User login
Rheumatology trials seem vulnerable to unblinding: Report
Until more is known about the potential for unblinding, clinicians need to keep in mind that patients and physicians could often guess accurately who was getting placebo or active drug, first author Cody Bruggemeyer, MD, a resident at the Medical College of Wisconsin, Milwaukee, said in an interview.
“It’s important that rheumatologists be aware of this potential issue and use their clinical reasoning and their ability to critically assess papers to evaluate the study design” of research on treatments, he said in an interview.
Dr. Bruggemeyer and coauthors at the Medical College of Wisconsin presented their assessment of the potential for unblinding in a Viewpoint article in The Lancet Rheumatology.
A sample of pivotal clinical trials
The authors selected a sample of pivotal studies of 14 commonly prescribed drugs for rheumatic conditions for which double-blind randomized controlled trials (RCTs) that compared the active ingredient with a placebo were available.
The 14 trials involved treatments classified as disease-modifying antirheumatic drugs (DMARDs), some of which were likely to produce side effects that placebos would not mimic, such as injection site and infusion reactions and difference in readings in lab reports, the authors wrote.
In their analysis, Dr. Bruggemeyer and colleagues evaluated discrepancies in the rates of adverse events reported between active drugs and placebos and classified the 14 studies as follows:
- High unblinding risk: Nine studies had a high estimated risk of unblinding, including trials of adalimumab with citrate (Humira), anakinra (Kineret), anifrolumab (Saphnelo), apremilast (Otezla), ixekizumab (Taltz), leflunomide (Arava), methotrexate, risankizumab (Skyrizi) and tofacitinib (Xeljanz).
- Moderate unblinding risk: Three studies had a moderate estimated risk of unblinding, including trials of azathioprine (Imuran), mycophenolate mofetil and tocilizumab (Actemra).
- Low unblinding risk: Two studies had a low estimated risk of unblinding. These involved tests of belimumab (Benlysta) and rituximab (Rituxan).
Many of the effectiveness measurements of treatments used in rheumatology depend on patients’ reports of relief of pain and other disease symptoms. For example, the widely used American College of Rheumatology 20% response for rheumatoid arthritis includes components that rely on patient and physician assessment of disease activity.
Unblinding risk to clinical trial validity
CTs are the highest level of evidence to establish efficacy, because the study design aims to mask whether the experimental treatment is a drug or placebo. In cases where patients and physicians are more likely to correctly detect use of an active drug, there can be biases that skew results toward reports of symptom improvement. Other patients’ views of their treatment may be distorted by accurate guesses that they have been given placebo, Dr. Bruggemeyer and coauthors wrote.
“The degree of these effects cannot be predicted, but they tend to erroneously inflate the perceived benefit of novel interventions,” they wrote.
The consequences of this unblinding may be minimal in cases where there’s a clear difference between the placebo and active drug, they said. As an example, they cited trials of interleukin-23 inhibitors for psoriasis, where skin clearance as measured by the Psoriatic Area and Severity Index 75 differed by more than 50% in absolute terms between the treatment and placebo groups.
But in other cases, there needs to be more attention paid to the potential role of unblinding, they wrote.
“Studies where effect sizes were small, contradictory, or dependent on subgroup analyses might be especially problematic, but commentary rarely reflects this issue or acknowledges the potential influence of unblinding,” they wrote.
In the paper, they call for more analysis of previous trials to look for unreported assessments of unblinding, while also asking that researchers consider surveying participants in future trials to evaluate the degree to which unblinding occurs.
“Advocacy from professional societies and the U.S. Food and Drug Administration itself might be necessary, but in the interim, rheumatologists should assume unblinding has occurred to some degree in most trials,” they wrote.
Unblinding measure needs validation
In an interview, Roy M. Fleischmann, MD, co–medical director of the Metroplex Clinical Research Center in Dallas, raised some objections to the paper. The paper addresses an interesting question about unblinding, but there should have been more work done, such as finding “a measure that is validated that can say whether you’ve been unblinded or not.”
He added that he was surprised the paper on unblinding in rheumatology trials was published in its current form.
“I would have sent it for a major rewrite” if asked to review this paper before publication, said Dr. Fleischmann, who as a reviewer for Lancet Rheumatology. “I would have said: ‘Okay, 90% of this paper is okay, but your gist is not correct.’ It should be: ‘Is this a problem?’ ”
Dr. Fleischmann said he would have recommended a different perspective to the paper. “That is, this could occur. Should we be looking at this, and how would we look at this?”
In the paper, the authors acknowledge their approach has not been validated, “but it highlights the potential effect of idiosyncratic adverse events,” they wrote.
There’s less funding in general for meta-research than for studies involving treatments, so researchers look for approaches that can be handled without requiring significant funding, and much of the research on the quality of research is conducted like this analysis of rheumatology trials, Michael Putman, MD, the corresponding author and is a rheumatologist and an assistant professor at the Medical College of Wisconsin, said in an interview.
“You’re mostly doing on a shoestring budget with yourself and trainees,” he said. Dr. Putman is an associate editor at the journal Rheumatology and also involved in meta-research, or efforts to understand how studies and trials answer questions about how medical treatments work.
In an Aug. 16 tweet, Dr. Putman said this issue of unintentional unblinding with rheumatology trials was something he’d “been ruminating about for awhile; took two all star trainees to push it over the top!”
One of the barriers to funding of meta-research is a tendency for major funding for medical studies to be focused on specific diseases or targets. With meta-research, it may be more difficult to explain how a specific project will advance efforts to treat or prevent a certain disease, Dr. Putman said.
“It’s a little more esoteric and maybe not quite as clear how these projects will move things forward,” Dr. Putman said.
In addition, the nature of meta-research is to question and often be critical of work that’s already been published, adding another hurdle in attempts to secure funding, he said.
Dr. Putman is supported by a Rheumatology Research Foundation Scientist Development Grant, receives research funding related to clinical trials by AbbVie and AstraZeneca, and consulting fees from Novartis. The other authors declared no competing interests.
Until more is known about the potential for unblinding, clinicians need to keep in mind that patients and physicians could often guess accurately who was getting placebo or active drug, first author Cody Bruggemeyer, MD, a resident at the Medical College of Wisconsin, Milwaukee, said in an interview.
“It’s important that rheumatologists be aware of this potential issue and use their clinical reasoning and their ability to critically assess papers to evaluate the study design” of research on treatments, he said in an interview.
Dr. Bruggemeyer and coauthors at the Medical College of Wisconsin presented their assessment of the potential for unblinding in a Viewpoint article in The Lancet Rheumatology.
A sample of pivotal clinical trials
The authors selected a sample of pivotal studies of 14 commonly prescribed drugs for rheumatic conditions for which double-blind randomized controlled trials (RCTs) that compared the active ingredient with a placebo were available.
The 14 trials involved treatments classified as disease-modifying antirheumatic drugs (DMARDs), some of which were likely to produce side effects that placebos would not mimic, such as injection site and infusion reactions and difference in readings in lab reports, the authors wrote.
In their analysis, Dr. Bruggemeyer and colleagues evaluated discrepancies in the rates of adverse events reported between active drugs and placebos and classified the 14 studies as follows:
- High unblinding risk: Nine studies had a high estimated risk of unblinding, including trials of adalimumab with citrate (Humira), anakinra (Kineret), anifrolumab (Saphnelo), apremilast (Otezla), ixekizumab (Taltz), leflunomide (Arava), methotrexate, risankizumab (Skyrizi) and tofacitinib (Xeljanz).
- Moderate unblinding risk: Three studies had a moderate estimated risk of unblinding, including trials of azathioprine (Imuran), mycophenolate mofetil and tocilizumab (Actemra).
- Low unblinding risk: Two studies had a low estimated risk of unblinding. These involved tests of belimumab (Benlysta) and rituximab (Rituxan).
Many of the effectiveness measurements of treatments used in rheumatology depend on patients’ reports of relief of pain and other disease symptoms. For example, the widely used American College of Rheumatology 20% response for rheumatoid arthritis includes components that rely on patient and physician assessment of disease activity.
Unblinding risk to clinical trial validity
CTs are the highest level of evidence to establish efficacy, because the study design aims to mask whether the experimental treatment is a drug or placebo. In cases where patients and physicians are more likely to correctly detect use of an active drug, there can be biases that skew results toward reports of symptom improvement. Other patients’ views of their treatment may be distorted by accurate guesses that they have been given placebo, Dr. Bruggemeyer and coauthors wrote.
“The degree of these effects cannot be predicted, but they tend to erroneously inflate the perceived benefit of novel interventions,” they wrote.
The consequences of this unblinding may be minimal in cases where there’s a clear difference between the placebo and active drug, they said. As an example, they cited trials of interleukin-23 inhibitors for psoriasis, where skin clearance as measured by the Psoriatic Area and Severity Index 75 differed by more than 50% in absolute terms between the treatment and placebo groups.
But in other cases, there needs to be more attention paid to the potential role of unblinding, they wrote.
“Studies where effect sizes were small, contradictory, or dependent on subgroup analyses might be especially problematic, but commentary rarely reflects this issue or acknowledges the potential influence of unblinding,” they wrote.
In the paper, they call for more analysis of previous trials to look for unreported assessments of unblinding, while also asking that researchers consider surveying participants in future trials to evaluate the degree to which unblinding occurs.
“Advocacy from professional societies and the U.S. Food and Drug Administration itself might be necessary, but in the interim, rheumatologists should assume unblinding has occurred to some degree in most trials,” they wrote.
Unblinding measure needs validation
In an interview, Roy M. Fleischmann, MD, co–medical director of the Metroplex Clinical Research Center in Dallas, raised some objections to the paper. The paper addresses an interesting question about unblinding, but there should have been more work done, such as finding “a measure that is validated that can say whether you’ve been unblinded or not.”
He added that he was surprised the paper on unblinding in rheumatology trials was published in its current form.
“I would have sent it for a major rewrite” if asked to review this paper before publication, said Dr. Fleischmann, who as a reviewer for Lancet Rheumatology. “I would have said: ‘Okay, 90% of this paper is okay, but your gist is not correct.’ It should be: ‘Is this a problem?’ ”
Dr. Fleischmann said he would have recommended a different perspective to the paper. “That is, this could occur. Should we be looking at this, and how would we look at this?”
In the paper, the authors acknowledge their approach has not been validated, “but it highlights the potential effect of idiosyncratic adverse events,” they wrote.
There’s less funding in general for meta-research than for studies involving treatments, so researchers look for approaches that can be handled without requiring significant funding, and much of the research on the quality of research is conducted like this analysis of rheumatology trials, Michael Putman, MD, the corresponding author and is a rheumatologist and an assistant professor at the Medical College of Wisconsin, said in an interview.
“You’re mostly doing on a shoestring budget with yourself and trainees,” he said. Dr. Putman is an associate editor at the journal Rheumatology and also involved in meta-research, or efforts to understand how studies and trials answer questions about how medical treatments work.
In an Aug. 16 tweet, Dr. Putman said this issue of unintentional unblinding with rheumatology trials was something he’d “been ruminating about for awhile; took two all star trainees to push it over the top!”
One of the barriers to funding of meta-research is a tendency for major funding for medical studies to be focused on specific diseases or targets. With meta-research, it may be more difficult to explain how a specific project will advance efforts to treat or prevent a certain disease, Dr. Putman said.
“It’s a little more esoteric and maybe not quite as clear how these projects will move things forward,” Dr. Putman said.
In addition, the nature of meta-research is to question and often be critical of work that’s already been published, adding another hurdle in attempts to secure funding, he said.
Dr. Putman is supported by a Rheumatology Research Foundation Scientist Development Grant, receives research funding related to clinical trials by AbbVie and AstraZeneca, and consulting fees from Novartis. The other authors declared no competing interests.
Until more is known about the potential for unblinding, clinicians need to keep in mind that patients and physicians could often guess accurately who was getting placebo or active drug, first author Cody Bruggemeyer, MD, a resident at the Medical College of Wisconsin, Milwaukee, said in an interview.
“It’s important that rheumatologists be aware of this potential issue and use their clinical reasoning and their ability to critically assess papers to evaluate the study design” of research on treatments, he said in an interview.
Dr. Bruggemeyer and coauthors at the Medical College of Wisconsin presented their assessment of the potential for unblinding in a Viewpoint article in The Lancet Rheumatology.
A sample of pivotal clinical trials
The authors selected a sample of pivotal studies of 14 commonly prescribed drugs for rheumatic conditions for which double-blind randomized controlled trials (RCTs) that compared the active ingredient with a placebo were available.
The 14 trials involved treatments classified as disease-modifying antirheumatic drugs (DMARDs), some of which were likely to produce side effects that placebos would not mimic, such as injection site and infusion reactions and difference in readings in lab reports, the authors wrote.
In their analysis, Dr. Bruggemeyer and colleagues evaluated discrepancies in the rates of adverse events reported between active drugs and placebos and classified the 14 studies as follows:
- High unblinding risk: Nine studies had a high estimated risk of unblinding, including trials of adalimumab with citrate (Humira), anakinra (Kineret), anifrolumab (Saphnelo), apremilast (Otezla), ixekizumab (Taltz), leflunomide (Arava), methotrexate, risankizumab (Skyrizi) and tofacitinib (Xeljanz).
- Moderate unblinding risk: Three studies had a moderate estimated risk of unblinding, including trials of azathioprine (Imuran), mycophenolate mofetil and tocilizumab (Actemra).
- Low unblinding risk: Two studies had a low estimated risk of unblinding. These involved tests of belimumab (Benlysta) and rituximab (Rituxan).
Many of the effectiveness measurements of treatments used in rheumatology depend on patients’ reports of relief of pain and other disease symptoms. For example, the widely used American College of Rheumatology 20% response for rheumatoid arthritis includes components that rely on patient and physician assessment of disease activity.
Unblinding risk to clinical trial validity
CTs are the highest level of evidence to establish efficacy, because the study design aims to mask whether the experimental treatment is a drug or placebo. In cases where patients and physicians are more likely to correctly detect use of an active drug, there can be biases that skew results toward reports of symptom improvement. Other patients’ views of their treatment may be distorted by accurate guesses that they have been given placebo, Dr. Bruggemeyer and coauthors wrote.
“The degree of these effects cannot be predicted, but they tend to erroneously inflate the perceived benefit of novel interventions,” they wrote.
The consequences of this unblinding may be minimal in cases where there’s a clear difference between the placebo and active drug, they said. As an example, they cited trials of interleukin-23 inhibitors for psoriasis, where skin clearance as measured by the Psoriatic Area and Severity Index 75 differed by more than 50% in absolute terms between the treatment and placebo groups.
But in other cases, there needs to be more attention paid to the potential role of unblinding, they wrote.
“Studies where effect sizes were small, contradictory, or dependent on subgroup analyses might be especially problematic, but commentary rarely reflects this issue or acknowledges the potential influence of unblinding,” they wrote.
In the paper, they call for more analysis of previous trials to look for unreported assessments of unblinding, while also asking that researchers consider surveying participants in future trials to evaluate the degree to which unblinding occurs.
“Advocacy from professional societies and the U.S. Food and Drug Administration itself might be necessary, but in the interim, rheumatologists should assume unblinding has occurred to some degree in most trials,” they wrote.
Unblinding measure needs validation
In an interview, Roy M. Fleischmann, MD, co–medical director of the Metroplex Clinical Research Center in Dallas, raised some objections to the paper. The paper addresses an interesting question about unblinding, but there should have been more work done, such as finding “a measure that is validated that can say whether you’ve been unblinded or not.”
He added that he was surprised the paper on unblinding in rheumatology trials was published in its current form.
“I would have sent it for a major rewrite” if asked to review this paper before publication, said Dr. Fleischmann, who as a reviewer for Lancet Rheumatology. “I would have said: ‘Okay, 90% of this paper is okay, but your gist is not correct.’ It should be: ‘Is this a problem?’ ”
Dr. Fleischmann said he would have recommended a different perspective to the paper. “That is, this could occur. Should we be looking at this, and how would we look at this?”
In the paper, the authors acknowledge their approach has not been validated, “but it highlights the potential effect of idiosyncratic adverse events,” they wrote.
There’s less funding in general for meta-research than for studies involving treatments, so researchers look for approaches that can be handled without requiring significant funding, and much of the research on the quality of research is conducted like this analysis of rheumatology trials, Michael Putman, MD, the corresponding author and is a rheumatologist and an assistant professor at the Medical College of Wisconsin, said in an interview.
“You’re mostly doing on a shoestring budget with yourself and trainees,” he said. Dr. Putman is an associate editor at the journal Rheumatology and also involved in meta-research, or efforts to understand how studies and trials answer questions about how medical treatments work.
In an Aug. 16 tweet, Dr. Putman said this issue of unintentional unblinding with rheumatology trials was something he’d “been ruminating about for awhile; took two all star trainees to push it over the top!”
One of the barriers to funding of meta-research is a tendency for major funding for medical studies to be focused on specific diseases or targets. With meta-research, it may be more difficult to explain how a specific project will advance efforts to treat or prevent a certain disease, Dr. Putman said.
“It’s a little more esoteric and maybe not quite as clear how these projects will move things forward,” Dr. Putman said.
In addition, the nature of meta-research is to question and often be critical of work that’s already been published, adding another hurdle in attempts to secure funding, he said.
Dr. Putman is supported by a Rheumatology Research Foundation Scientist Development Grant, receives research funding related to clinical trials by AbbVie and AstraZeneca, and consulting fees from Novartis. The other authors declared no competing interests.
FROM THE LANCET RHEUMATOLOGY
Financial Insecurity Among US Adults With Psoriasis
To the Editor:
Approximately 3% of the US population, or 6.9 million adults, is affected by psoriasis.1 Psoriasis has a substantial impact on quality of life and is associated with increased health care expenses and medication costs. In 2013, it was reported that the estimated US annual cost—direct, indirect, intangible, and comorbidity costs—of psoriasis for adults was $112 billion.2 We investigated the prevalence and sociodemographic characteristics of adult psoriasis patients (aged ≥20 years) with financial insecurity utilizing the 2009–2014 National Health and Nutrition Examination Survey (NHANES) data.3
We conducted a population-based, cross-sectional study focused on patients 20 years and older with psoriasis from the 2009-2014 NHANES database to evaluate financial insecurity. Financial insecurity was evaluated by 2 outcome variables. The primary outcome variable was assessed by the question “Are you covered by health insurance or some other kind of health care plan (including health insurance obtained through employment or purchased directly as well as government programs like Medicare and Medicaid that provide medical care or help pay medical bills)?”3 Our secondary outcome variable was evaluated by a reported annual household income of less than $20,000. P values in Table 1 were calculated using Pearson χ2 tests. In Table 2, multivariate logistic regressions were performed using Stata/MP 17 (StataCorp LLC) to analyze associations between outcome variables and sociodemographic characteristics. Additionally, we controlled for age, race/ethnicity, sex, education, marital status, US citizenship status, and tobacco use. Subsequently, relationships with P<.05 were considered statistically significant.
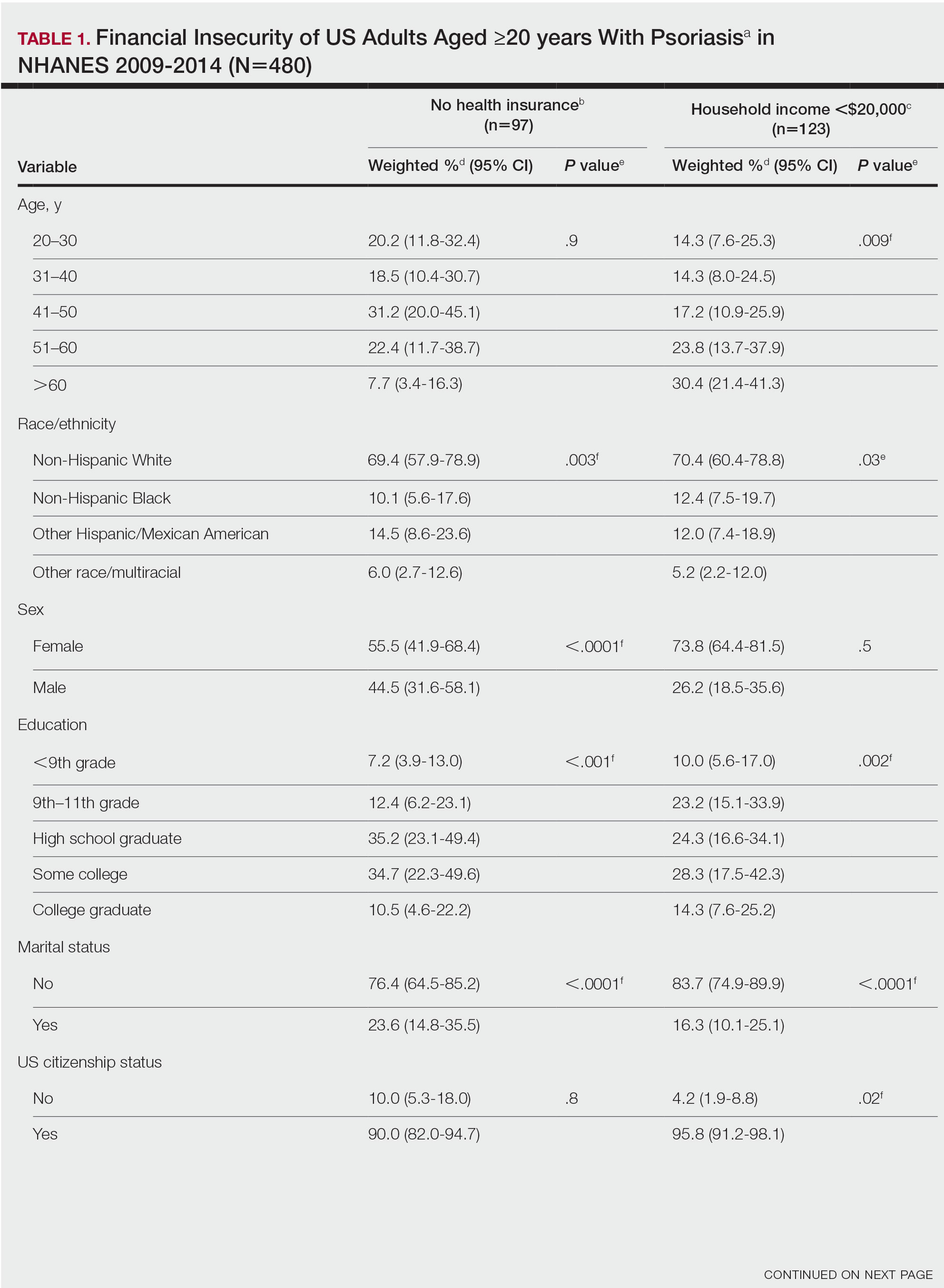

Our analysis comprised 480 individuals with psoriasis; 40 individuals were excluded from our analysis because they did not report annual household income and health insurance status (Table 1). Among the 480 individuals with psoriasis, approximately 16% (weighted) reported a lack of health insurance, and approximately 17% (weighted) reported an annual household income of less than $20,000. Among those who reported an annual household income of less than $20,000, approximately 38% (weighted) of them reported that they did not have health insurance.
Multivariate logistic regression analyses revealed that elderly individuals (aged >60 years), college graduates, married individuals, and US citizens had decreased odds of lacking health insurance (Table 2). Additionally, those with a history of tobacco use (adjusted odds ratio [AOR] 2.02; 95% CI, 1.00-4.05) were associated with lacking health insurance. Non-Hispanic Black individuals (AOR 2.26; 95% CI, 1.09-4.71) and US citizens (AOR 5.01; 95% CI, 1.28-19.63) had a significant association with an annual household income of less than $20,000 (P<.05). Lastly, males, those with education beyond ninth grade, and married individuals had a significantly decreased odds of having an annual household income of less than $20,000 (P<.05)(Table 2).

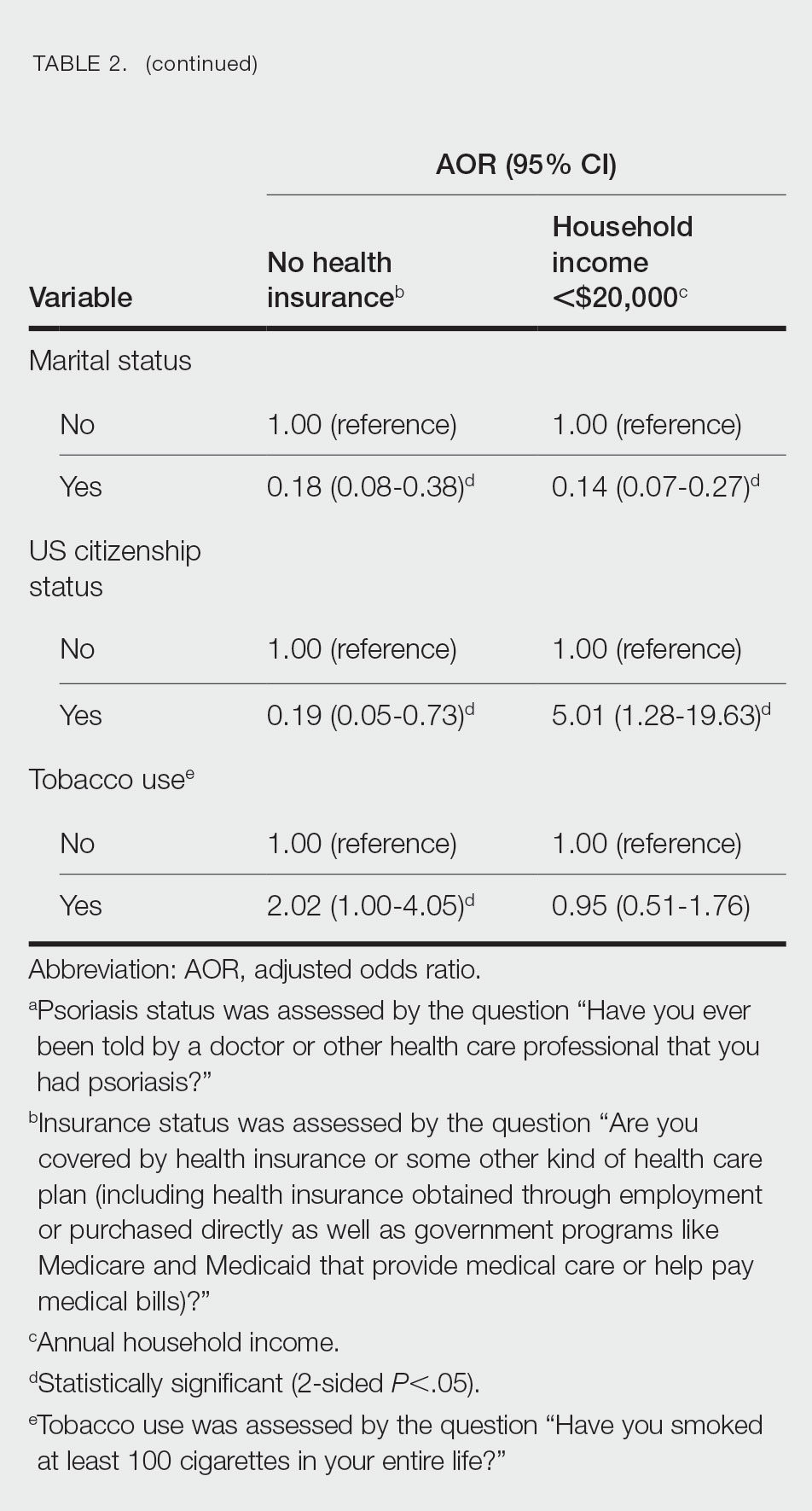
Our findings indicate that certain sociodemographic groups of psoriasis patients have an increased risk for being financially insecure. It is important to evaluate the cost of treatment, number of necessary visits to the office, and cost of transportation, as these factors can serve as a major economic burden to patients being managed for psoriasis.4 Additionally, the cost of biologics has been increasing over time.5 Taking all of this into account when caring for psoriasis patients is crucial, as understanding the financial status of patients can assist with determining appropriate individualized treatment regimens.
- Liu J, Thatiparthi A, Martin A, et al. Prevalence of psoriasis among adults in the US 2009-2010 and 2013-2014 National Health and Nutrition Examination Surveys. J Am Acad Dermatol. 2021;84:767-769. doi:10.1016/j.jaad.2020.10.035
- Brezinski EA, Dhillon JS, Armstrong AW. Economic burden of psoriasis in the United States: a systematic review. JAMA Dermatol. 2015;151:651-658. doi:10.1001/jamadermatol.2014.3593
- National Center for Health Statistics. NHANES questionnaires, datasets, and related documentation. Centers for Disease Control and Prevention website. Accessed June 22, 2023. https://wwwn.cdc.govnchs/nhanes/Default.aspx
- Maya-Rico AM, Londoño-García Á, Palacios-Barahona AU, et al. Out-of-pocket costs for patients with psoriasis in an outpatient dermatology referral service. An Bras Dermatol. 2021;96:295-300. doi:10.1016/j.abd.2020.09.004
- Cheng J, Feldman SR. The cost of biologics for psoriasis is increasing. Drugs Context. 2014;3:212266. doi:10.7573/dic.212266
To the Editor:
Approximately 3% of the US population, or 6.9 million adults, is affected by psoriasis.1 Psoriasis has a substantial impact on quality of life and is associated with increased health care expenses and medication costs. In 2013, it was reported that the estimated US annual cost—direct, indirect, intangible, and comorbidity costs—of psoriasis for adults was $112 billion.2 We investigated the prevalence and sociodemographic characteristics of adult psoriasis patients (aged ≥20 years) with financial insecurity utilizing the 2009–2014 National Health and Nutrition Examination Survey (NHANES) data.3
We conducted a population-based, cross-sectional study focused on patients 20 years and older with psoriasis from the 2009-2014 NHANES database to evaluate financial insecurity. Financial insecurity was evaluated by 2 outcome variables. The primary outcome variable was assessed by the question “Are you covered by health insurance or some other kind of health care plan (including health insurance obtained through employment or purchased directly as well as government programs like Medicare and Medicaid that provide medical care or help pay medical bills)?”3 Our secondary outcome variable was evaluated by a reported annual household income of less than $20,000. P values in Table 1 were calculated using Pearson χ2 tests. In Table 2, multivariate logistic regressions were performed using Stata/MP 17 (StataCorp LLC) to analyze associations between outcome variables and sociodemographic characteristics. Additionally, we controlled for age, race/ethnicity, sex, education, marital status, US citizenship status, and tobacco use. Subsequently, relationships with P<.05 were considered statistically significant.


Our analysis comprised 480 individuals with psoriasis; 40 individuals were excluded from our analysis because they did not report annual household income and health insurance status (Table 1). Among the 480 individuals with psoriasis, approximately 16% (weighted) reported a lack of health insurance, and approximately 17% (weighted) reported an annual household income of less than $20,000. Among those who reported an annual household income of less than $20,000, approximately 38% (weighted) of them reported that they did not have health insurance.
Multivariate logistic regression analyses revealed that elderly individuals (aged >60 years), college graduates, married individuals, and US citizens had decreased odds of lacking health insurance (Table 2). Additionally, those with a history of tobacco use (adjusted odds ratio [AOR] 2.02; 95% CI, 1.00-4.05) were associated with lacking health insurance. Non-Hispanic Black individuals (AOR 2.26; 95% CI, 1.09-4.71) and US citizens (AOR 5.01; 95% CI, 1.28-19.63) had a significant association with an annual household income of less than $20,000 (P<.05). Lastly, males, those with education beyond ninth grade, and married individuals had a significantly decreased odds of having an annual household income of less than $20,000 (P<.05)(Table 2).


Our findings indicate that certain sociodemographic groups of psoriasis patients have an increased risk for being financially insecure. It is important to evaluate the cost of treatment, number of necessary visits to the office, and cost of transportation, as these factors can serve as a major economic burden to patients being managed for psoriasis.4 Additionally, the cost of biologics has been increasing over time.5 Taking all of this into account when caring for psoriasis patients is crucial, as understanding the financial status of patients can assist with determining appropriate individualized treatment regimens.
To the Editor:
Approximately 3% of the US population, or 6.9 million adults, is affected by psoriasis.1 Psoriasis has a substantial impact on quality of life and is associated with increased health care expenses and medication costs. In 2013, it was reported that the estimated US annual cost—direct, indirect, intangible, and comorbidity costs—of psoriasis for adults was $112 billion.2 We investigated the prevalence and sociodemographic characteristics of adult psoriasis patients (aged ≥20 years) with financial insecurity utilizing the 2009–2014 National Health and Nutrition Examination Survey (NHANES) data.3
We conducted a population-based, cross-sectional study focused on patients 20 years and older with psoriasis from the 2009-2014 NHANES database to evaluate financial insecurity. Financial insecurity was evaluated by 2 outcome variables. The primary outcome variable was assessed by the question “Are you covered by health insurance or some other kind of health care plan (including health insurance obtained through employment or purchased directly as well as government programs like Medicare and Medicaid that provide medical care or help pay medical bills)?”3 Our secondary outcome variable was evaluated by a reported annual household income of less than $20,000. P values in Table 1 were calculated using Pearson χ2 tests. In Table 2, multivariate logistic regressions were performed using Stata/MP 17 (StataCorp LLC) to analyze associations between outcome variables and sociodemographic characteristics. Additionally, we controlled for age, race/ethnicity, sex, education, marital status, US citizenship status, and tobacco use. Subsequently, relationships with P<.05 were considered statistically significant.


Our analysis comprised 480 individuals with psoriasis; 40 individuals were excluded from our analysis because they did not report annual household income and health insurance status (Table 1). Among the 480 individuals with psoriasis, approximately 16% (weighted) reported a lack of health insurance, and approximately 17% (weighted) reported an annual household income of less than $20,000. Among those who reported an annual household income of less than $20,000, approximately 38% (weighted) of them reported that they did not have health insurance.
Multivariate logistic regression analyses revealed that elderly individuals (aged >60 years), college graduates, married individuals, and US citizens had decreased odds of lacking health insurance (Table 2). Additionally, those with a history of tobacco use (adjusted odds ratio [AOR] 2.02; 95% CI, 1.00-4.05) were associated with lacking health insurance. Non-Hispanic Black individuals (AOR 2.26; 95% CI, 1.09-4.71) and US citizens (AOR 5.01; 95% CI, 1.28-19.63) had a significant association with an annual household income of less than $20,000 (P<.05). Lastly, males, those with education beyond ninth grade, and married individuals had a significantly decreased odds of having an annual household income of less than $20,000 (P<.05)(Table 2).


Our findings indicate that certain sociodemographic groups of psoriasis patients have an increased risk for being financially insecure. It is important to evaluate the cost of treatment, number of necessary visits to the office, and cost of transportation, as these factors can serve as a major economic burden to patients being managed for psoriasis.4 Additionally, the cost of biologics has been increasing over time.5 Taking all of this into account when caring for psoriasis patients is crucial, as understanding the financial status of patients can assist with determining appropriate individualized treatment regimens.
- Liu J, Thatiparthi A, Martin A, et al. Prevalence of psoriasis among adults in the US 2009-2010 and 2013-2014 National Health and Nutrition Examination Surveys. J Am Acad Dermatol. 2021;84:767-769. doi:10.1016/j.jaad.2020.10.035
- Brezinski EA, Dhillon JS, Armstrong AW. Economic burden of psoriasis in the United States: a systematic review. JAMA Dermatol. 2015;151:651-658. doi:10.1001/jamadermatol.2014.3593
- National Center for Health Statistics. NHANES questionnaires, datasets, and related documentation. Centers for Disease Control and Prevention website. Accessed June 22, 2023. https://wwwn.cdc.govnchs/nhanes/Default.aspx
- Maya-Rico AM, Londoño-García Á, Palacios-Barahona AU, et al. Out-of-pocket costs for patients with psoriasis in an outpatient dermatology referral service. An Bras Dermatol. 2021;96:295-300. doi:10.1016/j.abd.2020.09.004
- Cheng J, Feldman SR. The cost of biologics for psoriasis is increasing. Drugs Context. 2014;3:212266. doi:10.7573/dic.212266
- Liu J, Thatiparthi A, Martin A, et al. Prevalence of psoriasis among adults in the US 2009-2010 and 2013-2014 National Health and Nutrition Examination Surveys. J Am Acad Dermatol. 2021;84:767-769. doi:10.1016/j.jaad.2020.10.035
- Brezinski EA, Dhillon JS, Armstrong AW. Economic burden of psoriasis in the United States: a systematic review. JAMA Dermatol. 2015;151:651-658. doi:10.1001/jamadermatol.2014.3593
- National Center for Health Statistics. NHANES questionnaires, datasets, and related documentation. Centers for Disease Control and Prevention website. Accessed June 22, 2023. https://wwwn.cdc.govnchs/nhanes/Default.aspx
- Maya-Rico AM, Londoño-García Á, Palacios-Barahona AU, et al. Out-of-pocket costs for patients with psoriasis in an outpatient dermatology referral service. An Bras Dermatol. 2021;96:295-300. doi:10.1016/j.abd.2020.09.004
- Cheng J, Feldman SR. The cost of biologics for psoriasis is increasing. Drugs Context. 2014;3:212266. doi:10.7573/dic.212266
Practice Points
- The economic burden on patients with psoriasis has been rising over time, as the disease impacts many aspects of patients’ lives.
- Various sociodemographic groups among patients with psoriasis are financially insecure. Knowing which groups are at higher risk for poor outcomes due to financial insecurity can assist with appropriate treatment regimens.
New evidence suggests genetic risk factors in hidradenitis suppurativa
Hidradenitis suppurativa (HS) is a chronic inflammatory skin condition with lesions that include deep-seated nodules and abscesses, draining tracts, and fibrotic scars, and has been reported to be highly heritable.
A genomewide association study (GWAS) that involved a meta-analysis of data from three large biobanks (UK Biobank, FinnGen, and BioVU) identified one significant locus and two suggestive loci. The researchers were able to replicate the association with HS for two loci near the SOX9 and KLF5 genes in BioVU.
In addition, they also looked at genetic correlations between HS and autoimmune and inflammatory diseases, and results suggested a positive association with inflammatory bowel disease, psoriasis, and type 2 diabetes.
However, while plausible, the variant associations near candidate genes do not prove a causal effect of these variants or genes on disease risk, and further study is needed.
“It’s possible that these genes aren’t the ones affected by the variations we describe, but both are very strong candidates,” said study author Christopher J. Sayed, MD, associate professor of dermatology, University of North Carolina at Chapel Hill. “This improves our understanding of HS as a disease that is likely related to genetic changes that result in dysregulated hair follicle maintenance, inflammation, and wound healing.”
In turn, this will allow clinicians to educate patients about the underlying genetic influences on HS, he explained, as opposed to stigmatizing misconceptions focusing only on weight, smoking, and hygiene.
“Larger studies are underway and will be needed to find variants that may predict things like disease severity and response to treatment, but this is a big first step in the right direction,” Dr. Sayed added.
The study was published online in JAMA Dermatology.
Loci identified
GWASs have been limited in HS, and previous research has not identified significant risk loci. In the current study, Dr. Sayed and colleagues sought to identify underlying genes and genetic mechanisms that may underlie pathogenesis in HS.
They performed a GWAS in a cohort of 720 patients who were part of the Hidradenitis Suppurativa Program for Research and Care Excellence (HS ProCARE) at the UNC department of dermatology, and controls from the National Longitudinal Study of Adolescent to Adult Health (Add Health) study, a U.S.-based study following adolescents through adulthood.
The UK Biobank (UKB) is a prospective biobank with about 500,000 individuals aged 40-69 years, and FinnGen collects and analyzes genome and health data from Finnish biobank participants. To increase power to detect associations, a GWAS was performed using participants from the UKB (247 HS cases, 453,048 controls). The HS ProCARE GWAS results were meta-analyzed with UKB, along with data from FinnGen (673 HS cases, 297,544 controls). This three-way meta-analysis revealed one genomewide significant locus and two suggestive loci.
The authors found that the most strongly associated variant, rs10512572 located on chromosome 17, showed the strongest association in FinnGen; at the second locus, the most strongly associated variant was rs17090189 located on chromosome 13 and also showed the strongest association in FinnGen; and at the third locus, the most strongly associated variant, rs5792315, located on chromosome 11, showed the strongest association in HS ProCARE.
Next, they tested the 10 most strongly associated variants at the three loci in the BioVU biobank, which has 290 HS cases, including 189 individuals of European ancestry and 101 individuals of African ancestry, with 64,234 and 12,105 controls, respectively. The locus on chromosome 17 was replicated in BioVU in the same direction of effect, while one variant at the chromosome 13 locus showed nominal evidence of association in the same direction.
In a four-way meta-analysis of BioVU combined with the other three studies, the chromosome locus became more significant and the chromosome 13 locus exceeded the genomewide significance threshold. In contrast, the chromosome 11 locus was not replicated in BioVU (P = .27).
The authors pointed out that variants at these loci are located in keratinocyte regulatory elements near the genes SOX9 and KLF5, which play a role in skin and follicular inflammation, but have not previously been associated with HS pathogenesis.
Finally, they looked to see if there were shared genetic components between HS and autoimmune and inflammatory diseases. A nominally significant genetic correlation was observed between HS and inflammatory bowel disease (P = .04), psoriasis (P = .03), and type 2 diabetes (P = .04), although none reached significance.
Different manifestation with genetic variant
In a related study, researchers assessed the prevalence of the NCSTN:c.671_682del variant among individuals with HS in Malta, the island country in the Mediterranean, which has a high prevalence of HS and its associated risk factors, particularly obesity.
Led by Dillon Mintoff, MD, of the department of pathology at the University of Malta, Msida, they conducted a cross-sectional study of 113 adults with HS. In this group, 14 (12.39%) were found to be heterozygous for the NCSTN:c.671_682del variant. Individuals who had this variant were more likely to develop symptoms earlier and to manifest them in atypical skin sites, including the scalp, neck, torso, and antecubital fossae. Additionally, even though their symptoms weren’t more severe than those without the variant, patients with the variant were more likely to require treatment with biologic agents.
Studies move genetics in HS forward
Writing in an accompanying editorial, Atlas Khan, PhD, and Lynn Petukhova, PhD, from Columbia University, New York, noted that both of these HS genetic studies “set a solid foundation for future studies aimed at understanding the biological and clinical relevance of new HS genetic evidence.”
“Each study suggests a series of experiments that will allow us to gain new knowledge about HS,” they wrote, including coming closer to providing patients with a genetic diagnosis.
In addition, evidence from the GWAS paper suggests that with “larger HS GWASs we will be able to better prioritize drug-repurposing studies,” concluded the editorialists. “Both of these goals will help to improve health outcomes for patients with HS and their family members.”
Dr. Sayed reported grants and/or personal fees from AbbVie, Novartis, UCB, Incyte, InflaRx, Alumis, and ChemoCentryx outside the submitted work; and serving as a volunteer member of the board of the HS Foundation and member of the European HS Foundation. The study in Malta was funded by the Government of Malta’s Tertiary Education Scholarship Scheme and Institutional Funds from the University of Malta. Dr. Mintoff reported grants from the Government of Malta Ministry for Education, Sport, Youth, Research and Innovation during the conduct of the study. Dr. Khan reported receiving grants from the National Institute of Diabetes and Digestive Kidney Diseases. Dr. Petukhova reported receiving grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Columbia University’s Precision Medicine Initiative, Herbert Irving Comprehensive Cancer Center, and Data Science Institute.
Hidradenitis suppurativa (HS) is a chronic inflammatory skin condition with lesions that include deep-seated nodules and abscesses, draining tracts, and fibrotic scars, and has been reported to be highly heritable.
A genomewide association study (GWAS) that involved a meta-analysis of data from three large biobanks (UK Biobank, FinnGen, and BioVU) identified one significant locus and two suggestive loci. The researchers were able to replicate the association with HS for two loci near the SOX9 and KLF5 genes in BioVU.
In addition, they also looked at genetic correlations between HS and autoimmune and inflammatory diseases, and results suggested a positive association with inflammatory bowel disease, psoriasis, and type 2 diabetes.
However, while plausible, the variant associations near candidate genes do not prove a causal effect of these variants or genes on disease risk, and further study is needed.
“It’s possible that these genes aren’t the ones affected by the variations we describe, but both are very strong candidates,” said study author Christopher J. Sayed, MD, associate professor of dermatology, University of North Carolina at Chapel Hill. “This improves our understanding of HS as a disease that is likely related to genetic changes that result in dysregulated hair follicle maintenance, inflammation, and wound healing.”
In turn, this will allow clinicians to educate patients about the underlying genetic influences on HS, he explained, as opposed to stigmatizing misconceptions focusing only on weight, smoking, and hygiene.
“Larger studies are underway and will be needed to find variants that may predict things like disease severity and response to treatment, but this is a big first step in the right direction,” Dr. Sayed added.
The study was published online in JAMA Dermatology.
Loci identified
GWASs have been limited in HS, and previous research has not identified significant risk loci. In the current study, Dr. Sayed and colleagues sought to identify underlying genes and genetic mechanisms that may underlie pathogenesis in HS.
They performed a GWAS in a cohort of 720 patients who were part of the Hidradenitis Suppurativa Program for Research and Care Excellence (HS ProCARE) at the UNC department of dermatology, and controls from the National Longitudinal Study of Adolescent to Adult Health (Add Health) study, a U.S.-based study following adolescents through adulthood.
The UK Biobank (UKB) is a prospective biobank with about 500,000 individuals aged 40-69 years, and FinnGen collects and analyzes genome and health data from Finnish biobank participants. To increase power to detect associations, a GWAS was performed using participants from the UKB (247 HS cases, 453,048 controls). The HS ProCARE GWAS results were meta-analyzed with UKB, along with data from FinnGen (673 HS cases, 297,544 controls). This three-way meta-analysis revealed one genomewide significant locus and two suggestive loci.
The authors found that the most strongly associated variant, rs10512572 located on chromosome 17, showed the strongest association in FinnGen; at the second locus, the most strongly associated variant was rs17090189 located on chromosome 13 and also showed the strongest association in FinnGen; and at the third locus, the most strongly associated variant, rs5792315, located on chromosome 11, showed the strongest association in HS ProCARE.
Next, they tested the 10 most strongly associated variants at the three loci in the BioVU biobank, which has 290 HS cases, including 189 individuals of European ancestry and 101 individuals of African ancestry, with 64,234 and 12,105 controls, respectively. The locus on chromosome 17 was replicated in BioVU in the same direction of effect, while one variant at the chromosome 13 locus showed nominal evidence of association in the same direction.
In a four-way meta-analysis of BioVU combined with the other three studies, the chromosome locus became more significant and the chromosome 13 locus exceeded the genomewide significance threshold. In contrast, the chromosome 11 locus was not replicated in BioVU (P = .27).
The authors pointed out that variants at these loci are located in keratinocyte regulatory elements near the genes SOX9 and KLF5, which play a role in skin and follicular inflammation, but have not previously been associated with HS pathogenesis.
Finally, they looked to see if there were shared genetic components between HS and autoimmune and inflammatory diseases. A nominally significant genetic correlation was observed between HS and inflammatory bowel disease (P = .04), psoriasis (P = .03), and type 2 diabetes (P = .04), although none reached significance.
Different manifestation with genetic variant
In a related study, researchers assessed the prevalence of the NCSTN:c.671_682del variant among individuals with HS in Malta, the island country in the Mediterranean, which has a high prevalence of HS and its associated risk factors, particularly obesity.
Led by Dillon Mintoff, MD, of the department of pathology at the University of Malta, Msida, they conducted a cross-sectional study of 113 adults with HS. In this group, 14 (12.39%) were found to be heterozygous for the NCSTN:c.671_682del variant. Individuals who had this variant were more likely to develop symptoms earlier and to manifest them in atypical skin sites, including the scalp, neck, torso, and antecubital fossae. Additionally, even though their symptoms weren’t more severe than those without the variant, patients with the variant were more likely to require treatment with biologic agents.
Studies move genetics in HS forward
Writing in an accompanying editorial, Atlas Khan, PhD, and Lynn Petukhova, PhD, from Columbia University, New York, noted that both of these HS genetic studies “set a solid foundation for future studies aimed at understanding the biological and clinical relevance of new HS genetic evidence.”
“Each study suggests a series of experiments that will allow us to gain new knowledge about HS,” they wrote, including coming closer to providing patients with a genetic diagnosis.
In addition, evidence from the GWAS paper suggests that with “larger HS GWASs we will be able to better prioritize drug-repurposing studies,” concluded the editorialists. “Both of these goals will help to improve health outcomes for patients with HS and their family members.”
Dr. Sayed reported grants and/or personal fees from AbbVie, Novartis, UCB, Incyte, InflaRx, Alumis, and ChemoCentryx outside the submitted work; and serving as a volunteer member of the board of the HS Foundation and member of the European HS Foundation. The study in Malta was funded by the Government of Malta’s Tertiary Education Scholarship Scheme and Institutional Funds from the University of Malta. Dr. Mintoff reported grants from the Government of Malta Ministry for Education, Sport, Youth, Research and Innovation during the conduct of the study. Dr. Khan reported receiving grants from the National Institute of Diabetes and Digestive Kidney Diseases. Dr. Petukhova reported receiving grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Columbia University’s Precision Medicine Initiative, Herbert Irving Comprehensive Cancer Center, and Data Science Institute.
Hidradenitis suppurativa (HS) is a chronic inflammatory skin condition with lesions that include deep-seated nodules and abscesses, draining tracts, and fibrotic scars, and has been reported to be highly heritable.
A genomewide association study (GWAS) that involved a meta-analysis of data from three large biobanks (UK Biobank, FinnGen, and BioVU) identified one significant locus and two suggestive loci. The researchers were able to replicate the association with HS for two loci near the SOX9 and KLF5 genes in BioVU.
In addition, they also looked at genetic correlations between HS and autoimmune and inflammatory diseases, and results suggested a positive association with inflammatory bowel disease, psoriasis, and type 2 diabetes.
However, while plausible, the variant associations near candidate genes do not prove a causal effect of these variants or genes on disease risk, and further study is needed.
“It’s possible that these genes aren’t the ones affected by the variations we describe, but both are very strong candidates,” said study author Christopher J. Sayed, MD, associate professor of dermatology, University of North Carolina at Chapel Hill. “This improves our understanding of HS as a disease that is likely related to genetic changes that result in dysregulated hair follicle maintenance, inflammation, and wound healing.”
In turn, this will allow clinicians to educate patients about the underlying genetic influences on HS, he explained, as opposed to stigmatizing misconceptions focusing only on weight, smoking, and hygiene.
“Larger studies are underway and will be needed to find variants that may predict things like disease severity and response to treatment, but this is a big first step in the right direction,” Dr. Sayed added.
The study was published online in JAMA Dermatology.
Loci identified
GWASs have been limited in HS, and previous research has not identified significant risk loci. In the current study, Dr. Sayed and colleagues sought to identify underlying genes and genetic mechanisms that may underlie pathogenesis in HS.
They performed a GWAS in a cohort of 720 patients who were part of the Hidradenitis Suppurativa Program for Research and Care Excellence (HS ProCARE) at the UNC department of dermatology, and controls from the National Longitudinal Study of Adolescent to Adult Health (Add Health) study, a U.S.-based study following adolescents through adulthood.
The UK Biobank (UKB) is a prospective biobank with about 500,000 individuals aged 40-69 years, and FinnGen collects and analyzes genome and health data from Finnish biobank participants. To increase power to detect associations, a GWAS was performed using participants from the UKB (247 HS cases, 453,048 controls). The HS ProCARE GWAS results were meta-analyzed with UKB, along with data from FinnGen (673 HS cases, 297,544 controls). This three-way meta-analysis revealed one genomewide significant locus and two suggestive loci.
The authors found that the most strongly associated variant, rs10512572 located on chromosome 17, showed the strongest association in FinnGen; at the second locus, the most strongly associated variant was rs17090189 located on chromosome 13 and also showed the strongest association in FinnGen; and at the third locus, the most strongly associated variant, rs5792315, located on chromosome 11, showed the strongest association in HS ProCARE.
Next, they tested the 10 most strongly associated variants at the three loci in the BioVU biobank, which has 290 HS cases, including 189 individuals of European ancestry and 101 individuals of African ancestry, with 64,234 and 12,105 controls, respectively. The locus on chromosome 17 was replicated in BioVU in the same direction of effect, while one variant at the chromosome 13 locus showed nominal evidence of association in the same direction.
In a four-way meta-analysis of BioVU combined with the other three studies, the chromosome locus became more significant and the chromosome 13 locus exceeded the genomewide significance threshold. In contrast, the chromosome 11 locus was not replicated in BioVU (P = .27).
The authors pointed out that variants at these loci are located in keratinocyte regulatory elements near the genes SOX9 and KLF5, which play a role in skin and follicular inflammation, but have not previously been associated with HS pathogenesis.
Finally, they looked to see if there were shared genetic components between HS and autoimmune and inflammatory diseases. A nominally significant genetic correlation was observed between HS and inflammatory bowel disease (P = .04), psoriasis (P = .03), and type 2 diabetes (P = .04), although none reached significance.
Different manifestation with genetic variant
In a related study, researchers assessed the prevalence of the NCSTN:c.671_682del variant among individuals with HS in Malta, the island country in the Mediterranean, which has a high prevalence of HS and its associated risk factors, particularly obesity.
Led by Dillon Mintoff, MD, of the department of pathology at the University of Malta, Msida, they conducted a cross-sectional study of 113 adults with HS. In this group, 14 (12.39%) were found to be heterozygous for the NCSTN:c.671_682del variant. Individuals who had this variant were more likely to develop symptoms earlier and to manifest them in atypical skin sites, including the scalp, neck, torso, and antecubital fossae. Additionally, even though their symptoms weren’t more severe than those without the variant, patients with the variant were more likely to require treatment with biologic agents.
Studies move genetics in HS forward
Writing in an accompanying editorial, Atlas Khan, PhD, and Lynn Petukhova, PhD, from Columbia University, New York, noted that both of these HS genetic studies “set a solid foundation for future studies aimed at understanding the biological and clinical relevance of new HS genetic evidence.”
“Each study suggests a series of experiments that will allow us to gain new knowledge about HS,” they wrote, including coming closer to providing patients with a genetic diagnosis.
In addition, evidence from the GWAS paper suggests that with “larger HS GWASs we will be able to better prioritize drug-repurposing studies,” concluded the editorialists. “Both of these goals will help to improve health outcomes for patients with HS and their family members.”
Dr. Sayed reported grants and/or personal fees from AbbVie, Novartis, UCB, Incyte, InflaRx, Alumis, and ChemoCentryx outside the submitted work; and serving as a volunteer member of the board of the HS Foundation and member of the European HS Foundation. The study in Malta was funded by the Government of Malta’s Tertiary Education Scholarship Scheme and Institutional Funds from the University of Malta. Dr. Mintoff reported grants from the Government of Malta Ministry for Education, Sport, Youth, Research and Innovation during the conduct of the study. Dr. Khan reported receiving grants from the National Institute of Diabetes and Digestive Kidney Diseases. Dr. Petukhova reported receiving grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Columbia University’s Precision Medicine Initiative, Herbert Irving Comprehensive Cancer Center, and Data Science Institute.
FROM JAMA DERMATOLOGY
PsA biomarkers move researchers closer to predictive test
In a new study, researchers report that they have found epigenetic methylation markers on 15 genes that appear to foreshadow psoriatic arthritis (PsA), a development that could bring scientists closer to developing a DNA test to predict which patients with psoriasis will develop the condition.
While no predictive test is in sight yet, the findings published in Arthritis & Rheumatology mark an important step, study lead author Omar F. Cruz-Correa, PhD, of the Psoriatic Arthritis Research Program in the University Health Network, Toronto, said in an interview. “In the future, markers like these could be measured by dermatologists and even general practitioners to help identify new psoriasis patients at a high risk of developing PsA,” he said. “Then both the health care team and the patients themselves could be more aware of their increased risk and the pressing need of closer monitoring for musculoskeletal symptoms. Once the first symptoms appear, treatment can be initiated early on, helping to prevent permanent joint damage.”
An estimated 30% of patients with psoriasis will develop PsA, too, putting them at higher risk of disability and death. According to Dr. Cruz-Correa, “one of the more pressing matters in PsA is the lack of means of predicting which psoriasis patients will develop PsA.”
DNA methylation, the topic of the new study, has already been linked to psoriasis and PsA. It’s “relatively easy to measure and helps regulate gene expression in response to environmental effects,” Dr. Cruz-Correa said. “DNA methylation is also appealing because it serves as an intermediary between environment and genetic factors as it’s transmitted between generations of cells and influenced by external factors.”
For the new study, researchers examined the DNA of 117 patients with psoriasis – 58 who went on to develop PsA (“converters”) and another 59 who were matched to converters but did not develop PsA (“nonconverters”). The patients were in a larger group of 700 patients with psoriasis who had the disease for a mean of about 17 years at the time of blood sampling.
Samples from converters were taken an average of 5.16 years (± 12.77 years) before PsA set in.
The researchers report that they found “36 highly relevant methylation markers … across 15 genes and several intergenic regions. A classification model relying on these markers identified converters and nonconverters with an area under the ROC curve of 0.9644.”
Statistically, this number is high and means that “the DNA methylation markers are really good at identifying psoriasis patients who will develop PsA and those that will not,” at least in this specific patient group, Dr. Cruz-Correa said.
At this point, the number of markers is a bit too high to develop a feasible DNA test to predict PsA, he said. “However, the results from our study have also pointed us toward some interesting metabolic pathways that may warrant further study.”
What’s next?
The first step forward “is the validation of these predictive DNA methylation markers in a wider population of patients with varied clinical and demographic characteristics. This would help assess the potential for generalization of such a test,” Dr. Cruz-Correa said. “A second step is to assess the potential impact of these methylation markers on disease activity and treatment response, which are clinical outcomes of great importance to patients.”
Meanwhile, he said, “there are ongoing efforts to shed light into how DNA methylation integrates with other epigenetic mechanisms like micro-RNAs to regulate gene expression in concert with one another. An integrative look into these mechanisms may be able to give insight into the pathogenesis of psoriatic disease in a way that has not been possible before.”
In an interview, Johann E. Gudjonsson, MD, PhD, professor of skin molecular immunology at the University of Michigan, Ann Arbor, said the study “is interesting and important as it indicates that there are changes in the blood that occur before the development of psoriatic arthritis. However, it does not provide much in terms of novel insights into the mechanisms involved and is still a long way away from being useful as a clinical predictor or biomarker.”
The National Psoriasis Foundation, Krembil Foundation, and Canadian Institutes of Health Research provided support for the study. Dr. Cruz-Correa reports support from the National Psoriasis Foundation and the Arthritis Society. Dr. Gudjonsson has no relevant financial relationships.
In a new study, researchers report that they have found epigenetic methylation markers on 15 genes that appear to foreshadow psoriatic arthritis (PsA), a development that could bring scientists closer to developing a DNA test to predict which patients with psoriasis will develop the condition.
While no predictive test is in sight yet, the findings published in Arthritis & Rheumatology mark an important step, study lead author Omar F. Cruz-Correa, PhD, of the Psoriatic Arthritis Research Program in the University Health Network, Toronto, said in an interview. “In the future, markers like these could be measured by dermatologists and even general practitioners to help identify new psoriasis patients at a high risk of developing PsA,” he said. “Then both the health care team and the patients themselves could be more aware of their increased risk and the pressing need of closer monitoring for musculoskeletal symptoms. Once the first symptoms appear, treatment can be initiated early on, helping to prevent permanent joint damage.”
An estimated 30% of patients with psoriasis will develop PsA, too, putting them at higher risk of disability and death. According to Dr. Cruz-Correa, “one of the more pressing matters in PsA is the lack of means of predicting which psoriasis patients will develop PsA.”
DNA methylation, the topic of the new study, has already been linked to psoriasis and PsA. It’s “relatively easy to measure and helps regulate gene expression in response to environmental effects,” Dr. Cruz-Correa said. “DNA methylation is also appealing because it serves as an intermediary between environment and genetic factors as it’s transmitted between generations of cells and influenced by external factors.”
For the new study, researchers examined the DNA of 117 patients with psoriasis – 58 who went on to develop PsA (“converters”) and another 59 who were matched to converters but did not develop PsA (“nonconverters”). The patients were in a larger group of 700 patients with psoriasis who had the disease for a mean of about 17 years at the time of blood sampling.
Samples from converters were taken an average of 5.16 years (± 12.77 years) before PsA set in.
The researchers report that they found “36 highly relevant methylation markers … across 15 genes and several intergenic regions. A classification model relying on these markers identified converters and nonconverters with an area under the ROC curve of 0.9644.”
Statistically, this number is high and means that “the DNA methylation markers are really good at identifying psoriasis patients who will develop PsA and those that will not,” at least in this specific patient group, Dr. Cruz-Correa said.
At this point, the number of markers is a bit too high to develop a feasible DNA test to predict PsA, he said. “However, the results from our study have also pointed us toward some interesting metabolic pathways that may warrant further study.”
What’s next?
The first step forward “is the validation of these predictive DNA methylation markers in a wider population of patients with varied clinical and demographic characteristics. This would help assess the potential for generalization of such a test,” Dr. Cruz-Correa said. “A second step is to assess the potential impact of these methylation markers on disease activity and treatment response, which are clinical outcomes of great importance to patients.”
Meanwhile, he said, “there are ongoing efforts to shed light into how DNA methylation integrates with other epigenetic mechanisms like micro-RNAs to regulate gene expression in concert with one another. An integrative look into these mechanisms may be able to give insight into the pathogenesis of psoriatic disease in a way that has not been possible before.”
In an interview, Johann E. Gudjonsson, MD, PhD, professor of skin molecular immunology at the University of Michigan, Ann Arbor, said the study “is interesting and important as it indicates that there are changes in the blood that occur before the development of psoriatic arthritis. However, it does not provide much in terms of novel insights into the mechanisms involved and is still a long way away from being useful as a clinical predictor or biomarker.”
The National Psoriasis Foundation, Krembil Foundation, and Canadian Institutes of Health Research provided support for the study. Dr. Cruz-Correa reports support from the National Psoriasis Foundation and the Arthritis Society. Dr. Gudjonsson has no relevant financial relationships.
In a new study, researchers report that they have found epigenetic methylation markers on 15 genes that appear to foreshadow psoriatic arthritis (PsA), a development that could bring scientists closer to developing a DNA test to predict which patients with psoriasis will develop the condition.
While no predictive test is in sight yet, the findings published in Arthritis & Rheumatology mark an important step, study lead author Omar F. Cruz-Correa, PhD, of the Psoriatic Arthritis Research Program in the University Health Network, Toronto, said in an interview. “In the future, markers like these could be measured by dermatologists and even general practitioners to help identify new psoriasis patients at a high risk of developing PsA,” he said. “Then both the health care team and the patients themselves could be more aware of their increased risk and the pressing need of closer monitoring for musculoskeletal symptoms. Once the first symptoms appear, treatment can be initiated early on, helping to prevent permanent joint damage.”
An estimated 30% of patients with psoriasis will develop PsA, too, putting them at higher risk of disability and death. According to Dr. Cruz-Correa, “one of the more pressing matters in PsA is the lack of means of predicting which psoriasis patients will develop PsA.”
DNA methylation, the topic of the new study, has already been linked to psoriasis and PsA. It’s “relatively easy to measure and helps regulate gene expression in response to environmental effects,” Dr. Cruz-Correa said. “DNA methylation is also appealing because it serves as an intermediary between environment and genetic factors as it’s transmitted between generations of cells and influenced by external factors.”
For the new study, researchers examined the DNA of 117 patients with psoriasis – 58 who went on to develop PsA (“converters”) and another 59 who were matched to converters but did not develop PsA (“nonconverters”). The patients were in a larger group of 700 patients with psoriasis who had the disease for a mean of about 17 years at the time of blood sampling.
Samples from converters were taken an average of 5.16 years (± 12.77 years) before PsA set in.
The researchers report that they found “36 highly relevant methylation markers … across 15 genes and several intergenic regions. A classification model relying on these markers identified converters and nonconverters with an area under the ROC curve of 0.9644.”
Statistically, this number is high and means that “the DNA methylation markers are really good at identifying psoriasis patients who will develop PsA and those that will not,” at least in this specific patient group, Dr. Cruz-Correa said.
At this point, the number of markers is a bit too high to develop a feasible DNA test to predict PsA, he said. “However, the results from our study have also pointed us toward some interesting metabolic pathways that may warrant further study.”
What’s next?
The first step forward “is the validation of these predictive DNA methylation markers in a wider population of patients with varied clinical and demographic characteristics. This would help assess the potential for generalization of such a test,” Dr. Cruz-Correa said. “A second step is to assess the potential impact of these methylation markers on disease activity and treatment response, which are clinical outcomes of great importance to patients.”
Meanwhile, he said, “there are ongoing efforts to shed light into how DNA methylation integrates with other epigenetic mechanisms like micro-RNAs to regulate gene expression in concert with one another. An integrative look into these mechanisms may be able to give insight into the pathogenesis of psoriatic disease in a way that has not been possible before.”
In an interview, Johann E. Gudjonsson, MD, PhD, professor of skin molecular immunology at the University of Michigan, Ann Arbor, said the study “is interesting and important as it indicates that there are changes in the blood that occur before the development of psoriatic arthritis. However, it does not provide much in terms of novel insights into the mechanisms involved and is still a long way away from being useful as a clinical predictor or biomarker.”
The National Psoriasis Foundation, Krembil Foundation, and Canadian Institutes of Health Research provided support for the study. Dr. Cruz-Correa reports support from the National Psoriasis Foundation and the Arthritis Society. Dr. Gudjonsson has no relevant financial relationships.
FROM ARTHRITIS & RHEUMATOLOGY
Study validates use of new psoriatic arthritis prediction tool
Though it requires further validation, researchers led by rheumatologist Lihi Eder, MD, PhD, of the Women’s College Research Institute at Women’s College Hospital, Toronto, characterized the development and validation of PRESTO as “an important first step in the development and testing of interventional strategies that may ultimately halt disease progression,” they wrote in their study of the tool, which published in Arthritis & Rheumatology. Dr. Eder presented a summary of progress on the effort at the 2023 annual meeting of the Canadian Rheumatology Association.
To develop and validate the tool, the researchers evaluated 635 patients from the University of Toronto Psoriasis Cohort, which was launched in 2006 as a prospective longitudinal cohort study to examine risk factors for the development of PsA among patients with psoriasis. Patients enrolled in the cohort have a dermatologist-confirmed diagnosis of psoriasis and are assessed by a rheumatologist prior to enrollment to exclude those with inflammatory arthritis in the past or at the time of assessment.
To develop prediction models for PsA, Dr. Eder and colleagues used information from the patient cohort demographics, psoriasis characteristics, comorbidities, medications, and musculoskeletal symptoms. Next, they used multivariable logistic regression models adjusting for covariates, duration of psoriasis, and the log duration at risk to estimate the probability of developing PsA within 1-year and 5-year time windows from consecutive study visits.
The mean age of the study participants was 47 years, 76% were White, and 57% were male; and they had psoriasis for a mean of 16 years. The researchers found that 51 patients developed PsA during the 1-year follow-up, and 71 developed PsA during the 5-year follow-up. The risk of developing PsA within 1 year was associated with younger age, male sex, family history of psoriasis, back stiffness, nail pitting, joint stiffness, use of biologic medications, patient global health, and pain severity (area under the curve, 72.3).
In addition, the risk of developing PsA within 5 years was associated with morning stiffness, psoriatic nail lesions, psoriasis severity, fatigue, pain, and use of systemic non-biologic medication or phototherapy (AUC, 74.9). Calibration plots showed reasonable agreement between predicted and observed probabilities.
“Interestingly, several previously reported risk factors for PsA, such as HLA-B27, family history of PsA, uveitis, and flexural psoriasis, were not included in the risk prediction model due to their scarcity in our cohort,” the researchers wrote. “This finding may be due to immortal time bias which can complicate the development of risk prediction models for PsA. Genetic factors or their surrogates (e.g., family history of PsA) are associated with the development of PsA concurrently or shortly after the onset of psoriasis.”
They acknowledged certain limitations of the study, including its relatively small sample size and questionable generalizability of the study findings, “as most of the patients were recruited from dermatology clinics leading to overrepresentation of moderate-severe psoriasis. Therefore, PRESTO will require an external validation to assess its performance in other populations of psoriasis patients with different characteristics.”
Saakshi Khattri, MD, a board-certified dermatologist, rheumatologist, and internist at the Icahn School of Medicine at Mount Sinai, New York, who was not involved in the study and was asked to comment on the results, characterized the PRESTO tool as “an interesting step in the right direction, but it’s the first step.”
Since dermatologists are usually the first point of contact for psoriasis patients, she added, “a risk calculator can be helpful, but the question remains: When do we refer them to a rheumatologist? If the risk comes to 5%, is that a low risk that doesn’t need referral to rheumatology? I don’t think those questions have been answered here. From a rheumatology perspective, does the risk calculator help me decide when to intervene? At present, I’m not sure it does. Perhaps a higher score might make us intervene sooner if our clinical exam doesn’t show swollen or tender joints.”
Clinical exam findings and history she considers as a rheumatologist before making treatment recommendations include the following: Are there swollen and tender joints? Does the patient report morning stiffness for upwards of 30 minutes? Do they have enthesitis or dactylitis? Is there axial involvement? “Imaging can help if there isn’t anything on clinical exam and the history is compelling and/or the patient has risk factors for PsA,” she said.
The study’s finding of biologic use being associated with risk of developing PsA at year 1 but not at year 5 is “confusing,” Dr. Khattri added. “My concern is, will that now dissuade our moderate to severe psoriasis patients from using biologics to clear their psoriasis? We know that biologics are indicated for moderate to severe psoriasis. We also know psoriasis is associated with increased cardiovascular risk and there’s data to suggest that treatment with biologics with its resultant decrease in systemic inflammation can decrease cardiovascular risk.”
The study was supported by a New Investigator Grant from the Physician Services Incorporated Foundation. Dr. Eder disclosed that she is supported by the Canada Research Chair in Inflammatory Rheumatic Diseases. Dr. Khattri reported that she is a member of the advisory board for UCB, Janssen, AbbVie, Regeneron, Sanofi, Lilly, Argenx, and Arcutis. She has also received research funds from Incyte, AbbVie, Leo, Galderma, Pfizer, and Acelyrin.
Though it requires further validation, researchers led by rheumatologist Lihi Eder, MD, PhD, of the Women’s College Research Institute at Women’s College Hospital, Toronto, characterized the development and validation of PRESTO as “an important first step in the development and testing of interventional strategies that may ultimately halt disease progression,” they wrote in their study of the tool, which published in Arthritis & Rheumatology. Dr. Eder presented a summary of progress on the effort at the 2023 annual meeting of the Canadian Rheumatology Association.
To develop and validate the tool, the researchers evaluated 635 patients from the University of Toronto Psoriasis Cohort, which was launched in 2006 as a prospective longitudinal cohort study to examine risk factors for the development of PsA among patients with psoriasis. Patients enrolled in the cohort have a dermatologist-confirmed diagnosis of psoriasis and are assessed by a rheumatologist prior to enrollment to exclude those with inflammatory arthritis in the past or at the time of assessment.
To develop prediction models for PsA, Dr. Eder and colleagues used information from the patient cohort demographics, psoriasis characteristics, comorbidities, medications, and musculoskeletal symptoms. Next, they used multivariable logistic regression models adjusting for covariates, duration of psoriasis, and the log duration at risk to estimate the probability of developing PsA within 1-year and 5-year time windows from consecutive study visits.
The mean age of the study participants was 47 years, 76% were White, and 57% were male; and they had psoriasis for a mean of 16 years. The researchers found that 51 patients developed PsA during the 1-year follow-up, and 71 developed PsA during the 5-year follow-up. The risk of developing PsA within 1 year was associated with younger age, male sex, family history of psoriasis, back stiffness, nail pitting, joint stiffness, use of biologic medications, patient global health, and pain severity (area under the curve, 72.3).
In addition, the risk of developing PsA within 5 years was associated with morning stiffness, psoriatic nail lesions, psoriasis severity, fatigue, pain, and use of systemic non-biologic medication or phototherapy (AUC, 74.9). Calibration plots showed reasonable agreement between predicted and observed probabilities.
“Interestingly, several previously reported risk factors for PsA, such as HLA-B27, family history of PsA, uveitis, and flexural psoriasis, were not included in the risk prediction model due to their scarcity in our cohort,” the researchers wrote. “This finding may be due to immortal time bias which can complicate the development of risk prediction models for PsA. Genetic factors or their surrogates (e.g., family history of PsA) are associated with the development of PsA concurrently or shortly after the onset of psoriasis.”
They acknowledged certain limitations of the study, including its relatively small sample size and questionable generalizability of the study findings, “as most of the patients were recruited from dermatology clinics leading to overrepresentation of moderate-severe psoriasis. Therefore, PRESTO will require an external validation to assess its performance in other populations of psoriasis patients with different characteristics.”
Saakshi Khattri, MD, a board-certified dermatologist, rheumatologist, and internist at the Icahn School of Medicine at Mount Sinai, New York, who was not involved in the study and was asked to comment on the results, characterized the PRESTO tool as “an interesting step in the right direction, but it’s the first step.”
Since dermatologists are usually the first point of contact for psoriasis patients, she added, “a risk calculator can be helpful, but the question remains: When do we refer them to a rheumatologist? If the risk comes to 5%, is that a low risk that doesn’t need referral to rheumatology? I don’t think those questions have been answered here. From a rheumatology perspective, does the risk calculator help me decide when to intervene? At present, I’m not sure it does. Perhaps a higher score might make us intervene sooner if our clinical exam doesn’t show swollen or tender joints.”
Clinical exam findings and history she considers as a rheumatologist before making treatment recommendations include the following: Are there swollen and tender joints? Does the patient report morning stiffness for upwards of 30 minutes? Do they have enthesitis or dactylitis? Is there axial involvement? “Imaging can help if there isn’t anything on clinical exam and the history is compelling and/or the patient has risk factors for PsA,” she said.
The study’s finding of biologic use being associated with risk of developing PsA at year 1 but not at year 5 is “confusing,” Dr. Khattri added. “My concern is, will that now dissuade our moderate to severe psoriasis patients from using biologics to clear their psoriasis? We know that biologics are indicated for moderate to severe psoriasis. We also know psoriasis is associated with increased cardiovascular risk and there’s data to suggest that treatment with biologics with its resultant decrease in systemic inflammation can decrease cardiovascular risk.”
The study was supported by a New Investigator Grant from the Physician Services Incorporated Foundation. Dr. Eder disclosed that she is supported by the Canada Research Chair in Inflammatory Rheumatic Diseases. Dr. Khattri reported that she is a member of the advisory board for UCB, Janssen, AbbVie, Regeneron, Sanofi, Lilly, Argenx, and Arcutis. She has also received research funds from Incyte, AbbVie, Leo, Galderma, Pfizer, and Acelyrin.
Though it requires further validation, researchers led by rheumatologist Lihi Eder, MD, PhD, of the Women’s College Research Institute at Women’s College Hospital, Toronto, characterized the development and validation of PRESTO as “an important first step in the development and testing of interventional strategies that may ultimately halt disease progression,” they wrote in their study of the tool, which published in Arthritis & Rheumatology. Dr. Eder presented a summary of progress on the effort at the 2023 annual meeting of the Canadian Rheumatology Association.
To develop and validate the tool, the researchers evaluated 635 patients from the University of Toronto Psoriasis Cohort, which was launched in 2006 as a prospective longitudinal cohort study to examine risk factors for the development of PsA among patients with psoriasis. Patients enrolled in the cohort have a dermatologist-confirmed diagnosis of psoriasis and are assessed by a rheumatologist prior to enrollment to exclude those with inflammatory arthritis in the past or at the time of assessment.
To develop prediction models for PsA, Dr. Eder and colleagues used information from the patient cohort demographics, psoriasis characteristics, comorbidities, medications, and musculoskeletal symptoms. Next, they used multivariable logistic regression models adjusting for covariates, duration of psoriasis, and the log duration at risk to estimate the probability of developing PsA within 1-year and 5-year time windows from consecutive study visits.
The mean age of the study participants was 47 years, 76% were White, and 57% were male; and they had psoriasis for a mean of 16 years. The researchers found that 51 patients developed PsA during the 1-year follow-up, and 71 developed PsA during the 5-year follow-up. The risk of developing PsA within 1 year was associated with younger age, male sex, family history of psoriasis, back stiffness, nail pitting, joint stiffness, use of biologic medications, patient global health, and pain severity (area under the curve, 72.3).
In addition, the risk of developing PsA within 5 years was associated with morning stiffness, psoriatic nail lesions, psoriasis severity, fatigue, pain, and use of systemic non-biologic medication or phototherapy (AUC, 74.9). Calibration plots showed reasonable agreement between predicted and observed probabilities.
“Interestingly, several previously reported risk factors for PsA, such as HLA-B27, family history of PsA, uveitis, and flexural psoriasis, were not included in the risk prediction model due to their scarcity in our cohort,” the researchers wrote. “This finding may be due to immortal time bias which can complicate the development of risk prediction models for PsA. Genetic factors or their surrogates (e.g., family history of PsA) are associated with the development of PsA concurrently or shortly after the onset of psoriasis.”
They acknowledged certain limitations of the study, including its relatively small sample size and questionable generalizability of the study findings, “as most of the patients were recruited from dermatology clinics leading to overrepresentation of moderate-severe psoriasis. Therefore, PRESTO will require an external validation to assess its performance in other populations of psoriasis patients with different characteristics.”
Saakshi Khattri, MD, a board-certified dermatologist, rheumatologist, and internist at the Icahn School of Medicine at Mount Sinai, New York, who was not involved in the study and was asked to comment on the results, characterized the PRESTO tool as “an interesting step in the right direction, but it’s the first step.”
Since dermatologists are usually the first point of contact for psoriasis patients, she added, “a risk calculator can be helpful, but the question remains: When do we refer them to a rheumatologist? If the risk comes to 5%, is that a low risk that doesn’t need referral to rheumatology? I don’t think those questions have been answered here. From a rheumatology perspective, does the risk calculator help me decide when to intervene? At present, I’m not sure it does. Perhaps a higher score might make us intervene sooner if our clinical exam doesn’t show swollen or tender joints.”
Clinical exam findings and history she considers as a rheumatologist before making treatment recommendations include the following: Are there swollen and tender joints? Does the patient report morning stiffness for upwards of 30 minutes? Do they have enthesitis or dactylitis? Is there axial involvement? “Imaging can help if there isn’t anything on clinical exam and the history is compelling and/or the patient has risk factors for PsA,” she said.
The study’s finding of biologic use being associated with risk of developing PsA at year 1 but not at year 5 is “confusing,” Dr. Khattri added. “My concern is, will that now dissuade our moderate to severe psoriasis patients from using biologics to clear their psoriasis? We know that biologics are indicated for moderate to severe psoriasis. We also know psoriasis is associated with increased cardiovascular risk and there’s data to suggest that treatment with biologics with its resultant decrease in systemic inflammation can decrease cardiovascular risk.”
The study was supported by a New Investigator Grant from the Physician Services Incorporated Foundation. Dr. Eder disclosed that she is supported by the Canada Research Chair in Inflammatory Rheumatic Diseases. Dr. Khattri reported that she is a member of the advisory board for UCB, Janssen, AbbVie, Regeneron, Sanofi, Lilly, Argenx, and Arcutis. She has also received research funds from Incyte, AbbVie, Leo, Galderma, Pfizer, and Acelyrin.
FROM ARTHRITIS AND RHEUMATOLOGY
Roflumilast cream appears safe, effective for children with psoriasis, researchers report
In patients aged 2-11 years, roflumilast cream was well tolerated and improved signs and symptoms of psoriasis over 4 weeks, according to results from a pair of phase two studies.
“Limited topical treatments are approved for children younger than 12 years old with psoriasis,” researchers led by Adelaide A. Hebert, MD, wrote in their abstract. The results were presented during a poster session at the annual meeting of the Society for Pediatric Dermatology.
Roflumilast cream 0.3% (Zoryve) is a once-daily, topical nonsteroidal treatment from Arcutis Biotherapeutics. A phosphodiesterase-4 inhibitor, it was approved by the Food and Drug Administration in 2022 for mild, moderate and severe psoriasis in individuals aged 12 and older, including intertriginous psoriasis.
For the analysis, Dr. Hebert, chief of pediatric dermatology at the University of Texas, Houston, and colleagues conducted two 4-week, phase 2, open-label safety studies of roflumilast cream 0.3%.
One, study 216, enrolled 10 children aged 2-5, and all but one were Black. The other, study 215, enrolled 20 children aged 6-11, and half were Black and nearly half were White. At baseline, patients had 2% or greater body surface area (BSA) involvement and an Investigator Global Assessment (IGA) score of at least mild.
Caregivers applied roflumilast cream to all affected areas once daily for 28 days. The researchers collected pharmacokinetic samples at week 2 and week 4. The primary endpoints were pharmacokinetic, safety, and tolerability.
Efficacy was evaluated as exploratory endpoints: An IGA of clear or almost clear plus a 2-grade or more improvement from baseline, a 50% or greater improvement and a 75% or greater improvement on the Psoriasis Area and Severity Index (PASI-50 and PASI-75), a 4-point or greater reduction in the Worst Itch–Numeric Rating Scale (WI-NRS) in patients with a baseline score of 4 or greater, a mean change from baseline in BSA, and improvement in the Children’s Dermatology Life Quality Index (CDLQI).
At baseline, the mean BSA was similar for patients enrolled in studies 216 and 215 (9.6% and 8.8%, respectively), and 80% of all patients had baseline IGA of moderate. By week 2, the mean roflumilast and N-oxide predose plasma concentrations among patients in the younger group were 2.15 and 22.4 ng/mL, compared with 3.15 and 28.9 ng/mL among those in the older group. At week 4, the mean roflumilast and N-oxide predose concentrations were 2.04 and 15.8 ng/mL in the younger group (study 216), compared with 1.68 and 15.7 ng/mL in the older group (study 215).
As for efficacy, 90% and 40% of patients in studies 216 and 215 achieved IGA success at week 4, respectively, while 90% and 50% achieved PASI-75, 90% and 40% achieved WI-NRS success, and the mean BSA reductions at week 4 were 79.1% and 44.4%. Meanwhile, one younger patient in study 216 reported a treatment-emergent adverse event (TEAE) of headache, which was considered mild, while four older patients in study 215 reported 8 TEAEs, which were considered mild and ranged from back pain to nasal congestion.
“The rapid onset of action was surprising but exceedingly rewarding for the subjects enrolled in the study,” Dr. Hebert told this news organization after the meeting. “The PASI scores and itch scores were markedly improved at the end of the 4-week clinical trial. Patient and parents alike were pleased to use a steroid-free option with once-daily application and rapid onset of action to help control plaque psoriasis.”
In the poster abstract, she and her coauthors concluded that “under maximal use conditions in children aged 2-11 years, roflumilast cream 0.3% was well tolerated and improved signs and symptoms of psoriasis with measured improvements in IGA score, PASI score, BSA involvement, CDLQI, and WI-NRS. Overall, pharmacokinetics, safety, tolerability, and efficacy in patients aged 2-11 years were consistent with prior results in adults and adolescents.”
The study was funded by Arcutis Biotherapeutics. Dr. Hebert reported that she is an investigator for Arcutis. About half the coauthors are employees of Arcutis, and the other half disclosed grants, research funding and/or honoraria from the company. Research grants from the company for this study were paid to the McGovern Medical School at the University of Texas.
In patients aged 2-11 years, roflumilast cream was well tolerated and improved signs and symptoms of psoriasis over 4 weeks, according to results from a pair of phase two studies.
“Limited topical treatments are approved for children younger than 12 years old with psoriasis,” researchers led by Adelaide A. Hebert, MD, wrote in their abstract. The results were presented during a poster session at the annual meeting of the Society for Pediatric Dermatology.
Roflumilast cream 0.3% (Zoryve) is a once-daily, topical nonsteroidal treatment from Arcutis Biotherapeutics. A phosphodiesterase-4 inhibitor, it was approved by the Food and Drug Administration in 2022 for mild, moderate and severe psoriasis in individuals aged 12 and older, including intertriginous psoriasis.
For the analysis, Dr. Hebert, chief of pediatric dermatology at the University of Texas, Houston, and colleagues conducted two 4-week, phase 2, open-label safety studies of roflumilast cream 0.3%.
One, study 216, enrolled 10 children aged 2-5, and all but one were Black. The other, study 215, enrolled 20 children aged 6-11, and half were Black and nearly half were White. At baseline, patients had 2% or greater body surface area (BSA) involvement and an Investigator Global Assessment (IGA) score of at least mild.
Caregivers applied roflumilast cream to all affected areas once daily for 28 days. The researchers collected pharmacokinetic samples at week 2 and week 4. The primary endpoints were pharmacokinetic, safety, and tolerability.
Efficacy was evaluated as exploratory endpoints: An IGA of clear or almost clear plus a 2-grade or more improvement from baseline, a 50% or greater improvement and a 75% or greater improvement on the Psoriasis Area and Severity Index (PASI-50 and PASI-75), a 4-point or greater reduction in the Worst Itch–Numeric Rating Scale (WI-NRS) in patients with a baseline score of 4 or greater, a mean change from baseline in BSA, and improvement in the Children’s Dermatology Life Quality Index (CDLQI).
At baseline, the mean BSA was similar for patients enrolled in studies 216 and 215 (9.6% and 8.8%, respectively), and 80% of all patients had baseline IGA of moderate. By week 2, the mean roflumilast and N-oxide predose plasma concentrations among patients in the younger group were 2.15 and 22.4 ng/mL, compared with 3.15 and 28.9 ng/mL among those in the older group. At week 4, the mean roflumilast and N-oxide predose concentrations were 2.04 and 15.8 ng/mL in the younger group (study 216), compared with 1.68 and 15.7 ng/mL in the older group (study 215).
As for efficacy, 90% and 40% of patients in studies 216 and 215 achieved IGA success at week 4, respectively, while 90% and 50% achieved PASI-75, 90% and 40% achieved WI-NRS success, and the mean BSA reductions at week 4 were 79.1% and 44.4%. Meanwhile, one younger patient in study 216 reported a treatment-emergent adverse event (TEAE) of headache, which was considered mild, while four older patients in study 215 reported 8 TEAEs, which were considered mild and ranged from back pain to nasal congestion.
“The rapid onset of action was surprising but exceedingly rewarding for the subjects enrolled in the study,” Dr. Hebert told this news organization after the meeting. “The PASI scores and itch scores were markedly improved at the end of the 4-week clinical trial. Patient and parents alike were pleased to use a steroid-free option with once-daily application and rapid onset of action to help control plaque psoriasis.”
In the poster abstract, she and her coauthors concluded that “under maximal use conditions in children aged 2-11 years, roflumilast cream 0.3% was well tolerated and improved signs and symptoms of psoriasis with measured improvements in IGA score, PASI score, BSA involvement, CDLQI, and WI-NRS. Overall, pharmacokinetics, safety, tolerability, and efficacy in patients aged 2-11 years were consistent with prior results in adults and adolescents.”
The study was funded by Arcutis Biotherapeutics. Dr. Hebert reported that she is an investigator for Arcutis. About half the coauthors are employees of Arcutis, and the other half disclosed grants, research funding and/or honoraria from the company. Research grants from the company for this study were paid to the McGovern Medical School at the University of Texas.
In patients aged 2-11 years, roflumilast cream was well tolerated and improved signs and symptoms of psoriasis over 4 weeks, according to results from a pair of phase two studies.
“Limited topical treatments are approved for children younger than 12 years old with psoriasis,” researchers led by Adelaide A. Hebert, MD, wrote in their abstract. The results were presented during a poster session at the annual meeting of the Society for Pediatric Dermatology.
Roflumilast cream 0.3% (Zoryve) is a once-daily, topical nonsteroidal treatment from Arcutis Biotherapeutics. A phosphodiesterase-4 inhibitor, it was approved by the Food and Drug Administration in 2022 for mild, moderate and severe psoriasis in individuals aged 12 and older, including intertriginous psoriasis.
For the analysis, Dr. Hebert, chief of pediatric dermatology at the University of Texas, Houston, and colleagues conducted two 4-week, phase 2, open-label safety studies of roflumilast cream 0.3%.
One, study 216, enrolled 10 children aged 2-5, and all but one were Black. The other, study 215, enrolled 20 children aged 6-11, and half were Black and nearly half were White. At baseline, patients had 2% or greater body surface area (BSA) involvement and an Investigator Global Assessment (IGA) score of at least mild.
Caregivers applied roflumilast cream to all affected areas once daily for 28 days. The researchers collected pharmacokinetic samples at week 2 and week 4. The primary endpoints were pharmacokinetic, safety, and tolerability.
Efficacy was evaluated as exploratory endpoints: An IGA of clear or almost clear plus a 2-grade or more improvement from baseline, a 50% or greater improvement and a 75% or greater improvement on the Psoriasis Area and Severity Index (PASI-50 and PASI-75), a 4-point or greater reduction in the Worst Itch–Numeric Rating Scale (WI-NRS) in patients with a baseline score of 4 or greater, a mean change from baseline in BSA, and improvement in the Children’s Dermatology Life Quality Index (CDLQI).
At baseline, the mean BSA was similar for patients enrolled in studies 216 and 215 (9.6% and 8.8%, respectively), and 80% of all patients had baseline IGA of moderate. By week 2, the mean roflumilast and N-oxide predose plasma concentrations among patients in the younger group were 2.15 and 22.4 ng/mL, compared with 3.15 and 28.9 ng/mL among those in the older group. At week 4, the mean roflumilast and N-oxide predose concentrations were 2.04 and 15.8 ng/mL in the younger group (study 216), compared with 1.68 and 15.7 ng/mL in the older group (study 215).
As for efficacy, 90% and 40% of patients in studies 216 and 215 achieved IGA success at week 4, respectively, while 90% and 50% achieved PASI-75, 90% and 40% achieved WI-NRS success, and the mean BSA reductions at week 4 were 79.1% and 44.4%. Meanwhile, one younger patient in study 216 reported a treatment-emergent adverse event (TEAE) of headache, which was considered mild, while four older patients in study 215 reported 8 TEAEs, which were considered mild and ranged from back pain to nasal congestion.
“The rapid onset of action was surprising but exceedingly rewarding for the subjects enrolled in the study,” Dr. Hebert told this news organization after the meeting. “The PASI scores and itch scores were markedly improved at the end of the 4-week clinical trial. Patient and parents alike were pleased to use a steroid-free option with once-daily application and rapid onset of action to help control plaque psoriasis.”
In the poster abstract, she and her coauthors concluded that “under maximal use conditions in children aged 2-11 years, roflumilast cream 0.3% was well tolerated and improved signs and symptoms of psoriasis with measured improvements in IGA score, PASI score, BSA involvement, CDLQI, and WI-NRS. Overall, pharmacokinetics, safety, tolerability, and efficacy in patients aged 2-11 years were consistent with prior results in adults and adolescents.”
The study was funded by Arcutis Biotherapeutics. Dr. Hebert reported that she is an investigator for Arcutis. About half the coauthors are employees of Arcutis, and the other half disclosed grants, research funding and/or honoraria from the company. Research grants from the company for this study were paid to the McGovern Medical School at the University of Texas.
FROM SPD 2023
What factors cause multiple biologic failure in psoriasis?
, results from a prospective cohort demonstrated.
“Prior cross-sectional and single-center studies have primarily analyzed therapeutic failure of a single biologic or biologics within one class,” researchers led by Wilson Liao, MD, professor and vice chair of research in the department of dermatology at the University of California, San Francisco, wrote in the study, published in the Journal of the American Academy of Dermatology. “However, failure of multiple biologics targeting different signaling pathways is common over the course of treatment. These ‘multiple biologic failure’ patients are not well-characterized, and the patterns of biologics attempted and sociodemographic or clinical features that may predict difficult treatment are incompletely studied.”
To bridge this gap, the researchers conducted a prospective cohort study from the CorEvitas Psoriasis Registry, which collected data from dermatologist-diagnosed patients with psoriasis who started or switched to a Food and Drug Administration (FDA)–approved systemic therapy for psoriasis during routine dermatology visits from April 15, 2015, to May 10, 2022. This period included data from 17,196 patients across 259 private and 209 academic sites from 580 physicians in the United States and Canada.
From this registry, Dr. Liao and colleagues identified 1,039 patients with 24 months or more of follow-up data, a confirmed index biologic start date, and valid baseline assessment data, and categorized them into three cohorts:
- 490 (47.2%) with good response (GR), defined as patients with 24 months or more of continued index biologic use by the last registry visit.
- 65 (6.3%) with multiple biologic failure (MBF), defined as patients administered two or more biologic agents of different mechanistic classes who discontinued these biologics because of physician-reported “inadequate initial response,” “failure to maintain initial response,” or “active disease” despite 90 or more days of use per biologic.
- 484 (46.6%) categorized as “other,” defined as patients failed by one biologic or who discontinued treatment for nonmedical reasons.
The researchers used multivariable logistic regression to identify sociodemographic, clinical, and patient-reported outcomes that differed between the MBF and GR groups. The mean age of the patients in the study was 49.1 years, 44.2% were female, 77.9% were White, 9.7% were Hispanic, and the mean duration of psoriasis was 11.5 years.
On multivariable logistic regression, factors associated with MBF, compared with those with GR, included female at birth (odds ratio [OR] = 2.29; confidence interval [CI], 1.11-4.72), history of hyperlipidemia (OR = 3.14; CI, 1.35-7.30), Medicaid insurance (OR = 4.53; CI, 1.40-14.60), prior nonbiologic systemic therapy (OR = 2.47; CI, 1.16-5.25), higher psoriasis duration (OR = 0.60 per standard deviation [SD]; CI, 0.38-0.94), and later index biologic initiation (OR = 0.37 per year; CI, 0.27-0.52). Sensitivity analysis revealed that the duration of prior nonbiologic systemic therapy use was not associated with MBF (OR = 0.99; CI, 0.94-1.02; P = 0.56).
“Interestingly, health-related behaviors (e.g., smoking, alcohol use) and location/extent of psoriasis were not important differentiators between MBF and GR,” the authors noted. “We might suspect these features to correlate with MBF, as numerous observational studies found associations between health-related behaviors or psoriasis severity and presence at difficult-to-treat locations, which often relates to biologic use.”
They acknowledged certain limitations of their study, including underrepresentation of ethnoracial minorities and male sex at birth relative to reported psoriasis epidemiology, “possibly reflecting participation bias and reduced access to specialty care, given that patients were enrolled into the registry by dermatologists,” they wrote. “Patient adherence to prescribed biologic regimens between registry visits was not evaluated.”
Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, who was asked to comment on the study, said that despite the rapid expansion in biologic therapies for psoriasis, “analysis of real-world use patterns and patient characteristics has been limited – particularly for those who have failed multiple treatments. These findings suggest that there indeed may be some key differences between patients who have had to cycle through multiple biologics versus those who have had a sustained satisfactory response on a single therapy, such as disease duration and previous nonbiologic treatments.”
However, he added, “while this prospective study utilized a robust approach to gather standard-of-care data across multiple clinical sites, the absolute number of patients with multiple biologic failures was low, and additional data for these kinds of patients are still highly needed.”
The study was sponsored by CorEvitas and supported through a partnership between CorEvitas and the National Psoriasis Foundation. Dr. Liao disclosed that he has received research grant funding from AbbVie, Amgen, Janssen, Leo, Novartis, Pfizer, Regeneron, and TRex Bio. Dr. Chovatiya disclosed ties with several pharmaceutical companies.
, results from a prospective cohort demonstrated.
“Prior cross-sectional and single-center studies have primarily analyzed therapeutic failure of a single biologic or biologics within one class,” researchers led by Wilson Liao, MD, professor and vice chair of research in the department of dermatology at the University of California, San Francisco, wrote in the study, published in the Journal of the American Academy of Dermatology. “However, failure of multiple biologics targeting different signaling pathways is common over the course of treatment. These ‘multiple biologic failure’ patients are not well-characterized, and the patterns of biologics attempted and sociodemographic or clinical features that may predict difficult treatment are incompletely studied.”
To bridge this gap, the researchers conducted a prospective cohort study from the CorEvitas Psoriasis Registry, which collected data from dermatologist-diagnosed patients with psoriasis who started or switched to a Food and Drug Administration (FDA)–approved systemic therapy for psoriasis during routine dermatology visits from April 15, 2015, to May 10, 2022. This period included data from 17,196 patients across 259 private and 209 academic sites from 580 physicians in the United States and Canada.
From this registry, Dr. Liao and colleagues identified 1,039 patients with 24 months or more of follow-up data, a confirmed index biologic start date, and valid baseline assessment data, and categorized them into three cohorts:
- 490 (47.2%) with good response (GR), defined as patients with 24 months or more of continued index biologic use by the last registry visit.
- 65 (6.3%) with multiple biologic failure (MBF), defined as patients administered two or more biologic agents of different mechanistic classes who discontinued these biologics because of physician-reported “inadequate initial response,” “failure to maintain initial response,” or “active disease” despite 90 or more days of use per biologic.
- 484 (46.6%) categorized as “other,” defined as patients failed by one biologic or who discontinued treatment for nonmedical reasons.
The researchers used multivariable logistic regression to identify sociodemographic, clinical, and patient-reported outcomes that differed between the MBF and GR groups. The mean age of the patients in the study was 49.1 years, 44.2% were female, 77.9% were White, 9.7% were Hispanic, and the mean duration of psoriasis was 11.5 years.
On multivariable logistic regression, factors associated with MBF, compared with those with GR, included female at birth (odds ratio [OR] = 2.29; confidence interval [CI], 1.11-4.72), history of hyperlipidemia (OR = 3.14; CI, 1.35-7.30), Medicaid insurance (OR = 4.53; CI, 1.40-14.60), prior nonbiologic systemic therapy (OR = 2.47; CI, 1.16-5.25), higher psoriasis duration (OR = 0.60 per standard deviation [SD]; CI, 0.38-0.94), and later index biologic initiation (OR = 0.37 per year; CI, 0.27-0.52). Sensitivity analysis revealed that the duration of prior nonbiologic systemic therapy use was not associated with MBF (OR = 0.99; CI, 0.94-1.02; P = 0.56).
“Interestingly, health-related behaviors (e.g., smoking, alcohol use) and location/extent of psoriasis were not important differentiators between MBF and GR,” the authors noted. “We might suspect these features to correlate with MBF, as numerous observational studies found associations between health-related behaviors or psoriasis severity and presence at difficult-to-treat locations, which often relates to biologic use.”
They acknowledged certain limitations of their study, including underrepresentation of ethnoracial minorities and male sex at birth relative to reported psoriasis epidemiology, “possibly reflecting participation bias and reduced access to specialty care, given that patients were enrolled into the registry by dermatologists,” they wrote. “Patient adherence to prescribed biologic regimens between registry visits was not evaluated.”
Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, who was asked to comment on the study, said that despite the rapid expansion in biologic therapies for psoriasis, “analysis of real-world use patterns and patient characteristics has been limited – particularly for those who have failed multiple treatments. These findings suggest that there indeed may be some key differences between patients who have had to cycle through multiple biologics versus those who have had a sustained satisfactory response on a single therapy, such as disease duration and previous nonbiologic treatments.”
However, he added, “while this prospective study utilized a robust approach to gather standard-of-care data across multiple clinical sites, the absolute number of patients with multiple biologic failures was low, and additional data for these kinds of patients are still highly needed.”
The study was sponsored by CorEvitas and supported through a partnership between CorEvitas and the National Psoriasis Foundation. Dr. Liao disclosed that he has received research grant funding from AbbVie, Amgen, Janssen, Leo, Novartis, Pfizer, Regeneron, and TRex Bio. Dr. Chovatiya disclosed ties with several pharmaceutical companies.
, results from a prospective cohort demonstrated.
“Prior cross-sectional and single-center studies have primarily analyzed therapeutic failure of a single biologic or biologics within one class,” researchers led by Wilson Liao, MD, professor and vice chair of research in the department of dermatology at the University of California, San Francisco, wrote in the study, published in the Journal of the American Academy of Dermatology. “However, failure of multiple biologics targeting different signaling pathways is common over the course of treatment. These ‘multiple biologic failure’ patients are not well-characterized, and the patterns of biologics attempted and sociodemographic or clinical features that may predict difficult treatment are incompletely studied.”
To bridge this gap, the researchers conducted a prospective cohort study from the CorEvitas Psoriasis Registry, which collected data from dermatologist-diagnosed patients with psoriasis who started or switched to a Food and Drug Administration (FDA)–approved systemic therapy for psoriasis during routine dermatology visits from April 15, 2015, to May 10, 2022. This period included data from 17,196 patients across 259 private and 209 academic sites from 580 physicians in the United States and Canada.
From this registry, Dr. Liao and colleagues identified 1,039 patients with 24 months or more of follow-up data, a confirmed index biologic start date, and valid baseline assessment data, and categorized them into three cohorts:
- 490 (47.2%) with good response (GR), defined as patients with 24 months or more of continued index biologic use by the last registry visit.
- 65 (6.3%) with multiple biologic failure (MBF), defined as patients administered two or more biologic agents of different mechanistic classes who discontinued these biologics because of physician-reported “inadequate initial response,” “failure to maintain initial response,” or “active disease” despite 90 or more days of use per biologic.
- 484 (46.6%) categorized as “other,” defined as patients failed by one biologic or who discontinued treatment for nonmedical reasons.
The researchers used multivariable logistic regression to identify sociodemographic, clinical, and patient-reported outcomes that differed between the MBF and GR groups. The mean age of the patients in the study was 49.1 years, 44.2% were female, 77.9% were White, 9.7% were Hispanic, and the mean duration of psoriasis was 11.5 years.
On multivariable logistic regression, factors associated with MBF, compared with those with GR, included female at birth (odds ratio [OR] = 2.29; confidence interval [CI], 1.11-4.72), history of hyperlipidemia (OR = 3.14; CI, 1.35-7.30), Medicaid insurance (OR = 4.53; CI, 1.40-14.60), prior nonbiologic systemic therapy (OR = 2.47; CI, 1.16-5.25), higher psoriasis duration (OR = 0.60 per standard deviation [SD]; CI, 0.38-0.94), and later index biologic initiation (OR = 0.37 per year; CI, 0.27-0.52). Sensitivity analysis revealed that the duration of prior nonbiologic systemic therapy use was not associated with MBF (OR = 0.99; CI, 0.94-1.02; P = 0.56).
“Interestingly, health-related behaviors (e.g., smoking, alcohol use) and location/extent of psoriasis were not important differentiators between MBF and GR,” the authors noted. “We might suspect these features to correlate with MBF, as numerous observational studies found associations between health-related behaviors or psoriasis severity and presence at difficult-to-treat locations, which often relates to biologic use.”
They acknowledged certain limitations of their study, including underrepresentation of ethnoracial minorities and male sex at birth relative to reported psoriasis epidemiology, “possibly reflecting participation bias and reduced access to specialty care, given that patients were enrolled into the registry by dermatologists,” they wrote. “Patient adherence to prescribed biologic regimens between registry visits was not evaluated.”
Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, who was asked to comment on the study, said that despite the rapid expansion in biologic therapies for psoriasis, “analysis of real-world use patterns and patient characteristics has been limited – particularly for those who have failed multiple treatments. These findings suggest that there indeed may be some key differences between patients who have had to cycle through multiple biologics versus those who have had a sustained satisfactory response on a single therapy, such as disease duration and previous nonbiologic treatments.”
However, he added, “while this prospective study utilized a robust approach to gather standard-of-care data across multiple clinical sites, the absolute number of patients with multiple biologic failures was low, and additional data for these kinds of patients are still highly needed.”
The study was sponsored by CorEvitas and supported through a partnership between CorEvitas and the National Psoriasis Foundation. Dr. Liao disclosed that he has received research grant funding from AbbVie, Amgen, Janssen, Leo, Novartis, Pfizer, Regeneron, and TRex Bio. Dr. Chovatiya disclosed ties with several pharmaceutical companies.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Could risk stratifying methotrexate users lead to less frequent testing?
A new model can predict which patients are more likely to experience side effects from long-term methotrexate (MTX) use, research suggests. Patients with a lower risk profile may benefit from less frequent testing, the authors hypothesize.
Most recommendations advise that patients initiating MTX therapy should get blood testing every 2-4 weeks to monitor for full blood count, liver function, urea electrolytes, and creatinine. After 6 months taking MTX, monitoring can be tapered to every 3 months. But Abhishek Abhishek, MD, PhD, professor of rheumatology and honorary consultant rheumatologist at Nottingham (England) University Hospitals NHS Trust and colleagues argue that abnormal results after the initial 6 months of treatment are “infrequent,” and patients may benefit from fewer tests throughout the year.
“Unnecessary blood tests waste patients’ time and health care resources, including the time of general practitioners and phlebotomists,” Dr. Abhishek and associates write. “It would be beneficial to predict the risk of clinically significant abnormal blood test results during long-term methotrexate treatment to inform the frequency of testing for individuals.”
Stratifying risk
In the study, published in the BMJ, researchers used the UK’s Clinical Practice Research Datalink (CPRD) to identify the electronic medical records of over 37,000 adult patients with an immune-mediated inflammatory disease who were prescribed MTX during 2007-2019. All included patients were prescribed MTX for at least 6 months. The main outcome was discontinuation of methotrexate because of abnormal blood test results. Around 62% of patients had rheumatoid arthritis and 22% had psoriasis or psoriatic arthritis.
Using these anonymized data, the group developed a risk stratification model using 11 clinical predictors. “The factors that went in the model are simple things that most patients can self-report or doctors can get from their patient’s medical records,” Dr. Abhishek told this news organization, including methotrexate dose, age, sex, and comorbidities. Dr. Abhishek emphasized that the model should be used only in patients who have continued taking MTX for at least 6 months and have already undergone more frequent initial testing.
The strongest individual predictors were diabetes (hazard ratio, 1.25), chronic kidney disease stage 3 (HR, 2.01), and previous cytopenia or raised liver enzyme levels during the first 6 months of MTX therapy (HR, 2.97). However, Dr. Abhishek emphasized that the individual factors were less important, noting that the model sums the risks to predict outcomes more accurately. Most patients (68.4%) were sorted into the low-risk cohort, with a less than 10% estimated risk of discontinuing MTX over the next 5 years. About one-fifth (20.9%) were categorized as moderate risk (10%-20% estimated risk over 5 years), and 10.7% were high risk, with a greater than 20% estimated risk of discontinuing the drug over 5 years.
The authors argue that low-risk patients could receive less frequent testing – perhaps every 6 months or annually, while moderate-risk patients would continue to be tested every 3 months. High-risk patients could potentially be tested with even greater frequently.
More research needed
The research involved “incredibly sophisticated statistical analysis,” said Daniel E. Furst, MD, professor emeritus of medicine at the University of California, Los Angeles, who was not involved with the study. However, the data do not yet support altering blood testing frequency based on this model.
“The hypothesis that not all patients have to be examined so frequently is a very reasonable hypothesis,” Dr. Furst said in an interview, and additional research is needed to corroborate it. The model also needs to be validated in patient populations outside of the United Kingdom, he added.
Dr. Abhishek agreed that validating the model in other patient populations is an important next step. “When we develop a tool [using] a one-nation data set, we want other researchers to then validate it in other countries’ data sets to make sure there is nothing odd about patients in the U.K. that makes the tool work well here but not in [the] U.S., Europe, or Asia, for example,” he said. Doing so should be relatively easy, he said, as the model is publicly available, and the information required is routinely collected during clinic visits.
To understand if less frequent testing might be appropriate for some patients, researchers would need to look at data registries like the Brigham and Women’s Hospital Rheumatoid Arthritis Sequential Study (BRASS) registry or CorEvitas registries “where the testing is done in a very regular way over the long haul,” Dr. Furst said. Analyzing these datasets, researchers could determine the testing intervals that would be most efficient for low- and high-risk patients.
A word of caution
While less frequent testing for long-term MTX therapy could likely have benefits, there is still some risk involved, cautioned Prabha Ranganathan, MD, professor of medicine at Washington University in St. Louis.
“Although most methotrexate toxicity occurs within the first 6 months of starting treatment, rare idiosyncratic toxicity can occur that does not correlate with the dose, duration, or method of how methotrexate is administered,” she wrote in an accompanying editorial. “Most rheumatologists can identify a handful of patients who receive methotrexate in their practice who develop sudden leukopenia or thrombocytopenia or transaminitis that is severe enough to warrant drug discontinuation.” While tools like this prediction model can be useful, clinicians need to consider each patient individually and use shared decision-making when monitoring for MTX toxicity, she advised.
“As in most of areas of medicine, the one-size-fits-all approach does not work for methotrexate users,” she noted.
This study was funded by the U.K. National Institute for Health and Care Research and Health Technology Assessment. Dr. Abhishek has received institutional research grants from AstraZeneca and Oxford Immunotech and personal fees from UpToDate, Springer, Cadila Pharmaceuticals, NGM Bio, Limbic, and Inflazome. Dr. Furst and Dr. Ranganathan report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new model can predict which patients are more likely to experience side effects from long-term methotrexate (MTX) use, research suggests. Patients with a lower risk profile may benefit from less frequent testing, the authors hypothesize.
Most recommendations advise that patients initiating MTX therapy should get blood testing every 2-4 weeks to monitor for full blood count, liver function, urea electrolytes, and creatinine. After 6 months taking MTX, monitoring can be tapered to every 3 months. But Abhishek Abhishek, MD, PhD, professor of rheumatology and honorary consultant rheumatologist at Nottingham (England) University Hospitals NHS Trust and colleagues argue that abnormal results after the initial 6 months of treatment are “infrequent,” and patients may benefit from fewer tests throughout the year.
“Unnecessary blood tests waste patients’ time and health care resources, including the time of general practitioners and phlebotomists,” Dr. Abhishek and associates write. “It would be beneficial to predict the risk of clinically significant abnormal blood test results during long-term methotrexate treatment to inform the frequency of testing for individuals.”
Stratifying risk
In the study, published in the BMJ, researchers used the UK’s Clinical Practice Research Datalink (CPRD) to identify the electronic medical records of over 37,000 adult patients with an immune-mediated inflammatory disease who were prescribed MTX during 2007-2019. All included patients were prescribed MTX for at least 6 months. The main outcome was discontinuation of methotrexate because of abnormal blood test results. Around 62% of patients had rheumatoid arthritis and 22% had psoriasis or psoriatic arthritis.
Using these anonymized data, the group developed a risk stratification model using 11 clinical predictors. “The factors that went in the model are simple things that most patients can self-report or doctors can get from their patient’s medical records,” Dr. Abhishek told this news organization, including methotrexate dose, age, sex, and comorbidities. Dr. Abhishek emphasized that the model should be used only in patients who have continued taking MTX for at least 6 months and have already undergone more frequent initial testing.
The strongest individual predictors were diabetes (hazard ratio, 1.25), chronic kidney disease stage 3 (HR, 2.01), and previous cytopenia or raised liver enzyme levels during the first 6 months of MTX therapy (HR, 2.97). However, Dr. Abhishek emphasized that the individual factors were less important, noting that the model sums the risks to predict outcomes more accurately. Most patients (68.4%) were sorted into the low-risk cohort, with a less than 10% estimated risk of discontinuing MTX over the next 5 years. About one-fifth (20.9%) were categorized as moderate risk (10%-20% estimated risk over 5 years), and 10.7% were high risk, with a greater than 20% estimated risk of discontinuing the drug over 5 years.
The authors argue that low-risk patients could receive less frequent testing – perhaps every 6 months or annually, while moderate-risk patients would continue to be tested every 3 months. High-risk patients could potentially be tested with even greater frequently.
More research needed
The research involved “incredibly sophisticated statistical analysis,” said Daniel E. Furst, MD, professor emeritus of medicine at the University of California, Los Angeles, who was not involved with the study. However, the data do not yet support altering blood testing frequency based on this model.
“The hypothesis that not all patients have to be examined so frequently is a very reasonable hypothesis,” Dr. Furst said in an interview, and additional research is needed to corroborate it. The model also needs to be validated in patient populations outside of the United Kingdom, he added.
Dr. Abhishek agreed that validating the model in other patient populations is an important next step. “When we develop a tool [using] a one-nation data set, we want other researchers to then validate it in other countries’ data sets to make sure there is nothing odd about patients in the U.K. that makes the tool work well here but not in [the] U.S., Europe, or Asia, for example,” he said. Doing so should be relatively easy, he said, as the model is publicly available, and the information required is routinely collected during clinic visits.
To understand if less frequent testing might be appropriate for some patients, researchers would need to look at data registries like the Brigham and Women’s Hospital Rheumatoid Arthritis Sequential Study (BRASS) registry or CorEvitas registries “where the testing is done in a very regular way over the long haul,” Dr. Furst said. Analyzing these datasets, researchers could determine the testing intervals that would be most efficient for low- and high-risk patients.
A word of caution
While less frequent testing for long-term MTX therapy could likely have benefits, there is still some risk involved, cautioned Prabha Ranganathan, MD, professor of medicine at Washington University in St. Louis.
“Although most methotrexate toxicity occurs within the first 6 months of starting treatment, rare idiosyncratic toxicity can occur that does not correlate with the dose, duration, or method of how methotrexate is administered,” she wrote in an accompanying editorial. “Most rheumatologists can identify a handful of patients who receive methotrexate in their practice who develop sudden leukopenia or thrombocytopenia or transaminitis that is severe enough to warrant drug discontinuation.” While tools like this prediction model can be useful, clinicians need to consider each patient individually and use shared decision-making when monitoring for MTX toxicity, she advised.
“As in most of areas of medicine, the one-size-fits-all approach does not work for methotrexate users,” she noted.
This study was funded by the U.K. National Institute for Health and Care Research and Health Technology Assessment. Dr. Abhishek has received institutional research grants from AstraZeneca and Oxford Immunotech and personal fees from UpToDate, Springer, Cadila Pharmaceuticals, NGM Bio, Limbic, and Inflazome. Dr. Furst and Dr. Ranganathan report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new model can predict which patients are more likely to experience side effects from long-term methotrexate (MTX) use, research suggests. Patients with a lower risk profile may benefit from less frequent testing, the authors hypothesize.
Most recommendations advise that patients initiating MTX therapy should get blood testing every 2-4 weeks to monitor for full blood count, liver function, urea electrolytes, and creatinine. After 6 months taking MTX, monitoring can be tapered to every 3 months. But Abhishek Abhishek, MD, PhD, professor of rheumatology and honorary consultant rheumatologist at Nottingham (England) University Hospitals NHS Trust and colleagues argue that abnormal results after the initial 6 months of treatment are “infrequent,” and patients may benefit from fewer tests throughout the year.
“Unnecessary blood tests waste patients’ time and health care resources, including the time of general practitioners and phlebotomists,” Dr. Abhishek and associates write. “It would be beneficial to predict the risk of clinically significant abnormal blood test results during long-term methotrexate treatment to inform the frequency of testing for individuals.”
Stratifying risk
In the study, published in the BMJ, researchers used the UK’s Clinical Practice Research Datalink (CPRD) to identify the electronic medical records of over 37,000 adult patients with an immune-mediated inflammatory disease who were prescribed MTX during 2007-2019. All included patients were prescribed MTX for at least 6 months. The main outcome was discontinuation of methotrexate because of abnormal blood test results. Around 62% of patients had rheumatoid arthritis and 22% had psoriasis or psoriatic arthritis.
Using these anonymized data, the group developed a risk stratification model using 11 clinical predictors. “The factors that went in the model are simple things that most patients can self-report or doctors can get from their patient’s medical records,” Dr. Abhishek told this news organization, including methotrexate dose, age, sex, and comorbidities. Dr. Abhishek emphasized that the model should be used only in patients who have continued taking MTX for at least 6 months and have already undergone more frequent initial testing.
The strongest individual predictors were diabetes (hazard ratio, 1.25), chronic kidney disease stage 3 (HR, 2.01), and previous cytopenia or raised liver enzyme levels during the first 6 months of MTX therapy (HR, 2.97). However, Dr. Abhishek emphasized that the individual factors were less important, noting that the model sums the risks to predict outcomes more accurately. Most patients (68.4%) were sorted into the low-risk cohort, with a less than 10% estimated risk of discontinuing MTX over the next 5 years. About one-fifth (20.9%) were categorized as moderate risk (10%-20% estimated risk over 5 years), and 10.7% were high risk, with a greater than 20% estimated risk of discontinuing the drug over 5 years.
The authors argue that low-risk patients could receive less frequent testing – perhaps every 6 months or annually, while moderate-risk patients would continue to be tested every 3 months. High-risk patients could potentially be tested with even greater frequently.
More research needed
The research involved “incredibly sophisticated statistical analysis,” said Daniel E. Furst, MD, professor emeritus of medicine at the University of California, Los Angeles, who was not involved with the study. However, the data do not yet support altering blood testing frequency based on this model.
“The hypothesis that not all patients have to be examined so frequently is a very reasonable hypothesis,” Dr. Furst said in an interview, and additional research is needed to corroborate it. The model also needs to be validated in patient populations outside of the United Kingdom, he added.
Dr. Abhishek agreed that validating the model in other patient populations is an important next step. “When we develop a tool [using] a one-nation data set, we want other researchers to then validate it in other countries’ data sets to make sure there is nothing odd about patients in the U.K. that makes the tool work well here but not in [the] U.S., Europe, or Asia, for example,” he said. Doing so should be relatively easy, he said, as the model is publicly available, and the information required is routinely collected during clinic visits.
To understand if less frequent testing might be appropriate for some patients, researchers would need to look at data registries like the Brigham and Women’s Hospital Rheumatoid Arthritis Sequential Study (BRASS) registry or CorEvitas registries “where the testing is done in a very regular way over the long haul,” Dr. Furst said. Analyzing these datasets, researchers could determine the testing intervals that would be most efficient for low- and high-risk patients.
A word of caution
While less frequent testing for long-term MTX therapy could likely have benefits, there is still some risk involved, cautioned Prabha Ranganathan, MD, professor of medicine at Washington University in St. Louis.
“Although most methotrexate toxicity occurs within the first 6 months of starting treatment, rare idiosyncratic toxicity can occur that does not correlate with the dose, duration, or method of how methotrexate is administered,” she wrote in an accompanying editorial. “Most rheumatologists can identify a handful of patients who receive methotrexate in their practice who develop sudden leukopenia or thrombocytopenia or transaminitis that is severe enough to warrant drug discontinuation.” While tools like this prediction model can be useful, clinicians need to consider each patient individually and use shared decision-making when monitoring for MTX toxicity, she advised.
“As in most of areas of medicine, the one-size-fits-all approach does not work for methotrexate users,” she noted.
This study was funded by the U.K. National Institute for Health and Care Research and Health Technology Assessment. Dr. Abhishek has received institutional research grants from AstraZeneca and Oxford Immunotech and personal fees from UpToDate, Springer, Cadila Pharmaceuticals, NGM Bio, Limbic, and Inflazome. Dr. Furst and Dr. Ranganathan report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE BMJ
Vitamin D deficiency linked to psoriasis severity
, suggesting that some people who increase their intake of the vitamin could better control this skin condition that affects up to 8 million people in the United States alone.
Brown University researchers studied almost 500 psoriasis cases taken from the National Health and Nutrition Examination Survey (NHANES), the scientists told attendees at the conference of the American Society for Nutrition. They compared the peoples’ reports on how much of their body surface was affected by psoriasis to vitamin D levels collected in blood samples.
“After adjusting for lifestyle factors such as smoking, the analysis showed that lower vitamin D levels and vitamin D deficiency were significantly associated with greater psoriasis severity,” the ASN said in a news release. “The researchers also found that patients with the least amount of body surface affected by psoriasis had the highest average vitamin D levels while those with the greatest affected area had the lowest average levels of vitamin D.”
The researchers said that people with psoriasis might improve their condition by getting more vitamin D in their diet and through supplements.
“Topical synthetic vitamin D creams are emerging as new therapies for psoriasis, but these usually require a doctor’s prescription,” said researcher Rachel K. Lim, an MD candidate at Brown University, Providence, R.I. “Our results suggest that a vitamin D–rich diet or oral vitamin D supplementation may also provide some benefit to psoriasis patients.”
The researchers said that vitamin D toxicity is rare but that people should consult with their medical caregivers before they start taking supplements.
A version of this article first appeared on WebMD.com.
, suggesting that some people who increase their intake of the vitamin could better control this skin condition that affects up to 8 million people in the United States alone.
Brown University researchers studied almost 500 psoriasis cases taken from the National Health and Nutrition Examination Survey (NHANES), the scientists told attendees at the conference of the American Society for Nutrition. They compared the peoples’ reports on how much of their body surface was affected by psoriasis to vitamin D levels collected in blood samples.
“After adjusting for lifestyle factors such as smoking, the analysis showed that lower vitamin D levels and vitamin D deficiency were significantly associated with greater psoriasis severity,” the ASN said in a news release. “The researchers also found that patients with the least amount of body surface affected by psoriasis had the highest average vitamin D levels while those with the greatest affected area had the lowest average levels of vitamin D.”
The researchers said that people with psoriasis might improve their condition by getting more vitamin D in their diet and through supplements.
“Topical synthetic vitamin D creams are emerging as new therapies for psoriasis, but these usually require a doctor’s prescription,” said researcher Rachel K. Lim, an MD candidate at Brown University, Providence, R.I. “Our results suggest that a vitamin D–rich diet or oral vitamin D supplementation may also provide some benefit to psoriasis patients.”
The researchers said that vitamin D toxicity is rare but that people should consult with their medical caregivers before they start taking supplements.
A version of this article first appeared on WebMD.com.
, suggesting that some people who increase their intake of the vitamin could better control this skin condition that affects up to 8 million people in the United States alone.
Brown University researchers studied almost 500 psoriasis cases taken from the National Health and Nutrition Examination Survey (NHANES), the scientists told attendees at the conference of the American Society for Nutrition. They compared the peoples’ reports on how much of their body surface was affected by psoriasis to vitamin D levels collected in blood samples.
“After adjusting for lifestyle factors such as smoking, the analysis showed that lower vitamin D levels and vitamin D deficiency were significantly associated with greater psoriasis severity,” the ASN said in a news release. “The researchers also found that patients with the least amount of body surface affected by psoriasis had the highest average vitamin D levels while those with the greatest affected area had the lowest average levels of vitamin D.”
The researchers said that people with psoriasis might improve their condition by getting more vitamin D in their diet and through supplements.
“Topical synthetic vitamin D creams are emerging as new therapies for psoriasis, but these usually require a doctor’s prescription,” said researcher Rachel K. Lim, an MD candidate at Brown University, Providence, R.I. “Our results suggest that a vitamin D–rich diet or oral vitamin D supplementation may also provide some benefit to psoriasis patients.”
The researchers said that vitamin D toxicity is rare but that people should consult with their medical caregivers before they start taking supplements.
A version of this article first appeared on WebMD.com.
FROM NUTRITION 2023
Humira biosimilars: Five things to know
The best-selling drug Humira (adalimumab) now faces competition in the United States after a 20-year monopoly. The first adalimumab biosimilar, Amjevita, launched in the United States on January 31, and in July, seven additional biosimilars became available. These drugs have the potential to lower prescription drug prices, but when and by how much remains to be seen.
Here’s what you need to know about adalimumab biosimilars.
What Humira biosimilars are now available?
Eight different biosimilars have launched in 2023 with discounts as large at 85% from Humira’s list price of $6,922. A few companies also offer two price points.
Three of these biosimilars – Hadlima, Hyrimoz, and Yuflyma – are available in high concentration formulations. This high concentration formulation makes up 85% of Humira prescriptions, according to a report from Goodroot, a collection of companies focused on lowering health care costs.
Cyltezo is currently the only adalimumab biosimilar with an interchangeability designation, meaning that a pharmacist can substitute the biosimilar for an equivalent Humira prescription without the intervention of a clinician. A total of 47 states allow for these substitutions without prior approval from a clinician, according to Goodroot, and the clinician must be notified of the switch within a certain time frame. A total of 40 states require that patients be notified of the switch before substitution.
However, it’s not clear if this interchangeability designation will prove an advantage for Cyltezo, as it is interchangeable with the lower concentration version of Humira that makes up just 15% of prescriptions.
Most of the companies behind these biosimilars are pursuing interchangeability designations for their drugs, except for Fresenius Kabi (Idacio) and Coherus (Yusimry).
A ninth biosimilar, Pfizer’s adalimumab-afzb (Abrilada), is not yet on the market and is currently awaiting an approval decision from the Food and Drug Administration to add an interchangeability designation to its prior approval for a low-concentration formulation.
Why are they priced differently?
The two price points offer different deals to payers. Pharmacy benefit managers make confidential agreements with drug manufacturers to get a discount – called a rebate – to get the drug on the PBM’s formulary. The PBM keeps a portion of that rebate, and the rest is passed on to the insurance company and patients. Biosimilars at a higher price point will likely offer larger rebates. Biosimilars offered at lower price points incorporate this discount up front in their list pricing and likely will not offer large rebates.
Will biosimilars be covered by payers?
Currently, biosimilars are being offered on formularies at parity with Humira, meaning they are on the same tier. The PBM companies OptumRx and Cigna Group’s Express Scripts will offer Amjevita (at both price points), Cyltezo, and Hyrimoz (at both price points).
“This decision allows our clients flexibility to provide access to the lower list price, so members in high-deductible plans and benefit designs with coinsurance can experience lower out-of-pocket costs,” said OptumRx spokesperson Isaac Sorensen in an email.
Mark Cuban Cost Plus Drug Company, which uses a direct-to-consumer model, will offer Yusimry for $567.27 on its website. SmithRx, a PBM based in San Francisco, announced it would partner with Cost Plus Drugs to offer Yusimry, adding that SmithRx members can use their insurance benefits to further reduce out-of-pocket costs. RxPreferred, another PBM, will also offer Yusimry through its partnership with Cuban’s company.
The news website Formulary Watch previously reported that CVS Caremark, another of the biggest PBMs, will be offering Amjevita, but as a nonpreferred brand, while Humira remains the preferred brand. CVS Caremark did not respond to a request for comment.
Will patients pay less?
Biosimilars have been touted as a potential solution to lower spending on biologic drugs, but it’s unknown if patients will ultimately benefit with lower out-of-pocket costs. It’s “impossible to predict” if the discount that third-party payers pay will be passed on to consumers, said Mark Fendrick, MD, who directs the University of Michigan Center for Value-based Insurance Design in Ann Arbor.
Generally, a consumer’s copay is a percentage of a drug’s list price, so it stands to reason that a low drug price would result in lower out-of-pocket payments. While this is mostly true, Humira has a successful copay assistance program to lower prescription costs for consumers. According to a 2022 IQVIA report, 82% of commercial prescriptions cost patients less than $10 for Humira because of this program.
To appeal to patients, biosimilar companies will need to offer similar savings, Dr. Fendrick added. “There will be some discontent if patients are actually asked to pay more out-of-pocket for a less expensive drug,” he said.
All eight companies behind these biosimilars are offering or will be launching copay saving programs, many which advertise copays as low as $0 per month for eligible patients.
How will Humira respond?
Marta Wosińska, PhD, a health care economist at the Brookings Institute, Washington, predicts payers will use these lower biosimilar prices to negotiate better deals with AbbVie, Humira’s manufacturer. “We have a lot of players coming into [the market] right now, so the competition is really fierce,” she said. In response, AbbVie will need to increase rebates on Humira and/or lower its price to compete with these biosimilars.
“The ball is in AbbVie’s court,” she said. “If [the company] is not willing to drop price sufficiently, then payers will start switching to biosimilars.”
Dr. Fendrick reported past financial relationships and consulting arrangements with AbbVie, Amgen, Arnold Ventures, Bayer, CareFirst, BlueCross BlueShield, and many other companies. Dr. Wosińska has received funding from Arnold Ventures and serves as an expert witness on antitrust cases involving generic medication.
A version of this article first appeared on Medscape.com.
The best-selling drug Humira (adalimumab) now faces competition in the United States after a 20-year monopoly. The first adalimumab biosimilar, Amjevita, launched in the United States on January 31, and in July, seven additional biosimilars became available. These drugs have the potential to lower prescription drug prices, but when and by how much remains to be seen.
Here’s what you need to know about adalimumab biosimilars.
What Humira biosimilars are now available?
Eight different biosimilars have launched in 2023 with discounts as large at 85% from Humira’s list price of $6,922. A few companies also offer two price points.

Three of these biosimilars – Hadlima, Hyrimoz, and Yuflyma – are available in high concentration formulations. This high concentration formulation makes up 85% of Humira prescriptions, according to a report from Goodroot, a collection of companies focused on lowering health care costs.
Cyltezo is currently the only adalimumab biosimilar with an interchangeability designation, meaning that a pharmacist can substitute the biosimilar for an equivalent Humira prescription without the intervention of a clinician. A total of 47 states allow for these substitutions without prior approval from a clinician, according to Goodroot, and the clinician must be notified of the switch within a certain time frame. A total of 40 states require that patients be notified of the switch before substitution.
However, it’s not clear if this interchangeability designation will prove an advantage for Cyltezo, as it is interchangeable with the lower concentration version of Humira that makes up just 15% of prescriptions.
Most of the companies behind these biosimilars are pursuing interchangeability designations for their drugs, except for Fresenius Kabi (Idacio) and Coherus (Yusimry).
A ninth biosimilar, Pfizer’s adalimumab-afzb (Abrilada), is not yet on the market and is currently awaiting an approval decision from the Food and Drug Administration to add an interchangeability designation to its prior approval for a low-concentration formulation.
Why are they priced differently?
The two price points offer different deals to payers. Pharmacy benefit managers make confidential agreements with drug manufacturers to get a discount – called a rebate – to get the drug on the PBM’s formulary. The PBM keeps a portion of that rebate, and the rest is passed on to the insurance company and patients. Biosimilars at a higher price point will likely offer larger rebates. Biosimilars offered at lower price points incorporate this discount up front in their list pricing and likely will not offer large rebates.
Will biosimilars be covered by payers?
Currently, biosimilars are being offered on formularies at parity with Humira, meaning they are on the same tier. The PBM companies OptumRx and Cigna Group’s Express Scripts will offer Amjevita (at both price points), Cyltezo, and Hyrimoz (at both price points).
“This decision allows our clients flexibility to provide access to the lower list price, so members in high-deductible plans and benefit designs with coinsurance can experience lower out-of-pocket costs,” said OptumRx spokesperson Isaac Sorensen in an email.
Mark Cuban Cost Plus Drug Company, which uses a direct-to-consumer model, will offer Yusimry for $567.27 on its website. SmithRx, a PBM based in San Francisco, announced it would partner with Cost Plus Drugs to offer Yusimry, adding that SmithRx members can use their insurance benefits to further reduce out-of-pocket costs. RxPreferred, another PBM, will also offer Yusimry through its partnership with Cuban’s company.
The news website Formulary Watch previously reported that CVS Caremark, another of the biggest PBMs, will be offering Amjevita, but as a nonpreferred brand, while Humira remains the preferred brand. CVS Caremark did not respond to a request for comment.
Will patients pay less?
Biosimilars have been touted as a potential solution to lower spending on biologic drugs, but it’s unknown if patients will ultimately benefit with lower out-of-pocket costs. It’s “impossible to predict” if the discount that third-party payers pay will be passed on to consumers, said Mark Fendrick, MD, who directs the University of Michigan Center for Value-based Insurance Design in Ann Arbor.
Generally, a consumer’s copay is a percentage of a drug’s list price, so it stands to reason that a low drug price would result in lower out-of-pocket payments. While this is mostly true, Humira has a successful copay assistance program to lower prescription costs for consumers. According to a 2022 IQVIA report, 82% of commercial prescriptions cost patients less than $10 for Humira because of this program.
To appeal to patients, biosimilar companies will need to offer similar savings, Dr. Fendrick added. “There will be some discontent if patients are actually asked to pay more out-of-pocket for a less expensive drug,” he said.
All eight companies behind these biosimilars are offering or will be launching copay saving programs, many which advertise copays as low as $0 per month for eligible patients.
How will Humira respond?
Marta Wosińska, PhD, a health care economist at the Brookings Institute, Washington, predicts payers will use these lower biosimilar prices to negotiate better deals with AbbVie, Humira’s manufacturer. “We have a lot of players coming into [the market] right now, so the competition is really fierce,” she said. In response, AbbVie will need to increase rebates on Humira and/or lower its price to compete with these biosimilars.
“The ball is in AbbVie’s court,” she said. “If [the company] is not willing to drop price sufficiently, then payers will start switching to biosimilars.”
Dr. Fendrick reported past financial relationships and consulting arrangements with AbbVie, Amgen, Arnold Ventures, Bayer, CareFirst, BlueCross BlueShield, and many other companies. Dr. Wosińska has received funding from Arnold Ventures and serves as an expert witness on antitrust cases involving generic medication.
A version of this article first appeared on Medscape.com.
The best-selling drug Humira (adalimumab) now faces competition in the United States after a 20-year monopoly. The first adalimumab biosimilar, Amjevita, launched in the United States on January 31, and in July, seven additional biosimilars became available. These drugs have the potential to lower prescription drug prices, but when and by how much remains to be seen.
Here’s what you need to know about adalimumab biosimilars.
What Humira biosimilars are now available?
Eight different biosimilars have launched in 2023 with discounts as large at 85% from Humira’s list price of $6,922. A few companies also offer two price points.

Three of these biosimilars – Hadlima, Hyrimoz, and Yuflyma – are available in high concentration formulations. This high concentration formulation makes up 85% of Humira prescriptions, according to a report from Goodroot, a collection of companies focused on lowering health care costs.
Cyltezo is currently the only adalimumab biosimilar with an interchangeability designation, meaning that a pharmacist can substitute the biosimilar for an equivalent Humira prescription without the intervention of a clinician. A total of 47 states allow for these substitutions without prior approval from a clinician, according to Goodroot, and the clinician must be notified of the switch within a certain time frame. A total of 40 states require that patients be notified of the switch before substitution.
However, it’s not clear if this interchangeability designation will prove an advantage for Cyltezo, as it is interchangeable with the lower concentration version of Humira that makes up just 15% of prescriptions.
Most of the companies behind these biosimilars are pursuing interchangeability designations for their drugs, except for Fresenius Kabi (Idacio) and Coherus (Yusimry).
A ninth biosimilar, Pfizer’s adalimumab-afzb (Abrilada), is not yet on the market and is currently awaiting an approval decision from the Food and Drug Administration to add an interchangeability designation to its prior approval for a low-concentration formulation.
Why are they priced differently?
The two price points offer different deals to payers. Pharmacy benefit managers make confidential agreements with drug manufacturers to get a discount – called a rebate – to get the drug on the PBM’s formulary. The PBM keeps a portion of that rebate, and the rest is passed on to the insurance company and patients. Biosimilars at a higher price point will likely offer larger rebates. Biosimilars offered at lower price points incorporate this discount up front in their list pricing and likely will not offer large rebates.
Will biosimilars be covered by payers?
Currently, biosimilars are being offered on formularies at parity with Humira, meaning they are on the same tier. The PBM companies OptumRx and Cigna Group’s Express Scripts will offer Amjevita (at both price points), Cyltezo, and Hyrimoz (at both price points).
“This decision allows our clients flexibility to provide access to the lower list price, so members in high-deductible plans and benefit designs with coinsurance can experience lower out-of-pocket costs,” said OptumRx spokesperson Isaac Sorensen in an email.
Mark Cuban Cost Plus Drug Company, which uses a direct-to-consumer model, will offer Yusimry for $567.27 on its website. SmithRx, a PBM based in San Francisco, announced it would partner with Cost Plus Drugs to offer Yusimry, adding that SmithRx members can use their insurance benefits to further reduce out-of-pocket costs. RxPreferred, another PBM, will also offer Yusimry through its partnership with Cuban’s company.
The news website Formulary Watch previously reported that CVS Caremark, another of the biggest PBMs, will be offering Amjevita, but as a nonpreferred brand, while Humira remains the preferred brand. CVS Caremark did not respond to a request for comment.
Will patients pay less?
Biosimilars have been touted as a potential solution to lower spending on biologic drugs, but it’s unknown if patients will ultimately benefit with lower out-of-pocket costs. It’s “impossible to predict” if the discount that third-party payers pay will be passed on to consumers, said Mark Fendrick, MD, who directs the University of Michigan Center for Value-based Insurance Design in Ann Arbor.
Generally, a consumer’s copay is a percentage of a drug’s list price, so it stands to reason that a low drug price would result in lower out-of-pocket payments. While this is mostly true, Humira has a successful copay assistance program to lower prescription costs for consumers. According to a 2022 IQVIA report, 82% of commercial prescriptions cost patients less than $10 for Humira because of this program.
To appeal to patients, biosimilar companies will need to offer similar savings, Dr. Fendrick added. “There will be some discontent if patients are actually asked to pay more out-of-pocket for a less expensive drug,” he said.
All eight companies behind these biosimilars are offering or will be launching copay saving programs, many which advertise copays as low as $0 per month for eligible patients.
How will Humira respond?
Marta Wosińska, PhD, a health care economist at the Brookings Institute, Washington, predicts payers will use these lower biosimilar prices to negotiate better deals with AbbVie, Humira’s manufacturer. “We have a lot of players coming into [the market] right now, so the competition is really fierce,” she said. In response, AbbVie will need to increase rebates on Humira and/or lower its price to compete with these biosimilars.
“The ball is in AbbVie’s court,” she said. “If [the company] is not willing to drop price sufficiently, then payers will start switching to biosimilars.”
Dr. Fendrick reported past financial relationships and consulting arrangements with AbbVie, Amgen, Arnold Ventures, Bayer, CareFirst, BlueCross BlueShield, and many other companies. Dr. Wosińska has received funding from Arnold Ventures and serves as an expert witness on antitrust cases involving generic medication.
A version of this article first appeared on Medscape.com.