compared with whites, based on national levels of heart failure hospitalizations, a reasonable surrogate for incidence rates, Boback Ziaeian, MD, reported at the annual scientific meeting of the Heart Failure Society of America.
The same period also showed a substantial relative improvement in the heart failure hospitalization rates among U.S. Hispanics, compared with whites, so that, by 2013, the ethnic disparity seen in 2002 between Hispanics and whites largely disappeared, reported Dr. Ziaeian, a cardiologist at the University of California, Los Angeles. The data he analyzed also showed that Asian Americans had the lowest heart failure hospitalization rates of any racial or ethnic group throughout the 11-year period, and that the incidence of heart failure fell more sharply in women than in men during the period, based on the hospitalization numbers.
Age-adjusted heart failure hospitalizations among whites dropped by 30%, and among African Americans by a nearly identical 29%. But this maintained a greater than twofold disparity in rates between the two groups. Among whites, the rate per 100,000 fell from 448 to 315; among African Americans, it dropped from 1,048 to 741. In 2013, the rate of heart failure hospitalizations was 2.4-fold higher in African Americans, compared with whites.
Heart failure hospitalizations fell among Hispanics from 650 per 100,000 to 337 per 100,000 in 2013, a 48% drop that brought the rate among Hispanics to nearly the same as among whites. Asian Americans remained the group with the least heart failure throughout the period, falling from 343 hospitalizations per 100,000 in 2002 to 181 per 100,000 in 2013, a 47% drop.
Among women, the age-adjusted rate per 100,000 fell from 486 to 311, a 36% drop, compared with a decrease from 582 to 431 per 100,000 in men, a 26% reduction. Lower incidence in women may reflect better risk factor control during the study period, compared with men, such as a higher rate of quiting smoking and better treatment compliance, Dr. Ziaeian suggested.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ORLANDO – The disparity in U.S. heart failure incidence continued undiminished during 2002-2013, with African Americans maintaining a steady 2.3-fold increased rate of heart failure, compared with whites, based on national levels of heart failure hospitalizations, a reasonable surrogate for incidence rates, Boback Ziaeian, MD, reported at the annual scientific meeting of the Heart Failure Society of America.
The same period also showed a substantial relative improvement in the heart failure hospitalization rates among U.S. Hispanics, compared with whites, so that, by 2013, the ethnic disparity seen in 2002 between Hispanics and whites largely disappeared, reported Dr. Ziaeian, a cardiologist at the University of California, Los Angeles. The data he analyzed also showed that Asian Americans had the lowest heart failure hospitalization rates of any racial or ethnic group throughout the 11-year period, and that the incidence of heart failure fell more sharply in women than in men during the period, based on the hospitalization numbers.
Age-adjusted heart failure hospitalizations among whites dropped by 30%, and among African Americans by a nearly identical 29%. But this maintained a greater than twofold disparity in rates between the two groups. Among whites, the rate per 100,000 fell from 448 to 315; among African Americans, it dropped from 1,048 to 741. In 2013, the rate of heart failure hospitalizations was 2.4-fold higher in African Americans, compared with whites.
Heart failure hospitalizations fell among Hispanics from 650 per 100,000 to 337 per 100,000 in 2013, a 48% drop that brought the rate among Hispanics to nearly the same as among whites. Asian Americans remained the group with the least heart failure throughout the period, falling from 343 hospitalizations per 100,000 in 2002 to 181 per 100,000 in 2013, a 47% drop.
Among women, the age-adjusted rate per 100,000 fell from 486 to 311, a 36% drop, compared with a decrease from 582 to 431 per 100,000 in men, a 26% reduction. Lower incidence in women may reflect better risk factor control during the study period, compared with men, such as a higher rate of quiting smoking and better treatment compliance, Dr. Ziaeian suggested.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT THE HFSA ANNUAL SCIENTIFIC MEETING
Key clinical point:
Major finding: In 2013, age-adjusted heart failure hospitalization was 741/100,000 in African Americans and 315/100,000 in whites.
Data source: The National Inpatient Sample and U.S. Census data.
Disclosures: Dr. Ziaeian had no disclosures.
Adaptive servo ventilation cuts atrial fib burden
ORLANDO – Adaptive servo ventilation produced a significant and clinically meaningful reduction in atrial fibrillation burden in patients with heart failure and sleep apnea in results from an exploratory, prospective, randomized study with 35 patients.
Adaptive servo ventilation (ASV) “may be an effective antiarrhythmic treatment producing a significant reduction in atrial fibrillation without clear evidence of being proarrhythmogenic,” Jonathan P. Piccini, MD, said at the annual scientific meeting of the Heart Failure Society of America. “Given the potential importance of this finding further studies should validate and quantify the efficacy of ASV for reducing atrial fibrillation in patients with or without heart failure.”
“A mound of data has shown that treating sleep apnea reduced arrhythmias, but until now it’s all been observational and retrospective,” Dr. Piccini, an electrophysiologist at Duke University in Durham, N.C., said in an interview. The study he reported is “the first time” the arrhythmia effects of a sleep apnea intervention, in this case ASV, was studied in a prospective, randomized way while using implanted devices to measure the antiarrhythmic effect of the treatment.
The new finding means that additional, larger studies are now needed, he said. “If patients have sleep apnea, treating the apnea may be an incredibly important way to prevent AF or reduce its burden”
The CAT-HF (Cardiovascular Improvements With Minute Ventilation-Targeted ASV Therapy in Heart Failure) trial was originally designed to randomize 215 heart failure patients with sleep disordered breathing – and who were hospitalized for heart failure – to optimal medical therapy with or without ASV at any of 15 centers in the United States and Germany. But in August 2015, results from the SERVE-HF (Treatment of Sleep-Disordered Breathing with Predominant Central Sleep Apnea by Adaptive Servo Ventilation in Patients with Heart Failure) trial, which generally had a similar design to CAT-HF, showed an unexpected danger from ASV in patients with central sleep apnea and heart failure with reduced ejection fraction (N Engl J Med. 2015 Sept 17;373[12]:1095-105). In SERVE-HF, ASV was associated with significant increases in all-cause and cardiovascular mortality. As a result, enrollment into CAT-HF stopped prematurely with just 126 patients entered, and ASV treatment of patients already enrolled came to a halt.
The primary endpoint in the underpowered and shortened CAT-HF study, survival without cardiovascular hospitalization and with improved functional capacity measured on a 6-minute walk test, showed similar outcomes in both the ASV and control arms. But in a prespecified subgroup analysis by baseline ejection fraction, the 24 patients with heart failure with preserved ejection fraction (19% of the CAT-HF enrollment) showed a statistically significant, 62% relative improvement in the primary endpoint linked with ASV treatment compared with similar patients who did not receive ASV, Christopher M. O’Connor, MD, professor of medicine at Duke University, reported in May 2016 at the European Heart Failure meeting in Florence.
Dr. Piccini’s report focused on a prespecified subgroup analysis of CAT-HF designed to examine the impact of ASV on arrhythmias. Assessment of the impact of ASV on atrial fibrillation was possible in 35 of the 126 patients in CAT-HF who had an implanted cardiac device (pacemaker, defibrillator, or cardiac resynchronization device) with an atrial lead, and assessment of ventricular arrhythmias occurred in 46 of the CAT-HF patients with an implanted high-voltage device (a defibrillator or resynchronization device) that allowed monitoring of ventricular arrhythmias.
For the atrial fibrillation analysis, the 35 patients averaged 60 years of age, and about 90% had a reduced ejection fraction. About two-thirds had an apnea-hypopnea index greater than 30.
The results showed that the 19 patients randomized to receive ASV had an average atrial fibrillation burden of 30% at baseline that dropped to 14% after 6 months of treatment. In contrast, the 16 patients in the control arm had a AF burden of 6% at baseline and 8% after 6 months. The between-group difference for change in AF burden was statistically significant, Dr. Piccini reported, with a burden that decreased by a relative 21% with ASV treatment and increased by a relative 31% in the control arm.
Analysis of the ventricular arrhythmia subgroup showed that ASV had no statistically significant impact for either lowering or raising ventricular tachyarrhythmias or fibrillations.
Trying to reconcile this AF benefit and lack of ventricular arrhythmia harm from ASV in CAT-HF with the excess in cardiovascular deaths seen with ASV in SERVE-HF, Dr. Piccini speculated that some of the SERVE-HF deaths may not have been related to arrhythmia.
“Sudden cardiac death adjudication is profoundly difficult, and does not always equal ventricular arrhythmia,” he said. “We need to consider that some of the adverse events in patients with severe central sleep apnea and low left ventricular ejection fraction [enrolled in SERVE-HF] may have been due to causes other than arrhythmias. The CAT-HF results should motivate investigations of alternative mechanisms of death in SERVE-HF.”
The CAT-HF trial was funded by ResMed, a company that markets adaptive servo ventilation equipment. Dr. Piccini has received research support from ResMed and from Janssen, Gilead, St. Jude, Spectranetics, and he has been a consultant to Janssen, Spectranetics, Medtronic, GSK and BMS-Pfizer. Dr. O’Connor has been a consultant to ResMed and to several other drug and device companies.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
A small prespecified sub-group of patients in the CAT-HF (Cardiovascuar improvements with minute ventilation-targeted ASV therapy in heart failure) trial randomized to adaptive servo ventilation (ASV) showed a 21% relative reduction in atrial fibrillation burden as compared to the control arm which had only 31% relative reduction. While the CAT-HF study was discontinued following results of SERVE-HF trial, this subgroup analysis included 35 patients (19 ASV arm; 16 control arm), the majority of whom had a reduced ejection fraction. This report poses interesting questions about effects of ASV on atrial fibrillation burden in those with reduced EF given the finding that central sleep apnea and Cheyne-Stokes respiration are shown to be associated with incident atrial fibrillation in older men (May et al. Am J Respir Crit Care Med 2016).
A small prespecified sub-group of patients in the CAT-HF (Cardiovascuar improvements with minute ventilation-targeted ASV therapy in heart failure) trial randomized to adaptive servo ventilation (ASV) showed a 21% relative reduction in atrial fibrillation burden as compared to the control arm which had only 31% relative reduction. While the CAT-HF study was discontinued following results of SERVE-HF trial, this subgroup analysis included 35 patients (19 ASV arm; 16 control arm), the majority of whom had a reduced ejection fraction. This report poses interesting questions about effects of ASV on atrial fibrillation burden in those with reduced EF given the finding that central sleep apnea and Cheyne-Stokes respiration are shown to be associated with incident atrial fibrillation in older men (May et al. Am J Respir Crit Care Med 2016).
A small prespecified sub-group of patients in the CAT-HF (Cardiovascuar improvements with minute ventilation-targeted ASV therapy in heart failure) trial randomized to adaptive servo ventilation (ASV) showed a 21% relative reduction in atrial fibrillation burden as compared to the control arm which had only 31% relative reduction. While the CAT-HF study was discontinued following results of SERVE-HF trial, this subgroup analysis included 35 patients (19 ASV arm; 16 control arm), the majority of whom had a reduced ejection fraction. This report poses interesting questions about effects of ASV on atrial fibrillation burden in those with reduced EF given the finding that central sleep apnea and Cheyne-Stokes respiration are shown to be associated with incident atrial fibrillation in older men (May et al. Am J Respir Crit Care Med 2016).
ORLANDO – Adaptive servo ventilation produced a significant and clinically meaningful reduction in atrial fibrillation burden in patients with heart failure and sleep apnea in results from an exploratory, prospective, randomized study with 35 patients.
Adaptive servo ventilation (ASV) “may be an effective antiarrhythmic treatment producing a significant reduction in atrial fibrillation without clear evidence of being proarrhythmogenic,” Jonathan P. Piccini, MD, said at the annual scientific meeting of the Heart Failure Society of America. “Given the potential importance of this finding further studies should validate and quantify the efficacy of ASV for reducing atrial fibrillation in patients with or without heart failure.”
“A mound of data has shown that treating sleep apnea reduced arrhythmias, but until now it’s all been observational and retrospective,” Dr. Piccini, an electrophysiologist at Duke University in Durham, N.C., said in an interview. The study he reported is “the first time” the arrhythmia effects of a sleep apnea intervention, in this case ASV, was studied in a prospective, randomized way while using implanted devices to measure the antiarrhythmic effect of the treatment.
The new finding means that additional, larger studies are now needed, he said. “If patients have sleep apnea, treating the apnea may be an incredibly important way to prevent AF or reduce its burden”
The CAT-HF (Cardiovascular Improvements With Minute Ventilation-Targeted ASV Therapy in Heart Failure) trial was originally designed to randomize 215 heart failure patients with sleep disordered breathing – and who were hospitalized for heart failure – to optimal medical therapy with or without ASV at any of 15 centers in the United States and Germany. But in August 2015, results from the SERVE-HF (Treatment of Sleep-Disordered Breathing with Predominant Central Sleep Apnea by Adaptive Servo Ventilation in Patients with Heart Failure) trial, which generally had a similar design to CAT-HF, showed an unexpected danger from ASV in patients with central sleep apnea and heart failure with reduced ejection fraction (N Engl J Med. 2015 Sept 17;373[12]:1095-105). In SERVE-HF, ASV was associated with significant increases in all-cause and cardiovascular mortality. As a result, enrollment into CAT-HF stopped prematurely with just 126 patients entered, and ASV treatment of patients already enrolled came to a halt.
The primary endpoint in the underpowered and shortened CAT-HF study, survival without cardiovascular hospitalization and with improved functional capacity measured on a 6-minute walk test, showed similar outcomes in both the ASV and control arms. But in a prespecified subgroup analysis by baseline ejection fraction, the 24 patients with heart failure with preserved ejection fraction (19% of the CAT-HF enrollment) showed a statistically significant, 62% relative improvement in the primary endpoint linked with ASV treatment compared with similar patients who did not receive ASV, Christopher M. O’Connor, MD, professor of medicine at Duke University, reported in May 2016 at the European Heart Failure meeting in Florence.
Dr. Piccini’s report focused on a prespecified subgroup analysis of CAT-HF designed to examine the impact of ASV on arrhythmias. Assessment of the impact of ASV on atrial fibrillation was possible in 35 of the 126 patients in CAT-HF who had an implanted cardiac device (pacemaker, defibrillator, or cardiac resynchronization device) with an atrial lead, and assessment of ventricular arrhythmias occurred in 46 of the CAT-HF patients with an implanted high-voltage device (a defibrillator or resynchronization device) that allowed monitoring of ventricular arrhythmias.
For the atrial fibrillation analysis, the 35 patients averaged 60 years of age, and about 90% had a reduced ejection fraction. About two-thirds had an apnea-hypopnea index greater than 30.
The results showed that the 19 patients randomized to receive ASV had an average atrial fibrillation burden of 30% at baseline that dropped to 14% after 6 months of treatment. In contrast, the 16 patients in the control arm had a AF burden of 6% at baseline and 8% after 6 months. The between-group difference for change in AF burden was statistically significant, Dr. Piccini reported, with a burden that decreased by a relative 21% with ASV treatment and increased by a relative 31% in the control arm.
Analysis of the ventricular arrhythmia subgroup showed that ASV had no statistically significant impact for either lowering or raising ventricular tachyarrhythmias or fibrillations.
Trying to reconcile this AF benefit and lack of ventricular arrhythmia harm from ASV in CAT-HF with the excess in cardiovascular deaths seen with ASV in SERVE-HF, Dr. Piccini speculated that some of the SERVE-HF deaths may not have been related to arrhythmia.
“Sudden cardiac death adjudication is profoundly difficult, and does not always equal ventricular arrhythmia,” he said. “We need to consider that some of the adverse events in patients with severe central sleep apnea and low left ventricular ejection fraction [enrolled in SERVE-HF] may have been due to causes other than arrhythmias. The CAT-HF results should motivate investigations of alternative mechanisms of death in SERVE-HF.”
The CAT-HF trial was funded by ResMed, a company that markets adaptive servo ventilation equipment. Dr. Piccini has received research support from ResMed and from Janssen, Gilead, St. Jude, Spectranetics, and he has been a consultant to Janssen, Spectranetics, Medtronic, GSK and BMS-Pfizer. Dr. O’Connor has been a consultant to ResMed and to several other drug and device companies.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ORLANDO – Adaptive servo ventilation produced a significant and clinically meaningful reduction in atrial fibrillation burden in patients with heart failure and sleep apnea in results from an exploratory, prospective, randomized study with 35 patients.
Adaptive servo ventilation (ASV) “may be an effective antiarrhythmic treatment producing a significant reduction in atrial fibrillation without clear evidence of being proarrhythmogenic,” Jonathan P. Piccini, MD, said at the annual scientific meeting of the Heart Failure Society of America. “Given the potential importance of this finding further studies should validate and quantify the efficacy of ASV for reducing atrial fibrillation in patients with or without heart failure.”
“A mound of data has shown that treating sleep apnea reduced arrhythmias, but until now it’s all been observational and retrospective,” Dr. Piccini, an electrophysiologist at Duke University in Durham, N.C., said in an interview. The study he reported is “the first time” the arrhythmia effects of a sleep apnea intervention, in this case ASV, was studied in a prospective, randomized way while using implanted devices to measure the antiarrhythmic effect of the treatment.
The new finding means that additional, larger studies are now needed, he said. “If patients have sleep apnea, treating the apnea may be an incredibly important way to prevent AF or reduce its burden”
The CAT-HF (Cardiovascular Improvements With Minute Ventilation-Targeted ASV Therapy in Heart Failure) trial was originally designed to randomize 215 heart failure patients with sleep disordered breathing – and who were hospitalized for heart failure – to optimal medical therapy with or without ASV at any of 15 centers in the United States and Germany. But in August 2015, results from the SERVE-HF (Treatment of Sleep-Disordered Breathing with Predominant Central Sleep Apnea by Adaptive Servo Ventilation in Patients with Heart Failure) trial, which generally had a similar design to CAT-HF, showed an unexpected danger from ASV in patients with central sleep apnea and heart failure with reduced ejection fraction (N Engl J Med. 2015 Sept 17;373[12]:1095-105). In SERVE-HF, ASV was associated with significant increases in all-cause and cardiovascular mortality. As a result, enrollment into CAT-HF stopped prematurely with just 126 patients entered, and ASV treatment of patients already enrolled came to a halt.
The primary endpoint in the underpowered and shortened CAT-HF study, survival without cardiovascular hospitalization and with improved functional capacity measured on a 6-minute walk test, showed similar outcomes in both the ASV and control arms. But in a prespecified subgroup analysis by baseline ejection fraction, the 24 patients with heart failure with preserved ejection fraction (19% of the CAT-HF enrollment) showed a statistically significant, 62% relative improvement in the primary endpoint linked with ASV treatment compared with similar patients who did not receive ASV, Christopher M. O’Connor, MD, professor of medicine at Duke University, reported in May 2016 at the European Heart Failure meeting in Florence.
Dr. Piccini’s report focused on a prespecified subgroup analysis of CAT-HF designed to examine the impact of ASV on arrhythmias. Assessment of the impact of ASV on atrial fibrillation was possible in 35 of the 126 patients in CAT-HF who had an implanted cardiac device (pacemaker, defibrillator, or cardiac resynchronization device) with an atrial lead, and assessment of ventricular arrhythmias occurred in 46 of the CAT-HF patients with an implanted high-voltage device (a defibrillator or resynchronization device) that allowed monitoring of ventricular arrhythmias.
For the atrial fibrillation analysis, the 35 patients averaged 60 years of age, and about 90% had a reduced ejection fraction. About two-thirds had an apnea-hypopnea index greater than 30.
The results showed that the 19 patients randomized to receive ASV had an average atrial fibrillation burden of 30% at baseline that dropped to 14% after 6 months of treatment. In contrast, the 16 patients in the control arm had a AF burden of 6% at baseline and 8% after 6 months. The between-group difference for change in AF burden was statistically significant, Dr. Piccini reported, with a burden that decreased by a relative 21% with ASV treatment and increased by a relative 31% in the control arm.
Analysis of the ventricular arrhythmia subgroup showed that ASV had no statistically significant impact for either lowering or raising ventricular tachyarrhythmias or fibrillations.
Trying to reconcile this AF benefit and lack of ventricular arrhythmia harm from ASV in CAT-HF with the excess in cardiovascular deaths seen with ASV in SERVE-HF, Dr. Piccini speculated that some of the SERVE-HF deaths may not have been related to arrhythmia.
“Sudden cardiac death adjudication is profoundly difficult, and does not always equal ventricular arrhythmia,” he said. “We need to consider that some of the adverse events in patients with severe central sleep apnea and low left ventricular ejection fraction [enrolled in SERVE-HF] may have been due to causes other than arrhythmias. The CAT-HF results should motivate investigations of alternative mechanisms of death in SERVE-HF.”
The CAT-HF trial was funded by ResMed, a company that markets adaptive servo ventilation equipment. Dr. Piccini has received research support from ResMed and from Janssen, Gilead, St. Jude, Spectranetics, and he has been a consultant to Janssen, Spectranetics, Medtronic, GSK and BMS-Pfizer. Dr. O’Connor has been a consultant to ResMed and to several other drug and device companies.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Key clinical point:
Major finding: After 6 months, ASV produced a relative 21% drop in atrial fibrillation burden, compared with increased burden in control patients.
Data source: CAT-HF, a multicenter randomized trial that enrolled 126 heart failure patients with sleep apnea.
Disclosures: The CAT-HF trial was funded by ResMed, a company that markets adaptive servo ventilation equipment. Dr. Piccini has received research support and/or consultant fees from ResMed, Janssen, Gilead, St. Jude, Spectranetics, Medtronic, GSK and BMS-Pfizer.
Advanced heart failure symptoms linked to mortality
ORLANDO – Advanced heart failure patients who are hospitalized for heart failure and have a higher symptom burden at discharge have a significantly increased rate of death or rehospitalization over the next 6 months, based on an analysis of 393 patients enrolled in a heart failure trial.
The strong link between severe symptom burden and poor near-term outcomes persisted despite adjustment for various markers of heart failure severity, suggesting that treatment aimed at reducing symptoms may be able to reduce mortality or heart failure hospitalization in advanced heart failure patients, Ellen K. Hummel, MD, said at the annual scientific meeting of the Heart Failure Society of America.
In her analysis, a severe symptom burden at the time of hospital discharge linked with an adjusted 2.9-fold increased mortality rate and a 2.5-fold increased rate of days dead or hospitalized during the next 6 months, said Dr. Hummel, a geriatric and palliative care specialist at the University of Michigan in Ann Arbor. These elevated rate ratios for patients with severe symptoms at hospital discharge were in comparison to the ratios for advanced heart failure patients in the study with no symptoms at discharge.
Three symptoms contributed to the symptom score she used in her analysis: fatigue, scored on a scale of 0-3; dyspnea, also scored 0-3; and gastrointestinal distress, scored as 0-2, creating a maximum score of 8. Her analysis categorized mild as a total score of 1-4 and severe as 5 or greater. In the study population she used for her analysis, patients enrolled in the multicenter ESCAPE (Evaluation Study of Congestive Heart Failure and Pulmonary Artery Catheterization Effectiveness) trial, 111 of the 393 evaluable patients (28%) had none of these symptoms, 239 (61%) had mild symptoms, and 43 (11%) had severe symptoms. Scoring was done by patients based on their subjective self-assessment at the time of hospital discharge.
The absolute, observed 6-month mortality rates were roughly 45% among patients with severe symptoms, about 17% in patients with mild symptoms, and about 12% in those with no symptoms.
The primary purpose of ESCAPE was to assess the impact that routine collection of data from a pulmonary artery catheter during hospitalization has on outcomes; the results showed no significant link between improved outcomes and getting these data (JAMA. 2005 Oct 5;294[13]:1625-33). The study ran during 2000-2003 at 26 centers in the United States and Canada. Of the 433 advanced heart failure patients enrolled in ESCAPE, 393 had complete records to allow the current analysis.
The adjustments that Dr. Hummel made in the proportional hazard analysis took into account New York Heart Association class, and severity of disease at the time of hospital discharge measured by the ESCAPE Discharge Risk Score. This score takes into account age, 6-minute walk distance, blood urea nitrogen, brain natriuretic peptide levels, blood pressure, selected drug treatments, sodium level, and history of cardiopulmonary resuscitation or mechanical ventilation.
Dr. Hummel had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ORLANDO – Advanced heart failure patients who are hospitalized for heart failure and have a higher symptom burden at discharge have a significantly increased rate of death or rehospitalization over the next 6 months, based on an analysis of 393 patients enrolled in a heart failure trial.
The strong link between severe symptom burden and poor near-term outcomes persisted despite adjustment for various markers of heart failure severity, suggesting that treatment aimed at reducing symptoms may be able to reduce mortality or heart failure hospitalization in advanced heart failure patients, Ellen K. Hummel, MD, said at the annual scientific meeting of the Heart Failure Society of America.
In her analysis, a severe symptom burden at the time of hospital discharge linked with an adjusted 2.9-fold increased mortality rate and a 2.5-fold increased rate of days dead or hospitalized during the next 6 months, said Dr. Hummel, a geriatric and palliative care specialist at the University of Michigan in Ann Arbor. These elevated rate ratios for patients with severe symptoms at hospital discharge were in comparison to the ratios for advanced heart failure patients in the study with no symptoms at discharge.
Three symptoms contributed to the symptom score she used in her analysis: fatigue, scored on a scale of 0-3; dyspnea, also scored 0-3; and gastrointestinal distress, scored as 0-2, creating a maximum score of 8. Her analysis categorized mild as a total score of 1-4 and severe as 5 or greater. In the study population she used for her analysis, patients enrolled in the multicenter ESCAPE (Evaluation Study of Congestive Heart Failure and Pulmonary Artery Catheterization Effectiveness) trial, 111 of the 393 evaluable patients (28%) had none of these symptoms, 239 (61%) had mild symptoms, and 43 (11%) had severe symptoms. Scoring was done by patients based on their subjective self-assessment at the time of hospital discharge.
The absolute, observed 6-month mortality rates were roughly 45% among patients with severe symptoms, about 17% in patients with mild symptoms, and about 12% in those with no symptoms.
The primary purpose of ESCAPE was to assess the impact that routine collection of data from a pulmonary artery catheter during hospitalization has on outcomes; the results showed no significant link between improved outcomes and getting these data (JAMA. 2005 Oct 5;294[13]:1625-33). The study ran during 2000-2003 at 26 centers in the United States and Canada. Of the 433 advanced heart failure patients enrolled in ESCAPE, 393 had complete records to allow the current analysis.
The adjustments that Dr. Hummel made in the proportional hazard analysis took into account New York Heart Association class, and severity of disease at the time of hospital discharge measured by the ESCAPE Discharge Risk Score. This score takes into account age, 6-minute walk distance, blood urea nitrogen, brain natriuretic peptide levels, blood pressure, selected drug treatments, sodium level, and history of cardiopulmonary resuscitation or mechanical ventilation.
Dr. Hummel had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ORLANDO – Advanced heart failure patients who are hospitalized for heart failure and have a higher symptom burden at discharge have a significantly increased rate of death or rehospitalization over the next 6 months, based on an analysis of 393 patients enrolled in a heart failure trial.
The strong link between severe symptom burden and poor near-term outcomes persisted despite adjustment for various markers of heart failure severity, suggesting that treatment aimed at reducing symptoms may be able to reduce mortality or heart failure hospitalization in advanced heart failure patients, Ellen K. Hummel, MD, said at the annual scientific meeting of the Heart Failure Society of America.
In her analysis, a severe symptom burden at the time of hospital discharge linked with an adjusted 2.9-fold increased mortality rate and a 2.5-fold increased rate of days dead or hospitalized during the next 6 months, said Dr. Hummel, a geriatric and palliative care specialist at the University of Michigan in Ann Arbor. These elevated rate ratios for patients with severe symptoms at hospital discharge were in comparison to the ratios for advanced heart failure patients in the study with no symptoms at discharge.
Three symptoms contributed to the symptom score she used in her analysis: fatigue, scored on a scale of 0-3; dyspnea, also scored 0-3; and gastrointestinal distress, scored as 0-2, creating a maximum score of 8. Her analysis categorized mild as a total score of 1-4 and severe as 5 or greater. In the study population she used for her analysis, patients enrolled in the multicenter ESCAPE (Evaluation Study of Congestive Heart Failure and Pulmonary Artery Catheterization Effectiveness) trial, 111 of the 393 evaluable patients (28%) had none of these symptoms, 239 (61%) had mild symptoms, and 43 (11%) had severe symptoms. Scoring was done by patients based on their subjective self-assessment at the time of hospital discharge.
The absolute, observed 6-month mortality rates were roughly 45% among patients with severe symptoms, about 17% in patients with mild symptoms, and about 12% in those with no symptoms.
The primary purpose of ESCAPE was to assess the impact that routine collection of data from a pulmonary artery catheter during hospitalization has on outcomes; the results showed no significant link between improved outcomes and getting these data (JAMA. 2005 Oct 5;294[13]:1625-33). The study ran during 2000-2003 at 26 centers in the United States and Canada. Of the 433 advanced heart failure patients enrolled in ESCAPE, 393 had complete records to allow the current analysis.
The adjustments that Dr. Hummel made in the proportional hazard analysis took into account New York Heart Association class, and severity of disease at the time of hospital discharge measured by the ESCAPE Discharge Risk Score. This score takes into account age, 6-minute walk distance, blood urea nitrogen, brain natriuretic peptide levels, blood pressure, selected drug treatments, sodium level, and history of cardiopulmonary resuscitation or mechanical ventilation.
Dr. Hummel had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT THE HFSA ANNUAL SCIENTIFIC MEETING
Key clinical point:
Major finding: Patients with severe symptoms at discharge had a 2.9-fold increased rate of death, compared with those with no symptoms.
Data source: A post hoc analysis of data collected from 393 patients enrolled in the ESCAPE trial.
Disclosures: Dr. Hummel had no relevant financial disclosures.
TOPCAT, a third time around
Shakespeare, in Romeo and Juliet, refers to the proverb, “A cat has nine lives. For three he plays, for three he strays, and for the last he stays.”
TOPCAT is back again, having randomized its first patient with heart failure with preserved ejection fraction (HFpEF) almost 10 years ago for its treatment with spironolactone (SPIRO), a mineralocorticoid receptor antagonist.
The first report of the results of TOPCAT in 2014 indicated that there was no benefit associate with SPIRO therapy tested in the 3,445 patients randomized in 244 sites around the world (N Engl J Med. 2014 Apr 10;370[15]:1383-92). A subsequent analysis of data carried out in 2015 reported a striking regional difference in the outcome of patients randomized in the 1,767 patients in the Americas, compared with the 1,678 randomized in Russia and Georgia (Circulation. 2015 Jan 6;131[1]:34-42). In the Americas, there was an 18% decrease in the primary event of death and heart failure rehospitalization (3.6% in the SPIRO vs. 4.9% in the placebo; hazard ratio, 0.82; P = .026). There was essentially no difference in the groups randomized in Russia and Georgia, which had a 1.6% placebo event rate.
And now in 2016, at the recent meeting of the Heart Failure Society of America, we were informed that there was no detectable level of blood canrenone, a metabolite of SPIRO, in 30% of the 66 randomized patients in Russia and Georgia, compared with 3% of the patients randomized in the Americas (Cardiology News. Oct 2016. p 8). These data tend to confirm that the patients randomized in Russia and Georgia were either undertreated or not treated. In fact, after examination of the baseline characteristics of the two groups it is possible that many of the patients may not have had heart failure at all.
So what are we left with? One thing that is clear is that the management of TOPCAT was flawed and constitutes an example of how not to run an international clinical trial. Can we make any conclusion about the benefit of SPIRO? TOPCAT initially was powered for over 3,515 patients and 630 events in order to achieve a 85% benefit. The current analysis has now narrowed the population down to 1,787 patients with 522 events with an 18% decrease (P = .02) in the primary end point. During the mean follow-up of 3.3 years there was a placebo mortality of 4.9%, which is impressive in the setting of concomitant beta-blocker and renin angiotensin-converting enzyme inhibitor therapy. The only significant adverse observation was a threefold occurrence in hyperkalemia (potassium greater than 5.5 mmols/L) in the 25.2% in the Americas group treated with SPIRO, compared with the Russian-Georgian patients
Unfortunately the answer is not entirely clear. We all know who HFpEF patients are when they walk into the clinic but identifying them for a clinical trial has been difficult if not impossible. As for me, I will choose to treat their hypertension aggressively (not an easy task) and prevent or suppress their arrhythmias. In that project I will use beta-blockers and SPIRO to prevent their next heart failure episode and hope for the best.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
Shakespeare, in Romeo and Juliet, refers to the proverb, “A cat has nine lives. For three he plays, for three he strays, and for the last he stays.”
TOPCAT is back again, having randomized its first patient with heart failure with preserved ejection fraction (HFpEF) almost 10 years ago for its treatment with spironolactone (SPIRO), a mineralocorticoid receptor antagonist.
The first report of the results of TOPCAT in 2014 indicated that there was no benefit associate with SPIRO therapy tested in the 3,445 patients randomized in 244 sites around the world (N Engl J Med. 2014 Apr 10;370[15]:1383-92). A subsequent analysis of data carried out in 2015 reported a striking regional difference in the outcome of patients randomized in the 1,767 patients in the Americas, compared with the 1,678 randomized in Russia and Georgia (Circulation. 2015 Jan 6;131[1]:34-42). In the Americas, there was an 18% decrease in the primary event of death and heart failure rehospitalization (3.6% in the SPIRO vs. 4.9% in the placebo; hazard ratio, 0.82; P = .026). There was essentially no difference in the groups randomized in Russia and Georgia, which had a 1.6% placebo event rate.
And now in 2016, at the recent meeting of the Heart Failure Society of America, we were informed that there was no detectable level of blood canrenone, a metabolite of SPIRO, in 30% of the 66 randomized patients in Russia and Georgia, compared with 3% of the patients randomized in the Americas (Cardiology News. Oct 2016. p 8). These data tend to confirm that the patients randomized in Russia and Georgia were either undertreated or not treated. In fact, after examination of the baseline characteristics of the two groups it is possible that many of the patients may not have had heart failure at all.
So what are we left with? One thing that is clear is that the management of TOPCAT was flawed and constitutes an example of how not to run an international clinical trial. Can we make any conclusion about the benefit of SPIRO? TOPCAT initially was powered for over 3,515 patients and 630 events in order to achieve a 85% benefit. The current analysis has now narrowed the population down to 1,787 patients with 522 events with an 18% decrease (P = .02) in the primary end point. During the mean follow-up of 3.3 years there was a placebo mortality of 4.9%, which is impressive in the setting of concomitant beta-blocker and renin angiotensin-converting enzyme inhibitor therapy. The only significant adverse observation was a threefold occurrence in hyperkalemia (potassium greater than 5.5 mmols/L) in the 25.2% in the Americas group treated with SPIRO, compared with the Russian-Georgian patients
Unfortunately the answer is not entirely clear. We all know who HFpEF patients are when they walk into the clinic but identifying them for a clinical trial has been difficult if not impossible. As for me, I will choose to treat their hypertension aggressively (not an easy task) and prevent or suppress their arrhythmias. In that project I will use beta-blockers and SPIRO to prevent their next heart failure episode and hope for the best.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
Shakespeare, in Romeo and Juliet, refers to the proverb, “A cat has nine lives. For three he plays, for three he strays, and for the last he stays.”
TOPCAT is back again, having randomized its first patient with heart failure with preserved ejection fraction (HFpEF) almost 10 years ago for its treatment with spironolactone (SPIRO), a mineralocorticoid receptor antagonist.
The first report of the results of TOPCAT in 2014 indicated that there was no benefit associate with SPIRO therapy tested in the 3,445 patients randomized in 244 sites around the world (N Engl J Med. 2014 Apr 10;370[15]:1383-92). A subsequent analysis of data carried out in 2015 reported a striking regional difference in the outcome of patients randomized in the 1,767 patients in the Americas, compared with the 1,678 randomized in Russia and Georgia (Circulation. 2015 Jan 6;131[1]:34-42). In the Americas, there was an 18% decrease in the primary event of death and heart failure rehospitalization (3.6% in the SPIRO vs. 4.9% in the placebo; hazard ratio, 0.82; P = .026). There was essentially no difference in the groups randomized in Russia and Georgia, which had a 1.6% placebo event rate.
And now in 2016, at the recent meeting of the Heart Failure Society of America, we were informed that there was no detectable level of blood canrenone, a metabolite of SPIRO, in 30% of the 66 randomized patients in Russia and Georgia, compared with 3% of the patients randomized in the Americas (Cardiology News. Oct 2016. p 8). These data tend to confirm that the patients randomized in Russia and Georgia were either undertreated or not treated. In fact, after examination of the baseline characteristics of the two groups it is possible that many of the patients may not have had heart failure at all.
So what are we left with? One thing that is clear is that the management of TOPCAT was flawed and constitutes an example of how not to run an international clinical trial. Can we make any conclusion about the benefit of SPIRO? TOPCAT initially was powered for over 3,515 patients and 630 events in order to achieve a 85% benefit. The current analysis has now narrowed the population down to 1,787 patients with 522 events with an 18% decrease (P = .02) in the primary end point. During the mean follow-up of 3.3 years there was a placebo mortality of 4.9%, which is impressive in the setting of concomitant beta-blocker and renin angiotensin-converting enzyme inhibitor therapy. The only significant adverse observation was a threefold occurrence in hyperkalemia (potassium greater than 5.5 mmols/L) in the 25.2% in the Americas group treated with SPIRO, compared with the Russian-Georgian patients
Unfortunately the answer is not entirely clear. We all know who HFpEF patients are when they walk into the clinic but identifying them for a clinical trial has been difficult if not impossible. As for me, I will choose to treat their hypertension aggressively (not an easy task) and prevent or suppress their arrhythmias. In that project I will use beta-blockers and SPIRO to prevent their next heart failure episode and hope for the best.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
Beta-blockers curb death risk in patients with primary prevention ICD
ROME – Beta-blocker therapy reduces the risks of all-cause mortality as well as cardiac death in patients with a left ventricular ejection fraction below 35% who get an implantable cardioverter-defibrillator for primary prevention, Laurent Fauchier, MD, PhD, reported at the annual congress of the European Society of Cardiology.
Some physicians have recently urged reconsideration of current guidelines recommending routine use of beta-blockers for prevention of cardiovascular events in certain groups of patients with coronary artery disease, including those with chronic heart failure who have received an ICD for primary prevention of sudden death. And indeed it’s true that the now–relatively old randomized trials of ICDs for primary prevention in patients with chronic heart failure don’t provide any real evidence that beta-blockers reduce mortality in this setting. In fact, the guideline recommendation for beta-blockade has been based upon expert opinion. This was the impetus for Dr. Fauchier and coinvestigators to conduct a large retrospective observational study in a contemporary cohort of heart failure patients who received an ICD for primary prevention during a recent 10-year period at the 12 largest centers in France.
Fifteen percent of the 3,975 French ICD recipients did not receive a beta-blocker. They differed from those who did in that they were on average 2 years older, had an absolute 5% lower ejection fraction, and were more likely to also receive cardiac resynchronization therapy. Propensity score matching based on these and 19 other baseline characteristics enabled investigators to assemble a cohort of 541 closely matched patient pairs, explained Dr. Fauchier, professor of cardiology at Francois Rabelais University in Tours, France.
During a mean follow-up of 3.2 years, the risk of all-cause mortality in ICD recipients not on a beta-blocker was 34% higher than in those who were. Moreover, their risk of cardiac death was 50% greater.
In contrast, beta-blocker therapy had no effect on the risks of sudden death or of appropriate or inappropriate shocks.
The finding that beta-blocker therapy doesn’t prevent sudden death in patients with an ICD for primary prevention has not previously been reported. However, it makes sense. The device prevents such events so effectively that a beta-blocker adds nothing further in that regard, according to Dr. Fauchier.
“Beta-blockers should continue to be used widely, as currently recommended, for heart failure in the specific setting of patients with prophylactic ICD implantation. You do not have the benefit for prevention of sudden death, but you still have all the benefit from preventing cardiac death,” the electrophysiologist concluded.
This study was supported by French governmental research grants. Dr. Fauchier reported serving as a consultant to Bayer, Pfizer, Boehringer Ingelheim, Medtronic, and Novartis.
ROME – Beta-blocker therapy reduces the risks of all-cause mortality as well as cardiac death in patients with a left ventricular ejection fraction below 35% who get an implantable cardioverter-defibrillator for primary prevention, Laurent Fauchier, MD, PhD, reported at the annual congress of the European Society of Cardiology.
Some physicians have recently urged reconsideration of current guidelines recommending routine use of beta-blockers for prevention of cardiovascular events in certain groups of patients with coronary artery disease, including those with chronic heart failure who have received an ICD for primary prevention of sudden death. And indeed it’s true that the now–relatively old randomized trials of ICDs for primary prevention in patients with chronic heart failure don’t provide any real evidence that beta-blockers reduce mortality in this setting. In fact, the guideline recommendation for beta-blockade has been based upon expert opinion. This was the impetus for Dr. Fauchier and coinvestigators to conduct a large retrospective observational study in a contemporary cohort of heart failure patients who received an ICD for primary prevention during a recent 10-year period at the 12 largest centers in France.
Fifteen percent of the 3,975 French ICD recipients did not receive a beta-blocker. They differed from those who did in that they were on average 2 years older, had an absolute 5% lower ejection fraction, and were more likely to also receive cardiac resynchronization therapy. Propensity score matching based on these and 19 other baseline characteristics enabled investigators to assemble a cohort of 541 closely matched patient pairs, explained Dr. Fauchier, professor of cardiology at Francois Rabelais University in Tours, France.
During a mean follow-up of 3.2 years, the risk of all-cause mortality in ICD recipients not on a beta-blocker was 34% higher than in those who were. Moreover, their risk of cardiac death was 50% greater.
In contrast, beta-blocker therapy had no effect on the risks of sudden death or of appropriate or inappropriate shocks.
The finding that beta-blocker therapy doesn’t prevent sudden death in patients with an ICD for primary prevention has not previously been reported. However, it makes sense. The device prevents such events so effectively that a beta-blocker adds nothing further in that regard, according to Dr. Fauchier.
“Beta-blockers should continue to be used widely, as currently recommended, for heart failure in the specific setting of patients with prophylactic ICD implantation. You do not have the benefit for prevention of sudden death, but you still have all the benefit from preventing cardiac death,” the electrophysiologist concluded.
This study was supported by French governmental research grants. Dr. Fauchier reported serving as a consultant to Bayer, Pfizer, Boehringer Ingelheim, Medtronic, and Novartis.
ROME – Beta-blocker therapy reduces the risks of all-cause mortality as well as cardiac death in patients with a left ventricular ejection fraction below 35% who get an implantable cardioverter-defibrillator for primary prevention, Laurent Fauchier, MD, PhD, reported at the annual congress of the European Society of Cardiology.
Some physicians have recently urged reconsideration of current guidelines recommending routine use of beta-blockers for prevention of cardiovascular events in certain groups of patients with coronary artery disease, including those with chronic heart failure who have received an ICD for primary prevention of sudden death. And indeed it’s true that the now–relatively old randomized trials of ICDs for primary prevention in patients with chronic heart failure don’t provide any real evidence that beta-blockers reduce mortality in this setting. In fact, the guideline recommendation for beta-blockade has been based upon expert opinion. This was the impetus for Dr. Fauchier and coinvestigators to conduct a large retrospective observational study in a contemporary cohort of heart failure patients who received an ICD for primary prevention during a recent 10-year period at the 12 largest centers in France.
Fifteen percent of the 3,975 French ICD recipients did not receive a beta-blocker. They differed from those who did in that they were on average 2 years older, had an absolute 5% lower ejection fraction, and were more likely to also receive cardiac resynchronization therapy. Propensity score matching based on these and 19 other baseline characteristics enabled investigators to assemble a cohort of 541 closely matched patient pairs, explained Dr. Fauchier, professor of cardiology at Francois Rabelais University in Tours, France.
During a mean follow-up of 3.2 years, the risk of all-cause mortality in ICD recipients not on a beta-blocker was 34% higher than in those who were. Moreover, their risk of cardiac death was 50% greater.
In contrast, beta-blocker therapy had no effect on the risks of sudden death or of appropriate or inappropriate shocks.
The finding that beta-blocker therapy doesn’t prevent sudden death in patients with an ICD for primary prevention has not previously been reported. However, it makes sense. The device prevents such events so effectively that a beta-blocker adds nothing further in that regard, according to Dr. Fauchier.
“Beta-blockers should continue to be used widely, as currently recommended, for heart failure in the specific setting of patients with prophylactic ICD implantation. You do not have the benefit for prevention of sudden death, but you still have all the benefit from preventing cardiac death,” the electrophysiologist concluded.
This study was supported by French governmental research grants. Dr. Fauchier reported serving as a consultant to Bayer, Pfizer, Boehringer Ingelheim, Medtronic, and Novartis.
AT THE ESC CONGRESS 2016
Key clinical point:
Major finding: Patients with heart failure with reduced ejection fraction who received an ICD for primary prevention and were not on a beta-blocker were at an adjusted 50% increased risk for cardiac death and 34% increased risk for all-cause mortality during 3.2 years of follow-up, but they were at no increased risk for sudden death.
Data source: A retrospective observational study of all of the nearly 4,000 patients who received a primary prevention ICD at the 12 largest French centers during a recent 10-year period.
Disclosures: This study was supported by French governmental research funds. The presenter reported serving as a consultant to Bayer, Pfizer, Boehringer Ingelheim, Medtronic, and Novartis.
Palliative care boosts heart failure patient outcomes
ORLANDO – Systematic introduction of palliative care interventions for patients with advanced heart failure improved patients’ quality of life and spurred their development of advanced-care preferences in a pair of independently performed, controlled, pilot studies.
But, despite demonstrating the ability of palliative-care interventions to help heart failure patients during their final months of life, the findings raised questions about the generalizability and reproducibility of palliative-care interventions that may depend on the skills and experience of the individual specialists who deliver the palliative care.
“Palliative care for patients with cardiovascular disease is in desperate need of good-quality evidence,” commented Larry A, Allen, MD, a heart failure cardiologist at the University of Colorado in Aurora and designated discussant for one of the two studies presented at the meeting. “We need large, randomized trials with clinical outcomes to look at patient outcomes from palliative-care interventions.”
The patients average 71 years old, about half were women, and about 40% were African Americans. They had been diagnosed with heart failure for an average of more than 5 years, all had advanced heart failure, about 60% spent at least half of their time awake immobilized in a bed or chair, and they had average NT-proBNP blood levels of greater than 10,000 pg/mL.
After 24 weeks of intervention, the palliative-care program produced both statistically significant and clinically meaningful improvements in two different measures of health-related quality of life, the Kansas City Cardiomyopathy Questionnaire (KCCQ) and the Functional Assessment of Chronic Illness Therapy – Palliative Care (FACIT-PAL). The KCCQ showed the palliative care intervention linked with an average rise of more than 9 points compared with patients in the control arm after adjustment for age and sex, a statistically significant increase on a scale where a 5-point rise is considered clinically meaningful. The FACIT-PAL showed an average, adjusted 11-point rise linked with the intervention, a statistically significant increase on a scale where an increase of at least 10 is judged clinically meaningful, reported Dr. Rogers, a heart failure cardiologist and professor of medicine at Duke University.
The palliative-care intervention also led to significant improvements in measures of spirituality, depression, and anxiety, but intervention had no impact on mortality.
“I like these endpoints and the idea that we can make quality-of-life better. These are very sick patients, with a predicted 6-month mortality of 50%. Patients reach a time when they don’t want to live longer but want better life quality for the days they still have,” he said in an interview.
The second report came from a single-center pilot study of 50 patients enrolled when they were hospitalized for acute decompensated heart failure and had at least one addition risk factor for poor prognosis such as age of at least 81 years, renal dysfunction, or a prior heart failure hospitalization within the past year. Patients randomized to the intervention arm underwent a structured evaluation based on the Serious Illness Conversation Guide and performed by a social worker experienced in palliative care and embedded in the heart failure clinical team. The primary endpoint of the SWAP-HF (Social Worker–Aided Palliative Care Intervention in High Risk Patients with Heart Failure) study was clinical-level documentation of advanced-care preferences by 6 months after the program began.
“Although more comprehensive, multidisciplinary palliative care interventions may also be effective, the focused approach [used in this study] may represent a cost-effective and scalable method for shepherding limited specialty resources to enhance the delivery of patient-centered care,” Dr. Desai said. In other words, a program with a social worker costs less than a two-person staff with a palliative-care physician and nurse practitioner.
Despite its relative simplicity, the SWAP-HF intervention had some unique aspects that make it generalizability uncertain, commented Dr. Allen. The embedding of a social worker on the heart failure team placed a professional with a “good understanding of social context” right on the scene with everyone else delivering care to the heart failure patient, a good strategy for minimizing fragmentation, he said. In addition, the place where the study was done, Brigham and Women’s Hospital, “is not your average hospital,” he noted,
In addition, the timing of the intervention studied during hospitalization may be problematic. Clinicians need to “be careful about patients making long-term decisions” about their care while they are hospitalized, a time when patients can be “ill, confused, and scared.” He cited recent findings from a study of hospital-based palliative-care interventions for family members of patients with chronic critical illness that did not reduce anxiety or depression symptoms among the treated family members and may have increased symptoms of posttraumatic stress disorder (JAMA. 2016 July 5;374[1]:51-62).
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
It’s very exciting to have these two studies presented at the Heart Failure Society of America’s annual meeting. Palliative-care research now receives funding from the National Institutes of Health, but consistently and successfully integrating palliative care into heart failure management still has a long way to go. In 2004, my colleagues and I published a set of consensus recommendations on how to apply palliative care methods to patients with advanced heart failure and what research needs existed for the field (J Card Fail. 2004 June;10[3]:200-9). Today, 12 years later, many of those research needs remain inadequately addressed.
Sarah J. Goodlin, MD , is chief of geriatrics at the Portland (Ore.) VA Medical Center. She had no disclosures. She made these comments as the designated discussant for Dr. Rogers’ report.
It’s very exciting to have these two studies presented at the Heart Failure Society of America’s annual meeting. Palliative-care research now receives funding from the National Institutes of Health, but consistently and successfully integrating palliative care into heart failure management still has a long way to go. In 2004, my colleagues and I published a set of consensus recommendations on how to apply palliative care methods to patients with advanced heart failure and what research needs existed for the field (J Card Fail. 2004 June;10[3]:200-9). Today, 12 years later, many of those research needs remain inadequately addressed.
Sarah J. Goodlin, MD , is chief of geriatrics at the Portland (Ore.) VA Medical Center. She had no disclosures. She made these comments as the designated discussant for Dr. Rogers’ report.
It’s very exciting to have these two studies presented at the Heart Failure Society of America’s annual meeting. Palliative-care research now receives funding from the National Institutes of Health, but consistently and successfully integrating palliative care into heart failure management still has a long way to go. In 2004, my colleagues and I published a set of consensus recommendations on how to apply palliative care methods to patients with advanced heart failure and what research needs existed for the field (J Card Fail. 2004 June;10[3]:200-9). Today, 12 years later, many of those research needs remain inadequately addressed.
Sarah J. Goodlin, MD , is chief of geriatrics at the Portland (Ore.) VA Medical Center. She had no disclosures. She made these comments as the designated discussant for Dr. Rogers’ report.
ORLANDO – Systematic introduction of palliative care interventions for patients with advanced heart failure improved patients’ quality of life and spurred their development of advanced-care preferences in a pair of independently performed, controlled, pilot studies.
But, despite demonstrating the ability of palliative-care interventions to help heart failure patients during their final months of life, the findings raised questions about the generalizability and reproducibility of palliative-care interventions that may depend on the skills and experience of the individual specialists who deliver the palliative care.
“Palliative care for patients with cardiovascular disease is in desperate need of good-quality evidence,” commented Larry A, Allen, MD, a heart failure cardiologist at the University of Colorado in Aurora and designated discussant for one of the two studies presented at the meeting. “We need large, randomized trials with clinical outcomes to look at patient outcomes from palliative-care interventions.”
The patients average 71 years old, about half were women, and about 40% were African Americans. They had been diagnosed with heart failure for an average of more than 5 years, all had advanced heart failure, about 60% spent at least half of their time awake immobilized in a bed or chair, and they had average NT-proBNP blood levels of greater than 10,000 pg/mL.
After 24 weeks of intervention, the palliative-care program produced both statistically significant and clinically meaningful improvements in two different measures of health-related quality of life, the Kansas City Cardiomyopathy Questionnaire (KCCQ) and the Functional Assessment of Chronic Illness Therapy – Palliative Care (FACIT-PAL). The KCCQ showed the palliative care intervention linked with an average rise of more than 9 points compared with patients in the control arm after adjustment for age and sex, a statistically significant increase on a scale where a 5-point rise is considered clinically meaningful. The FACIT-PAL showed an average, adjusted 11-point rise linked with the intervention, a statistically significant increase on a scale where an increase of at least 10 is judged clinically meaningful, reported Dr. Rogers, a heart failure cardiologist and professor of medicine at Duke University.
The palliative-care intervention also led to significant improvements in measures of spirituality, depression, and anxiety, but intervention had no impact on mortality.
“I like these endpoints and the idea that we can make quality-of-life better. These are very sick patients, with a predicted 6-month mortality of 50%. Patients reach a time when they don’t want to live longer but want better life quality for the days they still have,” he said in an interview.
The second report came from a single-center pilot study of 50 patients enrolled when they were hospitalized for acute decompensated heart failure and had at least one addition risk factor for poor prognosis such as age of at least 81 years, renal dysfunction, or a prior heart failure hospitalization within the past year. Patients randomized to the intervention arm underwent a structured evaluation based on the Serious Illness Conversation Guide and performed by a social worker experienced in palliative care and embedded in the heart failure clinical team. The primary endpoint of the SWAP-HF (Social Worker–Aided Palliative Care Intervention in High Risk Patients with Heart Failure) study was clinical-level documentation of advanced-care preferences by 6 months after the program began.
“Although more comprehensive, multidisciplinary palliative care interventions may also be effective, the focused approach [used in this study] may represent a cost-effective and scalable method for shepherding limited specialty resources to enhance the delivery of patient-centered care,” Dr. Desai said. In other words, a program with a social worker costs less than a two-person staff with a palliative-care physician and nurse practitioner.
Despite its relative simplicity, the SWAP-HF intervention had some unique aspects that make it generalizability uncertain, commented Dr. Allen. The embedding of a social worker on the heart failure team placed a professional with a “good understanding of social context” right on the scene with everyone else delivering care to the heart failure patient, a good strategy for minimizing fragmentation, he said. In addition, the place where the study was done, Brigham and Women’s Hospital, “is not your average hospital,” he noted,
In addition, the timing of the intervention studied during hospitalization may be problematic. Clinicians need to “be careful about patients making long-term decisions” about their care while they are hospitalized, a time when patients can be “ill, confused, and scared.” He cited recent findings from a study of hospital-based palliative-care interventions for family members of patients with chronic critical illness that did not reduce anxiety or depression symptoms among the treated family members and may have increased symptoms of posttraumatic stress disorder (JAMA. 2016 July 5;374[1]:51-62).
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ORLANDO – Systematic introduction of palliative care interventions for patients with advanced heart failure improved patients’ quality of life and spurred their development of advanced-care preferences in a pair of independently performed, controlled, pilot studies.
But, despite demonstrating the ability of palliative-care interventions to help heart failure patients during their final months of life, the findings raised questions about the generalizability and reproducibility of palliative-care interventions that may depend on the skills and experience of the individual specialists who deliver the palliative care.
“Palliative care for patients with cardiovascular disease is in desperate need of good-quality evidence,” commented Larry A, Allen, MD, a heart failure cardiologist at the University of Colorado in Aurora and designated discussant for one of the two studies presented at the meeting. “We need large, randomized trials with clinical outcomes to look at patient outcomes from palliative-care interventions.”
The patients average 71 years old, about half were women, and about 40% were African Americans. They had been diagnosed with heart failure for an average of more than 5 years, all had advanced heart failure, about 60% spent at least half of their time awake immobilized in a bed or chair, and they had average NT-proBNP blood levels of greater than 10,000 pg/mL.
After 24 weeks of intervention, the palliative-care program produced both statistically significant and clinically meaningful improvements in two different measures of health-related quality of life, the Kansas City Cardiomyopathy Questionnaire (KCCQ) and the Functional Assessment of Chronic Illness Therapy – Palliative Care (FACIT-PAL). The KCCQ showed the palliative care intervention linked with an average rise of more than 9 points compared with patients in the control arm after adjustment for age and sex, a statistically significant increase on a scale where a 5-point rise is considered clinically meaningful. The FACIT-PAL showed an average, adjusted 11-point rise linked with the intervention, a statistically significant increase on a scale where an increase of at least 10 is judged clinically meaningful, reported Dr. Rogers, a heart failure cardiologist and professor of medicine at Duke University.
The palliative-care intervention also led to significant improvements in measures of spirituality, depression, and anxiety, but intervention had no impact on mortality.
“I like these endpoints and the idea that we can make quality-of-life better. These are very sick patients, with a predicted 6-month mortality of 50%. Patients reach a time when they don’t want to live longer but want better life quality for the days they still have,” he said in an interview.
The second report came from a single-center pilot study of 50 patients enrolled when they were hospitalized for acute decompensated heart failure and had at least one addition risk factor for poor prognosis such as age of at least 81 years, renal dysfunction, or a prior heart failure hospitalization within the past year. Patients randomized to the intervention arm underwent a structured evaluation based on the Serious Illness Conversation Guide and performed by a social worker experienced in palliative care and embedded in the heart failure clinical team. The primary endpoint of the SWAP-HF (Social Worker–Aided Palliative Care Intervention in High Risk Patients with Heart Failure) study was clinical-level documentation of advanced-care preferences by 6 months after the program began.
“Although more comprehensive, multidisciplinary palliative care interventions may also be effective, the focused approach [used in this study] may represent a cost-effective and scalable method for shepherding limited specialty resources to enhance the delivery of patient-centered care,” Dr. Desai said. In other words, a program with a social worker costs less than a two-person staff with a palliative-care physician and nurse practitioner.
Despite its relative simplicity, the SWAP-HF intervention had some unique aspects that make it generalizability uncertain, commented Dr. Allen. The embedding of a social worker on the heart failure team placed a professional with a “good understanding of social context” right on the scene with everyone else delivering care to the heart failure patient, a good strategy for minimizing fragmentation, he said. In addition, the place where the study was done, Brigham and Women’s Hospital, “is not your average hospital,” he noted,
In addition, the timing of the intervention studied during hospitalization may be problematic. Clinicians need to “be careful about patients making long-term decisions” about their care while they are hospitalized, a time when patients can be “ill, confused, and scared.” He cited recent findings from a study of hospital-based palliative-care interventions for family members of patients with chronic critical illness that did not reduce anxiety or depression symptoms among the treated family members and may have increased symptoms of posttraumatic stress disorder (JAMA. 2016 July 5;374[1]:51-62).
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT THE HFSA ANNUAL SCIENTIFIC MEETING
Key clinical point:
Major finding: Palliative care measures boosted patients’ Kansas City Cardiomyopathy Questionnaire score by an average of 9 points over that of controls.
Data source: PAL-HF, a single-center study with 150 randomized patients with heart failure and SWAP-HF, a single-center study with 50 randomized patients.
Disclosures: Dr. Rogers, Dr. Allen, and Dr. Desai had no relevant disclosures.
Elevated troponins are serious business, even without an MI
Sometimes it seems like cardiac troponin testing has become nearly as ubiquitous as the CBC and the BMP. Concern over atypical presentations of MI has contributed to widespread use in emergency departments and hospitalized patients. But once the test comes back elevated, what do you do with that information?
Typically, the next step is to consult Cardiology, which is a reasonable request with or without a suspicion of MI. Frequently, invasive management is not an option; or perhaps the diagnosis is “type 2 MI.”1
A growing body of evidence is making it clear that any elevation in cardiac troponin is a serious predictor of risk and that the risk is highest if the patient is not having an MI.2 My colleagues and I recently conducted a cohort study of more than 700 veterans at our VA Medical Center addressing this question. We evaluated long-term mortality (6 years) comparing veterans who were diagnosed with MI with those who had troponin elevation and no clinical MI. The diagnostic determination was made for all subjects prospectively as part of a quality improvement project that sought to better care for MI patients at our facility. (In some cases, only single troponin values were measured so we cannot say that all patients in our investigation had a true type 2 MI.)
We found that veterans with an elevation in troponin that was not caused by MI had higher risk of mortality risk than did MI patients.3 The risk started to diverge at 30 days and was 42.0% at 1 year, compared with 29.0% for those with MI (odds ratio, 0.56; 95% confidence interval, 0.41-0.78). This risk continued to separate and, at 6 years, was 77.7% vs. 58.7% (OR, 0.41; 95% CI 0.30-0.56). Our observations agree with other recent publications; what we tried to do in advancing the literature was to construct a robust Cox proportional hazard model to try to better understand if the risk seen in these patients is just because of their being “sicker.”
We tried to capture a number of other acute illness states with variables including TIMI score, being in hospice care, having a “do not resuscitate” order, being in the ICU, receiving CPR, and having a fever or leukocytosis, etc. Despite this modeling, elevated troponin remained a significant predictor of risk. While several variables we modeled remained significant predictors of mortality, their distribution between our two cohorts did not explain the excess mortality risk associated with non-MI troponin.
Unfortunately, there are no viable treatment options specific for patients with non-MI troponin elevation and type 2 MI. Given that the causes are multiple and heterogeneous, there may not be a common pathway to target for reducing cardiovascular risk. Regardless, the observation of non-MI troponin or type 2 MI should be taken seriously and not be ignored.
In selected patients, particularly those without known coronary artery disease, it may be appropriate to perform diagnostic testing or risk assessment with noninvasive imaging prior to discharge. Those with coronary artery disease should be treated aggressively for prevention of future cardiovascular events with both medical therapy and risk factor reduction.
1. Thygesen K, Alpert JS, et al. Third universal definition of myocardial infarction. J Am Coll Cardiol. 2012;60:1581-98.
2. Alcalai R, Planer D, Culhaoglu A, Osman A, Pollak A and Lotan C. Acute coronary syndrome vs nonspecific troponin elevation: clinical predictors and survival analysis. Arch Intern Med. 2007;167:276-81.
3. Winchester DE, Burke L, Agarwal N, Schmalfuss C and Pepine CJ. Predictors of short- and long-term mortality in hospitalized veterans with elevated troponin. J Hosp Med. 2016 Jun 3. doi: 10.1002/jhm.2619.
David Winchester, MD, is assistant professor in the division of cardiovascular medicine at the University of Florida (Gainesville), and practices general cardiology at the Malcom Randall VA Medical Center, Gainesville.
Sometimes it seems like cardiac troponin testing has become nearly as ubiquitous as the CBC and the BMP. Concern over atypical presentations of MI has contributed to widespread use in emergency departments and hospitalized patients. But once the test comes back elevated, what do you do with that information?
Typically, the next step is to consult Cardiology, which is a reasonable request with or without a suspicion of MI. Frequently, invasive management is not an option; or perhaps the diagnosis is “type 2 MI.”1
A growing body of evidence is making it clear that any elevation in cardiac troponin is a serious predictor of risk and that the risk is highest if the patient is not having an MI.2 My colleagues and I recently conducted a cohort study of more than 700 veterans at our VA Medical Center addressing this question. We evaluated long-term mortality (6 years) comparing veterans who were diagnosed with MI with those who had troponin elevation and no clinical MI. The diagnostic determination was made for all subjects prospectively as part of a quality improvement project that sought to better care for MI patients at our facility. (In some cases, only single troponin values were measured so we cannot say that all patients in our investigation had a true type 2 MI.)
We found that veterans with an elevation in troponin that was not caused by MI had higher risk of mortality risk than did MI patients.3 The risk started to diverge at 30 days and was 42.0% at 1 year, compared with 29.0% for those with MI (odds ratio, 0.56; 95% confidence interval, 0.41-0.78). This risk continued to separate and, at 6 years, was 77.7% vs. 58.7% (OR, 0.41; 95% CI 0.30-0.56). Our observations agree with other recent publications; what we tried to do in advancing the literature was to construct a robust Cox proportional hazard model to try to better understand if the risk seen in these patients is just because of their being “sicker.”
We tried to capture a number of other acute illness states with variables including TIMI score, being in hospice care, having a “do not resuscitate” order, being in the ICU, receiving CPR, and having a fever or leukocytosis, etc. Despite this modeling, elevated troponin remained a significant predictor of risk. While several variables we modeled remained significant predictors of mortality, their distribution between our two cohorts did not explain the excess mortality risk associated with non-MI troponin.
Unfortunately, there are no viable treatment options specific for patients with non-MI troponin elevation and type 2 MI. Given that the causes are multiple and heterogeneous, there may not be a common pathway to target for reducing cardiovascular risk. Regardless, the observation of non-MI troponin or type 2 MI should be taken seriously and not be ignored.
In selected patients, particularly those without known coronary artery disease, it may be appropriate to perform diagnostic testing or risk assessment with noninvasive imaging prior to discharge. Those with coronary artery disease should be treated aggressively for prevention of future cardiovascular events with both medical therapy and risk factor reduction.
1. Thygesen K, Alpert JS, et al. Third universal definition of myocardial infarction. J Am Coll Cardiol. 2012;60:1581-98.
2. Alcalai R, Planer D, Culhaoglu A, Osman A, Pollak A and Lotan C. Acute coronary syndrome vs nonspecific troponin elevation: clinical predictors and survival analysis. Arch Intern Med. 2007;167:276-81.
3. Winchester DE, Burke L, Agarwal N, Schmalfuss C and Pepine CJ. Predictors of short- and long-term mortality in hospitalized veterans with elevated troponin. J Hosp Med. 2016 Jun 3. doi: 10.1002/jhm.2619.
David Winchester, MD, is assistant professor in the division of cardiovascular medicine at the University of Florida (Gainesville), and practices general cardiology at the Malcom Randall VA Medical Center, Gainesville.
Sometimes it seems like cardiac troponin testing has become nearly as ubiquitous as the CBC and the BMP. Concern over atypical presentations of MI has contributed to widespread use in emergency departments and hospitalized patients. But once the test comes back elevated, what do you do with that information?
Typically, the next step is to consult Cardiology, which is a reasonable request with or without a suspicion of MI. Frequently, invasive management is not an option; or perhaps the diagnosis is “type 2 MI.”1
A growing body of evidence is making it clear that any elevation in cardiac troponin is a serious predictor of risk and that the risk is highest if the patient is not having an MI.2 My colleagues and I recently conducted a cohort study of more than 700 veterans at our VA Medical Center addressing this question. We evaluated long-term mortality (6 years) comparing veterans who were diagnosed with MI with those who had troponin elevation and no clinical MI. The diagnostic determination was made for all subjects prospectively as part of a quality improvement project that sought to better care for MI patients at our facility. (In some cases, only single troponin values were measured so we cannot say that all patients in our investigation had a true type 2 MI.)
We found that veterans with an elevation in troponin that was not caused by MI had higher risk of mortality risk than did MI patients.3 The risk started to diverge at 30 days and was 42.0% at 1 year, compared with 29.0% for those with MI (odds ratio, 0.56; 95% confidence interval, 0.41-0.78). This risk continued to separate and, at 6 years, was 77.7% vs. 58.7% (OR, 0.41; 95% CI 0.30-0.56). Our observations agree with other recent publications; what we tried to do in advancing the literature was to construct a robust Cox proportional hazard model to try to better understand if the risk seen in these patients is just because of their being “sicker.”
We tried to capture a number of other acute illness states with variables including TIMI score, being in hospice care, having a “do not resuscitate” order, being in the ICU, receiving CPR, and having a fever or leukocytosis, etc. Despite this modeling, elevated troponin remained a significant predictor of risk. While several variables we modeled remained significant predictors of mortality, their distribution between our two cohorts did not explain the excess mortality risk associated with non-MI troponin.
Unfortunately, there are no viable treatment options specific for patients with non-MI troponin elevation and type 2 MI. Given that the causes are multiple and heterogeneous, there may not be a common pathway to target for reducing cardiovascular risk. Regardless, the observation of non-MI troponin or type 2 MI should be taken seriously and not be ignored.
In selected patients, particularly those without known coronary artery disease, it may be appropriate to perform diagnostic testing or risk assessment with noninvasive imaging prior to discharge. Those with coronary artery disease should be treated aggressively for prevention of future cardiovascular events with both medical therapy and risk factor reduction.
1. Thygesen K, Alpert JS, et al. Third universal definition of myocardial infarction. J Am Coll Cardiol. 2012;60:1581-98.
2. Alcalai R, Planer D, Culhaoglu A, Osman A, Pollak A and Lotan C. Acute coronary syndrome vs nonspecific troponin elevation: clinical predictors and survival analysis. Arch Intern Med. 2007;167:276-81.
3. Winchester DE, Burke L, Agarwal N, Schmalfuss C and Pepine CJ. Predictors of short- and long-term mortality in hospitalized veterans with elevated troponin. J Hosp Med. 2016 Jun 3. doi: 10.1002/jhm.2619.
David Winchester, MD, is assistant professor in the division of cardiovascular medicine at the University of Florida (Gainesville), and practices general cardiology at the Malcom Randall VA Medical Center, Gainesville.