User login
Acute Onset of Vitiligolike Depigmentation After Nivolumab Therapy for Systemic Melanoma
To the Editor:
Vitiligolike depigmentation has been known to develop around the sites of origin of melanoma or more rarely in patients treated with antimelanoma therapy.1 Vitiligo is characterized by white patchy depigmentation of the skin caused by the loss of functional melanocytes from the epidermis. The exact mechanisms of disease are unknown and multifactorial; however, autoimmunity plays a central role. Interferon gamma (IFN-γ), C-X-C chemokine ligand 10, and IL-22 have been identified as key mediators in an inflammatory cascade leading to the stimulation of the innate immune response against melanocyte antigens.2,3 Research suggests melanoma-associated vitiligolike leukoderma also results from an immune reaction directed against antigenic determinants shared by both normal and malignant melanocytes.3 Vitiligolike lesions have been associated with the use of immunomodulatory agents such as nivolumab, a fully humanized monoclonal IgG4 antibody, which blocks the programmed cell death protein 1 (PD-1) receptor that normally is expressed on T cells during the effector phase of T-cell activation.4,5 In the tumor microenvironment, the PD-1 receptor is stimulated, leading to downregulation of the T-cell effector function and destruction of T cells.5 Due to T-cell apoptosis and consequent suppression of the immune response, tumorigenesis continues. By inhibiting the PD-1 receptor, nivolumab increases the number of active T cells and antitumor response. However, the distressing side effect of vitiligolike depigmentation has been reported in 15% to 25% of treated patients.6
In a meta-analysis by Teulings et al,7 patients with new-onset vitiligo and malignant melanoma demonstrated a 2-fold decrease in cancer progression and a 4-fold decreased risk for death vs patients without vitiligo development. Thus, in patients with melanoma, vitiligolike depigmentation should be considered a good prognostic indicator as well as a visible sign of spontaneous or therapy-induced antihumoral immune response against melanocyte differentiation antigens, as it is associated with a notable survival benefit in patients receiving immunotherapy for metastatic melanoma.3 We describe a case of diffuse vitiligolike depigmentation that developed suddenly during nivolumab treatment, causing much distress to the patient.
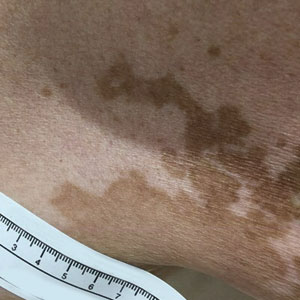
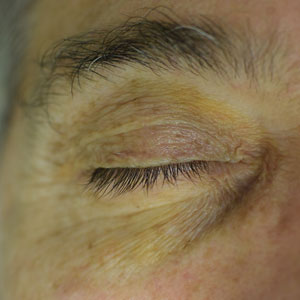
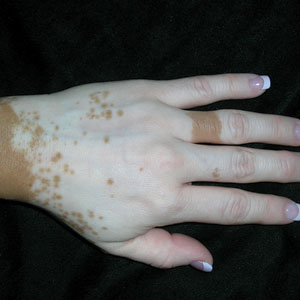
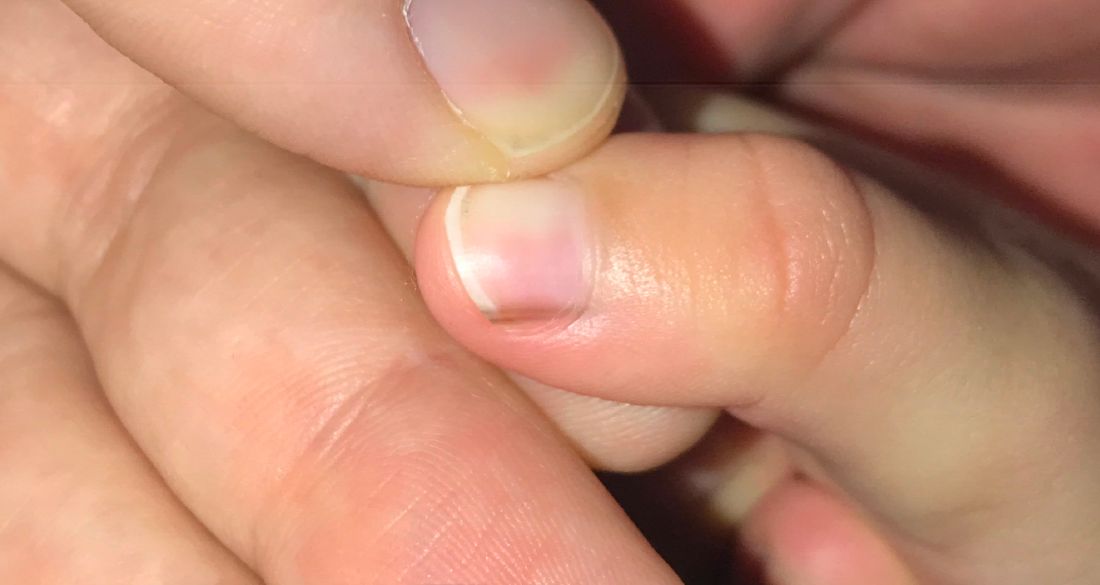
A 75-year-old woman presented to the clinic with a chief concern of sudden diffuse skin discoloration primarily affecting the face, hands, and extremities of 3 weeks’ duration. She had a medical history of metastatic melanoma—the site of the primary melanoma was never identified—and she was undergoing immune-modulating therapy with nivolumab. She was on her fifth month of treatment and was experiencing a robust therapeutic response with a reported 100% clearance of the metastatic melanoma as observed on a positron emission tomography scan. The patchy depigmentation of skin was causing her much distress. Physical examination revealed diffuse patches of hypopigmentation on the trunk, face, and extremities (Figure). Shave biopsies of the right lateral arm demonstrated changes consistent with vitiligo, with an adjacent biopsy illustrating normal skin characteristics. Triamcinolone ointment 0.1% was initiated, with instruction to apply it to affected areas twice daily for 2 weeks. However, there was no improvement, and she discontinued use.
At 3-month follow-up, the depigmentation persisted, prompting a trial of hydroquinone cream 4% to be used sparingly in cosmetically sensitive areas such as the face and dorsal aspects of the hands. Additionally, diligent photoprotection was advised. Upon re-evaluation 9 months later, the patient remained in cancer remission, continued nivolumab therapy, and reported improvement in the hypopigmentation with a more even skin color with topical hydroquinone use. She no longer noticed starkly contrasting hypopigmented patches.
Vitiligo is a benign skin condition characterized by white depigmented macules and patches. The key feature of the disorder is loss of functional melanocytes from the cutaneous epidermis and sometimes from the hair follicles, with various theories on the cause. It has been suggested that the disease is multifactorial, involving both genetics and environmental factors.2 Regardless of the exact mechanism, the result is always the same: loss of melanin pigment in cells due to loss of melanocytes.
Autoimmunity plays a central role in the causation of vitiligo and was first suspected as a possible cause due to the association of vitiligo with several other autoimmune disorders, such as thyroiditis.8 An epidemiological survey from the United Kingdom and North America (N=2624) found that 19.4% of vitiligo patients aged 20 years or older also reported a clinical history of autoimmune thyroid disease compared with 2.4% of the overall White population of the same age.9 Interferon gamma, C-X-C chemokine ligand 10, and IL-22 receptors stimulate the innate immune response, resulting in an overactive danger signaling cascade, which leads to proinflammatory signals against melanocyte antigens.2,3 The adaptive immune system also participates in the progression of vitiligo by activating dermal dendritic cells to attack melanocytes along with melanocyte-specific cytotoxic T cells.
Immunomodulatory agents utilized in the treatment of metastatic melanoma have been linked to vitiligolike depigmentation. In those receiving PD-1 immunotherapy for metastatic melanoma, vitiligolike lesions have been reported in 15% to 25% of patients.6 Typically, the PD-1 molecule has a regulatory function on effector T cells. Interaction of the PD-1 receptor with its ligands occurs primarily in peripheral tissue causing apoptosis and downregulation of effector T cells with the goal of decreasing collateral damage to surrounding tissues by active T cells.5 In the tumor microenvironment, however, suppression of the host’s immune response is enhanced by aberrant stimulation of the PD-1 receptor, causing downregulation of the T-cell effector function, T-cell destruction, and apoptosis, which results in continued tumor growth. Nivolumab, a fully humanized monoclonal IgG4 antibody, selectively inhibits the PD-1 receptor, disrupting the regulator pathway that would typically end in T-cell destruction.5 Accordingly, the population of active T cells is increased along with the antitumor response.4,10 Nivolumab exhibits success as an immunotherapeutic agent, with an overall survival rate in patients with metastatic melanoma undergoing nivolumab therapy of 41% to 42% at 3 years and 35% at 5 years.11 However, therapeutic manipulation of the host’s immune response does not come without a cost. Vitiligolike lesions have been reported in up to a quarter of patients receiving PD-1 immunotherapy for metastatic melanoma.6
The relationship between vitiligolike depigmentation and melanoma can be explained by the immune activation against antigens associated with melanoma that also are expressed by normal melanocytes. In clinical observations of patients with melanoma and patients with vitiligo, antibodies to human melanocyte antigens were present in 80% (24/30) of patients vs 7% (2/28) in the control group.12 The autoimmune response results from a cross-reaction of melanoma cells that share the same antigens as normal melanocytes, such as melanoma antigen recognized by T cells 1 (MART-1), gp100, and tyrosinase.13,14
Development of vitiligolike depigmentation in patients with metastatic melanoma treated with nivolumab has been reported to occur between 2 and 15 months after the start of PD-1 therapy. This side effect of treatment correlates with favorable clinical outcomes.15,16 Enhancing immune recognition of melanocytes in patients with melanoma confers a survival advantage, as studies by Koh et al17 and Norlund et al18 involving patients who developed vitiligolike hypopigmentation associated with malignant melanoma indicated a better prognosis than for those without hypopigmentation. The 5-year survival rate of patients with both malignant melanoma and vitiligo was reported as 60% to 67% when it was estimated that only 30% to 50% of patients should have survived that duration of time.17,18 Similarly, a systematic review of patients with melanoma stages III and IV reported that those with associated hypopigmentation had a 2- to 4-fold decreased risk of disease progression and death compared to patients without depigmentation.7
Use of traditional treatment therapies for vitiligo is based on the ability of the therapy to suppress the immune system. However, in patients with metastatic melanoma undergoing immune-modulating cancer therapies, traditional treatment options may counter the antitumor effects of the targeted immunotherapies and should be used with caution. Our patient displayed improvement in the appearance of her starkly contrasting hypopigmented patches with the use of hydroquinone cream 4%, which induced necrotic death of melanocytes by inhibiting the conversion of L-3,4-dihydroxyphenylalanine to melanin by tyrosinase.19 The effect achieved by using topical hydroquinone 4% was a lighter skin appearance in areas of application.
There is no cure for vitiligo, and although it is a benign condition, it can negatively impact a patient's quality of life. In some countries, vitiligo is confused with leprosy, resulting in a social stigma attached to the diagnosis. Many patients are frightened or embarrassed by the diagnosis of vitiligo and its effects, and they often experience discrimination.2 Patients with vitiligo also experience more psychological difficulties such as depression.20 The unpredictability of vitiligo is associated with negative emotions including fear of spreading the lesions, shame, insecurity, and sadness.21 Supportive care measures, including psychological support and counseling, are recommended. Additionally, upon initiation of anti–PD-1 therapies, expectations should be discussed with patients concerning the possibilities of depigmentation and associated treatment results. Although the occurrence of vitiligo may cause the patient concern, it should be communicated that its presence is a positive indicator of a vigorous antimelanoma immunity and an increased survival rate.7
Vitiligolike depigmentation is a known rare adverse effect of nivolumab treatment. Although aesthetically unfavorable for the patient, the development of vitiligolike lesions while undergoing immunotherapy for melanoma may be a sign of a promising clinical outcome due to an effective immune response to melanoma antigens. Our patient remains in remission without any evidence of melanoma after 9 months of therapy, which offers support for a promising outcome for melanoma patients who experience vitiligolike depigmentation.
- de Golian E, Kwong BY, Swetter SM, et al. Cutaneous complications of targeted melanoma therapy. Curr Treat Options Oncol. 2016;17:57.
- Ezzedine K, Eleftheriadou V, Whitton M, et al. Vitiligo. Lancet. 2015;386:74-84.
- Ortonne, JP, Passeron, T. Vitiligo and other disorders of hypopigmentation. In: Bolognia J, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:1087-1114.
- Opdivo. Package insert. Bristol-Myers Squibb Company; 2023.
- Ott PA, Hodi FS, Robert C. CTLA-4 and PD-1/PD-L1 blockade: new immunotherapeutic modalities with durable clinical benefit in melanoma patients. Clin Cancer Res. 2013;19:5300-5309.
- Hwang SJE, Carlos G, Wakade D, et al. Cutaneous adverse events (AEs) of anti-programmed cell death (PD)-1 therapy in patients with metastatic melanoma: a single-institution cohort. J Am Acad Dermatol. 2016;74:455-461.e1.
- Teulings HE, Limpens J, Jansen SN, et al. Vitiligo-like depigmentation in patients with stage III-IV melanoma receiving immunotherapy and its association with survival: a systematic review and meta-analysis. J Clin Oncol. 2015;33:773-781.
- Gey A, Diallo A, Seneschal J, et al. Autoimmune thyroid disease in vitiligo: multivariate analysis indicates intricate pathomechanisms. Br J Dermatol. 2013;168:756-761.
- Alkhateeb A, Fain PR, Thody A, et al. Epidemiology of vitiligo and associated autoimmune diseases in Caucasian probands and their families. Pigment Cell Res. 2003;16:208-214.
- Robert C, Long GV, Brady B, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372:320-330.
- Hodi FS, Kluger H, Sznol M, et al. Durable, long-term survival in previously treated patients with advanced melanoma who received nivolumab monotherapy in a phase I trial. Cancer Res. 2016;76(14 suppl):CT001.
- Cui J, Bystryn JC. Melanoma and vitiligo are associated with antibody responses to similar antigens on pigment cells. Arch Dermatol. 1995;131:314-318.
- Lynch SA, Bouchard BN, Vijayasaradhi S, et al. Antigens of melanocytes and melanoma. Cancer Metastasis Rev. 1991;10:141-150.
- Sanlorenzo M, Vujic I, Daud A, et al. Pembrolizumab cutaneous adverse events and their association with disease progression. JAMA Dermatol. 2015;15:1206-1212.
- Hua C, Boussemart L, Mateus C, et al. Association of vitiligo with tumor response in patients with metastatic melanoma treated with pembrolizumab. JAMA Dermatol. 2016;152:45-51.
- Nakamura Y, Tanaka R, Asami Y, et al. Correlation between vitiligo occurrence and clinical benefit in advanced melanoma patients treated with nivolumab: a multi-institutional retrospective study. J Dermatol. 2017;44:117-122.
- Koh HK, Sober AJ, Nakagawa H, et al. Malignant melanoma and vitiligo-like leukoderma: an electron microscope study. J Am Acad Dermatol. 1983;9:696-708.
- Nordlund JJ, Kirkwood JM, Forget BM, et al. Vitiligo in patients with metastatic melanoma: a good prognostic sign. J Am Acad Dermatol. 1983;9:689-696.
- Palumbo A, d’Ischia M, Misuraca G, et al. Mechanism of inhibition of melanogenesis by hydroquinone. Biochim Biophys Acta. 1991;1073:85-90.
- Lai YC, Yew YW, Kennedy C, et al. Vitiligo and depression: a systematic review and meta-analysis of observational studies. Br J Dermatol. 2017;177:708-718.
- Nogueira LSC, Zancanaro PCQ, Azambuja RD. Vitiligo and emotions. An Bras Dermatol. 2009;84:41-45.
To the Editor:
Vitiligolike depigmentation has been known to develop around the sites of origin of melanoma or more rarely in patients treated with antimelanoma therapy.1 Vitiligo is characterized by white patchy depigmentation of the skin caused by the loss of functional melanocytes from the epidermis. The exact mechanisms of disease are unknown and multifactorial; however, autoimmunity plays a central role. Interferon gamma (IFN-γ), C-X-C chemokine ligand 10, and IL-22 have been identified as key mediators in an inflammatory cascade leading to the stimulation of the innate immune response against melanocyte antigens.2,3 Research suggests melanoma-associated vitiligolike leukoderma also results from an immune reaction directed against antigenic determinants shared by both normal and malignant melanocytes.3 Vitiligolike lesions have been associated with the use of immunomodulatory agents such as nivolumab, a fully humanized monoclonal IgG4 antibody, which blocks the programmed cell death protein 1 (PD-1) receptor that normally is expressed on T cells during the effector phase of T-cell activation.4,5 In the tumor microenvironment, the PD-1 receptor is stimulated, leading to downregulation of the T-cell effector function and destruction of T cells.5 Due to T-cell apoptosis and consequent suppression of the immune response, tumorigenesis continues. By inhibiting the PD-1 receptor, nivolumab increases the number of active T cells and antitumor response. However, the distressing side effect of vitiligolike depigmentation has been reported in 15% to 25% of treated patients.6
In a meta-analysis by Teulings et al,7 patients with new-onset vitiligo and malignant melanoma demonstrated a 2-fold decrease in cancer progression and a 4-fold decreased risk for death vs patients without vitiligo development. Thus, in patients with melanoma, vitiligolike depigmentation should be considered a good prognostic indicator as well as a visible sign of spontaneous or therapy-induced antihumoral immune response against melanocyte differentiation antigens, as it is associated with a notable survival benefit in patients receiving immunotherapy for metastatic melanoma.3 We describe a case of diffuse vitiligolike depigmentation that developed suddenly during nivolumab treatment, causing much distress to the patient.
A 75-year-old woman presented to the clinic with a chief concern of sudden diffuse skin discoloration primarily affecting the face, hands, and extremities of 3 weeks’ duration. She had a medical history of metastatic melanoma—the site of the primary melanoma was never identified—and she was undergoing immune-modulating therapy with nivolumab. She was on her fifth month of treatment and was experiencing a robust therapeutic response with a reported 100% clearance of the metastatic melanoma as observed on a positron emission tomography scan. The patchy depigmentation of skin was causing her much distress. Physical examination revealed diffuse patches of hypopigmentation on the trunk, face, and extremities (Figure). Shave biopsies of the right lateral arm demonstrated changes consistent with vitiligo, with an adjacent biopsy illustrating normal skin characteristics. Triamcinolone ointment 0.1% was initiated, with instruction to apply it to affected areas twice daily for 2 weeks. However, there was no improvement, and she discontinued use.

At 3-month follow-up, the depigmentation persisted, prompting a trial of hydroquinone cream 4% to be used sparingly in cosmetically sensitive areas such as the face and dorsal aspects of the hands. Additionally, diligent photoprotection was advised. Upon re-evaluation 9 months later, the patient remained in cancer remission, continued nivolumab therapy, and reported improvement in the hypopigmentation with a more even skin color with topical hydroquinone use. She no longer noticed starkly contrasting hypopigmented patches.
Vitiligo is a benign skin condition characterized by white depigmented macules and patches. The key feature of the disorder is loss of functional melanocytes from the cutaneous epidermis and sometimes from the hair follicles, with various theories on the cause. It has been suggested that the disease is multifactorial, involving both genetics and environmental factors.2 Regardless of the exact mechanism, the result is always the same: loss of melanin pigment in cells due to loss of melanocytes.
Autoimmunity plays a central role in the causation of vitiligo and was first suspected as a possible cause due to the association of vitiligo with several other autoimmune disorders, such as thyroiditis.8 An epidemiological survey from the United Kingdom and North America (N=2624) found that 19.4% of vitiligo patients aged 20 years or older also reported a clinical history of autoimmune thyroid disease compared with 2.4% of the overall White population of the same age.9 Interferon gamma, C-X-C chemokine ligand 10, and IL-22 receptors stimulate the innate immune response, resulting in an overactive danger signaling cascade, which leads to proinflammatory signals against melanocyte antigens.2,3 The adaptive immune system also participates in the progression of vitiligo by activating dermal dendritic cells to attack melanocytes along with melanocyte-specific cytotoxic T cells.
Immunomodulatory agents utilized in the treatment of metastatic melanoma have been linked to vitiligolike depigmentation. In those receiving PD-1 immunotherapy for metastatic melanoma, vitiligolike lesions have been reported in 15% to 25% of patients.6 Typically, the PD-1 molecule has a regulatory function on effector T cells. Interaction of the PD-1 receptor with its ligands occurs primarily in peripheral tissue causing apoptosis and downregulation of effector T cells with the goal of decreasing collateral damage to surrounding tissues by active T cells.5 In the tumor microenvironment, however, suppression of the host’s immune response is enhanced by aberrant stimulation of the PD-1 receptor, causing downregulation of the T-cell effector function, T-cell destruction, and apoptosis, which results in continued tumor growth. Nivolumab, a fully humanized monoclonal IgG4 antibody, selectively inhibits the PD-1 receptor, disrupting the regulator pathway that would typically end in T-cell destruction.5 Accordingly, the population of active T cells is increased along with the antitumor response.4,10 Nivolumab exhibits success as an immunotherapeutic agent, with an overall survival rate in patients with metastatic melanoma undergoing nivolumab therapy of 41% to 42% at 3 years and 35% at 5 years.11 However, therapeutic manipulation of the host’s immune response does not come without a cost. Vitiligolike lesions have been reported in up to a quarter of patients receiving PD-1 immunotherapy for metastatic melanoma.6
The relationship between vitiligolike depigmentation and melanoma can be explained by the immune activation against antigens associated with melanoma that also are expressed by normal melanocytes. In clinical observations of patients with melanoma and patients with vitiligo, antibodies to human melanocyte antigens were present in 80% (24/30) of patients vs 7% (2/28) in the control group.12 The autoimmune response results from a cross-reaction of melanoma cells that share the same antigens as normal melanocytes, such as melanoma antigen recognized by T cells 1 (MART-1), gp100, and tyrosinase.13,14
Development of vitiligolike depigmentation in patients with metastatic melanoma treated with nivolumab has been reported to occur between 2 and 15 months after the start of PD-1 therapy. This side effect of treatment correlates with favorable clinical outcomes.15,16 Enhancing immune recognition of melanocytes in patients with melanoma confers a survival advantage, as studies by Koh et al17 and Norlund et al18 involving patients who developed vitiligolike hypopigmentation associated with malignant melanoma indicated a better prognosis than for those without hypopigmentation. The 5-year survival rate of patients with both malignant melanoma and vitiligo was reported as 60% to 67% when it was estimated that only 30% to 50% of patients should have survived that duration of time.17,18 Similarly, a systematic review of patients with melanoma stages III and IV reported that those with associated hypopigmentation had a 2- to 4-fold decreased risk of disease progression and death compared to patients without depigmentation.7
Use of traditional treatment therapies for vitiligo is based on the ability of the therapy to suppress the immune system. However, in patients with metastatic melanoma undergoing immune-modulating cancer therapies, traditional treatment options may counter the antitumor effects of the targeted immunotherapies and should be used with caution. Our patient displayed improvement in the appearance of her starkly contrasting hypopigmented patches with the use of hydroquinone cream 4%, which induced necrotic death of melanocytes by inhibiting the conversion of L-3,4-dihydroxyphenylalanine to melanin by tyrosinase.19 The effect achieved by using topical hydroquinone 4% was a lighter skin appearance in areas of application.
There is no cure for vitiligo, and although it is a benign condition, it can negatively impact a patient's quality of life. In some countries, vitiligo is confused with leprosy, resulting in a social stigma attached to the diagnosis. Many patients are frightened or embarrassed by the diagnosis of vitiligo and its effects, and they often experience discrimination.2 Patients with vitiligo also experience more psychological difficulties such as depression.20 The unpredictability of vitiligo is associated with negative emotions including fear of spreading the lesions, shame, insecurity, and sadness.21 Supportive care measures, including psychological support and counseling, are recommended. Additionally, upon initiation of anti–PD-1 therapies, expectations should be discussed with patients concerning the possibilities of depigmentation and associated treatment results. Although the occurrence of vitiligo may cause the patient concern, it should be communicated that its presence is a positive indicator of a vigorous antimelanoma immunity and an increased survival rate.7
Vitiligolike depigmentation is a known rare adverse effect of nivolumab treatment. Although aesthetically unfavorable for the patient, the development of vitiligolike lesions while undergoing immunotherapy for melanoma may be a sign of a promising clinical outcome due to an effective immune response to melanoma antigens. Our patient remains in remission without any evidence of melanoma after 9 months of therapy, which offers support for a promising outcome for melanoma patients who experience vitiligolike depigmentation.
To the Editor:
Vitiligolike depigmentation has been known to develop around the sites of origin of melanoma or more rarely in patients treated with antimelanoma therapy.1 Vitiligo is characterized by white patchy depigmentation of the skin caused by the loss of functional melanocytes from the epidermis. The exact mechanisms of disease are unknown and multifactorial; however, autoimmunity plays a central role. Interferon gamma (IFN-γ), C-X-C chemokine ligand 10, and IL-22 have been identified as key mediators in an inflammatory cascade leading to the stimulation of the innate immune response against melanocyte antigens.2,3 Research suggests melanoma-associated vitiligolike leukoderma also results from an immune reaction directed against antigenic determinants shared by both normal and malignant melanocytes.3 Vitiligolike lesions have been associated with the use of immunomodulatory agents such as nivolumab, a fully humanized monoclonal IgG4 antibody, which blocks the programmed cell death protein 1 (PD-1) receptor that normally is expressed on T cells during the effector phase of T-cell activation.4,5 In the tumor microenvironment, the PD-1 receptor is stimulated, leading to downregulation of the T-cell effector function and destruction of T cells.5 Due to T-cell apoptosis and consequent suppression of the immune response, tumorigenesis continues. By inhibiting the PD-1 receptor, nivolumab increases the number of active T cells and antitumor response. However, the distressing side effect of vitiligolike depigmentation has been reported in 15% to 25% of treated patients.6
In a meta-analysis by Teulings et al,7 patients with new-onset vitiligo and malignant melanoma demonstrated a 2-fold decrease in cancer progression and a 4-fold decreased risk for death vs patients without vitiligo development. Thus, in patients with melanoma, vitiligolike depigmentation should be considered a good prognostic indicator as well as a visible sign of spontaneous or therapy-induced antihumoral immune response against melanocyte differentiation antigens, as it is associated with a notable survival benefit in patients receiving immunotherapy for metastatic melanoma.3 We describe a case of diffuse vitiligolike depigmentation that developed suddenly during nivolumab treatment, causing much distress to the patient.
A 75-year-old woman presented to the clinic with a chief concern of sudden diffuse skin discoloration primarily affecting the face, hands, and extremities of 3 weeks’ duration. She had a medical history of metastatic melanoma—the site of the primary melanoma was never identified—and she was undergoing immune-modulating therapy with nivolumab. She was on her fifth month of treatment and was experiencing a robust therapeutic response with a reported 100% clearance of the metastatic melanoma as observed on a positron emission tomography scan. The patchy depigmentation of skin was causing her much distress. Physical examination revealed diffuse patches of hypopigmentation on the trunk, face, and extremities (Figure). Shave biopsies of the right lateral arm demonstrated changes consistent with vitiligo, with an adjacent biopsy illustrating normal skin characteristics. Triamcinolone ointment 0.1% was initiated, with instruction to apply it to affected areas twice daily for 2 weeks. However, there was no improvement, and she discontinued use.

At 3-month follow-up, the depigmentation persisted, prompting a trial of hydroquinone cream 4% to be used sparingly in cosmetically sensitive areas such as the face and dorsal aspects of the hands. Additionally, diligent photoprotection was advised. Upon re-evaluation 9 months later, the patient remained in cancer remission, continued nivolumab therapy, and reported improvement in the hypopigmentation with a more even skin color with topical hydroquinone use. She no longer noticed starkly contrasting hypopigmented patches.
Vitiligo is a benign skin condition characterized by white depigmented macules and patches. The key feature of the disorder is loss of functional melanocytes from the cutaneous epidermis and sometimes from the hair follicles, with various theories on the cause. It has been suggested that the disease is multifactorial, involving both genetics and environmental factors.2 Regardless of the exact mechanism, the result is always the same: loss of melanin pigment in cells due to loss of melanocytes.
Autoimmunity plays a central role in the causation of vitiligo and was first suspected as a possible cause due to the association of vitiligo with several other autoimmune disorders, such as thyroiditis.8 An epidemiological survey from the United Kingdom and North America (N=2624) found that 19.4% of vitiligo patients aged 20 years or older also reported a clinical history of autoimmune thyroid disease compared with 2.4% of the overall White population of the same age.9 Interferon gamma, C-X-C chemokine ligand 10, and IL-22 receptors stimulate the innate immune response, resulting in an overactive danger signaling cascade, which leads to proinflammatory signals against melanocyte antigens.2,3 The adaptive immune system also participates in the progression of vitiligo by activating dermal dendritic cells to attack melanocytes along with melanocyte-specific cytotoxic T cells.
Immunomodulatory agents utilized in the treatment of metastatic melanoma have been linked to vitiligolike depigmentation. In those receiving PD-1 immunotherapy for metastatic melanoma, vitiligolike lesions have been reported in 15% to 25% of patients.6 Typically, the PD-1 molecule has a regulatory function on effector T cells. Interaction of the PD-1 receptor with its ligands occurs primarily in peripheral tissue causing apoptosis and downregulation of effector T cells with the goal of decreasing collateral damage to surrounding tissues by active T cells.5 In the tumor microenvironment, however, suppression of the host’s immune response is enhanced by aberrant stimulation of the PD-1 receptor, causing downregulation of the T-cell effector function, T-cell destruction, and apoptosis, which results in continued tumor growth. Nivolumab, a fully humanized monoclonal IgG4 antibody, selectively inhibits the PD-1 receptor, disrupting the regulator pathway that would typically end in T-cell destruction.5 Accordingly, the population of active T cells is increased along with the antitumor response.4,10 Nivolumab exhibits success as an immunotherapeutic agent, with an overall survival rate in patients with metastatic melanoma undergoing nivolumab therapy of 41% to 42% at 3 years and 35% at 5 years.11 However, therapeutic manipulation of the host’s immune response does not come without a cost. Vitiligolike lesions have been reported in up to a quarter of patients receiving PD-1 immunotherapy for metastatic melanoma.6
The relationship between vitiligolike depigmentation and melanoma can be explained by the immune activation against antigens associated with melanoma that also are expressed by normal melanocytes. In clinical observations of patients with melanoma and patients with vitiligo, antibodies to human melanocyte antigens were present in 80% (24/30) of patients vs 7% (2/28) in the control group.12 The autoimmune response results from a cross-reaction of melanoma cells that share the same antigens as normal melanocytes, such as melanoma antigen recognized by T cells 1 (MART-1), gp100, and tyrosinase.13,14
Development of vitiligolike depigmentation in patients with metastatic melanoma treated with nivolumab has been reported to occur between 2 and 15 months after the start of PD-1 therapy. This side effect of treatment correlates with favorable clinical outcomes.15,16 Enhancing immune recognition of melanocytes in patients with melanoma confers a survival advantage, as studies by Koh et al17 and Norlund et al18 involving patients who developed vitiligolike hypopigmentation associated with malignant melanoma indicated a better prognosis than for those without hypopigmentation. The 5-year survival rate of patients with both malignant melanoma and vitiligo was reported as 60% to 67% when it was estimated that only 30% to 50% of patients should have survived that duration of time.17,18 Similarly, a systematic review of patients with melanoma stages III and IV reported that those with associated hypopigmentation had a 2- to 4-fold decreased risk of disease progression and death compared to patients without depigmentation.7
Use of traditional treatment therapies for vitiligo is based on the ability of the therapy to suppress the immune system. However, in patients with metastatic melanoma undergoing immune-modulating cancer therapies, traditional treatment options may counter the antitumor effects of the targeted immunotherapies and should be used with caution. Our patient displayed improvement in the appearance of her starkly contrasting hypopigmented patches with the use of hydroquinone cream 4%, which induced necrotic death of melanocytes by inhibiting the conversion of L-3,4-dihydroxyphenylalanine to melanin by tyrosinase.19 The effect achieved by using topical hydroquinone 4% was a lighter skin appearance in areas of application.
There is no cure for vitiligo, and although it is a benign condition, it can negatively impact a patient's quality of life. In some countries, vitiligo is confused with leprosy, resulting in a social stigma attached to the diagnosis. Many patients are frightened or embarrassed by the diagnosis of vitiligo and its effects, and they often experience discrimination.2 Patients with vitiligo also experience more psychological difficulties such as depression.20 The unpredictability of vitiligo is associated with negative emotions including fear of spreading the lesions, shame, insecurity, and sadness.21 Supportive care measures, including psychological support and counseling, are recommended. Additionally, upon initiation of anti–PD-1 therapies, expectations should be discussed with patients concerning the possibilities of depigmentation and associated treatment results. Although the occurrence of vitiligo may cause the patient concern, it should be communicated that its presence is a positive indicator of a vigorous antimelanoma immunity and an increased survival rate.7
Vitiligolike depigmentation is a known rare adverse effect of nivolumab treatment. Although aesthetically unfavorable for the patient, the development of vitiligolike lesions while undergoing immunotherapy for melanoma may be a sign of a promising clinical outcome due to an effective immune response to melanoma antigens. Our patient remains in remission without any evidence of melanoma after 9 months of therapy, which offers support for a promising outcome for melanoma patients who experience vitiligolike depigmentation.
- de Golian E, Kwong BY, Swetter SM, et al. Cutaneous complications of targeted melanoma therapy. Curr Treat Options Oncol. 2016;17:57.
- Ezzedine K, Eleftheriadou V, Whitton M, et al. Vitiligo. Lancet. 2015;386:74-84.
- Ortonne, JP, Passeron, T. Vitiligo and other disorders of hypopigmentation. In: Bolognia J, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:1087-1114.
- Opdivo. Package insert. Bristol-Myers Squibb Company; 2023.
- Ott PA, Hodi FS, Robert C. CTLA-4 and PD-1/PD-L1 blockade: new immunotherapeutic modalities with durable clinical benefit in melanoma patients. Clin Cancer Res. 2013;19:5300-5309.
- Hwang SJE, Carlos G, Wakade D, et al. Cutaneous adverse events (AEs) of anti-programmed cell death (PD)-1 therapy in patients with metastatic melanoma: a single-institution cohort. J Am Acad Dermatol. 2016;74:455-461.e1.
- Teulings HE, Limpens J, Jansen SN, et al. Vitiligo-like depigmentation in patients with stage III-IV melanoma receiving immunotherapy and its association with survival: a systematic review and meta-analysis. J Clin Oncol. 2015;33:773-781.
- Gey A, Diallo A, Seneschal J, et al. Autoimmune thyroid disease in vitiligo: multivariate analysis indicates intricate pathomechanisms. Br J Dermatol. 2013;168:756-761.
- Alkhateeb A, Fain PR, Thody A, et al. Epidemiology of vitiligo and associated autoimmune diseases in Caucasian probands and their families. Pigment Cell Res. 2003;16:208-214.
- Robert C, Long GV, Brady B, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372:320-330.
- Hodi FS, Kluger H, Sznol M, et al. Durable, long-term survival in previously treated patients with advanced melanoma who received nivolumab monotherapy in a phase I trial. Cancer Res. 2016;76(14 suppl):CT001.
- Cui J, Bystryn JC. Melanoma and vitiligo are associated with antibody responses to similar antigens on pigment cells. Arch Dermatol. 1995;131:314-318.
- Lynch SA, Bouchard BN, Vijayasaradhi S, et al. Antigens of melanocytes and melanoma. Cancer Metastasis Rev. 1991;10:141-150.
- Sanlorenzo M, Vujic I, Daud A, et al. Pembrolizumab cutaneous adverse events and their association with disease progression. JAMA Dermatol. 2015;15:1206-1212.
- Hua C, Boussemart L, Mateus C, et al. Association of vitiligo with tumor response in patients with metastatic melanoma treated with pembrolizumab. JAMA Dermatol. 2016;152:45-51.
- Nakamura Y, Tanaka R, Asami Y, et al. Correlation between vitiligo occurrence and clinical benefit in advanced melanoma patients treated with nivolumab: a multi-institutional retrospective study. J Dermatol. 2017;44:117-122.
- Koh HK, Sober AJ, Nakagawa H, et al. Malignant melanoma and vitiligo-like leukoderma: an electron microscope study. J Am Acad Dermatol. 1983;9:696-708.
- Nordlund JJ, Kirkwood JM, Forget BM, et al. Vitiligo in patients with metastatic melanoma: a good prognostic sign. J Am Acad Dermatol. 1983;9:689-696.
- Palumbo A, d’Ischia M, Misuraca G, et al. Mechanism of inhibition of melanogenesis by hydroquinone. Biochim Biophys Acta. 1991;1073:85-90.
- Lai YC, Yew YW, Kennedy C, et al. Vitiligo and depression: a systematic review and meta-analysis of observational studies. Br J Dermatol. 2017;177:708-718.
- Nogueira LSC, Zancanaro PCQ, Azambuja RD. Vitiligo and emotions. An Bras Dermatol. 2009;84:41-45.
- de Golian E, Kwong BY, Swetter SM, et al. Cutaneous complications of targeted melanoma therapy. Curr Treat Options Oncol. 2016;17:57.
- Ezzedine K, Eleftheriadou V, Whitton M, et al. Vitiligo. Lancet. 2015;386:74-84.
- Ortonne, JP, Passeron, T. Vitiligo and other disorders of hypopigmentation. In: Bolognia J, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2018:1087-1114.
- Opdivo. Package insert. Bristol-Myers Squibb Company; 2023.
- Ott PA, Hodi FS, Robert C. CTLA-4 and PD-1/PD-L1 blockade: new immunotherapeutic modalities with durable clinical benefit in melanoma patients. Clin Cancer Res. 2013;19:5300-5309.
- Hwang SJE, Carlos G, Wakade D, et al. Cutaneous adverse events (AEs) of anti-programmed cell death (PD)-1 therapy in patients with metastatic melanoma: a single-institution cohort. J Am Acad Dermatol. 2016;74:455-461.e1.
- Teulings HE, Limpens J, Jansen SN, et al. Vitiligo-like depigmentation in patients with stage III-IV melanoma receiving immunotherapy and its association with survival: a systematic review and meta-analysis. J Clin Oncol. 2015;33:773-781.
- Gey A, Diallo A, Seneschal J, et al. Autoimmune thyroid disease in vitiligo: multivariate analysis indicates intricate pathomechanisms. Br J Dermatol. 2013;168:756-761.
- Alkhateeb A, Fain PR, Thody A, et al. Epidemiology of vitiligo and associated autoimmune diseases in Caucasian probands and their families. Pigment Cell Res. 2003;16:208-214.
- Robert C, Long GV, Brady B, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372:320-330.
- Hodi FS, Kluger H, Sznol M, et al. Durable, long-term survival in previously treated patients with advanced melanoma who received nivolumab monotherapy in a phase I trial. Cancer Res. 2016;76(14 suppl):CT001.
- Cui J, Bystryn JC. Melanoma and vitiligo are associated with antibody responses to similar antigens on pigment cells. Arch Dermatol. 1995;131:314-318.
- Lynch SA, Bouchard BN, Vijayasaradhi S, et al. Antigens of melanocytes and melanoma. Cancer Metastasis Rev. 1991;10:141-150.
- Sanlorenzo M, Vujic I, Daud A, et al. Pembrolizumab cutaneous adverse events and their association with disease progression. JAMA Dermatol. 2015;15:1206-1212.
- Hua C, Boussemart L, Mateus C, et al. Association of vitiligo with tumor response in patients with metastatic melanoma treated with pembrolizumab. JAMA Dermatol. 2016;152:45-51.
- Nakamura Y, Tanaka R, Asami Y, et al. Correlation between vitiligo occurrence and clinical benefit in advanced melanoma patients treated with nivolumab: a multi-institutional retrospective study. J Dermatol. 2017;44:117-122.
- Koh HK, Sober AJ, Nakagawa H, et al. Malignant melanoma and vitiligo-like leukoderma: an electron microscope study. J Am Acad Dermatol. 1983;9:696-708.
- Nordlund JJ, Kirkwood JM, Forget BM, et al. Vitiligo in patients with metastatic melanoma: a good prognostic sign. J Am Acad Dermatol. 1983;9:689-696.
- Palumbo A, d’Ischia M, Misuraca G, et al. Mechanism of inhibition of melanogenesis by hydroquinone. Biochim Biophys Acta. 1991;1073:85-90.
- Lai YC, Yew YW, Kennedy C, et al. Vitiligo and depression: a systematic review and meta-analysis of observational studies. Br J Dermatol. 2017;177:708-718.
- Nogueira LSC, Zancanaro PCQ, Azambuja RD. Vitiligo and emotions. An Bras Dermatol. 2009;84:41-45.
Practice Points
- New-onset vitiligo coinciding with malignant melanoma should be considered a good prognostic indicator.
- Daily use of hydroquinone cream 4% in conjunction with diligent photoprotection was shown to even overall skin tone in a patient experiencing leukoderma from nivolumab therapy.
Bergamot
Citrus bergamia (bergamot) is a fruit tree thought to originate in the Mediterranean area; its fruit has been a part of the diet in that region since the early 18th century.1 Bergamot is known to confer antioxidant as well as anti-inflammatory activity, and yields proapoptotic effects in the sebaceous gland.2,3 The plant contains the natural furocoumarin bergapten, which is also known as 5-methoxypsoralen.4
5 In this capacity, bergamot oil has been used for photodynamic therapy of cutaneous conditions such as vitiligo.6 In fact, for several years 5-methoxypsoralen and 8-methoxypsoralen have been used to achieve acceptable clearance rates of psoriasis and vitiligo.7 This column focuses on bergapten, as well as the cutaneous conditions for which bergamot has been shown to have some benefits warranting application or further investigation.
Bergapten
In a 2021 literature review, Liang et al. cited the anti-inflammatory, antimicrobial, anticancer, and other salutary effects associated with bergapten. Based on numerous citations, they also cautioned about the phototoxicity of the compound combined with ultraviolet (UV) light while noting the photoactivation of bergapten for anticancer uses.4
The following year, Quetglas-Llabrés et al. acknowledged, in another literature review, the numerous preclinical and in vitro studies demonstrating the therapeutic activity of bergapten and highlighted clinical trials revealing notable lesion clearance rates of psoriasis or vitiligo imparted by oral or topical bergapten along with UV irradiation. Bergapten was also found to be effective as hypolipemic therapy.5
Anti-inflammatory topical uses
In a 2017 study by Han et al. of 10 essential oils, bergamot was among the investigated oils, all of which exhibited significant anti-proliferative activity in a preinflamed human dermal fibroblast system simulating chronic inflammation. Bergamot was among three essential oils that also suppressed protein molecules involved with inflammation, immune responses, and tissue remodeling, indicating anti-inflammatory and wound healing characteristics.8
More recently, Cristiano et al. reported that ultradeformable nanocarriers containing bergamot essential oil and ammonium glycyrrhizinate were demonstrated in healthy human volunteers to be characterized by the appropriate mean size, size distribution, surface charge, and long-term stability for topical administration. Topical administration on human volunteers also revealed greater activity of the combined agents as compared with a nanosystem loaded only with ammonium glycyrrhizinate. The researchers concluded that this combination of ingredients in ultradeformable vesicles shows potential as topical anti-inflammatory treatment.3
Acne
In a 2020 study using golden hamsters, Sun et al. assessed the effects of the juice and essential oils of bergamot and sweet orange on acne vulgaris engendered by excessive androgen secretion. Among 80 male hamsters randomly divided into 10 groups ranging from low to high doses, all results demonstrated improvement with treatment as seen by decreased growth rates of sebaceous glands, suppressed triglyceride accumulation, lowered inflammatory cytokine release, and apoptosis promotion in sebaceous glands. The authors noted that the essential oils yielded better dose-dependent effects than the juices.2
Psoriasis
In 2019, Perna et al. conducted a literature review on the effects of bergamot essential oil, extract, juice, and polyphenolic fraction on various health metrics. Thirty-one studies (20 involving humans with 1,709 subjects and 11 in rats and mice) were identified. Animal models indicated that bergamot essential oil (10 mg/kg or 20 mg/kg daily for 20 weeks) reduced psoriatic plaques, increased skin collagen content, and fostered hair growth and that bergamot juice (20 mg/kg) diminished proinflammatory cytokines. Human studies showed that bergamot extract and essential oil may reduce blood pressure and improve mental conditions.9
Vitiligo
In 2019, Shaaban et al. prepared elastic nanocarriers (spanlastics) to deliver psoralen-containing bergamot oil along with PUVB with the intention of harnessing melanogenic activity to treat vitiligo. Histopathologic assessment on rat skin was conducted before clinical treatment in patients with vitiligo. The spanlastics were deemed to be of suitable nanosize and deformable, yielding consistent bergamot oil release. The bergamot oil included in the nanocarrier was found to enhance photostability and photodynamic activity, with the researchers concluding that bergamot oil nanospanlastics with psoralen-UVB therapy shows potential as a vitiligo therapy.10
Two years later, Shaaban evaluated bergamot oil formulated in nanostructured lipid carriers as a photosensitizer for photodynamic treatment of vitiligo. The botanical oil was effectively used in the nanostructured lipid carriers with a gel consistency that delivered sustained release of the oil for 24 hours. Preclinical and clinical results in patients were encouraging for the topical photodynamic treatment of vitiligo, with the nanostructured lipid carriers improving the photostability and photodynamic activity of bergamot oil.6
Photoaging, photoprotection, and safety concerns
Three decades ago, an international cooperative study of the photophysical, photomutagenic, and photocarcinogenic characteristics of bergamot oil and the effect of UVA and UVB sunscreens found that UVB and UVA sunscreens at low concentration (0.5%-1%) in perfumes could not inhibit the phototoxicity of bergamot oil on human skin.11
In a 2015 study assessing the impact of 38% bergamot polyphenolic fraction (a highly concentrated Citrus bergamia fruit extract) on UVB-generated photoaging, Nisticò et al. found that the bergamot compound dose-dependently protected HaCaT cells against UVB-caused oxidative stress and photoaging markers. Suggesting that the high-antioxidant bergamot polyphenolic fraction has potential for use in skin care formulations, the researchers added that the extract seems to induce antiproliferative, immune-modulating, and antiaging activity.12In 2022, Alexa et al. performed in vitro tests and found that natural preparations containing bergamot, orange, and clove essential oils do not significantly alter physiological skin parameters and were deemed safe for topical use. An emulsion with bergamot essential oil was also found to reduce the viability of oral squamous cell carcinoma cells.13
Conclusion
As a photosensitizing agent, bergamot has an established role in skin care. Beyond its niche role in treatments for vitiligo and psoriasis, this botanical product appears to show potential as an anti-inflammatory agent as well as an ingredient to combat photoaging and skin cancer. Much more research is needed to elucidate the possible wider benefits of this Mediterranean staple.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions Inc., a SaaS company used to generate skin care routines in office and as an ecommerce solution. Write to her at dermnews@mdedge.com.
References
1. Juber M. Health benefits of bergamot. WebMD. November 29, 2022. Accessed March 21, 2023.
2. Sun P et al. Mediators Inflamm. 2020 Oct 6;2020:8868107.
3. Cristiano MC et al. Biomedicines. 2022 Apr 30;10(5):1039.
4. Liang Y et al. Phytother Res. 2021 Nov;35(11):6131-47.
5. Quetglas-Llabrés MM et al. Oxid Med Cell Longev. 2022 Apr 25;2022:8615242.
6. Shaaban M et al. Expert Opin Drug Deliv. 2021 Jan;18(1):139-50.
7. McNeely W, Goa KL. Drugs. 1998 Oct;56(4):667-90.
8. Han X, Beaumont C, Stevens N. Biochim Open. 2017 Apr 26;5:1-7.
9. Perna S et al. Food Sci Nutr. 2019 Jan 25;7(2):369-84.
10. Shaaban M et al. Drug Deliv Transl Res. 2019 Dec;9(6):1106-16.
11. Dubertret L et al. J Photochem Photobiol B. 1990 Nov;7(2-4):251-9.
12. Nisticò S et al. J Biol Regul Homeost Agents. 2015 Jul-Sep;29(3):723-8.
13. Alexa VT et al. Molecules. 2022 Feb 1;27(3):990.
Citrus bergamia (bergamot) is a fruit tree thought to originate in the Mediterranean area; its fruit has been a part of the diet in that region since the early 18th century.1 Bergamot is known to confer antioxidant as well as anti-inflammatory activity, and yields proapoptotic effects in the sebaceous gland.2,3 The plant contains the natural furocoumarin bergapten, which is also known as 5-methoxypsoralen.4
5 In this capacity, bergamot oil has been used for photodynamic therapy of cutaneous conditions such as vitiligo.6 In fact, for several years 5-methoxypsoralen and 8-methoxypsoralen have been used to achieve acceptable clearance rates of psoriasis and vitiligo.7 This column focuses on bergapten, as well as the cutaneous conditions for which bergamot has been shown to have some benefits warranting application or further investigation.
Bergapten
In a 2021 literature review, Liang et al. cited the anti-inflammatory, antimicrobial, anticancer, and other salutary effects associated with bergapten. Based on numerous citations, they also cautioned about the phototoxicity of the compound combined with ultraviolet (UV) light while noting the photoactivation of bergapten for anticancer uses.4
The following year, Quetglas-Llabrés et al. acknowledged, in another literature review, the numerous preclinical and in vitro studies demonstrating the therapeutic activity of bergapten and highlighted clinical trials revealing notable lesion clearance rates of psoriasis or vitiligo imparted by oral or topical bergapten along with UV irradiation. Bergapten was also found to be effective as hypolipemic therapy.5
Anti-inflammatory topical uses
In a 2017 study by Han et al. of 10 essential oils, bergamot was among the investigated oils, all of which exhibited significant anti-proliferative activity in a preinflamed human dermal fibroblast system simulating chronic inflammation. Bergamot was among three essential oils that also suppressed protein molecules involved with inflammation, immune responses, and tissue remodeling, indicating anti-inflammatory and wound healing characteristics.8
More recently, Cristiano et al. reported that ultradeformable nanocarriers containing bergamot essential oil and ammonium glycyrrhizinate were demonstrated in healthy human volunteers to be characterized by the appropriate mean size, size distribution, surface charge, and long-term stability for topical administration. Topical administration on human volunteers also revealed greater activity of the combined agents as compared with a nanosystem loaded only with ammonium glycyrrhizinate. The researchers concluded that this combination of ingredients in ultradeformable vesicles shows potential as topical anti-inflammatory treatment.3
Acne
In a 2020 study using golden hamsters, Sun et al. assessed the effects of the juice and essential oils of bergamot and sweet orange on acne vulgaris engendered by excessive androgen secretion. Among 80 male hamsters randomly divided into 10 groups ranging from low to high doses, all results demonstrated improvement with treatment as seen by decreased growth rates of sebaceous glands, suppressed triglyceride accumulation, lowered inflammatory cytokine release, and apoptosis promotion in sebaceous glands. The authors noted that the essential oils yielded better dose-dependent effects than the juices.2
Psoriasis
In 2019, Perna et al. conducted a literature review on the effects of bergamot essential oil, extract, juice, and polyphenolic fraction on various health metrics. Thirty-one studies (20 involving humans with 1,709 subjects and 11 in rats and mice) were identified. Animal models indicated that bergamot essential oil (10 mg/kg or 20 mg/kg daily for 20 weeks) reduced psoriatic plaques, increased skin collagen content, and fostered hair growth and that bergamot juice (20 mg/kg) diminished proinflammatory cytokines. Human studies showed that bergamot extract and essential oil may reduce blood pressure and improve mental conditions.9
Vitiligo
In 2019, Shaaban et al. prepared elastic nanocarriers (spanlastics) to deliver psoralen-containing bergamot oil along with PUVB with the intention of harnessing melanogenic activity to treat vitiligo. Histopathologic assessment on rat skin was conducted before clinical treatment in patients with vitiligo. The spanlastics were deemed to be of suitable nanosize and deformable, yielding consistent bergamot oil release. The bergamot oil included in the nanocarrier was found to enhance photostability and photodynamic activity, with the researchers concluding that bergamot oil nanospanlastics with psoralen-UVB therapy shows potential as a vitiligo therapy.10
Two years later, Shaaban evaluated bergamot oil formulated in nanostructured lipid carriers as a photosensitizer for photodynamic treatment of vitiligo. The botanical oil was effectively used in the nanostructured lipid carriers with a gel consistency that delivered sustained release of the oil for 24 hours. Preclinical and clinical results in patients were encouraging for the topical photodynamic treatment of vitiligo, with the nanostructured lipid carriers improving the photostability and photodynamic activity of bergamot oil.6
Photoaging, photoprotection, and safety concerns
Three decades ago, an international cooperative study of the photophysical, photomutagenic, and photocarcinogenic characteristics of bergamot oil and the effect of UVA and UVB sunscreens found that UVB and UVA sunscreens at low concentration (0.5%-1%) in perfumes could not inhibit the phototoxicity of bergamot oil on human skin.11
In a 2015 study assessing the impact of 38% bergamot polyphenolic fraction (a highly concentrated Citrus bergamia fruit extract) on UVB-generated photoaging, Nisticò et al. found that the bergamot compound dose-dependently protected HaCaT cells against UVB-caused oxidative stress and photoaging markers. Suggesting that the high-antioxidant bergamot polyphenolic fraction has potential for use in skin care formulations, the researchers added that the extract seems to induce antiproliferative, immune-modulating, and antiaging activity.12In 2022, Alexa et al. performed in vitro tests and found that natural preparations containing bergamot, orange, and clove essential oils do not significantly alter physiological skin parameters and were deemed safe for topical use. An emulsion with bergamot essential oil was also found to reduce the viability of oral squamous cell carcinoma cells.13
Conclusion
As a photosensitizing agent, bergamot has an established role in skin care. Beyond its niche role in treatments for vitiligo and psoriasis, this botanical product appears to show potential as an anti-inflammatory agent as well as an ingredient to combat photoaging and skin cancer. Much more research is needed to elucidate the possible wider benefits of this Mediterranean staple.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions Inc., a SaaS company used to generate skin care routines in office and as an ecommerce solution. Write to her at dermnews@mdedge.com.
References
1. Juber M. Health benefits of bergamot. WebMD. November 29, 2022. Accessed March 21, 2023.
2. Sun P et al. Mediators Inflamm. 2020 Oct 6;2020:8868107.
3. Cristiano MC et al. Biomedicines. 2022 Apr 30;10(5):1039.
4. Liang Y et al. Phytother Res. 2021 Nov;35(11):6131-47.
5. Quetglas-Llabrés MM et al. Oxid Med Cell Longev. 2022 Apr 25;2022:8615242.
6. Shaaban M et al. Expert Opin Drug Deliv. 2021 Jan;18(1):139-50.
7. McNeely W, Goa KL. Drugs. 1998 Oct;56(4):667-90.
8. Han X, Beaumont C, Stevens N. Biochim Open. 2017 Apr 26;5:1-7.
9. Perna S et al. Food Sci Nutr. 2019 Jan 25;7(2):369-84.
10. Shaaban M et al. Drug Deliv Transl Res. 2019 Dec;9(6):1106-16.
11. Dubertret L et al. J Photochem Photobiol B. 1990 Nov;7(2-4):251-9.
12. Nisticò S et al. J Biol Regul Homeost Agents. 2015 Jul-Sep;29(3):723-8.
13. Alexa VT et al. Molecules. 2022 Feb 1;27(3):990.
Citrus bergamia (bergamot) is a fruit tree thought to originate in the Mediterranean area; its fruit has been a part of the diet in that region since the early 18th century.1 Bergamot is known to confer antioxidant as well as anti-inflammatory activity, and yields proapoptotic effects in the sebaceous gland.2,3 The plant contains the natural furocoumarin bergapten, which is also known as 5-methoxypsoralen.4
5 In this capacity, bergamot oil has been used for photodynamic therapy of cutaneous conditions such as vitiligo.6 In fact, for several years 5-methoxypsoralen and 8-methoxypsoralen have been used to achieve acceptable clearance rates of psoriasis and vitiligo.7 This column focuses on bergapten, as well as the cutaneous conditions for which bergamot has been shown to have some benefits warranting application or further investigation.
Bergapten
In a 2021 literature review, Liang et al. cited the anti-inflammatory, antimicrobial, anticancer, and other salutary effects associated with bergapten. Based on numerous citations, they also cautioned about the phototoxicity of the compound combined with ultraviolet (UV) light while noting the photoactivation of bergapten for anticancer uses.4
The following year, Quetglas-Llabrés et al. acknowledged, in another literature review, the numerous preclinical and in vitro studies demonstrating the therapeutic activity of bergapten and highlighted clinical trials revealing notable lesion clearance rates of psoriasis or vitiligo imparted by oral or topical bergapten along with UV irradiation. Bergapten was also found to be effective as hypolipemic therapy.5
Anti-inflammatory topical uses
In a 2017 study by Han et al. of 10 essential oils, bergamot was among the investigated oils, all of which exhibited significant anti-proliferative activity in a preinflamed human dermal fibroblast system simulating chronic inflammation. Bergamot was among three essential oils that also suppressed protein molecules involved with inflammation, immune responses, and tissue remodeling, indicating anti-inflammatory and wound healing characteristics.8
More recently, Cristiano et al. reported that ultradeformable nanocarriers containing bergamot essential oil and ammonium glycyrrhizinate were demonstrated in healthy human volunteers to be characterized by the appropriate mean size, size distribution, surface charge, and long-term stability for topical administration. Topical administration on human volunteers also revealed greater activity of the combined agents as compared with a nanosystem loaded only with ammonium glycyrrhizinate. The researchers concluded that this combination of ingredients in ultradeformable vesicles shows potential as topical anti-inflammatory treatment.3
Acne
In a 2020 study using golden hamsters, Sun et al. assessed the effects of the juice and essential oils of bergamot and sweet orange on acne vulgaris engendered by excessive androgen secretion. Among 80 male hamsters randomly divided into 10 groups ranging from low to high doses, all results demonstrated improvement with treatment as seen by decreased growth rates of sebaceous glands, suppressed triglyceride accumulation, lowered inflammatory cytokine release, and apoptosis promotion in sebaceous glands. The authors noted that the essential oils yielded better dose-dependent effects than the juices.2
Psoriasis
In 2019, Perna et al. conducted a literature review on the effects of bergamot essential oil, extract, juice, and polyphenolic fraction on various health metrics. Thirty-one studies (20 involving humans with 1,709 subjects and 11 in rats and mice) were identified. Animal models indicated that bergamot essential oil (10 mg/kg or 20 mg/kg daily for 20 weeks) reduced psoriatic plaques, increased skin collagen content, and fostered hair growth and that bergamot juice (20 mg/kg) diminished proinflammatory cytokines. Human studies showed that bergamot extract and essential oil may reduce blood pressure and improve mental conditions.9
Vitiligo
In 2019, Shaaban et al. prepared elastic nanocarriers (spanlastics) to deliver psoralen-containing bergamot oil along with PUVB with the intention of harnessing melanogenic activity to treat vitiligo. Histopathologic assessment on rat skin was conducted before clinical treatment in patients with vitiligo. The spanlastics were deemed to be of suitable nanosize and deformable, yielding consistent bergamot oil release. The bergamot oil included in the nanocarrier was found to enhance photostability and photodynamic activity, with the researchers concluding that bergamot oil nanospanlastics with psoralen-UVB therapy shows potential as a vitiligo therapy.10
Two years later, Shaaban evaluated bergamot oil formulated in nanostructured lipid carriers as a photosensitizer for photodynamic treatment of vitiligo. The botanical oil was effectively used in the nanostructured lipid carriers with a gel consistency that delivered sustained release of the oil for 24 hours. Preclinical and clinical results in patients were encouraging for the topical photodynamic treatment of vitiligo, with the nanostructured lipid carriers improving the photostability and photodynamic activity of bergamot oil.6
Photoaging, photoprotection, and safety concerns
Three decades ago, an international cooperative study of the photophysical, photomutagenic, and photocarcinogenic characteristics of bergamot oil and the effect of UVA and UVB sunscreens found that UVB and UVA sunscreens at low concentration (0.5%-1%) in perfumes could not inhibit the phototoxicity of bergamot oil on human skin.11
In a 2015 study assessing the impact of 38% bergamot polyphenolic fraction (a highly concentrated Citrus bergamia fruit extract) on UVB-generated photoaging, Nisticò et al. found that the bergamot compound dose-dependently protected HaCaT cells against UVB-caused oxidative stress and photoaging markers. Suggesting that the high-antioxidant bergamot polyphenolic fraction has potential for use in skin care formulations, the researchers added that the extract seems to induce antiproliferative, immune-modulating, and antiaging activity.12In 2022, Alexa et al. performed in vitro tests and found that natural preparations containing bergamot, orange, and clove essential oils do not significantly alter physiological skin parameters and were deemed safe for topical use. An emulsion with bergamot essential oil was also found to reduce the viability of oral squamous cell carcinoma cells.13
Conclusion
As a photosensitizing agent, bergamot has an established role in skin care. Beyond its niche role in treatments for vitiligo and psoriasis, this botanical product appears to show potential as an anti-inflammatory agent as well as an ingredient to combat photoaging and skin cancer. Much more research is needed to elucidate the possible wider benefits of this Mediterranean staple.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions Inc., a SaaS company used to generate skin care routines in office and as an ecommerce solution. Write to her at dermnews@mdedge.com.
References
1. Juber M. Health benefits of bergamot. WebMD. November 29, 2022. Accessed March 21, 2023.
2. Sun P et al. Mediators Inflamm. 2020 Oct 6;2020:8868107.
3. Cristiano MC et al. Biomedicines. 2022 Apr 30;10(5):1039.
4. Liang Y et al. Phytother Res. 2021 Nov;35(11):6131-47.
5. Quetglas-Llabrés MM et al. Oxid Med Cell Longev. 2022 Apr 25;2022:8615242.
6. Shaaban M et al. Expert Opin Drug Deliv. 2021 Jan;18(1):139-50.
7. McNeely W, Goa KL. Drugs. 1998 Oct;56(4):667-90.
8. Han X, Beaumont C, Stevens N. Biochim Open. 2017 Apr 26;5:1-7.
9. Perna S et al. Food Sci Nutr. 2019 Jan 25;7(2):369-84.
10. Shaaban M et al. Drug Deliv Transl Res. 2019 Dec;9(6):1106-16.
11. Dubertret L et al. J Photochem Photobiol B. 1990 Nov;7(2-4):251-9.
12. Nisticò S et al. J Biol Regul Homeost Agents. 2015 Jul-Sep;29(3):723-8.
13. Alexa VT et al. Molecules. 2022 Feb 1;27(3):990.
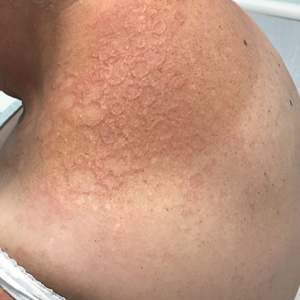
Annular Erythematous Plaques With Central Hypopigmentation on Sun-Exposed Skin
A biopsy showed a markedly elastotic dermis consisting of a palisading granulomatous inflammatory infiltrate and numerous multinucleated histiocytes (Figure). These histopathologic findings along with the clinical presentation confirmed a diagnosis of annular elastolytic granuloma (AEG). Treatment consisting of 3 months of oral minocycline, 2 months of oral doxycycline, and clobetasol ointment all failed. At that point, oral hydroxychloroquine was recommended. Our patient was lost to follow-up by dermatology, then subsequently was placed on hydroxychloroquine by rheumatology to treat both the osteoarthritis and AEG. A follow-up appointment with dermatology was planned for 3 months to monitor hydroxychloroquine treatment and monitor treatment progress; however, she did not follow-up or seek further treatment.
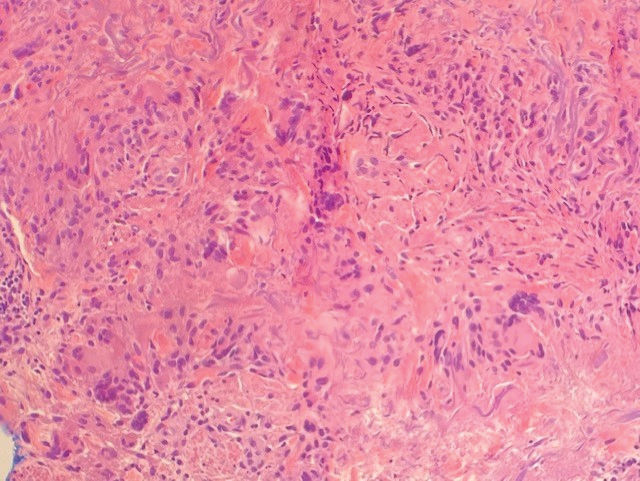
Annular elastolytic granuloma clinically is similar to granuloma annulare (GA), with both presenting as annular plaques surrounded by an elevated border.1 Although AEG clinically is distinct with hypopigmented atrophied plaque centers,2 a biopsy is required to confirm the lack of elastic tissue in zones of atrophy and the presence of multinucleated histiocytes.1,3 Lesions most commonly are seen clinically on sun-exposed areas in middle-aged White women; however, they rarely have been seen on frequently covered skin.4 Our case illustrates the striking photodistribution of AEG, especially on the posterior neck area. The clinical diagnoses of AEG, annular elastolytic giant cell granuloma, and GA in sun-exposed areas are synonymous and can be used interchangeably.5,6
Pathologies considered in the diagnosis of AEG include but are not limited to tinea corporis, annular lichen planus, erythema annulare centrifugum, and necrobiosis lipoidica. Scaling typically is absent in AEG, while tinea corporis presents with hyphae within the stratum corneum of the plaques.7 Papules along the periphery of annular lesions are more typical of annular lichen planus than AEG, and they tend to have a more purple hue.8 Erythema annulare centrifugum has annular erythematous plaques similar to those found in AEG but differs with scaling on the inner margins of these plaques. Histopathology presenting with a lymphocytic infiltrate surrounding vasculature and no indication of elastolytic degradation would further indicate a diagnosis of erythema annulare centrifugum.9 Histopathology showing necrobiosis, lipid depositions, and vascular wall thickenings is indicative of necrobiosis lipoidica.10
Similar to GA,11 the cause of AEG is idiopathic.2 Annular elastolytic granuloma and GA differ in the fact that elastin degradation is characteristic of AEG compared to collagen degradation in GA. It is suspected that elastin degradation in AEG patients is caused by an immune response triggering phagocytosis of elastin by multinucleated histiocytes.2 Actinic damage also is considered a possible cause of elastin fiber degradation in AEG.12 Granuloma annulare can be ruled out and the diagnosis of AEG confirmed with the absence of elastin fibers and mucin on pathology.13
Although there is no established first-line treatment of AEG, successful treatment has been achieved with antimalarial drugs paired with topical steroids.14 Treatment recommendations for AEG include minocycline, chloroquine, hydroxychloroquine, tranilast, and oral retinoids, as well as oral and topical steroids. In clinical cases where AEG occurs in the setting of a chronic disease such as diabetes mellitus, vascular occlusion, arthritis, or hypertension, treatment of underlying disease has been shown to resolve AEG symptoms.14
Although light therapy is not common for AEG, UV light radiation has demonstrated success in treating AEG.15,16 One study showed complete clearance of granulomatous papules after narrowband UVB treatment.15 Another study showed that 2 patients treated with psoralen plus UVA therapy reached complete clearance of AEG lasting at least 3 months after treatment.16
1. Lai JH, Murray SJ, Walsh NM. Evolution of granuloma annulare to mid-dermal elastolysis: report of a case and review of the literature. J Cutan Pathol. 2014;41:462-468. doi:10.1111/cup.12292 2. Klemke CD, Siebold D, Dippel E, et al. Generalised annular elastolytic giant cell granuloma. Dermatology. 2003;207:420-422. doi:10.1159/000074132 3. Limas C. The spectrum of primary cutaneous elastolytic granulomas and their distinction from granuloma annulare: a clinicopathological analysis. Histopathology. 2004;44:277-282. doi:10.1111/j.0309-0167.2004.01755.x 4. Revenga F, Rovira I, Pimentel J, et al. Annular elastolytic giant cell granuloma—actinic granuloma? Clin Exp Dermatol. 1996;21:51-53. 5. Hawryluk EB, Izikson L, English JC 3rd. Non-infectious granulomatous diseases of the skin and their associated systemic diseases: an evidence-based update to important clinical questions. Am J Clin Dermatol. 2010;11:171-181. doi:10.2165/11530080-000000000-00000 6. Berliner JG, Haemel A, LeBoit PE, et al. The sarcoidal variant of annular elastolytic granuloma. J Cutan Pathol. 2013;40:918-920. doi:10.1111/cup.12237 7. Pflederer RT, Ahmed S, Tonkovic-Capin V, et al. Annular polycyclic plaques on the chest and upper back [published online April 24, 2018]. JAAD Case Rep. 2018;4:405-407. doi:10.1016/j.jdcr.2017.07.022 8. Trayes KP, Savage K, Studdiford JS. Annular lesions: diagnosis and treatment. Am Fam Physician. 2018;98:283-291. 9. Weyers W, Diaz-Cascajo C, Weyers I. Erythema annulare centrifugum: results of a clinicopathologic study of 73 patients. Am J Dermatopathol. 2003;25:451-462. doi:10.1097/00000372-200312000-00001 10. Dowling GB, Jones EW. Atypical (annular) necrobiosis lipoidica of the face and scalp. a report of the clinical and histological features of 7 cases. Dermatologica. 1967;135:11-26. doi:10.1159/000254156 11. Piette EW, Rosenbach M. Granuloma annulare: pathogenesis, disease associations and triggers, and therapeutic options. J Am Acad Dermatol. 2016;75:467-479. doi:10.1016/j.jaad.2015 .03.055 12. O’Brien JP, Regan W. Actinically degenerate elastic tissue is the likely antigenic basis of actinic granuloma of the skin and of temporal arteritis [published correction appears in J Am Acad Dermatol. 2000; 42(1 pt 1):148]. J Am Acad Dermatol. 1999;40(2 pt 1):214-222. doi:10.1016/s0190-9622(99)70191-x 13. Rencic A, Nousari CH. Other rheumatologic diseases. In: Bolognia JL, Jorizzo JL, Rapini RP, et al, eds. Dermatology. 2nd ed. Elsevier Limited; 2008:600-601. 14. Burlando M, Herzum A, Cozzani E, et al. Can methotrexate be a successful treatment for unresponsive generalized annular elastolytic giant cell granuloma? case report and review of the literature. Dermatol Ther. 2021;34:E14705. doi:10.1111/dth.14705 15. Takata T, Ikeda M, Kodama H, et al. Regression of papular elastolytic giant cell granuloma using narrow-band UVB irradiation. Dermatology. 2006;212:77-79. doi:10.1159/000089028 16. Pérez-Pérez L, García-Gavín J, Allegue F, et al. Successful treatment of generalized elastolytic giant cell granuloma with psoralenultraviolet A. Photodermatol Photoimmunol Photomed. 2012;28:264-266. doi:10.1111/j.1600-0781.2012.00680.x
A biopsy showed a markedly elastotic dermis consisting of a palisading granulomatous inflammatory infiltrate and numerous multinucleated histiocytes (Figure). These histopathologic findings along with the clinical presentation confirmed a diagnosis of annular elastolytic granuloma (AEG). Treatment consisting of 3 months of oral minocycline, 2 months of oral doxycycline, and clobetasol ointment all failed. At that point, oral hydroxychloroquine was recommended. Our patient was lost to follow-up by dermatology, then subsequently was placed on hydroxychloroquine by rheumatology to treat both the osteoarthritis and AEG. A follow-up appointment with dermatology was planned for 3 months to monitor hydroxychloroquine treatment and monitor treatment progress; however, she did not follow-up or seek further treatment.

Annular elastolytic granuloma clinically is similar to granuloma annulare (GA), with both presenting as annular plaques surrounded by an elevated border.1 Although AEG clinically is distinct with hypopigmented atrophied plaque centers,2 a biopsy is required to confirm the lack of elastic tissue in zones of atrophy and the presence of multinucleated histiocytes.1,3 Lesions most commonly are seen clinically on sun-exposed areas in middle-aged White women; however, they rarely have been seen on frequently covered skin.4 Our case illustrates the striking photodistribution of AEG, especially on the posterior neck area. The clinical diagnoses of AEG, annular elastolytic giant cell granuloma, and GA in sun-exposed areas are synonymous and can be used interchangeably.5,6
Pathologies considered in the diagnosis of AEG include but are not limited to tinea corporis, annular lichen planus, erythema annulare centrifugum, and necrobiosis lipoidica. Scaling typically is absent in AEG, while tinea corporis presents with hyphae within the stratum corneum of the plaques.7 Papules along the periphery of annular lesions are more typical of annular lichen planus than AEG, and they tend to have a more purple hue.8 Erythema annulare centrifugum has annular erythematous plaques similar to those found in AEG but differs with scaling on the inner margins of these plaques. Histopathology presenting with a lymphocytic infiltrate surrounding vasculature and no indication of elastolytic degradation would further indicate a diagnosis of erythema annulare centrifugum.9 Histopathology showing necrobiosis, lipid depositions, and vascular wall thickenings is indicative of necrobiosis lipoidica.10
Similar to GA,11 the cause of AEG is idiopathic.2 Annular elastolytic granuloma and GA differ in the fact that elastin degradation is characteristic of AEG compared to collagen degradation in GA. It is suspected that elastin degradation in AEG patients is caused by an immune response triggering phagocytosis of elastin by multinucleated histiocytes.2 Actinic damage also is considered a possible cause of elastin fiber degradation in AEG.12 Granuloma annulare can be ruled out and the diagnosis of AEG confirmed with the absence of elastin fibers and mucin on pathology.13
Although there is no established first-line treatment of AEG, successful treatment has been achieved with antimalarial drugs paired with topical steroids.14 Treatment recommendations for AEG include minocycline, chloroquine, hydroxychloroquine, tranilast, and oral retinoids, as well as oral and topical steroids. In clinical cases where AEG occurs in the setting of a chronic disease such as diabetes mellitus, vascular occlusion, arthritis, or hypertension, treatment of underlying disease has been shown to resolve AEG symptoms.14
Although light therapy is not common for AEG, UV light radiation has demonstrated success in treating AEG.15,16 One study showed complete clearance of granulomatous papules after narrowband UVB treatment.15 Another study showed that 2 patients treated with psoralen plus UVA therapy reached complete clearance of AEG lasting at least 3 months after treatment.16
A biopsy showed a markedly elastotic dermis consisting of a palisading granulomatous inflammatory infiltrate and numerous multinucleated histiocytes (Figure). These histopathologic findings along with the clinical presentation confirmed a diagnosis of annular elastolytic granuloma (AEG). Treatment consisting of 3 months of oral minocycline, 2 months of oral doxycycline, and clobetasol ointment all failed. At that point, oral hydroxychloroquine was recommended. Our patient was lost to follow-up by dermatology, then subsequently was placed on hydroxychloroquine by rheumatology to treat both the osteoarthritis and AEG. A follow-up appointment with dermatology was planned for 3 months to monitor hydroxychloroquine treatment and monitor treatment progress; however, she did not follow-up or seek further treatment.

Annular elastolytic granuloma clinically is similar to granuloma annulare (GA), with both presenting as annular plaques surrounded by an elevated border.1 Although AEG clinically is distinct with hypopigmented atrophied plaque centers,2 a biopsy is required to confirm the lack of elastic tissue in zones of atrophy and the presence of multinucleated histiocytes.1,3 Lesions most commonly are seen clinically on sun-exposed areas in middle-aged White women; however, they rarely have been seen on frequently covered skin.4 Our case illustrates the striking photodistribution of AEG, especially on the posterior neck area. The clinical diagnoses of AEG, annular elastolytic giant cell granuloma, and GA in sun-exposed areas are synonymous and can be used interchangeably.5,6
Pathologies considered in the diagnosis of AEG include but are not limited to tinea corporis, annular lichen planus, erythema annulare centrifugum, and necrobiosis lipoidica. Scaling typically is absent in AEG, while tinea corporis presents with hyphae within the stratum corneum of the plaques.7 Papules along the periphery of annular lesions are more typical of annular lichen planus than AEG, and they tend to have a more purple hue.8 Erythema annulare centrifugum has annular erythematous plaques similar to those found in AEG but differs with scaling on the inner margins of these plaques. Histopathology presenting with a lymphocytic infiltrate surrounding vasculature and no indication of elastolytic degradation would further indicate a diagnosis of erythema annulare centrifugum.9 Histopathology showing necrobiosis, lipid depositions, and vascular wall thickenings is indicative of necrobiosis lipoidica.10
Similar to GA,11 the cause of AEG is idiopathic.2 Annular elastolytic granuloma and GA differ in the fact that elastin degradation is characteristic of AEG compared to collagen degradation in GA. It is suspected that elastin degradation in AEG patients is caused by an immune response triggering phagocytosis of elastin by multinucleated histiocytes.2 Actinic damage also is considered a possible cause of elastin fiber degradation in AEG.12 Granuloma annulare can be ruled out and the diagnosis of AEG confirmed with the absence of elastin fibers and mucin on pathology.13
Although there is no established first-line treatment of AEG, successful treatment has been achieved with antimalarial drugs paired with topical steroids.14 Treatment recommendations for AEG include minocycline, chloroquine, hydroxychloroquine, tranilast, and oral retinoids, as well as oral and topical steroids. In clinical cases where AEG occurs in the setting of a chronic disease such as diabetes mellitus, vascular occlusion, arthritis, or hypertension, treatment of underlying disease has been shown to resolve AEG symptoms.14
Although light therapy is not common for AEG, UV light radiation has demonstrated success in treating AEG.15,16 One study showed complete clearance of granulomatous papules after narrowband UVB treatment.15 Another study showed that 2 patients treated with psoralen plus UVA therapy reached complete clearance of AEG lasting at least 3 months after treatment.16
1. Lai JH, Murray SJ, Walsh NM. Evolution of granuloma annulare to mid-dermal elastolysis: report of a case and review of the literature. J Cutan Pathol. 2014;41:462-468. doi:10.1111/cup.12292 2. Klemke CD, Siebold D, Dippel E, et al. Generalised annular elastolytic giant cell granuloma. Dermatology. 2003;207:420-422. doi:10.1159/000074132 3. Limas C. The spectrum of primary cutaneous elastolytic granulomas and their distinction from granuloma annulare: a clinicopathological analysis. Histopathology. 2004;44:277-282. doi:10.1111/j.0309-0167.2004.01755.x 4. Revenga F, Rovira I, Pimentel J, et al. Annular elastolytic giant cell granuloma—actinic granuloma? Clin Exp Dermatol. 1996;21:51-53. 5. Hawryluk EB, Izikson L, English JC 3rd. Non-infectious granulomatous diseases of the skin and their associated systemic diseases: an evidence-based update to important clinical questions. Am J Clin Dermatol. 2010;11:171-181. doi:10.2165/11530080-000000000-00000 6. Berliner JG, Haemel A, LeBoit PE, et al. The sarcoidal variant of annular elastolytic granuloma. J Cutan Pathol. 2013;40:918-920. doi:10.1111/cup.12237 7. Pflederer RT, Ahmed S, Tonkovic-Capin V, et al. Annular polycyclic plaques on the chest and upper back [published online April 24, 2018]. JAAD Case Rep. 2018;4:405-407. doi:10.1016/j.jdcr.2017.07.022 8. Trayes KP, Savage K, Studdiford JS. Annular lesions: diagnosis and treatment. Am Fam Physician. 2018;98:283-291. 9. Weyers W, Diaz-Cascajo C, Weyers I. Erythema annulare centrifugum: results of a clinicopathologic study of 73 patients. Am J Dermatopathol. 2003;25:451-462. doi:10.1097/00000372-200312000-00001 10. Dowling GB, Jones EW. Atypical (annular) necrobiosis lipoidica of the face and scalp. a report of the clinical and histological features of 7 cases. Dermatologica. 1967;135:11-26. doi:10.1159/000254156 11. Piette EW, Rosenbach M. Granuloma annulare: pathogenesis, disease associations and triggers, and therapeutic options. J Am Acad Dermatol. 2016;75:467-479. doi:10.1016/j.jaad.2015 .03.055 12. O’Brien JP, Regan W. Actinically degenerate elastic tissue is the likely antigenic basis of actinic granuloma of the skin and of temporal arteritis [published correction appears in J Am Acad Dermatol. 2000; 42(1 pt 1):148]. J Am Acad Dermatol. 1999;40(2 pt 1):214-222. doi:10.1016/s0190-9622(99)70191-x 13. Rencic A, Nousari CH. Other rheumatologic diseases. In: Bolognia JL, Jorizzo JL, Rapini RP, et al, eds. Dermatology. 2nd ed. Elsevier Limited; 2008:600-601. 14. Burlando M, Herzum A, Cozzani E, et al. Can methotrexate be a successful treatment for unresponsive generalized annular elastolytic giant cell granuloma? case report and review of the literature. Dermatol Ther. 2021;34:E14705. doi:10.1111/dth.14705 15. Takata T, Ikeda M, Kodama H, et al. Regression of papular elastolytic giant cell granuloma using narrow-band UVB irradiation. Dermatology. 2006;212:77-79. doi:10.1159/000089028 16. Pérez-Pérez L, García-Gavín J, Allegue F, et al. Successful treatment of generalized elastolytic giant cell granuloma with psoralenultraviolet A. Photodermatol Photoimmunol Photomed. 2012;28:264-266. doi:10.1111/j.1600-0781.2012.00680.x
1. Lai JH, Murray SJ, Walsh NM. Evolution of granuloma annulare to mid-dermal elastolysis: report of a case and review of the literature. J Cutan Pathol. 2014;41:462-468. doi:10.1111/cup.12292 2. Klemke CD, Siebold D, Dippel E, et al. Generalised annular elastolytic giant cell granuloma. Dermatology. 2003;207:420-422. doi:10.1159/000074132 3. Limas C. The spectrum of primary cutaneous elastolytic granulomas and their distinction from granuloma annulare: a clinicopathological analysis. Histopathology. 2004;44:277-282. doi:10.1111/j.0309-0167.2004.01755.x 4. Revenga F, Rovira I, Pimentel J, et al. Annular elastolytic giant cell granuloma—actinic granuloma? Clin Exp Dermatol. 1996;21:51-53. 5. Hawryluk EB, Izikson L, English JC 3rd. Non-infectious granulomatous diseases of the skin and their associated systemic diseases: an evidence-based update to important clinical questions. Am J Clin Dermatol. 2010;11:171-181. doi:10.2165/11530080-000000000-00000 6. Berliner JG, Haemel A, LeBoit PE, et al. The sarcoidal variant of annular elastolytic granuloma. J Cutan Pathol. 2013;40:918-920. doi:10.1111/cup.12237 7. Pflederer RT, Ahmed S, Tonkovic-Capin V, et al. Annular polycyclic plaques on the chest and upper back [published online April 24, 2018]. JAAD Case Rep. 2018;4:405-407. doi:10.1016/j.jdcr.2017.07.022 8. Trayes KP, Savage K, Studdiford JS. Annular lesions: diagnosis and treatment. Am Fam Physician. 2018;98:283-291. 9. Weyers W, Diaz-Cascajo C, Weyers I. Erythema annulare centrifugum: results of a clinicopathologic study of 73 patients. Am J Dermatopathol. 2003;25:451-462. doi:10.1097/00000372-200312000-00001 10. Dowling GB, Jones EW. Atypical (annular) necrobiosis lipoidica of the face and scalp. a report of the clinical and histological features of 7 cases. Dermatologica. 1967;135:11-26. doi:10.1159/000254156 11. Piette EW, Rosenbach M. Granuloma annulare: pathogenesis, disease associations and triggers, and therapeutic options. J Am Acad Dermatol. 2016;75:467-479. doi:10.1016/j.jaad.2015 .03.055 12. O’Brien JP, Regan W. Actinically degenerate elastic tissue is the likely antigenic basis of actinic granuloma of the skin and of temporal arteritis [published correction appears in J Am Acad Dermatol. 2000; 42(1 pt 1):148]. J Am Acad Dermatol. 1999;40(2 pt 1):214-222. doi:10.1016/s0190-9622(99)70191-x 13. Rencic A, Nousari CH. Other rheumatologic diseases. In: Bolognia JL, Jorizzo JL, Rapini RP, et al, eds. Dermatology. 2nd ed. Elsevier Limited; 2008:600-601. 14. Burlando M, Herzum A, Cozzani E, et al. Can methotrexate be a successful treatment for unresponsive generalized annular elastolytic giant cell granuloma? case report and review of the literature. Dermatol Ther. 2021;34:E14705. doi:10.1111/dth.14705 15. Takata T, Ikeda M, Kodama H, et al. Regression of papular elastolytic giant cell granuloma using narrow-band UVB irradiation. Dermatology. 2006;212:77-79. doi:10.1159/000089028 16. Pérez-Pérez L, García-Gavín J, Allegue F, et al. Successful treatment of generalized elastolytic giant cell granuloma with psoralenultraviolet A. Photodermatol Photoimmunol Photomed. 2012;28:264-266. doi:10.1111/j.1600-0781.2012.00680.x
A 67-year-old White woman presented to our dermatology clinic with pruritic annular erythematous plaques with central hypopigmentation on the forearms, dorsal aspect of the hands, neck, and fingers of 3 to 4 months’ duration. The patient rated the severity of pruritus an 8 on a 10-point scale. A review of symptoms was positive for fatigue, joint pain, and headache. The patient had a history of type 2 diabetes mellitus, osteoarthritis, thyroid disease, and stage 3 renal failure. A punch biopsy from the left forearm was performed.
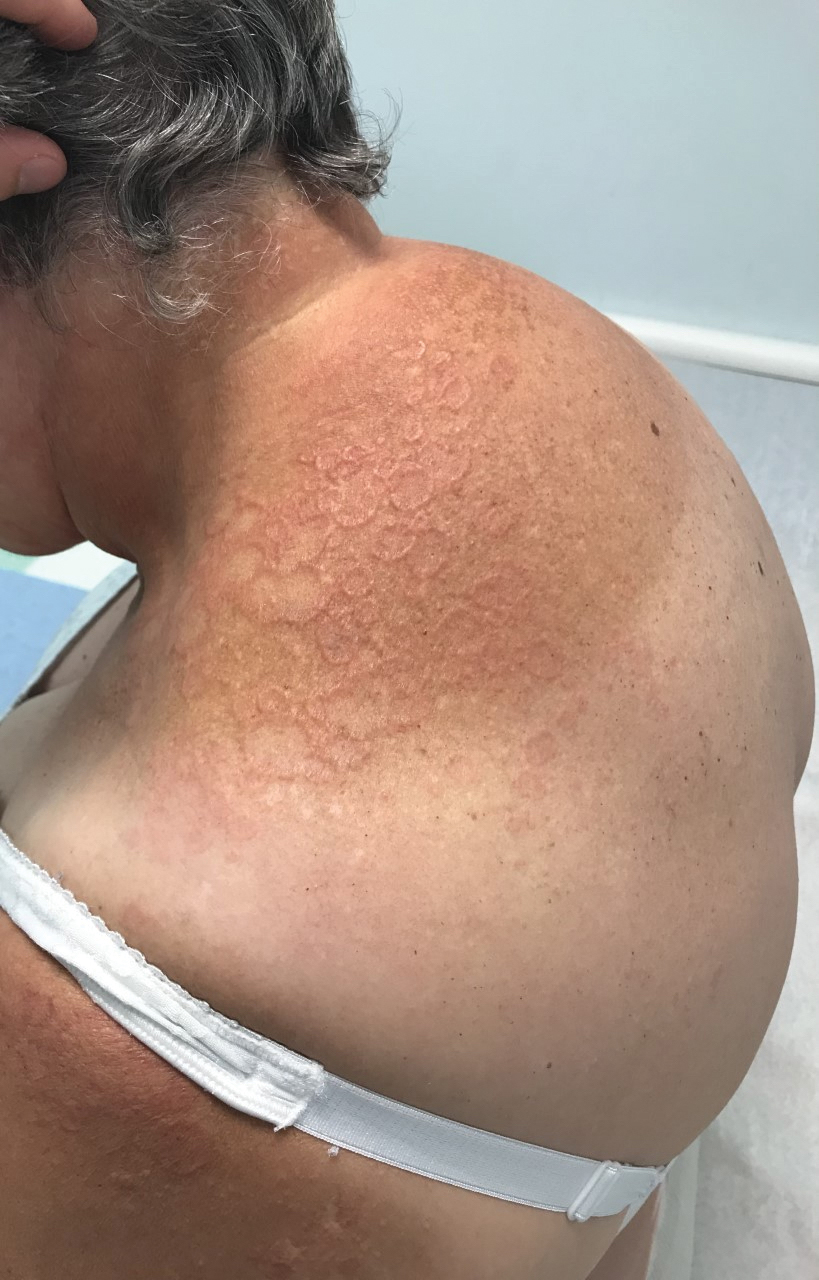
Periorbital Orange Spots
The Diagnosis: Orange Palpebral Spots
The clinical presentation of our patient was consistent with a diagnosis of orange palpebral spots (OPSs), an uncommon discoloration that most often appears in White patients in the fifth or sixth decades of life. Orange palpebral spots were first described in 2008 by Assouly et al1 in 27 patients (23 females and 4 males). In 2015, Belliveau et al2 expanded the designation to yellow-orange palpebral spots because they felt the term more fully expressed the color variations depicted in their patients; however, this term more frequently is used in ophthalmology.
Orange palpebral spots commonly appear as asymptomatic, yellow-orange, symmetric lesions with a predilection for the recessed areas of the superior eyelids but also can present on the canthi and inferior eyelids. The discolorations are more easily visible on fair skin and have been reported to measure from 10 to 15 mm in the long axis.3 Assouly et al1 described the orange spots as having indistinct margins, with borders similar to “sand on a sea shore.” Orange palpebral spots can be a persistent discoloration, and there are no reports of spontaneous regression. No known association with malignancy or systemic illness has been reported.
Case reports of OPSs describe histologic similarities between specimens, including increased adipose tissue and pigment-laden macrophages in the superficial dermis.2 The pigmented deposits sometimes may be found in the basal keratinocytes of the epidermis and turn black with Fontana-Masson stain.1 No inflammatory infiltrates, necrosis, or xanthomization are characteristically found. Stains for iron, mucin, and amyloid also have been negative.2
The cause of pigmentation in OPSs is unknown; however, lipofuscin deposits and high-situated adipocytes in the reticular dermis colored by carotenoids have been proposed as possible mechanisms.1 No unifying cause for pigmentation in the serum (eg, cholesterol, triglycerides, thyroid-stimulating hormone, free retinol, vitamin E, carotenoids) was found in 11 of 27 patients with OPSs assessed by Assouly et al.1 In one case, lipofuscin, a degradation product of lysosomes, was detected by microscopic autofluorescence in the superficial dermis. However, lipofuscin typically is a breakdown product associated with aging, and OPSs have been present in patients as young as 28 years.1 Local trauma related to eye rubbing is another theory that has been proposed due to the finding of melanin in the superficial dermis. However, the absence of hemosiderin deposits as well as the extensive duration of the discolorations makes local trauma a less likely explanation for the etiology of OPSs.2
The clinical differential diagnosis for OPSs includes xanthelasma, jaundice, and carotenoderma. Xanthelasma presents as elevated yellow plaques usually found over the medial aspect of the eyes. In contrast, OPSs are nonelevated with both orange and yellow hues typically present. Histologic samples of xanthelasma are characterized by lipid-laden macrophages (foam cells) in the dermis in contrast to the adipose tissue seen in OPSs that has not been phagocytized.1,2 The lack of scleral icterus made jaundice an unlikely diagnosis in our patient. Bilirubin elevations substantial enough to cause skin discoloration also would be expected to discolor the conjunctiva. In carotenoderma, carotenoids are deposited in the sweat and sebum of the stratum corneum with the orange pigmentation most prominent in regions of increased sweating such as the palms, soles, and nasolabial folds.4 Our patient’s lack of discoloration in places other than the periorbital region made carotenoderma less likely.
In the study by Assouly et al,1 10 of 11 patients who underwent laboratory analysis self-reported eating a diet rich in fruit and vegetables, though no standardized questionnaire was given. One patient was found to have an elevated vitamin E level, and in 5 cases there was an elevated level of β-cryptoxanthin. The significance of these elevations in such a small minority is unknown, and increased β-cryptoxanthin has been attributed to increased consumption of citrus fruits during the winter season. Our patient reported ingesting a daily oral supplement rich in carotenoids that constituted 60% of the daily value of vitamin E including mixed tocopherols as well as 90% of the daily value of vitamin A with many sources of carotenoids including beta-carotenes, lutein/zeaxanthin, lycopene, and astaxanthin. An invasive biopsy was not taken in this case, as OPSs largely are diagnosed clinically. Greater awareness and recognition of OPSs may help to identify common underlying causes for this unique diagnosis.
- Assouly P, Cavelier-Balloy B, Dupré T. Orange palpebral spots. Dermatology. 2008;216:166-170.
- Belliveau MJ, Odashiro AN, Harvey JT. Yellow-orange palpebral spots. Ophthalmology. 2015;122:2139-2140.
- Kluger N, Guillot B. Bilateral orange discoloration of the upper eyelids: a quiz. Acta Derm Venereol. 2011;91:211-212.
- Maharshak N, Shapiro J, Trau H. Carotenoderma—a review of the current literature. Int J Dermatol. 2003;42:178-181.
The Diagnosis: Orange Palpebral Spots
The clinical presentation of our patient was consistent with a diagnosis of orange palpebral spots (OPSs), an uncommon discoloration that most often appears in White patients in the fifth or sixth decades of life. Orange palpebral spots were first described in 2008 by Assouly et al1 in 27 patients (23 females and 4 males). In 2015, Belliveau et al2 expanded the designation to yellow-orange palpebral spots because they felt the term more fully expressed the color variations depicted in their patients; however, this term more frequently is used in ophthalmology.
Orange palpebral spots commonly appear as asymptomatic, yellow-orange, symmetric lesions with a predilection for the recessed areas of the superior eyelids but also can present on the canthi and inferior eyelids. The discolorations are more easily visible on fair skin and have been reported to measure from 10 to 15 mm in the long axis.3 Assouly et al1 described the orange spots as having indistinct margins, with borders similar to “sand on a sea shore.” Orange palpebral spots can be a persistent discoloration, and there are no reports of spontaneous regression. No known association with malignancy or systemic illness has been reported.
Case reports of OPSs describe histologic similarities between specimens, including increased adipose tissue and pigment-laden macrophages in the superficial dermis.2 The pigmented deposits sometimes may be found in the basal keratinocytes of the epidermis and turn black with Fontana-Masson stain.1 No inflammatory infiltrates, necrosis, or xanthomization are characteristically found. Stains for iron, mucin, and amyloid also have been negative.2
The cause of pigmentation in OPSs is unknown; however, lipofuscin deposits and high-situated adipocytes in the reticular dermis colored by carotenoids have been proposed as possible mechanisms.1 No unifying cause for pigmentation in the serum (eg, cholesterol, triglycerides, thyroid-stimulating hormone, free retinol, vitamin E, carotenoids) was found in 11 of 27 patients with OPSs assessed by Assouly et al.1 In one case, lipofuscin, a degradation product of lysosomes, was detected by microscopic autofluorescence in the superficial dermis. However, lipofuscin typically is a breakdown product associated with aging, and OPSs have been present in patients as young as 28 years.1 Local trauma related to eye rubbing is another theory that has been proposed due to the finding of melanin in the superficial dermis. However, the absence of hemosiderin deposits as well as the extensive duration of the discolorations makes local trauma a less likely explanation for the etiology of OPSs.2
The clinical differential diagnosis for OPSs includes xanthelasma, jaundice, and carotenoderma. Xanthelasma presents as elevated yellow plaques usually found over the medial aspect of the eyes. In contrast, OPSs are nonelevated with both orange and yellow hues typically present. Histologic samples of xanthelasma are characterized by lipid-laden macrophages (foam cells) in the dermis in contrast to the adipose tissue seen in OPSs that has not been phagocytized.1,2 The lack of scleral icterus made jaundice an unlikely diagnosis in our patient. Bilirubin elevations substantial enough to cause skin discoloration also would be expected to discolor the conjunctiva. In carotenoderma, carotenoids are deposited in the sweat and sebum of the stratum corneum with the orange pigmentation most prominent in regions of increased sweating such as the palms, soles, and nasolabial folds.4 Our patient’s lack of discoloration in places other than the periorbital region made carotenoderma less likely.
In the study by Assouly et al,1 10 of 11 patients who underwent laboratory analysis self-reported eating a diet rich in fruit and vegetables, though no standardized questionnaire was given. One patient was found to have an elevated vitamin E level, and in 5 cases there was an elevated level of β-cryptoxanthin. The significance of these elevations in such a small minority is unknown, and increased β-cryptoxanthin has been attributed to increased consumption of citrus fruits during the winter season. Our patient reported ingesting a daily oral supplement rich in carotenoids that constituted 60% of the daily value of vitamin E including mixed tocopherols as well as 90% of the daily value of vitamin A with many sources of carotenoids including beta-carotenes, lutein/zeaxanthin, lycopene, and astaxanthin. An invasive biopsy was not taken in this case, as OPSs largely are diagnosed clinically. Greater awareness and recognition of OPSs may help to identify common underlying causes for this unique diagnosis.
The Diagnosis: Orange Palpebral Spots
The clinical presentation of our patient was consistent with a diagnosis of orange palpebral spots (OPSs), an uncommon discoloration that most often appears in White patients in the fifth or sixth decades of life. Orange palpebral spots were first described in 2008 by Assouly et al1 in 27 patients (23 females and 4 males). In 2015, Belliveau et al2 expanded the designation to yellow-orange palpebral spots because they felt the term more fully expressed the color variations depicted in their patients; however, this term more frequently is used in ophthalmology.
Orange palpebral spots commonly appear as asymptomatic, yellow-orange, symmetric lesions with a predilection for the recessed areas of the superior eyelids but also can present on the canthi and inferior eyelids. The discolorations are more easily visible on fair skin and have been reported to measure from 10 to 15 mm in the long axis.3 Assouly et al1 described the orange spots as having indistinct margins, with borders similar to “sand on a sea shore.” Orange palpebral spots can be a persistent discoloration, and there are no reports of spontaneous regression. No known association with malignancy or systemic illness has been reported.
Case reports of OPSs describe histologic similarities between specimens, including increased adipose tissue and pigment-laden macrophages in the superficial dermis.2 The pigmented deposits sometimes may be found in the basal keratinocytes of the epidermis and turn black with Fontana-Masson stain.1 No inflammatory infiltrates, necrosis, or xanthomization are characteristically found. Stains for iron, mucin, and amyloid also have been negative.2
The cause of pigmentation in OPSs is unknown; however, lipofuscin deposits and high-situated adipocytes in the reticular dermis colored by carotenoids have been proposed as possible mechanisms.1 No unifying cause for pigmentation in the serum (eg, cholesterol, triglycerides, thyroid-stimulating hormone, free retinol, vitamin E, carotenoids) was found in 11 of 27 patients with OPSs assessed by Assouly et al.1 In one case, lipofuscin, a degradation product of lysosomes, was detected by microscopic autofluorescence in the superficial dermis. However, lipofuscin typically is a breakdown product associated with aging, and OPSs have been present in patients as young as 28 years.1 Local trauma related to eye rubbing is another theory that has been proposed due to the finding of melanin in the superficial dermis. However, the absence of hemosiderin deposits as well as the extensive duration of the discolorations makes local trauma a less likely explanation for the etiology of OPSs.2
The clinical differential diagnosis for OPSs includes xanthelasma, jaundice, and carotenoderma. Xanthelasma presents as elevated yellow plaques usually found over the medial aspect of the eyes. In contrast, OPSs are nonelevated with both orange and yellow hues typically present. Histologic samples of xanthelasma are characterized by lipid-laden macrophages (foam cells) in the dermis in contrast to the adipose tissue seen in OPSs that has not been phagocytized.1,2 The lack of scleral icterus made jaundice an unlikely diagnosis in our patient. Bilirubin elevations substantial enough to cause skin discoloration also would be expected to discolor the conjunctiva. In carotenoderma, carotenoids are deposited in the sweat and sebum of the stratum corneum with the orange pigmentation most prominent in regions of increased sweating such as the palms, soles, and nasolabial folds.4 Our patient’s lack of discoloration in places other than the periorbital region made carotenoderma less likely.
In the study by Assouly et al,1 10 of 11 patients who underwent laboratory analysis self-reported eating a diet rich in fruit and vegetables, though no standardized questionnaire was given. One patient was found to have an elevated vitamin E level, and in 5 cases there was an elevated level of β-cryptoxanthin. The significance of these elevations in such a small minority is unknown, and increased β-cryptoxanthin has been attributed to increased consumption of citrus fruits during the winter season. Our patient reported ingesting a daily oral supplement rich in carotenoids that constituted 60% of the daily value of vitamin E including mixed tocopherols as well as 90% of the daily value of vitamin A with many sources of carotenoids including beta-carotenes, lutein/zeaxanthin, lycopene, and astaxanthin. An invasive biopsy was not taken in this case, as OPSs largely are diagnosed clinically. Greater awareness and recognition of OPSs may help to identify common underlying causes for this unique diagnosis.
- Assouly P, Cavelier-Balloy B, Dupré T. Orange palpebral spots. Dermatology. 2008;216:166-170.
- Belliveau MJ, Odashiro AN, Harvey JT. Yellow-orange palpebral spots. Ophthalmology. 2015;122:2139-2140.
- Kluger N, Guillot B. Bilateral orange discoloration of the upper eyelids: a quiz. Acta Derm Venereol. 2011;91:211-212.
- Maharshak N, Shapiro J, Trau H. Carotenoderma—a review of the current literature. Int J Dermatol. 2003;42:178-181.
- Assouly P, Cavelier-Balloy B, Dupré T. Orange palpebral spots. Dermatology. 2008;216:166-170.
- Belliveau MJ, Odashiro AN, Harvey JT. Yellow-orange palpebral spots. Ophthalmology. 2015;122:2139-2140.
- Kluger N, Guillot B. Bilateral orange discoloration of the upper eyelids: a quiz. Acta Derm Venereol. 2011;91:211-212.
- Maharshak N, Shapiro J, Trau H. Carotenoderma—a review of the current literature. Int J Dermatol. 2003;42:178-181.
A 63-year-old White man with a history of melanoma presented to our dermatology clinic for evaluation of gradually worsening yellow discoloration around the eyes of 2 years’ duration. Physical examination revealed periorbital yellow-orange patches (top). The discolorations were nonelevated and nonpalpable. Dermoscopy revealed yellow blotches with sparing of the hair follicles (bottom). The remainder of the skin examination was unremarkable.
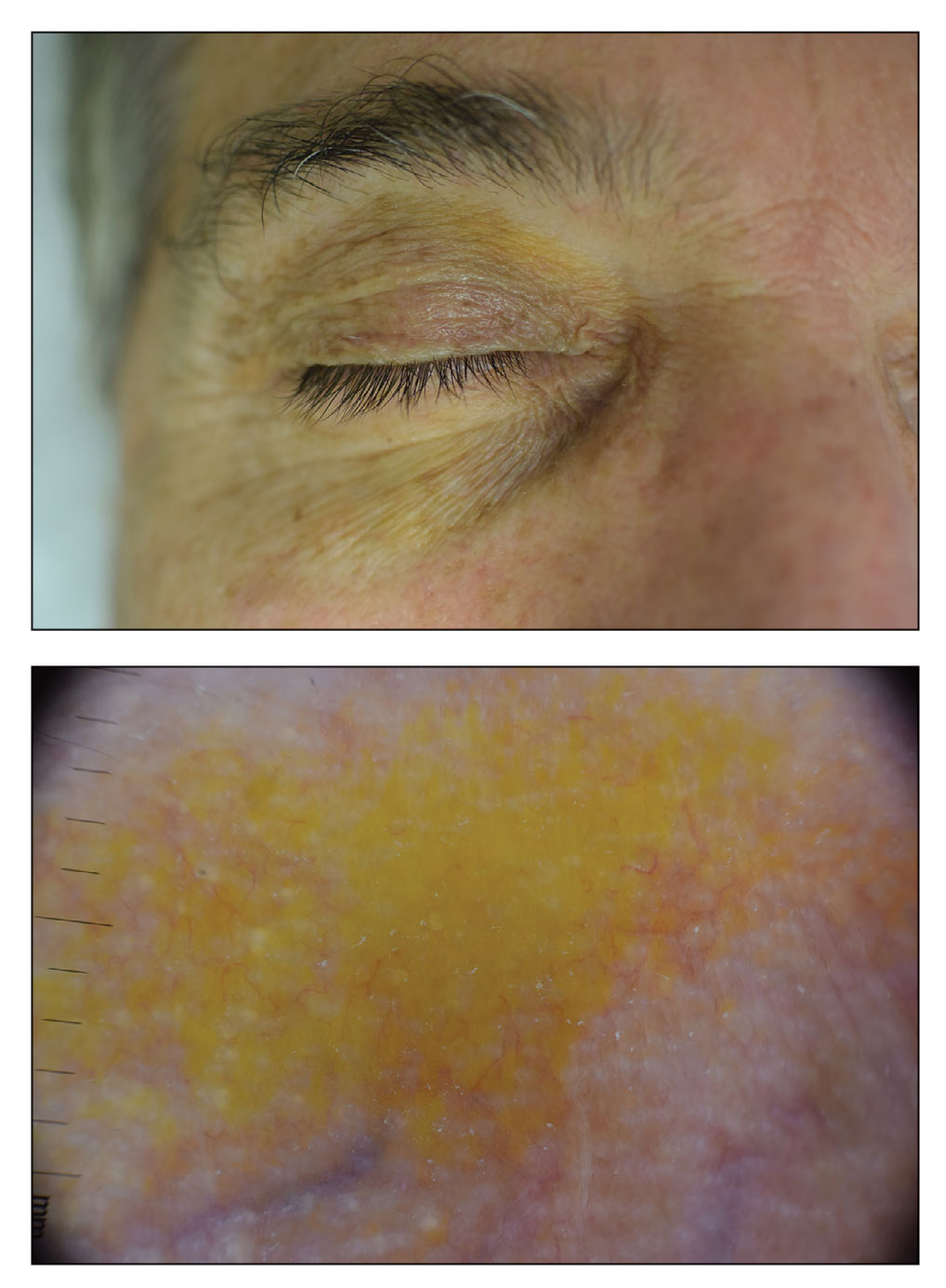
Vitiligo
THE COMPARISON
A Vitiligo in a young Hispanic female, which spared the area under a ring. The patient has spotty return of pigment on the hand after narrowband UVB treatment.
B Vitiligo on the hand in a young Hispanic male.
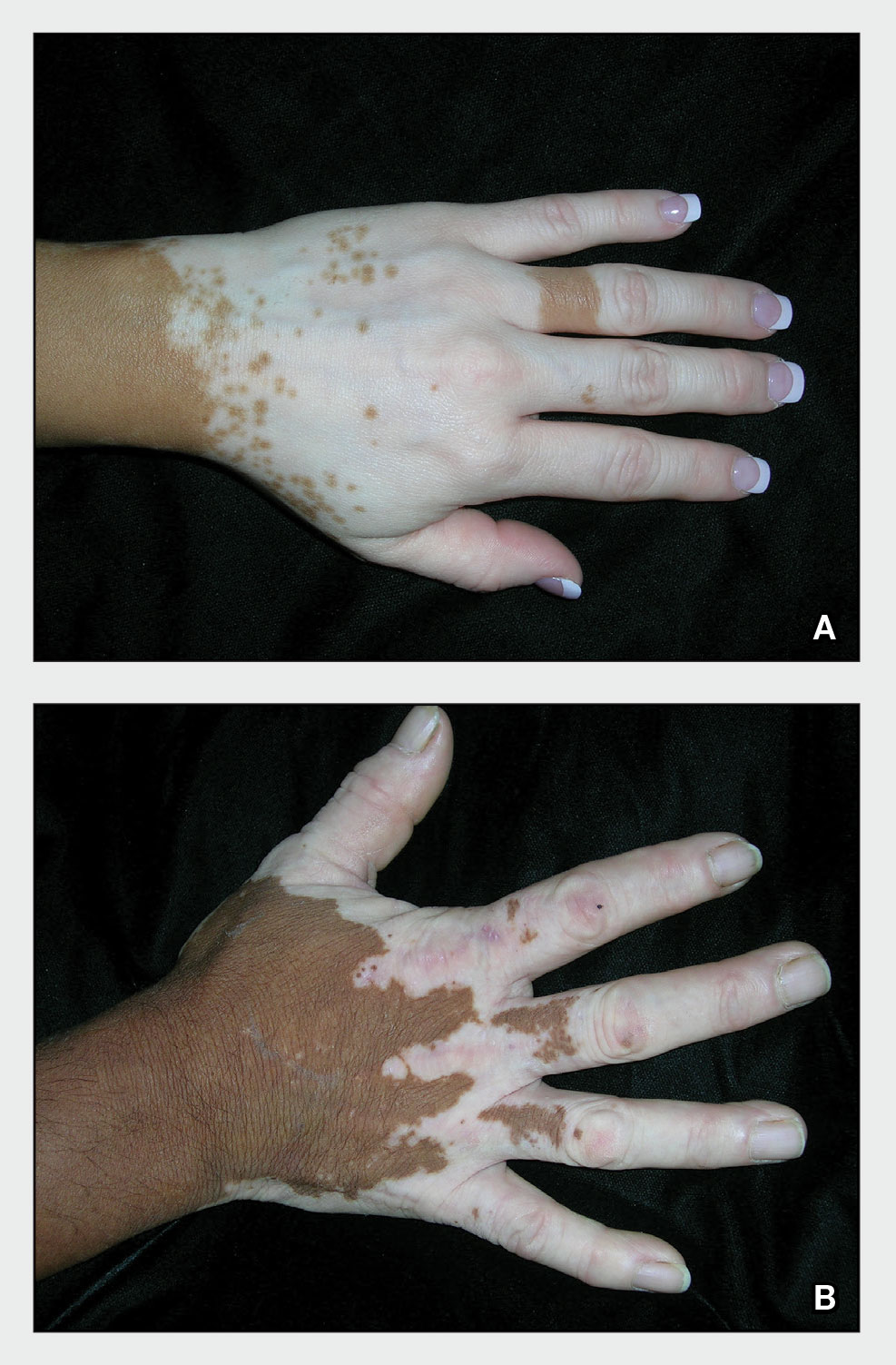
Vitiligo is a chronic autoimmune disorder characterized by areas of depigmented white patches on the skin due to the loss of melanocytes in the epidermis. Various theories on the pathogenesis of vitiligo exist; however, autoimmune destruction of melanocytes remains the leading hypothesis, followed by intrinsic defects in melanocytes.1 Vitiligo is associated with various autoimmune diseases but is most frequently reported in conjunction with thyroid disorders.2
Epidemiology
Vitiligo affects approximately 1% of the US population and up to 8% worldwide.2 There is no difference in prevalence between races or genders. Females typically acquire the disease earlier than males. Onset may occur at any age, although about half of patients will have vitiligo by 20 years of age.1
Key clinical features in people with darker skin tones
Bright white patches are characteristic of vitiligo. The patches typically are asymptomatic and often affect the hands (Figures A and B), perioral skin, feet, and scalp, as well as areas more vulnerable to friction and trauma, such as the elbows and knees.2 Trichrome lesions—consisting of varying zones of white (depigmented), lighter brown (hypopigmented), and normal skin—are most commonly seen in individuals with darker skin. Trichrome vitiligo is considered an actively progressing variant of vitiligo.2
An important distinction when diagnosing vitiligo is evaluating for segmental vs nonsegmental vitiligo. Although nonsegmental vitiligo—the more common subtype—is characterized by symmetric distribution and a less predictable course, segmental vitiligo manifests in a localized and unilateral distribution, often avoiding extension past the midline. Segmental vitiligo typically manifests at a younger age and follows a more rapidly stabilizing course.3
Worth noting
Given that stark contrasts between pigmented and depigmented lesions are more prominent in darker skin tones, vitiligo can be more socially stigmatizing and psychologically devastating in these patients.4,5
Treatment of vitiligo includes narrowband UVB (NB-UVB) light phototherapy, excimer laser, topical corticosteroids, topical calcineurin inhibitors such as tacrolimus and pimecrolimus, and surgical melanocyte transplantation.1 In July 2022, ruxolitinib cream 1.5% was approved by the US Food and Drug Administration (FDA) for nonsegmental vitiligo in patients 12 years and older.6,7 It is the only FDA-approved therapy for vitiligo. It is thought to work by inhibiting the Janus kinase– signal transducers and activators of the transcription pathway.6 However, topical ruxolitinib is expensive, costing more than $2000 for 60 g.8
Health disparity highlight
A 2021 study reviewing the coverage policies of 15 commercial health care insurance companies, 50 BlueCross BlueShield plans, Medicaid, Medicare, and Veterans Affairs plans found inequities in the insurance coverage patterns for therapies used to treat vitiligo. There were 2 commonly cited reasons for denying coverage for therapies: vitiligo was considered cosmetic and therapies were not FDA approved.7 In comparison, NB-UVB light phototherapy for psoriasis is not considered cosmetic and has a much higher insurance coverage rate.9,10 The out-of-pocket cost for a patient to purchase their own NB-UVB light phototherapy is more than $5000.11 Not all patients of color are economically disadvantaged, but in the United States, Black and Hispanic populations experience disproportionately higher rates of poverty (19% and 17%, respectively) compared to their White counterparts (8%).12
Final thoughts
US Food and Drug Administration approval of new drugs or new treatment indications comes after years of research discovery and large-scale trials. This pursuit of new discovery, however, is uneven. Vitiligo has historically been understudied and underfunded for research; this is common among several conditions adversely affecting people of color in the United States.13
- Rashighi M, Harris JE. Vitiligo pathogenesis and emerging treatments. Dermatol Clin. 2017;35:257-265. doi:10.1016/j.det.2016.11.014
- Alikhan A, Felsten LM, Daly M, et al. Vitiligo: a comprehensive overview part I. introduction, epidemiology, quality of life, diagnosis, differential diagnosis, associations, histopathology, etiology, and work-up. J Am Acad Dermatol. 2011;65:473-491. doi:10.1016/j.jaad.2010.11.061
- van Geel N, Speeckaert R. Segmental vitiligo. Dermatol Clin. 2017; 35:145-150. doi:10.1016/j.det.2016.11.005
- Grimes PE, Miller MM. Vitiligo: patient stories, self-esteem, and the psychological burden of disease. Int J Womens Dermatol. 2018;4:32-37. doi:10.1016/j.ijwd.2017.11.005
- Ezzedine K, Eleftheriadou V, Jones H, et al. Psychosocial effects of vitiligo: a systematic literature review [published online September 23, 2021]. Am J Clin Dermatol. 2021;22:757-774. doi:10.1007/s40257 -021-00631-6
- FDA approves topical treatment addressing repigmentation in vitiligo in patients aged 12 and older. News release. US Food and Drug Administration; July 19, 2022. Accessed December 27, 2022. https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-topical-treatment-addressing-repigmentation-vitiligo-patients -aged-12-and-older
- Blundell A, Sachar M, Gabel CK, et al. The scope of health insurance coverage of vitiligo treatments in the United States: implications for health care outcomes and disparities in children of color [published online July 16, 2021]. Pediatr Dermatol. 2021; 38(suppl 2):79-85. doi:10.1111/pde.14714
- Opzelura prices, coupons, and patient assistance programs. Drugs.com. Accessed January 10, 2023. https://www.drugs.com /price-guide/opzelura#:~:text=Opzelura%20Prices%2C%20 Coupons%20and%20Patient,on%20the%20pharmacy%20you%20visit
- Bhutani T, Liao W. A practical approach to home UVB phototherapy for the treatment of generalized psoriasis. Pract Dermatol. 2010;7:31-35.
- Castro Porto Silva Lopes F, Ahmed A. Insurance coverage for phototherapy for vitiligo in comparison to psoriasis and atopic dermatitis. SKIN The Journal of Cutaneous Medicine. 2022;6:217-224. https://doi.org/10.25251/skin.6.3.6
- Smith MP, Ly K, Thibodeaux Q, et al. Home phototherapy for patients with vitiligo: challenges and solutions. Clin Cosmet Investig Dermatol. 2019;12:451-459. doi:10.2147/CCID.S185798
- Shrider EA, Kollar M, Chen F, et al. Income and poverty in the United States: 2020. US Census Bureau. September 14, 2021. Accessed December 27, 2022. https://www.census.gov/library/publications/2021/demo/p60-273.html
- Whitton ME, Pinart M, Batchelor J, et al. Interventions for vitiligo. Cochrane Database Syst Rev. 2010;(1):CD003263. doi:10.1002/14651858.CD003263.pub4
THE COMPARISON
A Vitiligo in a young Hispanic female, which spared the area under a ring. The patient has spotty return of pigment on the hand after narrowband UVB treatment.
B Vitiligo on the hand in a young Hispanic male.

Vitiligo is a chronic autoimmune disorder characterized by areas of depigmented white patches on the skin due to the loss of melanocytes in the epidermis. Various theories on the pathogenesis of vitiligo exist; however, autoimmune destruction of melanocytes remains the leading hypothesis, followed by intrinsic defects in melanocytes.1 Vitiligo is associated with various autoimmune diseases but is most frequently reported in conjunction with thyroid disorders.2
Epidemiology
Vitiligo affects approximately 1% of the US population and up to 8% worldwide.2 There is no difference in prevalence between races or genders. Females typically acquire the disease earlier than males. Onset may occur at any age, although about half of patients will have vitiligo by 20 years of age.1
Key clinical features in people with darker skin tones
Bright white patches are characteristic of vitiligo. The patches typically are asymptomatic and often affect the hands (Figures A and B), perioral skin, feet, and scalp, as well as areas more vulnerable to friction and trauma, such as the elbows and knees.2 Trichrome lesions—consisting of varying zones of white (depigmented), lighter brown (hypopigmented), and normal skin—are most commonly seen in individuals with darker skin. Trichrome vitiligo is considered an actively progressing variant of vitiligo.2
An important distinction when diagnosing vitiligo is evaluating for segmental vs nonsegmental vitiligo. Although nonsegmental vitiligo—the more common subtype—is characterized by symmetric distribution and a less predictable course, segmental vitiligo manifests in a localized and unilateral distribution, often avoiding extension past the midline. Segmental vitiligo typically manifests at a younger age and follows a more rapidly stabilizing course.3
Worth noting
Given that stark contrasts between pigmented and depigmented lesions are more prominent in darker skin tones, vitiligo can be more socially stigmatizing and psychologically devastating in these patients.4,5
Treatment of vitiligo includes narrowband UVB (NB-UVB) light phototherapy, excimer laser, topical corticosteroids, topical calcineurin inhibitors such as tacrolimus and pimecrolimus, and surgical melanocyte transplantation.1 In July 2022, ruxolitinib cream 1.5% was approved by the US Food and Drug Administration (FDA) for nonsegmental vitiligo in patients 12 years and older.6,7 It is the only FDA-approved therapy for vitiligo. It is thought to work by inhibiting the Janus kinase– signal transducers and activators of the transcription pathway.6 However, topical ruxolitinib is expensive, costing more than $2000 for 60 g.8
Health disparity highlight
A 2021 study reviewing the coverage policies of 15 commercial health care insurance companies, 50 BlueCross BlueShield plans, Medicaid, Medicare, and Veterans Affairs plans found inequities in the insurance coverage patterns for therapies used to treat vitiligo. There were 2 commonly cited reasons for denying coverage for therapies: vitiligo was considered cosmetic and therapies were not FDA approved.7 In comparison, NB-UVB light phototherapy for psoriasis is not considered cosmetic and has a much higher insurance coverage rate.9,10 The out-of-pocket cost for a patient to purchase their own NB-UVB light phototherapy is more than $5000.11 Not all patients of color are economically disadvantaged, but in the United States, Black and Hispanic populations experience disproportionately higher rates of poverty (19% and 17%, respectively) compared to their White counterparts (8%).12
Final thoughts
US Food and Drug Administration approval of new drugs or new treatment indications comes after years of research discovery and large-scale trials. This pursuit of new discovery, however, is uneven. Vitiligo has historically been understudied and underfunded for research; this is common among several conditions adversely affecting people of color in the United States.13
THE COMPARISON
A Vitiligo in a young Hispanic female, which spared the area under a ring. The patient has spotty return of pigment on the hand after narrowband UVB treatment.
B Vitiligo on the hand in a young Hispanic male.

Vitiligo is a chronic autoimmune disorder characterized by areas of depigmented white patches on the skin due to the loss of melanocytes in the epidermis. Various theories on the pathogenesis of vitiligo exist; however, autoimmune destruction of melanocytes remains the leading hypothesis, followed by intrinsic defects in melanocytes.1 Vitiligo is associated with various autoimmune diseases but is most frequently reported in conjunction with thyroid disorders.2
Epidemiology
Vitiligo affects approximately 1% of the US population and up to 8% worldwide.2 There is no difference in prevalence between races or genders. Females typically acquire the disease earlier than males. Onset may occur at any age, although about half of patients will have vitiligo by 20 years of age.1
Key clinical features in people with darker skin tones
Bright white patches are characteristic of vitiligo. The patches typically are asymptomatic and often affect the hands (Figures A and B), perioral skin, feet, and scalp, as well as areas more vulnerable to friction and trauma, such as the elbows and knees.2 Trichrome lesions—consisting of varying zones of white (depigmented), lighter brown (hypopigmented), and normal skin—are most commonly seen in individuals with darker skin. Trichrome vitiligo is considered an actively progressing variant of vitiligo.2
An important distinction when diagnosing vitiligo is evaluating for segmental vs nonsegmental vitiligo. Although nonsegmental vitiligo—the more common subtype—is characterized by symmetric distribution and a less predictable course, segmental vitiligo manifests in a localized and unilateral distribution, often avoiding extension past the midline. Segmental vitiligo typically manifests at a younger age and follows a more rapidly stabilizing course.3
Worth noting
Given that stark contrasts between pigmented and depigmented lesions are more prominent in darker skin tones, vitiligo can be more socially stigmatizing and psychologically devastating in these patients.4,5
Treatment of vitiligo includes narrowband UVB (NB-UVB) light phototherapy, excimer laser, topical corticosteroids, topical calcineurin inhibitors such as tacrolimus and pimecrolimus, and surgical melanocyte transplantation.1 In July 2022, ruxolitinib cream 1.5% was approved by the US Food and Drug Administration (FDA) for nonsegmental vitiligo in patients 12 years and older.6,7 It is the only FDA-approved therapy for vitiligo. It is thought to work by inhibiting the Janus kinase– signal transducers and activators of the transcription pathway.6 However, topical ruxolitinib is expensive, costing more than $2000 for 60 g.8
Health disparity highlight
A 2021 study reviewing the coverage policies of 15 commercial health care insurance companies, 50 BlueCross BlueShield plans, Medicaid, Medicare, and Veterans Affairs plans found inequities in the insurance coverage patterns for therapies used to treat vitiligo. There were 2 commonly cited reasons for denying coverage for therapies: vitiligo was considered cosmetic and therapies were not FDA approved.7 In comparison, NB-UVB light phototherapy for psoriasis is not considered cosmetic and has a much higher insurance coverage rate.9,10 The out-of-pocket cost for a patient to purchase their own NB-UVB light phototherapy is more than $5000.11 Not all patients of color are economically disadvantaged, but in the United States, Black and Hispanic populations experience disproportionately higher rates of poverty (19% and 17%, respectively) compared to their White counterparts (8%).12
Final thoughts
US Food and Drug Administration approval of new drugs or new treatment indications comes after years of research discovery and large-scale trials. This pursuit of new discovery, however, is uneven. Vitiligo has historically been understudied and underfunded for research; this is common among several conditions adversely affecting people of color in the United States.13
- Rashighi M, Harris JE. Vitiligo pathogenesis and emerging treatments. Dermatol Clin. 2017;35:257-265. doi:10.1016/j.det.2016.11.014
- Alikhan A, Felsten LM, Daly M, et al. Vitiligo: a comprehensive overview part I. introduction, epidemiology, quality of life, diagnosis, differential diagnosis, associations, histopathology, etiology, and work-up. J Am Acad Dermatol. 2011;65:473-491. doi:10.1016/j.jaad.2010.11.061
- van Geel N, Speeckaert R. Segmental vitiligo. Dermatol Clin. 2017; 35:145-150. doi:10.1016/j.det.2016.11.005
- Grimes PE, Miller MM. Vitiligo: patient stories, self-esteem, and the psychological burden of disease. Int J Womens Dermatol. 2018;4:32-37. doi:10.1016/j.ijwd.2017.11.005
- Ezzedine K, Eleftheriadou V, Jones H, et al. Psychosocial effects of vitiligo: a systematic literature review [published online September 23, 2021]. Am J Clin Dermatol. 2021;22:757-774. doi:10.1007/s40257 -021-00631-6
- FDA approves topical treatment addressing repigmentation in vitiligo in patients aged 12 and older. News release. US Food and Drug Administration; July 19, 2022. Accessed December 27, 2022. https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-topical-treatment-addressing-repigmentation-vitiligo-patients -aged-12-and-older
- Blundell A, Sachar M, Gabel CK, et al. The scope of health insurance coverage of vitiligo treatments in the United States: implications for health care outcomes and disparities in children of color [published online July 16, 2021]. Pediatr Dermatol. 2021; 38(suppl 2):79-85. doi:10.1111/pde.14714
- Opzelura prices, coupons, and patient assistance programs. Drugs.com. Accessed January 10, 2023. https://www.drugs.com /price-guide/opzelura#:~:text=Opzelura%20Prices%2C%20 Coupons%20and%20Patient,on%20the%20pharmacy%20you%20visit
- Bhutani T, Liao W. A practical approach to home UVB phototherapy for the treatment of generalized psoriasis. Pract Dermatol. 2010;7:31-35.
- Castro Porto Silva Lopes F, Ahmed A. Insurance coverage for phototherapy for vitiligo in comparison to psoriasis and atopic dermatitis. SKIN The Journal of Cutaneous Medicine. 2022;6:217-224. https://doi.org/10.25251/skin.6.3.6
- Smith MP, Ly K, Thibodeaux Q, et al. Home phototherapy for patients with vitiligo: challenges and solutions. Clin Cosmet Investig Dermatol. 2019;12:451-459. doi:10.2147/CCID.S185798
- Shrider EA, Kollar M, Chen F, et al. Income and poverty in the United States: 2020. US Census Bureau. September 14, 2021. Accessed December 27, 2022. https://www.census.gov/library/publications/2021/demo/p60-273.html
- Whitton ME, Pinart M, Batchelor J, et al. Interventions for vitiligo. Cochrane Database Syst Rev. 2010;(1):CD003263. doi:10.1002/14651858.CD003263.pub4
- Rashighi M, Harris JE. Vitiligo pathogenesis and emerging treatments. Dermatol Clin. 2017;35:257-265. doi:10.1016/j.det.2016.11.014
- Alikhan A, Felsten LM, Daly M, et al. Vitiligo: a comprehensive overview part I. introduction, epidemiology, quality of life, diagnosis, differential diagnosis, associations, histopathology, etiology, and work-up. J Am Acad Dermatol. 2011;65:473-491. doi:10.1016/j.jaad.2010.11.061
- van Geel N, Speeckaert R. Segmental vitiligo. Dermatol Clin. 2017; 35:145-150. doi:10.1016/j.det.2016.11.005
- Grimes PE, Miller MM. Vitiligo: patient stories, self-esteem, and the psychological burden of disease. Int J Womens Dermatol. 2018;4:32-37. doi:10.1016/j.ijwd.2017.11.005
- Ezzedine K, Eleftheriadou V, Jones H, et al. Psychosocial effects of vitiligo: a systematic literature review [published online September 23, 2021]. Am J Clin Dermatol. 2021;22:757-774. doi:10.1007/s40257 -021-00631-6
- FDA approves topical treatment addressing repigmentation in vitiligo in patients aged 12 and older. News release. US Food and Drug Administration; July 19, 2022. Accessed December 27, 2022. https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-topical-treatment-addressing-repigmentation-vitiligo-patients -aged-12-and-older
- Blundell A, Sachar M, Gabel CK, et al. The scope of health insurance coverage of vitiligo treatments in the United States: implications for health care outcomes and disparities in children of color [published online July 16, 2021]. Pediatr Dermatol. 2021; 38(suppl 2):79-85. doi:10.1111/pde.14714
- Opzelura prices, coupons, and patient assistance programs. Drugs.com. Accessed January 10, 2023. https://www.drugs.com /price-guide/opzelura#:~:text=Opzelura%20Prices%2C%20 Coupons%20and%20Patient,on%20the%20pharmacy%20you%20visit
- Bhutani T, Liao W. A practical approach to home UVB phototherapy for the treatment of generalized psoriasis. Pract Dermatol. 2010;7:31-35.
- Castro Porto Silva Lopes F, Ahmed A. Insurance coverage for phototherapy for vitiligo in comparison to psoriasis and atopic dermatitis. SKIN The Journal of Cutaneous Medicine. 2022;6:217-224. https://doi.org/10.25251/skin.6.3.6
- Smith MP, Ly K, Thibodeaux Q, et al. Home phototherapy for patients with vitiligo: challenges and solutions. Clin Cosmet Investig Dermatol. 2019;12:451-459. doi:10.2147/CCID.S185798
- Shrider EA, Kollar M, Chen F, et al. Income and poverty in the United States: 2020. US Census Bureau. September 14, 2021. Accessed December 27, 2022. https://www.census.gov/library/publications/2021/demo/p60-273.html
- Whitton ME, Pinart M, Batchelor J, et al. Interventions for vitiligo. Cochrane Database Syst Rev. 2010;(1):CD003263. doi:10.1002/14651858.CD003263.pub4
Camellia japonica
The various Camellia species originated in Eastern Asia and are believed to have been introduced in northwestern Spain in the 18th century. Camellia japonica, a flowering evergreen tree with various medical and cosmetic applications, is found throughout Galicia, Spain, where it is cultivated as an ornamental plant, and is native to Japan, South Korea, and China.1-4 The flowers and seeds of C. japonica have been used in traditional medicine and cosmetics in East Asia, with the oil of C. japonica used there to restore skin elasticity and to enhance skin health.4-6
While the use of C. sinensis in traditional and modern medicine is much better researched, understood, and characterized, C. japonica is now being considered for various health benefits. This column will focus on the bioactivity and scientific support for dermatologic applications of C. japonica. It is worth noting that a dry oil known as tsubaki oil, derived from C. japonica and rich in oleic acid, polyphenols, as well as vitamins A, C, D, and E, is used for skin and hair care in moisturizers produced primarily in Japan.
Antioxidant activity
In 2005, Lee and colleagues determined that C. japonica leaf and flower extracts display antioxidant, antifungal, and antibacterial activities (with the latter showing greater gram-positive than gram-negative activity).8 Investigating the antioxidant characteristics of the ethanol extract of the C. japonica flower in 2011, Piao and colleagues reported that the botanical exerted scavenging activity against reactive oxygen species in human HaCaT keratinocytes and enhanced protein expression and function of the antioxidant enzymes superoxide dismutase, catalase, and glutathione peroxidase.9
Less than a decade later, Yoon and colleagues determined that C. japonica leaf extract contains high concentrations of vitamin E and rutin as well as other active constituents and that it exhibits antioxidant and antihyperuricemic activity in vitro and in vivo.4
Since then, Kim and colleagues have demonstrated, using cultured normal human dermal fibroblasts, that C. japonica flower extract effectively hindered urban air pollutants–induced reactive oxygen species synthesis. In ex vivo results, the investigators showed that the botanical agent suppressed matrix metalloproteinase (MMP)-1 expression, fostered collagen production, and decreased levels of pollutants-induced malondialdehyde. The authors concluded that C. japonica flower extract shows promise as a protective agent against pollutant-induced cutaneous damage.10
Anti-inflammatory and wound-healing activity
In 2012, Kim and colleagues found that C. japonica oil imparts anti-inflammatory activity via down-regulation of iNOS and COX-2 gene expression by suppressing of NF-KB and AP-1 signaling.6
Jeon and colleagues determined, in a 2018 investigation of 3,695 native plant extracts, that extracts from C. japonica fruit and stems improved induced pluripotent stem cell (iPSC) generation in mouse and human skin and enhanced wound healing in an in vivo mouse wound model. They suggested that their findings may point toward more effective approaches to developing clinical-grade iPSCs and wound-healing therapies.11
Cosmeceutical potential
Among the important bioactive ingredients present in C. japonica are phenolic compounds, terpenoids, and fatty acids, which are thought to account for the anti-inflammatory, antioxidant, antimicrobial, and anticancer activity associated with the plant.1 The high concentration of polyphenolic substances, in particular, is thought to at least partly account for the inclusion of C. japonica leaf extracts in antiaging cosmetics and cosmeceuticals.12 Specifically, some of the antioxidant substances found in C. japonica extracts include quercetin, quercetin-3-O-glucoside, quercitrin, and kaempferol.9
Wrinkle reduction and moisturization
In 2007, Jung and colleagues found that C. japonica oil activated collagen 1A2 promotion in human dermal fibroblast cells in a concentration-dependent fashion. The oil also suppressed MMP-1 functions and spurred the production of human type I procollagen. On human skin, C. japonica oil was tested on the upper back of 30 volunteers and failed to provoke any adverse reactions. The oil also diminished transepidermal water loss on the forearm. The researchers concluded that C. japonica oil merits consideration as an antiwrinkle ingredient in topical formulations.13
More recently, Choi and colleagues showed that ceramide nanoparticles developed through the use of natural oils derived from Korean traditional plants (including C. japonica, along with Panax ginseng, C. sinensis, Glycine max napjakong, and Glycine max seoritae) improve skin carrier functions and promote gene expressions needed for epidermal homeostasis. The expressions of the FLG, CASP14, and INV genes were notably enhanced by the tested formulation. The researchers observed from in vivo human studies that the application of the ceramide nanoparticles yielded more rapid recovery in impaired skin barriers than the control formulation. Amelioration of stratum corneum cohesion was also noted. The investigators concluded that this and other natural oil–derived ceramide nanoparticle formulations may represent the potential for developing better moisturizers for enhancing skin barrier function.14
Hair-growth promotion and skin-whitening activity
Early in 2021, Cho and colleagues demonstrated that C. japonica phytoplacenta extract spurred the up-regulation of the expression of hair growth–marker genes in human follicle dermal papilla cells in vitro. In clinical tests with 42 adult female volunteers, a solution with 0.5% C. japonica placenta extract raised moisture content of the scalp and reduced sebum levels, dead scalp keratin, and redness. The researchers concluded that C. japonica phytoplacenta extract displays promise as a scalp treatment and hair growth–promoting agent.2
Later that year, Ha and colleagues reported on their findings regarding the tyrosinase inhibitory activity of the essential oil of C. japonica seeds. They identified hexamethylcyclotrisiloxane (42.36%) and octamethylcyclotetrasiloxane (23.28%) as the main constituents of the oil, which demonstrated comparable inhibitory activity to arbutin (positive control) against mushroom tyrosinase. Melanogenesis was also significantly suppressed by C. japonica seed essential oil in B16F10 melanoma cells. The investigators concluded that the essential oil of C. japonica seeds exhibits robust antityrosinase activity and, therefore, warrants consideration as a skin-whitening agent.15
Conclusion
C. japonica is not as popular or well researched as another Camellia species, C. sinensis (the primary tea plant consumed globally and highly touted and appreciated for its multitude of health benefits), but it has its own history of traditional uses for medical and cosmetic purposes and is a subject of increasing research interest along with popular applications. Its antioxidant and anti-inflammatory properties are thought to be central in conferring the ability to protect the skin from aging. Its effects on the skin barrier help skin hydration. More research is necessary to elucidate the apparently widespread potential of this botanical agent that is already found in some over-the-counter products.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions, a SaaS company used to generate skin care routines in office and as an ecommerce solution. Write to her at dermnews@mdedge.com.
References
1. Pereira AG et al. Food Chem X. 2022 Feb 17;13:100258.
2. Cho WK et al. FEBS Open Bio. 2021 Mar;11(3):633-51.
3. Chung MY et al. Evolution. 2003 Jan;57(1):62-73.
4. Yoon IS et al. Int J Mol Med. 2017 Jun;39(6):1613-20.
5. Lee HH et al. Evid Based Complement Alternat Med. 2016;2016:9679867.
6. Kim S et al. BMB Rep. 2012 Mar;45(3):177-82.
7. Majumder S et al. Bull Nat Res Cen. 2020 Dec;44(1):1-4.
8. Lee SY et al. Korean Journal of Medicinal Crop Science. 2005;13(3):93-100.
9. Piao MJ et al. Int J Mol Sci. 2011;12(4):2618-30.
10. Kim M et al. BMC Complement Altern Med. 2019 Jan 28;19(1):30.
11. Jeon H et al. J Clin Med. 2018 Nov 20;7(11):449.
12. Mizutani T, Masaki H. Exp Dermatol. 2014 Oct;23 Suppl 1:23-6.
13. Jung E et al. J Ethnopharmacol. 2007 May 30;112(1):127-31.
14. Choi HK et al. J Cosmet Dermatol. 2022 Oct;21(10):4931-41.
15. Ha SY et al. Evid Based Complement Alternat Med. 2021 Nov 16;2021:6328767.
The various Camellia species originated in Eastern Asia and are believed to have been introduced in northwestern Spain in the 18th century. Camellia japonica, a flowering evergreen tree with various medical and cosmetic applications, is found throughout Galicia, Spain, where it is cultivated as an ornamental plant, and is native to Japan, South Korea, and China.1-4 The flowers and seeds of C. japonica have been used in traditional medicine and cosmetics in East Asia, with the oil of C. japonica used there to restore skin elasticity and to enhance skin health.4-6
While the use of C. sinensis in traditional and modern medicine is much better researched, understood, and characterized, C. japonica is now being considered for various health benefits. This column will focus on the bioactivity and scientific support for dermatologic applications of C. japonica. It is worth noting that a dry oil known as tsubaki oil, derived from C. japonica and rich in oleic acid, polyphenols, as well as vitamins A, C, D, and E, is used for skin and hair care in moisturizers produced primarily in Japan.
Antioxidant activity
In 2005, Lee and colleagues determined that C. japonica leaf and flower extracts display antioxidant, antifungal, and antibacterial activities (with the latter showing greater gram-positive than gram-negative activity).8 Investigating the antioxidant characteristics of the ethanol extract of the C. japonica flower in 2011, Piao and colleagues reported that the botanical exerted scavenging activity against reactive oxygen species in human HaCaT keratinocytes and enhanced protein expression and function of the antioxidant enzymes superoxide dismutase, catalase, and glutathione peroxidase.9
Less than a decade later, Yoon and colleagues determined that C. japonica leaf extract contains high concentrations of vitamin E and rutin as well as other active constituents and that it exhibits antioxidant and antihyperuricemic activity in vitro and in vivo.4
Since then, Kim and colleagues have demonstrated, using cultured normal human dermal fibroblasts, that C. japonica flower extract effectively hindered urban air pollutants–induced reactive oxygen species synthesis. In ex vivo results, the investigators showed that the botanical agent suppressed matrix metalloproteinase (MMP)-1 expression, fostered collagen production, and decreased levels of pollutants-induced malondialdehyde. The authors concluded that C. japonica flower extract shows promise as a protective agent against pollutant-induced cutaneous damage.10
Anti-inflammatory and wound-healing activity
In 2012, Kim and colleagues found that C. japonica oil imparts anti-inflammatory activity via down-regulation of iNOS and COX-2 gene expression by suppressing of NF-KB and AP-1 signaling.6
Jeon and colleagues determined, in a 2018 investigation of 3,695 native plant extracts, that extracts from C. japonica fruit and stems improved induced pluripotent stem cell (iPSC) generation in mouse and human skin and enhanced wound healing in an in vivo mouse wound model. They suggested that their findings may point toward more effective approaches to developing clinical-grade iPSCs and wound-healing therapies.11
Cosmeceutical potential
Among the important bioactive ingredients present in C. japonica are phenolic compounds, terpenoids, and fatty acids, which are thought to account for the anti-inflammatory, antioxidant, antimicrobial, and anticancer activity associated with the plant.1 The high concentration of polyphenolic substances, in particular, is thought to at least partly account for the inclusion of C. japonica leaf extracts in antiaging cosmetics and cosmeceuticals.12 Specifically, some of the antioxidant substances found in C. japonica extracts include quercetin, quercetin-3-O-glucoside, quercitrin, and kaempferol.9
Wrinkle reduction and moisturization
In 2007, Jung and colleagues found that C. japonica oil activated collagen 1A2 promotion in human dermal fibroblast cells in a concentration-dependent fashion. The oil also suppressed MMP-1 functions and spurred the production of human type I procollagen. On human skin, C. japonica oil was tested on the upper back of 30 volunteers and failed to provoke any adverse reactions. The oil also diminished transepidermal water loss on the forearm. The researchers concluded that C. japonica oil merits consideration as an antiwrinkle ingredient in topical formulations.13
More recently, Choi and colleagues showed that ceramide nanoparticles developed through the use of natural oils derived from Korean traditional plants (including C. japonica, along with Panax ginseng, C. sinensis, Glycine max napjakong, and Glycine max seoritae) improve skin carrier functions and promote gene expressions needed for epidermal homeostasis. The expressions of the FLG, CASP14, and INV genes were notably enhanced by the tested formulation. The researchers observed from in vivo human studies that the application of the ceramide nanoparticles yielded more rapid recovery in impaired skin barriers than the control formulation. Amelioration of stratum corneum cohesion was also noted. The investigators concluded that this and other natural oil–derived ceramide nanoparticle formulations may represent the potential for developing better moisturizers for enhancing skin barrier function.14
Hair-growth promotion and skin-whitening activity
Early in 2021, Cho and colleagues demonstrated that C. japonica phytoplacenta extract spurred the up-regulation of the expression of hair growth–marker genes in human follicle dermal papilla cells in vitro. In clinical tests with 42 adult female volunteers, a solution with 0.5% C. japonica placenta extract raised moisture content of the scalp and reduced sebum levels, dead scalp keratin, and redness. The researchers concluded that C. japonica phytoplacenta extract displays promise as a scalp treatment and hair growth–promoting agent.2
Later that year, Ha and colleagues reported on their findings regarding the tyrosinase inhibitory activity of the essential oil of C. japonica seeds. They identified hexamethylcyclotrisiloxane (42.36%) and octamethylcyclotetrasiloxane (23.28%) as the main constituents of the oil, which demonstrated comparable inhibitory activity to arbutin (positive control) against mushroom tyrosinase. Melanogenesis was also significantly suppressed by C. japonica seed essential oil in B16F10 melanoma cells. The investigators concluded that the essential oil of C. japonica seeds exhibits robust antityrosinase activity and, therefore, warrants consideration as a skin-whitening agent.15
Conclusion
C. japonica is not as popular or well researched as another Camellia species, C. sinensis (the primary tea plant consumed globally and highly touted and appreciated for its multitude of health benefits), but it has its own history of traditional uses for medical and cosmetic purposes and is a subject of increasing research interest along with popular applications. Its antioxidant and anti-inflammatory properties are thought to be central in conferring the ability to protect the skin from aging. Its effects on the skin barrier help skin hydration. More research is necessary to elucidate the apparently widespread potential of this botanical agent that is already found in some over-the-counter products.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions, a SaaS company used to generate skin care routines in office and as an ecommerce solution. Write to her at dermnews@mdedge.com.
References
1. Pereira AG et al. Food Chem X. 2022 Feb 17;13:100258.
2. Cho WK et al. FEBS Open Bio. 2021 Mar;11(3):633-51.
3. Chung MY et al. Evolution. 2003 Jan;57(1):62-73.
4. Yoon IS et al. Int J Mol Med. 2017 Jun;39(6):1613-20.
5. Lee HH et al. Evid Based Complement Alternat Med. 2016;2016:9679867.
6. Kim S et al. BMB Rep. 2012 Mar;45(3):177-82.
7. Majumder S et al. Bull Nat Res Cen. 2020 Dec;44(1):1-4.
8. Lee SY et al. Korean Journal of Medicinal Crop Science. 2005;13(3):93-100.
9. Piao MJ et al. Int J Mol Sci. 2011;12(4):2618-30.
10. Kim M et al. BMC Complement Altern Med. 2019 Jan 28;19(1):30.
11. Jeon H et al. J Clin Med. 2018 Nov 20;7(11):449.
12. Mizutani T, Masaki H. Exp Dermatol. 2014 Oct;23 Suppl 1:23-6.
13. Jung E et al. J Ethnopharmacol. 2007 May 30;112(1):127-31.
14. Choi HK et al. J Cosmet Dermatol. 2022 Oct;21(10):4931-41.
15. Ha SY et al. Evid Based Complement Alternat Med. 2021 Nov 16;2021:6328767.
The various Camellia species originated in Eastern Asia and are believed to have been introduced in northwestern Spain in the 18th century. Camellia japonica, a flowering evergreen tree with various medical and cosmetic applications, is found throughout Galicia, Spain, where it is cultivated as an ornamental plant, and is native to Japan, South Korea, and China.1-4 The flowers and seeds of C. japonica have been used in traditional medicine and cosmetics in East Asia, with the oil of C. japonica used there to restore skin elasticity and to enhance skin health.4-6
While the use of C. sinensis in traditional and modern medicine is much better researched, understood, and characterized, C. japonica is now being considered for various health benefits. This column will focus on the bioactivity and scientific support for dermatologic applications of C. japonica. It is worth noting that a dry oil known as tsubaki oil, derived from C. japonica and rich in oleic acid, polyphenols, as well as vitamins A, C, D, and E, is used for skin and hair care in moisturizers produced primarily in Japan.
Antioxidant activity
In 2005, Lee and colleagues determined that C. japonica leaf and flower extracts display antioxidant, antifungal, and antibacterial activities (with the latter showing greater gram-positive than gram-negative activity).8 Investigating the antioxidant characteristics of the ethanol extract of the C. japonica flower in 2011, Piao and colleagues reported that the botanical exerted scavenging activity against reactive oxygen species in human HaCaT keratinocytes and enhanced protein expression and function of the antioxidant enzymes superoxide dismutase, catalase, and glutathione peroxidase.9
Less than a decade later, Yoon and colleagues determined that C. japonica leaf extract contains high concentrations of vitamin E and rutin as well as other active constituents and that it exhibits antioxidant and antihyperuricemic activity in vitro and in vivo.4
Since then, Kim and colleagues have demonstrated, using cultured normal human dermal fibroblasts, that C. japonica flower extract effectively hindered urban air pollutants–induced reactive oxygen species synthesis. In ex vivo results, the investigators showed that the botanical agent suppressed matrix metalloproteinase (MMP)-1 expression, fostered collagen production, and decreased levels of pollutants-induced malondialdehyde. The authors concluded that C. japonica flower extract shows promise as a protective agent against pollutant-induced cutaneous damage.10
Anti-inflammatory and wound-healing activity
In 2012, Kim and colleagues found that C. japonica oil imparts anti-inflammatory activity via down-regulation of iNOS and COX-2 gene expression by suppressing of NF-KB and AP-1 signaling.6
Jeon and colleagues determined, in a 2018 investigation of 3,695 native plant extracts, that extracts from C. japonica fruit and stems improved induced pluripotent stem cell (iPSC) generation in mouse and human skin and enhanced wound healing in an in vivo mouse wound model. They suggested that their findings may point toward more effective approaches to developing clinical-grade iPSCs and wound-healing therapies.11
Cosmeceutical potential
Among the important bioactive ingredients present in C. japonica are phenolic compounds, terpenoids, and fatty acids, which are thought to account for the anti-inflammatory, antioxidant, antimicrobial, and anticancer activity associated with the plant.1 The high concentration of polyphenolic substances, in particular, is thought to at least partly account for the inclusion of C. japonica leaf extracts in antiaging cosmetics and cosmeceuticals.12 Specifically, some of the antioxidant substances found in C. japonica extracts include quercetin, quercetin-3-O-glucoside, quercitrin, and kaempferol.9
Wrinkle reduction and moisturization
In 2007, Jung and colleagues found that C. japonica oil activated collagen 1A2 promotion in human dermal fibroblast cells in a concentration-dependent fashion. The oil also suppressed MMP-1 functions and spurred the production of human type I procollagen. On human skin, C. japonica oil was tested on the upper back of 30 volunteers and failed to provoke any adverse reactions. The oil also diminished transepidermal water loss on the forearm. The researchers concluded that C. japonica oil merits consideration as an antiwrinkle ingredient in topical formulations.13
More recently, Choi and colleagues showed that ceramide nanoparticles developed through the use of natural oils derived from Korean traditional plants (including C. japonica, along with Panax ginseng, C. sinensis, Glycine max napjakong, and Glycine max seoritae) improve skin carrier functions and promote gene expressions needed for epidermal homeostasis. The expressions of the FLG, CASP14, and INV genes were notably enhanced by the tested formulation. The researchers observed from in vivo human studies that the application of the ceramide nanoparticles yielded more rapid recovery in impaired skin barriers than the control formulation. Amelioration of stratum corneum cohesion was also noted. The investigators concluded that this and other natural oil–derived ceramide nanoparticle formulations may represent the potential for developing better moisturizers for enhancing skin barrier function.14
Hair-growth promotion and skin-whitening activity
Early in 2021, Cho and colleagues demonstrated that C. japonica phytoplacenta extract spurred the up-regulation of the expression of hair growth–marker genes in human follicle dermal papilla cells in vitro. In clinical tests with 42 adult female volunteers, a solution with 0.5% C. japonica placenta extract raised moisture content of the scalp and reduced sebum levels, dead scalp keratin, and redness. The researchers concluded that C. japonica phytoplacenta extract displays promise as a scalp treatment and hair growth–promoting agent.2
Later that year, Ha and colleagues reported on their findings regarding the tyrosinase inhibitory activity of the essential oil of C. japonica seeds. They identified hexamethylcyclotrisiloxane (42.36%) and octamethylcyclotetrasiloxane (23.28%) as the main constituents of the oil, which demonstrated comparable inhibitory activity to arbutin (positive control) against mushroom tyrosinase. Melanogenesis was also significantly suppressed by C. japonica seed essential oil in B16F10 melanoma cells. The investigators concluded that the essential oil of C. japonica seeds exhibits robust antityrosinase activity and, therefore, warrants consideration as a skin-whitening agent.15
Conclusion
C. japonica is not as popular or well researched as another Camellia species, C. sinensis (the primary tea plant consumed globally and highly touted and appreciated for its multitude of health benefits), but it has its own history of traditional uses for medical and cosmetic purposes and is a subject of increasing research interest along with popular applications. Its antioxidant and anti-inflammatory properties are thought to be central in conferring the ability to protect the skin from aging. Its effects on the skin barrier help skin hydration. More research is necessary to elucidate the apparently widespread potential of this botanical agent that is already found in some over-the-counter products.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions, a SaaS company used to generate skin care routines in office and as an ecommerce solution. Write to her at dermnews@mdedge.com.
References
1. Pereira AG et al. Food Chem X. 2022 Feb 17;13:100258.
2. Cho WK et al. FEBS Open Bio. 2021 Mar;11(3):633-51.
3. Chung MY et al. Evolution. 2003 Jan;57(1):62-73.
4. Yoon IS et al. Int J Mol Med. 2017 Jun;39(6):1613-20.
5. Lee HH et al. Evid Based Complement Alternat Med. 2016;2016:9679867.
6. Kim S et al. BMB Rep. 2012 Mar;45(3):177-82.
7. Majumder S et al. Bull Nat Res Cen. 2020 Dec;44(1):1-4.
8. Lee SY et al. Korean Journal of Medicinal Crop Science. 2005;13(3):93-100.
9. Piao MJ et al. Int J Mol Sci. 2011;12(4):2618-30.
10. Kim M et al. BMC Complement Altern Med. 2019 Jan 28;19(1):30.
11. Jeon H et al. J Clin Med. 2018 Nov 20;7(11):449.
12. Mizutani T, Masaki H. Exp Dermatol. 2014 Oct;23 Suppl 1:23-6.
13. Jung E et al. J Ethnopharmacol. 2007 May 30;112(1):127-31.
14. Choi HK et al. J Cosmet Dermatol. 2022 Oct;21(10):4931-41.
15. Ha SY et al. Evid Based Complement Alternat Med. 2021 Nov 16;2021:6328767.
Pigmentary disorder experts welcome research explosion
ORLANDO – , a panel of experts said in a session on this topic at the ODAC Dermatology, Aesthetic & Surgical Conference.
The arrival of ruxolitinib cream, a topical JAK inhibitor – and oral JAK inhibitors, including ritlecitinib, a JAK3/TEC (tyrosine kinase expressed in hepatocellular carcinoma) inhibitor in clinical trials – is a welcome development for treatment of vitiligo, said John E. Harris, MD, PhD, chair of dermatology and director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester. Also in the pipeline is a kit for melanocyte-keratinocyte transplantation, which involves transplanting epidermal cells from one part of the body to another. This can be a challenging procedure but a kit would make it easier for a wider range of practitioners. (Topical ruxolitinib was approved by the Food and Drug Administration for treating nonsegmental vitiligo in July, 2022.)
“In the last 10 years, it’s just blown up and people care about vitiligo now,” Dr. Harris said, noting that vitiligo is more than a cosmetic issue, like gray hair or wrinkles. “Vitiligo is an autoimmune disease and now is being treated as such.”
Nada Elbuluk, MD, MSc, associate professor of dermatology at the University of Southern California, Los Angeles, said she’s pleased at the increasing availability of treatment options for hyperpigmentation, aside from hydroquinone, which is associated with an increased risk of adverse effects.
“We have more and more nonhydroquinone agents ... which is really nice because it expands our treatment armamentarium and what we can use to cycle people off of hydroquinone,” she said.
Some of these options include tranexamic acid and products containing azelaic acid or vitamin C.
Iltefat H. Hamzavi, MD, senior staff physician at Henry Ford Health System, Detroit, said that pigmentary disorders are receiving the recognition they deserve.
“I’m excited just about the intersection of society and science, the awareness that pigmentary abnormalities mean something, and they mean something across our society,” he said.
Dr. Elbuluk said that hyperpigmentation has “profound effects on quality of life” for patients.
“They are often more bothered by the darkening of the skin than the primary process that caused it,” she said. “It’s not uncommon that the chief complaint will say ‘dark spots’ and I walk in a room and it’s a patient who has acne. They don’t even say they’re here for acne. They just put ‘dark spots’ [down] because that’s what bothers them. That’s what lasts for so long after the acne is gone.”
The experts offered suggestions for managing these cases. Among her tips, Dr. Elbuluk said that for hyperpigmentation, physicians should not be afraid to biopsy the face – but suggested small, 2-millimeter specimens. In addition, “you can get common conditions in uncommon places,” she noted. “If you see something that looks like melasma off the face, it actually could be, so keep that in your differential.”
Dr. Hamzavi, who spoke about hypopigmentation disorders, said clinicians need to use an algorithm for diagnosis, considering features such as localized or diffuse, scale or no scale, as well as patient history, and other factors. For instance, a hypopigmented area that is localized and has a reddish central papule might lead a clinician to a diagnosis of hypopigmented sarcoidosis.
Using the algorithms, “you actually have to categorize these and then use your own experience. ... All of these elements can help you become a really good taxonomist – ultimately that’s what physicians are.”
He said it’s also important to know when it’s time to reconsider a diagnosis, such as when patients do not respond to traditional treatments. “If they don’t respond, re-categorize,” he said.
Speaking about vitiligo, Dr. Harris said it’s crucial to differentiate active vitiligo from inactive vitiligo and if it’s active, steps need to be taken to keep it from worsening..
Four signs of active vitiligo are a “confetti” pattern of clustered tiny macules of depigmentation, which will coalesce quickly into huge patches; tri-chrome vitiligo that includes a hypopigmented zone; linear areas of depigmentation (Koebner’s phenomenon) that look like scratches on the skin; and inflammatory vitiligo, with an erythematous ring around the edges of a depigmented area.
Dr. Harris disclosed ties with Incyte, Pfizer, Abbvie, Genzyme/Sanofi and other companies. Dr. Elbuluk disclosed ties with Avita, Incyte, Beiersdorf, and other companies. Dr. Hamzavi disclosed ties with AbbVie, Pfizer, Incyte, and other companies.
ORLANDO – , a panel of experts said in a session on this topic at the ODAC Dermatology, Aesthetic & Surgical Conference.
The arrival of ruxolitinib cream, a topical JAK inhibitor – and oral JAK inhibitors, including ritlecitinib, a JAK3/TEC (tyrosine kinase expressed in hepatocellular carcinoma) inhibitor in clinical trials – is a welcome development for treatment of vitiligo, said John E. Harris, MD, PhD, chair of dermatology and director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester. Also in the pipeline is a kit for melanocyte-keratinocyte transplantation, which involves transplanting epidermal cells from one part of the body to another. This can be a challenging procedure but a kit would make it easier for a wider range of practitioners. (Topical ruxolitinib was approved by the Food and Drug Administration for treating nonsegmental vitiligo in July, 2022.)
“In the last 10 years, it’s just blown up and people care about vitiligo now,” Dr. Harris said, noting that vitiligo is more than a cosmetic issue, like gray hair or wrinkles. “Vitiligo is an autoimmune disease and now is being treated as such.”
Nada Elbuluk, MD, MSc, associate professor of dermatology at the University of Southern California, Los Angeles, said she’s pleased at the increasing availability of treatment options for hyperpigmentation, aside from hydroquinone, which is associated with an increased risk of adverse effects.
“We have more and more nonhydroquinone agents ... which is really nice because it expands our treatment armamentarium and what we can use to cycle people off of hydroquinone,” she said.
Some of these options include tranexamic acid and products containing azelaic acid or vitamin C.
Iltefat H. Hamzavi, MD, senior staff physician at Henry Ford Health System, Detroit, said that pigmentary disorders are receiving the recognition they deserve.
“I’m excited just about the intersection of society and science, the awareness that pigmentary abnormalities mean something, and they mean something across our society,” he said.
Dr. Elbuluk said that hyperpigmentation has “profound effects on quality of life” for patients.
“They are often more bothered by the darkening of the skin than the primary process that caused it,” she said. “It’s not uncommon that the chief complaint will say ‘dark spots’ and I walk in a room and it’s a patient who has acne. They don’t even say they’re here for acne. They just put ‘dark spots’ [down] because that’s what bothers them. That’s what lasts for so long after the acne is gone.”
The experts offered suggestions for managing these cases. Among her tips, Dr. Elbuluk said that for hyperpigmentation, physicians should not be afraid to biopsy the face – but suggested small, 2-millimeter specimens. In addition, “you can get common conditions in uncommon places,” she noted. “If you see something that looks like melasma off the face, it actually could be, so keep that in your differential.”
Dr. Hamzavi, who spoke about hypopigmentation disorders, said clinicians need to use an algorithm for diagnosis, considering features such as localized or diffuse, scale or no scale, as well as patient history, and other factors. For instance, a hypopigmented area that is localized and has a reddish central papule might lead a clinician to a diagnosis of hypopigmented sarcoidosis.
Using the algorithms, “you actually have to categorize these and then use your own experience. ... All of these elements can help you become a really good taxonomist – ultimately that’s what physicians are.”
He said it’s also important to know when it’s time to reconsider a diagnosis, such as when patients do not respond to traditional treatments. “If they don’t respond, re-categorize,” he said.
Speaking about vitiligo, Dr. Harris said it’s crucial to differentiate active vitiligo from inactive vitiligo and if it’s active, steps need to be taken to keep it from worsening..
Four signs of active vitiligo are a “confetti” pattern of clustered tiny macules of depigmentation, which will coalesce quickly into huge patches; tri-chrome vitiligo that includes a hypopigmented zone; linear areas of depigmentation (Koebner’s phenomenon) that look like scratches on the skin; and inflammatory vitiligo, with an erythematous ring around the edges of a depigmented area.
Dr. Harris disclosed ties with Incyte, Pfizer, Abbvie, Genzyme/Sanofi and other companies. Dr. Elbuluk disclosed ties with Avita, Incyte, Beiersdorf, and other companies. Dr. Hamzavi disclosed ties with AbbVie, Pfizer, Incyte, and other companies.
ORLANDO – , a panel of experts said in a session on this topic at the ODAC Dermatology, Aesthetic & Surgical Conference.
The arrival of ruxolitinib cream, a topical JAK inhibitor – and oral JAK inhibitors, including ritlecitinib, a JAK3/TEC (tyrosine kinase expressed in hepatocellular carcinoma) inhibitor in clinical trials – is a welcome development for treatment of vitiligo, said John E. Harris, MD, PhD, chair of dermatology and director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester. Also in the pipeline is a kit for melanocyte-keratinocyte transplantation, which involves transplanting epidermal cells from one part of the body to another. This can be a challenging procedure but a kit would make it easier for a wider range of practitioners. (Topical ruxolitinib was approved by the Food and Drug Administration for treating nonsegmental vitiligo in July, 2022.)
“In the last 10 years, it’s just blown up and people care about vitiligo now,” Dr. Harris said, noting that vitiligo is more than a cosmetic issue, like gray hair or wrinkles. “Vitiligo is an autoimmune disease and now is being treated as such.”
Nada Elbuluk, MD, MSc, associate professor of dermatology at the University of Southern California, Los Angeles, said she’s pleased at the increasing availability of treatment options for hyperpigmentation, aside from hydroquinone, which is associated with an increased risk of adverse effects.
“We have more and more nonhydroquinone agents ... which is really nice because it expands our treatment armamentarium and what we can use to cycle people off of hydroquinone,” she said.
Some of these options include tranexamic acid and products containing azelaic acid or vitamin C.
Iltefat H. Hamzavi, MD, senior staff physician at Henry Ford Health System, Detroit, said that pigmentary disorders are receiving the recognition they deserve.
“I’m excited just about the intersection of society and science, the awareness that pigmentary abnormalities mean something, and they mean something across our society,” he said.
Dr. Elbuluk said that hyperpigmentation has “profound effects on quality of life” for patients.
“They are often more bothered by the darkening of the skin than the primary process that caused it,” she said. “It’s not uncommon that the chief complaint will say ‘dark spots’ and I walk in a room and it’s a patient who has acne. They don’t even say they’re here for acne. They just put ‘dark spots’ [down] because that’s what bothers them. That’s what lasts for so long after the acne is gone.”
The experts offered suggestions for managing these cases. Among her tips, Dr. Elbuluk said that for hyperpigmentation, physicians should not be afraid to biopsy the face – but suggested small, 2-millimeter specimens. In addition, “you can get common conditions in uncommon places,” she noted. “If you see something that looks like melasma off the face, it actually could be, so keep that in your differential.”
Dr. Hamzavi, who spoke about hypopigmentation disorders, said clinicians need to use an algorithm for diagnosis, considering features such as localized or diffuse, scale or no scale, as well as patient history, and other factors. For instance, a hypopigmented area that is localized and has a reddish central papule might lead a clinician to a diagnosis of hypopigmented sarcoidosis.
Using the algorithms, “you actually have to categorize these and then use your own experience. ... All of these elements can help you become a really good taxonomist – ultimately that’s what physicians are.”
He said it’s also important to know when it’s time to reconsider a diagnosis, such as when patients do not respond to traditional treatments. “If they don’t respond, re-categorize,” he said.
Speaking about vitiligo, Dr. Harris said it’s crucial to differentiate active vitiligo from inactive vitiligo and if it’s active, steps need to be taken to keep it from worsening..
Four signs of active vitiligo are a “confetti” pattern of clustered tiny macules of depigmentation, which will coalesce quickly into huge patches; tri-chrome vitiligo that includes a hypopigmented zone; linear areas of depigmentation (Koebner’s phenomenon) that look like scratches on the skin; and inflammatory vitiligo, with an erythematous ring around the edges of a depigmented area.
Dr. Harris disclosed ties with Incyte, Pfizer, Abbvie, Genzyme/Sanofi and other companies. Dr. Elbuluk disclosed ties with Avita, Incyte, Beiersdorf, and other companies. Dr. Hamzavi disclosed ties with AbbVie, Pfizer, Incyte, and other companies.
AT ODAC 2023
Microneedling With Bimatoprost to Treat Hypopigmented Skin Caused by Burn Scars
To the Editor:
Microneedling is a percutaneous collagen induction therapy frequently used in cosmetic dermatology to promote skin rejuvenation and hair growth and to treat scars by taking advantage of the body’s natural wound-healing cascade.1 The procedure works by generating thousands of microscopic wounds in the dermis with minimal damage to the epidermis, thus initiating the wound-healing cascade and subsequently promoting collagen production in a manner safe for all Fitzpatrick classification skin types.1-3 This therapy effectively treats scars by breaking down scarred collagen and replacing it with new healthy collagen. Microneedling also has application in drug delivery by increasing the permeability of the skin; the microwounds generated can serve as a portal for drug delivery.4
Bimatoprost is a prostaglandin analogue typically used to treat hypotrichosis and open-angle glaucoma.5-7 A known side effect of bimatoprost is hyperpigmentation of surrounding skin; the drug increases melanogenesis, melanocyte proliferation, and melanocyte dendricity, resulting in activation of the inflammatory response and subsequent prostaglandin release, which stimulates melanogenesis. This effect is similar to UV radiation–induced inflammation and hyperpigmentation.6,8
Capitalizing on this effect, a novel application of bimatoprost has been proposed—treating vitiligo, in which hypopigmentation results from destruction of melanocytes in certain areas of the skin. Bimatoprost ophthalmic solution 0.3% utilized as an off-label treatment for vitiligo has been shown to notably increase melanogenesis and return pigmentation to hypopigmented areas.8-10
A 32-year-old Black woman presented to our clinic with a 40×15-cm scar that was marked by postinflammatory hypopigmentation from a second-degree burn on the right proximal arm. The patient had been burned 5 months prior by boiling water that was spilled on the arm while cooking. She had immediately sought treatment at an emergency department and subsequently in a burn unit, where the burn was debrided twice; medication was not prescribed to continue treatment. The patient reported that the scarring and hypopigmentation had taken a psychologic toll; her hope was to have pigmentation restored to the affected area to boost her confidence.
Physical examination revealed that the burn wound had healed but visible scarring and severe hypopigmentation due to destroyed melanocytes remained (Figure 1). To inhibit inflammation and stimulate repigmentation, we prescribed the calcineurin inhibitor tacrolimus ointment 0.1% to be applied daily to the affected area. The patient returned to the clinic 1 month later. Perifollicular hyperpigmentation was noted at the site of the scar.
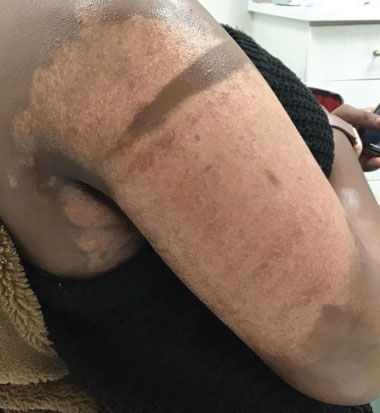
Monthly microneedling sessions with bimatoprost ophthalmic solution 0.3% were started. To avoid damaging any potentially remaining unhealed hypodermis and vasculature, the first microneedling session was performed with 9 needles set at minimal needle depth and frequency. The number of needles and their depth and frequency gradually were increased with each subsequent treatment. The patient continued tacrolimus ointment 0.1% throughout the course of treatment.
For each microneedling procedure, a handheld motorized microneedling device was applied to the skin at a depth of 0.25 mm, which was gradually increased until pinpoint petechiae were achieved. Bimatoprost ophthalmic solution 0.3% was then painted on the skin and allowed to absorb. Microneedling was performed again, ensuring that bimatoprost entered the skin in the area of the burn scar.
Microneedling procedures were performed monthly for 6 months, then once 3 months later, and once more 3 months later—8 treatments in total over the course of 1 year. Improvement in skin pigmentation was noted at each visit (Figure 2). Repigmentation was first noticed surrounding hair follicles; after later visits, it was observed that pigmentation began to spread from hair follicles to fill in remaining skin. The darkest areas of pigmentation were first noted around hair follicles; over time, melanocytes appeared to spontaneously regenerate and fill in surrounding areas as the scar continued to heal. The patient continued use of tacrolimus during the entire course of microneedling treatments and for the following 4 months. Sixteen months after initiation of treatment, the appearance of the skin was texturally smooth and returned to almost its original pigmentation (Figure 3).
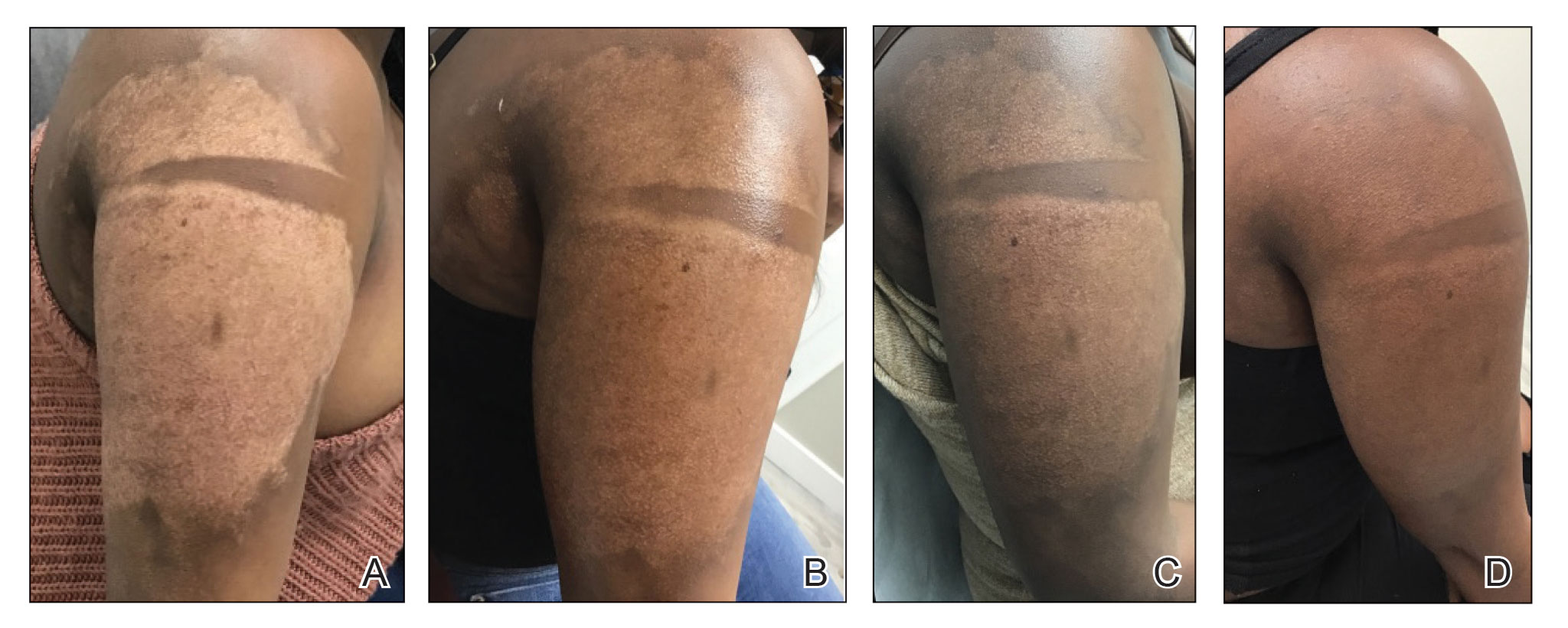
We report a successful outcome in a patient with a hypopigmented burn scar who was treated with bimatoprost administered with traditional microneedling and alongside a tacrolimus regimen. Tacrolimus ointment inhibited the inflammatory response to allow melanocytes to heal and regenerate; bimatoprost and microneedling promoted hyperpigmentation of hair follicles in the affected area, eventually restoring pigmentation to the entire area. Our patient was extremely satisfied with the results of this combination treatment. She has reported feeling more confident going out and wearing short-sleeved clothing. Percutaneous drug delivery of bimatoprost ophthalmic solution 0.3% combined with topical tacrolimus may be an effective treatment for skin repigmentation. Further investigation of this regimen is needed to develop standardized treatment protocols.
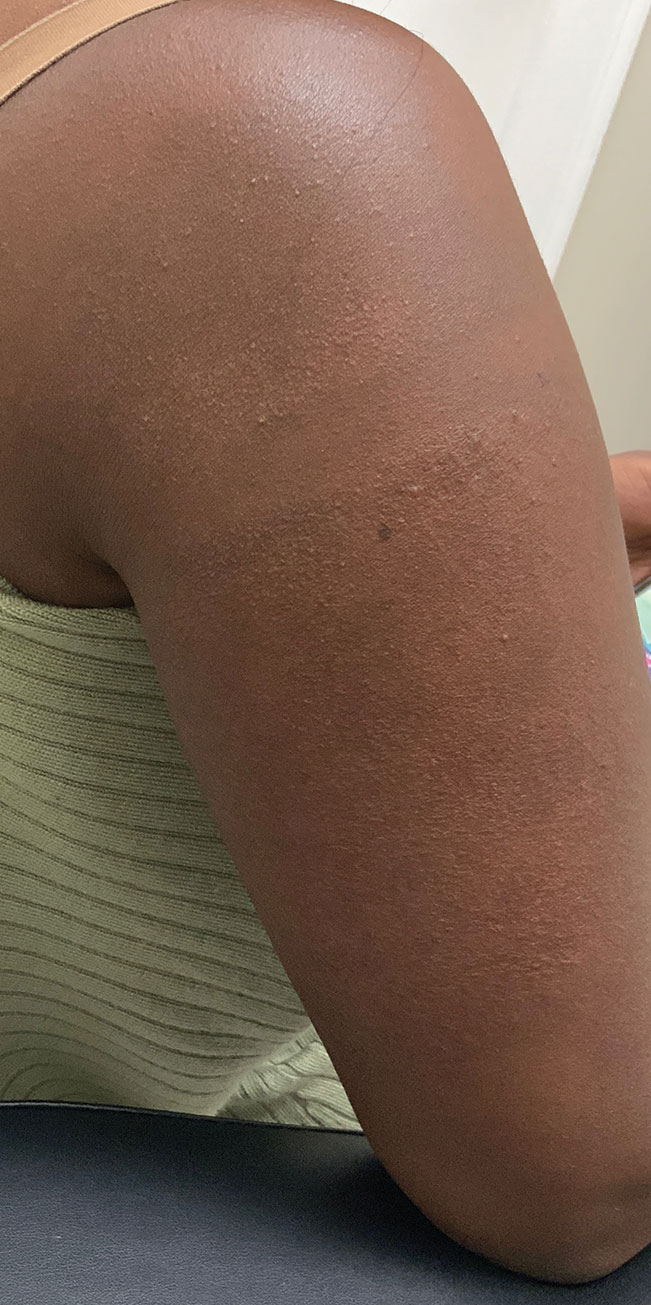
- Juhasz MLW, Cohen JL. Micro-needling for the treatment of scars: an update for clinicians. Clin Cosmet Investig Dermatol. 2020;13:997-1003. doi:10.2147/CCID.S267192
- Alster TS, Li MKY. Micro-needling of scars: a large prospective study with long-term follow-up. Plast Reconstr Surg. 2020;145:358-364. doi:10.1097/PRS.0000000000006462
- Aust MC, Knobloch K, Reimers K, et al. Percutaneous collagen induction therapy: an alternative treatment for burn scars. Burns. 2010;36:836-843. doi:10.1016/j.burns.2009.11.014
- Kim Y-C, Park J-H, Prausnitz MR. Microneedles for drug and vaccine delivery. Adv Drug Deliv Rev. 2012;64:1547-1568. doi:10.1016/j.addr.2012.04.005
- Doshi M, Edward DP, Osmanovic S. Clinical course of bimatoprost-induced periocular skin changes in Caucasians. Ophthalmology. 2006;113:1961-1967. doi:10.1016/j.ophtha.2006.05.041
- Kapur R, Osmanovic S, Toyran S, et al. Bimatoprost-induced periocular skin hyperpigmentation: histopathological study. Arch Ophthalmol. 2005;123:1541-1546. doi:10.1001/archopht.123.11.1541
- Priluck JC, Fu S. Latisse-induced periocular skin hyperpigmentation. Arch Ophthalmol. 2010;128:792-793. doi:10.1001/archophthalmol.2010.89
- Grimes PE. Bimatoprost 0.03% solution for the treatment of nonfacial vitiligo. J Drugs Dermatol. 2016;15:703-710.
- Barbulescu C, Goldstein N, Roop D, et al. Harnessing the power of regenerative therapy for vitiligo and alopecia areata. J Invest Dermatol. 2020;140: 29-37. doi:10.1016/j.jid.2019.03.1142
- Kanokrungsee S, Pruettivorawongse D, Rajatanavin N. Clinicaloutcomes of topical bimatoprost for nonsegmental facial vitiligo: a preliminary study. J Cosmet Dermatol. 2021;20:812-818. doi.org/10.1111/jocd.13648
To the Editor:
Microneedling is a percutaneous collagen induction therapy frequently used in cosmetic dermatology to promote skin rejuvenation and hair growth and to treat scars by taking advantage of the body’s natural wound-healing cascade.1 The procedure works by generating thousands of microscopic wounds in the dermis with minimal damage to the epidermis, thus initiating the wound-healing cascade and subsequently promoting collagen production in a manner safe for all Fitzpatrick classification skin types.1-3 This therapy effectively treats scars by breaking down scarred collagen and replacing it with new healthy collagen. Microneedling also has application in drug delivery by increasing the permeability of the skin; the microwounds generated can serve as a portal for drug delivery.4
Bimatoprost is a prostaglandin analogue typically used to treat hypotrichosis and open-angle glaucoma.5-7 A known side effect of bimatoprost is hyperpigmentation of surrounding skin; the drug increases melanogenesis, melanocyte proliferation, and melanocyte dendricity, resulting in activation of the inflammatory response and subsequent prostaglandin release, which stimulates melanogenesis. This effect is similar to UV radiation–induced inflammation and hyperpigmentation.6,8
Capitalizing on this effect, a novel application of bimatoprost has been proposed—treating vitiligo, in which hypopigmentation results from destruction of melanocytes in certain areas of the skin. Bimatoprost ophthalmic solution 0.3% utilized as an off-label treatment for vitiligo has been shown to notably increase melanogenesis and return pigmentation to hypopigmented areas.8-10
A 32-year-old Black woman presented to our clinic with a 40×15-cm scar that was marked by postinflammatory hypopigmentation from a second-degree burn on the right proximal arm. The patient had been burned 5 months prior by boiling water that was spilled on the arm while cooking. She had immediately sought treatment at an emergency department and subsequently in a burn unit, where the burn was debrided twice; medication was not prescribed to continue treatment. The patient reported that the scarring and hypopigmentation had taken a psychologic toll; her hope was to have pigmentation restored to the affected area to boost her confidence.
Physical examination revealed that the burn wound had healed but visible scarring and severe hypopigmentation due to destroyed melanocytes remained (Figure 1). To inhibit inflammation and stimulate repigmentation, we prescribed the calcineurin inhibitor tacrolimus ointment 0.1% to be applied daily to the affected area. The patient returned to the clinic 1 month later. Perifollicular hyperpigmentation was noted at the site of the scar.

Monthly microneedling sessions with bimatoprost ophthalmic solution 0.3% were started. To avoid damaging any potentially remaining unhealed hypodermis and vasculature, the first microneedling session was performed with 9 needles set at minimal needle depth and frequency. The number of needles and their depth and frequency gradually were increased with each subsequent treatment. The patient continued tacrolimus ointment 0.1% throughout the course of treatment.
For each microneedling procedure, a handheld motorized microneedling device was applied to the skin at a depth of 0.25 mm, which was gradually increased until pinpoint petechiae were achieved. Bimatoprost ophthalmic solution 0.3% was then painted on the skin and allowed to absorb. Microneedling was performed again, ensuring that bimatoprost entered the skin in the area of the burn scar.
Microneedling procedures were performed monthly for 6 months, then once 3 months later, and once more 3 months later—8 treatments in total over the course of 1 year. Improvement in skin pigmentation was noted at each visit (Figure 2). Repigmentation was first noticed surrounding hair follicles; after later visits, it was observed that pigmentation began to spread from hair follicles to fill in remaining skin. The darkest areas of pigmentation were first noted around hair follicles; over time, melanocytes appeared to spontaneously regenerate and fill in surrounding areas as the scar continued to heal. The patient continued use of tacrolimus during the entire course of microneedling treatments and for the following 4 months. Sixteen months after initiation of treatment, the appearance of the skin was texturally smooth and returned to almost its original pigmentation (Figure 3).

We report a successful outcome in a patient with a hypopigmented burn scar who was treated with bimatoprost administered with traditional microneedling and alongside a tacrolimus regimen. Tacrolimus ointment inhibited the inflammatory response to allow melanocytes to heal and regenerate; bimatoprost and microneedling promoted hyperpigmentation of hair follicles in the affected area, eventually restoring pigmentation to the entire area. Our patient was extremely satisfied with the results of this combination treatment. She has reported feeling more confident going out and wearing short-sleeved clothing. Percutaneous drug delivery of bimatoprost ophthalmic solution 0.3% combined with topical tacrolimus may be an effective treatment for skin repigmentation. Further investigation of this regimen is needed to develop standardized treatment protocols.

To the Editor:
Microneedling is a percutaneous collagen induction therapy frequently used in cosmetic dermatology to promote skin rejuvenation and hair growth and to treat scars by taking advantage of the body’s natural wound-healing cascade.1 The procedure works by generating thousands of microscopic wounds in the dermis with minimal damage to the epidermis, thus initiating the wound-healing cascade and subsequently promoting collagen production in a manner safe for all Fitzpatrick classification skin types.1-3 This therapy effectively treats scars by breaking down scarred collagen and replacing it with new healthy collagen. Microneedling also has application in drug delivery by increasing the permeability of the skin; the microwounds generated can serve as a portal for drug delivery.4
Bimatoprost is a prostaglandin analogue typically used to treat hypotrichosis and open-angle glaucoma.5-7 A known side effect of bimatoprost is hyperpigmentation of surrounding skin; the drug increases melanogenesis, melanocyte proliferation, and melanocyte dendricity, resulting in activation of the inflammatory response and subsequent prostaglandin release, which stimulates melanogenesis. This effect is similar to UV radiation–induced inflammation and hyperpigmentation.6,8
Capitalizing on this effect, a novel application of bimatoprost has been proposed—treating vitiligo, in which hypopigmentation results from destruction of melanocytes in certain areas of the skin. Bimatoprost ophthalmic solution 0.3% utilized as an off-label treatment for vitiligo has been shown to notably increase melanogenesis and return pigmentation to hypopigmented areas.8-10
A 32-year-old Black woman presented to our clinic with a 40×15-cm scar that was marked by postinflammatory hypopigmentation from a second-degree burn on the right proximal arm. The patient had been burned 5 months prior by boiling water that was spilled on the arm while cooking. She had immediately sought treatment at an emergency department and subsequently in a burn unit, where the burn was debrided twice; medication was not prescribed to continue treatment. The patient reported that the scarring and hypopigmentation had taken a psychologic toll; her hope was to have pigmentation restored to the affected area to boost her confidence.
Physical examination revealed that the burn wound had healed but visible scarring and severe hypopigmentation due to destroyed melanocytes remained (Figure 1). To inhibit inflammation and stimulate repigmentation, we prescribed the calcineurin inhibitor tacrolimus ointment 0.1% to be applied daily to the affected area. The patient returned to the clinic 1 month later. Perifollicular hyperpigmentation was noted at the site of the scar.

Monthly microneedling sessions with bimatoprost ophthalmic solution 0.3% were started. To avoid damaging any potentially remaining unhealed hypodermis and vasculature, the first microneedling session was performed with 9 needles set at minimal needle depth and frequency. The number of needles and their depth and frequency gradually were increased with each subsequent treatment. The patient continued tacrolimus ointment 0.1% throughout the course of treatment.
For each microneedling procedure, a handheld motorized microneedling device was applied to the skin at a depth of 0.25 mm, which was gradually increased until pinpoint petechiae were achieved. Bimatoprost ophthalmic solution 0.3% was then painted on the skin and allowed to absorb. Microneedling was performed again, ensuring that bimatoprost entered the skin in the area of the burn scar.
Microneedling procedures were performed monthly for 6 months, then once 3 months later, and once more 3 months later—8 treatments in total over the course of 1 year. Improvement in skin pigmentation was noted at each visit (Figure 2). Repigmentation was first noticed surrounding hair follicles; after later visits, it was observed that pigmentation began to spread from hair follicles to fill in remaining skin. The darkest areas of pigmentation were first noted around hair follicles; over time, melanocytes appeared to spontaneously regenerate and fill in surrounding areas as the scar continued to heal. The patient continued use of tacrolimus during the entire course of microneedling treatments and for the following 4 months. Sixteen months after initiation of treatment, the appearance of the skin was texturally smooth and returned to almost its original pigmentation (Figure 3).

We report a successful outcome in a patient with a hypopigmented burn scar who was treated with bimatoprost administered with traditional microneedling and alongside a tacrolimus regimen. Tacrolimus ointment inhibited the inflammatory response to allow melanocytes to heal and regenerate; bimatoprost and microneedling promoted hyperpigmentation of hair follicles in the affected area, eventually restoring pigmentation to the entire area. Our patient was extremely satisfied with the results of this combination treatment. She has reported feeling more confident going out and wearing short-sleeved clothing. Percutaneous drug delivery of bimatoprost ophthalmic solution 0.3% combined with topical tacrolimus may be an effective treatment for skin repigmentation. Further investigation of this regimen is needed to develop standardized treatment protocols.

- Juhasz MLW, Cohen JL. Micro-needling for the treatment of scars: an update for clinicians. Clin Cosmet Investig Dermatol. 2020;13:997-1003. doi:10.2147/CCID.S267192
- Alster TS, Li MKY. Micro-needling of scars: a large prospective study with long-term follow-up. Plast Reconstr Surg. 2020;145:358-364. doi:10.1097/PRS.0000000000006462
- Aust MC, Knobloch K, Reimers K, et al. Percutaneous collagen induction therapy: an alternative treatment for burn scars. Burns. 2010;36:836-843. doi:10.1016/j.burns.2009.11.014
- Kim Y-C, Park J-H, Prausnitz MR. Microneedles for drug and vaccine delivery. Adv Drug Deliv Rev. 2012;64:1547-1568. doi:10.1016/j.addr.2012.04.005
- Doshi M, Edward DP, Osmanovic S. Clinical course of bimatoprost-induced periocular skin changes in Caucasians. Ophthalmology. 2006;113:1961-1967. doi:10.1016/j.ophtha.2006.05.041
- Kapur R, Osmanovic S, Toyran S, et al. Bimatoprost-induced periocular skin hyperpigmentation: histopathological study. Arch Ophthalmol. 2005;123:1541-1546. doi:10.1001/archopht.123.11.1541
- Priluck JC, Fu S. Latisse-induced periocular skin hyperpigmentation. Arch Ophthalmol. 2010;128:792-793. doi:10.1001/archophthalmol.2010.89
- Grimes PE. Bimatoprost 0.03% solution for the treatment of nonfacial vitiligo. J Drugs Dermatol. 2016;15:703-710.
- Barbulescu C, Goldstein N, Roop D, et al. Harnessing the power of regenerative therapy for vitiligo and alopecia areata. J Invest Dermatol. 2020;140: 29-37. doi:10.1016/j.jid.2019.03.1142
- Kanokrungsee S, Pruettivorawongse D, Rajatanavin N. Clinicaloutcomes of topical bimatoprost for nonsegmental facial vitiligo: a preliminary study. J Cosmet Dermatol. 2021;20:812-818. doi.org/10.1111/jocd.13648
- Juhasz MLW, Cohen JL. Micro-needling for the treatment of scars: an update for clinicians. Clin Cosmet Investig Dermatol. 2020;13:997-1003. doi:10.2147/CCID.S267192
- Alster TS, Li MKY. Micro-needling of scars: a large prospective study with long-term follow-up. Plast Reconstr Surg. 2020;145:358-364. doi:10.1097/PRS.0000000000006462
- Aust MC, Knobloch K, Reimers K, et al. Percutaneous collagen induction therapy: an alternative treatment for burn scars. Burns. 2010;36:836-843. doi:10.1016/j.burns.2009.11.014
- Kim Y-C, Park J-H, Prausnitz MR. Microneedles for drug and vaccine delivery. Adv Drug Deliv Rev. 2012;64:1547-1568. doi:10.1016/j.addr.2012.04.005
- Doshi M, Edward DP, Osmanovic S. Clinical course of bimatoprost-induced periocular skin changes in Caucasians. Ophthalmology. 2006;113:1961-1967. doi:10.1016/j.ophtha.2006.05.041
- Kapur R, Osmanovic S, Toyran S, et al. Bimatoprost-induced periocular skin hyperpigmentation: histopathological study. Arch Ophthalmol. 2005;123:1541-1546. doi:10.1001/archopht.123.11.1541
- Priluck JC, Fu S. Latisse-induced periocular skin hyperpigmentation. Arch Ophthalmol. 2010;128:792-793. doi:10.1001/archophthalmol.2010.89
- Grimes PE. Bimatoprost 0.03% solution for the treatment of nonfacial vitiligo. J Drugs Dermatol. 2016;15:703-710.
- Barbulescu C, Goldstein N, Roop D, et al. Harnessing the power of regenerative therapy for vitiligo and alopecia areata. J Invest Dermatol. 2020;140: 29-37. doi:10.1016/j.jid.2019.03.1142
- Kanokrungsee S, Pruettivorawongse D, Rajatanavin N. Clinicaloutcomes of topical bimatoprost for nonsegmental facial vitiligo: a preliminary study. J Cosmet Dermatol. 2021;20:812-818. doi.org/10.1111/jocd.13648
PRACTICE POINTS
- Microneedling is a percutaneous collagen induction therapy that also may be used in drug delivery.
- Hypopigmentation can cause considerable distress for patients with skin of color.
- Percutaneous drug delivery of bimatoprost may be helpful in skin repigmentation.
A toddler presents with a dark line on a fingernail
Given the over 1-year history of an unchanging longitudinal band of pigment without extension to the proximal or lateral nailfolds or any other nail findings, the most likely diagnosis is benign longitudinal melanonychia.
Longitudinal melanonychia, also known as melanonychia striata, describes a brown to black streak of pigment extending from the nail matrix to the free edge of the nail.1,2
This disorder can occur secondary to a wide variety of benign and pathologic causes including lentigines, nevi, melanoma, chronic trauma, inflammatory skin diseases, systemic diseases, iatrogenic causes, and genetic syndromes.3 In melanocytic causes of longitudinal melanonychia, either melanocytic activation or hyperplasia drive pigmentary development leading to the brown to black band seen in the nail.4 Benign causes of longitudinal melanonychia include benign melanocyte activation, lentigo, and benign nevus.1
What’s the differential diagnosis?
The differential diagnosis for longitudinal melanonychia can include a wide variety of local and systemic causes. For our discussion, we will limit our differential to other locally involved disorders of the nail including subungual melanoma, subungual hematoma, onychomycosis, and glomus tumor.
Subungual melanoma is a rare subtype of acral lentiginous melanoma that most often presents as longitudinal melanonychia. Subungual melanoma is more common in those aged 50-70 years, individuals with personal or family history of melanoma or dysplastic nevus syndrome, and persons with African American, Native American, and Asian descent. Longitudinal melanonychia features that can be concerning for subungual melanoma include the presence of multiple colors, width greater than or equal to 3 mm, blurry borders, rapid increase in size, and extension to the proximal or lateral nailfolds (Hutchinson’s sign). Biopsy is required to make the diagnosis of subungual melanoma but is not necessary for melanonychia without atypical features.
Treatment of subungual melanoma depends on disease stage and can range from wide local excision of the nail apparatus to amputation of the affected digit and management with a medical oncologist. Given the absence of concerning neoplastic findings or personal or family history of melanoma, subungual melanoma is unlikely in this patient.
Subungual hematoma is an accumulation of blood underneath the nail plate that is typically the result of acute or chronic trauma to the distal phalanx. It can present as purple, red, pink, brown, or black discoloration under the nail plate and is most commonly found on the first toe. With acute trauma, pain is usually present upon initial injury. Subungual hematomas typically resolve on their own with normal nail growth. The absence of a history of trauma or pain, and the linear appearance of the lesion in our patient are inconsistent with a subungual hematoma.
Onychomycosis is a fungal infection of the nail caused by dermatophytes, nondermatophytes, or yeasts. It may present with longitudinal melanonychia; however, it more often presents with other nail abnormalities such as nail thickening, yellow discoloration, onycholysis, splitting, subungual hyperkeratosis, and nail plate destruction, which are not present in this patient. Furthermore, onychomycosis is more common in adults than children. Diagnosis is usually made with potassium hydroxide (KOH) preparations, histopathologic examination of nail clippings with a periodic acid-Schiff stain, fungal culture, or PCR.
Glomus tumor is a rare, benign neoplasm originating from cells of the glomus body. It is often found in the subungual region, in addition to other areas rich in glomus bodies such as the fingertips, palms, wrists, and forearms. Subungual glomus tumors present as a red, purple, or blueish lesions under the nail plate. Distal notching or an overlying longitudinal fissure may be present. Subungual glomus tumors are typically associated with pinpoint tenderness, paroxysmal pain, and cold sensitivity, features that are not present in our patient. The history and examination of our patient are much more consistent with benign longitudinal melanonychia.
It appears that melanoma associated with longitudinal melanonychia is very rare in children. According to one review published in 2020, only 12 cases of pediatric subungual melanoma have been reported.5 Recent series have observed longitudinal melanonychia in large sets of children, with findings that demonstrate that the vast majority of longitudinal melanonychia either stops progressing or regresses. These investigations therefore recommend serial observation of longitudinal melanonychia except in rare circumstances.6,7
Given the lack of troubling findings or concerning history, our patient was managed with observation. On follow-up 6 months later, he was found to have no change in his nail pigmentation.
Dr. Haft is an inflammatory skin disease fellow in the division of pediatric and adolescent dermatology; Ms. Sui is a research associate in the department of dermatology, division of pediatric and adolescent dermatology; and Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics, all at the University of California and Rady Children’s Hospital, San Diego. They have no relevant disclosures.
References
1. Mannava KA et al. Hand Surg. 2013;18(1):133-9.
2. Leung AKC et al. Int J Dermatol. 2019;58(11):1239-45.
3. Andre J and Lateur N. Dermatol Clin. 2006;24(3):329-39.
4. Lee DK and Lipner SR. Ann Med. 2022;54(1):694-712.
5. Smith RJ and Rubin AI. Curr Opin Pediatr. 2020;32(4):506-15. .
6. Matsui Y et al. J Am Acad Dermatol. 2022;86(4):946-8.
7. Lee JS et al. J Am Acad Dermatol. 2022;87(2):366-72.
Given the over 1-year history of an unchanging longitudinal band of pigment without extension to the proximal or lateral nailfolds or any other nail findings, the most likely diagnosis is benign longitudinal melanonychia.
Longitudinal melanonychia, also known as melanonychia striata, describes a brown to black streak of pigment extending from the nail matrix to the free edge of the nail.1,2
This disorder can occur secondary to a wide variety of benign and pathologic causes including lentigines, nevi, melanoma, chronic trauma, inflammatory skin diseases, systemic diseases, iatrogenic causes, and genetic syndromes.3 In melanocytic causes of longitudinal melanonychia, either melanocytic activation or hyperplasia drive pigmentary development leading to the brown to black band seen in the nail.4 Benign causes of longitudinal melanonychia include benign melanocyte activation, lentigo, and benign nevus.1
What’s the differential diagnosis?
The differential diagnosis for longitudinal melanonychia can include a wide variety of local and systemic causes. For our discussion, we will limit our differential to other locally involved disorders of the nail including subungual melanoma, subungual hematoma, onychomycosis, and glomus tumor.
Subungual melanoma is a rare subtype of acral lentiginous melanoma that most often presents as longitudinal melanonychia. Subungual melanoma is more common in those aged 50-70 years, individuals with personal or family history of melanoma or dysplastic nevus syndrome, and persons with African American, Native American, and Asian descent. Longitudinal melanonychia features that can be concerning for subungual melanoma include the presence of multiple colors, width greater than or equal to 3 mm, blurry borders, rapid increase in size, and extension to the proximal or lateral nailfolds (Hutchinson’s sign). Biopsy is required to make the diagnosis of subungual melanoma but is not necessary for melanonychia without atypical features.
Treatment of subungual melanoma depends on disease stage and can range from wide local excision of the nail apparatus to amputation of the affected digit and management with a medical oncologist. Given the absence of concerning neoplastic findings or personal or family history of melanoma, subungual melanoma is unlikely in this patient.
Subungual hematoma is an accumulation of blood underneath the nail plate that is typically the result of acute or chronic trauma to the distal phalanx. It can present as purple, red, pink, brown, or black discoloration under the nail plate and is most commonly found on the first toe. With acute trauma, pain is usually present upon initial injury. Subungual hematomas typically resolve on their own with normal nail growth. The absence of a history of trauma or pain, and the linear appearance of the lesion in our patient are inconsistent with a subungual hematoma.
Onychomycosis is a fungal infection of the nail caused by dermatophytes, nondermatophytes, or yeasts. It may present with longitudinal melanonychia; however, it more often presents with other nail abnormalities such as nail thickening, yellow discoloration, onycholysis, splitting, subungual hyperkeratosis, and nail plate destruction, which are not present in this patient. Furthermore, onychomycosis is more common in adults than children. Diagnosis is usually made with potassium hydroxide (KOH) preparations, histopathologic examination of nail clippings with a periodic acid-Schiff stain, fungal culture, or PCR.
Glomus tumor is a rare, benign neoplasm originating from cells of the glomus body. It is often found in the subungual region, in addition to other areas rich in glomus bodies such as the fingertips, palms, wrists, and forearms. Subungual glomus tumors present as a red, purple, or blueish lesions under the nail plate. Distal notching or an overlying longitudinal fissure may be present. Subungual glomus tumors are typically associated with pinpoint tenderness, paroxysmal pain, and cold sensitivity, features that are not present in our patient. The history and examination of our patient are much more consistent with benign longitudinal melanonychia.
It appears that melanoma associated with longitudinal melanonychia is very rare in children. According to one review published in 2020, only 12 cases of pediatric subungual melanoma have been reported.5 Recent series have observed longitudinal melanonychia in large sets of children, with findings that demonstrate that the vast majority of longitudinal melanonychia either stops progressing or regresses. These investigations therefore recommend serial observation of longitudinal melanonychia except in rare circumstances.6,7
Given the lack of troubling findings or concerning history, our patient was managed with observation. On follow-up 6 months later, he was found to have no change in his nail pigmentation.
Dr. Haft is an inflammatory skin disease fellow in the division of pediatric and adolescent dermatology; Ms. Sui is a research associate in the department of dermatology, division of pediatric and adolescent dermatology; and Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics, all at the University of California and Rady Children’s Hospital, San Diego. They have no relevant disclosures.
References
1. Mannava KA et al. Hand Surg. 2013;18(1):133-9.
2. Leung AKC et al. Int J Dermatol. 2019;58(11):1239-45.
3. Andre J and Lateur N. Dermatol Clin. 2006;24(3):329-39.
4. Lee DK and Lipner SR. Ann Med. 2022;54(1):694-712.
5. Smith RJ and Rubin AI. Curr Opin Pediatr. 2020;32(4):506-15. .
6. Matsui Y et al. J Am Acad Dermatol. 2022;86(4):946-8.
7. Lee JS et al. J Am Acad Dermatol. 2022;87(2):366-72.
Given the over 1-year history of an unchanging longitudinal band of pigment without extension to the proximal or lateral nailfolds or any other nail findings, the most likely diagnosis is benign longitudinal melanonychia.
Longitudinal melanonychia, also known as melanonychia striata, describes a brown to black streak of pigment extending from the nail matrix to the free edge of the nail.1,2
This disorder can occur secondary to a wide variety of benign and pathologic causes including lentigines, nevi, melanoma, chronic trauma, inflammatory skin diseases, systemic diseases, iatrogenic causes, and genetic syndromes.3 In melanocytic causes of longitudinal melanonychia, either melanocytic activation or hyperplasia drive pigmentary development leading to the brown to black band seen in the nail.4 Benign causes of longitudinal melanonychia include benign melanocyte activation, lentigo, and benign nevus.1
What’s the differential diagnosis?
The differential diagnosis for longitudinal melanonychia can include a wide variety of local and systemic causes. For our discussion, we will limit our differential to other locally involved disorders of the nail including subungual melanoma, subungual hematoma, onychomycosis, and glomus tumor.
Subungual melanoma is a rare subtype of acral lentiginous melanoma that most often presents as longitudinal melanonychia. Subungual melanoma is more common in those aged 50-70 years, individuals with personal or family history of melanoma or dysplastic nevus syndrome, and persons with African American, Native American, and Asian descent. Longitudinal melanonychia features that can be concerning for subungual melanoma include the presence of multiple colors, width greater than or equal to 3 mm, blurry borders, rapid increase in size, and extension to the proximal or lateral nailfolds (Hutchinson’s sign). Biopsy is required to make the diagnosis of subungual melanoma but is not necessary for melanonychia without atypical features.
Treatment of subungual melanoma depends on disease stage and can range from wide local excision of the nail apparatus to amputation of the affected digit and management with a medical oncologist. Given the absence of concerning neoplastic findings or personal or family history of melanoma, subungual melanoma is unlikely in this patient.
Subungual hematoma is an accumulation of blood underneath the nail plate that is typically the result of acute or chronic trauma to the distal phalanx. It can present as purple, red, pink, brown, or black discoloration under the nail plate and is most commonly found on the first toe. With acute trauma, pain is usually present upon initial injury. Subungual hematomas typically resolve on their own with normal nail growth. The absence of a history of trauma or pain, and the linear appearance of the lesion in our patient are inconsistent with a subungual hematoma.
Onychomycosis is a fungal infection of the nail caused by dermatophytes, nondermatophytes, or yeasts. It may present with longitudinal melanonychia; however, it more often presents with other nail abnormalities such as nail thickening, yellow discoloration, onycholysis, splitting, subungual hyperkeratosis, and nail plate destruction, which are not present in this patient. Furthermore, onychomycosis is more common in adults than children. Diagnosis is usually made with potassium hydroxide (KOH) preparations, histopathologic examination of nail clippings with a periodic acid-Schiff stain, fungal culture, or PCR.
Glomus tumor is a rare, benign neoplasm originating from cells of the glomus body. It is often found in the subungual region, in addition to other areas rich in glomus bodies such as the fingertips, palms, wrists, and forearms. Subungual glomus tumors present as a red, purple, or blueish lesions under the nail plate. Distal notching or an overlying longitudinal fissure may be present. Subungual glomus tumors are typically associated with pinpoint tenderness, paroxysmal pain, and cold sensitivity, features that are not present in our patient. The history and examination of our patient are much more consistent with benign longitudinal melanonychia.
It appears that melanoma associated with longitudinal melanonychia is very rare in children. According to one review published in 2020, only 12 cases of pediatric subungual melanoma have been reported.5 Recent series have observed longitudinal melanonychia in large sets of children, with findings that demonstrate that the vast majority of longitudinal melanonychia either stops progressing or regresses. These investigations therefore recommend serial observation of longitudinal melanonychia except in rare circumstances.6,7
Given the lack of troubling findings or concerning history, our patient was managed with observation. On follow-up 6 months later, he was found to have no change in his nail pigmentation.
Dr. Haft is an inflammatory skin disease fellow in the division of pediatric and adolescent dermatology; Ms. Sui is a research associate in the department of dermatology, division of pediatric and adolescent dermatology; and Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics, all at the University of California and Rady Children’s Hospital, San Diego. They have no relevant disclosures.
References
1. Mannava KA et al. Hand Surg. 2013;18(1):133-9.
2. Leung AKC et al. Int J Dermatol. 2019;58(11):1239-45.
3. Andre J and Lateur N. Dermatol Clin. 2006;24(3):329-39.
4. Lee DK and Lipner SR. Ann Med. 2022;54(1):694-712.
5. Smith RJ and Rubin AI. Curr Opin Pediatr. 2020;32(4):506-15. .
6. Matsui Y et al. J Am Acad Dermatol. 2022;86(4):946-8.
7. Lee JS et al. J Am Acad Dermatol. 2022;87(2):366-72.
Examination findings reveal a 2-mm brown longitudinal band on the radial aspect of the right thumbnail that does not extend into the proximal or lateral nailfolds. The rest of the skin and nail exam is unremarkable.
Long-term maintenance required in melasma patients
SAN DIEGO –
“They need to understand that melasma is going to require long-term maintenance,” Dr. Ortiz, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium.
Hydroquinone is a mainstay of melasma therapy, but instead of the commonly used 4% formulation, she prefers to use 12% hydroquinone with 6% kojic acid in VersaBase cream. “It’s a high concentration but the VersaBase makes it more tolerable,” she said. “I have patients take a pea-sized amount and mix it in a regular moisturizer. It’s too strong to spot treat, so it goes on the whole face.”
Mindful that chronic hydroquinone use can cause ochronosis (permanent darkening), she has patients alternate with a nonhydroquinone bleaching agent such as lignin peroxidase, oligopeptide, Lytera, Melaplex, 4-n-butylresorcinol, Cysteamine cream, tranexamic acid, or oral antioxidants. In a study sponsored by SkinMedica, investigators conducted a randomized, double-blind, half-face study in females with moderate to severe facial hyperpigmentation to assess the efficacy and tolerability of three new skin brightener formulations containing SMA-432, a prostaglandin E2 inhibitor, compared with 4% hydroquinone. They found that the nonhydroquinone skin formulations were better tolerated and were just as effective as 4% hydroquinone.
Chemical peels and laser treatments
Chemical peels are another treatment option for melasma, but Dr. Ortiz prefers glycolic peels over salicylic and other peels, “because there is no downtime,” she said.
As for laser-based approaches, melasma patients respond best to low energy devices such as the 1,927-nm fractional diode laser at a 3.75% density. “This also can increase the skin permeability of topicals, so when you’re combining it with hydroquinone it can be more effective,” she said.
In an observational study of 27 women with refractory melasma, with phototypes II-V, New York City–based dermatologist Arielle Kauvar, MD, combined microdermabrasion with the Q-switched Nd:YAG laser. “The settings she used were very low fluence, so there was no clinical endpoint or no whitening,” said Dr. Ortiz, vice president of the American Society for Laser Medicine and Surgery (ASLMS). Specifically, Dr. Kauvar used the laser at 1.6-2 J/cm2 with a 5- or 6-mm spot size immediately following microdermabrasion every 4 weeks; Patients received an average of 2.6 treatments, and were assessed 3-12 months after the last treatment. Study participants were on a standard skin care regimen of a broad spectrum sunscreen, hydroquinone, and tretinoin or vitamin C.
Most of the patients showed at least 50% clearance of melasma 1 month after the first treatment, and 81% showed more than 75% clearance of melasma; remission lasted at least 6 months.
“I personally prefer to use picosecond over Q-switched lasers, because they deliver the energy faster, and you can use a 1,064-nm picosecond laser that is safe in all skin types,” Dr. Ortiz said. “There is minimal downtime, and it doesn’t require anesthesia. You have to consider these things when you’re treating melasma, because this usually requires monthly treatments. If you do something that requires a week of downtime every month, it’s not practical for patients.”
In a study published in 2021, Dr. Ortiz and Tanya Greywal, MD, used three passes of the 1,064-nm Nd:YAG laser to treat melasma in 10 patients with skin types II-V. The device had a 650-microsecond pulse duration, a 6-mm spot size, and an energy mode of 11-14 J/cm2. The researchers observed a mean melasma improvement of 26%-50% as early as 3 weeks. “There was no downtime, and no anesthesia was required,” Dr. Ortiz said.
Researchers have discovered a vascular component to melasma, which may have treatment implications. Houston-based dermatologist Paul M. Friedman, MD, and his colleagues used spectrocolorimetry to detect an underlying prominent vascular component in a retrospective review of 11 patients with melasma, with skin types II-IV. They determined that melasma lesions exhibiting subtle or subclinical telangiectatic erythema may be improved by combining vascular-targeted laser therapy with fractional low-powered diode laser therapy.
“So, combining a vascular laser with a 1,927-nm fractional diode laser showed more improvement than with just the diode laser alone,” said Dr. Ortiz, who was not involved with the analysis.
To optimize results following the laser treatment of melasma, she uses one application of clobetasol immediately after the procedure. “This can help reduce swelling and inflammation to decrease the risk of postinflammatory hyperpigmentation,” she said. “You can also use a skin cooling system like Cryomodulation for controlled cooling.”
Tranexamic acid and PLE
Another strategy for melasma patients involves oral treatment with extract of Polypodium leucotomos (PLE), a fern from the Polypodiaceae family with antioxidant properties that has been shown to be photoprotective against UVA and UVB radiation. “I explain to my patients that it’s like an internal sunscreen,” Dr. Ortiz said. “It does not replace your external sunscreen, but it adds extra protection.”
In a pilot placebo-controlled study of patients with melasma on their normal regimen of hydroquinone and sunscreen, 40 Asian patients with melasma were randomized to receive either oral PLE supplementation or placebo for 12 weeks. The authors found that PLE significantly improved and accelerated the outcome reached with hydroquinone and sunscreen from about the first month of treatment, compared with placebo.
Dr. Ortiz discussed the role of oral tranexamic acid, an antifibrinolytic, procoagulant agent that is approved by the Food and Drug Administration for the treatment of menorrhagia and to prevent hemorrhage in patients with hemophilia undergoing tooth extractions. “This is a game changer for melasma treatment,” she said, but its use has been limited by the risk for thromboembolism.
In a study of 561 patients with melasma, 90% improved after a median treatment duration of 4 months, and only 7% had side effects, most commonly abdominal bloating and pain. Treatment was discontinued in one patient who developed a deep vein thrombosis, and was diagnosed with familial protein S deficiency.
The daily dosing of tranexamic acid for menorrhagia is 3,900 mg daily, while the dose for treating melasma has ranged from 500 mg to 1,500 mg per day, Dr. Ortiz said. It’s available as a 650-mg tablet in the United States. “I prescribe 325 mg twice a day, but studies have shown that 650 mg once a day is just as effective,” she said.
Prior to prescribing tranexamic acid, Dr. Ortiz does not order labs, but she performs an extensive history of current illness and does not prescribe it in patients with an increased risk of clotting, including people who smoke and those who take oral contraceptives or are on hormone supplementation. Use is also contraindicated in people with a current malignancy, those with a history of stroke or DVT, and those who have any clotting disorder.
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies. She is cochair of the Masters of Aesthetics Symposium.
SAN DIEGO –
“They need to understand that melasma is going to require long-term maintenance,” Dr. Ortiz, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium.
Hydroquinone is a mainstay of melasma therapy, but instead of the commonly used 4% formulation, she prefers to use 12% hydroquinone with 6% kojic acid in VersaBase cream. “It’s a high concentration but the VersaBase makes it more tolerable,” she said. “I have patients take a pea-sized amount and mix it in a regular moisturizer. It’s too strong to spot treat, so it goes on the whole face.”
Mindful that chronic hydroquinone use can cause ochronosis (permanent darkening), she has patients alternate with a nonhydroquinone bleaching agent such as lignin peroxidase, oligopeptide, Lytera, Melaplex, 4-n-butylresorcinol, Cysteamine cream, tranexamic acid, or oral antioxidants. In a study sponsored by SkinMedica, investigators conducted a randomized, double-blind, half-face study in females with moderate to severe facial hyperpigmentation to assess the efficacy and tolerability of three new skin brightener formulations containing SMA-432, a prostaglandin E2 inhibitor, compared with 4% hydroquinone. They found that the nonhydroquinone skin formulations were better tolerated and were just as effective as 4% hydroquinone.
Chemical peels and laser treatments
Chemical peels are another treatment option for melasma, but Dr. Ortiz prefers glycolic peels over salicylic and other peels, “because there is no downtime,” she said.
As for laser-based approaches, melasma patients respond best to low energy devices such as the 1,927-nm fractional diode laser at a 3.75% density. “This also can increase the skin permeability of topicals, so when you’re combining it with hydroquinone it can be more effective,” she said.
In an observational study of 27 women with refractory melasma, with phototypes II-V, New York City–based dermatologist Arielle Kauvar, MD, combined microdermabrasion with the Q-switched Nd:YAG laser. “The settings she used were very low fluence, so there was no clinical endpoint or no whitening,” said Dr. Ortiz, vice president of the American Society for Laser Medicine and Surgery (ASLMS). Specifically, Dr. Kauvar used the laser at 1.6-2 J/cm2 with a 5- or 6-mm spot size immediately following microdermabrasion every 4 weeks; Patients received an average of 2.6 treatments, and were assessed 3-12 months after the last treatment. Study participants were on a standard skin care regimen of a broad spectrum sunscreen, hydroquinone, and tretinoin or vitamin C.
Most of the patients showed at least 50% clearance of melasma 1 month after the first treatment, and 81% showed more than 75% clearance of melasma; remission lasted at least 6 months.
“I personally prefer to use picosecond over Q-switched lasers, because they deliver the energy faster, and you can use a 1,064-nm picosecond laser that is safe in all skin types,” Dr. Ortiz said. “There is minimal downtime, and it doesn’t require anesthesia. You have to consider these things when you’re treating melasma, because this usually requires monthly treatments. If you do something that requires a week of downtime every month, it’s not practical for patients.”
In a study published in 2021, Dr. Ortiz and Tanya Greywal, MD, used three passes of the 1,064-nm Nd:YAG laser to treat melasma in 10 patients with skin types II-V. The device had a 650-microsecond pulse duration, a 6-mm spot size, and an energy mode of 11-14 J/cm2. The researchers observed a mean melasma improvement of 26%-50% as early as 3 weeks. “There was no downtime, and no anesthesia was required,” Dr. Ortiz said.
Researchers have discovered a vascular component to melasma, which may have treatment implications. Houston-based dermatologist Paul M. Friedman, MD, and his colleagues used spectrocolorimetry to detect an underlying prominent vascular component in a retrospective review of 11 patients with melasma, with skin types II-IV. They determined that melasma lesions exhibiting subtle or subclinical telangiectatic erythema may be improved by combining vascular-targeted laser therapy with fractional low-powered diode laser therapy.
“So, combining a vascular laser with a 1,927-nm fractional diode laser showed more improvement than with just the diode laser alone,” said Dr. Ortiz, who was not involved with the analysis.
To optimize results following the laser treatment of melasma, she uses one application of clobetasol immediately after the procedure. “This can help reduce swelling and inflammation to decrease the risk of postinflammatory hyperpigmentation,” she said. “You can also use a skin cooling system like Cryomodulation for controlled cooling.”
Tranexamic acid and PLE
Another strategy for melasma patients involves oral treatment with extract of Polypodium leucotomos (PLE), a fern from the Polypodiaceae family with antioxidant properties that has been shown to be photoprotective against UVA and UVB radiation. “I explain to my patients that it’s like an internal sunscreen,” Dr. Ortiz said. “It does not replace your external sunscreen, but it adds extra protection.”
In a pilot placebo-controlled study of patients with melasma on their normal regimen of hydroquinone and sunscreen, 40 Asian patients with melasma were randomized to receive either oral PLE supplementation or placebo for 12 weeks. The authors found that PLE significantly improved and accelerated the outcome reached with hydroquinone and sunscreen from about the first month of treatment, compared with placebo.
Dr. Ortiz discussed the role of oral tranexamic acid, an antifibrinolytic, procoagulant agent that is approved by the Food and Drug Administration for the treatment of menorrhagia and to prevent hemorrhage in patients with hemophilia undergoing tooth extractions. “This is a game changer for melasma treatment,” she said, but its use has been limited by the risk for thromboembolism.
In a study of 561 patients with melasma, 90% improved after a median treatment duration of 4 months, and only 7% had side effects, most commonly abdominal bloating and pain. Treatment was discontinued in one patient who developed a deep vein thrombosis, and was diagnosed with familial protein S deficiency.
The daily dosing of tranexamic acid for menorrhagia is 3,900 mg daily, while the dose for treating melasma has ranged from 500 mg to 1,500 mg per day, Dr. Ortiz said. It’s available as a 650-mg tablet in the United States. “I prescribe 325 mg twice a day, but studies have shown that 650 mg once a day is just as effective,” she said.
Prior to prescribing tranexamic acid, Dr. Ortiz does not order labs, but she performs an extensive history of current illness and does not prescribe it in patients with an increased risk of clotting, including people who smoke and those who take oral contraceptives or are on hormone supplementation. Use is also contraindicated in people with a current malignancy, those with a history of stroke or DVT, and those who have any clotting disorder.
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies. She is cochair of the Masters of Aesthetics Symposium.
SAN DIEGO –
“They need to understand that melasma is going to require long-term maintenance,” Dr. Ortiz, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium.
Hydroquinone is a mainstay of melasma therapy, but instead of the commonly used 4% formulation, she prefers to use 12% hydroquinone with 6% kojic acid in VersaBase cream. “It’s a high concentration but the VersaBase makes it more tolerable,” she said. “I have patients take a pea-sized amount and mix it in a regular moisturizer. It’s too strong to spot treat, so it goes on the whole face.”
Mindful that chronic hydroquinone use can cause ochronosis (permanent darkening), she has patients alternate with a nonhydroquinone bleaching agent such as lignin peroxidase, oligopeptide, Lytera, Melaplex, 4-n-butylresorcinol, Cysteamine cream, tranexamic acid, or oral antioxidants. In a study sponsored by SkinMedica, investigators conducted a randomized, double-blind, half-face study in females with moderate to severe facial hyperpigmentation to assess the efficacy and tolerability of three new skin brightener formulations containing SMA-432, a prostaglandin E2 inhibitor, compared with 4% hydroquinone. They found that the nonhydroquinone skin formulations were better tolerated and were just as effective as 4% hydroquinone.
Chemical peels and laser treatments
Chemical peels are another treatment option for melasma, but Dr. Ortiz prefers glycolic peels over salicylic and other peels, “because there is no downtime,” she said.
As for laser-based approaches, melasma patients respond best to low energy devices such as the 1,927-nm fractional diode laser at a 3.75% density. “This also can increase the skin permeability of topicals, so when you’re combining it with hydroquinone it can be more effective,” she said.
In an observational study of 27 women with refractory melasma, with phototypes II-V, New York City–based dermatologist Arielle Kauvar, MD, combined microdermabrasion with the Q-switched Nd:YAG laser. “The settings she used were very low fluence, so there was no clinical endpoint or no whitening,” said Dr. Ortiz, vice president of the American Society for Laser Medicine and Surgery (ASLMS). Specifically, Dr. Kauvar used the laser at 1.6-2 J/cm2 with a 5- or 6-mm spot size immediately following microdermabrasion every 4 weeks; Patients received an average of 2.6 treatments, and were assessed 3-12 months after the last treatment. Study participants were on a standard skin care regimen of a broad spectrum sunscreen, hydroquinone, and tretinoin or vitamin C.
Most of the patients showed at least 50% clearance of melasma 1 month after the first treatment, and 81% showed more than 75% clearance of melasma; remission lasted at least 6 months.
“I personally prefer to use picosecond over Q-switched lasers, because they deliver the energy faster, and you can use a 1,064-nm picosecond laser that is safe in all skin types,” Dr. Ortiz said. “There is minimal downtime, and it doesn’t require anesthesia. You have to consider these things when you’re treating melasma, because this usually requires monthly treatments. If you do something that requires a week of downtime every month, it’s not practical for patients.”
In a study published in 2021, Dr. Ortiz and Tanya Greywal, MD, used three passes of the 1,064-nm Nd:YAG laser to treat melasma in 10 patients with skin types II-V. The device had a 650-microsecond pulse duration, a 6-mm spot size, and an energy mode of 11-14 J/cm2. The researchers observed a mean melasma improvement of 26%-50% as early as 3 weeks. “There was no downtime, and no anesthesia was required,” Dr. Ortiz said.
Researchers have discovered a vascular component to melasma, which may have treatment implications. Houston-based dermatologist Paul M. Friedman, MD, and his colleagues used spectrocolorimetry to detect an underlying prominent vascular component in a retrospective review of 11 patients with melasma, with skin types II-IV. They determined that melasma lesions exhibiting subtle or subclinical telangiectatic erythema may be improved by combining vascular-targeted laser therapy with fractional low-powered diode laser therapy.
“So, combining a vascular laser with a 1,927-nm fractional diode laser showed more improvement than with just the diode laser alone,” said Dr. Ortiz, who was not involved with the analysis.
To optimize results following the laser treatment of melasma, she uses one application of clobetasol immediately after the procedure. “This can help reduce swelling and inflammation to decrease the risk of postinflammatory hyperpigmentation,” she said. “You can also use a skin cooling system like Cryomodulation for controlled cooling.”
Tranexamic acid and PLE
Another strategy for melasma patients involves oral treatment with extract of Polypodium leucotomos (PLE), a fern from the Polypodiaceae family with antioxidant properties that has been shown to be photoprotective against UVA and UVB radiation. “I explain to my patients that it’s like an internal sunscreen,” Dr. Ortiz said. “It does not replace your external sunscreen, but it adds extra protection.”
In a pilot placebo-controlled study of patients with melasma on their normal regimen of hydroquinone and sunscreen, 40 Asian patients with melasma were randomized to receive either oral PLE supplementation or placebo for 12 weeks. The authors found that PLE significantly improved and accelerated the outcome reached with hydroquinone and sunscreen from about the first month of treatment, compared with placebo.
Dr. Ortiz discussed the role of oral tranexamic acid, an antifibrinolytic, procoagulant agent that is approved by the Food and Drug Administration for the treatment of menorrhagia and to prevent hemorrhage in patients with hemophilia undergoing tooth extractions. “This is a game changer for melasma treatment,” she said, but its use has been limited by the risk for thromboembolism.
In a study of 561 patients with melasma, 90% improved after a median treatment duration of 4 months, and only 7% had side effects, most commonly abdominal bloating and pain. Treatment was discontinued in one patient who developed a deep vein thrombosis, and was diagnosed with familial protein S deficiency.
The daily dosing of tranexamic acid for menorrhagia is 3,900 mg daily, while the dose for treating melasma has ranged from 500 mg to 1,500 mg per day, Dr. Ortiz said. It’s available as a 650-mg tablet in the United States. “I prescribe 325 mg twice a day, but studies have shown that 650 mg once a day is just as effective,” she said.
Prior to prescribing tranexamic acid, Dr. Ortiz does not order labs, but she performs an extensive history of current illness and does not prescribe it in patients with an increased risk of clotting, including people who smoke and those who take oral contraceptives or are on hormone supplementation. Use is also contraindicated in people with a current malignancy, those with a history of stroke or DVT, and those who have any clotting disorder.
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies. She is cochair of the Masters of Aesthetics Symposium.
AT MOAS 2022