User login
Topicals, PDT offer field treatment options for actinic keratoses
LAS VEGAS – The addition of field treatment to targeted lesion treatment may offer patients the best outcomes when addressing actinic keratoses (AKs), said Dr. David M. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk, Virginia.
Speaking at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar, Dr. Pariser said that the rationale for treating AKs stems from the fact that not all AKs will transform to squamous cell carcinoma (SCC). “We don’t know which ones will and which ones won’t.” Though treatment can be guided by a shared decision-making approach, a personal history of multiple skin cancers is an obvious reason for concern, as is any drug-related or disease-related immunosuppression, he added.
Available treatments for AKs include cryosurgery, curettage, and electrodessication; photodynamic therapy (PDT); and topical agents, including 5-fluorouracil (5-FU), diclofenac, imiquimod, and ingenol mebutate.
“Cryosurgery is what most of us do for most AKs,” said Dr. Pariser, observing that this is a bread-and-butter procedure that is part of the backbone of most dermatology practices. Cryosurgery, curettage, and electrodessication are all used to treat discrete lesions, especially for higher-grade AKs.
However, Dr. Pariser noted that many AKs occur in a “field of cancerization.” This is a larger area of photodamaged skin; the skin may not appear grossly abnormal, but deranged areas will be diffusely evident with fluorescence detection, “indicating that all of this area was damaged,” he said.
PDT and topical agents effectively provide field treatment, and do not just address discrete lesions, although field treatment is technically an off-label use for these modalities, he added.
Lower concentrations and shorter courses of topical agents may be effective and better tolerated by patients, Dr. Pariser said. In clinical trials, for example, the efficacy of imiquimod 2.5% and 3.75% has been comparable to the stronger 5% formulation. Treatment cycles lasting 2 rather than 3 weeks have also shown similar efficacy. The greater palatability of these regimens to patients may increase real-world adherence as well.
Cure rates for PDT range from 67% to 92% for both the blue light–aminolevulinic acid modality and the red light–methyl aminolevulinate more commonly used in Europe. These modalities also treat a broad field, offer cure rates comparable to other topical modalities, and result in less scarring than destructive modalities, Dr. Pariser said.
The pain of PDT may be understandably off-putting for some patients, he commented. In the United States, investigators are experimenting with shorter incubation times for the ALA topical solutions. One study found comparable reductions in the number of AKs by the end of the study period for 1 versus 2 or 3 hours of incubation, and a 2-hour incubation period with spot treatment only was also similarly effective. Clearance rates ranged from about 70%-80%, and all were highly statistically significant, compared with the vehicle-only arm. However, “a second treatment at week 8 seems to be necessary for improved efficacy,” Dr. Pariser said.
Another treatment strategy currently being investigated is “painless PDT,” where the agent is applied and the field is illuminated immediately. Photo-bleaching degrades the protoporphyrins as they are produced, dramatically reducing patient discomfort. Efficacy is promising, he added.
In Europe, “daylight PDT” is being explored – the photosensitizing agent is applied, and the patient simply goes about his or her daily business in normal daylight. This is a nearly painless therapy and shows promise for efficacy. However, said Dr. Pariser, the realities of practice economics make it very unlikely that daylight PDT will catch on in the United States, since the daylight exposure does not represent a billable procedure.
Dr. Pariser reiterated that regardless of the modality, “field therapy has better long-term efficacy rates and better sustained clearance no matter which one of the agents we use, and combination therapy [of lesional and field treatment] seems to be the most effective.”
Dr. Pariser disclosed that he is an investigator and consultant for DUSA Pharmaceuticals, Photocure, and LEO Pharma, and that he discussed off-label uses of drugs and devices.
SDEF and this news organization are owned by the same parent company.
On Twitter @karioakes
LAS VEGAS – The addition of field treatment to targeted lesion treatment may offer patients the best outcomes when addressing actinic keratoses (AKs), said Dr. David M. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk, Virginia.
Speaking at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar, Dr. Pariser said that the rationale for treating AKs stems from the fact that not all AKs will transform to squamous cell carcinoma (SCC). “We don’t know which ones will and which ones won’t.” Though treatment can be guided by a shared decision-making approach, a personal history of multiple skin cancers is an obvious reason for concern, as is any drug-related or disease-related immunosuppression, he added.
Available treatments for AKs include cryosurgery, curettage, and electrodessication; photodynamic therapy (PDT); and topical agents, including 5-fluorouracil (5-FU), diclofenac, imiquimod, and ingenol mebutate.
“Cryosurgery is what most of us do for most AKs,” said Dr. Pariser, observing that this is a bread-and-butter procedure that is part of the backbone of most dermatology practices. Cryosurgery, curettage, and electrodessication are all used to treat discrete lesions, especially for higher-grade AKs.
However, Dr. Pariser noted that many AKs occur in a “field of cancerization.” This is a larger area of photodamaged skin; the skin may not appear grossly abnormal, but deranged areas will be diffusely evident with fluorescence detection, “indicating that all of this area was damaged,” he said.
PDT and topical agents effectively provide field treatment, and do not just address discrete lesions, although field treatment is technically an off-label use for these modalities, he added.
Lower concentrations and shorter courses of topical agents may be effective and better tolerated by patients, Dr. Pariser said. In clinical trials, for example, the efficacy of imiquimod 2.5% and 3.75% has been comparable to the stronger 5% formulation. Treatment cycles lasting 2 rather than 3 weeks have also shown similar efficacy. The greater palatability of these regimens to patients may increase real-world adherence as well.
Cure rates for PDT range from 67% to 92% for both the blue light–aminolevulinic acid modality and the red light–methyl aminolevulinate more commonly used in Europe. These modalities also treat a broad field, offer cure rates comparable to other topical modalities, and result in less scarring than destructive modalities, Dr. Pariser said.
The pain of PDT may be understandably off-putting for some patients, he commented. In the United States, investigators are experimenting with shorter incubation times for the ALA topical solutions. One study found comparable reductions in the number of AKs by the end of the study period for 1 versus 2 or 3 hours of incubation, and a 2-hour incubation period with spot treatment only was also similarly effective. Clearance rates ranged from about 70%-80%, and all were highly statistically significant, compared with the vehicle-only arm. However, “a second treatment at week 8 seems to be necessary for improved efficacy,” Dr. Pariser said.
Another treatment strategy currently being investigated is “painless PDT,” where the agent is applied and the field is illuminated immediately. Photo-bleaching degrades the protoporphyrins as they are produced, dramatically reducing patient discomfort. Efficacy is promising, he added.
In Europe, “daylight PDT” is being explored – the photosensitizing agent is applied, and the patient simply goes about his or her daily business in normal daylight. This is a nearly painless therapy and shows promise for efficacy. However, said Dr. Pariser, the realities of practice economics make it very unlikely that daylight PDT will catch on in the United States, since the daylight exposure does not represent a billable procedure.
Dr. Pariser reiterated that regardless of the modality, “field therapy has better long-term efficacy rates and better sustained clearance no matter which one of the agents we use, and combination therapy [of lesional and field treatment] seems to be the most effective.”
Dr. Pariser disclosed that he is an investigator and consultant for DUSA Pharmaceuticals, Photocure, and LEO Pharma, and that he discussed off-label uses of drugs and devices.
SDEF and this news organization are owned by the same parent company.
On Twitter @karioakes
LAS VEGAS – The addition of field treatment to targeted lesion treatment may offer patients the best outcomes when addressing actinic keratoses (AKs), said Dr. David M. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk, Virginia.
Speaking at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar, Dr. Pariser said that the rationale for treating AKs stems from the fact that not all AKs will transform to squamous cell carcinoma (SCC). “We don’t know which ones will and which ones won’t.” Though treatment can be guided by a shared decision-making approach, a personal history of multiple skin cancers is an obvious reason for concern, as is any drug-related or disease-related immunosuppression, he added.
Available treatments for AKs include cryosurgery, curettage, and electrodessication; photodynamic therapy (PDT); and topical agents, including 5-fluorouracil (5-FU), diclofenac, imiquimod, and ingenol mebutate.
“Cryosurgery is what most of us do for most AKs,” said Dr. Pariser, observing that this is a bread-and-butter procedure that is part of the backbone of most dermatology practices. Cryosurgery, curettage, and electrodessication are all used to treat discrete lesions, especially for higher-grade AKs.
However, Dr. Pariser noted that many AKs occur in a “field of cancerization.” This is a larger area of photodamaged skin; the skin may not appear grossly abnormal, but deranged areas will be diffusely evident with fluorescence detection, “indicating that all of this area was damaged,” he said.
PDT and topical agents effectively provide field treatment, and do not just address discrete lesions, although field treatment is technically an off-label use for these modalities, he added.
Lower concentrations and shorter courses of topical agents may be effective and better tolerated by patients, Dr. Pariser said. In clinical trials, for example, the efficacy of imiquimod 2.5% and 3.75% has been comparable to the stronger 5% formulation. Treatment cycles lasting 2 rather than 3 weeks have also shown similar efficacy. The greater palatability of these regimens to patients may increase real-world adherence as well.
Cure rates for PDT range from 67% to 92% for both the blue light–aminolevulinic acid modality and the red light–methyl aminolevulinate more commonly used in Europe. These modalities also treat a broad field, offer cure rates comparable to other topical modalities, and result in less scarring than destructive modalities, Dr. Pariser said.
The pain of PDT may be understandably off-putting for some patients, he commented. In the United States, investigators are experimenting with shorter incubation times for the ALA topical solutions. One study found comparable reductions in the number of AKs by the end of the study period for 1 versus 2 or 3 hours of incubation, and a 2-hour incubation period with spot treatment only was also similarly effective. Clearance rates ranged from about 70%-80%, and all were highly statistically significant, compared with the vehicle-only arm. However, “a second treatment at week 8 seems to be necessary for improved efficacy,” Dr. Pariser said.
Another treatment strategy currently being investigated is “painless PDT,” where the agent is applied and the field is illuminated immediately. Photo-bleaching degrades the protoporphyrins as they are produced, dramatically reducing patient discomfort. Efficacy is promising, he added.
In Europe, “daylight PDT” is being explored – the photosensitizing agent is applied, and the patient simply goes about his or her daily business in normal daylight. This is a nearly painless therapy and shows promise for efficacy. However, said Dr. Pariser, the realities of practice economics make it very unlikely that daylight PDT will catch on in the United States, since the daylight exposure does not represent a billable procedure.
Dr. Pariser reiterated that regardless of the modality, “field therapy has better long-term efficacy rates and better sustained clearance no matter which one of the agents we use, and combination therapy [of lesional and field treatment] seems to be the most effective.”
Dr. Pariser disclosed that he is an investigator and consultant for DUSA Pharmaceuticals, Photocure, and LEO Pharma, and that he discussed off-label uses of drugs and devices.
SDEF and this news organization are owned by the same parent company.
On Twitter @karioakes
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Actinic keratosis: RCM images accord with histopathology
Reflectance Confocal Microscopy (RCM) images of actinic keratoses (AKs) accord well with histopatholgy images, and trained dermatologists can readily distinguish different grades of cytological atypia in the lesions using RCM images alone, according to a report published in the November issue of Journal of the European Academy of Dermatology and Venereology.
RCM is a noninvasive optical technology that enhances clinical diagnostic accuracy, permitting clinicians to assess cytologic and architectural aspects of the epidermis in vivo. Researchers hypothesized that RCM could be used to grade AKs by allowing clinicians to assess “the irregularity of the honeycombed pattern reflecting the architectural disarray of the epidermal spinous layer,” said Dr. Giovanni Pellacani of the department of dermatology, University of Modena and Reggio Emilia, Modena, Italy, and his associates.
In a prospective study, two independent pathologists examined micrographs taken from punch biopsies and RCM images from 48 consecutive cases of facial/scalp AKs collected in a database; they also assessed two benign samples from one young patient with no sun-damaged skin and one elderly patient with severely sun-damaged skin, which served as the controls.
They graded the lesions/samples, classifying 38 as low- or moderate-grade AKs, 10 as high-grade AKs, and the two control samples as normal or lowest-grade sun-damaged skin.
Then three “raters” (two dermatopathologists and Dr. Pellacani) evaluated the RCM images, which showed a representative horizontal section of the epidermis at the stratum spinosum, while the two dermatopathologist raters evaluated photomicrographs taken at histopathology examination, grading the keratinocyte atypia they observed.
RCM grading of keratinocyte atypia strongly correlated among the three RCM raters, and the correlations were strongest for higher-grade lesions. Histopathologic grading also strongly correlated between two pathologist raters. Most important, grading of keratinocytic atypia strongly correlated between these two groups: Raters of the RCM images consistently distinguished different grades of atypia accurately.
These findings show that RCM can allow noninvasive in vivo assessment of keratinocyte morphology that is comparable to that obtained with invasive histopathology, the investigators concluded (J Eur Acad Dermatol Venereol. 2015;29[11]: 2216-21).
“RCM enables one to quickly explore several lesions and skin areas, offering a quasi-histological view of the epidermis useful for the study of AKs. This may lead, in the future, to the use of noninvasive technologies, such as RCM, for studying and monitoring of AKs and other diseases characterized by alterations of keratinocyte morphology thus avoiding the burden of invasive biopsies in clinical trials,” Dr. Pellacani and his associates said.
“Other advantages of noninvasive skin assessments are the possibility to evaluate a larger number of lesions and perilesional areas, and to collect dynamic information on quasi-histological changes occurring during the course of the disease, without alteration induced by the healing process following skin biopsy. This may also facilitate the evaluation of the efficacy of the treatments for AK and field cancerization at cellular level of keratinocytes,” they added.
Reflectance Confocal Microscopy (RCM) images of actinic keratoses (AKs) accord well with histopatholgy images, and trained dermatologists can readily distinguish different grades of cytological atypia in the lesions using RCM images alone, according to a report published in the November issue of Journal of the European Academy of Dermatology and Venereology.
RCM is a noninvasive optical technology that enhances clinical diagnostic accuracy, permitting clinicians to assess cytologic and architectural aspects of the epidermis in vivo. Researchers hypothesized that RCM could be used to grade AKs by allowing clinicians to assess “the irregularity of the honeycombed pattern reflecting the architectural disarray of the epidermal spinous layer,” said Dr. Giovanni Pellacani of the department of dermatology, University of Modena and Reggio Emilia, Modena, Italy, and his associates.
In a prospective study, two independent pathologists examined micrographs taken from punch biopsies and RCM images from 48 consecutive cases of facial/scalp AKs collected in a database; they also assessed two benign samples from one young patient with no sun-damaged skin and one elderly patient with severely sun-damaged skin, which served as the controls.
They graded the lesions/samples, classifying 38 as low- or moderate-grade AKs, 10 as high-grade AKs, and the two control samples as normal or lowest-grade sun-damaged skin.
Then three “raters” (two dermatopathologists and Dr. Pellacani) evaluated the RCM images, which showed a representative horizontal section of the epidermis at the stratum spinosum, while the two dermatopathologist raters evaluated photomicrographs taken at histopathology examination, grading the keratinocyte atypia they observed.
RCM grading of keratinocyte atypia strongly correlated among the three RCM raters, and the correlations were strongest for higher-grade lesions. Histopathologic grading also strongly correlated between two pathologist raters. Most important, grading of keratinocytic atypia strongly correlated between these two groups: Raters of the RCM images consistently distinguished different grades of atypia accurately.
These findings show that RCM can allow noninvasive in vivo assessment of keratinocyte morphology that is comparable to that obtained with invasive histopathology, the investigators concluded (J Eur Acad Dermatol Venereol. 2015;29[11]: 2216-21).
“RCM enables one to quickly explore several lesions and skin areas, offering a quasi-histological view of the epidermis useful for the study of AKs. This may lead, in the future, to the use of noninvasive technologies, such as RCM, for studying and monitoring of AKs and other diseases characterized by alterations of keratinocyte morphology thus avoiding the burden of invasive biopsies in clinical trials,” Dr. Pellacani and his associates said.
“Other advantages of noninvasive skin assessments are the possibility to evaluate a larger number of lesions and perilesional areas, and to collect dynamic information on quasi-histological changes occurring during the course of the disease, without alteration induced by the healing process following skin biopsy. This may also facilitate the evaluation of the efficacy of the treatments for AK and field cancerization at cellular level of keratinocytes,” they added.
Reflectance Confocal Microscopy (RCM) images of actinic keratoses (AKs) accord well with histopatholgy images, and trained dermatologists can readily distinguish different grades of cytological atypia in the lesions using RCM images alone, according to a report published in the November issue of Journal of the European Academy of Dermatology and Venereology.
RCM is a noninvasive optical technology that enhances clinical diagnostic accuracy, permitting clinicians to assess cytologic and architectural aspects of the epidermis in vivo. Researchers hypothesized that RCM could be used to grade AKs by allowing clinicians to assess “the irregularity of the honeycombed pattern reflecting the architectural disarray of the epidermal spinous layer,” said Dr. Giovanni Pellacani of the department of dermatology, University of Modena and Reggio Emilia, Modena, Italy, and his associates.
In a prospective study, two independent pathologists examined micrographs taken from punch biopsies and RCM images from 48 consecutive cases of facial/scalp AKs collected in a database; they also assessed two benign samples from one young patient with no sun-damaged skin and one elderly patient with severely sun-damaged skin, which served as the controls.
They graded the lesions/samples, classifying 38 as low- or moderate-grade AKs, 10 as high-grade AKs, and the two control samples as normal or lowest-grade sun-damaged skin.
Then three “raters” (two dermatopathologists and Dr. Pellacani) evaluated the RCM images, which showed a representative horizontal section of the epidermis at the stratum spinosum, while the two dermatopathologist raters evaluated photomicrographs taken at histopathology examination, grading the keratinocyte atypia they observed.
RCM grading of keratinocyte atypia strongly correlated among the three RCM raters, and the correlations were strongest for higher-grade lesions. Histopathologic grading also strongly correlated between two pathologist raters. Most important, grading of keratinocytic atypia strongly correlated between these two groups: Raters of the RCM images consistently distinguished different grades of atypia accurately.
These findings show that RCM can allow noninvasive in vivo assessment of keratinocyte morphology that is comparable to that obtained with invasive histopathology, the investigators concluded (J Eur Acad Dermatol Venereol. 2015;29[11]: 2216-21).
“RCM enables one to quickly explore several lesions and skin areas, offering a quasi-histological view of the epidermis useful for the study of AKs. This may lead, in the future, to the use of noninvasive technologies, such as RCM, for studying and monitoring of AKs and other diseases characterized by alterations of keratinocyte morphology thus avoiding the burden of invasive biopsies in clinical trials,” Dr. Pellacani and his associates said.
“Other advantages of noninvasive skin assessments are the possibility to evaluate a larger number of lesions and perilesional areas, and to collect dynamic information on quasi-histological changes occurring during the course of the disease, without alteration induced by the healing process following skin biopsy. This may also facilitate the evaluation of the efficacy of the treatments for AK and field cancerization at cellular level of keratinocytes,” they added.
FROM THE JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Key clinical point: Reflectance confocal microscopy images of actinic keratoses accord well with histopathology images, allowing noninvasive assessment of the lesions.
Major finding: The grading of keratinocytic atypia strongly correlated between three clinicians who rated the RCM images and two who rated the histopathology micrographs.
Data source: The study compared results of expert assessments of 48 actinic keratoses using either RCM images or histopathology slides.
Disclosures: This study had no sponsor or funding source. Dr. Pellacani and his associates reported having no relevant financial disclosures.
ILDS establishes guidelines for treating AK patients
The International Leagues of Dermatological Societies (ILDS) in cooperation with the European Dermatology Forum has developed consensus-based guidelines for the treatment of actinic keratosis (AK), which are published in the Journal of the European Academy of Dermatology and Venereology.
“The guidelines were elaborated along adapted recommendations by the WHO guidelines review committee and the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) working group,” say R. N. Werner and colleagues of the Medical University of Berlin. The guidelines include recommendations for treatment of different subgroups of AK patients, how to make an AK diagnosis, how to assess AK patients, and how to define AK.
The ILDS recommends or suggests the following interventions for treating patients who have single AK lesions:
• Cryotherapy
• Curettage (discrete, hyperkeratotic lesions)
• 0.5% 5-fluorouracil (5-FU)
• 5% 5-FU
• 0.5% 5-FU + 10% salicylic acid (discrete, hyperkeratotic lesions)
• 3.75% imiquimod
• 5% imiquimod
• Ingenol mebutate 0.015% (lesions on the face or scalp) and ingenol mebutate 0.05% (lesions on the trunk or extremities)
• 5-aminolevulinic acid-photodynamic therapy (ALA-PDT)
• methylaminolevulinate-photodynamic therapy (MAL-PDT)
For patients with multiple AK lesions/field cancerization, the ILDS recommends* or suggests that patients use the following therapies:
• 0.5% 5-FU*
• 3.75% imiquimod*
• Ingenol mebutate 0.015% (lesions on the face or scalp) and ingenol mebutate 0.05% (lesions on the trunk or extremities)*
• ALA-PDT*
• MAL-PDT*
• Cryotherapy (patients with multiple lesions, especially for multiple discrete lesions; not suitable for the treatment of field cancerization)
• 3% diclofenac in 2.5% hyaluronic acid gel
• 5% 5-FU
• 0.5% 5-FU + 10% salicylic acid (discrete, hyperkeratotic lesions)
• 5% imiquimod
• 2.5% imiquimod
• CO2 laser and Er:YAG laser
For immunosuppressed AK patients, the ILDS suggests the following treatments:
• Cryotherapy (especially for single lesions or multiple discrete lesions; not suitable for the treatment of field cancerization);
• Curettage (discrete, hyperkeratotic lesions)
• 5% 5-FU
• 5% imiquimod
• ALA-PDT
• MAL-PDT
The ILDS additionally recommends that immunosuppressed AK patients not use CO2 laser and Er:YAG laser.
“Deviation from the recommendations may be justified or inevitable in specific situations. The ultimate judgment regarding patient care must be individualized and must be made by the physician and patient in light of all presenting circumstances,” the authors said. “International guidelines are intended to be adapted to national or regional circumstances” (J Eur Acad Dermatol Venereol. 2015;29:2069-79).
The “long version of the guidelines” is available as an online supplement. Additionally, a methods report, results report, and declarations of interest of the guidelines development have been published at doi: 10.1111/jdv.13179 in the Journal of the European Academy of Dermatology and Venereology (2015).
The International Leagues of Dermatological Societies (ILDS) in cooperation with the European Dermatology Forum has developed consensus-based guidelines for the treatment of actinic keratosis (AK), which are published in the Journal of the European Academy of Dermatology and Venereology.
“The guidelines were elaborated along adapted recommendations by the WHO guidelines review committee and the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) working group,” say R. N. Werner and colleagues of the Medical University of Berlin. The guidelines include recommendations for treatment of different subgroups of AK patients, how to make an AK diagnosis, how to assess AK patients, and how to define AK.
The ILDS recommends or suggests the following interventions for treating patients who have single AK lesions:
• Cryotherapy
• Curettage (discrete, hyperkeratotic lesions)
• 0.5% 5-fluorouracil (5-FU)
• 5% 5-FU
• 0.5% 5-FU + 10% salicylic acid (discrete, hyperkeratotic lesions)
• 3.75% imiquimod
• 5% imiquimod
• Ingenol mebutate 0.015% (lesions on the face or scalp) and ingenol mebutate 0.05% (lesions on the trunk or extremities)
• 5-aminolevulinic acid-photodynamic therapy (ALA-PDT)
• methylaminolevulinate-photodynamic therapy (MAL-PDT)
For patients with multiple AK lesions/field cancerization, the ILDS recommends* or suggests that patients use the following therapies:
• 0.5% 5-FU*
• 3.75% imiquimod*
• Ingenol mebutate 0.015% (lesions on the face or scalp) and ingenol mebutate 0.05% (lesions on the trunk or extremities)*
• ALA-PDT*
• MAL-PDT*
• Cryotherapy (patients with multiple lesions, especially for multiple discrete lesions; not suitable for the treatment of field cancerization)
• 3% diclofenac in 2.5% hyaluronic acid gel
• 5% 5-FU
• 0.5% 5-FU + 10% salicylic acid (discrete, hyperkeratotic lesions)
• 5% imiquimod
• 2.5% imiquimod
• CO2 laser and Er:YAG laser
For immunosuppressed AK patients, the ILDS suggests the following treatments:
• Cryotherapy (especially for single lesions or multiple discrete lesions; not suitable for the treatment of field cancerization);
• Curettage (discrete, hyperkeratotic lesions)
• 5% 5-FU
• 5% imiquimod
• ALA-PDT
• MAL-PDT
The ILDS additionally recommends that immunosuppressed AK patients not use CO2 laser and Er:YAG laser.
“Deviation from the recommendations may be justified or inevitable in specific situations. The ultimate judgment regarding patient care must be individualized and must be made by the physician and patient in light of all presenting circumstances,” the authors said. “International guidelines are intended to be adapted to national or regional circumstances” (J Eur Acad Dermatol Venereol. 2015;29:2069-79).
The “long version of the guidelines” is available as an online supplement. Additionally, a methods report, results report, and declarations of interest of the guidelines development have been published at doi: 10.1111/jdv.13179 in the Journal of the European Academy of Dermatology and Venereology (2015).
The International Leagues of Dermatological Societies (ILDS) in cooperation with the European Dermatology Forum has developed consensus-based guidelines for the treatment of actinic keratosis (AK), which are published in the Journal of the European Academy of Dermatology and Venereology.
“The guidelines were elaborated along adapted recommendations by the WHO guidelines review committee and the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) working group,” say R. N. Werner and colleagues of the Medical University of Berlin. The guidelines include recommendations for treatment of different subgroups of AK patients, how to make an AK diagnosis, how to assess AK patients, and how to define AK.
The ILDS recommends or suggests the following interventions for treating patients who have single AK lesions:
• Cryotherapy
• Curettage (discrete, hyperkeratotic lesions)
• 0.5% 5-fluorouracil (5-FU)
• 5% 5-FU
• 0.5% 5-FU + 10% salicylic acid (discrete, hyperkeratotic lesions)
• 3.75% imiquimod
• 5% imiquimod
• Ingenol mebutate 0.015% (lesions on the face or scalp) and ingenol mebutate 0.05% (lesions on the trunk or extremities)
• 5-aminolevulinic acid-photodynamic therapy (ALA-PDT)
• methylaminolevulinate-photodynamic therapy (MAL-PDT)
For patients with multiple AK lesions/field cancerization, the ILDS recommends* or suggests that patients use the following therapies:
• 0.5% 5-FU*
• 3.75% imiquimod*
• Ingenol mebutate 0.015% (lesions on the face or scalp) and ingenol mebutate 0.05% (lesions on the trunk or extremities)*
• ALA-PDT*
• MAL-PDT*
• Cryotherapy (patients with multiple lesions, especially for multiple discrete lesions; not suitable for the treatment of field cancerization)
• 3% diclofenac in 2.5% hyaluronic acid gel
• 5% 5-FU
• 0.5% 5-FU + 10% salicylic acid (discrete, hyperkeratotic lesions)
• 5% imiquimod
• 2.5% imiquimod
• CO2 laser and Er:YAG laser
For immunosuppressed AK patients, the ILDS suggests the following treatments:
• Cryotherapy (especially for single lesions or multiple discrete lesions; not suitable for the treatment of field cancerization);
• Curettage (discrete, hyperkeratotic lesions)
• 5% 5-FU
• 5% imiquimod
• ALA-PDT
• MAL-PDT
The ILDS additionally recommends that immunosuppressed AK patients not use CO2 laser and Er:YAG laser.
“Deviation from the recommendations may be justified or inevitable in specific situations. The ultimate judgment regarding patient care must be individualized and must be made by the physician and patient in light of all presenting circumstances,” the authors said. “International guidelines are intended to be adapted to national or regional circumstances” (J Eur Acad Dermatol Venereol. 2015;29:2069-79).
The “long version of the guidelines” is available as an online supplement. Additionally, a methods report, results report, and declarations of interest of the guidelines development have been published at doi: 10.1111/jdv.13179 in the Journal of the European Academy of Dermatology and Venereology (2015).
FROM JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Simultaneous equal to sequential treatment for actinic keratoses
Patients with multiple actinic keratoses (AKs) may be treated either sequentially or simultaneously with ingenol mebutate gel, according to the authors of a study that found no difference in outcomes or adverse effects from either treatment approach.
The phase IIIb study conducted in Italy and Spain enrolled 199 patients with two separate areas of clinically visible, nonhyperkeratotic AKs. Subjects were randomized to have the two areas (face/scalp and trunk/extremities) treated simultaneously (101 patients) or sequentially (98 patients) with 0.015% and 0.05% ingenol mebutate gel.
There were no significant differences in localized skin responses between the simultaneous and sequential treatment groups, based on the mean composite local skin response scores 3 days after treatment started, which were similar between the groups for the face/scalp and trunk/extremities applications. About 22% of patients in each group experienced adverse events.
At 8 weeks, the complete clearance rates also were not statistically different between the simultaneous and sequential groups (52.7% and 46.9%, respectively; P = .34), and patient satisfaction with treatment was similar for both treatment approaches. At that time, the number of AKs had dropped by a mean of 83.4% among those in the simultaneous group and 79.1% in the sequential group (P = .20).
“The favorable rate of complete clearance in the simultaneous treatment group means that patients can receive their treatment for both areas in one visit, rather than having to return to the clinic for a second cycle of treatment,” wrote Dr. Giovanni Pellacani of the department of dermatology at the University of Modena (Italy) and Reggio Emilia, and his coauthors (J Eur Acad Dermatol Venereol. 2015;29[11]:2192-8).
“Ultimately, the treatment schedule is based on agreement between the physician and the patient; this study helps to support the selection of the most appropriate regimen to treat AK in individual patients,” they commented.
The study was funded by LEO Pharma, the manufacturer of ingenol mebutate gel (Picato). Dr. Pellacani has received consultant fees from the company; three authors are employees of the company; and the other authors declared consultancies, honoraria and, grants from LEO Pharma and other pharmaceutical companies.
Patients with multiple actinic keratoses (AKs) may be treated either sequentially or simultaneously with ingenol mebutate gel, according to the authors of a study that found no difference in outcomes or adverse effects from either treatment approach.
The phase IIIb study conducted in Italy and Spain enrolled 199 patients with two separate areas of clinically visible, nonhyperkeratotic AKs. Subjects were randomized to have the two areas (face/scalp and trunk/extremities) treated simultaneously (101 patients) or sequentially (98 patients) with 0.015% and 0.05% ingenol mebutate gel.
There were no significant differences in localized skin responses between the simultaneous and sequential treatment groups, based on the mean composite local skin response scores 3 days after treatment started, which were similar between the groups for the face/scalp and trunk/extremities applications. About 22% of patients in each group experienced adverse events.
At 8 weeks, the complete clearance rates also were not statistically different between the simultaneous and sequential groups (52.7% and 46.9%, respectively; P = .34), and patient satisfaction with treatment was similar for both treatment approaches. At that time, the number of AKs had dropped by a mean of 83.4% among those in the simultaneous group and 79.1% in the sequential group (P = .20).
“The favorable rate of complete clearance in the simultaneous treatment group means that patients can receive their treatment for both areas in one visit, rather than having to return to the clinic for a second cycle of treatment,” wrote Dr. Giovanni Pellacani of the department of dermatology at the University of Modena (Italy) and Reggio Emilia, and his coauthors (J Eur Acad Dermatol Venereol. 2015;29[11]:2192-8).
“Ultimately, the treatment schedule is based on agreement between the physician and the patient; this study helps to support the selection of the most appropriate regimen to treat AK in individual patients,” they commented.
The study was funded by LEO Pharma, the manufacturer of ingenol mebutate gel (Picato). Dr. Pellacani has received consultant fees from the company; three authors are employees of the company; and the other authors declared consultancies, honoraria and, grants from LEO Pharma and other pharmaceutical companies.
Patients with multiple actinic keratoses (AKs) may be treated either sequentially or simultaneously with ingenol mebutate gel, according to the authors of a study that found no difference in outcomes or adverse effects from either treatment approach.
The phase IIIb study conducted in Italy and Spain enrolled 199 patients with two separate areas of clinically visible, nonhyperkeratotic AKs. Subjects were randomized to have the two areas (face/scalp and trunk/extremities) treated simultaneously (101 patients) or sequentially (98 patients) with 0.015% and 0.05% ingenol mebutate gel.
There were no significant differences in localized skin responses between the simultaneous and sequential treatment groups, based on the mean composite local skin response scores 3 days after treatment started, which were similar between the groups for the face/scalp and trunk/extremities applications. About 22% of patients in each group experienced adverse events.
At 8 weeks, the complete clearance rates also were not statistically different between the simultaneous and sequential groups (52.7% and 46.9%, respectively; P = .34), and patient satisfaction with treatment was similar for both treatment approaches. At that time, the number of AKs had dropped by a mean of 83.4% among those in the simultaneous group and 79.1% in the sequential group (P = .20).
“The favorable rate of complete clearance in the simultaneous treatment group means that patients can receive their treatment for both areas in one visit, rather than having to return to the clinic for a second cycle of treatment,” wrote Dr. Giovanni Pellacani of the department of dermatology at the University of Modena (Italy) and Reggio Emilia, and his coauthors (J Eur Acad Dermatol Venereol. 2015;29[11]:2192-8).
“Ultimately, the treatment schedule is based on agreement between the physician and the patient; this study helps to support the selection of the most appropriate regimen to treat AK in individual patients,” they commented.
The study was funded by LEO Pharma, the manufacturer of ingenol mebutate gel (Picato). Dr. Pellacani has received consultant fees from the company; three authors are employees of the company; and the other authors declared consultancies, honoraria and, grants from LEO Pharma and other pharmaceutical companies.
FROM THE JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Key clinical point: Patients with multiple AKs may be treated either sequentially or simultaneously with ingenol mebutate gel, with similar efficacy and safety outcomes.
Major finding: The incidence of localized skin responses, complete clearance rates, and patient treatment satisfaction were similar for simultaneous and sequential treatment approaches.
Data source: A phase IIIb randomized, multicenter, open-label, parallel-group study evaluated 199 patients with two separate areas of clinically visible, nonhyperkeratotic AKs.
Disclosures: The study was funded by ingenol mebutate gel manufacturer LEO Pharma. Three authors are employees of the company; the other authors declared consultancies, honoraria, and/or grants from LEO Pharma and other pharmaceutical companies.
EADV: Prophylactic photodynamic therapy benefits transplant recipients
COPENHAGEN – Twice-yearly prophylactic photodynamic therapy for primary prevention of actinic keratoses and squamous cell carcinomas is a novel and effective strategy that addresses the problem of accelerated photocarcinogenesis in organ transplant recipients, according to an interim analysis of a multinational, randomized, controlled trial.
“The overall aim is to prevent squamous cell carcinoma development. Photodynamic therapy is well established for secondary prevention of further AKs, and these very early data show that it can also be used for primary prevention in very high-risk patients,” Dr. Katrine Togsverd-Bo said at the annual congress of the European Academy of Dermatology and Venereology.
Accelerated carcinogenesis on sun-exposed skin is a major concern in organ transplant recipients (OTRs). They experience early onset of multiple AKs, with field cancerization and up to a 100-fold increased risk of squamous cell carcinomas (SCCs). Moreover, their SCCs are at substantially greater risk of metastasis than SCCs occurring in the general population, noted Dr. Togsverd-Bo of Bispebjerg Hospital and the University of Copenhagen.
She presented an interim analysis of an ongoing 5-year prospective randomized trial in 50 renal transplant recipients at academic dermatology centers in Copenhagen, Oslo, and Gothenburg, Sweden. All participants had clinically normal-appearing skin at baseline, with no history of AKs or SCCs. They are undergoing twice-yearly, split-side photodynamic therapy (PDT) on the face, forearm, and hand, with the opposite side serving as the untreated control.
To date, 25 patients have completed 3 years of the study. At 3 years of prospective follow-up by blinded evaluators, 50% of patients had AKs on their untreated side, compared with 26% on the prophylactic PDT side. The collective number of AKs on untreated skin was 43, compared with just 8 AKs on PDT-treated skin. Seven patients had AKs only on their untreated side, six had AKs on both sides, and none had any AKs only on their PDT-treated side.
The twice-yearly prophylactic PDT regimen consists of a 3-hour application of 20% methyl aminolevulinate as a photosensitizer followed by applications of a conventional LED light at 37 J/cm2.
Dr. Togsverd-Bo reported having no financial conflicts regarding her study.
COPENHAGEN – Twice-yearly prophylactic photodynamic therapy for primary prevention of actinic keratoses and squamous cell carcinomas is a novel and effective strategy that addresses the problem of accelerated photocarcinogenesis in organ transplant recipients, according to an interim analysis of a multinational, randomized, controlled trial.
“The overall aim is to prevent squamous cell carcinoma development. Photodynamic therapy is well established for secondary prevention of further AKs, and these very early data show that it can also be used for primary prevention in very high-risk patients,” Dr. Katrine Togsverd-Bo said at the annual congress of the European Academy of Dermatology and Venereology.
Accelerated carcinogenesis on sun-exposed skin is a major concern in organ transplant recipients (OTRs). They experience early onset of multiple AKs, with field cancerization and up to a 100-fold increased risk of squamous cell carcinomas (SCCs). Moreover, their SCCs are at substantially greater risk of metastasis than SCCs occurring in the general population, noted Dr. Togsverd-Bo of Bispebjerg Hospital and the University of Copenhagen.
She presented an interim analysis of an ongoing 5-year prospective randomized trial in 50 renal transplant recipients at academic dermatology centers in Copenhagen, Oslo, and Gothenburg, Sweden. All participants had clinically normal-appearing skin at baseline, with no history of AKs or SCCs. They are undergoing twice-yearly, split-side photodynamic therapy (PDT) on the face, forearm, and hand, with the opposite side serving as the untreated control.
To date, 25 patients have completed 3 years of the study. At 3 years of prospective follow-up by blinded evaluators, 50% of patients had AKs on their untreated side, compared with 26% on the prophylactic PDT side. The collective number of AKs on untreated skin was 43, compared with just 8 AKs on PDT-treated skin. Seven patients had AKs only on their untreated side, six had AKs on both sides, and none had any AKs only on their PDT-treated side.
The twice-yearly prophylactic PDT regimen consists of a 3-hour application of 20% methyl aminolevulinate as a photosensitizer followed by applications of a conventional LED light at 37 J/cm2.
Dr. Togsverd-Bo reported having no financial conflicts regarding her study.
COPENHAGEN – Twice-yearly prophylactic photodynamic therapy for primary prevention of actinic keratoses and squamous cell carcinomas is a novel and effective strategy that addresses the problem of accelerated photocarcinogenesis in organ transplant recipients, according to an interim analysis of a multinational, randomized, controlled trial.
“The overall aim is to prevent squamous cell carcinoma development. Photodynamic therapy is well established for secondary prevention of further AKs, and these very early data show that it can also be used for primary prevention in very high-risk patients,” Dr. Katrine Togsverd-Bo said at the annual congress of the European Academy of Dermatology and Venereology.
Accelerated carcinogenesis on sun-exposed skin is a major concern in organ transplant recipients (OTRs). They experience early onset of multiple AKs, with field cancerization and up to a 100-fold increased risk of squamous cell carcinomas (SCCs). Moreover, their SCCs are at substantially greater risk of metastasis than SCCs occurring in the general population, noted Dr. Togsverd-Bo of Bispebjerg Hospital and the University of Copenhagen.
She presented an interim analysis of an ongoing 5-year prospective randomized trial in 50 renal transplant recipients at academic dermatology centers in Copenhagen, Oslo, and Gothenburg, Sweden. All participants had clinically normal-appearing skin at baseline, with no history of AKs or SCCs. They are undergoing twice-yearly, split-side photodynamic therapy (PDT) on the face, forearm, and hand, with the opposite side serving as the untreated control.
To date, 25 patients have completed 3 years of the study. At 3 years of prospective follow-up by blinded evaluators, 50% of patients had AKs on their untreated side, compared with 26% on the prophylactic PDT side. The collective number of AKs on untreated skin was 43, compared with just 8 AKs on PDT-treated skin. Seven patients had AKs only on their untreated side, six had AKs on both sides, and none had any AKs only on their PDT-treated side.
The twice-yearly prophylactic PDT regimen consists of a 3-hour application of 20% methyl aminolevulinate as a photosensitizer followed by applications of a conventional LED light at 37 J/cm2.
Dr. Togsverd-Bo reported having no financial conflicts regarding her study.
AT THE EADV CONGRESS
Key clinical point: Prophylactic photodynamic therapy is a new and effective strategy for primary prevention of actinic keratoses and squamous cell carcinomas in organ transplant recipients.
Major finding: At 3 years of follow-up, 25 renal transplant recipients collectively had 8 actinic keratoses on the side of their face, forearms, and hands treated with twice-yearly prophylactic photodynamic therapy, compared with 43 AKs on the untreated control side.
Data source: This is an interim 3-year analysis from an ongoing 5-year prospective multinational, randomized, controlled trial involving 50 renal transplant recipients.
Disclosures: The presenter reported having no financial conflicts regarding this ongoing study.
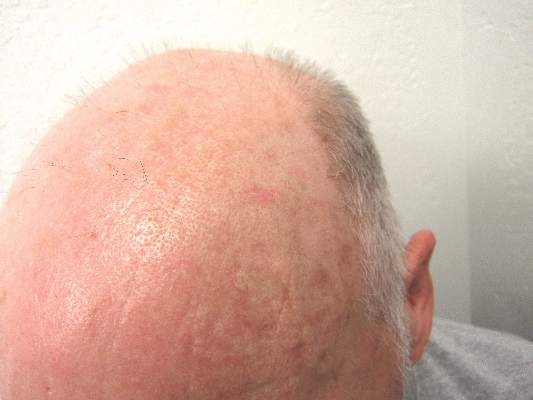
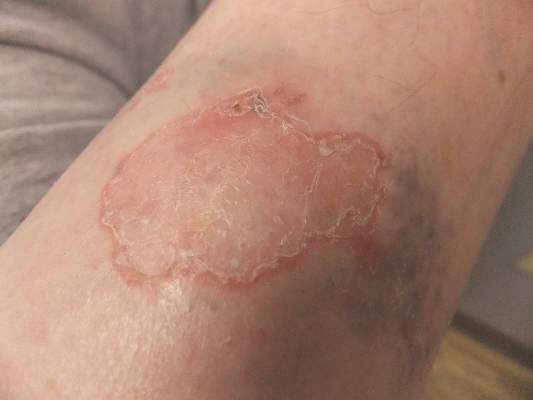
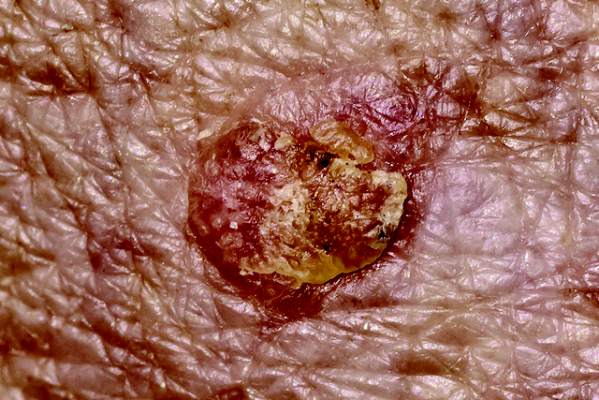
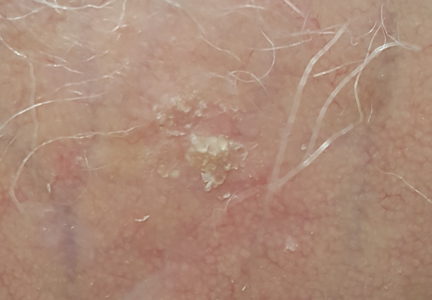
Solitary Lesion on the Left Ankle
The Diagnosis: Porokeratosis of Mibelli
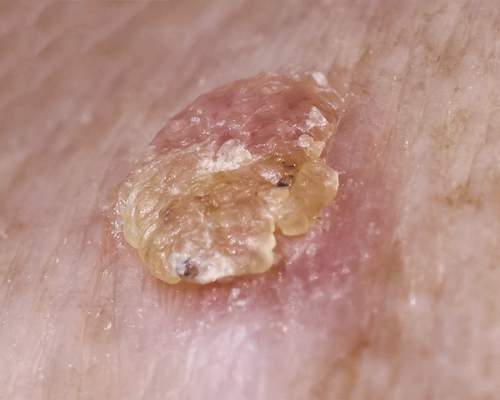
There are 5 variants of porokeratosis: disseminated superficial actinic porokeratosis (DSAP), linear porokeratosis, porokeratosis of Mibelli, porokeratosis palmaris et plantaris disseminata, and punctate porokeratosis. The most common type is DSAP,1 which is characterized by multiple lesions on the body, particularly in sun-exposed areas. The distinguishing feature of porokeratosis is the cornoid lamella, which is made up of parakeratotic cells extending through the stratum corneum. There also is a thin or absent granular layer beneath it (Figure).2
Patients generally present in the third and fourth decades of life.1 Risk factors for porokeratosis include sun exposure, immunosuppression, and genetics.2-4 Overexpression of the protein p53 in porokeratosis lesions has been demonstrated in studies investigating the genetics of porokeratosis.5,6 A study of Chinese families with DSAP identified 3 different loci associated with DSAP: DSAP1, DSAP2, and DSAP3.2 The progression to cancer has been noted in all types of porokeratosis lesions. Malignancies include squamous cell carcinoma, Bowen disease, and basal cell carcinoma.7,8
Many treatments have been tried for DSAP including cryotherapy, topical 5-fluorouracil, photodynamic therapy, and topical imiquimod with varying success.1 Our patient was treated with cryotherapy but had side effects from treatment including cellulitis and local infections with ulceration before finally healing.
Interestingly, our patient had a single lesion with pathology findings most consistent with DSAP at a later age. Although the pathology suggested DSAP, the size and solitary lesion was more consistent with porokeratosis of Mibelli. Porokeratosis of Mibelli can occur concurrently with DSAP,9 but we have not seen other lesions in this patient. We have educated our patient to be aware of other lesions that may occur in the future. Due to risk for malignant conversion, it is generally viewed as beneficial to treat patients who present with porokeratosis lesions. Our patient’s lesion ultimately cleared and he has not developed new lesions at 1-year follow-up.
Although DSAP generally presents in the third and fourth decades of life and porokeratosis of Mibelli during childhood, it is important to educate both dermatologists and primary care physicians to be aware of the possibility of both diagnoses in the elderly population.
- Rouhani P, Fischer M, Meehan S, et al. Disseminated superficial actinic porokeratosis. Dermatol Online J. 2012;18:24.
- Murase J, Gilliam AC, et al. Disseminated superficial actinic porokeratosis co-existing with linear and verrucous porokeratosis in an elderly woman: update on the genetics and clinical expression of porokeratosis. J Am Acad Dermatol. 2010;63:886-891.
- Lederman JS, Sober AJ, Lederman GS. Immunosuppression: a cause of porokeratosis? J Am Acad Dermatol. 1985;13:75-79.
- Hernandez MH, Lai CH, Mallory SB. Disseminated porokeratosis associated with chronic renal failure: a new type of disseminated porokeratosis? Arch Dermatol. 2000;136:1568-1569.
- Magee JW, McCalmont TH, LeBoit PE. Overexpression of p53 tumor suppressor protein in porokeratosis. Arch Dermatol. 1994;130:187-190.
- Arranz-Salas I, Sanz-Trelles A, Ojeda DB. p53 alterations in porokeratosis. J Cutan Pathol. 2003;30:455-458.
- Curnow P, Foley P, Baker C. Multiple squamous cell carcinomas complicating linear porokeratosis. Australas J Dermatol. 2003;44:136-139.
- Lee HR, Han TY, Son SJ, et al. Squamous cell carcinoma developing within lesions of disseminated superficial actinic porokeratosis. Ann Dermatol. 2011;23:536-538.
- Mehta V, Balachandran C. Simultaneous co-occurrence of porokeratosis of Mibelli with disseminated superficial actinic porokeratosis. Indian J Dermatol. 2009;54:390-391.
The Diagnosis: Porokeratosis of Mibelli
There are 5 variants of porokeratosis: disseminated superficial actinic porokeratosis (DSAP), linear porokeratosis, porokeratosis of Mibelli, porokeratosis palmaris et plantaris disseminata, and punctate porokeratosis. The most common type is DSAP,1 which is characterized by multiple lesions on the body, particularly in sun-exposed areas. The distinguishing feature of porokeratosis is the cornoid lamella, which is made up of parakeratotic cells extending through the stratum corneum. There also is a thin or absent granular layer beneath it (Figure).2
Patients generally present in the third and fourth decades of life.1 Risk factors for porokeratosis include sun exposure, immunosuppression, and genetics.2-4 Overexpression of the protein p53 in porokeratosis lesions has been demonstrated in studies investigating the genetics of porokeratosis.5,6 A study of Chinese families with DSAP identified 3 different loci associated with DSAP: DSAP1, DSAP2, and DSAP3.2 The progression to cancer has been noted in all types of porokeratosis lesions. Malignancies include squamous cell carcinoma, Bowen disease, and basal cell carcinoma.7,8
Many treatments have been tried for DSAP including cryotherapy, topical 5-fluorouracil, photodynamic therapy, and topical imiquimod with varying success.1 Our patient was treated with cryotherapy but had side effects from treatment including cellulitis and local infections with ulceration before finally healing.
Interestingly, our patient had a single lesion with pathology findings most consistent with DSAP at a later age. Although the pathology suggested DSAP, the size and solitary lesion was more consistent with porokeratosis of Mibelli. Porokeratosis of Mibelli can occur concurrently with DSAP,9 but we have not seen other lesions in this patient. We have educated our patient to be aware of other lesions that may occur in the future. Due to risk for malignant conversion, it is generally viewed as beneficial to treat patients who present with porokeratosis lesions. Our patient’s lesion ultimately cleared and he has not developed new lesions at 1-year follow-up.
Although DSAP generally presents in the third and fourth decades of life and porokeratosis of Mibelli during childhood, it is important to educate both dermatologists and primary care physicians to be aware of the possibility of both diagnoses in the elderly population.
The Diagnosis: Porokeratosis of Mibelli
There are 5 variants of porokeratosis: disseminated superficial actinic porokeratosis (DSAP), linear porokeratosis, porokeratosis of Mibelli, porokeratosis palmaris et plantaris disseminata, and punctate porokeratosis. The most common type is DSAP,1 which is characterized by multiple lesions on the body, particularly in sun-exposed areas. The distinguishing feature of porokeratosis is the cornoid lamella, which is made up of parakeratotic cells extending through the stratum corneum. There also is a thin or absent granular layer beneath it (Figure).2
Patients generally present in the third and fourth decades of life.1 Risk factors for porokeratosis include sun exposure, immunosuppression, and genetics.2-4 Overexpression of the protein p53 in porokeratosis lesions has been demonstrated in studies investigating the genetics of porokeratosis.5,6 A study of Chinese families with DSAP identified 3 different loci associated with DSAP: DSAP1, DSAP2, and DSAP3.2 The progression to cancer has been noted in all types of porokeratosis lesions. Malignancies include squamous cell carcinoma, Bowen disease, and basal cell carcinoma.7,8
Many treatments have been tried for DSAP including cryotherapy, topical 5-fluorouracil, photodynamic therapy, and topical imiquimod with varying success.1 Our patient was treated with cryotherapy but had side effects from treatment including cellulitis and local infections with ulceration before finally healing.
Interestingly, our patient had a single lesion with pathology findings most consistent with DSAP at a later age. Although the pathology suggested DSAP, the size and solitary lesion was more consistent with porokeratosis of Mibelli. Porokeratosis of Mibelli can occur concurrently with DSAP,9 but we have not seen other lesions in this patient. We have educated our patient to be aware of other lesions that may occur in the future. Due to risk for malignant conversion, it is generally viewed as beneficial to treat patients who present with porokeratosis lesions. Our patient’s lesion ultimately cleared and he has not developed new lesions at 1-year follow-up.
Although DSAP generally presents in the third and fourth decades of life and porokeratosis of Mibelli during childhood, it is important to educate both dermatologists and primary care physicians to be aware of the possibility of both diagnoses in the elderly population.
- Rouhani P, Fischer M, Meehan S, et al. Disseminated superficial actinic porokeratosis. Dermatol Online J. 2012;18:24.
- Murase J, Gilliam AC, et al. Disseminated superficial actinic porokeratosis co-existing with linear and verrucous porokeratosis in an elderly woman: update on the genetics and clinical expression of porokeratosis. J Am Acad Dermatol. 2010;63:886-891.
- Lederman JS, Sober AJ, Lederman GS. Immunosuppression: a cause of porokeratosis? J Am Acad Dermatol. 1985;13:75-79.
- Hernandez MH, Lai CH, Mallory SB. Disseminated porokeratosis associated with chronic renal failure: a new type of disseminated porokeratosis? Arch Dermatol. 2000;136:1568-1569.
- Magee JW, McCalmont TH, LeBoit PE. Overexpression of p53 tumor suppressor protein in porokeratosis. Arch Dermatol. 1994;130:187-190.
- Arranz-Salas I, Sanz-Trelles A, Ojeda DB. p53 alterations in porokeratosis. J Cutan Pathol. 2003;30:455-458.
- Curnow P, Foley P, Baker C. Multiple squamous cell carcinomas complicating linear porokeratosis. Australas J Dermatol. 2003;44:136-139.
- Lee HR, Han TY, Son SJ, et al. Squamous cell carcinoma developing within lesions of disseminated superficial actinic porokeratosis. Ann Dermatol. 2011;23:536-538.
- Mehta V, Balachandran C. Simultaneous co-occurrence of porokeratosis of Mibelli with disseminated superficial actinic porokeratosis. Indian J Dermatol. 2009;54:390-391.
- Rouhani P, Fischer M, Meehan S, et al. Disseminated superficial actinic porokeratosis. Dermatol Online J. 2012;18:24.
- Murase J, Gilliam AC, et al. Disseminated superficial actinic porokeratosis co-existing with linear and verrucous porokeratosis in an elderly woman: update on the genetics and clinical expression of porokeratosis. J Am Acad Dermatol. 2010;63:886-891.
- Lederman JS, Sober AJ, Lederman GS. Immunosuppression: a cause of porokeratosis? J Am Acad Dermatol. 1985;13:75-79.
- Hernandez MH, Lai CH, Mallory SB. Disseminated porokeratosis associated with chronic renal failure: a new type of disseminated porokeratosis? Arch Dermatol. 2000;136:1568-1569.
- Magee JW, McCalmont TH, LeBoit PE. Overexpression of p53 tumor suppressor protein in porokeratosis. Arch Dermatol. 1994;130:187-190.
- Arranz-Salas I, Sanz-Trelles A, Ojeda DB. p53 alterations in porokeratosis. J Cutan Pathol. 2003;30:455-458.
- Curnow P, Foley P, Baker C. Multiple squamous cell carcinomas complicating linear porokeratosis. Australas J Dermatol. 2003;44:136-139.
- Lee HR, Han TY, Son SJ, et al. Squamous cell carcinoma developing within lesions of disseminated superficial actinic porokeratosis. Ann Dermatol. 2011;23:536-538.
- Mehta V, Balachandran C. Simultaneous co-occurrence of porokeratosis of Mibelli with disseminated superficial actinic porokeratosis. Indian J Dermatol. 2009;54:390-391.

A 69-year-old white man presented with a solitary lesion on the left ankle. His medical history included hypertension and arthritis. He resided in Florida for 11 years but denied tanning and has had sensitive skin throughout his life. He had no other notable skin conditions, except for nummular eczema. He did not have a family history of skin cancer. Physical examination showed the single lesion on the left ankle.
PDT with daylight effective in clearing AKs of the face, scalp
Photodynamic therapy (PDT) using daylight was as effective as PDT with an artificial light source in clearing grade I actinic keratoses (AKs) of the face and scalp after 3 months, but was associated with less pain and less severe side effects in a small Italian study.
In addition, most of the patients preferred the treatment with daylight photodynamic therapy (DL-PDT), reported Dr. Maria Concetta Fargnoli of the University of L’Aquila (Italy) and her associates (J Eur Acad Dermatol Venereol. 2015 Oct;29[10]:1926-32).
The prospective intrapatient study compared the effects of DL-PDT with conventional PDT (c-PDT) at 3 months in 35 patients with multiple grade I AKs of the face and scalp in September and October 2013. Patients were treated with c-PDT on one side of the face and DL-PDT on the other side. For c-PDT, light therapy was administered after methyl aminolevulinate (MAL) cream was applied to the treatment area and occluded. For DL-PDT, MAL cream was applied to the treatment area, left uncovered for 30 minutes in the dark; patients then exposed these areas to daylight for 2 hours, between 11 a.m. and 3 p.m., after which the cream was wiped off.
At 3 months, the complete response rate of grade I AKs was 87% for DL-PDT and 91% for c-PDT, which was not a significant difference. It was less effective for grade II and III AKs in the study, though, and at 6 months, the recurrence rate for grade I AKs treated with DL-PDT was higher (17%) than for those treated with c-PDT (12%), with a P value less than .05.
Treatment with DL-PDT was associated with significantly less pain and less severe adverse events, with less erythema, crusting, and pustular eruption 2 days after treatment. In addition, 88% of the patients were more satisfied with DL-PDT, the authors said.
“Our study confirms that DL-PDT using MAL is an effective, safe, and convenient alternative for the treatment of grade I AKs ,” they concluded. “Interestingly, clinical response for AK I was significantly moderated by outdoor temperature, increasing at higher temperatures.”
The authors had no conflicts of interest. Dr. Fargnoli received a research grant from Galderma, Italy, but Galderma played no role in the study, according to the study’s disclosure statement. Galderma manufactures the MAL cream product used in the study.
Photodynamic therapy (PDT) using daylight was as effective as PDT with an artificial light source in clearing grade I actinic keratoses (AKs) of the face and scalp after 3 months, but was associated with less pain and less severe side effects in a small Italian study.
In addition, most of the patients preferred the treatment with daylight photodynamic therapy (DL-PDT), reported Dr. Maria Concetta Fargnoli of the University of L’Aquila (Italy) and her associates (J Eur Acad Dermatol Venereol. 2015 Oct;29[10]:1926-32).
The prospective intrapatient study compared the effects of DL-PDT with conventional PDT (c-PDT) at 3 months in 35 patients with multiple grade I AKs of the face and scalp in September and October 2013. Patients were treated with c-PDT on one side of the face and DL-PDT on the other side. For c-PDT, light therapy was administered after methyl aminolevulinate (MAL) cream was applied to the treatment area and occluded. For DL-PDT, MAL cream was applied to the treatment area, left uncovered for 30 minutes in the dark; patients then exposed these areas to daylight for 2 hours, between 11 a.m. and 3 p.m., after which the cream was wiped off.
At 3 months, the complete response rate of grade I AKs was 87% for DL-PDT and 91% for c-PDT, which was not a significant difference. It was less effective for grade II and III AKs in the study, though, and at 6 months, the recurrence rate for grade I AKs treated with DL-PDT was higher (17%) than for those treated with c-PDT (12%), with a P value less than .05.
Treatment with DL-PDT was associated with significantly less pain and less severe adverse events, with less erythema, crusting, and pustular eruption 2 days after treatment. In addition, 88% of the patients were more satisfied with DL-PDT, the authors said.
“Our study confirms that DL-PDT using MAL is an effective, safe, and convenient alternative for the treatment of grade I AKs ,” they concluded. “Interestingly, clinical response for AK I was significantly moderated by outdoor temperature, increasing at higher temperatures.”
The authors had no conflicts of interest. Dr. Fargnoli received a research grant from Galderma, Italy, but Galderma played no role in the study, according to the study’s disclosure statement. Galderma manufactures the MAL cream product used in the study.
Photodynamic therapy (PDT) using daylight was as effective as PDT with an artificial light source in clearing grade I actinic keratoses (AKs) of the face and scalp after 3 months, but was associated with less pain and less severe side effects in a small Italian study.
In addition, most of the patients preferred the treatment with daylight photodynamic therapy (DL-PDT), reported Dr. Maria Concetta Fargnoli of the University of L’Aquila (Italy) and her associates (J Eur Acad Dermatol Venereol. 2015 Oct;29[10]:1926-32).
The prospective intrapatient study compared the effects of DL-PDT with conventional PDT (c-PDT) at 3 months in 35 patients with multiple grade I AKs of the face and scalp in September and October 2013. Patients were treated with c-PDT on one side of the face and DL-PDT on the other side. For c-PDT, light therapy was administered after methyl aminolevulinate (MAL) cream was applied to the treatment area and occluded. For DL-PDT, MAL cream was applied to the treatment area, left uncovered for 30 minutes in the dark; patients then exposed these areas to daylight for 2 hours, between 11 a.m. and 3 p.m., after which the cream was wiped off.
At 3 months, the complete response rate of grade I AKs was 87% for DL-PDT and 91% for c-PDT, which was not a significant difference. It was less effective for grade II and III AKs in the study, though, and at 6 months, the recurrence rate for grade I AKs treated with DL-PDT was higher (17%) than for those treated with c-PDT (12%), with a P value less than .05.
Treatment with DL-PDT was associated with significantly less pain and less severe adverse events, with less erythema, crusting, and pustular eruption 2 days after treatment. In addition, 88% of the patients were more satisfied with DL-PDT, the authors said.
“Our study confirms that DL-PDT using MAL is an effective, safe, and convenient alternative for the treatment of grade I AKs ,” they concluded. “Interestingly, clinical response for AK I was significantly moderated by outdoor temperature, increasing at higher temperatures.”
The authors had no conflicts of interest. Dr. Fargnoli received a research grant from Galderma, Italy, but Galderma played no role in the study, according to the study’s disclosure statement. Galderma manufactures the MAL cream product used in the study.
FROM THE JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Key clinical point: Photodynamic therapy using daylight was as effective as PDT with an artificial light source in clearing grade I actinic keratoses of the face and scalp after 3 months, but was associated with less pain and less severe side effects, and was more acceptable to patients.
Major finding: At 3 months, the complete response rate of grade I AKs treated with daylight photodynamic therapy was 87% vs. 91% among those treated with conventional PDT (P = .16).
Data source: A prospective intrapatient, left-right study compared the effects of daylight PDT with methyl aminolevulinate cream and conventional PDT at 3 months in 35 patients with grade I AKs. c-PDT was also combined with application of the MAL cream.
Disclosures: The authors had no financial conflicts of interest. Dr. Fargnoli received a research grant from Galderma, Italy, but Galderma played no role in the study, according to the study’s disclosure statement. Galderma manufactures the MAL cream product used in the study.
Topical fluorouracil shows long-term benefit for actinic keratoses
One course of topical fluorouracil cream reduced the need for localized treatments and the number of actinic keratoses (AK) over a mean follow-up of 2.6 years in the Veterans Affairs Keratinocyte Carcinoma Chemoprevention (VAKCC) trial.
These results “indicate that treating a patient with a single course of fluorouracil would reduce the subsequent number of spot treatments and benefit care of patients with multiple AKs for longer than 2 years,” concluded Dr. Hyemin Pomerantz of the department of dermatoepidemiology at the Providence (R.I.) VA Medical Center and his coauthors. Previous studies on treating AKs with topical fluorouracil have followed up participants for less than 6 months, they pointed out (JAMA Dermatol. 2015;9:952-60).
In the randomized, double-blinded, placebo-controlled study, conducted from 2009 to 2011 at 12 VA dermatology clinics, participants received topical fluorouracil cream, 5%, or a vehicle control cream, applied twice a day for 4 weeks, and were followed up for a mean of 2.6 years. There were no significant differences in the baseline characteristics of the 468 participants randomized to receive fluorouracil cream and the 464 participants randomized to receive the control cream.
The mean total AK count on the face and ears in both groups was about 11. At 6 months, the mean number of AKs per participant had dropped to 3 in the fluorouracil group (a 73% reduction from baseline) vs. a mean of 8.1 in the control group, a 24% reduction from baseline (P less than .001). Over the study period, those treated with fluorouracil had significantly fewer AKs. Over the entire study, there was not a significant difference in the number of hypertrophic AKs between the two groups, although the number of hypertrophic AKs was lower in the treatment group at 6 months.
During the study period, more participants treated with fluorouracil had complete clearance of AKs on the face and ears, compared with the control group (P less than .001). Finally, the fluorouracil group required 2 spot treatments per visit per participant vs. 3.9 in the control group (P less than .001).
“Our findings highlight the long-term efficacy of topical fluorouracil cream in treating and preventing AKs. A single course of topical fluorouracil cream, 5%, treatment led to a sustained reduction of the number of AKs and subsequent AK treatments,” in the population of patients at high risk for basal cell and squamous cell carcinomas, the authors wrote.
The study was supported by the Office of Research and Development Cooperative Studies Program at the Department of Veterans Affairs. Three of the authors reported several disclosures, including serving as a consultant to several pharmaceutical companies; the others, including the lead author, had no disclosures.
One course of topical fluorouracil cream reduced the need for localized treatments and the number of actinic keratoses (AK) over a mean follow-up of 2.6 years in the Veterans Affairs Keratinocyte Carcinoma Chemoprevention (VAKCC) trial.
These results “indicate that treating a patient with a single course of fluorouracil would reduce the subsequent number of spot treatments and benefit care of patients with multiple AKs for longer than 2 years,” concluded Dr. Hyemin Pomerantz of the department of dermatoepidemiology at the Providence (R.I.) VA Medical Center and his coauthors. Previous studies on treating AKs with topical fluorouracil have followed up participants for less than 6 months, they pointed out (JAMA Dermatol. 2015;9:952-60).
In the randomized, double-blinded, placebo-controlled study, conducted from 2009 to 2011 at 12 VA dermatology clinics, participants received topical fluorouracil cream, 5%, or a vehicle control cream, applied twice a day for 4 weeks, and were followed up for a mean of 2.6 years. There were no significant differences in the baseline characteristics of the 468 participants randomized to receive fluorouracil cream and the 464 participants randomized to receive the control cream.
The mean total AK count on the face and ears in both groups was about 11. At 6 months, the mean number of AKs per participant had dropped to 3 in the fluorouracil group (a 73% reduction from baseline) vs. a mean of 8.1 in the control group, a 24% reduction from baseline (P less than .001). Over the study period, those treated with fluorouracil had significantly fewer AKs. Over the entire study, there was not a significant difference in the number of hypertrophic AKs between the two groups, although the number of hypertrophic AKs was lower in the treatment group at 6 months.
During the study period, more participants treated with fluorouracil had complete clearance of AKs on the face and ears, compared with the control group (P less than .001). Finally, the fluorouracil group required 2 spot treatments per visit per participant vs. 3.9 in the control group (P less than .001).
“Our findings highlight the long-term efficacy of topical fluorouracil cream in treating and preventing AKs. A single course of topical fluorouracil cream, 5%, treatment led to a sustained reduction of the number of AKs and subsequent AK treatments,” in the population of patients at high risk for basal cell and squamous cell carcinomas, the authors wrote.
The study was supported by the Office of Research and Development Cooperative Studies Program at the Department of Veterans Affairs. Three of the authors reported several disclosures, including serving as a consultant to several pharmaceutical companies; the others, including the lead author, had no disclosures.
One course of topical fluorouracil cream reduced the need for localized treatments and the number of actinic keratoses (AK) over a mean follow-up of 2.6 years in the Veterans Affairs Keratinocyte Carcinoma Chemoprevention (VAKCC) trial.
These results “indicate that treating a patient with a single course of fluorouracil would reduce the subsequent number of spot treatments and benefit care of patients with multiple AKs for longer than 2 years,” concluded Dr. Hyemin Pomerantz of the department of dermatoepidemiology at the Providence (R.I.) VA Medical Center and his coauthors. Previous studies on treating AKs with topical fluorouracil have followed up participants for less than 6 months, they pointed out (JAMA Dermatol. 2015;9:952-60).
In the randomized, double-blinded, placebo-controlled study, conducted from 2009 to 2011 at 12 VA dermatology clinics, participants received topical fluorouracil cream, 5%, or a vehicle control cream, applied twice a day for 4 weeks, and were followed up for a mean of 2.6 years. There were no significant differences in the baseline characteristics of the 468 participants randomized to receive fluorouracil cream and the 464 participants randomized to receive the control cream.
The mean total AK count on the face and ears in both groups was about 11. At 6 months, the mean number of AKs per participant had dropped to 3 in the fluorouracil group (a 73% reduction from baseline) vs. a mean of 8.1 in the control group, a 24% reduction from baseline (P less than .001). Over the study period, those treated with fluorouracil had significantly fewer AKs. Over the entire study, there was not a significant difference in the number of hypertrophic AKs between the two groups, although the number of hypertrophic AKs was lower in the treatment group at 6 months.
During the study period, more participants treated with fluorouracil had complete clearance of AKs on the face and ears, compared with the control group (P less than .001). Finally, the fluorouracil group required 2 spot treatments per visit per participant vs. 3.9 in the control group (P less than .001).
“Our findings highlight the long-term efficacy of topical fluorouracil cream in treating and preventing AKs. A single course of topical fluorouracil cream, 5%, treatment led to a sustained reduction of the number of AKs and subsequent AK treatments,” in the population of patients at high risk for basal cell and squamous cell carcinomas, the authors wrote.
The study was supported by the Office of Research and Development Cooperative Studies Program at the Department of Veterans Affairs. Three of the authors reported several disclosures, including serving as a consultant to several pharmaceutical companies; the others, including the lead author, had no disclosures.
FROM JAMA DERMATOLOGY
Key clinical point:One course of topical fluorouracil cream, 5%, decreased the need for localized treatment and the number of actinic keratoses long term.
Major finding: Participants whose AKs were treated with a course of fluorouracil cream, 5%, had significantly fewer AKs and required fewer treatments, compared with the control group, for over 2 years.
Data source: The randomized, double-blind study compared the effect of a course of topical fluorouracil cream with a vehicle cream on the number of AKs and other measures, over a mean follow-up of 2.6 years, in 932 patients treated at 12 VA dermatology clinics.
Disclosures: The study was supported by the Office of Research and Development Cooperative Studies Program at the Department of Veterans Affairs. Three of the authors reported several disclosures, including serving as a consultant to several pharmaceutical companies; the others had no disclosures.
Sunscreens with DNA repair enzymes might lessen AK progression
Patients with actinic keratosis who used UPF 50 sunscreen containing DNA repair enzymes improved significantly more on two measures of malignant progression than did those who used sunscreen alone, according to research published in the Journal of Drugs in Dermatology.
At 6 months, improvements in field cancerization and levels of cyclobutane pyrimidine dimers were significantly greater (P less than .001) for the sunscreen-plus-enzymes group compared with sunscreen-only patients, wrote Dr. Mauro Carducci of Centro Ortopedico di Quadrante in Omegna, Italy, and his associates.
The study is the first of its type to directly compare the clinical effects of two such topicals, the investigators wrote. The findings set the stage for longer, larger trials that are powered to assess the risk of progression to squamous cell carcinoma, they added.
For the study, 28 patients with AK were randomly assigned to use SPF 50 sunscreen alone or a formula that contained 1% photolyase from Anacystis nidulans and 1% endonuclease from Micrococcus luteus. Patients applied 2 mg/cm2 of sunscreen to treatment areas that contained 4-10 AKs. They were not allowed to use other topicals during the trial or for 2 weeks beforehand.
All of the patients were white and aged older than 65 years; three-quarters were men. The investigators used fluorescence diagnostics with methylaminolaevulinate to measure field cancerization, and analyzed skin biopsies to quantify CPD levels (J Drugs Dermatol. 2015;14[9]:986-90.).
Hyperkeratosis improved the same amount in both groups at month 6, according to the researchers. But field cancerizations dropped 29% from baseline in the sunscreen-plus-enzymes group, compared with a 10% decrease with sunscreen alone (P less than .0001). Likewise, CPD levels fell 61% from baseline in the sunscreen-plus-enzymes group compared with a 35% drop with sunscreen alone (P less than .0001).
Despite those significant differences, the study was not powered to detect differences in the risk of transformation to SCC, the researchers cautioned.
Biodue S.p.A. provided the methyl aminolevulinate used in the study. Dr. Enzo Emanuele, the study’s senior author, is a major shareholder of Living Research S.A.S., a privately held biomedical research organization that provided funding for the work. The other researchers reported no conflicts of interest.
Patients with actinic keratosis who used UPF 50 sunscreen containing DNA repair enzymes improved significantly more on two measures of malignant progression than did those who used sunscreen alone, according to research published in the Journal of Drugs in Dermatology.
At 6 months, improvements in field cancerization and levels of cyclobutane pyrimidine dimers were significantly greater (P less than .001) for the sunscreen-plus-enzymes group compared with sunscreen-only patients, wrote Dr. Mauro Carducci of Centro Ortopedico di Quadrante in Omegna, Italy, and his associates.
The study is the first of its type to directly compare the clinical effects of two such topicals, the investigators wrote. The findings set the stage for longer, larger trials that are powered to assess the risk of progression to squamous cell carcinoma, they added.
For the study, 28 patients with AK were randomly assigned to use SPF 50 sunscreen alone or a formula that contained 1% photolyase from Anacystis nidulans and 1% endonuclease from Micrococcus luteus. Patients applied 2 mg/cm2 of sunscreen to treatment areas that contained 4-10 AKs. They were not allowed to use other topicals during the trial or for 2 weeks beforehand.
All of the patients were white and aged older than 65 years; three-quarters were men. The investigators used fluorescence diagnostics with methylaminolaevulinate to measure field cancerization, and analyzed skin biopsies to quantify CPD levels (J Drugs Dermatol. 2015;14[9]:986-90.).
Hyperkeratosis improved the same amount in both groups at month 6, according to the researchers. But field cancerizations dropped 29% from baseline in the sunscreen-plus-enzymes group, compared with a 10% decrease with sunscreen alone (P less than .0001). Likewise, CPD levels fell 61% from baseline in the sunscreen-plus-enzymes group compared with a 35% drop with sunscreen alone (P less than .0001).
Despite those significant differences, the study was not powered to detect differences in the risk of transformation to SCC, the researchers cautioned.
Biodue S.p.A. provided the methyl aminolevulinate used in the study. Dr. Enzo Emanuele, the study’s senior author, is a major shareholder of Living Research S.A.S., a privately held biomedical research organization that provided funding for the work. The other researchers reported no conflicts of interest.
Patients with actinic keratosis who used UPF 50 sunscreen containing DNA repair enzymes improved significantly more on two measures of malignant progression than did those who used sunscreen alone, according to research published in the Journal of Drugs in Dermatology.
At 6 months, improvements in field cancerization and levels of cyclobutane pyrimidine dimers were significantly greater (P less than .001) for the sunscreen-plus-enzymes group compared with sunscreen-only patients, wrote Dr. Mauro Carducci of Centro Ortopedico di Quadrante in Omegna, Italy, and his associates.
The study is the first of its type to directly compare the clinical effects of two such topicals, the investigators wrote. The findings set the stage for longer, larger trials that are powered to assess the risk of progression to squamous cell carcinoma, they added.
For the study, 28 patients with AK were randomly assigned to use SPF 50 sunscreen alone or a formula that contained 1% photolyase from Anacystis nidulans and 1% endonuclease from Micrococcus luteus. Patients applied 2 mg/cm2 of sunscreen to treatment areas that contained 4-10 AKs. They were not allowed to use other topicals during the trial or for 2 weeks beforehand.
All of the patients were white and aged older than 65 years; three-quarters were men. The investigators used fluorescence diagnostics with methylaminolaevulinate to measure field cancerization, and analyzed skin biopsies to quantify CPD levels (J Drugs Dermatol. 2015;14[9]:986-90.).
Hyperkeratosis improved the same amount in both groups at month 6, according to the researchers. But field cancerizations dropped 29% from baseline in the sunscreen-plus-enzymes group, compared with a 10% decrease with sunscreen alone (P less than .0001). Likewise, CPD levels fell 61% from baseline in the sunscreen-plus-enzymes group compared with a 35% drop with sunscreen alone (P less than .0001).
Despite those significant differences, the study was not powered to detect differences in the risk of transformation to SCC, the researchers cautioned.
Biodue S.p.A. provided the methyl aminolevulinate used in the study. Dr. Enzo Emanuele, the study’s senior author, is a major shareholder of Living Research S.A.S., a privately held biomedical research organization that provided funding for the work. The other researchers reported no conflicts of interest.
FROM THE JOURNAL OF DRUGS IN DERMATOLOGY
Key clinical point:Sunscreen containing DNA repair enzymes might prevent malignant progression of actinic keratosis better than sunscreen alone.
Major finding: Field cancerization and cyclobutane pyrimidine dimer levels improved significantly more with sunscreen plus enzymes than with sunscreen only (P less than .0001 for each).
Data source: Six-month randomized trial of 28 patients with actinic keratosis.
Disclosures: Biodue S.p.A. provided the methyl aminolevulinate used in the study. Dr. Enzo Emanuele, the study’s senior author, is a major shareholder of Living Research S.A.S., a privately held biomedical research organization that provided funding for the work. The other researchers reported no conflicts of interest.
Therapies for Actinic Keratosis With a Focus on Cosmetic Outcomes
Actinic keratosis (AK), also referred to as solar keratosis or senile keratosis, is an intraepidermal proliferation of dysplastic keratinocytes that develops in response to chronic exposure to UV radiation. Actinic keratoses are among the most commonly encountered lesions seen by dermatologists, and it has been estimated that 60% of predisposed individuals older than 40 years have at least one AK.1,2 Prevalence is notably higher in light-skinned individuals and increases with age, presumably from higher cumulative sun exposure and decreased effectiveness of the immune system.1,3 It remains a point of contention as to whether or not AKs actually represent squamous cell carcinoma (SCC) in situ, but the potential for progression to invasive disease has been well demonstrated, as the majority of SCCs develop from preexisting AKs.4-6 The risk for progression to invasive disease for an individual AK has been estimated to range from 0.025% to 16% per year, with an average of approximately 8% in immunocompetent patients.7
The clinical morphology of AK can vary widely, but the most common presentation is an erythematous scaly macule, papule, or plaque on sun-exposed skin. The skin surrounding AKs typically shows evidence of solar damage with deep wrinkling, mottled pigmentation, scattered telangiectases, purpura, or xerosis (Figure). A variety of clinical variants with unique presentations exist, including atrophic, hypertrophic, acantholytic, lichenoid, bowenoid, and pigmented subtypes. Because more than 80% of AKs occur on highly visible areas such as the head, neck, back of the hands, and forearms, AKs can have an obvious detrimental effect on cosmetic appearance. Studies also have shown a strong association between AKs and decreased overall quality of life (QOL).3,8,9

Because of the risk for AK progression to invasive cancer along with its negative impact on cosmesis and QOL, clinicians generally opt to treat AKs. Numerous different treatment options exist, including topical medications, procedural modalities, and light-based therapies. Here, we review the efficacy of the most commonly utilized treatments and discuss the relevant cosmetic considerations and outcomes.
Topical Treatments
5-Fluorouracil
5-Fluorouracil (5-FU) is a US Food and Drug Administration (FDA)–approved, topically applied pyrimidine analogue that inhibits thymidylate synthase. The resulting suppression of DNA and RNA synthesis induces cell death with a preference for mitotically active cells.10 5-Fluorouracil has been used for more than 50 years as a treatment of AK and its efficacy is well established. A systematic review of 5 randomized controlled studies of topical 5-FU reported an average of 49% of 423 patients achieving complete lesion clearance with 5-FU cream 5% applied once or twice daily for up to 7 weeks.11 Some notable drawbacks of 5-FU, however, are application-site erythema, blistering, pruritus, necrosis, erosion, and pain. These effects often lead to premature cessation of therapy, but newer formulations of 5-FU cream 0.5% have shown good efficacy with better tolerability.12 A randomized, double-blind, multicenter, parallel-group study of 177 patients using 5-FU cream 0.5% once daily for either 1, 2, or 4 weeks demonstrated significant (P<.001) efficacy over vehicle gel in all treatment arms.13 The most effective therapy was 4 weeks of treatment, which achieved a mean 91.7% reduction in lesion count as assessed 1 month after cessation of therapy. The primary adverse effect (AE) reported in this trial was mild to moderate facial irritation, which generally resolved within 18 to 21 days after treatment cessation.13 Overall, 5-FU is a highly effective therapy for treating AKs that also can improve signs of photoaging, but patients should be aware of cosmetically unappealing effects that generally occur throughout therapy and during the immediate posttreatment period.14
Chemical Peels
Chemical peels traditionally employ acidic compounds to strip away outer layers of skin to variable depths depending on the concentration of the agent being applied. For treatment of AK, trichloroacetic acid (TCA) is a commonly employed cauterant that has shown efficacy comparable to topical 5-FU as well as ablative CO2 laser resurfacing.15 Trichloroacetic acid peels also are a convenient therapy, as good results can be achieved after a single treatment session. A split-face study of 15 patients treated with either a single application of 35% TCA and Jessner solution or twice-daily application of 5-FU cream 5% for 3 weeks demonstrated a reduction in 75% of visible AKs in both treatment arms over a 1-year follow-up period.16 Although 80% of patients self-reported considerable cosmetic improvement with both therapies, patient preference was reported to be in favor of the TCA peel, given its quick results and relatively mild side effects as compared to 5-FU. Treatment with chemical peels will result in temporary erythema and mild desquamation that usually resolves within 2 weeks; however, there are cases in which erythema has been reported to persist for several months.16 Adverse effects such as permanent scarring or pigmentation changes rarely are seen with TCA concentrations less than 45%.17 Caution should be used in patients with a history of herpes simplex virus, keloids, postinflammatory hyperpigmentation, radiation exposure, immunosuppression, and those unable or unwilling to use sunscreen and avoid sun exposure in the immediate posttreatment period.
Diclofenac Sodium
Diclofenac sodium (DFS) is an FDA-approved topical, nonsteroidal,
anti-inflammatory drug whose mechanism of action in the treatment of AK is thought to involve inhibition of the cyclooxygenase 2 enzyme.18 The resulting reduction of prostaglandins is believed to inhibit tumor angiogenesis, induce apoptosis, and inhibit cell differentiation.19-22 In a multicenter, double-blind, placebo-controlled study of 195 patients, application of DFS 3% in hyaluronan gel 2.5% twice daily for 60 days showed significant (P<.05) efficacy over placebo in achieving complete resolution of target lesions during a 30-day follow-up period (31% vs 10%). Furthermore, qualitative patient assessment of complete global improvement also was significantly (P<.05) higher in the active treatment group as compared to placebo (31% vs 10%).23 Additional studies of DFS 3% in hyaluronan gel 2.5% applied twice daily for 90 days have shown even higher rates of success, with complete resolution of target lesions in 40% to 58% of cases.24,25 This therapy also has been reported to substantially improve QOL following treatment completion.26 The most frequently cited AEs include pruritus, rash, dry skin, erythema, and application-site reactions. Overall, DFS is a
well-tolerated therapy with efficacy comparable to that of 5-FU but with a lower incidence of AEs
and higher patient satisfaction as determined in
2 head-to-head studies.27,28
ImiquimodImiquimod (IMQ) is an FDA-approved topical agent that functions as an immune response modifier via agonism of toll-like receptor 7.18 The resulting cytokine production and release enhances the innate and acquired immune responses leading to anticancer activity.29 The efficacy of IMQ for treatment of AK has been demonstrated in numerous well-designed clinical trials. A
meta-analysis of 5 randomized, double-blind trials including 1293 patients treated with IMQ cream 5%
2 to 3 times per week for 12 to 16 weeks reported complete clearance of AKs in 50% of patients treated with IMQ as compared to 5% of patients treated with vehicle.30 The most frequently reported AEs with this therapy include erythema, scabbing, flaking, and erosion. These effects generally resolve following cessation of treatment, and therapy is considered to be well tolerated; however, there are case reports of IMQ triggering or exacerbating existing inflammatory conditions.31 Imiquimod cream also is approved at 2.5% and 3.75% concentrations, which have demonstrated significant (P<.001) efficacy over placebo and a reduced incidence of AEs; complete clearance rates have been reported as 30.6% and 35.6%, respectively.32 Notably, a study comparing 75 patients randomized to either IMQ cream 5%
3 times per week for 4 weeks, 1 or 2 courses of cryosurgery, or 5-FU ointment 5% twice daily for 4 weeks reported that IMQ achieved significantly (P<.01) superior sustained clearance rates during a 12-month follow-up period over cryosurgery and 5-FU
(73% vs 4% vs 33%).33 Additionally, cosmetic outcomes as determined by both participants and investigators were reported as excellent at 12 months posttreatment in more than 80% of participants treated with IMQ. These excellent, long-lasting cosmetic outcomes also were determined to be significantly (P<.0001) superior to the cosmetic outcomes of 5-FU and cryotherapy, which both reported excellent outcomes in less than 10% of cases.33
Ingenol MebutateIngenol mebutate (IM) is a macrocyclic diterpene ester derived from the Euphorbia peplus plant that is FDA approved for the treatment of AK.1 Ingenol mebutate’s mechanism of action is thought to involve induction of cell death via disruption of the plasma membrane and mitochondria in addition to production of an inflammatory response, which produces tumor-specific antibodies and a large influx of neutrophils.34,35 The overall evidence for the efficacy of IM is strong. A combined analysis of 4 multicenter, randomized, double-blind studies of 1005 participants reported that IM gel 0.015% applied once daily for 3 days to the face or scalp was significantly superior (P<.001) to placebo in achieving complete clearance as assessed 54 days after completion of therapy (42.2% vs 3.7%) and that IM gel 0.05% applied once daily for 2 days to the trunk or extremities also was significantly superior (P<.001) to placebo in achieving complete clearance as determined 55 days after completion of therapy (34.1% vs 4.7%).36 A follow-up report to this study indicated that IM also appears to achieve long-lasting effects with an overall 87% decrease in total AKs at 12 months follow-up in both trial groups.37 Additionally, it has been recently reported that treatment with IM in these trials was associated with significantly higher overall treatment satisfaction (P<.001) and improved QOL (P<.001) as compared to vehicle.38 Cosmetic outcomes of IM therapy have been assessed in a trial analyzing the efficacy of IM gel 0.025% for 3 days or IM gel 0.05% for 2 or 3 days on nonfacial AKs. This study reported significantly (P<.0001) higher patient satisfaction with the cosmetic outcome at 8 weeks after therapy as compared to vehicle.34 Studies performed in mice have demonstrated that IM is able to promote collagen matrix turnover and impose dermal elasticity, which may contribute to these good cosmetic outcomes.39 The most common AEs of IM therapy are erythema, crusting, and flaking; these effects generally occur 3 to 8 days after starting treatment. These effects, however, generally are short lived and resolve within 2 weeks of treatment cessation when IM is applied to the face or scalp or 4 weeks when applied to the trunk or extremities.40 Overall, IM is a useful therapeutic option given its relatively short treatment course as compared to other topically applied agents, as well as its lasting efficacy, mild AEs, and good cosmetic outcomes.
Procedural Modalities
Surgical Procedures
Surgical approaches for the treatment of AK include excision, curettage with or without electrodesiccation, and dermabrasion. In the past, these modalities were used with greater frequency, but the advent of effective topical medications with lower risks of AEs has largely reduced their use.41 Excision may still be indicated in cases where SCC is suspected, and curettage can be used for treatment of thicker hypertrophic AKs.42 Although these approaches have not been evaluated in clinical trials, they are generally effective but require the use of local anesthetics and come with substantial risk for infection, permanent scarring, and hypopigmentation. Dermabrasion employs the use of a motorized device equipped with an abrasive material to physically remove superficial layers of the skin. Studies are limited, but this method has been reported as an effective treatment in a retrospective review of 23 participants in which 96% remained free of AKs at 1 year, 83% at 2 years, 64% at 4 years, and 54% at 5 years posttherapy.43 Notably, one split-face study of 40 participants treated with dermabrasion followed by 25% TCA on one side and either Jessner solution and 35% TCA or dermabrasion alone on the other side reported that the combination of dermabrasion with 25% TCA consistently produced excellent cosmetic results with nearly complete eradication of AKs.44 In general, however, cosmetic outcomes with dermabrasion are variable, as the technique is highly operator dependent and treatment is associated with notable discomfort as well as risk for scarring and permanent pigmentation alteration.
Cryotherapy
Cryotherapy remains one of the most commonly utilized treatments of AK and involves the delivery of liquid nitrogen via a spray device or a cotton tip applicator to rapidly freeze cells, thus causing cellular destruction via ice crystal formation and protein denaturation.45 Efficacy with this technique has been reported to be as high as 98.8% at 12 months follow-up, but more recent studies cite lower rates of success.46 A prospective multicenter study of 90 participants with 421 AKs on the face or scalp treated with a single freeze-thaw cycle of liquid nitrogen reported an overall complete response rate of 67.2% at 3 months posttherapy. Additionally, higher complete response rates were associated with longer freeze times, and cosmetic outcomes were reported as good to excellent in 94% of complete response lesions.47 Similar results were reported in an open-label, prospective, randomized, controlled clinical trial of 200 participants with 543 AKs, which compared a single freeze-thaw cycle with liquid nitrogen to a single session of CO2 laser ablation in the treatment of isolated AKs of the face and scalp.48 At 3 months posttherapy, complete clearance was observed in 71.6% of participants treated with cryotherapy and in 65.3% of participants treated with laser ablation (P=.532). At 12 months posttherapy, participants who originally showed complete response at 3 months were assessed for relapse. Complete clearance was preserved in 72.6% of participants treated with cryotherapy versus 21.9% of participants treated with laser ablation (P<.0001), and cosmetic outcomes were reported by participants as good or excellent at 3 months follow-up in more than 93% of participants for both treatment arms.48 Possible AEs of cryotherapy include pain during treatment, blister formation with possible hemorrhage, infection, scarring, and permanent pigmentary changes.47,48 Notably, the risk for hypopigmentation increases with longer freezing times, thus requiring clinicians to consider the balance between improved efficacy and reduced cosmetic outcomes.47
Light-Based Therapies
Laser Therapy
Ablative laser resurfacing with either the CO2 or erbium-doped:YAG (Er:YAG) laser utilizes light of specific wavelengths to selectively induce thermolysis and destruction of the epidermal layer. Both lasers have been studied as treatments of AK, but there is a lack of large, well-designed studies. In one small study of 14 participants treated with 1 to 2 passes of the CO2 laser, complete clearance was reported in all cases without any recurrences during a follow-up period of 6 to 24 months. Additionally, all participants in this study reported satisfaction with the cosmetic outcome.49 The CO2 laser also has demonstrated efficacy comparable to that of the TCA peel and 5-FU therapy in a prospective randomized trial of 34 patients with facial or scalp AKs who received either CO2 laser with 2 passes, 30% TCA peel, or 5-FU cream 5% twice daily for 3 weeks.15 Reduction in mean AK counts at 3 months posttherapy was significantly (P<.03) higher in all treatment arms as compared to the control group (92% for CO2 laser, 89% for TCA peel, and 83% for 5-FU cream). No significant (P=.31) difference in outcomes was noted among the different treatment arms.15 Similar results were reported for the Er:YAG laser in a small prospective study of 5 participants treated with 2 to 3 passes with the Er:YAG laser in which reduction in mean AK counts was reported as ranging from 86% to 96% at 3 months posttherapy.50 The Er:YAG laser in combination with the CO2 laser has shown notable long-term efficacy in achieving higher lesion clearance rates and sustained complete clearance rates over treatment with topical 5-FU.51 In a prospective randomized study of 55 par-ticipants with multiple AKs on the face or scalp, participants were assigned to receive either combination laser ablation with the Er:YAG and CO2 lasers down to the level of the papillary dermis or 5-FU cream 5% applied twice daily for 2 to 7 weeks until an appropriate clinical inflammatory response was achieved. At 12 months follow-up, the laser treatment group achieved significantly (P=.048) higher mean lesion clearance rates (91.1%) as compared to the 5-FU arm (76.6%) and significantly (P=.003) higher sustained complete clearance rates (59.3%) as compared to 5-FU (29.2%). The proportion of participants with an improvement in photoaging score at 12 months follow-up approached statistical significance (P=.07), with 74% of the laser-treated group showing improvement as compared to 43% of the 5-FU–treated group. Long-term, cosmetically unappealing side effects such as erythema and hypopigmentation occurred notably more often in the laser-treated group as compared to the 5-FU group.51 In summary, ablative lasers appear to be a highly effective therapy for AK but at the cost of increased risk for AEs such as permanent pigmentary changes, prolonged erythema lasting up to several months, and scarring.50,52-55
Fractional photothermolysis is a relatively new advancement in the field of laser therapy that has received FDA approval for the treatment of AK.56 This treatment works by creating multiple noncontiguous microscopic columns of thermal injury while sparing adjacent zones of viable tissue.57 Although there are limited studies involving the use of such lasers in the treatment of AK, initial findings suggest that 1927-nm thulium lasers may be more effective than 1550-nm erbium lasers in achieving lesion clearance. A trial of 14 participants who received 5 laser treatments with a 1550-nm fractionated erbium-doped fiber laser reported an average reduction in AK counts of 66.2% at 3 months follow-up and a 55.6% reduction at 6 months follow-up. A participant-determined marked or very significant improvement of lesions was reported in 83% of participants at 1 month posttreatment but only in 44% of participants at 6 months posttreatment.58 A similar trial of 24 participants treated with up to 4 treatment sessions of the fractionated 1927-nm thulium laser reported an 87.3% reduction in number of AKs at 3 months follow-up and an 86.6% reduction at 6 months follow-up.56 The primary advantage of fractional laser therapy is a faster recovery period generally lasting only 2 or 3 days as compared to 2 weeks or more with traditional ablative lasers, thus limiting the amount of time a patient must tolerate cosmetically unappealing erythema.59,60 The quick recovery time has been attributed to the fractional laser’s ability to preserve the stratum corneum and skin barrier, which also helps reduce the risk for other AEs such as scarring and infection.56,59-61 Additional studies are needed to better assess the true efficacy of fractional laser therapy, but treatment with the fractional 1927-nm thulium laser appears to be a promising and well-tolerated therapeutic option for treatment of AK with similar efficacy to traditional ablative lasers but with a lower risk of AEs.
Photodynamic TherapyPhotodynamic therapy (PDT) is an FDA-approved treatment that involves the use of a topical photosensitizing agent such as 5-aminolevulinic acid (ALA) or methyl aminovulinate (MAL) before exposure to an activating light source to generate reactive oxygen species that lead to cell death.62-65 Multiple PDT regimens with varying combinations of photosensitizers, incubation time, and light sources have been studied, but a 2012 Cochrane review determined that treatment with conventional formulations of MAL and ALA with either blue- or red-light PDT were similarly efficacious for treatment of individual AKs as compared to vehicle with blue- or red-light PDT. One exception was that longer incubation time (ie, 4 hours) with ALA resulted in better results than shorter incubation times (ie, 0.5, 1, 2 hours) with ALA.66
Standard PDT treatment with MAL also has consistently demonstrated superior efficacy in achieving complete clearance rates in addition to superior cosmetic outcomes over treatment with either cryotherapy, DFS, or 5-FU.67-73 Three studies in particular noted an excellent or good investigator-determined cosmetic outcome in 96% to 98% of participants treated with MAL-PDT.69,71,74 Photodynamic therapy with ALA also has been reported as superior over CO2 laser ablation for AK reduction as well as both patient and investigator overall satisfaction.75
More recently, several methods of improving photosensitizer delivery have been studied, which have demonstrated remarkable efficacy at achieving lesion clearance over standard cream formulations or application routines. One such method involves the use of gentle heating to increase photosensitizer uptake. In a split-extremity study of 20 participants who were treated with 20% ALA under occlusion for 1 hour with one side heated to 38.8°C, the heated side demonstrated significant (P<.0001) efficacy at achieving higher median clearance rates over control when evaluated at 2 and 6 months posttherapy.76 Notably, occlusion of ALA in itself during the incubation period also has been demonstrated to significantly (P<.0001) improve clearance rates.77 Another method involves the use of a new nanoemulsion-based formulation of ALA gel, known as BF-200 ALA, which has demonstrated remarkable efficacy over standard MAL cream and placebo in a long-term follow-up analysis of 2 prospective, randomized, controlled trials.78 In a similar vein, 3 prospective randomized trials with a minimum follow-up time of 3 months demonstrated that MAL-PDT in combination with fractional ablative laser pretreatment has significant (P<.02 in all trials) efficacy over MAL-PDT without pretreatment in achieving complete AK clearance. Although the cosmetic outcomes were good or excellent in 87% to 100% of patients, they were not significantly different from stand-alone MAL-PDT treatment in any of the trials.79-81 However, pretreatment with microneedling in MAL-PDT has been shown to achieve superior cosmetic outcomes over MAL-PDT without microneedling, according to one small split-face study of 10 participants.82
Overall, PDT is an excellent therapeutic option that is able to provide efficacious clearance of AKs as well as superior cosmetic outcomes. Common AEs of PDT include burning, itching, and stinging during therapy, but pain intensity decreases dramatically upon termination of illumination, with cessation of most symptoms by 12 hours posttherapy.73 Permanent pigmentation changes have been reported to occasionally occur following PDT therapy.81
Conclusion
When determining which therapy to use in a patient, clinicians must take into account a variety of factors such as patient preference, cost of treatment, availability, tolerance for AEs, and the need for field therapy. Although all therapies discussed within this article are effective and reasonable treatment choices, patients who are particularly concerned about cosmetic outcomes would most likely benefit from either IMQ or PDT, as the data for cosmetic outcomes with these therapies are the strongest. Combination or sequential treatments may be required in some cases and all patients should be monitored for lesion recurrence regardless of treatment choice. A summary of the therapies and key studies discussed here is available in the PDF.
- Lebwohl M. Actinic keratosis: epidemiology and progression to squamous cell carcinoma. Br J Dermatol. 2003;149(suppl 66):31-33.
- Drake LA, Ceilley RI, Cornelison RL, et al. Guidelines of care for actinic keratoses. Committee on Guidelines of Care. J Am Acad Dermatol. 1995;32:95-98.
- Salasche SJ. Epidemiology of actinic keratoses and squamous cell carcinoma. J Am Acad Dermatol. 2000;42(1, pt 2):4-7.
- Ackerman AB, Mones JM. Solar (actinic) keratosis is squamous cell carcinoma. Br J Dermatol. 2006;155:9-22.
- Anwar J, Wrone DA, Kimyai-Asadi A, et al. The development of actinic keratosis into invasive squamous cell carcinoma: evidence and evolving classification schemes. Clin Dermatol. 2004;22:189-196.
- Criscione VD, Weinstock MA, Naylor MF, et al. Actinic keratoses: natural history and risk of malignant transformation in the Veterans Affairs Topical Tretinoin Chemoprevention Trial. Cancer. 2009;115:2523-2530.
- Glogau RG. The risk of progression to invasive disease. J Am Acad Dermatol. 2000;42(1, pt 2):23-24.
- Esmann S, Jemec GB. Management of actinic keratosis patients: a qualitative study. J Dermatolog Treat. 2007;18:53-58.
- Weinstock MA, Lee KC, Chren MM, et al. Quality of life in the actinic neoplasia syndrome: the VA Topical Tretinoin Chemoprevention (VATTC) trial. J Am Acad Dermatol. 2009;61:207-215.
- Berman B, Villa AM, Ramirez CC. Mechanisms of action of new treatment modalities for actinic keratosis. J Drugs Dermatol. 2006;5:167-173.
- Askew DA, Mickan SM, Soyer HP. Effectiveness of 5-fluorouracil treatment for actinic keratosis: a systematic review of randomized controlled trials. Int J Dermatol. 2009;46:452-463.
- Levy S, Furst K, Chern W. A pharmacokinetic evaluation of 0.5% and 5% fluorouracil topical cream in patients with actinic keratosis. Clin Ther. 2001;23:908-920.
- Jorizzo J, Stewart D, Bucko A, et al. Randomized trial evaluating a new 0.5% fluorouracil formulation demonstrates efficacy after 1-, 2-, or 4-week treatment in patients with actinic keratosis. Cutis. 2002;70:335-359.
- Sachs DL, Kang S, Hammerberg C, et al. Topical fluorouracil for actinic keratoses and photoaging: a clinical and molecular analysis. Arch Dermatol. 2009;145:659-666.
- Hantash BM, Stewart DB, Cooper ZA, et al. Facial resurfacing for nonmelanoma skin cancer prophylaxis. Arch Dermatol. 2006;142:976-982.
- Lawrence N, Cox SE, Cockerell CJ, et al. A comparison of the efficacy and safety of Jessner’s solution and 35% trichloroacetic acid vs 5% fluorouracil in the treatment of widespread facial actinic keratoses. Arch Dermatol. 1995;131:176-181.
- Monheit GD. The Jessner’s + TCA peel: a medium-depth chemical peel. J Dermatol Surg Oncol. 1989;15:945-950.
- Hemmi H, Kaisho T, Takeuchi O, et al. Small anti-viral compounds activate immune cells via the TLR7 MyD88-dependent signaling pathway. Nat Immunol. 2002;3:196-200.
- Adamson DJ, Frew D, Tatoud R, et al. Diclofenac antagonizes peroxisome proliferator-activated receptor-gamma signaling. Mol Pharmacol. 2002;61:7-12.
- Alam CA, Seed MP, Willoughby DA. Angiostasis and vascular regression in chronic granulomatous inflammation induced by diclofenac in combination with hyaluronan in mice. J Pharm Pharmacol. 1995;47:407-411.
- Lu X, Xie W, Reed D, et al. Nonsteroidal antiinflammatory drugs cause apoptosis and induce cyclooxygenases in chicken embryo fibroblasts. Proc Natl Acad Sci USA. 1995;92:7961-7965.
- Seed MP, Brown JR, Freemantle CN, et al. The inhibition of colon-26 adenocarcinoma development and angiogenesis by topical diclofenac in 2.5% hyaluronan. Cancer Res. 1997;57:1625-1629.
- Rivers JK, Arlette J, Shear N, et al. Topical treatment of actinic keratoses with 3.0% diclofenac in 2.5% hyaluronan gel. Br J Dermatol. 2002;146:94-100.
- Wolf JE, Taylor JR, Tschen E, et al. Topical 3.0% diclo-fenac in 2.5% hyaluronan gel in the treatment of actinic keratoses. Int J Dermatol. 2001;40:709-713.
- Nelson C, Rigel D, Smith S, et al. Phase IV, open-label assessment of the treatment of actinic keratosis with 3.0% diclofenac sodium topical gel (Solaraze). J Drugs Dermatol. 2004;3:401-407.
- Pflugfelder A, Welter AK, Leiter U, et al. Open label randomized study comparing 3 months vs. 6 months treatment of actinic keratoses with 3% diclofenac in 2.5% hyaluronic acid gel: a trial of the German Dermatologic Cooperative Oncology Group. J Eur Acad Dermatol Venereol. 2012;26:48-53.
- Smith SR, Morhenn VB, Piacquadio DJ. Bilateral comparison of the efficacy and tolerability of 3% diclofenac sodium gel and 5% 5-fluorouracil cream in the treatment of actinic keratoses of the face and scalp. J Drugs Dermatol. 2006;5:156-159.
- Segatto MM, Dornelles SI, Silveira VB, et al. Comparative study of actinic keratosis treatment with 3% diclo- fenac sodium and 5% 5-fluorouracil. An Bras Dermatol. 2013;88:732-738.
- Vidal D. Topical imiquimod: mechanism of action and clinical applications. Mini Rev Med Chem. 2006;6:499-503.
- Hadley G, Derry S, Moore RA. Imiquimod for actinic keratosis: systematic review and meta-analysis. J Invest Dermatol. 2006;126:1251-1255.
- Caperton C, Berman B. Safety, efficacy, and patient acceptability of imiquimod for topical treatment of actinic keratoses. Clin Cosmet Investig Dermatol. 2011;4:35-40.
- Swanson N, Smith CC, Kaur M, et al. Imiquimod 2.5% and 3.75% for the treatment of actinic keratoses: two phase 3, multicenter, randomized, double-blind, placebo-controlled studies. J Drugs Dermatol. 2014;13:166-169.
- Krawtchenko N, Roewert-Huber J, Ulrich M, et al. A randomised study of topical 5% imiquimod vs. topical 5-fluorouracil vs. cryosurgery in immunocompetent patients with actinic keratoses: a comparison of clinical and histological outcomes including 1-year follow-up. Br J Dermatol. 2007;157(suppl 2):34-40.
- Anderson L, Schmieder GJ, Werschler WP, et al. Randomized, double-blind, double-dummy, vehicle-controlled study of ingenol mebutate gel 0.025% and 0.05% for actinic keratosis. J Am Acad Dermatol. 2009;60:934-943.
- Ogbourne SM, Suhrbier A, Jones B, et al. Antitumor activity of 3-ingenyl angelate: plasma membrane and mitochondrial disruption and necrotic cell death. Cancer Res. 2004;64:2833-2839.
- Lebwohl M, Swanson N, Anderson LL, et al. Ingenol mebutate gel for actinic keratosis. N Engl J Med. 2012;366:1010-1019.
- Lebwohl M, Shumack S, Stein-Gold L, et al. Long-term follow-up study of ingenol mebutate gel for the treatment of actinic keratoses. JAMA Dermatol. 2013;149:666-670.
- Augustin M, Tu JH, Knudsen KM, et al. Ingenol mebutate gel for actinic keratosis: the link between quality of life, treatment satisfaction, and clinical outcomes. J Am Acad Dermatol. 2015;72:816-821.
- Kane-Maguire N, Moseley R, Cozzi S, et al. Modulation of fibroblast phenotype and extracellular matrix composition by ingenol mebutate may be associated with scar resolution and improved dermal cosmesis. J Am Acad Dermatol. 2012;66:AB218.
- Martin G, Swanson N. Clinical findings using ingenol mebutate gel to treat actinic keratoses. J Am Acad Dermatol. 2013;68(1, suppl 1):S39-S48.
- Feldman SR, Fleischer AB, Williford PM, et al. Destructive procedures are the standard of care for treatment of actinic keratoses. J Am Acad Dermatol. 1999;40:43-47.
- Berlin JM. Current and emerging treatment strategies for the treatment of actinic keratosis. Clin Cosmet Investig Dermatol. 2010;3:119-126.
- Coleman WP, Yarborough JM, Mandy SH. Dermabrasion for prophylaxis and treatment of actinic keratoses. Dermatol Surg. 1996;22:17-21.
- Cooley JE, Casey DL, Kauffman CL. Manual resurfacing and trichloroacetic acid for the treatment of patients with widespread actinic damage. clinical and histologic observations. Dermatol Surg. 1997;23:373-379.
- Goldberg LH, Kaplan B, Vergilis-Kalner I, et al. Liquid nitrogen: temperature control in the treatment of actinic keratosis. Dermatol Surg. 2010;36:1956-1961.
- Lubritz RR, Smolewski SA. Cryosurgery cure rate of actinic keratoses. J Am Acad Dermatol. 1982;7:631-632.
- Thai KE, Fergin P, Freeman M, et al. A prospective study of the use of cryosurgery for the treatment of actinic keratoses. Int J Dermatol. 2004;43:687-692.
- Zane C, Facchinetti E, Rossi MT, et al. Cryotherapy is preferable to ablative CO2 laser for the treatment of isolated actinic keratoses of the face and scalp: a randomized clinical trial. Br J Dermatol. 2014;170:1114-1121.
- Trimas SJ, Ellis DA, Metz RD. The carbon dioxide laser. an alternative for the treatment of actinically damaged skin. Dermatol Surg. 1997;23:885-889.
- Jiang SB, Levine VJ, Nehal KS, et al. Er:YAG laser for the treatment of actinic keratoses. Dermatol Surg. 2000;26:437-440.
- Ostertag JU, Quaedvlieg PJ, Van der geer S, et al. A clinical comparison and long-term follow-up of topical 5-fluorouracil versus laser resurfacing in the treatment of widespread actinic keratoses. Lasers Surg Med. 2006;38:731-739.
- Iyer S, Friedli A, Bowes L, et al. Full face laser resurfacing: therapy and prophylaxis for actinic keratoses and non-melanoma skin cancer. Lasers Surg Med. 2004;34:114-119.
- Rubin MG. A peeler’s thoughts on skin improvement with chemical peels and laser resurfacing. Clin Plast Surg. 1997;24:407-409.
- Riggs K, Keller M, Humphreys TR. Ablative laser resurfacing: high-energy pulsed carbon dioxide and erbium:yttrium-aluminum-garnet. Clin Dermatol. 2007;25:462-473.
- Adrian RM. Pulsed carbon dioxide and long pulse 10-ms erbium-YAG laser resurfacing: a comparative clinical and histological study. J Cutan Laser Ther. 1999;1:197-202.
- Weiss ET, Brauer JA, Anolik R, et al. 1927-nm fractional resurfacing of facial actinic keratoses: a promising new therapeutic option. J Am Acad Dermatol. 2013; 68:98-102.
- Manstein D, Herron GS, Sink RK, et al. Fractional photothermolysis: a new concept for cutaneous remodeling using microscopic patterns of thermal injury. Lasers Surg Med. 2004;34:426-438.
- Katz TM, Goldberg LH, Marquez D, et al. Nonablative fractional photothermolysis for facial actinic keratoses: 6-month follow-up with histologic evaluation. J Am Acad Dermatol. 2011;65:349-356.
- Prens SP, De Vries K, Neumann HA, et al. Non-ablative fractional resurfacing in combination with topical tretinoin cream as a field treatment modality for multiple actinic keratosis: a pilot study and a review of other field treatment modalities. J Dermatolog Treat. 2013;24:227-231.
- Alexiades-Armenakas MR, Dover JS, Arndt KA. The spectrum of laser skin resurfacing: nonablative, fractional, and ablative laser resurfacing. J Am Acad Dermatol. 2008;58:719-737.
- Tannous Z. Fractional resurfacing. Clin Dermatol. 2007;25:480-486.
- Gold MH. Continuing medical education article-skin treatment: photodynamic therapy: indications and treatment. Aesthet Surg J. 2008;28:545-552.
- Juarranz A, Jaén P, Sanz-Rodríguez F, et al. Photodynamic therapy of cancer. basic principles and applications. Clin Transl Oncol. 2008;10:148-154.
- Juzeniene A, Peng Q, Moan J. Milestones in the development of photodynamic therapy and fluorescence diagnosis. Photochem Photobiol Sci. 2007;6:1234-1245.
- Moan J, Berg K. The photodegradation of porphyrins in cells can be used to estimate the lifetime of singlet oxygen. Photochem Photobiol. 1991;53:549-553.
- Gupta AK, Paquet M, Villanueva E, et al. Interventions for actinic keratoses. Cochrane Database Syst Rev. 2012;12:CD004415.
- Patel G, Armstrong AW, Eisen DB. Efficacy of photodynamic therapy vs other interventions in randomized clinical trials for the treatment of actinic keratoses: a systematic review and meta-analysis. JAMA Dermatol. 2014;150:1281-1288.
- Kaufmann R, Spelman L, Weightman W, et al. Multicentre intraindividual randomized trial of topical methyl aminolaevulinate-photodynamic therapy vs. cryotherapy for multiple actinic keratoses on the extremities. Br J Dermatol. 2008;158:994-999.
- Freeman M, Vinciullo C, Francis D, et al. A comparison of photodynamic therapy using topical methyl aminolevulinate (Metvix) with single cycle cryotherapy in patients with actinic keratosis: a prospective, randomized study. J Dermatolog Treat. 2003;14:99-106.
- Morton C, Campbell S, Gupta G, et al. Intraindividual, right-left comparison of topical methyl aminolaevulinate-photodynamic therapy and cryotherapy in subjects with actinic keratoses: a multicentre, randomized controlled study. Br J Dermatol. 2006;155:1029-1036.
- Pariser DM, Lowe NJ, Stewart DM, et al. Photodynamic therapy with topical methyl aminolevulinate for actinic keratosis: results of a prospective randomized multicenter trial. J Am Acad Dermatol. 2003;48:227-232.
- Zane C, Facchinetti E, Rossi MT, et al. A randomized clinical trial of photodynamic therapy with methyl aminolaevulinate vs. diclofenac 3% plus hyaluronic acid gel for the treatment of multiple actinic keratoses of the face and scalp. Br J Dermatol. 2014;170:1143-1150.
- Perrett CM, McGregor JM, Warwick J, et al. Treatment of post-transplant premalignant skin disease: a randomized intrapatient comparative study of 5-fluorouracil cream and topical photodynamic therapy. Br J Dermatol. 2007;156:320-328.
- Szeimies RM, Karrer S, Radakovic-Fijan S, et al. Photodynamic therapy using topical methyl 5-aminolevulinate compared with cryotherapy for actinic keratosis: a prospective, randomized study. J Am Acad Dermatol. 2002; 47:258-262.
- Scola N, Terras S, Georgas D, et al. A randomized, half-side comparative study of aminolaevulinate photodynamic therapy vs. CO(2) laser ablation in immunocompetent patients with multiple actinic keratoses. Br J Dermatol. 2012;167:1366-1373.
- Willey A, Anderson RR, Sakamoto FH. Temperature-modulated photodynamic therapy for the treatment of actinic keratosis on the extremities: a pilot study. Dermatol Surg. 2014;40:1094-1102.
- Pariser DM. Management of Actinic Keratoses: Treatment Selection and Optimizing Outcomes. Presented at: Winter Clinical Dermatology Conference Hawaii; January 18, 2015; Kaanapali, HI.
- Dirschka T, Radny P, Dominicus R, et al. Long-term (6 and 12 months) follow-up of two prospective, randomized, controlled phase III trials of photodynamic therapy with BF-200 ALA and methyl aminolaevulinate for the treatment of actinic keratosis. Br J Dermatol. 2013;168:825-836.
- Choi SH, Kim KH, Song KH. Efficacy of ablative fractional laser-assisted photodynamic therapy with short-incubation time for the treatment of facial and scalp actinic keratosis: 12-month follow-up results of a randomized, prospective, comparative trial. J Eur Acad Dermatol Venereol. 2015;29:1598-1605.
- Ko DY, Jeon SY, Kim KH, et al. Fractional erbium:YAG laser-assisted photodynamic therapy for facial actinic keratoses: a randomized, comparative, prospective study. J Eur Acad Dermatol Venereol. 2014;28:1529-1539.
- Togsverd-Ho K, Haak CS, Thaysen-Petersen D, et al. Intensified photodynamic therapy of actinic keratoses with fractional CO2 laser: a randomized clinical trial. Br J Dermatol. 2012;166:1262-1269.
- Torezan L, Chaves Y, Niwa A, et al. A pilot split-face study comparing conventional methyl aminolevulinate-photodynamic therapy (PDT) with microneedling-assisted PDT on actinically damaged skin. Dermatol Surg. 2013;39:1197-1201.
Actinic keratosis (AK), also referred to as solar keratosis or senile keratosis, is an intraepidermal proliferation of dysplastic keratinocytes that develops in response to chronic exposure to UV radiation. Actinic keratoses are among the most commonly encountered lesions seen by dermatologists, and it has been estimated that 60% of predisposed individuals older than 40 years have at least one AK.1,2 Prevalence is notably higher in light-skinned individuals and increases with age, presumably from higher cumulative sun exposure and decreased effectiveness of the immune system.1,3 It remains a point of contention as to whether or not AKs actually represent squamous cell carcinoma (SCC) in situ, but the potential for progression to invasive disease has been well demonstrated, as the majority of SCCs develop from preexisting AKs.4-6 The risk for progression to invasive disease for an individual AK has been estimated to range from 0.025% to 16% per year, with an average of approximately 8% in immunocompetent patients.7
The clinical morphology of AK can vary widely, but the most common presentation is an erythematous scaly macule, papule, or plaque on sun-exposed skin. The skin surrounding AKs typically shows evidence of solar damage with deep wrinkling, mottled pigmentation, scattered telangiectases, purpura, or xerosis (Figure). A variety of clinical variants with unique presentations exist, including atrophic, hypertrophic, acantholytic, lichenoid, bowenoid, and pigmented subtypes. Because more than 80% of AKs occur on highly visible areas such as the head, neck, back of the hands, and forearms, AKs can have an obvious detrimental effect on cosmetic appearance. Studies also have shown a strong association between AKs and decreased overall quality of life (QOL).3,8,9

Because of the risk for AK progression to invasive cancer along with its negative impact on cosmesis and QOL, clinicians generally opt to treat AKs. Numerous different treatment options exist, including topical medications, procedural modalities, and light-based therapies. Here, we review the efficacy of the most commonly utilized treatments and discuss the relevant cosmetic considerations and outcomes.
Topical Treatments
5-Fluorouracil
5-Fluorouracil (5-FU) is a US Food and Drug Administration (FDA)–approved, topically applied pyrimidine analogue that inhibits thymidylate synthase. The resulting suppression of DNA and RNA synthesis induces cell death with a preference for mitotically active cells.10 5-Fluorouracil has been used for more than 50 years as a treatment of AK and its efficacy is well established. A systematic review of 5 randomized controlled studies of topical 5-FU reported an average of 49% of 423 patients achieving complete lesion clearance with 5-FU cream 5% applied once or twice daily for up to 7 weeks.11 Some notable drawbacks of 5-FU, however, are application-site erythema, blistering, pruritus, necrosis, erosion, and pain. These effects often lead to premature cessation of therapy, but newer formulations of 5-FU cream 0.5% have shown good efficacy with better tolerability.12 A randomized, double-blind, multicenter, parallel-group study of 177 patients using 5-FU cream 0.5% once daily for either 1, 2, or 4 weeks demonstrated significant (P<.001) efficacy over vehicle gel in all treatment arms.13 The most effective therapy was 4 weeks of treatment, which achieved a mean 91.7% reduction in lesion count as assessed 1 month after cessation of therapy. The primary adverse effect (AE) reported in this trial was mild to moderate facial irritation, which generally resolved within 18 to 21 days after treatment cessation.13 Overall, 5-FU is a highly effective therapy for treating AKs that also can improve signs of photoaging, but patients should be aware of cosmetically unappealing effects that generally occur throughout therapy and during the immediate posttreatment period.14
Chemical Peels
Chemical peels traditionally employ acidic compounds to strip away outer layers of skin to variable depths depending on the concentration of the agent being applied. For treatment of AK, trichloroacetic acid (TCA) is a commonly employed cauterant that has shown efficacy comparable to topical 5-FU as well as ablative CO2 laser resurfacing.15 Trichloroacetic acid peels also are a convenient therapy, as good results can be achieved after a single treatment session. A split-face study of 15 patients treated with either a single application of 35% TCA and Jessner solution or twice-daily application of 5-FU cream 5% for 3 weeks demonstrated a reduction in 75% of visible AKs in both treatment arms over a 1-year follow-up period.16 Although 80% of patients self-reported considerable cosmetic improvement with both therapies, patient preference was reported to be in favor of the TCA peel, given its quick results and relatively mild side effects as compared to 5-FU. Treatment with chemical peels will result in temporary erythema and mild desquamation that usually resolves within 2 weeks; however, there are cases in which erythema has been reported to persist for several months.16 Adverse effects such as permanent scarring or pigmentation changes rarely are seen with TCA concentrations less than 45%.17 Caution should be used in patients with a history of herpes simplex virus, keloids, postinflammatory hyperpigmentation, radiation exposure, immunosuppression, and those unable or unwilling to use sunscreen and avoid sun exposure in the immediate posttreatment period.
Diclofenac Sodium
Diclofenac sodium (DFS) is an FDA-approved topical, nonsteroidal,
anti-inflammatory drug whose mechanism of action in the treatment of AK is thought to involve inhibition of the cyclooxygenase 2 enzyme.18 The resulting reduction of prostaglandins is believed to inhibit tumor angiogenesis, induce apoptosis, and inhibit cell differentiation.19-22 In a multicenter, double-blind, placebo-controlled study of 195 patients, application of DFS 3% in hyaluronan gel 2.5% twice daily for 60 days showed significant (P<.05) efficacy over placebo in achieving complete resolution of target lesions during a 30-day follow-up period (31% vs 10%). Furthermore, qualitative patient assessment of complete global improvement also was significantly (P<.05) higher in the active treatment group as compared to placebo (31% vs 10%).23 Additional studies of DFS 3% in hyaluronan gel 2.5% applied twice daily for 90 days have shown even higher rates of success, with complete resolution of target lesions in 40% to 58% of cases.24,25 This therapy also has been reported to substantially improve QOL following treatment completion.26 The most frequently cited AEs include pruritus, rash, dry skin, erythema, and application-site reactions. Overall, DFS is a
well-tolerated therapy with efficacy comparable to that of 5-FU but with a lower incidence of AEs
and higher patient satisfaction as determined in
2 head-to-head studies.27,28
ImiquimodImiquimod (IMQ) is an FDA-approved topical agent that functions as an immune response modifier via agonism of toll-like receptor 7.18 The resulting cytokine production and release enhances the innate and acquired immune responses leading to anticancer activity.29 The efficacy of IMQ for treatment of AK has been demonstrated in numerous well-designed clinical trials. A
meta-analysis of 5 randomized, double-blind trials including 1293 patients treated with IMQ cream 5%
2 to 3 times per week for 12 to 16 weeks reported complete clearance of AKs in 50% of patients treated with IMQ as compared to 5% of patients treated with vehicle.30 The most frequently reported AEs with this therapy include erythema, scabbing, flaking, and erosion. These effects generally resolve following cessation of treatment, and therapy is considered to be well tolerated; however, there are case reports of IMQ triggering or exacerbating existing inflammatory conditions.31 Imiquimod cream also is approved at 2.5% and 3.75% concentrations, which have demonstrated significant (P<.001) efficacy over placebo and a reduced incidence of AEs; complete clearance rates have been reported as 30.6% and 35.6%, respectively.32 Notably, a study comparing 75 patients randomized to either IMQ cream 5%
3 times per week for 4 weeks, 1 or 2 courses of cryosurgery, or 5-FU ointment 5% twice daily for 4 weeks reported that IMQ achieved significantly (P<.01) superior sustained clearance rates during a 12-month follow-up period over cryosurgery and 5-FU
(73% vs 4% vs 33%).33 Additionally, cosmetic outcomes as determined by both participants and investigators were reported as excellent at 12 months posttreatment in more than 80% of participants treated with IMQ. These excellent, long-lasting cosmetic outcomes also were determined to be significantly (P<.0001) superior to the cosmetic outcomes of 5-FU and cryotherapy, which both reported excellent outcomes in less than 10% of cases.33
Ingenol MebutateIngenol mebutate (IM) is a macrocyclic diterpene ester derived from the Euphorbia peplus plant that is FDA approved for the treatment of AK.1 Ingenol mebutate’s mechanism of action is thought to involve induction of cell death via disruption of the plasma membrane and mitochondria in addition to production of an inflammatory response, which produces tumor-specific antibodies and a large influx of neutrophils.34,35 The overall evidence for the efficacy of IM is strong. A combined analysis of 4 multicenter, randomized, double-blind studies of 1005 participants reported that IM gel 0.015% applied once daily for 3 days to the face or scalp was significantly superior (P<.001) to placebo in achieving complete clearance as assessed 54 days after completion of therapy (42.2% vs 3.7%) and that IM gel 0.05% applied once daily for 2 days to the trunk or extremities also was significantly superior (P<.001) to placebo in achieving complete clearance as determined 55 days after completion of therapy (34.1% vs 4.7%).36 A follow-up report to this study indicated that IM also appears to achieve long-lasting effects with an overall 87% decrease in total AKs at 12 months follow-up in both trial groups.37 Additionally, it has been recently reported that treatment with IM in these trials was associated with significantly higher overall treatment satisfaction (P<.001) and improved QOL (P<.001) as compared to vehicle.38 Cosmetic outcomes of IM therapy have been assessed in a trial analyzing the efficacy of IM gel 0.025% for 3 days or IM gel 0.05% for 2 or 3 days on nonfacial AKs. This study reported significantly (P<.0001) higher patient satisfaction with the cosmetic outcome at 8 weeks after therapy as compared to vehicle.34 Studies performed in mice have demonstrated that IM is able to promote collagen matrix turnover and impose dermal elasticity, which may contribute to these good cosmetic outcomes.39 The most common AEs of IM therapy are erythema, crusting, and flaking; these effects generally occur 3 to 8 days after starting treatment. These effects, however, generally are short lived and resolve within 2 weeks of treatment cessation when IM is applied to the face or scalp or 4 weeks when applied to the trunk or extremities.40 Overall, IM is a useful therapeutic option given its relatively short treatment course as compared to other topically applied agents, as well as its lasting efficacy, mild AEs, and good cosmetic outcomes.
Procedural Modalities
Surgical Procedures
Surgical approaches for the treatment of AK include excision, curettage with or without electrodesiccation, and dermabrasion. In the past, these modalities were used with greater frequency, but the advent of effective topical medications with lower risks of AEs has largely reduced their use.41 Excision may still be indicated in cases where SCC is suspected, and curettage can be used for treatment of thicker hypertrophic AKs.42 Although these approaches have not been evaluated in clinical trials, they are generally effective but require the use of local anesthetics and come with substantial risk for infection, permanent scarring, and hypopigmentation. Dermabrasion employs the use of a motorized device equipped with an abrasive material to physically remove superficial layers of the skin. Studies are limited, but this method has been reported as an effective treatment in a retrospective review of 23 participants in which 96% remained free of AKs at 1 year, 83% at 2 years, 64% at 4 years, and 54% at 5 years posttherapy.43 Notably, one split-face study of 40 participants treated with dermabrasion followed by 25% TCA on one side and either Jessner solution and 35% TCA or dermabrasion alone on the other side reported that the combination of dermabrasion with 25% TCA consistently produced excellent cosmetic results with nearly complete eradication of AKs.44 In general, however, cosmetic outcomes with dermabrasion are variable, as the technique is highly operator dependent and treatment is associated with notable discomfort as well as risk for scarring and permanent pigmentation alteration.
Cryotherapy
Cryotherapy remains one of the most commonly utilized treatments of AK and involves the delivery of liquid nitrogen via a spray device or a cotton tip applicator to rapidly freeze cells, thus causing cellular destruction via ice crystal formation and protein denaturation.45 Efficacy with this technique has been reported to be as high as 98.8% at 12 months follow-up, but more recent studies cite lower rates of success.46 A prospective multicenter study of 90 participants with 421 AKs on the face or scalp treated with a single freeze-thaw cycle of liquid nitrogen reported an overall complete response rate of 67.2% at 3 months posttherapy. Additionally, higher complete response rates were associated with longer freeze times, and cosmetic outcomes were reported as good to excellent in 94% of complete response lesions.47 Similar results were reported in an open-label, prospective, randomized, controlled clinical trial of 200 participants with 543 AKs, which compared a single freeze-thaw cycle with liquid nitrogen to a single session of CO2 laser ablation in the treatment of isolated AKs of the face and scalp.48 At 3 months posttherapy, complete clearance was observed in 71.6% of participants treated with cryotherapy and in 65.3% of participants treated with laser ablation (P=.532). At 12 months posttherapy, participants who originally showed complete response at 3 months were assessed for relapse. Complete clearance was preserved in 72.6% of participants treated with cryotherapy versus 21.9% of participants treated with laser ablation (P<.0001), and cosmetic outcomes were reported by participants as good or excellent at 3 months follow-up in more than 93% of participants for both treatment arms.48 Possible AEs of cryotherapy include pain during treatment, blister formation with possible hemorrhage, infection, scarring, and permanent pigmentary changes.47,48 Notably, the risk for hypopigmentation increases with longer freezing times, thus requiring clinicians to consider the balance between improved efficacy and reduced cosmetic outcomes.47
Light-Based Therapies
Laser Therapy
Ablative laser resurfacing with either the CO2 or erbium-doped:YAG (Er:YAG) laser utilizes light of specific wavelengths to selectively induce thermolysis and destruction of the epidermal layer. Both lasers have been studied as treatments of AK, but there is a lack of large, well-designed studies. In one small study of 14 participants treated with 1 to 2 passes of the CO2 laser, complete clearance was reported in all cases without any recurrences during a follow-up period of 6 to 24 months. Additionally, all participants in this study reported satisfaction with the cosmetic outcome.49 The CO2 laser also has demonstrated efficacy comparable to that of the TCA peel and 5-FU therapy in a prospective randomized trial of 34 patients with facial or scalp AKs who received either CO2 laser with 2 passes, 30% TCA peel, or 5-FU cream 5% twice daily for 3 weeks.15 Reduction in mean AK counts at 3 months posttherapy was significantly (P<.03) higher in all treatment arms as compared to the control group (92% for CO2 laser, 89% for TCA peel, and 83% for 5-FU cream). No significant (P=.31) difference in outcomes was noted among the different treatment arms.15 Similar results were reported for the Er:YAG laser in a small prospective study of 5 participants treated with 2 to 3 passes with the Er:YAG laser in which reduction in mean AK counts was reported as ranging from 86% to 96% at 3 months posttherapy.50 The Er:YAG laser in combination with the CO2 laser has shown notable long-term efficacy in achieving higher lesion clearance rates and sustained complete clearance rates over treatment with topical 5-FU.51 In a prospective randomized study of 55 par-ticipants with multiple AKs on the face or scalp, participants were assigned to receive either combination laser ablation with the Er:YAG and CO2 lasers down to the level of the papillary dermis or 5-FU cream 5% applied twice daily for 2 to 7 weeks until an appropriate clinical inflammatory response was achieved. At 12 months follow-up, the laser treatment group achieved significantly (P=.048) higher mean lesion clearance rates (91.1%) as compared to the 5-FU arm (76.6%) and significantly (P=.003) higher sustained complete clearance rates (59.3%) as compared to 5-FU (29.2%). The proportion of participants with an improvement in photoaging score at 12 months follow-up approached statistical significance (P=.07), with 74% of the laser-treated group showing improvement as compared to 43% of the 5-FU–treated group. Long-term, cosmetically unappealing side effects such as erythema and hypopigmentation occurred notably more often in the laser-treated group as compared to the 5-FU group.51 In summary, ablative lasers appear to be a highly effective therapy for AK but at the cost of increased risk for AEs such as permanent pigmentary changes, prolonged erythema lasting up to several months, and scarring.50,52-55
Fractional photothermolysis is a relatively new advancement in the field of laser therapy that has received FDA approval for the treatment of AK.56 This treatment works by creating multiple noncontiguous microscopic columns of thermal injury while sparing adjacent zones of viable tissue.57 Although there are limited studies involving the use of such lasers in the treatment of AK, initial findings suggest that 1927-nm thulium lasers may be more effective than 1550-nm erbium lasers in achieving lesion clearance. A trial of 14 participants who received 5 laser treatments with a 1550-nm fractionated erbium-doped fiber laser reported an average reduction in AK counts of 66.2% at 3 months follow-up and a 55.6% reduction at 6 months follow-up. A participant-determined marked or very significant improvement of lesions was reported in 83% of participants at 1 month posttreatment but only in 44% of participants at 6 months posttreatment.58 A similar trial of 24 participants treated with up to 4 treatment sessions of the fractionated 1927-nm thulium laser reported an 87.3% reduction in number of AKs at 3 months follow-up and an 86.6% reduction at 6 months follow-up.56 The primary advantage of fractional laser therapy is a faster recovery period generally lasting only 2 or 3 days as compared to 2 weeks or more with traditional ablative lasers, thus limiting the amount of time a patient must tolerate cosmetically unappealing erythema.59,60 The quick recovery time has been attributed to the fractional laser’s ability to preserve the stratum corneum and skin barrier, which also helps reduce the risk for other AEs such as scarring and infection.56,59-61 Additional studies are needed to better assess the true efficacy of fractional laser therapy, but treatment with the fractional 1927-nm thulium laser appears to be a promising and well-tolerated therapeutic option for treatment of AK with similar efficacy to traditional ablative lasers but with a lower risk of AEs.
Photodynamic TherapyPhotodynamic therapy (PDT) is an FDA-approved treatment that involves the use of a topical photosensitizing agent such as 5-aminolevulinic acid (ALA) or methyl aminovulinate (MAL) before exposure to an activating light source to generate reactive oxygen species that lead to cell death.62-65 Multiple PDT regimens with varying combinations of photosensitizers, incubation time, and light sources have been studied, but a 2012 Cochrane review determined that treatment with conventional formulations of MAL and ALA with either blue- or red-light PDT were similarly efficacious for treatment of individual AKs as compared to vehicle with blue- or red-light PDT. One exception was that longer incubation time (ie, 4 hours) with ALA resulted in better results than shorter incubation times (ie, 0.5, 1, 2 hours) with ALA.66
Standard PDT treatment with MAL also has consistently demonstrated superior efficacy in achieving complete clearance rates in addition to superior cosmetic outcomes over treatment with either cryotherapy, DFS, or 5-FU.67-73 Three studies in particular noted an excellent or good investigator-determined cosmetic outcome in 96% to 98% of participants treated with MAL-PDT.69,71,74 Photodynamic therapy with ALA also has been reported as superior over CO2 laser ablation for AK reduction as well as both patient and investigator overall satisfaction.75
More recently, several methods of improving photosensitizer delivery have been studied, which have demonstrated remarkable efficacy at achieving lesion clearance over standard cream formulations or application routines. One such method involves the use of gentle heating to increase photosensitizer uptake. In a split-extremity study of 20 participants who were treated with 20% ALA under occlusion for 1 hour with one side heated to 38.8°C, the heated side demonstrated significant (P<.0001) efficacy at achieving higher median clearance rates over control when evaluated at 2 and 6 months posttherapy.76 Notably, occlusion of ALA in itself during the incubation period also has been demonstrated to significantly (P<.0001) improve clearance rates.77 Another method involves the use of a new nanoemulsion-based formulation of ALA gel, known as BF-200 ALA, which has demonstrated remarkable efficacy over standard MAL cream and placebo in a long-term follow-up analysis of 2 prospective, randomized, controlled trials.78 In a similar vein, 3 prospective randomized trials with a minimum follow-up time of 3 months demonstrated that MAL-PDT in combination with fractional ablative laser pretreatment has significant (P<.02 in all trials) efficacy over MAL-PDT without pretreatment in achieving complete AK clearance. Although the cosmetic outcomes were good or excellent in 87% to 100% of patients, they were not significantly different from stand-alone MAL-PDT treatment in any of the trials.79-81 However, pretreatment with microneedling in MAL-PDT has been shown to achieve superior cosmetic outcomes over MAL-PDT without microneedling, according to one small split-face study of 10 participants.82
Overall, PDT is an excellent therapeutic option that is able to provide efficacious clearance of AKs as well as superior cosmetic outcomes. Common AEs of PDT include burning, itching, and stinging during therapy, but pain intensity decreases dramatically upon termination of illumination, with cessation of most symptoms by 12 hours posttherapy.73 Permanent pigmentation changes have been reported to occasionally occur following PDT therapy.81
Conclusion
When determining which therapy to use in a patient, clinicians must take into account a variety of factors such as patient preference, cost of treatment, availability, tolerance for AEs, and the need for field therapy. Although all therapies discussed within this article are effective and reasonable treatment choices, patients who are particularly concerned about cosmetic outcomes would most likely benefit from either IMQ or PDT, as the data for cosmetic outcomes with these therapies are the strongest. Combination or sequential treatments may be required in some cases and all patients should be monitored for lesion recurrence regardless of treatment choice. A summary of the therapies and key studies discussed here is available in the PDF.
Actinic keratosis (AK), also referred to as solar keratosis or senile keratosis, is an intraepidermal proliferation of dysplastic keratinocytes that develops in response to chronic exposure to UV radiation. Actinic keratoses are among the most commonly encountered lesions seen by dermatologists, and it has been estimated that 60% of predisposed individuals older than 40 years have at least one AK.1,2 Prevalence is notably higher in light-skinned individuals and increases with age, presumably from higher cumulative sun exposure and decreased effectiveness of the immune system.1,3 It remains a point of contention as to whether or not AKs actually represent squamous cell carcinoma (SCC) in situ, but the potential for progression to invasive disease has been well demonstrated, as the majority of SCCs develop from preexisting AKs.4-6 The risk for progression to invasive disease for an individual AK has been estimated to range from 0.025% to 16% per year, with an average of approximately 8% in immunocompetent patients.7
The clinical morphology of AK can vary widely, but the most common presentation is an erythematous scaly macule, papule, or plaque on sun-exposed skin. The skin surrounding AKs typically shows evidence of solar damage with deep wrinkling, mottled pigmentation, scattered telangiectases, purpura, or xerosis (Figure). A variety of clinical variants with unique presentations exist, including atrophic, hypertrophic, acantholytic, lichenoid, bowenoid, and pigmented subtypes. Because more than 80% of AKs occur on highly visible areas such as the head, neck, back of the hands, and forearms, AKs can have an obvious detrimental effect on cosmetic appearance. Studies also have shown a strong association between AKs and decreased overall quality of life (QOL).3,8,9

Because of the risk for AK progression to invasive cancer along with its negative impact on cosmesis and QOL, clinicians generally opt to treat AKs. Numerous different treatment options exist, including topical medications, procedural modalities, and light-based therapies. Here, we review the efficacy of the most commonly utilized treatments and discuss the relevant cosmetic considerations and outcomes.
Topical Treatments
5-Fluorouracil
5-Fluorouracil (5-FU) is a US Food and Drug Administration (FDA)–approved, topically applied pyrimidine analogue that inhibits thymidylate synthase. The resulting suppression of DNA and RNA synthesis induces cell death with a preference for mitotically active cells.10 5-Fluorouracil has been used for more than 50 years as a treatment of AK and its efficacy is well established. A systematic review of 5 randomized controlled studies of topical 5-FU reported an average of 49% of 423 patients achieving complete lesion clearance with 5-FU cream 5% applied once or twice daily for up to 7 weeks.11 Some notable drawbacks of 5-FU, however, are application-site erythema, blistering, pruritus, necrosis, erosion, and pain. These effects often lead to premature cessation of therapy, but newer formulations of 5-FU cream 0.5% have shown good efficacy with better tolerability.12 A randomized, double-blind, multicenter, parallel-group study of 177 patients using 5-FU cream 0.5% once daily for either 1, 2, or 4 weeks demonstrated significant (P<.001) efficacy over vehicle gel in all treatment arms.13 The most effective therapy was 4 weeks of treatment, which achieved a mean 91.7% reduction in lesion count as assessed 1 month after cessation of therapy. The primary adverse effect (AE) reported in this trial was mild to moderate facial irritation, which generally resolved within 18 to 21 days after treatment cessation.13 Overall, 5-FU is a highly effective therapy for treating AKs that also can improve signs of photoaging, but patients should be aware of cosmetically unappealing effects that generally occur throughout therapy and during the immediate posttreatment period.14
Chemical Peels
Chemical peels traditionally employ acidic compounds to strip away outer layers of skin to variable depths depending on the concentration of the agent being applied. For treatment of AK, trichloroacetic acid (TCA) is a commonly employed cauterant that has shown efficacy comparable to topical 5-FU as well as ablative CO2 laser resurfacing.15 Trichloroacetic acid peels also are a convenient therapy, as good results can be achieved after a single treatment session. A split-face study of 15 patients treated with either a single application of 35% TCA and Jessner solution or twice-daily application of 5-FU cream 5% for 3 weeks demonstrated a reduction in 75% of visible AKs in both treatment arms over a 1-year follow-up period.16 Although 80% of patients self-reported considerable cosmetic improvement with both therapies, patient preference was reported to be in favor of the TCA peel, given its quick results and relatively mild side effects as compared to 5-FU. Treatment with chemical peels will result in temporary erythema and mild desquamation that usually resolves within 2 weeks; however, there are cases in which erythema has been reported to persist for several months.16 Adverse effects such as permanent scarring or pigmentation changes rarely are seen with TCA concentrations less than 45%.17 Caution should be used in patients with a history of herpes simplex virus, keloids, postinflammatory hyperpigmentation, radiation exposure, immunosuppression, and those unable or unwilling to use sunscreen and avoid sun exposure in the immediate posttreatment period.
Diclofenac Sodium
Diclofenac sodium (DFS) is an FDA-approved topical, nonsteroidal,
anti-inflammatory drug whose mechanism of action in the treatment of AK is thought to involve inhibition of the cyclooxygenase 2 enzyme.18 The resulting reduction of prostaglandins is believed to inhibit tumor angiogenesis, induce apoptosis, and inhibit cell differentiation.19-22 In a multicenter, double-blind, placebo-controlled study of 195 patients, application of DFS 3% in hyaluronan gel 2.5% twice daily for 60 days showed significant (P<.05) efficacy over placebo in achieving complete resolution of target lesions during a 30-day follow-up period (31% vs 10%). Furthermore, qualitative patient assessment of complete global improvement also was significantly (P<.05) higher in the active treatment group as compared to placebo (31% vs 10%).23 Additional studies of DFS 3% in hyaluronan gel 2.5% applied twice daily for 90 days have shown even higher rates of success, with complete resolution of target lesions in 40% to 58% of cases.24,25 This therapy also has been reported to substantially improve QOL following treatment completion.26 The most frequently cited AEs include pruritus, rash, dry skin, erythema, and application-site reactions. Overall, DFS is a
well-tolerated therapy with efficacy comparable to that of 5-FU but with a lower incidence of AEs
and higher patient satisfaction as determined in
2 head-to-head studies.27,28
ImiquimodImiquimod (IMQ) is an FDA-approved topical agent that functions as an immune response modifier via agonism of toll-like receptor 7.18 The resulting cytokine production and release enhances the innate and acquired immune responses leading to anticancer activity.29 The efficacy of IMQ for treatment of AK has been demonstrated in numerous well-designed clinical trials. A
meta-analysis of 5 randomized, double-blind trials including 1293 patients treated with IMQ cream 5%
2 to 3 times per week for 12 to 16 weeks reported complete clearance of AKs in 50% of patients treated with IMQ as compared to 5% of patients treated with vehicle.30 The most frequently reported AEs with this therapy include erythema, scabbing, flaking, and erosion. These effects generally resolve following cessation of treatment, and therapy is considered to be well tolerated; however, there are case reports of IMQ triggering or exacerbating existing inflammatory conditions.31 Imiquimod cream also is approved at 2.5% and 3.75% concentrations, which have demonstrated significant (P<.001) efficacy over placebo and a reduced incidence of AEs; complete clearance rates have been reported as 30.6% and 35.6%, respectively.32 Notably, a study comparing 75 patients randomized to either IMQ cream 5%
3 times per week for 4 weeks, 1 or 2 courses of cryosurgery, or 5-FU ointment 5% twice daily for 4 weeks reported that IMQ achieved significantly (P<.01) superior sustained clearance rates during a 12-month follow-up period over cryosurgery and 5-FU
(73% vs 4% vs 33%).33 Additionally, cosmetic outcomes as determined by both participants and investigators were reported as excellent at 12 months posttreatment in more than 80% of participants treated with IMQ. These excellent, long-lasting cosmetic outcomes also were determined to be significantly (P<.0001) superior to the cosmetic outcomes of 5-FU and cryotherapy, which both reported excellent outcomes in less than 10% of cases.33
Ingenol MebutateIngenol mebutate (IM) is a macrocyclic diterpene ester derived from the Euphorbia peplus plant that is FDA approved for the treatment of AK.1 Ingenol mebutate’s mechanism of action is thought to involve induction of cell death via disruption of the plasma membrane and mitochondria in addition to production of an inflammatory response, which produces tumor-specific antibodies and a large influx of neutrophils.34,35 The overall evidence for the efficacy of IM is strong. A combined analysis of 4 multicenter, randomized, double-blind studies of 1005 participants reported that IM gel 0.015% applied once daily for 3 days to the face or scalp was significantly superior (P<.001) to placebo in achieving complete clearance as assessed 54 days after completion of therapy (42.2% vs 3.7%) and that IM gel 0.05% applied once daily for 2 days to the trunk or extremities also was significantly superior (P<.001) to placebo in achieving complete clearance as determined 55 days after completion of therapy (34.1% vs 4.7%).36 A follow-up report to this study indicated that IM also appears to achieve long-lasting effects with an overall 87% decrease in total AKs at 12 months follow-up in both trial groups.37 Additionally, it has been recently reported that treatment with IM in these trials was associated with significantly higher overall treatment satisfaction (P<.001) and improved QOL (P<.001) as compared to vehicle.38 Cosmetic outcomes of IM therapy have been assessed in a trial analyzing the efficacy of IM gel 0.025% for 3 days or IM gel 0.05% for 2 or 3 days on nonfacial AKs. This study reported significantly (P<.0001) higher patient satisfaction with the cosmetic outcome at 8 weeks after therapy as compared to vehicle.34 Studies performed in mice have demonstrated that IM is able to promote collagen matrix turnover and impose dermal elasticity, which may contribute to these good cosmetic outcomes.39 The most common AEs of IM therapy are erythema, crusting, and flaking; these effects generally occur 3 to 8 days after starting treatment. These effects, however, generally are short lived and resolve within 2 weeks of treatment cessation when IM is applied to the face or scalp or 4 weeks when applied to the trunk or extremities.40 Overall, IM is a useful therapeutic option given its relatively short treatment course as compared to other topically applied agents, as well as its lasting efficacy, mild AEs, and good cosmetic outcomes.
Procedural Modalities
Surgical Procedures
Surgical approaches for the treatment of AK include excision, curettage with or without electrodesiccation, and dermabrasion. In the past, these modalities were used with greater frequency, but the advent of effective topical medications with lower risks of AEs has largely reduced their use.41 Excision may still be indicated in cases where SCC is suspected, and curettage can be used for treatment of thicker hypertrophic AKs.42 Although these approaches have not been evaluated in clinical trials, they are generally effective but require the use of local anesthetics and come with substantial risk for infection, permanent scarring, and hypopigmentation. Dermabrasion employs the use of a motorized device equipped with an abrasive material to physically remove superficial layers of the skin. Studies are limited, but this method has been reported as an effective treatment in a retrospective review of 23 participants in which 96% remained free of AKs at 1 year, 83% at 2 years, 64% at 4 years, and 54% at 5 years posttherapy.43 Notably, one split-face study of 40 participants treated with dermabrasion followed by 25% TCA on one side and either Jessner solution and 35% TCA or dermabrasion alone on the other side reported that the combination of dermabrasion with 25% TCA consistently produced excellent cosmetic results with nearly complete eradication of AKs.44 In general, however, cosmetic outcomes with dermabrasion are variable, as the technique is highly operator dependent and treatment is associated with notable discomfort as well as risk for scarring and permanent pigmentation alteration.
Cryotherapy
Cryotherapy remains one of the most commonly utilized treatments of AK and involves the delivery of liquid nitrogen via a spray device or a cotton tip applicator to rapidly freeze cells, thus causing cellular destruction via ice crystal formation and protein denaturation.45 Efficacy with this technique has been reported to be as high as 98.8% at 12 months follow-up, but more recent studies cite lower rates of success.46 A prospective multicenter study of 90 participants with 421 AKs on the face or scalp treated with a single freeze-thaw cycle of liquid nitrogen reported an overall complete response rate of 67.2% at 3 months posttherapy. Additionally, higher complete response rates were associated with longer freeze times, and cosmetic outcomes were reported as good to excellent in 94% of complete response lesions.47 Similar results were reported in an open-label, prospective, randomized, controlled clinical trial of 200 participants with 543 AKs, which compared a single freeze-thaw cycle with liquid nitrogen to a single session of CO2 laser ablation in the treatment of isolated AKs of the face and scalp.48 At 3 months posttherapy, complete clearance was observed in 71.6% of participants treated with cryotherapy and in 65.3% of participants treated with laser ablation (P=.532). At 12 months posttherapy, participants who originally showed complete response at 3 months were assessed for relapse. Complete clearance was preserved in 72.6% of participants treated with cryotherapy versus 21.9% of participants treated with laser ablation (P<.0001), and cosmetic outcomes were reported by participants as good or excellent at 3 months follow-up in more than 93% of participants for both treatment arms.48 Possible AEs of cryotherapy include pain during treatment, blister formation with possible hemorrhage, infection, scarring, and permanent pigmentary changes.47,48 Notably, the risk for hypopigmentation increases with longer freezing times, thus requiring clinicians to consider the balance between improved efficacy and reduced cosmetic outcomes.47
Light-Based Therapies
Laser Therapy
Ablative laser resurfacing with either the CO2 or erbium-doped:YAG (Er:YAG) laser utilizes light of specific wavelengths to selectively induce thermolysis and destruction of the epidermal layer. Both lasers have been studied as treatments of AK, but there is a lack of large, well-designed studies. In one small study of 14 participants treated with 1 to 2 passes of the CO2 laser, complete clearance was reported in all cases without any recurrences during a follow-up period of 6 to 24 months. Additionally, all participants in this study reported satisfaction with the cosmetic outcome.49 The CO2 laser also has demonstrated efficacy comparable to that of the TCA peel and 5-FU therapy in a prospective randomized trial of 34 patients with facial or scalp AKs who received either CO2 laser with 2 passes, 30% TCA peel, or 5-FU cream 5% twice daily for 3 weeks.15 Reduction in mean AK counts at 3 months posttherapy was significantly (P<.03) higher in all treatment arms as compared to the control group (92% for CO2 laser, 89% for TCA peel, and 83% for 5-FU cream). No significant (P=.31) difference in outcomes was noted among the different treatment arms.15 Similar results were reported for the Er:YAG laser in a small prospective study of 5 participants treated with 2 to 3 passes with the Er:YAG laser in which reduction in mean AK counts was reported as ranging from 86% to 96% at 3 months posttherapy.50 The Er:YAG laser in combination with the CO2 laser has shown notable long-term efficacy in achieving higher lesion clearance rates and sustained complete clearance rates over treatment with topical 5-FU.51 In a prospective randomized study of 55 par-ticipants with multiple AKs on the face or scalp, participants were assigned to receive either combination laser ablation with the Er:YAG and CO2 lasers down to the level of the papillary dermis or 5-FU cream 5% applied twice daily for 2 to 7 weeks until an appropriate clinical inflammatory response was achieved. At 12 months follow-up, the laser treatment group achieved significantly (P=.048) higher mean lesion clearance rates (91.1%) as compared to the 5-FU arm (76.6%) and significantly (P=.003) higher sustained complete clearance rates (59.3%) as compared to 5-FU (29.2%). The proportion of participants with an improvement in photoaging score at 12 months follow-up approached statistical significance (P=.07), with 74% of the laser-treated group showing improvement as compared to 43% of the 5-FU–treated group. Long-term, cosmetically unappealing side effects such as erythema and hypopigmentation occurred notably more often in the laser-treated group as compared to the 5-FU group.51 In summary, ablative lasers appear to be a highly effective therapy for AK but at the cost of increased risk for AEs such as permanent pigmentary changes, prolonged erythema lasting up to several months, and scarring.50,52-55
Fractional photothermolysis is a relatively new advancement in the field of laser therapy that has received FDA approval for the treatment of AK.56 This treatment works by creating multiple noncontiguous microscopic columns of thermal injury while sparing adjacent zones of viable tissue.57 Although there are limited studies involving the use of such lasers in the treatment of AK, initial findings suggest that 1927-nm thulium lasers may be more effective than 1550-nm erbium lasers in achieving lesion clearance. A trial of 14 participants who received 5 laser treatments with a 1550-nm fractionated erbium-doped fiber laser reported an average reduction in AK counts of 66.2% at 3 months follow-up and a 55.6% reduction at 6 months follow-up. A participant-determined marked or very significant improvement of lesions was reported in 83% of participants at 1 month posttreatment but only in 44% of participants at 6 months posttreatment.58 A similar trial of 24 participants treated with up to 4 treatment sessions of the fractionated 1927-nm thulium laser reported an 87.3% reduction in number of AKs at 3 months follow-up and an 86.6% reduction at 6 months follow-up.56 The primary advantage of fractional laser therapy is a faster recovery period generally lasting only 2 or 3 days as compared to 2 weeks or more with traditional ablative lasers, thus limiting the amount of time a patient must tolerate cosmetically unappealing erythema.59,60 The quick recovery time has been attributed to the fractional laser’s ability to preserve the stratum corneum and skin barrier, which also helps reduce the risk for other AEs such as scarring and infection.56,59-61 Additional studies are needed to better assess the true efficacy of fractional laser therapy, but treatment with the fractional 1927-nm thulium laser appears to be a promising and well-tolerated therapeutic option for treatment of AK with similar efficacy to traditional ablative lasers but with a lower risk of AEs.
Photodynamic TherapyPhotodynamic therapy (PDT) is an FDA-approved treatment that involves the use of a topical photosensitizing agent such as 5-aminolevulinic acid (ALA) or methyl aminovulinate (MAL) before exposure to an activating light source to generate reactive oxygen species that lead to cell death.62-65 Multiple PDT regimens with varying combinations of photosensitizers, incubation time, and light sources have been studied, but a 2012 Cochrane review determined that treatment with conventional formulations of MAL and ALA with either blue- or red-light PDT were similarly efficacious for treatment of individual AKs as compared to vehicle with blue- or red-light PDT. One exception was that longer incubation time (ie, 4 hours) with ALA resulted in better results than shorter incubation times (ie, 0.5, 1, 2 hours) with ALA.66
Standard PDT treatment with MAL also has consistently demonstrated superior efficacy in achieving complete clearance rates in addition to superior cosmetic outcomes over treatment with either cryotherapy, DFS, or 5-FU.67-73 Three studies in particular noted an excellent or good investigator-determined cosmetic outcome in 96% to 98% of participants treated with MAL-PDT.69,71,74 Photodynamic therapy with ALA also has been reported as superior over CO2 laser ablation for AK reduction as well as both patient and investigator overall satisfaction.75
More recently, several methods of improving photosensitizer delivery have been studied, which have demonstrated remarkable efficacy at achieving lesion clearance over standard cream formulations or application routines. One such method involves the use of gentle heating to increase photosensitizer uptake. In a split-extremity study of 20 participants who were treated with 20% ALA under occlusion for 1 hour with one side heated to 38.8°C, the heated side demonstrated significant (P<.0001) efficacy at achieving higher median clearance rates over control when evaluated at 2 and 6 months posttherapy.76 Notably, occlusion of ALA in itself during the incubation period also has been demonstrated to significantly (P<.0001) improve clearance rates.77 Another method involves the use of a new nanoemulsion-based formulation of ALA gel, known as BF-200 ALA, which has demonstrated remarkable efficacy over standard MAL cream and placebo in a long-term follow-up analysis of 2 prospective, randomized, controlled trials.78 In a similar vein, 3 prospective randomized trials with a minimum follow-up time of 3 months demonstrated that MAL-PDT in combination with fractional ablative laser pretreatment has significant (P<.02 in all trials) efficacy over MAL-PDT without pretreatment in achieving complete AK clearance. Although the cosmetic outcomes were good or excellent in 87% to 100% of patients, they were not significantly different from stand-alone MAL-PDT treatment in any of the trials.79-81 However, pretreatment with microneedling in MAL-PDT has been shown to achieve superior cosmetic outcomes over MAL-PDT without microneedling, according to one small split-face study of 10 participants.82
Overall, PDT is an excellent therapeutic option that is able to provide efficacious clearance of AKs as well as superior cosmetic outcomes. Common AEs of PDT include burning, itching, and stinging during therapy, but pain intensity decreases dramatically upon termination of illumination, with cessation of most symptoms by 12 hours posttherapy.73 Permanent pigmentation changes have been reported to occasionally occur following PDT therapy.81
Conclusion
When determining which therapy to use in a patient, clinicians must take into account a variety of factors such as patient preference, cost of treatment, availability, tolerance for AEs, and the need for field therapy. Although all therapies discussed within this article are effective and reasonable treatment choices, patients who are particularly concerned about cosmetic outcomes would most likely benefit from either IMQ or PDT, as the data for cosmetic outcomes with these therapies are the strongest. Combination or sequential treatments may be required in some cases and all patients should be monitored for lesion recurrence regardless of treatment choice. A summary of the therapies and key studies discussed here is available in the PDF.
- Lebwohl M. Actinic keratosis: epidemiology and progression to squamous cell carcinoma. Br J Dermatol. 2003;149(suppl 66):31-33.
- Drake LA, Ceilley RI, Cornelison RL, et al. Guidelines of care for actinic keratoses. Committee on Guidelines of Care. J Am Acad Dermatol. 1995;32:95-98.
- Salasche SJ. Epidemiology of actinic keratoses and squamous cell carcinoma. J Am Acad Dermatol. 2000;42(1, pt 2):4-7.
- Ackerman AB, Mones JM. Solar (actinic) keratosis is squamous cell carcinoma. Br J Dermatol. 2006;155:9-22.
- Anwar J, Wrone DA, Kimyai-Asadi A, et al. The development of actinic keratosis into invasive squamous cell carcinoma: evidence and evolving classification schemes. Clin Dermatol. 2004;22:189-196.
- Criscione VD, Weinstock MA, Naylor MF, et al. Actinic keratoses: natural history and risk of malignant transformation in the Veterans Affairs Topical Tretinoin Chemoprevention Trial. Cancer. 2009;115:2523-2530.
- Glogau RG. The risk of progression to invasive disease. J Am Acad Dermatol. 2000;42(1, pt 2):23-24.
- Esmann S, Jemec GB. Management of actinic keratosis patients: a qualitative study. J Dermatolog Treat. 2007;18:53-58.
- Weinstock MA, Lee KC, Chren MM, et al. Quality of life in the actinic neoplasia syndrome: the VA Topical Tretinoin Chemoprevention (VATTC) trial. J Am Acad Dermatol. 2009;61:207-215.
- Berman B, Villa AM, Ramirez CC. Mechanisms of action of new treatment modalities for actinic keratosis. J Drugs Dermatol. 2006;5:167-173.
- Askew DA, Mickan SM, Soyer HP. Effectiveness of 5-fluorouracil treatment for actinic keratosis: a systematic review of randomized controlled trials. Int J Dermatol. 2009;46:452-463.
- Levy S, Furst K, Chern W. A pharmacokinetic evaluation of 0.5% and 5% fluorouracil topical cream in patients with actinic keratosis. Clin Ther. 2001;23:908-920.
- Jorizzo J, Stewart D, Bucko A, et al. Randomized trial evaluating a new 0.5% fluorouracil formulation demonstrates efficacy after 1-, 2-, or 4-week treatment in patients with actinic keratosis. Cutis. 2002;70:335-359.
- Sachs DL, Kang S, Hammerberg C, et al. Topical fluorouracil for actinic keratoses and photoaging: a clinical and molecular analysis. Arch Dermatol. 2009;145:659-666.
- Hantash BM, Stewart DB, Cooper ZA, et al. Facial resurfacing for nonmelanoma skin cancer prophylaxis. Arch Dermatol. 2006;142:976-982.
- Lawrence N, Cox SE, Cockerell CJ, et al. A comparison of the efficacy and safety of Jessner’s solution and 35% trichloroacetic acid vs 5% fluorouracil in the treatment of widespread facial actinic keratoses. Arch Dermatol. 1995;131:176-181.
- Monheit GD. The Jessner’s + TCA peel: a medium-depth chemical peel. J Dermatol Surg Oncol. 1989;15:945-950.
- Hemmi H, Kaisho T, Takeuchi O, et al. Small anti-viral compounds activate immune cells via the TLR7 MyD88-dependent signaling pathway. Nat Immunol. 2002;3:196-200.
- Adamson DJ, Frew D, Tatoud R, et al. Diclofenac antagonizes peroxisome proliferator-activated receptor-gamma signaling. Mol Pharmacol. 2002;61:7-12.
- Alam CA, Seed MP, Willoughby DA. Angiostasis and vascular regression in chronic granulomatous inflammation induced by diclofenac in combination with hyaluronan in mice. J Pharm Pharmacol. 1995;47:407-411.
- Lu X, Xie W, Reed D, et al. Nonsteroidal antiinflammatory drugs cause apoptosis and induce cyclooxygenases in chicken embryo fibroblasts. Proc Natl Acad Sci USA. 1995;92:7961-7965.
- Seed MP, Brown JR, Freemantle CN, et al. The inhibition of colon-26 adenocarcinoma development and angiogenesis by topical diclofenac in 2.5% hyaluronan. Cancer Res. 1997;57:1625-1629.
- Rivers JK, Arlette J, Shear N, et al. Topical treatment of actinic keratoses with 3.0% diclofenac in 2.5% hyaluronan gel. Br J Dermatol. 2002;146:94-100.
- Wolf JE, Taylor JR, Tschen E, et al. Topical 3.0% diclo-fenac in 2.5% hyaluronan gel in the treatment of actinic keratoses. Int J Dermatol. 2001;40:709-713.
- Nelson C, Rigel D, Smith S, et al. Phase IV, open-label assessment of the treatment of actinic keratosis with 3.0% diclofenac sodium topical gel (Solaraze). J Drugs Dermatol. 2004;3:401-407.
- Pflugfelder A, Welter AK, Leiter U, et al. Open label randomized study comparing 3 months vs. 6 months treatment of actinic keratoses with 3% diclofenac in 2.5% hyaluronic acid gel: a trial of the German Dermatologic Cooperative Oncology Group. J Eur Acad Dermatol Venereol. 2012;26:48-53.
- Smith SR, Morhenn VB, Piacquadio DJ. Bilateral comparison of the efficacy and tolerability of 3% diclofenac sodium gel and 5% 5-fluorouracil cream in the treatment of actinic keratoses of the face and scalp. J Drugs Dermatol. 2006;5:156-159.
- Segatto MM, Dornelles SI, Silveira VB, et al. Comparative study of actinic keratosis treatment with 3% diclo- fenac sodium and 5% 5-fluorouracil. An Bras Dermatol. 2013;88:732-738.
- Vidal D. Topical imiquimod: mechanism of action and clinical applications. Mini Rev Med Chem. 2006;6:499-503.
- Hadley G, Derry S, Moore RA. Imiquimod for actinic keratosis: systematic review and meta-analysis. J Invest Dermatol. 2006;126:1251-1255.
- Caperton C, Berman B. Safety, efficacy, and patient acceptability of imiquimod for topical treatment of actinic keratoses. Clin Cosmet Investig Dermatol. 2011;4:35-40.
- Swanson N, Smith CC, Kaur M, et al. Imiquimod 2.5% and 3.75% for the treatment of actinic keratoses: two phase 3, multicenter, randomized, double-blind, placebo-controlled studies. J Drugs Dermatol. 2014;13:166-169.
- Krawtchenko N, Roewert-Huber J, Ulrich M, et al. A randomised study of topical 5% imiquimod vs. topical 5-fluorouracil vs. cryosurgery in immunocompetent patients with actinic keratoses: a comparison of clinical and histological outcomes including 1-year follow-up. Br J Dermatol. 2007;157(suppl 2):34-40.
- Anderson L, Schmieder GJ, Werschler WP, et al. Randomized, double-blind, double-dummy, vehicle-controlled study of ingenol mebutate gel 0.025% and 0.05% for actinic keratosis. J Am Acad Dermatol. 2009;60:934-943.
- Ogbourne SM, Suhrbier A, Jones B, et al. Antitumor activity of 3-ingenyl angelate: plasma membrane and mitochondrial disruption and necrotic cell death. Cancer Res. 2004;64:2833-2839.
- Lebwohl M, Swanson N, Anderson LL, et al. Ingenol mebutate gel for actinic keratosis. N Engl J Med. 2012;366:1010-1019.
- Lebwohl M, Shumack S, Stein-Gold L, et al. Long-term follow-up study of ingenol mebutate gel for the treatment of actinic keratoses. JAMA Dermatol. 2013;149:666-670.
- Augustin M, Tu JH, Knudsen KM, et al. Ingenol mebutate gel for actinic keratosis: the link between quality of life, treatment satisfaction, and clinical outcomes. J Am Acad Dermatol. 2015;72:816-821.
- Kane-Maguire N, Moseley R, Cozzi S, et al. Modulation of fibroblast phenotype and extracellular matrix composition by ingenol mebutate may be associated with scar resolution and improved dermal cosmesis. J Am Acad Dermatol. 2012;66:AB218.
- Martin G, Swanson N. Clinical findings using ingenol mebutate gel to treat actinic keratoses. J Am Acad Dermatol. 2013;68(1, suppl 1):S39-S48.
- Feldman SR, Fleischer AB, Williford PM, et al. Destructive procedures are the standard of care for treatment of actinic keratoses. J Am Acad Dermatol. 1999;40:43-47.
- Berlin JM. Current and emerging treatment strategies for the treatment of actinic keratosis. Clin Cosmet Investig Dermatol. 2010;3:119-126.
- Coleman WP, Yarborough JM, Mandy SH. Dermabrasion for prophylaxis and treatment of actinic keratoses. Dermatol Surg. 1996;22:17-21.
- Cooley JE, Casey DL, Kauffman CL. Manual resurfacing and trichloroacetic acid for the treatment of patients with widespread actinic damage. clinical and histologic observations. Dermatol Surg. 1997;23:373-379.
- Goldberg LH, Kaplan B, Vergilis-Kalner I, et al. Liquid nitrogen: temperature control in the treatment of actinic keratosis. Dermatol Surg. 2010;36:1956-1961.
- Lubritz RR, Smolewski SA. Cryosurgery cure rate of actinic keratoses. J Am Acad Dermatol. 1982;7:631-632.
- Thai KE, Fergin P, Freeman M, et al. A prospective study of the use of cryosurgery for the treatment of actinic keratoses. Int J Dermatol. 2004;43:687-692.
- Zane C, Facchinetti E, Rossi MT, et al. Cryotherapy is preferable to ablative CO2 laser for the treatment of isolated actinic keratoses of the face and scalp: a randomized clinical trial. Br J Dermatol. 2014;170:1114-1121.
- Trimas SJ, Ellis DA, Metz RD. The carbon dioxide laser. an alternative for the treatment of actinically damaged skin. Dermatol Surg. 1997;23:885-889.
- Jiang SB, Levine VJ, Nehal KS, et al. Er:YAG laser for the treatment of actinic keratoses. Dermatol Surg. 2000;26:437-440.
- Ostertag JU, Quaedvlieg PJ, Van der geer S, et al. A clinical comparison and long-term follow-up of topical 5-fluorouracil versus laser resurfacing in the treatment of widespread actinic keratoses. Lasers Surg Med. 2006;38:731-739.
- Iyer S, Friedli A, Bowes L, et al. Full face laser resurfacing: therapy and prophylaxis for actinic keratoses and non-melanoma skin cancer. Lasers Surg Med. 2004;34:114-119.
- Rubin MG. A peeler’s thoughts on skin improvement with chemical peels and laser resurfacing. Clin Plast Surg. 1997;24:407-409.
- Riggs K, Keller M, Humphreys TR. Ablative laser resurfacing: high-energy pulsed carbon dioxide and erbium:yttrium-aluminum-garnet. Clin Dermatol. 2007;25:462-473.
- Adrian RM. Pulsed carbon dioxide and long pulse 10-ms erbium-YAG laser resurfacing: a comparative clinical and histological study. J Cutan Laser Ther. 1999;1:197-202.
- Weiss ET, Brauer JA, Anolik R, et al. 1927-nm fractional resurfacing of facial actinic keratoses: a promising new therapeutic option. J Am Acad Dermatol. 2013; 68:98-102.
- Manstein D, Herron GS, Sink RK, et al. Fractional photothermolysis: a new concept for cutaneous remodeling using microscopic patterns of thermal injury. Lasers Surg Med. 2004;34:426-438.
- Katz TM, Goldberg LH, Marquez D, et al. Nonablative fractional photothermolysis for facial actinic keratoses: 6-month follow-up with histologic evaluation. J Am Acad Dermatol. 2011;65:349-356.
- Prens SP, De Vries K, Neumann HA, et al. Non-ablative fractional resurfacing in combination with topical tretinoin cream as a field treatment modality for multiple actinic keratosis: a pilot study and a review of other field treatment modalities. J Dermatolog Treat. 2013;24:227-231.
- Alexiades-Armenakas MR, Dover JS, Arndt KA. The spectrum of laser skin resurfacing: nonablative, fractional, and ablative laser resurfacing. J Am Acad Dermatol. 2008;58:719-737.
- Tannous Z. Fractional resurfacing. Clin Dermatol. 2007;25:480-486.
- Gold MH. Continuing medical education article-skin treatment: photodynamic therapy: indications and treatment. Aesthet Surg J. 2008;28:545-552.
- Juarranz A, Jaén P, Sanz-Rodríguez F, et al. Photodynamic therapy of cancer. basic principles and applications. Clin Transl Oncol. 2008;10:148-154.
- Juzeniene A, Peng Q, Moan J. Milestones in the development of photodynamic therapy and fluorescence diagnosis. Photochem Photobiol Sci. 2007;6:1234-1245.
- Moan J, Berg K. The photodegradation of porphyrins in cells can be used to estimate the lifetime of singlet oxygen. Photochem Photobiol. 1991;53:549-553.
- Gupta AK, Paquet M, Villanueva E, et al. Interventions for actinic keratoses. Cochrane Database Syst Rev. 2012;12:CD004415.
- Patel G, Armstrong AW, Eisen DB. Efficacy of photodynamic therapy vs other interventions in randomized clinical trials for the treatment of actinic keratoses: a systematic review and meta-analysis. JAMA Dermatol. 2014;150:1281-1288.
- Kaufmann R, Spelman L, Weightman W, et al. Multicentre intraindividual randomized trial of topical methyl aminolaevulinate-photodynamic therapy vs. cryotherapy for multiple actinic keratoses on the extremities. Br J Dermatol. 2008;158:994-999.
- Freeman M, Vinciullo C, Francis D, et al. A comparison of photodynamic therapy using topical methyl aminolevulinate (Metvix) with single cycle cryotherapy in patients with actinic keratosis: a prospective, randomized study. J Dermatolog Treat. 2003;14:99-106.
- Morton C, Campbell S, Gupta G, et al. Intraindividual, right-left comparison of topical methyl aminolaevulinate-photodynamic therapy and cryotherapy in subjects with actinic keratoses: a multicentre, randomized controlled study. Br J Dermatol. 2006;155:1029-1036.
- Pariser DM, Lowe NJ, Stewart DM, et al. Photodynamic therapy with topical methyl aminolevulinate for actinic keratosis: results of a prospective randomized multicenter trial. J Am Acad Dermatol. 2003;48:227-232.
- Zane C, Facchinetti E, Rossi MT, et al. A randomized clinical trial of photodynamic therapy with methyl aminolaevulinate vs. diclofenac 3% plus hyaluronic acid gel for the treatment of multiple actinic keratoses of the face and scalp. Br J Dermatol. 2014;170:1143-1150.
- Perrett CM, McGregor JM, Warwick J, et al. Treatment of post-transplant premalignant skin disease: a randomized intrapatient comparative study of 5-fluorouracil cream and topical photodynamic therapy. Br J Dermatol. 2007;156:320-328.
- Szeimies RM, Karrer S, Radakovic-Fijan S, et al. Photodynamic therapy using topical methyl 5-aminolevulinate compared with cryotherapy for actinic keratosis: a prospective, randomized study. J Am Acad Dermatol. 2002; 47:258-262.
- Scola N, Terras S, Georgas D, et al. A randomized, half-side comparative study of aminolaevulinate photodynamic therapy vs. CO(2) laser ablation in immunocompetent patients with multiple actinic keratoses. Br J Dermatol. 2012;167:1366-1373.
- Willey A, Anderson RR, Sakamoto FH. Temperature-modulated photodynamic therapy for the treatment of actinic keratosis on the extremities: a pilot study. Dermatol Surg. 2014;40:1094-1102.
- Pariser DM. Management of Actinic Keratoses: Treatment Selection and Optimizing Outcomes. Presented at: Winter Clinical Dermatology Conference Hawaii; January 18, 2015; Kaanapali, HI.
- Dirschka T, Radny P, Dominicus R, et al. Long-term (6 and 12 months) follow-up of two prospective, randomized, controlled phase III trials of photodynamic therapy with BF-200 ALA and methyl aminolaevulinate for the treatment of actinic keratosis. Br J Dermatol. 2013;168:825-836.
- Choi SH, Kim KH, Song KH. Efficacy of ablative fractional laser-assisted photodynamic therapy with short-incubation time for the treatment of facial and scalp actinic keratosis: 12-month follow-up results of a randomized, prospective, comparative trial. J Eur Acad Dermatol Venereol. 2015;29:1598-1605.
- Ko DY, Jeon SY, Kim KH, et al. Fractional erbium:YAG laser-assisted photodynamic therapy for facial actinic keratoses: a randomized, comparative, prospective study. J Eur Acad Dermatol Venereol. 2014;28:1529-1539.
- Togsverd-Ho K, Haak CS, Thaysen-Petersen D, et al. Intensified photodynamic therapy of actinic keratoses with fractional CO2 laser: a randomized clinical trial. Br J Dermatol. 2012;166:1262-1269.
- Torezan L, Chaves Y, Niwa A, et al. A pilot split-face study comparing conventional methyl aminolevulinate-photodynamic therapy (PDT) with microneedling-assisted PDT on actinically damaged skin. Dermatol Surg. 2013;39:1197-1201.
- Lebwohl M. Actinic keratosis: epidemiology and progression to squamous cell carcinoma. Br J Dermatol. 2003;149(suppl 66):31-33.
- Drake LA, Ceilley RI, Cornelison RL, et al. Guidelines of care for actinic keratoses. Committee on Guidelines of Care. J Am Acad Dermatol. 1995;32:95-98.
- Salasche SJ. Epidemiology of actinic keratoses and squamous cell carcinoma. J Am Acad Dermatol. 2000;42(1, pt 2):4-7.
- Ackerman AB, Mones JM. Solar (actinic) keratosis is squamous cell carcinoma. Br J Dermatol. 2006;155:9-22.
- Anwar J, Wrone DA, Kimyai-Asadi A, et al. The development of actinic keratosis into invasive squamous cell carcinoma: evidence and evolving classification schemes. Clin Dermatol. 2004;22:189-196.
- Criscione VD, Weinstock MA, Naylor MF, et al. Actinic keratoses: natural history and risk of malignant transformation in the Veterans Affairs Topical Tretinoin Chemoprevention Trial. Cancer. 2009;115:2523-2530.
- Glogau RG. The risk of progression to invasive disease. J Am Acad Dermatol. 2000;42(1, pt 2):23-24.
- Esmann S, Jemec GB. Management of actinic keratosis patients: a qualitative study. J Dermatolog Treat. 2007;18:53-58.
- Weinstock MA, Lee KC, Chren MM, et al. Quality of life in the actinic neoplasia syndrome: the VA Topical Tretinoin Chemoprevention (VATTC) trial. J Am Acad Dermatol. 2009;61:207-215.
- Berman B, Villa AM, Ramirez CC. Mechanisms of action of new treatment modalities for actinic keratosis. J Drugs Dermatol. 2006;5:167-173.
- Askew DA, Mickan SM, Soyer HP. Effectiveness of 5-fluorouracil treatment for actinic keratosis: a systematic review of randomized controlled trials. Int J Dermatol. 2009;46:452-463.
- Levy S, Furst K, Chern W. A pharmacokinetic evaluation of 0.5% and 5% fluorouracil topical cream in patients with actinic keratosis. Clin Ther. 2001;23:908-920.
- Jorizzo J, Stewart D, Bucko A, et al. Randomized trial evaluating a new 0.5% fluorouracil formulation demonstrates efficacy after 1-, 2-, or 4-week treatment in patients with actinic keratosis. Cutis. 2002;70:335-359.
- Sachs DL, Kang S, Hammerberg C, et al. Topical fluorouracil for actinic keratoses and photoaging: a clinical and molecular analysis. Arch Dermatol. 2009;145:659-666.
- Hantash BM, Stewart DB, Cooper ZA, et al. Facial resurfacing for nonmelanoma skin cancer prophylaxis. Arch Dermatol. 2006;142:976-982.
- Lawrence N, Cox SE, Cockerell CJ, et al. A comparison of the efficacy and safety of Jessner’s solution and 35% trichloroacetic acid vs 5% fluorouracil in the treatment of widespread facial actinic keratoses. Arch Dermatol. 1995;131:176-181.
- Monheit GD. The Jessner’s + TCA peel: a medium-depth chemical peel. J Dermatol Surg Oncol. 1989;15:945-950.
- Hemmi H, Kaisho T, Takeuchi O, et al. Small anti-viral compounds activate immune cells via the TLR7 MyD88-dependent signaling pathway. Nat Immunol. 2002;3:196-200.
- Adamson DJ, Frew D, Tatoud R, et al. Diclofenac antagonizes peroxisome proliferator-activated receptor-gamma signaling. Mol Pharmacol. 2002;61:7-12.
- Alam CA, Seed MP, Willoughby DA. Angiostasis and vascular regression in chronic granulomatous inflammation induced by diclofenac in combination with hyaluronan in mice. J Pharm Pharmacol. 1995;47:407-411.
- Lu X, Xie W, Reed D, et al. Nonsteroidal antiinflammatory drugs cause apoptosis and induce cyclooxygenases in chicken embryo fibroblasts. Proc Natl Acad Sci USA. 1995;92:7961-7965.
- Seed MP, Brown JR, Freemantle CN, et al. The inhibition of colon-26 adenocarcinoma development and angiogenesis by topical diclofenac in 2.5% hyaluronan. Cancer Res. 1997;57:1625-1629.
- Rivers JK, Arlette J, Shear N, et al. Topical treatment of actinic keratoses with 3.0% diclofenac in 2.5% hyaluronan gel. Br J Dermatol. 2002;146:94-100.
- Wolf JE, Taylor JR, Tschen E, et al. Topical 3.0% diclo-fenac in 2.5% hyaluronan gel in the treatment of actinic keratoses. Int J Dermatol. 2001;40:709-713.
- Nelson C, Rigel D, Smith S, et al. Phase IV, open-label assessment of the treatment of actinic keratosis with 3.0% diclofenac sodium topical gel (Solaraze). J Drugs Dermatol. 2004;3:401-407.
- Pflugfelder A, Welter AK, Leiter U, et al. Open label randomized study comparing 3 months vs. 6 months treatment of actinic keratoses with 3% diclofenac in 2.5% hyaluronic acid gel: a trial of the German Dermatologic Cooperative Oncology Group. J Eur Acad Dermatol Venereol. 2012;26:48-53.
- Smith SR, Morhenn VB, Piacquadio DJ. Bilateral comparison of the efficacy and tolerability of 3% diclofenac sodium gel and 5% 5-fluorouracil cream in the treatment of actinic keratoses of the face and scalp. J Drugs Dermatol. 2006;5:156-159.
- Segatto MM, Dornelles SI, Silveira VB, et al. Comparative study of actinic keratosis treatment with 3% diclo- fenac sodium and 5% 5-fluorouracil. An Bras Dermatol. 2013;88:732-738.
- Vidal D. Topical imiquimod: mechanism of action and clinical applications. Mini Rev Med Chem. 2006;6:499-503.
- Hadley G, Derry S, Moore RA. Imiquimod for actinic keratosis: systematic review and meta-analysis. J Invest Dermatol. 2006;126:1251-1255.
- Caperton C, Berman B. Safety, efficacy, and patient acceptability of imiquimod for topical treatment of actinic keratoses. Clin Cosmet Investig Dermatol. 2011;4:35-40.
- Swanson N, Smith CC, Kaur M, et al. Imiquimod 2.5% and 3.75% for the treatment of actinic keratoses: two phase 3, multicenter, randomized, double-blind, placebo-controlled studies. J Drugs Dermatol. 2014;13:166-169.
- Krawtchenko N, Roewert-Huber J, Ulrich M, et al. A randomised study of topical 5% imiquimod vs. topical 5-fluorouracil vs. cryosurgery in immunocompetent patients with actinic keratoses: a comparison of clinical and histological outcomes including 1-year follow-up. Br J Dermatol. 2007;157(suppl 2):34-40.
- Anderson L, Schmieder GJ, Werschler WP, et al. Randomized, double-blind, double-dummy, vehicle-controlled study of ingenol mebutate gel 0.025% and 0.05% for actinic keratosis. J Am Acad Dermatol. 2009;60:934-943.
- Ogbourne SM, Suhrbier A, Jones B, et al. Antitumor activity of 3-ingenyl angelate: plasma membrane and mitochondrial disruption and necrotic cell death. Cancer Res. 2004;64:2833-2839.
- Lebwohl M, Swanson N, Anderson LL, et al. Ingenol mebutate gel for actinic keratosis. N Engl J Med. 2012;366:1010-1019.
- Lebwohl M, Shumack S, Stein-Gold L, et al. Long-term follow-up study of ingenol mebutate gel for the treatment of actinic keratoses. JAMA Dermatol. 2013;149:666-670.
- Augustin M, Tu JH, Knudsen KM, et al. Ingenol mebutate gel for actinic keratosis: the link between quality of life, treatment satisfaction, and clinical outcomes. J Am Acad Dermatol. 2015;72:816-821.
- Kane-Maguire N, Moseley R, Cozzi S, et al. Modulation of fibroblast phenotype and extracellular matrix composition by ingenol mebutate may be associated with scar resolution and improved dermal cosmesis. J Am Acad Dermatol. 2012;66:AB218.
- Martin G, Swanson N. Clinical findings using ingenol mebutate gel to treat actinic keratoses. J Am Acad Dermatol. 2013;68(1, suppl 1):S39-S48.
- Feldman SR, Fleischer AB, Williford PM, et al. Destructive procedures are the standard of care for treatment of actinic keratoses. J Am Acad Dermatol. 1999;40:43-47.
- Berlin JM. Current and emerging treatment strategies for the treatment of actinic keratosis. Clin Cosmet Investig Dermatol. 2010;3:119-126.
- Coleman WP, Yarborough JM, Mandy SH. Dermabrasion for prophylaxis and treatment of actinic keratoses. Dermatol Surg. 1996;22:17-21.
- Cooley JE, Casey DL, Kauffman CL. Manual resurfacing and trichloroacetic acid for the treatment of patients with widespread actinic damage. clinical and histologic observations. Dermatol Surg. 1997;23:373-379.
- Goldberg LH, Kaplan B, Vergilis-Kalner I, et al. Liquid nitrogen: temperature control in the treatment of actinic keratosis. Dermatol Surg. 2010;36:1956-1961.
- Lubritz RR, Smolewski SA. Cryosurgery cure rate of actinic keratoses. J Am Acad Dermatol. 1982;7:631-632.
- Thai KE, Fergin P, Freeman M, et al. A prospective study of the use of cryosurgery for the treatment of actinic keratoses. Int J Dermatol. 2004;43:687-692.
- Zane C, Facchinetti E, Rossi MT, et al. Cryotherapy is preferable to ablative CO2 laser for the treatment of isolated actinic keratoses of the face and scalp: a randomized clinical trial. Br J Dermatol. 2014;170:1114-1121.
- Trimas SJ, Ellis DA, Metz RD. The carbon dioxide laser. an alternative for the treatment of actinically damaged skin. Dermatol Surg. 1997;23:885-889.
- Jiang SB, Levine VJ, Nehal KS, et al. Er:YAG laser for the treatment of actinic keratoses. Dermatol Surg. 2000;26:437-440.
- Ostertag JU, Quaedvlieg PJ, Van der geer S, et al. A clinical comparison and long-term follow-up of topical 5-fluorouracil versus laser resurfacing in the treatment of widespread actinic keratoses. Lasers Surg Med. 2006;38:731-739.
- Iyer S, Friedli A, Bowes L, et al. Full face laser resurfacing: therapy and prophylaxis for actinic keratoses and non-melanoma skin cancer. Lasers Surg Med. 2004;34:114-119.
- Rubin MG. A peeler’s thoughts on skin improvement with chemical peels and laser resurfacing. Clin Plast Surg. 1997;24:407-409.
- Riggs K, Keller M, Humphreys TR. Ablative laser resurfacing: high-energy pulsed carbon dioxide and erbium:yttrium-aluminum-garnet. Clin Dermatol. 2007;25:462-473.
- Adrian RM. Pulsed carbon dioxide and long pulse 10-ms erbium-YAG laser resurfacing: a comparative clinical and histological study. J Cutan Laser Ther. 1999;1:197-202.
- Weiss ET, Brauer JA, Anolik R, et al. 1927-nm fractional resurfacing of facial actinic keratoses: a promising new therapeutic option. J Am Acad Dermatol. 2013; 68:98-102.
- Manstein D, Herron GS, Sink RK, et al. Fractional photothermolysis: a new concept for cutaneous remodeling using microscopic patterns of thermal injury. Lasers Surg Med. 2004;34:426-438.
- Katz TM, Goldberg LH, Marquez D, et al. Nonablative fractional photothermolysis for facial actinic keratoses: 6-month follow-up with histologic evaluation. J Am Acad Dermatol. 2011;65:349-356.
- Prens SP, De Vries K, Neumann HA, et al. Non-ablative fractional resurfacing in combination with topical tretinoin cream as a field treatment modality for multiple actinic keratosis: a pilot study and a review of other field treatment modalities. J Dermatolog Treat. 2013;24:227-231.
- Alexiades-Armenakas MR, Dover JS, Arndt KA. The spectrum of laser skin resurfacing: nonablative, fractional, and ablative laser resurfacing. J Am Acad Dermatol. 2008;58:719-737.
- Tannous Z. Fractional resurfacing. Clin Dermatol. 2007;25:480-486.
- Gold MH. Continuing medical education article-skin treatment: photodynamic therapy: indications and treatment. Aesthet Surg J. 2008;28:545-552.
- Juarranz A, Jaén P, Sanz-Rodríguez F, et al. Photodynamic therapy of cancer. basic principles and applications. Clin Transl Oncol. 2008;10:148-154.
- Juzeniene A, Peng Q, Moan J. Milestones in the development of photodynamic therapy and fluorescence diagnosis. Photochem Photobiol Sci. 2007;6:1234-1245.
- Moan J, Berg K. The photodegradation of porphyrins in cells can be used to estimate the lifetime of singlet oxygen. Photochem Photobiol. 1991;53:549-553.
- Gupta AK, Paquet M, Villanueva E, et al. Interventions for actinic keratoses. Cochrane Database Syst Rev. 2012;12:CD004415.
- Patel G, Armstrong AW, Eisen DB. Efficacy of photodynamic therapy vs other interventions in randomized clinical trials for the treatment of actinic keratoses: a systematic review and meta-analysis. JAMA Dermatol. 2014;150:1281-1288.
- Kaufmann R, Spelman L, Weightman W, et al. Multicentre intraindividual randomized trial of topical methyl aminolaevulinate-photodynamic therapy vs. cryotherapy for multiple actinic keratoses on the extremities. Br J Dermatol. 2008;158:994-999.
- Freeman M, Vinciullo C, Francis D, et al. A comparison of photodynamic therapy using topical methyl aminolevulinate (Metvix) with single cycle cryotherapy in patients with actinic keratosis: a prospective, randomized study. J Dermatolog Treat. 2003;14:99-106.
- Morton C, Campbell S, Gupta G, et al. Intraindividual, right-left comparison of topical methyl aminolaevulinate-photodynamic therapy and cryotherapy in subjects with actinic keratoses: a multicentre, randomized controlled study. Br J Dermatol. 2006;155:1029-1036.
- Pariser DM, Lowe NJ, Stewart DM, et al. Photodynamic therapy with topical methyl aminolevulinate for actinic keratosis: results of a prospective randomized multicenter trial. J Am Acad Dermatol. 2003;48:227-232.
- Zane C, Facchinetti E, Rossi MT, et al. A randomized clinical trial of photodynamic therapy with methyl aminolaevulinate vs. diclofenac 3% plus hyaluronic acid gel for the treatment of multiple actinic keratoses of the face and scalp. Br J Dermatol. 2014;170:1143-1150.
- Perrett CM, McGregor JM, Warwick J, et al. Treatment of post-transplant premalignant skin disease: a randomized intrapatient comparative study of 5-fluorouracil cream and topical photodynamic therapy. Br J Dermatol. 2007;156:320-328.
- Szeimies RM, Karrer S, Radakovic-Fijan S, et al. Photodynamic therapy using topical methyl 5-aminolevulinate compared with cryotherapy for actinic keratosis: a prospective, randomized study. J Am Acad Dermatol. 2002; 47:258-262.
- Scola N, Terras S, Georgas D, et al. A randomized, half-side comparative study of aminolaevulinate photodynamic therapy vs. CO(2) laser ablation in immunocompetent patients with multiple actinic keratoses. Br J Dermatol. 2012;167:1366-1373.
- Willey A, Anderson RR, Sakamoto FH. Temperature-modulated photodynamic therapy for the treatment of actinic keratosis on the extremities: a pilot study. Dermatol Surg. 2014;40:1094-1102.
- Pariser DM. Management of Actinic Keratoses: Treatment Selection and Optimizing Outcomes. Presented at: Winter Clinical Dermatology Conference Hawaii; January 18, 2015; Kaanapali, HI.
- Dirschka T, Radny P, Dominicus R, et al. Long-term (6 and 12 months) follow-up of two prospective, randomized, controlled phase III trials of photodynamic therapy with BF-200 ALA and methyl aminolaevulinate for the treatment of actinic keratosis. Br J Dermatol. 2013;168:825-836.
- Choi SH, Kim KH, Song KH. Efficacy of ablative fractional laser-assisted photodynamic therapy with short-incubation time for the treatment of facial and scalp actinic keratosis: 12-month follow-up results of a randomized, prospective, comparative trial. J Eur Acad Dermatol Venereol. 2015;29:1598-1605.
- Ko DY, Jeon SY, Kim KH, et al. Fractional erbium:YAG laser-assisted photodynamic therapy for facial actinic keratoses: a randomized, comparative, prospective study. J Eur Acad Dermatol Venereol. 2014;28:1529-1539.
- Togsverd-Ho K, Haak CS, Thaysen-Petersen D, et al. Intensified photodynamic therapy of actinic keratoses with fractional CO2 laser: a randomized clinical trial. Br J Dermatol. 2012;166:1262-1269.
- Torezan L, Chaves Y, Niwa A, et al. A pilot split-face study comparing conventional methyl aminolevulinate-photodynamic therapy (PDT) with microneedling-assisted PDT on actinically damaged skin. Dermatol Surg. 2013;39:1197-1201.
Practice Points
- In addition to their risk for progression to malignancy, actinic keratoses (AKs) can have negative impacts on cosmetic appearance and quality of life.
- A variety of topical medications, procedural modalities, and light-based therapies are available for treatment of AKs, which offer varying degrees of efficacy for clearance of lesions and cosmetic outcomes. Based on the current data, imiquimod and photodynamic therapy are the treatments most likely to provide an excellent cosmetic outcome.