User login
Improving Nephropathy Screening in Appalachian Patients With Diabetes Using Practice-Wide Outreach
From West Virginia University, Morgantown, WV.
Abstract
Objective: To describe the strategies a family medicine clinic in Appalachia utilized to increase nephropathy screening rates as well as to explore the factors predictive of nephropathy screening in patients with diabetes.
Design: This quality improvement project targeted the points in the care process when patients are lost to follow-up for nephropathy screening.
Setting and participants: Patients with diabetes cared for by a primary care provider (PCP) at an academic family medicine practice in Appalachia from January 2018 to November 2018.
Interventions: Bulk orders for albumin-to-creatinine (ACR) testing and urine collection during clinic visit, enhanced patient communication through bulk communication reminders and individual patient outreach, and education of clinic providers.
Measurements: Demographic data and monthly nephropathy screening rates.
Results: The nephropathy screening rate increased by 6.2% during the project. Older patients living closer to the clinic who visited their PCP 3 or more times per year were the most likely to be screened.
Conclusion: Combining team-based interventions with quality control monitoring can significantly improve compliance with recommended nephropathy treatment and screening in rural patients with diabetes at a family medicine clinic.
Keywords: rural; kidney disease; albumin-to-creatinine ratio; electronic health record.
According to the Centers for Disease Control and Prevention (CDC), an estimated 30.3 million people in the United States—about 9.4% of the population—have been diagnosed with diabetes.1 Diabetes is the seventh leading cause of death in the United States, and it contributes to other leading causes of death: heart disease and stroke.1 Diabetes also is related to high morbidity risk and is a leading cause of chronic kidney disease.1 The total cost of diagnosed diabetes was estimated at $327 billion in direct medical costs and reduced productivity.2
Residents of Appalachia bear a disproportionate burden of diabetes and other related negative health outcomes; these outcomes are influenced by a number of factors, including socioeconomic status, poverty, rurality, and health care access. Rates of chronic disease, such as diabetes, are most pronounced in Appalachia’s most economically distressed counties.3-5 In 2011, the CDC labeled a 644-county area the “diabetes belt,” which included most of Appalachia.6 As a result of this elevated prevalence of diabetes in Appalachia as compared to the rest of the country, complications directly associated with diabetes are more commonly observed in Appalachian residents. One of the most damaging complications is diabetic nephropathy.
Diabetic nephropathy results from damage to the microvasculature of the kidney due to inadequately controlled blood glucose. This, in turn, leads to decreased renal function, eventually leading to clinically significant renal disease. The long-term complications associated with nephropathy can include many comorbid conditions, the most serious of which are progression to end-stage renal disease, dialysis requirement, and early mortality. Diabetic nephropathy affects approximately 40% of patients with type 1 and type 2 diabetes.7,8
One way to prevent complications of diabetic nephropathy, in addition to good glycemic control in patients with diabetes, is early and regular screening. Currently, the American Diabetes Association (ADA) recommends yearly screening for diabetic nephropathy in the form of a urine albumin-to-creatinine ratio (ACR) for patients 18 to 75 years of age.2 This screening to detect diabetic nephropathy is recognized as a marker of quality care by many public and private insurance agencies and medical specialty associations, such as the Centers for Medicare and Medicaid Services.
Many patients with diabetes are cared for by primary care providers (PCP), and these PCP appointments provide an opportune time to screen and appropriately treat nephropathy. Screening opportunities are often missed, however, due to time constraints and competing health priorities. There are also a number of other factors specific to the Appalachian region that reduce the likelihood of screening for diabetic nephropathy, such as a lack of health insurance, the need to travel long distances to see a PCP, work and household responsibilities, low levels of education and health literacy, and a mistrust of outsiders regarding personal matters, including health.9-11 While nephropathy can have a detrimental impact on patients across populations, it is of particular concern for a state located in the heart of Appalachia, such as West Virginia.
Given the disproportionate burden of diabetes in this region and the potentially severe consequences of undetected nephropathy, clinicians from an academic family medicine clinic in West Virginia undertook a quality improvement project to increase the rate of nephropathy screening and treatment among patients with diabetes. This article describes the intervention strategies the team utilized to increase nephropathy screening and treatment in patients 18 to 75 years of age who met quality measures for nephropathy screening or treatment in the previous 12 months and explores the factors most predictive of nephropathy screening in Appalachian patients in this age group. It also reports the challenges and opportunities encountered and offers suggestions for other providers and clinics attempting to increase their nephropathy screening rates.
Methods
Setting and Study Population
The study population included patients ages 18 to 75 years under the care of providers in an academic family medicine practice in West Virginia who had been diagnosed with diabetes mellitus. The study focused on those patients overdue for diabetic nephropathy screening (ie, had not been screened in previous 12 months). The project began in January 2018 with a screening rate of 83.8%. The goal of this project was to increase this compliance metric by at least 5%. The project protocol was submitted to the West Virginia University Institutional Review Board, and, because it is a quality improvement project, permission was given to proceed without a board review.
Interventions
The team identified and implemented several interventions intended to reduce screening barriers and increase the screening rate.
Bulk orders for ACR and urine collection during clinic visits. Prior to initiation of this project, it was left to individual clinic providers to order nephropathy screening for patients with diabetes during a clinic visit; after receiving the order for “random urine microalbumin/creatinine ratio,” patients then had to travel to a lab to provide a urine sample. For this project and moving forward, the team changed to the procedure of initiating bulk ACR orders and collecting urine samples during clinic visits from all patients ages 18 to 75 years who have diabetes.
Bulk communication reminders. Since many patients with diabetes may not have realized they were overdue for nephropathy screening, the team began sending out bulk communication reminders through either the institution’s electronic health record (EHR; MyChart) or postal service–delivered physical letters (according to patient communication preferences) to remind patients that they were due for screening and to encourage them to schedule an appointment or keep a previously scheduled appointment with their PCP.
Individual patient outreach. A team of pharmacy students led by a licensed pharmacist in the family medicine clinic contacted patients overdue for screening even after bulk communication reminders went out. The students telephoned patients 2 to 3 months following the bulk communication. The students obtained an updated list of patients with diabetes ages 18 to 75 years from an EHR quality report. They began by prescreening the patients on the overdue list for potential candidacy for an angiotensin-converting enzyme (ACE) inhibitor or an angiotensin II receptor blocker (ARB). Screening for candidacy included evaluation of recent blood pressure readings, electrolytes (ie, basic metabolic panel), and ACR. If the students determined a patient was a candidate, they presented the patient to the preceptor for verification and then reached out to the provider with a recommendation. If the provider agreed, the student contacted the patient by telephone for medication counseling and education. The remaining patients determined not to be candidates for ACE inhibitors or ARBs were contacted by the pharmacy students by telephone to remind them that laboratory work was pending. Up to 3 phone call attempts were made before patients were determined to be unreachable. Students left voice mails with generic reminders if a patient could not be reached. If a patient answered, the student provided a reminder but also reviewed indications for lab work, the reason why the provider wished for follow-up, and updated lab hours. Students also followed up with the results of the work-up, as appropriate. During this outreach process, the student team encountered a number of patients who had moved or changed to a PCP outside of the family medicine clinic. In these cases, the EHR was updated and those patients were removed from the list of patients altogether.
Education of clinic providers. Clinic providers were educated during faculty and resident meetings and didactic learning sessions on identifying patients within the EHR who are due for nephropathy screening. They also received instruction on how to update the EHR to reflect completed screenings.
Data Analysis
All analyses in this study were conducted using SAS (version 9.4, 2013, SAS Institute Inc., Cary, NC). Descriptive analyses were conducted to summarize basic patient demographic information. To compare patients screened within the previous 12 months to those patients overdue for screening, 2-sample t-tests were used to examine differences in patients’ age, HbA1c, ACR, and creatinine level and the distance (in miles) between the patient’s home and the clinic. Chi-square analyses were used to examine the relationship between whether a patient was recently screened for nephropathy and the patient’s insurance, number of patient visits in the previous 12 months, and provider level. Logistic regression analyses were conducted to control for covariates and to explore which factors were most predictive of nephropathy screening. All tests were 2-tailed, and P values less than 0.05 were considered statistically significant.
Results
Patient Characteristics
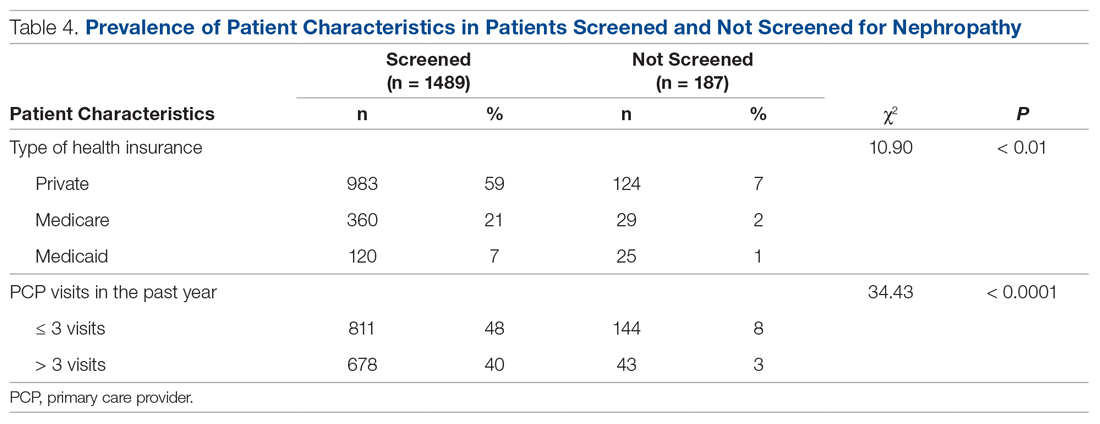
There were 1676 family medicine clinic patients with diabetes between 18 and 75 years of age (Table 1 and Table 2). Of the total sample, 1489 (88.8%) had completed screening for nephropathy in the 12 months prior to evaluation, and 67.5%, 23.7%, and 8.8% of patients had private insurance, Medicare, and Medicaid, respectively. 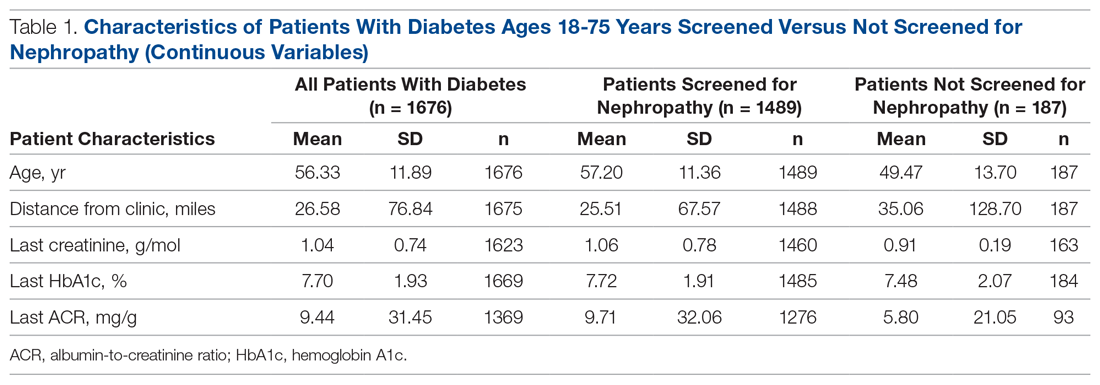
The mean (SD) age of the patients was 56.3 (11.9) years. The mean distance between the patient’s home and the clinic was 26.6 (76.8) miles. The mean number of visits was 3.6 (2.9) per year, and 43.0% of the patientvisited the clinic more than 3 times in a year. The mean values for HbA1c (%), creatinine (g/mol), and ACR (mg/g) were 7.7 (1.9), 1.0 (0.7), and 9.4 (31.4), respectively.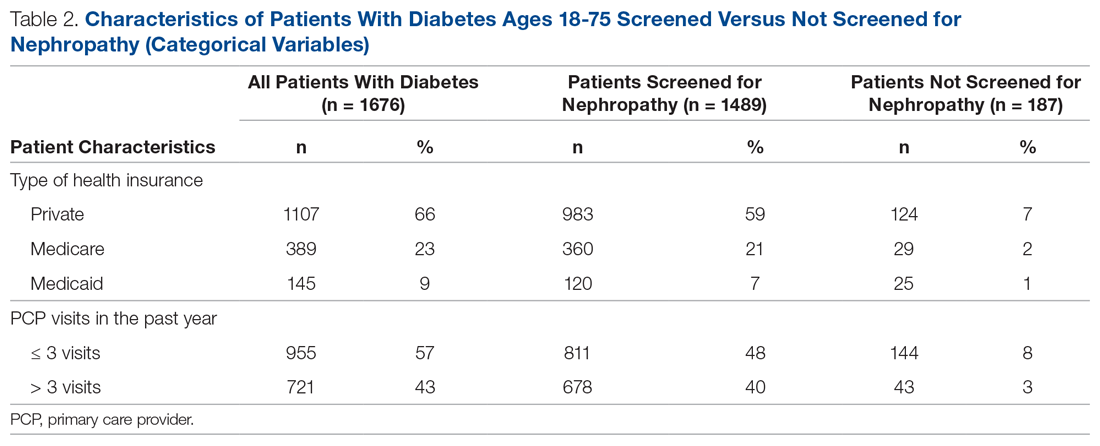
Screening of Patients for Nephropathy
Patients with Medicare and private insurance were more likely to have completed the nephropathy screening than those with Medicaid (92.5% versus 88.8% versus 82.8%, P = 0.004; Table 3 and Table 4). 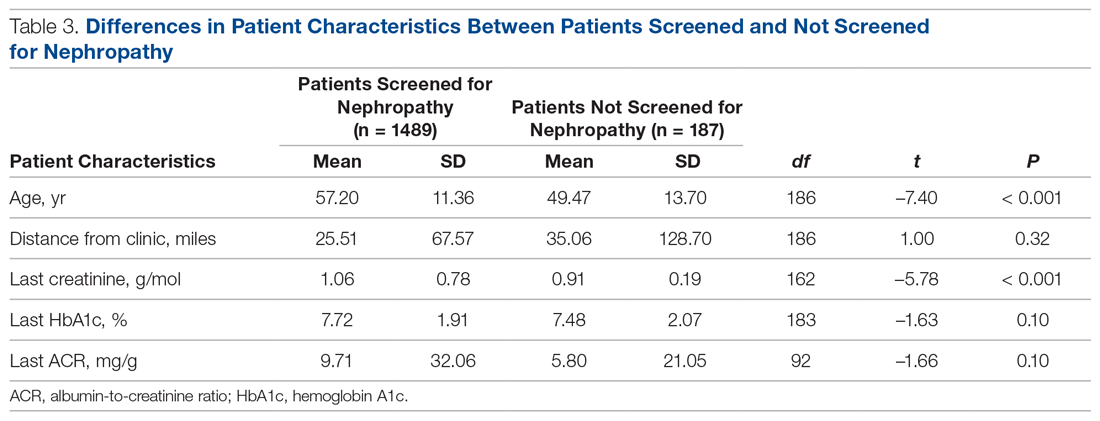
Changes in Screening Rate
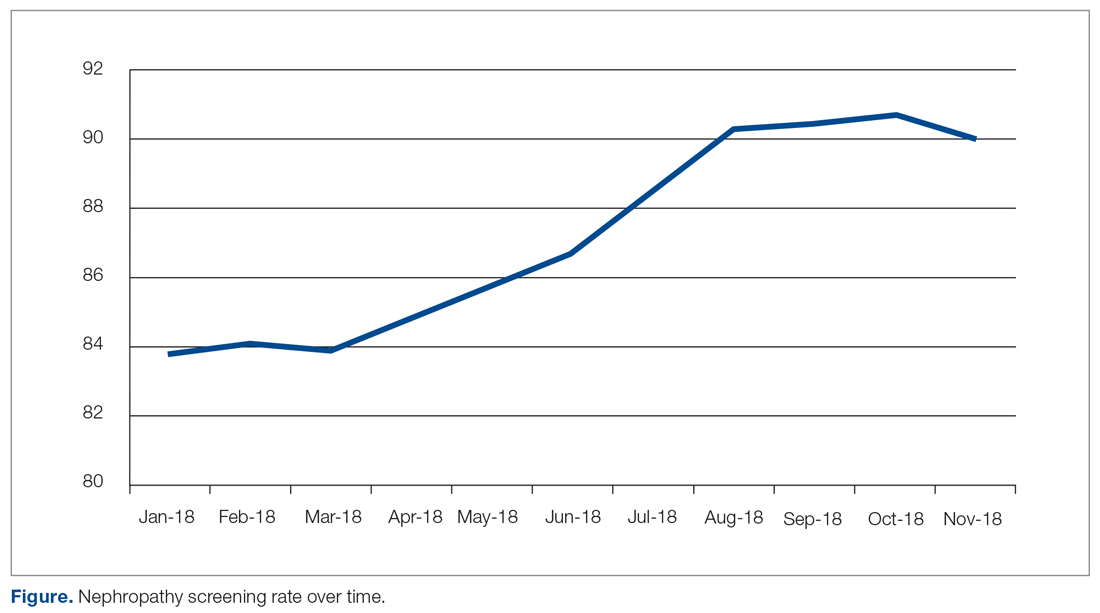
The practice-wide screening rate was 83.8% at the start of this project in January 2018. The screening rate steadily increased throughout 2018, reaching 90.3% in August 2018, and then leveled off around 90% when the project was concluded at the end of November 2018 (Figure). As an added benefit of the increased screening rates, a number of patients were initiated on an ACE inhibitor or ARB based on the team’s screening efforts.
Predictors of Nephropathy Screening
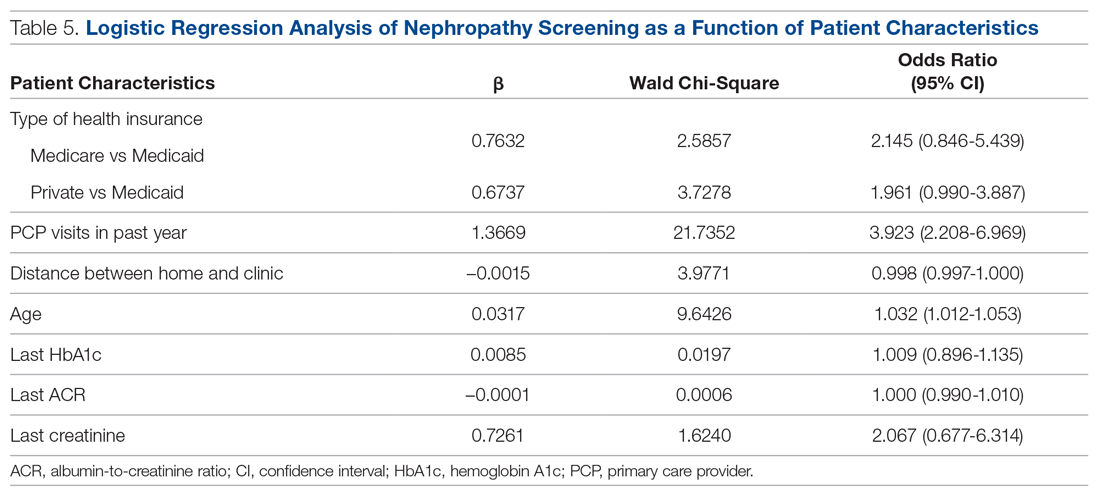
A logistic regression analysis was conducted with nephropathy screening (screened or not screened) as the outcome and 7 patient characteristics as predictors: type of insurance (private, Medicare, or Medicaid), PCP visits in the past 12 months (≤ 3 or > 3), distance in miles of the patient’s residence from the clinic, age, last HbA1c value, last ACR value, and last creatinine value. A test of the full model with all 7 predictors was statistically significant (χ2 (8) = 57.77, P < 0.001). Table 5 shows regression coefficients, Wald statistics, and 95% confidence intervals for odds ratios for each of the 7 predictors. According to the Wald criterion, 3 patient characteristics were significant predictors of nephropathy screening: age, distance between the patient’s home and clinic, and number of PCP visits in the past 12 months. After adjusting for the covariates, there were still significant associations between the nephropathy screening status and age ( χ2(1) = 9.64, P < 0.01); distance between the patient’s home and the clinic (χ2(1) = 3.98, P < 0.05); and the number of PCP visits in the previous year (χ2(1) = 21.74, P < 0.001). With each 1-year increment in age, the odds of completing the nephropathy screening increased by 3.2%. With each 1-mile increase in the distance between the patient’s home and clinic, the odds of completing the nephropathy screening decreased by 0.2%. Patients who visited the clinic more than 3 times in a year were 3.9 times (95% confidence interval, 2.2-7.0) more likely to complete the nephropathy screening than their counterparts who visited fewer than 3 times per year.
In summary, older patients living within about 164 miles of the clinic (ie, within 1 standard deviation from the average miles between patient’s homes and the clinic) who visited their PCP 3 or more times per year were the most likely to be screened.
Discussion
Diabetic nephropathy is a critical issue facing family medicine providers and patients. The morbidity and mortality costs are significant, as diabetic nephropathy is the leading cause of end-stage renal disease. While the ADA recommends annual ACR screening in patients with diabetes and prescription of ACE inhibitors or ARBs in patients who qualify, many patients do not receive these interventions, despite following up with a provider.12-15 There is no current literature that indicates the compliance rates in the rural setting. Due to health disparities in the rural setting noted in the literature, it could be hypothesized that these individuals are at high risk of not meeting these screening and treatment recommendations.16,17 Limited access to care and resources, gaps in insurance coverage, and lower health literacy are a few barriers identified in the rural population that may influence whether these measures are met.17
Considering the disease burden of diabetes and its related complications, including nephropathy, consistent screening is necessary to reduce diabetes-related burdens and cost, while also increasing the quality of life for patients with diabetes. All parties must be involved to ensure appropriate compliance and treatment. Our institution’s implementation of quality improvement strategies has key implications for nephropathy screening and treatment efforts in rural settings.
An additional step of having a health care provider (other than the PCP) screen all patients who are not meeting the standard allows for identification of gaps in care. In our quality improvement workflow, the clinical pharmacist screened all patients for candidacy for ACE inhibitor/ARB therapy. While only a small percentage of patients qualified, many of these patients had previously been on therapy and were discontinued for an unknown reason or were stopped due to an acute condition (eg, acute kidney injury) and never restarted after recovery. Other patients required additional education that therapy would be utilized for nephroprotection versus blood pressure management (secondary to an elevated ACR). This highlights the importance of transitions of care and ongoing, intensive education, not only during initial diagnosis but also throughout the disease-state progression.
Utilization of EHRs and telephone outreach are additional aspects of care that can be provided. Our improved rates of compliance with these care interventions parallel findings from previous studies.15,18 Optimization of an institution’s EHR can aid in standardization of care, workflow management, and communication with patients, as well as alert nursing or support staff of screening needs. Techniques such as best practice reminders, patient chart messages, and nursing-entered physician alerts on daily schedules have been shown to increase rates of compliance with nephropathy standards. These findings underscore an additional opportunity for nursing and support staff to be better integrated into care.
Despite the success of this quality improvement initiative, there remain some limitations. The processes we used in this project may not be applicable to every institution and may have limited external validity. Primarily, while these processes may be implemented at some sites, without additional support staff (ie, extra nursing staff, pharmacists) and students to aid in patient outreach, success may be limited due to provider time constraints. Additionally, our workflow process demonstrates significant incorporation of an EHR system for patient outreach. Institutions and/or clinics that heavily rely on paper charts and paper outreach may face barriers with bulk orders (eg, ACR) and messages, interventions that streamlined our population health management. Finally, this project focuses on only 1 aspect of population health management for patients with diabetes. While nephropathy is a critical aspect of caring for individuals with diabetes, this patient outreach does not address retinopathy screening, HbA1c control, or vaccination rates, which are other components of care.
Conclusion
Although this evaluation does not provide insight into why patients were not treated or screened, it demonstrates processes to improve compliance in patients with diabetic nephropathy. Rural health care facilities require an ongoing program of change and evaluation, with the aim to improve the provision of services, increase screening, and encourage team member involvement in health promotion. This study demonstrates that combining team-based interventions with quality control monitoring can significantly improve compliance with recommended nephropathy screening and treatment in rural patients with diabetes at a family medicine clinic.
Corresponding author: Amie M. Ashcraft, West Virginia University, Department of Family Medicine, 1 Medical Center Drive, Box 9152, Morgantown, WV 26506; amashcraft@hsc.wvu.edu.
Financial disclosures: None.
Acknowledgment: The authors thank the faculty, residents, nurses, and clinic staff for their hard work and dedication to this effort: Umama Sadia, Michelle Prestoza, Richard Dattola, Greg Doyle, Dana King, Mike Maroon, Kendra Under, Judy Siebert, Christine Snyder, Rachel Burge, Meagan Gribble, Lisa Metts, Kelsey Samek, Sarah Deavers, Amber Kitzmiller, Angela Lamp, Tina Waldeck, and Andrea Sukeruksa.
1. Centers for Disease Control and Prevention (CDC). National diabetes statistics report. Estimates of diabetes and its burden in the United States. Atlanta, GA: CDC; 2017www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed December 20, 2020.
2. American Diabetes Association (ADA). Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917-928.
3. Wood L. Trends in national and regional economic distress, 1960-2000. Washington, DC: Appalachian Regional Commission; 2005.
4. Barker L, Crespo R, Gerzoff RB, et al. Residence in a distressed county in Appalachia as a risk factor for diabetes, Behavioral Risk Factor Surveillance System, 2006-2007. Prev Chronic Dis. 2010;7:A104.
5. Barker L, Kirtland KA, Gregg E, et al. Geographic distribution of diagnosed diabetes in the United States: A diabetes belt. Am J Prev Med. 2011;40:434-439.
6. Gross JL, de Azevedo MJ, Silveiro SP, et al. Diabetic nephropathy: Diagnosis, prevention, and treatment. Diabetes Care. 2005;28:164-176.
7. United States Renal Data System (USRDS). Annual data report. Ann Arbor, MI: USRDS; 2018. www.usrds.org/2018/view/Default.aspx. Accessed December 20, 2020.
8. Halverson JA, Bichak G. Underlying socioeconomic factors influencing health disparities in the Appalachian region. Washington, DC: Appalachian Regional Commission; 2008.
9. Shell R, Tudiver F. Barriers to cancer screening by rural Appalachian primary care providers. J Rural Health. 2004;20:368-373.
10. Hatcher J, Dignan MB, Schoenberg N. How do rural health care providers and patients view barriers to colorectal cancer screening? Insights from Appalachian Kentucky. Nurs Clin North Am. 2011;46:181-192.
11. Scott S, McSpirit S. The suspicious, untrusting hillbilly in political-economic contexts: Stereotypes and social trust in the Appalachian coalfields. Pract Anthropol. 2014;36:42-46.
12. Kirkman MS, Williams SR, Caffrey HH, Marrero DG. Impact of a program to improve adherence to diabetes guidelines by primary care physicians. Diabetes Care. 2002;25:1946-1951.
13. Byun SH, Ma SH, Jun JK, et al. Screening for diabetic retinopathy and nephropathy in patients with diabetes: A nationwide survey in Korea. PLoS One. 2013;8:e62991.
14. Flood D, Garcia P, Douglas K, et al. Screening for chronic kidney disease in a community-based diabetes cohort in rural Guatemala: A cross-sectional study. BMJ Open. 2018;8:e019778.
15. Anabtawi A, Mathew LM. Improving compliance with screening of diabetic patients for microalbuminuria in primary care practice. ISRN Endocrinology. 2013:893913.
16. Tonks SA, Makwana S, Salanitro AH, et al. Quality of diabetes mellitus care by rural primary care physicians. J Rural Health. 2012;28:364-371.
17. Douthit N, Kiv S, Dwolatzky T, Biswas S. Exposing some important barriers to health care access in the rural USA. Public Health. 2015;129:611-620.
18. Weber V, Bloom F, Pierdon S, Wood C. Employing the electronic health record to improve diabetes care: a multifaceted intervention in an integrated delivery system. J Gen Intern Med. 2008;23:379-382.
From West Virginia University, Morgantown, WV.
Abstract
Objective: To describe the strategies a family medicine clinic in Appalachia utilized to increase nephropathy screening rates as well as to explore the factors predictive of nephropathy screening in patients with diabetes.
Design: This quality improvement project targeted the points in the care process when patients are lost to follow-up for nephropathy screening.
Setting and participants: Patients with diabetes cared for by a primary care provider (PCP) at an academic family medicine practice in Appalachia from January 2018 to November 2018.
Interventions: Bulk orders for albumin-to-creatinine (ACR) testing and urine collection during clinic visit, enhanced patient communication through bulk communication reminders and individual patient outreach, and education of clinic providers.
Measurements: Demographic data and monthly nephropathy screening rates.
Results: The nephropathy screening rate increased by 6.2% during the project. Older patients living closer to the clinic who visited their PCP 3 or more times per year were the most likely to be screened.
Conclusion: Combining team-based interventions with quality control monitoring can significantly improve compliance with recommended nephropathy treatment and screening in rural patients with diabetes at a family medicine clinic.
Keywords: rural; kidney disease; albumin-to-creatinine ratio; electronic health record.
According to the Centers for Disease Control and Prevention (CDC), an estimated 30.3 million people in the United States—about 9.4% of the population—have been diagnosed with diabetes.1 Diabetes is the seventh leading cause of death in the United States, and it contributes to other leading causes of death: heart disease and stroke.1 Diabetes also is related to high morbidity risk and is a leading cause of chronic kidney disease.1 The total cost of diagnosed diabetes was estimated at $327 billion in direct medical costs and reduced productivity.2
Residents of Appalachia bear a disproportionate burden of diabetes and other related negative health outcomes; these outcomes are influenced by a number of factors, including socioeconomic status, poverty, rurality, and health care access. Rates of chronic disease, such as diabetes, are most pronounced in Appalachia’s most economically distressed counties.3-5 In 2011, the CDC labeled a 644-county area the “diabetes belt,” which included most of Appalachia.6 As a result of this elevated prevalence of diabetes in Appalachia as compared to the rest of the country, complications directly associated with diabetes are more commonly observed in Appalachian residents. One of the most damaging complications is diabetic nephropathy.
Diabetic nephropathy results from damage to the microvasculature of the kidney due to inadequately controlled blood glucose. This, in turn, leads to decreased renal function, eventually leading to clinically significant renal disease. The long-term complications associated with nephropathy can include many comorbid conditions, the most serious of which are progression to end-stage renal disease, dialysis requirement, and early mortality. Diabetic nephropathy affects approximately 40% of patients with type 1 and type 2 diabetes.7,8
One way to prevent complications of diabetic nephropathy, in addition to good glycemic control in patients with diabetes, is early and regular screening. Currently, the American Diabetes Association (ADA) recommends yearly screening for diabetic nephropathy in the form of a urine albumin-to-creatinine ratio (ACR) for patients 18 to 75 years of age.2 This screening to detect diabetic nephropathy is recognized as a marker of quality care by many public and private insurance agencies and medical specialty associations, such as the Centers for Medicare and Medicaid Services.
Many patients with diabetes are cared for by primary care providers (PCP), and these PCP appointments provide an opportune time to screen and appropriately treat nephropathy. Screening opportunities are often missed, however, due to time constraints and competing health priorities. There are also a number of other factors specific to the Appalachian region that reduce the likelihood of screening for diabetic nephropathy, such as a lack of health insurance, the need to travel long distances to see a PCP, work and household responsibilities, low levels of education and health literacy, and a mistrust of outsiders regarding personal matters, including health.9-11 While nephropathy can have a detrimental impact on patients across populations, it is of particular concern for a state located in the heart of Appalachia, such as West Virginia.
Given the disproportionate burden of diabetes in this region and the potentially severe consequences of undetected nephropathy, clinicians from an academic family medicine clinic in West Virginia undertook a quality improvement project to increase the rate of nephropathy screening and treatment among patients with diabetes. This article describes the intervention strategies the team utilized to increase nephropathy screening and treatment in patients 18 to 75 years of age who met quality measures for nephropathy screening or treatment in the previous 12 months and explores the factors most predictive of nephropathy screening in Appalachian patients in this age group. It also reports the challenges and opportunities encountered and offers suggestions for other providers and clinics attempting to increase their nephropathy screening rates.
Methods
Setting and Study Population
The study population included patients ages 18 to 75 years under the care of providers in an academic family medicine practice in West Virginia who had been diagnosed with diabetes mellitus. The study focused on those patients overdue for diabetic nephropathy screening (ie, had not been screened in previous 12 months). The project began in January 2018 with a screening rate of 83.8%. The goal of this project was to increase this compliance metric by at least 5%. The project protocol was submitted to the West Virginia University Institutional Review Board, and, because it is a quality improvement project, permission was given to proceed without a board review.
Interventions
The team identified and implemented several interventions intended to reduce screening barriers and increase the screening rate.
Bulk orders for ACR and urine collection during clinic visits. Prior to initiation of this project, it was left to individual clinic providers to order nephropathy screening for patients with diabetes during a clinic visit; after receiving the order for “random urine microalbumin/creatinine ratio,” patients then had to travel to a lab to provide a urine sample. For this project and moving forward, the team changed to the procedure of initiating bulk ACR orders and collecting urine samples during clinic visits from all patients ages 18 to 75 years who have diabetes.
Bulk communication reminders. Since many patients with diabetes may not have realized they were overdue for nephropathy screening, the team began sending out bulk communication reminders through either the institution’s electronic health record (EHR; MyChart) or postal service–delivered physical letters (according to patient communication preferences) to remind patients that they were due for screening and to encourage them to schedule an appointment or keep a previously scheduled appointment with their PCP.
Individual patient outreach. A team of pharmacy students led by a licensed pharmacist in the family medicine clinic contacted patients overdue for screening even after bulk communication reminders went out. The students telephoned patients 2 to 3 months following the bulk communication. The students obtained an updated list of patients with diabetes ages 18 to 75 years from an EHR quality report. They began by prescreening the patients on the overdue list for potential candidacy for an angiotensin-converting enzyme (ACE) inhibitor or an angiotensin II receptor blocker (ARB). Screening for candidacy included evaluation of recent blood pressure readings, electrolytes (ie, basic metabolic panel), and ACR. If the students determined a patient was a candidate, they presented the patient to the preceptor for verification and then reached out to the provider with a recommendation. If the provider agreed, the student contacted the patient by telephone for medication counseling and education. The remaining patients determined not to be candidates for ACE inhibitors or ARBs were contacted by the pharmacy students by telephone to remind them that laboratory work was pending. Up to 3 phone call attempts were made before patients were determined to be unreachable. Students left voice mails with generic reminders if a patient could not be reached. If a patient answered, the student provided a reminder but also reviewed indications for lab work, the reason why the provider wished for follow-up, and updated lab hours. Students also followed up with the results of the work-up, as appropriate. During this outreach process, the student team encountered a number of patients who had moved or changed to a PCP outside of the family medicine clinic. In these cases, the EHR was updated and those patients were removed from the list of patients altogether.
Education of clinic providers. Clinic providers were educated during faculty and resident meetings and didactic learning sessions on identifying patients within the EHR who are due for nephropathy screening. They also received instruction on how to update the EHR to reflect completed screenings.
Data Analysis
All analyses in this study were conducted using SAS (version 9.4, 2013, SAS Institute Inc., Cary, NC). Descriptive analyses were conducted to summarize basic patient demographic information. To compare patients screened within the previous 12 months to those patients overdue for screening, 2-sample t-tests were used to examine differences in patients’ age, HbA1c, ACR, and creatinine level and the distance (in miles) between the patient’s home and the clinic. Chi-square analyses were used to examine the relationship between whether a patient was recently screened for nephropathy and the patient’s insurance, number of patient visits in the previous 12 months, and provider level. Logistic regression analyses were conducted to control for covariates and to explore which factors were most predictive of nephropathy screening. All tests were 2-tailed, and P values less than 0.05 were considered statistically significant.
Results
Patient Characteristics
There were 1676 family medicine clinic patients with diabetes between 18 and 75 years of age (Table 1 and Table 2). Of the total sample, 1489 (88.8%) had completed screening for nephropathy in the 12 months prior to evaluation, and 67.5%, 23.7%, and 8.8% of patients had private insurance, Medicare, and Medicaid, respectively. 
The mean (SD) age of the patients was 56.3 (11.9) years. The mean distance between the patient’s home and the clinic was 26.6 (76.8) miles. The mean number of visits was 3.6 (2.9) per year, and 43.0% of the patientvisited the clinic more than 3 times in a year. The mean values for HbA1c (%), creatinine (g/mol), and ACR (mg/g) were 7.7 (1.9), 1.0 (0.7), and 9.4 (31.4), respectively.
Screening of Patients for Nephropathy
Patients with Medicare and private insurance were more likely to have completed the nephropathy screening than those with Medicaid (92.5% versus 88.8% versus 82.8%, P = 0.004; Table 3 and Table 4). 

Changes in Screening Rate
The practice-wide screening rate was 83.8% at the start of this project in January 2018. The screening rate steadily increased throughout 2018, reaching 90.3% in August 2018, and then leveled off around 90% when the project was concluded at the end of November 2018 (Figure). As an added benefit of the increased screening rates, a number of patients were initiated on an ACE inhibitor or ARB based on the team’s screening efforts.

Predictors of Nephropathy Screening
A logistic regression analysis was conducted with nephropathy screening (screened or not screened) as the outcome and 7 patient characteristics as predictors: type of insurance (private, Medicare, or Medicaid), PCP visits in the past 12 months (≤ 3 or > 3), distance in miles of the patient’s residence from the clinic, age, last HbA1c value, last ACR value, and last creatinine value. A test of the full model with all 7 predictors was statistically significant (χ2 (8) = 57.77, P < 0.001). Table 5 shows regression coefficients, Wald statistics, and 95% confidence intervals for odds ratios for each of the 7 predictors. According to the Wald criterion, 3 patient characteristics were significant predictors of nephropathy screening: age, distance between the patient’s home and clinic, and number of PCP visits in the past 12 months. After adjusting for the covariates, there were still significant associations between the nephropathy screening status and age ( χ2(1) = 9.64, P < 0.01); distance between the patient’s home and the clinic (χ2(1) = 3.98, P < 0.05); and the number of PCP visits in the previous year (χ2(1) = 21.74, P < 0.001). With each 1-year increment in age, the odds of completing the nephropathy screening increased by 3.2%. With each 1-mile increase in the distance between the patient’s home and clinic, the odds of completing the nephropathy screening decreased by 0.2%. Patients who visited the clinic more than 3 times in a year were 3.9 times (95% confidence interval, 2.2-7.0) more likely to complete the nephropathy screening than their counterparts who visited fewer than 3 times per year.
In summary, older patients living within about 164 miles of the clinic (ie, within 1 standard deviation from the average miles between patient’s homes and the clinic) who visited their PCP 3 or more times per year were the most likely to be screened.

Discussion
Diabetic nephropathy is a critical issue facing family medicine providers and patients. The morbidity and mortality costs are significant, as diabetic nephropathy is the leading cause of end-stage renal disease. While the ADA recommends annual ACR screening in patients with diabetes and prescription of ACE inhibitors or ARBs in patients who qualify, many patients do not receive these interventions, despite following up with a provider.12-15 There is no current literature that indicates the compliance rates in the rural setting. Due to health disparities in the rural setting noted in the literature, it could be hypothesized that these individuals are at high risk of not meeting these screening and treatment recommendations.16,17 Limited access to care and resources, gaps in insurance coverage, and lower health literacy are a few barriers identified in the rural population that may influence whether these measures are met.17
Considering the disease burden of diabetes and its related complications, including nephropathy, consistent screening is necessary to reduce diabetes-related burdens and cost, while also increasing the quality of life for patients with diabetes. All parties must be involved to ensure appropriate compliance and treatment. Our institution’s implementation of quality improvement strategies has key implications for nephropathy screening and treatment efforts in rural settings.
An additional step of having a health care provider (other than the PCP) screen all patients who are not meeting the standard allows for identification of gaps in care. In our quality improvement workflow, the clinical pharmacist screened all patients for candidacy for ACE inhibitor/ARB therapy. While only a small percentage of patients qualified, many of these patients had previously been on therapy and were discontinued for an unknown reason or were stopped due to an acute condition (eg, acute kidney injury) and never restarted after recovery. Other patients required additional education that therapy would be utilized for nephroprotection versus blood pressure management (secondary to an elevated ACR). This highlights the importance of transitions of care and ongoing, intensive education, not only during initial diagnosis but also throughout the disease-state progression.
Utilization of EHRs and telephone outreach are additional aspects of care that can be provided. Our improved rates of compliance with these care interventions parallel findings from previous studies.15,18 Optimization of an institution’s EHR can aid in standardization of care, workflow management, and communication with patients, as well as alert nursing or support staff of screening needs. Techniques such as best practice reminders, patient chart messages, and nursing-entered physician alerts on daily schedules have been shown to increase rates of compliance with nephropathy standards. These findings underscore an additional opportunity for nursing and support staff to be better integrated into care.
Despite the success of this quality improvement initiative, there remain some limitations. The processes we used in this project may not be applicable to every institution and may have limited external validity. Primarily, while these processes may be implemented at some sites, without additional support staff (ie, extra nursing staff, pharmacists) and students to aid in patient outreach, success may be limited due to provider time constraints. Additionally, our workflow process demonstrates significant incorporation of an EHR system for patient outreach. Institutions and/or clinics that heavily rely on paper charts and paper outreach may face barriers with bulk orders (eg, ACR) and messages, interventions that streamlined our population health management. Finally, this project focuses on only 1 aspect of population health management for patients with diabetes. While nephropathy is a critical aspect of caring for individuals with diabetes, this patient outreach does not address retinopathy screening, HbA1c control, or vaccination rates, which are other components of care.
Conclusion
Although this evaluation does not provide insight into why patients were not treated or screened, it demonstrates processes to improve compliance in patients with diabetic nephropathy. Rural health care facilities require an ongoing program of change and evaluation, with the aim to improve the provision of services, increase screening, and encourage team member involvement in health promotion. This study demonstrates that combining team-based interventions with quality control monitoring can significantly improve compliance with recommended nephropathy screening and treatment in rural patients with diabetes at a family medicine clinic.
Corresponding author: Amie M. Ashcraft, West Virginia University, Department of Family Medicine, 1 Medical Center Drive, Box 9152, Morgantown, WV 26506; amashcraft@hsc.wvu.edu.
Financial disclosures: None.
Acknowledgment: The authors thank the faculty, residents, nurses, and clinic staff for their hard work and dedication to this effort: Umama Sadia, Michelle Prestoza, Richard Dattola, Greg Doyle, Dana King, Mike Maroon, Kendra Under, Judy Siebert, Christine Snyder, Rachel Burge, Meagan Gribble, Lisa Metts, Kelsey Samek, Sarah Deavers, Amber Kitzmiller, Angela Lamp, Tina Waldeck, and Andrea Sukeruksa.
From West Virginia University, Morgantown, WV.
Abstract
Objective: To describe the strategies a family medicine clinic in Appalachia utilized to increase nephropathy screening rates as well as to explore the factors predictive of nephropathy screening in patients with diabetes.
Design: This quality improvement project targeted the points in the care process when patients are lost to follow-up for nephropathy screening.
Setting and participants: Patients with diabetes cared for by a primary care provider (PCP) at an academic family medicine practice in Appalachia from January 2018 to November 2018.
Interventions: Bulk orders for albumin-to-creatinine (ACR) testing and urine collection during clinic visit, enhanced patient communication through bulk communication reminders and individual patient outreach, and education of clinic providers.
Measurements: Demographic data and monthly nephropathy screening rates.
Results: The nephropathy screening rate increased by 6.2% during the project. Older patients living closer to the clinic who visited their PCP 3 or more times per year were the most likely to be screened.
Conclusion: Combining team-based interventions with quality control monitoring can significantly improve compliance with recommended nephropathy treatment and screening in rural patients with diabetes at a family medicine clinic.
Keywords: rural; kidney disease; albumin-to-creatinine ratio; electronic health record.
According to the Centers for Disease Control and Prevention (CDC), an estimated 30.3 million people in the United States—about 9.4% of the population—have been diagnosed with diabetes.1 Diabetes is the seventh leading cause of death in the United States, and it contributes to other leading causes of death: heart disease and stroke.1 Diabetes also is related to high morbidity risk and is a leading cause of chronic kidney disease.1 The total cost of diagnosed diabetes was estimated at $327 billion in direct medical costs and reduced productivity.2
Residents of Appalachia bear a disproportionate burden of diabetes and other related negative health outcomes; these outcomes are influenced by a number of factors, including socioeconomic status, poverty, rurality, and health care access. Rates of chronic disease, such as diabetes, are most pronounced in Appalachia’s most economically distressed counties.3-5 In 2011, the CDC labeled a 644-county area the “diabetes belt,” which included most of Appalachia.6 As a result of this elevated prevalence of diabetes in Appalachia as compared to the rest of the country, complications directly associated with diabetes are more commonly observed in Appalachian residents. One of the most damaging complications is diabetic nephropathy.
Diabetic nephropathy results from damage to the microvasculature of the kidney due to inadequately controlled blood glucose. This, in turn, leads to decreased renal function, eventually leading to clinically significant renal disease. The long-term complications associated with nephropathy can include many comorbid conditions, the most serious of which are progression to end-stage renal disease, dialysis requirement, and early mortality. Diabetic nephropathy affects approximately 40% of patients with type 1 and type 2 diabetes.7,8
One way to prevent complications of diabetic nephropathy, in addition to good glycemic control in patients with diabetes, is early and regular screening. Currently, the American Diabetes Association (ADA) recommends yearly screening for diabetic nephropathy in the form of a urine albumin-to-creatinine ratio (ACR) for patients 18 to 75 years of age.2 This screening to detect diabetic nephropathy is recognized as a marker of quality care by many public and private insurance agencies and medical specialty associations, such as the Centers for Medicare and Medicaid Services.
Many patients with diabetes are cared for by primary care providers (PCP), and these PCP appointments provide an opportune time to screen and appropriately treat nephropathy. Screening opportunities are often missed, however, due to time constraints and competing health priorities. There are also a number of other factors specific to the Appalachian region that reduce the likelihood of screening for diabetic nephropathy, such as a lack of health insurance, the need to travel long distances to see a PCP, work and household responsibilities, low levels of education and health literacy, and a mistrust of outsiders regarding personal matters, including health.9-11 While nephropathy can have a detrimental impact on patients across populations, it is of particular concern for a state located in the heart of Appalachia, such as West Virginia.
Given the disproportionate burden of diabetes in this region and the potentially severe consequences of undetected nephropathy, clinicians from an academic family medicine clinic in West Virginia undertook a quality improvement project to increase the rate of nephropathy screening and treatment among patients with diabetes. This article describes the intervention strategies the team utilized to increase nephropathy screening and treatment in patients 18 to 75 years of age who met quality measures for nephropathy screening or treatment in the previous 12 months and explores the factors most predictive of nephropathy screening in Appalachian patients in this age group. It also reports the challenges and opportunities encountered and offers suggestions for other providers and clinics attempting to increase their nephropathy screening rates.
Methods
Setting and Study Population
The study population included patients ages 18 to 75 years under the care of providers in an academic family medicine practice in West Virginia who had been diagnosed with diabetes mellitus. The study focused on those patients overdue for diabetic nephropathy screening (ie, had not been screened in previous 12 months). The project began in January 2018 with a screening rate of 83.8%. The goal of this project was to increase this compliance metric by at least 5%. The project protocol was submitted to the West Virginia University Institutional Review Board, and, because it is a quality improvement project, permission was given to proceed without a board review.
Interventions
The team identified and implemented several interventions intended to reduce screening barriers and increase the screening rate.
Bulk orders for ACR and urine collection during clinic visits. Prior to initiation of this project, it was left to individual clinic providers to order nephropathy screening for patients with diabetes during a clinic visit; after receiving the order for “random urine microalbumin/creatinine ratio,” patients then had to travel to a lab to provide a urine sample. For this project and moving forward, the team changed to the procedure of initiating bulk ACR orders and collecting urine samples during clinic visits from all patients ages 18 to 75 years who have diabetes.
Bulk communication reminders. Since many patients with diabetes may not have realized they were overdue for nephropathy screening, the team began sending out bulk communication reminders through either the institution’s electronic health record (EHR; MyChart) or postal service–delivered physical letters (according to patient communication preferences) to remind patients that they were due for screening and to encourage them to schedule an appointment or keep a previously scheduled appointment with their PCP.
Individual patient outreach. A team of pharmacy students led by a licensed pharmacist in the family medicine clinic contacted patients overdue for screening even after bulk communication reminders went out. The students telephoned patients 2 to 3 months following the bulk communication. The students obtained an updated list of patients with diabetes ages 18 to 75 years from an EHR quality report. They began by prescreening the patients on the overdue list for potential candidacy for an angiotensin-converting enzyme (ACE) inhibitor or an angiotensin II receptor blocker (ARB). Screening for candidacy included evaluation of recent blood pressure readings, electrolytes (ie, basic metabolic panel), and ACR. If the students determined a patient was a candidate, they presented the patient to the preceptor for verification and then reached out to the provider with a recommendation. If the provider agreed, the student contacted the patient by telephone for medication counseling and education. The remaining patients determined not to be candidates for ACE inhibitors or ARBs were contacted by the pharmacy students by telephone to remind them that laboratory work was pending. Up to 3 phone call attempts were made before patients were determined to be unreachable. Students left voice mails with generic reminders if a patient could not be reached. If a patient answered, the student provided a reminder but also reviewed indications for lab work, the reason why the provider wished for follow-up, and updated lab hours. Students also followed up with the results of the work-up, as appropriate. During this outreach process, the student team encountered a number of patients who had moved or changed to a PCP outside of the family medicine clinic. In these cases, the EHR was updated and those patients were removed from the list of patients altogether.
Education of clinic providers. Clinic providers were educated during faculty and resident meetings and didactic learning sessions on identifying patients within the EHR who are due for nephropathy screening. They also received instruction on how to update the EHR to reflect completed screenings.
Data Analysis
All analyses in this study were conducted using SAS (version 9.4, 2013, SAS Institute Inc., Cary, NC). Descriptive analyses were conducted to summarize basic patient demographic information. To compare patients screened within the previous 12 months to those patients overdue for screening, 2-sample t-tests were used to examine differences in patients’ age, HbA1c, ACR, and creatinine level and the distance (in miles) between the patient’s home and the clinic. Chi-square analyses were used to examine the relationship between whether a patient was recently screened for nephropathy and the patient’s insurance, number of patient visits in the previous 12 months, and provider level. Logistic regression analyses were conducted to control for covariates and to explore which factors were most predictive of nephropathy screening. All tests were 2-tailed, and P values less than 0.05 were considered statistically significant.
Results
Patient Characteristics
There were 1676 family medicine clinic patients with diabetes between 18 and 75 years of age (Table 1 and Table 2). Of the total sample, 1489 (88.8%) had completed screening for nephropathy in the 12 months prior to evaluation, and 67.5%, 23.7%, and 8.8% of patients had private insurance, Medicare, and Medicaid, respectively. 
The mean (SD) age of the patients was 56.3 (11.9) years. The mean distance between the patient’s home and the clinic was 26.6 (76.8) miles. The mean number of visits was 3.6 (2.9) per year, and 43.0% of the patientvisited the clinic more than 3 times in a year. The mean values for HbA1c (%), creatinine (g/mol), and ACR (mg/g) were 7.7 (1.9), 1.0 (0.7), and 9.4 (31.4), respectively.
Screening of Patients for Nephropathy
Patients with Medicare and private insurance were more likely to have completed the nephropathy screening than those with Medicaid (92.5% versus 88.8% versus 82.8%, P = 0.004; Table 3 and Table 4). 

Changes in Screening Rate
The practice-wide screening rate was 83.8% at the start of this project in January 2018. The screening rate steadily increased throughout 2018, reaching 90.3% in August 2018, and then leveled off around 90% when the project was concluded at the end of November 2018 (Figure). As an added benefit of the increased screening rates, a number of patients were initiated on an ACE inhibitor or ARB based on the team’s screening efforts.

Predictors of Nephropathy Screening
A logistic regression analysis was conducted with nephropathy screening (screened or not screened) as the outcome and 7 patient characteristics as predictors: type of insurance (private, Medicare, or Medicaid), PCP visits in the past 12 months (≤ 3 or > 3), distance in miles of the patient’s residence from the clinic, age, last HbA1c value, last ACR value, and last creatinine value. A test of the full model with all 7 predictors was statistically significant (χ2 (8) = 57.77, P < 0.001). Table 5 shows regression coefficients, Wald statistics, and 95% confidence intervals for odds ratios for each of the 7 predictors. According to the Wald criterion, 3 patient characteristics were significant predictors of nephropathy screening: age, distance between the patient’s home and clinic, and number of PCP visits in the past 12 months. After adjusting for the covariates, there were still significant associations between the nephropathy screening status and age ( χ2(1) = 9.64, P < 0.01); distance between the patient’s home and the clinic (χ2(1) = 3.98, P < 0.05); and the number of PCP visits in the previous year (χ2(1) = 21.74, P < 0.001). With each 1-year increment in age, the odds of completing the nephropathy screening increased by 3.2%. With each 1-mile increase in the distance between the patient’s home and clinic, the odds of completing the nephropathy screening decreased by 0.2%. Patients who visited the clinic more than 3 times in a year were 3.9 times (95% confidence interval, 2.2-7.0) more likely to complete the nephropathy screening than their counterparts who visited fewer than 3 times per year.
In summary, older patients living within about 164 miles of the clinic (ie, within 1 standard deviation from the average miles between patient’s homes and the clinic) who visited their PCP 3 or more times per year were the most likely to be screened.

Discussion
Diabetic nephropathy is a critical issue facing family medicine providers and patients. The morbidity and mortality costs are significant, as diabetic nephropathy is the leading cause of end-stage renal disease. While the ADA recommends annual ACR screening in patients with diabetes and prescription of ACE inhibitors or ARBs in patients who qualify, many patients do not receive these interventions, despite following up with a provider.12-15 There is no current literature that indicates the compliance rates in the rural setting. Due to health disparities in the rural setting noted in the literature, it could be hypothesized that these individuals are at high risk of not meeting these screening and treatment recommendations.16,17 Limited access to care and resources, gaps in insurance coverage, and lower health literacy are a few barriers identified in the rural population that may influence whether these measures are met.17
Considering the disease burden of diabetes and its related complications, including nephropathy, consistent screening is necessary to reduce diabetes-related burdens and cost, while also increasing the quality of life for patients with diabetes. All parties must be involved to ensure appropriate compliance and treatment. Our institution’s implementation of quality improvement strategies has key implications for nephropathy screening and treatment efforts in rural settings.
An additional step of having a health care provider (other than the PCP) screen all patients who are not meeting the standard allows for identification of gaps in care. In our quality improvement workflow, the clinical pharmacist screened all patients for candidacy for ACE inhibitor/ARB therapy. While only a small percentage of patients qualified, many of these patients had previously been on therapy and were discontinued for an unknown reason or were stopped due to an acute condition (eg, acute kidney injury) and never restarted after recovery. Other patients required additional education that therapy would be utilized for nephroprotection versus blood pressure management (secondary to an elevated ACR). This highlights the importance of transitions of care and ongoing, intensive education, not only during initial diagnosis but also throughout the disease-state progression.
Utilization of EHRs and telephone outreach are additional aspects of care that can be provided. Our improved rates of compliance with these care interventions parallel findings from previous studies.15,18 Optimization of an institution’s EHR can aid in standardization of care, workflow management, and communication with patients, as well as alert nursing or support staff of screening needs. Techniques such as best practice reminders, patient chart messages, and nursing-entered physician alerts on daily schedules have been shown to increase rates of compliance with nephropathy standards. These findings underscore an additional opportunity for nursing and support staff to be better integrated into care.
Despite the success of this quality improvement initiative, there remain some limitations. The processes we used in this project may not be applicable to every institution and may have limited external validity. Primarily, while these processes may be implemented at some sites, without additional support staff (ie, extra nursing staff, pharmacists) and students to aid in patient outreach, success may be limited due to provider time constraints. Additionally, our workflow process demonstrates significant incorporation of an EHR system for patient outreach. Institutions and/or clinics that heavily rely on paper charts and paper outreach may face barriers with bulk orders (eg, ACR) and messages, interventions that streamlined our population health management. Finally, this project focuses on only 1 aspect of population health management for patients with diabetes. While nephropathy is a critical aspect of caring for individuals with diabetes, this patient outreach does not address retinopathy screening, HbA1c control, or vaccination rates, which are other components of care.
Conclusion
Although this evaluation does not provide insight into why patients were not treated or screened, it demonstrates processes to improve compliance in patients with diabetic nephropathy. Rural health care facilities require an ongoing program of change and evaluation, with the aim to improve the provision of services, increase screening, and encourage team member involvement in health promotion. This study demonstrates that combining team-based interventions with quality control monitoring can significantly improve compliance with recommended nephropathy screening and treatment in rural patients with diabetes at a family medicine clinic.
Corresponding author: Amie M. Ashcraft, West Virginia University, Department of Family Medicine, 1 Medical Center Drive, Box 9152, Morgantown, WV 26506; amashcraft@hsc.wvu.edu.
Financial disclosures: None.
Acknowledgment: The authors thank the faculty, residents, nurses, and clinic staff for their hard work and dedication to this effort: Umama Sadia, Michelle Prestoza, Richard Dattola, Greg Doyle, Dana King, Mike Maroon, Kendra Under, Judy Siebert, Christine Snyder, Rachel Burge, Meagan Gribble, Lisa Metts, Kelsey Samek, Sarah Deavers, Amber Kitzmiller, Angela Lamp, Tina Waldeck, and Andrea Sukeruksa.
1. Centers for Disease Control and Prevention (CDC). National diabetes statistics report. Estimates of diabetes and its burden in the United States. Atlanta, GA: CDC; 2017www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed December 20, 2020.
2. American Diabetes Association (ADA). Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917-928.
3. Wood L. Trends in national and regional economic distress, 1960-2000. Washington, DC: Appalachian Regional Commission; 2005.
4. Barker L, Crespo R, Gerzoff RB, et al. Residence in a distressed county in Appalachia as a risk factor for diabetes, Behavioral Risk Factor Surveillance System, 2006-2007. Prev Chronic Dis. 2010;7:A104.
5. Barker L, Kirtland KA, Gregg E, et al. Geographic distribution of diagnosed diabetes in the United States: A diabetes belt. Am J Prev Med. 2011;40:434-439.
6. Gross JL, de Azevedo MJ, Silveiro SP, et al. Diabetic nephropathy: Diagnosis, prevention, and treatment. Diabetes Care. 2005;28:164-176.
7. United States Renal Data System (USRDS). Annual data report. Ann Arbor, MI: USRDS; 2018. www.usrds.org/2018/view/Default.aspx. Accessed December 20, 2020.
8. Halverson JA, Bichak G. Underlying socioeconomic factors influencing health disparities in the Appalachian region. Washington, DC: Appalachian Regional Commission; 2008.
9. Shell R, Tudiver F. Barriers to cancer screening by rural Appalachian primary care providers. J Rural Health. 2004;20:368-373.
10. Hatcher J, Dignan MB, Schoenberg N. How do rural health care providers and patients view barriers to colorectal cancer screening? Insights from Appalachian Kentucky. Nurs Clin North Am. 2011;46:181-192.
11. Scott S, McSpirit S. The suspicious, untrusting hillbilly in political-economic contexts: Stereotypes and social trust in the Appalachian coalfields. Pract Anthropol. 2014;36:42-46.
12. Kirkman MS, Williams SR, Caffrey HH, Marrero DG. Impact of a program to improve adherence to diabetes guidelines by primary care physicians. Diabetes Care. 2002;25:1946-1951.
13. Byun SH, Ma SH, Jun JK, et al. Screening for diabetic retinopathy and nephropathy in patients with diabetes: A nationwide survey in Korea. PLoS One. 2013;8:e62991.
14. Flood D, Garcia P, Douglas K, et al. Screening for chronic kidney disease in a community-based diabetes cohort in rural Guatemala: A cross-sectional study. BMJ Open. 2018;8:e019778.
15. Anabtawi A, Mathew LM. Improving compliance with screening of diabetic patients for microalbuminuria in primary care practice. ISRN Endocrinology. 2013:893913.
16. Tonks SA, Makwana S, Salanitro AH, et al. Quality of diabetes mellitus care by rural primary care physicians. J Rural Health. 2012;28:364-371.
17. Douthit N, Kiv S, Dwolatzky T, Biswas S. Exposing some important barriers to health care access in the rural USA. Public Health. 2015;129:611-620.
18. Weber V, Bloom F, Pierdon S, Wood C. Employing the electronic health record to improve diabetes care: a multifaceted intervention in an integrated delivery system. J Gen Intern Med. 2008;23:379-382.
1. Centers for Disease Control and Prevention (CDC). National diabetes statistics report. Estimates of diabetes and its burden in the United States. Atlanta, GA: CDC; 2017www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed December 20, 2020.
2. American Diabetes Association (ADA). Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917-928.
3. Wood L. Trends in national and regional economic distress, 1960-2000. Washington, DC: Appalachian Regional Commission; 2005.
4. Barker L, Crespo R, Gerzoff RB, et al. Residence in a distressed county in Appalachia as a risk factor for diabetes, Behavioral Risk Factor Surveillance System, 2006-2007. Prev Chronic Dis. 2010;7:A104.
5. Barker L, Kirtland KA, Gregg E, et al. Geographic distribution of diagnosed diabetes in the United States: A diabetes belt. Am J Prev Med. 2011;40:434-439.
6. Gross JL, de Azevedo MJ, Silveiro SP, et al. Diabetic nephropathy: Diagnosis, prevention, and treatment. Diabetes Care. 2005;28:164-176.
7. United States Renal Data System (USRDS). Annual data report. Ann Arbor, MI: USRDS; 2018. www.usrds.org/2018/view/Default.aspx. Accessed December 20, 2020.
8. Halverson JA, Bichak G. Underlying socioeconomic factors influencing health disparities in the Appalachian region. Washington, DC: Appalachian Regional Commission; 2008.
9. Shell R, Tudiver F. Barriers to cancer screening by rural Appalachian primary care providers. J Rural Health. 2004;20:368-373.
10. Hatcher J, Dignan MB, Schoenberg N. How do rural health care providers and patients view barriers to colorectal cancer screening? Insights from Appalachian Kentucky. Nurs Clin North Am. 2011;46:181-192.
11. Scott S, McSpirit S. The suspicious, untrusting hillbilly in political-economic contexts: Stereotypes and social trust in the Appalachian coalfields. Pract Anthropol. 2014;36:42-46.
12. Kirkman MS, Williams SR, Caffrey HH, Marrero DG. Impact of a program to improve adherence to diabetes guidelines by primary care physicians. Diabetes Care. 2002;25:1946-1951.
13. Byun SH, Ma SH, Jun JK, et al. Screening for diabetic retinopathy and nephropathy in patients with diabetes: A nationwide survey in Korea. PLoS One. 2013;8:e62991.
14. Flood D, Garcia P, Douglas K, et al. Screening for chronic kidney disease in a community-based diabetes cohort in rural Guatemala: A cross-sectional study. BMJ Open. 2018;8:e019778.
15. Anabtawi A, Mathew LM. Improving compliance with screening of diabetic patients for microalbuminuria in primary care practice. ISRN Endocrinology. 2013:893913.
16. Tonks SA, Makwana S, Salanitro AH, et al. Quality of diabetes mellitus care by rural primary care physicians. J Rural Health. 2012;28:364-371.
17. Douthit N, Kiv S, Dwolatzky T, Biswas S. Exposing some important barriers to health care access in the rural USA. Public Health. 2015;129:611-620.
18. Weber V, Bloom F, Pierdon S, Wood C. Employing the electronic health record to improve diabetes care: a multifaceted intervention in an integrated delivery system. J Gen Intern Med. 2008;23:379-382.
Noninjectable modes of insulin delivery coming of age
LOS ANGELES – Injections may be the most common way for patients with diabetes to take insulin, but other modes of delivery are coming of age.
George Grunberger, MD, chairman of the Grunberger Diabetes Institute in Bloomfield Township, Mich., said that at least seven different agents that are being studied for the oral delivery of biologics for diabetes.
He outlined several at the World Congress on Insulin Resistance, Diabetes & Cardiovascular Disease.
Oral insulin
ORMD-0801 from Oramed is an oral insulin capsule that prevents enzyme degradation and enhances intestinal absorption. Top-line, unpublished findings from a phase 2 study, which the company announced in November 2019, showed that ORMD-0801 significantly reduced hemoglobin A1c levels in patients with type 2 diabetes who were inadequately controlled on other standard-of-care drugs. ORMD-0801 dosed once daily reduced HbA1c by 0.60%, compared with 0.06% by placebo. “We’ll see when it’s going to wind up in the clinic,” Dr. Grunberger said. Oramed is also developing an oral glucagonlike peptide–1 analogue capsule, ORMD-0901, which has potential to be the first orally ingestible GLP-1 analogue.
Inhaled and absorbed insulin
Technosphere insulin (Affreza) is a novel inhalation powder for the treatment of diabetes that was developed by MannKind and approved by the Food and Drug Administration in 2014. Clinical studies have shown that Technosphere insulin delivers insulin with an ultrarapid pharmacokinetic profile that is different from all other insulin products, but similar to natural insulin release. “The idea was to develop a more patient-friendly device to deliver insulin directly into the lungs,” said Dr. Grunberger, who is also a clinical professor of internal medicine and molecular medicine and genetics at Wayne State University, Detroit. “When you inhale this into the lungs, there is one cell layer between the air sac and the circulation, so it works very quickly. The idea is to try to avoid injecting insulin to see if it helps. This is a prandial insulin – you inhale it before meals. The whole idea is that hopefully, you can reduce any fear of delayed postprandial hyperglycemia.”
In a randomized trial of 353 patients with inadequately controlled type 2 diabetes, those in the Technosphere insulin arm significantly reduced HbA1c by 0.8% from a baseline of 8.3%, compared with the placebo arm, which was reduced by 0.4% (P less than .0001; Diabetes Care. 2015;38[12]:2274-81). A greater number of patients treated with Technosphere insulin achieved an HbA1c of 7.0% or less, compared with placebo (38% vs. 19%; P = .0005). Dr. Grunberger noted that, in clinical trials lasting up to 2 years, patients treated with Technosphere insulin had a 40-mL greater decline from baseline in forced expiratory volume in 1 second (FEV1 ), compared with patients treated with comparator antidiabetes treatments. “But once you stop using the drug, FEV1 reverts to normal,” he said. “So, there does not appear to be lasting damage to your lungs and respiratory ability.”
In another development, Oral-Lyn from Generex Biotechnology, which delivers insulin through the oral mucosa, is being evaluated as a potential treatment option. In 2015, Generex partnered with the University of Toronto’s Center for Molecular Design and Preformulations to increase the bioavailability of insulin in the product and to reduce the number of sprays required to achieve effective prandial glucose control. In 2019, the company formed the NuGenerex Diabetes Research Center, which intended to accelerate the development of the reformulated Oral-Lyn-2, for type 2 diabetes, and Altsulin, for the treatment of type 1 diabetes. The programs are expected to initiate in the first quarter of 2020.
In the meantime, studies of intranasally delivered insulin continue to advance. “It works. It lowers glucose, but there is a whole slew of knowledge now about how it can also improve neurocognitive function,” Dr. Grunberger said.
Oral GLP-1 receptor agonists
Oral versions of glucagonlike peptide–1 (GLP-1) receptor agonists are also emerging as a treatment option. The FDA recently approved the first oral GLP-1 receptor agonist, semaglutide bound in the absorption enhancer sodium N‐(8‐[2‐hydroxybenzoyl] amino) caprylate (SNAC). According to data from manufacturer Novo Nordisk, SNAC facilitates local increase of pH, which leads to a higher solubility. SNAC interacts with cell membranes of gastric mucosa, facilitating absorption within 30 minutes, “so the drug can penetrate the mucosa without lasting damage,” Dr. Grunberger said. The SNAC effect is size dependent and fully reversible.
In PIONEER 3, researchers found that, in adults with type 2 diabetes uncontrolled with metformin with or without sulfonylurea, oral semaglutide at dosages of 7 and 14 mg/day resulted in significantly greater reductions in HbA1c over 26 weeks, compared with sitagliptin, but there was no significant benefit with the 3-mg/d dosage (JAMA. 2019;321[15]:1466-80). In PIONEER 4, researchers compared the efficacy and safety of oral semaglutide with subcutaneous liraglutide (Lancet. 2019;394[10192]:P39-50). “There was no difference in HbA1c effect between the two groups, but oral semaglutide beat out sitagliptin in terms of weight loss,” Dr. Grunberger said. “It’s going to be interesting to see what’s going to happen in the marketplace as the drug gets widely launched.”
Nasal glucagon
He closed out his presentation by discussing the July 2019 FDA approval of Eli Lilly’s nasal glucagon for severe hypoglycemia – the first such treatment that can be administered without an injection. The nasally administered dry powder, known as Baqsimi, is a welcome alternative to current glucagon kits, “which contain multiple components,” said Dr. Grunberger, who is also a past president of the American Association of Clinical Endocrinologists. An adult pivotal study showed that supraphysiologic levels of glucagon were achieved within 5 minutes with both nasal and intramuscular glucagon (Diabetes Care. 2016;39[2]:264-70). Headache and nasal symptoms occurred more frequently with nasal glucagon, but most were resolved within 1 day. In addition, nausea and vomiting occurred at similar frequencies with nasal and intramuscular glucacon, and most cases were resolved within 1 day.
Similar results were observed in a pediatric study of 48 patients with type 1 diabetes who were older than 4 years, (Diabetes Care. 2016;39[4]:555-62).
Dr. Grunberger disclosed that has research contracts with Medtronic and Eli Lilly, and that he serves on speakers bureaus of Eli Lilly, Janssen, Novo Nordisk, and Sanofi.
LOS ANGELES – Injections may be the most common way for patients with diabetes to take insulin, but other modes of delivery are coming of age.
George Grunberger, MD, chairman of the Grunberger Diabetes Institute in Bloomfield Township, Mich., said that at least seven different agents that are being studied for the oral delivery of biologics for diabetes.
He outlined several at the World Congress on Insulin Resistance, Diabetes & Cardiovascular Disease.
Oral insulin
ORMD-0801 from Oramed is an oral insulin capsule that prevents enzyme degradation and enhances intestinal absorption. Top-line, unpublished findings from a phase 2 study, which the company announced in November 2019, showed that ORMD-0801 significantly reduced hemoglobin A1c levels in patients with type 2 diabetes who were inadequately controlled on other standard-of-care drugs. ORMD-0801 dosed once daily reduced HbA1c by 0.60%, compared with 0.06% by placebo. “We’ll see when it’s going to wind up in the clinic,” Dr. Grunberger said. Oramed is also developing an oral glucagonlike peptide–1 analogue capsule, ORMD-0901, which has potential to be the first orally ingestible GLP-1 analogue.
Inhaled and absorbed insulin
Technosphere insulin (Affreza) is a novel inhalation powder for the treatment of diabetes that was developed by MannKind and approved by the Food and Drug Administration in 2014. Clinical studies have shown that Technosphere insulin delivers insulin with an ultrarapid pharmacokinetic profile that is different from all other insulin products, but similar to natural insulin release. “The idea was to develop a more patient-friendly device to deliver insulin directly into the lungs,” said Dr. Grunberger, who is also a clinical professor of internal medicine and molecular medicine and genetics at Wayne State University, Detroit. “When you inhale this into the lungs, there is one cell layer between the air sac and the circulation, so it works very quickly. The idea is to try to avoid injecting insulin to see if it helps. This is a prandial insulin – you inhale it before meals. The whole idea is that hopefully, you can reduce any fear of delayed postprandial hyperglycemia.”
In a randomized trial of 353 patients with inadequately controlled type 2 diabetes, those in the Technosphere insulin arm significantly reduced HbA1c by 0.8% from a baseline of 8.3%, compared with the placebo arm, which was reduced by 0.4% (P less than .0001; Diabetes Care. 2015;38[12]:2274-81). A greater number of patients treated with Technosphere insulin achieved an HbA1c of 7.0% or less, compared with placebo (38% vs. 19%; P = .0005). Dr. Grunberger noted that, in clinical trials lasting up to 2 years, patients treated with Technosphere insulin had a 40-mL greater decline from baseline in forced expiratory volume in 1 second (FEV1 ), compared with patients treated with comparator antidiabetes treatments. “But once you stop using the drug, FEV1 reverts to normal,” he said. “So, there does not appear to be lasting damage to your lungs and respiratory ability.”
In another development, Oral-Lyn from Generex Biotechnology, which delivers insulin through the oral mucosa, is being evaluated as a potential treatment option. In 2015, Generex partnered with the University of Toronto’s Center for Molecular Design and Preformulations to increase the bioavailability of insulin in the product and to reduce the number of sprays required to achieve effective prandial glucose control. In 2019, the company formed the NuGenerex Diabetes Research Center, which intended to accelerate the development of the reformulated Oral-Lyn-2, for type 2 diabetes, and Altsulin, for the treatment of type 1 diabetes. The programs are expected to initiate in the first quarter of 2020.
In the meantime, studies of intranasally delivered insulin continue to advance. “It works. It lowers glucose, but there is a whole slew of knowledge now about how it can also improve neurocognitive function,” Dr. Grunberger said.
Oral GLP-1 receptor agonists
Oral versions of glucagonlike peptide–1 (GLP-1) receptor agonists are also emerging as a treatment option. The FDA recently approved the first oral GLP-1 receptor agonist, semaglutide bound in the absorption enhancer sodium N‐(8‐[2‐hydroxybenzoyl] amino) caprylate (SNAC). According to data from manufacturer Novo Nordisk, SNAC facilitates local increase of pH, which leads to a higher solubility. SNAC interacts with cell membranes of gastric mucosa, facilitating absorption within 30 minutes, “so the drug can penetrate the mucosa without lasting damage,” Dr. Grunberger said. The SNAC effect is size dependent and fully reversible.
In PIONEER 3, researchers found that, in adults with type 2 diabetes uncontrolled with metformin with or without sulfonylurea, oral semaglutide at dosages of 7 and 14 mg/day resulted in significantly greater reductions in HbA1c over 26 weeks, compared with sitagliptin, but there was no significant benefit with the 3-mg/d dosage (JAMA. 2019;321[15]:1466-80). In PIONEER 4, researchers compared the efficacy and safety of oral semaglutide with subcutaneous liraglutide (Lancet. 2019;394[10192]:P39-50). “There was no difference in HbA1c effect between the two groups, but oral semaglutide beat out sitagliptin in terms of weight loss,” Dr. Grunberger said. “It’s going to be interesting to see what’s going to happen in the marketplace as the drug gets widely launched.”
Nasal glucagon
He closed out his presentation by discussing the July 2019 FDA approval of Eli Lilly’s nasal glucagon for severe hypoglycemia – the first such treatment that can be administered without an injection. The nasally administered dry powder, known as Baqsimi, is a welcome alternative to current glucagon kits, “which contain multiple components,” said Dr. Grunberger, who is also a past president of the American Association of Clinical Endocrinologists. An adult pivotal study showed that supraphysiologic levels of glucagon were achieved within 5 minutes with both nasal and intramuscular glucagon (Diabetes Care. 2016;39[2]:264-70). Headache and nasal symptoms occurred more frequently with nasal glucagon, but most were resolved within 1 day. In addition, nausea and vomiting occurred at similar frequencies with nasal and intramuscular glucacon, and most cases were resolved within 1 day.
Similar results were observed in a pediatric study of 48 patients with type 1 diabetes who were older than 4 years, (Diabetes Care. 2016;39[4]:555-62).
Dr. Grunberger disclosed that has research contracts with Medtronic and Eli Lilly, and that he serves on speakers bureaus of Eli Lilly, Janssen, Novo Nordisk, and Sanofi.
LOS ANGELES – Injections may be the most common way for patients with diabetes to take insulin, but other modes of delivery are coming of age.
George Grunberger, MD, chairman of the Grunberger Diabetes Institute in Bloomfield Township, Mich., said that at least seven different agents that are being studied for the oral delivery of biologics for diabetes.
He outlined several at the World Congress on Insulin Resistance, Diabetes & Cardiovascular Disease.
Oral insulin
ORMD-0801 from Oramed is an oral insulin capsule that prevents enzyme degradation and enhances intestinal absorption. Top-line, unpublished findings from a phase 2 study, which the company announced in November 2019, showed that ORMD-0801 significantly reduced hemoglobin A1c levels in patients with type 2 diabetes who were inadequately controlled on other standard-of-care drugs. ORMD-0801 dosed once daily reduced HbA1c by 0.60%, compared with 0.06% by placebo. “We’ll see when it’s going to wind up in the clinic,” Dr. Grunberger said. Oramed is also developing an oral glucagonlike peptide–1 analogue capsule, ORMD-0901, which has potential to be the first orally ingestible GLP-1 analogue.
Inhaled and absorbed insulin
Technosphere insulin (Affreza) is a novel inhalation powder for the treatment of diabetes that was developed by MannKind and approved by the Food and Drug Administration in 2014. Clinical studies have shown that Technosphere insulin delivers insulin with an ultrarapid pharmacokinetic profile that is different from all other insulin products, but similar to natural insulin release. “The idea was to develop a more patient-friendly device to deliver insulin directly into the lungs,” said Dr. Grunberger, who is also a clinical professor of internal medicine and molecular medicine and genetics at Wayne State University, Detroit. “When you inhale this into the lungs, there is one cell layer between the air sac and the circulation, so it works very quickly. The idea is to try to avoid injecting insulin to see if it helps. This is a prandial insulin – you inhale it before meals. The whole idea is that hopefully, you can reduce any fear of delayed postprandial hyperglycemia.”
In a randomized trial of 353 patients with inadequately controlled type 2 diabetes, those in the Technosphere insulin arm significantly reduced HbA1c by 0.8% from a baseline of 8.3%, compared with the placebo arm, which was reduced by 0.4% (P less than .0001; Diabetes Care. 2015;38[12]:2274-81). A greater number of patients treated with Technosphere insulin achieved an HbA1c of 7.0% or less, compared with placebo (38% vs. 19%; P = .0005). Dr. Grunberger noted that, in clinical trials lasting up to 2 years, patients treated with Technosphere insulin had a 40-mL greater decline from baseline in forced expiratory volume in 1 second (FEV1 ), compared with patients treated with comparator antidiabetes treatments. “But once you stop using the drug, FEV1 reverts to normal,” he said. “So, there does not appear to be lasting damage to your lungs and respiratory ability.”
In another development, Oral-Lyn from Generex Biotechnology, which delivers insulin through the oral mucosa, is being evaluated as a potential treatment option. In 2015, Generex partnered with the University of Toronto’s Center for Molecular Design and Preformulations to increase the bioavailability of insulin in the product and to reduce the number of sprays required to achieve effective prandial glucose control. In 2019, the company formed the NuGenerex Diabetes Research Center, which intended to accelerate the development of the reformulated Oral-Lyn-2, for type 2 diabetes, and Altsulin, for the treatment of type 1 diabetes. The programs are expected to initiate in the first quarter of 2020.
In the meantime, studies of intranasally delivered insulin continue to advance. “It works. It lowers glucose, but there is a whole slew of knowledge now about how it can also improve neurocognitive function,” Dr. Grunberger said.
Oral GLP-1 receptor agonists
Oral versions of glucagonlike peptide–1 (GLP-1) receptor agonists are also emerging as a treatment option. The FDA recently approved the first oral GLP-1 receptor agonist, semaglutide bound in the absorption enhancer sodium N‐(8‐[2‐hydroxybenzoyl] amino) caprylate (SNAC). According to data from manufacturer Novo Nordisk, SNAC facilitates local increase of pH, which leads to a higher solubility. SNAC interacts with cell membranes of gastric mucosa, facilitating absorption within 30 minutes, “so the drug can penetrate the mucosa without lasting damage,” Dr. Grunberger said. The SNAC effect is size dependent and fully reversible.
In PIONEER 3, researchers found that, in adults with type 2 diabetes uncontrolled with metformin with or without sulfonylurea, oral semaglutide at dosages of 7 and 14 mg/day resulted in significantly greater reductions in HbA1c over 26 weeks, compared with sitagliptin, but there was no significant benefit with the 3-mg/d dosage (JAMA. 2019;321[15]:1466-80). In PIONEER 4, researchers compared the efficacy and safety of oral semaglutide with subcutaneous liraglutide (Lancet. 2019;394[10192]:P39-50). “There was no difference in HbA1c effect between the two groups, but oral semaglutide beat out sitagliptin in terms of weight loss,” Dr. Grunberger said. “It’s going to be interesting to see what’s going to happen in the marketplace as the drug gets widely launched.”
Nasal glucagon
He closed out his presentation by discussing the July 2019 FDA approval of Eli Lilly’s nasal glucagon for severe hypoglycemia – the first such treatment that can be administered without an injection. The nasally administered dry powder, known as Baqsimi, is a welcome alternative to current glucagon kits, “which contain multiple components,” said Dr. Grunberger, who is also a past president of the American Association of Clinical Endocrinologists. An adult pivotal study showed that supraphysiologic levels of glucagon were achieved within 5 minutes with both nasal and intramuscular glucagon (Diabetes Care. 2016;39[2]:264-70). Headache and nasal symptoms occurred more frequently with nasal glucagon, but most were resolved within 1 day. In addition, nausea and vomiting occurred at similar frequencies with nasal and intramuscular glucacon, and most cases were resolved within 1 day.
Similar results were observed in a pediatric study of 48 patients with type 1 diabetes who were older than 4 years, (Diabetes Care. 2016;39[4]:555-62).
Dr. Grunberger disclosed that has research contracts with Medtronic and Eli Lilly, and that he serves on speakers bureaus of Eli Lilly, Janssen, Novo Nordisk, and Sanofi.
EXPERT ANALYSIS FROM WCIRDC 2019
Zoledronate promotes postdenosumab bone retention
Women with osteoporosis who received a single infusion of zoledronate after discontinuing denosumab (Prolia) maintained bone mineral density at both the lumbar spine and the total hip, based on data from 120 individuals.
Although denosumab is often prescribed for postmenopausal osteoporosis, its effects disappear when treatment ends, wrote Judith Everts-Graber, MD, of OsteoRheuma Bern (Switzerland), and colleagues. In addition, recent reports of increased fractures in osteoporotic women after denosumab discontinuation highlight the need for subsequent therapy, but no protocol has been established.
In a study published in the Journal of Bone and Mineral Research, the investigators reviewed data from women aged older than 48 years with postmenopausal osteoporosis who were treated with denosumab between Aug. 1, 2010, and March 31, 2019. The women received four or more injections of 60 mg denosumab administered at 6-month intervals, followed by a single infusion of 5 mg zoledronate 6 months after the final denosumab injection. Patients were evaluated using dual-energy x-ray absorptiometry and vertebral fracture assessment every 2 years after starting denosumab; the average duration of treatment was 3 years.
At an average of 2.5 years after discontinuing denosumab, women who received zoledronate retained 66% of bone mineral density (BMD) gains at the lumbar spine, 49% at the total hip, and 57% at the femoral neck. In addition, three patients developed symptomatic single vertebral fractures and four patients developed peripheral fractures between 1 and 3 years after their last denosumab injections, but none of these patients sustained multiple fractures.
All bone loss occurred within 18 months of denosumab discontinuation, and no significant differences appeared between patients with gains in BMD greater than or less than 9%.
The study findings were limited by several factors, including the retrospective design and the lack of a control group, the researchers noted. However, they collected data from 11 of 28 patients who did not follow the treatment recommendations and did not receive zoledronate after discontinuing denosumab. “As expected, BMD of the lumbar spine and total hip decreased to baseline,” they wrote. In addition, 2 of the 11 patients experienced multiple vertebral fractures.
A single 5-mg infusion of zoledronate “may be a promising step in identifying sequential long-term treatment strategies for osteoporosis,” the researchers concluded. “Nevertheless, each patient requires an individualized surveillance and treatment plan after denosumab discontinuation, including BMD assessment, evaluation of bone turnover markers and consideration of individual clinical risk factors, in particular prevalent fragility fractures.”
The study was funded by OsteoRheuma Bern. The researchers reported having no financial conflicts.
SOURCE: Everts-Graber J et al. J Bone Miner Res. 2020 Jan 28. doi: 10.1002/jbmr.3962.
Women with osteoporosis who received a single infusion of zoledronate after discontinuing denosumab (Prolia) maintained bone mineral density at both the lumbar spine and the total hip, based on data from 120 individuals.
Although denosumab is often prescribed for postmenopausal osteoporosis, its effects disappear when treatment ends, wrote Judith Everts-Graber, MD, of OsteoRheuma Bern (Switzerland), and colleagues. In addition, recent reports of increased fractures in osteoporotic women after denosumab discontinuation highlight the need for subsequent therapy, but no protocol has been established.
In a study published in the Journal of Bone and Mineral Research, the investigators reviewed data from women aged older than 48 years with postmenopausal osteoporosis who were treated with denosumab between Aug. 1, 2010, and March 31, 2019. The women received four or more injections of 60 mg denosumab administered at 6-month intervals, followed by a single infusion of 5 mg zoledronate 6 months after the final denosumab injection. Patients were evaluated using dual-energy x-ray absorptiometry and vertebral fracture assessment every 2 years after starting denosumab; the average duration of treatment was 3 years.
At an average of 2.5 years after discontinuing denosumab, women who received zoledronate retained 66% of bone mineral density (BMD) gains at the lumbar spine, 49% at the total hip, and 57% at the femoral neck. In addition, three patients developed symptomatic single vertebral fractures and four patients developed peripheral fractures between 1 and 3 years after their last denosumab injections, but none of these patients sustained multiple fractures.
All bone loss occurred within 18 months of denosumab discontinuation, and no significant differences appeared between patients with gains in BMD greater than or less than 9%.
The study findings were limited by several factors, including the retrospective design and the lack of a control group, the researchers noted. However, they collected data from 11 of 28 patients who did not follow the treatment recommendations and did not receive zoledronate after discontinuing denosumab. “As expected, BMD of the lumbar spine and total hip decreased to baseline,” they wrote. In addition, 2 of the 11 patients experienced multiple vertebral fractures.
A single 5-mg infusion of zoledronate “may be a promising step in identifying sequential long-term treatment strategies for osteoporosis,” the researchers concluded. “Nevertheless, each patient requires an individualized surveillance and treatment plan after denosumab discontinuation, including BMD assessment, evaluation of bone turnover markers and consideration of individual clinical risk factors, in particular prevalent fragility fractures.”
The study was funded by OsteoRheuma Bern. The researchers reported having no financial conflicts.
SOURCE: Everts-Graber J et al. J Bone Miner Res. 2020 Jan 28. doi: 10.1002/jbmr.3962.
Women with osteoporosis who received a single infusion of zoledronate after discontinuing denosumab (Prolia) maintained bone mineral density at both the lumbar spine and the total hip, based on data from 120 individuals.
Although denosumab is often prescribed for postmenopausal osteoporosis, its effects disappear when treatment ends, wrote Judith Everts-Graber, MD, of OsteoRheuma Bern (Switzerland), and colleagues. In addition, recent reports of increased fractures in osteoporotic women after denosumab discontinuation highlight the need for subsequent therapy, but no protocol has been established.
In a study published in the Journal of Bone and Mineral Research, the investigators reviewed data from women aged older than 48 years with postmenopausal osteoporosis who were treated with denosumab between Aug. 1, 2010, and March 31, 2019. The women received four or more injections of 60 mg denosumab administered at 6-month intervals, followed by a single infusion of 5 mg zoledronate 6 months after the final denosumab injection. Patients were evaluated using dual-energy x-ray absorptiometry and vertebral fracture assessment every 2 years after starting denosumab; the average duration of treatment was 3 years.
At an average of 2.5 years after discontinuing denosumab, women who received zoledronate retained 66% of bone mineral density (BMD) gains at the lumbar spine, 49% at the total hip, and 57% at the femoral neck. In addition, three patients developed symptomatic single vertebral fractures and four patients developed peripheral fractures between 1 and 3 years after their last denosumab injections, but none of these patients sustained multiple fractures.
All bone loss occurred within 18 months of denosumab discontinuation, and no significant differences appeared between patients with gains in BMD greater than or less than 9%.
The study findings were limited by several factors, including the retrospective design and the lack of a control group, the researchers noted. However, they collected data from 11 of 28 patients who did not follow the treatment recommendations and did not receive zoledronate after discontinuing denosumab. “As expected, BMD of the lumbar spine and total hip decreased to baseline,” they wrote. In addition, 2 of the 11 patients experienced multiple vertebral fractures.
A single 5-mg infusion of zoledronate “may be a promising step in identifying sequential long-term treatment strategies for osteoporosis,” the researchers concluded. “Nevertheless, each patient requires an individualized surveillance and treatment plan after denosumab discontinuation, including BMD assessment, evaluation of bone turnover markers and consideration of individual clinical risk factors, in particular prevalent fragility fractures.”
The study was funded by OsteoRheuma Bern. The researchers reported having no financial conflicts.
SOURCE: Everts-Graber J et al. J Bone Miner Res. 2020 Jan 28. doi: 10.1002/jbmr.3962.
FROM THE JOURNAL OF BONE AND MINERAL RESEARCH
Right hip and pelvic pain
A 65-year-old man with a history of remote colon cancer, peptic ulcer disease, gastroesophageal reflux disease (GERD), and bilateral knee replacements presented with right groin and hip pain of more than a year’s duration. The patient described his hip pain as aching and said that it had worsened over the previous 6 months, interfering with his sleep. He said the pain worsened following activity, and it briefly felt better following an intra-articular corticosteroid injection into his right hip. The patient denied recent trauma or fracture and said he had no scalp pain, hearing loss, or spinal tenderness. Physical examination showed limited range of motion of the right hip and mild tenderness to palpation. Laboratory values were within normal limits. X-rays of the pelvis (Figure 1A) and right hip (Figure 1B) were ordered.
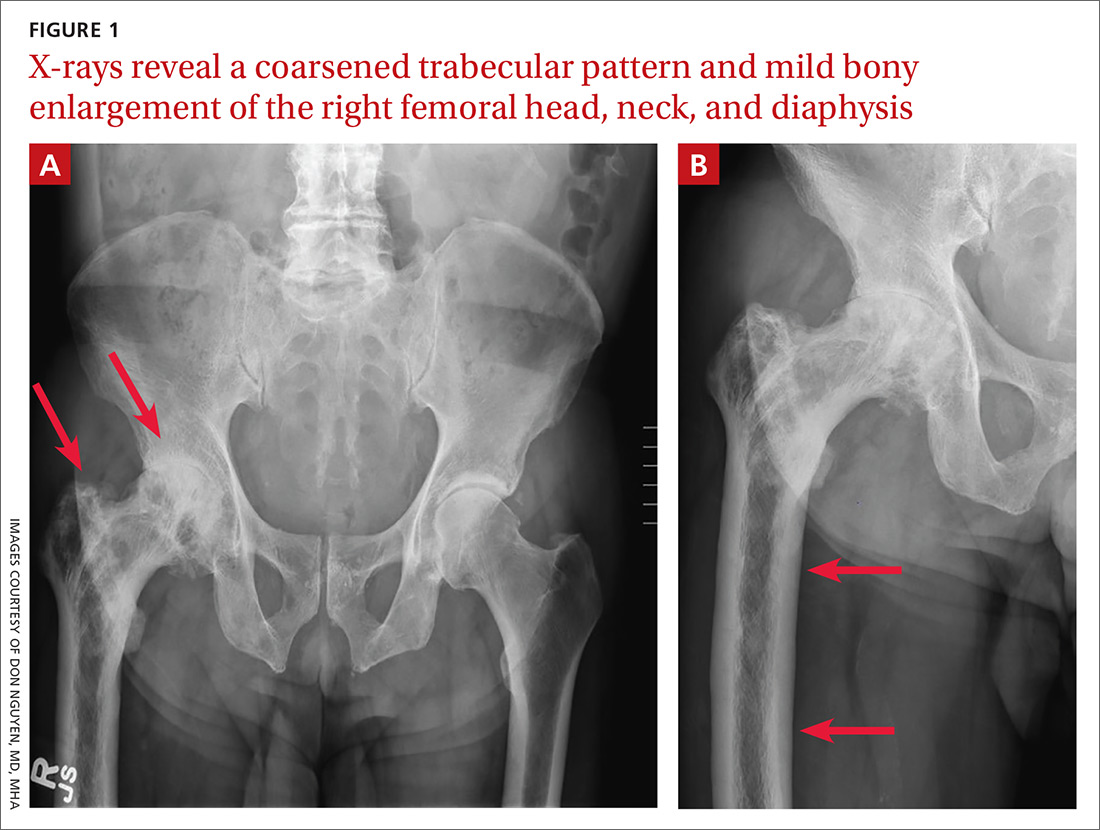
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Paget disease of bone
Based on the patient’s clinical history and initial imaging studies, which showed characteristic trabecular thickening with bony enlargement of the right femur, we suspected that he had Paget disease of bone. This was confirmed on subsequent whole-body 99mTc-MDP bone scan (Figure 2), which revealed corresponding diffuse increased radiotracer uptake of the right femur. There was no scintigraphic evidence of osseous involvement of the skull, spine, or pelvis.
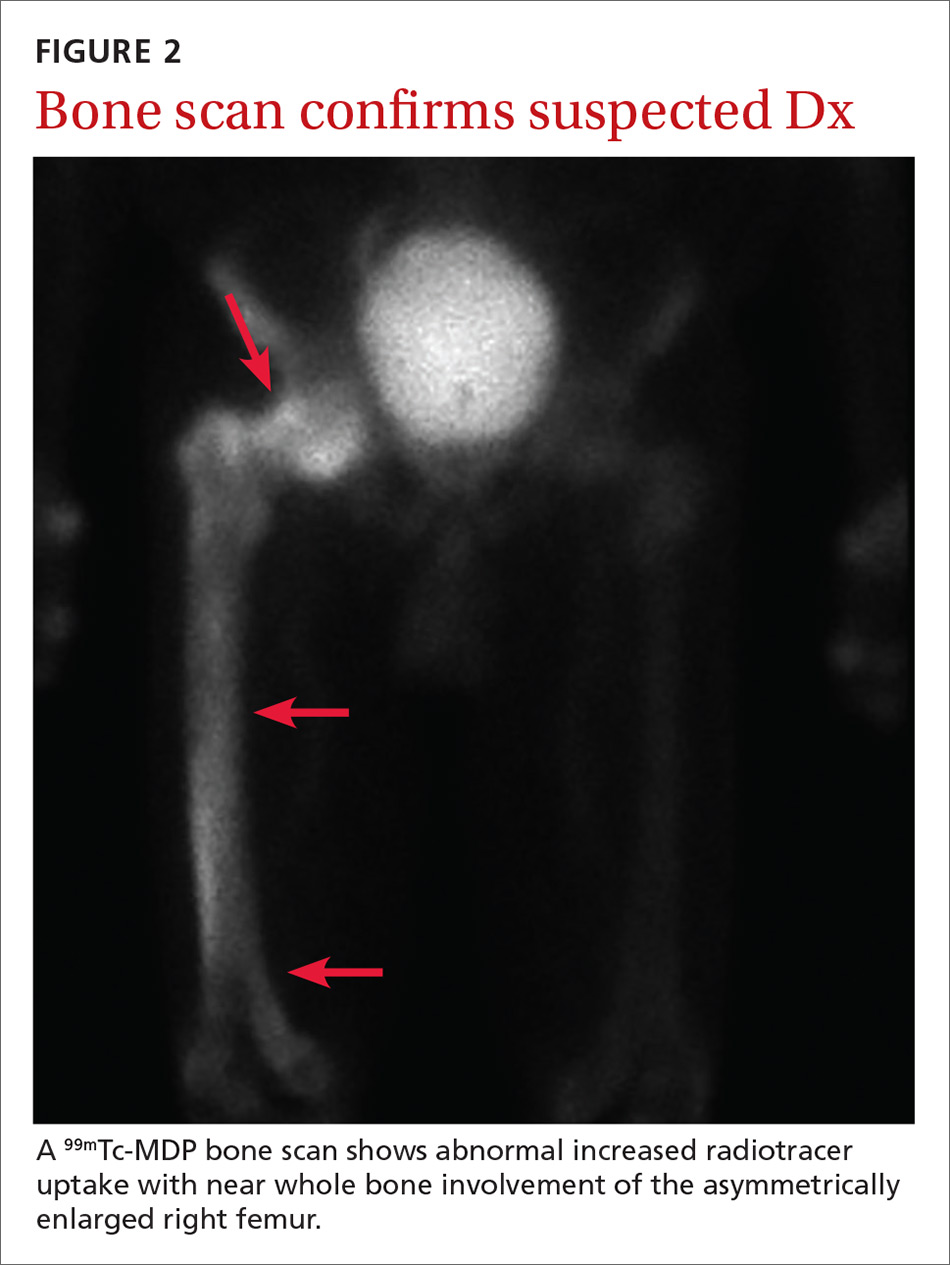
Epidemiology/incidence. Paget disease, also known as osteitis deformans, is fairly common in the aging population, with a prevalence ranging from 2% to almost 10%.1,2 Although onset before age 40 is rare, the diagnosis should be considered in younger patients, given the high prevalence. There is a slight male predominance, and the disease is more common in the United Kingdom and Western Europe, as well as in countries settled by European immigrants.3
Both genetic and environmental causes are believed to contribute to the pathogenesis of Paget disease. Mutations in the gene encoding sequestosome 1 (SQSTM1) can be seen in the autosomal dominant familial type (25%-50% of these cases), as well as in sporadic cases.4 Environmental influence has also been postulated as a possible cause, with a viral etiology (eg, chronic measles infection) being the most cited.5
Most patients will be asymptomatic
Paget disease can affect any bone in the body, although the skull, spine, pelvis, and long bones of the lower extremity are the most commonly affected sites.2 Most patients with Paget disease are asymptomatic. When symptoms are present, they either result from direct involvement of the bone or are secondary to bone overgrowth and deformity.
Direct involvement manifests as deep, constant bone pain that is worse at night. Symptoms related to bone overgrowth and deformity include spinal stenosis and related neurologic abnormalities, increased skull size, hearing loss (impingement of cranial nerve VIII), pathologic fracture (most commonly of the femur), and deformity such as protrusio acetabuli or femoral or tibial bowing.6 High-output heart failure and abnormalities in calcium and phosphate balance are uncommon but do occur.
Continue to: Degeneration into osteosarcoma...
Degeneration into osteosarcoma is a rare but almost invariably fatal complication of Paget disease, with an incidence of 0.2% to 1%.7 It clinically manifests as increased bone pain that is poorly responsive to medical therapy, local swelling, and pathologic fracture.8
Radiography is key to the work-up
The diagnosis of Paget disease is primarily radiographic. Early in the disease process, lytic lesions with thinning of the cortex will be noted. Later in the disease, there will be a mixed lytic/sclerotic phase, in which enlargement of the bone, a thickened cortex, and coarsened trabeculae are observed.
Characteristic radiographic findings. Focal lytic lesions in the skull are known as osteoporosis circumscripta. In the sclerotic phase, there is a thickening of the calvaria (termed “cotton wool”). Lesions involving the long bones will begin at the proximal or distal subchondral region and progress toward the diaphysis, with a sharp oblique delineation between involved bone and normal bone; this is described as “blade of grass” or “flame-shaped.”9
Within the pelvis, there will be cortical thickening and sclerosis with enlargement of the iliac wing. Within the spine, there will be enlarged vertebrae with a thickened sclerotic border, resulting in a “picture frame” appearance. Later in the disease, the sclerosis will involve the entire vertebrae (termed “ivory vertebra”).10
Additional testing options include magnetic resonance imaging (MRI), bone scintigraphy, laboratory testing, and biopsy.
Continue to: MRI is recommended...
MRI is recommended when degeneration into osteosarcoma is present—indicated by permeative lesions with cortical breakthrough and a soft-tissue mass. MRI is helpful to further characterize the lesion. Absence of the normal fatty marrow on T1-weighted images would be concerning for tumor involvement.
Bone scintigraphy is used to determine the extent of disease. It will show increased uptake when the lesions are active.
Laboratory testing. Serum alkaline phosphatase (sAP) is frequently elevated in patients with Paget disease (normal range, 20-140 IU/L) and reflects the extent and activity of disease. However, this correlation is not always reliable; it depends on monostotic vs polyostotic involvement, as well as which bones are involved. For example, sAP levels may be markedly elevated when the skull is involved but normal when other bones are involved.11 In patients with elevated sAP, serum calcium and 25-hydroxyvitamin D measurements should be obtained in anticipation of bisphosphonate treatment.
Biopsy. If the radiographic findings are typical for Paget disease, bone biopsy is not indicated. However, the main competing diagnosis to consider is malignancy; in atypical cases when imaging is unable to elucidate an underlying tumor, biopsy would be warranted.
Differentiating Paget disease from sclerotic metastasis is important. In metastasis, there will be no trabecular coarsening or enlargement of the bone.
Continue to: Bisphosphonates are a Tx mainstay
Bisphosphonates are a Tx mainstay
Indications for treatment include symptomatic or asymptomatic disease with any of the following: elevated sAP with pagetic changes at sites where complications could occur; sAP more than 2 to 4 times the upper limit of normal; normal sAP with abnormal bone scintigraphy at a site where complications could occur; planned surgery at an active pagetic site; and hypercalcemia in association with immobilization in patients with polyostotic disease.
Newer generation nitrogen-containing bisphosphonates are the mainstay of treatment; they ease pain, slow bone turnover, and promote deposition of normal lamellar bone, which over time will normalize sAP levels.12 The most frequently used and studied bisphosphonates include oral alendronate, oral risedronate, and intravenous zoledronic acid.13
Prior to treatment initiation, the patient should have documented normal serum levels of calcium, phosphorus, and 25-hydroxyvitamin D, and these levels should be monitored throughout the first year of treatment. All patients should receive supplemental vitamin D and calcium to avoid hypocalcemia. sAP should be measured at 3 to 6 months to assess the initial response to therapy. Once the levels equilibrate, sAP can be measured once or twice a year to asses bone activity.14
Our patient was referred to Endocrinology for management of Paget disease of his right hip and femur. Lab values, including sAP and liver function test results, were normal. The patient was prescribed a zoledronic acid infusion (Reclast). At 4-week follow-up, the patient reported moderate relief of bone pain and improved sleep.
CORRESPONDENCE
Don Nguyen, MD, MHA, Brigham and Women’s Hospital, Department of Radiology, 75 Francis Street, Boston, MA 02115; dnguyen42@bwh.harvard.edu
1. Altman RD, Bloch DA, Hochberg MC, et al. Prevalence of pelvic Paget’s disease of bone in the United States. J Bone Miner Res. 2000;15:461-465.
2. Singer F. Paget’s disease of bone. In: Feingold KR, Anawalt B, Boyce A, et al, eds. Endotext. South Dartmouth, MA: MDText.com, Inc.; 2000.
3. Merashli M, Jawad A. Paget’s disease of bone among various ethnic groups. Sultan Qaboos Univ Med J. 2015;15:E22-E26.
4. Hocking LJ, Lucas GJ, Daroszewska A, et al. Domain-specific mutations in sequestosome 1 (SQSTM1) cause familial and sporadic Paget’s disease. Hum Mol Genet. 2002;11:2735-2739.
5. Reddy SV, Kurihara N, Menaa C, et al. Osteoclasts formed by measles virus-infected osteoclast precursors from hCD46 transgenic mice express characteristics of pagetic osteoclasts. Endocrinology. 2001;142:2898-2905.
6. Moore TE, King AR, Kathol MH, et al. Sarcoma in Paget disease of bone: clinical, radiologic, and pathologic features in 22 cases. AJR Am J Roentgenol. 1991;156:1199-1203.
7. van Staa TP, Selby P, Leufkens HG, et al. Incidence and natural history of Paget’s disease of bone in England and Wales. J Bone Miner Res. 2002;17:465-471.
8. Hansen MF, Seton M, Merchant A. Osteosarcoma in Paget’s disease of bone. J Bone Miner Res. 2006;21(suppl 2):P58-P63.
9. Wittenberg K. The blade of grass sign. Radiology. 2001;221:199-200.
10. Dennis JM. The solitary dense vertebral body. Radiology. 1961;77:618-621.
11. Seton M. Paget’s disease of bone. In: Hochberg MC, Silman AJ, Smolen JS, et al, eds. Rheumatology. 4th ed. Philadelphia, PA: Mosby (Elsevier); 2008:2003.
12. Reid IR, Nicholson GC, Weinstein RS, et al. Biochemical and radiologic improvement in Paget’s disease of bone treated with alendronate: a randomized, placebo-controlled trial. Am J Med. 1996;101:341-348.
13. Siris ES, Lyles KW, Singer FR, et al. Medical management of Paget’s disease of bone: indications for treatment and review of current therapies. J Bone Miner Res. 2006;21(suppl 2):P94-P98.
14. Alvarez L, Peris P, Guañabens N, et al. Long-term biochemical response after bisphosphonate therapy in Paget’s disease of bone: proposed intervals for monitoring treatment. Rheumatology (Oxford). 2004;43:869-874.
A 65-year-old man with a history of remote colon cancer, peptic ulcer disease, gastroesophageal reflux disease (GERD), and bilateral knee replacements presented with right groin and hip pain of more than a year’s duration. The patient described his hip pain as aching and said that it had worsened over the previous 6 months, interfering with his sleep. He said the pain worsened following activity, and it briefly felt better following an intra-articular corticosteroid injection into his right hip. The patient denied recent trauma or fracture and said he had no scalp pain, hearing loss, or spinal tenderness. Physical examination showed limited range of motion of the right hip and mild tenderness to palpation. Laboratory values were within normal limits. X-rays of the pelvis (Figure 1A) and right hip (Figure 1B) were ordered.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Paget disease of bone
Based on the patient’s clinical history and initial imaging studies, which showed characteristic trabecular thickening with bony enlargement of the right femur, we suspected that he had Paget disease of bone. This was confirmed on subsequent whole-body 99mTc-MDP bone scan (Figure 2), which revealed corresponding diffuse increased radiotracer uptake of the right femur. There was no scintigraphic evidence of osseous involvement of the skull, spine, or pelvis.

Epidemiology/incidence. Paget disease, also known as osteitis deformans, is fairly common in the aging population, with a prevalence ranging from 2% to almost 10%.1,2 Although onset before age 40 is rare, the diagnosis should be considered in younger patients, given the high prevalence. There is a slight male predominance, and the disease is more common in the United Kingdom and Western Europe, as well as in countries settled by European immigrants.3
Both genetic and environmental causes are believed to contribute to the pathogenesis of Paget disease. Mutations in the gene encoding sequestosome 1 (SQSTM1) can be seen in the autosomal dominant familial type (25%-50% of these cases), as well as in sporadic cases.4 Environmental influence has also been postulated as a possible cause, with a viral etiology (eg, chronic measles infection) being the most cited.5
Most patients will be asymptomatic
Paget disease can affect any bone in the body, although the skull, spine, pelvis, and long bones of the lower extremity are the most commonly affected sites.2 Most patients with Paget disease are asymptomatic. When symptoms are present, they either result from direct involvement of the bone or are secondary to bone overgrowth and deformity.
Direct involvement manifests as deep, constant bone pain that is worse at night. Symptoms related to bone overgrowth and deformity include spinal stenosis and related neurologic abnormalities, increased skull size, hearing loss (impingement of cranial nerve VIII), pathologic fracture (most commonly of the femur), and deformity such as protrusio acetabuli or femoral or tibial bowing.6 High-output heart failure and abnormalities in calcium and phosphate balance are uncommon but do occur.
Continue to: Degeneration into osteosarcoma...
Degeneration into osteosarcoma is a rare but almost invariably fatal complication of Paget disease, with an incidence of 0.2% to 1%.7 It clinically manifests as increased bone pain that is poorly responsive to medical therapy, local swelling, and pathologic fracture.8
Radiography is key to the work-up
The diagnosis of Paget disease is primarily radiographic. Early in the disease process, lytic lesions with thinning of the cortex will be noted. Later in the disease, there will be a mixed lytic/sclerotic phase, in which enlargement of the bone, a thickened cortex, and coarsened trabeculae are observed.
Characteristic radiographic findings. Focal lytic lesions in the skull are known as osteoporosis circumscripta. In the sclerotic phase, there is a thickening of the calvaria (termed “cotton wool”). Lesions involving the long bones will begin at the proximal or distal subchondral region and progress toward the diaphysis, with a sharp oblique delineation between involved bone and normal bone; this is described as “blade of grass” or “flame-shaped.”9
Within the pelvis, there will be cortical thickening and sclerosis with enlargement of the iliac wing. Within the spine, there will be enlarged vertebrae with a thickened sclerotic border, resulting in a “picture frame” appearance. Later in the disease, the sclerosis will involve the entire vertebrae (termed “ivory vertebra”).10
Additional testing options include magnetic resonance imaging (MRI), bone scintigraphy, laboratory testing, and biopsy.
Continue to: MRI is recommended...
MRI is recommended when degeneration into osteosarcoma is present—indicated by permeative lesions with cortical breakthrough and a soft-tissue mass. MRI is helpful to further characterize the lesion. Absence of the normal fatty marrow on T1-weighted images would be concerning for tumor involvement.
Bone scintigraphy is used to determine the extent of disease. It will show increased uptake when the lesions are active.
Laboratory testing. Serum alkaline phosphatase (sAP) is frequently elevated in patients with Paget disease (normal range, 20-140 IU/L) and reflects the extent and activity of disease. However, this correlation is not always reliable; it depends on monostotic vs polyostotic involvement, as well as which bones are involved. For example, sAP levels may be markedly elevated when the skull is involved but normal when other bones are involved.11 In patients with elevated sAP, serum calcium and 25-hydroxyvitamin D measurements should be obtained in anticipation of bisphosphonate treatment.
Biopsy. If the radiographic findings are typical for Paget disease, bone biopsy is not indicated. However, the main competing diagnosis to consider is malignancy; in atypical cases when imaging is unable to elucidate an underlying tumor, biopsy would be warranted.
Differentiating Paget disease from sclerotic metastasis is important. In metastasis, there will be no trabecular coarsening or enlargement of the bone.
Continue to: Bisphosphonates are a Tx mainstay
Bisphosphonates are a Tx mainstay
Indications for treatment include symptomatic or asymptomatic disease with any of the following: elevated sAP with pagetic changes at sites where complications could occur; sAP more than 2 to 4 times the upper limit of normal; normal sAP with abnormal bone scintigraphy at a site where complications could occur; planned surgery at an active pagetic site; and hypercalcemia in association with immobilization in patients with polyostotic disease.
Newer generation nitrogen-containing bisphosphonates are the mainstay of treatment; they ease pain, slow bone turnover, and promote deposition of normal lamellar bone, which over time will normalize sAP levels.12 The most frequently used and studied bisphosphonates include oral alendronate, oral risedronate, and intravenous zoledronic acid.13
Prior to treatment initiation, the patient should have documented normal serum levels of calcium, phosphorus, and 25-hydroxyvitamin D, and these levels should be monitored throughout the first year of treatment. All patients should receive supplemental vitamin D and calcium to avoid hypocalcemia. sAP should be measured at 3 to 6 months to assess the initial response to therapy. Once the levels equilibrate, sAP can be measured once or twice a year to asses bone activity.14
Our patient was referred to Endocrinology for management of Paget disease of his right hip and femur. Lab values, including sAP and liver function test results, were normal. The patient was prescribed a zoledronic acid infusion (Reclast). At 4-week follow-up, the patient reported moderate relief of bone pain and improved sleep.
CORRESPONDENCE
Don Nguyen, MD, MHA, Brigham and Women’s Hospital, Department of Radiology, 75 Francis Street, Boston, MA 02115; dnguyen42@bwh.harvard.edu
A 65-year-old man with a history of remote colon cancer, peptic ulcer disease, gastroesophageal reflux disease (GERD), and bilateral knee replacements presented with right groin and hip pain of more than a year’s duration. The patient described his hip pain as aching and said that it had worsened over the previous 6 months, interfering with his sleep. He said the pain worsened following activity, and it briefly felt better following an intra-articular corticosteroid injection into his right hip. The patient denied recent trauma or fracture and said he had no scalp pain, hearing loss, or spinal tenderness. Physical examination showed limited range of motion of the right hip and mild tenderness to palpation. Laboratory values were within normal limits. X-rays of the pelvis (Figure 1A) and right hip (Figure 1B) were ordered.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Paget disease of bone
Based on the patient’s clinical history and initial imaging studies, which showed characteristic trabecular thickening with bony enlargement of the right femur, we suspected that he had Paget disease of bone. This was confirmed on subsequent whole-body 99mTc-MDP bone scan (Figure 2), which revealed corresponding diffuse increased radiotracer uptake of the right femur. There was no scintigraphic evidence of osseous involvement of the skull, spine, or pelvis.

Epidemiology/incidence. Paget disease, also known as osteitis deformans, is fairly common in the aging population, with a prevalence ranging from 2% to almost 10%.1,2 Although onset before age 40 is rare, the diagnosis should be considered in younger patients, given the high prevalence. There is a slight male predominance, and the disease is more common in the United Kingdom and Western Europe, as well as in countries settled by European immigrants.3
Both genetic and environmental causes are believed to contribute to the pathogenesis of Paget disease. Mutations in the gene encoding sequestosome 1 (SQSTM1) can be seen in the autosomal dominant familial type (25%-50% of these cases), as well as in sporadic cases.4 Environmental influence has also been postulated as a possible cause, with a viral etiology (eg, chronic measles infection) being the most cited.5
Most patients will be asymptomatic
Paget disease can affect any bone in the body, although the skull, spine, pelvis, and long bones of the lower extremity are the most commonly affected sites.2 Most patients with Paget disease are asymptomatic. When symptoms are present, they either result from direct involvement of the bone or are secondary to bone overgrowth and deformity.
Direct involvement manifests as deep, constant bone pain that is worse at night. Symptoms related to bone overgrowth and deformity include spinal stenosis and related neurologic abnormalities, increased skull size, hearing loss (impingement of cranial nerve VIII), pathologic fracture (most commonly of the femur), and deformity such as protrusio acetabuli or femoral or tibial bowing.6 High-output heart failure and abnormalities in calcium and phosphate balance are uncommon but do occur.
Continue to: Degeneration into osteosarcoma...
Degeneration into osteosarcoma is a rare but almost invariably fatal complication of Paget disease, with an incidence of 0.2% to 1%.7 It clinically manifests as increased bone pain that is poorly responsive to medical therapy, local swelling, and pathologic fracture.8
Radiography is key to the work-up
The diagnosis of Paget disease is primarily radiographic. Early in the disease process, lytic lesions with thinning of the cortex will be noted. Later in the disease, there will be a mixed lytic/sclerotic phase, in which enlargement of the bone, a thickened cortex, and coarsened trabeculae are observed.
Characteristic radiographic findings. Focal lytic lesions in the skull are known as osteoporosis circumscripta. In the sclerotic phase, there is a thickening of the calvaria (termed “cotton wool”). Lesions involving the long bones will begin at the proximal or distal subchondral region and progress toward the diaphysis, with a sharp oblique delineation between involved bone and normal bone; this is described as “blade of grass” or “flame-shaped.”9
Within the pelvis, there will be cortical thickening and sclerosis with enlargement of the iliac wing. Within the spine, there will be enlarged vertebrae with a thickened sclerotic border, resulting in a “picture frame” appearance. Later in the disease, the sclerosis will involve the entire vertebrae (termed “ivory vertebra”).10
Additional testing options include magnetic resonance imaging (MRI), bone scintigraphy, laboratory testing, and biopsy.
Continue to: MRI is recommended...
MRI is recommended when degeneration into osteosarcoma is present—indicated by permeative lesions with cortical breakthrough and a soft-tissue mass. MRI is helpful to further characterize the lesion. Absence of the normal fatty marrow on T1-weighted images would be concerning for tumor involvement.
Bone scintigraphy is used to determine the extent of disease. It will show increased uptake when the lesions are active.
Laboratory testing. Serum alkaline phosphatase (sAP) is frequently elevated in patients with Paget disease (normal range, 20-140 IU/L) and reflects the extent and activity of disease. However, this correlation is not always reliable; it depends on monostotic vs polyostotic involvement, as well as which bones are involved. For example, sAP levels may be markedly elevated when the skull is involved but normal when other bones are involved.11 In patients with elevated sAP, serum calcium and 25-hydroxyvitamin D measurements should be obtained in anticipation of bisphosphonate treatment.
Biopsy. If the radiographic findings are typical for Paget disease, bone biopsy is not indicated. However, the main competing diagnosis to consider is malignancy; in atypical cases when imaging is unable to elucidate an underlying tumor, biopsy would be warranted.
Differentiating Paget disease from sclerotic metastasis is important. In metastasis, there will be no trabecular coarsening or enlargement of the bone.
Continue to: Bisphosphonates are a Tx mainstay
Bisphosphonates are a Tx mainstay
Indications for treatment include symptomatic or asymptomatic disease with any of the following: elevated sAP with pagetic changes at sites where complications could occur; sAP more than 2 to 4 times the upper limit of normal; normal sAP with abnormal bone scintigraphy at a site where complications could occur; planned surgery at an active pagetic site; and hypercalcemia in association with immobilization in patients with polyostotic disease.
Newer generation nitrogen-containing bisphosphonates are the mainstay of treatment; they ease pain, slow bone turnover, and promote deposition of normal lamellar bone, which over time will normalize sAP levels.12 The most frequently used and studied bisphosphonates include oral alendronate, oral risedronate, and intravenous zoledronic acid.13
Prior to treatment initiation, the patient should have documented normal serum levels of calcium, phosphorus, and 25-hydroxyvitamin D, and these levels should be monitored throughout the first year of treatment. All patients should receive supplemental vitamin D and calcium to avoid hypocalcemia. sAP should be measured at 3 to 6 months to assess the initial response to therapy. Once the levels equilibrate, sAP can be measured once or twice a year to asses bone activity.14
Our patient was referred to Endocrinology for management of Paget disease of his right hip and femur. Lab values, including sAP and liver function test results, were normal. The patient was prescribed a zoledronic acid infusion (Reclast). At 4-week follow-up, the patient reported moderate relief of bone pain and improved sleep.
CORRESPONDENCE
Don Nguyen, MD, MHA, Brigham and Women’s Hospital, Department of Radiology, 75 Francis Street, Boston, MA 02115; dnguyen42@bwh.harvard.edu
1. Altman RD, Bloch DA, Hochberg MC, et al. Prevalence of pelvic Paget’s disease of bone in the United States. J Bone Miner Res. 2000;15:461-465.
2. Singer F. Paget’s disease of bone. In: Feingold KR, Anawalt B, Boyce A, et al, eds. Endotext. South Dartmouth, MA: MDText.com, Inc.; 2000.
3. Merashli M, Jawad A. Paget’s disease of bone among various ethnic groups. Sultan Qaboos Univ Med J. 2015;15:E22-E26.
4. Hocking LJ, Lucas GJ, Daroszewska A, et al. Domain-specific mutations in sequestosome 1 (SQSTM1) cause familial and sporadic Paget’s disease. Hum Mol Genet. 2002;11:2735-2739.
5. Reddy SV, Kurihara N, Menaa C, et al. Osteoclasts formed by measles virus-infected osteoclast precursors from hCD46 transgenic mice express characteristics of pagetic osteoclasts. Endocrinology. 2001;142:2898-2905.
6. Moore TE, King AR, Kathol MH, et al. Sarcoma in Paget disease of bone: clinical, radiologic, and pathologic features in 22 cases. AJR Am J Roentgenol. 1991;156:1199-1203.
7. van Staa TP, Selby P, Leufkens HG, et al. Incidence and natural history of Paget’s disease of bone in England and Wales. J Bone Miner Res. 2002;17:465-471.
8. Hansen MF, Seton M, Merchant A. Osteosarcoma in Paget’s disease of bone. J Bone Miner Res. 2006;21(suppl 2):P58-P63.
9. Wittenberg K. The blade of grass sign. Radiology. 2001;221:199-200.
10. Dennis JM. The solitary dense vertebral body. Radiology. 1961;77:618-621.
11. Seton M. Paget’s disease of bone. In: Hochberg MC, Silman AJ, Smolen JS, et al, eds. Rheumatology. 4th ed. Philadelphia, PA: Mosby (Elsevier); 2008:2003.
12. Reid IR, Nicholson GC, Weinstein RS, et al. Biochemical and radiologic improvement in Paget’s disease of bone treated with alendronate: a randomized, placebo-controlled trial. Am J Med. 1996;101:341-348.
13. Siris ES, Lyles KW, Singer FR, et al. Medical management of Paget’s disease of bone: indications for treatment and review of current therapies. J Bone Miner Res. 2006;21(suppl 2):P94-P98.
14. Alvarez L, Peris P, Guañabens N, et al. Long-term biochemical response after bisphosphonate therapy in Paget’s disease of bone: proposed intervals for monitoring treatment. Rheumatology (Oxford). 2004;43:869-874.
1. Altman RD, Bloch DA, Hochberg MC, et al. Prevalence of pelvic Paget’s disease of bone in the United States. J Bone Miner Res. 2000;15:461-465.
2. Singer F. Paget’s disease of bone. In: Feingold KR, Anawalt B, Boyce A, et al, eds. Endotext. South Dartmouth, MA: MDText.com, Inc.; 2000.
3. Merashli M, Jawad A. Paget’s disease of bone among various ethnic groups. Sultan Qaboos Univ Med J. 2015;15:E22-E26.
4. Hocking LJ, Lucas GJ, Daroszewska A, et al. Domain-specific mutations in sequestosome 1 (SQSTM1) cause familial and sporadic Paget’s disease. Hum Mol Genet. 2002;11:2735-2739.
5. Reddy SV, Kurihara N, Menaa C, et al. Osteoclasts formed by measles virus-infected osteoclast precursors from hCD46 transgenic mice express characteristics of pagetic osteoclasts. Endocrinology. 2001;142:2898-2905.
6. Moore TE, King AR, Kathol MH, et al. Sarcoma in Paget disease of bone: clinical, radiologic, and pathologic features in 22 cases. AJR Am J Roentgenol. 1991;156:1199-1203.
7. van Staa TP, Selby P, Leufkens HG, et al. Incidence and natural history of Paget’s disease of bone in England and Wales. J Bone Miner Res. 2002;17:465-471.
8. Hansen MF, Seton M, Merchant A. Osteosarcoma in Paget’s disease of bone. J Bone Miner Res. 2006;21(suppl 2):P58-P63.
9. Wittenberg K. The blade of grass sign. Radiology. 2001;221:199-200.
10. Dennis JM. The solitary dense vertebral body. Radiology. 1961;77:618-621.
11. Seton M. Paget’s disease of bone. In: Hochberg MC, Silman AJ, Smolen JS, et al, eds. Rheumatology. 4th ed. Philadelphia, PA: Mosby (Elsevier); 2008:2003.
12. Reid IR, Nicholson GC, Weinstein RS, et al. Biochemical and radiologic improvement in Paget’s disease of bone treated with alendronate: a randomized, placebo-controlled trial. Am J Med. 1996;101:341-348.
13. Siris ES, Lyles KW, Singer FR, et al. Medical management of Paget’s disease of bone: indications for treatment and review of current therapies. J Bone Miner Res. 2006;21(suppl 2):P94-P98.
14. Alvarez L, Peris P, Guañabens N, et al. Long-term biochemical response after bisphosphonate therapy in Paget’s disease of bone: proposed intervals for monitoring treatment. Rheumatology (Oxford). 2004;43:869-874.
33-year-old man • flaccid paralysis in limbs • 30-lb weight loss • thyromegaly without nodules • Dx?
THE CASE
A 33-year-old Hispanic man with no significant past medical history presented to the emergency department with generalized flaccid paralysis in both arms and legs. Two days before, he had been working on a construction site in hot weather. The following day, he woke up with very little energy or strength to perform his daily activities, and he had pain in the inguinal area and both calves. He denied taking any medications or supplements.
The patient had complete muscle weakness and was unable to move his arms and legs. He reported dysphagia and an unintentional weight loss of 30 lb during the previous month.
On physical examination, the patient’s vital signs were within the normal range, and mild thyromegaly without nodules was present. Neurologic examination revealed decreased deep tendon reflexes with intact sensation. Muscle strength in his arms and legs was 0/5.
Initial laboratory test results included a potassium level of 2.2 mEq/L (normal range, 3.5–5 mEq/L) and normal acid-basic status that was confirmed by an arterial blood gas measurement. Serum magnesium was 1.6 mg/dL (normal range, 1.6–2.5 mg/dL); phosphorus, 1.9 mg/dL (normal range, 2.7–4.5 mg/dL); and random urinary potassium, 16 mEq/L (normal range, 25–125 mEq/L). An initial chest x-ray was normal, and an electrocardiogram showed a prolonged QT interval, flattening of the T wave, and a prominent U wave consistent with hypokalemia.
THE DIAGNOSIS
The initial clinical diagnosis was hypokalemic paralysis. The patient was treated with intravenous (IV) potassium chloride 40 mEq
Evaluation of the patient’s hypokalemia revealed the following: thyroid-stimulating hormone (TSH) level, < 0.01 microIU/mL (normal range, 0.27–4.2 microIU/mL); free T4 (thyroxine) level, 4.47 ng/dL (normal range, 0.08–1.70 ng/dL); total T3 (triiodothyronine) level, 17.5 ng/dL (normal range, 2.6–4.4 ng/dL).
The patient was diagnosed with hypokalemic periodic paralysis (HPP) secondary to thyrotoxicosis, also known as thyrotoxicosis periodic paralysis (TPP). His hyperthyroidism was treated with oral atenolol 25 mg/d and oral methimazole 10 mg tid.
Continue to: Within a few hours...
Within a few hours of this treatment, the patient experienced significant improvement in muscle strength and complete resolution of weakness in his arms and legs. Serial measurements of potassium levels normalized.
Further workup revealed that the patient’s thyroid-stimulating immunoglobulin (TSI) was 4.2 on the TSI index (normal, ≤ 1.3) and his thyroid peroxidase (TPO) antibody level was 133.4 IU/mL (normal, < 34 IU/mL). Ultrasonography showed decreased echogenicity of the thyroid gland, consistent with the acute phase of Hashimoto thyroiditis or Graves disease.
The patient was unaware that he had any thyroid disorder previously. He was a private-pay, undocumented immigrant and did not have a regular primary care physician. On discharge, he was referred to a local primary care physician as well as an endocrinologist. He was discharged on atenolol and methimazole.
DISCUSSION
A rare neuromuscular disorder known as periodic paralysis can be precipitated by a hypokalemic or hyperkalemic state; HPP is more common and can be either familial (a defect in the gene) or acquired (secondary to thyrotoxicosis; TPP).1,2 In both forms of periodic paralysis, patients present with hypokalemia and paralysis. Physicians need to look closely at thyroid lab test results so as not to miss the cause of the paralysis.
TPP is most commonly seen in Asian populations, and 95% of cases reported occur in males, despite the higher incidence of hyperthyroidism in females.3 TPP can be precipitated by emotional stress, steroid use, beta-adrenergic bronchodilators, heavy exercise, fasting, or high-carbohydrate meals.2-4 In our patient, heavy exercise and fasting likely were the triggers.
Continue to: The pathophysiology for the hypokalemia...
The pathophysiology for the hypokalemia in TPP is thought to involve the sodium/potassium–adenosine triphosphatase (Na+/K+–ATPase) pump. This pump activity is increased in skeletal muscle and platelets in patients with TPP vs patients with thyrotoxicosis alone.3,5
The role of Hashimoto thyrotoxicosis. Most acquired cases of TPP are mainly secondary to Graves disease with elevated levels of TSI and mildly elevated or normal levels of TPO. In this case, the patient was in the acute phase of Hashimoto thyrotoxicosis (“hashitoxicosis”) with elevated levels of TPO and only mildly elevated TSI.Imaging studies to support the diagnosis, such as a thyroid uptake scan or ultrasonography, are not necessary to determine the cause of thyrotoxicosis. In the absence of test results for TPO and TSI antibodies, however, a scan can be helpful.6,7
Treatment of TPP consists of early recognition and supportive management by correcting the potassium deficit; failure to do so could cause severe complications, such as respiratory failure and psychosis.8 Because of the risk for rebound hyperkalemia, serial potassium levels must be measured until a stable potassium level in the normal range is achieved.
Nonselective beta-blockers, such as propranolol (3 mg/kg) 4 times per day, have been reported to ameliorate the periodic paralysis and prevent rebound hyperkalemia.9 Finally, restoring a euthyroid state will prevent the patient from experiencing future attacks.
THE TAKEAWAY
Few medical conditions result in complete muscle paralysis in a matter of hours. Clinicians should consider the possibility of TPP in any patient who presents with acute onset of paralysis.
CORRESPONDENCE
Jorge Luis Chavez, MD; 8405 E. San Pedro Drive, Scottsdale, AZ 85258; jorgeluischavezmd@yahoo.com.
1. Fontaine B. Periodic paralysis. Adv Genet. 2008;63:3-23.
2. Ober KP. Thyrotoxic periodic paralysis in the United States. Report of 7 cases and review of the literature. Medicine (Baltimore).1992;71:109-120.
3. Lin YF, Wu CC, Pei D, et al. Diagnosing thyrotoxic periodic paralysis in the ED. Am J Emerg Med. 2003;21:339-342.
4. Yu TS, Tseng CF, Chuang YY, et al. Potassium chloride supplementation alone may not improve hypokalemia in thyrotoxic hypokalemic periodic paralysis. J Emerg Med. 2007;32:263-265.
5. Chan A, Shinde R, Chow CC, et al. In vivo and in vitro sodium pump activity in subjects with thyrotoxic periodic paralysis. BMJ. 1991;303:1096-1099.
6. Harsch IA, Hahn EG, Strobel D. Hashitoxicosis—three cases and a review of the literature. Eur Endocrinol. 2008;4:70-72. 7. Pou Ucha JL. Imaging in hyperthyroidism. In: Díaz-Soto G, ed. Thyroid Disorders: Focus on Hyperthyroidism. InTechOpen; 2014. www.intechopen.com/books/thyroid-disorders-focus-on-hyperthyroidism/imaging-in-hyperthyroidism. Accessed January 14, 2020.
8. Abbasi B, Sharif Z, Sprabery LR. Hypokalemic thyrotoxic periodic paralysis with thyrotoxic psychosis and hypercapnic respiratory failure. Am J Med Sci. 2010;340:147-153.
9. Lin SH, Lin YF. Propranolol rapidly reverses paralysis, hypokalemia, and hypophosphatemia in thyrotoxic periodic paralysis. Am J Kidney Dis. 2001;37:620-623.
THE CASE
A 33-year-old Hispanic man with no significant past medical history presented to the emergency department with generalized flaccid paralysis in both arms and legs. Two days before, he had been working on a construction site in hot weather. The following day, he woke up with very little energy or strength to perform his daily activities, and he had pain in the inguinal area and both calves. He denied taking any medications or supplements.
The patient had complete muscle weakness and was unable to move his arms and legs. He reported dysphagia and an unintentional weight loss of 30 lb during the previous month.
On physical examination, the patient’s vital signs were within the normal range, and mild thyromegaly without nodules was present. Neurologic examination revealed decreased deep tendon reflexes with intact sensation. Muscle strength in his arms and legs was 0/5.
Initial laboratory test results included a potassium level of 2.2 mEq/L (normal range, 3.5–5 mEq/L) and normal acid-basic status that was confirmed by an arterial blood gas measurement. Serum magnesium was 1.6 mg/dL (normal range, 1.6–2.5 mg/dL); phosphorus, 1.9 mg/dL (normal range, 2.7–4.5 mg/dL); and random urinary potassium, 16 mEq/L (normal range, 25–125 mEq/L). An initial chest x-ray was normal, and an electrocardiogram showed a prolonged QT interval, flattening of the T wave, and a prominent U wave consistent with hypokalemia.
THE DIAGNOSIS
The initial clinical diagnosis was hypokalemic paralysis. The patient was treated with intravenous (IV) potassium chloride 40 mEq
Evaluation of the patient’s hypokalemia revealed the following: thyroid-stimulating hormone (TSH) level, < 0.01 microIU/mL (normal range, 0.27–4.2 microIU/mL); free T4 (thyroxine) level, 4.47 ng/dL (normal range, 0.08–1.70 ng/dL); total T3 (triiodothyronine) level, 17.5 ng/dL (normal range, 2.6–4.4 ng/dL).
The patient was diagnosed with hypokalemic periodic paralysis (HPP) secondary to thyrotoxicosis, also known as thyrotoxicosis periodic paralysis (TPP). His hyperthyroidism was treated with oral atenolol 25 mg/d and oral methimazole 10 mg tid.
Continue to: Within a few hours...
Within a few hours of this treatment, the patient experienced significant improvement in muscle strength and complete resolution of weakness in his arms and legs. Serial measurements of potassium levels normalized.
Further workup revealed that the patient’s thyroid-stimulating immunoglobulin (TSI) was 4.2 on the TSI index (normal, ≤ 1.3) and his thyroid peroxidase (TPO) antibody level was 133.4 IU/mL (normal, < 34 IU/mL). Ultrasonography showed decreased echogenicity of the thyroid gland, consistent with the acute phase of Hashimoto thyroiditis or Graves disease.
The patient was unaware that he had any thyroid disorder previously. He was a private-pay, undocumented immigrant and did not have a regular primary care physician. On discharge, he was referred to a local primary care physician as well as an endocrinologist. He was discharged on atenolol and methimazole.
DISCUSSION
A rare neuromuscular disorder known as periodic paralysis can be precipitated by a hypokalemic or hyperkalemic state; HPP is more common and can be either familial (a defect in the gene) or acquired (secondary to thyrotoxicosis; TPP).1,2 In both forms of periodic paralysis, patients present with hypokalemia and paralysis. Physicians need to look closely at thyroid lab test results so as not to miss the cause of the paralysis.
TPP is most commonly seen in Asian populations, and 95% of cases reported occur in males, despite the higher incidence of hyperthyroidism in females.3 TPP can be precipitated by emotional stress, steroid use, beta-adrenergic bronchodilators, heavy exercise, fasting, or high-carbohydrate meals.2-4 In our patient, heavy exercise and fasting likely were the triggers.
Continue to: The pathophysiology for the hypokalemia...
The pathophysiology for the hypokalemia in TPP is thought to involve the sodium/potassium–adenosine triphosphatase (Na+/K+–ATPase) pump. This pump activity is increased in skeletal muscle and platelets in patients with TPP vs patients with thyrotoxicosis alone.3,5
The role of Hashimoto thyrotoxicosis. Most acquired cases of TPP are mainly secondary to Graves disease with elevated levels of TSI and mildly elevated or normal levels of TPO. In this case, the patient was in the acute phase of Hashimoto thyrotoxicosis (“hashitoxicosis”) with elevated levels of TPO and only mildly elevated TSI.Imaging studies to support the diagnosis, such as a thyroid uptake scan or ultrasonography, are not necessary to determine the cause of thyrotoxicosis. In the absence of test results for TPO and TSI antibodies, however, a scan can be helpful.6,7
Treatment of TPP consists of early recognition and supportive management by correcting the potassium deficit; failure to do so could cause severe complications, such as respiratory failure and psychosis.8 Because of the risk for rebound hyperkalemia, serial potassium levels must be measured until a stable potassium level in the normal range is achieved.
Nonselective beta-blockers, such as propranolol (3 mg/kg) 4 times per day, have been reported to ameliorate the periodic paralysis and prevent rebound hyperkalemia.9 Finally, restoring a euthyroid state will prevent the patient from experiencing future attacks.
THE TAKEAWAY
Few medical conditions result in complete muscle paralysis in a matter of hours. Clinicians should consider the possibility of TPP in any patient who presents with acute onset of paralysis.
CORRESPONDENCE
Jorge Luis Chavez, MD; 8405 E. San Pedro Drive, Scottsdale, AZ 85258; jorgeluischavezmd@yahoo.com.
THE CASE
A 33-year-old Hispanic man with no significant past medical history presented to the emergency department with generalized flaccid paralysis in both arms and legs. Two days before, he had been working on a construction site in hot weather. The following day, he woke up with very little energy or strength to perform his daily activities, and he had pain in the inguinal area and both calves. He denied taking any medications or supplements.
The patient had complete muscle weakness and was unable to move his arms and legs. He reported dysphagia and an unintentional weight loss of 30 lb during the previous month.
On physical examination, the patient’s vital signs were within the normal range, and mild thyromegaly without nodules was present. Neurologic examination revealed decreased deep tendon reflexes with intact sensation. Muscle strength in his arms and legs was 0/5.
Initial laboratory test results included a potassium level of 2.2 mEq/L (normal range, 3.5–5 mEq/L) and normal acid-basic status that was confirmed by an arterial blood gas measurement. Serum magnesium was 1.6 mg/dL (normal range, 1.6–2.5 mg/dL); phosphorus, 1.9 mg/dL (normal range, 2.7–4.5 mg/dL); and random urinary potassium, 16 mEq/L (normal range, 25–125 mEq/L). An initial chest x-ray was normal, and an electrocardiogram showed a prolonged QT interval, flattening of the T wave, and a prominent U wave consistent with hypokalemia.
THE DIAGNOSIS
The initial clinical diagnosis was hypokalemic paralysis. The patient was treated with intravenous (IV) potassium chloride 40 mEq
Evaluation of the patient’s hypokalemia revealed the following: thyroid-stimulating hormone (TSH) level, < 0.01 microIU/mL (normal range, 0.27–4.2 microIU/mL); free T4 (thyroxine) level, 4.47 ng/dL (normal range, 0.08–1.70 ng/dL); total T3 (triiodothyronine) level, 17.5 ng/dL (normal range, 2.6–4.4 ng/dL).
The patient was diagnosed with hypokalemic periodic paralysis (HPP) secondary to thyrotoxicosis, also known as thyrotoxicosis periodic paralysis (TPP). His hyperthyroidism was treated with oral atenolol 25 mg/d and oral methimazole 10 mg tid.
Continue to: Within a few hours...
Within a few hours of this treatment, the patient experienced significant improvement in muscle strength and complete resolution of weakness in his arms and legs. Serial measurements of potassium levels normalized.
Further workup revealed that the patient’s thyroid-stimulating immunoglobulin (TSI) was 4.2 on the TSI index (normal, ≤ 1.3) and his thyroid peroxidase (TPO) antibody level was 133.4 IU/mL (normal, < 34 IU/mL). Ultrasonography showed decreased echogenicity of the thyroid gland, consistent with the acute phase of Hashimoto thyroiditis or Graves disease.
The patient was unaware that he had any thyroid disorder previously. He was a private-pay, undocumented immigrant and did not have a regular primary care physician. On discharge, he was referred to a local primary care physician as well as an endocrinologist. He was discharged on atenolol and methimazole.
DISCUSSION
A rare neuromuscular disorder known as periodic paralysis can be precipitated by a hypokalemic or hyperkalemic state; HPP is more common and can be either familial (a defect in the gene) or acquired (secondary to thyrotoxicosis; TPP).1,2 In both forms of periodic paralysis, patients present with hypokalemia and paralysis. Physicians need to look closely at thyroid lab test results so as not to miss the cause of the paralysis.
TPP is most commonly seen in Asian populations, and 95% of cases reported occur in males, despite the higher incidence of hyperthyroidism in females.3 TPP can be precipitated by emotional stress, steroid use, beta-adrenergic bronchodilators, heavy exercise, fasting, or high-carbohydrate meals.2-4 In our patient, heavy exercise and fasting likely were the triggers.
Continue to: The pathophysiology for the hypokalemia...
The pathophysiology for the hypokalemia in TPP is thought to involve the sodium/potassium–adenosine triphosphatase (Na+/K+–ATPase) pump. This pump activity is increased in skeletal muscle and platelets in patients with TPP vs patients with thyrotoxicosis alone.3,5
The role of Hashimoto thyrotoxicosis. Most acquired cases of TPP are mainly secondary to Graves disease with elevated levels of TSI and mildly elevated or normal levels of TPO. In this case, the patient was in the acute phase of Hashimoto thyrotoxicosis (“hashitoxicosis”) with elevated levels of TPO and only mildly elevated TSI.Imaging studies to support the diagnosis, such as a thyroid uptake scan or ultrasonography, are not necessary to determine the cause of thyrotoxicosis. In the absence of test results for TPO and TSI antibodies, however, a scan can be helpful.6,7
Treatment of TPP consists of early recognition and supportive management by correcting the potassium deficit; failure to do so could cause severe complications, such as respiratory failure and psychosis.8 Because of the risk for rebound hyperkalemia, serial potassium levels must be measured until a stable potassium level in the normal range is achieved.
Nonselective beta-blockers, such as propranolol (3 mg/kg) 4 times per day, have been reported to ameliorate the periodic paralysis and prevent rebound hyperkalemia.9 Finally, restoring a euthyroid state will prevent the patient from experiencing future attacks.
THE TAKEAWAY
Few medical conditions result in complete muscle paralysis in a matter of hours. Clinicians should consider the possibility of TPP in any patient who presents with acute onset of paralysis.
CORRESPONDENCE
Jorge Luis Chavez, MD; 8405 E. San Pedro Drive, Scottsdale, AZ 85258; jorgeluischavezmd@yahoo.com.
1. Fontaine B. Periodic paralysis. Adv Genet. 2008;63:3-23.
2. Ober KP. Thyrotoxic periodic paralysis in the United States. Report of 7 cases and review of the literature. Medicine (Baltimore).1992;71:109-120.
3. Lin YF, Wu CC, Pei D, et al. Diagnosing thyrotoxic periodic paralysis in the ED. Am J Emerg Med. 2003;21:339-342.
4. Yu TS, Tseng CF, Chuang YY, et al. Potassium chloride supplementation alone may not improve hypokalemia in thyrotoxic hypokalemic periodic paralysis. J Emerg Med. 2007;32:263-265.
5. Chan A, Shinde R, Chow CC, et al. In vivo and in vitro sodium pump activity in subjects with thyrotoxic periodic paralysis. BMJ. 1991;303:1096-1099.
6. Harsch IA, Hahn EG, Strobel D. Hashitoxicosis—three cases and a review of the literature. Eur Endocrinol. 2008;4:70-72. 7. Pou Ucha JL. Imaging in hyperthyroidism. In: Díaz-Soto G, ed. Thyroid Disorders: Focus on Hyperthyroidism. InTechOpen; 2014. www.intechopen.com/books/thyroid-disorders-focus-on-hyperthyroidism/imaging-in-hyperthyroidism. Accessed January 14, 2020.
8. Abbasi B, Sharif Z, Sprabery LR. Hypokalemic thyrotoxic periodic paralysis with thyrotoxic psychosis and hypercapnic respiratory failure. Am J Med Sci. 2010;340:147-153.
9. Lin SH, Lin YF. Propranolol rapidly reverses paralysis, hypokalemia, and hypophosphatemia in thyrotoxic periodic paralysis. Am J Kidney Dis. 2001;37:620-623.
1. Fontaine B. Periodic paralysis. Adv Genet. 2008;63:3-23.
2. Ober KP. Thyrotoxic periodic paralysis in the United States. Report of 7 cases and review of the literature. Medicine (Baltimore).1992;71:109-120.
3. Lin YF, Wu CC, Pei D, et al. Diagnosing thyrotoxic periodic paralysis in the ED. Am J Emerg Med. 2003;21:339-342.
4. Yu TS, Tseng CF, Chuang YY, et al. Potassium chloride supplementation alone may not improve hypokalemia in thyrotoxic hypokalemic periodic paralysis. J Emerg Med. 2007;32:263-265.
5. Chan A, Shinde R, Chow CC, et al. In vivo and in vitro sodium pump activity in subjects with thyrotoxic periodic paralysis. BMJ. 1991;303:1096-1099.
6. Harsch IA, Hahn EG, Strobel D. Hashitoxicosis—three cases and a review of the literature. Eur Endocrinol. 2008;4:70-72. 7. Pou Ucha JL. Imaging in hyperthyroidism. In: Díaz-Soto G, ed. Thyroid Disorders: Focus on Hyperthyroidism. InTechOpen; 2014. www.intechopen.com/books/thyroid-disorders-focus-on-hyperthyroidism/imaging-in-hyperthyroidism. Accessed January 14, 2020.
8. Abbasi B, Sharif Z, Sprabery LR. Hypokalemic thyrotoxic periodic paralysis with thyrotoxic psychosis and hypercapnic respiratory failure. Am J Med Sci. 2010;340:147-153.
9. Lin SH, Lin YF. Propranolol rapidly reverses paralysis, hypokalemia, and hypophosphatemia in thyrotoxic periodic paralysis. Am J Kidney Dis. 2001;37:620-623.
Teprotumumab gets FDA go-ahead for thyroid eye disease
according to a press release.
Thyroid eye disease is a rare, progressive, autoimmune condition that causes the eyes to bulge (proptosis) and can lead to blindness. Until now, treatment has focused on managing its symptoms – which can include eye pain, double vision, or sensitivity to light – with steroids, and in some cases, multiple invasive surgeries.
The human monoclonal antibody and a targeted inhibitor of the insulinlike growth factor-1 receptor is administered to patients once every 3 weeks, for a total of eight infusions, according to a statement from Horizon Therapeutics, which manufactures the drug.
The approval was based on the findings from two similarly designed, parallel-group studies (Studies 1 and 2) involving 170 patients with thyroid eye disease who were randomized to receive either teprotumumab or placebo. Of those receiving the study drug, 71% in Study 1 and 83% in Study 2 had a reduction of more than 2 mm in eye protrusion, compared with 20% and 10%, respectively, among the placebo participants.
The most common adverse reactions in patients receiving teprotumumab were muscle spasm, nausea, alopecia, diarrhea, fatigue, and hyperglycemia. The treatment is contraindicated for pregnancy.
according to a press release.
Thyroid eye disease is a rare, progressive, autoimmune condition that causes the eyes to bulge (proptosis) and can lead to blindness. Until now, treatment has focused on managing its symptoms – which can include eye pain, double vision, or sensitivity to light – with steroids, and in some cases, multiple invasive surgeries.
The human monoclonal antibody and a targeted inhibitor of the insulinlike growth factor-1 receptor is administered to patients once every 3 weeks, for a total of eight infusions, according to a statement from Horizon Therapeutics, which manufactures the drug.
The approval was based on the findings from two similarly designed, parallel-group studies (Studies 1 and 2) involving 170 patients with thyroid eye disease who were randomized to receive either teprotumumab or placebo. Of those receiving the study drug, 71% in Study 1 and 83% in Study 2 had a reduction of more than 2 mm in eye protrusion, compared with 20% and 10%, respectively, among the placebo participants.
The most common adverse reactions in patients receiving teprotumumab were muscle spasm, nausea, alopecia, diarrhea, fatigue, and hyperglycemia. The treatment is contraindicated for pregnancy.
according to a press release.
Thyroid eye disease is a rare, progressive, autoimmune condition that causes the eyes to bulge (proptosis) and can lead to blindness. Until now, treatment has focused on managing its symptoms – which can include eye pain, double vision, or sensitivity to light – with steroids, and in some cases, multiple invasive surgeries.
The human monoclonal antibody and a targeted inhibitor of the insulinlike growth factor-1 receptor is administered to patients once every 3 weeks, for a total of eight infusions, according to a statement from Horizon Therapeutics, which manufactures the drug.
The approval was based on the findings from two similarly designed, parallel-group studies (Studies 1 and 2) involving 170 patients with thyroid eye disease who were randomized to receive either teprotumumab or placebo. Of those receiving the study drug, 71% in Study 1 and 83% in Study 2 had a reduction of more than 2 mm in eye protrusion, compared with 20% and 10%, respectively, among the placebo participants.
The most common adverse reactions in patients receiving teprotumumab were muscle spasm, nausea, alopecia, diarrhea, fatigue, and hyperglycemia. The treatment is contraindicated for pregnancy.
FROM THE FDA
Testosterone gel increases LV mass in older men
PHILADELPHIA – Testosterone gel for treatment of hypogonadism in older men boosted their left ventricular mass by 3.5% in a single year in the multicenter, double-blind, placebo-controlled Testosterone Cardiovascular Trial, although the clinical implications of this impressive increase remain unclear, Elizabeth Hutchins, MD, reported at the American Heart Association scientific sessions.
“I do think these results should be considered as part of the safety profile for testosterone gel and also represent an interesting and understudied area for future research,” said Dr. Hutchins, a hospitalist affiliated with the Los Angeles Biomedical Research Center at Harbor-UCLA Medical Center.
The Testosterone Cardiovascular Trial was one of seven coordinated placebo-controlled, double-blind clinical trials of the impact of raising serum testosterone levels in older men with low testosterone. Some results of what are known as the TTrials have previously been reported (Endocr Rev. 2018 Jun 1;39[3]:369-86).
Dr. Hutchins presented new findings on the effect of treatment with 1% topical testosterone gel on body surface area–indexed left ventricular mass. The trial utilized a widely prescribed, commercially available product known as AndroGel. The study included 123 men over age 65 with low serum testosterone and coronary CT angiography images obtained at baseline and again after 1 year of double-blind testosterone gel or placebo. More than 80% of the men were above age 75, half were obese, more than two-thirds had hypertension, and 30% had diabetes.
The men initially applied 5 g of the testosterone gel daily, providing 15 mg/day of testosterone, with subsequent dosing adjustments as needed based on serum testosterone levels measured at a central laboratory. Participants were evaluated in office visits with serum testosterone measurements every 3 months. Testosterone levels in the men assigned to active treatment quickly rose to normal range and stayed there for the full 12 months, while the placebo-treated controls continued to have below-normal testosterone throughout the trial.
The key study finding was that LV mass indexed to body surface area rose significantly in the testosterone gel group, from an average of 71.5 g/m2 at baseline to 74.8 g/m2 at 1 year. That’s a statistically significant 3.5% increase. In contrast, LV mass remained flat across the year in controls: 73.8 g/m2 at baseline and 73.3 g/m2 at 12 months.
There was, however, no change over time in left or right atrial or ventricular chamber volumes in the testosterone gel recipients, nor in the controls.
Session comoderator Eric D. Peterson, MD, professor of medicine and a cardiologist at Duke University in Durham, N.C., said that “this is a very important topic,” then posed a provocative question to Dr. Hutchins: “If the intervention had been running instead of testosterone gel, would the results have looked similar, and would you be concluding that there should be a warning around the use of running?”
Dr. Hutchins replied that she’s given that question much thought.
“Of course, exercise leads to LV hypertrophy and we consider that to be good muscle, and high blood pressure leads to LV hypertrophy and we consider that bad muscle. So which one is it in this case? From what I can find in the literature, it seems that incremental increases in LV mass in the absence of being an athlete are deleterious. But I think we would need outcomes-based research to really answer that question,” she said.
Dr. Hutchins noted that this was the first-ever randomized controlled trial to measure the effect of testosterone therapy on LV mass in humans. The documented increase achieved with 1 year of testosterone gel doesn’t come close to reaching the threshold of LV hypertrophy, which is about 125 g/m2 for men. But evidence from animal and observational human studies suggests that even in the absence of LV hypertrophy, increases in LV mass are associated with increased mortality, she added.
She reported having no financial conflicts regarding her study, sponsored by the National Institutes of Health.
SOURCE: Hutchins E. AHA 2019, Session FS.AOS.04.
PHILADELPHIA – Testosterone gel for treatment of hypogonadism in older men boosted their left ventricular mass by 3.5% in a single year in the multicenter, double-blind, placebo-controlled Testosterone Cardiovascular Trial, although the clinical implications of this impressive increase remain unclear, Elizabeth Hutchins, MD, reported at the American Heart Association scientific sessions.
“I do think these results should be considered as part of the safety profile for testosterone gel and also represent an interesting and understudied area for future research,” said Dr. Hutchins, a hospitalist affiliated with the Los Angeles Biomedical Research Center at Harbor-UCLA Medical Center.
The Testosterone Cardiovascular Trial was one of seven coordinated placebo-controlled, double-blind clinical trials of the impact of raising serum testosterone levels in older men with low testosterone. Some results of what are known as the TTrials have previously been reported (Endocr Rev. 2018 Jun 1;39[3]:369-86).
Dr. Hutchins presented new findings on the effect of treatment with 1% topical testosterone gel on body surface area–indexed left ventricular mass. The trial utilized a widely prescribed, commercially available product known as AndroGel. The study included 123 men over age 65 with low serum testosterone and coronary CT angiography images obtained at baseline and again after 1 year of double-blind testosterone gel or placebo. More than 80% of the men were above age 75, half were obese, more than two-thirds had hypertension, and 30% had diabetes.
The men initially applied 5 g of the testosterone gel daily, providing 15 mg/day of testosterone, with subsequent dosing adjustments as needed based on serum testosterone levels measured at a central laboratory. Participants were evaluated in office visits with serum testosterone measurements every 3 months. Testosterone levels in the men assigned to active treatment quickly rose to normal range and stayed there for the full 12 months, while the placebo-treated controls continued to have below-normal testosterone throughout the trial.
The key study finding was that LV mass indexed to body surface area rose significantly in the testosterone gel group, from an average of 71.5 g/m2 at baseline to 74.8 g/m2 at 1 year. That’s a statistically significant 3.5% increase. In contrast, LV mass remained flat across the year in controls: 73.8 g/m2 at baseline and 73.3 g/m2 at 12 months.
There was, however, no change over time in left or right atrial or ventricular chamber volumes in the testosterone gel recipients, nor in the controls.
Session comoderator Eric D. Peterson, MD, professor of medicine and a cardiologist at Duke University in Durham, N.C., said that “this is a very important topic,” then posed a provocative question to Dr. Hutchins: “If the intervention had been running instead of testosterone gel, would the results have looked similar, and would you be concluding that there should be a warning around the use of running?”
Dr. Hutchins replied that she’s given that question much thought.
“Of course, exercise leads to LV hypertrophy and we consider that to be good muscle, and high blood pressure leads to LV hypertrophy and we consider that bad muscle. So which one is it in this case? From what I can find in the literature, it seems that incremental increases in LV mass in the absence of being an athlete are deleterious. But I think we would need outcomes-based research to really answer that question,” she said.
Dr. Hutchins noted that this was the first-ever randomized controlled trial to measure the effect of testosterone therapy on LV mass in humans. The documented increase achieved with 1 year of testosterone gel doesn’t come close to reaching the threshold of LV hypertrophy, which is about 125 g/m2 for men. But evidence from animal and observational human studies suggests that even in the absence of LV hypertrophy, increases in LV mass are associated with increased mortality, she added.
She reported having no financial conflicts regarding her study, sponsored by the National Institutes of Health.
SOURCE: Hutchins E. AHA 2019, Session FS.AOS.04.
PHILADELPHIA – Testosterone gel for treatment of hypogonadism in older men boosted their left ventricular mass by 3.5% in a single year in the multicenter, double-blind, placebo-controlled Testosterone Cardiovascular Trial, although the clinical implications of this impressive increase remain unclear, Elizabeth Hutchins, MD, reported at the American Heart Association scientific sessions.
“I do think these results should be considered as part of the safety profile for testosterone gel and also represent an interesting and understudied area for future research,” said Dr. Hutchins, a hospitalist affiliated with the Los Angeles Biomedical Research Center at Harbor-UCLA Medical Center.
The Testosterone Cardiovascular Trial was one of seven coordinated placebo-controlled, double-blind clinical trials of the impact of raising serum testosterone levels in older men with low testosterone. Some results of what are known as the TTrials have previously been reported (Endocr Rev. 2018 Jun 1;39[3]:369-86).
Dr. Hutchins presented new findings on the effect of treatment with 1% topical testosterone gel on body surface area–indexed left ventricular mass. The trial utilized a widely prescribed, commercially available product known as AndroGel. The study included 123 men over age 65 with low serum testosterone and coronary CT angiography images obtained at baseline and again after 1 year of double-blind testosterone gel or placebo. More than 80% of the men were above age 75, half were obese, more than two-thirds had hypertension, and 30% had diabetes.
The men initially applied 5 g of the testosterone gel daily, providing 15 mg/day of testosterone, with subsequent dosing adjustments as needed based on serum testosterone levels measured at a central laboratory. Participants were evaluated in office visits with serum testosterone measurements every 3 months. Testosterone levels in the men assigned to active treatment quickly rose to normal range and stayed there for the full 12 months, while the placebo-treated controls continued to have below-normal testosterone throughout the trial.
The key study finding was that LV mass indexed to body surface area rose significantly in the testosterone gel group, from an average of 71.5 g/m2 at baseline to 74.8 g/m2 at 1 year. That’s a statistically significant 3.5% increase. In contrast, LV mass remained flat across the year in controls: 73.8 g/m2 at baseline and 73.3 g/m2 at 12 months.
There was, however, no change over time in left or right atrial or ventricular chamber volumes in the testosterone gel recipients, nor in the controls.
Session comoderator Eric D. Peterson, MD, professor of medicine and a cardiologist at Duke University in Durham, N.C., said that “this is a very important topic,” then posed a provocative question to Dr. Hutchins: “If the intervention had been running instead of testosterone gel, would the results have looked similar, and would you be concluding that there should be a warning around the use of running?”
Dr. Hutchins replied that she’s given that question much thought.
“Of course, exercise leads to LV hypertrophy and we consider that to be good muscle, and high blood pressure leads to LV hypertrophy and we consider that bad muscle. So which one is it in this case? From what I can find in the literature, it seems that incremental increases in LV mass in the absence of being an athlete are deleterious. But I think we would need outcomes-based research to really answer that question,” she said.
Dr. Hutchins noted that this was the first-ever randomized controlled trial to measure the effect of testosterone therapy on LV mass in humans. The documented increase achieved with 1 year of testosterone gel doesn’t come close to reaching the threshold of LV hypertrophy, which is about 125 g/m2 for men. But evidence from animal and observational human studies suggests that even in the absence of LV hypertrophy, increases in LV mass are associated with increased mortality, she added.
She reported having no financial conflicts regarding her study, sponsored by the National Institutes of Health.
SOURCE: Hutchins E. AHA 2019, Session FS.AOS.04.
REPORTING FROM AHA 2019
Treatment of heart failure with preserved ejection fraction is a work in progress
LOS ANGELES – When it comes to the optimal treatment of patients with heart failure with preserved ejection fraction and diabetes, cardiologists like Mark T. Kearney, MB ChB, MD, remain stumped.
“Over the years, the diagnosis of heart failure with preserved ejection fraction has been notoriously difficult [to treat], controversial, and ultimately involves aggressive catheterization of the heart to assess diastolic dysfunction, complex echocardiography, and invasive tests,” Dr. Kearney said at the World Congress on Insulin Resistance, Diabetes, and Cardiovascular Disease. “These patients have an ejection fraction of over 50% and classic signs and symptoms of heart failure. Studies of beta-blockers, angiotensin-converting enzyme inhibitors, and angiotensin II receptor blockers have been unsuccessful in this group of patients. We’re at the beginning of a journey in understanding this disorder, and it’s important, because more and more patients present to us with signs and symptoms of heart failure with an ejection fraction greater than 50%.”
In a recent analysis of 1,797 patients with chronic heart failure, Dr. Kearney, British Heart Foundation Professor of Cardiovascular and Diabetes Research at the Leeds (England) Institute of Cardiovascular and Metabolic Medicine, and colleagues examined whether beta-blockers and ACE inhibitors were associated with differential effects on mortality in patients with and without diabetes (Diabetes Care. 2018;41:136-42). Mean follow-up was 4 years.
For the ACE inhibitor component of the trial, the researchers correlated the dose of ramipril to outcomes and found that each milligram increase of ramipril reduced the risk of death by about 3%. “In the nondiabetic patients who did not receive an ACE inhibitor, mortality was about 60% – worse than most cancers,” Dr. Kearney said. “In patients with diabetes, there was a similar pattern. If you didn’t get an ACE inhibitor, mortality was 70%. So, if you get patients on an optimal dose of an ACE inhibitor, you improve their mortality substantially, whether they have diabetes or not.”
The beta-blocker component of the trial yielded similar results. “Among patients who did not receive a beta-blocker, the mortality was about 70% at 5 years – really terrible,” he said. “Every milligram of bisoprolol was associated with a reduction in mortality of about 9%. So, if a patient gets on an optimal dose of a beta-blocker and they have diabetes, it’s associated with prolongation of life over a year.”
Dr. Kearney said that patients often do not want to take an increased dose of a beta-blocker because of concerns about side effects, such as tiredness. “They ask me what the side effects of an increased dose would be. My answer is: ‘It will make you live longer.’ Usually, they’ll respond by agreeing to have a little bit more of the beta-blocker. The message here is, if you have a patient with ejection fraction heart failure and diabetes, get them on the optimal dose of a beta-blocker, even at the expense of an ACE inhibitor.”
In 2016, the European Society of Cardiology introduced guidelines for physicians to make a diagnosis of heart failure with preserved ejection fraction. The guidelines mandate that a diagnosis requires signs and symptoms of heart failure, elevated levels of natriuretic peptide, and echocardiographic abnormalities of cardiac structure and/or function in the presence of a left ventricular ejection fraction of 50% or more (Eur J Heart Fail. 2016;18[8]:891-975).
“Signs and symptoms of heart failure, elevated BNP [brain natriuretic peptide], and echocardiography allow us to make a diagnosis of heart failure with preserved ejection fraction,” Dr. Kearney, who is also dean of the Leeds University School of Medicine. “But we don’t know the outcome of these patients, we don’t know how to treat them, and we don’t know the impact on hospitalizations.”
In a large, unpublished cohort study conducted at Leeds, Dr. Kearney and colleagues evaluated how many patients met criteria for heart failure with reduced ejection fraction or heart failure with preserved ejection fraction after undergoing a BNP measurement. Ultimately, 959 patients met criteria. After assessment, 23% had no heart failure, 44% had heart failure with preserved ejection fraction, and 33% had heart failure with reduced ejection fraction. They found that patients with preserved ejection fraction were older (mean age, 84 years); were more likely to be female; and had less ischemia, less diabetes, and more hypertension. In addition, patients with preserved ejection fraction had significantly better survival than patients with reduced ejection fraction over 5 years follow-up.
“What was really interesting were the findings related to hospitalization,” he said. “All 959 patients accounted for 20,517 days in the hospital over 5 years, which is the equivalent of 1 patient occupying a hospital bed for 56 years. This disorder [heart failure with preserved ejection fraction], despite having a lower mortality than heart failure with reduced ejection fraction, leads to a significant burden on health care systems.”
Among patients with preserved ejection fraction, 82% were hospitalized for a noncardiovascular cause, 6.9% because of heart failure, and 11% were caused by other cardiovascular causes. Most of the hospital admissions were because of chest infections, falls, and other frailty-linked causes. “This link between systemic frailty and heart failure with preserved ejection fraction warrants further investigation,” Dr. Kearney said. “This is a major burden on patient hospital care.”
When the researchers examined outcomes in patients with and without diabetes, those with diabetes were younger, more likely to be male, and have a higher body mass index. They found that, in the presence of diabetes, mortality was increased in heart failure with preserved and reduced ejection fraction. “So, even at the age of 81 or 82, diabetes changes the pathophysiology of mortality in what was previously believed to be a benign disease,” he said.
In a subset analysis of patients with and without diabetes who were not taking a beta-blocker, there did not seem to be increased sympathetic activation in the patients with diabetes and heart failure with preserved ejection fraction, nor a difference in heart rate between the nondiabetic patients and patients with diabetes. However, among patients with heart failure with reduced ejection fraction, those with diabetes had an increased heart rate.
“Is heart failure with preserved ejection fraction in diabetes benign? I think the answer is no,” Dr. Kearney said. “It increases hospitalization and is a major burden on health care systems. What should we do? We deal with comorbidity and fall risk. It’s good old-fashioned doctoring, really. We address frailty and respiratory tract infections, but the key thing here is that we need more research.”
Dr. Kearney reported having no relevant financial disclosures.
LOS ANGELES – When it comes to the optimal treatment of patients with heart failure with preserved ejection fraction and diabetes, cardiologists like Mark T. Kearney, MB ChB, MD, remain stumped.
“Over the years, the diagnosis of heart failure with preserved ejection fraction has been notoriously difficult [to treat], controversial, and ultimately involves aggressive catheterization of the heart to assess diastolic dysfunction, complex echocardiography, and invasive tests,” Dr. Kearney said at the World Congress on Insulin Resistance, Diabetes, and Cardiovascular Disease. “These patients have an ejection fraction of over 50% and classic signs and symptoms of heart failure. Studies of beta-blockers, angiotensin-converting enzyme inhibitors, and angiotensin II receptor blockers have been unsuccessful in this group of patients. We’re at the beginning of a journey in understanding this disorder, and it’s important, because more and more patients present to us with signs and symptoms of heart failure with an ejection fraction greater than 50%.”
In a recent analysis of 1,797 patients with chronic heart failure, Dr. Kearney, British Heart Foundation Professor of Cardiovascular and Diabetes Research at the Leeds (England) Institute of Cardiovascular and Metabolic Medicine, and colleagues examined whether beta-blockers and ACE inhibitors were associated with differential effects on mortality in patients with and without diabetes (Diabetes Care. 2018;41:136-42). Mean follow-up was 4 years.
For the ACE inhibitor component of the trial, the researchers correlated the dose of ramipril to outcomes and found that each milligram increase of ramipril reduced the risk of death by about 3%. “In the nondiabetic patients who did not receive an ACE inhibitor, mortality was about 60% – worse than most cancers,” Dr. Kearney said. “In patients with diabetes, there was a similar pattern. If you didn’t get an ACE inhibitor, mortality was 70%. So, if you get patients on an optimal dose of an ACE inhibitor, you improve their mortality substantially, whether they have diabetes or not.”
The beta-blocker component of the trial yielded similar results. “Among patients who did not receive a beta-blocker, the mortality was about 70% at 5 years – really terrible,” he said. “Every milligram of bisoprolol was associated with a reduction in mortality of about 9%. So, if a patient gets on an optimal dose of a beta-blocker and they have diabetes, it’s associated with prolongation of life over a year.”
Dr. Kearney said that patients often do not want to take an increased dose of a beta-blocker because of concerns about side effects, such as tiredness. “They ask me what the side effects of an increased dose would be. My answer is: ‘It will make you live longer.’ Usually, they’ll respond by agreeing to have a little bit more of the beta-blocker. The message here is, if you have a patient with ejection fraction heart failure and diabetes, get them on the optimal dose of a beta-blocker, even at the expense of an ACE inhibitor.”
In 2016, the European Society of Cardiology introduced guidelines for physicians to make a diagnosis of heart failure with preserved ejection fraction. The guidelines mandate that a diagnosis requires signs and symptoms of heart failure, elevated levels of natriuretic peptide, and echocardiographic abnormalities of cardiac structure and/or function in the presence of a left ventricular ejection fraction of 50% or more (Eur J Heart Fail. 2016;18[8]:891-975).
“Signs and symptoms of heart failure, elevated BNP [brain natriuretic peptide], and echocardiography allow us to make a diagnosis of heart failure with preserved ejection fraction,” Dr. Kearney, who is also dean of the Leeds University School of Medicine. “But we don’t know the outcome of these patients, we don’t know how to treat them, and we don’t know the impact on hospitalizations.”
In a large, unpublished cohort study conducted at Leeds, Dr. Kearney and colleagues evaluated how many patients met criteria for heart failure with reduced ejection fraction or heart failure with preserved ejection fraction after undergoing a BNP measurement. Ultimately, 959 patients met criteria. After assessment, 23% had no heart failure, 44% had heart failure with preserved ejection fraction, and 33% had heart failure with reduced ejection fraction. They found that patients with preserved ejection fraction were older (mean age, 84 years); were more likely to be female; and had less ischemia, less diabetes, and more hypertension. In addition, patients with preserved ejection fraction had significantly better survival than patients with reduced ejection fraction over 5 years follow-up.
“What was really interesting were the findings related to hospitalization,” he said. “All 959 patients accounted for 20,517 days in the hospital over 5 years, which is the equivalent of 1 patient occupying a hospital bed for 56 years. This disorder [heart failure with preserved ejection fraction], despite having a lower mortality than heart failure with reduced ejection fraction, leads to a significant burden on health care systems.”
Among patients with preserved ejection fraction, 82% were hospitalized for a noncardiovascular cause, 6.9% because of heart failure, and 11% were caused by other cardiovascular causes. Most of the hospital admissions were because of chest infections, falls, and other frailty-linked causes. “This link between systemic frailty and heart failure with preserved ejection fraction warrants further investigation,” Dr. Kearney said. “This is a major burden on patient hospital care.”
When the researchers examined outcomes in patients with and without diabetes, those with diabetes were younger, more likely to be male, and have a higher body mass index. They found that, in the presence of diabetes, mortality was increased in heart failure with preserved and reduced ejection fraction. “So, even at the age of 81 or 82, diabetes changes the pathophysiology of mortality in what was previously believed to be a benign disease,” he said.
In a subset analysis of patients with and without diabetes who were not taking a beta-blocker, there did not seem to be increased sympathetic activation in the patients with diabetes and heart failure with preserved ejection fraction, nor a difference in heart rate between the nondiabetic patients and patients with diabetes. However, among patients with heart failure with reduced ejection fraction, those with diabetes had an increased heart rate.
“Is heart failure with preserved ejection fraction in diabetes benign? I think the answer is no,” Dr. Kearney said. “It increases hospitalization and is a major burden on health care systems. What should we do? We deal with comorbidity and fall risk. It’s good old-fashioned doctoring, really. We address frailty and respiratory tract infections, but the key thing here is that we need more research.”
Dr. Kearney reported having no relevant financial disclosures.
LOS ANGELES – When it comes to the optimal treatment of patients with heart failure with preserved ejection fraction and diabetes, cardiologists like Mark T. Kearney, MB ChB, MD, remain stumped.
“Over the years, the diagnosis of heart failure with preserved ejection fraction has been notoriously difficult [to treat], controversial, and ultimately involves aggressive catheterization of the heart to assess diastolic dysfunction, complex echocardiography, and invasive tests,” Dr. Kearney said at the World Congress on Insulin Resistance, Diabetes, and Cardiovascular Disease. “These patients have an ejection fraction of over 50% and classic signs and symptoms of heart failure. Studies of beta-blockers, angiotensin-converting enzyme inhibitors, and angiotensin II receptor blockers have been unsuccessful in this group of patients. We’re at the beginning of a journey in understanding this disorder, and it’s important, because more and more patients present to us with signs and symptoms of heart failure with an ejection fraction greater than 50%.”
In a recent analysis of 1,797 patients with chronic heart failure, Dr. Kearney, British Heart Foundation Professor of Cardiovascular and Diabetes Research at the Leeds (England) Institute of Cardiovascular and Metabolic Medicine, and colleagues examined whether beta-blockers and ACE inhibitors were associated with differential effects on mortality in patients with and without diabetes (Diabetes Care. 2018;41:136-42). Mean follow-up was 4 years.
For the ACE inhibitor component of the trial, the researchers correlated the dose of ramipril to outcomes and found that each milligram increase of ramipril reduced the risk of death by about 3%. “In the nondiabetic patients who did not receive an ACE inhibitor, mortality was about 60% – worse than most cancers,” Dr. Kearney said. “In patients with diabetes, there was a similar pattern. If you didn’t get an ACE inhibitor, mortality was 70%. So, if you get patients on an optimal dose of an ACE inhibitor, you improve their mortality substantially, whether they have diabetes or not.”
The beta-blocker component of the trial yielded similar results. “Among patients who did not receive a beta-blocker, the mortality was about 70% at 5 years – really terrible,” he said. “Every milligram of bisoprolol was associated with a reduction in mortality of about 9%. So, if a patient gets on an optimal dose of a beta-blocker and they have diabetes, it’s associated with prolongation of life over a year.”
Dr. Kearney said that patients often do not want to take an increased dose of a beta-blocker because of concerns about side effects, such as tiredness. “They ask me what the side effects of an increased dose would be. My answer is: ‘It will make you live longer.’ Usually, they’ll respond by agreeing to have a little bit more of the beta-blocker. The message here is, if you have a patient with ejection fraction heart failure and diabetes, get them on the optimal dose of a beta-blocker, even at the expense of an ACE inhibitor.”
In 2016, the European Society of Cardiology introduced guidelines for physicians to make a diagnosis of heart failure with preserved ejection fraction. The guidelines mandate that a diagnosis requires signs and symptoms of heart failure, elevated levels of natriuretic peptide, and echocardiographic abnormalities of cardiac structure and/or function in the presence of a left ventricular ejection fraction of 50% or more (Eur J Heart Fail. 2016;18[8]:891-975).
“Signs and symptoms of heart failure, elevated BNP [brain natriuretic peptide], and echocardiography allow us to make a diagnosis of heart failure with preserved ejection fraction,” Dr. Kearney, who is also dean of the Leeds University School of Medicine. “But we don’t know the outcome of these patients, we don’t know how to treat them, and we don’t know the impact on hospitalizations.”
In a large, unpublished cohort study conducted at Leeds, Dr. Kearney and colleagues evaluated how many patients met criteria for heart failure with reduced ejection fraction or heart failure with preserved ejection fraction after undergoing a BNP measurement. Ultimately, 959 patients met criteria. After assessment, 23% had no heart failure, 44% had heart failure with preserved ejection fraction, and 33% had heart failure with reduced ejection fraction. They found that patients with preserved ejection fraction were older (mean age, 84 years); were more likely to be female; and had less ischemia, less diabetes, and more hypertension. In addition, patients with preserved ejection fraction had significantly better survival than patients with reduced ejection fraction over 5 years follow-up.
“What was really interesting were the findings related to hospitalization,” he said. “All 959 patients accounted for 20,517 days in the hospital over 5 years, which is the equivalent of 1 patient occupying a hospital bed for 56 years. This disorder [heart failure with preserved ejection fraction], despite having a lower mortality than heart failure with reduced ejection fraction, leads to a significant burden on health care systems.”
Among patients with preserved ejection fraction, 82% were hospitalized for a noncardiovascular cause, 6.9% because of heart failure, and 11% were caused by other cardiovascular causes. Most of the hospital admissions were because of chest infections, falls, and other frailty-linked causes. “This link between systemic frailty and heart failure with preserved ejection fraction warrants further investigation,” Dr. Kearney said. “This is a major burden on patient hospital care.”
When the researchers examined outcomes in patients with and without diabetes, those with diabetes were younger, more likely to be male, and have a higher body mass index. They found that, in the presence of diabetes, mortality was increased in heart failure with preserved and reduced ejection fraction. “So, even at the age of 81 or 82, diabetes changes the pathophysiology of mortality in what was previously believed to be a benign disease,” he said.
In a subset analysis of patients with and without diabetes who were not taking a beta-blocker, there did not seem to be increased sympathetic activation in the patients with diabetes and heart failure with preserved ejection fraction, nor a difference in heart rate between the nondiabetic patients and patients with diabetes. However, among patients with heart failure with reduced ejection fraction, those with diabetes had an increased heart rate.
“Is heart failure with preserved ejection fraction in diabetes benign? I think the answer is no,” Dr. Kearney said. “It increases hospitalization and is a major burden on health care systems. What should we do? We deal with comorbidity and fall risk. It’s good old-fashioned doctoring, really. We address frailty and respiratory tract infections, but the key thing here is that we need more research.”
Dr. Kearney reported having no relevant financial disclosures.
EXPERT ANALYSIS FROM WCIRDC 2019
New guideline for testosterone treatment in men with ‘low T’
The American College of Physicians has released new clinical guidelines providing practical recommendations for testosterone therapy in adult men with age-related low testosterone.
The evidence-based recommendations target all clinicians and were published online January 6, 2020, in Annals of Internal Medicine, highlighting data from a systematic review of evidence on the efficacy and safety of testosterone treatment in adult men with age-related low testosterone.
Serum testosterone levels drop as men age, starting in their mid-30s, and approximately 20% of American men older than 60 years have low testosterone.
However, no widely accepted testosterone threshold level exists that represents a measure below which symptoms of androgen deficiency and adverse health outcomes occur.
In addition, the role of testosterone therapy in managing this patient population is controversial.
“The purpose of this American College of Physicians guideline is to present recommendations based on the best available evidence on the benefits, harms, and costs of testosterone treatment in adult men with age-related low testosterone,” write Amir Qaseem, MD, PhD, MHA, from the American College of Physicians, Philadelphia, and colleagues.
“This guideline does not address screening or diagnosis of hypogonadism or monitoring of testosterone levels,” the authors note.
In particular, the recommendations suggest that clinicians should initiate testosterone treatment in these patients only to help them improve their sexual function.
According to the authors, moderate-certainty evidence from seven trials involving testosterone treatment in adult men with age-related low testosterone showed a small improvement in global sexual function, whereas low-certainty evidence from seven trials showed a small improvement in erectile function.
By contrast, the guideline emphasizes that clinicians should avoid prescribing testosterone treatment for any other concern in this population. Available evidence demonstrates little to no improvement in physical function, depressive symptoms, energy and vitality, or cognition among these men after receiving testosterone treatment, the authors stress.
ACP recommends that clinicians should reassess men’s symptoms within 12 months of testosterone treatment initiation, with regular reevaluations during subsequent follow up. Clinicians should discontinue treatment in men if sexual function fails to improve.
The guideline also recommends using intramuscular formulations of testosterone treatment for this patient population instead of transdermal ones, because intramuscular formulations cost less and have similar clinical effectiveness and harms.
“The annual cost in 2016 per beneficiary for TRT [testosterone replacement therapy] was $2,135.32 for the transdermal and $156.24 for the intramuscular formulation, according to paid pharmaceutical claims provided in the 2016 Medicare Part D Drug Claims data,” the authors write.
In an accompanying editorial, E. Victor Adlin, MD, of Temple University, Philadelphia, notes that these new ACP guidelines mostly mirror those recently proposed by both the Endocrine Society and the American Urological Association.
However, he predicts that many clinicians will question the ACP’s recommendation to favor use of intramuscular over transdermal formulations of testosterone.
Although Dr. Adlin acknowledges the lower cost of intramuscular preparations as a major consideration, he explains that “the need for an intramuscular injection every 1-4 weeks is a potential barrier to adherence, and some patients require visits to a health care facility for the injections, which may add to the expense.”
Fluctuating blood testosterone levels after each injection may also result in irregular symptom relief and difficulty achieving the desired blood level, he adds. “Individual preference may vary widely in the choice of testosterone therapy.”
Overall, Dr. Adlin stresses that a patient-clinician discussion should serve as the foundation for starting testosterone therapy in men with age-related low testosterone, with the patient playing a central role in treatment decision making.
This guideline was developed with financial support from the American College of Physicians’ operating budget. Study author Carrie Horwitch reports serving as a fiduciary officer for the Washington State Medical Association. Jennifer S. Lin, a member of the ACP Clinical Guidelines Committee, reports being an employee of Kaiser Permanente. Robert McLean, another member of the committee, reports being an employee of Northeast Medical Group. The remaining authors and the editorialist have disclosed no relevant financial relationships.
A version of this story appeared on Medscape.com.
The American College of Physicians has released new clinical guidelines providing practical recommendations for testosterone therapy in adult men with age-related low testosterone.
The evidence-based recommendations target all clinicians and were published online January 6, 2020, in Annals of Internal Medicine, highlighting data from a systematic review of evidence on the efficacy and safety of testosterone treatment in adult men with age-related low testosterone.
Serum testosterone levels drop as men age, starting in their mid-30s, and approximately 20% of American men older than 60 years have low testosterone.
However, no widely accepted testosterone threshold level exists that represents a measure below which symptoms of androgen deficiency and adverse health outcomes occur.
In addition, the role of testosterone therapy in managing this patient population is controversial.
“The purpose of this American College of Physicians guideline is to present recommendations based on the best available evidence on the benefits, harms, and costs of testosterone treatment in adult men with age-related low testosterone,” write Amir Qaseem, MD, PhD, MHA, from the American College of Physicians, Philadelphia, and colleagues.
“This guideline does not address screening or diagnosis of hypogonadism or monitoring of testosterone levels,” the authors note.
In particular, the recommendations suggest that clinicians should initiate testosterone treatment in these patients only to help them improve their sexual function.
According to the authors, moderate-certainty evidence from seven trials involving testosterone treatment in adult men with age-related low testosterone showed a small improvement in global sexual function, whereas low-certainty evidence from seven trials showed a small improvement in erectile function.
By contrast, the guideline emphasizes that clinicians should avoid prescribing testosterone treatment for any other concern in this population. Available evidence demonstrates little to no improvement in physical function, depressive symptoms, energy and vitality, or cognition among these men after receiving testosterone treatment, the authors stress.
ACP recommends that clinicians should reassess men’s symptoms within 12 months of testosterone treatment initiation, with regular reevaluations during subsequent follow up. Clinicians should discontinue treatment in men if sexual function fails to improve.
The guideline also recommends using intramuscular formulations of testosterone treatment for this patient population instead of transdermal ones, because intramuscular formulations cost less and have similar clinical effectiveness and harms.
“The annual cost in 2016 per beneficiary for TRT [testosterone replacement therapy] was $2,135.32 for the transdermal and $156.24 for the intramuscular formulation, according to paid pharmaceutical claims provided in the 2016 Medicare Part D Drug Claims data,” the authors write.
In an accompanying editorial, E. Victor Adlin, MD, of Temple University, Philadelphia, notes that these new ACP guidelines mostly mirror those recently proposed by both the Endocrine Society and the American Urological Association.
However, he predicts that many clinicians will question the ACP’s recommendation to favor use of intramuscular over transdermal formulations of testosterone.
Although Dr. Adlin acknowledges the lower cost of intramuscular preparations as a major consideration, he explains that “the need for an intramuscular injection every 1-4 weeks is a potential barrier to adherence, and some patients require visits to a health care facility for the injections, which may add to the expense.”
Fluctuating blood testosterone levels after each injection may also result in irregular symptom relief and difficulty achieving the desired blood level, he adds. “Individual preference may vary widely in the choice of testosterone therapy.”
Overall, Dr. Adlin stresses that a patient-clinician discussion should serve as the foundation for starting testosterone therapy in men with age-related low testosterone, with the patient playing a central role in treatment decision making.
This guideline was developed with financial support from the American College of Physicians’ operating budget. Study author Carrie Horwitch reports serving as a fiduciary officer for the Washington State Medical Association. Jennifer S. Lin, a member of the ACP Clinical Guidelines Committee, reports being an employee of Kaiser Permanente. Robert McLean, another member of the committee, reports being an employee of Northeast Medical Group. The remaining authors and the editorialist have disclosed no relevant financial relationships.
A version of this story appeared on Medscape.com.
The American College of Physicians has released new clinical guidelines providing practical recommendations for testosterone therapy in adult men with age-related low testosterone.
The evidence-based recommendations target all clinicians and were published online January 6, 2020, in Annals of Internal Medicine, highlighting data from a systematic review of evidence on the efficacy and safety of testosterone treatment in adult men with age-related low testosterone.
Serum testosterone levels drop as men age, starting in their mid-30s, and approximately 20% of American men older than 60 years have low testosterone.
However, no widely accepted testosterone threshold level exists that represents a measure below which symptoms of androgen deficiency and adverse health outcomes occur.
In addition, the role of testosterone therapy in managing this patient population is controversial.
“The purpose of this American College of Physicians guideline is to present recommendations based on the best available evidence on the benefits, harms, and costs of testosterone treatment in adult men with age-related low testosterone,” write Amir Qaseem, MD, PhD, MHA, from the American College of Physicians, Philadelphia, and colleagues.
“This guideline does not address screening or diagnosis of hypogonadism or monitoring of testosterone levels,” the authors note.
In particular, the recommendations suggest that clinicians should initiate testosterone treatment in these patients only to help them improve their sexual function.
According to the authors, moderate-certainty evidence from seven trials involving testosterone treatment in adult men with age-related low testosterone showed a small improvement in global sexual function, whereas low-certainty evidence from seven trials showed a small improvement in erectile function.
By contrast, the guideline emphasizes that clinicians should avoid prescribing testosterone treatment for any other concern in this population. Available evidence demonstrates little to no improvement in physical function, depressive symptoms, energy and vitality, or cognition among these men after receiving testosterone treatment, the authors stress.
ACP recommends that clinicians should reassess men’s symptoms within 12 months of testosterone treatment initiation, with regular reevaluations during subsequent follow up. Clinicians should discontinue treatment in men if sexual function fails to improve.
The guideline also recommends using intramuscular formulations of testosterone treatment for this patient population instead of transdermal ones, because intramuscular formulations cost less and have similar clinical effectiveness and harms.
“The annual cost in 2016 per beneficiary for TRT [testosterone replacement therapy] was $2,135.32 for the transdermal and $156.24 for the intramuscular formulation, according to paid pharmaceutical claims provided in the 2016 Medicare Part D Drug Claims data,” the authors write.
In an accompanying editorial, E. Victor Adlin, MD, of Temple University, Philadelphia, notes that these new ACP guidelines mostly mirror those recently proposed by both the Endocrine Society and the American Urological Association.
However, he predicts that many clinicians will question the ACP’s recommendation to favor use of intramuscular over transdermal formulations of testosterone.
Although Dr. Adlin acknowledges the lower cost of intramuscular preparations as a major consideration, he explains that “the need for an intramuscular injection every 1-4 weeks is a potential barrier to adherence, and some patients require visits to a health care facility for the injections, which may add to the expense.”
Fluctuating blood testosterone levels after each injection may also result in irregular symptom relief and difficulty achieving the desired blood level, he adds. “Individual preference may vary widely in the choice of testosterone therapy.”
Overall, Dr. Adlin stresses that a patient-clinician discussion should serve as the foundation for starting testosterone therapy in men with age-related low testosterone, with the patient playing a central role in treatment decision making.
This guideline was developed with financial support from the American College of Physicians’ operating budget. Study author Carrie Horwitch reports serving as a fiduciary officer for the Washington State Medical Association. Jennifer S. Lin, a member of the ACP Clinical Guidelines Committee, reports being an employee of Kaiser Permanente. Robert McLean, another member of the committee, reports being an employee of Northeast Medical Group. The remaining authors and the editorialist have disclosed no relevant financial relationships.
A version of this story appeared on Medscape.com.
Anorexia linked to low bone density, osteoporosis
A new study has reinforced the link between anorexia nervosa and reduced bone mineral density (BMD), especially in patients with lower body mass index.
“Our large study raises further concerns that [anorexia nervosa] has significant deleterious effects on BMD,” wrote Cassandra Workman, MD, of the Eating Recovery Center in Denver and coauthors. The study was published in Bone.
To determine the degree of low BMD in patients with certain severe eating disorders, the researchers reviewed the medical records of 336 patients with either anorexia nervosa–restricting subtype (AN-R) or anorexia nervosa–binge/purge subtype (AN-BP) who had been admitted to a treatment facility in Denver. Bone density was assessed using dual-energy x-ray absorptiometry, with osteopenia being diagnosed for an average BMD z score between –1.0 and –2.0 and osteoporosis being diagnosed for an average BMD z score of less than –2.0. The average age of the patients was 27 years (standard deviation, 9.12; range, 18-69), and 91% (n = 305) were women.
Across the sample, the average BMD z score was –1.67 (SD, 1.21), and 43.5% of the sample met the established criteria for low BMD.
Patients with AN-R had slightly lower z scores (–1.79; SD, 1.31), compared with patients with AN-BP (–1.54; SD, 1.08; P = .06), but the severity of osteoporosis was greater in patients with AN-R, compared with patients with AN-BP (chi-square, 7.40; P less than .01).
The authors acknowledged their study’s limitations, including the use of retrospective data from the patient charts, which did not allow for assessment of follow-up improvements or longer-term effects. In addition, they noted that extrapolation of their findings may be problematic because all the patients were from a single site and the data might be representative of “a more ill population than a true cross section of the eating disorder population.”
The authors reported no conflicts of interest.
SOURCE: Workman C et al. Bone. 2019 Nov 23. doi: 10.1016/j.bone.2019.115161.
A new study has reinforced the link between anorexia nervosa and reduced bone mineral density (BMD), especially in patients with lower body mass index.
“Our large study raises further concerns that [anorexia nervosa] has significant deleterious effects on BMD,” wrote Cassandra Workman, MD, of the Eating Recovery Center in Denver and coauthors. The study was published in Bone.
To determine the degree of low BMD in patients with certain severe eating disorders, the researchers reviewed the medical records of 336 patients with either anorexia nervosa–restricting subtype (AN-R) or anorexia nervosa–binge/purge subtype (AN-BP) who had been admitted to a treatment facility in Denver. Bone density was assessed using dual-energy x-ray absorptiometry, with osteopenia being diagnosed for an average BMD z score between –1.0 and –2.0 and osteoporosis being diagnosed for an average BMD z score of less than –2.0. The average age of the patients was 27 years (standard deviation, 9.12; range, 18-69), and 91% (n = 305) were women.
Across the sample, the average BMD z score was –1.67 (SD, 1.21), and 43.5% of the sample met the established criteria for low BMD.
Patients with AN-R had slightly lower z scores (–1.79; SD, 1.31), compared with patients with AN-BP (–1.54; SD, 1.08; P = .06), but the severity of osteoporosis was greater in patients with AN-R, compared with patients with AN-BP (chi-square, 7.40; P less than .01).
The authors acknowledged their study’s limitations, including the use of retrospective data from the patient charts, which did not allow for assessment of follow-up improvements or longer-term effects. In addition, they noted that extrapolation of their findings may be problematic because all the patients were from a single site and the data might be representative of “a more ill population than a true cross section of the eating disorder population.”
The authors reported no conflicts of interest.
SOURCE: Workman C et al. Bone. 2019 Nov 23. doi: 10.1016/j.bone.2019.115161.
A new study has reinforced the link between anorexia nervosa and reduced bone mineral density (BMD), especially in patients with lower body mass index.
“Our large study raises further concerns that [anorexia nervosa] has significant deleterious effects on BMD,” wrote Cassandra Workman, MD, of the Eating Recovery Center in Denver and coauthors. The study was published in Bone.
To determine the degree of low BMD in patients with certain severe eating disorders, the researchers reviewed the medical records of 336 patients with either anorexia nervosa–restricting subtype (AN-R) or anorexia nervosa–binge/purge subtype (AN-BP) who had been admitted to a treatment facility in Denver. Bone density was assessed using dual-energy x-ray absorptiometry, with osteopenia being diagnosed for an average BMD z score between –1.0 and –2.0 and osteoporosis being diagnosed for an average BMD z score of less than –2.0. The average age of the patients was 27 years (standard deviation, 9.12; range, 18-69), and 91% (n = 305) were women.
Across the sample, the average BMD z score was –1.67 (SD, 1.21), and 43.5% of the sample met the established criteria for low BMD.
Patients with AN-R had slightly lower z scores (–1.79; SD, 1.31), compared with patients with AN-BP (–1.54; SD, 1.08; P = .06), but the severity of osteoporosis was greater in patients with AN-R, compared with patients with AN-BP (chi-square, 7.40; P less than .01).
The authors acknowledged their study’s limitations, including the use of retrospective data from the patient charts, which did not allow for assessment of follow-up improvements or longer-term effects. In addition, they noted that extrapolation of their findings may be problematic because all the patients were from a single site and the data might be representative of “a more ill population than a true cross section of the eating disorder population.”
The authors reported no conflicts of interest.
SOURCE: Workman C et al. Bone. 2019 Nov 23. doi: 10.1016/j.bone.2019.115161.
FROM BONE