User login
Inhibitor exhibits activity against resistant AML

Preclinical research indicates that a novel inhibitor can overcome resistance-conferring FLT3 mutations in acute myeloid leukemia (AML).
The MERTK/FLT3 inhibitor MRX-2843 induced apoptosis and inhibited colony formation in AML cell lines and primary patient samples expressing MERTK and/or FLT3-ITD.
MRX-2843 also improved survival in mouse models of AML, including cases where tumors were resistant to the FLT3 inhibitor quizartinib.
Douglas Graham, MD, PhD, of the University of Colorado in Aurora, and his colleagues conducted this research and reported the results in JCI Insight.
The researchers previously showed that the receptor tyrosine kinase MERTK is overexpressed in 80% to 90% of AMLs and contributes to leukemogenesis.
With the current study, they showed that MRX-2843 abrogates activation of MERTK, FLT3, and their downstream effectors. And this translates to antileukemic activity in vitro and in vivo.
MRX-2843 inhibited MERTK signaling, induced cell death, and abolished oncogenic phenotypes in AML cells. The drug also exhibited therapeutic activity in a MERTK-dependent xenograft model.
MRX-2843 was able to stop the activation of FLT3 and its signaling pathways almost completely. The researchers said this suggests the drug has somewhat higher cellular potency against FLT3 relative to MERTK.
In mouse models of FLT3-ITD AML, MRX-2843 significantly prolonged survival when compared to vehicle control.
The researchers also said MRX-2843 selectively inhibited colony formation in primary AML patient samples. Primary human MERTK-expressing leukemic blasts, with or without FLT3-ITD mutations, proved sensitive to treatment with MRX-2843.
In addition, MRX-2843 increased survival and decreased peripheral disease burden in patient-derived xenograft models of AML—both MERTK+FLT3-WT and MERTK+FLT3-ITD models.
Finally, the researchers found that MRX-2843 was active against quizartinib-resistant FLT3-mutant proteins, induced apoptosis and inhibited colony formation in quizartinib-resistant FLT3-ITD cell lines, and prolonged survival in quizartinib-resistant FLT3-ITD xenograft models. ![]()

Preclinical research indicates that a novel inhibitor can overcome resistance-conferring FLT3 mutations in acute myeloid leukemia (AML).
The MERTK/FLT3 inhibitor MRX-2843 induced apoptosis and inhibited colony formation in AML cell lines and primary patient samples expressing MERTK and/or FLT3-ITD.
MRX-2843 also improved survival in mouse models of AML, including cases where tumors were resistant to the FLT3 inhibitor quizartinib.
Douglas Graham, MD, PhD, of the University of Colorado in Aurora, and his colleagues conducted this research and reported the results in JCI Insight.
The researchers previously showed that the receptor tyrosine kinase MERTK is overexpressed in 80% to 90% of AMLs and contributes to leukemogenesis.
With the current study, they showed that MRX-2843 abrogates activation of MERTK, FLT3, and their downstream effectors. And this translates to antileukemic activity in vitro and in vivo.
MRX-2843 inhibited MERTK signaling, induced cell death, and abolished oncogenic phenotypes in AML cells. The drug also exhibited therapeutic activity in a MERTK-dependent xenograft model.
MRX-2843 was able to stop the activation of FLT3 and its signaling pathways almost completely. The researchers said this suggests the drug has somewhat higher cellular potency against FLT3 relative to MERTK.
In mouse models of FLT3-ITD AML, MRX-2843 significantly prolonged survival when compared to vehicle control.
The researchers also said MRX-2843 selectively inhibited colony formation in primary AML patient samples. Primary human MERTK-expressing leukemic blasts, with or without FLT3-ITD mutations, proved sensitive to treatment with MRX-2843.
In addition, MRX-2843 increased survival and decreased peripheral disease burden in patient-derived xenograft models of AML—both MERTK+FLT3-WT and MERTK+FLT3-ITD models.
Finally, the researchers found that MRX-2843 was active against quizartinib-resistant FLT3-mutant proteins, induced apoptosis and inhibited colony formation in quizartinib-resistant FLT3-ITD cell lines, and prolonged survival in quizartinib-resistant FLT3-ITD xenograft models. ![]()

Preclinical research indicates that a novel inhibitor can overcome resistance-conferring FLT3 mutations in acute myeloid leukemia (AML).
The MERTK/FLT3 inhibitor MRX-2843 induced apoptosis and inhibited colony formation in AML cell lines and primary patient samples expressing MERTK and/or FLT3-ITD.
MRX-2843 also improved survival in mouse models of AML, including cases where tumors were resistant to the FLT3 inhibitor quizartinib.
Douglas Graham, MD, PhD, of the University of Colorado in Aurora, and his colleagues conducted this research and reported the results in JCI Insight.
The researchers previously showed that the receptor tyrosine kinase MERTK is overexpressed in 80% to 90% of AMLs and contributes to leukemogenesis.
With the current study, they showed that MRX-2843 abrogates activation of MERTK, FLT3, and their downstream effectors. And this translates to antileukemic activity in vitro and in vivo.
MRX-2843 inhibited MERTK signaling, induced cell death, and abolished oncogenic phenotypes in AML cells. The drug also exhibited therapeutic activity in a MERTK-dependent xenograft model.
MRX-2843 was able to stop the activation of FLT3 and its signaling pathways almost completely. The researchers said this suggests the drug has somewhat higher cellular potency against FLT3 relative to MERTK.
In mouse models of FLT3-ITD AML, MRX-2843 significantly prolonged survival when compared to vehicle control.
The researchers also said MRX-2843 selectively inhibited colony formation in primary AML patient samples. Primary human MERTK-expressing leukemic blasts, with or without FLT3-ITD mutations, proved sensitive to treatment with MRX-2843.
In addition, MRX-2843 increased survival and decreased peripheral disease burden in patient-derived xenograft models of AML—both MERTK+FLT3-WT and MERTK+FLT3-ITD models.
Finally, the researchers found that MRX-2843 was active against quizartinib-resistant FLT3-mutant proteins, induced apoptosis and inhibited colony formation in quizartinib-resistant FLT3-ITD cell lines, and prolonged survival in quizartinib-resistant FLT3-ITD xenograft models. ![]()
Germline mutations linked to hematologic malignancies

A new study suggests mutations in the gene DDX41 occur in families where hematologic malignancies are common.
Previous research showed that both germline and acquired DDX41 mutations occur in families with multiple cases of late-onset myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML).
The new study, published in Blood, has linked germline mutations in DDX41 to chronic myeloid leukemia and lymphomas as well.
“This is the first gene identified in families with lymphoma and represents a major breakthrough for the field,” said study author Hamish Scott, PhD, of the University of Adelaide in South Australia.
“Researchers are recognizing now that genetic predisposition to blood cancer is more common than previously thought, and our study shows the importance of taking a thorough family history at diagnosis.”
To conduct this study, Dr Scott and his colleagues screened 2 cohorts of families with a range of hematologic disorders (malignant and non-malignant). One cohort included 240 individuals from 93 families in Australia. The other included 246 individuals from 198 families in the US.
In all, 9 of the families (3%) had germline DDX41 mutations.
Three families carried the recurrent p.D140Gfs*2 mutation, which was linked to AML.
One family carried a germline mutation—p.R525H, c.1574G.A—that was previously described only as a somatic mutation at the time of progression to MDS or AML. In the current study, the mutation was again linked to MDS and AML.
Five families carried novel DDX41 mutations.
One of these mutations was a germline substitution—c.435-2_435-1delAGinsCA—that was linked to MDS in 1 family.
Two families had a missense start-loss substitution—c.3G.A, p.M1I—that was linked to MDS, AML, chronic myeloid leukemia, and non-Hodgkin lymphoma.
One family had a DDX41 missense variant—c.490C.T, p.R164W. This was linked to Hodgkin and non-Hodgkin lymphoma (including 3 cases of follicular lymphoma). There was a possible link to multiple myeloma as well, but the diagnosis could not be confirmed.
And 1 family had a missense mutation in the helicase domain—p.G530D—that was linked to AML.
“DDX41 is a new type of cancer predisposition gene, and we are still investigating its function,” Dr Scott noted.
“But it appears to have dual roles in regulating the correct expression of genes in the cell and also enabling the immune system to respond to threats such as bacteria and viruses, as well as the development of cancer cells. Immunotherapy is a promising approach for cancer treatment, and our research to understand the function of DDX41 will help design better therapies.” ![]()

A new study suggests mutations in the gene DDX41 occur in families where hematologic malignancies are common.
Previous research showed that both germline and acquired DDX41 mutations occur in families with multiple cases of late-onset myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML).
The new study, published in Blood, has linked germline mutations in DDX41 to chronic myeloid leukemia and lymphomas as well.
“This is the first gene identified in families with lymphoma and represents a major breakthrough for the field,” said study author Hamish Scott, PhD, of the University of Adelaide in South Australia.
“Researchers are recognizing now that genetic predisposition to blood cancer is more common than previously thought, and our study shows the importance of taking a thorough family history at diagnosis.”
To conduct this study, Dr Scott and his colleagues screened 2 cohorts of families with a range of hematologic disorders (malignant and non-malignant). One cohort included 240 individuals from 93 families in Australia. The other included 246 individuals from 198 families in the US.
In all, 9 of the families (3%) had germline DDX41 mutations.
Three families carried the recurrent p.D140Gfs*2 mutation, which was linked to AML.
One family carried a germline mutation—p.R525H, c.1574G.A—that was previously described only as a somatic mutation at the time of progression to MDS or AML. In the current study, the mutation was again linked to MDS and AML.
Five families carried novel DDX41 mutations.
One of these mutations was a germline substitution—c.435-2_435-1delAGinsCA—that was linked to MDS in 1 family.
Two families had a missense start-loss substitution—c.3G.A, p.M1I—that was linked to MDS, AML, chronic myeloid leukemia, and non-Hodgkin lymphoma.
One family had a DDX41 missense variant—c.490C.T, p.R164W. This was linked to Hodgkin and non-Hodgkin lymphoma (including 3 cases of follicular lymphoma). There was a possible link to multiple myeloma as well, but the diagnosis could not be confirmed.
And 1 family had a missense mutation in the helicase domain—p.G530D—that was linked to AML.
“DDX41 is a new type of cancer predisposition gene, and we are still investigating its function,” Dr Scott noted.
“But it appears to have dual roles in regulating the correct expression of genes in the cell and also enabling the immune system to respond to threats such as bacteria and viruses, as well as the development of cancer cells. Immunotherapy is a promising approach for cancer treatment, and our research to understand the function of DDX41 will help design better therapies.” ![]()

A new study suggests mutations in the gene DDX41 occur in families where hematologic malignancies are common.
Previous research showed that both germline and acquired DDX41 mutations occur in families with multiple cases of late-onset myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML).
The new study, published in Blood, has linked germline mutations in DDX41 to chronic myeloid leukemia and lymphomas as well.
“This is the first gene identified in families with lymphoma and represents a major breakthrough for the field,” said study author Hamish Scott, PhD, of the University of Adelaide in South Australia.
“Researchers are recognizing now that genetic predisposition to blood cancer is more common than previously thought, and our study shows the importance of taking a thorough family history at diagnosis.”
To conduct this study, Dr Scott and his colleagues screened 2 cohorts of families with a range of hematologic disorders (malignant and non-malignant). One cohort included 240 individuals from 93 families in Australia. The other included 246 individuals from 198 families in the US.
In all, 9 of the families (3%) had germline DDX41 mutations.
Three families carried the recurrent p.D140Gfs*2 mutation, which was linked to AML.
One family carried a germline mutation—p.R525H, c.1574G.A—that was previously described only as a somatic mutation at the time of progression to MDS or AML. In the current study, the mutation was again linked to MDS and AML.
Five families carried novel DDX41 mutations.
One of these mutations was a germline substitution—c.435-2_435-1delAGinsCA—that was linked to MDS in 1 family.
Two families had a missense start-loss substitution—c.3G.A, p.M1I—that was linked to MDS, AML, chronic myeloid leukemia, and non-Hodgkin lymphoma.
One family had a DDX41 missense variant—c.490C.T, p.R164W. This was linked to Hodgkin and non-Hodgkin lymphoma (including 3 cases of follicular lymphoma). There was a possible link to multiple myeloma as well, but the diagnosis could not be confirmed.
And 1 family had a missense mutation in the helicase domain—p.G530D—that was linked to AML.
“DDX41 is a new type of cancer predisposition gene, and we are still investigating its function,” Dr Scott noted.
“But it appears to have dual roles in regulating the correct expression of genes in the cell and also enabling the immune system to respond to threats such as bacteria and viruses, as well as the development of cancer cells. Immunotherapy is a promising approach for cancer treatment, and our research to understand the function of DDX41 will help design better therapies.” ![]()
Team identifies potential target for aggressive AML

Photo by Rhoda Baer
Research published in The Journal of Clinical Investigation has revealed a potential therapeutic target for an aggressive form of acute myeloid leukemia (AML).
Investigators studied AML characterized by overexpression of the gene meningioma-1 (MN1), which is not a druggable target.
The team found that MN1 overexpression induces aggressive AML that is dependent on a gene expression program controlled by 2 histone methyltransferases.
And 1 of these histone methyltransferases can be targeted by drugs currently in clinical development.
To make these discoveries, the investigators forced expression of MN1 in mice, which induced AML, and looked for changes in other genes. They found that MN1 overexpression prompted the activation of genes already linked to AML development—HoxA9 and Meis1.
HoxA9 and Meis1 are key targets of the histone methyltransferases Mll1 and Dot1l. It turned out that Mll1 and Dotl1 are essential for creating the environment MN1 needs to cause AML.
“In mice, we put MN1 in first, leading to AML,” explained study author Kathrin Bernt, MD, of the University of Colorado Anschutz Medical Campus.
“Then, we knocked out these chromatin-regulating molecules, Mll1 or Dot1l. When we did that, the leukemia collapsed.”
The investigators also studied samples from AML patients and found that samples with overexpression of MN1 and HOXA9 were sensitive to the DOT1L inhibitor EPZ004777. The drug induced dose-dependent decreases in cell growth and the fraction of cycling cells, as well as an increase in apoptosis.
Anticancer agents targeting DOT1L are already in clinical trials. One such inhibitor, EPZ-5676, is being tested in a phase 1 trial of pediatric patients with aggressive leukemias (NCT02141828).
“The existing trial targets patients with rearrangements in the gene MLL1,” Dr Bernt noted. “Our study shows another subset of patients that may benefit from this or other therapies aimed at DOT1L inhibition—namely, patients with MN1 overexpression.”
Dr Bernt added, however, that the investigators must still determine the cutoff of MN1 overexpression at which AML is susceptible to DOT1L inhibition.
“Overexpression exists along a spectrum,” she explained. “At what degree of MN1 overexpression does it become clinically significant?” ![]()

Photo by Rhoda Baer
Research published in The Journal of Clinical Investigation has revealed a potential therapeutic target for an aggressive form of acute myeloid leukemia (AML).
Investigators studied AML characterized by overexpression of the gene meningioma-1 (MN1), which is not a druggable target.
The team found that MN1 overexpression induces aggressive AML that is dependent on a gene expression program controlled by 2 histone methyltransferases.
And 1 of these histone methyltransferases can be targeted by drugs currently in clinical development.
To make these discoveries, the investigators forced expression of MN1 in mice, which induced AML, and looked for changes in other genes. They found that MN1 overexpression prompted the activation of genes already linked to AML development—HoxA9 and Meis1.
HoxA9 and Meis1 are key targets of the histone methyltransferases Mll1 and Dot1l. It turned out that Mll1 and Dotl1 are essential for creating the environment MN1 needs to cause AML.
“In mice, we put MN1 in first, leading to AML,” explained study author Kathrin Bernt, MD, of the University of Colorado Anschutz Medical Campus.
“Then, we knocked out these chromatin-regulating molecules, Mll1 or Dot1l. When we did that, the leukemia collapsed.”
The investigators also studied samples from AML patients and found that samples with overexpression of MN1 and HOXA9 were sensitive to the DOT1L inhibitor EPZ004777. The drug induced dose-dependent decreases in cell growth and the fraction of cycling cells, as well as an increase in apoptosis.
Anticancer agents targeting DOT1L are already in clinical trials. One such inhibitor, EPZ-5676, is being tested in a phase 1 trial of pediatric patients with aggressive leukemias (NCT02141828).
“The existing trial targets patients with rearrangements in the gene MLL1,” Dr Bernt noted. “Our study shows another subset of patients that may benefit from this or other therapies aimed at DOT1L inhibition—namely, patients with MN1 overexpression.”
Dr Bernt added, however, that the investigators must still determine the cutoff of MN1 overexpression at which AML is susceptible to DOT1L inhibition.
“Overexpression exists along a spectrum,” she explained. “At what degree of MN1 overexpression does it become clinically significant?” ![]()

Photo by Rhoda Baer
Research published in The Journal of Clinical Investigation has revealed a potential therapeutic target for an aggressive form of acute myeloid leukemia (AML).
Investigators studied AML characterized by overexpression of the gene meningioma-1 (MN1), which is not a druggable target.
The team found that MN1 overexpression induces aggressive AML that is dependent on a gene expression program controlled by 2 histone methyltransferases.
And 1 of these histone methyltransferases can be targeted by drugs currently in clinical development.
To make these discoveries, the investigators forced expression of MN1 in mice, which induced AML, and looked for changes in other genes. They found that MN1 overexpression prompted the activation of genes already linked to AML development—HoxA9 and Meis1.
HoxA9 and Meis1 are key targets of the histone methyltransferases Mll1 and Dot1l. It turned out that Mll1 and Dotl1 are essential for creating the environment MN1 needs to cause AML.
“In mice, we put MN1 in first, leading to AML,” explained study author Kathrin Bernt, MD, of the University of Colorado Anschutz Medical Campus.
“Then, we knocked out these chromatin-regulating molecules, Mll1 or Dot1l. When we did that, the leukemia collapsed.”
The investigators also studied samples from AML patients and found that samples with overexpression of MN1 and HOXA9 were sensitive to the DOT1L inhibitor EPZ004777. The drug induced dose-dependent decreases in cell growth and the fraction of cycling cells, as well as an increase in apoptosis.
Anticancer agents targeting DOT1L are already in clinical trials. One such inhibitor, EPZ-5676, is being tested in a phase 1 trial of pediatric patients with aggressive leukemias (NCT02141828).
“The existing trial targets patients with rearrangements in the gene MLL1,” Dr Bernt noted. “Our study shows another subset of patients that may benefit from this or other therapies aimed at DOT1L inhibition—namely, patients with MN1 overexpression.”
Dr Bernt added, however, that the investigators must still determine the cutoff of MN1 overexpression at which AML is susceptible to DOT1L inhibition.
“Overexpression exists along a spectrum,” she explained. “At what degree of MN1 overexpression does it become clinically significant?” ![]()
Children’s cancer survival steadily increasing
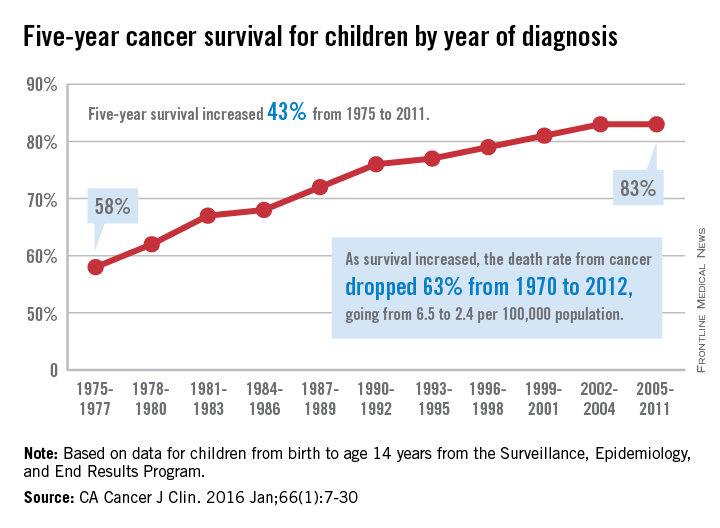
The 5-year cancer survival rate for children younger than 15 years old is up by 43% since 1975, according to investigators from the American Cancer Society.
The 5-year survival rate for all cancers showed a statistically significant rise from 58% in 1975 to 83% in 2011, said Rebecca L. Siegel and her associates at the ACS (CA Cancer J Clin. 2016 Jan;66[1]:7-30).
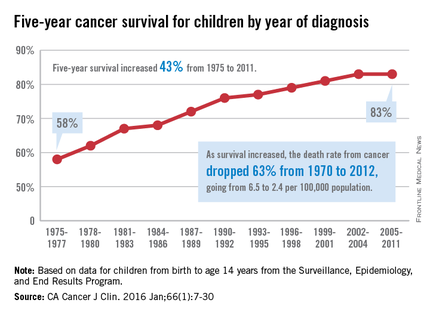
“The substantial progress for all of the major childhood cancers reflects both improvements in treatment and high levels of participation in clinical trials,” they wrote.
Survival for cancers of the brain and nervous system – now the leading cause of cancer death for those younger than 20 years old – increased from 57% in 1975 to 74% in 2011. The next-most-common cause of cancer death in children and adolescents is leukemia, and 5-year survival for acute myeloid leukemia went from 19% in 1975 to 67% in 2011, while 5-year survival for acute lymphocytic leukemia rose from 57% to 91% over that time period, the investigators reported.
The authors reported no conflicts of interest.
The 5-year cancer survival rate for children younger than 15 years old is up by 43% since 1975, according to investigators from the American Cancer Society.
The 5-year survival rate for all cancers showed a statistically significant rise from 58% in 1975 to 83% in 2011, said Rebecca L. Siegel and her associates at the ACS (CA Cancer J Clin. 2016 Jan;66[1]:7-30).

“The substantial progress for all of the major childhood cancers reflects both improvements in treatment and high levels of participation in clinical trials,” they wrote.
Survival for cancers of the brain and nervous system – now the leading cause of cancer death for those younger than 20 years old – increased from 57% in 1975 to 74% in 2011. The next-most-common cause of cancer death in children and adolescents is leukemia, and 5-year survival for acute myeloid leukemia went from 19% in 1975 to 67% in 2011, while 5-year survival for acute lymphocytic leukemia rose from 57% to 91% over that time period, the investigators reported.
The authors reported no conflicts of interest.
The 5-year cancer survival rate for children younger than 15 years old is up by 43% since 1975, according to investigators from the American Cancer Society.
The 5-year survival rate for all cancers showed a statistically significant rise from 58% in 1975 to 83% in 2011, said Rebecca L. Siegel and her associates at the ACS (CA Cancer J Clin. 2016 Jan;66[1]:7-30).

“The substantial progress for all of the major childhood cancers reflects both improvements in treatment and high levels of participation in clinical trials,” they wrote.
Survival for cancers of the brain and nervous system – now the leading cause of cancer death for those younger than 20 years old – increased from 57% in 1975 to 74% in 2011. The next-most-common cause of cancer death in children and adolescents is leukemia, and 5-year survival for acute myeloid leukemia went from 19% in 1975 to 67% in 2011, while 5-year survival for acute lymphocytic leukemia rose from 57% to 91% over that time period, the investigators reported.
The authors reported no conflicts of interest.
FROM CA: A CANCER JOURNAL FOR CLINICIANS
Combining inhibitors to treat AML

Preclinical research has revealed a treatment approach that could prove effective against acute myeloid leukemia (AML).
Researchers tested the IAP inhibitor birinapant in combination with p38 inhibitors and observed antileukemic activity in mouse models of AML and samples from patients with the disease.
Combination treatment proved more effective than either agent alone, and the combination was less toxic than single-agent chemotherapy.
Najoua Lalaoui, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia, and her colleagues conducted this research and relayed the results in an article published in Cancer Cell.
The researchers generated several mouse models of AML—MLL-ENL ± NRasG12D, MLL-AF9 ± NrasG12D, AML1-ETO9a + NrasG12D, CBFβ-MYH11 + NrasG12D, NUP98-HoxA9, and HoxA9/Meis1.
In these models, the team tested birinapant with 1 of 2 p38 inhibitors—LY2228820 or SCIO-469—or with the MK2 inhibitor PF-3644022. They said each combination “dramatically” increased cell death, when compared to birinapant alone, in most models. The exceptions were AML1-ETO9a + NrasG12D and CBFβ-MYH11 + NrasG12D.
Next, the researchers tested LY2228820 plus birinapant in samples from 8 AML patients. The samples had FLT3-ITD mutations (patients 1, 2, 4, 6, and 7), a FLT3 D835 missense mutation (patient 4), nucleophosmin exon-12 mutations (patients 2 and 4), an MLL translocation (patient 3), inv(3) (patient 1), and inv(16) (patient 8).
All 8 samples were sensitive to birinapant alone. And although LY2228820 alone did not induce cell death in any of the samples, the drug had a synergistic effect with birinapant in 4 of the samples (patients 2, 3, 4, and 7).
The researchers also found that peripheral blood mononuclear cells from healthy donors proved more resistant to combination LY2228820 (at 10 µM) and birinapant (at 500 nM) than to cytarabine (10 µM), daunorubicin (at 0.4 µM), or idarubicin (at 0.4 µM).
In addition, 4 weeks of treatment with birinapant and LY2228820 was well-tolerated in mice without tumors.
Finally, the researchers tested birinapant and LY2228820, either alone or in combination, in mouse models of MLL-ENL, MLL-AF9, and NRasG12D mutant/MLL-AF9/Luc AML.
Combination treatment prolonged survival in all 3 models, when compared with mice that received single agents or no treatment. However, unlike in the MLL-ENL and MLL-AF9 models, the combination was unable to cure NRasG12D mutant/MLL-AF9/Luc mice of their leukemia.
“Our findings have made us hopeful that a combination of birinapant and a p38 inhibitor may be more effective in treating AML than current therapies and also have less toxicity for patients,” Dr Lalaoui said.
“We tested forms of AML that are highly resistant to chemotherapy and found that birinapant and p38 inhibitors could even kill these cancer cells, which is great news.”
Birinapant is being developed by TetraLogic Pharmaceuticals Corporation, and some of the researchers involved in this work reported relationships with the company. ![]()

Preclinical research has revealed a treatment approach that could prove effective against acute myeloid leukemia (AML).
Researchers tested the IAP inhibitor birinapant in combination with p38 inhibitors and observed antileukemic activity in mouse models of AML and samples from patients with the disease.
Combination treatment proved more effective than either agent alone, and the combination was less toxic than single-agent chemotherapy.
Najoua Lalaoui, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia, and her colleagues conducted this research and relayed the results in an article published in Cancer Cell.
The researchers generated several mouse models of AML—MLL-ENL ± NRasG12D, MLL-AF9 ± NrasG12D, AML1-ETO9a + NrasG12D, CBFβ-MYH11 + NrasG12D, NUP98-HoxA9, and HoxA9/Meis1.
In these models, the team tested birinapant with 1 of 2 p38 inhibitors—LY2228820 or SCIO-469—or with the MK2 inhibitor PF-3644022. They said each combination “dramatically” increased cell death, when compared to birinapant alone, in most models. The exceptions were AML1-ETO9a + NrasG12D and CBFβ-MYH11 + NrasG12D.
Next, the researchers tested LY2228820 plus birinapant in samples from 8 AML patients. The samples had FLT3-ITD mutations (patients 1, 2, 4, 6, and 7), a FLT3 D835 missense mutation (patient 4), nucleophosmin exon-12 mutations (patients 2 and 4), an MLL translocation (patient 3), inv(3) (patient 1), and inv(16) (patient 8).
All 8 samples were sensitive to birinapant alone. And although LY2228820 alone did not induce cell death in any of the samples, the drug had a synergistic effect with birinapant in 4 of the samples (patients 2, 3, 4, and 7).
The researchers also found that peripheral blood mononuclear cells from healthy donors proved more resistant to combination LY2228820 (at 10 µM) and birinapant (at 500 nM) than to cytarabine (10 µM), daunorubicin (at 0.4 µM), or idarubicin (at 0.4 µM).
In addition, 4 weeks of treatment with birinapant and LY2228820 was well-tolerated in mice without tumors.
Finally, the researchers tested birinapant and LY2228820, either alone or in combination, in mouse models of MLL-ENL, MLL-AF9, and NRasG12D mutant/MLL-AF9/Luc AML.
Combination treatment prolonged survival in all 3 models, when compared with mice that received single agents or no treatment. However, unlike in the MLL-ENL and MLL-AF9 models, the combination was unable to cure NRasG12D mutant/MLL-AF9/Luc mice of their leukemia.
“Our findings have made us hopeful that a combination of birinapant and a p38 inhibitor may be more effective in treating AML than current therapies and also have less toxicity for patients,” Dr Lalaoui said.
“We tested forms of AML that are highly resistant to chemotherapy and found that birinapant and p38 inhibitors could even kill these cancer cells, which is great news.”
Birinapant is being developed by TetraLogic Pharmaceuticals Corporation, and some of the researchers involved in this work reported relationships with the company. ![]()

Preclinical research has revealed a treatment approach that could prove effective against acute myeloid leukemia (AML).
Researchers tested the IAP inhibitor birinapant in combination with p38 inhibitors and observed antileukemic activity in mouse models of AML and samples from patients with the disease.
Combination treatment proved more effective than either agent alone, and the combination was less toxic than single-agent chemotherapy.
Najoua Lalaoui, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia, and her colleagues conducted this research and relayed the results in an article published in Cancer Cell.
The researchers generated several mouse models of AML—MLL-ENL ± NRasG12D, MLL-AF9 ± NrasG12D, AML1-ETO9a + NrasG12D, CBFβ-MYH11 + NrasG12D, NUP98-HoxA9, and HoxA9/Meis1.
In these models, the team tested birinapant with 1 of 2 p38 inhibitors—LY2228820 or SCIO-469—or with the MK2 inhibitor PF-3644022. They said each combination “dramatically” increased cell death, when compared to birinapant alone, in most models. The exceptions were AML1-ETO9a + NrasG12D and CBFβ-MYH11 + NrasG12D.
Next, the researchers tested LY2228820 plus birinapant in samples from 8 AML patients. The samples had FLT3-ITD mutations (patients 1, 2, 4, 6, and 7), a FLT3 D835 missense mutation (patient 4), nucleophosmin exon-12 mutations (patients 2 and 4), an MLL translocation (patient 3), inv(3) (patient 1), and inv(16) (patient 8).
All 8 samples were sensitive to birinapant alone. And although LY2228820 alone did not induce cell death in any of the samples, the drug had a synergistic effect with birinapant in 4 of the samples (patients 2, 3, 4, and 7).
The researchers also found that peripheral blood mononuclear cells from healthy donors proved more resistant to combination LY2228820 (at 10 µM) and birinapant (at 500 nM) than to cytarabine (10 µM), daunorubicin (at 0.4 µM), or idarubicin (at 0.4 µM).
In addition, 4 weeks of treatment with birinapant and LY2228820 was well-tolerated in mice without tumors.
Finally, the researchers tested birinapant and LY2228820, either alone or in combination, in mouse models of MLL-ENL, MLL-AF9, and NRasG12D mutant/MLL-AF9/Luc AML.
Combination treatment prolonged survival in all 3 models, when compared with mice that received single agents or no treatment. However, unlike in the MLL-ENL and MLL-AF9 models, the combination was unable to cure NRasG12D mutant/MLL-AF9/Luc mice of their leukemia.
“Our findings have made us hopeful that a combination of birinapant and a p38 inhibitor may be more effective in treating AML than current therapies and also have less toxicity for patients,” Dr Lalaoui said.
“We tested forms of AML that are highly resistant to chemotherapy and found that birinapant and p38 inhibitors could even kill these cancer cells, which is great news.”
Birinapant is being developed by TetraLogic Pharmaceuticals Corporation, and some of the researchers involved in this work reported relationships with the company. ![]()
EC grants venetoclax orphan designation for AML

The European Commission has granted orphan drug designation for the oral BCL-2 inhibitor venetoclax to treat acute myeloid leukemia (AML).
The EC grants orphan designation to products intended to treat, prevent, or diagnose a life-threatening condition affecting up to 5 in 10,000 people in the
European Union. The product must provide significant benefit to those affected by the condition.
Orphan drug designation from the EC provides companies with certain development incentives, including protocol assistance, a type of scientific advice specific for orphan drugs, and 10 years of market exclusivity once the drug is on the market.
Venetoclax is being developed by AbbVie in partnership with Genentech and Roche.
Phase 2 study
Results from a phase 2 study of venetoclax in AML were presented at ASH 2014. At that time, the trial had enrolled 32 patients, 30 of whom had relapsed or refractory disease. Patients had a median age of 71 (range, 19 to 84), and half were male.
The overall response rate was 15.5%, with 1 patient achieving a complete response (CR) and 4 achieving a CR with incomplete count recovery (CRi).
The researchers noted that 3 of the patients who had a CR/CRi had IDH mutations. Two of these patients also achieved minimal residual disease negativity.
The median bone marrow blast count in evaluable patients decreased 36% after treatment, and 6 patients (19%) had at least a 50% reduction in bone marrow blasts.
Common adverse events following treatment (occurring in at least 25% of patients) included nausea, diarrhea, fatigue, neutropenia, and vomiting.
Grade 3/4 adverse events (occurring in 3 or more patients) included febrile neutropenia, anemia, and pneumonia. No patient died as a result of treatment-related adverse events. ![]()

The European Commission has granted orphan drug designation for the oral BCL-2 inhibitor venetoclax to treat acute myeloid leukemia (AML).
The EC grants orphan designation to products intended to treat, prevent, or diagnose a life-threatening condition affecting up to 5 in 10,000 people in the
European Union. The product must provide significant benefit to those affected by the condition.
Orphan drug designation from the EC provides companies with certain development incentives, including protocol assistance, a type of scientific advice specific for orphan drugs, and 10 years of market exclusivity once the drug is on the market.
Venetoclax is being developed by AbbVie in partnership with Genentech and Roche.
Phase 2 study
Results from a phase 2 study of venetoclax in AML were presented at ASH 2014. At that time, the trial had enrolled 32 patients, 30 of whom had relapsed or refractory disease. Patients had a median age of 71 (range, 19 to 84), and half were male.
The overall response rate was 15.5%, with 1 patient achieving a complete response (CR) and 4 achieving a CR with incomplete count recovery (CRi).
The researchers noted that 3 of the patients who had a CR/CRi had IDH mutations. Two of these patients also achieved minimal residual disease negativity.
The median bone marrow blast count in evaluable patients decreased 36% after treatment, and 6 patients (19%) had at least a 50% reduction in bone marrow blasts.
Common adverse events following treatment (occurring in at least 25% of patients) included nausea, diarrhea, fatigue, neutropenia, and vomiting.
Grade 3/4 adverse events (occurring in 3 or more patients) included febrile neutropenia, anemia, and pneumonia. No patient died as a result of treatment-related adverse events. ![]()

The European Commission has granted orphan drug designation for the oral BCL-2 inhibitor venetoclax to treat acute myeloid leukemia (AML).
The EC grants orphan designation to products intended to treat, prevent, or diagnose a life-threatening condition affecting up to 5 in 10,000 people in the
European Union. The product must provide significant benefit to those affected by the condition.
Orphan drug designation from the EC provides companies with certain development incentives, including protocol assistance, a type of scientific advice specific for orphan drugs, and 10 years of market exclusivity once the drug is on the market.
Venetoclax is being developed by AbbVie in partnership with Genentech and Roche.
Phase 2 study
Results from a phase 2 study of venetoclax in AML were presented at ASH 2014. At that time, the trial had enrolled 32 patients, 30 of whom had relapsed or refractory disease. Patients had a median age of 71 (range, 19 to 84), and half were male.
The overall response rate was 15.5%, with 1 patient achieving a complete response (CR) and 4 achieving a CR with incomplete count recovery (CRi).
The researchers noted that 3 of the patients who had a CR/CRi had IDH mutations. Two of these patients also achieved minimal residual disease negativity.
The median bone marrow blast count in evaluable patients decreased 36% after treatment, and 6 patients (19%) had at least a 50% reduction in bone marrow blasts.
Common adverse events following treatment (occurring in at least 25% of patients) included nausea, diarrhea, fatigue, neutropenia, and vomiting.
Grade 3/4 adverse events (occurring in 3 or more patients) included febrile neutropenia, anemia, and pneumonia. No patient died as a result of treatment-related adverse events. ![]()
DNA delivery vehicles may circumvent drug resistance in AML

Image courtesy of PNAS
DNA origami nanostructures may be used to overcome drug resistance in acute myeloid leukemia (AML), according to preclinical research published in the journal Small.
Researchers found they could create these nanostructures in 10 minutes and load them with the anthracycline daunorubicin.
When the team introduced the structures to daunorubicin-resistant AML cells, the drug delivery vehicles entered the cells via endocytosis.
This allowed the drug to bypass defenses in the cell membrane that are effective against the free drug.
Once the nanostructures broke down, daunorubicin flooded the cells and killed them off.
Other research groups have used this delivery technique to overcome drug resistance in solid tumors, but this is the first time researchers have shown the same technique works on drug-resistant leukemia cells.
To create the DNA origami nanostructures, the researchers used the genome of a common bacteriophage and synthetic strands that were designed to fold up the bacteriophage DNA.
Although the folded-up shape performs a function, the DNA itself does not, explained Patrick Halley, a graduate student at The Ohio State University in Columbus.
“[T]he DNA capsule doesn’t do anything except hold a shape,” Halley said. “It’s just a static, rigid structure that carries things. It doesn’t encode any proteins or do anything else that we normally think of DNA as doing.”
The researchers tested the DNA origami nanostructures in AML cell lines that had developed resistance to daunorubicin. When molecules of daunorubicin enter these cells, the cells recognize the drug molecules and eject them through openings in the cell wall.
“Cancer cells have novel ways of resisting drugs, like these ‘pumps,’ and the exciting part of packaging the drug this way is that we can circumvent those defenses so that the drug accumulates in the cancer cell and causes it to die,” said John Byrd, MD, of The Ohio State University.
“Potentially, we can also tailor these structures to make them deliver drugs selectively to cancer cells and not to other parts of the body where they can cause side effects.”
In tests, the resistant AML cells effectively absorbed molecules of daunorubicin when they were hidden inside the rod-shaped nanostructures.
The researchers tracked the nanostructures inside the cells using fluorescent tags. Each structure measures about 15 nanometers wide and 100 nanometers long, and each has 4 hollow, open-ended interior compartments.
Study author Christopher Lucas, PhD, of The Ohio State University, said the design of the nanostructures maximizes the surface area available to carry the drug.
“The way daunorubicin works is it tucks into the cancer cell’s DNA and prevents it from replicating,” Dr Lucas said. “So we designed a capsule structure that would have lots of accessible DNA base-pairs for it to tuck into. When the capsule breaks down, the drug molecules are freed to flood the cell.”
The researchers said they designed the nanostructures to be strong and stable so they wouldn’t fully disintegrate and release the bulk of the drug until it was too late for the cells to eject them.
And that’s what the team observed with a fluorescence microscope. The cells drew the nanostructures into the organelles that would normally digest them (if they were food).
When the nanostructures broke down, the drug flooded the cells and caused them to disintegrate. Most cells died within the first 15 hours after consuming the nanostructures.
“DNA origami nanostructures have a lot of potential for drug delivery, not just for making effective drug delivery vehicles, but enabling new ways to study drug delivery,” said Carlos Castro, PhD, of The Ohio State University.
“For instance, we can vary the shape or mechanical stiffness of a structure very precisely and see how that affects entry into cells.”
Dr Castro said he hopes to create a streamlined and economically viable process for building DNA origami nanostructures as part of a modular drug delivery system.
Dr Byrd said the technique should work on most any form of drug-resistant cancer if further research shows it can be translated to animal models. ![]()

Image courtesy of PNAS
DNA origami nanostructures may be used to overcome drug resistance in acute myeloid leukemia (AML), according to preclinical research published in the journal Small.
Researchers found they could create these nanostructures in 10 minutes and load them with the anthracycline daunorubicin.
When the team introduced the structures to daunorubicin-resistant AML cells, the drug delivery vehicles entered the cells via endocytosis.
This allowed the drug to bypass defenses in the cell membrane that are effective against the free drug.
Once the nanostructures broke down, daunorubicin flooded the cells and killed them off.
Other research groups have used this delivery technique to overcome drug resistance in solid tumors, but this is the first time researchers have shown the same technique works on drug-resistant leukemia cells.
To create the DNA origami nanostructures, the researchers used the genome of a common bacteriophage and synthetic strands that were designed to fold up the bacteriophage DNA.
Although the folded-up shape performs a function, the DNA itself does not, explained Patrick Halley, a graduate student at The Ohio State University in Columbus.
“[T]he DNA capsule doesn’t do anything except hold a shape,” Halley said. “It’s just a static, rigid structure that carries things. It doesn’t encode any proteins or do anything else that we normally think of DNA as doing.”
The researchers tested the DNA origami nanostructures in AML cell lines that had developed resistance to daunorubicin. When molecules of daunorubicin enter these cells, the cells recognize the drug molecules and eject them through openings in the cell wall.
“Cancer cells have novel ways of resisting drugs, like these ‘pumps,’ and the exciting part of packaging the drug this way is that we can circumvent those defenses so that the drug accumulates in the cancer cell and causes it to die,” said John Byrd, MD, of The Ohio State University.
“Potentially, we can also tailor these structures to make them deliver drugs selectively to cancer cells and not to other parts of the body where they can cause side effects.”
In tests, the resistant AML cells effectively absorbed molecules of daunorubicin when they were hidden inside the rod-shaped nanostructures.
The researchers tracked the nanostructures inside the cells using fluorescent tags. Each structure measures about 15 nanometers wide and 100 nanometers long, and each has 4 hollow, open-ended interior compartments.
Study author Christopher Lucas, PhD, of The Ohio State University, said the design of the nanostructures maximizes the surface area available to carry the drug.
“The way daunorubicin works is it tucks into the cancer cell’s DNA and prevents it from replicating,” Dr Lucas said. “So we designed a capsule structure that would have lots of accessible DNA base-pairs for it to tuck into. When the capsule breaks down, the drug molecules are freed to flood the cell.”
The researchers said they designed the nanostructures to be strong and stable so they wouldn’t fully disintegrate and release the bulk of the drug until it was too late for the cells to eject them.
And that’s what the team observed with a fluorescence microscope. The cells drew the nanostructures into the organelles that would normally digest them (if they were food).
When the nanostructures broke down, the drug flooded the cells and caused them to disintegrate. Most cells died within the first 15 hours after consuming the nanostructures.
“DNA origami nanostructures have a lot of potential for drug delivery, not just for making effective drug delivery vehicles, but enabling new ways to study drug delivery,” said Carlos Castro, PhD, of The Ohio State University.
“For instance, we can vary the shape or mechanical stiffness of a structure very precisely and see how that affects entry into cells.”
Dr Castro said he hopes to create a streamlined and economically viable process for building DNA origami nanostructures as part of a modular drug delivery system.
Dr Byrd said the technique should work on most any form of drug-resistant cancer if further research shows it can be translated to animal models. ![]()

Image courtesy of PNAS
DNA origami nanostructures may be used to overcome drug resistance in acute myeloid leukemia (AML), according to preclinical research published in the journal Small.
Researchers found they could create these nanostructures in 10 minutes and load them with the anthracycline daunorubicin.
When the team introduced the structures to daunorubicin-resistant AML cells, the drug delivery vehicles entered the cells via endocytosis.
This allowed the drug to bypass defenses in the cell membrane that are effective against the free drug.
Once the nanostructures broke down, daunorubicin flooded the cells and killed them off.
Other research groups have used this delivery technique to overcome drug resistance in solid tumors, but this is the first time researchers have shown the same technique works on drug-resistant leukemia cells.
To create the DNA origami nanostructures, the researchers used the genome of a common bacteriophage and synthetic strands that were designed to fold up the bacteriophage DNA.
Although the folded-up shape performs a function, the DNA itself does not, explained Patrick Halley, a graduate student at The Ohio State University in Columbus.
“[T]he DNA capsule doesn’t do anything except hold a shape,” Halley said. “It’s just a static, rigid structure that carries things. It doesn’t encode any proteins or do anything else that we normally think of DNA as doing.”
The researchers tested the DNA origami nanostructures in AML cell lines that had developed resistance to daunorubicin. When molecules of daunorubicin enter these cells, the cells recognize the drug molecules and eject them through openings in the cell wall.
“Cancer cells have novel ways of resisting drugs, like these ‘pumps,’ and the exciting part of packaging the drug this way is that we can circumvent those defenses so that the drug accumulates in the cancer cell and causes it to die,” said John Byrd, MD, of The Ohio State University.
“Potentially, we can also tailor these structures to make them deliver drugs selectively to cancer cells and not to other parts of the body where they can cause side effects.”
In tests, the resistant AML cells effectively absorbed molecules of daunorubicin when they were hidden inside the rod-shaped nanostructures.
The researchers tracked the nanostructures inside the cells using fluorescent tags. Each structure measures about 15 nanometers wide and 100 nanometers long, and each has 4 hollow, open-ended interior compartments.
Study author Christopher Lucas, PhD, of The Ohio State University, said the design of the nanostructures maximizes the surface area available to carry the drug.
“The way daunorubicin works is it tucks into the cancer cell’s DNA and prevents it from replicating,” Dr Lucas said. “So we designed a capsule structure that would have lots of accessible DNA base-pairs for it to tuck into. When the capsule breaks down, the drug molecules are freed to flood the cell.”
The researchers said they designed the nanostructures to be strong and stable so they wouldn’t fully disintegrate and release the bulk of the drug until it was too late for the cells to eject them.
And that’s what the team observed with a fluorescence microscope. The cells drew the nanostructures into the organelles that would normally digest them (if they were food).
When the nanostructures broke down, the drug flooded the cells and caused them to disintegrate. Most cells died within the first 15 hours after consuming the nanostructures.
“DNA origami nanostructures have a lot of potential for drug delivery, not just for making effective drug delivery vehicles, but enabling new ways to study drug delivery,” said Carlos Castro, PhD, of The Ohio State University.
“For instance, we can vary the shape or mechanical stiffness of a structure very precisely and see how that affects entry into cells.”
Dr Castro said he hopes to create a streamlined and economically viable process for building DNA origami nanostructures as part of a modular drug delivery system.
Dr Byrd said the technique should work on most any form of drug-resistant cancer if further research shows it can be translated to animal models. ![]()
Factors appear to confer poor survival in AML

receiving chemotherapy
Photo by Rhoda Baer
A study published in the British Journal of Hematology has revealed factors that appear to affect survival in patients with acute myeloid leukemia (AML).
The research showed that death was more likely among AML patients treated at centers not affiliated with the National Cancer Institute (NCI).
Death was also more likely for black patients, older patients, those without health insurance, and those who lived in poorer neighborhoods.
“Our study reveals that survival inequalities persist among vulnerable patients with acute myeloid leukemia, such as the uninsured, those of black race/ethnicity, and adolescents and young adults,” said study author Renata Abrahão, MD, of Cancer Prevention Institute of California.
“This study can serve as a baseline to compare changes in survival that may result from potential improvements in health insurance coverage following the implementation of the Affordable Care Act.”
Dr Abrahão and her colleagues analyzed 3935 AML patients who were 39 or younger between 1988 and 2011. The team used data from the California Cancer Registry, which participates in the Surveillance, Epidemiology and End Results program of the NCI.
The data revealed an increase over time in the 5-year survival rate, from 32.9% in 1988–1995 to 50% in 2004–2011. However, 58% of the patients (n=2272) died during follow-up. The overall median follow-up was 10 years, and the median time to death was 0.9 years.
A multivariate analysis revealed several subgroups of patients with worse survival.
Older patients had a greater risk of death when compared to patients ages 0 to 9. The hazard ratio (HR) was 1.23 for patients ages 10 to 19, 1.34 for patients ages 20 to 29, and 1.55 for patients ages 30 to 39.
Black patients had an increased risk of death as well. When compared with white patients, the HR was 1.27 for black patients, 1.05 for Hispanic patients, and 0.98 for Asian/Pacific Islanders.
Patients living in the neighborhoods with the lowest socioeconomic status had an HR of 1.14. And patients who received their initial care at a hospital not affiliated with the NCI had an HR of 1.18.
Health insurance information was only available for patients diagnosed from 1996 to 2011. Among these patients, the risk of death was higher among uninsured patients (HR=1.34) than among privately insured patients, but there was no difference between privately and publicly insured patients.
Explaining the findings
The researchers said AML diagnosis in older children, adolescents, and young adults may require more intensive treatment than in young children, which may lead to a higher probability of treatment-related complications. And recent studies have shown the biology of pediatric AML differs from adult AML, which may lead to a favorable prognosis in younger patients.
In addition, older children, adolescents, and young adults are less likely to participate in clinical trials and more likely to receive treatment at hospitals not affiliated with the NCI, when compared to younger children.
The researchers said it is not clear what factors accounted for the inferior survival observed among black patients. The team speculated that genetics may contribute to the difference in chemotherapy response or that black patients had less access to chemotherapy and other treatments such as hematopoietic stem cell transplant.
The association between lower socioeconomic status and death suggests a lack of access to treatment. The same can be said for the association between death and a lack of insurance.
“[T]his study showed that survival after AML remains low among young patients and highlights the need for new therapeutic regimens to treat this disease with various subtypes,” Dr Abrahão said.
“We emphasized the importance of linking population-based data with genetic and clinical information contained in the patients’ medical records in order to better understand the causes of survival inequalities.” ![]()

receiving chemotherapy
Photo by Rhoda Baer
A study published in the British Journal of Hematology has revealed factors that appear to affect survival in patients with acute myeloid leukemia (AML).
The research showed that death was more likely among AML patients treated at centers not affiliated with the National Cancer Institute (NCI).
Death was also more likely for black patients, older patients, those without health insurance, and those who lived in poorer neighborhoods.
“Our study reveals that survival inequalities persist among vulnerable patients with acute myeloid leukemia, such as the uninsured, those of black race/ethnicity, and adolescents and young adults,” said study author Renata Abrahão, MD, of Cancer Prevention Institute of California.
“This study can serve as a baseline to compare changes in survival that may result from potential improvements in health insurance coverage following the implementation of the Affordable Care Act.”
Dr Abrahão and her colleagues analyzed 3935 AML patients who were 39 or younger between 1988 and 2011. The team used data from the California Cancer Registry, which participates in the Surveillance, Epidemiology and End Results program of the NCI.
The data revealed an increase over time in the 5-year survival rate, from 32.9% in 1988–1995 to 50% in 2004–2011. However, 58% of the patients (n=2272) died during follow-up. The overall median follow-up was 10 years, and the median time to death was 0.9 years.
A multivariate analysis revealed several subgroups of patients with worse survival.
Older patients had a greater risk of death when compared to patients ages 0 to 9. The hazard ratio (HR) was 1.23 for patients ages 10 to 19, 1.34 for patients ages 20 to 29, and 1.55 for patients ages 30 to 39.
Black patients had an increased risk of death as well. When compared with white patients, the HR was 1.27 for black patients, 1.05 for Hispanic patients, and 0.98 for Asian/Pacific Islanders.
Patients living in the neighborhoods with the lowest socioeconomic status had an HR of 1.14. And patients who received their initial care at a hospital not affiliated with the NCI had an HR of 1.18.
Health insurance information was only available for patients diagnosed from 1996 to 2011. Among these patients, the risk of death was higher among uninsured patients (HR=1.34) than among privately insured patients, but there was no difference between privately and publicly insured patients.
Explaining the findings
The researchers said AML diagnosis in older children, adolescents, and young adults may require more intensive treatment than in young children, which may lead to a higher probability of treatment-related complications. And recent studies have shown the biology of pediatric AML differs from adult AML, which may lead to a favorable prognosis in younger patients.
In addition, older children, adolescents, and young adults are less likely to participate in clinical trials and more likely to receive treatment at hospitals not affiliated with the NCI, when compared to younger children.
The researchers said it is not clear what factors accounted for the inferior survival observed among black patients. The team speculated that genetics may contribute to the difference in chemotherapy response or that black patients had less access to chemotherapy and other treatments such as hematopoietic stem cell transplant.
The association between lower socioeconomic status and death suggests a lack of access to treatment. The same can be said for the association between death and a lack of insurance.
“[T]his study showed that survival after AML remains low among young patients and highlights the need for new therapeutic regimens to treat this disease with various subtypes,” Dr Abrahão said.
“We emphasized the importance of linking population-based data with genetic and clinical information contained in the patients’ medical records in order to better understand the causes of survival inequalities.” ![]()

receiving chemotherapy
Photo by Rhoda Baer
A study published in the British Journal of Hematology has revealed factors that appear to affect survival in patients with acute myeloid leukemia (AML).
The research showed that death was more likely among AML patients treated at centers not affiliated with the National Cancer Institute (NCI).
Death was also more likely for black patients, older patients, those without health insurance, and those who lived in poorer neighborhoods.
“Our study reveals that survival inequalities persist among vulnerable patients with acute myeloid leukemia, such as the uninsured, those of black race/ethnicity, and adolescents and young adults,” said study author Renata Abrahão, MD, of Cancer Prevention Institute of California.
“This study can serve as a baseline to compare changes in survival that may result from potential improvements in health insurance coverage following the implementation of the Affordable Care Act.”
Dr Abrahão and her colleagues analyzed 3935 AML patients who were 39 or younger between 1988 and 2011. The team used data from the California Cancer Registry, which participates in the Surveillance, Epidemiology and End Results program of the NCI.
The data revealed an increase over time in the 5-year survival rate, from 32.9% in 1988–1995 to 50% in 2004–2011. However, 58% of the patients (n=2272) died during follow-up. The overall median follow-up was 10 years, and the median time to death was 0.9 years.
A multivariate analysis revealed several subgroups of patients with worse survival.
Older patients had a greater risk of death when compared to patients ages 0 to 9. The hazard ratio (HR) was 1.23 for patients ages 10 to 19, 1.34 for patients ages 20 to 29, and 1.55 for patients ages 30 to 39.
Black patients had an increased risk of death as well. When compared with white patients, the HR was 1.27 for black patients, 1.05 for Hispanic patients, and 0.98 for Asian/Pacific Islanders.
Patients living in the neighborhoods with the lowest socioeconomic status had an HR of 1.14. And patients who received their initial care at a hospital not affiliated with the NCI had an HR of 1.18.
Health insurance information was only available for patients diagnosed from 1996 to 2011. Among these patients, the risk of death was higher among uninsured patients (HR=1.34) than among privately insured patients, but there was no difference between privately and publicly insured patients.
Explaining the findings
The researchers said AML diagnosis in older children, adolescents, and young adults may require more intensive treatment than in young children, which may lead to a higher probability of treatment-related complications. And recent studies have shown the biology of pediatric AML differs from adult AML, which may lead to a favorable prognosis in younger patients.
In addition, older children, adolescents, and young adults are less likely to participate in clinical trials and more likely to receive treatment at hospitals not affiliated with the NCI, when compared to younger children.
The researchers said it is not clear what factors accounted for the inferior survival observed among black patients. The team speculated that genetics may contribute to the difference in chemotherapy response or that black patients had less access to chemotherapy and other treatments such as hematopoietic stem cell transplant.
The association between lower socioeconomic status and death suggests a lack of access to treatment. The same can be said for the association between death and a lack of insurance.
“[T]his study showed that survival after AML remains low among young patients and highlights the need for new therapeutic regimens to treat this disease with various subtypes,” Dr Abrahão said.
“We emphasized the importance of linking population-based data with genetic and clinical information contained in the patients’ medical records in order to better understand the causes of survival inequalities.”
Drug granted breakthrough designation for AML
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for midostaurin (PKC412) to treat acute myeloid leukemia (AML).
Midostaurin is a multi-targeted kinase inhibitor being developed for adults with newly diagnosed AML who are FLT3-positive, as detected by an FDA-approved test, and who are eligible to receive standard induction and consolidation chemotherapy.
Breakthrough therapy designation is intended to expedite the development and review of new medicines intended to treat serious or life-threatening conditions. The therapy must demonstrate substantial improvement over an available therapy on at least one clinically significant endpoint.
The designation includes all of the fast track program features, as well as more intensive FDA guidance on an efficient drug development program.
Phase 3 trial
The breakthrough designation for midostaurin is primarily based on the results of the phase 3 RATIFY trial, which were presented at the 2015 ASH Annual Meeting.
The trial included 717 patients with newly diagnosed, FLT3-positive AML who were younger than 60 at enrollment. All of the patients received standard induction and consolidation therapy. Roughly half also received midostaurin (n=360), while the other half received placebo (n=357).
Patients who received midostaurin experienced a significant improvement in overall survival (hazard ratio=0.77, P=0.0074). The median overall survival was 74.4 months in the midostaurin arm and 25.6 months in the placebo arm.
The median event-free survival was 8 months in the midostaurin arm and 3.6 months in the placebo arm (P=0.0032). The 5-year event-free survival was 27.5% for midostaurin and 19.3% for placebo.
There was no significant difference between the treatment arms with regard to most non-hematologic grade 3/4 adverse events. The exception was rash/desquamation, which occurred in 13% of patients in the midostaurin arm and 8% of patients in the placebo arm (P=0.02).
Other grade 3/4 non-hematologic events occurring in 10% of patients or more included, in the midostaurin and placebo arms, respectively: febrile neutropenia (81%, 82%), infection (40%, 38%), diarrhea (15%, 16%), hypokalemia (13%, 17%), pain (13%, 13%), other infection (12%, 12%), ALT/SGPT (12%, 9%), and fatigue (9%, 11%).
There were 18 deaths (5%) in the midostaurin arm and 19 (5.3%) in the placebo arm during induction and consolidation.
Midostaurin development
Novartis has opened a Global Individual Patient Program (compassionate use program) and a US Expanded Treatment Protocol (ETP) to enable midostaurin access. Patients 18 years of age and older with newly diagnosed FLT3-mutated AML who are able to receive standard induction and consolidation therapy will be considered.
To help identify patients who may have a FLT3 mutation and potentially benefit from treatment with midostaurin, Novartis is collaborating with Invivoscribe Technologies, Inc. which is leading regulatory submissions for a companion diagnostic.
Midostaurin is also being investigated for the treatment of aggressive systemic mastocytosis/mast cell leukemia.

The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for midostaurin (PKC412) to treat acute myeloid leukemia (AML).
Midostaurin is a multi-targeted kinase inhibitor being developed for adults with newly diagnosed AML who are FLT3-positive, as detected by an FDA-approved test, and who are eligible to receive standard induction and consolidation chemotherapy.
Breakthrough therapy designation is intended to expedite the development and review of new medicines intended to treat serious or life-threatening conditions. The therapy must demonstrate substantial improvement over an available therapy on at least one clinically significant endpoint.
The designation includes all of the fast track program features, as well as more intensive FDA guidance on an efficient drug development program.
Phase 3 trial
The breakthrough designation for midostaurin is primarily based on the results of the phase 3 RATIFY trial, which were presented at the 2015 ASH Annual Meeting.
The trial included 717 patients with newly diagnosed, FLT3-positive AML who were younger than 60 at enrollment. All of the patients received standard induction and consolidation therapy. Roughly half also received midostaurin (n=360), while the other half received placebo (n=357).
Patients who received midostaurin experienced a significant improvement in overall survival (hazard ratio=0.77, P=0.0074). The median overall survival was 74.4 months in the midostaurin arm and 25.6 months in the placebo arm.
The median event-free survival was 8 months in the midostaurin arm and 3.6 months in the placebo arm (P=0.0032). The 5-year event-free survival was 27.5% for midostaurin and 19.3% for placebo.
There was no significant difference between the treatment arms with regard to most non-hematologic grade 3/4 adverse events. The exception was rash/desquamation, which occurred in 13% of patients in the midostaurin arm and 8% of patients in the placebo arm (P=0.02).
Other grade 3/4 non-hematologic events occurring in 10% of patients or more included, in the midostaurin and placebo arms, respectively: febrile neutropenia (81%, 82%), infection (40%, 38%), diarrhea (15%, 16%), hypokalemia (13%, 17%), pain (13%, 13%), other infection (12%, 12%), ALT/SGPT (12%, 9%), and fatigue (9%, 11%).
There were 18 deaths (5%) in the midostaurin arm and 19 (5.3%) in the placebo arm during induction and consolidation.
Midostaurin development
Novartis has opened a Global Individual Patient Program (compassionate use program) and a US Expanded Treatment Protocol (ETP) to enable midostaurin access. Patients 18 years of age and older with newly diagnosed FLT3-mutated AML who are able to receive standard induction and consolidation therapy will be considered.
To help identify patients who may have a FLT3 mutation and potentially benefit from treatment with midostaurin, Novartis is collaborating with Invivoscribe Technologies, Inc. which is leading regulatory submissions for a companion diagnostic.
Midostaurin is also being investigated for the treatment of aggressive systemic mastocytosis/mast cell leukemia.

The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for midostaurin (PKC412) to treat acute myeloid leukemia (AML).
Midostaurin is a multi-targeted kinase inhibitor being developed for adults with newly diagnosed AML who are FLT3-positive, as detected by an FDA-approved test, and who are eligible to receive standard induction and consolidation chemotherapy.
Breakthrough therapy designation is intended to expedite the development and review of new medicines intended to treat serious or life-threatening conditions. The therapy must demonstrate substantial improvement over an available therapy on at least one clinically significant endpoint.
The designation includes all of the fast track program features, as well as more intensive FDA guidance on an efficient drug development program.
Phase 3 trial
The breakthrough designation for midostaurin is primarily based on the results of the phase 3 RATIFY trial, which were presented at the 2015 ASH Annual Meeting.
The trial included 717 patients with newly diagnosed, FLT3-positive AML who were younger than 60 at enrollment. All of the patients received standard induction and consolidation therapy. Roughly half also received midostaurin (n=360), while the other half received placebo (n=357).
Patients who received midostaurin experienced a significant improvement in overall survival (hazard ratio=0.77, P=0.0074). The median overall survival was 74.4 months in the midostaurin arm and 25.6 months in the placebo arm.
The median event-free survival was 8 months in the midostaurin arm and 3.6 months in the placebo arm (P=0.0032). The 5-year event-free survival was 27.5% for midostaurin and 19.3% for placebo.
There was no significant difference between the treatment arms with regard to most non-hematologic grade 3/4 adverse events. The exception was rash/desquamation, which occurred in 13% of patients in the midostaurin arm and 8% of patients in the placebo arm (P=0.02).
Other grade 3/4 non-hematologic events occurring in 10% of patients or more included, in the midostaurin and placebo arms, respectively: febrile neutropenia (81%, 82%), infection (40%, 38%), diarrhea (15%, 16%), hypokalemia (13%, 17%), pain (13%, 13%), other infection (12%, 12%), ALT/SGPT (12%, 9%), and fatigue (9%, 11%).
There were 18 deaths (5%) in the midostaurin arm and 19 (5.3%) in the placebo arm during induction and consolidation.
Midostaurin development
Novartis has opened a Global Individual Patient Program (compassionate use program) and a US Expanded Treatment Protocol (ETP) to enable midostaurin access. Patients 18 years of age and older with newly diagnosed FLT3-mutated AML who are able to receive standard induction and consolidation therapy will be considered.
To help identify patients who may have a FLT3 mutation and potentially benefit from treatment with midostaurin, Novartis is collaborating with Invivoscribe Technologies, Inc. which is leading regulatory submissions for a companion diagnostic.
Midostaurin is also being investigated for the treatment of aggressive systemic mastocytosis/mast cell leukemia.
FDA gives breakthrough status to midostaurin for AML
An experimental treatment targeting a form of acute myeloid leukemia has been designated a breakthrough therapy by the Food and Drug Administration, according to the drug’s manufacturer.
Midostaurin (Novartis) is an oral drug used alongside standard chemotherapy for adults with newly-diagnosed AML who are positive for the FMS-like tyrosine 3 (FLT-3) mutation and can undergo chemotherapy. AML has the lowest survival rate of all leukemias, and about one-third of AML patients have the FLT-3 mutation.
The FDA’s breakthrough therapy designation, in place since 2012, is an intensive form of fast-tracking in which the agency offers the manufacturer more guidance on an efficient drug development program and a higher level of organizational support, though future approval is not guaranteed. To qualify, a therapy must come with preliminary clinical evidence demonstrating substantial improvement on at least one clinically significant endpoint over available therapy, according to the agency.
Results from a phase III clinical trial, presented in December 2015 at the 57th annual meeting of the American Society of Hematology, showed that among 717 patients randomized to receive midostaurin alongside standard induction and consolidation chemotherapy or the same chemotherapy protocol alone, the midostaurin group saw significant improvement in overall survival (hazard ratio, 0.77, P = .0074).
Mean OS for patients in the midostaurin arm was 74.7 months (95% CI: 31.7, not attained), compared with 25.6 months for the placebo arm (18.6, 42.9). Median follow-up in the study was 57 months for surviving patients.
In a news release Feb. 19, Novartis said that midostaurin would be submitted for FDA approval for FLT-3-positive AML and that the company had launched compassionate use and expanded access programs allowing newly diagnosed patients aged 18 and older to receive midostaurin alongside standard induction and consolidation therapy. No targeted AML treatments are currently approved by FDA.
FLT3 is a receptor tyrosine kinase that plays a role in the proliferation in the number of certain blood cells. Midostaurin is a multi-targeted kinase inhibitor that is also being investigated for the treatment of aggressive systemic mast cell leukemia, according to Novartis.
An experimental treatment targeting a form of acute myeloid leukemia has been designated a breakthrough therapy by the Food and Drug Administration, according to the drug’s manufacturer.
Midostaurin (Novartis) is an oral drug used alongside standard chemotherapy for adults with newly-diagnosed AML who are positive for the FMS-like tyrosine 3 (FLT-3) mutation and can undergo chemotherapy. AML has the lowest survival rate of all leukemias, and about one-third of AML patients have the FLT-3 mutation.
The FDA’s breakthrough therapy designation, in place since 2012, is an intensive form of fast-tracking in which the agency offers the manufacturer more guidance on an efficient drug development program and a higher level of organizational support, though future approval is not guaranteed. To qualify, a therapy must come with preliminary clinical evidence demonstrating substantial improvement on at least one clinically significant endpoint over available therapy, according to the agency.
Results from a phase III clinical trial, presented in December 2015 at the 57th annual meeting of the American Society of Hematology, showed that among 717 patients randomized to receive midostaurin alongside standard induction and consolidation chemotherapy or the same chemotherapy protocol alone, the midostaurin group saw significant improvement in overall survival (hazard ratio, 0.77, P = .0074).
Mean OS for patients in the midostaurin arm was 74.7 months (95% CI: 31.7, not attained), compared with 25.6 months for the placebo arm (18.6, 42.9). Median follow-up in the study was 57 months for surviving patients.
In a news release Feb. 19, Novartis said that midostaurin would be submitted for FDA approval for FLT-3-positive AML and that the company had launched compassionate use and expanded access programs allowing newly diagnosed patients aged 18 and older to receive midostaurin alongside standard induction and consolidation therapy. No targeted AML treatments are currently approved by FDA.
FLT3 is a receptor tyrosine kinase that plays a role in the proliferation in the number of certain blood cells. Midostaurin is a multi-targeted kinase inhibitor that is also being investigated for the treatment of aggressive systemic mast cell leukemia, according to Novartis.
An experimental treatment targeting a form of acute myeloid leukemia has been designated a breakthrough therapy by the Food and Drug Administration, according to the drug’s manufacturer.
Midostaurin (Novartis) is an oral drug used alongside standard chemotherapy for adults with newly-diagnosed AML who are positive for the FMS-like tyrosine 3 (FLT-3) mutation and can undergo chemotherapy. AML has the lowest survival rate of all leukemias, and about one-third of AML patients have the FLT-3 mutation.
The FDA’s breakthrough therapy designation, in place since 2012, is an intensive form of fast-tracking in which the agency offers the manufacturer more guidance on an efficient drug development program and a higher level of organizational support, though future approval is not guaranteed. To qualify, a therapy must come with preliminary clinical evidence demonstrating substantial improvement on at least one clinically significant endpoint over available therapy, according to the agency.
Results from a phase III clinical trial, presented in December 2015 at the 57th annual meeting of the American Society of Hematology, showed that among 717 patients randomized to receive midostaurin alongside standard induction and consolidation chemotherapy or the same chemotherapy protocol alone, the midostaurin group saw significant improvement in overall survival (hazard ratio, 0.77, P = .0074).
Mean OS for patients in the midostaurin arm was 74.7 months (95% CI: 31.7, not attained), compared with 25.6 months for the placebo arm (18.6, 42.9). Median follow-up in the study was 57 months for surviving patients.
In a news release Feb. 19, Novartis said that midostaurin would be submitted for FDA approval for FLT-3-positive AML and that the company had launched compassionate use and expanded access programs allowing newly diagnosed patients aged 18 and older to receive midostaurin alongside standard induction and consolidation therapy. No targeted AML treatments are currently approved by FDA.
FLT3 is a receptor tyrosine kinase that plays a role in the proliferation in the number of certain blood cells. Midostaurin is a multi-targeted kinase inhibitor that is also being investigated for the treatment of aggressive systemic mast cell leukemia, according to Novartis.