User login
Pence named COVID-19 point person as CDC reports possible community spread
Vice President Mike Pence will be the White House point person quarterbacking the administration’s response to COVID-19, although President Donald Trump was quick to dismiss the notion that he is a so-called coronavirus “czar.”
President Trump introduced Vice President Pence in this role during a Feb. 26 press conference. The same night, officials at the Centers for Disease Control and Prevention announced the first case of possible community spread of the novel coronavirus in the United States.
“I am going to be putting our vice president, Mike Pence, in charge, and Mike will be working with the professionals, the doctors, and everybody else that is working” on this, President Trump said.
“Mike is going to be in charge and Mike will report back to me, but he’s got a certain talent for this,” President Trump continued, noting that while Vice President Pence was governor of Indiana, his was the first state to have a patient affected by the 2014 Middle East Respiratory Syndrome coronavirus (MERS-CoV) outbreak, so he has experience in a similar situation.
“I know full well the importance of presidential leadership, the importance of administration leadership, and the vital role of partnerships of state and local governments and health authorities in responding to the potential threat of dangerous infectious diseases,” Vice President Pence said.
He said that his role will be to continue to meet with the Coronavirus Task Force and bring to the president “the best options for action and to see to the safety and well being and health of the American people. I will also be continuing to reach out to governors [and] state and local officials.”
Vice President Pence said he will also be working with Congress to ensure that resources are available.
It was noted during the press conference that some members of Congress consider the $2.5 billion in emergency appropriations requested by the White House to be inadequate and that the legislative branch is working to provide more funding.
Vice President Pence’s new role does not change the command structure of the Coronavirus Task Force, which is currently led by Department of Health & Human Services Secretary Alex Azar.
Speaking at the press conference, Secretary Azar noted that he is still chairman of the task force. “Having the vice president gives me the biggest stick one can have in the government on this whole-of-government approach.”
He emphatically stated, “not in the least,” in response to a question about whether he felt he was being replaced. “When this was mentioned to me, I said I was delighted that I get to have the vice president helping in this way. Delighted.”
The announcement came as President Trump continued to downplay the threat of the coronavirus to U.S. citizens, going so far as to contradict CDC officials who have stated that it is a matter of when, not if, there will be community spread in the United States.
“I don’t think it’s inevitable,” President Trump said. “I think that there’s a chance that it could get worse. There’s a chance it could get fairly substantially worse, but nothing’s inevitable.”
Immediately after President Trump wrapped up his statement, however, the CDC formally announced the first case of possible community spread of the coronavirus. In a statement issued to the press, the agency announced the 15th confirmed case in the United States, a person in California “who reportedly did not have relevant travel history or exposure to another known patient” with the coronavirus.
“This case was detected through the U.S. public health system – picked up by astute clinicians,” CDC added, noting it will continue to provide updates on the evolving situation.
Vice President Mike Pence will be the White House point person quarterbacking the administration’s response to COVID-19, although President Donald Trump was quick to dismiss the notion that he is a so-called coronavirus “czar.”
President Trump introduced Vice President Pence in this role during a Feb. 26 press conference. The same night, officials at the Centers for Disease Control and Prevention announced the first case of possible community spread of the novel coronavirus in the United States.
“I am going to be putting our vice president, Mike Pence, in charge, and Mike will be working with the professionals, the doctors, and everybody else that is working” on this, President Trump said.
“Mike is going to be in charge and Mike will report back to me, but he’s got a certain talent for this,” President Trump continued, noting that while Vice President Pence was governor of Indiana, his was the first state to have a patient affected by the 2014 Middle East Respiratory Syndrome coronavirus (MERS-CoV) outbreak, so he has experience in a similar situation.
“I know full well the importance of presidential leadership, the importance of administration leadership, and the vital role of partnerships of state and local governments and health authorities in responding to the potential threat of dangerous infectious diseases,” Vice President Pence said.
He said that his role will be to continue to meet with the Coronavirus Task Force and bring to the president “the best options for action and to see to the safety and well being and health of the American people. I will also be continuing to reach out to governors [and] state and local officials.”
Vice President Pence said he will also be working with Congress to ensure that resources are available.
It was noted during the press conference that some members of Congress consider the $2.5 billion in emergency appropriations requested by the White House to be inadequate and that the legislative branch is working to provide more funding.
Vice President Pence’s new role does not change the command structure of the Coronavirus Task Force, which is currently led by Department of Health & Human Services Secretary Alex Azar.
Speaking at the press conference, Secretary Azar noted that he is still chairman of the task force. “Having the vice president gives me the biggest stick one can have in the government on this whole-of-government approach.”
He emphatically stated, “not in the least,” in response to a question about whether he felt he was being replaced. “When this was mentioned to me, I said I was delighted that I get to have the vice president helping in this way. Delighted.”
The announcement came as President Trump continued to downplay the threat of the coronavirus to U.S. citizens, going so far as to contradict CDC officials who have stated that it is a matter of when, not if, there will be community spread in the United States.
“I don’t think it’s inevitable,” President Trump said. “I think that there’s a chance that it could get worse. There’s a chance it could get fairly substantially worse, but nothing’s inevitable.”
Immediately after President Trump wrapped up his statement, however, the CDC formally announced the first case of possible community spread of the coronavirus. In a statement issued to the press, the agency announced the 15th confirmed case in the United States, a person in California “who reportedly did not have relevant travel history or exposure to another known patient” with the coronavirus.
“This case was detected through the U.S. public health system – picked up by astute clinicians,” CDC added, noting it will continue to provide updates on the evolving situation.
Vice President Mike Pence will be the White House point person quarterbacking the administration’s response to COVID-19, although President Donald Trump was quick to dismiss the notion that he is a so-called coronavirus “czar.”
President Trump introduced Vice President Pence in this role during a Feb. 26 press conference. The same night, officials at the Centers for Disease Control and Prevention announced the first case of possible community spread of the novel coronavirus in the United States.
“I am going to be putting our vice president, Mike Pence, in charge, and Mike will be working with the professionals, the doctors, and everybody else that is working” on this, President Trump said.
“Mike is going to be in charge and Mike will report back to me, but he’s got a certain talent for this,” President Trump continued, noting that while Vice President Pence was governor of Indiana, his was the first state to have a patient affected by the 2014 Middle East Respiratory Syndrome coronavirus (MERS-CoV) outbreak, so he has experience in a similar situation.
“I know full well the importance of presidential leadership, the importance of administration leadership, and the vital role of partnerships of state and local governments and health authorities in responding to the potential threat of dangerous infectious diseases,” Vice President Pence said.
He said that his role will be to continue to meet with the Coronavirus Task Force and bring to the president “the best options for action and to see to the safety and well being and health of the American people. I will also be continuing to reach out to governors [and] state and local officials.”
Vice President Pence said he will also be working with Congress to ensure that resources are available.
It was noted during the press conference that some members of Congress consider the $2.5 billion in emergency appropriations requested by the White House to be inadequate and that the legislative branch is working to provide more funding.
Vice President Pence’s new role does not change the command structure of the Coronavirus Task Force, which is currently led by Department of Health & Human Services Secretary Alex Azar.
Speaking at the press conference, Secretary Azar noted that he is still chairman of the task force. “Having the vice president gives me the biggest stick one can have in the government on this whole-of-government approach.”
He emphatically stated, “not in the least,” in response to a question about whether he felt he was being replaced. “When this was mentioned to me, I said I was delighted that I get to have the vice president helping in this way. Delighted.”
The announcement came as President Trump continued to downplay the threat of the coronavirus to U.S. citizens, going so far as to contradict CDC officials who have stated that it is a matter of when, not if, there will be community spread in the United States.
“I don’t think it’s inevitable,” President Trump said. “I think that there’s a chance that it could get worse. There’s a chance it could get fairly substantially worse, but nothing’s inevitable.”
Immediately after President Trump wrapped up his statement, however, the CDC formally announced the first case of possible community spread of the coronavirus. In a statement issued to the press, the agency announced the 15th confirmed case in the United States, a person in California “who reportedly did not have relevant travel history or exposure to another known patient” with the coronavirus.
“This case was detected through the U.S. public health system – picked up by astute clinicians,” CDC added, noting it will continue to provide updates on the evolving situation.
COVID-19: Time to ‘take the risk of scaring people’
It’s past time to call the novel coronavirus, COVID-19, a pandemic and “time to push people to prepare, and guide their prep,” according to risk communication experts.
Medical messaging about containing or stopping the spread of the virus is doing more harm than good, write Peter Sandman, PhD, and Jody Lanard, MD, both based in New York City, in a recent blog post.
“We are near-certain that the desperate-sounding last-ditch containment messaging of recent days is contributing to a massive global misperception,” they warn.
“The most crucial (and overdue) risk communication task … is to help people visualize their communities when ‘keeping it out’ – containment – is no longer relevant.”
That message is embraced by several experts who spoke to Medscape Medical News.
“I’m jealous of what [they] have written: It is so clear, so correct, and so practical,” said David Fisman, MD, MPH, professor of epidemiology at the University of Toronto, Canada. “I think WHO [World Health Organization] is shying away from the P word,” he continued, referring to the organization’s continuing decision not to call the outbreak a pandemic.
“I fully support exactly what [Sandman and Lanard] are saying,” said Michael Osterholm, PhD, MPH, professor of environmental health sciences and director of the Center for Infectious Disease Research and Policy (CIDRAP) at the University of Minnesota in Minneapolis.
Sandman and Lanard write. “Hardly any officials are telling civil society and the general public how to get ready for this pandemic.”
Effective communication should inform people of what to expect now, they continue: “[T]he end of most quarantines, travel restrictions, contact tracing, and other measures designed to keep ‘them’ from infecting ‘us,’ and the switch to measures like canceling mass events designed to keep us from infecting each other.”
Among the new messages that should be delivered are things like:
- Stockpiling nonperishable food and prescription meds.
- Considering care of sick family members.
- Cross-training work personnel so one person’s absence won’t derail an organization’s ability to function.
“We hope that governments and healthcare institutions are using this time wisely,” Sandman and Lanard continue. “We know that ordinary citizens are not being asked to do so. In most countries … ordinary citizens have not been asked to prepare. Instead, they have been led to expect that their governments will keep the virus from their doors.”
This article first appeared on Medscape.com.
It’s past time to call the novel coronavirus, COVID-19, a pandemic and “time to push people to prepare, and guide their prep,” according to risk communication experts.
Medical messaging about containing or stopping the spread of the virus is doing more harm than good, write Peter Sandman, PhD, and Jody Lanard, MD, both based in New York City, in a recent blog post.
“We are near-certain that the desperate-sounding last-ditch containment messaging of recent days is contributing to a massive global misperception,” they warn.
“The most crucial (and overdue) risk communication task … is to help people visualize their communities when ‘keeping it out’ – containment – is no longer relevant.”
That message is embraced by several experts who spoke to Medscape Medical News.
“I’m jealous of what [they] have written: It is so clear, so correct, and so practical,” said David Fisman, MD, MPH, professor of epidemiology at the University of Toronto, Canada. “I think WHO [World Health Organization] is shying away from the P word,” he continued, referring to the organization’s continuing decision not to call the outbreak a pandemic.
“I fully support exactly what [Sandman and Lanard] are saying,” said Michael Osterholm, PhD, MPH, professor of environmental health sciences and director of the Center for Infectious Disease Research and Policy (CIDRAP) at the University of Minnesota in Minneapolis.
Sandman and Lanard write. “Hardly any officials are telling civil society and the general public how to get ready for this pandemic.”
Effective communication should inform people of what to expect now, they continue: “[T]he end of most quarantines, travel restrictions, contact tracing, and other measures designed to keep ‘them’ from infecting ‘us,’ and the switch to measures like canceling mass events designed to keep us from infecting each other.”
Among the new messages that should be delivered are things like:
- Stockpiling nonperishable food and prescription meds.
- Considering care of sick family members.
- Cross-training work personnel so one person’s absence won’t derail an organization’s ability to function.
“We hope that governments and healthcare institutions are using this time wisely,” Sandman and Lanard continue. “We know that ordinary citizens are not being asked to do so. In most countries … ordinary citizens have not been asked to prepare. Instead, they have been led to expect that their governments will keep the virus from their doors.”
This article first appeared on Medscape.com.
It’s past time to call the novel coronavirus, COVID-19, a pandemic and “time to push people to prepare, and guide their prep,” according to risk communication experts.
Medical messaging about containing or stopping the spread of the virus is doing more harm than good, write Peter Sandman, PhD, and Jody Lanard, MD, both based in New York City, in a recent blog post.
“We are near-certain that the desperate-sounding last-ditch containment messaging of recent days is contributing to a massive global misperception,” they warn.
“The most crucial (and overdue) risk communication task … is to help people visualize their communities when ‘keeping it out’ – containment – is no longer relevant.”
That message is embraced by several experts who spoke to Medscape Medical News.
“I’m jealous of what [they] have written: It is so clear, so correct, and so practical,” said David Fisman, MD, MPH, professor of epidemiology at the University of Toronto, Canada. “I think WHO [World Health Organization] is shying away from the P word,” he continued, referring to the organization’s continuing decision not to call the outbreak a pandemic.
“I fully support exactly what [Sandman and Lanard] are saying,” said Michael Osterholm, PhD, MPH, professor of environmental health sciences and director of the Center for Infectious Disease Research and Policy (CIDRAP) at the University of Minnesota in Minneapolis.
Sandman and Lanard write. “Hardly any officials are telling civil society and the general public how to get ready for this pandemic.”
Effective communication should inform people of what to expect now, they continue: “[T]he end of most quarantines, travel restrictions, contact tracing, and other measures designed to keep ‘them’ from infecting ‘us,’ and the switch to measures like canceling mass events designed to keep us from infecting each other.”
Among the new messages that should be delivered are things like:
- Stockpiling nonperishable food and prescription meds.
- Considering care of sick family members.
- Cross-training work personnel so one person’s absence won’t derail an organization’s ability to function.
“We hope that governments and healthcare institutions are using this time wisely,” Sandman and Lanard continue. “We know that ordinary citizens are not being asked to do so. In most countries … ordinary citizens have not been asked to prepare. Instead, they have been led to expect that their governments will keep the virus from their doors.”
This article first appeared on Medscape.com.
CDC expects eventual community spread of coronavirus in U.S.
“We have for many weeks been saying that, while we hope this is not going to be severe, we are planning as if it is,” Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the CDC, said during a Feb. 25, 2020, telebriefing with reporters. “The data over the last week and the spread in other countries has certainly raised our level of concern and raised our level expectation that we are going to have community spread here.”
Dr. Messonnier noted that the coronavirus is now showing signs of community spread without a known source of exposure in a number of countries, including in Hong Kong, Iran, Italy, Japan, Singapore, South Korea, Taiwan, and Thailand. This has now raised the belief that there will be more widespread outbreaks in the United States.
“What we still don’t know is what that will look like,” she said. “As many of you know, we can have community spread in the United States and have it be reasonably mild. We can have community spread in the U.S. and have it be very severe. That is what we don’t completely know yet and we certainly also don’t exactly know when it is going to happen.”
She reiterated the number of actions being taken to slow the potential spread in the United States, including detecting, tracking, and isolating all cases, as well as restricting travel into the United States and issuing travel advisories for countries where coronavirus outbreaks are known.
“We are doing this with the goal of slowing the introduction of this new virus into the U.S. and buying us more time to prepare,” Dr. Messonnier said, noting the containment strategies have been largely successful, though it will be more difficult as more countries experience community spread of the virus.
Dr. Messonnier also reiterated that at this time there are no vaccines and no medicines to treat the coronavirus. She stressed the need to adhere to nonpharmaceutical interventions (NPIs), as they will be “the most important tools in our response to this virus.”
She said the NPIs will vary based on the severity of the outbreak in any given local community and include personal protective measures that individuals can take every day (many of which mirror the recommendations for preventing the spread of the seasonal flu virus), community NPIs that involve social distancing measures designed to keep people away from others, and environmental NPIs such as surface cleaning measures.
CDC’s latest warning comes as parent agency the Department of Health & Human Services is seeking $2.5 billion in funds from Congress to address the coronavirus outbreak.
During a separate press conference on the same day, HHS Secretary Alex Azar noted that there are five major priorities related to those funds, which would be used in the current year, including expansion of surveillance work within the influenza surveillance network; supporting public health preparedness and response for state and local governments; support the development of therapeutics and the development of vaccines; and the purchase of personal protective equipment for national stockpiles.
Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Disease at the National Institutes of Health, added during the press conference that vaccine work is in progress and could be ready for phase 1 testing within a month and a half. If all goes well, it would still be at least 12 - 18 months following the completion of a phase 2 trial before it could be produced for mass consumption.
“It is certainly conceivable that this issue with this coronavirus will go well beyond this season into next season,” Dr. Fauci said. “So a vaccine may not solve the problems of the next couple of months, but it certainly would be an important tool that we would have and we will keep you posted on that.”
He also mentioned that NIAID is looking at a number of candidates for therapeutic treatment of coronavirus. He highlighted Gilead’s remdesivir, a nucleotide analog, as one which undergoing two trials – a randomized controlled trial in China and a copy of that trial in Nebraska among patients with the coronavirus who were taken from the Diamond Princess cruise line in Japan.
“I am optimistic that we will at least get an answer if we do have do have a therapy that really is a gamechanger because then we could do something from the standpoint of intervention for those who are sick,” Dr. Fauci said.
UPDATE: This story was updated 2/25 at 4:51 p.m. ET
“We have for many weeks been saying that, while we hope this is not going to be severe, we are planning as if it is,” Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the CDC, said during a Feb. 25, 2020, telebriefing with reporters. “The data over the last week and the spread in other countries has certainly raised our level of concern and raised our level expectation that we are going to have community spread here.”
Dr. Messonnier noted that the coronavirus is now showing signs of community spread without a known source of exposure in a number of countries, including in Hong Kong, Iran, Italy, Japan, Singapore, South Korea, Taiwan, and Thailand. This has now raised the belief that there will be more widespread outbreaks in the United States.
“What we still don’t know is what that will look like,” she said. “As many of you know, we can have community spread in the United States and have it be reasonably mild. We can have community spread in the U.S. and have it be very severe. That is what we don’t completely know yet and we certainly also don’t exactly know when it is going to happen.”
She reiterated the number of actions being taken to slow the potential spread in the United States, including detecting, tracking, and isolating all cases, as well as restricting travel into the United States and issuing travel advisories for countries where coronavirus outbreaks are known.
“We are doing this with the goal of slowing the introduction of this new virus into the U.S. and buying us more time to prepare,” Dr. Messonnier said, noting the containment strategies have been largely successful, though it will be more difficult as more countries experience community spread of the virus.
Dr. Messonnier also reiterated that at this time there are no vaccines and no medicines to treat the coronavirus. She stressed the need to adhere to nonpharmaceutical interventions (NPIs), as they will be “the most important tools in our response to this virus.”
She said the NPIs will vary based on the severity of the outbreak in any given local community and include personal protective measures that individuals can take every day (many of which mirror the recommendations for preventing the spread of the seasonal flu virus), community NPIs that involve social distancing measures designed to keep people away from others, and environmental NPIs such as surface cleaning measures.
CDC’s latest warning comes as parent agency the Department of Health & Human Services is seeking $2.5 billion in funds from Congress to address the coronavirus outbreak.
During a separate press conference on the same day, HHS Secretary Alex Azar noted that there are five major priorities related to those funds, which would be used in the current year, including expansion of surveillance work within the influenza surveillance network; supporting public health preparedness and response for state and local governments; support the development of therapeutics and the development of vaccines; and the purchase of personal protective equipment for national stockpiles.
Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Disease at the National Institutes of Health, added during the press conference that vaccine work is in progress and could be ready for phase 1 testing within a month and a half. If all goes well, it would still be at least 12 - 18 months following the completion of a phase 2 trial before it could be produced for mass consumption.
“It is certainly conceivable that this issue with this coronavirus will go well beyond this season into next season,” Dr. Fauci said. “So a vaccine may not solve the problems of the next couple of months, but it certainly would be an important tool that we would have and we will keep you posted on that.”
He also mentioned that NIAID is looking at a number of candidates for therapeutic treatment of coronavirus. He highlighted Gilead’s remdesivir, a nucleotide analog, as one which undergoing two trials – a randomized controlled trial in China and a copy of that trial in Nebraska among patients with the coronavirus who were taken from the Diamond Princess cruise line in Japan.
“I am optimistic that we will at least get an answer if we do have do have a therapy that really is a gamechanger because then we could do something from the standpoint of intervention for those who are sick,” Dr. Fauci said.
UPDATE: This story was updated 2/25 at 4:51 p.m. ET
“We have for many weeks been saying that, while we hope this is not going to be severe, we are planning as if it is,” Nancy Messonnier, MD, director of the National Center for Immunization and Respiratory Diseases at the CDC, said during a Feb. 25, 2020, telebriefing with reporters. “The data over the last week and the spread in other countries has certainly raised our level of concern and raised our level expectation that we are going to have community spread here.”
Dr. Messonnier noted that the coronavirus is now showing signs of community spread without a known source of exposure in a number of countries, including in Hong Kong, Iran, Italy, Japan, Singapore, South Korea, Taiwan, and Thailand. This has now raised the belief that there will be more widespread outbreaks in the United States.
“What we still don’t know is what that will look like,” she said. “As many of you know, we can have community spread in the United States and have it be reasonably mild. We can have community spread in the U.S. and have it be very severe. That is what we don’t completely know yet and we certainly also don’t exactly know when it is going to happen.”
She reiterated the number of actions being taken to slow the potential spread in the United States, including detecting, tracking, and isolating all cases, as well as restricting travel into the United States and issuing travel advisories for countries where coronavirus outbreaks are known.
“We are doing this with the goal of slowing the introduction of this new virus into the U.S. and buying us more time to prepare,” Dr. Messonnier said, noting the containment strategies have been largely successful, though it will be more difficult as more countries experience community spread of the virus.
Dr. Messonnier also reiterated that at this time there are no vaccines and no medicines to treat the coronavirus. She stressed the need to adhere to nonpharmaceutical interventions (NPIs), as they will be “the most important tools in our response to this virus.”
She said the NPIs will vary based on the severity of the outbreak in any given local community and include personal protective measures that individuals can take every day (many of which mirror the recommendations for preventing the spread of the seasonal flu virus), community NPIs that involve social distancing measures designed to keep people away from others, and environmental NPIs such as surface cleaning measures.
CDC’s latest warning comes as parent agency the Department of Health & Human Services is seeking $2.5 billion in funds from Congress to address the coronavirus outbreak.
During a separate press conference on the same day, HHS Secretary Alex Azar noted that there are five major priorities related to those funds, which would be used in the current year, including expansion of surveillance work within the influenza surveillance network; supporting public health preparedness and response for state and local governments; support the development of therapeutics and the development of vaccines; and the purchase of personal protective equipment for national stockpiles.
Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Disease at the National Institutes of Health, added during the press conference that vaccine work is in progress and could be ready for phase 1 testing within a month and a half. If all goes well, it would still be at least 12 - 18 months following the completion of a phase 2 trial before it could be produced for mass consumption.
“It is certainly conceivable that this issue with this coronavirus will go well beyond this season into next season,” Dr. Fauci said. “So a vaccine may not solve the problems of the next couple of months, but it certainly would be an important tool that we would have and we will keep you posted on that.”
He also mentioned that NIAID is looking at a number of candidates for therapeutic treatment of coronavirus. He highlighted Gilead’s remdesivir, a nucleotide analog, as one which undergoing two trials – a randomized controlled trial in China and a copy of that trial in Nebraska among patients with the coronavirus who were taken from the Diamond Princess cruise line in Japan.
“I am optimistic that we will at least get an answer if we do have do have a therapy that really is a gamechanger because then we could do something from the standpoint of intervention for those who are sick,” Dr. Fauci said.
UPDATE: This story was updated 2/25 at 4:51 p.m. ET
ID Blog: SARS-CoV-2 – What’s in a name?
Coming up with a moniker for the new coronavirus shows the perils of naming names.
There is no Baby Book of Names or hurricane alphabet to readily name diseases and their causal entities. Throughout history and even in the modern era, a host of considerations have intruded on the decision as to what to call these blights upon humanity. Names have varied from inflammatory to misleading, from colloquial to scientific. And when it concerns a new epidemiological entity such as the latest coronavirus outbreak originating in China, health organizations, media, politicians, scientific taxonomy commissions, and the public at large all have a stake in the naming.
From “Wuhan virus” to “novel coronavirus-2019” to “COVID-19 virus,” the name of the new coronavirus that first appeared in China has been evolving to its now official designation: SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2). But where did the final name come from, how does such a name become official, and who makes it so?
Virus taxonomy
The Coronavirus Study Group (CSG) of the International Committee on Taxonomy of Viruses (ICTV) named the new coronavirus SARS-CoV-2 based upon its genetic relationship to the original SARS-CoV that caused an outbreak of disease in 2002–2003.
According to the ICTV website, the first internationally organized attempts to introduce order into the bewildering variety of viruses took place at the International Congress of Microbiology held in Moscow in 1966 where a committee was created that later became the ICTV and was given the task of developing a single, universal taxonomic scheme for all the viruses infecting animals, plants, fungi, bacteria, and archaea. The ICTV was created as a committee of the virology division of the International Union of Microbiological Societies and is governed by statutes approved by the virology division. Virus classification and nomenclature are subject to rules set out in an International Code.
These designate that: “The universal virus classification system shall employ the hierarchical levels of realm, subrealm, kingdom, subkingdom, phylum, subphylum, class, subclass, order, suborder, family, subfamily, genus, subgenus and species.”
Many of the topmost areas of classification are based on whether the viruses are DNA or RNA, single or double stranded, and have a simple protein shell or a complex lipoprotein envelope. Other levels of classification include host species, type of replication, and type of diseases they cause, the later exemplified in the SARS designation for this virus.
There are 98 international study groups (SGs) covering all major virus orders, families, and genera that are part of the ICTV, and it was the one dedicated to the single-stranded RNA coronaviruses, the CSG, that came up with the SARS-CoV-2 name and first referenced it in their Feb 11 publication in the Cold Springs Harbor preprint journal bioRxiv.
“Based on phylogeny, taxonomy and established practice, the CSG formally recognizes this virus as a sister to severe acute respiratory syndrome coronaviruses (SARS-CoVs) of the species severe acute respiratory syndrome–related coronavirus and designates it as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2),” they wrote.
According to the National Center for Biotechnology Information Taxonomy Browser, with respect to the original SARS CoV virus, of which this is a relative, the full taxonomic designation is: Viruses, Riboviria, Nidovirales, Cornidovirineae, Coronaviridae, Orthocoronavirinae, Betacoronavirus, Sarbecovirus.
The problem with naming names
The World Health Organization currently is not using the official scientific name of the virus, but rather is merely labeling it with regard to the disease: COVID-19, which simply refers to coronavirus disease 2019.
They are following a modern standard by which disease names avoid inflammatory connotations with people and places. Too often in the past from syphilis as the “French pox,” the 1918 influenza as the “Spanish flu,” AIDS as the “gay plague,” Middle East Respiratory Syndrome (MERS), and the currently named “WuFlu,” which made an appearance early in the new outbreak and which is symbolic of a sudden wave of anti-Asian, and specifically Chinese, prejudice.
Chinatown districts even in the United States are being affected economically through unwarranted fear associated with the virus. And there have been equivalently virulent outbreaks of hate speech against Asian individuals in places untouched by the new virus.
However, although SARS-CoV-2 as a name avoids such problems, different considerations led the WHO to reject it in its discussions, determining that its use ties it to tightly to the much more deadly SARS-CoV-1 virus in the public mind, risking greater fear and panic, especially in Asia, where SARS-CoV-1 had the biggest impact.
Back in 1896, William Sykes, MD, writing in the first flush of the triumph of germ theory in modern medicine, attempted to give some guidance to how medical science should best come up with new names of diseases by merging the demands of common parlance with those of taxonomic legitimacy. His “On the Origin and History of Disease-Names,” published in the Lancet, had clearcut advice: “It is vain to attempt to replace a folk name or one widely adopted by the people by a new one deliberately coined by scholars, and this for the following reasons: first, whatever names may be accepted by medical men must be translated by them into the vernacular of their patients, and by a resulting reaction the vernacular name comes to be the commoner one with themselves; and, secondly, there is no continuity or unchangeableness in the terms invented by savants, which are amended, improved upon, and displaced by the next writer on the subject, or, even more absurdly still, by the very inventors themselves in a subsequent publication.”
This is the reason that virus taxonomy provides names based upon unchangeable scientific descriptors of the actual disease causing entity, as illustrated by the decisions of the ICTV. In addition, the genomic sequences being provided by the scientific community are all being organized under the SARS-CoV-2 name and thus are cementing that moniker as the only acceptable scientific one.
Whether the rest of the world universally adopts SARS-CoV-2 as a name is still in question. If the outbreak spreads significantly beyond its current limits, fear and confusion – and simply the need for a more familiar-sounding label – may lead the general public to adopt more colloquial designations than those that science attempts to impose, as Dr. Sykes suggested back in 1896. That remains to be seen.
mlesney@mdedge.com
Mark Lesney is the managing editor of MDedge.com/IDPractioner. He has a PhD in plant virology and a PhD in the history of science, with a focus on the history of biotechnology and medicine. He has served as an adjunct assistant professor of the department of biochemistry and molecular & cellular biology at Georgetown University, Washington.
Coming up with a moniker for the new coronavirus shows the perils of naming names.
Coming up with a moniker for the new coronavirus shows the perils of naming names.
There is no Baby Book of Names or hurricane alphabet to readily name diseases and their causal entities. Throughout history and even in the modern era, a host of considerations have intruded on the decision as to what to call these blights upon humanity. Names have varied from inflammatory to misleading, from colloquial to scientific. And when it concerns a new epidemiological entity such as the latest coronavirus outbreak originating in China, health organizations, media, politicians, scientific taxonomy commissions, and the public at large all have a stake in the naming.
From “Wuhan virus” to “novel coronavirus-2019” to “COVID-19 virus,” the name of the new coronavirus that first appeared in China has been evolving to its now official designation: SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2). But where did the final name come from, how does such a name become official, and who makes it so?
Virus taxonomy
The Coronavirus Study Group (CSG) of the International Committee on Taxonomy of Viruses (ICTV) named the new coronavirus SARS-CoV-2 based upon its genetic relationship to the original SARS-CoV that caused an outbreak of disease in 2002–2003.
According to the ICTV website, the first internationally organized attempts to introduce order into the bewildering variety of viruses took place at the International Congress of Microbiology held in Moscow in 1966 where a committee was created that later became the ICTV and was given the task of developing a single, universal taxonomic scheme for all the viruses infecting animals, plants, fungi, bacteria, and archaea. The ICTV was created as a committee of the virology division of the International Union of Microbiological Societies and is governed by statutes approved by the virology division. Virus classification and nomenclature are subject to rules set out in an International Code.
These designate that: “The universal virus classification system shall employ the hierarchical levels of realm, subrealm, kingdom, subkingdom, phylum, subphylum, class, subclass, order, suborder, family, subfamily, genus, subgenus and species.”
Many of the topmost areas of classification are based on whether the viruses are DNA or RNA, single or double stranded, and have a simple protein shell or a complex lipoprotein envelope. Other levels of classification include host species, type of replication, and type of diseases they cause, the later exemplified in the SARS designation for this virus.
There are 98 international study groups (SGs) covering all major virus orders, families, and genera that are part of the ICTV, and it was the one dedicated to the single-stranded RNA coronaviruses, the CSG, that came up with the SARS-CoV-2 name and first referenced it in their Feb 11 publication in the Cold Springs Harbor preprint journal bioRxiv.
“Based on phylogeny, taxonomy and established practice, the CSG formally recognizes this virus as a sister to severe acute respiratory syndrome coronaviruses (SARS-CoVs) of the species severe acute respiratory syndrome–related coronavirus and designates it as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2),” they wrote.
According to the National Center for Biotechnology Information Taxonomy Browser, with respect to the original SARS CoV virus, of which this is a relative, the full taxonomic designation is: Viruses, Riboviria, Nidovirales, Cornidovirineae, Coronaviridae, Orthocoronavirinae, Betacoronavirus, Sarbecovirus.
The problem with naming names
The World Health Organization currently is not using the official scientific name of the virus, but rather is merely labeling it with regard to the disease: COVID-19, which simply refers to coronavirus disease 2019.
They are following a modern standard by which disease names avoid inflammatory connotations with people and places. Too often in the past from syphilis as the “French pox,” the 1918 influenza as the “Spanish flu,” AIDS as the “gay plague,” Middle East Respiratory Syndrome (MERS), and the currently named “WuFlu,” which made an appearance early in the new outbreak and which is symbolic of a sudden wave of anti-Asian, and specifically Chinese, prejudice.
Chinatown districts even in the United States are being affected economically through unwarranted fear associated with the virus. And there have been equivalently virulent outbreaks of hate speech against Asian individuals in places untouched by the new virus.
However, although SARS-CoV-2 as a name avoids such problems, different considerations led the WHO to reject it in its discussions, determining that its use ties it to tightly to the much more deadly SARS-CoV-1 virus in the public mind, risking greater fear and panic, especially in Asia, where SARS-CoV-1 had the biggest impact.
Back in 1896, William Sykes, MD, writing in the first flush of the triumph of germ theory in modern medicine, attempted to give some guidance to how medical science should best come up with new names of diseases by merging the demands of common parlance with those of taxonomic legitimacy. His “On the Origin and History of Disease-Names,” published in the Lancet, had clearcut advice: “It is vain to attempt to replace a folk name or one widely adopted by the people by a new one deliberately coined by scholars, and this for the following reasons: first, whatever names may be accepted by medical men must be translated by them into the vernacular of their patients, and by a resulting reaction the vernacular name comes to be the commoner one with themselves; and, secondly, there is no continuity or unchangeableness in the terms invented by savants, which are amended, improved upon, and displaced by the next writer on the subject, or, even more absurdly still, by the very inventors themselves in a subsequent publication.”
This is the reason that virus taxonomy provides names based upon unchangeable scientific descriptors of the actual disease causing entity, as illustrated by the decisions of the ICTV. In addition, the genomic sequences being provided by the scientific community are all being organized under the SARS-CoV-2 name and thus are cementing that moniker as the only acceptable scientific one.
Whether the rest of the world universally adopts SARS-CoV-2 as a name is still in question. If the outbreak spreads significantly beyond its current limits, fear and confusion – and simply the need for a more familiar-sounding label – may lead the general public to adopt more colloquial designations than those that science attempts to impose, as Dr. Sykes suggested back in 1896. That remains to be seen.
mlesney@mdedge.com
Mark Lesney is the managing editor of MDedge.com/IDPractioner. He has a PhD in plant virology and a PhD in the history of science, with a focus on the history of biotechnology and medicine. He has served as an adjunct assistant professor of the department of biochemistry and molecular & cellular biology at Georgetown University, Washington.
There is no Baby Book of Names or hurricane alphabet to readily name diseases and their causal entities. Throughout history and even in the modern era, a host of considerations have intruded on the decision as to what to call these blights upon humanity. Names have varied from inflammatory to misleading, from colloquial to scientific. And when it concerns a new epidemiological entity such as the latest coronavirus outbreak originating in China, health organizations, media, politicians, scientific taxonomy commissions, and the public at large all have a stake in the naming.
From “Wuhan virus” to “novel coronavirus-2019” to “COVID-19 virus,” the name of the new coronavirus that first appeared in China has been evolving to its now official designation: SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2). But where did the final name come from, how does such a name become official, and who makes it so?
Virus taxonomy
The Coronavirus Study Group (CSG) of the International Committee on Taxonomy of Viruses (ICTV) named the new coronavirus SARS-CoV-2 based upon its genetic relationship to the original SARS-CoV that caused an outbreak of disease in 2002–2003.
According to the ICTV website, the first internationally organized attempts to introduce order into the bewildering variety of viruses took place at the International Congress of Microbiology held in Moscow in 1966 where a committee was created that later became the ICTV and was given the task of developing a single, universal taxonomic scheme for all the viruses infecting animals, plants, fungi, bacteria, and archaea. The ICTV was created as a committee of the virology division of the International Union of Microbiological Societies and is governed by statutes approved by the virology division. Virus classification and nomenclature are subject to rules set out in an International Code.
These designate that: “The universal virus classification system shall employ the hierarchical levels of realm, subrealm, kingdom, subkingdom, phylum, subphylum, class, subclass, order, suborder, family, subfamily, genus, subgenus and species.”
Many of the topmost areas of classification are based on whether the viruses are DNA or RNA, single or double stranded, and have a simple protein shell or a complex lipoprotein envelope. Other levels of classification include host species, type of replication, and type of diseases they cause, the later exemplified in the SARS designation for this virus.
There are 98 international study groups (SGs) covering all major virus orders, families, and genera that are part of the ICTV, and it was the one dedicated to the single-stranded RNA coronaviruses, the CSG, that came up with the SARS-CoV-2 name and first referenced it in their Feb 11 publication in the Cold Springs Harbor preprint journal bioRxiv.
“Based on phylogeny, taxonomy and established practice, the CSG formally recognizes this virus as a sister to severe acute respiratory syndrome coronaviruses (SARS-CoVs) of the species severe acute respiratory syndrome–related coronavirus and designates it as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2),” they wrote.
According to the National Center for Biotechnology Information Taxonomy Browser, with respect to the original SARS CoV virus, of which this is a relative, the full taxonomic designation is: Viruses, Riboviria, Nidovirales, Cornidovirineae, Coronaviridae, Orthocoronavirinae, Betacoronavirus, Sarbecovirus.
The problem with naming names
The World Health Organization currently is not using the official scientific name of the virus, but rather is merely labeling it with regard to the disease: COVID-19, which simply refers to coronavirus disease 2019.
They are following a modern standard by which disease names avoid inflammatory connotations with people and places. Too often in the past from syphilis as the “French pox,” the 1918 influenza as the “Spanish flu,” AIDS as the “gay plague,” Middle East Respiratory Syndrome (MERS), and the currently named “WuFlu,” which made an appearance early in the new outbreak and which is symbolic of a sudden wave of anti-Asian, and specifically Chinese, prejudice.
Chinatown districts even in the United States are being affected economically through unwarranted fear associated with the virus. And there have been equivalently virulent outbreaks of hate speech against Asian individuals in places untouched by the new virus.
However, although SARS-CoV-2 as a name avoids such problems, different considerations led the WHO to reject it in its discussions, determining that its use ties it to tightly to the much more deadly SARS-CoV-1 virus in the public mind, risking greater fear and panic, especially in Asia, where SARS-CoV-1 had the biggest impact.
Back in 1896, William Sykes, MD, writing in the first flush of the triumph of germ theory in modern medicine, attempted to give some guidance to how medical science should best come up with new names of diseases by merging the demands of common parlance with those of taxonomic legitimacy. His “On the Origin and History of Disease-Names,” published in the Lancet, had clearcut advice: “It is vain to attempt to replace a folk name or one widely adopted by the people by a new one deliberately coined by scholars, and this for the following reasons: first, whatever names may be accepted by medical men must be translated by them into the vernacular of their patients, and by a resulting reaction the vernacular name comes to be the commoner one with themselves; and, secondly, there is no continuity or unchangeableness in the terms invented by savants, which are amended, improved upon, and displaced by the next writer on the subject, or, even more absurdly still, by the very inventors themselves in a subsequent publication.”
This is the reason that virus taxonomy provides names based upon unchangeable scientific descriptors of the actual disease causing entity, as illustrated by the decisions of the ICTV. In addition, the genomic sequences being provided by the scientific community are all being organized under the SARS-CoV-2 name and thus are cementing that moniker as the only acceptable scientific one.
Whether the rest of the world universally adopts SARS-CoV-2 as a name is still in question. If the outbreak spreads significantly beyond its current limits, fear and confusion – and simply the need for a more familiar-sounding label – may lead the general public to adopt more colloquial designations than those that science attempts to impose, as Dr. Sykes suggested back in 1896. That remains to be seen.
mlesney@mdedge.com
Mark Lesney is the managing editor of MDedge.com/IDPractioner. He has a PhD in plant virology and a PhD in the history of science, with a focus on the history of biotechnology and medicine. He has served as an adjunct assistant professor of the department of biochemistry and molecular & cellular biology at Georgetown University, Washington.
China’s health authorities release large coronavirus case series
The Chinese Center for Disease Control and Prevention has released the largest case series to date for novel coronavirus 2019 (COVID-19), and a summary of key findings appears in JAMA.
- The virus, which spread from a single city to a whole country in only 30 days, has so far has caused over 72,314 cases as of Feb. 11, 2020, and 1,023 fatalities (2.3%) overall.
- The age distribution shows that most of the cases (87%) occurred in patients aged 30-79 years, while 10% were in patients 29 years and younger and 3% at 80 years and older.
- Following the SARS outbreak in 2002-2003, the Chinese government adjusted its epidemic response protocol. For example, according to the summary, while there were 300 cases and 5 deaths with SARS before the Chinese government reported it to the World Health Organization, there were only 27 cases and no deaths with COVID-19 before it was reported to that agency.
- A major goal, the authors wrote, is to buy enough time for scientific research, hopefully before the disease has become too widespread.
The summary argues that, while some measures the Chinese government has taken could be seen as extreme, the overall benefits and lives saved outweigh the potential infringement on civil liberties. It also suggests that countries need to work together in situations like this because disease pathogens do not respect geopolitical borders.
SOURCE: Wu Z, McGoogan JM. JAMA. 2020 Feb 24. doi: 10.1001/jama.2020.2648.
The Chinese Center for Disease Control and Prevention has released the largest case series to date for novel coronavirus 2019 (COVID-19), and a summary of key findings appears in JAMA.
- The virus, which spread from a single city to a whole country in only 30 days, has so far has caused over 72,314 cases as of Feb. 11, 2020, and 1,023 fatalities (2.3%) overall.
- The age distribution shows that most of the cases (87%) occurred in patients aged 30-79 years, while 10% were in patients 29 years and younger and 3% at 80 years and older.
- Following the SARS outbreak in 2002-2003, the Chinese government adjusted its epidemic response protocol. For example, according to the summary, while there were 300 cases and 5 deaths with SARS before the Chinese government reported it to the World Health Organization, there were only 27 cases and no deaths with COVID-19 before it was reported to that agency.
- A major goal, the authors wrote, is to buy enough time for scientific research, hopefully before the disease has become too widespread.
The summary argues that, while some measures the Chinese government has taken could be seen as extreme, the overall benefits and lives saved outweigh the potential infringement on civil liberties. It also suggests that countries need to work together in situations like this because disease pathogens do not respect geopolitical borders.
SOURCE: Wu Z, McGoogan JM. JAMA. 2020 Feb 24. doi: 10.1001/jama.2020.2648.
The Chinese Center for Disease Control and Prevention has released the largest case series to date for novel coronavirus 2019 (COVID-19), and a summary of key findings appears in JAMA.
- The virus, which spread from a single city to a whole country in only 30 days, has so far has caused over 72,314 cases as of Feb. 11, 2020, and 1,023 fatalities (2.3%) overall.
- The age distribution shows that most of the cases (87%) occurred in patients aged 30-79 years, while 10% were in patients 29 years and younger and 3% at 80 years and older.
- Following the SARS outbreak in 2002-2003, the Chinese government adjusted its epidemic response protocol. For example, according to the summary, while there were 300 cases and 5 deaths with SARS before the Chinese government reported it to the World Health Organization, there were only 27 cases and no deaths with COVID-19 before it was reported to that agency.
- A major goal, the authors wrote, is to buy enough time for scientific research, hopefully before the disease has become too widespread.
The summary argues that, while some measures the Chinese government has taken could be seen as extreme, the overall benefits and lives saved outweigh the potential infringement on civil liberties. It also suggests that countries need to work together in situations like this because disease pathogens do not respect geopolitical borders.
SOURCE: Wu Z, McGoogan JM. JAMA. 2020 Feb 24. doi: 10.1001/jama.2020.2648.
FROM JAMA
WHO urges that ‘window of opportunity’ on containing novel coronavirus not be missed
As of 6 a.m. Geneva time, Feb. 21, China reported 75,567 cases of COVID-19 and 2,239 deaths, including a total of 892 new confirmed cases that were reported in China in the past 24 hours, with 118 deaths, stated Tedros Adhanom Ghebreyesus, MD, World Health Organization Director-General, in a Feb. 21 news conference on the COVID-19 outbreak.
What he described as “the significant decline in newly reported cases” is partly because of a change in reporting in which China switched from including “clinically diagnosed” cases to reporting only “suspected” and “laboratory confirmed cases.” The reporting procedure changed because the medical facilities in Wuhan regained the capability of checking all suspected cases with laboratory tests. As a result, some cases that were clinically confirmed were subtracted from the total because they tested negative, said Dr. Ghebreyesus.
Although the number of cases in Hubuei province continues to decline, the WHO is concerned about an increase seen in Shandong province and they are seeking more information. Outside China, there are now 1,152 cases in 26 countries and eight deaths. “Although the number of cases outside of China remains small, we are concerned about the cases with no clear epidemiological link, such as travel history to China, or contact with a confirmed case,” said Dr. Ghebreyesus. “Apart from the Diamond Princess cruise ship, the Republic of Korea now has the most cases outside China, and we are working closely with that government to understand the transmission dynamics that led to this increase.”
“We are also concerned about the increase of cases in the Islamic Republic of Iran, where there are now 18 cases and four deaths in just the past 2 days.”
“Our particular concern is for COVID-19 to spread in countries with weaker health systems,” he said, adding that tomorrow, he will address an emergency meeting of African health ministers held jointly by the African Union and the African Centres for Disease Control and Prevention on dealing with COVID-19.
Dr. Ghebreyesus also announced that today the WHO has designated six special envoys on COVID-19 to provide strategic advice and high-level political advocacy and engagement in different parts of the world.
In his remarks, Dr. Ghebreyesus particularly stressed that: “The measures that China and other countries have taken have given us a fighting chance of containing the spread of the virus. We call on all countries to continue their commitment for containment measures, while preparing for community transmission if it occurs. We must not look back and regret that we failed to take advantage of the window of opportunity that we have now.”
In the question and answer period, Dr. Ghebreyesus specifically addressed the issue of misinformation and conspiracy theories being promulgated by certain individuals and on social media about the source of the virus, especially those people who believe that it was designed in a Chinese virus laboratory. Scientists play an important role in refuting such particular misinformation, he said, and research must continue to track down the actual source in nature.
To that regard, a paper published online in the Lancet on Feb. 19, provided a consensus statement by more than 25 health scientists outside of China stating: “We stand together to strongly condemn conspiracy theories suggesting that COVID-19 does not have a natural origin. Scientists from multiple countries have published and analyzed genomes of the causative agent, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and they overwhelmingly conclude that this coronavirus originated in wildlife, as have so many other emerging pathogens.”
The WHO issues daily coronavirus disease (COVID-2019) situation reports on its website and provides these telebriefing updates daily.
As of 6 a.m. Geneva time, Feb. 21, China reported 75,567 cases of COVID-19 and 2,239 deaths, including a total of 892 new confirmed cases that were reported in China in the past 24 hours, with 118 deaths, stated Tedros Adhanom Ghebreyesus, MD, World Health Organization Director-General, in a Feb. 21 news conference on the COVID-19 outbreak.
What he described as “the significant decline in newly reported cases” is partly because of a change in reporting in which China switched from including “clinically diagnosed” cases to reporting only “suspected” and “laboratory confirmed cases.” The reporting procedure changed because the medical facilities in Wuhan regained the capability of checking all suspected cases with laboratory tests. As a result, some cases that were clinically confirmed were subtracted from the total because they tested negative, said Dr. Ghebreyesus.
Although the number of cases in Hubuei province continues to decline, the WHO is concerned about an increase seen in Shandong province and they are seeking more information. Outside China, there are now 1,152 cases in 26 countries and eight deaths. “Although the number of cases outside of China remains small, we are concerned about the cases with no clear epidemiological link, such as travel history to China, or contact with a confirmed case,” said Dr. Ghebreyesus. “Apart from the Diamond Princess cruise ship, the Republic of Korea now has the most cases outside China, and we are working closely with that government to understand the transmission dynamics that led to this increase.”
“We are also concerned about the increase of cases in the Islamic Republic of Iran, where there are now 18 cases and four deaths in just the past 2 days.”
“Our particular concern is for COVID-19 to spread in countries with weaker health systems,” he said, adding that tomorrow, he will address an emergency meeting of African health ministers held jointly by the African Union and the African Centres for Disease Control and Prevention on dealing with COVID-19.
Dr. Ghebreyesus also announced that today the WHO has designated six special envoys on COVID-19 to provide strategic advice and high-level political advocacy and engagement in different parts of the world.
In his remarks, Dr. Ghebreyesus particularly stressed that: “The measures that China and other countries have taken have given us a fighting chance of containing the spread of the virus. We call on all countries to continue their commitment for containment measures, while preparing for community transmission if it occurs. We must not look back and regret that we failed to take advantage of the window of opportunity that we have now.”
In the question and answer period, Dr. Ghebreyesus specifically addressed the issue of misinformation and conspiracy theories being promulgated by certain individuals and on social media about the source of the virus, especially those people who believe that it was designed in a Chinese virus laboratory. Scientists play an important role in refuting such particular misinformation, he said, and research must continue to track down the actual source in nature.
To that regard, a paper published online in the Lancet on Feb. 19, provided a consensus statement by more than 25 health scientists outside of China stating: “We stand together to strongly condemn conspiracy theories suggesting that COVID-19 does not have a natural origin. Scientists from multiple countries have published and analyzed genomes of the causative agent, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and they overwhelmingly conclude that this coronavirus originated in wildlife, as have so many other emerging pathogens.”
The WHO issues daily coronavirus disease (COVID-2019) situation reports on its website and provides these telebriefing updates daily.
As of 6 a.m. Geneva time, Feb. 21, China reported 75,567 cases of COVID-19 and 2,239 deaths, including a total of 892 new confirmed cases that were reported in China in the past 24 hours, with 118 deaths, stated Tedros Adhanom Ghebreyesus, MD, World Health Organization Director-General, in a Feb. 21 news conference on the COVID-19 outbreak.
What he described as “the significant decline in newly reported cases” is partly because of a change in reporting in which China switched from including “clinically diagnosed” cases to reporting only “suspected” and “laboratory confirmed cases.” The reporting procedure changed because the medical facilities in Wuhan regained the capability of checking all suspected cases with laboratory tests. As a result, some cases that were clinically confirmed were subtracted from the total because they tested negative, said Dr. Ghebreyesus.
Although the number of cases in Hubuei province continues to decline, the WHO is concerned about an increase seen in Shandong province and they are seeking more information. Outside China, there are now 1,152 cases in 26 countries and eight deaths. “Although the number of cases outside of China remains small, we are concerned about the cases with no clear epidemiological link, such as travel history to China, or contact with a confirmed case,” said Dr. Ghebreyesus. “Apart from the Diamond Princess cruise ship, the Republic of Korea now has the most cases outside China, and we are working closely with that government to understand the transmission dynamics that led to this increase.”
“We are also concerned about the increase of cases in the Islamic Republic of Iran, where there are now 18 cases and four deaths in just the past 2 days.”
“Our particular concern is for COVID-19 to spread in countries with weaker health systems,” he said, adding that tomorrow, he will address an emergency meeting of African health ministers held jointly by the African Union and the African Centres for Disease Control and Prevention on dealing with COVID-19.
Dr. Ghebreyesus also announced that today the WHO has designated six special envoys on COVID-19 to provide strategic advice and high-level political advocacy and engagement in different parts of the world.
In his remarks, Dr. Ghebreyesus particularly stressed that: “The measures that China and other countries have taken have given us a fighting chance of containing the spread of the virus. We call on all countries to continue their commitment for containment measures, while preparing for community transmission if it occurs. We must not look back and regret that we failed to take advantage of the window of opportunity that we have now.”
In the question and answer period, Dr. Ghebreyesus specifically addressed the issue of misinformation and conspiracy theories being promulgated by certain individuals and on social media about the source of the virus, especially those people who believe that it was designed in a Chinese virus laboratory. Scientists play an important role in refuting such particular misinformation, he said, and research must continue to track down the actual source in nature.
To that regard, a paper published online in the Lancet on Feb. 19, provided a consensus statement by more than 25 health scientists outside of China stating: “We stand together to strongly condemn conspiracy theories suggesting that COVID-19 does not have a natural origin. Scientists from multiple countries have published and analyzed genomes of the causative agent, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and they overwhelmingly conclude that this coronavirus originated in wildlife, as have so many other emerging pathogens.”
The WHO issues daily coronavirus disease (COVID-2019) situation reports on its website and provides these telebriefing updates daily.
REPORTING FROM A WHO TELEBRIEFING
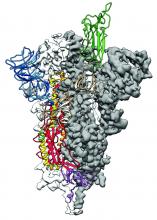
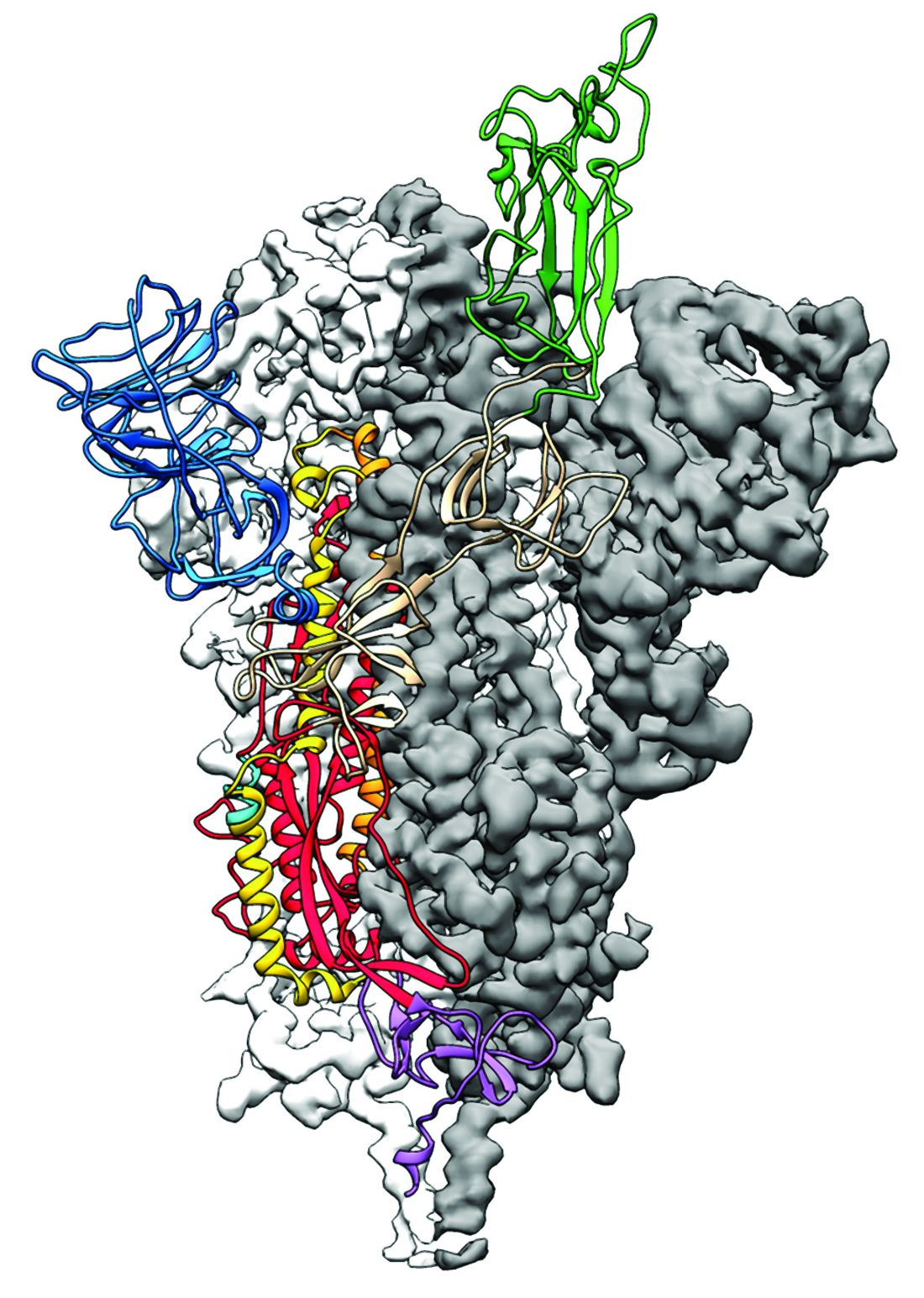
2019-nCoV: Structure, characteristics of key potential therapy target determined
Researchers have identified the structure of a protein that could turn out to be a potential vaccine target for the 2019-nCoV.
As is typical of other coronaviruses, 2019-nCoV makes use of a densely glycosylated spike protein to gain entry into host cells. The spike protein is a trimeric class I fusion protein that exists in a metastable prefusion conformation that undergoes a dramatic structural rearrangement to fuse the viral membrane with the host-cell membrane, according to Daniel Wrapp of the University of Texas at Austin and colleagues.
The researchers performed a study to synthesize and determine the 3-D structure of the spike protein because it is a logical target for vaccine development and for the development of targeted therapeutics for COVID-19, the disease caused by the virus.
“As soon as we knew this was a coronavirus, we felt we had to jump at it,” senior author Jason S. McLellan, PhD, associate professor of molecular science, said in a press release from the University, “because we could be one of the first ones to get this structure. We knew exactly what mutations to put into this because we’ve already shown these mutations work for a bunch of other coronaviruses.”
Because recent reports by other researchers demonstrated that 2019-nCoV and SARS-CoV spike proteins share the same functional host-cell receptor–angiotensin-converting enzyme 2 (ACE2), Dr. McLellan and his colleagues examined the relation between the two viruses. They found biophysical and structural evidence that the 2019-nCoV spike protein binds ACE2 with higher affinity than the closely related SARS-CoV spike protein. “The high affinity of 2019-nCoV S for human ACE2 may contribute to the apparent ease with which 2019-nCoV can spread from human-to-human; however, additional studies are needed to investigate this possibility,” the researchers wrote.
Focusing their attention on the receptor-binding domain (RBD) of the 2019-nCoV spike protein, they tested several published SARS-CoV RBD-specific monoclonal antibodies against it and found that these antibodies showed no appreciable binding to 2019-nCoV spike protein, which suggests limited antibody cross-reactivity. For this reason, they suggested that future antibody isolation and therapeutic design efforts will benefit from specifically using 2019-nCoV spike proteins as probes.
“This information will support precision vaccine design and discovery of anti-viral therapeutics, accelerating medical countermeasure development,” they concluded.
The research was supported in part by an National Institutes of Health/National Institute of Allergy and Infectious Diseases grant and by intramural funding from the National Institute of Allergy and Infectious Diseases. Four authors are inventors on US patent application No. 62/412,703 (Prefusion Coronavirus Spike Proteins and Their Use) and all are inventors on US patent application No. 62/972,886 (2019-nCoV Vaccine).
SOURCE: Wrapp D et al. Science. 2020 Feb 19. doi: 10.1126/science.abb2507.
Researchers have identified the structure of a protein that could turn out to be a potential vaccine target for the 2019-nCoV.
As is typical of other coronaviruses, 2019-nCoV makes use of a densely glycosylated spike protein to gain entry into host cells. The spike protein is a trimeric class I fusion protein that exists in a metastable prefusion conformation that undergoes a dramatic structural rearrangement to fuse the viral membrane with the host-cell membrane, according to Daniel Wrapp of the University of Texas at Austin and colleagues.
The researchers performed a study to synthesize and determine the 3-D structure of the spike protein because it is a logical target for vaccine development and for the development of targeted therapeutics for COVID-19, the disease caused by the virus.
“As soon as we knew this was a coronavirus, we felt we had to jump at it,” senior author Jason S. McLellan, PhD, associate professor of molecular science, said in a press release from the University, “because we could be one of the first ones to get this structure. We knew exactly what mutations to put into this because we’ve already shown these mutations work for a bunch of other coronaviruses.”
Because recent reports by other researchers demonstrated that 2019-nCoV and SARS-CoV spike proteins share the same functional host-cell receptor–angiotensin-converting enzyme 2 (ACE2), Dr. McLellan and his colleagues examined the relation between the two viruses. They found biophysical and structural evidence that the 2019-nCoV spike protein binds ACE2 with higher affinity than the closely related SARS-CoV spike protein. “The high affinity of 2019-nCoV S for human ACE2 may contribute to the apparent ease with which 2019-nCoV can spread from human-to-human; however, additional studies are needed to investigate this possibility,” the researchers wrote.
Focusing their attention on the receptor-binding domain (RBD) of the 2019-nCoV spike protein, they tested several published SARS-CoV RBD-specific monoclonal antibodies against it and found that these antibodies showed no appreciable binding to 2019-nCoV spike protein, which suggests limited antibody cross-reactivity. For this reason, they suggested that future antibody isolation and therapeutic design efforts will benefit from specifically using 2019-nCoV spike proteins as probes.
“This information will support precision vaccine design and discovery of anti-viral therapeutics, accelerating medical countermeasure development,” they concluded.
The research was supported in part by an National Institutes of Health/National Institute of Allergy and Infectious Diseases grant and by intramural funding from the National Institute of Allergy and Infectious Diseases. Four authors are inventors on US patent application No. 62/412,703 (Prefusion Coronavirus Spike Proteins and Their Use) and all are inventors on US patent application No. 62/972,886 (2019-nCoV Vaccine).
SOURCE: Wrapp D et al. Science. 2020 Feb 19. doi: 10.1126/science.abb2507.
Researchers have identified the structure of a protein that could turn out to be a potential vaccine target for the 2019-nCoV.
As is typical of other coronaviruses, 2019-nCoV makes use of a densely glycosylated spike protein to gain entry into host cells. The spike protein is a trimeric class I fusion protein that exists in a metastable prefusion conformation that undergoes a dramatic structural rearrangement to fuse the viral membrane with the host-cell membrane, according to Daniel Wrapp of the University of Texas at Austin and colleagues.
The researchers performed a study to synthesize and determine the 3-D structure of the spike protein because it is a logical target for vaccine development and for the development of targeted therapeutics for COVID-19, the disease caused by the virus.
“As soon as we knew this was a coronavirus, we felt we had to jump at it,” senior author Jason S. McLellan, PhD, associate professor of molecular science, said in a press release from the University, “because we could be one of the first ones to get this structure. We knew exactly what mutations to put into this because we’ve already shown these mutations work for a bunch of other coronaviruses.”
Because recent reports by other researchers demonstrated that 2019-nCoV and SARS-CoV spike proteins share the same functional host-cell receptor–angiotensin-converting enzyme 2 (ACE2), Dr. McLellan and his colleagues examined the relation between the two viruses. They found biophysical and structural evidence that the 2019-nCoV spike protein binds ACE2 with higher affinity than the closely related SARS-CoV spike protein. “The high affinity of 2019-nCoV S for human ACE2 may contribute to the apparent ease with which 2019-nCoV can spread from human-to-human; however, additional studies are needed to investigate this possibility,” the researchers wrote.
Focusing their attention on the receptor-binding domain (RBD) of the 2019-nCoV spike protein, they tested several published SARS-CoV RBD-specific monoclonal antibodies against it and found that these antibodies showed no appreciable binding to 2019-nCoV spike protein, which suggests limited antibody cross-reactivity. For this reason, they suggested that future antibody isolation and therapeutic design efforts will benefit from specifically using 2019-nCoV spike proteins as probes.
“This information will support precision vaccine design and discovery of anti-viral therapeutics, accelerating medical countermeasure development,” they concluded.
The research was supported in part by an National Institutes of Health/National Institute of Allergy and Infectious Diseases grant and by intramural funding from the National Institute of Allergy and Infectious Diseases. Four authors are inventors on US patent application No. 62/412,703 (Prefusion Coronavirus Spike Proteins and Their Use) and all are inventors on US patent application No. 62/972,886 (2019-nCoV Vaccine).
SOURCE: Wrapp D et al. Science. 2020 Feb 19. doi: 10.1126/science.abb2507.
FROM SCIENCE
Doctors look to existing drugs in coronavirus fight
COVID-19, the infection caused by the newly identified coronavirus, is a currently a disease with no pharmaceutical weapons against it. There’s no vaccine to prevent it, and no drugs can treat it.
But researchers are racing to change that. A vaccine could be ready to test as soon as April. More than two dozen studies have already been registered on ClinicalTrials.gov, a website that tracks research. These studies aim to test everything from traditional Chinese medicine to vitamin C, stem cells, steroids, and medications that fight other viruses, like the flu and HIV. The hope is that something about how these repurposed remedies work will help patients who are desperately ill with no other prospects.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, says this is all part of the playbook for brand-new diseases. “There’s a lot of empiric guessing,” he says. “They’re going to propose a whole lot of drugs that already exist. They’re going to say, here’s the data that shows it blocks the virus” in a test tube. But test tubes aren’t people, and many drugs that seem to work in a lab won’t end up helping patients.
Coronaviruses are especially hard to stop once they invade the body. Unlike many other kinds of viruses, they have a fail-safe against tampering – a “proofreader” that constantly inspects their code, looking for errors, including the potentially life-saving errors that drugs could introduce.
Dr. Fauci said that researchers will be able to make better guesses about how to help people when they can try drugs in animals. “We don’t have an animal model yet of the new coronavirus. When we do get an animal model, that will be a big boon to drugs because then, you can clearly test them in a physiological way, whether they work,” he says.
Looking to drugs for HIV and flu
One of the drugs already under study is the combination of two HIV medications: lopinavir and ritonavir (Kaletra). Kaletra stops viruses by interfering with the enzymes they need to infect cells, called proteases.
One study being done at the Guangzhou Eighth People’s Hospital in China is testing Kaletra against Arbidol, an antiviral drug approved in China and Russia to treat the flu. Two groups of patients will take the medications along with standard care. A third group in the study will receive only standard care, typically supportive therapy with oxygen and IV fluids that are meant to support the body so the immune system can fight off a virus on its own.
An Ebola drug gets a second look
One repurposed drug generating a lot of buzz is an experimental infusion called remdesivir (Xembify). It was originally tested against the Ebola virus. While it didn’t work for that infection, it has been shown to shut down the new coronavirus, at least in test tubes. It’s been given to a small number of COVID-19 patients already, including one in Washington state.
In order to have better evidence of how well it may work in people, two studies in Beijing are comparing remdesivir to a dummy pill to see if the drug can help patients with both mild and severe symptoms recover from their illnesses. Viruses work by infecting cells, taking over their machinery, and getting them to crank out more copies of the virus, which then goes on to infect more cells. Remdesivir is a mimic that fools a virus into replacing one of its four building blocks with a chemical fake. Once in the virus’s blueprints, the imposter acts like a stop sign that keeps the virus from copying itself.
Other kinds of drugs in the same class – called nucleotide analogs – are used to attack cancer and other infectious viruses like hepatitis.
Last week, Chinese scientists published study showing remdesivir was effective against the new coronavirus, 2019-nCoV. Out of seven drugs tested, only remdesivir and an older drug called chloroquine (Aralen), which is used to treat malaria, worked, at least in test tubes. “It functions like a knife that just cuts off the RNA strand,” says Mark Denison, MD, a pediatric infectious disease specialist at Vanderbilt University in Nashville. “They can’t replicate any more. It stops them from doing that.” Dr. Denison is part of a team of researchers in Tennessee and North Carolina that discovered remdesivir could stop coronaviruses, like severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS), in test tubes and animals. He has studied coronaviruses in his lab for 30 years. He knew they would pose a threat again. “We’re shocked, but not surprised, that this has happened again,” he says of the China-based outbreak of 2019-nCoV.
After the SARS outbreak, which infected more than 8,000 people in 26 countries during 2002-2003, and MERS, which has infected nearly 2,500 people in 27 countries since 2012, researchers knew they had to start looking for treatments that would work against coronaviruses. Dr. Denison reached out to Gilead Sciences, a company best known for its antiviral medications that treat HIV and hepatitis C, and asked it to send drug candidates for him to test on coronaviruses. “The idea was that we didn’t want a drug that would just work against SARS or MERS,” he says. “We wanted drugs that worked against every coronavirus.”
Many of the agents he tried didn’t work until Dr. Denison and his team knocked out the virus’s pesky proofreader. Remdesivir seems to be able to defeat the proofreader, though Dr. Denison admits that he does not know how the drug gets around a virus’s defenses. He has a grant from the National Institutes of Health to study that. Gilead has been giving remdesivir to “a small number” of coronavirus patients in the United States and Europe on a compassionate basis.
One of those patients was a 35-year-old man in Everett, Wash., who had gotten pneumonia after being infected with the new coronavirus during a trip to see family in Wuhan, China, the epicenter of the outbreak. His doctors started IV remdesivir on the evening of his 7th day in the hospital. On the 8th day, he improved. He was well enough to stop using oxygen. Signs of pneumonia were gone. He got his appetite back. His case was recently published in the New England Journal of Medicine, igniting a firestorm of interest in the therapy.
Unfortunately, though, even Dr. Denison says a single person’s case isn’t enough proof that the medication can treat the new coronavirus. The patient, who has not been identified, was getting expert care. He may have improved on his own, despite getting the drug. He said the challenge in people will be to find out two things: whether the medication can block the spread of virus in the body and whether it can reverse the disease. “You can remove the source of injury, but you still have the injury,” he said. Other important questions include how soon the drug may need to be given after infection for it work and whether it may cause significant side effects.
A promising pill
Another drug, a nucleoside analog, that appears to be able to defeat the coronavirus proofreader, EIDD-2801, was developed by Emory University in Atlanta. It was originally intended to treat the flu but has shown some effectiveness against coronaviruses like SARS and MERS.
The FDA recently reached out to Emory asking if it had any drug candidates that might work against the new coronavirus. “It’s a good shot on goal here,” says George Painter, PhD, CEO of Drug Innovation Ventures at Emory. EIDD-2801 can be taken as a pill, which makes it easier to use outside of a hospital setting.
“The capsules for the trial are being made at the end of this month. So we’re close,” Painter says. “We’re right on the edge.”
While these early tests are just getting started, and it will be months until researchers have results, the World Health Organization has sounded a note of caution.
In new guidelines for the clinical management of COVID-19, the WHO reminded doctors and patients that there’s not enough evidence to recommend any specific treatment for infected patients.
Right now, the guidelines recommend that doctors offer supportive care to help the body fight off an infection on its own.
The organization says unlicensed treatments should be given only in the context of clinical trials that have been ethically reviewed or with strict clinical monitoring in emergencies.
This article first appeared on WebMD.com.
COVID-19, the infection caused by the newly identified coronavirus, is a currently a disease with no pharmaceutical weapons against it. There’s no vaccine to prevent it, and no drugs can treat it.
But researchers are racing to change that. A vaccine could be ready to test as soon as April. More than two dozen studies have already been registered on ClinicalTrials.gov, a website that tracks research. These studies aim to test everything from traditional Chinese medicine to vitamin C, stem cells, steroids, and medications that fight other viruses, like the flu and HIV. The hope is that something about how these repurposed remedies work will help patients who are desperately ill with no other prospects.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, says this is all part of the playbook for brand-new diseases. “There’s a lot of empiric guessing,” he says. “They’re going to propose a whole lot of drugs that already exist. They’re going to say, here’s the data that shows it blocks the virus” in a test tube. But test tubes aren’t people, and many drugs that seem to work in a lab won’t end up helping patients.
Coronaviruses are especially hard to stop once they invade the body. Unlike many other kinds of viruses, they have a fail-safe against tampering – a “proofreader” that constantly inspects their code, looking for errors, including the potentially life-saving errors that drugs could introduce.
Dr. Fauci said that researchers will be able to make better guesses about how to help people when they can try drugs in animals. “We don’t have an animal model yet of the new coronavirus. When we do get an animal model, that will be a big boon to drugs because then, you can clearly test them in a physiological way, whether they work,” he says.
Looking to drugs for HIV and flu
One of the drugs already under study is the combination of two HIV medications: lopinavir and ritonavir (Kaletra). Kaletra stops viruses by interfering with the enzymes they need to infect cells, called proteases.
One study being done at the Guangzhou Eighth People’s Hospital in China is testing Kaletra against Arbidol, an antiviral drug approved in China and Russia to treat the flu. Two groups of patients will take the medications along with standard care. A third group in the study will receive only standard care, typically supportive therapy with oxygen and IV fluids that are meant to support the body so the immune system can fight off a virus on its own.
An Ebola drug gets a second look
One repurposed drug generating a lot of buzz is an experimental infusion called remdesivir (Xembify). It was originally tested against the Ebola virus. While it didn’t work for that infection, it has been shown to shut down the new coronavirus, at least in test tubes. It’s been given to a small number of COVID-19 patients already, including one in Washington state.
In order to have better evidence of how well it may work in people, two studies in Beijing are comparing remdesivir to a dummy pill to see if the drug can help patients with both mild and severe symptoms recover from their illnesses. Viruses work by infecting cells, taking over their machinery, and getting them to crank out more copies of the virus, which then goes on to infect more cells. Remdesivir is a mimic that fools a virus into replacing one of its four building blocks with a chemical fake. Once in the virus’s blueprints, the imposter acts like a stop sign that keeps the virus from copying itself.
Other kinds of drugs in the same class – called nucleotide analogs – are used to attack cancer and other infectious viruses like hepatitis.
Last week, Chinese scientists published study showing remdesivir was effective against the new coronavirus, 2019-nCoV. Out of seven drugs tested, only remdesivir and an older drug called chloroquine (Aralen), which is used to treat malaria, worked, at least in test tubes. “It functions like a knife that just cuts off the RNA strand,” says Mark Denison, MD, a pediatric infectious disease specialist at Vanderbilt University in Nashville. “They can’t replicate any more. It stops them from doing that.” Dr. Denison is part of a team of researchers in Tennessee and North Carolina that discovered remdesivir could stop coronaviruses, like severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS), in test tubes and animals. He has studied coronaviruses in his lab for 30 years. He knew they would pose a threat again. “We’re shocked, but not surprised, that this has happened again,” he says of the China-based outbreak of 2019-nCoV.
After the SARS outbreak, which infected more than 8,000 people in 26 countries during 2002-2003, and MERS, which has infected nearly 2,500 people in 27 countries since 2012, researchers knew they had to start looking for treatments that would work against coronaviruses. Dr. Denison reached out to Gilead Sciences, a company best known for its antiviral medications that treat HIV and hepatitis C, and asked it to send drug candidates for him to test on coronaviruses. “The idea was that we didn’t want a drug that would just work against SARS or MERS,” he says. “We wanted drugs that worked against every coronavirus.”
Many of the agents he tried didn’t work until Dr. Denison and his team knocked out the virus’s pesky proofreader. Remdesivir seems to be able to defeat the proofreader, though Dr. Denison admits that he does not know how the drug gets around a virus’s defenses. He has a grant from the National Institutes of Health to study that. Gilead has been giving remdesivir to “a small number” of coronavirus patients in the United States and Europe on a compassionate basis.
One of those patients was a 35-year-old man in Everett, Wash., who had gotten pneumonia after being infected with the new coronavirus during a trip to see family in Wuhan, China, the epicenter of the outbreak. His doctors started IV remdesivir on the evening of his 7th day in the hospital. On the 8th day, he improved. He was well enough to stop using oxygen. Signs of pneumonia were gone. He got his appetite back. His case was recently published in the New England Journal of Medicine, igniting a firestorm of interest in the therapy.
Unfortunately, though, even Dr. Denison says a single person’s case isn’t enough proof that the medication can treat the new coronavirus. The patient, who has not been identified, was getting expert care. He may have improved on his own, despite getting the drug. He said the challenge in people will be to find out two things: whether the medication can block the spread of virus in the body and whether it can reverse the disease. “You can remove the source of injury, but you still have the injury,” he said. Other important questions include how soon the drug may need to be given after infection for it work and whether it may cause significant side effects.
A promising pill
Another drug, a nucleoside analog, that appears to be able to defeat the coronavirus proofreader, EIDD-2801, was developed by Emory University in Atlanta. It was originally intended to treat the flu but has shown some effectiveness against coronaviruses like SARS and MERS.
The FDA recently reached out to Emory asking if it had any drug candidates that might work against the new coronavirus. “It’s a good shot on goal here,” says George Painter, PhD, CEO of Drug Innovation Ventures at Emory. EIDD-2801 can be taken as a pill, which makes it easier to use outside of a hospital setting.
“The capsules for the trial are being made at the end of this month. So we’re close,” Painter says. “We’re right on the edge.”
While these early tests are just getting started, and it will be months until researchers have results, the World Health Organization has sounded a note of caution.
In new guidelines for the clinical management of COVID-19, the WHO reminded doctors and patients that there’s not enough evidence to recommend any specific treatment for infected patients.
Right now, the guidelines recommend that doctors offer supportive care to help the body fight off an infection on its own.
The organization says unlicensed treatments should be given only in the context of clinical trials that have been ethically reviewed or with strict clinical monitoring in emergencies.
This article first appeared on WebMD.com.
COVID-19, the infection caused by the newly identified coronavirus, is a currently a disease with no pharmaceutical weapons against it. There’s no vaccine to prevent it, and no drugs can treat it.
But researchers are racing to change that. A vaccine could be ready to test as soon as April. More than two dozen studies have already been registered on ClinicalTrials.gov, a website that tracks research. These studies aim to test everything from traditional Chinese medicine to vitamin C, stem cells, steroids, and medications that fight other viruses, like the flu and HIV. The hope is that something about how these repurposed remedies work will help patients who are desperately ill with no other prospects.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, says this is all part of the playbook for brand-new diseases. “There’s a lot of empiric guessing,” he says. “They’re going to propose a whole lot of drugs that already exist. They’re going to say, here’s the data that shows it blocks the virus” in a test tube. But test tubes aren’t people, and many drugs that seem to work in a lab won’t end up helping patients.
Coronaviruses are especially hard to stop once they invade the body. Unlike many other kinds of viruses, they have a fail-safe against tampering – a “proofreader” that constantly inspects their code, looking for errors, including the potentially life-saving errors that drugs could introduce.
Dr. Fauci said that researchers will be able to make better guesses about how to help people when they can try drugs in animals. “We don’t have an animal model yet of the new coronavirus. When we do get an animal model, that will be a big boon to drugs because then, you can clearly test them in a physiological way, whether they work,” he says.
Looking to drugs for HIV and flu
One of the drugs already under study is the combination of two HIV medications: lopinavir and ritonavir (Kaletra). Kaletra stops viruses by interfering with the enzymes they need to infect cells, called proteases.
One study being done at the Guangzhou Eighth People’s Hospital in China is testing Kaletra against Arbidol, an antiviral drug approved in China and Russia to treat the flu. Two groups of patients will take the medications along with standard care. A third group in the study will receive only standard care, typically supportive therapy with oxygen and IV fluids that are meant to support the body so the immune system can fight off a virus on its own.
An Ebola drug gets a second look
One repurposed drug generating a lot of buzz is an experimental infusion called remdesivir (Xembify). It was originally tested against the Ebola virus. While it didn’t work for that infection, it has been shown to shut down the new coronavirus, at least in test tubes. It’s been given to a small number of COVID-19 patients already, including one in Washington state.
In order to have better evidence of how well it may work in people, two studies in Beijing are comparing remdesivir to a dummy pill to see if the drug can help patients with both mild and severe symptoms recover from their illnesses. Viruses work by infecting cells, taking over their machinery, and getting them to crank out more copies of the virus, which then goes on to infect more cells. Remdesivir is a mimic that fools a virus into replacing one of its four building blocks with a chemical fake. Once in the virus’s blueprints, the imposter acts like a stop sign that keeps the virus from copying itself.
Other kinds of drugs in the same class – called nucleotide analogs – are used to attack cancer and other infectious viruses like hepatitis.
Last week, Chinese scientists published study showing remdesivir was effective against the new coronavirus, 2019-nCoV. Out of seven drugs tested, only remdesivir and an older drug called chloroquine (Aralen), which is used to treat malaria, worked, at least in test tubes. “It functions like a knife that just cuts off the RNA strand,” says Mark Denison, MD, a pediatric infectious disease specialist at Vanderbilt University in Nashville. “They can’t replicate any more. It stops them from doing that.” Dr. Denison is part of a team of researchers in Tennessee and North Carolina that discovered remdesivir could stop coronaviruses, like severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS), in test tubes and animals. He has studied coronaviruses in his lab for 30 years. He knew they would pose a threat again. “We’re shocked, but not surprised, that this has happened again,” he says of the China-based outbreak of 2019-nCoV.
After the SARS outbreak, which infected more than 8,000 people in 26 countries during 2002-2003, and MERS, which has infected nearly 2,500 people in 27 countries since 2012, researchers knew they had to start looking for treatments that would work against coronaviruses. Dr. Denison reached out to Gilead Sciences, a company best known for its antiviral medications that treat HIV and hepatitis C, and asked it to send drug candidates for him to test on coronaviruses. “The idea was that we didn’t want a drug that would just work against SARS or MERS,” he says. “We wanted drugs that worked against every coronavirus.”
Many of the agents he tried didn’t work until Dr. Denison and his team knocked out the virus’s pesky proofreader. Remdesivir seems to be able to defeat the proofreader, though Dr. Denison admits that he does not know how the drug gets around a virus’s defenses. He has a grant from the National Institutes of Health to study that. Gilead has been giving remdesivir to “a small number” of coronavirus patients in the United States and Europe on a compassionate basis.
One of those patients was a 35-year-old man in Everett, Wash., who had gotten pneumonia after being infected with the new coronavirus during a trip to see family in Wuhan, China, the epicenter of the outbreak. His doctors started IV remdesivir on the evening of his 7th day in the hospital. On the 8th day, he improved. He was well enough to stop using oxygen. Signs of pneumonia were gone. He got his appetite back. His case was recently published in the New England Journal of Medicine, igniting a firestorm of interest in the therapy.
Unfortunately, though, even Dr. Denison says a single person’s case isn’t enough proof that the medication can treat the new coronavirus. The patient, who has not been identified, was getting expert care. He may have improved on his own, despite getting the drug. He said the challenge in people will be to find out two things: whether the medication can block the spread of virus in the body and whether it can reverse the disease. “You can remove the source of injury, but you still have the injury,” he said. Other important questions include how soon the drug may need to be given after infection for it work and whether it may cause significant side effects.
A promising pill
Another drug, a nucleoside analog, that appears to be able to defeat the coronavirus proofreader, EIDD-2801, was developed by Emory University in Atlanta. It was originally intended to treat the flu but has shown some effectiveness against coronaviruses like SARS and MERS.
The FDA recently reached out to Emory asking if it had any drug candidates that might work against the new coronavirus. “It’s a good shot on goal here,” says George Painter, PhD, CEO of Drug Innovation Ventures at Emory. EIDD-2801 can be taken as a pill, which makes it easier to use outside of a hospital setting.
“The capsules for the trial are being made at the end of this month. So we’re close,” Painter says. “We’re right on the edge.”
While these early tests are just getting started, and it will be months until researchers have results, the World Health Organization has sounded a note of caution.
In new guidelines for the clinical management of COVID-19, the WHO reminded doctors and patients that there’s not enough evidence to recommend any specific treatment for infected patients.
Right now, the guidelines recommend that doctors offer supportive care to help the body fight off an infection on its own.
The organization says unlicensed treatments should be given only in the context of clinical trials that have been ethically reviewed or with strict clinical monitoring in emergencies.
This article first appeared on WebMD.com.
Infection with 2019 novel coronavirus extends to infants
between Dec. 8, 2019, and Feb. 6, 2020, based on data from the Chinese central government and local health departments.
“As of February 6, 2020, China reported 31,211 confirmed cases of COVID-19 and 637 fatalities,” wrote Min Wei, MD, of Wuhan University, China, and colleagues. However, “few infections in children have been reported.”
In a research letter published in JAMA, the investigators reviewed data from nine infants aged 28 days to 1 year who were hospitalized with a diagnosis of COVID-19 between Dec. 8, 2019, and Feb. 6, 2020. The ages of the infants ranged from 1 month to 11 months, and seven were female. The patients included two children from Beijing, two from Hainan, and one each from the areas of Guangdong, Anhui, Shanghai, Zhejiang, and Guizhou.
All infected infants had at least one infected family member, and the infants’ infections occurred after the family members’ infections; seven infants lived in Wuhan or had family members who had visited Wuhan.
One of the infants had no symptoms but tested positive for the 2019 novel coronavirus, and two others had a diagnosis but missing information on any symptoms. Fever occurred in four patients, and mild upper respiratory tract symptoms occurred in two patients.
None of the infants died, and none reported severe complications or the need for intensive care or mechanical ventilation, the investigators said. The fact that most of the infants were female might suggest that they are more susceptible to the virus than males, although overall COVID-19 viral infections have been more common in adult men, especially those with chronic comorbidities, Dr. Wei and associates noted.
The study findings were limited by the small sample size and lack of symptom data for some patients, the researchers said. However, the results confirm that the COVID-19 virus is transmissible to infants younger than 1 year, and adult caregivers should exercise protective measures including wearing masks, washing hands before contact with infants, and routinely sterilizing toys and tableware, they emphasized.
The study was supported by the National Natural Science Foundation of China and the Fundamental Research Funds for the Central Universities. The researchers had no financial conflicts to disclose.
SOURCE: Wei M et al. JAMA. 2020 Feb 14. doi:10.1001/jama.2020.2131.
between Dec. 8, 2019, and Feb. 6, 2020, based on data from the Chinese central government and local health departments.
“As of February 6, 2020, China reported 31,211 confirmed cases of COVID-19 and 637 fatalities,” wrote Min Wei, MD, of Wuhan University, China, and colleagues. However, “few infections in children have been reported.”
In a research letter published in JAMA, the investigators reviewed data from nine infants aged 28 days to 1 year who were hospitalized with a diagnosis of COVID-19 between Dec. 8, 2019, and Feb. 6, 2020. The ages of the infants ranged from 1 month to 11 months, and seven were female. The patients included two children from Beijing, two from Hainan, and one each from the areas of Guangdong, Anhui, Shanghai, Zhejiang, and Guizhou.
All infected infants had at least one infected family member, and the infants’ infections occurred after the family members’ infections; seven infants lived in Wuhan or had family members who had visited Wuhan.
One of the infants had no symptoms but tested positive for the 2019 novel coronavirus, and two others had a diagnosis but missing information on any symptoms. Fever occurred in four patients, and mild upper respiratory tract symptoms occurred in two patients.
None of the infants died, and none reported severe complications or the need for intensive care or mechanical ventilation, the investigators said. The fact that most of the infants were female might suggest that they are more susceptible to the virus than males, although overall COVID-19 viral infections have been more common in adult men, especially those with chronic comorbidities, Dr. Wei and associates noted.
The study findings were limited by the small sample size and lack of symptom data for some patients, the researchers said. However, the results confirm that the COVID-19 virus is transmissible to infants younger than 1 year, and adult caregivers should exercise protective measures including wearing masks, washing hands before contact with infants, and routinely sterilizing toys and tableware, they emphasized.
The study was supported by the National Natural Science Foundation of China and the Fundamental Research Funds for the Central Universities. The researchers had no financial conflicts to disclose.
SOURCE: Wei M et al. JAMA. 2020 Feb 14. doi:10.1001/jama.2020.2131.
between Dec. 8, 2019, and Feb. 6, 2020, based on data from the Chinese central government and local health departments.
“As of February 6, 2020, China reported 31,211 confirmed cases of COVID-19 and 637 fatalities,” wrote Min Wei, MD, of Wuhan University, China, and colleagues. However, “few infections in children have been reported.”
In a research letter published in JAMA, the investigators reviewed data from nine infants aged 28 days to 1 year who were hospitalized with a diagnosis of COVID-19 between Dec. 8, 2019, and Feb. 6, 2020. The ages of the infants ranged from 1 month to 11 months, and seven were female. The patients included two children from Beijing, two from Hainan, and one each from the areas of Guangdong, Anhui, Shanghai, Zhejiang, and Guizhou.
All infected infants had at least one infected family member, and the infants’ infections occurred after the family members’ infections; seven infants lived in Wuhan or had family members who had visited Wuhan.
One of the infants had no symptoms but tested positive for the 2019 novel coronavirus, and two others had a diagnosis but missing information on any symptoms. Fever occurred in four patients, and mild upper respiratory tract symptoms occurred in two patients.
None of the infants died, and none reported severe complications or the need for intensive care or mechanical ventilation, the investigators said. The fact that most of the infants were female might suggest that they are more susceptible to the virus than males, although overall COVID-19 viral infections have been more common in adult men, especially those with chronic comorbidities, Dr. Wei and associates noted.
The study findings were limited by the small sample size and lack of symptom data for some patients, the researchers said. However, the results confirm that the COVID-19 virus is transmissible to infants younger than 1 year, and adult caregivers should exercise protective measures including wearing masks, washing hands before contact with infants, and routinely sterilizing toys and tableware, they emphasized.
The study was supported by the National Natural Science Foundation of China and the Fundamental Research Funds for the Central Universities. The researchers had no financial conflicts to disclose.
SOURCE: Wei M et al. JAMA. 2020 Feb 14. doi:10.1001/jama.2020.2131.
FROM JAMA
As novel coronavirus outbreak evolves, critical care providers need to be prepared
ORLANDO – While the impact of the 2019 novel coronavirus outbreak on hospitals outside of China remains to be determined, there are several practical points critical care professionals need to know to be prepared in the face of this dynamic and rapidly evolving outbreak, speakers said at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
“Priorities for us in our hospitals are early detection, infection prevention, staff safety, and obviously, taking care of sick people,” said Ryan C. Maves, MD, of the Naval Medical Center San Diego in a special session on the 2019 Novel Coronavirus outbreak.*
Approximately 72,000 cases of coronavirus disease 2019 (COVID-19) had been reported as of Feb. 17, 2020, the day of Dr. Maves’ talk, according to statistics from Johns Hopkins Center for Science and Engineering in Baltimore. A total of 1,775 deaths had been recorded, nearly all of which were in Hubei Province, the central point of the outbreak. In the United States, the number of cases stood at 15, with no deaths reported.
While the dynamics of the 2019 novel coronavirus are still being learned, the estimated range of spread for droplet transmission is 2 meters, according to Dr. Maves. The duration of environmental persistence is not yet known, but he said that other coronaviruses persist in low-humidity conditions for up to 4 days.
The number of secondary cases that arise from a primary infection, or R0, is estimated to be between 1.5 and 3, though it can change as exposure evolves; by comparison, the R0 for H1N1 influenza has been reported as 1.5, while measles is 12-18, indicating that it is “very contagious,” said Dr. Maves. Severe acute respiratory syndrome had an initial R0 of about 3.5, which he said declined rapidly to 0.7 as environmental and policy controls were put into place.
Critical care professionals need to know how to identify patients at risk of having COVID-19 and determine whether they need further work-up, according to Dr. Maves, who highlighted recent criteria released by the Centers for Disease Control and Prevention.
The highest-risk category, he said, are individuals exposed to a laboratory-confirmed coronavirus case, which along with fever or signs and symptoms of a lower respiratory illness would be sufficient to classify them as a “person of interest” requiring further evaluation for disease. A history of travel from Hubei Province plus fever and signs/symptoms of lower respiratory illness would also meet criteria for evaluation, according to the CDC, while travel to mainland China would also meet the threshold, if those symptoms required hospitalization.
The CDC also published a step-wise flowchart to evaluate patients who may have been exposed to the 2019 novel coronavirus. According to that flowchart, if an individual has traveled to China or had close contact with someone infected with the 2019 Novel Coronavirus within 14 days of symptoms, and that individual has fever or symptoms of lower respiratory illness such as cough or shortness of breath, then providers should isolate that individual and assess clinical status, in addition to contacting the local health department.
Laura E. Evans, MD, MS, FCCM, of New York University, said she might recommend providers “flip the script” on that CDC algorithm when it comes to identifying patients who may have been exposed.
“I think perhaps what we should be doing at sites of entry is not talking about travel as the first question, but rather fever or symptoms of lower respiratory illnesses as the first question, and use that as the opportunity to implement risk mitigation at that stage,” Dr. Evans said in a presentation on preparing for COVID-19.
Even with “substantial uncertainty” about the potential impact of the 2019 Novel Coronavirus, a significant influx of seriously ill patients would put strain the U.S. health care delivery system, she added.
“None of us have tons of extra capacity in our emergency departments, inpatient units, or ICUs, and I think we need to be prepared for that,” she added. “We need to know what our process is for ‘identify, isolate, and inform,’ and we need to be testing that now.”
Dr. Maves and Dr. Evans both reported that they had no financial conflicts of interest to report. Dr. Maves indicated that the views expressed in his presentation did not necessarily reflect the official policy or position of the Department of the Navy, Department of Defense, or the United States government.
*Correction, 2/19/20: An earlier version of this article misstated the location of the naval center.
ORLANDO – While the impact of the 2019 novel coronavirus outbreak on hospitals outside of China remains to be determined, there are several practical points critical care professionals need to know to be prepared in the face of this dynamic and rapidly evolving outbreak, speakers said at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
“Priorities for us in our hospitals are early detection, infection prevention, staff safety, and obviously, taking care of sick people,” said Ryan C. Maves, MD, of the Naval Medical Center San Diego in a special session on the 2019 Novel Coronavirus outbreak.*
Approximately 72,000 cases of coronavirus disease 2019 (COVID-19) had been reported as of Feb. 17, 2020, the day of Dr. Maves’ talk, according to statistics from Johns Hopkins Center for Science and Engineering in Baltimore. A total of 1,775 deaths had been recorded, nearly all of which were in Hubei Province, the central point of the outbreak. In the United States, the number of cases stood at 15, with no deaths reported.
While the dynamics of the 2019 novel coronavirus are still being learned, the estimated range of spread for droplet transmission is 2 meters, according to Dr. Maves. The duration of environmental persistence is not yet known, but he said that other coronaviruses persist in low-humidity conditions for up to 4 days.
The number of secondary cases that arise from a primary infection, or R0, is estimated to be between 1.5 and 3, though it can change as exposure evolves; by comparison, the R0 for H1N1 influenza has been reported as 1.5, while measles is 12-18, indicating that it is “very contagious,” said Dr. Maves. Severe acute respiratory syndrome had an initial R0 of about 3.5, which he said declined rapidly to 0.7 as environmental and policy controls were put into place.
Critical care professionals need to know how to identify patients at risk of having COVID-19 and determine whether they need further work-up, according to Dr. Maves, who highlighted recent criteria released by the Centers for Disease Control and Prevention.
The highest-risk category, he said, are individuals exposed to a laboratory-confirmed coronavirus case, which along with fever or signs and symptoms of a lower respiratory illness would be sufficient to classify them as a “person of interest” requiring further evaluation for disease. A history of travel from Hubei Province plus fever and signs/symptoms of lower respiratory illness would also meet criteria for evaluation, according to the CDC, while travel to mainland China would also meet the threshold, if those symptoms required hospitalization.
The CDC also published a step-wise flowchart to evaluate patients who may have been exposed to the 2019 novel coronavirus. According to that flowchart, if an individual has traveled to China or had close contact with someone infected with the 2019 Novel Coronavirus within 14 days of symptoms, and that individual has fever or symptoms of lower respiratory illness such as cough or shortness of breath, then providers should isolate that individual and assess clinical status, in addition to contacting the local health department.
Laura E. Evans, MD, MS, FCCM, of New York University, said she might recommend providers “flip the script” on that CDC algorithm when it comes to identifying patients who may have been exposed.
“I think perhaps what we should be doing at sites of entry is not talking about travel as the first question, but rather fever or symptoms of lower respiratory illnesses as the first question, and use that as the opportunity to implement risk mitigation at that stage,” Dr. Evans said in a presentation on preparing for COVID-19.
Even with “substantial uncertainty” about the potential impact of the 2019 Novel Coronavirus, a significant influx of seriously ill patients would put strain the U.S. health care delivery system, she added.
“None of us have tons of extra capacity in our emergency departments, inpatient units, or ICUs, and I think we need to be prepared for that,” she added. “We need to know what our process is for ‘identify, isolate, and inform,’ and we need to be testing that now.”
Dr. Maves and Dr. Evans both reported that they had no financial conflicts of interest to report. Dr. Maves indicated that the views expressed in his presentation did not necessarily reflect the official policy or position of the Department of the Navy, Department of Defense, or the United States government.
*Correction, 2/19/20: An earlier version of this article misstated the location of the naval center.
ORLANDO – While the impact of the 2019 novel coronavirus outbreak on hospitals outside of China remains to be determined, there are several practical points critical care professionals need to know to be prepared in the face of this dynamic and rapidly evolving outbreak, speakers said at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
“Priorities for us in our hospitals are early detection, infection prevention, staff safety, and obviously, taking care of sick people,” said Ryan C. Maves, MD, of the Naval Medical Center San Diego in a special session on the 2019 Novel Coronavirus outbreak.*
Approximately 72,000 cases of coronavirus disease 2019 (COVID-19) had been reported as of Feb. 17, 2020, the day of Dr. Maves’ talk, according to statistics from Johns Hopkins Center for Science and Engineering in Baltimore. A total of 1,775 deaths had been recorded, nearly all of which were in Hubei Province, the central point of the outbreak. In the United States, the number of cases stood at 15, with no deaths reported.
While the dynamics of the 2019 novel coronavirus are still being learned, the estimated range of spread for droplet transmission is 2 meters, according to Dr. Maves. The duration of environmental persistence is not yet known, but he said that other coronaviruses persist in low-humidity conditions for up to 4 days.
The number of secondary cases that arise from a primary infection, or R0, is estimated to be between 1.5 and 3, though it can change as exposure evolves; by comparison, the R0 for H1N1 influenza has been reported as 1.5, while measles is 12-18, indicating that it is “very contagious,” said Dr. Maves. Severe acute respiratory syndrome had an initial R0 of about 3.5, which he said declined rapidly to 0.7 as environmental and policy controls were put into place.
Critical care professionals need to know how to identify patients at risk of having COVID-19 and determine whether they need further work-up, according to Dr. Maves, who highlighted recent criteria released by the Centers for Disease Control and Prevention.
The highest-risk category, he said, are individuals exposed to a laboratory-confirmed coronavirus case, which along with fever or signs and symptoms of a lower respiratory illness would be sufficient to classify them as a “person of interest” requiring further evaluation for disease. A history of travel from Hubei Province plus fever and signs/symptoms of lower respiratory illness would also meet criteria for evaluation, according to the CDC, while travel to mainland China would also meet the threshold, if those symptoms required hospitalization.
The CDC also published a step-wise flowchart to evaluate patients who may have been exposed to the 2019 novel coronavirus. According to that flowchart, if an individual has traveled to China or had close contact with someone infected with the 2019 Novel Coronavirus within 14 days of symptoms, and that individual has fever or symptoms of lower respiratory illness such as cough or shortness of breath, then providers should isolate that individual and assess clinical status, in addition to contacting the local health department.
Laura E. Evans, MD, MS, FCCM, of New York University, said she might recommend providers “flip the script” on that CDC algorithm when it comes to identifying patients who may have been exposed.
“I think perhaps what we should be doing at sites of entry is not talking about travel as the first question, but rather fever or symptoms of lower respiratory illnesses as the first question, and use that as the opportunity to implement risk mitigation at that stage,” Dr. Evans said in a presentation on preparing for COVID-19.
Even with “substantial uncertainty” about the potential impact of the 2019 Novel Coronavirus, a significant influx of seriously ill patients would put strain the U.S. health care delivery system, she added.
“None of us have tons of extra capacity in our emergency departments, inpatient units, or ICUs, and I think we need to be prepared for that,” she added. “We need to know what our process is for ‘identify, isolate, and inform,’ and we need to be testing that now.”
Dr. Maves and Dr. Evans both reported that they had no financial conflicts of interest to report. Dr. Maves indicated that the views expressed in his presentation did not necessarily reflect the official policy or position of the Department of the Navy, Department of Defense, or the United States government.
*Correction, 2/19/20: An earlier version of this article misstated the location of the naval center.
EXPERT ANALYSIS FROM CCC49