User login
Large study amplifies evidence of COVID vaccine safety in pregnancy
The research team wrote in the BMJ that their reassuring findings – drawn from a registry of all births in Ontario over an 8-month period – “can inform evidence-based decision-making” about COVID vaccination during pregnancy.
Previous research has found that pregnant patients are at higher risk of severe complications and death if they become infected with COVID and that vaccination before or during pregnancy prevents such outcomes and reduces the risk of newborn infection, noted Jeffrey Ecker, chief of obstetrics and gynecology at Massachusetts General Hospital, Boston.
This new study “adds to a growing body of information arguing clearly and reassuringly that vaccination during pregnancy is not associated with complications during pregnancy,” said Dr. Ecker, who was not involved in the new study.
He added that it “should help obstetric providers further reassure those who are hesitant that vaccination is safe and best both for the pregnant patient and their pregnancy.”
Methods and results
For the new study, researchers tapped a provincial registry of all live and stillborn infants with a gestational age of at least 20 weeks or birth weight of at least 500 g. Unique health card numbers were used to link birth records to a database of COVID vaccinations.
Of 85,162 infants born from May through December of 2021, 43,099 (50.6%) were born to individuals who received at least one vaccine dose during pregnancy. Among those, 99.7% received an mRNA vaccine such as Pfizer-BioNTech or Moderna.
Vaccination during pregnancy was not associated with greater risk of overall preterm birth (6.5% among vaccinated individuals versus 6.9% among unvaccinated; hazard ratio, 1.02; 95% confidence interval, 0.96-1.08), spontaneous preterm birth (3.7% versus 4.4%; hazard ratio, 0.96; 95% CI, 0.90-1.03) or very preterm birth (0.59% versus 0.89%; hazard ratio, 0.80; 95% CI, 0.67-0.95).
Likewise, no increase was observed in the risk of an infant being small for gestational age at birth (9.1% versus 9.2%; hazard ratio, 0.98; 95% CI, 0.93-1.03).
The researchers observed a reduction in the risk of stillbirth, even after adjusting for potential confounders. Stillbirths occurred in 0.25% of vaccinated individuals, compared with 0.44% of unvaccinated individuals (hazard ratio, 0.65; 95% CI, 0.51-0.84).
A reduced risk of stillbirth – albeit to a smaller degree – was also found in a Scandinavian registry study that included 28,506 babies born to individuals who were vaccinated during pregnancy.
“Collectively, the findings from these two studies are reassuring and are consistent with no increased risk of stillbirth after COVID-19 vaccination during pregnancy. In contrast, COVID-19 disease during pregnancy has been associated with an increased risk of stillbirth,” the researchers wrote.
Findings did not vary by which mRNA vaccine a mother received, the number of doses she received, or the trimester in which a vaccine was given, the researchers reported.
Stillbirth findings will be ‘very reassuring’ for patients
The lead investigator, Deshayne Fell, PhD, said in an interview, the fact that the study comprised the entire population of pregnant people in Ontario during the study period “increases our confidence” about the validity and relevance of the findings for other geographic settings.
Dr. Fell, an associate professor in epidemiology and public health at the University of Ottawa and a scientist at the Children’s Hospital of Eastern Ontario Research Institute, Ottawa, said the evaluation of stillbirth in particular, “a rare but devastating outcome,” will be “very reassuring and useful for clinical counseling.”
A limitation cited by the research team included a lack of data on vaccination prior to pregnancy.
In the new study, Dr, Ecker said, “Though the investigators were able to adjust for many variables they cannot be certain that some unmeasured variable that, accordingly, was not adjusted for does not hide a small risk. This seems very unlikely, however.”
The Canadian research team said similar studies of non-mRNA COVID vaccines “should be a research priority.” However, such studies are not underway in Canada, where only mRNA vaccines are used in pregnancy, Dr. Fell said.
This study was supported by the Public Health Agency of Canada.
Dr. Fell and Dr. Ecker reported no competing financial interests.
The research team wrote in the BMJ that their reassuring findings – drawn from a registry of all births in Ontario over an 8-month period – “can inform evidence-based decision-making” about COVID vaccination during pregnancy.
Previous research has found that pregnant patients are at higher risk of severe complications and death if they become infected with COVID and that vaccination before or during pregnancy prevents such outcomes and reduces the risk of newborn infection, noted Jeffrey Ecker, chief of obstetrics and gynecology at Massachusetts General Hospital, Boston.
This new study “adds to a growing body of information arguing clearly and reassuringly that vaccination during pregnancy is not associated with complications during pregnancy,” said Dr. Ecker, who was not involved in the new study.
He added that it “should help obstetric providers further reassure those who are hesitant that vaccination is safe and best both for the pregnant patient and their pregnancy.”
Methods and results
For the new study, researchers tapped a provincial registry of all live and stillborn infants with a gestational age of at least 20 weeks or birth weight of at least 500 g. Unique health card numbers were used to link birth records to a database of COVID vaccinations.
Of 85,162 infants born from May through December of 2021, 43,099 (50.6%) were born to individuals who received at least one vaccine dose during pregnancy. Among those, 99.7% received an mRNA vaccine such as Pfizer-BioNTech or Moderna.
Vaccination during pregnancy was not associated with greater risk of overall preterm birth (6.5% among vaccinated individuals versus 6.9% among unvaccinated; hazard ratio, 1.02; 95% confidence interval, 0.96-1.08), spontaneous preterm birth (3.7% versus 4.4%; hazard ratio, 0.96; 95% CI, 0.90-1.03) or very preterm birth (0.59% versus 0.89%; hazard ratio, 0.80; 95% CI, 0.67-0.95).
Likewise, no increase was observed in the risk of an infant being small for gestational age at birth (9.1% versus 9.2%; hazard ratio, 0.98; 95% CI, 0.93-1.03).
The researchers observed a reduction in the risk of stillbirth, even after adjusting for potential confounders. Stillbirths occurred in 0.25% of vaccinated individuals, compared with 0.44% of unvaccinated individuals (hazard ratio, 0.65; 95% CI, 0.51-0.84).
A reduced risk of stillbirth – albeit to a smaller degree – was also found in a Scandinavian registry study that included 28,506 babies born to individuals who were vaccinated during pregnancy.
“Collectively, the findings from these two studies are reassuring and are consistent with no increased risk of stillbirth after COVID-19 vaccination during pregnancy. In contrast, COVID-19 disease during pregnancy has been associated with an increased risk of stillbirth,” the researchers wrote.
Findings did not vary by which mRNA vaccine a mother received, the number of doses she received, or the trimester in which a vaccine was given, the researchers reported.
Stillbirth findings will be ‘very reassuring’ for patients
The lead investigator, Deshayne Fell, PhD, said in an interview, the fact that the study comprised the entire population of pregnant people in Ontario during the study period “increases our confidence” about the validity and relevance of the findings for other geographic settings.
Dr. Fell, an associate professor in epidemiology and public health at the University of Ottawa and a scientist at the Children’s Hospital of Eastern Ontario Research Institute, Ottawa, said the evaluation of stillbirth in particular, “a rare but devastating outcome,” will be “very reassuring and useful for clinical counseling.”
A limitation cited by the research team included a lack of data on vaccination prior to pregnancy.
In the new study, Dr, Ecker said, “Though the investigators were able to adjust for many variables they cannot be certain that some unmeasured variable that, accordingly, was not adjusted for does not hide a small risk. This seems very unlikely, however.”
The Canadian research team said similar studies of non-mRNA COVID vaccines “should be a research priority.” However, such studies are not underway in Canada, where only mRNA vaccines are used in pregnancy, Dr. Fell said.
This study was supported by the Public Health Agency of Canada.
Dr. Fell and Dr. Ecker reported no competing financial interests.
The research team wrote in the BMJ that their reassuring findings – drawn from a registry of all births in Ontario over an 8-month period – “can inform evidence-based decision-making” about COVID vaccination during pregnancy.
Previous research has found that pregnant patients are at higher risk of severe complications and death if they become infected with COVID and that vaccination before or during pregnancy prevents such outcomes and reduces the risk of newborn infection, noted Jeffrey Ecker, chief of obstetrics and gynecology at Massachusetts General Hospital, Boston.
This new study “adds to a growing body of information arguing clearly and reassuringly that vaccination during pregnancy is not associated with complications during pregnancy,” said Dr. Ecker, who was not involved in the new study.
He added that it “should help obstetric providers further reassure those who are hesitant that vaccination is safe and best both for the pregnant patient and their pregnancy.”
Methods and results
For the new study, researchers tapped a provincial registry of all live and stillborn infants with a gestational age of at least 20 weeks or birth weight of at least 500 g. Unique health card numbers were used to link birth records to a database of COVID vaccinations.
Of 85,162 infants born from May through December of 2021, 43,099 (50.6%) were born to individuals who received at least one vaccine dose during pregnancy. Among those, 99.7% received an mRNA vaccine such as Pfizer-BioNTech or Moderna.
Vaccination during pregnancy was not associated with greater risk of overall preterm birth (6.5% among vaccinated individuals versus 6.9% among unvaccinated; hazard ratio, 1.02; 95% confidence interval, 0.96-1.08), spontaneous preterm birth (3.7% versus 4.4%; hazard ratio, 0.96; 95% CI, 0.90-1.03) or very preterm birth (0.59% versus 0.89%; hazard ratio, 0.80; 95% CI, 0.67-0.95).
Likewise, no increase was observed in the risk of an infant being small for gestational age at birth (9.1% versus 9.2%; hazard ratio, 0.98; 95% CI, 0.93-1.03).
The researchers observed a reduction in the risk of stillbirth, even after adjusting for potential confounders. Stillbirths occurred in 0.25% of vaccinated individuals, compared with 0.44% of unvaccinated individuals (hazard ratio, 0.65; 95% CI, 0.51-0.84).
A reduced risk of stillbirth – albeit to a smaller degree – was also found in a Scandinavian registry study that included 28,506 babies born to individuals who were vaccinated during pregnancy.
“Collectively, the findings from these two studies are reassuring and are consistent with no increased risk of stillbirth after COVID-19 vaccination during pregnancy. In contrast, COVID-19 disease during pregnancy has been associated with an increased risk of stillbirth,” the researchers wrote.
Findings did not vary by which mRNA vaccine a mother received, the number of doses she received, or the trimester in which a vaccine was given, the researchers reported.
Stillbirth findings will be ‘very reassuring’ for patients
The lead investigator, Deshayne Fell, PhD, said in an interview, the fact that the study comprised the entire population of pregnant people in Ontario during the study period “increases our confidence” about the validity and relevance of the findings for other geographic settings.
Dr. Fell, an associate professor in epidemiology and public health at the University of Ottawa and a scientist at the Children’s Hospital of Eastern Ontario Research Institute, Ottawa, said the evaluation of stillbirth in particular, “a rare but devastating outcome,” will be “very reassuring and useful for clinical counseling.”
A limitation cited by the research team included a lack of data on vaccination prior to pregnancy.
In the new study, Dr, Ecker said, “Though the investigators were able to adjust for many variables they cannot be certain that some unmeasured variable that, accordingly, was not adjusted for does not hide a small risk. This seems very unlikely, however.”
The Canadian research team said similar studies of non-mRNA COVID vaccines “should be a research priority.” However, such studies are not underway in Canada, where only mRNA vaccines are used in pregnancy, Dr. Fell said.
This study was supported by the Public Health Agency of Canada.
Dr. Fell and Dr. Ecker reported no competing financial interests.
FROM BMJ
Acute otitis media pneumococcal disease burden in children due to serotypes not included in vaccines
My group in Rochester, N.Y., examined the current pneumococcal serotypes causing AOM in children. From our data, we can determine the PCV13 vaccine types that escape prevention and cause AOM and understand what effect to expect from the new pneumococcal conjugate vaccines (PCVs) that will be coming soon. There are limited data from middle ear fluid (MEF) cultures on which to base such analyses. Tympanocentesis is the preferred method for securing MEF for culture and our group is unique in providing such data to the Centers for Disease Control and publishing our results on a periodic basis to inform clinicians.
Pneumococci are the second most common cause of acute otitis media (AOM) since the introduction of pneumococcal conjugate vaccines (PCVs) more than 2 decades ago.1,2 Pneumococcal AOM causes more severe acute disease and more often causes suppurative complications than Haemophilus influenzae, which is the most common cause of AOM. Prevention of pneumococcal AOM will be a highly relevant contributor to cost-effectiveness analyses for the anticipated introduction of PCV15 (Merck) and PCV20 (Pfizer). Both PCV15 and PCV20 have been licensed for adult use; PCV15 licensure for infants and children occurred in June 2022 for invasive pneumococcal disease and is anticipated in the near future for PCV20. They are improvements over PCV13 because they add serotypes that cause invasive pneumococcal diseases, although less so for prevention of AOM, on the basis of our data.
Nasopharyngeal colonization is a necessary pathogenic step in progression to pneumococcal disease. However, not all strains of pneumococci expressing different capsular serotypes are equally virulent and likely to cause disease. In PCV-vaccinated populations, vaccine pressure and antibiotic resistance drive PCV serotype replacement with nonvaccine serotypes (NVTs), gradually reducing the net effectiveness of the vaccines. Therefore, knowledge of prevalent NVTs colonizing the nasopharynx identifies future pneumococcal serotypes most likely to emerge as pathogenic.
We published an effectiveness study of PCV13.3 A relative reduction of 86% in AOM caused by strains expressing PCV13 serotypes was observed in the first few years after PCV13 introduction. The greatest reduction in MEF samples was in serotype 19A, with a relative reduction of 91%. However, over time the vaccine type efficacy of PCV13 against MEF-positive pneumococcal AOM has eroded. There was no clear efficacy against serotype 3, and we still observed cases of serotype 19A and 19F. PCV13 vaccine failures have been even more frequent in Europe (nearly 30% of pneumococcal AOM in Europe is caused by vaccine serotypes) than our data indicate, where about 10% of AOM is caused by PCV13 serotypes.
In our most recent publication covering 2015-2019, we described results from 589 children, aged 6-36 months, from whom we collected 2,042 nasopharyngeal samples.2,4 During AOM, 495 MEF samples from 319 AOM-infected children were collected (during bilateral infections, tympanocentesis was performed in both ears). Whether bacteria were isolated was based per AOM case, not per tap. The average age of children with AOM was 15 months (range 6-31 months). The three most prevalent nasopharyngeal pneumococcal serotypes were 35B, 23B, and 15B/C. Serotype 35B was the most common at AOM visits in both the nasopharynx and MEF samples followed by serotype 15B/C. Nonsusceptibility among pneumococci to penicillin, azithromycin, and multiple other antibiotics was high. Increasing resistance to ceftriaxone was also observed.
Based on our results, if PCV15 (PCV13 + 22F and 33F) effectiveness is identical to PCV13 for the included serotypes and 100% efficacy for the added serotypes is presumed, PCV15 will reduce pneumococcal AOMs by 8%, pneumococcal nasopharyngeal colonization events at onset of AOM by 6%, and pneumococcal nasopharyngeal colonization events during health by 3%. As for the projected reductions brought about by PCV20 (PCV15 + 8, 10A, 11A, 12F, and 15B), presuming serotype 15B is efficacious against serotype 15C and 100% efficacy for the added serotypes, PCV20 will reduce pneumococcal AOMs by 22%, pneumococcal nasopharyngeal colonization events at onset of AOM by 20%, and pneumococcal nasopharyngeal colonization events during health by 3% (Figure).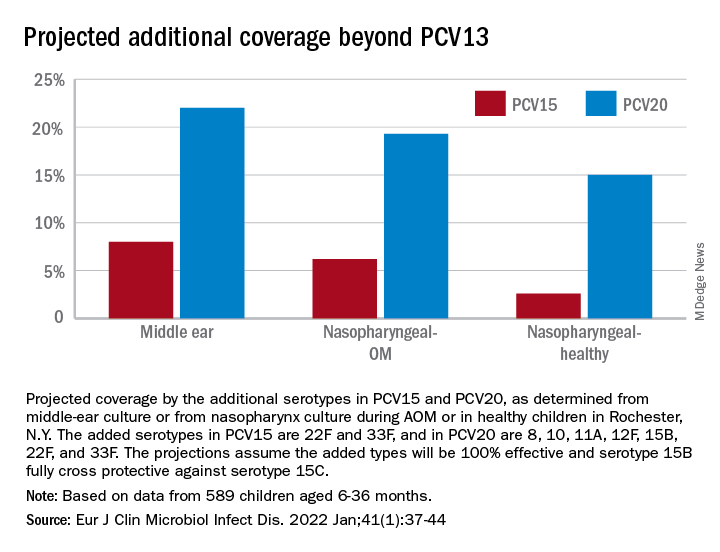
The CDC estimated that, in 2004, pneumococcal disease in the United States caused 4 million illness episodes, 22,000 deaths, 445,000 hospitalizations, 774,000 emergency department visits, 5 million outpatient visits, and 4.1 million outpatient antibiotic prescriptions. Direct medical costs totaled $3.5 billion. Pneumonia (866,000 cases) accounted for 22% of all cases and 72% of pneumococcal costs. AOM and sinusitis (1.5 million cases each) composed 75% of cases and 16% of direct medical costs.5 However, if indirect costs are taken into account, such as work loss by parents of young children, the cost of pneumococcal disease caused by AOM alone may exceed $6 billion annually6 and become dominant in the cost-effectiveness analysis in high-income countries.
Despite widespread use of PCV13, Pneumococcus has shown its resilience under vaccine pressure such that the organism remains a very common AOM pathogen. All-cause AOM has declined modestly and pneumococcal AOM caused by the specific serotypes in PCVs has declined dramatically since the introduction of PCVs. However, the burden of pneumococcal AOM disease is still considerable.
The notion that strains expressing serotypes that were not included in PCV7 were less virulent was proven wrong within a few years after introduction of PCV7, with the emergence of strains expressing serotype 19A, and others. The same cycle occurred after introduction of PCV13. It appears to take about 4 years after introduction of a PCV before peak effectiveness is achieved – which then begins to erode with emergence of NVTs. First, the NVTs are observed to colonize the nasopharynx as commensals and then from among those strains new disease-causing strains emerge.
At the most recent meeting of the International Society of Pneumococci and Pneumococcal Diseases in Toronto in June, many presentations focused on the fact that PCVs elicit highly effective protective serotype-specific antibodies to the capsular polysaccharides of included types. However, 100 serotypes are known. The limitations of PCVs are becoming increasingly apparent. They are costly and consume a large portion of the Vaccines for Children budget. Children in the developing world remain largely unvaccinated because of the high cost. NVTs that have emerged to cause disease vary by country, vary by adult vs. pediatric populations, and are dynamically changing year to year. Forthcoming PCVs of 15 and 20 serotypes will be even more costly than PCV13, will not include many newly emerged serotypes, and will probably likewise encounter “serotype replacement” because of high immune evasion by pneumococci.
When Merck and Pfizer made their decisions on serotype composition for PCV15 and PCV20, respectively, they were based on available data at the time regarding predominant serotypes causing invasive pneumococcal disease in countries that had the best data and would be the market for their products. However, from the time of the decision to licensure of vaccine is many years, and during that time the pneumococcal serotypes have changed, more so for AOM, and I predict more change will occur in the future.
In the past 3 years, Dr. Pichichero has received honoraria from Merck to attend 1-day consulting meetings and his institution has received investigator-initiated research grants to study aspects of PCV15. In the past 3 years, he was reimbursed for expenses to attend the ISPPD meeting in Toronto to present a poster on potential efficacy of PCV20 to prevent complicated AOM.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (N.Y.) General Hospital.
References
1. Kaur R et al. Pediatrics. 2017;140(3).
2. Kaur R et al. Eur J Clin Microbiol Infect Dis. 2021;41:37-44..
3. Pichichero M et al. Lancet Child Adolesc Health. 2018;2(8):561-8.
4. Zhou F et al. Pediatrics. 2008;121(2):253-60.
5. Huang SS et al. Vaccine. 2011;29(18):3398-412.
6. Casey JR and Pichichero ME. Clin Pediatr (Phila). 2014;53(9):865-73. .
My group in Rochester, N.Y., examined the current pneumococcal serotypes causing AOM in children. From our data, we can determine the PCV13 vaccine types that escape prevention and cause AOM and understand what effect to expect from the new pneumococcal conjugate vaccines (PCVs) that will be coming soon. There are limited data from middle ear fluid (MEF) cultures on which to base such analyses. Tympanocentesis is the preferred method for securing MEF for culture and our group is unique in providing such data to the Centers for Disease Control and publishing our results on a periodic basis to inform clinicians.
Pneumococci are the second most common cause of acute otitis media (AOM) since the introduction of pneumococcal conjugate vaccines (PCVs) more than 2 decades ago.1,2 Pneumococcal AOM causes more severe acute disease and more often causes suppurative complications than Haemophilus influenzae, which is the most common cause of AOM. Prevention of pneumococcal AOM will be a highly relevant contributor to cost-effectiveness analyses for the anticipated introduction of PCV15 (Merck) and PCV20 (Pfizer). Both PCV15 and PCV20 have been licensed for adult use; PCV15 licensure for infants and children occurred in June 2022 for invasive pneumococcal disease and is anticipated in the near future for PCV20. They are improvements over PCV13 because they add serotypes that cause invasive pneumococcal diseases, although less so for prevention of AOM, on the basis of our data.
Nasopharyngeal colonization is a necessary pathogenic step in progression to pneumococcal disease. However, not all strains of pneumococci expressing different capsular serotypes are equally virulent and likely to cause disease. In PCV-vaccinated populations, vaccine pressure and antibiotic resistance drive PCV serotype replacement with nonvaccine serotypes (NVTs), gradually reducing the net effectiveness of the vaccines. Therefore, knowledge of prevalent NVTs colonizing the nasopharynx identifies future pneumococcal serotypes most likely to emerge as pathogenic.
We published an effectiveness study of PCV13.3 A relative reduction of 86% in AOM caused by strains expressing PCV13 serotypes was observed in the first few years after PCV13 introduction. The greatest reduction in MEF samples was in serotype 19A, with a relative reduction of 91%. However, over time the vaccine type efficacy of PCV13 against MEF-positive pneumococcal AOM has eroded. There was no clear efficacy against serotype 3, and we still observed cases of serotype 19A and 19F. PCV13 vaccine failures have been even more frequent in Europe (nearly 30% of pneumococcal AOM in Europe is caused by vaccine serotypes) than our data indicate, where about 10% of AOM is caused by PCV13 serotypes.
In our most recent publication covering 2015-2019, we described results from 589 children, aged 6-36 months, from whom we collected 2,042 nasopharyngeal samples.2,4 During AOM, 495 MEF samples from 319 AOM-infected children were collected (during bilateral infections, tympanocentesis was performed in both ears). Whether bacteria were isolated was based per AOM case, not per tap. The average age of children with AOM was 15 months (range 6-31 months). The three most prevalent nasopharyngeal pneumococcal serotypes were 35B, 23B, and 15B/C. Serotype 35B was the most common at AOM visits in both the nasopharynx and MEF samples followed by serotype 15B/C. Nonsusceptibility among pneumococci to penicillin, azithromycin, and multiple other antibiotics was high. Increasing resistance to ceftriaxone was also observed.
Based on our results, if PCV15 (PCV13 + 22F and 33F) effectiveness is identical to PCV13 for the included serotypes and 100% efficacy for the added serotypes is presumed, PCV15 will reduce pneumococcal AOMs by 8%, pneumococcal nasopharyngeal colonization events at onset of AOM by 6%, and pneumococcal nasopharyngeal colonization events during health by 3%. As for the projected reductions brought about by PCV20 (PCV15 + 8, 10A, 11A, 12F, and 15B), presuming serotype 15B is efficacious against serotype 15C and 100% efficacy for the added serotypes, PCV20 will reduce pneumococcal AOMs by 22%, pneumococcal nasopharyngeal colonization events at onset of AOM by 20%, and pneumococcal nasopharyngeal colonization events during health by 3% (Figure).
The CDC estimated that, in 2004, pneumococcal disease in the United States caused 4 million illness episodes, 22,000 deaths, 445,000 hospitalizations, 774,000 emergency department visits, 5 million outpatient visits, and 4.1 million outpatient antibiotic prescriptions. Direct medical costs totaled $3.5 billion. Pneumonia (866,000 cases) accounted for 22% of all cases and 72% of pneumococcal costs. AOM and sinusitis (1.5 million cases each) composed 75% of cases and 16% of direct medical costs.5 However, if indirect costs are taken into account, such as work loss by parents of young children, the cost of pneumococcal disease caused by AOM alone may exceed $6 billion annually6 and become dominant in the cost-effectiveness analysis in high-income countries.
Despite widespread use of PCV13, Pneumococcus has shown its resilience under vaccine pressure such that the organism remains a very common AOM pathogen. All-cause AOM has declined modestly and pneumococcal AOM caused by the specific serotypes in PCVs has declined dramatically since the introduction of PCVs. However, the burden of pneumococcal AOM disease is still considerable.
The notion that strains expressing serotypes that were not included in PCV7 were less virulent was proven wrong within a few years after introduction of PCV7, with the emergence of strains expressing serotype 19A, and others. The same cycle occurred after introduction of PCV13. It appears to take about 4 years after introduction of a PCV before peak effectiveness is achieved – which then begins to erode with emergence of NVTs. First, the NVTs are observed to colonize the nasopharynx as commensals and then from among those strains new disease-causing strains emerge.
At the most recent meeting of the International Society of Pneumococci and Pneumococcal Diseases in Toronto in June, many presentations focused on the fact that PCVs elicit highly effective protective serotype-specific antibodies to the capsular polysaccharides of included types. However, 100 serotypes are known. The limitations of PCVs are becoming increasingly apparent. They are costly and consume a large portion of the Vaccines for Children budget. Children in the developing world remain largely unvaccinated because of the high cost. NVTs that have emerged to cause disease vary by country, vary by adult vs. pediatric populations, and are dynamically changing year to year. Forthcoming PCVs of 15 and 20 serotypes will be even more costly than PCV13, will not include many newly emerged serotypes, and will probably likewise encounter “serotype replacement” because of high immune evasion by pneumococci.
When Merck and Pfizer made their decisions on serotype composition for PCV15 and PCV20, respectively, they were based on available data at the time regarding predominant serotypes causing invasive pneumococcal disease in countries that had the best data and would be the market for their products. However, from the time of the decision to licensure of vaccine is many years, and during that time the pneumococcal serotypes have changed, more so for AOM, and I predict more change will occur in the future.
In the past 3 years, Dr. Pichichero has received honoraria from Merck to attend 1-day consulting meetings and his institution has received investigator-initiated research grants to study aspects of PCV15. In the past 3 years, he was reimbursed for expenses to attend the ISPPD meeting in Toronto to present a poster on potential efficacy of PCV20 to prevent complicated AOM.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (N.Y.) General Hospital.
References
1. Kaur R et al. Pediatrics. 2017;140(3).
2. Kaur R et al. Eur J Clin Microbiol Infect Dis. 2021;41:37-44..
3. Pichichero M et al. Lancet Child Adolesc Health. 2018;2(8):561-8.
4. Zhou F et al. Pediatrics. 2008;121(2):253-60.
5. Huang SS et al. Vaccine. 2011;29(18):3398-412.
6. Casey JR and Pichichero ME. Clin Pediatr (Phila). 2014;53(9):865-73. .
My group in Rochester, N.Y., examined the current pneumococcal serotypes causing AOM in children. From our data, we can determine the PCV13 vaccine types that escape prevention and cause AOM and understand what effect to expect from the new pneumococcal conjugate vaccines (PCVs) that will be coming soon. There are limited data from middle ear fluid (MEF) cultures on which to base such analyses. Tympanocentesis is the preferred method for securing MEF for culture and our group is unique in providing such data to the Centers for Disease Control and publishing our results on a periodic basis to inform clinicians.
Pneumococci are the second most common cause of acute otitis media (AOM) since the introduction of pneumococcal conjugate vaccines (PCVs) more than 2 decades ago.1,2 Pneumococcal AOM causes more severe acute disease and more often causes suppurative complications than Haemophilus influenzae, which is the most common cause of AOM. Prevention of pneumococcal AOM will be a highly relevant contributor to cost-effectiveness analyses for the anticipated introduction of PCV15 (Merck) and PCV20 (Pfizer). Both PCV15 and PCV20 have been licensed for adult use; PCV15 licensure for infants and children occurred in June 2022 for invasive pneumococcal disease and is anticipated in the near future for PCV20. They are improvements over PCV13 because they add serotypes that cause invasive pneumococcal diseases, although less so for prevention of AOM, on the basis of our data.
Nasopharyngeal colonization is a necessary pathogenic step in progression to pneumococcal disease. However, not all strains of pneumococci expressing different capsular serotypes are equally virulent and likely to cause disease. In PCV-vaccinated populations, vaccine pressure and antibiotic resistance drive PCV serotype replacement with nonvaccine serotypes (NVTs), gradually reducing the net effectiveness of the vaccines. Therefore, knowledge of prevalent NVTs colonizing the nasopharynx identifies future pneumococcal serotypes most likely to emerge as pathogenic.
We published an effectiveness study of PCV13.3 A relative reduction of 86% in AOM caused by strains expressing PCV13 serotypes was observed in the first few years after PCV13 introduction. The greatest reduction in MEF samples was in serotype 19A, with a relative reduction of 91%. However, over time the vaccine type efficacy of PCV13 against MEF-positive pneumococcal AOM has eroded. There was no clear efficacy against serotype 3, and we still observed cases of serotype 19A and 19F. PCV13 vaccine failures have been even more frequent in Europe (nearly 30% of pneumococcal AOM in Europe is caused by vaccine serotypes) than our data indicate, where about 10% of AOM is caused by PCV13 serotypes.
In our most recent publication covering 2015-2019, we described results from 589 children, aged 6-36 months, from whom we collected 2,042 nasopharyngeal samples.2,4 During AOM, 495 MEF samples from 319 AOM-infected children were collected (during bilateral infections, tympanocentesis was performed in both ears). Whether bacteria were isolated was based per AOM case, not per tap. The average age of children with AOM was 15 months (range 6-31 months). The three most prevalent nasopharyngeal pneumococcal serotypes were 35B, 23B, and 15B/C. Serotype 35B was the most common at AOM visits in both the nasopharynx and MEF samples followed by serotype 15B/C. Nonsusceptibility among pneumococci to penicillin, azithromycin, and multiple other antibiotics was high. Increasing resistance to ceftriaxone was also observed.
Based on our results, if PCV15 (PCV13 + 22F and 33F) effectiveness is identical to PCV13 for the included serotypes and 100% efficacy for the added serotypes is presumed, PCV15 will reduce pneumococcal AOMs by 8%, pneumococcal nasopharyngeal colonization events at onset of AOM by 6%, and pneumococcal nasopharyngeal colonization events during health by 3%. As for the projected reductions brought about by PCV20 (PCV15 + 8, 10A, 11A, 12F, and 15B), presuming serotype 15B is efficacious against serotype 15C and 100% efficacy for the added serotypes, PCV20 will reduce pneumococcal AOMs by 22%, pneumococcal nasopharyngeal colonization events at onset of AOM by 20%, and pneumococcal nasopharyngeal colonization events during health by 3% (Figure).
The CDC estimated that, in 2004, pneumococcal disease in the United States caused 4 million illness episodes, 22,000 deaths, 445,000 hospitalizations, 774,000 emergency department visits, 5 million outpatient visits, and 4.1 million outpatient antibiotic prescriptions. Direct medical costs totaled $3.5 billion. Pneumonia (866,000 cases) accounted for 22% of all cases and 72% of pneumococcal costs. AOM and sinusitis (1.5 million cases each) composed 75% of cases and 16% of direct medical costs.5 However, if indirect costs are taken into account, such as work loss by parents of young children, the cost of pneumococcal disease caused by AOM alone may exceed $6 billion annually6 and become dominant in the cost-effectiveness analysis in high-income countries.
Despite widespread use of PCV13, Pneumococcus has shown its resilience under vaccine pressure such that the organism remains a very common AOM pathogen. All-cause AOM has declined modestly and pneumococcal AOM caused by the specific serotypes in PCVs has declined dramatically since the introduction of PCVs. However, the burden of pneumococcal AOM disease is still considerable.
The notion that strains expressing serotypes that were not included in PCV7 were less virulent was proven wrong within a few years after introduction of PCV7, with the emergence of strains expressing serotype 19A, and others. The same cycle occurred after introduction of PCV13. It appears to take about 4 years after introduction of a PCV before peak effectiveness is achieved – which then begins to erode with emergence of NVTs. First, the NVTs are observed to colonize the nasopharynx as commensals and then from among those strains new disease-causing strains emerge.
At the most recent meeting of the International Society of Pneumococci and Pneumococcal Diseases in Toronto in June, many presentations focused on the fact that PCVs elicit highly effective protective serotype-specific antibodies to the capsular polysaccharides of included types. However, 100 serotypes are known. The limitations of PCVs are becoming increasingly apparent. They are costly and consume a large portion of the Vaccines for Children budget. Children in the developing world remain largely unvaccinated because of the high cost. NVTs that have emerged to cause disease vary by country, vary by adult vs. pediatric populations, and are dynamically changing year to year. Forthcoming PCVs of 15 and 20 serotypes will be even more costly than PCV13, will not include many newly emerged serotypes, and will probably likewise encounter “serotype replacement” because of high immune evasion by pneumococci.
When Merck and Pfizer made their decisions on serotype composition for PCV15 and PCV20, respectively, they were based on available data at the time regarding predominant serotypes causing invasive pneumococcal disease in countries that had the best data and would be the market for their products. However, from the time of the decision to licensure of vaccine is many years, and during that time the pneumococcal serotypes have changed, more so for AOM, and I predict more change will occur in the future.
In the past 3 years, Dr. Pichichero has received honoraria from Merck to attend 1-day consulting meetings and his institution has received investigator-initiated research grants to study aspects of PCV15. In the past 3 years, he was reimbursed for expenses to attend the ISPPD meeting in Toronto to present a poster on potential efficacy of PCV20 to prevent complicated AOM.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (N.Y.) General Hospital.
References
1. Kaur R et al. Pediatrics. 2017;140(3).
2. Kaur R et al. Eur J Clin Microbiol Infect Dis. 2021;41:37-44..
3. Pichichero M et al. Lancet Child Adolesc Health. 2018;2(8):561-8.
4. Zhou F et al. Pediatrics. 2008;121(2):253-60.
5. Huang SS et al. Vaccine. 2011;29(18):3398-412.
6. Casey JR and Pichichero ME. Clin Pediatr (Phila). 2014;53(9):865-73. .
NYC switching children’s COVID vaccine sites to monkeypox
The city health department said demand for children’s COVID vaccines had been on the downswing at the clinics, which opened in late June. Meanwhile, monkeypox cases have increased, with the city declaring it a public health emergency July 30.
“We always planned to transition vaccination for very young children to providers,” the city’s health department said in a statement, according to Spectrum News NY1. “Due to the ongoing monkeypox emergency, we transitioned some of these sites to administer monkeypox vaccine.”
All the COVID vaccine sites for children will close by Aug. 14, Spectrum News NY1 said. It’s unclear if the other sites will transition to monkeypox vaccine.
No appointments for children’s COVID vaccinations had to be canceled, the city said. The plan is that children now needing the COVID vaccine can go to doctors, pharmacies, or the health department clinics.
Manhattan City Councilwoman Gale Brewer urged the health department to keep the kids’ COVID vaccine sites open through the fall.
“I strongly urge you to maintain these family-friendly sites, at least until mid-September so that children who are going to day care and school can get vaccinated,” Brewer wrote. City schools open Sept. 8
Ms. Brewer noted that the city-run sites administered the Moderna vaccines, while many doctors and neighborhood health clinics use the Pfizer vaccine. That could be a problem for a child that had not finished the Moderna regimen or for families that prefer Moderna.
According to the city health department, 2,130 people in New York City had tested positive for monkeypox as of Aug. 12.
On Friday, the city announced 9,000 additional monkeypox vaccines would be made available the morning of Aug. 13.
A version of this article first appeared on WebMD.com.
The city health department said demand for children’s COVID vaccines had been on the downswing at the clinics, which opened in late June. Meanwhile, monkeypox cases have increased, with the city declaring it a public health emergency July 30.
“We always planned to transition vaccination for very young children to providers,” the city’s health department said in a statement, according to Spectrum News NY1. “Due to the ongoing monkeypox emergency, we transitioned some of these sites to administer monkeypox vaccine.”
All the COVID vaccine sites for children will close by Aug. 14, Spectrum News NY1 said. It’s unclear if the other sites will transition to monkeypox vaccine.
No appointments for children’s COVID vaccinations had to be canceled, the city said. The plan is that children now needing the COVID vaccine can go to doctors, pharmacies, or the health department clinics.
Manhattan City Councilwoman Gale Brewer urged the health department to keep the kids’ COVID vaccine sites open through the fall.
“I strongly urge you to maintain these family-friendly sites, at least until mid-September so that children who are going to day care and school can get vaccinated,” Brewer wrote. City schools open Sept. 8
Ms. Brewer noted that the city-run sites administered the Moderna vaccines, while many doctors and neighborhood health clinics use the Pfizer vaccine. That could be a problem for a child that had not finished the Moderna regimen or for families that prefer Moderna.
According to the city health department, 2,130 people in New York City had tested positive for monkeypox as of Aug. 12.
On Friday, the city announced 9,000 additional monkeypox vaccines would be made available the morning of Aug. 13.
A version of this article first appeared on WebMD.com.
The city health department said demand for children’s COVID vaccines had been on the downswing at the clinics, which opened in late June. Meanwhile, monkeypox cases have increased, with the city declaring it a public health emergency July 30.
“We always planned to transition vaccination for very young children to providers,” the city’s health department said in a statement, according to Spectrum News NY1. “Due to the ongoing monkeypox emergency, we transitioned some of these sites to administer monkeypox vaccine.”
All the COVID vaccine sites for children will close by Aug. 14, Spectrum News NY1 said. It’s unclear if the other sites will transition to monkeypox vaccine.
No appointments for children’s COVID vaccinations had to be canceled, the city said. The plan is that children now needing the COVID vaccine can go to doctors, pharmacies, or the health department clinics.
Manhattan City Councilwoman Gale Brewer urged the health department to keep the kids’ COVID vaccine sites open through the fall.
“I strongly urge you to maintain these family-friendly sites, at least until mid-September so that children who are going to day care and school can get vaccinated,” Brewer wrote. City schools open Sept. 8
Ms. Brewer noted that the city-run sites administered the Moderna vaccines, while many doctors and neighborhood health clinics use the Pfizer vaccine. That could be a problem for a child that had not finished the Moderna regimen or for families that prefer Moderna.
According to the city health department, 2,130 people in New York City had tested positive for monkeypox as of Aug. 12.
On Friday, the city announced 9,000 additional monkeypox vaccines would be made available the morning of Aug. 13.
A version of this article first appeared on WebMD.com.
Seniors intend to receive variant-specific COVID booster in coming months
of 2022.
That finding comes from a new poll by researchers at the University of Michigan, Ann Arbor, who also report that when it comes to the shots, people appear to be putting more trust in their health care professionals than in public health authorities.
“When you are a doctor, you are a trusted source of medical information,” said Preeti Malani, MD, MSJ, an infectious disease specialist at the University of Michigan. “Use the ongoing conversation with your patient as an opportunity to answer their questions and counter any confusion.”
The vaccination campaign appears to be having a rub-off effect, too. More people say they’re likely to receive vaccines and boosters for other infections, such as flu, if they have already been vaccinated and boosted against COVID-19.
Inside the poll
Dr. Malani and her colleagues, who published their findings on the National Poll on Healthy Aging’s website, asked 1,024 adults older than 50 about their attitudes on COVID-19 vaccinations and their history of receiving the injections. The questions covered topics including whether the individual had contracted COVID, COVID vaccine doses, and the prevalence of a health care clinician’s opinion on vaccines and boosters. The poll was conducted July 21-26.
The researchers chose the age range of 50-65 years because this group is an important population for new booster shots that target specific variants of the SARS-CoV-2 virus that causes COVID-19.
Only 19% of people aged 50-64 and 44% of those older than 65 said they had received both their first and second COVID-19 booster shots. What’s more, 17% of people said they had not received any doses of a COVID-19 vaccine.
The vast majority (77%) of respondents said their clinician’s recommendations were “very important” or “somewhat important” in their decision to receive the vaccine.
Dr. Malani said that in her practice, patients have expressed hesitation about COVID-19 vaccines because of concerns about the potential side effects of the shots.
Monica Gandhi, MD, MPH, professor of medicine at the University of California, San Francisco, noted that Americans now appear to trust their physicians more than public health authorities such as the U.S. Centers for Disease Control and Prevention when it comes to COVID-19.
“More people are trusting their providers’ opinions [more] than the CDC or other public health agencies. That speaks volumes to me,” Dr. Gandhi said.
Among the more surprising findings of the poll, according to the researchers, was the number of people who said they had yet to contract COVID-19: 50% of those aged 50-64, and 69% of those older than 65. (Another 12% of those aged 50-64 said they were unsure if they’d ever had the infection.)
Dr. Malani said she hoped future studies would explore in depth the people who remain uninfected with COVID-19.
“We focus a lot on the science of COVID,” she said. “But we need to turn our attention to the behavioral aspects and how to address them.”
A version of this article first appeared on Medscape.com.
of 2022.
That finding comes from a new poll by researchers at the University of Michigan, Ann Arbor, who also report that when it comes to the shots, people appear to be putting more trust in their health care professionals than in public health authorities.
“When you are a doctor, you are a trusted source of medical information,” said Preeti Malani, MD, MSJ, an infectious disease specialist at the University of Michigan. “Use the ongoing conversation with your patient as an opportunity to answer their questions and counter any confusion.”
The vaccination campaign appears to be having a rub-off effect, too. More people say they’re likely to receive vaccines and boosters for other infections, such as flu, if they have already been vaccinated and boosted against COVID-19.
Inside the poll
Dr. Malani and her colleagues, who published their findings on the National Poll on Healthy Aging’s website, asked 1,024 adults older than 50 about their attitudes on COVID-19 vaccinations and their history of receiving the injections. The questions covered topics including whether the individual had contracted COVID, COVID vaccine doses, and the prevalence of a health care clinician’s opinion on vaccines and boosters. The poll was conducted July 21-26.
The researchers chose the age range of 50-65 years because this group is an important population for new booster shots that target specific variants of the SARS-CoV-2 virus that causes COVID-19.
Only 19% of people aged 50-64 and 44% of those older than 65 said they had received both their first and second COVID-19 booster shots. What’s more, 17% of people said they had not received any doses of a COVID-19 vaccine.
The vast majority (77%) of respondents said their clinician’s recommendations were “very important” or “somewhat important” in their decision to receive the vaccine.
Dr. Malani said that in her practice, patients have expressed hesitation about COVID-19 vaccines because of concerns about the potential side effects of the shots.
Monica Gandhi, MD, MPH, professor of medicine at the University of California, San Francisco, noted that Americans now appear to trust their physicians more than public health authorities such as the U.S. Centers for Disease Control and Prevention when it comes to COVID-19.
“More people are trusting their providers’ opinions [more] than the CDC or other public health agencies. That speaks volumes to me,” Dr. Gandhi said.
Among the more surprising findings of the poll, according to the researchers, was the number of people who said they had yet to contract COVID-19: 50% of those aged 50-64, and 69% of those older than 65. (Another 12% of those aged 50-64 said they were unsure if they’d ever had the infection.)
Dr. Malani said she hoped future studies would explore in depth the people who remain uninfected with COVID-19.
“We focus a lot on the science of COVID,” she said. “But we need to turn our attention to the behavioral aspects and how to address them.”
A version of this article first appeared on Medscape.com.
of 2022.
That finding comes from a new poll by researchers at the University of Michigan, Ann Arbor, who also report that when it comes to the shots, people appear to be putting more trust in their health care professionals than in public health authorities.
“When you are a doctor, you are a trusted source of medical information,” said Preeti Malani, MD, MSJ, an infectious disease specialist at the University of Michigan. “Use the ongoing conversation with your patient as an opportunity to answer their questions and counter any confusion.”
The vaccination campaign appears to be having a rub-off effect, too. More people say they’re likely to receive vaccines and boosters for other infections, such as flu, if they have already been vaccinated and boosted against COVID-19.
Inside the poll
Dr. Malani and her colleagues, who published their findings on the National Poll on Healthy Aging’s website, asked 1,024 adults older than 50 about their attitudes on COVID-19 vaccinations and their history of receiving the injections. The questions covered topics including whether the individual had contracted COVID, COVID vaccine doses, and the prevalence of a health care clinician’s opinion on vaccines and boosters. The poll was conducted July 21-26.
The researchers chose the age range of 50-65 years because this group is an important population for new booster shots that target specific variants of the SARS-CoV-2 virus that causes COVID-19.
Only 19% of people aged 50-64 and 44% of those older than 65 said they had received both their first and second COVID-19 booster shots. What’s more, 17% of people said they had not received any doses of a COVID-19 vaccine.
The vast majority (77%) of respondents said their clinician’s recommendations were “very important” or “somewhat important” in their decision to receive the vaccine.
Dr. Malani said that in her practice, patients have expressed hesitation about COVID-19 vaccines because of concerns about the potential side effects of the shots.
Monica Gandhi, MD, MPH, professor of medicine at the University of California, San Francisco, noted that Americans now appear to trust their physicians more than public health authorities such as the U.S. Centers for Disease Control and Prevention when it comes to COVID-19.
“More people are trusting their providers’ opinions [more] than the CDC or other public health agencies. That speaks volumes to me,” Dr. Gandhi said.
Among the more surprising findings of the poll, according to the researchers, was the number of people who said they had yet to contract COVID-19: 50% of those aged 50-64, and 69% of those older than 65. (Another 12% of those aged 50-64 said they were unsure if they’d ever had the infection.)
Dr. Malani said she hoped future studies would explore in depth the people who remain uninfected with COVID-19.
“We focus a lot on the science of COVID,” she said. “But we need to turn our attention to the behavioral aspects and how to address them.”
A version of this article first appeared on Medscape.com.
Federal Health Care Data Trends 2022
Federal Health Care Data Trends (click to view the digital edition) is a special supplement to Federal Practitioner highlighting the latest research and study outcomes related to the health of veteran and active-duty populations.
In this issue:
- Vaccinations
- Mental Health and Related Disorders
- LGBTQ+ Veterans
- Military Sexual Trauma
- Sleep Disorders
- Respiratory Illnesses
- HIV Care in the VA
- Rheumatologic Diseases
- The Cancer-Obesity Connection
- Skin Health for Active-Duty Personnel
- Contraception
- Chronic Kidney Disease
- Cardiovascular Diseases
- Neurologic Disorders
- Hearing, Vision, and Balance
Federal Practitioner would like to thank the following experts for their review of content and helpful guidance in developing this issue:
Kelvin N.V. Bush, MD, FACC, CCDS; Sonya Borrero, MD, MS; Kenneth L. Cameron, PhD, MPH, ATC, FNATA; Jason DeViva, PhD; Ellen Lockard Edens, MD; Leonard E. Egede, MD, MS; Amy Justice, MD, PhD; Stephanie Knudson, MD; Willis H. Lyford, MD; Sarah O. Meadows, PhD; Tamara Schult, PhD, MPH; Eric L. Singman, MD, PhD; Art Wallace, MD, PhD; Elizabeth Waterhouse, MD, FAAN
Federal Health Care Data Trends (click to view the digital edition) is a special supplement to Federal Practitioner highlighting the latest research and study outcomes related to the health of veteran and active-duty populations.
In this issue:
- Vaccinations
- Mental Health and Related Disorders
- LGBTQ+ Veterans
- Military Sexual Trauma
- Sleep Disorders
- Respiratory Illnesses
- HIV Care in the VA
- Rheumatologic Diseases
- The Cancer-Obesity Connection
- Skin Health for Active-Duty Personnel
- Contraception
- Chronic Kidney Disease
- Cardiovascular Diseases
- Neurologic Disorders
- Hearing, Vision, and Balance
Federal Practitioner would like to thank the following experts for their review of content and helpful guidance in developing this issue:
Kelvin N.V. Bush, MD, FACC, CCDS; Sonya Borrero, MD, MS; Kenneth L. Cameron, PhD, MPH, ATC, FNATA; Jason DeViva, PhD; Ellen Lockard Edens, MD; Leonard E. Egede, MD, MS; Amy Justice, MD, PhD; Stephanie Knudson, MD; Willis H. Lyford, MD; Sarah O. Meadows, PhD; Tamara Schult, PhD, MPH; Eric L. Singman, MD, PhD; Art Wallace, MD, PhD; Elizabeth Waterhouse, MD, FAAN
Federal Health Care Data Trends (click to view the digital edition) is a special supplement to Federal Practitioner highlighting the latest research and study outcomes related to the health of veteran and active-duty populations.
In this issue:
- Vaccinations
- Mental Health and Related Disorders
- LGBTQ+ Veterans
- Military Sexual Trauma
- Sleep Disorders
- Respiratory Illnesses
- HIV Care in the VA
- Rheumatologic Diseases
- The Cancer-Obesity Connection
- Skin Health for Active-Duty Personnel
- Contraception
- Chronic Kidney Disease
- Cardiovascular Diseases
- Neurologic Disorders
- Hearing, Vision, and Balance
Federal Practitioner would like to thank the following experts for their review of content and helpful guidance in developing this issue:
Kelvin N.V. Bush, MD, FACC, CCDS; Sonya Borrero, MD, MS; Kenneth L. Cameron, PhD, MPH, ATC, FNATA; Jason DeViva, PhD; Ellen Lockard Edens, MD; Leonard E. Egede, MD, MS; Amy Justice, MD, PhD; Stephanie Knudson, MD; Willis H. Lyford, MD; Sarah O. Meadows, PhD; Tamara Schult, PhD, MPH; Eric L. Singman, MD, PhD; Art Wallace, MD, PhD; Elizabeth Waterhouse, MD, FAAN
FDA authorizes intradermal use of Jynneos vaccine for monkeypox
The Food and Drug Administration on Aug. 9 authorized intradermal administration of the Jynneos vaccine for the treatment of monkeypox. The process, approved specifically for high-risk patients, was passed under the administration’s Emergency Use Authorization. It follows the decision on Aug. 4 by the U.S. Department of Health and Human Services to declare monkeypox a public health emergency. Intradermal administration will allow providers to get five doses out of a one-dose vial.
This news organization will update this article as more information becomes available.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration on Aug. 9 authorized intradermal administration of the Jynneos vaccine for the treatment of monkeypox. The process, approved specifically for high-risk patients, was passed under the administration’s Emergency Use Authorization. It follows the decision on Aug. 4 by the U.S. Department of Health and Human Services to declare monkeypox a public health emergency. Intradermal administration will allow providers to get five doses out of a one-dose vial.
This news organization will update this article as more information becomes available.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration on Aug. 9 authorized intradermal administration of the Jynneos vaccine for the treatment of monkeypox. The process, approved specifically for high-risk patients, was passed under the administration’s Emergency Use Authorization. It follows the decision on Aug. 4 by the U.S. Department of Health and Human Services to declare monkeypox a public health emergency. Intradermal administration will allow providers to get five doses out of a one-dose vial.
This news organization will update this article as more information becomes available.
A version of this article first appeared on Medscape.com.
How well do vaccines protect against long COVID?
New York City veterinarian Erin Kulick used to be a weekend warrior. Only 2½ years ago, the 38-year-old new mother played ultimate Frisbee and flag football with friends. She went for regular 30-minute runs to burn off stress.
Now, Dr. Kulick is usually so exhausted, she can’t walk nonstop for 15 minutes. She recently tried to take her 4-year-old son, Cooper, to the American Museum of Natural History for his first visit, but ended up on a bench outside the museum, sobbing in the rain, because she couldn’t even get through the first hurdle of standing in line. “I just wanted to be there with my kid,” she said.
Dr. Kulick got sick with COVID-19 at the start of the pandemic in March 2020, 9 months before the first vaccine would be approved. Now she is among the estimated one in five infected Americans, or 19%, whose symptoms developed into long COVID.
Dr. Kulick also is now vaccinated and boosted. Had a vaccine been available sooner, could it have protected her from long COVID?
Evidence is starting to show it’s likely.
“The best way not to have long COVID is not to have COVID at all,” said Leora Horwitz, MD, a professor of population health and medicine at New York University. “To the extent that vaccination can prevent you from getting COVID at all, then it helps to reduce long COVID.”
And People with more serious initial illness appear more likely to have prolonged symptoms, but those with milder disease can certainly get it, too.
“You’re more likely to have long COVID with more severe disease, and we have ample evidence that vaccination reduces the severity of disease,” Dr. Horwitz said. “We also now have quite a lot of evidence that vaccination does reduce your risk of long COVID – probably because it reduces your risk of severe disease.”
There is little consensus about how much vaccines can lower the risk of long-term COVID symptoms, but several studies suggest that number lies anywhere from 15% to more than 80%.
That might seem like a big variation, but infectious disease experts argue that trying to interpret the gap isn’t as important as noticing what’s consistent across all these studies: “Vaccines do offer some protection, but it’s incomplete,” said Ziyad Al-Aly, MD, chief of research and development at the Veterans Affairs St. Louis Health Care System. Dr. Al-Aly, who has led several large studies on long COVID, said focusing on the fact that vaccines do offer some protection is a much better public health message than looking at the different levels of risk.
“Vaccines do a miraculous job for what they were designed to do,” said Dr. Al-Aly. “Vaccines were designed to reduce the risk of hospitalization ... and for that, vaccines are still holding up, even with all the changes in the virus.”
Still, Elena Azzolini, MD, PhD, head of the Humanitas Research Hospital’s vaccination center in Milan, thinks some studies may have underestimated the level of long COVID protection from vaccines because of limits in the study methods, such as not including enough women, who are more affected by long COVID. Her recent study, which looked at 2,560 health care professionals working in nine Italian centers from March 2020 to April 2022, focused on the risk for healthy women and men in their 20s to their 70s.
In the paper, Dr. Azzolini and associates reported that two or three doses of vaccine reduced the risk of hospitalization from COVID-19 from 42% among those who are unvaccinated to 16%-17%. In other words, they found unvaccinated people in the study were nearly three times as likely to have serious symptoms for longer than 4 weeks.
But Dr. Azzolini and Dr. Al-Aly still say that, even for the vaccinated, as long as COVID is around, masks are necessary. That’s because current vaccines don’t do enough to reduce transmission, said Dr. Al-Aly. “The only way that can really help [stop] transmission is covering our nose and mouth with a mask.”
How vaccinations affect people who already have long COVID
Some long COVID patients have said they got better after they get boosted, while some say they’re getting worse, said Dr. Horwitz, who is also a lead investigator at the National Institutes of Health’s flagship RECOVER program, a 4-year research project to study long COVID across the United States. (The NIH is still recruiting volunteers for these studies, which are also open to people who have never had COVID.)
One study published in the British Medical Journal analyzed survey data of more than 28,000 people infected with COVID in the United Kingdom and found a 13% reduction in long-term symptoms after a first dose of the vaccine, although it was unclear from the data if the improvement was sustained.
A second dose was associated with another 8% improvement over a 2-month period. “It’s reassuring that we see an average modest improvement in symptoms, not an average worsening in symptoms,” said Daniel Ayoubkhani, principal statistician at the U.K. Office for National Statistics and lead author of the study. Of course, the experience will differ among different people.
“It doesn’t appear that vaccination is the silver bullet that’s going to eradicate long COVID,” he said, but evidence from multiple studies suggests vaccines may help people with long-term symptoms.
Akiko Iwasaki, PhD, an immunobiologist at Yale University, New Haven, Conn., told a White House summit in July that one of the best ways to prevent long COVID is to develop the next generation of vaccines that also prevent milder cases by blocking transmission in the first place.
Back in New York, Dr. Kulick is now triple vaccinated. She’s due for a fourth dose soon but admits she’s “terrified every time” that she’s going to get sicker.
In her Facebook support group for long COVID, she reads that most people with prolonged symptoms handle it well. She has also noticed some of her symptoms eased after her first two doses of vaccine.
Since being diagnosed, Dr. Kulick learned she has a genetic condition, Ehlers-Danlos syndrome, which affects connective tissues that support skin, joints, organs, and blood vessels, and which her doctors say may have made her more prone to long COVID. She’s also being screened for autoimmune diseases, but for now, the only relief she has found has come from long COVID physical therapy, changes to her diet, and integrative medicine.
Dr. Kulick is still trying to figure out how she can get better while keeping her long hours at her veterinary job – and her health benefits. She is thankful her husband is a devoted caregiver to their son and a professional jazz musician with a schedule that allows for some flexibility.
“But it’s really hard when every week feels like I’ve run a marathon,” she said. “I can barely make it through.”
A version of this article first appeared on WebMD.com.
New York City veterinarian Erin Kulick used to be a weekend warrior. Only 2½ years ago, the 38-year-old new mother played ultimate Frisbee and flag football with friends. She went for regular 30-minute runs to burn off stress.
Now, Dr. Kulick is usually so exhausted, she can’t walk nonstop for 15 minutes. She recently tried to take her 4-year-old son, Cooper, to the American Museum of Natural History for his first visit, but ended up on a bench outside the museum, sobbing in the rain, because she couldn’t even get through the first hurdle of standing in line. “I just wanted to be there with my kid,” she said.
Dr. Kulick got sick with COVID-19 at the start of the pandemic in March 2020, 9 months before the first vaccine would be approved. Now she is among the estimated one in five infected Americans, or 19%, whose symptoms developed into long COVID.
Dr. Kulick also is now vaccinated and boosted. Had a vaccine been available sooner, could it have protected her from long COVID?
Evidence is starting to show it’s likely.
“The best way not to have long COVID is not to have COVID at all,” said Leora Horwitz, MD, a professor of population health and medicine at New York University. “To the extent that vaccination can prevent you from getting COVID at all, then it helps to reduce long COVID.”
And People with more serious initial illness appear more likely to have prolonged symptoms, but those with milder disease can certainly get it, too.
“You’re more likely to have long COVID with more severe disease, and we have ample evidence that vaccination reduces the severity of disease,” Dr. Horwitz said. “We also now have quite a lot of evidence that vaccination does reduce your risk of long COVID – probably because it reduces your risk of severe disease.”
There is little consensus about how much vaccines can lower the risk of long-term COVID symptoms, but several studies suggest that number lies anywhere from 15% to more than 80%.
That might seem like a big variation, but infectious disease experts argue that trying to interpret the gap isn’t as important as noticing what’s consistent across all these studies: “Vaccines do offer some protection, but it’s incomplete,” said Ziyad Al-Aly, MD, chief of research and development at the Veterans Affairs St. Louis Health Care System. Dr. Al-Aly, who has led several large studies on long COVID, said focusing on the fact that vaccines do offer some protection is a much better public health message than looking at the different levels of risk.
“Vaccines do a miraculous job for what they were designed to do,” said Dr. Al-Aly. “Vaccines were designed to reduce the risk of hospitalization ... and for that, vaccines are still holding up, even with all the changes in the virus.”
Still, Elena Azzolini, MD, PhD, head of the Humanitas Research Hospital’s vaccination center in Milan, thinks some studies may have underestimated the level of long COVID protection from vaccines because of limits in the study methods, such as not including enough women, who are more affected by long COVID. Her recent study, which looked at 2,560 health care professionals working in nine Italian centers from March 2020 to April 2022, focused on the risk for healthy women and men in their 20s to their 70s.
In the paper, Dr. Azzolini and associates reported that two or three doses of vaccine reduced the risk of hospitalization from COVID-19 from 42% among those who are unvaccinated to 16%-17%. In other words, they found unvaccinated people in the study were nearly three times as likely to have serious symptoms for longer than 4 weeks.
But Dr. Azzolini and Dr. Al-Aly still say that, even for the vaccinated, as long as COVID is around, masks are necessary. That’s because current vaccines don’t do enough to reduce transmission, said Dr. Al-Aly. “The only way that can really help [stop] transmission is covering our nose and mouth with a mask.”
How vaccinations affect people who already have long COVID
Some long COVID patients have said they got better after they get boosted, while some say they’re getting worse, said Dr. Horwitz, who is also a lead investigator at the National Institutes of Health’s flagship RECOVER program, a 4-year research project to study long COVID across the United States. (The NIH is still recruiting volunteers for these studies, which are also open to people who have never had COVID.)
One study published in the British Medical Journal analyzed survey data of more than 28,000 people infected with COVID in the United Kingdom and found a 13% reduction in long-term symptoms after a first dose of the vaccine, although it was unclear from the data if the improvement was sustained.
A second dose was associated with another 8% improvement over a 2-month period. “It’s reassuring that we see an average modest improvement in symptoms, not an average worsening in symptoms,” said Daniel Ayoubkhani, principal statistician at the U.K. Office for National Statistics and lead author of the study. Of course, the experience will differ among different people.
“It doesn’t appear that vaccination is the silver bullet that’s going to eradicate long COVID,” he said, but evidence from multiple studies suggests vaccines may help people with long-term symptoms.
Akiko Iwasaki, PhD, an immunobiologist at Yale University, New Haven, Conn., told a White House summit in July that one of the best ways to prevent long COVID is to develop the next generation of vaccines that also prevent milder cases by blocking transmission in the first place.
Back in New York, Dr. Kulick is now triple vaccinated. She’s due for a fourth dose soon but admits she’s “terrified every time” that she’s going to get sicker.
In her Facebook support group for long COVID, she reads that most people with prolonged symptoms handle it well. She has also noticed some of her symptoms eased after her first two doses of vaccine.
Since being diagnosed, Dr. Kulick learned she has a genetic condition, Ehlers-Danlos syndrome, which affects connective tissues that support skin, joints, organs, and blood vessels, and which her doctors say may have made her more prone to long COVID. She’s also being screened for autoimmune diseases, but for now, the only relief she has found has come from long COVID physical therapy, changes to her diet, and integrative medicine.
Dr. Kulick is still trying to figure out how she can get better while keeping her long hours at her veterinary job – and her health benefits. She is thankful her husband is a devoted caregiver to their son and a professional jazz musician with a schedule that allows for some flexibility.
“But it’s really hard when every week feels like I’ve run a marathon,” she said. “I can barely make it through.”
A version of this article first appeared on WebMD.com.
New York City veterinarian Erin Kulick used to be a weekend warrior. Only 2½ years ago, the 38-year-old new mother played ultimate Frisbee and flag football with friends. She went for regular 30-minute runs to burn off stress.
Now, Dr. Kulick is usually so exhausted, she can’t walk nonstop for 15 minutes. She recently tried to take her 4-year-old son, Cooper, to the American Museum of Natural History for his first visit, but ended up on a bench outside the museum, sobbing in the rain, because she couldn’t even get through the first hurdle of standing in line. “I just wanted to be there with my kid,” she said.
Dr. Kulick got sick with COVID-19 at the start of the pandemic in March 2020, 9 months before the first vaccine would be approved. Now she is among the estimated one in five infected Americans, or 19%, whose symptoms developed into long COVID.
Dr. Kulick also is now vaccinated and boosted. Had a vaccine been available sooner, could it have protected her from long COVID?
Evidence is starting to show it’s likely.
“The best way not to have long COVID is not to have COVID at all,” said Leora Horwitz, MD, a professor of population health and medicine at New York University. “To the extent that vaccination can prevent you from getting COVID at all, then it helps to reduce long COVID.”
And People with more serious initial illness appear more likely to have prolonged symptoms, but those with milder disease can certainly get it, too.
“You’re more likely to have long COVID with more severe disease, and we have ample evidence that vaccination reduces the severity of disease,” Dr. Horwitz said. “We also now have quite a lot of evidence that vaccination does reduce your risk of long COVID – probably because it reduces your risk of severe disease.”
There is little consensus about how much vaccines can lower the risk of long-term COVID symptoms, but several studies suggest that number lies anywhere from 15% to more than 80%.
That might seem like a big variation, but infectious disease experts argue that trying to interpret the gap isn’t as important as noticing what’s consistent across all these studies: “Vaccines do offer some protection, but it’s incomplete,” said Ziyad Al-Aly, MD, chief of research and development at the Veterans Affairs St. Louis Health Care System. Dr. Al-Aly, who has led several large studies on long COVID, said focusing on the fact that vaccines do offer some protection is a much better public health message than looking at the different levels of risk.
“Vaccines do a miraculous job for what they were designed to do,” said Dr. Al-Aly. “Vaccines were designed to reduce the risk of hospitalization ... and for that, vaccines are still holding up, even with all the changes in the virus.”
Still, Elena Azzolini, MD, PhD, head of the Humanitas Research Hospital’s vaccination center in Milan, thinks some studies may have underestimated the level of long COVID protection from vaccines because of limits in the study methods, such as not including enough women, who are more affected by long COVID. Her recent study, which looked at 2,560 health care professionals working in nine Italian centers from March 2020 to April 2022, focused on the risk for healthy women and men in their 20s to their 70s.
In the paper, Dr. Azzolini and associates reported that two or three doses of vaccine reduced the risk of hospitalization from COVID-19 from 42% among those who are unvaccinated to 16%-17%. In other words, they found unvaccinated people in the study were nearly three times as likely to have serious symptoms for longer than 4 weeks.
But Dr. Azzolini and Dr. Al-Aly still say that, even for the vaccinated, as long as COVID is around, masks are necessary. That’s because current vaccines don’t do enough to reduce transmission, said Dr. Al-Aly. “The only way that can really help [stop] transmission is covering our nose and mouth with a mask.”
How vaccinations affect people who already have long COVID
Some long COVID patients have said they got better after they get boosted, while some say they’re getting worse, said Dr. Horwitz, who is also a lead investigator at the National Institutes of Health’s flagship RECOVER program, a 4-year research project to study long COVID across the United States. (The NIH is still recruiting volunteers for these studies, which are also open to people who have never had COVID.)
One study published in the British Medical Journal analyzed survey data of more than 28,000 people infected with COVID in the United Kingdom and found a 13% reduction in long-term symptoms after a first dose of the vaccine, although it was unclear from the data if the improvement was sustained.
A second dose was associated with another 8% improvement over a 2-month period. “It’s reassuring that we see an average modest improvement in symptoms, not an average worsening in symptoms,” said Daniel Ayoubkhani, principal statistician at the U.K. Office for National Statistics and lead author of the study. Of course, the experience will differ among different people.
“It doesn’t appear that vaccination is the silver bullet that’s going to eradicate long COVID,” he said, but evidence from multiple studies suggests vaccines may help people with long-term symptoms.
Akiko Iwasaki, PhD, an immunobiologist at Yale University, New Haven, Conn., told a White House summit in July that one of the best ways to prevent long COVID is to develop the next generation of vaccines that also prevent milder cases by blocking transmission in the first place.
Back in New York, Dr. Kulick is now triple vaccinated. She’s due for a fourth dose soon but admits she’s “terrified every time” that she’s going to get sicker.
In her Facebook support group for long COVID, she reads that most people with prolonged symptoms handle it well. She has also noticed some of her symptoms eased after her first two doses of vaccine.
Since being diagnosed, Dr. Kulick learned she has a genetic condition, Ehlers-Danlos syndrome, which affects connective tissues that support skin, joints, organs, and blood vessels, and which her doctors say may have made her more prone to long COVID. She’s also being screened for autoimmune diseases, but for now, the only relief she has found has come from long COVID physical therapy, changes to her diet, and integrative medicine.
Dr. Kulick is still trying to figure out how she can get better while keeping her long hours at her veterinary job – and her health benefits. She is thankful her husband is a devoted caregiver to their son and a professional jazz musician with a schedule that allows for some flexibility.
“But it’s really hard when every week feels like I’ve run a marathon,” she said. “I can barely make it through.”
A version of this article first appeared on WebMD.com.
ACR makes changes to adult, pediatric vaccinations guidance
Patients with rheumatic and musculoskeletal diseases may need additional vaccines or different versions of vaccines they were not previously recommended to receive, according to updated guidelines from the American College of Rheumatology (ACR) on vaccinations for these patients. The new guidelines pertain to routine vaccinations for adults and children and are based on the most current evidence. They include recommendations on whether to hold certain medications before or after vaccination. They do not include recommendations regarding COVID-19 vaccines.
For guidance on COVID-19 vaccine timing and frequency, the ACR directs physicians to the CDC’s recommendations for people with mild or severe immunosuppression and the ACR’s previous clinical guidance summary on the topic, last revised in February 2022. The recommendations in the new guidance differ from ACR’s guidance on COVID-19 vaccines on whether and when to hold immunosuppressive medications when patients receive nonlive vaccines. The new guidelines now align more closely with those of EULAR, the Infectious Diseases Society of America, and the CDC’s recommendations for human papillomavirus (HPV), pneumococcal, and shingles vaccines.
Vaccinations in this population are particularly important because “a leading cause of morbidity and mortality in those with rheumatic diseases is infections, due to the detrimental impact immunosuppression has on the ability for the patient to properly clear the pathogen,” Alfred Kim, MD, PhD, professor of rheumatology at Washington University, St. Louis, told this news organization. While immunosuppressive medications are the most common reason patients with these conditions may have impaired immune function, “some of our patients with autoimmune disease also have a preexisting immunodeficiency that can inherently blunt immune responses to either infection or vaccination,” Dr. Kim explained.
“The authors of the guidelines have done a really nice job of making distinct recommendations based on the mechanism of action of various immunosuppressive medications,” Dr. Kim said. “This helps simplify the process of deciding the timing of vaccination for the health provider, especially for those on multiple immunosuppressives who represent an important proportion of our patients with rheumatic diseases.”
The main change to the guidelines for children, aside from those related to flu vaccination, is in regard to rotavirus vaccination for infants exposed to tumor necrosis factor (TNF) inhibitors or rituximab in utero. Infants prenatally exposed to rituximab should not receive the rotavirus vaccine until they are older than 6 months. Those exposed prenatally to TNF inhibitors should receive the rotavirus vaccine on time, according to the CDC schedule for all infants.
The new rotavirus recommendations follow data showing that immune responses to rotavirus are blunted in those with infliximab exposure, according to Dr. Kim.
“Thus, this poses a serious theoretical risk in newborns with mothers on [a TNF inhibitor] of ineffective clearance of rotavirus infections,” Dr. Kim said in an interview. “While rotavirus infections are quite common with typically self-limiting disease, sometimes requiring hydration to counteract diarrhea-induced dehydration, this can become severe in these newborns that have [a TNF inhibitor] in their system.”
For adults, the ACR issued the following expanded indications for four vaccines for patients currently taking immunosuppressive medication:
- Patients aged 18 and older should receive the recombinant zoster vaccine against shingles.
- For patients aged 27-44 who weren’t previously vaccinated against HPV, the HPV vaccine is “conditionally recommended.”
- Patients younger than 65 should receive the pneumococcal vaccine.
- Patients aged 19-64 are conditionally recommended to receive the high-dose or adjuvanted flu vaccine rather than the regular-dose flu vaccine.
The guidelines also conditionally recommend that all patients aged 65 and older who have rheumatic or musculoskeletal diseases receive the high-dose or adjuvanted flu vaccine, regardless of whether they are taking immunosuppressive medication. Another new conditional recommendation is to give multiple vaccinations to patients on the same day, rather than give individual vaccines on different days.
The guidelines make conditional recommendations regarding flu and nonlive attenuated vaccines for those taking methotrexate, rituximab, or glucocorticoids. Methotrexate should be held for 2 weeks after flu vaccination as long as disease activity allows it, but patients who are taking methotrexate should continue taking it for any other nonlive attenuated vaccinations.
“Non-rheumatology providers, such as general pediatricians and internists, are encouraged to give the influenza vaccination and then consult with the patient’s rheumatology provider about holding methotrexate to avoid a missed vaccination opportunity,” the guidelines state.
Patients taking rituximab should receive the flu vaccine on schedule and continue taking rituximab. However, for these patients, the guidelines recommend to “delay any subsequent rituximab dosing for at least two weeks after influenza vaccination if disease activity allows.”
“Because of the relatively short time period between the rollout of the influenza vaccine and its season, we can’t always wait to time the B-cell depletion dosage,” Dr. Kim said. “Also, it is not always easy to synchronize the patient’s B-cell depletion dosing schedule to the influenza vaccine rollout. Thus, we now just recommend getting the influenza vaccine regardless of the patient’s last B-cell depletion dosage despite its known strong attenuation of optimal immune responses.”
For other nonlive attenuated vaccines, providers should time vaccination for when the next rituximab dose is due and then hold the drug for at least 2 weeks thereafter, providing time for the B cells to mount a response before rituximab depletes B cells again.
Patients taking less than 20 mg of prednisone daily should still receive the flu vaccine and other nonlive attenuated vaccines. Those taking 20 mg or more of prednisone each day should still receive the flu vaccine, but other vaccines should be deferred until their dose of glucocorticoids has been tapered down to less than 20 mg daily.
Patients taking all other immunosuppressive medications should continue taking them for the flu vaccine and other nonlive attenuated vaccinations, but it is conditionally recommended that live attenuated vaccines be deferred. For any patient with a rheumatic and musculoskeletal disease, regardless of disease activity, it is conditionally recommended that all routine nonlive attenuated vaccines be administered.
For live attenuated virus vaccines, the ACR provides a chart on which immunosuppressive medications to hold and for how long. Glucocorticoids, methotrexate, azathioprine, leflunomide, mycophenolate mofetil, calcineurin inhibitors, and oral cyclophosphamide should all be held 4 weeks before and 4 weeks after administration of a live attenuated vaccine. For those taking JAK inhibitors, the medication should be halted 1 week before administration of a live vaccine and should continue to be withheld for 4 weeks after.
For most other biologics, the ACR recommends holding the medication for one dosing interval before the live vaccine and 4 weeks thereafter. The main exception is rituximab, which should be held for 6 months before a live vaccine and then for 4 more weeks thereafter.
For patients receiving intravenous immunoglobulin, the drug should be held for 8-11 months before they are administered a live attenuated vaccine, depending on the dosage, and then 4 weeks after vaccination, regardless of dosage.
To reassure people with rheumatic disease who may have anxiety or concerns about receiving immunizations, whether taking immunosuppressive medication or not, Dr. Kim said it’s important to provide lots of education to patients.
“Fear and emotion have replaced facts, and data as a leading factor in decision-making, as seen with COVID-19,” Dr. Kim said. “The reality is that a small minority of people will have any issues with most vaccines, which include disease flares, adverse events, or acquisition of an autoimmune disease. We are not saying there is zero risk, rather, that the risk is quite small. This is where shared decision-making between the health care provider and the patient must be done effectively to enable the patient to properly weigh risk versus benefit.”
Dr. Kim has relationships with GlaxoSmithKline, Aurinia Pharmaceuticals, Kypha, Pfizer, Alexion Pharmaceuticals, AstraZeneca, Exagen Diagnostics, and Foghorn Therapeutics.
A version of this article first appeared on Medscape.com.
Patients with rheumatic and musculoskeletal diseases may need additional vaccines or different versions of vaccines they were not previously recommended to receive, according to updated guidelines from the American College of Rheumatology (ACR) on vaccinations for these patients. The new guidelines pertain to routine vaccinations for adults and children and are based on the most current evidence. They include recommendations on whether to hold certain medications before or after vaccination. They do not include recommendations regarding COVID-19 vaccines.
For guidance on COVID-19 vaccine timing and frequency, the ACR directs physicians to the CDC’s recommendations for people with mild or severe immunosuppression and the ACR’s previous clinical guidance summary on the topic, last revised in February 2022. The recommendations in the new guidance differ from ACR’s guidance on COVID-19 vaccines on whether and when to hold immunosuppressive medications when patients receive nonlive vaccines. The new guidelines now align more closely with those of EULAR, the Infectious Diseases Society of America, and the CDC’s recommendations for human papillomavirus (HPV), pneumococcal, and shingles vaccines.
Vaccinations in this population are particularly important because “a leading cause of morbidity and mortality in those with rheumatic diseases is infections, due to the detrimental impact immunosuppression has on the ability for the patient to properly clear the pathogen,” Alfred Kim, MD, PhD, professor of rheumatology at Washington University, St. Louis, told this news organization. While immunosuppressive medications are the most common reason patients with these conditions may have impaired immune function, “some of our patients with autoimmune disease also have a preexisting immunodeficiency that can inherently blunt immune responses to either infection or vaccination,” Dr. Kim explained.
“The authors of the guidelines have done a really nice job of making distinct recommendations based on the mechanism of action of various immunosuppressive medications,” Dr. Kim said. “This helps simplify the process of deciding the timing of vaccination for the health provider, especially for those on multiple immunosuppressives who represent an important proportion of our patients with rheumatic diseases.”
The main change to the guidelines for children, aside from those related to flu vaccination, is in regard to rotavirus vaccination for infants exposed to tumor necrosis factor (TNF) inhibitors or rituximab in utero. Infants prenatally exposed to rituximab should not receive the rotavirus vaccine until they are older than 6 months. Those exposed prenatally to TNF inhibitors should receive the rotavirus vaccine on time, according to the CDC schedule for all infants.
The new rotavirus recommendations follow data showing that immune responses to rotavirus are blunted in those with infliximab exposure, according to Dr. Kim.
“Thus, this poses a serious theoretical risk in newborns with mothers on [a TNF inhibitor] of ineffective clearance of rotavirus infections,” Dr. Kim said in an interview. “While rotavirus infections are quite common with typically self-limiting disease, sometimes requiring hydration to counteract diarrhea-induced dehydration, this can become severe in these newborns that have [a TNF inhibitor] in their system.”
For adults, the ACR issued the following expanded indications for four vaccines for patients currently taking immunosuppressive medication:
- Patients aged 18 and older should receive the recombinant zoster vaccine against shingles.
- For patients aged 27-44 who weren’t previously vaccinated against HPV, the HPV vaccine is “conditionally recommended.”
- Patients younger than 65 should receive the pneumococcal vaccine.
- Patients aged 19-64 are conditionally recommended to receive the high-dose or adjuvanted flu vaccine rather than the regular-dose flu vaccine.
The guidelines also conditionally recommend that all patients aged 65 and older who have rheumatic or musculoskeletal diseases receive the high-dose or adjuvanted flu vaccine, regardless of whether they are taking immunosuppressive medication. Another new conditional recommendation is to give multiple vaccinations to patients on the same day, rather than give individual vaccines on different days.
The guidelines make conditional recommendations regarding flu and nonlive attenuated vaccines for those taking methotrexate, rituximab, or glucocorticoids. Methotrexate should be held for 2 weeks after flu vaccination as long as disease activity allows it, but patients who are taking methotrexate should continue taking it for any other nonlive attenuated vaccinations.
“Non-rheumatology providers, such as general pediatricians and internists, are encouraged to give the influenza vaccination and then consult with the patient’s rheumatology provider about holding methotrexate to avoid a missed vaccination opportunity,” the guidelines state.
Patients taking rituximab should receive the flu vaccine on schedule and continue taking rituximab. However, for these patients, the guidelines recommend to “delay any subsequent rituximab dosing for at least two weeks after influenza vaccination if disease activity allows.”
“Because of the relatively short time period between the rollout of the influenza vaccine and its season, we can’t always wait to time the B-cell depletion dosage,” Dr. Kim said. “Also, it is not always easy to synchronize the patient’s B-cell depletion dosing schedule to the influenza vaccine rollout. Thus, we now just recommend getting the influenza vaccine regardless of the patient’s last B-cell depletion dosage despite its known strong attenuation of optimal immune responses.”
For other nonlive attenuated vaccines, providers should time vaccination for when the next rituximab dose is due and then hold the drug for at least 2 weeks thereafter, providing time for the B cells to mount a response before rituximab depletes B cells again.
Patients taking less than 20 mg of prednisone daily should still receive the flu vaccine and other nonlive attenuated vaccines. Those taking 20 mg or more of prednisone each day should still receive the flu vaccine, but other vaccines should be deferred until their dose of glucocorticoids has been tapered down to less than 20 mg daily.
Patients taking all other immunosuppressive medications should continue taking them for the flu vaccine and other nonlive attenuated vaccinations, but it is conditionally recommended that live attenuated vaccines be deferred. For any patient with a rheumatic and musculoskeletal disease, regardless of disease activity, it is conditionally recommended that all routine nonlive attenuated vaccines be administered.
For live attenuated virus vaccines, the ACR provides a chart on which immunosuppressive medications to hold and for how long. Glucocorticoids, methotrexate, azathioprine, leflunomide, mycophenolate mofetil, calcineurin inhibitors, and oral cyclophosphamide should all be held 4 weeks before and 4 weeks after administration of a live attenuated vaccine. For those taking JAK inhibitors, the medication should be halted 1 week before administration of a live vaccine and should continue to be withheld for 4 weeks after.
For most other biologics, the ACR recommends holding the medication for one dosing interval before the live vaccine and 4 weeks thereafter. The main exception is rituximab, which should be held for 6 months before a live vaccine and then for 4 more weeks thereafter.
For patients receiving intravenous immunoglobulin, the drug should be held for 8-11 months before they are administered a live attenuated vaccine, depending on the dosage, and then 4 weeks after vaccination, regardless of dosage.
To reassure people with rheumatic disease who may have anxiety or concerns about receiving immunizations, whether taking immunosuppressive medication or not, Dr. Kim said it’s important to provide lots of education to patients.
“Fear and emotion have replaced facts, and data as a leading factor in decision-making, as seen with COVID-19,” Dr. Kim said. “The reality is that a small minority of people will have any issues with most vaccines, which include disease flares, adverse events, or acquisition of an autoimmune disease. We are not saying there is zero risk, rather, that the risk is quite small. This is where shared decision-making between the health care provider and the patient must be done effectively to enable the patient to properly weigh risk versus benefit.”
Dr. Kim has relationships with GlaxoSmithKline, Aurinia Pharmaceuticals, Kypha, Pfizer, Alexion Pharmaceuticals, AstraZeneca, Exagen Diagnostics, and Foghorn Therapeutics.
A version of this article first appeared on Medscape.com.
Patients with rheumatic and musculoskeletal diseases may need additional vaccines or different versions of vaccines they were not previously recommended to receive, according to updated guidelines from the American College of Rheumatology (ACR) on vaccinations for these patients. The new guidelines pertain to routine vaccinations for adults and children and are based on the most current evidence. They include recommendations on whether to hold certain medications before or after vaccination. They do not include recommendations regarding COVID-19 vaccines.
For guidance on COVID-19 vaccine timing and frequency, the ACR directs physicians to the CDC’s recommendations for people with mild or severe immunosuppression and the ACR’s previous clinical guidance summary on the topic, last revised in February 2022. The recommendations in the new guidance differ from ACR’s guidance on COVID-19 vaccines on whether and when to hold immunosuppressive medications when patients receive nonlive vaccines. The new guidelines now align more closely with those of EULAR, the Infectious Diseases Society of America, and the CDC’s recommendations for human papillomavirus (HPV), pneumococcal, and shingles vaccines.
Vaccinations in this population are particularly important because “a leading cause of morbidity and mortality in those with rheumatic diseases is infections, due to the detrimental impact immunosuppression has on the ability for the patient to properly clear the pathogen,” Alfred Kim, MD, PhD, professor of rheumatology at Washington University, St. Louis, told this news organization. While immunosuppressive medications are the most common reason patients with these conditions may have impaired immune function, “some of our patients with autoimmune disease also have a preexisting immunodeficiency that can inherently blunt immune responses to either infection or vaccination,” Dr. Kim explained.
“The authors of the guidelines have done a really nice job of making distinct recommendations based on the mechanism of action of various immunosuppressive medications,” Dr. Kim said. “This helps simplify the process of deciding the timing of vaccination for the health provider, especially for those on multiple immunosuppressives who represent an important proportion of our patients with rheumatic diseases.”
The main change to the guidelines for children, aside from those related to flu vaccination, is in regard to rotavirus vaccination for infants exposed to tumor necrosis factor (TNF) inhibitors or rituximab in utero. Infants prenatally exposed to rituximab should not receive the rotavirus vaccine until they are older than 6 months. Those exposed prenatally to TNF inhibitors should receive the rotavirus vaccine on time, according to the CDC schedule for all infants.
The new rotavirus recommendations follow data showing that immune responses to rotavirus are blunted in those with infliximab exposure, according to Dr. Kim.
“Thus, this poses a serious theoretical risk in newborns with mothers on [a TNF inhibitor] of ineffective clearance of rotavirus infections,” Dr. Kim said in an interview. “While rotavirus infections are quite common with typically self-limiting disease, sometimes requiring hydration to counteract diarrhea-induced dehydration, this can become severe in these newborns that have [a TNF inhibitor] in their system.”
For adults, the ACR issued the following expanded indications for four vaccines for patients currently taking immunosuppressive medication:
- Patients aged 18 and older should receive the recombinant zoster vaccine against shingles.
- For patients aged 27-44 who weren’t previously vaccinated against HPV, the HPV vaccine is “conditionally recommended.”
- Patients younger than 65 should receive the pneumococcal vaccine.
- Patients aged 19-64 are conditionally recommended to receive the high-dose or adjuvanted flu vaccine rather than the regular-dose flu vaccine.
The guidelines also conditionally recommend that all patients aged 65 and older who have rheumatic or musculoskeletal diseases receive the high-dose or adjuvanted flu vaccine, regardless of whether they are taking immunosuppressive medication. Another new conditional recommendation is to give multiple vaccinations to patients on the same day, rather than give individual vaccines on different days.
The guidelines make conditional recommendations regarding flu and nonlive attenuated vaccines for those taking methotrexate, rituximab, or glucocorticoids. Methotrexate should be held for 2 weeks after flu vaccination as long as disease activity allows it, but patients who are taking methotrexate should continue taking it for any other nonlive attenuated vaccinations.
“Non-rheumatology providers, such as general pediatricians and internists, are encouraged to give the influenza vaccination and then consult with the patient’s rheumatology provider about holding methotrexate to avoid a missed vaccination opportunity,” the guidelines state.
Patients taking rituximab should receive the flu vaccine on schedule and continue taking rituximab. However, for these patients, the guidelines recommend to “delay any subsequent rituximab dosing for at least two weeks after influenza vaccination if disease activity allows.”
“Because of the relatively short time period between the rollout of the influenza vaccine and its season, we can’t always wait to time the B-cell depletion dosage,” Dr. Kim said. “Also, it is not always easy to synchronize the patient’s B-cell depletion dosing schedule to the influenza vaccine rollout. Thus, we now just recommend getting the influenza vaccine regardless of the patient’s last B-cell depletion dosage despite its known strong attenuation of optimal immune responses.”
For other nonlive attenuated vaccines, providers should time vaccination for when the next rituximab dose is due and then hold the drug for at least 2 weeks thereafter, providing time for the B cells to mount a response before rituximab depletes B cells again.
Patients taking less than 20 mg of prednisone daily should still receive the flu vaccine and other nonlive attenuated vaccines. Those taking 20 mg or more of prednisone each day should still receive the flu vaccine, but other vaccines should be deferred until their dose of glucocorticoids has been tapered down to less than 20 mg daily.
Patients taking all other immunosuppressive medications should continue taking them for the flu vaccine and other nonlive attenuated vaccinations, but it is conditionally recommended that live attenuated vaccines be deferred. For any patient with a rheumatic and musculoskeletal disease, regardless of disease activity, it is conditionally recommended that all routine nonlive attenuated vaccines be administered.
For live attenuated virus vaccines, the ACR provides a chart on which immunosuppressive medications to hold and for how long. Glucocorticoids, methotrexate, azathioprine, leflunomide, mycophenolate mofetil, calcineurin inhibitors, and oral cyclophosphamide should all be held 4 weeks before and 4 weeks after administration of a live attenuated vaccine. For those taking JAK inhibitors, the medication should be halted 1 week before administration of a live vaccine and should continue to be withheld for 4 weeks after.
For most other biologics, the ACR recommends holding the medication for one dosing interval before the live vaccine and 4 weeks thereafter. The main exception is rituximab, which should be held for 6 months before a live vaccine and then for 4 more weeks thereafter.
For patients receiving intravenous immunoglobulin, the drug should be held for 8-11 months before they are administered a live attenuated vaccine, depending on the dosage, and then 4 weeks after vaccination, regardless of dosage.
To reassure people with rheumatic disease who may have anxiety or concerns about receiving immunizations, whether taking immunosuppressive medication or not, Dr. Kim said it’s important to provide lots of education to patients.
“Fear and emotion have replaced facts, and data as a leading factor in decision-making, as seen with COVID-19,” Dr. Kim said. “The reality is that a small minority of people will have any issues with most vaccines, which include disease flares, adverse events, or acquisition of an autoimmune disease. We are not saying there is zero risk, rather, that the risk is quite small. This is where shared decision-making between the health care provider and the patient must be done effectively to enable the patient to properly weigh risk versus benefit.”
Dr. Kim has relationships with GlaxoSmithKline, Aurinia Pharmaceuticals, Kypha, Pfizer, Alexion Pharmaceuticals, AstraZeneca, Exagen Diagnostics, and Foghorn Therapeutics.
A version of this article first appeared on Medscape.com.
‘Self-boosting’ vaccines could be immunizations of the future
Most vaccines don’t come as one-shot deals. A series of boosters is needed to step up immunity to COVID-19, tetanus, and other infectious threats over time.
But what if you could receive just one shot that boosts itself whenever you need a bump in protection?
Researchers at the Massachusetts Institute of Technology (MIT) have developed microparticles that could be used to create self-boosting vaccines that deliver their contents at carefully set time points. In a new study published in the journal Science Advances, the scientists describe how they tune the particles to release the goods at the right time and offer insights on how they can keep the particles stable until then.
How self-boosting vaccines could work
The team developed tiny particles that look like coffee cups – except instead of your favorite brew, they’re filled with vaccine.
“You can put the lid on, and then inject it into the body, and once the lid breaks, whatever is in there is released,” says study author Ana Jaklenec, PhD, a research scientist at MIT’s Koch Institute for Integrative Cancer Research.
To make the tiny cups, the researchers use various polymers already used in medical applications, such as dissolvable stitches. Then they fill the cups with vaccine material that is dried and combined with sugars and other stabilizers.
The particles can be made in various shapes and fine-tuned using polymers with different properties. Some polymers last longer in the body than others, so their choice helps determine how long everything will stay stable under the skin after the injection and when the particles will release their cargo. It could be days or months after the injection.
One challenge is that as the particles open, the environment around them becomes more acidic. The team is working on ways to curb that acidity to make the vaccine material more stable.
“We have ongoing research that has produced some really, really exciting results about their stability and [shows] that you’re able to maintain really sensitive vaccines, stable for a good period of time,” says study author Morteza Sarmadi, PhD, a research specialist at the Koch Institute.
The potential public health impact
This research, funded by the Bill & Melinda Gates Foundation, started with the developing world in mind.
“The intent was actually helping people in the developing world, because a lot of times, people don’t come back for a second injection,” says study author Robert Langer, ScD, the David H. Koch Institute professor at MIT.
But a one-shot plan could benefit the developed world, too. One reason is that self-boosting vaccines could help those who get one achieve higher antibody responses than they would with just one dose. That could mean more protection for the person and the population, because as people develop stronger immunity, germs may have less of a chance to evolve and spread.
Take the COVID-19 pandemic, for example. Only 67% of Americans are fully vaccinated, and most people eligible for first and second boosters haven’t gotten them. New variants, such as the recent Omicron ones, continue to emerge and infect.
“I think those variants would have had a lot less chance to come about if everybody that had gotten vaccinated the first time got repeat injections, which they didn’t,” says Dr. Langer.
Self-boosting vaccines could also benefit infants, children who fear shots, and older adults who have a hard time getting health care.
Also, because the vaccine material is encapsulated and its release can be staggered, this technology might help people receive multiple vaccines at the same time that must now be given separately.
What comes next
The team is testing self-boosting polio and hepatitis vaccines in non-human primates. A small trial in healthy humans might follow within the next few years.
“We think that there’s really high potential for this technology, and we hope it can be developed and get to the human phase very soon,” says Dr. Jaklenec.
In smaller animal models, they are exploring the potential of self-boosting mRNA vaccines. They’re also working with scientists who are studying HIV vaccines.
“There has been some recent progress where very complex regimens seem to be working, but they’re not practical,” says Dr. Jaklenec. “And so, this is where this particular technology could be useful, because you have to prime and boost with different things, and this allows you to do that.”
This system could also extend beyond vaccines and be used to deliver cancer therapies, hormones, and biologics in a shot.
Through new work with researchers at Georgia Tech University, the team will study the potential of giving self-boosting vaccines through 3D-printed microneedles. These vaccines, which would stick on your skin like a bandage, could be self-administered and deployed globally in response to local outbreaks.
A version of this article first appeared on WebMD.com.
Most vaccines don’t come as one-shot deals. A series of boosters is needed to step up immunity to COVID-19, tetanus, and other infectious threats over time.
But what if you could receive just one shot that boosts itself whenever you need a bump in protection?
Researchers at the Massachusetts Institute of Technology (MIT) have developed microparticles that could be used to create self-boosting vaccines that deliver their contents at carefully set time points. In a new study published in the journal Science Advances, the scientists describe how they tune the particles to release the goods at the right time and offer insights on how they can keep the particles stable until then.
How self-boosting vaccines could work
The team developed tiny particles that look like coffee cups – except instead of your favorite brew, they’re filled with vaccine.
“You can put the lid on, and then inject it into the body, and once the lid breaks, whatever is in there is released,” says study author Ana Jaklenec, PhD, a research scientist at MIT’s Koch Institute for Integrative Cancer Research.
To make the tiny cups, the researchers use various polymers already used in medical applications, such as dissolvable stitches. Then they fill the cups with vaccine material that is dried and combined with sugars and other stabilizers.
The particles can be made in various shapes and fine-tuned using polymers with different properties. Some polymers last longer in the body than others, so their choice helps determine how long everything will stay stable under the skin after the injection and when the particles will release their cargo. It could be days or months after the injection.
One challenge is that as the particles open, the environment around them becomes more acidic. The team is working on ways to curb that acidity to make the vaccine material more stable.
“We have ongoing research that has produced some really, really exciting results about their stability and [shows] that you’re able to maintain really sensitive vaccines, stable for a good period of time,” says study author Morteza Sarmadi, PhD, a research specialist at the Koch Institute.
The potential public health impact
This research, funded by the Bill & Melinda Gates Foundation, started with the developing world in mind.
“The intent was actually helping people in the developing world, because a lot of times, people don’t come back for a second injection,” says study author Robert Langer, ScD, the David H. Koch Institute professor at MIT.
But a one-shot plan could benefit the developed world, too. One reason is that self-boosting vaccines could help those who get one achieve higher antibody responses than they would with just one dose. That could mean more protection for the person and the population, because as people develop stronger immunity, germs may have less of a chance to evolve and spread.
Take the COVID-19 pandemic, for example. Only 67% of Americans are fully vaccinated, and most people eligible for first and second boosters haven’t gotten them. New variants, such as the recent Omicron ones, continue to emerge and infect.
“I think those variants would have had a lot less chance to come about if everybody that had gotten vaccinated the first time got repeat injections, which they didn’t,” says Dr. Langer.
Self-boosting vaccines could also benefit infants, children who fear shots, and older adults who have a hard time getting health care.
Also, because the vaccine material is encapsulated and its release can be staggered, this technology might help people receive multiple vaccines at the same time that must now be given separately.
What comes next
The team is testing self-boosting polio and hepatitis vaccines in non-human primates. A small trial in healthy humans might follow within the next few years.
“We think that there’s really high potential for this technology, and we hope it can be developed and get to the human phase very soon,” says Dr. Jaklenec.
In smaller animal models, they are exploring the potential of self-boosting mRNA vaccines. They’re also working with scientists who are studying HIV vaccines.
“There has been some recent progress where very complex regimens seem to be working, but they’re not practical,” says Dr. Jaklenec. “And so, this is where this particular technology could be useful, because you have to prime and boost with different things, and this allows you to do that.”
This system could also extend beyond vaccines and be used to deliver cancer therapies, hormones, and biologics in a shot.
Through new work with researchers at Georgia Tech University, the team will study the potential of giving self-boosting vaccines through 3D-printed microneedles. These vaccines, which would stick on your skin like a bandage, could be self-administered and deployed globally in response to local outbreaks.
A version of this article first appeared on WebMD.com.
Most vaccines don’t come as one-shot deals. A series of boosters is needed to step up immunity to COVID-19, tetanus, and other infectious threats over time.
But what if you could receive just one shot that boosts itself whenever you need a bump in protection?
Researchers at the Massachusetts Institute of Technology (MIT) have developed microparticles that could be used to create self-boosting vaccines that deliver their contents at carefully set time points. In a new study published in the journal Science Advances, the scientists describe how they tune the particles to release the goods at the right time and offer insights on how they can keep the particles stable until then.
How self-boosting vaccines could work
The team developed tiny particles that look like coffee cups – except instead of your favorite brew, they’re filled with vaccine.
“You can put the lid on, and then inject it into the body, and once the lid breaks, whatever is in there is released,” says study author Ana Jaklenec, PhD, a research scientist at MIT’s Koch Institute for Integrative Cancer Research.
To make the tiny cups, the researchers use various polymers already used in medical applications, such as dissolvable stitches. Then they fill the cups with vaccine material that is dried and combined with sugars and other stabilizers.
The particles can be made in various shapes and fine-tuned using polymers with different properties. Some polymers last longer in the body than others, so their choice helps determine how long everything will stay stable under the skin after the injection and when the particles will release their cargo. It could be days or months after the injection.
One challenge is that as the particles open, the environment around them becomes more acidic. The team is working on ways to curb that acidity to make the vaccine material more stable.
“We have ongoing research that has produced some really, really exciting results about their stability and [shows] that you’re able to maintain really sensitive vaccines, stable for a good period of time,” says study author Morteza Sarmadi, PhD, a research specialist at the Koch Institute.
The potential public health impact
This research, funded by the Bill & Melinda Gates Foundation, started with the developing world in mind.
“The intent was actually helping people in the developing world, because a lot of times, people don’t come back for a second injection,” says study author Robert Langer, ScD, the David H. Koch Institute professor at MIT.
But a one-shot plan could benefit the developed world, too. One reason is that self-boosting vaccines could help those who get one achieve higher antibody responses than they would with just one dose. That could mean more protection for the person and the population, because as people develop stronger immunity, germs may have less of a chance to evolve and spread.
Take the COVID-19 pandemic, for example. Only 67% of Americans are fully vaccinated, and most people eligible for first and second boosters haven’t gotten them. New variants, such as the recent Omicron ones, continue to emerge and infect.
“I think those variants would have had a lot less chance to come about if everybody that had gotten vaccinated the first time got repeat injections, which they didn’t,” says Dr. Langer.
Self-boosting vaccines could also benefit infants, children who fear shots, and older adults who have a hard time getting health care.
Also, because the vaccine material is encapsulated and its release can be staggered, this technology might help people receive multiple vaccines at the same time that must now be given separately.
What comes next
The team is testing self-boosting polio and hepatitis vaccines in non-human primates. A small trial in healthy humans might follow within the next few years.
“We think that there’s really high potential for this technology, and we hope it can be developed and get to the human phase very soon,” says Dr. Jaklenec.
In smaller animal models, they are exploring the potential of self-boosting mRNA vaccines. They’re also working with scientists who are studying HIV vaccines.
“There has been some recent progress where very complex regimens seem to be working, but they’re not practical,” says Dr. Jaklenec. “And so, this is where this particular technology could be useful, because you have to prime and boost with different things, and this allows you to do that.”
This system could also extend beyond vaccines and be used to deliver cancer therapies, hormones, and biologics in a shot.
Through new work with researchers at Georgia Tech University, the team will study the potential of giving self-boosting vaccines through 3D-printed microneedles. These vaccines, which would stick on your skin like a bandage, could be self-administered and deployed globally in response to local outbreaks.
A version of this article first appeared on WebMD.com.
FROM SCIENCE ADVANCES
White House declares monkeypox a public health emergency
There have been more than 6,600 reported cases of the disease in the United States, up from less than 5,000 cases reported last week.
“This public health emergency will allow us to explore additional strategies to get vaccines and treatments more quickly out in the affected communities. And it will allow us to get more data from jurisdictions so we can effectively track and attack this outbreak,” Robert Fenton, who was named as the national monkeypox response coordinator this week, said at a news briefing Aug. 4.
Those who catch the virus usually have fever-like symptoms, followed by red lesions on the body that can raise and develop pus. Those at highest risk of monkeypox are gay and bisexual men, as well as men who have sex with other men. There are between 1.6 million and 1.7 million Americans in this high-risk group, Health and Human Services Secretary Xavier Becerra said at the briefing.
The Jynneos vaccine is being distributed to protect against monkeypox and can prevent severe symptoms. It’s mostly going to those with the greatest risk of catching the virus.
Last week, the Biden administration made over 1.1 million doses of the Jynneos vaccine available – of which over 600,000 doses have already been distributed across the country – and have secured over 6.9 million Jynneos doses altogether.
Around 786,000 vaccines have already been allocated, and the first doses were shipped this week. States will be able to order more doses beginning Aug. 15. If a state has used 90% or more of its vaccine supply, it will be eligible to order more doses before Aug. 15, according to Dawn O’Connell, JD, assistant secretary for preparedness and response at the U.S. Department of Health and Human Services.
An additional 150,000 doses will be added to the national stockpile in September, with more doses to come later this year, Ms. O’Connell says.
The administration is also stressing the importance of monkeypox testing and says it can now distribute 80,000 monkeypox tests per week.
An antiviral drug – known as TPOXX – is also available to treat severe cases of monkeypox. Around 1,700,000 doses are available in the Strategic National Stockpile, public health officials say.
“We are prepared to take our response to the next level, and we urge every American to take this seriously and to take responsibility to help us tackle this virus,” Secretary Becerra told reporters.
The White House says it will continue reaching out to doctors, public health partners, LGBTQ advocates, and other impacted communities.
“The public health emergency further raises awareness about monkeypox, which will encourage clinicians to test for it,” Rochelle Walensky, MD, director of the Centers for Disease Control and Prevention, said at the briefing.
This week, President Joe Biden appointed a new White House monkeypox response team. Besides Mr. Fenton as the response coordinator, Demetre Daskalakis, MD, will serve as the White House national monkeypox response deputy coordinator. He is the director of the CDC’s Division of HIV Prevention.
“This virus is moving fast. This is a unique outbreak that is spreading faster than previous outbreaks,” Mr. Fenton told reporters Aug. 4. “That’s why the president asked me to explore everything we can do to combat monkeypox and protect communities at risk.”
This article was updated 8/4/22.
There have been more than 6,600 reported cases of the disease in the United States, up from less than 5,000 cases reported last week.
“This public health emergency will allow us to explore additional strategies to get vaccines and treatments more quickly out in the affected communities. And it will allow us to get more data from jurisdictions so we can effectively track and attack this outbreak,” Robert Fenton, who was named as the national monkeypox response coordinator this week, said at a news briefing Aug. 4.
Those who catch the virus usually have fever-like symptoms, followed by red lesions on the body that can raise and develop pus. Those at highest risk of monkeypox are gay and bisexual men, as well as men who have sex with other men. There are between 1.6 million and 1.7 million Americans in this high-risk group, Health and Human Services Secretary Xavier Becerra said at the briefing.
The Jynneos vaccine is being distributed to protect against monkeypox and can prevent severe symptoms. It’s mostly going to those with the greatest risk of catching the virus.
Last week, the Biden administration made over 1.1 million doses of the Jynneos vaccine available – of which over 600,000 doses have already been distributed across the country – and have secured over 6.9 million Jynneos doses altogether.
Around 786,000 vaccines have already been allocated, and the first doses were shipped this week. States will be able to order more doses beginning Aug. 15. If a state has used 90% or more of its vaccine supply, it will be eligible to order more doses before Aug. 15, according to Dawn O’Connell, JD, assistant secretary for preparedness and response at the U.S. Department of Health and Human Services.
An additional 150,000 doses will be added to the national stockpile in September, with more doses to come later this year, Ms. O’Connell says.
The administration is also stressing the importance of monkeypox testing and says it can now distribute 80,000 monkeypox tests per week.
An antiviral drug – known as TPOXX – is also available to treat severe cases of monkeypox. Around 1,700,000 doses are available in the Strategic National Stockpile, public health officials say.
“We are prepared to take our response to the next level, and we urge every American to take this seriously and to take responsibility to help us tackle this virus,” Secretary Becerra told reporters.
The White House says it will continue reaching out to doctors, public health partners, LGBTQ advocates, and other impacted communities.
“The public health emergency further raises awareness about monkeypox, which will encourage clinicians to test for it,” Rochelle Walensky, MD, director of the Centers for Disease Control and Prevention, said at the briefing.
This week, President Joe Biden appointed a new White House monkeypox response team. Besides Mr. Fenton as the response coordinator, Demetre Daskalakis, MD, will serve as the White House national monkeypox response deputy coordinator. He is the director of the CDC’s Division of HIV Prevention.
“This virus is moving fast. This is a unique outbreak that is spreading faster than previous outbreaks,” Mr. Fenton told reporters Aug. 4. “That’s why the president asked me to explore everything we can do to combat monkeypox and protect communities at risk.”
This article was updated 8/4/22.
There have been more than 6,600 reported cases of the disease in the United States, up from less than 5,000 cases reported last week.
“This public health emergency will allow us to explore additional strategies to get vaccines and treatments more quickly out in the affected communities. And it will allow us to get more data from jurisdictions so we can effectively track and attack this outbreak,” Robert Fenton, who was named as the national monkeypox response coordinator this week, said at a news briefing Aug. 4.
Those who catch the virus usually have fever-like symptoms, followed by red lesions on the body that can raise and develop pus. Those at highest risk of monkeypox are gay and bisexual men, as well as men who have sex with other men. There are between 1.6 million and 1.7 million Americans in this high-risk group, Health and Human Services Secretary Xavier Becerra said at the briefing.
The Jynneos vaccine is being distributed to protect against monkeypox and can prevent severe symptoms. It’s mostly going to those with the greatest risk of catching the virus.
Last week, the Biden administration made over 1.1 million doses of the Jynneos vaccine available – of which over 600,000 doses have already been distributed across the country – and have secured over 6.9 million Jynneos doses altogether.
Around 786,000 vaccines have already been allocated, and the first doses were shipped this week. States will be able to order more doses beginning Aug. 15. If a state has used 90% or more of its vaccine supply, it will be eligible to order more doses before Aug. 15, according to Dawn O’Connell, JD, assistant secretary for preparedness and response at the U.S. Department of Health and Human Services.
An additional 150,000 doses will be added to the national stockpile in September, with more doses to come later this year, Ms. O’Connell says.
The administration is also stressing the importance of monkeypox testing and says it can now distribute 80,000 monkeypox tests per week.
An antiviral drug – known as TPOXX – is also available to treat severe cases of monkeypox. Around 1,700,000 doses are available in the Strategic National Stockpile, public health officials say.
“We are prepared to take our response to the next level, and we urge every American to take this seriously and to take responsibility to help us tackle this virus,” Secretary Becerra told reporters.
The White House says it will continue reaching out to doctors, public health partners, LGBTQ advocates, and other impacted communities.
“The public health emergency further raises awareness about monkeypox, which will encourage clinicians to test for it,” Rochelle Walensky, MD, director of the Centers for Disease Control and Prevention, said at the briefing.
This week, President Joe Biden appointed a new White House monkeypox response team. Besides Mr. Fenton as the response coordinator, Demetre Daskalakis, MD, will serve as the White House national monkeypox response deputy coordinator. He is the director of the CDC’s Division of HIV Prevention.
“This virus is moving fast. This is a unique outbreak that is spreading faster than previous outbreaks,” Mr. Fenton told reporters Aug. 4. “That’s why the president asked me to explore everything we can do to combat monkeypox and protect communities at risk.”
This article was updated 8/4/22.




