User login
Bariatric Surgery + Medical Therapy: Effective Tx for T2DM?

A 46-year-old woman presents with a BMI of 28, a 4-year history of type 2 diabetes mellitus (T2DM), and an A1C of 9.8%. The patient is currently being treated with intensive medical therapy (IMT), including metformin 2000 mg/d, sitagliptin 100 mg/d, and insulin glargine 12 U/d, with minimal change in A1C. Should you recommend bariatric surgery?
One in 11 Americans has diabetes, and at least 95% of those have T2DM.2,3 The treatment of T2DM is generally multimodal to target the various mechanisms that cause hyperglycemia. Strategies may include making lifestyle modifications, decreasing insulin resistance, increasing insulin secretion, replacing insulin, and targeting incretin-hormonal pathways.
The American Diabetes Association (ADA) recommends diet, exercise, and behavioral modifications as firstline therapy for diabetes management, but these methods are often inadequate.2 In addition to various pharmacotherapeutic strategies for some populations with T2DM, the ADA recommends bariatric surgery for those with a BMI ≥ 35 and uncontrolled hyperglycemia.2,4
However, this recommendation is based only on short-term studies. For example, in a single-center, nonblinded RCT of 60 patients with a BMI ≥ 35, the average baseline A1C levels of 8.65 ± 1.45% were reduced to 7.7 ± 0.6% in the IMT group and to 6.4 ± 1.4% in the gastric-bypass group at 2 years.5 In another study, a randomized double-blind trial involving 60 moderately obese patients (BMI, 25-35), gastric bypass yielded better outcomes than sleeve gastrectomy: 93% of patients in the former group and 47% of those in the latter group achieved remission of T2DM over a 12-month period.6
The current study by Schauer et al examined the long-term outcomes of IMT alone vs bariatric surgery with IMT for the treatment of T2DM in patients who are overweight or obese.1
STUDY SUMMARY
5-year follow-up: surgery + IMT works
This study was a 5-year follow-up of a nonblinded, single-center RCT comparing IMT alone to IMT with Roux-en-Y gastric bypass or sleeve gastrectomy in 150 patients with T2DM.1 Patients were included if they were ages 20 to 60, had a BMI of 27 to 43, and had an A1C > 7%. Patients with a history of bariatric surgery, complex abdominal surgery, or uncontrolled medical or psychiatric disorders were excluded.
Patients were randomly placed in a 1:1:1 fashion into 3 groups: IMT (as defined by the ADA) only, IMT and gastric bypass, or IMT and sleeve gastrectomy. The primary outcome was the number of patients with an A1C ≤ 6%. Secondary outcomes included weight loss, glucose control, lipid levels, blood pressure, medication use, renal function, adverse effects, ophthalmologic outcomes, and quality of life.
Continue to: Of the 150 patients...
Of the 150 patients, 1 died during the follow-up period, leaving 149. Of these, 134 completed the 5-year follow-up. Eight patients in the IMT group and 1 patient in the sleeve gastrectomy group never initiated assigned treatment, and 6 patients were lost to follow-up. One patient from the IMT group and 1 patient from the sleeve gastrectomy group crossed over to the gastric bypass group.
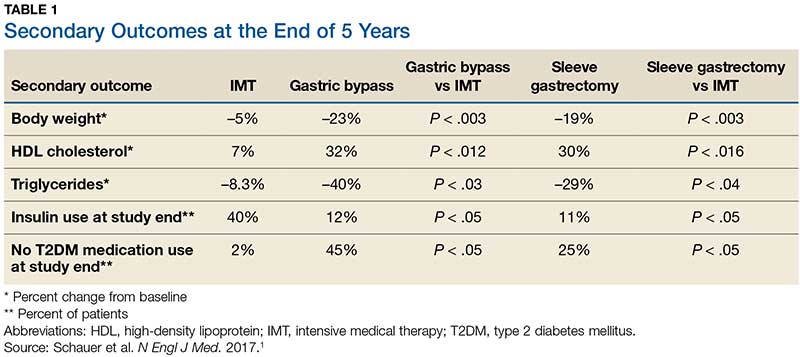
Results. More patients in the bariatric surgery and sleeve gastrectomy groups achieved an A1C of ≤ 6% than in the IMT group (14 of 49 gastric bypass patients, 11 of 47 sleeve gastrectomy patients, and 2 of 38 IMT patients). Compared with those in the IMT group, the patients in the 2 surgery groups showed greater reductions from baseline in body weight and triglyceride levels and greater increases from baseline in HDL cholesterol levels; they also required less antidiabetes medication for glycemic control (see Table).1
WHAT’S NEW?
Big benefits, minimal adverse effects
Prior studies evaluating the effect of gastric bypass surgery on diabetes were observational or had a shorter follow-up duration. This study demonstrates that bariatric surgery plus IMT has long-term benefits with minimal adverse events, compared with IMT alone.1,5 Additionally, this study supports recommendations for bariatric surgery as treatment for T2DM in patients with a BMI ≥ 27, which is below the starting BMI (35) recommended by the ADA.1,4
CAVEATS
Surgery is not without risks
The risk for surgical complications—eg, gastrointestinal bleeding, severe hypoglycemia requiring intervention, and ketoacidosis—in this patient population is significant.1 Other potential complications include gastrointestinal leak, stroke, and infection.1 Additionally, long-term complications from bariatric surgery are emerging and include choledocholithiasis, intestinal obstruction, and esophageal pathology.7 Extensive patient counseling is necessary to ensure that patients make an informed decision regarding surgery.
This study utilized surrogate markers (A1C, lipid levels, and body weight) as disease-oriented outcome measures. Patient-oriented outcomes, such as morbidity and mortality, were not explored in this study.
Continue to: Due to the small sample size...
Due to the small sample size of the study, it is unclear if the outcomes of the 2 surgery groups were significantly different. Patients who underwent gastric bypass surgery had more weight loss and used less diabetes medication at the end of follow-up, compared with patients who underwent sleeve gastrectomy. More information is needed to determine which gastric surgery is preferable for the treatment of T2DM while minimizing adverse effects. However, both of the procedures had outcomes superior to those of IMT, and selection of a particular type of surgery should be a joint decision between the patient and provider.
CHALLENGES TO IMPLEMENTATION
Access and cost may be barriers
The major barriers to implementation are access to, and cost of, bariatric surgery.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
Copyright © 2019. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice (2019;68[2]:102-104).
1. Schauer PR, Bhatt DL, Kirwan JP, et al; STAMPEDE Investigators. Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med. 2017;376:641-651.
2. American Diabetes Association. Obesity management for the treatment of type 2 diabetes: standards of medical care in diabetes—2019. Diabetes Care. 2019;42(suppl 1):S81-S89.
3. CDC. National Diabetes Statistics Report, 2017. Atlanta, GA: CDC, US Department of Health and Human Services; 2017. www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed June 27, 2019.
4. Rubino F, Nathan DM, Eckel RH, et al. Metabolic surgery in the treatment algorithm for type 2 diabetes: a joint statement by international diabetes organizations. Diabetes Care. 2016;39:861-877.
5. Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366:1577-1585.
6. Lee WJ, Chong K, Ser KH, et al. Gastric bypass vs sleeve gastrectomy for type 2 diabetes mellitus: a randomized controlled trial. Arch Surg. 2011; 146:143-148.
7. Schulman AR, Thompson CC. Complications of bariatric surgery: what you can expect to see in your GI practice. Am J Gastroenterol. 2017;112:1640-1655.

A 46-year-old woman presents with a BMI of 28, a 4-year history of type 2 diabetes mellitus (T2DM), and an A1C of 9.8%. The patient is currently being treated with intensive medical therapy (IMT), including metformin 2000 mg/d, sitagliptin 100 mg/d, and insulin glargine 12 U/d, with minimal change in A1C. Should you recommend bariatric surgery?
One in 11 Americans has diabetes, and at least 95% of those have T2DM.2,3 The treatment of T2DM is generally multimodal to target the various mechanisms that cause hyperglycemia. Strategies may include making lifestyle modifications, decreasing insulin resistance, increasing insulin secretion, replacing insulin, and targeting incretin-hormonal pathways.
The American Diabetes Association (ADA) recommends diet, exercise, and behavioral modifications as firstline therapy for diabetes management, but these methods are often inadequate.2 In addition to various pharmacotherapeutic strategies for some populations with T2DM, the ADA recommends bariatric surgery for those with a BMI ≥ 35 and uncontrolled hyperglycemia.2,4
However, this recommendation is based only on short-term studies. For example, in a single-center, nonblinded RCT of 60 patients with a BMI ≥ 35, the average baseline A1C levels of 8.65 ± 1.45% were reduced to 7.7 ± 0.6% in the IMT group and to 6.4 ± 1.4% in the gastric-bypass group at 2 years.5 In another study, a randomized double-blind trial involving 60 moderately obese patients (BMI, 25-35), gastric bypass yielded better outcomes than sleeve gastrectomy: 93% of patients in the former group and 47% of those in the latter group achieved remission of T2DM over a 12-month period.6
The current study by Schauer et al examined the long-term outcomes of IMT alone vs bariatric surgery with IMT for the treatment of T2DM in patients who are overweight or obese.1
STUDY SUMMARY
5-year follow-up: surgery + IMT works
This study was a 5-year follow-up of a nonblinded, single-center RCT comparing IMT alone to IMT with Roux-en-Y gastric bypass or sleeve gastrectomy in 150 patients with T2DM.1 Patients were included if they were ages 20 to 60, had a BMI of 27 to 43, and had an A1C > 7%. Patients with a history of bariatric surgery, complex abdominal surgery, or uncontrolled medical or psychiatric disorders were excluded.
Patients were randomly placed in a 1:1:1 fashion into 3 groups: IMT (as defined by the ADA) only, IMT and gastric bypass, or IMT and sleeve gastrectomy. The primary outcome was the number of patients with an A1C ≤ 6%. Secondary outcomes included weight loss, glucose control, lipid levels, blood pressure, medication use, renal function, adverse effects, ophthalmologic outcomes, and quality of life.
Continue to: Of the 150 patients...
Of the 150 patients, 1 died during the follow-up period, leaving 149. Of these, 134 completed the 5-year follow-up. Eight patients in the IMT group and 1 patient in the sleeve gastrectomy group never initiated assigned treatment, and 6 patients were lost to follow-up. One patient from the IMT group and 1 patient from the sleeve gastrectomy group crossed over to the gastric bypass group.
Results. More patients in the bariatric surgery and sleeve gastrectomy groups achieved an A1C of ≤ 6% than in the IMT group (14 of 49 gastric bypass patients, 11 of 47 sleeve gastrectomy patients, and 2 of 38 IMT patients). Compared with those in the IMT group, the patients in the 2 surgery groups showed greater reductions from baseline in body weight and triglyceride levels and greater increases from baseline in HDL cholesterol levels; they also required less antidiabetes medication for glycemic control (see Table).1

WHAT’S NEW?
Big benefits, minimal adverse effects
Prior studies evaluating the effect of gastric bypass surgery on diabetes were observational or had a shorter follow-up duration. This study demonstrates that bariatric surgery plus IMT has long-term benefits with minimal adverse events, compared with IMT alone.1,5 Additionally, this study supports recommendations for bariatric surgery as treatment for T2DM in patients with a BMI ≥ 27, which is below the starting BMI (35) recommended by the ADA.1,4
CAVEATS
Surgery is not without risks
The risk for surgical complications—eg, gastrointestinal bleeding, severe hypoglycemia requiring intervention, and ketoacidosis—in this patient population is significant.1 Other potential complications include gastrointestinal leak, stroke, and infection.1 Additionally, long-term complications from bariatric surgery are emerging and include choledocholithiasis, intestinal obstruction, and esophageal pathology.7 Extensive patient counseling is necessary to ensure that patients make an informed decision regarding surgery.
This study utilized surrogate markers (A1C, lipid levels, and body weight) as disease-oriented outcome measures. Patient-oriented outcomes, such as morbidity and mortality, were not explored in this study.
Continue to: Due to the small sample size...
Due to the small sample size of the study, it is unclear if the outcomes of the 2 surgery groups were significantly different. Patients who underwent gastric bypass surgery had more weight loss and used less diabetes medication at the end of follow-up, compared with patients who underwent sleeve gastrectomy. More information is needed to determine which gastric surgery is preferable for the treatment of T2DM while minimizing adverse effects. However, both of the procedures had outcomes superior to those of IMT, and selection of a particular type of surgery should be a joint decision between the patient and provider.
CHALLENGES TO IMPLEMENTATION
Access and cost may be barriers
The major barriers to implementation are access to, and cost of, bariatric surgery.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
Copyright © 2019. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice (2019;68[2]:102-104).

A 46-year-old woman presents with a BMI of 28, a 4-year history of type 2 diabetes mellitus (T2DM), and an A1C of 9.8%. The patient is currently being treated with intensive medical therapy (IMT), including metformin 2000 mg/d, sitagliptin 100 mg/d, and insulin glargine 12 U/d, with minimal change in A1C. Should you recommend bariatric surgery?
One in 11 Americans has diabetes, and at least 95% of those have T2DM.2,3 The treatment of T2DM is generally multimodal to target the various mechanisms that cause hyperglycemia. Strategies may include making lifestyle modifications, decreasing insulin resistance, increasing insulin secretion, replacing insulin, and targeting incretin-hormonal pathways.
The American Diabetes Association (ADA) recommends diet, exercise, and behavioral modifications as firstline therapy for diabetes management, but these methods are often inadequate.2 In addition to various pharmacotherapeutic strategies for some populations with T2DM, the ADA recommends bariatric surgery for those with a BMI ≥ 35 and uncontrolled hyperglycemia.2,4
However, this recommendation is based only on short-term studies. For example, in a single-center, nonblinded RCT of 60 patients with a BMI ≥ 35, the average baseline A1C levels of 8.65 ± 1.45% were reduced to 7.7 ± 0.6% in the IMT group and to 6.4 ± 1.4% in the gastric-bypass group at 2 years.5 In another study, a randomized double-blind trial involving 60 moderately obese patients (BMI, 25-35), gastric bypass yielded better outcomes than sleeve gastrectomy: 93% of patients in the former group and 47% of those in the latter group achieved remission of T2DM over a 12-month period.6
The current study by Schauer et al examined the long-term outcomes of IMT alone vs bariatric surgery with IMT for the treatment of T2DM in patients who are overweight or obese.1
STUDY SUMMARY
5-year follow-up: surgery + IMT works
This study was a 5-year follow-up of a nonblinded, single-center RCT comparing IMT alone to IMT with Roux-en-Y gastric bypass or sleeve gastrectomy in 150 patients with T2DM.1 Patients were included if they were ages 20 to 60, had a BMI of 27 to 43, and had an A1C > 7%. Patients with a history of bariatric surgery, complex abdominal surgery, or uncontrolled medical or psychiatric disorders were excluded.
Patients were randomly placed in a 1:1:1 fashion into 3 groups: IMT (as defined by the ADA) only, IMT and gastric bypass, or IMT and sleeve gastrectomy. The primary outcome was the number of patients with an A1C ≤ 6%. Secondary outcomes included weight loss, glucose control, lipid levels, blood pressure, medication use, renal function, adverse effects, ophthalmologic outcomes, and quality of life.
Continue to: Of the 150 patients...
Of the 150 patients, 1 died during the follow-up period, leaving 149. Of these, 134 completed the 5-year follow-up. Eight patients in the IMT group and 1 patient in the sleeve gastrectomy group never initiated assigned treatment, and 6 patients were lost to follow-up. One patient from the IMT group and 1 patient from the sleeve gastrectomy group crossed over to the gastric bypass group.
Results. More patients in the bariatric surgery and sleeve gastrectomy groups achieved an A1C of ≤ 6% than in the IMT group (14 of 49 gastric bypass patients, 11 of 47 sleeve gastrectomy patients, and 2 of 38 IMT patients). Compared with those in the IMT group, the patients in the 2 surgery groups showed greater reductions from baseline in body weight and triglyceride levels and greater increases from baseline in HDL cholesterol levels; they also required less antidiabetes medication for glycemic control (see Table).1

WHAT’S NEW?
Big benefits, minimal adverse effects
Prior studies evaluating the effect of gastric bypass surgery on diabetes were observational or had a shorter follow-up duration. This study demonstrates that bariatric surgery plus IMT has long-term benefits with minimal adverse events, compared with IMT alone.1,5 Additionally, this study supports recommendations for bariatric surgery as treatment for T2DM in patients with a BMI ≥ 27, which is below the starting BMI (35) recommended by the ADA.1,4
CAVEATS
Surgery is not without risks
The risk for surgical complications—eg, gastrointestinal bleeding, severe hypoglycemia requiring intervention, and ketoacidosis—in this patient population is significant.1 Other potential complications include gastrointestinal leak, stroke, and infection.1 Additionally, long-term complications from bariatric surgery are emerging and include choledocholithiasis, intestinal obstruction, and esophageal pathology.7 Extensive patient counseling is necessary to ensure that patients make an informed decision regarding surgery.
This study utilized surrogate markers (A1C, lipid levels, and body weight) as disease-oriented outcome measures. Patient-oriented outcomes, such as morbidity and mortality, were not explored in this study.
Continue to: Due to the small sample size...
Due to the small sample size of the study, it is unclear if the outcomes of the 2 surgery groups were significantly different. Patients who underwent gastric bypass surgery had more weight loss and used less diabetes medication at the end of follow-up, compared with patients who underwent sleeve gastrectomy. More information is needed to determine which gastric surgery is preferable for the treatment of T2DM while minimizing adverse effects. However, both of the procedures had outcomes superior to those of IMT, and selection of a particular type of surgery should be a joint decision between the patient and provider.
CHALLENGES TO IMPLEMENTATION
Access and cost may be barriers
The major barriers to implementation are access to, and cost of, bariatric surgery.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
Copyright © 2019. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice (2019;68[2]:102-104).
1. Schauer PR, Bhatt DL, Kirwan JP, et al; STAMPEDE Investigators. Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med. 2017;376:641-651.
2. American Diabetes Association. Obesity management for the treatment of type 2 diabetes: standards of medical care in diabetes—2019. Diabetes Care. 2019;42(suppl 1):S81-S89.
3. CDC. National Diabetes Statistics Report, 2017. Atlanta, GA: CDC, US Department of Health and Human Services; 2017. www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed June 27, 2019.
4. Rubino F, Nathan DM, Eckel RH, et al. Metabolic surgery in the treatment algorithm for type 2 diabetes: a joint statement by international diabetes organizations. Diabetes Care. 2016;39:861-877.
5. Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366:1577-1585.
6. Lee WJ, Chong K, Ser KH, et al. Gastric bypass vs sleeve gastrectomy for type 2 diabetes mellitus: a randomized controlled trial. Arch Surg. 2011; 146:143-148.
7. Schulman AR, Thompson CC. Complications of bariatric surgery: what you can expect to see in your GI practice. Am J Gastroenterol. 2017;112:1640-1655.
1. Schauer PR, Bhatt DL, Kirwan JP, et al; STAMPEDE Investigators. Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med. 2017;376:641-651.
2. American Diabetes Association. Obesity management for the treatment of type 2 diabetes: standards of medical care in diabetes—2019. Diabetes Care. 2019;42(suppl 1):S81-S89.
3. CDC. National Diabetes Statistics Report, 2017. Atlanta, GA: CDC, US Department of Health and Human Services; 2017. www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed June 27, 2019.
4. Rubino F, Nathan DM, Eckel RH, et al. Metabolic surgery in the treatment algorithm for type 2 diabetes: a joint statement by international diabetes organizations. Diabetes Care. 2016;39:861-877.
5. Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366:1577-1585.
6. Lee WJ, Chong K, Ser KH, et al. Gastric bypass vs sleeve gastrectomy for type 2 diabetes mellitus: a randomized controlled trial. Arch Surg. 2011; 146:143-148.
7. Schulman AR, Thompson CC. Complications of bariatric surgery: what you can expect to see in your GI practice. Am J Gastroenterol. 2017;112:1640-1655.
Intervention tied to fewer depressive symptoms, more weight loss
Adults with obesity and depression who participated in a program that addressed weight and mood saw improvement in weight loss and depressive symptoms at 12 months, results of a randomized, controlled trial of almost 350 patients show.
“To our knowledge, this study was the first and largest RTC of integrated collaborative care for coexisting obesity and depression,” wrote Jun Ma, MD, PhD, of the Institute of Health Research and Policy at the University of Illinois at Chicago, and colleagues.
Dr. Ma and colleagues enrolled 409 patients in the RAINBOW (Research Aimed at Improving Both Mood and Weight) trial between September 2014 and January 2017 from family and internal medicine departments at four medical centers in California. The RAINBOW intervention combined usual care with a weight loss treatment program used in diabetes prevention, problem-solving therapy, and prescriptions for antidepressants if indicated. About 71% of the trial participants were non-Hispanic white adults, 70% were women, and 69% had a college education.
Half the patients were randomized to receive usual care consisting of seeing personal physicians, receiving information on obesity and depression services at the clinic, and wireless activity-tracking devices. Patients were enrolled in the trial if they scored at least 10 points in the nine-item Patient Health Questionaire (PHQ-9) and had a body mass index (BMI) of 30 or higher, or a BMI of 27 or higher in Asian adults. The mean age in the cohort was 51.0 years, the mean BMI was 37.7, and the mean PHQ-9 score was 13.8.
Of the 344 patients (84.1%) who completed follow-up at 12 months, there was a decrease in mean BMI from 36.7 to 35.9 for patients who received the collaborative care intervention, compared with no change in BMI for patients who received usual care alone (between-group mean difference, −0.7; 95% confidence interval, −1.1 to −0.2; P = .01). Depressive symptoms also improved in the intervention group, with mean 20-item Depression Symptom Checklist scores decreasing from 1.5 at baseline to 1.1 at 12 months, compared with a decrease from 1.5 at baseline to 1.4 at 12 months in the usual-care group (between-group mean difference, −0.2; 95% CI, −0.4 to 0; P = .01). Overall, there were 47 adverse events or serious adverse events, with 27 events in the collaborative-care intervention group and 20 events in the usual-care group involving musculoskeletal injuries such as fracture and meniscus tear.
In addition, they cited the relative demographic homogeneity of the study sample as one of several limitations.
The study was funded in part by Palo Alto Medical Foundation Research Institute, the University of Illinois at Chicago, and an award from the National Heart, Lung, and Blood Institute. One author, Philip W. Lavori, PhD, reported receiving personal fees from Palo Alto Medical Foundation Research Institute. The other authors reported no relevant financial disclosures.
SOURCE: Ma J et al. JAMA. 2019. doi: 10.1001/jama2019.0557.
Adults with obesity and depression who participated in a program that addressed weight and mood saw improvement in weight loss and depressive symptoms at 12 months, results of a randomized, controlled trial of almost 350 patients show.
“To our knowledge, this study was the first and largest RTC of integrated collaborative care for coexisting obesity and depression,” wrote Jun Ma, MD, PhD, of the Institute of Health Research and Policy at the University of Illinois at Chicago, and colleagues.
Dr. Ma and colleagues enrolled 409 patients in the RAINBOW (Research Aimed at Improving Both Mood and Weight) trial between September 2014 and January 2017 from family and internal medicine departments at four medical centers in California. The RAINBOW intervention combined usual care with a weight loss treatment program used in diabetes prevention, problem-solving therapy, and prescriptions for antidepressants if indicated. About 71% of the trial participants were non-Hispanic white adults, 70% were women, and 69% had a college education.
Half the patients were randomized to receive usual care consisting of seeing personal physicians, receiving information on obesity and depression services at the clinic, and wireless activity-tracking devices. Patients were enrolled in the trial if they scored at least 10 points in the nine-item Patient Health Questionaire (PHQ-9) and had a body mass index (BMI) of 30 or higher, or a BMI of 27 or higher in Asian adults. The mean age in the cohort was 51.0 years, the mean BMI was 37.7, and the mean PHQ-9 score was 13.8.
Of the 344 patients (84.1%) who completed follow-up at 12 months, there was a decrease in mean BMI from 36.7 to 35.9 for patients who received the collaborative care intervention, compared with no change in BMI for patients who received usual care alone (between-group mean difference, −0.7; 95% confidence interval, −1.1 to −0.2; P = .01). Depressive symptoms also improved in the intervention group, with mean 20-item Depression Symptom Checklist scores decreasing from 1.5 at baseline to 1.1 at 12 months, compared with a decrease from 1.5 at baseline to 1.4 at 12 months in the usual-care group (between-group mean difference, −0.2; 95% CI, −0.4 to 0; P = .01). Overall, there were 47 adverse events or serious adverse events, with 27 events in the collaborative-care intervention group and 20 events in the usual-care group involving musculoskeletal injuries such as fracture and meniscus tear.
In addition, they cited the relative demographic homogeneity of the study sample as one of several limitations.
The study was funded in part by Palo Alto Medical Foundation Research Institute, the University of Illinois at Chicago, and an award from the National Heart, Lung, and Blood Institute. One author, Philip W. Lavori, PhD, reported receiving personal fees from Palo Alto Medical Foundation Research Institute. The other authors reported no relevant financial disclosures.
SOURCE: Ma J et al. JAMA. 2019. doi: 10.1001/jama2019.0557.
Adults with obesity and depression who participated in a program that addressed weight and mood saw improvement in weight loss and depressive symptoms at 12 months, results of a randomized, controlled trial of almost 350 patients show.
“To our knowledge, this study was the first and largest RTC of integrated collaborative care for coexisting obesity and depression,” wrote Jun Ma, MD, PhD, of the Institute of Health Research and Policy at the University of Illinois at Chicago, and colleagues.
Dr. Ma and colleagues enrolled 409 patients in the RAINBOW (Research Aimed at Improving Both Mood and Weight) trial between September 2014 and January 2017 from family and internal medicine departments at four medical centers in California. The RAINBOW intervention combined usual care with a weight loss treatment program used in diabetes prevention, problem-solving therapy, and prescriptions for antidepressants if indicated. About 71% of the trial participants were non-Hispanic white adults, 70% were women, and 69% had a college education.
Half the patients were randomized to receive usual care consisting of seeing personal physicians, receiving information on obesity and depression services at the clinic, and wireless activity-tracking devices. Patients were enrolled in the trial if they scored at least 10 points in the nine-item Patient Health Questionaire (PHQ-9) and had a body mass index (BMI) of 30 or higher, or a BMI of 27 or higher in Asian adults. The mean age in the cohort was 51.0 years, the mean BMI was 37.7, and the mean PHQ-9 score was 13.8.
Of the 344 patients (84.1%) who completed follow-up at 12 months, there was a decrease in mean BMI from 36.7 to 35.9 for patients who received the collaborative care intervention, compared with no change in BMI for patients who received usual care alone (between-group mean difference, −0.7; 95% confidence interval, −1.1 to −0.2; P = .01). Depressive symptoms also improved in the intervention group, with mean 20-item Depression Symptom Checklist scores decreasing from 1.5 at baseline to 1.1 at 12 months, compared with a decrease from 1.5 at baseline to 1.4 at 12 months in the usual-care group (between-group mean difference, −0.2; 95% CI, −0.4 to 0; P = .01). Overall, there were 47 adverse events or serious adverse events, with 27 events in the collaborative-care intervention group and 20 events in the usual-care group involving musculoskeletal injuries such as fracture and meniscus tear.
In addition, they cited the relative demographic homogeneity of the study sample as one of several limitations.
The study was funded in part by Palo Alto Medical Foundation Research Institute, the University of Illinois at Chicago, and an award from the National Heart, Lung, and Blood Institute. One author, Philip W. Lavori, PhD, reported receiving personal fees from Palo Alto Medical Foundation Research Institute. The other authors reported no relevant financial disclosures.
SOURCE: Ma J et al. JAMA. 2019. doi: 10.1001/jama2019.0557.
FROM JAMA
New findings cast more doubt on ‘fat-but-fit’ theory
SAN FRANCISCO – Can you be “fat but fit” if you’re obese but don’t suffer from metabolic syndrome? Some advocates have claimed you can, but new findings presented at the annual scientific sessions of the American Diabetes Association provide more evidence that those extra pounds translate to extra cardiac risk.
Fat-but-fit is a misnomer, Yvonne Commodore-Mensah, PhD, RN, assistant professor at Johns Hopkins School of Nursing, Baltimore, said in an interview. “The metabolically healthy obese are not so healthy. [We found] they had a higher risk of heart disease than people who were metabolically healthy and nonobese.”
Studies began supporting the fat-but-fit “paradox” in the late 1990s. They showed “that all-cause and CVD [cardiovascular] mortality risk in obese individuals, as defined by body mass index (BMI), body fat percentage, or waist circumference, who are fit (i.e., cardiorespiratory fitness level above the age-specific and sex-specific 20th percentile) is not significantly different from their normal-weight and fit counterparts” (Br J Sports Med. 2018;52[3]:151-3).
However, a 2017 study had found that “metabolically healthy obese individuals had a higher risk of coronary heart disease, cerebrovascular disease, and heart failure [compared with] normal weight, metabolically healthy individuals” (J Am Coll Cardiol. 2017;70[12]:1429-37). And a 2016 meta-analysis of 22 studies had produced similar results but also found that metabolically healthy obese individuals were better off, cardiac-health–wise, than those of normal weight who were metabolically unhealthy (Eur J Prev Cardiol. 2016;23[9]:956-66).
Dr. Commodore-Mensah and colleagues sought to establish through their study whether there was evidence of subclinical heart disease in people who are considered obese but metabolically healthy (Abstract 272-OR).
They tracked 11,884 participants in the Atherosclerosis Risk in Communities Study (ARIC) from 1990-1992 to 2016-2018. The study, which continues today, includes participants in suburban Minneapolis; Jackson, Miss.; Forsyth County, N.C.; and Washington County, Md.
None of the participants had previous cardiovascular disease at baseline (1990-1992). The researchers divided the participants into four groups at baseline: Nonobese (with metabolic syndrome, 20% of the total number of participants; or without metabolic syndrome, 51%) and obese (with metabolic syndrome, 20%; or without metabolic syndrome, 9%).
The average age range in the groups was 56-57 years. The percentage of women in the groups ranged from 53% to 58%, except for the obese and metabolically healthy group (73%). The percentage of black participants in the groups ranged from 17% (nonobese, metabolically unhealthy) to 45% (obese, metabolically healthy).
“People who were younger, women, and black were more likely to be classified as metabolically healthy obese,” Dr. Commodore-Mensah said.
According to one adjusted model with a median follow-up of 16 years and a total of 3,560 events, obese participants had a higher risk of incident cardiovascular disease, compared with their nonobese counterparts, regardless of whether they had metabolic syndrome.
When compared with the nonobese, metabolically healthy group, the risk grew in the nonobese, metabolically unhealthy group (hazard ratio, .24; 95% confidence interval, 1.12-1.36), as well as in the obese, metabolically healthy (HR, 1.33; 95% CI, 1.15-1.53) and the obese, metabolically unhealthy (HR, 2.11; 95% CI, 1.90-2.35) groups.
The researchers also focused on the cardiac biomarker known as high-sensitive cardiac troponin T (hs-cTnT), which indicates chronic myocardial damage. “This biomarker provides us with a window to the heart,” Dr. Commodore-Mensah said.
According to previous findings reported in 2014, ARIC participants who had hs-cTnT levels of 14 ng/L or higher were much more likely than were those with undetectable levels to suffer from heart failure, death from any cause, and coronary heart disease (JACC Heart Fail. 2014;2[6]:600-7).
Based on an analysis of the hs-cTnT levels in the present study, the researchers believe obese, metabolically healthy participants fell in the intermediate range of excess subclinical myocardial damage, between the nonobese and the obese participants who are also metabolically unhealthy.
“This group is not protected from heart disease,” Dr. Commodore-Mensah said. “They should be targeted, and they would benefit from behavioral changes, such as modifying their diet and increasing physical activity levels.”
The study is funded by the National Institutes of Health. Dr. Commodore-Mensah and six coauthors reported no relevant disclosures. Two coauthors reported various disclosures.
SAN FRANCISCO – Can you be “fat but fit” if you’re obese but don’t suffer from metabolic syndrome? Some advocates have claimed you can, but new findings presented at the annual scientific sessions of the American Diabetes Association provide more evidence that those extra pounds translate to extra cardiac risk.
Fat-but-fit is a misnomer, Yvonne Commodore-Mensah, PhD, RN, assistant professor at Johns Hopkins School of Nursing, Baltimore, said in an interview. “The metabolically healthy obese are not so healthy. [We found] they had a higher risk of heart disease than people who were metabolically healthy and nonobese.”
Studies began supporting the fat-but-fit “paradox” in the late 1990s. They showed “that all-cause and CVD [cardiovascular] mortality risk in obese individuals, as defined by body mass index (BMI), body fat percentage, or waist circumference, who are fit (i.e., cardiorespiratory fitness level above the age-specific and sex-specific 20th percentile) is not significantly different from their normal-weight and fit counterparts” (Br J Sports Med. 2018;52[3]:151-3).
However, a 2017 study had found that “metabolically healthy obese individuals had a higher risk of coronary heart disease, cerebrovascular disease, and heart failure [compared with] normal weight, metabolically healthy individuals” (J Am Coll Cardiol. 2017;70[12]:1429-37). And a 2016 meta-analysis of 22 studies had produced similar results but also found that metabolically healthy obese individuals were better off, cardiac-health–wise, than those of normal weight who were metabolically unhealthy (Eur J Prev Cardiol. 2016;23[9]:956-66).
Dr. Commodore-Mensah and colleagues sought to establish through their study whether there was evidence of subclinical heart disease in people who are considered obese but metabolically healthy (Abstract 272-OR).
They tracked 11,884 participants in the Atherosclerosis Risk in Communities Study (ARIC) from 1990-1992 to 2016-2018. The study, which continues today, includes participants in suburban Minneapolis; Jackson, Miss.; Forsyth County, N.C.; and Washington County, Md.
None of the participants had previous cardiovascular disease at baseline (1990-1992). The researchers divided the participants into four groups at baseline: Nonobese (with metabolic syndrome, 20% of the total number of participants; or without metabolic syndrome, 51%) and obese (with metabolic syndrome, 20%; or without metabolic syndrome, 9%).
The average age range in the groups was 56-57 years. The percentage of women in the groups ranged from 53% to 58%, except for the obese and metabolically healthy group (73%). The percentage of black participants in the groups ranged from 17% (nonobese, metabolically unhealthy) to 45% (obese, metabolically healthy).
“People who were younger, women, and black were more likely to be classified as metabolically healthy obese,” Dr. Commodore-Mensah said.
According to one adjusted model with a median follow-up of 16 years and a total of 3,560 events, obese participants had a higher risk of incident cardiovascular disease, compared with their nonobese counterparts, regardless of whether they had metabolic syndrome.
When compared with the nonobese, metabolically healthy group, the risk grew in the nonobese, metabolically unhealthy group (hazard ratio, .24; 95% confidence interval, 1.12-1.36), as well as in the obese, metabolically healthy (HR, 1.33; 95% CI, 1.15-1.53) and the obese, metabolically unhealthy (HR, 2.11; 95% CI, 1.90-2.35) groups.
The researchers also focused on the cardiac biomarker known as high-sensitive cardiac troponin T (hs-cTnT), which indicates chronic myocardial damage. “This biomarker provides us with a window to the heart,” Dr. Commodore-Mensah said.
According to previous findings reported in 2014, ARIC participants who had hs-cTnT levels of 14 ng/L or higher were much more likely than were those with undetectable levels to suffer from heart failure, death from any cause, and coronary heart disease (JACC Heart Fail. 2014;2[6]:600-7).
Based on an analysis of the hs-cTnT levels in the present study, the researchers believe obese, metabolically healthy participants fell in the intermediate range of excess subclinical myocardial damage, between the nonobese and the obese participants who are also metabolically unhealthy.
“This group is not protected from heart disease,” Dr. Commodore-Mensah said. “They should be targeted, and they would benefit from behavioral changes, such as modifying their diet and increasing physical activity levels.”
The study is funded by the National Institutes of Health. Dr. Commodore-Mensah and six coauthors reported no relevant disclosures. Two coauthors reported various disclosures.
SAN FRANCISCO – Can you be “fat but fit” if you’re obese but don’t suffer from metabolic syndrome? Some advocates have claimed you can, but new findings presented at the annual scientific sessions of the American Diabetes Association provide more evidence that those extra pounds translate to extra cardiac risk.
Fat-but-fit is a misnomer, Yvonne Commodore-Mensah, PhD, RN, assistant professor at Johns Hopkins School of Nursing, Baltimore, said in an interview. “The metabolically healthy obese are not so healthy. [We found] they had a higher risk of heart disease than people who were metabolically healthy and nonobese.”
Studies began supporting the fat-but-fit “paradox” in the late 1990s. They showed “that all-cause and CVD [cardiovascular] mortality risk in obese individuals, as defined by body mass index (BMI), body fat percentage, or waist circumference, who are fit (i.e., cardiorespiratory fitness level above the age-specific and sex-specific 20th percentile) is not significantly different from their normal-weight and fit counterparts” (Br J Sports Med. 2018;52[3]:151-3).
However, a 2017 study had found that “metabolically healthy obese individuals had a higher risk of coronary heart disease, cerebrovascular disease, and heart failure [compared with] normal weight, metabolically healthy individuals” (J Am Coll Cardiol. 2017;70[12]:1429-37). And a 2016 meta-analysis of 22 studies had produced similar results but also found that metabolically healthy obese individuals were better off, cardiac-health–wise, than those of normal weight who were metabolically unhealthy (Eur J Prev Cardiol. 2016;23[9]:956-66).
Dr. Commodore-Mensah and colleagues sought to establish through their study whether there was evidence of subclinical heart disease in people who are considered obese but metabolically healthy (Abstract 272-OR).
They tracked 11,884 participants in the Atherosclerosis Risk in Communities Study (ARIC) from 1990-1992 to 2016-2018. The study, which continues today, includes participants in suburban Minneapolis; Jackson, Miss.; Forsyth County, N.C.; and Washington County, Md.
None of the participants had previous cardiovascular disease at baseline (1990-1992). The researchers divided the participants into four groups at baseline: Nonobese (with metabolic syndrome, 20% of the total number of participants; or without metabolic syndrome, 51%) and obese (with metabolic syndrome, 20%; or without metabolic syndrome, 9%).
The average age range in the groups was 56-57 years. The percentage of women in the groups ranged from 53% to 58%, except for the obese and metabolically healthy group (73%). The percentage of black participants in the groups ranged from 17% (nonobese, metabolically unhealthy) to 45% (obese, metabolically healthy).
“People who were younger, women, and black were more likely to be classified as metabolically healthy obese,” Dr. Commodore-Mensah said.
According to one adjusted model with a median follow-up of 16 years and a total of 3,560 events, obese participants had a higher risk of incident cardiovascular disease, compared with their nonobese counterparts, regardless of whether they had metabolic syndrome.
When compared with the nonobese, metabolically healthy group, the risk grew in the nonobese, metabolically unhealthy group (hazard ratio, .24; 95% confidence interval, 1.12-1.36), as well as in the obese, metabolically healthy (HR, 1.33; 95% CI, 1.15-1.53) and the obese, metabolically unhealthy (HR, 2.11; 95% CI, 1.90-2.35) groups.
The researchers also focused on the cardiac biomarker known as high-sensitive cardiac troponin T (hs-cTnT), which indicates chronic myocardial damage. “This biomarker provides us with a window to the heart,” Dr. Commodore-Mensah said.
According to previous findings reported in 2014, ARIC participants who had hs-cTnT levels of 14 ng/L or higher were much more likely than were those with undetectable levels to suffer from heart failure, death from any cause, and coronary heart disease (JACC Heart Fail. 2014;2[6]:600-7).
Based on an analysis of the hs-cTnT levels in the present study, the researchers believe obese, metabolically healthy participants fell in the intermediate range of excess subclinical myocardial damage, between the nonobese and the obese participants who are also metabolically unhealthy.
“This group is not protected from heart disease,” Dr. Commodore-Mensah said. “They should be targeted, and they would benefit from behavioral changes, such as modifying their diet and increasing physical activity levels.”
The study is funded by the National Institutes of Health. Dr. Commodore-Mensah and six coauthors reported no relevant disclosures. Two coauthors reported various disclosures.
REPORTING FROM ADA 2019
Formal weight loss programs improve NAFLD
For patients with nonalcoholic fatty liver disease (NAFLD), formal weight loss programs lead to statistically and clinically significant improvements in biomarkers of liver disease, based on a recent meta-analysis.
The findings support changing NAFLD guidelines to recommend weight loss interventions, according to lead author Dimitrios A. Koutoukidis, PhD, of the University of Oxford, UK, and colleagues.
“Clinical guidelines around the world recommend physicians offer advice on lifestyle modification, which mostly includes weight loss through hypoenergetic diets and increased physical activity,” the investigators wrote in JAMA Internal Medicine.“However, whether clinicians provide advice and the type of advice they give vary greatly, and guidelines rarely specifically recommend treatment programs to support weight loss,” they added.
To investigate associations between methods of weight loss and improvements in NAFLD, the investigators screened for studies involving behavioral weight loss programs, pharmacotherapy, or bariatric surgery, alone or in combination. To limit confounding, studies combining weight loss with other potential treatments, such as medications, were excluded. Weight loss interventions were compared to liver disease outcomes associated with lower-intensity weight loss intervention or none or minimal weight loss support, using at least 1 reported biomarker of liver disease.
The literature search returned 22 eligible studies involving 2,588 patients. The investigators found that more intensive weight loss programs were associated with greater weight loss than lower intensity methods (-3.61 kg; I2 = 95%). Multiple biomarkers of liver disease showed significant improvements in association with formal weight loss programs, including histologically or radiologically measured liver steatosis (standardized mean difference: -1.48; I2 = 94%), histologic NAFLD activity score (-0.92; I2= 95%), presence of nonalcoholic steatohepatitis (OR, 0.14; I2 =0%), alanine aminotransferase (-9.81 U/L; I2= 97%), aspartate transaminase (-4.84 U/L; I2 = 96%), alkaline phosphatase (-5.53 U/L; I2 = 96%), and gamma-glutamyl transferase (-4.35 U/L; I2 = 92%). Weight loss interventions were not significantly associated with histologic liver fibrosis or inflammation, the investigators noted.
“The advantages [of weight loss interventions] seem to be greater in people who are overweight and with NAFLD, but our exploratory results suggest that weight loss interventions might still be beneficial in the minority of people with healthy weight and NAFLD,” the investigators wrote. “Clinicians may use these findings to counsel people with NAFLD on the expected clinically significant improvements in liver biomarkers after weight loss and direct the patients toward valuable interventions.”
“The accumulated evidence supports changing the clinical guidelines and routine practice to recommend formal weight loss programs to treat people with NAFLD,” the investigators concluded.
The study was funded by the National Institute for Health Research (NIHR) Oxford Biomedical Research Centre and the Oxford NIHR Collaboration and Leadership in Applied Health Research. The investigators reported grants for other research from Cambridge Weight Plan.
SOURCE: Koutoukidis et al. JAMA Int Med. 2019 Jul 1. doi: 10.1001/jamainternmed.2019.2248.
The AGA Practice guide on Obesity and Weight management, Education and Resources (POWER) paper provides physicians with a comprehensive, multi-disciplinary process to guide and personalize innovative obesity care for safe and effective weight management. Learn more at https://www.gastro.org/practice-guidance/practice-updates/obesity-practice-guide
For patients with nonalcoholic fatty liver disease (NAFLD), formal weight loss programs lead to statistically and clinically significant improvements in biomarkers of liver disease, based on a recent meta-analysis.
The findings support changing NAFLD guidelines to recommend weight loss interventions, according to lead author Dimitrios A. Koutoukidis, PhD, of the University of Oxford, UK, and colleagues.
“Clinical guidelines around the world recommend physicians offer advice on lifestyle modification, which mostly includes weight loss through hypoenergetic diets and increased physical activity,” the investigators wrote in JAMA Internal Medicine.“However, whether clinicians provide advice and the type of advice they give vary greatly, and guidelines rarely specifically recommend treatment programs to support weight loss,” they added.
To investigate associations between methods of weight loss and improvements in NAFLD, the investigators screened for studies involving behavioral weight loss programs, pharmacotherapy, or bariatric surgery, alone or in combination. To limit confounding, studies combining weight loss with other potential treatments, such as medications, were excluded. Weight loss interventions were compared to liver disease outcomes associated with lower-intensity weight loss intervention or none or minimal weight loss support, using at least 1 reported biomarker of liver disease.
The literature search returned 22 eligible studies involving 2,588 patients. The investigators found that more intensive weight loss programs were associated with greater weight loss than lower intensity methods (-3.61 kg; I2 = 95%). Multiple biomarkers of liver disease showed significant improvements in association with formal weight loss programs, including histologically or radiologically measured liver steatosis (standardized mean difference: -1.48; I2 = 94%), histologic NAFLD activity score (-0.92; I2= 95%), presence of nonalcoholic steatohepatitis (OR, 0.14; I2 =0%), alanine aminotransferase (-9.81 U/L; I2= 97%), aspartate transaminase (-4.84 U/L; I2 = 96%), alkaline phosphatase (-5.53 U/L; I2 = 96%), and gamma-glutamyl transferase (-4.35 U/L; I2 = 92%). Weight loss interventions were not significantly associated with histologic liver fibrosis or inflammation, the investigators noted.
“The advantages [of weight loss interventions] seem to be greater in people who are overweight and with NAFLD, but our exploratory results suggest that weight loss interventions might still be beneficial in the minority of people with healthy weight and NAFLD,” the investigators wrote. “Clinicians may use these findings to counsel people with NAFLD on the expected clinically significant improvements in liver biomarkers after weight loss and direct the patients toward valuable interventions.”
“The accumulated evidence supports changing the clinical guidelines and routine practice to recommend formal weight loss programs to treat people with NAFLD,” the investigators concluded.
The study was funded by the National Institute for Health Research (NIHR) Oxford Biomedical Research Centre and the Oxford NIHR Collaboration and Leadership in Applied Health Research. The investigators reported grants for other research from Cambridge Weight Plan.
SOURCE: Koutoukidis et al. JAMA Int Med. 2019 Jul 1. doi: 10.1001/jamainternmed.2019.2248.
The AGA Practice guide on Obesity and Weight management, Education and Resources (POWER) paper provides physicians with a comprehensive, multi-disciplinary process to guide and personalize innovative obesity care for safe and effective weight management. Learn more at https://www.gastro.org/practice-guidance/practice-updates/obesity-practice-guide
For patients with nonalcoholic fatty liver disease (NAFLD), formal weight loss programs lead to statistically and clinically significant improvements in biomarkers of liver disease, based on a recent meta-analysis.
The findings support changing NAFLD guidelines to recommend weight loss interventions, according to lead author Dimitrios A. Koutoukidis, PhD, of the University of Oxford, UK, and colleagues.
“Clinical guidelines around the world recommend physicians offer advice on lifestyle modification, which mostly includes weight loss through hypoenergetic diets and increased physical activity,” the investigators wrote in JAMA Internal Medicine.“However, whether clinicians provide advice and the type of advice they give vary greatly, and guidelines rarely specifically recommend treatment programs to support weight loss,” they added.
To investigate associations between methods of weight loss and improvements in NAFLD, the investigators screened for studies involving behavioral weight loss programs, pharmacotherapy, or bariatric surgery, alone or in combination. To limit confounding, studies combining weight loss with other potential treatments, such as medications, were excluded. Weight loss interventions were compared to liver disease outcomes associated with lower-intensity weight loss intervention or none or minimal weight loss support, using at least 1 reported biomarker of liver disease.
The literature search returned 22 eligible studies involving 2,588 patients. The investigators found that more intensive weight loss programs were associated with greater weight loss than lower intensity methods (-3.61 kg; I2 = 95%). Multiple biomarkers of liver disease showed significant improvements in association with formal weight loss programs, including histologically or radiologically measured liver steatosis (standardized mean difference: -1.48; I2 = 94%), histologic NAFLD activity score (-0.92; I2= 95%), presence of nonalcoholic steatohepatitis (OR, 0.14; I2 =0%), alanine aminotransferase (-9.81 U/L; I2= 97%), aspartate transaminase (-4.84 U/L; I2 = 96%), alkaline phosphatase (-5.53 U/L; I2 = 96%), and gamma-glutamyl transferase (-4.35 U/L; I2 = 92%). Weight loss interventions were not significantly associated with histologic liver fibrosis or inflammation, the investigators noted.
“The advantages [of weight loss interventions] seem to be greater in people who are overweight and with NAFLD, but our exploratory results suggest that weight loss interventions might still be beneficial in the minority of people with healthy weight and NAFLD,” the investigators wrote. “Clinicians may use these findings to counsel people with NAFLD on the expected clinically significant improvements in liver biomarkers after weight loss and direct the patients toward valuable interventions.”
“The accumulated evidence supports changing the clinical guidelines and routine practice to recommend formal weight loss programs to treat people with NAFLD,” the investigators concluded.
The study was funded by the National Institute for Health Research (NIHR) Oxford Biomedical Research Centre and the Oxford NIHR Collaboration and Leadership in Applied Health Research. The investigators reported grants for other research from Cambridge Weight Plan.
SOURCE: Koutoukidis et al. JAMA Int Med. 2019 Jul 1. doi: 10.1001/jamainternmed.2019.2248.
The AGA Practice guide on Obesity and Weight management, Education and Resources (POWER) paper provides physicians with a comprehensive, multi-disciplinary process to guide and personalize innovative obesity care for safe and effective weight management. Learn more at https://www.gastro.org/practice-guidance/practice-updates/obesity-practice-guide
FROM JAMA INTERNAL MEDICINE
Key clinical point: Major finding: Weight loss interventions were associated with significantly decreased alanine aminotransferase (-9.81 U/L; I2 = 97%).
Study details: A meta-analysis of randomized clinicals involving weight loss interventions for patients with nonalcoholic fatty liver disease (NAFLD).
Disclosures: The study was funded by the National Institute for Health Research (NIHR) Oxford Biomedical Research Centre and the Oxford NIHR Collaboration and Leadership in Applied Health Research. The investigators reported grants for other research from Cambridge Weight Plan.
Source: Koutoukidis et al. JAMA Int Med. 2019 Jul 1. doi: 10.1001/jamainternmed.2019.2248.
Past studies have attempted to investigate the relationship between weight loss and nonalcoholic fatty liver disease (NAFLD), but they did so with various interventions and outcomes measures. Fortunately, the study by Dr. Koutoukidis and colleagues helps clear up this variability with a well-conducted systematic review. The results offer a convincing case that formal weight loss programs should be a cornerstone of NALFD treatment, based on improvements in blood, histologic, and radiologic biomarkers of liver disease. Since pharmacologic options for NAFLD are limited, these findings are particularly important.
Although the study did not reveal improvements in fibrosis or inflammation with weight loss, this is likely due to the scarcity of trials with histologic measures or long-term follow-up. Where long-term follow-up was available, weight loss was not maintained, disallowing clear conclusions. Still, other studies have shown that sustained weight loss is associated with improvements in fibrosis and mortality, so clinicians should feel encouraged that formal weight loss programs for patients with NAFLD likely have life-saving consequences.
Elizabeth Adler, MD and Danielle Brandman, MD , are with the University of California, San Francisco. Dr. Brandman reported financial affiliations with Conatus, Gilead, and Allergan. Their remarks are adapted from an accompanying editorial (JAMA Int Med. 2019 Jul 1. doi: 10.1001/jamainternmed.2019.2244 ).
Click for Credit: Roux-en-Y for diabetes; Exercise & fall prevention; more
Here are 5 articles from the July issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Cloud of inconsistency hangs over cannabis data
To take the posttest, go to: https://bit.ly/2NfjaDS
Expires February 6, 2020
2. Roux-en-Y achieves diabetes remission in majority of patients
To take the posttest, go to: https://bit.ly/2x9hLnE
Expires February 6, 2020
3. Socioeconomic status, race found to impact CPAP compliance
To take the posttest, go to: https://bit.ly/2RBpLa9
Expires February 8, 2020
4. Exercise type matters for fall prevention among elderly
To take the posttest, go to: https://bit.ly/2X26OUh
Expires February 12, 2020
5. Adult HIV patients should receive standard vaccinations, with caveats
To take the posttest, go to: https://bit.ly/2X1S7LV
Expires February 12, 2020
Here are 5 articles from the July issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Cloud of inconsistency hangs over cannabis data
To take the posttest, go to: https://bit.ly/2NfjaDS
Expires February 6, 2020
2. Roux-en-Y achieves diabetes remission in majority of patients
To take the posttest, go to: https://bit.ly/2x9hLnE
Expires February 6, 2020
3. Socioeconomic status, race found to impact CPAP compliance
To take the posttest, go to: https://bit.ly/2RBpLa9
Expires February 8, 2020
4. Exercise type matters for fall prevention among elderly
To take the posttest, go to: https://bit.ly/2X26OUh
Expires February 12, 2020
5. Adult HIV patients should receive standard vaccinations, with caveats
To take the posttest, go to: https://bit.ly/2X1S7LV
Expires February 12, 2020
Here are 5 articles from the July issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Cloud of inconsistency hangs over cannabis data
To take the posttest, go to: https://bit.ly/2NfjaDS
Expires February 6, 2020
2. Roux-en-Y achieves diabetes remission in majority of patients
To take the posttest, go to: https://bit.ly/2x9hLnE
Expires February 6, 2020
3. Socioeconomic status, race found to impact CPAP compliance
To take the posttest, go to: https://bit.ly/2RBpLa9
Expires February 8, 2020
4. Exercise type matters for fall prevention among elderly
To take the posttest, go to: https://bit.ly/2X26OUh
Expires February 12, 2020
5. Adult HIV patients should receive standard vaccinations, with caveats
To take the posttest, go to: https://bit.ly/2X1S7LV
Expires February 12, 2020
Mortality risk from mild to moderate OSA modified by age
SAN ANTONIO – , results from a large longitudinal analysis showed.
“The association between severe OSA and significant morbidity and mortality – particularly cardiovascular in nature – is well established,” the study’s first author, Alexandros N. Vgontzas, MD, said at the annual meeting of the Associated Professional Sleep Societies. “In contrast, mild to moderate OSA is highly prevalent in the general population but its association with morbidity and mortality is not well established.”
In an effort to examine the association between mild to moderate OSA and all-cause mortality, Dr. Vgontzas and colleagues drew from the Penn State Adult Cohort, a random general population sample of 1,741 men and women who were studied in the sleep lab with an 8-hour polysomnography at baseline and followed for a mean of 19.2 years for all-cause mortality.
The researchers retrieved mortality data from the Centers for Disease Control and Prevention’s National Death Index and defined mild OSA as an apnea/hypopnea index (AHI) of 5-14.9 events per hour, while moderate OSA was defined as an AHI of 15-29.9 events per hour. They used Cox proportional hazards regression to estimate all-cause mortality and adjusted for race, sex, body mass index, smoking, hypertension, diabetes, heart problems, and stroke.
Dr. Vgontzas, of the Sleep Research and Treatment Center at Penn State University, Hershey, Pa., reported that 596 individuals have died since the study began. On adjusted analysis, patients with an AHI between 5 and 29 were 1.28 times as likely to die overall (P = .019). The researchers found that the association with mortality was stronger among patients younger than age 60, compared with those aged 60 and older. The hazard ratio was 1.44 for study participants younger than age 60 (P = .027), and 1.14 for those aged 60 and older (P = .34).
“Mild to moderate sleep apnea is associated with significant all-cause mortality risk, but the strength of the association decreases markedly with age,” Dr. Vgontzas concluded. “These findings are in line with previous findings that the association of mild to moderate OSA with cardiometabolic risk is modified by age and suggests that OSA in older adults is a distinctly different phenotype than in the young and middle-aged.”
The explanation for the association remains unclear. “Is it because the people of older age have some kind of genetic protection, or is because their sleep apnea is milder?” he asked. “We don’t have the data to tell.”
Dr. Vgontzas reported having no financial disclosures.
SOURCE: Vgontzas A et al. SLEEP 2019, abstract 0504.
SAN ANTONIO – , results from a large longitudinal analysis showed.
“The association between severe OSA and significant morbidity and mortality – particularly cardiovascular in nature – is well established,” the study’s first author, Alexandros N. Vgontzas, MD, said at the annual meeting of the Associated Professional Sleep Societies. “In contrast, mild to moderate OSA is highly prevalent in the general population but its association with morbidity and mortality is not well established.”
In an effort to examine the association between mild to moderate OSA and all-cause mortality, Dr. Vgontzas and colleagues drew from the Penn State Adult Cohort, a random general population sample of 1,741 men and women who were studied in the sleep lab with an 8-hour polysomnography at baseline and followed for a mean of 19.2 years for all-cause mortality.
The researchers retrieved mortality data from the Centers for Disease Control and Prevention’s National Death Index and defined mild OSA as an apnea/hypopnea index (AHI) of 5-14.9 events per hour, while moderate OSA was defined as an AHI of 15-29.9 events per hour. They used Cox proportional hazards regression to estimate all-cause mortality and adjusted for race, sex, body mass index, smoking, hypertension, diabetes, heart problems, and stroke.
Dr. Vgontzas, of the Sleep Research and Treatment Center at Penn State University, Hershey, Pa., reported that 596 individuals have died since the study began. On adjusted analysis, patients with an AHI between 5 and 29 were 1.28 times as likely to die overall (P = .019). The researchers found that the association with mortality was stronger among patients younger than age 60, compared with those aged 60 and older. The hazard ratio was 1.44 for study participants younger than age 60 (P = .027), and 1.14 for those aged 60 and older (P = .34).
“Mild to moderate sleep apnea is associated with significant all-cause mortality risk, but the strength of the association decreases markedly with age,” Dr. Vgontzas concluded. “These findings are in line with previous findings that the association of mild to moderate OSA with cardiometabolic risk is modified by age and suggests that OSA in older adults is a distinctly different phenotype than in the young and middle-aged.”
The explanation for the association remains unclear. “Is it because the people of older age have some kind of genetic protection, or is because their sleep apnea is milder?” he asked. “We don’t have the data to tell.”
Dr. Vgontzas reported having no financial disclosures.
SOURCE: Vgontzas A et al. SLEEP 2019, abstract 0504.
SAN ANTONIO – , results from a large longitudinal analysis showed.
“The association between severe OSA and significant morbidity and mortality – particularly cardiovascular in nature – is well established,” the study’s first author, Alexandros N. Vgontzas, MD, said at the annual meeting of the Associated Professional Sleep Societies. “In contrast, mild to moderate OSA is highly prevalent in the general population but its association with morbidity and mortality is not well established.”
In an effort to examine the association between mild to moderate OSA and all-cause mortality, Dr. Vgontzas and colleagues drew from the Penn State Adult Cohort, a random general population sample of 1,741 men and women who were studied in the sleep lab with an 8-hour polysomnography at baseline and followed for a mean of 19.2 years for all-cause mortality.
The researchers retrieved mortality data from the Centers for Disease Control and Prevention’s National Death Index and defined mild OSA as an apnea/hypopnea index (AHI) of 5-14.9 events per hour, while moderate OSA was defined as an AHI of 15-29.9 events per hour. They used Cox proportional hazards regression to estimate all-cause mortality and adjusted for race, sex, body mass index, smoking, hypertension, diabetes, heart problems, and stroke.
Dr. Vgontzas, of the Sleep Research and Treatment Center at Penn State University, Hershey, Pa., reported that 596 individuals have died since the study began. On adjusted analysis, patients with an AHI between 5 and 29 were 1.28 times as likely to die overall (P = .019). The researchers found that the association with mortality was stronger among patients younger than age 60, compared with those aged 60 and older. The hazard ratio was 1.44 for study participants younger than age 60 (P = .027), and 1.14 for those aged 60 and older (P = .34).
“Mild to moderate sleep apnea is associated with significant all-cause mortality risk, but the strength of the association decreases markedly with age,” Dr. Vgontzas concluded. “These findings are in line with previous findings that the association of mild to moderate OSA with cardiometabolic risk is modified by age and suggests that OSA in older adults is a distinctly different phenotype than in the young and middle-aged.”
The explanation for the association remains unclear. “Is it because the people of older age have some kind of genetic protection, or is because their sleep apnea is milder?” he asked. “We don’t have the data to tell.”
Dr. Vgontzas reported having no financial disclosures.
SOURCE: Vgontzas A et al. SLEEP 2019, abstract 0504.
REPORTING FROM SLEEP 2019
Key clinical point: Among adults with mild to moderate obstructive sleep apnea, the risk of mortality is highest among those younger than age 60.
Major finding: The hazard ratio for mortality was 1.44 for study participants younger than age 60 (P = .027), and 1.14 for those aged 60 and older (P = .34).
Study details: An analysis of 1,741 men and women from the Penn State Adult Cohort.
Disclosures: Dr. Vgontzas reported having no financial disclosures.
Source: Vgontzas A et al. SLEEP 2019, Abstract 0504.
Daytime eating schedule found to help with weight management
SAN ANTONIO – In adults of normal weight, a small controlled study has shown that a , independent of caloric intake.
The findings come from an 8-week controlled trial presented at the annual meeting of the Associated Professional Sleep Societies, which set out to examine the impact of a daytime versus delayed eating schedule on body mass, adiposity, and energy homeostasis in adults of normal weight.
“It is best to stop eating as early as possible in the day, and to not eat late at night,” the study’s first author, Namni Goel, PhD, said in an interview at the meeting. “There’s an open question in our field: Should you stop eating at 7:00 p.m.? 8:00 p.m.? My own feeling is, the longer it is between when you stop eating and go to bed, the better off you are metabolically.”
Dr. Goel, associate professor in the division of sleep and chronobiology in the department of psychiatry at the University of Pennsylvania, Philadelphia, and colleagues enrolled 12 healthy adults to participate in a randomized cross-over study in free-living conditions. Three meals and two snacks consisting of comparable energy and macronutrient content were provided during two 8-week counterbalanced phases: 1) daytime eating (food consumed between 8:00 a.m. and 7:00 p.m, and 2) delayed eating (food consumed between 12:00 p.m. and 11:00 p.m. A 2-week washout period occurred between the conditions. “What we wanted to do is just manipulate the timing of eating and we provided all of the meals so we could control the caloric intake,” Dr. Goel said.
The researchers asked participants to maintain a sleep-wake cycle between 11:00 p.m. and 9:00 a.m. (verified by wrist actigraphy) and to limit physical activity. They assessed weight, adiposity, energy metabolism, and hormonal markers during four inpatient visits: 1) baseline; 2) after the first eating condition; 3) after the washout period, before the second eating condition began; and 4) after the second eating condition. They used two-way analysis of variance and Cohen’s d effect sizes to examine changes in anthropometrics and metabolic measures affected by eating schedule (daytime vs. delayed) and time (before vs. after each eating schedule).
The mean age of 12 study participants was 26 years; five were females. Their mean body mass index was 21.9 kg/m2. Dr. Goel reported that participants had excellent adherence to assigned eating schedules, with no differences between the conditions. Weight was decreased on the daytime vs. delayed eating schedule. Specifically, Cohen’s d effect sizes were 0.57 overall: 1.16 for females and 0.33 for males, all in the small to large range. Resting energy expenditure, respiratory quotient, and trunk fat percentage/leg fat percentage were decreased on the daytime vs. delayed eating condition, with Cohen’s d effect sizes of 0.45-1.02, all in the medium to large range. In addition, total cholesterol and insulin were decreased on the daytime eating condition (medium effect sizes of 0.60 and 0.57, respectively), while triglycerides and glucose were increased on the delayed condition (medium effect sizes of 0.46 and 0.52, respectively).
Weight, adiposity, energy metabolism, and hormonal measures did not differ significantly between the pre-daytime and pre-delayed eating conditions, suggesting that they returned to pre-condition levels after the washout period.
“One of the things we’re advocating is that with consistent daytime eating, you can lose weight and/or remain at weight maintenance,” Dr. Goel said. “Consistency is very important. Across 8 weeks, you’re becoming metabolically healthier because you’re not eating that late-night meal or snack. We had shown in previous sleep loss studies that people were eating 500 calories late in the evening on consecutive nights and gaining a substantial amount of weight.”
She and her colleagues are currently enrolling obese individuals into a similarly designed trial, “where we expect much bigger changes metabolically,” she said. The study was funded by a grant from the National Institutes of Health. Dr. Goel reported having no financial disclosures.
SOURCE: Goel N et al. SLEEP 2019, Abstract 0036.
SAN ANTONIO – In adults of normal weight, a small controlled study has shown that a , independent of caloric intake.
The findings come from an 8-week controlled trial presented at the annual meeting of the Associated Professional Sleep Societies, which set out to examine the impact of a daytime versus delayed eating schedule on body mass, adiposity, and energy homeostasis in adults of normal weight.
“It is best to stop eating as early as possible in the day, and to not eat late at night,” the study’s first author, Namni Goel, PhD, said in an interview at the meeting. “There’s an open question in our field: Should you stop eating at 7:00 p.m.? 8:00 p.m.? My own feeling is, the longer it is between when you stop eating and go to bed, the better off you are metabolically.”
Dr. Goel, associate professor in the division of sleep and chronobiology in the department of psychiatry at the University of Pennsylvania, Philadelphia, and colleagues enrolled 12 healthy adults to participate in a randomized cross-over study in free-living conditions. Three meals and two snacks consisting of comparable energy and macronutrient content were provided during two 8-week counterbalanced phases: 1) daytime eating (food consumed between 8:00 a.m. and 7:00 p.m, and 2) delayed eating (food consumed between 12:00 p.m. and 11:00 p.m. A 2-week washout period occurred between the conditions. “What we wanted to do is just manipulate the timing of eating and we provided all of the meals so we could control the caloric intake,” Dr. Goel said.
The researchers asked participants to maintain a sleep-wake cycle between 11:00 p.m. and 9:00 a.m. (verified by wrist actigraphy) and to limit physical activity. They assessed weight, adiposity, energy metabolism, and hormonal markers during four inpatient visits: 1) baseline; 2) after the first eating condition; 3) after the washout period, before the second eating condition began; and 4) after the second eating condition. They used two-way analysis of variance and Cohen’s d effect sizes to examine changes in anthropometrics and metabolic measures affected by eating schedule (daytime vs. delayed) and time (before vs. after each eating schedule).
The mean age of 12 study participants was 26 years; five were females. Their mean body mass index was 21.9 kg/m2. Dr. Goel reported that participants had excellent adherence to assigned eating schedules, with no differences between the conditions. Weight was decreased on the daytime vs. delayed eating schedule. Specifically, Cohen’s d effect sizes were 0.57 overall: 1.16 for females and 0.33 for males, all in the small to large range. Resting energy expenditure, respiratory quotient, and trunk fat percentage/leg fat percentage were decreased on the daytime vs. delayed eating condition, with Cohen’s d effect sizes of 0.45-1.02, all in the medium to large range. In addition, total cholesterol and insulin were decreased on the daytime eating condition (medium effect sizes of 0.60 and 0.57, respectively), while triglycerides and glucose were increased on the delayed condition (medium effect sizes of 0.46 and 0.52, respectively).
Weight, adiposity, energy metabolism, and hormonal measures did not differ significantly between the pre-daytime and pre-delayed eating conditions, suggesting that they returned to pre-condition levels after the washout period.
“One of the things we’re advocating is that with consistent daytime eating, you can lose weight and/or remain at weight maintenance,” Dr. Goel said. “Consistency is very important. Across 8 weeks, you’re becoming metabolically healthier because you’re not eating that late-night meal or snack. We had shown in previous sleep loss studies that people were eating 500 calories late in the evening on consecutive nights and gaining a substantial amount of weight.”
She and her colleagues are currently enrolling obese individuals into a similarly designed trial, “where we expect much bigger changes metabolically,” she said. The study was funded by a grant from the National Institutes of Health. Dr. Goel reported having no financial disclosures.
SOURCE: Goel N et al. SLEEP 2019, Abstract 0036.
SAN ANTONIO – In adults of normal weight, a small controlled study has shown that a , independent of caloric intake.
The findings come from an 8-week controlled trial presented at the annual meeting of the Associated Professional Sleep Societies, which set out to examine the impact of a daytime versus delayed eating schedule on body mass, adiposity, and energy homeostasis in adults of normal weight.
“It is best to stop eating as early as possible in the day, and to not eat late at night,” the study’s first author, Namni Goel, PhD, said in an interview at the meeting. “There’s an open question in our field: Should you stop eating at 7:00 p.m.? 8:00 p.m.? My own feeling is, the longer it is between when you stop eating and go to bed, the better off you are metabolically.”
Dr. Goel, associate professor in the division of sleep and chronobiology in the department of psychiatry at the University of Pennsylvania, Philadelphia, and colleagues enrolled 12 healthy adults to participate in a randomized cross-over study in free-living conditions. Three meals and two snacks consisting of comparable energy and macronutrient content were provided during two 8-week counterbalanced phases: 1) daytime eating (food consumed between 8:00 a.m. and 7:00 p.m, and 2) delayed eating (food consumed between 12:00 p.m. and 11:00 p.m. A 2-week washout period occurred between the conditions. “What we wanted to do is just manipulate the timing of eating and we provided all of the meals so we could control the caloric intake,” Dr. Goel said.
The researchers asked participants to maintain a sleep-wake cycle between 11:00 p.m. and 9:00 a.m. (verified by wrist actigraphy) and to limit physical activity. They assessed weight, adiposity, energy metabolism, and hormonal markers during four inpatient visits: 1) baseline; 2) after the first eating condition; 3) after the washout period, before the second eating condition began; and 4) after the second eating condition. They used two-way analysis of variance and Cohen’s d effect sizes to examine changes in anthropometrics and metabolic measures affected by eating schedule (daytime vs. delayed) and time (before vs. after each eating schedule).
The mean age of 12 study participants was 26 years; five were females. Their mean body mass index was 21.9 kg/m2. Dr. Goel reported that participants had excellent adherence to assigned eating schedules, with no differences between the conditions. Weight was decreased on the daytime vs. delayed eating schedule. Specifically, Cohen’s d effect sizes were 0.57 overall: 1.16 for females and 0.33 for males, all in the small to large range. Resting energy expenditure, respiratory quotient, and trunk fat percentage/leg fat percentage were decreased on the daytime vs. delayed eating condition, with Cohen’s d effect sizes of 0.45-1.02, all in the medium to large range. In addition, total cholesterol and insulin were decreased on the daytime eating condition (medium effect sizes of 0.60 and 0.57, respectively), while triglycerides and glucose were increased on the delayed condition (medium effect sizes of 0.46 and 0.52, respectively).
Weight, adiposity, energy metabolism, and hormonal measures did not differ significantly between the pre-daytime and pre-delayed eating conditions, suggesting that they returned to pre-condition levels after the washout period.
“One of the things we’re advocating is that with consistent daytime eating, you can lose weight and/or remain at weight maintenance,” Dr. Goel said. “Consistency is very important. Across 8 weeks, you’re becoming metabolically healthier because you’re not eating that late-night meal or snack. We had shown in previous sleep loss studies that people were eating 500 calories late in the evening on consecutive nights and gaining a substantial amount of weight.”
She and her colleagues are currently enrolling obese individuals into a similarly designed trial, “where we expect much bigger changes metabolically,” she said. The study was funded by a grant from the National Institutes of Health. Dr. Goel reported having no financial disclosures.
SOURCE: Goel N et al. SLEEP 2019, Abstract 0036.
REPORTING FROM SLEEP 2019
Key clinical point: A daytime eating schedule is likely beneficial for weight management and metabolic health.
Major finding: Weight was decreased on the daytime vs. delayed eating schedule with Cohen’s d effect of 0.57 overall: 1.16 for females and 0.33 for males, all in the small to large range.
Study details: A randomized trial of 12 healthy adults with normal body weight.
Disclosures: The study was funded by a grant from the National Institutes of Health. Dr. Goel reported having no financial disclosures.
Source: Goel N et al. SLEEP 2019, Abstract 0036.
Obesity and overweight declined among lower-income kids
The combined rate of , according to a study in JAMA.
Liping Pan, MD, MPH, of the Centers for Disease Control and Prevention and colleagues used data from the WIC Participant and Program Characteristics survey from 2010, 2012, 2014, and 2016 for 12,403,629 children aged 2-4 years from 50 states, Washington, D.C., and 5 territories. In addition to a –3.2% change (95% confidence interval, –3.3% to –3.2%) in adjusted prevalence difference for the combined rate of obesity and overweight seen between 2010 and 2016, the researchers found the crude prevalence decreased from 32.5% to 29.1%. A decrease was also seen for obesity alone (crude prevalence, 15.9% to 13.9%; adjusted prevalence difference, –1.9%; 95% CI, –1.9% to –1.8%).
One of the limitations of the study is that the characteristics of enrolled children might differ from those of children not enrolled in this WIC program; however, the researchers noted that they accounted for many demographic characteristics in the trend analyses.
“Reasons for the declines in obesity among young children in WIC remain undetermined but may include WIC food package revisions and local, state, and national initiatives,” they wrote.
SOURCE: Pan L et al. JAMA. 2019 Jun 18;321(23):2364-6.
The combined rate of , according to a study in JAMA.
Liping Pan, MD, MPH, of the Centers for Disease Control and Prevention and colleagues used data from the WIC Participant and Program Characteristics survey from 2010, 2012, 2014, and 2016 for 12,403,629 children aged 2-4 years from 50 states, Washington, D.C., and 5 territories. In addition to a –3.2% change (95% confidence interval, –3.3% to –3.2%) in adjusted prevalence difference for the combined rate of obesity and overweight seen between 2010 and 2016, the researchers found the crude prevalence decreased from 32.5% to 29.1%. A decrease was also seen for obesity alone (crude prevalence, 15.9% to 13.9%; adjusted prevalence difference, –1.9%; 95% CI, –1.9% to –1.8%).
One of the limitations of the study is that the characteristics of enrolled children might differ from those of children not enrolled in this WIC program; however, the researchers noted that they accounted for many demographic characteristics in the trend analyses.
“Reasons for the declines in obesity among young children in WIC remain undetermined but may include WIC food package revisions and local, state, and national initiatives,” they wrote.
SOURCE: Pan L et al. JAMA. 2019 Jun 18;321(23):2364-6.
The combined rate of , according to a study in JAMA.
Liping Pan, MD, MPH, of the Centers for Disease Control and Prevention and colleagues used data from the WIC Participant and Program Characteristics survey from 2010, 2012, 2014, and 2016 for 12,403,629 children aged 2-4 years from 50 states, Washington, D.C., and 5 territories. In addition to a –3.2% change (95% confidence interval, –3.3% to –3.2%) in adjusted prevalence difference for the combined rate of obesity and overweight seen between 2010 and 2016, the researchers found the crude prevalence decreased from 32.5% to 29.1%. A decrease was also seen for obesity alone (crude prevalence, 15.9% to 13.9%; adjusted prevalence difference, –1.9%; 95% CI, –1.9% to –1.8%).
One of the limitations of the study is that the characteristics of enrolled children might differ from those of children not enrolled in this WIC program; however, the researchers noted that they accounted for many demographic characteristics in the trend analyses.
“Reasons for the declines in obesity among young children in WIC remain undetermined but may include WIC food package revisions and local, state, and national initiatives,” they wrote.
SOURCE: Pan L et al. JAMA. 2019 Jun 18;321(23):2364-6.
FROM JAMA
Physical activity prevalence shows urban/rural divide
according to the Centers for Disease Control and Prevention.
The prevalence of meeting the aerobic and muscle-strengthening recommendations in the 2008 Physical Activity Guidelines for Americans rose from 18.2% of adults in 2008 to 24.3% in 2017, but despite that increase, “insufficient participation in physical activity remains a public health concern,” Geoffrey P. Whitfield, PhD, and his associates said in the Morbidity and Mortality Weekly Report.
There was progress among both urban and rural residents, but those in rural areas were behind at the start of the study period in 2008 and remained behind in 2017. The prevalence of meeting the activity guideline started at 13.3% for rural residents and 19.4% for urbanites and rose to 19.6% and 25.3%, respectively, in 2017 – that’s an annual percentage point change of 0.5% for each population, the investigators reported. Rates among women were well below those of men in both populations.
Rural communities may lack the infrastructure, such as sidewalks, schoolyards, and parks, to support physical activities, or rural residents may get more exercise through occupational and domestic tasks, rather than through the leisure-time activities that are the focus of the National Health Interview Survey, which was the source of the study data, Dr. Whitfield and his associates suggested.
The 2008 federal guidelines recommend that adults get 150-300 minutes of moderate-intensity or 75-150 minutes of vigorous-intensity aerobic physical activity per week, along with muscle-strengthening activities of at least moderate intensity involving all major muscle groups on 2 or more days each week.
SOURCE: Whitfield GP et al. MMWR. 2019 Jun 14;68(23):514-8.
according to the Centers for Disease Control and Prevention.

The prevalence of meeting the aerobic and muscle-strengthening recommendations in the 2008 Physical Activity Guidelines for Americans rose from 18.2% of adults in 2008 to 24.3% in 2017, but despite that increase, “insufficient participation in physical activity remains a public health concern,” Geoffrey P. Whitfield, PhD, and his associates said in the Morbidity and Mortality Weekly Report.
There was progress among both urban and rural residents, but those in rural areas were behind at the start of the study period in 2008 and remained behind in 2017. The prevalence of meeting the activity guideline started at 13.3% for rural residents and 19.4% for urbanites and rose to 19.6% and 25.3%, respectively, in 2017 – that’s an annual percentage point change of 0.5% for each population, the investigators reported. Rates among women were well below those of men in both populations.
Rural communities may lack the infrastructure, such as sidewalks, schoolyards, and parks, to support physical activities, or rural residents may get more exercise through occupational and domestic tasks, rather than through the leisure-time activities that are the focus of the National Health Interview Survey, which was the source of the study data, Dr. Whitfield and his associates suggested.
The 2008 federal guidelines recommend that adults get 150-300 minutes of moderate-intensity or 75-150 minutes of vigorous-intensity aerobic physical activity per week, along with muscle-strengthening activities of at least moderate intensity involving all major muscle groups on 2 or more days each week.
SOURCE: Whitfield GP et al. MMWR. 2019 Jun 14;68(23):514-8.
according to the Centers for Disease Control and Prevention.

The prevalence of meeting the aerobic and muscle-strengthening recommendations in the 2008 Physical Activity Guidelines for Americans rose from 18.2% of adults in 2008 to 24.3% in 2017, but despite that increase, “insufficient participation in physical activity remains a public health concern,” Geoffrey P. Whitfield, PhD, and his associates said in the Morbidity and Mortality Weekly Report.
There was progress among both urban and rural residents, but those in rural areas were behind at the start of the study period in 2008 and remained behind in 2017. The prevalence of meeting the activity guideline started at 13.3% for rural residents and 19.4% for urbanites and rose to 19.6% and 25.3%, respectively, in 2017 – that’s an annual percentage point change of 0.5% for each population, the investigators reported. Rates among women were well below those of men in both populations.
Rural communities may lack the infrastructure, such as sidewalks, schoolyards, and parks, to support physical activities, or rural residents may get more exercise through occupational and domestic tasks, rather than through the leisure-time activities that are the focus of the National Health Interview Survey, which was the source of the study data, Dr. Whitfield and his associates suggested.
The 2008 federal guidelines recommend that adults get 150-300 minutes of moderate-intensity or 75-150 minutes of vigorous-intensity aerobic physical activity per week, along with muscle-strengthening activities of at least moderate intensity involving all major muscle groups on 2 or more days each week.
SOURCE: Whitfield GP et al. MMWR. 2019 Jun 14;68(23):514-8.
FROM MMWR
Weight loss in knee OA patients sustained with liraglutide over 1 year
MADRID – The glucagonlike peptide–1 receptor agonist liraglutide appears to be effective for keeping weight off following an intensive weight-loss program in patients with knee osteoarthritis, according to a randomized, double-blind, placebo-controlled trial presented at the European Congress of Rheumatology.
However, even though the 8-week intensive dietary program led to substantial weight loss and significant improvement in pain, additional weight loss of nearly 2.5 kg over 52 weeks of daily liraglutide treatment did not translate into more pain control.
According to study author Lars Erik Kristensen, MD, PhD, this is the first randomized trial to test the ability of liraglutide to provide a sustained weight loss in OA patients. The Food and Drug Administration indication for liraglutide is as an adjunct to diet and exercise for glycemic control in type 2 diabetes mellitus.
The study compared liraglutide against placebo in patients who had completed an intensive weight-control program in which the median loss was 12.46 kg. They were followed for 52 weeks.
At the end of follow-up, patients in the placebo group had gained a mean of 1.17 kg while those randomized to liraglutide lost an additional 2.76 kg. The between-group difference of 3.93 kg was statistically significant (P = .008).
“We believe that liraglutide is a promising agent for sustained weight loss in OA patients,” concluded Dr. Kristensen, a clinical researcher in rheumatology in the Parker Institute at Bispebjerg-Frederiksberg Hospital in Copenhagen.
In the single-center study, 156 patients were enrolled and randomized. In an initial 8-week diet intervention undertaken by both groups, an intensive program for weight loss included average daily calorie intakes of less than 800 kcal along with dietetic counseling. Patients were monitored for daily activities.
The majority of patients achieved a 10% or greater loss of total body weight during the intensive program before initiating 3 mg of once-daily liraglutide or a placebo.
Over the course of 52 weeks, the attrition from the study was relatively low. Among the 80 patients randomized to liraglutide, only 2 were lost because of noncompliance. Another 12 participants left the study before completion, 10 of whom did so for treatment-associated adverse effects. In the placebo arm, four patients were noncompliant, four left for treatment-associated adverse effects, and five left for other reasons.
Following the 8-week intensive dietary program, there was 11.86-point improvement in the pain subscale of the Knee and Osteoarthritis Outcome Score, confirming a substantial symptomatic benefit from this degree of weight loss. While this improvement in pain score was sustained at 52 weeks in both groups, the additional weight loss in the liraglutide arm did not lead to additional pain control.
The lack of additional pain control in the liraglutide group was disappointing, and the reason is unclear, but Dr. Kristensen emphasized that the persistent improvement in pain control was a positive result. In patients who are overweight or obese, regardless of whether they have concomitant OA, weight loss is not only difficult to achieve but difficult to sustain even after a successful intervention.
Dr. Kristensen reported financial relationships with multiple pharmaceutical companies. The trial received funding from Novo Nordisk.
SOURCE: Kristensen LE et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):71-2. Abstract OP0011. doi: 10.1136/annrheumdis-2019-eular.1375.
MADRID – The glucagonlike peptide–1 receptor agonist liraglutide appears to be effective for keeping weight off following an intensive weight-loss program in patients with knee osteoarthritis, according to a randomized, double-blind, placebo-controlled trial presented at the European Congress of Rheumatology.
However, even though the 8-week intensive dietary program led to substantial weight loss and significant improvement in pain, additional weight loss of nearly 2.5 kg over 52 weeks of daily liraglutide treatment did not translate into more pain control.
According to study author Lars Erik Kristensen, MD, PhD, this is the first randomized trial to test the ability of liraglutide to provide a sustained weight loss in OA patients. The Food and Drug Administration indication for liraglutide is as an adjunct to diet and exercise for glycemic control in type 2 diabetes mellitus.
The study compared liraglutide against placebo in patients who had completed an intensive weight-control program in which the median loss was 12.46 kg. They were followed for 52 weeks.
At the end of follow-up, patients in the placebo group had gained a mean of 1.17 kg while those randomized to liraglutide lost an additional 2.76 kg. The between-group difference of 3.93 kg was statistically significant (P = .008).
“We believe that liraglutide is a promising agent for sustained weight loss in OA patients,” concluded Dr. Kristensen, a clinical researcher in rheumatology in the Parker Institute at Bispebjerg-Frederiksberg Hospital in Copenhagen.
In the single-center study, 156 patients were enrolled and randomized. In an initial 8-week diet intervention undertaken by both groups, an intensive program for weight loss included average daily calorie intakes of less than 800 kcal along with dietetic counseling. Patients were monitored for daily activities.
The majority of patients achieved a 10% or greater loss of total body weight during the intensive program before initiating 3 mg of once-daily liraglutide or a placebo.
Over the course of 52 weeks, the attrition from the study was relatively low. Among the 80 patients randomized to liraglutide, only 2 were lost because of noncompliance. Another 12 participants left the study before completion, 10 of whom did so for treatment-associated adverse effects. In the placebo arm, four patients were noncompliant, four left for treatment-associated adverse effects, and five left for other reasons.
Following the 8-week intensive dietary program, there was 11.86-point improvement in the pain subscale of the Knee and Osteoarthritis Outcome Score, confirming a substantial symptomatic benefit from this degree of weight loss. While this improvement in pain score was sustained at 52 weeks in both groups, the additional weight loss in the liraglutide arm did not lead to additional pain control.
The lack of additional pain control in the liraglutide group was disappointing, and the reason is unclear, but Dr. Kristensen emphasized that the persistent improvement in pain control was a positive result. In patients who are overweight or obese, regardless of whether they have concomitant OA, weight loss is not only difficult to achieve but difficult to sustain even after a successful intervention.
Dr. Kristensen reported financial relationships with multiple pharmaceutical companies. The trial received funding from Novo Nordisk.
SOURCE: Kristensen LE et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):71-2. Abstract OP0011. doi: 10.1136/annrheumdis-2019-eular.1375.
MADRID – The glucagonlike peptide–1 receptor agonist liraglutide appears to be effective for keeping weight off following an intensive weight-loss program in patients with knee osteoarthritis, according to a randomized, double-blind, placebo-controlled trial presented at the European Congress of Rheumatology.
However, even though the 8-week intensive dietary program led to substantial weight loss and significant improvement in pain, additional weight loss of nearly 2.5 kg over 52 weeks of daily liraglutide treatment did not translate into more pain control.
According to study author Lars Erik Kristensen, MD, PhD, this is the first randomized trial to test the ability of liraglutide to provide a sustained weight loss in OA patients. The Food and Drug Administration indication for liraglutide is as an adjunct to diet and exercise for glycemic control in type 2 diabetes mellitus.
The study compared liraglutide against placebo in patients who had completed an intensive weight-control program in which the median loss was 12.46 kg. They were followed for 52 weeks.
At the end of follow-up, patients in the placebo group had gained a mean of 1.17 kg while those randomized to liraglutide lost an additional 2.76 kg. The between-group difference of 3.93 kg was statistically significant (P = .008).
“We believe that liraglutide is a promising agent for sustained weight loss in OA patients,” concluded Dr. Kristensen, a clinical researcher in rheumatology in the Parker Institute at Bispebjerg-Frederiksberg Hospital in Copenhagen.
In the single-center study, 156 patients were enrolled and randomized. In an initial 8-week diet intervention undertaken by both groups, an intensive program for weight loss included average daily calorie intakes of less than 800 kcal along with dietetic counseling. Patients were monitored for daily activities.
The majority of patients achieved a 10% or greater loss of total body weight during the intensive program before initiating 3 mg of once-daily liraglutide or a placebo.
Over the course of 52 weeks, the attrition from the study was relatively low. Among the 80 patients randomized to liraglutide, only 2 were lost because of noncompliance. Another 12 participants left the study before completion, 10 of whom did so for treatment-associated adverse effects. In the placebo arm, four patients were noncompliant, four left for treatment-associated adverse effects, and five left for other reasons.
Following the 8-week intensive dietary program, there was 11.86-point improvement in the pain subscale of the Knee and Osteoarthritis Outcome Score, confirming a substantial symptomatic benefit from this degree of weight loss. While this improvement in pain score was sustained at 52 weeks in both groups, the additional weight loss in the liraglutide arm did not lead to additional pain control.
The lack of additional pain control in the liraglutide group was disappointing, and the reason is unclear, but Dr. Kristensen emphasized that the persistent improvement in pain control was a positive result. In patients who are overweight or obese, regardless of whether they have concomitant OA, weight loss is not only difficult to achieve but difficult to sustain even after a successful intervention.
Dr. Kristensen reported financial relationships with multiple pharmaceutical companies. The trial received funding from Novo Nordisk.
SOURCE: Kristensen LE et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):71-2. Abstract OP0011. doi: 10.1136/annrheumdis-2019-eular.1375.
REPORTING FROM EULAR 2019 CONGRESS