User login
Studying Documentation
In 1968, Weed highlighted the importance of medical documentation when he proposed a single format for notes.[1, 2] Since then, sweeping changes in the technology, the purposes, and the requirements of clinical record keeping have encouraged steady growth of a literature devoted to the chart. Specifically, over the past half century, computers, lawsuits, regulations, and the use of documentation as a tool of billing have all contributed to the transformation of hospital records. In addition, mounting pressure to shorten inpatient stays, the vastly increased complexity of care, and a growing number of diagnostic possibilities have combined to make medical documentation far more prolific and far less leisurely. All these changes have stimulated a boom in documentation research coinciding, productively, with an era of rapid advances in the conduct of clinical trials and statistical rigor. However, in important respects research into medical documentation today is not asking the right questions, either in the formulation of hypotheses or in the choice of methodology. Forms of clinical communication that do not involve order sets or notes are widespread, growing in sophistication, and increasingly relevant to new concepts of healthcare as a team enterprise; but documentation research has not embraced this development. At the same time, methodologically, the field suffers from a persistent professional bias in the choice of research outcomes, a bias that limits the interpretation of results by neglecting what happens to the patient.
In assessing the chart as a communication device and the effect of changes in documentation, it is increasingly necessary to study direct interpersonal communication as an alternative and partner to writing notes. In particular, 3 recent developments in healthcare emphasize the importance of broadening our concepts of clinical communication. First, the need for discussion in the medical record has become less pressing because of technical improvements in person‐to‐person communication. Second, the electronic health record, by creating discipline‐defined chart views, has helped equalize the stature of different healthcare disciplines but also Balkanized the chart, making direct interdisciplinary communication more necessary. Third, changes in reimbursement are redefining medical goals in such a way that only teams of healthcare providers in close and constant personal communication can achieve them.
Rapid adoption of electronic health records has encouraged researchers studying documentation or information technology to focus on computer formats as defining the range of all possible communication strategies. And certainly there is a broad range of formats: electronic progress notes may be free text or multiple choice, typed or dictated, copy forwarded or composed daily, institutionally templated or self‐templated, furnished with or free from prompts and pop‐ups. However, it is not only, and perhaps not even principally, the electronic record that has changed how clinicians communicate with each other. The technology of discussion over the last 2 decades has become instant, utterly mobile, device independent, and capable of connecting all the patient's caregivers at once to each other and to the medical record in text, picture, and sound. That the same communications upheaval has visited practically every other aspect of our lives diminishes perhaps the visibility of this new virtual team in healthcare but not its importance.
The electronic record certainly plays a role in facilitating communication, through simultaneous chart access and in many other ways, but even more significant is the effect that computerization has had on equalizing the roles of different disciplines and by doing so in fragmenting the medical record. A computerized record expands and reorganizes the chart, changing it from a single authoritative book read by all to an almost limitless array of chart views, each read by some. All viewers (patient, clinician or researcher, administrator, reviewer or coder) can, with equal claim to consulting the chart, categorize, compare, combine, and format data elements from 1 or many encounters, whether inpatient or ambulatory. Typically, an electronic item of patient information may have several authors and uses but has no owner. Data are entered by protocol and in different guises into many aspects of patient care as components of notes, flow sheets, summaries, pop‐ups, and order sets unique to each of a number of disciplines. As the electronic record equalizes but also separates members of the healthcare team, interdisciplinary personal communication becomes more, not less, important.
Recent and impending reimbursement reform proves also to be a means of democratizing medical care and enforcing better interdisciplinary communication. The basis for hospital reimbursement has evolved over decades from day rates to payments for specific diseases, a system under which profit margins are in theory determined by the interdisciplinary efficiency with which diseases are managed by all care givers and the accuracy with which that management is documented. The next, seemingly inexorable, step in the evolution of reimbursement will result in further democratization of care givers: a single combined disease episode payment will be divided among all those involved in a course of treatment that may span many months and require many disciplines and many types of intervention. Payment reform makes the success of a visiting nurse as important to the net reimbursement of a disease episode as the success of an orthopedic surgeon; for if the visiting nurse does not do well the patient will be readmitted or require more office services. In this sense, payment reform, like the electronic record, tends both to equalize the importance of different healthcare roles and to require their enhanced communication.
As these changes in technology and reimbursement evolve, the study of medical documentation must increasingly address medical communication more generally. It is entirely possible, for example, that an individual daily progress note, whose preparation consumes so many hours and removes caretakers from patients, will no longer serve any demonstrable purpose.[3, 4] It may be that consensus summaries will prove more useful in clarifying one's own thinking and incorporating that of others than will a daily, solo chart soliloquy in free or imported text. It is conceivable that contrasting views will be best presented not as a debate in the progress notes but as a plan mutually agreed upon earlier in the decision‐making process. These are the kind of broader questions that investigators in medical documentation should be pursuing.
Another problem in studies of documentation is a pervasive professional bias in the choice of end points. Studies tend to evaluate documentary practices not by their effect on patients but by their impact on physicians or nurses. Success is measured by clinician satisfaction, percent adoption, and note length or timing; note quality is judged using a checklist derived from professional surveys.[5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15] End points like these will often make 1 document look better than another in a results section, but it is the relation between communication strategies and healthcare outcomes that determines whether 1 approach or another is of benefit to the patient.
For example, an important current debate is whether free text adds essential nuance to a note or is simply nostalgia, a relic of the 3‐ring binder.[16, 17, 18] This debate can be resolved convincingly only if improvement with the use or abolition of free text is measured in terms of patient outcomes or resource consumption. Again, if it is important to know whether progress notes of a particular length or structure create less handover confusion, then changes in medical error rates is a more persuasive way to evaluate this issue than a change in physician opinion. It may be a good question whether briefer notes will free nurses and doctors to spend more time at the bedside, but along with recording bedside time that study should also measure improvement in reacting to important changes of clinical status. With today's technology, group phone discussions could perhaps successfully replace examining each other's notes, but the measure of success should be improved hospital efficiency or a decline in errors and readmissions.
The questions we ask in our research today create the treatments and policies of tomorrow. Our studies must address communications in a larger sense, must encompass all the settings in which an episode of care occurs, and must focus on patient outcomes and use of resources. The measured end points of an intervention should of course be sensitive to the particular setting where the intervention takes place, or else small and location‐specific gains will be missed. However, real health effects and robust measures of efficiency must take the place of word counts, inclusion checklists, and clinician adoption or satisfaction in the design of documentation studies.
A great national experiment is underway involving the deployment of information technology, the expansion and empowerment of healthcare teams, and the retargeting of economic incentives. The experimental hypothesis is that technology will increase medical efficiency and will benefit patient well‐being only if these are in fact the purposes, and if teamwork is the principal means, of providing medical care. We should seize this time of change as an opportunity to measure and demonstrably improve the contribution of medical documentation and communication to the efficient and long‐term remission of disease.
- . Medical records that guide and teach. N Engl J Med. 1968;278(12):593–600.
- . Medical records that guide and teach. N Engl J Med. 1968;278(12):652–257.
- , , , . Use of electronic clinical documentation: time spent and team interactions. J Am Med Inform Assoc. 2011;18(2):112–117.
- , , , , , . The influence of integrated electronic medical records and computerized nursing notes on nurses' time spent in documentation. Comput Inform Nurs. 2012;30(6):287–292.
- , , , , . The hybrid progress note: semiautomating daily progress notes to achieve high‐quality documentation and improve provider efficiency. Am J Med Qual. 2013;28(1):25–32.
- , , , . Preliminary development of the physician documentation quality instrument. J Am Med Inform Assoc. 2008;15(4):534–541.
- , , , , . Evaluation of residents' delivery notes after a simulated shoulder dystocia. Obstet Gynecol. 2004;104(4):667–670.
- , , , , , . Validity evidence for a patient note scoring rubric based on the new patient note format of the United States Medical Licensing Examination. Acad Med. 2013;88(10):1552–1557.
- , , , . Quality of outpatient clinical notes: a stakeholder definition derived through qualitative research. BMC Health Serv Res. 2012;12:407.
- , , . Definition, structure, content, use and impacts of electronic health records: a review of the research literature. Int J Med Inform. 2008;77(5):291–304.
- , , , et al. Randomised trial comparing the recording ability of a novel, electronic emergency documentation system with the AHA paper cardiac arrest record [published online ahead of print July 29, 2013]. Emerg Med J. doi: 10.1136/emermed‐2013‐202512.
- , , , et al. Generating clinical notes for electronic health record systems. Appl Clin Inform. 2010;1(3):232–243.
- , , . The effects of EMR deployment on doctors' work practices: a qualitative study in the emergency department of a teaching hospital. Int J Med Inform. 2012;81(3):204–217.
- , , , , . Comparison of handheld computer‐assisted and conventional paper chart documentation of medical records. A randomized, controlled trial. J Bone Joint Surg Am. 2004;86A(3):553–560.
- , , , , . Assessing quality and efficiency of discharge summaries. Am J Med Qual. 2005;20(6):337–343.
- , , , , , . Physicians' attitudes towards copy and pasting in electronic note writing. J Gen Intern Med. 2009;24(1):63–68.
- , , , . Association of medical directors of information systems consensus on inpatient electronic health record documentation. Appl Clin Inform. 2013;4(2):293–303.
- , , . Method of electronic health record documentation and quality of primary care. Am Med Inform Assoc. 2012;19(6):1019–1024.
In 1968, Weed highlighted the importance of medical documentation when he proposed a single format for notes.[1, 2] Since then, sweeping changes in the technology, the purposes, and the requirements of clinical record keeping have encouraged steady growth of a literature devoted to the chart. Specifically, over the past half century, computers, lawsuits, regulations, and the use of documentation as a tool of billing have all contributed to the transformation of hospital records. In addition, mounting pressure to shorten inpatient stays, the vastly increased complexity of care, and a growing number of diagnostic possibilities have combined to make medical documentation far more prolific and far less leisurely. All these changes have stimulated a boom in documentation research coinciding, productively, with an era of rapid advances in the conduct of clinical trials and statistical rigor. However, in important respects research into medical documentation today is not asking the right questions, either in the formulation of hypotheses or in the choice of methodology. Forms of clinical communication that do not involve order sets or notes are widespread, growing in sophistication, and increasingly relevant to new concepts of healthcare as a team enterprise; but documentation research has not embraced this development. At the same time, methodologically, the field suffers from a persistent professional bias in the choice of research outcomes, a bias that limits the interpretation of results by neglecting what happens to the patient.
In assessing the chart as a communication device and the effect of changes in documentation, it is increasingly necessary to study direct interpersonal communication as an alternative and partner to writing notes. In particular, 3 recent developments in healthcare emphasize the importance of broadening our concepts of clinical communication. First, the need for discussion in the medical record has become less pressing because of technical improvements in person‐to‐person communication. Second, the electronic health record, by creating discipline‐defined chart views, has helped equalize the stature of different healthcare disciplines but also Balkanized the chart, making direct interdisciplinary communication more necessary. Third, changes in reimbursement are redefining medical goals in such a way that only teams of healthcare providers in close and constant personal communication can achieve them.
Rapid adoption of electronic health records has encouraged researchers studying documentation or information technology to focus on computer formats as defining the range of all possible communication strategies. And certainly there is a broad range of formats: electronic progress notes may be free text or multiple choice, typed or dictated, copy forwarded or composed daily, institutionally templated or self‐templated, furnished with or free from prompts and pop‐ups. However, it is not only, and perhaps not even principally, the electronic record that has changed how clinicians communicate with each other. The technology of discussion over the last 2 decades has become instant, utterly mobile, device independent, and capable of connecting all the patient's caregivers at once to each other and to the medical record in text, picture, and sound. That the same communications upheaval has visited practically every other aspect of our lives diminishes perhaps the visibility of this new virtual team in healthcare but not its importance.
The electronic record certainly plays a role in facilitating communication, through simultaneous chart access and in many other ways, but even more significant is the effect that computerization has had on equalizing the roles of different disciplines and by doing so in fragmenting the medical record. A computerized record expands and reorganizes the chart, changing it from a single authoritative book read by all to an almost limitless array of chart views, each read by some. All viewers (patient, clinician or researcher, administrator, reviewer or coder) can, with equal claim to consulting the chart, categorize, compare, combine, and format data elements from 1 or many encounters, whether inpatient or ambulatory. Typically, an electronic item of patient information may have several authors and uses but has no owner. Data are entered by protocol and in different guises into many aspects of patient care as components of notes, flow sheets, summaries, pop‐ups, and order sets unique to each of a number of disciplines. As the electronic record equalizes but also separates members of the healthcare team, interdisciplinary personal communication becomes more, not less, important.
Recent and impending reimbursement reform proves also to be a means of democratizing medical care and enforcing better interdisciplinary communication. The basis for hospital reimbursement has evolved over decades from day rates to payments for specific diseases, a system under which profit margins are in theory determined by the interdisciplinary efficiency with which diseases are managed by all care givers and the accuracy with which that management is documented. The next, seemingly inexorable, step in the evolution of reimbursement will result in further democratization of care givers: a single combined disease episode payment will be divided among all those involved in a course of treatment that may span many months and require many disciplines and many types of intervention. Payment reform makes the success of a visiting nurse as important to the net reimbursement of a disease episode as the success of an orthopedic surgeon; for if the visiting nurse does not do well the patient will be readmitted or require more office services. In this sense, payment reform, like the electronic record, tends both to equalize the importance of different healthcare roles and to require their enhanced communication.
As these changes in technology and reimbursement evolve, the study of medical documentation must increasingly address medical communication more generally. It is entirely possible, for example, that an individual daily progress note, whose preparation consumes so many hours and removes caretakers from patients, will no longer serve any demonstrable purpose.[3, 4] It may be that consensus summaries will prove more useful in clarifying one's own thinking and incorporating that of others than will a daily, solo chart soliloquy in free or imported text. It is conceivable that contrasting views will be best presented not as a debate in the progress notes but as a plan mutually agreed upon earlier in the decision‐making process. These are the kind of broader questions that investigators in medical documentation should be pursuing.
Another problem in studies of documentation is a pervasive professional bias in the choice of end points. Studies tend to evaluate documentary practices not by their effect on patients but by their impact on physicians or nurses. Success is measured by clinician satisfaction, percent adoption, and note length or timing; note quality is judged using a checklist derived from professional surveys.[5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15] End points like these will often make 1 document look better than another in a results section, but it is the relation between communication strategies and healthcare outcomes that determines whether 1 approach or another is of benefit to the patient.
For example, an important current debate is whether free text adds essential nuance to a note or is simply nostalgia, a relic of the 3‐ring binder.[16, 17, 18] This debate can be resolved convincingly only if improvement with the use or abolition of free text is measured in terms of patient outcomes or resource consumption. Again, if it is important to know whether progress notes of a particular length or structure create less handover confusion, then changes in medical error rates is a more persuasive way to evaluate this issue than a change in physician opinion. It may be a good question whether briefer notes will free nurses and doctors to spend more time at the bedside, but along with recording bedside time that study should also measure improvement in reacting to important changes of clinical status. With today's technology, group phone discussions could perhaps successfully replace examining each other's notes, but the measure of success should be improved hospital efficiency or a decline in errors and readmissions.
The questions we ask in our research today create the treatments and policies of tomorrow. Our studies must address communications in a larger sense, must encompass all the settings in which an episode of care occurs, and must focus on patient outcomes and use of resources. The measured end points of an intervention should of course be sensitive to the particular setting where the intervention takes place, or else small and location‐specific gains will be missed. However, real health effects and robust measures of efficiency must take the place of word counts, inclusion checklists, and clinician adoption or satisfaction in the design of documentation studies.
A great national experiment is underway involving the deployment of information technology, the expansion and empowerment of healthcare teams, and the retargeting of economic incentives. The experimental hypothesis is that technology will increase medical efficiency and will benefit patient well‐being only if these are in fact the purposes, and if teamwork is the principal means, of providing medical care. We should seize this time of change as an opportunity to measure and demonstrably improve the contribution of medical documentation and communication to the efficient and long‐term remission of disease.
In 1968, Weed highlighted the importance of medical documentation when he proposed a single format for notes.[1, 2] Since then, sweeping changes in the technology, the purposes, and the requirements of clinical record keeping have encouraged steady growth of a literature devoted to the chart. Specifically, over the past half century, computers, lawsuits, regulations, and the use of documentation as a tool of billing have all contributed to the transformation of hospital records. In addition, mounting pressure to shorten inpatient stays, the vastly increased complexity of care, and a growing number of diagnostic possibilities have combined to make medical documentation far more prolific and far less leisurely. All these changes have stimulated a boom in documentation research coinciding, productively, with an era of rapid advances in the conduct of clinical trials and statistical rigor. However, in important respects research into medical documentation today is not asking the right questions, either in the formulation of hypotheses or in the choice of methodology. Forms of clinical communication that do not involve order sets or notes are widespread, growing in sophistication, and increasingly relevant to new concepts of healthcare as a team enterprise; but documentation research has not embraced this development. At the same time, methodologically, the field suffers from a persistent professional bias in the choice of research outcomes, a bias that limits the interpretation of results by neglecting what happens to the patient.
In assessing the chart as a communication device and the effect of changes in documentation, it is increasingly necessary to study direct interpersonal communication as an alternative and partner to writing notes. In particular, 3 recent developments in healthcare emphasize the importance of broadening our concepts of clinical communication. First, the need for discussion in the medical record has become less pressing because of technical improvements in person‐to‐person communication. Second, the electronic health record, by creating discipline‐defined chart views, has helped equalize the stature of different healthcare disciplines but also Balkanized the chart, making direct interdisciplinary communication more necessary. Third, changes in reimbursement are redefining medical goals in such a way that only teams of healthcare providers in close and constant personal communication can achieve them.
Rapid adoption of electronic health records has encouraged researchers studying documentation or information technology to focus on computer formats as defining the range of all possible communication strategies. And certainly there is a broad range of formats: electronic progress notes may be free text or multiple choice, typed or dictated, copy forwarded or composed daily, institutionally templated or self‐templated, furnished with or free from prompts and pop‐ups. However, it is not only, and perhaps not even principally, the electronic record that has changed how clinicians communicate with each other. The technology of discussion over the last 2 decades has become instant, utterly mobile, device independent, and capable of connecting all the patient's caregivers at once to each other and to the medical record in text, picture, and sound. That the same communications upheaval has visited practically every other aspect of our lives diminishes perhaps the visibility of this new virtual team in healthcare but not its importance.
The electronic record certainly plays a role in facilitating communication, through simultaneous chart access and in many other ways, but even more significant is the effect that computerization has had on equalizing the roles of different disciplines and by doing so in fragmenting the medical record. A computerized record expands and reorganizes the chart, changing it from a single authoritative book read by all to an almost limitless array of chart views, each read by some. All viewers (patient, clinician or researcher, administrator, reviewer or coder) can, with equal claim to consulting the chart, categorize, compare, combine, and format data elements from 1 or many encounters, whether inpatient or ambulatory. Typically, an electronic item of patient information may have several authors and uses but has no owner. Data are entered by protocol and in different guises into many aspects of patient care as components of notes, flow sheets, summaries, pop‐ups, and order sets unique to each of a number of disciplines. As the electronic record equalizes but also separates members of the healthcare team, interdisciplinary personal communication becomes more, not less, important.
Recent and impending reimbursement reform proves also to be a means of democratizing medical care and enforcing better interdisciplinary communication. The basis for hospital reimbursement has evolved over decades from day rates to payments for specific diseases, a system under which profit margins are in theory determined by the interdisciplinary efficiency with which diseases are managed by all care givers and the accuracy with which that management is documented. The next, seemingly inexorable, step in the evolution of reimbursement will result in further democratization of care givers: a single combined disease episode payment will be divided among all those involved in a course of treatment that may span many months and require many disciplines and many types of intervention. Payment reform makes the success of a visiting nurse as important to the net reimbursement of a disease episode as the success of an orthopedic surgeon; for if the visiting nurse does not do well the patient will be readmitted or require more office services. In this sense, payment reform, like the electronic record, tends both to equalize the importance of different healthcare roles and to require their enhanced communication.
As these changes in technology and reimbursement evolve, the study of medical documentation must increasingly address medical communication more generally. It is entirely possible, for example, that an individual daily progress note, whose preparation consumes so many hours and removes caretakers from patients, will no longer serve any demonstrable purpose.[3, 4] It may be that consensus summaries will prove more useful in clarifying one's own thinking and incorporating that of others than will a daily, solo chart soliloquy in free or imported text. It is conceivable that contrasting views will be best presented not as a debate in the progress notes but as a plan mutually agreed upon earlier in the decision‐making process. These are the kind of broader questions that investigators in medical documentation should be pursuing.
Another problem in studies of documentation is a pervasive professional bias in the choice of end points. Studies tend to evaluate documentary practices not by their effect on patients but by their impact on physicians or nurses. Success is measured by clinician satisfaction, percent adoption, and note length or timing; note quality is judged using a checklist derived from professional surveys.[5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15] End points like these will often make 1 document look better than another in a results section, but it is the relation between communication strategies and healthcare outcomes that determines whether 1 approach or another is of benefit to the patient.
For example, an important current debate is whether free text adds essential nuance to a note or is simply nostalgia, a relic of the 3‐ring binder.[16, 17, 18] This debate can be resolved convincingly only if improvement with the use or abolition of free text is measured in terms of patient outcomes or resource consumption. Again, if it is important to know whether progress notes of a particular length or structure create less handover confusion, then changes in medical error rates is a more persuasive way to evaluate this issue than a change in physician opinion. It may be a good question whether briefer notes will free nurses and doctors to spend more time at the bedside, but along with recording bedside time that study should also measure improvement in reacting to important changes of clinical status. With today's technology, group phone discussions could perhaps successfully replace examining each other's notes, but the measure of success should be improved hospital efficiency or a decline in errors and readmissions.
The questions we ask in our research today create the treatments and policies of tomorrow. Our studies must address communications in a larger sense, must encompass all the settings in which an episode of care occurs, and must focus on patient outcomes and use of resources. The measured end points of an intervention should of course be sensitive to the particular setting where the intervention takes place, or else small and location‐specific gains will be missed. However, real health effects and robust measures of efficiency must take the place of word counts, inclusion checklists, and clinician adoption or satisfaction in the design of documentation studies.
A great national experiment is underway involving the deployment of information technology, the expansion and empowerment of healthcare teams, and the retargeting of economic incentives. The experimental hypothesis is that technology will increase medical efficiency and will benefit patient well‐being only if these are in fact the purposes, and if teamwork is the principal means, of providing medical care. We should seize this time of change as an opportunity to measure and demonstrably improve the contribution of medical documentation and communication to the efficient and long‐term remission of disease.
- . Medical records that guide and teach. N Engl J Med. 1968;278(12):593–600.
- . Medical records that guide and teach. N Engl J Med. 1968;278(12):652–257.
- , , , . Use of electronic clinical documentation: time spent and team interactions. J Am Med Inform Assoc. 2011;18(2):112–117.
- , , , , , . The influence of integrated electronic medical records and computerized nursing notes on nurses' time spent in documentation. Comput Inform Nurs. 2012;30(6):287–292.
- , , , , . The hybrid progress note: semiautomating daily progress notes to achieve high‐quality documentation and improve provider efficiency. Am J Med Qual. 2013;28(1):25–32.
- , , , . Preliminary development of the physician documentation quality instrument. J Am Med Inform Assoc. 2008;15(4):534–541.
- , , , , . Evaluation of residents' delivery notes after a simulated shoulder dystocia. Obstet Gynecol. 2004;104(4):667–670.
- , , , , , . Validity evidence for a patient note scoring rubric based on the new patient note format of the United States Medical Licensing Examination. Acad Med. 2013;88(10):1552–1557.
- , , , . Quality of outpatient clinical notes: a stakeholder definition derived through qualitative research. BMC Health Serv Res. 2012;12:407.
- , , . Definition, structure, content, use and impacts of electronic health records: a review of the research literature. Int J Med Inform. 2008;77(5):291–304.
- , , , et al. Randomised trial comparing the recording ability of a novel, electronic emergency documentation system with the AHA paper cardiac arrest record [published online ahead of print July 29, 2013]. Emerg Med J. doi: 10.1136/emermed‐2013‐202512.
- , , , et al. Generating clinical notes for electronic health record systems. Appl Clin Inform. 2010;1(3):232–243.
- , , . The effects of EMR deployment on doctors' work practices: a qualitative study in the emergency department of a teaching hospital. Int J Med Inform. 2012;81(3):204–217.
- , , , , . Comparison of handheld computer‐assisted and conventional paper chart documentation of medical records. A randomized, controlled trial. J Bone Joint Surg Am. 2004;86A(3):553–560.
- , , , , . Assessing quality and efficiency of discharge summaries. Am J Med Qual. 2005;20(6):337–343.
- , , , , , . Physicians' attitudes towards copy and pasting in electronic note writing. J Gen Intern Med. 2009;24(1):63–68.
- , , , . Association of medical directors of information systems consensus on inpatient electronic health record documentation. Appl Clin Inform. 2013;4(2):293–303.
- , , . Method of electronic health record documentation and quality of primary care. Am Med Inform Assoc. 2012;19(6):1019–1024.
- . Medical records that guide and teach. N Engl J Med. 1968;278(12):593–600.
- . Medical records that guide and teach. N Engl J Med. 1968;278(12):652–257.
- , , , . Use of electronic clinical documentation: time spent and team interactions. J Am Med Inform Assoc. 2011;18(2):112–117.
- , , , , , . The influence of integrated electronic medical records and computerized nursing notes on nurses' time spent in documentation. Comput Inform Nurs. 2012;30(6):287–292.
- , , , , . The hybrid progress note: semiautomating daily progress notes to achieve high‐quality documentation and improve provider efficiency. Am J Med Qual. 2013;28(1):25–32.
- , , , . Preliminary development of the physician documentation quality instrument. J Am Med Inform Assoc. 2008;15(4):534–541.
- , , , , . Evaluation of residents' delivery notes after a simulated shoulder dystocia. Obstet Gynecol. 2004;104(4):667–670.
- , , , , , . Validity evidence for a patient note scoring rubric based on the new patient note format of the United States Medical Licensing Examination. Acad Med. 2013;88(10):1552–1557.
- , , , . Quality of outpatient clinical notes: a stakeholder definition derived through qualitative research. BMC Health Serv Res. 2012;12:407.
- , , . Definition, structure, content, use and impacts of electronic health records: a review of the research literature. Int J Med Inform. 2008;77(5):291–304.
- , , , et al. Randomised trial comparing the recording ability of a novel, electronic emergency documentation system with the AHA paper cardiac arrest record [published online ahead of print July 29, 2013]. Emerg Med J. doi: 10.1136/emermed‐2013‐202512.
- , , , et al. Generating clinical notes for electronic health record systems. Appl Clin Inform. 2010;1(3):232–243.
- , , . The effects of EMR deployment on doctors' work practices: a qualitative study in the emergency department of a teaching hospital. Int J Med Inform. 2012;81(3):204–217.
- , , , , . Comparison of handheld computer‐assisted and conventional paper chart documentation of medical records. A randomized, controlled trial. J Bone Joint Surg Am. 2004;86A(3):553–560.
- , , , , . Assessing quality and efficiency of discharge summaries. Am J Med Qual. 2005;20(6):337–343.
- , , , , , . Physicians' attitudes towards copy and pasting in electronic note writing. J Gen Intern Med. 2009;24(1):63–68.
- , , , . Association of medical directors of information systems consensus on inpatient electronic health record documentation. Appl Clin Inform. 2013;4(2):293–303.
- , , . Method of electronic health record documentation and quality of primary care. Am Med Inform Assoc. 2012;19(6):1019–1024.
Sleep disturbances in cancer patients: Underrecognized and undertreated
Many cancer patients don't sleep well, for a variety of reasons. It is an important problem: not only does poor sleep worsen quality of life, it may affect prognosis. Moreover, treatment is available.
Yet many physicians caring for cancer patients do not ask about sleep problems, underestimating their impact or focusing on more urgent problems. Also, patients may not want to bring up the topic because they consider poor sleep to be unavoidable and untreatable and because they fear that reporting it may shift the focus of their treatment from trying to cure the cancer to easing its symptoms.
This practical review will help health care professionals avoid the common barriers to diagnosis and treatment of poor sleep in cancer patients. Because there are few data on other sleep disorders such as sleep apnea and restless leg syndrome, we will focus on the most common one in cancer patients—insomnia—and its effects on other symptoms and quality of life.
MORE PATIENTS SURVIVE CANCER NOW
Today, more patients are surviving cancer, but cancer symptoms and the side effects of surgery, chemotherapy, and radiation therapy may persist for years.1,2 The most common complaints include cancer-related fatigue, leg restlessness, anxiety, insomnia, and excessive sleepiness.3
Sleep disturbances appear to contribute to the other problems and are relatively easier to quantify. Most studies of sleep disorders in cancer patients have looked specifically at insomnia,4 although a few have explored the prevalence of other sleep disorders, such as sleep-disordered breathing and limb movements during sleep.5
The International Classification of Sleep Disorders, 2nd edition,6 defines insomnia as difficulty going to sleep or staying asleep (the latter defined as waking up in the middle of the night, with wakeful episodes lasting more than 30 minutes), early-morning awakenings (waking 30 minutes or more before the intended time), or nonrestorative sleep, causing significant distress or impairment of day-time functioning.
INSOMNIA WORSENS QUALITY OF LIFE
Insomnia significantly worsens quality of life in cancer patients, and if it can be detected and effectively treated, quality of life is likely to improve. Studies in cancer patients have found that those with insomnia:
- Were less able to cope with stress and carry on their activities of daily living3
- Were much less able to function and reported more pain, less energy, and greater difficulty in dealing with emotional problems7
- Had poor quality of life, both physically and emotionally.3,8
PERHAPS MORE THAN HALF OF CANCER PATIENTS HAVE INSOMNIA
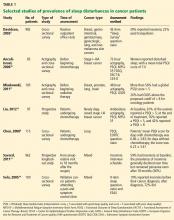
Depending on the methods used and populations studied, at least 30% and perhaps more than half of patients with cancer have insomnia (Table 1).3,4,8–14 It is one of the most commonly reported complaints in this group,15–17 and it occurs before, during, and after treatment of cancer.
Although the prevalence may differ in various cancers, it is still higher than in the general population. In a study of about 450 patients with cancer or depression and 300 healthy volunteers, 62% of the cancer patients reported moderate to severe sleep disturbance, compared with 52% of the depressed patients and 30% of the healthy volunteers.18
When Davidson et al3 surveyed nearly 1,000 cancer patients, one-third said they had insomnia. The problem was most prevalent in lung and breast cancer patients.
In a longitudinal study by Savard et al,13 the prevalence of insomnia declined over time but remained high even at the end of 18 months. It was more prevalent in patients with gynecologic and breast cancer than in those with prostate cancer.13,19
SLEEP PROBLEMS ARE UNDERREPORTED
Sleep problems in cancer patients often go unrecognized because patients do not report them. In a survey of 150 patients,20 44% reported having had sleep problems during the preceding month. However, only one-third of those with sleep problems told their health care providers. This highlights the need for physicians to address sleep complaints in cancer patients at every visit and, if needed, to refer them to a sleep specialist for further evaluation and management.
INSOMNIA IS OFTEN ASSOCIATED WITH OTHER PROBLEMS
Many things can interfere with sleep in cancer patients: the cancer itself (eg, pain due to tumor invasion), medical treatments (eg, narcotics, chemotherapy, neuroleptics, sympathomimetics, steroids, sedative hypnotics), psychosocial disturbances (eg, depression, anxiety, stress), and comorbid medical issues.
In this population, insomnia is often part of a cluster of symptoms that includes pain, fatigue, depression, and anxiety. These act synergistically, worsening quality of life.21–24
Cancer-related fatigue and insomnia
Cancer-related fatigue is a distressing, persistent, subjective sense of tiredness or exhaustion that is related to cancer or cancer treatment, that is not proportional to recent activity and that interferes with usual functioning.25 It has been reported by up to 90% of cancer patients in some studies.26–28
Cancer-related fatigue worsens quality of life and is one of the most distressing and persistent symptoms experienced before, during, and after cancer treatment.29,30 Furthermore, it can lead to sleep disturbances and daytime somnolence and further aggravate insomnia.31,32 The two conditions are often reported as part of a cluster of interrelated symptoms that include pain, depression, and loss of concentration and other cognitive functions, suggesting that they may share a common etiology.33–35
Åhsberg et al36 examined different aspects of perceived cancer-related fatigue in patients undergoing radiotherapy and found correlations between lack of energy, sleepiness, and cancer-related fatigue.
Current understanding of the possible link between cancer-related fatigue and insomnia suggests that interventions targeting the insomnia and daytime sleepiness could decrease the fatigue as well.31
Pain and insomnia in cancer patients
Pain is reported by 60% to 90% of patients with advanced cancer,37,38 its intensity usually varying with the extent of disease. Too often, it is inadequately controlled.39 Furthermore, it is thought to contribute to insomnia.40
In a study of more than 1,600 cancer patients, nearly 60% reported insomnia in addition to pain.41 The severity of pain directly correlated with the probability of insomnia.
Conversely, research suggests that sleep disturbances, primarily insomnia, can increase cancer patients’ sensitivity to pain.42 One hypothesis is that adequate sleep is needed to promote processes relevant to recovery from pain, both physiologic (ie, tissue repair) and psychological (ie, transient cessation of the perception of pain signals).43
Paradoxically, opioids can worsen insomnia
Cancer pain is often treated with opioids, which, paradoxically, can cause or worsen insomnia.
Although opioids induce sleep, they also depress respiration, and at night, they can cause or worsen sleep-disordered breathing (obstructive or central sleep apnea or ataxic breathing), leading to episodes of hypoxia, arousals, and fragmented sleep.44 Moreover, opioids can lead to daytime sedation. Further, psychostimulants such as methylphenidate, given to counteract opioid-induced sedation, can cause anxiety and insomnia. Thus, the interaction between cancer-related pain, insomnia, and pain management leads to a vicious cycle. Understanding this process, we can try to break the cycle and help patients with cancer sleep better.
However, how best to treat sleep-disordered breathing in patients taking opioids long-term is not well established.
In general, the primary intervention is to reduce the opioid dose. Practitioners should continually assess the need for these drugs and consider referral to a drug-behavior treatment center to help with discontinuation of opioid use when deemed medically appropriate.45 Other strategies include positive airway pressure ventilation including continuous positive airway pressure, bilevel pressure devices with backup rate, or adaptive servoventilators. In some cases oxygen supplementation may be required.
Sleep-disordered breathing, when recognized and diagnosed, should be managed in partnership with a sleep specialist.
Depression and insomnia in cancer patients
By some estimates, up to half of cancer patients suffer from depression at some point in their illness.28 And not without reason: these patients face uncertainty about their life, and this often results in depression or anxiety.46
Many cancer patients with depression also have insomnia.28 Indeed, patients with persistent insomnia are at greater risk of developing psychological disorders such as depression and anxiety.47
In a survey of cancer patients, insomnia symptoms were more often attributed to thoughts or concerns about health, family, friends, the cancer diagnosis, and finances than to the actual physical effects of cancer.48
CANCER TREATMENT AND INSOMNIA
Many cancer patients experience sleep disturbances even before starting treatment.49 Liu et al50 showed that, in 76 women about to undergo chemotherapy for breast cancer, those who already had sleep disturbances, fatigue, and depression had more problems, and more severe problems, during chemotherapy.
Radiation therapy and chemotherapy have been reported to cause or precipitate insomnia (Table 2).8,13
Hormonal therapy and biological therapy can also cause or worsen preexisting insomnia.51,52 For example, androgen deprivation therapy for prostate cancer and hormonal therapy for breast cancer are often associated with sleep problems.49,50 Possible mechanisms of insomnia include hot flashes, night sweats, and anxiety caused by such treatments. Biological agents such as interferons, interleukins, and tumor necrosis factor (TNF) alpha, which are often used to treat malignant melanoma, can affect the sleep-wake cycle, leading to insomnia.53
Corticosteroids sharply raise serum cortisol levels, which can lead to insomnia. Cancer patients receiving dexamethasone to prevent radiation-induced emesis experienced more insomnia than patients who did not receive dexamethasone.54
IMMUNOLOGIC BASIS OF INSOMNIA IN CANCER PATIENTS
Cancer cells produce inflammatory cytokines such as interleukin 1 (IL-1), interleukin 6 (IL-6), and TNF alpha, and inflammation plays a role in tumor progression and possibly tumorigenesis.55
Specific cytokines also help regulate the sleep-wake cycle. Levels of IL-6 and TNF alpha peak during sleep, and daytime IL-6 levels are inversely related to the amount of nocturnal sleep.56 Vgontzas et al57 showed that although mean levels of 24-hour IL-6 and TNF alpha secretion were not significantly different in patients with insomnia vs healthy controls, chronic insomnia was associated with a shift in IL-6 and TNF alpha secretion from nighttime to daytime.57
Cancer and its treatment can affect secretion of the cytokines that play a role in the sleep-wake cycle. Thus, the sleep disturbances associated with cancer may also be related to the abnormalities in cytokine levels caused by either cancer or its treatment.
Mills et al58 found that inflammatory markers such as vascular endothelial growth factor and soluble intercellular adhesion molecule-1 were significantly elevated during chemotherapy in breast cancer patients, and the elevated vascular endothelial growth factor levels were associated with poorer sleep during treatment.
Further research is warranted to establish causality, to help us understand the mechanisms of insomnia and other cancer symptoms, and to develop new treatments for these complaints.
POOR SLEEP AND CANCER RISK AND OUTCOMES
Sleep disturbances have negative health consequences in cancer. Their impact ranges from plausible carcinogenesis to affecting the course of the disease and cancer survival.
Poor sleep and risk of cancer
Epidemiologic studies have examined a possible link between circadian rhythm disruption and breast cancer risk, using both direct measures such as melatonin levels and indirect measures such as sleep duration and shift work. (Melatonin production is related to sleep duration, and night-shift work leads to disruption of sleep pattern and quality of sleep, thus lowering melatonin levels.59)
The findings were mixed. Breast cancer risk was significantly and inversely associated with urinary melatonin levels (6-sulfatoxymelatonin) in the Nurses’ Health Study II,60 but not in the Guernsey III study in the United Kingdom.61 Breast cancer risk was significantly lower with longer sleep duration in Finnish women62 and in Chinese women in Singapore,63 but not in American women.64,65 Results of three cohort studies66–68 and two case-control studies69,70 suggested a higher breast cancer risk in women who work evening or overnight shifts. Shorter sleep duration was associated with a higher risk of colorectal adenomas.71
These studies make a strong case for an association of cancer with circadian rhythm disruption and shorter sleep duration, possibly from an effect on melatonin levels. However, one should be cautious in interpreting epidemiologic studies: although they show sleep disturbances to be associated with cancer risk, they do not establish causality.
Insomnia and cancer outcomes
Evidence is growing that sleep disturbances may affect compliance with treatment, immune function, and outcomes—including survival—in cancer patients.23,24
In patients newly diagnosed with various types of cancer, Degner and Sloan72 showed that those who suffered from insomnia, nausea, poor appetite, and pain had a lower survival rate at 5 years, independent of the cancer stage. However, no separate analyses were performed to examine the specific influence of insomnia on cancer survival.
Thompson and Li73 analyzed data from 101 breast cancer patients with available Oncotype DX recurrence scores (a proprietary genetic test performed on tumor tissue that predicts the likelihood of recurrence). The scores were strongly correlated with average hours of sleep per night before breast cancer diagnosis, with fewer hours of sleep associated with a higher (worse) score.
Since these studies were retrospective and merely suggest associations, prospective studies, using more standardized questionnaires and objective measures, are needed to establish causality and to further our understanding of the mechanisms involved.
HELPING CANCER PATIENTS SLEEP BETTER
Insomnia is generally diagnosed with a thorough history that includes sleep, medical issues, substance use, and psychiatric issues. The sleep history should include specific insomniarelated complaints, presleep conditions and habits, sleep-wake habits, other sleep-related symptoms, and daytime consequences. To obtain the information, one can use questionnaires, sleep logs, psychological screening tests, and bed-partner interviews.74
Managing insomnia involves both pharmacologic and nonpharmacologic treatment. It is also important to treat the associated disorders such as depression and anxiety disorders that often accompany insomnia. Long-term management of cancer patients should not be limited to surveillance of cancer but should also involve aggressive treatment of clusters of symptoms such as insomnia, cancer-related fatigue, and pain to yield better long-term quality of life.75–77
Nonpharmacologic treatment: Cognitive-behavioral therapy
Nonpharmacologic interventions use psychological and behavioral therapies. The American Academy of Sleep Medicine guidelines recommend cognitive behavioral therapy for all patients with insomnia, either alone or in combination with hypnotic medications.
Cognitive-behavioral therapy for insomnia includes various components that help the patient learn coping skills and ways to prevent or mitigate the severity of future episodes (Table 3). Various randomized controlled trials found it to be effective for treating insomnia in the general population.77–79
Several studies found that cognitive-behavioral therapy for insomnia was effective in cancer patients, not only improving sleep quality but also decreasing psychological distress, resulting in better overall quality of life.80,81
Savard et al81 conducted a randomized controlled trial of cognitive-behavioral therapy for insomnia in 57 patients with breast cancer, examining subjective and objective sleep measures, psychological functioning, quality of life, and immunologic responses. They found significant improvements in sleep efficiency, mood, quality of life, depression, anxiety, and need for sleep medications. Improvements in subjective sleep measures persisted on 12-month follow-up.
Berger et al,82 in another randomized controlled trial, assessed behavioral therapy using stimulus control, modified sleep restriction, relaxation therapy, and sleep hygiene in breast cancer patients receiving adjuvant chemotherapy. Behavioral therapy improved sleep quality over time, as measured by the Pittsburgh Sleep Quality Index.
Espie et al83 evaluated the effect of cognitive-behavioral therapy on prostate, colorectal, gynecologic, and breast cancer patients, with similar results.83
Cognitive-behavioral therapy is at least as effective as drug therapy for insomnia in the general population. In the limited studies done in cancer patients, it has been shown to be effective irrespective of the type of cancer and is associated with better long-term outcomes. It diminishes the distress associated with early insomnia, can reduce anxiety, and can promote sleep.
A National Institutes of Health conference on insomnia concluded that cognitivebehavioral therapy is at least as effective as medications for brief treatment of chronic insomnia and that its beneficial effects, in contrast to those produced by medications, may last beyond the termination of treatment.84
It is important to think about numerous factors when considering options such as cognitive-behavioral therapy, as patients with cancer have different complications that may affect sleep quality, such as cancer-related fatigue, cancer-related depression, psychological reactions to the disease, side effects of treatment, and cancer-related pain. These need to be addressed as well.
If cognitive-behavioral therapy is not available, self-help interventions (eg, written material, videos, television and Internet resources) can be used. These have several advantages over professionally administered interventions, including greater accessibility, less burden for the patient, and lower cost. Research is under way evaluating this approach in cancer patients.85
Drug therapy
The focus of therapy should be to treat underlying disorders that may be causing or contributing to insomnia. However, a substantial number of patients may need to be assessed for pharmacotherapy for insomnia.
Sleep problems in the general population are commonly treated with drugs, and most of the recommendations in cancer patients are based on experience in the general population. However, sleep medications should be used cautiously in cancer patients, since to our knowledge there have been no studies of these agents in patients with cancer.
Side effects also need to be considered. For example, sleep medications can profoundly worsen cancer-related fatigue.
Hypnotics are often prescribed for cancer patients.86,87 A study in five major oncology centers showed that about half of the 1,500 patients were prescribed at least one psychotropic drug.86 In this study, hypnotics were the most frequently prescribed drugs, accounting for 48% of total prescriptions, and 44% of the psychotropic prescriptions were written for sleep.
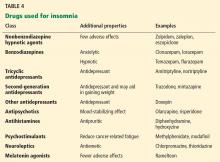
Benzodiazepine receptor agonists such as zaleplon, zolpidem, and eszopiclone can be used for problems with falling asleep and staying asleep.88,89 They are better tolerated than older, long-acting benzodiazepines,90 which can cause alterations in sleep-cycle architecture or rebound insomnia. The earlier agents can also cause adverse effects such as tolerance, drowsiness, and cognitive impairment.
A National Institutes of Health conference stated that benzodiazepine receptor agonists are efficacious in the short-term management of insomnia and that their adverse effects are much less frequent and severe than those of the benzodiazepines or other sedating drugs.84 It also stated that all antidepressants, antihistamines (H1 receptor antagonists), and anti-psychotics have potentially significant adverse effects that raise concerns about their risk-to-benefit ratio and their suitability as treatment for chronic insomnia.
Benzodiazepines are commonly prescribed for insomnia. They increase sleep efficiency, decrease arousals, and increase stage 2 sleep.
Melatonin receptor agonists have been approved by the US Food and Drug Administration for treating insomnia. A recent meta-analysis of eight studies in healthy patients showed improvements in subjective and objective sleep outcomes with the use of ramelteon.91 The dosages primarily used were 4 to 32 mg. However, most of the studies used a dosage of 4 to 8 mg.
Antidepressants. Some of the antidepressants are also used for insomnia, but they can cause daytime fatigue.
Mirtazapine was shown to be effective for insomnia and coexistent mood disorder in cancer patients, but larger trials are needed.92
A recent clinical trial with secondary data analyses evaluated the effect of paroxetine on insomnia, depression, and fatigue in patients with cancer. Paroxetine significantly reduced insomnia in both depressed and nondepressed patients after 2 to 3 weeks of treatment.93
Table 4 summarizes classes of drugs used for insomnia and their additional therapeutic properties.
- Ness KK, Wall MM, Oakes JM, Robison LL, Gurney JG. Physical performance limitations and participation restrictions among cancer survivors: a population-based study. Ann Epidemiol 2006; 16:197–205.
- Deimling GT, Bowman KF, Sterns S, Wagner LJ, Kahana B. Cancer-related health worries and psychological distress among older adult, long-term cancer survivors. Psychooncology 2006; 15:306–320.
- Davidson JR, MacLean AW, Brundage MD, Schulze K. Sleep disturbance in cancer patients. Soc Sci Med 2002; 54:1309–1321.
- Savard J, Morin CM. Insomnia in the context of cancer: a review of a neglected problem. J Clin Oncol 2001; 19:895–908.
- Payne RJ, Hier MP, Kost KM, et al. High prevalence of obstructive sleep apnea among patients with head and neck cancer. J Otolaryngol 2005; 34:304–311.
- American Academy of Sleep Medicine. International Classification of Sleep Disorders—Second Edition (ICSD-2); 2005.
- Fortner BV, Stepanski EJ, Wang SC, Kasprowicz S, Durrence HH. Sleep and quality of life in breast cancer patients. J Pain Symptom Manage 2002; 24:471–480.
- Chen ML, Yu CT, Yang CH. Sleep disturbances and quality of life in lung cancer patients undergoing chemotherapy. Lung Cancer 2008; 62:391–400.
- Liu L, Ancoli-Israel S. Sleep disturbances in cancer. Psychiatr Ann 2008; 38:627–634.
- Ancoli-Israel S, Liu L, Marler MR, et al. Fatigue, sleep, and circadian rhythms prior to chemotherapy for breast cancer. Support Care Cancer 2006; 14:201–209.
- Miaskowski C, Lee K, Dunn L, et al. Sleep-wake circadian activity rhythm parameters and fatigue in oncology patients before the initiation of radiation therapy. Cancer Nurs 2011; 34:255–268.
- Liu L, Rissling M, Natarajan L, et al. The longitudinal relationship between fatigue and sleep in breast cancer patients undergoing chemotherapy. Sleep 2012; 35:237–245.
- Savard J, Ivers H, Villa J, Caplette-Gingras A, Morin CM. Natural course of insomnia comorbid with cancer: an 18-month longitudinal study. J Clin Oncol 2011; 29:3580–3586.
- Sela RA, Watanabe S, Nekolaichuk CL. Sleep disturbances in palliative cancer patients attending a pain and symptom control clinic. Palliat Support Care 2005; 3:23–31.
- Mao JJ, Armstrong K, Bowman MA, Xie SX, Kadakia R, Farrar JT. Symptom burden among cancer survivors: impact of age and comorbidity. J Am Board Fam Med 2007; 20:434–443.
- Schroevers MJ, Ranchor AV, Sanderman R. The role of age at the onset of cancer in relation to survivors’ long-term adjustment: a controlled comparison over an eight-year period. Psychooncology 2004; 13:740–752.
- Stein KD, Syrjala KL, Andrykowski MA. Physical and psychological long-term and late effects of cancer. Cancer 2008; 112(suppl 11):2577–2592.
- Anderson KO, Getto CJ, Mendoza TR, et al. Fatigue and sleep disturbance in patients with cancer, patients with clinical depression, and community-dwelling adults. J Pain Symptom Manage 2003; 25:307–318.
- Savard J, Villa J, Ivers H, Simard S, Morin CM. Prevalence, natural course, and risk factors of insomnia comorbid with cancer over a 2-month period. J Clin Oncol 2009; 27:5233–5239.
- Engstrom CA, Strohl RA, Rose L, Lewandowski L, Stefanek ME. Sleep alterations in cancer patients. Cancer Nurs 1999; 22:143–148.
- Hoffman A, Given BA, von Eye A, Given CW, Gift AG. A study on the relationship between fatigue, pain, insomnia, and gender in persons with lung cancer. Oncol Nurs Forum 2006; 33:404.
- Hoffman AJ, Given BA, von Eye A, Gift AG, Given CW. Relationships among pain, fatigue, insomnia, and gender in persons with lung cancer. Oncol Nurs Forum 2007; 34:785–792.
- Shapiro SL, Bootzin RR, Figueredo AJ, Lopez AM, Schwartz GE. The efficacy of mindfulness-based stress reduction in the treatment of disturbance in women with breast cancer: an exploratory study. J Psychosom Res 2003; 54:85–91.
- Shapiro SL, Lopez AM, Schwartz GE, et al. Quality of life and breast cancer: relationship to psychosocial variables. J Clin Psychol 2001; 57:501–519.
- Mock V, Atkinson A, Barsevick A, et al; National Comprehensive Cancer Network. NCCN practice guidelines for cancer-related fatigue. Oncology (Williston Park) 2000; 14:151–161.
- Cella D, Davis K, Breitbart W, Curt G;Fatigue Coalition. Cancer-related fatigue: prevalence of proposed diagnostic criteria in a United States sample of cancer survivors. J Clin Oncol 2001; 19:3385–3391.
- Sateia MJ, Lang BJ. Sleep and cancer: recent developments. Curr Oncol Rep 2008; 10:309–318.
- Ahluwalia M. Fatigue, pain, and depression among older adults with cancer: still underrecognized and undertreated. Geriatrics and Aging 2008; 11:495–501.
- Enderlin CA, Coleman EA, Cole C, Richards KC, Hutchins LF, Sherman AC. Sleep across chemotherapy treatment: a growing concern for women older than 50 with breast cancer. Oncol Nurs Forum 2010; 37:461–A3.
- Winningham ML, Nail LM, Burke MB, et al. Fatigue and the cancer experience: the state of the knowledge. Oncol Nurs Forum 1994; 21:23–36.
- Berger AM, Mitchell SA. Modifying cancer-related fatigue by optimizing sleep quality. J Natl Compr Canc Netw 2008; 6:3–13.
- Anderson KO, Getto CJ, Mendoza TR, et al. Fatigue and sleep disturbance in patients with cancer, patients with clinical depression, and community-dwelling adults. J Pain Symptom Manage 2003; 25:307–318.
- Armstrong TS, Cohen MZ, Eriksen LR, Hickey JV. Symptom clusters in oncology patients and implications for symptom research in people with primary brain tumors. J Nurs Scholarsh 2004; 36:197–206.
- Dodd MJ, Miaskowski C, Lee KA. Occurrence of symptom clusters. J Natl Cancer Inst Monogr 2004;76–78.
- Paice JA. Assessment of symptom clusters in people with cancer. J Natl Cancer Inst Monogr 2004;98–102.
- Åhsberg E, Fürst CJ. Dimensions of fatigue during radiotherapy—an application of the Swedish Occupational Fatigue Inventory (SOFI) on cancer patients. Acta Oncol 2001; 40:37–43.
- Foley KM. The treatment of cancer pain. N Engl J Med 1985; 313:84–95.
- Twycross RG, Fairfield S. Pain in far-advanced cancer. Pain 1982; 14:303–310.
- Cleeland CS, Gonin R, Hatfield AK, et al. Pain and its treatment in outpatients with metastatic cancer. N Engl J Med 1994; 330:592–596.
- Fleming L, Gillespie S, Espie CA. The development and impact of insomnia on cancer survivors: Psychooncology 2010; 19:991–996.
- Grond S, Zech D, Diefenbach C, Bischoff A. Prevalence and pattern of symptoms in patients with cancer pain: a prospective evaluation of 1635 cancer patients referred to a pain clinic. J Pain Symptom Manage 1994; 9:372–382.
- Smith MT, Haythornthwaite JA. How do sleep disturbance and chronic pain inter-relate? Insights from the longitudinal and cognitive-behavioral clinical trials literature. Sleep Med Rev 2004; 8:119–132.
- Lewin DS, Dahl RE. Importance of sleep in the management of pediatric pain. J Dev Behav Pediatr 1999; 20:244–252.
- Yue HJ, Guilleminault C. Opioid medication and sleep-disordered breathing. Med Clin North Am 2010; 94:435–446.
- Teichtahl H, Wang D. Sleep-disordered breathing with chronic opioid use. Expert Opin Drug Saf 2007; 6:641–649.
- Ancoli-Israel S, Moore PJ, Jones V. The relationship between fatigue and sleep in cancer patients: a review. Eur J Cancer Care (Engl) 2001; 10:245–255.
- Perlis ML, Giles DE, Buysse DJ, Tu X, Kupfer DJ. Self-reported sleep disturbance as a prodromal symptom in recurrent depression. J Affect Disord 1997; 42:209–212.
- Stone P, Hardy J, Broadley K, Tookman AJ, Kurowska A, A’Hern R. Fatigue in advanced cancer: a prospective controlled cross-sectional study. Br J Cancer 1999; 79:1479–1486.
- Cimprich B. Pretreatment symptom distress in women newly diagnosed with breast cancer. Cancer Nurs 1999; 22:185–194.
- Liu L, Fiorentino L, Natarajan L, et al. Pre-treatment symptom cluster in breast cancer patients is associated with worse sleep, fatigue and depression during chemotherapy. Psychooncology 2009; 18:187–194.
- Savard J, Hervouet S, Ivers H. Prostate cancer treatments and their side effects are associated with increased insomnia. Psychooncology 2013; 22:1381–1388.
- Fenlon DR, Corner JL, Haviland J. Menopausal hot flushes after breast cancer. Eur J Cancer Care (Engl) 2009; 18:140–148.
- Miller AH, Ancoli-Israel S, Bower JE, Capuron L, Irwin MR. Neuroendocrine-immune mechanisms of behavioral comorbidities in patients with cancer. J Clin Oncol 2008; 26:971–982.
- Kirkbride P, Bezjak A, Pater J, et al. Dexamethasone for the prophylaxis of radiation-induced emesis: a National Cancer Institute of Canada Clinical Trials Group phase III study. J Clin Oncol 2000; 18:1960–1966.
- Coussens LM, Werb Z. Inflammation and cancer. Nature 2002; 420:860–867.
- Vgontzas AN, Chrousos GP. Sleep, the hypothalamic-pituitary-adrenal axis, and cytokines: multiple interactions and disturbances in sleep disorders. Endocrinol Metab Clin North Am 2002; 31:15–36.
- Vgontzas AN, Zoumakis M, Papanicolaou DA, et al. Chronic insomnia is associated with a shift of interleukin-6 and tumor necrosis factor secretion from nighttime to daytime. Metabolism 2002; 51:887–892.
- Mills PJ, Parker B, Jones V, et al. The effects of standard anthracycline-based chemotherapy on soluble ICAM-1 and vascular endothelial growth factor levels in breast cancer. Clin Cancer Res 2004; 10:4998–5003.
- Reiter RJ, Tan DX, Korkmaz A, et al. Light at night, chronodisruption, melatonin suppression, and cancer risk: a review. Crit Rev Oncog 2007; 13:303–328.
- Schernhammer ES, Hankinson SE. Urinary melatonin levels and breast cancer risk. J Natl Cancer Inst 2005; 97:1084–1087.
- Travis RC, Allen DS, Fentiman IS, Key TJ. Melatonin and breast cancer: a prospective study. J Natl Cancer Inst 2004; 96:475–482.
- Verkasalo PK, Lillberg K, Stevens RG, et al. Sleep duration and breast cancer: a prospective cohort study. Cancer Res 2005; 65:9595–9600.
- Wu AH, Wang R, Koh WP, Stanczyk FZ, Lee HP, Yu MC. Sleep duration, melatonin and breast cancer among Chinese women in Singapore. Carcinogenesis 2008; 29:1244–1248.
- McElroy JA, Newcomb PA, Titus-Ernstoff L, Trentham-Dietz A, Hampton JM, Egan KM. Duration of sleep and breast cancer risk in a large population-based case-control study. J Sleep Res 2006; 15:241–249.
- Pinheiro SP, Schernhammer ES, Tworoger SS, Michels KB. A prospective study on habitual duration of sleep and incidence of breast cancer in a large cohort of women. Cancer Res 2006; 66:5521–5525.
- Lie JA, Roessink J, Kjaerheim K. Breast cancer and night work among Norwegian nurses. Cancer Causes Control 2006; 17:39–44.
- Schernhammer ES, Kroenke CH, Laden F, Hankinson SE. Night work and risk of breast cancer. Epidemiology 2006; 17:108–111.
- Schernhammer ES, Laden F, Speizer FE, et al. Rotating night shifts and risk of breast cancer in women participating in the Nurses’ Health Study. J Natl Cancer Inst 2001; 93:1563–1568.
- Davis S, Mirick DK, Stevens RG. Night shift work, light at night, and risk of breast cancer. J Natl Cancer Inst 2001; 93:1557–1562.
- Hansen J. Light at night, shiftwork, and breast cancer risk. J Natl Cancer Inst 2001; 93:1513–1515.
- Thompson CL, Larkin EK, Patel S, Berger NA, Redline S, Li L. Short duration of sleep increases risk of colorectal adenoma. Cancer 2011; 117:841–847.
- Degner LF, Sloan JA. Symptom distress in newly diagnosed ambulatory cancer patients and as a predictor of survival in lung cancer. J Pain Symptom Manage 1995; 10:423–431.
- Thompson CL, Li L. Association of sleep duration and breast cancer OncotypeDX recurrence score. Breast Cancer Res Treat 2012; 134:1291–1295.
- Schutte-Rodin S, Broch L, Buysse D, Dorsey C, Sateia M. Clinical guideline for the evaluation and management of chronic insomnia in adults. J Clin Sleep Med 2008; 4:487–504.
- Fan HG, Houédé-Tchen N, Yi QL, et al. Fatigue, menopausal symptoms, and cognitive function in women after adjuvant chemotherapy for breast cancer: 1- and 2-year follow-up of a prospective controlled study. J Clin Oncol 2005; 23:8025–8032.
- Ganz PA. Late effects of cancer and its treatment. Semin Oncol Nurs 2001; 17:241–248.
- Lee TS, Kilbreath SL, Refshauge KM, Pendlebury SC, Beith JM, Lee MJ. Quality of life of women treated with radiotherapy for breast cancer. Support Care Cancer 2008; 16:399–405.
- National Institutes of Health. National Institutes of Health state of the science conference statement on manifestations and management of chronic insomnia in adults, June 13–15, 2005. Sleep 2005; 28:1049–1057.
- Smith MT, Huang MI, Manber R. Cognitive behavior therapy for chronic insomnia occurring within the context of medical and psychiatric disorders. Clin Psychol Rev 2005; 25:559–592.
- Quesnel C, Savard J, Simard S, Ivers H, Morin CM. Efficacy of cognitive-behavioral therapy for insomnia in women treated for nonmetastatic breast cancer. J Consult Clin Psychol 2003; 71:189–200.
- Savard J, Simard S, Ivers H, Morin CM. Randomized study on the efficacy of cognitive-behavioral therapy for insomnia secondary to breast cancer, part I: sleep and psychological effects. J Clin Oncol 2005; 23:6083–6096.
- Berger AM, Kuhn BR, Farr LA, et al. Behavioral therapy intervention trial to improve sleep quality and cancer-related fatigue. Psychooncology 2009; 18:634–646.
- Espie CA, Fleming L, Cassidy J, et al. Randomized controlled clinical effectiveness trial of cognitive behavior therapy compared with treatment as usual for persistent insomnia in patients with cancer. J Clin Oncol 2008; 26:4651–4658.
- National Institutes of Health. National Institutes of Health state of the science conference statement on manifestations and management of chronic insomnia in adults, June 13–15, 2005. Sleep 2005; 28:1049–1057.
- Savard J, Villa J, Simard S, Ivers H, Morin CM. Feasibility of a self-help treatment for insomnia comorbid with cancer. Psychooncology 2011; 20:1013–1019.
- Derogatis LR, Feldstein M, Morrow G, et al. A survey of psychotropic drug prescriptions in an oncology population. Cancer 1979; 44:1919–1929.
- Stiefel FC, Kornblith AB, Holland JC. Changes in the prescription patterns of psychotropic drugs for cancer patients during a 10-year period. Cancer 1990; 65:1048–1053.
- Minton O, Richardson A, Sharpe M, Hotopf M, Stone P. A systematic review and meta-analysis of the pharmacological treatment of cancer-related fatigue. J Natl Cancer Inst 2008; 100:1155–1166.
- Minton O, Stone P, Richardson A, Sharpe M, Hotopf M. Drug therapy for the management of cancer-related fatigue. Cochrane Database Syst Rev 2008;CD006704.
- Krystal AD, Walsh JK, Laska E, et al. Sustained efficacy of eszopiclone over 6 months of nightly treatment: results of a randomized, double-blind, placebo-controlled study in adults with chronic insomnia. Sleep 2003; 26:793–799.
- Liu J, Wang LN. Ramelteon in the treatment of chronic insomnia: systematic review and meta-analysis. Int J Clin Pract 2012; 66:867–873.
- Cankurtaran ES, Ozalp E, Soygur H, Akbiyik DI, Turhan L, Alkis N. Mirtazapine improves sleep and lowers anxiety and depression in cancer patients: superiority over imipramine. Support Care Cancer 2008; 16:1291–1298.
- Palesh OG, Mustian KM, Peppone LJ, et al. Impact of paroxetine on sleep problems in 426 cancer patients receiving chemotherapy: a trial from the University of Rochester Cancer Center Community Clinical Oncology Program. Sleep Med 2012; 13:1184–1190.
Many cancer patients don't sleep well, for a variety of reasons. It is an important problem: not only does poor sleep worsen quality of life, it may affect prognosis. Moreover, treatment is available.
Yet many physicians caring for cancer patients do not ask about sleep problems, underestimating their impact or focusing on more urgent problems. Also, patients may not want to bring up the topic because they consider poor sleep to be unavoidable and untreatable and because they fear that reporting it may shift the focus of their treatment from trying to cure the cancer to easing its symptoms.
This practical review will help health care professionals avoid the common barriers to diagnosis and treatment of poor sleep in cancer patients. Because there are few data on other sleep disorders such as sleep apnea and restless leg syndrome, we will focus on the most common one in cancer patients—insomnia—and its effects on other symptoms and quality of life.
MORE PATIENTS SURVIVE CANCER NOW
Today, more patients are surviving cancer, but cancer symptoms and the side effects of surgery, chemotherapy, and radiation therapy may persist for years.1,2 The most common complaints include cancer-related fatigue, leg restlessness, anxiety, insomnia, and excessive sleepiness.3
Sleep disturbances appear to contribute to the other problems and are relatively easier to quantify. Most studies of sleep disorders in cancer patients have looked specifically at insomnia,4 although a few have explored the prevalence of other sleep disorders, such as sleep-disordered breathing and limb movements during sleep.5
The International Classification of Sleep Disorders, 2nd edition,6 defines insomnia as difficulty going to sleep or staying asleep (the latter defined as waking up in the middle of the night, with wakeful episodes lasting more than 30 minutes), early-morning awakenings (waking 30 minutes or more before the intended time), or nonrestorative sleep, causing significant distress or impairment of day-time functioning.
INSOMNIA WORSENS QUALITY OF LIFE
Insomnia significantly worsens quality of life in cancer patients, and if it can be detected and effectively treated, quality of life is likely to improve. Studies in cancer patients have found that those with insomnia:
- Were less able to cope with stress and carry on their activities of daily living3
- Were much less able to function and reported more pain, less energy, and greater difficulty in dealing with emotional problems7
- Had poor quality of life, both physically and emotionally.3,8
PERHAPS MORE THAN HALF OF CANCER PATIENTS HAVE INSOMNIA
Depending on the methods used and populations studied, at least 30% and perhaps more than half of patients with cancer have insomnia (Table 1).3,4,8–14 It is one of the most commonly reported complaints in this group,15–17 and it occurs before, during, and after treatment of cancer.
Although the prevalence may differ in various cancers, it is still higher than in the general population. In a study of about 450 patients with cancer or depression and 300 healthy volunteers, 62% of the cancer patients reported moderate to severe sleep disturbance, compared with 52% of the depressed patients and 30% of the healthy volunteers.18
When Davidson et al3 surveyed nearly 1,000 cancer patients, one-third said they had insomnia. The problem was most prevalent in lung and breast cancer patients.
In a longitudinal study by Savard et al,13 the prevalence of insomnia declined over time but remained high even at the end of 18 months. It was more prevalent in patients with gynecologic and breast cancer than in those with prostate cancer.13,19
SLEEP PROBLEMS ARE UNDERREPORTED
Sleep problems in cancer patients often go unrecognized because patients do not report them. In a survey of 150 patients,20 44% reported having had sleep problems during the preceding month. However, only one-third of those with sleep problems told their health care providers. This highlights the need for physicians to address sleep complaints in cancer patients at every visit and, if needed, to refer them to a sleep specialist for further evaluation and management.
INSOMNIA IS OFTEN ASSOCIATED WITH OTHER PROBLEMS
Many things can interfere with sleep in cancer patients: the cancer itself (eg, pain due to tumor invasion), medical treatments (eg, narcotics, chemotherapy, neuroleptics, sympathomimetics, steroids, sedative hypnotics), psychosocial disturbances (eg, depression, anxiety, stress), and comorbid medical issues.
In this population, insomnia is often part of a cluster of symptoms that includes pain, fatigue, depression, and anxiety. These act synergistically, worsening quality of life.21–24
Cancer-related fatigue and insomnia
Cancer-related fatigue is a distressing, persistent, subjective sense of tiredness or exhaustion that is related to cancer or cancer treatment, that is not proportional to recent activity and that interferes with usual functioning.25 It has been reported by up to 90% of cancer patients in some studies.26–28
Cancer-related fatigue worsens quality of life and is one of the most distressing and persistent symptoms experienced before, during, and after cancer treatment.29,30 Furthermore, it can lead to sleep disturbances and daytime somnolence and further aggravate insomnia.31,32 The two conditions are often reported as part of a cluster of interrelated symptoms that include pain, depression, and loss of concentration and other cognitive functions, suggesting that they may share a common etiology.33–35
Åhsberg et al36 examined different aspects of perceived cancer-related fatigue in patients undergoing radiotherapy and found correlations between lack of energy, sleepiness, and cancer-related fatigue.
Current understanding of the possible link between cancer-related fatigue and insomnia suggests that interventions targeting the insomnia and daytime sleepiness could decrease the fatigue as well.31
Pain and insomnia in cancer patients
Pain is reported by 60% to 90% of patients with advanced cancer,37,38 its intensity usually varying with the extent of disease. Too often, it is inadequately controlled.39 Furthermore, it is thought to contribute to insomnia.40
In a study of more than 1,600 cancer patients, nearly 60% reported insomnia in addition to pain.41 The severity of pain directly correlated with the probability of insomnia.
Conversely, research suggests that sleep disturbances, primarily insomnia, can increase cancer patients’ sensitivity to pain.42 One hypothesis is that adequate sleep is needed to promote processes relevant to recovery from pain, both physiologic (ie, tissue repair) and psychological (ie, transient cessation of the perception of pain signals).43
Paradoxically, opioids can worsen insomnia
Cancer pain is often treated with opioids, which, paradoxically, can cause or worsen insomnia.
Although opioids induce sleep, they also depress respiration, and at night, they can cause or worsen sleep-disordered breathing (obstructive or central sleep apnea or ataxic breathing), leading to episodes of hypoxia, arousals, and fragmented sleep.44 Moreover, opioids can lead to daytime sedation. Further, psychostimulants such as methylphenidate, given to counteract opioid-induced sedation, can cause anxiety and insomnia. Thus, the interaction between cancer-related pain, insomnia, and pain management leads to a vicious cycle. Understanding this process, we can try to break the cycle and help patients with cancer sleep better.
However, how best to treat sleep-disordered breathing in patients taking opioids long-term is not well established.
In general, the primary intervention is to reduce the opioid dose. Practitioners should continually assess the need for these drugs and consider referral to a drug-behavior treatment center to help with discontinuation of opioid use when deemed medically appropriate.45 Other strategies include positive airway pressure ventilation including continuous positive airway pressure, bilevel pressure devices with backup rate, or adaptive servoventilators. In some cases oxygen supplementation may be required.
Sleep-disordered breathing, when recognized and diagnosed, should be managed in partnership with a sleep specialist.
Depression and insomnia in cancer patients
By some estimates, up to half of cancer patients suffer from depression at some point in their illness.28 And not without reason: these patients face uncertainty about their life, and this often results in depression or anxiety.46
Many cancer patients with depression also have insomnia.28 Indeed, patients with persistent insomnia are at greater risk of developing psychological disorders such as depression and anxiety.47
In a survey of cancer patients, insomnia symptoms were more often attributed to thoughts or concerns about health, family, friends, the cancer diagnosis, and finances than to the actual physical effects of cancer.48
CANCER TREATMENT AND INSOMNIA
Many cancer patients experience sleep disturbances even before starting treatment.49 Liu et al50 showed that, in 76 women about to undergo chemotherapy for breast cancer, those who already had sleep disturbances, fatigue, and depression had more problems, and more severe problems, during chemotherapy.
Radiation therapy and chemotherapy have been reported to cause or precipitate insomnia (Table 2).8,13
Hormonal therapy and biological therapy can also cause or worsen preexisting insomnia.51,52 For example, androgen deprivation therapy for prostate cancer and hormonal therapy for breast cancer are often associated with sleep problems.49,50 Possible mechanisms of insomnia include hot flashes, night sweats, and anxiety caused by such treatments. Biological agents such as interferons, interleukins, and tumor necrosis factor (TNF) alpha, which are often used to treat malignant melanoma, can affect the sleep-wake cycle, leading to insomnia.53
Corticosteroids sharply raise serum cortisol levels, which can lead to insomnia. Cancer patients receiving dexamethasone to prevent radiation-induced emesis experienced more insomnia than patients who did not receive dexamethasone.54
IMMUNOLOGIC BASIS OF INSOMNIA IN CANCER PATIENTS
Cancer cells produce inflammatory cytokines such as interleukin 1 (IL-1), interleukin 6 (IL-6), and TNF alpha, and inflammation plays a role in tumor progression and possibly tumorigenesis.55
Specific cytokines also help regulate the sleep-wake cycle. Levels of IL-6 and TNF alpha peak during sleep, and daytime IL-6 levels are inversely related to the amount of nocturnal sleep.56 Vgontzas et al57 showed that although mean levels of 24-hour IL-6 and TNF alpha secretion were not significantly different in patients with insomnia vs healthy controls, chronic insomnia was associated with a shift in IL-6 and TNF alpha secretion from nighttime to daytime.57
Cancer and its treatment can affect secretion of the cytokines that play a role in the sleep-wake cycle. Thus, the sleep disturbances associated with cancer may also be related to the abnormalities in cytokine levels caused by either cancer or its treatment.
Mills et al58 found that inflammatory markers such as vascular endothelial growth factor and soluble intercellular adhesion molecule-1 were significantly elevated during chemotherapy in breast cancer patients, and the elevated vascular endothelial growth factor levels were associated with poorer sleep during treatment.
Further research is warranted to establish causality, to help us understand the mechanisms of insomnia and other cancer symptoms, and to develop new treatments for these complaints.
POOR SLEEP AND CANCER RISK AND OUTCOMES
Sleep disturbances have negative health consequences in cancer. Their impact ranges from plausible carcinogenesis to affecting the course of the disease and cancer survival.
Poor sleep and risk of cancer
Epidemiologic studies have examined a possible link between circadian rhythm disruption and breast cancer risk, using both direct measures such as melatonin levels and indirect measures such as sleep duration and shift work. (Melatonin production is related to sleep duration, and night-shift work leads to disruption of sleep pattern and quality of sleep, thus lowering melatonin levels.59)
The findings were mixed. Breast cancer risk was significantly and inversely associated with urinary melatonin levels (6-sulfatoxymelatonin) in the Nurses’ Health Study II,60 but not in the Guernsey III study in the United Kingdom.61 Breast cancer risk was significantly lower with longer sleep duration in Finnish women62 and in Chinese women in Singapore,63 but not in American women.64,65 Results of three cohort studies66–68 and two case-control studies69,70 suggested a higher breast cancer risk in women who work evening or overnight shifts. Shorter sleep duration was associated with a higher risk of colorectal adenomas.71
These studies make a strong case for an association of cancer with circadian rhythm disruption and shorter sleep duration, possibly from an effect on melatonin levels. However, one should be cautious in interpreting epidemiologic studies: although they show sleep disturbances to be associated with cancer risk, they do not establish causality.
Insomnia and cancer outcomes
Evidence is growing that sleep disturbances may affect compliance with treatment, immune function, and outcomes—including survival—in cancer patients.23,24
In patients newly diagnosed with various types of cancer, Degner and Sloan72 showed that those who suffered from insomnia, nausea, poor appetite, and pain had a lower survival rate at 5 years, independent of the cancer stage. However, no separate analyses were performed to examine the specific influence of insomnia on cancer survival.
Thompson and Li73 analyzed data from 101 breast cancer patients with available Oncotype DX recurrence scores (a proprietary genetic test performed on tumor tissue that predicts the likelihood of recurrence). The scores were strongly correlated with average hours of sleep per night before breast cancer diagnosis, with fewer hours of sleep associated with a higher (worse) score.
Since these studies were retrospective and merely suggest associations, prospective studies, using more standardized questionnaires and objective measures, are needed to establish causality and to further our understanding of the mechanisms involved.
HELPING CANCER PATIENTS SLEEP BETTER
Insomnia is generally diagnosed with a thorough history that includes sleep, medical issues, substance use, and psychiatric issues. The sleep history should include specific insomniarelated complaints, presleep conditions and habits, sleep-wake habits, other sleep-related symptoms, and daytime consequences. To obtain the information, one can use questionnaires, sleep logs, psychological screening tests, and bed-partner interviews.74
Managing insomnia involves both pharmacologic and nonpharmacologic treatment. It is also important to treat the associated disorders such as depression and anxiety disorders that often accompany insomnia. Long-term management of cancer patients should not be limited to surveillance of cancer but should also involve aggressive treatment of clusters of symptoms such as insomnia, cancer-related fatigue, and pain to yield better long-term quality of life.75–77
Nonpharmacologic treatment: Cognitive-behavioral therapy
Nonpharmacologic interventions use psychological and behavioral therapies. The American Academy of Sleep Medicine guidelines recommend cognitive behavioral therapy for all patients with insomnia, either alone or in combination with hypnotic medications.
Cognitive-behavioral therapy for insomnia includes various components that help the patient learn coping skills and ways to prevent or mitigate the severity of future episodes (Table 3). Various randomized controlled trials found it to be effective for treating insomnia in the general population.77–79
Several studies found that cognitive-behavioral therapy for insomnia was effective in cancer patients, not only improving sleep quality but also decreasing psychological distress, resulting in better overall quality of life.80,81
Savard et al81 conducted a randomized controlled trial of cognitive-behavioral therapy for insomnia in 57 patients with breast cancer, examining subjective and objective sleep measures, psychological functioning, quality of life, and immunologic responses. They found significant improvements in sleep efficiency, mood, quality of life, depression, anxiety, and need for sleep medications. Improvements in subjective sleep measures persisted on 12-month follow-up.
Berger et al,82 in another randomized controlled trial, assessed behavioral therapy using stimulus control, modified sleep restriction, relaxation therapy, and sleep hygiene in breast cancer patients receiving adjuvant chemotherapy. Behavioral therapy improved sleep quality over time, as measured by the Pittsburgh Sleep Quality Index.
Espie et al83 evaluated the effect of cognitive-behavioral therapy on prostate, colorectal, gynecologic, and breast cancer patients, with similar results.83
Cognitive-behavioral therapy is at least as effective as drug therapy for insomnia in the general population. In the limited studies done in cancer patients, it has been shown to be effective irrespective of the type of cancer and is associated with better long-term outcomes. It diminishes the distress associated with early insomnia, can reduce anxiety, and can promote sleep.
A National Institutes of Health conference on insomnia concluded that cognitivebehavioral therapy is at least as effective as medications for brief treatment of chronic insomnia and that its beneficial effects, in contrast to those produced by medications, may last beyond the termination of treatment.84
It is important to think about numerous factors when considering options such as cognitive-behavioral therapy, as patients with cancer have different complications that may affect sleep quality, such as cancer-related fatigue, cancer-related depression, psychological reactions to the disease, side effects of treatment, and cancer-related pain. These need to be addressed as well.
If cognitive-behavioral therapy is not available, self-help interventions (eg, written material, videos, television and Internet resources) can be used. These have several advantages over professionally administered interventions, including greater accessibility, less burden for the patient, and lower cost. Research is under way evaluating this approach in cancer patients.85
Drug therapy
The focus of therapy should be to treat underlying disorders that may be causing or contributing to insomnia. However, a substantial number of patients may need to be assessed for pharmacotherapy for insomnia.
Sleep problems in the general population are commonly treated with drugs, and most of the recommendations in cancer patients are based on experience in the general population. However, sleep medications should be used cautiously in cancer patients, since to our knowledge there have been no studies of these agents in patients with cancer.
Side effects also need to be considered. For example, sleep medications can profoundly worsen cancer-related fatigue.
Hypnotics are often prescribed for cancer patients.86,87 A study in five major oncology centers showed that about half of the 1,500 patients were prescribed at least one psychotropic drug.86 In this study, hypnotics were the most frequently prescribed drugs, accounting for 48% of total prescriptions, and 44% of the psychotropic prescriptions were written for sleep.
Benzodiazepine receptor agonists such as zaleplon, zolpidem, and eszopiclone can be used for problems with falling asleep and staying asleep.88,89 They are better tolerated than older, long-acting benzodiazepines,90 which can cause alterations in sleep-cycle architecture or rebound insomnia. The earlier agents can also cause adverse effects such as tolerance, drowsiness, and cognitive impairment.
A National Institutes of Health conference stated that benzodiazepine receptor agonists are efficacious in the short-term management of insomnia and that their adverse effects are much less frequent and severe than those of the benzodiazepines or other sedating drugs.84 It also stated that all antidepressants, antihistamines (H1 receptor antagonists), and anti-psychotics have potentially significant adverse effects that raise concerns about their risk-to-benefit ratio and their suitability as treatment for chronic insomnia.
Benzodiazepines are commonly prescribed for insomnia. They increase sleep efficiency, decrease arousals, and increase stage 2 sleep.
Melatonin receptor agonists have been approved by the US Food and Drug Administration for treating insomnia. A recent meta-analysis of eight studies in healthy patients showed improvements in subjective and objective sleep outcomes with the use of ramelteon.91 The dosages primarily used were 4 to 32 mg. However, most of the studies used a dosage of 4 to 8 mg.
Antidepressants. Some of the antidepressants are also used for insomnia, but they can cause daytime fatigue.
Mirtazapine was shown to be effective for insomnia and coexistent mood disorder in cancer patients, but larger trials are needed.92
A recent clinical trial with secondary data analyses evaluated the effect of paroxetine on insomnia, depression, and fatigue in patients with cancer. Paroxetine significantly reduced insomnia in both depressed and nondepressed patients after 2 to 3 weeks of treatment.93
Table 4 summarizes classes of drugs used for insomnia and their additional therapeutic properties.
Many cancer patients don't sleep well, for a variety of reasons. It is an important problem: not only does poor sleep worsen quality of life, it may affect prognosis. Moreover, treatment is available.
Yet many physicians caring for cancer patients do not ask about sleep problems, underestimating their impact or focusing on more urgent problems. Also, patients may not want to bring up the topic because they consider poor sleep to be unavoidable and untreatable and because they fear that reporting it may shift the focus of their treatment from trying to cure the cancer to easing its symptoms.
This practical review will help health care professionals avoid the common barriers to diagnosis and treatment of poor sleep in cancer patients. Because there are few data on other sleep disorders such as sleep apnea and restless leg syndrome, we will focus on the most common one in cancer patients—insomnia—and its effects on other symptoms and quality of life.
MORE PATIENTS SURVIVE CANCER NOW
Today, more patients are surviving cancer, but cancer symptoms and the side effects of surgery, chemotherapy, and radiation therapy may persist for years.1,2 The most common complaints include cancer-related fatigue, leg restlessness, anxiety, insomnia, and excessive sleepiness.3
Sleep disturbances appear to contribute to the other problems and are relatively easier to quantify. Most studies of sleep disorders in cancer patients have looked specifically at insomnia,4 although a few have explored the prevalence of other sleep disorders, such as sleep-disordered breathing and limb movements during sleep.5
The International Classification of Sleep Disorders, 2nd edition,6 defines insomnia as difficulty going to sleep or staying asleep (the latter defined as waking up in the middle of the night, with wakeful episodes lasting more than 30 minutes), early-morning awakenings (waking 30 minutes or more before the intended time), or nonrestorative sleep, causing significant distress or impairment of day-time functioning.
INSOMNIA WORSENS QUALITY OF LIFE
Insomnia significantly worsens quality of life in cancer patients, and if it can be detected and effectively treated, quality of life is likely to improve. Studies in cancer patients have found that those with insomnia:
- Were less able to cope with stress and carry on their activities of daily living3
- Were much less able to function and reported more pain, less energy, and greater difficulty in dealing with emotional problems7
- Had poor quality of life, both physically and emotionally.3,8
PERHAPS MORE THAN HALF OF CANCER PATIENTS HAVE INSOMNIA
Depending on the methods used and populations studied, at least 30% and perhaps more than half of patients with cancer have insomnia (Table 1).3,4,8–14 It is one of the most commonly reported complaints in this group,15–17 and it occurs before, during, and after treatment of cancer.
Although the prevalence may differ in various cancers, it is still higher than in the general population. In a study of about 450 patients with cancer or depression and 300 healthy volunteers, 62% of the cancer patients reported moderate to severe sleep disturbance, compared with 52% of the depressed patients and 30% of the healthy volunteers.18
When Davidson et al3 surveyed nearly 1,000 cancer patients, one-third said they had insomnia. The problem was most prevalent in lung and breast cancer patients.
In a longitudinal study by Savard et al,13 the prevalence of insomnia declined over time but remained high even at the end of 18 months. It was more prevalent in patients with gynecologic and breast cancer than in those with prostate cancer.13,19
SLEEP PROBLEMS ARE UNDERREPORTED
Sleep problems in cancer patients often go unrecognized because patients do not report them. In a survey of 150 patients,20 44% reported having had sleep problems during the preceding month. However, only one-third of those with sleep problems told their health care providers. This highlights the need for physicians to address sleep complaints in cancer patients at every visit and, if needed, to refer them to a sleep specialist for further evaluation and management.
INSOMNIA IS OFTEN ASSOCIATED WITH OTHER PROBLEMS
Many things can interfere with sleep in cancer patients: the cancer itself (eg, pain due to tumor invasion), medical treatments (eg, narcotics, chemotherapy, neuroleptics, sympathomimetics, steroids, sedative hypnotics), psychosocial disturbances (eg, depression, anxiety, stress), and comorbid medical issues.
In this population, insomnia is often part of a cluster of symptoms that includes pain, fatigue, depression, and anxiety. These act synergistically, worsening quality of life.21–24
Cancer-related fatigue and insomnia
Cancer-related fatigue is a distressing, persistent, subjective sense of tiredness or exhaustion that is related to cancer or cancer treatment, that is not proportional to recent activity and that interferes with usual functioning.25 It has been reported by up to 90% of cancer patients in some studies.26–28
Cancer-related fatigue worsens quality of life and is one of the most distressing and persistent symptoms experienced before, during, and after cancer treatment.29,30 Furthermore, it can lead to sleep disturbances and daytime somnolence and further aggravate insomnia.31,32 The two conditions are often reported as part of a cluster of interrelated symptoms that include pain, depression, and loss of concentration and other cognitive functions, suggesting that they may share a common etiology.33–35
Åhsberg et al36 examined different aspects of perceived cancer-related fatigue in patients undergoing radiotherapy and found correlations between lack of energy, sleepiness, and cancer-related fatigue.
Current understanding of the possible link between cancer-related fatigue and insomnia suggests that interventions targeting the insomnia and daytime sleepiness could decrease the fatigue as well.31
Pain and insomnia in cancer patients
Pain is reported by 60% to 90% of patients with advanced cancer,37,38 its intensity usually varying with the extent of disease. Too often, it is inadequately controlled.39 Furthermore, it is thought to contribute to insomnia.40
In a study of more than 1,600 cancer patients, nearly 60% reported insomnia in addition to pain.41 The severity of pain directly correlated with the probability of insomnia.
Conversely, research suggests that sleep disturbances, primarily insomnia, can increase cancer patients’ sensitivity to pain.42 One hypothesis is that adequate sleep is needed to promote processes relevant to recovery from pain, both physiologic (ie, tissue repair) and psychological (ie, transient cessation of the perception of pain signals).43
Paradoxically, opioids can worsen insomnia
Cancer pain is often treated with opioids, which, paradoxically, can cause or worsen insomnia.
Although opioids induce sleep, they also depress respiration, and at night, they can cause or worsen sleep-disordered breathing (obstructive or central sleep apnea or ataxic breathing), leading to episodes of hypoxia, arousals, and fragmented sleep.44 Moreover, opioids can lead to daytime sedation. Further, psychostimulants such as methylphenidate, given to counteract opioid-induced sedation, can cause anxiety and insomnia. Thus, the interaction between cancer-related pain, insomnia, and pain management leads to a vicious cycle. Understanding this process, we can try to break the cycle and help patients with cancer sleep better.
However, how best to treat sleep-disordered breathing in patients taking opioids long-term is not well established.
In general, the primary intervention is to reduce the opioid dose. Practitioners should continually assess the need for these drugs and consider referral to a drug-behavior treatment center to help with discontinuation of opioid use when deemed medically appropriate.45 Other strategies include positive airway pressure ventilation including continuous positive airway pressure, bilevel pressure devices with backup rate, or adaptive servoventilators. In some cases oxygen supplementation may be required.
Sleep-disordered breathing, when recognized and diagnosed, should be managed in partnership with a sleep specialist.
Depression and insomnia in cancer patients
By some estimates, up to half of cancer patients suffer from depression at some point in their illness.28 And not without reason: these patients face uncertainty about their life, and this often results in depression or anxiety.46
Many cancer patients with depression also have insomnia.28 Indeed, patients with persistent insomnia are at greater risk of developing psychological disorders such as depression and anxiety.47
In a survey of cancer patients, insomnia symptoms were more often attributed to thoughts or concerns about health, family, friends, the cancer diagnosis, and finances than to the actual physical effects of cancer.48
CANCER TREATMENT AND INSOMNIA
Many cancer patients experience sleep disturbances even before starting treatment.49 Liu et al50 showed that, in 76 women about to undergo chemotherapy for breast cancer, those who already had sleep disturbances, fatigue, and depression had more problems, and more severe problems, during chemotherapy.
Radiation therapy and chemotherapy have been reported to cause or precipitate insomnia (Table 2).8,13
Hormonal therapy and biological therapy can also cause or worsen preexisting insomnia.51,52 For example, androgen deprivation therapy for prostate cancer and hormonal therapy for breast cancer are often associated with sleep problems.49,50 Possible mechanisms of insomnia include hot flashes, night sweats, and anxiety caused by such treatments. Biological agents such as interferons, interleukins, and tumor necrosis factor (TNF) alpha, which are often used to treat malignant melanoma, can affect the sleep-wake cycle, leading to insomnia.53
Corticosteroids sharply raise serum cortisol levels, which can lead to insomnia. Cancer patients receiving dexamethasone to prevent radiation-induced emesis experienced more insomnia than patients who did not receive dexamethasone.54
IMMUNOLOGIC BASIS OF INSOMNIA IN CANCER PATIENTS
Cancer cells produce inflammatory cytokines such as interleukin 1 (IL-1), interleukin 6 (IL-6), and TNF alpha, and inflammation plays a role in tumor progression and possibly tumorigenesis.55
Specific cytokines also help regulate the sleep-wake cycle. Levels of IL-6 and TNF alpha peak during sleep, and daytime IL-6 levels are inversely related to the amount of nocturnal sleep.56 Vgontzas et al57 showed that although mean levels of 24-hour IL-6 and TNF alpha secretion were not significantly different in patients with insomnia vs healthy controls, chronic insomnia was associated with a shift in IL-6 and TNF alpha secretion from nighttime to daytime.57
Cancer and its treatment can affect secretion of the cytokines that play a role in the sleep-wake cycle. Thus, the sleep disturbances associated with cancer may also be related to the abnormalities in cytokine levels caused by either cancer or its treatment.
Mills et al58 found that inflammatory markers such as vascular endothelial growth factor and soluble intercellular adhesion molecule-1 were significantly elevated during chemotherapy in breast cancer patients, and the elevated vascular endothelial growth factor levels were associated with poorer sleep during treatment.
Further research is warranted to establish causality, to help us understand the mechanisms of insomnia and other cancer symptoms, and to develop new treatments for these complaints.
POOR SLEEP AND CANCER RISK AND OUTCOMES
Sleep disturbances have negative health consequences in cancer. Their impact ranges from plausible carcinogenesis to affecting the course of the disease and cancer survival.
Poor sleep and risk of cancer
Epidemiologic studies have examined a possible link between circadian rhythm disruption and breast cancer risk, using both direct measures such as melatonin levels and indirect measures such as sleep duration and shift work. (Melatonin production is related to sleep duration, and night-shift work leads to disruption of sleep pattern and quality of sleep, thus lowering melatonin levels.59)
The findings were mixed. Breast cancer risk was significantly and inversely associated with urinary melatonin levels (6-sulfatoxymelatonin) in the Nurses’ Health Study II,60 but not in the Guernsey III study in the United Kingdom.61 Breast cancer risk was significantly lower with longer sleep duration in Finnish women62 and in Chinese women in Singapore,63 but not in American women.64,65 Results of three cohort studies66–68 and two case-control studies69,70 suggested a higher breast cancer risk in women who work evening or overnight shifts. Shorter sleep duration was associated with a higher risk of colorectal adenomas.71
These studies make a strong case for an association of cancer with circadian rhythm disruption and shorter sleep duration, possibly from an effect on melatonin levels. However, one should be cautious in interpreting epidemiologic studies: although they show sleep disturbances to be associated with cancer risk, they do not establish causality.
Insomnia and cancer outcomes
Evidence is growing that sleep disturbances may affect compliance with treatment, immune function, and outcomes—including survival—in cancer patients.23,24
In patients newly diagnosed with various types of cancer, Degner and Sloan72 showed that those who suffered from insomnia, nausea, poor appetite, and pain had a lower survival rate at 5 years, independent of the cancer stage. However, no separate analyses were performed to examine the specific influence of insomnia on cancer survival.
Thompson and Li73 analyzed data from 101 breast cancer patients with available Oncotype DX recurrence scores (a proprietary genetic test performed on tumor tissue that predicts the likelihood of recurrence). The scores were strongly correlated with average hours of sleep per night before breast cancer diagnosis, with fewer hours of sleep associated with a higher (worse) score.
Since these studies were retrospective and merely suggest associations, prospective studies, using more standardized questionnaires and objective measures, are needed to establish causality and to further our understanding of the mechanisms involved.
HELPING CANCER PATIENTS SLEEP BETTER
Insomnia is generally diagnosed with a thorough history that includes sleep, medical issues, substance use, and psychiatric issues. The sleep history should include specific insomniarelated complaints, presleep conditions and habits, sleep-wake habits, other sleep-related symptoms, and daytime consequences. To obtain the information, one can use questionnaires, sleep logs, psychological screening tests, and bed-partner interviews.74
Managing insomnia involves both pharmacologic and nonpharmacologic treatment. It is also important to treat the associated disorders such as depression and anxiety disorders that often accompany insomnia. Long-term management of cancer patients should not be limited to surveillance of cancer but should also involve aggressive treatment of clusters of symptoms such as insomnia, cancer-related fatigue, and pain to yield better long-term quality of life.75–77
Nonpharmacologic treatment: Cognitive-behavioral therapy
Nonpharmacologic interventions use psychological and behavioral therapies. The American Academy of Sleep Medicine guidelines recommend cognitive behavioral therapy for all patients with insomnia, either alone or in combination with hypnotic medications.
Cognitive-behavioral therapy for insomnia includes various components that help the patient learn coping skills and ways to prevent or mitigate the severity of future episodes (Table 3). Various randomized controlled trials found it to be effective for treating insomnia in the general population.77–79
Several studies found that cognitive-behavioral therapy for insomnia was effective in cancer patients, not only improving sleep quality but also decreasing psychological distress, resulting in better overall quality of life.80,81
Savard et al81 conducted a randomized controlled trial of cognitive-behavioral therapy for insomnia in 57 patients with breast cancer, examining subjective and objective sleep measures, psychological functioning, quality of life, and immunologic responses. They found significant improvements in sleep efficiency, mood, quality of life, depression, anxiety, and need for sleep medications. Improvements in subjective sleep measures persisted on 12-month follow-up.
Berger et al,82 in another randomized controlled trial, assessed behavioral therapy using stimulus control, modified sleep restriction, relaxation therapy, and sleep hygiene in breast cancer patients receiving adjuvant chemotherapy. Behavioral therapy improved sleep quality over time, as measured by the Pittsburgh Sleep Quality Index.
Espie et al83 evaluated the effect of cognitive-behavioral therapy on prostate, colorectal, gynecologic, and breast cancer patients, with similar results.83
Cognitive-behavioral therapy is at least as effective as drug therapy for insomnia in the general population. In the limited studies done in cancer patients, it has been shown to be effective irrespective of the type of cancer and is associated with better long-term outcomes. It diminishes the distress associated with early insomnia, can reduce anxiety, and can promote sleep.
A National Institutes of Health conference on insomnia concluded that cognitivebehavioral therapy is at least as effective as medications for brief treatment of chronic insomnia and that its beneficial effects, in contrast to those produced by medications, may last beyond the termination of treatment.84
It is important to think about numerous factors when considering options such as cognitive-behavioral therapy, as patients with cancer have different complications that may affect sleep quality, such as cancer-related fatigue, cancer-related depression, psychological reactions to the disease, side effects of treatment, and cancer-related pain. These need to be addressed as well.
If cognitive-behavioral therapy is not available, self-help interventions (eg, written material, videos, television and Internet resources) can be used. These have several advantages over professionally administered interventions, including greater accessibility, less burden for the patient, and lower cost. Research is under way evaluating this approach in cancer patients.85
Drug therapy
The focus of therapy should be to treat underlying disorders that may be causing or contributing to insomnia. However, a substantial number of patients may need to be assessed for pharmacotherapy for insomnia.
Sleep problems in the general population are commonly treated with drugs, and most of the recommendations in cancer patients are based on experience in the general population. However, sleep medications should be used cautiously in cancer patients, since to our knowledge there have been no studies of these agents in patients with cancer.
Side effects also need to be considered. For example, sleep medications can profoundly worsen cancer-related fatigue.
Hypnotics are often prescribed for cancer patients.86,87 A study in five major oncology centers showed that about half of the 1,500 patients were prescribed at least one psychotropic drug.86 In this study, hypnotics were the most frequently prescribed drugs, accounting for 48% of total prescriptions, and 44% of the psychotropic prescriptions were written for sleep.
Benzodiazepine receptor agonists such as zaleplon, zolpidem, and eszopiclone can be used for problems with falling asleep and staying asleep.88,89 They are better tolerated than older, long-acting benzodiazepines,90 which can cause alterations in sleep-cycle architecture or rebound insomnia. The earlier agents can also cause adverse effects such as tolerance, drowsiness, and cognitive impairment.
A National Institutes of Health conference stated that benzodiazepine receptor agonists are efficacious in the short-term management of insomnia and that their adverse effects are much less frequent and severe than those of the benzodiazepines or other sedating drugs.84 It also stated that all antidepressants, antihistamines (H1 receptor antagonists), and anti-psychotics have potentially significant adverse effects that raise concerns about their risk-to-benefit ratio and their suitability as treatment for chronic insomnia.
Benzodiazepines are commonly prescribed for insomnia. They increase sleep efficiency, decrease arousals, and increase stage 2 sleep.
Melatonin receptor agonists have been approved by the US Food and Drug Administration for treating insomnia. A recent meta-analysis of eight studies in healthy patients showed improvements in subjective and objective sleep outcomes with the use of ramelteon.91 The dosages primarily used were 4 to 32 mg. However, most of the studies used a dosage of 4 to 8 mg.
Antidepressants. Some of the antidepressants are also used for insomnia, but they can cause daytime fatigue.
Mirtazapine was shown to be effective for insomnia and coexistent mood disorder in cancer patients, but larger trials are needed.92
A recent clinical trial with secondary data analyses evaluated the effect of paroxetine on insomnia, depression, and fatigue in patients with cancer. Paroxetine significantly reduced insomnia in both depressed and nondepressed patients after 2 to 3 weeks of treatment.93
Table 4 summarizes classes of drugs used for insomnia and their additional therapeutic properties.
- Ness KK, Wall MM, Oakes JM, Robison LL, Gurney JG. Physical performance limitations and participation restrictions among cancer survivors: a population-based study. Ann Epidemiol 2006; 16:197–205.
- Deimling GT, Bowman KF, Sterns S, Wagner LJ, Kahana B. Cancer-related health worries and psychological distress among older adult, long-term cancer survivors. Psychooncology 2006; 15:306–320.
- Davidson JR, MacLean AW, Brundage MD, Schulze K. Sleep disturbance in cancer patients. Soc Sci Med 2002; 54:1309–1321.
- Savard J, Morin CM. Insomnia in the context of cancer: a review of a neglected problem. J Clin Oncol 2001; 19:895–908.
- Payne RJ, Hier MP, Kost KM, et al. High prevalence of obstructive sleep apnea among patients with head and neck cancer. J Otolaryngol 2005; 34:304–311.
- American Academy of Sleep Medicine. International Classification of Sleep Disorders—Second Edition (ICSD-2); 2005.
- Fortner BV, Stepanski EJ, Wang SC, Kasprowicz S, Durrence HH. Sleep and quality of life in breast cancer patients. J Pain Symptom Manage 2002; 24:471–480.
- Chen ML, Yu CT, Yang CH. Sleep disturbances and quality of life in lung cancer patients undergoing chemotherapy. Lung Cancer 2008; 62:391–400.
- Liu L, Ancoli-Israel S. Sleep disturbances in cancer. Psychiatr Ann 2008; 38:627–634.
- Ancoli-Israel S, Liu L, Marler MR, et al. Fatigue, sleep, and circadian rhythms prior to chemotherapy for breast cancer. Support Care Cancer 2006; 14:201–209.
- Miaskowski C, Lee K, Dunn L, et al. Sleep-wake circadian activity rhythm parameters and fatigue in oncology patients before the initiation of radiation therapy. Cancer Nurs 2011; 34:255–268.
- Liu L, Rissling M, Natarajan L, et al. The longitudinal relationship between fatigue and sleep in breast cancer patients undergoing chemotherapy. Sleep 2012; 35:237–245.
- Savard J, Ivers H, Villa J, Caplette-Gingras A, Morin CM. Natural course of insomnia comorbid with cancer: an 18-month longitudinal study. J Clin Oncol 2011; 29:3580–3586.
- Sela RA, Watanabe S, Nekolaichuk CL. Sleep disturbances in palliative cancer patients attending a pain and symptom control clinic. Palliat Support Care 2005; 3:23–31.
- Mao JJ, Armstrong K, Bowman MA, Xie SX, Kadakia R, Farrar JT. Symptom burden among cancer survivors: impact of age and comorbidity. J Am Board Fam Med 2007; 20:434–443.
- Schroevers MJ, Ranchor AV, Sanderman R. The role of age at the onset of cancer in relation to survivors’ long-term adjustment: a controlled comparison over an eight-year period. Psychooncology 2004; 13:740–752.
- Stein KD, Syrjala KL, Andrykowski MA. Physical and psychological long-term and late effects of cancer. Cancer 2008; 112(suppl 11):2577–2592.
- Anderson KO, Getto CJ, Mendoza TR, et al. Fatigue and sleep disturbance in patients with cancer, patients with clinical depression, and community-dwelling adults. J Pain Symptom Manage 2003; 25:307–318.
- Savard J, Villa J, Ivers H, Simard S, Morin CM. Prevalence, natural course, and risk factors of insomnia comorbid with cancer over a 2-month period. J Clin Oncol 2009; 27:5233–5239.
- Engstrom CA, Strohl RA, Rose L, Lewandowski L, Stefanek ME. Sleep alterations in cancer patients. Cancer Nurs 1999; 22:143–148.
- Hoffman A, Given BA, von Eye A, Given CW, Gift AG. A study on the relationship between fatigue, pain, insomnia, and gender in persons with lung cancer. Oncol Nurs Forum 2006; 33:404.
- Hoffman AJ, Given BA, von Eye A, Gift AG, Given CW. Relationships among pain, fatigue, insomnia, and gender in persons with lung cancer. Oncol Nurs Forum 2007; 34:785–792.
- Shapiro SL, Bootzin RR, Figueredo AJ, Lopez AM, Schwartz GE. The efficacy of mindfulness-based stress reduction in the treatment of disturbance in women with breast cancer: an exploratory study. J Psychosom Res 2003; 54:85–91.
- Shapiro SL, Lopez AM, Schwartz GE, et al. Quality of life and breast cancer: relationship to psychosocial variables. J Clin Psychol 2001; 57:501–519.
- Mock V, Atkinson A, Barsevick A, et al; National Comprehensive Cancer Network. NCCN practice guidelines for cancer-related fatigue. Oncology (Williston Park) 2000; 14:151–161.
- Cella D, Davis K, Breitbart W, Curt G;Fatigue Coalition. Cancer-related fatigue: prevalence of proposed diagnostic criteria in a United States sample of cancer survivors. J Clin Oncol 2001; 19:3385–3391.
- Sateia MJ, Lang BJ. Sleep and cancer: recent developments. Curr Oncol Rep 2008; 10:309–318.
- Ahluwalia M. Fatigue, pain, and depression among older adults with cancer: still underrecognized and undertreated. Geriatrics and Aging 2008; 11:495–501.
- Enderlin CA, Coleman EA, Cole C, Richards KC, Hutchins LF, Sherman AC. Sleep across chemotherapy treatment: a growing concern for women older than 50 with breast cancer. Oncol Nurs Forum 2010; 37:461–A3.
- Winningham ML, Nail LM, Burke MB, et al. Fatigue and the cancer experience: the state of the knowledge. Oncol Nurs Forum 1994; 21:23–36.
- Berger AM, Mitchell SA. Modifying cancer-related fatigue by optimizing sleep quality. J Natl Compr Canc Netw 2008; 6:3–13.
- Anderson KO, Getto CJ, Mendoza TR, et al. Fatigue and sleep disturbance in patients with cancer, patients with clinical depression, and community-dwelling adults. J Pain Symptom Manage 2003; 25:307–318.
- Armstrong TS, Cohen MZ, Eriksen LR, Hickey JV. Symptom clusters in oncology patients and implications for symptom research in people with primary brain tumors. J Nurs Scholarsh 2004; 36:197–206.
- Dodd MJ, Miaskowski C, Lee KA. Occurrence of symptom clusters. J Natl Cancer Inst Monogr 2004;76–78.
- Paice JA. Assessment of symptom clusters in people with cancer. J Natl Cancer Inst Monogr 2004;98–102.
- Åhsberg E, Fürst CJ. Dimensions of fatigue during radiotherapy—an application of the Swedish Occupational Fatigue Inventory (SOFI) on cancer patients. Acta Oncol 2001; 40:37–43.
- Foley KM. The treatment of cancer pain. N Engl J Med 1985; 313:84–95.
- Twycross RG, Fairfield S. Pain in far-advanced cancer. Pain 1982; 14:303–310.
- Cleeland CS, Gonin R, Hatfield AK, et al. Pain and its treatment in outpatients with metastatic cancer. N Engl J Med 1994; 330:592–596.
- Fleming L, Gillespie S, Espie CA. The development and impact of insomnia on cancer survivors: Psychooncology 2010; 19:991–996.
- Grond S, Zech D, Diefenbach C, Bischoff A. Prevalence and pattern of symptoms in patients with cancer pain: a prospective evaluation of 1635 cancer patients referred to a pain clinic. J Pain Symptom Manage 1994; 9:372–382.
- Smith MT, Haythornthwaite JA. How do sleep disturbance and chronic pain inter-relate? Insights from the longitudinal and cognitive-behavioral clinical trials literature. Sleep Med Rev 2004; 8:119–132.
- Lewin DS, Dahl RE. Importance of sleep in the management of pediatric pain. J Dev Behav Pediatr 1999; 20:244–252.
- Yue HJ, Guilleminault C. Opioid medication and sleep-disordered breathing. Med Clin North Am 2010; 94:435–446.
- Teichtahl H, Wang D. Sleep-disordered breathing with chronic opioid use. Expert Opin Drug Saf 2007; 6:641–649.
- Ancoli-Israel S, Moore PJ, Jones V. The relationship between fatigue and sleep in cancer patients: a review. Eur J Cancer Care (Engl) 2001; 10:245–255.
- Perlis ML, Giles DE, Buysse DJ, Tu X, Kupfer DJ. Self-reported sleep disturbance as a prodromal symptom in recurrent depression. J Affect Disord 1997; 42:209–212.
- Stone P, Hardy J, Broadley K, Tookman AJ, Kurowska A, A’Hern R. Fatigue in advanced cancer: a prospective controlled cross-sectional study. Br J Cancer 1999; 79:1479–1486.
- Cimprich B. Pretreatment symptom distress in women newly diagnosed with breast cancer. Cancer Nurs 1999; 22:185–194.
- Liu L, Fiorentino L, Natarajan L, et al. Pre-treatment symptom cluster in breast cancer patients is associated with worse sleep, fatigue and depression during chemotherapy. Psychooncology 2009; 18:187–194.
- Savard J, Hervouet S, Ivers H. Prostate cancer treatments and their side effects are associated with increased insomnia. Psychooncology 2013; 22:1381–1388.
- Fenlon DR, Corner JL, Haviland J. Menopausal hot flushes after breast cancer. Eur J Cancer Care (Engl) 2009; 18:140–148.
- Miller AH, Ancoli-Israel S, Bower JE, Capuron L, Irwin MR. Neuroendocrine-immune mechanisms of behavioral comorbidities in patients with cancer. J Clin Oncol 2008; 26:971–982.
- Kirkbride P, Bezjak A, Pater J, et al. Dexamethasone for the prophylaxis of radiation-induced emesis: a National Cancer Institute of Canada Clinical Trials Group phase III study. J Clin Oncol 2000; 18:1960–1966.
- Coussens LM, Werb Z. Inflammation and cancer. Nature 2002; 420:860–867.
- Vgontzas AN, Chrousos GP. Sleep, the hypothalamic-pituitary-adrenal axis, and cytokines: multiple interactions and disturbances in sleep disorders. Endocrinol Metab Clin North Am 2002; 31:15–36.
- Vgontzas AN, Zoumakis M, Papanicolaou DA, et al. Chronic insomnia is associated with a shift of interleukin-6 and tumor necrosis factor secretion from nighttime to daytime. Metabolism 2002; 51:887–892.
- Mills PJ, Parker B, Jones V, et al. The effects of standard anthracycline-based chemotherapy on soluble ICAM-1 and vascular endothelial growth factor levels in breast cancer. Clin Cancer Res 2004; 10:4998–5003.
- Reiter RJ, Tan DX, Korkmaz A, et al. Light at night, chronodisruption, melatonin suppression, and cancer risk: a review. Crit Rev Oncog 2007; 13:303–328.
- Schernhammer ES, Hankinson SE. Urinary melatonin levels and breast cancer risk. J Natl Cancer Inst 2005; 97:1084–1087.
- Travis RC, Allen DS, Fentiman IS, Key TJ. Melatonin and breast cancer: a prospective study. J Natl Cancer Inst 2004; 96:475–482.
- Verkasalo PK, Lillberg K, Stevens RG, et al. Sleep duration and breast cancer: a prospective cohort study. Cancer Res 2005; 65:9595–9600.
- Wu AH, Wang R, Koh WP, Stanczyk FZ, Lee HP, Yu MC. Sleep duration, melatonin and breast cancer among Chinese women in Singapore. Carcinogenesis 2008; 29:1244–1248.
- McElroy JA, Newcomb PA, Titus-Ernstoff L, Trentham-Dietz A, Hampton JM, Egan KM. Duration of sleep and breast cancer risk in a large population-based case-control study. J Sleep Res 2006; 15:241–249.
- Pinheiro SP, Schernhammer ES, Tworoger SS, Michels KB. A prospective study on habitual duration of sleep and incidence of breast cancer in a large cohort of women. Cancer Res 2006; 66:5521–5525.
- Lie JA, Roessink J, Kjaerheim K. Breast cancer and night work among Norwegian nurses. Cancer Causes Control 2006; 17:39–44.
- Schernhammer ES, Kroenke CH, Laden F, Hankinson SE. Night work and risk of breast cancer. Epidemiology 2006; 17:108–111.
- Schernhammer ES, Laden F, Speizer FE, et al. Rotating night shifts and risk of breast cancer in women participating in the Nurses’ Health Study. J Natl Cancer Inst 2001; 93:1563–1568.
- Davis S, Mirick DK, Stevens RG. Night shift work, light at night, and risk of breast cancer. J Natl Cancer Inst 2001; 93:1557–1562.
- Hansen J. Light at night, shiftwork, and breast cancer risk. J Natl Cancer Inst 2001; 93:1513–1515.
- Thompson CL, Larkin EK, Patel S, Berger NA, Redline S, Li L. Short duration of sleep increases risk of colorectal adenoma. Cancer 2011; 117:841–847.
- Degner LF, Sloan JA. Symptom distress in newly diagnosed ambulatory cancer patients and as a predictor of survival in lung cancer. J Pain Symptom Manage 1995; 10:423–431.
- Thompson CL, Li L. Association of sleep duration and breast cancer OncotypeDX recurrence score. Breast Cancer Res Treat 2012; 134:1291–1295.
- Schutte-Rodin S, Broch L, Buysse D, Dorsey C, Sateia M. Clinical guideline for the evaluation and management of chronic insomnia in adults. J Clin Sleep Med 2008; 4:487–504.
- Fan HG, Houédé-Tchen N, Yi QL, et al. Fatigue, menopausal symptoms, and cognitive function in women after adjuvant chemotherapy for breast cancer: 1- and 2-year follow-up of a prospective controlled study. J Clin Oncol 2005; 23:8025–8032.
- Ganz PA. Late effects of cancer and its treatment. Semin Oncol Nurs 2001; 17:241–248.
- Lee TS, Kilbreath SL, Refshauge KM, Pendlebury SC, Beith JM, Lee MJ. Quality of life of women treated with radiotherapy for breast cancer. Support Care Cancer 2008; 16:399–405.
- National Institutes of Health. National Institutes of Health state of the science conference statement on manifestations and management of chronic insomnia in adults, June 13–15, 2005. Sleep 2005; 28:1049–1057.
- Smith MT, Huang MI, Manber R. Cognitive behavior therapy for chronic insomnia occurring within the context of medical and psychiatric disorders. Clin Psychol Rev 2005; 25:559–592.
- Quesnel C, Savard J, Simard S, Ivers H, Morin CM. Efficacy of cognitive-behavioral therapy for insomnia in women treated for nonmetastatic breast cancer. J Consult Clin Psychol 2003; 71:189–200.
- Savard J, Simard S, Ivers H, Morin CM. Randomized study on the efficacy of cognitive-behavioral therapy for insomnia secondary to breast cancer, part I: sleep and psychological effects. J Clin Oncol 2005; 23:6083–6096.
- Berger AM, Kuhn BR, Farr LA, et al. Behavioral therapy intervention trial to improve sleep quality and cancer-related fatigue. Psychooncology 2009; 18:634–646.
- Espie CA, Fleming L, Cassidy J, et al. Randomized controlled clinical effectiveness trial of cognitive behavior therapy compared with treatment as usual for persistent insomnia in patients with cancer. J Clin Oncol 2008; 26:4651–4658.
- National Institutes of Health. National Institutes of Health state of the science conference statement on manifestations and management of chronic insomnia in adults, June 13–15, 2005. Sleep 2005; 28:1049–1057.
- Savard J, Villa J, Simard S, Ivers H, Morin CM. Feasibility of a self-help treatment for insomnia comorbid with cancer. Psychooncology 2011; 20:1013–1019.
- Derogatis LR, Feldstein M, Morrow G, et al. A survey of psychotropic drug prescriptions in an oncology population. Cancer 1979; 44:1919–1929.
- Stiefel FC, Kornblith AB, Holland JC. Changes in the prescription patterns of psychotropic drugs for cancer patients during a 10-year period. Cancer 1990; 65:1048–1053.
- Minton O, Richardson A, Sharpe M, Hotopf M, Stone P. A systematic review and meta-analysis of the pharmacological treatment of cancer-related fatigue. J Natl Cancer Inst 2008; 100:1155–1166.
- Minton O, Stone P, Richardson A, Sharpe M, Hotopf M. Drug therapy for the management of cancer-related fatigue. Cochrane Database Syst Rev 2008;CD006704.
- Krystal AD, Walsh JK, Laska E, et al. Sustained efficacy of eszopiclone over 6 months of nightly treatment: results of a randomized, double-blind, placebo-controlled study in adults with chronic insomnia. Sleep 2003; 26:793–799.
- Liu J, Wang LN. Ramelteon in the treatment of chronic insomnia: systematic review and meta-analysis. Int J Clin Pract 2012; 66:867–873.
- Cankurtaran ES, Ozalp E, Soygur H, Akbiyik DI, Turhan L, Alkis N. Mirtazapine improves sleep and lowers anxiety and depression in cancer patients: superiority over imipramine. Support Care Cancer 2008; 16:1291–1298.
- Palesh OG, Mustian KM, Peppone LJ, et al. Impact of paroxetine on sleep problems in 426 cancer patients receiving chemotherapy: a trial from the University of Rochester Cancer Center Community Clinical Oncology Program. Sleep Med 2012; 13:1184–1190.
- Ness KK, Wall MM, Oakes JM, Robison LL, Gurney JG. Physical performance limitations and participation restrictions among cancer survivors: a population-based study. Ann Epidemiol 2006; 16:197–205.
- Deimling GT, Bowman KF, Sterns S, Wagner LJ, Kahana B. Cancer-related health worries and psychological distress among older adult, long-term cancer survivors. Psychooncology 2006; 15:306–320.
- Davidson JR, MacLean AW, Brundage MD, Schulze K. Sleep disturbance in cancer patients. Soc Sci Med 2002; 54:1309–1321.
- Savard J, Morin CM. Insomnia in the context of cancer: a review of a neglected problem. J Clin Oncol 2001; 19:895–908.
- Payne RJ, Hier MP, Kost KM, et al. High prevalence of obstructive sleep apnea among patients with head and neck cancer. J Otolaryngol 2005; 34:304–311.
- American Academy of Sleep Medicine. International Classification of Sleep Disorders—Second Edition (ICSD-2); 2005.
- Fortner BV, Stepanski EJ, Wang SC, Kasprowicz S, Durrence HH. Sleep and quality of life in breast cancer patients. J Pain Symptom Manage 2002; 24:471–480.
- Chen ML, Yu CT, Yang CH. Sleep disturbances and quality of life in lung cancer patients undergoing chemotherapy. Lung Cancer 2008; 62:391–400.
- Liu L, Ancoli-Israel S. Sleep disturbances in cancer. Psychiatr Ann 2008; 38:627–634.
- Ancoli-Israel S, Liu L, Marler MR, et al. Fatigue, sleep, and circadian rhythms prior to chemotherapy for breast cancer. Support Care Cancer 2006; 14:201–209.
- Miaskowski C, Lee K, Dunn L, et al. Sleep-wake circadian activity rhythm parameters and fatigue in oncology patients before the initiation of radiation therapy. Cancer Nurs 2011; 34:255–268.
- Liu L, Rissling M, Natarajan L, et al. The longitudinal relationship between fatigue and sleep in breast cancer patients undergoing chemotherapy. Sleep 2012; 35:237–245.
- Savard J, Ivers H, Villa J, Caplette-Gingras A, Morin CM. Natural course of insomnia comorbid with cancer: an 18-month longitudinal study. J Clin Oncol 2011; 29:3580–3586.
- Sela RA, Watanabe S, Nekolaichuk CL. Sleep disturbances in palliative cancer patients attending a pain and symptom control clinic. Palliat Support Care 2005; 3:23–31.
- Mao JJ, Armstrong K, Bowman MA, Xie SX, Kadakia R, Farrar JT. Symptom burden among cancer survivors: impact of age and comorbidity. J Am Board Fam Med 2007; 20:434–443.
- Schroevers MJ, Ranchor AV, Sanderman R. The role of age at the onset of cancer in relation to survivors’ long-term adjustment: a controlled comparison over an eight-year period. Psychooncology 2004; 13:740–752.
- Stein KD, Syrjala KL, Andrykowski MA. Physical and psychological long-term and late effects of cancer. Cancer 2008; 112(suppl 11):2577–2592.
- Anderson KO, Getto CJ, Mendoza TR, et al. Fatigue and sleep disturbance in patients with cancer, patients with clinical depression, and community-dwelling adults. J Pain Symptom Manage 2003; 25:307–318.
- Savard J, Villa J, Ivers H, Simard S, Morin CM. Prevalence, natural course, and risk factors of insomnia comorbid with cancer over a 2-month period. J Clin Oncol 2009; 27:5233–5239.
- Engstrom CA, Strohl RA, Rose L, Lewandowski L, Stefanek ME. Sleep alterations in cancer patients. Cancer Nurs 1999; 22:143–148.
- Hoffman A, Given BA, von Eye A, Given CW, Gift AG. A study on the relationship between fatigue, pain, insomnia, and gender in persons with lung cancer. Oncol Nurs Forum 2006; 33:404.
- Hoffman AJ, Given BA, von Eye A, Gift AG, Given CW. Relationships among pain, fatigue, insomnia, and gender in persons with lung cancer. Oncol Nurs Forum 2007; 34:785–792.
- Shapiro SL, Bootzin RR, Figueredo AJ, Lopez AM, Schwartz GE. The efficacy of mindfulness-based stress reduction in the treatment of disturbance in women with breast cancer: an exploratory study. J Psychosom Res 2003; 54:85–91.
- Shapiro SL, Lopez AM, Schwartz GE, et al. Quality of life and breast cancer: relationship to psychosocial variables. J Clin Psychol 2001; 57:501–519.
- Mock V, Atkinson A, Barsevick A, et al; National Comprehensive Cancer Network. NCCN practice guidelines for cancer-related fatigue. Oncology (Williston Park) 2000; 14:151–161.
- Cella D, Davis K, Breitbart W, Curt G;Fatigue Coalition. Cancer-related fatigue: prevalence of proposed diagnostic criteria in a United States sample of cancer survivors. J Clin Oncol 2001; 19:3385–3391.
- Sateia MJ, Lang BJ. Sleep and cancer: recent developments. Curr Oncol Rep 2008; 10:309–318.
- Ahluwalia M. Fatigue, pain, and depression among older adults with cancer: still underrecognized and undertreated. Geriatrics and Aging 2008; 11:495–501.
- Enderlin CA, Coleman EA, Cole C, Richards KC, Hutchins LF, Sherman AC. Sleep across chemotherapy treatment: a growing concern for women older than 50 with breast cancer. Oncol Nurs Forum 2010; 37:461–A3.
- Winningham ML, Nail LM, Burke MB, et al. Fatigue and the cancer experience: the state of the knowledge. Oncol Nurs Forum 1994; 21:23–36.
- Berger AM, Mitchell SA. Modifying cancer-related fatigue by optimizing sleep quality. J Natl Compr Canc Netw 2008; 6:3–13.
- Anderson KO, Getto CJ, Mendoza TR, et al. Fatigue and sleep disturbance in patients with cancer, patients with clinical depression, and community-dwelling adults. J Pain Symptom Manage 2003; 25:307–318.
- Armstrong TS, Cohen MZ, Eriksen LR, Hickey JV. Symptom clusters in oncology patients and implications for symptom research in people with primary brain tumors. J Nurs Scholarsh 2004; 36:197–206.
- Dodd MJ, Miaskowski C, Lee KA. Occurrence of symptom clusters. J Natl Cancer Inst Monogr 2004;76–78.
- Paice JA. Assessment of symptom clusters in people with cancer. J Natl Cancer Inst Monogr 2004;98–102.
- Åhsberg E, Fürst CJ. Dimensions of fatigue during radiotherapy—an application of the Swedish Occupational Fatigue Inventory (SOFI) on cancer patients. Acta Oncol 2001; 40:37–43.
- Foley KM. The treatment of cancer pain. N Engl J Med 1985; 313:84–95.
- Twycross RG, Fairfield S. Pain in far-advanced cancer. Pain 1982; 14:303–310.
- Cleeland CS, Gonin R, Hatfield AK, et al. Pain and its treatment in outpatients with metastatic cancer. N Engl J Med 1994; 330:592–596.
- Fleming L, Gillespie S, Espie CA. The development and impact of insomnia on cancer survivors: Psychooncology 2010; 19:991–996.
- Grond S, Zech D, Diefenbach C, Bischoff A. Prevalence and pattern of symptoms in patients with cancer pain: a prospective evaluation of 1635 cancer patients referred to a pain clinic. J Pain Symptom Manage 1994; 9:372–382.
- Smith MT, Haythornthwaite JA. How do sleep disturbance and chronic pain inter-relate? Insights from the longitudinal and cognitive-behavioral clinical trials literature. Sleep Med Rev 2004; 8:119–132.
- Lewin DS, Dahl RE. Importance of sleep in the management of pediatric pain. J Dev Behav Pediatr 1999; 20:244–252.
- Yue HJ, Guilleminault C. Opioid medication and sleep-disordered breathing. Med Clin North Am 2010; 94:435–446.
- Teichtahl H, Wang D. Sleep-disordered breathing with chronic opioid use. Expert Opin Drug Saf 2007; 6:641–649.
- Ancoli-Israel S, Moore PJ, Jones V. The relationship between fatigue and sleep in cancer patients: a review. Eur J Cancer Care (Engl) 2001; 10:245–255.
- Perlis ML, Giles DE, Buysse DJ, Tu X, Kupfer DJ. Self-reported sleep disturbance as a prodromal symptom in recurrent depression. J Affect Disord 1997; 42:209–212.
- Stone P, Hardy J, Broadley K, Tookman AJ, Kurowska A, A’Hern R. Fatigue in advanced cancer: a prospective controlled cross-sectional study. Br J Cancer 1999; 79:1479–1486.
- Cimprich B. Pretreatment symptom distress in women newly diagnosed with breast cancer. Cancer Nurs 1999; 22:185–194.
- Liu L, Fiorentino L, Natarajan L, et al. Pre-treatment symptom cluster in breast cancer patients is associated with worse sleep, fatigue and depression during chemotherapy. Psychooncology 2009; 18:187–194.
- Savard J, Hervouet S, Ivers H. Prostate cancer treatments and their side effects are associated with increased insomnia. Psychooncology 2013; 22:1381–1388.
- Fenlon DR, Corner JL, Haviland J. Menopausal hot flushes after breast cancer. Eur J Cancer Care (Engl) 2009; 18:140–148.
- Miller AH, Ancoli-Israel S, Bower JE, Capuron L, Irwin MR. Neuroendocrine-immune mechanisms of behavioral comorbidities in patients with cancer. J Clin Oncol 2008; 26:971–982.
- Kirkbride P, Bezjak A, Pater J, et al. Dexamethasone for the prophylaxis of radiation-induced emesis: a National Cancer Institute of Canada Clinical Trials Group phase III study. J Clin Oncol 2000; 18:1960–1966.
- Coussens LM, Werb Z. Inflammation and cancer. Nature 2002; 420:860–867.
- Vgontzas AN, Chrousos GP. Sleep, the hypothalamic-pituitary-adrenal axis, and cytokines: multiple interactions and disturbances in sleep disorders. Endocrinol Metab Clin North Am 2002; 31:15–36.
- Vgontzas AN, Zoumakis M, Papanicolaou DA, et al. Chronic insomnia is associated with a shift of interleukin-6 and tumor necrosis factor secretion from nighttime to daytime. Metabolism 2002; 51:887–892.
- Mills PJ, Parker B, Jones V, et al. The effects of standard anthracycline-based chemotherapy on soluble ICAM-1 and vascular endothelial growth factor levels in breast cancer. Clin Cancer Res 2004; 10:4998–5003.
- Reiter RJ, Tan DX, Korkmaz A, et al. Light at night, chronodisruption, melatonin suppression, and cancer risk: a review. Crit Rev Oncog 2007; 13:303–328.
- Schernhammer ES, Hankinson SE. Urinary melatonin levels and breast cancer risk. J Natl Cancer Inst 2005; 97:1084–1087.
- Travis RC, Allen DS, Fentiman IS, Key TJ. Melatonin and breast cancer: a prospective study. J Natl Cancer Inst 2004; 96:475–482.
- Verkasalo PK, Lillberg K, Stevens RG, et al. Sleep duration and breast cancer: a prospective cohort study. Cancer Res 2005; 65:9595–9600.
- Wu AH, Wang R, Koh WP, Stanczyk FZ, Lee HP, Yu MC. Sleep duration, melatonin and breast cancer among Chinese women in Singapore. Carcinogenesis 2008; 29:1244–1248.
- McElroy JA, Newcomb PA, Titus-Ernstoff L, Trentham-Dietz A, Hampton JM, Egan KM. Duration of sleep and breast cancer risk in a large population-based case-control study. J Sleep Res 2006; 15:241–249.
- Pinheiro SP, Schernhammer ES, Tworoger SS, Michels KB. A prospective study on habitual duration of sleep and incidence of breast cancer in a large cohort of women. Cancer Res 2006; 66:5521–5525.
- Lie JA, Roessink J, Kjaerheim K. Breast cancer and night work among Norwegian nurses. Cancer Causes Control 2006; 17:39–44.
- Schernhammer ES, Kroenke CH, Laden F, Hankinson SE. Night work and risk of breast cancer. Epidemiology 2006; 17:108–111.
- Schernhammer ES, Laden F, Speizer FE, et al. Rotating night shifts and risk of breast cancer in women participating in the Nurses’ Health Study. J Natl Cancer Inst 2001; 93:1563–1568.
- Davis S, Mirick DK, Stevens RG. Night shift work, light at night, and risk of breast cancer. J Natl Cancer Inst 2001; 93:1557–1562.
- Hansen J. Light at night, shiftwork, and breast cancer risk. J Natl Cancer Inst 2001; 93:1513–1515.
- Thompson CL, Larkin EK, Patel S, Berger NA, Redline S, Li L. Short duration of sleep increases risk of colorectal adenoma. Cancer 2011; 117:841–847.
- Degner LF, Sloan JA. Symptom distress in newly diagnosed ambulatory cancer patients and as a predictor of survival in lung cancer. J Pain Symptom Manage 1995; 10:423–431.
- Thompson CL, Li L. Association of sleep duration and breast cancer OncotypeDX recurrence score. Breast Cancer Res Treat 2012; 134:1291–1295.
- Schutte-Rodin S, Broch L, Buysse D, Dorsey C, Sateia M. Clinical guideline for the evaluation and management of chronic insomnia in adults. J Clin Sleep Med 2008; 4:487–504.
- Fan HG, Houédé-Tchen N, Yi QL, et al. Fatigue, menopausal symptoms, and cognitive function in women after adjuvant chemotherapy for breast cancer: 1- and 2-year follow-up of a prospective controlled study. J Clin Oncol 2005; 23:8025–8032.
- Ganz PA. Late effects of cancer and its treatment. Semin Oncol Nurs 2001; 17:241–248.
- Lee TS, Kilbreath SL, Refshauge KM, Pendlebury SC, Beith JM, Lee MJ. Quality of life of women treated with radiotherapy for breast cancer. Support Care Cancer 2008; 16:399–405.
- National Institutes of Health. National Institutes of Health state of the science conference statement on manifestations and management of chronic insomnia in adults, June 13–15, 2005. Sleep 2005; 28:1049–1057.
- Smith MT, Huang MI, Manber R. Cognitive behavior therapy for chronic insomnia occurring within the context of medical and psychiatric disorders. Clin Psychol Rev 2005; 25:559–592.
- Quesnel C, Savard J, Simard S, Ivers H, Morin CM. Efficacy of cognitive-behavioral therapy for insomnia in women treated for nonmetastatic breast cancer. J Consult Clin Psychol 2003; 71:189–200.
- Savard J, Simard S, Ivers H, Morin CM. Randomized study on the efficacy of cognitive-behavioral therapy for insomnia secondary to breast cancer, part I: sleep and psychological effects. J Clin Oncol 2005; 23:6083–6096.
- Berger AM, Kuhn BR, Farr LA, et al. Behavioral therapy intervention trial to improve sleep quality and cancer-related fatigue. Psychooncology 2009; 18:634–646.
- Espie CA, Fleming L, Cassidy J, et al. Randomized controlled clinical effectiveness trial of cognitive behavior therapy compared with treatment as usual for persistent insomnia in patients with cancer. J Clin Oncol 2008; 26:4651–4658.
- National Institutes of Health. National Institutes of Health state of the science conference statement on manifestations and management of chronic insomnia in adults, June 13–15, 2005. Sleep 2005; 28:1049–1057.
- Savard J, Villa J, Simard S, Ivers H, Morin CM. Feasibility of a self-help treatment for insomnia comorbid with cancer. Psychooncology 2011; 20:1013–1019.
- Derogatis LR, Feldstein M, Morrow G, et al. A survey of psychotropic drug prescriptions in an oncology population. Cancer 1979; 44:1919–1929.
- Stiefel FC, Kornblith AB, Holland JC. Changes in the prescription patterns of psychotropic drugs for cancer patients during a 10-year period. Cancer 1990; 65:1048–1053.
- Minton O, Richardson A, Sharpe M, Hotopf M, Stone P. A systematic review and meta-analysis of the pharmacological treatment of cancer-related fatigue. J Natl Cancer Inst 2008; 100:1155–1166.
- Minton O, Stone P, Richardson A, Sharpe M, Hotopf M. Drug therapy for the management of cancer-related fatigue. Cochrane Database Syst Rev 2008;CD006704.
- Krystal AD, Walsh JK, Laska E, et al. Sustained efficacy of eszopiclone over 6 months of nightly treatment: results of a randomized, double-blind, placebo-controlled study in adults with chronic insomnia. Sleep 2003; 26:793–799.
- Liu J, Wang LN. Ramelteon in the treatment of chronic insomnia: systematic review and meta-analysis. Int J Clin Pract 2012; 66:867–873.
- Cankurtaran ES, Ozalp E, Soygur H, Akbiyik DI, Turhan L, Alkis N. Mirtazapine improves sleep and lowers anxiety and depression in cancer patients: superiority over imipramine. Support Care Cancer 2008; 16:1291–1298.
- Palesh OG, Mustian KM, Peppone LJ, et al. Impact of paroxetine on sleep problems in 426 cancer patients receiving chemotherapy: a trial from the University of Rochester Cancer Center Community Clinical Oncology Program. Sleep Med 2012; 13:1184–1190.
KEY POINTS
- Sleep disturbances, primarily insomnia, profoundly affect all aspects of quality of life.
- Insomnia can be caused or worsened by a number of other conditions, such as pain, fatigue, depression, and anxiety, and these in turn can be worsened by insomnia.
- Cognitive-behavioral therapy is the treatment of choice for chronic insomnia. Underlying problems should be addressed.
- Drugs are often prescribed to help cancer patients sleep but should be used with caution, as there is limited information from clinical trials in this population.
Myasthenia gravis: Newer therapies offer sustained improvement
Current therapies for myasthenia gravis can help most patients achieve sustained improvement. The overall prognosis has dramatically improved over the last 4 decades: the mortality rate used to be 75%; now it is 4.5%.1
Myasthenia gravis is the most common disorder of neuromuscular junction transmission and is also one of the best characterized autoimmune diseases. However, its symptoms—primarily weakness—vary from patient to patient, and in the same patient, by time of day and over longer time periods. The variation in symptoms can be very confusing to undiagnosed patients and puzzling to unsuspecting physicians. Such diagnostic uncertainty can give the patient additional frustration and emotional stress, which in turn exacerbate his or her condition.
In this review, we will give an overview of the pathogenesis, clinical manifestations, diagnosis, and treatment of myasthenia gravis.
TWO PEAKS IN INCIDENCE BY AGE
The annual incidence of myasthenia gravis is approximately 10 to 20 new cases per million, with a prevalence of about 150 to 200 per million.2
The age of onset has a bimodal distribution, with an early incidence peak in the second to third decade with a female predominance and a late peak in the 6th to the 8th decade with a male predominance.2
Myasthenia gravis is commonly associated with several other autoimmune disorders, including hypothyroidism, hyperthyroidism, systemic lupus erythematosus, rheumatoid arthritis, vitiligo, diabetes, and, more recently recognized, neuromyelitis optica.3
ANTIBODIES AGAINST AChR AND MuSK
In most cases of myasthenia gravis the patient has autoimmune antibodies against constituents of the neuromuscular junction, specifically acetylcholine receptor (AChR) and muscle-specific tyrosine kinase (MuSK) (Figure 1).
AChR antibody-positive myasthenia gravis
When antibodies bind to AChR on the postsynaptic membrane, they cross-link neighboring AChR units, which are absorbed into the muscle fiber and are broken up.4 In addition, the complement system is activated to mediate further damage on the postsynaptic membrane.
AChR antibodies may come from germinal centers of the thymus, where clustered myoid cells express AChR on the plasma membrane surface.5 About 60% of AChR antibody-positive myasthenia gravis patients have an enlarged thymus, and 10% have a thymoma—a tumor of the epithelial cells of this organ. Conversely, about 15% of patients with a thymoma have clinical myasthenia gravis, and an additional 20% possess antibodies against AChR in the serum without myasthenic symptoms.5
MuSK antibody-positive myasthenia gravis
Like AChR, MuSK is a transmembrane component of the postsynaptic neuromuscular junction. During formation of the neuromuscular junction, MuSK is activated through the binding of agrin (a nerve-derived proteoglycan) to lipoprotein-related protein 4 (LRP4), after which complicated intracellular signaling promotes the assembly and stabilization of AChR.6
Unlike AChR antibodies, antibodies against MuSK do not activate the complement system, and complement fixation is not essential for clinical myasthenic symptoms to appear.7 Also, myasthenia gravis with MuSK antibodies is rarely associated with thymoma.8
The precise mechanism by which MuSK antibody impairs transmission at the neuromuscular junction has been a mystery until recently. Animal models, including MuSK-mutant mice and mice injected with MuSK protein or with purified immunoglobulin G from patients with this disease, have revealed a significant reduction of AChR clusters and destruction of neuromuscular junction structures.7,9–12
In addition, MuSK antibodies produce pre-synaptic dysfunction, manifesting as a reduction of acetylcholine content. This information is based on studies in mice and on in vitro electrophysiologic analyses of neuromuscular junctions from a patient with this disease.7,9–13
Finally, MuSK antibodies may indirectly affect the recycling of acetylcholine. After post-synaptic activation, acetylcholine is normally hydrolized by acetylcholinesterase, which is located in the synaptic cleft but anchored to MuSK on the postsynaptic membrane. MuSK antibodies block the binding of MuSK to acetylcholinesterase, possibly leading to less accumulation of acetylcholinesterase.14 This process may explain why patients with MuSK antibody-positive myasthenia gravis tend to respond poorly to acetylcholinesterase inhibitors (more about this below).
Seronegative myasthenia gravis
In a series of 562 consecutive patients with generalized weakness due to myasthenia gravis, 92% were positive for AChR antibody, 3% were positive for MuSK antibody, and 5% were seronegative (possessing neither antibody).15 In contrast, about 50% of patients with purely ocular myasthenia gravis (ie, with isolated weakness of the levator palpebrae superioris, orbicularis oculi, or oculomotor muscles) are seropositive for AChR antibody. Only a few ocular MuSK antibody-positive cases have been described, leaving the rest seronegative. Rarely, both antibodies can be detected in the same patient.16
In patients who are negative for AChR antibodies at the time of disease onset, sero-conversion may occur later during the course. Repeating serologic testing 6 to 12 months later may detect AChR antibodies in approximately 15% of patients who were initially seronegative.15,17
The clinical presentation, electrophysiologic findings, thymic pathologic findings, and treatment responses are similar in AChR antibody-positive and seronegative myasthenia gravis.17 Muscle biopsy study in seronegative cases demonstrates a loss of AChR as well.18
Based on these observations, it has been proposed that seronegative patients may have low-affinity antibodies that can bind to tightly clustered AChRs on the postsynaptic membrane but escape detection by routine radioimmunoassays in a solution phase. With a sensitive cell-based immunofluorescence assay, low-affinity antibodies to clustered AChRs were detected in 66% of patients with generalized myasthenia gravis and in 50% of those with ocular myasthenia gravis who were seronegative on standard assays.19,20 These low-affinity AChR antibodies can also activate complement in vitro, increasing the likelihood that they are pathogenic. However, assays to detect low-affinity AChR antibodies are not yet commercially available.
Within the past year, three research groups independently reported detecting antibodies to LRP4 in 2% to 50% of seronegative myasthenia gravis patients. This wide variation in the prevalence of LRP4 antibodies could be related to patient ethnicity and methods of detection.21–23 LRP4 is a receptor for agrin and is required for agrin-induced MuSK activation and AChR clustering. LRP antibodies can activate complement; therefore, it is plausible that LRP4 antibody binding leads to AChR loss on the postsynaptic membrane. However, additional study is needed to determine if LRP4 antibodies are truly pathogenic in myasthenia gravis.
A DISORDER OF FATIGABLE WEAKNESS
Myasthenia gravis is a disorder of fatigable weakness producing fluctuating symptoms. Symptoms related to the involvement of specific muscle groups are listed in Table 1. Muscle weakness is often worse later in the day or after exercise.
Ocular myasthenia gravis accounts for about 15% of all cases. Of patients initially presenting with ocular symptoms only, twothirds will ultimately develop generalized symptoms, most within the first 2 years.24 No factor has been identified that predicts conversion from an ocular to a generalized form.
Several clinical phenotypes of MuSK antibody-positive myasthenia gravis have been described. An oculobulbar form presents with diplopia, ptosis, dysarthria, and profound atrophy of the muscles of the tongue and face. A restricted myopathic form presents with prominent neck, shoulder, and respiratory weakness without ocular involvement. A third form is a combination of ocular and proximal limb weakness, indistinguishable from AChR antibody-positive disease.25
MuSK antibody-positive patients do not respond as well to acetylcholinesterase inhibitors as AChR antibody-positive patients do. In one study, nearly 70% of MuSK antibody-positive patients demonstrated no response, poor tolerance, or cholinergic hypersensitivity to these agents.25 Fortunately, most MuSK antibody-positive patients have a favorable response to immunosuppressive therapy—sometimes a dramatic improvement after plasmapheresis.8
DIAGNOSIS OF MYASTHENIA GRAVIS
The common differential diagnoses for myasthenia gravis are listed in Table 2.
The essential feature of myasthenia gravis is fluctuating muscle weakness, often with fatigue. Many patients complain of weakness of specific muscle groups after their repeated usage. Pain is generally a less conspicuous symptom, and generalized fatigue without objective weakness is inconsistent with myasthenia gravis.
Signs of muscle weakness may include droopy eyelids, diplopia, inability to hold the head straight, difficulty swallowing or chewing, speech disturbances, difficulty breathing, and difficulty raising the arms or rising from the sitting position. A historical pattern of ptosis alternating from one eye to the other is fairly characteristic of myasthenia gravis.
The weakness of orbicularis oculi is easily identified on examination by prying open the eyes during forced eye closure. Limb weakness is usually more significant in the arms than in the legs. An often-neglected feature of myasthenia gravis is finger extensor weakness with a relative sparing of other distal hand muscles.2
The ice-pack test is performed by placing a small bag of ice over the ptotic eye for 2 to 5 minutes and assessing the degree of ptosis for any noticeable improvement. This test is not very helpful for assessing ocular motor weakness.
The edrophonium (Tensilon) test can be used for patients with ptosis or ophthalmoparesis. Edrophonium, a short-acting acetylcholinesterase inhibitor, is given intravenously while the patient is observed for objective improvement. The patient’s cardiovascular status should be monitored for arrhythmias and hypotension. Atropine should be immediately available in case severe bradycardia develops.
The ice-pack test and the edrophonium test can give false-negative and false-positive results, and the diagnosis of myasthenia gravis must be verified by other diagnostic tests.
Testing for antibodies
Testing for circulating AChR antibodies, MuSK antibodies, or both is the first step in the laboratory confirmation of myasthenia gravis.
There are three AChR antibody subtypes: binding, blocking, and modulating. Binding antibodies are present in 80% to 90% of patients with generalized myasthenia gravis and 50% of those with ocular myasthenia gravis. Testing for blocking and modulating AChR antibodies increases the sensitivity by less than 5% when added to testing for binding antibodies.
AChR antibody titers correlate poorly with disease severity between patients. However, in individual patients, antibody titers tend to go down in parallel with clinical improvement.
MuSK antibody is detected in nearly half of myasthenia gravis patients with generalized weakness who are negative for AChR antibody.
Electrophysiologic tests
Electrophysiologic tests can usually confirm the diagnosis of seronegative myasthenia gravis. They are also helpful in seropositive patients who have unusual clinical features or a poor response to treatment.
Repetitive nerve stimulation studies use a slow rate (2–5 Hz) of repetitive electrical stimulation. The study is positive if the motor response declines by more than 10%. However, a decremental response is not specific for myasthenia gravis, as it may be seen in other neuromuscular disorders such as motor neuron disease or Lambert-Eaton myasthenic syndrome.
This test is technically easier to do in distal muscles than in proximal muscles, but less sensitive. Therefore, proximal muscles such as the trapezius or facial muscles are usually also sampled to maximize the yield. To further maximize the sensitivity, muscles being tested should be warm, and acetylcholinesterase inhibitors should be withheld for 12 hours before.
Repetitive nerve stimulation studies in distal muscles are positive in approximately 75% of patients with generalized myasthenia gravis and in 30% with ocular myasthenia gravis.26
Single-fiber electromyography is more technically demanding than repetitive nerve stimulation and is less widely available. It is usually performed with a special needle electrode that can simultaneously identify action potentials arising from individual muscle fibers innervated by the same axon.
Variability in time of the second action potential relative to the first is called “jitter.” Abnormal jitter is seen in more than 95% of patients with generalized myasthenia gravis and in 85% to 90% of those with ocular myasthenia gravis.26,27 However, abnormal jitter can also be seen in other neuromuscular diseases such as motor neuron disease or in neuromuscular junctional disorders such as Lambert-Eaton myasthenic syndrome.
Imaging studies
Chest computed tomography or magnetic resonance imaging with contrast should be performed in all myasthenia gravis patients to look for a thymoma.
TREATMENT OF MYASTHENIA GRAVIS
Acetylcholinesterase inhibitors
As a reasonable first therapy in mild cases of myasthenia gravis, acetylcholinesterase inhibitors slow down the degradation of acetylcholine and prolong its effect in the neuromuscular junction, but they are not disease-modifying and their benefits are mild.
Pyridostigmine is the usual choice of acetylcholinesterase inhibitor. Its onset of action is rapid (15 to 30 minutes) and its action lasts for 3 to 4 hours. For most patients, the effective dosage range is 60 mg to 90 mg every 4 to 6 hours. A long-acting form is also available and can be given as a single nighttime dose.
Immunomodulating therapy
Patients who have moderate to severe symptoms require some form of immunomodulating therapy.
Plasmapheresis or intravenous immune globulin is reserved for patients with severe or rapidly worsening disease because their beneficial effects can be seen within the first week of treatment.
Longer-acting immunotherapies (corticosteroids, azathioprine, mycophenolate mofetil and others) have a slower onset of responses but provide sustained benefits. Which drug to use depends on factors such as comorbidity, side effects, and cost.
Drugs to avoid
A number of medications can exacerbate weakness in myasthenia gravis and should be avoided or used with caution. The list is long, but ones that deserve the most attention are penicillamine, interferons, procainamide, quinidine, and antibiotics, including quinolones and aminoglycosides. A more comprehensive list of medications that may exacerbate myasthenia gravis symptoms can be found in a review by Keesey.2
RAPID INDUCTION IMMUNOTHERAPIES : PLASMAPHERESIS, IMMUNE GLOBULIN
Both plasmapheresis and intravenous immune globulin act quickly over days, but in most patients their effects last only a few weeks. Both are used as rescue therapies for myasthenic crises, bridging therapy to slow-acting immunotherapeutic agents, or maintenance treatment for poorly controlled cases.
Several retrospective studies have confirmed the efficacy of plasmapheresis in more than 80% of patients with generalized symptoms.28,29
In a randomized trial in patients with generalized therapies, intravenous immune globulin improved muscle strength in the group of patients with severe symptoms.30 The effective dosage of intravenous immune globulin varies from 1 to 2 g/kg without observed difference between doses.31 Trials comparing the efficacy of intravenous immune globulin and plasmapheresis in acute and severe myasthenia gravis did not reveal a difference in efficacy.32,33 Intravenous immune globulin at a minimal dose of 0.4 g/kg every 3 months has been successfully used as a long-term maintenance monotherapy, and such a role could be expanded to more patients with further studies.34
The choice between plasmapheresis and intravenous immune globulin is often based on the ability of a patient to tolerate each treatment and on the availability of the plasmapheresis procedure. Intravenous immune globulin is easier to administer, is associated with fewer adverse events related to vascular access, and is therefore more appropriate than plasmapheresis in some centers.
CHRONIC MAINTENANCE IMMUNOMODULATING TREATMENT
Corticosteroids
Prednisone, the most commonly used agent, leads to remission or marked improvement in 70% to 80% of patients with ocular or generalized myasthenia gravis.35 It may also reduce the progression of ocular myasthenia gravis to the generalized form.36
The effective dose of prednisone depends on the severity and distribution of symptoms. Some patients may need up to 1.0 mg/kg/day (usually 50 to 80 mg per day). In patients with mild to moderate symptoms, a lower maximal dosage such as 20 to 40 mg per day can be sufficient.
Within 1 to 2 weeks after starting high-dose prednisone, up to 50% of patients may develop a transient deterioration, including possible precipitation of a myasthenic crisis.37 For this reason, high-dose prednisone is commonly started only in hospitalized patients who are also receiving plasmapheresis or intravenous immune globulin. Otherwise, an outpatient dose-escalation protocol can be used to achieve a target dose over several weeks.
Prednisone tapering can begin after the patient has been on the maximal dose for 1 to 2 months and significant improvement is evident. A monthly tapering of 5 to 10 mg is preferred, then more slowly after the daily dose reaches 30 mg. The usual maintenance dose averages about 5 mg daily.
Common side effects of prednisone include weight gain, cushingoid features, easy bruising, cataracts, glaucoma, hypertension, diabetes, dyslipidemia, and osteoporosis. Patients are advised to take supplemental calcium (1,500 mg per day) and vitamin D (400 to 800 IU per day). For those most at risk of osteoporosis, treatment with a bisphosphonate should be considered.
Other immunotherapeutic agents are often needed, either to replace the corticosteroid or to permit use of lower doses of it. Because of their delayed onset of action, starting such corticosteroid-sparing agents early in the course is often necessary. These agents are often initially combined with high-dose prednisone, with an eventual goal of weaning off prednisone entirely. This strategy offers the advantage of relatively rapid induction while avoiding the long-term adverse effects of corticosteroid treatment.
Azathioprine
Azathioprine doesn’t begin to show a beneficial effect in myasthenia gravis for 6 to 12 months, and it often reaches its maximal efficacy only after 1 to 2 years of treatment.38
In a study of 78 myasthenia gravis patients, 91% improved when treated with azathioprine alone or together with prednisone.39 In another study using azathioprine and prednisolone for generalized myasthenia gravis, nearly two-thirds of patients came off prednisolone while maintaining remission for 3 years.38
A typical maintenance dose is 2 to 3 mg/kg/day. Common side effects are nausea, vomiting, and malaise. Less frequent side effects include hematologic abnormalities, abnormal liver function, and pancreatitis. Monthly monitoring of complete blood cell counts and liver function tests is warranted for the first 6 months, then less often.
One in 300 people in the general population is homozygous for a mutant allele in the thiopurine methyltransferase (TPMT) gene. Patients with this genotype should not receive azathioprine because of the risk of life-threatening bone marrow suppression.40 A slightly increased risk of various forms of lymphoma has been documented.41
Mycophenolate mofetil
A well-tolerated medication with few side effects, mycophenolate mofetil is being used more in myasthenia gravis. The results of two recent randomized trials suggested that it is not effective in improving myasthenia gravis symptoms or sparing prednisone dosage when used for 90 days or 36 weeks.42,43 However, extensive clinical experience supports its longterm efficacy in myasthenia gravis.
In a retrospective study of 85 patients with generalized myasthenia gravis, mycophenolate at doses of 1 to 3 g daily improved symptoms in 73% and produced remission in 50%. Steroid dosage was reduced in 71% of patients.44
Another retrospective study, with 102 patients, verified a slow development of clinical benefit after months of mycophenolate therapy alone or in combination with prednisone. Approximately 50% of patients achieved a minimal manifestation status after 6 to 12 months of mycophenolate treatment. Eventually, at 24 months of treatment, 80% of patients had a desirable outcome of minimal clinical manifestation or better, 55% of patients were able to come off prednisone entirely, and 75% were taking less than 7.5 mg of prednisone per day.45
Common side effects of mycophenolate include nausea, diarrhea, and infections such as urinary tract infections and herpes reactivation. The complete blood cell count needs to be monitored frequently during the first 6 months of therapy. Leukopenia can occur but rarely necessitates stopping mycophenolate. Long-term safety data are lacking, but so far there has been no clearly increased risk of malignancy.
Mycophenolate exposure in pregnancy results in a high incidence of major fetal malformations. Therefore, its use in pregnant patients is discouraged, and women of child-bearing age should use effective contraception.46
Cyclosporine
A randomized trial in a small number of patients suggested that cyclosporine is fairly effective as monotherapy.47 Its onset of action in myasthenia gravis is faster than that of other corticosteroid-sparing agents, and clinical benefit can often be observed as early as 1 to 2 months. A dose of 5 mg/kg/day and a maintenance serum level of 100 to 150 ng/mL are generally recommended. However, renal, hepatic, and hematologic toxicities and interactions with other medications make cyclosporine a less attractive choice.
Methotrexate
A randomized trial evaluated the utility of methotrexate as a steroid-sparing agent compared with azathioprine.48 At 24 months, its steroid-sparing effect was similar to that of azathioprine, and the prednisone dosage had been reduced in more than 50% of patients.
Another phase II trial studying the efficacy of methotrexate in myasthenia gravis is under way.49
Rituximab
Rituximab is a monoclonal antibody against B-cell membrane marker CD20. A growing number of case series support its efficacy in patients with severe generalized myasthenia gravis refractory to multiple immunosuppressants.16,50 It seems particularly effective for MuSK antibody-positive disease, reducing MuSK antibody titers and having a treatment effect that lasts for years.
The standard dosage is 375 mg/m2 per week for 4 consecutive weeks. Peripheral B cells tend to be depleted within 2 weeks after the first infusion, while T-cell populations remain unchanged.50
A minimal infusion reaction such as flushing and chills can be seen with the first infusion. Patients may be more susceptible to certain infections such as reactivation of herpes zoster, but overall rituximab is well tolerated. Rare cases of progressive multifocal leukoencephalopathy have been reported in patients taking it, but none have occurred so far in myasthenia gravis treatment.
Cyclophosphamide
Cyclophosphamide is an alkylating agent that reduces proliferation of both B and T cells. It can be effective in myasthenia gravis, but potentially serious side effects limit its use. It should be reserved for the small percentage of cases that are refractory to other immunotherapies.
Thymectomy
Surgical treatment should be considered for patients with thymoma. If the tumor cannot be surgically resected, chemoradiotherapy can be considered for relief of myasthenic symptoms and for prevention of local invasion.
Thymomas recur in a minority of patients many years after the initial resection, sometimes without myasthenia gravis symptoms. A recurrence of symptoms does not necessarily indicate a recurrence of thymoma. The lack of correlation between myasthenia gravis symptoms and thymoma recurrence highlights the importance of radiologic follow-up in these patients.
For patients without thymoma, many experts believe that thymectomy is beneficial in patients under age 60 who have generalized myasthenia gravis. The likelihood of medication-free remission is about twice as high, and the likelihood of becoming asymptomatic is about one and a half times higher after thymectomy.51 However, it takes up to several years for the benefits of thymectomy to manifest, and thymectomy does not guarantee protection from developing AChR antibody-positive myasthenia gravis in the future.
The optimal timing of thymectomy is not well established; however, the procedure is usually recommended within the first 3 years of diagnosis.52 The response rates from thymectomy are similar for AChR antibody-positive and seronegative patients. In general, thymectomy for MuSK antibody-positive patients has not been effective, and its role in ocular myasthenia gravis is unclear.2,53
- Alshekhlee A, Miles JD, Katirji B, Preston DC, Kaminski HJ. Incidence and mortality rates of myasthenia gravis and myasthenic crisis in US hospitals. Neurology 2009; 72:1548–1554.
- Keesey JC. Clinical evaluation and management of myasthenia gravis. Muscle Nerve 2004; 29:484–505.
- Leite MI, Coutinho E, Lana-Peixoto M, et al. Myasthenia gravis and neuromyelitis optica spectrum disorder: a multicenter study of 16 patients. Neurology 2012; 78:1601–1607.
- Drachman DB, Angus CW, Adams RN, Michelson JD, Hoffman GJ. Myasthenic antibodies cross-link acetylcholine receptors to accelerate degradation. N Engl J Med 1978; 298:1116–1122.
- Fujii Y. The thymus, thymoma and myasthenia gravis. Surg Today 2013; 43:461–466.
- Evoli A, Lindstrom J. Myasthenia gravis with antibodies to MuSK: another step toward solving mystery? Neurology 2011; 77:1783–1784.
- Mori S, Kubo S, Akiyoshi T, et al. Antibodies against muscle-specific kinase impair both presynaptic and postsynaptic functions in a murine model of myasthenia gravis. Am J Pathol 2012; 180:798–810.
- Guptill JT, Sanders DB, Evoli A. Anti-MuSK antibody myasthenia gravis: clinical findings and response to treatment in two large cohorts. Muscle Nerve 2011; 44:36–40.
- Chevessier F, Girard E, Molgó J, et al. A mouse model for congenital myasthenic syndrome due to MuSK mutations reveals defects in structure and function of neuromuscular junctions. Hum Mol Genet 2008; 17:3577–3595.
- Richman DP, Nishi K, Morell SW, et al. Acute severe animal model of anti-muscle-specific kinase myasthenia: combined postsynaptic and presynaptic changes. Arch Neurol 2012; 69:453–460.
- Klooster R, Plomp JJ, Huijbers MG, et al. Muscle-specific kinase myasthenia gravis IgG4 autoantibodies cause severe neuromuscular junction dysfunction in mice. Brain 2012; 135:1081–1101.
- Viegas S, Jacobson L, Waters P, et al. Passive and active immunization models of MuSK-Ab positive myasthenia: electrophysiological evidence for pre and postsynaptic defects. Exp Neurol 2012; 234:506–512.
- Niks EH, Kuks JB, Wokke JH, et al. Pre- and postsynaptic neuromuscular junction abnormalities in musk myasthenia. Muscle Nerve 2010; 42:283–288.
- Kawakami Y, Ito M, Hirayama M, et al. Anti-MuSK autoantibodies block binding of collagen Q to MuSK. Neurology 2011; 77:1819–1826.
- Chan KH, Lachance DH, Harper CM, Lennon VA. Frequency of seronegativity in adult-acquired generalized myasthenia gravis. Muscle Nerve 2007; 36:651–658.
- Collongues N, Casez O, Lacour A, et al. Rituximab in refractory and non-refractory myasthenia: a retrospective multicenter study. Muscle Nerve 2012; 46:687–691.
- Sanders DB, Andrews PI, Howard JF, Massey JM. Seronegative myasthenia gravis. Neurology 1997; 48(suppl 5):40S–45S.
- Shiraishi H, Motomura M, Yoshimura T, et al. Acetylcholine receptors loss and postsynaptic damage in MuSK antibody-positive myasthenia gravis. Ann Neurol 2005; 57:289–293.
- Leite MI, Jacob S, Viegas S, et al. IgG1 antibodies to acetylcholine receptors in ‘seronegative’ myasthenia gravis. Brain 2008; 131:1940–1952.
- Jacob S, Viegas S, Leite MI, et al. Presence and pathogenic relevance of antibodies to clustered acetylcholine receptor in ocular and generalized myasthenia gravis. Arch Neurol 2012; 69:994–1001.
- Higuchi O, Hamuro J, Motomura M, Yamanashi Y. Autoantibodies to low-density lipoprotein receptor-related protein 4 in myasthenia gravis. Ann Neurol 2011; 69:418–422.
- Pevzner A, Schoser B, Peters K, et al. Anti-LRP4 autoantibodies in AChR- and MuSK-antibody-negative myasthenia gravis. J Neurol 2012; 259:427–435.
- Zhang B, Tzartos JS, Belimezi M, et al. Autoantibodies to lipoprotein-related protein 4 in patients with double-seronegative myasthenia gravis. Arch Neurol 2012; 69:445–451.
- Kupersmith MJ, Latkany R, Homel P. Development of generalized disease at 2 years in patients with ocular myasthenia gravis. Arch Neurol 2003; 60:243–248.
- Pasnoor M, Wolfe GI, Nations S, et al. Clinical findings in MuSK-antibody positive myasthenia gravis: a US experience. Muscle Nerve 2010; 41:370–374.
- Oh SJ, Kim DE, Kuruoglu R, Bradley RJ, Dwyer D. Diagnostic sensitivity of the laboratory tests in myasthenia gravis. Muscle Nerve 1992; 15:720–724.
- Sanders DB, Stålberg EV. AAEM minimonograph #25: single-fiber electromyography. Muscle Nerve 1996; 19:1069–1083.
- Lazo-Langner A, Espinosa-Poblano I, Tirado-Cárdenas N, et al. Therapeutic plasma exchange in Mexico: experience from a single institution. Am J Hematol 2002; 70:16–21.
- Carandina-Maffeis R, Nucci A, Marques JF, et al. Plasmapheresis in the treatment of myasthenia gravis: retrospective study of 26 patients. Arq Neuropsiquiatr 2004; 62:391–395.
- Zinman L, Ng E, Bril V. IV immunoglobulin in patients with myasthenia gravis: a randomized controlled trial. Neurology 2007; 68:837–841.
- Gajdos P, Tranchant C, Clair B, et al; Myasthenia Gravis Clinical Study Group. Treatment of myasthenia gravis exacerbation with intravenous immunoglobulin: a randomized double-blind clinical trial. Arch Neurol 2005; 62:1689–1693.
- Rønager J, Ravnborg M, Hermansen I, Vorstrup S. Immunoglobulin treatment versus plasma exchange in patients with chronic moderate to severe myasthenia gravis. Artif Organs 2001; 25:967–973.
- Barth D, Nabavi Nouri M, Ng E, Nwe P, Bril V. Comparison of IVIg and PLEX in patients with myasthenia gravis. Neurology 2011; 76:2017–2023.
- Wegner B, Ahmed I. Intravenous immunoglobulin monotherapy in long-term treatment of myasthenia gravis. Clin Neurol Neurosurg 2002; 105:3–8.
- Pascuzzi RM, Coslett HB, Johns TR. Long-term corticosteroid treatment of myasthenia gravis: report of 116 patients. Ann Neurol 1984; 15:291–298.
- Monsul NT, Patwa HS, Knorr AM, Lesser RL, Goldstein JM. The effect of prednisone on the progression from ocular to generalized myasthenia gravis. J Neurol Sci 2004; 217:131–133.
- Miller RG, Milner-Brown HS, Mirka A. Prednisone-induced worsening of neuromuscular function in myasthenia gravis. Neurology 1986; 36:729–732.
- Palace J, Newsom-Davis J, Lecky B. A randomized double-blind trial of prednisolone alone or with azathioprine in myasthenia gravis. Myasthenia Gravis Study Group. Neurology 1998; 50:1778–1783.
- Mertens HG, Hertel G, Reuther P, Ricker K. Effect of immunosuppressive drugs (azathioprine). Ann N Y Acad Sci 1981; 377:691–699.
- Relling MV, Gardner EE, Sandborn WJ, et al; Clinical Pharmacogenetics Implementation Consortium. Clinical Pharmacogenetics Implementation Consortium guidelines for thiopurine methyltransferase genotype and thiopurine dosing. Clin Pharmacol Ther 2011; 89:387–391.
- Finelli PF. Primary CNS lymphoma in myasthenic on long-term azathioprine. J Neurooncol 2005; 74:91–92.
- Sanders DB, Hart IK, Mantegazza R, et al. An international, phase III, randomized trial of mycophenolate mofetil in myasthenia gravis. Neurology 2008; 71:400–406.
- Muscle Study Group. A trial of mycophenolate mofetil with prednisone as initial immunotherapy in myasthenia gravis. Neurology 2008; 71:394–399.
- Meriggioli MN, Ciafaloni E, Al-Hayk KA, et al. Mycophenolate mofetil for myasthenia gravis: an analysis of efficacy, safety, and tolerability. Neurology 2003; 61:1438–1440.
- Hehir MK, Burns TM, Alpers J, Conaway MR, Sawa M, Sanders DB. Mycophenolate mofetil in AChR-antibody-positive myasthenia gravis: outcomes in 102 patients. Muscle Nerve 2010; 41:593–598.
- Merlob P, Stahl B, Klinger G. Tetrada of the possible mycophenolate mofetil embryopathy: a review. Reprod Toxicol 2009; 28:105–108.
- Tindall RS, Rollins JA, Phillips JT, Greenlee RG, Wells L, Belendiuk G. Preliminary results of a double-blind, randomized, placebo-controlled trial of cyclosporine in myasthenia gravis. N Engl J Med 1987; 316:719–724.
- Heckmann JM, Rawoot A, Bateman K, Renison R, Badri M. A single-blinded trial of methotrexate versus azathioprine as steroid-sparing agents in generalized myasthenia gravis. BMC Neurol 2011; 11:97.
- Pasnoor M, He J, Herbelin L, Dimachkie M, Barohn RJ; Muscle Study Group. Phase II trial of methotrexate in myasthenia gravis. Ann N Y Acad Sci 2012; 1275:23–28.
- Díaz-Manera J, Martínez-Hernández E, Querol L, et al. Long-lasting treatment effect of rituximab in MuSK myasthenia. Neurology 2012; 78:189–193.
- Gronseth GS, Barohn RJ. Practice parameter: thymectomy for autoimmune myasthenia gravis (an evidence-based review): Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2000; 55:7–15.
- Kumar V, Kaminski HJ. Treatment of myasthenia gravis. Curr Neurol Neurosci Rep 2011; 11:89–96.
- Pompeo E, Tacconi F, Massa R, Mineo D, Nahmias S, Mineo TC. Long-term outcome of thoracoscopic extended thymectomy for nonthymomatous myasthenia gravis. Eur J Cardiothorac Surg 2009; 36:164–169.
Current therapies for myasthenia gravis can help most patients achieve sustained improvement. The overall prognosis has dramatically improved over the last 4 decades: the mortality rate used to be 75%; now it is 4.5%.1
Myasthenia gravis is the most common disorder of neuromuscular junction transmission and is also one of the best characterized autoimmune diseases. However, its symptoms—primarily weakness—vary from patient to patient, and in the same patient, by time of day and over longer time periods. The variation in symptoms can be very confusing to undiagnosed patients and puzzling to unsuspecting physicians. Such diagnostic uncertainty can give the patient additional frustration and emotional stress, which in turn exacerbate his or her condition.
In this review, we will give an overview of the pathogenesis, clinical manifestations, diagnosis, and treatment of myasthenia gravis.
TWO PEAKS IN INCIDENCE BY AGE
The annual incidence of myasthenia gravis is approximately 10 to 20 new cases per million, with a prevalence of about 150 to 200 per million.2
The age of onset has a bimodal distribution, with an early incidence peak in the second to third decade with a female predominance and a late peak in the 6th to the 8th decade with a male predominance.2
Myasthenia gravis is commonly associated with several other autoimmune disorders, including hypothyroidism, hyperthyroidism, systemic lupus erythematosus, rheumatoid arthritis, vitiligo, diabetes, and, more recently recognized, neuromyelitis optica.3
ANTIBODIES AGAINST AChR AND MuSK
In most cases of myasthenia gravis the patient has autoimmune antibodies against constituents of the neuromuscular junction, specifically acetylcholine receptor (AChR) and muscle-specific tyrosine kinase (MuSK) (Figure 1).
AChR antibody-positive myasthenia gravis
When antibodies bind to AChR on the postsynaptic membrane, they cross-link neighboring AChR units, which are absorbed into the muscle fiber and are broken up.4 In addition, the complement system is activated to mediate further damage on the postsynaptic membrane.
AChR antibodies may come from germinal centers of the thymus, where clustered myoid cells express AChR on the plasma membrane surface.5 About 60% of AChR antibody-positive myasthenia gravis patients have an enlarged thymus, and 10% have a thymoma—a tumor of the epithelial cells of this organ. Conversely, about 15% of patients with a thymoma have clinical myasthenia gravis, and an additional 20% possess antibodies against AChR in the serum without myasthenic symptoms.5
MuSK antibody-positive myasthenia gravis
Like AChR, MuSK is a transmembrane component of the postsynaptic neuromuscular junction. During formation of the neuromuscular junction, MuSK is activated through the binding of agrin (a nerve-derived proteoglycan) to lipoprotein-related protein 4 (LRP4), after which complicated intracellular signaling promotes the assembly and stabilization of AChR.6
Unlike AChR antibodies, antibodies against MuSK do not activate the complement system, and complement fixation is not essential for clinical myasthenic symptoms to appear.7 Also, myasthenia gravis with MuSK antibodies is rarely associated with thymoma.8
The precise mechanism by which MuSK antibody impairs transmission at the neuromuscular junction has been a mystery until recently. Animal models, including MuSK-mutant mice and mice injected with MuSK protein or with purified immunoglobulin G from patients with this disease, have revealed a significant reduction of AChR clusters and destruction of neuromuscular junction structures.7,9–12
In addition, MuSK antibodies produce pre-synaptic dysfunction, manifesting as a reduction of acetylcholine content. This information is based on studies in mice and on in vitro electrophysiologic analyses of neuromuscular junctions from a patient with this disease.7,9–13
Finally, MuSK antibodies may indirectly affect the recycling of acetylcholine. After post-synaptic activation, acetylcholine is normally hydrolized by acetylcholinesterase, which is located in the synaptic cleft but anchored to MuSK on the postsynaptic membrane. MuSK antibodies block the binding of MuSK to acetylcholinesterase, possibly leading to less accumulation of acetylcholinesterase.14 This process may explain why patients with MuSK antibody-positive myasthenia gravis tend to respond poorly to acetylcholinesterase inhibitors (more about this below).
Seronegative myasthenia gravis
In a series of 562 consecutive patients with generalized weakness due to myasthenia gravis, 92% were positive for AChR antibody, 3% were positive for MuSK antibody, and 5% were seronegative (possessing neither antibody).15 In contrast, about 50% of patients with purely ocular myasthenia gravis (ie, with isolated weakness of the levator palpebrae superioris, orbicularis oculi, or oculomotor muscles) are seropositive for AChR antibody. Only a few ocular MuSK antibody-positive cases have been described, leaving the rest seronegative. Rarely, both antibodies can be detected in the same patient.16
In patients who are negative for AChR antibodies at the time of disease onset, sero-conversion may occur later during the course. Repeating serologic testing 6 to 12 months later may detect AChR antibodies in approximately 15% of patients who were initially seronegative.15,17
The clinical presentation, electrophysiologic findings, thymic pathologic findings, and treatment responses are similar in AChR antibody-positive and seronegative myasthenia gravis.17 Muscle biopsy study in seronegative cases demonstrates a loss of AChR as well.18
Based on these observations, it has been proposed that seronegative patients may have low-affinity antibodies that can bind to tightly clustered AChRs on the postsynaptic membrane but escape detection by routine radioimmunoassays in a solution phase. With a sensitive cell-based immunofluorescence assay, low-affinity antibodies to clustered AChRs were detected in 66% of patients with generalized myasthenia gravis and in 50% of those with ocular myasthenia gravis who were seronegative on standard assays.19,20 These low-affinity AChR antibodies can also activate complement in vitro, increasing the likelihood that they are pathogenic. However, assays to detect low-affinity AChR antibodies are not yet commercially available.
Within the past year, three research groups independently reported detecting antibodies to LRP4 in 2% to 50% of seronegative myasthenia gravis patients. This wide variation in the prevalence of LRP4 antibodies could be related to patient ethnicity and methods of detection.21–23 LRP4 is a receptor for agrin and is required for agrin-induced MuSK activation and AChR clustering. LRP antibodies can activate complement; therefore, it is plausible that LRP4 antibody binding leads to AChR loss on the postsynaptic membrane. However, additional study is needed to determine if LRP4 antibodies are truly pathogenic in myasthenia gravis.
A DISORDER OF FATIGABLE WEAKNESS
Myasthenia gravis is a disorder of fatigable weakness producing fluctuating symptoms. Symptoms related to the involvement of specific muscle groups are listed in Table 1. Muscle weakness is often worse later in the day or after exercise.
Ocular myasthenia gravis accounts for about 15% of all cases. Of patients initially presenting with ocular symptoms only, twothirds will ultimately develop generalized symptoms, most within the first 2 years.24 No factor has been identified that predicts conversion from an ocular to a generalized form.
Several clinical phenotypes of MuSK antibody-positive myasthenia gravis have been described. An oculobulbar form presents with diplopia, ptosis, dysarthria, and profound atrophy of the muscles of the tongue and face. A restricted myopathic form presents with prominent neck, shoulder, and respiratory weakness without ocular involvement. A third form is a combination of ocular and proximal limb weakness, indistinguishable from AChR antibody-positive disease.25
MuSK antibody-positive patients do not respond as well to acetylcholinesterase inhibitors as AChR antibody-positive patients do. In one study, nearly 70% of MuSK antibody-positive patients demonstrated no response, poor tolerance, or cholinergic hypersensitivity to these agents.25 Fortunately, most MuSK antibody-positive patients have a favorable response to immunosuppressive therapy—sometimes a dramatic improvement after plasmapheresis.8
DIAGNOSIS OF MYASTHENIA GRAVIS
The common differential diagnoses for myasthenia gravis are listed in Table 2.
The essential feature of myasthenia gravis is fluctuating muscle weakness, often with fatigue. Many patients complain of weakness of specific muscle groups after their repeated usage. Pain is generally a less conspicuous symptom, and generalized fatigue without objective weakness is inconsistent with myasthenia gravis.
Signs of muscle weakness may include droopy eyelids, diplopia, inability to hold the head straight, difficulty swallowing or chewing, speech disturbances, difficulty breathing, and difficulty raising the arms or rising from the sitting position. A historical pattern of ptosis alternating from one eye to the other is fairly characteristic of myasthenia gravis.
The weakness of orbicularis oculi is easily identified on examination by prying open the eyes during forced eye closure. Limb weakness is usually more significant in the arms than in the legs. An often-neglected feature of myasthenia gravis is finger extensor weakness with a relative sparing of other distal hand muscles.2
The ice-pack test is performed by placing a small bag of ice over the ptotic eye for 2 to 5 minutes and assessing the degree of ptosis for any noticeable improvement. This test is not very helpful for assessing ocular motor weakness.
The edrophonium (Tensilon) test can be used for patients with ptosis or ophthalmoparesis. Edrophonium, a short-acting acetylcholinesterase inhibitor, is given intravenously while the patient is observed for objective improvement. The patient’s cardiovascular status should be monitored for arrhythmias and hypotension. Atropine should be immediately available in case severe bradycardia develops.
The ice-pack test and the edrophonium test can give false-negative and false-positive results, and the diagnosis of myasthenia gravis must be verified by other diagnostic tests.
Testing for antibodies
Testing for circulating AChR antibodies, MuSK antibodies, or both is the first step in the laboratory confirmation of myasthenia gravis.
There are three AChR antibody subtypes: binding, blocking, and modulating. Binding antibodies are present in 80% to 90% of patients with generalized myasthenia gravis and 50% of those with ocular myasthenia gravis. Testing for blocking and modulating AChR antibodies increases the sensitivity by less than 5% when added to testing for binding antibodies.
AChR antibody titers correlate poorly with disease severity between patients. However, in individual patients, antibody titers tend to go down in parallel with clinical improvement.
MuSK antibody is detected in nearly half of myasthenia gravis patients with generalized weakness who are negative for AChR antibody.
Electrophysiologic tests
Electrophysiologic tests can usually confirm the diagnosis of seronegative myasthenia gravis. They are also helpful in seropositive patients who have unusual clinical features or a poor response to treatment.
Repetitive nerve stimulation studies use a slow rate (2–5 Hz) of repetitive electrical stimulation. The study is positive if the motor response declines by more than 10%. However, a decremental response is not specific for myasthenia gravis, as it may be seen in other neuromuscular disorders such as motor neuron disease or Lambert-Eaton myasthenic syndrome.
This test is technically easier to do in distal muscles than in proximal muscles, but less sensitive. Therefore, proximal muscles such as the trapezius or facial muscles are usually also sampled to maximize the yield. To further maximize the sensitivity, muscles being tested should be warm, and acetylcholinesterase inhibitors should be withheld for 12 hours before.
Repetitive nerve stimulation studies in distal muscles are positive in approximately 75% of patients with generalized myasthenia gravis and in 30% with ocular myasthenia gravis.26
Single-fiber electromyography is more technically demanding than repetitive nerve stimulation and is less widely available. It is usually performed with a special needle electrode that can simultaneously identify action potentials arising from individual muscle fibers innervated by the same axon.
Variability in time of the second action potential relative to the first is called “jitter.” Abnormal jitter is seen in more than 95% of patients with generalized myasthenia gravis and in 85% to 90% of those with ocular myasthenia gravis.26,27 However, abnormal jitter can also be seen in other neuromuscular diseases such as motor neuron disease or in neuromuscular junctional disorders such as Lambert-Eaton myasthenic syndrome.
Imaging studies
Chest computed tomography or magnetic resonance imaging with contrast should be performed in all myasthenia gravis patients to look for a thymoma.
TREATMENT OF MYASTHENIA GRAVIS
Acetylcholinesterase inhibitors
As a reasonable first therapy in mild cases of myasthenia gravis, acetylcholinesterase inhibitors slow down the degradation of acetylcholine and prolong its effect in the neuromuscular junction, but they are not disease-modifying and their benefits are mild.
Pyridostigmine is the usual choice of acetylcholinesterase inhibitor. Its onset of action is rapid (15 to 30 minutes) and its action lasts for 3 to 4 hours. For most patients, the effective dosage range is 60 mg to 90 mg every 4 to 6 hours. A long-acting form is also available and can be given as a single nighttime dose.
Immunomodulating therapy
Patients who have moderate to severe symptoms require some form of immunomodulating therapy.
Plasmapheresis or intravenous immune globulin is reserved for patients with severe or rapidly worsening disease because their beneficial effects can be seen within the first week of treatment.
Longer-acting immunotherapies (corticosteroids, azathioprine, mycophenolate mofetil and others) have a slower onset of responses but provide sustained benefits. Which drug to use depends on factors such as comorbidity, side effects, and cost.
Drugs to avoid
A number of medications can exacerbate weakness in myasthenia gravis and should be avoided or used with caution. The list is long, but ones that deserve the most attention are penicillamine, interferons, procainamide, quinidine, and antibiotics, including quinolones and aminoglycosides. A more comprehensive list of medications that may exacerbate myasthenia gravis symptoms can be found in a review by Keesey.2
RAPID INDUCTION IMMUNOTHERAPIES : PLASMAPHERESIS, IMMUNE GLOBULIN
Both plasmapheresis and intravenous immune globulin act quickly over days, but in most patients their effects last only a few weeks. Both are used as rescue therapies for myasthenic crises, bridging therapy to slow-acting immunotherapeutic agents, or maintenance treatment for poorly controlled cases.
Several retrospective studies have confirmed the efficacy of plasmapheresis in more than 80% of patients with generalized symptoms.28,29
In a randomized trial in patients with generalized therapies, intravenous immune globulin improved muscle strength in the group of patients with severe symptoms.30 The effective dosage of intravenous immune globulin varies from 1 to 2 g/kg without observed difference between doses.31 Trials comparing the efficacy of intravenous immune globulin and plasmapheresis in acute and severe myasthenia gravis did not reveal a difference in efficacy.32,33 Intravenous immune globulin at a minimal dose of 0.4 g/kg every 3 months has been successfully used as a long-term maintenance monotherapy, and such a role could be expanded to more patients with further studies.34
The choice between plasmapheresis and intravenous immune globulin is often based on the ability of a patient to tolerate each treatment and on the availability of the plasmapheresis procedure. Intravenous immune globulin is easier to administer, is associated with fewer adverse events related to vascular access, and is therefore more appropriate than plasmapheresis in some centers.
CHRONIC MAINTENANCE IMMUNOMODULATING TREATMENT
Corticosteroids
Prednisone, the most commonly used agent, leads to remission or marked improvement in 70% to 80% of patients with ocular or generalized myasthenia gravis.35 It may also reduce the progression of ocular myasthenia gravis to the generalized form.36
The effective dose of prednisone depends on the severity and distribution of symptoms. Some patients may need up to 1.0 mg/kg/day (usually 50 to 80 mg per day). In patients with mild to moderate symptoms, a lower maximal dosage such as 20 to 40 mg per day can be sufficient.
Within 1 to 2 weeks after starting high-dose prednisone, up to 50% of patients may develop a transient deterioration, including possible precipitation of a myasthenic crisis.37 For this reason, high-dose prednisone is commonly started only in hospitalized patients who are also receiving plasmapheresis or intravenous immune globulin. Otherwise, an outpatient dose-escalation protocol can be used to achieve a target dose over several weeks.
Prednisone tapering can begin after the patient has been on the maximal dose for 1 to 2 months and significant improvement is evident. A monthly tapering of 5 to 10 mg is preferred, then more slowly after the daily dose reaches 30 mg. The usual maintenance dose averages about 5 mg daily.
Common side effects of prednisone include weight gain, cushingoid features, easy bruising, cataracts, glaucoma, hypertension, diabetes, dyslipidemia, and osteoporosis. Patients are advised to take supplemental calcium (1,500 mg per day) and vitamin D (400 to 800 IU per day). For those most at risk of osteoporosis, treatment with a bisphosphonate should be considered.
Other immunotherapeutic agents are often needed, either to replace the corticosteroid or to permit use of lower doses of it. Because of their delayed onset of action, starting such corticosteroid-sparing agents early in the course is often necessary. These agents are often initially combined with high-dose prednisone, with an eventual goal of weaning off prednisone entirely. This strategy offers the advantage of relatively rapid induction while avoiding the long-term adverse effects of corticosteroid treatment.
Azathioprine
Azathioprine doesn’t begin to show a beneficial effect in myasthenia gravis for 6 to 12 months, and it often reaches its maximal efficacy only after 1 to 2 years of treatment.38
In a study of 78 myasthenia gravis patients, 91% improved when treated with azathioprine alone or together with prednisone.39 In another study using azathioprine and prednisolone for generalized myasthenia gravis, nearly two-thirds of patients came off prednisolone while maintaining remission for 3 years.38
A typical maintenance dose is 2 to 3 mg/kg/day. Common side effects are nausea, vomiting, and malaise. Less frequent side effects include hematologic abnormalities, abnormal liver function, and pancreatitis. Monthly monitoring of complete blood cell counts and liver function tests is warranted for the first 6 months, then less often.
One in 300 people in the general population is homozygous for a mutant allele in the thiopurine methyltransferase (TPMT) gene. Patients with this genotype should not receive azathioprine because of the risk of life-threatening bone marrow suppression.40 A slightly increased risk of various forms of lymphoma has been documented.41
Mycophenolate mofetil
A well-tolerated medication with few side effects, mycophenolate mofetil is being used more in myasthenia gravis. The results of two recent randomized trials suggested that it is not effective in improving myasthenia gravis symptoms or sparing prednisone dosage when used for 90 days or 36 weeks.42,43 However, extensive clinical experience supports its longterm efficacy in myasthenia gravis.
In a retrospective study of 85 patients with generalized myasthenia gravis, mycophenolate at doses of 1 to 3 g daily improved symptoms in 73% and produced remission in 50%. Steroid dosage was reduced in 71% of patients.44
Another retrospective study, with 102 patients, verified a slow development of clinical benefit after months of mycophenolate therapy alone or in combination with prednisone. Approximately 50% of patients achieved a minimal manifestation status after 6 to 12 months of mycophenolate treatment. Eventually, at 24 months of treatment, 80% of patients had a desirable outcome of minimal clinical manifestation or better, 55% of patients were able to come off prednisone entirely, and 75% were taking less than 7.5 mg of prednisone per day.45
Common side effects of mycophenolate include nausea, diarrhea, and infections such as urinary tract infections and herpes reactivation. The complete blood cell count needs to be monitored frequently during the first 6 months of therapy. Leukopenia can occur but rarely necessitates stopping mycophenolate. Long-term safety data are lacking, but so far there has been no clearly increased risk of malignancy.
Mycophenolate exposure in pregnancy results in a high incidence of major fetal malformations. Therefore, its use in pregnant patients is discouraged, and women of child-bearing age should use effective contraception.46
Cyclosporine
A randomized trial in a small number of patients suggested that cyclosporine is fairly effective as monotherapy.47 Its onset of action in myasthenia gravis is faster than that of other corticosteroid-sparing agents, and clinical benefit can often be observed as early as 1 to 2 months. A dose of 5 mg/kg/day and a maintenance serum level of 100 to 150 ng/mL are generally recommended. However, renal, hepatic, and hematologic toxicities and interactions with other medications make cyclosporine a less attractive choice.
Methotrexate
A randomized trial evaluated the utility of methotrexate as a steroid-sparing agent compared with azathioprine.48 At 24 months, its steroid-sparing effect was similar to that of azathioprine, and the prednisone dosage had been reduced in more than 50% of patients.
Another phase II trial studying the efficacy of methotrexate in myasthenia gravis is under way.49
Rituximab
Rituximab is a monoclonal antibody against B-cell membrane marker CD20. A growing number of case series support its efficacy in patients with severe generalized myasthenia gravis refractory to multiple immunosuppressants.16,50 It seems particularly effective for MuSK antibody-positive disease, reducing MuSK antibody titers and having a treatment effect that lasts for years.
The standard dosage is 375 mg/m2 per week for 4 consecutive weeks. Peripheral B cells tend to be depleted within 2 weeks after the first infusion, while T-cell populations remain unchanged.50
A minimal infusion reaction such as flushing and chills can be seen with the first infusion. Patients may be more susceptible to certain infections such as reactivation of herpes zoster, but overall rituximab is well tolerated. Rare cases of progressive multifocal leukoencephalopathy have been reported in patients taking it, but none have occurred so far in myasthenia gravis treatment.
Cyclophosphamide
Cyclophosphamide is an alkylating agent that reduces proliferation of both B and T cells. It can be effective in myasthenia gravis, but potentially serious side effects limit its use. It should be reserved for the small percentage of cases that are refractory to other immunotherapies.
Thymectomy
Surgical treatment should be considered for patients with thymoma. If the tumor cannot be surgically resected, chemoradiotherapy can be considered for relief of myasthenic symptoms and for prevention of local invasion.
Thymomas recur in a minority of patients many years after the initial resection, sometimes without myasthenia gravis symptoms. A recurrence of symptoms does not necessarily indicate a recurrence of thymoma. The lack of correlation between myasthenia gravis symptoms and thymoma recurrence highlights the importance of radiologic follow-up in these patients.
For patients without thymoma, many experts believe that thymectomy is beneficial in patients under age 60 who have generalized myasthenia gravis. The likelihood of medication-free remission is about twice as high, and the likelihood of becoming asymptomatic is about one and a half times higher after thymectomy.51 However, it takes up to several years for the benefits of thymectomy to manifest, and thymectomy does not guarantee protection from developing AChR antibody-positive myasthenia gravis in the future.
The optimal timing of thymectomy is not well established; however, the procedure is usually recommended within the first 3 years of diagnosis.52 The response rates from thymectomy are similar for AChR antibody-positive and seronegative patients. In general, thymectomy for MuSK antibody-positive patients has not been effective, and its role in ocular myasthenia gravis is unclear.2,53
Current therapies for myasthenia gravis can help most patients achieve sustained improvement. The overall prognosis has dramatically improved over the last 4 decades: the mortality rate used to be 75%; now it is 4.5%.1
Myasthenia gravis is the most common disorder of neuromuscular junction transmission and is also one of the best characterized autoimmune diseases. However, its symptoms—primarily weakness—vary from patient to patient, and in the same patient, by time of day and over longer time periods. The variation in symptoms can be very confusing to undiagnosed patients and puzzling to unsuspecting physicians. Such diagnostic uncertainty can give the patient additional frustration and emotional stress, which in turn exacerbate his or her condition.
In this review, we will give an overview of the pathogenesis, clinical manifestations, diagnosis, and treatment of myasthenia gravis.
TWO PEAKS IN INCIDENCE BY AGE
The annual incidence of myasthenia gravis is approximately 10 to 20 new cases per million, with a prevalence of about 150 to 200 per million.2
The age of onset has a bimodal distribution, with an early incidence peak in the second to third decade with a female predominance and a late peak in the 6th to the 8th decade with a male predominance.2
Myasthenia gravis is commonly associated with several other autoimmune disorders, including hypothyroidism, hyperthyroidism, systemic lupus erythematosus, rheumatoid arthritis, vitiligo, diabetes, and, more recently recognized, neuromyelitis optica.3
ANTIBODIES AGAINST AChR AND MuSK
In most cases of myasthenia gravis the patient has autoimmune antibodies against constituents of the neuromuscular junction, specifically acetylcholine receptor (AChR) and muscle-specific tyrosine kinase (MuSK) (Figure 1).
AChR antibody-positive myasthenia gravis
When antibodies bind to AChR on the postsynaptic membrane, they cross-link neighboring AChR units, which are absorbed into the muscle fiber and are broken up.4 In addition, the complement system is activated to mediate further damage on the postsynaptic membrane.
AChR antibodies may come from germinal centers of the thymus, where clustered myoid cells express AChR on the plasma membrane surface.5 About 60% of AChR antibody-positive myasthenia gravis patients have an enlarged thymus, and 10% have a thymoma—a tumor of the epithelial cells of this organ. Conversely, about 15% of patients with a thymoma have clinical myasthenia gravis, and an additional 20% possess antibodies against AChR in the serum without myasthenic symptoms.5
MuSK antibody-positive myasthenia gravis
Like AChR, MuSK is a transmembrane component of the postsynaptic neuromuscular junction. During formation of the neuromuscular junction, MuSK is activated through the binding of agrin (a nerve-derived proteoglycan) to lipoprotein-related protein 4 (LRP4), after which complicated intracellular signaling promotes the assembly and stabilization of AChR.6
Unlike AChR antibodies, antibodies against MuSK do not activate the complement system, and complement fixation is not essential for clinical myasthenic symptoms to appear.7 Also, myasthenia gravis with MuSK antibodies is rarely associated with thymoma.8
The precise mechanism by which MuSK antibody impairs transmission at the neuromuscular junction has been a mystery until recently. Animal models, including MuSK-mutant mice and mice injected with MuSK protein or with purified immunoglobulin G from patients with this disease, have revealed a significant reduction of AChR clusters and destruction of neuromuscular junction structures.7,9–12
In addition, MuSK antibodies produce pre-synaptic dysfunction, manifesting as a reduction of acetylcholine content. This information is based on studies in mice and on in vitro electrophysiologic analyses of neuromuscular junctions from a patient with this disease.7,9–13
Finally, MuSK antibodies may indirectly affect the recycling of acetylcholine. After post-synaptic activation, acetylcholine is normally hydrolized by acetylcholinesterase, which is located in the synaptic cleft but anchored to MuSK on the postsynaptic membrane. MuSK antibodies block the binding of MuSK to acetylcholinesterase, possibly leading to less accumulation of acetylcholinesterase.14 This process may explain why patients with MuSK antibody-positive myasthenia gravis tend to respond poorly to acetylcholinesterase inhibitors (more about this below).
Seronegative myasthenia gravis
In a series of 562 consecutive patients with generalized weakness due to myasthenia gravis, 92% were positive for AChR antibody, 3% were positive for MuSK antibody, and 5% were seronegative (possessing neither antibody).15 In contrast, about 50% of patients with purely ocular myasthenia gravis (ie, with isolated weakness of the levator palpebrae superioris, orbicularis oculi, or oculomotor muscles) are seropositive for AChR antibody. Only a few ocular MuSK antibody-positive cases have been described, leaving the rest seronegative. Rarely, both antibodies can be detected in the same patient.16
In patients who are negative for AChR antibodies at the time of disease onset, sero-conversion may occur later during the course. Repeating serologic testing 6 to 12 months later may detect AChR antibodies in approximately 15% of patients who were initially seronegative.15,17
The clinical presentation, electrophysiologic findings, thymic pathologic findings, and treatment responses are similar in AChR antibody-positive and seronegative myasthenia gravis.17 Muscle biopsy study in seronegative cases demonstrates a loss of AChR as well.18
Based on these observations, it has been proposed that seronegative patients may have low-affinity antibodies that can bind to tightly clustered AChRs on the postsynaptic membrane but escape detection by routine radioimmunoassays in a solution phase. With a sensitive cell-based immunofluorescence assay, low-affinity antibodies to clustered AChRs were detected in 66% of patients with generalized myasthenia gravis and in 50% of those with ocular myasthenia gravis who were seronegative on standard assays.19,20 These low-affinity AChR antibodies can also activate complement in vitro, increasing the likelihood that they are pathogenic. However, assays to detect low-affinity AChR antibodies are not yet commercially available.
Within the past year, three research groups independently reported detecting antibodies to LRP4 in 2% to 50% of seronegative myasthenia gravis patients. This wide variation in the prevalence of LRP4 antibodies could be related to patient ethnicity and methods of detection.21–23 LRP4 is a receptor for agrin and is required for agrin-induced MuSK activation and AChR clustering. LRP antibodies can activate complement; therefore, it is plausible that LRP4 antibody binding leads to AChR loss on the postsynaptic membrane. However, additional study is needed to determine if LRP4 antibodies are truly pathogenic in myasthenia gravis.
A DISORDER OF FATIGABLE WEAKNESS
Myasthenia gravis is a disorder of fatigable weakness producing fluctuating symptoms. Symptoms related to the involvement of specific muscle groups are listed in Table 1. Muscle weakness is often worse later in the day or after exercise.
Ocular myasthenia gravis accounts for about 15% of all cases. Of patients initially presenting with ocular symptoms only, twothirds will ultimately develop generalized symptoms, most within the first 2 years.24 No factor has been identified that predicts conversion from an ocular to a generalized form.
Several clinical phenotypes of MuSK antibody-positive myasthenia gravis have been described. An oculobulbar form presents with diplopia, ptosis, dysarthria, and profound atrophy of the muscles of the tongue and face. A restricted myopathic form presents with prominent neck, shoulder, and respiratory weakness without ocular involvement. A third form is a combination of ocular and proximal limb weakness, indistinguishable from AChR antibody-positive disease.25
MuSK antibody-positive patients do not respond as well to acetylcholinesterase inhibitors as AChR antibody-positive patients do. In one study, nearly 70% of MuSK antibody-positive patients demonstrated no response, poor tolerance, or cholinergic hypersensitivity to these agents.25 Fortunately, most MuSK antibody-positive patients have a favorable response to immunosuppressive therapy—sometimes a dramatic improvement after plasmapheresis.8
DIAGNOSIS OF MYASTHENIA GRAVIS
The common differential diagnoses for myasthenia gravis are listed in Table 2.
The essential feature of myasthenia gravis is fluctuating muscle weakness, often with fatigue. Many patients complain of weakness of specific muscle groups after their repeated usage. Pain is generally a less conspicuous symptom, and generalized fatigue without objective weakness is inconsistent with myasthenia gravis.
Signs of muscle weakness may include droopy eyelids, diplopia, inability to hold the head straight, difficulty swallowing or chewing, speech disturbances, difficulty breathing, and difficulty raising the arms or rising from the sitting position. A historical pattern of ptosis alternating from one eye to the other is fairly characteristic of myasthenia gravis.
The weakness of orbicularis oculi is easily identified on examination by prying open the eyes during forced eye closure. Limb weakness is usually more significant in the arms than in the legs. An often-neglected feature of myasthenia gravis is finger extensor weakness with a relative sparing of other distal hand muscles.2
The ice-pack test is performed by placing a small bag of ice over the ptotic eye for 2 to 5 minutes and assessing the degree of ptosis for any noticeable improvement. This test is not very helpful for assessing ocular motor weakness.
The edrophonium (Tensilon) test can be used for patients with ptosis or ophthalmoparesis. Edrophonium, a short-acting acetylcholinesterase inhibitor, is given intravenously while the patient is observed for objective improvement. The patient’s cardiovascular status should be monitored for arrhythmias and hypotension. Atropine should be immediately available in case severe bradycardia develops.
The ice-pack test and the edrophonium test can give false-negative and false-positive results, and the diagnosis of myasthenia gravis must be verified by other diagnostic tests.
Testing for antibodies
Testing for circulating AChR antibodies, MuSK antibodies, or both is the first step in the laboratory confirmation of myasthenia gravis.
There are three AChR antibody subtypes: binding, blocking, and modulating. Binding antibodies are present in 80% to 90% of patients with generalized myasthenia gravis and 50% of those with ocular myasthenia gravis. Testing for blocking and modulating AChR antibodies increases the sensitivity by less than 5% when added to testing for binding antibodies.
AChR antibody titers correlate poorly with disease severity between patients. However, in individual patients, antibody titers tend to go down in parallel with clinical improvement.
MuSK antibody is detected in nearly half of myasthenia gravis patients with generalized weakness who are negative for AChR antibody.
Electrophysiologic tests
Electrophysiologic tests can usually confirm the diagnosis of seronegative myasthenia gravis. They are also helpful in seropositive patients who have unusual clinical features or a poor response to treatment.
Repetitive nerve stimulation studies use a slow rate (2–5 Hz) of repetitive electrical stimulation. The study is positive if the motor response declines by more than 10%. However, a decremental response is not specific for myasthenia gravis, as it may be seen in other neuromuscular disorders such as motor neuron disease or Lambert-Eaton myasthenic syndrome.
This test is technically easier to do in distal muscles than in proximal muscles, but less sensitive. Therefore, proximal muscles such as the trapezius or facial muscles are usually also sampled to maximize the yield. To further maximize the sensitivity, muscles being tested should be warm, and acetylcholinesterase inhibitors should be withheld for 12 hours before.
Repetitive nerve stimulation studies in distal muscles are positive in approximately 75% of patients with generalized myasthenia gravis and in 30% with ocular myasthenia gravis.26
Single-fiber electromyography is more technically demanding than repetitive nerve stimulation and is less widely available. It is usually performed with a special needle electrode that can simultaneously identify action potentials arising from individual muscle fibers innervated by the same axon.
Variability in time of the second action potential relative to the first is called “jitter.” Abnormal jitter is seen in more than 95% of patients with generalized myasthenia gravis and in 85% to 90% of those with ocular myasthenia gravis.26,27 However, abnormal jitter can also be seen in other neuromuscular diseases such as motor neuron disease or in neuromuscular junctional disorders such as Lambert-Eaton myasthenic syndrome.
Imaging studies
Chest computed tomography or magnetic resonance imaging with contrast should be performed in all myasthenia gravis patients to look for a thymoma.
TREATMENT OF MYASTHENIA GRAVIS
Acetylcholinesterase inhibitors
As a reasonable first therapy in mild cases of myasthenia gravis, acetylcholinesterase inhibitors slow down the degradation of acetylcholine and prolong its effect in the neuromuscular junction, but they are not disease-modifying and their benefits are mild.
Pyridostigmine is the usual choice of acetylcholinesterase inhibitor. Its onset of action is rapid (15 to 30 minutes) and its action lasts for 3 to 4 hours. For most patients, the effective dosage range is 60 mg to 90 mg every 4 to 6 hours. A long-acting form is also available and can be given as a single nighttime dose.
Immunomodulating therapy
Patients who have moderate to severe symptoms require some form of immunomodulating therapy.
Plasmapheresis or intravenous immune globulin is reserved for patients with severe or rapidly worsening disease because their beneficial effects can be seen within the first week of treatment.
Longer-acting immunotherapies (corticosteroids, azathioprine, mycophenolate mofetil and others) have a slower onset of responses but provide sustained benefits. Which drug to use depends on factors such as comorbidity, side effects, and cost.
Drugs to avoid
A number of medications can exacerbate weakness in myasthenia gravis and should be avoided or used with caution. The list is long, but ones that deserve the most attention are penicillamine, interferons, procainamide, quinidine, and antibiotics, including quinolones and aminoglycosides. A more comprehensive list of medications that may exacerbate myasthenia gravis symptoms can be found in a review by Keesey.2
RAPID INDUCTION IMMUNOTHERAPIES : PLASMAPHERESIS, IMMUNE GLOBULIN
Both plasmapheresis and intravenous immune globulin act quickly over days, but in most patients their effects last only a few weeks. Both are used as rescue therapies for myasthenic crises, bridging therapy to slow-acting immunotherapeutic agents, or maintenance treatment for poorly controlled cases.
Several retrospective studies have confirmed the efficacy of plasmapheresis in more than 80% of patients with generalized symptoms.28,29
In a randomized trial in patients with generalized therapies, intravenous immune globulin improved muscle strength in the group of patients with severe symptoms.30 The effective dosage of intravenous immune globulin varies from 1 to 2 g/kg without observed difference between doses.31 Trials comparing the efficacy of intravenous immune globulin and plasmapheresis in acute and severe myasthenia gravis did not reveal a difference in efficacy.32,33 Intravenous immune globulin at a minimal dose of 0.4 g/kg every 3 months has been successfully used as a long-term maintenance monotherapy, and such a role could be expanded to more patients with further studies.34
The choice between plasmapheresis and intravenous immune globulin is often based on the ability of a patient to tolerate each treatment and on the availability of the plasmapheresis procedure. Intravenous immune globulin is easier to administer, is associated with fewer adverse events related to vascular access, and is therefore more appropriate than plasmapheresis in some centers.
CHRONIC MAINTENANCE IMMUNOMODULATING TREATMENT
Corticosteroids
Prednisone, the most commonly used agent, leads to remission or marked improvement in 70% to 80% of patients with ocular or generalized myasthenia gravis.35 It may also reduce the progression of ocular myasthenia gravis to the generalized form.36
The effective dose of prednisone depends on the severity and distribution of symptoms. Some patients may need up to 1.0 mg/kg/day (usually 50 to 80 mg per day). In patients with mild to moderate symptoms, a lower maximal dosage such as 20 to 40 mg per day can be sufficient.
Within 1 to 2 weeks after starting high-dose prednisone, up to 50% of patients may develop a transient deterioration, including possible precipitation of a myasthenic crisis.37 For this reason, high-dose prednisone is commonly started only in hospitalized patients who are also receiving plasmapheresis or intravenous immune globulin. Otherwise, an outpatient dose-escalation protocol can be used to achieve a target dose over several weeks.
Prednisone tapering can begin after the patient has been on the maximal dose for 1 to 2 months and significant improvement is evident. A monthly tapering of 5 to 10 mg is preferred, then more slowly after the daily dose reaches 30 mg. The usual maintenance dose averages about 5 mg daily.
Common side effects of prednisone include weight gain, cushingoid features, easy bruising, cataracts, glaucoma, hypertension, diabetes, dyslipidemia, and osteoporosis. Patients are advised to take supplemental calcium (1,500 mg per day) and vitamin D (400 to 800 IU per day). For those most at risk of osteoporosis, treatment with a bisphosphonate should be considered.
Other immunotherapeutic agents are often needed, either to replace the corticosteroid or to permit use of lower doses of it. Because of their delayed onset of action, starting such corticosteroid-sparing agents early in the course is often necessary. These agents are often initially combined with high-dose prednisone, with an eventual goal of weaning off prednisone entirely. This strategy offers the advantage of relatively rapid induction while avoiding the long-term adverse effects of corticosteroid treatment.
Azathioprine
Azathioprine doesn’t begin to show a beneficial effect in myasthenia gravis for 6 to 12 months, and it often reaches its maximal efficacy only after 1 to 2 years of treatment.38
In a study of 78 myasthenia gravis patients, 91% improved when treated with azathioprine alone or together with prednisone.39 In another study using azathioprine and prednisolone for generalized myasthenia gravis, nearly two-thirds of patients came off prednisolone while maintaining remission for 3 years.38
A typical maintenance dose is 2 to 3 mg/kg/day. Common side effects are nausea, vomiting, and malaise. Less frequent side effects include hematologic abnormalities, abnormal liver function, and pancreatitis. Monthly monitoring of complete blood cell counts and liver function tests is warranted for the first 6 months, then less often.
One in 300 people in the general population is homozygous for a mutant allele in the thiopurine methyltransferase (TPMT) gene. Patients with this genotype should not receive azathioprine because of the risk of life-threatening bone marrow suppression.40 A slightly increased risk of various forms of lymphoma has been documented.41
Mycophenolate mofetil
A well-tolerated medication with few side effects, mycophenolate mofetil is being used more in myasthenia gravis. The results of two recent randomized trials suggested that it is not effective in improving myasthenia gravis symptoms or sparing prednisone dosage when used for 90 days or 36 weeks.42,43 However, extensive clinical experience supports its longterm efficacy in myasthenia gravis.
In a retrospective study of 85 patients with generalized myasthenia gravis, mycophenolate at doses of 1 to 3 g daily improved symptoms in 73% and produced remission in 50%. Steroid dosage was reduced in 71% of patients.44
Another retrospective study, with 102 patients, verified a slow development of clinical benefit after months of mycophenolate therapy alone or in combination with prednisone. Approximately 50% of patients achieved a minimal manifestation status after 6 to 12 months of mycophenolate treatment. Eventually, at 24 months of treatment, 80% of patients had a desirable outcome of minimal clinical manifestation or better, 55% of patients were able to come off prednisone entirely, and 75% were taking less than 7.5 mg of prednisone per day.45
Common side effects of mycophenolate include nausea, diarrhea, and infections such as urinary tract infections and herpes reactivation. The complete blood cell count needs to be monitored frequently during the first 6 months of therapy. Leukopenia can occur but rarely necessitates stopping mycophenolate. Long-term safety data are lacking, but so far there has been no clearly increased risk of malignancy.
Mycophenolate exposure in pregnancy results in a high incidence of major fetal malformations. Therefore, its use in pregnant patients is discouraged, and women of child-bearing age should use effective contraception.46
Cyclosporine
A randomized trial in a small number of patients suggested that cyclosporine is fairly effective as monotherapy.47 Its onset of action in myasthenia gravis is faster than that of other corticosteroid-sparing agents, and clinical benefit can often be observed as early as 1 to 2 months. A dose of 5 mg/kg/day and a maintenance serum level of 100 to 150 ng/mL are generally recommended. However, renal, hepatic, and hematologic toxicities and interactions with other medications make cyclosporine a less attractive choice.
Methotrexate
A randomized trial evaluated the utility of methotrexate as a steroid-sparing agent compared with azathioprine.48 At 24 months, its steroid-sparing effect was similar to that of azathioprine, and the prednisone dosage had been reduced in more than 50% of patients.
Another phase II trial studying the efficacy of methotrexate in myasthenia gravis is under way.49
Rituximab
Rituximab is a monoclonal antibody against B-cell membrane marker CD20. A growing number of case series support its efficacy in patients with severe generalized myasthenia gravis refractory to multiple immunosuppressants.16,50 It seems particularly effective for MuSK antibody-positive disease, reducing MuSK antibody titers and having a treatment effect that lasts for years.
The standard dosage is 375 mg/m2 per week for 4 consecutive weeks. Peripheral B cells tend to be depleted within 2 weeks after the first infusion, while T-cell populations remain unchanged.50
A minimal infusion reaction such as flushing and chills can be seen with the first infusion. Patients may be more susceptible to certain infections such as reactivation of herpes zoster, but overall rituximab is well tolerated. Rare cases of progressive multifocal leukoencephalopathy have been reported in patients taking it, but none have occurred so far in myasthenia gravis treatment.
Cyclophosphamide
Cyclophosphamide is an alkylating agent that reduces proliferation of both B and T cells. It can be effective in myasthenia gravis, but potentially serious side effects limit its use. It should be reserved for the small percentage of cases that are refractory to other immunotherapies.
Thymectomy
Surgical treatment should be considered for patients with thymoma. If the tumor cannot be surgically resected, chemoradiotherapy can be considered for relief of myasthenic symptoms and for prevention of local invasion.
Thymomas recur in a minority of patients many years after the initial resection, sometimes without myasthenia gravis symptoms. A recurrence of symptoms does not necessarily indicate a recurrence of thymoma. The lack of correlation between myasthenia gravis symptoms and thymoma recurrence highlights the importance of radiologic follow-up in these patients.
For patients without thymoma, many experts believe that thymectomy is beneficial in patients under age 60 who have generalized myasthenia gravis. The likelihood of medication-free remission is about twice as high, and the likelihood of becoming asymptomatic is about one and a half times higher after thymectomy.51 However, it takes up to several years for the benefits of thymectomy to manifest, and thymectomy does not guarantee protection from developing AChR antibody-positive myasthenia gravis in the future.
The optimal timing of thymectomy is not well established; however, the procedure is usually recommended within the first 3 years of diagnosis.52 The response rates from thymectomy are similar for AChR antibody-positive and seronegative patients. In general, thymectomy for MuSK antibody-positive patients has not been effective, and its role in ocular myasthenia gravis is unclear.2,53
- Alshekhlee A, Miles JD, Katirji B, Preston DC, Kaminski HJ. Incidence and mortality rates of myasthenia gravis and myasthenic crisis in US hospitals. Neurology 2009; 72:1548–1554.
- Keesey JC. Clinical evaluation and management of myasthenia gravis. Muscle Nerve 2004; 29:484–505.
- Leite MI, Coutinho E, Lana-Peixoto M, et al. Myasthenia gravis and neuromyelitis optica spectrum disorder: a multicenter study of 16 patients. Neurology 2012; 78:1601–1607.
- Drachman DB, Angus CW, Adams RN, Michelson JD, Hoffman GJ. Myasthenic antibodies cross-link acetylcholine receptors to accelerate degradation. N Engl J Med 1978; 298:1116–1122.
- Fujii Y. The thymus, thymoma and myasthenia gravis. Surg Today 2013; 43:461–466.
- Evoli A, Lindstrom J. Myasthenia gravis with antibodies to MuSK: another step toward solving mystery? Neurology 2011; 77:1783–1784.
- Mori S, Kubo S, Akiyoshi T, et al. Antibodies against muscle-specific kinase impair both presynaptic and postsynaptic functions in a murine model of myasthenia gravis. Am J Pathol 2012; 180:798–810.
- Guptill JT, Sanders DB, Evoli A. Anti-MuSK antibody myasthenia gravis: clinical findings and response to treatment in two large cohorts. Muscle Nerve 2011; 44:36–40.
- Chevessier F, Girard E, Molgó J, et al. A mouse model for congenital myasthenic syndrome due to MuSK mutations reveals defects in structure and function of neuromuscular junctions. Hum Mol Genet 2008; 17:3577–3595.
- Richman DP, Nishi K, Morell SW, et al. Acute severe animal model of anti-muscle-specific kinase myasthenia: combined postsynaptic and presynaptic changes. Arch Neurol 2012; 69:453–460.
- Klooster R, Plomp JJ, Huijbers MG, et al. Muscle-specific kinase myasthenia gravis IgG4 autoantibodies cause severe neuromuscular junction dysfunction in mice. Brain 2012; 135:1081–1101.
- Viegas S, Jacobson L, Waters P, et al. Passive and active immunization models of MuSK-Ab positive myasthenia: electrophysiological evidence for pre and postsynaptic defects. Exp Neurol 2012; 234:506–512.
- Niks EH, Kuks JB, Wokke JH, et al. Pre- and postsynaptic neuromuscular junction abnormalities in musk myasthenia. Muscle Nerve 2010; 42:283–288.
- Kawakami Y, Ito M, Hirayama M, et al. Anti-MuSK autoantibodies block binding of collagen Q to MuSK. Neurology 2011; 77:1819–1826.
- Chan KH, Lachance DH, Harper CM, Lennon VA. Frequency of seronegativity in adult-acquired generalized myasthenia gravis. Muscle Nerve 2007; 36:651–658.
- Collongues N, Casez O, Lacour A, et al. Rituximab in refractory and non-refractory myasthenia: a retrospective multicenter study. Muscle Nerve 2012; 46:687–691.
- Sanders DB, Andrews PI, Howard JF, Massey JM. Seronegative myasthenia gravis. Neurology 1997; 48(suppl 5):40S–45S.
- Shiraishi H, Motomura M, Yoshimura T, et al. Acetylcholine receptors loss and postsynaptic damage in MuSK antibody-positive myasthenia gravis. Ann Neurol 2005; 57:289–293.
- Leite MI, Jacob S, Viegas S, et al. IgG1 antibodies to acetylcholine receptors in ‘seronegative’ myasthenia gravis. Brain 2008; 131:1940–1952.
- Jacob S, Viegas S, Leite MI, et al. Presence and pathogenic relevance of antibodies to clustered acetylcholine receptor in ocular and generalized myasthenia gravis. Arch Neurol 2012; 69:994–1001.
- Higuchi O, Hamuro J, Motomura M, Yamanashi Y. Autoantibodies to low-density lipoprotein receptor-related protein 4 in myasthenia gravis. Ann Neurol 2011; 69:418–422.
- Pevzner A, Schoser B, Peters K, et al. Anti-LRP4 autoantibodies in AChR- and MuSK-antibody-negative myasthenia gravis. J Neurol 2012; 259:427–435.
- Zhang B, Tzartos JS, Belimezi M, et al. Autoantibodies to lipoprotein-related protein 4 in patients with double-seronegative myasthenia gravis. Arch Neurol 2012; 69:445–451.
- Kupersmith MJ, Latkany R, Homel P. Development of generalized disease at 2 years in patients with ocular myasthenia gravis. Arch Neurol 2003; 60:243–248.
- Pasnoor M, Wolfe GI, Nations S, et al. Clinical findings in MuSK-antibody positive myasthenia gravis: a US experience. Muscle Nerve 2010; 41:370–374.
- Oh SJ, Kim DE, Kuruoglu R, Bradley RJ, Dwyer D. Diagnostic sensitivity of the laboratory tests in myasthenia gravis. Muscle Nerve 1992; 15:720–724.
- Sanders DB, Stålberg EV. AAEM minimonograph #25: single-fiber electromyography. Muscle Nerve 1996; 19:1069–1083.
- Lazo-Langner A, Espinosa-Poblano I, Tirado-Cárdenas N, et al. Therapeutic plasma exchange in Mexico: experience from a single institution. Am J Hematol 2002; 70:16–21.
- Carandina-Maffeis R, Nucci A, Marques JF, et al. Plasmapheresis in the treatment of myasthenia gravis: retrospective study of 26 patients. Arq Neuropsiquiatr 2004; 62:391–395.
- Zinman L, Ng E, Bril V. IV immunoglobulin in patients with myasthenia gravis: a randomized controlled trial. Neurology 2007; 68:837–841.
- Gajdos P, Tranchant C, Clair B, et al; Myasthenia Gravis Clinical Study Group. Treatment of myasthenia gravis exacerbation with intravenous immunoglobulin: a randomized double-blind clinical trial. Arch Neurol 2005; 62:1689–1693.
- Rønager J, Ravnborg M, Hermansen I, Vorstrup S. Immunoglobulin treatment versus plasma exchange in patients with chronic moderate to severe myasthenia gravis. Artif Organs 2001; 25:967–973.
- Barth D, Nabavi Nouri M, Ng E, Nwe P, Bril V. Comparison of IVIg and PLEX in patients with myasthenia gravis. Neurology 2011; 76:2017–2023.
- Wegner B, Ahmed I. Intravenous immunoglobulin monotherapy in long-term treatment of myasthenia gravis. Clin Neurol Neurosurg 2002; 105:3–8.
- Pascuzzi RM, Coslett HB, Johns TR. Long-term corticosteroid treatment of myasthenia gravis: report of 116 patients. Ann Neurol 1984; 15:291–298.
- Monsul NT, Patwa HS, Knorr AM, Lesser RL, Goldstein JM. The effect of prednisone on the progression from ocular to generalized myasthenia gravis. J Neurol Sci 2004; 217:131–133.
- Miller RG, Milner-Brown HS, Mirka A. Prednisone-induced worsening of neuromuscular function in myasthenia gravis. Neurology 1986; 36:729–732.
- Palace J, Newsom-Davis J, Lecky B. A randomized double-blind trial of prednisolone alone or with azathioprine in myasthenia gravis. Myasthenia Gravis Study Group. Neurology 1998; 50:1778–1783.
- Mertens HG, Hertel G, Reuther P, Ricker K. Effect of immunosuppressive drugs (azathioprine). Ann N Y Acad Sci 1981; 377:691–699.
- Relling MV, Gardner EE, Sandborn WJ, et al; Clinical Pharmacogenetics Implementation Consortium. Clinical Pharmacogenetics Implementation Consortium guidelines for thiopurine methyltransferase genotype and thiopurine dosing. Clin Pharmacol Ther 2011; 89:387–391.
- Finelli PF. Primary CNS lymphoma in myasthenic on long-term azathioprine. J Neurooncol 2005; 74:91–92.
- Sanders DB, Hart IK, Mantegazza R, et al. An international, phase III, randomized trial of mycophenolate mofetil in myasthenia gravis. Neurology 2008; 71:400–406.
- Muscle Study Group. A trial of mycophenolate mofetil with prednisone as initial immunotherapy in myasthenia gravis. Neurology 2008; 71:394–399.
- Meriggioli MN, Ciafaloni E, Al-Hayk KA, et al. Mycophenolate mofetil for myasthenia gravis: an analysis of efficacy, safety, and tolerability. Neurology 2003; 61:1438–1440.
- Hehir MK, Burns TM, Alpers J, Conaway MR, Sawa M, Sanders DB. Mycophenolate mofetil in AChR-antibody-positive myasthenia gravis: outcomes in 102 patients. Muscle Nerve 2010; 41:593–598.
- Merlob P, Stahl B, Klinger G. Tetrada of the possible mycophenolate mofetil embryopathy: a review. Reprod Toxicol 2009; 28:105–108.
- Tindall RS, Rollins JA, Phillips JT, Greenlee RG, Wells L, Belendiuk G. Preliminary results of a double-blind, randomized, placebo-controlled trial of cyclosporine in myasthenia gravis. N Engl J Med 1987; 316:719–724.
- Heckmann JM, Rawoot A, Bateman K, Renison R, Badri M. A single-blinded trial of methotrexate versus azathioprine as steroid-sparing agents in generalized myasthenia gravis. BMC Neurol 2011; 11:97.
- Pasnoor M, He J, Herbelin L, Dimachkie M, Barohn RJ; Muscle Study Group. Phase II trial of methotrexate in myasthenia gravis. Ann N Y Acad Sci 2012; 1275:23–28.
- Díaz-Manera J, Martínez-Hernández E, Querol L, et al. Long-lasting treatment effect of rituximab in MuSK myasthenia. Neurology 2012; 78:189–193.
- Gronseth GS, Barohn RJ. Practice parameter: thymectomy for autoimmune myasthenia gravis (an evidence-based review): Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2000; 55:7–15.
- Kumar V, Kaminski HJ. Treatment of myasthenia gravis. Curr Neurol Neurosci Rep 2011; 11:89–96.
- Pompeo E, Tacconi F, Massa R, Mineo D, Nahmias S, Mineo TC. Long-term outcome of thoracoscopic extended thymectomy for nonthymomatous myasthenia gravis. Eur J Cardiothorac Surg 2009; 36:164–169.
- Alshekhlee A, Miles JD, Katirji B, Preston DC, Kaminski HJ. Incidence and mortality rates of myasthenia gravis and myasthenic crisis in US hospitals. Neurology 2009; 72:1548–1554.
- Keesey JC. Clinical evaluation and management of myasthenia gravis. Muscle Nerve 2004; 29:484–505.
- Leite MI, Coutinho E, Lana-Peixoto M, et al. Myasthenia gravis and neuromyelitis optica spectrum disorder: a multicenter study of 16 patients. Neurology 2012; 78:1601–1607.
- Drachman DB, Angus CW, Adams RN, Michelson JD, Hoffman GJ. Myasthenic antibodies cross-link acetylcholine receptors to accelerate degradation. N Engl J Med 1978; 298:1116–1122.
- Fujii Y. The thymus, thymoma and myasthenia gravis. Surg Today 2013; 43:461–466.
- Evoli A, Lindstrom J. Myasthenia gravis with antibodies to MuSK: another step toward solving mystery? Neurology 2011; 77:1783–1784.
- Mori S, Kubo S, Akiyoshi T, et al. Antibodies against muscle-specific kinase impair both presynaptic and postsynaptic functions in a murine model of myasthenia gravis. Am J Pathol 2012; 180:798–810.
- Guptill JT, Sanders DB, Evoli A. Anti-MuSK antibody myasthenia gravis: clinical findings and response to treatment in two large cohorts. Muscle Nerve 2011; 44:36–40.
- Chevessier F, Girard E, Molgó J, et al. A mouse model for congenital myasthenic syndrome due to MuSK mutations reveals defects in structure and function of neuromuscular junctions. Hum Mol Genet 2008; 17:3577–3595.
- Richman DP, Nishi K, Morell SW, et al. Acute severe animal model of anti-muscle-specific kinase myasthenia: combined postsynaptic and presynaptic changes. Arch Neurol 2012; 69:453–460.
- Klooster R, Plomp JJ, Huijbers MG, et al. Muscle-specific kinase myasthenia gravis IgG4 autoantibodies cause severe neuromuscular junction dysfunction in mice. Brain 2012; 135:1081–1101.
- Viegas S, Jacobson L, Waters P, et al. Passive and active immunization models of MuSK-Ab positive myasthenia: electrophysiological evidence for pre and postsynaptic defects. Exp Neurol 2012; 234:506–512.
- Niks EH, Kuks JB, Wokke JH, et al. Pre- and postsynaptic neuromuscular junction abnormalities in musk myasthenia. Muscle Nerve 2010; 42:283–288.
- Kawakami Y, Ito M, Hirayama M, et al. Anti-MuSK autoantibodies block binding of collagen Q to MuSK. Neurology 2011; 77:1819–1826.
- Chan KH, Lachance DH, Harper CM, Lennon VA. Frequency of seronegativity in adult-acquired generalized myasthenia gravis. Muscle Nerve 2007; 36:651–658.
- Collongues N, Casez O, Lacour A, et al. Rituximab in refractory and non-refractory myasthenia: a retrospective multicenter study. Muscle Nerve 2012; 46:687–691.
- Sanders DB, Andrews PI, Howard JF, Massey JM. Seronegative myasthenia gravis. Neurology 1997; 48(suppl 5):40S–45S.
- Shiraishi H, Motomura M, Yoshimura T, et al. Acetylcholine receptors loss and postsynaptic damage in MuSK antibody-positive myasthenia gravis. Ann Neurol 2005; 57:289–293.
- Leite MI, Jacob S, Viegas S, et al. IgG1 antibodies to acetylcholine receptors in ‘seronegative’ myasthenia gravis. Brain 2008; 131:1940–1952.
- Jacob S, Viegas S, Leite MI, et al. Presence and pathogenic relevance of antibodies to clustered acetylcholine receptor in ocular and generalized myasthenia gravis. Arch Neurol 2012; 69:994–1001.
- Higuchi O, Hamuro J, Motomura M, Yamanashi Y. Autoantibodies to low-density lipoprotein receptor-related protein 4 in myasthenia gravis. Ann Neurol 2011; 69:418–422.
- Pevzner A, Schoser B, Peters K, et al. Anti-LRP4 autoantibodies in AChR- and MuSK-antibody-negative myasthenia gravis. J Neurol 2012; 259:427–435.
- Zhang B, Tzartos JS, Belimezi M, et al. Autoantibodies to lipoprotein-related protein 4 in patients with double-seronegative myasthenia gravis. Arch Neurol 2012; 69:445–451.
- Kupersmith MJ, Latkany R, Homel P. Development of generalized disease at 2 years in patients with ocular myasthenia gravis. Arch Neurol 2003; 60:243–248.
- Pasnoor M, Wolfe GI, Nations S, et al. Clinical findings in MuSK-antibody positive myasthenia gravis: a US experience. Muscle Nerve 2010; 41:370–374.
- Oh SJ, Kim DE, Kuruoglu R, Bradley RJ, Dwyer D. Diagnostic sensitivity of the laboratory tests in myasthenia gravis. Muscle Nerve 1992; 15:720–724.
- Sanders DB, Stålberg EV. AAEM minimonograph #25: single-fiber electromyography. Muscle Nerve 1996; 19:1069–1083.
- Lazo-Langner A, Espinosa-Poblano I, Tirado-Cárdenas N, et al. Therapeutic plasma exchange in Mexico: experience from a single institution. Am J Hematol 2002; 70:16–21.
- Carandina-Maffeis R, Nucci A, Marques JF, et al. Plasmapheresis in the treatment of myasthenia gravis: retrospective study of 26 patients. Arq Neuropsiquiatr 2004; 62:391–395.
- Zinman L, Ng E, Bril V. IV immunoglobulin in patients with myasthenia gravis: a randomized controlled trial. Neurology 2007; 68:837–841.
- Gajdos P, Tranchant C, Clair B, et al; Myasthenia Gravis Clinical Study Group. Treatment of myasthenia gravis exacerbation with intravenous immunoglobulin: a randomized double-blind clinical trial. Arch Neurol 2005; 62:1689–1693.
- Rønager J, Ravnborg M, Hermansen I, Vorstrup S. Immunoglobulin treatment versus plasma exchange in patients with chronic moderate to severe myasthenia gravis. Artif Organs 2001; 25:967–973.
- Barth D, Nabavi Nouri M, Ng E, Nwe P, Bril V. Comparison of IVIg and PLEX in patients with myasthenia gravis. Neurology 2011; 76:2017–2023.
- Wegner B, Ahmed I. Intravenous immunoglobulin monotherapy in long-term treatment of myasthenia gravis. Clin Neurol Neurosurg 2002; 105:3–8.
- Pascuzzi RM, Coslett HB, Johns TR. Long-term corticosteroid treatment of myasthenia gravis: report of 116 patients. Ann Neurol 1984; 15:291–298.
- Monsul NT, Patwa HS, Knorr AM, Lesser RL, Goldstein JM. The effect of prednisone on the progression from ocular to generalized myasthenia gravis. J Neurol Sci 2004; 217:131–133.
- Miller RG, Milner-Brown HS, Mirka A. Prednisone-induced worsening of neuromuscular function in myasthenia gravis. Neurology 1986; 36:729–732.
- Palace J, Newsom-Davis J, Lecky B. A randomized double-blind trial of prednisolone alone or with azathioprine in myasthenia gravis. Myasthenia Gravis Study Group. Neurology 1998; 50:1778–1783.
- Mertens HG, Hertel G, Reuther P, Ricker K. Effect of immunosuppressive drugs (azathioprine). Ann N Y Acad Sci 1981; 377:691–699.
- Relling MV, Gardner EE, Sandborn WJ, et al; Clinical Pharmacogenetics Implementation Consortium. Clinical Pharmacogenetics Implementation Consortium guidelines for thiopurine methyltransferase genotype and thiopurine dosing. Clin Pharmacol Ther 2011; 89:387–391.
- Finelli PF. Primary CNS lymphoma in myasthenic on long-term azathioprine. J Neurooncol 2005; 74:91–92.
- Sanders DB, Hart IK, Mantegazza R, et al. An international, phase III, randomized trial of mycophenolate mofetil in myasthenia gravis. Neurology 2008; 71:400–406.
- Muscle Study Group. A trial of mycophenolate mofetil with prednisone as initial immunotherapy in myasthenia gravis. Neurology 2008; 71:394–399.
- Meriggioli MN, Ciafaloni E, Al-Hayk KA, et al. Mycophenolate mofetil for myasthenia gravis: an analysis of efficacy, safety, and tolerability. Neurology 2003; 61:1438–1440.
- Hehir MK, Burns TM, Alpers J, Conaway MR, Sawa M, Sanders DB. Mycophenolate mofetil in AChR-antibody-positive myasthenia gravis: outcomes in 102 patients. Muscle Nerve 2010; 41:593–598.
- Merlob P, Stahl B, Klinger G. Tetrada of the possible mycophenolate mofetil embryopathy: a review. Reprod Toxicol 2009; 28:105–108.
- Tindall RS, Rollins JA, Phillips JT, Greenlee RG, Wells L, Belendiuk G. Preliminary results of a double-blind, randomized, placebo-controlled trial of cyclosporine in myasthenia gravis. N Engl J Med 1987; 316:719–724.
- Heckmann JM, Rawoot A, Bateman K, Renison R, Badri M. A single-blinded trial of methotrexate versus azathioprine as steroid-sparing agents in generalized myasthenia gravis. BMC Neurol 2011; 11:97.
- Pasnoor M, He J, Herbelin L, Dimachkie M, Barohn RJ; Muscle Study Group. Phase II trial of methotrexate in myasthenia gravis. Ann N Y Acad Sci 2012; 1275:23–28.
- Díaz-Manera J, Martínez-Hernández E, Querol L, et al. Long-lasting treatment effect of rituximab in MuSK myasthenia. Neurology 2012; 78:189–193.
- Gronseth GS, Barohn RJ. Practice parameter: thymectomy for autoimmune myasthenia gravis (an evidence-based review): Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2000; 55:7–15.
- Kumar V, Kaminski HJ. Treatment of myasthenia gravis. Curr Neurol Neurosci Rep 2011; 11:89–96.
- Pompeo E, Tacconi F, Massa R, Mineo D, Nahmias S, Mineo TC. Long-term outcome of thoracoscopic extended thymectomy for nonthymomatous myasthenia gravis. Eur J Cardiothorac Surg 2009; 36:164–169.
KEY POINTS
- In most cases of myasthenia gravis, the patient has antibodies against acetylcholine receptor (AChR) or musclespecific tyrosine kinase (MuSK).
- Myasthenia gravis is diagnosed by clinical signs, bedside tests (the ice-pack test or the edrophonium test), serologic tests for AChR antibodies or MuSK antibodies, and electrophysiologic tests.
- Acetylcholinesterase inhibitors are the first-step therapy, but patients who have moderate to severe symptoms require some form of immunomodulating therapy.
- A number of drugs can exacerbate weakness in myasthenia gravis and should be avoided or used with caution. These include penicillamine, interferons, procainamide, quinidine, and antibiotics such as quinolones and aminoglycosides.
A 66-year-old woman with an enlarged tongue
A 66-year-old woman was prompted by her dentist to seek medical attention for an unusually enlarged, smooth-appearing tongue (Figure 1). She also complained of fatigue, dyspnea on exertion, and tingling of her hands.
Basic laboratory tests showed normocytic anemia and renal insufficiency. Her thyrotropin level was within normal limits. Serum protein electrophoresis showed a monoclonal M-spike, which prompted a bone marrow biopsy that was diagnostic of multiple myeloma.
Transthoracic echocardiography revealed diffuse hypokinesis with a restrictive filling pattern, myocardial thickening, and moderate mitral and tricuspid regurgitation, highly suggestive of an infiltrative cardiomyopathy. Biopsy of the right ventricle confirmed cardiac amyloidosis of the amyloid immunoglobulin light chain (AL) subtype.
The patient underwent chemotherapy, followed by autologous stem-cell transplantation. She achieved successful remission, and her cardiomyopathy was compensated.
AMYLOIDOSIS IS HETEROGENEOUS
Amyloidosis is a heterogeneous syndrome characterized by abnormal folding of proteins that deposit as insoluble fibrils in different tissues, impairing both structure and function. Virchow was the first to describe amyloid (from amylon, Greek for starch) as an abnormal material seen in postmortem examination of the liver. On Congo red staining, the extracellular proteins appear as salmon-red conglomerates, which also show apple-green birefringence under polarized light.
Amyloidosis can be localized but more often represents a systemic process, often associated with a plasma cell dyscrasia such as multiple myeloma.
Modern classification is based on the precursor protein,1 eg:
- Light chains (AL)
- Acute-phase protein (AA)
- Beta-2-microglobulin (Aß2M)
- Transthyretin (ATTR; occurring in senile systemic amyloidosis)
- Other proteins (occurring in various forms of hereditary systemic amyloidosis).
AMYLOIDOSIS AND THE TONGUE
Macroglossia is defined as protrusion of the tongue beyond the alveolar ridge of the teeth at rest. When caused by amyloidosis, it is most often associated with the systemic AL variant and is present in 10% to 23% of patients with this subtype.2
On physical examination, tongue enlargement can present with lateral indentations, with a smooth contour or with nodular deposits. Less often, bullous lesions, vesicles, and ulcers can also be seen, particularly on the lips. Infiltration of salivary glands can result in xerostomia. Functional symptoms, such as hypogeusia, dysarthria, dysphagia, dysphonia, and, in advanced cases, upper-airway dysfunction can result from restricted mobility of the tongue and tethering to deeper structures.
Surgical management may be necessary if severe obstructive symptoms are present, but infiltrative lesions tend to recur.
AMYLOIDOSIS AND THE HEART
Cardiac involvement in amyloidosis is currently the primary determinant of prognosis.3 It is more often seen in the AL, senile, and hereditary forms. It usually manifests as diastolic heart failure, but angina, orthostatic hypotension, dysrhythmias, and syncope can also occur. Systolic dysfunction is typically a late finding in the course of the disease.
Although an electrocardiographic pattern of low voltage in the precordial and limb leads has been classically associated with cardiac amyloidosis, only 30% of patients with the senile and hereditary forms show this feature.4 Left ventricular hypertrophy on electrocardiography is thought to be uncommon, but it has been reported in 16% of patients with AL amyloidosis and biopsy-proven cardiac involvement.5 Cardiac troponin levels can be elevated, as seen in other infiltrative cardiomyopathies.3 The diagnosis can be established by endomyocardial biopsy or indirectly by cardiac imaging and a positive extracardiac biopsy.
Drug therapy is supportive and mainly involves diuretics, since angiotensin-converting enzyme inhibitors, beta-blockers, and calcium channel blockers may cause hypotension and exacerbate myocardial dysfunction. The specific treatment varies depending on the underlying cause of amyloidosis.
- Westermark P, Benson MD, Buxbaum JN, et al. A primer of amyloid nomenclature. Amyloid 2007; 14:179–183.
- Kyle RA, Gertz MA. Primary systemic amyloidosis: clinical and laboratory features in 474 cases. Semin Hematol 1995; 32:45–59.
- Kapoor P, Thenappan T, Singh E, Kumar S, Greipp PR. Cardiac amyloidosis: a practical approach to diagnosis and management. Am J Med 2011; 124:1006–1015.
- Ng B, Connors LH, Davidoff R, Skinner M, Falk RH. Senile systemic amyloidosis presenting with heart failure: a comparison with light chain-associated amyloidosis. Arch Intern Med 2005; 165:1425–1429.
- Murtagh B, Hammill SC, Gertz MA, Kyle RA, Tajik AJ, Grogan M. Electrocardiographic findings in primary systemic amyloidosis and biopsy-proven cardiac involvement. Am J Cardiol 2005; 95:535–537.
A 66-year-old woman was prompted by her dentist to seek medical attention for an unusually enlarged, smooth-appearing tongue (Figure 1). She also complained of fatigue, dyspnea on exertion, and tingling of her hands.
Basic laboratory tests showed normocytic anemia and renal insufficiency. Her thyrotropin level was within normal limits. Serum protein electrophoresis showed a monoclonal M-spike, which prompted a bone marrow biopsy that was diagnostic of multiple myeloma.
Transthoracic echocardiography revealed diffuse hypokinesis with a restrictive filling pattern, myocardial thickening, and moderate mitral and tricuspid regurgitation, highly suggestive of an infiltrative cardiomyopathy. Biopsy of the right ventricle confirmed cardiac amyloidosis of the amyloid immunoglobulin light chain (AL) subtype.
The patient underwent chemotherapy, followed by autologous stem-cell transplantation. She achieved successful remission, and her cardiomyopathy was compensated.
AMYLOIDOSIS IS HETEROGENEOUS
Amyloidosis is a heterogeneous syndrome characterized by abnormal folding of proteins that deposit as insoluble fibrils in different tissues, impairing both structure and function. Virchow was the first to describe amyloid (from amylon, Greek for starch) as an abnormal material seen in postmortem examination of the liver. On Congo red staining, the extracellular proteins appear as salmon-red conglomerates, which also show apple-green birefringence under polarized light.
Amyloidosis can be localized but more often represents a systemic process, often associated with a plasma cell dyscrasia such as multiple myeloma.
Modern classification is based on the precursor protein,1 eg:
- Light chains (AL)
- Acute-phase protein (AA)
- Beta-2-microglobulin (Aß2M)
- Transthyretin (ATTR; occurring in senile systemic amyloidosis)
- Other proteins (occurring in various forms of hereditary systemic amyloidosis).
AMYLOIDOSIS AND THE TONGUE
Macroglossia is defined as protrusion of the tongue beyond the alveolar ridge of the teeth at rest. When caused by amyloidosis, it is most often associated with the systemic AL variant and is present in 10% to 23% of patients with this subtype.2
On physical examination, tongue enlargement can present with lateral indentations, with a smooth contour or with nodular deposits. Less often, bullous lesions, vesicles, and ulcers can also be seen, particularly on the lips. Infiltration of salivary glands can result in xerostomia. Functional symptoms, such as hypogeusia, dysarthria, dysphagia, dysphonia, and, in advanced cases, upper-airway dysfunction can result from restricted mobility of the tongue and tethering to deeper structures.
Surgical management may be necessary if severe obstructive symptoms are present, but infiltrative lesions tend to recur.
AMYLOIDOSIS AND THE HEART
Cardiac involvement in amyloidosis is currently the primary determinant of prognosis.3 It is more often seen in the AL, senile, and hereditary forms. It usually manifests as diastolic heart failure, but angina, orthostatic hypotension, dysrhythmias, and syncope can also occur. Systolic dysfunction is typically a late finding in the course of the disease.
Although an electrocardiographic pattern of low voltage in the precordial and limb leads has been classically associated with cardiac amyloidosis, only 30% of patients with the senile and hereditary forms show this feature.4 Left ventricular hypertrophy on electrocardiography is thought to be uncommon, but it has been reported in 16% of patients with AL amyloidosis and biopsy-proven cardiac involvement.5 Cardiac troponin levels can be elevated, as seen in other infiltrative cardiomyopathies.3 The diagnosis can be established by endomyocardial biopsy or indirectly by cardiac imaging and a positive extracardiac biopsy.
Drug therapy is supportive and mainly involves diuretics, since angiotensin-converting enzyme inhibitors, beta-blockers, and calcium channel blockers may cause hypotension and exacerbate myocardial dysfunction. The specific treatment varies depending on the underlying cause of amyloidosis.
A 66-year-old woman was prompted by her dentist to seek medical attention for an unusually enlarged, smooth-appearing tongue (Figure 1). She also complained of fatigue, dyspnea on exertion, and tingling of her hands.
Basic laboratory tests showed normocytic anemia and renal insufficiency. Her thyrotropin level was within normal limits. Serum protein electrophoresis showed a monoclonal M-spike, which prompted a bone marrow biopsy that was diagnostic of multiple myeloma.
Transthoracic echocardiography revealed diffuse hypokinesis with a restrictive filling pattern, myocardial thickening, and moderate mitral and tricuspid regurgitation, highly suggestive of an infiltrative cardiomyopathy. Biopsy of the right ventricle confirmed cardiac amyloidosis of the amyloid immunoglobulin light chain (AL) subtype.
The patient underwent chemotherapy, followed by autologous stem-cell transplantation. She achieved successful remission, and her cardiomyopathy was compensated.
AMYLOIDOSIS IS HETEROGENEOUS
Amyloidosis is a heterogeneous syndrome characterized by abnormal folding of proteins that deposit as insoluble fibrils in different tissues, impairing both structure and function. Virchow was the first to describe amyloid (from amylon, Greek for starch) as an abnormal material seen in postmortem examination of the liver. On Congo red staining, the extracellular proteins appear as salmon-red conglomerates, which also show apple-green birefringence under polarized light.
Amyloidosis can be localized but more often represents a systemic process, often associated with a plasma cell dyscrasia such as multiple myeloma.
Modern classification is based on the precursor protein,1 eg:
- Light chains (AL)
- Acute-phase protein (AA)
- Beta-2-microglobulin (Aß2M)
- Transthyretin (ATTR; occurring in senile systemic amyloidosis)
- Other proteins (occurring in various forms of hereditary systemic amyloidosis).
AMYLOIDOSIS AND THE TONGUE
Macroglossia is defined as protrusion of the tongue beyond the alveolar ridge of the teeth at rest. When caused by amyloidosis, it is most often associated with the systemic AL variant and is present in 10% to 23% of patients with this subtype.2
On physical examination, tongue enlargement can present with lateral indentations, with a smooth contour or with nodular deposits. Less often, bullous lesions, vesicles, and ulcers can also be seen, particularly on the lips. Infiltration of salivary glands can result in xerostomia. Functional symptoms, such as hypogeusia, dysarthria, dysphagia, dysphonia, and, in advanced cases, upper-airway dysfunction can result from restricted mobility of the tongue and tethering to deeper structures.
Surgical management may be necessary if severe obstructive symptoms are present, but infiltrative lesions tend to recur.
AMYLOIDOSIS AND THE HEART
Cardiac involvement in amyloidosis is currently the primary determinant of prognosis.3 It is more often seen in the AL, senile, and hereditary forms. It usually manifests as diastolic heart failure, but angina, orthostatic hypotension, dysrhythmias, and syncope can also occur. Systolic dysfunction is typically a late finding in the course of the disease.
Although an electrocardiographic pattern of low voltage in the precordial and limb leads has been classically associated with cardiac amyloidosis, only 30% of patients with the senile and hereditary forms show this feature.4 Left ventricular hypertrophy on electrocardiography is thought to be uncommon, but it has been reported in 16% of patients with AL amyloidosis and biopsy-proven cardiac involvement.5 Cardiac troponin levels can be elevated, as seen in other infiltrative cardiomyopathies.3 The diagnosis can be established by endomyocardial biopsy or indirectly by cardiac imaging and a positive extracardiac biopsy.
Drug therapy is supportive and mainly involves diuretics, since angiotensin-converting enzyme inhibitors, beta-blockers, and calcium channel blockers may cause hypotension and exacerbate myocardial dysfunction. The specific treatment varies depending on the underlying cause of amyloidosis.
- Westermark P, Benson MD, Buxbaum JN, et al. A primer of amyloid nomenclature. Amyloid 2007; 14:179–183.
- Kyle RA, Gertz MA. Primary systemic amyloidosis: clinical and laboratory features in 474 cases. Semin Hematol 1995; 32:45–59.
- Kapoor P, Thenappan T, Singh E, Kumar S, Greipp PR. Cardiac amyloidosis: a practical approach to diagnosis and management. Am J Med 2011; 124:1006–1015.
- Ng B, Connors LH, Davidoff R, Skinner M, Falk RH. Senile systemic amyloidosis presenting with heart failure: a comparison with light chain-associated amyloidosis. Arch Intern Med 2005; 165:1425–1429.
- Murtagh B, Hammill SC, Gertz MA, Kyle RA, Tajik AJ, Grogan M. Electrocardiographic findings in primary systemic amyloidosis and biopsy-proven cardiac involvement. Am J Cardiol 2005; 95:535–537.
- Westermark P, Benson MD, Buxbaum JN, et al. A primer of amyloid nomenclature. Amyloid 2007; 14:179–183.
- Kyle RA, Gertz MA. Primary systemic amyloidosis: clinical and laboratory features in 474 cases. Semin Hematol 1995; 32:45–59.
- Kapoor P, Thenappan T, Singh E, Kumar S, Greipp PR. Cardiac amyloidosis: a practical approach to diagnosis and management. Am J Med 2011; 124:1006–1015.
- Ng B, Connors LH, Davidoff R, Skinner M, Falk RH. Senile systemic amyloidosis presenting with heart failure: a comparison with light chain-associated amyloidosis. Arch Intern Med 2005; 165:1425–1429.
- Murtagh B, Hammill SC, Gertz MA, Kyle RA, Tajik AJ, Grogan M. Electrocardiographic findings in primary systemic amyloidosis and biopsy-proven cardiac involvement. Am J Cardiol 2005; 95:535–537.
Dyspnea after treatment of recurrent urinary tract infection
A 71-year-old woman came to the hospital because of generalized weakness, fatigue, and exertional dyspnea.
She had a history of anemia, recurrent urinary tract infections, and hyperactive bladder. She had been taking nitrofurantoin for a urinary tract infection and phenazopyridine for dysuria, and she noticed that her urine was dark-colored.
She was of northern European descent. She was unaware of any family history of blood-related disorders. She had been admitted to the hospital 6 weeks earlier for symptomatic anemia after taking nitrofurantoin for a urinary tract infection. At that time, she received 2 units of packed red blood cells and then was discharged. Follow-up blood work done 2 weeks later—including a glucose-6 phosphate dehydrogenase (G6PD) assay—was normal.
On physical examination, she was pale and weak. Her hemoglobin level was 5.5 g/dL (reference range 14.0–17.5), with normal white blood cell and platelet counts and an elevated reticulocyte count. A comprehensive metabolic panel showed elevated indirect bilirubin and lactate dehydrogenase levels. A direct Coombs test for autoimmune hemolytic anemia was negative, as was a haptoglobin assay to look for intravascular hemolytic anemia. G6PD levels were normal, yet a peripheral blood smear (Figure 1) showed features of G6PD deficiency.
What was the cause of her anemia?
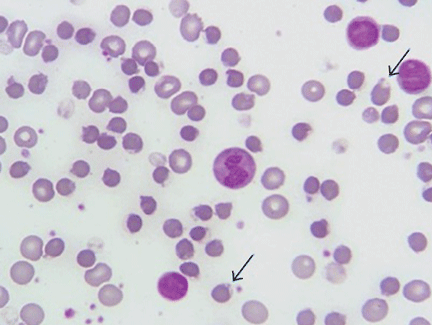
GLUCOSE-6-PHOSPHATE DEHYDROGENASE DEFICIENCY
G6PD deficiency is an X-linked disorder1 that can present as hemolytic anemia. Symptoms of hemolysis can range from mild to severe on exposure to an inciting agent. Men are more commonly affected than women, and affected women are mostly heterozygous. The severity of hemolysis in heterozygous women depends on inactivation of the unaffected X chromosome in some cells.
When exposed to oxidizing agents, people with G6PD deficiency do not have enough nicotinamide adenine dinucleotide phosphate to protect red blood cells.2 This leads to oxidative denaturation of hemoglobin, formation of methemoglobin, and denaturation of globulin. These products are insoluble; they collect in red blood cells and are called Heinz bodies.3 When red blood cells containing Heinz bodies pass through the liver and spleen, the insoluble masses are taken up by macrophages, causing hemolysis and the formation of “bite cells”4 (so named because macrophages “bite” the Heinz bodies out of the red blood cells).
Patients with G6PD deficiency have all the clinical features of hemolytic anemia. On laboratory testing, the Coombs test is negative, the G6PD level is low, and the peripheral smear shows bite cells. The G6PD level is falsely normal or elevated during acute hemolysis because red blood cells deficient in G6PD are removed from circulation and replaced by young red blood cells. The G6PD level is also elevated after blood transfusion. Thus, the G6PD level should be tested 3 months after an acute event.
Hemolysis in G6PD is usually intermittent and self-limited. No treatment is needed except for avoidance of triggers and transfusion for symptomatic anemia. Of note, triggers include some of the drugs commonly used for urinary tract infections (sulfa drugs, nitrofurantoin, phenazopyridine) and antimalarials. Fava beans are also known to cause hemolytic crisis. A complete list of things to avoid can be found at www.g6pd.org/en/G6PDDeficiency/SafeUnsafe/DaEvitare_ISS-it.
There is no commercially available genetic testing kit for G6PD deficiency. Mutation analysis and G6PD gene sequencing are possible but are neither routinely done nor widely available.
BACK TO OUR PATIENT
Our patient’s hemolytic anemia was most likely drug-induced, secondary to a relative deficiency of G6PD. She had been taking nitrofurantoin and phenazopyridine; both of these are oxidizing agents and are known to cause acute hemolytic anemia in people with G6PD deficiency. The G6PD level can be normal after a recent blood transfusion and, as in our patient, during an acute episode of hemolysis.
Because of the strong suspicion of G6PD deficiency, both drugs were stopped when the patient was discharged from the hospital. She did not take either drug for 3 months. Her G6PD level was then retested and was found to be low, confirming the diagnosis. The patient was then advised not to take those drugs again. Since then, her hemoglobin level has remained stable and she has not needed any more blood transfusions.
- Mason PJ, Bautista JM, Gilsanz F. G6PD deficiency: the genotype-phenotype association. Blood Rev 2007; 21:267–283.
- Arese P, De Flora A. Pathophysiology of hemolysis in glucose-6-phosphate dehydrogenase deficiency. Semin Hematol 1990; 27:1–40.
- Jacob HS. Mechanisms of Heinz body formation and attachment to red cell membrane. Semin Hematol 1970; 7:341–354.
- Rifkind RA. Heinz body anemia: an ultrastructural study. II. Red cell sequestration and destruction. Blood 1965; 26:433–448.
A 71-year-old woman came to the hospital because of generalized weakness, fatigue, and exertional dyspnea.
She had a history of anemia, recurrent urinary tract infections, and hyperactive bladder. She had been taking nitrofurantoin for a urinary tract infection and phenazopyridine for dysuria, and she noticed that her urine was dark-colored.
She was of northern European descent. She was unaware of any family history of blood-related disorders. She had been admitted to the hospital 6 weeks earlier for symptomatic anemia after taking nitrofurantoin for a urinary tract infection. At that time, she received 2 units of packed red blood cells and then was discharged. Follow-up blood work done 2 weeks later—including a glucose-6 phosphate dehydrogenase (G6PD) assay—was normal.
On physical examination, she was pale and weak. Her hemoglobin level was 5.5 g/dL (reference range 14.0–17.5), with normal white blood cell and platelet counts and an elevated reticulocyte count. A comprehensive metabolic panel showed elevated indirect bilirubin and lactate dehydrogenase levels. A direct Coombs test for autoimmune hemolytic anemia was negative, as was a haptoglobin assay to look for intravascular hemolytic anemia. G6PD levels were normal, yet a peripheral blood smear (Figure 1) showed features of G6PD deficiency.
What was the cause of her anemia?

GLUCOSE-6-PHOSPHATE DEHYDROGENASE DEFICIENCY
G6PD deficiency is an X-linked disorder1 that can present as hemolytic anemia. Symptoms of hemolysis can range from mild to severe on exposure to an inciting agent. Men are more commonly affected than women, and affected women are mostly heterozygous. The severity of hemolysis in heterozygous women depends on inactivation of the unaffected X chromosome in some cells.
When exposed to oxidizing agents, people with G6PD deficiency do not have enough nicotinamide adenine dinucleotide phosphate to protect red blood cells.2 This leads to oxidative denaturation of hemoglobin, formation of methemoglobin, and denaturation of globulin. These products are insoluble; they collect in red blood cells and are called Heinz bodies.3 When red blood cells containing Heinz bodies pass through the liver and spleen, the insoluble masses are taken up by macrophages, causing hemolysis and the formation of “bite cells”4 (so named because macrophages “bite” the Heinz bodies out of the red blood cells).
Patients with G6PD deficiency have all the clinical features of hemolytic anemia. On laboratory testing, the Coombs test is negative, the G6PD level is low, and the peripheral smear shows bite cells. The G6PD level is falsely normal or elevated during acute hemolysis because red blood cells deficient in G6PD are removed from circulation and replaced by young red blood cells. The G6PD level is also elevated after blood transfusion. Thus, the G6PD level should be tested 3 months after an acute event.
Hemolysis in G6PD is usually intermittent and self-limited. No treatment is needed except for avoidance of triggers and transfusion for symptomatic anemia. Of note, triggers include some of the drugs commonly used for urinary tract infections (sulfa drugs, nitrofurantoin, phenazopyridine) and antimalarials. Fava beans are also known to cause hemolytic crisis. A complete list of things to avoid can be found at www.g6pd.org/en/G6PDDeficiency/SafeUnsafe/DaEvitare_ISS-it.
There is no commercially available genetic testing kit for G6PD deficiency. Mutation analysis and G6PD gene sequencing are possible but are neither routinely done nor widely available.
BACK TO OUR PATIENT
Our patient’s hemolytic anemia was most likely drug-induced, secondary to a relative deficiency of G6PD. She had been taking nitrofurantoin and phenazopyridine; both of these are oxidizing agents and are known to cause acute hemolytic anemia in people with G6PD deficiency. The G6PD level can be normal after a recent blood transfusion and, as in our patient, during an acute episode of hemolysis.
Because of the strong suspicion of G6PD deficiency, both drugs were stopped when the patient was discharged from the hospital. She did not take either drug for 3 months. Her G6PD level was then retested and was found to be low, confirming the diagnosis. The patient was then advised not to take those drugs again. Since then, her hemoglobin level has remained stable and she has not needed any more blood transfusions.
A 71-year-old woman came to the hospital because of generalized weakness, fatigue, and exertional dyspnea.
She had a history of anemia, recurrent urinary tract infections, and hyperactive bladder. She had been taking nitrofurantoin for a urinary tract infection and phenazopyridine for dysuria, and she noticed that her urine was dark-colored.
She was of northern European descent. She was unaware of any family history of blood-related disorders. She had been admitted to the hospital 6 weeks earlier for symptomatic anemia after taking nitrofurantoin for a urinary tract infection. At that time, she received 2 units of packed red blood cells and then was discharged. Follow-up blood work done 2 weeks later—including a glucose-6 phosphate dehydrogenase (G6PD) assay—was normal.
On physical examination, she was pale and weak. Her hemoglobin level was 5.5 g/dL (reference range 14.0–17.5), with normal white blood cell and platelet counts and an elevated reticulocyte count. A comprehensive metabolic panel showed elevated indirect bilirubin and lactate dehydrogenase levels. A direct Coombs test for autoimmune hemolytic anemia was negative, as was a haptoglobin assay to look for intravascular hemolytic anemia. G6PD levels were normal, yet a peripheral blood smear (Figure 1) showed features of G6PD deficiency.
What was the cause of her anemia?

GLUCOSE-6-PHOSPHATE DEHYDROGENASE DEFICIENCY
G6PD deficiency is an X-linked disorder1 that can present as hemolytic anemia. Symptoms of hemolysis can range from mild to severe on exposure to an inciting agent. Men are more commonly affected than women, and affected women are mostly heterozygous. The severity of hemolysis in heterozygous women depends on inactivation of the unaffected X chromosome in some cells.
When exposed to oxidizing agents, people with G6PD deficiency do not have enough nicotinamide adenine dinucleotide phosphate to protect red blood cells.2 This leads to oxidative denaturation of hemoglobin, formation of methemoglobin, and denaturation of globulin. These products are insoluble; they collect in red blood cells and are called Heinz bodies.3 When red blood cells containing Heinz bodies pass through the liver and spleen, the insoluble masses are taken up by macrophages, causing hemolysis and the formation of “bite cells”4 (so named because macrophages “bite” the Heinz bodies out of the red blood cells).
Patients with G6PD deficiency have all the clinical features of hemolytic anemia. On laboratory testing, the Coombs test is negative, the G6PD level is low, and the peripheral smear shows bite cells. The G6PD level is falsely normal or elevated during acute hemolysis because red blood cells deficient in G6PD are removed from circulation and replaced by young red blood cells. The G6PD level is also elevated after blood transfusion. Thus, the G6PD level should be tested 3 months after an acute event.
Hemolysis in G6PD is usually intermittent and self-limited. No treatment is needed except for avoidance of triggers and transfusion for symptomatic anemia. Of note, triggers include some of the drugs commonly used for urinary tract infections (sulfa drugs, nitrofurantoin, phenazopyridine) and antimalarials. Fava beans are also known to cause hemolytic crisis. A complete list of things to avoid can be found at www.g6pd.org/en/G6PDDeficiency/SafeUnsafe/DaEvitare_ISS-it.
There is no commercially available genetic testing kit for G6PD deficiency. Mutation analysis and G6PD gene sequencing are possible but are neither routinely done nor widely available.
BACK TO OUR PATIENT
Our patient’s hemolytic anemia was most likely drug-induced, secondary to a relative deficiency of G6PD. She had been taking nitrofurantoin and phenazopyridine; both of these are oxidizing agents and are known to cause acute hemolytic anemia in people with G6PD deficiency. The G6PD level can be normal after a recent blood transfusion and, as in our patient, during an acute episode of hemolysis.
Because of the strong suspicion of G6PD deficiency, both drugs were stopped when the patient was discharged from the hospital. She did not take either drug for 3 months. Her G6PD level was then retested and was found to be low, confirming the diagnosis. The patient was then advised not to take those drugs again. Since then, her hemoglobin level has remained stable and she has not needed any more blood transfusions.
- Mason PJ, Bautista JM, Gilsanz F. G6PD deficiency: the genotype-phenotype association. Blood Rev 2007; 21:267–283.
- Arese P, De Flora A. Pathophysiology of hemolysis in glucose-6-phosphate dehydrogenase deficiency. Semin Hematol 1990; 27:1–40.
- Jacob HS. Mechanisms of Heinz body formation and attachment to red cell membrane. Semin Hematol 1970; 7:341–354.
- Rifkind RA. Heinz body anemia: an ultrastructural study. II. Red cell sequestration and destruction. Blood 1965; 26:433–448.
- Mason PJ, Bautista JM, Gilsanz F. G6PD deficiency: the genotype-phenotype association. Blood Rev 2007; 21:267–283.
- Arese P, De Flora A. Pathophysiology of hemolysis in glucose-6-phosphate dehydrogenase deficiency. Semin Hematol 1990; 27:1–40.
- Jacob HS. Mechanisms of Heinz body formation and attachment to red cell membrane. Semin Hematol 1970; 7:341–354.
- Rifkind RA. Heinz body anemia: an ultrastructural study. II. Red cell sequestration and destruction. Blood 1965; 26:433–448.
Canagliflozin: Improving diabetes by making urine sweet
Glycosuria used to be a sign of uncontrolled diabetes and was something to be corrected, not a therapeutic mechanism. But now we have a new class of drugs that lower plasma glucose levels by increasing the renal excretion of glucose.
Here, we will review canagliflozin, the first in a new class of drugs for type 2 diabetes: how it works, who is a candidate for it, and what to watch out for.
THE NEED FOR NEW DIABETES DRUGS
Diabetes mellitus affects more than 25.8 million people in the United States—8.3% of the population—and this staggering number is rising.1 Among US residents age 65 and older, more than 10.9 million (26.9%) have diabetes.1 People with uncontrolled diabetes are at risk of microvascular complications such as retinopathy, nephropathy, and neuropathy, as well as cardiovascular disease. Diabetes is the leading cause of blindness, chronic kidney disease, and nontraumatic lower-limb amputation in the United States.1
Type 2 diabetes accounts for more than 90% of cases of diabetes in the United States, Europe, and Canada.2 It is characterized by insulin resistance, decreased beta-cell function, and progressive beta-cell decline.3
Current American Diabetes Association guidelines for the treatment of diabetes recommend a hemoglobin A1c target of less than 7.0%.4 Initial management includes lifestyle modifications such as changes in diet and an increase in exercise, as well as consideration of metformin treatment at the same time. If glucose levels remain uncontrolled despite these efforts, other drugs should be added.
A number of oral and injectable antihyperglycemic drugs are available to help achieve this goal, though none is without risk of adverse effects. Those available up to now include metformin, sulfonylureas, meglitinides, alpha-glucosidase inhibitors, thiazolidinediones, gliptins, glucagon-like peptide-1 agonists, amylin analogues, colesevelam, dopamine agonists, and insulin.5 Most of the available antihyperglycemics target the liver, pancreas, gut, and muscle to improve insulin sensitivity, reduce insulin resistance, or stimulate insulin secretion.
Despite the abundance of agents, type 2 diabetes remains uncontrolled in many patients. Only 57.1% of participants with previously diagnosed diabetes in the 2003–2006 National Health and Nutrition Examination Survey were at the hemoglobin A1c goal of less than 7.0%.6 Possible reasons for failure include adverse effects such as hypoglycemia, weight gain, and gastrointestinal symptoms resulting in discontinued use, nonadherence to the prescribed regimen, and failure to increase the dosage or to add additional agents, including insulin, to optimize glycemic control as beta-cell function declines over time.
HOW THE KIDNEYS HANDLE GLUCOSE
In the kidney, glucose is filtered in the glomerulus and then is reabsorbed in the proximal tubule. Normally, the filtered glucose is all reabsorbed unless the glucose load exceeds the kidney’s absorptive capacity. Membrane proteins called sodium-glucose cotransporters reabsorb glucose at the proximal tubule and return it into the peripheral circulation. Glucose enters the tubular epithelial cell with sodium by passive cotransport via the sodiumglucose cotransporters, and then exits on the other side via the glucose transporter GLUT in the basolateral membrane.
Two sodium-glucose transporters that act in the proximal tubule of the kidney have been identified: SGLT1 and SGLT2. SGLT2 reabsorbs most of the glucose in the early segment of the proximal tubule, while SLGT1 reabsorbs the remaining glucose at the distal end.7 SGLT2 is responsible for more than 90% of renal tubular reabsorption of glucose and is found only in the proximal tubule, whereas SGLT1 is found mainly in the gastrointestinal tract.8
Patients with type 2 diabetes have a higher capacity for glucose reabsorption in the proximal tubule as a result of the up-regulation of SGLT2.9
SGLT2 INHIBITORS AND TYPE 2 DIABETES
Drugs that inhibit SGLT2 block reabsorption of glucose in the proximal tubule, lowering the renal threshold for glucose and thereby increasing urinary glucose excretion and lowering the serum glucose level in patients with hyperglycemia. This mechanism of action is insulin-independent.
On March 29, 2013, canagliflozin became the first SGLT2 inhibitor to be approved in the United States for the treatment of type 2 diabetes.10 However, it is not the first of its class to be introduced.
Dapagliflozin was the first SGLT2 inhibitor approved in Europe and has been available there since November 2012. However, the US Food and Drug Administration withheld its approval in the United States in January 2012 because of concerns of a possible association with cancer, specifically breast and bladder cancers, as well as possible liver injury.10 Canagliflozin does not appear to share this risk.
Several other SGLT2 inhibitors may soon be available. Empagliflozin is in phase III trials, and the manufacturer has filed for approval in the United States. Ipragliflozin is awaiting approval in Japan.
CANAGLIFLOZIN: PHARMACOKINETICS AND THERAPEUTIC EFFICACY
Canagliflozin reaches its peak plasma concentration within 1 to 2 hours of oral administration.11 Its half-life is 10.6 hours with a 100-mg dose and 13.1 hours with a 300-mg dose. A steady state is typically achieved in 4 to 5 days.11
Canagliflozin lowers fasting plasma glucose and hemoglobin A1c levels in a dose-dependent manner.10,11 These effects are independent of age, sex, body mass index, and race.12 Postprandial glucose levels are also lowered.
Other potential benefits of canagliflozin include lowering of the systolic blood pressure and, especially important in obese people with type 2 diabetes, weight loss.12 Aside from metformin, which occasionally results in modest weight loss, other oral drugs used in treating type 2 diabetes are weight-neutral or can cause weight gain.
Trials of canagliflozin
Nine phase III trials of canagliflozin have enrolled 10,285 patients, in one of the largest clinical trial programs in type 2 diabetes to date.10 Several of these trials evaluated canagliflozin as monotherapy, whereas others assessed its effect as an add-on therapy in combination with another antihyperglycemic agent such as a sulfonylurea, metformin, pioglitazone, or insulin. There has not yet been a trial directly comparing canagliflozin with metformin.
Four of the placebo-controlled trials evaluated canagliflozin as monotherapy, canagliflozin added to metformin alone, canagliflozin added to metformin plus glimepiride, and canagliflozin added to metformin plus pioglitazone.
When canagliflozin was used as monotherapy, hemoglobin A1c levels at 26 weeks were an absolute 0.91% lower in the canagliflozin 100 mg/day group than in the placebo group, and an absolute 1.16% lower in the canagliflozin 300 mg/day group than in the placebo group (P < .001 for both).12 Patients lost 2.8% of their body weight with canagliflozin 100 mg and 3.3% with canagliflozin 300 mg, compared with 0.6% with placebo. Systolic blood pressure fell by a mean of 3.7 mm Hg with the 100-mg dose and by a mean of 5.4 mm Hg with the 300-mg dose compared with placebo (P < .001 for both dose groups).12
When canagliflozin was added to metformin, with glimepiride as the comparator drug, there was a 5.2% weight reduction with the 100-mg dose, a 5.7% reduction with 300 mg, and a 1% gain with glimepiride. Hemoglobin A1c fell about equally in the three groups.11
When canagliflozin was added to metformin and a sulfonylurea, with sitagliptin as the comparator third drug, the 300-mg canagliflozin dosage group had a 2.8% weight reduction.11
WHAT ARE THE ADVERSE EFFECTS?
Overall, canagliflozin seems to be well tolerated. The most common adverse effects reported in the clinical trials were genital yeast infections, urinary tract infections, and increased urination.
Genital yeast infections were more common in women than in men, occurring in 10.4% of women who received canagliflozin 100 mg and in 11.4% of women who received 300 mg, compared with only 3.2% in the placebo group.11
Urinary tract infections occurred in 5.9% of the 100-mg group and in 4.3% of the 300-mg group, compared with 4.0% of the placebo group.11
Postural hypotension. Lowering of blood pressure and symptoms of postural hypotension were also reported, and these may be attributed to the drug’s mild osmotic diuretic effect. The risk of adverse effects of volume depletion was dose-dependent; in patients over age 75, they occurred in 4.9% of those taking 100 mg and in 8.7% of those taking 300 mg, compared with 2.6% of those in the placebo or active-comparator groups.11 Therefore, one should exercise particular caution when starting this drug in the elderly or in patients taking diuretics or multiple antihypertensive drugs.
Hypoglycemia. When canagliflozin was used as monotherapy, the incidence of hypoglycemia over 26 weeks was similar to that with placebo, occurring in 3.6% of the 100-mg group, 3.0% of the 300-mg group, and 2.6% of the placebo group.12 Canagliflozin was associated with fewer episodes of hypoglycemia than were sulfonylureas, and the number of episodes was similar to that in patients taking gliptins. There was a higher overall incidence of hypoglycemia when canagliflozin was used in combination with a sulfonylurea or with insulin than when it was used as monotherapy.11
Hyperkalemia. Patients with moderate renal impairment or who are on potassiumsparing drugs or drugs that interfere with the renin-angiotensin-aldosterone system may be at higher risk of hyperkalemia, so close monitoring of potassium is recommended. There was also a dose-dependent increase in serum phosphate and magnesium levels, more notably in patients with moderate renal impairment within the first 3 weeks of starting the drug.11
Patients on canagliflozin who are also taking digoxin, ritonavir, phenytoin, phenobarbital, or rifampin should be closely monitored because of the risk of drug-drug interactions.11 Specifically, there was an increase in mean peak digoxin concentrations when used with canagliflozin 300 mg, and the use of phenytoin, phenobarbital, and ritonavir decreased the efficacy of canagliflozin.
WHAT ARE THE CARDIOVASCULAR RISKS OR LONG-TERM CONCERNS?
Dose-dependent increases in low-density lipoprotein cholesterol (LDL-C) may be seen with canagliflozin. Mean changes from baseline compared with placebo were 4.4 mg/dL (4.5%) with canagliflozin 100 mg and 8.3 mg/dL (8%) with canagliflozin 300 mg.11
There was also an increase in non-high-density lipoprotein cholesterol (non-HDL-C).12 Compared with placebo, mean non-HDL-C levels rose by 2.1 mg/dL (1.5%) with canagliflozin 100 mg and 5.1 mg/dL (3.6%) with 300 mg.11
In the 26-week canagliflozin monotherapy trial, archived blood samples in a small subgroup of patients (n = 349) were measured for apolipoprotein-B, which was found to increase by 1.2% with canagliflozin 100 mg and 3.5% with canagliflozin 300 mg, compared with 0.9% in the placebo group.12
Although small, the increase in LDL-C seen with this drug could be a concern, as diabetic patients are already at higher risk of cardiovascular events. The mechanism of this increase is not yet known, though it may be related to metabolic changes from urinary glucose excretion.12
The Canagliflozin Cardiovascular Assessment Study (CANVAS) is a randomized placebo-controlled trial in more than 4,000 patients with type 2 diabetes who have a history of or are at high risk of cardiovascular events. Currently under way, it is evaluating the occurrence of major adverse cardiovascular events (the primary end point) in patients randomized to receive canagliflozin 100 mg, canagliflozin 300 mg, or placebo once daily for up to 4 years. Secondary end points will be the drug’s effects on fasting plasma insulin and glucose, progression of albuminuria, body weight, blood pressure, HDL-C, LDL-C, bone mineral density, markers of bone turnover, and body composition.10 This trial will run for 9 years, to be completed in 2018.13
The CANVAS investigators have already reported that within the first month of treatment, 13 patients taking canagliflozin suffered a major cardiovascular event, including stroke (one of which was fatal) compared with just one patient taking placebo. These events were not seen after the first month. The hazard ratio for major adverse cardiovascular events within the first 30 days was 6.49, but this dropped to 0.89 after the first 30 days.10
Additional issues that should be addressed in long-term postmarketing studies include possible relationships with cancers and pancreatitis and the safety of the drug in pregnancy and in children with diabetes.10
WHO IS A CANDIDATE FOR THIS DRUG?
Canagliflozin is approved for use as monotherapy in addition to lifestyle modifications. It is also approved for use with other antihyperglycemic drugs, including metformin.
Obese patients with type 2 diabetes and normal kidney function may have the greatest benefit. Because of canagliflozin’s insulinin-dependent mechanism of action, patients with both early and late type 2 diabetes may benefit from its ability to lower hemoglobin A1c and blood glucose.14
Although it can be used in patients with moderate (but not severe) kidney disease, canagliflozin does not appear to be as effective in these patients, who had higher rates of adverse effects.11 It is not indicated for patients with type 1 diabetes, type 2 diabetes with ketonuria, or end-stage renal disease (estimated glomerular filtration rate < 45 mL/min or receiving dialysis).11 It also is not yet recommended for use in pregnant women or patients under age 18.
The recommended starting dose of canagliflozin is 100 mg once daily, taken with breakfast. This can be increased to 300 mg once daily if tolerated. However, patients with an estimated glomerular filtration rate of 45 to 60 mL/min should not exceed the 100- mg dose. No dose adjustment is required in patients with mild to moderate hepatic impairment. It is not recommended, however, in patients with severe hepatic impairment.11
Comment. Although canagliflozin is approved as monotherapy, metformin remains my choice for first-line oral therapy. Because canagliflozin is more expensive and its long-term affects are still relatively unknown, I prefer to use it as an adjunct, and believe it will be a useful addition, especially in obese patients who are seeking to lose weight.
WHAT IS THE COST OF THIS DRUG?
The suggested price is $10.53 per tablet (AmerisourceBergen), which is comparable to that of other newer drugs for type 2 diabetes.
THE BOTTOM LINE
The availability of canagliflozin as an additional oral antihyperglycemic option may prove helpful in managing patients with type 2 diabetes who experience adverse effects with other antihyperglycemic drugs.
As with any new drug, questions remain about the long-term risks of canagliflozin. However, it seems to be well tolerated, especially in patients with normal kidney function, and poses a low risk of hypoglycemia. The slight increase in LDL-C may prompt more aggressive lipid management. Whether blood pressure-lowering and weight loss will offset this increase in LDL-C is yet to be determined. Ongoing studies will help to further elucidate whether there is an increased risk of cardiovascular events.
Finally, canagliflozin distinguishes itself from other oral diabetes drugs by its added benefit of weight loss, an appealing side effect, especially in the growing population of obese individuals with type 2 diabetes mellitus.
- Centers for Disease Control and Prevention (CDC). Diabetes data and trends. www.cdc.gov/diabetes/statistics/. Accessed September 6, 2013.
- National Diabetes Information Clearinghouse (NDIC), National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). National diabetes statistics, 2011. www.diabetes.niddk.nih.gov/dm/pubs/statistics/. Accessed September 6, 2013.
- Campbell RK. Fate of the beta-cell in the pathophysiology of type 2 diabetes. J Am Pharm Assoc (2003). 2009; 49(suppl 1):S10–S15.
- American Diabetes Association. Executive summary: standards of medical care in diabetes—2012. Diabetes Care 2012; 35(suppl 1):S4–S10.
- Blonde L. Current antihyperglycemic treatment strategies for patients with type 2 diabetes mellitus. Cleve Clin J Med 2009; 76(suppl 5):S4–S11.
- Cheung BM, Ong KL, Cherny SS, Sham PC, Tso AW, Lam KS. Diabetes prevalence and therapeutic target achievement in the United States, 1999 to 2006. Am J Med 2009; 122:443–453.
- Vallon V. The proximal tubule in the pathophysiology of the diabetic kidney. Am J Physiol Regul Integr Comp Physiol 2011; 300:R1009–R1022.
- DeFronzo RA, Davidson JA, Del Prato S. The role of the kidneys in glucose homeostasis: a new path towards normalizing glycaemia. Diabetes Obes Metab 2012; 14:5–14.
- Pfister M, Whaley JM, Zhang L, List JF. Inhibition of SGLT2: a novel strategy for treatment of type 2 diabetes mellitus. Clin Pharmacol Ther 2011; 89:621–625.
- Food and Drug Administration (FDA). FDA Briefing Document. NDA 204042. Invokana (canagliflozin) Tablets. www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/EndocrinologicandMetabolicDrugsAdvisoryCommittee/UCM334550.pdf. Accessed September 6, 2013.
- INVOKANA (canagliflozin) tablets, for oral use. Prescribing Information. Janssen Pharmaceuticals, Inc. www.janssenpharmaceuticalsinc.com/assets/invokana_prescribing_info.pdf. Accessed September 6, 2013.
- Stenlöf K, Cefalu WT, Kim KA, et al. Efficacy and safety of canagliflozin monotherapy in subjects with type 2 diabetes mellitus inadequately controlled with diet and exercise. Diabetes Obes Metab 2013; 15:372–382.
- US National Institutes of Health. ClinicalTrials.gov. CANVAS—CA Nagliflozin cardio Vascular Assessment Study. http://clinicaltrials.gov/show/NCT01032629. Accessed September 6, 2013.
- Devineni D, Morrow L, Hompesch M, et al. Canagliflozin improves glycaemic control over 28 days in subjects with type 2 diabetes not optimally controlled on insulin. Diabetes Obes Metab 2012; 14:539–545.
Glycosuria used to be a sign of uncontrolled diabetes and was something to be corrected, not a therapeutic mechanism. But now we have a new class of drugs that lower plasma glucose levels by increasing the renal excretion of glucose.
Here, we will review canagliflozin, the first in a new class of drugs for type 2 diabetes: how it works, who is a candidate for it, and what to watch out for.
THE NEED FOR NEW DIABETES DRUGS
Diabetes mellitus affects more than 25.8 million people in the United States—8.3% of the population—and this staggering number is rising.1 Among US residents age 65 and older, more than 10.9 million (26.9%) have diabetes.1 People with uncontrolled diabetes are at risk of microvascular complications such as retinopathy, nephropathy, and neuropathy, as well as cardiovascular disease. Diabetes is the leading cause of blindness, chronic kidney disease, and nontraumatic lower-limb amputation in the United States.1
Type 2 diabetes accounts for more than 90% of cases of diabetes in the United States, Europe, and Canada.2 It is characterized by insulin resistance, decreased beta-cell function, and progressive beta-cell decline.3
Current American Diabetes Association guidelines for the treatment of diabetes recommend a hemoglobin A1c target of less than 7.0%.4 Initial management includes lifestyle modifications such as changes in diet and an increase in exercise, as well as consideration of metformin treatment at the same time. If glucose levels remain uncontrolled despite these efforts, other drugs should be added.
A number of oral and injectable antihyperglycemic drugs are available to help achieve this goal, though none is without risk of adverse effects. Those available up to now include metformin, sulfonylureas, meglitinides, alpha-glucosidase inhibitors, thiazolidinediones, gliptins, glucagon-like peptide-1 agonists, amylin analogues, colesevelam, dopamine agonists, and insulin.5 Most of the available antihyperglycemics target the liver, pancreas, gut, and muscle to improve insulin sensitivity, reduce insulin resistance, or stimulate insulin secretion.
Despite the abundance of agents, type 2 diabetes remains uncontrolled in many patients. Only 57.1% of participants with previously diagnosed diabetes in the 2003–2006 National Health and Nutrition Examination Survey were at the hemoglobin A1c goal of less than 7.0%.6 Possible reasons for failure include adverse effects such as hypoglycemia, weight gain, and gastrointestinal symptoms resulting in discontinued use, nonadherence to the prescribed regimen, and failure to increase the dosage or to add additional agents, including insulin, to optimize glycemic control as beta-cell function declines over time.
HOW THE KIDNEYS HANDLE GLUCOSE
In the kidney, glucose is filtered in the glomerulus and then is reabsorbed in the proximal tubule. Normally, the filtered glucose is all reabsorbed unless the glucose load exceeds the kidney’s absorptive capacity. Membrane proteins called sodium-glucose cotransporters reabsorb glucose at the proximal tubule and return it into the peripheral circulation. Glucose enters the tubular epithelial cell with sodium by passive cotransport via the sodiumglucose cotransporters, and then exits on the other side via the glucose transporter GLUT in the basolateral membrane.
Two sodium-glucose transporters that act in the proximal tubule of the kidney have been identified: SGLT1 and SGLT2. SGLT2 reabsorbs most of the glucose in the early segment of the proximal tubule, while SLGT1 reabsorbs the remaining glucose at the distal end.7 SGLT2 is responsible for more than 90% of renal tubular reabsorption of glucose and is found only in the proximal tubule, whereas SGLT1 is found mainly in the gastrointestinal tract.8
Patients with type 2 diabetes have a higher capacity for glucose reabsorption in the proximal tubule as a result of the up-regulation of SGLT2.9
SGLT2 INHIBITORS AND TYPE 2 DIABETES
Drugs that inhibit SGLT2 block reabsorption of glucose in the proximal tubule, lowering the renal threshold for glucose and thereby increasing urinary glucose excretion and lowering the serum glucose level in patients with hyperglycemia. This mechanism of action is insulin-independent.
On March 29, 2013, canagliflozin became the first SGLT2 inhibitor to be approved in the United States for the treatment of type 2 diabetes.10 However, it is not the first of its class to be introduced.
Dapagliflozin was the first SGLT2 inhibitor approved in Europe and has been available there since November 2012. However, the US Food and Drug Administration withheld its approval in the United States in January 2012 because of concerns of a possible association with cancer, specifically breast and bladder cancers, as well as possible liver injury.10 Canagliflozin does not appear to share this risk.
Several other SGLT2 inhibitors may soon be available. Empagliflozin is in phase III trials, and the manufacturer has filed for approval in the United States. Ipragliflozin is awaiting approval in Japan.
CANAGLIFLOZIN: PHARMACOKINETICS AND THERAPEUTIC EFFICACY
Canagliflozin reaches its peak plasma concentration within 1 to 2 hours of oral administration.11 Its half-life is 10.6 hours with a 100-mg dose and 13.1 hours with a 300-mg dose. A steady state is typically achieved in 4 to 5 days.11
Canagliflozin lowers fasting plasma glucose and hemoglobin A1c levels in a dose-dependent manner.10,11 These effects are independent of age, sex, body mass index, and race.12 Postprandial glucose levels are also lowered.
Other potential benefits of canagliflozin include lowering of the systolic blood pressure and, especially important in obese people with type 2 diabetes, weight loss.12 Aside from metformin, which occasionally results in modest weight loss, other oral drugs used in treating type 2 diabetes are weight-neutral or can cause weight gain.
Trials of canagliflozin
Nine phase III trials of canagliflozin have enrolled 10,285 patients, in one of the largest clinical trial programs in type 2 diabetes to date.10 Several of these trials evaluated canagliflozin as monotherapy, whereas others assessed its effect as an add-on therapy in combination with another antihyperglycemic agent such as a sulfonylurea, metformin, pioglitazone, or insulin. There has not yet been a trial directly comparing canagliflozin with metformin.
Four of the placebo-controlled trials evaluated canagliflozin as monotherapy, canagliflozin added to metformin alone, canagliflozin added to metformin plus glimepiride, and canagliflozin added to metformin plus pioglitazone.
When canagliflozin was used as monotherapy, hemoglobin A1c levels at 26 weeks were an absolute 0.91% lower in the canagliflozin 100 mg/day group than in the placebo group, and an absolute 1.16% lower in the canagliflozin 300 mg/day group than in the placebo group (P < .001 for both).12 Patients lost 2.8% of their body weight with canagliflozin 100 mg and 3.3% with canagliflozin 300 mg, compared with 0.6% with placebo. Systolic blood pressure fell by a mean of 3.7 mm Hg with the 100-mg dose and by a mean of 5.4 mm Hg with the 300-mg dose compared with placebo (P < .001 for both dose groups).12
When canagliflozin was added to metformin, with glimepiride as the comparator drug, there was a 5.2% weight reduction with the 100-mg dose, a 5.7% reduction with 300 mg, and a 1% gain with glimepiride. Hemoglobin A1c fell about equally in the three groups.11
When canagliflozin was added to metformin and a sulfonylurea, with sitagliptin as the comparator third drug, the 300-mg canagliflozin dosage group had a 2.8% weight reduction.11
WHAT ARE THE ADVERSE EFFECTS?
Overall, canagliflozin seems to be well tolerated. The most common adverse effects reported in the clinical trials were genital yeast infections, urinary tract infections, and increased urination.
Genital yeast infections were more common in women than in men, occurring in 10.4% of women who received canagliflozin 100 mg and in 11.4% of women who received 300 mg, compared with only 3.2% in the placebo group.11
Urinary tract infections occurred in 5.9% of the 100-mg group and in 4.3% of the 300-mg group, compared with 4.0% of the placebo group.11
Postural hypotension. Lowering of blood pressure and symptoms of postural hypotension were also reported, and these may be attributed to the drug’s mild osmotic diuretic effect. The risk of adverse effects of volume depletion was dose-dependent; in patients over age 75, they occurred in 4.9% of those taking 100 mg and in 8.7% of those taking 300 mg, compared with 2.6% of those in the placebo or active-comparator groups.11 Therefore, one should exercise particular caution when starting this drug in the elderly or in patients taking diuretics or multiple antihypertensive drugs.
Hypoglycemia. When canagliflozin was used as monotherapy, the incidence of hypoglycemia over 26 weeks was similar to that with placebo, occurring in 3.6% of the 100-mg group, 3.0% of the 300-mg group, and 2.6% of the placebo group.12 Canagliflozin was associated with fewer episodes of hypoglycemia than were sulfonylureas, and the number of episodes was similar to that in patients taking gliptins. There was a higher overall incidence of hypoglycemia when canagliflozin was used in combination with a sulfonylurea or with insulin than when it was used as monotherapy.11
Hyperkalemia. Patients with moderate renal impairment or who are on potassiumsparing drugs or drugs that interfere with the renin-angiotensin-aldosterone system may be at higher risk of hyperkalemia, so close monitoring of potassium is recommended. There was also a dose-dependent increase in serum phosphate and magnesium levels, more notably in patients with moderate renal impairment within the first 3 weeks of starting the drug.11
Patients on canagliflozin who are also taking digoxin, ritonavir, phenytoin, phenobarbital, or rifampin should be closely monitored because of the risk of drug-drug interactions.11 Specifically, there was an increase in mean peak digoxin concentrations when used with canagliflozin 300 mg, and the use of phenytoin, phenobarbital, and ritonavir decreased the efficacy of canagliflozin.
WHAT ARE THE CARDIOVASCULAR RISKS OR LONG-TERM CONCERNS?
Dose-dependent increases in low-density lipoprotein cholesterol (LDL-C) may be seen with canagliflozin. Mean changes from baseline compared with placebo were 4.4 mg/dL (4.5%) with canagliflozin 100 mg and 8.3 mg/dL (8%) with canagliflozin 300 mg.11
There was also an increase in non-high-density lipoprotein cholesterol (non-HDL-C).12 Compared with placebo, mean non-HDL-C levels rose by 2.1 mg/dL (1.5%) with canagliflozin 100 mg and 5.1 mg/dL (3.6%) with 300 mg.11
In the 26-week canagliflozin monotherapy trial, archived blood samples in a small subgroup of patients (n = 349) were measured for apolipoprotein-B, which was found to increase by 1.2% with canagliflozin 100 mg and 3.5% with canagliflozin 300 mg, compared with 0.9% in the placebo group.12
Although small, the increase in LDL-C seen with this drug could be a concern, as diabetic patients are already at higher risk of cardiovascular events. The mechanism of this increase is not yet known, though it may be related to metabolic changes from urinary glucose excretion.12
The Canagliflozin Cardiovascular Assessment Study (CANVAS) is a randomized placebo-controlled trial in more than 4,000 patients with type 2 diabetes who have a history of or are at high risk of cardiovascular events. Currently under way, it is evaluating the occurrence of major adverse cardiovascular events (the primary end point) in patients randomized to receive canagliflozin 100 mg, canagliflozin 300 mg, or placebo once daily for up to 4 years. Secondary end points will be the drug’s effects on fasting plasma insulin and glucose, progression of albuminuria, body weight, blood pressure, HDL-C, LDL-C, bone mineral density, markers of bone turnover, and body composition.10 This trial will run for 9 years, to be completed in 2018.13
The CANVAS investigators have already reported that within the first month of treatment, 13 patients taking canagliflozin suffered a major cardiovascular event, including stroke (one of which was fatal) compared with just one patient taking placebo. These events were not seen after the first month. The hazard ratio for major adverse cardiovascular events within the first 30 days was 6.49, but this dropped to 0.89 after the first 30 days.10
Additional issues that should be addressed in long-term postmarketing studies include possible relationships with cancers and pancreatitis and the safety of the drug in pregnancy and in children with diabetes.10
WHO IS A CANDIDATE FOR THIS DRUG?
Canagliflozin is approved for use as monotherapy in addition to lifestyle modifications. It is also approved for use with other antihyperglycemic drugs, including metformin.
Obese patients with type 2 diabetes and normal kidney function may have the greatest benefit. Because of canagliflozin’s insulinin-dependent mechanism of action, patients with both early and late type 2 diabetes may benefit from its ability to lower hemoglobin A1c and blood glucose.14
Although it can be used in patients with moderate (but not severe) kidney disease, canagliflozin does not appear to be as effective in these patients, who had higher rates of adverse effects.11 It is not indicated for patients with type 1 diabetes, type 2 diabetes with ketonuria, or end-stage renal disease (estimated glomerular filtration rate < 45 mL/min or receiving dialysis).11 It also is not yet recommended for use in pregnant women or patients under age 18.
The recommended starting dose of canagliflozin is 100 mg once daily, taken with breakfast. This can be increased to 300 mg once daily if tolerated. However, patients with an estimated glomerular filtration rate of 45 to 60 mL/min should not exceed the 100- mg dose. No dose adjustment is required in patients with mild to moderate hepatic impairment. It is not recommended, however, in patients with severe hepatic impairment.11
Comment. Although canagliflozin is approved as monotherapy, metformin remains my choice for first-line oral therapy. Because canagliflozin is more expensive and its long-term affects are still relatively unknown, I prefer to use it as an adjunct, and believe it will be a useful addition, especially in obese patients who are seeking to lose weight.
WHAT IS THE COST OF THIS DRUG?
The suggested price is $10.53 per tablet (AmerisourceBergen), which is comparable to that of other newer drugs for type 2 diabetes.
THE BOTTOM LINE
The availability of canagliflozin as an additional oral antihyperglycemic option may prove helpful in managing patients with type 2 diabetes who experience adverse effects with other antihyperglycemic drugs.
As with any new drug, questions remain about the long-term risks of canagliflozin. However, it seems to be well tolerated, especially in patients with normal kidney function, and poses a low risk of hypoglycemia. The slight increase in LDL-C may prompt more aggressive lipid management. Whether blood pressure-lowering and weight loss will offset this increase in LDL-C is yet to be determined. Ongoing studies will help to further elucidate whether there is an increased risk of cardiovascular events.
Finally, canagliflozin distinguishes itself from other oral diabetes drugs by its added benefit of weight loss, an appealing side effect, especially in the growing population of obese individuals with type 2 diabetes mellitus.
Glycosuria used to be a sign of uncontrolled diabetes and was something to be corrected, not a therapeutic mechanism. But now we have a new class of drugs that lower plasma glucose levels by increasing the renal excretion of glucose.
Here, we will review canagliflozin, the first in a new class of drugs for type 2 diabetes: how it works, who is a candidate for it, and what to watch out for.
THE NEED FOR NEW DIABETES DRUGS
Diabetes mellitus affects more than 25.8 million people in the United States—8.3% of the population—and this staggering number is rising.1 Among US residents age 65 and older, more than 10.9 million (26.9%) have diabetes.1 People with uncontrolled diabetes are at risk of microvascular complications such as retinopathy, nephropathy, and neuropathy, as well as cardiovascular disease. Diabetes is the leading cause of blindness, chronic kidney disease, and nontraumatic lower-limb amputation in the United States.1
Type 2 diabetes accounts for more than 90% of cases of diabetes in the United States, Europe, and Canada.2 It is characterized by insulin resistance, decreased beta-cell function, and progressive beta-cell decline.3
Current American Diabetes Association guidelines for the treatment of diabetes recommend a hemoglobin A1c target of less than 7.0%.4 Initial management includes lifestyle modifications such as changes in diet and an increase in exercise, as well as consideration of metformin treatment at the same time. If glucose levels remain uncontrolled despite these efforts, other drugs should be added.
A number of oral and injectable antihyperglycemic drugs are available to help achieve this goal, though none is without risk of adverse effects. Those available up to now include metformin, sulfonylureas, meglitinides, alpha-glucosidase inhibitors, thiazolidinediones, gliptins, glucagon-like peptide-1 agonists, amylin analogues, colesevelam, dopamine agonists, and insulin.5 Most of the available antihyperglycemics target the liver, pancreas, gut, and muscle to improve insulin sensitivity, reduce insulin resistance, or stimulate insulin secretion.
Despite the abundance of agents, type 2 diabetes remains uncontrolled in many patients. Only 57.1% of participants with previously diagnosed diabetes in the 2003–2006 National Health and Nutrition Examination Survey were at the hemoglobin A1c goal of less than 7.0%.6 Possible reasons for failure include adverse effects such as hypoglycemia, weight gain, and gastrointestinal symptoms resulting in discontinued use, nonadherence to the prescribed regimen, and failure to increase the dosage or to add additional agents, including insulin, to optimize glycemic control as beta-cell function declines over time.
HOW THE KIDNEYS HANDLE GLUCOSE
In the kidney, glucose is filtered in the glomerulus and then is reabsorbed in the proximal tubule. Normally, the filtered glucose is all reabsorbed unless the glucose load exceeds the kidney’s absorptive capacity. Membrane proteins called sodium-glucose cotransporters reabsorb glucose at the proximal tubule and return it into the peripheral circulation. Glucose enters the tubular epithelial cell with sodium by passive cotransport via the sodiumglucose cotransporters, and then exits on the other side via the glucose transporter GLUT in the basolateral membrane.
Two sodium-glucose transporters that act in the proximal tubule of the kidney have been identified: SGLT1 and SGLT2. SGLT2 reabsorbs most of the glucose in the early segment of the proximal tubule, while SLGT1 reabsorbs the remaining glucose at the distal end.7 SGLT2 is responsible for more than 90% of renal tubular reabsorption of glucose and is found only in the proximal tubule, whereas SGLT1 is found mainly in the gastrointestinal tract.8
Patients with type 2 diabetes have a higher capacity for glucose reabsorption in the proximal tubule as a result of the up-regulation of SGLT2.9
SGLT2 INHIBITORS AND TYPE 2 DIABETES
Drugs that inhibit SGLT2 block reabsorption of glucose in the proximal tubule, lowering the renal threshold for glucose and thereby increasing urinary glucose excretion and lowering the serum glucose level in patients with hyperglycemia. This mechanism of action is insulin-independent.
On March 29, 2013, canagliflozin became the first SGLT2 inhibitor to be approved in the United States for the treatment of type 2 diabetes.10 However, it is not the first of its class to be introduced.
Dapagliflozin was the first SGLT2 inhibitor approved in Europe and has been available there since November 2012. However, the US Food and Drug Administration withheld its approval in the United States in January 2012 because of concerns of a possible association with cancer, specifically breast and bladder cancers, as well as possible liver injury.10 Canagliflozin does not appear to share this risk.
Several other SGLT2 inhibitors may soon be available. Empagliflozin is in phase III trials, and the manufacturer has filed for approval in the United States. Ipragliflozin is awaiting approval in Japan.
CANAGLIFLOZIN: PHARMACOKINETICS AND THERAPEUTIC EFFICACY
Canagliflozin reaches its peak plasma concentration within 1 to 2 hours of oral administration.11 Its half-life is 10.6 hours with a 100-mg dose and 13.1 hours with a 300-mg dose. A steady state is typically achieved in 4 to 5 days.11
Canagliflozin lowers fasting plasma glucose and hemoglobin A1c levels in a dose-dependent manner.10,11 These effects are independent of age, sex, body mass index, and race.12 Postprandial glucose levels are also lowered.
Other potential benefits of canagliflozin include lowering of the systolic blood pressure and, especially important in obese people with type 2 diabetes, weight loss.12 Aside from metformin, which occasionally results in modest weight loss, other oral drugs used in treating type 2 diabetes are weight-neutral or can cause weight gain.
Trials of canagliflozin
Nine phase III trials of canagliflozin have enrolled 10,285 patients, in one of the largest clinical trial programs in type 2 diabetes to date.10 Several of these trials evaluated canagliflozin as monotherapy, whereas others assessed its effect as an add-on therapy in combination with another antihyperglycemic agent such as a sulfonylurea, metformin, pioglitazone, or insulin. There has not yet been a trial directly comparing canagliflozin with metformin.
Four of the placebo-controlled trials evaluated canagliflozin as monotherapy, canagliflozin added to metformin alone, canagliflozin added to metformin plus glimepiride, and canagliflozin added to metformin plus pioglitazone.
When canagliflozin was used as monotherapy, hemoglobin A1c levels at 26 weeks were an absolute 0.91% lower in the canagliflozin 100 mg/day group than in the placebo group, and an absolute 1.16% lower in the canagliflozin 300 mg/day group than in the placebo group (P < .001 for both).12 Patients lost 2.8% of their body weight with canagliflozin 100 mg and 3.3% with canagliflozin 300 mg, compared with 0.6% with placebo. Systolic blood pressure fell by a mean of 3.7 mm Hg with the 100-mg dose and by a mean of 5.4 mm Hg with the 300-mg dose compared with placebo (P < .001 for both dose groups).12
When canagliflozin was added to metformin, with glimepiride as the comparator drug, there was a 5.2% weight reduction with the 100-mg dose, a 5.7% reduction with 300 mg, and a 1% gain with glimepiride. Hemoglobin A1c fell about equally in the three groups.11
When canagliflozin was added to metformin and a sulfonylurea, with sitagliptin as the comparator third drug, the 300-mg canagliflozin dosage group had a 2.8% weight reduction.11
WHAT ARE THE ADVERSE EFFECTS?
Overall, canagliflozin seems to be well tolerated. The most common adverse effects reported in the clinical trials were genital yeast infections, urinary tract infections, and increased urination.
Genital yeast infections were more common in women than in men, occurring in 10.4% of women who received canagliflozin 100 mg and in 11.4% of women who received 300 mg, compared with only 3.2% in the placebo group.11
Urinary tract infections occurred in 5.9% of the 100-mg group and in 4.3% of the 300-mg group, compared with 4.0% of the placebo group.11
Postural hypotension. Lowering of blood pressure and symptoms of postural hypotension were also reported, and these may be attributed to the drug’s mild osmotic diuretic effect. The risk of adverse effects of volume depletion was dose-dependent; in patients over age 75, they occurred in 4.9% of those taking 100 mg and in 8.7% of those taking 300 mg, compared with 2.6% of those in the placebo or active-comparator groups.11 Therefore, one should exercise particular caution when starting this drug in the elderly or in patients taking diuretics or multiple antihypertensive drugs.
Hypoglycemia. When canagliflozin was used as monotherapy, the incidence of hypoglycemia over 26 weeks was similar to that with placebo, occurring in 3.6% of the 100-mg group, 3.0% of the 300-mg group, and 2.6% of the placebo group.12 Canagliflozin was associated with fewer episodes of hypoglycemia than were sulfonylureas, and the number of episodes was similar to that in patients taking gliptins. There was a higher overall incidence of hypoglycemia when canagliflozin was used in combination with a sulfonylurea or with insulin than when it was used as monotherapy.11
Hyperkalemia. Patients with moderate renal impairment or who are on potassiumsparing drugs or drugs that interfere with the renin-angiotensin-aldosterone system may be at higher risk of hyperkalemia, so close monitoring of potassium is recommended. There was also a dose-dependent increase in serum phosphate and magnesium levels, more notably in patients with moderate renal impairment within the first 3 weeks of starting the drug.11
Patients on canagliflozin who are also taking digoxin, ritonavir, phenytoin, phenobarbital, or rifampin should be closely monitored because of the risk of drug-drug interactions.11 Specifically, there was an increase in mean peak digoxin concentrations when used with canagliflozin 300 mg, and the use of phenytoin, phenobarbital, and ritonavir decreased the efficacy of canagliflozin.
WHAT ARE THE CARDIOVASCULAR RISKS OR LONG-TERM CONCERNS?
Dose-dependent increases in low-density lipoprotein cholesterol (LDL-C) may be seen with canagliflozin. Mean changes from baseline compared with placebo were 4.4 mg/dL (4.5%) with canagliflozin 100 mg and 8.3 mg/dL (8%) with canagliflozin 300 mg.11
There was also an increase in non-high-density lipoprotein cholesterol (non-HDL-C).12 Compared with placebo, mean non-HDL-C levels rose by 2.1 mg/dL (1.5%) with canagliflozin 100 mg and 5.1 mg/dL (3.6%) with 300 mg.11
In the 26-week canagliflozin monotherapy trial, archived blood samples in a small subgroup of patients (n = 349) were measured for apolipoprotein-B, which was found to increase by 1.2% with canagliflozin 100 mg and 3.5% with canagliflozin 300 mg, compared with 0.9% in the placebo group.12
Although small, the increase in LDL-C seen with this drug could be a concern, as diabetic patients are already at higher risk of cardiovascular events. The mechanism of this increase is not yet known, though it may be related to metabolic changes from urinary glucose excretion.12
The Canagliflozin Cardiovascular Assessment Study (CANVAS) is a randomized placebo-controlled trial in more than 4,000 patients with type 2 diabetes who have a history of or are at high risk of cardiovascular events. Currently under way, it is evaluating the occurrence of major adverse cardiovascular events (the primary end point) in patients randomized to receive canagliflozin 100 mg, canagliflozin 300 mg, or placebo once daily for up to 4 years. Secondary end points will be the drug’s effects on fasting plasma insulin and glucose, progression of albuminuria, body weight, blood pressure, HDL-C, LDL-C, bone mineral density, markers of bone turnover, and body composition.10 This trial will run for 9 years, to be completed in 2018.13
The CANVAS investigators have already reported that within the first month of treatment, 13 patients taking canagliflozin suffered a major cardiovascular event, including stroke (one of which was fatal) compared with just one patient taking placebo. These events were not seen after the first month. The hazard ratio for major adverse cardiovascular events within the first 30 days was 6.49, but this dropped to 0.89 after the first 30 days.10
Additional issues that should be addressed in long-term postmarketing studies include possible relationships with cancers and pancreatitis and the safety of the drug in pregnancy and in children with diabetes.10
WHO IS A CANDIDATE FOR THIS DRUG?
Canagliflozin is approved for use as monotherapy in addition to lifestyle modifications. It is also approved for use with other antihyperglycemic drugs, including metformin.
Obese patients with type 2 diabetes and normal kidney function may have the greatest benefit. Because of canagliflozin’s insulinin-dependent mechanism of action, patients with both early and late type 2 diabetes may benefit from its ability to lower hemoglobin A1c and blood glucose.14
Although it can be used in patients with moderate (but not severe) kidney disease, canagliflozin does not appear to be as effective in these patients, who had higher rates of adverse effects.11 It is not indicated for patients with type 1 diabetes, type 2 diabetes with ketonuria, or end-stage renal disease (estimated glomerular filtration rate < 45 mL/min or receiving dialysis).11 It also is not yet recommended for use in pregnant women or patients under age 18.
The recommended starting dose of canagliflozin is 100 mg once daily, taken with breakfast. This can be increased to 300 mg once daily if tolerated. However, patients with an estimated glomerular filtration rate of 45 to 60 mL/min should not exceed the 100- mg dose. No dose adjustment is required in patients with mild to moderate hepatic impairment. It is not recommended, however, in patients with severe hepatic impairment.11
Comment. Although canagliflozin is approved as monotherapy, metformin remains my choice for first-line oral therapy. Because canagliflozin is more expensive and its long-term affects are still relatively unknown, I prefer to use it as an adjunct, and believe it will be a useful addition, especially in obese patients who are seeking to lose weight.
WHAT IS THE COST OF THIS DRUG?
The suggested price is $10.53 per tablet (AmerisourceBergen), which is comparable to that of other newer drugs for type 2 diabetes.
THE BOTTOM LINE
The availability of canagliflozin as an additional oral antihyperglycemic option may prove helpful in managing patients with type 2 diabetes who experience adverse effects with other antihyperglycemic drugs.
As with any new drug, questions remain about the long-term risks of canagliflozin. However, it seems to be well tolerated, especially in patients with normal kidney function, and poses a low risk of hypoglycemia. The slight increase in LDL-C may prompt more aggressive lipid management. Whether blood pressure-lowering and weight loss will offset this increase in LDL-C is yet to be determined. Ongoing studies will help to further elucidate whether there is an increased risk of cardiovascular events.
Finally, canagliflozin distinguishes itself from other oral diabetes drugs by its added benefit of weight loss, an appealing side effect, especially in the growing population of obese individuals with type 2 diabetes mellitus.
- Centers for Disease Control and Prevention (CDC). Diabetes data and trends. www.cdc.gov/diabetes/statistics/. Accessed September 6, 2013.
- National Diabetes Information Clearinghouse (NDIC), National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). National diabetes statistics, 2011. www.diabetes.niddk.nih.gov/dm/pubs/statistics/. Accessed September 6, 2013.
- Campbell RK. Fate of the beta-cell in the pathophysiology of type 2 diabetes. J Am Pharm Assoc (2003). 2009; 49(suppl 1):S10–S15.
- American Diabetes Association. Executive summary: standards of medical care in diabetes—2012. Diabetes Care 2012; 35(suppl 1):S4–S10.
- Blonde L. Current antihyperglycemic treatment strategies for patients with type 2 diabetes mellitus. Cleve Clin J Med 2009; 76(suppl 5):S4–S11.
- Cheung BM, Ong KL, Cherny SS, Sham PC, Tso AW, Lam KS. Diabetes prevalence and therapeutic target achievement in the United States, 1999 to 2006. Am J Med 2009; 122:443–453.
- Vallon V. The proximal tubule in the pathophysiology of the diabetic kidney. Am J Physiol Regul Integr Comp Physiol 2011; 300:R1009–R1022.
- DeFronzo RA, Davidson JA, Del Prato S. The role of the kidneys in glucose homeostasis: a new path towards normalizing glycaemia. Diabetes Obes Metab 2012; 14:5–14.
- Pfister M, Whaley JM, Zhang L, List JF. Inhibition of SGLT2: a novel strategy for treatment of type 2 diabetes mellitus. Clin Pharmacol Ther 2011; 89:621–625.
- Food and Drug Administration (FDA). FDA Briefing Document. NDA 204042. Invokana (canagliflozin) Tablets. www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/EndocrinologicandMetabolicDrugsAdvisoryCommittee/UCM334550.pdf. Accessed September 6, 2013.
- INVOKANA (canagliflozin) tablets, for oral use. Prescribing Information. Janssen Pharmaceuticals, Inc. www.janssenpharmaceuticalsinc.com/assets/invokana_prescribing_info.pdf. Accessed September 6, 2013.
- Stenlöf K, Cefalu WT, Kim KA, et al. Efficacy and safety of canagliflozin monotherapy in subjects with type 2 diabetes mellitus inadequately controlled with diet and exercise. Diabetes Obes Metab 2013; 15:372–382.
- US National Institutes of Health. ClinicalTrials.gov. CANVAS—CA Nagliflozin cardio Vascular Assessment Study. http://clinicaltrials.gov/show/NCT01032629. Accessed September 6, 2013.
- Devineni D, Morrow L, Hompesch M, et al. Canagliflozin improves glycaemic control over 28 days in subjects with type 2 diabetes not optimally controlled on insulin. Diabetes Obes Metab 2012; 14:539–545.
- Centers for Disease Control and Prevention (CDC). Diabetes data and trends. www.cdc.gov/diabetes/statistics/. Accessed September 6, 2013.
- National Diabetes Information Clearinghouse (NDIC), National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). National diabetes statistics, 2011. www.diabetes.niddk.nih.gov/dm/pubs/statistics/. Accessed September 6, 2013.
- Campbell RK. Fate of the beta-cell in the pathophysiology of type 2 diabetes. J Am Pharm Assoc (2003). 2009; 49(suppl 1):S10–S15.
- American Diabetes Association. Executive summary: standards of medical care in diabetes—2012. Diabetes Care 2012; 35(suppl 1):S4–S10.
- Blonde L. Current antihyperglycemic treatment strategies for patients with type 2 diabetes mellitus. Cleve Clin J Med 2009; 76(suppl 5):S4–S11.
- Cheung BM, Ong KL, Cherny SS, Sham PC, Tso AW, Lam KS. Diabetes prevalence and therapeutic target achievement in the United States, 1999 to 2006. Am J Med 2009; 122:443–453.
- Vallon V. The proximal tubule in the pathophysiology of the diabetic kidney. Am J Physiol Regul Integr Comp Physiol 2011; 300:R1009–R1022.
- DeFronzo RA, Davidson JA, Del Prato S. The role of the kidneys in glucose homeostasis: a new path towards normalizing glycaemia. Diabetes Obes Metab 2012; 14:5–14.
- Pfister M, Whaley JM, Zhang L, List JF. Inhibition of SGLT2: a novel strategy for treatment of type 2 diabetes mellitus. Clin Pharmacol Ther 2011; 89:621–625.
- Food and Drug Administration (FDA). FDA Briefing Document. NDA 204042. Invokana (canagliflozin) Tablets. www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/EndocrinologicandMetabolicDrugsAdvisoryCommittee/UCM334550.pdf. Accessed September 6, 2013.
- INVOKANA (canagliflozin) tablets, for oral use. Prescribing Information. Janssen Pharmaceuticals, Inc. www.janssenpharmaceuticalsinc.com/assets/invokana_prescribing_info.pdf. Accessed September 6, 2013.
- Stenlöf K, Cefalu WT, Kim KA, et al. Efficacy and safety of canagliflozin monotherapy in subjects with type 2 diabetes mellitus inadequately controlled with diet and exercise. Diabetes Obes Metab 2013; 15:372–382.
- US National Institutes of Health. ClinicalTrials.gov. CANVAS—CA Nagliflozin cardio Vascular Assessment Study. http://clinicaltrials.gov/show/NCT01032629. Accessed September 6, 2013.
- Devineni D, Morrow L, Hompesch M, et al. Canagliflozin improves glycaemic control over 28 days in subjects with type 2 diabetes not optimally controlled on insulin. Diabetes Obes Metab 2012; 14:539–545.
KEY POINTS
- Type 2 diabetes is ubiquitous and, despite an abundance of agents, often remains uncontrolled.
- Canagliflozin and other drugs of its class cause glucose to be spilled in the urine by reducing the amount reabsorbed by the kidney.
- In clinical trials, canagliflozin lowered hemoglobin A1c levels by approximately 1 absolute percentage point.
- Beyond the adverse effects to be expected from the mechanism of action of the drug (ie, genital yeast infections, urinary tract infections, and hypotension caused by osmotic diuresis), canagliflozin seems to increase plasma levels of low-density lipoprotein cholesterol. This may be worrisome, as diabetic patients are already at increased risk of cardiovascular disease.
When people with diabetes go to surgery
Over the past decade, recommendations about the ideal glucose target in hospitalized diabetic patients have fluctuated. The controversy has extended to diabetic patients in various types of intensive care units and to those headed to the operating room. Although proposals exist on how to manage diabetes in the operating room, including intraoperative insulin infusions, outcomes probably depend more on how glucose is managed during the patient’s postoperative stay in the hospital. For patients who are less critically ill and less medically complex, continuous insulin infusions are used infrequently, and insulin is often prescribed by algorithm or, archaically, by some form of “catch-up” sliding scale. Studies indicate that even the fairly loose glucose target of 70 to 180 mg/dL is achieved consistently in only a few patients.1
In view of a number of observations, including the link between hyperglycemia and postoperative wound infections, studies were designed to test the hypothesis that aggressively keeping glucose levels quite low in critically ill and postoperative diabetic patients would be beneficial. Instead, most of these studies found that overly tight glucose control in these settings led to untoward outcomes—and not only as the result of hypoglycemic episodes. Aiming for a modest serum glucose target of 150 to 200 mg/dL can significantly reduce the postoperative death rate, but the beneficial reduction is no greater if the target is less than 150 mg/dL.
With a looser glucose target, pre- and perioperative management of insulin-dependent diabetic patients can be simplified. Dobri and Lansang discuss the key practical principles of managing insulin before the patient goes to the operating suite. They emphasize relevant pearls of insulin physiology and discuss several scenarios we often encounter.
In fact, the principles they review are equally useful to remember when we admit diabetic patients to the hospital with orders to keep them “npo” while planning and awaiting tests or other procedures. A key take-home point is that severely insulinopenic patients require some exogenous basal insulin, even when not eating.
- Lopes R, Albrecht A, Williams J, et al. Postoperative glucose control following coronary artery bypass graft surgery: predictors and clinical outcomes. J Am Coll Cardiol 2013; 61:e1601.
Over the past decade, recommendations about the ideal glucose target in hospitalized diabetic patients have fluctuated. The controversy has extended to diabetic patients in various types of intensive care units and to those headed to the operating room. Although proposals exist on how to manage diabetes in the operating room, including intraoperative insulin infusions, outcomes probably depend more on how glucose is managed during the patient’s postoperative stay in the hospital. For patients who are less critically ill and less medically complex, continuous insulin infusions are used infrequently, and insulin is often prescribed by algorithm or, archaically, by some form of “catch-up” sliding scale. Studies indicate that even the fairly loose glucose target of 70 to 180 mg/dL is achieved consistently in only a few patients.1
In view of a number of observations, including the link between hyperglycemia and postoperative wound infections, studies were designed to test the hypothesis that aggressively keeping glucose levels quite low in critically ill and postoperative diabetic patients would be beneficial. Instead, most of these studies found that overly tight glucose control in these settings led to untoward outcomes—and not only as the result of hypoglycemic episodes. Aiming for a modest serum glucose target of 150 to 200 mg/dL can significantly reduce the postoperative death rate, but the beneficial reduction is no greater if the target is less than 150 mg/dL.
With a looser glucose target, pre- and perioperative management of insulin-dependent diabetic patients can be simplified. Dobri and Lansang discuss the key practical principles of managing insulin before the patient goes to the operating suite. They emphasize relevant pearls of insulin physiology and discuss several scenarios we often encounter.
In fact, the principles they review are equally useful to remember when we admit diabetic patients to the hospital with orders to keep them “npo” while planning and awaiting tests or other procedures. A key take-home point is that severely insulinopenic patients require some exogenous basal insulin, even when not eating.
Over the past decade, recommendations about the ideal glucose target in hospitalized diabetic patients have fluctuated. The controversy has extended to diabetic patients in various types of intensive care units and to those headed to the operating room. Although proposals exist on how to manage diabetes in the operating room, including intraoperative insulin infusions, outcomes probably depend more on how glucose is managed during the patient’s postoperative stay in the hospital. For patients who are less critically ill and less medically complex, continuous insulin infusions are used infrequently, and insulin is often prescribed by algorithm or, archaically, by some form of “catch-up” sliding scale. Studies indicate that even the fairly loose glucose target of 70 to 180 mg/dL is achieved consistently in only a few patients.1
In view of a number of observations, including the link between hyperglycemia and postoperative wound infections, studies were designed to test the hypothesis that aggressively keeping glucose levels quite low in critically ill and postoperative diabetic patients would be beneficial. Instead, most of these studies found that overly tight glucose control in these settings led to untoward outcomes—and not only as the result of hypoglycemic episodes. Aiming for a modest serum glucose target of 150 to 200 mg/dL can significantly reduce the postoperative death rate, but the beneficial reduction is no greater if the target is less than 150 mg/dL.
With a looser glucose target, pre- and perioperative management of insulin-dependent diabetic patients can be simplified. Dobri and Lansang discuss the key practical principles of managing insulin before the patient goes to the operating suite. They emphasize relevant pearls of insulin physiology and discuss several scenarios we often encounter.
In fact, the principles they review are equally useful to remember when we admit diabetic patients to the hospital with orders to keep them “npo” while planning and awaiting tests or other procedures. A key take-home point is that severely insulinopenic patients require some exogenous basal insulin, even when not eating.
- Lopes R, Albrecht A, Williams J, et al. Postoperative glucose control following coronary artery bypass graft surgery: predictors and clinical outcomes. J Am Coll Cardiol 2013; 61:e1601.
- Lopes R, Albrecht A, Williams J, et al. Postoperative glucose control following coronary artery bypass graft surgery: predictors and clinical outcomes. J Am Coll Cardiol 2013; 61:e1601.
How should we manage insulin therapy before surgery?
Continuing at least part of the basal insulin is the reasonable, physiologic approach to controlling glucose levels before surgery in patients with diabetes. The process involves three basic steps:
- Ascertaining the type of diabetes
- Adjusting the basal insulin dosage
- Stopping the prandial insulin.
The steps are the same whether the surgery is major or minor. These recommendations are based on general principles of insulin action, data from large databases of surgical inpatients, and expert clinical experience translated into standardized protocols.1,2
WHY CONTINUE THE INSULIN?
Stopping or decreasing insulin because of a fear of hypoglycemia is not appropriate, as the resulting hyperglycemia can lead to delayed wound healing, wound infection, fluid and electrolyte shifts, diabetic ketoacidosis, and hyperosmolar states.
Insulin inhibits both gluconeogenesis and conversion of glycogen to glucose, processes that occur regardless of food intake. It also inhibits degradation of fats to fatty acids and of fatty acids to ketones. This is why inadequate insulin dosing can lead to uncontrolled hyperglycemia and even ketoacidosis, and thus why long-acting insulin is needed in a fasting state.
STEP 1: ASCERTAIN THE TYPE OF DIABETES
Does the patient have type 1 or type 2 diabetes, and does that even matter?
The type of diabetes should not matter, since ideally the insulin should be dosed the same for both types. However, the consequences of inappropriate insulin management may be different.
Usually, the type of diabetes can be ascertained by the history. If the patient was diagnosed at age 40 or later and was on oral medication for years before insulin was started, then he or she most likely has type 2. If the patient was younger than 40 at the time of diagnosis, was lean, and was started on insulin within a year of diagnosis, then he or she likely has type 1.
If this information is not available or is unreliable and the patient has been on insulin for many years, we recommend viewing the patient as being insulinopenic, ie, not producing enough insulin endogenously and thus requiring insulin at all times.
Though checking for antibody markers of type 1 diabetes might give a more definitive answer, it is not practical before surgery.
In the setting of surgical stress, withholding the basal insulin preoperatively and just giving a small dose of fast-acting (see Table 1 for the different classes of insulin) or shortacting insulin as part of a sliding scale (ie, insulin given only when the blood glucose reaches a certain high level) can send a patient with type 1 diabetes into diabetic ketoacidosis by the end of the day. This is less likely to occur in a patient with type 2 diabetes with some endogenous insulin secretion.
STEP 2: ADJUST THE BASAL INSULIN
Basal insulin is the insulin that the healthy person’s body produces when fasting. For a diabetic patient already on insulin, basal insulin is insulin injected to prevent ketogenesis, glycogenolysis, and gluconeogenesis in the fasting state.
If the basal insulin is long-acting
Long-acting insulins have a relatively peakless profile and, when properly dosed, should not result in hypoglycemia when a patient is fasting.
Preoperatively, the patient should take it as close as possible to the usual time of injection. This could be at home either at bedtime the night before surgery or the morning of surgery. If there is concern for hypoglycemia, the injection can be given when the patient is at the hospital.
- If the patient does not tend to have hypoglycemic episodes and the total daily basal insulin dose is roughly the same as the total daily mealtime (prandial) dose (eg, 50% basal, 50% prandial ratio), the full dose of basal insulin can be given.3
Example: If the patient is on insulin glargine 30 U at bedtime and insulin lispro 10 U with each meal and does not have hypoglycemic episodes, then insulin glargine 30 U should be taken at bedtime.
- If the patient has hypoglycemic episodes at home, then the basal insulin can be reduced by 25%.3
Example: If the patient is on insulin glargine 30 U at bedtime and insulin glulisine 10 U with each meal (appropriate proportion of doses, similar to the example above) but has hypoglycemic episodes at home on this regimen, then only 22 U of insulin glargine should be taken at bedtime.
- If the patient’s regimen has disproportionately more basal insulin than mealtime insulin, then the total daily doses can be added and half can be given as the basal insulin.
Example: If the patient is on insulin detemir 30 U every morning at 6 am and insulin aspart 6 U with each meal and has no hypoglycemic episodes, then 24 U of insulin detemir should be taken in the morning (ie, half of the total of 30 + 6 + 6 + 6).
- In the less common scenario of diabetes managed only with basal insulin (no other diabetes injections or oral agents), then half of the dose can be given.
- If the patient is on twice-daily long-acting basal insulin, then both the dose the night before surgery and the dose the morning of surgery should be adjusted.
If the basal insulin is intermediate-acting
The intermediate-acting insulin neutral protamine Hagedorn (NPH) is usually given twice a day because of its profile (Table 1).
- On the night before surgery, the full dose of NPH insulin should be taken, unless the patient will now skip a nighttime meal because of taking nothing by mouth, in which case the dose can be decreased by 25%.1
- On the morning of surgery, since the patient will be skipping breakfast and probably also lunch, the dose should be reduced by 50%.3,4
Special situation: Premixed insulins
Premixed insulins (70/30, 75/25) are a combination of intermediate-acting insulin and either fast-acting or short-acting insulin. In other words, they are combinations of basal and prandial insulin. Their use is thus not ideal in the preoperative period. There are two options in these situations.
One option is to switch to a regimen that includes long-acting insulin. If the patient is admitted for surgery, then the hospital staff can change the insulin regimen to long-acting basal insulin. A quick formula for conversion is to add all the premixed insulin doses and give half as basal insulin on the morning of surgery, similar to the scenario above for the patient with long-acting basal insulin that was out of proportion to the prandial insulin injections.
For example, if the usual regimen is insulin 70/30 NPH/Regular, 60 U with breakfast, 30 U with dinner, then the patient can take 45 U of insulin glargine (which is half of 60 + 30) in the morning or evening before surgery.
Another option is to adjust the dose of pre-mixed insulin. Sometimes it is not feasible or economical to change the patient’s premixed insulin just before surgery. In these situations, the patient can take half of the morning dose, followed by dextrose-containing intravenous fluids and blood glucose checks.
We recommend preoperatively giving at least part of the patient’s previous basal insulin, regardless of the type of diabetes, the type of surgery, or the fasting period.
STEP 3: STOP THE PRANDIAL INSULIN
Prandial insulin—given before each meal to cover the carbohydrates to be consumed—should be stopped the morning of surgery.3,4
WHAT ABOUT SLIDING SCALE INSULIN?
Using a sliding scale alone has no known benefit. Although it can be a quick fix to correct a high glucose level, it should be added to the basal insulin and not used as the sole insulin therapy. If a sliding scale is used, fast-acting insulin (aspart, glulisine, lispro) is preferred over regular insulin because of the more rapid onset and shorter duration of action.
Patients already using a supplemental insulin scale can apply it to correct a blood glucose above 200 mg/dL on the morning of surgery.
MAINTENANCE FLUIDS
As long as glucose levels are not very elevated (ie, > 200 mg/dL), after 12 hours on a nothing-by-mouth regimen, provide dextrose in the IV fluid to prevent hypoglycemia (eg, the patient received long-acting insulin and the glucose levels are running low) or to prevent starvation ketosis, which may result in ketones in the blood or urine. We recommend 5% dextrose in half-normal (0.45%) saline at 50 to 75 mL/hour as maintenance fluid; the infusion rate should be lower if fluid overload is a concern.
POSTOPERATIVE INSULIN MANAGEMENT
Once patients are discharged and can go back to their previous routine, they can restart their usual insulin regimen the same evening. The prandial insulin will be resumed when the regular diet is reintroduced, and the doses will be adjusted according to food intake.
- Joshi GP, Chung F, Vann MA, et al; Society for Ambulatory Anesthesia. Society for Ambulatory Anesthesia consensus statement on perioperative blood glucose management in diabetic patients undergoing ambulatory surgery. Anesth Analg 2010; 111:1378–1387.
- DiNardo M, Donihi AC, Forte P, Gieraltowski L, Korytkowski M. Standardized glycemic management and perioperative glycemic outcomes in patients with diabetes mellitus who undergo same-day surgery. Endocr Pract 2011; 17:404–411.
- Vann MA. Perioperative management of ambulatory surgical patients with diabetes mellitus. Curr Opin Anaesthesiol 2009; 22:718–724.
- Meneghini LF. Perioperative management of diabetes: translating evidence into practice. Cleve Clin J Med 2009; 76(suppl 4):S53–S59.
Continuing at least part of the basal insulin is the reasonable, physiologic approach to controlling glucose levels before surgery in patients with diabetes. The process involves three basic steps:
- Ascertaining the type of diabetes
- Adjusting the basal insulin dosage
- Stopping the prandial insulin.
The steps are the same whether the surgery is major or minor. These recommendations are based on general principles of insulin action, data from large databases of surgical inpatients, and expert clinical experience translated into standardized protocols.1,2
WHY CONTINUE THE INSULIN?
Stopping or decreasing insulin because of a fear of hypoglycemia is not appropriate, as the resulting hyperglycemia can lead to delayed wound healing, wound infection, fluid and electrolyte shifts, diabetic ketoacidosis, and hyperosmolar states.
Insulin inhibits both gluconeogenesis and conversion of glycogen to glucose, processes that occur regardless of food intake. It also inhibits degradation of fats to fatty acids and of fatty acids to ketones. This is why inadequate insulin dosing can lead to uncontrolled hyperglycemia and even ketoacidosis, and thus why long-acting insulin is needed in a fasting state.
STEP 1: ASCERTAIN THE TYPE OF DIABETES
Does the patient have type 1 or type 2 diabetes, and does that even matter?
The type of diabetes should not matter, since ideally the insulin should be dosed the same for both types. However, the consequences of inappropriate insulin management may be different.
Usually, the type of diabetes can be ascertained by the history. If the patient was diagnosed at age 40 or later and was on oral medication for years before insulin was started, then he or she most likely has type 2. If the patient was younger than 40 at the time of diagnosis, was lean, and was started on insulin within a year of diagnosis, then he or she likely has type 1.
If this information is not available or is unreliable and the patient has been on insulin for many years, we recommend viewing the patient as being insulinopenic, ie, not producing enough insulin endogenously and thus requiring insulin at all times.
Though checking for antibody markers of type 1 diabetes might give a more definitive answer, it is not practical before surgery.
In the setting of surgical stress, withholding the basal insulin preoperatively and just giving a small dose of fast-acting (see Table 1 for the different classes of insulin) or shortacting insulin as part of a sliding scale (ie, insulin given only when the blood glucose reaches a certain high level) can send a patient with type 1 diabetes into diabetic ketoacidosis by the end of the day. This is less likely to occur in a patient with type 2 diabetes with some endogenous insulin secretion.
STEP 2: ADJUST THE BASAL INSULIN
Basal insulin is the insulin that the healthy person’s body produces when fasting. For a diabetic patient already on insulin, basal insulin is insulin injected to prevent ketogenesis, glycogenolysis, and gluconeogenesis in the fasting state.
If the basal insulin is long-acting
Long-acting insulins have a relatively peakless profile and, when properly dosed, should not result in hypoglycemia when a patient is fasting.
Preoperatively, the patient should take it as close as possible to the usual time of injection. This could be at home either at bedtime the night before surgery or the morning of surgery. If there is concern for hypoglycemia, the injection can be given when the patient is at the hospital.
- If the patient does not tend to have hypoglycemic episodes and the total daily basal insulin dose is roughly the same as the total daily mealtime (prandial) dose (eg, 50% basal, 50% prandial ratio), the full dose of basal insulin can be given.3
Example: If the patient is on insulin glargine 30 U at bedtime and insulin lispro 10 U with each meal and does not have hypoglycemic episodes, then insulin glargine 30 U should be taken at bedtime.
- If the patient has hypoglycemic episodes at home, then the basal insulin can be reduced by 25%.3
Example: If the patient is on insulin glargine 30 U at bedtime and insulin glulisine 10 U with each meal (appropriate proportion of doses, similar to the example above) but has hypoglycemic episodes at home on this regimen, then only 22 U of insulin glargine should be taken at bedtime.
- If the patient’s regimen has disproportionately more basal insulin than mealtime insulin, then the total daily doses can be added and half can be given as the basal insulin.
Example: If the patient is on insulin detemir 30 U every morning at 6 am and insulin aspart 6 U with each meal and has no hypoglycemic episodes, then 24 U of insulin detemir should be taken in the morning (ie, half of the total of 30 + 6 + 6 + 6).
- In the less common scenario of diabetes managed only with basal insulin (no other diabetes injections or oral agents), then half of the dose can be given.
- If the patient is on twice-daily long-acting basal insulin, then both the dose the night before surgery and the dose the morning of surgery should be adjusted.
If the basal insulin is intermediate-acting
The intermediate-acting insulin neutral protamine Hagedorn (NPH) is usually given twice a day because of its profile (Table 1).
- On the night before surgery, the full dose of NPH insulin should be taken, unless the patient will now skip a nighttime meal because of taking nothing by mouth, in which case the dose can be decreased by 25%.1
- On the morning of surgery, since the patient will be skipping breakfast and probably also lunch, the dose should be reduced by 50%.3,4
Special situation: Premixed insulins
Premixed insulins (70/30, 75/25) are a combination of intermediate-acting insulin and either fast-acting or short-acting insulin. In other words, they are combinations of basal and prandial insulin. Their use is thus not ideal in the preoperative period. There are two options in these situations.
One option is to switch to a regimen that includes long-acting insulin. If the patient is admitted for surgery, then the hospital staff can change the insulin regimen to long-acting basal insulin. A quick formula for conversion is to add all the premixed insulin doses and give half as basal insulin on the morning of surgery, similar to the scenario above for the patient with long-acting basal insulin that was out of proportion to the prandial insulin injections.
For example, if the usual regimen is insulin 70/30 NPH/Regular, 60 U with breakfast, 30 U with dinner, then the patient can take 45 U of insulin glargine (which is half of 60 + 30) in the morning or evening before surgery.
Another option is to adjust the dose of pre-mixed insulin. Sometimes it is not feasible or economical to change the patient’s premixed insulin just before surgery. In these situations, the patient can take half of the morning dose, followed by dextrose-containing intravenous fluids and blood glucose checks.
We recommend preoperatively giving at least part of the patient’s previous basal insulin, regardless of the type of diabetes, the type of surgery, or the fasting period.
STEP 3: STOP THE PRANDIAL INSULIN
Prandial insulin—given before each meal to cover the carbohydrates to be consumed—should be stopped the morning of surgery.3,4
WHAT ABOUT SLIDING SCALE INSULIN?
Using a sliding scale alone has no known benefit. Although it can be a quick fix to correct a high glucose level, it should be added to the basal insulin and not used as the sole insulin therapy. If a sliding scale is used, fast-acting insulin (aspart, glulisine, lispro) is preferred over regular insulin because of the more rapid onset and shorter duration of action.
Patients already using a supplemental insulin scale can apply it to correct a blood glucose above 200 mg/dL on the morning of surgery.
MAINTENANCE FLUIDS
As long as glucose levels are not very elevated (ie, > 200 mg/dL), after 12 hours on a nothing-by-mouth regimen, provide dextrose in the IV fluid to prevent hypoglycemia (eg, the patient received long-acting insulin and the glucose levels are running low) or to prevent starvation ketosis, which may result in ketones in the blood or urine. We recommend 5% dextrose in half-normal (0.45%) saline at 50 to 75 mL/hour as maintenance fluid; the infusion rate should be lower if fluid overload is a concern.
POSTOPERATIVE INSULIN MANAGEMENT
Once patients are discharged and can go back to their previous routine, they can restart their usual insulin regimen the same evening. The prandial insulin will be resumed when the regular diet is reintroduced, and the doses will be adjusted according to food intake.
Continuing at least part of the basal insulin is the reasonable, physiologic approach to controlling glucose levels before surgery in patients with diabetes. The process involves three basic steps:
- Ascertaining the type of diabetes
- Adjusting the basal insulin dosage
- Stopping the prandial insulin.
The steps are the same whether the surgery is major or minor. These recommendations are based on general principles of insulin action, data from large databases of surgical inpatients, and expert clinical experience translated into standardized protocols.1,2
WHY CONTINUE THE INSULIN?
Stopping or decreasing insulin because of a fear of hypoglycemia is not appropriate, as the resulting hyperglycemia can lead to delayed wound healing, wound infection, fluid and electrolyte shifts, diabetic ketoacidosis, and hyperosmolar states.
Insulin inhibits both gluconeogenesis and conversion of glycogen to glucose, processes that occur regardless of food intake. It also inhibits degradation of fats to fatty acids and of fatty acids to ketones. This is why inadequate insulin dosing can lead to uncontrolled hyperglycemia and even ketoacidosis, and thus why long-acting insulin is needed in a fasting state.
STEP 1: ASCERTAIN THE TYPE OF DIABETES
Does the patient have type 1 or type 2 diabetes, and does that even matter?
The type of diabetes should not matter, since ideally the insulin should be dosed the same for both types. However, the consequences of inappropriate insulin management may be different.
Usually, the type of diabetes can be ascertained by the history. If the patient was diagnosed at age 40 or later and was on oral medication for years before insulin was started, then he or she most likely has type 2. If the patient was younger than 40 at the time of diagnosis, was lean, and was started on insulin within a year of diagnosis, then he or she likely has type 1.
If this information is not available or is unreliable and the patient has been on insulin for many years, we recommend viewing the patient as being insulinopenic, ie, not producing enough insulin endogenously and thus requiring insulin at all times.
Though checking for antibody markers of type 1 diabetes might give a more definitive answer, it is not practical before surgery.
In the setting of surgical stress, withholding the basal insulin preoperatively and just giving a small dose of fast-acting (see Table 1 for the different classes of insulin) or shortacting insulin as part of a sliding scale (ie, insulin given only when the blood glucose reaches a certain high level) can send a patient with type 1 diabetes into diabetic ketoacidosis by the end of the day. This is less likely to occur in a patient with type 2 diabetes with some endogenous insulin secretion.
STEP 2: ADJUST THE BASAL INSULIN
Basal insulin is the insulin that the healthy person’s body produces when fasting. For a diabetic patient already on insulin, basal insulin is insulin injected to prevent ketogenesis, glycogenolysis, and gluconeogenesis in the fasting state.
If the basal insulin is long-acting
Long-acting insulins have a relatively peakless profile and, when properly dosed, should not result in hypoglycemia when a patient is fasting.
Preoperatively, the patient should take it as close as possible to the usual time of injection. This could be at home either at bedtime the night before surgery or the morning of surgery. If there is concern for hypoglycemia, the injection can be given when the patient is at the hospital.
- If the patient does not tend to have hypoglycemic episodes and the total daily basal insulin dose is roughly the same as the total daily mealtime (prandial) dose (eg, 50% basal, 50% prandial ratio), the full dose of basal insulin can be given.3
Example: If the patient is on insulin glargine 30 U at bedtime and insulin lispro 10 U with each meal and does not have hypoglycemic episodes, then insulin glargine 30 U should be taken at bedtime.
- If the patient has hypoglycemic episodes at home, then the basal insulin can be reduced by 25%.3
Example: If the patient is on insulin glargine 30 U at bedtime and insulin glulisine 10 U with each meal (appropriate proportion of doses, similar to the example above) but has hypoglycemic episodes at home on this regimen, then only 22 U of insulin glargine should be taken at bedtime.
- If the patient’s regimen has disproportionately more basal insulin than mealtime insulin, then the total daily doses can be added and half can be given as the basal insulin.
Example: If the patient is on insulin detemir 30 U every morning at 6 am and insulin aspart 6 U with each meal and has no hypoglycemic episodes, then 24 U of insulin detemir should be taken in the morning (ie, half of the total of 30 + 6 + 6 + 6).
- In the less common scenario of diabetes managed only with basal insulin (no other diabetes injections or oral agents), then half of the dose can be given.
- If the patient is on twice-daily long-acting basal insulin, then both the dose the night before surgery and the dose the morning of surgery should be adjusted.
If the basal insulin is intermediate-acting
The intermediate-acting insulin neutral protamine Hagedorn (NPH) is usually given twice a day because of its profile (Table 1).
- On the night before surgery, the full dose of NPH insulin should be taken, unless the patient will now skip a nighttime meal because of taking nothing by mouth, in which case the dose can be decreased by 25%.1
- On the morning of surgery, since the patient will be skipping breakfast and probably also lunch, the dose should be reduced by 50%.3,4
Special situation: Premixed insulins
Premixed insulins (70/30, 75/25) are a combination of intermediate-acting insulin and either fast-acting or short-acting insulin. In other words, they are combinations of basal and prandial insulin. Their use is thus not ideal in the preoperative period. There are two options in these situations.
One option is to switch to a regimen that includes long-acting insulin. If the patient is admitted for surgery, then the hospital staff can change the insulin regimen to long-acting basal insulin. A quick formula for conversion is to add all the premixed insulin doses and give half as basal insulin on the morning of surgery, similar to the scenario above for the patient with long-acting basal insulin that was out of proportion to the prandial insulin injections.
For example, if the usual regimen is insulin 70/30 NPH/Regular, 60 U with breakfast, 30 U with dinner, then the patient can take 45 U of insulin glargine (which is half of 60 + 30) in the morning or evening before surgery.
Another option is to adjust the dose of pre-mixed insulin. Sometimes it is not feasible or economical to change the patient’s premixed insulin just before surgery. In these situations, the patient can take half of the morning dose, followed by dextrose-containing intravenous fluids and blood glucose checks.
We recommend preoperatively giving at least part of the patient’s previous basal insulin, regardless of the type of diabetes, the type of surgery, or the fasting period.
STEP 3: STOP THE PRANDIAL INSULIN
Prandial insulin—given before each meal to cover the carbohydrates to be consumed—should be stopped the morning of surgery.3,4
WHAT ABOUT SLIDING SCALE INSULIN?
Using a sliding scale alone has no known benefit. Although it can be a quick fix to correct a high glucose level, it should be added to the basal insulin and not used as the sole insulin therapy. If a sliding scale is used, fast-acting insulin (aspart, glulisine, lispro) is preferred over regular insulin because of the more rapid onset and shorter duration of action.
Patients already using a supplemental insulin scale can apply it to correct a blood glucose above 200 mg/dL on the morning of surgery.
MAINTENANCE FLUIDS
As long as glucose levels are not very elevated (ie, > 200 mg/dL), after 12 hours on a nothing-by-mouth regimen, provide dextrose in the IV fluid to prevent hypoglycemia (eg, the patient received long-acting insulin and the glucose levels are running low) or to prevent starvation ketosis, which may result in ketones in the blood or urine. We recommend 5% dextrose in half-normal (0.45%) saline at 50 to 75 mL/hour as maintenance fluid; the infusion rate should be lower if fluid overload is a concern.
POSTOPERATIVE INSULIN MANAGEMENT
Once patients are discharged and can go back to their previous routine, they can restart their usual insulin regimen the same evening. The prandial insulin will be resumed when the regular diet is reintroduced, and the doses will be adjusted according to food intake.
- Joshi GP, Chung F, Vann MA, et al; Society for Ambulatory Anesthesia. Society for Ambulatory Anesthesia consensus statement on perioperative blood glucose management in diabetic patients undergoing ambulatory surgery. Anesth Analg 2010; 111:1378–1387.
- DiNardo M, Donihi AC, Forte P, Gieraltowski L, Korytkowski M. Standardized glycemic management and perioperative glycemic outcomes in patients with diabetes mellitus who undergo same-day surgery. Endocr Pract 2011; 17:404–411.
- Vann MA. Perioperative management of ambulatory surgical patients with diabetes mellitus. Curr Opin Anaesthesiol 2009; 22:718–724.
- Meneghini LF. Perioperative management of diabetes: translating evidence into practice. Cleve Clin J Med 2009; 76(suppl 4):S53–S59.
- Joshi GP, Chung F, Vann MA, et al; Society for Ambulatory Anesthesia. Society for Ambulatory Anesthesia consensus statement on perioperative blood glucose management in diabetic patients undergoing ambulatory surgery. Anesth Analg 2010; 111:1378–1387.
- DiNardo M, Donihi AC, Forte P, Gieraltowski L, Korytkowski M. Standardized glycemic management and perioperative glycemic outcomes in patients with diabetes mellitus who undergo same-day surgery. Endocr Pract 2011; 17:404–411.
- Vann MA. Perioperative management of ambulatory surgical patients with diabetes mellitus. Curr Opin Anaesthesiol 2009; 22:718–724.
- Meneghini LF. Perioperative management of diabetes: translating evidence into practice. Cleve Clin J Med 2009; 76(suppl 4):S53–S59.
BioniCare® in the Treatment of Arthritis of the Hand and Wrist
Peter A. Holt, MD
Associate Professor of Medicine
The Johns Hopkins University School of Medicine
Baltimore, Maryland
Tuna Ozyurekoglu, MD
Christine M. Kleinert Institute for Hand and Microsurgery
Louisville, Kentucky
Shaili Deveshwar, MD
Sports Medicine and Orthopedic Center
Greensboro, North Carolina
Edmund J. MacLaughlin, MD
Cambridge, Maryland
Shirley W. Pang, MD
St. Jude Heritage Medical Group
Fullerton, California
Jack S. Tuber, DO
SunValley Arthritis Center
Peoria, Arizona
Joy Schechtman, DO
SunValley Arthritis CenterPeoria, Arizona
Thomas M. Zizic, MD
Associate Professor of Medicine
The Johns Hopkins University School of Medicine
Baltimore, Maryland
Peter A. Holt, MD
Associate Professor of Medicine
The Johns Hopkins University School of Medicine
Baltimore, Maryland
Tuna Ozyurekoglu, MD
Christine M. Kleinert Institute for Hand and Microsurgery
Louisville, Kentucky
Shaili Deveshwar, MD
Sports Medicine and Orthopedic Center
Greensboro, North Carolina
Edmund J. MacLaughlin, MD
Cambridge, Maryland
Shirley W. Pang, MD
St. Jude Heritage Medical Group
Fullerton, California
Jack S. Tuber, DO
SunValley Arthritis Center
Peoria, Arizona
Joy Schechtman, DO
SunValley Arthritis CenterPeoria, Arizona
Thomas M. Zizic, MD
Associate Professor of Medicine
The Johns Hopkins University School of Medicine
Baltimore, Maryland
Peter A. Holt, MD
Associate Professor of Medicine
The Johns Hopkins University School of Medicine
Baltimore, Maryland
Tuna Ozyurekoglu, MD
Christine M. Kleinert Institute for Hand and Microsurgery
Louisville, Kentucky
Shaili Deveshwar, MD
Sports Medicine and Orthopedic Center
Greensboro, North Carolina
Edmund J. MacLaughlin, MD
Cambridge, Maryland
Shirley W. Pang, MD
St. Jude Heritage Medical Group
Fullerton, California
Jack S. Tuber, DO
SunValley Arthritis Center
Peoria, Arizona
Joy Schechtman, DO
SunValley Arthritis CenterPeoria, Arizona
Thomas M. Zizic, MD
Associate Professor of Medicine
The Johns Hopkins University School of Medicine
Baltimore, Maryland