User login
Nilotinib plus chemotherapy pays off for older patients with Ph+ALL
SAN FRANCISCO– The study was small but encouraging: Among 47 older patients with newly diagnosed acute lymphoblastic leukemia positive for the Philadelphia chromosome, 41 had a complete hematologic response to a combination of chemotherapy and the targeted agent nilotinib (Tasigna), report investigators from a European consortium.
“The data I have presented show that the combination of nilotinib with this age-adapted chemotherapy is highly effective. We do have quite a reasonable overall survival estimate at 2 years of just more than 70%,” said Dr. Oliver Ottmann of Goethe University in Frankfurt, on behalf of colleagues in the European Working Group for Adult ALL (EWALL).
The study also shows that although some centers are reluctant to offer allogeneic stem cell transplantation (SCT) to older patients, it is still a viable treatment option in this population, Dr. Ottmann said at a briefing at the annual meeting of the American Society of Hematology.
Although older patients with newly diagnosed Philadelphia-positive (Ph+) ALL have a high complete hematologic response rate (CHR) with imatinib (Gleevec), they generally have a poor prognosis because of a high rate of relapse.
Because nilotinib, a potent inhibitor of the ABL kinase, has good efficacy in the chronic and accelerated phase of Ph+ chronic myeloid leukemia, the EWALL investigators initiated a study to evaluate it in combination with chemotherapy in the front-line setting.
Adults aged 55 years and older with ALL positive for the Philadelphia chromosome and/or BCR-ABL1 fusion who were treatment naive or had not received therapy other than corticosteroids, single-dose vincristine, or three doses of cyclophosphamide were eligible.
Details of the combination regimen are available online.
Briefly, following a prephase with dexamethasone and optional cyclophosphamide, patients receive nilotinib 400 mg twice daily starting with induction and continuously thereafter. During induction, nilotinib is given with intravenous injections of vincristine and dexamethasone for 4 weeks, followed by consolidation with nilotinib, methotrexate, asparaginase and cytarabine. Maintenance consists of nilotinib, 6-mercaptopurine, and methotrexate once weekly for 1 month then every other month, and dexamethasone and vincristine in 2 month intervals up to 24 months.
The data Dr. Ottmann reported come from an interim analysis of the ongoing study. As of August 2014, data on 47 of 56 patients was available for an efficacy analysis, As noted before, the rate of CHR was 87%, occurring in 41 of 47 patients. The treatment evoked a partial response or no response in 2 patients, and there was one death during the induction phase. Additionally, three patients discontinued therapy early and were not included in the assessment, but at least one had a complete response later on, Dr. Ottmann noted.
The median time to a complete response (CR) was 41 days, but CRs occurred as early as 25 days and as late as 62 days after the start of therapy. The remissions at the time of data cutoff appeared to be durable, but follow-up is still early, he said.
Overall survival at a median follow-up for all patients of 8.6 months was 72.7% at 30 months for patients who did not undergo SCT (allowed under the protocol), and 67.1% at 30 months for patients who underwent SCT. This difference was not significant, but only nine patients at the time of data cutoff had undergone transplantation.
“It will be interesting to see how this will proceed if the transplant-free patients will do as well as the others,” Dr. Ottmann said.
An analysis of molecular response by minimal residual disease (MRD) time point showed a significant further increase with the consolidation chemotherapy and kinase inhibitor, emphasizing that “continuing the treatment in this form emphasizes the depth of response. If we then look at the rate of MRD negativity using high quality assays, then a quarter of the patients have undetectable polymerase chain reaction during the consolidation cycles, and approximately 80% achieves something that we call a major molecular response,” he said.
Dr. Ottmann did not provide updated safety data, but at the time of the data cutoff, there had been 34 serious adverse events reported, 11 of which occurred during induction, 16 during consolidation, 6 during maintenance, and 1 following discontinuation. The most-common adverse events were infections and neutropenic fevers. Single serious adverse events included metabolic, cardiovascular, neurologic, renal, and hepatic events.
The trial is expected to be completed in the next few months.
SAN FRANCISCO– The study was small but encouraging: Among 47 older patients with newly diagnosed acute lymphoblastic leukemia positive for the Philadelphia chromosome, 41 had a complete hematologic response to a combination of chemotherapy and the targeted agent nilotinib (Tasigna), report investigators from a European consortium.
“The data I have presented show that the combination of nilotinib with this age-adapted chemotherapy is highly effective. We do have quite a reasonable overall survival estimate at 2 years of just more than 70%,” said Dr. Oliver Ottmann of Goethe University in Frankfurt, on behalf of colleagues in the European Working Group for Adult ALL (EWALL).
The study also shows that although some centers are reluctant to offer allogeneic stem cell transplantation (SCT) to older patients, it is still a viable treatment option in this population, Dr. Ottmann said at a briefing at the annual meeting of the American Society of Hematology.
Although older patients with newly diagnosed Philadelphia-positive (Ph+) ALL have a high complete hematologic response rate (CHR) with imatinib (Gleevec), they generally have a poor prognosis because of a high rate of relapse.
Because nilotinib, a potent inhibitor of the ABL kinase, has good efficacy in the chronic and accelerated phase of Ph+ chronic myeloid leukemia, the EWALL investigators initiated a study to evaluate it in combination with chemotherapy in the front-line setting.
Adults aged 55 years and older with ALL positive for the Philadelphia chromosome and/or BCR-ABL1 fusion who were treatment naive or had not received therapy other than corticosteroids, single-dose vincristine, or three doses of cyclophosphamide were eligible.
Details of the combination regimen are available online.
Briefly, following a prephase with dexamethasone and optional cyclophosphamide, patients receive nilotinib 400 mg twice daily starting with induction and continuously thereafter. During induction, nilotinib is given with intravenous injections of vincristine and dexamethasone for 4 weeks, followed by consolidation with nilotinib, methotrexate, asparaginase and cytarabine. Maintenance consists of nilotinib, 6-mercaptopurine, and methotrexate once weekly for 1 month then every other month, and dexamethasone and vincristine in 2 month intervals up to 24 months.
The data Dr. Ottmann reported come from an interim analysis of the ongoing study. As of August 2014, data on 47 of 56 patients was available for an efficacy analysis, As noted before, the rate of CHR was 87%, occurring in 41 of 47 patients. The treatment evoked a partial response or no response in 2 patients, and there was one death during the induction phase. Additionally, three patients discontinued therapy early and were not included in the assessment, but at least one had a complete response later on, Dr. Ottmann noted.
The median time to a complete response (CR) was 41 days, but CRs occurred as early as 25 days and as late as 62 days after the start of therapy. The remissions at the time of data cutoff appeared to be durable, but follow-up is still early, he said.
Overall survival at a median follow-up for all patients of 8.6 months was 72.7% at 30 months for patients who did not undergo SCT (allowed under the protocol), and 67.1% at 30 months for patients who underwent SCT. This difference was not significant, but only nine patients at the time of data cutoff had undergone transplantation.
“It will be interesting to see how this will proceed if the transplant-free patients will do as well as the others,” Dr. Ottmann said.
An analysis of molecular response by minimal residual disease (MRD) time point showed a significant further increase with the consolidation chemotherapy and kinase inhibitor, emphasizing that “continuing the treatment in this form emphasizes the depth of response. If we then look at the rate of MRD negativity using high quality assays, then a quarter of the patients have undetectable polymerase chain reaction during the consolidation cycles, and approximately 80% achieves something that we call a major molecular response,” he said.
Dr. Ottmann did not provide updated safety data, but at the time of the data cutoff, there had been 34 serious adverse events reported, 11 of which occurred during induction, 16 during consolidation, 6 during maintenance, and 1 following discontinuation. The most-common adverse events were infections and neutropenic fevers. Single serious adverse events included metabolic, cardiovascular, neurologic, renal, and hepatic events.
The trial is expected to be completed in the next few months.
SAN FRANCISCO– The study was small but encouraging: Among 47 older patients with newly diagnosed acute lymphoblastic leukemia positive for the Philadelphia chromosome, 41 had a complete hematologic response to a combination of chemotherapy and the targeted agent nilotinib (Tasigna), report investigators from a European consortium.
“The data I have presented show that the combination of nilotinib with this age-adapted chemotherapy is highly effective. We do have quite a reasonable overall survival estimate at 2 years of just more than 70%,” said Dr. Oliver Ottmann of Goethe University in Frankfurt, on behalf of colleagues in the European Working Group for Adult ALL (EWALL).
The study also shows that although some centers are reluctant to offer allogeneic stem cell transplantation (SCT) to older patients, it is still a viable treatment option in this population, Dr. Ottmann said at a briefing at the annual meeting of the American Society of Hematology.
Although older patients with newly diagnosed Philadelphia-positive (Ph+) ALL have a high complete hematologic response rate (CHR) with imatinib (Gleevec), they generally have a poor prognosis because of a high rate of relapse.
Because nilotinib, a potent inhibitor of the ABL kinase, has good efficacy in the chronic and accelerated phase of Ph+ chronic myeloid leukemia, the EWALL investigators initiated a study to evaluate it in combination with chemotherapy in the front-line setting.
Adults aged 55 years and older with ALL positive for the Philadelphia chromosome and/or BCR-ABL1 fusion who were treatment naive or had not received therapy other than corticosteroids, single-dose vincristine, or three doses of cyclophosphamide were eligible.
Details of the combination regimen are available online.
Briefly, following a prephase with dexamethasone and optional cyclophosphamide, patients receive nilotinib 400 mg twice daily starting with induction and continuously thereafter. During induction, nilotinib is given with intravenous injections of vincristine and dexamethasone for 4 weeks, followed by consolidation with nilotinib, methotrexate, asparaginase and cytarabine. Maintenance consists of nilotinib, 6-mercaptopurine, and methotrexate once weekly for 1 month then every other month, and dexamethasone and vincristine in 2 month intervals up to 24 months.
The data Dr. Ottmann reported come from an interim analysis of the ongoing study. As of August 2014, data on 47 of 56 patients was available for an efficacy analysis, As noted before, the rate of CHR was 87%, occurring in 41 of 47 patients. The treatment evoked a partial response or no response in 2 patients, and there was one death during the induction phase. Additionally, three patients discontinued therapy early and were not included in the assessment, but at least one had a complete response later on, Dr. Ottmann noted.
The median time to a complete response (CR) was 41 days, but CRs occurred as early as 25 days and as late as 62 days after the start of therapy. The remissions at the time of data cutoff appeared to be durable, but follow-up is still early, he said.
Overall survival at a median follow-up for all patients of 8.6 months was 72.7% at 30 months for patients who did not undergo SCT (allowed under the protocol), and 67.1% at 30 months for patients who underwent SCT. This difference was not significant, but only nine patients at the time of data cutoff had undergone transplantation.
“It will be interesting to see how this will proceed if the transplant-free patients will do as well as the others,” Dr. Ottmann said.
An analysis of molecular response by minimal residual disease (MRD) time point showed a significant further increase with the consolidation chemotherapy and kinase inhibitor, emphasizing that “continuing the treatment in this form emphasizes the depth of response. If we then look at the rate of MRD negativity using high quality assays, then a quarter of the patients have undetectable polymerase chain reaction during the consolidation cycles, and approximately 80% achieves something that we call a major molecular response,” he said.
Dr. Ottmann did not provide updated safety data, but at the time of the data cutoff, there had been 34 serious adverse events reported, 11 of which occurred during induction, 16 during consolidation, 6 during maintenance, and 1 following discontinuation. The most-common adverse events were infections and neutropenic fevers. Single serious adverse events included metabolic, cardiovascular, neurologic, renal, and hepatic events.
The trial is expected to be completed in the next few months.
Key clinical point: Combining a kinase inhibitor with an intensive chemotherapy regimen produces a high complete response rate in patients age 55 and older who have Ph+All.
Major finding: The complete hematologic response rate for the combination of chemotherapy and nilotinib was 87%.
Data source: Investigator initiated study in 56 patients with acute lymphoblastic leukemia positive for the Philadelphia chromosome or BCR/ABL fusion.
Disclosures: The study was sponsored by participating institution. Dr. Ottmann disclosed consultancy, honoraria, and research funding from Novartis, maker of nilotinib.
Young adults with ALL have better survival with pediatric regimens
SAN FRANCISCO– Teens and young adults don’t like being treated like children, but they should make an exception when it comes to acute lymphoblastic leukemia therapy, because pediatric ALL regimens are associated with significantly better event-free and overall survival among patients with ALL from the ages of 16 to 40 years.
Those findings come from a clinical trial 14 years in the making, in which 296 adolescents and young adults (AYA) with ALL were treated with an intensive pediatric chemotherapy combination regiment rather than a less-intensive adult regimen. At 2-year follow-up, the rate of overall survival (OS) was 78%, with the median overall survival not yet reached, and the event-free survival (EFS) rate was 66%, reported Dr. Wendy Stock from the University of Chicago Medical Center.
In contrast, EFS rates among AYA treated with adult regimens have historically ranged from 35-40%, Dr. Stock said at a press briefing at the annual meeting of the American Society of Hematology.
“These data really started 14 years ago at this ASH meeting when we presented data showing that young adults ages 16 to 20 who were treated on adult cooperative group studies in the United States fared much worse than the same age group who were treated on pediatric studies,” she said.
In 2008, Dr. Stock and her colleagues published a study (Blood 2008;112:1646-54) showing that AYAs treated under Children’s Cancer Group (CCG) protocols had an overall survival rate at 7 years of 67% and an EFS rate of 63%. In contrast, AYAs treated under Cancer and Leukemia Group B (CALGB) protocols had an OS of 46% and EFS of only 34%. The risk for worse outcomes was approximately twofold among adolescents treated with adult regimens, compared with those treated with pediatric regimens. The findings were similar in studies from France, the United Kingdom, and the Netherlands, Dr. Stock noted.
The investigators determined at that time that, under the CCG regimens, the teen and young adult patients received earlier and more intensive central nervous system prophylaxis and higher doses of nonmyelosuppressive drugs, especially glucocorticoids, vincristine, and pegylated asparignase, while those on the CALGB regimens received higher doses of myelosuppressive agents such as anthracyclines.
Because their original findings came from a retrospective study, the investigators decided to launch a prospective study,US Intergroup trial C10403, designed to evaluate outcomes among patients with ALL from the ages of 16-40 years when they were treated with a pediatric regimen by adult hematologists/oncologists in the cooperative group setting.
A total of 296 eligible patients with a median age of 25 years were enrolled. The patients had newly diagnosed ALL of T-cell or B-cell lineage; patients with Burkitt’s type ALL or ALL positive for the Philadelphia chromosome were excluded. The patients were treated with a regimen identical to the Capizzi methotrexate arm of the Children’s Oncology Group AALL0232 study. The regimen consisted of four intensive courses: remission induction, remission consolidation, interim maintenance and delayed intensification, and prolonged maintenance therapy. Patients who had an M2 marrow response after remission induction (more than 5% but less than 25% lymphoblasts) received an extended remission induction course of therapy.
As noted before, the EFS rate was 66% and the median EFS duration was 59 months. The 2-year overall survival rate was 79%. EFS rates were similar between patients with B-cell lineage (65%) and T-cell lineage (68%) ALL, and there were no significant differences in EFS or OS by sex or by age.
There were five (2%) treatment-related deaths during protocol therapy, including two cases of liver failure, both occurring during induction; two infections (one in the induction phase and one in the consolidation phase); and one ventricular arrhythmia (during induction). Treatment toxicities in general were similar to those seen in the standard therapy of the COG AALL0232 trial, although patients in the current study had an increase in risk for thrombosis and early hyperbilirubinemia.
“Our outcomes are similar to other prospective international studies which apply pediatric regimens to the young adult population of acute lymphoblastic leukemia,” Dr. Stock said.
In analyses of biological factors that affect outcome, the investigators found that white blood cell counts above 30,000/uL were associated with worse EFS and OS, and that the presence of a BCR-ABL1-like signature and overexpression of the gene CRLF2 were common and associated with significantly worse survival.
The investigators plan to use the study as a basis for future studies incorporating target antibodies and kinase inhibitors in an attempt to improve survival further by eradicating minimal residual disease, Dr. Stock said.
SAN FRANCISCO– Teens and young adults don’t like being treated like children, but they should make an exception when it comes to acute lymphoblastic leukemia therapy, because pediatric ALL regimens are associated with significantly better event-free and overall survival among patients with ALL from the ages of 16 to 40 years.
Those findings come from a clinical trial 14 years in the making, in which 296 adolescents and young adults (AYA) with ALL were treated with an intensive pediatric chemotherapy combination regiment rather than a less-intensive adult regimen. At 2-year follow-up, the rate of overall survival (OS) was 78%, with the median overall survival not yet reached, and the event-free survival (EFS) rate was 66%, reported Dr. Wendy Stock from the University of Chicago Medical Center.
In contrast, EFS rates among AYA treated with adult regimens have historically ranged from 35-40%, Dr. Stock said at a press briefing at the annual meeting of the American Society of Hematology.
“These data really started 14 years ago at this ASH meeting when we presented data showing that young adults ages 16 to 20 who were treated on adult cooperative group studies in the United States fared much worse than the same age group who were treated on pediatric studies,” she said.
In 2008, Dr. Stock and her colleagues published a study (Blood 2008;112:1646-54) showing that AYAs treated under Children’s Cancer Group (CCG) protocols had an overall survival rate at 7 years of 67% and an EFS rate of 63%. In contrast, AYAs treated under Cancer and Leukemia Group B (CALGB) protocols had an OS of 46% and EFS of only 34%. The risk for worse outcomes was approximately twofold among adolescents treated with adult regimens, compared with those treated with pediatric regimens. The findings were similar in studies from France, the United Kingdom, and the Netherlands, Dr. Stock noted.
The investigators determined at that time that, under the CCG regimens, the teen and young adult patients received earlier and more intensive central nervous system prophylaxis and higher doses of nonmyelosuppressive drugs, especially glucocorticoids, vincristine, and pegylated asparignase, while those on the CALGB regimens received higher doses of myelosuppressive agents such as anthracyclines.
Because their original findings came from a retrospective study, the investigators decided to launch a prospective study,US Intergroup trial C10403, designed to evaluate outcomes among patients with ALL from the ages of 16-40 years when they were treated with a pediatric regimen by adult hematologists/oncologists in the cooperative group setting.
A total of 296 eligible patients with a median age of 25 years were enrolled. The patients had newly diagnosed ALL of T-cell or B-cell lineage; patients with Burkitt’s type ALL or ALL positive for the Philadelphia chromosome were excluded. The patients were treated with a regimen identical to the Capizzi methotrexate arm of the Children’s Oncology Group AALL0232 study. The regimen consisted of four intensive courses: remission induction, remission consolidation, interim maintenance and delayed intensification, and prolonged maintenance therapy. Patients who had an M2 marrow response after remission induction (more than 5% but less than 25% lymphoblasts) received an extended remission induction course of therapy.
As noted before, the EFS rate was 66% and the median EFS duration was 59 months. The 2-year overall survival rate was 79%. EFS rates were similar between patients with B-cell lineage (65%) and T-cell lineage (68%) ALL, and there were no significant differences in EFS or OS by sex or by age.
There were five (2%) treatment-related deaths during protocol therapy, including two cases of liver failure, both occurring during induction; two infections (one in the induction phase and one in the consolidation phase); and one ventricular arrhythmia (during induction). Treatment toxicities in general were similar to those seen in the standard therapy of the COG AALL0232 trial, although patients in the current study had an increase in risk for thrombosis and early hyperbilirubinemia.
“Our outcomes are similar to other prospective international studies which apply pediatric regimens to the young adult population of acute lymphoblastic leukemia,” Dr. Stock said.
In analyses of biological factors that affect outcome, the investigators found that white blood cell counts above 30,000/uL were associated with worse EFS and OS, and that the presence of a BCR-ABL1-like signature and overexpression of the gene CRLF2 were common and associated with significantly worse survival.
The investigators plan to use the study as a basis for future studies incorporating target antibodies and kinase inhibitors in an attempt to improve survival further by eradicating minimal residual disease, Dr. Stock said.
SAN FRANCISCO– Teens and young adults don’t like being treated like children, but they should make an exception when it comes to acute lymphoblastic leukemia therapy, because pediatric ALL regimens are associated with significantly better event-free and overall survival among patients with ALL from the ages of 16 to 40 years.
Those findings come from a clinical trial 14 years in the making, in which 296 adolescents and young adults (AYA) with ALL were treated with an intensive pediatric chemotherapy combination regiment rather than a less-intensive adult regimen. At 2-year follow-up, the rate of overall survival (OS) was 78%, with the median overall survival not yet reached, and the event-free survival (EFS) rate was 66%, reported Dr. Wendy Stock from the University of Chicago Medical Center.
In contrast, EFS rates among AYA treated with adult regimens have historically ranged from 35-40%, Dr. Stock said at a press briefing at the annual meeting of the American Society of Hematology.
“These data really started 14 years ago at this ASH meeting when we presented data showing that young adults ages 16 to 20 who were treated on adult cooperative group studies in the United States fared much worse than the same age group who were treated on pediatric studies,” she said.
In 2008, Dr. Stock and her colleagues published a study (Blood 2008;112:1646-54) showing that AYAs treated under Children’s Cancer Group (CCG) protocols had an overall survival rate at 7 years of 67% and an EFS rate of 63%. In contrast, AYAs treated under Cancer and Leukemia Group B (CALGB) protocols had an OS of 46% and EFS of only 34%. The risk for worse outcomes was approximately twofold among adolescents treated with adult regimens, compared with those treated with pediatric regimens. The findings were similar in studies from France, the United Kingdom, and the Netherlands, Dr. Stock noted.
The investigators determined at that time that, under the CCG regimens, the teen and young adult patients received earlier and more intensive central nervous system prophylaxis and higher doses of nonmyelosuppressive drugs, especially glucocorticoids, vincristine, and pegylated asparignase, while those on the CALGB regimens received higher doses of myelosuppressive agents such as anthracyclines.
Because their original findings came from a retrospective study, the investigators decided to launch a prospective study,US Intergroup trial C10403, designed to evaluate outcomes among patients with ALL from the ages of 16-40 years when they were treated with a pediatric regimen by adult hematologists/oncologists in the cooperative group setting.
A total of 296 eligible patients with a median age of 25 years were enrolled. The patients had newly diagnosed ALL of T-cell or B-cell lineage; patients with Burkitt’s type ALL or ALL positive for the Philadelphia chromosome were excluded. The patients were treated with a regimen identical to the Capizzi methotrexate arm of the Children’s Oncology Group AALL0232 study. The regimen consisted of four intensive courses: remission induction, remission consolidation, interim maintenance and delayed intensification, and prolonged maintenance therapy. Patients who had an M2 marrow response after remission induction (more than 5% but less than 25% lymphoblasts) received an extended remission induction course of therapy.
As noted before, the EFS rate was 66% and the median EFS duration was 59 months. The 2-year overall survival rate was 79%. EFS rates were similar between patients with B-cell lineage (65%) and T-cell lineage (68%) ALL, and there were no significant differences in EFS or OS by sex or by age.
There were five (2%) treatment-related deaths during protocol therapy, including two cases of liver failure, both occurring during induction; two infections (one in the induction phase and one in the consolidation phase); and one ventricular arrhythmia (during induction). Treatment toxicities in general were similar to those seen in the standard therapy of the COG AALL0232 trial, although patients in the current study had an increase in risk for thrombosis and early hyperbilirubinemia.
“Our outcomes are similar to other prospective international studies which apply pediatric regimens to the young adult population of acute lymphoblastic leukemia,” Dr. Stock said.
In analyses of biological factors that affect outcome, the investigators found that white blood cell counts above 30,000/uL were associated with worse EFS and OS, and that the presence of a BCR-ABL1-like signature and overexpression of the gene CRLF2 were common and associated with significantly worse survival.
The investigators plan to use the study as a basis for future studies incorporating target antibodies and kinase inhibitors in an attempt to improve survival further by eradicating minimal residual disease, Dr. Stock said.
Key clinical point: Adolescents and young adults with acute lymphoblastic leukemia should be treated with a pediatric rather than an adult ALL regimen.
Major finding: Overall survival was 78% and event-free survival rate was 66%, compared with 46% and 34% for young adults treated with adult-style regimens in the past.
Data source: Prospective trial with 296 patients from the ages of 16-40 years with ALL.
Disclosures: The study is supported by the National Institutes of Health. Dr. Stock disclosed serving as an advisor and receiving research funding from Sigma-Tau Pharmaceuticals.
FDA approves first anti-CD19 drug

Credit: NIAID
The US Food and Drug Administration (FDA) has granted accelerated approval for blinatumomab (Blincyto) to treat adults with relapsed or refractory Philadelphia chromosome-negative precursor B-cell acute lymphoblastic leukemia (ALL).
Blinatumomab is a bispecific T-cell engager (BiTE®) antibody construct designed to direct the body’s T cells against target cells expressing CD19, a protein found on the surface of B cells.
Blinatumomab is the first anti-CD19 drug to receive FDA approval. It will be available as a 35 mcg single-use vial.
“Immunotherapies, especially Blincyto with its unique mechanism of action, are particularly promising for patients with leukemia,” said Richard Pazdur, MD, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research.
“Recognizing the potential of this novel therapy, the FDA worked proactively with the sponsor under our breakthrough therapy designation program to facilitate the approval of this novel agent.”
Approval details
The FDA granted blinatumomab accelerated approval based on clinical data showing the drug has an effect on a surrogate endpoint reasonably likely to predict clinical benefit to patients.
Before granting the drug full approval, the FDA is requiring that blinatumomab’s manufacturer, Amgen, conduct a study to verify that the drug improves survival in patients with relapsed or refractory Philadelphia-negative precursor B-cell ALL.
Blinatumomab was approved with a boxed warning detailing the risk of cytokine release syndrome and neurological toxicities. The FDA also approved the drug with a Risk Evaluation and Mitigation Strategy, which consists of a communication plan to inform healthcare providers about the serious risks and the potential for preparation and administration errors.
Blinatumomab was approved more than 5 months ahead of the date the FDA was scheduled to complete review of the drug’s application (May 19, 2015).
The agency had granted blinatumomab breakthrough therapy designation, priority review, and orphan product designation because Amgen demonstrated through preliminary clinical evidence that the drug may offer a substantial improvement over available therapies.
That evidence came in the form of a phase 2 study, the results of which were presented at the 19th Congress of the European Hematology Association (EHA) in June.
Trial results
Researchers evaluated blinatumomab monotherapy in 189 patients with relapsed or refractory B-cell ALL and a median age of 39 (range, 18-79). The patients received blinatumomab by continuous intravenous infusion—4 weeks on and 2 weeks off—for up to 5 cycles.
Thirty-three percent of patients achieved a complete remission, and 9% achieved a complete remission with partial hematologic recovery. Seventy-one percent of these patients were negative for minimal residual disease.
The median relapse-free survival was 5.9 months.
Major toxicities were related to cytokine release syndrome, but cytopenias and central nervous system events were also common. The most frequent adverse events were pyrexia (59%), headache (35%), and febrile neutropenia (29%).
The most frequent grade 3 or higher adverse events were febrile neutropenia (26%), anemia (15%), and neutropenia (15%). Two percent of patients had grade 3 or higher cytokine release syndrome.
The most common grade 3 or higher nervous system events were headache (4%), encephalopathy (3%), and ataxia (2%). Three patients (2%) had grade 5 events considered treatment-related—2 with sepsis and 1 with Candida infection. ![]()

Credit: NIAID
The US Food and Drug Administration (FDA) has granted accelerated approval for blinatumomab (Blincyto) to treat adults with relapsed or refractory Philadelphia chromosome-negative precursor B-cell acute lymphoblastic leukemia (ALL).
Blinatumomab is a bispecific T-cell engager (BiTE®) antibody construct designed to direct the body’s T cells against target cells expressing CD19, a protein found on the surface of B cells.
Blinatumomab is the first anti-CD19 drug to receive FDA approval. It will be available as a 35 mcg single-use vial.
“Immunotherapies, especially Blincyto with its unique mechanism of action, are particularly promising for patients with leukemia,” said Richard Pazdur, MD, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research.
“Recognizing the potential of this novel therapy, the FDA worked proactively with the sponsor under our breakthrough therapy designation program to facilitate the approval of this novel agent.”
Approval details
The FDA granted blinatumomab accelerated approval based on clinical data showing the drug has an effect on a surrogate endpoint reasonably likely to predict clinical benefit to patients.
Before granting the drug full approval, the FDA is requiring that blinatumomab’s manufacturer, Amgen, conduct a study to verify that the drug improves survival in patients with relapsed or refractory Philadelphia-negative precursor B-cell ALL.
Blinatumomab was approved with a boxed warning detailing the risk of cytokine release syndrome and neurological toxicities. The FDA also approved the drug with a Risk Evaluation and Mitigation Strategy, which consists of a communication plan to inform healthcare providers about the serious risks and the potential for preparation and administration errors.
Blinatumomab was approved more than 5 months ahead of the date the FDA was scheduled to complete review of the drug’s application (May 19, 2015).
The agency had granted blinatumomab breakthrough therapy designation, priority review, and orphan product designation because Amgen demonstrated through preliminary clinical evidence that the drug may offer a substantial improvement over available therapies.
That evidence came in the form of a phase 2 study, the results of which were presented at the 19th Congress of the European Hematology Association (EHA) in June.
Trial results
Researchers evaluated blinatumomab monotherapy in 189 patients with relapsed or refractory B-cell ALL and a median age of 39 (range, 18-79). The patients received blinatumomab by continuous intravenous infusion—4 weeks on and 2 weeks off—for up to 5 cycles.
Thirty-three percent of patients achieved a complete remission, and 9% achieved a complete remission with partial hematologic recovery. Seventy-one percent of these patients were negative for minimal residual disease.
The median relapse-free survival was 5.9 months.
Major toxicities were related to cytokine release syndrome, but cytopenias and central nervous system events were also common. The most frequent adverse events were pyrexia (59%), headache (35%), and febrile neutropenia (29%).
The most frequent grade 3 or higher adverse events were febrile neutropenia (26%), anemia (15%), and neutropenia (15%). Two percent of patients had grade 3 or higher cytokine release syndrome.
The most common grade 3 or higher nervous system events were headache (4%), encephalopathy (3%), and ataxia (2%). Three patients (2%) had grade 5 events considered treatment-related—2 with sepsis and 1 with Candida infection. ![]()

Credit: NIAID
The US Food and Drug Administration (FDA) has granted accelerated approval for blinatumomab (Blincyto) to treat adults with relapsed or refractory Philadelphia chromosome-negative precursor B-cell acute lymphoblastic leukemia (ALL).
Blinatumomab is a bispecific T-cell engager (BiTE®) antibody construct designed to direct the body’s T cells against target cells expressing CD19, a protein found on the surface of B cells.
Blinatumomab is the first anti-CD19 drug to receive FDA approval. It will be available as a 35 mcg single-use vial.
“Immunotherapies, especially Blincyto with its unique mechanism of action, are particularly promising for patients with leukemia,” said Richard Pazdur, MD, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research.
“Recognizing the potential of this novel therapy, the FDA worked proactively with the sponsor under our breakthrough therapy designation program to facilitate the approval of this novel agent.”
Approval details
The FDA granted blinatumomab accelerated approval based on clinical data showing the drug has an effect on a surrogate endpoint reasonably likely to predict clinical benefit to patients.
Before granting the drug full approval, the FDA is requiring that blinatumomab’s manufacturer, Amgen, conduct a study to verify that the drug improves survival in patients with relapsed or refractory Philadelphia-negative precursor B-cell ALL.
Blinatumomab was approved with a boxed warning detailing the risk of cytokine release syndrome and neurological toxicities. The FDA also approved the drug with a Risk Evaluation and Mitigation Strategy, which consists of a communication plan to inform healthcare providers about the serious risks and the potential for preparation and administration errors.
Blinatumomab was approved more than 5 months ahead of the date the FDA was scheduled to complete review of the drug’s application (May 19, 2015).
The agency had granted blinatumomab breakthrough therapy designation, priority review, and orphan product designation because Amgen demonstrated through preliminary clinical evidence that the drug may offer a substantial improvement over available therapies.
That evidence came in the form of a phase 2 study, the results of which were presented at the 19th Congress of the European Hematology Association (EHA) in June.
Trial results
Researchers evaluated blinatumomab monotherapy in 189 patients with relapsed or refractory B-cell ALL and a median age of 39 (range, 18-79). The patients received blinatumomab by continuous intravenous infusion—4 weeks on and 2 weeks off—for up to 5 cycles.
Thirty-three percent of patients achieved a complete remission, and 9% achieved a complete remission with partial hematologic recovery. Seventy-one percent of these patients were negative for minimal residual disease.
The median relapse-free survival was 5.9 months.
Major toxicities were related to cytokine release syndrome, but cytopenias and central nervous system events were also common. The most frequent adverse events were pyrexia (59%), headache (35%), and febrile neutropenia (29%).
The most frequent grade 3 or higher adverse events were febrile neutropenia (26%), anemia (15%), and neutropenia (15%). Two percent of patients had grade 3 or higher cytokine release syndrome.
The most common grade 3 or higher nervous system events were headache (4%), encephalopathy (3%), and ataxia (2%). Three patients (2%) had grade 5 events considered treatment-related—2 with sepsis and 1 with Candida infection. ![]()
Leukemia survival rates vary widely across the globe

Credit: Bill Branson
A large, international study has revealed sizable differences in cancer survival rates between countries.
In particular, investigators observed a wide gap in survival for children with acute lymphoblastic leukemia (ALL).
The most recent 5-year survival rate was less than 60% in several countries—and as low as 16% in one nation—but more than 90% in other countries.
This indicates major deficiencies in managing this largely curable disease, according to researchers.
Results from this study, known as CONCORD-2, appear in The Lancet alongside a linked comment article.
Claudia Allemani, PhD, of the London School of Hygiene & Tropical Medicine in the UK, and her colleagues analyzed individual patient data from 279 cancer registries in 67 countries.
The team reported 5-year survival estimates for 25.7 million cancer patients diagnosed with 1 of 10 common cancers—stomach, colon, rectum, liver, lung, breast (women), cervix, ovary, prostate, and leukemia (n=873,588)—from 1995 through 2009.
The investigators also reported survival estimates for 74,343 children diagnosed with ALL during that period.
Even after they adjusted for differences between countries and regions in the risk of death from other causes by age, sex, and race, and over time, the researchers found very large variations between countries in survival for specific cancers.
Adult leukemia
For adults diagnosed with leukemia from 2005 through 2009, the 5-year net survival was 50% to 60% in 21 countries in North America, west Asia, Europe, and Oceania. Survival rates were generally much lower in the 15 participating Asian countries than in other parts of the world.
Survival rates tended to be low in east Asia—ranging from 19% in Japan to 23% in South Korea and Taiwan—but higher in west Asia—ranging from 33% in Turkey to 53% in Qatar. And results were mixed in other Asian countries—ranging from 7% in Jordan to 40% in Indonesia.
ALL in children
For children diagnosed with ALL from 2005 through 2009, 5-year survival was 90% or higher in Austria, Belgium, Canada, Germany, and Norway. It was 80% to 89% in 21 countries on various continents.
However, 5-year survival was lower than 60% in several countries and the lowest—16%—in Jordan.
The range of survival estimates for childhood ALL in Central/South America and Asia was much lower than the range in North America, Europe, and Oceania.
Solid tumor malignancies
The study also showed that liver and lung cancer have the worst prognosis among the 10 cancers examined, with 5-year survival of less than 20% in both developed and developing countries. The researchers said this suggests most patients still visit their doctors too late for treatment to be effective.
Five-year survival from breast and colorectal cancers increased in most developed countries and in South America (Brazil, Colombia, and Ecuador) from the beginning of the study period to the end. These trends likely reflect earlier diagnosis and the availability of better treatment options, according to the investigators.
Stomach cancer survival was higher in Southeast Asia than in other regions. This is likely a result of intensive diagnostic activity, early stage at diagnosis, and radical surgery, the researchers said. And this suggests important lessons could be learned from these countries about diagnosis and treatment.
Cervical and ovarian cancers showed particularly wide differences in survival, and overall improvements during the study period were slight.
“Our findings show that, in some countries, cancer is far more lethal than in others,” Dr Allemani said. “In the 21st century, there should not be such a dramatic gulf in survival. The majority of the variability in survival is probably due to factors that can be changed, such as the availability and quality of diagnostic and treatment services.”
“The findings can be used to evaluate the extent to which investment in healthcare systems is improving their effectiveness. We expect them to act as a stimulus for politicians to improve health policy and invest in healthcare.” ![]()

Credit: Bill Branson
A large, international study has revealed sizable differences in cancer survival rates between countries.
In particular, investigators observed a wide gap in survival for children with acute lymphoblastic leukemia (ALL).
The most recent 5-year survival rate was less than 60% in several countries—and as low as 16% in one nation—but more than 90% in other countries.
This indicates major deficiencies in managing this largely curable disease, according to researchers.
Results from this study, known as CONCORD-2, appear in The Lancet alongside a linked comment article.
Claudia Allemani, PhD, of the London School of Hygiene & Tropical Medicine in the UK, and her colleagues analyzed individual patient data from 279 cancer registries in 67 countries.
The team reported 5-year survival estimates for 25.7 million cancer patients diagnosed with 1 of 10 common cancers—stomach, colon, rectum, liver, lung, breast (women), cervix, ovary, prostate, and leukemia (n=873,588)—from 1995 through 2009.
The investigators also reported survival estimates for 74,343 children diagnosed with ALL during that period.
Even after they adjusted for differences between countries and regions in the risk of death from other causes by age, sex, and race, and over time, the researchers found very large variations between countries in survival for specific cancers.
Adult leukemia
For adults diagnosed with leukemia from 2005 through 2009, the 5-year net survival was 50% to 60% in 21 countries in North America, west Asia, Europe, and Oceania. Survival rates were generally much lower in the 15 participating Asian countries than in other parts of the world.
Survival rates tended to be low in east Asia—ranging from 19% in Japan to 23% in South Korea and Taiwan—but higher in west Asia—ranging from 33% in Turkey to 53% in Qatar. And results were mixed in other Asian countries—ranging from 7% in Jordan to 40% in Indonesia.
ALL in children
For children diagnosed with ALL from 2005 through 2009, 5-year survival was 90% or higher in Austria, Belgium, Canada, Germany, and Norway. It was 80% to 89% in 21 countries on various continents.
However, 5-year survival was lower than 60% in several countries and the lowest—16%—in Jordan.
The range of survival estimates for childhood ALL in Central/South America and Asia was much lower than the range in North America, Europe, and Oceania.
Solid tumor malignancies
The study also showed that liver and lung cancer have the worst prognosis among the 10 cancers examined, with 5-year survival of less than 20% in both developed and developing countries. The researchers said this suggests most patients still visit their doctors too late for treatment to be effective.
Five-year survival from breast and colorectal cancers increased in most developed countries and in South America (Brazil, Colombia, and Ecuador) from the beginning of the study period to the end. These trends likely reflect earlier diagnosis and the availability of better treatment options, according to the investigators.
Stomach cancer survival was higher in Southeast Asia than in other regions. This is likely a result of intensive diagnostic activity, early stage at diagnosis, and radical surgery, the researchers said. And this suggests important lessons could be learned from these countries about diagnosis and treatment.
Cervical and ovarian cancers showed particularly wide differences in survival, and overall improvements during the study period were slight.
“Our findings show that, in some countries, cancer is far more lethal than in others,” Dr Allemani said. “In the 21st century, there should not be such a dramatic gulf in survival. The majority of the variability in survival is probably due to factors that can be changed, such as the availability and quality of diagnostic and treatment services.”
“The findings can be used to evaluate the extent to which investment in healthcare systems is improving their effectiveness. We expect them to act as a stimulus for politicians to improve health policy and invest in healthcare.” ![]()

Credit: Bill Branson
A large, international study has revealed sizable differences in cancer survival rates between countries.
In particular, investigators observed a wide gap in survival for children with acute lymphoblastic leukemia (ALL).
The most recent 5-year survival rate was less than 60% in several countries—and as low as 16% in one nation—but more than 90% in other countries.
This indicates major deficiencies in managing this largely curable disease, according to researchers.
Results from this study, known as CONCORD-2, appear in The Lancet alongside a linked comment article.
Claudia Allemani, PhD, of the London School of Hygiene & Tropical Medicine in the UK, and her colleagues analyzed individual patient data from 279 cancer registries in 67 countries.
The team reported 5-year survival estimates for 25.7 million cancer patients diagnosed with 1 of 10 common cancers—stomach, colon, rectum, liver, lung, breast (women), cervix, ovary, prostate, and leukemia (n=873,588)—from 1995 through 2009.
The investigators also reported survival estimates for 74,343 children diagnosed with ALL during that period.
Even after they adjusted for differences between countries and regions in the risk of death from other causes by age, sex, and race, and over time, the researchers found very large variations between countries in survival for specific cancers.
Adult leukemia
For adults diagnosed with leukemia from 2005 through 2009, the 5-year net survival was 50% to 60% in 21 countries in North America, west Asia, Europe, and Oceania. Survival rates were generally much lower in the 15 participating Asian countries than in other parts of the world.
Survival rates tended to be low in east Asia—ranging from 19% in Japan to 23% in South Korea and Taiwan—but higher in west Asia—ranging from 33% in Turkey to 53% in Qatar. And results were mixed in other Asian countries—ranging from 7% in Jordan to 40% in Indonesia.
ALL in children
For children diagnosed with ALL from 2005 through 2009, 5-year survival was 90% or higher in Austria, Belgium, Canada, Germany, and Norway. It was 80% to 89% in 21 countries on various continents.
However, 5-year survival was lower than 60% in several countries and the lowest—16%—in Jordan.
The range of survival estimates for childhood ALL in Central/South America and Asia was much lower than the range in North America, Europe, and Oceania.
Solid tumor malignancies
The study also showed that liver and lung cancer have the worst prognosis among the 10 cancers examined, with 5-year survival of less than 20% in both developed and developing countries. The researchers said this suggests most patients still visit their doctors too late for treatment to be effective.
Five-year survival from breast and colorectal cancers increased in most developed countries and in South America (Brazil, Colombia, and Ecuador) from the beginning of the study period to the end. These trends likely reflect earlier diagnosis and the availability of better treatment options, according to the investigators.
Stomach cancer survival was higher in Southeast Asia than in other regions. This is likely a result of intensive diagnostic activity, early stage at diagnosis, and radical surgery, the researchers said. And this suggests important lessons could be learned from these countries about diagnosis and treatment.
Cervical and ovarian cancers showed particularly wide differences in survival, and overall improvements during the study period were slight.
“Our findings show that, in some countries, cancer is far more lethal than in others,” Dr Allemani said. “In the 21st century, there should not be such a dramatic gulf in survival. The majority of the variability in survival is probably due to factors that can be changed, such as the availability and quality of diagnostic and treatment services.”
“The findings can be used to evaluate the extent to which investment in healthcare systems is improving their effectiveness. We expect them to act as a stimulus for politicians to improve health policy and invest in healthcare.” ![]()
CAR T-cell therapy gets breakthrough designation

Credit: Charles Haymond
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for JCAR015, a chimeric antigen receptor (CAR) T-cell therapy, to treat patients with relapsed or refractory B-cell acute lymphoblastic leukemia (ALL).
Breakthrough designation is designed to help accelerate the development and review of new drugs for serious or life-threatening conditions.
The designation comes with potential benefits, including intensive FDA guidance and eligibility for priority review. It is granted to applicants when preliminary clinical evidence indicates the drug may demonstrate substantial improvement over existing therapies on one or more clinical endpoints.
The FDA recently granted JCAR015 orphan designation to treat ALL as well.
JCAR015 consists of autologous T cells expressing a CD19-specific, CD28/CD3z CAR. The treatment has shown promise in an ongoing phase 1 trial of patients with B-cell ALL.
Initial results from this study were published in Science Translational Medicine last year and in February. Updated results were presented at the AACR Annual Meeting in April.
At that point, the researchers had enrolled 22 adult patients with relapsed or refractory B-ALL who were minimal residual disease-positive or were in first complete remission at enrollment. Patients in complete remission were monitored and only received JCAR015 if they relapsed.
The remaining patients received re-induction chemotherapy (physician’s choice), followed by an infusion of JCAR015. After treatment, patients could receive allogeneic transplant, a different salvage therapy, or monitoring.
Eighty-two percent of patients achieved a complete response to JCAR015. The average time to complete response was about 24.5 days.
Twelve of the responders were eligible for transplant. Of the 8 patients who ultimately underwent transplant and survived, 1 relapsed, but the rest remained in remission.
Ten patients had died at the time of the AACR presentation. Six deaths were a result of disease relapse or progression, and 2 patients died of complications from stem cell transplant.
The 2 remaining deaths prompted a temporary suspension of enrollment in this trial. Those deaths were related to complications from cytokine release syndrome. One patient died of cardiovascular disease, and the other died following persistent seizure activity.
So researchers at the Memorial Sloan-Kettering Cancer Center, where the trial is being conducted, reviewed these cases. The results prompted them to amend trial enrollment criteria and dosing recommendations. Now, patients with cardiac disease are ineligible to receive JCAR015.
And the T-cell dose a patient receives will depend on the extent of his or her disease. The hope is that this will reduce the risk of cytokine release syndrome and any resulting seizures. The researchers also noted that the monoclonal antibody tocilizumab has proven effective in treating cytokine release syndrome.
In addition to this trial, JCAR015 is under investigation at Memorial Sloan-Kettering Cancer Center in a phase 1 trial of patients with relapsed and refractory non-Hodgkin lymphoma.
Two other CAR T-cell product candidates under development by the same company, Juno Therapeutics, Inc., are being tested in clinical trials as well. JCAR017 is under investigation at Seattle Children’s Hospital for relapsed/refractory CD19-positive pediatric leukemia.
JCAR014 is being tested for refractory chronic lymphocytic leukemia, non-Hodgkin lymphoma, and ALL at the Fred Hutchinson Cancer Research Center. Data on these programs are set to be presented at the 56th ASH Annual Meeting next week in San Francisco. ![]()

Credit: Charles Haymond
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for JCAR015, a chimeric antigen receptor (CAR) T-cell therapy, to treat patients with relapsed or refractory B-cell acute lymphoblastic leukemia (ALL).
Breakthrough designation is designed to help accelerate the development and review of new drugs for serious or life-threatening conditions.
The designation comes with potential benefits, including intensive FDA guidance and eligibility for priority review. It is granted to applicants when preliminary clinical evidence indicates the drug may demonstrate substantial improvement over existing therapies on one or more clinical endpoints.
The FDA recently granted JCAR015 orphan designation to treat ALL as well.
JCAR015 consists of autologous T cells expressing a CD19-specific, CD28/CD3z CAR. The treatment has shown promise in an ongoing phase 1 trial of patients with B-cell ALL.
Initial results from this study were published in Science Translational Medicine last year and in February. Updated results were presented at the AACR Annual Meeting in April.
At that point, the researchers had enrolled 22 adult patients with relapsed or refractory B-ALL who were minimal residual disease-positive or were in first complete remission at enrollment. Patients in complete remission were monitored and only received JCAR015 if they relapsed.
The remaining patients received re-induction chemotherapy (physician’s choice), followed by an infusion of JCAR015. After treatment, patients could receive allogeneic transplant, a different salvage therapy, or monitoring.
Eighty-two percent of patients achieved a complete response to JCAR015. The average time to complete response was about 24.5 days.
Twelve of the responders were eligible for transplant. Of the 8 patients who ultimately underwent transplant and survived, 1 relapsed, but the rest remained in remission.
Ten patients had died at the time of the AACR presentation. Six deaths were a result of disease relapse or progression, and 2 patients died of complications from stem cell transplant.
The 2 remaining deaths prompted a temporary suspension of enrollment in this trial. Those deaths were related to complications from cytokine release syndrome. One patient died of cardiovascular disease, and the other died following persistent seizure activity.
So researchers at the Memorial Sloan-Kettering Cancer Center, where the trial is being conducted, reviewed these cases. The results prompted them to amend trial enrollment criteria and dosing recommendations. Now, patients with cardiac disease are ineligible to receive JCAR015.
And the T-cell dose a patient receives will depend on the extent of his or her disease. The hope is that this will reduce the risk of cytokine release syndrome and any resulting seizures. The researchers also noted that the monoclonal antibody tocilizumab has proven effective in treating cytokine release syndrome.
In addition to this trial, JCAR015 is under investigation at Memorial Sloan-Kettering Cancer Center in a phase 1 trial of patients with relapsed and refractory non-Hodgkin lymphoma.
Two other CAR T-cell product candidates under development by the same company, Juno Therapeutics, Inc., are being tested in clinical trials as well. JCAR017 is under investigation at Seattle Children’s Hospital for relapsed/refractory CD19-positive pediatric leukemia.
JCAR014 is being tested for refractory chronic lymphocytic leukemia, non-Hodgkin lymphoma, and ALL at the Fred Hutchinson Cancer Research Center. Data on these programs are set to be presented at the 56th ASH Annual Meeting next week in San Francisco. ![]()

Credit: Charles Haymond
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for JCAR015, a chimeric antigen receptor (CAR) T-cell therapy, to treat patients with relapsed or refractory B-cell acute lymphoblastic leukemia (ALL).
Breakthrough designation is designed to help accelerate the development and review of new drugs for serious or life-threatening conditions.
The designation comes with potential benefits, including intensive FDA guidance and eligibility for priority review. It is granted to applicants when preliminary clinical evidence indicates the drug may demonstrate substantial improvement over existing therapies on one or more clinical endpoints.
The FDA recently granted JCAR015 orphan designation to treat ALL as well.
JCAR015 consists of autologous T cells expressing a CD19-specific, CD28/CD3z CAR. The treatment has shown promise in an ongoing phase 1 trial of patients with B-cell ALL.
Initial results from this study were published in Science Translational Medicine last year and in February. Updated results were presented at the AACR Annual Meeting in April.
At that point, the researchers had enrolled 22 adult patients with relapsed or refractory B-ALL who were minimal residual disease-positive or were in first complete remission at enrollment. Patients in complete remission were monitored and only received JCAR015 if they relapsed.
The remaining patients received re-induction chemotherapy (physician’s choice), followed by an infusion of JCAR015. After treatment, patients could receive allogeneic transplant, a different salvage therapy, or monitoring.
Eighty-two percent of patients achieved a complete response to JCAR015. The average time to complete response was about 24.5 days.
Twelve of the responders were eligible for transplant. Of the 8 patients who ultimately underwent transplant and survived, 1 relapsed, but the rest remained in remission.
Ten patients had died at the time of the AACR presentation. Six deaths were a result of disease relapse or progression, and 2 patients died of complications from stem cell transplant.
The 2 remaining deaths prompted a temporary suspension of enrollment in this trial. Those deaths were related to complications from cytokine release syndrome. One patient died of cardiovascular disease, and the other died following persistent seizure activity.
So researchers at the Memorial Sloan-Kettering Cancer Center, where the trial is being conducted, reviewed these cases. The results prompted them to amend trial enrollment criteria and dosing recommendations. Now, patients with cardiac disease are ineligible to receive JCAR015.
And the T-cell dose a patient receives will depend on the extent of his or her disease. The hope is that this will reduce the risk of cytokine release syndrome and any resulting seizures. The researchers also noted that the monoclonal antibody tocilizumab has proven effective in treating cytokine release syndrome.
In addition to this trial, JCAR015 is under investigation at Memorial Sloan-Kettering Cancer Center in a phase 1 trial of patients with relapsed and refractory non-Hodgkin lymphoma.
Two other CAR T-cell product candidates under development by the same company, Juno Therapeutics, Inc., are being tested in clinical trials as well. JCAR017 is under investigation at Seattle Children’s Hospital for relapsed/refractory CD19-positive pediatric leukemia.
JCAR014 is being tested for refractory chronic lymphocytic leukemia, non-Hodgkin lymphoma, and ALL at the Fred Hutchinson Cancer Research Center. Data on these programs are set to be presented at the 56th ASH Annual Meeting next week in San Francisco. ![]()
FDA grants CAR T-cell therapy orphan designation

The US Food and Drug Administration (FDA) has granted orphan drug designation for the chimeric antigen receptor (CAR) T-cell therapy JCAR015 to treat acute lymphoblastic leukemia (ALL).
The designation will provide the product’s developer, Juno Therapeutics, with multiple benefits, including the availability of grant money, certain tax credits, and 7 years of market exclusivity, as well as the possibility of an expedited regulatory process.
JCAR015 consists of autologous T cells expressing a CD19-specific, CD28/CD3z CAR. The treatment has shown promise in an ongoing phase 1 trial of patients with B-cell ALL.
Initial results from this study were published in Science Translational Medicine last year and in February. Updated results were presented at the AACR Annual Meeting in April.
At that point, the researchers had enrolled 22 adult patients with relapsed or refractory B-ALL who were minimal residual disease-positive or were in first complete remission at enrollment. Patients in complete remission were monitored and only received JCAR015 if they relapsed.
The remaining patients received re-induction chemotherapy (physician’s choice), followed by an infusion of JCAR015. After treatment, patients could receive allogeneic transplant, a different salvage therapy, or monitoring.
Eighty-two percent of patients achieved a complete response to JCAR015. The average time to complete response was about 24.5 days.
Twelve of the responders were eligible for transplant. Of the 8 patients who ultimately underwent transplant and survived, 1 relapsed, but the rest remained in remission.
Ten patients had died at the time of the AACR presentation. Six deaths were a result of disease relapse or progression, and 2 patients died of complications from stem cell transplant.
The 2 remaining deaths prompted a temporary suspension of enrollment in this trial.
Those deaths were related to complications from cytokine release syndrome. One patient died of cardiovascular disease, and the other died following “persistent seizure activity.”
So researchers at the Memorial Sloan-Kettering Cancer Center, where the trial is being conducted, reviewed these cases.
The results prompted them to amend trial enrollment criteria and dosing recommendations. Now, patients with cardiac disease are ineligible to receive JCAR015.
And the T-cell dose a patient receives will depend on the extent of his or her disease. The hope is that this will reduce the risk of cytokine release syndrome and any resulting seizures.
The researchers also noted that the monoclonal antibody tocilizumab has proven effective in treating cytokine release syndrome.
In addition to this trial, JCAR015 is under investigation in another phase 1 trial of patients with relapsed and refractory non-Hodgkin lymphoma. ![]()

The US Food and Drug Administration (FDA) has granted orphan drug designation for the chimeric antigen receptor (CAR) T-cell therapy JCAR015 to treat acute lymphoblastic leukemia (ALL).
The designation will provide the product’s developer, Juno Therapeutics, with multiple benefits, including the availability of grant money, certain tax credits, and 7 years of market exclusivity, as well as the possibility of an expedited regulatory process.
JCAR015 consists of autologous T cells expressing a CD19-specific, CD28/CD3z CAR. The treatment has shown promise in an ongoing phase 1 trial of patients with B-cell ALL.
Initial results from this study were published in Science Translational Medicine last year and in February. Updated results were presented at the AACR Annual Meeting in April.
At that point, the researchers had enrolled 22 adult patients with relapsed or refractory B-ALL who were minimal residual disease-positive or were in first complete remission at enrollment. Patients in complete remission were monitored and only received JCAR015 if they relapsed.
The remaining patients received re-induction chemotherapy (physician’s choice), followed by an infusion of JCAR015. After treatment, patients could receive allogeneic transplant, a different salvage therapy, or monitoring.
Eighty-two percent of patients achieved a complete response to JCAR015. The average time to complete response was about 24.5 days.
Twelve of the responders were eligible for transplant. Of the 8 patients who ultimately underwent transplant and survived, 1 relapsed, but the rest remained in remission.
Ten patients had died at the time of the AACR presentation. Six deaths were a result of disease relapse or progression, and 2 patients died of complications from stem cell transplant.
The 2 remaining deaths prompted a temporary suspension of enrollment in this trial.
Those deaths were related to complications from cytokine release syndrome. One patient died of cardiovascular disease, and the other died following “persistent seizure activity.”
So researchers at the Memorial Sloan-Kettering Cancer Center, where the trial is being conducted, reviewed these cases.
The results prompted them to amend trial enrollment criteria and dosing recommendations. Now, patients with cardiac disease are ineligible to receive JCAR015.
And the T-cell dose a patient receives will depend on the extent of his or her disease. The hope is that this will reduce the risk of cytokine release syndrome and any resulting seizures.
The researchers also noted that the monoclonal antibody tocilizumab has proven effective in treating cytokine release syndrome.
In addition to this trial, JCAR015 is under investigation in another phase 1 trial of patients with relapsed and refractory non-Hodgkin lymphoma. ![]()

The US Food and Drug Administration (FDA) has granted orphan drug designation for the chimeric antigen receptor (CAR) T-cell therapy JCAR015 to treat acute lymphoblastic leukemia (ALL).
The designation will provide the product’s developer, Juno Therapeutics, with multiple benefits, including the availability of grant money, certain tax credits, and 7 years of market exclusivity, as well as the possibility of an expedited regulatory process.
JCAR015 consists of autologous T cells expressing a CD19-specific, CD28/CD3z CAR. The treatment has shown promise in an ongoing phase 1 trial of patients with B-cell ALL.
Initial results from this study were published in Science Translational Medicine last year and in February. Updated results were presented at the AACR Annual Meeting in April.
At that point, the researchers had enrolled 22 adult patients with relapsed or refractory B-ALL who were minimal residual disease-positive or were in first complete remission at enrollment. Patients in complete remission were monitored and only received JCAR015 if they relapsed.
The remaining patients received re-induction chemotherapy (physician’s choice), followed by an infusion of JCAR015. After treatment, patients could receive allogeneic transplant, a different salvage therapy, or monitoring.
Eighty-two percent of patients achieved a complete response to JCAR015. The average time to complete response was about 24.5 days.
Twelve of the responders were eligible for transplant. Of the 8 patients who ultimately underwent transplant and survived, 1 relapsed, but the rest remained in remission.
Ten patients had died at the time of the AACR presentation. Six deaths were a result of disease relapse or progression, and 2 patients died of complications from stem cell transplant.
The 2 remaining deaths prompted a temporary suspension of enrollment in this trial.
Those deaths were related to complications from cytokine release syndrome. One patient died of cardiovascular disease, and the other died following “persistent seizure activity.”
So researchers at the Memorial Sloan-Kettering Cancer Center, where the trial is being conducted, reviewed these cases.
The results prompted them to amend trial enrollment criteria and dosing recommendations. Now, patients with cardiac disease are ineligible to receive JCAR015.
And the T-cell dose a patient receives will depend on the extent of his or her disease. The hope is that this will reduce the risk of cytokine release syndrome and any resulting seizures.
The researchers also noted that the monoclonal antibody tocilizumab has proven effective in treating cytokine release syndrome.
In addition to this trial, JCAR015 is under investigation in another phase 1 trial of patients with relapsed and refractory non-Hodgkin lymphoma. ![]()
RNAi therapy may eliminate resistance in B-ALL
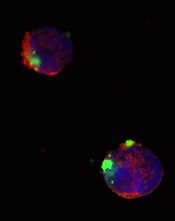
CD22ΔE12-siRNA nanoparticle
Credit: Fatih Uckun
Researchers have identified a molecular target for RNA interference (RNAi) therapy, and they believe that, by targeting this molecular lesion, they may be able to eliminate drug resistance in B-lineage acute lymphoblastic leukemia (ALL).
“We knew that we could kill chemotherapy-resistant leukemia cells if we only knew what made them so resistant,” said study author Fatih Uckun, MD, PhD, of the Children’s Hospital Los Angeles in California.
“Once we determined the mechanism, the next step was obvious—to rationally design a drug that would take out that specific target.”
The target is a defective gene that results in the production of abnormal CD22—CD22ΔE12. The researchers found that pediatric and adult B-lineage lymphoid malignancies are characterized by a high incidence of CD22ΔE12.
The group’s experiments revealed a causal link between CD22ΔE12 and the aggressive biology of B-ALL cells. When the researchers knocked down CD22ΔE12 in primary B-ALL cells using small interfering RNA (siRNA), they observed inhibition of clonogenicity.
The team therefore theorized that the siRNA-mediated depletion of CD22ΔE12 might be useful for treating B-ALL.
So they designed a nanoparticle formulation of CD22ΔE12-siRNA in which a polypeptide functions as the delivery system. The particle has a diameter of 100 nanometers.
Experiments showed that the nanoparticle could deliver its siRNA cargo into the cytosol of ALL-1 cells, thereby depleting CD22ΔE12 and inhibiting leukemic cell growth in vitro.
The researchers said that further development of this nanoparticle or other nanoformulation platforms for CD22ΔE12-siRNA could facilitate an effective therapeutic RNAi strategy to treat aggressive or chemotherapy-resistant B-lineage lymphoid malignancies.
“The goal is to translate our recent research discoveries in nanotechnology and biotherapy into effective patient-tailored treatment programs for the most common form of childhood cancer,” Dr Uckun said.
He and his colleagues described this research in EBioMedicine. ![]()

CD22ΔE12-siRNA nanoparticle
Credit: Fatih Uckun
Researchers have identified a molecular target for RNA interference (RNAi) therapy, and they believe that, by targeting this molecular lesion, they may be able to eliminate drug resistance in B-lineage acute lymphoblastic leukemia (ALL).
“We knew that we could kill chemotherapy-resistant leukemia cells if we only knew what made them so resistant,” said study author Fatih Uckun, MD, PhD, of the Children’s Hospital Los Angeles in California.
“Once we determined the mechanism, the next step was obvious—to rationally design a drug that would take out that specific target.”
The target is a defective gene that results in the production of abnormal CD22—CD22ΔE12. The researchers found that pediatric and adult B-lineage lymphoid malignancies are characterized by a high incidence of CD22ΔE12.
The group’s experiments revealed a causal link between CD22ΔE12 and the aggressive biology of B-ALL cells. When the researchers knocked down CD22ΔE12 in primary B-ALL cells using small interfering RNA (siRNA), they observed inhibition of clonogenicity.
The team therefore theorized that the siRNA-mediated depletion of CD22ΔE12 might be useful for treating B-ALL.
So they designed a nanoparticle formulation of CD22ΔE12-siRNA in which a polypeptide functions as the delivery system. The particle has a diameter of 100 nanometers.
Experiments showed that the nanoparticle could deliver its siRNA cargo into the cytosol of ALL-1 cells, thereby depleting CD22ΔE12 and inhibiting leukemic cell growth in vitro.
The researchers said that further development of this nanoparticle or other nanoformulation platforms for CD22ΔE12-siRNA could facilitate an effective therapeutic RNAi strategy to treat aggressive or chemotherapy-resistant B-lineage lymphoid malignancies.
“The goal is to translate our recent research discoveries in nanotechnology and biotherapy into effective patient-tailored treatment programs for the most common form of childhood cancer,” Dr Uckun said.
He and his colleagues described this research in EBioMedicine. ![]()

CD22ΔE12-siRNA nanoparticle
Credit: Fatih Uckun
Researchers have identified a molecular target for RNA interference (RNAi) therapy, and they believe that, by targeting this molecular lesion, they may be able to eliminate drug resistance in B-lineage acute lymphoblastic leukemia (ALL).
“We knew that we could kill chemotherapy-resistant leukemia cells if we only knew what made them so resistant,” said study author Fatih Uckun, MD, PhD, of the Children’s Hospital Los Angeles in California.
“Once we determined the mechanism, the next step was obvious—to rationally design a drug that would take out that specific target.”
The target is a defective gene that results in the production of abnormal CD22—CD22ΔE12. The researchers found that pediatric and adult B-lineage lymphoid malignancies are characterized by a high incidence of CD22ΔE12.
The group’s experiments revealed a causal link between CD22ΔE12 and the aggressive biology of B-ALL cells. When the researchers knocked down CD22ΔE12 in primary B-ALL cells using small interfering RNA (siRNA), they observed inhibition of clonogenicity.
The team therefore theorized that the siRNA-mediated depletion of CD22ΔE12 might be useful for treating B-ALL.
So they designed a nanoparticle formulation of CD22ΔE12-siRNA in which a polypeptide functions as the delivery system. The particle has a diameter of 100 nanometers.
Experiments showed that the nanoparticle could deliver its siRNA cargo into the cytosol of ALL-1 cells, thereby depleting CD22ΔE12 and inhibiting leukemic cell growth in vitro.
The researchers said that further development of this nanoparticle or other nanoformulation platforms for CD22ΔE12-siRNA could facilitate an effective therapeutic RNAi strategy to treat aggressive or chemotherapy-resistant B-lineage lymphoid malignancies.
“The goal is to translate our recent research discoveries in nanotechnology and biotherapy into effective patient-tailored treatment programs for the most common form of childhood cancer,” Dr Uckun said.
He and his colleagues described this research in EBioMedicine. ![]()
Risk-directed therapy improves survival in ALL

Credit: St Jude Biomedical
Communications
Risk-directed therapy can level the playing field for patients with BCR-ABL1-like acute lymphoblastic leukemia (ALL), new research suggests.
Using minimal residual disease (MRD) and other risk factors to guide treatment intensity allowed patients withBCR-ABL1-like ALL to have survival rates comparable to those of other B-ALL patients.
The research also revealed that not all BCR-ABL1-like ALL patients have high-risk disease.
Ching-Hon Pui, MD, of St Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues reported these results in the Journal of Clinical Oncology.
Patients with BCR-ABL1-like ALL tend to have poor outcomes, so Dr Pui and his colleagues evaluated the utility of risk-directed therapy in these and other B-ALL patients.
The team assessed patients enrolled in the Total Therapy XV study between 2000 and 2007. They were 1 to 18 years of age at diagnosis. There were 344 subjects with adequate samples for gene expression profiling.
Forty patients (11.6%) had BCR-ABL1-like ALL. They were significantly more likely than other study participants to be male, have Down syndrome, and have higher MRD levels on day 19 and at the end of induction.
The researchers monitored patients and adjusted their treatment intensity based on MRD at days 19 and 42. The treatment regimen was described in JAMA in 2009.
The MRD monitoring combined with conventional risk factors, such as patient age and white blood count at diagnosis, demonstrated that BCR-ABL1-like ALL is not a uniformly high-risk disease.
Forty percent of BCR-ABL1-like ALL patients were actually classified as having low-risk disease because they had other favorable clinical or biological features and no MRD at the end of remission induction.
The other 60% were classified as having standard-risk or high-risk disease. The group included 6 patients who underwent transplant.
There were no significant differences in event-free or overall survival in patients with BCR-ABL1-like ALL and the other ALL patients. At 5 years, event-free survival was 90.0% ± 4.7% and 88.4% ± 1.9%, respectively. And 5-year overall survival was 92.5% ± 4.2% and 95.1% ± 1.3%, respectively.
When available, more sophisticated genetic testing should be used to identify which of the B-ALL patients with high levels of MRD have the BCR-ABL1-like ALL subtype, Dr Pui said. Many of these patients have genetic alterations that make them responsive to tyrosine kinase inhibitors (TKIs) and possibly other targeted therapies.
For example, in this study, the researchers evaluated genetic abnormalities in 25 BCR-ABL1-like ALL patients. Eleven had a genomic rearrangement of CRLF2, 6 had fusion transcripts responsive to ABL TKIs or JAK inhibitors, and 7 had mutations involving the Ras signaling pathway.
“In the future, genetic testing will likely be used at diagnosis to identify [BCR-ABL1-like ALL] and direct patients to the best targeted therapy,” Dr Pui said, “possibly including some drugs that are currently experimental.” ![]()

Credit: St Jude Biomedical
Communications
Risk-directed therapy can level the playing field for patients with BCR-ABL1-like acute lymphoblastic leukemia (ALL), new research suggests.
Using minimal residual disease (MRD) and other risk factors to guide treatment intensity allowed patients withBCR-ABL1-like ALL to have survival rates comparable to those of other B-ALL patients.
The research also revealed that not all BCR-ABL1-like ALL patients have high-risk disease.
Ching-Hon Pui, MD, of St Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues reported these results in the Journal of Clinical Oncology.
Patients with BCR-ABL1-like ALL tend to have poor outcomes, so Dr Pui and his colleagues evaluated the utility of risk-directed therapy in these and other B-ALL patients.
The team assessed patients enrolled in the Total Therapy XV study between 2000 and 2007. They were 1 to 18 years of age at diagnosis. There were 344 subjects with adequate samples for gene expression profiling.
Forty patients (11.6%) had BCR-ABL1-like ALL. They were significantly more likely than other study participants to be male, have Down syndrome, and have higher MRD levels on day 19 and at the end of induction.
The researchers monitored patients and adjusted their treatment intensity based on MRD at days 19 and 42. The treatment regimen was described in JAMA in 2009.
The MRD monitoring combined with conventional risk factors, such as patient age and white blood count at diagnosis, demonstrated that BCR-ABL1-like ALL is not a uniformly high-risk disease.
Forty percent of BCR-ABL1-like ALL patients were actually classified as having low-risk disease because they had other favorable clinical or biological features and no MRD at the end of remission induction.
The other 60% were classified as having standard-risk or high-risk disease. The group included 6 patients who underwent transplant.
There were no significant differences in event-free or overall survival in patients with BCR-ABL1-like ALL and the other ALL patients. At 5 years, event-free survival was 90.0% ± 4.7% and 88.4% ± 1.9%, respectively. And 5-year overall survival was 92.5% ± 4.2% and 95.1% ± 1.3%, respectively.
When available, more sophisticated genetic testing should be used to identify which of the B-ALL patients with high levels of MRD have the BCR-ABL1-like ALL subtype, Dr Pui said. Many of these patients have genetic alterations that make them responsive to tyrosine kinase inhibitors (TKIs) and possibly other targeted therapies.
For example, in this study, the researchers evaluated genetic abnormalities in 25 BCR-ABL1-like ALL patients. Eleven had a genomic rearrangement of CRLF2, 6 had fusion transcripts responsive to ABL TKIs or JAK inhibitors, and 7 had mutations involving the Ras signaling pathway.
“In the future, genetic testing will likely be used at diagnosis to identify [BCR-ABL1-like ALL] and direct patients to the best targeted therapy,” Dr Pui said, “possibly including some drugs that are currently experimental.” ![]()

Credit: St Jude Biomedical
Communications
Risk-directed therapy can level the playing field for patients with BCR-ABL1-like acute lymphoblastic leukemia (ALL), new research suggests.
Using minimal residual disease (MRD) and other risk factors to guide treatment intensity allowed patients withBCR-ABL1-like ALL to have survival rates comparable to those of other B-ALL patients.
The research also revealed that not all BCR-ABL1-like ALL patients have high-risk disease.
Ching-Hon Pui, MD, of St Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues reported these results in the Journal of Clinical Oncology.
Patients with BCR-ABL1-like ALL tend to have poor outcomes, so Dr Pui and his colleagues evaluated the utility of risk-directed therapy in these and other B-ALL patients.
The team assessed patients enrolled in the Total Therapy XV study between 2000 and 2007. They were 1 to 18 years of age at diagnosis. There were 344 subjects with adequate samples for gene expression profiling.
Forty patients (11.6%) had BCR-ABL1-like ALL. They were significantly more likely than other study participants to be male, have Down syndrome, and have higher MRD levels on day 19 and at the end of induction.
The researchers monitored patients and adjusted their treatment intensity based on MRD at days 19 and 42. The treatment regimen was described in JAMA in 2009.
The MRD monitoring combined with conventional risk factors, such as patient age and white blood count at diagnosis, demonstrated that BCR-ABL1-like ALL is not a uniformly high-risk disease.
Forty percent of BCR-ABL1-like ALL patients were actually classified as having low-risk disease because they had other favorable clinical or biological features and no MRD at the end of remission induction.
The other 60% were classified as having standard-risk or high-risk disease. The group included 6 patients who underwent transplant.
There were no significant differences in event-free or overall survival in patients with BCR-ABL1-like ALL and the other ALL patients. At 5 years, event-free survival was 90.0% ± 4.7% and 88.4% ± 1.9%, respectively. And 5-year overall survival was 92.5% ± 4.2% and 95.1% ± 1.3%, respectively.
When available, more sophisticated genetic testing should be used to identify which of the B-ALL patients with high levels of MRD have the BCR-ABL1-like ALL subtype, Dr Pui said. Many of these patients have genetic alterations that make them responsive to tyrosine kinase inhibitors (TKIs) and possibly other targeted therapies.
For example, in this study, the researchers evaluated genetic abnormalities in 25 BCR-ABL1-like ALL patients. Eleven had a genomic rearrangement of CRLF2, 6 had fusion transcripts responsive to ABL TKIs or JAK inhibitors, and 7 had mutations involving the Ras signaling pathway.
“In the future, genetic testing will likely be used at diagnosis to identify [BCR-ABL1-like ALL] and direct patients to the best targeted therapy,” Dr Pui said, “possibly including some drugs that are currently experimental.” ![]()
Product appears safe, effective in rel/ref ALL

Credit: Bill Branson
Eryaspase, a product consisting of L-asparaginase encapsulated in red blood cells, may be safer and more effective than native E coli L-asparaginase, results of a phase 2/3 study suggest.
In patients with relapsed or refractory acute lymphoblastic leukemia (ALL), eryaspase given in combination with chemotherapy produced a higher complete response rate and fewer allergic reactions than native E coli L-asparaginase in combination with chemotherapy.
Eryaspase was also well-tolerated in patients who had previously experienced allergic reactions to L-asparaginase.
ERYTECH Pharma, the company developing eryaspase, recently announced these results from the GRASPIVOTALL trial.
The study included 80 children and adults with relapsed or refractory ALL, who were randomized to 1 of 3 treatment arms.
In the first 2 arms, researchers compared eryaspase to native E coli L-asparaginase, both in combination with standard chemotherapy (COOPRALL), in patients without prior allergies to L-asparaginase. In the third arm, the team evaluated eryaspase for patients who have experienced allergic reactions related to asparaginase in their first-line treatment.
The primary endpoint of the study consisted of 2 objectives: (a) superior safety, expressed as a significant reduction of the incidence of allergic reactions with eryaspase compared to native L-asparaginase, and (b) noninferior duration of asparaginase activity above the threshold of 100 IU/L during the induction phase in the nonallergic patients. Both endpoints needed to be met for the study to be considered positive.
The main secondary efficacy endpoints included complete response, minimal residual disease, event-free survival, and overall survival.
At 1 year of follow-up, both primary endpoints were met. There was a significant reduction of allergic reactions in the eryaspase arm compared to the native L-asparaginase arm—0% (0/26) and 42.9% (12/28), respectively (P<0.001).
And there was a significant increase in the duration of circulating asparaginase activity in the eryaspase arm compared to the native L-asparaginase arm. Asparaginase levels were maintained above 100 IU/L for an average of 20.5 days with up to 2 injections during the first month of treatment with eryaspase, compared to 9.2 days in the native L-asparaginase arm, with up to 8 injections of L-asparaginase (P<0.001).
In addition, the complete response rate was higher with eryaspase. At the end of the induction phase, 71.4% of patients (n=15) in the eryaspase arm had a complete response, compared to 42.3% of patients (n=11) in the native L-asparaginase arm.
Finally, the study showed that eryaspase is well-tolerated by patients with previous allergies to L-asparaginase. Two of the 26 patients with prior allergies to L-asparaginase experienced mild allergic reactions to eryaspase.
“The results of this study are an important step forward for the treatment of ALL patients that are at risk to receive L-asparaginase, which remains an important unmet medical need,” said Yves Betrand, MD, of the Institute for Pediatric Hematology and Oncology in Lyon, France.
“The virtual absence of allergic reactions, also in patients with prior allergies to L-asparaginase, is very encouraging.”
The analysis of additional secondary and exploratory endpoints for this study is ongoing. ERYTECH said results will be available later this year and are set to be presented at an upcoming scientific conference.
Based on the results of this and earlier studies of eryaspase, ERYTECH intends to submit its application for European Marketing Authorization in the first half of 2015.
The company also plans to accelerate the product’s development in ALL in the US and to launch phase 2 clinical trials in additional oncology indications with high unmet medical need. A phase 2b study of eryaspase in acute myeloid leukemia is already underway, with more than half of the patients enrolled. ![]()

Credit: Bill Branson
Eryaspase, a product consisting of L-asparaginase encapsulated in red blood cells, may be safer and more effective than native E coli L-asparaginase, results of a phase 2/3 study suggest.
In patients with relapsed or refractory acute lymphoblastic leukemia (ALL), eryaspase given in combination with chemotherapy produced a higher complete response rate and fewer allergic reactions than native E coli L-asparaginase in combination with chemotherapy.
Eryaspase was also well-tolerated in patients who had previously experienced allergic reactions to L-asparaginase.
ERYTECH Pharma, the company developing eryaspase, recently announced these results from the GRASPIVOTALL trial.
The study included 80 children and adults with relapsed or refractory ALL, who were randomized to 1 of 3 treatment arms.
In the first 2 arms, researchers compared eryaspase to native E coli L-asparaginase, both in combination with standard chemotherapy (COOPRALL), in patients without prior allergies to L-asparaginase. In the third arm, the team evaluated eryaspase for patients who have experienced allergic reactions related to asparaginase in their first-line treatment.
The primary endpoint of the study consisted of 2 objectives: (a) superior safety, expressed as a significant reduction of the incidence of allergic reactions with eryaspase compared to native L-asparaginase, and (b) noninferior duration of asparaginase activity above the threshold of 100 IU/L during the induction phase in the nonallergic patients. Both endpoints needed to be met for the study to be considered positive.
The main secondary efficacy endpoints included complete response, minimal residual disease, event-free survival, and overall survival.
At 1 year of follow-up, both primary endpoints were met. There was a significant reduction of allergic reactions in the eryaspase arm compared to the native L-asparaginase arm—0% (0/26) and 42.9% (12/28), respectively (P<0.001).
And there was a significant increase in the duration of circulating asparaginase activity in the eryaspase arm compared to the native L-asparaginase arm. Asparaginase levels were maintained above 100 IU/L for an average of 20.5 days with up to 2 injections during the first month of treatment with eryaspase, compared to 9.2 days in the native L-asparaginase arm, with up to 8 injections of L-asparaginase (P<0.001).
In addition, the complete response rate was higher with eryaspase. At the end of the induction phase, 71.4% of patients (n=15) in the eryaspase arm had a complete response, compared to 42.3% of patients (n=11) in the native L-asparaginase arm.
Finally, the study showed that eryaspase is well-tolerated by patients with previous allergies to L-asparaginase. Two of the 26 patients with prior allergies to L-asparaginase experienced mild allergic reactions to eryaspase.
“The results of this study are an important step forward for the treatment of ALL patients that are at risk to receive L-asparaginase, which remains an important unmet medical need,” said Yves Betrand, MD, of the Institute for Pediatric Hematology and Oncology in Lyon, France.
“The virtual absence of allergic reactions, also in patients with prior allergies to L-asparaginase, is very encouraging.”
The analysis of additional secondary and exploratory endpoints for this study is ongoing. ERYTECH said results will be available later this year and are set to be presented at an upcoming scientific conference.
Based on the results of this and earlier studies of eryaspase, ERYTECH intends to submit its application for European Marketing Authorization in the first half of 2015.
The company also plans to accelerate the product’s development in ALL in the US and to launch phase 2 clinical trials in additional oncology indications with high unmet medical need. A phase 2b study of eryaspase in acute myeloid leukemia is already underway, with more than half of the patients enrolled. ![]()

Credit: Bill Branson
Eryaspase, a product consisting of L-asparaginase encapsulated in red blood cells, may be safer and more effective than native E coli L-asparaginase, results of a phase 2/3 study suggest.
In patients with relapsed or refractory acute lymphoblastic leukemia (ALL), eryaspase given in combination with chemotherapy produced a higher complete response rate and fewer allergic reactions than native E coli L-asparaginase in combination with chemotherapy.
Eryaspase was also well-tolerated in patients who had previously experienced allergic reactions to L-asparaginase.
ERYTECH Pharma, the company developing eryaspase, recently announced these results from the GRASPIVOTALL trial.
The study included 80 children and adults with relapsed or refractory ALL, who were randomized to 1 of 3 treatment arms.
In the first 2 arms, researchers compared eryaspase to native E coli L-asparaginase, both in combination with standard chemotherapy (COOPRALL), in patients without prior allergies to L-asparaginase. In the third arm, the team evaluated eryaspase for patients who have experienced allergic reactions related to asparaginase in their first-line treatment.
The primary endpoint of the study consisted of 2 objectives: (a) superior safety, expressed as a significant reduction of the incidence of allergic reactions with eryaspase compared to native L-asparaginase, and (b) noninferior duration of asparaginase activity above the threshold of 100 IU/L during the induction phase in the nonallergic patients. Both endpoints needed to be met for the study to be considered positive.
The main secondary efficacy endpoints included complete response, minimal residual disease, event-free survival, and overall survival.
At 1 year of follow-up, both primary endpoints were met. There was a significant reduction of allergic reactions in the eryaspase arm compared to the native L-asparaginase arm—0% (0/26) and 42.9% (12/28), respectively (P<0.001).
And there was a significant increase in the duration of circulating asparaginase activity in the eryaspase arm compared to the native L-asparaginase arm. Asparaginase levels were maintained above 100 IU/L for an average of 20.5 days with up to 2 injections during the first month of treatment with eryaspase, compared to 9.2 days in the native L-asparaginase arm, with up to 8 injections of L-asparaginase (P<0.001).
In addition, the complete response rate was higher with eryaspase. At the end of the induction phase, 71.4% of patients (n=15) in the eryaspase arm had a complete response, compared to 42.3% of patients (n=11) in the native L-asparaginase arm.
Finally, the study showed that eryaspase is well-tolerated by patients with previous allergies to L-asparaginase. Two of the 26 patients with prior allergies to L-asparaginase experienced mild allergic reactions to eryaspase.
“The results of this study are an important step forward for the treatment of ALL patients that are at risk to receive L-asparaginase, which remains an important unmet medical need,” said Yves Betrand, MD, of the Institute for Pediatric Hematology and Oncology in Lyon, France.
“The virtual absence of allergic reactions, also in patients with prior allergies to L-asparaginase, is very encouraging.”
The analysis of additional secondary and exploratory endpoints for this study is ongoing. ERYTECH said results will be available later this year and are set to be presented at an upcoming scientific conference.
Based on the results of this and earlier studies of eryaspase, ERYTECH intends to submit its application for European Marketing Authorization in the first half of 2015.
The company also plans to accelerate the product’s development in ALL in the US and to launch phase 2 clinical trials in additional oncology indications with high unmet medical need. A phase 2b study of eryaspase in acute myeloid leukemia is already underway, with more than half of the patients enrolled. ![]()
Targeting drug resistance earlier

Credit: PNAS
PHILADELPHIA—A combination treatment strategy that takes tumor evolution into account could help us avoid drug resistance in hematologic malignancies, researchers say.
Preclinical experiments suggest we can prevent resistance by starting secondary treatment prior to relapse.
For example, a patient receiving dasatinib for acute lymphoblastic leukemia (ALL) could benefit from receiving crizotinib or foretinib during the early stages of clonal evolution.
The researchers described this strategy in a poster at the AACR conference Hematologic Malignancies: Translating Discoveries to Novel Therapies.
“Our goal is to identify vulnerabilities in cancer across stages of tumor evolution while it is developing resistance to initial treatment, to help guide the design of drug combination strategies,” said Douglas Lauffenburger, PhD, of the Koch Institute for Integrative Cancer Research at MIT in Boston.
“There may be many stages in a tumor evolution while under treatment that may make them vulnerable to already existing therapies. Rather than waiting for the tumor to become resistant to the first treatment and then thinking about a second-line drug to use, we can capitalize on opportunities that exploit vulnerabilities at different early stages, as the tumor is evolving to become resistant to the first drug.”
Dr Lauffenburger and his colleagues used a combination of computational and experimental approaches to identify drugs likely to be effective against a murine ALL cell line as the cells evolve.
To develop drug combinations based on the characteristics of evolving tumors, the researchers used escalating doses of imatinib, dasatinib, nilotinib, foretinib, and crizotinib on ALL cells.
As some cells exhibited resistance to a particular drug, the team treated the resistant cells with other drugs to check for cross-resistance. They found that resistant cells surviving at low multiples of the original drug dose actually demonstrated sensitization to certain other drugs, with the sensitization abrogated at higher doses.
Specifically, cell populations that were resistant to dasatinib at 1x and 2x IC90 became even more sensitive to crizotinib and foretinib. However, the sensitivity was lost at IC90 4x and above.
“Instead of only looking for the most resistant population of ALL cells at the end of this selection process, we monitored for drug sensitivity of the cells at each stage of the dose escalation,” Dr Lauffenburger explained.
“This led us to discover the vulnerabilities of a tumor at different stages of clonal evolution, a phenomenon we would have missed if we only analyzed for drug sensitivity at the last stage of this process, which is equivalent to when a patient has relapsed.” ![]()

Credit: PNAS
PHILADELPHIA—A combination treatment strategy that takes tumor evolution into account could help us avoid drug resistance in hematologic malignancies, researchers say.
Preclinical experiments suggest we can prevent resistance by starting secondary treatment prior to relapse.
For example, a patient receiving dasatinib for acute lymphoblastic leukemia (ALL) could benefit from receiving crizotinib or foretinib during the early stages of clonal evolution.
The researchers described this strategy in a poster at the AACR conference Hematologic Malignancies: Translating Discoveries to Novel Therapies.
“Our goal is to identify vulnerabilities in cancer across stages of tumor evolution while it is developing resistance to initial treatment, to help guide the design of drug combination strategies,” said Douglas Lauffenburger, PhD, of the Koch Institute for Integrative Cancer Research at MIT in Boston.
“There may be many stages in a tumor evolution while under treatment that may make them vulnerable to already existing therapies. Rather than waiting for the tumor to become resistant to the first treatment and then thinking about a second-line drug to use, we can capitalize on opportunities that exploit vulnerabilities at different early stages, as the tumor is evolving to become resistant to the first drug.”
Dr Lauffenburger and his colleagues used a combination of computational and experimental approaches to identify drugs likely to be effective against a murine ALL cell line as the cells evolve.
To develop drug combinations based on the characteristics of evolving tumors, the researchers used escalating doses of imatinib, dasatinib, nilotinib, foretinib, and crizotinib on ALL cells.
As some cells exhibited resistance to a particular drug, the team treated the resistant cells with other drugs to check for cross-resistance. They found that resistant cells surviving at low multiples of the original drug dose actually demonstrated sensitization to certain other drugs, with the sensitization abrogated at higher doses.
Specifically, cell populations that were resistant to dasatinib at 1x and 2x IC90 became even more sensitive to crizotinib and foretinib. However, the sensitivity was lost at IC90 4x and above.
“Instead of only looking for the most resistant population of ALL cells at the end of this selection process, we monitored for drug sensitivity of the cells at each stage of the dose escalation,” Dr Lauffenburger explained.
“This led us to discover the vulnerabilities of a tumor at different stages of clonal evolution, a phenomenon we would have missed if we only analyzed for drug sensitivity at the last stage of this process, which is equivalent to when a patient has relapsed.” ![]()

Credit: PNAS
PHILADELPHIA—A combination treatment strategy that takes tumor evolution into account could help us avoid drug resistance in hematologic malignancies, researchers say.
Preclinical experiments suggest we can prevent resistance by starting secondary treatment prior to relapse.
For example, a patient receiving dasatinib for acute lymphoblastic leukemia (ALL) could benefit from receiving crizotinib or foretinib during the early stages of clonal evolution.
The researchers described this strategy in a poster at the AACR conference Hematologic Malignancies: Translating Discoveries to Novel Therapies.
“Our goal is to identify vulnerabilities in cancer across stages of tumor evolution while it is developing resistance to initial treatment, to help guide the design of drug combination strategies,” said Douglas Lauffenburger, PhD, of the Koch Institute for Integrative Cancer Research at MIT in Boston.
“There may be many stages in a tumor evolution while under treatment that may make them vulnerable to already existing therapies. Rather than waiting for the tumor to become resistant to the first treatment and then thinking about a second-line drug to use, we can capitalize on opportunities that exploit vulnerabilities at different early stages, as the tumor is evolving to become resistant to the first drug.”
Dr Lauffenburger and his colleagues used a combination of computational and experimental approaches to identify drugs likely to be effective against a murine ALL cell line as the cells evolve.
To develop drug combinations based on the characteristics of evolving tumors, the researchers used escalating doses of imatinib, dasatinib, nilotinib, foretinib, and crizotinib on ALL cells.
As some cells exhibited resistance to a particular drug, the team treated the resistant cells with other drugs to check for cross-resistance. They found that resistant cells surviving at low multiples of the original drug dose actually demonstrated sensitization to certain other drugs, with the sensitization abrogated at higher doses.
Specifically, cell populations that were resistant to dasatinib at 1x and 2x IC90 became even more sensitive to crizotinib and foretinib. However, the sensitivity was lost at IC90 4x and above.
“Instead of only looking for the most resistant population of ALL cells at the end of this selection process, we monitored for drug sensitivity of the cells at each stage of the dose escalation,” Dr Lauffenburger explained.
“This led us to discover the vulnerabilities of a tumor at different stages of clonal evolution, a phenomenon we would have missed if we only analyzed for drug sensitivity at the last stage of this process, which is equivalent to when a patient has relapsed.” ![]()



