User login
Concurrent Atopic Dermatitis and Psoriasis Vulgaris: Implications for Targeted Biologic Therapy
Psoriasis vulgaris is a chronic inflammatory skin condition associated with notable elevation in helper T cell (TH) production of TH1/TH17-mediated inflammatory cytokines, including IL-17A.1 Upon binding of IL-17A to IL-17 receptors in the skin, an inflammatory cascade is triggered, resulting in the classic clinical appearance of psoriasis. Moderate to severe psoriasis often is managed by suppressing TH1/TH17-mediated inflammation using targeted immune therapy such as secukinumab, an IL-17A inhibitor.2 Atopic dermatitis (AD), another chronic inflammatory dermatosis, is associated with substantial elevation in TH2-mediated inflammatory cytokines, such as IL-4.3 Dupilumab, which interacts with IL-4R, disrupts the IL-4 and IL-13 signaling pathways and demonstrates considerable efficacy in the treatment of moderate to severe AD.4
A case series has shown that suppression of the TH1/TH17-mediated inflammation of psoriasis may paradoxically result in the development of TH2-mediated AD.5 Similarly, a recent case report described a patient who developed psoriasis following treatment of AD with dupilumab.6 Herein, we describe a patient with a history of psoriasis that was well controlled with secukinumab who developed severe refractory erythrodermic AD that resolved with dupilumab treatment. Following clearance of AD with dupilumab, he exhibited psoriasis recurrence.
Case Report
A 39-year-old man with a lifelong history of psoriasis was admitted to the hospital for management of severe erythroderma. Four years prior, secukinumab was initiated for treatment of psoriasis, resulting in excellent clinical response. He discontinued secukinumab after 2 years of treatment because of insurance coverage issues and managed his condition with only topical corticosteroids. He restarted secukinumab 10 months before admission because of a psoriasis flare. Shortly after resuming secukinumab, he developed a severe exfoliative erythroderma that was not responsive to corticosteroids, etanercept, methotrexate, or ustekinumab.
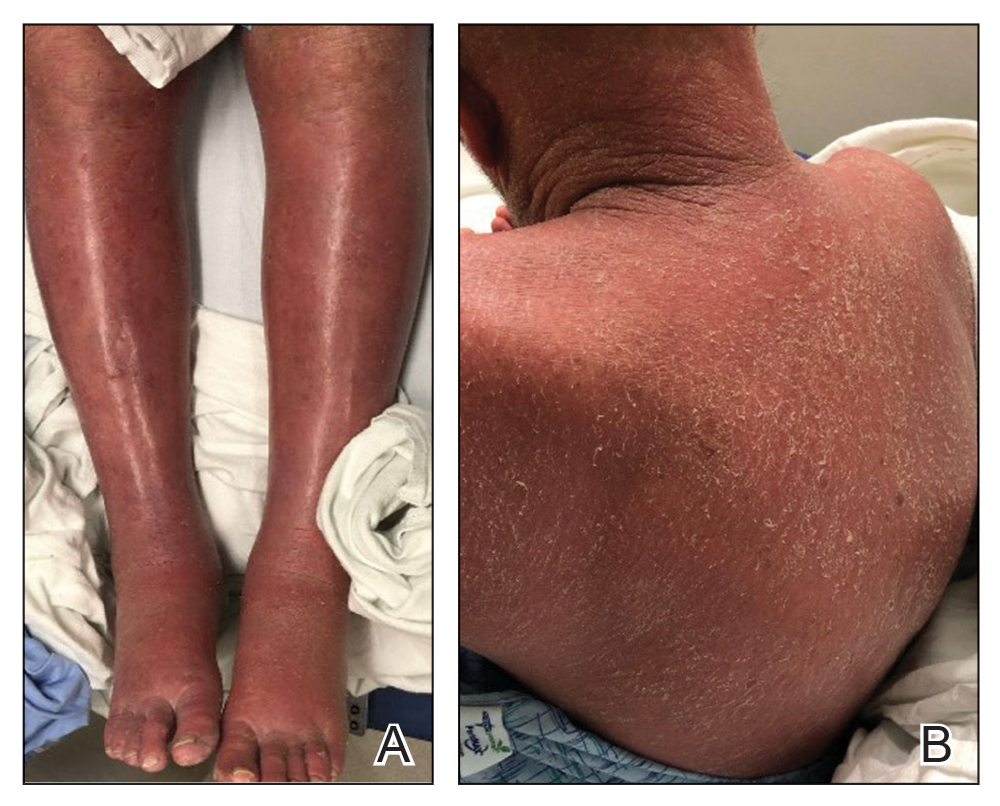
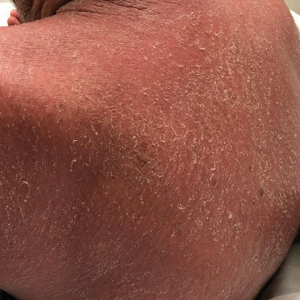
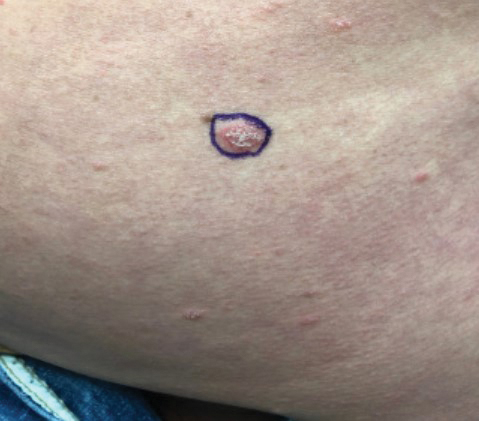
On initial presentation, physical examination revealed diffuse erythema and scaling with associated edema of the face, trunk, and extremities (Figure 1). A biopsy from the patient’s right arm demonstrated a superficial perivascular inflammatory infiltrate composed of lymphocytes, histiocytes, and scattered eosinophils consistent with spongiotic dermatitis (Figure 2). Cyclosporine 225 mg twice daily and topical corticosteroids were started.
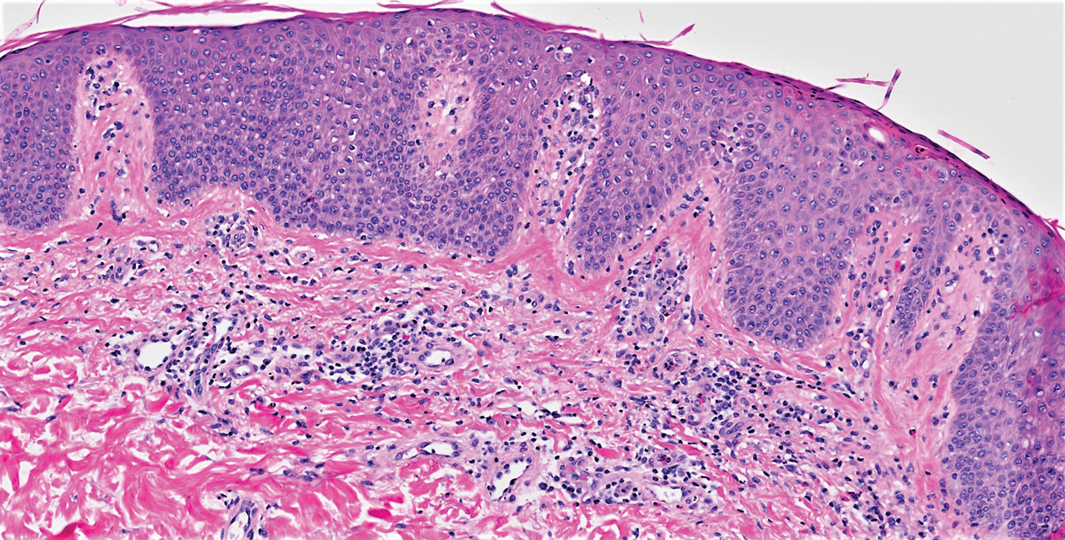
Over the next several months, the patient had several admissions secondary to recurrent skin abscesses in the setting of refractory erythroderma. He underwent trials of infliximab, corticosteroids, intravenous immunoglobulin, guselkumab, and acitretin with minimal improvement. He underwent an extensive laboratory and radiologic workup, which was notable for cyclical peripheral eosinophilia and elevated IgE levels correlating with the erythroderma flares. A second biopsy was obtained and continued to demonstrate changes consistent with AD.
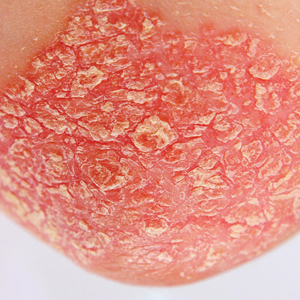
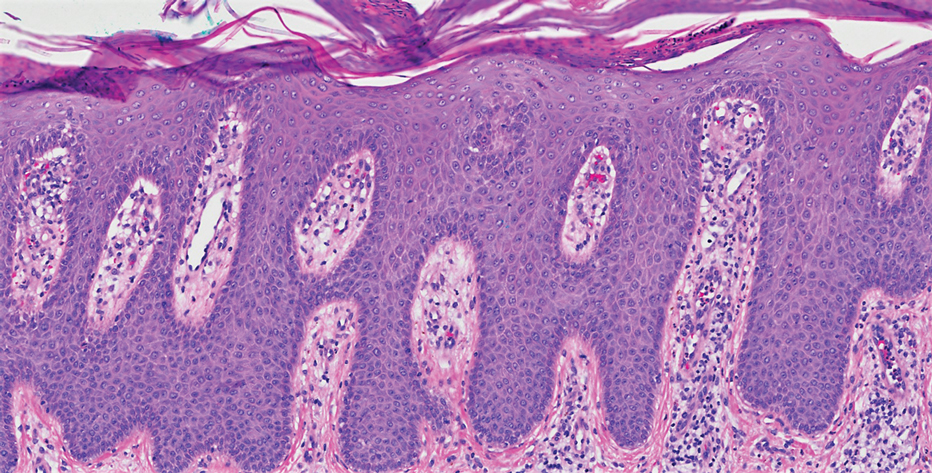
Four months after the initial hospitalization, all psoriasis medications were stopped, and the patient was started on dupilumab 300 mg/2 mL every 2 weeks and an 8-week oral prednisone taper. This combination led to notable clinical improvement and resolution of peripheral eosinophilia. Several months after disease remission, he began to develop worsening erythema and pruritus on the trunk and extremities, followed by the development of new psoriatic lesions (Figure 3) with a biopsy consistent with psoriasis (Figure 4). The patient was continued on dupilumab, but cyclosporine was added. The patient self-discontinued dupilumab owing to injection-site discomfort and has been slowly weaning off oral cyclosporine with 1 to 2 remaining eczematous plaques and 1 to 2 psoriatic plaques managed by topical corticosteroids.
Comment
We present a patient with psoriasis that was well controlled on secukinumab who developed severe AD following treatment with secukinumab. The AD resolved following treatment with dupilumab and a tapering dose of prednisone. However, after several months of treatment with dupilumab alone, he began to develop psoriatic lesions again. This case supports findings in a case series describing the development of AD in patients with psoriasis treated with IL-17 inhibitors5 and a recent case report describing a patient with AD who developed psoriasis following treatment with an IL-4/IL-13 inhibitor.6
Recognized adverse effects demonstrate biologic medications’ contributions to both normal as well as aberrant immunologic responses. For example, IL-17 plays an essential role in innate and adaptive immune responses against infections at mucosal and cutaneous interfaces, as demonstrated by chronic mucocutaneous candidiasis in patients with genetic defects in IL-17–related pathways.7 Similarly, in patients taking IL-17 antagonists, an increase in the incidence of Candida infections has been observed.8 In patients with concurrent psoriasis and inflammatory bowel disease (IBD), treatment with IL-17 inhibitors is contraindicated due to the risk of exacerbating the IBD. This observation is somewhat paradoxical, as increased IL-17 release by TH17 cells is implicated in the pathogenesis of IBD.9 Interestingly, it is now thought that IL-17 may play a protective role in T-cell–driven intestinal inflammation through induction of protective intestinal epithelial gene expression and increased mucosal defense against gut microbes, explaining the worsening of IBD in patients on IL-17 inhibitors.10 These adverse effects illustrate the complicated and varied roles biologic medications play in immunologic response.
Given that TH1 and TH2 exert opposing immune mechanisms, it is uncommon for psoriasis and AD to coexist in a single patient. However, patients who exhibit concurrent findings may represent a unique population in which psoriasis and AD coexist, perhaps because of an underlying genetic predisposition. Moreover, targeted treatment of pathways unique to these disease processes may result in paradoxical flaring of the nontargeted pathway. It also is possible that inhibition of a specific T-cell pathway in a subset of patients will result in an immunologic imbalance, favoring increased activity of the opposing pathway in the absence of coexisting disease. In the case presented here, the findings may be explained by secukinumab’s inhibition of TH1/TH17-mediated inflammation, which resulted in a shift to a TH2-mediated inflammatory response manifesting as AD, as well as dupilumab’s inhibition of TH2-mediated inflammation, which caused a shift back to TH1-mediated inflammatory pathways. Additionally, for patients with changing morphologies exacerbated by biologic medications, alternative diagnoses, such as cutaneous T-cell lymphoma, may be considered.
Conclusion
We report an unusual case of secukinumab-induced AD in a patient with psoriasis that resolved following several months of treatment with dupilumab and a tapering dose of prednisone. Subsequently, this same patient developed re-emergence of psoriatic lesions with continued use of dupilumab, which was eventually discontinued by the patient despite appropriate disease control. In addition to illustrating the underlying pathophysiologic mechanisms of 2 common inflammatory dermatologic conditions, this case highlights how pharmacologic interventions targeted at specific immunologic pathways may have unintended consequences. Further investigation into the effects of targeted biologics on the TH1/TH2 immune axis is warranted to better understand the mechanism and possible implications of the phenotypic switching presented in this case.
- Diani M, Altomare G, Reali E. T helper cell subsets in clinical manifestations of psoriasis. J Immunol Res. 2016;2016:7692024.
- Langley RG, Elewski BE, Lebwohl M, et al. Secukinumab in plaque psoriasis—results of two phase 3 trials. N Engl J Med. 2014;371:326-338.
- van der Heijden FL, Wierenga EA, Bos JD, et al. High frequency of IL-4-producing CD4+ allergen-specific T lymphocytes in atopic dermatitis lesional skin. J Invest Dermatol. 1991;97:389-394.
- Beck LA, Thaçi D, Hamilton JD, et al. Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. N Engl J Med. 2014;371:130-139.
- Lai FYX, Higgins E, Smith CH, et al. Morphologic switch from psoriasiform to eczematous dermatitis after anti-IL-17 therapy: a case series. JAMA Dermatol. 2019;155:1082-1084.
- Varma A, Levitt J. Dupilumab-induced phenotype switching from atopic dermatitis to psoriasis. JAAD Case Rep. 2020;6:217-218.
- Ling Y, Puel A. IL-17 and infections. Actas Dermosifiliogr. 2014;105(suppl 1):34-40.
- Saunte DM, Mrowietz U, Puig L, et al. Candida infections in patients with psoriasis and psoriatic arthritis treated with interleukin-17 inhibitors and their practical management. Br J Dermatol. 2017;177:47-62.
- Hölttä V, Klemetti P, Sipponen T, et al. IL-23/IL-17 immunity as a hallmark of Crohn’s disease. Inflamm Bowel Dis. 2008;14:1175-1184.
- Smith MK, Pai J, Panaccione R, et al. Crohn’s-like disease in a patient exposed to anti-interleukin-17 blockade (ixekizumab) for the treatment of chronic plaque psoriasis: a case report. BMC Gastroenterol. 2019;19:162.
Psoriasis vulgaris is a chronic inflammatory skin condition associated with notable elevation in helper T cell (TH) production of TH1/TH17-mediated inflammatory cytokines, including IL-17A.1 Upon binding of IL-17A to IL-17 receptors in the skin, an inflammatory cascade is triggered, resulting in the classic clinical appearance of psoriasis. Moderate to severe psoriasis often is managed by suppressing TH1/TH17-mediated inflammation using targeted immune therapy such as secukinumab, an IL-17A inhibitor.2 Atopic dermatitis (AD), another chronic inflammatory dermatosis, is associated with substantial elevation in TH2-mediated inflammatory cytokines, such as IL-4.3 Dupilumab, which interacts with IL-4R, disrupts the IL-4 and IL-13 signaling pathways and demonstrates considerable efficacy in the treatment of moderate to severe AD.4
A case series has shown that suppression of the TH1/TH17-mediated inflammation of psoriasis may paradoxically result in the development of TH2-mediated AD.5 Similarly, a recent case report described a patient who developed psoriasis following treatment of AD with dupilumab.6 Herein, we describe a patient with a history of psoriasis that was well controlled with secukinumab who developed severe refractory erythrodermic AD that resolved with dupilumab treatment. Following clearance of AD with dupilumab, he exhibited psoriasis recurrence.
Case Report
A 39-year-old man with a lifelong history of psoriasis was admitted to the hospital for management of severe erythroderma. Four years prior, secukinumab was initiated for treatment of psoriasis, resulting in excellent clinical response. He discontinued secukinumab after 2 years of treatment because of insurance coverage issues and managed his condition with only topical corticosteroids. He restarted secukinumab 10 months before admission because of a psoriasis flare. Shortly after resuming secukinumab, he developed a severe exfoliative erythroderma that was not responsive to corticosteroids, etanercept, methotrexate, or ustekinumab.

On initial presentation, physical examination revealed diffuse erythema and scaling with associated edema of the face, trunk, and extremities (Figure 1). A biopsy from the patient’s right arm demonstrated a superficial perivascular inflammatory infiltrate composed of lymphocytes, histiocytes, and scattered eosinophils consistent with spongiotic dermatitis (Figure 2). Cyclosporine 225 mg twice daily and topical corticosteroids were started.

Over the next several months, the patient had several admissions secondary to recurrent skin abscesses in the setting of refractory erythroderma. He underwent trials of infliximab, corticosteroids, intravenous immunoglobulin, guselkumab, and acitretin with minimal improvement. He underwent an extensive laboratory and radiologic workup, which was notable for cyclical peripheral eosinophilia and elevated IgE levels correlating with the erythroderma flares. A second biopsy was obtained and continued to demonstrate changes consistent with AD.

Four months after the initial hospitalization, all psoriasis medications were stopped, and the patient was started on dupilumab 300 mg/2 mL every 2 weeks and an 8-week oral prednisone taper. This combination led to notable clinical improvement and resolution of peripheral eosinophilia. Several months after disease remission, he began to develop worsening erythema and pruritus on the trunk and extremities, followed by the development of new psoriatic lesions (Figure 3) with a biopsy consistent with psoriasis (Figure 4). The patient was continued on dupilumab, but cyclosporine was added. The patient self-discontinued dupilumab owing to injection-site discomfort and has been slowly weaning off oral cyclosporine with 1 to 2 remaining eczematous plaques and 1 to 2 psoriatic plaques managed by topical corticosteroids.

Comment
We present a patient with psoriasis that was well controlled on secukinumab who developed severe AD following treatment with secukinumab. The AD resolved following treatment with dupilumab and a tapering dose of prednisone. However, after several months of treatment with dupilumab alone, he began to develop psoriatic lesions again. This case supports findings in a case series describing the development of AD in patients with psoriasis treated with IL-17 inhibitors5 and a recent case report describing a patient with AD who developed psoriasis following treatment with an IL-4/IL-13 inhibitor.6
Recognized adverse effects demonstrate biologic medications’ contributions to both normal as well as aberrant immunologic responses. For example, IL-17 plays an essential role in innate and adaptive immune responses against infections at mucosal and cutaneous interfaces, as demonstrated by chronic mucocutaneous candidiasis in patients with genetic defects in IL-17–related pathways.7 Similarly, in patients taking IL-17 antagonists, an increase in the incidence of Candida infections has been observed.8 In patients with concurrent psoriasis and inflammatory bowel disease (IBD), treatment with IL-17 inhibitors is contraindicated due to the risk of exacerbating the IBD. This observation is somewhat paradoxical, as increased IL-17 release by TH17 cells is implicated in the pathogenesis of IBD.9 Interestingly, it is now thought that IL-17 may play a protective role in T-cell–driven intestinal inflammation through induction of protective intestinal epithelial gene expression and increased mucosal defense against gut microbes, explaining the worsening of IBD in patients on IL-17 inhibitors.10 These adverse effects illustrate the complicated and varied roles biologic medications play in immunologic response.
Given that TH1 and TH2 exert opposing immune mechanisms, it is uncommon for psoriasis and AD to coexist in a single patient. However, patients who exhibit concurrent findings may represent a unique population in which psoriasis and AD coexist, perhaps because of an underlying genetic predisposition. Moreover, targeted treatment of pathways unique to these disease processes may result in paradoxical flaring of the nontargeted pathway. It also is possible that inhibition of a specific T-cell pathway in a subset of patients will result in an immunologic imbalance, favoring increased activity of the opposing pathway in the absence of coexisting disease. In the case presented here, the findings may be explained by secukinumab’s inhibition of TH1/TH17-mediated inflammation, which resulted in a shift to a TH2-mediated inflammatory response manifesting as AD, as well as dupilumab’s inhibition of TH2-mediated inflammation, which caused a shift back to TH1-mediated inflammatory pathways. Additionally, for patients with changing morphologies exacerbated by biologic medications, alternative diagnoses, such as cutaneous T-cell lymphoma, may be considered.
Conclusion
We report an unusual case of secukinumab-induced AD in a patient with psoriasis that resolved following several months of treatment with dupilumab and a tapering dose of prednisone. Subsequently, this same patient developed re-emergence of psoriatic lesions with continued use of dupilumab, which was eventually discontinued by the patient despite appropriate disease control. In addition to illustrating the underlying pathophysiologic mechanisms of 2 common inflammatory dermatologic conditions, this case highlights how pharmacologic interventions targeted at specific immunologic pathways may have unintended consequences. Further investigation into the effects of targeted biologics on the TH1/TH2 immune axis is warranted to better understand the mechanism and possible implications of the phenotypic switching presented in this case.
Psoriasis vulgaris is a chronic inflammatory skin condition associated with notable elevation in helper T cell (TH) production of TH1/TH17-mediated inflammatory cytokines, including IL-17A.1 Upon binding of IL-17A to IL-17 receptors in the skin, an inflammatory cascade is triggered, resulting in the classic clinical appearance of psoriasis. Moderate to severe psoriasis often is managed by suppressing TH1/TH17-mediated inflammation using targeted immune therapy such as secukinumab, an IL-17A inhibitor.2 Atopic dermatitis (AD), another chronic inflammatory dermatosis, is associated with substantial elevation in TH2-mediated inflammatory cytokines, such as IL-4.3 Dupilumab, which interacts with IL-4R, disrupts the IL-4 and IL-13 signaling pathways and demonstrates considerable efficacy in the treatment of moderate to severe AD.4
A case series has shown that suppression of the TH1/TH17-mediated inflammation of psoriasis may paradoxically result in the development of TH2-mediated AD.5 Similarly, a recent case report described a patient who developed psoriasis following treatment of AD with dupilumab.6 Herein, we describe a patient with a history of psoriasis that was well controlled with secukinumab who developed severe refractory erythrodermic AD that resolved with dupilumab treatment. Following clearance of AD with dupilumab, he exhibited psoriasis recurrence.
Case Report
A 39-year-old man with a lifelong history of psoriasis was admitted to the hospital for management of severe erythroderma. Four years prior, secukinumab was initiated for treatment of psoriasis, resulting in excellent clinical response. He discontinued secukinumab after 2 years of treatment because of insurance coverage issues and managed his condition with only topical corticosteroids. He restarted secukinumab 10 months before admission because of a psoriasis flare. Shortly after resuming secukinumab, he developed a severe exfoliative erythroderma that was not responsive to corticosteroids, etanercept, methotrexate, or ustekinumab.

On initial presentation, physical examination revealed diffuse erythema and scaling with associated edema of the face, trunk, and extremities (Figure 1). A biopsy from the patient’s right arm demonstrated a superficial perivascular inflammatory infiltrate composed of lymphocytes, histiocytes, and scattered eosinophils consistent with spongiotic dermatitis (Figure 2). Cyclosporine 225 mg twice daily and topical corticosteroids were started.

Over the next several months, the patient had several admissions secondary to recurrent skin abscesses in the setting of refractory erythroderma. He underwent trials of infliximab, corticosteroids, intravenous immunoglobulin, guselkumab, and acitretin with minimal improvement. He underwent an extensive laboratory and radiologic workup, which was notable for cyclical peripheral eosinophilia and elevated IgE levels correlating with the erythroderma flares. A second biopsy was obtained and continued to demonstrate changes consistent with AD.

Four months after the initial hospitalization, all psoriasis medications were stopped, and the patient was started on dupilumab 300 mg/2 mL every 2 weeks and an 8-week oral prednisone taper. This combination led to notable clinical improvement and resolution of peripheral eosinophilia. Several months after disease remission, he began to develop worsening erythema and pruritus on the trunk and extremities, followed by the development of new psoriatic lesions (Figure 3) with a biopsy consistent with psoriasis (Figure 4). The patient was continued on dupilumab, but cyclosporine was added. The patient self-discontinued dupilumab owing to injection-site discomfort and has been slowly weaning off oral cyclosporine with 1 to 2 remaining eczematous plaques and 1 to 2 psoriatic plaques managed by topical corticosteroids.

Comment
We present a patient with psoriasis that was well controlled on secukinumab who developed severe AD following treatment with secukinumab. The AD resolved following treatment with dupilumab and a tapering dose of prednisone. However, after several months of treatment with dupilumab alone, he began to develop psoriatic lesions again. This case supports findings in a case series describing the development of AD in patients with psoriasis treated with IL-17 inhibitors5 and a recent case report describing a patient with AD who developed psoriasis following treatment with an IL-4/IL-13 inhibitor.6
Recognized adverse effects demonstrate biologic medications’ contributions to both normal as well as aberrant immunologic responses. For example, IL-17 plays an essential role in innate and adaptive immune responses against infections at mucosal and cutaneous interfaces, as demonstrated by chronic mucocutaneous candidiasis in patients with genetic defects in IL-17–related pathways.7 Similarly, in patients taking IL-17 antagonists, an increase in the incidence of Candida infections has been observed.8 In patients with concurrent psoriasis and inflammatory bowel disease (IBD), treatment with IL-17 inhibitors is contraindicated due to the risk of exacerbating the IBD. This observation is somewhat paradoxical, as increased IL-17 release by TH17 cells is implicated in the pathogenesis of IBD.9 Interestingly, it is now thought that IL-17 may play a protective role in T-cell–driven intestinal inflammation through induction of protective intestinal epithelial gene expression and increased mucosal defense against gut microbes, explaining the worsening of IBD in patients on IL-17 inhibitors.10 These adverse effects illustrate the complicated and varied roles biologic medications play in immunologic response.
Given that TH1 and TH2 exert opposing immune mechanisms, it is uncommon for psoriasis and AD to coexist in a single patient. However, patients who exhibit concurrent findings may represent a unique population in which psoriasis and AD coexist, perhaps because of an underlying genetic predisposition. Moreover, targeted treatment of pathways unique to these disease processes may result in paradoxical flaring of the nontargeted pathway. It also is possible that inhibition of a specific T-cell pathway in a subset of patients will result in an immunologic imbalance, favoring increased activity of the opposing pathway in the absence of coexisting disease. In the case presented here, the findings may be explained by secukinumab’s inhibition of TH1/TH17-mediated inflammation, which resulted in a shift to a TH2-mediated inflammatory response manifesting as AD, as well as dupilumab’s inhibition of TH2-mediated inflammation, which caused a shift back to TH1-mediated inflammatory pathways. Additionally, for patients with changing morphologies exacerbated by biologic medications, alternative diagnoses, such as cutaneous T-cell lymphoma, may be considered.
Conclusion
We report an unusual case of secukinumab-induced AD in a patient with psoriasis that resolved following several months of treatment with dupilumab and a tapering dose of prednisone. Subsequently, this same patient developed re-emergence of psoriatic lesions with continued use of dupilumab, which was eventually discontinued by the patient despite appropriate disease control. In addition to illustrating the underlying pathophysiologic mechanisms of 2 common inflammatory dermatologic conditions, this case highlights how pharmacologic interventions targeted at specific immunologic pathways may have unintended consequences. Further investigation into the effects of targeted biologics on the TH1/TH2 immune axis is warranted to better understand the mechanism and possible implications of the phenotypic switching presented in this case.
- Diani M, Altomare G, Reali E. T helper cell subsets in clinical manifestations of psoriasis. J Immunol Res. 2016;2016:7692024.
- Langley RG, Elewski BE, Lebwohl M, et al. Secukinumab in plaque psoriasis—results of two phase 3 trials. N Engl J Med. 2014;371:326-338.
- van der Heijden FL, Wierenga EA, Bos JD, et al. High frequency of IL-4-producing CD4+ allergen-specific T lymphocytes in atopic dermatitis lesional skin. J Invest Dermatol. 1991;97:389-394.
- Beck LA, Thaçi D, Hamilton JD, et al. Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. N Engl J Med. 2014;371:130-139.
- Lai FYX, Higgins E, Smith CH, et al. Morphologic switch from psoriasiform to eczematous dermatitis after anti-IL-17 therapy: a case series. JAMA Dermatol. 2019;155:1082-1084.
- Varma A, Levitt J. Dupilumab-induced phenotype switching from atopic dermatitis to psoriasis. JAAD Case Rep. 2020;6:217-218.
- Ling Y, Puel A. IL-17 and infections. Actas Dermosifiliogr. 2014;105(suppl 1):34-40.
- Saunte DM, Mrowietz U, Puig L, et al. Candida infections in patients with psoriasis and psoriatic arthritis treated with interleukin-17 inhibitors and their practical management. Br J Dermatol. 2017;177:47-62.
- Hölttä V, Klemetti P, Sipponen T, et al. IL-23/IL-17 immunity as a hallmark of Crohn’s disease. Inflamm Bowel Dis. 2008;14:1175-1184.
- Smith MK, Pai J, Panaccione R, et al. Crohn’s-like disease in a patient exposed to anti-interleukin-17 blockade (ixekizumab) for the treatment of chronic plaque psoriasis: a case report. BMC Gastroenterol. 2019;19:162.
- Diani M, Altomare G, Reali E. T helper cell subsets in clinical manifestations of psoriasis. J Immunol Res. 2016;2016:7692024.
- Langley RG, Elewski BE, Lebwohl M, et al. Secukinumab in plaque psoriasis—results of two phase 3 trials. N Engl J Med. 2014;371:326-338.
- van der Heijden FL, Wierenga EA, Bos JD, et al. High frequency of IL-4-producing CD4+ allergen-specific T lymphocytes in atopic dermatitis lesional skin. J Invest Dermatol. 1991;97:389-394.
- Beck LA, Thaçi D, Hamilton JD, et al. Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. N Engl J Med. 2014;371:130-139.
- Lai FYX, Higgins E, Smith CH, et al. Morphologic switch from psoriasiform to eczematous dermatitis after anti-IL-17 therapy: a case series. JAMA Dermatol. 2019;155:1082-1084.
- Varma A, Levitt J. Dupilumab-induced phenotype switching from atopic dermatitis to psoriasis. JAAD Case Rep. 2020;6:217-218.
- Ling Y, Puel A. IL-17 and infections. Actas Dermosifiliogr. 2014;105(suppl 1):34-40.
- Saunte DM, Mrowietz U, Puig L, et al. Candida infections in patients with psoriasis and psoriatic arthritis treated with interleukin-17 inhibitors and their practical management. Br J Dermatol. 2017;177:47-62.
- Hölttä V, Klemetti P, Sipponen T, et al. IL-23/IL-17 immunity as a hallmark of Crohn’s disease. Inflamm Bowel Dis. 2008;14:1175-1184.
- Smith MK, Pai J, Panaccione R, et al. Crohn’s-like disease in a patient exposed to anti-interleukin-17 blockade (ixekizumab) for the treatment of chronic plaque psoriasis: a case report. BMC Gastroenterol. 2019;19:162.
Practice Points
- Treatment of psoriasis vulgaris, a helper T cell TH1/TH17-mediated skin condition, with secukinumab may result in phenotypic switching to TH2-mediated atopic dermatitis.
- Atopic dermatitis responds well to dupilumab but may result in phenotypic switching to psoriasis.
- Biologic therapies targeted at specific immunologic pathways may have unintended consequences on the TH1/TH2 immune axis.
Just Like Rock and Roll, Topical Medications for Psoriasis Are Here to Stay
When I finished my dermatology training in 1986, the only moving parts in the skin that I recall were keratinocytes moving upward from the basal layer of the epidermis until they were desquamated 4 or 5 weeks later and hairs growing within their follicles until they were shed. Now we are learning about countless cytokines, chemokines, interleukins, antibodies, receptors, enzymes, and cell types, as well as their associated pathways, at an endless pace. Every day I am looking in my inbox to sign up for the “Cytokine of the Month” club! Despite the challenges of sorting through what is relevant clinically, it is a very exciting time. Coupled with this myriad of fundamental science is the emergence of newer therapies that are more directly targeting specific disease states and dramatically changing the lives of patients. We see prominent examples of these therapeutic results every day in patients we treat, especially with psoriasis and atopic dermatitis. Importantly, there also is hope for patients with notoriously refractory skin disorders, such as hidradenitis suppurativa, alopecia areata, and vitiligo, as newer therapies are being thoroughly studied in clinical trials.
Despite the best advances in therapy that we currently have available and those anticipated in the foreseeable future, patients with chronic dermatoses such as psoriasis and atopic dermatitis still require prolonged constant or frequently used intermittent therapies to adequately control their disease. Fortunately, as dermatologists we understand the importance of proper skin care and topical medications as well as how to incorporate them in the management plan. To date, specifically with psoriasis, we have a variety of brand and generic topical corticosteroids, calcipotriene (vitamin D analogue), and tazarotene (retinoid), as well as combination formulations, in our toolbox to help manage localized areas of involvement.1 This includes both patients with more limited psoriasis and those responding favorably to systemic therapy but who still develop some new or persistent areas of localized psoriatic lesions. New data with the brand formulation of calcipotriene–betamethasone dipropionate (Cal-BDP) foam applied once daily shows that after adequate control is achieved, continued application to the affected sites twice weekly is superior to vehicle in preventing relapse of psoriasis.2 A highly cosmetically acceptable Cal-BDP cream incorporating a unique vehicle technology has been US Food and Drug Administration (FDA) approved for once-daily use for plaque psoriasis, overcoming the compatibility difficulties encountered in combining both active ingredients in an aqueous-based formulation and also optimizing the delivery of the active ingredients into the skin. This Cal-BDP cream demonstrated efficacy superior to a brand Cal-BDP suspension, rapid reduction in pruritus, and favorable tolerability and safety.3 Another combination formulation that is FDA approved for plaque psoriasis with once-daily application that has been shown to be effective and safe is halobetasol propionate–tazarotene lotion. This formulation contains lower concentrations of both active ingredients than those normally used in a barrier-friendly polymeric emulsion vehicle, allowing for augmented delivery of both active ingredients into the skin than with the individual agents applied separately and sequentially.4,5 In the best of circumstances, most patients with psoriasis still require use of topical therapy and appreciate its availability. Just like on any menu, it is good to have multiple good options.
What else does this psoriasis management story need? A pipeline! I am happy to tell you that with topical therapy, 2 nonsteroidal agents are under development with completion of phase 2 and phase 3 trials submitted to the FDA to evaluate for approval for psoriasis. They are tapinarof cream, an aryl hydrocarbon receptor agonist, and roflumilast cream, a phosphodiesterase 4 (PDE4) inhibitor. Both of these modes of action involve intracellular pathways that are highly conserved in humans and are ubiquitously present in structural and hematopoietic cells.
Topical application of tapinarof cream once daily has been shown to be effective and safe for plaque psoriasis, is well tolerated with some reports of folliculitis observed that did not typically interfere with use, exhibits a remittive effect in patients achieving clearance on therapy, and is devoid of any systemic safety signals with both short-term and long-term use.6-8 It also is currently under evaluation for atopic dermatitis. Topical roflumilast cream once daily has been shown to be effective and safe for plaque psoriasis as well as intertriginous psoriasis; is well tolerated including negligible rates of skin tolerability reactions such as stinging and burning; and is devoid of systemic safety signals, including those often observed with oral PDE4 inhibitor therapy (apremilast).9,10 In addition, roflumilast has been shown to be more inherently potent in PDE4 inhibition activity than crisaborole and apremilast.11 Roflumilast cream also is being studied for atopic dermatitis and a foam formulation is being evaluated for seborrheic dermatitis. Importantly, both tapinarof and roflumilast are not corticosteroids and are not associated with adverse effects observed with topical corticosteroid therapy, such as atrophy, striae, telangiectasia, and hypothalamic-pituitary-adrenal axis suppression. This provides a sense of comfort for clinicians and patients, as potential side effects associated with more prolonged topical corticosteroid therapy are common and lingering concerns.
To summarize, topical therapy for psoriasis is here to stay, just like all the rock and roll we have more access to than ever through expanded modern-day radio access and several music streaming sources, most of which are on demand. Also available to us are some viable current options, including a few newer brand formulations. New nonsteroidal agents with favorable data thus far are on the horizon, providing their own inherent efficacy and safety, which appear to be advantageous thus far. As the late Ric Ocasek of the Cars sang, “Let the good times roll.”
- Lebwohl MG, Van de Kerkhof PCM. Psoriasis. In: Lebwohl MG, Heymann WR, Berth-Jones J, et al, eds. Treatment of Skin Disease: Comprehensive Therapeutic Strategies. 4th ed. Elsevier Saunders; 2014:640-650.
- Lebwohl M, Kircik L, Lacour JP, et al. Twice-weekly topical calcipotriene/betamethasone dipropionate foam as proactive management of plaque psoriasis increases time in remission and is well tolerated over 52 weeks (PSO-LONG trial). J Am Acad Dermatol. 2021;84:1269-1277.
- Wynzora (calcipotriene and betamethasone dipropionate) cream, for topical use. Package insert. EPI Health, LLC; 2020.
- Ramachandran V, Bertus B, Bashyam AM, et al. Treating psoriasis with halobetasol propionate and tazarotene combination: a review of phase II and III clinical trials. Ann Pharmacother. 2020;54:872-878.
- Lebwohl MG, Tanghetti EA, Stein Gold L, et al. Fixed-combination halobetasol propionate and tazarotene in the treatment of psoriasis: narrative review of mechanisms of action and therapeutic benefits. Dermatol Ther (Heidelb). 2021;11:1157-1174.
- Bissonnette R, Stein Gold L, Rubenstein DS, et al. Tapinarof in the treatment of psoriasis: a review of the unique mechanism of action of a novel therapeutic aryl hydrocarbon receptor-modulating agent. J Am Acad Dermatol. 2021;84:1059-1067.
- Lebwohl MG, Stein Gold L, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229.
- Jett JE, McLaughlin M, Lee MS, et al. Tapinarof cream 1% for extensive plaque psoriasis: a maximal use trial on safety, tolerability, and pharmacokinetics [published online October 28, 2021]. Am J Clin Dermatol. doi:10.100/s40257-021-00641-4
- Lebwohl MG, Papp KA, Stein Gold L, et al. Trial of roflumilast cream for chronic plaque psoriasis. N Engl J Med. 2020;383:229-239.
- Papp KA, Gooderham M, Droege M, et al. Roflumilast cream improves signs and symptoms of plaque psoriasis: results from a phase 1/2a randomized, controlled study. J Drugs Dermatol. 2020;19:734-740.
- Dong C, Virtucio C, Zemska O, et al. Treatment of skin inflammation with benzoxaborole phosphodiesterase inhibitors: selectivity, cellular activity, and effect on cytokines associated with skin inflammation and skin architecture changes. J Pharmacol Exp Ther. 2016;358:413-422.
When I finished my dermatology training in 1986, the only moving parts in the skin that I recall were keratinocytes moving upward from the basal layer of the epidermis until they were desquamated 4 or 5 weeks later and hairs growing within their follicles until they were shed. Now we are learning about countless cytokines, chemokines, interleukins, antibodies, receptors, enzymes, and cell types, as well as their associated pathways, at an endless pace. Every day I am looking in my inbox to sign up for the “Cytokine of the Month” club! Despite the challenges of sorting through what is relevant clinically, it is a very exciting time. Coupled with this myriad of fundamental science is the emergence of newer therapies that are more directly targeting specific disease states and dramatically changing the lives of patients. We see prominent examples of these therapeutic results every day in patients we treat, especially with psoriasis and atopic dermatitis. Importantly, there also is hope for patients with notoriously refractory skin disorders, such as hidradenitis suppurativa, alopecia areata, and vitiligo, as newer therapies are being thoroughly studied in clinical trials.
Despite the best advances in therapy that we currently have available and those anticipated in the foreseeable future, patients with chronic dermatoses such as psoriasis and atopic dermatitis still require prolonged constant or frequently used intermittent therapies to adequately control their disease. Fortunately, as dermatologists we understand the importance of proper skin care and topical medications as well as how to incorporate them in the management plan. To date, specifically with psoriasis, we have a variety of brand and generic topical corticosteroids, calcipotriene (vitamin D analogue), and tazarotene (retinoid), as well as combination formulations, in our toolbox to help manage localized areas of involvement.1 This includes both patients with more limited psoriasis and those responding favorably to systemic therapy but who still develop some new or persistent areas of localized psoriatic lesions. New data with the brand formulation of calcipotriene–betamethasone dipropionate (Cal-BDP) foam applied once daily shows that after adequate control is achieved, continued application to the affected sites twice weekly is superior to vehicle in preventing relapse of psoriasis.2 A highly cosmetically acceptable Cal-BDP cream incorporating a unique vehicle technology has been US Food and Drug Administration (FDA) approved for once-daily use for plaque psoriasis, overcoming the compatibility difficulties encountered in combining both active ingredients in an aqueous-based formulation and also optimizing the delivery of the active ingredients into the skin. This Cal-BDP cream demonstrated efficacy superior to a brand Cal-BDP suspension, rapid reduction in pruritus, and favorable tolerability and safety.3 Another combination formulation that is FDA approved for plaque psoriasis with once-daily application that has been shown to be effective and safe is halobetasol propionate–tazarotene lotion. This formulation contains lower concentrations of both active ingredients than those normally used in a barrier-friendly polymeric emulsion vehicle, allowing for augmented delivery of both active ingredients into the skin than with the individual agents applied separately and sequentially.4,5 In the best of circumstances, most patients with psoriasis still require use of topical therapy and appreciate its availability. Just like on any menu, it is good to have multiple good options.
What else does this psoriasis management story need? A pipeline! I am happy to tell you that with topical therapy, 2 nonsteroidal agents are under development with completion of phase 2 and phase 3 trials submitted to the FDA to evaluate for approval for psoriasis. They are tapinarof cream, an aryl hydrocarbon receptor agonist, and roflumilast cream, a phosphodiesterase 4 (PDE4) inhibitor. Both of these modes of action involve intracellular pathways that are highly conserved in humans and are ubiquitously present in structural and hematopoietic cells.
Topical application of tapinarof cream once daily has been shown to be effective and safe for plaque psoriasis, is well tolerated with some reports of folliculitis observed that did not typically interfere with use, exhibits a remittive effect in patients achieving clearance on therapy, and is devoid of any systemic safety signals with both short-term and long-term use.6-8 It also is currently under evaluation for atopic dermatitis. Topical roflumilast cream once daily has been shown to be effective and safe for plaque psoriasis as well as intertriginous psoriasis; is well tolerated including negligible rates of skin tolerability reactions such as stinging and burning; and is devoid of systemic safety signals, including those often observed with oral PDE4 inhibitor therapy (apremilast).9,10 In addition, roflumilast has been shown to be more inherently potent in PDE4 inhibition activity than crisaborole and apremilast.11 Roflumilast cream also is being studied for atopic dermatitis and a foam formulation is being evaluated for seborrheic dermatitis. Importantly, both tapinarof and roflumilast are not corticosteroids and are not associated with adverse effects observed with topical corticosteroid therapy, such as atrophy, striae, telangiectasia, and hypothalamic-pituitary-adrenal axis suppression. This provides a sense of comfort for clinicians and patients, as potential side effects associated with more prolonged topical corticosteroid therapy are common and lingering concerns.
To summarize, topical therapy for psoriasis is here to stay, just like all the rock and roll we have more access to than ever through expanded modern-day radio access and several music streaming sources, most of which are on demand. Also available to us are some viable current options, including a few newer brand formulations. New nonsteroidal agents with favorable data thus far are on the horizon, providing their own inherent efficacy and safety, which appear to be advantageous thus far. As the late Ric Ocasek of the Cars sang, “Let the good times roll.”
When I finished my dermatology training in 1986, the only moving parts in the skin that I recall were keratinocytes moving upward from the basal layer of the epidermis until they were desquamated 4 or 5 weeks later and hairs growing within their follicles until they were shed. Now we are learning about countless cytokines, chemokines, interleukins, antibodies, receptors, enzymes, and cell types, as well as their associated pathways, at an endless pace. Every day I am looking in my inbox to sign up for the “Cytokine of the Month” club! Despite the challenges of sorting through what is relevant clinically, it is a very exciting time. Coupled with this myriad of fundamental science is the emergence of newer therapies that are more directly targeting specific disease states and dramatically changing the lives of patients. We see prominent examples of these therapeutic results every day in patients we treat, especially with psoriasis and atopic dermatitis. Importantly, there also is hope for patients with notoriously refractory skin disorders, such as hidradenitis suppurativa, alopecia areata, and vitiligo, as newer therapies are being thoroughly studied in clinical trials.
Despite the best advances in therapy that we currently have available and those anticipated in the foreseeable future, patients with chronic dermatoses such as psoriasis and atopic dermatitis still require prolonged constant or frequently used intermittent therapies to adequately control their disease. Fortunately, as dermatologists we understand the importance of proper skin care and topical medications as well as how to incorporate them in the management plan. To date, specifically with psoriasis, we have a variety of brand and generic topical corticosteroids, calcipotriene (vitamin D analogue), and tazarotene (retinoid), as well as combination formulations, in our toolbox to help manage localized areas of involvement.1 This includes both patients with more limited psoriasis and those responding favorably to systemic therapy but who still develop some new or persistent areas of localized psoriatic lesions. New data with the brand formulation of calcipotriene–betamethasone dipropionate (Cal-BDP) foam applied once daily shows that after adequate control is achieved, continued application to the affected sites twice weekly is superior to vehicle in preventing relapse of psoriasis.2 A highly cosmetically acceptable Cal-BDP cream incorporating a unique vehicle technology has been US Food and Drug Administration (FDA) approved for once-daily use for plaque psoriasis, overcoming the compatibility difficulties encountered in combining both active ingredients in an aqueous-based formulation and also optimizing the delivery of the active ingredients into the skin. This Cal-BDP cream demonstrated efficacy superior to a brand Cal-BDP suspension, rapid reduction in pruritus, and favorable tolerability and safety.3 Another combination formulation that is FDA approved for plaque psoriasis with once-daily application that has been shown to be effective and safe is halobetasol propionate–tazarotene lotion. This formulation contains lower concentrations of both active ingredients than those normally used in a barrier-friendly polymeric emulsion vehicle, allowing for augmented delivery of both active ingredients into the skin than with the individual agents applied separately and sequentially.4,5 In the best of circumstances, most patients with psoriasis still require use of topical therapy and appreciate its availability. Just like on any menu, it is good to have multiple good options.
What else does this psoriasis management story need? A pipeline! I am happy to tell you that with topical therapy, 2 nonsteroidal agents are under development with completion of phase 2 and phase 3 trials submitted to the FDA to evaluate for approval for psoriasis. They are tapinarof cream, an aryl hydrocarbon receptor agonist, and roflumilast cream, a phosphodiesterase 4 (PDE4) inhibitor. Both of these modes of action involve intracellular pathways that are highly conserved in humans and are ubiquitously present in structural and hematopoietic cells.
Topical application of tapinarof cream once daily has been shown to be effective and safe for plaque psoriasis, is well tolerated with some reports of folliculitis observed that did not typically interfere with use, exhibits a remittive effect in patients achieving clearance on therapy, and is devoid of any systemic safety signals with both short-term and long-term use.6-8 It also is currently under evaluation for atopic dermatitis. Topical roflumilast cream once daily has been shown to be effective and safe for plaque psoriasis as well as intertriginous psoriasis; is well tolerated including negligible rates of skin tolerability reactions such as stinging and burning; and is devoid of systemic safety signals, including those often observed with oral PDE4 inhibitor therapy (apremilast).9,10 In addition, roflumilast has been shown to be more inherently potent in PDE4 inhibition activity than crisaborole and apremilast.11 Roflumilast cream also is being studied for atopic dermatitis and a foam formulation is being evaluated for seborrheic dermatitis. Importantly, both tapinarof and roflumilast are not corticosteroids and are not associated with adverse effects observed with topical corticosteroid therapy, such as atrophy, striae, telangiectasia, and hypothalamic-pituitary-adrenal axis suppression. This provides a sense of comfort for clinicians and patients, as potential side effects associated with more prolonged topical corticosteroid therapy are common and lingering concerns.
To summarize, topical therapy for psoriasis is here to stay, just like all the rock and roll we have more access to than ever through expanded modern-day radio access and several music streaming sources, most of which are on demand. Also available to us are some viable current options, including a few newer brand formulations. New nonsteroidal agents with favorable data thus far are on the horizon, providing their own inherent efficacy and safety, which appear to be advantageous thus far. As the late Ric Ocasek of the Cars sang, “Let the good times roll.”
- Lebwohl MG, Van de Kerkhof PCM. Psoriasis. In: Lebwohl MG, Heymann WR, Berth-Jones J, et al, eds. Treatment of Skin Disease: Comprehensive Therapeutic Strategies. 4th ed. Elsevier Saunders; 2014:640-650.
- Lebwohl M, Kircik L, Lacour JP, et al. Twice-weekly topical calcipotriene/betamethasone dipropionate foam as proactive management of plaque psoriasis increases time in remission and is well tolerated over 52 weeks (PSO-LONG trial). J Am Acad Dermatol. 2021;84:1269-1277.
- Wynzora (calcipotriene and betamethasone dipropionate) cream, for topical use. Package insert. EPI Health, LLC; 2020.
- Ramachandran V, Bertus B, Bashyam AM, et al. Treating psoriasis with halobetasol propionate and tazarotene combination: a review of phase II and III clinical trials. Ann Pharmacother. 2020;54:872-878.
- Lebwohl MG, Tanghetti EA, Stein Gold L, et al. Fixed-combination halobetasol propionate and tazarotene in the treatment of psoriasis: narrative review of mechanisms of action and therapeutic benefits. Dermatol Ther (Heidelb). 2021;11:1157-1174.
- Bissonnette R, Stein Gold L, Rubenstein DS, et al. Tapinarof in the treatment of psoriasis: a review of the unique mechanism of action of a novel therapeutic aryl hydrocarbon receptor-modulating agent. J Am Acad Dermatol. 2021;84:1059-1067.
- Lebwohl MG, Stein Gold L, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229.
- Jett JE, McLaughlin M, Lee MS, et al. Tapinarof cream 1% for extensive plaque psoriasis: a maximal use trial on safety, tolerability, and pharmacokinetics [published online October 28, 2021]. Am J Clin Dermatol. doi:10.100/s40257-021-00641-4
- Lebwohl MG, Papp KA, Stein Gold L, et al. Trial of roflumilast cream for chronic plaque psoriasis. N Engl J Med. 2020;383:229-239.
- Papp KA, Gooderham M, Droege M, et al. Roflumilast cream improves signs and symptoms of plaque psoriasis: results from a phase 1/2a randomized, controlled study. J Drugs Dermatol. 2020;19:734-740.
- Dong C, Virtucio C, Zemska O, et al. Treatment of skin inflammation with benzoxaborole phosphodiesterase inhibitors: selectivity, cellular activity, and effect on cytokines associated with skin inflammation and skin architecture changes. J Pharmacol Exp Ther. 2016;358:413-422.
- Lebwohl MG, Van de Kerkhof PCM. Psoriasis. In: Lebwohl MG, Heymann WR, Berth-Jones J, et al, eds. Treatment of Skin Disease: Comprehensive Therapeutic Strategies. 4th ed. Elsevier Saunders; 2014:640-650.
- Lebwohl M, Kircik L, Lacour JP, et al. Twice-weekly topical calcipotriene/betamethasone dipropionate foam as proactive management of plaque psoriasis increases time in remission and is well tolerated over 52 weeks (PSO-LONG trial). J Am Acad Dermatol. 2021;84:1269-1277.
- Wynzora (calcipotriene and betamethasone dipropionate) cream, for topical use. Package insert. EPI Health, LLC; 2020.
- Ramachandran V, Bertus B, Bashyam AM, et al. Treating psoriasis with halobetasol propionate and tazarotene combination: a review of phase II and III clinical trials. Ann Pharmacother. 2020;54:872-878.
- Lebwohl MG, Tanghetti EA, Stein Gold L, et al. Fixed-combination halobetasol propionate and tazarotene in the treatment of psoriasis: narrative review of mechanisms of action and therapeutic benefits. Dermatol Ther (Heidelb). 2021;11:1157-1174.
- Bissonnette R, Stein Gold L, Rubenstein DS, et al. Tapinarof in the treatment of psoriasis: a review of the unique mechanism of action of a novel therapeutic aryl hydrocarbon receptor-modulating agent. J Am Acad Dermatol. 2021;84:1059-1067.
- Lebwohl MG, Stein Gold L, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229.
- Jett JE, McLaughlin M, Lee MS, et al. Tapinarof cream 1% for extensive plaque psoriasis: a maximal use trial on safety, tolerability, and pharmacokinetics [published online October 28, 2021]. Am J Clin Dermatol. doi:10.100/s40257-021-00641-4
- Lebwohl MG, Papp KA, Stein Gold L, et al. Trial of roflumilast cream for chronic plaque psoriasis. N Engl J Med. 2020;383:229-239.
- Papp KA, Gooderham M, Droege M, et al. Roflumilast cream improves signs and symptoms of plaque psoriasis: results from a phase 1/2a randomized, controlled study. J Drugs Dermatol. 2020;19:734-740.
- Dong C, Virtucio C, Zemska O, et al. Treatment of skin inflammation with benzoxaborole phosphodiesterase inhibitors: selectivity, cellular activity, and effect on cytokines associated with skin inflammation and skin architecture changes. J Pharmacol Exp Ther. 2016;358:413-422.
Adjunctive Use of Halobetasol Propionate–Tazarotene in Biologic-Experienced Patients With Psoriasis
Psoriasis is a common chronic immunologic skin disease that affects approximately 7.4 million adults in the United States1 and more than 100 million individuals worldwide.2 Patients with psoriasis have a potentially heightened risk for cardiometabolic diseases, psychiatric disorders, and psoriatic arthritis,3 as well as impaired quality of life (QOL).4 Psoriasis also is associated with increased health care costs5 and may result in substantial socioeconomic repercussions for affected patients.6,7
Psoriasis treatments focus on relieving symptoms and improving patient QOL. Systemic therapy has been the mainstay of treatment for moderate to severe psoriasis.8 Although topical therapy usually is applied to treat mild symptoms, it also can be used as an adjunct to enhance efficacy of other treatment approaches.9 The National Psoriasis Foundation (NPF) recommends a treat-to-target (TTT) strategy for plaque psoriasis, the most common form of psoriasis, with a target response of attaining affected body surface area (BSA) of 1% or lower at 3 months after treatment initiation, allowing for regular assessments of treatment responses.10
Not all patients with moderate to severe psoriasis can achieve a satisfactory response with systemic biologic monotherapy.11 Switching to a new biologic improves responses in some but not all cases12 and could be associated with new safety issues and additional costs. Combinations of biologics with phototherapy, nonbiologic systemic agents, or topical medications were found to be more effective than biologics alone,9,11 though long-term safety studies are needed for biologics combined with other systemic inverventions.11
A lotion containing a fixed combination of halobetasol propionate (HP) 0.01%, a corticosteroid, and tazarotene (TAZ) 0.045%, a retinoid, is indicated for plaque psoriasis in adults.13 Two randomized, controlled, phase 3 trials demonstrated the rapid and sustained efficacy of HP-TAZ in treating moderate to severe plaque psoriasis without any safety concerns.14,15 However, combining HP-TAZ lotion with biologics has not been examined yet, to our knowledge.
This open-label study evaluated the effectiveness and safety of adjunctive HP-TAZ lotion in adult patients with moderate to severe plaque psoriasis who were being treated with biologics in a real-world setting. Potential cost savings with the addition of topical HP-TAZ to ongoing biologics vs switching to a new biologic also were assessed.
Methods
Study Design and Participants—A single-center, institutional review board–approved, open-label study evaluated adjunctive therapy with HP 0.01%–TAZ 0.045% lotion in patients with psoriasis being treated with biologic agents. The study was conducted in accordance with the ethical principles of the Declaration of Helsinki and in compliance with Good Clinical Practices. All patients provided written informed consent before enrollment.
Male and nonpregnant female patients (aged ≥18 years)with moderate to severe chronic plaque psoriasis and a BSA of 2% to 10% who were being treated with biologics for at least 24 weeks at baseline were enrolled. Patients were excluded if they had used oral systemic medications for psoriasis (≤4 weeks), other topical antipsoriatic therapies (≤14 days), UVB phototherapy (≤2 weeks), and psoralen plus UVA phototherapy (≤4 weeks) prior to study initiation. Concomitant use of steroid-free topical emollients or low-potency topical steroids and appropriate interventions deemed necessary by the investigator were allowed.
Although participants maintained their prescribed biologics for the duration of the study, HP-TAZ lotion also was applied once daily for 8 weeks, followed by once every other day for an additional 4 weeks. Participants then continued with biologics only for the last 4 weeks of the study.
Study Outcome Measures—Disease severity and treatment efficacy were assessed by affected BSA, Physician Global Assessment (PGA) score, composite BSA×PGA score, and participant-reported Dermatology Life Quality Index (DLQI). The primary end point was the proportion of participants achieving a BSA of 0% to 1% (NPF TTT status) at week 8. Secondary end points included the proportions of participants with BSA of 0% to 1% at weeks 12 and 16; BSA×PGA score at weeks 8, 12, and 16; and improvements in BSA, PGA, and DLQI at weeks 8, 12, and 16.
Adverse events (AEs) that occurred after the signing of the informed consent and for the duration of the participant’s participation were recorded, regardless of causality. Physical examinations were performed at screening; baseline; and weeks 8, 12, and 16 to document any clinically significant abnormalities. Localized skin reactions were assessed for tolerability of the study drug, with any reaction requiring concomitant therapy recorded as an AE.
The likelihood of switching to a new biologic regimen was assessed by the investigator for each participant at baseline and weeks 8, 12, and 16. Participants with unacceptable responses to their treatments (BSA >3%) were reported as likely to be considered for switching biologics by the investigator.
Pharmacoeconomic Evaluation—Potential cost savings were evaluated for the addition of HP-TAZ lotion to ongoing biologics vs switching to a new biologic. Cost comparisons were made in participants for whom the investigator would likely have switched biologics at baseline but not at the end of the study. For maintaining the same biologic with adjunctive topical HP-TAZ, total cost was estimated by adding the cost for 12 weeks (once daily for 8 weeks and once every other day for 4 weeks) of the HP-TAZ lotion to that of 16-week maintenance dosing with the biologic. The projected cost for switching to a new biologic for 16 weeks of treatment was based on both induction and maintenance dosing as recommended in its product label. Prices were obtained from the 2020 average wholesale price specialty pharmacy reports (BioPlus Specialty Pharmacy Services [https://www.bioplusrx.com]).
Data Handling—Enrollment of approximately 25 participants was desired for the study. Data on disease severity and participant-reported outcomes were assessed using descriptive statistics. Adverse events were summarized descriptively by incidence, severity, and relationship to the study drug. All participants with data available at a measured time point were included in the analyses for that time point.
Results
Participant Disposition and Demographics—Twenty-five participants (15 male and 10 female) were included in the study (Table 1). Seven participants discontinued the study for the following reasons: AEs (n=4), patient choice (n=2), and noncompliance (n=1).

The average age of the participants was 50 years, the majority were White (76.0% [19/25]) andnon-Hispanic (88.0% [22/25]), and the mean duration of chronic plaque psoriasis was 18.9 years (Table 1). Participants had been receiving biologic monotherapy for at least 24 weeks prior to enrollment, most commonly ustekinumab (32.0% [8/25])(Table 1). None had achieved the NPF TTT status with their biologics. At baseline, mean (SD) affected BSA, PGA, BSA×PGA, and participant-reported DLQI were 4.16% (2.04%), 2.84 (0.55), 11.88 (6.39), and 4.00 (4.74), respectively.
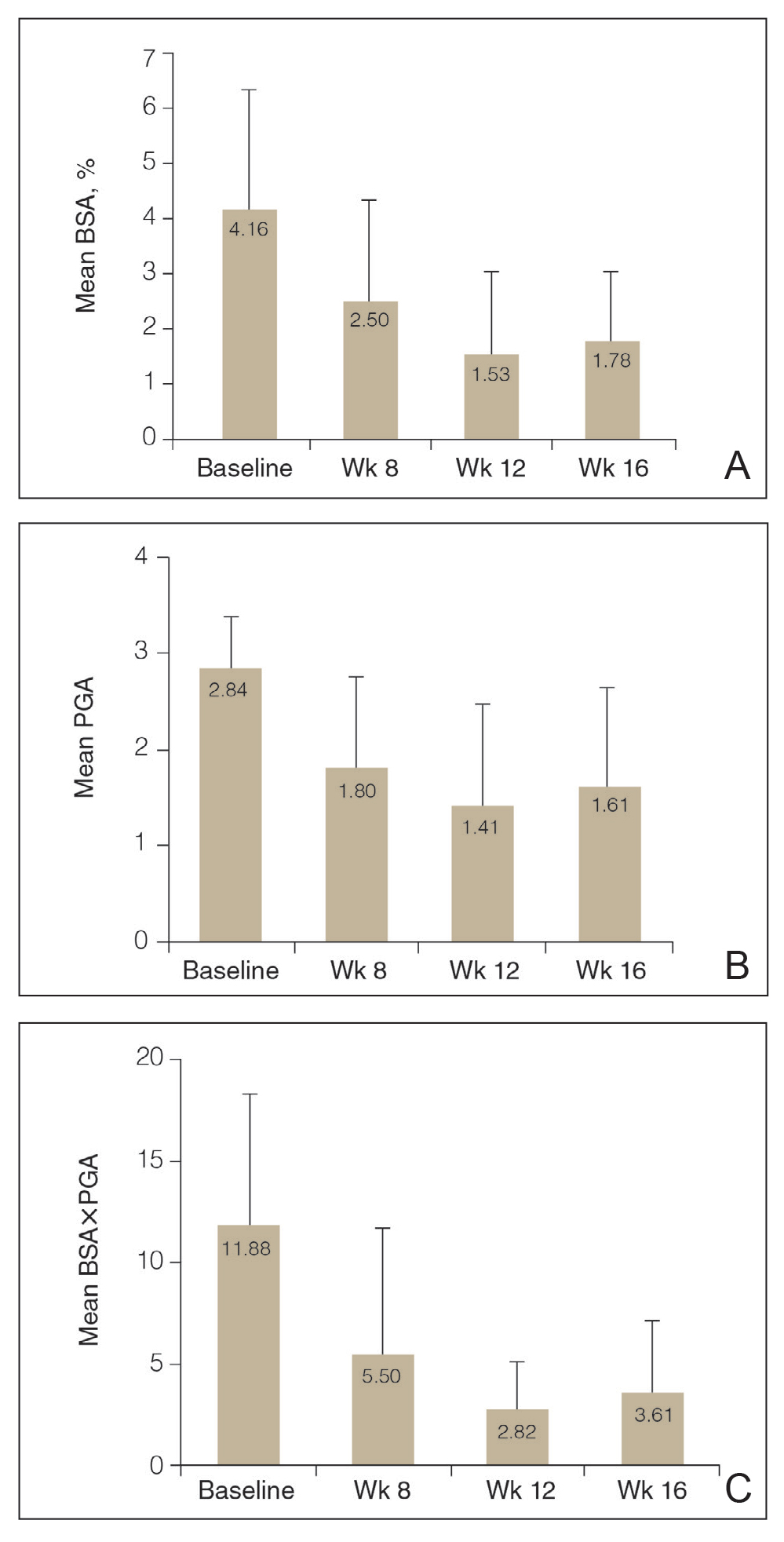
Efficacy Assessment—Application of HP-TAZ lotion in addition to the participants’ existing biologic therapy reduced severity of the disease, as evidenced by the reductions in mean BSA, PGA, and BSA×PGA. After 8 weeks of once-daily concomitant HP-TAZ use with biologic, mean BSA and PGA dropped by approximately 40% and 37%, respectively (Figures 1A and 1B). A greater reduction (54%) was found for mean BSA×PGA (Figure 1C). Disease severity continued to improve when the application schedule for HP-TAZ was changed to once every other day for 4 weeks, as mean BSA, PGA, and BSA×PGA decreased further at week 12. These beneficial effects were sustained during the last 4 weeks of the study after HP-TAZ was discontinued, with reductions of 57%, 43%, and 70% from baseline for mean BSA, PGA, and BSA×PGA, respectively (Figure 1).
The proportion of participants who achieved NPF TTT status increased from 0% at baseline to 20.0% (5/20) at week 8 with once-daily use of HP-TAZ plus biologic for 8 weeks (Figure 2). At week 12, more participants (64.7% [11/17]) achieved the treatment goal after application of HP-TAZ once every other day with biologic for 4 weeks. Most participants maintained NPF TTT status after HP-TAZ was discontinued; at week 16, 50.0% (9/18) attained the NPF treatment goal (Figure 2).
![Proportion of participants achieving National Psoriasis Foundation target-to-treat status (body surface area [BSA] ≤1%) at baseline and weeks 8, 12, and 16 Proportion of participants achieving National Psoriasis Foundation target-to-treat status (body surface area [BSA] ≤1%) at baseline and weeks 8, 12, and 16](https://cdn.mdedge.com/files/s3fs-public/Bagel_0222_2.JPG)
The mean DLQI score decreased from 4.00 at baseline to 2.45 after 8 weeks of concomitant use of once-daily HP-TAZ with biologic, reflecting a 39% score reduction. An additional 4 weeks of adjunctive HP-TAZ applied once every other day with biologic further decreased the DLQI score to 2.18 at week 12. Mean DLQI remained similar (2.33) after another 4 weeks of biologics alone. The proportion of participants reporting a DLQI score of 0 to 1 increased from 40% (10/25) at baseline to 60% (12/20) at week 8 and 76.5% (13/17) at week 12 with adjunctive HP-TAZ lotion use with biologic. At week 16, a DLQI score of 0 to 1 was reported in 61.1% (11/18) of participants after receiving only biologics for 4 weeks.
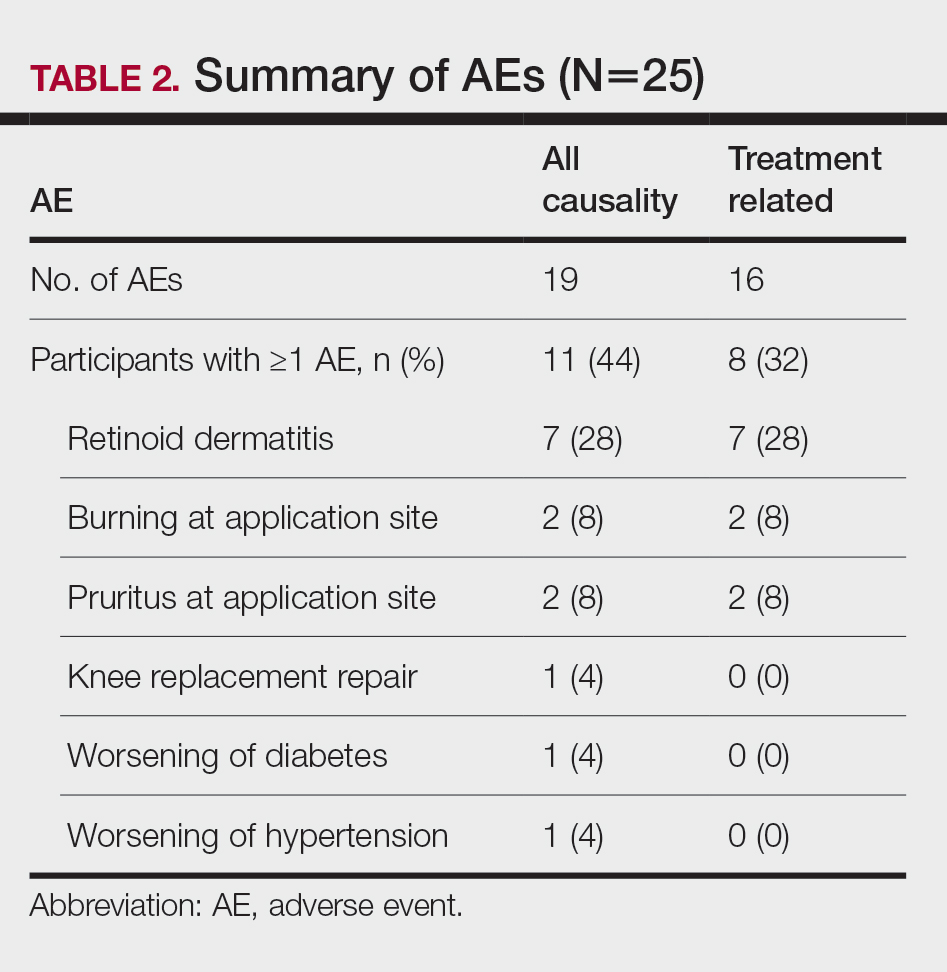
Safety Assessment—A total of 19 AEs were reported in 11 participants during the study; 16 AEs were considered treatment related in 8 participants (Table 2). The most common AEs were retinoid dermatitis (28% [7/25]), burning at the application site (8% [2/25]), and pruritus at the application site (8% [2/25]), all of which were considered related to the treatment. Among all AEs, 12 were mild in severity, and the remaining 7 were moderate. Adverse events led to early study termination in 4 participants, all with retinoid dermatitis as the primary reason.
Likelihood of Switching Biologics—At baseline, almost 90% (22/25) of participants were rated as likely to switch biologics by the investigator due to unacceptable responses to their currently prescribed biologics (BSA >3%)(Figure 3). The likelihood was greatly reduced by concomitant HP-TAZ, as the proportion of participants defined as nonresponders to their biologic decreased to 35% (7/20) with 8-week adjunctive application of once-daily HP-TAZ with biologic and further decreased to 23.5% (4/17) with another 4 weeks of adjunctive HP-TAZ applied every other day plus biologic. At week 16, after 4 weeks of biologics alone, the proportion was maintained at 33.3% (6/18).
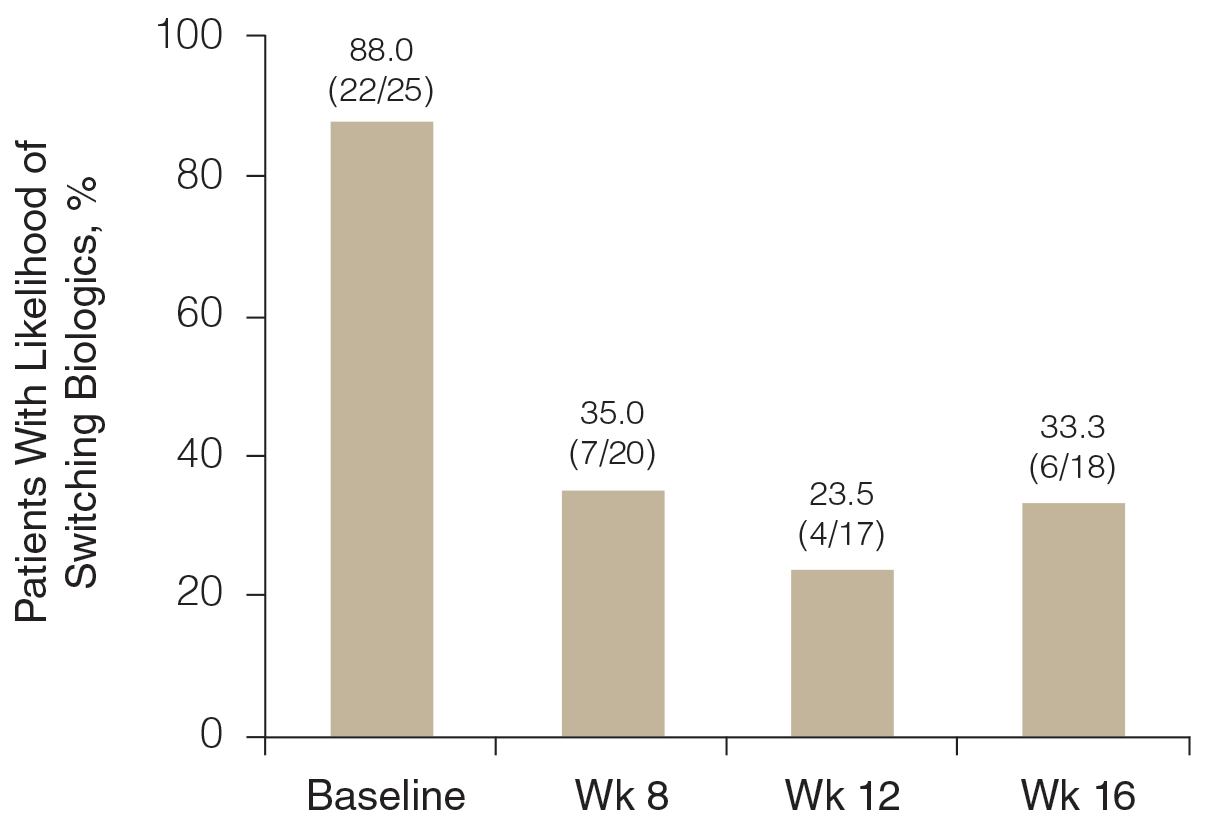
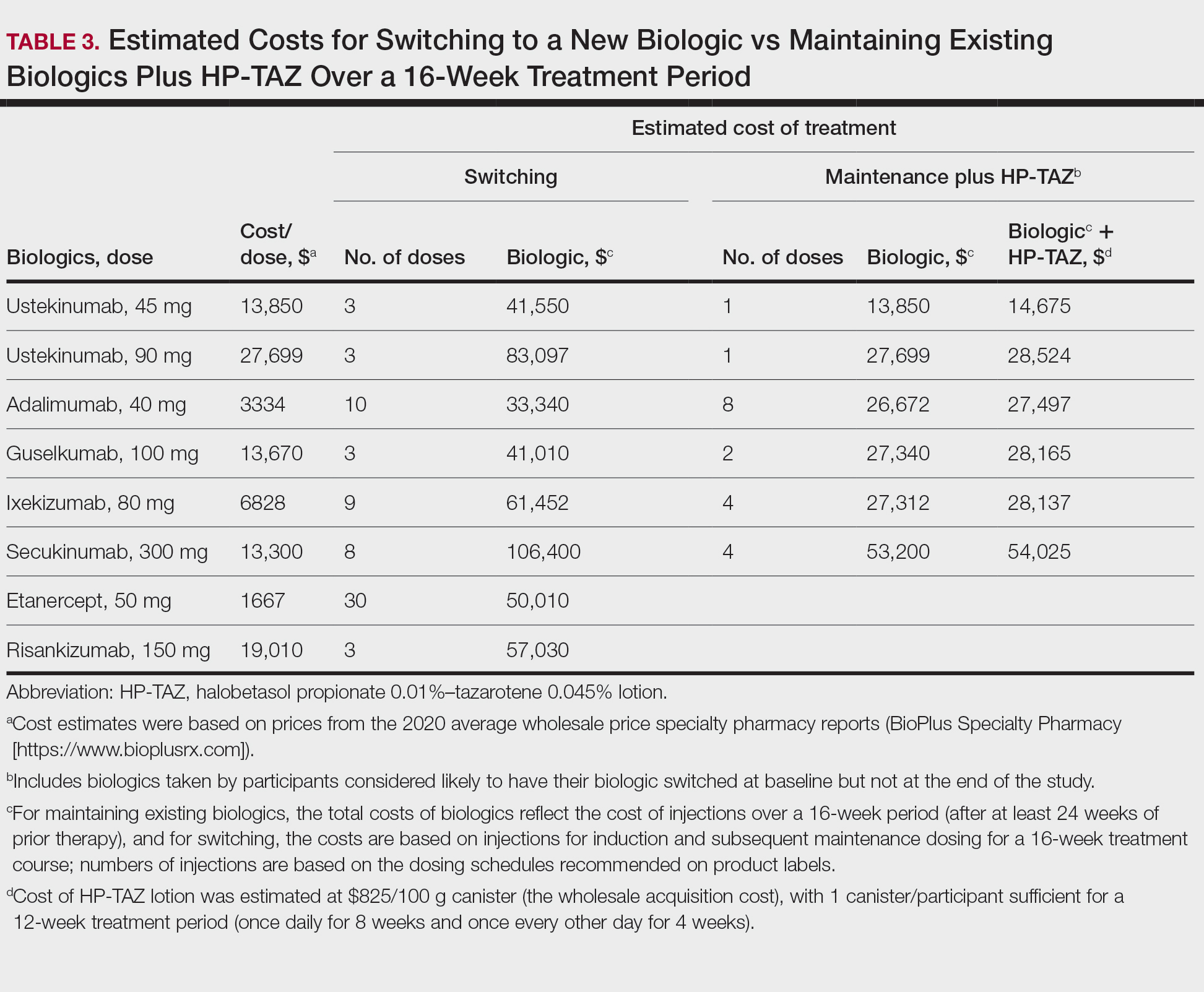
Pharmacoeconomics of Adding Topical HP-TAZ vs Switching Biologics—In the participants whom the investigator reported as likely to switch biologics at baseline, 9 had improvements in disease control such that switching biologics was no longer considered necessary for them at week 16. Potential cost savings with adjunctive therapy of HP-TAZ plus biologic vs switching biologics were therefore evaluated in these 9 participants, who were receiving ustekinumab, adalimumab, guselkumab, ixekizumab, and secukinumab during the study (Table 3). The estimated total cost of 16-week maintenance dosing of biologics plus adjunct HP-TAZ lotion ranged from $14,675 (ustekinumab 45 mg) to $54,025 (secukinumab 300 mg), while switching to other most commonly prescribed biologics for 16 weeks would cost an estimated $33,340 to $106,400 (induction and subsequent maintenance phases)(Table 3). Most biologic plus HP-TAZ combinations were estimated to cost less than $30,000, potentially saving $4816 to $91,725 compared with switching to any of the other 7 biologics (Table 3). The relatively more expensive maintenance combination containing secukinumab plus HP-TAZ ($54,025) appeared to be a less expensive option when compared with switching to ustekinumab (90 mg)($83,097), ixekizumab (80 mg)($61,452), or risankizumab (150 mg)($57,030) as an alternative biologic.
Comment
Adjunctive Use of HP-TAZ Lotion—In the present study, we showed that adjunctive HP-TAZ lotion improved biologic treatment response and reduced disease severity in participants with moderate to severe psoriasis whose symptoms could not be adequately controlled by 24 weeks or more of biologic monotherapy in a real-world setting. Disease activity decreased as evidenced by reductions in all assessed effectiveness variables, including BSA involvement, PGA score, composite BSA×PGA score, and participant-reported DLQI score. Half of the participants achieved NPF TTT status at the end of the study. The treatment was well tolerated with no unexpected safety concerns. Compared with switching to a new biologic, adding topical HP-TAZ to ongoing biologics appeared to be a more cost-effective approach to enhance treatment effects. Our results suggest that adjunctive use of HP-TAZ lotion may be a safe, effective, and economical option for patients with psoriasis who are failing their ongoing biologic monotherapy.
Treat-to-Target Status—The NPF-recommended target response to a treatment for plaque psoriasis is BSA of 1% or lower at 3 months postinitiation.10 Patients in the current study had major psoriasis activity at study entry despite being treated with a biologic for at least 24 weeks, as none had attained NPF TTT status at baseline. Because the time period of prior biologic treatment (at least 24 weeks) is much longer than the 3 months suggested by NPF, we believe that we were observing a true failure of the biologic rather than a slow onset of treatment effects in these patients at the time of enrollment. By week 12, with the addition of HP-TAZ lotion to the biologic, a high rate of participants achieved NPF TTT status (64.7%), with most participants being able to maintain this TTT status at study end after 4 weeks of biologic alone. Most participants also reported no impact of psoriasis on their QOL (DLQI, 0–116; 76.5%) at week 12. Improvements we found in disease control with adjunctive HP-TAZ lotion plus biologic support prior reports showing enhanced responses when a topical medication was added to a biologic.17,18 Reductions in psoriasis activity after 8 weeks of combined biologics plus once-daily HP-TAZ also are consistent with 2 phase 3 RCTs in which a monotherapy of HP-TAZ lotion used once daily for 8 weeks reduced BSA and DLQI.15 Notably, in the current study, disease severity continued to decrease when dosing of HP-TAZ was reduced to once every other day for 4 weeks, and the improvements were maintained even after the adjunct topical therapy was discontinued.
Safety Profile of HP-TAZ Lotion—The overall safety profile in our study also was consistent with that previously reported for HP-TAZ lotion,15,19-21 with no new safety signals observed. The combination treatment was well tolerated, with most reported AEs being mild in severity. Adverse events were mostly related to application-site reactions, the most common being dermatitis (28%), which was likely attributable to the TAZ component of the topical regimen.15
Likelihood of Switching Biologics—Reduced disease activity was reflected by a decrease in the percentage of participants the investigator considered likely to change biologics, which was 88.0% at baseline but only 33.3% at the end of the study. Although switching to a different biologic agent can improve treatment effect,22 it could lead to a substantial increase in health care costs and use of resources compared with no switch.5 We found that switching to one of the other most commonly prescribed biologics could incur $4816 to $91,725 in additional costs in most cases when compared with the combination strategy we evaluated over the 16-week treatment period. Therefore, concomitant use of HP-TAZ lotion with the ongoing biologics would be a potentially more economical alternative for patients to achieve acceptable responses or the NPF TTT goal. Moreover, combination with an adjunctive topical medication could avoid potential risks associated with switching, such as new AEs with new biologic regimens or disease flare during any washout period sometimes adopted for switching biologics.
Study Limitations—Our estimated costs were based on average wholesale prices and did not reflect net prices paid by patients or health plans due to the lack of known discount rates.
Conclusion
In this real-world study, patients with psoriasis that failed to respond to biologic monotherapy had improved disease control and QOL and reported no new safety concerns with adjunctive use of HP-TAZ lotion. Adding HP-TAZ to the ongoing biologics could be a more cost-effective option vs switching biologics for patients whose psoriasis symptoms could not be controlled with biologic monotherapy. Taken together, our results support the use of HP-TAZ lotion as an effective and safe adjunctive topical therapy in combination with biologics for psoriasis treatment.
Acknowledgments—We acknowledge the medical writing assistance provided by Hui Zhang, PhD, and Kathleen Ohleth, PhD, from Precise Publications LLC (Far Hills, New Jersey), which was funded by Ortho Dermatologics.
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516.
- Global Report on Psoriasis. World Health Organization; 2016. Accessed January 11, 2022. https://apps.who.int/iris/handle/10665/204417
- Takeshita J, Grewal S, Langan SM, et al. Psoriasis and comorbid diseases: epidemiology. J Am Acad Dermatol. 2017;76:377-390.
- Moller AH, Erntoft S, Vinding GR, et al. A systematic literature review to compare quality of life in psoriasis with other chronic diseases using EQ-5D-derived utility values. Patient Relat Outcome Meas. 2015;6:167-177.
- Feldman SR, Tian H, Wang X, et al. Health care utilization and cost associated with biologic treatment patterns among patients with moderate to severe psoriasis: analyses from a large U.S. claims database. J Manag Care Spec Pharm. 2019;25:479-488.
- Thomsen SF, Skov L, Dodge R, et al. Socioeconomic costs and health inequalities from psoriasis: a cohort study. Dermatology. 2019;235:372-379.
- Fowler JF, Duh MS, Rovba L, et al. The impact of psoriasis on health care costs and patient work loss. J Am Acad Dermatol. 2008;59:772-780.
- Menter A, Gottlieb A, Feldman SR, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 1. overview of psoriasis and guidelines of care for the treatment of psoriasis with biologics. J Am Acad Dermatol. 2008;58:826-850.
- Bagel J, Gold LS. Combining topical psoriasis treatment to enhance systemic and phototherapy: a review of the literature. J Drugs Dermatol. 2017;16:1209-1222.
- Armstrong AW, Siegel MP, Bagel J, et al. From the Medical Board of the National Psoriasis Foundation: treatment targets for plaque psoriasis. J Am Acad Dermatol. 2017;76:290-298.
- Armstrong AW, Bagel J, Van Voorhees AS, et al. Combining biologic therapies with other systemic treatments in psoriasis: evidence-based, best-practice recommendations from the Medical Board of the National Psoriasis Foundation. JAMA Dermatol. 2015;151:432-438.
- Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80:1029-1072.
- Duobrii. Prescribing information. Bausch Health Companies Inc; 2019.
- Sugarman JL, Weiss J, Tanghetti EA, et al. Safety and efficacy of a fixed combination halobetasol and tazarotene lotion in the treatment of moderate-to-severe plaque psoriasis: a pooled analysis of two phase 3 studies. J Drugs Dermatol. 2018;17:855-861.
- Gold LS, Lebwohl MG, Sugarman JL, et al. Safety and efficacy of a fixed combination of halobetasol and tazarotene in the treatment of moderate-to-severe plaque psoriasis: results of 2 phase 3 randomized controlled trials. J Am Acad Dermatol. 2018;79:287-293.
- Finlay AY. Current severe psoriasis and the rule of tens. Br J Dermatol. 2005;152:861-867.
- Campione E, Mazzotta A, Paterno EJ, et al. Effect of calcipotriol on etanercept partial responder psoriasis vulgaris and psoriatic arthritis patients. Acta Derm Venereol. 2009;89:288-291.
- Bagel J, Zapata J, Nelson E. A prospective, open-label study evaluating adjunctive calcipotriene 0.005%/betamethasone dipropionate 0.064% foam in psoriasis patients with inadequate response to biologic therapy. J Drugs Dermatol. 2018;17:845-850.
- Sugarman JL, Gold LS, Lebwohl MG, et al. A phase 2, multicenter, double-blind, randomized, vehicle controlled clinical study to assess the safety and efficacy of a halobetasol/tazarotene fixed combination in the treatment of plaque psoriasis. J Drugs Dermatol. 2017;16:197-204.
- Lebwohl MG, Sugarman JL, Gold LS, et al. Long-term safety results from a phase 3 open-label study of a fixed combination halobetasol propionate 0.01% and tazarotene 0.045% lotion in moderate-to-severe plaque psoriasis. J Am Acad Dermatol. 2019;80:282-285.
- Bhatia ND, Pariser DM, Kircik L, et al. Safety and efficacy of a halobetasol 0.01%/tazarotene 0.045% fixed combination lotion in the treatment of moderate-to-severe plaque psoriasis: a comparison with halobetasol propionate 0.05% cream. J Clin Aesthet Dermatol. 2018;11:15-19.
- Wang TS, Tsai TF. Biologics switch in psoriasis. Immunotherapy. 2019;11:531-541.
Psoriasis is a common chronic immunologic skin disease that affects approximately 7.4 million adults in the United States1 and more than 100 million individuals worldwide.2 Patients with psoriasis have a potentially heightened risk for cardiometabolic diseases, psychiatric disorders, and psoriatic arthritis,3 as well as impaired quality of life (QOL).4 Psoriasis also is associated with increased health care costs5 and may result in substantial socioeconomic repercussions for affected patients.6,7
Psoriasis treatments focus on relieving symptoms and improving patient QOL. Systemic therapy has been the mainstay of treatment for moderate to severe psoriasis.8 Although topical therapy usually is applied to treat mild symptoms, it also can be used as an adjunct to enhance efficacy of other treatment approaches.9 The National Psoriasis Foundation (NPF) recommends a treat-to-target (TTT) strategy for plaque psoriasis, the most common form of psoriasis, with a target response of attaining affected body surface area (BSA) of 1% or lower at 3 months after treatment initiation, allowing for regular assessments of treatment responses.10
Not all patients with moderate to severe psoriasis can achieve a satisfactory response with systemic biologic monotherapy.11 Switching to a new biologic improves responses in some but not all cases12 and could be associated with new safety issues and additional costs. Combinations of biologics with phototherapy, nonbiologic systemic agents, or topical medications were found to be more effective than biologics alone,9,11 though long-term safety studies are needed for biologics combined with other systemic inverventions.11
A lotion containing a fixed combination of halobetasol propionate (HP) 0.01%, a corticosteroid, and tazarotene (TAZ) 0.045%, a retinoid, is indicated for plaque psoriasis in adults.13 Two randomized, controlled, phase 3 trials demonstrated the rapid and sustained efficacy of HP-TAZ in treating moderate to severe plaque psoriasis without any safety concerns.14,15 However, combining HP-TAZ lotion with biologics has not been examined yet, to our knowledge.
This open-label study evaluated the effectiveness and safety of adjunctive HP-TAZ lotion in adult patients with moderate to severe plaque psoriasis who were being treated with biologics in a real-world setting. Potential cost savings with the addition of topical HP-TAZ to ongoing biologics vs switching to a new biologic also were assessed.
Methods
Study Design and Participants—A single-center, institutional review board–approved, open-label study evaluated adjunctive therapy with HP 0.01%–TAZ 0.045% lotion in patients with psoriasis being treated with biologic agents. The study was conducted in accordance with the ethical principles of the Declaration of Helsinki and in compliance with Good Clinical Practices. All patients provided written informed consent before enrollment.
Male and nonpregnant female patients (aged ≥18 years)with moderate to severe chronic plaque psoriasis and a BSA of 2% to 10% who were being treated with biologics for at least 24 weeks at baseline were enrolled. Patients were excluded if they had used oral systemic medications for psoriasis (≤4 weeks), other topical antipsoriatic therapies (≤14 days), UVB phototherapy (≤2 weeks), and psoralen plus UVA phototherapy (≤4 weeks) prior to study initiation. Concomitant use of steroid-free topical emollients or low-potency topical steroids and appropriate interventions deemed necessary by the investigator were allowed.
Although participants maintained their prescribed biologics for the duration of the study, HP-TAZ lotion also was applied once daily for 8 weeks, followed by once every other day for an additional 4 weeks. Participants then continued with biologics only for the last 4 weeks of the study.
Study Outcome Measures—Disease severity and treatment efficacy were assessed by affected BSA, Physician Global Assessment (PGA) score, composite BSA×PGA score, and participant-reported Dermatology Life Quality Index (DLQI). The primary end point was the proportion of participants achieving a BSA of 0% to 1% (NPF TTT status) at week 8. Secondary end points included the proportions of participants with BSA of 0% to 1% at weeks 12 and 16; BSA×PGA score at weeks 8, 12, and 16; and improvements in BSA, PGA, and DLQI at weeks 8, 12, and 16.
Adverse events (AEs) that occurred after the signing of the informed consent and for the duration of the participant’s participation were recorded, regardless of causality. Physical examinations were performed at screening; baseline; and weeks 8, 12, and 16 to document any clinically significant abnormalities. Localized skin reactions were assessed for tolerability of the study drug, with any reaction requiring concomitant therapy recorded as an AE.
The likelihood of switching to a new biologic regimen was assessed by the investigator for each participant at baseline and weeks 8, 12, and 16. Participants with unacceptable responses to their treatments (BSA >3%) were reported as likely to be considered for switching biologics by the investigator.
Pharmacoeconomic Evaluation—Potential cost savings were evaluated for the addition of HP-TAZ lotion to ongoing biologics vs switching to a new biologic. Cost comparisons were made in participants for whom the investigator would likely have switched biologics at baseline but not at the end of the study. For maintaining the same biologic with adjunctive topical HP-TAZ, total cost was estimated by adding the cost for 12 weeks (once daily for 8 weeks and once every other day for 4 weeks) of the HP-TAZ lotion to that of 16-week maintenance dosing with the biologic. The projected cost for switching to a new biologic for 16 weeks of treatment was based on both induction and maintenance dosing as recommended in its product label. Prices were obtained from the 2020 average wholesale price specialty pharmacy reports (BioPlus Specialty Pharmacy Services [https://www.bioplusrx.com]).
Data Handling—Enrollment of approximately 25 participants was desired for the study. Data on disease severity and participant-reported outcomes were assessed using descriptive statistics. Adverse events were summarized descriptively by incidence, severity, and relationship to the study drug. All participants with data available at a measured time point were included in the analyses for that time point.
Results
Participant Disposition and Demographics—Twenty-five participants (15 male and 10 female) were included in the study (Table 1). Seven participants discontinued the study for the following reasons: AEs (n=4), patient choice (n=2), and noncompliance (n=1).

The average age of the participants was 50 years, the majority were White (76.0% [19/25]) andnon-Hispanic (88.0% [22/25]), and the mean duration of chronic plaque psoriasis was 18.9 years (Table 1). Participants had been receiving biologic monotherapy for at least 24 weeks prior to enrollment, most commonly ustekinumab (32.0% [8/25])(Table 1). None had achieved the NPF TTT status with their biologics. At baseline, mean (SD) affected BSA, PGA, BSA×PGA, and participant-reported DLQI were 4.16% (2.04%), 2.84 (0.55), 11.88 (6.39), and 4.00 (4.74), respectively.
Efficacy Assessment—Application of HP-TAZ lotion in addition to the participants’ existing biologic therapy reduced severity of the disease, as evidenced by the reductions in mean BSA, PGA, and BSA×PGA. After 8 weeks of once-daily concomitant HP-TAZ use with biologic, mean BSA and PGA dropped by approximately 40% and 37%, respectively (Figures 1A and 1B). A greater reduction (54%) was found for mean BSA×PGA (Figure 1C). Disease severity continued to improve when the application schedule for HP-TAZ was changed to once every other day for 4 weeks, as mean BSA, PGA, and BSA×PGA decreased further at week 12. These beneficial effects were sustained during the last 4 weeks of the study after HP-TAZ was discontinued, with reductions of 57%, 43%, and 70% from baseline for mean BSA, PGA, and BSA×PGA, respectively (Figure 1).

The proportion of participants who achieved NPF TTT status increased from 0% at baseline to 20.0% (5/20) at week 8 with once-daily use of HP-TAZ plus biologic for 8 weeks (Figure 2). At week 12, more participants (64.7% [11/17]) achieved the treatment goal after application of HP-TAZ once every other day with biologic for 4 weeks. Most participants maintained NPF TTT status after HP-TAZ was discontinued; at week 16, 50.0% (9/18) attained the NPF treatment goal (Figure 2).
![Proportion of participants achieving National Psoriasis Foundation target-to-treat status (body surface area [BSA] ≤1%) at baseline and weeks 8, 12, and 16 Proportion of participants achieving National Psoriasis Foundation target-to-treat status (body surface area [BSA] ≤1%) at baseline and weeks 8, 12, and 16](https://cdn.mdedge.com/files/s3fs-public/Bagel_0222_2.JPG)
The mean DLQI score decreased from 4.00 at baseline to 2.45 after 8 weeks of concomitant use of once-daily HP-TAZ with biologic, reflecting a 39% score reduction. An additional 4 weeks of adjunctive HP-TAZ applied once every other day with biologic further decreased the DLQI score to 2.18 at week 12. Mean DLQI remained similar (2.33) after another 4 weeks of biologics alone. The proportion of participants reporting a DLQI score of 0 to 1 increased from 40% (10/25) at baseline to 60% (12/20) at week 8 and 76.5% (13/17) at week 12 with adjunctive HP-TAZ lotion use with biologic. At week 16, a DLQI score of 0 to 1 was reported in 61.1% (11/18) of participants after receiving only biologics for 4 weeks.
Safety Assessment—A total of 19 AEs were reported in 11 participants during the study; 16 AEs were considered treatment related in 8 participants (Table 2). The most common AEs were retinoid dermatitis (28% [7/25]), burning at the application site (8% [2/25]), and pruritus at the application site (8% [2/25]), all of which were considered related to the treatment. Among all AEs, 12 were mild in severity, and the remaining 7 were moderate. Adverse events led to early study termination in 4 participants, all with retinoid dermatitis as the primary reason.

Likelihood of Switching Biologics—At baseline, almost 90% (22/25) of participants were rated as likely to switch biologics by the investigator due to unacceptable responses to their currently prescribed biologics (BSA >3%)(Figure 3). The likelihood was greatly reduced by concomitant HP-TAZ, as the proportion of participants defined as nonresponders to their biologic decreased to 35% (7/20) with 8-week adjunctive application of once-daily HP-TAZ with biologic and further decreased to 23.5% (4/17) with another 4 weeks of adjunctive HP-TAZ applied every other day plus biologic. At week 16, after 4 weeks of biologics alone, the proportion was maintained at 33.3% (6/18).

Pharmacoeconomics of Adding Topical HP-TAZ vs Switching Biologics—In the participants whom the investigator reported as likely to switch biologics at baseline, 9 had improvements in disease control such that switching biologics was no longer considered necessary for them at week 16. Potential cost savings with adjunctive therapy of HP-TAZ plus biologic vs switching biologics were therefore evaluated in these 9 participants, who were receiving ustekinumab, adalimumab, guselkumab, ixekizumab, and secukinumab during the study (Table 3). The estimated total cost of 16-week maintenance dosing of biologics plus adjunct HP-TAZ lotion ranged from $14,675 (ustekinumab 45 mg) to $54,025 (secukinumab 300 mg), while switching to other most commonly prescribed biologics for 16 weeks would cost an estimated $33,340 to $106,400 (induction and subsequent maintenance phases)(Table 3). Most biologic plus HP-TAZ combinations were estimated to cost less than $30,000, potentially saving $4816 to $91,725 compared with switching to any of the other 7 biologics (Table 3). The relatively more expensive maintenance combination containing secukinumab plus HP-TAZ ($54,025) appeared to be a less expensive option when compared with switching to ustekinumab (90 mg)($83,097), ixekizumab (80 mg)($61,452), or risankizumab (150 mg)($57,030) as an alternative biologic.

Comment
Adjunctive Use of HP-TAZ Lotion—In the present study, we showed that adjunctive HP-TAZ lotion improved biologic treatment response and reduced disease severity in participants with moderate to severe psoriasis whose symptoms could not be adequately controlled by 24 weeks or more of biologic monotherapy in a real-world setting. Disease activity decreased as evidenced by reductions in all assessed effectiveness variables, including BSA involvement, PGA score, composite BSA×PGA score, and participant-reported DLQI score. Half of the participants achieved NPF TTT status at the end of the study. The treatment was well tolerated with no unexpected safety concerns. Compared with switching to a new biologic, adding topical HP-TAZ to ongoing biologics appeared to be a more cost-effective approach to enhance treatment effects. Our results suggest that adjunctive use of HP-TAZ lotion may be a safe, effective, and economical option for patients with psoriasis who are failing their ongoing biologic monotherapy.
Treat-to-Target Status—The NPF-recommended target response to a treatment for plaque psoriasis is BSA of 1% or lower at 3 months postinitiation.10 Patients in the current study had major psoriasis activity at study entry despite being treated with a biologic for at least 24 weeks, as none had attained NPF TTT status at baseline. Because the time period of prior biologic treatment (at least 24 weeks) is much longer than the 3 months suggested by NPF, we believe that we were observing a true failure of the biologic rather than a slow onset of treatment effects in these patients at the time of enrollment. By week 12, with the addition of HP-TAZ lotion to the biologic, a high rate of participants achieved NPF TTT status (64.7%), with most participants being able to maintain this TTT status at study end after 4 weeks of biologic alone. Most participants also reported no impact of psoriasis on their QOL (DLQI, 0–116; 76.5%) at week 12. Improvements we found in disease control with adjunctive HP-TAZ lotion plus biologic support prior reports showing enhanced responses when a topical medication was added to a biologic.17,18 Reductions in psoriasis activity after 8 weeks of combined biologics plus once-daily HP-TAZ also are consistent with 2 phase 3 RCTs in which a monotherapy of HP-TAZ lotion used once daily for 8 weeks reduced BSA and DLQI.15 Notably, in the current study, disease severity continued to decrease when dosing of HP-TAZ was reduced to once every other day for 4 weeks, and the improvements were maintained even after the adjunct topical therapy was discontinued.
Safety Profile of HP-TAZ Lotion—The overall safety profile in our study also was consistent with that previously reported for HP-TAZ lotion,15,19-21 with no new safety signals observed. The combination treatment was well tolerated, with most reported AEs being mild in severity. Adverse events were mostly related to application-site reactions, the most common being dermatitis (28%), which was likely attributable to the TAZ component of the topical regimen.15
Likelihood of Switching Biologics—Reduced disease activity was reflected by a decrease in the percentage of participants the investigator considered likely to change biologics, which was 88.0% at baseline but only 33.3% at the end of the study. Although switching to a different biologic agent can improve treatment effect,22 it could lead to a substantial increase in health care costs and use of resources compared with no switch.5 We found that switching to one of the other most commonly prescribed biologics could incur $4816 to $91,725 in additional costs in most cases when compared with the combination strategy we evaluated over the 16-week treatment period. Therefore, concomitant use of HP-TAZ lotion with the ongoing biologics would be a potentially more economical alternative for patients to achieve acceptable responses or the NPF TTT goal. Moreover, combination with an adjunctive topical medication could avoid potential risks associated with switching, such as new AEs with new biologic regimens or disease flare during any washout period sometimes adopted for switching biologics.
Study Limitations—Our estimated costs were based on average wholesale prices and did not reflect net prices paid by patients or health plans due to the lack of known discount rates.
Conclusion
In this real-world study, patients with psoriasis that failed to respond to biologic monotherapy had improved disease control and QOL and reported no new safety concerns with adjunctive use of HP-TAZ lotion. Adding HP-TAZ to the ongoing biologics could be a more cost-effective option vs switching biologics for patients whose psoriasis symptoms could not be controlled with biologic monotherapy. Taken together, our results support the use of HP-TAZ lotion as an effective and safe adjunctive topical therapy in combination with biologics for psoriasis treatment.
Acknowledgments—We acknowledge the medical writing assistance provided by Hui Zhang, PhD, and Kathleen Ohleth, PhD, from Precise Publications LLC (Far Hills, New Jersey), which was funded by Ortho Dermatologics.
Psoriasis is a common chronic immunologic skin disease that affects approximately 7.4 million adults in the United States1 and more than 100 million individuals worldwide.2 Patients with psoriasis have a potentially heightened risk for cardiometabolic diseases, psychiatric disorders, and psoriatic arthritis,3 as well as impaired quality of life (QOL).4 Psoriasis also is associated with increased health care costs5 and may result in substantial socioeconomic repercussions for affected patients.6,7
Psoriasis treatments focus on relieving symptoms and improving patient QOL. Systemic therapy has been the mainstay of treatment for moderate to severe psoriasis.8 Although topical therapy usually is applied to treat mild symptoms, it also can be used as an adjunct to enhance efficacy of other treatment approaches.9 The National Psoriasis Foundation (NPF) recommends a treat-to-target (TTT) strategy for plaque psoriasis, the most common form of psoriasis, with a target response of attaining affected body surface area (BSA) of 1% or lower at 3 months after treatment initiation, allowing for regular assessments of treatment responses.10
Not all patients with moderate to severe psoriasis can achieve a satisfactory response with systemic biologic monotherapy.11 Switching to a new biologic improves responses in some but not all cases12 and could be associated with new safety issues and additional costs. Combinations of biologics with phototherapy, nonbiologic systemic agents, or topical medications were found to be more effective than biologics alone,9,11 though long-term safety studies are needed for biologics combined with other systemic inverventions.11
A lotion containing a fixed combination of halobetasol propionate (HP) 0.01%, a corticosteroid, and tazarotene (TAZ) 0.045%, a retinoid, is indicated for plaque psoriasis in adults.13 Two randomized, controlled, phase 3 trials demonstrated the rapid and sustained efficacy of HP-TAZ in treating moderate to severe plaque psoriasis without any safety concerns.14,15 However, combining HP-TAZ lotion with biologics has not been examined yet, to our knowledge.
This open-label study evaluated the effectiveness and safety of adjunctive HP-TAZ lotion in adult patients with moderate to severe plaque psoriasis who were being treated with biologics in a real-world setting. Potential cost savings with the addition of topical HP-TAZ to ongoing biologics vs switching to a new biologic also were assessed.
Methods
Study Design and Participants—A single-center, institutional review board–approved, open-label study evaluated adjunctive therapy with HP 0.01%–TAZ 0.045% lotion in patients with psoriasis being treated with biologic agents. The study was conducted in accordance with the ethical principles of the Declaration of Helsinki and in compliance with Good Clinical Practices. All patients provided written informed consent before enrollment.
Male and nonpregnant female patients (aged ≥18 years)with moderate to severe chronic plaque psoriasis and a BSA of 2% to 10% who were being treated with biologics for at least 24 weeks at baseline were enrolled. Patients were excluded if they had used oral systemic medications for psoriasis (≤4 weeks), other topical antipsoriatic therapies (≤14 days), UVB phototherapy (≤2 weeks), and psoralen plus UVA phototherapy (≤4 weeks) prior to study initiation. Concomitant use of steroid-free topical emollients or low-potency topical steroids and appropriate interventions deemed necessary by the investigator were allowed.
Although participants maintained their prescribed biologics for the duration of the study, HP-TAZ lotion also was applied once daily for 8 weeks, followed by once every other day for an additional 4 weeks. Participants then continued with biologics only for the last 4 weeks of the study.
Study Outcome Measures—Disease severity and treatment efficacy were assessed by affected BSA, Physician Global Assessment (PGA) score, composite BSA×PGA score, and participant-reported Dermatology Life Quality Index (DLQI). The primary end point was the proportion of participants achieving a BSA of 0% to 1% (NPF TTT status) at week 8. Secondary end points included the proportions of participants with BSA of 0% to 1% at weeks 12 and 16; BSA×PGA score at weeks 8, 12, and 16; and improvements in BSA, PGA, and DLQI at weeks 8, 12, and 16.
Adverse events (AEs) that occurred after the signing of the informed consent and for the duration of the participant’s participation were recorded, regardless of causality. Physical examinations were performed at screening; baseline; and weeks 8, 12, and 16 to document any clinically significant abnormalities. Localized skin reactions were assessed for tolerability of the study drug, with any reaction requiring concomitant therapy recorded as an AE.
The likelihood of switching to a new biologic regimen was assessed by the investigator for each participant at baseline and weeks 8, 12, and 16. Participants with unacceptable responses to their treatments (BSA >3%) were reported as likely to be considered for switching biologics by the investigator.
Pharmacoeconomic Evaluation—Potential cost savings were evaluated for the addition of HP-TAZ lotion to ongoing biologics vs switching to a new biologic. Cost comparisons were made in participants for whom the investigator would likely have switched biologics at baseline but not at the end of the study. For maintaining the same biologic with adjunctive topical HP-TAZ, total cost was estimated by adding the cost for 12 weeks (once daily for 8 weeks and once every other day for 4 weeks) of the HP-TAZ lotion to that of 16-week maintenance dosing with the biologic. The projected cost for switching to a new biologic for 16 weeks of treatment was based on both induction and maintenance dosing as recommended in its product label. Prices were obtained from the 2020 average wholesale price specialty pharmacy reports (BioPlus Specialty Pharmacy Services [https://www.bioplusrx.com]).
Data Handling—Enrollment of approximately 25 participants was desired for the study. Data on disease severity and participant-reported outcomes were assessed using descriptive statistics. Adverse events were summarized descriptively by incidence, severity, and relationship to the study drug. All participants with data available at a measured time point were included in the analyses for that time point.
Results
Participant Disposition and Demographics—Twenty-five participants (15 male and 10 female) were included in the study (Table 1). Seven participants discontinued the study for the following reasons: AEs (n=4), patient choice (n=2), and noncompliance (n=1).

The average age of the participants was 50 years, the majority were White (76.0% [19/25]) andnon-Hispanic (88.0% [22/25]), and the mean duration of chronic plaque psoriasis was 18.9 years (Table 1). Participants had been receiving biologic monotherapy for at least 24 weeks prior to enrollment, most commonly ustekinumab (32.0% [8/25])(Table 1). None had achieved the NPF TTT status with their biologics. At baseline, mean (SD) affected BSA, PGA, BSA×PGA, and participant-reported DLQI were 4.16% (2.04%), 2.84 (0.55), 11.88 (6.39), and 4.00 (4.74), respectively.
Efficacy Assessment—Application of HP-TAZ lotion in addition to the participants’ existing biologic therapy reduced severity of the disease, as evidenced by the reductions in mean BSA, PGA, and BSA×PGA. After 8 weeks of once-daily concomitant HP-TAZ use with biologic, mean BSA and PGA dropped by approximately 40% and 37%, respectively (Figures 1A and 1B). A greater reduction (54%) was found for mean BSA×PGA (Figure 1C). Disease severity continued to improve when the application schedule for HP-TAZ was changed to once every other day for 4 weeks, as mean BSA, PGA, and BSA×PGA decreased further at week 12. These beneficial effects were sustained during the last 4 weeks of the study after HP-TAZ was discontinued, with reductions of 57%, 43%, and 70% from baseline for mean BSA, PGA, and BSA×PGA, respectively (Figure 1).

The proportion of participants who achieved NPF TTT status increased from 0% at baseline to 20.0% (5/20) at week 8 with once-daily use of HP-TAZ plus biologic for 8 weeks (Figure 2). At week 12, more participants (64.7% [11/17]) achieved the treatment goal after application of HP-TAZ once every other day with biologic for 4 weeks. Most participants maintained NPF TTT status after HP-TAZ was discontinued; at week 16, 50.0% (9/18) attained the NPF treatment goal (Figure 2).
![Proportion of participants achieving National Psoriasis Foundation target-to-treat status (body surface area [BSA] ≤1%) at baseline and weeks 8, 12, and 16 Proportion of participants achieving National Psoriasis Foundation target-to-treat status (body surface area [BSA] ≤1%) at baseline and weeks 8, 12, and 16](https://cdn.mdedge.com/files/s3fs-public/Bagel_0222_2.JPG)
The mean DLQI score decreased from 4.00 at baseline to 2.45 after 8 weeks of concomitant use of once-daily HP-TAZ with biologic, reflecting a 39% score reduction. An additional 4 weeks of adjunctive HP-TAZ applied once every other day with biologic further decreased the DLQI score to 2.18 at week 12. Mean DLQI remained similar (2.33) after another 4 weeks of biologics alone. The proportion of participants reporting a DLQI score of 0 to 1 increased from 40% (10/25) at baseline to 60% (12/20) at week 8 and 76.5% (13/17) at week 12 with adjunctive HP-TAZ lotion use with biologic. At week 16, a DLQI score of 0 to 1 was reported in 61.1% (11/18) of participants after receiving only biologics for 4 weeks.
Safety Assessment—A total of 19 AEs were reported in 11 participants during the study; 16 AEs were considered treatment related in 8 participants (Table 2). The most common AEs were retinoid dermatitis (28% [7/25]), burning at the application site (8% [2/25]), and pruritus at the application site (8% [2/25]), all of which were considered related to the treatment. Among all AEs, 12 were mild in severity, and the remaining 7 were moderate. Adverse events led to early study termination in 4 participants, all with retinoid dermatitis as the primary reason.

Likelihood of Switching Biologics—At baseline, almost 90% (22/25) of participants were rated as likely to switch biologics by the investigator due to unacceptable responses to their currently prescribed biologics (BSA >3%)(Figure 3). The likelihood was greatly reduced by concomitant HP-TAZ, as the proportion of participants defined as nonresponders to their biologic decreased to 35% (7/20) with 8-week adjunctive application of once-daily HP-TAZ with biologic and further decreased to 23.5% (4/17) with another 4 weeks of adjunctive HP-TAZ applied every other day plus biologic. At week 16, after 4 weeks of biologics alone, the proportion was maintained at 33.3% (6/18).

Pharmacoeconomics of Adding Topical HP-TAZ vs Switching Biologics—In the participants whom the investigator reported as likely to switch biologics at baseline, 9 had improvements in disease control such that switching biologics was no longer considered necessary for them at week 16. Potential cost savings with adjunctive therapy of HP-TAZ plus biologic vs switching biologics were therefore evaluated in these 9 participants, who were receiving ustekinumab, adalimumab, guselkumab, ixekizumab, and secukinumab during the study (Table 3). The estimated total cost of 16-week maintenance dosing of biologics plus adjunct HP-TAZ lotion ranged from $14,675 (ustekinumab 45 mg) to $54,025 (secukinumab 300 mg), while switching to other most commonly prescribed biologics for 16 weeks would cost an estimated $33,340 to $106,400 (induction and subsequent maintenance phases)(Table 3). Most biologic plus HP-TAZ combinations were estimated to cost less than $30,000, potentially saving $4816 to $91,725 compared with switching to any of the other 7 biologics (Table 3). The relatively more expensive maintenance combination containing secukinumab plus HP-TAZ ($54,025) appeared to be a less expensive option when compared with switching to ustekinumab (90 mg)($83,097), ixekizumab (80 mg)($61,452), or risankizumab (150 mg)($57,030) as an alternative biologic.

Comment
Adjunctive Use of HP-TAZ Lotion—In the present study, we showed that adjunctive HP-TAZ lotion improved biologic treatment response and reduced disease severity in participants with moderate to severe psoriasis whose symptoms could not be adequately controlled by 24 weeks or more of biologic monotherapy in a real-world setting. Disease activity decreased as evidenced by reductions in all assessed effectiveness variables, including BSA involvement, PGA score, composite BSA×PGA score, and participant-reported DLQI score. Half of the participants achieved NPF TTT status at the end of the study. The treatment was well tolerated with no unexpected safety concerns. Compared with switching to a new biologic, adding topical HP-TAZ to ongoing biologics appeared to be a more cost-effective approach to enhance treatment effects. Our results suggest that adjunctive use of HP-TAZ lotion may be a safe, effective, and economical option for patients with psoriasis who are failing their ongoing biologic monotherapy.
Treat-to-Target Status—The NPF-recommended target response to a treatment for plaque psoriasis is BSA of 1% or lower at 3 months postinitiation.10 Patients in the current study had major psoriasis activity at study entry despite being treated with a biologic for at least 24 weeks, as none had attained NPF TTT status at baseline. Because the time period of prior biologic treatment (at least 24 weeks) is much longer than the 3 months suggested by NPF, we believe that we were observing a true failure of the biologic rather than a slow onset of treatment effects in these patients at the time of enrollment. By week 12, with the addition of HP-TAZ lotion to the biologic, a high rate of participants achieved NPF TTT status (64.7%), with most participants being able to maintain this TTT status at study end after 4 weeks of biologic alone. Most participants also reported no impact of psoriasis on their QOL (DLQI, 0–116; 76.5%) at week 12. Improvements we found in disease control with adjunctive HP-TAZ lotion plus biologic support prior reports showing enhanced responses when a topical medication was added to a biologic.17,18 Reductions in psoriasis activity after 8 weeks of combined biologics plus once-daily HP-TAZ also are consistent with 2 phase 3 RCTs in which a monotherapy of HP-TAZ lotion used once daily for 8 weeks reduced BSA and DLQI.15 Notably, in the current study, disease severity continued to decrease when dosing of HP-TAZ was reduced to once every other day for 4 weeks, and the improvements were maintained even after the adjunct topical therapy was discontinued.
Safety Profile of HP-TAZ Lotion—The overall safety profile in our study also was consistent with that previously reported for HP-TAZ lotion,15,19-21 with no new safety signals observed. The combination treatment was well tolerated, with most reported AEs being mild in severity. Adverse events were mostly related to application-site reactions, the most common being dermatitis (28%), which was likely attributable to the TAZ component of the topical regimen.15
Likelihood of Switching Biologics—Reduced disease activity was reflected by a decrease in the percentage of participants the investigator considered likely to change biologics, which was 88.0% at baseline but only 33.3% at the end of the study. Although switching to a different biologic agent can improve treatment effect,22 it could lead to a substantial increase in health care costs and use of resources compared with no switch.5 We found that switching to one of the other most commonly prescribed biologics could incur $4816 to $91,725 in additional costs in most cases when compared with the combination strategy we evaluated over the 16-week treatment period. Therefore, concomitant use of HP-TAZ lotion with the ongoing biologics would be a potentially more economical alternative for patients to achieve acceptable responses or the NPF TTT goal. Moreover, combination with an adjunctive topical medication could avoid potential risks associated with switching, such as new AEs with new biologic regimens or disease flare during any washout period sometimes adopted for switching biologics.
Study Limitations—Our estimated costs were based on average wholesale prices and did not reflect net prices paid by patients or health plans due to the lack of known discount rates.
Conclusion
In this real-world study, patients with psoriasis that failed to respond to biologic monotherapy had improved disease control and QOL and reported no new safety concerns with adjunctive use of HP-TAZ lotion. Adding HP-TAZ to the ongoing biologics could be a more cost-effective option vs switching biologics for patients whose psoriasis symptoms could not be controlled with biologic monotherapy. Taken together, our results support the use of HP-TAZ lotion as an effective and safe adjunctive topical therapy in combination with biologics for psoriasis treatment.
Acknowledgments—We acknowledge the medical writing assistance provided by Hui Zhang, PhD, and Kathleen Ohleth, PhD, from Precise Publications LLC (Far Hills, New Jersey), which was funded by Ortho Dermatologics.
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516.
- Global Report on Psoriasis. World Health Organization; 2016. Accessed January 11, 2022. https://apps.who.int/iris/handle/10665/204417
- Takeshita J, Grewal S, Langan SM, et al. Psoriasis and comorbid diseases: epidemiology. J Am Acad Dermatol. 2017;76:377-390.
- Moller AH, Erntoft S, Vinding GR, et al. A systematic literature review to compare quality of life in psoriasis with other chronic diseases using EQ-5D-derived utility values. Patient Relat Outcome Meas. 2015;6:167-177.
- Feldman SR, Tian H, Wang X, et al. Health care utilization and cost associated with biologic treatment patterns among patients with moderate to severe psoriasis: analyses from a large U.S. claims database. J Manag Care Spec Pharm. 2019;25:479-488.
- Thomsen SF, Skov L, Dodge R, et al. Socioeconomic costs and health inequalities from psoriasis: a cohort study. Dermatology. 2019;235:372-379.
- Fowler JF, Duh MS, Rovba L, et al. The impact of psoriasis on health care costs and patient work loss. J Am Acad Dermatol. 2008;59:772-780.
- Menter A, Gottlieb A, Feldman SR, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 1. overview of psoriasis and guidelines of care for the treatment of psoriasis with biologics. J Am Acad Dermatol. 2008;58:826-850.
- Bagel J, Gold LS. Combining topical psoriasis treatment to enhance systemic and phototherapy: a review of the literature. J Drugs Dermatol. 2017;16:1209-1222.
- Armstrong AW, Siegel MP, Bagel J, et al. From the Medical Board of the National Psoriasis Foundation: treatment targets for plaque psoriasis. J Am Acad Dermatol. 2017;76:290-298.
- Armstrong AW, Bagel J, Van Voorhees AS, et al. Combining biologic therapies with other systemic treatments in psoriasis: evidence-based, best-practice recommendations from the Medical Board of the National Psoriasis Foundation. JAMA Dermatol. 2015;151:432-438.
- Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80:1029-1072.
- Duobrii. Prescribing information. Bausch Health Companies Inc; 2019.
- Sugarman JL, Weiss J, Tanghetti EA, et al. Safety and efficacy of a fixed combination halobetasol and tazarotene lotion in the treatment of moderate-to-severe plaque psoriasis: a pooled analysis of two phase 3 studies. J Drugs Dermatol. 2018;17:855-861.
- Gold LS, Lebwohl MG, Sugarman JL, et al. Safety and efficacy of a fixed combination of halobetasol and tazarotene in the treatment of moderate-to-severe plaque psoriasis: results of 2 phase 3 randomized controlled trials. J Am Acad Dermatol. 2018;79:287-293.
- Finlay AY. Current severe psoriasis and the rule of tens. Br J Dermatol. 2005;152:861-867.
- Campione E, Mazzotta A, Paterno EJ, et al. Effect of calcipotriol on etanercept partial responder psoriasis vulgaris and psoriatic arthritis patients. Acta Derm Venereol. 2009;89:288-291.
- Bagel J, Zapata J, Nelson E. A prospective, open-label study evaluating adjunctive calcipotriene 0.005%/betamethasone dipropionate 0.064% foam in psoriasis patients with inadequate response to biologic therapy. J Drugs Dermatol. 2018;17:845-850.
- Sugarman JL, Gold LS, Lebwohl MG, et al. A phase 2, multicenter, double-blind, randomized, vehicle controlled clinical study to assess the safety and efficacy of a halobetasol/tazarotene fixed combination in the treatment of plaque psoriasis. J Drugs Dermatol. 2017;16:197-204.
- Lebwohl MG, Sugarman JL, Gold LS, et al. Long-term safety results from a phase 3 open-label study of a fixed combination halobetasol propionate 0.01% and tazarotene 0.045% lotion in moderate-to-severe plaque psoriasis. J Am Acad Dermatol. 2019;80:282-285.
- Bhatia ND, Pariser DM, Kircik L, et al. Safety and efficacy of a halobetasol 0.01%/tazarotene 0.045% fixed combination lotion in the treatment of moderate-to-severe plaque psoriasis: a comparison with halobetasol propionate 0.05% cream. J Clin Aesthet Dermatol. 2018;11:15-19.
- Wang TS, Tsai TF. Biologics switch in psoriasis. Immunotherapy. 2019;11:531-541.
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516.
- Global Report on Psoriasis. World Health Organization; 2016. Accessed January 11, 2022. https://apps.who.int/iris/handle/10665/204417
- Takeshita J, Grewal S, Langan SM, et al. Psoriasis and comorbid diseases: epidemiology. J Am Acad Dermatol. 2017;76:377-390.
- Moller AH, Erntoft S, Vinding GR, et al. A systematic literature review to compare quality of life in psoriasis with other chronic diseases using EQ-5D-derived utility values. Patient Relat Outcome Meas. 2015;6:167-177.
- Feldman SR, Tian H, Wang X, et al. Health care utilization and cost associated with biologic treatment patterns among patients with moderate to severe psoriasis: analyses from a large U.S. claims database. J Manag Care Spec Pharm. 2019;25:479-488.
- Thomsen SF, Skov L, Dodge R, et al. Socioeconomic costs and health inequalities from psoriasis: a cohort study. Dermatology. 2019;235:372-379.
- Fowler JF, Duh MS, Rovba L, et al. The impact of psoriasis on health care costs and patient work loss. J Am Acad Dermatol. 2008;59:772-780.
- Menter A, Gottlieb A, Feldman SR, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 1. overview of psoriasis and guidelines of care for the treatment of psoriasis with biologics. J Am Acad Dermatol. 2008;58:826-850.
- Bagel J, Gold LS. Combining topical psoriasis treatment to enhance systemic and phototherapy: a review of the literature. J Drugs Dermatol. 2017;16:1209-1222.
- Armstrong AW, Siegel MP, Bagel J, et al. From the Medical Board of the National Psoriasis Foundation: treatment targets for plaque psoriasis. J Am Acad Dermatol. 2017;76:290-298.
- Armstrong AW, Bagel J, Van Voorhees AS, et al. Combining biologic therapies with other systemic treatments in psoriasis: evidence-based, best-practice recommendations from the Medical Board of the National Psoriasis Foundation. JAMA Dermatol. 2015;151:432-438.
- Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80:1029-1072.
- Duobrii. Prescribing information. Bausch Health Companies Inc; 2019.
- Sugarman JL, Weiss J, Tanghetti EA, et al. Safety and efficacy of a fixed combination halobetasol and tazarotene lotion in the treatment of moderate-to-severe plaque psoriasis: a pooled analysis of two phase 3 studies. J Drugs Dermatol. 2018;17:855-861.
- Gold LS, Lebwohl MG, Sugarman JL, et al. Safety and efficacy of a fixed combination of halobetasol and tazarotene in the treatment of moderate-to-severe plaque psoriasis: results of 2 phase 3 randomized controlled trials. J Am Acad Dermatol. 2018;79:287-293.
- Finlay AY. Current severe psoriasis and the rule of tens. Br J Dermatol. 2005;152:861-867.
- Campione E, Mazzotta A, Paterno EJ, et al. Effect of calcipotriol on etanercept partial responder psoriasis vulgaris and psoriatic arthritis patients. Acta Derm Venereol. 2009;89:288-291.
- Bagel J, Zapata J, Nelson E. A prospective, open-label study evaluating adjunctive calcipotriene 0.005%/betamethasone dipropionate 0.064% foam in psoriasis patients with inadequate response to biologic therapy. J Drugs Dermatol. 2018;17:845-850.
- Sugarman JL, Gold LS, Lebwohl MG, et al. A phase 2, multicenter, double-blind, randomized, vehicle controlled clinical study to assess the safety and efficacy of a halobetasol/tazarotene fixed combination in the treatment of plaque psoriasis. J Drugs Dermatol. 2017;16:197-204.
- Lebwohl MG, Sugarman JL, Gold LS, et al. Long-term safety results from a phase 3 open-label study of a fixed combination halobetasol propionate 0.01% and tazarotene 0.045% lotion in moderate-to-severe plaque psoriasis. J Am Acad Dermatol. 2019;80:282-285.
- Bhatia ND, Pariser DM, Kircik L, et al. Safety and efficacy of a halobetasol 0.01%/tazarotene 0.045% fixed combination lotion in the treatment of moderate-to-severe plaque psoriasis: a comparison with halobetasol propionate 0.05% cream. J Clin Aesthet Dermatol. 2018;11:15-19.
- Wang TS, Tsai TF. Biologics switch in psoriasis. Immunotherapy. 2019;11:531-541.
Practice Points
- Although monotherapy with biologic agents is effective to treat psoriasis, some patients do not achieve a satisfactory response.
- Adjunctive therapy with halobetasol propionate (HP) 0.01%–tazarotene (TAZ) 0.045% lotion can improve responses to biologic treatment in patients whose psoriasis symptoms could not be adequately controlled by biologic monotherapy.
- Adjunctive use of HP-TAZ lotion in addition to biologics was well tolerated.
- Compared with switching to a new biologic regimen, adding a topical regimen of HP-TAZ lotion to ongoing biologics may be a more cost-effective approach to enhance treatment effects.
Guttate Psoriasis Following COVID-19 Infection
Psoriasis is an inflammatory skin condition affecting 1% to 5% of the world population. 1 Guttate psoriasis is a subgroup of psoriasis that most commonly presents as raindroplike, erythematous, silvery, scaly papules. There have been limited reports of guttate psoriasis caused by rhinovirus and COVID-19 infection, but a PubMed search of articles indexed for MEDLINE using the term COVID-19 guttate psoriasis yielded only 3 documented cases of a psoriatic flare secondary to SARS-CoV-2 infection. 1-4 Herein, we detail a case in which a patient with mild SARS-CoV-2 infection who did not have a personal or family history of psoriasis experienced a moderate psoriatic flare 3 weeks after diagnosis of COVID-19.
Case Report
A 55-year-old woman was diagnosed with COVID-19 after SARS-CoV-2 RNA was detected from a nasopharyngeal swab. She reported moderate fatigue but no other symptoms. At the time of infection, she was not taking medications and reported neither a personal nor family history of psoriasis.
Three weeks after the COVID-19 diagnosis, she reported erythematous scaly papules only on the trunk and backs of the legs. Two months after the COVID-19 diagnosis, she was evaluated in our practice and diagnosed with guttate psoriasis. The patient refused biopsy. Physical examination revealed that the affected body surface area had increased to 5%; erythematous, silvery, scaly papules were found on the trunk, anterior and posterior legs, and lateral thighs (Figure). At the time of evaluation, she did not report joint pain or nail changes.
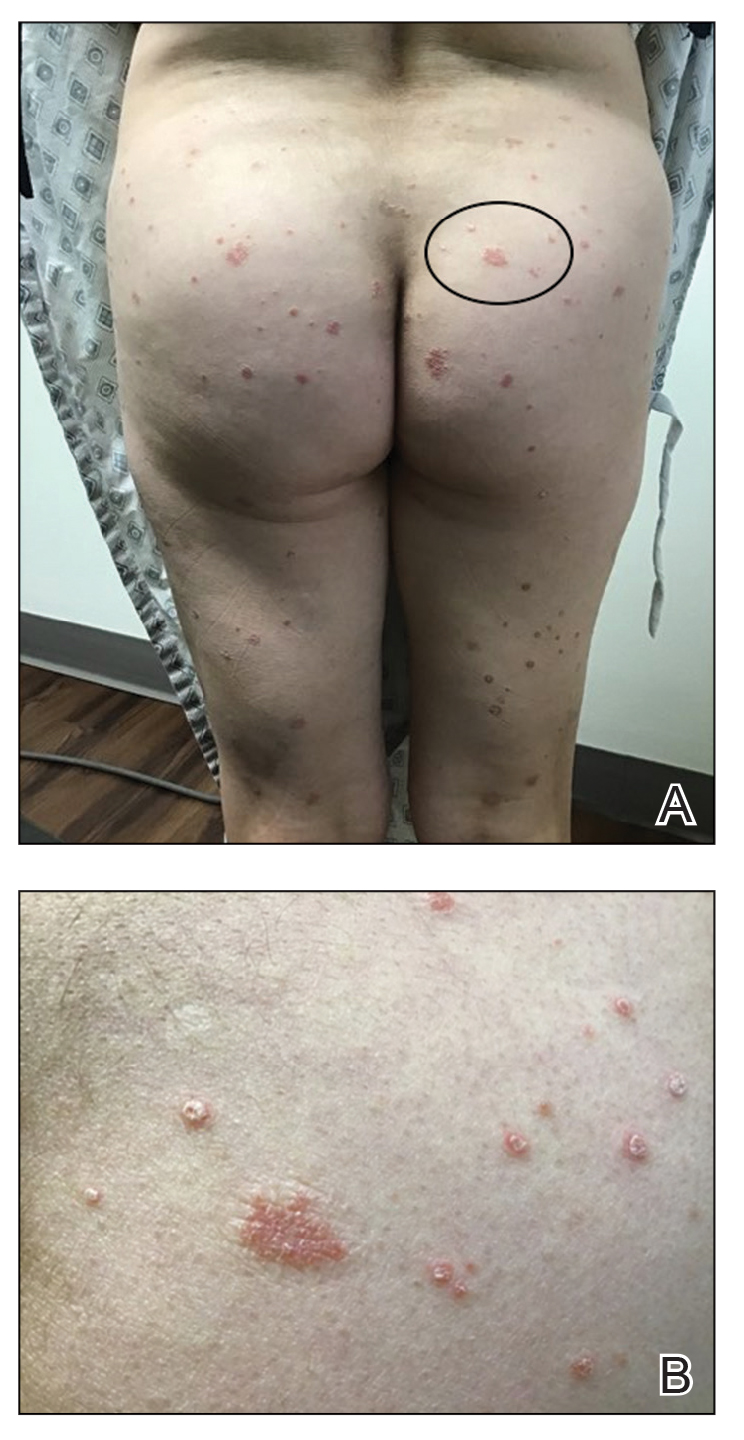
The patient was treated with triamcinolone acetonide cream 0.1% twice daily for 2 to 4 weeks. The guttate psoriasis resolved.
Comment
A sudden psoriatic flare can be linked to dysregulation of the innate immune response. Guttate psoriasis and generalized plaque-type psoriasis are postulated to have similar pathogenetic mechanisms, but guttate psoriasis is the only type of psoriasis that originates from viral infection. Initially, viral RNA will stimulate the toll-like receptor 3 protein, leading to increased production of the pathogenic cytokine IL-36γ and pathogenic chemokine CXCL8 (also known as IL-8), both of which are biomarkers for psoriasis.1 Specifically, IL-36γ and CXCL8 are known to further stimulate the proinflammatory cascade during the innate immune response displayed in guttate psoriasis.5,6
Our patient had a mild case of COVID-19, and she first reported the erythematous and scaly papules 3 weeks after infection. Dysregulation of proinflammatory cytokines must have started in the initial stages—within 7 days—of the viral infection. Guttate psoriasis arises within 3 weeks of infection with other viral and bacterial triggers, most commonly with streptococcal infections.1
Rodríguez et al7 described a phenomenon in which both SARS-CoV-2 and Middle East respiratory syndrome, both caused by a coronavirus, can lead to a reduction of type I interferon, which in turn leads to failure of control of viral replication during initial stages of a viral infection. This triggers an increase in proinflammatory cytokines and chemokines, including IL‐36γ and CXCL8. This pathologic mechanism might apply to SARS-CoV-2, as demonstrated in our patient’s sudden psoriatic flare 3 weeks after the COVID-19 diagnosis. However, further investigation and quantification of the putatively involved cytokines is necessary for confirmation.
Conclusion
Psoriasis, a chronic inflammatory skin condition, has been linked predominantly to genetic and environmental factors. Guttate psoriasis as a secondary reaction after streptococcal tonsillar and respiratory infections has been reported.1
Our case is the fourth documented case of guttate psoriasis secondary to COVID-19 infection.2-4 However, it is the second documented case of a patient with a diagnosis of guttate psoriasis secondary to COVID-19 infection who had neither a personal nor family history of psoriasis.
Because SARS-CoV-2 is a novel virus, the long-term effects of COVID-19 remain unclear. We report this case and its findings to introduce a novel clinical manifestation of SARS-CoV-2 infection.
- Sbidian E, Madrange M, Viguier M, et al. Respiratory virus infection triggers acute psoriasis flares across different clinical subtypes and genetic backgrounds. Br J Dermatol. 2019;181:1304-1306. doi:10.1111/bjd.18203
- Gananandan K, Sacks B, Ewing I. Guttate psoriasis secondary to COVID-19. BMJ Case Rep. 2020;13:e237367. doi:10.1136/bcr-2020-237367
- Rouai M, Rabhi F, Mansouri N, et al. New-onset guttate psoriasis secondary to COVID-19. Clin Case Rep. 2021;9:e04542. doi:10.1002/ccr3.4542
- Agarwal A, Tripathy T, Kar BR. Guttate flare in a patient with chronic plaque psoriasis following COVID-19 infection: a case report. J Cosmet Dermatol. 2021;20:3064-3065. doi:10.1111/jocd.14396
- Madonna S, Girolomoni G, Dinarello CA, et al. The significance of IL-36 hyperactivation and IL-36R targeting in psoriasis. Int J Mol Sci. 2019;20:3318. doi:10.3390/ijms20133318
- Nedoszytko B, Sokołowska-Wojdyło M, Ruckemann-Dziurdzin´ska K, et al. Chemokines and cytokines network in the pathogenesis of the inflammatory skin diseases: atopic dermatitis, psoriasis and skin mastocytosis. Postepy Dermatol Alergol. 2014;31:84-91. doi:10.5114/pdia.2014.40920
- Rodríguez Y, Novelli L, Rojas M, et al. Autoinflammatory and autoimmune conditions at the crossroad of COVID-19. J Autoimmun. 2020;114:102506. doi:10.1016/j.jaut.2020.102506
Psoriasis is an inflammatory skin condition affecting 1% to 5% of the world population. 1 Guttate psoriasis is a subgroup of psoriasis that most commonly presents as raindroplike, erythematous, silvery, scaly papules. There have been limited reports of guttate psoriasis caused by rhinovirus and COVID-19 infection, but a PubMed search of articles indexed for MEDLINE using the term COVID-19 guttate psoriasis yielded only 3 documented cases of a psoriatic flare secondary to SARS-CoV-2 infection. 1-4 Herein, we detail a case in which a patient with mild SARS-CoV-2 infection who did not have a personal or family history of psoriasis experienced a moderate psoriatic flare 3 weeks after diagnosis of COVID-19.
Case Report
A 55-year-old woman was diagnosed with COVID-19 after SARS-CoV-2 RNA was detected from a nasopharyngeal swab. She reported moderate fatigue but no other symptoms. At the time of infection, she was not taking medications and reported neither a personal nor family history of psoriasis.
Three weeks after the COVID-19 diagnosis, she reported erythematous scaly papules only on the trunk and backs of the legs. Two months after the COVID-19 diagnosis, she was evaluated in our practice and diagnosed with guttate psoriasis. The patient refused biopsy. Physical examination revealed that the affected body surface area had increased to 5%; erythematous, silvery, scaly papules were found on the trunk, anterior and posterior legs, and lateral thighs (Figure). At the time of evaluation, she did not report joint pain or nail changes.

The patient was treated with triamcinolone acetonide cream 0.1% twice daily for 2 to 4 weeks. The guttate psoriasis resolved.
Comment
A sudden psoriatic flare can be linked to dysregulation of the innate immune response. Guttate psoriasis and generalized plaque-type psoriasis are postulated to have similar pathogenetic mechanisms, but guttate psoriasis is the only type of psoriasis that originates from viral infection. Initially, viral RNA will stimulate the toll-like receptor 3 protein, leading to increased production of the pathogenic cytokine IL-36γ and pathogenic chemokine CXCL8 (also known as IL-8), both of which are biomarkers for psoriasis.1 Specifically, IL-36γ and CXCL8 are known to further stimulate the proinflammatory cascade during the innate immune response displayed in guttate psoriasis.5,6
Our patient had a mild case of COVID-19, and she first reported the erythematous and scaly papules 3 weeks after infection. Dysregulation of proinflammatory cytokines must have started in the initial stages—within 7 days—of the viral infection. Guttate psoriasis arises within 3 weeks of infection with other viral and bacterial triggers, most commonly with streptococcal infections.1
Rodríguez et al7 described a phenomenon in which both SARS-CoV-2 and Middle East respiratory syndrome, both caused by a coronavirus, can lead to a reduction of type I interferon, which in turn leads to failure of control of viral replication during initial stages of a viral infection. This triggers an increase in proinflammatory cytokines and chemokines, including IL‐36γ and CXCL8. This pathologic mechanism might apply to SARS-CoV-2, as demonstrated in our patient’s sudden psoriatic flare 3 weeks after the COVID-19 diagnosis. However, further investigation and quantification of the putatively involved cytokines is necessary for confirmation.
Conclusion
Psoriasis, a chronic inflammatory skin condition, has been linked predominantly to genetic and environmental factors. Guttate psoriasis as a secondary reaction after streptococcal tonsillar and respiratory infections has been reported.1
Our case is the fourth documented case of guttate psoriasis secondary to COVID-19 infection.2-4 However, it is the second documented case of a patient with a diagnosis of guttate psoriasis secondary to COVID-19 infection who had neither a personal nor family history of psoriasis.
Because SARS-CoV-2 is a novel virus, the long-term effects of COVID-19 remain unclear. We report this case and its findings to introduce a novel clinical manifestation of SARS-CoV-2 infection.
Psoriasis is an inflammatory skin condition affecting 1% to 5% of the world population. 1 Guttate psoriasis is a subgroup of psoriasis that most commonly presents as raindroplike, erythematous, silvery, scaly papules. There have been limited reports of guttate psoriasis caused by rhinovirus and COVID-19 infection, but a PubMed search of articles indexed for MEDLINE using the term COVID-19 guttate psoriasis yielded only 3 documented cases of a psoriatic flare secondary to SARS-CoV-2 infection. 1-4 Herein, we detail a case in which a patient with mild SARS-CoV-2 infection who did not have a personal or family history of psoriasis experienced a moderate psoriatic flare 3 weeks after diagnosis of COVID-19.
Case Report
A 55-year-old woman was diagnosed with COVID-19 after SARS-CoV-2 RNA was detected from a nasopharyngeal swab. She reported moderate fatigue but no other symptoms. At the time of infection, she was not taking medications and reported neither a personal nor family history of psoriasis.
Three weeks after the COVID-19 diagnosis, she reported erythematous scaly papules only on the trunk and backs of the legs. Two months after the COVID-19 diagnosis, she was evaluated in our practice and diagnosed with guttate psoriasis. The patient refused biopsy. Physical examination revealed that the affected body surface area had increased to 5%; erythematous, silvery, scaly papules were found on the trunk, anterior and posterior legs, and lateral thighs (Figure). At the time of evaluation, she did not report joint pain or nail changes.

The patient was treated with triamcinolone acetonide cream 0.1% twice daily for 2 to 4 weeks. The guttate psoriasis resolved.
Comment
A sudden psoriatic flare can be linked to dysregulation of the innate immune response. Guttate psoriasis and generalized plaque-type psoriasis are postulated to have similar pathogenetic mechanisms, but guttate psoriasis is the only type of psoriasis that originates from viral infection. Initially, viral RNA will stimulate the toll-like receptor 3 protein, leading to increased production of the pathogenic cytokine IL-36γ and pathogenic chemokine CXCL8 (also known as IL-8), both of which are biomarkers for psoriasis.1 Specifically, IL-36γ and CXCL8 are known to further stimulate the proinflammatory cascade during the innate immune response displayed in guttate psoriasis.5,6
Our patient had a mild case of COVID-19, and she first reported the erythematous and scaly papules 3 weeks after infection. Dysregulation of proinflammatory cytokines must have started in the initial stages—within 7 days—of the viral infection. Guttate psoriasis arises within 3 weeks of infection with other viral and bacterial triggers, most commonly with streptococcal infections.1
Rodríguez et al7 described a phenomenon in which both SARS-CoV-2 and Middle East respiratory syndrome, both caused by a coronavirus, can lead to a reduction of type I interferon, which in turn leads to failure of control of viral replication during initial stages of a viral infection. This triggers an increase in proinflammatory cytokines and chemokines, including IL‐36γ and CXCL8. This pathologic mechanism might apply to SARS-CoV-2, as demonstrated in our patient’s sudden psoriatic flare 3 weeks after the COVID-19 diagnosis. However, further investigation and quantification of the putatively involved cytokines is necessary for confirmation.
Conclusion
Psoriasis, a chronic inflammatory skin condition, has been linked predominantly to genetic and environmental factors. Guttate psoriasis as a secondary reaction after streptococcal tonsillar and respiratory infections has been reported.1
Our case is the fourth documented case of guttate psoriasis secondary to COVID-19 infection.2-4 However, it is the second documented case of a patient with a diagnosis of guttate psoriasis secondary to COVID-19 infection who had neither a personal nor family history of psoriasis.
Because SARS-CoV-2 is a novel virus, the long-term effects of COVID-19 remain unclear. We report this case and its findings to introduce a novel clinical manifestation of SARS-CoV-2 infection.
- Sbidian E, Madrange M, Viguier M, et al. Respiratory virus infection triggers acute psoriasis flares across different clinical subtypes and genetic backgrounds. Br J Dermatol. 2019;181:1304-1306. doi:10.1111/bjd.18203
- Gananandan K, Sacks B, Ewing I. Guttate psoriasis secondary to COVID-19. BMJ Case Rep. 2020;13:e237367. doi:10.1136/bcr-2020-237367
- Rouai M, Rabhi F, Mansouri N, et al. New-onset guttate psoriasis secondary to COVID-19. Clin Case Rep. 2021;9:e04542. doi:10.1002/ccr3.4542
- Agarwal A, Tripathy T, Kar BR. Guttate flare in a patient with chronic plaque psoriasis following COVID-19 infection: a case report. J Cosmet Dermatol. 2021;20:3064-3065. doi:10.1111/jocd.14396
- Madonna S, Girolomoni G, Dinarello CA, et al. The significance of IL-36 hyperactivation and IL-36R targeting in psoriasis. Int J Mol Sci. 2019;20:3318. doi:10.3390/ijms20133318
- Nedoszytko B, Sokołowska-Wojdyło M, Ruckemann-Dziurdzin´ska K, et al. Chemokines and cytokines network in the pathogenesis of the inflammatory skin diseases: atopic dermatitis, psoriasis and skin mastocytosis. Postepy Dermatol Alergol. 2014;31:84-91. doi:10.5114/pdia.2014.40920
- Rodríguez Y, Novelli L, Rojas M, et al. Autoinflammatory and autoimmune conditions at the crossroad of COVID-19. J Autoimmun. 2020;114:102506. doi:10.1016/j.jaut.2020.102506
- Sbidian E, Madrange M, Viguier M, et al. Respiratory virus infection triggers acute psoriasis flares across different clinical subtypes and genetic backgrounds. Br J Dermatol. 2019;181:1304-1306. doi:10.1111/bjd.18203
- Gananandan K, Sacks B, Ewing I. Guttate psoriasis secondary to COVID-19. BMJ Case Rep. 2020;13:e237367. doi:10.1136/bcr-2020-237367
- Rouai M, Rabhi F, Mansouri N, et al. New-onset guttate psoriasis secondary to COVID-19. Clin Case Rep. 2021;9:e04542. doi:10.1002/ccr3.4542
- Agarwal A, Tripathy T, Kar BR. Guttate flare in a patient with chronic plaque psoriasis following COVID-19 infection: a case report. J Cosmet Dermatol. 2021;20:3064-3065. doi:10.1111/jocd.14396
- Madonna S, Girolomoni G, Dinarello CA, et al. The significance of IL-36 hyperactivation and IL-36R targeting in psoriasis. Int J Mol Sci. 2019;20:3318. doi:10.3390/ijms20133318
- Nedoszytko B, Sokołowska-Wojdyło M, Ruckemann-Dziurdzin´ska K, et al. Chemokines and cytokines network in the pathogenesis of the inflammatory skin diseases: atopic dermatitis, psoriasis and skin mastocytosis. Postepy Dermatol Alergol. 2014;31:84-91. doi:10.5114/pdia.2014.40920
- Rodríguez Y, Novelli L, Rojas M, et al. Autoinflammatory and autoimmune conditions at the crossroad of COVID-19. J Autoimmun. 2020;114:102506. doi:10.1016/j.jaut.2020.102506
Practice Points
- Guttate psoriasis is the only type of psoriasis that originates from viral infection.
- Dysregulation of proinflammatory cytokines during COVID-19 infection in our patient led to development of guttate psoriasis 3 weeks later.
Severe Acute Systemic Reaction After the First Injections of Ixekizumab
Case Report
A 39-year-old woman who was otherwise healthy presented with fatigue, malaise, a resolving rash, focal lymphadenopathy, increasing distal arthritis, dactylitis, resolving ecchymoses, and acute onycholysis of 1 week’s duration that developed 13 days after initiating ixekizumab. The patient had a history of psoriasis and psoriatic arthritis for more than 10 years. She had been successfully treated in the past for psoriasis with adalimumab for several years; however, adalimumab was discontinued after an episode of Clostridium difficile colitis. The patient had a negative purified protein derivative (tuberculin) test prior to starting biologics as she works in the health care field. Routine follow-up purified protein derivative (tuberculin) test was positive. She discontinued all therapy for psoriasis and psoriatic arthritis prior to being appropriately treated for 6 months under the care of infectious disease physicians. She then had several pregnancies and chose to restart biologic treatment after weaning her third child from breastfeeding, as her skin and joint disease were notably flaring.
Ustekinumab was chosen to shift treatment away from tumor necrosis factor (TNF) α inhibitors. The patient's condition was under relatively good control for 1 year; however, she experienced notable gastrointestinal tract upset (ie, intermittent diarrhea and constipation), despite multiple negative tests for C difficile. The patient was referred to see a gastroenterologist but never followed up. Due to long-term low-grade gastrointestinal problems, ustekinumab was discontinued, and the gastrointestinal symptoms resolved without treatment.
Given the side effects noted with TNF-α and IL-12/23 inhibitors and the fact that the patient’s cutaneous and joint disease were notable, the decision was made to start the IL-17A inhibitor ixekizumab. The patient administered 2 injections, one in each thigh. Within 12 hours, she experienced severe injection-site pain. The pain was so severe that it woke her from sleep the night of the first injections. She then developed severe pain in the right axilla that limited upper extremity mobility. Within 48 hours, she developed an erythematous, nonpruritic, nonscaly, mottled rash on the right breast that began to resolve within 24 hours without treatment. In addition, 3 days after the injections, she developed ecchymoses on the trunk and extremities without any identifiable trauma, severe acute onycholysis in several fingernails (Figure 1) and toenails, dactylitis such that she could not wear her wedding ring, and a flare of psoriatic arthritis in the fingers and ankles.
At the current presentation (2 weeks after the injections), the patient reported malaise, flulike symptoms, and low-grade intermittent fevers. Results from a hematology panel displayed leukopenia at 2.69×103/μL (reference range, 3.54–9.06×103/μL) and thrombocytopenia at 114×103/μL (reference range, 165–415×103/μL).1 Her most recent laboratory results before the ixekizumab injections displayed a white blood cell count level at 4.6×103/μL and platelet count at 159×103/μL. C-reactive protein and erythrocyte sedimentation rate were within reference range. A shave biopsy of an erythematous nodule on the proximal interphalangeal joint of the fourth finger on the right hand displayed spongiotic dermatitis with eosinophils (Figure 2).
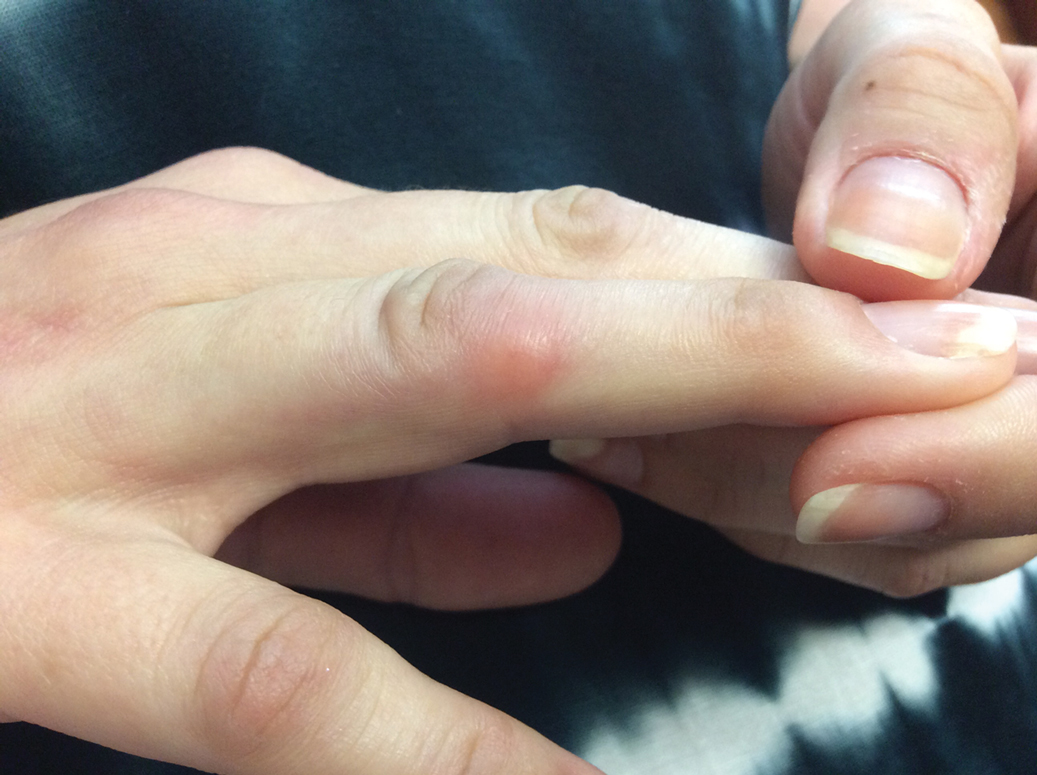
Interestingly, the psoriatic plaques on the scalp, trunk, and extremities had nearly completely resolved after only the first 2 injections. However, given the side effects, the second dose of ixekizumab was held, repeat laboratory tests were ordered to ensure normalization of cytopenia, and the patient was transitioned to pulse-dose topical steroids to control the remaining psoriatic plaques.
One week after presentation (3 weeks after the initial injections), the patient’s systemic symptoms had almost completely resolved, and she denied any further concerns. Her fingernails and toenails, however, continued to show the changes of onycholysis noted at the visit.
Comment
Ixekizumab is a human IgG4 monoclonal antibody that binds to IL-17A, one of the cytokines involved in the pathogenesis of psoriasis. The monoclonal antibody prevents its attachment to the IL-17 receptor, which inhibits the release of further cytokines and chemokines, decreasing the inflammatory and immune response.2
Ixekizumab was approved by the US Food and Drug Administration for plaque psoriasis after 3 clinical trials—UNCOVER-1, UNCOVER-2, and UNCOVER-3—were performed. In UNCOVER-3, the most common side effects that occurred—nasopharyngitis, upper respiratory tract infection, injection-site reaction, arthralgia, headache, and infections (specifically candidiasis)—generally were well tolerated. More serious adverse events included cardiovascular and cerebrovascular events, inflammatory bowel disease, and nonmelanoma skin cancer.3
Notable laboratory abnormalities that have been documented from ixekizumab include elevated liver function tests (eg, alanine aminotransferase, aspartate aminotransferase, bilirubin, and alkaline phosphatase), as well as leukopenia, neutropenia, and thrombocytopenia.4 Although short-term thrombocytopenia, as described in our patient, provides an explanation for the bruising noted on observation, it is unusual to note such notable ecchymoses within days of the first injection.
Onycholysis has not been documented as a side effect of ixekizumab; however, it has been reported as an adverse event from other biologic medications. Sfikakis et al5 reported 5 patients who developed psoriatic skin lesions after treatment with 3 different anti-TNF biologics—infliximab, adalimumab, or etanercept—fo
The exact pathophysiology of these adverse events has not been clearly understood, but it has been proposed that anti-TNF biologics may initiate an autoimmune reaction in the skin and nails, leading to paradoxical psoriasis and nail changes such as onycholysis. Tumor necrosis factor may have a regulatory role in the skin that prevents autoreactive T cells, such as cutaneous lymphocyte antigen–expressing T cells that promote the formation of psoriasiform lesions. By inhibiting TNF, there can be an underlying activation of autoreactive T cells that leads to tissue destruction in the skin and nails.6 Anti-TNF biologics also could increase CXCR3, a chemokine receptor that allows autoreactive T cells to enter the skin and cause pathology.7
IL-17A and IL-17F also have been shown to upregulate the expression of TNF receptor II in synoviocytes,8 which demonstrates that IL-17 works in synergy with TNF-α to promote an inflammatory reaction.9 Due to the inhibitory effects of ixekizumab, psoriatic arthritis should theoretically improve. However, if there is an alteration in the inflammatory sequence, then the regulatory role of TNF could be suppressed and psoriatic arthritis could become exacerbated. Additionally, its associated symptoms, such as dactylitis, could develop, as seen in our patient.4 Because psoriatic arthritis is closely associated with nail changes of psoriasis, it is conceivable that acute arthritic flares and acute onycholysis are both induced by the same cytokine dysregulation. Further studies and a larger patient population need to be evaluated to determine the exact cause of the acute exacerbation of psoriatic arthritis with concomitant nail changes as noted in our patient.
Acute onycholysis (within 72 hours) is a rare side effect of ixekizumab. It can be postulated that our patient’s severe acute onycholysis associated with a flare of psoriatic arthritis could be due to idiosyncratic immune dysregulation, promoting the activity of autoreactive T cells. The pharmacologic effects of ixekizumab occur through the inhibition of IL-17. We propose that by inhibiting IL-17 with associated TNF alterations, an altered inflammatory cascade could promote an autoimmune reaction leading to the described pathology.
- Kratz A, Pesce MA, Basner RC, et al. Laboratory values of clinical importance. In: Kasper D, Fauci A, Hauser S, et al, eds. Harrison’s Principles of Internal Medicine. 19th ed. McGraw-Hill; 2014.
- Ixekizumab. Package insert. Eli Lilly & Co; 2017.
- Gordon KB, Blauvelt A, Papp KA, et al. Phase 3 trials of ixekizumab in moderate-to-severe plaque psoriasis. N Engl J Med. 2016;375:345-356.
- Leonardi C, Matheson R, Zachariae C, et al. Anti-interleukin-17 monoclonal antibody ixekizumab in chronic plaque psoriasis. N Engl J Med. 2012;366:1190-1199.
- Sfikakis PP, Iliopoulos A, Elezoglou A, et al. Psoriasis induced by anti-tumor necrosis factor therapy: a paradoxical adverse reaction. Arthritis Rheum. 2005;52:2513-2518.
- Berg EL, Yoshino T, Rott LS, et al. The cutaneous lymphocyte antigen is a skin lymphocyte homing receptor for the vascular lectin endothelial cell-leukocyte adhesion molecule 1. J Exp Med. 1991;174:1461-1466.
- Flier J, Boorsma DM, van Beek PJ, et al. Differential expression of CXCR3 targeting chemokines CXCL10, CXCL9, and CXCL11 in different types of skin inflammation. J Pathol. 2001;194:398-405.
- Zrioual S, Ecochard R, Tournadre A, et al. Genome-wide comparison between IL-17A- and IL-17F-induced effects in human rheumatoid arthritis synoviocytes. J Immunol. 2009;182:3112-3120.
- Gaffen SL. The role of interleukin-17 in the pathogenesis of rheumatoid arthritis. Curr Rheumatol Rep. 2009;11:365-370.
Case Report
A 39-year-old woman who was otherwise healthy presented with fatigue, malaise, a resolving rash, focal lymphadenopathy, increasing distal arthritis, dactylitis, resolving ecchymoses, and acute onycholysis of 1 week’s duration that developed 13 days after initiating ixekizumab. The patient had a history of psoriasis and psoriatic arthritis for more than 10 years. She had been successfully treated in the past for psoriasis with adalimumab for several years; however, adalimumab was discontinued after an episode of Clostridium difficile colitis. The patient had a negative purified protein derivative (tuberculin) test prior to starting biologics as she works in the health care field. Routine follow-up purified protein derivative (tuberculin) test was positive. She discontinued all therapy for psoriasis and psoriatic arthritis prior to being appropriately treated for 6 months under the care of infectious disease physicians. She then had several pregnancies and chose to restart biologic treatment after weaning her third child from breastfeeding, as her skin and joint disease were notably flaring.
Ustekinumab was chosen to shift treatment away from tumor necrosis factor (TNF) α inhibitors. The patient's condition was under relatively good control for 1 year; however, she experienced notable gastrointestinal tract upset (ie, intermittent diarrhea and constipation), despite multiple negative tests for C difficile. The patient was referred to see a gastroenterologist but never followed up. Due to long-term low-grade gastrointestinal problems, ustekinumab was discontinued, and the gastrointestinal symptoms resolved without treatment.
Given the side effects noted with TNF-α and IL-12/23 inhibitors and the fact that the patient’s cutaneous and joint disease were notable, the decision was made to start the IL-17A inhibitor ixekizumab. The patient administered 2 injections, one in each thigh. Within 12 hours, she experienced severe injection-site pain. The pain was so severe that it woke her from sleep the night of the first injections. She then developed severe pain in the right axilla that limited upper extremity mobility. Within 48 hours, she developed an erythematous, nonpruritic, nonscaly, mottled rash on the right breast that began to resolve within 24 hours without treatment. In addition, 3 days after the injections, she developed ecchymoses on the trunk and extremities without any identifiable trauma, severe acute onycholysis in several fingernails (Figure 1) and toenails, dactylitis such that she could not wear her wedding ring, and a flare of psoriatic arthritis in the fingers and ankles.

At the current presentation (2 weeks after the injections), the patient reported malaise, flulike symptoms, and low-grade intermittent fevers. Results from a hematology panel displayed leukopenia at 2.69×103/μL (reference range, 3.54–9.06×103/μL) and thrombocytopenia at 114×103/μL (reference range, 165–415×103/μL).1 Her most recent laboratory results before the ixekizumab injections displayed a white blood cell count level at 4.6×103/μL and platelet count at 159×103/μL. C-reactive protein and erythrocyte sedimentation rate were within reference range. A shave biopsy of an erythematous nodule on the proximal interphalangeal joint of the fourth finger on the right hand displayed spongiotic dermatitis with eosinophils (Figure 2).

Interestingly, the psoriatic plaques on the scalp, trunk, and extremities had nearly completely resolved after only the first 2 injections. However, given the side effects, the second dose of ixekizumab was held, repeat laboratory tests were ordered to ensure normalization of cytopenia, and the patient was transitioned to pulse-dose topical steroids to control the remaining psoriatic plaques.
One week after presentation (3 weeks after the initial injections), the patient’s systemic symptoms had almost completely resolved, and she denied any further concerns. Her fingernails and toenails, however, continued to show the changes of onycholysis noted at the visit.
Comment
Ixekizumab is a human IgG4 monoclonal antibody that binds to IL-17A, one of the cytokines involved in the pathogenesis of psoriasis. The monoclonal antibody prevents its attachment to the IL-17 receptor, which inhibits the release of further cytokines and chemokines, decreasing the inflammatory and immune response.2
Ixekizumab was approved by the US Food and Drug Administration for plaque psoriasis after 3 clinical trials—UNCOVER-1, UNCOVER-2, and UNCOVER-3—were performed. In UNCOVER-3, the most common side effects that occurred—nasopharyngitis, upper respiratory tract infection, injection-site reaction, arthralgia, headache, and infections (specifically candidiasis)—generally were well tolerated. More serious adverse events included cardiovascular and cerebrovascular events, inflammatory bowel disease, and nonmelanoma skin cancer.3
Notable laboratory abnormalities that have been documented from ixekizumab include elevated liver function tests (eg, alanine aminotransferase, aspartate aminotransferase, bilirubin, and alkaline phosphatase), as well as leukopenia, neutropenia, and thrombocytopenia.4 Although short-term thrombocytopenia, as described in our patient, provides an explanation for the bruising noted on observation, it is unusual to note such notable ecchymoses within days of the first injection.
Onycholysis has not been documented as a side effect of ixekizumab; however, it has been reported as an adverse event from other biologic medications. Sfikakis et al5 reported 5 patients who developed psoriatic skin lesions after treatment with 3 different anti-TNF biologics—infliximab, adalimumab, or etanercept—fo
The exact pathophysiology of these adverse events has not been clearly understood, but it has been proposed that anti-TNF biologics may initiate an autoimmune reaction in the skin and nails, leading to paradoxical psoriasis and nail changes such as onycholysis. Tumor necrosis factor may have a regulatory role in the skin that prevents autoreactive T cells, such as cutaneous lymphocyte antigen–expressing T cells that promote the formation of psoriasiform lesions. By inhibiting TNF, there can be an underlying activation of autoreactive T cells that leads to tissue destruction in the skin and nails.6 Anti-TNF biologics also could increase CXCR3, a chemokine receptor that allows autoreactive T cells to enter the skin and cause pathology.7
IL-17A and IL-17F also have been shown to upregulate the expression of TNF receptor II in synoviocytes,8 which demonstrates that IL-17 works in synergy with TNF-α to promote an inflammatory reaction.9 Due to the inhibitory effects of ixekizumab, psoriatic arthritis should theoretically improve. However, if there is an alteration in the inflammatory sequence, then the regulatory role of TNF could be suppressed and psoriatic arthritis could become exacerbated. Additionally, its associated symptoms, such as dactylitis, could develop, as seen in our patient.4 Because psoriatic arthritis is closely associated with nail changes of psoriasis, it is conceivable that acute arthritic flares and acute onycholysis are both induced by the same cytokine dysregulation. Further studies and a larger patient population need to be evaluated to determine the exact cause of the acute exacerbation of psoriatic arthritis with concomitant nail changes as noted in our patient.
Acute onycholysis (within 72 hours) is a rare side effect of ixekizumab. It can be postulated that our patient’s severe acute onycholysis associated with a flare of psoriatic arthritis could be due to idiosyncratic immune dysregulation, promoting the activity of autoreactive T cells. The pharmacologic effects of ixekizumab occur through the inhibition of IL-17. We propose that by inhibiting IL-17 with associated TNF alterations, an altered inflammatory cascade could promote an autoimmune reaction leading to the described pathology.
Case Report
A 39-year-old woman who was otherwise healthy presented with fatigue, malaise, a resolving rash, focal lymphadenopathy, increasing distal arthritis, dactylitis, resolving ecchymoses, and acute onycholysis of 1 week’s duration that developed 13 days after initiating ixekizumab. The patient had a history of psoriasis and psoriatic arthritis for more than 10 years. She had been successfully treated in the past for psoriasis with adalimumab for several years; however, adalimumab was discontinued after an episode of Clostridium difficile colitis. The patient had a negative purified protein derivative (tuberculin) test prior to starting biologics as she works in the health care field. Routine follow-up purified protein derivative (tuberculin) test was positive. She discontinued all therapy for psoriasis and psoriatic arthritis prior to being appropriately treated for 6 months under the care of infectious disease physicians. She then had several pregnancies and chose to restart biologic treatment after weaning her third child from breastfeeding, as her skin and joint disease were notably flaring.
Ustekinumab was chosen to shift treatment away from tumor necrosis factor (TNF) α inhibitors. The patient's condition was under relatively good control for 1 year; however, she experienced notable gastrointestinal tract upset (ie, intermittent diarrhea and constipation), despite multiple negative tests for C difficile. The patient was referred to see a gastroenterologist but never followed up. Due to long-term low-grade gastrointestinal problems, ustekinumab was discontinued, and the gastrointestinal symptoms resolved without treatment.
Given the side effects noted with TNF-α and IL-12/23 inhibitors and the fact that the patient’s cutaneous and joint disease were notable, the decision was made to start the IL-17A inhibitor ixekizumab. The patient administered 2 injections, one in each thigh. Within 12 hours, she experienced severe injection-site pain. The pain was so severe that it woke her from sleep the night of the first injections. She then developed severe pain in the right axilla that limited upper extremity mobility. Within 48 hours, she developed an erythematous, nonpruritic, nonscaly, mottled rash on the right breast that began to resolve within 24 hours without treatment. In addition, 3 days after the injections, she developed ecchymoses on the trunk and extremities without any identifiable trauma, severe acute onycholysis in several fingernails (Figure 1) and toenails, dactylitis such that she could not wear her wedding ring, and a flare of psoriatic arthritis in the fingers and ankles.

At the current presentation (2 weeks after the injections), the patient reported malaise, flulike symptoms, and low-grade intermittent fevers. Results from a hematology panel displayed leukopenia at 2.69×103/μL (reference range, 3.54–9.06×103/μL) and thrombocytopenia at 114×103/μL (reference range, 165–415×103/μL).1 Her most recent laboratory results before the ixekizumab injections displayed a white blood cell count level at 4.6×103/μL and platelet count at 159×103/μL. C-reactive protein and erythrocyte sedimentation rate were within reference range. A shave biopsy of an erythematous nodule on the proximal interphalangeal joint of the fourth finger on the right hand displayed spongiotic dermatitis with eosinophils (Figure 2).

Interestingly, the psoriatic plaques on the scalp, trunk, and extremities had nearly completely resolved after only the first 2 injections. However, given the side effects, the second dose of ixekizumab was held, repeat laboratory tests were ordered to ensure normalization of cytopenia, and the patient was transitioned to pulse-dose topical steroids to control the remaining psoriatic plaques.
One week after presentation (3 weeks after the initial injections), the patient’s systemic symptoms had almost completely resolved, and she denied any further concerns. Her fingernails and toenails, however, continued to show the changes of onycholysis noted at the visit.
Comment
Ixekizumab is a human IgG4 monoclonal antibody that binds to IL-17A, one of the cytokines involved in the pathogenesis of psoriasis. The monoclonal antibody prevents its attachment to the IL-17 receptor, which inhibits the release of further cytokines and chemokines, decreasing the inflammatory and immune response.2
Ixekizumab was approved by the US Food and Drug Administration for plaque psoriasis after 3 clinical trials—UNCOVER-1, UNCOVER-2, and UNCOVER-3—were performed. In UNCOVER-3, the most common side effects that occurred—nasopharyngitis, upper respiratory tract infection, injection-site reaction, arthralgia, headache, and infections (specifically candidiasis)—generally were well tolerated. More serious adverse events included cardiovascular and cerebrovascular events, inflammatory bowel disease, and nonmelanoma skin cancer.3
Notable laboratory abnormalities that have been documented from ixekizumab include elevated liver function tests (eg, alanine aminotransferase, aspartate aminotransferase, bilirubin, and alkaline phosphatase), as well as leukopenia, neutropenia, and thrombocytopenia.4 Although short-term thrombocytopenia, as described in our patient, provides an explanation for the bruising noted on observation, it is unusual to note such notable ecchymoses within days of the first injection.
Onycholysis has not been documented as a side effect of ixekizumab; however, it has been reported as an adverse event from other biologic medications. Sfikakis et al5 reported 5 patients who developed psoriatic skin lesions after treatment with 3 different anti-TNF biologics—infliximab, adalimumab, or etanercept—fo
The exact pathophysiology of these adverse events has not been clearly understood, but it has been proposed that anti-TNF biologics may initiate an autoimmune reaction in the skin and nails, leading to paradoxical psoriasis and nail changes such as onycholysis. Tumor necrosis factor may have a regulatory role in the skin that prevents autoreactive T cells, such as cutaneous lymphocyte antigen–expressing T cells that promote the formation of psoriasiform lesions. By inhibiting TNF, there can be an underlying activation of autoreactive T cells that leads to tissue destruction in the skin and nails.6 Anti-TNF biologics also could increase CXCR3, a chemokine receptor that allows autoreactive T cells to enter the skin and cause pathology.7
IL-17A and IL-17F also have been shown to upregulate the expression of TNF receptor II in synoviocytes,8 which demonstrates that IL-17 works in synergy with TNF-α to promote an inflammatory reaction.9 Due to the inhibitory effects of ixekizumab, psoriatic arthritis should theoretically improve. However, if there is an alteration in the inflammatory sequence, then the regulatory role of TNF could be suppressed and psoriatic arthritis could become exacerbated. Additionally, its associated symptoms, such as dactylitis, could develop, as seen in our patient.4 Because psoriatic arthritis is closely associated with nail changes of psoriasis, it is conceivable that acute arthritic flares and acute onycholysis are both induced by the same cytokine dysregulation. Further studies and a larger patient population need to be evaluated to determine the exact cause of the acute exacerbation of psoriatic arthritis with concomitant nail changes as noted in our patient.
Acute onycholysis (within 72 hours) is a rare side effect of ixekizumab. It can be postulated that our patient’s severe acute onycholysis associated with a flare of psoriatic arthritis could be due to idiosyncratic immune dysregulation, promoting the activity of autoreactive T cells. The pharmacologic effects of ixekizumab occur through the inhibition of IL-17. We propose that by inhibiting IL-17 with associated TNF alterations, an altered inflammatory cascade could promote an autoimmune reaction leading to the described pathology.
- Kratz A, Pesce MA, Basner RC, et al. Laboratory values of clinical importance. In: Kasper D, Fauci A, Hauser S, et al, eds. Harrison’s Principles of Internal Medicine. 19th ed. McGraw-Hill; 2014.
- Ixekizumab. Package insert. Eli Lilly & Co; 2017.
- Gordon KB, Blauvelt A, Papp KA, et al. Phase 3 trials of ixekizumab in moderate-to-severe plaque psoriasis. N Engl J Med. 2016;375:345-356.
- Leonardi C, Matheson R, Zachariae C, et al. Anti-interleukin-17 monoclonal antibody ixekizumab in chronic plaque psoriasis. N Engl J Med. 2012;366:1190-1199.
- Sfikakis PP, Iliopoulos A, Elezoglou A, et al. Psoriasis induced by anti-tumor necrosis factor therapy: a paradoxical adverse reaction. Arthritis Rheum. 2005;52:2513-2518.
- Berg EL, Yoshino T, Rott LS, et al. The cutaneous lymphocyte antigen is a skin lymphocyte homing receptor for the vascular lectin endothelial cell-leukocyte adhesion molecule 1. J Exp Med. 1991;174:1461-1466.
- Flier J, Boorsma DM, van Beek PJ, et al. Differential expression of CXCR3 targeting chemokines CXCL10, CXCL9, and CXCL11 in different types of skin inflammation. J Pathol. 2001;194:398-405.
- Zrioual S, Ecochard R, Tournadre A, et al. Genome-wide comparison between IL-17A- and IL-17F-induced effects in human rheumatoid arthritis synoviocytes. J Immunol. 2009;182:3112-3120.
- Gaffen SL. The role of interleukin-17 in the pathogenesis of rheumatoid arthritis. Curr Rheumatol Rep. 2009;11:365-370.
- Kratz A, Pesce MA, Basner RC, et al. Laboratory values of clinical importance. In: Kasper D, Fauci A, Hauser S, et al, eds. Harrison’s Principles of Internal Medicine. 19th ed. McGraw-Hill; 2014.
- Ixekizumab. Package insert. Eli Lilly & Co; 2017.
- Gordon KB, Blauvelt A, Papp KA, et al. Phase 3 trials of ixekizumab in moderate-to-severe plaque psoriasis. N Engl J Med. 2016;375:345-356.
- Leonardi C, Matheson R, Zachariae C, et al. Anti-interleukin-17 monoclonal antibody ixekizumab in chronic plaque psoriasis. N Engl J Med. 2012;366:1190-1199.
- Sfikakis PP, Iliopoulos A, Elezoglou A, et al. Psoriasis induced by anti-tumor necrosis factor therapy: a paradoxical adverse reaction. Arthritis Rheum. 2005;52:2513-2518.
- Berg EL, Yoshino T, Rott LS, et al. The cutaneous lymphocyte antigen is a skin lymphocyte homing receptor for the vascular lectin endothelial cell-leukocyte adhesion molecule 1. J Exp Med. 1991;174:1461-1466.
- Flier J, Boorsma DM, van Beek PJ, et al. Differential expression of CXCR3 targeting chemokines CXCL10, CXCL9, and CXCL11 in different types of skin inflammation. J Pathol. 2001;194:398-405.
- Zrioual S, Ecochard R, Tournadre A, et al. Genome-wide comparison between IL-17A- and IL-17F-induced effects in human rheumatoid arthritis synoviocytes. J Immunol. 2009;182:3112-3120.
- Gaffen SL. The role of interleukin-17 in the pathogenesis of rheumatoid arthritis. Curr Rheumatol Rep. 2009;11:365-370.
Practice Points
- Psoriasis is an autoimmune disorder with a predominance of CD4+ and CD8+ T cells that release cytokines, such as tumor necrosis factor 11α and interleukins, which promote inflammation in the skin and joints and is associated with systemic inflammation predisposing patients to cardiovascular disease.
- Common adverse effects of most biologic medications for psoriasis include injection-site pain and rash, fever, malaise, back pain, urticaria and flushing, edema, dyspnea, and nausea.
- Ixekizumab is a humanized IL-17A antagonist intended for adults with moderate to severe psoriasis. Certain rare side effects specific to ixekizumab include inflammatory bowel disease, thrombocytopenia, severe injection-site reactions, and candidiasis.
- Acute onycholysis and acute exacerbation of arthritis/dactylitis are rare side effects of ixekizumab therapy.
A dermatologist-led model for CVD prevention in psoriasis may be feasible
A – may be feasible, given the positive perspectives expressed by both clinicians and patients in a set of electronic surveys, researchers say.
In an analysis of survey responses from 183 dermatologists and 322 patients, John S. Barbieri, MD, MBA, and coinvestigators found that more than two-thirds of dermatologists (69.3%) agreed it “seems doable” to check lipids and calculate a 10-year cardiovascular risk score, and over one-third (36.1%) agreed they could prescribe statins when indicated.
The patient survey was distributed through the National Psoriasis Foundation to individuals who were seeing a dermatologist or rheumatologist for psoriatic disease; the clinician survey was distributed through the American Academy of Dermatology to dermatologists who reported caring for patients with psoriasis. (A survey of rheumatologists was similarly conducted, but the number of participants fell short of the needed sample size.)
Most patients surveyed indicated they would be receptive to their dermatologist (or rheumatologist) playing a larger role in screening and managing CVD risk, and that they would be similarly likely to follow recommendations regarding risk screening and management whether the advice came their dermatologist/rheumatologist or from their PCP.
The clinician survey focused on lipids and statin use, and did not address other elements of risk management. Still, the researchers see their findings as an early but promising step in finding better models to improve cardiovascular outcomes for patients with psoriatic disease, who too often do not engage with their PCPs despite their increased risk of CVD and a higher risk of premature mortality from CVD.
Fewer than half of commercially insured adults aged under 65 years visit a PCP each year, the researchers noted. And among the patients in their survey, approximately 20% did not have a PCP or had not seen their PCP in the past year.
Other research has shown that only a small minority of patients with psoriasis have an encounter with their PCP within a year of establishing care with their dermatologist, and that “over half of patients with psoriasis have undetected risk factors like dyslipidemia or hypertension,” Dr. Barbieri, of the department of dermatology at Brigham and Women’s Hospital, Boston, said in an interview.
“There’s a gap here, a missing link in the chain of cardiovascular disease prevention,” he said. “What if the dermatologist or rheumatologist could be more engaged in [CV] risk protection? ... It’s the idea of meeting the patients where they are.”
The surveys
The clinician survey focused on statins because of their ease of use, efficacy and safety, and the need for minimal monitoring, Dr. Barbieri said in the interview. “On the spectrum of things you can do for cardiovascular disease prevention, it’s one of the easiest ones.”
In an accompanying editorial, cardiologists Michael S. Garshick, MD, MS, and Jeffrey S. Berger, MD, MS, both of the department of medicine, New York University, wrote that, “despite the well-described association between psoriasis and CVD, only 35% of patients with psoriasis diagnosed with hyperlipidemia are adequately treated with statin therapy.”
“For many of these patients, their dermatologist or rheumatologist may be their only source of contact with the health care system,” they added.
Most studies targeting CVD risk in psoriasis have focused on targeting psoriatic inflammation, and few studies have explored strategies to improve modifiable CVD risk factor control with pharmacological therapy, they said.
In addition to the questions about receptiveness to identifying and potentially treating CVD risk with statins, the dermatologist survey included a best-worst scaling choice experiment to assess preferences for implementation approaches. Dermatologists were asked to rank their preferences for eight implementation strategies that have been shown in published studies to help increase statin prescribing rates.
The three highest-ranked strategies among dermatologists were clinical decision support, physician educational outreach, and patient education materials. The lowest-ranked strategies were comparisons with peers, a pay-for-performance option, and a mobile app/texting service to remind patients to undergo CVD risk screening.
Of the 183 dermatologists in the survey, 28.4% were from academic settings, 11.5% were from multispecialty groups, and 45.4% were from dermatology groups. (A low response rate of 5.2% for dermatologists raises some questions about the generalizability of the findings, Dr. Garshick and Dr. Berger noted in their editorial.)
Where to go from here?
Asked to comment on the results, Jashin J. Wu, MD, founder and CEO of the Dermatology Research and Education Foundation, Irvine, Calif., who was not involved with the study, said that a larger role in CVD risk management is “not likely to find traction with everyday dermatologists.”
“It’s already a big ask for community dermatologists to go through the approval process to get biologics for patients, so I don’t think many would be willing to add more to their plate by taking a bigger role in CVD management,” he said in an interview. He generally has not prescribed statins, “as I don’t feel that is in my scope of work.”
In the interview, Dr. Barbieri said that a parallel qualitative study, not yet published, has looked at the facilitators and barriers – including time constraints and concern about scope of practice – to statin prescribing and other elements of cardiovascular risk reduction.
All told, he said, a centralized care coordinator model may be the best approach to engage the dermatologist more in CVD prevention, including lipid management, but to also “offload some of the management responsibility.”
In this model, which is partially described by Dr. Barbieri and colleagues, the dermatologist (or rheumatologist) would educate the patient, measure blood pressure and check a lipid panel, and refer the patient to a coordinator who would, in turn, collect more information and calculate a 10-year CVD risk score.
Using a protocol-driven clinical decision support approach, the care coordinator would provide counseling about diet, exercise, and smoking cessation, and about whether statin therapy or blood pressure management is indicated.
“That coordinator would be in a good position to help the patient work with their PCP, if they have one, to find a PCP if they don’t, or to use telemedicine or work with their dermatologist or rheumatologist,” Dr. Barbieri said.
The centralized care coordinator service could be funded through grants, charitable funds, and patient assistance funds so that it is free to patients, he said, and could possibly be “housed in the National Psoriasis Foundation.”
Dr. Barbieri said he and his colleagues plan to design a clinical trial to test whether such a model can be adopted in practice and whether it can improve outcomes associated with CVD risk management.
In their editorial, Dr. Garshick and Dr. Berger, who is director of NYU Langone’s Center for the Prevention of Cardiovascular Disease, wrote that many patients with psoriatic disease have or are at risk for cardiometabolic conditions, and that CVD risk reduction should extend beyond lipid management to include blood pressure, glucose lowering, obesity management, and antiplatelet therapy.
The joint AAD-NPF guidelines for the management and treatment of psoriasis with awareness and attention to comorbidities, published in 2019, were among the first to formally recognize the enhanced CVD risk of patients with psoriasis, they noted.
The guidelines call upon dermatologists to inform patients of the psoriasis-CVD association and ensure their patients are engaged with their PCP or cardiologist for appropriate screening. Now, the editorialists say, “moving the needle forward includes refining and developing modifiable CVD risk reduction strategies for patients with psoriasis, and collaboration between the fields of dermatology, rheumatology, and cardiology is key.”
Incorporating a preventive cardiologist into combined dermatology-rheumatology clinics, or partnering as a freestanding cardioinflammatory clinic, also have potential to improve CVD risk, they wrote.
The survey study was supported by a grant from the NPF Psoriasis Prevention Initiative. Dr. Barbieri reported no conflicts of interest. Several authors disclosed consulting fees and grants from numerous pharmaceutical companies. Dr. Berger reported receiving personal fees from Janssen and grants from AstraZeneca outside of the submitted work. Dr. Garshick reported receiving personal fees from AbbVie outside of the submitted work.
A – may be feasible, given the positive perspectives expressed by both clinicians and patients in a set of electronic surveys, researchers say.
In an analysis of survey responses from 183 dermatologists and 322 patients, John S. Barbieri, MD, MBA, and coinvestigators found that more than two-thirds of dermatologists (69.3%) agreed it “seems doable” to check lipids and calculate a 10-year cardiovascular risk score, and over one-third (36.1%) agreed they could prescribe statins when indicated.
The patient survey was distributed through the National Psoriasis Foundation to individuals who were seeing a dermatologist or rheumatologist for psoriatic disease; the clinician survey was distributed through the American Academy of Dermatology to dermatologists who reported caring for patients with psoriasis. (A survey of rheumatologists was similarly conducted, but the number of participants fell short of the needed sample size.)
Most patients surveyed indicated they would be receptive to their dermatologist (or rheumatologist) playing a larger role in screening and managing CVD risk, and that they would be similarly likely to follow recommendations regarding risk screening and management whether the advice came their dermatologist/rheumatologist or from their PCP.
The clinician survey focused on lipids and statin use, and did not address other elements of risk management. Still, the researchers see their findings as an early but promising step in finding better models to improve cardiovascular outcomes for patients with psoriatic disease, who too often do not engage with their PCPs despite their increased risk of CVD and a higher risk of premature mortality from CVD.
Fewer than half of commercially insured adults aged under 65 years visit a PCP each year, the researchers noted. And among the patients in their survey, approximately 20% did not have a PCP or had not seen their PCP in the past year.
Other research has shown that only a small minority of patients with psoriasis have an encounter with their PCP within a year of establishing care with their dermatologist, and that “over half of patients with psoriasis have undetected risk factors like dyslipidemia or hypertension,” Dr. Barbieri, of the department of dermatology at Brigham and Women’s Hospital, Boston, said in an interview.
“There’s a gap here, a missing link in the chain of cardiovascular disease prevention,” he said. “What if the dermatologist or rheumatologist could be more engaged in [CV] risk protection? ... It’s the idea of meeting the patients where they are.”
The surveys
The clinician survey focused on statins because of their ease of use, efficacy and safety, and the need for minimal monitoring, Dr. Barbieri said in the interview. “On the spectrum of things you can do for cardiovascular disease prevention, it’s one of the easiest ones.”
In an accompanying editorial, cardiologists Michael S. Garshick, MD, MS, and Jeffrey S. Berger, MD, MS, both of the department of medicine, New York University, wrote that, “despite the well-described association between psoriasis and CVD, only 35% of patients with psoriasis diagnosed with hyperlipidemia are adequately treated with statin therapy.”
“For many of these patients, their dermatologist or rheumatologist may be their only source of contact with the health care system,” they added.
Most studies targeting CVD risk in psoriasis have focused on targeting psoriatic inflammation, and few studies have explored strategies to improve modifiable CVD risk factor control with pharmacological therapy, they said.
In addition to the questions about receptiveness to identifying and potentially treating CVD risk with statins, the dermatologist survey included a best-worst scaling choice experiment to assess preferences for implementation approaches. Dermatologists were asked to rank their preferences for eight implementation strategies that have been shown in published studies to help increase statin prescribing rates.
The three highest-ranked strategies among dermatologists were clinical decision support, physician educational outreach, and patient education materials. The lowest-ranked strategies were comparisons with peers, a pay-for-performance option, and a mobile app/texting service to remind patients to undergo CVD risk screening.
Of the 183 dermatologists in the survey, 28.4% were from academic settings, 11.5% were from multispecialty groups, and 45.4% were from dermatology groups. (A low response rate of 5.2% for dermatologists raises some questions about the generalizability of the findings, Dr. Garshick and Dr. Berger noted in their editorial.)
Where to go from here?
Asked to comment on the results, Jashin J. Wu, MD, founder and CEO of the Dermatology Research and Education Foundation, Irvine, Calif., who was not involved with the study, said that a larger role in CVD risk management is “not likely to find traction with everyday dermatologists.”
“It’s already a big ask for community dermatologists to go through the approval process to get biologics for patients, so I don’t think many would be willing to add more to their plate by taking a bigger role in CVD management,” he said in an interview. He generally has not prescribed statins, “as I don’t feel that is in my scope of work.”
In the interview, Dr. Barbieri said that a parallel qualitative study, not yet published, has looked at the facilitators and barriers – including time constraints and concern about scope of practice – to statin prescribing and other elements of cardiovascular risk reduction.
All told, he said, a centralized care coordinator model may be the best approach to engage the dermatologist more in CVD prevention, including lipid management, but to also “offload some of the management responsibility.”
In this model, which is partially described by Dr. Barbieri and colleagues, the dermatologist (or rheumatologist) would educate the patient, measure blood pressure and check a lipid panel, and refer the patient to a coordinator who would, in turn, collect more information and calculate a 10-year CVD risk score.
Using a protocol-driven clinical decision support approach, the care coordinator would provide counseling about diet, exercise, and smoking cessation, and about whether statin therapy or blood pressure management is indicated.
“That coordinator would be in a good position to help the patient work with their PCP, if they have one, to find a PCP if they don’t, or to use telemedicine or work with their dermatologist or rheumatologist,” Dr. Barbieri said.
The centralized care coordinator service could be funded through grants, charitable funds, and patient assistance funds so that it is free to patients, he said, and could possibly be “housed in the National Psoriasis Foundation.”
Dr. Barbieri said he and his colleagues plan to design a clinical trial to test whether such a model can be adopted in practice and whether it can improve outcomes associated with CVD risk management.
In their editorial, Dr. Garshick and Dr. Berger, who is director of NYU Langone’s Center for the Prevention of Cardiovascular Disease, wrote that many patients with psoriatic disease have or are at risk for cardiometabolic conditions, and that CVD risk reduction should extend beyond lipid management to include blood pressure, glucose lowering, obesity management, and antiplatelet therapy.
The joint AAD-NPF guidelines for the management and treatment of psoriasis with awareness and attention to comorbidities, published in 2019, were among the first to formally recognize the enhanced CVD risk of patients with psoriasis, they noted.
The guidelines call upon dermatologists to inform patients of the psoriasis-CVD association and ensure their patients are engaged with their PCP or cardiologist for appropriate screening. Now, the editorialists say, “moving the needle forward includes refining and developing modifiable CVD risk reduction strategies for patients with psoriasis, and collaboration between the fields of dermatology, rheumatology, and cardiology is key.”
Incorporating a preventive cardiologist into combined dermatology-rheumatology clinics, or partnering as a freestanding cardioinflammatory clinic, also have potential to improve CVD risk, they wrote.
The survey study was supported by a grant from the NPF Psoriasis Prevention Initiative. Dr. Barbieri reported no conflicts of interest. Several authors disclosed consulting fees and grants from numerous pharmaceutical companies. Dr. Berger reported receiving personal fees from Janssen and grants from AstraZeneca outside of the submitted work. Dr. Garshick reported receiving personal fees from AbbVie outside of the submitted work.
A – may be feasible, given the positive perspectives expressed by both clinicians and patients in a set of electronic surveys, researchers say.
In an analysis of survey responses from 183 dermatologists and 322 patients, John S. Barbieri, MD, MBA, and coinvestigators found that more than two-thirds of dermatologists (69.3%) agreed it “seems doable” to check lipids and calculate a 10-year cardiovascular risk score, and over one-third (36.1%) agreed they could prescribe statins when indicated.
The patient survey was distributed through the National Psoriasis Foundation to individuals who were seeing a dermatologist or rheumatologist for psoriatic disease; the clinician survey was distributed through the American Academy of Dermatology to dermatologists who reported caring for patients with psoriasis. (A survey of rheumatologists was similarly conducted, but the number of participants fell short of the needed sample size.)
Most patients surveyed indicated they would be receptive to their dermatologist (or rheumatologist) playing a larger role in screening and managing CVD risk, and that they would be similarly likely to follow recommendations regarding risk screening and management whether the advice came their dermatologist/rheumatologist or from their PCP.
The clinician survey focused on lipids and statin use, and did not address other elements of risk management. Still, the researchers see their findings as an early but promising step in finding better models to improve cardiovascular outcomes for patients with psoriatic disease, who too often do not engage with their PCPs despite their increased risk of CVD and a higher risk of premature mortality from CVD.
Fewer than half of commercially insured adults aged under 65 years visit a PCP each year, the researchers noted. And among the patients in their survey, approximately 20% did not have a PCP or had not seen their PCP in the past year.
Other research has shown that only a small minority of patients with psoriasis have an encounter with their PCP within a year of establishing care with their dermatologist, and that “over half of patients with psoriasis have undetected risk factors like dyslipidemia or hypertension,” Dr. Barbieri, of the department of dermatology at Brigham and Women’s Hospital, Boston, said in an interview.
“There’s a gap here, a missing link in the chain of cardiovascular disease prevention,” he said. “What if the dermatologist or rheumatologist could be more engaged in [CV] risk protection? ... It’s the idea of meeting the patients where they are.”
The surveys
The clinician survey focused on statins because of their ease of use, efficacy and safety, and the need for minimal monitoring, Dr. Barbieri said in the interview. “On the spectrum of things you can do for cardiovascular disease prevention, it’s one of the easiest ones.”
In an accompanying editorial, cardiologists Michael S. Garshick, MD, MS, and Jeffrey S. Berger, MD, MS, both of the department of medicine, New York University, wrote that, “despite the well-described association between psoriasis and CVD, only 35% of patients with psoriasis diagnosed with hyperlipidemia are adequately treated with statin therapy.”
“For many of these patients, their dermatologist or rheumatologist may be their only source of contact with the health care system,” they added.
Most studies targeting CVD risk in psoriasis have focused on targeting psoriatic inflammation, and few studies have explored strategies to improve modifiable CVD risk factor control with pharmacological therapy, they said.
In addition to the questions about receptiveness to identifying and potentially treating CVD risk with statins, the dermatologist survey included a best-worst scaling choice experiment to assess preferences for implementation approaches. Dermatologists were asked to rank their preferences for eight implementation strategies that have been shown in published studies to help increase statin prescribing rates.
The three highest-ranked strategies among dermatologists were clinical decision support, physician educational outreach, and patient education materials. The lowest-ranked strategies were comparisons with peers, a pay-for-performance option, and a mobile app/texting service to remind patients to undergo CVD risk screening.
Of the 183 dermatologists in the survey, 28.4% were from academic settings, 11.5% were from multispecialty groups, and 45.4% were from dermatology groups. (A low response rate of 5.2% for dermatologists raises some questions about the generalizability of the findings, Dr. Garshick and Dr. Berger noted in their editorial.)
Where to go from here?
Asked to comment on the results, Jashin J. Wu, MD, founder and CEO of the Dermatology Research and Education Foundation, Irvine, Calif., who was not involved with the study, said that a larger role in CVD risk management is “not likely to find traction with everyday dermatologists.”
“It’s already a big ask for community dermatologists to go through the approval process to get biologics for patients, so I don’t think many would be willing to add more to their plate by taking a bigger role in CVD management,” he said in an interview. He generally has not prescribed statins, “as I don’t feel that is in my scope of work.”
In the interview, Dr. Barbieri said that a parallel qualitative study, not yet published, has looked at the facilitators and barriers – including time constraints and concern about scope of practice – to statin prescribing and other elements of cardiovascular risk reduction.
All told, he said, a centralized care coordinator model may be the best approach to engage the dermatologist more in CVD prevention, including lipid management, but to also “offload some of the management responsibility.”
In this model, which is partially described by Dr. Barbieri and colleagues, the dermatologist (or rheumatologist) would educate the patient, measure blood pressure and check a lipid panel, and refer the patient to a coordinator who would, in turn, collect more information and calculate a 10-year CVD risk score.
Using a protocol-driven clinical decision support approach, the care coordinator would provide counseling about diet, exercise, and smoking cessation, and about whether statin therapy or blood pressure management is indicated.
“That coordinator would be in a good position to help the patient work with their PCP, if they have one, to find a PCP if they don’t, or to use telemedicine or work with their dermatologist or rheumatologist,” Dr. Barbieri said.
The centralized care coordinator service could be funded through grants, charitable funds, and patient assistance funds so that it is free to patients, he said, and could possibly be “housed in the National Psoriasis Foundation.”
Dr. Barbieri said he and his colleagues plan to design a clinical trial to test whether such a model can be adopted in practice and whether it can improve outcomes associated with CVD risk management.
In their editorial, Dr. Garshick and Dr. Berger, who is director of NYU Langone’s Center for the Prevention of Cardiovascular Disease, wrote that many patients with psoriatic disease have or are at risk for cardiometabolic conditions, and that CVD risk reduction should extend beyond lipid management to include blood pressure, glucose lowering, obesity management, and antiplatelet therapy.
The joint AAD-NPF guidelines for the management and treatment of psoriasis with awareness and attention to comorbidities, published in 2019, were among the first to formally recognize the enhanced CVD risk of patients with psoriasis, they noted.
The guidelines call upon dermatologists to inform patients of the psoriasis-CVD association and ensure their patients are engaged with their PCP or cardiologist for appropriate screening. Now, the editorialists say, “moving the needle forward includes refining and developing modifiable CVD risk reduction strategies for patients with psoriasis, and collaboration between the fields of dermatology, rheumatology, and cardiology is key.”
Incorporating a preventive cardiologist into combined dermatology-rheumatology clinics, or partnering as a freestanding cardioinflammatory clinic, also have potential to improve CVD risk, they wrote.
The survey study was supported by a grant from the NPF Psoriasis Prevention Initiative. Dr. Barbieri reported no conflicts of interest. Several authors disclosed consulting fees and grants from numerous pharmaceutical companies. Dr. Berger reported receiving personal fees from Janssen and grants from AstraZeneca outside of the submitted work. Dr. Garshick reported receiving personal fees from AbbVie outside of the submitted work.
FROM JAMA DERMATOLOGY
More frequent secukinumab dosing found to benefit overweight psoriasis patients
, results from a multicenter, double-blind, parallel-group trial showed.
The more frequent dosing was also associated with comparable safety, consistent with the established secukinumab safety profile.
“Weight may have an impact on pharmacokinetics and, therefore, on the clinical outcome of biologic treatment for psoriasis,” Matthias Augustin, MD, and colleagues wrote in the study, published recently in the British Journal of Dermatology. “Dose optimization may be highly beneficial for patients with higher body weight,” they noted, adding that their study supports previous study findings and pharmacokinetic/pharmacodynamic modelling data, showing that secukinumab dosed every 2 weeks “leads to a clinically and statistically significant advantage in PASI 90 response,” compared with standard dosing every 4 weeks in patients who weight 90 kg (about 198 pounds) or more, after 16 weeks of treatment, which was maintained until week 52.
For the study, Dr. Augustin, of the Institute for Health Services Research in Dermatology and Nursing at University Medical Center Hamburg-Eppendorf (Germany), and colleagues randomized 331 patients with moderate to severe chronic plaque psoriasis who weighed 90 kg or more to receive secukinumab 300 mg every 2 weeks, or secukinumab 300 mg every 4 weeks. The mean age of the patients was 47 years, 75% were male, 92% were White, and their mean body weight was 111.1 kg, with a mean body mass index of 36.1 kg/m2.
Patients who did not achieve a Psoriasis Area and Severity Index (PASI) 90 at week 16 on the monthly regimen (Q4W) either remained on that regimen or were up-titrated to dosing every 2 weeks (Q2W). Of the 331 patients, 165 received Q2W dosing and 166 received Q4W dosing. The researchers found that, at 16 weeks, patients in the Q2W dosing group had significantly higher PASI 90 responses, compared with those in the Q4W group (73.2% vs. 55.5%, respectively; P = .0003; odds ratio estimate, 2.3).
At 52 weeks, a greater proportion of patients in the Q2W group maintained responses to several outcome measures, compared with those in the Q4W group, including PASI 75 (88.9% vs. 74.8%), PASI 90 (76.4% vs. 52.4%), and PASI 100 (46.7% vs. 27.3%) scores; Investigator’s Global Assessment score of 0 or 1 (75.9% vs. 55.6%); and Dermatology Life Quality Index scores of 0 or 1 (66.1% vs. 48.8%).
In addition, those who had not had a PASI 90 response at week 16 who were up-titrated to Q2W dosing demonstrated higher efficacy responses at week 32, compared with those who remained on the Q4W regimen, with PASI 90 scores of 37.7% versus 16.5%, respectively.
Both regimens were well-tolerated, consistent with the known secukinumab safety profile; safety was comparable in the treatment arms, and there was “no clear dose-response relationship seen” for the incidence of overall adverse events, serious AEs, and AEs leading to discontinuation of the study treatment, “or AEs related to the identified risks” of infections, hypersensitivity, neutropenia and potential risk of major adverse cardiovascular events, the authors wrote.
“Despite more frequent dosing, the incidence of Candida infections was numerically lower in the Q2W group versus the Q4W group,” although there were not many cases, three patients versus six patients, respectively.
Need for individualized treatment
“Despite a decades-long revolution in development of highly efficacious biologic treatments for psoriasis, we are only in the early stages of developing personalized clinical approaches,” said Raj Chovatiya, MD, PhD, a dermatologist at Northwestern University, Chicago, who was asked to comment on the study. “The need for individualized treatment in psoriasis is very real; not every patient may respond to therapy in the same way. Obesity is one important comorbidity of psoriasis, and increased body mass index may be associated with variable treatment outcomes with systemic therapy.”
The data from this study, he added, “suggest that dose optimization may be an important strategy to enhance psoriasis clearance in patients with suboptimal treatment outcomes on standard dosing, including those with increased weight. Future studies should examine optimal regimen of biologic therapy across a variety of patient factors.”
The study was funded by Novartis, the manufacturer of secukinumab (Cosentyx); several authors were company employees. Dr. Augustin disclosed that he has served as a consultant for or has been a paid speaker for clinical trials sponsored by companies that manufacture drugs used for the treatment of psoriasis, including AbbVie, Almirall, Amgen, Biogen, Boehringer Ingelheim, Celgene, Centocor, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, LEO Pharma, Medac, Merck, MSD, Novartis, Pfizer, UCB, and Xenoport. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arcutis, Arena, Incyte, Pfizer, Regeneron, and Sanofi Genzyme.
, results from a multicenter, double-blind, parallel-group trial showed.
The more frequent dosing was also associated with comparable safety, consistent with the established secukinumab safety profile.
“Weight may have an impact on pharmacokinetics and, therefore, on the clinical outcome of biologic treatment for psoriasis,” Matthias Augustin, MD, and colleagues wrote in the study, published recently in the British Journal of Dermatology. “Dose optimization may be highly beneficial for patients with higher body weight,” they noted, adding that their study supports previous study findings and pharmacokinetic/pharmacodynamic modelling data, showing that secukinumab dosed every 2 weeks “leads to a clinically and statistically significant advantage in PASI 90 response,” compared with standard dosing every 4 weeks in patients who weight 90 kg (about 198 pounds) or more, after 16 weeks of treatment, which was maintained until week 52.
For the study, Dr. Augustin, of the Institute for Health Services Research in Dermatology and Nursing at University Medical Center Hamburg-Eppendorf (Germany), and colleagues randomized 331 patients with moderate to severe chronic plaque psoriasis who weighed 90 kg or more to receive secukinumab 300 mg every 2 weeks, or secukinumab 300 mg every 4 weeks. The mean age of the patients was 47 years, 75% were male, 92% were White, and their mean body weight was 111.1 kg, with a mean body mass index of 36.1 kg/m2.
Patients who did not achieve a Psoriasis Area and Severity Index (PASI) 90 at week 16 on the monthly regimen (Q4W) either remained on that regimen or were up-titrated to dosing every 2 weeks (Q2W). Of the 331 patients, 165 received Q2W dosing and 166 received Q4W dosing. The researchers found that, at 16 weeks, patients in the Q2W dosing group had significantly higher PASI 90 responses, compared with those in the Q4W group (73.2% vs. 55.5%, respectively; P = .0003; odds ratio estimate, 2.3).
At 52 weeks, a greater proportion of patients in the Q2W group maintained responses to several outcome measures, compared with those in the Q4W group, including PASI 75 (88.9% vs. 74.8%), PASI 90 (76.4% vs. 52.4%), and PASI 100 (46.7% vs. 27.3%) scores; Investigator’s Global Assessment score of 0 or 1 (75.9% vs. 55.6%); and Dermatology Life Quality Index scores of 0 or 1 (66.1% vs. 48.8%).
In addition, those who had not had a PASI 90 response at week 16 who were up-titrated to Q2W dosing demonstrated higher efficacy responses at week 32, compared with those who remained on the Q4W regimen, with PASI 90 scores of 37.7% versus 16.5%, respectively.
Both regimens were well-tolerated, consistent with the known secukinumab safety profile; safety was comparable in the treatment arms, and there was “no clear dose-response relationship seen” for the incidence of overall adverse events, serious AEs, and AEs leading to discontinuation of the study treatment, “or AEs related to the identified risks” of infections, hypersensitivity, neutropenia and potential risk of major adverse cardiovascular events, the authors wrote.
“Despite more frequent dosing, the incidence of Candida infections was numerically lower in the Q2W group versus the Q4W group,” although there were not many cases, three patients versus six patients, respectively.
Need for individualized treatment
“Despite a decades-long revolution in development of highly efficacious biologic treatments for psoriasis, we are only in the early stages of developing personalized clinical approaches,” said Raj Chovatiya, MD, PhD, a dermatologist at Northwestern University, Chicago, who was asked to comment on the study. “The need for individualized treatment in psoriasis is very real; not every patient may respond to therapy in the same way. Obesity is one important comorbidity of psoriasis, and increased body mass index may be associated with variable treatment outcomes with systemic therapy.”
The data from this study, he added, “suggest that dose optimization may be an important strategy to enhance psoriasis clearance in patients with suboptimal treatment outcomes on standard dosing, including those with increased weight. Future studies should examine optimal regimen of biologic therapy across a variety of patient factors.”
The study was funded by Novartis, the manufacturer of secukinumab (Cosentyx); several authors were company employees. Dr. Augustin disclosed that he has served as a consultant for or has been a paid speaker for clinical trials sponsored by companies that manufacture drugs used for the treatment of psoriasis, including AbbVie, Almirall, Amgen, Biogen, Boehringer Ingelheim, Celgene, Centocor, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, LEO Pharma, Medac, Merck, MSD, Novartis, Pfizer, UCB, and Xenoport. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arcutis, Arena, Incyte, Pfizer, Regeneron, and Sanofi Genzyme.
, results from a multicenter, double-blind, parallel-group trial showed.
The more frequent dosing was also associated with comparable safety, consistent with the established secukinumab safety profile.
“Weight may have an impact on pharmacokinetics and, therefore, on the clinical outcome of biologic treatment for psoriasis,” Matthias Augustin, MD, and colleagues wrote in the study, published recently in the British Journal of Dermatology. “Dose optimization may be highly beneficial for patients with higher body weight,” they noted, adding that their study supports previous study findings and pharmacokinetic/pharmacodynamic modelling data, showing that secukinumab dosed every 2 weeks “leads to a clinically and statistically significant advantage in PASI 90 response,” compared with standard dosing every 4 weeks in patients who weight 90 kg (about 198 pounds) or more, after 16 weeks of treatment, which was maintained until week 52.
For the study, Dr. Augustin, of the Institute for Health Services Research in Dermatology and Nursing at University Medical Center Hamburg-Eppendorf (Germany), and colleagues randomized 331 patients with moderate to severe chronic plaque psoriasis who weighed 90 kg or more to receive secukinumab 300 mg every 2 weeks, or secukinumab 300 mg every 4 weeks. The mean age of the patients was 47 years, 75% were male, 92% were White, and their mean body weight was 111.1 kg, with a mean body mass index of 36.1 kg/m2.
Patients who did not achieve a Psoriasis Area and Severity Index (PASI) 90 at week 16 on the monthly regimen (Q4W) either remained on that regimen or were up-titrated to dosing every 2 weeks (Q2W). Of the 331 patients, 165 received Q2W dosing and 166 received Q4W dosing. The researchers found that, at 16 weeks, patients in the Q2W dosing group had significantly higher PASI 90 responses, compared with those in the Q4W group (73.2% vs. 55.5%, respectively; P = .0003; odds ratio estimate, 2.3).
At 52 weeks, a greater proportion of patients in the Q2W group maintained responses to several outcome measures, compared with those in the Q4W group, including PASI 75 (88.9% vs. 74.8%), PASI 90 (76.4% vs. 52.4%), and PASI 100 (46.7% vs. 27.3%) scores; Investigator’s Global Assessment score of 0 or 1 (75.9% vs. 55.6%); and Dermatology Life Quality Index scores of 0 or 1 (66.1% vs. 48.8%).
In addition, those who had not had a PASI 90 response at week 16 who were up-titrated to Q2W dosing demonstrated higher efficacy responses at week 32, compared with those who remained on the Q4W regimen, with PASI 90 scores of 37.7% versus 16.5%, respectively.
Both regimens were well-tolerated, consistent with the known secukinumab safety profile; safety was comparable in the treatment arms, and there was “no clear dose-response relationship seen” for the incidence of overall adverse events, serious AEs, and AEs leading to discontinuation of the study treatment, “or AEs related to the identified risks” of infections, hypersensitivity, neutropenia and potential risk of major adverse cardiovascular events, the authors wrote.
“Despite more frequent dosing, the incidence of Candida infections was numerically lower in the Q2W group versus the Q4W group,” although there were not many cases, three patients versus six patients, respectively.
Need for individualized treatment
“Despite a decades-long revolution in development of highly efficacious biologic treatments for psoriasis, we are only in the early stages of developing personalized clinical approaches,” said Raj Chovatiya, MD, PhD, a dermatologist at Northwestern University, Chicago, who was asked to comment on the study. “The need for individualized treatment in psoriasis is very real; not every patient may respond to therapy in the same way. Obesity is one important comorbidity of psoriasis, and increased body mass index may be associated with variable treatment outcomes with systemic therapy.”
The data from this study, he added, “suggest that dose optimization may be an important strategy to enhance psoriasis clearance in patients with suboptimal treatment outcomes on standard dosing, including those with increased weight. Future studies should examine optimal regimen of biologic therapy across a variety of patient factors.”
The study was funded by Novartis, the manufacturer of secukinumab (Cosentyx); several authors were company employees. Dr. Augustin disclosed that he has served as a consultant for or has been a paid speaker for clinical trials sponsored by companies that manufacture drugs used for the treatment of psoriasis, including AbbVie, Almirall, Amgen, Biogen, Boehringer Ingelheim, Celgene, Centocor, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, LEO Pharma, Medac, Merck, MSD, Novartis, Pfizer, UCB, and Xenoport. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arcutis, Arena, Incyte, Pfizer, Regeneron, and Sanofi Genzyme.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Proactive infliximab monitoring found best for sustaining control of inflammatory diseases
A new study has found that proactive therapeutic drug monitoring (TDM) with maintenance infliximab is more effective than standard therapy in sustaining control of immune-mediated inflammatory diseases.
The findings from the Norwegian Drug Monitoring B (NOR-DRUM B) trial, published Dec. 21, 2021, in JAMA, provide greater support to the usefulness of TDM in proactively monitoring serum drug levels and antidrug antibodies to infliximab, which has been previously shown to have benefit in patients with inflammatory bowel disease, but leave the benefits of proactive versus reactive monitoring and the cost-effectiveness of the approach in individual immune-mediated inflammatory diseases still open to questioning.
TDM is ‘not the holy grail,’ and that’s OK
“This is an important milestone in the field of TDM with biologics for immunoinflammatory diseases,” Niels Vande Casteele, PharmD, PhD, of the University of California, San Diego, told this news organization. He was not involved in the study.
“When you read through the study, you can see the authors used the TAXIT trial results to inform their study design and the sample size,” he added, referencing his 2015 study on infliximab guide dosing for patients with inflammatory bowel disease, “the first-ever randomized, controlled trial of proactive TDM with any biologic.”
For the TAXIT study’s primary outcome of clinical and biochemical remission at 1 year, “continued concentration-based dosing was not superior to clinically based dosing for achieving remission.” But in regard to their secondary outcome of sustained remission, their results were quite similar to the results of NOR-DRUM B.
“If anything, we already showed a benefit of proactive TDM in 2015,” he said, “but I’m very glad that the authors looked at the trial design and teased out where TDM could be the most important and have the biggest impact, which is to maintain that sustained disease remission over a prolonged period.”
As for next steps, Dr. Vande Casteele noted that TDM isn’t a one-size-fits-all upgrade for drug treatments. But that doesn’t mean it won’t be very useful in many patients.
“What the paper is saying, and what we’ve been finding all along, is that TDM is not the holy grail,” he said. “But it is a tool in the physicians’ toolbox to optimize treatments and maximize efficacy, and there are some patients who truly benefit from it.”
Study details
To determine if proactive TDM with infliximab led to more sustained disease control than standard therapy, first author Silje Watterdal Syversen, MD, PhD, of Diakonhjemmet Hospital in Oslo, and coauthors conducted a 52-week, randomized, parallel-group, open-label trial. From 20 Norwegian hospitals, they recruited 458 patients with rheumatoid arthritis (n = 80), spondyloarthritis (n = 138), psoriatic arthritis (n = 54), ulcerative colitis (n = 81), Crohn’s disease (n = 68), or psoriasis (n = 37) who were undergoing maintenance therapy with the biologic.
The 454 patients who received at least one randomly allocated dose of infliximab were treated with one of two strategies: TDM (n = 227) or standard therapy (n = 227). The TDM group received dose and interval adjustments based on an algorithm that factored in serum drug levels and antidrug antibodies. The standard therapy group was treated on the basis of clinical judgment and physician discretion. The average age across groups was roughly 45 years, and just under 50% were women.
Overall, sustained disease control without worsening was achieved in 167 patients (73.6%) in the TDM group and 127 patients (55.9%) in the standard therapy group, with an estimated adjusted difference of 17.6% (95% confidence interval, 9.0%-26.2%; P < .001). The estimated hazard ratio of disease worsening was 2.1 (95% CI, 1.5-2.9) for standard therapy, compared with TDM. A total of 27 patients (15%) in the standard therapy group and 21 patients (9.2%) in the TDM group developed significant levels of antidrug antibodies, defined here as 50 mcg/L or more.
A total of 34 patients discontinued infliximab in each group; in the TDM group, most discontinued because of antidrug antibody formation, while the main reason for discontinuing in the standard therapy group was disease worsening. Adverse events were reported in 137 patients (60%) in the TDM group and 142 patients (63%) in the standard therapy group.
Removing barriers to TDM
It’s not clear that proactive TDM will benefit treatment with all biologic disease-modifying antirheumatic drugs (bDMARDs), but the findings from Dr. Syversen and colleagues state the clear value of using drug monitoring to guide maintenance therapy with infliximab, Zachary S. Wallace, MD, and Jeffrey A. Sparks, MD, wrote in an accompanying editorial.
“The relatively large sample size and rigorous study design ... helped to overcome some limitations of previous observational studies and small clinical trials that yielded conflicting results regarding TDM,” they added, noting that these findings contrasted somewhat with the NOR-DRUM A trial in which TDM did not improve remission induction in patients initiating infliximab therapy.
Along those lines, they recognized that TDM appears to have a greater effect in patients on maintenance infliximab, compared with those just starting the drug, surmising – among several explanations – that achieving remission in someone beginning treatment is a more difficult outcome to achieve than controlling disease in a patient already in remission.
For now, more clinical trials assessing specific diseases and involving other bDMARDs are needed; Dr. Wallace and Dr. Sparks stated that it’s time to remove barriers to implementing TDM – including the need for medical insurance preauthorization before increasing drug doses – and potentially “introduce a new era in treatment approach to maintenance therapy for patients with immune-mediated inflammatory diseases.”
The authors acknowledged their study’s limitations, including disease worsening being measured in part by patient-physician consensus and thus potentially subject to bias. In addition, they did not have the statistical ability to test TDM effectiveness in each of the six disease groups, noting that “these diseases have inherent differences, and findings may not be completely generalizable across groups.”
The study was funded by grants from the Norwegian Regional Health Authorities and the South-Eastern Norway Regional Health Authorities. The authors reported numerous potential conflicts of interest, including receiving personal fees and grants from various pharmaceutical companies. Dr. Wallace and Dr. Sparks also reported receiving research support and fees from pharmaceutical companies. Dr. Vande Casteele reported receiving research grants and personal fees from multiple pharmaceutical companies, all outside of the reviewed work.
A version of this article first appeared on Medscape.com.
A new study has found that proactive therapeutic drug monitoring (TDM) with maintenance infliximab is more effective than standard therapy in sustaining control of immune-mediated inflammatory diseases.
The findings from the Norwegian Drug Monitoring B (NOR-DRUM B) trial, published Dec. 21, 2021, in JAMA, provide greater support to the usefulness of TDM in proactively monitoring serum drug levels and antidrug antibodies to infliximab, which has been previously shown to have benefit in patients with inflammatory bowel disease, but leave the benefits of proactive versus reactive monitoring and the cost-effectiveness of the approach in individual immune-mediated inflammatory diseases still open to questioning.
TDM is ‘not the holy grail,’ and that’s OK
“This is an important milestone in the field of TDM with biologics for immunoinflammatory diseases,” Niels Vande Casteele, PharmD, PhD, of the University of California, San Diego, told this news organization. He was not involved in the study.
“When you read through the study, you can see the authors used the TAXIT trial results to inform their study design and the sample size,” he added, referencing his 2015 study on infliximab guide dosing for patients with inflammatory bowel disease, “the first-ever randomized, controlled trial of proactive TDM with any biologic.”
For the TAXIT study’s primary outcome of clinical and biochemical remission at 1 year, “continued concentration-based dosing was not superior to clinically based dosing for achieving remission.” But in regard to their secondary outcome of sustained remission, their results were quite similar to the results of NOR-DRUM B.
“If anything, we already showed a benefit of proactive TDM in 2015,” he said, “but I’m very glad that the authors looked at the trial design and teased out where TDM could be the most important and have the biggest impact, which is to maintain that sustained disease remission over a prolonged period.”
As for next steps, Dr. Vande Casteele noted that TDM isn’t a one-size-fits-all upgrade for drug treatments. But that doesn’t mean it won’t be very useful in many patients.
“What the paper is saying, and what we’ve been finding all along, is that TDM is not the holy grail,” he said. “But it is a tool in the physicians’ toolbox to optimize treatments and maximize efficacy, and there are some patients who truly benefit from it.”
Study details
To determine if proactive TDM with infliximab led to more sustained disease control than standard therapy, first author Silje Watterdal Syversen, MD, PhD, of Diakonhjemmet Hospital in Oslo, and coauthors conducted a 52-week, randomized, parallel-group, open-label trial. From 20 Norwegian hospitals, they recruited 458 patients with rheumatoid arthritis (n = 80), spondyloarthritis (n = 138), psoriatic arthritis (n = 54), ulcerative colitis (n = 81), Crohn’s disease (n = 68), or psoriasis (n = 37) who were undergoing maintenance therapy with the biologic.
The 454 patients who received at least one randomly allocated dose of infliximab were treated with one of two strategies: TDM (n = 227) or standard therapy (n = 227). The TDM group received dose and interval adjustments based on an algorithm that factored in serum drug levels and antidrug antibodies. The standard therapy group was treated on the basis of clinical judgment and physician discretion. The average age across groups was roughly 45 years, and just under 50% were women.
Overall, sustained disease control without worsening was achieved in 167 patients (73.6%) in the TDM group and 127 patients (55.9%) in the standard therapy group, with an estimated adjusted difference of 17.6% (95% confidence interval, 9.0%-26.2%; P < .001). The estimated hazard ratio of disease worsening was 2.1 (95% CI, 1.5-2.9) for standard therapy, compared with TDM. A total of 27 patients (15%) in the standard therapy group and 21 patients (9.2%) in the TDM group developed significant levels of antidrug antibodies, defined here as 50 mcg/L or more.
A total of 34 patients discontinued infliximab in each group; in the TDM group, most discontinued because of antidrug antibody formation, while the main reason for discontinuing in the standard therapy group was disease worsening. Adverse events were reported in 137 patients (60%) in the TDM group and 142 patients (63%) in the standard therapy group.
Removing barriers to TDM
It’s not clear that proactive TDM will benefit treatment with all biologic disease-modifying antirheumatic drugs (bDMARDs), but the findings from Dr. Syversen and colleagues state the clear value of using drug monitoring to guide maintenance therapy with infliximab, Zachary S. Wallace, MD, and Jeffrey A. Sparks, MD, wrote in an accompanying editorial.
“The relatively large sample size and rigorous study design ... helped to overcome some limitations of previous observational studies and small clinical trials that yielded conflicting results regarding TDM,” they added, noting that these findings contrasted somewhat with the NOR-DRUM A trial in which TDM did not improve remission induction in patients initiating infliximab therapy.
Along those lines, they recognized that TDM appears to have a greater effect in patients on maintenance infliximab, compared with those just starting the drug, surmising – among several explanations – that achieving remission in someone beginning treatment is a more difficult outcome to achieve than controlling disease in a patient already in remission.
For now, more clinical trials assessing specific diseases and involving other bDMARDs are needed; Dr. Wallace and Dr. Sparks stated that it’s time to remove barriers to implementing TDM – including the need for medical insurance preauthorization before increasing drug doses – and potentially “introduce a new era in treatment approach to maintenance therapy for patients with immune-mediated inflammatory diseases.”
The authors acknowledged their study’s limitations, including disease worsening being measured in part by patient-physician consensus and thus potentially subject to bias. In addition, they did not have the statistical ability to test TDM effectiveness in each of the six disease groups, noting that “these diseases have inherent differences, and findings may not be completely generalizable across groups.”
The study was funded by grants from the Norwegian Regional Health Authorities and the South-Eastern Norway Regional Health Authorities. The authors reported numerous potential conflicts of interest, including receiving personal fees and grants from various pharmaceutical companies. Dr. Wallace and Dr. Sparks also reported receiving research support and fees from pharmaceutical companies. Dr. Vande Casteele reported receiving research grants and personal fees from multiple pharmaceutical companies, all outside of the reviewed work.
A version of this article first appeared on Medscape.com.
A new study has found that proactive therapeutic drug monitoring (TDM) with maintenance infliximab is more effective than standard therapy in sustaining control of immune-mediated inflammatory diseases.
The findings from the Norwegian Drug Monitoring B (NOR-DRUM B) trial, published Dec. 21, 2021, in JAMA, provide greater support to the usefulness of TDM in proactively monitoring serum drug levels and antidrug antibodies to infliximab, which has been previously shown to have benefit in patients with inflammatory bowel disease, but leave the benefits of proactive versus reactive monitoring and the cost-effectiveness of the approach in individual immune-mediated inflammatory diseases still open to questioning.
TDM is ‘not the holy grail,’ and that’s OK
“This is an important milestone in the field of TDM with biologics for immunoinflammatory diseases,” Niels Vande Casteele, PharmD, PhD, of the University of California, San Diego, told this news organization. He was not involved in the study.
“When you read through the study, you can see the authors used the TAXIT trial results to inform their study design and the sample size,” he added, referencing his 2015 study on infliximab guide dosing for patients with inflammatory bowel disease, “the first-ever randomized, controlled trial of proactive TDM with any biologic.”
For the TAXIT study’s primary outcome of clinical and biochemical remission at 1 year, “continued concentration-based dosing was not superior to clinically based dosing for achieving remission.” But in regard to their secondary outcome of sustained remission, their results were quite similar to the results of NOR-DRUM B.
“If anything, we already showed a benefit of proactive TDM in 2015,” he said, “but I’m very glad that the authors looked at the trial design and teased out where TDM could be the most important and have the biggest impact, which is to maintain that sustained disease remission over a prolonged period.”
As for next steps, Dr. Vande Casteele noted that TDM isn’t a one-size-fits-all upgrade for drug treatments. But that doesn’t mean it won’t be very useful in many patients.
“What the paper is saying, and what we’ve been finding all along, is that TDM is not the holy grail,” he said. “But it is a tool in the physicians’ toolbox to optimize treatments and maximize efficacy, and there are some patients who truly benefit from it.”
Study details
To determine if proactive TDM with infliximab led to more sustained disease control than standard therapy, first author Silje Watterdal Syversen, MD, PhD, of Diakonhjemmet Hospital in Oslo, and coauthors conducted a 52-week, randomized, parallel-group, open-label trial. From 20 Norwegian hospitals, they recruited 458 patients with rheumatoid arthritis (n = 80), spondyloarthritis (n = 138), psoriatic arthritis (n = 54), ulcerative colitis (n = 81), Crohn’s disease (n = 68), or psoriasis (n = 37) who were undergoing maintenance therapy with the biologic.
The 454 patients who received at least one randomly allocated dose of infliximab were treated with one of two strategies: TDM (n = 227) or standard therapy (n = 227). The TDM group received dose and interval adjustments based on an algorithm that factored in serum drug levels and antidrug antibodies. The standard therapy group was treated on the basis of clinical judgment and physician discretion. The average age across groups was roughly 45 years, and just under 50% were women.
Overall, sustained disease control without worsening was achieved in 167 patients (73.6%) in the TDM group and 127 patients (55.9%) in the standard therapy group, with an estimated adjusted difference of 17.6% (95% confidence interval, 9.0%-26.2%; P < .001). The estimated hazard ratio of disease worsening was 2.1 (95% CI, 1.5-2.9) for standard therapy, compared with TDM. A total of 27 patients (15%) in the standard therapy group and 21 patients (9.2%) in the TDM group developed significant levels of antidrug antibodies, defined here as 50 mcg/L or more.
A total of 34 patients discontinued infliximab in each group; in the TDM group, most discontinued because of antidrug antibody formation, while the main reason for discontinuing in the standard therapy group was disease worsening. Adverse events were reported in 137 patients (60%) in the TDM group and 142 patients (63%) in the standard therapy group.
Removing barriers to TDM
It’s not clear that proactive TDM will benefit treatment with all biologic disease-modifying antirheumatic drugs (bDMARDs), but the findings from Dr. Syversen and colleagues state the clear value of using drug monitoring to guide maintenance therapy with infliximab, Zachary S. Wallace, MD, and Jeffrey A. Sparks, MD, wrote in an accompanying editorial.
“The relatively large sample size and rigorous study design ... helped to overcome some limitations of previous observational studies and small clinical trials that yielded conflicting results regarding TDM,” they added, noting that these findings contrasted somewhat with the NOR-DRUM A trial in which TDM did not improve remission induction in patients initiating infliximab therapy.
Along those lines, they recognized that TDM appears to have a greater effect in patients on maintenance infliximab, compared with those just starting the drug, surmising – among several explanations – that achieving remission in someone beginning treatment is a more difficult outcome to achieve than controlling disease in a patient already in remission.
For now, more clinical trials assessing specific diseases and involving other bDMARDs are needed; Dr. Wallace and Dr. Sparks stated that it’s time to remove barriers to implementing TDM – including the need for medical insurance preauthorization before increasing drug doses – and potentially “introduce a new era in treatment approach to maintenance therapy for patients with immune-mediated inflammatory diseases.”
The authors acknowledged their study’s limitations, including disease worsening being measured in part by patient-physician consensus and thus potentially subject to bias. In addition, they did not have the statistical ability to test TDM effectiveness in each of the six disease groups, noting that “these diseases have inherent differences, and findings may not be completely generalizable across groups.”
The study was funded by grants from the Norwegian Regional Health Authorities and the South-Eastern Norway Regional Health Authorities. The authors reported numerous potential conflicts of interest, including receiving personal fees and grants from various pharmaceutical companies. Dr. Wallace and Dr. Sparks also reported receiving research support and fees from pharmaceutical companies. Dr. Vande Casteele reported receiving research grants and personal fees from multiple pharmaceutical companies, all outside of the reviewed work.
A version of this article first appeared on Medscape.com.
FROM JAMA
Case series show no consensus on treatment for palmoplantar pustulosis, generalized pustular psoriasis
that evaluated the characteristics and course of the disease in patients diagnosed with PPP or GPP.
“These case series confirm the rarity of both generalized pustular psoriasis and palmoplantar pustulosis (PPP) and highlight the persistence of symptoms over time and the lack of effective treatment options available to patients,” Megan H. Noe, MD, MPH, MSCE, first author of both case series and assistant professor of dermatology, Harvard Medical School, and a dermatologist at Brigham and Women’s Hospital, both in Boston, said in an interview. In both studies, she added, “more than 20 different therapies were utilized, demonstrating a lack of consensus regarding effective treatment.”
The two case series were published in JAMA Dermatology.
Palmoplantar pustulosis
In the case series of 197 patients with PPP , data were obtained from a retrospective review at 20 academic dermatology practices in the United States between January 2007 and December 2018. The patients were mostly women (73.6%) who were White (60.9%), with a mean age of 53 years; 38.1% were current smokers, and 27.4% were former smokers, and the mean follow-up time was 22.1 months. About half (48.2%) of patients who presented to their respective centers had skin pain, 19.8% had problems using their hands and feet, 12.7% had arthralgias, and 2% had myalgias. Clinicians who examined these patients found pustules on the palms (80.2%), soles (76.7%), and both palms and soles (59.9%); some nail unit involvement was reported in 10.2%.
Patients were treated with a variety of topical therapies, systemic steroids, systemic anti-infectives, and systemic psoriasis therapies, Dr. Noe and colleagues said. The most common initial treatments included a topical steroid (84.8%), with the vast majority of clinicians using a high-potency topical steroid (153 of 167 patients; 91.6%), or topical therapy only (64.5%).
Other initial treatments used were other types of topical medications in 34 of the patients in the series (17.3%), such as a vitamin D analogue in 27 patients (79.4%); oral systemic treatments such as acitretin in 27 patients (13.7%) or methotrexate in 22 patients (11.2%); narrowband UVB phototherapy in 15 patients (7.7%); systemic steroids in 10 patients (5.1%); or systemic antibiotics in 9 patients (4.6%). Less commonly used were biologic agents like adalimumab, used in 6 patients (3.1%).
The researchers also examined health care utilization in 128 patients and found that 82% had at least one follow-up visit, 31.3% required two to three follow-up visits, and 18.8% had five or more follow-up visits. When adjusted to account for age and sex, there was a decreased risk of requiring five or more healthcare visits per year for women (odds ratio, 0.49; 95% confidence interval, 0.25-0.95)
Generalized pustular psoriasis
Dr. Noe and colleagues also evaluated 95 patients with GPP in a retrospective longitudinal case series of patients treated at 20 academic dermatology practices in the United States between January 2007 and December 2018. As in the PPP group, most patients in the GPP case series were women (70.5%), and over half were White (53.7%); the mean age was 50.3 years old, and the mean follow-up time was 19.8 months. A majority of patients with GPP were never-smokers (52.6%) or former smokers (20%). When patients with GPP initially presented to the study sites, 36.8% were admitted as inpatients, 9.5% presented in the emergency department, and 53.7% presented in an outpatient or ambulatory dermatology setting.
GPP commonly appeared on the trunk and extremities, but was “also reported on the scalp, face, genitals, nail unit, and mucous membranes in a minority of patients,” the researchers said. Overall, 62.1% of patients had skin pain, 26.2% had joint pain, 16.8% reported tachycardia, and 9.5% reported fever. Hypertension, depression, diabetes, chronic kidney disease, and hypothyroidism were common comorbidities of GPP, the researchers noted.
Clinicians reported treating GPP with topical steroids (86.3%) and topical treatments alone (32.3%). Oral systemic treatments such as acitretin (24.2%), cyclosporine (22.1%), and methotrexate (13.7%) were also used, as well as systemic steroids (20%). Other treatments used were narrowband UVB phototherapy (5.3%) and biologic agents like adalimumab (4.2%) and infliximab (4.2%).
For 53 patients with follow-up data of at least 6 months, 19 (35.8%) had been hospitalized because of their symptoms, and 8 patients were hospitalized for further GPP-specific concerns. Patients with GPP had a median 3.2 dermatology visits per year and a maximum of 18 visits. A model that was adjusted for age and sex showed women were at a decreased risk for being admitted to the hospital or emergency department in the follow-up period (odds ratio, 0.19; 95% confidence interval, 0.04-0.83).
PPP and GPP in practice
Sylvia Hsu, MD, professor and chair of the department of dermatology at Temple University, Philadelphia, who was not involved with the research, noted that most dermatologists will see few, if any, cases of PPP and GPP in a year. At her center, she estimated that she sees about one PPP case per week, and one or two cases of GPP a year. In general, she said that her clinical experience matched what was found by the authors of both case series.
For patients with PPP, “I would say the average dermatologist would probably start out with a superpotent topical steroid like clobetasol or halobetasol ointment,” Dr. Hsu said.
If they are not of childbearing age, she added, she would also prescribe acitretin, which she avoids giving to patients of childbearing age because of its teratogenicity. “Acitretin has the reputation that it doesn’t work well or fast for psoriasis. It doesn’t work well or fast for plaque-type psoriasis, but it works well and fast for pustular psoriasis,” she said.
In place of acitretin, Dr. Hsu recommended cyclosporine for a patient of childbearing age as a short-term solution to resolve symptoms before transitioning them to another therapy. “A woman of childbearing age, you put on cyclosporine, you’ve got to transition to something else,” she said. “And so many times you wean them off, the pustular psoriasis comes back because the topical steroid doesn’t work that well.”
One possible option is the interluekin-23 inhibitor guselkumab (approved by the Food and Drug Administration for treating moderate to severe plaque psoriasis and psoriatic arthritis) but cost and effectiveness can be a factor. Although studies have shown efficacy, biologics as treatments for PPP are “hit or miss,” Dr. Hsu said.
Regarding use of systemic therapies, Dr. Hsu cautioned against using them to treat plaque-type psoriasis. “We always learn, don’t use a systemic steroid like prednisone to treat psoriasis because it helps, but it comes back with a vengeance,” she said. “Sometimes when you treat plaque-type psoriasis with prednisone, it could come back with a vengeance, and it can come back as generalized pustular psoriasis.”
For patients with GPP, “you need a quick fix” because of the painful symptoms associated with the disease, Dr. Hsu said. In this case, she recommended cyclosporine and said she would avoid prescribing topical medications. “You’re going to have to give an oral drug because usually when we’re seeing somebody with GPP, they’re either a hospital consult or they just walked in the door,” she said. After prescribing cyclosporine, you would transition to another treatment like a biologic “as quickly as you can” with the knowledge that the biologic “may or may not work.”
New treatment options needed
Commenting on both case series in a related editorial, Edward W. Cowen, MD, MHSc, senior clinician and head of the dermatology consultation service in the dermatology branch of the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Bethesda, Md., said that “much of the clinical presentation of pustular disease remains a mystery,” including why tobacco use is a risk factor for developing pustular psoriasis, and why tumor necrosis factor inhibitors “induce pustular disease in a small number of patients” with psoriasis vulgaris.
“Most importantly, we still do not know if localized and generalized pustular psoriasis all truly represent different variants of the same disease process, and if not, which biologic treatment represents the best option for a given clinical variant,” he wrote.
Dr. Cowen noted that the multi-institutional approach to collecting the retrospective data in these case series could be used as a “basic framework to build on for future clinical trials for rare skin diseases such as pustular psoriasis.”
In the interview, Dr. Noe said that she hoped that the “Pustular Psoriasis in the US Research Group” she and her coauthors created for the case series could help with the development of prospective clinical trials. “For pustular psoriasis and other rare diseases in dermatology, multi-institutional collaborations are necessary to conduct prospective research,” she said.
“While not directly studied in our research, I think it is important to consider the negative impact on quality of life, experienced by patients with pustular psoriasis. In our study, many patients experienced exacerbations of their disease over time, and it is important to consider the impact this has on patients,” she said in the interview. “Continued research on pustular psoriasis is necessary to decrease the negative impact of these diseases on the lives of our patients.”
The case series were funded in part by an institutional grant from Boehringer Ingelheim. The authors report relationships with various pharmaceutical and biopharmaceutical companies, technology companies, medical publishing companies, medical journals, and medical societies with connections to the topic area in the form of serving in roles as a chief medical editor, consultant, data safety monitoring board member, deputy editor, principal investigator, research investigator, scientific adviser, or speaker; or having received grants, honoraria, personal fees, or research funding. Dr. Cowen has no disclosures. Dr. Hsu reports serving on a Boehringer Ingelheim advisory board for a product being evaluated as a potential treatment for GPP.
that evaluated the characteristics and course of the disease in patients diagnosed with PPP or GPP.
“These case series confirm the rarity of both generalized pustular psoriasis and palmoplantar pustulosis (PPP) and highlight the persistence of symptoms over time and the lack of effective treatment options available to patients,” Megan H. Noe, MD, MPH, MSCE, first author of both case series and assistant professor of dermatology, Harvard Medical School, and a dermatologist at Brigham and Women’s Hospital, both in Boston, said in an interview. In both studies, she added, “more than 20 different therapies were utilized, demonstrating a lack of consensus regarding effective treatment.”
The two case series were published in JAMA Dermatology.
Palmoplantar pustulosis
In the case series of 197 patients with PPP , data were obtained from a retrospective review at 20 academic dermatology practices in the United States between January 2007 and December 2018. The patients were mostly women (73.6%) who were White (60.9%), with a mean age of 53 years; 38.1% were current smokers, and 27.4% were former smokers, and the mean follow-up time was 22.1 months. About half (48.2%) of patients who presented to their respective centers had skin pain, 19.8% had problems using their hands and feet, 12.7% had arthralgias, and 2% had myalgias. Clinicians who examined these patients found pustules on the palms (80.2%), soles (76.7%), and both palms and soles (59.9%); some nail unit involvement was reported in 10.2%.
Patients were treated with a variety of topical therapies, systemic steroids, systemic anti-infectives, and systemic psoriasis therapies, Dr. Noe and colleagues said. The most common initial treatments included a topical steroid (84.8%), with the vast majority of clinicians using a high-potency topical steroid (153 of 167 patients; 91.6%), or topical therapy only (64.5%).
Other initial treatments used were other types of topical medications in 34 of the patients in the series (17.3%), such as a vitamin D analogue in 27 patients (79.4%); oral systemic treatments such as acitretin in 27 patients (13.7%) or methotrexate in 22 patients (11.2%); narrowband UVB phototherapy in 15 patients (7.7%); systemic steroids in 10 patients (5.1%); or systemic antibiotics in 9 patients (4.6%). Less commonly used were biologic agents like adalimumab, used in 6 patients (3.1%).
The researchers also examined health care utilization in 128 patients and found that 82% had at least one follow-up visit, 31.3% required two to three follow-up visits, and 18.8% had five or more follow-up visits. When adjusted to account for age and sex, there was a decreased risk of requiring five or more healthcare visits per year for women (odds ratio, 0.49; 95% confidence interval, 0.25-0.95)
Generalized pustular psoriasis
Dr. Noe and colleagues also evaluated 95 patients with GPP in a retrospective longitudinal case series of patients treated at 20 academic dermatology practices in the United States between January 2007 and December 2018. As in the PPP group, most patients in the GPP case series were women (70.5%), and over half were White (53.7%); the mean age was 50.3 years old, and the mean follow-up time was 19.8 months. A majority of patients with GPP were never-smokers (52.6%) or former smokers (20%). When patients with GPP initially presented to the study sites, 36.8% were admitted as inpatients, 9.5% presented in the emergency department, and 53.7% presented in an outpatient or ambulatory dermatology setting.
GPP commonly appeared on the trunk and extremities, but was “also reported on the scalp, face, genitals, nail unit, and mucous membranes in a minority of patients,” the researchers said. Overall, 62.1% of patients had skin pain, 26.2% had joint pain, 16.8% reported tachycardia, and 9.5% reported fever. Hypertension, depression, diabetes, chronic kidney disease, and hypothyroidism were common comorbidities of GPP, the researchers noted.
Clinicians reported treating GPP with topical steroids (86.3%) and topical treatments alone (32.3%). Oral systemic treatments such as acitretin (24.2%), cyclosporine (22.1%), and methotrexate (13.7%) were also used, as well as systemic steroids (20%). Other treatments used were narrowband UVB phototherapy (5.3%) and biologic agents like adalimumab (4.2%) and infliximab (4.2%).
For 53 patients with follow-up data of at least 6 months, 19 (35.8%) had been hospitalized because of their symptoms, and 8 patients were hospitalized for further GPP-specific concerns. Patients with GPP had a median 3.2 dermatology visits per year and a maximum of 18 visits. A model that was adjusted for age and sex showed women were at a decreased risk for being admitted to the hospital or emergency department in the follow-up period (odds ratio, 0.19; 95% confidence interval, 0.04-0.83).
PPP and GPP in practice
Sylvia Hsu, MD, professor and chair of the department of dermatology at Temple University, Philadelphia, who was not involved with the research, noted that most dermatologists will see few, if any, cases of PPP and GPP in a year. At her center, she estimated that she sees about one PPP case per week, and one or two cases of GPP a year. In general, she said that her clinical experience matched what was found by the authors of both case series.
For patients with PPP, “I would say the average dermatologist would probably start out with a superpotent topical steroid like clobetasol or halobetasol ointment,” Dr. Hsu said.
If they are not of childbearing age, she added, she would also prescribe acitretin, which she avoids giving to patients of childbearing age because of its teratogenicity. “Acitretin has the reputation that it doesn’t work well or fast for psoriasis. It doesn’t work well or fast for plaque-type psoriasis, but it works well and fast for pustular psoriasis,” she said.
In place of acitretin, Dr. Hsu recommended cyclosporine for a patient of childbearing age as a short-term solution to resolve symptoms before transitioning them to another therapy. “A woman of childbearing age, you put on cyclosporine, you’ve got to transition to something else,” she said. “And so many times you wean them off, the pustular psoriasis comes back because the topical steroid doesn’t work that well.”
One possible option is the interluekin-23 inhibitor guselkumab (approved by the Food and Drug Administration for treating moderate to severe plaque psoriasis and psoriatic arthritis) but cost and effectiveness can be a factor. Although studies have shown efficacy, biologics as treatments for PPP are “hit or miss,” Dr. Hsu said.
Regarding use of systemic therapies, Dr. Hsu cautioned against using them to treat plaque-type psoriasis. “We always learn, don’t use a systemic steroid like prednisone to treat psoriasis because it helps, but it comes back with a vengeance,” she said. “Sometimes when you treat plaque-type psoriasis with prednisone, it could come back with a vengeance, and it can come back as generalized pustular psoriasis.”
For patients with GPP, “you need a quick fix” because of the painful symptoms associated with the disease, Dr. Hsu said. In this case, she recommended cyclosporine and said she would avoid prescribing topical medications. “You’re going to have to give an oral drug because usually when we’re seeing somebody with GPP, they’re either a hospital consult or they just walked in the door,” she said. After prescribing cyclosporine, you would transition to another treatment like a biologic “as quickly as you can” with the knowledge that the biologic “may or may not work.”
New treatment options needed
Commenting on both case series in a related editorial, Edward W. Cowen, MD, MHSc, senior clinician and head of the dermatology consultation service in the dermatology branch of the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Bethesda, Md., said that “much of the clinical presentation of pustular disease remains a mystery,” including why tobacco use is a risk factor for developing pustular psoriasis, and why tumor necrosis factor inhibitors “induce pustular disease in a small number of patients” with psoriasis vulgaris.
“Most importantly, we still do not know if localized and generalized pustular psoriasis all truly represent different variants of the same disease process, and if not, which biologic treatment represents the best option for a given clinical variant,” he wrote.
Dr. Cowen noted that the multi-institutional approach to collecting the retrospective data in these case series could be used as a “basic framework to build on for future clinical trials for rare skin diseases such as pustular psoriasis.”
In the interview, Dr. Noe said that she hoped that the “Pustular Psoriasis in the US Research Group” she and her coauthors created for the case series could help with the development of prospective clinical trials. “For pustular psoriasis and other rare diseases in dermatology, multi-institutional collaborations are necessary to conduct prospective research,” she said.
“While not directly studied in our research, I think it is important to consider the negative impact on quality of life, experienced by patients with pustular psoriasis. In our study, many patients experienced exacerbations of their disease over time, and it is important to consider the impact this has on patients,” she said in the interview. “Continued research on pustular psoriasis is necessary to decrease the negative impact of these diseases on the lives of our patients.”
The case series were funded in part by an institutional grant from Boehringer Ingelheim. The authors report relationships with various pharmaceutical and biopharmaceutical companies, technology companies, medical publishing companies, medical journals, and medical societies with connections to the topic area in the form of serving in roles as a chief medical editor, consultant, data safety monitoring board member, deputy editor, principal investigator, research investigator, scientific adviser, or speaker; or having received grants, honoraria, personal fees, or research funding. Dr. Cowen has no disclosures. Dr. Hsu reports serving on a Boehringer Ingelheim advisory board for a product being evaluated as a potential treatment for GPP.
that evaluated the characteristics and course of the disease in patients diagnosed with PPP or GPP.
“These case series confirm the rarity of both generalized pustular psoriasis and palmoplantar pustulosis (PPP) and highlight the persistence of symptoms over time and the lack of effective treatment options available to patients,” Megan H. Noe, MD, MPH, MSCE, first author of both case series and assistant professor of dermatology, Harvard Medical School, and a dermatologist at Brigham and Women’s Hospital, both in Boston, said in an interview. In both studies, she added, “more than 20 different therapies were utilized, demonstrating a lack of consensus regarding effective treatment.”
The two case series were published in JAMA Dermatology.
Palmoplantar pustulosis
In the case series of 197 patients with PPP , data were obtained from a retrospective review at 20 academic dermatology practices in the United States between January 2007 and December 2018. The patients were mostly women (73.6%) who were White (60.9%), with a mean age of 53 years; 38.1% were current smokers, and 27.4% were former smokers, and the mean follow-up time was 22.1 months. About half (48.2%) of patients who presented to their respective centers had skin pain, 19.8% had problems using their hands and feet, 12.7% had arthralgias, and 2% had myalgias. Clinicians who examined these patients found pustules on the palms (80.2%), soles (76.7%), and both palms and soles (59.9%); some nail unit involvement was reported in 10.2%.
Patients were treated with a variety of topical therapies, systemic steroids, systemic anti-infectives, and systemic psoriasis therapies, Dr. Noe and colleagues said. The most common initial treatments included a topical steroid (84.8%), with the vast majority of clinicians using a high-potency topical steroid (153 of 167 patients; 91.6%), or topical therapy only (64.5%).
Other initial treatments used were other types of topical medications in 34 of the patients in the series (17.3%), such as a vitamin D analogue in 27 patients (79.4%); oral systemic treatments such as acitretin in 27 patients (13.7%) or methotrexate in 22 patients (11.2%); narrowband UVB phototherapy in 15 patients (7.7%); systemic steroids in 10 patients (5.1%); or systemic antibiotics in 9 patients (4.6%). Less commonly used were biologic agents like adalimumab, used in 6 patients (3.1%).
The researchers also examined health care utilization in 128 patients and found that 82% had at least one follow-up visit, 31.3% required two to three follow-up visits, and 18.8% had five or more follow-up visits. When adjusted to account for age and sex, there was a decreased risk of requiring five or more healthcare visits per year for women (odds ratio, 0.49; 95% confidence interval, 0.25-0.95)
Generalized pustular psoriasis
Dr. Noe and colleagues also evaluated 95 patients with GPP in a retrospective longitudinal case series of patients treated at 20 academic dermatology practices in the United States between January 2007 and December 2018. As in the PPP group, most patients in the GPP case series were women (70.5%), and over half were White (53.7%); the mean age was 50.3 years old, and the mean follow-up time was 19.8 months. A majority of patients with GPP were never-smokers (52.6%) or former smokers (20%). When patients with GPP initially presented to the study sites, 36.8% were admitted as inpatients, 9.5% presented in the emergency department, and 53.7% presented in an outpatient or ambulatory dermatology setting.
GPP commonly appeared on the trunk and extremities, but was “also reported on the scalp, face, genitals, nail unit, and mucous membranes in a minority of patients,” the researchers said. Overall, 62.1% of patients had skin pain, 26.2% had joint pain, 16.8% reported tachycardia, and 9.5% reported fever. Hypertension, depression, diabetes, chronic kidney disease, and hypothyroidism were common comorbidities of GPP, the researchers noted.
Clinicians reported treating GPP with topical steroids (86.3%) and topical treatments alone (32.3%). Oral systemic treatments such as acitretin (24.2%), cyclosporine (22.1%), and methotrexate (13.7%) were also used, as well as systemic steroids (20%). Other treatments used were narrowband UVB phototherapy (5.3%) and biologic agents like adalimumab (4.2%) and infliximab (4.2%).
For 53 patients with follow-up data of at least 6 months, 19 (35.8%) had been hospitalized because of their symptoms, and 8 patients were hospitalized for further GPP-specific concerns. Patients with GPP had a median 3.2 dermatology visits per year and a maximum of 18 visits. A model that was adjusted for age and sex showed women were at a decreased risk for being admitted to the hospital or emergency department in the follow-up period (odds ratio, 0.19; 95% confidence interval, 0.04-0.83).
PPP and GPP in practice
Sylvia Hsu, MD, professor and chair of the department of dermatology at Temple University, Philadelphia, who was not involved with the research, noted that most dermatologists will see few, if any, cases of PPP and GPP in a year. At her center, she estimated that she sees about one PPP case per week, and one or two cases of GPP a year. In general, she said that her clinical experience matched what was found by the authors of both case series.
For patients with PPP, “I would say the average dermatologist would probably start out with a superpotent topical steroid like clobetasol or halobetasol ointment,” Dr. Hsu said.
If they are not of childbearing age, she added, she would also prescribe acitretin, which she avoids giving to patients of childbearing age because of its teratogenicity. “Acitretin has the reputation that it doesn’t work well or fast for psoriasis. It doesn’t work well or fast for plaque-type psoriasis, but it works well and fast for pustular psoriasis,” she said.
In place of acitretin, Dr. Hsu recommended cyclosporine for a patient of childbearing age as a short-term solution to resolve symptoms before transitioning them to another therapy. “A woman of childbearing age, you put on cyclosporine, you’ve got to transition to something else,” she said. “And so many times you wean them off, the pustular psoriasis comes back because the topical steroid doesn’t work that well.”
One possible option is the interluekin-23 inhibitor guselkumab (approved by the Food and Drug Administration for treating moderate to severe plaque psoriasis and psoriatic arthritis) but cost and effectiveness can be a factor. Although studies have shown efficacy, biologics as treatments for PPP are “hit or miss,” Dr. Hsu said.
Regarding use of systemic therapies, Dr. Hsu cautioned against using them to treat plaque-type psoriasis. “We always learn, don’t use a systemic steroid like prednisone to treat psoriasis because it helps, but it comes back with a vengeance,” she said. “Sometimes when you treat plaque-type psoriasis with prednisone, it could come back with a vengeance, and it can come back as generalized pustular psoriasis.”
For patients with GPP, “you need a quick fix” because of the painful symptoms associated with the disease, Dr. Hsu said. In this case, she recommended cyclosporine and said she would avoid prescribing topical medications. “You’re going to have to give an oral drug because usually when we’re seeing somebody with GPP, they’re either a hospital consult or they just walked in the door,” she said. After prescribing cyclosporine, you would transition to another treatment like a biologic “as quickly as you can” with the knowledge that the biologic “may or may not work.”
New treatment options needed
Commenting on both case series in a related editorial, Edward W. Cowen, MD, MHSc, senior clinician and head of the dermatology consultation service in the dermatology branch of the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Bethesda, Md., said that “much of the clinical presentation of pustular disease remains a mystery,” including why tobacco use is a risk factor for developing pustular psoriasis, and why tumor necrosis factor inhibitors “induce pustular disease in a small number of patients” with psoriasis vulgaris.
“Most importantly, we still do not know if localized and generalized pustular psoriasis all truly represent different variants of the same disease process, and if not, which biologic treatment represents the best option for a given clinical variant,” he wrote.
Dr. Cowen noted that the multi-institutional approach to collecting the retrospective data in these case series could be used as a “basic framework to build on for future clinical trials for rare skin diseases such as pustular psoriasis.”
In the interview, Dr. Noe said that she hoped that the “Pustular Psoriasis in the US Research Group” she and her coauthors created for the case series could help with the development of prospective clinical trials. “For pustular psoriasis and other rare diseases in dermatology, multi-institutional collaborations are necessary to conduct prospective research,” she said.
“While not directly studied in our research, I think it is important to consider the negative impact on quality of life, experienced by patients with pustular psoriasis. In our study, many patients experienced exacerbations of their disease over time, and it is important to consider the impact this has on patients,” she said in the interview. “Continued research on pustular psoriasis is necessary to decrease the negative impact of these diseases on the lives of our patients.”
The case series were funded in part by an institutional grant from Boehringer Ingelheim. The authors report relationships with various pharmaceutical and biopharmaceutical companies, technology companies, medical publishing companies, medical journals, and medical societies with connections to the topic area in the form of serving in roles as a chief medical editor, consultant, data safety monitoring board member, deputy editor, principal investigator, research investigator, scientific adviser, or speaker; or having received grants, honoraria, personal fees, or research funding. Dr. Cowen has no disclosures. Dr. Hsu reports serving on a Boehringer Ingelheim advisory board for a product being evaluated as a potential treatment for GPP.
FROM JAMA DERMATOLOGY
FDA to review PDE4-inhibitor roflumilast for psoriasis
The , according to a statement from the manufacturer.
Roflumilast cream (also known as ARQ-151) is a small molecule inhibitor of PDE4, an enzyme that increases proinflammatory mediators and decreases anti-inflammatory mediators. PDE4 is an established treatment target in dermatology: The FDA approved PDE-4 inhibitor crisaborole (Eucrisa) as a topical treatment for mild to moderate atopic dermatitis in 2016, and an oral PDE-4 inhibitor, orismilast, is being studied for the treatment of plaque psoriasis.
Topical roflumilast, if approved, would be the first topical PDE4 inhibitor for psoriasis in particular, according to the Arcutis Biotherapeutics statement. The cream is designed for use on the entire body, including the face and sensitive intertriginous areas.
The NDA is based on data from a pair of phase 3 randomized, double-blind 8-week studies known as DERMIS 1 and DERMIS 2 (Trials of PDE4 Inhibition with Roflumilast for the Management of Plaque Psoriasis” One and Two) and a long-term phase 2b open-label study.
DERMIS 1 and DERMIS 2 were identical multinational, multicenter studies designed to assess the safety and efficacy of 0.3% roflumilast cream. In the studies, roflumilast met its primary endpoint and patients treated with it demonstrated an Investigator Global Assessment (IGA) success rate of 42.4% compared with 6.1% for the vehicle control (P < .0001), and 37.5% compared with 6.9% for the vehicle control (P < .0001), in the DERMIS 1 and 2 trials, respectively, according to Arcutis.
In the phase 2b study, the treatment effect lasted for 52-64 weeks. Roflumilast was well tolerated across the three studies.
Overall, the most common adverse events reported in the studies were diarrhea (3%), headache (2%), insomnia (1%), nausea (1%), upper respiratory tract infections (1%), and urinary tract infections (1%).
Roflumilast also showed statistically significant improvement compared to a vehicle on secondary endpoints including Intertriginous IGA (I-IGA) Success, Psoriasis Area Severity Index-75 (PASI-75), reductions in itch as measured by the Worst Itch-Numerical Rating Scale (WI-NRS), and patient perceptions of symptoms based on the Psoriasis Symptoms Diary (PSD).
The FDA has set a Prescription Drug User Fee Act (PDUFA) target action date of July 29, 2022, according to the manufacturer’s statement. An oral formulation of roflumilast was approved by the FDA in 2011, for reducing the risk of exacerbations of chronic obstructive pulmonary disease (COPD) in patients with severe COPD.
The , according to a statement from the manufacturer.
Roflumilast cream (also known as ARQ-151) is a small molecule inhibitor of PDE4, an enzyme that increases proinflammatory mediators and decreases anti-inflammatory mediators. PDE4 is an established treatment target in dermatology: The FDA approved PDE-4 inhibitor crisaborole (Eucrisa) as a topical treatment for mild to moderate atopic dermatitis in 2016, and an oral PDE-4 inhibitor, orismilast, is being studied for the treatment of plaque psoriasis.
Topical roflumilast, if approved, would be the first topical PDE4 inhibitor for psoriasis in particular, according to the Arcutis Biotherapeutics statement. The cream is designed for use on the entire body, including the face and sensitive intertriginous areas.
The NDA is based on data from a pair of phase 3 randomized, double-blind 8-week studies known as DERMIS 1 and DERMIS 2 (Trials of PDE4 Inhibition with Roflumilast for the Management of Plaque Psoriasis” One and Two) and a long-term phase 2b open-label study.
DERMIS 1 and DERMIS 2 were identical multinational, multicenter studies designed to assess the safety and efficacy of 0.3% roflumilast cream. In the studies, roflumilast met its primary endpoint and patients treated with it demonstrated an Investigator Global Assessment (IGA) success rate of 42.4% compared with 6.1% for the vehicle control (P < .0001), and 37.5% compared with 6.9% for the vehicle control (P < .0001), in the DERMIS 1 and 2 trials, respectively, according to Arcutis.
In the phase 2b study, the treatment effect lasted for 52-64 weeks. Roflumilast was well tolerated across the three studies.
Overall, the most common adverse events reported in the studies were diarrhea (3%), headache (2%), insomnia (1%), nausea (1%), upper respiratory tract infections (1%), and urinary tract infections (1%).
Roflumilast also showed statistically significant improvement compared to a vehicle on secondary endpoints including Intertriginous IGA (I-IGA) Success, Psoriasis Area Severity Index-75 (PASI-75), reductions in itch as measured by the Worst Itch-Numerical Rating Scale (WI-NRS), and patient perceptions of symptoms based on the Psoriasis Symptoms Diary (PSD).
The FDA has set a Prescription Drug User Fee Act (PDUFA) target action date of July 29, 2022, according to the manufacturer’s statement. An oral formulation of roflumilast was approved by the FDA in 2011, for reducing the risk of exacerbations of chronic obstructive pulmonary disease (COPD) in patients with severe COPD.
The , according to a statement from the manufacturer.
Roflumilast cream (also known as ARQ-151) is a small molecule inhibitor of PDE4, an enzyme that increases proinflammatory mediators and decreases anti-inflammatory mediators. PDE4 is an established treatment target in dermatology: The FDA approved PDE-4 inhibitor crisaborole (Eucrisa) as a topical treatment for mild to moderate atopic dermatitis in 2016, and an oral PDE-4 inhibitor, orismilast, is being studied for the treatment of plaque psoriasis.
Topical roflumilast, if approved, would be the first topical PDE4 inhibitor for psoriasis in particular, according to the Arcutis Biotherapeutics statement. The cream is designed for use on the entire body, including the face and sensitive intertriginous areas.
The NDA is based on data from a pair of phase 3 randomized, double-blind 8-week studies known as DERMIS 1 and DERMIS 2 (Trials of PDE4 Inhibition with Roflumilast for the Management of Plaque Psoriasis” One and Two) and a long-term phase 2b open-label study.
DERMIS 1 and DERMIS 2 were identical multinational, multicenter studies designed to assess the safety and efficacy of 0.3% roflumilast cream. In the studies, roflumilast met its primary endpoint and patients treated with it demonstrated an Investigator Global Assessment (IGA) success rate of 42.4% compared with 6.1% for the vehicle control (P < .0001), and 37.5% compared with 6.9% for the vehicle control (P < .0001), in the DERMIS 1 and 2 trials, respectively, according to Arcutis.
In the phase 2b study, the treatment effect lasted for 52-64 weeks. Roflumilast was well tolerated across the three studies.
Overall, the most common adverse events reported in the studies were diarrhea (3%), headache (2%), insomnia (1%), nausea (1%), upper respiratory tract infections (1%), and urinary tract infections (1%).
Roflumilast also showed statistically significant improvement compared to a vehicle on secondary endpoints including Intertriginous IGA (I-IGA) Success, Psoriasis Area Severity Index-75 (PASI-75), reductions in itch as measured by the Worst Itch-Numerical Rating Scale (WI-NRS), and patient perceptions of symptoms based on the Psoriasis Symptoms Diary (PSD).
The FDA has set a Prescription Drug User Fee Act (PDUFA) target action date of July 29, 2022, according to the manufacturer’s statement. An oral formulation of roflumilast was approved by the FDA in 2011, for reducing the risk of exacerbations of chronic obstructive pulmonary disease (COPD) in patients with severe COPD.