User login
How to help runners steer clear of injury
Approximately 60 million people in the United States run for exercise at least once a calendar year, with approximately 11 million of them running > 100 days a year.1,2 Running is an affordable, convenient, and efficient form of exercise, whose benefits include a decrease in the risk of all-cause early mortality, cancer, and diabetes; an improved lipid profile; and better mental health.3
However, running is also the cause of a significant percentage of exercise-associated injuries: More than 60% of runners report overuse injury annually.4 Given the high incidence of running-related injury, an important component of primary care is accurately diagnosing and managing such injuries and counseling patients about how to prevent them.

This article reviews risk factors for running-related injury and summarizes evidence-based recommendations for prevention.
CASE
During a health maintenance examination, Clara K, a 47-year-old woman who is obese (body mass index [BMI], 34) and has bilateral knee osteoarthritis (OA), inquires about establishing a weight-loss strategy. Ms. K is interested in starting an exercise regimen involving running but is worried about provoking a flare of OA pain.
Risk factors for running injuries
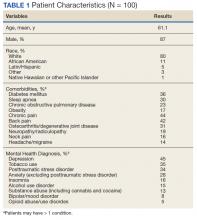
Several risk factors—some modifiable, others nonmodifiable—are associated with running-related injury (TABLE 14-16). In addition, research suggests that other variables once thought to be risk factors, such as running surface and the Q-angle (described later), are not associated with running-related injury.
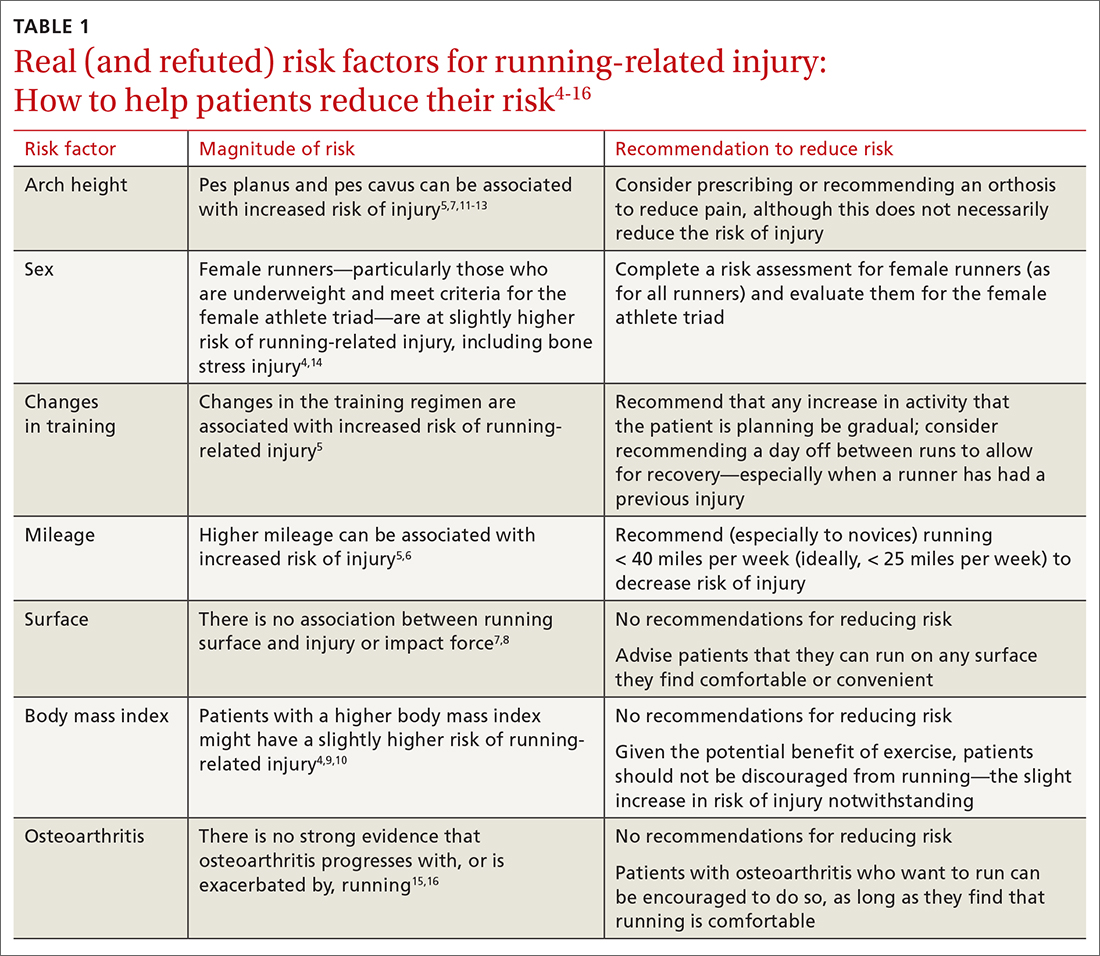
Modifiable risk factors
Changes in a training regimen or type of training. Many runners escalate training regimens as their fitness improves. Increasing mileage and changing the type of training (such as introducing hills or interval training) are independent risk factors for sustaining injury.5
The traditional recommendation has been for a runner to slowly increase or modify training with a 10% weekly increase in mileage or intensity.17 However, a randomized controlled trial failed to show a lower incidence of injury among amateur runners who adopted a graded exercise program.18 Regardless: It is still prudent to recommend a gradual increase in activity, such as taking ≥ 1 day off between running workouts or starting with a walking or jogging program, especially when there is a history of injury.19
Continue to: Excessive mileage
Excessive mileage. Many runners aspire to complete high-mileage runs. There is low-quality evidence demonstrating that high-mileage running, especially > 40 miles per week, is associated with increased risk of running-related injury.5 Injuries that occur with higher mileage are more often those of the hip and hamstring.5 A study noted that running ≤ 25 miles a week was protective against calf injury.6
Overall, there is little evidence to show that high-mileage running is associated with increased risk of running-related injury. However, this is still a risk factor that you should address with patients who have a running program—especially novices and those who ramp up mileage quickly.
Type of surface. Access to running surfaces—concrete, pavement, trails, treadmills, and athletic tracks—varies by time of day and season. Softer surfaces include treadmill, tracks, and trails; harder surfaces include asphalt and concrete.
There are limited data linking running surface with risk of injury.7 A study did not find an association between peak impact force based on running surface8; the authors hypothesized that runners compensate for a harder surface by making kinematic adjustments to minimize impact. With no strong evidence to link running-related injury to a particular running surface, patients should not be restricted to a softer running surface unless they notice a difference in comfort, because it is likely that they can compensate for a harder surface by adapting their gait.
Patients can therefore be counseled to run locally on sidewalks and neighborhood streets—if safe to do so—instead of obtaining a gym membership or driving to run on a trail. Such reassurance can increase a patient’s access to running and reduce barriers to exercise.
Continue to: BMI
BMI. Elevated BMI increases joint contact forces, which might increase risk of pain and injury.20 Results of studies investigating the link between BMI and running injury are mixed; some report that, in regard to bone stress injury, overweight BMI (> 25) is a risk factor for male runners and underweight BMI (< 18.5) is a risk factor for female runners.4,6 An observational study concluded that, among half-marathon and marathon runners, there was no significant increase in race-related injury, based on BMI.9 However, another study showed a higher rate of running-related injury in novice runners who had a higher BMI.10 A prospective cohort study found that runners with a higher BMI reported increased knee stiffness, which can place a runner at higher risk of overuse injury.4
Although these results conflict, there is consistency in the finding that obese novice runners are likely at increased risk of running-related injury; it is reasonable, therefore, for you to discuss strategies to reduce the risk of other modifiable factors, especially among obese novice runners. Patients with a higher BMI should not be discouraged from running, because exercise in combination with healthy eating habits is essential to decrease the myriad adverse health outcomes associated with obesity.
Female runners with a lower BMI, especially in the presence of other components of the female athlete triad (inadequate nutrition, amenorrhea, and low bone density), should be counseled about their increased risk of bone stress injury.21 Notably, a study of female US Navy recruits randomized to receive a trial of dietary supplementation of vitamin D plus calcium, or placebo, showed a 21% lower incidence of bone stress injury in the active-treatment group.22 To mitigate risk of injury associated with low BMI and the female athlete triad, therefore, a multidisciplinary approach of nutrition intervention, dietary optimization of vitamin D and calcium, and, possibly, activity modification should be implemented when appropriate.
Running gait. A study using 2-dimensional gait analysis to visualize biomechanical running patterns in injured and noninjured runners found that, in regard to mechanical variables, running-related injury was most strongly associated with contralateral pelvic drop.23 Gait retraining can be employed to help decrease contralateral pelvic drop.24 In addition, pelvic drop is often a result of weak gluteal muscles, and can be improved by doing strengthening exercises at home or with physical therapy.
Longer stride is also associated with running-related injury.25 A study showed improvement in patellofemoral pain by having runners increase stride rate by 10%, which reduces stride length to a significant degree.25,26 These improvements were maintained at 1-month and 3-month follow-up, and required only 1 gait retraining session.
Continue to: Get analysis is not feasible...
Gait analysis is not feasible in most primary care clinics. Instead, patients who run and (1) in whom pain persists despite more traditional treatments and (2) who have had recurring injury should be referred to a gait lab for analysis, usually by a physical therapist.
Nonmodifiable risk factors
Arch height. A high arch (pes cavus) is associated with increased risk of running-related injury, including bone stress injury, Achilles tendinopathy, plantar fasciitis, and patellofemoral pain syndrome.5 The mechanism of injury is thought to be increased forefoot loading forces.1
A review article showed that patients with pes cavus have reduced pain when using an orthosis, although there is no associated decrease in the risk of injury.5 To the contrary, a prospective study concluded that arch height was unrelated to increased risk of running-related injury.7
Evidence regarding flat feet (pes planus) and risk of injury is also mixed. Some studies show that pes planus is not associated with increased risk of injury in athletes.12 A cross-sectional study in older patients showed those with pes planus morphology had a higher rate of knee pain and wearing away of medial compartment cartilage.13 Because this study comprised only older adults, it is not generalizable to runners—nor can conclusions be drawn about causation, given the cross-sectional nature of the study.
Although a foot orthosis can correct mechanical differences caused by pes planus morphology, there is not enough evidence to conclude that correction results in a lower rate of injury. In sum, data are mixed with regard to arch height as a risk factor for running-related injury.
Continue to: Patients with...
Patients with pes planus or pes cavus should not be discouraged from running, however. If they experience pain with running, they might benefit from a trial of arch support inserts; or consider referral to an orthotist for evaluation for a custom orthosis.
Sex. Based on a prospective cohort study, female runners have a slightly higher rate of running injury than male counterparts.4 Similarly, a study showed that female military members generally had a higher incidence of stress fractures than male military members—specifically, femoral shaft and neck stress fractures.14 Runners who fall in the spectrum of the female athlete triad, as described earlier, are particularly vulnerable to bone stress injury. It is reasonable, therefore, to review risk factors for injury with female runners (as it is with all runners), especially those who have sustained a prior running-related injury.
Increased Q-angle (an obsolete risk factor). The Q-angle is approximated by drawing a line from the anterior superior iliac spine to the patella and a second line from the patella to the tibial tubercle. In males, a normal Q-angle is 14°; in females, 17° (SD = 4.5°). The Q-angle can be obtained by goniometric or radiographic measurement.
An increased Q-angle had been considered an intrinsic risk factor for running injury but has not been shown to be associated with increased risk of running-related injury or patellofemoral pain syndrome.27,28 Because the Q-angle is not a clinically relevant tool in assessing risk of injury, do not routinely measure it or include it in risk-factor counseling.
OA. Based on a systematic review of observational studies, data are inconclusive with regard to whether running contributes to, or is protective against, knee OA.15 In a large cohort study, running (1) was protective against development of hip OA and (2) decreased the risk of requiring hip replacement.29 This finding was supported by animal-model research that concluded that it is inactivity that results in thinning of articular cartilage.29 In addition, a systematic review of randomized controlled trials concluded that knee joint-loading exercises are not harmful to articular cartilage (this is low-quality evidence, however).16
Continue to: Given that there...
Given that there are no high-quality studies suggesting that running contributes to or exacerbates OA, patients with OA can be counseled to start or continue running as tolerated because the health benefit of running likely outweighs risk. Patients with pre-existing moderate-to-severe OA might report knee and hip pain that is already exacerbated by certain activities; if a high-impact activity, such as running, makes that pain worse, exercise counseling that you provide can be tailored to include lower-impact alternatives, such as swimming, cycling, or an elliptical workout.
CASE
In response to Ms. K’s interest in beginning an exercise regimen that includes running, you perform a complete routine pre-participation evaluation and appropriate cardiac screening. You discuss risk factors for running injury, focusing on modifiable risk factors.
Ms. K is perimenopausal but reports a history of regular menstrual cycles. She eats a relatively well-balanced diet. You advise that her BMI should not restrict her from incorporating running into her fitness regimen. Also, you reassure her that she should not restrict running based on a diagnosis of OA; instead, you advise her to monitor her symptoms and reconsider her program if running makes her knee pain worse.
At this point, Ms. K is ready to run. She tells you that, based on your guidance, she feels more comfortable and safe starting a running program.
Preventing injury
After reviewing risk factors for running-related injury with patients, encourage other evidence-based methods of reducing that risk.
Continue to: Shoes
Shoes
The running shoe industry offers a variety of running shoes, from minimalist shoes to cushioned stability shoes that vary based on the amount of cushioning, level of motion control, and amount of heel-to-toe drop. With so many options, new runners might wonder which shoes can reduce their risk of injury and how they should select a pair.
Stability. A characteristic of running shoes promoted by the industry is their stability: ie, their motion control. Stability shoes are marketed to runners who overpronate and therefore limit motion to prevent overpronation. The benefit of stability shoes, or stability insoles, is unclear.30 A randomized controlled trial showed that, in runners who overpronate, motion-control shoes reduced their risk of injury.31 However, another study assessed whether shoes that had been “prescribed” based on foot morphology and stride reduced the risk of injury (compared to neutral, cushioned shoes) and found no change in the incidence of soft-tissue injury.32 Given no strong evidence to suggest otherwise, runners can be advised to buy shoes based on comfort rather than on foot morphology or running stride.
Heel-to-toe drop. Another component of shoe variability is heel-to-toe drop (the height difference between heel and forefoot). A study suggests that moderate-to-high (8-12 mm) heel-to-toe drop is associated with a reduced risk of running injury.33 Barefoot running shoes, which, typically, have no heel-to-toe drop, are associated with increased risk of injury—specifically, foot stress fracture (especially in runners who are even moderately overweight).34,35
Shoe age and shoe wear can be modified to reduce injury. There is evidence that running shoes lose approximately 50% of cushioning after 300 to 500 miles of use.36 Another study found that rotating running shoes—ideally, different types or brands—can lead to fewer running-related injuries.37
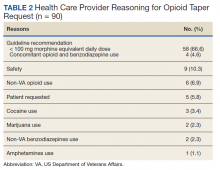
In general, patients can be counseled to use shoes that feel comfortable, as long as they replace them regularly (TABLE 2). Runners can also consider alternating pairs of different running shoes between runs. Overweight runners should avoid minimally cushioned and low heel-to-toe drop running shoes.

Continue to: Cross-training
Cross-training
Cross-training exercises for runners include cycling, an elliptical workout, swimming, and weightlifting. Incorporating cross-training can be protective against running injury because cross-training requires different movement patterns, prevents overuse, and equalizes muscle imbalances that occur with running.7 In addition, replacing running with a cross-training activity can decrease weekly running time and mileage, which can further reduce risk of running-related injury.7 Runners—especially higher-mileage runners—should be encouraged to incorporate cross-training into their workout regimen to decrease their risk of injury.
Stretching. The authors of a Cochrane review concluded that there is no significant reduction in injury associated with hamstring or gastrocnemius stretching.32 A small randomized, controlled, crossover study concluded that participants subjectively felt their performance was better when warm-ups included stretching.38 This perceived improvement in performance was similar between groups who completed dynamic or static stretching. However, no difference was noted in flexibility or objective performance between groups who stretched or did not stretch before activity.
Although there is no supporting evidence that stretching reduces the risk of injury, stretching is a low-risk intervention. Because stretching might provide subjective benefit to runners, you need not discourage patients from including this activity in their running program.
CORRESPONDENCE
Kartik Sidhar, MD, 15370 Huff Way, Brookfield, WI, 53005; kartiksidhar@gmail.com
1. Brown CR Jr. Common injuries from running. In: Imboden JB, Hellerman, DB, Stone JH, eds. Current Diagnosis & Treatment: Rheumatology. 3rd ed. McGraw-Hill; 2013.
2. Lange D. Running & jogging - statistic and facts. Statista Web site. November 16, 2020. Accessed March 28, 2021. www.statista.com/topics/1743/running-and-jogging/
3. Warburton DER, Bredin SSD. Health benefits of physical activity: a systematic review of current systematic reviews. Curr Opin Cardiol. 2017;32:541-556. doi:10.1097/HCO.0000000000000437
4. Messier SP, Martin DF, Mihalko SL, et al. A 2-year prospective cohort study of overuse running injuries: The Runners and Injury Longitudinal Study (TRAILS). Am J Sports Med. 2018;46:2211-2221. doi:10.1177/0363546518773755
5. Fields KB, Sykes JC, Walker KM, et al. Prevention of running injuries. Curr Sports Med Rep. 2010;9:176-182. doi:10.1249/JSR.0b013e3181de7ec5
6. van der Worp MP, ten Haaf DSM, van Cingel R. Injuries in runners; a systematic review on risk factors and sex differences. PLoS One. 2015;10:1-18. doi:10.1371/journal.pone.0114937
7. Taunton JE, Ryan MB, Clement DB, et al. A prospective study of running injuries: the Vancouver Sun Run “In Training” clinics. Br J Sports Med. 2003;37:239-244. doi:10.1136/bjsm.37.3.239
8. Dixon SJ, Collop AC, Batt ME. Surface effects on ground reaction forces and lower extremity kinematics in running. Med Sci Sports Exerc. 2000;32:1919-1926. doi:10.1097/00005768-200011000-00016
9. Vadeboncoeur TF, Silvers SM, Taylor WC, et al. Impact of a high body mass index on lower extremity injury in marathon/half-marathon participants. J Phys Act Health. 2012;9:96-103. doi:10.1123/jpah.9.1.96
10. Buist I, Bredeweg SW. Higher risk of injury in overweight novice runners. Br J Sports Med. 2011;45:338. http://dx.doi.org/10.1136/bjsm.2011.084038.79
11. Cowan DN, Jones BH, Robinson JR. Foot morphologic characteristics and risk of Exercise-related injury. Arch Fam Med. 1993;2:773-777. doi:10.1001/archfami.2.7.773
12. Michelson JD, Durant DM, McFarland E. The injury risk associated with pes planus in athletes. Foot Ankle Int. 2002;23:629-633. doi: 10.1177/107110070202300708
13. Gross KD, Felson DT, Niu J, et al. Association of flat feet with knee pain and cartilage damage in older adults. Arthritis Care Res (Hoboken). 2011;63:937-944. doi:10.1002/acr.20431
14. Waterman BR, Gun B, Bader JO, et al. Epidemiology of lower extremity stress fractures in the United States military. Mil Med. 2016;181:1308-1313. doi:10.7205/MILMED-D-15-00571
15. Timmins KA, Leech RD, Batt ME, et al. Running and knee osteoarthritis: a systematic review and meta-analysis. Am J Sports Med. 2017;45:1447-1457. doi:10.1177/0363546516657531
16. Bricca A, Juhl CB, Steultjens M, et al. Impact of exercise on articular cartilage in people at risk of, or with established, knee osteoarthritis: a systematic review of randomised controlled trials. Br J Sports Med. 2019;53:940-947. doi:10.1136/bjsports-2017-098661
17. Johnston CAM, Taunton JE, Lloyd-Smith DR, et al. Preventing running injuries. Practical approach for family doctors. Can Fam Physician. 2003;49:1101-1109.
18. Buist I, Bredeweg SW, van Mechelen W, et al. No effect of a graded training program on the number of running-related injuries in novice runners: a randomized controlled trial. Am J Sports Med. 2008;36:33-39. doi:10.1177/0363546507307505
19. Warden SJ, Davis IS, Fredericson M. Management and prevention of bone stress injuries in long-distance runners. J Orthop Sports Phys Ther. 2014;44:749-765. doi:10.2519/jospt.2014.5334
20. Kim N, Browning RC, Lerner ZF. The effects of pediatric obesity on patellofemoral joint contact force during walking. Gait Posture. 2019;73:209-214. doi:10.1016/j.gaitpost.2019.07.307
21. Tenforde AS, Kraus E, Fredericson M. Bone stress injuries in runners. Phys Med Rehabil Clin N Am. 2016;27:139-149. doi:10.1016/j.pmr.2015.08.008
22. Lappe J, Cullen D, Haynatzki G, et al. Calcium and vitamin D supplementation decreases incidence of stress fractures in female navy recruits. J Bone Miner Res. 2008;23:741-749. doi:10.1359/jbmr.080102
23. Bramah C, Preece SJ, Gill N, et al. Is there a pathological gait associated with common soft tissue running injuries? Am J Sports Med. 2018;46:3023-3031. doi:10.1177/0363546518793657
24. Willy RW, Scholz PT, Davis IS. Mirror gait retraining for the treatment of patellofemoral pain in female runners. Clin Biomech (Bristol Avon). 2012;27:1045-1051. doi:10.1016/j.clinbiomech.2012.07.011
25. Schubert AG, Kempf J, Heiderscheit BC. Influence of stride frequency and length on running mechanics: a systematic review. Sports Health. 2014;6:210-217. doi:10.1177/1941738113508544
26. Bramah C, Preece SJ, Gill N et al. A 10% increase in step rate improves running kinematics and clinical outcomes in runners with patellofemoral pain at 4 weeks and 3 months. Am J Sports Med. 2019;47:3406-3413. doi: 10.1177/0363546519879693
27. Ramskov D, Jensen ML, Obling K, et al. No association between q-angle and foot posture with running-related injuries: a 10 week prospective follow-up study. Int J Sports Phys Ther. 2013;8:407-415.
28. Almeida GPL, Silva AP, França FJR, et al. Q-angle in patellofemoral pain: relationship with dynamic knee valgus, hip abductor torque, pain and function. Rev Bras Ortop. 2016;51:181-186. doi:10.1016/j.rboe.2016.01.010
29. Williams PT. Effects of running and walking on osteoarthritis and hip replacement risk. Med Sci Sports Exerc. 2013;45:1292-1297. doi:10.1249/MSS.0b013e3182885f26
30. Nigg BM, Baltich J, Hoerzer S, et al. Running shoes and running injuries: mythbusting and a proposal for two new paradigms: ‘Preferred movement path’ and ‘comfort filter.’ Br J Sports Med. 2015;49:1290-1294. doi:10.1136/bjsports-2015-095054
31. Malisoux L, Chambon N, Delattre N, et al. Injury risk in runners using standard or motion control shoes: a randomised controlled trial with participant and assessor blinding. Br J Sports Med. 2016;50:481-487. doi:10.1136/bjsports-2015-095031
32. Yeung SS, Yeung EW, Gillespie LD. Interventions for preventing lower limb soft-tissue running injuries. Cochrane Database Syst Rev. 2011(7):CD001256. doi:10.1002/14651858.cd001256.pub2
33. Malisoux L, Chambon N, Urhausen A, et al. Influence of the heel-to-toe drop of standard cushioned running shoes on injury risk in leisure-time runners: a randomized controlled trial with 6-month follow-up. Am J Sports Med. 2016;44:2933-2940. doi:10.1177/0363546516654690
34. Ryan M, Elashi M, Newsham-West R, et al. Examining injury risk and pain perception in runners using minimalist footwear. Br J Sports Med. 2014;48:1257-1262. doi:10.1136/bjsports-2012-092061
35. Fuller JT, Thewlis D, Buckley JD, et al.. Body mass and weekly training distance influence the pain and injuries experienced by runners using minimalist shoes: a randomized controlled trial. Am J Sports Med. 2017;45:1162-1170. doi:10.1177/0363546516682497
36. Cook SD, Kester MA, Brunet ME. Shock absorption characteristics of running shoes. Am J Sports Med. 1985;13:248-253. doi.org/10.1177/036354658501300406
37. Malisoux L, Ramesh J, Mann R, et al. Can parallel use of different running shoes decrease running-related injury risk? Scand J Med Sci Sport. 2015;25:110-115. doi:10.1111/sms.12154
38. Blazevich AJ, Gill ND, Kvorning T, et al. No effect of muscle stretching within a full, dynamic warm-up on athletic performance. Med Sci Sports Exerc. 2018;50:1258-1266. doi:10.1249/MSS.0000000000001539
Approximately 60 million people in the United States run for exercise at least once a calendar year, with approximately 11 million of them running > 100 days a year.1,2 Running is an affordable, convenient, and efficient form of exercise, whose benefits include a decrease in the risk of all-cause early mortality, cancer, and diabetes; an improved lipid profile; and better mental health.3
However, running is also the cause of a significant percentage of exercise-associated injuries: More than 60% of runners report overuse injury annually.4 Given the high incidence of running-related injury, an important component of primary care is accurately diagnosing and managing such injuries and counseling patients about how to prevent them.

This article reviews risk factors for running-related injury and summarizes evidence-based recommendations for prevention.
CASE
During a health maintenance examination, Clara K, a 47-year-old woman who is obese (body mass index [BMI], 34) and has bilateral knee osteoarthritis (OA), inquires about establishing a weight-loss strategy. Ms. K is interested in starting an exercise regimen involving running but is worried about provoking a flare of OA pain.
Risk factors for running injuries
Several risk factors—some modifiable, others nonmodifiable—are associated with running-related injury (TABLE 14-16). In addition, research suggests that other variables once thought to be risk factors, such as running surface and the Q-angle (described later), are not associated with running-related injury.

Modifiable risk factors
Changes in a training regimen or type of training. Many runners escalate training regimens as their fitness improves. Increasing mileage and changing the type of training (such as introducing hills or interval training) are independent risk factors for sustaining injury.5
The traditional recommendation has been for a runner to slowly increase or modify training with a 10% weekly increase in mileage or intensity.17 However, a randomized controlled trial failed to show a lower incidence of injury among amateur runners who adopted a graded exercise program.18 Regardless: It is still prudent to recommend a gradual increase in activity, such as taking ≥ 1 day off between running workouts or starting with a walking or jogging program, especially when there is a history of injury.19
Continue to: Excessive mileage
Excessive mileage. Many runners aspire to complete high-mileage runs. There is low-quality evidence demonstrating that high-mileage running, especially > 40 miles per week, is associated with increased risk of running-related injury.5 Injuries that occur with higher mileage are more often those of the hip and hamstring.5 A study noted that running ≤ 25 miles a week was protective against calf injury.6
Overall, there is little evidence to show that high-mileage running is associated with increased risk of running-related injury. However, this is still a risk factor that you should address with patients who have a running program—especially novices and those who ramp up mileage quickly.
Type of surface. Access to running surfaces—concrete, pavement, trails, treadmills, and athletic tracks—varies by time of day and season. Softer surfaces include treadmill, tracks, and trails; harder surfaces include asphalt and concrete.
There are limited data linking running surface with risk of injury.7 A study did not find an association between peak impact force based on running surface8; the authors hypothesized that runners compensate for a harder surface by making kinematic adjustments to minimize impact. With no strong evidence to link running-related injury to a particular running surface, patients should not be restricted to a softer running surface unless they notice a difference in comfort, because it is likely that they can compensate for a harder surface by adapting their gait.
Patients can therefore be counseled to run locally on sidewalks and neighborhood streets—if safe to do so—instead of obtaining a gym membership or driving to run on a trail. Such reassurance can increase a patient’s access to running and reduce barriers to exercise.
Continue to: BMI
BMI. Elevated BMI increases joint contact forces, which might increase risk of pain and injury.20 Results of studies investigating the link between BMI and running injury are mixed; some report that, in regard to bone stress injury, overweight BMI (> 25) is a risk factor for male runners and underweight BMI (< 18.5) is a risk factor for female runners.4,6 An observational study concluded that, among half-marathon and marathon runners, there was no significant increase in race-related injury, based on BMI.9 However, another study showed a higher rate of running-related injury in novice runners who had a higher BMI.10 A prospective cohort study found that runners with a higher BMI reported increased knee stiffness, which can place a runner at higher risk of overuse injury.4
Although these results conflict, there is consistency in the finding that obese novice runners are likely at increased risk of running-related injury; it is reasonable, therefore, for you to discuss strategies to reduce the risk of other modifiable factors, especially among obese novice runners. Patients with a higher BMI should not be discouraged from running, because exercise in combination with healthy eating habits is essential to decrease the myriad adverse health outcomes associated with obesity.
Female runners with a lower BMI, especially in the presence of other components of the female athlete triad (inadequate nutrition, amenorrhea, and low bone density), should be counseled about their increased risk of bone stress injury.21 Notably, a study of female US Navy recruits randomized to receive a trial of dietary supplementation of vitamin D plus calcium, or placebo, showed a 21% lower incidence of bone stress injury in the active-treatment group.22 To mitigate risk of injury associated with low BMI and the female athlete triad, therefore, a multidisciplinary approach of nutrition intervention, dietary optimization of vitamin D and calcium, and, possibly, activity modification should be implemented when appropriate.
Running gait. A study using 2-dimensional gait analysis to visualize biomechanical running patterns in injured and noninjured runners found that, in regard to mechanical variables, running-related injury was most strongly associated with contralateral pelvic drop.23 Gait retraining can be employed to help decrease contralateral pelvic drop.24 In addition, pelvic drop is often a result of weak gluteal muscles, and can be improved by doing strengthening exercises at home or with physical therapy.
Longer stride is also associated with running-related injury.25 A study showed improvement in patellofemoral pain by having runners increase stride rate by 10%, which reduces stride length to a significant degree.25,26 These improvements were maintained at 1-month and 3-month follow-up, and required only 1 gait retraining session.
Continue to: Get analysis is not feasible...
Gait analysis is not feasible in most primary care clinics. Instead, patients who run and (1) in whom pain persists despite more traditional treatments and (2) who have had recurring injury should be referred to a gait lab for analysis, usually by a physical therapist.
Nonmodifiable risk factors
Arch height. A high arch (pes cavus) is associated with increased risk of running-related injury, including bone stress injury, Achilles tendinopathy, plantar fasciitis, and patellofemoral pain syndrome.5 The mechanism of injury is thought to be increased forefoot loading forces.1
A review article showed that patients with pes cavus have reduced pain when using an orthosis, although there is no associated decrease in the risk of injury.5 To the contrary, a prospective study concluded that arch height was unrelated to increased risk of running-related injury.7
Evidence regarding flat feet (pes planus) and risk of injury is also mixed. Some studies show that pes planus is not associated with increased risk of injury in athletes.12 A cross-sectional study in older patients showed those with pes planus morphology had a higher rate of knee pain and wearing away of medial compartment cartilage.13 Because this study comprised only older adults, it is not generalizable to runners—nor can conclusions be drawn about causation, given the cross-sectional nature of the study.
Although a foot orthosis can correct mechanical differences caused by pes planus morphology, there is not enough evidence to conclude that correction results in a lower rate of injury. In sum, data are mixed with regard to arch height as a risk factor for running-related injury.
Continue to: Patients with...
Patients with pes planus or pes cavus should not be discouraged from running, however. If they experience pain with running, they might benefit from a trial of arch support inserts; or consider referral to an orthotist for evaluation for a custom orthosis.
Sex. Based on a prospective cohort study, female runners have a slightly higher rate of running injury than male counterparts.4 Similarly, a study showed that female military members generally had a higher incidence of stress fractures than male military members—specifically, femoral shaft and neck stress fractures.14 Runners who fall in the spectrum of the female athlete triad, as described earlier, are particularly vulnerable to bone stress injury. It is reasonable, therefore, to review risk factors for injury with female runners (as it is with all runners), especially those who have sustained a prior running-related injury.
Increased Q-angle (an obsolete risk factor). The Q-angle is approximated by drawing a line from the anterior superior iliac spine to the patella and a second line from the patella to the tibial tubercle. In males, a normal Q-angle is 14°; in females, 17° (SD = 4.5°). The Q-angle can be obtained by goniometric or radiographic measurement.
An increased Q-angle had been considered an intrinsic risk factor for running injury but has not been shown to be associated with increased risk of running-related injury or patellofemoral pain syndrome.27,28 Because the Q-angle is not a clinically relevant tool in assessing risk of injury, do not routinely measure it or include it in risk-factor counseling.
OA. Based on a systematic review of observational studies, data are inconclusive with regard to whether running contributes to, or is protective against, knee OA.15 In a large cohort study, running (1) was protective against development of hip OA and (2) decreased the risk of requiring hip replacement.29 This finding was supported by animal-model research that concluded that it is inactivity that results in thinning of articular cartilage.29 In addition, a systematic review of randomized controlled trials concluded that knee joint-loading exercises are not harmful to articular cartilage (this is low-quality evidence, however).16
Continue to: Given that there...
Given that there are no high-quality studies suggesting that running contributes to or exacerbates OA, patients with OA can be counseled to start or continue running as tolerated because the health benefit of running likely outweighs risk. Patients with pre-existing moderate-to-severe OA might report knee and hip pain that is already exacerbated by certain activities; if a high-impact activity, such as running, makes that pain worse, exercise counseling that you provide can be tailored to include lower-impact alternatives, such as swimming, cycling, or an elliptical workout.
CASE
In response to Ms. K’s interest in beginning an exercise regimen that includes running, you perform a complete routine pre-participation evaluation and appropriate cardiac screening. You discuss risk factors for running injury, focusing on modifiable risk factors.
Ms. K is perimenopausal but reports a history of regular menstrual cycles. She eats a relatively well-balanced diet. You advise that her BMI should not restrict her from incorporating running into her fitness regimen. Also, you reassure her that she should not restrict running based on a diagnosis of OA; instead, you advise her to monitor her symptoms and reconsider her program if running makes her knee pain worse.
At this point, Ms. K is ready to run. She tells you that, based on your guidance, she feels more comfortable and safe starting a running program.
Preventing injury
After reviewing risk factors for running-related injury with patients, encourage other evidence-based methods of reducing that risk.
Continue to: Shoes
Shoes
The running shoe industry offers a variety of running shoes, from minimalist shoes to cushioned stability shoes that vary based on the amount of cushioning, level of motion control, and amount of heel-to-toe drop. With so many options, new runners might wonder which shoes can reduce their risk of injury and how they should select a pair.
Stability. A characteristic of running shoes promoted by the industry is their stability: ie, their motion control. Stability shoes are marketed to runners who overpronate and therefore limit motion to prevent overpronation. The benefit of stability shoes, or stability insoles, is unclear.30 A randomized controlled trial showed that, in runners who overpronate, motion-control shoes reduced their risk of injury.31 However, another study assessed whether shoes that had been “prescribed” based on foot morphology and stride reduced the risk of injury (compared to neutral, cushioned shoes) and found no change in the incidence of soft-tissue injury.32 Given no strong evidence to suggest otherwise, runners can be advised to buy shoes based on comfort rather than on foot morphology or running stride.
Heel-to-toe drop. Another component of shoe variability is heel-to-toe drop (the height difference between heel and forefoot). A study suggests that moderate-to-high (8-12 mm) heel-to-toe drop is associated with a reduced risk of running injury.33 Barefoot running shoes, which, typically, have no heel-to-toe drop, are associated with increased risk of injury—specifically, foot stress fracture (especially in runners who are even moderately overweight).34,35
Shoe age and shoe wear can be modified to reduce injury. There is evidence that running shoes lose approximately 50% of cushioning after 300 to 500 miles of use.36 Another study found that rotating running shoes—ideally, different types or brands—can lead to fewer running-related injuries.37
In general, patients can be counseled to use shoes that feel comfortable, as long as they replace them regularly (TABLE 2). Runners can also consider alternating pairs of different running shoes between runs. Overweight runners should avoid minimally cushioned and low heel-to-toe drop running shoes.

Continue to: Cross-training
Cross-training
Cross-training exercises for runners include cycling, an elliptical workout, swimming, and weightlifting. Incorporating cross-training can be protective against running injury because cross-training requires different movement patterns, prevents overuse, and equalizes muscle imbalances that occur with running.7 In addition, replacing running with a cross-training activity can decrease weekly running time and mileage, which can further reduce risk of running-related injury.7 Runners—especially higher-mileage runners—should be encouraged to incorporate cross-training into their workout regimen to decrease their risk of injury.
Stretching. The authors of a Cochrane review concluded that there is no significant reduction in injury associated with hamstring or gastrocnemius stretching.32 A small randomized, controlled, crossover study concluded that participants subjectively felt their performance was better when warm-ups included stretching.38 This perceived improvement in performance was similar between groups who completed dynamic or static stretching. However, no difference was noted in flexibility or objective performance between groups who stretched or did not stretch before activity.
Although there is no supporting evidence that stretching reduces the risk of injury, stretching is a low-risk intervention. Because stretching might provide subjective benefit to runners, you need not discourage patients from including this activity in their running program.
CORRESPONDENCE
Kartik Sidhar, MD, 15370 Huff Way, Brookfield, WI, 53005; kartiksidhar@gmail.com
Approximately 60 million people in the United States run for exercise at least once a calendar year, with approximately 11 million of them running > 100 days a year.1,2 Running is an affordable, convenient, and efficient form of exercise, whose benefits include a decrease in the risk of all-cause early mortality, cancer, and diabetes; an improved lipid profile; and better mental health.3
However, running is also the cause of a significant percentage of exercise-associated injuries: More than 60% of runners report overuse injury annually.4 Given the high incidence of running-related injury, an important component of primary care is accurately diagnosing and managing such injuries and counseling patients about how to prevent them.

This article reviews risk factors for running-related injury and summarizes evidence-based recommendations for prevention.
CASE
During a health maintenance examination, Clara K, a 47-year-old woman who is obese (body mass index [BMI], 34) and has bilateral knee osteoarthritis (OA), inquires about establishing a weight-loss strategy. Ms. K is interested in starting an exercise regimen involving running but is worried about provoking a flare of OA pain.
Risk factors for running injuries
Several risk factors—some modifiable, others nonmodifiable—are associated with running-related injury (TABLE 14-16). In addition, research suggests that other variables once thought to be risk factors, such as running surface and the Q-angle (described later), are not associated with running-related injury.

Modifiable risk factors
Changes in a training regimen or type of training. Many runners escalate training regimens as their fitness improves. Increasing mileage and changing the type of training (such as introducing hills or interval training) are independent risk factors for sustaining injury.5
The traditional recommendation has been for a runner to slowly increase or modify training with a 10% weekly increase in mileage or intensity.17 However, a randomized controlled trial failed to show a lower incidence of injury among amateur runners who adopted a graded exercise program.18 Regardless: It is still prudent to recommend a gradual increase in activity, such as taking ≥ 1 day off between running workouts or starting with a walking or jogging program, especially when there is a history of injury.19
Continue to: Excessive mileage
Excessive mileage. Many runners aspire to complete high-mileage runs. There is low-quality evidence demonstrating that high-mileage running, especially > 40 miles per week, is associated with increased risk of running-related injury.5 Injuries that occur with higher mileage are more often those of the hip and hamstring.5 A study noted that running ≤ 25 miles a week was protective against calf injury.6
Overall, there is little evidence to show that high-mileage running is associated with increased risk of running-related injury. However, this is still a risk factor that you should address with patients who have a running program—especially novices and those who ramp up mileage quickly.
Type of surface. Access to running surfaces—concrete, pavement, trails, treadmills, and athletic tracks—varies by time of day and season. Softer surfaces include treadmill, tracks, and trails; harder surfaces include asphalt and concrete.
There are limited data linking running surface with risk of injury.7 A study did not find an association between peak impact force based on running surface8; the authors hypothesized that runners compensate for a harder surface by making kinematic adjustments to minimize impact. With no strong evidence to link running-related injury to a particular running surface, patients should not be restricted to a softer running surface unless they notice a difference in comfort, because it is likely that they can compensate for a harder surface by adapting their gait.
Patients can therefore be counseled to run locally on sidewalks and neighborhood streets—if safe to do so—instead of obtaining a gym membership or driving to run on a trail. Such reassurance can increase a patient’s access to running and reduce barriers to exercise.
Continue to: BMI
BMI. Elevated BMI increases joint contact forces, which might increase risk of pain and injury.20 Results of studies investigating the link between BMI and running injury are mixed; some report that, in regard to bone stress injury, overweight BMI (> 25) is a risk factor for male runners and underweight BMI (< 18.5) is a risk factor for female runners.4,6 An observational study concluded that, among half-marathon and marathon runners, there was no significant increase in race-related injury, based on BMI.9 However, another study showed a higher rate of running-related injury in novice runners who had a higher BMI.10 A prospective cohort study found that runners with a higher BMI reported increased knee stiffness, which can place a runner at higher risk of overuse injury.4
Although these results conflict, there is consistency in the finding that obese novice runners are likely at increased risk of running-related injury; it is reasonable, therefore, for you to discuss strategies to reduce the risk of other modifiable factors, especially among obese novice runners. Patients with a higher BMI should not be discouraged from running, because exercise in combination with healthy eating habits is essential to decrease the myriad adverse health outcomes associated with obesity.
Female runners with a lower BMI, especially in the presence of other components of the female athlete triad (inadequate nutrition, amenorrhea, and low bone density), should be counseled about their increased risk of bone stress injury.21 Notably, a study of female US Navy recruits randomized to receive a trial of dietary supplementation of vitamin D plus calcium, or placebo, showed a 21% lower incidence of bone stress injury in the active-treatment group.22 To mitigate risk of injury associated with low BMI and the female athlete triad, therefore, a multidisciplinary approach of nutrition intervention, dietary optimization of vitamin D and calcium, and, possibly, activity modification should be implemented when appropriate.
Running gait. A study using 2-dimensional gait analysis to visualize biomechanical running patterns in injured and noninjured runners found that, in regard to mechanical variables, running-related injury was most strongly associated with contralateral pelvic drop.23 Gait retraining can be employed to help decrease contralateral pelvic drop.24 In addition, pelvic drop is often a result of weak gluteal muscles, and can be improved by doing strengthening exercises at home or with physical therapy.
Longer stride is also associated with running-related injury.25 A study showed improvement in patellofemoral pain by having runners increase stride rate by 10%, which reduces stride length to a significant degree.25,26 These improvements were maintained at 1-month and 3-month follow-up, and required only 1 gait retraining session.
Continue to: Get analysis is not feasible...
Gait analysis is not feasible in most primary care clinics. Instead, patients who run and (1) in whom pain persists despite more traditional treatments and (2) who have had recurring injury should be referred to a gait lab for analysis, usually by a physical therapist.
Nonmodifiable risk factors
Arch height. A high arch (pes cavus) is associated with increased risk of running-related injury, including bone stress injury, Achilles tendinopathy, plantar fasciitis, and patellofemoral pain syndrome.5 The mechanism of injury is thought to be increased forefoot loading forces.1
A review article showed that patients with pes cavus have reduced pain when using an orthosis, although there is no associated decrease in the risk of injury.5 To the contrary, a prospective study concluded that arch height was unrelated to increased risk of running-related injury.7
Evidence regarding flat feet (pes planus) and risk of injury is also mixed. Some studies show that pes planus is not associated with increased risk of injury in athletes.12 A cross-sectional study in older patients showed those with pes planus morphology had a higher rate of knee pain and wearing away of medial compartment cartilage.13 Because this study comprised only older adults, it is not generalizable to runners—nor can conclusions be drawn about causation, given the cross-sectional nature of the study.
Although a foot orthosis can correct mechanical differences caused by pes planus morphology, there is not enough evidence to conclude that correction results in a lower rate of injury. In sum, data are mixed with regard to arch height as a risk factor for running-related injury.
Continue to: Patients with...
Patients with pes planus or pes cavus should not be discouraged from running, however. If they experience pain with running, they might benefit from a trial of arch support inserts; or consider referral to an orthotist for evaluation for a custom orthosis.
Sex. Based on a prospective cohort study, female runners have a slightly higher rate of running injury than male counterparts.4 Similarly, a study showed that female military members generally had a higher incidence of stress fractures than male military members—specifically, femoral shaft and neck stress fractures.14 Runners who fall in the spectrum of the female athlete triad, as described earlier, are particularly vulnerable to bone stress injury. It is reasonable, therefore, to review risk factors for injury with female runners (as it is with all runners), especially those who have sustained a prior running-related injury.
Increased Q-angle (an obsolete risk factor). The Q-angle is approximated by drawing a line from the anterior superior iliac spine to the patella and a second line from the patella to the tibial tubercle. In males, a normal Q-angle is 14°; in females, 17° (SD = 4.5°). The Q-angle can be obtained by goniometric or radiographic measurement.
An increased Q-angle had been considered an intrinsic risk factor for running injury but has not been shown to be associated with increased risk of running-related injury or patellofemoral pain syndrome.27,28 Because the Q-angle is not a clinically relevant tool in assessing risk of injury, do not routinely measure it or include it in risk-factor counseling.
OA. Based on a systematic review of observational studies, data are inconclusive with regard to whether running contributes to, or is protective against, knee OA.15 In a large cohort study, running (1) was protective against development of hip OA and (2) decreased the risk of requiring hip replacement.29 This finding was supported by animal-model research that concluded that it is inactivity that results in thinning of articular cartilage.29 In addition, a systematic review of randomized controlled trials concluded that knee joint-loading exercises are not harmful to articular cartilage (this is low-quality evidence, however).16
Continue to: Given that there...
Given that there are no high-quality studies suggesting that running contributes to or exacerbates OA, patients with OA can be counseled to start or continue running as tolerated because the health benefit of running likely outweighs risk. Patients with pre-existing moderate-to-severe OA might report knee and hip pain that is already exacerbated by certain activities; if a high-impact activity, such as running, makes that pain worse, exercise counseling that you provide can be tailored to include lower-impact alternatives, such as swimming, cycling, or an elliptical workout.
CASE
In response to Ms. K’s interest in beginning an exercise regimen that includes running, you perform a complete routine pre-participation evaluation and appropriate cardiac screening. You discuss risk factors for running injury, focusing on modifiable risk factors.
Ms. K is perimenopausal but reports a history of regular menstrual cycles. She eats a relatively well-balanced diet. You advise that her BMI should not restrict her from incorporating running into her fitness regimen. Also, you reassure her that she should not restrict running based on a diagnosis of OA; instead, you advise her to monitor her symptoms and reconsider her program if running makes her knee pain worse.
At this point, Ms. K is ready to run. She tells you that, based on your guidance, she feels more comfortable and safe starting a running program.
Preventing injury
After reviewing risk factors for running-related injury with patients, encourage other evidence-based methods of reducing that risk.
Continue to: Shoes
Shoes
The running shoe industry offers a variety of running shoes, from minimalist shoes to cushioned stability shoes that vary based on the amount of cushioning, level of motion control, and amount of heel-to-toe drop. With so many options, new runners might wonder which shoes can reduce their risk of injury and how they should select a pair.
Stability. A characteristic of running shoes promoted by the industry is their stability: ie, their motion control. Stability shoes are marketed to runners who overpronate and therefore limit motion to prevent overpronation. The benefit of stability shoes, or stability insoles, is unclear.30 A randomized controlled trial showed that, in runners who overpronate, motion-control shoes reduced their risk of injury.31 However, another study assessed whether shoes that had been “prescribed” based on foot morphology and stride reduced the risk of injury (compared to neutral, cushioned shoes) and found no change in the incidence of soft-tissue injury.32 Given no strong evidence to suggest otherwise, runners can be advised to buy shoes based on comfort rather than on foot morphology or running stride.
Heel-to-toe drop. Another component of shoe variability is heel-to-toe drop (the height difference between heel and forefoot). A study suggests that moderate-to-high (8-12 mm) heel-to-toe drop is associated with a reduced risk of running injury.33 Barefoot running shoes, which, typically, have no heel-to-toe drop, are associated with increased risk of injury—specifically, foot stress fracture (especially in runners who are even moderately overweight).34,35
Shoe age and shoe wear can be modified to reduce injury. There is evidence that running shoes lose approximately 50% of cushioning after 300 to 500 miles of use.36 Another study found that rotating running shoes—ideally, different types or brands—can lead to fewer running-related injuries.37
In general, patients can be counseled to use shoes that feel comfortable, as long as they replace them regularly (TABLE 2). Runners can also consider alternating pairs of different running shoes between runs. Overweight runners should avoid minimally cushioned and low heel-to-toe drop running shoes.

Continue to: Cross-training
Cross-training
Cross-training exercises for runners include cycling, an elliptical workout, swimming, and weightlifting. Incorporating cross-training can be protective against running injury because cross-training requires different movement patterns, prevents overuse, and equalizes muscle imbalances that occur with running.7 In addition, replacing running with a cross-training activity can decrease weekly running time and mileage, which can further reduce risk of running-related injury.7 Runners—especially higher-mileage runners—should be encouraged to incorporate cross-training into their workout regimen to decrease their risk of injury.
Stretching. The authors of a Cochrane review concluded that there is no significant reduction in injury associated with hamstring or gastrocnemius stretching.32 A small randomized, controlled, crossover study concluded that participants subjectively felt their performance was better when warm-ups included stretching.38 This perceived improvement in performance was similar between groups who completed dynamic or static stretching. However, no difference was noted in flexibility or objective performance between groups who stretched or did not stretch before activity.
Although there is no supporting evidence that stretching reduces the risk of injury, stretching is a low-risk intervention. Because stretching might provide subjective benefit to runners, you need not discourage patients from including this activity in their running program.
CORRESPONDENCE
Kartik Sidhar, MD, 15370 Huff Way, Brookfield, WI, 53005; kartiksidhar@gmail.com
1. Brown CR Jr. Common injuries from running. In: Imboden JB, Hellerman, DB, Stone JH, eds. Current Diagnosis & Treatment: Rheumatology. 3rd ed. McGraw-Hill; 2013.
2. Lange D. Running & jogging - statistic and facts. Statista Web site. November 16, 2020. Accessed March 28, 2021. www.statista.com/topics/1743/running-and-jogging/
3. Warburton DER, Bredin SSD. Health benefits of physical activity: a systematic review of current systematic reviews. Curr Opin Cardiol. 2017;32:541-556. doi:10.1097/HCO.0000000000000437
4. Messier SP, Martin DF, Mihalko SL, et al. A 2-year prospective cohort study of overuse running injuries: The Runners and Injury Longitudinal Study (TRAILS). Am J Sports Med. 2018;46:2211-2221. doi:10.1177/0363546518773755
5. Fields KB, Sykes JC, Walker KM, et al. Prevention of running injuries. Curr Sports Med Rep. 2010;9:176-182. doi:10.1249/JSR.0b013e3181de7ec5
6. van der Worp MP, ten Haaf DSM, van Cingel R. Injuries in runners; a systematic review on risk factors and sex differences. PLoS One. 2015;10:1-18. doi:10.1371/journal.pone.0114937
7. Taunton JE, Ryan MB, Clement DB, et al. A prospective study of running injuries: the Vancouver Sun Run “In Training” clinics. Br J Sports Med. 2003;37:239-244. doi:10.1136/bjsm.37.3.239
8. Dixon SJ, Collop AC, Batt ME. Surface effects on ground reaction forces and lower extremity kinematics in running. Med Sci Sports Exerc. 2000;32:1919-1926. doi:10.1097/00005768-200011000-00016
9. Vadeboncoeur TF, Silvers SM, Taylor WC, et al. Impact of a high body mass index on lower extremity injury in marathon/half-marathon participants. J Phys Act Health. 2012;9:96-103. doi:10.1123/jpah.9.1.96
10. Buist I, Bredeweg SW. Higher risk of injury in overweight novice runners. Br J Sports Med. 2011;45:338. http://dx.doi.org/10.1136/bjsm.2011.084038.79
11. Cowan DN, Jones BH, Robinson JR. Foot morphologic characteristics and risk of Exercise-related injury. Arch Fam Med. 1993;2:773-777. doi:10.1001/archfami.2.7.773
12. Michelson JD, Durant DM, McFarland E. The injury risk associated with pes planus in athletes. Foot Ankle Int. 2002;23:629-633. doi: 10.1177/107110070202300708
13. Gross KD, Felson DT, Niu J, et al. Association of flat feet with knee pain and cartilage damage in older adults. Arthritis Care Res (Hoboken). 2011;63:937-944. doi:10.1002/acr.20431
14. Waterman BR, Gun B, Bader JO, et al. Epidemiology of lower extremity stress fractures in the United States military. Mil Med. 2016;181:1308-1313. doi:10.7205/MILMED-D-15-00571
15. Timmins KA, Leech RD, Batt ME, et al. Running and knee osteoarthritis: a systematic review and meta-analysis. Am J Sports Med. 2017;45:1447-1457. doi:10.1177/0363546516657531
16. Bricca A, Juhl CB, Steultjens M, et al. Impact of exercise on articular cartilage in people at risk of, or with established, knee osteoarthritis: a systematic review of randomised controlled trials. Br J Sports Med. 2019;53:940-947. doi:10.1136/bjsports-2017-098661
17. Johnston CAM, Taunton JE, Lloyd-Smith DR, et al. Preventing running injuries. Practical approach for family doctors. Can Fam Physician. 2003;49:1101-1109.
18. Buist I, Bredeweg SW, van Mechelen W, et al. No effect of a graded training program on the number of running-related injuries in novice runners: a randomized controlled trial. Am J Sports Med. 2008;36:33-39. doi:10.1177/0363546507307505
19. Warden SJ, Davis IS, Fredericson M. Management and prevention of bone stress injuries in long-distance runners. J Orthop Sports Phys Ther. 2014;44:749-765. doi:10.2519/jospt.2014.5334
20. Kim N, Browning RC, Lerner ZF. The effects of pediatric obesity on patellofemoral joint contact force during walking. Gait Posture. 2019;73:209-214. doi:10.1016/j.gaitpost.2019.07.307
21. Tenforde AS, Kraus E, Fredericson M. Bone stress injuries in runners. Phys Med Rehabil Clin N Am. 2016;27:139-149. doi:10.1016/j.pmr.2015.08.008
22. Lappe J, Cullen D, Haynatzki G, et al. Calcium and vitamin D supplementation decreases incidence of stress fractures in female navy recruits. J Bone Miner Res. 2008;23:741-749. doi:10.1359/jbmr.080102
23. Bramah C, Preece SJ, Gill N, et al. Is there a pathological gait associated with common soft tissue running injuries? Am J Sports Med. 2018;46:3023-3031. doi:10.1177/0363546518793657
24. Willy RW, Scholz PT, Davis IS. Mirror gait retraining for the treatment of patellofemoral pain in female runners. Clin Biomech (Bristol Avon). 2012;27:1045-1051. doi:10.1016/j.clinbiomech.2012.07.011
25. Schubert AG, Kempf J, Heiderscheit BC. Influence of stride frequency and length on running mechanics: a systematic review. Sports Health. 2014;6:210-217. doi:10.1177/1941738113508544
26. Bramah C, Preece SJ, Gill N et al. A 10% increase in step rate improves running kinematics and clinical outcomes in runners with patellofemoral pain at 4 weeks and 3 months. Am J Sports Med. 2019;47:3406-3413. doi: 10.1177/0363546519879693
27. Ramskov D, Jensen ML, Obling K, et al. No association between q-angle and foot posture with running-related injuries: a 10 week prospective follow-up study. Int J Sports Phys Ther. 2013;8:407-415.
28. Almeida GPL, Silva AP, França FJR, et al. Q-angle in patellofemoral pain: relationship with dynamic knee valgus, hip abductor torque, pain and function. Rev Bras Ortop. 2016;51:181-186. doi:10.1016/j.rboe.2016.01.010
29. Williams PT. Effects of running and walking on osteoarthritis and hip replacement risk. Med Sci Sports Exerc. 2013;45:1292-1297. doi:10.1249/MSS.0b013e3182885f26
30. Nigg BM, Baltich J, Hoerzer S, et al. Running shoes and running injuries: mythbusting and a proposal for two new paradigms: ‘Preferred movement path’ and ‘comfort filter.’ Br J Sports Med. 2015;49:1290-1294. doi:10.1136/bjsports-2015-095054
31. Malisoux L, Chambon N, Delattre N, et al. Injury risk in runners using standard or motion control shoes: a randomised controlled trial with participant and assessor blinding. Br J Sports Med. 2016;50:481-487. doi:10.1136/bjsports-2015-095031
32. Yeung SS, Yeung EW, Gillespie LD. Interventions for preventing lower limb soft-tissue running injuries. Cochrane Database Syst Rev. 2011(7):CD001256. doi:10.1002/14651858.cd001256.pub2
33. Malisoux L, Chambon N, Urhausen A, et al. Influence of the heel-to-toe drop of standard cushioned running shoes on injury risk in leisure-time runners: a randomized controlled trial with 6-month follow-up. Am J Sports Med. 2016;44:2933-2940. doi:10.1177/0363546516654690
34. Ryan M, Elashi M, Newsham-West R, et al. Examining injury risk and pain perception in runners using minimalist footwear. Br J Sports Med. 2014;48:1257-1262. doi:10.1136/bjsports-2012-092061
35. Fuller JT, Thewlis D, Buckley JD, et al.. Body mass and weekly training distance influence the pain and injuries experienced by runners using minimalist shoes: a randomized controlled trial. Am J Sports Med. 2017;45:1162-1170. doi:10.1177/0363546516682497
36. Cook SD, Kester MA, Brunet ME. Shock absorption characteristics of running shoes. Am J Sports Med. 1985;13:248-253. doi.org/10.1177/036354658501300406
37. Malisoux L, Ramesh J, Mann R, et al. Can parallel use of different running shoes decrease running-related injury risk? Scand J Med Sci Sport. 2015;25:110-115. doi:10.1111/sms.12154
38. Blazevich AJ, Gill ND, Kvorning T, et al. No effect of muscle stretching within a full, dynamic warm-up on athletic performance. Med Sci Sports Exerc. 2018;50:1258-1266. doi:10.1249/MSS.0000000000001539
1. Brown CR Jr. Common injuries from running. In: Imboden JB, Hellerman, DB, Stone JH, eds. Current Diagnosis & Treatment: Rheumatology. 3rd ed. McGraw-Hill; 2013.
2. Lange D. Running & jogging - statistic and facts. Statista Web site. November 16, 2020. Accessed March 28, 2021. www.statista.com/topics/1743/running-and-jogging/
3. Warburton DER, Bredin SSD. Health benefits of physical activity: a systematic review of current systematic reviews. Curr Opin Cardiol. 2017;32:541-556. doi:10.1097/HCO.0000000000000437
4. Messier SP, Martin DF, Mihalko SL, et al. A 2-year prospective cohort study of overuse running injuries: The Runners and Injury Longitudinal Study (TRAILS). Am J Sports Med. 2018;46:2211-2221. doi:10.1177/0363546518773755
5. Fields KB, Sykes JC, Walker KM, et al. Prevention of running injuries. Curr Sports Med Rep. 2010;9:176-182. doi:10.1249/JSR.0b013e3181de7ec5
6. van der Worp MP, ten Haaf DSM, van Cingel R. Injuries in runners; a systematic review on risk factors and sex differences. PLoS One. 2015;10:1-18. doi:10.1371/journal.pone.0114937
7. Taunton JE, Ryan MB, Clement DB, et al. A prospective study of running injuries: the Vancouver Sun Run “In Training” clinics. Br J Sports Med. 2003;37:239-244. doi:10.1136/bjsm.37.3.239
8. Dixon SJ, Collop AC, Batt ME. Surface effects on ground reaction forces and lower extremity kinematics in running. Med Sci Sports Exerc. 2000;32:1919-1926. doi:10.1097/00005768-200011000-00016
9. Vadeboncoeur TF, Silvers SM, Taylor WC, et al. Impact of a high body mass index on lower extremity injury in marathon/half-marathon participants. J Phys Act Health. 2012;9:96-103. doi:10.1123/jpah.9.1.96
10. Buist I, Bredeweg SW. Higher risk of injury in overweight novice runners. Br J Sports Med. 2011;45:338. http://dx.doi.org/10.1136/bjsm.2011.084038.79
11. Cowan DN, Jones BH, Robinson JR. Foot morphologic characteristics and risk of Exercise-related injury. Arch Fam Med. 1993;2:773-777. doi:10.1001/archfami.2.7.773
12. Michelson JD, Durant DM, McFarland E. The injury risk associated with pes planus in athletes. Foot Ankle Int. 2002;23:629-633. doi: 10.1177/107110070202300708
13. Gross KD, Felson DT, Niu J, et al. Association of flat feet with knee pain and cartilage damage in older adults. Arthritis Care Res (Hoboken). 2011;63:937-944. doi:10.1002/acr.20431
14. Waterman BR, Gun B, Bader JO, et al. Epidemiology of lower extremity stress fractures in the United States military. Mil Med. 2016;181:1308-1313. doi:10.7205/MILMED-D-15-00571
15. Timmins KA, Leech RD, Batt ME, et al. Running and knee osteoarthritis: a systematic review and meta-analysis. Am J Sports Med. 2017;45:1447-1457. doi:10.1177/0363546516657531
16. Bricca A, Juhl CB, Steultjens M, et al. Impact of exercise on articular cartilage in people at risk of, or with established, knee osteoarthritis: a systematic review of randomised controlled trials. Br J Sports Med. 2019;53:940-947. doi:10.1136/bjsports-2017-098661
17. Johnston CAM, Taunton JE, Lloyd-Smith DR, et al. Preventing running injuries. Practical approach for family doctors. Can Fam Physician. 2003;49:1101-1109.
18. Buist I, Bredeweg SW, van Mechelen W, et al. No effect of a graded training program on the number of running-related injuries in novice runners: a randomized controlled trial. Am J Sports Med. 2008;36:33-39. doi:10.1177/0363546507307505
19. Warden SJ, Davis IS, Fredericson M. Management and prevention of bone stress injuries in long-distance runners. J Orthop Sports Phys Ther. 2014;44:749-765. doi:10.2519/jospt.2014.5334
20. Kim N, Browning RC, Lerner ZF. The effects of pediatric obesity on patellofemoral joint contact force during walking. Gait Posture. 2019;73:209-214. doi:10.1016/j.gaitpost.2019.07.307
21. Tenforde AS, Kraus E, Fredericson M. Bone stress injuries in runners. Phys Med Rehabil Clin N Am. 2016;27:139-149. doi:10.1016/j.pmr.2015.08.008
22. Lappe J, Cullen D, Haynatzki G, et al. Calcium and vitamin D supplementation decreases incidence of stress fractures in female navy recruits. J Bone Miner Res. 2008;23:741-749. doi:10.1359/jbmr.080102
23. Bramah C, Preece SJ, Gill N, et al. Is there a pathological gait associated with common soft tissue running injuries? Am J Sports Med. 2018;46:3023-3031. doi:10.1177/0363546518793657
24. Willy RW, Scholz PT, Davis IS. Mirror gait retraining for the treatment of patellofemoral pain in female runners. Clin Biomech (Bristol Avon). 2012;27:1045-1051. doi:10.1016/j.clinbiomech.2012.07.011
25. Schubert AG, Kempf J, Heiderscheit BC. Influence of stride frequency and length on running mechanics: a systematic review. Sports Health. 2014;6:210-217. doi:10.1177/1941738113508544
26. Bramah C, Preece SJ, Gill N et al. A 10% increase in step rate improves running kinematics and clinical outcomes in runners with patellofemoral pain at 4 weeks and 3 months. Am J Sports Med. 2019;47:3406-3413. doi: 10.1177/0363546519879693
27. Ramskov D, Jensen ML, Obling K, et al. No association between q-angle and foot posture with running-related injuries: a 10 week prospective follow-up study. Int J Sports Phys Ther. 2013;8:407-415.
28. Almeida GPL, Silva AP, França FJR, et al. Q-angle in patellofemoral pain: relationship with dynamic knee valgus, hip abductor torque, pain and function. Rev Bras Ortop. 2016;51:181-186. doi:10.1016/j.rboe.2016.01.010
29. Williams PT. Effects of running and walking on osteoarthritis and hip replacement risk. Med Sci Sports Exerc. 2013;45:1292-1297. doi:10.1249/MSS.0b013e3182885f26
30. Nigg BM, Baltich J, Hoerzer S, et al. Running shoes and running injuries: mythbusting and a proposal for two new paradigms: ‘Preferred movement path’ and ‘comfort filter.’ Br J Sports Med. 2015;49:1290-1294. doi:10.1136/bjsports-2015-095054
31. Malisoux L, Chambon N, Delattre N, et al. Injury risk in runners using standard or motion control shoes: a randomised controlled trial with participant and assessor blinding. Br J Sports Med. 2016;50:481-487. doi:10.1136/bjsports-2015-095031
32. Yeung SS, Yeung EW, Gillespie LD. Interventions for preventing lower limb soft-tissue running injuries. Cochrane Database Syst Rev. 2011(7):CD001256. doi:10.1002/14651858.cd001256.pub2
33. Malisoux L, Chambon N, Urhausen A, et al. Influence of the heel-to-toe drop of standard cushioned running shoes on injury risk in leisure-time runners: a randomized controlled trial with 6-month follow-up. Am J Sports Med. 2016;44:2933-2940. doi:10.1177/0363546516654690
34. Ryan M, Elashi M, Newsham-West R, et al. Examining injury risk and pain perception in runners using minimalist footwear. Br J Sports Med. 2014;48:1257-1262. doi:10.1136/bjsports-2012-092061
35. Fuller JT, Thewlis D, Buckley JD, et al.. Body mass and weekly training distance influence the pain and injuries experienced by runners using minimalist shoes: a randomized controlled trial. Am J Sports Med. 2017;45:1162-1170. doi:10.1177/0363546516682497
36. Cook SD, Kester MA, Brunet ME. Shock absorption characteristics of running shoes. Am J Sports Med. 1985;13:248-253. doi.org/10.1177/036354658501300406
37. Malisoux L, Ramesh J, Mann R, et al. Can parallel use of different running shoes decrease running-related injury risk? Scand J Med Sci Sport. 2015;25:110-115. doi:10.1111/sms.12154
38. Blazevich AJ, Gill ND, Kvorning T, et al. No effect of muscle stretching within a full, dynamic warm-up on athletic performance. Med Sci Sports Exerc. 2018;50:1258-1266. doi:10.1249/MSS.0000000000001539
PRACTICE RECOMMENDATIONS
› Counsel runners to cross-train, replace shoes regularly, and use shoes with moderate-to-high (8-12 mm) heel-to-toe drop. C
› Don’t discourage running for exercise, as long as it is tolerated, in patients who have osteoarthritis. C
› Encourage moderation in running distance and intensity, especially in novice runners. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Veteran and Provider Perspectives on Telehealth for Vocational Rehabilitation Services
Vocational rehabilitation (VR) interventions are offered through Compensated Work Therapy (CWT) as part of clinical care in the Veterans Health Administration (VHA) to improve employment and quality of life outcomes for veterans with life-altering disabilities.1–5 CWT vocational services range from assessment, vocational counseling, and treatment plan development to job placement, coaching, and follow-along support.1 However, many veterans receive care in community-based clinics that are not staffed with a VR specialist (VRS) to provide these services.6–8 Telehealth may increase patient access to VR, especially for rural veterans and those with travel barriers, but it is not known whether veterans and VRS would find this to be a satisfactory service delivery method.8,9 This paper examines veteran and VRS provider perspectives on VR provided by telehealth (VRtele) as part of a VHA clinical demonstration project. To our knowledge, this is the first report of using real-time, clinic-based VRtele.
Methods
The Rural Veterans Supported Employment Telerehabilitation Initiative (RVSETI) was conducted as a field-initiated demonstration project at 2 US Department of Veterans Affairs (VA) medical centers (VAMCs) in Florida between 2014 and 2016: James A. Haley Veterans’ Hospital & Clinics (Tampa) and Malcom Randall VAMC (Gainesville). This retrospective evaluation of its first year did not require institutional review board approval as it was determined to be a quality improvement project by the local research service.
The patient population for the project was veterans with disabilities who were referred by clinical consults to the CWT service, a recovery-oriented vocational program. During the project years, veterans were offered the option of receiving VR services, such as supported employment, community-based employment services, or vocational assistance, through VRtele rather than traditional face-to-face meetings. The specific interventions delivered included patient orientation, interview assessment, treatment plan development, referral activities, vocational counseling, assessment of workplace for accommodation needs, vocational case management, and other employment supports. VR staff participating in the project included 2 VR supervisors, 1 supported employment mentor trainer, and 5 VRSs.
Each clinic was set up for VRtele, and codes were added to the electronic health record (EHR) to ensure proper documentation. Participating VRSs completed teleconferencing training, including a skills assessment using the equipment for real-time, high-quality video streaming over an encrypted network to provide services in a patient’s home or other remote locations. VRS staff provided veterans with instructions on using a VA-provided tablet or their own device and assisted them with establishing connectivity with the network. Video equipment included speakers, camera, and headphones connected to the desktop computer or laptop of the VRS. A patient’s first VRtele
Demographic data, primary diagnosis, VR usage data, and zip codes of participating veterans were extracted from the EHR. Veterans completed a 2-part satisfaction survey administered 90 days after enrollment and at discharge. Part 1 was composed of 15 items, most with a 5-point Likert scale (higher ratings indicated greater satisfaction), on various aspects of the VRtele experience, such as audio and video quality and wait times.10 Part 2 addressed VR services and the VRS and consisted of 8 Likert scale items with the option to add a comment for each and 2 open-ended items that asked the participant to list what they liked best and least about VRtele.
Semistructured, in-person 30- to 60-minute interviews were conducted with VRSs at the initiation of VRtele
After ≥ 2 months of VRtele use
Analyses
Descriptive statistics were used for EHR data and satisfaction surveys. For qualitative analysis, each transcript was read in full by 2 researchers to get an overview of the data, and a rapid analysis approach was used to identify central themes focused on how technology was used and the experiences of the participants.11,12 Relevant text for each topic was tabulated, and a summary table was created that highlighted overlapping ideas discussed by the interviewees as well as differences.
Results
Of the 22 veterans who participated in the project, 11 completed satisfaction surveys and 4 participated in qualitative interviews. The rural and nonrural groups did not differ demographically or by diagnosis, which was predominantly mental health related. Only 1 veteran in each group owned a tablet; the majority of both groups required VA-issued devices: 80% (n = 8) rural and 91.7% (n = 11) nonrural. The number of VRtele sessions for the groups also was similar, 53 for rural and 60 for nonrural, as was the mean (SD) number of sessions per veteran: 5.3 (SD, 3.2) rural and 5.0 (SD, 2.5) urban. Overall, 63 miles per session were saved, mostly for rural veterans, and the number of mean (SD) miles saved per veteran was greater for rural than nonrural veterans: 379.2 (243.0) and 256.1 (275.9), respectively. One veteran who moved to a different state during the program continued VRtele at the new location. In a qualitative sampling of 5 VRtele sessions, all the VRSs used office desktop computers.
Level of satisfaction with aspects of VRtele related to the technology rated was consistently > 4 on the Likert scale. The lowest mean (SD) ratings were 4.2 (1.0) for audio quality and 4.4 (0.5) for video quality, and the highest rating was given for equipment operation explanation and privacy was respected, 4.9 (0.3) for both. All questions related to satisfaction with services were also rated high: The mean (SD) lowest ratings were 4.3 (1.0) given to both vocational needs 4.3 (1.0) and tasks effectively helped achieve goals 4.3 (0.7). The highest mean (SD) ratings were 4.6 (0.5) given to VR program service explained and 4.7 (0.5) for appointment timeliness.
Qualitative Results
At first, some VRSs thought the teleconferencing system might be difficult or awkward to use, but they found it easier to set up than expected and seamless to use. VRS staff reported being surprised at how well it worked despite some issues that occurred with loading the software. Once loaded, however, the connection worked well, one VRS noting that following step-by-step instructions solved the problem. Some VRSs indicated they did not invite all the veterans on their caseload to participate in VRtele due to concerns with the patient’s familiarity with technology, but one VRS stated, “I haven’t had anybody that failed to do a [session] that I couldn’t get them up and running within a few minutes.”
When working in the community, VRSs reported using laptops for VRtele but found that these devices were unreliable due to lack of internet access and were slow to start; several VRSs thought tablets would have been more helpful. Some veterans reported technical glitches, lack of comfort with technology, or a problem with sound due to a tablet’s protective case blocking the speakers. To solve the sound issue, a veteran used headphones. This veteran also explained that the log-on process required a new password every time, so he would keep a pen and paper ready to write it down. Because signing in and setting up takes a little time, this veteran and his VRS agreed to start connecting 5 minutes before their meeting time to allow for that set- up time.
Initially, some VRSs expressed concern that transitioning to VRtele would affect the quality of interactions with the veterans. However, VRSs also identified strengths of VRtele, including flexibility, saved time, and increased interaction. One VRS discussed a veteran’s adaptation by saying, “I think he feels even more involved in his plan [and] enjoys the increased interaction.” Veterans reported enjoying using tablets and identified the main strength of VRtele as being able to talk face-to-face with the VRS. Echoing the VRSs, veterans reported teleconferencing saved time by avoiding travel and enabled spontaneous meetings. One of the veterans summed up the benefits of using VRtele: “I’d rather just connect. It’s going to take us 40 to 50 minutes [to meet in person] when we can just connect right here and it takes 15 to 20. We don’t have to go through the driving.… So this right here, doing it ahead of time and having the appointment, it’s a lot easier.”
In their interviews, VRSs talked about enjoying VRtele. A VRS explained: “It makes it a lot easier. It makes me feel less guilty. This way [veterans] don’t have to use their gas money, use their time. I know [the veteran] had something else he needed to do today.” Thus, both veterans and VRSs were satisfied with their VRtele experiences.
Discussion
This first report on the perspective of providers and veterans using VRtele suggests that it is a viable option for service delivery and that is highly satisfactory for serving veterans with disabilities, many of whom live in rural areas or have travel barriers. These findings are consistent with data on telerehabilitation for veterans with cognitive, physical, and mental disabilities.13-22 Further, the data support the notion of using VRtele to facilitate long-term VR follow-up for persons with disabilities, as illustrated by successful continuation of vocational services after a veteran moved out of state.23
Similar to other reports, our experience highlighted 2 factors that affect successful VRtele: (1) Troubleshooting technology barriers for both VR providers and clients; and (2) supportive leadership to facilitate implementation
Changes to technology and increased usage of VA Video Connect may indicate that the barriers identified from the earlier process described here have been diminished or eliminated. More evaluation is needed to assess whether system upgrades have increased ease of use and access for veterans with disabilities.
Conclusions
Encouragingly, this clinical demonstration project showed that both providers and clients recognize the benefits of VRtele. Patient satisfaction and decreased travel costs were clear advantages to using VRtele for this small group of veterans who had barriers to care due to travel or disability barriers. As this program evaluation was limited by a small sample, absence of a comparison group, and lack of outcome data (eg, employment rates, hours, wages, retention), future research is needed on implementation and outcomes of VRtele
Acknowledgments
The authors thank Lynn Dirk, MAMC, for substantial editorial assistance. This material was based on work supported by Rural Veterans Supported Employment TeleRehabilitation Initiative (RVSETI), funded by the VA Office of Rural Health (Project # N08-FY14Q3-S2-P01222) and by support of the VA Health Services Research and Development Service. This work was presented in part at the 114th Annual Meeting of the American Anthropological Association at Denver, Colorado, November 21, 2015; a field-based Health Services Research and Development Service meeting, US Department of Veterans Affairs at Washington, DC, September 12, 2016; and the 2016 Annual Conference of the American Congress for Rehabilitation Medicine at Chicago, Illinois, October-November 2016.
1. Abraham KM, Yosef M, Resnick SG, Zivin K. Competitive employment outcomes among veterans in VHA therapeutic and supported employment services programs. Psychiatr Serv. 2017;68(9):938-946. doi:10.1176/appi.ps.201600412
2. Davis LL, Kyriakides TC, Suris AM, et al. Effect of evidence-based supported employment vs transitional work on achieving steady work among veterans with posttraumatic stress disorder: a randomized clinical trial. JAMA Psychiatry. 2018;75(4):316. doi:10.1001/jamapsychiatry.2017.4472
3. Ottomanelli L, Goetz LL, Suris A, et al. Effectiveness of supported employment for veterans with spinal cord injuries: results from a randomized multisite study. Arch Phys Med Rehabil. 2012;93(5):740-747. doi:10.1016/j.apmr.2012.01.002
4. Ottomanelli L, Goetz LL, Barnett SD, et al. Individual placement and support in spinal cord injury: a longitudinal observational study of employment outcomes. Arch Phys Med Rehabil. 2017;98(8):1567-1575. doi:10.1016/j.apmr.2016.12.010
5. Cotner BA, Ottomanelli L, O’Connor DR, Njoh EN, Barnett SD, Miech EJ. Quality of life outcomes for veterans with spinal cord injury receiving individual placement and support (IPS). Top Spinal Cord Inj Rehabil. 2018;24(4):325-335. doi:10.1310/sci17-00046
6. Metzel DS, Giordano A. Locations of employment services and people with disabilities: a geographical analysis of accessibility. J Disabil Policy Stud. 2007;18(2):88-97. doi:10.1177/10442073070180020501
7. Landon T, Connor A, McKnight-Lizotte M, Peña J. Rehabilitation counseling in rural settings: a phenomenological study on barriers and supports. J Rehabil. 2019;85(2):47-57.
8. Riemer-Reiss M. Vocational rehabilitation counseling at a distance: Challenges, strategies and ethics to consider. J Rehabil. 2000;66(1):11-17.
9. Schmeler MR, Schein RM, McCue M, Betz K. Telerehabilitation clinical and vocational applications for assistive technology: research, opportunities, and challenges. Int J Telerehabilitation. 2009;1(1):59-72.
10. Levy CE, Silverman E, Jia H, Geiss M, Omura D. Effects of physical therapy delivery via home video telerehabilitation on functional and health-related quality of life outcomes. J Rehabil Res Dev. 2015;52(3):361-370. doi:10.1682/JRRD.2014.10.0239
11. McMullen CK, Ash JS, Sittig DF, et al. Rapid assessment of clinical information systems in the healthcare setting: an efficient method for time-pressed evaluation. Methods Inf Med. 2011;50(4):299-307. doi:10.3414/ME10-01-0042
12. Averill JB. Matrix analysis as a complementary analytic strategy in qualitative inquiry. Qual Health Res. 2002;12(6):855-866.
13. Egede LE, Acierno R, Knapp RG, et al. Psychotherapy for depression in older veterans via telemedicine: a randomised, open-label, non-inferiority trial. Lancet Psychiatry. 2015;2(8):693-701. doi:10.1016/S2215-0366(15)00122-4
14. Fortney JC, Pyne JM, Edlund MJ, et al. A randomized trial of telemedicine-based collaborative care for depression. J Gen Intern Med. 2007;22(8):1086-1093. doi:10.1007/s11606-007-0201-9
15. Fortney JC, Pyne JM, Kimbrell TA, et al. Telemedicine-based collaborative care for posttraumatic stress disorder: a randomized clinical trial. JAMA Psychiatry. 2015;72(1):58. doi:10.1001/jamapsychiatry.2014.1575
16. Grubbs KM, Fortney JC, Dean T, Williams JS, Godleski L. A comparison of mental health diagnoses treated via interactive video and face to face in the Veterans Healthcare Administration. Telemed E-Health. 2015;21(7):564-566. doi:10.1089/tmj.2014.0152
17. Agostini M, Moja L, Banzi R, et al. Telerehabilitation and recovery of motor function: a systematic review and meta-analysis. J Telemed Telecare. 2015;21(4):202-213. doi:10.1177/1357633X15572201
18. Bergquist TF, Thompson K, Gehl C, Munoz Pineda J. Satisfaction ratings after receiving internet-based cognitive rehabilitation in persons with memory impairments after severe acquired brain injury. Telemed E-Health. 2010;16(4):417-423. doi:10.1089/tmj.2009.0118
19. Brennan DM, Georgeadis AC, Baron CR, Barker LM. The effect of videoconference-based telerehabilitation on story retelling performance by brain-injured subjects and its implications for remote speech-language therapy. Telemed J E Health. 2004;10(2):147-154. doi:10.1089/tmj.2004.10.147
20. Dallolio L, Menarini M, China S, et al. Functional and clinical outcomes of telemedicine in patients with spinal cord injury. Arch Phys Med Rehabil. 2008;89(12):2332-2341. doi:10.1016/j.apmr.2008.06.012
21. Houlihan BV, Jette A, Friedman RH, et al. A pilot study of a telehealth intervention for persons with spinal cord dysfunction. Spinal Cord. 2013;51(9):715-720.doi:10.1038/sc.2013.45
22. Smith MW, Hill ML, Hopkins KL, Kiratli BJ, Cronkite RC. A modeled analysis of telehealth methods for treating pressure ulcers after spinal cord injury. Int J Telemed Appl. 2012;2012:1-10. doi:10.1155/2012/729492
23. Balcazar FE, Keys CB, Davis M, Lardon C, Jones C. Strengths and challenges of intervention research in vocational rehabilitation: an illustration of agency-university collaboration. J Rehabil. 2005;71(2):40-48.
24. Martinez RN, Hogan TP, Balbale S, et al. Sociotechnical perspective on implementing clinical video telehealth for veterans with spinal cord injuries and disorders. Telemed J E Health. 2017;23(7):567-576. doi:10.1089/tmj.2016.0200
25. Martinez RN, Hogan TP, Lones K, et al. Evaluation and treatment of mild traumatic brain injury through the implementation of clinical video telehealth: provider perspectives from the Veterans Health Administration. PM R. 2017;9(3):231-240. doi:10.1016/j.pmrj.2016.07.002
26. Smith AC, Thomas E, Snoswell CL, et al. Telehealth for global emergencies: implications for coronavirus disease 2019 (COVID-19). J Telemed Telecare. 2020;26(5):309-313. doi:10.1177/1357633X20916567
27. Cowper-Ripley DC, Jia H, Wang X, et al. Trends in VA telerehabilitation patients and encounters over time and by rurality. Fed Pract. 2019; 36(3):122-128.
28. US Department of Veterans Affairs. Veterans VA Video Connect. Published May 22, 2020. Accessed May 29, 2020. https://mobile.va.gov/app/va-video-connect#AppDescription.
29. US Department of Veterans Affairs. VA telehealth at home. Accessed May 29, 2020. https://telehealth.va.gov/type/home
Vocational rehabilitation (VR) interventions are offered through Compensated Work Therapy (CWT) as part of clinical care in the Veterans Health Administration (VHA) to improve employment and quality of life outcomes for veterans with life-altering disabilities.1–5 CWT vocational services range from assessment, vocational counseling, and treatment plan development to job placement, coaching, and follow-along support.1 However, many veterans receive care in community-based clinics that are not staffed with a VR specialist (VRS) to provide these services.6–8 Telehealth may increase patient access to VR, especially for rural veterans and those with travel barriers, but it is not known whether veterans and VRS would find this to be a satisfactory service delivery method.8,9 This paper examines veteran and VRS provider perspectives on VR provided by telehealth (VRtele) as part of a VHA clinical demonstration project. To our knowledge, this is the first report of using real-time, clinic-based VRtele.
Methods
The Rural Veterans Supported Employment Telerehabilitation Initiative (RVSETI) was conducted as a field-initiated demonstration project at 2 US Department of Veterans Affairs (VA) medical centers (VAMCs) in Florida between 2014 and 2016: James A. Haley Veterans’ Hospital & Clinics (Tampa) and Malcom Randall VAMC (Gainesville). This retrospective evaluation of its first year did not require institutional review board approval as it was determined to be a quality improvement project by the local research service.
The patient population for the project was veterans with disabilities who were referred by clinical consults to the CWT service, a recovery-oriented vocational program. During the project years, veterans were offered the option of receiving VR services, such as supported employment, community-based employment services, or vocational assistance, through VRtele rather than traditional face-to-face meetings. The specific interventions delivered included patient orientation, interview assessment, treatment plan development, referral activities, vocational counseling, assessment of workplace for accommodation needs, vocational case management, and other employment supports. VR staff participating in the project included 2 VR supervisors, 1 supported employment mentor trainer, and 5 VRSs.
Each clinic was set up for VRtele, and codes were added to the electronic health record (EHR) to ensure proper documentation. Participating VRSs completed teleconferencing training, including a skills assessment using the equipment for real-time, high-quality video streaming over an encrypted network to provide services in a patient’s home or other remote locations. VRS staff provided veterans with instructions on using a VA-provided tablet or their own device and assisted them with establishing connectivity with the network. Video equipment included speakers, camera, and headphones connected to the desktop computer or laptop of the VRS. A patient’s first VRtele
Demographic data, primary diagnosis, VR usage data, and zip codes of participating veterans were extracted from the EHR. Veterans completed a 2-part satisfaction survey administered 90 days after enrollment and at discharge. Part 1 was composed of 15 items, most with a 5-point Likert scale (higher ratings indicated greater satisfaction), on various aspects of the VRtele experience, such as audio and video quality and wait times.10 Part 2 addressed VR services and the VRS and consisted of 8 Likert scale items with the option to add a comment for each and 2 open-ended items that asked the participant to list what they liked best and least about VRtele.
Semistructured, in-person 30- to 60-minute interviews were conducted with VRSs at the initiation of VRtele
After ≥ 2 months of VRtele use
Analyses
Descriptive statistics were used for EHR data and satisfaction surveys. For qualitative analysis, each transcript was read in full by 2 researchers to get an overview of the data, and a rapid analysis approach was used to identify central themes focused on how technology was used and the experiences of the participants.11,12 Relevant text for each topic was tabulated, and a summary table was created that highlighted overlapping ideas discussed by the interviewees as well as differences.
Results
Of the 22 veterans who participated in the project, 11 completed satisfaction surveys and 4 participated in qualitative interviews. The rural and nonrural groups did not differ demographically or by diagnosis, which was predominantly mental health related. Only 1 veteran in each group owned a tablet; the majority of both groups required VA-issued devices: 80% (n = 8) rural and 91.7% (n = 11) nonrural. The number of VRtele sessions for the groups also was similar, 53 for rural and 60 for nonrural, as was the mean (SD) number of sessions per veteran: 5.3 (SD, 3.2) rural and 5.0 (SD, 2.5) urban. Overall, 63 miles per session were saved, mostly for rural veterans, and the number of mean (SD) miles saved per veteran was greater for rural than nonrural veterans: 379.2 (243.0) and 256.1 (275.9), respectively. One veteran who moved to a different state during the program continued VRtele at the new location. In a qualitative sampling of 5 VRtele sessions, all the VRSs used office desktop computers.
Level of satisfaction with aspects of VRtele related to the technology rated was consistently > 4 on the Likert scale. The lowest mean (SD) ratings were 4.2 (1.0) for audio quality and 4.4 (0.5) for video quality, and the highest rating was given for equipment operation explanation and privacy was respected, 4.9 (0.3) for both. All questions related to satisfaction with services were also rated high: The mean (SD) lowest ratings were 4.3 (1.0) given to both vocational needs 4.3 (1.0) and tasks effectively helped achieve goals 4.3 (0.7). The highest mean (SD) ratings were 4.6 (0.5) given to VR program service explained and 4.7 (0.5) for appointment timeliness.
Qualitative Results
At first, some VRSs thought the teleconferencing system might be difficult or awkward to use, but they found it easier to set up than expected and seamless to use. VRS staff reported being surprised at how well it worked despite some issues that occurred with loading the software. Once loaded, however, the connection worked well, one VRS noting that following step-by-step instructions solved the problem. Some VRSs indicated they did not invite all the veterans on their caseload to participate in VRtele due to concerns with the patient’s familiarity with technology, but one VRS stated, “I haven’t had anybody that failed to do a [session] that I couldn’t get them up and running within a few minutes.”
When working in the community, VRSs reported using laptops for VRtele but found that these devices were unreliable due to lack of internet access and were slow to start; several VRSs thought tablets would have been more helpful. Some veterans reported technical glitches, lack of comfort with technology, or a problem with sound due to a tablet’s protective case blocking the speakers. To solve the sound issue, a veteran used headphones. This veteran also explained that the log-on process required a new password every time, so he would keep a pen and paper ready to write it down. Because signing in and setting up takes a little time, this veteran and his VRS agreed to start connecting 5 minutes before their meeting time to allow for that set- up time.
Initially, some VRSs expressed concern that transitioning to VRtele would affect the quality of interactions with the veterans. However, VRSs also identified strengths of VRtele, including flexibility, saved time, and increased interaction. One VRS discussed a veteran’s adaptation by saying, “I think he feels even more involved in his plan [and] enjoys the increased interaction.” Veterans reported enjoying using tablets and identified the main strength of VRtele as being able to talk face-to-face with the VRS. Echoing the VRSs, veterans reported teleconferencing saved time by avoiding travel and enabled spontaneous meetings. One of the veterans summed up the benefits of using VRtele: “I’d rather just connect. It’s going to take us 40 to 50 minutes [to meet in person] when we can just connect right here and it takes 15 to 20. We don’t have to go through the driving.… So this right here, doing it ahead of time and having the appointment, it’s a lot easier.”
In their interviews, VRSs talked about enjoying VRtele. A VRS explained: “It makes it a lot easier. It makes me feel less guilty. This way [veterans] don’t have to use their gas money, use their time. I know [the veteran] had something else he needed to do today.” Thus, both veterans and VRSs were satisfied with their VRtele experiences.
Discussion
This first report on the perspective of providers and veterans using VRtele suggests that it is a viable option for service delivery and that is highly satisfactory for serving veterans with disabilities, many of whom live in rural areas or have travel barriers. These findings are consistent with data on telerehabilitation for veterans with cognitive, physical, and mental disabilities.13-22 Further, the data support the notion of using VRtele to facilitate long-term VR follow-up for persons with disabilities, as illustrated by successful continuation of vocational services after a veteran moved out of state.23
Similar to other reports, our experience highlighted 2 factors that affect successful VRtele: (1) Troubleshooting technology barriers for both VR providers and clients; and (2) supportive leadership to facilitate implementation
Changes to technology and increased usage of VA Video Connect may indicate that the barriers identified from the earlier process described here have been diminished or eliminated. More evaluation is needed to assess whether system upgrades have increased ease of use and access for veterans with disabilities.
Conclusions
Encouragingly, this clinical demonstration project showed that both providers and clients recognize the benefits of VRtele. Patient satisfaction and decreased travel costs were clear advantages to using VRtele for this small group of veterans who had barriers to care due to travel or disability barriers. As this program evaluation was limited by a small sample, absence of a comparison group, and lack of outcome data (eg, employment rates, hours, wages, retention), future research is needed on implementation and outcomes of VRtele
Acknowledgments
The authors thank Lynn Dirk, MAMC, for substantial editorial assistance. This material was based on work supported by Rural Veterans Supported Employment TeleRehabilitation Initiative (RVSETI), funded by the VA Office of Rural Health (Project # N08-FY14Q3-S2-P01222) and by support of the VA Health Services Research and Development Service. This work was presented in part at the 114th Annual Meeting of the American Anthropological Association at Denver, Colorado, November 21, 2015; a field-based Health Services Research and Development Service meeting, US Department of Veterans Affairs at Washington, DC, September 12, 2016; and the 2016 Annual Conference of the American Congress for Rehabilitation Medicine at Chicago, Illinois, October-November 2016.
Vocational rehabilitation (VR) interventions are offered through Compensated Work Therapy (CWT) as part of clinical care in the Veterans Health Administration (VHA) to improve employment and quality of life outcomes for veterans with life-altering disabilities.1–5 CWT vocational services range from assessment, vocational counseling, and treatment plan development to job placement, coaching, and follow-along support.1 However, many veterans receive care in community-based clinics that are not staffed with a VR specialist (VRS) to provide these services.6–8 Telehealth may increase patient access to VR, especially for rural veterans and those with travel barriers, but it is not known whether veterans and VRS would find this to be a satisfactory service delivery method.8,9 This paper examines veteran and VRS provider perspectives on VR provided by telehealth (VRtele) as part of a VHA clinical demonstration project. To our knowledge, this is the first report of using real-time, clinic-based VRtele.
Methods
The Rural Veterans Supported Employment Telerehabilitation Initiative (RVSETI) was conducted as a field-initiated demonstration project at 2 US Department of Veterans Affairs (VA) medical centers (VAMCs) in Florida between 2014 and 2016: James A. Haley Veterans’ Hospital & Clinics (Tampa) and Malcom Randall VAMC (Gainesville). This retrospective evaluation of its first year did not require institutional review board approval as it was determined to be a quality improvement project by the local research service.
The patient population for the project was veterans with disabilities who were referred by clinical consults to the CWT service, a recovery-oriented vocational program. During the project years, veterans were offered the option of receiving VR services, such as supported employment, community-based employment services, or vocational assistance, through VRtele rather than traditional face-to-face meetings. The specific interventions delivered included patient orientation, interview assessment, treatment plan development, referral activities, vocational counseling, assessment of workplace for accommodation needs, vocational case management, and other employment supports. VR staff participating in the project included 2 VR supervisors, 1 supported employment mentor trainer, and 5 VRSs.
Each clinic was set up for VRtele, and codes were added to the electronic health record (EHR) to ensure proper documentation. Participating VRSs completed teleconferencing training, including a skills assessment using the equipment for real-time, high-quality video streaming over an encrypted network to provide services in a patient’s home or other remote locations. VRS staff provided veterans with instructions on using a VA-provided tablet or their own device and assisted them with establishing connectivity with the network. Video equipment included speakers, camera, and headphones connected to the desktop computer or laptop of the VRS. A patient’s first VRtele
Demographic data, primary diagnosis, VR usage data, and zip codes of participating veterans were extracted from the EHR. Veterans completed a 2-part satisfaction survey administered 90 days after enrollment and at discharge. Part 1 was composed of 15 items, most with a 5-point Likert scale (higher ratings indicated greater satisfaction), on various aspects of the VRtele experience, such as audio and video quality and wait times.10 Part 2 addressed VR services and the VRS and consisted of 8 Likert scale items with the option to add a comment for each and 2 open-ended items that asked the participant to list what they liked best and least about VRtele.
Semistructured, in-person 30- to 60-minute interviews were conducted with VRSs at the initiation of VRtele
After ≥ 2 months of VRtele use
Analyses
Descriptive statistics were used for EHR data and satisfaction surveys. For qualitative analysis, each transcript was read in full by 2 researchers to get an overview of the data, and a rapid analysis approach was used to identify central themes focused on how technology was used and the experiences of the participants.11,12 Relevant text for each topic was tabulated, and a summary table was created that highlighted overlapping ideas discussed by the interviewees as well as differences.
Results
Of the 22 veterans who participated in the project, 11 completed satisfaction surveys and 4 participated in qualitative interviews. The rural and nonrural groups did not differ demographically or by diagnosis, which was predominantly mental health related. Only 1 veteran in each group owned a tablet; the majority of both groups required VA-issued devices: 80% (n = 8) rural and 91.7% (n = 11) nonrural. The number of VRtele sessions for the groups also was similar, 53 for rural and 60 for nonrural, as was the mean (SD) number of sessions per veteran: 5.3 (SD, 3.2) rural and 5.0 (SD, 2.5) urban. Overall, 63 miles per session were saved, mostly for rural veterans, and the number of mean (SD) miles saved per veteran was greater for rural than nonrural veterans: 379.2 (243.0) and 256.1 (275.9), respectively. One veteran who moved to a different state during the program continued VRtele at the new location. In a qualitative sampling of 5 VRtele sessions, all the VRSs used office desktop computers.
Level of satisfaction with aspects of VRtele related to the technology rated was consistently > 4 on the Likert scale. The lowest mean (SD) ratings were 4.2 (1.0) for audio quality and 4.4 (0.5) for video quality, and the highest rating was given for equipment operation explanation and privacy was respected, 4.9 (0.3) for both. All questions related to satisfaction with services were also rated high: The mean (SD) lowest ratings were 4.3 (1.0) given to both vocational needs 4.3 (1.0) and tasks effectively helped achieve goals 4.3 (0.7). The highest mean (SD) ratings were 4.6 (0.5) given to VR program service explained and 4.7 (0.5) for appointment timeliness.
Qualitative Results
At first, some VRSs thought the teleconferencing system might be difficult or awkward to use, but they found it easier to set up than expected and seamless to use. VRS staff reported being surprised at how well it worked despite some issues that occurred with loading the software. Once loaded, however, the connection worked well, one VRS noting that following step-by-step instructions solved the problem. Some VRSs indicated they did not invite all the veterans on their caseload to participate in VRtele due to concerns with the patient’s familiarity with technology, but one VRS stated, “I haven’t had anybody that failed to do a [session] that I couldn’t get them up and running within a few minutes.”
When working in the community, VRSs reported using laptops for VRtele but found that these devices were unreliable due to lack of internet access and were slow to start; several VRSs thought tablets would have been more helpful. Some veterans reported technical glitches, lack of comfort with technology, or a problem with sound due to a tablet’s protective case blocking the speakers. To solve the sound issue, a veteran used headphones. This veteran also explained that the log-on process required a new password every time, so he would keep a pen and paper ready to write it down. Because signing in and setting up takes a little time, this veteran and his VRS agreed to start connecting 5 minutes before their meeting time to allow for that set- up time.
Initially, some VRSs expressed concern that transitioning to VRtele would affect the quality of interactions with the veterans. However, VRSs also identified strengths of VRtele, including flexibility, saved time, and increased interaction. One VRS discussed a veteran’s adaptation by saying, “I think he feels even more involved in his plan [and] enjoys the increased interaction.” Veterans reported enjoying using tablets and identified the main strength of VRtele as being able to talk face-to-face with the VRS. Echoing the VRSs, veterans reported teleconferencing saved time by avoiding travel and enabled spontaneous meetings. One of the veterans summed up the benefits of using VRtele: “I’d rather just connect. It’s going to take us 40 to 50 minutes [to meet in person] when we can just connect right here and it takes 15 to 20. We don’t have to go through the driving.… So this right here, doing it ahead of time and having the appointment, it’s a lot easier.”
In their interviews, VRSs talked about enjoying VRtele. A VRS explained: “It makes it a lot easier. It makes me feel less guilty. This way [veterans] don’t have to use their gas money, use their time. I know [the veteran] had something else he needed to do today.” Thus, both veterans and VRSs were satisfied with their VRtele experiences.
Discussion
This first report on the perspective of providers and veterans using VRtele suggests that it is a viable option for service delivery and that is highly satisfactory for serving veterans with disabilities, many of whom live in rural areas or have travel barriers. These findings are consistent with data on telerehabilitation for veterans with cognitive, physical, and mental disabilities.13-22 Further, the data support the notion of using VRtele to facilitate long-term VR follow-up for persons with disabilities, as illustrated by successful continuation of vocational services after a veteran moved out of state.23
Similar to other reports, our experience highlighted 2 factors that affect successful VRtele: (1) Troubleshooting technology barriers for both VR providers and clients; and (2) supportive leadership to facilitate implementation
Changes to technology and increased usage of VA Video Connect may indicate that the barriers identified from the earlier process described here have been diminished or eliminated. More evaluation is needed to assess whether system upgrades have increased ease of use and access for veterans with disabilities.
Conclusions
Encouragingly, this clinical demonstration project showed that both providers and clients recognize the benefits of VRtele. Patient satisfaction and decreased travel costs were clear advantages to using VRtele for this small group of veterans who had barriers to care due to travel or disability barriers. As this program evaluation was limited by a small sample, absence of a comparison group, and lack of outcome data (eg, employment rates, hours, wages, retention), future research is needed on implementation and outcomes of VRtele
Acknowledgments
The authors thank Lynn Dirk, MAMC, for substantial editorial assistance. This material was based on work supported by Rural Veterans Supported Employment TeleRehabilitation Initiative (RVSETI), funded by the VA Office of Rural Health (Project # N08-FY14Q3-S2-P01222) and by support of the VA Health Services Research and Development Service. This work was presented in part at the 114th Annual Meeting of the American Anthropological Association at Denver, Colorado, November 21, 2015; a field-based Health Services Research and Development Service meeting, US Department of Veterans Affairs at Washington, DC, September 12, 2016; and the 2016 Annual Conference of the American Congress for Rehabilitation Medicine at Chicago, Illinois, October-November 2016.
1. Abraham KM, Yosef M, Resnick SG, Zivin K. Competitive employment outcomes among veterans in VHA therapeutic and supported employment services programs. Psychiatr Serv. 2017;68(9):938-946. doi:10.1176/appi.ps.201600412
2. Davis LL, Kyriakides TC, Suris AM, et al. Effect of evidence-based supported employment vs transitional work on achieving steady work among veterans with posttraumatic stress disorder: a randomized clinical trial. JAMA Psychiatry. 2018;75(4):316. doi:10.1001/jamapsychiatry.2017.4472
3. Ottomanelli L, Goetz LL, Suris A, et al. Effectiveness of supported employment for veterans with spinal cord injuries: results from a randomized multisite study. Arch Phys Med Rehabil. 2012;93(5):740-747. doi:10.1016/j.apmr.2012.01.002
4. Ottomanelli L, Goetz LL, Barnett SD, et al. Individual placement and support in spinal cord injury: a longitudinal observational study of employment outcomes. Arch Phys Med Rehabil. 2017;98(8):1567-1575. doi:10.1016/j.apmr.2016.12.010
5. Cotner BA, Ottomanelli L, O’Connor DR, Njoh EN, Barnett SD, Miech EJ. Quality of life outcomes for veterans with spinal cord injury receiving individual placement and support (IPS). Top Spinal Cord Inj Rehabil. 2018;24(4):325-335. doi:10.1310/sci17-00046
6. Metzel DS, Giordano A. Locations of employment services and people with disabilities: a geographical analysis of accessibility. J Disabil Policy Stud. 2007;18(2):88-97. doi:10.1177/10442073070180020501
7. Landon T, Connor A, McKnight-Lizotte M, Peña J. Rehabilitation counseling in rural settings: a phenomenological study on barriers and supports. J Rehabil. 2019;85(2):47-57.
8. Riemer-Reiss M. Vocational rehabilitation counseling at a distance: Challenges, strategies and ethics to consider. J Rehabil. 2000;66(1):11-17.
9. Schmeler MR, Schein RM, McCue M, Betz K. Telerehabilitation clinical and vocational applications for assistive technology: research, opportunities, and challenges. Int J Telerehabilitation. 2009;1(1):59-72.
10. Levy CE, Silverman E, Jia H, Geiss M, Omura D. Effects of physical therapy delivery via home video telerehabilitation on functional and health-related quality of life outcomes. J Rehabil Res Dev. 2015;52(3):361-370. doi:10.1682/JRRD.2014.10.0239
11. McMullen CK, Ash JS, Sittig DF, et al. Rapid assessment of clinical information systems in the healthcare setting: an efficient method for time-pressed evaluation. Methods Inf Med. 2011;50(4):299-307. doi:10.3414/ME10-01-0042
12. Averill JB. Matrix analysis as a complementary analytic strategy in qualitative inquiry. Qual Health Res. 2002;12(6):855-866.
13. Egede LE, Acierno R, Knapp RG, et al. Psychotherapy for depression in older veterans via telemedicine: a randomised, open-label, non-inferiority trial. Lancet Psychiatry. 2015;2(8):693-701. doi:10.1016/S2215-0366(15)00122-4
14. Fortney JC, Pyne JM, Edlund MJ, et al. A randomized trial of telemedicine-based collaborative care for depression. J Gen Intern Med. 2007;22(8):1086-1093. doi:10.1007/s11606-007-0201-9
15. Fortney JC, Pyne JM, Kimbrell TA, et al. Telemedicine-based collaborative care for posttraumatic stress disorder: a randomized clinical trial. JAMA Psychiatry. 2015;72(1):58. doi:10.1001/jamapsychiatry.2014.1575
16. Grubbs KM, Fortney JC, Dean T, Williams JS, Godleski L. A comparison of mental health diagnoses treated via interactive video and face to face in the Veterans Healthcare Administration. Telemed E-Health. 2015;21(7):564-566. doi:10.1089/tmj.2014.0152
17. Agostini M, Moja L, Banzi R, et al. Telerehabilitation and recovery of motor function: a systematic review and meta-analysis. J Telemed Telecare. 2015;21(4):202-213. doi:10.1177/1357633X15572201
18. Bergquist TF, Thompson K, Gehl C, Munoz Pineda J. Satisfaction ratings after receiving internet-based cognitive rehabilitation in persons with memory impairments after severe acquired brain injury. Telemed E-Health. 2010;16(4):417-423. doi:10.1089/tmj.2009.0118
19. Brennan DM, Georgeadis AC, Baron CR, Barker LM. The effect of videoconference-based telerehabilitation on story retelling performance by brain-injured subjects and its implications for remote speech-language therapy. Telemed J E Health. 2004;10(2):147-154. doi:10.1089/tmj.2004.10.147
20. Dallolio L, Menarini M, China S, et al. Functional and clinical outcomes of telemedicine in patients with spinal cord injury. Arch Phys Med Rehabil. 2008;89(12):2332-2341. doi:10.1016/j.apmr.2008.06.012
21. Houlihan BV, Jette A, Friedman RH, et al. A pilot study of a telehealth intervention for persons with spinal cord dysfunction. Spinal Cord. 2013;51(9):715-720.doi:10.1038/sc.2013.45
22. Smith MW, Hill ML, Hopkins KL, Kiratli BJ, Cronkite RC. A modeled analysis of telehealth methods for treating pressure ulcers after spinal cord injury. Int J Telemed Appl. 2012;2012:1-10. doi:10.1155/2012/729492
23. Balcazar FE, Keys CB, Davis M, Lardon C, Jones C. Strengths and challenges of intervention research in vocational rehabilitation: an illustration of agency-university collaboration. J Rehabil. 2005;71(2):40-48.
24. Martinez RN, Hogan TP, Balbale S, et al. Sociotechnical perspective on implementing clinical video telehealth for veterans with spinal cord injuries and disorders. Telemed J E Health. 2017;23(7):567-576. doi:10.1089/tmj.2016.0200
25. Martinez RN, Hogan TP, Lones K, et al. Evaluation and treatment of mild traumatic brain injury through the implementation of clinical video telehealth: provider perspectives from the Veterans Health Administration. PM R. 2017;9(3):231-240. doi:10.1016/j.pmrj.2016.07.002
26. Smith AC, Thomas E, Snoswell CL, et al. Telehealth for global emergencies: implications for coronavirus disease 2019 (COVID-19). J Telemed Telecare. 2020;26(5):309-313. doi:10.1177/1357633X20916567
27. Cowper-Ripley DC, Jia H, Wang X, et al. Trends in VA telerehabilitation patients and encounters over time and by rurality. Fed Pract. 2019; 36(3):122-128.
28. US Department of Veterans Affairs. Veterans VA Video Connect. Published May 22, 2020. Accessed May 29, 2020. https://mobile.va.gov/app/va-video-connect#AppDescription.
29. US Department of Veterans Affairs. VA telehealth at home. Accessed May 29, 2020. https://telehealth.va.gov/type/home
1. Abraham KM, Yosef M, Resnick SG, Zivin K. Competitive employment outcomes among veterans in VHA therapeutic and supported employment services programs. Psychiatr Serv. 2017;68(9):938-946. doi:10.1176/appi.ps.201600412
2. Davis LL, Kyriakides TC, Suris AM, et al. Effect of evidence-based supported employment vs transitional work on achieving steady work among veterans with posttraumatic stress disorder: a randomized clinical trial. JAMA Psychiatry. 2018;75(4):316. doi:10.1001/jamapsychiatry.2017.4472
3. Ottomanelli L, Goetz LL, Suris A, et al. Effectiveness of supported employment for veterans with spinal cord injuries: results from a randomized multisite study. Arch Phys Med Rehabil. 2012;93(5):740-747. doi:10.1016/j.apmr.2012.01.002
4. Ottomanelli L, Goetz LL, Barnett SD, et al. Individual placement and support in spinal cord injury: a longitudinal observational study of employment outcomes. Arch Phys Med Rehabil. 2017;98(8):1567-1575. doi:10.1016/j.apmr.2016.12.010
5. Cotner BA, Ottomanelli L, O’Connor DR, Njoh EN, Barnett SD, Miech EJ. Quality of life outcomes for veterans with spinal cord injury receiving individual placement and support (IPS). Top Spinal Cord Inj Rehabil. 2018;24(4):325-335. doi:10.1310/sci17-00046
6. Metzel DS, Giordano A. Locations of employment services and people with disabilities: a geographical analysis of accessibility. J Disabil Policy Stud. 2007;18(2):88-97. doi:10.1177/10442073070180020501
7. Landon T, Connor A, McKnight-Lizotte M, Peña J. Rehabilitation counseling in rural settings: a phenomenological study on barriers and supports. J Rehabil. 2019;85(2):47-57.
8. Riemer-Reiss M. Vocational rehabilitation counseling at a distance: Challenges, strategies and ethics to consider. J Rehabil. 2000;66(1):11-17.
9. Schmeler MR, Schein RM, McCue M, Betz K. Telerehabilitation clinical and vocational applications for assistive technology: research, opportunities, and challenges. Int J Telerehabilitation. 2009;1(1):59-72.
10. Levy CE, Silverman E, Jia H, Geiss M, Omura D. Effects of physical therapy delivery via home video telerehabilitation on functional and health-related quality of life outcomes. J Rehabil Res Dev. 2015;52(3):361-370. doi:10.1682/JRRD.2014.10.0239
11. McMullen CK, Ash JS, Sittig DF, et al. Rapid assessment of clinical information systems in the healthcare setting: an efficient method for time-pressed evaluation. Methods Inf Med. 2011;50(4):299-307. doi:10.3414/ME10-01-0042
12. Averill JB. Matrix analysis as a complementary analytic strategy in qualitative inquiry. Qual Health Res. 2002;12(6):855-866.
13. Egede LE, Acierno R, Knapp RG, et al. Psychotherapy for depression in older veterans via telemedicine: a randomised, open-label, non-inferiority trial. Lancet Psychiatry. 2015;2(8):693-701. doi:10.1016/S2215-0366(15)00122-4
14. Fortney JC, Pyne JM, Edlund MJ, et al. A randomized trial of telemedicine-based collaborative care for depression. J Gen Intern Med. 2007;22(8):1086-1093. doi:10.1007/s11606-007-0201-9
15. Fortney JC, Pyne JM, Kimbrell TA, et al. Telemedicine-based collaborative care for posttraumatic stress disorder: a randomized clinical trial. JAMA Psychiatry. 2015;72(1):58. doi:10.1001/jamapsychiatry.2014.1575
16. Grubbs KM, Fortney JC, Dean T, Williams JS, Godleski L. A comparison of mental health diagnoses treated via interactive video and face to face in the Veterans Healthcare Administration. Telemed E-Health. 2015;21(7):564-566. doi:10.1089/tmj.2014.0152
17. Agostini M, Moja L, Banzi R, et al. Telerehabilitation and recovery of motor function: a systematic review and meta-analysis. J Telemed Telecare. 2015;21(4):202-213. doi:10.1177/1357633X15572201
18. Bergquist TF, Thompson K, Gehl C, Munoz Pineda J. Satisfaction ratings after receiving internet-based cognitive rehabilitation in persons with memory impairments after severe acquired brain injury. Telemed E-Health. 2010;16(4):417-423. doi:10.1089/tmj.2009.0118
19. Brennan DM, Georgeadis AC, Baron CR, Barker LM. The effect of videoconference-based telerehabilitation on story retelling performance by brain-injured subjects and its implications for remote speech-language therapy. Telemed J E Health. 2004;10(2):147-154. doi:10.1089/tmj.2004.10.147
20. Dallolio L, Menarini M, China S, et al. Functional and clinical outcomes of telemedicine in patients with spinal cord injury. Arch Phys Med Rehabil. 2008;89(12):2332-2341. doi:10.1016/j.apmr.2008.06.012
21. Houlihan BV, Jette A, Friedman RH, et al. A pilot study of a telehealth intervention for persons with spinal cord dysfunction. Spinal Cord. 2013;51(9):715-720.doi:10.1038/sc.2013.45
22. Smith MW, Hill ML, Hopkins KL, Kiratli BJ, Cronkite RC. A modeled analysis of telehealth methods for treating pressure ulcers after spinal cord injury. Int J Telemed Appl. 2012;2012:1-10. doi:10.1155/2012/729492
23. Balcazar FE, Keys CB, Davis M, Lardon C, Jones C. Strengths and challenges of intervention research in vocational rehabilitation: an illustration of agency-university collaboration. J Rehabil. 2005;71(2):40-48.
24. Martinez RN, Hogan TP, Balbale S, et al. Sociotechnical perspective on implementing clinical video telehealth for veterans with spinal cord injuries and disorders. Telemed J E Health. 2017;23(7):567-576. doi:10.1089/tmj.2016.0200
25. Martinez RN, Hogan TP, Lones K, et al. Evaluation and treatment of mild traumatic brain injury through the implementation of clinical video telehealth: provider perspectives from the Veterans Health Administration. PM R. 2017;9(3):231-240. doi:10.1016/j.pmrj.2016.07.002
26. Smith AC, Thomas E, Snoswell CL, et al. Telehealth for global emergencies: implications for coronavirus disease 2019 (COVID-19). J Telemed Telecare. 2020;26(5):309-313. doi:10.1177/1357633X20916567
27. Cowper-Ripley DC, Jia H, Wang X, et al. Trends in VA telerehabilitation patients and encounters over time and by rurality. Fed Pract. 2019; 36(3):122-128.
28. US Department of Veterans Affairs. Veterans VA Video Connect. Published May 22, 2020. Accessed May 29, 2020. https://mobile.va.gov/app/va-video-connect#AppDescription.
29. US Department of Veterans Affairs. VA telehealth at home. Accessed May 29, 2020. https://telehealth.va.gov/type/home
Outcomes Associated With Pharmacist- Led Consult Service for Opioid Tapering and Pharmacotherapy
In the late 1980s and early 1990s, an emphasis on better pain management led health care professionals (HCPs) to increase prescribing of opioids to better manage patient’s pain. In 1991, 76 million prescriptions were written for opioids in the United States, and by 2011, the number had nearly tripled to 219 million.1 Overdose rates increased as well, nearly tripling from 1999 to 2014.2 Of the 52,404 US deaths from drug overdoses in the in 2015, 63% involved an opioid.2
Opioid Safety Initiative
In response to the growing opioid epidemic, the US Department of Veterans Affairs (VA) created the Opioid Safety Initiative in 2014.3 This comprehensive, multifaceted initiative was designed to improve the care and safety of veterans managed with opioid therapy and promote rational opioid prescribing and monitoring. In 2016 the Centers for Disease Control and Prevention (CDC) issued guidelines for opioid prescriptions, and the following year the VA and the US Department of Defense (DoD) updated the VA/DoD Clinical Practice Guidelines for Opioid Therapy for Chronic Pain (VA/DoD guidelines).4,5 After the release of these guidelines, the use of opioid tapers expanded. However, due to public outcry of forced opioid tapering in 2019, the US Food and Drug Administration updated its opioid labeling requirements to provide clearer guidance on opioid tapers for tolerant patients.6,7
As a result, HCPs began to develop various strategies to balance the safety and efficacy of opioid use in patients with chronic pain. The West Palm Beach VA Medical Center (WPBVAMC) in Florida has a Pain Clinic that includes 2 pain management clinical pharmacy specialists (CPSs) with specialized training in pain management, who are uniquely qualified to assess and evaluate medication therapy in complex pain patient cases. These CPSs were involved in the face-to-face management of patients requiring specialized pain care and participated in a pain pharmacy electronic consult (eConsult) service to document pain management consultative recommendations for patients appropriate for management at the primary care level. This formalized process increased specialty pain care access for veterans whose pain was managed by primary care providers (PCPs).
The pain pharmacy eConsult service was initiated at the WPBVAMC in June 2013 to assist PCPs in the management of outpatients with chronic pain. The eConsult service includes evaluation of a patient’s electronic health records (EHRs) by CPSs. The eConsult service also provided PCPs with the option to engage a pharmacist who could provide recommendations for opioid dosing conversion, opioid tapering, pain pharmacotherapy, or drug screen interpretation, without the necessity for an additional patient visit.
Subsequent to the release of the 2016 CDC (and later the 2017 VA/DoD) guidelines recommending reducing morphine equivalent daily dose (MEDD) levels, the WPBVAMC had a large increase in pain eConsult requests for opioid tapering and opioid pharmacotherapy. A 3.4-fold increase in requests occurred in March, April, and May vs the following 9 months, and a nearly 4-fold increase in requests for opioid tapers during the same period. However, the impact of the completed eConsults was unclear. Therefore, the primary objective of this study was to assess the effect of CPS services for opioid tapering and opioid pharmacotherapy by quantifying the number of recommendations accepted/implemented by PCPs. The secondary objectives included evaluating harms associated with the recommendations (eg, increase in visits to the emergency department [ED], hospitalizations, suicide attempts, or PCP visits) and provider satisfaction.
Methods
A retrospective chart review was completed to assess data of patients from the WPBVAMC and its associated community-based outpatient clinics (CBOCs). The project was approved by the WPBVAMC Scientific Advisory Committee as part of the facility’s performance improvement efforts.
Included patients had a pain pharmacy eConsult placed between April 1, 2016 and March 31, 2017. EHRs were reviewed and only eConsults for opioid pharmacotherapy recommendation or opioid tapers were evaluated. eConsults were excluded if the request was discontinued, completed by a HCP other than the pain CPS, or placed for an opioid dose conversion, nonopioid pharmacotherapy, or drug screen interpretation.
Data for analyses were entered into Microsoft Excel 2016 and were securely saved and accessible to relevant researchers. Patient protected health information used during patient care remained confidential.
Demographic data were collected, including age, gender, race, pertinent medical comorbidities (eg, diabetes mellitus, sleep apnea), and mental health comorbidities. Pain scores were collected at baseline and 6-months postconsult. Pain medications used by patients were noted at baseline and 6 months postconsult, including concomitant opioid and benzodiazepine use, MEDD, and other pain medication. The duration of time needed by pain CPS to complete each eConsult and total time from eConsult entered to HCP implementation of the initial recommendation was collected. The number of actionable recommendations (eg, changes in drug therapy, urine drug screens [UDSs], and referrals to other services also were recorded and reviewed 6 months postconsult to determine the number and percentage of recommendations implemented by the HCP. The EHR was examined to determine adverse events (AEs) (eg, any documentation of suicide attempt, calls to the Veterans Crisis Line, or death 6 month postconsult). Collected data also included new eConsults, the reason for opioid tapering either by HCP or patient, and assessment of economic harms (count of the number of visits to ED, hospitalizations, or unscheduled PCP visits with uncontrolled pain as chief reason within 6 months postconsult). Last, PCPs were sent a survey to assess their satisfaction with the pain eConsult service.
Results
Of 517 eConsults received from April 1, 2016 to March 31, 2017, 285 (55.1%) met inclusion criteria (Figure). Using a random number generator, 100 eConsults were further reviewed for outcomes of interest.
In this cohort, the mean age was 61 years, 87% were male, and 80% were White individuals. Most patients (83%) had ≥ 1 mental health comorbidity, and 53% had ≥ 2, with depressive symptoms, tobacco use, and/or posttraumatic stress disorder the most common diagnoses (Table 1). Eighty-seven percent of eConsults were for opioid tapers and the remaining 13% were for opioid pharmacotherapy.
The median pain score at time of consult was 6 on a 10-point scale, with no change at 6 months postconsult. However, 41% of patients overall had a median 3.3-point drop in pain score, 17% had no change in pain score, and 42% had a median 2.6-point increase in pain score.
At time of consult, 24% of patients had an opioid and benzodiazepine prescribed concurrently. At the time of the initial request, the mean MEDD was 177.5 mg (median, 165; range, 0-577.5). At 6 months postconsult, the average MEDD was 71 mg (median, 90; range, 0-450) for a mean 44% MEDD decrease. Eighteen percent of patients had no change in MEDD, and 5% had an increase.
One concern was the number of patients whose pain management regimen consisted of either opioids as monotherapy or a combination of opioids and skeletal muscle relaxants (SMRs), which can increase the opioid overdose risk and are not indicated for long-term use (except for baclofen for spasticity). Thirty-five percent of patients were taking either opioid monotherapy or opioids and SMRs for chronic pain management at time of consult and 28% were taking opioid monotherapy or opioids and SMRs 6 months postconsult.
Electronic Consults
Table 2 describes the reasons eConsults were requested. The most common reason was to taper the dose to be in compliance with the CDC 2016 guideline recommendation of MEDD < 90 mg, which was later increased to 100 mg by the VA/DoD guideline.
On average, eConsults were completed within a mean of 11.5 days of the PCP request, including nights and weekends. The CPS spent a mean 66.8 minutes to complete each eConsult. Once the eConsult was completed, PCPs took a mean of 9 days to initiate the primary recommendation. This 9-day average does not include 11 eConsults with no accepted recommendations and 11 eConsults for which the PCP implemented the primary recommendation before the CPS completed the consult, most likely due to a phone call or direct contact with the CPS at the time the eConsult was ordered.
A mean 3.5 actionable recommendations were made by the CPS and a mean 1.6 recommendations were implemented within 6 months by the PCP. At least 1 recommendation was accepted/implemented for 89% of patients, with a mean 55% recommendations that were accepted/implemented. Eleven percent of the eConsult final recommendations were not accepted by PCPs and clear documentation of the reasons were not provided.
Adverse Outcomes
In the 6 months postconsult, 11 patients (7 men and 4 women) experienced 32 AEs (Table 3). Eight patients had 15 ED visits, with 3 of the visits resulting in hospitalizations, 8 patients had 9 unscheduled PCP visits, 1 patient reported suicidal ideation and 2 patients made a total of 4 calls to the Veterans Crisis Line. There were also 2 deaths; however, both were due to end-stage disease (cirrhosis and amyotrophic lateral sclerosis) and not believed to be related to eConsult recommendations.
Eight patients had a history of substance use disorders (SUDs) and 8 had a history of a mood disorder or psychosis. One patient had both SUD and a mood/psychosis-related mental health disorder, including a reported suicidal attempt/ideation at an ED visit and a subsequent hospitalization. A similar number of AEs occurred in patients with decreases in MEDD of 0 to 24% compared with those that received more aggressive tapers of 75 to 100% (Table 4).
Nine patients were reconsulted, with only 1 secondary to the PCP not implementing recommendations from the initial consult. No factors were found that correlated with likelihood of a patient being reconsulted.
Surveys on PCP satisfaction with the eConsult service were completed by 29 of the 55 PCPs. PCP feedback was generally positive with nearly 90% of PCPs planning to use the service in the future as well as recommending use to other providers.
PCPs also were given the option to indicate the most important factor for overall satisfaction with eConsult service (time, access, safety, expectations or confidence). Safety was provider’s top choice with time being a close second.
Discussion
Most (89%) PCPs accepted at least 1 recommendation from the completed eConsult, and MEDDs decreased by 60%, likely reducing the patient’s risk of overdose or other AEs from opioids. There also was a slight reduction in patient’s mean pain scores; however, 41% had a decrease and 42% had an increase in pain scores. There was no clear relationship when pain scores were compared with MEDDs, likely giving credence to the idea that pain scores are largely subjective and an unreliable surrogate marker for assessing effectiveness of analgesic regimens.
Eleven patients experienced AEs, including 1 patient for whom the recommendations were not implemented by the PCP. Eight of the 11 had multiple AEs. One interesting finding was that 7 of the 11 patients with an AE tested positive for unexpected substances on routine UDS or were arrested for driving while intoxicated (DWI). However, only 3 of the 7 had an active SUD diagnosis. With 25% of the AEs coming from patients with a history of SUD, it is important that any history of SUD be documented in the EHR. Maintaining this documentation can be especially difficult if patients switch VA medical centers or receive services outside the VA. Thorough and accurate history and chart review should ideally be completed before prescribing opioids.
Guidelines
While the PCPs were following VA/DoD and CDC recommendations for opioid tapering to < 100 or 90 mg MEDD, respectively, there is weak evidence in these guidelines to support specific MEDD cutoffs. The CDC guidelines even state, “a single dosage threshold for safe opioid use could not be identified.”5 One of the largest issues when using MEDD as a cutoff is the lack of agreement on its calculation. In 2014, Nuckols and colleagues al conducted a study to compare the existing guidelines on the use of opioids for chronic pain. While 13 guidelines were considered eligible, most recommendations were supported only by observational data or expert recommendations, and there was no consensus on what constitutes a “morphine equivalent.”8 Currently there is no universally accepted opioid-conversion method, resulting in a substantial problem when calculating a MEDD.9 A survey of 8 online opioid dose conversion tools found a -55% to +242% variation.10 As Fudin and colleagues concluded in response to the large variations found in these various analyses, the studies “unequivocally disqualify the validity of embracing MEDD to assess risk in any meaningful statistical way.”11 Pharmacogenetics, drug tolerance, drug-drug interactions, body surface area, and organ function are patient- specific factors that are not taken into consideration when relying solely on a MEDD calculation. Tapering to lowest functional dose rather than a specific number or cutoff may be a more effective way to treat patients, and providers should use the guidelines as recommendations and not a hardline mandate.
At 6 months, 6 patients were receiving no pain medications from the VA, and 24 of the patients were tapered from their opiate to discontinuation. It is unclear whether patients are no longer taking opioids or switched their care to non-VA providers to receive medications, including opioids, privately. This is difficult to verify, though a prescription drug monitoring program (PDMP) could be used to assess patient adherence. As many of the patients that were tapered due to identification of aberrant behaviors, lack of continuity of care across health care systems may result in future patient harm.
The results of this analysis highlight the importance of checking PDMP databases and routine UDSs when prescribing opioids—there can be serious safety concerns if patients are taking other prescribed or illicit medications. However, care must be taken; there were 2 instances of patients’ chronic opioid prescriptions discontinued by their VA provider after a review of the PDMP showed they had received non-VA opioids. In both cases, the quantity and doses received were small (counts of ≤ 12) and were received more than 6 months prior to the check of the PDMP. While this constitutes a breach of the Informed Consent for long-term opioid use, if there are no other concerning behaviors, it may be more prudent to review the informed consent with the patient and discuss why the behavior is a breach to ensure that patients and PCPs continue to work as a team to manage chronic pain.
Limitations
The study population was one limitation of this project. While data suggest that chronic pain affects women more than men, this study’s population was only 13% female. Thirty percent of the women in this study had an AE compared with only 8% of the men. Additional limitations included use of problem list for comorbidities, as lists may be inaccurate or outdated, and limiting the monitoring of AE to only 6 months. As some tapers were not initiated immediately and some taper schedules can last several months to years; therefor, outcomes may have been higher if patients were followed longer. Many of the patients with AEs had increased ED visits or unscheduled primary care visits as the tapers went on and their pain worsened, but the visits were outside the 6-month time frame for data collection. An additional weakness of this review included assessing a pain score, but not functional status, which may be a better predictor of the effectiveness of a patient’s pain management regimen. This assessment is needed in future studies for more reliable data. Finally, PCP survey results also should be viewed with caution. The current survey had only 29 respondents, and the 2014 survey had only 10 respondents and did not include CBOC providers.
Conclusion
A pain eConsult service managed by CPSs specializing in pain management can assist patients and PCPs with opioid therapy recommendations in a safe and timely manner, reducing risk of overdose secondary to high dose opioid therapy and with limited harm to patients.
1. National Institute on Drug Abuse. Increased drug availability is associated with increased use and overdose. Published June 9, 2020. Accessed February 19, 2021. https://www.drugabuse.gov/publications/research-reports/prescription-opioids-heroin/increased-drug-availability-associated-increased-use-overdose
2. Rudd RA, Seth P, David F, Scholl L. Increases in drug and opioid-involved overdose deaths - United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452. Published 2016 Dec 30.doi:10.15585/mmwr.mm655051e1
3. US Department of Veterans Affairs, Office of Inspector General. Healthcare inspection – VA patterns of dispensing take-home opioids and monitoring patients on opioid therapy. Report 14-00895-163. Published May 14, 2014. Accessed February 2, 2021. https://www.va.gov/oig/pubs/VAOIG-14-00895-163.pdf
4. US Department of Veterans Affairs, US Department of Defense, Opioid Therapy for Chronic Pain Work Group. VA/DoD clinical practice guidelines for opioid therapy for chronic pain. Version 3.0. Published December 2017. Accessed February 2, 2021. https://www.va.gov/HOMELESS/nchav/resources/docs/mental-health/substance-abuse/VA_DoD-CLINICAL-PRACTICE-GUIDELINE-FOR-OPIOID-THERAPY-FOR-CHRONIC-PAIN-508.pdf
5. Dowell D, Haegerich TM, Chou R. CDC Guideline for Prescribing Opioids for Chronic Pain - United States, 2016 [published correction appears in MMWR Recomm Rep. 2016;65(11):295]. MMWR Recomm Rep. 2016;65(1):1-49. Published 2016 Mar 18. doi:10.15585/mmwr.rr6501e1.
6. US Food and Drug Administration. (2019). FDA identifies harm reported from sudden discontinuation of opioid pain medicines and requires label changes to guide prescribers on gradual, individualized tapering. Updated April 17, 2019. Accessed February 2, 2021. https://www.fda.gov/drugs/fda-drug-safety-podcasts/fda-identifies-harm-reported-sudden-discontinuation-opioid-pain-medicines-and-requires-label-changes
7. Dowell D, Haegerich T, Chou R. No Shortcuts to Safer Opioid Prescribing. N Engl J Med. 2019;380(24):2285-2287. doi:10.1056/NEJMp1904190
8. Nuckols TK, Anderson L, Popescu I, et al. Opioid prescribing: a systematic review and critical appraisal of guidelines for chronic pain. Ann Intern Med. 2014;160(1):38-47. doi:10.7326/0003-4819-160-1-201401070-00732
9. Rennick A, Atkinson T, Cimino NM, Strassels SA, McPherson ML, Fudin J. Variability in Opioid Equivalence Calculations. Pain Med. 2016;17(5):892-898. doi:10.1111/pme.12920
10. Shaw K, Fudin J. Evaluation and comparison of online equianalgesic opioid dose conversion calculators. Pract Pain Manag. 2013;13(7):61-66.
11. Fudin J, Pratt Cleary J, Schatman ME. The MEDD myth: the impact of pseudoscience on pain research and prescribing-guideline development. J Pain Res. 2016;9:153-156. Published 2016 Mar 23. doi:10.2147/JPR.S107794
In the late 1980s and early 1990s, an emphasis on better pain management led health care professionals (HCPs) to increase prescribing of opioids to better manage patient’s pain. In 1991, 76 million prescriptions were written for opioids in the United States, and by 2011, the number had nearly tripled to 219 million.1 Overdose rates increased as well, nearly tripling from 1999 to 2014.2 Of the 52,404 US deaths from drug overdoses in the in 2015, 63% involved an opioid.2
Opioid Safety Initiative
In response to the growing opioid epidemic, the US Department of Veterans Affairs (VA) created the Opioid Safety Initiative in 2014.3 This comprehensive, multifaceted initiative was designed to improve the care and safety of veterans managed with opioid therapy and promote rational opioid prescribing and monitoring. In 2016 the Centers for Disease Control and Prevention (CDC) issued guidelines for opioid prescriptions, and the following year the VA and the US Department of Defense (DoD) updated the VA/DoD Clinical Practice Guidelines for Opioid Therapy for Chronic Pain (VA/DoD guidelines).4,5 After the release of these guidelines, the use of opioid tapers expanded. However, due to public outcry of forced opioid tapering in 2019, the US Food and Drug Administration updated its opioid labeling requirements to provide clearer guidance on opioid tapers for tolerant patients.6,7
As a result, HCPs began to develop various strategies to balance the safety and efficacy of opioid use in patients with chronic pain. The West Palm Beach VA Medical Center (WPBVAMC) in Florida has a Pain Clinic that includes 2 pain management clinical pharmacy specialists (CPSs) with specialized training in pain management, who are uniquely qualified to assess and evaluate medication therapy in complex pain patient cases. These CPSs were involved in the face-to-face management of patients requiring specialized pain care and participated in a pain pharmacy electronic consult (eConsult) service to document pain management consultative recommendations for patients appropriate for management at the primary care level. This formalized process increased specialty pain care access for veterans whose pain was managed by primary care providers (PCPs).
The pain pharmacy eConsult service was initiated at the WPBVAMC in June 2013 to assist PCPs in the management of outpatients with chronic pain. The eConsult service includes evaluation of a patient’s electronic health records (EHRs) by CPSs. The eConsult service also provided PCPs with the option to engage a pharmacist who could provide recommendations for opioid dosing conversion, opioid tapering, pain pharmacotherapy, or drug screen interpretation, without the necessity for an additional patient visit.
Subsequent to the release of the 2016 CDC (and later the 2017 VA/DoD) guidelines recommending reducing morphine equivalent daily dose (MEDD) levels, the WPBVAMC had a large increase in pain eConsult requests for opioid tapering and opioid pharmacotherapy. A 3.4-fold increase in requests occurred in March, April, and May vs the following 9 months, and a nearly 4-fold increase in requests for opioid tapers during the same period. However, the impact of the completed eConsults was unclear. Therefore, the primary objective of this study was to assess the effect of CPS services for opioid tapering and opioid pharmacotherapy by quantifying the number of recommendations accepted/implemented by PCPs. The secondary objectives included evaluating harms associated with the recommendations (eg, increase in visits to the emergency department [ED], hospitalizations, suicide attempts, or PCP visits) and provider satisfaction.
Methods
A retrospective chart review was completed to assess data of patients from the WPBVAMC and its associated community-based outpatient clinics (CBOCs). The project was approved by the WPBVAMC Scientific Advisory Committee as part of the facility’s performance improvement efforts.
Included patients had a pain pharmacy eConsult placed between April 1, 2016 and March 31, 2017. EHRs were reviewed and only eConsults for opioid pharmacotherapy recommendation or opioid tapers were evaluated. eConsults were excluded if the request was discontinued, completed by a HCP other than the pain CPS, or placed for an opioid dose conversion, nonopioid pharmacotherapy, or drug screen interpretation.
Data for analyses were entered into Microsoft Excel 2016 and were securely saved and accessible to relevant researchers. Patient protected health information used during patient care remained confidential.
Demographic data were collected, including age, gender, race, pertinent medical comorbidities (eg, diabetes mellitus, sleep apnea), and mental health comorbidities. Pain scores were collected at baseline and 6-months postconsult. Pain medications used by patients were noted at baseline and 6 months postconsult, including concomitant opioid and benzodiazepine use, MEDD, and other pain medication. The duration of time needed by pain CPS to complete each eConsult and total time from eConsult entered to HCP implementation of the initial recommendation was collected. The number of actionable recommendations (eg, changes in drug therapy, urine drug screens [UDSs], and referrals to other services also were recorded and reviewed 6 months postconsult to determine the number and percentage of recommendations implemented by the HCP. The EHR was examined to determine adverse events (AEs) (eg, any documentation of suicide attempt, calls to the Veterans Crisis Line, or death 6 month postconsult). Collected data also included new eConsults, the reason for opioid tapering either by HCP or patient, and assessment of economic harms (count of the number of visits to ED, hospitalizations, or unscheduled PCP visits with uncontrolled pain as chief reason within 6 months postconsult). Last, PCPs were sent a survey to assess their satisfaction with the pain eConsult service.
Results
Of 517 eConsults received from April 1, 2016 to March 31, 2017, 285 (55.1%) met inclusion criteria (Figure). Using a random number generator, 100 eConsults were further reviewed for outcomes of interest.
In this cohort, the mean age was 61 years, 87% were male, and 80% were White individuals. Most patients (83%) had ≥ 1 mental health comorbidity, and 53% had ≥ 2, with depressive symptoms, tobacco use, and/or posttraumatic stress disorder the most common diagnoses (Table 1). Eighty-seven percent of eConsults were for opioid tapers and the remaining 13% were for opioid pharmacotherapy.
The median pain score at time of consult was 6 on a 10-point scale, with no change at 6 months postconsult. However, 41% of patients overall had a median 3.3-point drop in pain score, 17% had no change in pain score, and 42% had a median 2.6-point increase in pain score.
At time of consult, 24% of patients had an opioid and benzodiazepine prescribed concurrently. At the time of the initial request, the mean MEDD was 177.5 mg (median, 165; range, 0-577.5). At 6 months postconsult, the average MEDD was 71 mg (median, 90; range, 0-450) for a mean 44% MEDD decrease. Eighteen percent of patients had no change in MEDD, and 5% had an increase.
One concern was the number of patients whose pain management regimen consisted of either opioids as monotherapy or a combination of opioids and skeletal muscle relaxants (SMRs), which can increase the opioid overdose risk and are not indicated for long-term use (except for baclofen for spasticity). Thirty-five percent of patients were taking either opioid monotherapy or opioids and SMRs for chronic pain management at time of consult and 28% were taking opioid monotherapy or opioids and SMRs 6 months postconsult.
Electronic Consults
Table 2 describes the reasons eConsults were requested. The most common reason was to taper the dose to be in compliance with the CDC 2016 guideline recommendation of MEDD < 90 mg, which was later increased to 100 mg by the VA/DoD guideline.
On average, eConsults were completed within a mean of 11.5 days of the PCP request, including nights and weekends. The CPS spent a mean 66.8 minutes to complete each eConsult. Once the eConsult was completed, PCPs took a mean of 9 days to initiate the primary recommendation. This 9-day average does not include 11 eConsults with no accepted recommendations and 11 eConsults for which the PCP implemented the primary recommendation before the CPS completed the consult, most likely due to a phone call or direct contact with the CPS at the time the eConsult was ordered.
A mean 3.5 actionable recommendations were made by the CPS and a mean 1.6 recommendations were implemented within 6 months by the PCP. At least 1 recommendation was accepted/implemented for 89% of patients, with a mean 55% recommendations that were accepted/implemented. Eleven percent of the eConsult final recommendations were not accepted by PCPs and clear documentation of the reasons were not provided.
Adverse Outcomes
In the 6 months postconsult, 11 patients (7 men and 4 women) experienced 32 AEs (Table 3). Eight patients had 15 ED visits, with 3 of the visits resulting in hospitalizations, 8 patients had 9 unscheduled PCP visits, 1 patient reported suicidal ideation and 2 patients made a total of 4 calls to the Veterans Crisis Line. There were also 2 deaths; however, both were due to end-stage disease (cirrhosis and amyotrophic lateral sclerosis) and not believed to be related to eConsult recommendations.
Eight patients had a history of substance use disorders (SUDs) and 8 had a history of a mood disorder or psychosis. One patient had both SUD and a mood/psychosis-related mental health disorder, including a reported suicidal attempt/ideation at an ED visit and a subsequent hospitalization. A similar number of AEs occurred in patients with decreases in MEDD of 0 to 24% compared with those that received more aggressive tapers of 75 to 100% (Table 4).
Nine patients were reconsulted, with only 1 secondary to the PCP not implementing recommendations from the initial consult. No factors were found that correlated with likelihood of a patient being reconsulted.
Surveys on PCP satisfaction with the eConsult service were completed by 29 of the 55 PCPs. PCP feedback was generally positive with nearly 90% of PCPs planning to use the service in the future as well as recommending use to other providers.
PCPs also were given the option to indicate the most important factor for overall satisfaction with eConsult service (time, access, safety, expectations or confidence). Safety was provider’s top choice with time being a close second.
Discussion
Most (89%) PCPs accepted at least 1 recommendation from the completed eConsult, and MEDDs decreased by 60%, likely reducing the patient’s risk of overdose or other AEs from opioids. There also was a slight reduction in patient’s mean pain scores; however, 41% had a decrease and 42% had an increase in pain scores. There was no clear relationship when pain scores were compared with MEDDs, likely giving credence to the idea that pain scores are largely subjective and an unreliable surrogate marker for assessing effectiveness of analgesic regimens.
Eleven patients experienced AEs, including 1 patient for whom the recommendations were not implemented by the PCP. Eight of the 11 had multiple AEs. One interesting finding was that 7 of the 11 patients with an AE tested positive for unexpected substances on routine UDS or were arrested for driving while intoxicated (DWI). However, only 3 of the 7 had an active SUD diagnosis. With 25% of the AEs coming from patients with a history of SUD, it is important that any history of SUD be documented in the EHR. Maintaining this documentation can be especially difficult if patients switch VA medical centers or receive services outside the VA. Thorough and accurate history and chart review should ideally be completed before prescribing opioids.
Guidelines
While the PCPs were following VA/DoD and CDC recommendations for opioid tapering to < 100 or 90 mg MEDD, respectively, there is weak evidence in these guidelines to support specific MEDD cutoffs. The CDC guidelines even state, “a single dosage threshold for safe opioid use could not be identified.”5 One of the largest issues when using MEDD as a cutoff is the lack of agreement on its calculation. In 2014, Nuckols and colleagues al conducted a study to compare the existing guidelines on the use of opioids for chronic pain. While 13 guidelines were considered eligible, most recommendations were supported only by observational data or expert recommendations, and there was no consensus on what constitutes a “morphine equivalent.”8 Currently there is no universally accepted opioid-conversion method, resulting in a substantial problem when calculating a MEDD.9 A survey of 8 online opioid dose conversion tools found a -55% to +242% variation.10 As Fudin and colleagues concluded in response to the large variations found in these various analyses, the studies “unequivocally disqualify the validity of embracing MEDD to assess risk in any meaningful statistical way.”11 Pharmacogenetics, drug tolerance, drug-drug interactions, body surface area, and organ function are patient- specific factors that are not taken into consideration when relying solely on a MEDD calculation. Tapering to lowest functional dose rather than a specific number or cutoff may be a more effective way to treat patients, and providers should use the guidelines as recommendations and not a hardline mandate.
At 6 months, 6 patients were receiving no pain medications from the VA, and 24 of the patients were tapered from their opiate to discontinuation. It is unclear whether patients are no longer taking opioids or switched their care to non-VA providers to receive medications, including opioids, privately. This is difficult to verify, though a prescription drug monitoring program (PDMP) could be used to assess patient adherence. As many of the patients that were tapered due to identification of aberrant behaviors, lack of continuity of care across health care systems may result in future patient harm.
The results of this analysis highlight the importance of checking PDMP databases and routine UDSs when prescribing opioids—there can be serious safety concerns if patients are taking other prescribed or illicit medications. However, care must be taken; there were 2 instances of patients’ chronic opioid prescriptions discontinued by their VA provider after a review of the PDMP showed they had received non-VA opioids. In both cases, the quantity and doses received were small (counts of ≤ 12) and were received more than 6 months prior to the check of the PDMP. While this constitutes a breach of the Informed Consent for long-term opioid use, if there are no other concerning behaviors, it may be more prudent to review the informed consent with the patient and discuss why the behavior is a breach to ensure that patients and PCPs continue to work as a team to manage chronic pain.
Limitations
The study population was one limitation of this project. While data suggest that chronic pain affects women more than men, this study’s population was only 13% female. Thirty percent of the women in this study had an AE compared with only 8% of the men. Additional limitations included use of problem list for comorbidities, as lists may be inaccurate or outdated, and limiting the monitoring of AE to only 6 months. As some tapers were not initiated immediately and some taper schedules can last several months to years; therefor, outcomes may have been higher if patients were followed longer. Many of the patients with AEs had increased ED visits or unscheduled primary care visits as the tapers went on and their pain worsened, but the visits were outside the 6-month time frame for data collection. An additional weakness of this review included assessing a pain score, but not functional status, which may be a better predictor of the effectiveness of a patient’s pain management regimen. This assessment is needed in future studies for more reliable data. Finally, PCP survey results also should be viewed with caution. The current survey had only 29 respondents, and the 2014 survey had only 10 respondents and did not include CBOC providers.
Conclusion
A pain eConsult service managed by CPSs specializing in pain management can assist patients and PCPs with opioid therapy recommendations in a safe and timely manner, reducing risk of overdose secondary to high dose opioid therapy and with limited harm to patients.
In the late 1980s and early 1990s, an emphasis on better pain management led health care professionals (HCPs) to increase prescribing of opioids to better manage patient’s pain. In 1991, 76 million prescriptions were written for opioids in the United States, and by 2011, the number had nearly tripled to 219 million.1 Overdose rates increased as well, nearly tripling from 1999 to 2014.2 Of the 52,404 US deaths from drug overdoses in the in 2015, 63% involved an opioid.2
Opioid Safety Initiative
In response to the growing opioid epidemic, the US Department of Veterans Affairs (VA) created the Opioid Safety Initiative in 2014.3 This comprehensive, multifaceted initiative was designed to improve the care and safety of veterans managed with opioid therapy and promote rational opioid prescribing and monitoring. In 2016 the Centers for Disease Control and Prevention (CDC) issued guidelines for opioid prescriptions, and the following year the VA and the US Department of Defense (DoD) updated the VA/DoD Clinical Practice Guidelines for Opioid Therapy for Chronic Pain (VA/DoD guidelines).4,5 After the release of these guidelines, the use of opioid tapers expanded. However, due to public outcry of forced opioid tapering in 2019, the US Food and Drug Administration updated its opioid labeling requirements to provide clearer guidance on opioid tapers for tolerant patients.6,7
As a result, HCPs began to develop various strategies to balance the safety and efficacy of opioid use in patients with chronic pain. The West Palm Beach VA Medical Center (WPBVAMC) in Florida has a Pain Clinic that includes 2 pain management clinical pharmacy specialists (CPSs) with specialized training in pain management, who are uniquely qualified to assess and evaluate medication therapy in complex pain patient cases. These CPSs were involved in the face-to-face management of patients requiring specialized pain care and participated in a pain pharmacy electronic consult (eConsult) service to document pain management consultative recommendations for patients appropriate for management at the primary care level. This formalized process increased specialty pain care access for veterans whose pain was managed by primary care providers (PCPs).
The pain pharmacy eConsult service was initiated at the WPBVAMC in June 2013 to assist PCPs in the management of outpatients with chronic pain. The eConsult service includes evaluation of a patient’s electronic health records (EHRs) by CPSs. The eConsult service also provided PCPs with the option to engage a pharmacist who could provide recommendations for opioid dosing conversion, opioid tapering, pain pharmacotherapy, or drug screen interpretation, without the necessity for an additional patient visit.
Subsequent to the release of the 2016 CDC (and later the 2017 VA/DoD) guidelines recommending reducing morphine equivalent daily dose (MEDD) levels, the WPBVAMC had a large increase in pain eConsult requests for opioid tapering and opioid pharmacotherapy. A 3.4-fold increase in requests occurred in March, April, and May vs the following 9 months, and a nearly 4-fold increase in requests for opioid tapers during the same period. However, the impact of the completed eConsults was unclear. Therefore, the primary objective of this study was to assess the effect of CPS services for opioid tapering and opioid pharmacotherapy by quantifying the number of recommendations accepted/implemented by PCPs. The secondary objectives included evaluating harms associated with the recommendations (eg, increase in visits to the emergency department [ED], hospitalizations, suicide attempts, or PCP visits) and provider satisfaction.
Methods
A retrospective chart review was completed to assess data of patients from the WPBVAMC and its associated community-based outpatient clinics (CBOCs). The project was approved by the WPBVAMC Scientific Advisory Committee as part of the facility’s performance improvement efforts.
Included patients had a pain pharmacy eConsult placed between April 1, 2016 and March 31, 2017. EHRs were reviewed and only eConsults for opioid pharmacotherapy recommendation or opioid tapers were evaluated. eConsults were excluded if the request was discontinued, completed by a HCP other than the pain CPS, or placed for an opioid dose conversion, nonopioid pharmacotherapy, or drug screen interpretation.
Data for analyses were entered into Microsoft Excel 2016 and were securely saved and accessible to relevant researchers. Patient protected health information used during patient care remained confidential.
Demographic data were collected, including age, gender, race, pertinent medical comorbidities (eg, diabetes mellitus, sleep apnea), and mental health comorbidities. Pain scores were collected at baseline and 6-months postconsult. Pain medications used by patients were noted at baseline and 6 months postconsult, including concomitant opioid and benzodiazepine use, MEDD, and other pain medication. The duration of time needed by pain CPS to complete each eConsult and total time from eConsult entered to HCP implementation of the initial recommendation was collected. The number of actionable recommendations (eg, changes in drug therapy, urine drug screens [UDSs], and referrals to other services also were recorded and reviewed 6 months postconsult to determine the number and percentage of recommendations implemented by the HCP. The EHR was examined to determine adverse events (AEs) (eg, any documentation of suicide attempt, calls to the Veterans Crisis Line, or death 6 month postconsult). Collected data also included new eConsults, the reason for opioid tapering either by HCP or patient, and assessment of economic harms (count of the number of visits to ED, hospitalizations, or unscheduled PCP visits with uncontrolled pain as chief reason within 6 months postconsult). Last, PCPs were sent a survey to assess their satisfaction with the pain eConsult service.
Results
Of 517 eConsults received from April 1, 2016 to March 31, 2017, 285 (55.1%) met inclusion criteria (Figure). Using a random number generator, 100 eConsults were further reviewed for outcomes of interest.
In this cohort, the mean age was 61 years, 87% were male, and 80% were White individuals. Most patients (83%) had ≥ 1 mental health comorbidity, and 53% had ≥ 2, with depressive symptoms, tobacco use, and/or posttraumatic stress disorder the most common diagnoses (Table 1). Eighty-seven percent of eConsults were for opioid tapers and the remaining 13% were for opioid pharmacotherapy.
The median pain score at time of consult was 6 on a 10-point scale, with no change at 6 months postconsult. However, 41% of patients overall had a median 3.3-point drop in pain score, 17% had no change in pain score, and 42% had a median 2.6-point increase in pain score.
At time of consult, 24% of patients had an opioid and benzodiazepine prescribed concurrently. At the time of the initial request, the mean MEDD was 177.5 mg (median, 165; range, 0-577.5). At 6 months postconsult, the average MEDD was 71 mg (median, 90; range, 0-450) for a mean 44% MEDD decrease. Eighteen percent of patients had no change in MEDD, and 5% had an increase.
One concern was the number of patients whose pain management regimen consisted of either opioids as monotherapy or a combination of opioids and skeletal muscle relaxants (SMRs), which can increase the opioid overdose risk and are not indicated for long-term use (except for baclofen for spasticity). Thirty-five percent of patients were taking either opioid monotherapy or opioids and SMRs for chronic pain management at time of consult and 28% were taking opioid monotherapy or opioids and SMRs 6 months postconsult.
Electronic Consults
Table 2 describes the reasons eConsults were requested. The most common reason was to taper the dose to be in compliance with the CDC 2016 guideline recommendation of MEDD < 90 mg, which was later increased to 100 mg by the VA/DoD guideline.
On average, eConsults were completed within a mean of 11.5 days of the PCP request, including nights and weekends. The CPS spent a mean 66.8 minutes to complete each eConsult. Once the eConsult was completed, PCPs took a mean of 9 days to initiate the primary recommendation. This 9-day average does not include 11 eConsults with no accepted recommendations and 11 eConsults for which the PCP implemented the primary recommendation before the CPS completed the consult, most likely due to a phone call or direct contact with the CPS at the time the eConsult was ordered.
A mean 3.5 actionable recommendations were made by the CPS and a mean 1.6 recommendations were implemented within 6 months by the PCP. At least 1 recommendation was accepted/implemented for 89% of patients, with a mean 55% recommendations that were accepted/implemented. Eleven percent of the eConsult final recommendations were not accepted by PCPs and clear documentation of the reasons were not provided.
Adverse Outcomes
In the 6 months postconsult, 11 patients (7 men and 4 women) experienced 32 AEs (Table 3). Eight patients had 15 ED visits, with 3 of the visits resulting in hospitalizations, 8 patients had 9 unscheduled PCP visits, 1 patient reported suicidal ideation and 2 patients made a total of 4 calls to the Veterans Crisis Line. There were also 2 deaths; however, both were due to end-stage disease (cirrhosis and amyotrophic lateral sclerosis) and not believed to be related to eConsult recommendations.
Eight patients had a history of substance use disorders (SUDs) and 8 had a history of a mood disorder or psychosis. One patient had both SUD and a mood/psychosis-related mental health disorder, including a reported suicidal attempt/ideation at an ED visit and a subsequent hospitalization. A similar number of AEs occurred in patients with decreases in MEDD of 0 to 24% compared with those that received more aggressive tapers of 75 to 100% (Table 4).
Nine patients were reconsulted, with only 1 secondary to the PCP not implementing recommendations from the initial consult. No factors were found that correlated with likelihood of a patient being reconsulted.
Surveys on PCP satisfaction with the eConsult service were completed by 29 of the 55 PCPs. PCP feedback was generally positive with nearly 90% of PCPs planning to use the service in the future as well as recommending use to other providers.
PCPs also were given the option to indicate the most important factor for overall satisfaction with eConsult service (time, access, safety, expectations or confidence). Safety was provider’s top choice with time being a close second.
Discussion
Most (89%) PCPs accepted at least 1 recommendation from the completed eConsult, and MEDDs decreased by 60%, likely reducing the patient’s risk of overdose or other AEs from opioids. There also was a slight reduction in patient’s mean pain scores; however, 41% had a decrease and 42% had an increase in pain scores. There was no clear relationship when pain scores were compared with MEDDs, likely giving credence to the idea that pain scores are largely subjective and an unreliable surrogate marker for assessing effectiveness of analgesic regimens.
Eleven patients experienced AEs, including 1 patient for whom the recommendations were not implemented by the PCP. Eight of the 11 had multiple AEs. One interesting finding was that 7 of the 11 patients with an AE tested positive for unexpected substances on routine UDS or were arrested for driving while intoxicated (DWI). However, only 3 of the 7 had an active SUD diagnosis. With 25% of the AEs coming from patients with a history of SUD, it is important that any history of SUD be documented in the EHR. Maintaining this documentation can be especially difficult if patients switch VA medical centers or receive services outside the VA. Thorough and accurate history and chart review should ideally be completed before prescribing opioids.
Guidelines
While the PCPs were following VA/DoD and CDC recommendations for opioid tapering to < 100 or 90 mg MEDD, respectively, there is weak evidence in these guidelines to support specific MEDD cutoffs. The CDC guidelines even state, “a single dosage threshold for safe opioid use could not be identified.”5 One of the largest issues when using MEDD as a cutoff is the lack of agreement on its calculation. In 2014, Nuckols and colleagues al conducted a study to compare the existing guidelines on the use of opioids for chronic pain. While 13 guidelines were considered eligible, most recommendations were supported only by observational data or expert recommendations, and there was no consensus on what constitutes a “morphine equivalent.”8 Currently there is no universally accepted opioid-conversion method, resulting in a substantial problem when calculating a MEDD.9 A survey of 8 online opioid dose conversion tools found a -55% to +242% variation.10 As Fudin and colleagues concluded in response to the large variations found in these various analyses, the studies “unequivocally disqualify the validity of embracing MEDD to assess risk in any meaningful statistical way.”11 Pharmacogenetics, drug tolerance, drug-drug interactions, body surface area, and organ function are patient- specific factors that are not taken into consideration when relying solely on a MEDD calculation. Tapering to lowest functional dose rather than a specific number or cutoff may be a more effective way to treat patients, and providers should use the guidelines as recommendations and not a hardline mandate.
At 6 months, 6 patients were receiving no pain medications from the VA, and 24 of the patients were tapered from their opiate to discontinuation. It is unclear whether patients are no longer taking opioids or switched their care to non-VA providers to receive medications, including opioids, privately. This is difficult to verify, though a prescription drug monitoring program (PDMP) could be used to assess patient adherence. As many of the patients that were tapered due to identification of aberrant behaviors, lack of continuity of care across health care systems may result in future patient harm.
The results of this analysis highlight the importance of checking PDMP databases and routine UDSs when prescribing opioids—there can be serious safety concerns if patients are taking other prescribed or illicit medications. However, care must be taken; there were 2 instances of patients’ chronic opioid prescriptions discontinued by their VA provider after a review of the PDMP showed they had received non-VA opioids. In both cases, the quantity and doses received were small (counts of ≤ 12) and were received more than 6 months prior to the check of the PDMP. While this constitutes a breach of the Informed Consent for long-term opioid use, if there are no other concerning behaviors, it may be more prudent to review the informed consent with the patient and discuss why the behavior is a breach to ensure that patients and PCPs continue to work as a team to manage chronic pain.
Limitations
The study population was one limitation of this project. While data suggest that chronic pain affects women more than men, this study’s population was only 13% female. Thirty percent of the women in this study had an AE compared with only 8% of the men. Additional limitations included use of problem list for comorbidities, as lists may be inaccurate or outdated, and limiting the monitoring of AE to only 6 months. As some tapers were not initiated immediately and some taper schedules can last several months to years; therefor, outcomes may have been higher if patients were followed longer. Many of the patients with AEs had increased ED visits or unscheduled primary care visits as the tapers went on and their pain worsened, but the visits were outside the 6-month time frame for data collection. An additional weakness of this review included assessing a pain score, but not functional status, which may be a better predictor of the effectiveness of a patient’s pain management regimen. This assessment is needed in future studies for more reliable data. Finally, PCP survey results also should be viewed with caution. The current survey had only 29 respondents, and the 2014 survey had only 10 respondents and did not include CBOC providers.
Conclusion
A pain eConsult service managed by CPSs specializing in pain management can assist patients and PCPs with opioid therapy recommendations in a safe and timely manner, reducing risk of overdose secondary to high dose opioid therapy and with limited harm to patients.
1. National Institute on Drug Abuse. Increased drug availability is associated with increased use and overdose. Published June 9, 2020. Accessed February 19, 2021. https://www.drugabuse.gov/publications/research-reports/prescription-opioids-heroin/increased-drug-availability-associated-increased-use-overdose
2. Rudd RA, Seth P, David F, Scholl L. Increases in drug and opioid-involved overdose deaths - United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452. Published 2016 Dec 30.doi:10.15585/mmwr.mm655051e1
3. US Department of Veterans Affairs, Office of Inspector General. Healthcare inspection – VA patterns of dispensing take-home opioids and monitoring patients on opioid therapy. Report 14-00895-163. Published May 14, 2014. Accessed February 2, 2021. https://www.va.gov/oig/pubs/VAOIG-14-00895-163.pdf
4. US Department of Veterans Affairs, US Department of Defense, Opioid Therapy for Chronic Pain Work Group. VA/DoD clinical practice guidelines for opioid therapy for chronic pain. Version 3.0. Published December 2017. Accessed February 2, 2021. https://www.va.gov/HOMELESS/nchav/resources/docs/mental-health/substance-abuse/VA_DoD-CLINICAL-PRACTICE-GUIDELINE-FOR-OPIOID-THERAPY-FOR-CHRONIC-PAIN-508.pdf
5. Dowell D, Haegerich TM, Chou R. CDC Guideline for Prescribing Opioids for Chronic Pain - United States, 2016 [published correction appears in MMWR Recomm Rep. 2016;65(11):295]. MMWR Recomm Rep. 2016;65(1):1-49. Published 2016 Mar 18. doi:10.15585/mmwr.rr6501e1.
6. US Food and Drug Administration. (2019). FDA identifies harm reported from sudden discontinuation of opioid pain medicines and requires label changes to guide prescribers on gradual, individualized tapering. Updated April 17, 2019. Accessed February 2, 2021. https://www.fda.gov/drugs/fda-drug-safety-podcasts/fda-identifies-harm-reported-sudden-discontinuation-opioid-pain-medicines-and-requires-label-changes
7. Dowell D, Haegerich T, Chou R. No Shortcuts to Safer Opioid Prescribing. N Engl J Med. 2019;380(24):2285-2287. doi:10.1056/NEJMp1904190
8. Nuckols TK, Anderson L, Popescu I, et al. Opioid prescribing: a systematic review and critical appraisal of guidelines for chronic pain. Ann Intern Med. 2014;160(1):38-47. doi:10.7326/0003-4819-160-1-201401070-00732
9. Rennick A, Atkinson T, Cimino NM, Strassels SA, McPherson ML, Fudin J. Variability in Opioid Equivalence Calculations. Pain Med. 2016;17(5):892-898. doi:10.1111/pme.12920
10. Shaw K, Fudin J. Evaluation and comparison of online equianalgesic opioid dose conversion calculators. Pract Pain Manag. 2013;13(7):61-66.
11. Fudin J, Pratt Cleary J, Schatman ME. The MEDD myth: the impact of pseudoscience on pain research and prescribing-guideline development. J Pain Res. 2016;9:153-156. Published 2016 Mar 23. doi:10.2147/JPR.S107794
1. National Institute on Drug Abuse. Increased drug availability is associated with increased use and overdose. Published June 9, 2020. Accessed February 19, 2021. https://www.drugabuse.gov/publications/research-reports/prescription-opioids-heroin/increased-drug-availability-associated-increased-use-overdose
2. Rudd RA, Seth P, David F, Scholl L. Increases in drug and opioid-involved overdose deaths - United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2016;65(50-51):1445-1452. Published 2016 Dec 30.doi:10.15585/mmwr.mm655051e1
3. US Department of Veterans Affairs, Office of Inspector General. Healthcare inspection – VA patterns of dispensing take-home opioids and monitoring patients on opioid therapy. Report 14-00895-163. Published May 14, 2014. Accessed February 2, 2021. https://www.va.gov/oig/pubs/VAOIG-14-00895-163.pdf
4. US Department of Veterans Affairs, US Department of Defense, Opioid Therapy for Chronic Pain Work Group. VA/DoD clinical practice guidelines for opioid therapy for chronic pain. Version 3.0. Published December 2017. Accessed February 2, 2021. https://www.va.gov/HOMELESS/nchav/resources/docs/mental-health/substance-abuse/VA_DoD-CLINICAL-PRACTICE-GUIDELINE-FOR-OPIOID-THERAPY-FOR-CHRONIC-PAIN-508.pdf
5. Dowell D, Haegerich TM, Chou R. CDC Guideline for Prescribing Opioids for Chronic Pain - United States, 2016 [published correction appears in MMWR Recomm Rep. 2016;65(11):295]. MMWR Recomm Rep. 2016;65(1):1-49. Published 2016 Mar 18. doi:10.15585/mmwr.rr6501e1.
6. US Food and Drug Administration. (2019). FDA identifies harm reported from sudden discontinuation of opioid pain medicines and requires label changes to guide prescribers on gradual, individualized tapering. Updated April 17, 2019. Accessed February 2, 2021. https://www.fda.gov/drugs/fda-drug-safety-podcasts/fda-identifies-harm-reported-sudden-discontinuation-opioid-pain-medicines-and-requires-label-changes
7. Dowell D, Haegerich T, Chou R. No Shortcuts to Safer Opioid Prescribing. N Engl J Med. 2019;380(24):2285-2287. doi:10.1056/NEJMp1904190
8. Nuckols TK, Anderson L, Popescu I, et al. Opioid prescribing: a systematic review and critical appraisal of guidelines for chronic pain. Ann Intern Med. 2014;160(1):38-47. doi:10.7326/0003-4819-160-1-201401070-00732
9. Rennick A, Atkinson T, Cimino NM, Strassels SA, McPherson ML, Fudin J. Variability in Opioid Equivalence Calculations. Pain Med. 2016;17(5):892-898. doi:10.1111/pme.12920
10. Shaw K, Fudin J. Evaluation and comparison of online equianalgesic opioid dose conversion calculators. Pract Pain Manag. 2013;13(7):61-66.
11. Fudin J, Pratt Cleary J, Schatman ME. The MEDD myth: the impact of pseudoscience on pain research and prescribing-guideline development. J Pain Res. 2016;9:153-156. Published 2016 Mar 23. doi:10.2147/JPR.S107794
Reduction of Opioid Use With Enhanced Recovery Program for Total Knee Arthroplasty
Total knee arthroplasty (TKA) is one of the most common surgical procedures in the United States. The volume of TKAs is projected to substantially increase over the next 30 years.1 Adequate pain control after TKA is critically important to achieve early mobilization, shorten the length of hospital stay, and reduce postoperative complications. The evolution and inclusion of multimodal pain-management protocols have had a major impact on the clinical outcomes for TKA patients.2,3
Pain-management protocols typically use several modalities to control pain throughout the perioperative period. Multimodal opioid and nonopioid oral medications are administered during the pre- and postoperative periods and often involve a combination of acetaminophen, gabapentinoids, and cyclooxygenase-2 inhibitors.4 Peripheral nerve blocks and central neuraxial blockades are widely used and have been shown to be effective in reducing postoperative pain as well as overall opioid consumption.5,6 Finally, intraoperative periarticular injections have been shown to reduce postoperative pain and opioid consumption as well as improve patient satisfaction scores.7-9 These strategies are routinely used in TKA with the goal of minimizing overall opioid consumption and adverse events, reducing perioperative complications, and improving patient satisfaction.
Periarticular injections during surgery are an integral part of the multimodal pain-management protocols, though no consensus has been reached on proper injection formulation or technique. Liposomal bupivacaine is a local anesthetic depot formulation approved by the US Food and Drug Administration for surgical patients. The reported results have been discrepant regarding the efficacy of using liposomal bupivacaine injection in patients with TKA. Several studies have reported no added benefit of liposomal bupivacaine in contrast to a mixture of local anesthetics.10,11 Other studies have demonstrated superior pain relief.12 Many factors may contribute to the discrepant data, such as injection techniques, infiltration volume, and the assessment tools used to measure efficacy and safety.13
The US Department of Veterans Affairs (VA) Veterans Health Administration (VHA) provides care to a large patient population. Many of the patients in that system have high-risk profiles, including medical comorbidities; exposure to chronic pain and opioid use; and psychological and central nervous system injuries, including posttraumatic stress disorder and traumatic brain injury. Hadlandsmyth and colleagues reported increased risk of prolonged opioid use in VA patients after TKA surgery.14 They found that 20% of the patients were still on long-term opioids more than 90 days after TKA.
The purpose of this study was to evaluate the efficacy of the implementation of a comprehensive enhanced recovery after surgery (ERAS) protocol at a regional VA medical center. We hypothesize that the addition of liposomal bupivacaine in a multidisciplinary ERAS protocol would reduce the length of hospital stay and opioid consumption without any deleterious effects on postoperative outcomes.
Methods
A postoperative recovery protocol was implemented in 2013 at VA North Texas Health Care System (VANTHCS) in Dallas, and many of the patients continued to have issues with satisfactory pain control, prolonged length of stay, and extended opioid consumption postoperatively. A multimodal pain-management protocol and multidisciplinary perioperative case-management protocol were implemented in 2016 to further improve the clinical outcomes of patients undergoing TKA surgery. The senior surgeon (JM) organized a multidisciplinary team of health care providers to identify and implement potential solutions. This task force met weekly and consisted of surgeons, anesthesiologists, certified registered nurse anesthetists, orthopedic physician assistants, a nurse coordinator, a physical therapist, and an occupational therapist, as well as operating room, postanesthesia care unit (PACU), and surgical ward nurses. In addition, the staff from the home health agencies and social services attended the weekly meetings.
We conducted a retrospective review of all patients who had undergone unilateral TKA from 2013 to 2018 at VANTHCS. This was a consecutive, unselected cohort. All patients were under the care of a single surgeon using identical implant systems and identical surgical techniques. This study was approved by the institutional review board at VANTHCS. Patients were divided into 2 distinct and consecutive cohorts. The standard of care (SOC) group included all patients from 2013 to 2016. The ERAS group included all patients after the institution of the standardized protocol until the end of the study period.
Data on patient demographics, the American Society of Anesthesiologists risk classification, and preoperative functional status were extracted. Anesthesia techniques included either general endotracheal anesthesia or subarachnoid block with monitored anesthesia care. The quantity of the opioids given during surgery, in the PACU, during the inpatient stay, as discharge prescriptions, and as refills of the narcotic prescriptions up to 3 months postsurgery were recorded. All opioids were converted into morphine equivalent dosages (MED) in order to be properly analyzed using the statistical methodologies described in the statistical section.15 The VHA is a closed health care delivery system; therefore, all of the prescriptions ordered by surgery providers were recorded in the electronic health record.
ERAS Protocol
The SOC cohort was predominantly managed with general endotracheal anesthesia. The ERAS group was predominantly managed with subarachnoid blocks (Table 1). For the ERAS protocol preoperatively, the patients were administered oral gabapentin 300 mg, acetaminophen 650 mg, and oxycodone 20 mg, and IV ondansetron 4 mg. Intraoperatively, minimal opioids were used. In the PACU, the patients received dilaudid 0.25 mg IV as needed every 15 minutes for up to 1 mg/h. The nursing staff was trained to use the visual analog pain scale scores to titrate the medication. During the inpatient stay, patients received 1 g IV acetaminophen every 6 hours for 3 doses. The patients thereafter received oral acetaminophen as needed. Other medications in the multimodal pain-management protocol included gabapentin 300 mg twice daily, meloxicam 15 mg daily, and oxycodone 10 mg every 4 hours as needed. Rescue medication for insufficient pain relief was dilaudid 0.25 mg IV every 15 minutes for visual analog pain scale > 8. On discharge, the patients received a prescription of 30 tablets of hydrocodone 10 mg.
Periarticular Injections
Intraoperatively, all patients in the SOC and ERAS groups received periarticular injections. The liposomal bupivacaine injection was added to the standard injection mixture for the ERAS group. For the SOC group, the total volume of 100 ml was divided into 10 separate 10 cc syringes, and for the ERAS group, the total volume of 140 ml was divided into 14 separate 10 cc syringes. The SOC group injections were performed with an 18-gauge needle and the periarticular soft tissues grossly infiltrated. The ERAS group injections were done with more attention to anatomical detail. Injection sites for the ERAS group included the posterior joint capsule, the medial compartment, the lateral compartment, the tibial fat pad, the quadriceps and the patellar tendon, the femoral and tibial periosteum circumferentially, and the anterior joint capsule. Each needle-stick in the ERAS group delivered 1 to 1.5 ml through a 22-gauge needle to each compartment of the knee.
Outcome Variable
The primary outcome measure was total oral MED intraoperatively, in the PACU, during the hospital inpatient stay, in the hospital discharge prescription, and during the 3-month period after hospital discharge. Incidence of nausea and vomiting during the inpatient stay and any narcotic use at 6 months postsurgery were secondary binary outcomes.
Statistical Analysis
Demographic data and the clinical characteristics for the entire group were described using the sample mean and SD for continuous variables and the frequency and percentage for categorical variables. Differences between the 2 cohorts were analyzed using a 2-independent-sample t test and Fisher exact test.
The estimation of the total oral MED throughout all phases of care was done using a separate Poisson model due to the data being not normally distributed. A log-linear regression model was used to evaluate the main effect of ERAS vs the SOC cohort on the total oral MED used. Finally, a separate multiple logistic regression model was used to estimate the odds of postoperative nausea and vomiting and narcotic use at 6 months postsurgery between the cohorts. The adjusted odds ratio (OR) was estimated from the logistic model. Age, sex, body mass index, preoperative functional independence score, narcotic use within 3 months prior to surgery, anesthesia type used (subarachnoid block with monitored anesthesia care vs general endotracheal anesthesia), and postoperative complications (yes/no) were included as covariates in each model. The length of hospital stay and the above-mentioned factors were also included as covariates in the model estimating the total oral MED during the hospital stay, on hospital discharge, during the 3-month period after hospital discharge, and at 6 months following hospital discharge.
Statistical analysis was done using SAS version 9.4. The level of significance was set at α = 0.05 (2 tailed), and we implemented the false discovery rate (FDR) procedure to control false positives over multiple tests.16
Results
Two hundred forty-nine patients had 296 elective unilateral TKAs in this study from 2013 through 2018. Thirty-one patients had both unilateral TKAs under the SOC protocol; 5 patients had both unilateral TKAs under the ERAS protocol. Eleven of the patients who eventually had both knees replaced had 1 operation under each protocol The SOC group included 196 TKAs and the ERAS group included 100 TKAs. Of the 196 SOC patients, 94% were male. The mean age was 68.2 years (range, 48-86). The length of hospital stay ranged from 36.6 to 664.3 hours. Of the 100 ERAS patients, 96% were male (Table 2). The mean age was 66.7 years (range, 48-85). The length of hospital stay ranged from 12.5 to 45 hours.
Perioperative Opioid Use
Of the SOC patients, 99.0% received narcotics intraoperatively (range, 0-198 mg MED), and 74.5% received narcotics during PACU recovery (range, 0-141 mg MED). The total oral MED during the hospital stay for the SOC patients ranged from 10 to 2,946 mg. Of the ERAS patients, 86% received no narcotics during surgery (range, 0-110 mg MED), and 98% received no narcotics during PACU recovery (range, 0-65 mg MED). The total oral MED during the hospital stay for the ERAS patients ranged from 10 to 240 mg.
The MED used was significantly lower for the ERAS patients than it was for the SOC patients during surgery (10.5 mg vs 57.4 mg, P = .0001, FDR = .0002) and in the PACU (1.3 mg vs 13.6 mg, P = .0002, FDR = .0004), during the inpatient stay (66.7 mg vs 169.5 mg, P = .0001, FDR = .0002), and on hospital discharge (419.3 mg vs 776.7 mg, P = .0001, FDR = .0002). However, there was no significant difference in the total MED prescriptions filled between patients on the ERAS protocol vs those who received SOC during the 3-month period after hospital discharge (858.3 mg vs 1126.1 mg, P = .29, FDR = .29)(Table 3).
Finally, the logistic regression analysis, adjusting for the covariates demonstrated that the ERAS patients were less likely to take narcotics at 6 months following hospital discharge (OR, 0.23; P = .013; FDR = .018) and less likely to have postoperative nausea and vomiting (OR, 0.18; P = .019; FDR = .02) than SOC patients. There was no statistically significant difference between complication rates for the SOC and ERAS groups, which were 11.2% and 5.0%, respectively, with an overall complication rate of 9.1% (P = .09)(Table 4).
Discussion
Orthopedic surgery has been associated with long-term opioid use and misuse. Orthopedic surgeons are frequently among the highest prescribers of narcotics. According to Volkow and colleagues, orthopedic surgeons were the fourth largest prescribers of opioids in 2009, behind primary care physicians, internists, and dentists.17 The opioid crisis in the United States is well recognized. In 2017, > 70,000 deaths occurred due to drug overdoses, with 68% involving a prescription or illicit opioid. The Centers for Disease Control and Prevention has estimated a total economic burden of $78.5 billion per year as a direct result of misused prescribed opioids.18 This includes the cost of health care, lost productivity, addiction treatment, and the impact on the criminal justice system.
The current opioid crisis places further emphasis on opioid-reducing or sparing techniques in patients undergoing TKA. The use of liposomal bupivacaine for intraoperative periarticular injection is debated in the literature regarding its efficacy and whether it should be included in multimodal protocols. Researchers have argued that liposomal bupivacaine is not superior to regular bupivacaine and because of its increased cost is not justified.19,20 A meta-analysis from Zhao and colleagues showed no difference in pain control and functional recovery when comparing liposomal bupivacaine and control.21 In a randomized clinical trial, Schroer and colleagues matched liposomal bupivacaine against regular bupivacaine and found no difference in pain scores and similar narcotic use during hospitalization.22
Studies evaluating liposomal bupivacaine have demonstrated postoperative benefits in pain relief and potential opioid consumption.23 In a multicenter randomized controlled trial, Barrington and colleagues noted improved pain control at 6 and 12 hours after surgery with liposomal bupivacaine as a periarticular injection vs ropivacaine, though results were similar when compared with intrathecal morphine.24 Snyder and colleagues reported higher patient satisfaction in pain control and overall experience as well as decreased MED consumption in the PACU and on postoperative days 0 to 2 when using liposomal bupivacaine vs a multidrug cocktail for periarticular injection.25
The PILLAR trial, an industry-sponsored study, was designed to compare the effects of local infiltration anesthesia with and without liposomal bupivacaine with emphasis on a meticulous standardized infiltration technique. In our study, we used a similar technique with an expanded volume of injection solution to 140 ml that was delivered throughout the knee in a series of 14 syringes. Each needle-stick delivered 1 to 1.5 ml through a 22-gauge needle to each compartment of the knee. Infiltration technique has varied among the literature focused on periarticular injections.
In our experience, a standard infiltration technique is critical to the effective delivery of liposomal bupivacaine throughout all compartments of the knee and to obtaining reproducible pain control. The importance of injection technique cannot be overemphasized, and variations can be seen in studies published to date.26 Well-designed trials are needed to address this key component.
There have been limited data focused on the veteran population regarding postoperative pain-management strategies and recovery pathways either with or without liposomal bupivacaine. In a retrospective review, Sakamoto and colleagues found VA patients undergoing TKA had reduced opioid use in the first 24 hours after primary TKA with the use of intraoperative liposomal bupivacaine.27 The VA population has been shown to be at high risk for opioid misuse. The prevalence of comorbidities such as traumatic brain injury, posttraumatic stress disorder, and depression in the VA population also places them at risk for polypharmacy of central nervous system–acting medications.28 This emphasizes the importance of multimodal strategies, which can limit or eliminate narcotics in the perioperative period. The implementation of our ERAS protocol reduced opioid use during intraoperative, PACU, and inpatient hospital stay.
While the financial implications of our recovery protocol were not a primary focus of this study, there are many notable benefits on the overall inpatient cost to the VHA. According to the Health Economics Resource Center, the average daily cost of stay while under VA care for an inpatient surgical bed increased from $4,831 in 2013 to $6,220 in 2018.29 Our reduction in length of stay between our cohorts is 44.5 hours, which translates to a substantial financial savings per patient after protocol implementation. A more detailed look at the financial aspect of our protocol would need to be performed to evaluate the financial impact of other aspects of our protocol, such as the elimination of patient-controlled anesthesia and the reduction in total narcotics prescribed in the postoperative global period.
Limitations
The limitations of this study include its retrospective study design. With the VHA patient population, it may be subject to selection bias, as the population is mostly older and predominantly male compared with that of the general population. This could potentially influence the efficacy of our protocol on a population of patients with more women. In a recent study by Perruccio and colleagues, sex was found to moderate the effects of comorbidities, low back pain, and depressive symptoms on postoperative pain in patients undergoing TKA.30
With regard to outpatient narcotic prescriptions, although we cannot fully know whether these filled prescriptions were used for pain control, it is a reasonable assumption that patients who are dealing with continued postoperative or chronic pain issues will fill these prescriptions or seek refills. It is important to note that the data on prescriptions and refills in the 3-month postoperative period include all narcotic prescriptions filled by any VHA prescriber and are not specifically limited to our orthopedic team. For outpatient narcotic use, we were not able to access accurate pill counts for any discharge prescriptions or subsequent refills that were given throughout the VA system. We were able to report on total prescriptions filled in the first 3 months following TKA.
We calculated total oral MEDs to better understand the amount of narcotics being distributed throughout our population of patients. We believe this provides important information about the overall narcotic burden in the veteran population. There was no significant difference between the SOC and ERAS groups regarding oral MED prescribed in the 3-month postoperative period; however, at the 6-month follow-up visit, only 16% of patients in the ERAS group were taking any type of narcotic vs 37.2% in the SOC group (P = .0002).
Conclusions
A multidisciplinary ERAS protocol implemented at VANTHCS was effective in reducing length of stay and opioid burden throughout all phases of surgical care in our patients undergoing primary TKA. Patient and nursing education seem to be critical components to the implementation of a successful multimodal pain protocol. Reducing the narcotic burden has valuable financial and medical benefits in this at-risk population.
1. Inacio MCS, Paxton EW, Graves SE, Namba RS, Nemes S. Projected increase in total knee arthroplasty in the United States - an alternative projection model. Osteoarthritis Cartilage. 2017;25(11):1797-1803. doi:10.1016/j.joca.2017.07.022
2. Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of Postoperative pain: a clinical practice guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council [published correction appears in J Pain. 2016 Apr;17(4):508-10. Dosage error in article text]. J Pain. 2016;17(2):131-157. doi:10.1016/j.jpain.2015.12.008
3. Moucha CS, Weiser MC, Levin EJ. Current Strategies in anesthesia and analgesia for total knee arthroplasty. J Am Acad Orthop Surg. 2016;24(2):60-73. doi:10.5435/JAAOS-D-14-00259
4. Parvizi J, Miller AG, Gandhi K. Multimodal pain management after total joint arthroplasty. J Bone Joint Surg Am. 2011;93(11):1075-1084. doi:10.2106/JBJS.J.01095
5. Jenstrup MT, Jæger P, Lund J, et al. Effects of adductor-canal-blockade on pain and ambulation after total knee arthroplasty: a randomized study. Acta Anaesthesiol Scand. 2012;56(3):357-364. doi:10.1111/j.1399-6576.2011.02621.x
6. Macfarlane AJ, Prasad GA, Chan VW, Brull R. Does regional anesthesia improve outcome after total knee arthroplasty?. Clin Orthop Relat Res. 2009;467(9):2379-2402. doi:10.1007/s11999-008-0666-9
7. Parvataneni HK, Shah VP, Howard H, Cole N, Ranawat AS, Ranawat CS. Controlling pain after total hip and knee arthroplasty using a multimodal protocol with local periarticular injections: a prospective randomized study. J Arthroplasty. 2007;22(6)(suppl 2):33-38. doi:10.1016/j.arth.2007.03.034
8. Busch CA, Shore BJ, Bhandari R, et al. Efficacy of periarticular multimodal drug injection in total knee arthroplasty. A randomized trial. J Bone Joint Surg Am. 2006;88(5):959-963. doi:10.2106/JBJS.E.00344
9. Lamplot JD, Wagner ER, Manning DW. Multimodal pain management in total knee arthroplasty: a prospective randomized controlled trial. J Arthroplasty. 2014;29(2):329-334. doi:10.1016/j.arth.2013.06.005
10. Hyland SJ, Deliberato DG, Fada RA, Romanelli MJ, Collins CL, Wasielewski RC. Liposomal bupivacaine versus standard periarticular injection in total knee arthroplasty with regional anesthesia: a prospective randomized controlled trial. J Arthroplasty. 2019;34(3):488-494. doi:10.1016/j.arth.2018.11.026
11. Barrington JW, Lovald ST, Ong KL, Watson HN, Emerson RH Jr. Postoperative pain after primary total knee arthroplasty: comparison of local injection analgesic cocktails and the role of demographic and surgical factors. J Arthroplasty. 2016;31(9) (suppl):288-292. doi:10.1016/j.arth.2016.05.002
12. Bramlett K, Onel E, Viscusi ER, Jones K. A randomized, double-blind, dose-ranging study comparing wound infiltration of DepoFoam bupivacaine, an extended-release liposomal bupivacaine, to bupivacaine HCl for postsurgical analgesia in total knee arthroplasty. Knee. 2012;19(5):530-536. doi:10.1016/j.knee.2011.12.004
13. Mont MA, Beaver WB, Dysart SH, Barrington JW, Del Gaizo D. Local infiltration analgesia with liposomal bupivacaine improves pain scores and reduces opioid use after total knee arthroplasty: results of a randomized controlled trial. J Arthroplasty. 2018;33(1):90-96. doi:10.1016/j.arth.2017.07.024
14. Hadlandsmyth K, Vander Weg MW, McCoy KD, Mosher HJ, Vaughan-Sarrazin MS, Lund BC. Risk for prolonged opioid use following total knee arthroplasty in veterans. J Arthroplasty. 2018;33(1):119-123. doi:10.1016/j.arth.2017.08.022
15. Nielsen S, Degenhardt L, Hoban B, Gisev N. A synthesis of oral morphine equivalents (OME) for opioid utilisation studies. Pharmacoepidemiol Drug Saf. 2016;25(6):733-737. doi:10.1002/pds.3945
16. Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Statist Soc B. 1995;57(1):289-300. doi:10.1111/j.2517-6161.1995.tb02031.x
17. Volkow ND, McLellan TA, Cotto JH, Karithanom M, Weiss SRB. Characteristics of opioid prescriptions in 2009. JAMA. 2011;305(13):1299-1301. doi:10.1001/jama.2011.401
18. Scholl L, Seth P, Kariisa M, Wilson N, Baldwin G. Drug and opioid-involved overdose deaths - United States, 2013-2017. MMWR Morb Mortal Wkly Rep. 2018;67(5152):1419-1427. doi:10.15585/mmwr.mm675152e1
19. Pichler L, Poeran J, Zubizarreta N, et al. Liposomal bupivacaine does not reduce inpatient opioid prescription or related complications after knee arthroplasty: a database analysis. Anesthesiology. 2018;129(4):689-699. doi:10.1097/ALN.0000000000002267
20. Jain RK, Porat MD, Klingenstein GG, Reid JJ, Post RE, Schoifet SD. The AAHKS Clinical Research Award: liposomal bupivacaine and periarticular injection are not superior to single-shot intra-articular injection for pain control in total knee arthroplasty. J Arthroplasty. 2016;31(9)(suppl):22-25. doi:10.1016/j.arth.2016.03.036
21. Zhao B, Ma X, Zhang J, Ma J, Cao Q. The efficacy of local liposomal bupivacaine infiltration on pain and recovery after total joint arthroplasty: a systematic review and meta-analysis of randomized controlled trials. Medicine (Baltimore). 2019;98(3):e14092. doi:10.1097/MD.0000000000014092
22. Schroer WC, Diesfeld PG, LeMarr AR, Morton DJ, Reedy ME. Does extended-release liposomal bupivacaine better control pain than bupivacaine after total knee arthroplasty (TKA)? A prospective, randomized clinical trial. J Arthroplasty. 2015;30(9)(suppl):64-67. doi:10.1016/j.arth.2015.01.059
23. Ma J, Zhang W, Yao S. Liposomal bupivacaine infiltration versus femoral nerve block for pain control in total knee arthroplasty: a systematic review and meta-analysis. Int J Surg. 2016;36(Pt A): 44-55. doi:10.1016/j.ijsu.2016.10.007
24. Barrington JW, Emerson RH, Lovald ST, Lombardi AV, Berend KR. No difference in early analgesia between liposomal bupivacaine injection and intrathecal morphine after TKA. Clin Orthop Relat Res. 2017;475(1):94-105. doi:10.1007/s11999-016-4931-z
25. Snyder MA, Scheuerman CM, Gregg JL, Ruhnke CJ, Eten K. Improving total knee arthroplasty perioperative pain management using a periarticular injection with bupivacaine liposomal suspension. Arthroplast Today. 2016;2(1):37-42. doi:10.1016/j.artd.2015.05.005
26. Kuang MJ,Du Y, Ma JX, He W, Fu L, Ma XL. The efficacy of liposomal bupivacaine using periarticular injection in total knee arthroplasty: a systematic review and meta-analysis. J Arthroplasty. 2017;32(4):1395-1402. doi:10.1016/j.arth.2016.12.025
27. Sakamoto B, Keiser S, Meldrum R, Harker G, Freese A. Efficacy of liposomal bupivacaine infiltration on the management of total knee arthroplasty. JAMA Surg. 2017;152(1):90-95. doi:10.1001/jamasurg.2016.3474
28. Collett GA, Song K, Jaramillo CA, Potter JS, Finley EP, Pugh MJ. Prevalence of central nervous system polypharmacy and associations with overdose and suicide-related behaviors in Iraq and Afghanistan war veterans in VA care 2010-2011. Drugs Real World Outcomes. 2016;3(1):45-52. doi:10.1007/s40801-015-0055-0
29. US Department of Veterans Affairs. HERC inpatient average cost data. Updated April 2, 2021. Accessed April 16, 2021. https://www.herc.research.va.gov/include/page.asp?id=inpatient#herc-inpat-avg-cost
30. Perruccio AV, Fitzpatrick J, Power JD, et al. Sex-modified effects of depression, low back pain, and comorbidities on pain after total knee arthroplasty for osteoarthritis. Arthritis Care Res (Hoboken). 2020;72(8):1074-1080. doi:10.1002/acr.24002
Total knee arthroplasty (TKA) is one of the most common surgical procedures in the United States. The volume of TKAs is projected to substantially increase over the next 30 years.1 Adequate pain control after TKA is critically important to achieve early mobilization, shorten the length of hospital stay, and reduce postoperative complications. The evolution and inclusion of multimodal pain-management protocols have had a major impact on the clinical outcomes for TKA patients.2,3
Pain-management protocols typically use several modalities to control pain throughout the perioperative period. Multimodal opioid and nonopioid oral medications are administered during the pre- and postoperative periods and often involve a combination of acetaminophen, gabapentinoids, and cyclooxygenase-2 inhibitors.4 Peripheral nerve blocks and central neuraxial blockades are widely used and have been shown to be effective in reducing postoperative pain as well as overall opioid consumption.5,6 Finally, intraoperative periarticular injections have been shown to reduce postoperative pain and opioid consumption as well as improve patient satisfaction scores.7-9 These strategies are routinely used in TKA with the goal of minimizing overall opioid consumption and adverse events, reducing perioperative complications, and improving patient satisfaction.
Periarticular injections during surgery are an integral part of the multimodal pain-management protocols, though no consensus has been reached on proper injection formulation or technique. Liposomal bupivacaine is a local anesthetic depot formulation approved by the US Food and Drug Administration for surgical patients. The reported results have been discrepant regarding the efficacy of using liposomal bupivacaine injection in patients with TKA. Several studies have reported no added benefit of liposomal bupivacaine in contrast to a mixture of local anesthetics.10,11 Other studies have demonstrated superior pain relief.12 Many factors may contribute to the discrepant data, such as injection techniques, infiltration volume, and the assessment tools used to measure efficacy and safety.13
The US Department of Veterans Affairs (VA) Veterans Health Administration (VHA) provides care to a large patient population. Many of the patients in that system have high-risk profiles, including medical comorbidities; exposure to chronic pain and opioid use; and psychological and central nervous system injuries, including posttraumatic stress disorder and traumatic brain injury. Hadlandsmyth and colleagues reported increased risk of prolonged opioid use in VA patients after TKA surgery.14 They found that 20% of the patients were still on long-term opioids more than 90 days after TKA.
The purpose of this study was to evaluate the efficacy of the implementation of a comprehensive enhanced recovery after surgery (ERAS) protocol at a regional VA medical center. We hypothesize that the addition of liposomal bupivacaine in a multidisciplinary ERAS protocol would reduce the length of hospital stay and opioid consumption without any deleterious effects on postoperative outcomes.
Methods
A postoperative recovery protocol was implemented in 2013 at VA North Texas Health Care System (VANTHCS) in Dallas, and many of the patients continued to have issues with satisfactory pain control, prolonged length of stay, and extended opioid consumption postoperatively. A multimodal pain-management protocol and multidisciplinary perioperative case-management protocol were implemented in 2016 to further improve the clinical outcomes of patients undergoing TKA surgery. The senior surgeon (JM) organized a multidisciplinary team of health care providers to identify and implement potential solutions. This task force met weekly and consisted of surgeons, anesthesiologists, certified registered nurse anesthetists, orthopedic physician assistants, a nurse coordinator, a physical therapist, and an occupational therapist, as well as operating room, postanesthesia care unit (PACU), and surgical ward nurses. In addition, the staff from the home health agencies and social services attended the weekly meetings.
We conducted a retrospective review of all patients who had undergone unilateral TKA from 2013 to 2018 at VANTHCS. This was a consecutive, unselected cohort. All patients were under the care of a single surgeon using identical implant systems and identical surgical techniques. This study was approved by the institutional review board at VANTHCS. Patients were divided into 2 distinct and consecutive cohorts. The standard of care (SOC) group included all patients from 2013 to 2016. The ERAS group included all patients after the institution of the standardized protocol until the end of the study period.
Data on patient demographics, the American Society of Anesthesiologists risk classification, and preoperative functional status were extracted. Anesthesia techniques included either general endotracheal anesthesia or subarachnoid block with monitored anesthesia care. The quantity of the opioids given during surgery, in the PACU, during the inpatient stay, as discharge prescriptions, and as refills of the narcotic prescriptions up to 3 months postsurgery were recorded. All opioids were converted into morphine equivalent dosages (MED) in order to be properly analyzed using the statistical methodologies described in the statistical section.15 The VHA is a closed health care delivery system; therefore, all of the prescriptions ordered by surgery providers were recorded in the electronic health record.
ERAS Protocol
The SOC cohort was predominantly managed with general endotracheal anesthesia. The ERAS group was predominantly managed with subarachnoid blocks (Table 1). For the ERAS protocol preoperatively, the patients were administered oral gabapentin 300 mg, acetaminophen 650 mg, and oxycodone 20 mg, and IV ondansetron 4 mg. Intraoperatively, minimal opioids were used. In the PACU, the patients received dilaudid 0.25 mg IV as needed every 15 minutes for up to 1 mg/h. The nursing staff was trained to use the visual analog pain scale scores to titrate the medication. During the inpatient stay, patients received 1 g IV acetaminophen every 6 hours for 3 doses. The patients thereafter received oral acetaminophen as needed. Other medications in the multimodal pain-management protocol included gabapentin 300 mg twice daily, meloxicam 15 mg daily, and oxycodone 10 mg every 4 hours as needed. Rescue medication for insufficient pain relief was dilaudid 0.25 mg IV every 15 minutes for visual analog pain scale > 8. On discharge, the patients received a prescription of 30 tablets of hydrocodone 10 mg.
Periarticular Injections
Intraoperatively, all patients in the SOC and ERAS groups received periarticular injections. The liposomal bupivacaine injection was added to the standard injection mixture for the ERAS group. For the SOC group, the total volume of 100 ml was divided into 10 separate 10 cc syringes, and for the ERAS group, the total volume of 140 ml was divided into 14 separate 10 cc syringes. The SOC group injections were performed with an 18-gauge needle and the periarticular soft tissues grossly infiltrated. The ERAS group injections were done with more attention to anatomical detail. Injection sites for the ERAS group included the posterior joint capsule, the medial compartment, the lateral compartment, the tibial fat pad, the quadriceps and the patellar tendon, the femoral and tibial periosteum circumferentially, and the anterior joint capsule. Each needle-stick in the ERAS group delivered 1 to 1.5 ml through a 22-gauge needle to each compartment of the knee.
Outcome Variable
The primary outcome measure was total oral MED intraoperatively, in the PACU, during the hospital inpatient stay, in the hospital discharge prescription, and during the 3-month period after hospital discharge. Incidence of nausea and vomiting during the inpatient stay and any narcotic use at 6 months postsurgery were secondary binary outcomes.
Statistical Analysis
Demographic data and the clinical characteristics for the entire group were described using the sample mean and SD for continuous variables and the frequency and percentage for categorical variables. Differences between the 2 cohorts were analyzed using a 2-independent-sample t test and Fisher exact test.
The estimation of the total oral MED throughout all phases of care was done using a separate Poisson model due to the data being not normally distributed. A log-linear regression model was used to evaluate the main effect of ERAS vs the SOC cohort on the total oral MED used. Finally, a separate multiple logistic regression model was used to estimate the odds of postoperative nausea and vomiting and narcotic use at 6 months postsurgery between the cohorts. The adjusted odds ratio (OR) was estimated from the logistic model. Age, sex, body mass index, preoperative functional independence score, narcotic use within 3 months prior to surgery, anesthesia type used (subarachnoid block with monitored anesthesia care vs general endotracheal anesthesia), and postoperative complications (yes/no) were included as covariates in each model. The length of hospital stay and the above-mentioned factors were also included as covariates in the model estimating the total oral MED during the hospital stay, on hospital discharge, during the 3-month period after hospital discharge, and at 6 months following hospital discharge.
Statistical analysis was done using SAS version 9.4. The level of significance was set at α = 0.05 (2 tailed), and we implemented the false discovery rate (FDR) procedure to control false positives over multiple tests.16
Results
Two hundred forty-nine patients had 296 elective unilateral TKAs in this study from 2013 through 2018. Thirty-one patients had both unilateral TKAs under the SOC protocol; 5 patients had both unilateral TKAs under the ERAS protocol. Eleven of the patients who eventually had both knees replaced had 1 operation under each protocol The SOC group included 196 TKAs and the ERAS group included 100 TKAs. Of the 196 SOC patients, 94% were male. The mean age was 68.2 years (range, 48-86). The length of hospital stay ranged from 36.6 to 664.3 hours. Of the 100 ERAS patients, 96% were male (Table 2). The mean age was 66.7 years (range, 48-85). The length of hospital stay ranged from 12.5 to 45 hours.
Perioperative Opioid Use
Of the SOC patients, 99.0% received narcotics intraoperatively (range, 0-198 mg MED), and 74.5% received narcotics during PACU recovery (range, 0-141 mg MED). The total oral MED during the hospital stay for the SOC patients ranged from 10 to 2,946 mg. Of the ERAS patients, 86% received no narcotics during surgery (range, 0-110 mg MED), and 98% received no narcotics during PACU recovery (range, 0-65 mg MED). The total oral MED during the hospital stay for the ERAS patients ranged from 10 to 240 mg.
The MED used was significantly lower for the ERAS patients than it was for the SOC patients during surgery (10.5 mg vs 57.4 mg, P = .0001, FDR = .0002) and in the PACU (1.3 mg vs 13.6 mg, P = .0002, FDR = .0004), during the inpatient stay (66.7 mg vs 169.5 mg, P = .0001, FDR = .0002), and on hospital discharge (419.3 mg vs 776.7 mg, P = .0001, FDR = .0002). However, there was no significant difference in the total MED prescriptions filled between patients on the ERAS protocol vs those who received SOC during the 3-month period after hospital discharge (858.3 mg vs 1126.1 mg, P = .29, FDR = .29)(Table 3).
Finally, the logistic regression analysis, adjusting for the covariates demonstrated that the ERAS patients were less likely to take narcotics at 6 months following hospital discharge (OR, 0.23; P = .013; FDR = .018) and less likely to have postoperative nausea and vomiting (OR, 0.18; P = .019; FDR = .02) than SOC patients. There was no statistically significant difference between complication rates for the SOC and ERAS groups, which were 11.2% and 5.0%, respectively, with an overall complication rate of 9.1% (P = .09)(Table 4).
Discussion
Orthopedic surgery has been associated with long-term opioid use and misuse. Orthopedic surgeons are frequently among the highest prescribers of narcotics. According to Volkow and colleagues, orthopedic surgeons were the fourth largest prescribers of opioids in 2009, behind primary care physicians, internists, and dentists.17 The opioid crisis in the United States is well recognized. In 2017, > 70,000 deaths occurred due to drug overdoses, with 68% involving a prescription or illicit opioid. The Centers for Disease Control and Prevention has estimated a total economic burden of $78.5 billion per year as a direct result of misused prescribed opioids.18 This includes the cost of health care, lost productivity, addiction treatment, and the impact on the criminal justice system.
The current opioid crisis places further emphasis on opioid-reducing or sparing techniques in patients undergoing TKA. The use of liposomal bupivacaine for intraoperative periarticular injection is debated in the literature regarding its efficacy and whether it should be included in multimodal protocols. Researchers have argued that liposomal bupivacaine is not superior to regular bupivacaine and because of its increased cost is not justified.19,20 A meta-analysis from Zhao and colleagues showed no difference in pain control and functional recovery when comparing liposomal bupivacaine and control.21 In a randomized clinical trial, Schroer and colleagues matched liposomal bupivacaine against regular bupivacaine and found no difference in pain scores and similar narcotic use during hospitalization.22
Studies evaluating liposomal bupivacaine have demonstrated postoperative benefits in pain relief and potential opioid consumption.23 In a multicenter randomized controlled trial, Barrington and colleagues noted improved pain control at 6 and 12 hours after surgery with liposomal bupivacaine as a periarticular injection vs ropivacaine, though results were similar when compared with intrathecal morphine.24 Snyder and colleagues reported higher patient satisfaction in pain control and overall experience as well as decreased MED consumption in the PACU and on postoperative days 0 to 2 when using liposomal bupivacaine vs a multidrug cocktail for periarticular injection.25
The PILLAR trial, an industry-sponsored study, was designed to compare the effects of local infiltration anesthesia with and without liposomal bupivacaine with emphasis on a meticulous standardized infiltration technique. In our study, we used a similar technique with an expanded volume of injection solution to 140 ml that was delivered throughout the knee in a series of 14 syringes. Each needle-stick delivered 1 to 1.5 ml through a 22-gauge needle to each compartment of the knee. Infiltration technique has varied among the literature focused on periarticular injections.
In our experience, a standard infiltration technique is critical to the effective delivery of liposomal bupivacaine throughout all compartments of the knee and to obtaining reproducible pain control. The importance of injection technique cannot be overemphasized, and variations can be seen in studies published to date.26 Well-designed trials are needed to address this key component.
There have been limited data focused on the veteran population regarding postoperative pain-management strategies and recovery pathways either with or without liposomal bupivacaine. In a retrospective review, Sakamoto and colleagues found VA patients undergoing TKA had reduced opioid use in the first 24 hours after primary TKA with the use of intraoperative liposomal bupivacaine.27 The VA population has been shown to be at high risk for opioid misuse. The prevalence of comorbidities such as traumatic brain injury, posttraumatic stress disorder, and depression in the VA population also places them at risk for polypharmacy of central nervous system–acting medications.28 This emphasizes the importance of multimodal strategies, which can limit or eliminate narcotics in the perioperative period. The implementation of our ERAS protocol reduced opioid use during intraoperative, PACU, and inpatient hospital stay.
While the financial implications of our recovery protocol were not a primary focus of this study, there are many notable benefits on the overall inpatient cost to the VHA. According to the Health Economics Resource Center, the average daily cost of stay while under VA care for an inpatient surgical bed increased from $4,831 in 2013 to $6,220 in 2018.29 Our reduction in length of stay between our cohorts is 44.5 hours, which translates to a substantial financial savings per patient after protocol implementation. A more detailed look at the financial aspect of our protocol would need to be performed to evaluate the financial impact of other aspects of our protocol, such as the elimination of patient-controlled anesthesia and the reduction in total narcotics prescribed in the postoperative global period.
Limitations
The limitations of this study include its retrospective study design. With the VHA patient population, it may be subject to selection bias, as the population is mostly older and predominantly male compared with that of the general population. This could potentially influence the efficacy of our protocol on a population of patients with more women. In a recent study by Perruccio and colleagues, sex was found to moderate the effects of comorbidities, low back pain, and depressive symptoms on postoperative pain in patients undergoing TKA.30
With regard to outpatient narcotic prescriptions, although we cannot fully know whether these filled prescriptions were used for pain control, it is a reasonable assumption that patients who are dealing with continued postoperative or chronic pain issues will fill these prescriptions or seek refills. It is important to note that the data on prescriptions and refills in the 3-month postoperative period include all narcotic prescriptions filled by any VHA prescriber and are not specifically limited to our orthopedic team. For outpatient narcotic use, we were not able to access accurate pill counts for any discharge prescriptions or subsequent refills that were given throughout the VA system. We were able to report on total prescriptions filled in the first 3 months following TKA.
We calculated total oral MEDs to better understand the amount of narcotics being distributed throughout our population of patients. We believe this provides important information about the overall narcotic burden in the veteran population. There was no significant difference between the SOC and ERAS groups regarding oral MED prescribed in the 3-month postoperative period; however, at the 6-month follow-up visit, only 16% of patients in the ERAS group were taking any type of narcotic vs 37.2% in the SOC group (P = .0002).
Conclusions
A multidisciplinary ERAS protocol implemented at VANTHCS was effective in reducing length of stay and opioid burden throughout all phases of surgical care in our patients undergoing primary TKA. Patient and nursing education seem to be critical components to the implementation of a successful multimodal pain protocol. Reducing the narcotic burden has valuable financial and medical benefits in this at-risk population.
Total knee arthroplasty (TKA) is one of the most common surgical procedures in the United States. The volume of TKAs is projected to substantially increase over the next 30 years.1 Adequate pain control after TKA is critically important to achieve early mobilization, shorten the length of hospital stay, and reduce postoperative complications. The evolution and inclusion of multimodal pain-management protocols have had a major impact on the clinical outcomes for TKA patients.2,3
Pain-management protocols typically use several modalities to control pain throughout the perioperative period. Multimodal opioid and nonopioid oral medications are administered during the pre- and postoperative periods and often involve a combination of acetaminophen, gabapentinoids, and cyclooxygenase-2 inhibitors.4 Peripheral nerve blocks and central neuraxial blockades are widely used and have been shown to be effective in reducing postoperative pain as well as overall opioid consumption.5,6 Finally, intraoperative periarticular injections have been shown to reduce postoperative pain and opioid consumption as well as improve patient satisfaction scores.7-9 These strategies are routinely used in TKA with the goal of minimizing overall opioid consumption and adverse events, reducing perioperative complications, and improving patient satisfaction.
Periarticular injections during surgery are an integral part of the multimodal pain-management protocols, though no consensus has been reached on proper injection formulation or technique. Liposomal bupivacaine is a local anesthetic depot formulation approved by the US Food and Drug Administration for surgical patients. The reported results have been discrepant regarding the efficacy of using liposomal bupivacaine injection in patients with TKA. Several studies have reported no added benefit of liposomal bupivacaine in contrast to a mixture of local anesthetics.10,11 Other studies have demonstrated superior pain relief.12 Many factors may contribute to the discrepant data, such as injection techniques, infiltration volume, and the assessment tools used to measure efficacy and safety.13
The US Department of Veterans Affairs (VA) Veterans Health Administration (VHA) provides care to a large patient population. Many of the patients in that system have high-risk profiles, including medical comorbidities; exposure to chronic pain and opioid use; and psychological and central nervous system injuries, including posttraumatic stress disorder and traumatic brain injury. Hadlandsmyth and colleagues reported increased risk of prolonged opioid use in VA patients after TKA surgery.14 They found that 20% of the patients were still on long-term opioids more than 90 days after TKA.
The purpose of this study was to evaluate the efficacy of the implementation of a comprehensive enhanced recovery after surgery (ERAS) protocol at a regional VA medical center. We hypothesize that the addition of liposomal bupivacaine in a multidisciplinary ERAS protocol would reduce the length of hospital stay and opioid consumption without any deleterious effects on postoperative outcomes.
Methods
A postoperative recovery protocol was implemented in 2013 at VA North Texas Health Care System (VANTHCS) in Dallas, and many of the patients continued to have issues with satisfactory pain control, prolonged length of stay, and extended opioid consumption postoperatively. A multimodal pain-management protocol and multidisciplinary perioperative case-management protocol were implemented in 2016 to further improve the clinical outcomes of patients undergoing TKA surgery. The senior surgeon (JM) organized a multidisciplinary team of health care providers to identify and implement potential solutions. This task force met weekly and consisted of surgeons, anesthesiologists, certified registered nurse anesthetists, orthopedic physician assistants, a nurse coordinator, a physical therapist, and an occupational therapist, as well as operating room, postanesthesia care unit (PACU), and surgical ward nurses. In addition, the staff from the home health agencies and social services attended the weekly meetings.
We conducted a retrospective review of all patients who had undergone unilateral TKA from 2013 to 2018 at VANTHCS. This was a consecutive, unselected cohort. All patients were under the care of a single surgeon using identical implant systems and identical surgical techniques. This study was approved by the institutional review board at VANTHCS. Patients were divided into 2 distinct and consecutive cohorts. The standard of care (SOC) group included all patients from 2013 to 2016. The ERAS group included all patients after the institution of the standardized protocol until the end of the study period.
Data on patient demographics, the American Society of Anesthesiologists risk classification, and preoperative functional status were extracted. Anesthesia techniques included either general endotracheal anesthesia or subarachnoid block with monitored anesthesia care. The quantity of the opioids given during surgery, in the PACU, during the inpatient stay, as discharge prescriptions, and as refills of the narcotic prescriptions up to 3 months postsurgery were recorded. All opioids were converted into morphine equivalent dosages (MED) in order to be properly analyzed using the statistical methodologies described in the statistical section.15 The VHA is a closed health care delivery system; therefore, all of the prescriptions ordered by surgery providers were recorded in the electronic health record.
ERAS Protocol
The SOC cohort was predominantly managed with general endotracheal anesthesia. The ERAS group was predominantly managed with subarachnoid blocks (Table 1). For the ERAS protocol preoperatively, the patients were administered oral gabapentin 300 mg, acetaminophen 650 mg, and oxycodone 20 mg, and IV ondansetron 4 mg. Intraoperatively, minimal opioids were used. In the PACU, the patients received dilaudid 0.25 mg IV as needed every 15 minutes for up to 1 mg/h. The nursing staff was trained to use the visual analog pain scale scores to titrate the medication. During the inpatient stay, patients received 1 g IV acetaminophen every 6 hours for 3 doses. The patients thereafter received oral acetaminophen as needed. Other medications in the multimodal pain-management protocol included gabapentin 300 mg twice daily, meloxicam 15 mg daily, and oxycodone 10 mg every 4 hours as needed. Rescue medication for insufficient pain relief was dilaudid 0.25 mg IV every 15 minutes for visual analog pain scale > 8. On discharge, the patients received a prescription of 30 tablets of hydrocodone 10 mg.
Periarticular Injections
Intraoperatively, all patients in the SOC and ERAS groups received periarticular injections. The liposomal bupivacaine injection was added to the standard injection mixture for the ERAS group. For the SOC group, the total volume of 100 ml was divided into 10 separate 10 cc syringes, and for the ERAS group, the total volume of 140 ml was divided into 14 separate 10 cc syringes. The SOC group injections were performed with an 18-gauge needle and the periarticular soft tissues grossly infiltrated. The ERAS group injections were done with more attention to anatomical detail. Injection sites for the ERAS group included the posterior joint capsule, the medial compartment, the lateral compartment, the tibial fat pad, the quadriceps and the patellar tendon, the femoral and tibial periosteum circumferentially, and the anterior joint capsule. Each needle-stick in the ERAS group delivered 1 to 1.5 ml through a 22-gauge needle to each compartment of the knee.
Outcome Variable
The primary outcome measure was total oral MED intraoperatively, in the PACU, during the hospital inpatient stay, in the hospital discharge prescription, and during the 3-month period after hospital discharge. Incidence of nausea and vomiting during the inpatient stay and any narcotic use at 6 months postsurgery were secondary binary outcomes.
Statistical Analysis
Demographic data and the clinical characteristics for the entire group were described using the sample mean and SD for continuous variables and the frequency and percentage for categorical variables. Differences between the 2 cohorts were analyzed using a 2-independent-sample t test and Fisher exact test.
The estimation of the total oral MED throughout all phases of care was done using a separate Poisson model due to the data being not normally distributed. A log-linear regression model was used to evaluate the main effect of ERAS vs the SOC cohort on the total oral MED used. Finally, a separate multiple logistic regression model was used to estimate the odds of postoperative nausea and vomiting and narcotic use at 6 months postsurgery between the cohorts. The adjusted odds ratio (OR) was estimated from the logistic model. Age, sex, body mass index, preoperative functional independence score, narcotic use within 3 months prior to surgery, anesthesia type used (subarachnoid block with monitored anesthesia care vs general endotracheal anesthesia), and postoperative complications (yes/no) were included as covariates in each model. The length of hospital stay and the above-mentioned factors were also included as covariates in the model estimating the total oral MED during the hospital stay, on hospital discharge, during the 3-month period after hospital discharge, and at 6 months following hospital discharge.
Statistical analysis was done using SAS version 9.4. The level of significance was set at α = 0.05 (2 tailed), and we implemented the false discovery rate (FDR) procedure to control false positives over multiple tests.16
Results
Two hundred forty-nine patients had 296 elective unilateral TKAs in this study from 2013 through 2018. Thirty-one patients had both unilateral TKAs under the SOC protocol; 5 patients had both unilateral TKAs under the ERAS protocol. Eleven of the patients who eventually had both knees replaced had 1 operation under each protocol The SOC group included 196 TKAs and the ERAS group included 100 TKAs. Of the 196 SOC patients, 94% were male. The mean age was 68.2 years (range, 48-86). The length of hospital stay ranged from 36.6 to 664.3 hours. Of the 100 ERAS patients, 96% were male (Table 2). The mean age was 66.7 years (range, 48-85). The length of hospital stay ranged from 12.5 to 45 hours.
Perioperative Opioid Use
Of the SOC patients, 99.0% received narcotics intraoperatively (range, 0-198 mg MED), and 74.5% received narcotics during PACU recovery (range, 0-141 mg MED). The total oral MED during the hospital stay for the SOC patients ranged from 10 to 2,946 mg. Of the ERAS patients, 86% received no narcotics during surgery (range, 0-110 mg MED), and 98% received no narcotics during PACU recovery (range, 0-65 mg MED). The total oral MED during the hospital stay for the ERAS patients ranged from 10 to 240 mg.
The MED used was significantly lower for the ERAS patients than it was for the SOC patients during surgery (10.5 mg vs 57.4 mg, P = .0001, FDR = .0002) and in the PACU (1.3 mg vs 13.6 mg, P = .0002, FDR = .0004), during the inpatient stay (66.7 mg vs 169.5 mg, P = .0001, FDR = .0002), and on hospital discharge (419.3 mg vs 776.7 mg, P = .0001, FDR = .0002). However, there was no significant difference in the total MED prescriptions filled between patients on the ERAS protocol vs those who received SOC during the 3-month period after hospital discharge (858.3 mg vs 1126.1 mg, P = .29, FDR = .29)(Table 3).
Finally, the logistic regression analysis, adjusting for the covariates demonstrated that the ERAS patients were less likely to take narcotics at 6 months following hospital discharge (OR, 0.23; P = .013; FDR = .018) and less likely to have postoperative nausea and vomiting (OR, 0.18; P = .019; FDR = .02) than SOC patients. There was no statistically significant difference between complication rates for the SOC and ERAS groups, which were 11.2% and 5.0%, respectively, with an overall complication rate of 9.1% (P = .09)(Table 4).
Discussion
Orthopedic surgery has been associated with long-term opioid use and misuse. Orthopedic surgeons are frequently among the highest prescribers of narcotics. According to Volkow and colleagues, orthopedic surgeons were the fourth largest prescribers of opioids in 2009, behind primary care physicians, internists, and dentists.17 The opioid crisis in the United States is well recognized. In 2017, > 70,000 deaths occurred due to drug overdoses, with 68% involving a prescription or illicit opioid. The Centers for Disease Control and Prevention has estimated a total economic burden of $78.5 billion per year as a direct result of misused prescribed opioids.18 This includes the cost of health care, lost productivity, addiction treatment, and the impact on the criminal justice system.
The current opioid crisis places further emphasis on opioid-reducing or sparing techniques in patients undergoing TKA. The use of liposomal bupivacaine for intraoperative periarticular injection is debated in the literature regarding its efficacy and whether it should be included in multimodal protocols. Researchers have argued that liposomal bupivacaine is not superior to regular bupivacaine and because of its increased cost is not justified.19,20 A meta-analysis from Zhao and colleagues showed no difference in pain control and functional recovery when comparing liposomal bupivacaine and control.21 In a randomized clinical trial, Schroer and colleagues matched liposomal bupivacaine against regular bupivacaine and found no difference in pain scores and similar narcotic use during hospitalization.22
Studies evaluating liposomal bupivacaine have demonstrated postoperative benefits in pain relief and potential opioid consumption.23 In a multicenter randomized controlled trial, Barrington and colleagues noted improved pain control at 6 and 12 hours after surgery with liposomal bupivacaine as a periarticular injection vs ropivacaine, though results were similar when compared with intrathecal morphine.24 Snyder and colleagues reported higher patient satisfaction in pain control and overall experience as well as decreased MED consumption in the PACU and on postoperative days 0 to 2 when using liposomal bupivacaine vs a multidrug cocktail for periarticular injection.25
The PILLAR trial, an industry-sponsored study, was designed to compare the effects of local infiltration anesthesia with and without liposomal bupivacaine with emphasis on a meticulous standardized infiltration technique. In our study, we used a similar technique with an expanded volume of injection solution to 140 ml that was delivered throughout the knee in a series of 14 syringes. Each needle-stick delivered 1 to 1.5 ml through a 22-gauge needle to each compartment of the knee. Infiltration technique has varied among the literature focused on periarticular injections.
In our experience, a standard infiltration technique is critical to the effective delivery of liposomal bupivacaine throughout all compartments of the knee and to obtaining reproducible pain control. The importance of injection technique cannot be overemphasized, and variations can be seen in studies published to date.26 Well-designed trials are needed to address this key component.
There have been limited data focused on the veteran population regarding postoperative pain-management strategies and recovery pathways either with or without liposomal bupivacaine. In a retrospective review, Sakamoto and colleagues found VA patients undergoing TKA had reduced opioid use in the first 24 hours after primary TKA with the use of intraoperative liposomal bupivacaine.27 The VA population has been shown to be at high risk for opioid misuse. The prevalence of comorbidities such as traumatic brain injury, posttraumatic stress disorder, and depression in the VA population also places them at risk for polypharmacy of central nervous system–acting medications.28 This emphasizes the importance of multimodal strategies, which can limit or eliminate narcotics in the perioperative period. The implementation of our ERAS protocol reduced opioid use during intraoperative, PACU, and inpatient hospital stay.
While the financial implications of our recovery protocol were not a primary focus of this study, there are many notable benefits on the overall inpatient cost to the VHA. According to the Health Economics Resource Center, the average daily cost of stay while under VA care for an inpatient surgical bed increased from $4,831 in 2013 to $6,220 in 2018.29 Our reduction in length of stay between our cohorts is 44.5 hours, which translates to a substantial financial savings per patient after protocol implementation. A more detailed look at the financial aspect of our protocol would need to be performed to evaluate the financial impact of other aspects of our protocol, such as the elimination of patient-controlled anesthesia and the reduction in total narcotics prescribed in the postoperative global period.
Limitations
The limitations of this study include its retrospective study design. With the VHA patient population, it may be subject to selection bias, as the population is mostly older and predominantly male compared with that of the general population. This could potentially influence the efficacy of our protocol on a population of patients with more women. In a recent study by Perruccio and colleagues, sex was found to moderate the effects of comorbidities, low back pain, and depressive symptoms on postoperative pain in patients undergoing TKA.30
With regard to outpatient narcotic prescriptions, although we cannot fully know whether these filled prescriptions were used for pain control, it is a reasonable assumption that patients who are dealing with continued postoperative or chronic pain issues will fill these prescriptions or seek refills. It is important to note that the data on prescriptions and refills in the 3-month postoperative period include all narcotic prescriptions filled by any VHA prescriber and are not specifically limited to our orthopedic team. For outpatient narcotic use, we were not able to access accurate pill counts for any discharge prescriptions or subsequent refills that were given throughout the VA system. We were able to report on total prescriptions filled in the first 3 months following TKA.
We calculated total oral MEDs to better understand the amount of narcotics being distributed throughout our population of patients. We believe this provides important information about the overall narcotic burden in the veteran population. There was no significant difference between the SOC and ERAS groups regarding oral MED prescribed in the 3-month postoperative period; however, at the 6-month follow-up visit, only 16% of patients in the ERAS group were taking any type of narcotic vs 37.2% in the SOC group (P = .0002).
Conclusions
A multidisciplinary ERAS protocol implemented at VANTHCS was effective in reducing length of stay and opioid burden throughout all phases of surgical care in our patients undergoing primary TKA. Patient and nursing education seem to be critical components to the implementation of a successful multimodal pain protocol. Reducing the narcotic burden has valuable financial and medical benefits in this at-risk population.
1. Inacio MCS, Paxton EW, Graves SE, Namba RS, Nemes S. Projected increase in total knee arthroplasty in the United States - an alternative projection model. Osteoarthritis Cartilage. 2017;25(11):1797-1803. doi:10.1016/j.joca.2017.07.022
2. Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of Postoperative pain: a clinical practice guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council [published correction appears in J Pain. 2016 Apr;17(4):508-10. Dosage error in article text]. J Pain. 2016;17(2):131-157. doi:10.1016/j.jpain.2015.12.008
3. Moucha CS, Weiser MC, Levin EJ. Current Strategies in anesthesia and analgesia for total knee arthroplasty. J Am Acad Orthop Surg. 2016;24(2):60-73. doi:10.5435/JAAOS-D-14-00259
4. Parvizi J, Miller AG, Gandhi K. Multimodal pain management after total joint arthroplasty. J Bone Joint Surg Am. 2011;93(11):1075-1084. doi:10.2106/JBJS.J.01095
5. Jenstrup MT, Jæger P, Lund J, et al. Effects of adductor-canal-blockade on pain and ambulation after total knee arthroplasty: a randomized study. Acta Anaesthesiol Scand. 2012;56(3):357-364. doi:10.1111/j.1399-6576.2011.02621.x
6. Macfarlane AJ, Prasad GA, Chan VW, Brull R. Does regional anesthesia improve outcome after total knee arthroplasty?. Clin Orthop Relat Res. 2009;467(9):2379-2402. doi:10.1007/s11999-008-0666-9
7. Parvataneni HK, Shah VP, Howard H, Cole N, Ranawat AS, Ranawat CS. Controlling pain after total hip and knee arthroplasty using a multimodal protocol with local periarticular injections: a prospective randomized study. J Arthroplasty. 2007;22(6)(suppl 2):33-38. doi:10.1016/j.arth.2007.03.034
8. Busch CA, Shore BJ, Bhandari R, et al. Efficacy of periarticular multimodal drug injection in total knee arthroplasty. A randomized trial. J Bone Joint Surg Am. 2006;88(5):959-963. doi:10.2106/JBJS.E.00344
9. Lamplot JD, Wagner ER, Manning DW. Multimodal pain management in total knee arthroplasty: a prospective randomized controlled trial. J Arthroplasty. 2014;29(2):329-334. doi:10.1016/j.arth.2013.06.005
10. Hyland SJ, Deliberato DG, Fada RA, Romanelli MJ, Collins CL, Wasielewski RC. Liposomal bupivacaine versus standard periarticular injection in total knee arthroplasty with regional anesthesia: a prospective randomized controlled trial. J Arthroplasty. 2019;34(3):488-494. doi:10.1016/j.arth.2018.11.026
11. Barrington JW, Lovald ST, Ong KL, Watson HN, Emerson RH Jr. Postoperative pain after primary total knee arthroplasty: comparison of local injection analgesic cocktails and the role of demographic and surgical factors. J Arthroplasty. 2016;31(9) (suppl):288-292. doi:10.1016/j.arth.2016.05.002
12. Bramlett K, Onel E, Viscusi ER, Jones K. A randomized, double-blind, dose-ranging study comparing wound infiltration of DepoFoam bupivacaine, an extended-release liposomal bupivacaine, to bupivacaine HCl for postsurgical analgesia in total knee arthroplasty. Knee. 2012;19(5):530-536. doi:10.1016/j.knee.2011.12.004
13. Mont MA, Beaver WB, Dysart SH, Barrington JW, Del Gaizo D. Local infiltration analgesia with liposomal bupivacaine improves pain scores and reduces opioid use after total knee arthroplasty: results of a randomized controlled trial. J Arthroplasty. 2018;33(1):90-96. doi:10.1016/j.arth.2017.07.024
14. Hadlandsmyth K, Vander Weg MW, McCoy KD, Mosher HJ, Vaughan-Sarrazin MS, Lund BC. Risk for prolonged opioid use following total knee arthroplasty in veterans. J Arthroplasty. 2018;33(1):119-123. doi:10.1016/j.arth.2017.08.022
15. Nielsen S, Degenhardt L, Hoban B, Gisev N. A synthesis of oral morphine equivalents (OME) for opioid utilisation studies. Pharmacoepidemiol Drug Saf. 2016;25(6):733-737. doi:10.1002/pds.3945
16. Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Statist Soc B. 1995;57(1):289-300. doi:10.1111/j.2517-6161.1995.tb02031.x
17. Volkow ND, McLellan TA, Cotto JH, Karithanom M, Weiss SRB. Characteristics of opioid prescriptions in 2009. JAMA. 2011;305(13):1299-1301. doi:10.1001/jama.2011.401
18. Scholl L, Seth P, Kariisa M, Wilson N, Baldwin G. Drug and opioid-involved overdose deaths - United States, 2013-2017. MMWR Morb Mortal Wkly Rep. 2018;67(5152):1419-1427. doi:10.15585/mmwr.mm675152e1
19. Pichler L, Poeran J, Zubizarreta N, et al. Liposomal bupivacaine does not reduce inpatient opioid prescription or related complications after knee arthroplasty: a database analysis. Anesthesiology. 2018;129(4):689-699. doi:10.1097/ALN.0000000000002267
20. Jain RK, Porat MD, Klingenstein GG, Reid JJ, Post RE, Schoifet SD. The AAHKS Clinical Research Award: liposomal bupivacaine and periarticular injection are not superior to single-shot intra-articular injection for pain control in total knee arthroplasty. J Arthroplasty. 2016;31(9)(suppl):22-25. doi:10.1016/j.arth.2016.03.036
21. Zhao B, Ma X, Zhang J, Ma J, Cao Q. The efficacy of local liposomal bupivacaine infiltration on pain and recovery after total joint arthroplasty: a systematic review and meta-analysis of randomized controlled trials. Medicine (Baltimore). 2019;98(3):e14092. doi:10.1097/MD.0000000000014092
22. Schroer WC, Diesfeld PG, LeMarr AR, Morton DJ, Reedy ME. Does extended-release liposomal bupivacaine better control pain than bupivacaine after total knee arthroplasty (TKA)? A prospective, randomized clinical trial. J Arthroplasty. 2015;30(9)(suppl):64-67. doi:10.1016/j.arth.2015.01.059
23. Ma J, Zhang W, Yao S. Liposomal bupivacaine infiltration versus femoral nerve block for pain control in total knee arthroplasty: a systematic review and meta-analysis. Int J Surg. 2016;36(Pt A): 44-55. doi:10.1016/j.ijsu.2016.10.007
24. Barrington JW, Emerson RH, Lovald ST, Lombardi AV, Berend KR. No difference in early analgesia between liposomal bupivacaine injection and intrathecal morphine after TKA. Clin Orthop Relat Res. 2017;475(1):94-105. doi:10.1007/s11999-016-4931-z
25. Snyder MA, Scheuerman CM, Gregg JL, Ruhnke CJ, Eten K. Improving total knee arthroplasty perioperative pain management using a periarticular injection with bupivacaine liposomal suspension. Arthroplast Today. 2016;2(1):37-42. doi:10.1016/j.artd.2015.05.005
26. Kuang MJ,Du Y, Ma JX, He W, Fu L, Ma XL. The efficacy of liposomal bupivacaine using periarticular injection in total knee arthroplasty: a systematic review and meta-analysis. J Arthroplasty. 2017;32(4):1395-1402. doi:10.1016/j.arth.2016.12.025
27. Sakamoto B, Keiser S, Meldrum R, Harker G, Freese A. Efficacy of liposomal bupivacaine infiltration on the management of total knee arthroplasty. JAMA Surg. 2017;152(1):90-95. doi:10.1001/jamasurg.2016.3474
28. Collett GA, Song K, Jaramillo CA, Potter JS, Finley EP, Pugh MJ. Prevalence of central nervous system polypharmacy and associations with overdose and suicide-related behaviors in Iraq and Afghanistan war veterans in VA care 2010-2011. Drugs Real World Outcomes. 2016;3(1):45-52. doi:10.1007/s40801-015-0055-0
29. US Department of Veterans Affairs. HERC inpatient average cost data. Updated April 2, 2021. Accessed April 16, 2021. https://www.herc.research.va.gov/include/page.asp?id=inpatient#herc-inpat-avg-cost
30. Perruccio AV, Fitzpatrick J, Power JD, et al. Sex-modified effects of depression, low back pain, and comorbidities on pain after total knee arthroplasty for osteoarthritis. Arthritis Care Res (Hoboken). 2020;72(8):1074-1080. doi:10.1002/acr.24002
1. Inacio MCS, Paxton EW, Graves SE, Namba RS, Nemes S. Projected increase in total knee arthroplasty in the United States - an alternative projection model. Osteoarthritis Cartilage. 2017;25(11):1797-1803. doi:10.1016/j.joca.2017.07.022
2. Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of Postoperative pain: a clinical practice guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council [published correction appears in J Pain. 2016 Apr;17(4):508-10. Dosage error in article text]. J Pain. 2016;17(2):131-157. doi:10.1016/j.jpain.2015.12.008
3. Moucha CS, Weiser MC, Levin EJ. Current Strategies in anesthesia and analgesia for total knee arthroplasty. J Am Acad Orthop Surg. 2016;24(2):60-73. doi:10.5435/JAAOS-D-14-00259
4. Parvizi J, Miller AG, Gandhi K. Multimodal pain management after total joint arthroplasty. J Bone Joint Surg Am. 2011;93(11):1075-1084. doi:10.2106/JBJS.J.01095
5. Jenstrup MT, Jæger P, Lund J, et al. Effects of adductor-canal-blockade on pain and ambulation after total knee arthroplasty: a randomized study. Acta Anaesthesiol Scand. 2012;56(3):357-364. doi:10.1111/j.1399-6576.2011.02621.x
6. Macfarlane AJ, Prasad GA, Chan VW, Brull R. Does regional anesthesia improve outcome after total knee arthroplasty?. Clin Orthop Relat Res. 2009;467(9):2379-2402. doi:10.1007/s11999-008-0666-9
7. Parvataneni HK, Shah VP, Howard H, Cole N, Ranawat AS, Ranawat CS. Controlling pain after total hip and knee arthroplasty using a multimodal protocol with local periarticular injections: a prospective randomized study. J Arthroplasty. 2007;22(6)(suppl 2):33-38. doi:10.1016/j.arth.2007.03.034
8. Busch CA, Shore BJ, Bhandari R, et al. Efficacy of periarticular multimodal drug injection in total knee arthroplasty. A randomized trial. J Bone Joint Surg Am. 2006;88(5):959-963. doi:10.2106/JBJS.E.00344
9. Lamplot JD, Wagner ER, Manning DW. Multimodal pain management in total knee arthroplasty: a prospective randomized controlled trial. J Arthroplasty. 2014;29(2):329-334. doi:10.1016/j.arth.2013.06.005
10. Hyland SJ, Deliberato DG, Fada RA, Romanelli MJ, Collins CL, Wasielewski RC. Liposomal bupivacaine versus standard periarticular injection in total knee arthroplasty with regional anesthesia: a prospective randomized controlled trial. J Arthroplasty. 2019;34(3):488-494. doi:10.1016/j.arth.2018.11.026
11. Barrington JW, Lovald ST, Ong KL, Watson HN, Emerson RH Jr. Postoperative pain after primary total knee arthroplasty: comparison of local injection analgesic cocktails and the role of demographic and surgical factors. J Arthroplasty. 2016;31(9) (suppl):288-292. doi:10.1016/j.arth.2016.05.002
12. Bramlett K, Onel E, Viscusi ER, Jones K. A randomized, double-blind, dose-ranging study comparing wound infiltration of DepoFoam bupivacaine, an extended-release liposomal bupivacaine, to bupivacaine HCl for postsurgical analgesia in total knee arthroplasty. Knee. 2012;19(5):530-536. doi:10.1016/j.knee.2011.12.004
13. Mont MA, Beaver WB, Dysart SH, Barrington JW, Del Gaizo D. Local infiltration analgesia with liposomal bupivacaine improves pain scores and reduces opioid use after total knee arthroplasty: results of a randomized controlled trial. J Arthroplasty. 2018;33(1):90-96. doi:10.1016/j.arth.2017.07.024
14. Hadlandsmyth K, Vander Weg MW, McCoy KD, Mosher HJ, Vaughan-Sarrazin MS, Lund BC. Risk for prolonged opioid use following total knee arthroplasty in veterans. J Arthroplasty. 2018;33(1):119-123. doi:10.1016/j.arth.2017.08.022
15. Nielsen S, Degenhardt L, Hoban B, Gisev N. A synthesis of oral morphine equivalents (OME) for opioid utilisation studies. Pharmacoepidemiol Drug Saf. 2016;25(6):733-737. doi:10.1002/pds.3945
16. Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Statist Soc B. 1995;57(1):289-300. doi:10.1111/j.2517-6161.1995.tb02031.x
17. Volkow ND, McLellan TA, Cotto JH, Karithanom M, Weiss SRB. Characteristics of opioid prescriptions in 2009. JAMA. 2011;305(13):1299-1301. doi:10.1001/jama.2011.401
18. Scholl L, Seth P, Kariisa M, Wilson N, Baldwin G. Drug and opioid-involved overdose deaths - United States, 2013-2017. MMWR Morb Mortal Wkly Rep. 2018;67(5152):1419-1427. doi:10.15585/mmwr.mm675152e1
19. Pichler L, Poeran J, Zubizarreta N, et al. Liposomal bupivacaine does not reduce inpatient opioid prescription or related complications after knee arthroplasty: a database analysis. Anesthesiology. 2018;129(4):689-699. doi:10.1097/ALN.0000000000002267
20. Jain RK, Porat MD, Klingenstein GG, Reid JJ, Post RE, Schoifet SD. The AAHKS Clinical Research Award: liposomal bupivacaine and periarticular injection are not superior to single-shot intra-articular injection for pain control in total knee arthroplasty. J Arthroplasty. 2016;31(9)(suppl):22-25. doi:10.1016/j.arth.2016.03.036
21. Zhao B, Ma X, Zhang J, Ma J, Cao Q. The efficacy of local liposomal bupivacaine infiltration on pain and recovery after total joint arthroplasty: a systematic review and meta-analysis of randomized controlled trials. Medicine (Baltimore). 2019;98(3):e14092. doi:10.1097/MD.0000000000014092
22. Schroer WC, Diesfeld PG, LeMarr AR, Morton DJ, Reedy ME. Does extended-release liposomal bupivacaine better control pain than bupivacaine after total knee arthroplasty (TKA)? A prospective, randomized clinical trial. J Arthroplasty. 2015;30(9)(suppl):64-67. doi:10.1016/j.arth.2015.01.059
23. Ma J, Zhang W, Yao S. Liposomal bupivacaine infiltration versus femoral nerve block for pain control in total knee arthroplasty: a systematic review and meta-analysis. Int J Surg. 2016;36(Pt A): 44-55. doi:10.1016/j.ijsu.2016.10.007
24. Barrington JW, Emerson RH, Lovald ST, Lombardi AV, Berend KR. No difference in early analgesia between liposomal bupivacaine injection and intrathecal morphine after TKA. Clin Orthop Relat Res. 2017;475(1):94-105. doi:10.1007/s11999-016-4931-z
25. Snyder MA, Scheuerman CM, Gregg JL, Ruhnke CJ, Eten K. Improving total knee arthroplasty perioperative pain management using a periarticular injection with bupivacaine liposomal suspension. Arthroplast Today. 2016;2(1):37-42. doi:10.1016/j.artd.2015.05.005
26. Kuang MJ,Du Y, Ma JX, He W, Fu L, Ma XL. The efficacy of liposomal bupivacaine using periarticular injection in total knee arthroplasty: a systematic review and meta-analysis. J Arthroplasty. 2017;32(4):1395-1402. doi:10.1016/j.arth.2016.12.025
27. Sakamoto B, Keiser S, Meldrum R, Harker G, Freese A. Efficacy of liposomal bupivacaine infiltration on the management of total knee arthroplasty. JAMA Surg. 2017;152(1):90-95. doi:10.1001/jamasurg.2016.3474
28. Collett GA, Song K, Jaramillo CA, Potter JS, Finley EP, Pugh MJ. Prevalence of central nervous system polypharmacy and associations with overdose and suicide-related behaviors in Iraq and Afghanistan war veterans in VA care 2010-2011. Drugs Real World Outcomes. 2016;3(1):45-52. doi:10.1007/s40801-015-0055-0
29. US Department of Veterans Affairs. HERC inpatient average cost data. Updated April 2, 2021. Accessed April 16, 2021. https://www.herc.research.va.gov/include/page.asp?id=inpatient#herc-inpat-avg-cost
30. Perruccio AV, Fitzpatrick J, Power JD, et al. Sex-modified effects of depression, low back pain, and comorbidities on pain after total knee arthroplasty for osteoarthritis. Arthritis Care Res (Hoboken). 2020;72(8):1074-1080. doi:10.1002/acr.24002
NSAIDs don’t make COVID-19 worse in hospitalized patients
NSAIDs don’t boost the risk of more severe disease or death in hospitalized patients with COVID-19, a new study finds.
“To our knowledge, our prospective study includes the largest number of patients admitted to hospital with COVID-19 to date, and adds to the literature on the safety of NSAIDs and in-hospital outcomes. NSAIDs do not appear to increase the risk of worse in-hospital outcomes ...” the study authors wrote. “NSAIDs are an important analgesic modality and have a vital opioid-sparing role in pain management. Patients and clinicians should be reassured by these findings that NSAIDs are safe in the context of the pandemic.”
The report was published online May 7 in The Lancet Rheumatology and led by clinical research fellow Thomas M. Drake, MBChB, of the University of Edinburgh’s Usher Institute.
For more than a year, researchers worldwide have debated about whether NSAIDs spell trouble for people at risk of COVID-19. In March 2020, French health officials announced that use of the painkillers such as NSAIDs may increase the severity of the disease, and they recommended that patients take acetaminophen instead. The National Health Service in the United Kingdom made a similar recommendation. But other agencies didn’t believe there was enough evidence to support ditching NSAIDs, and recent research studies published in Annals of the Rheumatic Diseases and PLoS Medicine suggested they may be right.
For the new study, researchers identified 72,179 patients who were treated for COVID-19 in British hospitals during January-August 2020. About 56% were men, 74% were White, and 6% took NSAIDs on a regular basis before they entered the hospital. The average age was 70.
The researchers examined whether the patients in either group were more or less likely to die in the hospital, be admitted into a critical care unit, need oxygen treatment, need a ventilator, or suffer kidney injury.
In terms of outcomes, there weren’t any major gaps between the groups overall. The differences in most comparisons were statistically insignificant. For example, 31% of those who didn’t take NSAIDs died vs. 30% of those who did (P = .227). In both groups, 14% required critical care admission (P = .476).
The researchers then focused on two matched groups of 4,205 patients: One group used NSAIDs regularly, and the other group didn’t. The difference in risk of death in those who took NSAIDs vs. those who didn’t was statistically insignificant (odds ratio, 0.95; 95% confidence interval, 0.84-1.07; P = .35). Other comparisons were also statistically insignificant.
The findings offer insight into whether the use of NSAIDs might actually be helpful for patients who develop COVID-19. Scientists believe that COVID-19 is linked to inflammation in the body, and NSAIDs, of course, reduce inflammation. But the researchers didn’t turn up any sign of a benefit.
The new study has some weaknesses: It doesn’t say anything about whether NSAIDs have an impact on whether people get COVID-19 in the first place. Researchers don’t know if high use of NSAIDs may affect the severity of the disease. And it doesn’t examine the potential effect of acetaminophen, although other research suggests the drug also may not cause harm in patients with COVID-19.
Still, the researchers say the study is the largest of its kind to look at the use of NSAIDs by patients who are admitted to the hospital with COVID-19. “Considering all the evidence, if there was an extreme effect of NSAIDs on COVID-19 outcomes or severity, this would have been observed in one or more of the studies that have been done, including the present study,” they wrote.
In a commentary that accompanied the study, three physicians from hospitals in Denmark, led by Kristian Kragholm, MD, of Aalborg University Hospital, praised the research and wrote that it adds to “a growing body of evidence” that NSAIDs don’t make things worse for patients with COVID-19.
The study was funded by the U.K. National Institute for Health Research and the U.K. Medical Research Council. The study and commentary authors reported no relevant disclosures.
NSAIDs don’t boost the risk of more severe disease or death in hospitalized patients with COVID-19, a new study finds.
“To our knowledge, our prospective study includes the largest number of patients admitted to hospital with COVID-19 to date, and adds to the literature on the safety of NSAIDs and in-hospital outcomes. NSAIDs do not appear to increase the risk of worse in-hospital outcomes ...” the study authors wrote. “NSAIDs are an important analgesic modality and have a vital opioid-sparing role in pain management. Patients and clinicians should be reassured by these findings that NSAIDs are safe in the context of the pandemic.”
The report was published online May 7 in The Lancet Rheumatology and led by clinical research fellow Thomas M. Drake, MBChB, of the University of Edinburgh’s Usher Institute.
For more than a year, researchers worldwide have debated about whether NSAIDs spell trouble for people at risk of COVID-19. In March 2020, French health officials announced that use of the painkillers such as NSAIDs may increase the severity of the disease, and they recommended that patients take acetaminophen instead. The National Health Service in the United Kingdom made a similar recommendation. But other agencies didn’t believe there was enough evidence to support ditching NSAIDs, and recent research studies published in Annals of the Rheumatic Diseases and PLoS Medicine suggested they may be right.
For the new study, researchers identified 72,179 patients who were treated for COVID-19 in British hospitals during January-August 2020. About 56% were men, 74% were White, and 6% took NSAIDs on a regular basis before they entered the hospital. The average age was 70.
The researchers examined whether the patients in either group were more or less likely to die in the hospital, be admitted into a critical care unit, need oxygen treatment, need a ventilator, or suffer kidney injury.
In terms of outcomes, there weren’t any major gaps between the groups overall. The differences in most comparisons were statistically insignificant. For example, 31% of those who didn’t take NSAIDs died vs. 30% of those who did (P = .227). In both groups, 14% required critical care admission (P = .476).
The researchers then focused on two matched groups of 4,205 patients: One group used NSAIDs regularly, and the other group didn’t. The difference in risk of death in those who took NSAIDs vs. those who didn’t was statistically insignificant (odds ratio, 0.95; 95% confidence interval, 0.84-1.07; P = .35). Other comparisons were also statistically insignificant.
The findings offer insight into whether the use of NSAIDs might actually be helpful for patients who develop COVID-19. Scientists believe that COVID-19 is linked to inflammation in the body, and NSAIDs, of course, reduce inflammation. But the researchers didn’t turn up any sign of a benefit.
The new study has some weaknesses: It doesn’t say anything about whether NSAIDs have an impact on whether people get COVID-19 in the first place. Researchers don’t know if high use of NSAIDs may affect the severity of the disease. And it doesn’t examine the potential effect of acetaminophen, although other research suggests the drug also may not cause harm in patients with COVID-19.
Still, the researchers say the study is the largest of its kind to look at the use of NSAIDs by patients who are admitted to the hospital with COVID-19. “Considering all the evidence, if there was an extreme effect of NSAIDs on COVID-19 outcomes or severity, this would have been observed in one or more of the studies that have been done, including the present study,” they wrote.
In a commentary that accompanied the study, three physicians from hospitals in Denmark, led by Kristian Kragholm, MD, of Aalborg University Hospital, praised the research and wrote that it adds to “a growing body of evidence” that NSAIDs don’t make things worse for patients with COVID-19.
The study was funded by the U.K. National Institute for Health Research and the U.K. Medical Research Council. The study and commentary authors reported no relevant disclosures.
NSAIDs don’t boost the risk of more severe disease or death in hospitalized patients with COVID-19, a new study finds.
“To our knowledge, our prospective study includes the largest number of patients admitted to hospital with COVID-19 to date, and adds to the literature on the safety of NSAIDs and in-hospital outcomes. NSAIDs do not appear to increase the risk of worse in-hospital outcomes ...” the study authors wrote. “NSAIDs are an important analgesic modality and have a vital opioid-sparing role in pain management. Patients and clinicians should be reassured by these findings that NSAIDs are safe in the context of the pandemic.”
The report was published online May 7 in The Lancet Rheumatology and led by clinical research fellow Thomas M. Drake, MBChB, of the University of Edinburgh’s Usher Institute.
For more than a year, researchers worldwide have debated about whether NSAIDs spell trouble for people at risk of COVID-19. In March 2020, French health officials announced that use of the painkillers such as NSAIDs may increase the severity of the disease, and they recommended that patients take acetaminophen instead. The National Health Service in the United Kingdom made a similar recommendation. But other agencies didn’t believe there was enough evidence to support ditching NSAIDs, and recent research studies published in Annals of the Rheumatic Diseases and PLoS Medicine suggested they may be right.
For the new study, researchers identified 72,179 patients who were treated for COVID-19 in British hospitals during January-August 2020. About 56% were men, 74% were White, and 6% took NSAIDs on a regular basis before they entered the hospital. The average age was 70.
The researchers examined whether the patients in either group were more or less likely to die in the hospital, be admitted into a critical care unit, need oxygen treatment, need a ventilator, or suffer kidney injury.
In terms of outcomes, there weren’t any major gaps between the groups overall. The differences in most comparisons were statistically insignificant. For example, 31% of those who didn’t take NSAIDs died vs. 30% of those who did (P = .227). In both groups, 14% required critical care admission (P = .476).
The researchers then focused on two matched groups of 4,205 patients: One group used NSAIDs regularly, and the other group didn’t. The difference in risk of death in those who took NSAIDs vs. those who didn’t was statistically insignificant (odds ratio, 0.95; 95% confidence interval, 0.84-1.07; P = .35). Other comparisons were also statistically insignificant.
The findings offer insight into whether the use of NSAIDs might actually be helpful for patients who develop COVID-19. Scientists believe that COVID-19 is linked to inflammation in the body, and NSAIDs, of course, reduce inflammation. But the researchers didn’t turn up any sign of a benefit.
The new study has some weaknesses: It doesn’t say anything about whether NSAIDs have an impact on whether people get COVID-19 in the first place. Researchers don’t know if high use of NSAIDs may affect the severity of the disease. And it doesn’t examine the potential effect of acetaminophen, although other research suggests the drug also may not cause harm in patients with COVID-19.
Still, the researchers say the study is the largest of its kind to look at the use of NSAIDs by patients who are admitted to the hospital with COVID-19. “Considering all the evidence, if there was an extreme effect of NSAIDs on COVID-19 outcomes or severity, this would have been observed in one or more of the studies that have been done, including the present study,” they wrote.
In a commentary that accompanied the study, three physicians from hospitals in Denmark, led by Kristian Kragholm, MD, of Aalborg University Hospital, praised the research and wrote that it adds to “a growing body of evidence” that NSAIDs don’t make things worse for patients with COVID-19.
The study was funded by the U.K. National Institute for Health Research and the U.K. Medical Research Council. The study and commentary authors reported no relevant disclosures.
FROM THE LANCET RHEUMATOLOGY
Transcranial brain stimulation can modulate placebo and nocebo experiences
, according to results of a new study published in the Proceedings of the National Academy of Sciences (PNAS).
“Placebo and nocebo effects are a critical component of clinical care and efficacy studies,” said senior author Jian Kong, MD, associate professor in the department of psychiatry at Massachusetts General Hospital, Charlestown campus. “Harnessing these effects in clinical practice and research could facilitate the development of new pain management methods,” he said. “Healing may involve multiple components: the self-healing properties of the body; the nonspecific effects of treatment (i.e., placebo effect); and the specific effect of a physical or pharmacologic intervention. Therefore, enhancing the placebo effect may ultimately boost the overall therapeutic effect of existing treatment,” he explained, emphasizing that the results are preliminary and should be interpreted with caution.
The authors noted that reducing nocebo effects could also be a major benefit “since patients discontinue prescribed medications, make unnecessary medical visits, and take additional medications to counteract adverse effects that are actually nocebo effects.”
Testing the hypothesis
The randomized, double-blind, sham-controlled study used transcranial direct current stimulation (tDCS), which delivers an electrical current to the brain via scalp electrodes. The aim was to see if stimulating the dorsolateral prefrontal cortex with tDCS could alter the brain’s perception of placebo and nocebo experiences.
The study included 81 participants (37 females, mean age: 27.4 years), who were randomized into one of three tDCS groups (anodal, cathodal, or sham).
All participants were first conditioned to believe that an inert cream was either lidocaine or capsaicin and that this cream could either dull the impact of a painful heat stimulus (placebo analgesia) or exacerbate it (nocebo hyperalgesia). Participants were then placed into a functional MRI scanner where tDCS was initiated. Painful stimuli were then applied to spots on their forearms where they believed they had either lidocaine, capsaicin, or a neutral control cream and they rated the pain using the Gracely Sensory Scale.
Placebo analgesia was defined as the difference between perceived pain intensity where participants believed they had lidocaine cream compared with where they believed they had control cream. Nocebo hyperalgesia was defined as the difference between perceived pain intensity where they believed they had capsaicin cream compared with where they believed they had control cream.
The researchers found that compared with sham tDCS, cathodal tDCS showed significant effects in increasing placebo analgesia and brain responses in the ventromedial prefrontal cortex (vmPFC), while anodal tDCS showed significant effects in inhibiting nocebo hyperalgesia and brain responses in the insula.
“The potential to enhance salubrious placebo effects and/or diminish treatment-interfering nocebo effects may have clinical significance,” the authors noted. “For example, clinical studies have suggested that expectancy is positively associated with chronic pain improvement, and using conditioning-like expectancy manipulation, we have shown that significantly boosting expectancy can improve treatment outcome.”
Proof of concept
Asked to comment on the study, Brian E. McGeeney, MD, of the John R. Graham Headache Center at Brigham and Women’s Faulkner Hospital in Boston, said “the findings are a proof of concept that it is possible to use noninvasive brain stimulation to modulate placebo and nocebo pain effects.”
Although the findings do not have immediate clinical application, they are “exciting” and “break new ground in expectancy research,” he said.
“It is important to recognize that the researchers are trying to utilize a purported expectancy mechanism rather than attempting to alter placebo/nocebo by verbal and other cues. It remains to be seen whether the manipulation of brief experimental pain like this can translate into altered chronic pain over time, the main clinical goal. Current tDCS therapy for various reasons is necessarily brief and one can ask whether there are meaningful changes from brief stimulation. Such results can foster speculation as to whether direct strategic placement of intracranial stimulation leads could result in more longstanding similar benefits.”
Dr. Kong holds equity in a startup company (MNT) and a pending patent to develop new peripheral neuromodulation tools, but declares no conflict of interest. All other authors declare no conflict of interest.
, according to results of a new study published in the Proceedings of the National Academy of Sciences (PNAS).
“Placebo and nocebo effects are a critical component of clinical care and efficacy studies,” said senior author Jian Kong, MD, associate professor in the department of psychiatry at Massachusetts General Hospital, Charlestown campus. “Harnessing these effects in clinical practice and research could facilitate the development of new pain management methods,” he said. “Healing may involve multiple components: the self-healing properties of the body; the nonspecific effects of treatment (i.e., placebo effect); and the specific effect of a physical or pharmacologic intervention. Therefore, enhancing the placebo effect may ultimately boost the overall therapeutic effect of existing treatment,” he explained, emphasizing that the results are preliminary and should be interpreted with caution.
The authors noted that reducing nocebo effects could also be a major benefit “since patients discontinue prescribed medications, make unnecessary medical visits, and take additional medications to counteract adverse effects that are actually nocebo effects.”
Testing the hypothesis
The randomized, double-blind, sham-controlled study used transcranial direct current stimulation (tDCS), which delivers an electrical current to the brain via scalp electrodes. The aim was to see if stimulating the dorsolateral prefrontal cortex with tDCS could alter the brain’s perception of placebo and nocebo experiences.
The study included 81 participants (37 females, mean age: 27.4 years), who were randomized into one of three tDCS groups (anodal, cathodal, or sham).
All participants were first conditioned to believe that an inert cream was either lidocaine or capsaicin and that this cream could either dull the impact of a painful heat stimulus (placebo analgesia) or exacerbate it (nocebo hyperalgesia). Participants were then placed into a functional MRI scanner where tDCS was initiated. Painful stimuli were then applied to spots on their forearms where they believed they had either lidocaine, capsaicin, or a neutral control cream and they rated the pain using the Gracely Sensory Scale.
Placebo analgesia was defined as the difference between perceived pain intensity where participants believed they had lidocaine cream compared with where they believed they had control cream. Nocebo hyperalgesia was defined as the difference between perceived pain intensity where they believed they had capsaicin cream compared with where they believed they had control cream.
The researchers found that compared with sham tDCS, cathodal tDCS showed significant effects in increasing placebo analgesia and brain responses in the ventromedial prefrontal cortex (vmPFC), while anodal tDCS showed significant effects in inhibiting nocebo hyperalgesia and brain responses in the insula.
“The potential to enhance salubrious placebo effects and/or diminish treatment-interfering nocebo effects may have clinical significance,” the authors noted. “For example, clinical studies have suggested that expectancy is positively associated with chronic pain improvement, and using conditioning-like expectancy manipulation, we have shown that significantly boosting expectancy can improve treatment outcome.”
Proof of concept
Asked to comment on the study, Brian E. McGeeney, MD, of the John R. Graham Headache Center at Brigham and Women’s Faulkner Hospital in Boston, said “the findings are a proof of concept that it is possible to use noninvasive brain stimulation to modulate placebo and nocebo pain effects.”
Although the findings do not have immediate clinical application, they are “exciting” and “break new ground in expectancy research,” he said.
“It is important to recognize that the researchers are trying to utilize a purported expectancy mechanism rather than attempting to alter placebo/nocebo by verbal and other cues. It remains to be seen whether the manipulation of brief experimental pain like this can translate into altered chronic pain over time, the main clinical goal. Current tDCS therapy for various reasons is necessarily brief and one can ask whether there are meaningful changes from brief stimulation. Such results can foster speculation as to whether direct strategic placement of intracranial stimulation leads could result in more longstanding similar benefits.”
Dr. Kong holds equity in a startup company (MNT) and a pending patent to develop new peripheral neuromodulation tools, but declares no conflict of interest. All other authors declare no conflict of interest.
, according to results of a new study published in the Proceedings of the National Academy of Sciences (PNAS).
“Placebo and nocebo effects are a critical component of clinical care and efficacy studies,” said senior author Jian Kong, MD, associate professor in the department of psychiatry at Massachusetts General Hospital, Charlestown campus. “Harnessing these effects in clinical practice and research could facilitate the development of new pain management methods,” he said. “Healing may involve multiple components: the self-healing properties of the body; the nonspecific effects of treatment (i.e., placebo effect); and the specific effect of a physical or pharmacologic intervention. Therefore, enhancing the placebo effect may ultimately boost the overall therapeutic effect of existing treatment,” he explained, emphasizing that the results are preliminary and should be interpreted with caution.
The authors noted that reducing nocebo effects could also be a major benefit “since patients discontinue prescribed medications, make unnecessary medical visits, and take additional medications to counteract adverse effects that are actually nocebo effects.”
Testing the hypothesis
The randomized, double-blind, sham-controlled study used transcranial direct current stimulation (tDCS), which delivers an electrical current to the brain via scalp electrodes. The aim was to see if stimulating the dorsolateral prefrontal cortex with tDCS could alter the brain’s perception of placebo and nocebo experiences.
The study included 81 participants (37 females, mean age: 27.4 years), who were randomized into one of three tDCS groups (anodal, cathodal, or sham).
All participants were first conditioned to believe that an inert cream was either lidocaine or capsaicin and that this cream could either dull the impact of a painful heat stimulus (placebo analgesia) or exacerbate it (nocebo hyperalgesia). Participants were then placed into a functional MRI scanner where tDCS was initiated. Painful stimuli were then applied to spots on their forearms where they believed they had either lidocaine, capsaicin, or a neutral control cream and they rated the pain using the Gracely Sensory Scale.
Placebo analgesia was defined as the difference between perceived pain intensity where participants believed they had lidocaine cream compared with where they believed they had control cream. Nocebo hyperalgesia was defined as the difference between perceived pain intensity where they believed they had capsaicin cream compared with where they believed they had control cream.
The researchers found that compared with sham tDCS, cathodal tDCS showed significant effects in increasing placebo analgesia and brain responses in the ventromedial prefrontal cortex (vmPFC), while anodal tDCS showed significant effects in inhibiting nocebo hyperalgesia and brain responses in the insula.
“The potential to enhance salubrious placebo effects and/or diminish treatment-interfering nocebo effects may have clinical significance,” the authors noted. “For example, clinical studies have suggested that expectancy is positively associated with chronic pain improvement, and using conditioning-like expectancy manipulation, we have shown that significantly boosting expectancy can improve treatment outcome.”
Proof of concept
Asked to comment on the study, Brian E. McGeeney, MD, of the John R. Graham Headache Center at Brigham and Women’s Faulkner Hospital in Boston, said “the findings are a proof of concept that it is possible to use noninvasive brain stimulation to modulate placebo and nocebo pain effects.”
Although the findings do not have immediate clinical application, they are “exciting” and “break new ground in expectancy research,” he said.
“It is important to recognize that the researchers are trying to utilize a purported expectancy mechanism rather than attempting to alter placebo/nocebo by verbal and other cues. It remains to be seen whether the manipulation of brief experimental pain like this can translate into altered chronic pain over time, the main clinical goal. Current tDCS therapy for various reasons is necessarily brief and one can ask whether there are meaningful changes from brief stimulation. Such results can foster speculation as to whether direct strategic placement of intracranial stimulation leads could result in more longstanding similar benefits.”
Dr. Kong holds equity in a startup company (MNT) and a pending patent to develop new peripheral neuromodulation tools, but declares no conflict of interest. All other authors declare no conflict of interest.
FROM PNAS
Hypertension worsened by commonly used prescription meds
Nearly half of these American adults had hypertension, and in this subgroup, 18.5% reported using a prescription drug known to increase blood pressure. The most widely used class of agents with this effect was antidepressants, used by 8.7%; followed by nonsteroidal anti-inflammatory drugs (NSAIDs), used by 6.5%; steroids, 1.9%; estrogens, 1.7%; and several other agents each used by fewer than 1% of the study cohort, John Vitarello, MD, said during a press briefing on reports from the upcoming annual scientific sessions of the American College of Cardiology.
He and his associates estimated that this use of prescription drugs known to raise blood pressure could be what stands in the way of some 560,000-2.2 million Americans from having their hypertension under control, depending on the exact blood pressure impact that various pressure-increasing drugs have and presuming that half of those on blood pressure increasing agents could stop them and switch to alternative agents, said Dr. Vitarello, a researcher at Beth Israel Deaconess Medical Center in Boston.
He also highlighted that the study assessed only prescription drugs and did not examine OTC drug use, which may be especially relevant for the many people who regularly take NSAIDs.
“Clinicians should review the prescription and OTC drug use of patients with hypertension and consider stopping drugs that increase blood pressure or switching the patient to alternatives” that are blood pressure neutral, Dr. Vitarello said during the briefing. He cautioned that maintaining hypertensive patients on agents that raise their blood pressure can result in “prescribing cascades” where taking drugs that boost blood pressure results in need for intensified antihypertensive treatment.
An opportunity for NSAID alternatives
“This study hopefully raises awareness that there is a very high use of medications that increase blood pressure, and use of OTC agents could increase the rate even higher” said Eugene Yang, MD, a cardiologist and codirector of the Cardiovascular Wellness and Prevention Program of the University of Washington, Seattle. Substituting for certain antidepressant agents may often not be realistic, but an opportunity exists for reducing NSAID use, a class also linked with an increased risk for bleeding and other adverse effects, Dr. Yang said during the briefing. Minimizing use of NSAIDs including ibuprofen and naproxen use “is something to think about,” he suggested.
“The effect of NSAIDs on blood pressure is not well studied and can vary from person to person” noted Dr. Vitarello, who added that higher NSAID dosages and more prolonged use likely increase the risk for an adverse effect on blood pressure. One reasonable option is to encourage patients to use an alternative class of pain reliever such as acetaminophen.
It remains “a challenge” to discern differences in adverse blood pressure effects, and in all adverse cardiovascular effects among different NSAIDs, said Dr. Yang. Results from “some studies show that certain NSAIDs may be safer, but other studies did not. We need to be very careful using NSAIDs because, on average, they increase blood pressure by about 3 mm Hg. We need to be mindful to try to prescribe alternative agents, like acetaminophen.”
A decade of data from NHANES
The analysis run by Dr. Vitarello and associates used data from 27,599 American adults included in the NHANES during 2009-2018, and focused on the 44% who either had an average blood pressure measurement of at least 130/80 mm Hg or reported having ever been told by a clinician that they had hypertension. The NHANES assessments included the prescription medications taken by each participant. The prevalence of using at least one prescription drug known to raise blood pressure was 24% among women and 14% among men, and 4% of those with hypertension were on two or more pressure-increasing agents.
The researchers based their identification of pressure-increasing prescription drugs on the list included in the 2017 guideline for managing high blood pressure from the American College of Cardiology and American Heart Association. This list specifies that the antidepressants that raise blood pressure are the monoamine oxidase inhibitors, serotonin norepinephrine reuptake inhibitors, and tricyclic antidepressants.
Dr. Vitarello and Dr. Yang had no disclosures.
Nearly half of these American adults had hypertension, and in this subgroup, 18.5% reported using a prescription drug known to increase blood pressure. The most widely used class of agents with this effect was antidepressants, used by 8.7%; followed by nonsteroidal anti-inflammatory drugs (NSAIDs), used by 6.5%; steroids, 1.9%; estrogens, 1.7%; and several other agents each used by fewer than 1% of the study cohort, John Vitarello, MD, said during a press briefing on reports from the upcoming annual scientific sessions of the American College of Cardiology.
He and his associates estimated that this use of prescription drugs known to raise blood pressure could be what stands in the way of some 560,000-2.2 million Americans from having their hypertension under control, depending on the exact blood pressure impact that various pressure-increasing drugs have and presuming that half of those on blood pressure increasing agents could stop them and switch to alternative agents, said Dr. Vitarello, a researcher at Beth Israel Deaconess Medical Center in Boston.
He also highlighted that the study assessed only prescription drugs and did not examine OTC drug use, which may be especially relevant for the many people who regularly take NSAIDs.
“Clinicians should review the prescription and OTC drug use of patients with hypertension and consider stopping drugs that increase blood pressure or switching the patient to alternatives” that are blood pressure neutral, Dr. Vitarello said during the briefing. He cautioned that maintaining hypertensive patients on agents that raise their blood pressure can result in “prescribing cascades” where taking drugs that boost blood pressure results in need for intensified antihypertensive treatment.
An opportunity for NSAID alternatives
“This study hopefully raises awareness that there is a very high use of medications that increase blood pressure, and use of OTC agents could increase the rate even higher” said Eugene Yang, MD, a cardiologist and codirector of the Cardiovascular Wellness and Prevention Program of the University of Washington, Seattle. Substituting for certain antidepressant agents may often not be realistic, but an opportunity exists for reducing NSAID use, a class also linked with an increased risk for bleeding and other adverse effects, Dr. Yang said during the briefing. Minimizing use of NSAIDs including ibuprofen and naproxen use “is something to think about,” he suggested.
“The effect of NSAIDs on blood pressure is not well studied and can vary from person to person” noted Dr. Vitarello, who added that higher NSAID dosages and more prolonged use likely increase the risk for an adverse effect on blood pressure. One reasonable option is to encourage patients to use an alternative class of pain reliever such as acetaminophen.
It remains “a challenge” to discern differences in adverse blood pressure effects, and in all adverse cardiovascular effects among different NSAIDs, said Dr. Yang. Results from “some studies show that certain NSAIDs may be safer, but other studies did not. We need to be very careful using NSAIDs because, on average, they increase blood pressure by about 3 mm Hg. We need to be mindful to try to prescribe alternative agents, like acetaminophen.”
A decade of data from NHANES
The analysis run by Dr. Vitarello and associates used data from 27,599 American adults included in the NHANES during 2009-2018, and focused on the 44% who either had an average blood pressure measurement of at least 130/80 mm Hg or reported having ever been told by a clinician that they had hypertension. The NHANES assessments included the prescription medications taken by each participant. The prevalence of using at least one prescription drug known to raise blood pressure was 24% among women and 14% among men, and 4% of those with hypertension were on two or more pressure-increasing agents.
The researchers based their identification of pressure-increasing prescription drugs on the list included in the 2017 guideline for managing high blood pressure from the American College of Cardiology and American Heart Association. This list specifies that the antidepressants that raise blood pressure are the monoamine oxidase inhibitors, serotonin norepinephrine reuptake inhibitors, and tricyclic antidepressants.
Dr. Vitarello and Dr. Yang had no disclosures.
Nearly half of these American adults had hypertension, and in this subgroup, 18.5% reported using a prescription drug known to increase blood pressure. The most widely used class of agents with this effect was antidepressants, used by 8.7%; followed by nonsteroidal anti-inflammatory drugs (NSAIDs), used by 6.5%; steroids, 1.9%; estrogens, 1.7%; and several other agents each used by fewer than 1% of the study cohort, John Vitarello, MD, said during a press briefing on reports from the upcoming annual scientific sessions of the American College of Cardiology.
He and his associates estimated that this use of prescription drugs known to raise blood pressure could be what stands in the way of some 560,000-2.2 million Americans from having their hypertension under control, depending on the exact blood pressure impact that various pressure-increasing drugs have and presuming that half of those on blood pressure increasing agents could stop them and switch to alternative agents, said Dr. Vitarello, a researcher at Beth Israel Deaconess Medical Center in Boston.
He also highlighted that the study assessed only prescription drugs and did not examine OTC drug use, which may be especially relevant for the many people who regularly take NSAIDs.
“Clinicians should review the prescription and OTC drug use of patients with hypertension and consider stopping drugs that increase blood pressure or switching the patient to alternatives” that are blood pressure neutral, Dr. Vitarello said during the briefing. He cautioned that maintaining hypertensive patients on agents that raise their blood pressure can result in “prescribing cascades” where taking drugs that boost blood pressure results in need for intensified antihypertensive treatment.
An opportunity for NSAID alternatives
“This study hopefully raises awareness that there is a very high use of medications that increase blood pressure, and use of OTC agents could increase the rate even higher” said Eugene Yang, MD, a cardiologist and codirector of the Cardiovascular Wellness and Prevention Program of the University of Washington, Seattle. Substituting for certain antidepressant agents may often not be realistic, but an opportunity exists for reducing NSAID use, a class also linked with an increased risk for bleeding and other adverse effects, Dr. Yang said during the briefing. Minimizing use of NSAIDs including ibuprofen and naproxen use “is something to think about,” he suggested.
“The effect of NSAIDs on blood pressure is not well studied and can vary from person to person” noted Dr. Vitarello, who added that higher NSAID dosages and more prolonged use likely increase the risk for an adverse effect on blood pressure. One reasonable option is to encourage patients to use an alternative class of pain reliever such as acetaminophen.
It remains “a challenge” to discern differences in adverse blood pressure effects, and in all adverse cardiovascular effects among different NSAIDs, said Dr. Yang. Results from “some studies show that certain NSAIDs may be safer, but other studies did not. We need to be very careful using NSAIDs because, on average, they increase blood pressure by about 3 mm Hg. We need to be mindful to try to prescribe alternative agents, like acetaminophen.”
A decade of data from NHANES
The analysis run by Dr. Vitarello and associates used data from 27,599 American adults included in the NHANES during 2009-2018, and focused on the 44% who either had an average blood pressure measurement of at least 130/80 mm Hg or reported having ever been told by a clinician that they had hypertension. The NHANES assessments included the prescription medications taken by each participant. The prevalence of using at least one prescription drug known to raise blood pressure was 24% among women and 14% among men, and 4% of those with hypertension were on two or more pressure-increasing agents.
The researchers based their identification of pressure-increasing prescription drugs on the list included in the 2017 guideline for managing high blood pressure from the American College of Cardiology and American Heart Association. This list specifies that the antidepressants that raise blood pressure are the monoamine oxidase inhibitors, serotonin norepinephrine reuptake inhibitors, and tricyclic antidepressants.
Dr. Vitarello and Dr. Yang had no disclosures.
FROM ACC 2021
Pediatric cancer survivors at risk for opioid misuse
Survivors of childhood cancers are at increased risk for prescription opioid misuse compared with their peers, a review of a claims database revealed.
Among more than 8,000 patients age 21 or younger who had completed treatment for hematologic, central nervous system, bone, or gonadal cancers, survivors were significantly more likely than were their peers to have an opioid prescription, longer duration of prescription, and higher daily doses of opioids, and to have opioid prescriptions overlapping for a week or more, reported Xu Ji, PhD, of Emory University in Atlanta.
Teenage and young adult patients were at higher risk than were patients younger than 12, and the risk was highest among patients who had been treated for bone malignancies, as well as those who had undergone any hematopoietic stem cell transplant.
“These findings suggest that health care providers who regularly see survivors should explore nonopioid options to help prevent opioid misuse, and screen for potential misuse in those who actually receive opioids,” she said in an oral abstract presented during the annual meeting of the American Society of Pediatric Hematology/Oncology.
“This is a really important topic, and something that’s probably been underinvestigated and underexplored in our patient population,” said session comoderator Sheri Spunt, MD, Endowed Professor of Pediatric Cancer at Stanford (Calif.) University.
Database review
Dr. Ji and colleagues used the IBM MarketScan Commercial Claims and Encounters database from 2009 to 2018 to examine prescription opioid use, potential misuse, and substance use disorders in pediatric cancer survivors in the first year after completion of therapy, and to identify factors associated with risk for misuse or substance use disorders. Specifically, the period of interest was the first year after completion of all treatments, including surgery, chemotherapy, radiation, and stem cell transplant (Abstract 2015).
They looked at deidentified records on any opioid prescription and for treatment of any opioid use or substance use disorder (alcohol, psychotherapeutic drugs, marijuana, or illicit drug use disorders).
They defined indicators of potential misuse as either prescriptions for long-acting or extended-release opioids for acute pain conditions; opioid and benzodiazepine prescriptions overlapping by a week or more; opioid prescriptions overlapping by a week or more; high daily opioid dosage (prescribed daily dose of 100 or greater morphine milligram equivalent [MME]; and/or opioid dose escalation (an increase of at least 50% in mean MMEs per month twice consecutively within 1 year).
They compared outcomes between a total of 8,635 survivors and 44,175 controls, matched on a 1:5 basis with survivors by age, sex, and region, and continuous enrollment during the 1-year posttherapy period.
In each of three age categories – 0 to 11 years, 12 to 17 years, and 18 years and older – survivors were significantly more likely to have received an opioid prescription, at 15% for the youngest survivors vs. 2% of controls, 25% vs. 8% for 12- to 17-year-olds, and 28% vs. 12% for those 18 and older (P < .01 for all three comparisons).
Survivors were also significantly more likely to have any indicator of potential misuse (1.6% vs. 0.1%, 4.6% vs. 0.5%, and 7.4% vs. 1.2%, respectively, P < .001 for all) and both the youngest and oldest groups (but not 12- to 17-year-olds) were significantly more like to have opioid or substance use disorder (0.4% vs. 0% for 0-11 years, 5.76% vs. 4.2% for 18 years and older, P < .001 for both).
Among patients with any opioid prescription, survivors were significantly more likely than were controls of any age to have indicators for potential misuse. For example, 13% of survivors aged 18 years and older had prescriptions for high opioid doses, compared with 5% of controls, and 12% had prescription overlap, vs. 2%.
Compared with patients with leukemia, patients treated for bone malignancies had a 6% greater risk for having any indicator of misuse, while patients with other malignancies were at slightly lower risk for misuse than those who completed leukemia therapy.
Patients who received any stem cell transplant had an 8.4% greater risk for misuse compared with patients who had surgery only.
Opioids pre- and posttreatment?
“Being someone who takes care of a lot of bone cancer patients, I do see patients with these issues,” Dr. Spunt said.
Audience member Jack H. Staddon, MD, PhD, of the Billings (Montana) Clinic, noted the possibility that opioid use during treatment may have been carried on into the posttreatment period, and asked whether use of narcotics during treatment was an independent risk factor for posttreatment narcotic use or misuse.
The researchers plan to investigate this question in future studies, Dr. Ji replied.
They did not report a study funding source. Dr. Ji and coauthors and Dr. Staddon reported no relevant disclosures.
Survivors of childhood cancers are at increased risk for prescription opioid misuse compared with their peers, a review of a claims database revealed.
Among more than 8,000 patients age 21 or younger who had completed treatment for hematologic, central nervous system, bone, or gonadal cancers, survivors were significantly more likely than were their peers to have an opioid prescription, longer duration of prescription, and higher daily doses of opioids, and to have opioid prescriptions overlapping for a week or more, reported Xu Ji, PhD, of Emory University in Atlanta.
Teenage and young adult patients were at higher risk than were patients younger than 12, and the risk was highest among patients who had been treated for bone malignancies, as well as those who had undergone any hematopoietic stem cell transplant.
“These findings suggest that health care providers who regularly see survivors should explore nonopioid options to help prevent opioid misuse, and screen for potential misuse in those who actually receive opioids,” she said in an oral abstract presented during the annual meeting of the American Society of Pediatric Hematology/Oncology.
“This is a really important topic, and something that’s probably been underinvestigated and underexplored in our patient population,” said session comoderator Sheri Spunt, MD, Endowed Professor of Pediatric Cancer at Stanford (Calif.) University.
Database review
Dr. Ji and colleagues used the IBM MarketScan Commercial Claims and Encounters database from 2009 to 2018 to examine prescription opioid use, potential misuse, and substance use disorders in pediatric cancer survivors in the first year after completion of therapy, and to identify factors associated with risk for misuse or substance use disorders. Specifically, the period of interest was the first year after completion of all treatments, including surgery, chemotherapy, radiation, and stem cell transplant (Abstract 2015).
They looked at deidentified records on any opioid prescription and for treatment of any opioid use or substance use disorder (alcohol, psychotherapeutic drugs, marijuana, or illicit drug use disorders).
They defined indicators of potential misuse as either prescriptions for long-acting or extended-release opioids for acute pain conditions; opioid and benzodiazepine prescriptions overlapping by a week or more; opioid prescriptions overlapping by a week or more; high daily opioid dosage (prescribed daily dose of 100 or greater morphine milligram equivalent [MME]; and/or opioid dose escalation (an increase of at least 50% in mean MMEs per month twice consecutively within 1 year).
They compared outcomes between a total of 8,635 survivors and 44,175 controls, matched on a 1:5 basis with survivors by age, sex, and region, and continuous enrollment during the 1-year posttherapy period.
In each of three age categories – 0 to 11 years, 12 to 17 years, and 18 years and older – survivors were significantly more likely to have received an opioid prescription, at 15% for the youngest survivors vs. 2% of controls, 25% vs. 8% for 12- to 17-year-olds, and 28% vs. 12% for those 18 and older (P < .01 for all three comparisons).
Survivors were also significantly more likely to have any indicator of potential misuse (1.6% vs. 0.1%, 4.6% vs. 0.5%, and 7.4% vs. 1.2%, respectively, P < .001 for all) and both the youngest and oldest groups (but not 12- to 17-year-olds) were significantly more like to have opioid or substance use disorder (0.4% vs. 0% for 0-11 years, 5.76% vs. 4.2% for 18 years and older, P < .001 for both).
Among patients with any opioid prescription, survivors were significantly more likely than were controls of any age to have indicators for potential misuse. For example, 13% of survivors aged 18 years and older had prescriptions for high opioid doses, compared with 5% of controls, and 12% had prescription overlap, vs. 2%.
Compared with patients with leukemia, patients treated for bone malignancies had a 6% greater risk for having any indicator of misuse, while patients with other malignancies were at slightly lower risk for misuse than those who completed leukemia therapy.
Patients who received any stem cell transplant had an 8.4% greater risk for misuse compared with patients who had surgery only.
Opioids pre- and posttreatment?
“Being someone who takes care of a lot of bone cancer patients, I do see patients with these issues,” Dr. Spunt said.
Audience member Jack H. Staddon, MD, PhD, of the Billings (Montana) Clinic, noted the possibility that opioid use during treatment may have been carried on into the posttreatment period, and asked whether use of narcotics during treatment was an independent risk factor for posttreatment narcotic use or misuse.
The researchers plan to investigate this question in future studies, Dr. Ji replied.
They did not report a study funding source. Dr. Ji and coauthors and Dr. Staddon reported no relevant disclosures.
Survivors of childhood cancers are at increased risk for prescription opioid misuse compared with their peers, a review of a claims database revealed.
Among more than 8,000 patients age 21 or younger who had completed treatment for hematologic, central nervous system, bone, or gonadal cancers, survivors were significantly more likely than were their peers to have an opioid prescription, longer duration of prescription, and higher daily doses of opioids, and to have opioid prescriptions overlapping for a week or more, reported Xu Ji, PhD, of Emory University in Atlanta.
Teenage and young adult patients were at higher risk than were patients younger than 12, and the risk was highest among patients who had been treated for bone malignancies, as well as those who had undergone any hematopoietic stem cell transplant.
“These findings suggest that health care providers who regularly see survivors should explore nonopioid options to help prevent opioid misuse, and screen for potential misuse in those who actually receive opioids,” she said in an oral abstract presented during the annual meeting of the American Society of Pediatric Hematology/Oncology.
“This is a really important topic, and something that’s probably been underinvestigated and underexplored in our patient population,” said session comoderator Sheri Spunt, MD, Endowed Professor of Pediatric Cancer at Stanford (Calif.) University.
Database review
Dr. Ji and colleagues used the IBM MarketScan Commercial Claims and Encounters database from 2009 to 2018 to examine prescription opioid use, potential misuse, and substance use disorders in pediatric cancer survivors in the first year after completion of therapy, and to identify factors associated with risk for misuse or substance use disorders. Specifically, the period of interest was the first year after completion of all treatments, including surgery, chemotherapy, radiation, and stem cell transplant (Abstract 2015).
They looked at deidentified records on any opioid prescription and for treatment of any opioid use or substance use disorder (alcohol, psychotherapeutic drugs, marijuana, or illicit drug use disorders).
They defined indicators of potential misuse as either prescriptions for long-acting or extended-release opioids for acute pain conditions; opioid and benzodiazepine prescriptions overlapping by a week or more; opioid prescriptions overlapping by a week or more; high daily opioid dosage (prescribed daily dose of 100 or greater morphine milligram equivalent [MME]; and/or opioid dose escalation (an increase of at least 50% in mean MMEs per month twice consecutively within 1 year).
They compared outcomes between a total of 8,635 survivors and 44,175 controls, matched on a 1:5 basis with survivors by age, sex, and region, and continuous enrollment during the 1-year posttherapy period.
In each of three age categories – 0 to 11 years, 12 to 17 years, and 18 years and older – survivors were significantly more likely to have received an opioid prescription, at 15% for the youngest survivors vs. 2% of controls, 25% vs. 8% for 12- to 17-year-olds, and 28% vs. 12% for those 18 and older (P < .01 for all three comparisons).
Survivors were also significantly more likely to have any indicator of potential misuse (1.6% vs. 0.1%, 4.6% vs. 0.5%, and 7.4% vs. 1.2%, respectively, P < .001 for all) and both the youngest and oldest groups (but not 12- to 17-year-olds) were significantly more like to have opioid or substance use disorder (0.4% vs. 0% for 0-11 years, 5.76% vs. 4.2% for 18 years and older, P < .001 for both).
Among patients with any opioid prescription, survivors were significantly more likely than were controls of any age to have indicators for potential misuse. For example, 13% of survivors aged 18 years and older had prescriptions for high opioid doses, compared with 5% of controls, and 12% had prescription overlap, vs. 2%.
Compared with patients with leukemia, patients treated for bone malignancies had a 6% greater risk for having any indicator of misuse, while patients with other malignancies were at slightly lower risk for misuse than those who completed leukemia therapy.
Patients who received any stem cell transplant had an 8.4% greater risk for misuse compared with patients who had surgery only.
Opioids pre- and posttreatment?
“Being someone who takes care of a lot of bone cancer patients, I do see patients with these issues,” Dr. Spunt said.
Audience member Jack H. Staddon, MD, PhD, of the Billings (Montana) Clinic, noted the possibility that opioid use during treatment may have been carried on into the posttreatment period, and asked whether use of narcotics during treatment was an independent risk factor for posttreatment narcotic use or misuse.
The researchers plan to investigate this question in future studies, Dr. Ji replied.
They did not report a study funding source. Dr. Ji and coauthors and Dr. Staddon reported no relevant disclosures.
FROM 2021 ASPHO CONFERENCE
High variability found in studies assessing hemophilia-related pain
Chronic pain is a common condition among people with hemophilia and is associated with joint deterioration because of repeated joint bleeds. This systematic review and meta-analysis aimed to determine the prevalence of chronic pain because of hemophilia and to analyze its interference in the lives of patients, according to Ana Cristina Paredes, a PhD student at the University of Minho, Braga, Portugal, and colleagues.
The manuscripts included in the study, which was published online in the Journal of Pain, were mostly observational, cross-sectional studies and one prospective investigation, published between 2009 and 2019.
The issue of pain is particularly important among people with hemophilia, as many adult patients suffer from distinct degrees of arthropathy and associated chronic pain, due to the lifelong occurrence of hemarthrosis, the authors noted. In an important distinction, according to the authors, people with hemophilia may therefore experience both acute pain during bleeds and chronic pain caused by joint deterioration. Acute pain ceases with the resolution of the bleeding episode, but the chronic pain is significantly more challenging, since it persists in time and may trigger changes in the nervous system, leading to peripheral or central sensitization.
Data in the assessed studies were collected from a variety of sources: hemophilia centers, online surveys, by mail, or through a national database, with return rates ranging from 29.2% to 98%. Overall, these studies comprised 4,772 adults, with individual sample sizes ranging from 21 to 2,253 patients, the authors added.
Conflicting results
Overall, there was a widely varying prevalence of hemophilia-related chronic pain reported across studies. Additionally, methodologies and sample characteristics varied widely. The meta-analyses revealed high heterogeneity between studies, and, therefore, pooled prevalence estimates values must be interpreted with caution, the authors stated.
All of the 11 selected studies included for meta-analysis and review reported on the prevalence of chronic pain caused by hemophilia. Chronic pain was assessed using direct questions developed by the authors in eight studies and using the European Haemophilia Therapy Standardization Board definition in three studies. The prevalence for global samples ranged widely from 17% to 84%.
Although there was high heterogeneity, the random-effects meta-analysis including all studies demonstrated a pooled prevalence of 46% of patients reporting chronic pain. Subgroup analyses of studies including all disease severities (mild, moderate, and severe; seven studies) revealed a pooled prevalence of 48%, but also with high heterogeneity. Looking at severe patients only (six studies), the chronic pain prevalence ranged from 33% to 86.4%, with a pooled prevalence of 53% and high heterogeneity, the authors added.
The wide disparity of the chronic pain prevalence seen across the studies is likely because of the fact that some investigations inquired about pain without distinguishing between acute (hemarthrosis-related) or chronic (arthropathy-related) pain, and without clarifying if the only focus is pain caused by hemophilia, or including all causes of pain complaints, according to the researchers.
“Concerning hemophilia-related chronic pain interference, it is striking that the existing literature does not distinguish between the impact of acute or chronic pain. Such a distinction is needed and should be made in future studies to ensure accurate accounts of hemophilia-related pain and to fully understand its interference according to the type of pain (acute vs. chronic). This information is relevant to promote targeted and effective treatment approaches,” the researchers concluded.
The research was supported by a Novo Nordisk HERO Research Grant 2015, the Portuguese Foundation for Science and Technology, and the Foundation for Science and Technology in Portugal. The authors declared they had no conflicts of interest.
Chronic pain is a common condition among people with hemophilia and is associated with joint deterioration because of repeated joint bleeds. This systematic review and meta-analysis aimed to determine the prevalence of chronic pain because of hemophilia and to analyze its interference in the lives of patients, according to Ana Cristina Paredes, a PhD student at the University of Minho, Braga, Portugal, and colleagues.
The manuscripts included in the study, which was published online in the Journal of Pain, were mostly observational, cross-sectional studies and one prospective investigation, published between 2009 and 2019.
The issue of pain is particularly important among people with hemophilia, as many adult patients suffer from distinct degrees of arthropathy and associated chronic pain, due to the lifelong occurrence of hemarthrosis, the authors noted. In an important distinction, according to the authors, people with hemophilia may therefore experience both acute pain during bleeds and chronic pain caused by joint deterioration. Acute pain ceases with the resolution of the bleeding episode, but the chronic pain is significantly more challenging, since it persists in time and may trigger changes in the nervous system, leading to peripheral or central sensitization.
Data in the assessed studies were collected from a variety of sources: hemophilia centers, online surveys, by mail, or through a national database, with return rates ranging from 29.2% to 98%. Overall, these studies comprised 4,772 adults, with individual sample sizes ranging from 21 to 2,253 patients, the authors added.
Conflicting results
Overall, there was a widely varying prevalence of hemophilia-related chronic pain reported across studies. Additionally, methodologies and sample characteristics varied widely. The meta-analyses revealed high heterogeneity between studies, and, therefore, pooled prevalence estimates values must be interpreted with caution, the authors stated.
All of the 11 selected studies included for meta-analysis and review reported on the prevalence of chronic pain caused by hemophilia. Chronic pain was assessed using direct questions developed by the authors in eight studies and using the European Haemophilia Therapy Standardization Board definition in three studies. The prevalence for global samples ranged widely from 17% to 84%.
Although there was high heterogeneity, the random-effects meta-analysis including all studies demonstrated a pooled prevalence of 46% of patients reporting chronic pain. Subgroup analyses of studies including all disease severities (mild, moderate, and severe; seven studies) revealed a pooled prevalence of 48%, but also with high heterogeneity. Looking at severe patients only (six studies), the chronic pain prevalence ranged from 33% to 86.4%, with a pooled prevalence of 53% and high heterogeneity, the authors added.
The wide disparity of the chronic pain prevalence seen across the studies is likely because of the fact that some investigations inquired about pain without distinguishing between acute (hemarthrosis-related) or chronic (arthropathy-related) pain, and without clarifying if the only focus is pain caused by hemophilia, or including all causes of pain complaints, according to the researchers.
“Concerning hemophilia-related chronic pain interference, it is striking that the existing literature does not distinguish between the impact of acute or chronic pain. Such a distinction is needed and should be made in future studies to ensure accurate accounts of hemophilia-related pain and to fully understand its interference according to the type of pain (acute vs. chronic). This information is relevant to promote targeted and effective treatment approaches,” the researchers concluded.
The research was supported by a Novo Nordisk HERO Research Grant 2015, the Portuguese Foundation for Science and Technology, and the Foundation for Science and Technology in Portugal. The authors declared they had no conflicts of interest.
Chronic pain is a common condition among people with hemophilia and is associated with joint deterioration because of repeated joint bleeds. This systematic review and meta-analysis aimed to determine the prevalence of chronic pain because of hemophilia and to analyze its interference in the lives of patients, according to Ana Cristina Paredes, a PhD student at the University of Minho, Braga, Portugal, and colleagues.
The manuscripts included in the study, which was published online in the Journal of Pain, were mostly observational, cross-sectional studies and one prospective investigation, published between 2009 and 2019.
The issue of pain is particularly important among people with hemophilia, as many adult patients suffer from distinct degrees of arthropathy and associated chronic pain, due to the lifelong occurrence of hemarthrosis, the authors noted. In an important distinction, according to the authors, people with hemophilia may therefore experience both acute pain during bleeds and chronic pain caused by joint deterioration. Acute pain ceases with the resolution of the bleeding episode, but the chronic pain is significantly more challenging, since it persists in time and may trigger changes in the nervous system, leading to peripheral or central sensitization.
Data in the assessed studies were collected from a variety of sources: hemophilia centers, online surveys, by mail, or through a national database, with return rates ranging from 29.2% to 98%. Overall, these studies comprised 4,772 adults, with individual sample sizes ranging from 21 to 2,253 patients, the authors added.
Conflicting results
Overall, there was a widely varying prevalence of hemophilia-related chronic pain reported across studies. Additionally, methodologies and sample characteristics varied widely. The meta-analyses revealed high heterogeneity between studies, and, therefore, pooled prevalence estimates values must be interpreted with caution, the authors stated.
All of the 11 selected studies included for meta-analysis and review reported on the prevalence of chronic pain caused by hemophilia. Chronic pain was assessed using direct questions developed by the authors in eight studies and using the European Haemophilia Therapy Standardization Board definition in three studies. The prevalence for global samples ranged widely from 17% to 84%.
Although there was high heterogeneity, the random-effects meta-analysis including all studies demonstrated a pooled prevalence of 46% of patients reporting chronic pain. Subgroup analyses of studies including all disease severities (mild, moderate, and severe; seven studies) revealed a pooled prevalence of 48%, but also with high heterogeneity. Looking at severe patients only (six studies), the chronic pain prevalence ranged from 33% to 86.4%, with a pooled prevalence of 53% and high heterogeneity, the authors added.
The wide disparity of the chronic pain prevalence seen across the studies is likely because of the fact that some investigations inquired about pain without distinguishing between acute (hemarthrosis-related) or chronic (arthropathy-related) pain, and without clarifying if the only focus is pain caused by hemophilia, or including all causes of pain complaints, according to the researchers.
“Concerning hemophilia-related chronic pain interference, it is striking that the existing literature does not distinguish between the impact of acute or chronic pain. Such a distinction is needed and should be made in future studies to ensure accurate accounts of hemophilia-related pain and to fully understand its interference according to the type of pain (acute vs. chronic). This information is relevant to promote targeted and effective treatment approaches,” the researchers concluded.
The research was supported by a Novo Nordisk HERO Research Grant 2015, the Portuguese Foundation for Science and Technology, and the Foundation for Science and Technology in Portugal. The authors declared they had no conflicts of interest.
FROM THE JOURNAL OF PAIN
FDA OKs higher-dose naloxone nasal spray for opioid overdose
The Food and Drug Administration has approved a higher-dose naloxone hydrochloride nasal spray (Kloxxado) for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression.
Kloxxado delivers 8 mg of naloxone into the nasal cavity, which is twice as much as the 4 mg of naloxone contained in Narcan nasal spray.
When administered quickly, naloxone can counter opioid overdose effects, usually within minutes. A higher dose of naloxone provides an additional option for the treatment of opioid overdoses, the FDA said in a news release.
“This approval meets another critical need in combating opioid overdose,” Patrizia Cavazzoni, MD, director, FDA Center for Drug Evaluation and Research, said in the release.
“Addressing the opioid crisis is a top priority for the FDA, and we will continue our efforts to increase access to naloxone and place this important medicine in the hands of those who need it most,” said Dr. Cavazzoni.
In a company news release announcing the approval, manufacturer Hikma Pharmaceuticals noted that a recent survey of community organizations in which the 4-mg naloxone nasal spray had been distributed showed that for 34% of attempted reversals, two or more doses of naloxone were used.
A separate study found that the percentage of overdose-related emergency medical service calls in the United States that led to the administration of multiple doses of naloxone increased to 21% during the period of 2013-2016, which represents a 43% increase over 4 years.
“The approval of Kloxxado is an important step in providing patients, friends, and family members – as well as the public health community – with an important new option for treating opioid overdose,” Brian Hoffmann, president of Hikma Generics, said in the release.
The FDA approved Kloxxado through the 505(b)(2) regulatory pathway, which allows the agency to refer to previous findings of safety and efficacy for an already-approved product, as well as to review findings from further studies of the product.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved a higher-dose naloxone hydrochloride nasal spray (Kloxxado) for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression.
Kloxxado delivers 8 mg of naloxone into the nasal cavity, which is twice as much as the 4 mg of naloxone contained in Narcan nasal spray.
When administered quickly, naloxone can counter opioid overdose effects, usually within minutes. A higher dose of naloxone provides an additional option for the treatment of opioid overdoses, the FDA said in a news release.
“This approval meets another critical need in combating opioid overdose,” Patrizia Cavazzoni, MD, director, FDA Center for Drug Evaluation and Research, said in the release.
“Addressing the opioid crisis is a top priority for the FDA, and we will continue our efforts to increase access to naloxone and place this important medicine in the hands of those who need it most,” said Dr. Cavazzoni.
In a company news release announcing the approval, manufacturer Hikma Pharmaceuticals noted that a recent survey of community organizations in which the 4-mg naloxone nasal spray had been distributed showed that for 34% of attempted reversals, two or more doses of naloxone were used.
A separate study found that the percentage of overdose-related emergency medical service calls in the United States that led to the administration of multiple doses of naloxone increased to 21% during the period of 2013-2016, which represents a 43% increase over 4 years.
“The approval of Kloxxado is an important step in providing patients, friends, and family members – as well as the public health community – with an important new option for treating opioid overdose,” Brian Hoffmann, president of Hikma Generics, said in the release.
The FDA approved Kloxxado through the 505(b)(2) regulatory pathway, which allows the agency to refer to previous findings of safety and efficacy for an already-approved product, as well as to review findings from further studies of the product.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved a higher-dose naloxone hydrochloride nasal spray (Kloxxado) for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression.
Kloxxado delivers 8 mg of naloxone into the nasal cavity, which is twice as much as the 4 mg of naloxone contained in Narcan nasal spray.
When administered quickly, naloxone can counter opioid overdose effects, usually within minutes. A higher dose of naloxone provides an additional option for the treatment of opioid overdoses, the FDA said in a news release.
“This approval meets another critical need in combating opioid overdose,” Patrizia Cavazzoni, MD, director, FDA Center for Drug Evaluation and Research, said in the release.
“Addressing the opioid crisis is a top priority for the FDA, and we will continue our efforts to increase access to naloxone and place this important medicine in the hands of those who need it most,” said Dr. Cavazzoni.
In a company news release announcing the approval, manufacturer Hikma Pharmaceuticals noted that a recent survey of community organizations in which the 4-mg naloxone nasal spray had been distributed showed that for 34% of attempted reversals, two or more doses of naloxone were used.
A separate study found that the percentage of overdose-related emergency medical service calls in the United States that led to the administration of multiple doses of naloxone increased to 21% during the period of 2013-2016, which represents a 43% increase over 4 years.
“The approval of Kloxxado is an important step in providing patients, friends, and family members – as well as the public health community – with an important new option for treating opioid overdose,” Brian Hoffmann, president of Hikma Generics, said in the release.
The FDA approved Kloxxado through the 505(b)(2) regulatory pathway, which allows the agency to refer to previous findings of safety and efficacy for an already-approved product, as well as to review findings from further studies of the product.
A version of this article first appeared on Medscape.com.