User login
Determining Efficacy of Antibacterial Hand Wash Products
Determining Efficacy of Antibacterial Hand Wash Products
This promotional supplement was supported by American Cleaning Institute® and the Personal Care Products Council.
•Topics
•Faculty/Faculty Disclosures
To view the supplement, click the image above.
- 1994 FDA-Proposed Antiseptic Hand Wash Efficacy Criteria
- Need for Link Between Bacterial Log Reduction and Disease
- New Model for Efficacy Evaluation of Antibacterial Hand Washes
- New Scientific Model Links Bacterial Reduction with Reduction in Disease
- Expert Panel Review of the New Model for Efficacy Evaluation
- Expert Panel Consensus Statement
- Expanding the Scope of the New Scientific Model to Other Health Care Settings
- Moving Forward
Francis H. Kruszewski, PhD, DABT
Director, Human Health and Safety
American Cleaning Institute® (formerly the Soap and Detergent Association)
Washington, DC
Francis H. Kruszewski, PhD, DABT is employed by American Cleaning Institute® (formerly the Soap and Detergent Association)
John F. Krowka, PhD
Senior Environmental Scientist
The Personal Care Products Council
Washington, DC
John F. Krowka, PhD is employed by The Personal Care Products Council
Copyright © 2011 Elsevier Inc.
Determining Efficacy of Antibacterial Hand Wash Products
This promotional supplement was supported by American Cleaning Institute® and the Personal Care Products Council.
•Topics
•Faculty/Faculty Disclosures
To view the supplement, click the image above.
- 1994 FDA-Proposed Antiseptic Hand Wash Efficacy Criteria
- Need for Link Between Bacterial Log Reduction and Disease
- New Model for Efficacy Evaluation of Antibacterial Hand Washes
- New Scientific Model Links Bacterial Reduction with Reduction in Disease
- Expert Panel Review of the New Model for Efficacy Evaluation
- Expert Panel Consensus Statement
- Expanding the Scope of the New Scientific Model to Other Health Care Settings
- Moving Forward
Francis H. Kruszewski, PhD, DABT
Director, Human Health and Safety
American Cleaning Institute® (formerly the Soap and Detergent Association)
Washington, DC
Francis H. Kruszewski, PhD, DABT is employed by American Cleaning Institute® (formerly the Soap and Detergent Association)
John F. Krowka, PhD
Senior Environmental Scientist
The Personal Care Products Council
Washington, DC
John F. Krowka, PhD is employed by The Personal Care Products Council
Copyright © 2011 Elsevier Inc.
Determining Efficacy of Antibacterial Hand Wash Products
This promotional supplement was supported by American Cleaning Institute® and the Personal Care Products Council.
•Topics
•Faculty/Faculty Disclosures
To view the supplement, click the image above.
- 1994 FDA-Proposed Antiseptic Hand Wash Efficacy Criteria
- Need for Link Between Bacterial Log Reduction and Disease
- New Model for Efficacy Evaluation of Antibacterial Hand Washes
- New Scientific Model Links Bacterial Reduction with Reduction in Disease
- Expert Panel Review of the New Model for Efficacy Evaluation
- Expert Panel Consensus Statement
- Expanding the Scope of the New Scientific Model to Other Health Care Settings
- Moving Forward
Francis H. Kruszewski, PhD, DABT
Director, Human Health and Safety
American Cleaning Institute® (formerly the Soap and Detergent Association)
Washington, DC
Francis H. Kruszewski, PhD, DABT is employed by American Cleaning Institute® (formerly the Soap and Detergent Association)
John F. Krowka, PhD
Senior Environmental Scientist
The Personal Care Products Council
Washington, DC
John F. Krowka, PhD is employed by The Personal Care Products Council
Copyright © 2011 Elsevier Inc.
Survey of Academic Hospitalist Leaders
Hospitalists are hospital‐based physicians whose primary professional focus is patient care, education, research, and administrative activities related to hospital medicine.1 Initially, community‐based hospitals were far more likely to employ hospitalists than academic centers. However, today most academic centers employ hospitalist models and it is now a fully recognized entity in academic settings.2
While much has been written about the structure, business operations, and potential benefits of nonteaching (clinical) hospitalist programs,3, 4 there is little known about the current state of academic hospitalist programs or their challenges. For example, who are the leaders of academic hospitalist medicine groups? Given the youth of the field, are academic hospitalists receiving adequate mentorship and are they advancing academically? What are future directions and goals for academic hospitalist groups?
To better understand academic hospitalist programs, we surveyed division chiefs and academic hospitalist leaders to explore existing business models and operations, the status of mentorship, and key issues in growth and retention.
Methods
Sites and Subjects
We targeted potential hospital medicine group leaders by identifying academic medical centers using Association of American Medical Colleges (AAMC), the Accreditation Council for Graduate Medical Education (ACGME), the Association of Chiefs of General Internal Medicine (ACGIM), and the Society of Hospital Medicine (SHM) lists of sites with teaching missions. We then used publicly available data (eg, from websites maintained by the sites) to identify physician leaders who: 1) self identified as a leader of a hospitalist group at an academic medical center (or a Chief of Division of General Internal Medicine which managed a hospitalist group) in the SHM database, 2) were listed as such on the website, or 3) were members of ACGIM and listed as a hospitalist group leader at a university based medical center.
Survey Development
Our survey was based on questions used in previous research by the authors,5 with additional questions regarding operations of academic hospitalist programs, growth and retention of hospitalists, and mentorship developed by the study authors. Questions were pretested among a selected group of members of the Society of General Internal Medicine (SGIM) Academic Hospitalist Task Force and the SHM Academic Hospitalist Interest Group, after which the survey was refined and converted into its electronic form.
Survey Methods
The email survey process began in April 2007 with an initial survey sent to those physicians identified using preexisting data, as described. Our survey asked first if recipients were directly responsible for the oversight of a hospitalist group (eg, the division chief or director of the hospital medicine group) and if they practiced at an academic medical center. Only respondents who answered yes to both of these criteria were invited to respond to our survey. Those who felt the survey did not apply to them were invited to forward the email survey on to the appropriate person at their site or respond that their hospital had no hospital medicine service. Subsequent reminder emails were sent to nonrespondents at 10‐day intervals up to a total of four times. This survey was granted exempt status from the UCSF Institutional Review Board.
Statistical Methods
Response rates and frequencies and distribution of survey responses were analyzed using univariable statistics.
Results
Characteristics of Responding Sites
We received responses from 57 (40%) of the academic sites identified as having an academic hospital medicine group. Hospitalist group leaders at responding sites had been in their current position 3.8 years, graduated medical school approximately 15 years prior, and were either Assistant (40%), Associate (32%), or Full Professors (23%). Group leaders reported that the vast majority (91%) of group full‐time members were in junior faculty positions (Instructor or Assistant Professor), who were working full‐time. On average, responding programs were 6 years old (formed in 2001) and currently had 10.0 total full time equivalents (FTEs). A total of 38 of the groups (67%) were part of the larger Division of General Internal Medicine, whereas 9 groups (16 %) were their own division within the Department of Medicine. The remaining 17% were part of another division.
Mentorship Practices In Academic Hospital Medicine Groups
As one mechanism of mentorship, annual performance reviews were offered in most programs (88%). These were usually performed by the general medicine division chief or hospitalist leader. Mentoring relationships for clinician investigators (CI) were most often from personnel outside the hospitalist group, whereas clinician‐educators (CE) most often were mentored by faculty inside the group.
Hospitalist Leaders' Priorities and Impressions of Growth, Opportunities, Career Development and Barriers
Hospitalist leaders reported the highest priorities for hospitalist leaders were developing research and teaching programs, and minimizing turnover. Other priorities included achieving financial stability, applying for extramural funding, and reducing clinical workload (Table 2). Only 14% of respondents noted that becoming a separate division was a priority.0
| Characteristic | n (%) |
|---|---|
| |
| Group leader characteristics | |
| Academic rank | |
| Assistant professor/other | 26 (45) |
| Associate professor | 18 (32) |
| Full professor | 13 (23) |
| Years in position (mean, range) | 3.8 (2.07.0) |
| Group characteristics | |
| Hospital medicine place in school of medicine | |
| Within the department of medicine | 55 (98) |
| Separate division | 9 (16) |
| Within division of general medicine | 38 (67) |
| Other | 9 (16) |
| Not in the department of medicine | 1 (2) |
| Program size (mean, range) | |
| Number of hospitalists in program | 10 (718) |
| Number of FTE | 11 (3.512) |
| FTE's hired in past 2 years (July 2005 to survey date) | 4.0 (2.27.0) |
| Hospitalist activities | |
| Medicine consultation | 52 (91) |
| Quality improvement projects | 52 (91) |
| Nonteaching attending | 44 (77) |
| Comanagement of surgical patients | 44 (77) |
| 24‐hour coverage | 24 (61) |
| Manage patient transfer requests | 32 (56) |
| Peer review/morbidity and mortality | 31 (54) |
| Education program leadership | 29 (51) |
| Medical student program leadership | 29 (51) |
| Palliative care program | 23 (40) |
| Preoperative clinic | 23 (40) |
| Emergency department triage | 14 (25) |
| Post discharge follow‐up clinic | 13 (23) |
| Skill nursing facility coverage | 4 (7) |
| Other | 15 (26) |
| Mentorship Activity | n (%) |
|---|---|
| Programs performing annual reviews with faculty | 50 (88) |
| Who performs the annual review? | |
| General Medicine Division Chief | 9 (18) |
| Hospitalist leader | 18 (36) |
| Both | 13 (26) |
| Other (eg, Department Chair, Chief Medical Officer) | 10 (20) |
| Who is the primary source of mentorship for clinician‐educators? | |
| Senior faculty within the group | 43 (77) |
| Generalist faculty outside the group, but within the institution | 6 (11) |
| Subspecialty Internal Medicine faculty outside the group, but within the institution | 3 (5) |
| Non‐Internal Medicine (eg, surgeon, epidemiologist) outside the group, but within the institution | 0 (0) |
| Faculty from another institution | 0 (0) |
| Don't know | 4 (7) |
| Who is the primary source of mentorship for clinician‐investigators? | |
| Senior faculty within the group | 6 (12) |
| Generalist faculty outside the group, but within the institution | 13 (25) |
| Subspecialty internal medicine faculty outside the group, but within the institution | 6 (12) |
| Non‐Internal Medicine (eg, surgeon, epidemiologist) outside the group, but within the institution | 2 (4) |
| Faculty from another institution | 3 (6) |
| Don't know | 2 (4) |
| Not applicable; no clinician investigators | 20 (38) |
| Highest Priority, n (%) | Intermediate Priority, n (%) | Lowest Priority, n (%) | Not a Priority, n (%) | NA, n (%) | |
|---|---|---|---|---|---|
| Reducing individual faculty clinical workload | 9 (16) | 22 (3) | 11 (2) | 14 (2) | 0 (0) |
| Achieving financial stability | 13 (24) | 30 (55) | 6 (11) | 6 (11) | 0 (0) |
| Minimizing turnover | 22 (39) | 27 (48) | 6 (11) | 1 (2) | 0 (0) |
| Developing teaching programs | 22 (39) | 29 (52) | 3 (5) | 2 (4) | 0 (0) |
| Becoming a separate division | 3 (5) | 5 (9) | 11 (20) | 23 (41) | 14 (25) |
| Developing research | 25 (45) | 18 (32) | 5 (9) | 6 (11) | 2 (4) |
| Applying for extramural funding | 10 (18) | 24 (43) | 10 (18) | 8 (14) | 4 (7) |
| Strongly Agree, n (%) | Agree, n (%) | Neutral, n (%) | Disagree, n (%) | Strongly Disagree, n (%) | NA, n (%) | |
|---|---|---|---|---|---|---|
| Growth and sustainability | ||||||
| Availability of funds is limiting expansion of academic functions (eg, education and research) | 20 (36) | 21 (38) | 5 (9) | 7 (12) | 3 (5) | 0 (0) |
| Availability of funds is limiting expansion of clinical functions (eg, development of new services) | 11 (20) | 17 (30) | 14 (25) | 10 (18) | 4 (7) | 0 (0) |
| My faculty are developing sustainable nonclinical activities | 9 (16) | 23 (41) | 12 (21) | 9 (16) | 3 (5) | 0 (0) |
| Career development | ||||||
| Mentorship is a major issue for my clinician‐educator faculty | 14 (25) | 28 (50) | 7 (12) | 4 (7) | 1 (2) | 2 (4) |
| Mentorship is a major issue for my research faculty | 22 (40) | 10 (18) | 4 (7) | 3 (5) | 2 (4) | 14 (25) |
| External support for hospital medicine group | ||||||
| There is investment in the development of academic functions of our hospitalist program from my hospital | 4 (7) | 12 (21) | 10 (18) | 22 (39) | 8 (14) | 0 (0) |
| There is investment in the development of academic functions of our hospitalist program from the Department of Medicine | 22 (40) | 17 (31) | 8 (15) | 4 (7) | 2 (4) | 2 (4) |
In general, academic hospitalist leaders reported that Departments of Medicine and Divisions of General Medicine (where applicable) were invested in the development of their academic functions. Yet, more than half of program directors reported that hospitals were not supportive. Moreover, lack of funds limited the expansion of their academic or clinical functions (Table 3). Additionally, while the majority either strongly agree or agree that their faculty are developing sustainable nonclinical activities (57%), they perceive that they are at risk for burnout (69%), and that lack of mentorship is a major issue for both CE (75%) and research faculty (58%). Lastly, while program directors strongly agree or agree (71%) that their hospitalist groups are respected by other academic physicians, they additionally strongly agree or agree that their Departments of Medicine (58%) and other Divisions (78%) view their hospitalist program as a clinical service rather than an academic program.
Discussion
Our survey provides a unique snapshot of academic hospitalist groups, highlighting a perceived lack of support and respect for their programs, a need to increase education and scholarly activities, and a desire to better prepare faculty for academic promotion.
Academic hospitalist groups and leaders reflected what one would expect from a field that is just over a decade old. Program leaders were relatively new to their position, as were their division group members. As a result, it is not surprising that most of the academic hospitalist leaders identified mentorship as a major issue. We were encouraged to see that most programs were offering annual reviews. However, the majority of these annual reviews were performed by the group leaders, many of whom are relatively junior (40% Assistant Professors) and may not be experienced in mentoring and performing annual reviews. Importantly, the absence of a mentor (or a high‐quality, experienced one) among physicians, and specifically hospitalists, may result in fewer peer‐reviewed first author and non‐peer‐reviewed publications, and less experience leading a teaching session at a national meeting.6 Research suggests that effective mentoring may help faculty increase career satisfaction and productivity and reduce their risk for burn‐out.7 Hospitalist groups might benefit nationally from focusing specifically on finding adequate mentorship either within or outside their groups. In addition, national organizations such as the SHM and the SGIM could potentially help these groups and individual hospitalists in creating mentorship networks and a mentoring infrastructure.
Academic hospitalist leaders were concerned about the ability of their faculty to develop sustainable nonclinical activities and scholarship. Notably, more than 40% of surveyed leaders agreed or strongly agreed that their faculty were not developing sustainable nonclinical activities. For individual faculty, the inability to develop scholarly activities and engage in academic pursuits may create challenges in getting promoted by traditional academic pathways. Some have recognized this issue and tried to develop practical solutions.2 In addition, academic hospitalists often engage in nonclinical activities such as quality improvement or patient safety which do not fit in the traditional tripartite mission of academics (clinical care, education, and research). In this survey, more than 90% of groups were engaged in quality improvement projects and over half in peer review exercises (Table 1). As many of these scholarly activities require innovation, sophisticated data analysis, and can have far‐reaching and substantial impacts on healthcare, some have argued these should be considered as part of the promotion process.8 Notably, the SGIM Academic Hospitalist Taskforce has created the Quality Portfolio, a structured adjunct to promotions packets to organize and document work in quality improvement and patient safety.9
While there were few CI in the divisions surveyed, building CI programs was a major priority of programs. In programs reporting the presence of CI's, they report limited access to research support. This highlights the potential role and benefit of post residency training in designing and conducting clinical research whether in a traditional general internal medicine fellowship or in 1 of the many growing hospital medicine fellowships.10 There also appears to be a need for funding to support the research careers of junior hospitalists. While access to effective mentorship is integrally linked to achieving increased academic accomplishments, there is certainly an ample call for research in the areas of quality improvement, patient safety; systems‐based practice, hospital efficiency, transitions of care,11 perioperative medicine,12 and education.2, 13, 14 While providing lower costs per admission and lower lengths of stay, hospitalists seem well‐positioned to spearhead active research in cost‐effectiveness in the hospital.14 Additionally, a quality portfolio, documenting such quality improvement projects, has been suggested as an effective means to provide a record of this work for academic promotion.9
The diverse activities of today's hospitalists are transforming the traditional view of academic work and are critical to the growth of hospitals, patient care, and development of the field of hospital medicine itself. Until these areas are fully embraced as legitimate areas of academic productivity and scholarship, the academic advancement of hospitalists will be slow.
It is unclear from our survey if academic hospitalist programs are truly getting the support they need to succeed. On one hand, there was general agreement that the Departments of Medicine and Divisions of General and Hospital Medicine were invested in the development of the academic accomplishments. Yet, the majority of program directors believed that they are viewed by the Department or Division as a clinical rather than an academic program. Moreover, over half of program directors report that their hospital was not supportive and therefore have limited the expansion of their hospitalist groups' educational and research activities. Lastly, for a large majority of programs, unavailable funding also acted to limit growth and expansion of academic functions. In a mere 2 decades, Emergency Medicine has become one of the largest US specialties and yet research and funding in the field have been lagging and are limiting academic expansion. Junior faculty seeking research careers struggled to find support and mentorship within their emergency medicine divisions.15 Challenges faced by academic emergency medicine provide important historical perspective for the even more rapidly growing field of academic hospital medicine. Learning from the Academic Emergency Medicine experience, academic hospitalists should proactively identify scholarship and research opportunities unique to hospitalist and fitting the needs of academic institutions. Involvement in national medical organizations, such as SGIM‐SHM‐ACGIM Academic Hospitalist Academy, or the SGIM Academic Hospitalist Task Force, where skill and career development is the focus, will undoubtedly promote the success of academic hospitalist. Expanding valuable niches of expertise, such as quality control, perioperative medicine and care transitions, create an indispensible component of hospital care. Lastly mentoring programs for academic hospitalist within SHM and SGIM are also essential for networking and career development. There are several limitations to our study. Our response rate of 40% was relatively low, and our results may not be representative of all academic hospitalist division chiefs and their programs, may be overstating the perceived difficulties of the survey sample, or conversely missing a large portion too overwhelmed by current duties who lacked the time to complete the survey. Having said this, our survey methodology targeted sites where we could identify potentialnot confirmedhospitalist groups and hospitalist group leaders. For this reason, our response rate could be higher (if some of our contacts were in error). Our results are a cross‐sectional survey based on self report and are subject to recall bias. In addition, our study was carried out in 2007, and while issues such as mentorship may remain important, our results regarding financial arrangements may not be applicable to the current economic climate. Finally, while improving mentorship was identified as a principle objective for program leaders, we did not explore the existing quality of mentorship, nor perceived shortfalls. This should be the subject of future exploration.
The vast majority of academic hospital medicine programs continue to view inadequate support, expanding research, mentorship, and academic promotion as critical issues for the future. Thus, further understanding of these features, and interventions to allow for success, are of crucial importance in the continued development of academic hospitalists. Our study supports the need for mentoring and career development programs, targeting academic hospitalists and their leaders. In addition, attention should be paid to activities that support career fit, creating sustainable and viable job descriptions for academic hospitalists, and preventing burnout.16 At the same time we must expand the traditional view of scholarship and training and advocate for promotion criteria that value the unique contributions of hospitalists to become in line with the broad areas that hospitalists work.
- Anonymous. Definition of a hospitalist.2009. Society of Hospital Medicine Homepage/General Information. Available at: http://www.hospitalmedicine.org/AM/Template.cfm?Section=Hospitalist_Definition4:240–246.
- ,,,,.Resource‐based relative value scale analysis between teaching and nonteaching hospitalist services.Health Care Management.2009;1(28):81–85.
- ,,, et al.Effects of physician experience on costs and outcomes on an academic general medicine service.Ann Intern Med.2002;137:866–874.
- ,,,.Hospitalists and the practice of inpatient medicine.Ann Intern Med.1999;130:343–349.
- ,,,,,.Rates, predictors and consequences of low career satisfaction and burnout in academic hospital medicine.J Hosp Med.2009;4(S1):24–25.
- ,.Mentoring faculty in academic medicine: a new paradigm?.J Gen Intern Med.2005;20(9):866–870.
- ,.Clinicians in quality improvement: a new career pathway in academic medicine.JAMA.2009;301(7):766–768.
- ,,,.Academic hospitalist taskforce quality portfolio rationale and development. 02/23/2009; Quality portfolio introduction. Available at:http://www.sgim.org/index.cfm?pageId=846. Accessed July 2010.
- ,,,.Hospital medicine fellowships: works in progress.Am J Med.2006;119:72e1–72e7.
- ,,,.Promoting effective care transitions of hospital discharge. A review of key issues for hospitalists.J. Hosp Med.2007;2:314–323.
- ,.Hospitalists and anesthesiologists as perioperative physicians: are their roles complementary?Proc (Bayl Univ Med Cent).2007;20(2):140–142.
- .A systematic review of outcomes and quality measures in adult patients cared for by hospitalists vs nonhospitalist.Mayo Clin Proc.2009;84(3):248–254.
- .Reflections: the hospitalist movement a decade later.J Hosp Med.2006;1:248–252.
- .Anyone, Anything, Anytime A History of Emergency Medicine.1st ed.Philadelphia, PA:Mosby‐Elsevier;2006.
- ,,, et al.Career fit and burnout among academic faculty.Arch Intern Med.2009;169(10):990–995.
Hospitalists are hospital‐based physicians whose primary professional focus is patient care, education, research, and administrative activities related to hospital medicine.1 Initially, community‐based hospitals were far more likely to employ hospitalists than academic centers. However, today most academic centers employ hospitalist models and it is now a fully recognized entity in academic settings.2
While much has been written about the structure, business operations, and potential benefits of nonteaching (clinical) hospitalist programs,3, 4 there is little known about the current state of academic hospitalist programs or their challenges. For example, who are the leaders of academic hospitalist medicine groups? Given the youth of the field, are academic hospitalists receiving adequate mentorship and are they advancing academically? What are future directions and goals for academic hospitalist groups?
To better understand academic hospitalist programs, we surveyed division chiefs and academic hospitalist leaders to explore existing business models and operations, the status of mentorship, and key issues in growth and retention.
Methods
Sites and Subjects
We targeted potential hospital medicine group leaders by identifying academic medical centers using Association of American Medical Colleges (AAMC), the Accreditation Council for Graduate Medical Education (ACGME), the Association of Chiefs of General Internal Medicine (ACGIM), and the Society of Hospital Medicine (SHM) lists of sites with teaching missions. We then used publicly available data (eg, from websites maintained by the sites) to identify physician leaders who: 1) self identified as a leader of a hospitalist group at an academic medical center (or a Chief of Division of General Internal Medicine which managed a hospitalist group) in the SHM database, 2) were listed as such on the website, or 3) were members of ACGIM and listed as a hospitalist group leader at a university based medical center.
Survey Development
Our survey was based on questions used in previous research by the authors,5 with additional questions regarding operations of academic hospitalist programs, growth and retention of hospitalists, and mentorship developed by the study authors. Questions were pretested among a selected group of members of the Society of General Internal Medicine (SGIM) Academic Hospitalist Task Force and the SHM Academic Hospitalist Interest Group, after which the survey was refined and converted into its electronic form.
Survey Methods
The email survey process began in April 2007 with an initial survey sent to those physicians identified using preexisting data, as described. Our survey asked first if recipients were directly responsible for the oversight of a hospitalist group (eg, the division chief or director of the hospital medicine group) and if they practiced at an academic medical center. Only respondents who answered yes to both of these criteria were invited to respond to our survey. Those who felt the survey did not apply to them were invited to forward the email survey on to the appropriate person at their site or respond that their hospital had no hospital medicine service. Subsequent reminder emails were sent to nonrespondents at 10‐day intervals up to a total of four times. This survey was granted exempt status from the UCSF Institutional Review Board.
Statistical Methods
Response rates and frequencies and distribution of survey responses were analyzed using univariable statistics.
Results
Characteristics of Responding Sites
We received responses from 57 (40%) of the academic sites identified as having an academic hospital medicine group. Hospitalist group leaders at responding sites had been in their current position 3.8 years, graduated medical school approximately 15 years prior, and were either Assistant (40%), Associate (32%), or Full Professors (23%). Group leaders reported that the vast majority (91%) of group full‐time members were in junior faculty positions (Instructor or Assistant Professor), who were working full‐time. On average, responding programs were 6 years old (formed in 2001) and currently had 10.0 total full time equivalents (FTEs). A total of 38 of the groups (67%) were part of the larger Division of General Internal Medicine, whereas 9 groups (16 %) were their own division within the Department of Medicine. The remaining 17% were part of another division.
Mentorship Practices In Academic Hospital Medicine Groups
As one mechanism of mentorship, annual performance reviews were offered in most programs (88%). These were usually performed by the general medicine division chief or hospitalist leader. Mentoring relationships for clinician investigators (CI) were most often from personnel outside the hospitalist group, whereas clinician‐educators (CE) most often were mentored by faculty inside the group.
Hospitalist Leaders' Priorities and Impressions of Growth, Opportunities, Career Development and Barriers
Hospitalist leaders reported the highest priorities for hospitalist leaders were developing research and teaching programs, and minimizing turnover. Other priorities included achieving financial stability, applying for extramural funding, and reducing clinical workload (Table 2). Only 14% of respondents noted that becoming a separate division was a priority.0
| Characteristic | n (%) |
|---|---|
| |
| Group leader characteristics | |
| Academic rank | |
| Assistant professor/other | 26 (45) |
| Associate professor | 18 (32) |
| Full professor | 13 (23) |
| Years in position (mean, range) | 3.8 (2.07.0) |
| Group characteristics | |
| Hospital medicine place in school of medicine | |
| Within the department of medicine | 55 (98) |
| Separate division | 9 (16) |
| Within division of general medicine | 38 (67) |
| Other | 9 (16) |
| Not in the department of medicine | 1 (2) |
| Program size (mean, range) | |
| Number of hospitalists in program | 10 (718) |
| Number of FTE | 11 (3.512) |
| FTE's hired in past 2 years (July 2005 to survey date) | 4.0 (2.27.0) |
| Hospitalist activities | |
| Medicine consultation | 52 (91) |
| Quality improvement projects | 52 (91) |
| Nonteaching attending | 44 (77) |
| Comanagement of surgical patients | 44 (77) |
| 24‐hour coverage | 24 (61) |
| Manage patient transfer requests | 32 (56) |
| Peer review/morbidity and mortality | 31 (54) |
| Education program leadership | 29 (51) |
| Medical student program leadership | 29 (51) |
| Palliative care program | 23 (40) |
| Preoperative clinic | 23 (40) |
| Emergency department triage | 14 (25) |
| Post discharge follow‐up clinic | 13 (23) |
| Skill nursing facility coverage | 4 (7) |
| Other | 15 (26) |
| Mentorship Activity | n (%) |
|---|---|
| Programs performing annual reviews with faculty | 50 (88) |
| Who performs the annual review? | |
| General Medicine Division Chief | 9 (18) |
| Hospitalist leader | 18 (36) |
| Both | 13 (26) |
| Other (eg, Department Chair, Chief Medical Officer) | 10 (20) |
| Who is the primary source of mentorship for clinician‐educators? | |
| Senior faculty within the group | 43 (77) |
| Generalist faculty outside the group, but within the institution | 6 (11) |
| Subspecialty Internal Medicine faculty outside the group, but within the institution | 3 (5) |
| Non‐Internal Medicine (eg, surgeon, epidemiologist) outside the group, but within the institution | 0 (0) |
| Faculty from another institution | 0 (0) |
| Don't know | 4 (7) |
| Who is the primary source of mentorship for clinician‐investigators? | |
| Senior faculty within the group | 6 (12) |
| Generalist faculty outside the group, but within the institution | 13 (25) |
| Subspecialty internal medicine faculty outside the group, but within the institution | 6 (12) |
| Non‐Internal Medicine (eg, surgeon, epidemiologist) outside the group, but within the institution | 2 (4) |
| Faculty from another institution | 3 (6) |
| Don't know | 2 (4) |
| Not applicable; no clinician investigators | 20 (38) |
| Highest Priority, n (%) | Intermediate Priority, n (%) | Lowest Priority, n (%) | Not a Priority, n (%) | NA, n (%) | |
|---|---|---|---|---|---|
| Reducing individual faculty clinical workload | 9 (16) | 22 (3) | 11 (2) | 14 (2) | 0 (0) |
| Achieving financial stability | 13 (24) | 30 (55) | 6 (11) | 6 (11) | 0 (0) |
| Minimizing turnover | 22 (39) | 27 (48) | 6 (11) | 1 (2) | 0 (0) |
| Developing teaching programs | 22 (39) | 29 (52) | 3 (5) | 2 (4) | 0 (0) |
| Becoming a separate division | 3 (5) | 5 (9) | 11 (20) | 23 (41) | 14 (25) |
| Developing research | 25 (45) | 18 (32) | 5 (9) | 6 (11) | 2 (4) |
| Applying for extramural funding | 10 (18) | 24 (43) | 10 (18) | 8 (14) | 4 (7) |
| Strongly Agree, n (%) | Agree, n (%) | Neutral, n (%) | Disagree, n (%) | Strongly Disagree, n (%) | NA, n (%) | |
|---|---|---|---|---|---|---|
| Growth and sustainability | ||||||
| Availability of funds is limiting expansion of academic functions (eg, education and research) | 20 (36) | 21 (38) | 5 (9) | 7 (12) | 3 (5) | 0 (0) |
| Availability of funds is limiting expansion of clinical functions (eg, development of new services) | 11 (20) | 17 (30) | 14 (25) | 10 (18) | 4 (7) | 0 (0) |
| My faculty are developing sustainable nonclinical activities | 9 (16) | 23 (41) | 12 (21) | 9 (16) | 3 (5) | 0 (0) |
| Career development | ||||||
| Mentorship is a major issue for my clinician‐educator faculty | 14 (25) | 28 (50) | 7 (12) | 4 (7) | 1 (2) | 2 (4) |
| Mentorship is a major issue for my research faculty | 22 (40) | 10 (18) | 4 (7) | 3 (5) | 2 (4) | 14 (25) |
| External support for hospital medicine group | ||||||
| There is investment in the development of academic functions of our hospitalist program from my hospital | 4 (7) | 12 (21) | 10 (18) | 22 (39) | 8 (14) | 0 (0) |
| There is investment in the development of academic functions of our hospitalist program from the Department of Medicine | 22 (40) | 17 (31) | 8 (15) | 4 (7) | 2 (4) | 2 (4) |
In general, academic hospitalist leaders reported that Departments of Medicine and Divisions of General Medicine (where applicable) were invested in the development of their academic functions. Yet, more than half of program directors reported that hospitals were not supportive. Moreover, lack of funds limited the expansion of their academic or clinical functions (Table 3). Additionally, while the majority either strongly agree or agree that their faculty are developing sustainable nonclinical activities (57%), they perceive that they are at risk for burnout (69%), and that lack of mentorship is a major issue for both CE (75%) and research faculty (58%). Lastly, while program directors strongly agree or agree (71%) that their hospitalist groups are respected by other academic physicians, they additionally strongly agree or agree that their Departments of Medicine (58%) and other Divisions (78%) view their hospitalist program as a clinical service rather than an academic program.
Discussion
Our survey provides a unique snapshot of academic hospitalist groups, highlighting a perceived lack of support and respect for their programs, a need to increase education and scholarly activities, and a desire to better prepare faculty for academic promotion.
Academic hospitalist groups and leaders reflected what one would expect from a field that is just over a decade old. Program leaders were relatively new to their position, as were their division group members. As a result, it is not surprising that most of the academic hospitalist leaders identified mentorship as a major issue. We were encouraged to see that most programs were offering annual reviews. However, the majority of these annual reviews were performed by the group leaders, many of whom are relatively junior (40% Assistant Professors) and may not be experienced in mentoring and performing annual reviews. Importantly, the absence of a mentor (or a high‐quality, experienced one) among physicians, and specifically hospitalists, may result in fewer peer‐reviewed first author and non‐peer‐reviewed publications, and less experience leading a teaching session at a national meeting.6 Research suggests that effective mentoring may help faculty increase career satisfaction and productivity and reduce their risk for burn‐out.7 Hospitalist groups might benefit nationally from focusing specifically on finding adequate mentorship either within or outside their groups. In addition, national organizations such as the SHM and the SGIM could potentially help these groups and individual hospitalists in creating mentorship networks and a mentoring infrastructure.
Academic hospitalist leaders were concerned about the ability of their faculty to develop sustainable nonclinical activities and scholarship. Notably, more than 40% of surveyed leaders agreed or strongly agreed that their faculty were not developing sustainable nonclinical activities. For individual faculty, the inability to develop scholarly activities and engage in academic pursuits may create challenges in getting promoted by traditional academic pathways. Some have recognized this issue and tried to develop practical solutions.2 In addition, academic hospitalists often engage in nonclinical activities such as quality improvement or patient safety which do not fit in the traditional tripartite mission of academics (clinical care, education, and research). In this survey, more than 90% of groups were engaged in quality improvement projects and over half in peer review exercises (Table 1). As many of these scholarly activities require innovation, sophisticated data analysis, and can have far‐reaching and substantial impacts on healthcare, some have argued these should be considered as part of the promotion process.8 Notably, the SGIM Academic Hospitalist Taskforce has created the Quality Portfolio, a structured adjunct to promotions packets to organize and document work in quality improvement and patient safety.9
While there were few CI in the divisions surveyed, building CI programs was a major priority of programs. In programs reporting the presence of CI's, they report limited access to research support. This highlights the potential role and benefit of post residency training in designing and conducting clinical research whether in a traditional general internal medicine fellowship or in 1 of the many growing hospital medicine fellowships.10 There also appears to be a need for funding to support the research careers of junior hospitalists. While access to effective mentorship is integrally linked to achieving increased academic accomplishments, there is certainly an ample call for research in the areas of quality improvement, patient safety; systems‐based practice, hospital efficiency, transitions of care,11 perioperative medicine,12 and education.2, 13, 14 While providing lower costs per admission and lower lengths of stay, hospitalists seem well‐positioned to spearhead active research in cost‐effectiveness in the hospital.14 Additionally, a quality portfolio, documenting such quality improvement projects, has been suggested as an effective means to provide a record of this work for academic promotion.9
The diverse activities of today's hospitalists are transforming the traditional view of academic work and are critical to the growth of hospitals, patient care, and development of the field of hospital medicine itself. Until these areas are fully embraced as legitimate areas of academic productivity and scholarship, the academic advancement of hospitalists will be slow.
It is unclear from our survey if academic hospitalist programs are truly getting the support they need to succeed. On one hand, there was general agreement that the Departments of Medicine and Divisions of General and Hospital Medicine were invested in the development of the academic accomplishments. Yet, the majority of program directors believed that they are viewed by the Department or Division as a clinical rather than an academic program. Moreover, over half of program directors report that their hospital was not supportive and therefore have limited the expansion of their hospitalist groups' educational and research activities. Lastly, for a large majority of programs, unavailable funding also acted to limit growth and expansion of academic functions. In a mere 2 decades, Emergency Medicine has become one of the largest US specialties and yet research and funding in the field have been lagging and are limiting academic expansion. Junior faculty seeking research careers struggled to find support and mentorship within their emergency medicine divisions.15 Challenges faced by academic emergency medicine provide important historical perspective for the even more rapidly growing field of academic hospital medicine. Learning from the Academic Emergency Medicine experience, academic hospitalists should proactively identify scholarship and research opportunities unique to hospitalist and fitting the needs of academic institutions. Involvement in national medical organizations, such as SGIM‐SHM‐ACGIM Academic Hospitalist Academy, or the SGIM Academic Hospitalist Task Force, where skill and career development is the focus, will undoubtedly promote the success of academic hospitalist. Expanding valuable niches of expertise, such as quality control, perioperative medicine and care transitions, create an indispensible component of hospital care. Lastly mentoring programs for academic hospitalist within SHM and SGIM are also essential for networking and career development. There are several limitations to our study. Our response rate of 40% was relatively low, and our results may not be representative of all academic hospitalist division chiefs and their programs, may be overstating the perceived difficulties of the survey sample, or conversely missing a large portion too overwhelmed by current duties who lacked the time to complete the survey. Having said this, our survey methodology targeted sites where we could identify potentialnot confirmedhospitalist groups and hospitalist group leaders. For this reason, our response rate could be higher (if some of our contacts were in error). Our results are a cross‐sectional survey based on self report and are subject to recall bias. In addition, our study was carried out in 2007, and while issues such as mentorship may remain important, our results regarding financial arrangements may not be applicable to the current economic climate. Finally, while improving mentorship was identified as a principle objective for program leaders, we did not explore the existing quality of mentorship, nor perceived shortfalls. This should be the subject of future exploration.
The vast majority of academic hospital medicine programs continue to view inadequate support, expanding research, mentorship, and academic promotion as critical issues for the future. Thus, further understanding of these features, and interventions to allow for success, are of crucial importance in the continued development of academic hospitalists. Our study supports the need for mentoring and career development programs, targeting academic hospitalists and their leaders. In addition, attention should be paid to activities that support career fit, creating sustainable and viable job descriptions for academic hospitalists, and preventing burnout.16 At the same time we must expand the traditional view of scholarship and training and advocate for promotion criteria that value the unique contributions of hospitalists to become in line with the broad areas that hospitalists work.
Hospitalists are hospital‐based physicians whose primary professional focus is patient care, education, research, and administrative activities related to hospital medicine.1 Initially, community‐based hospitals were far more likely to employ hospitalists than academic centers. However, today most academic centers employ hospitalist models and it is now a fully recognized entity in academic settings.2
While much has been written about the structure, business operations, and potential benefits of nonteaching (clinical) hospitalist programs,3, 4 there is little known about the current state of academic hospitalist programs or their challenges. For example, who are the leaders of academic hospitalist medicine groups? Given the youth of the field, are academic hospitalists receiving adequate mentorship and are they advancing academically? What are future directions and goals for academic hospitalist groups?
To better understand academic hospitalist programs, we surveyed division chiefs and academic hospitalist leaders to explore existing business models and operations, the status of mentorship, and key issues in growth and retention.
Methods
Sites and Subjects
We targeted potential hospital medicine group leaders by identifying academic medical centers using Association of American Medical Colleges (AAMC), the Accreditation Council for Graduate Medical Education (ACGME), the Association of Chiefs of General Internal Medicine (ACGIM), and the Society of Hospital Medicine (SHM) lists of sites with teaching missions. We then used publicly available data (eg, from websites maintained by the sites) to identify physician leaders who: 1) self identified as a leader of a hospitalist group at an academic medical center (or a Chief of Division of General Internal Medicine which managed a hospitalist group) in the SHM database, 2) were listed as such on the website, or 3) were members of ACGIM and listed as a hospitalist group leader at a university based medical center.
Survey Development
Our survey was based on questions used in previous research by the authors,5 with additional questions regarding operations of academic hospitalist programs, growth and retention of hospitalists, and mentorship developed by the study authors. Questions were pretested among a selected group of members of the Society of General Internal Medicine (SGIM) Academic Hospitalist Task Force and the SHM Academic Hospitalist Interest Group, after which the survey was refined and converted into its electronic form.
Survey Methods
The email survey process began in April 2007 with an initial survey sent to those physicians identified using preexisting data, as described. Our survey asked first if recipients were directly responsible for the oversight of a hospitalist group (eg, the division chief or director of the hospital medicine group) and if they practiced at an academic medical center. Only respondents who answered yes to both of these criteria were invited to respond to our survey. Those who felt the survey did not apply to them were invited to forward the email survey on to the appropriate person at their site or respond that their hospital had no hospital medicine service. Subsequent reminder emails were sent to nonrespondents at 10‐day intervals up to a total of four times. This survey was granted exempt status from the UCSF Institutional Review Board.
Statistical Methods
Response rates and frequencies and distribution of survey responses were analyzed using univariable statistics.
Results
Characteristics of Responding Sites
We received responses from 57 (40%) of the academic sites identified as having an academic hospital medicine group. Hospitalist group leaders at responding sites had been in their current position 3.8 years, graduated medical school approximately 15 years prior, and were either Assistant (40%), Associate (32%), or Full Professors (23%). Group leaders reported that the vast majority (91%) of group full‐time members were in junior faculty positions (Instructor or Assistant Professor), who were working full‐time. On average, responding programs were 6 years old (formed in 2001) and currently had 10.0 total full time equivalents (FTEs). A total of 38 of the groups (67%) were part of the larger Division of General Internal Medicine, whereas 9 groups (16 %) were their own division within the Department of Medicine. The remaining 17% were part of another division.
Mentorship Practices In Academic Hospital Medicine Groups
As one mechanism of mentorship, annual performance reviews were offered in most programs (88%). These were usually performed by the general medicine division chief or hospitalist leader. Mentoring relationships for clinician investigators (CI) were most often from personnel outside the hospitalist group, whereas clinician‐educators (CE) most often were mentored by faculty inside the group.
Hospitalist Leaders' Priorities and Impressions of Growth, Opportunities, Career Development and Barriers
Hospitalist leaders reported the highest priorities for hospitalist leaders were developing research and teaching programs, and minimizing turnover. Other priorities included achieving financial stability, applying for extramural funding, and reducing clinical workload (Table 2). Only 14% of respondents noted that becoming a separate division was a priority.0
| Characteristic | n (%) |
|---|---|
| |
| Group leader characteristics | |
| Academic rank | |
| Assistant professor/other | 26 (45) |
| Associate professor | 18 (32) |
| Full professor | 13 (23) |
| Years in position (mean, range) | 3.8 (2.07.0) |
| Group characteristics | |
| Hospital medicine place in school of medicine | |
| Within the department of medicine | 55 (98) |
| Separate division | 9 (16) |
| Within division of general medicine | 38 (67) |
| Other | 9 (16) |
| Not in the department of medicine | 1 (2) |
| Program size (mean, range) | |
| Number of hospitalists in program | 10 (718) |
| Number of FTE | 11 (3.512) |
| FTE's hired in past 2 years (July 2005 to survey date) | 4.0 (2.27.0) |
| Hospitalist activities | |
| Medicine consultation | 52 (91) |
| Quality improvement projects | 52 (91) |
| Nonteaching attending | 44 (77) |
| Comanagement of surgical patients | 44 (77) |
| 24‐hour coverage | 24 (61) |
| Manage patient transfer requests | 32 (56) |
| Peer review/morbidity and mortality | 31 (54) |
| Education program leadership | 29 (51) |
| Medical student program leadership | 29 (51) |
| Palliative care program | 23 (40) |
| Preoperative clinic | 23 (40) |
| Emergency department triage | 14 (25) |
| Post discharge follow‐up clinic | 13 (23) |
| Skill nursing facility coverage | 4 (7) |
| Other | 15 (26) |
| Mentorship Activity | n (%) |
|---|---|
| Programs performing annual reviews with faculty | 50 (88) |
| Who performs the annual review? | |
| General Medicine Division Chief | 9 (18) |
| Hospitalist leader | 18 (36) |
| Both | 13 (26) |
| Other (eg, Department Chair, Chief Medical Officer) | 10 (20) |
| Who is the primary source of mentorship for clinician‐educators? | |
| Senior faculty within the group | 43 (77) |
| Generalist faculty outside the group, but within the institution | 6 (11) |
| Subspecialty Internal Medicine faculty outside the group, but within the institution | 3 (5) |
| Non‐Internal Medicine (eg, surgeon, epidemiologist) outside the group, but within the institution | 0 (0) |
| Faculty from another institution | 0 (0) |
| Don't know | 4 (7) |
| Who is the primary source of mentorship for clinician‐investigators? | |
| Senior faculty within the group | 6 (12) |
| Generalist faculty outside the group, but within the institution | 13 (25) |
| Subspecialty internal medicine faculty outside the group, but within the institution | 6 (12) |
| Non‐Internal Medicine (eg, surgeon, epidemiologist) outside the group, but within the institution | 2 (4) |
| Faculty from another institution | 3 (6) |
| Don't know | 2 (4) |
| Not applicable; no clinician investigators | 20 (38) |
| Highest Priority, n (%) | Intermediate Priority, n (%) | Lowest Priority, n (%) | Not a Priority, n (%) | NA, n (%) | |
|---|---|---|---|---|---|
| Reducing individual faculty clinical workload | 9 (16) | 22 (3) | 11 (2) | 14 (2) | 0 (0) |
| Achieving financial stability | 13 (24) | 30 (55) | 6 (11) | 6 (11) | 0 (0) |
| Minimizing turnover | 22 (39) | 27 (48) | 6 (11) | 1 (2) | 0 (0) |
| Developing teaching programs | 22 (39) | 29 (52) | 3 (5) | 2 (4) | 0 (0) |
| Becoming a separate division | 3 (5) | 5 (9) | 11 (20) | 23 (41) | 14 (25) |
| Developing research | 25 (45) | 18 (32) | 5 (9) | 6 (11) | 2 (4) |
| Applying for extramural funding | 10 (18) | 24 (43) | 10 (18) | 8 (14) | 4 (7) |
| Strongly Agree, n (%) | Agree, n (%) | Neutral, n (%) | Disagree, n (%) | Strongly Disagree, n (%) | NA, n (%) | |
|---|---|---|---|---|---|---|
| Growth and sustainability | ||||||
| Availability of funds is limiting expansion of academic functions (eg, education and research) | 20 (36) | 21 (38) | 5 (9) | 7 (12) | 3 (5) | 0 (0) |
| Availability of funds is limiting expansion of clinical functions (eg, development of new services) | 11 (20) | 17 (30) | 14 (25) | 10 (18) | 4 (7) | 0 (0) |
| My faculty are developing sustainable nonclinical activities | 9 (16) | 23 (41) | 12 (21) | 9 (16) | 3 (5) | 0 (0) |
| Career development | ||||||
| Mentorship is a major issue for my clinician‐educator faculty | 14 (25) | 28 (50) | 7 (12) | 4 (7) | 1 (2) | 2 (4) |
| Mentorship is a major issue for my research faculty | 22 (40) | 10 (18) | 4 (7) | 3 (5) | 2 (4) | 14 (25) |
| External support for hospital medicine group | ||||||
| There is investment in the development of academic functions of our hospitalist program from my hospital | 4 (7) | 12 (21) | 10 (18) | 22 (39) | 8 (14) | 0 (0) |
| There is investment in the development of academic functions of our hospitalist program from the Department of Medicine | 22 (40) | 17 (31) | 8 (15) | 4 (7) | 2 (4) | 2 (4) |
In general, academic hospitalist leaders reported that Departments of Medicine and Divisions of General Medicine (where applicable) were invested in the development of their academic functions. Yet, more than half of program directors reported that hospitals were not supportive. Moreover, lack of funds limited the expansion of their academic or clinical functions (Table 3). Additionally, while the majority either strongly agree or agree that their faculty are developing sustainable nonclinical activities (57%), they perceive that they are at risk for burnout (69%), and that lack of mentorship is a major issue for both CE (75%) and research faculty (58%). Lastly, while program directors strongly agree or agree (71%) that their hospitalist groups are respected by other academic physicians, they additionally strongly agree or agree that their Departments of Medicine (58%) and other Divisions (78%) view their hospitalist program as a clinical service rather than an academic program.
Discussion
Our survey provides a unique snapshot of academic hospitalist groups, highlighting a perceived lack of support and respect for their programs, a need to increase education and scholarly activities, and a desire to better prepare faculty for academic promotion.
Academic hospitalist groups and leaders reflected what one would expect from a field that is just over a decade old. Program leaders were relatively new to their position, as were their division group members. As a result, it is not surprising that most of the academic hospitalist leaders identified mentorship as a major issue. We were encouraged to see that most programs were offering annual reviews. However, the majority of these annual reviews were performed by the group leaders, many of whom are relatively junior (40% Assistant Professors) and may not be experienced in mentoring and performing annual reviews. Importantly, the absence of a mentor (or a high‐quality, experienced one) among physicians, and specifically hospitalists, may result in fewer peer‐reviewed first author and non‐peer‐reviewed publications, and less experience leading a teaching session at a national meeting.6 Research suggests that effective mentoring may help faculty increase career satisfaction and productivity and reduce their risk for burn‐out.7 Hospitalist groups might benefit nationally from focusing specifically on finding adequate mentorship either within or outside their groups. In addition, national organizations such as the SHM and the SGIM could potentially help these groups and individual hospitalists in creating mentorship networks and a mentoring infrastructure.
Academic hospitalist leaders were concerned about the ability of their faculty to develop sustainable nonclinical activities and scholarship. Notably, more than 40% of surveyed leaders agreed or strongly agreed that their faculty were not developing sustainable nonclinical activities. For individual faculty, the inability to develop scholarly activities and engage in academic pursuits may create challenges in getting promoted by traditional academic pathways. Some have recognized this issue and tried to develop practical solutions.2 In addition, academic hospitalists often engage in nonclinical activities such as quality improvement or patient safety which do not fit in the traditional tripartite mission of academics (clinical care, education, and research). In this survey, more than 90% of groups were engaged in quality improvement projects and over half in peer review exercises (Table 1). As many of these scholarly activities require innovation, sophisticated data analysis, and can have far‐reaching and substantial impacts on healthcare, some have argued these should be considered as part of the promotion process.8 Notably, the SGIM Academic Hospitalist Taskforce has created the Quality Portfolio, a structured adjunct to promotions packets to organize and document work in quality improvement and patient safety.9
While there were few CI in the divisions surveyed, building CI programs was a major priority of programs. In programs reporting the presence of CI's, they report limited access to research support. This highlights the potential role and benefit of post residency training in designing and conducting clinical research whether in a traditional general internal medicine fellowship or in 1 of the many growing hospital medicine fellowships.10 There also appears to be a need for funding to support the research careers of junior hospitalists. While access to effective mentorship is integrally linked to achieving increased academic accomplishments, there is certainly an ample call for research in the areas of quality improvement, patient safety; systems‐based practice, hospital efficiency, transitions of care,11 perioperative medicine,12 and education.2, 13, 14 While providing lower costs per admission and lower lengths of stay, hospitalists seem well‐positioned to spearhead active research in cost‐effectiveness in the hospital.14 Additionally, a quality portfolio, documenting such quality improvement projects, has been suggested as an effective means to provide a record of this work for academic promotion.9
The diverse activities of today's hospitalists are transforming the traditional view of academic work and are critical to the growth of hospitals, patient care, and development of the field of hospital medicine itself. Until these areas are fully embraced as legitimate areas of academic productivity and scholarship, the academic advancement of hospitalists will be slow.
It is unclear from our survey if academic hospitalist programs are truly getting the support they need to succeed. On one hand, there was general agreement that the Departments of Medicine and Divisions of General and Hospital Medicine were invested in the development of the academic accomplishments. Yet, the majority of program directors believed that they are viewed by the Department or Division as a clinical rather than an academic program. Moreover, over half of program directors report that their hospital was not supportive and therefore have limited the expansion of their hospitalist groups' educational and research activities. Lastly, for a large majority of programs, unavailable funding also acted to limit growth and expansion of academic functions. In a mere 2 decades, Emergency Medicine has become one of the largest US specialties and yet research and funding in the field have been lagging and are limiting academic expansion. Junior faculty seeking research careers struggled to find support and mentorship within their emergency medicine divisions.15 Challenges faced by academic emergency medicine provide important historical perspective for the even more rapidly growing field of academic hospital medicine. Learning from the Academic Emergency Medicine experience, academic hospitalists should proactively identify scholarship and research opportunities unique to hospitalist and fitting the needs of academic institutions. Involvement in national medical organizations, such as SGIM‐SHM‐ACGIM Academic Hospitalist Academy, or the SGIM Academic Hospitalist Task Force, where skill and career development is the focus, will undoubtedly promote the success of academic hospitalist. Expanding valuable niches of expertise, such as quality control, perioperative medicine and care transitions, create an indispensible component of hospital care. Lastly mentoring programs for academic hospitalist within SHM and SGIM are also essential for networking and career development. There are several limitations to our study. Our response rate of 40% was relatively low, and our results may not be representative of all academic hospitalist division chiefs and their programs, may be overstating the perceived difficulties of the survey sample, or conversely missing a large portion too overwhelmed by current duties who lacked the time to complete the survey. Having said this, our survey methodology targeted sites where we could identify potentialnot confirmedhospitalist groups and hospitalist group leaders. For this reason, our response rate could be higher (if some of our contacts were in error). Our results are a cross‐sectional survey based on self report and are subject to recall bias. In addition, our study was carried out in 2007, and while issues such as mentorship may remain important, our results regarding financial arrangements may not be applicable to the current economic climate. Finally, while improving mentorship was identified as a principle objective for program leaders, we did not explore the existing quality of mentorship, nor perceived shortfalls. This should be the subject of future exploration.
The vast majority of academic hospital medicine programs continue to view inadequate support, expanding research, mentorship, and academic promotion as critical issues for the future. Thus, further understanding of these features, and interventions to allow for success, are of crucial importance in the continued development of academic hospitalists. Our study supports the need for mentoring and career development programs, targeting academic hospitalists and their leaders. In addition, attention should be paid to activities that support career fit, creating sustainable and viable job descriptions for academic hospitalists, and preventing burnout.16 At the same time we must expand the traditional view of scholarship and training and advocate for promotion criteria that value the unique contributions of hospitalists to become in line with the broad areas that hospitalists work.
- Anonymous. Definition of a hospitalist.2009. Society of Hospital Medicine Homepage/General Information. Available at: http://www.hospitalmedicine.org/AM/Template.cfm?Section=Hospitalist_Definition4:240–246.
- ,,,,.Resource‐based relative value scale analysis between teaching and nonteaching hospitalist services.Health Care Management.2009;1(28):81–85.
- ,,, et al.Effects of physician experience on costs and outcomes on an academic general medicine service.Ann Intern Med.2002;137:866–874.
- ,,,.Hospitalists and the practice of inpatient medicine.Ann Intern Med.1999;130:343–349.
- ,,,,,.Rates, predictors and consequences of low career satisfaction and burnout in academic hospital medicine.J Hosp Med.2009;4(S1):24–25.
- ,.Mentoring faculty in academic medicine: a new paradigm?.J Gen Intern Med.2005;20(9):866–870.
- ,.Clinicians in quality improvement: a new career pathway in academic medicine.JAMA.2009;301(7):766–768.
- ,,,.Academic hospitalist taskforce quality portfolio rationale and development. 02/23/2009; Quality portfolio introduction. Available at:http://www.sgim.org/index.cfm?pageId=846. Accessed July 2010.
- ,,,.Hospital medicine fellowships: works in progress.Am J Med.2006;119:72e1–72e7.
- ,,,.Promoting effective care transitions of hospital discharge. A review of key issues for hospitalists.J. Hosp Med.2007;2:314–323.
- ,.Hospitalists and anesthesiologists as perioperative physicians: are their roles complementary?Proc (Bayl Univ Med Cent).2007;20(2):140–142.
- .A systematic review of outcomes and quality measures in adult patients cared for by hospitalists vs nonhospitalist.Mayo Clin Proc.2009;84(3):248–254.
- .Reflections: the hospitalist movement a decade later.J Hosp Med.2006;1:248–252.
- .Anyone, Anything, Anytime A History of Emergency Medicine.1st ed.Philadelphia, PA:Mosby‐Elsevier;2006.
- ,,, et al.Career fit and burnout among academic faculty.Arch Intern Med.2009;169(10):990–995.
- Anonymous. Definition of a hospitalist.2009. Society of Hospital Medicine Homepage/General Information. Available at: http://www.hospitalmedicine.org/AM/Template.cfm?Section=Hospitalist_Definition4:240–246.
- ,,,,.Resource‐based relative value scale analysis between teaching and nonteaching hospitalist services.Health Care Management.2009;1(28):81–85.
- ,,, et al.Effects of physician experience on costs and outcomes on an academic general medicine service.Ann Intern Med.2002;137:866–874.
- ,,,.Hospitalists and the practice of inpatient medicine.Ann Intern Med.1999;130:343–349.
- ,,,,,.Rates, predictors and consequences of low career satisfaction and burnout in academic hospital medicine.J Hosp Med.2009;4(S1):24–25.
- ,.Mentoring faculty in academic medicine: a new paradigm?.J Gen Intern Med.2005;20(9):866–870.
- ,.Clinicians in quality improvement: a new career pathway in academic medicine.JAMA.2009;301(7):766–768.
- ,,,.Academic hospitalist taskforce quality portfolio rationale and development. 02/23/2009; Quality portfolio introduction. Available at:http://www.sgim.org/index.cfm?pageId=846. Accessed July 2010.
- ,,,.Hospital medicine fellowships: works in progress.Am J Med.2006;119:72e1–72e7.
- ,,,.Promoting effective care transitions of hospital discharge. A review of key issues for hospitalists.J. Hosp Med.2007;2:314–323.
- ,.Hospitalists and anesthesiologists as perioperative physicians: are their roles complementary?Proc (Bayl Univ Med Cent).2007;20(2):140–142.
- .A systematic review of outcomes and quality measures in adult patients cared for by hospitalists vs nonhospitalist.Mayo Clin Proc.2009;84(3):248–254.
- .Reflections: the hospitalist movement a decade later.J Hosp Med.2006;1:248–252.
- .Anyone, Anything, Anytime A History of Emergency Medicine.1st ed.Philadelphia, PA:Mosby‐Elsevier;2006.
- ,,, et al.Career fit and burnout among academic faculty.Arch Intern Med.2009;169(10):990–995.
Copyright © 2011 Society of Hospital Medicine
RIP Conference Provides Peer Mentoring
The research‐in‐progress (RIP) conference is commonplace in academia, but there are no studies that objectively characterize its value. Bringing faculty together away from revenue‐generating activities carries a significant cost. As such, measuring the success of such gatherings is necessary.
Mentors are an invaluable influence on the careers of junior faculty members, helping them to produce high‐quality research.13 Unfortunately, some divisions lack mentorship to support the academic needs of less experienced faculty.1 Peer mentorship may be a solution. RIP sessions represent an opportunity to intentionally formalize peer mentoring. Further, these sessions can facilitate collaborations as individuals become aware of colleagues' interests. The goal of this study was to assess the value of the research‐in‐progress conference initiated within the hospitalist division at our institution.
Methods
Study Design
This cohort study was conducted to evaluate the value of the RIP conference among hospitalists in our division and the academic outcomes of the projects.
Setting and Participants
The study took place at Johns Hopkins Bayview Medical Center (JHBMC), a 335‐bed university‐affiliated medical center in Baltimore, Maryland. The hospitalist division consists of faculty physicians, nurse practitioners, and physician assistants (20.06 FTE physicians and 7.41 FTE midlevel providers). Twelve (54%) of our faculty members are female, and the mean age of providers is 35.7 years. The providers have been practicing hospitalist medicine for 3.0 years on average; 2 (9%) are clinical associates, 16 (73%) are instructors, and 3 (14%) are assistant professors.
All faculty members presenting at the RIP session were members of the division. A senior faculty member (a professor in the Division of General Internal Medicine) helps to coordinate the conference. The group's research assistant was present at the sessions and was charged with data collection and collation.
The Johns Hopkins University institutional review board approved the study.
The Research in Progress Conference
During the 2009 academic year, our division held 15 RIP sessions. At each session, 1 faculty member presented a research proposal. The goal of each session was to provide a forum where faculty members could share their research ideas (specific aims, hypotheses, planned design, outcome measures, analytic plans, and preliminary results [if applicable]) in order to receive feedback. The senior faculty member met with the presenter prior to each session in order to: (1) ensure that half the RIP time was reserved for discussion and (2) review the presenter's goals so these would be made explicit to peers. The coordinator of the RIP conference facilitated the discussion, solicited input from all attendees, and encouraged constructive criticism.
Evaluation, Data Collection, and Analysis
At the end of each session, attendees (who were exclusively members of the hospitalist division) were asked to complete an anonymous survey. The 1‐page instrument was designed (1) with input from curriculum development experts4 and (2) after a review of the literature about RIP conferences. These steps conferred content validity to the instrument, which assessed perceptions about the session's quality and what was learned. Five‐point Likert scales were used to characterize the conference's success in several areas, including being intellectually/professionally stimulating and keeping them apprised of their colleagues' interests. The survey also assessed the participatory nature of the conference (balance of presentation vs discussion), its climate (extremely critical vs extremely supportive), and how the conference assisted the presenter. The presenters completed a distinct survey related to how helpful the conference was in improving/enhancing their projects. A final open‐ended section invited additional comments. The instrument was piloted and iteratively revised before its use in this study.
For the projects presented, we assessed the percentage that resulted in a peer‐reviewed publication or a presentation at a national meeting.
Results
The mean number of attendees at the RIP sessions was 9.6 persons. A total of 143 evaluations were completed. All 15 presenters (100%) completed their assessments. The research ideas presented spanned a breadth of topics in clinical research, quality improvement, policy, and professional development (Table 1).
| Session | Date | Presenter | Topic | Evaluations Completed |
|---|---|---|---|---|
| 1 | 7/2008 | Dr. CS | Hospital medicine in Canada versus the United States | 7 |
| 2 | 7/2008 | Dr. RT | Procedures by hospitalists | 9 |
| 3 | 8/2008 | Dr. MA | Clostridium difficile treatment in the hospital | 11 |
| 4 | 8/2008 | Dr. EH | Active bed management | 6 |
| 5 | 9/2008 | Dr. AS | Medication reconciliation for geriatric inpatients | 10 |
| 6 | 9/2008 | Dr. DT | Time‐motion study of hospitalists | 10 |
| 7 | 10/2008 | Dr. KV | e‐Triage pilot | 16 |
| 8 | 11/2008 | Dr. EH | Assessing clinical performance of hospitalists | 7 |
| 9 | 12/2008 | Dr. SC | Trends and implications of hospitalists' morale | 8 |
| 10 | 1/2009 | Dr. TB | Lessons learned: tracking urinary catheter use at Bayview | 11 |
| 11 | 2/2009 | Dr. FK | Utilizing audit and feedback to improve performance in tobacco dependence counseling | 12 |
| 12 | 3/2009 | Dr. MK | Survivorship care plans | 7 |
| 13 | 4/2009 | Dr. DK | Outpatient provider preference for discharge summary format/style/length | 7 |
| 14 | 5/2009 | Dr. RW | Comparing preoperative consults done by hospitalists and cardiologists | 11 |
| 15 | 6/2009 | Dr. AK | Development of Web‐based messaging tool for providers | 12 |
Presenter Perspective
All 15 presenters (100%) felt a lot or tremendously supported during their sessions. Thirteen physicians (86%) believed that the sessions were a lot or tremendously helpful in advancing their projects. The presenters believed that the guidance and discussions related to their research ideas, aims, hypotheses, and plans were most helpful for advancing their projects (Table 2).
| Not at All, n (%) | A Little, n (%) | Some, n (%) | A Lot, n (%) | Tremendously, n (%) | |
|---|---|---|---|---|---|
| General questions: | |||||
| Intellectually/professionally stimulating | 0 (0) | 0 (0) | 0 (0) | 5 (33) | 10 (66) |
| Feeling supported by your colleagues in your scholarly pursuits | 0 (0) | 0 (0) | 0 (0) | 4 (27) | 11 (73) |
| Session helpful in the following areas: | |||||
| Advancing your project | 0 (0) | 0 (0) | 2 (13) | 5 (33) | 8 (53) |
| Generated new hypotheses | 1 (6) | 3 (20) | 5 (33) | 5 (33) | 1 (6) |
| Clarification of research questions | 0 (0) | 2 (13) | 4 (27) | 7 (47) | 2 (13) |
| Ideas for alternate methods | 1 (6) | 1 (6) | 2 (13) | 7 (47) | 4 (27) |
| New outcomes suggested | 1 (6) | 2 (13) | 2 (13) | 5 (33) | 5 (33) |
| Strategies to improve or enhance data collection | 0 (0) | 2 (13) | 0 (0) | 8 (53) | 5 (33) |
| Suggestions for alternate analyses or analytical strategies | 1 (1) | 1 (6) | 4 (27) | 5 (33) | 4 (27) |
| Input into what is most novel/emnteresting about this work | 0 (0) | 2 (13) | 3 (20) | 6 (40) | 4 (27) |
| Guidance about the implications of the work | 1 (6) | 2 (13) | 1 (6) | 7 (47) | 4 (27) |
| Ideas about next steps or future direction/studies | 0 (0) | 0 (0) | 3 (21) | 8 (57) | 3 (21) |
Examples of the written comments are:
I was overwhelmed by how engaged people were in my project.
The process of preparing for the session and then the discussion both helped my thinking. Colleagues were very supportive.
I am so glad I heard these comments and received this feedback now, rather than from peer reviewers selected by a journal to review my study. It would have been a much more difficult situation to fix at that later time.
Attendee Perspective
The majority of attendees (123 of 143, 86%) found the sessions to be a lot or extremely stimulating, and almost all (96%) were a lot or extremely satisfied with how the RIP sessions kept them abreast of their colleagues' academic interests. In addition, 92% judged the session's climate to be a lot or extremely supportive, and 88% deemed the balance of presentation to discussion to be just right. Attendees believed that they were most helpful to the presenter in terms of conceiving ideas for alternative methods to be used to answer the research question and in providing strategies to improve data collection (Table 3).
| Insight Offered | n (%) |
|---|---|
| Ideas for alternate methods | 92 (64%) |
| Strategies to improve data collection | 85 (59.4%) |
| New hypotheses generated | 84 (58.7%) |
| Ideas for next steps/future direction/studies | 83 (58%) |
| New outcomes suggested that should be considered | 69 (48%) |
| Clarification of the research questions | 61 (43%) |
| Input about what is most novel/emnteresting about the work | 60 (42%) |
| Guidance about the real implications of the work | 59 (41%) |
| Suggestions for alternate analyses or analytical strategies | 51 (36%) |
The free text comments primarily addressed how the presenters' research ideas were helped by the session:
There were great ideas for improvementincluding practical approaches for recruitment.
The session made me think of the daily routine things that we do that could be studied.
There were some great ideas to help Dr. A make the study more simple, doable, and practical. There were also some good ideas regarding potential sources of funding.
Academic Success
Of the 15 projects, 6 have been published in peer‐reviewed journals as first‐ or senior‐authored publications.510 Of these, 3 were presented at national meetings prior to publication. Four additional projects have been presented at a national society's annual meeting, all of which are being prepared for publication. Of the remaining 5 presentations, 4 were terminated because of the low likelihood of academic success. The remaining project is ongoing.
Comparatively, scholarly output in the prior year by the 24 physicians in the hospitalist group was 4 first‐ or senior‐authored publications in peer‐reviewed journals and 3 presentations at national meetings.
Discussion
In this article, we report our experience with the RIP conference. The sessions were perceived to be intellectually stimulating and supportive, whereas the discussions proved helpful in advancing project ideas. Ample discussion time and good attendance were thought to be critical to the success.
To our knowledge, this is the first article gathering feedback from attendees and presenters at a RIP conference and to track academic outcomes. Several types of meetings have been established within faculty and trainee groups to support and encourage scholarly activities.11, 12 The benefits of peer collaboration and peer mentoring have been described in the literature.13, 14 For example, Edwards described the success of shortstop meetings among small groups of faculty members every 4‐6 weeks in which discussions of research projects and mutual feedback would occur.15 Santucci described peer‐mentored research development meetings, with increased research productivity.12
Mentoring is critically important for academic success in medicine.1619 When divisions have limited senior mentors available, peer mentoring has proven to be indispensable as a mechanism to support faculty members.2022 The RIP conference provided a forum for peer mentoring and provided a partial solution to the limited resource of experienced research mentors in the division. The RIP sessions appear to have helped to bring the majority of presented ideas to academic fruition. Perhaps even more important, the sessions were able to terminate studies judged to have low academic promise before the faculty had invested significant time.
Several limitations of our study should be considered. First, this study involved a research‐in‐progress conference coordinated for a group of hospitalist physicians at 1 institution, and the results may not be generalizable. Second, although attendance was good at each conference, some faculty members did not come to many sessions. It is possible that those not attending may have rated the sessions differently. Session evaluations were anonymous, and we do not know whether specific attendees rated all sessions highly, thereby resulting in some degree of clustering. Third, this study did not compare the effectiveness of the RIP conference with other peer‐mentorship models. Finally, our study was uncontrolled. Although it would not be possible to restrict specific faculty from presenting at or attending the RIP conference, we intend to more carefully collect attendance data to see whether there might be a dose‐response effect with respect to participation in this conference and academic success.
In conclusion, our RIP conference was perceived as valuable by our group and was associated with academic success. In our division, the RIP conference serves as a way to operationalize peer mentoring. Our findings may help other groups to refine either the focus or format of their RIP sessions and those wishing to initiate such a conference.
- ,,, et al.Junior faculty members' mentoring relationships and their professional development in US medical schools.Acad Med.1998;73:318–323.
- ,.Mentors, Advisors and Role Models in Graduate and Professional Education.Washington, DC:Association of Academic Health Centers;1996.
- ,.Characteristics of the successful researcher and implications for faculty development.J Med Educ.1986;61:22–31.
- ,,.Curriculum Development for Medical Education: A Six‐Step Approach.2nd ed.Baltimore, MD:The Johns Hopkins University Press;2009.
- ,,, et al.Characteristics of hospitalists and hospitalist programs in the United States and Canada.J Clin Outcomes Meas.2009;16:69–74
- ,,, et al.Procedures performed by hospitalist and non‐hospitalist general internists.J Gen Intern Med.2010;25:448–452.
- ,,, et al.Intravenous immunoglobulin for the treatment of severe Clostridium difficile colitis: an observational study and review of the literature [review].J Hosp Med.2010;5:E1–E9.
- ,,, et al.Active bed management by hospitalists and emergency department throughput.Ann Intern Med.2008;149:804–811.
- ,,, et al.Transitioning to breast cancer survivorship: perspectives of patients, cancer specialists, and primary care providers.J Gen Intern Med.2009;24(Suppl 2):S459–S466.
- ,,, et al.Utilizing audit and feedback to improve hospitalists' performance in tobacco dependence counseling.Nicotine Tob Res.2010;12:797–800.
- ,,, et al.An internal medicine interest group research program can improve scholarly productivity of medical students and foster mentoring relationships with internists.Teach Learn Med.2008;20:163–167.
- ,,, et al.Peer‐mentored research development meeting: a model for successful peer mentoring among junior level researchers.Acad Psychiatry.2008;32:493–497.
- ,,, et al.Mentoring junior faculty in geriatric oncology: report from the cancer and aging research group.J Clin Oncol.2008;26:3125–3127.
- ,,the Canadian Critical Care Trials Group.Investigator‐led clinical research consortia: the Canadian Critical Care Trials Group.Crit Care Med.2009;37(1):S165–S172.
- .“Short stops”: peer support of scholarly activity.Acad Med.2002;77:939.
- ,,,,,,.Mentorship in academic general internal medicine. Results of a survey of mentors.J Gen Intern Med.2005;20:1014–1018.
- ,,, et al.Making the most of mentors: a guide for mentees.Acad Med.2009;84:140–144.
- ,,.Mentoring in academic medicine: a systematic review.JAMA.2006;296:1103–1115.
- ,,, et al.Assessing the role of influential mentors in the research development of primary care fellows.Acad Med.2004;79:865–872.
- ,,.Peer group mentoring of junior faculty.Acad Psychiatry.2008;32:230–235.
- ,,,.Facilitated peer mentorship: a pilot program for academic advancement of female medical faculty.J Womens Health.2008;17:1009–1015.
- ,Mentoring faculty in academic medicine. A new paradigm?J Gen Intern Med.2005;20:866–870.
The research‐in‐progress (RIP) conference is commonplace in academia, but there are no studies that objectively characterize its value. Bringing faculty together away from revenue‐generating activities carries a significant cost. As such, measuring the success of such gatherings is necessary.
Mentors are an invaluable influence on the careers of junior faculty members, helping them to produce high‐quality research.13 Unfortunately, some divisions lack mentorship to support the academic needs of less experienced faculty.1 Peer mentorship may be a solution. RIP sessions represent an opportunity to intentionally formalize peer mentoring. Further, these sessions can facilitate collaborations as individuals become aware of colleagues' interests. The goal of this study was to assess the value of the research‐in‐progress conference initiated within the hospitalist division at our institution.
Methods
Study Design
This cohort study was conducted to evaluate the value of the RIP conference among hospitalists in our division and the academic outcomes of the projects.
Setting and Participants
The study took place at Johns Hopkins Bayview Medical Center (JHBMC), a 335‐bed university‐affiliated medical center in Baltimore, Maryland. The hospitalist division consists of faculty physicians, nurse practitioners, and physician assistants (20.06 FTE physicians and 7.41 FTE midlevel providers). Twelve (54%) of our faculty members are female, and the mean age of providers is 35.7 years. The providers have been practicing hospitalist medicine for 3.0 years on average; 2 (9%) are clinical associates, 16 (73%) are instructors, and 3 (14%) are assistant professors.
All faculty members presenting at the RIP session were members of the division. A senior faculty member (a professor in the Division of General Internal Medicine) helps to coordinate the conference. The group's research assistant was present at the sessions and was charged with data collection and collation.
The Johns Hopkins University institutional review board approved the study.
The Research in Progress Conference
During the 2009 academic year, our division held 15 RIP sessions. At each session, 1 faculty member presented a research proposal. The goal of each session was to provide a forum where faculty members could share their research ideas (specific aims, hypotheses, planned design, outcome measures, analytic plans, and preliminary results [if applicable]) in order to receive feedback. The senior faculty member met with the presenter prior to each session in order to: (1) ensure that half the RIP time was reserved for discussion and (2) review the presenter's goals so these would be made explicit to peers. The coordinator of the RIP conference facilitated the discussion, solicited input from all attendees, and encouraged constructive criticism.
Evaluation, Data Collection, and Analysis
At the end of each session, attendees (who were exclusively members of the hospitalist division) were asked to complete an anonymous survey. The 1‐page instrument was designed (1) with input from curriculum development experts4 and (2) after a review of the literature about RIP conferences. These steps conferred content validity to the instrument, which assessed perceptions about the session's quality and what was learned. Five‐point Likert scales were used to characterize the conference's success in several areas, including being intellectually/professionally stimulating and keeping them apprised of their colleagues' interests. The survey also assessed the participatory nature of the conference (balance of presentation vs discussion), its climate (extremely critical vs extremely supportive), and how the conference assisted the presenter. The presenters completed a distinct survey related to how helpful the conference was in improving/enhancing their projects. A final open‐ended section invited additional comments. The instrument was piloted and iteratively revised before its use in this study.
For the projects presented, we assessed the percentage that resulted in a peer‐reviewed publication or a presentation at a national meeting.
Results
The mean number of attendees at the RIP sessions was 9.6 persons. A total of 143 evaluations were completed. All 15 presenters (100%) completed their assessments. The research ideas presented spanned a breadth of topics in clinical research, quality improvement, policy, and professional development (Table 1).
| Session | Date | Presenter | Topic | Evaluations Completed |
|---|---|---|---|---|
| 1 | 7/2008 | Dr. CS | Hospital medicine in Canada versus the United States | 7 |
| 2 | 7/2008 | Dr. RT | Procedures by hospitalists | 9 |
| 3 | 8/2008 | Dr. MA | Clostridium difficile treatment in the hospital | 11 |
| 4 | 8/2008 | Dr. EH | Active bed management | 6 |
| 5 | 9/2008 | Dr. AS | Medication reconciliation for geriatric inpatients | 10 |
| 6 | 9/2008 | Dr. DT | Time‐motion study of hospitalists | 10 |
| 7 | 10/2008 | Dr. KV | e‐Triage pilot | 16 |
| 8 | 11/2008 | Dr. EH | Assessing clinical performance of hospitalists | 7 |
| 9 | 12/2008 | Dr. SC | Trends and implications of hospitalists' morale | 8 |
| 10 | 1/2009 | Dr. TB | Lessons learned: tracking urinary catheter use at Bayview | 11 |
| 11 | 2/2009 | Dr. FK | Utilizing audit and feedback to improve performance in tobacco dependence counseling | 12 |
| 12 | 3/2009 | Dr. MK | Survivorship care plans | 7 |
| 13 | 4/2009 | Dr. DK | Outpatient provider preference for discharge summary format/style/length | 7 |
| 14 | 5/2009 | Dr. RW | Comparing preoperative consults done by hospitalists and cardiologists | 11 |
| 15 | 6/2009 | Dr. AK | Development of Web‐based messaging tool for providers | 12 |
Presenter Perspective
All 15 presenters (100%) felt a lot or tremendously supported during their sessions. Thirteen physicians (86%) believed that the sessions were a lot or tremendously helpful in advancing their projects. The presenters believed that the guidance and discussions related to their research ideas, aims, hypotheses, and plans were most helpful for advancing their projects (Table 2).
| Not at All, n (%) | A Little, n (%) | Some, n (%) | A Lot, n (%) | Tremendously, n (%) | |
|---|---|---|---|---|---|
| General questions: | |||||
| Intellectually/professionally stimulating | 0 (0) | 0 (0) | 0 (0) | 5 (33) | 10 (66) |
| Feeling supported by your colleagues in your scholarly pursuits | 0 (0) | 0 (0) | 0 (0) | 4 (27) | 11 (73) |
| Session helpful in the following areas: | |||||
| Advancing your project | 0 (0) | 0 (0) | 2 (13) | 5 (33) | 8 (53) |
| Generated new hypotheses | 1 (6) | 3 (20) | 5 (33) | 5 (33) | 1 (6) |
| Clarification of research questions | 0 (0) | 2 (13) | 4 (27) | 7 (47) | 2 (13) |
| Ideas for alternate methods | 1 (6) | 1 (6) | 2 (13) | 7 (47) | 4 (27) |
| New outcomes suggested | 1 (6) | 2 (13) | 2 (13) | 5 (33) | 5 (33) |
| Strategies to improve or enhance data collection | 0 (0) | 2 (13) | 0 (0) | 8 (53) | 5 (33) |
| Suggestions for alternate analyses or analytical strategies | 1 (1) | 1 (6) | 4 (27) | 5 (33) | 4 (27) |
| Input into what is most novel/emnteresting about this work | 0 (0) | 2 (13) | 3 (20) | 6 (40) | 4 (27) |
| Guidance about the implications of the work | 1 (6) | 2 (13) | 1 (6) | 7 (47) | 4 (27) |
| Ideas about next steps or future direction/studies | 0 (0) | 0 (0) | 3 (21) | 8 (57) | 3 (21) |
Examples of the written comments are:
I was overwhelmed by how engaged people were in my project.
The process of preparing for the session and then the discussion both helped my thinking. Colleagues were very supportive.
I am so glad I heard these comments and received this feedback now, rather than from peer reviewers selected by a journal to review my study. It would have been a much more difficult situation to fix at that later time.
Attendee Perspective
The majority of attendees (123 of 143, 86%) found the sessions to be a lot or extremely stimulating, and almost all (96%) were a lot or extremely satisfied with how the RIP sessions kept them abreast of their colleagues' academic interests. In addition, 92% judged the session's climate to be a lot or extremely supportive, and 88% deemed the balance of presentation to discussion to be just right. Attendees believed that they were most helpful to the presenter in terms of conceiving ideas for alternative methods to be used to answer the research question and in providing strategies to improve data collection (Table 3).
| Insight Offered | n (%) |
|---|---|
| Ideas for alternate methods | 92 (64%) |
| Strategies to improve data collection | 85 (59.4%) |
| New hypotheses generated | 84 (58.7%) |
| Ideas for next steps/future direction/studies | 83 (58%) |
| New outcomes suggested that should be considered | 69 (48%) |
| Clarification of the research questions | 61 (43%) |
| Input about what is most novel/emnteresting about the work | 60 (42%) |
| Guidance about the real implications of the work | 59 (41%) |
| Suggestions for alternate analyses or analytical strategies | 51 (36%) |
The free text comments primarily addressed how the presenters' research ideas were helped by the session:
There were great ideas for improvementincluding practical approaches for recruitment.
The session made me think of the daily routine things that we do that could be studied.
There were some great ideas to help Dr. A make the study more simple, doable, and practical. There were also some good ideas regarding potential sources of funding.
Academic Success
Of the 15 projects, 6 have been published in peer‐reviewed journals as first‐ or senior‐authored publications.510 Of these, 3 were presented at national meetings prior to publication. Four additional projects have been presented at a national society's annual meeting, all of which are being prepared for publication. Of the remaining 5 presentations, 4 were terminated because of the low likelihood of academic success. The remaining project is ongoing.
Comparatively, scholarly output in the prior year by the 24 physicians in the hospitalist group was 4 first‐ or senior‐authored publications in peer‐reviewed journals and 3 presentations at national meetings.
Discussion
In this article, we report our experience with the RIP conference. The sessions were perceived to be intellectually stimulating and supportive, whereas the discussions proved helpful in advancing project ideas. Ample discussion time and good attendance were thought to be critical to the success.
To our knowledge, this is the first article gathering feedback from attendees and presenters at a RIP conference and to track academic outcomes. Several types of meetings have been established within faculty and trainee groups to support and encourage scholarly activities.11, 12 The benefits of peer collaboration and peer mentoring have been described in the literature.13, 14 For example, Edwards described the success of shortstop meetings among small groups of faculty members every 4‐6 weeks in which discussions of research projects and mutual feedback would occur.15 Santucci described peer‐mentored research development meetings, with increased research productivity.12
Mentoring is critically important for academic success in medicine.1619 When divisions have limited senior mentors available, peer mentoring has proven to be indispensable as a mechanism to support faculty members.2022 The RIP conference provided a forum for peer mentoring and provided a partial solution to the limited resource of experienced research mentors in the division. The RIP sessions appear to have helped to bring the majority of presented ideas to academic fruition. Perhaps even more important, the sessions were able to terminate studies judged to have low academic promise before the faculty had invested significant time.
Several limitations of our study should be considered. First, this study involved a research‐in‐progress conference coordinated for a group of hospitalist physicians at 1 institution, and the results may not be generalizable. Second, although attendance was good at each conference, some faculty members did not come to many sessions. It is possible that those not attending may have rated the sessions differently. Session evaluations were anonymous, and we do not know whether specific attendees rated all sessions highly, thereby resulting in some degree of clustering. Third, this study did not compare the effectiveness of the RIP conference with other peer‐mentorship models. Finally, our study was uncontrolled. Although it would not be possible to restrict specific faculty from presenting at or attending the RIP conference, we intend to more carefully collect attendance data to see whether there might be a dose‐response effect with respect to participation in this conference and academic success.
In conclusion, our RIP conference was perceived as valuable by our group and was associated with academic success. In our division, the RIP conference serves as a way to operationalize peer mentoring. Our findings may help other groups to refine either the focus or format of their RIP sessions and those wishing to initiate such a conference.
The research‐in‐progress (RIP) conference is commonplace in academia, but there are no studies that objectively characterize its value. Bringing faculty together away from revenue‐generating activities carries a significant cost. As such, measuring the success of such gatherings is necessary.
Mentors are an invaluable influence on the careers of junior faculty members, helping them to produce high‐quality research.13 Unfortunately, some divisions lack mentorship to support the academic needs of less experienced faculty.1 Peer mentorship may be a solution. RIP sessions represent an opportunity to intentionally formalize peer mentoring. Further, these sessions can facilitate collaborations as individuals become aware of colleagues' interests. The goal of this study was to assess the value of the research‐in‐progress conference initiated within the hospitalist division at our institution.
Methods
Study Design
This cohort study was conducted to evaluate the value of the RIP conference among hospitalists in our division and the academic outcomes of the projects.
Setting and Participants
The study took place at Johns Hopkins Bayview Medical Center (JHBMC), a 335‐bed university‐affiliated medical center in Baltimore, Maryland. The hospitalist division consists of faculty physicians, nurse practitioners, and physician assistants (20.06 FTE physicians and 7.41 FTE midlevel providers). Twelve (54%) of our faculty members are female, and the mean age of providers is 35.7 years. The providers have been practicing hospitalist medicine for 3.0 years on average; 2 (9%) are clinical associates, 16 (73%) are instructors, and 3 (14%) are assistant professors.
All faculty members presenting at the RIP session were members of the division. A senior faculty member (a professor in the Division of General Internal Medicine) helps to coordinate the conference. The group's research assistant was present at the sessions and was charged with data collection and collation.
The Johns Hopkins University institutional review board approved the study.
The Research in Progress Conference
During the 2009 academic year, our division held 15 RIP sessions. At each session, 1 faculty member presented a research proposal. The goal of each session was to provide a forum where faculty members could share their research ideas (specific aims, hypotheses, planned design, outcome measures, analytic plans, and preliminary results [if applicable]) in order to receive feedback. The senior faculty member met with the presenter prior to each session in order to: (1) ensure that half the RIP time was reserved for discussion and (2) review the presenter's goals so these would be made explicit to peers. The coordinator of the RIP conference facilitated the discussion, solicited input from all attendees, and encouraged constructive criticism.
Evaluation, Data Collection, and Analysis
At the end of each session, attendees (who were exclusively members of the hospitalist division) were asked to complete an anonymous survey. The 1‐page instrument was designed (1) with input from curriculum development experts4 and (2) after a review of the literature about RIP conferences. These steps conferred content validity to the instrument, which assessed perceptions about the session's quality and what was learned. Five‐point Likert scales were used to characterize the conference's success in several areas, including being intellectually/professionally stimulating and keeping them apprised of their colleagues' interests. The survey also assessed the participatory nature of the conference (balance of presentation vs discussion), its climate (extremely critical vs extremely supportive), and how the conference assisted the presenter. The presenters completed a distinct survey related to how helpful the conference was in improving/enhancing their projects. A final open‐ended section invited additional comments. The instrument was piloted and iteratively revised before its use in this study.
For the projects presented, we assessed the percentage that resulted in a peer‐reviewed publication or a presentation at a national meeting.
Results
The mean number of attendees at the RIP sessions was 9.6 persons. A total of 143 evaluations were completed. All 15 presenters (100%) completed their assessments. The research ideas presented spanned a breadth of topics in clinical research, quality improvement, policy, and professional development (Table 1).
| Session | Date | Presenter | Topic | Evaluations Completed |
|---|---|---|---|---|
| 1 | 7/2008 | Dr. CS | Hospital medicine in Canada versus the United States | 7 |
| 2 | 7/2008 | Dr. RT | Procedures by hospitalists | 9 |
| 3 | 8/2008 | Dr. MA | Clostridium difficile treatment in the hospital | 11 |
| 4 | 8/2008 | Dr. EH | Active bed management | 6 |
| 5 | 9/2008 | Dr. AS | Medication reconciliation for geriatric inpatients | 10 |
| 6 | 9/2008 | Dr. DT | Time‐motion study of hospitalists | 10 |
| 7 | 10/2008 | Dr. KV | e‐Triage pilot | 16 |
| 8 | 11/2008 | Dr. EH | Assessing clinical performance of hospitalists | 7 |
| 9 | 12/2008 | Dr. SC | Trends and implications of hospitalists' morale | 8 |
| 10 | 1/2009 | Dr. TB | Lessons learned: tracking urinary catheter use at Bayview | 11 |
| 11 | 2/2009 | Dr. FK | Utilizing audit and feedback to improve performance in tobacco dependence counseling | 12 |
| 12 | 3/2009 | Dr. MK | Survivorship care plans | 7 |
| 13 | 4/2009 | Dr. DK | Outpatient provider preference for discharge summary format/style/length | 7 |
| 14 | 5/2009 | Dr. RW | Comparing preoperative consults done by hospitalists and cardiologists | 11 |
| 15 | 6/2009 | Dr. AK | Development of Web‐based messaging tool for providers | 12 |
Presenter Perspective
All 15 presenters (100%) felt a lot or tremendously supported during their sessions. Thirteen physicians (86%) believed that the sessions were a lot or tremendously helpful in advancing their projects. The presenters believed that the guidance and discussions related to their research ideas, aims, hypotheses, and plans were most helpful for advancing their projects (Table 2).
| Not at All, n (%) | A Little, n (%) | Some, n (%) | A Lot, n (%) | Tremendously, n (%) | |
|---|---|---|---|---|---|
| General questions: | |||||
| Intellectually/professionally stimulating | 0 (0) | 0 (0) | 0 (0) | 5 (33) | 10 (66) |
| Feeling supported by your colleagues in your scholarly pursuits | 0 (0) | 0 (0) | 0 (0) | 4 (27) | 11 (73) |
| Session helpful in the following areas: | |||||
| Advancing your project | 0 (0) | 0 (0) | 2 (13) | 5 (33) | 8 (53) |
| Generated new hypotheses | 1 (6) | 3 (20) | 5 (33) | 5 (33) | 1 (6) |
| Clarification of research questions | 0 (0) | 2 (13) | 4 (27) | 7 (47) | 2 (13) |
| Ideas for alternate methods | 1 (6) | 1 (6) | 2 (13) | 7 (47) | 4 (27) |
| New outcomes suggested | 1 (6) | 2 (13) | 2 (13) | 5 (33) | 5 (33) |
| Strategies to improve or enhance data collection | 0 (0) | 2 (13) | 0 (0) | 8 (53) | 5 (33) |
| Suggestions for alternate analyses or analytical strategies | 1 (1) | 1 (6) | 4 (27) | 5 (33) | 4 (27) |
| Input into what is most novel/emnteresting about this work | 0 (0) | 2 (13) | 3 (20) | 6 (40) | 4 (27) |
| Guidance about the implications of the work | 1 (6) | 2 (13) | 1 (6) | 7 (47) | 4 (27) |
| Ideas about next steps or future direction/studies | 0 (0) | 0 (0) | 3 (21) | 8 (57) | 3 (21) |
Examples of the written comments are:
I was overwhelmed by how engaged people were in my project.
The process of preparing for the session and then the discussion both helped my thinking. Colleagues were very supportive.
I am so glad I heard these comments and received this feedback now, rather than from peer reviewers selected by a journal to review my study. It would have been a much more difficult situation to fix at that later time.
Attendee Perspective
The majority of attendees (123 of 143, 86%) found the sessions to be a lot or extremely stimulating, and almost all (96%) were a lot or extremely satisfied with how the RIP sessions kept them abreast of their colleagues' academic interests. In addition, 92% judged the session's climate to be a lot or extremely supportive, and 88% deemed the balance of presentation to discussion to be just right. Attendees believed that they were most helpful to the presenter in terms of conceiving ideas for alternative methods to be used to answer the research question and in providing strategies to improve data collection (Table 3).
| Insight Offered | n (%) |
|---|---|
| Ideas for alternate methods | 92 (64%) |
| Strategies to improve data collection | 85 (59.4%) |
| New hypotheses generated | 84 (58.7%) |
| Ideas for next steps/future direction/studies | 83 (58%) |
| New outcomes suggested that should be considered | 69 (48%) |
| Clarification of the research questions | 61 (43%) |
| Input about what is most novel/emnteresting about the work | 60 (42%) |
| Guidance about the real implications of the work | 59 (41%) |
| Suggestions for alternate analyses or analytical strategies | 51 (36%) |
The free text comments primarily addressed how the presenters' research ideas were helped by the session:
There were great ideas for improvementincluding practical approaches for recruitment.
The session made me think of the daily routine things that we do that could be studied.
There were some great ideas to help Dr. A make the study more simple, doable, and practical. There were also some good ideas regarding potential sources of funding.
Academic Success
Of the 15 projects, 6 have been published in peer‐reviewed journals as first‐ or senior‐authored publications.510 Of these, 3 were presented at national meetings prior to publication. Four additional projects have been presented at a national society's annual meeting, all of which are being prepared for publication. Of the remaining 5 presentations, 4 were terminated because of the low likelihood of academic success. The remaining project is ongoing.
Comparatively, scholarly output in the prior year by the 24 physicians in the hospitalist group was 4 first‐ or senior‐authored publications in peer‐reviewed journals and 3 presentations at national meetings.
Discussion
In this article, we report our experience with the RIP conference. The sessions were perceived to be intellectually stimulating and supportive, whereas the discussions proved helpful in advancing project ideas. Ample discussion time and good attendance were thought to be critical to the success.
To our knowledge, this is the first article gathering feedback from attendees and presenters at a RIP conference and to track academic outcomes. Several types of meetings have been established within faculty and trainee groups to support and encourage scholarly activities.11, 12 The benefits of peer collaboration and peer mentoring have been described in the literature.13, 14 For example, Edwards described the success of shortstop meetings among small groups of faculty members every 4‐6 weeks in which discussions of research projects and mutual feedback would occur.15 Santucci described peer‐mentored research development meetings, with increased research productivity.12
Mentoring is critically important for academic success in medicine.1619 When divisions have limited senior mentors available, peer mentoring has proven to be indispensable as a mechanism to support faculty members.2022 The RIP conference provided a forum for peer mentoring and provided a partial solution to the limited resource of experienced research mentors in the division. The RIP sessions appear to have helped to bring the majority of presented ideas to academic fruition. Perhaps even more important, the sessions were able to terminate studies judged to have low academic promise before the faculty had invested significant time.
Several limitations of our study should be considered. First, this study involved a research‐in‐progress conference coordinated for a group of hospitalist physicians at 1 institution, and the results may not be generalizable. Second, although attendance was good at each conference, some faculty members did not come to many sessions. It is possible that those not attending may have rated the sessions differently. Session evaluations were anonymous, and we do not know whether specific attendees rated all sessions highly, thereby resulting in some degree of clustering. Third, this study did not compare the effectiveness of the RIP conference with other peer‐mentorship models. Finally, our study was uncontrolled. Although it would not be possible to restrict specific faculty from presenting at or attending the RIP conference, we intend to more carefully collect attendance data to see whether there might be a dose‐response effect with respect to participation in this conference and academic success.
In conclusion, our RIP conference was perceived as valuable by our group and was associated with academic success. In our division, the RIP conference serves as a way to operationalize peer mentoring. Our findings may help other groups to refine either the focus or format of their RIP sessions and those wishing to initiate such a conference.
- ,,, et al.Junior faculty members' mentoring relationships and their professional development in US medical schools.Acad Med.1998;73:318–323.
- ,.Mentors, Advisors and Role Models in Graduate and Professional Education.Washington, DC:Association of Academic Health Centers;1996.
- ,.Characteristics of the successful researcher and implications for faculty development.J Med Educ.1986;61:22–31.
- ,,.Curriculum Development for Medical Education: A Six‐Step Approach.2nd ed.Baltimore, MD:The Johns Hopkins University Press;2009.
- ,,, et al.Characteristics of hospitalists and hospitalist programs in the United States and Canada.J Clin Outcomes Meas.2009;16:69–74
- ,,, et al.Procedures performed by hospitalist and non‐hospitalist general internists.J Gen Intern Med.2010;25:448–452.
- ,,, et al.Intravenous immunoglobulin for the treatment of severe Clostridium difficile colitis: an observational study and review of the literature [review].J Hosp Med.2010;5:E1–E9.
- ,,, et al.Active bed management by hospitalists and emergency department throughput.Ann Intern Med.2008;149:804–811.
- ,,, et al.Transitioning to breast cancer survivorship: perspectives of patients, cancer specialists, and primary care providers.J Gen Intern Med.2009;24(Suppl 2):S459–S466.
- ,,, et al.Utilizing audit and feedback to improve hospitalists' performance in tobacco dependence counseling.Nicotine Tob Res.2010;12:797–800.
- ,,, et al.An internal medicine interest group research program can improve scholarly productivity of medical students and foster mentoring relationships with internists.Teach Learn Med.2008;20:163–167.
- ,,, et al.Peer‐mentored research development meeting: a model for successful peer mentoring among junior level researchers.Acad Psychiatry.2008;32:493–497.
- ,,, et al.Mentoring junior faculty in geriatric oncology: report from the cancer and aging research group.J Clin Oncol.2008;26:3125–3127.
- ,,the Canadian Critical Care Trials Group.Investigator‐led clinical research consortia: the Canadian Critical Care Trials Group.Crit Care Med.2009;37(1):S165–S172.
- .“Short stops”: peer support of scholarly activity.Acad Med.2002;77:939.
- ,,,,,,.Mentorship in academic general internal medicine. Results of a survey of mentors.J Gen Intern Med.2005;20:1014–1018.
- ,,, et al.Making the most of mentors: a guide for mentees.Acad Med.2009;84:140–144.
- ,,.Mentoring in academic medicine: a systematic review.JAMA.2006;296:1103–1115.
- ,,, et al.Assessing the role of influential mentors in the research development of primary care fellows.Acad Med.2004;79:865–872.
- ,,.Peer group mentoring of junior faculty.Acad Psychiatry.2008;32:230–235.
- ,,,.Facilitated peer mentorship: a pilot program for academic advancement of female medical faculty.J Womens Health.2008;17:1009–1015.
- ,Mentoring faculty in academic medicine. A new paradigm?J Gen Intern Med.2005;20:866–870.
- ,,, et al.Junior faculty members' mentoring relationships and their professional development in US medical schools.Acad Med.1998;73:318–323.
- ,.Mentors, Advisors and Role Models in Graduate and Professional Education.Washington, DC:Association of Academic Health Centers;1996.
- ,.Characteristics of the successful researcher and implications for faculty development.J Med Educ.1986;61:22–31.
- ,,.Curriculum Development for Medical Education: A Six‐Step Approach.2nd ed.Baltimore, MD:The Johns Hopkins University Press;2009.
- ,,, et al.Characteristics of hospitalists and hospitalist programs in the United States and Canada.J Clin Outcomes Meas.2009;16:69–74
- ,,, et al.Procedures performed by hospitalist and non‐hospitalist general internists.J Gen Intern Med.2010;25:448–452.
- ,,, et al.Intravenous immunoglobulin for the treatment of severe Clostridium difficile colitis: an observational study and review of the literature [review].J Hosp Med.2010;5:E1–E9.
- ,,, et al.Active bed management by hospitalists and emergency department throughput.Ann Intern Med.2008;149:804–811.
- ,,, et al.Transitioning to breast cancer survivorship: perspectives of patients, cancer specialists, and primary care providers.J Gen Intern Med.2009;24(Suppl 2):S459–S466.
- ,,, et al.Utilizing audit and feedback to improve hospitalists' performance in tobacco dependence counseling.Nicotine Tob Res.2010;12:797–800.
- ,,, et al.An internal medicine interest group research program can improve scholarly productivity of medical students and foster mentoring relationships with internists.Teach Learn Med.2008;20:163–167.
- ,,, et al.Peer‐mentored research development meeting: a model for successful peer mentoring among junior level researchers.Acad Psychiatry.2008;32:493–497.
- ,,, et al.Mentoring junior faculty in geriatric oncology: report from the cancer and aging research group.J Clin Oncol.2008;26:3125–3127.
- ,,the Canadian Critical Care Trials Group.Investigator‐led clinical research consortia: the Canadian Critical Care Trials Group.Crit Care Med.2009;37(1):S165–S172.
- .“Short stops”: peer support of scholarly activity.Acad Med.2002;77:939.
- ,,,,,,.Mentorship in academic general internal medicine. Results of a survey of mentors.J Gen Intern Med.2005;20:1014–1018.
- ,,, et al.Making the most of mentors: a guide for mentees.Acad Med.2009;84:140–144.
- ,,.Mentoring in academic medicine: a systematic review.JAMA.2006;296:1103–1115.
- ,,, et al.Assessing the role of influential mentors in the research development of primary care fellows.Acad Med.2004;79:865–872.
- ,,.Peer group mentoring of junior faculty.Acad Psychiatry.2008;32:230–235.
- ,,,.Facilitated peer mentorship: a pilot program for academic advancement of female medical faculty.J Womens Health.2008;17:1009–1015.
- ,Mentoring faculty in academic medicine. A new paradigm?J Gen Intern Med.2005;20:866–870.
Copyright © 2011 Society of Hospital Medicine
Off‐hours care: not so off
The word housestaff came from an era when you moved into the hospital (house) for your internship (staff). In those days, call every other night meant you missed half the learning opportunities. In those days, supervision was as remote as it could get. My, have things changed.
In 2003, the Accreditation Council for Graduate Medical Education (ACGME) implemented duty hour standards that called for restricting resident duty hours to no more than 80 hours per week, and no more than 24 continuous hours with an additional 6 hours for transfer of care.1 The expectation was that limiting work hours would have positive effects on outcomes, such as resident fatigue, the educational experience, and patient safety. A recent systematic review commissioned by the ACGME reviewed three decades worth of research on duty hours and patient safety. It reviewed laboratory studies of resident performance on clinical simulations, and tests of cognitive and fine motor skills under conditions of sleep deprivation. The results of these studies imply that limiting residents' duty hours should have positive effects on patient safety. However, that assumption has not been borne out. Paradoxically, examination of studies of alternative duty hour schedules, as well as the follow‐up studies on the impact of the New York duty hour rules and ACGME duty hour regulations have documented few measurable effects on patient safety.2 Thus, the simplistic view that fewer hours worked equals better patient care deserves a second‐look. In fact, a cause‐and‐effect relationship between resident fatigue and medical errors has yet to be proven.
The hospitalist movement was in full swing by 2003 and had already transformed the care of hospitalized patients. The early studies of hospitalists focused on cost and comparing outcomes with those of other providers. As many studies showed the economic benefit of hospitalists, residency programs began redesigning their inpatient services with hospitalists at the helm. Institutions sponsoring graduate medical education programs saw hospitalists as a necessary resource to enable and maintain compliance with duty hour reform. Some institutions created hospitalist‐only services with no housestaff (nonteaching services). Others created hospitalist teaching services that used indirect supervision at night from a hospitalist on‐call from home, while others had on‐site hospitalists at night that provided direct supervision of housestaff. It is clear that hospitalists are a versatile and valuable resource.
In this issue of the Journal of Hospital Medicine, Khanna and colleagues3 describe a retrospective medical record review at a large, urban academic hospital that evaluates the potential association between night admissions by night‐shiftbased hospitalists and a resident night float system, and hospitalization‐relevant outcomes. The outcomes included length of stay, hospital charges, Intensive Care Unit (ICU) transfers, Emergency Department visits within 30 days of discharge, 30‐day readmission rates, and poor outcomes within the first 24 hours of admission. Their results showed that night or weekend admission was not associated with worse hospitalization‐relevant outcomes. The extension of on‐site faculty supervision to nights, as well as awards for nursing excellence, likely had a strong positive impact on these outcomes. In some cases, night or weekend admission was associated with better outcomes, particularly in terms of ICU transfer during hospitalization and hospital charges. Maybe off‐hours care is not so off after all.
I am not sure where we are going, but the duty hour standards have been revised again. The new duty hour standards limit continuous duty hours for 1st‐year residents to 16, and all others to 24. Upper‐level residents can stay an additional 4 hours for transfer of care.4 Functionally, this proposed standard eliminates overnight call for 1st‐year residents. Although I would never advocate returning to the days of yore where autonomy and independence were the rule, I mourn the loss of extended shifts for 1st‐year residents. The loss of the extended shift will come at the expense of overnight call in many programs. I worry about the loss of some unique teaching moments for all residents, and the increased number of transitions in a patients' care. The hospital is a different place at night. It is less chaotic with fewer visitors, and there are less interruptions in patient care. There is time for teaching and learning because the work intensity is lower. The bedside teaching among senior and junior residents, during an acute patient episode in the middle of the night, is an experience that many of us would recount as an invaluable part of our training. It is also a time when on‐site faculty supervision and nurse staffing ratios are typically reduced. In other words, the entire system of care is different at night. This dichotomy in day versus night medicine has spurred research into whether patient outcomes differed between the two. Various investigators have looked at the differences in care, and there is some data to support that patient care outcomes are worse at night than during the day.58
The results from Khanna and colleagues3 are welcome news, since graduate medical education is undergoing a paradigm shift with further reductions in duty hours. Hospital‐based medicine is a 24‐hour operation, 7 days a week. We don't get to pick when patients are admitted or need care. In fact, this study demonstrates that 58% of the admissions came after 7 PM. It turns out that this pattern of admissions is not atypical.9 In light of the new standards, programs are left deciding where to deploy their 1st‐year resident resources. There are essentially two types of models to provide patient care off‐hours in the absence of an extended shift option: night float and night medicine. Night float implies that a trainee, usually a resident, comes from a different site or rotation to provide nocturnal care of a group of patients. That resident may do one isolated night of coverage or several nights in a row. The resident may perform cross‐cover functions, admit patients, or do both. Night medicine refers to having teams of housestaff at night along with an educational program as part of a rotation. The latter implies that trainees will get the full educational experience whether working days or nights. If one designs a model of care that puts 1st‐year residents where the volume is, they would come in late and leave early in the morning the next day. Both create more handoffs in patient care, which critics of these models have said lead to worse patient outcomes. This research challenges such earlier work and should make us all feel more comfortable that care at night does not need to be different if the proper systems are in place to manage it.
The graduate medical education community needs to take a step back and ask: Where are we going? As a long‐standing program director, I don't know anymore. As training has become competency‐based, and milestones are being developed for each level of training, we need to ask if reducing hours further is in the best interests of graduate medical education and, ultimately, the general public. I have long been interested in work intensity as a contributor to resident fatigue, burnout, and errors. A systematic review of resident workload and work activity looked at 21 studies examining resident time allocation and suggests that at least one‐third of resident time is spent on activities of limited educational value. Eight of the nine studies evaluating the impact of resident workload on patient care showed a negative impact on outcomes, including length of stay, mortality, patient satisfaction, medication errors, and lab utilization.10 If we are going to further restrict duty hours, then we need to transform the learning environment in a similar fashion. It does not seem logical to restrict duty hours further without a major redesign of the learning environment. I fear that all we have done is create more work compression because the system of care will be further strained in its weakest areas.
Hospitalists are uniquely poised to mitigate some of these concerns by being available for direct and indirect supervision, as well as being highly skilled at working in our complex systems. They are experts in systems‐based practice. The extension of direct faculty supervision to nights may be a major factor in eliminating the variances in care between nights and days. Not every program or institution will have the ability to employ night hospitalists, let alone have them in a supervisory and teaching role. Those that do can take great comfort in this research. Those that don't must challenge themselves to think about how to get there.
We all share the collective goal of producing the next generation of well‐trained physicians who will practice safe and effective care. As we begin to embrace competency‐based training, the prism through which we view patient safety must extend beyond duty hours and include learning environment reform. While the Khanna et al.3 study supports that off‐hours care is no longer off, we need to be careful that we don't drift too far towards shift work as the fundamental structure of medical training. In the beginning, residents lived in the hospital, thus the term. As we move through these stormy seas, we don't want to trade patient safety for transient trainees. There is a middle ground here that will be informed by future research. There is something to be said for living in your house.
- ACGME.Duty hour requirements. Available at: http://www.acgme.org/acWebsite/dutyHours/dh_ComProgrRequirmentsDutyHours0707.pdf. Accessed August 16,2010.
- ,,, et al.Systematic review of the literature on the impact of variation in residents' duty hour schedules on patient safety. Available at: http://acgme‐2010standards.org/pdf/Jefferson_Medical_College_Duty_Hours_Review.pdf. Accessed August 18,2010.
- ,,, et al.The association between night or weekend admission and hospitalization‐relevant patient outcomes.J Hosp Med2011;6:10–14.
- ACGME.Proposed duty hour standards. Available at: http://acgme‐2010standards.org/pdf/Proposed_Standards.pdf. Accessed August 16,2010.
- ,.Mortality among patients admitted to hospitals on weekends as compared with weekdays.N Engl J Med.2001;345(9):663–668.
- ,,, et al.Survival from in‐hospital cardiac arrest during nights and weekends.JAMA.2008;299(7):785–792.
- ,,,,.What effect does inpatient physician specialty and experience have on clinical outcomes and resource utilization on a general medicine service?J Gen Intern Med.2004;19:395–401.
- ,,,,,.Implementation of a voluntary hospitalist service at a community teaching hospital: improved clinical efficiency and patient outcomes.Ann Intern Med.2002;137:859–865.
- .Like night and day—shedding light on off‐hours care.N Engl J Med.2008;358(20):2091–2093.
- ,,.Systematic review of resident workload and work activity: impact on resident education and patient care. Available at: http://acgme‐2010standards.org/pdf/Resident_Duty_Hours_and_Related_Topics.pdf. Accessed on August 16,2010.
The word housestaff came from an era when you moved into the hospital (house) for your internship (staff). In those days, call every other night meant you missed half the learning opportunities. In those days, supervision was as remote as it could get. My, have things changed.
In 2003, the Accreditation Council for Graduate Medical Education (ACGME) implemented duty hour standards that called for restricting resident duty hours to no more than 80 hours per week, and no more than 24 continuous hours with an additional 6 hours for transfer of care.1 The expectation was that limiting work hours would have positive effects on outcomes, such as resident fatigue, the educational experience, and patient safety. A recent systematic review commissioned by the ACGME reviewed three decades worth of research on duty hours and patient safety. It reviewed laboratory studies of resident performance on clinical simulations, and tests of cognitive and fine motor skills under conditions of sleep deprivation. The results of these studies imply that limiting residents' duty hours should have positive effects on patient safety. However, that assumption has not been borne out. Paradoxically, examination of studies of alternative duty hour schedules, as well as the follow‐up studies on the impact of the New York duty hour rules and ACGME duty hour regulations have documented few measurable effects on patient safety.2 Thus, the simplistic view that fewer hours worked equals better patient care deserves a second‐look. In fact, a cause‐and‐effect relationship between resident fatigue and medical errors has yet to be proven.
The hospitalist movement was in full swing by 2003 and had already transformed the care of hospitalized patients. The early studies of hospitalists focused on cost and comparing outcomes with those of other providers. As many studies showed the economic benefit of hospitalists, residency programs began redesigning their inpatient services with hospitalists at the helm. Institutions sponsoring graduate medical education programs saw hospitalists as a necessary resource to enable and maintain compliance with duty hour reform. Some institutions created hospitalist‐only services with no housestaff (nonteaching services). Others created hospitalist teaching services that used indirect supervision at night from a hospitalist on‐call from home, while others had on‐site hospitalists at night that provided direct supervision of housestaff. It is clear that hospitalists are a versatile and valuable resource.
In this issue of the Journal of Hospital Medicine, Khanna and colleagues3 describe a retrospective medical record review at a large, urban academic hospital that evaluates the potential association between night admissions by night‐shiftbased hospitalists and a resident night float system, and hospitalization‐relevant outcomes. The outcomes included length of stay, hospital charges, Intensive Care Unit (ICU) transfers, Emergency Department visits within 30 days of discharge, 30‐day readmission rates, and poor outcomes within the first 24 hours of admission. Their results showed that night or weekend admission was not associated with worse hospitalization‐relevant outcomes. The extension of on‐site faculty supervision to nights, as well as awards for nursing excellence, likely had a strong positive impact on these outcomes. In some cases, night or weekend admission was associated with better outcomes, particularly in terms of ICU transfer during hospitalization and hospital charges. Maybe off‐hours care is not so off after all.
I am not sure where we are going, but the duty hour standards have been revised again. The new duty hour standards limit continuous duty hours for 1st‐year residents to 16, and all others to 24. Upper‐level residents can stay an additional 4 hours for transfer of care.4 Functionally, this proposed standard eliminates overnight call for 1st‐year residents. Although I would never advocate returning to the days of yore where autonomy and independence were the rule, I mourn the loss of extended shifts for 1st‐year residents. The loss of the extended shift will come at the expense of overnight call in many programs. I worry about the loss of some unique teaching moments for all residents, and the increased number of transitions in a patients' care. The hospital is a different place at night. It is less chaotic with fewer visitors, and there are less interruptions in patient care. There is time for teaching and learning because the work intensity is lower. The bedside teaching among senior and junior residents, during an acute patient episode in the middle of the night, is an experience that many of us would recount as an invaluable part of our training. It is also a time when on‐site faculty supervision and nurse staffing ratios are typically reduced. In other words, the entire system of care is different at night. This dichotomy in day versus night medicine has spurred research into whether patient outcomes differed between the two. Various investigators have looked at the differences in care, and there is some data to support that patient care outcomes are worse at night than during the day.58
The results from Khanna and colleagues3 are welcome news, since graduate medical education is undergoing a paradigm shift with further reductions in duty hours. Hospital‐based medicine is a 24‐hour operation, 7 days a week. We don't get to pick when patients are admitted or need care. In fact, this study demonstrates that 58% of the admissions came after 7 PM. It turns out that this pattern of admissions is not atypical.9 In light of the new standards, programs are left deciding where to deploy their 1st‐year resident resources. There are essentially two types of models to provide patient care off‐hours in the absence of an extended shift option: night float and night medicine. Night float implies that a trainee, usually a resident, comes from a different site or rotation to provide nocturnal care of a group of patients. That resident may do one isolated night of coverage or several nights in a row. The resident may perform cross‐cover functions, admit patients, or do both. Night medicine refers to having teams of housestaff at night along with an educational program as part of a rotation. The latter implies that trainees will get the full educational experience whether working days or nights. If one designs a model of care that puts 1st‐year residents where the volume is, they would come in late and leave early in the morning the next day. Both create more handoffs in patient care, which critics of these models have said lead to worse patient outcomes. This research challenges such earlier work and should make us all feel more comfortable that care at night does not need to be different if the proper systems are in place to manage it.
The graduate medical education community needs to take a step back and ask: Where are we going? As a long‐standing program director, I don't know anymore. As training has become competency‐based, and milestones are being developed for each level of training, we need to ask if reducing hours further is in the best interests of graduate medical education and, ultimately, the general public. I have long been interested in work intensity as a contributor to resident fatigue, burnout, and errors. A systematic review of resident workload and work activity looked at 21 studies examining resident time allocation and suggests that at least one‐third of resident time is spent on activities of limited educational value. Eight of the nine studies evaluating the impact of resident workload on patient care showed a negative impact on outcomes, including length of stay, mortality, patient satisfaction, medication errors, and lab utilization.10 If we are going to further restrict duty hours, then we need to transform the learning environment in a similar fashion. It does not seem logical to restrict duty hours further without a major redesign of the learning environment. I fear that all we have done is create more work compression because the system of care will be further strained in its weakest areas.
Hospitalists are uniquely poised to mitigate some of these concerns by being available for direct and indirect supervision, as well as being highly skilled at working in our complex systems. They are experts in systems‐based practice. The extension of direct faculty supervision to nights may be a major factor in eliminating the variances in care between nights and days. Not every program or institution will have the ability to employ night hospitalists, let alone have them in a supervisory and teaching role. Those that do can take great comfort in this research. Those that don't must challenge themselves to think about how to get there.
We all share the collective goal of producing the next generation of well‐trained physicians who will practice safe and effective care. As we begin to embrace competency‐based training, the prism through which we view patient safety must extend beyond duty hours and include learning environment reform. While the Khanna et al.3 study supports that off‐hours care is no longer off, we need to be careful that we don't drift too far towards shift work as the fundamental structure of medical training. In the beginning, residents lived in the hospital, thus the term. As we move through these stormy seas, we don't want to trade patient safety for transient trainees. There is a middle ground here that will be informed by future research. There is something to be said for living in your house.
The word housestaff came from an era when you moved into the hospital (house) for your internship (staff). In those days, call every other night meant you missed half the learning opportunities. In those days, supervision was as remote as it could get. My, have things changed.
In 2003, the Accreditation Council for Graduate Medical Education (ACGME) implemented duty hour standards that called for restricting resident duty hours to no more than 80 hours per week, and no more than 24 continuous hours with an additional 6 hours for transfer of care.1 The expectation was that limiting work hours would have positive effects on outcomes, such as resident fatigue, the educational experience, and patient safety. A recent systematic review commissioned by the ACGME reviewed three decades worth of research on duty hours and patient safety. It reviewed laboratory studies of resident performance on clinical simulations, and tests of cognitive and fine motor skills under conditions of sleep deprivation. The results of these studies imply that limiting residents' duty hours should have positive effects on patient safety. However, that assumption has not been borne out. Paradoxically, examination of studies of alternative duty hour schedules, as well as the follow‐up studies on the impact of the New York duty hour rules and ACGME duty hour regulations have documented few measurable effects on patient safety.2 Thus, the simplistic view that fewer hours worked equals better patient care deserves a second‐look. In fact, a cause‐and‐effect relationship between resident fatigue and medical errors has yet to be proven.
The hospitalist movement was in full swing by 2003 and had already transformed the care of hospitalized patients. The early studies of hospitalists focused on cost and comparing outcomes with those of other providers. As many studies showed the economic benefit of hospitalists, residency programs began redesigning their inpatient services with hospitalists at the helm. Institutions sponsoring graduate medical education programs saw hospitalists as a necessary resource to enable and maintain compliance with duty hour reform. Some institutions created hospitalist‐only services with no housestaff (nonteaching services). Others created hospitalist teaching services that used indirect supervision at night from a hospitalist on‐call from home, while others had on‐site hospitalists at night that provided direct supervision of housestaff. It is clear that hospitalists are a versatile and valuable resource.
In this issue of the Journal of Hospital Medicine, Khanna and colleagues3 describe a retrospective medical record review at a large, urban academic hospital that evaluates the potential association between night admissions by night‐shiftbased hospitalists and a resident night float system, and hospitalization‐relevant outcomes. The outcomes included length of stay, hospital charges, Intensive Care Unit (ICU) transfers, Emergency Department visits within 30 days of discharge, 30‐day readmission rates, and poor outcomes within the first 24 hours of admission. Their results showed that night or weekend admission was not associated with worse hospitalization‐relevant outcomes. The extension of on‐site faculty supervision to nights, as well as awards for nursing excellence, likely had a strong positive impact on these outcomes. In some cases, night or weekend admission was associated with better outcomes, particularly in terms of ICU transfer during hospitalization and hospital charges. Maybe off‐hours care is not so off after all.
I am not sure where we are going, but the duty hour standards have been revised again. The new duty hour standards limit continuous duty hours for 1st‐year residents to 16, and all others to 24. Upper‐level residents can stay an additional 4 hours for transfer of care.4 Functionally, this proposed standard eliminates overnight call for 1st‐year residents. Although I would never advocate returning to the days of yore where autonomy and independence were the rule, I mourn the loss of extended shifts for 1st‐year residents. The loss of the extended shift will come at the expense of overnight call in many programs. I worry about the loss of some unique teaching moments for all residents, and the increased number of transitions in a patients' care. The hospital is a different place at night. It is less chaotic with fewer visitors, and there are less interruptions in patient care. There is time for teaching and learning because the work intensity is lower. The bedside teaching among senior and junior residents, during an acute patient episode in the middle of the night, is an experience that many of us would recount as an invaluable part of our training. It is also a time when on‐site faculty supervision and nurse staffing ratios are typically reduced. In other words, the entire system of care is different at night. This dichotomy in day versus night medicine has spurred research into whether patient outcomes differed between the two. Various investigators have looked at the differences in care, and there is some data to support that patient care outcomes are worse at night than during the day.58
The results from Khanna and colleagues3 are welcome news, since graduate medical education is undergoing a paradigm shift with further reductions in duty hours. Hospital‐based medicine is a 24‐hour operation, 7 days a week. We don't get to pick when patients are admitted or need care. In fact, this study demonstrates that 58% of the admissions came after 7 PM. It turns out that this pattern of admissions is not atypical.9 In light of the new standards, programs are left deciding where to deploy their 1st‐year resident resources. There are essentially two types of models to provide patient care off‐hours in the absence of an extended shift option: night float and night medicine. Night float implies that a trainee, usually a resident, comes from a different site or rotation to provide nocturnal care of a group of patients. That resident may do one isolated night of coverage or several nights in a row. The resident may perform cross‐cover functions, admit patients, or do both. Night medicine refers to having teams of housestaff at night along with an educational program as part of a rotation. The latter implies that trainees will get the full educational experience whether working days or nights. If one designs a model of care that puts 1st‐year residents where the volume is, they would come in late and leave early in the morning the next day. Both create more handoffs in patient care, which critics of these models have said lead to worse patient outcomes. This research challenges such earlier work and should make us all feel more comfortable that care at night does not need to be different if the proper systems are in place to manage it.
The graduate medical education community needs to take a step back and ask: Where are we going? As a long‐standing program director, I don't know anymore. As training has become competency‐based, and milestones are being developed for each level of training, we need to ask if reducing hours further is in the best interests of graduate medical education and, ultimately, the general public. I have long been interested in work intensity as a contributor to resident fatigue, burnout, and errors. A systematic review of resident workload and work activity looked at 21 studies examining resident time allocation and suggests that at least one‐third of resident time is spent on activities of limited educational value. Eight of the nine studies evaluating the impact of resident workload on patient care showed a negative impact on outcomes, including length of stay, mortality, patient satisfaction, medication errors, and lab utilization.10 If we are going to further restrict duty hours, then we need to transform the learning environment in a similar fashion. It does not seem logical to restrict duty hours further without a major redesign of the learning environment. I fear that all we have done is create more work compression because the system of care will be further strained in its weakest areas.
Hospitalists are uniquely poised to mitigate some of these concerns by being available for direct and indirect supervision, as well as being highly skilled at working in our complex systems. They are experts in systems‐based practice. The extension of direct faculty supervision to nights may be a major factor in eliminating the variances in care between nights and days. Not every program or institution will have the ability to employ night hospitalists, let alone have them in a supervisory and teaching role. Those that do can take great comfort in this research. Those that don't must challenge themselves to think about how to get there.
We all share the collective goal of producing the next generation of well‐trained physicians who will practice safe and effective care. As we begin to embrace competency‐based training, the prism through which we view patient safety must extend beyond duty hours and include learning environment reform. While the Khanna et al.3 study supports that off‐hours care is no longer off, we need to be careful that we don't drift too far towards shift work as the fundamental structure of medical training. In the beginning, residents lived in the hospital, thus the term. As we move through these stormy seas, we don't want to trade patient safety for transient trainees. There is a middle ground here that will be informed by future research. There is something to be said for living in your house.
- ACGME.Duty hour requirements. Available at: http://www.acgme.org/acWebsite/dutyHours/dh_ComProgrRequirmentsDutyHours0707.pdf. Accessed August 16,2010.
- ,,, et al.Systematic review of the literature on the impact of variation in residents' duty hour schedules on patient safety. Available at: http://acgme‐2010standards.org/pdf/Jefferson_Medical_College_Duty_Hours_Review.pdf. Accessed August 18,2010.
- ,,, et al.The association between night or weekend admission and hospitalization‐relevant patient outcomes.J Hosp Med2011;6:10–14.
- ACGME.Proposed duty hour standards. Available at: http://acgme‐2010standards.org/pdf/Proposed_Standards.pdf. Accessed August 16,2010.
- ,.Mortality among patients admitted to hospitals on weekends as compared with weekdays.N Engl J Med.2001;345(9):663–668.
- ,,, et al.Survival from in‐hospital cardiac arrest during nights and weekends.JAMA.2008;299(7):785–792.
- ,,,,.What effect does inpatient physician specialty and experience have on clinical outcomes and resource utilization on a general medicine service?J Gen Intern Med.2004;19:395–401.
- ,,,,,.Implementation of a voluntary hospitalist service at a community teaching hospital: improved clinical efficiency and patient outcomes.Ann Intern Med.2002;137:859–865.
- .Like night and day—shedding light on off‐hours care.N Engl J Med.2008;358(20):2091–2093.
- ,,.Systematic review of resident workload and work activity: impact on resident education and patient care. Available at: http://acgme‐2010standards.org/pdf/Resident_Duty_Hours_and_Related_Topics.pdf. Accessed on August 16,2010.
- ACGME.Duty hour requirements. Available at: http://www.acgme.org/acWebsite/dutyHours/dh_ComProgrRequirmentsDutyHours0707.pdf. Accessed August 16,2010.
- ,,, et al.Systematic review of the literature on the impact of variation in residents' duty hour schedules on patient safety. Available at: http://acgme‐2010standards.org/pdf/Jefferson_Medical_College_Duty_Hours_Review.pdf. Accessed August 18,2010.
- ,,, et al.The association between night or weekend admission and hospitalization‐relevant patient outcomes.J Hosp Med2011;6:10–14.
- ACGME.Proposed duty hour standards. Available at: http://acgme‐2010standards.org/pdf/Proposed_Standards.pdf. Accessed August 16,2010.
- ,.Mortality among patients admitted to hospitals on weekends as compared with weekdays.N Engl J Med.2001;345(9):663–668.
- ,,, et al.Survival from in‐hospital cardiac arrest during nights and weekends.JAMA.2008;299(7):785–792.
- ,,,,.What effect does inpatient physician specialty and experience have on clinical outcomes and resource utilization on a general medicine service?J Gen Intern Med.2004;19:395–401.
- ,,,,,.Implementation of a voluntary hospitalist service at a community teaching hospital: improved clinical efficiency and patient outcomes.Ann Intern Med.2002;137:859–865.
- .Like night and day—shedding light on off‐hours care.N Engl J Med.2008;358(20):2091–2093.
- ,,.Systematic review of resident workload and work activity: impact on resident education and patient care. Available at: http://acgme‐2010standards.org/pdf/Resident_Duty_Hours_and_Related_Topics.pdf. Accessed on August 16,2010.
Discharge Summary Completion
Discharge summaries (DS) correlate with rates of rehospitalization1, 2 and adverse events after discharge.3 The Joint Commission on the Accreditation of Healthcare Organizations acknowledges their importance and mandates that certain elements be included.4 Thus far, however, DS are not standardized across institutions and there is no expectation that they be available at postdischarge visits. There have been numerous attempts to improve the quality of DS by using more structured formats or computer generated summaries with positive results in term of comprehensiveness, clarity, and practitioner satisfaction58 but with persistence of serious errors and omissions.9
Postgraduate training is often the first opportunity for physicians to learn information transfer management skills. Unfortunately, DS are created by house staff who have minimal training in this area11 and feel like they have to learn by osmosis,12 resulting in poor quality DS and lack of availability at the point of care.1315
Previous research suggested that individualized feedback sessions for Internal Medicine residents improved the quality of certain aspects of their completed DS.10 We postulated that an audit and feedback educational intervention on DS for first year geriatric medicine fellows would also improve their quality. This technique involves chart or case review of clinical practice behaviors for a specific task followed by recommendation of new behaviors when applicable.16 Audit and feedback incorporates adult learning theory,1719 an essential part of continuous quality improvement that fits within the Accreditation Council for Graduate Medical Education (ACGME) competency of practice based learning and improvement,20 as an educational activity.
Methods
Setting
We conducted a preintervention post intervention study at the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai Medical Center (MSMC) in New York City between July 1, 2006 and June 30, 2007. The study received an exemption from the MSMC Institutional Review Board. First year geriatric medicine fellows at MSMC were required to complete 2 months of inpatient service; the first during the first 6 months of the academic year and the second during the last 6 months of the year. Fellows dictated all DS, which were transcribed and routed for signature to the attending of record. Prior to our study, a discharge summary template consisting of 21 items was developed for clinical use. Template items, agreed upon by an expert internal panel of geriatricians and interprofessional faculty, were selected for their importance in assuring a safe transition of older adults from the inpatient to the outpatient setting.
Participants
All 5 first‐year fellows at the Brookdale Department of Geriatrics and Palliative Medicine at MSMC were invited to participate in the study.
Intervention
Audit #1
All available DS for each fellow's first month of inpatient service were audited for completeness of the 21 item discharge summary template by 1 author (AD). The 21 items were focused on 4 distinct periods of the hospitalization: admission, hospital course, discharge planning, and postdischarge care (Figure 1).
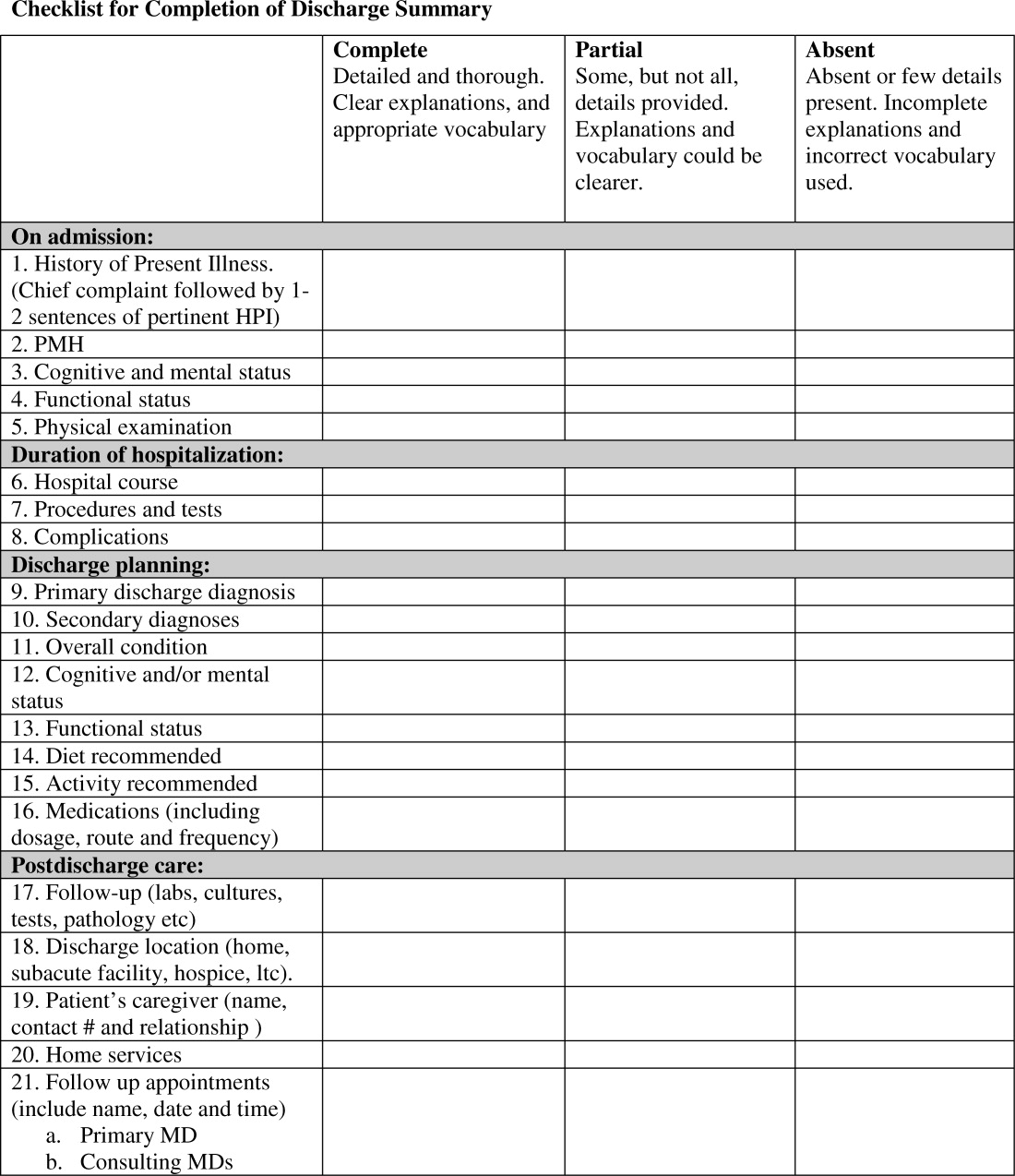
Content under each of the 21 items was classified as complete, partially complete, or absent. An item was considered complete if most information was present and appropriate medical terms were used, partially complete if information was unclear, and absent if no information was present for that area of the DS. To ensure investigator reliability, a random sample of 25% of each fellow's DS was scored by 2 additional investigators (RK and HF) and all disagreements were reviewed and resolved by consensus.
Feedback
Between December 2006 and January 2007, one‐on‐one formative feedback sessions were scheduled. The sessions were approximately 30 minutes long, confidential, performed by 1 of the authors (AD) and followed a written format. During these sessions, each fellow received the results of their discharge summary audit, each partially complete or absent item was discussed, and the importance of DS was emphasized.
Audit #2
All available DS for each fellow's second month of inpatient service were audited for completeness, using the same 21 item assessment tool and the same scoring system.
Statistical Analysis
To determine the impact of our audit and feedback intervention, we compared scores before and after formative feedback sessions, both overall and for the composite discharge summary scores for each of the 4 domains of care: admission, hospital course, discharge‐planning, and postdischarge care. Scores were dichotomized as being complete or partially complete or absent. We used generalized estimating equations to account for the clustering of DS within fellows. Analyses were performed using SAS 9.1 (SAS Institute, Inc., Cary, NC). All statistical tests were 2‐tailed and used a type I error rate of 0.01 to account for multiple comparisons.
Results
Five fellows participated, 4 of whom were women; 2 were in postgraduate year 4, 3 in year 5. A total of 158 DS were audited, 89 prefeedback and 79 postfeedback. Each fellow dictated an average of 17 DS during each inpatient month.
During Audit #1, the 21 item DS were complete among 71%, incomplete among 18%, absent among 11%. Admission items, hospital course items, and discharge planning items were complete among 70%, 78%, and 77% of DS respectively, but postdischarge items were complete among only 57%. Examining individual items, the lowest completion rates were found for test result follow‐up (42%), caregiver information (10%), and home services (64%), as well for assessment at admission and discharge of cognitive and mental status (56% and 53% respectively) and functional status (57% and 40%). Of note, all these items are of particular importance to geriatric care.
After receiving the audit and feedback intervention, fellows were more likely to complete all required discharge summary data when compared to prior‐to‐feedback (91% vs. 71%, P 0.001). Discharge summary completeness improved for all composite outcomes examining the four domains of care: admission (93% vs. 70%, P 0.001), hospital course (93% vs. 78%, P 0.001), discharge planning (93% vs. 77%, P 0.02), and postdischarge care (83% vs. 57%., P 0.001) (Table 1).
| Criteria | Preintervention | Postintervention | P Value* | ||
|---|---|---|---|---|---|
| Complete | Absent | Complete | Absent | ||
| |||||
| Admission composite (5 items) | 70 (3585) | 30 (1565) | 93 (79100) | 7 (021) | 0.001 |
| HPI | 79 (38100) | 21 (1563) | 100 | 0 | 0.001 |
| PMH | 94 (75100) | 5 (025) | 99 (93100) | 1 (07) | 0.001 |
| Cognitive/mental status | 56 (1979) | 44 (2182) | 99 (93100) | 1 (07) | 0.001 |
| Functional status | 57 (2588) | 43 (1375) | 97 (89100) | 2 (010) | 0.001 |
| Physical exam | 63 (19100) | 37 (082) | 72 (0100) | 28 (5100) | 0.27 |
| Hospital course composite (3 items) | 78 (2593) | 22 (775) | 93 (76100) | 7 (023) | 0.001 |
| Hospital course | 84 (25100) | 15 (076) | 99 (93100) | 1 (07) | 0.001 |
| Procedures and tests | 70 (690) | 30 (1094) | 90 (57100) | 10 (043) | 0.001 |
| Complications | 80 (4490) | 20 (556) | 90 (77100) | 10 (023) | 0.07 |
| Discharge planning composite (8 items) | 77 (4989) | 22 (1151) | 93 (64100) | 7 (036) | 0.02 |
| Primary diagnosis | 93 (75100) | 6 (026) | 100 | 0 | 0.03 |
| Secondary diagnosis | 82 (56100) | 18 (044) | 100 | 0 | 0.002 |
| Overall condition | 81 (38100) | 19 (062) | 86 (21100) | 14 (079) | 0.47 |
| Cognitive/mental status | 53 (1380) | 57 (2088) | 97 (93100) | 3 (07) | 0.001 |
| Functional status | 40 (1381) | 50 (1988) | 99 (93100) | 1 (07) | 0.001 |
| Diet | 89 (63100) | 12 (538) | 81 (0100) | 19 (0100) | 0.25 |
| Activity | 89 (69100) | 11 (032) | 82 (0100) | 18 (0100) | 0.49 |
| Medications | 83 (50100) | 17 (050) | 100 | 0 | 0.002 |
| Postdischarge care composite (5 items) | 57 (4183) | 43 (1759) | 83 (6998) | 18 (231) | 0.001 |
| F/U results | 42 (1190) | 58 (1089) | 81 (50100) | 20 (050) | 0.02 |
| Discharge location | 92 (88100) | 8 (012) | 100 | 0 | 0.02 |
| Caregiver info | 10 (025) | 89 (75100) | 48 (795) | 52 (584) | 0.001 |
| Home services | 64 (32100) | 35 (068) | 87 (7195) | 12 (029) | 0.001 |
| F/U appointments | 78 (33100) | 23 (067) | 96 (86100) | 4 (014) | 0.001 |
| Overall composite (21 items) | 71 (4287) | 29 (1358) | 91 (7399) | 9 (227) | 0.001 |
Discussion
Our study found that audit and feedback sessions significantly improved the completeness of DS dictated by geriatric medicine fellows at 1 academic medical center. Before feedback, completeness was high in most traditional areas of the DS including admission data, hospital course, and discharge planning, but was low in other areas critical for safe transitions of older adults such as postdischarge care, test follow‐up, caregiver information, and cognitive and functional status changes. These findings were surprising, as using a template should render a completion rate close to 100%. Notably, during feedback sessions, fellows suggested low completion rates were due to lack of awareness regarding the importance of completing all 21 items of the template and missing documentation in patient medical records.
Feedback sessions dramatically improved overall completeness of subsequent DS and in most of areas of specific importance for geriatric care, although we remain uncertain why all areas did not show improvement (for example, caregiver information completion remained low). One possible explanation is the lack of accurate documentation for all necessary items in the hospital medical record. Moreover, we did not observe completion improvement for other items, ie, diet and activity. Overall, we believe that drawing attention to areas of particular importance to geriatric care transitions and providing learners with individual reports on their performance increased their awareness and motivated changes to their practice, improving discharge summary completion.
Our study has limitations. This study was a pilot intervention without a control group, because of time and budgetary constraints. Also, we were unable to assess for sustainability because the fellows studied for this project graduated after the second audit. Third, we studied discharge summary completion; further research should focus on accuracy of discharge summary content. Finally, while we did not use any advanced technologies or materials, faculty time required to conduct the audit and feedback in this study was estimated at 45 hours. In our opinion this estimate would classify our audit and feedback intervention as a low external cost and moderately‐high human cost intervention, which may represent a potential barrier to generalizability. On the other hand, we believe that even an audit of a small sample of DS done by a physician could provide valuable data for feedback and would involve less faculty time.
Our finding that audit and feedback sessions improved the completeness of DS among house‐staff is important for 2 reasons. First, we were able to demonstrate that focused feedback targeted to areas of particular importance to the transition of older adults changed subsequent behavior and resulted in improved documentation of these areas. Second, our study provides evidence of a programmatic approach to address the ACGME competency of practice‐based learning and improvement. We believe that our intervention can be reproduced by training programs across the country and are hopeful that such interventions will result in improved patient outcomes during critical care transitions such as hospital discharge.
- ,Quality assessment of a discharge summary system.CMAJ.1995;152:1437–1442.
- ,,Accuracy of information on medicines in hospital discharge summaries.Intern Med J.2006;36:221–225.
- ,,,,The incidence and severity of adverse events affecting patients after discharge from the hospital.Ann Intern Med.2003;138:161–167.
- Available at: http://www.jointcommission.org./. The Joint Commission Requirements/Hospitals/Record of Care/Patient safety. Accessed July2010.
- ,,,,General practitioners' attitudes to computer‐generated surgical discharge letters.Med J Aust.1992;157(6):380–382.
- ,,,Do primary care physicians prefer dictated or computer‐generated discharge summaries?Am J Dis Child.1993;147(9):986–988.
- ,,,,,Evaluation of a computer‐generated discharge summary for patients with acute coronary syndromes.Br J Gen Pract.1998;48(429):1163–1164.
- ,,, et al.Creating a better discharge summary: improvement in quality and timeliness using an electronic dischanrge summary,J Hosp Med.2009;4(4):219–225.
- ,Communication with general practitioners after accident and emergency attendance: computer generated letters are often deficient.Emerg Med J.2003;20(3):256–257.
- ,,Evaluation of electronic discharge summaries: a comparison of documentation in electronic and handwritten discharge summaries.Int J Med Inform.2008;77:613–620.
- ,,,,,Are discharge summaries teachable? The effects of a discharge summary curriculum on the quality of discharge summaries in an internal medicine residency program.Acad Med.2006;81:S5–S8.
- ,,Experience of medical senior house officers in preparing discharge summaries.BMJ.1996;312:350.
- ,,,,,Deficits in communication and information transfer between hospital‐based and primary care physicians: implications for patient safety and continuity of care.JAMA.2007;297:831–841.
- ,,,Primary care physician attitudes regarding communication with hospitalists.Dis Mon.2002;48:218–229.
- ,,Improving the continuity of care following discharge of patients hospitalized with heart failure: is the discharge summary adequate?Can J Cardiol.2003;19:365–370.
- Effects of continuing medical education on improving physician clinical care and patient health: a review of systematic reviews.Int J Technol Assess Health Care.2005;21:380–385.
- ,,, et al.Impact of feedback and didactic sessions on the reporting behavior of upper endoscopic findings by physicians and nurses.Clin Gastroenterol Hepatol.2007;5:326–330.
- ,,Prospective assessment of the impact of feedback on colonoscopy performance.Aliment Pharmacol Ther.2006;24:313–318.
- ,,, et al.Inpatient care to community care: improving clinical handover in the private mental health setting.Med J Aust.2009;190(11 Suppl):S144–S149.
- Available at: http://www.Acgme.org, Record of care, Treatment, and Serives, Standard RC.02.04.01. Accessed July2010.
Discharge summaries (DS) correlate with rates of rehospitalization1, 2 and adverse events after discharge.3 The Joint Commission on the Accreditation of Healthcare Organizations acknowledges their importance and mandates that certain elements be included.4 Thus far, however, DS are not standardized across institutions and there is no expectation that they be available at postdischarge visits. There have been numerous attempts to improve the quality of DS by using more structured formats or computer generated summaries with positive results in term of comprehensiveness, clarity, and practitioner satisfaction58 but with persistence of serious errors and omissions.9
Postgraduate training is often the first opportunity for physicians to learn information transfer management skills. Unfortunately, DS are created by house staff who have minimal training in this area11 and feel like they have to learn by osmosis,12 resulting in poor quality DS and lack of availability at the point of care.1315
Previous research suggested that individualized feedback sessions for Internal Medicine residents improved the quality of certain aspects of their completed DS.10 We postulated that an audit and feedback educational intervention on DS for first year geriatric medicine fellows would also improve their quality. This technique involves chart or case review of clinical practice behaviors for a specific task followed by recommendation of new behaviors when applicable.16 Audit and feedback incorporates adult learning theory,1719 an essential part of continuous quality improvement that fits within the Accreditation Council for Graduate Medical Education (ACGME) competency of practice based learning and improvement,20 as an educational activity.
Methods
Setting
We conducted a preintervention post intervention study at the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai Medical Center (MSMC) in New York City between July 1, 2006 and June 30, 2007. The study received an exemption from the MSMC Institutional Review Board. First year geriatric medicine fellows at MSMC were required to complete 2 months of inpatient service; the first during the first 6 months of the academic year and the second during the last 6 months of the year. Fellows dictated all DS, which were transcribed and routed for signature to the attending of record. Prior to our study, a discharge summary template consisting of 21 items was developed for clinical use. Template items, agreed upon by an expert internal panel of geriatricians and interprofessional faculty, were selected for their importance in assuring a safe transition of older adults from the inpatient to the outpatient setting.
Participants
All 5 first‐year fellows at the Brookdale Department of Geriatrics and Palliative Medicine at MSMC were invited to participate in the study.
Intervention
Audit #1
All available DS for each fellow's first month of inpatient service were audited for completeness of the 21 item discharge summary template by 1 author (AD). The 21 items were focused on 4 distinct periods of the hospitalization: admission, hospital course, discharge planning, and postdischarge care (Figure 1).

Content under each of the 21 items was classified as complete, partially complete, or absent. An item was considered complete if most information was present and appropriate medical terms were used, partially complete if information was unclear, and absent if no information was present for that area of the DS. To ensure investigator reliability, a random sample of 25% of each fellow's DS was scored by 2 additional investigators (RK and HF) and all disagreements were reviewed and resolved by consensus.
Feedback
Between December 2006 and January 2007, one‐on‐one formative feedback sessions were scheduled. The sessions were approximately 30 minutes long, confidential, performed by 1 of the authors (AD) and followed a written format. During these sessions, each fellow received the results of their discharge summary audit, each partially complete or absent item was discussed, and the importance of DS was emphasized.
Audit #2
All available DS for each fellow's second month of inpatient service were audited for completeness, using the same 21 item assessment tool and the same scoring system.
Statistical Analysis
To determine the impact of our audit and feedback intervention, we compared scores before and after formative feedback sessions, both overall and for the composite discharge summary scores for each of the 4 domains of care: admission, hospital course, discharge‐planning, and postdischarge care. Scores were dichotomized as being complete or partially complete or absent. We used generalized estimating equations to account for the clustering of DS within fellows. Analyses were performed using SAS 9.1 (SAS Institute, Inc., Cary, NC). All statistical tests were 2‐tailed and used a type I error rate of 0.01 to account for multiple comparisons.
Results
Five fellows participated, 4 of whom were women; 2 were in postgraduate year 4, 3 in year 5. A total of 158 DS were audited, 89 prefeedback and 79 postfeedback. Each fellow dictated an average of 17 DS during each inpatient month.
During Audit #1, the 21 item DS were complete among 71%, incomplete among 18%, absent among 11%. Admission items, hospital course items, and discharge planning items were complete among 70%, 78%, and 77% of DS respectively, but postdischarge items were complete among only 57%. Examining individual items, the lowest completion rates were found for test result follow‐up (42%), caregiver information (10%), and home services (64%), as well for assessment at admission and discharge of cognitive and mental status (56% and 53% respectively) and functional status (57% and 40%). Of note, all these items are of particular importance to geriatric care.
After receiving the audit and feedback intervention, fellows were more likely to complete all required discharge summary data when compared to prior‐to‐feedback (91% vs. 71%, P 0.001). Discharge summary completeness improved for all composite outcomes examining the four domains of care: admission (93% vs. 70%, P 0.001), hospital course (93% vs. 78%, P 0.001), discharge planning (93% vs. 77%, P 0.02), and postdischarge care (83% vs. 57%., P 0.001) (Table 1).
| Criteria | Preintervention | Postintervention | P Value* | ||
|---|---|---|---|---|---|
| Complete | Absent | Complete | Absent | ||
| |||||
| Admission composite (5 items) | 70 (3585) | 30 (1565) | 93 (79100) | 7 (021) | 0.001 |
| HPI | 79 (38100) | 21 (1563) | 100 | 0 | 0.001 |
| PMH | 94 (75100) | 5 (025) | 99 (93100) | 1 (07) | 0.001 |
| Cognitive/mental status | 56 (1979) | 44 (2182) | 99 (93100) | 1 (07) | 0.001 |
| Functional status | 57 (2588) | 43 (1375) | 97 (89100) | 2 (010) | 0.001 |
| Physical exam | 63 (19100) | 37 (082) | 72 (0100) | 28 (5100) | 0.27 |
| Hospital course composite (3 items) | 78 (2593) | 22 (775) | 93 (76100) | 7 (023) | 0.001 |
| Hospital course | 84 (25100) | 15 (076) | 99 (93100) | 1 (07) | 0.001 |
| Procedures and tests | 70 (690) | 30 (1094) | 90 (57100) | 10 (043) | 0.001 |
| Complications | 80 (4490) | 20 (556) | 90 (77100) | 10 (023) | 0.07 |
| Discharge planning composite (8 items) | 77 (4989) | 22 (1151) | 93 (64100) | 7 (036) | 0.02 |
| Primary diagnosis | 93 (75100) | 6 (026) | 100 | 0 | 0.03 |
| Secondary diagnosis | 82 (56100) | 18 (044) | 100 | 0 | 0.002 |
| Overall condition | 81 (38100) | 19 (062) | 86 (21100) | 14 (079) | 0.47 |
| Cognitive/mental status | 53 (1380) | 57 (2088) | 97 (93100) | 3 (07) | 0.001 |
| Functional status | 40 (1381) | 50 (1988) | 99 (93100) | 1 (07) | 0.001 |
| Diet | 89 (63100) | 12 (538) | 81 (0100) | 19 (0100) | 0.25 |
| Activity | 89 (69100) | 11 (032) | 82 (0100) | 18 (0100) | 0.49 |
| Medications | 83 (50100) | 17 (050) | 100 | 0 | 0.002 |
| Postdischarge care composite (5 items) | 57 (4183) | 43 (1759) | 83 (6998) | 18 (231) | 0.001 |
| F/U results | 42 (1190) | 58 (1089) | 81 (50100) | 20 (050) | 0.02 |
| Discharge location | 92 (88100) | 8 (012) | 100 | 0 | 0.02 |
| Caregiver info | 10 (025) | 89 (75100) | 48 (795) | 52 (584) | 0.001 |
| Home services | 64 (32100) | 35 (068) | 87 (7195) | 12 (029) | 0.001 |
| F/U appointments | 78 (33100) | 23 (067) | 96 (86100) | 4 (014) | 0.001 |
| Overall composite (21 items) | 71 (4287) | 29 (1358) | 91 (7399) | 9 (227) | 0.001 |
Discussion
Our study found that audit and feedback sessions significantly improved the completeness of DS dictated by geriatric medicine fellows at 1 academic medical center. Before feedback, completeness was high in most traditional areas of the DS including admission data, hospital course, and discharge planning, but was low in other areas critical for safe transitions of older adults such as postdischarge care, test follow‐up, caregiver information, and cognitive and functional status changes. These findings were surprising, as using a template should render a completion rate close to 100%. Notably, during feedback sessions, fellows suggested low completion rates were due to lack of awareness regarding the importance of completing all 21 items of the template and missing documentation in patient medical records.
Feedback sessions dramatically improved overall completeness of subsequent DS and in most of areas of specific importance for geriatric care, although we remain uncertain why all areas did not show improvement (for example, caregiver information completion remained low). One possible explanation is the lack of accurate documentation for all necessary items in the hospital medical record. Moreover, we did not observe completion improvement for other items, ie, diet and activity. Overall, we believe that drawing attention to areas of particular importance to geriatric care transitions and providing learners with individual reports on their performance increased their awareness and motivated changes to their practice, improving discharge summary completion.
Our study has limitations. This study was a pilot intervention without a control group, because of time and budgetary constraints. Also, we were unable to assess for sustainability because the fellows studied for this project graduated after the second audit. Third, we studied discharge summary completion; further research should focus on accuracy of discharge summary content. Finally, while we did not use any advanced technologies or materials, faculty time required to conduct the audit and feedback in this study was estimated at 45 hours. In our opinion this estimate would classify our audit and feedback intervention as a low external cost and moderately‐high human cost intervention, which may represent a potential barrier to generalizability. On the other hand, we believe that even an audit of a small sample of DS done by a physician could provide valuable data for feedback and would involve less faculty time.
Our finding that audit and feedback sessions improved the completeness of DS among house‐staff is important for 2 reasons. First, we were able to demonstrate that focused feedback targeted to areas of particular importance to the transition of older adults changed subsequent behavior and resulted in improved documentation of these areas. Second, our study provides evidence of a programmatic approach to address the ACGME competency of practice‐based learning and improvement. We believe that our intervention can be reproduced by training programs across the country and are hopeful that such interventions will result in improved patient outcomes during critical care transitions such as hospital discharge.
Discharge summaries (DS) correlate with rates of rehospitalization1, 2 and adverse events after discharge.3 The Joint Commission on the Accreditation of Healthcare Organizations acknowledges their importance and mandates that certain elements be included.4 Thus far, however, DS are not standardized across institutions and there is no expectation that they be available at postdischarge visits. There have been numerous attempts to improve the quality of DS by using more structured formats or computer generated summaries with positive results in term of comprehensiveness, clarity, and practitioner satisfaction58 but with persistence of serious errors and omissions.9
Postgraduate training is often the first opportunity for physicians to learn information transfer management skills. Unfortunately, DS are created by house staff who have minimal training in this area11 and feel like they have to learn by osmosis,12 resulting in poor quality DS and lack of availability at the point of care.1315
Previous research suggested that individualized feedback sessions for Internal Medicine residents improved the quality of certain aspects of their completed DS.10 We postulated that an audit and feedback educational intervention on DS for first year geriatric medicine fellows would also improve their quality. This technique involves chart or case review of clinical practice behaviors for a specific task followed by recommendation of new behaviors when applicable.16 Audit and feedback incorporates adult learning theory,1719 an essential part of continuous quality improvement that fits within the Accreditation Council for Graduate Medical Education (ACGME) competency of practice based learning and improvement,20 as an educational activity.
Methods
Setting
We conducted a preintervention post intervention study at the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai Medical Center (MSMC) in New York City between July 1, 2006 and June 30, 2007. The study received an exemption from the MSMC Institutional Review Board. First year geriatric medicine fellows at MSMC were required to complete 2 months of inpatient service; the first during the first 6 months of the academic year and the second during the last 6 months of the year. Fellows dictated all DS, which were transcribed and routed for signature to the attending of record. Prior to our study, a discharge summary template consisting of 21 items was developed for clinical use. Template items, agreed upon by an expert internal panel of geriatricians and interprofessional faculty, were selected for their importance in assuring a safe transition of older adults from the inpatient to the outpatient setting.
Participants
All 5 first‐year fellows at the Brookdale Department of Geriatrics and Palliative Medicine at MSMC were invited to participate in the study.
Intervention
Audit #1
All available DS for each fellow's first month of inpatient service were audited for completeness of the 21 item discharge summary template by 1 author (AD). The 21 items were focused on 4 distinct periods of the hospitalization: admission, hospital course, discharge planning, and postdischarge care (Figure 1).

Content under each of the 21 items was classified as complete, partially complete, or absent. An item was considered complete if most information was present and appropriate medical terms were used, partially complete if information was unclear, and absent if no information was present for that area of the DS. To ensure investigator reliability, a random sample of 25% of each fellow's DS was scored by 2 additional investigators (RK and HF) and all disagreements were reviewed and resolved by consensus.
Feedback
Between December 2006 and January 2007, one‐on‐one formative feedback sessions were scheduled. The sessions were approximately 30 minutes long, confidential, performed by 1 of the authors (AD) and followed a written format. During these sessions, each fellow received the results of their discharge summary audit, each partially complete or absent item was discussed, and the importance of DS was emphasized.
Audit #2
All available DS for each fellow's second month of inpatient service were audited for completeness, using the same 21 item assessment tool and the same scoring system.
Statistical Analysis
To determine the impact of our audit and feedback intervention, we compared scores before and after formative feedback sessions, both overall and for the composite discharge summary scores for each of the 4 domains of care: admission, hospital course, discharge‐planning, and postdischarge care. Scores were dichotomized as being complete or partially complete or absent. We used generalized estimating equations to account for the clustering of DS within fellows. Analyses were performed using SAS 9.1 (SAS Institute, Inc., Cary, NC). All statistical tests were 2‐tailed and used a type I error rate of 0.01 to account for multiple comparisons.
Results
Five fellows participated, 4 of whom were women; 2 were in postgraduate year 4, 3 in year 5. A total of 158 DS were audited, 89 prefeedback and 79 postfeedback. Each fellow dictated an average of 17 DS during each inpatient month.
During Audit #1, the 21 item DS were complete among 71%, incomplete among 18%, absent among 11%. Admission items, hospital course items, and discharge planning items were complete among 70%, 78%, and 77% of DS respectively, but postdischarge items were complete among only 57%. Examining individual items, the lowest completion rates were found for test result follow‐up (42%), caregiver information (10%), and home services (64%), as well for assessment at admission and discharge of cognitive and mental status (56% and 53% respectively) and functional status (57% and 40%). Of note, all these items are of particular importance to geriatric care.
After receiving the audit and feedback intervention, fellows were more likely to complete all required discharge summary data when compared to prior‐to‐feedback (91% vs. 71%, P 0.001). Discharge summary completeness improved for all composite outcomes examining the four domains of care: admission (93% vs. 70%, P 0.001), hospital course (93% vs. 78%, P 0.001), discharge planning (93% vs. 77%, P 0.02), and postdischarge care (83% vs. 57%., P 0.001) (Table 1).
| Criteria | Preintervention | Postintervention | P Value* | ||
|---|---|---|---|---|---|
| Complete | Absent | Complete | Absent | ||
| |||||
| Admission composite (5 items) | 70 (3585) | 30 (1565) | 93 (79100) | 7 (021) | 0.001 |
| HPI | 79 (38100) | 21 (1563) | 100 | 0 | 0.001 |
| PMH | 94 (75100) | 5 (025) | 99 (93100) | 1 (07) | 0.001 |
| Cognitive/mental status | 56 (1979) | 44 (2182) | 99 (93100) | 1 (07) | 0.001 |
| Functional status | 57 (2588) | 43 (1375) | 97 (89100) | 2 (010) | 0.001 |
| Physical exam | 63 (19100) | 37 (082) | 72 (0100) | 28 (5100) | 0.27 |
| Hospital course composite (3 items) | 78 (2593) | 22 (775) | 93 (76100) | 7 (023) | 0.001 |
| Hospital course | 84 (25100) | 15 (076) | 99 (93100) | 1 (07) | 0.001 |
| Procedures and tests | 70 (690) | 30 (1094) | 90 (57100) | 10 (043) | 0.001 |
| Complications | 80 (4490) | 20 (556) | 90 (77100) | 10 (023) | 0.07 |
| Discharge planning composite (8 items) | 77 (4989) | 22 (1151) | 93 (64100) | 7 (036) | 0.02 |
| Primary diagnosis | 93 (75100) | 6 (026) | 100 | 0 | 0.03 |
| Secondary diagnosis | 82 (56100) | 18 (044) | 100 | 0 | 0.002 |
| Overall condition | 81 (38100) | 19 (062) | 86 (21100) | 14 (079) | 0.47 |
| Cognitive/mental status | 53 (1380) | 57 (2088) | 97 (93100) | 3 (07) | 0.001 |
| Functional status | 40 (1381) | 50 (1988) | 99 (93100) | 1 (07) | 0.001 |
| Diet | 89 (63100) | 12 (538) | 81 (0100) | 19 (0100) | 0.25 |
| Activity | 89 (69100) | 11 (032) | 82 (0100) | 18 (0100) | 0.49 |
| Medications | 83 (50100) | 17 (050) | 100 | 0 | 0.002 |
| Postdischarge care composite (5 items) | 57 (4183) | 43 (1759) | 83 (6998) | 18 (231) | 0.001 |
| F/U results | 42 (1190) | 58 (1089) | 81 (50100) | 20 (050) | 0.02 |
| Discharge location | 92 (88100) | 8 (012) | 100 | 0 | 0.02 |
| Caregiver info | 10 (025) | 89 (75100) | 48 (795) | 52 (584) | 0.001 |
| Home services | 64 (32100) | 35 (068) | 87 (7195) | 12 (029) | 0.001 |
| F/U appointments | 78 (33100) | 23 (067) | 96 (86100) | 4 (014) | 0.001 |
| Overall composite (21 items) | 71 (4287) | 29 (1358) | 91 (7399) | 9 (227) | 0.001 |
Discussion
Our study found that audit and feedback sessions significantly improved the completeness of DS dictated by geriatric medicine fellows at 1 academic medical center. Before feedback, completeness was high in most traditional areas of the DS including admission data, hospital course, and discharge planning, but was low in other areas critical for safe transitions of older adults such as postdischarge care, test follow‐up, caregiver information, and cognitive and functional status changes. These findings were surprising, as using a template should render a completion rate close to 100%. Notably, during feedback sessions, fellows suggested low completion rates were due to lack of awareness regarding the importance of completing all 21 items of the template and missing documentation in patient medical records.
Feedback sessions dramatically improved overall completeness of subsequent DS and in most of areas of specific importance for geriatric care, although we remain uncertain why all areas did not show improvement (for example, caregiver information completion remained low). One possible explanation is the lack of accurate documentation for all necessary items in the hospital medical record. Moreover, we did not observe completion improvement for other items, ie, diet and activity. Overall, we believe that drawing attention to areas of particular importance to geriatric care transitions and providing learners with individual reports on their performance increased their awareness and motivated changes to their practice, improving discharge summary completion.
Our study has limitations. This study was a pilot intervention without a control group, because of time and budgetary constraints. Also, we were unable to assess for sustainability because the fellows studied for this project graduated after the second audit. Third, we studied discharge summary completion; further research should focus on accuracy of discharge summary content. Finally, while we did not use any advanced technologies or materials, faculty time required to conduct the audit and feedback in this study was estimated at 45 hours. In our opinion this estimate would classify our audit and feedback intervention as a low external cost and moderately‐high human cost intervention, which may represent a potential barrier to generalizability. On the other hand, we believe that even an audit of a small sample of DS done by a physician could provide valuable data for feedback and would involve less faculty time.
Our finding that audit and feedback sessions improved the completeness of DS among house‐staff is important for 2 reasons. First, we were able to demonstrate that focused feedback targeted to areas of particular importance to the transition of older adults changed subsequent behavior and resulted in improved documentation of these areas. Second, our study provides evidence of a programmatic approach to address the ACGME competency of practice‐based learning and improvement. We believe that our intervention can be reproduced by training programs across the country and are hopeful that such interventions will result in improved patient outcomes during critical care transitions such as hospital discharge.
- ,Quality assessment of a discharge summary system.CMAJ.1995;152:1437–1442.
- ,,Accuracy of information on medicines in hospital discharge summaries.Intern Med J.2006;36:221–225.
- ,,,,The incidence and severity of adverse events affecting patients after discharge from the hospital.Ann Intern Med.2003;138:161–167.
- Available at: http://www.jointcommission.org./. The Joint Commission Requirements/Hospitals/Record of Care/Patient safety. Accessed July2010.
- ,,,,General practitioners' attitudes to computer‐generated surgical discharge letters.Med J Aust.1992;157(6):380–382.
- ,,,Do primary care physicians prefer dictated or computer‐generated discharge summaries?Am J Dis Child.1993;147(9):986–988.
- ,,,,,Evaluation of a computer‐generated discharge summary for patients with acute coronary syndromes.Br J Gen Pract.1998;48(429):1163–1164.
- ,,, et al.Creating a better discharge summary: improvement in quality and timeliness using an electronic dischanrge summary,J Hosp Med.2009;4(4):219–225.
- ,Communication with general practitioners after accident and emergency attendance: computer generated letters are often deficient.Emerg Med J.2003;20(3):256–257.
- ,,Evaluation of electronic discharge summaries: a comparison of documentation in electronic and handwritten discharge summaries.Int J Med Inform.2008;77:613–620.
- ,,,,,Are discharge summaries teachable? The effects of a discharge summary curriculum on the quality of discharge summaries in an internal medicine residency program.Acad Med.2006;81:S5–S8.
- ,,Experience of medical senior house officers in preparing discharge summaries.BMJ.1996;312:350.
- ,,,,,Deficits in communication and information transfer between hospital‐based and primary care physicians: implications for patient safety and continuity of care.JAMA.2007;297:831–841.
- ,,,Primary care physician attitudes regarding communication with hospitalists.Dis Mon.2002;48:218–229.
- ,,Improving the continuity of care following discharge of patients hospitalized with heart failure: is the discharge summary adequate?Can J Cardiol.2003;19:365–370.
- Effects of continuing medical education on improving physician clinical care and patient health: a review of systematic reviews.Int J Technol Assess Health Care.2005;21:380–385.
- ,,, et al.Impact of feedback and didactic sessions on the reporting behavior of upper endoscopic findings by physicians and nurses.Clin Gastroenterol Hepatol.2007;5:326–330.
- ,,Prospective assessment of the impact of feedback on colonoscopy performance.Aliment Pharmacol Ther.2006;24:313–318.
- ,,, et al.Inpatient care to community care: improving clinical handover in the private mental health setting.Med J Aust.2009;190(11 Suppl):S144–S149.
- Available at: http://www.Acgme.org, Record of care, Treatment, and Serives, Standard RC.02.04.01. Accessed July2010.
- ,Quality assessment of a discharge summary system.CMAJ.1995;152:1437–1442.
- ,,Accuracy of information on medicines in hospital discharge summaries.Intern Med J.2006;36:221–225.
- ,,,,The incidence and severity of adverse events affecting patients after discharge from the hospital.Ann Intern Med.2003;138:161–167.
- Available at: http://www.jointcommission.org./. The Joint Commission Requirements/Hospitals/Record of Care/Patient safety. Accessed July2010.
- ,,,,General practitioners' attitudes to computer‐generated surgical discharge letters.Med J Aust.1992;157(6):380–382.
- ,,,Do primary care physicians prefer dictated or computer‐generated discharge summaries?Am J Dis Child.1993;147(9):986–988.
- ,,,,,Evaluation of a computer‐generated discharge summary for patients with acute coronary syndromes.Br J Gen Pract.1998;48(429):1163–1164.
- ,,, et al.Creating a better discharge summary: improvement in quality and timeliness using an electronic dischanrge summary,J Hosp Med.2009;4(4):219–225.
- ,Communication with general practitioners after accident and emergency attendance: computer generated letters are often deficient.Emerg Med J.2003;20(3):256–257.
- ,,Evaluation of electronic discharge summaries: a comparison of documentation in electronic and handwritten discharge summaries.Int J Med Inform.2008;77:613–620.
- ,,,,,Are discharge summaries teachable? The effects of a discharge summary curriculum on the quality of discharge summaries in an internal medicine residency program.Acad Med.2006;81:S5–S8.
- ,,Experience of medical senior house officers in preparing discharge summaries.BMJ.1996;312:350.
- ,,,,,Deficits in communication and information transfer between hospital‐based and primary care physicians: implications for patient safety and continuity of care.JAMA.2007;297:831–841.
- ,,,Primary care physician attitudes regarding communication with hospitalists.Dis Mon.2002;48:218–229.
- ,,Improving the continuity of care following discharge of patients hospitalized with heart failure: is the discharge summary adequate?Can J Cardiol.2003;19:365–370.
- Effects of continuing medical education on improving physician clinical care and patient health: a review of systematic reviews.Int J Technol Assess Health Care.2005;21:380–385.
- ,,, et al.Impact of feedback and didactic sessions on the reporting behavior of upper endoscopic findings by physicians and nurses.Clin Gastroenterol Hepatol.2007;5:326–330.
- ,,Prospective assessment of the impact of feedback on colonoscopy performance.Aliment Pharmacol Ther.2006;24:313–318.
- ,,, et al.Inpatient care to community care: improving clinical handover in the private mental health setting.Med J Aust.2009;190(11 Suppl):S144–S149.
- Available at: http://www.Acgme.org, Record of care, Treatment, and Serives, Standard RC.02.04.01. Accessed July2010.
Continuing Medical Education Program in
If you wish to receive credit for this activity, which beginson the next page, please refer to the website:
Accreditation and Designation Statement
Blackwell Futura Media Services designates this educational activity for a 1 AMA PRA Category 1 Credit. Physicians should only claim credit commensurate with the extent of their participation in the activity.
Blackwell Futura Media Services is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Educational Objectives
Continuous participation in the Journal of Hospital Medicine CME program will enable learners to be better able to:
-
Interpret clinical guidelines and their applications for higher quality and more efficient care for all hospitalized patients.
-
Describe the standard of care for common illnesses and conditions treated in the hospital; such as pneumonia, COPD exacerbation, acute coronary syndrome, HF exacerbation, glycemic control, venous thromboembolic disease, stroke, etc.
-
Discuss evidence‐based recommendations involving transitions of care, including the hospital discharge process.
-
Gain insights into the roles of hospitalists as medical educators, researchers, medical ethicists, palliative care providers, and hospital‐based geriatricians.
-
Incorporate best practices for hospitalist administration, including quality improvement, patient safety, practice management, leadership, and demonstrating hospitalist value.
-
Identify evidence‐based best practices and trends for both adult and pediatric hospital medicine.
Instructions on Receiving Credit
For information on applicability and acceptance of continuing medical education credit for this activity, please consult your professional licensing board.
This activity is designed to be completed within the time designated on the title page; physicians should claim only those credits that reflect the time actually spent in the activity. To successfully earn credit, participants must complete the activity during the valid credit period that is noted on the title page.
Follow these steps to earn credit:
-
Log on to
www.blackwellpublishing.com/cme . -
Read the target audience, learning objectives, and author disclosures.
-
Read the article in print or online format.
-
Reflect on the article.
-
Access the CME Exam, and choose the best answer to each question.
-
Complete the required evaluation component of the activity.
If you wish to receive credit for this activity, which beginson the next page, please refer to the website:
Accreditation and Designation Statement
Blackwell Futura Media Services designates this educational activity for a 1 AMA PRA Category 1 Credit. Physicians should only claim credit commensurate with the extent of their participation in the activity.
Blackwell Futura Media Services is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Educational Objectives
Continuous participation in the Journal of Hospital Medicine CME program will enable learners to be better able to:
-
Interpret clinical guidelines and their applications for higher quality and more efficient care for all hospitalized patients.
-
Describe the standard of care for common illnesses and conditions treated in the hospital; such as pneumonia, COPD exacerbation, acute coronary syndrome, HF exacerbation, glycemic control, venous thromboembolic disease, stroke, etc.
-
Discuss evidence‐based recommendations involving transitions of care, including the hospital discharge process.
-
Gain insights into the roles of hospitalists as medical educators, researchers, medical ethicists, palliative care providers, and hospital‐based geriatricians.
-
Incorporate best practices for hospitalist administration, including quality improvement, patient safety, practice management, leadership, and demonstrating hospitalist value.
-
Identify evidence‐based best practices and trends for both adult and pediatric hospital medicine.
Instructions on Receiving Credit
For information on applicability and acceptance of continuing medical education credit for this activity, please consult your professional licensing board.
This activity is designed to be completed within the time designated on the title page; physicians should claim only those credits that reflect the time actually spent in the activity. To successfully earn credit, participants must complete the activity during the valid credit period that is noted on the title page.
Follow these steps to earn credit:
-
Log on to
www.blackwellpublishing.com/cme . -
Read the target audience, learning objectives, and author disclosures.
-
Read the article in print or online format.
-
Reflect on the article.
-
Access the CME Exam, and choose the best answer to each question.
-
Complete the required evaluation component of the activity.
If you wish to receive credit for this activity, which beginson the next page, please refer to the website:
Accreditation and Designation Statement
Blackwell Futura Media Services designates this educational activity for a 1 AMA PRA Category 1 Credit. Physicians should only claim credit commensurate with the extent of their participation in the activity.
Blackwell Futura Media Services is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Educational Objectives
Continuous participation in the Journal of Hospital Medicine CME program will enable learners to be better able to:
-
Interpret clinical guidelines and their applications for higher quality and more efficient care for all hospitalized patients.
-
Describe the standard of care for common illnesses and conditions treated in the hospital; such as pneumonia, COPD exacerbation, acute coronary syndrome, HF exacerbation, glycemic control, venous thromboembolic disease, stroke, etc.
-
Discuss evidence‐based recommendations involving transitions of care, including the hospital discharge process.
-
Gain insights into the roles of hospitalists as medical educators, researchers, medical ethicists, palliative care providers, and hospital‐based geriatricians.
-
Incorporate best practices for hospitalist administration, including quality improvement, patient safety, practice management, leadership, and demonstrating hospitalist value.
-
Identify evidence‐based best practices and trends for both adult and pediatric hospital medicine.
Instructions on Receiving Credit
For information on applicability and acceptance of continuing medical education credit for this activity, please consult your professional licensing board.
This activity is designed to be completed within the time designated on the title page; physicians should claim only those credits that reflect the time actually spent in the activity. To successfully earn credit, participants must complete the activity during the valid credit period that is noted on the title page.
Follow these steps to earn credit:
-
Log on to
www.blackwellpublishing.com/cme . -
Read the target audience, learning objectives, and author disclosures.
-
Read the article in print or online format.
-
Reflect on the article.
-
Access the CME Exam, and choose the best answer to each question.
-
Complete the required evaluation component of the activity.
The need for mentors in the odyssey of the academic hospitalist
This issue of the Journal of Hospital Medicine features an important contribution concerning the current state of academic hospital medicine. The survey of 57 hospitalists revealed what many of us already suspected: the state of mentorship in academic hospitalist groups is unsatisfactory.1
While the conclusion is alarming, it is also not surprising. Over the past decade academic medical centers enthusiastically hired hospitalists to improve efficiency for inpatient services and to lessen the effect of Accreditation Council for Graduate Medical Education (ACGME) regulations on duty hours and patient caps. Few departments of internal medicine, however, hired academic hospitalists with the intent of creating academic divisions. Thus many institutions appear to view hospitalists primarily as hospital employees ignoring their potential academic contributions, and as a result it should not be a surprise that many hospitalist groups lack the mentorship infrastructure of other divisions within a typical Department of Medicine. Compounding the hospital employee problem, the new field of academic hospital medicine has emerged only in the last decade, a time frame that has resulted in very few hospitalists qualified to serve as senior mentors.
We cannot easily remove these limitations: the past is the past, and over time, hospital medicine will mature and develop more senior mentors. But what should we do until that maturation occurs? We believe that the academic work of hospitalists, both in education and research (Quality and Patient Safety) are important endeavors too valuable to be left to chance. With 30,000 hospitalists delivering care, it is critical that research in the optimal delivery of this care be performed, targeting systems improvements to enact anticipated outcomes in quality and patient safety. The physicians who are regularly and intimately involved in this system of inpatient care delivery, the hospitalists, are best suited for identifying the unique features of the inpatient care system needing improvement. Mentorship is essential in ensuring the advancement of both areas, and the sustainability of hospital medicine in medical academe. The article by Harrison et al.1 both establishes the depth of these issues and provides important insights into potential solutions for closing this mentorship gap while the field matures.
Utilizing Other Mentors
No measure of systems change will make young hospitalists immediately experienced, such that they have the sophistication to be senior mentors for younger hospitalists. But we can compensate for this temporary gap in mentorship experience. First, in the next 5 to 10 years, young academic hospitalists need explicit direction from those within Departments of Medicine who do have this mentorship experience, even if these mentors do not work in hospital medicine. Mentors within General Internal Medicine or the subspecialties can still provide the guidance and support to ensure that academic hospitalists are engaging in the appropriate endeavors toward promotion and intellectual growth. Second, academic hospitalists have to seek out mentorship from afar through their participation in the organizations primarily devoted to the academic welfare of hospitalists: The Society of Hospital Medicine (SHM) and the Society of General Internal Medicine (SGIM). Both organizations sponsor mentorship programs, and regular attendance at regional and/or national meetings (followed by email correspondence) can greatly improve an academic hospitalist's career trajectory. Finally, midlevel and senior‐level hospitalists have to learn mentorship skills; mere experience in the field does not ensure acquisition of the necessary mentorship skills, anymore than experience in medicine ensures teaching skills. Mentorship is its own skill set, and receiving appropriate training via the SHM or SGIM national meetings or the Academic Hospitalist Academy (referenced below) is critical.
Defining Academic Expectations for Hospitalists
Harrison et al.1 note that academic hospitalists felt there was a lack of respect for the scholarly work that hospitalists do as part of their job, raising the proposition that the mentorship dearth for academic hospitalists might result from currently available mentors not knowing what to say. Even if mentors were plentiful today, we still must ask the question, What would the mentor advise the young hospitalist to do? The academic hospitalist offers extraordinary value to the Department, but in a way that is different from the standard R0RO‐1 Grant paradigm. Even if hospitalists acquire extramural funding, it will likely come from sources different from the National Institutes of Health (NIH): Agency for Healthcare Research and Quality (AHRQ), foundations (eg, The Robert Wood Johnson Foundation or The John A. Hartford Foundation), intramural hospital‐originating funding, etc. And while extramural funding may be a measure of a hospitalist's contribution to the Department, it should not be the only measure of the hospitalist's career development. There are 2 ways to get rich: acquire more money, or spend less money. Academic hospitalists, unlike other specialties in Medicine, are likely to fall into the latter category, by offering decreased hospital costs (ie, decreased length of stay, decreased never events, etc.). Further, hospitalists may save in opportunity costs: the hospitalist staffing a ward service is less costly than a subspecialist who could be performing procedures, or a basic science researcher who could be acquiring grants. The problem today is that there is no way to quantify this decreased loss, and having this sort of metric will greatly enable mentors to provide hospitalists with ways of showing value to the department outside of the standard NIH grant paradigm. The Quality Portfolio developed by the SGIM and the forthcoming Benchmarks for Academic Hospitalists Promotion white paper (as developed by the SHM's Academic Practice and Promotion Committee) will greatly improve the substance of mentorship for academic hospitalists.2 Leaders of academic hospital medicine must learn to educate chairs of medicine and medical school deans as to the value‐added services intrinsic in the integration of hospitalists into the academic environment.
Having an Academic Plan
Mentorship is a 2‐way relationship: the mentor has responsibilities, but so too, does the mentee. As we wait for the hospitalist field to further develop, new academic hospitalists must become proactive in seeking guidance in career development. The Academic Hospitalist Academy, cosponsored by SHM, SGIM, and ACLGIM, is an example of this type of training.3 As a part of this course, participants learn of the rules and the opportunities for success in academic hospital medicine. Success for academic hospitalist groups will likely follow from understanding what success looks like. The Academy provides an excellent program for distributing that knowledge.
Research Training in Hospital Medicine
Many traditionalists would insist that Hospital Medicine could evoke the same training paradigm as other subspecialties in medicine (ie, fellowships). Unfortunately there are not a sufficient number of GME‐funded positions to handle the number of hospitalists required to advance the mission of academic hospital medicine. Moreover, fellowship training for every academic hospitalist would be unlikely to produce the desired results of improving the delivery of inpatient care. The academic agenda for the hospitalist depends on understanding the hospital system, and then executing improvements that lead to safer, more efficient and effective care. In this way, the academic hospitalist academic training is much more akin to a Master of Business Administration (MBA) than it is to a Bachelor of Science (BS) degree: namely, via job immersion, the hospitalist develops a greater systems understanding that should inform his or her academic career. Thus, a fellowship right out of residency may not have the same urgency for the hospitalist as it does for the subspecialist. Nevertheless, those hospitalists seeking an academic scholarly career will experience major benefits from fellowship training. Academic hospitalists need not focus only on the few existing hospitalist fellowships; they can obtain the necessary training in research skills via a general medicine fellowship, of which there are many. For this cohort of hospitalists, we strongly encourage training in a general medicine, health services, or outcomes research fellowship, with an emphasis on research techniques as they apply to the measurement of quality, patient safety, and/or clinical education.
With respect to academic hospitalists, it is likely that nothing is as important as the question of mentorship. Even the hardest working hospitalist can lose their way without guidance and a roadmap; the mentor is central to both. But the lost opportunity is not borne by the individual physician alone; the academic department loses too. Because the hospitalist's value depends on sufficient familiarity with a specific system prior to leveraging improvements, the department accrues maximal benefits in efficiency and effectiveness only if it can maintain retention for at least 2 years.4 The turnover carries major costs; recruitment costs money, and every new hospitalist engenders major start‐up costs. Faculty members who become completely integrated into the department have higher retention rates than those who consider themselves outside the main stream. Mentorship will greatly increase the probability that hospitalists will progress and feel the importance to the department.
Academic hospital medicine must strive over the next 5 to 10 years to become totally integrated in the academic culture of every institution. This task will take great leadership both at the local level and at a national level. We agree with the authors that the SHM and the SGIM can both provide important assistance to young hospital medicine groups. We applaud the authors of this article for making explicit this next major challenge for the field.
- ,,,.Survey of US academic hospitalist leaders about mentorship and academic activities in hospitalist groups.J Hosp Med.2011;6:5–9.
- ,,,.Quality portfolio introduction – academic hospitalist taskforce quality portfolio rationale and development. Society of General Internal Medicine Website,2010. Available at:http://www.sgim.org/index.cfm?pageId=846. Accessed September 2010.
- The Academic Hospitalist Academy Website,2010. Available at:http://www.academichospitalist.org. Accessed September 2010.
- ,,, et al.Effects of physician experience on costs and outcomes on an academic general medicine service.Ann Intern Med.2002;137:866–874.
This issue of the Journal of Hospital Medicine features an important contribution concerning the current state of academic hospital medicine. The survey of 57 hospitalists revealed what many of us already suspected: the state of mentorship in academic hospitalist groups is unsatisfactory.1
While the conclusion is alarming, it is also not surprising. Over the past decade academic medical centers enthusiastically hired hospitalists to improve efficiency for inpatient services and to lessen the effect of Accreditation Council for Graduate Medical Education (ACGME) regulations on duty hours and patient caps. Few departments of internal medicine, however, hired academic hospitalists with the intent of creating academic divisions. Thus many institutions appear to view hospitalists primarily as hospital employees ignoring their potential academic contributions, and as a result it should not be a surprise that many hospitalist groups lack the mentorship infrastructure of other divisions within a typical Department of Medicine. Compounding the hospital employee problem, the new field of academic hospital medicine has emerged only in the last decade, a time frame that has resulted in very few hospitalists qualified to serve as senior mentors.
We cannot easily remove these limitations: the past is the past, and over time, hospital medicine will mature and develop more senior mentors. But what should we do until that maturation occurs? We believe that the academic work of hospitalists, both in education and research (Quality and Patient Safety) are important endeavors too valuable to be left to chance. With 30,000 hospitalists delivering care, it is critical that research in the optimal delivery of this care be performed, targeting systems improvements to enact anticipated outcomes in quality and patient safety. The physicians who are regularly and intimately involved in this system of inpatient care delivery, the hospitalists, are best suited for identifying the unique features of the inpatient care system needing improvement. Mentorship is essential in ensuring the advancement of both areas, and the sustainability of hospital medicine in medical academe. The article by Harrison et al.1 both establishes the depth of these issues and provides important insights into potential solutions for closing this mentorship gap while the field matures.
Utilizing Other Mentors
No measure of systems change will make young hospitalists immediately experienced, such that they have the sophistication to be senior mentors for younger hospitalists. But we can compensate for this temporary gap in mentorship experience. First, in the next 5 to 10 years, young academic hospitalists need explicit direction from those within Departments of Medicine who do have this mentorship experience, even if these mentors do not work in hospital medicine. Mentors within General Internal Medicine or the subspecialties can still provide the guidance and support to ensure that academic hospitalists are engaging in the appropriate endeavors toward promotion and intellectual growth. Second, academic hospitalists have to seek out mentorship from afar through their participation in the organizations primarily devoted to the academic welfare of hospitalists: The Society of Hospital Medicine (SHM) and the Society of General Internal Medicine (SGIM). Both organizations sponsor mentorship programs, and regular attendance at regional and/or national meetings (followed by email correspondence) can greatly improve an academic hospitalist's career trajectory. Finally, midlevel and senior‐level hospitalists have to learn mentorship skills; mere experience in the field does not ensure acquisition of the necessary mentorship skills, anymore than experience in medicine ensures teaching skills. Mentorship is its own skill set, and receiving appropriate training via the SHM or SGIM national meetings or the Academic Hospitalist Academy (referenced below) is critical.
Defining Academic Expectations for Hospitalists
Harrison et al.1 note that academic hospitalists felt there was a lack of respect for the scholarly work that hospitalists do as part of their job, raising the proposition that the mentorship dearth for academic hospitalists might result from currently available mentors not knowing what to say. Even if mentors were plentiful today, we still must ask the question, What would the mentor advise the young hospitalist to do? The academic hospitalist offers extraordinary value to the Department, but in a way that is different from the standard R0RO‐1 Grant paradigm. Even if hospitalists acquire extramural funding, it will likely come from sources different from the National Institutes of Health (NIH): Agency for Healthcare Research and Quality (AHRQ), foundations (eg, The Robert Wood Johnson Foundation or The John A. Hartford Foundation), intramural hospital‐originating funding, etc. And while extramural funding may be a measure of a hospitalist's contribution to the Department, it should not be the only measure of the hospitalist's career development. There are 2 ways to get rich: acquire more money, or spend less money. Academic hospitalists, unlike other specialties in Medicine, are likely to fall into the latter category, by offering decreased hospital costs (ie, decreased length of stay, decreased never events, etc.). Further, hospitalists may save in opportunity costs: the hospitalist staffing a ward service is less costly than a subspecialist who could be performing procedures, or a basic science researcher who could be acquiring grants. The problem today is that there is no way to quantify this decreased loss, and having this sort of metric will greatly enable mentors to provide hospitalists with ways of showing value to the department outside of the standard NIH grant paradigm. The Quality Portfolio developed by the SGIM and the forthcoming Benchmarks for Academic Hospitalists Promotion white paper (as developed by the SHM's Academic Practice and Promotion Committee) will greatly improve the substance of mentorship for academic hospitalists.2 Leaders of academic hospital medicine must learn to educate chairs of medicine and medical school deans as to the value‐added services intrinsic in the integration of hospitalists into the academic environment.
Having an Academic Plan
Mentorship is a 2‐way relationship: the mentor has responsibilities, but so too, does the mentee. As we wait for the hospitalist field to further develop, new academic hospitalists must become proactive in seeking guidance in career development. The Academic Hospitalist Academy, cosponsored by SHM, SGIM, and ACLGIM, is an example of this type of training.3 As a part of this course, participants learn of the rules and the opportunities for success in academic hospital medicine. Success for academic hospitalist groups will likely follow from understanding what success looks like. The Academy provides an excellent program for distributing that knowledge.
Research Training in Hospital Medicine
Many traditionalists would insist that Hospital Medicine could evoke the same training paradigm as other subspecialties in medicine (ie, fellowships). Unfortunately there are not a sufficient number of GME‐funded positions to handle the number of hospitalists required to advance the mission of academic hospital medicine. Moreover, fellowship training for every academic hospitalist would be unlikely to produce the desired results of improving the delivery of inpatient care. The academic agenda for the hospitalist depends on understanding the hospital system, and then executing improvements that lead to safer, more efficient and effective care. In this way, the academic hospitalist academic training is much more akin to a Master of Business Administration (MBA) than it is to a Bachelor of Science (BS) degree: namely, via job immersion, the hospitalist develops a greater systems understanding that should inform his or her academic career. Thus, a fellowship right out of residency may not have the same urgency for the hospitalist as it does for the subspecialist. Nevertheless, those hospitalists seeking an academic scholarly career will experience major benefits from fellowship training. Academic hospitalists need not focus only on the few existing hospitalist fellowships; they can obtain the necessary training in research skills via a general medicine fellowship, of which there are many. For this cohort of hospitalists, we strongly encourage training in a general medicine, health services, or outcomes research fellowship, with an emphasis on research techniques as they apply to the measurement of quality, patient safety, and/or clinical education.
With respect to academic hospitalists, it is likely that nothing is as important as the question of mentorship. Even the hardest working hospitalist can lose their way without guidance and a roadmap; the mentor is central to both. But the lost opportunity is not borne by the individual physician alone; the academic department loses too. Because the hospitalist's value depends on sufficient familiarity with a specific system prior to leveraging improvements, the department accrues maximal benefits in efficiency and effectiveness only if it can maintain retention for at least 2 years.4 The turnover carries major costs; recruitment costs money, and every new hospitalist engenders major start‐up costs. Faculty members who become completely integrated into the department have higher retention rates than those who consider themselves outside the main stream. Mentorship will greatly increase the probability that hospitalists will progress and feel the importance to the department.
Academic hospital medicine must strive over the next 5 to 10 years to become totally integrated in the academic culture of every institution. This task will take great leadership both at the local level and at a national level. We agree with the authors that the SHM and the SGIM can both provide important assistance to young hospital medicine groups. We applaud the authors of this article for making explicit this next major challenge for the field.
This issue of the Journal of Hospital Medicine features an important contribution concerning the current state of academic hospital medicine. The survey of 57 hospitalists revealed what many of us already suspected: the state of mentorship in academic hospitalist groups is unsatisfactory.1
While the conclusion is alarming, it is also not surprising. Over the past decade academic medical centers enthusiastically hired hospitalists to improve efficiency for inpatient services and to lessen the effect of Accreditation Council for Graduate Medical Education (ACGME) regulations on duty hours and patient caps. Few departments of internal medicine, however, hired academic hospitalists with the intent of creating academic divisions. Thus many institutions appear to view hospitalists primarily as hospital employees ignoring their potential academic contributions, and as a result it should not be a surprise that many hospitalist groups lack the mentorship infrastructure of other divisions within a typical Department of Medicine. Compounding the hospital employee problem, the new field of academic hospital medicine has emerged only in the last decade, a time frame that has resulted in very few hospitalists qualified to serve as senior mentors.
We cannot easily remove these limitations: the past is the past, and over time, hospital medicine will mature and develop more senior mentors. But what should we do until that maturation occurs? We believe that the academic work of hospitalists, both in education and research (Quality and Patient Safety) are important endeavors too valuable to be left to chance. With 30,000 hospitalists delivering care, it is critical that research in the optimal delivery of this care be performed, targeting systems improvements to enact anticipated outcomes in quality and patient safety. The physicians who are regularly and intimately involved in this system of inpatient care delivery, the hospitalists, are best suited for identifying the unique features of the inpatient care system needing improvement. Mentorship is essential in ensuring the advancement of both areas, and the sustainability of hospital medicine in medical academe. The article by Harrison et al.1 both establishes the depth of these issues and provides important insights into potential solutions for closing this mentorship gap while the field matures.
Utilizing Other Mentors
No measure of systems change will make young hospitalists immediately experienced, such that they have the sophistication to be senior mentors for younger hospitalists. But we can compensate for this temporary gap in mentorship experience. First, in the next 5 to 10 years, young academic hospitalists need explicit direction from those within Departments of Medicine who do have this mentorship experience, even if these mentors do not work in hospital medicine. Mentors within General Internal Medicine or the subspecialties can still provide the guidance and support to ensure that academic hospitalists are engaging in the appropriate endeavors toward promotion and intellectual growth. Second, academic hospitalists have to seek out mentorship from afar through their participation in the organizations primarily devoted to the academic welfare of hospitalists: The Society of Hospital Medicine (SHM) and the Society of General Internal Medicine (SGIM). Both organizations sponsor mentorship programs, and regular attendance at regional and/or national meetings (followed by email correspondence) can greatly improve an academic hospitalist's career trajectory. Finally, midlevel and senior‐level hospitalists have to learn mentorship skills; mere experience in the field does not ensure acquisition of the necessary mentorship skills, anymore than experience in medicine ensures teaching skills. Mentorship is its own skill set, and receiving appropriate training via the SHM or SGIM national meetings or the Academic Hospitalist Academy (referenced below) is critical.
Defining Academic Expectations for Hospitalists
Harrison et al.1 note that academic hospitalists felt there was a lack of respect for the scholarly work that hospitalists do as part of their job, raising the proposition that the mentorship dearth for academic hospitalists might result from currently available mentors not knowing what to say. Even if mentors were plentiful today, we still must ask the question, What would the mentor advise the young hospitalist to do? The academic hospitalist offers extraordinary value to the Department, but in a way that is different from the standard R0RO‐1 Grant paradigm. Even if hospitalists acquire extramural funding, it will likely come from sources different from the National Institutes of Health (NIH): Agency for Healthcare Research and Quality (AHRQ), foundations (eg, The Robert Wood Johnson Foundation or The John A. Hartford Foundation), intramural hospital‐originating funding, etc. And while extramural funding may be a measure of a hospitalist's contribution to the Department, it should not be the only measure of the hospitalist's career development. There are 2 ways to get rich: acquire more money, or spend less money. Academic hospitalists, unlike other specialties in Medicine, are likely to fall into the latter category, by offering decreased hospital costs (ie, decreased length of stay, decreased never events, etc.). Further, hospitalists may save in opportunity costs: the hospitalist staffing a ward service is less costly than a subspecialist who could be performing procedures, or a basic science researcher who could be acquiring grants. The problem today is that there is no way to quantify this decreased loss, and having this sort of metric will greatly enable mentors to provide hospitalists with ways of showing value to the department outside of the standard NIH grant paradigm. The Quality Portfolio developed by the SGIM and the forthcoming Benchmarks for Academic Hospitalists Promotion white paper (as developed by the SHM's Academic Practice and Promotion Committee) will greatly improve the substance of mentorship for academic hospitalists.2 Leaders of academic hospital medicine must learn to educate chairs of medicine and medical school deans as to the value‐added services intrinsic in the integration of hospitalists into the academic environment.
Having an Academic Plan
Mentorship is a 2‐way relationship: the mentor has responsibilities, but so too, does the mentee. As we wait for the hospitalist field to further develop, new academic hospitalists must become proactive in seeking guidance in career development. The Academic Hospitalist Academy, cosponsored by SHM, SGIM, and ACLGIM, is an example of this type of training.3 As a part of this course, participants learn of the rules and the opportunities for success in academic hospital medicine. Success for academic hospitalist groups will likely follow from understanding what success looks like. The Academy provides an excellent program for distributing that knowledge.
Research Training in Hospital Medicine
Many traditionalists would insist that Hospital Medicine could evoke the same training paradigm as other subspecialties in medicine (ie, fellowships). Unfortunately there are not a sufficient number of GME‐funded positions to handle the number of hospitalists required to advance the mission of academic hospital medicine. Moreover, fellowship training for every academic hospitalist would be unlikely to produce the desired results of improving the delivery of inpatient care. The academic agenda for the hospitalist depends on understanding the hospital system, and then executing improvements that lead to safer, more efficient and effective care. In this way, the academic hospitalist academic training is much more akin to a Master of Business Administration (MBA) than it is to a Bachelor of Science (BS) degree: namely, via job immersion, the hospitalist develops a greater systems understanding that should inform his or her academic career. Thus, a fellowship right out of residency may not have the same urgency for the hospitalist as it does for the subspecialist. Nevertheless, those hospitalists seeking an academic scholarly career will experience major benefits from fellowship training. Academic hospitalists need not focus only on the few existing hospitalist fellowships; they can obtain the necessary training in research skills via a general medicine fellowship, of which there are many. For this cohort of hospitalists, we strongly encourage training in a general medicine, health services, or outcomes research fellowship, with an emphasis on research techniques as they apply to the measurement of quality, patient safety, and/or clinical education.
With respect to academic hospitalists, it is likely that nothing is as important as the question of mentorship. Even the hardest working hospitalist can lose their way without guidance and a roadmap; the mentor is central to both. But the lost opportunity is not borne by the individual physician alone; the academic department loses too. Because the hospitalist's value depends on sufficient familiarity with a specific system prior to leveraging improvements, the department accrues maximal benefits in efficiency and effectiveness only if it can maintain retention for at least 2 years.4 The turnover carries major costs; recruitment costs money, and every new hospitalist engenders major start‐up costs. Faculty members who become completely integrated into the department have higher retention rates than those who consider themselves outside the main stream. Mentorship will greatly increase the probability that hospitalists will progress and feel the importance to the department.
Academic hospital medicine must strive over the next 5 to 10 years to become totally integrated in the academic culture of every institution. This task will take great leadership both at the local level and at a national level. We agree with the authors that the SHM and the SGIM can both provide important assistance to young hospital medicine groups. We applaud the authors of this article for making explicit this next major challenge for the field.
- ,,,.Survey of US academic hospitalist leaders about mentorship and academic activities in hospitalist groups.J Hosp Med.2011;6:5–9.
- ,,,.Quality portfolio introduction – academic hospitalist taskforce quality portfolio rationale and development. Society of General Internal Medicine Website,2010. Available at:http://www.sgim.org/index.cfm?pageId=846. Accessed September 2010.
- The Academic Hospitalist Academy Website,2010. Available at:http://www.academichospitalist.org. Accessed September 2010.
- ,,, et al.Effects of physician experience on costs and outcomes on an academic general medicine service.Ann Intern Med.2002;137:866–874.
- ,,,.Survey of US academic hospitalist leaders about mentorship and academic activities in hospitalist groups.J Hosp Med.2011;6:5–9.
- ,,,.Quality portfolio introduction – academic hospitalist taskforce quality portfolio rationale and development. Society of General Internal Medicine Website,2010. Available at:http://www.sgim.org/index.cfm?pageId=846. Accessed September 2010.
- The Academic Hospitalist Academy Website,2010. Available at:http://www.academichospitalist.org. Accessed September 2010.
- ,,, et al.Effects of physician experience on costs and outcomes on an academic general medicine service.Ann Intern Med.2002;137:866–874.
Rude Awakening
A hospitalist-turned-sleep specialist at Beth Israel Deaconess Medical Center (BIDMC) in Boston wants hospitalists to be aware of a recent report that showed 80.5% of hospitalized patients were at high risk for obstructive sleep apnea (OSA)—but she doesn't want you to lose any sleep over it.
Suzie Bertisch, MD, MPH, a physician in the Division of Pulmonary, Critical Care, and Sleep Medicine at BIDMC, says hospitalists should stay current with OSA research because of its growing prevalence. However, because there is little evidence-based data on just how much of a complicating factor the condition is, she doesn't suggest hospitalists be too concerned.
"It's the reality that we've all seen a patient who has sleep apnea," says Dr. Bertisch, an instructor at Harvard Medical School who helped lead a session on OSA at HM10. "It's important to keep an eye on it because it could become a bigger deal. We're not sure now what it is."
The study, the results of which were announced in November by Loyola University Health System in Maywood, Ill., was based on a one-day survey of 195 patients who were given an eight-question OSA screening questionnaire known as STOP-BANG. Patients who answered “yes” to three of the questions were considered high-risk.
Dr. Bertisch notes that since the general prevalence of OSA usually is cited at about 2% to 4% of the population (about 12 million Americans, according to the National Institutes of Health), the low threshold for determining risk via the STOP-BANG test likely contributed to the outsized percentage of those considered high-risk.
The researchers suggested the results could prod hospitals, and by extension hospitalists, to administer the STOP-BANG test to gauge risk.
Dr. Bertisch, who gave up hospitalist duties in recent months to focus on sleep research, says she expects more research to be conducted in order to help answer questions about how sleep apnea affects other health conditions. “It’s an emerging field,” she says.
A hospitalist-turned-sleep specialist at Beth Israel Deaconess Medical Center (BIDMC) in Boston wants hospitalists to be aware of a recent report that showed 80.5% of hospitalized patients were at high risk for obstructive sleep apnea (OSA)—but she doesn't want you to lose any sleep over it.
Suzie Bertisch, MD, MPH, a physician in the Division of Pulmonary, Critical Care, and Sleep Medicine at BIDMC, says hospitalists should stay current with OSA research because of its growing prevalence. However, because there is little evidence-based data on just how much of a complicating factor the condition is, she doesn't suggest hospitalists be too concerned.
"It's the reality that we've all seen a patient who has sleep apnea," says Dr. Bertisch, an instructor at Harvard Medical School who helped lead a session on OSA at HM10. "It's important to keep an eye on it because it could become a bigger deal. We're not sure now what it is."
The study, the results of which were announced in November by Loyola University Health System in Maywood, Ill., was based on a one-day survey of 195 patients who were given an eight-question OSA screening questionnaire known as STOP-BANG. Patients who answered “yes” to three of the questions were considered high-risk.
Dr. Bertisch notes that since the general prevalence of OSA usually is cited at about 2% to 4% of the population (about 12 million Americans, according to the National Institutes of Health), the low threshold for determining risk via the STOP-BANG test likely contributed to the outsized percentage of those considered high-risk.
The researchers suggested the results could prod hospitals, and by extension hospitalists, to administer the STOP-BANG test to gauge risk.
Dr. Bertisch, who gave up hospitalist duties in recent months to focus on sleep research, says she expects more research to be conducted in order to help answer questions about how sleep apnea affects other health conditions. “It’s an emerging field,” she says.
A hospitalist-turned-sleep specialist at Beth Israel Deaconess Medical Center (BIDMC) in Boston wants hospitalists to be aware of a recent report that showed 80.5% of hospitalized patients were at high risk for obstructive sleep apnea (OSA)—but she doesn't want you to lose any sleep over it.
Suzie Bertisch, MD, MPH, a physician in the Division of Pulmonary, Critical Care, and Sleep Medicine at BIDMC, says hospitalists should stay current with OSA research because of its growing prevalence. However, because there is little evidence-based data on just how much of a complicating factor the condition is, she doesn't suggest hospitalists be too concerned.
"It's the reality that we've all seen a patient who has sleep apnea," says Dr. Bertisch, an instructor at Harvard Medical School who helped lead a session on OSA at HM10. "It's important to keep an eye on it because it could become a bigger deal. We're not sure now what it is."
The study, the results of which were announced in November by Loyola University Health System in Maywood, Ill., was based on a one-day survey of 195 patients who were given an eight-question OSA screening questionnaire known as STOP-BANG. Patients who answered “yes” to three of the questions were considered high-risk.
Dr. Bertisch notes that since the general prevalence of OSA usually is cited at about 2% to 4% of the population (about 12 million Americans, according to the National Institutes of Health), the low threshold for determining risk via the STOP-BANG test likely contributed to the outsized percentage of those considered high-risk.
The researchers suggested the results could prod hospitals, and by extension hospitalists, to administer the STOP-BANG test to gauge risk.
Dr. Bertisch, who gave up hospitalist duties in recent months to focus on sleep research, says she expects more research to be conducted in order to help answer questions about how sleep apnea affects other health conditions. “It’s an emerging field,” she says.
Dr. Dermatologist, Meet the Hospitalist
Dermatology is about sleuthing and putting things together that don't go together, says Lindy Fox, MD, founder of the interdisciplinary dermatology hospitalist consultation service at the University of California at San Francisco (UCSF). She recommends hospitalists learn basic descriptions of dermatological conditions, categories, and terminology. That way, when they make a phone presentation of symptoms to a consulting dermatologist, the specialist can determine whether the patient needs an in-person consultation.
Differential diagnosis of a dermatological condition considers history, duration, timing, waxing, and waning, Dr. Fox told participants in the hands-on Hospitalist Mini-College at UCSF last October.
"We worry about pain much more than about itching. We worry about medications and drug eruptions. A family history is important, and a social history, especially for patients who travel. But the most important thing is morphology," she says. "What is the primary lesion? Learn to recognize the primary lesion and the differential diagnosis will follow."
It is a concern that fewer dermatologists have any presence in the hospital, and the resulting demands on hospitalists will only increase, Dr. Fox says. At UCSF, she and two colleagues consult on patients who have skin diseases that are severe enough to require hospitalization, or who develop a cutaneous manifestation of the disease for which they were admitted or as a consequence of treatment of that disease. They closely collaborate with UCSF's hospitist service, as well as teach residents and, at the bedside, internists. This approach, however, is rare, mainly limited to academic medical centers, she says.
"There are young dermatologists out there who want to stay in the hospital and work with internists but the structure often isn't there," Dr. Fox says. "What hospitalists can do is help to facilitate these relationships for dermatologists who want to work in the hospital."
Dermatology is about sleuthing and putting things together that don't go together, says Lindy Fox, MD, founder of the interdisciplinary dermatology hospitalist consultation service at the University of California at San Francisco (UCSF). She recommends hospitalists learn basic descriptions of dermatological conditions, categories, and terminology. That way, when they make a phone presentation of symptoms to a consulting dermatologist, the specialist can determine whether the patient needs an in-person consultation.
Differential diagnosis of a dermatological condition considers history, duration, timing, waxing, and waning, Dr. Fox told participants in the hands-on Hospitalist Mini-College at UCSF last October.
"We worry about pain much more than about itching. We worry about medications and drug eruptions. A family history is important, and a social history, especially for patients who travel. But the most important thing is morphology," she says. "What is the primary lesion? Learn to recognize the primary lesion and the differential diagnosis will follow."
It is a concern that fewer dermatologists have any presence in the hospital, and the resulting demands on hospitalists will only increase, Dr. Fox says. At UCSF, she and two colleagues consult on patients who have skin diseases that are severe enough to require hospitalization, or who develop a cutaneous manifestation of the disease for which they were admitted or as a consequence of treatment of that disease. They closely collaborate with UCSF's hospitist service, as well as teach residents and, at the bedside, internists. This approach, however, is rare, mainly limited to academic medical centers, she says.
"There are young dermatologists out there who want to stay in the hospital and work with internists but the structure often isn't there," Dr. Fox says. "What hospitalists can do is help to facilitate these relationships for dermatologists who want to work in the hospital."
Dermatology is about sleuthing and putting things together that don't go together, says Lindy Fox, MD, founder of the interdisciplinary dermatology hospitalist consultation service at the University of California at San Francisco (UCSF). She recommends hospitalists learn basic descriptions of dermatological conditions, categories, and terminology. That way, when they make a phone presentation of symptoms to a consulting dermatologist, the specialist can determine whether the patient needs an in-person consultation.
Differential diagnosis of a dermatological condition considers history, duration, timing, waxing, and waning, Dr. Fox told participants in the hands-on Hospitalist Mini-College at UCSF last October.
"We worry about pain much more than about itching. We worry about medications and drug eruptions. A family history is important, and a social history, especially for patients who travel. But the most important thing is morphology," she says. "What is the primary lesion? Learn to recognize the primary lesion and the differential diagnosis will follow."
It is a concern that fewer dermatologists have any presence in the hospital, and the resulting demands on hospitalists will only increase, Dr. Fox says. At UCSF, she and two colleagues consult on patients who have skin diseases that are severe enough to require hospitalization, or who develop a cutaneous manifestation of the disease for which they were admitted or as a consequence of treatment of that disease. They closely collaborate with UCSF's hospitist service, as well as teach residents and, at the bedside, internists. This approach, however, is rare, mainly limited to academic medical centers, she says.
"There are young dermatologists out there who want to stay in the hospital and work with internists but the structure often isn't there," Dr. Fox says. "What hospitalists can do is help to facilitate these relationships for dermatologists who want to work in the hospital."
Antimicrobial Stewardship
Nosocomial, or hospital‐acquired, infections (HAIs) are a major cause of patient morbidity and mortality in the United States and other countries.15 In 2002, approximately 1.7 million HAIs occurred in US hospitals and were associated with an estimated 98,987 deaths.1 Of particular note, increasing percentages of HAIs are now caused by antimicrobial‐resistant pathogens, which have been linked with increases in morbidity, mortality, length of hospital stay, and healthcare costs.6
The 2004 data summary from the United States National Nosocomial Infections Surveillance (NNIS) System Report highlighted substantial increases for year 2003 versus 1998 through 2002 in vancomycin‐resistant enterococci (VRE); methicillin‐resistant Staphylococcus aureus; Klebsiella pneumoniae resistant to third‐generation cephalosporins; and Pseudomonas aeruginosa resistant to imipenem, quinolones, or third‐generation cephalosporins.7 Other gram‐negative bacteria of concern include Escherichia coli and Acinetobacter baumannii, as well as Enterobacter cloacae and E. aerogenes.8, 9
The increasing number of multidrug‐resistant (MDR) gram‐negative bacteria within the healthcare setting is particularly concerning.1013 Too frequently, clinicians in the United States now encounter gram‐negative bacteria species that are resistant to many, and occasionally all, currently available antibiotics. For many of these MDR gram‐negative pathogens the antimicrobials that potentially remain active (eg, aminoglycosides and polymyxins) are often more toxic and less efficacious for some infections.14 Particularly problematic is that the pharmaceutical industry's developmental pipeline for new antibiotics, with novel mechanisms of action that might be used against MDR gram‐negative pathogens, has virtually come to a standstill.15, 16 Even if an investigational drug was in phase 2 or 3 trials right now or entered the US Food and Drug Administration (FDA) Fast Track Development Program, it would be at least 10 or 15 years before that drug would be available on the US market.
What this means is that the clinician's current antibiotic armamentarium is all they can expect in the foreseeable future. It also means that special care needs to be taken to optimally use currently available agents to ensure continued activity against the pathogens encountered in the hospital (and community) setting, now and in the future. Maximizing clinical outcomes, while minimizing the emergence and spread of antimicrobial resistance (and other adverse effects associated with suboptimal antimicrobial drug use), falls under the purview of antimicrobial stewardship, the focus of this paper.
Antimicrobial StewardshipWhy Is It Needed, What Is It, and What Are Its Goals?
Inappropriate Antimicrobial Use
Early in the onset of many infections, the data needed to make a rational, informed decision about specific antibiotic therapy are usually unavailable. For many infections, therapy cannot be delayed waiting for microbiology or other findings, and broad‐spectrum empiric therapy is begun on the basis of educated guesses made from the patient's presentation and characteristics, and local or hospital antibiograms. In addition, for many serious infections, delay in antimicrobial therapy will increase patient morbidity and mortality. Generally, what occurs is the decision to treat empirically with one or more broad‐spectrum antibiotic agents, which are then continued for the entire course of therapy. Opportunities are often missed to tailor therapy later in the course of infection when microbiologic or other data are available. There is also a tendency for spiraling empiricism to occur when a patient is not doing well with initial therapy; additional agents with broad antimicrobial activity, including antifungals and antivirals, are added to the therapeutic regimen, often in a haphazard way.17
Besides the perceived need to prescribe broad‐spectrum and/or multiple antibiotics to cover possible or perceived resistant or uncommon pathogens, a number of other factors contribute to inappropriate antibiotic or antimicrobial use. Many times antimicrobials are initiated when no infection exists, such as for asymptomatic bacteruria, noninfectious pulmonary conditions, or endotracheal tube or Foley catheter colonization. Another example of inappropriate use is treating for longer than needed to eradicate infection. All of these events intensify the exposure of bacteria colonizing or infecting the patient to multiple anti‐infective drugs and increase the chances for selection of an MDR pathogen.
Examining antibiotic usage at the hospital level, approximately 60% of adult patients admitted to US hospitals receive at least 1 dose of an antibiotic agent during their stay (range: 44%74% for individual hospitals).18, 19 Similarly, at Wake Forest University Baptist Medical Center (WFUBMC), approximately 75% of inpatients receive antimicrobials at some point during their hospitalization (Ohl, unpublished data, 2007). One recent example by Hecker and colleagues conducted in a 650‐bed, university‐affiliated US hospital reported 30% of the total days of antibiotic therapy received by adult non‐ICU inpatients was unnecessary.20 The most common reasons for unnecessary use were administration for longer than recommended durations, administration for a noninfectious or nonbacterial syndrome, and treatment of colonizing or contaminating microorganisms.
Consequences of antibiotic misuse
Unwanted consequences of antimicrobial therapy include increased morbidity and mortality, adverse drug reactions, increased length of hospital stay and hospitalization costs, predisposition to secondary infections, and emergence and selection of drug‐resistant organisms.21, 22 Selection or induction of antimicrobial resistance and promotion of secondary infection with Clostridium difficileparticularly with new, more toxigenic strains23are of particular concern in the current hospital environment.22 These untoward consequences can be seen as a calculated risk of antibiotic therapy for any single‐treated patient, or as an undesired outcome measure for excessive use at the level of the healthcare institution. For example, a 7‐day course of a third‐generation cephalosporin in a particular patient increases the risk of subsequent infection from an extended‐spectrum beta‐lactamase (ESBL)‐producing gram‐negative rod. For the institution as a whole, excessive use of this antimicrobial will increase the overall prevalence and number of infections due to this troublesome resistance factor.
Definition and Goals of Antimicrobial Stewardship
The above studies show a clear need for improved, more careful and prudent use of antimicrobials, which is key to antimicrobial stewardship. Building on the definition given by the Infectious Diseases Society of America and Society for Healthcare Epidemiology of America (IDSA/SHEA),24 antimicrobial stewardship is essentially a system of personnel, informatics, data collection, and policy/procedures that promotes the optimal selection, dosing, and duration of therapy for antimicrobial agents throughout the course of their use. An effective antimicrobial stewardship program will limit inappropriate and excessive antimicrobial use, but more importantly improve and optimize therapy for the individual infected patient.
The goals of antimicrobial stewardship are listed in Table 1.24, 25 It is important to recognize that the primary goals of antimicrobial stewardship are not the reduction of healthcare costsand certainly not the reduction of drug acquisition or usage costs. As the 2007 IDSA/SHEA guidelines for institutional development of an antimicrobial stewardship program make clear, the primary goal is to focus on patient care; that is, to optimize clinical outcomes, while minimizing unintended consequences of antimicrobial use (emergence of resistance, selection of pathogenic organisms, and adverse drug reactions).24
| Prevent or slow the emergence of antimicrobial resistance |
| Optimize selection, dose and duration of therapy |
| Reduce adverse drug events, including secondary infection (eg, C. difficile antibiotic‐associated diarrhea) |
| Reduce morbidity and mortality |
| Reduce length of stay |
| Reduce healthcare expenditures |
Reduced healthcare costs without an adverse effect on quality of patient care is, however, a legitimate secondary goal of antimicrobial stewardship, and will result from optimized clinical outcomes and decreased potential collateral damage associated with pharmacotherapy. Unfortunately, it is much more difficult to measure the impact of an antimicrobial stewardship program on emergence of resistance than on drug acquisition or usage costs. As a consequence, reduction in drug acquisition/usage costs has too often been viewed as the primary (and sometimes only) justification for implementing an antimicrobial stewardship program.26
Finally, the role of effective infection control cannot be overemphasized. Infection control is clearly necessary and often sufficient to reduce HAIs. However, a comprehensive infection control program, combined with an effective antimicrobial stewardship agenda, synergistically limit the emergence and spread of antimicrobial‐resistant bacteria, reduce HAIs, control resistance, and improve overall inpatient care.24, 27 Hence, when instituting an antimicrobial stewardship program, it is essential to ensure the hospital or other healthcare institution already has a robust hospital epidemiology and infection control program in placeor to simultaneously institute one.
Constructing an Antimicrobial Stewardship Program
Infectious Diseases Society of America and Society for Healthcare Epidemiology of America Guidelines
Whereas the value of antimicrobial stewardship is widely appreciated, actually taking the steps to set up a healthcare facility program can be daunting. The guidelines established by the IDSA/SHEA for developing an institutional program represent a valuable resource and suggest that the best programs are comprehensivetaking into account local antimicrobial use and resistance patterns, as well as available resources.24 The size and nature of the institution can make a big difference in determining what program to set up and what elements it should entail; what works at one institution might not work as well at another. The program components and effectiveness of each will differ for community versus academic medical centers. A comprehensive program includes active monitoring, fostering of appropriate antimicrobial use, and collaboration with an effective infection control program as well as other hospital entities. The role of a multidisciplinary team, with administrative support, is particularly underscored in the guidelines. According to the guidelines, core members of the multidisciplinary team should include an infectious diseases physician and a clinical pharmacist with infectious diseases training. It should also ideally include a clinical microbiologist, information system specialist, infection control professional, and hospital epidemiologist.24 It is important that all members of the team are passionate about the program, oversee its implementation and daily functions, and have some sense of ownership of it. Compensation for its primary participants is crucial. Compensation not only ensures that adequate time is available for executing the daily activities of the program, but it also helps impart a greater sense of program ownership. Process and outcome measures of the program (discussed below) should be included in the performance evaluations of the compensated key participants.
Although the guidelines indicate that an infectious diseases physician should act as the program leader, this might not always be feasible or necessary. Many of the hospitals most in need of improved antimicrobial stewardship simply do not have an infectious diseases physician available to them. In addition, a lot of community hospitals share their infectious diseases physician on a consultative basis with other medical centers and facilities, and that particular specialist may not have a lot of time to invest in the program. Where having an infectious diseases physician as a core member and leader of the team is beneficial, it is not absolutely necessary. A similar argument can be made concerning the inclusion of a clinical pharmacist with infectious diseases training as a core member. Not all hospitals have or can find a clinical pharmacist with formal infectious diseases training through a didactic pharmacy residency program.
If an infectious diseases physician or clinical pharmacist with formal infectious diseases training is not available at a given institution, the team will need to include others ready to assume a greater leadership role. Although not mentioned in the guidelines, hospital medicine specialists and hospitalists are well‐suited to take on this role and can be integral to leadership of the multidisciplinary team. Hospitalists have knowledge of the hospital where they support a wide range of services and, at least in some cases, may have fewer time constraints than a subspecialty. In addition, hospital leadership and administration more often reach out to hospitalists to oversee patient quality care and safety improvement projects, the realm to which antimicrobial stewardship belongs. Regarding clinical pharmacists, an alternative to formal residency training for PharmDs are online certification programs such as MAD‐ID (Making a Difference in Infectious Diseases Pharmacotherapy), the Society of Infectious Diseases Pharmacists, or via a limited number of state medical societies.28, 29 Such certification programs should increase the number of pharmacists and PharmDs with infectious diseases training in the near future.
Antimicrobial stewardship is best considered a medical staff, rather than primary hospital, function. Individuals from the medical staff, and particularly medical staff leadership, are most adept in employing the 3 Cs that are important when constructing, implementing, and operating an institutional antimicrobial stewardship programconceptualization, communication, and coercion. Conceptualization deals with understanding what needs to be done, why it needs to be done, and how to do it, whereas communication is making sure the providers of antimicrobials receive and understand this information. Coercion might seem like a strong term, but it refers to the pressure exerted by thought leaders and others involved in the process to get things done within the institution, including all units or departments. Although ultimate responsibility for an antimicrobial stewardship program should probably lie with the medical staff, the IDSA/SHEA guidelines correctly indicate that support and collaboration of hospital administration, medical staff leadership, and local providers are essential to the success of any such program.24
A Case Study: the Wake Forest University Baptist Medical Center (WFUBMC) Program
Figure 1A,B provides an overview of the general structure of the antimicrobial stewardship program at WFUBMC implemented in 2000. To establish and provide the information needed for day‐to‐day operations of a stewardship program at WFUBMC, data are needed on how, where, and by whom antibiotics are used within the institution. In addition, microbiology data, including the frequency and susceptibility of infecting pathogens, are essential. Obtaining these data often requires the help and cooperation of the information technology (IT) staff at the institution. Considerable time and effort may be required at the outset, but once information system programs are established, ongoing data mining is much easier. At the time of program initiation, it was decided to assess antibiotic use or density (amount of drug per inpatient geographic unit) using the defined‐daily‐dose (DDD) method. This entails assigning a predetermined weight of administered antibiotic as a dose and dividing by a denominator of 1000 patient days. Subsequently, days of therapy per drug has been found to be a more accurate measure of antimicrobial consumption. When developing a program, it is vital to first obtain baseline usage data. Such data should include, if possible, a detailed inventory of usage within different units of the hospital or for particular services, or sometimes even for a particular provider with a history of high antimicrobial usage. Ongoing measurement over time allows the impact of new stewardship interventions and guidelines to be measured, as well as identifying potential new problem areas in usage.
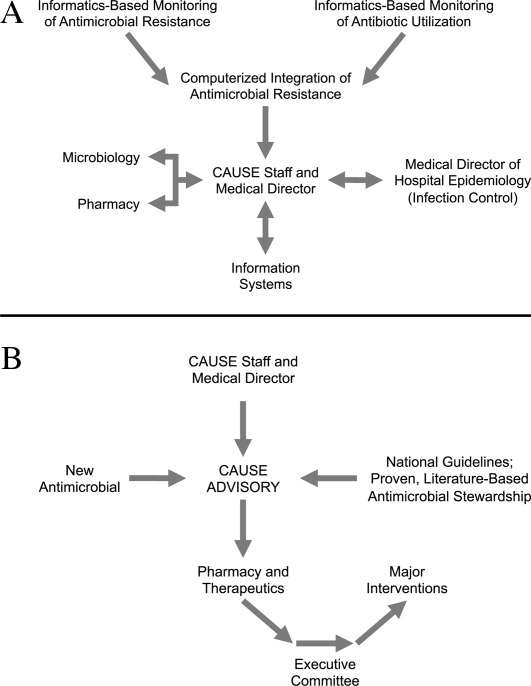
Good microbiology data are also essential to determine problem pathogens at the institution and where they are located. Such data are useful not only to define areas of resistance (potentially warranting changes in antimicrobial policies to alter selection pressures), but also for gathering information necessary for defining local guidelines for antimicrobial use. For example, if the local antibiograms show that a particular pathogen in the hospital ICU has a particular resistance pattern, then initial empiric therapy for patients at risk of infection with those organisms should be chosen to cover the problematic resistant pathogen. Once subsequent microbiology data become available, patients not infected with the pathogen can be de‐escalated to a more narrow‐spectrum antibiotic.
As illustrated in Figure 1A, at WFUBMC, all the collected data are integrated to provide information concerning antibiotic density, usage, and patterns of antimicrobial resistance. This information is received by the staff of the antimicrobial stewardship program, which at WFUBMC is called the Center for Antimicrobial Utilization, Stewardship and Epidemiology (CAUSE). The CAUSE staff works with the day‐to‐day elements of program administration and operations and includes 2 infectious diseases physicians and 2 infectious disease PharmDs. The CAUSE staff works very closely with the microbiology laboratory, hospital pharmacy, and the medical director of hospital epidemiology and infection control.
The CAUSE program at WFUBMC administratively functions through an advisory board committee that includes thought leaders from different medical specialties and patient units of the hospitalparticularly those with high antimicrobial usage, such as hematology/oncology, pulmonary, critical care, and transplantation. The CAUSE staff and advisory board exist to exchange ideas concerning what is working or not working and where problem areas may be, and to propose possible changes in antimicrobial practices at the institution. In addition, thought leaders on the advisory board also receive and evaluate information from various sources about new antimicrobial agents and national guidelines, and, in turn, help disseminate this information to the hospital personnel who will be involved in program implementation.
At WFUBMC, it is the advisory board committee, working in conjunction with the CAUSE staff/medical director, that presents antimicrobials for formulary consideration to the Pharmacy and Therapeutics (P&T) Committee, in addition to any major interventions CAUSE and its advisory board feel are indicated. The P&T committee then reports to the medical staff executive committee and hospital leadership. As should be evident, the approach to stewardship at WFUBMC is medical staff‐driven, rather than a function of administrative constituents.
Finally, no matter how well‐organized an antimicrobial stewardship program is, it will not be fully successful if the entire medical staff does not buy into the process and agree with the need for the proposed changes and interventions involving the practice of antimicrobial therapy. It is important to spend some time early in program development to ensure that the need for an antimicrobial stewardship program, the process, and the outcomes (both in terms of patient care and clinical outcomes at the institution) are clearly communicated to the medical staff, and that their full commitment and cooperation are enlisted. In cases where hospital‐wide infection or resistance rates are known and antimicrobial utilization data are available, it is important to present such information in an understandable and convincing manner that makes the case for a proposed change or intervention, not only at the hospital level but also at the level of the patient.
Elements of a Successful Program: Basic Strategies
Potential strategies or elements of an antimicrobial stewardship program are listed in Table 2. Two evidence‐based fundamental or core strategies have been recommended by the IDSA/SHEA guidelines24 and implemented at numerous institutions with various levels of success. The first is a so‐called back‐end approach to modifying antimicrobial therapy on the basis of prospective audit of antimicrobial use, with intervention and feedback to the provider. The second is a front‐end approach using formulary restriction and preauthorization requirements for specific antimicrobial agents. Various supplemental strategies, including large group and patient case‐based education, guidelines and clinical pathways, antimicrobial order forms, and computerized clinical decision support, are also recommended.
| Program Element | Advantages | Disadvantages | Comments |
|---|---|---|---|
| Prospective Audit and Feedback | Proven in clinical studies to reduce and modify antimicrobial consumption, improve selected clinical outcomes, and decrease antimicrobial expenditures Provides one‐on‐one patient‐centered education to the clinician Allows optimization of anti‐infective pharmacology | Adherence to stewardship interventions by the clinician is voluntary Resource intensive Requires a greater amount of team member training and experience in anti‐infective therapy | Back‐end approach Identify and intervene on patients already started on antimicrobials Interventions include changing, streamlining, de‐escalation, pharmacodynamic/dose optimization, IV to PO switch, and limitation of duration of therapy |
| Restriction or Preauthorization | Proven in clinical studies to reduce and modify antimicrobial consumption, improve selected clinical outcomes, and decrease antimicrobial expenditures Together with infection control effective in controlling outbreaks of resistant or secondary pathogens (such as C. diff) | Less appealing to clinicians Loss of prescriber autonomy Potential need for after‐hours service Time intensive Potential for delay in antimicrobial administration | Front‐end approach Formulary restriction or contact a stewardship team member to obtain authorization to prescribe a selected antimicrobial Each intervention is a mini‐consult |
| Large Group Education | Can reach a large number of prescribers in a short period of time Effective for communicating the need and rationale for subsequent stewardship interventions | Not particularly effective in changing prescribing behavior without other interventions Rapid extinction of gained knowledge | Grand rounds or clinical staff meeting venues Provides information to prescribers and thought leader clinicians on justification for stewardship Feedback antimicrobial susceptibility and use data to clinicians |
| Guidelines and Pathways | Limits variation in therapy of infectious diseases Best evidence‐based Assists in adherence with regulatory and third‐party payer stipulations | Often not utilized unless combined with other stewardship strategies or elements | Best if local data and conditions are used to adapt guidelines to a specific institution |
| Computerized Physician Order Entry and Clinical Decision Support | Shown in limited clinical studies to reduce and modify antimicrobial consumption, improve selected clinical outcomes, and decrease antimicrobial expenditures Once established can greatly assist with implementation of guidelines and best‐evidence therapy. Reduces adverse events related to antimicrobials | Resource intensive during design and implementation Expensive Not readily available | Often entails modification of existing or purchasing of additional informatics |
| Microbiology Interventions | Potential to improve antimicrobial use and anti‐infective therapy for the individual patient | Not well studied | Includes cascade reporting to hide antimicrobial susceptibilities that might promote suboptimal therapy (eg, fluoroquinolone susceptibility for invasive S. aureus) Assistance with choices of automated susceptibility profile, communication of new or changes in testing protocols Preauthorization of susceptibility testing for unconventional antibiotics |
| Rapid Diagnostics | Provides opportunity for early targeted therapy Assists with de‐escalation Shown in very limited studies to decrease antimicrobial consumption and improve clinical outcomes | Not readily available Expensive | Includes PCR and antigen testing of clinical specimens or early culture growth with rapid turnaround of test results |
| Antimicrobial Cycling | Potential to decrease antimicrobial resistance for an institution or geographic unit | Not consistently shown in clinical trials to improve clinical outcomes or decrease resistance Often increases antimicrobial consumption Extremely labor intensive to ensure adherence | Changing antimicrobial protocols periodically in an attempt to reduce selection pressure for resistance |
Prospective Audit With Intervention and Feedback
This approach usually involves the use of an antimicrobial support team that reviews initial or ongoing therapy and then intervenes to provide feedback and suggested modifications to the medical care provider to improve therapy. This can be done by an infectious diseases physician, a clinical pharmacist, or a hospitalist or internist with expertise in antimicrobial therapy. The aim is to provide patient‐specific education and/or suggest changes to antimicrobial utilization (when needed) to improve and streamline therapy. Suggested interventions could include discontinuing or changing 1 or more drugs, switching intravenous to oral drug administration, and suggesting a short‐course duration of therapy. Occasionally, suggestions are made when appropriate to actually escalate or intensify therapy to increase efficacy. Identification of patients for targeting or focusing prospective audit and feedback efforts typically involves using computer surveillance to single out problem antimicrobials or problematic usage, given local resistance patterns or patient characteristics.24 Examples could include a focus on asymptomatic bacteruria, excessive duration of therapy for ventilator‐associated pneumonia, or overzealous use of certain classes of antimicrobials. Another potential activity for a prospective audit and feedback team is to review reports of patient‐specific blood and sterile body fluid culture results matched to the patient's current antimicrobial therapy. This allows for daily review of the appropriateness of therapy for potentially serious infections. Some patients seen by the antibiotic support team may be referred for infectious diseases or other expert consultative opinion if their infections or therapy are felt to be too complicated for routine prospective audit and feedback recommendations.
A number of studies have demonstrated that strategies involving prospective audit with intervention and feedback can improve antibiotic stewardship, as measured by reductions in inappropriate antibiotic use,30 reduced antibiotic costs or overall consumption,3135 greater compliance with hospital treatment guidelines or policies33, 36, 37 and, in some cases, reduced number of infections due to C difficile infection32, 37 or resistant pathogens.31, 32, 37 Prospective audit with feedback is probably the best and most effective core strategy for a community hospital program where other interventions are cumbersome or not as well tolerated by the medical staff. One potential disadvantage of the prospective audit with intervention and feedback approach is that medical provider adherence is largely voluntary. The team can make suggestions, but if the provider disagrees or is unobtainable, the suggestion is never implemented. Also, this strategy can also be resource‐intensive from a personnel perspective.
Formulary Restriction and Preauthorization
The other major strategy used to achieve antimicrobial stewardship goals involves antimicrobial restriction. This can be accomplished either by not including the particular antimicrobial agent on the hospital formulary or by requiring the medical provider to obtain preauthorization before prescribing a restricted drug. A pager system or telephone call is often used for preauthorization, whereby the clinician wishing to prescribe a particular agent calls or pages a member of the stewardship team in order to obtain prescribing permission. When using preauthorization, it is important that the individuals who receive the calls actually see patients and have clinical experience and the respect of the medical staff, as each call may be a mini‐consult. Oftentimes, the provider or prescriber making the call is asking for suggestions as to what antimicrobial might be used, and not simply to obtain authorization to use a drug that is otherwise restricted. Studies have shown that effective interventions supporting stewardship are better provided by attending infectious diseases staff or clinical pharmacists, rather than persons in training.38
Regarding the identification of antimicrobials for restriction, a program should preferentially choose those drugs that are involved in therapy for complex patients and infections. It is also a reasonable approach for drugs that are, or have the potential to be, overused for certain infections where alternatives exist. For work‐horse antimicrobials, those drugs overused or misused for several different infectious diseases, prospective audit and feedback is arguably a better strategy to reduce and modulate consumption.
Formulary restriction and preauthorization is clearly effective in modulating antimicrobial use. A large number of studies have demonstrated reductions in antibiotic drug use, and often in cost, after hospital implementation of a formulary restriction or preauthorization approach to antimicrobial stewardship.3947 It has been more difficult to demonstrate other benefits associated with this approach, although there is some support for its aid in controlling nosocomial infection outbreaks. Restriction of clindamycin48 (or clindamycin, cefotaxime, and vancomycin27) has been shown to control outbreaks of nosocomial C difficileassociated diarrhea and VRE, respectively. More recently, Internet‐based antimicrobial restriction programs49, 50 and a computerized (electronic) approval system51 have been demonstrated to reduce antibiotic use at tertiary hospitals.
Some studies have reported increased antibiotic drug susceptibilities after implementation of institutional preauthorization policies,45, 46, 51 and at least 1 reported a decreased incidence of ceftazidime‐resistant Klebsiella species after instituting a preapproval policy for cephalosporins.52 However, there is concern that restricting 1 class of antibiotics and replacing it with another will simply replace 1 resistant species with another, the so‐called squeezing the balloon effect.53 This was observed in the latter study, where a 44% reduction in ceftazidime‐resistant Klebsiella species at the hospital was accompanied by a 69% increase in incidence of imipenem‐resistant P aeruginosa.52 To assess and enable response to possible squeezing the balloon effects, the guidelines recommend monitoring overall trends in antimicrobial use for institutions using preauthorization strategies.
Possible disadvantages of preauthorization and restriction include perceived loss of autonomy for prescribers, the potential need for all‐hours support, inaccurate or misleading information from the prescriber (leading to inappropriate recommendations),24 and significant delay in stat antimicrobial administration.54 Delay in antimicrobial administration due to the time required to obtain preauthorization and have the approval communicated to the pharmacy was not observed when studied as a process measure at WFUBMC (Ohl, unpublished data, 2008).
A study by Linkin and colleagues showed that 39% of telephone calls for preauthorization of a restricted antimicrobial contained an inaccuracy in at least 1 type of patient data.38 A follow‐up by the same group demonstrated that inaccurate communication was significantly associated with inappropriate antimicrobial recommendations (odds ratio [OR] 2.2; P = .03); this was particularly the case for inaccuracies in microbiologic data (OR 7.5; P = .002).55 Also, if all‐hours support is not provided, at least 1 study has shown some physicians may engage in stealth dosing, that is, avoiding having to obtain preauthorization for restricted antimicrobials by waiting until off‐hours to place orders.56 The latter can be dealt with by following up on such orders with a prospective audit and feedback component of the program. Preauthorization is usually more difficult to employ and less accepted in non‐academic medical centers. Prospective audit and feedback may be more appropriate in such settings.
Supplemental Strategies
A number of additional options are available to supplement the 2 core strategies just described, and are listed in Table 2. Education is generally considered an essential component of any effective antimicrobial stewardship program, but it generally has little lasting impact on providers' behavior, unless it is incorporated with other active interventions.24 In particular, the large group or Grand Roundstype education, where someone describes what needs to be done and why, typically does not produce lasting behavioral changes. There might be, and often is, some short‐term modification, but long‐lasting change at the provider level requires consistent and repeated educational endeavors. Such large group educational venues are more effective and better used as a forum to describe or garner support for an impending stewardship program or intervention, rather than to teach a specific practice.
Using the antimicrobial stewardship program to adapt national guidelines to local antimicrobial use, microbiology, and resistance patterns57, 58 or using clinical (critical) pathways59 has also been shown to improve antimicrobial utilization at hospitals. National guidelines generally enjoy widespread support, but they commonly lack specific information about how to implement recommendations at a given hospital or how to incorporate local data relevant for decision making. A 2006 report by Beardsley and coworkers provides a model from WFUBMC on how local microbiologic data can be used to modify national treatment guidelines to better serve the needs of patients treated at a particular institution.60 Using American Thoracic Society (ATS) and IDSA guidelines for the management of hospital‐acquired pneumonia (HAP), together with local data on the most common bacterial pathogens and their susceptibility to piperacillin‐tazobactam, cefepime, ciprofloxacin, and amikacin (based on length of hospitalization), the WFUBMC CAUSE Advisory Board developed institution‐specific HAP guidelines. The new guidelines divided the ATS/IDSA late onset/risk of the MDR pathogens group of patients into 2 subcategories, early‐late and late‐late pneumonias. Also, unlike the national guidelines, the new guidelines did not recommend ciprofloxacin as empiric therapy, instead recommending amikacin as a component of regimens targeting late‐late pneumonias.
Newer (and in some cases not so new) information technologies can be adapted to healthcare delivery and prescriber support to improve antimicrobial stewardship. These include computer decision support61 and alert systems6265; computerized physician order entry (CPOE)66, 67; electronic medical records24; electronic retrieval of treatment guidelines or clinical texts68; and personal digital assistant (PDA) applications providing information on pathogens, diagnosis, medication, and treatment.68, 69 In addition, computer‐based surveillance64, 70, 71 and Web‐based systems for antimicrobial approval; automated clinical decision support; and/or enhanced real‐time communication between prescribers and other members of the antimicrobial stewardship team show promise for antimicrobial stewardship programs.49, 50
Computer‐assisted decision support has been shown to improve or reduce antibiotic‐susceptibility mismatches (improve selection of effective therapy), overall antibiotic use, excess antimicrobial dosages, excessive‐dose days, selection of antimicrobials for which the patient was poorly matched in terms of allergies, and antimicrobial‐related adverse events, as well as reduce antimicrobial drug costs, total hospital costs, and length of hospital stay.7277 For their part, CPOE systems have been shown to improve compliance with treatment guidelines, decrease medication and other medical errors, shorten length of hospital stay, and decrease pharmaceutical costs.66, 67, 78 In many cases, CPOE systems can now be modified to include some clinical decision support to improve antimicrobial use.78
The IDSA/SHEA guidelines note that antimicrobial decisions can be improved through use of CPOE, clinical decision support, and electronic medical records that enable incorporation of data on patient‐specific microbiology cultures and susceptibilities, hepatic and renal function, drug interactions, allergies, and cost. They also point out that computer‐based surveillance can facilitate good stewardship by enabling more efficient targeting of antimicrobial interventions, tracking of antimicrobial resistance patterns, and identification of HAIs and adverse drug events.24 Recently, a few proprietary informatics programs that perform such functions for the hospital epidemiologist and antimicrobial steward have become available, including but not limited to TheraDoc (Salt Lake City, UT), SafetySurveillor (Premier, Inc., Charlotte, NC), and BD Protect (BD Diagnostics, Austin, TX). Perhaps one of the best‐known comprehensive hospital information systems that incorporates and integrates several information technologies to improve patient care at the level of the prescriber is the Health Evaluation through Logical Processing (HELP) system at LDS Hospital in Salt Lake City, Utah.7981 Unfortunately, these programs are expensive, need considerable time for installation and validation, and do not always perform the functions needed by the medical center. The medical community has generally been slow to incorporate healthcare information technology to improve antimicrobial use or general medical care, but in the last few years more hospitals are finding their merit.
On the basis of evidence currently available, the 2007 guidelines do not recommend the routine use of antimicrobial cycling or combination therapy to prevent or reduce antimicrobial resistance. Such strategies, where at first glance might intuitively seem to make sense, have not been shown to improve patient care, improve antimicrobial choices, or reduce antimicrobial resistance. In addition, antimicrobial cycling in particular is difficult to implement and labor intensive to oversee.24
One strategy for improving antimicrobial stewardship not mentioned in the 2007 IDSA/SHEA guidelines, but might become increasingly important in the future, is the use of rapid molecular diagnostic testing. Knowing the identity of the causative pathogen sooner or being able to rapidly rule out certain pathogens should enable better decision‐making. During the 2009/2010 influenza season with H1N1 influenza, WFUBMC was able to implement rapid viral testing and learned some things that enabled improvement of hospital practices. It was found that approximately 10% to 15% of the pneumonias in immunocompromised patients at the center were not bacterial but viral, the pathogens being respiratory syncytial virus (RSV) or metapneumovirus (Ohl, unpublished data, 2010). Upon finding a viral etiology to a lower respiratory tract infection, rapid de‐escalation of antibiotic therapy was possible. If rapid diagnostics are to be performed, it is important that there are systems in place to respond quickly to the findings, so the benefits of having rapid data can be realized.
Evaluating Antimicrobial Stewardship Programs
Two general types of measures are used to evaluate the effectiveness of antimicrobial stewardship: process and outcome. As with most things done in the hospital, process measures are easier. They measure surrogate impacts of a program, accountability, resource use, and cost effectiveness. In essence, process measures evaluate whether the program accomplished what it set out to do in terms of changing certain processes or prescriber behaviors. It is important to measure resource use, as this helps to continue funding and to keep workers involved in the project. Good programs will save money; this can easily be measured, even if it is just as simple as going to the hospital pharmacy and looking at the cost of antimicrobials provided per patient day.
Outcomes like decreases in particular infections, less emergence of antimicrobial resistance, or other patient‐specific measures are likely more important in the big picture, but they are also much more difficult to measure. For example, where one would like to measure changes in pathogen resistance after making some changes in antimicrobial stewardship, it often takes years before the benefits of a particular intervention or change materialize in terms of less resistance or reduced emergence of resistance. If that type of change is to be measured, then one needs to be persistent and continue measurements over a long period of time. In addition, given the protracted amount of time before these outcomes may be observed, a number of other changes are likely to happen that coincide with the antimicrobial stewardship interventions and make assessment of causality difficult and biased.
Having said that, a number of studies have demonstrated a relationship between antibiotic restriction48, 8285 or other antimicrobial stewardship policies32, 86 and decreases in nosocomial C difficile infections or disease. Figure 2 illustrates the impact of a nonrestrictive antimicrobial stewardship program at a secondary/tertiary‐care hospital in Quebec, Canada, on an epidemic of C difficileassociated disease (CDAD) that occurred at the institution during the latter portion of 2003.86 Following program implementation, and the major drop in targeted antibiotic consumption, the incidence of CDAD also significantly decreased. Earlier implementation of infection control measures had no effect on CDAD incidence.
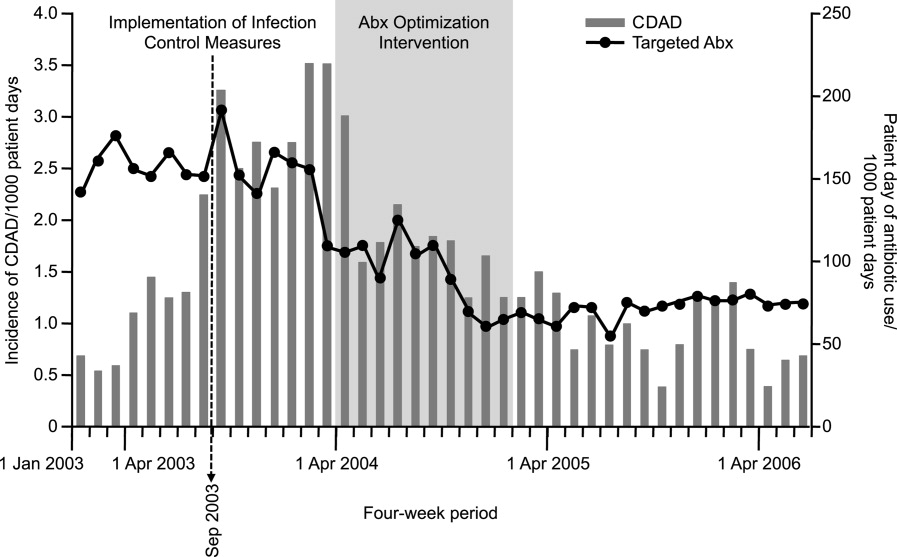
A smaller number of studies have reported decreases in resistant gram‐negative bacteria following implementation of antimicrobial stewardship programs. For example, Meyer and colleagues reported a marked reduction in ceftazidime‐resistant K pneumoniae at a 487‐bed general hospital in New York City after implementation of enhanced ceftazidime restriction and barrier precautions following an outbreak of infections caused by the resistant K pneumoniae.87 Similarly, Carling and coworkers reported a significant decrease in nosocomial infections caused by resistant Enterobacteriaceae following implementation of a multidisciplinary antibiotic stewardship program to minimize inappropriate use of third‐generation cephalosporins (Figure 3).32 More recently, a retrospective, longitudinal, multicenter analysis of a consortium of 22 academic health centers in the United States showed that incidence rates of carbapenem‐resistant P aeruginosa were lower at hospitals that restricted carbapenems than those that did not (P = .01).88
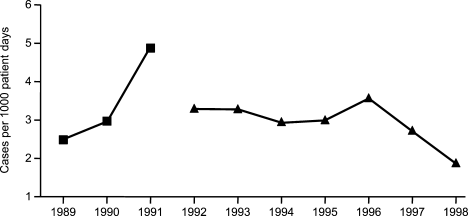
Evidence suggesting a beneficial impact of antimicrobial stewardship programs on resistance in gram‐positive organisms is limited. More specifically, the study by Carling and colleagues reported an apparent decrease in VRE rates following implementation of their program to reduce inappropriate use of third‐generation cephalosporins.32 The hospital had VRE rates similar to other NNIS System hospitals prior to beginning the program, but after antibiotic stewardship measures were implemented, the VRE rate began to drop, falling to 6% by 1999. This should be compared with a VRE rate of 24% for similar NNIS System hospitals in 1999.
As far as reducing healthcare costs, Figure 4A illustrates the direct antimicrobial cost savings at WFUBMC after implementation of the CAUSE antimicrobial stewardship program, and Figure 4B after supplemental interventions were implemented. Although decreasing antimicrobial cost is important, one would like to show decreases in overall healthcare costs associated with an antimicrobial stewardship program. Unfortunately, this is often a little more difficult to demonstrate. Extrapolations, however, may be possible. Because antimicrobial resistance, adverse drug effects, and secondary unintended infections such as C difficile colitis have been linked with increased patient morbidity and mortality, longer hospital stays, and increased healthcare costs,6, 89, 90 improved antimicrobial stewardship is expected to optimize patient care and lower overall healthcare costs. A study in a large tertiary care academic medical center estimated more than $4.25 million in total healthcare savings over 1 year with a stewardship program using both preauthorization and, to a lesser extent, prospective audit and feedback.91 Despite the fact cost saving should not be a primary goal of an antimicrobial stewardship program, lower antimicrobial costs can help keep a program funded and buttress a proposal for an antimicrobial stewardship program to hospital leadership.
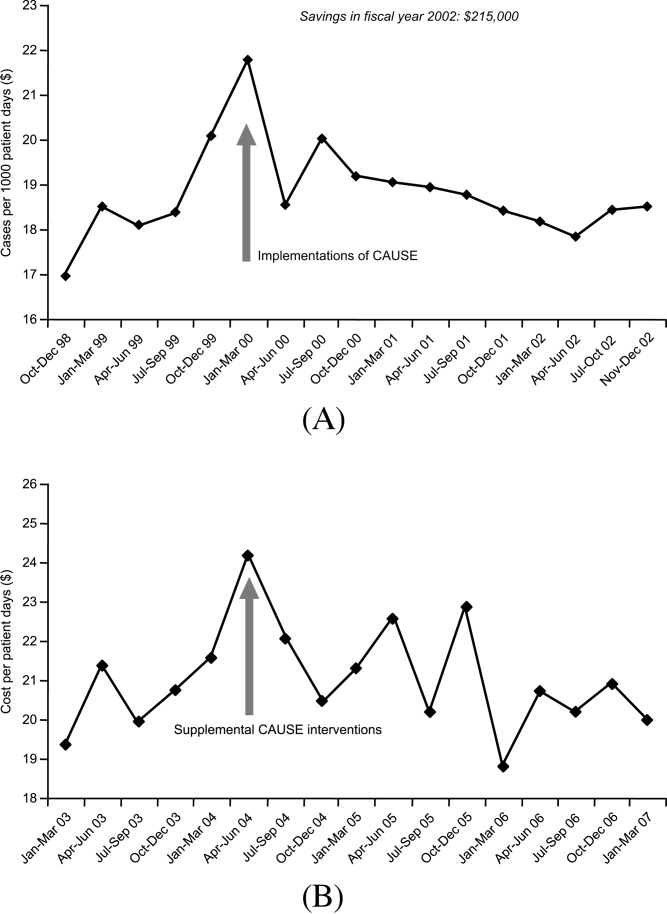
Many hospitals recognize other areas where an antimicrobial stewardship program can demonstrate its value. This includes implementation of a rapid change in drug utilization during antimicrobial supply shortages and assistance with regulatory mandates and surgical infection prophylaxis. Stewardship teams often assist microbiology with protocols for microbiology reporting, antibiograms, planning for susceptibility testing, and communicating changes in microbiology tests or protocols to clinicians.
Conclusions
Overuse or misuse of antibiotics and other antimicrobials for hospital inpatients is relatively common, and can be associated with several unintended negative consequences. Improving medical care necessarily includes better use of antimicrobials to optimize outcomes and preserve the effectiveness of currently available agents. Further, an important additional consequence of effective antimicrobial stewardship and improved patient care is typically a lowering of overall healthcare costs. The recent 2007 IDSA/SHEA guidelines provide recommendations for developing an institutional program to enhance antimicrobial stewardship. However, individual institutions need to look closely at their own systems and patients to develop an antimicrobial stewardship program that best serves the needs of their hospital and the people it serves.
- ,, et al.Estimating health care‐associated infections and deaths in U.S. hospitals, 2002.Public Health Rep.2007;122:160–166.
- ,,.Healthcare‐associated infection in Shiraz, Iran 2004–2005.J Hosp Infect.2008;69:283–287.
- ,,,,.Healthcare‐associated infections in Finnish acute care hospitals: a national prevalence survey, 2005.J Hosp Infect.2008;69:288–294.
- .Historical and changing epidemiology of healthcare‐associated infections.J Hosp Infect.2009;73:296–304.
- ,,, et al.Four country healthcare associated infection prevalence survey 2006: overview of the results.J Hosp Infect.2008;69:230–248.
- ,,.Clinical and economic burden of antimicrobial resistance.Expert Rev Anti Infect Ther.2008;6:751–763.
- National Nosocomial Infections Surveillance (NNIS) System Report, data summary from January 1992 through June 2004, issued October 2004.Am J Infect Control.2004;32:470–485.
- .Bad bugs, no drugs: no ESCAPE revisited.Clin Infect Dis.2009;49:992–993.
- .Federal funding for the study of antimicrobial resistance in nosocomial pathogens: no ESKAPE.J Infect Dis.2008;197:1079–1081.
- ,.Resistant gram‐negative bacilli: a neglected healthcare crisis?Am J Health Syst Pharm.2007;64:S3–S21; quizS22–S24.
- ,,, et al.Antimicrobial resistance among gram‐negative bacilli causing infections in intensive care unit patients in the United States between 1993 and 2004.J Clin Microbiol.2007;45:3352–3359.
- ,,.The emerging threat of multidrug‐resistant gram‐negative organisms in long‐term care facilities.J Gerontol A Biol Sci Med Sci.2009;64:138–141.
- ,,,,.Influx of multidrug‐resistant, gram‐negative bacteria in the hospital setting and the role of elderly patients with bacterial bloodstream infection.Infect Control Hosp Epidemiol.2009;30:325–331.
- ,,.Emergence of extensively drug‐resistant and pandrug‐resistant Gram‐negative bacilli in Europe.Euro Surveill.2008;13(47)pii:19045.
- ,,, et al.The epidemic of antibiotic‐resistant infections: a call to action for the medical community from the Infectious Diseases Society of America.Clin Infect Dis.2008;46:155–164.
- ,,,,,.Bad bugs need drugs: an update on the development pipeline from the Antimicrobial Availability Task Force of the Infectious Diseases Society of America.Clin Infect Dis.2006;42:657–668.
- ,.Observations on spiraling empiricism: its causes, allure, and perils, with particular reference to antibiotic therapy.Am J Med.1989;87:201–206.
- ,.Variability in rates of use of antibacterials among 130 US hospitals and risk‐adjustment models for interhospital comparison.Infect Control Hosp Epidemiol.2008;29:203–211.
- ,,,.Trends in antibacterial use in US academic health centers: 2002 to 2006.Arch Intern Med.2008;168:2254–2260.
- ,,,,.Unnecessary use of antimicrobials in hospitalized patients: current patterns of misuse with an emphasis on the antianaerobic spectrum of activity.Arch Intern Med.2003;163:972–978.
- Polk RE, Fishman NO, eds.Antimicrobial Stewardship.7th ed.Philadelphia, PA:Churchill Livingstone Elsevier;2010. Mandell GL, Bennett JE, Dolin R, eds. Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases; No. 1.
- .Collateral damage and what the future might hold. The need to balance prudent antibiotic utilization and stewardship with effective patient management.Int J Infect Dis.2006;10:S17–S24.
- ,,, et al.A predominantly clonal multi‐institutional outbreak of Clostridium difficile‐associated diarrhea with high morbidity and mortality.N Engl J Med.2005;353:2442–2449.
- ,, et al.Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship.Clin Infect Dis.2007;44:159–177.
- .Antimicrobial stewardship.Semin Infect Contr.2001;1:210–221.
- ,,,,.Insights from the Society of Infectious Diseases Pharmacists on antimicrobial stewardship guidelines from the Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America.Pharmacotherapy.2009;29:593–607.
- ,,,,,.Manipulation of a hospital antimicrobial formulary to control an outbreak of vancomycin‐resistant enterococci.Clin Infect Dis.1996;23:1020–1025.
- MAD‐ID: Making a Difference in Infectious Diseases Pharmacotherapy. Available at http://www.mad‐id.com. Accessed August 17,2010.
- Society of Infectious Diseases Pharmacists. The implementation of antimicrobial stewardship using a multidisciplinary approach. CME program. Available at http://www.esymposia.ashp.org/cemantimicrobial/. Accessed August 17,2010.
- ,,, et al.Academic detailing to improve use of broad‐spectrum antibiotics at an academic medical center.Arch Intern Med.2001;161:1897–1902.
- ,,, et al.A hospitalwide intervention program to optimize the quality of antibiotic use: impact on prescribing practice, antibiotic consumption, cost savings, and bacterial resistance.Clin Infect Dis.2003;37:180–186.
- ,,,,.Favorable impact of a multidisciplinary antibiotic management program conducted during 7 years.Infect Control Hosp Epidemiol.2003;24:699–706.
- ,,, et al.Antimicrobial stewardship program directed at broad‐spectrum intravenous antibiotics prescription in a tertiary hospital.Eur J Clin Microbiol Infect Dis.2009;28:1447–1456.
- ,,,,,.Antibiotic optimization: an evaluation of patient safety and economic outcomes.Arch Intern Med.1997;157:1689–1694.
- .Concurrent antibiotic review programs: a role for infectious diseases specialists at small community hospitals.Clin Infect Dis.2003;37:742–743.
- ,,,,.Improving antimicrobial use in the hospital setting by providing usage feedback to prescribing physicians.Infect Control Hosp Epidemiol.2006;27:378–382.
- ,,, et al.Successful use of feedback to improve antibiotic prescribing and reduce Clostridium difficile infection: a controlled interrupted time series.J Antimicrob Chemother.2007;59:990–995.
- ,,,,.Inaccurate communications in telephone calls to an antimicrobial stewardship program.Infect Control Hosp Epidemiol.2006;27:688–694.
- ,,,,.Effect of a vancomycin restriction policy on ordering practices during an outbreak of vancomycin‐resistant Enterococcus faecium.Arch Intern Med.1997;157:1132–1136.
- ,.Impact of voluntary vs enforced compliance of third‐generation cephalosporin use in a teaching hospital.Arch Intern Med.1992;152:554–557.
- ,,.Cost containment through restriction of cephalosporins.Am J Hosp Pharm.1981;38:1897–1900.
- ,.Controlling cephalosporin and aminoglycoside costs through pharmacy and therapeutics committee restrictions.Am J Hosp Pharm.1985;42:1343–1347.
- ,.Enforcing a policy for restricting antimicrobial drug use.Am J Health Syst Pharm.1995;52:1433–1435.
- ,,,,,.Changes in antibiotic use, cost and consumption after an antibiotic restriction policy applied by infectious disease specialists.Jpn J Infect Dis.2005;58:338–343.
- ,,,,,.Impact of an antibiotic restriction policy on hospital expenditures and bacterial susceptibilities: a lesson from a pediatric institution in a developing country.Pediatr Infect Dis J.2000;19:200–206.
- ,,,,,.Effects of requiring prior authorization for selected antimicrobials: expenditures, susceptibilities, and clinical outcomes.Clin Infect Dis.1997;25:230–239.
- ,,,.Antibiotic cost savings from formulary restrictions and physician monitoring in a medical‐school‐affiliated hospital.Am J Med.1987;83:817–823.
- ,,,,.Decrease in nosocomial Clostridium difficile‐associated diarrhea by restricting clindamycin use.Ann Intern Med.1994;120:272–277.
- ,,, et al.A World Wide Web‐based antimicrobial stewardship program improves efficiency, communication, and user satisfaction and reduces cost in a tertiary care pediatric medical center.Clin Infect Dis.2008;47:747–753.
- ,,, et al.Impact of a web‐based antimicrobial approval system on broad‐spectrum cephalosporin use at a teaching hospital.Med J Aust.2003;178:386–390.
- ,,, et al.Electronic antibiotic stewardship: reduced consumption of broad‐spectrum antibiotics using a computerized antimicrobial approval system in a hospital setting.J Antimicrob Chemother.2008;62:608–616.
- ,,, et al.Class restriction of cephalosporin use to control total cephalosporin resistance in nosocomial Klebsiella.JAMA.1998;280:1233–1237.
- .Antibiotic resistance: squeezing the balloon?JAMA.1998;280:1270–1271.
- ,,.Impact of a restrictive antimicrobial policy on the process and timing of antimicrobial administration.J Hosp Med.2010;5:E41–E45.
- ,,, et al.Effect of communication errors during calls to an antimicrobial stewardship program.Infect Control Hosp Epidemiol.2007;28:1374–1381.
- ,,,,,.Evaluation of antimicrobial therapy orders circumventing an antimicrobial stewardship program: investigating the strategy of “stealth dosing”.Infect Control Hosp Epidemiol.2007;28:551–556.
- ,,,,,.Experience with a clinical guideline for the treatment of ventilator‐associated pneumonia.Crit Care Med.2001;29:1109–1115.
- ,,,,.Short‐course empiric antibiotic therapy for patients with pulmonary infiltrates in the intensive care unit: a proposed solution for indiscriminate antibiotic prescription.Am J Respir Crit Care Med.2000;162:505–511.
- ,,,,,.A controlled trial of a critical pathway for treatment of community‐acquired pneumonia. CAPITAL Study Investigators. Community‐Acquired Pneumonia Intervention Trial Assessing Levofloxacin.JAMA.2000;283:749–755.
- ,,,,,.Using local microbiologic data to develop institution‐specific guidelines for the treatment of hospital‐acquired pneumonia.Chest.2006;130:787–793.
- .Expert clinical decision support systems to enhance antimicrobial stewardship programs: insights from the Society of Infectious Diseases Pharmacists.Pharmacotherapy.2005;25:1116–1125.
- ,,,,,.Improved perioperative antibiotic use and reduced surgical wound infections through use of computer decision analysis.Infect Control Hosp Epidemiol.1989;10:316–320.
- ,,,,.Impact of a computer‐generated alert system prompting review of antibiotic use in hospitals.J Antimicrob Chemother.2009;63:1058–1063.
- ,,,,.Therapeutic antibiotic monitoring: surveillance using a computerized expert system.Am J Med.1990;88:43–48.
- ,,,,.Improvement of intraoperative antibiotic prophylaxis in prolonged cardiac surgery by automated alerts in the operating room.Infect Control Hosp Epidemiol.2003;24:13–16.
- ,.Computer physician order entry: benefits, costs, and issues.Ann Intern Med.2003;139:31–39.
- .Computerized physician order entry in the critical care and general inpatient setting: a narrative review.J Crit Care.2004;19:271–278.
- ,,.Information technology for optimizing the management of infectious diseases.Am J Health Syst Pharm.2006;63:957–965.
- ,,.Personal digital assistant infectious diseases applications for health care professionals.Clin Infect Dis.2003;36:1018–1029.
- ,,,.Computerized surveillance of adverse drug events in hospital patients.JAMA.1991;266:2847–2851.
- ,,, et al.Computer surveillance of hospital‐acquired infections and antibiotic use.JAMA.1986;256:1007–1011.
- ,,,,.Improving empiric antibiotic selection using computer decision support.Arch Intern Med.1994;154:878–884.
- ,,,.Evaluation of a computer‐assisted antibiotic‐dose monitor.Ann Pharmacother.1999;33:1026–1031.
- ,,, et al.A computer‐assisted management program for antibiotics and other antiinfective agents.N Engl J Med.1998;338:232–238.
- ,,, et al.Impact of a computerized clinical decision support system on reducing inappropriate antimicrobial use: a randomized controlled trial.J Am Med Inform Assoc.2006;13:378–384.
- ,,,.Development and impact of a computerized pediatric antiinfective decision support program.Pediatrics.2001;108:E75.
- ,,,,.Computerized antimicrobial decision support: an offline evaluation of a database‐driven empiric antimicrobial guidance program in hospitalized patients with a bloodstream infection.Int J Med Inform.2004;73:455–460.
- ,,, et al.The impact of computerized physician order entry on medication error prevention.J Am Med Inform Assoc.1999;6:313–321.
- .Maximizing appropriate antibiotic prophylaxis for surgical patients: an update from LDS Hospital, Salt Lake City.Clin Infect Dis.2001;33Suppl 2:S78–S83.
- ,,,,.The HELP system and its application to infection control.J Hosp Infect.1991;18Suppl A:424–431.
- ,,.Decision support in medicine: lessons from the HELP system.Int J Med Inform.2003;69:273–284.
- ,,,,,.Hospital‐wide restriction of clindamycin: effect on the incidence of Clostridium difficile‐associated diarrhea and cost.Ann Intern Med.1998;128:989–995.
- ,,,,,.An antibiotic policy associated with reduced risk of Clostridium difficile‐associated diarrhoea.Age Ageing.1999;28:578–580.
- ,,, et al.Successful control of Clostridium difficile infection in an elderly care unit through use of a restrictive antibiotic policy.J Antimicrob Chemother.1997;40:707–711.
- ,,,,,.Antibiotic prescribing policy and Clostridium difficile diarrhoea.QJM.2004;97:423–429.
- ,,,,.Impact of a reduction in the use of high‐risk antibiotics on the course of an epidemic of Clostridium difficile‐associated disease caused by the hypervirulent NAP1/027 strain.Clin Infect Dis.2007;45Suppl 2:S112–S121.
- ,,,,.Nosocomial outbreak of Klebsiella infection resistant to late‐generation cephalosporins.Ann Intern Med.1993;119:353–358.
- ,,.Relationship of carbapenem restriction in 22 university teaching hospitals to carbapenem use and carbapenem‐resistant Pseudomonas aeruginosa.Antimicrob Agents Chemother.2009;53:1983–1986.
- .The relationship between antimicrobial resistance and patient outcomes: mortality, length of hospital stay, and health care costs.Clin Infect Dis.2006;42Suppl 2:S82–S89.
- ,,, et al.Hospital and societal costs of antimicrobial‐resistant infections in a Chicago teaching hospital: implications for antibiotic stewardship.Clin Infect Dis.2009;49:1175–1184.
- .Antimicrobial stewardship.Am J Med.2006;119:S53–S61; discussionS62–S70.
Nosocomial, or hospital‐acquired, infections (HAIs) are a major cause of patient morbidity and mortality in the United States and other countries.15 In 2002, approximately 1.7 million HAIs occurred in US hospitals and were associated with an estimated 98,987 deaths.1 Of particular note, increasing percentages of HAIs are now caused by antimicrobial‐resistant pathogens, which have been linked with increases in morbidity, mortality, length of hospital stay, and healthcare costs.6
The 2004 data summary from the United States National Nosocomial Infections Surveillance (NNIS) System Report highlighted substantial increases for year 2003 versus 1998 through 2002 in vancomycin‐resistant enterococci (VRE); methicillin‐resistant Staphylococcus aureus; Klebsiella pneumoniae resistant to third‐generation cephalosporins; and Pseudomonas aeruginosa resistant to imipenem, quinolones, or third‐generation cephalosporins.7 Other gram‐negative bacteria of concern include Escherichia coli and Acinetobacter baumannii, as well as Enterobacter cloacae and E. aerogenes.8, 9
The increasing number of multidrug‐resistant (MDR) gram‐negative bacteria within the healthcare setting is particularly concerning.1013 Too frequently, clinicians in the United States now encounter gram‐negative bacteria species that are resistant to many, and occasionally all, currently available antibiotics. For many of these MDR gram‐negative pathogens the antimicrobials that potentially remain active (eg, aminoglycosides and polymyxins) are often more toxic and less efficacious for some infections.14 Particularly problematic is that the pharmaceutical industry's developmental pipeline for new antibiotics, with novel mechanisms of action that might be used against MDR gram‐negative pathogens, has virtually come to a standstill.15, 16 Even if an investigational drug was in phase 2 or 3 trials right now or entered the US Food and Drug Administration (FDA) Fast Track Development Program, it would be at least 10 or 15 years before that drug would be available on the US market.
What this means is that the clinician's current antibiotic armamentarium is all they can expect in the foreseeable future. It also means that special care needs to be taken to optimally use currently available agents to ensure continued activity against the pathogens encountered in the hospital (and community) setting, now and in the future. Maximizing clinical outcomes, while minimizing the emergence and spread of antimicrobial resistance (and other adverse effects associated with suboptimal antimicrobial drug use), falls under the purview of antimicrobial stewardship, the focus of this paper.
Antimicrobial StewardshipWhy Is It Needed, What Is It, and What Are Its Goals?
Inappropriate Antimicrobial Use
Early in the onset of many infections, the data needed to make a rational, informed decision about specific antibiotic therapy are usually unavailable. For many infections, therapy cannot be delayed waiting for microbiology or other findings, and broad‐spectrum empiric therapy is begun on the basis of educated guesses made from the patient's presentation and characteristics, and local or hospital antibiograms. In addition, for many serious infections, delay in antimicrobial therapy will increase patient morbidity and mortality. Generally, what occurs is the decision to treat empirically with one or more broad‐spectrum antibiotic agents, which are then continued for the entire course of therapy. Opportunities are often missed to tailor therapy later in the course of infection when microbiologic or other data are available. There is also a tendency for spiraling empiricism to occur when a patient is not doing well with initial therapy; additional agents with broad antimicrobial activity, including antifungals and antivirals, are added to the therapeutic regimen, often in a haphazard way.17
Besides the perceived need to prescribe broad‐spectrum and/or multiple antibiotics to cover possible or perceived resistant or uncommon pathogens, a number of other factors contribute to inappropriate antibiotic or antimicrobial use. Many times antimicrobials are initiated when no infection exists, such as for asymptomatic bacteruria, noninfectious pulmonary conditions, or endotracheal tube or Foley catheter colonization. Another example of inappropriate use is treating for longer than needed to eradicate infection. All of these events intensify the exposure of bacteria colonizing or infecting the patient to multiple anti‐infective drugs and increase the chances for selection of an MDR pathogen.
Examining antibiotic usage at the hospital level, approximately 60% of adult patients admitted to US hospitals receive at least 1 dose of an antibiotic agent during their stay (range: 44%74% for individual hospitals).18, 19 Similarly, at Wake Forest University Baptist Medical Center (WFUBMC), approximately 75% of inpatients receive antimicrobials at some point during their hospitalization (Ohl, unpublished data, 2007). One recent example by Hecker and colleagues conducted in a 650‐bed, university‐affiliated US hospital reported 30% of the total days of antibiotic therapy received by adult non‐ICU inpatients was unnecessary.20 The most common reasons for unnecessary use were administration for longer than recommended durations, administration for a noninfectious or nonbacterial syndrome, and treatment of colonizing or contaminating microorganisms.
Consequences of antibiotic misuse
Unwanted consequences of antimicrobial therapy include increased morbidity and mortality, adverse drug reactions, increased length of hospital stay and hospitalization costs, predisposition to secondary infections, and emergence and selection of drug‐resistant organisms.21, 22 Selection or induction of antimicrobial resistance and promotion of secondary infection with Clostridium difficileparticularly with new, more toxigenic strains23are of particular concern in the current hospital environment.22 These untoward consequences can be seen as a calculated risk of antibiotic therapy for any single‐treated patient, or as an undesired outcome measure for excessive use at the level of the healthcare institution. For example, a 7‐day course of a third‐generation cephalosporin in a particular patient increases the risk of subsequent infection from an extended‐spectrum beta‐lactamase (ESBL)‐producing gram‐negative rod. For the institution as a whole, excessive use of this antimicrobial will increase the overall prevalence and number of infections due to this troublesome resistance factor.
Definition and Goals of Antimicrobial Stewardship
The above studies show a clear need for improved, more careful and prudent use of antimicrobials, which is key to antimicrobial stewardship. Building on the definition given by the Infectious Diseases Society of America and Society for Healthcare Epidemiology of America (IDSA/SHEA),24 antimicrobial stewardship is essentially a system of personnel, informatics, data collection, and policy/procedures that promotes the optimal selection, dosing, and duration of therapy for antimicrobial agents throughout the course of their use. An effective antimicrobial stewardship program will limit inappropriate and excessive antimicrobial use, but more importantly improve and optimize therapy for the individual infected patient.
The goals of antimicrobial stewardship are listed in Table 1.24, 25 It is important to recognize that the primary goals of antimicrobial stewardship are not the reduction of healthcare costsand certainly not the reduction of drug acquisition or usage costs. As the 2007 IDSA/SHEA guidelines for institutional development of an antimicrobial stewardship program make clear, the primary goal is to focus on patient care; that is, to optimize clinical outcomes, while minimizing unintended consequences of antimicrobial use (emergence of resistance, selection of pathogenic organisms, and adverse drug reactions).24
| Prevent or slow the emergence of antimicrobial resistance |
| Optimize selection, dose and duration of therapy |
| Reduce adverse drug events, including secondary infection (eg, C. difficile antibiotic‐associated diarrhea) |
| Reduce morbidity and mortality |
| Reduce length of stay |
| Reduce healthcare expenditures |
Reduced healthcare costs without an adverse effect on quality of patient care is, however, a legitimate secondary goal of antimicrobial stewardship, and will result from optimized clinical outcomes and decreased potential collateral damage associated with pharmacotherapy. Unfortunately, it is much more difficult to measure the impact of an antimicrobial stewardship program on emergence of resistance than on drug acquisition or usage costs. As a consequence, reduction in drug acquisition/usage costs has too often been viewed as the primary (and sometimes only) justification for implementing an antimicrobial stewardship program.26
Finally, the role of effective infection control cannot be overemphasized. Infection control is clearly necessary and often sufficient to reduce HAIs. However, a comprehensive infection control program, combined with an effective antimicrobial stewardship agenda, synergistically limit the emergence and spread of antimicrobial‐resistant bacteria, reduce HAIs, control resistance, and improve overall inpatient care.24, 27 Hence, when instituting an antimicrobial stewardship program, it is essential to ensure the hospital or other healthcare institution already has a robust hospital epidemiology and infection control program in placeor to simultaneously institute one.
Constructing an Antimicrobial Stewardship Program
Infectious Diseases Society of America and Society for Healthcare Epidemiology of America Guidelines
Whereas the value of antimicrobial stewardship is widely appreciated, actually taking the steps to set up a healthcare facility program can be daunting. The guidelines established by the IDSA/SHEA for developing an institutional program represent a valuable resource and suggest that the best programs are comprehensivetaking into account local antimicrobial use and resistance patterns, as well as available resources.24 The size and nature of the institution can make a big difference in determining what program to set up and what elements it should entail; what works at one institution might not work as well at another. The program components and effectiveness of each will differ for community versus academic medical centers. A comprehensive program includes active monitoring, fostering of appropriate antimicrobial use, and collaboration with an effective infection control program as well as other hospital entities. The role of a multidisciplinary team, with administrative support, is particularly underscored in the guidelines. According to the guidelines, core members of the multidisciplinary team should include an infectious diseases physician and a clinical pharmacist with infectious diseases training. It should also ideally include a clinical microbiologist, information system specialist, infection control professional, and hospital epidemiologist.24 It is important that all members of the team are passionate about the program, oversee its implementation and daily functions, and have some sense of ownership of it. Compensation for its primary participants is crucial. Compensation not only ensures that adequate time is available for executing the daily activities of the program, but it also helps impart a greater sense of program ownership. Process and outcome measures of the program (discussed below) should be included in the performance evaluations of the compensated key participants.
Although the guidelines indicate that an infectious diseases physician should act as the program leader, this might not always be feasible or necessary. Many of the hospitals most in need of improved antimicrobial stewardship simply do not have an infectious diseases physician available to them. In addition, a lot of community hospitals share their infectious diseases physician on a consultative basis with other medical centers and facilities, and that particular specialist may not have a lot of time to invest in the program. Where having an infectious diseases physician as a core member and leader of the team is beneficial, it is not absolutely necessary. A similar argument can be made concerning the inclusion of a clinical pharmacist with infectious diseases training as a core member. Not all hospitals have or can find a clinical pharmacist with formal infectious diseases training through a didactic pharmacy residency program.
If an infectious diseases physician or clinical pharmacist with formal infectious diseases training is not available at a given institution, the team will need to include others ready to assume a greater leadership role. Although not mentioned in the guidelines, hospital medicine specialists and hospitalists are well‐suited to take on this role and can be integral to leadership of the multidisciplinary team. Hospitalists have knowledge of the hospital where they support a wide range of services and, at least in some cases, may have fewer time constraints than a subspecialty. In addition, hospital leadership and administration more often reach out to hospitalists to oversee patient quality care and safety improvement projects, the realm to which antimicrobial stewardship belongs. Regarding clinical pharmacists, an alternative to formal residency training for PharmDs are online certification programs such as MAD‐ID (Making a Difference in Infectious Diseases Pharmacotherapy), the Society of Infectious Diseases Pharmacists, or via a limited number of state medical societies.28, 29 Such certification programs should increase the number of pharmacists and PharmDs with infectious diseases training in the near future.
Antimicrobial stewardship is best considered a medical staff, rather than primary hospital, function. Individuals from the medical staff, and particularly medical staff leadership, are most adept in employing the 3 Cs that are important when constructing, implementing, and operating an institutional antimicrobial stewardship programconceptualization, communication, and coercion. Conceptualization deals with understanding what needs to be done, why it needs to be done, and how to do it, whereas communication is making sure the providers of antimicrobials receive and understand this information. Coercion might seem like a strong term, but it refers to the pressure exerted by thought leaders and others involved in the process to get things done within the institution, including all units or departments. Although ultimate responsibility for an antimicrobial stewardship program should probably lie with the medical staff, the IDSA/SHEA guidelines correctly indicate that support and collaboration of hospital administration, medical staff leadership, and local providers are essential to the success of any such program.24
A Case Study: the Wake Forest University Baptist Medical Center (WFUBMC) Program
Figure 1A,B provides an overview of the general structure of the antimicrobial stewardship program at WFUBMC implemented in 2000. To establish and provide the information needed for day‐to‐day operations of a stewardship program at WFUBMC, data are needed on how, where, and by whom antibiotics are used within the institution. In addition, microbiology data, including the frequency and susceptibility of infecting pathogens, are essential. Obtaining these data often requires the help and cooperation of the information technology (IT) staff at the institution. Considerable time and effort may be required at the outset, but once information system programs are established, ongoing data mining is much easier. At the time of program initiation, it was decided to assess antibiotic use or density (amount of drug per inpatient geographic unit) using the defined‐daily‐dose (DDD) method. This entails assigning a predetermined weight of administered antibiotic as a dose and dividing by a denominator of 1000 patient days. Subsequently, days of therapy per drug has been found to be a more accurate measure of antimicrobial consumption. When developing a program, it is vital to first obtain baseline usage data. Such data should include, if possible, a detailed inventory of usage within different units of the hospital or for particular services, or sometimes even for a particular provider with a history of high antimicrobial usage. Ongoing measurement over time allows the impact of new stewardship interventions and guidelines to be measured, as well as identifying potential new problem areas in usage.

Good microbiology data are also essential to determine problem pathogens at the institution and where they are located. Such data are useful not only to define areas of resistance (potentially warranting changes in antimicrobial policies to alter selection pressures), but also for gathering information necessary for defining local guidelines for antimicrobial use. For example, if the local antibiograms show that a particular pathogen in the hospital ICU has a particular resistance pattern, then initial empiric therapy for patients at risk of infection with those organisms should be chosen to cover the problematic resistant pathogen. Once subsequent microbiology data become available, patients not infected with the pathogen can be de‐escalated to a more narrow‐spectrum antibiotic.
As illustrated in Figure 1A, at WFUBMC, all the collected data are integrated to provide information concerning antibiotic density, usage, and patterns of antimicrobial resistance. This information is received by the staff of the antimicrobial stewardship program, which at WFUBMC is called the Center for Antimicrobial Utilization, Stewardship and Epidemiology (CAUSE). The CAUSE staff works with the day‐to‐day elements of program administration and operations and includes 2 infectious diseases physicians and 2 infectious disease PharmDs. The CAUSE staff works very closely with the microbiology laboratory, hospital pharmacy, and the medical director of hospital epidemiology and infection control.
The CAUSE program at WFUBMC administratively functions through an advisory board committee that includes thought leaders from different medical specialties and patient units of the hospitalparticularly those with high antimicrobial usage, such as hematology/oncology, pulmonary, critical care, and transplantation. The CAUSE staff and advisory board exist to exchange ideas concerning what is working or not working and where problem areas may be, and to propose possible changes in antimicrobial practices at the institution. In addition, thought leaders on the advisory board also receive and evaluate information from various sources about new antimicrobial agents and national guidelines, and, in turn, help disseminate this information to the hospital personnel who will be involved in program implementation.
At WFUBMC, it is the advisory board committee, working in conjunction with the CAUSE staff/medical director, that presents antimicrobials for formulary consideration to the Pharmacy and Therapeutics (P&T) Committee, in addition to any major interventions CAUSE and its advisory board feel are indicated. The P&T committee then reports to the medical staff executive committee and hospital leadership. As should be evident, the approach to stewardship at WFUBMC is medical staff‐driven, rather than a function of administrative constituents.
Finally, no matter how well‐organized an antimicrobial stewardship program is, it will not be fully successful if the entire medical staff does not buy into the process and agree with the need for the proposed changes and interventions involving the practice of antimicrobial therapy. It is important to spend some time early in program development to ensure that the need for an antimicrobial stewardship program, the process, and the outcomes (both in terms of patient care and clinical outcomes at the institution) are clearly communicated to the medical staff, and that their full commitment and cooperation are enlisted. In cases where hospital‐wide infection or resistance rates are known and antimicrobial utilization data are available, it is important to present such information in an understandable and convincing manner that makes the case for a proposed change or intervention, not only at the hospital level but also at the level of the patient.
Elements of a Successful Program: Basic Strategies
Potential strategies or elements of an antimicrobial stewardship program are listed in Table 2. Two evidence‐based fundamental or core strategies have been recommended by the IDSA/SHEA guidelines24 and implemented at numerous institutions with various levels of success. The first is a so‐called back‐end approach to modifying antimicrobial therapy on the basis of prospective audit of antimicrobial use, with intervention and feedback to the provider. The second is a front‐end approach using formulary restriction and preauthorization requirements for specific antimicrobial agents. Various supplemental strategies, including large group and patient case‐based education, guidelines and clinical pathways, antimicrobial order forms, and computerized clinical decision support, are also recommended.
| Program Element | Advantages | Disadvantages | Comments |
|---|---|---|---|
| Prospective Audit and Feedback | Proven in clinical studies to reduce and modify antimicrobial consumption, improve selected clinical outcomes, and decrease antimicrobial expenditures Provides one‐on‐one patient‐centered education to the clinician Allows optimization of anti‐infective pharmacology | Adherence to stewardship interventions by the clinician is voluntary Resource intensive Requires a greater amount of team member training and experience in anti‐infective therapy | Back‐end approach Identify and intervene on patients already started on antimicrobials Interventions include changing, streamlining, de‐escalation, pharmacodynamic/dose optimization, IV to PO switch, and limitation of duration of therapy |
| Restriction or Preauthorization | Proven in clinical studies to reduce and modify antimicrobial consumption, improve selected clinical outcomes, and decrease antimicrobial expenditures Together with infection control effective in controlling outbreaks of resistant or secondary pathogens (such as C. diff) | Less appealing to clinicians Loss of prescriber autonomy Potential need for after‐hours service Time intensive Potential for delay in antimicrobial administration | Front‐end approach Formulary restriction or contact a stewardship team member to obtain authorization to prescribe a selected antimicrobial Each intervention is a mini‐consult |
| Large Group Education | Can reach a large number of prescribers in a short period of time Effective for communicating the need and rationale for subsequent stewardship interventions | Not particularly effective in changing prescribing behavior without other interventions Rapid extinction of gained knowledge | Grand rounds or clinical staff meeting venues Provides information to prescribers and thought leader clinicians on justification for stewardship Feedback antimicrobial susceptibility and use data to clinicians |
| Guidelines and Pathways | Limits variation in therapy of infectious diseases Best evidence‐based Assists in adherence with regulatory and third‐party payer stipulations | Often not utilized unless combined with other stewardship strategies or elements | Best if local data and conditions are used to adapt guidelines to a specific institution |
| Computerized Physician Order Entry and Clinical Decision Support | Shown in limited clinical studies to reduce and modify antimicrobial consumption, improve selected clinical outcomes, and decrease antimicrobial expenditures Once established can greatly assist with implementation of guidelines and best‐evidence therapy. Reduces adverse events related to antimicrobials | Resource intensive during design and implementation Expensive Not readily available | Often entails modification of existing or purchasing of additional informatics |
| Microbiology Interventions | Potential to improve antimicrobial use and anti‐infective therapy for the individual patient | Not well studied | Includes cascade reporting to hide antimicrobial susceptibilities that might promote suboptimal therapy (eg, fluoroquinolone susceptibility for invasive S. aureus) Assistance with choices of automated susceptibility profile, communication of new or changes in testing protocols Preauthorization of susceptibility testing for unconventional antibiotics |
| Rapid Diagnostics | Provides opportunity for early targeted therapy Assists with de‐escalation Shown in very limited studies to decrease antimicrobial consumption and improve clinical outcomes | Not readily available Expensive | Includes PCR and antigen testing of clinical specimens or early culture growth with rapid turnaround of test results |
| Antimicrobial Cycling | Potential to decrease antimicrobial resistance for an institution or geographic unit | Not consistently shown in clinical trials to improve clinical outcomes or decrease resistance Often increases antimicrobial consumption Extremely labor intensive to ensure adherence | Changing antimicrobial protocols periodically in an attempt to reduce selection pressure for resistance |
Prospective Audit With Intervention and Feedback
This approach usually involves the use of an antimicrobial support team that reviews initial or ongoing therapy and then intervenes to provide feedback and suggested modifications to the medical care provider to improve therapy. This can be done by an infectious diseases physician, a clinical pharmacist, or a hospitalist or internist with expertise in antimicrobial therapy. The aim is to provide patient‐specific education and/or suggest changes to antimicrobial utilization (when needed) to improve and streamline therapy. Suggested interventions could include discontinuing or changing 1 or more drugs, switching intravenous to oral drug administration, and suggesting a short‐course duration of therapy. Occasionally, suggestions are made when appropriate to actually escalate or intensify therapy to increase efficacy. Identification of patients for targeting or focusing prospective audit and feedback efforts typically involves using computer surveillance to single out problem antimicrobials or problematic usage, given local resistance patterns or patient characteristics.24 Examples could include a focus on asymptomatic bacteruria, excessive duration of therapy for ventilator‐associated pneumonia, or overzealous use of certain classes of antimicrobials. Another potential activity for a prospective audit and feedback team is to review reports of patient‐specific blood and sterile body fluid culture results matched to the patient's current antimicrobial therapy. This allows for daily review of the appropriateness of therapy for potentially serious infections. Some patients seen by the antibiotic support team may be referred for infectious diseases or other expert consultative opinion if their infections or therapy are felt to be too complicated for routine prospective audit and feedback recommendations.
A number of studies have demonstrated that strategies involving prospective audit with intervention and feedback can improve antibiotic stewardship, as measured by reductions in inappropriate antibiotic use,30 reduced antibiotic costs or overall consumption,3135 greater compliance with hospital treatment guidelines or policies33, 36, 37 and, in some cases, reduced number of infections due to C difficile infection32, 37 or resistant pathogens.31, 32, 37 Prospective audit with feedback is probably the best and most effective core strategy for a community hospital program where other interventions are cumbersome or not as well tolerated by the medical staff. One potential disadvantage of the prospective audit with intervention and feedback approach is that medical provider adherence is largely voluntary. The team can make suggestions, but if the provider disagrees or is unobtainable, the suggestion is never implemented. Also, this strategy can also be resource‐intensive from a personnel perspective.
Formulary Restriction and Preauthorization
The other major strategy used to achieve antimicrobial stewardship goals involves antimicrobial restriction. This can be accomplished either by not including the particular antimicrobial agent on the hospital formulary or by requiring the medical provider to obtain preauthorization before prescribing a restricted drug. A pager system or telephone call is often used for preauthorization, whereby the clinician wishing to prescribe a particular agent calls or pages a member of the stewardship team in order to obtain prescribing permission. When using preauthorization, it is important that the individuals who receive the calls actually see patients and have clinical experience and the respect of the medical staff, as each call may be a mini‐consult. Oftentimes, the provider or prescriber making the call is asking for suggestions as to what antimicrobial might be used, and not simply to obtain authorization to use a drug that is otherwise restricted. Studies have shown that effective interventions supporting stewardship are better provided by attending infectious diseases staff or clinical pharmacists, rather than persons in training.38
Regarding the identification of antimicrobials for restriction, a program should preferentially choose those drugs that are involved in therapy for complex patients and infections. It is also a reasonable approach for drugs that are, or have the potential to be, overused for certain infections where alternatives exist. For work‐horse antimicrobials, those drugs overused or misused for several different infectious diseases, prospective audit and feedback is arguably a better strategy to reduce and modulate consumption.
Formulary restriction and preauthorization is clearly effective in modulating antimicrobial use. A large number of studies have demonstrated reductions in antibiotic drug use, and often in cost, after hospital implementation of a formulary restriction or preauthorization approach to antimicrobial stewardship.3947 It has been more difficult to demonstrate other benefits associated with this approach, although there is some support for its aid in controlling nosocomial infection outbreaks. Restriction of clindamycin48 (or clindamycin, cefotaxime, and vancomycin27) has been shown to control outbreaks of nosocomial C difficileassociated diarrhea and VRE, respectively. More recently, Internet‐based antimicrobial restriction programs49, 50 and a computerized (electronic) approval system51 have been demonstrated to reduce antibiotic use at tertiary hospitals.
Some studies have reported increased antibiotic drug susceptibilities after implementation of institutional preauthorization policies,45, 46, 51 and at least 1 reported a decreased incidence of ceftazidime‐resistant Klebsiella species after instituting a preapproval policy for cephalosporins.52 However, there is concern that restricting 1 class of antibiotics and replacing it with another will simply replace 1 resistant species with another, the so‐called squeezing the balloon effect.53 This was observed in the latter study, where a 44% reduction in ceftazidime‐resistant Klebsiella species at the hospital was accompanied by a 69% increase in incidence of imipenem‐resistant P aeruginosa.52 To assess and enable response to possible squeezing the balloon effects, the guidelines recommend monitoring overall trends in antimicrobial use for institutions using preauthorization strategies.
Possible disadvantages of preauthorization and restriction include perceived loss of autonomy for prescribers, the potential need for all‐hours support, inaccurate or misleading information from the prescriber (leading to inappropriate recommendations),24 and significant delay in stat antimicrobial administration.54 Delay in antimicrobial administration due to the time required to obtain preauthorization and have the approval communicated to the pharmacy was not observed when studied as a process measure at WFUBMC (Ohl, unpublished data, 2008).
A study by Linkin and colleagues showed that 39% of telephone calls for preauthorization of a restricted antimicrobial contained an inaccuracy in at least 1 type of patient data.38 A follow‐up by the same group demonstrated that inaccurate communication was significantly associated with inappropriate antimicrobial recommendations (odds ratio [OR] 2.2; P = .03); this was particularly the case for inaccuracies in microbiologic data (OR 7.5; P = .002).55 Also, if all‐hours support is not provided, at least 1 study has shown some physicians may engage in stealth dosing, that is, avoiding having to obtain preauthorization for restricted antimicrobials by waiting until off‐hours to place orders.56 The latter can be dealt with by following up on such orders with a prospective audit and feedback component of the program. Preauthorization is usually more difficult to employ and less accepted in non‐academic medical centers. Prospective audit and feedback may be more appropriate in such settings.
Supplemental Strategies
A number of additional options are available to supplement the 2 core strategies just described, and are listed in Table 2. Education is generally considered an essential component of any effective antimicrobial stewardship program, but it generally has little lasting impact on providers' behavior, unless it is incorporated with other active interventions.24 In particular, the large group or Grand Roundstype education, where someone describes what needs to be done and why, typically does not produce lasting behavioral changes. There might be, and often is, some short‐term modification, but long‐lasting change at the provider level requires consistent and repeated educational endeavors. Such large group educational venues are more effective and better used as a forum to describe or garner support for an impending stewardship program or intervention, rather than to teach a specific practice.
Using the antimicrobial stewardship program to adapt national guidelines to local antimicrobial use, microbiology, and resistance patterns57, 58 or using clinical (critical) pathways59 has also been shown to improve antimicrobial utilization at hospitals. National guidelines generally enjoy widespread support, but they commonly lack specific information about how to implement recommendations at a given hospital or how to incorporate local data relevant for decision making. A 2006 report by Beardsley and coworkers provides a model from WFUBMC on how local microbiologic data can be used to modify national treatment guidelines to better serve the needs of patients treated at a particular institution.60 Using American Thoracic Society (ATS) and IDSA guidelines for the management of hospital‐acquired pneumonia (HAP), together with local data on the most common bacterial pathogens and their susceptibility to piperacillin‐tazobactam, cefepime, ciprofloxacin, and amikacin (based on length of hospitalization), the WFUBMC CAUSE Advisory Board developed institution‐specific HAP guidelines. The new guidelines divided the ATS/IDSA late onset/risk of the MDR pathogens group of patients into 2 subcategories, early‐late and late‐late pneumonias. Also, unlike the national guidelines, the new guidelines did not recommend ciprofloxacin as empiric therapy, instead recommending amikacin as a component of regimens targeting late‐late pneumonias.
Newer (and in some cases not so new) information technologies can be adapted to healthcare delivery and prescriber support to improve antimicrobial stewardship. These include computer decision support61 and alert systems6265; computerized physician order entry (CPOE)66, 67; electronic medical records24; electronic retrieval of treatment guidelines or clinical texts68; and personal digital assistant (PDA) applications providing information on pathogens, diagnosis, medication, and treatment.68, 69 In addition, computer‐based surveillance64, 70, 71 and Web‐based systems for antimicrobial approval; automated clinical decision support; and/or enhanced real‐time communication between prescribers and other members of the antimicrobial stewardship team show promise for antimicrobial stewardship programs.49, 50
Computer‐assisted decision support has been shown to improve or reduce antibiotic‐susceptibility mismatches (improve selection of effective therapy), overall antibiotic use, excess antimicrobial dosages, excessive‐dose days, selection of antimicrobials for which the patient was poorly matched in terms of allergies, and antimicrobial‐related adverse events, as well as reduce antimicrobial drug costs, total hospital costs, and length of hospital stay.7277 For their part, CPOE systems have been shown to improve compliance with treatment guidelines, decrease medication and other medical errors, shorten length of hospital stay, and decrease pharmaceutical costs.66, 67, 78 In many cases, CPOE systems can now be modified to include some clinical decision support to improve antimicrobial use.78
The IDSA/SHEA guidelines note that antimicrobial decisions can be improved through use of CPOE, clinical decision support, and electronic medical records that enable incorporation of data on patient‐specific microbiology cultures and susceptibilities, hepatic and renal function, drug interactions, allergies, and cost. They also point out that computer‐based surveillance can facilitate good stewardship by enabling more efficient targeting of antimicrobial interventions, tracking of antimicrobial resistance patterns, and identification of HAIs and adverse drug events.24 Recently, a few proprietary informatics programs that perform such functions for the hospital epidemiologist and antimicrobial steward have become available, including but not limited to TheraDoc (Salt Lake City, UT), SafetySurveillor (Premier, Inc., Charlotte, NC), and BD Protect (BD Diagnostics, Austin, TX). Perhaps one of the best‐known comprehensive hospital information systems that incorporates and integrates several information technologies to improve patient care at the level of the prescriber is the Health Evaluation through Logical Processing (HELP) system at LDS Hospital in Salt Lake City, Utah.7981 Unfortunately, these programs are expensive, need considerable time for installation and validation, and do not always perform the functions needed by the medical center. The medical community has generally been slow to incorporate healthcare information technology to improve antimicrobial use or general medical care, but in the last few years more hospitals are finding their merit.
On the basis of evidence currently available, the 2007 guidelines do not recommend the routine use of antimicrobial cycling or combination therapy to prevent or reduce antimicrobial resistance. Such strategies, where at first glance might intuitively seem to make sense, have not been shown to improve patient care, improve antimicrobial choices, or reduce antimicrobial resistance. In addition, antimicrobial cycling in particular is difficult to implement and labor intensive to oversee.24
One strategy for improving antimicrobial stewardship not mentioned in the 2007 IDSA/SHEA guidelines, but might become increasingly important in the future, is the use of rapid molecular diagnostic testing. Knowing the identity of the causative pathogen sooner or being able to rapidly rule out certain pathogens should enable better decision‐making. During the 2009/2010 influenza season with H1N1 influenza, WFUBMC was able to implement rapid viral testing and learned some things that enabled improvement of hospital practices. It was found that approximately 10% to 15% of the pneumonias in immunocompromised patients at the center were not bacterial but viral, the pathogens being respiratory syncytial virus (RSV) or metapneumovirus (Ohl, unpublished data, 2010). Upon finding a viral etiology to a lower respiratory tract infection, rapid de‐escalation of antibiotic therapy was possible. If rapid diagnostics are to be performed, it is important that there are systems in place to respond quickly to the findings, so the benefits of having rapid data can be realized.
Evaluating Antimicrobial Stewardship Programs
Two general types of measures are used to evaluate the effectiveness of antimicrobial stewardship: process and outcome. As with most things done in the hospital, process measures are easier. They measure surrogate impacts of a program, accountability, resource use, and cost effectiveness. In essence, process measures evaluate whether the program accomplished what it set out to do in terms of changing certain processes or prescriber behaviors. It is important to measure resource use, as this helps to continue funding and to keep workers involved in the project. Good programs will save money; this can easily be measured, even if it is just as simple as going to the hospital pharmacy and looking at the cost of antimicrobials provided per patient day.
Outcomes like decreases in particular infections, less emergence of antimicrobial resistance, or other patient‐specific measures are likely more important in the big picture, but they are also much more difficult to measure. For example, where one would like to measure changes in pathogen resistance after making some changes in antimicrobial stewardship, it often takes years before the benefits of a particular intervention or change materialize in terms of less resistance or reduced emergence of resistance. If that type of change is to be measured, then one needs to be persistent and continue measurements over a long period of time. In addition, given the protracted amount of time before these outcomes may be observed, a number of other changes are likely to happen that coincide with the antimicrobial stewardship interventions and make assessment of causality difficult and biased.
Having said that, a number of studies have demonstrated a relationship between antibiotic restriction48, 8285 or other antimicrobial stewardship policies32, 86 and decreases in nosocomial C difficile infections or disease. Figure 2 illustrates the impact of a nonrestrictive antimicrobial stewardship program at a secondary/tertiary‐care hospital in Quebec, Canada, on an epidemic of C difficileassociated disease (CDAD) that occurred at the institution during the latter portion of 2003.86 Following program implementation, and the major drop in targeted antibiotic consumption, the incidence of CDAD also significantly decreased. Earlier implementation of infection control measures had no effect on CDAD incidence.

A smaller number of studies have reported decreases in resistant gram‐negative bacteria following implementation of antimicrobial stewardship programs. For example, Meyer and colleagues reported a marked reduction in ceftazidime‐resistant K pneumoniae at a 487‐bed general hospital in New York City after implementation of enhanced ceftazidime restriction and barrier precautions following an outbreak of infections caused by the resistant K pneumoniae.87 Similarly, Carling and coworkers reported a significant decrease in nosocomial infections caused by resistant Enterobacteriaceae following implementation of a multidisciplinary antibiotic stewardship program to minimize inappropriate use of third‐generation cephalosporins (Figure 3).32 More recently, a retrospective, longitudinal, multicenter analysis of a consortium of 22 academic health centers in the United States showed that incidence rates of carbapenem‐resistant P aeruginosa were lower at hospitals that restricted carbapenems than those that did not (P = .01).88

Evidence suggesting a beneficial impact of antimicrobial stewardship programs on resistance in gram‐positive organisms is limited. More specifically, the study by Carling and colleagues reported an apparent decrease in VRE rates following implementation of their program to reduce inappropriate use of third‐generation cephalosporins.32 The hospital had VRE rates similar to other NNIS System hospitals prior to beginning the program, but after antibiotic stewardship measures were implemented, the VRE rate began to drop, falling to 6% by 1999. This should be compared with a VRE rate of 24% for similar NNIS System hospitals in 1999.
As far as reducing healthcare costs, Figure 4A illustrates the direct antimicrobial cost savings at WFUBMC after implementation of the CAUSE antimicrobial stewardship program, and Figure 4B after supplemental interventions were implemented. Although decreasing antimicrobial cost is important, one would like to show decreases in overall healthcare costs associated with an antimicrobial stewardship program. Unfortunately, this is often a little more difficult to demonstrate. Extrapolations, however, may be possible. Because antimicrobial resistance, adverse drug effects, and secondary unintended infections such as C difficile colitis have been linked with increased patient morbidity and mortality, longer hospital stays, and increased healthcare costs,6, 89, 90 improved antimicrobial stewardship is expected to optimize patient care and lower overall healthcare costs. A study in a large tertiary care academic medical center estimated more than $4.25 million in total healthcare savings over 1 year with a stewardship program using both preauthorization and, to a lesser extent, prospective audit and feedback.91 Despite the fact cost saving should not be a primary goal of an antimicrobial stewardship program, lower antimicrobial costs can help keep a program funded and buttress a proposal for an antimicrobial stewardship program to hospital leadership.

Many hospitals recognize other areas where an antimicrobial stewardship program can demonstrate its value. This includes implementation of a rapid change in drug utilization during antimicrobial supply shortages and assistance with regulatory mandates and surgical infection prophylaxis. Stewardship teams often assist microbiology with protocols for microbiology reporting, antibiograms, planning for susceptibility testing, and communicating changes in microbiology tests or protocols to clinicians.
Conclusions
Overuse or misuse of antibiotics and other antimicrobials for hospital inpatients is relatively common, and can be associated with several unintended negative consequences. Improving medical care necessarily includes better use of antimicrobials to optimize outcomes and preserve the effectiveness of currently available agents. Further, an important additional consequence of effective antimicrobial stewardship and improved patient care is typically a lowering of overall healthcare costs. The recent 2007 IDSA/SHEA guidelines provide recommendations for developing an institutional program to enhance antimicrobial stewardship. However, individual institutions need to look closely at their own systems and patients to develop an antimicrobial stewardship program that best serves the needs of their hospital and the people it serves.
Nosocomial, or hospital‐acquired, infections (HAIs) are a major cause of patient morbidity and mortality in the United States and other countries.15 In 2002, approximately 1.7 million HAIs occurred in US hospitals and were associated with an estimated 98,987 deaths.1 Of particular note, increasing percentages of HAIs are now caused by antimicrobial‐resistant pathogens, which have been linked with increases in morbidity, mortality, length of hospital stay, and healthcare costs.6
The 2004 data summary from the United States National Nosocomial Infections Surveillance (NNIS) System Report highlighted substantial increases for year 2003 versus 1998 through 2002 in vancomycin‐resistant enterococci (VRE); methicillin‐resistant Staphylococcus aureus; Klebsiella pneumoniae resistant to third‐generation cephalosporins; and Pseudomonas aeruginosa resistant to imipenem, quinolones, or third‐generation cephalosporins.7 Other gram‐negative bacteria of concern include Escherichia coli and Acinetobacter baumannii, as well as Enterobacter cloacae and E. aerogenes.8, 9
The increasing number of multidrug‐resistant (MDR) gram‐negative bacteria within the healthcare setting is particularly concerning.1013 Too frequently, clinicians in the United States now encounter gram‐negative bacteria species that are resistant to many, and occasionally all, currently available antibiotics. For many of these MDR gram‐negative pathogens the antimicrobials that potentially remain active (eg, aminoglycosides and polymyxins) are often more toxic and less efficacious for some infections.14 Particularly problematic is that the pharmaceutical industry's developmental pipeline for new antibiotics, with novel mechanisms of action that might be used against MDR gram‐negative pathogens, has virtually come to a standstill.15, 16 Even if an investigational drug was in phase 2 or 3 trials right now or entered the US Food and Drug Administration (FDA) Fast Track Development Program, it would be at least 10 or 15 years before that drug would be available on the US market.
What this means is that the clinician's current antibiotic armamentarium is all they can expect in the foreseeable future. It also means that special care needs to be taken to optimally use currently available agents to ensure continued activity against the pathogens encountered in the hospital (and community) setting, now and in the future. Maximizing clinical outcomes, while minimizing the emergence and spread of antimicrobial resistance (and other adverse effects associated with suboptimal antimicrobial drug use), falls under the purview of antimicrobial stewardship, the focus of this paper.
Antimicrobial StewardshipWhy Is It Needed, What Is It, and What Are Its Goals?
Inappropriate Antimicrobial Use
Early in the onset of many infections, the data needed to make a rational, informed decision about specific antibiotic therapy are usually unavailable. For many infections, therapy cannot be delayed waiting for microbiology or other findings, and broad‐spectrum empiric therapy is begun on the basis of educated guesses made from the patient's presentation and characteristics, and local or hospital antibiograms. In addition, for many serious infections, delay in antimicrobial therapy will increase patient morbidity and mortality. Generally, what occurs is the decision to treat empirically with one or more broad‐spectrum antibiotic agents, which are then continued for the entire course of therapy. Opportunities are often missed to tailor therapy later in the course of infection when microbiologic or other data are available. There is also a tendency for spiraling empiricism to occur when a patient is not doing well with initial therapy; additional agents with broad antimicrobial activity, including antifungals and antivirals, are added to the therapeutic regimen, often in a haphazard way.17
Besides the perceived need to prescribe broad‐spectrum and/or multiple antibiotics to cover possible or perceived resistant or uncommon pathogens, a number of other factors contribute to inappropriate antibiotic or antimicrobial use. Many times antimicrobials are initiated when no infection exists, such as for asymptomatic bacteruria, noninfectious pulmonary conditions, or endotracheal tube or Foley catheter colonization. Another example of inappropriate use is treating for longer than needed to eradicate infection. All of these events intensify the exposure of bacteria colonizing or infecting the patient to multiple anti‐infective drugs and increase the chances for selection of an MDR pathogen.
Examining antibiotic usage at the hospital level, approximately 60% of adult patients admitted to US hospitals receive at least 1 dose of an antibiotic agent during their stay (range: 44%74% for individual hospitals).18, 19 Similarly, at Wake Forest University Baptist Medical Center (WFUBMC), approximately 75% of inpatients receive antimicrobials at some point during their hospitalization (Ohl, unpublished data, 2007). One recent example by Hecker and colleagues conducted in a 650‐bed, university‐affiliated US hospital reported 30% of the total days of antibiotic therapy received by adult non‐ICU inpatients was unnecessary.20 The most common reasons for unnecessary use were administration for longer than recommended durations, administration for a noninfectious or nonbacterial syndrome, and treatment of colonizing or contaminating microorganisms.
Consequences of antibiotic misuse
Unwanted consequences of antimicrobial therapy include increased morbidity and mortality, adverse drug reactions, increased length of hospital stay and hospitalization costs, predisposition to secondary infections, and emergence and selection of drug‐resistant organisms.21, 22 Selection or induction of antimicrobial resistance and promotion of secondary infection with Clostridium difficileparticularly with new, more toxigenic strains23are of particular concern in the current hospital environment.22 These untoward consequences can be seen as a calculated risk of antibiotic therapy for any single‐treated patient, or as an undesired outcome measure for excessive use at the level of the healthcare institution. For example, a 7‐day course of a third‐generation cephalosporin in a particular patient increases the risk of subsequent infection from an extended‐spectrum beta‐lactamase (ESBL)‐producing gram‐negative rod. For the institution as a whole, excessive use of this antimicrobial will increase the overall prevalence and number of infections due to this troublesome resistance factor.
Definition and Goals of Antimicrobial Stewardship
The above studies show a clear need for improved, more careful and prudent use of antimicrobials, which is key to antimicrobial stewardship. Building on the definition given by the Infectious Diseases Society of America and Society for Healthcare Epidemiology of America (IDSA/SHEA),24 antimicrobial stewardship is essentially a system of personnel, informatics, data collection, and policy/procedures that promotes the optimal selection, dosing, and duration of therapy for antimicrobial agents throughout the course of their use. An effective antimicrobial stewardship program will limit inappropriate and excessive antimicrobial use, but more importantly improve and optimize therapy for the individual infected patient.
The goals of antimicrobial stewardship are listed in Table 1.24, 25 It is important to recognize that the primary goals of antimicrobial stewardship are not the reduction of healthcare costsand certainly not the reduction of drug acquisition or usage costs. As the 2007 IDSA/SHEA guidelines for institutional development of an antimicrobial stewardship program make clear, the primary goal is to focus on patient care; that is, to optimize clinical outcomes, while minimizing unintended consequences of antimicrobial use (emergence of resistance, selection of pathogenic organisms, and adverse drug reactions).24
| Prevent or slow the emergence of antimicrobial resistance |
| Optimize selection, dose and duration of therapy |
| Reduce adverse drug events, including secondary infection (eg, C. difficile antibiotic‐associated diarrhea) |
| Reduce morbidity and mortality |
| Reduce length of stay |
| Reduce healthcare expenditures |
Reduced healthcare costs without an adverse effect on quality of patient care is, however, a legitimate secondary goal of antimicrobial stewardship, and will result from optimized clinical outcomes and decreased potential collateral damage associated with pharmacotherapy. Unfortunately, it is much more difficult to measure the impact of an antimicrobial stewardship program on emergence of resistance than on drug acquisition or usage costs. As a consequence, reduction in drug acquisition/usage costs has too often been viewed as the primary (and sometimes only) justification for implementing an antimicrobial stewardship program.26
Finally, the role of effective infection control cannot be overemphasized. Infection control is clearly necessary and often sufficient to reduce HAIs. However, a comprehensive infection control program, combined with an effective antimicrobial stewardship agenda, synergistically limit the emergence and spread of antimicrobial‐resistant bacteria, reduce HAIs, control resistance, and improve overall inpatient care.24, 27 Hence, when instituting an antimicrobial stewardship program, it is essential to ensure the hospital or other healthcare institution already has a robust hospital epidemiology and infection control program in placeor to simultaneously institute one.
Constructing an Antimicrobial Stewardship Program
Infectious Diseases Society of America and Society for Healthcare Epidemiology of America Guidelines
Whereas the value of antimicrobial stewardship is widely appreciated, actually taking the steps to set up a healthcare facility program can be daunting. The guidelines established by the IDSA/SHEA for developing an institutional program represent a valuable resource and suggest that the best programs are comprehensivetaking into account local antimicrobial use and resistance patterns, as well as available resources.24 The size and nature of the institution can make a big difference in determining what program to set up and what elements it should entail; what works at one institution might not work as well at another. The program components and effectiveness of each will differ for community versus academic medical centers. A comprehensive program includes active monitoring, fostering of appropriate antimicrobial use, and collaboration with an effective infection control program as well as other hospital entities. The role of a multidisciplinary team, with administrative support, is particularly underscored in the guidelines. According to the guidelines, core members of the multidisciplinary team should include an infectious diseases physician and a clinical pharmacist with infectious diseases training. It should also ideally include a clinical microbiologist, information system specialist, infection control professional, and hospital epidemiologist.24 It is important that all members of the team are passionate about the program, oversee its implementation and daily functions, and have some sense of ownership of it. Compensation for its primary participants is crucial. Compensation not only ensures that adequate time is available for executing the daily activities of the program, but it also helps impart a greater sense of program ownership. Process and outcome measures of the program (discussed below) should be included in the performance evaluations of the compensated key participants.
Although the guidelines indicate that an infectious diseases physician should act as the program leader, this might not always be feasible or necessary. Many of the hospitals most in need of improved antimicrobial stewardship simply do not have an infectious diseases physician available to them. In addition, a lot of community hospitals share their infectious diseases physician on a consultative basis with other medical centers and facilities, and that particular specialist may not have a lot of time to invest in the program. Where having an infectious diseases physician as a core member and leader of the team is beneficial, it is not absolutely necessary. A similar argument can be made concerning the inclusion of a clinical pharmacist with infectious diseases training as a core member. Not all hospitals have or can find a clinical pharmacist with formal infectious diseases training through a didactic pharmacy residency program.
If an infectious diseases physician or clinical pharmacist with formal infectious diseases training is not available at a given institution, the team will need to include others ready to assume a greater leadership role. Although not mentioned in the guidelines, hospital medicine specialists and hospitalists are well‐suited to take on this role and can be integral to leadership of the multidisciplinary team. Hospitalists have knowledge of the hospital where they support a wide range of services and, at least in some cases, may have fewer time constraints than a subspecialty. In addition, hospital leadership and administration more often reach out to hospitalists to oversee patient quality care and safety improvement projects, the realm to which antimicrobial stewardship belongs. Regarding clinical pharmacists, an alternative to formal residency training for PharmDs are online certification programs such as MAD‐ID (Making a Difference in Infectious Diseases Pharmacotherapy), the Society of Infectious Diseases Pharmacists, or via a limited number of state medical societies.28, 29 Such certification programs should increase the number of pharmacists and PharmDs with infectious diseases training in the near future.
Antimicrobial stewardship is best considered a medical staff, rather than primary hospital, function. Individuals from the medical staff, and particularly medical staff leadership, are most adept in employing the 3 Cs that are important when constructing, implementing, and operating an institutional antimicrobial stewardship programconceptualization, communication, and coercion. Conceptualization deals with understanding what needs to be done, why it needs to be done, and how to do it, whereas communication is making sure the providers of antimicrobials receive and understand this information. Coercion might seem like a strong term, but it refers to the pressure exerted by thought leaders and others involved in the process to get things done within the institution, including all units or departments. Although ultimate responsibility for an antimicrobial stewardship program should probably lie with the medical staff, the IDSA/SHEA guidelines correctly indicate that support and collaboration of hospital administration, medical staff leadership, and local providers are essential to the success of any such program.24
A Case Study: the Wake Forest University Baptist Medical Center (WFUBMC) Program
Figure 1A,B provides an overview of the general structure of the antimicrobial stewardship program at WFUBMC implemented in 2000. To establish and provide the information needed for day‐to‐day operations of a stewardship program at WFUBMC, data are needed on how, where, and by whom antibiotics are used within the institution. In addition, microbiology data, including the frequency and susceptibility of infecting pathogens, are essential. Obtaining these data often requires the help and cooperation of the information technology (IT) staff at the institution. Considerable time and effort may be required at the outset, but once information system programs are established, ongoing data mining is much easier. At the time of program initiation, it was decided to assess antibiotic use or density (amount of drug per inpatient geographic unit) using the defined‐daily‐dose (DDD) method. This entails assigning a predetermined weight of administered antibiotic as a dose and dividing by a denominator of 1000 patient days. Subsequently, days of therapy per drug has been found to be a more accurate measure of antimicrobial consumption. When developing a program, it is vital to first obtain baseline usage data. Such data should include, if possible, a detailed inventory of usage within different units of the hospital or for particular services, or sometimes even for a particular provider with a history of high antimicrobial usage. Ongoing measurement over time allows the impact of new stewardship interventions and guidelines to be measured, as well as identifying potential new problem areas in usage.

Good microbiology data are also essential to determine problem pathogens at the institution and where they are located. Such data are useful not only to define areas of resistance (potentially warranting changes in antimicrobial policies to alter selection pressures), but also for gathering information necessary for defining local guidelines for antimicrobial use. For example, if the local antibiograms show that a particular pathogen in the hospital ICU has a particular resistance pattern, then initial empiric therapy for patients at risk of infection with those organisms should be chosen to cover the problematic resistant pathogen. Once subsequent microbiology data become available, patients not infected with the pathogen can be de‐escalated to a more narrow‐spectrum antibiotic.
As illustrated in Figure 1A, at WFUBMC, all the collected data are integrated to provide information concerning antibiotic density, usage, and patterns of antimicrobial resistance. This information is received by the staff of the antimicrobial stewardship program, which at WFUBMC is called the Center for Antimicrobial Utilization, Stewardship and Epidemiology (CAUSE). The CAUSE staff works with the day‐to‐day elements of program administration and operations and includes 2 infectious diseases physicians and 2 infectious disease PharmDs. The CAUSE staff works very closely with the microbiology laboratory, hospital pharmacy, and the medical director of hospital epidemiology and infection control.
The CAUSE program at WFUBMC administratively functions through an advisory board committee that includes thought leaders from different medical specialties and patient units of the hospitalparticularly those with high antimicrobial usage, such as hematology/oncology, pulmonary, critical care, and transplantation. The CAUSE staff and advisory board exist to exchange ideas concerning what is working or not working and where problem areas may be, and to propose possible changes in antimicrobial practices at the institution. In addition, thought leaders on the advisory board also receive and evaluate information from various sources about new antimicrobial agents and national guidelines, and, in turn, help disseminate this information to the hospital personnel who will be involved in program implementation.
At WFUBMC, it is the advisory board committee, working in conjunction with the CAUSE staff/medical director, that presents antimicrobials for formulary consideration to the Pharmacy and Therapeutics (P&T) Committee, in addition to any major interventions CAUSE and its advisory board feel are indicated. The P&T committee then reports to the medical staff executive committee and hospital leadership. As should be evident, the approach to stewardship at WFUBMC is medical staff‐driven, rather than a function of administrative constituents.
Finally, no matter how well‐organized an antimicrobial stewardship program is, it will not be fully successful if the entire medical staff does not buy into the process and agree with the need for the proposed changes and interventions involving the practice of antimicrobial therapy. It is important to spend some time early in program development to ensure that the need for an antimicrobial stewardship program, the process, and the outcomes (both in terms of patient care and clinical outcomes at the institution) are clearly communicated to the medical staff, and that their full commitment and cooperation are enlisted. In cases where hospital‐wide infection or resistance rates are known and antimicrobial utilization data are available, it is important to present such information in an understandable and convincing manner that makes the case for a proposed change or intervention, not only at the hospital level but also at the level of the patient.
Elements of a Successful Program: Basic Strategies
Potential strategies or elements of an antimicrobial stewardship program are listed in Table 2. Two evidence‐based fundamental or core strategies have been recommended by the IDSA/SHEA guidelines24 and implemented at numerous institutions with various levels of success. The first is a so‐called back‐end approach to modifying antimicrobial therapy on the basis of prospective audit of antimicrobial use, with intervention and feedback to the provider. The second is a front‐end approach using formulary restriction and preauthorization requirements for specific antimicrobial agents. Various supplemental strategies, including large group and patient case‐based education, guidelines and clinical pathways, antimicrobial order forms, and computerized clinical decision support, are also recommended.
| Program Element | Advantages | Disadvantages | Comments |
|---|---|---|---|
| Prospective Audit and Feedback | Proven in clinical studies to reduce and modify antimicrobial consumption, improve selected clinical outcomes, and decrease antimicrobial expenditures Provides one‐on‐one patient‐centered education to the clinician Allows optimization of anti‐infective pharmacology | Adherence to stewardship interventions by the clinician is voluntary Resource intensive Requires a greater amount of team member training and experience in anti‐infective therapy | Back‐end approach Identify and intervene on patients already started on antimicrobials Interventions include changing, streamlining, de‐escalation, pharmacodynamic/dose optimization, IV to PO switch, and limitation of duration of therapy |
| Restriction or Preauthorization | Proven in clinical studies to reduce and modify antimicrobial consumption, improve selected clinical outcomes, and decrease antimicrobial expenditures Together with infection control effective in controlling outbreaks of resistant or secondary pathogens (such as C. diff) | Less appealing to clinicians Loss of prescriber autonomy Potential need for after‐hours service Time intensive Potential for delay in antimicrobial administration | Front‐end approach Formulary restriction or contact a stewardship team member to obtain authorization to prescribe a selected antimicrobial Each intervention is a mini‐consult |
| Large Group Education | Can reach a large number of prescribers in a short period of time Effective for communicating the need and rationale for subsequent stewardship interventions | Not particularly effective in changing prescribing behavior without other interventions Rapid extinction of gained knowledge | Grand rounds or clinical staff meeting venues Provides information to prescribers and thought leader clinicians on justification for stewardship Feedback antimicrobial susceptibility and use data to clinicians |
| Guidelines and Pathways | Limits variation in therapy of infectious diseases Best evidence‐based Assists in adherence with regulatory and third‐party payer stipulations | Often not utilized unless combined with other stewardship strategies or elements | Best if local data and conditions are used to adapt guidelines to a specific institution |
| Computerized Physician Order Entry and Clinical Decision Support | Shown in limited clinical studies to reduce and modify antimicrobial consumption, improve selected clinical outcomes, and decrease antimicrobial expenditures Once established can greatly assist with implementation of guidelines and best‐evidence therapy. Reduces adverse events related to antimicrobials | Resource intensive during design and implementation Expensive Not readily available | Often entails modification of existing or purchasing of additional informatics |
| Microbiology Interventions | Potential to improve antimicrobial use and anti‐infective therapy for the individual patient | Not well studied | Includes cascade reporting to hide antimicrobial susceptibilities that might promote suboptimal therapy (eg, fluoroquinolone susceptibility for invasive S. aureus) Assistance with choices of automated susceptibility profile, communication of new or changes in testing protocols Preauthorization of susceptibility testing for unconventional antibiotics |
| Rapid Diagnostics | Provides opportunity for early targeted therapy Assists with de‐escalation Shown in very limited studies to decrease antimicrobial consumption and improve clinical outcomes | Not readily available Expensive | Includes PCR and antigen testing of clinical specimens or early culture growth with rapid turnaround of test results |
| Antimicrobial Cycling | Potential to decrease antimicrobial resistance for an institution or geographic unit | Not consistently shown in clinical trials to improve clinical outcomes or decrease resistance Often increases antimicrobial consumption Extremely labor intensive to ensure adherence | Changing antimicrobial protocols periodically in an attempt to reduce selection pressure for resistance |
Prospective Audit With Intervention and Feedback
This approach usually involves the use of an antimicrobial support team that reviews initial or ongoing therapy and then intervenes to provide feedback and suggested modifications to the medical care provider to improve therapy. This can be done by an infectious diseases physician, a clinical pharmacist, or a hospitalist or internist with expertise in antimicrobial therapy. The aim is to provide patient‐specific education and/or suggest changes to antimicrobial utilization (when needed) to improve and streamline therapy. Suggested interventions could include discontinuing or changing 1 or more drugs, switching intravenous to oral drug administration, and suggesting a short‐course duration of therapy. Occasionally, suggestions are made when appropriate to actually escalate or intensify therapy to increase efficacy. Identification of patients for targeting or focusing prospective audit and feedback efforts typically involves using computer surveillance to single out problem antimicrobials or problematic usage, given local resistance patterns or patient characteristics.24 Examples could include a focus on asymptomatic bacteruria, excessive duration of therapy for ventilator‐associated pneumonia, or overzealous use of certain classes of antimicrobials. Another potential activity for a prospective audit and feedback team is to review reports of patient‐specific blood and sterile body fluid culture results matched to the patient's current antimicrobial therapy. This allows for daily review of the appropriateness of therapy for potentially serious infections. Some patients seen by the antibiotic support team may be referred for infectious diseases or other expert consultative opinion if their infections or therapy are felt to be too complicated for routine prospective audit and feedback recommendations.
A number of studies have demonstrated that strategies involving prospective audit with intervention and feedback can improve antibiotic stewardship, as measured by reductions in inappropriate antibiotic use,30 reduced antibiotic costs or overall consumption,3135 greater compliance with hospital treatment guidelines or policies33, 36, 37 and, in some cases, reduced number of infections due to C difficile infection32, 37 or resistant pathogens.31, 32, 37 Prospective audit with feedback is probably the best and most effective core strategy for a community hospital program where other interventions are cumbersome or not as well tolerated by the medical staff. One potential disadvantage of the prospective audit with intervention and feedback approach is that medical provider adherence is largely voluntary. The team can make suggestions, but if the provider disagrees or is unobtainable, the suggestion is never implemented. Also, this strategy can also be resource‐intensive from a personnel perspective.
Formulary Restriction and Preauthorization
The other major strategy used to achieve antimicrobial stewardship goals involves antimicrobial restriction. This can be accomplished either by not including the particular antimicrobial agent on the hospital formulary or by requiring the medical provider to obtain preauthorization before prescribing a restricted drug. A pager system or telephone call is often used for preauthorization, whereby the clinician wishing to prescribe a particular agent calls or pages a member of the stewardship team in order to obtain prescribing permission. When using preauthorization, it is important that the individuals who receive the calls actually see patients and have clinical experience and the respect of the medical staff, as each call may be a mini‐consult. Oftentimes, the provider or prescriber making the call is asking for suggestions as to what antimicrobial might be used, and not simply to obtain authorization to use a drug that is otherwise restricted. Studies have shown that effective interventions supporting stewardship are better provided by attending infectious diseases staff or clinical pharmacists, rather than persons in training.38
Regarding the identification of antimicrobials for restriction, a program should preferentially choose those drugs that are involved in therapy for complex patients and infections. It is also a reasonable approach for drugs that are, or have the potential to be, overused for certain infections where alternatives exist. For work‐horse antimicrobials, those drugs overused or misused for several different infectious diseases, prospective audit and feedback is arguably a better strategy to reduce and modulate consumption.
Formulary restriction and preauthorization is clearly effective in modulating antimicrobial use. A large number of studies have demonstrated reductions in antibiotic drug use, and often in cost, after hospital implementation of a formulary restriction or preauthorization approach to antimicrobial stewardship.3947 It has been more difficult to demonstrate other benefits associated with this approach, although there is some support for its aid in controlling nosocomial infection outbreaks. Restriction of clindamycin48 (or clindamycin, cefotaxime, and vancomycin27) has been shown to control outbreaks of nosocomial C difficileassociated diarrhea and VRE, respectively. More recently, Internet‐based antimicrobial restriction programs49, 50 and a computerized (electronic) approval system51 have been demonstrated to reduce antibiotic use at tertiary hospitals.
Some studies have reported increased antibiotic drug susceptibilities after implementation of institutional preauthorization policies,45, 46, 51 and at least 1 reported a decreased incidence of ceftazidime‐resistant Klebsiella species after instituting a preapproval policy for cephalosporins.52 However, there is concern that restricting 1 class of antibiotics and replacing it with another will simply replace 1 resistant species with another, the so‐called squeezing the balloon effect.53 This was observed in the latter study, where a 44% reduction in ceftazidime‐resistant Klebsiella species at the hospital was accompanied by a 69% increase in incidence of imipenem‐resistant P aeruginosa.52 To assess and enable response to possible squeezing the balloon effects, the guidelines recommend monitoring overall trends in antimicrobial use for institutions using preauthorization strategies.
Possible disadvantages of preauthorization and restriction include perceived loss of autonomy for prescribers, the potential need for all‐hours support, inaccurate or misleading information from the prescriber (leading to inappropriate recommendations),24 and significant delay in stat antimicrobial administration.54 Delay in antimicrobial administration due to the time required to obtain preauthorization and have the approval communicated to the pharmacy was not observed when studied as a process measure at WFUBMC (Ohl, unpublished data, 2008).
A study by Linkin and colleagues showed that 39% of telephone calls for preauthorization of a restricted antimicrobial contained an inaccuracy in at least 1 type of patient data.38 A follow‐up by the same group demonstrated that inaccurate communication was significantly associated with inappropriate antimicrobial recommendations (odds ratio [OR] 2.2; P = .03); this was particularly the case for inaccuracies in microbiologic data (OR 7.5; P = .002).55 Also, if all‐hours support is not provided, at least 1 study has shown some physicians may engage in stealth dosing, that is, avoiding having to obtain preauthorization for restricted antimicrobials by waiting until off‐hours to place orders.56 The latter can be dealt with by following up on such orders with a prospective audit and feedback component of the program. Preauthorization is usually more difficult to employ and less accepted in non‐academic medical centers. Prospective audit and feedback may be more appropriate in such settings.
Supplemental Strategies
A number of additional options are available to supplement the 2 core strategies just described, and are listed in Table 2. Education is generally considered an essential component of any effective antimicrobial stewardship program, but it generally has little lasting impact on providers' behavior, unless it is incorporated with other active interventions.24 In particular, the large group or Grand Roundstype education, where someone describes what needs to be done and why, typically does not produce lasting behavioral changes. There might be, and often is, some short‐term modification, but long‐lasting change at the provider level requires consistent and repeated educational endeavors. Such large group educational venues are more effective and better used as a forum to describe or garner support for an impending stewardship program or intervention, rather than to teach a specific practice.
Using the antimicrobial stewardship program to adapt national guidelines to local antimicrobial use, microbiology, and resistance patterns57, 58 or using clinical (critical) pathways59 has also been shown to improve antimicrobial utilization at hospitals. National guidelines generally enjoy widespread support, but they commonly lack specific information about how to implement recommendations at a given hospital or how to incorporate local data relevant for decision making. A 2006 report by Beardsley and coworkers provides a model from WFUBMC on how local microbiologic data can be used to modify national treatment guidelines to better serve the needs of patients treated at a particular institution.60 Using American Thoracic Society (ATS) and IDSA guidelines for the management of hospital‐acquired pneumonia (HAP), together with local data on the most common bacterial pathogens and their susceptibility to piperacillin‐tazobactam, cefepime, ciprofloxacin, and amikacin (based on length of hospitalization), the WFUBMC CAUSE Advisory Board developed institution‐specific HAP guidelines. The new guidelines divided the ATS/IDSA late onset/risk of the MDR pathogens group of patients into 2 subcategories, early‐late and late‐late pneumonias. Also, unlike the national guidelines, the new guidelines did not recommend ciprofloxacin as empiric therapy, instead recommending amikacin as a component of regimens targeting late‐late pneumonias.
Newer (and in some cases not so new) information technologies can be adapted to healthcare delivery and prescriber support to improve antimicrobial stewardship. These include computer decision support61 and alert systems6265; computerized physician order entry (CPOE)66, 67; electronic medical records24; electronic retrieval of treatment guidelines or clinical texts68; and personal digital assistant (PDA) applications providing information on pathogens, diagnosis, medication, and treatment.68, 69 In addition, computer‐based surveillance64, 70, 71 and Web‐based systems for antimicrobial approval; automated clinical decision support; and/or enhanced real‐time communication between prescribers and other members of the antimicrobial stewardship team show promise for antimicrobial stewardship programs.49, 50
Computer‐assisted decision support has been shown to improve or reduce antibiotic‐susceptibility mismatches (improve selection of effective therapy), overall antibiotic use, excess antimicrobial dosages, excessive‐dose days, selection of antimicrobials for which the patient was poorly matched in terms of allergies, and antimicrobial‐related adverse events, as well as reduce antimicrobial drug costs, total hospital costs, and length of hospital stay.7277 For their part, CPOE systems have been shown to improve compliance with treatment guidelines, decrease medication and other medical errors, shorten length of hospital stay, and decrease pharmaceutical costs.66, 67, 78 In many cases, CPOE systems can now be modified to include some clinical decision support to improve antimicrobial use.78
The IDSA/SHEA guidelines note that antimicrobial decisions can be improved through use of CPOE, clinical decision support, and electronic medical records that enable incorporation of data on patient‐specific microbiology cultures and susceptibilities, hepatic and renal function, drug interactions, allergies, and cost. They also point out that computer‐based surveillance can facilitate good stewardship by enabling more efficient targeting of antimicrobial interventions, tracking of antimicrobial resistance patterns, and identification of HAIs and adverse drug events.24 Recently, a few proprietary informatics programs that perform such functions for the hospital epidemiologist and antimicrobial steward have become available, including but not limited to TheraDoc (Salt Lake City, UT), SafetySurveillor (Premier, Inc., Charlotte, NC), and BD Protect (BD Diagnostics, Austin, TX). Perhaps one of the best‐known comprehensive hospital information systems that incorporates and integrates several information technologies to improve patient care at the level of the prescriber is the Health Evaluation through Logical Processing (HELP) system at LDS Hospital in Salt Lake City, Utah.7981 Unfortunately, these programs are expensive, need considerable time for installation and validation, and do not always perform the functions needed by the medical center. The medical community has generally been slow to incorporate healthcare information technology to improve antimicrobial use or general medical care, but in the last few years more hospitals are finding their merit.
On the basis of evidence currently available, the 2007 guidelines do not recommend the routine use of antimicrobial cycling or combination therapy to prevent or reduce antimicrobial resistance. Such strategies, where at first glance might intuitively seem to make sense, have not been shown to improve patient care, improve antimicrobial choices, or reduce antimicrobial resistance. In addition, antimicrobial cycling in particular is difficult to implement and labor intensive to oversee.24
One strategy for improving antimicrobial stewardship not mentioned in the 2007 IDSA/SHEA guidelines, but might become increasingly important in the future, is the use of rapid molecular diagnostic testing. Knowing the identity of the causative pathogen sooner or being able to rapidly rule out certain pathogens should enable better decision‐making. During the 2009/2010 influenza season with H1N1 influenza, WFUBMC was able to implement rapid viral testing and learned some things that enabled improvement of hospital practices. It was found that approximately 10% to 15% of the pneumonias in immunocompromised patients at the center were not bacterial but viral, the pathogens being respiratory syncytial virus (RSV) or metapneumovirus (Ohl, unpublished data, 2010). Upon finding a viral etiology to a lower respiratory tract infection, rapid de‐escalation of antibiotic therapy was possible. If rapid diagnostics are to be performed, it is important that there are systems in place to respond quickly to the findings, so the benefits of having rapid data can be realized.
Evaluating Antimicrobial Stewardship Programs
Two general types of measures are used to evaluate the effectiveness of antimicrobial stewardship: process and outcome. As with most things done in the hospital, process measures are easier. They measure surrogate impacts of a program, accountability, resource use, and cost effectiveness. In essence, process measures evaluate whether the program accomplished what it set out to do in terms of changing certain processes or prescriber behaviors. It is important to measure resource use, as this helps to continue funding and to keep workers involved in the project. Good programs will save money; this can easily be measured, even if it is just as simple as going to the hospital pharmacy and looking at the cost of antimicrobials provided per patient day.
Outcomes like decreases in particular infections, less emergence of antimicrobial resistance, or other patient‐specific measures are likely more important in the big picture, but they are also much more difficult to measure. For example, where one would like to measure changes in pathogen resistance after making some changes in antimicrobial stewardship, it often takes years before the benefits of a particular intervention or change materialize in terms of less resistance or reduced emergence of resistance. If that type of change is to be measured, then one needs to be persistent and continue measurements over a long period of time. In addition, given the protracted amount of time before these outcomes may be observed, a number of other changes are likely to happen that coincide with the antimicrobial stewardship interventions and make assessment of causality difficult and biased.
Having said that, a number of studies have demonstrated a relationship between antibiotic restriction48, 8285 or other antimicrobial stewardship policies32, 86 and decreases in nosocomial C difficile infections or disease. Figure 2 illustrates the impact of a nonrestrictive antimicrobial stewardship program at a secondary/tertiary‐care hospital in Quebec, Canada, on an epidemic of C difficileassociated disease (CDAD) that occurred at the institution during the latter portion of 2003.86 Following program implementation, and the major drop in targeted antibiotic consumption, the incidence of CDAD also significantly decreased. Earlier implementation of infection control measures had no effect on CDAD incidence.

A smaller number of studies have reported decreases in resistant gram‐negative bacteria following implementation of antimicrobial stewardship programs. For example, Meyer and colleagues reported a marked reduction in ceftazidime‐resistant K pneumoniae at a 487‐bed general hospital in New York City after implementation of enhanced ceftazidime restriction and barrier precautions following an outbreak of infections caused by the resistant K pneumoniae.87 Similarly, Carling and coworkers reported a significant decrease in nosocomial infections caused by resistant Enterobacteriaceae following implementation of a multidisciplinary antibiotic stewardship program to minimize inappropriate use of third‐generation cephalosporins (Figure 3).32 More recently, a retrospective, longitudinal, multicenter analysis of a consortium of 22 academic health centers in the United States showed that incidence rates of carbapenem‐resistant P aeruginosa were lower at hospitals that restricted carbapenems than those that did not (P = .01).88

Evidence suggesting a beneficial impact of antimicrobial stewardship programs on resistance in gram‐positive organisms is limited. More specifically, the study by Carling and colleagues reported an apparent decrease in VRE rates following implementation of their program to reduce inappropriate use of third‐generation cephalosporins.32 The hospital had VRE rates similar to other NNIS System hospitals prior to beginning the program, but after antibiotic stewardship measures were implemented, the VRE rate began to drop, falling to 6% by 1999. This should be compared with a VRE rate of 24% for similar NNIS System hospitals in 1999.
As far as reducing healthcare costs, Figure 4A illustrates the direct antimicrobial cost savings at WFUBMC after implementation of the CAUSE antimicrobial stewardship program, and Figure 4B after supplemental interventions were implemented. Although decreasing antimicrobial cost is important, one would like to show decreases in overall healthcare costs associated with an antimicrobial stewardship program. Unfortunately, this is often a little more difficult to demonstrate. Extrapolations, however, may be possible. Because antimicrobial resistance, adverse drug effects, and secondary unintended infections such as C difficile colitis have been linked with increased patient morbidity and mortality, longer hospital stays, and increased healthcare costs,6, 89, 90 improved antimicrobial stewardship is expected to optimize patient care and lower overall healthcare costs. A study in a large tertiary care academic medical center estimated more than $4.25 million in total healthcare savings over 1 year with a stewardship program using both preauthorization and, to a lesser extent, prospective audit and feedback.91 Despite the fact cost saving should not be a primary goal of an antimicrobial stewardship program, lower antimicrobial costs can help keep a program funded and buttress a proposal for an antimicrobial stewardship program to hospital leadership.

Many hospitals recognize other areas where an antimicrobial stewardship program can demonstrate its value. This includes implementation of a rapid change in drug utilization during antimicrobial supply shortages and assistance with regulatory mandates and surgical infection prophylaxis. Stewardship teams often assist microbiology with protocols for microbiology reporting, antibiograms, planning for susceptibility testing, and communicating changes in microbiology tests or protocols to clinicians.
Conclusions
Overuse or misuse of antibiotics and other antimicrobials for hospital inpatients is relatively common, and can be associated with several unintended negative consequences. Improving medical care necessarily includes better use of antimicrobials to optimize outcomes and preserve the effectiveness of currently available agents. Further, an important additional consequence of effective antimicrobial stewardship and improved patient care is typically a lowering of overall healthcare costs. The recent 2007 IDSA/SHEA guidelines provide recommendations for developing an institutional program to enhance antimicrobial stewardship. However, individual institutions need to look closely at their own systems and patients to develop an antimicrobial stewardship program that best serves the needs of their hospital and the people it serves.
- ,, et al.Estimating health care‐associated infections and deaths in U.S. hospitals, 2002.Public Health Rep.2007;122:160–166.
- ,,.Healthcare‐associated infection in Shiraz, Iran 2004–2005.J Hosp Infect.2008;69:283–287.
- ,,,,.Healthcare‐associated infections in Finnish acute care hospitals: a national prevalence survey, 2005.J Hosp Infect.2008;69:288–294.
- .Historical and changing epidemiology of healthcare‐associated infections.J Hosp Infect.2009;73:296–304.
- ,,, et al.Four country healthcare associated infection prevalence survey 2006: overview of the results.J Hosp Infect.2008;69:230–248.
- ,,.Clinical and economic burden of antimicrobial resistance.Expert Rev Anti Infect Ther.2008;6:751–763.
- National Nosocomial Infections Surveillance (NNIS) System Report, data summary from January 1992 through June 2004, issued October 2004.Am J Infect Control.2004;32:470–485.
- .Bad bugs, no drugs: no ESCAPE revisited.Clin Infect Dis.2009;49:992–993.
- .Federal funding for the study of antimicrobial resistance in nosocomial pathogens: no ESKAPE.J Infect Dis.2008;197:1079–1081.
- ,.Resistant gram‐negative bacilli: a neglected healthcare crisis?Am J Health Syst Pharm.2007;64:S3–S21; quizS22–S24.
- ,,, et al.Antimicrobial resistance among gram‐negative bacilli causing infections in intensive care unit patients in the United States between 1993 and 2004.J Clin Microbiol.2007;45:3352–3359.
- ,,.The emerging threat of multidrug‐resistant gram‐negative organisms in long‐term care facilities.J Gerontol A Biol Sci Med Sci.2009;64:138–141.
- ,,,,.Influx of multidrug‐resistant, gram‐negative bacteria in the hospital setting and the role of elderly patients with bacterial bloodstream infection.Infect Control Hosp Epidemiol.2009;30:325–331.
- ,,.Emergence of extensively drug‐resistant and pandrug‐resistant Gram‐negative bacilli in Europe.Euro Surveill.2008;13(47)pii:19045.
- ,,, et al.The epidemic of antibiotic‐resistant infections: a call to action for the medical community from the Infectious Diseases Society of America.Clin Infect Dis.2008;46:155–164.
- ,,,,,.Bad bugs need drugs: an update on the development pipeline from the Antimicrobial Availability Task Force of the Infectious Diseases Society of America.Clin Infect Dis.2006;42:657–668.
- ,.Observations on spiraling empiricism: its causes, allure, and perils, with particular reference to antibiotic therapy.Am J Med.1989;87:201–206.
- ,.Variability in rates of use of antibacterials among 130 US hospitals and risk‐adjustment models for interhospital comparison.Infect Control Hosp Epidemiol.2008;29:203–211.
- ,,,.Trends in antibacterial use in US academic health centers: 2002 to 2006.Arch Intern Med.2008;168:2254–2260.
- ,,,,.Unnecessary use of antimicrobials in hospitalized patients: current patterns of misuse with an emphasis on the antianaerobic spectrum of activity.Arch Intern Med.2003;163:972–978.
- Polk RE, Fishman NO, eds.Antimicrobial Stewardship.7th ed.Philadelphia, PA:Churchill Livingstone Elsevier;2010. Mandell GL, Bennett JE, Dolin R, eds. Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases; No. 1.
- .Collateral damage and what the future might hold. The need to balance prudent antibiotic utilization and stewardship with effective patient management.Int J Infect Dis.2006;10:S17–S24.
- ,,, et al.A predominantly clonal multi‐institutional outbreak of Clostridium difficile‐associated diarrhea with high morbidity and mortality.N Engl J Med.2005;353:2442–2449.
- ,, et al.Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship.Clin Infect Dis.2007;44:159–177.
- .Antimicrobial stewardship.Semin Infect Contr.2001;1:210–221.
- ,,,,.Insights from the Society of Infectious Diseases Pharmacists on antimicrobial stewardship guidelines from the Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America.Pharmacotherapy.2009;29:593–607.
- ,,,,,.Manipulation of a hospital antimicrobial formulary to control an outbreak of vancomycin‐resistant enterococci.Clin Infect Dis.1996;23:1020–1025.
- MAD‐ID: Making a Difference in Infectious Diseases Pharmacotherapy. Available at http://www.mad‐id.com. Accessed August 17,2010.
- Society of Infectious Diseases Pharmacists. The implementation of antimicrobial stewardship using a multidisciplinary approach. CME program. Available at http://www.esymposia.ashp.org/cemantimicrobial/. Accessed August 17,2010.
- ,,, et al.Academic detailing to improve use of broad‐spectrum antibiotics at an academic medical center.Arch Intern Med.2001;161:1897–1902.
- ,,, et al.A hospitalwide intervention program to optimize the quality of antibiotic use: impact on prescribing practice, antibiotic consumption, cost savings, and bacterial resistance.Clin Infect Dis.2003;37:180–186.
- ,,,,.Favorable impact of a multidisciplinary antibiotic management program conducted during 7 years.Infect Control Hosp Epidemiol.2003;24:699–706.
- ,,, et al.Antimicrobial stewardship program directed at broad‐spectrum intravenous antibiotics prescription in a tertiary hospital.Eur J Clin Microbiol Infect Dis.2009;28:1447–1456.
- ,,,,,.Antibiotic optimization: an evaluation of patient safety and economic outcomes.Arch Intern Med.1997;157:1689–1694.
- .Concurrent antibiotic review programs: a role for infectious diseases specialists at small community hospitals.Clin Infect Dis.2003;37:742–743.
- ,,,,.Improving antimicrobial use in the hospital setting by providing usage feedback to prescribing physicians.Infect Control Hosp Epidemiol.2006;27:378–382.
- ,,, et al.Successful use of feedback to improve antibiotic prescribing and reduce Clostridium difficile infection: a controlled interrupted time series.J Antimicrob Chemother.2007;59:990–995.
- ,,,,.Inaccurate communications in telephone calls to an antimicrobial stewardship program.Infect Control Hosp Epidemiol.2006;27:688–694.
- ,,,,.Effect of a vancomycin restriction policy on ordering practices during an outbreak of vancomycin‐resistant Enterococcus faecium.Arch Intern Med.1997;157:1132–1136.
- ,.Impact of voluntary vs enforced compliance of third‐generation cephalosporin use in a teaching hospital.Arch Intern Med.1992;152:554–557.
- ,,.Cost containment through restriction of cephalosporins.Am J Hosp Pharm.1981;38:1897–1900.
- ,.Controlling cephalosporin and aminoglycoside costs through pharmacy and therapeutics committee restrictions.Am J Hosp Pharm.1985;42:1343–1347.
- ,.Enforcing a policy for restricting antimicrobial drug use.Am J Health Syst Pharm.1995;52:1433–1435.
- ,,,,,.Changes in antibiotic use, cost and consumption after an antibiotic restriction policy applied by infectious disease specialists.Jpn J Infect Dis.2005;58:338–343.
- ,,,,,.Impact of an antibiotic restriction policy on hospital expenditures and bacterial susceptibilities: a lesson from a pediatric institution in a developing country.Pediatr Infect Dis J.2000;19:200–206.
- ,,,,,.Effects of requiring prior authorization for selected antimicrobials: expenditures, susceptibilities, and clinical outcomes.Clin Infect Dis.1997;25:230–239.
- ,,,.Antibiotic cost savings from formulary restrictions and physician monitoring in a medical‐school‐affiliated hospital.Am J Med.1987;83:817–823.
- ,,,,.Decrease in nosocomial Clostridium difficile‐associated diarrhea by restricting clindamycin use.Ann Intern Med.1994;120:272–277.
- ,,, et al.A World Wide Web‐based antimicrobial stewardship program improves efficiency, communication, and user satisfaction and reduces cost in a tertiary care pediatric medical center.Clin Infect Dis.2008;47:747–753.
- ,,, et al.Impact of a web‐based antimicrobial approval system on broad‐spectrum cephalosporin use at a teaching hospital.Med J Aust.2003;178:386–390.
- ,,, et al.Electronic antibiotic stewardship: reduced consumption of broad‐spectrum antibiotics using a computerized antimicrobial approval system in a hospital setting.J Antimicrob Chemother.2008;62:608–616.
- ,,, et al.Class restriction of cephalosporin use to control total cephalosporin resistance in nosocomial Klebsiella.JAMA.1998;280:1233–1237.
- .Antibiotic resistance: squeezing the balloon?JAMA.1998;280:1270–1271.
- ,,.Impact of a restrictive antimicrobial policy on the process and timing of antimicrobial administration.J Hosp Med.2010;5:E41–E45.
- ,,, et al.Effect of communication errors during calls to an antimicrobial stewardship program.Infect Control Hosp Epidemiol.2007;28:1374–1381.
- ,,,,,.Evaluation of antimicrobial therapy orders circumventing an antimicrobial stewardship program: investigating the strategy of “stealth dosing”.Infect Control Hosp Epidemiol.2007;28:551–556.
- ,,,,,.Experience with a clinical guideline for the treatment of ventilator‐associated pneumonia.Crit Care Med.2001;29:1109–1115.
- ,,,,.Short‐course empiric antibiotic therapy for patients with pulmonary infiltrates in the intensive care unit: a proposed solution for indiscriminate antibiotic prescription.Am J Respir Crit Care Med.2000;162:505–511.
- ,,,,,.A controlled trial of a critical pathway for treatment of community‐acquired pneumonia. CAPITAL Study Investigators. Community‐Acquired Pneumonia Intervention Trial Assessing Levofloxacin.JAMA.2000;283:749–755.
- ,,,,,.Using local microbiologic data to develop institution‐specific guidelines for the treatment of hospital‐acquired pneumonia.Chest.2006;130:787–793.
- .Expert clinical decision support systems to enhance antimicrobial stewardship programs: insights from the Society of Infectious Diseases Pharmacists.Pharmacotherapy.2005;25:1116–1125.
- ,,,,,.Improved perioperative antibiotic use and reduced surgical wound infections through use of computer decision analysis.Infect Control Hosp Epidemiol.1989;10:316–320.
- ,,,,.Impact of a computer‐generated alert system prompting review of antibiotic use in hospitals.J Antimicrob Chemother.2009;63:1058–1063.
- ,,,,.Therapeutic antibiotic monitoring: surveillance using a computerized expert system.Am J Med.1990;88:43–48.
- ,,,,.Improvement of intraoperative antibiotic prophylaxis in prolonged cardiac surgery by automated alerts in the operating room.Infect Control Hosp Epidemiol.2003;24:13–16.
- ,.Computer physician order entry: benefits, costs, and issues.Ann Intern Med.2003;139:31–39.
- .Computerized physician order entry in the critical care and general inpatient setting: a narrative review.J Crit Care.2004;19:271–278.
- ,,.Information technology for optimizing the management of infectious diseases.Am J Health Syst Pharm.2006;63:957–965.
- ,,.Personal digital assistant infectious diseases applications for health care professionals.Clin Infect Dis.2003;36:1018–1029.
- ,,,.Computerized surveillance of adverse drug events in hospital patients.JAMA.1991;266:2847–2851.
- ,,, et al.Computer surveillance of hospital‐acquired infections and antibiotic use.JAMA.1986;256:1007–1011.
- ,,,,.Improving empiric antibiotic selection using computer decision support.Arch Intern Med.1994;154:878–884.
- ,,,.Evaluation of a computer‐assisted antibiotic‐dose monitor.Ann Pharmacother.1999;33:1026–1031.
- ,,, et al.A computer‐assisted management program for antibiotics and other antiinfective agents.N Engl J Med.1998;338:232–238.
- ,,, et al.Impact of a computerized clinical decision support system on reducing inappropriate antimicrobial use: a randomized controlled trial.J Am Med Inform Assoc.2006;13:378–384.
- ,,,.Development and impact of a computerized pediatric antiinfective decision support program.Pediatrics.2001;108:E75.
- ,,,,.Computerized antimicrobial decision support: an offline evaluation of a database‐driven empiric antimicrobial guidance program in hospitalized patients with a bloodstream infection.Int J Med Inform.2004;73:455–460.
- ,,, et al.The impact of computerized physician order entry on medication error prevention.J Am Med Inform Assoc.1999;6:313–321.
- .Maximizing appropriate antibiotic prophylaxis for surgical patients: an update from LDS Hospital, Salt Lake City.Clin Infect Dis.2001;33Suppl 2:S78–S83.
- ,,,,.The HELP system and its application to infection control.J Hosp Infect.1991;18Suppl A:424–431.
- ,,.Decision support in medicine: lessons from the HELP system.Int J Med Inform.2003;69:273–284.
- ,,,,,.Hospital‐wide restriction of clindamycin: effect on the incidence of Clostridium difficile‐associated diarrhea and cost.Ann Intern Med.1998;128:989–995.
- ,,,,,.An antibiotic policy associated with reduced risk of Clostridium difficile‐associated diarrhoea.Age Ageing.1999;28:578–580.
- ,,, et al.Successful control of Clostridium difficile infection in an elderly care unit through use of a restrictive antibiotic policy.J Antimicrob Chemother.1997;40:707–711.
- ,,,,,.Antibiotic prescribing policy and Clostridium difficile diarrhoea.QJM.2004;97:423–429.
- ,,,,.Impact of a reduction in the use of high‐risk antibiotics on the course of an epidemic of Clostridium difficile‐associated disease caused by the hypervirulent NAP1/027 strain.Clin Infect Dis.2007;45Suppl 2:S112–S121.
- ,,,,.Nosocomial outbreak of Klebsiella infection resistant to late‐generation cephalosporins.Ann Intern Med.1993;119:353–358.
- ,,.Relationship of carbapenem restriction in 22 university teaching hospitals to carbapenem use and carbapenem‐resistant Pseudomonas aeruginosa.Antimicrob Agents Chemother.2009;53:1983–1986.
- .The relationship between antimicrobial resistance and patient outcomes: mortality, length of hospital stay, and health care costs.Clin Infect Dis.2006;42Suppl 2:S82–S89.
- ,,, et al.Hospital and societal costs of antimicrobial‐resistant infections in a Chicago teaching hospital: implications for antibiotic stewardship.Clin Infect Dis.2009;49:1175–1184.
- .Antimicrobial stewardship.Am J Med.2006;119:S53–S61; discussionS62–S70.
- ,, et al.Estimating health care‐associated infections and deaths in U.S. hospitals, 2002.Public Health Rep.2007;122:160–166.
- ,,.Healthcare‐associated infection in Shiraz, Iran 2004–2005.J Hosp Infect.2008;69:283–287.
- ,,,,.Healthcare‐associated infections in Finnish acute care hospitals: a national prevalence survey, 2005.J Hosp Infect.2008;69:288–294.
- .Historical and changing epidemiology of healthcare‐associated infections.J Hosp Infect.2009;73:296–304.
- ,,, et al.Four country healthcare associated infection prevalence survey 2006: overview of the results.J Hosp Infect.2008;69:230–248.
- ,,.Clinical and economic burden of antimicrobial resistance.Expert Rev Anti Infect Ther.2008;6:751–763.
- National Nosocomial Infections Surveillance (NNIS) System Report, data summary from January 1992 through June 2004, issued October 2004.Am J Infect Control.2004;32:470–485.
- .Bad bugs, no drugs: no ESCAPE revisited.Clin Infect Dis.2009;49:992–993.
- .Federal funding for the study of antimicrobial resistance in nosocomial pathogens: no ESKAPE.J Infect Dis.2008;197:1079–1081.
- ,.Resistant gram‐negative bacilli: a neglected healthcare crisis?Am J Health Syst Pharm.2007;64:S3–S21; quizS22–S24.
- ,,, et al.Antimicrobial resistance among gram‐negative bacilli causing infections in intensive care unit patients in the United States between 1993 and 2004.J Clin Microbiol.2007;45:3352–3359.
- ,,.The emerging threat of multidrug‐resistant gram‐negative organisms in long‐term care facilities.J Gerontol A Biol Sci Med Sci.2009;64:138–141.
- ,,,,.Influx of multidrug‐resistant, gram‐negative bacteria in the hospital setting and the role of elderly patients with bacterial bloodstream infection.Infect Control Hosp Epidemiol.2009;30:325–331.
- ,,.Emergence of extensively drug‐resistant and pandrug‐resistant Gram‐negative bacilli in Europe.Euro Surveill.2008;13(47)pii:19045.
- ,,, et al.The epidemic of antibiotic‐resistant infections: a call to action for the medical community from the Infectious Diseases Society of America.Clin Infect Dis.2008;46:155–164.
- ,,,,,.Bad bugs need drugs: an update on the development pipeline from the Antimicrobial Availability Task Force of the Infectious Diseases Society of America.Clin Infect Dis.2006;42:657–668.
- ,.Observations on spiraling empiricism: its causes, allure, and perils, with particular reference to antibiotic therapy.Am J Med.1989;87:201–206.
- ,.Variability in rates of use of antibacterials among 130 US hospitals and risk‐adjustment models for interhospital comparison.Infect Control Hosp Epidemiol.2008;29:203–211.
- ,,,.Trends in antibacterial use in US academic health centers: 2002 to 2006.Arch Intern Med.2008;168:2254–2260.
- ,,,,.Unnecessary use of antimicrobials in hospitalized patients: current patterns of misuse with an emphasis on the antianaerobic spectrum of activity.Arch Intern Med.2003;163:972–978.
- Polk RE, Fishman NO, eds.Antimicrobial Stewardship.7th ed.Philadelphia, PA:Churchill Livingstone Elsevier;2010. Mandell GL, Bennett JE, Dolin R, eds. Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases; No. 1.
- .Collateral damage and what the future might hold. The need to balance prudent antibiotic utilization and stewardship with effective patient management.Int J Infect Dis.2006;10:S17–S24.
- ,,, et al.A predominantly clonal multi‐institutional outbreak of Clostridium difficile‐associated diarrhea with high morbidity and mortality.N Engl J Med.2005;353:2442–2449.
- ,, et al.Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship.Clin Infect Dis.2007;44:159–177.
- .Antimicrobial stewardship.Semin Infect Contr.2001;1:210–221.
- ,,,,.Insights from the Society of Infectious Diseases Pharmacists on antimicrobial stewardship guidelines from the Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America.Pharmacotherapy.2009;29:593–607.
- ,,,,,.Manipulation of a hospital antimicrobial formulary to control an outbreak of vancomycin‐resistant enterococci.Clin Infect Dis.1996;23:1020–1025.
- MAD‐ID: Making a Difference in Infectious Diseases Pharmacotherapy. Available at http://www.mad‐id.com. Accessed August 17,2010.
- Society of Infectious Diseases Pharmacists. The implementation of antimicrobial stewardship using a multidisciplinary approach. CME program. Available at http://www.esymposia.ashp.org/cemantimicrobial/. Accessed August 17,2010.
- ,,, et al.Academic detailing to improve use of broad‐spectrum antibiotics at an academic medical center.Arch Intern Med.2001;161:1897–1902.
- ,,, et al.A hospitalwide intervention program to optimize the quality of antibiotic use: impact on prescribing practice, antibiotic consumption, cost savings, and bacterial resistance.Clin Infect Dis.2003;37:180–186.
- ,,,,.Favorable impact of a multidisciplinary antibiotic management program conducted during 7 years.Infect Control Hosp Epidemiol.2003;24:699–706.
- ,,, et al.Antimicrobial stewardship program directed at broad‐spectrum intravenous antibiotics prescription in a tertiary hospital.Eur J Clin Microbiol Infect Dis.2009;28:1447–1456.
- ,,,,,.Antibiotic optimization: an evaluation of patient safety and economic outcomes.Arch Intern Med.1997;157:1689–1694.
- .Concurrent antibiotic review programs: a role for infectious diseases specialists at small community hospitals.Clin Infect Dis.2003;37:742–743.
- ,,,,.Improving antimicrobial use in the hospital setting by providing usage feedback to prescribing physicians.Infect Control Hosp Epidemiol.2006;27:378–382.
- ,,, et al.Successful use of feedback to improve antibiotic prescribing and reduce Clostridium difficile infection: a controlled interrupted time series.J Antimicrob Chemother.2007;59:990–995.
- ,,,,.Inaccurate communications in telephone calls to an antimicrobial stewardship program.Infect Control Hosp Epidemiol.2006;27:688–694.
- ,,,,.Effect of a vancomycin restriction policy on ordering practices during an outbreak of vancomycin‐resistant Enterococcus faecium.Arch Intern Med.1997;157:1132–1136.
- ,.Impact of voluntary vs enforced compliance of third‐generation cephalosporin use in a teaching hospital.Arch Intern Med.1992;152:554–557.
- ,,.Cost containment through restriction of cephalosporins.Am J Hosp Pharm.1981;38:1897–1900.
- ,.Controlling cephalosporin and aminoglycoside costs through pharmacy and therapeutics committee restrictions.Am J Hosp Pharm.1985;42:1343–1347.
- ,.Enforcing a policy for restricting antimicrobial drug use.Am J Health Syst Pharm.1995;52:1433–1435.
- ,,,,,.Changes in antibiotic use, cost and consumption after an antibiotic restriction policy applied by infectious disease specialists.Jpn J Infect Dis.2005;58:338–343.
- ,,,,,.Impact of an antibiotic restriction policy on hospital expenditures and bacterial susceptibilities: a lesson from a pediatric institution in a developing country.Pediatr Infect Dis J.2000;19:200–206.
- ,,,,,.Effects of requiring prior authorization for selected antimicrobials: expenditures, susceptibilities, and clinical outcomes.Clin Infect Dis.1997;25:230–239.
- ,,,.Antibiotic cost savings from formulary restrictions and physician monitoring in a medical‐school‐affiliated hospital.Am J Med.1987;83:817–823.
- ,,,,.Decrease in nosocomial Clostridium difficile‐associated diarrhea by restricting clindamycin use.Ann Intern Med.1994;120:272–277.
- ,,, et al.A World Wide Web‐based antimicrobial stewardship program improves efficiency, communication, and user satisfaction and reduces cost in a tertiary care pediatric medical center.Clin Infect Dis.2008;47:747–753.
- ,,, et al.Impact of a web‐based antimicrobial approval system on broad‐spectrum cephalosporin use at a teaching hospital.Med J Aust.2003;178:386–390.
- ,,, et al.Electronic antibiotic stewardship: reduced consumption of broad‐spectrum antibiotics using a computerized antimicrobial approval system in a hospital setting.J Antimicrob Chemother.2008;62:608–616.
- ,,, et al.Class restriction of cephalosporin use to control total cephalosporin resistance in nosocomial Klebsiella.JAMA.1998;280:1233–1237.
- .Antibiotic resistance: squeezing the balloon?JAMA.1998;280:1270–1271.
- ,,.Impact of a restrictive antimicrobial policy on the process and timing of antimicrobial administration.J Hosp Med.2010;5:E41–E45.
- ,,, et al.Effect of communication errors during calls to an antimicrobial stewardship program.Infect Control Hosp Epidemiol.2007;28:1374–1381.
- ,,,,,.Evaluation of antimicrobial therapy orders circumventing an antimicrobial stewardship program: investigating the strategy of “stealth dosing”.Infect Control Hosp Epidemiol.2007;28:551–556.
- ,,,,,.Experience with a clinical guideline for the treatment of ventilator‐associated pneumonia.Crit Care Med.2001;29:1109–1115.
- ,,,,.Short‐course empiric antibiotic therapy for patients with pulmonary infiltrates in the intensive care unit: a proposed solution for indiscriminate antibiotic prescription.Am J Respir Crit Care Med.2000;162:505–511.
- ,,,,,.A controlled trial of a critical pathway for treatment of community‐acquired pneumonia. CAPITAL Study Investigators. Community‐Acquired Pneumonia Intervention Trial Assessing Levofloxacin.JAMA.2000;283:749–755.
- ,,,,,.Using local microbiologic data to develop institution‐specific guidelines for the treatment of hospital‐acquired pneumonia.Chest.2006;130:787–793.
- .Expert clinical decision support systems to enhance antimicrobial stewardship programs: insights from the Society of Infectious Diseases Pharmacists.Pharmacotherapy.2005;25:1116–1125.
- ,,,,,.Improved perioperative antibiotic use and reduced surgical wound infections through use of computer decision analysis.Infect Control Hosp Epidemiol.1989;10:316–320.
- ,,,,.Impact of a computer‐generated alert system prompting review of antibiotic use in hospitals.J Antimicrob Chemother.2009;63:1058–1063.
- ,,,,.Therapeutic antibiotic monitoring: surveillance using a computerized expert system.Am J Med.1990;88:43–48.
- ,,,,.Improvement of intraoperative antibiotic prophylaxis in prolonged cardiac surgery by automated alerts in the operating room.Infect Control Hosp Epidemiol.2003;24:13–16.
- ,.Computer physician order entry: benefits, costs, and issues.Ann Intern Med.2003;139:31–39.
- .Computerized physician order entry in the critical care and general inpatient setting: a narrative review.J Crit Care.2004;19:271–278.
- ,,.Information technology for optimizing the management of infectious diseases.Am J Health Syst Pharm.2006;63:957–965.
- ,,.Personal digital assistant infectious diseases applications for health care professionals.Clin Infect Dis.2003;36:1018–1029.
- ,,,.Computerized surveillance of adverse drug events in hospital patients.JAMA.1991;266:2847–2851.
- ,,, et al.Computer surveillance of hospital‐acquired infections and antibiotic use.JAMA.1986;256:1007–1011.
- ,,,,.Improving empiric antibiotic selection using computer decision support.Arch Intern Med.1994;154:878–884.
- ,,,.Evaluation of a computer‐assisted antibiotic‐dose monitor.Ann Pharmacother.1999;33:1026–1031.
- ,,, et al.A computer‐assisted management program for antibiotics and other antiinfective agents.N Engl J Med.1998;338:232–238.
- ,,, et al.Impact of a computerized clinical decision support system on reducing inappropriate antimicrobial use: a randomized controlled trial.J Am Med Inform Assoc.2006;13:378–384.
- ,,,.Development and impact of a computerized pediatric antiinfective decision support program.Pediatrics.2001;108:E75.
- ,,,,.Computerized antimicrobial decision support: an offline evaluation of a database‐driven empiric antimicrobial guidance program in hospitalized patients with a bloodstream infection.Int J Med Inform.2004;73:455–460.
- ,,, et al.The impact of computerized physician order entry on medication error prevention.J Am Med Inform Assoc.1999;6:313–321.
- .Maximizing appropriate antibiotic prophylaxis for surgical patients: an update from LDS Hospital, Salt Lake City.Clin Infect Dis.2001;33Suppl 2:S78–S83.
- ,,,,.The HELP system and its application to infection control.J Hosp Infect.1991;18Suppl A:424–431.
- ,,.Decision support in medicine: lessons from the HELP system.Int J Med Inform.2003;69:273–284.
- ,,,,,.Hospital‐wide restriction of clindamycin: effect on the incidence of Clostridium difficile‐associated diarrhea and cost.Ann Intern Med.1998;128:989–995.
- ,,,,,.An antibiotic policy associated with reduced risk of Clostridium difficile‐associated diarrhoea.Age Ageing.1999;28:578–580.
- ,,, et al.Successful control of Clostridium difficile infection in an elderly care unit through use of a restrictive antibiotic policy.J Antimicrob Chemother.1997;40:707–711.
- ,,,,,.Antibiotic prescribing policy and Clostridium difficile diarrhoea.QJM.2004;97:423–429.
- ,,,,.Impact of a reduction in the use of high‐risk antibiotics on the course of an epidemic of Clostridium difficile‐associated disease caused by the hypervirulent NAP1/027 strain.Clin Infect Dis.2007;45Suppl 2:S112–S121.
- ,,,,.Nosocomial outbreak of Klebsiella infection resistant to late‐generation cephalosporins.Ann Intern Med.1993;119:353–358.
- ,,.Relationship of carbapenem restriction in 22 university teaching hospitals to carbapenem use and carbapenem‐resistant Pseudomonas aeruginosa.Antimicrob Agents Chemother.2009;53:1983–1986.
- .The relationship between antimicrobial resistance and patient outcomes: mortality, length of hospital stay, and health care costs.Clin Infect Dis.2006;42Suppl 2:S82–S89.
- ,,, et al.Hospital and societal costs of antimicrobial‐resistant infections in a Chicago teaching hospital: implications for antibiotic stewardship.Clin Infect Dis.2009;49:1175–1184.
- .Antimicrobial stewardship.Am J Med.2006;119:S53–S61; discussionS62–S70.