User login
Electronic Screen Exposure Is Associated With Migraine in Young Adults
Too much time in front of electronic screens can increase the risk of headaches and migraine in young adults, according to research published online ahead of print December 2, 2015, in Cephalalgia.
Previous studies have observed associations between screen time exposure and headaches in children between ages 10 and 12 and adolescents. Data also have found an association between screen time exposure and low-back and shoulder pain in adolescents. “This [information] had led to speculation that the high amount of screen time exposure among students of higher education institutions may be correlated with the high prevalence of headache and migraine observed in this population,” said Ilaria Montagni, PhD, a research fellow at the University of Bordeaux in Talence, France.
Dr. Montagni and her coinvestigators studied 4,927 individuals in France, all of whom were age 18 or older and part of the Internet-based Students Health Research Enterprise (i-Share) project cohort, which is an ongoing, prospective, population-based study of students at French-speaking universities and institutions of higher education. The mean age of the students was 20.8, and 75.5% were female.
Subjects completed self-reported surveys on the average amount of time they spend in front of screens during the following five activities: computer or tablet work, playing video games on a computer or tablet, Internet surfing on a computer or tablet, watching videos on a computer or tablet, and using a smartphone. All questions were scored using a five-point scale. A score of 0 indicated never, 1 indicated less than 30 minutes, 2 indicated 30 minutes to two hours, 3 indicated four to eight hours, and 5 indicated eight hours or more. Scores from the surveys were divided into quartiles of very low, low, high, and very high screen-time exposure.
Surveys also asked if participants had experienced any headaches that had lasted several hours in the previous 12 months. Participants who answered negatively were classified as “no headache,” while those who answered positively were asked a series of follow-up questions related to symptom type and severity, sensitivity to light or sound, nausea, vomiting, and disruption of daily routines. To establish a classification of migraine, the investigators used the “probable migraine” category of the International Classification of Headache Disorders, third edition. With these data, the investigators used multinomial logistic regression models to calculate odds ratios of any relationship between screen time exposure and the presence and severity of headaches.
Of the 4,927 participants, 2,773 (56.3%) reported no headaches. In all, 710 (14.4%) participants reported a nonmigraine headache, 791 (16.1%) reported migraine without aura, and 653 (13.3%) reported migraine with aura. In comparisons against very low screen time exposure, very high exposure increased the likelihood of having migraine by 37%, which resulted in a statistically significant 50% greater odds for migraine without aura, but not for migraine with aura.
“Students reporting very high screen time exposure were more likely to be male, to be older, to have higher BMI, and to consume cannabis [and] were also more likely to report nonmigraine headache or migraine,” the authors noted. Furthermore, higher exposure to screens was a significant indicator of recurrent headaches in adolescent males, and the same indicator was seen in adolescent females who spent more time on the computer and in front of the TV.
—Deepak Chitnis
Suggested Reading
Montagni I, Guichard E, Carpenet C, et al. Screen time exposure and reporting of headaches in young adults: A cross-sectional study. Cephalalgia. 2015 Dec 2 [Epub ahead of print].
Too much time in front of electronic screens can increase the risk of headaches and migraine in young adults, according to research published online ahead of print December 2, 2015, in Cephalalgia.
Previous studies have observed associations between screen time exposure and headaches in children between ages 10 and 12 and adolescents. Data also have found an association between screen time exposure and low-back and shoulder pain in adolescents. “This [information] had led to speculation that the high amount of screen time exposure among students of higher education institutions may be correlated with the high prevalence of headache and migraine observed in this population,” said Ilaria Montagni, PhD, a research fellow at the University of Bordeaux in Talence, France.
Dr. Montagni and her coinvestigators studied 4,927 individuals in France, all of whom were age 18 or older and part of the Internet-based Students Health Research Enterprise (i-Share) project cohort, which is an ongoing, prospective, population-based study of students at French-speaking universities and institutions of higher education. The mean age of the students was 20.8, and 75.5% were female.
Subjects completed self-reported surveys on the average amount of time they spend in front of screens during the following five activities: computer or tablet work, playing video games on a computer or tablet, Internet surfing on a computer or tablet, watching videos on a computer or tablet, and using a smartphone. All questions were scored using a five-point scale. A score of 0 indicated never, 1 indicated less than 30 minutes, 2 indicated 30 minutes to two hours, 3 indicated four to eight hours, and 5 indicated eight hours or more. Scores from the surveys were divided into quartiles of very low, low, high, and very high screen-time exposure.
Surveys also asked if participants had experienced any headaches that had lasted several hours in the previous 12 months. Participants who answered negatively were classified as “no headache,” while those who answered positively were asked a series of follow-up questions related to symptom type and severity, sensitivity to light or sound, nausea, vomiting, and disruption of daily routines. To establish a classification of migraine, the investigators used the “probable migraine” category of the International Classification of Headache Disorders, third edition. With these data, the investigators used multinomial logistic regression models to calculate odds ratios of any relationship between screen time exposure and the presence and severity of headaches.
Of the 4,927 participants, 2,773 (56.3%) reported no headaches. In all, 710 (14.4%) participants reported a nonmigraine headache, 791 (16.1%) reported migraine without aura, and 653 (13.3%) reported migraine with aura. In comparisons against very low screen time exposure, very high exposure increased the likelihood of having migraine by 37%, which resulted in a statistically significant 50% greater odds for migraine without aura, but not for migraine with aura.
“Students reporting very high screen time exposure were more likely to be male, to be older, to have higher BMI, and to consume cannabis [and] were also more likely to report nonmigraine headache or migraine,” the authors noted. Furthermore, higher exposure to screens was a significant indicator of recurrent headaches in adolescent males, and the same indicator was seen in adolescent females who spent more time on the computer and in front of the TV.
—Deepak Chitnis
Too much time in front of electronic screens can increase the risk of headaches and migraine in young adults, according to research published online ahead of print December 2, 2015, in Cephalalgia.
Previous studies have observed associations between screen time exposure and headaches in children between ages 10 and 12 and adolescents. Data also have found an association between screen time exposure and low-back and shoulder pain in adolescents. “This [information] had led to speculation that the high amount of screen time exposure among students of higher education institutions may be correlated with the high prevalence of headache and migraine observed in this population,” said Ilaria Montagni, PhD, a research fellow at the University of Bordeaux in Talence, France.
Dr. Montagni and her coinvestigators studied 4,927 individuals in France, all of whom were age 18 or older and part of the Internet-based Students Health Research Enterprise (i-Share) project cohort, which is an ongoing, prospective, population-based study of students at French-speaking universities and institutions of higher education. The mean age of the students was 20.8, and 75.5% were female.
Subjects completed self-reported surveys on the average amount of time they spend in front of screens during the following five activities: computer or tablet work, playing video games on a computer or tablet, Internet surfing on a computer or tablet, watching videos on a computer or tablet, and using a smartphone. All questions were scored using a five-point scale. A score of 0 indicated never, 1 indicated less than 30 minutes, 2 indicated 30 minutes to two hours, 3 indicated four to eight hours, and 5 indicated eight hours or more. Scores from the surveys were divided into quartiles of very low, low, high, and very high screen-time exposure.
Surveys also asked if participants had experienced any headaches that had lasted several hours in the previous 12 months. Participants who answered negatively were classified as “no headache,” while those who answered positively were asked a series of follow-up questions related to symptom type and severity, sensitivity to light or sound, nausea, vomiting, and disruption of daily routines. To establish a classification of migraine, the investigators used the “probable migraine” category of the International Classification of Headache Disorders, third edition. With these data, the investigators used multinomial logistic regression models to calculate odds ratios of any relationship between screen time exposure and the presence and severity of headaches.
Of the 4,927 participants, 2,773 (56.3%) reported no headaches. In all, 710 (14.4%) participants reported a nonmigraine headache, 791 (16.1%) reported migraine without aura, and 653 (13.3%) reported migraine with aura. In comparisons against very low screen time exposure, very high exposure increased the likelihood of having migraine by 37%, which resulted in a statistically significant 50% greater odds for migraine without aura, but not for migraine with aura.
“Students reporting very high screen time exposure were more likely to be male, to be older, to have higher BMI, and to consume cannabis [and] were also more likely to report nonmigraine headache or migraine,” the authors noted. Furthermore, higher exposure to screens was a significant indicator of recurrent headaches in adolescent males, and the same indicator was seen in adolescent females who spent more time on the computer and in front of the TV.
—Deepak Chitnis
Suggested Reading
Montagni I, Guichard E, Carpenet C, et al. Screen time exposure and reporting of headaches in young adults: A cross-sectional study. Cephalalgia. 2015 Dec 2 [Epub ahead of print].
Suggested Reading
Montagni I, Guichard E, Carpenet C, et al. Screen time exposure and reporting of headaches in young adults: A cross-sectional study. Cephalalgia. 2015 Dec 2 [Epub ahead of print].
Strategies for Success in Risk-Based Payment Models
Risk-based payment models are becoming increasingly common in healthcare. Programs and initiatives such as Medicare Advantage, accountable care organizations (ACOs), and bundled payments are tasking hospitals, physicians, post-acute, and other providers with better managing patient care to improve outcomes and lower costs.
According to the Kaiser Family Foundation, Medicare Advantage—through which providers accept full risk to treat patients for a fixed annual rate—grew by more than 1 million beneficiaries between March 2014 and March 2015. Today, 31%, or almost one in three Medicare beneficiaries, are enrolled in a Medicare Advantage plan.
The much newer ACO and bundled payment models are also growing quickly. And by the end of 2018, the Department of Health & Human Services would like to move more than 50% of Medicare payments into such models.
According to healthcare intelligence firm Leavitt Partners, in early 2015 there were 744 active ACOs in the U.S., up from just 64 at the same time in 2011. Those organizations, through which providers collaborate to manage the cost and quality of care for a patient population, grew by about 4.5 million covered lives between the beginning of 2014 and 2015, reaching 23.5 million participants.
The somewhat similar Bundled Payment for Care Improvement (BPCI) initiative had more than 2,100 participating providers as of August 2015, including acute-care hospitals, skilled nursing facilities (SNFs), physician group practices, long-term care hospitals, inpatient rehabilitation facilities, and home health agencies working together to assume financial risk for certain episodes of care based upon specific diagnosis-related groups (DRGs).
As this growth continues, hospitalists are uniquely positioned to drive success in risk-based payment models if they can become more “longitudinally” involved in patient care rather than focused solely on inpatient services. That means partnering with providers outside the hospital, extending their own practice beyond the hospital, and leveraging technology.
Partnering outside the Hospital
In a recent series of “On the Horizon” articles in The Hospitalist, Win Whitcomb, MD, MHM, argues that hospitalists hoping to capitalize on savings from the BPCI program must think “beyond the four walls” of the hospital to how they can impact the cost and quality of care in the post-acute setting as well as readmission rates. I couldn’t agree more.
The “buying power” hospitalists wield with their referral patterns puts them in a unique position to influence the quality of care at post-acute facilities, thereby benefiting hospitals and their patients.
Traditionally, a hospitalist discharging a patient to a SNF would not ordinarily recommend a particular facility. They likely would leave the mandated “patient choice” presentation to the case manager. And that presentation normally would consist of a list of facilities in the market, with little to no quality or cost information associated with any of the facilities. This process essentially becomes a roll of the dice as to whether the best facility is chosen for particular patients and their conditions.
Under risk-based payment models, hospitalists should consider working with SNFs to help them develop clinical protocols that reduce costly lengths of stay and hospital readmission rates. In return for improving their efficiency and care, the hospitalists can help increase referrals to those SNFs by designing a process (in partnership with the discharge-planning staff) that will provide information on cost and quality of post-discharge options for patients and families. Given the information, patients and families are much more likely to choose a facility that benefits them and, in turn, benefits the system.
Broaden Your Practice
In addition, hospitalists should consider hiring or designating personnel to help manage patients in a post-discharge environment.
In particular, HM groups are increasingly partnering with or hiring “SNFists” to manage the patients they discharge from hospitals. As the name suggests, SNFists are dedicated to treating patients in the SNF environment, just as hospitalists treat patients in the hospital. Nurse practitioners and physician assistants are playing an increasingly important role in this space.
By partnering with SNFists (internal or external to the hospital medicine practice), hospitalists can ensure better continuity of care after discharge and a higher level of care than patients typically receive in a post-acute setting. Often, SNFists are able to detect declines in a patient’s condition early enough to intervene and prevent a hospital readmission. Similarly, their supervision and communication with the hospitalist helps ensure the patient is following the discharge plan and more likely to achieve a prompt recovery.
Leveraging Technology
Though providers across healthcare settings are embracing technology systems such as electronic medical records, most continue to struggle with the lack of system interoperability for adequately sharing patient information.
On a local level, hospitalists can work with post-acute and other partnering providers to help identify what, if any, existing medical record technologies can be made to interface with one another—or if there are any adequate workarounds to facilitate the transfer of patient information and support the continuity of care post-discharge.
Other technology tools, such as telemedicine programs or remote patient monitoring, may also be options hospitalists may want to champion as a way to help manage episodes of care that extend beyond the acute-care hospital stay.
Looking Ahead
There’s no doubt risk-based payment models will continue to gain prevalence in the healthcare market. By thinking beyond the hospital, hospitalists can take a more active role in achieving success in these new models. TH
Risk-based payment models are becoming increasingly common in healthcare. Programs and initiatives such as Medicare Advantage, accountable care organizations (ACOs), and bundled payments are tasking hospitals, physicians, post-acute, and other providers with better managing patient care to improve outcomes and lower costs.
According to the Kaiser Family Foundation, Medicare Advantage—through which providers accept full risk to treat patients for a fixed annual rate—grew by more than 1 million beneficiaries between March 2014 and March 2015. Today, 31%, or almost one in three Medicare beneficiaries, are enrolled in a Medicare Advantage plan.
The much newer ACO and bundled payment models are also growing quickly. And by the end of 2018, the Department of Health & Human Services would like to move more than 50% of Medicare payments into such models.
According to healthcare intelligence firm Leavitt Partners, in early 2015 there were 744 active ACOs in the U.S., up from just 64 at the same time in 2011. Those organizations, through which providers collaborate to manage the cost and quality of care for a patient population, grew by about 4.5 million covered lives between the beginning of 2014 and 2015, reaching 23.5 million participants.
The somewhat similar Bundled Payment for Care Improvement (BPCI) initiative had more than 2,100 participating providers as of August 2015, including acute-care hospitals, skilled nursing facilities (SNFs), physician group practices, long-term care hospitals, inpatient rehabilitation facilities, and home health agencies working together to assume financial risk for certain episodes of care based upon specific diagnosis-related groups (DRGs).
As this growth continues, hospitalists are uniquely positioned to drive success in risk-based payment models if they can become more “longitudinally” involved in patient care rather than focused solely on inpatient services. That means partnering with providers outside the hospital, extending their own practice beyond the hospital, and leveraging technology.
Partnering outside the Hospital
In a recent series of “On the Horizon” articles in The Hospitalist, Win Whitcomb, MD, MHM, argues that hospitalists hoping to capitalize on savings from the BPCI program must think “beyond the four walls” of the hospital to how they can impact the cost and quality of care in the post-acute setting as well as readmission rates. I couldn’t agree more.
The “buying power” hospitalists wield with their referral patterns puts them in a unique position to influence the quality of care at post-acute facilities, thereby benefiting hospitals and their patients.
Traditionally, a hospitalist discharging a patient to a SNF would not ordinarily recommend a particular facility. They likely would leave the mandated “patient choice” presentation to the case manager. And that presentation normally would consist of a list of facilities in the market, with little to no quality or cost information associated with any of the facilities. This process essentially becomes a roll of the dice as to whether the best facility is chosen for particular patients and their conditions.
Under risk-based payment models, hospitalists should consider working with SNFs to help them develop clinical protocols that reduce costly lengths of stay and hospital readmission rates. In return for improving their efficiency and care, the hospitalists can help increase referrals to those SNFs by designing a process (in partnership with the discharge-planning staff) that will provide information on cost and quality of post-discharge options for patients and families. Given the information, patients and families are much more likely to choose a facility that benefits them and, in turn, benefits the system.
Broaden Your Practice
In addition, hospitalists should consider hiring or designating personnel to help manage patients in a post-discharge environment.
In particular, HM groups are increasingly partnering with or hiring “SNFists” to manage the patients they discharge from hospitals. As the name suggests, SNFists are dedicated to treating patients in the SNF environment, just as hospitalists treat patients in the hospital. Nurse practitioners and physician assistants are playing an increasingly important role in this space.
By partnering with SNFists (internal or external to the hospital medicine practice), hospitalists can ensure better continuity of care after discharge and a higher level of care than patients typically receive in a post-acute setting. Often, SNFists are able to detect declines in a patient’s condition early enough to intervene and prevent a hospital readmission. Similarly, their supervision and communication with the hospitalist helps ensure the patient is following the discharge plan and more likely to achieve a prompt recovery.
Leveraging Technology
Though providers across healthcare settings are embracing technology systems such as electronic medical records, most continue to struggle with the lack of system interoperability for adequately sharing patient information.
On a local level, hospitalists can work with post-acute and other partnering providers to help identify what, if any, existing medical record technologies can be made to interface with one another—or if there are any adequate workarounds to facilitate the transfer of patient information and support the continuity of care post-discharge.
Other technology tools, such as telemedicine programs or remote patient monitoring, may also be options hospitalists may want to champion as a way to help manage episodes of care that extend beyond the acute-care hospital stay.
Looking Ahead
There’s no doubt risk-based payment models will continue to gain prevalence in the healthcare market. By thinking beyond the hospital, hospitalists can take a more active role in achieving success in these new models. TH
Risk-based payment models are becoming increasingly common in healthcare. Programs and initiatives such as Medicare Advantage, accountable care organizations (ACOs), and bundled payments are tasking hospitals, physicians, post-acute, and other providers with better managing patient care to improve outcomes and lower costs.
According to the Kaiser Family Foundation, Medicare Advantage—through which providers accept full risk to treat patients for a fixed annual rate—grew by more than 1 million beneficiaries between March 2014 and March 2015. Today, 31%, or almost one in three Medicare beneficiaries, are enrolled in a Medicare Advantage plan.
The much newer ACO and bundled payment models are also growing quickly. And by the end of 2018, the Department of Health & Human Services would like to move more than 50% of Medicare payments into such models.
According to healthcare intelligence firm Leavitt Partners, in early 2015 there were 744 active ACOs in the U.S., up from just 64 at the same time in 2011. Those organizations, through which providers collaborate to manage the cost and quality of care for a patient population, grew by about 4.5 million covered lives between the beginning of 2014 and 2015, reaching 23.5 million participants.
The somewhat similar Bundled Payment for Care Improvement (BPCI) initiative had more than 2,100 participating providers as of August 2015, including acute-care hospitals, skilled nursing facilities (SNFs), physician group practices, long-term care hospitals, inpatient rehabilitation facilities, and home health agencies working together to assume financial risk for certain episodes of care based upon specific diagnosis-related groups (DRGs).
As this growth continues, hospitalists are uniquely positioned to drive success in risk-based payment models if they can become more “longitudinally” involved in patient care rather than focused solely on inpatient services. That means partnering with providers outside the hospital, extending their own practice beyond the hospital, and leveraging technology.
Partnering outside the Hospital
In a recent series of “On the Horizon” articles in The Hospitalist, Win Whitcomb, MD, MHM, argues that hospitalists hoping to capitalize on savings from the BPCI program must think “beyond the four walls” of the hospital to how they can impact the cost and quality of care in the post-acute setting as well as readmission rates. I couldn’t agree more.
The “buying power” hospitalists wield with their referral patterns puts them in a unique position to influence the quality of care at post-acute facilities, thereby benefiting hospitals and their patients.
Traditionally, a hospitalist discharging a patient to a SNF would not ordinarily recommend a particular facility. They likely would leave the mandated “patient choice” presentation to the case manager. And that presentation normally would consist of a list of facilities in the market, with little to no quality or cost information associated with any of the facilities. This process essentially becomes a roll of the dice as to whether the best facility is chosen for particular patients and their conditions.
Under risk-based payment models, hospitalists should consider working with SNFs to help them develop clinical protocols that reduce costly lengths of stay and hospital readmission rates. In return for improving their efficiency and care, the hospitalists can help increase referrals to those SNFs by designing a process (in partnership with the discharge-planning staff) that will provide information on cost and quality of post-discharge options for patients and families. Given the information, patients and families are much more likely to choose a facility that benefits them and, in turn, benefits the system.
Broaden Your Practice
In addition, hospitalists should consider hiring or designating personnel to help manage patients in a post-discharge environment.
In particular, HM groups are increasingly partnering with or hiring “SNFists” to manage the patients they discharge from hospitals. As the name suggests, SNFists are dedicated to treating patients in the SNF environment, just as hospitalists treat patients in the hospital. Nurse practitioners and physician assistants are playing an increasingly important role in this space.
By partnering with SNFists (internal or external to the hospital medicine practice), hospitalists can ensure better continuity of care after discharge and a higher level of care than patients typically receive in a post-acute setting. Often, SNFists are able to detect declines in a patient’s condition early enough to intervene and prevent a hospital readmission. Similarly, their supervision and communication with the hospitalist helps ensure the patient is following the discharge plan and more likely to achieve a prompt recovery.
Leveraging Technology
Though providers across healthcare settings are embracing technology systems such as electronic medical records, most continue to struggle with the lack of system interoperability for adequately sharing patient information.
On a local level, hospitalists can work with post-acute and other partnering providers to help identify what, if any, existing medical record technologies can be made to interface with one another—or if there are any adequate workarounds to facilitate the transfer of patient information and support the continuity of care post-discharge.
Other technology tools, such as telemedicine programs or remote patient monitoring, may also be options hospitalists may want to champion as a way to help manage episodes of care that extend beyond the acute-care hospital stay.
Looking Ahead
There’s no doubt risk-based payment models will continue to gain prevalence in the healthcare market. By thinking beyond the hospital, hospitalists can take a more active role in achieving success in these new models. TH
Study suggests chidamide could treat rel/ref CTCL too

Photo by Larry Young
SAN FRANCISCO—The oral histone deacetylase inhibitor chidamide can elicit responses in patients with relapsed or refractory cutaneous T-cell Lymphoma (CTCL), a new study suggests.
Chidamide has already demonstrated efficacy against relapsed or refractory peripheral T-cell lymphoma and has been approved for this indication in China.
Now, results of a phase 2 trial suggest chidamide might be a feasible treatment option for relapsed or refractory CTCL as well.
Yuankai Shi, MD, PhD, of the Chinese Academy of Medical Science & Peking Union Medical College in Beijing, China, discussed this trial at the 8th Annual T-cell Lymphoma Forum. The trial is sponsored by Chipscreen Biosciences Ltd.
He presented results observed in 50 patients with relapsed/refractory CTCL. They had a median age of 47 (range, 26-75), and half were male. Most patients had stage II disease (44%), 20% percent had stage III, and 18% each had stage I and IV. The median time from diagnosis was 2 years.
Patients were randomized to receive chidamide at 30 mg twice a week for 2 weeks out of a 3-week cycle (n=12), 4 weeks out of a 6-week cycle (n=13), or 30 mg twice a week without a drug-free holiday (n=25).
The objective response rate was 32% for the entire cohort, 33% for the 3-week cycle arm, 23% for the 6-week cycle arm, and 36% for the successive dosing arm.
There was 1 complete response, and it occurred in the successive dosing arm. There were 15 partial responses—4 in the 3-week arm, 3 in the 6-week arm, and 9 in the successive dosing arm.
The median duration of response was 92 days overall (range, 78-106), 50 days in the 3-week arm (range, 26-130), 92 days in the 6-week arm (range, 84-99), and 169 days in the successive dosing arm (range, 58-279).
The median progression-free survival was 85 days overall (range, 78-92), 84 days in the 3-week arm (range, 43-126), 81 days in the 6-week arm (range, 39-222), and 88 days in the successive dosing arm (range, 58-261).
Dr Shi noted that the major toxicities associated with chidamide were hematologic and gastrointestinal in nature, and they were controllable.
The incidence of adverse events (AEs) was 83% overall, 92% in the 3-week arm, 85% in the 6-week arm, and 77% in the successive dosing arm. The incidence of grade 3 or higher AEs was 23%, 23%, 38%, and 15%, respectively.
The most common AEs were thrombocytopenia (33%, 39%, 23%, and 35%, respectively), leucopenia (29%, 54%, 31%, and 15%, respectively), fatigue (17%, 23%, 23%, and 12%, respectively), nausea (13%, 23%, 8%, and 12%, respectively), diarrhea (10%, 8%, 8%, and 12%, respectively), fever (8%, 0%, 15%, and 8%, respectively), and anemia (8%, 8%, 15%, and 4%, respectively).
There were 2 serious AEs. One patient in the 3-week arm was hospitalized for fever and lung infection, and 1 patient in the successive dosing arm was hospitalized for hyperglycemia.
Dr Shi said these results suggest chidamide is effective and tolerable for patients with relapsed/refractory CTCL. And, based on the overall profiles of the 3 dosing regimens, successive dosing of chidamide at 30 mg twice a week is recommended. ![]()

Photo by Larry Young
SAN FRANCISCO—The oral histone deacetylase inhibitor chidamide can elicit responses in patients with relapsed or refractory cutaneous T-cell Lymphoma (CTCL), a new study suggests.
Chidamide has already demonstrated efficacy against relapsed or refractory peripheral T-cell lymphoma and has been approved for this indication in China.
Now, results of a phase 2 trial suggest chidamide might be a feasible treatment option for relapsed or refractory CTCL as well.
Yuankai Shi, MD, PhD, of the Chinese Academy of Medical Science & Peking Union Medical College in Beijing, China, discussed this trial at the 8th Annual T-cell Lymphoma Forum. The trial is sponsored by Chipscreen Biosciences Ltd.
He presented results observed in 50 patients with relapsed/refractory CTCL. They had a median age of 47 (range, 26-75), and half were male. Most patients had stage II disease (44%), 20% percent had stage III, and 18% each had stage I and IV. The median time from diagnosis was 2 years.
Patients were randomized to receive chidamide at 30 mg twice a week for 2 weeks out of a 3-week cycle (n=12), 4 weeks out of a 6-week cycle (n=13), or 30 mg twice a week without a drug-free holiday (n=25).
The objective response rate was 32% for the entire cohort, 33% for the 3-week cycle arm, 23% for the 6-week cycle arm, and 36% for the successive dosing arm.
There was 1 complete response, and it occurred in the successive dosing arm. There were 15 partial responses—4 in the 3-week arm, 3 in the 6-week arm, and 9 in the successive dosing arm.
The median duration of response was 92 days overall (range, 78-106), 50 days in the 3-week arm (range, 26-130), 92 days in the 6-week arm (range, 84-99), and 169 days in the successive dosing arm (range, 58-279).
The median progression-free survival was 85 days overall (range, 78-92), 84 days in the 3-week arm (range, 43-126), 81 days in the 6-week arm (range, 39-222), and 88 days in the successive dosing arm (range, 58-261).
Dr Shi noted that the major toxicities associated with chidamide were hematologic and gastrointestinal in nature, and they were controllable.
The incidence of adverse events (AEs) was 83% overall, 92% in the 3-week arm, 85% in the 6-week arm, and 77% in the successive dosing arm. The incidence of grade 3 or higher AEs was 23%, 23%, 38%, and 15%, respectively.
The most common AEs were thrombocytopenia (33%, 39%, 23%, and 35%, respectively), leucopenia (29%, 54%, 31%, and 15%, respectively), fatigue (17%, 23%, 23%, and 12%, respectively), nausea (13%, 23%, 8%, and 12%, respectively), diarrhea (10%, 8%, 8%, and 12%, respectively), fever (8%, 0%, 15%, and 8%, respectively), and anemia (8%, 8%, 15%, and 4%, respectively).
There were 2 serious AEs. One patient in the 3-week arm was hospitalized for fever and lung infection, and 1 patient in the successive dosing arm was hospitalized for hyperglycemia.
Dr Shi said these results suggest chidamide is effective and tolerable for patients with relapsed/refractory CTCL. And, based on the overall profiles of the 3 dosing regimens, successive dosing of chidamide at 30 mg twice a week is recommended. ![]()

Photo by Larry Young
SAN FRANCISCO—The oral histone deacetylase inhibitor chidamide can elicit responses in patients with relapsed or refractory cutaneous T-cell Lymphoma (CTCL), a new study suggests.
Chidamide has already demonstrated efficacy against relapsed or refractory peripheral T-cell lymphoma and has been approved for this indication in China.
Now, results of a phase 2 trial suggest chidamide might be a feasible treatment option for relapsed or refractory CTCL as well.
Yuankai Shi, MD, PhD, of the Chinese Academy of Medical Science & Peking Union Medical College in Beijing, China, discussed this trial at the 8th Annual T-cell Lymphoma Forum. The trial is sponsored by Chipscreen Biosciences Ltd.
He presented results observed in 50 patients with relapsed/refractory CTCL. They had a median age of 47 (range, 26-75), and half were male. Most patients had stage II disease (44%), 20% percent had stage III, and 18% each had stage I and IV. The median time from diagnosis was 2 years.
Patients were randomized to receive chidamide at 30 mg twice a week for 2 weeks out of a 3-week cycle (n=12), 4 weeks out of a 6-week cycle (n=13), or 30 mg twice a week without a drug-free holiday (n=25).
The objective response rate was 32% for the entire cohort, 33% for the 3-week cycle arm, 23% for the 6-week cycle arm, and 36% for the successive dosing arm.
There was 1 complete response, and it occurred in the successive dosing arm. There were 15 partial responses—4 in the 3-week arm, 3 in the 6-week arm, and 9 in the successive dosing arm.
The median duration of response was 92 days overall (range, 78-106), 50 days in the 3-week arm (range, 26-130), 92 days in the 6-week arm (range, 84-99), and 169 days in the successive dosing arm (range, 58-279).
The median progression-free survival was 85 days overall (range, 78-92), 84 days in the 3-week arm (range, 43-126), 81 days in the 6-week arm (range, 39-222), and 88 days in the successive dosing arm (range, 58-261).
Dr Shi noted that the major toxicities associated with chidamide were hematologic and gastrointestinal in nature, and they were controllable.
The incidence of adverse events (AEs) was 83% overall, 92% in the 3-week arm, 85% in the 6-week arm, and 77% in the successive dosing arm. The incidence of grade 3 or higher AEs was 23%, 23%, 38%, and 15%, respectively.
The most common AEs were thrombocytopenia (33%, 39%, 23%, and 35%, respectively), leucopenia (29%, 54%, 31%, and 15%, respectively), fatigue (17%, 23%, 23%, and 12%, respectively), nausea (13%, 23%, 8%, and 12%, respectively), diarrhea (10%, 8%, 8%, and 12%, respectively), fever (8%, 0%, 15%, and 8%, respectively), and anemia (8%, 8%, 15%, and 4%, respectively).
There were 2 serious AEs. One patient in the 3-week arm was hospitalized for fever and lung infection, and 1 patient in the successive dosing arm was hospitalized for hyperglycemia.
Dr Shi said these results suggest chidamide is effective and tolerable for patients with relapsed/refractory CTCL. And, based on the overall profiles of the 3 dosing regimens, successive dosing of chidamide at 30 mg twice a week is recommended. ![]()
When loved ones get cancer, people turn to the Web

chemotherapy
Photo by Rhoda Baer
Loved ones of cancer patients often turn to the Internet for further information about the disease, but they are less inclined to seek emotional support from social media forums, according to a study published in Computers, Informatics, Nursing.
It is fairly common for loved ones of cancer patients to develop depression or anxiety disorders as a result of the diagnosis, but there aren’t many studies focusing specifically on cancer patients’ caregivers and family members, said study author Carolyn Lauckner, PhD, of the University of Georgia in Athens.
“I think, sometimes, the loved ones and caregivers get forgotten about,” she said. “And that’s why I wanted to research this population to see if there are ways that we can better support these individuals.”
Dr Lauckner surveyed 191 people whose loved ones were diagnosed with cancer in the past year or who were currently acting as caregivers to someone with cancer. The motivation behind the research was personal for Dr Lauckner.
“I went through a period of time where I had 3 loved ones diagnosed within a short amount of time,” she said. “I had these experiences where I heard about the diagnosis and I would go online to look it up, and then I would immediately become terrified and freak out about all the stuff I read online.”
Like Dr Lauckner, more than 75% of the subjects she surveyed searched online for information on a loved one’s disease. Most looked for treatment options, prevention strategies and risk factors, and prognosis information.
“I was pleasantly surprised by the amount of people who said that they were looking for prevention information online and detection information,” Dr Lauckner said. “[T]hat shows that not only are they concerned for their loved one but they’re also concerned about how they themselves can avoid cancer, which, from a public health perspective, is great.”
Respondents were less inclined to view blogs or go online to hear about others’ cancer experiences. These kinds of sites were linked to negative emotions for participants, such as fear, sadness, and anger.
“A lot of people, especially in the cancer realm, they will use blogs or discussion posts to vent and to talk about the harsh realities of living with an illness,” Dr Lauckner said.
“And while I think that that is beneficial for both the person who is writing it and potentially for some people who want an idea of what to expect, when someone is dealing with the prospect of their loved one having to go through that experience, it can be extremely distressing.”
The most commonly visited websites were those of charitable organizations like the American Cancer Society, which were associated with positive emotions. Dr Lauckner said she found this information encouraging because it shows that participants were consulting reliable sources of information and not being swayed by personal accounts as much.
Dr Lauckner ultimately wants to build on the information gleaned in this study to determine the most effective use of social media and technology to distribute cancer prevention and risk reduction messages to the public. ![]()

chemotherapy
Photo by Rhoda Baer
Loved ones of cancer patients often turn to the Internet for further information about the disease, but they are less inclined to seek emotional support from social media forums, according to a study published in Computers, Informatics, Nursing.
It is fairly common for loved ones of cancer patients to develop depression or anxiety disorders as a result of the diagnosis, but there aren’t many studies focusing specifically on cancer patients’ caregivers and family members, said study author Carolyn Lauckner, PhD, of the University of Georgia in Athens.
“I think, sometimes, the loved ones and caregivers get forgotten about,” she said. “And that’s why I wanted to research this population to see if there are ways that we can better support these individuals.”
Dr Lauckner surveyed 191 people whose loved ones were diagnosed with cancer in the past year or who were currently acting as caregivers to someone with cancer. The motivation behind the research was personal for Dr Lauckner.
“I went through a period of time where I had 3 loved ones diagnosed within a short amount of time,” she said. “I had these experiences where I heard about the diagnosis and I would go online to look it up, and then I would immediately become terrified and freak out about all the stuff I read online.”
Like Dr Lauckner, more than 75% of the subjects she surveyed searched online for information on a loved one’s disease. Most looked for treatment options, prevention strategies and risk factors, and prognosis information.
“I was pleasantly surprised by the amount of people who said that they were looking for prevention information online and detection information,” Dr Lauckner said. “[T]hat shows that not only are they concerned for their loved one but they’re also concerned about how they themselves can avoid cancer, which, from a public health perspective, is great.”
Respondents were less inclined to view blogs or go online to hear about others’ cancer experiences. These kinds of sites were linked to negative emotions for participants, such as fear, sadness, and anger.
“A lot of people, especially in the cancer realm, they will use blogs or discussion posts to vent and to talk about the harsh realities of living with an illness,” Dr Lauckner said.
“And while I think that that is beneficial for both the person who is writing it and potentially for some people who want an idea of what to expect, when someone is dealing with the prospect of their loved one having to go through that experience, it can be extremely distressing.”
The most commonly visited websites were those of charitable organizations like the American Cancer Society, which were associated with positive emotions. Dr Lauckner said she found this information encouraging because it shows that participants were consulting reliable sources of information and not being swayed by personal accounts as much.
Dr Lauckner ultimately wants to build on the information gleaned in this study to determine the most effective use of social media and technology to distribute cancer prevention and risk reduction messages to the public. ![]()

chemotherapy
Photo by Rhoda Baer
Loved ones of cancer patients often turn to the Internet for further information about the disease, but they are less inclined to seek emotional support from social media forums, according to a study published in Computers, Informatics, Nursing.
It is fairly common for loved ones of cancer patients to develop depression or anxiety disorders as a result of the diagnosis, but there aren’t many studies focusing specifically on cancer patients’ caregivers and family members, said study author Carolyn Lauckner, PhD, of the University of Georgia in Athens.
“I think, sometimes, the loved ones and caregivers get forgotten about,” she said. “And that’s why I wanted to research this population to see if there are ways that we can better support these individuals.”
Dr Lauckner surveyed 191 people whose loved ones were diagnosed with cancer in the past year or who were currently acting as caregivers to someone with cancer. The motivation behind the research was personal for Dr Lauckner.
“I went through a period of time where I had 3 loved ones diagnosed within a short amount of time,” she said. “I had these experiences where I heard about the diagnosis and I would go online to look it up, and then I would immediately become terrified and freak out about all the stuff I read online.”
Like Dr Lauckner, more than 75% of the subjects she surveyed searched online for information on a loved one’s disease. Most looked for treatment options, prevention strategies and risk factors, and prognosis information.
“I was pleasantly surprised by the amount of people who said that they were looking for prevention information online and detection information,” Dr Lauckner said. “[T]hat shows that not only are they concerned for their loved one but they’re also concerned about how they themselves can avoid cancer, which, from a public health perspective, is great.”
Respondents were less inclined to view blogs or go online to hear about others’ cancer experiences. These kinds of sites were linked to negative emotions for participants, such as fear, sadness, and anger.
“A lot of people, especially in the cancer realm, they will use blogs or discussion posts to vent and to talk about the harsh realities of living with an illness,” Dr Lauckner said.
“And while I think that that is beneficial for both the person who is writing it and potentially for some people who want an idea of what to expect, when someone is dealing with the prospect of their loved one having to go through that experience, it can be extremely distressing.”
The most commonly visited websites were those of charitable organizations like the American Cancer Society, which were associated with positive emotions. Dr Lauckner said she found this information encouraging because it shows that participants were consulting reliable sources of information and not being swayed by personal accounts as much.
Dr Lauckner ultimately wants to build on the information gleaned in this study to determine the most effective use of social media and technology to distribute cancer prevention and risk reduction messages to the public. ![]()
Bile acid supports production of HSCs, team says

Photo by Åsa Hansdotter
Experiments in pregnant mice indicate that bile acid supports the production of hematopoietic stem cells (HSCs) in fetuses.
The work revealed large amounts of bile acids inside mouse fetuses and suggested these acids are transferred from the mothers via the placenta to help the fetuses produce HSCs.
“Fetuses produce small amounts of bile acids on their own, but here we are talking about much larger quantities,” said study author Kenichi Miharada, PhD, of Lund University in Sweden.
“The bile acid appears to be produced by the mother and then transferred to the fetus via the placenta.”
Dr Miharada and his colleagues detailed this discovery in Cell Stem Cell.
The investigators already knew that bile acid is produced in the fetal liver, but they did not know why.
With this study, they discovered that bile acid supports the production of HSCs in the fetal liver and enables them to develop normally. The additional contribution from the mother is important for the fetus to develop normally.
And although a large part of bile acid is toxic for cells, it undergoes a purification process when transferred through the placenta, letting only harmless bile acid through to the fetus.
“Our hypothesis is that the consequence of a damaged placenta, which, for various reasons, is unable to transfer bile acids to the fetus, can lead to leukemia or other blood diseases later in life, and we will continue our research to see if this hypothesis holds up,” Dr Miharada said.
He and his colleagues also said this work has implications for producing HSCs that could treat these blood diseases.
The problem with making HSCs proliferate outside the body is that the artificial growth gives rise to an accumulation of abnormal proteins in the endoplasmic reticulum (ER). This ER stress, if severe and chronic, causes cell death.
Dr Miharada and his colleagues previously showed it is possible to reduce ER stress chemically by adding bile acids to the cell culture. Bile acids, which are produced naturally in the liver and stored in the gallbladder, support protein production during the cell division process.
“Compared to other ways of trying to develop stem cells to treat blood diseases, this method is safer and quicker because it does not involve using any artificial substances or any genetic modifications, merely a substance that already exists inside the body,” Dr Miharada explained. ![]()

Photo by Åsa Hansdotter
Experiments in pregnant mice indicate that bile acid supports the production of hematopoietic stem cells (HSCs) in fetuses.
The work revealed large amounts of bile acids inside mouse fetuses and suggested these acids are transferred from the mothers via the placenta to help the fetuses produce HSCs.
“Fetuses produce small amounts of bile acids on their own, but here we are talking about much larger quantities,” said study author Kenichi Miharada, PhD, of Lund University in Sweden.
“The bile acid appears to be produced by the mother and then transferred to the fetus via the placenta.”
Dr Miharada and his colleagues detailed this discovery in Cell Stem Cell.
The investigators already knew that bile acid is produced in the fetal liver, but they did not know why.
With this study, they discovered that bile acid supports the production of HSCs in the fetal liver and enables them to develop normally. The additional contribution from the mother is important for the fetus to develop normally.
And although a large part of bile acid is toxic for cells, it undergoes a purification process when transferred through the placenta, letting only harmless bile acid through to the fetus.
“Our hypothesis is that the consequence of a damaged placenta, which, for various reasons, is unable to transfer bile acids to the fetus, can lead to leukemia or other blood diseases later in life, and we will continue our research to see if this hypothesis holds up,” Dr Miharada said.
He and his colleagues also said this work has implications for producing HSCs that could treat these blood diseases.
The problem with making HSCs proliferate outside the body is that the artificial growth gives rise to an accumulation of abnormal proteins in the endoplasmic reticulum (ER). This ER stress, if severe and chronic, causes cell death.
Dr Miharada and his colleagues previously showed it is possible to reduce ER stress chemically by adding bile acids to the cell culture. Bile acids, which are produced naturally in the liver and stored in the gallbladder, support protein production during the cell division process.
“Compared to other ways of trying to develop stem cells to treat blood diseases, this method is safer and quicker because it does not involve using any artificial substances or any genetic modifications, merely a substance that already exists inside the body,” Dr Miharada explained. ![]()

Photo by Åsa Hansdotter
Experiments in pregnant mice indicate that bile acid supports the production of hematopoietic stem cells (HSCs) in fetuses.
The work revealed large amounts of bile acids inside mouse fetuses and suggested these acids are transferred from the mothers via the placenta to help the fetuses produce HSCs.
“Fetuses produce small amounts of bile acids on their own, but here we are talking about much larger quantities,” said study author Kenichi Miharada, PhD, of Lund University in Sweden.
“The bile acid appears to be produced by the mother and then transferred to the fetus via the placenta.”
Dr Miharada and his colleagues detailed this discovery in Cell Stem Cell.
The investigators already knew that bile acid is produced in the fetal liver, but they did not know why.
With this study, they discovered that bile acid supports the production of HSCs in the fetal liver and enables them to develop normally. The additional contribution from the mother is important for the fetus to develop normally.
And although a large part of bile acid is toxic for cells, it undergoes a purification process when transferred through the placenta, letting only harmless bile acid through to the fetus.
“Our hypothesis is that the consequence of a damaged placenta, which, for various reasons, is unable to transfer bile acids to the fetus, can lead to leukemia or other blood diseases later in life, and we will continue our research to see if this hypothesis holds up,” Dr Miharada said.
He and his colleagues also said this work has implications for producing HSCs that could treat these blood diseases.
The problem with making HSCs proliferate outside the body is that the artificial growth gives rise to an accumulation of abnormal proteins in the endoplasmic reticulum (ER). This ER stress, if severe and chronic, causes cell death.
Dr Miharada and his colleagues previously showed it is possible to reduce ER stress chemically by adding bile acids to the cell culture. Bile acids, which are produced naturally in the liver and stored in the gallbladder, support protein production during the cell division process.
“Compared to other ways of trying to develop stem cells to treat blood diseases, this method is safer and quicker because it does not involve using any artificial substances or any genetic modifications, merely a substance that already exists inside the body,” Dr Miharada explained. ![]()
Group identifies SNPs associated with MBD
New research has revealed single-nucleotide polymorphisms (SNPs) that may increase the risk of bone disease in patients with multiple myeloma (MM).
One of these SNPs affects a gene encoding osteoprotegerin (OPG), a signaling protein that helps regulate the density of bones.
In patients with MM bone disease (MBD), bones are broken down faster than they can be repaired, resulting in fractures and lesions.
OPG is known to keep the numbers of osteoclasts in check, so it follows that mutations adversely affecting the gene may contribute to excessive bone breakdown.
Richard Houlston, MD, PhD, of The Institute of Cancer Research in London, UK, and his colleagues reported these findings in Leukemia.
The researchers analyzed the DNA of 3774 MM patients and used imaging techniques to detect which of these patients had MBD and which did not. The team used statistical analysis to compare sequencing data from the group of patients who developed MBD with those who did not.
The analysis revealed 9 SNPs that showed an association with MBD and reached genome-wide significance. They were all located in the same region at 8q24.12 and were in strong linkage disequilibrium (LD).
The strongest association at 8q24.12 was observed with the SNP rs4407910, which localizes 19Kb 3’ to the gene encoding TNFRSF11B, also known as OPG (odds ratio=1.38, P=4.02×10−9).
The researchers also found a “promising association” for MBD with the SNP rs74676832 at 19q13.43, which is located within a 29 kb region of LD intergenic to ZNF444 and GALP (odds ratio=1.97, P=9.33×10–7).
The team said these findings demonstrate that germline variation influences MBD, and they highlight the importance of the RANK/RANKL/OPG pathway in MBD development.
Furthermore, the work could enable scientists to develop new strategies to prevent MBD development and identify which patients might benefit from treatment with existing bone therapies such as bisphosphonates.
“It is really important to understand why some patients with myeloma are severely affected by bone disease,” Dr Houlston said. “This study tells us that at least part of the answer is in the patient’s DNA, greatly improving our understanding of the disease.”
“We want to be able to identify which patients are most at risk and which treatments are most likely to help them. This study gives us clues that can begin to help us do this. In the longer term, understanding the factors that contributed to bone breakdown could help us find new treatments for the disease.” ![]()

New research has revealed single-nucleotide polymorphisms (SNPs) that may increase the risk of bone disease in patients with multiple myeloma (MM).
One of these SNPs affects a gene encoding osteoprotegerin (OPG), a signaling protein that helps regulate the density of bones.
In patients with MM bone disease (MBD), bones are broken down faster than they can be repaired, resulting in fractures and lesions.
OPG is known to keep the numbers of osteoclasts in check, so it follows that mutations adversely affecting the gene may contribute to excessive bone breakdown.
Richard Houlston, MD, PhD, of The Institute of Cancer Research in London, UK, and his colleagues reported these findings in Leukemia.
The researchers analyzed the DNA of 3774 MM patients and used imaging techniques to detect which of these patients had MBD and which did not. The team used statistical analysis to compare sequencing data from the group of patients who developed MBD with those who did not.
The analysis revealed 9 SNPs that showed an association with MBD and reached genome-wide significance. They were all located in the same region at 8q24.12 and were in strong linkage disequilibrium (LD).
The strongest association at 8q24.12 was observed with the SNP rs4407910, which localizes 19Kb 3’ to the gene encoding TNFRSF11B, also known as OPG (odds ratio=1.38, P=4.02×10−9).
The researchers also found a “promising association” for MBD with the SNP rs74676832 at 19q13.43, which is located within a 29 kb region of LD intergenic to ZNF444 and GALP (odds ratio=1.97, P=9.33×10–7).
The team said these findings demonstrate that germline variation influences MBD, and they highlight the importance of the RANK/RANKL/OPG pathway in MBD development.
Furthermore, the work could enable scientists to develop new strategies to prevent MBD development and identify which patients might benefit from treatment with existing bone therapies such as bisphosphonates.
“It is really important to understand why some patients with myeloma are severely affected by bone disease,” Dr Houlston said. “This study tells us that at least part of the answer is in the patient’s DNA, greatly improving our understanding of the disease.”
“We want to be able to identify which patients are most at risk and which treatments are most likely to help them. This study gives us clues that can begin to help us do this. In the longer term, understanding the factors that contributed to bone breakdown could help us find new treatments for the disease.” ![]()

New research has revealed single-nucleotide polymorphisms (SNPs) that may increase the risk of bone disease in patients with multiple myeloma (MM).
One of these SNPs affects a gene encoding osteoprotegerin (OPG), a signaling protein that helps regulate the density of bones.
In patients with MM bone disease (MBD), bones are broken down faster than they can be repaired, resulting in fractures and lesions.
OPG is known to keep the numbers of osteoclasts in check, so it follows that mutations adversely affecting the gene may contribute to excessive bone breakdown.
Richard Houlston, MD, PhD, of The Institute of Cancer Research in London, UK, and his colleagues reported these findings in Leukemia.
The researchers analyzed the DNA of 3774 MM patients and used imaging techniques to detect which of these patients had MBD and which did not. The team used statistical analysis to compare sequencing data from the group of patients who developed MBD with those who did not.
The analysis revealed 9 SNPs that showed an association with MBD and reached genome-wide significance. They were all located in the same region at 8q24.12 and were in strong linkage disequilibrium (LD).
The strongest association at 8q24.12 was observed with the SNP rs4407910, which localizes 19Kb 3’ to the gene encoding TNFRSF11B, also known as OPG (odds ratio=1.38, P=4.02×10−9).
The researchers also found a “promising association” for MBD with the SNP rs74676832 at 19q13.43, which is located within a 29 kb region of LD intergenic to ZNF444 and GALP (odds ratio=1.97, P=9.33×10–7).
The team said these findings demonstrate that germline variation influences MBD, and they highlight the importance of the RANK/RANKL/OPG pathway in MBD development.
Furthermore, the work could enable scientists to develop new strategies to prevent MBD development and identify which patients might benefit from treatment with existing bone therapies such as bisphosphonates.
“It is really important to understand why some patients with myeloma are severely affected by bone disease,” Dr Houlston said. “This study tells us that at least part of the answer is in the patient’s DNA, greatly improving our understanding of the disease.”
“We want to be able to identify which patients are most at risk and which treatments are most likely to help them. This study gives us clues that can begin to help us do this. In the longer term, understanding the factors that contributed to bone breakdown could help us find new treatments for the disease.” ![]()
Readmission Analysis Using Fault Tree
As physicians strive to increase the value of healthcare delivery, there has been increased focus on improving the quality of care that patients receive while lowering per capita costs. A provision of the Affordable Care Act implemented in 2012 identified all‐cause 30‐day readmission rates as a measure of hospital quality, and as part of the Act's Hospital Readmission and Reduction Program, Medicare now penalizes hospitals with higher than expected all‐cause readmissions rates for adult patients with certain conditions by lowering reimbursements.[1] Although readmissions are not yet commonly used to determine reimbursements for pediatric hospitals, several states are penalizing higher than expected readmission rates for Medicaid enrollees,[2, 3] using an imprecise algorithm to determine which readmissions resulted from low‐quality care during the index admission.[4, 5, 6]
There is growing concern, however, that readmission rates are not an accurate gauge of the quality of care patients receive while in the hospital or during the discharge process to prepare them for their transition home.[7, 8, 9, 10] This is especially true in pediatric settings, where overall readmission rates are much lower than in adult settings, many readmissions are expected as part of a patient's planned course of care, and variation in readmission rates between hospitals is correlated with the percentage of patients with certain complex chronic conditions.[1, 7, 11] Thus, there is increasing agreement that hospitals and external evaluators need to shift the focus from all‐cause readmissions to a reliable, consistent, and fair measure of potentially preventable readmissions.[12, 13] In addition to being a more useful quality metric, analyzing preventable readmissions will help hospitals focus resources on patients with potentially modifiable risk factors and develop meaningful quality‐improvement initiatives to improve inpatient care as well as the discharge process to prepare families for their transition to home.[14]
Although previous studies have attempted to distinguish preventable from nonpreventable readmissions, many reported significant challenges in completing reviews efficiently, achieving consistency in how readmissions were classified, and attaining consensus on final determinations.[12, 13, 14] Studies have also demonstrated that the algorithms some states are using to streamline preventability reviews and determine reimbursements overestimate the rate of potentially preventable readmissions.[4, 5, 6]
To increase the efficiency of preventability reviews and reduce the subjectivity involved in reaching final determinations, while still accounting for the nuances necessary to conduct a fair review, a quality‐improvement team from the Division of General Pediatrics at The Children's Hospital of Philadelphia (CHOP) implemented a fault tree analysis tool based on a framework developed by Howard Parker at Intermountain Primary Children's Hospital. The CHOP team coded this framework into a secure Web‐based data‐collection tool in the form of a decision tree to guide reviewers through a logical progression of questions that result in 1 of 18 root causes of readmissions, 8 of which are considered potentially preventable. We hypothesized that this method would help reviewers efficiently reach consensus on the root causes of hospital readmissions, and thus help the division and the hospital focus efforts on developing relevant quality‐improvement initiatives.
METHODS
Inclusion Criteria and Study Design
This study was conducted at CHOP, a 535‐bed urban, tertiary‐care, freestanding children's hospital with approximately 29,000 annual discharges. Of those discharges, 7000 to 8000 are from the general pediatrics service, meaning that the attending of record was a general pediatrician. Patients were included in the study if (1) they were discharged from the general pediatrics service between January 2014 and December 2014, and (2) they were readmitted to the hospital, for any reason, within 15 days of discharge. Because this analysis was done as part of a quality‐improvement initiative, it focuses on 15‐day, early readmissions to target cases with a higher probability of being potentially preventable from the perspective of the hospital care team.[10, 12, 13] Patients under observation status during the index admission or the readmission were included. However, patients who returned to the emergency department but were not admitted to an inpatient unit were excluded. Objective details about each case, including the patient's name, demographics, chart number, and diagnosis code, were pre‐loaded from EPIC (Epic Systems Corp., Verona, WI) into REDCap (Research Electronic Data Capture;
A panel of 10 general pediatricians divided up the cases to perform retrospective chart reviews. For each case, REDCap guided reviewers through the fault tree analysis. Reviewers met monthly to discuss difficult cases and reach consensus on any identified ambiguities in the process. After all cases were reviewed once, 3 panel members independently reviewed a random selection of cases to measure inter‐rater reliability and confirm reproducibility of final determinations. The inter‐rater reliability statistic was calculated using Stata 12.1 (StataCorp LP, College Station, TX). During chart reviews, panel members were not blinded to the identity of physicians and other staff members caring for the patients under review. CHOP's institutional review board determined this study to be exempt from ongoing review.
Fault Tree Analysis
Using the decision tree framework for analyzing readmissions that was developed at Intermountain Primary Children's Hospital, the REDCap tool prompted reviewers with a series of sequential questions, each with mutually exclusive options. Using imbedded branching logic to select follow‐up questions, the tool guided reviewers to 1 of 18 terminal nodes, each representing a potential root cause of the readmission. Of those 18 potential causes, 8 were considered potentially preventable. A diagram of the fault tree framework, color coded to indicate which nodes were considered potentially preventable, is shown in Figure 1.
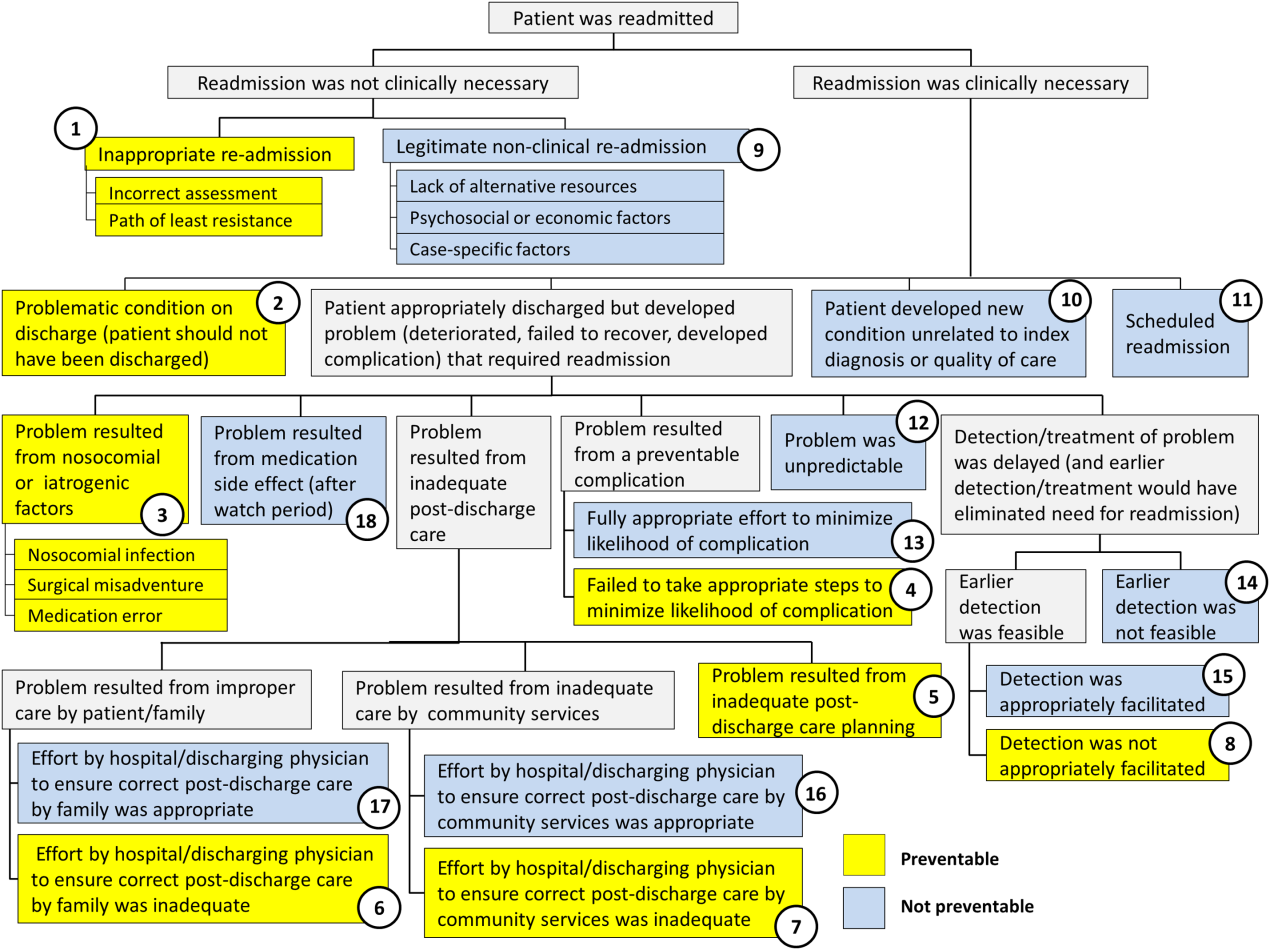
RESULTS
In 2014, 7252 patients were discharged from the general pediatrics service at CHOP. Of those patients, 248 were readmitted within 15 days for an overall general pediatrics 15‐day readmission rate of 3.4%.
Preventability Analysis
Of the 248 readmissions, 233 (94.0%) were considered not preventable. The most common cause for readmission, which accounted for 145 cases (58.5%), was a patient developing an unpredictable problem related to the index diagnosis or a natural progression of the disease that required readmission. The second most common cause, which accounted for 53 cases (21.4%), was a patient developing a new condition unrelated to the index diagnosis or a readmission unrelated to the quality of care received during the index stay. The third most frequent cause, which accounted for 11 cases (4.4%), was a legitimate nonclinical readmission due to lack of alternative resources, psychosocial or economic factors, or case‐specific factors. Other nonpreventable causes of readmission, including scheduled readmissions, each accounted for 7 or fewer cases and <3% of total readmissions.
The 15 readmissions considered potentially preventable accounted for 6.0% of total readmissions and 0.2% of total discharges from the general pediatrics service in 2014. The most common cause of preventable readmissions, which accounted for 6 cases, was premature discharge. The second most common cause, which accounted for 4 cases, was a problem resulting from nosocomial or iatrogenic factors. Other potentially preventable causes included delayed detection of problem (3 cases), inappropriate readmission (1 case), and inadequate postdischarge care planning (1 case).
A breakdown of fault tree results, including examples of cases associated with each terminal node, is shown in Table 1. Information about general pediatrics patients and readmitted patients is included in Tables 2 and 3. A breakdown of determinations for each reviewer is included in Supporting Table 1 in the online version of this article.
| Fault Tree Terminal Node | Root Cause of Readmission | No. of Cases | % of Total Readmissions | % Within Preventability Category | % of Total Discharges |
|---|---|---|---|---|---|
| |||||
| 2 (Potentially Preventable) | Problematic condition on discharge. Example:* Index admission: Infant with history of prematurity admitted with RSV and rhinovirus bronchiolitis. Had some waxing and waning symptoms. Just prior to discharge, noted to have increased work of breathing related to feeds. Readmission: 12 hours later with tachypnea, retractions, and hypoxia. | 6 | 2.4% | 40.0% | 0.08% |
| 3 (Potentially Preventable) | Nosocomial/Iatrogenic factors. Example*: Index admission: Toddler admitted with fever and neutropenia. Treated with antibiotics 24 hours. Diagnosed with viral illness and discharged home. Readmission: symptomatic Clostridum difficile infection. | 4 | 1.6% | 26.7% | 0.06% |
| 8 (Potentially Preventable) | Detection/treatment of problem was delayed and not appropriately facilitated. Example:* Index admission: Preteen admitted with abdominal pain, concern for appendicitis. Ultrasound and abdominal MRI negative for appendicitis. Symptoms improved. Tolerated PO. Readmission: 3 days later with similar abdominal pain. Diagnosed with constipation with significant improvement following clean‐out. | 3 | 1.2% | 20.0% | 0.04% |
| 1 (Potentially Preventable) | Inappropriate readmission. Example:* Index admission: Infant with laryngomalacia admitted with bronchiolitis. Readmission: Continued mild bronchiolitis symptoms but did not require oxygen or suctioning, normal CXR. | 1 | 0.4% | 6.7% | 0.01% |
| 5 (Potentially Preventable) | Resulted from inadequate postdischarge care planning. Example:* Index diagnosis: Infant with vomiting, prior admissions, and extensive evaluation, diagnosed with milk protein allergy and GERD. PPI increased. Readmission: Persistent symptoms, required NGT feeds supplementation. | 1 | 0.4% | 6.7% | 0.01% |
| 4 (Potentially Preventable) | Resulted from a preventable complication and hospital/physician did not take the appropriate steps to minimize likelihood of complication. | ||||
| 6 (Potentially Preventable) | Resulted from improper care by patient/family and effort by hospital/physician to ensure correct postdischarge care was inadequate. | ||||
| 7 (Potentially Preventable) | Resulted from inadequate care by community services and effort by hospital/physician to ensure correct postdischarge care was inadequate. | ||||
| 15 | 6.0% | 100% | 0.2% | ||
| 12 (Not Preventable) | Problem was unpredictable. Example:* Index admission: Infant admitted with gastroenteritis and dehydration with an anion gap metabolic acidosis. Vomiting and diarrhea improved, rehydrated, acidosis improved. Readmission: 1 day later, presented with emesis and fussiness. Readmitted for metabolic acidosis. | 145 | 58.5% | 62.2% | 2.00% |
| 10 (Not Preventable) | Patient developed new condition unrelated to index diagnosis or quality of care. Example:* Index admission: Toddler admitted with cellulitis. Readmission: Bronchiolitis (did not meet CDC guidelines for nosocomial infection). | 53 | 21.4% | 22.7% | 0.73% |
| 9 (Not Preventable) | Legitimate nonclinical readmission. Example:* Index admission: Infant admitted with second episode of bronchiolitis. Readmission: 4 days later with mild diarrhea. Tolerated PO challenge in emergency department. Admitted due to parental anxiety. | 11 | 4.4% | 4.7% | 0.15% |
| 17 (Not Preventable) | Problem resulted from improper care by patient/family but effort by hospital/physician to ensure correct postdischarge care was appropriate. Example:* Index admission: Infant admitted with diarrhea, diagnosed with milk protein allergy. Discharged on soy formula. Readmission: Developed vomiting and diarrhea with cow milk formula. | 7 | 2.8% | 3.0% | 0.10% |
| 11 (Not Preventable) | Scheduled readmission. Example:* Index admission: Infant with conjunctivitis and preseptal cellulitis with nasolacrimal duct obstruction. Readmission: Postoperatively following scheduled nasolacrimal duct repair. | 7 | 2.8% | 3.0% | 0.10% |
| 14 (Not Preventable) | Detection/treatment of problem was delayed, but earlier detection was not feasible. Example:* Index admission: Preteen admitted with fever, abdominal pain, and elevated inflammatory markers. Fever resolved and symptoms improved. Diagnosed with unspecified viral infection. Readmission: 4 days later with lower extremity pyomyositis and possible osteomyelitis. | 4 | 1.6% | 1.7% | 0.06% |
| 15 (Not Preventable) | Detection/treatment of problem was delayed, earlier detection was feasible, but detection was appropriately facilitated. Example:* Index admission: Infant with history of laryngomalacia and GER admitted with an ALTE. No events during hospitalization. Appropriate workup and cleared by consultants for discharge. Zantac increased. Readmission: Infant had similar ALTE events within a week after discharge. Ultimately underwent supraglottoplasty. | 2 | 0.8% | 0.9% | 0.03% |
| 13 (Not Preventable) | Resulted from preventable complication but efforts to minimize likelihood were appropriate. Example:* Index admission: Patient on GJ feeds admitted for dislodged GJ. Extensive conversations between primary team and multiple consulting services regarding best type of tube. Determined that no other tube options were appropriate. Temporizing measures were initiated. Readmission: GJ tube dislodged again. | 2 | 0.8% | 0.9% | 0.03% |
| 18 (Not Preventable) | Resulted from medication side effect (after watch period). Example:* Index admission: Preteen with MSSA bacteremia spread to other organs. Sent home on appropriate IV antibiotics. Readmission: Fever, rash, increased LFTs. Blood cultures negative. Presumed drug reaction. Fevers resolved with alternate medication. | 2 | 0.8% | 0.9% | 0.03% |
| 16 (Not Preventable) | Resulted from inadequate care by community services, but effort by hospital/physician to ensure correct postdischarge care was appropriate. | ||||
| 233 | 94.0% | 100% | 3.2% | ||
| Fault Tree Terminal Node | Root Cause of Potentially Preventable Readmission with Case Descriptions* |
|---|---|
| |
| 2 (Potentially Preventable) | Problematic condition on discharge |
| Case 1: Index admission: Infant with history of prematurity admitted with RSV and rhinovirus bronchiolitis. Had some waxing and waning symptoms. Just prior to discharge, noted to have increased work of breathing related to feeds. Readmission: 12 hours later with tachypnea, retractions, and hypoxia. | |
| Case 2: Index admission: Toddler admitted with febrile seizure in setting of gastroenteritis. Poor PO intake during hospitalization. Readmission: 1 day later with dehydration. | |
| Case 3: Index admission: Infant admitted with a prolonged complex febrile seizure. Workup included an unremarkable lumbar puncture. No additional seizures. No inpatient imaging obtained. Readmission: Abnormal outpatient MRI requiring intervention. | |
| Case 4: Index admission: Teenager with wheezing and history of chronic daily symptoms. Discharged <24 hours later on albuterol every 4 hours and prednisone. Readmission: 1 day later, seen by primary care physician with persistent asthma flare. | |
| Case 5: Index admission: Exfull‐term infant admitted with bronchiolitis, early in course. At time of discharge, had been off oxygen for 24 hours, but last recorded respiratory rate was >70. Readmission: 1 day later due to continued tachypnea and increased work of breathing. No hypoxia. CXR normal. | |
| Case 6: Exfull‐term infant admitted with bilious emesis, diarrhea, and dehydration. Ultrasound of pylorus, UGI, and BMP all normal. Tolerated oral intake but had emesis and loose stools prior to discharge. Readmission: <48 hours later with severe metabolic acidosis. | |
| 3 (Potentially Preventable) | Nosocomial/ematrogenic factors |
| Case 1: Index admission: Toddler admitted with fever and neutropenia. Treated with antibiotics 24 hours. Diagnosed with viral illness and discharged home. Readmission: Symptomatic Clostridum difficile infection. | |
| Case 2: Index admission: Patient with autism admitted with viral gastroenteritis. Readmission: Presumed nosocominal upper respiratory infection. | |
| Case 3: Index admission: Infant admitted with bronchiolitis. Recovered from initial infection. Readmission: New upper respiratory infection and presumed nosocomial infection. | |
| Case 4: Index admission: <28‐day‐old full‐term neonate presenting with neonatal fever and rash. Full septic workup performed and all cultures negative at 24 hours. Readmission: CSF culture positive at 36 hours and readmitted while awaiting speciation. Discharged once culture grew out a contaminant. | |
| 8 (Potentially Preventable) | Detection/treatment of problem was delayed and/or not appropriately facilitated |
| Case 1: Index admission: Preteen admitted with abdominal pain, concern for appendicitis. Ultrasound and MRI abdomen negative for appendicitis. Symptoms improved. Tolerated PO. Readmission: 3 days later with similar abdominal pain. Diagnosed with constipation with significant improvement following clean‐out. | |
| Case 2: Index admission: Infant with history of macrocephaly presented with fever and full fontanelle. Head CT showed mild prominence of the extra‐axial space, and lumbar puncture was normal. Readmission: Patient developed torticollis. MRI demonstrated a malignant lesion. | |
| Case 3: Index admission: School‐age child with RLQ abdominal pain, fever, leukocytosis, and indeterminate RLQ abdominal ultrasound. Twelve‐hour observation with no further fevers. Pain and appetite improved. Readmission: 1 day later with fever, anorexia, and abdominal pain. RLQ ultrasound unchanged. Appendectomy performed with inflamed appendix. | |
| 1 (Potentially Preventable) | Inappropriate readmission |
| Case 1: Index admission: Infant with laryngomalacia admitted with bronchiolitis. Readmission: Continued mild bronchiolitis symptoms but did not require oxygen or suctioning. Normal CXR. | |
| 5 (Potentially Preventable) | Resulted from inadequate postdischarge care planning |
| Case 1: Index diagnosis: Infant with vomiting, prior admissions, and extensive evaluation, diagnosed with milk protein allergy and GERD. PPI increased. Readmission: Persistent symptoms, required NGT feeds supplementation. | |
| All General Pediatrics Patients in 2014 | General Pediatric Readmitted Patients in 2014 | ||||
|---|---|---|---|---|---|
| Major Diagnosis Category at Index Admission | No. | % | Major Diagnosis Category at Index Admission | No. | % |
| |||||
| Respiratory | 2,723 | 37.5% | Respiratory | 79 | 31.9% |
| Digestive | 748 | 10.3% | Digestive | 41 | 16.5% |
| Ear, nose, mouth, throat | 675 | 9.3% | Ear, nose, mouth, throat | 24 | 9.7% |
| Skin, subcutaneous tissue | 480 | 6.6% | Musculoskeletal and connective tissue | 14 | 5.6% |
| Infectious, parasitic, systemic | 455 | 6.3% | Nervous | 13 | 5.2% |
| Factors influencing health status | 359 | 5.0% | Endocrine, nutritional, metabolic | 13 | 5.2% |
| Endocrine, nutritional, metabolic | 339 | 4.7% | Infectious, parasitic, systemic | 12 | 4.8% |
| Nervous | 239 | 3.3% | Newborn, neonate, perinatal period | 11 | 4.4% |
| Musculoskeletal and connective tissue | 228 | 3.1% | Hepatobiliary system and pancreas | 8 | 3.2% |
| Newborn, neonate, perinatal period | 206 | 2.8% | Skin, subcutaneous tissue | 8 | 3.2% |
| Other* | 800 | 11.0% | Other | 25 | 10.1% |
| Total | 7,252 | 100% | Total | 248 | 100% |
Inter‐Rater Reliability Analysis
A random selection of 50 cases (20% of total readmissions) was selected for a second review to test the tool's inter‐rater reliability. The second review resulted in the same terminal node for 44 (86%) of the cross‐checked files ( = 0.79; 95% confidence interval: 0.60‐0.98). Of the 6 cross‐checked files that ended at different nodes, 5 resulted in the same final determination about preventability. Only 1 of the cross‐checks (2% of total cross‐checked files) resulted in a different conclusion about preventability.
Efficiency Analysis
Reviewers reported that using the tool to reach a determination about preventability took approximately 20 minutes per case. Thus, initial reviews on the 248 cases required approximately 82.6 reviewer hours. Divided across 10 reviewers, this resulted in 8 to 9 hours of review time per reviewer over the year.
DISCUSSION
As part of an effort to direct quality‐improvement initiatives, this project used a Web‐based fault tree tool to identify root causes of general pediatrics readmissions at a freestanding children's hospital and classify them as either preventable or not preventable. The project also investigated the efficiency and inter‐rater reliability of the tool, which was designed to systematically guide physicians through the chart review process to a final determination about preventability. The project confirmed that using the tool helped reviewers reach final determinations about preventability efficiently with a high degree of consistency. It also confirmed that only a very small percentage of general pediatrics 15‐day readmissions are potentially preventable. Specifically, potentially preventable readmissions accounted for only 6.0% of total readmissions and 0.2% of general pediatrics discharges in 2014. Although our analysis focused on 15‐day readmissions, the fault tree methodology can be applied to any timeframe.
Previous studies attempting to distinguish preventable from nonpreventable readmissions, which used a range of methodologies to reach final determinations, reported that their review process was both time intensive and highly subjective. One study, which had 4 reviewers independently review charts and assign each case a preventability score on a 5‐point Likert scale, reported that reviewers disagreed on the final determination in 62.5% of cases.[12] Another study had 2 physicians independently review a selection of cases and assign a preventability score on a scale from 0 to 3. Scores for the 2 reviewers were added together, and cases above a certain composite threshold were classified as preventable. Despite being time‐intensive, this method resulted in only moderate agreement among physicians about the likelihood of preventability (weighted statistic of 0.44).[14] A more recent study, in which 2 physicians independently classified readmissions into 1 of 4 predefined categories, also reported only moderate agreement between reviewers ( = 0.44).[13] Other methods that have been reported include classifying readmissions as preventable only if multiple reviewers independently agreed, and using a third reviewer as a tie‐breaker.[14]
In an attempt to identify potentially preventable readmissions without using chart reviews, 3M (St. Paul, MN) developed its Potentially Preventable Readmissions software (3M‐PPR), which uses administrative data to identify which readmissions were potentially preventable. Although this automated approach is less time intensive, evidence suggests that due to a lack of nuance, the algorithm significantly overestimates the percentage of readmissions that are potentially preventable.[4, 5] A study that used 3M‐PPR to assess 1.7 million hospitalizations across 58 children's hospitals found that the algorithm classified 81% of sickle cell crisis and asthma readmissions, and 83% of bronchiolitis readmissions as potentially preventable.[10, 11] However, many readmissions for asthma and bronchiolitis are due to social factors that are outside of a hospital's direct control,[4, 5] and at many hospitals, readmissions for sickle cell crisis are part of a high‐value care model that weighs length of stay against potential readmissions. In addition, when assessing readmissions 7, 15, and 30 days after discharge, the algorithm classified almost the same percentage as potentially preventable, which is inconsistent with the notion that readmissions are more likely to have been preventable if they occurred closer to the initial discharge.[4, 13] Another study that assessed the performance of the software in the adult population reported that the algorithm performed with 85% sensitivity, but only 28% specificity.[5, 6]
The results of this quality‐improvement project indicate that using the fault tree tool to guide physicians through the chart review process helped address some of the shortcomings of methods reported in previous studies, by increasing the efficiency and reducing the subjectivity of final determinations, while still accounting for the nuances necessary to conduct a fair review. Because the tool provided a systematic framework for reviews, each case was completed in approximately 20 minutes, and because the process was the same for all reviewers, inter‐rater reliability was extremely high. In 86% of cross‐checked cases, the second reviewer ended at the same terminal node in the decision tree as the original reviewer, and in 98% of cross‐checked cases the second reviewer reached the same conclusion about preventability, even if they did not end at the same terminal node. Even accounting for agreement due to chance, the statistic of 0.79 confirmed that there was substantial agreement among reviewers about final determinations. Because the tool is easily adaptable, other hospitals can adopt this framework for their own preventability reviews and quality‐improvement initiatives.
Using the fault tree tool to access root causes of all 15‐day general pediatric readmissions helped the division focus quality‐improvement efforts on the most common causes of potentially preventable readmissions. Because 40% of potentially preventable readmissions were due to premature discharges, this prompted quality‐improvement teams to focus efforts on improving and clarifying the division's discharge criteria and clinical pathways. The division also initiated processes to improve discharge planning, including improved teaching of discharge instructions and having families pick up prescriptions prior to discharge.
Although these results did help the division identify a few areas of focus to potentially reduce readmissions, the fact that the overall 15‐day readmission rate for general pediatrics, as well as the percentage of readmissions and total discharges that were deemed potentially preventable, were so low (3.4%, 6.0%, and 0.2%, respectively), supports those who question whether prioritizing pediatric readmissions is the best place for hospitals to focus quality‐improvement efforts.[10, 12, 15, 16] As these results indicate, most pediatric readmissions are not preventable, and thus consistent with an efficient, effective, timely, patient‐centered, and equitable health system. Other studies have also shown that because overall and condition‐specific readmissions at pediatric hospitals are low, few pediatric hospitals are high or low performing for readmissions, and thus readmission rates are likely not a good measure of hospital quality.[8]
However, other condition‐specific studies of readmissions in pediatrics have indicated that there are some areas of opportunity to identify populations at high risk for readmission. One study found that although pneumonia‐specific 30‐day readmission rates in a national cohort of children hospitalized with pneumonia was only 3.1%, the chances of readmission were higher for children <1 year old, children with chronic comorbidities or complicated pneumonia, and children cared for in hospitals with lower volumes of pneumonia admissions.[17] Another study found that 17.1% of adolescents in a statewide database were readmitted post‐tonsillectomy for pain, nausea, and dehydration.[18] Thus, adapting the tool to identify root causes of condition‐specific or procedure‐specific readmissions, especially for surgical patients, may be an area of opportunity for future quality‐improvement efforts.[5] However, for general pediatrics, shifting the focus from reducing readmissions to improving the quality of care patients receive in the hospital, improving the discharge process, and adopting a population health approach to mitigate external risk factors, may be appropriate.
This project was subject to limitations. First, because it was conducted at a single site and only on general pediatrics patients, results may not be generalizable to other hospitals or other pediatric divisions. Thus, future studies might use the fault tree framework to assess preventability of pediatric readmissions in other divisions or specialties. Second, because readmissions to other hospitals were not included in the sample, the overall readmissions rate is likely underestimated.[19] However, it is unclear how this would affect the rate of potentially preventable readmissions. Third, although the fault tree framework reduced the subjectivity of the review process, there is still a degree of subjectivity inherent at each decision node. To minimize this, reviewers should try to discuss and come to consensus on how they are making determinations at each juncture in the decision tree. Similarly, because reviewers' answers to decision‐tree questions rely heavily on chart documentation, reviews may be compromised by unclear or incomplete documentation. For example, if information about steps the hospital team took to prepare a family for discharge were not properly documented, it would be difficult to determine whether appropriate steps were taken to minimize the likelihood of a complication. In the case of insufficient documentation of relevant social concerns, cases may be incorrectly classified as preventable, because addressing social issues is often not within a hospital's direct control. Finally, because reviewers were not blinded to the original discharging physician, there may have been some unconscious bias of unknown direction in the reviews.
CONCLUSION
Using the Web‐based fault tree tool helped physicians to identify the root causes of hospital readmissions and classify them as preventable or not preventable in a standardized, efficient, and consistent way, while still accounting for the nuances necessary to conduct a fair review. Thus, other hospitals should consider adopting this framework for their own preventability reviews and quality‐improvement initiatives. However, this project also confirmed that only a very small percentage of general pediatrics 15‐day readmissions are potentially preventable, suggesting that general pediatrics readmissions are not an appropriate measure of hospital quality. Instead, adapting the tool to identify root causes of condition‐specific or procedure‐specific readmission rates may be an area of opportunity for future quality‐improvement efforts.
Disclosures: This work was supported through internal funds from The Children's Hospital of Philadelphia. The authors have no financial interests, relationships or affiliations relevant to the subject matter or materials discussed in the article to disclose. The authors have no potential conflicts of interest relevant to the subject matter or materials discussed in the article to disclose.
- , . Pediatric readmissions as a hospital quality measure. JAMA. 2013;309(4):396–398.
- Texas Health and Human Services Commission. Potentially preventable readmissions in the Texas Medicaid population, state fiscal year 2012. Available at: http://www.hhsc.state.tx.us/reports/2013/ppr‐report.pdf. Published November 2013. Accessed August 16, 2015.
- Illinois Department of Healthcare and Family Services. Quality initiative to reduce hospital potentially preventable readmissions (PPR): Status update. Available at: http://www.illinois.gov/hfs/SiteCollectionDocuments/PPRPolicyStatusUpdate.pdf. Published September 3, 2014. Accessed August 16, 2015.
- , , , et al. Rates and impact of potentially preventable readmissions at children's hospitals. J Pediatr. 2015;166(3):613–619.e615.
- , . Preventing pediatric readmissions: which ones and how? J Pediatr. 2015;166(3):519–520.
- , , , , , . Manual and automated methods for identifying potentially preventable readmissions: a comparison in a large healthcare system. BMC Med Inform Decis Mak. 2014;14:28.
- , . Section on hospital medicine leadership and staff. Hosp Pediatr. 2013;3(4):390–393.
- , , , et al. Measuring hospital quality using pediatric readmission and revisit rates. Pediatrics. 2013;132(3):429–436.
- , . Hospital readmissions—not just a measure of quality. JAMA. 2011;306(16):1796–1797.
- , . Preventing readmissions in children: how do we do that? Hosp Pediatr. 2015;5(11):602–604.
- , , , et al. Pediatric readmission prevalence and variability across hospitals. JAMA. 2013;309(4):372–380.
- , , , , , . Preventability of early readmissions at a children's hospital. Pediatrics. 2013;131(1):e171–e181.
- , , , et al. An examination of physician‐, caregiver‐, and disease‐related factors associated with readmission from a pediatric hospital medicine service. Hosp Pediatr. 2015;5(11):566–573.
- , , , et al. Clinical preventability of 30‐day readmission after percutaneous coronary intervention. J Am Heart Assoc. 2014;3(5):e001290.
- . 3M algorithm overestimates preventable pediatric readmissions. Hospitalist News website. Available at: http://www.ehospitalistnews.com/specialty‐focus/pediatrics/single‐article‐page/3m‐algorithm‐overestimates‐preventable‐pediatric‐readmissions.html. Published August 16, 2013. Accessed August 16, 2015.
- . The 30‐day readmission rate: not a quality measure but an accountability measure. An Ounce of Evidence: Health Policy blog. Available at: https://blogs.sph.harvard.edu/ashish‐jha/?s=30‐day+readmission+rate. Published February 14, 2013. Accessed August 16, 2015.
- , , , et al. Readmissions among children previously hospitalized with pneumonia. Pediatrics. 2014;134(1):100–109.
- , , . A population‐based study of acute care revisits following tonsillectomy. J Pediatr. 2015;166(3):607–612.e605.
- , , , et al. Same‐hospital readmission rates as a measure of pediatric quality of care. JAMA Pediatr. 2015;169(10):905–912.
As physicians strive to increase the value of healthcare delivery, there has been increased focus on improving the quality of care that patients receive while lowering per capita costs. A provision of the Affordable Care Act implemented in 2012 identified all‐cause 30‐day readmission rates as a measure of hospital quality, and as part of the Act's Hospital Readmission and Reduction Program, Medicare now penalizes hospitals with higher than expected all‐cause readmissions rates for adult patients with certain conditions by lowering reimbursements.[1] Although readmissions are not yet commonly used to determine reimbursements for pediatric hospitals, several states are penalizing higher than expected readmission rates for Medicaid enrollees,[2, 3] using an imprecise algorithm to determine which readmissions resulted from low‐quality care during the index admission.[4, 5, 6]
There is growing concern, however, that readmission rates are not an accurate gauge of the quality of care patients receive while in the hospital or during the discharge process to prepare them for their transition home.[7, 8, 9, 10] This is especially true in pediatric settings, where overall readmission rates are much lower than in adult settings, many readmissions are expected as part of a patient's planned course of care, and variation in readmission rates between hospitals is correlated with the percentage of patients with certain complex chronic conditions.[1, 7, 11] Thus, there is increasing agreement that hospitals and external evaluators need to shift the focus from all‐cause readmissions to a reliable, consistent, and fair measure of potentially preventable readmissions.[12, 13] In addition to being a more useful quality metric, analyzing preventable readmissions will help hospitals focus resources on patients with potentially modifiable risk factors and develop meaningful quality‐improvement initiatives to improve inpatient care as well as the discharge process to prepare families for their transition to home.[14]
Although previous studies have attempted to distinguish preventable from nonpreventable readmissions, many reported significant challenges in completing reviews efficiently, achieving consistency in how readmissions were classified, and attaining consensus on final determinations.[12, 13, 14] Studies have also demonstrated that the algorithms some states are using to streamline preventability reviews and determine reimbursements overestimate the rate of potentially preventable readmissions.[4, 5, 6]
To increase the efficiency of preventability reviews and reduce the subjectivity involved in reaching final determinations, while still accounting for the nuances necessary to conduct a fair review, a quality‐improvement team from the Division of General Pediatrics at The Children's Hospital of Philadelphia (CHOP) implemented a fault tree analysis tool based on a framework developed by Howard Parker at Intermountain Primary Children's Hospital. The CHOP team coded this framework into a secure Web‐based data‐collection tool in the form of a decision tree to guide reviewers through a logical progression of questions that result in 1 of 18 root causes of readmissions, 8 of which are considered potentially preventable. We hypothesized that this method would help reviewers efficiently reach consensus on the root causes of hospital readmissions, and thus help the division and the hospital focus efforts on developing relevant quality‐improvement initiatives.
METHODS
Inclusion Criteria and Study Design
This study was conducted at CHOP, a 535‐bed urban, tertiary‐care, freestanding children's hospital with approximately 29,000 annual discharges. Of those discharges, 7000 to 8000 are from the general pediatrics service, meaning that the attending of record was a general pediatrician. Patients were included in the study if (1) they were discharged from the general pediatrics service between January 2014 and December 2014, and (2) they were readmitted to the hospital, for any reason, within 15 days of discharge. Because this analysis was done as part of a quality‐improvement initiative, it focuses on 15‐day, early readmissions to target cases with a higher probability of being potentially preventable from the perspective of the hospital care team.[10, 12, 13] Patients under observation status during the index admission or the readmission were included. However, patients who returned to the emergency department but were not admitted to an inpatient unit were excluded. Objective details about each case, including the patient's name, demographics, chart number, and diagnosis code, were pre‐loaded from EPIC (Epic Systems Corp., Verona, WI) into REDCap (Research Electronic Data Capture;
A panel of 10 general pediatricians divided up the cases to perform retrospective chart reviews. For each case, REDCap guided reviewers through the fault tree analysis. Reviewers met monthly to discuss difficult cases and reach consensus on any identified ambiguities in the process. After all cases were reviewed once, 3 panel members independently reviewed a random selection of cases to measure inter‐rater reliability and confirm reproducibility of final determinations. The inter‐rater reliability statistic was calculated using Stata 12.1 (StataCorp LP, College Station, TX). During chart reviews, panel members were not blinded to the identity of physicians and other staff members caring for the patients under review. CHOP's institutional review board determined this study to be exempt from ongoing review.
Fault Tree Analysis
Using the decision tree framework for analyzing readmissions that was developed at Intermountain Primary Children's Hospital, the REDCap tool prompted reviewers with a series of sequential questions, each with mutually exclusive options. Using imbedded branching logic to select follow‐up questions, the tool guided reviewers to 1 of 18 terminal nodes, each representing a potential root cause of the readmission. Of those 18 potential causes, 8 were considered potentially preventable. A diagram of the fault tree framework, color coded to indicate which nodes were considered potentially preventable, is shown in Figure 1.

RESULTS
In 2014, 7252 patients were discharged from the general pediatrics service at CHOP. Of those patients, 248 were readmitted within 15 days for an overall general pediatrics 15‐day readmission rate of 3.4%.
Preventability Analysis
Of the 248 readmissions, 233 (94.0%) were considered not preventable. The most common cause for readmission, which accounted for 145 cases (58.5%), was a patient developing an unpredictable problem related to the index diagnosis or a natural progression of the disease that required readmission. The second most common cause, which accounted for 53 cases (21.4%), was a patient developing a new condition unrelated to the index diagnosis or a readmission unrelated to the quality of care received during the index stay. The third most frequent cause, which accounted for 11 cases (4.4%), was a legitimate nonclinical readmission due to lack of alternative resources, psychosocial or economic factors, or case‐specific factors. Other nonpreventable causes of readmission, including scheduled readmissions, each accounted for 7 or fewer cases and <3% of total readmissions.
The 15 readmissions considered potentially preventable accounted for 6.0% of total readmissions and 0.2% of total discharges from the general pediatrics service in 2014. The most common cause of preventable readmissions, which accounted for 6 cases, was premature discharge. The second most common cause, which accounted for 4 cases, was a problem resulting from nosocomial or iatrogenic factors. Other potentially preventable causes included delayed detection of problem (3 cases), inappropriate readmission (1 case), and inadequate postdischarge care planning (1 case).
A breakdown of fault tree results, including examples of cases associated with each terminal node, is shown in Table 1. Information about general pediatrics patients and readmitted patients is included in Tables 2 and 3. A breakdown of determinations for each reviewer is included in Supporting Table 1 in the online version of this article.
| Fault Tree Terminal Node | Root Cause of Readmission | No. of Cases | % of Total Readmissions | % Within Preventability Category | % of Total Discharges |
|---|---|---|---|---|---|
| |||||
| 2 (Potentially Preventable) | Problematic condition on discharge. Example:* Index admission: Infant with history of prematurity admitted with RSV and rhinovirus bronchiolitis. Had some waxing and waning symptoms. Just prior to discharge, noted to have increased work of breathing related to feeds. Readmission: 12 hours later with tachypnea, retractions, and hypoxia. | 6 | 2.4% | 40.0% | 0.08% |
| 3 (Potentially Preventable) | Nosocomial/Iatrogenic factors. Example*: Index admission: Toddler admitted with fever and neutropenia. Treated with antibiotics 24 hours. Diagnosed with viral illness and discharged home. Readmission: symptomatic Clostridum difficile infection. | 4 | 1.6% | 26.7% | 0.06% |
| 8 (Potentially Preventable) | Detection/treatment of problem was delayed and not appropriately facilitated. Example:* Index admission: Preteen admitted with abdominal pain, concern for appendicitis. Ultrasound and abdominal MRI negative for appendicitis. Symptoms improved. Tolerated PO. Readmission: 3 days later with similar abdominal pain. Diagnosed with constipation with significant improvement following clean‐out. | 3 | 1.2% | 20.0% | 0.04% |
| 1 (Potentially Preventable) | Inappropriate readmission. Example:* Index admission: Infant with laryngomalacia admitted with bronchiolitis. Readmission: Continued mild bronchiolitis symptoms but did not require oxygen or suctioning, normal CXR. | 1 | 0.4% | 6.7% | 0.01% |
| 5 (Potentially Preventable) | Resulted from inadequate postdischarge care planning. Example:* Index diagnosis: Infant with vomiting, prior admissions, and extensive evaluation, diagnosed with milk protein allergy and GERD. PPI increased. Readmission: Persistent symptoms, required NGT feeds supplementation. | 1 | 0.4% | 6.7% | 0.01% |
| 4 (Potentially Preventable) | Resulted from a preventable complication and hospital/physician did not take the appropriate steps to minimize likelihood of complication. | ||||
| 6 (Potentially Preventable) | Resulted from improper care by patient/family and effort by hospital/physician to ensure correct postdischarge care was inadequate. | ||||
| 7 (Potentially Preventable) | Resulted from inadequate care by community services and effort by hospital/physician to ensure correct postdischarge care was inadequate. | ||||
| 15 | 6.0% | 100% | 0.2% | ||
| 12 (Not Preventable) | Problem was unpredictable. Example:* Index admission: Infant admitted with gastroenteritis and dehydration with an anion gap metabolic acidosis. Vomiting and diarrhea improved, rehydrated, acidosis improved. Readmission: 1 day later, presented with emesis and fussiness. Readmitted for metabolic acidosis. | 145 | 58.5% | 62.2% | 2.00% |
| 10 (Not Preventable) | Patient developed new condition unrelated to index diagnosis or quality of care. Example:* Index admission: Toddler admitted with cellulitis. Readmission: Bronchiolitis (did not meet CDC guidelines for nosocomial infection). | 53 | 21.4% | 22.7% | 0.73% |
| 9 (Not Preventable) | Legitimate nonclinical readmission. Example:* Index admission: Infant admitted with second episode of bronchiolitis. Readmission: 4 days later with mild diarrhea. Tolerated PO challenge in emergency department. Admitted due to parental anxiety. | 11 | 4.4% | 4.7% | 0.15% |
| 17 (Not Preventable) | Problem resulted from improper care by patient/family but effort by hospital/physician to ensure correct postdischarge care was appropriate. Example:* Index admission: Infant admitted with diarrhea, diagnosed with milk protein allergy. Discharged on soy formula. Readmission: Developed vomiting and diarrhea with cow milk formula. | 7 | 2.8% | 3.0% | 0.10% |
| 11 (Not Preventable) | Scheduled readmission. Example:* Index admission: Infant with conjunctivitis and preseptal cellulitis with nasolacrimal duct obstruction. Readmission: Postoperatively following scheduled nasolacrimal duct repair. | 7 | 2.8% | 3.0% | 0.10% |
| 14 (Not Preventable) | Detection/treatment of problem was delayed, but earlier detection was not feasible. Example:* Index admission: Preteen admitted with fever, abdominal pain, and elevated inflammatory markers. Fever resolved and symptoms improved. Diagnosed with unspecified viral infection. Readmission: 4 days later with lower extremity pyomyositis and possible osteomyelitis. | 4 | 1.6% | 1.7% | 0.06% |
| 15 (Not Preventable) | Detection/treatment of problem was delayed, earlier detection was feasible, but detection was appropriately facilitated. Example:* Index admission: Infant with history of laryngomalacia and GER admitted with an ALTE. No events during hospitalization. Appropriate workup and cleared by consultants for discharge. Zantac increased. Readmission: Infant had similar ALTE events within a week after discharge. Ultimately underwent supraglottoplasty. | 2 | 0.8% | 0.9% | 0.03% |
| 13 (Not Preventable) | Resulted from preventable complication but efforts to minimize likelihood were appropriate. Example:* Index admission: Patient on GJ feeds admitted for dislodged GJ. Extensive conversations between primary team and multiple consulting services regarding best type of tube. Determined that no other tube options were appropriate. Temporizing measures were initiated. Readmission: GJ tube dislodged again. | 2 | 0.8% | 0.9% | 0.03% |
| 18 (Not Preventable) | Resulted from medication side effect (after watch period). Example:* Index admission: Preteen with MSSA bacteremia spread to other organs. Sent home on appropriate IV antibiotics. Readmission: Fever, rash, increased LFTs. Blood cultures negative. Presumed drug reaction. Fevers resolved with alternate medication. | 2 | 0.8% | 0.9% | 0.03% |
| 16 (Not Preventable) | Resulted from inadequate care by community services, but effort by hospital/physician to ensure correct postdischarge care was appropriate. | ||||
| 233 | 94.0% | 100% | 3.2% | ||
| Fault Tree Terminal Node | Root Cause of Potentially Preventable Readmission with Case Descriptions* |
|---|---|
| |
| 2 (Potentially Preventable) | Problematic condition on discharge |
| Case 1: Index admission: Infant with history of prematurity admitted with RSV and rhinovirus bronchiolitis. Had some waxing and waning symptoms. Just prior to discharge, noted to have increased work of breathing related to feeds. Readmission: 12 hours later with tachypnea, retractions, and hypoxia. | |
| Case 2: Index admission: Toddler admitted with febrile seizure in setting of gastroenteritis. Poor PO intake during hospitalization. Readmission: 1 day later with dehydration. | |
| Case 3: Index admission: Infant admitted with a prolonged complex febrile seizure. Workup included an unremarkable lumbar puncture. No additional seizures. No inpatient imaging obtained. Readmission: Abnormal outpatient MRI requiring intervention. | |
| Case 4: Index admission: Teenager with wheezing and history of chronic daily symptoms. Discharged <24 hours later on albuterol every 4 hours and prednisone. Readmission: 1 day later, seen by primary care physician with persistent asthma flare. | |
| Case 5: Index admission: Exfull‐term infant admitted with bronchiolitis, early in course. At time of discharge, had been off oxygen for 24 hours, but last recorded respiratory rate was >70. Readmission: 1 day later due to continued tachypnea and increased work of breathing. No hypoxia. CXR normal. | |
| Case 6: Exfull‐term infant admitted with bilious emesis, diarrhea, and dehydration. Ultrasound of pylorus, UGI, and BMP all normal. Tolerated oral intake but had emesis and loose stools prior to discharge. Readmission: <48 hours later with severe metabolic acidosis. | |
| 3 (Potentially Preventable) | Nosocomial/ematrogenic factors |
| Case 1: Index admission: Toddler admitted with fever and neutropenia. Treated with antibiotics 24 hours. Diagnosed with viral illness and discharged home. Readmission: Symptomatic Clostridum difficile infection. | |
| Case 2: Index admission: Patient with autism admitted with viral gastroenteritis. Readmission: Presumed nosocominal upper respiratory infection. | |
| Case 3: Index admission: Infant admitted with bronchiolitis. Recovered from initial infection. Readmission: New upper respiratory infection and presumed nosocomial infection. | |
| Case 4: Index admission: <28‐day‐old full‐term neonate presenting with neonatal fever and rash. Full septic workup performed and all cultures negative at 24 hours. Readmission: CSF culture positive at 36 hours and readmitted while awaiting speciation. Discharged once culture grew out a contaminant. | |
| 8 (Potentially Preventable) | Detection/treatment of problem was delayed and/or not appropriately facilitated |
| Case 1: Index admission: Preteen admitted with abdominal pain, concern for appendicitis. Ultrasound and MRI abdomen negative for appendicitis. Symptoms improved. Tolerated PO. Readmission: 3 days later with similar abdominal pain. Diagnosed with constipation with significant improvement following clean‐out. | |
| Case 2: Index admission: Infant with history of macrocephaly presented with fever and full fontanelle. Head CT showed mild prominence of the extra‐axial space, and lumbar puncture was normal. Readmission: Patient developed torticollis. MRI demonstrated a malignant lesion. | |
| Case 3: Index admission: School‐age child with RLQ abdominal pain, fever, leukocytosis, and indeterminate RLQ abdominal ultrasound. Twelve‐hour observation with no further fevers. Pain and appetite improved. Readmission: 1 day later with fever, anorexia, and abdominal pain. RLQ ultrasound unchanged. Appendectomy performed with inflamed appendix. | |
| 1 (Potentially Preventable) | Inappropriate readmission |
| Case 1: Index admission: Infant with laryngomalacia admitted with bronchiolitis. Readmission: Continued mild bronchiolitis symptoms but did not require oxygen or suctioning. Normal CXR. | |
| 5 (Potentially Preventable) | Resulted from inadequate postdischarge care planning |
| Case 1: Index diagnosis: Infant with vomiting, prior admissions, and extensive evaluation, diagnosed with milk protein allergy and GERD. PPI increased. Readmission: Persistent symptoms, required NGT feeds supplementation. | |
| All General Pediatrics Patients in 2014 | General Pediatric Readmitted Patients in 2014 | ||||
|---|---|---|---|---|---|
| Major Diagnosis Category at Index Admission | No. | % | Major Diagnosis Category at Index Admission | No. | % |
| |||||
| Respiratory | 2,723 | 37.5% | Respiratory | 79 | 31.9% |
| Digestive | 748 | 10.3% | Digestive | 41 | 16.5% |
| Ear, nose, mouth, throat | 675 | 9.3% | Ear, nose, mouth, throat | 24 | 9.7% |
| Skin, subcutaneous tissue | 480 | 6.6% | Musculoskeletal and connective tissue | 14 | 5.6% |
| Infectious, parasitic, systemic | 455 | 6.3% | Nervous | 13 | 5.2% |
| Factors influencing health status | 359 | 5.0% | Endocrine, nutritional, metabolic | 13 | 5.2% |
| Endocrine, nutritional, metabolic | 339 | 4.7% | Infectious, parasitic, systemic | 12 | 4.8% |
| Nervous | 239 | 3.3% | Newborn, neonate, perinatal period | 11 | 4.4% |
| Musculoskeletal and connective tissue | 228 | 3.1% | Hepatobiliary system and pancreas | 8 | 3.2% |
| Newborn, neonate, perinatal period | 206 | 2.8% | Skin, subcutaneous tissue | 8 | 3.2% |
| Other* | 800 | 11.0% | Other | 25 | 10.1% |
| Total | 7,252 | 100% | Total | 248 | 100% |
Inter‐Rater Reliability Analysis
A random selection of 50 cases (20% of total readmissions) was selected for a second review to test the tool's inter‐rater reliability. The second review resulted in the same terminal node for 44 (86%) of the cross‐checked files ( = 0.79; 95% confidence interval: 0.60‐0.98). Of the 6 cross‐checked files that ended at different nodes, 5 resulted in the same final determination about preventability. Only 1 of the cross‐checks (2% of total cross‐checked files) resulted in a different conclusion about preventability.
Efficiency Analysis
Reviewers reported that using the tool to reach a determination about preventability took approximately 20 minutes per case. Thus, initial reviews on the 248 cases required approximately 82.6 reviewer hours. Divided across 10 reviewers, this resulted in 8 to 9 hours of review time per reviewer over the year.
DISCUSSION
As part of an effort to direct quality‐improvement initiatives, this project used a Web‐based fault tree tool to identify root causes of general pediatrics readmissions at a freestanding children's hospital and classify them as either preventable or not preventable. The project also investigated the efficiency and inter‐rater reliability of the tool, which was designed to systematically guide physicians through the chart review process to a final determination about preventability. The project confirmed that using the tool helped reviewers reach final determinations about preventability efficiently with a high degree of consistency. It also confirmed that only a very small percentage of general pediatrics 15‐day readmissions are potentially preventable. Specifically, potentially preventable readmissions accounted for only 6.0% of total readmissions and 0.2% of general pediatrics discharges in 2014. Although our analysis focused on 15‐day readmissions, the fault tree methodology can be applied to any timeframe.
Previous studies attempting to distinguish preventable from nonpreventable readmissions, which used a range of methodologies to reach final determinations, reported that their review process was both time intensive and highly subjective. One study, which had 4 reviewers independently review charts and assign each case a preventability score on a 5‐point Likert scale, reported that reviewers disagreed on the final determination in 62.5% of cases.[12] Another study had 2 physicians independently review a selection of cases and assign a preventability score on a scale from 0 to 3. Scores for the 2 reviewers were added together, and cases above a certain composite threshold were classified as preventable. Despite being time‐intensive, this method resulted in only moderate agreement among physicians about the likelihood of preventability (weighted statistic of 0.44).[14] A more recent study, in which 2 physicians independently classified readmissions into 1 of 4 predefined categories, also reported only moderate agreement between reviewers ( = 0.44).[13] Other methods that have been reported include classifying readmissions as preventable only if multiple reviewers independently agreed, and using a third reviewer as a tie‐breaker.[14]
In an attempt to identify potentially preventable readmissions without using chart reviews, 3M (St. Paul, MN) developed its Potentially Preventable Readmissions software (3M‐PPR), which uses administrative data to identify which readmissions were potentially preventable. Although this automated approach is less time intensive, evidence suggests that due to a lack of nuance, the algorithm significantly overestimates the percentage of readmissions that are potentially preventable.[4, 5] A study that used 3M‐PPR to assess 1.7 million hospitalizations across 58 children's hospitals found that the algorithm classified 81% of sickle cell crisis and asthma readmissions, and 83% of bronchiolitis readmissions as potentially preventable.[10, 11] However, many readmissions for asthma and bronchiolitis are due to social factors that are outside of a hospital's direct control,[4, 5] and at many hospitals, readmissions for sickle cell crisis are part of a high‐value care model that weighs length of stay against potential readmissions. In addition, when assessing readmissions 7, 15, and 30 days after discharge, the algorithm classified almost the same percentage as potentially preventable, which is inconsistent with the notion that readmissions are more likely to have been preventable if they occurred closer to the initial discharge.[4, 13] Another study that assessed the performance of the software in the adult population reported that the algorithm performed with 85% sensitivity, but only 28% specificity.[5, 6]
The results of this quality‐improvement project indicate that using the fault tree tool to guide physicians through the chart review process helped address some of the shortcomings of methods reported in previous studies, by increasing the efficiency and reducing the subjectivity of final determinations, while still accounting for the nuances necessary to conduct a fair review. Because the tool provided a systematic framework for reviews, each case was completed in approximately 20 minutes, and because the process was the same for all reviewers, inter‐rater reliability was extremely high. In 86% of cross‐checked cases, the second reviewer ended at the same terminal node in the decision tree as the original reviewer, and in 98% of cross‐checked cases the second reviewer reached the same conclusion about preventability, even if they did not end at the same terminal node. Even accounting for agreement due to chance, the statistic of 0.79 confirmed that there was substantial agreement among reviewers about final determinations. Because the tool is easily adaptable, other hospitals can adopt this framework for their own preventability reviews and quality‐improvement initiatives.
Using the fault tree tool to access root causes of all 15‐day general pediatric readmissions helped the division focus quality‐improvement efforts on the most common causes of potentially preventable readmissions. Because 40% of potentially preventable readmissions were due to premature discharges, this prompted quality‐improvement teams to focus efforts on improving and clarifying the division's discharge criteria and clinical pathways. The division also initiated processes to improve discharge planning, including improved teaching of discharge instructions and having families pick up prescriptions prior to discharge.
Although these results did help the division identify a few areas of focus to potentially reduce readmissions, the fact that the overall 15‐day readmission rate for general pediatrics, as well as the percentage of readmissions and total discharges that were deemed potentially preventable, were so low (3.4%, 6.0%, and 0.2%, respectively), supports those who question whether prioritizing pediatric readmissions is the best place for hospitals to focus quality‐improvement efforts.[10, 12, 15, 16] As these results indicate, most pediatric readmissions are not preventable, and thus consistent with an efficient, effective, timely, patient‐centered, and equitable health system. Other studies have also shown that because overall and condition‐specific readmissions at pediatric hospitals are low, few pediatric hospitals are high or low performing for readmissions, and thus readmission rates are likely not a good measure of hospital quality.[8]
However, other condition‐specific studies of readmissions in pediatrics have indicated that there are some areas of opportunity to identify populations at high risk for readmission. One study found that although pneumonia‐specific 30‐day readmission rates in a national cohort of children hospitalized with pneumonia was only 3.1%, the chances of readmission were higher for children <1 year old, children with chronic comorbidities or complicated pneumonia, and children cared for in hospitals with lower volumes of pneumonia admissions.[17] Another study found that 17.1% of adolescents in a statewide database were readmitted post‐tonsillectomy for pain, nausea, and dehydration.[18] Thus, adapting the tool to identify root causes of condition‐specific or procedure‐specific readmissions, especially for surgical patients, may be an area of opportunity for future quality‐improvement efforts.[5] However, for general pediatrics, shifting the focus from reducing readmissions to improving the quality of care patients receive in the hospital, improving the discharge process, and adopting a population health approach to mitigate external risk factors, may be appropriate.
This project was subject to limitations. First, because it was conducted at a single site and only on general pediatrics patients, results may not be generalizable to other hospitals or other pediatric divisions. Thus, future studies might use the fault tree framework to assess preventability of pediatric readmissions in other divisions or specialties. Second, because readmissions to other hospitals were not included in the sample, the overall readmissions rate is likely underestimated.[19] However, it is unclear how this would affect the rate of potentially preventable readmissions. Third, although the fault tree framework reduced the subjectivity of the review process, there is still a degree of subjectivity inherent at each decision node. To minimize this, reviewers should try to discuss and come to consensus on how they are making determinations at each juncture in the decision tree. Similarly, because reviewers' answers to decision‐tree questions rely heavily on chart documentation, reviews may be compromised by unclear or incomplete documentation. For example, if information about steps the hospital team took to prepare a family for discharge were not properly documented, it would be difficult to determine whether appropriate steps were taken to minimize the likelihood of a complication. In the case of insufficient documentation of relevant social concerns, cases may be incorrectly classified as preventable, because addressing social issues is often not within a hospital's direct control. Finally, because reviewers were not blinded to the original discharging physician, there may have been some unconscious bias of unknown direction in the reviews.
CONCLUSION
Using the Web‐based fault tree tool helped physicians to identify the root causes of hospital readmissions and classify them as preventable or not preventable in a standardized, efficient, and consistent way, while still accounting for the nuances necessary to conduct a fair review. Thus, other hospitals should consider adopting this framework for their own preventability reviews and quality‐improvement initiatives. However, this project also confirmed that only a very small percentage of general pediatrics 15‐day readmissions are potentially preventable, suggesting that general pediatrics readmissions are not an appropriate measure of hospital quality. Instead, adapting the tool to identify root causes of condition‐specific or procedure‐specific readmission rates may be an area of opportunity for future quality‐improvement efforts.
Disclosures: This work was supported through internal funds from The Children's Hospital of Philadelphia. The authors have no financial interests, relationships or affiliations relevant to the subject matter or materials discussed in the article to disclose. The authors have no potential conflicts of interest relevant to the subject matter or materials discussed in the article to disclose.
As physicians strive to increase the value of healthcare delivery, there has been increased focus on improving the quality of care that patients receive while lowering per capita costs. A provision of the Affordable Care Act implemented in 2012 identified all‐cause 30‐day readmission rates as a measure of hospital quality, and as part of the Act's Hospital Readmission and Reduction Program, Medicare now penalizes hospitals with higher than expected all‐cause readmissions rates for adult patients with certain conditions by lowering reimbursements.[1] Although readmissions are not yet commonly used to determine reimbursements for pediatric hospitals, several states are penalizing higher than expected readmission rates for Medicaid enrollees,[2, 3] using an imprecise algorithm to determine which readmissions resulted from low‐quality care during the index admission.[4, 5, 6]
There is growing concern, however, that readmission rates are not an accurate gauge of the quality of care patients receive while in the hospital or during the discharge process to prepare them for their transition home.[7, 8, 9, 10] This is especially true in pediatric settings, where overall readmission rates are much lower than in adult settings, many readmissions are expected as part of a patient's planned course of care, and variation in readmission rates between hospitals is correlated with the percentage of patients with certain complex chronic conditions.[1, 7, 11] Thus, there is increasing agreement that hospitals and external evaluators need to shift the focus from all‐cause readmissions to a reliable, consistent, and fair measure of potentially preventable readmissions.[12, 13] In addition to being a more useful quality metric, analyzing preventable readmissions will help hospitals focus resources on patients with potentially modifiable risk factors and develop meaningful quality‐improvement initiatives to improve inpatient care as well as the discharge process to prepare families for their transition to home.[14]
Although previous studies have attempted to distinguish preventable from nonpreventable readmissions, many reported significant challenges in completing reviews efficiently, achieving consistency in how readmissions were classified, and attaining consensus on final determinations.[12, 13, 14] Studies have also demonstrated that the algorithms some states are using to streamline preventability reviews and determine reimbursements overestimate the rate of potentially preventable readmissions.[4, 5, 6]
To increase the efficiency of preventability reviews and reduce the subjectivity involved in reaching final determinations, while still accounting for the nuances necessary to conduct a fair review, a quality‐improvement team from the Division of General Pediatrics at The Children's Hospital of Philadelphia (CHOP) implemented a fault tree analysis tool based on a framework developed by Howard Parker at Intermountain Primary Children's Hospital. The CHOP team coded this framework into a secure Web‐based data‐collection tool in the form of a decision tree to guide reviewers through a logical progression of questions that result in 1 of 18 root causes of readmissions, 8 of which are considered potentially preventable. We hypothesized that this method would help reviewers efficiently reach consensus on the root causes of hospital readmissions, and thus help the division and the hospital focus efforts on developing relevant quality‐improvement initiatives.
METHODS
Inclusion Criteria and Study Design
This study was conducted at CHOP, a 535‐bed urban, tertiary‐care, freestanding children's hospital with approximately 29,000 annual discharges. Of those discharges, 7000 to 8000 are from the general pediatrics service, meaning that the attending of record was a general pediatrician. Patients were included in the study if (1) they were discharged from the general pediatrics service between January 2014 and December 2014, and (2) they were readmitted to the hospital, for any reason, within 15 days of discharge. Because this analysis was done as part of a quality‐improvement initiative, it focuses on 15‐day, early readmissions to target cases with a higher probability of being potentially preventable from the perspective of the hospital care team.[10, 12, 13] Patients under observation status during the index admission or the readmission were included. However, patients who returned to the emergency department but were not admitted to an inpatient unit were excluded. Objective details about each case, including the patient's name, demographics, chart number, and diagnosis code, were pre‐loaded from EPIC (Epic Systems Corp., Verona, WI) into REDCap (Research Electronic Data Capture;
A panel of 10 general pediatricians divided up the cases to perform retrospective chart reviews. For each case, REDCap guided reviewers through the fault tree analysis. Reviewers met monthly to discuss difficult cases and reach consensus on any identified ambiguities in the process. After all cases were reviewed once, 3 panel members independently reviewed a random selection of cases to measure inter‐rater reliability and confirm reproducibility of final determinations. The inter‐rater reliability statistic was calculated using Stata 12.1 (StataCorp LP, College Station, TX). During chart reviews, panel members were not blinded to the identity of physicians and other staff members caring for the patients under review. CHOP's institutional review board determined this study to be exempt from ongoing review.
Fault Tree Analysis
Using the decision tree framework for analyzing readmissions that was developed at Intermountain Primary Children's Hospital, the REDCap tool prompted reviewers with a series of sequential questions, each with mutually exclusive options. Using imbedded branching logic to select follow‐up questions, the tool guided reviewers to 1 of 18 terminal nodes, each representing a potential root cause of the readmission. Of those 18 potential causes, 8 were considered potentially preventable. A diagram of the fault tree framework, color coded to indicate which nodes were considered potentially preventable, is shown in Figure 1.

RESULTS
In 2014, 7252 patients were discharged from the general pediatrics service at CHOP. Of those patients, 248 were readmitted within 15 days for an overall general pediatrics 15‐day readmission rate of 3.4%.
Preventability Analysis
Of the 248 readmissions, 233 (94.0%) were considered not preventable. The most common cause for readmission, which accounted for 145 cases (58.5%), was a patient developing an unpredictable problem related to the index diagnosis or a natural progression of the disease that required readmission. The second most common cause, which accounted for 53 cases (21.4%), was a patient developing a new condition unrelated to the index diagnosis or a readmission unrelated to the quality of care received during the index stay. The third most frequent cause, which accounted for 11 cases (4.4%), was a legitimate nonclinical readmission due to lack of alternative resources, psychosocial or economic factors, or case‐specific factors. Other nonpreventable causes of readmission, including scheduled readmissions, each accounted for 7 or fewer cases and <3% of total readmissions.
The 15 readmissions considered potentially preventable accounted for 6.0% of total readmissions and 0.2% of total discharges from the general pediatrics service in 2014. The most common cause of preventable readmissions, which accounted for 6 cases, was premature discharge. The second most common cause, which accounted for 4 cases, was a problem resulting from nosocomial or iatrogenic factors. Other potentially preventable causes included delayed detection of problem (3 cases), inappropriate readmission (1 case), and inadequate postdischarge care planning (1 case).
A breakdown of fault tree results, including examples of cases associated with each terminal node, is shown in Table 1. Information about general pediatrics patients and readmitted patients is included in Tables 2 and 3. A breakdown of determinations for each reviewer is included in Supporting Table 1 in the online version of this article.
| Fault Tree Terminal Node | Root Cause of Readmission | No. of Cases | % of Total Readmissions | % Within Preventability Category | % of Total Discharges |
|---|---|---|---|---|---|
| |||||
| 2 (Potentially Preventable) | Problematic condition on discharge. Example:* Index admission: Infant with history of prematurity admitted with RSV and rhinovirus bronchiolitis. Had some waxing and waning symptoms. Just prior to discharge, noted to have increased work of breathing related to feeds. Readmission: 12 hours later with tachypnea, retractions, and hypoxia. | 6 | 2.4% | 40.0% | 0.08% |
| 3 (Potentially Preventable) | Nosocomial/Iatrogenic factors. Example*: Index admission: Toddler admitted with fever and neutropenia. Treated with antibiotics 24 hours. Diagnosed with viral illness and discharged home. Readmission: symptomatic Clostridum difficile infection. | 4 | 1.6% | 26.7% | 0.06% |
| 8 (Potentially Preventable) | Detection/treatment of problem was delayed and not appropriately facilitated. Example:* Index admission: Preteen admitted with abdominal pain, concern for appendicitis. Ultrasound and abdominal MRI negative for appendicitis. Symptoms improved. Tolerated PO. Readmission: 3 days later with similar abdominal pain. Diagnosed with constipation with significant improvement following clean‐out. | 3 | 1.2% | 20.0% | 0.04% |
| 1 (Potentially Preventable) | Inappropriate readmission. Example:* Index admission: Infant with laryngomalacia admitted with bronchiolitis. Readmission: Continued mild bronchiolitis symptoms but did not require oxygen or suctioning, normal CXR. | 1 | 0.4% | 6.7% | 0.01% |
| 5 (Potentially Preventable) | Resulted from inadequate postdischarge care planning. Example:* Index diagnosis: Infant with vomiting, prior admissions, and extensive evaluation, diagnosed with milk protein allergy and GERD. PPI increased. Readmission: Persistent symptoms, required NGT feeds supplementation. | 1 | 0.4% | 6.7% | 0.01% |
| 4 (Potentially Preventable) | Resulted from a preventable complication and hospital/physician did not take the appropriate steps to minimize likelihood of complication. | ||||
| 6 (Potentially Preventable) | Resulted from improper care by patient/family and effort by hospital/physician to ensure correct postdischarge care was inadequate. | ||||
| 7 (Potentially Preventable) | Resulted from inadequate care by community services and effort by hospital/physician to ensure correct postdischarge care was inadequate. | ||||
| 15 | 6.0% | 100% | 0.2% | ||
| 12 (Not Preventable) | Problem was unpredictable. Example:* Index admission: Infant admitted with gastroenteritis and dehydration with an anion gap metabolic acidosis. Vomiting and diarrhea improved, rehydrated, acidosis improved. Readmission: 1 day later, presented with emesis and fussiness. Readmitted for metabolic acidosis. | 145 | 58.5% | 62.2% | 2.00% |
| 10 (Not Preventable) | Patient developed new condition unrelated to index diagnosis or quality of care. Example:* Index admission: Toddler admitted with cellulitis. Readmission: Bronchiolitis (did not meet CDC guidelines for nosocomial infection). | 53 | 21.4% | 22.7% | 0.73% |
| 9 (Not Preventable) | Legitimate nonclinical readmission. Example:* Index admission: Infant admitted with second episode of bronchiolitis. Readmission: 4 days later with mild diarrhea. Tolerated PO challenge in emergency department. Admitted due to parental anxiety. | 11 | 4.4% | 4.7% | 0.15% |
| 17 (Not Preventable) | Problem resulted from improper care by patient/family but effort by hospital/physician to ensure correct postdischarge care was appropriate. Example:* Index admission: Infant admitted with diarrhea, diagnosed with milk protein allergy. Discharged on soy formula. Readmission: Developed vomiting and diarrhea with cow milk formula. | 7 | 2.8% | 3.0% | 0.10% |
| 11 (Not Preventable) | Scheduled readmission. Example:* Index admission: Infant with conjunctivitis and preseptal cellulitis with nasolacrimal duct obstruction. Readmission: Postoperatively following scheduled nasolacrimal duct repair. | 7 | 2.8% | 3.0% | 0.10% |
| 14 (Not Preventable) | Detection/treatment of problem was delayed, but earlier detection was not feasible. Example:* Index admission: Preteen admitted with fever, abdominal pain, and elevated inflammatory markers. Fever resolved and symptoms improved. Diagnosed with unspecified viral infection. Readmission: 4 days later with lower extremity pyomyositis and possible osteomyelitis. | 4 | 1.6% | 1.7% | 0.06% |
| 15 (Not Preventable) | Detection/treatment of problem was delayed, earlier detection was feasible, but detection was appropriately facilitated. Example:* Index admission: Infant with history of laryngomalacia and GER admitted with an ALTE. No events during hospitalization. Appropriate workup and cleared by consultants for discharge. Zantac increased. Readmission: Infant had similar ALTE events within a week after discharge. Ultimately underwent supraglottoplasty. | 2 | 0.8% | 0.9% | 0.03% |
| 13 (Not Preventable) | Resulted from preventable complication but efforts to minimize likelihood were appropriate. Example:* Index admission: Patient on GJ feeds admitted for dislodged GJ. Extensive conversations between primary team and multiple consulting services regarding best type of tube. Determined that no other tube options were appropriate. Temporizing measures were initiated. Readmission: GJ tube dislodged again. | 2 | 0.8% | 0.9% | 0.03% |
| 18 (Not Preventable) | Resulted from medication side effect (after watch period). Example:* Index admission: Preteen with MSSA bacteremia spread to other organs. Sent home on appropriate IV antibiotics. Readmission: Fever, rash, increased LFTs. Blood cultures negative. Presumed drug reaction. Fevers resolved with alternate medication. | 2 | 0.8% | 0.9% | 0.03% |
| 16 (Not Preventable) | Resulted from inadequate care by community services, but effort by hospital/physician to ensure correct postdischarge care was appropriate. | ||||
| 233 | 94.0% | 100% | 3.2% | ||
| Fault Tree Terminal Node | Root Cause of Potentially Preventable Readmission with Case Descriptions* |
|---|---|
| |
| 2 (Potentially Preventable) | Problematic condition on discharge |
| Case 1: Index admission: Infant with history of prematurity admitted with RSV and rhinovirus bronchiolitis. Had some waxing and waning symptoms. Just prior to discharge, noted to have increased work of breathing related to feeds. Readmission: 12 hours later with tachypnea, retractions, and hypoxia. | |
| Case 2: Index admission: Toddler admitted with febrile seizure in setting of gastroenteritis. Poor PO intake during hospitalization. Readmission: 1 day later with dehydration. | |
| Case 3: Index admission: Infant admitted with a prolonged complex febrile seizure. Workup included an unremarkable lumbar puncture. No additional seizures. No inpatient imaging obtained. Readmission: Abnormal outpatient MRI requiring intervention. | |
| Case 4: Index admission: Teenager with wheezing and history of chronic daily symptoms. Discharged <24 hours later on albuterol every 4 hours and prednisone. Readmission: 1 day later, seen by primary care physician with persistent asthma flare. | |
| Case 5: Index admission: Exfull‐term infant admitted with bronchiolitis, early in course. At time of discharge, had been off oxygen for 24 hours, but last recorded respiratory rate was >70. Readmission: 1 day later due to continued tachypnea and increased work of breathing. No hypoxia. CXR normal. | |
| Case 6: Exfull‐term infant admitted with bilious emesis, diarrhea, and dehydration. Ultrasound of pylorus, UGI, and BMP all normal. Tolerated oral intake but had emesis and loose stools prior to discharge. Readmission: <48 hours later with severe metabolic acidosis. | |
| 3 (Potentially Preventable) | Nosocomial/ematrogenic factors |
| Case 1: Index admission: Toddler admitted with fever and neutropenia. Treated with antibiotics 24 hours. Diagnosed with viral illness and discharged home. Readmission: Symptomatic Clostridum difficile infection. | |
| Case 2: Index admission: Patient with autism admitted with viral gastroenteritis. Readmission: Presumed nosocominal upper respiratory infection. | |
| Case 3: Index admission: Infant admitted with bronchiolitis. Recovered from initial infection. Readmission: New upper respiratory infection and presumed nosocomial infection. | |
| Case 4: Index admission: <28‐day‐old full‐term neonate presenting with neonatal fever and rash. Full septic workup performed and all cultures negative at 24 hours. Readmission: CSF culture positive at 36 hours and readmitted while awaiting speciation. Discharged once culture grew out a contaminant. | |
| 8 (Potentially Preventable) | Detection/treatment of problem was delayed and/or not appropriately facilitated |
| Case 1: Index admission: Preteen admitted with abdominal pain, concern for appendicitis. Ultrasound and MRI abdomen negative for appendicitis. Symptoms improved. Tolerated PO. Readmission: 3 days later with similar abdominal pain. Diagnosed with constipation with significant improvement following clean‐out. | |
| Case 2: Index admission: Infant with history of macrocephaly presented with fever and full fontanelle. Head CT showed mild prominence of the extra‐axial space, and lumbar puncture was normal. Readmission: Patient developed torticollis. MRI demonstrated a malignant lesion. | |
| Case 3: Index admission: School‐age child with RLQ abdominal pain, fever, leukocytosis, and indeterminate RLQ abdominal ultrasound. Twelve‐hour observation with no further fevers. Pain and appetite improved. Readmission: 1 day later with fever, anorexia, and abdominal pain. RLQ ultrasound unchanged. Appendectomy performed with inflamed appendix. | |
| 1 (Potentially Preventable) | Inappropriate readmission |
| Case 1: Index admission: Infant with laryngomalacia admitted with bronchiolitis. Readmission: Continued mild bronchiolitis symptoms but did not require oxygen or suctioning. Normal CXR. | |
| 5 (Potentially Preventable) | Resulted from inadequate postdischarge care planning |
| Case 1: Index diagnosis: Infant with vomiting, prior admissions, and extensive evaluation, diagnosed with milk protein allergy and GERD. PPI increased. Readmission: Persistent symptoms, required NGT feeds supplementation. | |
| All General Pediatrics Patients in 2014 | General Pediatric Readmitted Patients in 2014 | ||||
|---|---|---|---|---|---|
| Major Diagnosis Category at Index Admission | No. | % | Major Diagnosis Category at Index Admission | No. | % |
| |||||
| Respiratory | 2,723 | 37.5% | Respiratory | 79 | 31.9% |
| Digestive | 748 | 10.3% | Digestive | 41 | 16.5% |
| Ear, nose, mouth, throat | 675 | 9.3% | Ear, nose, mouth, throat | 24 | 9.7% |
| Skin, subcutaneous tissue | 480 | 6.6% | Musculoskeletal and connective tissue | 14 | 5.6% |
| Infectious, parasitic, systemic | 455 | 6.3% | Nervous | 13 | 5.2% |
| Factors influencing health status | 359 | 5.0% | Endocrine, nutritional, metabolic | 13 | 5.2% |
| Endocrine, nutritional, metabolic | 339 | 4.7% | Infectious, parasitic, systemic | 12 | 4.8% |
| Nervous | 239 | 3.3% | Newborn, neonate, perinatal period | 11 | 4.4% |
| Musculoskeletal and connective tissue | 228 | 3.1% | Hepatobiliary system and pancreas | 8 | 3.2% |
| Newborn, neonate, perinatal period | 206 | 2.8% | Skin, subcutaneous tissue | 8 | 3.2% |
| Other* | 800 | 11.0% | Other | 25 | 10.1% |
| Total | 7,252 | 100% | Total | 248 | 100% |
Inter‐Rater Reliability Analysis
A random selection of 50 cases (20% of total readmissions) was selected for a second review to test the tool's inter‐rater reliability. The second review resulted in the same terminal node for 44 (86%) of the cross‐checked files ( = 0.79; 95% confidence interval: 0.60‐0.98). Of the 6 cross‐checked files that ended at different nodes, 5 resulted in the same final determination about preventability. Only 1 of the cross‐checks (2% of total cross‐checked files) resulted in a different conclusion about preventability.
Efficiency Analysis
Reviewers reported that using the tool to reach a determination about preventability took approximately 20 minutes per case. Thus, initial reviews on the 248 cases required approximately 82.6 reviewer hours. Divided across 10 reviewers, this resulted in 8 to 9 hours of review time per reviewer over the year.
DISCUSSION
As part of an effort to direct quality‐improvement initiatives, this project used a Web‐based fault tree tool to identify root causes of general pediatrics readmissions at a freestanding children's hospital and classify them as either preventable or not preventable. The project also investigated the efficiency and inter‐rater reliability of the tool, which was designed to systematically guide physicians through the chart review process to a final determination about preventability. The project confirmed that using the tool helped reviewers reach final determinations about preventability efficiently with a high degree of consistency. It also confirmed that only a very small percentage of general pediatrics 15‐day readmissions are potentially preventable. Specifically, potentially preventable readmissions accounted for only 6.0% of total readmissions and 0.2% of general pediatrics discharges in 2014. Although our analysis focused on 15‐day readmissions, the fault tree methodology can be applied to any timeframe.
Previous studies attempting to distinguish preventable from nonpreventable readmissions, which used a range of methodologies to reach final determinations, reported that their review process was both time intensive and highly subjective. One study, which had 4 reviewers independently review charts and assign each case a preventability score on a 5‐point Likert scale, reported that reviewers disagreed on the final determination in 62.5% of cases.[12] Another study had 2 physicians independently review a selection of cases and assign a preventability score on a scale from 0 to 3. Scores for the 2 reviewers were added together, and cases above a certain composite threshold were classified as preventable. Despite being time‐intensive, this method resulted in only moderate agreement among physicians about the likelihood of preventability (weighted statistic of 0.44).[14] A more recent study, in which 2 physicians independently classified readmissions into 1 of 4 predefined categories, also reported only moderate agreement between reviewers ( = 0.44).[13] Other methods that have been reported include classifying readmissions as preventable only if multiple reviewers independently agreed, and using a third reviewer as a tie‐breaker.[14]
In an attempt to identify potentially preventable readmissions without using chart reviews, 3M (St. Paul, MN) developed its Potentially Preventable Readmissions software (3M‐PPR), which uses administrative data to identify which readmissions were potentially preventable. Although this automated approach is less time intensive, evidence suggests that due to a lack of nuance, the algorithm significantly overestimates the percentage of readmissions that are potentially preventable.[4, 5] A study that used 3M‐PPR to assess 1.7 million hospitalizations across 58 children's hospitals found that the algorithm classified 81% of sickle cell crisis and asthma readmissions, and 83% of bronchiolitis readmissions as potentially preventable.[10, 11] However, many readmissions for asthma and bronchiolitis are due to social factors that are outside of a hospital's direct control,[4, 5] and at many hospitals, readmissions for sickle cell crisis are part of a high‐value care model that weighs length of stay against potential readmissions. In addition, when assessing readmissions 7, 15, and 30 days after discharge, the algorithm classified almost the same percentage as potentially preventable, which is inconsistent with the notion that readmissions are more likely to have been preventable if they occurred closer to the initial discharge.[4, 13] Another study that assessed the performance of the software in the adult population reported that the algorithm performed with 85% sensitivity, but only 28% specificity.[5, 6]
The results of this quality‐improvement project indicate that using the fault tree tool to guide physicians through the chart review process helped address some of the shortcomings of methods reported in previous studies, by increasing the efficiency and reducing the subjectivity of final determinations, while still accounting for the nuances necessary to conduct a fair review. Because the tool provided a systematic framework for reviews, each case was completed in approximately 20 minutes, and because the process was the same for all reviewers, inter‐rater reliability was extremely high. In 86% of cross‐checked cases, the second reviewer ended at the same terminal node in the decision tree as the original reviewer, and in 98% of cross‐checked cases the second reviewer reached the same conclusion about preventability, even if they did not end at the same terminal node. Even accounting for agreement due to chance, the statistic of 0.79 confirmed that there was substantial agreement among reviewers about final determinations. Because the tool is easily adaptable, other hospitals can adopt this framework for their own preventability reviews and quality‐improvement initiatives.
Using the fault tree tool to access root causes of all 15‐day general pediatric readmissions helped the division focus quality‐improvement efforts on the most common causes of potentially preventable readmissions. Because 40% of potentially preventable readmissions were due to premature discharges, this prompted quality‐improvement teams to focus efforts on improving and clarifying the division's discharge criteria and clinical pathways. The division also initiated processes to improve discharge planning, including improved teaching of discharge instructions and having families pick up prescriptions prior to discharge.
Although these results did help the division identify a few areas of focus to potentially reduce readmissions, the fact that the overall 15‐day readmission rate for general pediatrics, as well as the percentage of readmissions and total discharges that were deemed potentially preventable, were so low (3.4%, 6.0%, and 0.2%, respectively), supports those who question whether prioritizing pediatric readmissions is the best place for hospitals to focus quality‐improvement efforts.[10, 12, 15, 16] As these results indicate, most pediatric readmissions are not preventable, and thus consistent with an efficient, effective, timely, patient‐centered, and equitable health system. Other studies have also shown that because overall and condition‐specific readmissions at pediatric hospitals are low, few pediatric hospitals are high or low performing for readmissions, and thus readmission rates are likely not a good measure of hospital quality.[8]
However, other condition‐specific studies of readmissions in pediatrics have indicated that there are some areas of opportunity to identify populations at high risk for readmission. One study found that although pneumonia‐specific 30‐day readmission rates in a national cohort of children hospitalized with pneumonia was only 3.1%, the chances of readmission were higher for children <1 year old, children with chronic comorbidities or complicated pneumonia, and children cared for in hospitals with lower volumes of pneumonia admissions.[17] Another study found that 17.1% of adolescents in a statewide database were readmitted post‐tonsillectomy for pain, nausea, and dehydration.[18] Thus, adapting the tool to identify root causes of condition‐specific or procedure‐specific readmissions, especially for surgical patients, may be an area of opportunity for future quality‐improvement efforts.[5] However, for general pediatrics, shifting the focus from reducing readmissions to improving the quality of care patients receive in the hospital, improving the discharge process, and adopting a population health approach to mitigate external risk factors, may be appropriate.
This project was subject to limitations. First, because it was conducted at a single site and only on general pediatrics patients, results may not be generalizable to other hospitals or other pediatric divisions. Thus, future studies might use the fault tree framework to assess preventability of pediatric readmissions in other divisions or specialties. Second, because readmissions to other hospitals were not included in the sample, the overall readmissions rate is likely underestimated.[19] However, it is unclear how this would affect the rate of potentially preventable readmissions. Third, although the fault tree framework reduced the subjectivity of the review process, there is still a degree of subjectivity inherent at each decision node. To minimize this, reviewers should try to discuss and come to consensus on how they are making determinations at each juncture in the decision tree. Similarly, because reviewers' answers to decision‐tree questions rely heavily on chart documentation, reviews may be compromised by unclear or incomplete documentation. For example, if information about steps the hospital team took to prepare a family for discharge were not properly documented, it would be difficult to determine whether appropriate steps were taken to minimize the likelihood of a complication. In the case of insufficient documentation of relevant social concerns, cases may be incorrectly classified as preventable, because addressing social issues is often not within a hospital's direct control. Finally, because reviewers were not blinded to the original discharging physician, there may have been some unconscious bias of unknown direction in the reviews.
CONCLUSION
Using the Web‐based fault tree tool helped physicians to identify the root causes of hospital readmissions and classify them as preventable or not preventable in a standardized, efficient, and consistent way, while still accounting for the nuances necessary to conduct a fair review. Thus, other hospitals should consider adopting this framework for their own preventability reviews and quality‐improvement initiatives. However, this project also confirmed that only a very small percentage of general pediatrics 15‐day readmissions are potentially preventable, suggesting that general pediatrics readmissions are not an appropriate measure of hospital quality. Instead, adapting the tool to identify root causes of condition‐specific or procedure‐specific readmission rates may be an area of opportunity for future quality‐improvement efforts.
Disclosures: This work was supported through internal funds from The Children's Hospital of Philadelphia. The authors have no financial interests, relationships or affiliations relevant to the subject matter or materials discussed in the article to disclose. The authors have no potential conflicts of interest relevant to the subject matter or materials discussed in the article to disclose.
- , . Pediatric readmissions as a hospital quality measure. JAMA. 2013;309(4):396–398.
- Texas Health and Human Services Commission. Potentially preventable readmissions in the Texas Medicaid population, state fiscal year 2012. Available at: http://www.hhsc.state.tx.us/reports/2013/ppr‐report.pdf. Published November 2013. Accessed August 16, 2015.
- Illinois Department of Healthcare and Family Services. Quality initiative to reduce hospital potentially preventable readmissions (PPR): Status update. Available at: http://www.illinois.gov/hfs/SiteCollectionDocuments/PPRPolicyStatusUpdate.pdf. Published September 3, 2014. Accessed August 16, 2015.
- , , , et al. Rates and impact of potentially preventable readmissions at children's hospitals. J Pediatr. 2015;166(3):613–619.e615.
- , . Preventing pediatric readmissions: which ones and how? J Pediatr. 2015;166(3):519–520.
- , , , , , . Manual and automated methods for identifying potentially preventable readmissions: a comparison in a large healthcare system. BMC Med Inform Decis Mak. 2014;14:28.
- , . Section on hospital medicine leadership and staff. Hosp Pediatr. 2013;3(4):390–393.
- , , , et al. Measuring hospital quality using pediatric readmission and revisit rates. Pediatrics. 2013;132(3):429–436.
- , . Hospital readmissions—not just a measure of quality. JAMA. 2011;306(16):1796–1797.
- , . Preventing readmissions in children: how do we do that? Hosp Pediatr. 2015;5(11):602–604.
- , , , et al. Pediatric readmission prevalence and variability across hospitals. JAMA. 2013;309(4):372–380.
- , , , , , . Preventability of early readmissions at a children's hospital. Pediatrics. 2013;131(1):e171–e181.
- , , , et al. An examination of physician‐, caregiver‐, and disease‐related factors associated with readmission from a pediatric hospital medicine service. Hosp Pediatr. 2015;5(11):566–573.
- , , , et al. Clinical preventability of 30‐day readmission after percutaneous coronary intervention. J Am Heart Assoc. 2014;3(5):e001290.
- . 3M algorithm overestimates preventable pediatric readmissions. Hospitalist News website. Available at: http://www.ehospitalistnews.com/specialty‐focus/pediatrics/single‐article‐page/3m‐algorithm‐overestimates‐preventable‐pediatric‐readmissions.html. Published August 16, 2013. Accessed August 16, 2015.
- . The 30‐day readmission rate: not a quality measure but an accountability measure. An Ounce of Evidence: Health Policy blog. Available at: https://blogs.sph.harvard.edu/ashish‐jha/?s=30‐day+readmission+rate. Published February 14, 2013. Accessed August 16, 2015.
- , , , et al. Readmissions among children previously hospitalized with pneumonia. Pediatrics. 2014;134(1):100–109.
- , , . A population‐based study of acute care revisits following tonsillectomy. J Pediatr. 2015;166(3):607–612.e605.
- , , , et al. Same‐hospital readmission rates as a measure of pediatric quality of care. JAMA Pediatr. 2015;169(10):905–912.
- , . Pediatric readmissions as a hospital quality measure. JAMA. 2013;309(4):396–398.
- Texas Health and Human Services Commission. Potentially preventable readmissions in the Texas Medicaid population, state fiscal year 2012. Available at: http://www.hhsc.state.tx.us/reports/2013/ppr‐report.pdf. Published November 2013. Accessed August 16, 2015.
- Illinois Department of Healthcare and Family Services. Quality initiative to reduce hospital potentially preventable readmissions (PPR): Status update. Available at: http://www.illinois.gov/hfs/SiteCollectionDocuments/PPRPolicyStatusUpdate.pdf. Published September 3, 2014. Accessed August 16, 2015.
- , , , et al. Rates and impact of potentially preventable readmissions at children's hospitals. J Pediatr. 2015;166(3):613–619.e615.
- , . Preventing pediatric readmissions: which ones and how? J Pediatr. 2015;166(3):519–520.
- , , , , , . Manual and automated methods for identifying potentially preventable readmissions: a comparison in a large healthcare system. BMC Med Inform Decis Mak. 2014;14:28.
- , . Section on hospital medicine leadership and staff. Hosp Pediatr. 2013;3(4):390–393.
- , , , et al. Measuring hospital quality using pediatric readmission and revisit rates. Pediatrics. 2013;132(3):429–436.
- , . Hospital readmissions—not just a measure of quality. JAMA. 2011;306(16):1796–1797.
- , . Preventing readmissions in children: how do we do that? Hosp Pediatr. 2015;5(11):602–604.
- , , , et al. Pediatric readmission prevalence and variability across hospitals. JAMA. 2013;309(4):372–380.
- , , , , , . Preventability of early readmissions at a children's hospital. Pediatrics. 2013;131(1):e171–e181.
- , , , et al. An examination of physician‐, caregiver‐, and disease‐related factors associated with readmission from a pediatric hospital medicine service. Hosp Pediatr. 2015;5(11):566–573.
- , , , et al. Clinical preventability of 30‐day readmission after percutaneous coronary intervention. J Am Heart Assoc. 2014;3(5):e001290.
- . 3M algorithm overestimates preventable pediatric readmissions. Hospitalist News website. Available at: http://www.ehospitalistnews.com/specialty‐focus/pediatrics/single‐article‐page/3m‐algorithm‐overestimates‐preventable‐pediatric‐readmissions.html. Published August 16, 2013. Accessed August 16, 2015.
- . The 30‐day readmission rate: not a quality measure but an accountability measure. An Ounce of Evidence: Health Policy blog. Available at: https://blogs.sph.harvard.edu/ashish‐jha/?s=30‐day+readmission+rate. Published February 14, 2013. Accessed August 16, 2015.
- , , , et al. Readmissions among children previously hospitalized with pneumonia. Pediatrics. 2014;134(1):100–109.
- , , . A population‐based study of acute care revisits following tonsillectomy. J Pediatr. 2015;166(3):607–612.e605.
- , , , et al. Same‐hospital readmission rates as a measure of pediatric quality of care. JAMA Pediatr. 2015;169(10):905–912.
© 2016 Society of Hospital Medicine
Hereditary Cancer Testing in a Value Based World: The Evolving Standard of Care
Commemorating the 20th anniversary of BRCA genetic testing, we are increasingly conscious of the social, economic, and medico-legal impacts of genetic cancer risk on both personal and public health. An ever-growing number of medical societies and expert panels have now charged clinicians with the duty to evaluate and manage their patients’ cancer risk profiles, with particular attention to those individuals carrying genetic mutations that confer elevated cancer susceptibility, a process commonly known as hereditary cancer risk assessment, or HCRA.

Click here to download the PDF.
This article is written for women’s health care clinicians, and will address the current status of hereditary cancer genetic testing, focusing on the evolution of pan-cancer, multigene panel testing in the context of value-based medicine.
Click on the video below to watch Dr. Frieder discuss Hereditary Cancer Testing.
Commemorating the 20th anniversary of BRCA genetic testing, we are increasingly conscious of the social, economic, and medico-legal impacts of genetic cancer risk on both personal and public health. An ever-growing number of medical societies and expert panels have now charged clinicians with the duty to evaluate and manage their patients’ cancer risk profiles, with particular attention to those individuals carrying genetic mutations that confer elevated cancer susceptibility, a process commonly known as hereditary cancer risk assessment, or HCRA.

Click here to download the PDF.
This article is written for women’s health care clinicians, and will address the current status of hereditary cancer genetic testing, focusing on the evolution of pan-cancer, multigene panel testing in the context of value-based medicine.
Click on the video below to watch Dr. Frieder discuss Hereditary Cancer Testing.
Commemorating the 20th anniversary of BRCA genetic testing, we are increasingly conscious of the social, economic, and medico-legal impacts of genetic cancer risk on both personal and public health. An ever-growing number of medical societies and expert panels have now charged clinicians with the duty to evaluate and manage their patients’ cancer risk profiles, with particular attention to those individuals carrying genetic mutations that confer elevated cancer susceptibility, a process commonly known as hereditary cancer risk assessment, or HCRA.

Click here to download the PDF.
This article is written for women’s health care clinicians, and will address the current status of hereditary cancer genetic testing, focusing on the evolution of pan-cancer, multigene panel testing in the context of value-based medicine.
Click on the video below to watch Dr. Frieder discuss Hereditary Cancer Testing.
Hepatitis C May Increase Risk of Parkinson’s Disease
Hepatitis C infection may increase the risk of Parkinson’s disease, according to a nationwide population-based study published online ahead of print December 23, 2015, in Neurology. Researchers analyzed 10 years of data from the Taiwan National Health Insurance Research Database, which included 49,967 patients with viral hepatitis—35,619 with hepatitis B infection, 10,286 with hepatitis C, and 4,062 with both—and 199,868 noninfected controls.
Individuals with hepatitis C infection had a 29% greater incidence of Parkinson’s disease after adjustment for confounders such as sex, age, heart disease, stroke, and head injury. The researchers found no significant associations between hepatitis B or coinfection and Parkinson’s disease risk.
Age was the most common risk factor for Parkinson’s disease across all cohorts, and in the control group, comorbidities such as hyperlipidemia, hypertension, ischemic heart disease, diabetes, and head injury all were associated with a significant increase in the risk of Parkinson’s disease. Among individuals with hepatitis C infection, however, only ischemic heart disease and head injury remained significantly associated with Parkinson’s disease risk.
The possibility of an association between hepatitis C infection and Parkinson’s disease has emerged recently, with evidence showing that the virus is neurotropic and can replicate in the CNS, reported Hsin-Hsi Tsai, MD, a neurologist at the National Taiwan University Hospital in Taipei, and coauthors.
“Parkinsonism is rarely a described feature in patients with hepatitis C virus. However, a recent study has discovered that hepatitis C virus can induce dopaminergic neuron death, suggesting a possible association between hepatitis C virus infection and” Parkinson’s disease, said the authors.
The study also showed that the association between hepatitis C infection and Parkinson’s disease was more significant in individuals younger than 65, who had a 61% greater risk of developing the neurodegenerative disease.
“Some of the risk factors for hepatitis C virus infection, such as illicit drug use and associated behaviors, may be confounding factors in this age group,” said the authors. They pointed out, however, that in Taiwan, use of IV drugs was not known to be a risk factor for infection. Commenting on a possible mechanism for the association between hepatitis C infection and Parkinson’s disease, Dr. Tsai and associates suggested that the hepatitis C virus could be a possible viral candidate for triggering the neuroinflammation that is a characteristic feature of Parkinson’s disease.
“An earlier imaging study that involved using magnetic resonance spectroscopy to investigate the cerebral effect of hepatitis C virus showed that chronic hepatitis C virus infection was associated with elevated choline/creatinine ratios, a biomarker indicating inflammatory and infective conditions, in the basal ganglia and white matter,” they said.
—Bianca Nogrady
Suggested Reading
Tsai HH, Liou HH, Muo CH, et al. Hepatitis C virus infection as a risk factor for Parkinson disease: A nationwide cohort study. Neurology. 2015 Dec 23 [Epub ahead of print].
Hepatitis C infection may increase the risk of Parkinson’s disease, according to a nationwide population-based study published online ahead of print December 23, 2015, in Neurology. Researchers analyzed 10 years of data from the Taiwan National Health Insurance Research Database, which included 49,967 patients with viral hepatitis—35,619 with hepatitis B infection, 10,286 with hepatitis C, and 4,062 with both—and 199,868 noninfected controls.
Individuals with hepatitis C infection had a 29% greater incidence of Parkinson’s disease after adjustment for confounders such as sex, age, heart disease, stroke, and head injury. The researchers found no significant associations between hepatitis B or coinfection and Parkinson’s disease risk.
Age was the most common risk factor for Parkinson’s disease across all cohorts, and in the control group, comorbidities such as hyperlipidemia, hypertension, ischemic heart disease, diabetes, and head injury all were associated with a significant increase in the risk of Parkinson’s disease. Among individuals with hepatitis C infection, however, only ischemic heart disease and head injury remained significantly associated with Parkinson’s disease risk.
The possibility of an association between hepatitis C infection and Parkinson’s disease has emerged recently, with evidence showing that the virus is neurotropic and can replicate in the CNS, reported Hsin-Hsi Tsai, MD, a neurologist at the National Taiwan University Hospital in Taipei, and coauthors.
“Parkinsonism is rarely a described feature in patients with hepatitis C virus. However, a recent study has discovered that hepatitis C virus can induce dopaminergic neuron death, suggesting a possible association between hepatitis C virus infection and” Parkinson’s disease, said the authors.
The study also showed that the association between hepatitis C infection and Parkinson’s disease was more significant in individuals younger than 65, who had a 61% greater risk of developing the neurodegenerative disease.
“Some of the risk factors for hepatitis C virus infection, such as illicit drug use and associated behaviors, may be confounding factors in this age group,” said the authors. They pointed out, however, that in Taiwan, use of IV drugs was not known to be a risk factor for infection. Commenting on a possible mechanism for the association between hepatitis C infection and Parkinson’s disease, Dr. Tsai and associates suggested that the hepatitis C virus could be a possible viral candidate for triggering the neuroinflammation that is a characteristic feature of Parkinson’s disease.
“An earlier imaging study that involved using magnetic resonance spectroscopy to investigate the cerebral effect of hepatitis C virus showed that chronic hepatitis C virus infection was associated with elevated choline/creatinine ratios, a biomarker indicating inflammatory and infective conditions, in the basal ganglia and white matter,” they said.
—Bianca Nogrady
Hepatitis C infection may increase the risk of Parkinson’s disease, according to a nationwide population-based study published online ahead of print December 23, 2015, in Neurology. Researchers analyzed 10 years of data from the Taiwan National Health Insurance Research Database, which included 49,967 patients with viral hepatitis—35,619 with hepatitis B infection, 10,286 with hepatitis C, and 4,062 with both—and 199,868 noninfected controls.
Individuals with hepatitis C infection had a 29% greater incidence of Parkinson’s disease after adjustment for confounders such as sex, age, heart disease, stroke, and head injury. The researchers found no significant associations between hepatitis B or coinfection and Parkinson’s disease risk.
Age was the most common risk factor for Parkinson’s disease across all cohorts, and in the control group, comorbidities such as hyperlipidemia, hypertension, ischemic heart disease, diabetes, and head injury all were associated with a significant increase in the risk of Parkinson’s disease. Among individuals with hepatitis C infection, however, only ischemic heart disease and head injury remained significantly associated with Parkinson’s disease risk.
The possibility of an association between hepatitis C infection and Parkinson’s disease has emerged recently, with evidence showing that the virus is neurotropic and can replicate in the CNS, reported Hsin-Hsi Tsai, MD, a neurologist at the National Taiwan University Hospital in Taipei, and coauthors.
“Parkinsonism is rarely a described feature in patients with hepatitis C virus. However, a recent study has discovered that hepatitis C virus can induce dopaminergic neuron death, suggesting a possible association between hepatitis C virus infection and” Parkinson’s disease, said the authors.
The study also showed that the association between hepatitis C infection and Parkinson’s disease was more significant in individuals younger than 65, who had a 61% greater risk of developing the neurodegenerative disease.
“Some of the risk factors for hepatitis C virus infection, such as illicit drug use and associated behaviors, may be confounding factors in this age group,” said the authors. They pointed out, however, that in Taiwan, use of IV drugs was not known to be a risk factor for infection. Commenting on a possible mechanism for the association between hepatitis C infection and Parkinson’s disease, Dr. Tsai and associates suggested that the hepatitis C virus could be a possible viral candidate for triggering the neuroinflammation that is a characteristic feature of Parkinson’s disease.
“An earlier imaging study that involved using magnetic resonance spectroscopy to investigate the cerebral effect of hepatitis C virus showed that chronic hepatitis C virus infection was associated with elevated choline/creatinine ratios, a biomarker indicating inflammatory and infective conditions, in the basal ganglia and white matter,” they said.
—Bianca Nogrady
Suggested Reading
Tsai HH, Liou HH, Muo CH, et al. Hepatitis C virus infection as a risk factor for Parkinson disease: A nationwide cohort study. Neurology. 2015 Dec 23 [Epub ahead of print].
Suggested Reading
Tsai HH, Liou HH, Muo CH, et al. Hepatitis C virus infection as a risk factor for Parkinson disease: A nationwide cohort study. Neurology. 2015 Dec 23 [Epub ahead of print].
High Urate Concentration May Protect Men Against Parkinson’s Disease
Men with a high plasma urate concentration have a decreased risk of developing Parkinson’s disease, independent of potential risk factors such as age, smoking, and caffeine intake, according to research published online ahead of print January 13 in Neurology. Plasma urate concentration appears to have no association with risk of Parkinson’s disease among women, however. For men, urate could protect against Parkinson’s disease risk or slow the progression of preclinical Parkinson’s disease, according to the authors.
“Our findings, together with previous observations that urate can be elevated by administration of its precursor inosine, which is generally safe and tolerable, in early Parkinson’s disease, provide strong evidence supporting the design of a randomized trial of urate elevation in patients with early Parkinson’s disease or pre-Parkinson syndrome,” said Xiang Gao, MD, PhD, Director of the Nutritional Epidemiology Laboratory at Pennsylvania State University in University Park.
Dr. Gao and colleagues examined blood samples for more than 90,000 men and women who participated in the Health Professionals Follow-up Study, the Nurses’ Health Study, or the Cancer Prevention Study II Nutrition. The researchers confirmed cases of Parkinson’s disease based on a detailed questionnaire that the treating neurologist or internists completed, or through a movement disorder specialist’s review of medical records. Only patients with definite or probable Parkinson’s disease were included in the analysis. Between one and six controls were selected randomly for each case. The investigators used questionnaires to collect data on potential confounders, including age, smoking status, height, weight, chronic diseases, and consumption of caffeinated coffee and alcohol.
Dr. Gao’s group identified 388 new incident cases of Parkinson’s disease since blood collection and matched them with 1,267 controls. Higher baseline urate concentrations were associated with lower risk of Parkinson’s disease in men, but not in women. The multivariate-adjusted risk ratios of Parkinson’s disease, comparing the highest and lowest quartiles of urate, were 0.63 in men and 1.04 in women. Adjusting the data for cardiovascular factors, including history of cardiovascular disease and diabetes, did not affect the results.
The researchers pooled the results of their study with those of three previous investigations that included 325 patients with Parkinson’s disease. The pooled risk ratios comparing the highest and lowest categories of urate were 0.63 in men and 0.89 in women.
Animal and human studies suggest that urate is a neuroprotective agent, noted the authors. Their own previous research indicates that urate may slow disease progression during the preclinical stage of Parkinson’s disease.
The strengths of the current study include its prospective design, large sample size, and availability of information on covariates that may confound the potential association between serum urate and Parkinson’s disease risk, according to the authors. The study was based on a single measure of plasma urate, however, and did not account for within-person variability in urate levels. In addition, the results may be difficult to generalize because the majority of participants were Caucasian and had high educational attainment and socioeconomic status.
—Erik Greb
Suggested Reading
Gao X, O’Reilly ÉJ, Schwarzschild MA, Ascherio A. Prospective study of plasma urate and risk of Parkinson disease in men and women. Neurology. 2016 Jan 13 [Epub ahead of print].
Men with a high plasma urate concentration have a decreased risk of developing Parkinson’s disease, independent of potential risk factors such as age, smoking, and caffeine intake, according to research published online ahead of print January 13 in Neurology. Plasma urate concentration appears to have no association with risk of Parkinson’s disease among women, however. For men, urate could protect against Parkinson’s disease risk or slow the progression of preclinical Parkinson’s disease, according to the authors.
“Our findings, together with previous observations that urate can be elevated by administration of its precursor inosine, which is generally safe and tolerable, in early Parkinson’s disease, provide strong evidence supporting the design of a randomized trial of urate elevation in patients with early Parkinson’s disease or pre-Parkinson syndrome,” said Xiang Gao, MD, PhD, Director of the Nutritional Epidemiology Laboratory at Pennsylvania State University in University Park.
Dr. Gao and colleagues examined blood samples for more than 90,000 men and women who participated in the Health Professionals Follow-up Study, the Nurses’ Health Study, or the Cancer Prevention Study II Nutrition. The researchers confirmed cases of Parkinson’s disease based on a detailed questionnaire that the treating neurologist or internists completed, or through a movement disorder specialist’s review of medical records. Only patients with definite or probable Parkinson’s disease were included in the analysis. Between one and six controls were selected randomly for each case. The investigators used questionnaires to collect data on potential confounders, including age, smoking status, height, weight, chronic diseases, and consumption of caffeinated coffee and alcohol.
Dr. Gao’s group identified 388 new incident cases of Parkinson’s disease since blood collection and matched them with 1,267 controls. Higher baseline urate concentrations were associated with lower risk of Parkinson’s disease in men, but not in women. The multivariate-adjusted risk ratios of Parkinson’s disease, comparing the highest and lowest quartiles of urate, were 0.63 in men and 1.04 in women. Adjusting the data for cardiovascular factors, including history of cardiovascular disease and diabetes, did not affect the results.
The researchers pooled the results of their study with those of three previous investigations that included 325 patients with Parkinson’s disease. The pooled risk ratios comparing the highest and lowest categories of urate were 0.63 in men and 0.89 in women.
Animal and human studies suggest that urate is a neuroprotective agent, noted the authors. Their own previous research indicates that urate may slow disease progression during the preclinical stage of Parkinson’s disease.
The strengths of the current study include its prospective design, large sample size, and availability of information on covariates that may confound the potential association between serum urate and Parkinson’s disease risk, according to the authors. The study was based on a single measure of plasma urate, however, and did not account for within-person variability in urate levels. In addition, the results may be difficult to generalize because the majority of participants were Caucasian and had high educational attainment and socioeconomic status.
—Erik Greb
Men with a high plasma urate concentration have a decreased risk of developing Parkinson’s disease, independent of potential risk factors such as age, smoking, and caffeine intake, according to research published online ahead of print January 13 in Neurology. Plasma urate concentration appears to have no association with risk of Parkinson’s disease among women, however. For men, urate could protect against Parkinson’s disease risk or slow the progression of preclinical Parkinson’s disease, according to the authors.
“Our findings, together with previous observations that urate can be elevated by administration of its precursor inosine, which is generally safe and tolerable, in early Parkinson’s disease, provide strong evidence supporting the design of a randomized trial of urate elevation in patients with early Parkinson’s disease or pre-Parkinson syndrome,” said Xiang Gao, MD, PhD, Director of the Nutritional Epidemiology Laboratory at Pennsylvania State University in University Park.
Dr. Gao and colleagues examined blood samples for more than 90,000 men and women who participated in the Health Professionals Follow-up Study, the Nurses’ Health Study, or the Cancer Prevention Study II Nutrition. The researchers confirmed cases of Parkinson’s disease based on a detailed questionnaire that the treating neurologist or internists completed, or through a movement disorder specialist’s review of medical records. Only patients with definite or probable Parkinson’s disease were included in the analysis. Between one and six controls were selected randomly for each case. The investigators used questionnaires to collect data on potential confounders, including age, smoking status, height, weight, chronic diseases, and consumption of caffeinated coffee and alcohol.
Dr. Gao’s group identified 388 new incident cases of Parkinson’s disease since blood collection and matched them with 1,267 controls. Higher baseline urate concentrations were associated with lower risk of Parkinson’s disease in men, but not in women. The multivariate-adjusted risk ratios of Parkinson’s disease, comparing the highest and lowest quartiles of urate, were 0.63 in men and 1.04 in women. Adjusting the data for cardiovascular factors, including history of cardiovascular disease and diabetes, did not affect the results.
The researchers pooled the results of their study with those of three previous investigations that included 325 patients with Parkinson’s disease. The pooled risk ratios comparing the highest and lowest categories of urate were 0.63 in men and 0.89 in women.
Animal and human studies suggest that urate is a neuroprotective agent, noted the authors. Their own previous research indicates that urate may slow disease progression during the preclinical stage of Parkinson’s disease.
The strengths of the current study include its prospective design, large sample size, and availability of information on covariates that may confound the potential association between serum urate and Parkinson’s disease risk, according to the authors. The study was based on a single measure of plasma urate, however, and did not account for within-person variability in urate levels. In addition, the results may be difficult to generalize because the majority of participants were Caucasian and had high educational attainment and socioeconomic status.
—Erik Greb
Suggested Reading
Gao X, O’Reilly ÉJ, Schwarzschild MA, Ascherio A. Prospective study of plasma urate and risk of Parkinson disease in men and women. Neurology. 2016 Jan 13 [Epub ahead of print].
Suggested Reading
Gao X, O’Reilly ÉJ, Schwarzschild MA, Ascherio A. Prospective study of plasma urate and risk of Parkinson disease in men and women. Neurology. 2016 Jan 13 [Epub ahead of print].