User login
Joint effort: CBD not just innocent bystander in weed
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
I visited a legal cannabis dispensary in Massachusetts a few years ago, mostly to see what the hype was about. There I was, knowing basically nothing about pot, as the gentle stoner behind the counter explained to me the differences between the various strains. Acapulco Gold is buoyant and energizing; Purple Kush is sleepy, relaxed, dissociative. Here’s a strain that makes you feel nostalgic; here’s one that helps you focus. It was as complicated and as oddly specific as a fancy wine tasting – and, I had a feeling, about as reliable.
It’s a plant, after all, and though delta-9-tetrahydrocannabinol (THC) is the chemical responsible for its euphoric effects, it is far from the only substance in there.
The second most important compound in cannabis is cannabidiol, and most people will tell you that CBD is the gentle yin to THC’s paranoiac yang. Hence your local ganja barista reminding you that, if you don›t want all those anxiety-inducing side effects of THC, grab a strain with a nice CBD balance.
But is it true? A new study appearing in JAMA Network Open suggests, in fact, that it’s quite the opposite. This study is from Austin Zamarripa and colleagues, who clearly sit at the researcher cool kids table.
Eighteen adults who had abstained from marijuana use for at least a month participated in this trial (which is way more fun than anything we do in my lab at Yale). In random order, separated by at least a week, they ate some special brownies.
Condition one was a control brownie, condition two was a brownie containing 20 mg of THC, and condition three was a brownie containing 20 mg of THC and 640 mg of CBD. Participants were assigned each condition in random order, separated by at least a week.
A side note on doses for those of you who, like me, are not totally weed literate. A dose of 20 mg of THC is about a third of what you might find in a typical joint these days (though it’s about double the THC content of a joint in the ‘70s – I believe the technical term is “doobie”). And 640 mg of CBD is a decent dose, as 5 mg per kilogram is what some folks start with to achieve therapeutic effects.
Both THC and CBD interact with the cytochrome p450 system in the liver. This matters when you’re ingesting them instead of smoking them because you have first-pass metabolism to contend with. And, because of that p450 inhibition, it’s possible that CBD might actually increase the amount of THC that gets into your bloodstream from the brownie, or gummy, or pizza sauce, or whatever.
Let’s get to the results, starting with blood THC concentration. It’s not subtle. With CBD on board the THC concentration rises higher faster, with roughly double the area under the curve.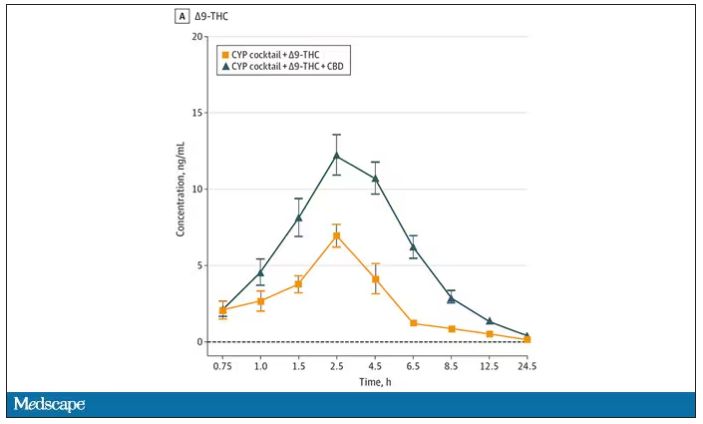
And, unsurprisingly, the subjective experience correlated with those higher levels. Individuals rated the “drug effect” higher with the combo. But, interestingly, the “pleasant” drug effect didn’t change much, while the unpleasant effects were substantially higher. No mitigation of THC anxiety here – quite the opposite. CBD made the anxiety worse.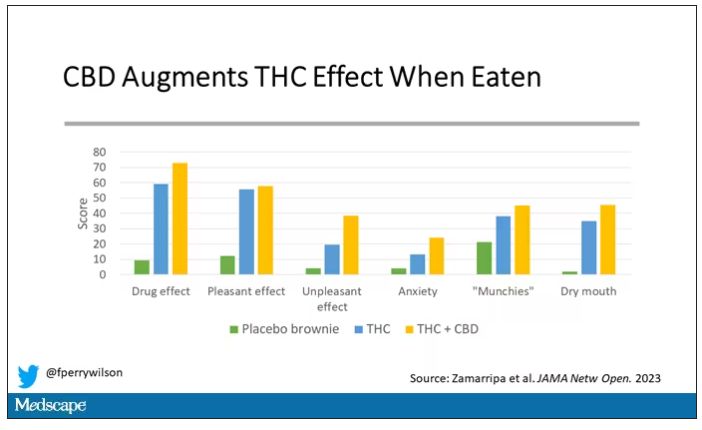
Cognitive effects were equally profound. Scores on a digit symbol substitution test and a paced serial addition task were all substantially worse when CBD was mixed with THC.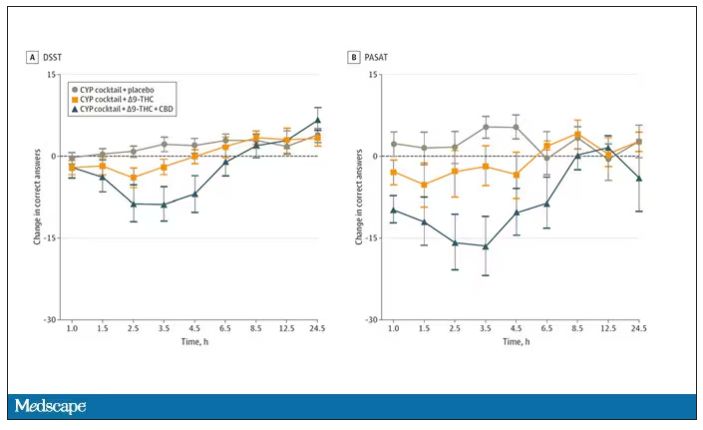
And for those of you who want some more objective measures, check out the heart rate. Despite the purported “calming” nature of CBD, heart rates were way higher when individuals were exposed to both chemicals.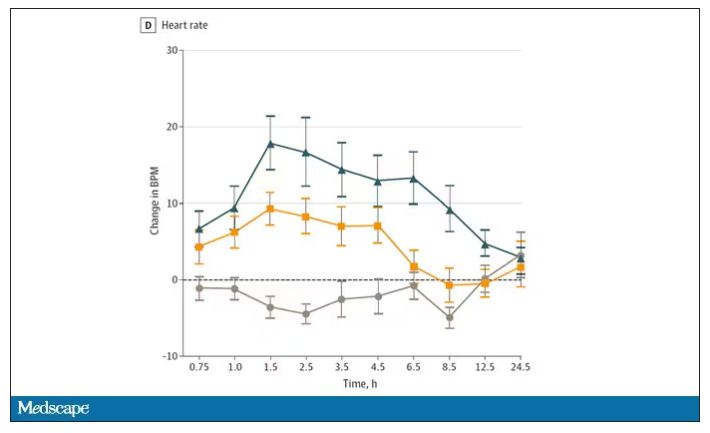
The picture here is quite clear, though the mechanism is not. At least when talking edibles, CBD enhances the effects of THC, and not necessarily for the better. It may be that CBD is competing with some of the proteins that metabolize THC, thus prolonging its effects. CBD may also directly inhibit those enzymes. But whatever the case, I think we can safely say the myth that CBD makes the effects of THC more mild or more tolerable is busted.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale University’s Clinical and Translational Research Accelerator in New Haven, Conn.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
I visited a legal cannabis dispensary in Massachusetts a few years ago, mostly to see what the hype was about. There I was, knowing basically nothing about pot, as the gentle stoner behind the counter explained to me the differences between the various strains. Acapulco Gold is buoyant and energizing; Purple Kush is sleepy, relaxed, dissociative. Here’s a strain that makes you feel nostalgic; here’s one that helps you focus. It was as complicated and as oddly specific as a fancy wine tasting – and, I had a feeling, about as reliable.
It’s a plant, after all, and though delta-9-tetrahydrocannabinol (THC) is the chemical responsible for its euphoric effects, it is far from the only substance in there.
The second most important compound in cannabis is cannabidiol, and most people will tell you that CBD is the gentle yin to THC’s paranoiac yang. Hence your local ganja barista reminding you that, if you don›t want all those anxiety-inducing side effects of THC, grab a strain with a nice CBD balance.
But is it true? A new study appearing in JAMA Network Open suggests, in fact, that it’s quite the opposite. This study is from Austin Zamarripa and colleagues, who clearly sit at the researcher cool kids table.
Eighteen adults who had abstained from marijuana use for at least a month participated in this trial (which is way more fun than anything we do in my lab at Yale). In random order, separated by at least a week, they ate some special brownies.
Condition one was a control brownie, condition two was a brownie containing 20 mg of THC, and condition three was a brownie containing 20 mg of THC and 640 mg of CBD. Participants were assigned each condition in random order, separated by at least a week.
A side note on doses for those of you who, like me, are not totally weed literate. A dose of 20 mg of THC is about a third of what you might find in a typical joint these days (though it’s about double the THC content of a joint in the ‘70s – I believe the technical term is “doobie”). And 640 mg of CBD is a decent dose, as 5 mg per kilogram is what some folks start with to achieve therapeutic effects.
Both THC and CBD interact with the cytochrome p450 system in the liver. This matters when you’re ingesting them instead of smoking them because you have first-pass metabolism to contend with. And, because of that p450 inhibition, it’s possible that CBD might actually increase the amount of THC that gets into your bloodstream from the brownie, or gummy, or pizza sauce, or whatever.
Let’s get to the results, starting with blood THC concentration. It’s not subtle. With CBD on board the THC concentration rises higher faster, with roughly double the area under the curve.
And, unsurprisingly, the subjective experience correlated with those higher levels. Individuals rated the “drug effect” higher with the combo. But, interestingly, the “pleasant” drug effect didn’t change much, while the unpleasant effects were substantially higher. No mitigation of THC anxiety here – quite the opposite. CBD made the anxiety worse.
Cognitive effects were equally profound. Scores on a digit symbol substitution test and a paced serial addition task were all substantially worse when CBD was mixed with THC.
And for those of you who want some more objective measures, check out the heart rate. Despite the purported “calming” nature of CBD, heart rates were way higher when individuals were exposed to both chemicals.
The picture here is quite clear, though the mechanism is not. At least when talking edibles, CBD enhances the effects of THC, and not necessarily for the better. It may be that CBD is competing with some of the proteins that metabolize THC, thus prolonging its effects. CBD may also directly inhibit those enzymes. But whatever the case, I think we can safely say the myth that CBD makes the effects of THC more mild or more tolerable is busted.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale University’s Clinical and Translational Research Accelerator in New Haven, Conn.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
I visited a legal cannabis dispensary in Massachusetts a few years ago, mostly to see what the hype was about. There I was, knowing basically nothing about pot, as the gentle stoner behind the counter explained to me the differences between the various strains. Acapulco Gold is buoyant and energizing; Purple Kush is sleepy, relaxed, dissociative. Here’s a strain that makes you feel nostalgic; here’s one that helps you focus. It was as complicated and as oddly specific as a fancy wine tasting – and, I had a feeling, about as reliable.
It’s a plant, after all, and though delta-9-tetrahydrocannabinol (THC) is the chemical responsible for its euphoric effects, it is far from the only substance in there.
The second most important compound in cannabis is cannabidiol, and most people will tell you that CBD is the gentle yin to THC’s paranoiac yang. Hence your local ganja barista reminding you that, if you don›t want all those anxiety-inducing side effects of THC, grab a strain with a nice CBD balance.
But is it true? A new study appearing in JAMA Network Open suggests, in fact, that it’s quite the opposite. This study is from Austin Zamarripa and colleagues, who clearly sit at the researcher cool kids table.
Eighteen adults who had abstained from marijuana use for at least a month participated in this trial (which is way more fun than anything we do in my lab at Yale). In random order, separated by at least a week, they ate some special brownies.
Condition one was a control brownie, condition two was a brownie containing 20 mg of THC, and condition three was a brownie containing 20 mg of THC and 640 mg of CBD. Participants were assigned each condition in random order, separated by at least a week.
A side note on doses for those of you who, like me, are not totally weed literate. A dose of 20 mg of THC is about a third of what you might find in a typical joint these days (though it’s about double the THC content of a joint in the ‘70s – I believe the technical term is “doobie”). And 640 mg of CBD is a decent dose, as 5 mg per kilogram is what some folks start with to achieve therapeutic effects.
Both THC and CBD interact with the cytochrome p450 system in the liver. This matters when you’re ingesting them instead of smoking them because you have first-pass metabolism to contend with. And, because of that p450 inhibition, it’s possible that CBD might actually increase the amount of THC that gets into your bloodstream from the brownie, or gummy, or pizza sauce, or whatever.
Let’s get to the results, starting with blood THC concentration. It’s not subtle. With CBD on board the THC concentration rises higher faster, with roughly double the area under the curve.
And, unsurprisingly, the subjective experience correlated with those higher levels. Individuals rated the “drug effect” higher with the combo. But, interestingly, the “pleasant” drug effect didn’t change much, while the unpleasant effects were substantially higher. No mitigation of THC anxiety here – quite the opposite. CBD made the anxiety worse.
Cognitive effects were equally profound. Scores on a digit symbol substitution test and a paced serial addition task were all substantially worse when CBD was mixed with THC.
And for those of you who want some more objective measures, check out the heart rate. Despite the purported “calming” nature of CBD, heart rates were way higher when individuals were exposed to both chemicals.
The picture here is quite clear, though the mechanism is not. At least when talking edibles, CBD enhances the effects of THC, and not necessarily for the better. It may be that CBD is competing with some of the proteins that metabolize THC, thus prolonging its effects. CBD may also directly inhibit those enzymes. But whatever the case, I think we can safely say the myth that CBD makes the effects of THC more mild or more tolerable is busted.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale University’s Clinical and Translational Research Accelerator in New Haven, Conn.
A version of this article first appeared on Medscape.com.
Health plans get very poor scores for access to autoimmune drugs
Both public and private health plans score poorly when it comes to providing access to autoimmune medication, according to a report commissioned by the Autoimmune Association and Let My Doctors Decide, a national partnership of health care professionals. The analysis, published Jan. 26, found that 75% of insurers in the United States have policies that can limit coverage for Food and Drug Administration–approved medications for Crohn’s disease, lupus nephritis, multiple sclerosis, psoriasis, psoriatic arthritis, rheumatoid arthritis, and ulcerative colitis.
“Choice among health plans is a hallmark of the American health insurance system, yet this analysis shows that people living with autoimmune conditions have few, if any, coverage choices that do not involve significant to severe access restrictions,” the authors wrote.
The study looked at three common utilization management policies by health plans that can limit coverage of certain medications: step therapy, formulary/tier placement, and prior authorization. To compare health plans, researchers weighted these policies using a point system. Each medication indicated for each condition was given a score of 0-4 based on access restrictions in a health plan. If a plan used step therapy, it received one point, and requiring prior authorization added an additional point. They also added points based on where a drug appeared on a plan’s formulary. A lower total score meant fewer access barriers. The numbers were then added, and each health plan received a grade of A, B, C, or F based on their average score. The datasets and analysis were provided and performed by the data analytics firm MMIT.
Nearly 9 in 10 Medicare plans received a C or worse for coverage of medication received via mail order or the pharmacy. In commercial plans, the majority of plans scored Cs or Fs for six of the seven conditions, excluding lupus nephritis, where 67% of all commercial health plans scored a B for access to these medications.
Physician-administered medications tended to receive poorer coverage than drugs received via pharmacy. Across all conditions, 65% of Medicare Advantage plans scored an F for physician-administered medication access. For both psoriasis and multiple sclerosis, at least 80% of Medicare plans earned failing scores because of these restrictions. Coverage was poorer on both commercial and health exchange plans, where across all conditions, 83% achieved failing scores. Two exceptions were the Southern and Northern California PPO plans by the Kaiser Foundation Health Plan. Out of the largest 25 health plans in the United States, these two plans earned As in coverage for physician-administered medications across all seven autoimmune conditions.
The report shows “a growing disconnect between science and health insurance benefit designs that were developed in the 1960s and 1970s,” Kenneth Thorpe, PhD, of Emory University, Atlanta, said in an interview. Insurers originally designed these benefits to prevent excessive utilization in a population of mostly acutely ill patients, he said, whereas now, 90% of healthcare spending is linked to chronic conditions. For these patients, research shows that incentivizing patients to adhere to medications results in fewer hospitalizations and, therefore, more cost savings, Thorpe noted. These plans also do not consider that there is no average patient, he said, and healthcare providers should be able to match each patient to the best treatment option for them rather than trying out other less expensive medications first. “To the extent that physicians can have the flexibility to provide medications and treatments to patients that are going to have the best clinical response, that’s better outcomes at lower cost,” Dr. Thorpe said. While research shows heterogeneity in patient outcomes with different medication, “benefit designs from the past just don’t recognize that.”
Neither America’s Health Insurance Plans nor Pharmaceutical Care Management Association responded to a request for comment.
Quardricos Driskell, executive director of Let My Doctors Decide and vice president of government relations and public policy at the Autoimmune Association, hopes the study will spur action by policy makers and health plans to improve access to medications for the people who need them. Another larger point of the report is to “uphold the sanctity of protecting the doctor and patient relationship,” he said in an interview, adding “that decisions fundamentally need to be made not by insurance plans or middleman pharmacy benefit managers, but by the provider and patient.”
Mr. Driskell and Dr. Thorpe reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Both public and private health plans score poorly when it comes to providing access to autoimmune medication, according to a report commissioned by the Autoimmune Association and Let My Doctors Decide, a national partnership of health care professionals. The analysis, published Jan. 26, found that 75% of insurers in the United States have policies that can limit coverage for Food and Drug Administration–approved medications for Crohn’s disease, lupus nephritis, multiple sclerosis, psoriasis, psoriatic arthritis, rheumatoid arthritis, and ulcerative colitis.
“Choice among health plans is a hallmark of the American health insurance system, yet this analysis shows that people living with autoimmune conditions have few, if any, coverage choices that do not involve significant to severe access restrictions,” the authors wrote.
The study looked at three common utilization management policies by health plans that can limit coverage of certain medications: step therapy, formulary/tier placement, and prior authorization. To compare health plans, researchers weighted these policies using a point system. Each medication indicated for each condition was given a score of 0-4 based on access restrictions in a health plan. If a plan used step therapy, it received one point, and requiring prior authorization added an additional point. They also added points based on where a drug appeared on a plan’s formulary. A lower total score meant fewer access barriers. The numbers were then added, and each health plan received a grade of A, B, C, or F based on their average score. The datasets and analysis were provided and performed by the data analytics firm MMIT.
Nearly 9 in 10 Medicare plans received a C or worse for coverage of medication received via mail order or the pharmacy. In commercial plans, the majority of plans scored Cs or Fs for six of the seven conditions, excluding lupus nephritis, where 67% of all commercial health plans scored a B for access to these medications.
Physician-administered medications tended to receive poorer coverage than drugs received via pharmacy. Across all conditions, 65% of Medicare Advantage plans scored an F for physician-administered medication access. For both psoriasis and multiple sclerosis, at least 80% of Medicare plans earned failing scores because of these restrictions. Coverage was poorer on both commercial and health exchange plans, where across all conditions, 83% achieved failing scores. Two exceptions were the Southern and Northern California PPO plans by the Kaiser Foundation Health Plan. Out of the largest 25 health plans in the United States, these two plans earned As in coverage for physician-administered medications across all seven autoimmune conditions.
The report shows “a growing disconnect between science and health insurance benefit designs that were developed in the 1960s and 1970s,” Kenneth Thorpe, PhD, of Emory University, Atlanta, said in an interview. Insurers originally designed these benefits to prevent excessive utilization in a population of mostly acutely ill patients, he said, whereas now, 90% of healthcare spending is linked to chronic conditions. For these patients, research shows that incentivizing patients to adhere to medications results in fewer hospitalizations and, therefore, more cost savings, Thorpe noted. These plans also do not consider that there is no average patient, he said, and healthcare providers should be able to match each patient to the best treatment option for them rather than trying out other less expensive medications first. “To the extent that physicians can have the flexibility to provide medications and treatments to patients that are going to have the best clinical response, that’s better outcomes at lower cost,” Dr. Thorpe said. While research shows heterogeneity in patient outcomes with different medication, “benefit designs from the past just don’t recognize that.”
Neither America’s Health Insurance Plans nor Pharmaceutical Care Management Association responded to a request for comment.
Quardricos Driskell, executive director of Let My Doctors Decide and vice president of government relations and public policy at the Autoimmune Association, hopes the study will spur action by policy makers and health plans to improve access to medications for the people who need them. Another larger point of the report is to “uphold the sanctity of protecting the doctor and patient relationship,” he said in an interview, adding “that decisions fundamentally need to be made not by insurance plans or middleman pharmacy benefit managers, but by the provider and patient.”
Mr. Driskell and Dr. Thorpe reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Both public and private health plans score poorly when it comes to providing access to autoimmune medication, according to a report commissioned by the Autoimmune Association and Let My Doctors Decide, a national partnership of health care professionals. The analysis, published Jan. 26, found that 75% of insurers in the United States have policies that can limit coverage for Food and Drug Administration–approved medications for Crohn’s disease, lupus nephritis, multiple sclerosis, psoriasis, psoriatic arthritis, rheumatoid arthritis, and ulcerative colitis.
“Choice among health plans is a hallmark of the American health insurance system, yet this analysis shows that people living with autoimmune conditions have few, if any, coverage choices that do not involve significant to severe access restrictions,” the authors wrote.
The study looked at three common utilization management policies by health plans that can limit coverage of certain medications: step therapy, formulary/tier placement, and prior authorization. To compare health plans, researchers weighted these policies using a point system. Each medication indicated for each condition was given a score of 0-4 based on access restrictions in a health plan. If a plan used step therapy, it received one point, and requiring prior authorization added an additional point. They also added points based on where a drug appeared on a plan’s formulary. A lower total score meant fewer access barriers. The numbers were then added, and each health plan received a grade of A, B, C, or F based on their average score. The datasets and analysis were provided and performed by the data analytics firm MMIT.
Nearly 9 in 10 Medicare plans received a C or worse for coverage of medication received via mail order or the pharmacy. In commercial plans, the majority of plans scored Cs or Fs for six of the seven conditions, excluding lupus nephritis, where 67% of all commercial health plans scored a B for access to these medications.
Physician-administered medications tended to receive poorer coverage than drugs received via pharmacy. Across all conditions, 65% of Medicare Advantage plans scored an F for physician-administered medication access. For both psoriasis and multiple sclerosis, at least 80% of Medicare plans earned failing scores because of these restrictions. Coverage was poorer on both commercial and health exchange plans, where across all conditions, 83% achieved failing scores. Two exceptions were the Southern and Northern California PPO plans by the Kaiser Foundation Health Plan. Out of the largest 25 health plans in the United States, these two plans earned As in coverage for physician-administered medications across all seven autoimmune conditions.
The report shows “a growing disconnect between science and health insurance benefit designs that were developed in the 1960s and 1970s,” Kenneth Thorpe, PhD, of Emory University, Atlanta, said in an interview. Insurers originally designed these benefits to prevent excessive utilization in a population of mostly acutely ill patients, he said, whereas now, 90% of healthcare spending is linked to chronic conditions. For these patients, research shows that incentivizing patients to adhere to medications results in fewer hospitalizations and, therefore, more cost savings, Thorpe noted. These plans also do not consider that there is no average patient, he said, and healthcare providers should be able to match each patient to the best treatment option for them rather than trying out other less expensive medications first. “To the extent that physicians can have the flexibility to provide medications and treatments to patients that are going to have the best clinical response, that’s better outcomes at lower cost,” Dr. Thorpe said. While research shows heterogeneity in patient outcomes with different medication, “benefit designs from the past just don’t recognize that.”
Neither America’s Health Insurance Plans nor Pharmaceutical Care Management Association responded to a request for comment.
Quardricos Driskell, executive director of Let My Doctors Decide and vice president of government relations and public policy at the Autoimmune Association, hopes the study will spur action by policy makers and health plans to improve access to medications for the people who need them. Another larger point of the report is to “uphold the sanctity of protecting the doctor and patient relationship,” he said in an interview, adding “that decisions fundamentally need to be made not by insurance plans or middleman pharmacy benefit managers, but by the provider and patient.”
Mr. Driskell and Dr. Thorpe reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Antenatal corticosteroids: Fresh answers to old questions
Giving corticosteroids to pregnant women at risk for preterm birth before 34 weeks of gestational age has been the standard of care since the 1990s, but a few scenarios for their use remain up for debate. Two studies presented this week at the 2023 meeting sponsored by the Society for Maternal–Fetal Medicine provided some fresh insight into the practice that could help clinicians better manage pregnant patients.
Neurodevelopmental outcomes in late preterm
First, should antenatal corticosteroids (ACS) be given to mothers who present with late preterm labor, defined as 34-36 weeks’ gestational age?
A landmark randomized clinical trial published in 2016 demonstrated that use of ACS in mothers in late preterm labor reduced severe respiratory complications. That practice has largely been adopted by clinicians. The only downside, according to the researchers, was that infants whose mothers received steroid therapy were more likely to develop hypoglycemia. The condition is self-limiting, but studies have raised concern about the potential long-term risk of neurocognitive or psychological outcomes in infants with hypoglycemia.
Cynthia Gyamfi-Bannerman, MD, MSc, endowed chair and professor of obstetrics, gynecology, and reproductive sciences at the University of California, San Diego, led the 2016 study. Her team was unable to secure funding for their originally planned follow-up study of the infants 2 years later. But once the American College of Obstetricians and Gynecologists endorsed the practice and more women received ACS in the late preterm period, Dr. Gyamfi-Bannerman and her colleagues felt the need to “follow up the infants just to see what the outcomes are from a neurodevelopmental standpoint,” she said.
Dr. Gyamfi-Bannerman and colleagues recruited children older than age 6 from the original trial whose parents were willing to have them participate in a follow-up study. A total of 949 from the initial 2,831 cohort completed cognitive testing and received assessments for cerebral palsy, social impairment within the autism spectrum, and behavioral and emotional problems.
At the SMFM conference, Dr. Gyamfi-Bannerman reported no differences in the primary outcome of cognitive function between those whose mothers had received a single course of betamethasone and those who did not, or any differences in rates of the other outcomes.
Kathy Zhang-Rutledge, MD, a maternal-fetal medicine specialist who practices with Obstetrix Maternal Fetal Medicine Group of Houston, part of Pediatrix Medical Group, said she was glad to see a study that addressed the potential long-term adverse events associated with ACS in the late preterm period.
“Having this pretty large study – with really good neurological testing results – should help reassure clinicians that this is something they should consider adopting in their practice,” Dr. Zhang-Rutledge said.
Are boosters better?
The second unresolved question was if a repeat course of ACS should be administered when a woman at risk for preterm birth receives a course of steroids but does not deliver in the following 7 days.
Any benefits to the initial course of ACS wear off after a week. As a result, clinicians often give booster courses 7 days after the first dose if the infant is likely to be delivered in the following week. A 2009 study showed this approach may protect infants from respiratory problems, but data on long-term outcomes have been weak.
ACOG guidelines say to “consider” a booster dose in women who are less than 34 weeks’ gestation at risk for preterm delivery within 7 days.
The exception is when the mother already has experienced preterm prelabor rupture of membranes (PPROM), because ACS may increase the risk for infection for both mother and child. ACOG doesn’t take a stand on use of booster doses for PPROM, citing a lack of data to show that potential benefits outweigh the potential risks of this approach.
A recent multicenter, double-blinded, randomized clinical trial attempted to fill that void in knowledge. Between 2016 and 2022, 194 women with PPROM and gestational age less than 32 weeks who had received an initial ACS course at least 7 days prior to randomization received a booster course of ACS or saline placebo.
“Our primary outcome was designed to be like the prior rescue study (in 2009) that we did with patients with intact membranes,” said Andrew Combs, MD, PhD, a maternal-fetal medicine specialist at Pediatrix Medical Group in Sunrise, Fla., who participated in the earlier study. “It was a composite of neonatal morbidity that was any one of a variety of outcomes including respiratory distress syndrome, intraventricular hemorrhage, necrotizing enterocolitis, and neonatal death.”
The primary outcome occurred in 64% of women who received booster ACS and 66% with placebo (odds ratio, 0.82; 95% confidence interval, 0.43-1.57), according to Dr. Combs, who presented the findings at SMFM.
Although the study was not powered to detect significant differences in specific outcomes, the rate of neonatal sepsis was not higher in the ACS group, suggesting that ACS may be safe if membranes have ruptured, the researchers reported. But because the booster course of ACS did not prevent respiratory morbidity, clinicians may wonder what to do with the findings.
Niraj Chavan, MD, an associate professor in the department of obstetrics, gynecology, and women’s health at Saint Louis University, said he was unsure how the study would affect clinical practice.
The relatively small sample number of patients prevented analysis of specific outcomes and subgroup analyses of important variables such as race, ethnicity, gestational age, and other comorbid conditions in the mothers, he said. So clinicians still must weigh potential risks and benefits on a case-by-case basis.
“You have to think about it in buckets,” he said, “One is conditions that would increase the risk for neonatal morbidity. The other is the risk for infection, both for the mom and the baby.”
But for Dr. Combs, the interpretation of the findings was simpler: “We concluded that there’s no indication to give a booster course of steroids after a week has elapsed in patients with ruptured membranes.”
The study presented by Dr. Gyamfi-Bannerman was funded by the National Institute of Child Health and Human Development. The study presented by Dr. Combs was funded by MEDNAX Center for Research, Education, and Quality, which in 2022 was renamed Pediatrix Center for Research, Education,and Quality. Dr. Combs is an employee of Pediatrix Medical Group but has no conflicts of interest. Dr. Gyamfi-Bannerman, Dr. Zhang-Rutledge, and Dr. Chavan report no relevant financial relationships.
Ann Thomas is a pediatrician and epidemiologist in Portland, Ore.
A version of this article originally appeared on Medscape.com.
Giving corticosteroids to pregnant women at risk for preterm birth before 34 weeks of gestational age has been the standard of care since the 1990s, but a few scenarios for their use remain up for debate. Two studies presented this week at the 2023 meeting sponsored by the Society for Maternal–Fetal Medicine provided some fresh insight into the practice that could help clinicians better manage pregnant patients.
Neurodevelopmental outcomes in late preterm
First, should antenatal corticosteroids (ACS) be given to mothers who present with late preterm labor, defined as 34-36 weeks’ gestational age?
A landmark randomized clinical trial published in 2016 demonstrated that use of ACS in mothers in late preterm labor reduced severe respiratory complications. That practice has largely been adopted by clinicians. The only downside, according to the researchers, was that infants whose mothers received steroid therapy were more likely to develop hypoglycemia. The condition is self-limiting, but studies have raised concern about the potential long-term risk of neurocognitive or psychological outcomes in infants with hypoglycemia.
Cynthia Gyamfi-Bannerman, MD, MSc, endowed chair and professor of obstetrics, gynecology, and reproductive sciences at the University of California, San Diego, led the 2016 study. Her team was unable to secure funding for their originally planned follow-up study of the infants 2 years later. But once the American College of Obstetricians and Gynecologists endorsed the practice and more women received ACS in the late preterm period, Dr. Gyamfi-Bannerman and her colleagues felt the need to “follow up the infants just to see what the outcomes are from a neurodevelopmental standpoint,” she said.
Dr. Gyamfi-Bannerman and colleagues recruited children older than age 6 from the original trial whose parents were willing to have them participate in a follow-up study. A total of 949 from the initial 2,831 cohort completed cognitive testing and received assessments for cerebral palsy, social impairment within the autism spectrum, and behavioral and emotional problems.
At the SMFM conference, Dr. Gyamfi-Bannerman reported no differences in the primary outcome of cognitive function between those whose mothers had received a single course of betamethasone and those who did not, or any differences in rates of the other outcomes.
Kathy Zhang-Rutledge, MD, a maternal-fetal medicine specialist who practices with Obstetrix Maternal Fetal Medicine Group of Houston, part of Pediatrix Medical Group, said she was glad to see a study that addressed the potential long-term adverse events associated with ACS in the late preterm period.
“Having this pretty large study – with really good neurological testing results – should help reassure clinicians that this is something they should consider adopting in their practice,” Dr. Zhang-Rutledge said.
Are boosters better?
The second unresolved question was if a repeat course of ACS should be administered when a woman at risk for preterm birth receives a course of steroids but does not deliver in the following 7 days.
Any benefits to the initial course of ACS wear off after a week. As a result, clinicians often give booster courses 7 days after the first dose if the infant is likely to be delivered in the following week. A 2009 study showed this approach may protect infants from respiratory problems, but data on long-term outcomes have been weak.
ACOG guidelines say to “consider” a booster dose in women who are less than 34 weeks’ gestation at risk for preterm delivery within 7 days.
The exception is when the mother already has experienced preterm prelabor rupture of membranes (PPROM), because ACS may increase the risk for infection for both mother and child. ACOG doesn’t take a stand on use of booster doses for PPROM, citing a lack of data to show that potential benefits outweigh the potential risks of this approach.
A recent multicenter, double-blinded, randomized clinical trial attempted to fill that void in knowledge. Between 2016 and 2022, 194 women with PPROM and gestational age less than 32 weeks who had received an initial ACS course at least 7 days prior to randomization received a booster course of ACS or saline placebo.
“Our primary outcome was designed to be like the prior rescue study (in 2009) that we did with patients with intact membranes,” said Andrew Combs, MD, PhD, a maternal-fetal medicine specialist at Pediatrix Medical Group in Sunrise, Fla., who participated in the earlier study. “It was a composite of neonatal morbidity that was any one of a variety of outcomes including respiratory distress syndrome, intraventricular hemorrhage, necrotizing enterocolitis, and neonatal death.”
The primary outcome occurred in 64% of women who received booster ACS and 66% with placebo (odds ratio, 0.82; 95% confidence interval, 0.43-1.57), according to Dr. Combs, who presented the findings at SMFM.
Although the study was not powered to detect significant differences in specific outcomes, the rate of neonatal sepsis was not higher in the ACS group, suggesting that ACS may be safe if membranes have ruptured, the researchers reported. But because the booster course of ACS did not prevent respiratory morbidity, clinicians may wonder what to do with the findings.
Niraj Chavan, MD, an associate professor in the department of obstetrics, gynecology, and women’s health at Saint Louis University, said he was unsure how the study would affect clinical practice.
The relatively small sample number of patients prevented analysis of specific outcomes and subgroup analyses of important variables such as race, ethnicity, gestational age, and other comorbid conditions in the mothers, he said. So clinicians still must weigh potential risks and benefits on a case-by-case basis.
“You have to think about it in buckets,” he said, “One is conditions that would increase the risk for neonatal morbidity. The other is the risk for infection, both for the mom and the baby.”
But for Dr. Combs, the interpretation of the findings was simpler: “We concluded that there’s no indication to give a booster course of steroids after a week has elapsed in patients with ruptured membranes.”
The study presented by Dr. Gyamfi-Bannerman was funded by the National Institute of Child Health and Human Development. The study presented by Dr. Combs was funded by MEDNAX Center for Research, Education, and Quality, which in 2022 was renamed Pediatrix Center for Research, Education,and Quality. Dr. Combs is an employee of Pediatrix Medical Group but has no conflicts of interest. Dr. Gyamfi-Bannerman, Dr. Zhang-Rutledge, and Dr. Chavan report no relevant financial relationships.
Ann Thomas is a pediatrician and epidemiologist in Portland, Ore.
A version of this article originally appeared on Medscape.com.
Giving corticosteroids to pregnant women at risk for preterm birth before 34 weeks of gestational age has been the standard of care since the 1990s, but a few scenarios for their use remain up for debate. Two studies presented this week at the 2023 meeting sponsored by the Society for Maternal–Fetal Medicine provided some fresh insight into the practice that could help clinicians better manage pregnant patients.
Neurodevelopmental outcomes in late preterm
First, should antenatal corticosteroids (ACS) be given to mothers who present with late preterm labor, defined as 34-36 weeks’ gestational age?
A landmark randomized clinical trial published in 2016 demonstrated that use of ACS in mothers in late preterm labor reduced severe respiratory complications. That practice has largely been adopted by clinicians. The only downside, according to the researchers, was that infants whose mothers received steroid therapy were more likely to develop hypoglycemia. The condition is self-limiting, but studies have raised concern about the potential long-term risk of neurocognitive or psychological outcomes in infants with hypoglycemia.
Cynthia Gyamfi-Bannerman, MD, MSc, endowed chair and professor of obstetrics, gynecology, and reproductive sciences at the University of California, San Diego, led the 2016 study. Her team was unable to secure funding for their originally planned follow-up study of the infants 2 years later. But once the American College of Obstetricians and Gynecologists endorsed the practice and more women received ACS in the late preterm period, Dr. Gyamfi-Bannerman and her colleagues felt the need to “follow up the infants just to see what the outcomes are from a neurodevelopmental standpoint,” she said.
Dr. Gyamfi-Bannerman and colleagues recruited children older than age 6 from the original trial whose parents were willing to have them participate in a follow-up study. A total of 949 from the initial 2,831 cohort completed cognitive testing and received assessments for cerebral palsy, social impairment within the autism spectrum, and behavioral and emotional problems.
At the SMFM conference, Dr. Gyamfi-Bannerman reported no differences in the primary outcome of cognitive function between those whose mothers had received a single course of betamethasone and those who did not, or any differences in rates of the other outcomes.
Kathy Zhang-Rutledge, MD, a maternal-fetal medicine specialist who practices with Obstetrix Maternal Fetal Medicine Group of Houston, part of Pediatrix Medical Group, said she was glad to see a study that addressed the potential long-term adverse events associated with ACS in the late preterm period.
“Having this pretty large study – with really good neurological testing results – should help reassure clinicians that this is something they should consider adopting in their practice,” Dr. Zhang-Rutledge said.
Are boosters better?
The second unresolved question was if a repeat course of ACS should be administered when a woman at risk for preterm birth receives a course of steroids but does not deliver in the following 7 days.
Any benefits to the initial course of ACS wear off after a week. As a result, clinicians often give booster courses 7 days after the first dose if the infant is likely to be delivered in the following week. A 2009 study showed this approach may protect infants from respiratory problems, but data on long-term outcomes have been weak.
ACOG guidelines say to “consider” a booster dose in women who are less than 34 weeks’ gestation at risk for preterm delivery within 7 days.
The exception is when the mother already has experienced preterm prelabor rupture of membranes (PPROM), because ACS may increase the risk for infection for both mother and child. ACOG doesn’t take a stand on use of booster doses for PPROM, citing a lack of data to show that potential benefits outweigh the potential risks of this approach.
A recent multicenter, double-blinded, randomized clinical trial attempted to fill that void in knowledge. Between 2016 and 2022, 194 women with PPROM and gestational age less than 32 weeks who had received an initial ACS course at least 7 days prior to randomization received a booster course of ACS or saline placebo.
“Our primary outcome was designed to be like the prior rescue study (in 2009) that we did with patients with intact membranes,” said Andrew Combs, MD, PhD, a maternal-fetal medicine specialist at Pediatrix Medical Group in Sunrise, Fla., who participated in the earlier study. “It was a composite of neonatal morbidity that was any one of a variety of outcomes including respiratory distress syndrome, intraventricular hemorrhage, necrotizing enterocolitis, and neonatal death.”
The primary outcome occurred in 64% of women who received booster ACS and 66% with placebo (odds ratio, 0.82; 95% confidence interval, 0.43-1.57), according to Dr. Combs, who presented the findings at SMFM.
Although the study was not powered to detect significant differences in specific outcomes, the rate of neonatal sepsis was not higher in the ACS group, suggesting that ACS may be safe if membranes have ruptured, the researchers reported. But because the booster course of ACS did not prevent respiratory morbidity, clinicians may wonder what to do with the findings.
Niraj Chavan, MD, an associate professor in the department of obstetrics, gynecology, and women’s health at Saint Louis University, said he was unsure how the study would affect clinical practice.
The relatively small sample number of patients prevented analysis of specific outcomes and subgroup analyses of important variables such as race, ethnicity, gestational age, and other comorbid conditions in the mothers, he said. So clinicians still must weigh potential risks and benefits on a case-by-case basis.
“You have to think about it in buckets,” he said, “One is conditions that would increase the risk for neonatal morbidity. The other is the risk for infection, both for the mom and the baby.”
But for Dr. Combs, the interpretation of the findings was simpler: “We concluded that there’s no indication to give a booster course of steroids after a week has elapsed in patients with ruptured membranes.”
The study presented by Dr. Gyamfi-Bannerman was funded by the National Institute of Child Health and Human Development. The study presented by Dr. Combs was funded by MEDNAX Center for Research, Education, and Quality, which in 2022 was renamed Pediatrix Center for Research, Education,and Quality. Dr. Combs is an employee of Pediatrix Medical Group but has no conflicts of interest. Dr. Gyamfi-Bannerman, Dr. Zhang-Rutledge, and Dr. Chavan report no relevant financial relationships.
Ann Thomas is a pediatrician and epidemiologist in Portland, Ore.
A version of this article originally appeared on Medscape.com.
FROM THE PREGNANCY MEETING
UnitedHealthcare tried to deny coverage to a chronically ill patient. He fought back, exposing the insurer’s inner workings.
In May 2021, a nurse at UnitedHealthcare called a colleague to share some welcome news about a problem the two had been grappling with for weeks.
United provided the health insurance plan for students at Penn State University. It was a large and potentially lucrative account: lots of young, healthy students paying premiums in, not too many huge medical reimbursements going out.
But Christopher McNaughton suffered from a crippling case of ulcerative colitis – an ailment that caused him to develop severe arthritis, debilitating diarrhea, numbing fatigue, and life-threatening blood clots. His medical bills were running nearly $2 million a year.
United had flagged Mr. McNaughton’s case as a “high dollar account,” and the company was reviewing whether it needed to keep paying for the expensive cocktail of drugs crafted by a Mayo Clinic specialist that had brought Mr. McNaughton’s disease under control after he’d been through years of misery.
On the 2021 phone call, which was recorded by the company, nurse Victoria Kavanaugh told her colleague that a doctor contracted by United to review the case had concluded that Mr. McNaughton’s treatment was “not medically necessary.” Her colleague, Dave Opperman, reacted to the news with a long laugh.
“I knew that was coming,” said Mr. Opperman, who heads up a United subsidiary that brokered the health insurance contract between United and Penn State. “I did too,” Ms. Kavanaugh replied.
Mr. Opperman then complained about Mr. McNaughton’s mother, whom he referred to as “this woman,” for “screaming and yelling” and “throwing tantrums” during calls with United.
The pair agreed that any appeal of the United doctor’s denial of the treatment would be a waste of the family’s time and money.
“We’re still gonna say no,” Mr. Opperman said.
More than 200 million Americans are covered by private health insurance. But data from state and federal regulators shows that insurers reject about 1 in 7 claims for treatment. Many people, faced with fighting insurance companies, simply give up: One study found that Americans file formal appeals on only 0.1% of claims denied by insurers under the Affordable Care Act.
Insurers have wide discretion in crafting what is covered by their policies, beyond some basic services mandated by federal and state law. They often deny claims for services that they deem not “medically necessary.”
When United refused to pay for Mr. McNaughton’s treatment for that reason, his family did something unusual. They fought back with a lawsuit, which uncovered a trove of materials, including internal emails and tape-recorded exchanges among company employees. Those records offer an extraordinary behind-the-scenes look at how one of America’s leading health care insurers relentlessly fought to reduce spending on care, even as its profits rose to record levels.
As United reviewed Mr. McNaughton’s treatment, he and his family were often in the dark about what was happening or their rights. Meanwhile, United employees misrepresented critical findings and ignored warnings from doctors about the risks of altering Mr. McNaughton’s drug plan.
At one point, court records show, United inaccurately reported to Penn State and the family that Mr. McNaughton’s doctor had agreed to lower the doses of his medication. Another time, a doctor paid by United concluded that denying payments for Mr. McNaughton’s treatment could put his health at risk, but the company buried his report and did not consider its findings. The insurer did, however, consider a report submitted by a company doctor who rubber-stamped the recommendation of a United nurse to reject paying for the treatment.
United declined to answer specific questions about the case, even after Mr. McNaughton signed a release provided by the insurer to allow it to discuss details of his interactions with the company. United noted that it ultimately paid for all of Mr. McNaughton’s treatments. In a written response, United spokesperson Maria Gordon Shydlo wrote that the company’s guiding concern was Mr. McNaughton’s well-being.
“Mr. McNaughton’s treatment involves medication dosages that far exceed [Food and Drug Administration] guidelines,” the statement said. “In cases like this, we review treatment plans based on current clinical guidelines to help ensure patient safety.”
But the records reviewed by ProPublica show that United had another, equally urgent goal in dealing with Mr. McNaughton. In emails, officials calculated what Mr. McNaughton was costing them to keep his crippling disease at bay and how much they would save if they forced him to undergo a cheaper treatment that had already failed him. As the family pressed the company to back down, first through Penn State and then through a lawsuit, the United officials handling the case bristled.
“This is just unbelievable,” Ms. Kavanaugh said of Mr. McNaughton’s family in one call to discuss his case. ”They’re just really pushing the envelope, and I’m surprised, like I don’t even know what to say.”
The same meal every day
Now 31, Mr. McNaughton grew up in State College, Pa., just blocks from the Penn State campus. Both of his parents are faculty members at the university.
In the winter of 2014, Mr. McNaughton was halfway through his junior year at Bard College in New York. At 6 feet, 4 inches tall, he was a guard on the basketball team and had started most of the team’s games since the start of his sophomore year. He was majoring in psychology.
When Mr. McNaughton returned to school after the winter holiday break, he started to experience frequent bouts of bloody diarrhea. After just a few days on campus, he went home to State College, where doctors diagnosed him with a severe case of ulcerative colitis.
A chronic inflammatory bowel disease that causes swelling and ulcers in the digestive tract, ulcerative colitis has no cure, and ongoing treatment is needed to alleviate symptoms and prevent serious health complications. The majority of cases produce mild to moderate symptoms. Mr. McNaughton’s case was severe.
Treatments for ulcerative colitis include steroids and special drugs known as biologics that work to reduce inflammation in the large intestine.
Mr. McNaughton, however, failed to get meaningful relief from the drugs his doctors initially prescribed. He was experiencing bloody diarrhea up to 20 times a day, with such severe stomach pain that he spent much of his day curled up on a couch. He had little appetite and lost 50 pounds. Severe anemia left him fatigued. He suffered from other conditions related to his colitis, including crippling arthritis. He was hospitalized several times to treat dangerous blood clots.
For 2 years, in an effort to help alleviate his symptoms, he ate the same meals every day: Rice Chex cereal and scrambled eggs for breakfast, a cup of white rice with plain chicken breast for lunch, and a similar meal for dinner, occasionally swapping in tilapia.
His hometown doctors referred him to a specialist at the University of Pittsburgh, who tried unsuccessfully to bring his disease under control. That doctor ended up referring Mr. McNaughton to Edward V. Loftus Jr., MD, at the Mayo Clinic in Rochester, Minn., which has been ranked as the best gastroenterology hospital in the country every year since 1990 by U.S. News & World Report.
For his first visit with Dr. Loftus in May 2015, Mr. McNaughton and his mother, Janice Light, charted hospitals along the 900-mile drive from Pennsylvania to Minnesota in case they needed medical help along the way.
Mornings were the hardest. Mr. McNaughton often spent several hours in the bathroom at the start of the day. To prepare for his meeting with Dr. Loftus, he set his alarm for 3:30 a.m. so he could be ready for the 7:30 a.m. appointment. Even with that preparation, he had to stop twice to use a bathroom on the 5-minute walk from the hotel to the clinic. When they met, Dr. Loftus looked at Mr. McNaughton and told him that he appeared incapacitated. It was, he told the student, as if Mr. McNaughton were chained to the bathroom, with no outside life. He had not been able to return to school and spent most days indoors, managing his symptoms as best he could.
Mr. McNaughton had tried a number of medications by this point, none of which worked. This pattern would repeat itself during the first couple of years that Dr. Loftus treated him.
In addition to trying to find a treatment that would bring Mr. McNaughton’s colitis into remission, Dr. Loftus wanted to wean him off the steroid prednisone, which he had been taking since his initial diagnosis in 2014. The drug is commonly prescribed to colitis patients to control inflammation, but prolonged use can lead to severe side effects including cataracts, osteoporosis, increased risk of infection, and fatigue. Mr. McNaughton also experienced “moon face,” a side effect caused by the shifting of fat deposits that results in the face becoming puffy and rounder.
In 2018, Dr. Loftus and Mr. McNaughton decided to try an unusual regimen. Many patients with inflammatory bowel diseases such as colitis take a single biologic drug as treatment. Whereas traditional drugs are chemically synthesized, biologics are manufactured in living systems, such as plant or animal cells. A year’s supply of an individual biologic drug can cost up to $500,000. They are often given through infusions in a medical facility, which adds to the cost.
Mr. McNaughton had tried individual biologics, and then two in combination, without much success. He and Dr. Loftus then agreed to try two biologic drugs together at doses well above those recommended by the Food and Drug Administration. The federal Agency for Healthcare Research and Quality estimates one in five prescriptions written today are for off-label uses.
There are drawbacks to the practice. Since some uses and doses of particular drugs have not been extensively studied, the risks and efficacy of using them off-label are not well known. Also, some drug manufacturers have improperly pushed off-label usage of their products to boost sales despite little or no evidence to support their use in those situations. Like many leading experts and researchers in his field, Dr. Loftus has been paid to do consulting related to the biologic drugs taken by Mr. McNaughton. The payments related to those drugs have ranged from a total of $1,440 in 2020 to $51,235 in 2018. Dr. Loftus said much of his work with pharmaceutical companies was related to conducting clinical trials on new drugs.
In cases of off-label prescribing, patients are depending upon their doctors’ expertise and experience with the drug. “In this case, I was comfortable that the potential benefits to Chris outweighed the risks,” Dr. Loftus said.
There was evidence that the treatment plan for Mr. McNaughton might work, including studies that had found dual biologic therapy to be efficacious and safe. The two drugs he takes, Entyvio and Remicade, have the same purpose – to reduce inflammation in the large intestine – but each works differently in the body. Remicade, marketed by Janssen Biotech, targets a protein that causes inflammation. Entyvio, made by Takeda Pharmaceuticals, works by preventing an excess of white blood cells from entering into the gastrointestinal tract.
As for any suggestion by United doctors that his treatment plan for Mr. McNaughton was out of bounds or dangerous, Dr. Loftus said “my treatment of Chris was not clinically inappropriate – as was shown by Chris’ positive outcome.”
The unusual high-dose combination of two biologic drugs produced a remarkable change in Mr. McNaughton. He no longer had blood in his stool, and his trips to the bathroom were cut from 20 times a day to 3 or 4. He was able to eat different foods and put on weight. He had more energy. He tapered off prednisone.
“If you told me in 2015 that I would be living like this, I would have asked where do I sign up,” Mr. McNaughton said of the change he experienced with the new drug regimen.
When he first started the new treatment, Mr. McNaughton was covered under his family’s plan, and all his bills were paid. Mr. McNaughton enrolled at the university in 2020. Before switching to United’s plan for students, Mr. McNaughton and his parents consulted with a health advocacy service offered to faculty members. A benefits specialist assured them the drugs taken by Mr. McNaughton would be covered by United.
Mr. McNaughton joined the student plan in July 2020, and his infusions that month and the following month were paid for by United. In September, the insurer indicated payment on his claims was “pending,” something it did for his other claims that came in during the rest of the year.
Mr. McNaughton and his family were worried. They called United to make sure there wasn’t a problem; the insurer told them, they said, that it only needed to check his medical records. When the family called again, United told them it had the documentation needed, they said. United, in a court filing last year, said it received two calls from the family and each time indicated that all of the necessary medical records had not yet been received.
In January 2021, Mr. McNaughton received a new explanation of benefits for the prior months. All of the claims for his care, beginning in September, were no longer “pending.” They were stamped “DENIED.” The total outstanding bill for his treatment was $807,086.
When Mr. McNaughton’s mother reached a United customer service representative the next day to ask why bills that had been paid in the summer were being denied for the fall, the representative told her the account was being reviewed because of “a high dollar amount on the claims,” according to a recording of the call.
Misrepresentations
With United refusing to pay, the family was terrified of being stuck with medical bills that would bankrupt them and deprive Mr. McNaughton of treatment that they considered miraculous.
They turned to Penn State for help. Ms. Light and Mr. McNaughton’s father, David McNaughton, hoped their position as faculty members would make the school more willing to intervene on their behalf.
“After more than 30 years on faculty, my husband and I know that this is not how Penn State would want its students to be treated,” Ms. Light wrote to a school official in February 2021.
In response to questions from ProPublica, Penn State spokesperson Lisa Powers wrote that “supporting the health and well-being of our students is always of primary importance” and that “our hearts go out to any student and family impacted by a serious medical condition.” The university, she wrote, does “not comment on students’ individual circumstances or disclose information from their records.” Mr. McNaughton offered to grant Penn State whatever permissions it needed to speak about his case with ProPublica. The school, however, wrote that it would not comment “even if confidentiality has been waived.”
The family appealed to school administrators. Because the effectiveness of biologics wanes in some patients if doses are skipped, Mr. McNaughton and his parents were worried about even a delay in treatment. His doctor wrote that if he missed scheduled infusions of the drugs, there was “a high likelihood they would no longer be effective.”
During a conference call arranged by Penn State officials on March 5, 2021, United agreed to pay for Mr. McNaughton’s care through the end of the plan year that August. Penn State immediately notified the family of the “wonderful news” while also apologizing for “the stress this has caused Chris and your family.”
Behind the scenes, Mr. McNaughton’s review had “gone all the way to the top” at United’s student health plan division, Ms. Kavanaugh, the nurse, said in a recorded conversation.
The family’s relief was short-lived. A month later, United started another review of Mr. McNaughton’s care, overseen by Ms. Kavanaugh, to determine if it would pay for the treatment in the upcoming plan year.
The nurse sent the Mr. McNaughton case to a company called Medical Review Institute of America. Insurers often turn to companies like MRIoA to review coverage decisions involving expensive treatments or specialized care.
Ms. Kavanaugh, who was assigned to a special investigations unit at United, let her feelings about the matter be known in a recorded telephone call with a representative of MRIoA.
“This school apparently is a big client of ours,” she said. She then shared her opinion of Mr. McNaughton’s treatment. “Really this is a case of a kid who’s getting a drug way too much, like too much of a dose,” Ms. Kavanaugh said. She said it was “insane that they would even think that this is reasonable” and “to be honest with you, they’re awfully pushy considering that we are paying through the end of this school year.”
On a call with an outside contractor, the United nurse claimed Mr. McNaughton was on a higher dose of medication than the FDA approved, which is a common practice.
MRIoA sent the case to Vikas Pabby, MD, a gastroenterologist at UCLA Health and a professor at the university’s medical school. His May 2021 review of Mr. McNaughton’s case was just one of more than 300 Dr. Pabby did for MRIoA that month, for which he was paid $23,000 in total, according to a log of his work produced in the lawsuit.
In a May 4, 2021, report, Dr. Pabby concluded Mr. McNaughton’s treatment was not medically necessary, because United’s policies for the two drugs taken by Mr. McNaughton did not support using them in combination.
Insurers spell out what services they cover in plan policies, lengthy documents that can be confusing and difficult to understand. Many policies, such as Mr. McNaughton’s, contain a provision that treatments and procedures must be “medically necessary” in order to be covered. The definition of medically necessary differs by plan. Some don’t even define the term. Mr. McNaughton’s policy contains a five-part definition, including that the treatment must be “in accordance with the standards of good medical policy” and “the most appropriate supply or level of service which can be safely provided.”
Behind the scenes at United, Mr. Opperman and Ms. Kavanaugh agreed that if Mr. McNaughton were to appeal Dr. Pabby’s decision, the insurer would simply rule against him. “I just think it’s a waste of money and time to appeal and send it to another one when we know we’re gonna get the same answer,” Mr. Opperman said, according to a recording in court files. At Mr. Opperman’s urging, United decided to skip the usual appeals process and arrange for Dr. Pabby to have a so-called “peer-to-peer” discussion with Dr. Loftus, the Mayo physician treating Mr. McNaughton. Such a conversation, in which a patient’s doctor talks with an insurance company’s doctor to advocate for the prescribed treatment, usually occurs only after a customer has appealed a denial and the appeal has been rejected.
When Ms. Kavanaugh called Dr. Loftus’ office to set up a conversation with Dr. Pabby, she explained it was an urgent matter and had been requested by Mr. McNaughton. “You know I’ve just gotten to know Christopher,” she explained, although she had never spoken with him. “We’re trying to advocate and help and get this peer-to-peer set up.”
Mr. McNaughton, meanwhile, had no idea at the time that a United doctor had decided his treatment was unnecessary and that the insurer was trying to set up a phone call with his physician.
In the peer-to-peer conversation, Dr. Loftus told Dr. Pabby that Mr. McNaughton had “a very complicated case” and that lower doses had not worked for him, according to an internal MRIoA memo.
Following his conversation with Dr. Loftus, Dr. Pabby created a second report for United. He recommended the insurer pay for both drugs, but at reduced doses. He added new language saying that the safety of using both drugs at the higher levels “is not established.”
When Ms. Kavanaugh shared the May 12 decision from Dr. Pabby with others at United, her boss responded with an email calling it “great news.”
Then Mr. Opperman sent an email that puzzled the McNaughtons.
In it, Mr. Opperman claimed that Dr. Loftus and Dr. Pabby had agreed that Mr. McNaughton should be on significantly lower doses of both drugs. He said Dr. Loftus “will work with the patient to start titrating them down to a normal dose range.” Mr. Opperman wrote that United would cover Mr. McNaughton’s treatment in the coming year, but only at the reduced doses. Mr. Opperman did not respond to emails and phone messages seeking comment.
Mr. McNaughton didn’t believe a word of it. He had already tried and failed treatment with those drugs at lower doses, and it was Dr. Loftus who had upped the doses, leading to his remission from severe colitis.
The only thing that made sense to Mr. McNaughton was that the treatment United said it would now pay for was dramatically cheaper – saving the company at least hundreds of thousands of dollars a year – than his prescribed treatment because it sliced the size of the doses by more than half.
When the family contacted Dr. Loftus for an explanation, they were outraged by what they heard. Dr. Loftus told them that he had never recommended lowering the dosage. In a letter, Dr. Loftus wrote that changing Mr. McNaughton’s treatment “would have serious detrimental effects on both his short term and long term health and could potentially involve life threatening complications. This would ultimately incur far greater medical costs. Chris was on the doses suggested by United Healthcare before, and they were not at all effective.”
It would not be until the lawsuit that it would become clear how Dr. Loftus’ conversations had been so seriously misrepresented.
Under questioning by Mr. McNaughton’s lawyers, Ms. Kavanaugh acknowledged that she was the source of the incorrect claim that Mr. McNaughton’s doctor had agreed to a change in treatment.
“I incorrectly made an assumption that they had come to some sort of agreement,” she said in a deposition last August. “It was my first peer-to-peer. I did not realize that that simply does not occur.”
Ms. Kavanaugh did not respond to emails and telephone messages seeking comment.
When the McNaughtons first learned of Mr. Opperman’s inaccurate report of the phone call with Dr. Loftus, it unnerved them. They started to question if their case would be fairly reviewed.
“When we got the denial and they lied about what Dr. Loftus said, it just hit me that none of this matters,” Mr. McNaughton said. “They will just say or do anything to get rid of me. It delegitimized the entire review process. When I got that denial, I was crushed.”
A buried report
While the family tried to sort out the inaccurate report, United continued putting the McNaughton case in front of more company doctors.
On May 21, 2021, United sent the case to one of its own doctors, Nady Cates, MD, for an additional review. The review was marked “escalated issue.” Dr. Cates is a United medical director, a title used by many insurers for physicians who review cases. It is work he has been doing as an employee of health insurers since 1989 and at United since 2010. He has not practiced medicine since the early 1990s.
Dr. Cates, in a deposition, said he stopped seeing patients because of the long hours involved and because “AIDS was coming around then. I was seeing a lot of military folks who had venereal diseases, and I guess I was concerned about being exposed.” He transitioned to reviewing paperwork for the insurance industry, he said, because “I guess I was a chicken.”
When he had practiced, Dr. Cates said, he hadn’t treated patients with ulcerative colitis and had referred those cases to a gastroenterologist.
He said his review of Mr. McNaughton’s case primarily involved reading a United nurse’s recommendation to deny his care and making sure “that there wasn’t a decimal place that was out of line.” He said he copied and pasted the nurse’s recommendation and typed “agree” on his review of Mr. McNaughton’s case.
Dr. Cates said that he does about a hundred reviews a week. He said that in his reviews he typically checks to see if any medications are prescribed in accordance with the insurer’s guidelines, and if not, he denies it. United’s policies, he said, prevented him from considering that Mr. McNaughton had failed other treatments or that Dr. Loftus was a leading expert in his field.
“You are giving zero weight to the treating doctor’s opinion on the necessity of the treatment regimen?” a lawyer asked Dr. Cates in his deposition. He responded, “Yeah.”
Attempts to contact Dr. Cates for comment were unsuccessful.
At the same time Dr. Cates was looking at Mr. McNaughton’s case, yet another review was underway at MRIoA. United said it sent the case back to MRIoA after the insurer received the letter from Dr. Loftus warning of the life-threatening complications that might occur if the dosages were reduced.
On May 24, 2021, the new report requested by MRIoA arrived. It came to a completely different conclusion than all of the previous reviews.
Nitin Kumar, MD, a gastroenterologist in Illinois, concluded that Mr. McNaughton’s established treatment plan was not only medically necessary and appropriate but that lowering his doses “can result in a lack of effective therapy of Ulcerative Colitis, with complications of uncontrolled disease (including dysplasia leading to colorectal cancer), flare, hospitalization, need for surgery, and toxic megacolon.”
Unlike other doctors who produced reports for United, Dr. Kumar discussed the harm that Mr. McNaughton might suffer if United required him to change his treatment. “His disease is significantly severe, with diagnosis at a young age,” Dr. Kumar wrote. “He has failed every biologic medication class recommended by guidelines. Therefore, guidelines can no longer be applied in this case.” He cited six studies of patients using two biologic drugs together and wrote that they revealed no significant safety issues and found the therapy to be “broadly successful.”
When Ms. Kavanaugh learned of Dr. Kumar’s report, she quickly moved to quash it and get the case returned to Dr. Pabby, according to her deposition.
In a recorded telephone call, Ms. Kavanaugh told an MRIoA representative that “I had asked that this go back through Dr. Pabby, and it went through a different doctor and they had a much different result.” After further discussion, the MRIoA representative agreed to send the case back to Dr. Pabby. “I appreciate that,” Ms. Kavanaugh replied. “I just want to make sure, because, I mean, it’s obviously a very different result than what we’ve been getting on this case.”
MRIoA case notes show that at 7:04 a.m. on May 25, 2021, Dr. Pabby was assigned to take a look at the case for the third time. At 7:27 a.m., the notes indicate, Dr. Pabby again rejected Mr. McNaughton’s treatment plan. While noting it was “difficult to control” Mr. McNaughton’s ulcerative colitis, Dr. Pabby added that his doses “far exceed what is approved by literature” and that the “safety of the requested doses is not supported by literature.”
In a deposition, Ms. Kavanaugh said that after she opened the Kumar report and read that he was supporting Mr. McNaughton’s current treatment plan, she immediately spoke to her supervisor, who told her to call MRIoA and have the case sent back to Dr. Pabby for review.
Ms. Kavanaugh said she didn’t save a copy of the Kumar report, nor did she forward it to anyone at United or to officials at Penn State who had been inquiring about the McNaughton case. “I didn’t because it shouldn’t have existed,” she said. “It should have gone back to Dr. Pabby.”
When asked if the Kumar report caused her any concerns given his warning that Mr. McNaughton risked cancer or hospitalization if his regimen were changed, Ms. Kavanaugh said she didn’t read his full report. “I saw that it was not the correct doctor, I saw the initial outcome and I was asked to send it back,” she said. Ms. Kavanaugh added, “I have a lot of empathy for this member, but it needed to go back to the peer-to-peer reviewer.”
In a court filing, United said Ms. Kavanaugh was correct in insisting that Dr. Pabby conduct the review and that MRIoA confirmed that Dr. Pabby should have been the one doing the review.
The Kumar report was not provided to Mr. McNaughton when his lawyer, Jonathan M. Gesk, first asked United and MRIoA for any reviews of the case. Mr. Gesk discovered it by accident when he was listening to a recorded telephone call produced by United in which Ms. Kavanaugh mentioned a report number Mr. Gesk had not heard before. He then called MRIoA, which confirmed the report existed and eventually provided it to him.
Dr. Pabby asked ProPublica to direct any questions about his involvement in the matter to MRIoA. The company did not respond to questions from ProPublica about the case.
A sense of hopelessness
When Mr. McNaughton enrolled at Penn State in 2020, it brought a sense of normalcy that he had lost when he was first diagnosed with colitis. He still needed monthly hours-long infusions and suffered occasional flare-ups and symptoms, but he was attending classes in person and living a life similar to the one he had before his diagnosis.
It was a striking contrast to the previous 6 years, which he had spent largely confined to his parents’ house in State College. The frequent bouts of diarrhea made it difficult to go out. He didn’t talk much to friends and spent as much time as he could studying potential treatments and reviewing ongoing clinical trials. He tried to keep up with the occasional online course, but his disease made it difficult to make any real progress toward a degree.
United, in correspondence with Mr. McNaughton, noted that its review of his care was “not a treatment decision. Treatment decisions are made between you and your physician.” But by threatening not to pay for his medications, or only to pay for a different regimen, Mr. McNaughton said, United was in fact attempting to dictate his treatment. From his perspective, the insurer was playing doctor, making decisions without ever examining him or even speaking to him.
The idea of changing his treatment or stopping it altogether caused constant worry for Mr. McNaughton, exacerbating his colitis and triggering physical symptoms, according to his doctors. Those included a large ulcer on his leg and welts under his skin on his thighs and shin that made his leg muscles stiff and painful to the point where he couldn’t bend his leg or walk properly. There were daily migraines and severe stomach pain. “I was consumed with this situation,” Mr. McNaughton said. “My path was unconventional, but I was proud of myself for fighting back and finishing school and getting my life back on track. I thought they were singling me out. My biggest fear was going back to the hell.”
Mr. McNaughton said he contemplated suicide on several occasions, dreading a return to a life where he was housebound or hospitalized.
Mr. McNaughton and his parents talked about his possibly moving to Canada where his grandmother lived and seeking treatment there under the nation’s government health plan.
Dr. Loftus connected Mr. McNaughton with a psychologist who specializes in helping patients with chronic digestive diseases.
The psychologist, Tiffany Taft, PsyD, said Mr. McNaughton was not an unusual case. About one in three patients with diseases like colitis suffer from medical trauma or PTSD related to it, she said, often the result of issues related to getting appropriate treatment approved by insurers.
“You get into hopelessness,” she said of the depression that accompanies fighting with insurance companies over care. “They feel like ‘I can’t fix that. I am screwed.’ When you can’t control things with what an insurance company is doing, anxiety, PTSD and depression get mixed together.”
In the case of Mr. McNaughton, Dr. Taft said, he was being treated by one of the best gastroenterologists in the world, was doing well with his treatment, and then was suddenly notified he might be on the hook for nearly a million dollars in medical charges without access to his medications. “It sends you immediately into panic about all these horrific things that could happen,” Dr. Taft said. The physical and mental symptoms Mr. McNaughton suffered after his care was threatened were “triggered” by the stress he experienced, she said.
In early June 2021, United informed Mr. McNaughton in a letter that it would not cover the cost of his treatment regimen in the next academic year, starting in August. The insurer said it would pay only for a treatment plan that called for a significant reduction in the doses of the drugs he took.
United wrote that the decision came after his “records have been reviewed three times and the medical reviewers have concluded that the medication as prescribed does not meet the Medical Necessity requirement of the plan.”
In August 2021, Mr. McNaughton filed a federal lawsuit accusing United of acting in bad faith and unreasonably making treatment decisions based on financial concerns and not what was the best and most effective treatment. It claims United had a duty to find information that supported Mr. McNaughton’s claim for treatment rather than looking for ways to deny coverage.
United, in a court filing, said it did not breach any duty it owed to Mr. McNaughton and acted in good faith. On Sept. 20, 2021, a month after filing the lawsuit, and with United again balking at paying for his treatment, Mr. McNaughton asked a judge to grant a temporary restraining order requiring United to pay for his care. With the looming threat of a court hearing on the motion, United quickly agreed to cover the cost of Mr. McNaughton’s treatment through the end of the 2021-2022 academic year. It also dropped a demand requiring Mr. McNaughton to settle the matter as a condition of the insurer paying for his treatment as prescribed by Dr. Loftus, according to an email sent by United’s lawyer.
The cost of treatment
It is not surprising that insurers are carefully scrutinizing the care of patients treated with biologics, which are among the most expensive medications on the market. Biologics are considered specialty drugs, a class that includes the best-selling Humira, used to treat arthritis. Specialty drug spending in the United States is expected to reach $505 billion in 2023, according to an estimate from Optum, United’s health services division. The Institute for Clinical and Economic Review, a nonprofit that analyzes the value of drugs, found in 2020 that the biologic drugs used to treat patients like Mr. McNaughton are often effective but overpriced for their therapeutic benefit. To be judged cost-effective by ICER, the biologics should sell at a steep discount to their current market price, the panel found.
A panel convened by ICER to review its analysis cautioned that insurance coverage “should be structured to prevent situations in which patients are forced to choose a treatment approach on the basis of cost.” ICER also found examples where insurance company policies failed to keep pace with updates to clinical practice guidelines based on emerging research.
United officials did not make the cost of treatment an issue when discussing Mr. McNaughton’s care with Penn State administrators or the family.
Bill Truxal, the president of UnitedHealthcare StudentResources, the company’s student health plan division, told a Penn State official that the insurer wanted the “best for the student” and it had “nothing to do with cost,” according to notes the official took of the conversation.
Behind the scenes, however, the price of Mr. McNaughton’s care was front and center at United.
In one email, Mr. Opperman asked about the cost difference if the insurer insisted on paying only for greatly reduced doses of the biologic drugs. Ms. Kavanaugh responded that the insurer had paid $1.1 million in claims for Mr. McNaughton’s care as of the middle of May 2021. If the reduced doses had been in place, the amount would have been cut to $260,218, she wrote.
United was keeping close tabs on Mr. McNaughton at the highest levels of the company. On Aug. 2, 2021, Mr. Opperman notified Mr. Truxal and a United lawyer that Mr. McNaughton “has just purchased the plan again for the 21-22 school year.”
A month later, Ms. Kavanaugh shared another calculation with United executives showing that the insurer spent over $1.7 million on Mr. McNaughton in the prior plan year.
United officials strategized about how to best explain why it was reviewing Mr. McNaughton’s drug regimen, according to an internal email. They pointed to a justification often used by health insurers when denying claims. “As the cost of healthcare continues to climb to soaring heights, it has been determined that a judicious review of these drugs should be included” in order to “make healthcare more affordable for our members,” Ms. Kavanaugh offered as a potential talking point in an April 23, 2021, email.
Three days later, UnitedHealth Group filed an annual statement with the U.S. Securities and Exchange Commission disclosing its pay for top executives in the prior year. Then-CEO David Wichmann was paid $17.9 million in salary and other compensation in 2020. Wichmann retired early the following year, and his total compensation that year exceeded $140 million, according to calculations in a compensation database maintained by the Star Tribune in Minneapolis. The newspaper said the amount was the most paid to an executive in the state since it started tracking pay more than 2 decades ago. About $110 million of that total came from Wichmann exercising stock options accumulated during his stewardship.
The McNaughtons were well aware of the financial situation at United. They looked at publicly available financial results and annual reports. Last year, United reported a profit of $20.1 billion on revenues of $324.2 billion.
When discussing the case with Penn State, Ms. Light said, she told university administrators that United could pay for a year of her son’s treatment using just minutes’ worth of profit.
‘Betrayed’
Mr. McNaughton has been able to continue receiving his infusions for now, anyway. In October, United notified him it was once again reviewing his care, although the insurer quickly reversed course when his lawyer intervened. United, in a court filing, said the review was a mistake and that it had erred in putting Mr. McNaughton’s claims into pending status.
Mr. McNaughton said he is fortunate his parents were employed at the same school he was attending, which was critical in getting the attention of administrators there. But that help had its limits.
In June 2021, just a week after United told Mr. McNaughton it would not cover his treatment plan in the upcoming plan year, Penn State essentially walked away from the matter.
In an email to the McNaughtons and United, Penn State Associate Vice President for Student Affairs Andrea Dowhower wrote that administrators “have observed an unfortunate breakdown in communication” between Mr. McNaughton and his family and the university health insurance plan, “which appears from our perspective to have resulted in a standstill between the two parties.” While she proposed some potential steps to help settle the matter, she wrote that “Penn State’s role in this process is as a resource for students like Chris who, for whatever reason, have experienced difficulty navigating the complex world of health insurance.” The university’s role “is limited,” she wrote, and the school “simply must leave” the issue of the best treatment for Mr. McNaughton to “the appropriate health care professionals.”
In a statement, a Penn State spokesperson wrote that “as a third party in this arrangement, the University’s role is limited and Penn State officials can only help a student manage an issue based on information that a student/family, medical personnel, and/or insurance provider give – with the hope that all information is accurate and that the lines of communication remain open between the insured and the insurer.”
Penn State declined to provide financial information about the plan. However, the university and United share at least one tie that they have not publicly disclosed.
When the McNaughtons first reached out to the university for help, they were referred to the school’s student health insurance coordinator. The official, Heather Klinger, wrote in an email to the family in February 2021 that “I appreciate your trusting me to resolve this for you.”
In April 2022, United began paying Ms. Klinger’s salary, an arrangement which is not noted on the university website. Ms. Klinger appears in the online staff directory on the Penn State University Health Services web page, and has a university phone number, a university address, and a Penn State email listed as her contact. The school said she has maintained a part-time status with the university to allow her to access relevant data systems at both the university and United.
The university said students “benefit” from having a United employee to handle questions about insurance coverage and that the arrangement is “not uncommon” for student health plans.
The family was dismayed to learn that Ms. Klinger was now a full-time employee of United.
“We did feel betrayed,” Ms. Light said. Ms. Klinger did not respond to an email seeking comment.
Mr. McNaughton’s fight to maintain his treatment regimen has come at a cost of time, debilitating stress, and depression. “My biggest fear is realizing I might have to do this every year of my life,” he said.
Mr. McNaughton said one motivation for his lawsuit was to expose how insurers like United make decisions about what care they will pay for and what they will not. The case remains pending, a court docket shows.
He has been accepted to Penn State’s law school. He hopes to become a health care lawyer working for patients who find themselves in situations similar to his.
He plans to re-enroll in the United health care plan when he starts school next fall.
This story was originally published on ProPublica. ProPublica is a nonprofit newsroom that investigates abuses of power. Sign up to receive the biggest stories as soon as they’re published.
In May 2021, a nurse at UnitedHealthcare called a colleague to share some welcome news about a problem the two had been grappling with for weeks.
United provided the health insurance plan for students at Penn State University. It was a large and potentially lucrative account: lots of young, healthy students paying premiums in, not too many huge medical reimbursements going out.
But Christopher McNaughton suffered from a crippling case of ulcerative colitis – an ailment that caused him to develop severe arthritis, debilitating diarrhea, numbing fatigue, and life-threatening blood clots. His medical bills were running nearly $2 million a year.
United had flagged Mr. McNaughton’s case as a “high dollar account,” and the company was reviewing whether it needed to keep paying for the expensive cocktail of drugs crafted by a Mayo Clinic specialist that had brought Mr. McNaughton’s disease under control after he’d been through years of misery.
On the 2021 phone call, which was recorded by the company, nurse Victoria Kavanaugh told her colleague that a doctor contracted by United to review the case had concluded that Mr. McNaughton’s treatment was “not medically necessary.” Her colleague, Dave Opperman, reacted to the news with a long laugh.
“I knew that was coming,” said Mr. Opperman, who heads up a United subsidiary that brokered the health insurance contract between United and Penn State. “I did too,” Ms. Kavanaugh replied.
Mr. Opperman then complained about Mr. McNaughton’s mother, whom he referred to as “this woman,” for “screaming and yelling” and “throwing tantrums” during calls with United.
The pair agreed that any appeal of the United doctor’s denial of the treatment would be a waste of the family’s time and money.
“We’re still gonna say no,” Mr. Opperman said.
More than 200 million Americans are covered by private health insurance. But data from state and federal regulators shows that insurers reject about 1 in 7 claims for treatment. Many people, faced with fighting insurance companies, simply give up: One study found that Americans file formal appeals on only 0.1% of claims denied by insurers under the Affordable Care Act.
Insurers have wide discretion in crafting what is covered by their policies, beyond some basic services mandated by federal and state law. They often deny claims for services that they deem not “medically necessary.”
When United refused to pay for Mr. McNaughton’s treatment for that reason, his family did something unusual. They fought back with a lawsuit, which uncovered a trove of materials, including internal emails and tape-recorded exchanges among company employees. Those records offer an extraordinary behind-the-scenes look at how one of America’s leading health care insurers relentlessly fought to reduce spending on care, even as its profits rose to record levels.
As United reviewed Mr. McNaughton’s treatment, he and his family were often in the dark about what was happening or their rights. Meanwhile, United employees misrepresented critical findings and ignored warnings from doctors about the risks of altering Mr. McNaughton’s drug plan.
At one point, court records show, United inaccurately reported to Penn State and the family that Mr. McNaughton’s doctor had agreed to lower the doses of his medication. Another time, a doctor paid by United concluded that denying payments for Mr. McNaughton’s treatment could put his health at risk, but the company buried his report and did not consider its findings. The insurer did, however, consider a report submitted by a company doctor who rubber-stamped the recommendation of a United nurse to reject paying for the treatment.
United declined to answer specific questions about the case, even after Mr. McNaughton signed a release provided by the insurer to allow it to discuss details of his interactions with the company. United noted that it ultimately paid for all of Mr. McNaughton’s treatments. In a written response, United spokesperson Maria Gordon Shydlo wrote that the company’s guiding concern was Mr. McNaughton’s well-being.
“Mr. McNaughton’s treatment involves medication dosages that far exceed [Food and Drug Administration] guidelines,” the statement said. “In cases like this, we review treatment plans based on current clinical guidelines to help ensure patient safety.”
But the records reviewed by ProPublica show that United had another, equally urgent goal in dealing with Mr. McNaughton. In emails, officials calculated what Mr. McNaughton was costing them to keep his crippling disease at bay and how much they would save if they forced him to undergo a cheaper treatment that had already failed him. As the family pressed the company to back down, first through Penn State and then through a lawsuit, the United officials handling the case bristled.
“This is just unbelievable,” Ms. Kavanaugh said of Mr. McNaughton’s family in one call to discuss his case. ”They’re just really pushing the envelope, and I’m surprised, like I don’t even know what to say.”
The same meal every day
Now 31, Mr. McNaughton grew up in State College, Pa., just blocks from the Penn State campus. Both of his parents are faculty members at the university.
In the winter of 2014, Mr. McNaughton was halfway through his junior year at Bard College in New York. At 6 feet, 4 inches tall, he was a guard on the basketball team and had started most of the team’s games since the start of his sophomore year. He was majoring in psychology.
When Mr. McNaughton returned to school after the winter holiday break, he started to experience frequent bouts of bloody diarrhea. After just a few days on campus, he went home to State College, where doctors diagnosed him with a severe case of ulcerative colitis.
A chronic inflammatory bowel disease that causes swelling and ulcers in the digestive tract, ulcerative colitis has no cure, and ongoing treatment is needed to alleviate symptoms and prevent serious health complications. The majority of cases produce mild to moderate symptoms. Mr. McNaughton’s case was severe.
Treatments for ulcerative colitis include steroids and special drugs known as biologics that work to reduce inflammation in the large intestine.
Mr. McNaughton, however, failed to get meaningful relief from the drugs his doctors initially prescribed. He was experiencing bloody diarrhea up to 20 times a day, with such severe stomach pain that he spent much of his day curled up on a couch. He had little appetite and lost 50 pounds. Severe anemia left him fatigued. He suffered from other conditions related to his colitis, including crippling arthritis. He was hospitalized several times to treat dangerous blood clots.
For 2 years, in an effort to help alleviate his symptoms, he ate the same meals every day: Rice Chex cereal and scrambled eggs for breakfast, a cup of white rice with plain chicken breast for lunch, and a similar meal for dinner, occasionally swapping in tilapia.
His hometown doctors referred him to a specialist at the University of Pittsburgh, who tried unsuccessfully to bring his disease under control. That doctor ended up referring Mr. McNaughton to Edward V. Loftus Jr., MD, at the Mayo Clinic in Rochester, Minn., which has been ranked as the best gastroenterology hospital in the country every year since 1990 by U.S. News & World Report.
For his first visit with Dr. Loftus in May 2015, Mr. McNaughton and his mother, Janice Light, charted hospitals along the 900-mile drive from Pennsylvania to Minnesota in case they needed medical help along the way.
Mornings were the hardest. Mr. McNaughton often spent several hours in the bathroom at the start of the day. To prepare for his meeting with Dr. Loftus, he set his alarm for 3:30 a.m. so he could be ready for the 7:30 a.m. appointment. Even with that preparation, he had to stop twice to use a bathroom on the 5-minute walk from the hotel to the clinic. When they met, Dr. Loftus looked at Mr. McNaughton and told him that he appeared incapacitated. It was, he told the student, as if Mr. McNaughton were chained to the bathroom, with no outside life. He had not been able to return to school and spent most days indoors, managing his symptoms as best he could.
Mr. McNaughton had tried a number of medications by this point, none of which worked. This pattern would repeat itself during the first couple of years that Dr. Loftus treated him.
In addition to trying to find a treatment that would bring Mr. McNaughton’s colitis into remission, Dr. Loftus wanted to wean him off the steroid prednisone, which he had been taking since his initial diagnosis in 2014. The drug is commonly prescribed to colitis patients to control inflammation, but prolonged use can lead to severe side effects including cataracts, osteoporosis, increased risk of infection, and fatigue. Mr. McNaughton also experienced “moon face,” a side effect caused by the shifting of fat deposits that results in the face becoming puffy and rounder.
In 2018, Dr. Loftus and Mr. McNaughton decided to try an unusual regimen. Many patients with inflammatory bowel diseases such as colitis take a single biologic drug as treatment. Whereas traditional drugs are chemically synthesized, biologics are manufactured in living systems, such as plant or animal cells. A year’s supply of an individual biologic drug can cost up to $500,000. They are often given through infusions in a medical facility, which adds to the cost.
Mr. McNaughton had tried individual biologics, and then two in combination, without much success. He and Dr. Loftus then agreed to try two biologic drugs together at doses well above those recommended by the Food and Drug Administration. The federal Agency for Healthcare Research and Quality estimates one in five prescriptions written today are for off-label uses.
There are drawbacks to the practice. Since some uses and doses of particular drugs have not been extensively studied, the risks and efficacy of using them off-label are not well known. Also, some drug manufacturers have improperly pushed off-label usage of their products to boost sales despite little or no evidence to support their use in those situations. Like many leading experts and researchers in his field, Dr. Loftus has been paid to do consulting related to the biologic drugs taken by Mr. McNaughton. The payments related to those drugs have ranged from a total of $1,440 in 2020 to $51,235 in 2018. Dr. Loftus said much of his work with pharmaceutical companies was related to conducting clinical trials on new drugs.
In cases of off-label prescribing, patients are depending upon their doctors’ expertise and experience with the drug. “In this case, I was comfortable that the potential benefits to Chris outweighed the risks,” Dr. Loftus said.
There was evidence that the treatment plan for Mr. McNaughton might work, including studies that had found dual biologic therapy to be efficacious and safe. The two drugs he takes, Entyvio and Remicade, have the same purpose – to reduce inflammation in the large intestine – but each works differently in the body. Remicade, marketed by Janssen Biotech, targets a protein that causes inflammation. Entyvio, made by Takeda Pharmaceuticals, works by preventing an excess of white blood cells from entering into the gastrointestinal tract.
As for any suggestion by United doctors that his treatment plan for Mr. McNaughton was out of bounds or dangerous, Dr. Loftus said “my treatment of Chris was not clinically inappropriate – as was shown by Chris’ positive outcome.”
The unusual high-dose combination of two biologic drugs produced a remarkable change in Mr. McNaughton. He no longer had blood in his stool, and his trips to the bathroom were cut from 20 times a day to 3 or 4. He was able to eat different foods and put on weight. He had more energy. He tapered off prednisone.
“If you told me in 2015 that I would be living like this, I would have asked where do I sign up,” Mr. McNaughton said of the change he experienced with the new drug regimen.
When he first started the new treatment, Mr. McNaughton was covered under his family’s plan, and all his bills were paid. Mr. McNaughton enrolled at the university in 2020. Before switching to United’s plan for students, Mr. McNaughton and his parents consulted with a health advocacy service offered to faculty members. A benefits specialist assured them the drugs taken by Mr. McNaughton would be covered by United.
Mr. McNaughton joined the student plan in July 2020, and his infusions that month and the following month were paid for by United. In September, the insurer indicated payment on his claims was “pending,” something it did for his other claims that came in during the rest of the year.
Mr. McNaughton and his family were worried. They called United to make sure there wasn’t a problem; the insurer told them, they said, that it only needed to check his medical records. When the family called again, United told them it had the documentation needed, they said. United, in a court filing last year, said it received two calls from the family and each time indicated that all of the necessary medical records had not yet been received.
In January 2021, Mr. McNaughton received a new explanation of benefits for the prior months. All of the claims for his care, beginning in September, were no longer “pending.” They were stamped “DENIED.” The total outstanding bill for his treatment was $807,086.
When Mr. McNaughton’s mother reached a United customer service representative the next day to ask why bills that had been paid in the summer were being denied for the fall, the representative told her the account was being reviewed because of “a high dollar amount on the claims,” according to a recording of the call.
Misrepresentations
With United refusing to pay, the family was terrified of being stuck with medical bills that would bankrupt them and deprive Mr. McNaughton of treatment that they considered miraculous.
They turned to Penn State for help. Ms. Light and Mr. McNaughton’s father, David McNaughton, hoped their position as faculty members would make the school more willing to intervene on their behalf.
“After more than 30 years on faculty, my husband and I know that this is not how Penn State would want its students to be treated,” Ms. Light wrote to a school official in February 2021.
In response to questions from ProPublica, Penn State spokesperson Lisa Powers wrote that “supporting the health and well-being of our students is always of primary importance” and that “our hearts go out to any student and family impacted by a serious medical condition.” The university, she wrote, does “not comment on students’ individual circumstances or disclose information from their records.” Mr. McNaughton offered to grant Penn State whatever permissions it needed to speak about his case with ProPublica. The school, however, wrote that it would not comment “even if confidentiality has been waived.”
The family appealed to school administrators. Because the effectiveness of biologics wanes in some patients if doses are skipped, Mr. McNaughton and his parents were worried about even a delay in treatment. His doctor wrote that if he missed scheduled infusions of the drugs, there was “a high likelihood they would no longer be effective.”
During a conference call arranged by Penn State officials on March 5, 2021, United agreed to pay for Mr. McNaughton’s care through the end of the plan year that August. Penn State immediately notified the family of the “wonderful news” while also apologizing for “the stress this has caused Chris and your family.”
Behind the scenes, Mr. McNaughton’s review had “gone all the way to the top” at United’s student health plan division, Ms. Kavanaugh, the nurse, said in a recorded conversation.
The family’s relief was short-lived. A month later, United started another review of Mr. McNaughton’s care, overseen by Ms. Kavanaugh, to determine if it would pay for the treatment in the upcoming plan year.
The nurse sent the Mr. McNaughton case to a company called Medical Review Institute of America. Insurers often turn to companies like MRIoA to review coverage decisions involving expensive treatments or specialized care.
Ms. Kavanaugh, who was assigned to a special investigations unit at United, let her feelings about the matter be known in a recorded telephone call with a representative of MRIoA.
“This school apparently is a big client of ours,” she said. She then shared her opinion of Mr. McNaughton’s treatment. “Really this is a case of a kid who’s getting a drug way too much, like too much of a dose,” Ms. Kavanaugh said. She said it was “insane that they would even think that this is reasonable” and “to be honest with you, they’re awfully pushy considering that we are paying through the end of this school year.”
On a call with an outside contractor, the United nurse claimed Mr. McNaughton was on a higher dose of medication than the FDA approved, which is a common practice.
MRIoA sent the case to Vikas Pabby, MD, a gastroenterologist at UCLA Health and a professor at the university’s medical school. His May 2021 review of Mr. McNaughton’s case was just one of more than 300 Dr. Pabby did for MRIoA that month, for which he was paid $23,000 in total, according to a log of his work produced in the lawsuit.
In a May 4, 2021, report, Dr. Pabby concluded Mr. McNaughton’s treatment was not medically necessary, because United’s policies for the two drugs taken by Mr. McNaughton did not support using them in combination.
Insurers spell out what services they cover in plan policies, lengthy documents that can be confusing and difficult to understand. Many policies, such as Mr. McNaughton’s, contain a provision that treatments and procedures must be “medically necessary” in order to be covered. The definition of medically necessary differs by plan. Some don’t even define the term. Mr. McNaughton’s policy contains a five-part definition, including that the treatment must be “in accordance with the standards of good medical policy” and “the most appropriate supply or level of service which can be safely provided.”
Behind the scenes at United, Mr. Opperman and Ms. Kavanaugh agreed that if Mr. McNaughton were to appeal Dr. Pabby’s decision, the insurer would simply rule against him. “I just think it’s a waste of money and time to appeal and send it to another one when we know we’re gonna get the same answer,” Mr. Opperman said, according to a recording in court files. At Mr. Opperman’s urging, United decided to skip the usual appeals process and arrange for Dr. Pabby to have a so-called “peer-to-peer” discussion with Dr. Loftus, the Mayo physician treating Mr. McNaughton. Such a conversation, in which a patient’s doctor talks with an insurance company’s doctor to advocate for the prescribed treatment, usually occurs only after a customer has appealed a denial and the appeal has been rejected.
When Ms. Kavanaugh called Dr. Loftus’ office to set up a conversation with Dr. Pabby, she explained it was an urgent matter and had been requested by Mr. McNaughton. “You know I’ve just gotten to know Christopher,” she explained, although she had never spoken with him. “We’re trying to advocate and help and get this peer-to-peer set up.”
Mr. McNaughton, meanwhile, had no idea at the time that a United doctor had decided his treatment was unnecessary and that the insurer was trying to set up a phone call with his physician.
In the peer-to-peer conversation, Dr. Loftus told Dr. Pabby that Mr. McNaughton had “a very complicated case” and that lower doses had not worked for him, according to an internal MRIoA memo.
Following his conversation with Dr. Loftus, Dr. Pabby created a second report for United. He recommended the insurer pay for both drugs, but at reduced doses. He added new language saying that the safety of using both drugs at the higher levels “is not established.”
When Ms. Kavanaugh shared the May 12 decision from Dr. Pabby with others at United, her boss responded with an email calling it “great news.”
Then Mr. Opperman sent an email that puzzled the McNaughtons.
In it, Mr. Opperman claimed that Dr. Loftus and Dr. Pabby had agreed that Mr. McNaughton should be on significantly lower doses of both drugs. He said Dr. Loftus “will work with the patient to start titrating them down to a normal dose range.” Mr. Opperman wrote that United would cover Mr. McNaughton’s treatment in the coming year, but only at the reduced doses. Mr. Opperman did not respond to emails and phone messages seeking comment.
Mr. McNaughton didn’t believe a word of it. He had already tried and failed treatment with those drugs at lower doses, and it was Dr. Loftus who had upped the doses, leading to his remission from severe colitis.
The only thing that made sense to Mr. McNaughton was that the treatment United said it would now pay for was dramatically cheaper – saving the company at least hundreds of thousands of dollars a year – than his prescribed treatment because it sliced the size of the doses by more than half.
When the family contacted Dr. Loftus for an explanation, they were outraged by what they heard. Dr. Loftus told them that he had never recommended lowering the dosage. In a letter, Dr. Loftus wrote that changing Mr. McNaughton’s treatment “would have serious detrimental effects on both his short term and long term health and could potentially involve life threatening complications. This would ultimately incur far greater medical costs. Chris was on the doses suggested by United Healthcare before, and they were not at all effective.”
It would not be until the lawsuit that it would become clear how Dr. Loftus’ conversations had been so seriously misrepresented.
Under questioning by Mr. McNaughton’s lawyers, Ms. Kavanaugh acknowledged that she was the source of the incorrect claim that Mr. McNaughton’s doctor had agreed to a change in treatment.
“I incorrectly made an assumption that they had come to some sort of agreement,” she said in a deposition last August. “It was my first peer-to-peer. I did not realize that that simply does not occur.”
Ms. Kavanaugh did not respond to emails and telephone messages seeking comment.
When the McNaughtons first learned of Mr. Opperman’s inaccurate report of the phone call with Dr. Loftus, it unnerved them. They started to question if their case would be fairly reviewed.
“When we got the denial and they lied about what Dr. Loftus said, it just hit me that none of this matters,” Mr. McNaughton said. “They will just say or do anything to get rid of me. It delegitimized the entire review process. When I got that denial, I was crushed.”
A buried report
While the family tried to sort out the inaccurate report, United continued putting the McNaughton case in front of more company doctors.
On May 21, 2021, United sent the case to one of its own doctors, Nady Cates, MD, for an additional review. The review was marked “escalated issue.” Dr. Cates is a United medical director, a title used by many insurers for physicians who review cases. It is work he has been doing as an employee of health insurers since 1989 and at United since 2010. He has not practiced medicine since the early 1990s.
Dr. Cates, in a deposition, said he stopped seeing patients because of the long hours involved and because “AIDS was coming around then. I was seeing a lot of military folks who had venereal diseases, and I guess I was concerned about being exposed.” He transitioned to reviewing paperwork for the insurance industry, he said, because “I guess I was a chicken.”
When he had practiced, Dr. Cates said, he hadn’t treated patients with ulcerative colitis and had referred those cases to a gastroenterologist.
He said his review of Mr. McNaughton’s case primarily involved reading a United nurse’s recommendation to deny his care and making sure “that there wasn’t a decimal place that was out of line.” He said he copied and pasted the nurse’s recommendation and typed “agree” on his review of Mr. McNaughton’s case.
Dr. Cates said that he does about a hundred reviews a week. He said that in his reviews he typically checks to see if any medications are prescribed in accordance with the insurer’s guidelines, and if not, he denies it. United’s policies, he said, prevented him from considering that Mr. McNaughton had failed other treatments or that Dr. Loftus was a leading expert in his field.
“You are giving zero weight to the treating doctor’s opinion on the necessity of the treatment regimen?” a lawyer asked Dr. Cates in his deposition. He responded, “Yeah.”
Attempts to contact Dr. Cates for comment were unsuccessful.
At the same time Dr. Cates was looking at Mr. McNaughton’s case, yet another review was underway at MRIoA. United said it sent the case back to MRIoA after the insurer received the letter from Dr. Loftus warning of the life-threatening complications that might occur if the dosages were reduced.
On May 24, 2021, the new report requested by MRIoA arrived. It came to a completely different conclusion than all of the previous reviews.
Nitin Kumar, MD, a gastroenterologist in Illinois, concluded that Mr. McNaughton’s established treatment plan was not only medically necessary and appropriate but that lowering his doses “can result in a lack of effective therapy of Ulcerative Colitis, with complications of uncontrolled disease (including dysplasia leading to colorectal cancer), flare, hospitalization, need for surgery, and toxic megacolon.”
Unlike other doctors who produced reports for United, Dr. Kumar discussed the harm that Mr. McNaughton might suffer if United required him to change his treatment. “His disease is significantly severe, with diagnosis at a young age,” Dr. Kumar wrote. “He has failed every biologic medication class recommended by guidelines. Therefore, guidelines can no longer be applied in this case.” He cited six studies of patients using two biologic drugs together and wrote that they revealed no significant safety issues and found the therapy to be “broadly successful.”
When Ms. Kavanaugh learned of Dr. Kumar’s report, she quickly moved to quash it and get the case returned to Dr. Pabby, according to her deposition.
In a recorded telephone call, Ms. Kavanaugh told an MRIoA representative that “I had asked that this go back through Dr. Pabby, and it went through a different doctor and they had a much different result.” After further discussion, the MRIoA representative agreed to send the case back to Dr. Pabby. “I appreciate that,” Ms. Kavanaugh replied. “I just want to make sure, because, I mean, it’s obviously a very different result than what we’ve been getting on this case.”
MRIoA case notes show that at 7:04 a.m. on May 25, 2021, Dr. Pabby was assigned to take a look at the case for the third time. At 7:27 a.m., the notes indicate, Dr. Pabby again rejected Mr. McNaughton’s treatment plan. While noting it was “difficult to control” Mr. McNaughton’s ulcerative colitis, Dr. Pabby added that his doses “far exceed what is approved by literature” and that the “safety of the requested doses is not supported by literature.”
In a deposition, Ms. Kavanaugh said that after she opened the Kumar report and read that he was supporting Mr. McNaughton’s current treatment plan, she immediately spoke to her supervisor, who told her to call MRIoA and have the case sent back to Dr. Pabby for review.
Ms. Kavanaugh said she didn’t save a copy of the Kumar report, nor did she forward it to anyone at United or to officials at Penn State who had been inquiring about the McNaughton case. “I didn’t because it shouldn’t have existed,” she said. “It should have gone back to Dr. Pabby.”
When asked if the Kumar report caused her any concerns given his warning that Mr. McNaughton risked cancer or hospitalization if his regimen were changed, Ms. Kavanaugh said she didn’t read his full report. “I saw that it was not the correct doctor, I saw the initial outcome and I was asked to send it back,” she said. Ms. Kavanaugh added, “I have a lot of empathy for this member, but it needed to go back to the peer-to-peer reviewer.”
In a court filing, United said Ms. Kavanaugh was correct in insisting that Dr. Pabby conduct the review and that MRIoA confirmed that Dr. Pabby should have been the one doing the review.
The Kumar report was not provided to Mr. McNaughton when his lawyer, Jonathan M. Gesk, first asked United and MRIoA for any reviews of the case. Mr. Gesk discovered it by accident when he was listening to a recorded telephone call produced by United in which Ms. Kavanaugh mentioned a report number Mr. Gesk had not heard before. He then called MRIoA, which confirmed the report existed and eventually provided it to him.
Dr. Pabby asked ProPublica to direct any questions about his involvement in the matter to MRIoA. The company did not respond to questions from ProPublica about the case.
A sense of hopelessness
When Mr. McNaughton enrolled at Penn State in 2020, it brought a sense of normalcy that he had lost when he was first diagnosed with colitis. He still needed monthly hours-long infusions and suffered occasional flare-ups and symptoms, but he was attending classes in person and living a life similar to the one he had before his diagnosis.
It was a striking contrast to the previous 6 years, which he had spent largely confined to his parents’ house in State College. The frequent bouts of diarrhea made it difficult to go out. He didn’t talk much to friends and spent as much time as he could studying potential treatments and reviewing ongoing clinical trials. He tried to keep up with the occasional online course, but his disease made it difficult to make any real progress toward a degree.
United, in correspondence with Mr. McNaughton, noted that its review of his care was “not a treatment decision. Treatment decisions are made between you and your physician.” But by threatening not to pay for his medications, or only to pay for a different regimen, Mr. McNaughton said, United was in fact attempting to dictate his treatment. From his perspective, the insurer was playing doctor, making decisions without ever examining him or even speaking to him.
The idea of changing his treatment or stopping it altogether caused constant worry for Mr. McNaughton, exacerbating his colitis and triggering physical symptoms, according to his doctors. Those included a large ulcer on his leg and welts under his skin on his thighs and shin that made his leg muscles stiff and painful to the point where he couldn’t bend his leg or walk properly. There were daily migraines and severe stomach pain. “I was consumed with this situation,” Mr. McNaughton said. “My path was unconventional, but I was proud of myself for fighting back and finishing school and getting my life back on track. I thought they were singling me out. My biggest fear was going back to the hell.”
Mr. McNaughton said he contemplated suicide on several occasions, dreading a return to a life where he was housebound or hospitalized.
Mr. McNaughton and his parents talked about his possibly moving to Canada where his grandmother lived and seeking treatment there under the nation’s government health plan.
Dr. Loftus connected Mr. McNaughton with a psychologist who specializes in helping patients with chronic digestive diseases.
The psychologist, Tiffany Taft, PsyD, said Mr. McNaughton was not an unusual case. About one in three patients with diseases like colitis suffer from medical trauma or PTSD related to it, she said, often the result of issues related to getting appropriate treatment approved by insurers.
“You get into hopelessness,” she said of the depression that accompanies fighting with insurance companies over care. “They feel like ‘I can’t fix that. I am screwed.’ When you can’t control things with what an insurance company is doing, anxiety, PTSD and depression get mixed together.”
In the case of Mr. McNaughton, Dr. Taft said, he was being treated by one of the best gastroenterologists in the world, was doing well with his treatment, and then was suddenly notified he might be on the hook for nearly a million dollars in medical charges without access to his medications. “It sends you immediately into panic about all these horrific things that could happen,” Dr. Taft said. The physical and mental symptoms Mr. McNaughton suffered after his care was threatened were “triggered” by the stress he experienced, she said.
In early June 2021, United informed Mr. McNaughton in a letter that it would not cover the cost of his treatment regimen in the next academic year, starting in August. The insurer said it would pay only for a treatment plan that called for a significant reduction in the doses of the drugs he took.
United wrote that the decision came after his “records have been reviewed three times and the medical reviewers have concluded that the medication as prescribed does not meet the Medical Necessity requirement of the plan.”
In August 2021, Mr. McNaughton filed a federal lawsuit accusing United of acting in bad faith and unreasonably making treatment decisions based on financial concerns and not what was the best and most effective treatment. It claims United had a duty to find information that supported Mr. McNaughton’s claim for treatment rather than looking for ways to deny coverage.
United, in a court filing, said it did not breach any duty it owed to Mr. McNaughton and acted in good faith. On Sept. 20, 2021, a month after filing the lawsuit, and with United again balking at paying for his treatment, Mr. McNaughton asked a judge to grant a temporary restraining order requiring United to pay for his care. With the looming threat of a court hearing on the motion, United quickly agreed to cover the cost of Mr. McNaughton’s treatment through the end of the 2021-2022 academic year. It also dropped a demand requiring Mr. McNaughton to settle the matter as a condition of the insurer paying for his treatment as prescribed by Dr. Loftus, according to an email sent by United’s lawyer.
The cost of treatment
It is not surprising that insurers are carefully scrutinizing the care of patients treated with biologics, which are among the most expensive medications on the market. Biologics are considered specialty drugs, a class that includes the best-selling Humira, used to treat arthritis. Specialty drug spending in the United States is expected to reach $505 billion in 2023, according to an estimate from Optum, United’s health services division. The Institute for Clinical and Economic Review, a nonprofit that analyzes the value of drugs, found in 2020 that the biologic drugs used to treat patients like Mr. McNaughton are often effective but overpriced for their therapeutic benefit. To be judged cost-effective by ICER, the biologics should sell at a steep discount to their current market price, the panel found.
A panel convened by ICER to review its analysis cautioned that insurance coverage “should be structured to prevent situations in which patients are forced to choose a treatment approach on the basis of cost.” ICER also found examples where insurance company policies failed to keep pace with updates to clinical practice guidelines based on emerging research.
United officials did not make the cost of treatment an issue when discussing Mr. McNaughton’s care with Penn State administrators or the family.
Bill Truxal, the president of UnitedHealthcare StudentResources, the company’s student health plan division, told a Penn State official that the insurer wanted the “best for the student” and it had “nothing to do with cost,” according to notes the official took of the conversation.
Behind the scenes, however, the price of Mr. McNaughton’s care was front and center at United.
In one email, Mr. Opperman asked about the cost difference if the insurer insisted on paying only for greatly reduced doses of the biologic drugs. Ms. Kavanaugh responded that the insurer had paid $1.1 million in claims for Mr. McNaughton’s care as of the middle of May 2021. If the reduced doses had been in place, the amount would have been cut to $260,218, she wrote.
United was keeping close tabs on Mr. McNaughton at the highest levels of the company. On Aug. 2, 2021, Mr. Opperman notified Mr. Truxal and a United lawyer that Mr. McNaughton “has just purchased the plan again for the 21-22 school year.”
A month later, Ms. Kavanaugh shared another calculation with United executives showing that the insurer spent over $1.7 million on Mr. McNaughton in the prior plan year.
United officials strategized about how to best explain why it was reviewing Mr. McNaughton’s drug regimen, according to an internal email. They pointed to a justification often used by health insurers when denying claims. “As the cost of healthcare continues to climb to soaring heights, it has been determined that a judicious review of these drugs should be included” in order to “make healthcare more affordable for our members,” Ms. Kavanaugh offered as a potential talking point in an April 23, 2021, email.
Three days later, UnitedHealth Group filed an annual statement with the U.S. Securities and Exchange Commission disclosing its pay for top executives in the prior year. Then-CEO David Wichmann was paid $17.9 million in salary and other compensation in 2020. Wichmann retired early the following year, and his total compensation that year exceeded $140 million, according to calculations in a compensation database maintained by the Star Tribune in Minneapolis. The newspaper said the amount was the most paid to an executive in the state since it started tracking pay more than 2 decades ago. About $110 million of that total came from Wichmann exercising stock options accumulated during his stewardship.
The McNaughtons were well aware of the financial situation at United. They looked at publicly available financial results and annual reports. Last year, United reported a profit of $20.1 billion on revenues of $324.2 billion.
When discussing the case with Penn State, Ms. Light said, she told university administrators that United could pay for a year of her son’s treatment using just minutes’ worth of profit.
‘Betrayed’
Mr. McNaughton has been able to continue receiving his infusions for now, anyway. In October, United notified him it was once again reviewing his care, although the insurer quickly reversed course when his lawyer intervened. United, in a court filing, said the review was a mistake and that it had erred in putting Mr. McNaughton’s claims into pending status.
Mr. McNaughton said he is fortunate his parents were employed at the same school he was attending, which was critical in getting the attention of administrators there. But that help had its limits.
In June 2021, just a week after United told Mr. McNaughton it would not cover his treatment plan in the upcoming plan year, Penn State essentially walked away from the matter.
In an email to the McNaughtons and United, Penn State Associate Vice President for Student Affairs Andrea Dowhower wrote that administrators “have observed an unfortunate breakdown in communication” between Mr. McNaughton and his family and the university health insurance plan, “which appears from our perspective to have resulted in a standstill between the two parties.” While she proposed some potential steps to help settle the matter, she wrote that “Penn State’s role in this process is as a resource for students like Chris who, for whatever reason, have experienced difficulty navigating the complex world of health insurance.” The university’s role “is limited,” she wrote, and the school “simply must leave” the issue of the best treatment for Mr. McNaughton to “the appropriate health care professionals.”
In a statement, a Penn State spokesperson wrote that “as a third party in this arrangement, the University’s role is limited and Penn State officials can only help a student manage an issue based on information that a student/family, medical personnel, and/or insurance provider give – with the hope that all information is accurate and that the lines of communication remain open between the insured and the insurer.”
Penn State declined to provide financial information about the plan. However, the university and United share at least one tie that they have not publicly disclosed.
When the McNaughtons first reached out to the university for help, they were referred to the school’s student health insurance coordinator. The official, Heather Klinger, wrote in an email to the family in February 2021 that “I appreciate your trusting me to resolve this for you.”
In April 2022, United began paying Ms. Klinger’s salary, an arrangement which is not noted on the university website. Ms. Klinger appears in the online staff directory on the Penn State University Health Services web page, and has a university phone number, a university address, and a Penn State email listed as her contact. The school said she has maintained a part-time status with the university to allow her to access relevant data systems at both the university and United.
The university said students “benefit” from having a United employee to handle questions about insurance coverage and that the arrangement is “not uncommon” for student health plans.
The family was dismayed to learn that Ms. Klinger was now a full-time employee of United.
“We did feel betrayed,” Ms. Light said. Ms. Klinger did not respond to an email seeking comment.
Mr. McNaughton’s fight to maintain his treatment regimen has come at a cost of time, debilitating stress, and depression. “My biggest fear is realizing I might have to do this every year of my life,” he said.
Mr. McNaughton said one motivation for his lawsuit was to expose how insurers like United make decisions about what care they will pay for and what they will not. The case remains pending, a court docket shows.
He has been accepted to Penn State’s law school. He hopes to become a health care lawyer working for patients who find themselves in situations similar to his.
He plans to re-enroll in the United health care plan when he starts school next fall.
This story was originally published on ProPublica. ProPublica is a nonprofit newsroom that investigates abuses of power. Sign up to receive the biggest stories as soon as they’re published.
In May 2021, a nurse at UnitedHealthcare called a colleague to share some welcome news about a problem the two had been grappling with for weeks.
United provided the health insurance plan for students at Penn State University. It was a large and potentially lucrative account: lots of young, healthy students paying premiums in, not too many huge medical reimbursements going out.
But Christopher McNaughton suffered from a crippling case of ulcerative colitis – an ailment that caused him to develop severe arthritis, debilitating diarrhea, numbing fatigue, and life-threatening blood clots. His medical bills were running nearly $2 million a year.
United had flagged Mr. McNaughton’s case as a “high dollar account,” and the company was reviewing whether it needed to keep paying for the expensive cocktail of drugs crafted by a Mayo Clinic specialist that had brought Mr. McNaughton’s disease under control after he’d been through years of misery.
On the 2021 phone call, which was recorded by the company, nurse Victoria Kavanaugh told her colleague that a doctor contracted by United to review the case had concluded that Mr. McNaughton’s treatment was “not medically necessary.” Her colleague, Dave Opperman, reacted to the news with a long laugh.
“I knew that was coming,” said Mr. Opperman, who heads up a United subsidiary that brokered the health insurance contract between United and Penn State. “I did too,” Ms. Kavanaugh replied.
Mr. Opperman then complained about Mr. McNaughton’s mother, whom he referred to as “this woman,” for “screaming and yelling” and “throwing tantrums” during calls with United.
The pair agreed that any appeal of the United doctor’s denial of the treatment would be a waste of the family’s time and money.
“We’re still gonna say no,” Mr. Opperman said.
More than 200 million Americans are covered by private health insurance. But data from state and federal regulators shows that insurers reject about 1 in 7 claims for treatment. Many people, faced with fighting insurance companies, simply give up: One study found that Americans file formal appeals on only 0.1% of claims denied by insurers under the Affordable Care Act.
Insurers have wide discretion in crafting what is covered by their policies, beyond some basic services mandated by federal and state law. They often deny claims for services that they deem not “medically necessary.”
When United refused to pay for Mr. McNaughton’s treatment for that reason, his family did something unusual. They fought back with a lawsuit, which uncovered a trove of materials, including internal emails and tape-recorded exchanges among company employees. Those records offer an extraordinary behind-the-scenes look at how one of America’s leading health care insurers relentlessly fought to reduce spending on care, even as its profits rose to record levels.
As United reviewed Mr. McNaughton’s treatment, he and his family were often in the dark about what was happening or their rights. Meanwhile, United employees misrepresented critical findings and ignored warnings from doctors about the risks of altering Mr. McNaughton’s drug plan.
At one point, court records show, United inaccurately reported to Penn State and the family that Mr. McNaughton’s doctor had agreed to lower the doses of his medication. Another time, a doctor paid by United concluded that denying payments for Mr. McNaughton’s treatment could put his health at risk, but the company buried his report and did not consider its findings. The insurer did, however, consider a report submitted by a company doctor who rubber-stamped the recommendation of a United nurse to reject paying for the treatment.
United declined to answer specific questions about the case, even after Mr. McNaughton signed a release provided by the insurer to allow it to discuss details of his interactions with the company. United noted that it ultimately paid for all of Mr. McNaughton’s treatments. In a written response, United spokesperson Maria Gordon Shydlo wrote that the company’s guiding concern was Mr. McNaughton’s well-being.
“Mr. McNaughton’s treatment involves medication dosages that far exceed [Food and Drug Administration] guidelines,” the statement said. “In cases like this, we review treatment plans based on current clinical guidelines to help ensure patient safety.”
But the records reviewed by ProPublica show that United had another, equally urgent goal in dealing with Mr. McNaughton. In emails, officials calculated what Mr. McNaughton was costing them to keep his crippling disease at bay and how much they would save if they forced him to undergo a cheaper treatment that had already failed him. As the family pressed the company to back down, first through Penn State and then through a lawsuit, the United officials handling the case bristled.
“This is just unbelievable,” Ms. Kavanaugh said of Mr. McNaughton’s family in one call to discuss his case. ”They’re just really pushing the envelope, and I’m surprised, like I don’t even know what to say.”
The same meal every day
Now 31, Mr. McNaughton grew up in State College, Pa., just blocks from the Penn State campus. Both of his parents are faculty members at the university.
In the winter of 2014, Mr. McNaughton was halfway through his junior year at Bard College in New York. At 6 feet, 4 inches tall, he was a guard on the basketball team and had started most of the team’s games since the start of his sophomore year. He was majoring in psychology.
When Mr. McNaughton returned to school after the winter holiday break, he started to experience frequent bouts of bloody diarrhea. After just a few days on campus, he went home to State College, where doctors diagnosed him with a severe case of ulcerative colitis.
A chronic inflammatory bowel disease that causes swelling and ulcers in the digestive tract, ulcerative colitis has no cure, and ongoing treatment is needed to alleviate symptoms and prevent serious health complications. The majority of cases produce mild to moderate symptoms. Mr. McNaughton’s case was severe.
Treatments for ulcerative colitis include steroids and special drugs known as biologics that work to reduce inflammation in the large intestine.
Mr. McNaughton, however, failed to get meaningful relief from the drugs his doctors initially prescribed. He was experiencing bloody diarrhea up to 20 times a day, with such severe stomach pain that he spent much of his day curled up on a couch. He had little appetite and lost 50 pounds. Severe anemia left him fatigued. He suffered from other conditions related to his colitis, including crippling arthritis. He was hospitalized several times to treat dangerous blood clots.
For 2 years, in an effort to help alleviate his symptoms, he ate the same meals every day: Rice Chex cereal and scrambled eggs for breakfast, a cup of white rice with plain chicken breast for lunch, and a similar meal for dinner, occasionally swapping in tilapia.
His hometown doctors referred him to a specialist at the University of Pittsburgh, who tried unsuccessfully to bring his disease under control. That doctor ended up referring Mr. McNaughton to Edward V. Loftus Jr., MD, at the Mayo Clinic in Rochester, Minn., which has been ranked as the best gastroenterology hospital in the country every year since 1990 by U.S. News & World Report.
For his first visit with Dr. Loftus in May 2015, Mr. McNaughton and his mother, Janice Light, charted hospitals along the 900-mile drive from Pennsylvania to Minnesota in case they needed medical help along the way.
Mornings were the hardest. Mr. McNaughton often spent several hours in the bathroom at the start of the day. To prepare for his meeting with Dr. Loftus, he set his alarm for 3:30 a.m. so he could be ready for the 7:30 a.m. appointment. Even with that preparation, he had to stop twice to use a bathroom on the 5-minute walk from the hotel to the clinic. When they met, Dr. Loftus looked at Mr. McNaughton and told him that he appeared incapacitated. It was, he told the student, as if Mr. McNaughton were chained to the bathroom, with no outside life. He had not been able to return to school and spent most days indoors, managing his symptoms as best he could.
Mr. McNaughton had tried a number of medications by this point, none of which worked. This pattern would repeat itself during the first couple of years that Dr. Loftus treated him.
In addition to trying to find a treatment that would bring Mr. McNaughton’s colitis into remission, Dr. Loftus wanted to wean him off the steroid prednisone, which he had been taking since his initial diagnosis in 2014. The drug is commonly prescribed to colitis patients to control inflammation, but prolonged use can lead to severe side effects including cataracts, osteoporosis, increased risk of infection, and fatigue. Mr. McNaughton also experienced “moon face,” a side effect caused by the shifting of fat deposits that results in the face becoming puffy and rounder.
In 2018, Dr. Loftus and Mr. McNaughton decided to try an unusual regimen. Many patients with inflammatory bowel diseases such as colitis take a single biologic drug as treatment. Whereas traditional drugs are chemically synthesized, biologics are manufactured in living systems, such as plant or animal cells. A year’s supply of an individual biologic drug can cost up to $500,000. They are often given through infusions in a medical facility, which adds to the cost.
Mr. McNaughton had tried individual biologics, and then two in combination, without much success. He and Dr. Loftus then agreed to try two biologic drugs together at doses well above those recommended by the Food and Drug Administration. The federal Agency for Healthcare Research and Quality estimates one in five prescriptions written today are for off-label uses.
There are drawbacks to the practice. Since some uses and doses of particular drugs have not been extensively studied, the risks and efficacy of using them off-label are not well known. Also, some drug manufacturers have improperly pushed off-label usage of their products to boost sales despite little or no evidence to support their use in those situations. Like many leading experts and researchers in his field, Dr. Loftus has been paid to do consulting related to the biologic drugs taken by Mr. McNaughton. The payments related to those drugs have ranged from a total of $1,440 in 2020 to $51,235 in 2018. Dr. Loftus said much of his work with pharmaceutical companies was related to conducting clinical trials on new drugs.
In cases of off-label prescribing, patients are depending upon their doctors’ expertise and experience with the drug. “In this case, I was comfortable that the potential benefits to Chris outweighed the risks,” Dr. Loftus said.
There was evidence that the treatment plan for Mr. McNaughton might work, including studies that had found dual biologic therapy to be efficacious and safe. The two drugs he takes, Entyvio and Remicade, have the same purpose – to reduce inflammation in the large intestine – but each works differently in the body. Remicade, marketed by Janssen Biotech, targets a protein that causes inflammation. Entyvio, made by Takeda Pharmaceuticals, works by preventing an excess of white blood cells from entering into the gastrointestinal tract.
As for any suggestion by United doctors that his treatment plan for Mr. McNaughton was out of bounds or dangerous, Dr. Loftus said “my treatment of Chris was not clinically inappropriate – as was shown by Chris’ positive outcome.”
The unusual high-dose combination of two biologic drugs produced a remarkable change in Mr. McNaughton. He no longer had blood in his stool, and his trips to the bathroom were cut from 20 times a day to 3 or 4. He was able to eat different foods and put on weight. He had more energy. He tapered off prednisone.
“If you told me in 2015 that I would be living like this, I would have asked where do I sign up,” Mr. McNaughton said of the change he experienced with the new drug regimen.
When he first started the new treatment, Mr. McNaughton was covered under his family’s plan, and all his bills were paid. Mr. McNaughton enrolled at the university in 2020. Before switching to United’s plan for students, Mr. McNaughton and his parents consulted with a health advocacy service offered to faculty members. A benefits specialist assured them the drugs taken by Mr. McNaughton would be covered by United.
Mr. McNaughton joined the student plan in July 2020, and his infusions that month and the following month were paid for by United. In September, the insurer indicated payment on his claims was “pending,” something it did for his other claims that came in during the rest of the year.
Mr. McNaughton and his family were worried. They called United to make sure there wasn’t a problem; the insurer told them, they said, that it only needed to check his medical records. When the family called again, United told them it had the documentation needed, they said. United, in a court filing last year, said it received two calls from the family and each time indicated that all of the necessary medical records had not yet been received.
In January 2021, Mr. McNaughton received a new explanation of benefits for the prior months. All of the claims for his care, beginning in September, were no longer “pending.” They were stamped “DENIED.” The total outstanding bill for his treatment was $807,086.
When Mr. McNaughton’s mother reached a United customer service representative the next day to ask why bills that had been paid in the summer were being denied for the fall, the representative told her the account was being reviewed because of “a high dollar amount on the claims,” according to a recording of the call.
Misrepresentations
With United refusing to pay, the family was terrified of being stuck with medical bills that would bankrupt them and deprive Mr. McNaughton of treatment that they considered miraculous.
They turned to Penn State for help. Ms. Light and Mr. McNaughton’s father, David McNaughton, hoped their position as faculty members would make the school more willing to intervene on their behalf.
“After more than 30 years on faculty, my husband and I know that this is not how Penn State would want its students to be treated,” Ms. Light wrote to a school official in February 2021.
In response to questions from ProPublica, Penn State spokesperson Lisa Powers wrote that “supporting the health and well-being of our students is always of primary importance” and that “our hearts go out to any student and family impacted by a serious medical condition.” The university, she wrote, does “not comment on students’ individual circumstances or disclose information from their records.” Mr. McNaughton offered to grant Penn State whatever permissions it needed to speak about his case with ProPublica. The school, however, wrote that it would not comment “even if confidentiality has been waived.”
The family appealed to school administrators. Because the effectiveness of biologics wanes in some patients if doses are skipped, Mr. McNaughton and his parents were worried about even a delay in treatment. His doctor wrote that if he missed scheduled infusions of the drugs, there was “a high likelihood they would no longer be effective.”
During a conference call arranged by Penn State officials on March 5, 2021, United agreed to pay for Mr. McNaughton’s care through the end of the plan year that August. Penn State immediately notified the family of the “wonderful news” while also apologizing for “the stress this has caused Chris and your family.”
Behind the scenes, Mr. McNaughton’s review had “gone all the way to the top” at United’s student health plan division, Ms. Kavanaugh, the nurse, said in a recorded conversation.
The family’s relief was short-lived. A month later, United started another review of Mr. McNaughton’s care, overseen by Ms. Kavanaugh, to determine if it would pay for the treatment in the upcoming plan year.
The nurse sent the Mr. McNaughton case to a company called Medical Review Institute of America. Insurers often turn to companies like MRIoA to review coverage decisions involving expensive treatments or specialized care.
Ms. Kavanaugh, who was assigned to a special investigations unit at United, let her feelings about the matter be known in a recorded telephone call with a representative of MRIoA.
“This school apparently is a big client of ours,” she said. She then shared her opinion of Mr. McNaughton’s treatment. “Really this is a case of a kid who’s getting a drug way too much, like too much of a dose,” Ms. Kavanaugh said. She said it was “insane that they would even think that this is reasonable” and “to be honest with you, they’re awfully pushy considering that we are paying through the end of this school year.”
On a call with an outside contractor, the United nurse claimed Mr. McNaughton was on a higher dose of medication than the FDA approved, which is a common practice.
MRIoA sent the case to Vikas Pabby, MD, a gastroenterologist at UCLA Health and a professor at the university’s medical school. His May 2021 review of Mr. McNaughton’s case was just one of more than 300 Dr. Pabby did for MRIoA that month, for which he was paid $23,000 in total, according to a log of his work produced in the lawsuit.
In a May 4, 2021, report, Dr. Pabby concluded Mr. McNaughton’s treatment was not medically necessary, because United’s policies for the two drugs taken by Mr. McNaughton did not support using them in combination.
Insurers spell out what services they cover in plan policies, lengthy documents that can be confusing and difficult to understand. Many policies, such as Mr. McNaughton’s, contain a provision that treatments and procedures must be “medically necessary” in order to be covered. The definition of medically necessary differs by plan. Some don’t even define the term. Mr. McNaughton’s policy contains a five-part definition, including that the treatment must be “in accordance with the standards of good medical policy” and “the most appropriate supply or level of service which can be safely provided.”
Behind the scenes at United, Mr. Opperman and Ms. Kavanaugh agreed that if Mr. McNaughton were to appeal Dr. Pabby’s decision, the insurer would simply rule against him. “I just think it’s a waste of money and time to appeal and send it to another one when we know we’re gonna get the same answer,” Mr. Opperman said, according to a recording in court files. At Mr. Opperman’s urging, United decided to skip the usual appeals process and arrange for Dr. Pabby to have a so-called “peer-to-peer” discussion with Dr. Loftus, the Mayo physician treating Mr. McNaughton. Such a conversation, in which a patient’s doctor talks with an insurance company’s doctor to advocate for the prescribed treatment, usually occurs only after a customer has appealed a denial and the appeal has been rejected.
When Ms. Kavanaugh called Dr. Loftus’ office to set up a conversation with Dr. Pabby, she explained it was an urgent matter and had been requested by Mr. McNaughton. “You know I’ve just gotten to know Christopher,” she explained, although she had never spoken with him. “We’re trying to advocate and help and get this peer-to-peer set up.”
Mr. McNaughton, meanwhile, had no idea at the time that a United doctor had decided his treatment was unnecessary and that the insurer was trying to set up a phone call with his physician.
In the peer-to-peer conversation, Dr. Loftus told Dr. Pabby that Mr. McNaughton had “a very complicated case” and that lower doses had not worked for him, according to an internal MRIoA memo.
Following his conversation with Dr. Loftus, Dr. Pabby created a second report for United. He recommended the insurer pay for both drugs, but at reduced doses. He added new language saying that the safety of using both drugs at the higher levels “is not established.”
When Ms. Kavanaugh shared the May 12 decision from Dr. Pabby with others at United, her boss responded with an email calling it “great news.”
Then Mr. Opperman sent an email that puzzled the McNaughtons.
In it, Mr. Opperman claimed that Dr. Loftus and Dr. Pabby had agreed that Mr. McNaughton should be on significantly lower doses of both drugs. He said Dr. Loftus “will work with the patient to start titrating them down to a normal dose range.” Mr. Opperman wrote that United would cover Mr. McNaughton’s treatment in the coming year, but only at the reduced doses. Mr. Opperman did not respond to emails and phone messages seeking comment.
Mr. McNaughton didn’t believe a word of it. He had already tried and failed treatment with those drugs at lower doses, and it was Dr. Loftus who had upped the doses, leading to his remission from severe colitis.
The only thing that made sense to Mr. McNaughton was that the treatment United said it would now pay for was dramatically cheaper – saving the company at least hundreds of thousands of dollars a year – than his prescribed treatment because it sliced the size of the doses by more than half.
When the family contacted Dr. Loftus for an explanation, they were outraged by what they heard. Dr. Loftus told them that he had never recommended lowering the dosage. In a letter, Dr. Loftus wrote that changing Mr. McNaughton’s treatment “would have serious detrimental effects on both his short term and long term health and could potentially involve life threatening complications. This would ultimately incur far greater medical costs. Chris was on the doses suggested by United Healthcare before, and they were not at all effective.”
It would not be until the lawsuit that it would become clear how Dr. Loftus’ conversations had been so seriously misrepresented.
Under questioning by Mr. McNaughton’s lawyers, Ms. Kavanaugh acknowledged that she was the source of the incorrect claim that Mr. McNaughton’s doctor had agreed to a change in treatment.
“I incorrectly made an assumption that they had come to some sort of agreement,” she said in a deposition last August. “It was my first peer-to-peer. I did not realize that that simply does not occur.”
Ms. Kavanaugh did not respond to emails and telephone messages seeking comment.
When the McNaughtons first learned of Mr. Opperman’s inaccurate report of the phone call with Dr. Loftus, it unnerved them. They started to question if their case would be fairly reviewed.
“When we got the denial and they lied about what Dr. Loftus said, it just hit me that none of this matters,” Mr. McNaughton said. “They will just say or do anything to get rid of me. It delegitimized the entire review process. When I got that denial, I was crushed.”
A buried report
While the family tried to sort out the inaccurate report, United continued putting the McNaughton case in front of more company doctors.
On May 21, 2021, United sent the case to one of its own doctors, Nady Cates, MD, for an additional review. The review was marked “escalated issue.” Dr. Cates is a United medical director, a title used by many insurers for physicians who review cases. It is work he has been doing as an employee of health insurers since 1989 and at United since 2010. He has not practiced medicine since the early 1990s.
Dr. Cates, in a deposition, said he stopped seeing patients because of the long hours involved and because “AIDS was coming around then. I was seeing a lot of military folks who had venereal diseases, and I guess I was concerned about being exposed.” He transitioned to reviewing paperwork for the insurance industry, he said, because “I guess I was a chicken.”
When he had practiced, Dr. Cates said, he hadn’t treated patients with ulcerative colitis and had referred those cases to a gastroenterologist.
He said his review of Mr. McNaughton’s case primarily involved reading a United nurse’s recommendation to deny his care and making sure “that there wasn’t a decimal place that was out of line.” He said he copied and pasted the nurse’s recommendation and typed “agree” on his review of Mr. McNaughton’s case.
Dr. Cates said that he does about a hundred reviews a week. He said that in his reviews he typically checks to see if any medications are prescribed in accordance with the insurer’s guidelines, and if not, he denies it. United’s policies, he said, prevented him from considering that Mr. McNaughton had failed other treatments or that Dr. Loftus was a leading expert in his field.
“You are giving zero weight to the treating doctor’s opinion on the necessity of the treatment regimen?” a lawyer asked Dr. Cates in his deposition. He responded, “Yeah.”
Attempts to contact Dr. Cates for comment were unsuccessful.
At the same time Dr. Cates was looking at Mr. McNaughton’s case, yet another review was underway at MRIoA. United said it sent the case back to MRIoA after the insurer received the letter from Dr. Loftus warning of the life-threatening complications that might occur if the dosages were reduced.
On May 24, 2021, the new report requested by MRIoA arrived. It came to a completely different conclusion than all of the previous reviews.
Nitin Kumar, MD, a gastroenterologist in Illinois, concluded that Mr. McNaughton’s established treatment plan was not only medically necessary and appropriate but that lowering his doses “can result in a lack of effective therapy of Ulcerative Colitis, with complications of uncontrolled disease (including dysplasia leading to colorectal cancer), flare, hospitalization, need for surgery, and toxic megacolon.”
Unlike other doctors who produced reports for United, Dr. Kumar discussed the harm that Mr. McNaughton might suffer if United required him to change his treatment. “His disease is significantly severe, with diagnosis at a young age,” Dr. Kumar wrote. “He has failed every biologic medication class recommended by guidelines. Therefore, guidelines can no longer be applied in this case.” He cited six studies of patients using two biologic drugs together and wrote that they revealed no significant safety issues and found the therapy to be “broadly successful.”
When Ms. Kavanaugh learned of Dr. Kumar’s report, she quickly moved to quash it and get the case returned to Dr. Pabby, according to her deposition.
In a recorded telephone call, Ms. Kavanaugh told an MRIoA representative that “I had asked that this go back through Dr. Pabby, and it went through a different doctor and they had a much different result.” After further discussion, the MRIoA representative agreed to send the case back to Dr. Pabby. “I appreciate that,” Ms. Kavanaugh replied. “I just want to make sure, because, I mean, it’s obviously a very different result than what we’ve been getting on this case.”
MRIoA case notes show that at 7:04 a.m. on May 25, 2021, Dr. Pabby was assigned to take a look at the case for the third time. At 7:27 a.m., the notes indicate, Dr. Pabby again rejected Mr. McNaughton’s treatment plan. While noting it was “difficult to control” Mr. McNaughton’s ulcerative colitis, Dr. Pabby added that his doses “far exceed what is approved by literature” and that the “safety of the requested doses is not supported by literature.”
In a deposition, Ms. Kavanaugh said that after she opened the Kumar report and read that he was supporting Mr. McNaughton’s current treatment plan, she immediately spoke to her supervisor, who told her to call MRIoA and have the case sent back to Dr. Pabby for review.
Ms. Kavanaugh said she didn’t save a copy of the Kumar report, nor did she forward it to anyone at United or to officials at Penn State who had been inquiring about the McNaughton case. “I didn’t because it shouldn’t have existed,” she said. “It should have gone back to Dr. Pabby.”
When asked if the Kumar report caused her any concerns given his warning that Mr. McNaughton risked cancer or hospitalization if his regimen were changed, Ms. Kavanaugh said she didn’t read his full report. “I saw that it was not the correct doctor, I saw the initial outcome and I was asked to send it back,” she said. Ms. Kavanaugh added, “I have a lot of empathy for this member, but it needed to go back to the peer-to-peer reviewer.”
In a court filing, United said Ms. Kavanaugh was correct in insisting that Dr. Pabby conduct the review and that MRIoA confirmed that Dr. Pabby should have been the one doing the review.
The Kumar report was not provided to Mr. McNaughton when his lawyer, Jonathan M. Gesk, first asked United and MRIoA for any reviews of the case. Mr. Gesk discovered it by accident when he was listening to a recorded telephone call produced by United in which Ms. Kavanaugh mentioned a report number Mr. Gesk had not heard before. He then called MRIoA, which confirmed the report existed and eventually provided it to him.
Dr. Pabby asked ProPublica to direct any questions about his involvement in the matter to MRIoA. The company did not respond to questions from ProPublica about the case.
A sense of hopelessness
When Mr. McNaughton enrolled at Penn State in 2020, it brought a sense of normalcy that he had lost when he was first diagnosed with colitis. He still needed monthly hours-long infusions and suffered occasional flare-ups and symptoms, but he was attending classes in person and living a life similar to the one he had before his diagnosis.
It was a striking contrast to the previous 6 years, which he had spent largely confined to his parents’ house in State College. The frequent bouts of diarrhea made it difficult to go out. He didn’t talk much to friends and spent as much time as he could studying potential treatments and reviewing ongoing clinical trials. He tried to keep up with the occasional online course, but his disease made it difficult to make any real progress toward a degree.
United, in correspondence with Mr. McNaughton, noted that its review of his care was “not a treatment decision. Treatment decisions are made between you and your physician.” But by threatening not to pay for his medications, or only to pay for a different regimen, Mr. McNaughton said, United was in fact attempting to dictate his treatment. From his perspective, the insurer was playing doctor, making decisions without ever examining him or even speaking to him.
The idea of changing his treatment or stopping it altogether caused constant worry for Mr. McNaughton, exacerbating his colitis and triggering physical symptoms, according to his doctors. Those included a large ulcer on his leg and welts under his skin on his thighs and shin that made his leg muscles stiff and painful to the point where he couldn’t bend his leg or walk properly. There were daily migraines and severe stomach pain. “I was consumed with this situation,” Mr. McNaughton said. “My path was unconventional, but I was proud of myself for fighting back and finishing school and getting my life back on track. I thought they were singling me out. My biggest fear was going back to the hell.”
Mr. McNaughton said he contemplated suicide on several occasions, dreading a return to a life where he was housebound or hospitalized.
Mr. McNaughton and his parents talked about his possibly moving to Canada where his grandmother lived and seeking treatment there under the nation’s government health plan.
Dr. Loftus connected Mr. McNaughton with a psychologist who specializes in helping patients with chronic digestive diseases.
The psychologist, Tiffany Taft, PsyD, said Mr. McNaughton was not an unusual case. About one in three patients with diseases like colitis suffer from medical trauma or PTSD related to it, she said, often the result of issues related to getting appropriate treatment approved by insurers.
“You get into hopelessness,” she said of the depression that accompanies fighting with insurance companies over care. “They feel like ‘I can’t fix that. I am screwed.’ When you can’t control things with what an insurance company is doing, anxiety, PTSD and depression get mixed together.”
In the case of Mr. McNaughton, Dr. Taft said, he was being treated by one of the best gastroenterologists in the world, was doing well with his treatment, and then was suddenly notified he might be on the hook for nearly a million dollars in medical charges without access to his medications. “It sends you immediately into panic about all these horrific things that could happen,” Dr. Taft said. The physical and mental symptoms Mr. McNaughton suffered after his care was threatened were “triggered” by the stress he experienced, she said.
In early June 2021, United informed Mr. McNaughton in a letter that it would not cover the cost of his treatment regimen in the next academic year, starting in August. The insurer said it would pay only for a treatment plan that called for a significant reduction in the doses of the drugs he took.
United wrote that the decision came after his “records have been reviewed three times and the medical reviewers have concluded that the medication as prescribed does not meet the Medical Necessity requirement of the plan.”
In August 2021, Mr. McNaughton filed a federal lawsuit accusing United of acting in bad faith and unreasonably making treatment decisions based on financial concerns and not what was the best and most effective treatment. It claims United had a duty to find information that supported Mr. McNaughton’s claim for treatment rather than looking for ways to deny coverage.
United, in a court filing, said it did not breach any duty it owed to Mr. McNaughton and acted in good faith. On Sept. 20, 2021, a month after filing the lawsuit, and with United again balking at paying for his treatment, Mr. McNaughton asked a judge to grant a temporary restraining order requiring United to pay for his care. With the looming threat of a court hearing on the motion, United quickly agreed to cover the cost of Mr. McNaughton’s treatment through the end of the 2021-2022 academic year. It also dropped a demand requiring Mr. McNaughton to settle the matter as a condition of the insurer paying for his treatment as prescribed by Dr. Loftus, according to an email sent by United’s lawyer.
The cost of treatment
It is not surprising that insurers are carefully scrutinizing the care of patients treated with biologics, which are among the most expensive medications on the market. Biologics are considered specialty drugs, a class that includes the best-selling Humira, used to treat arthritis. Specialty drug spending in the United States is expected to reach $505 billion in 2023, according to an estimate from Optum, United’s health services division. The Institute for Clinical and Economic Review, a nonprofit that analyzes the value of drugs, found in 2020 that the biologic drugs used to treat patients like Mr. McNaughton are often effective but overpriced for their therapeutic benefit. To be judged cost-effective by ICER, the biologics should sell at a steep discount to their current market price, the panel found.
A panel convened by ICER to review its analysis cautioned that insurance coverage “should be structured to prevent situations in which patients are forced to choose a treatment approach on the basis of cost.” ICER also found examples where insurance company policies failed to keep pace with updates to clinical practice guidelines based on emerging research.
United officials did not make the cost of treatment an issue when discussing Mr. McNaughton’s care with Penn State administrators or the family.
Bill Truxal, the president of UnitedHealthcare StudentResources, the company’s student health plan division, told a Penn State official that the insurer wanted the “best for the student” and it had “nothing to do with cost,” according to notes the official took of the conversation.
Behind the scenes, however, the price of Mr. McNaughton’s care was front and center at United.
In one email, Mr. Opperman asked about the cost difference if the insurer insisted on paying only for greatly reduced doses of the biologic drugs. Ms. Kavanaugh responded that the insurer had paid $1.1 million in claims for Mr. McNaughton’s care as of the middle of May 2021. If the reduced doses had been in place, the amount would have been cut to $260,218, she wrote.
United was keeping close tabs on Mr. McNaughton at the highest levels of the company. On Aug. 2, 2021, Mr. Opperman notified Mr. Truxal and a United lawyer that Mr. McNaughton “has just purchased the plan again for the 21-22 school year.”
A month later, Ms. Kavanaugh shared another calculation with United executives showing that the insurer spent over $1.7 million on Mr. McNaughton in the prior plan year.
United officials strategized about how to best explain why it was reviewing Mr. McNaughton’s drug regimen, according to an internal email. They pointed to a justification often used by health insurers when denying claims. “As the cost of healthcare continues to climb to soaring heights, it has been determined that a judicious review of these drugs should be included” in order to “make healthcare more affordable for our members,” Ms. Kavanaugh offered as a potential talking point in an April 23, 2021, email.
Three days later, UnitedHealth Group filed an annual statement with the U.S. Securities and Exchange Commission disclosing its pay for top executives in the prior year. Then-CEO David Wichmann was paid $17.9 million in salary and other compensation in 2020. Wichmann retired early the following year, and his total compensation that year exceeded $140 million, according to calculations in a compensation database maintained by the Star Tribune in Minneapolis. The newspaper said the amount was the most paid to an executive in the state since it started tracking pay more than 2 decades ago. About $110 million of that total came from Wichmann exercising stock options accumulated during his stewardship.
The McNaughtons were well aware of the financial situation at United. They looked at publicly available financial results and annual reports. Last year, United reported a profit of $20.1 billion on revenues of $324.2 billion.
When discussing the case with Penn State, Ms. Light said, she told university administrators that United could pay for a year of her son’s treatment using just minutes’ worth of profit.
‘Betrayed’
Mr. McNaughton has been able to continue receiving his infusions for now, anyway. In October, United notified him it was once again reviewing his care, although the insurer quickly reversed course when his lawyer intervened. United, in a court filing, said the review was a mistake and that it had erred in putting Mr. McNaughton’s claims into pending status.
Mr. McNaughton said he is fortunate his parents were employed at the same school he was attending, which was critical in getting the attention of administrators there. But that help had its limits.
In June 2021, just a week after United told Mr. McNaughton it would not cover his treatment plan in the upcoming plan year, Penn State essentially walked away from the matter.
In an email to the McNaughtons and United, Penn State Associate Vice President for Student Affairs Andrea Dowhower wrote that administrators “have observed an unfortunate breakdown in communication” between Mr. McNaughton and his family and the university health insurance plan, “which appears from our perspective to have resulted in a standstill between the two parties.” While she proposed some potential steps to help settle the matter, she wrote that “Penn State’s role in this process is as a resource for students like Chris who, for whatever reason, have experienced difficulty navigating the complex world of health insurance.” The university’s role “is limited,” she wrote, and the school “simply must leave” the issue of the best treatment for Mr. McNaughton to “the appropriate health care professionals.”
In a statement, a Penn State spokesperson wrote that “as a third party in this arrangement, the University’s role is limited and Penn State officials can only help a student manage an issue based on information that a student/family, medical personnel, and/or insurance provider give – with the hope that all information is accurate and that the lines of communication remain open between the insured and the insurer.”
Penn State declined to provide financial information about the plan. However, the university and United share at least one tie that they have not publicly disclosed.
When the McNaughtons first reached out to the university for help, they were referred to the school’s student health insurance coordinator. The official, Heather Klinger, wrote in an email to the family in February 2021 that “I appreciate your trusting me to resolve this for you.”
In April 2022, United began paying Ms. Klinger’s salary, an arrangement which is not noted on the university website. Ms. Klinger appears in the online staff directory on the Penn State University Health Services web page, and has a university phone number, a university address, and a Penn State email listed as her contact. The school said she has maintained a part-time status with the university to allow her to access relevant data systems at both the university and United.
The university said students “benefit” from having a United employee to handle questions about insurance coverage and that the arrangement is “not uncommon” for student health plans.
The family was dismayed to learn that Ms. Klinger was now a full-time employee of United.
“We did feel betrayed,” Ms. Light said. Ms. Klinger did not respond to an email seeking comment.
Mr. McNaughton’s fight to maintain his treatment regimen has come at a cost of time, debilitating stress, and depression. “My biggest fear is realizing I might have to do this every year of my life,” he said.
Mr. McNaughton said one motivation for his lawsuit was to expose how insurers like United make decisions about what care they will pay for and what they will not. The case remains pending, a court docket shows.
He has been accepted to Penn State’s law school. He hopes to become a health care lawyer working for patients who find themselves in situations similar to his.
He plans to re-enroll in the United health care plan when he starts school next fall.
This story was originally published on ProPublica. ProPublica is a nonprofit newsroom that investigates abuses of power. Sign up to receive the biggest stories as soon as they’re published.
STROKE AF at 3 years: High AFib rate after atherosclerotic stroke
In the STROKE AF study, among patients who had a stroke presumably caused by atherosclerosis, the rate of atrial fibrillation (AFib) was almost 22% at 3 years, as detected by continuous monitoring.
The 3-year results from the study were presented by Lee H. Schwamm, MD, of Massachusetts General Hospital, Boston, at the International Stroke Conference presented by the American Stroke Association, a division of the American Heart Association.
Dr. Schwamm said the high rate of AFib detection in this study suggests that continuous monitoring for AFib should be considered for a larger population of stroke patients, rather than just those with cryptogenic stroke.
“We found a much higher rate of AF[ib] than we expected in this population of patients who have had an atherosclerotic stroke,” Dr. Schwamm said in an interview.
“These AF[ib] occurrences were found by a device, so they are known as ‘device-documented AF[ib].’ The patient is not generally aware of symptoms, but 67% of the AF[ib] episodes lasted for more than 1 hour, showing that this is not trivial AF[ib]. This is meaningful AF[ib],” he said.
Dr. Schwamm said the major question is whether these cases of AFib that are detected with a device warrant treatment with anticoagulation. He noted that, in this study, clinicians decided to provide anticoagulation to 70%-80% of patients in whom AFib was detected.
“If we think it deserves treatment, then we have to look for it. And if we care about finding AF[ib], we have no choice but to monitor continuously,” he said.
“If this data doesn’t convince you that AF[ib] is present in this population, I don’t think any data will. Because it is consistent, it accumulates over time and looks remarkably similar to a set of data that we have all become very comfortable with – the CRYSTAL-AF study in patients with cryptogenic stroke,” he stated.
Dr. Schwamm noted that the STROKE AF trial was not based on the cause of the index stroke; rather, it was asking whether there are risk factors that could contribute to the 25% stroke recurrence rate in this population that are not covered in current guidelines.
“I’m really trying to move away from the anchor that I was trained in, which is to figure out the cause of the last stroke to help decide how to prevent the next stroke, towards more of a probabilistic model – of what is all the information I have at my disposal and how do I act on it to prevent the next stroke? We have to start thinking differently about building models for future stroke risk and determining therapy based on that,” he commented.
Changing practice
ISC 2023 program chair Tudor Jovin, MD, Cooper Neurological Institute, Cherry Hill, N.J., and moderator of the session at which the results were presented, discussed the STROKE AF results in a highlights presentation.
“To me as clinician, these results are even more relevant than those at 12 months,” Dr. Jovin said. “The lesson I took is that AF[ib] is even more prevalent than we thought. The burden of AF[ib] is significant in these patients, and it doesn’t seem to be limited to a particular time. These are very thought-provoking results which are going to change clinical practice. I think the threshold for long-term monitoring will be lower.”
Comoderator Lauren Sansing, MD, Yale University, New Haven, Conn., added: “This study shows that the longer we monitor, the more patients with AF[ib] we are likely to pick up. And because in two-thirds of patients with AF[ib], it lasted longer than 1 hour, I do believe this was clinically relevant AF[ib]. The question now is, do we monitor everyone? I think it puts the burden on us to search for AF[ib] in our patients.”
In his presentation, Dr. Schwamm explained that, on the basis of the CRYSTAL-AF study, insertable cardiac monitoring devices are frequently used to identify poststroke AFib in patients with cryptogenic stroke. In the device-monitored arm of that study, AFib was detected in 12.4% of patients over 12 months versus 2.0% in the control arm.
“However, we don’t know how often AF[ib] is detected in other presumed stroke types – largely those due to atherosclerosis,” he said.
He pointed out that, at present, long-term monitoring post stroke for the detection of AFib is not currently recommended for patients with ischemic stroke, owing to presumed small-vessel occlusion or large-artery atherosclerosis.
“In these patients, we are not suspecting AF[ib] because we believe the cause of the stroke was not embolic. But we wanted to investigate what the AF[ib] risk is in these patients, who often have multiple stroke risk factors,” he said.
The trial enrolled 496 patients at 33 centers in the United States. Eligible patients were aged 60 years or older or aged 50-59 years with at least one additional stroke risk factor and had an index stroke that was attributed to large-artery or small-vessel disease. Patients were randomly assigned either to continuous monitoring with the Reveal LINQ device (Medtronic) or to the control arm following site-specific standard of care for AFib detection.
Dr. Schwamm noted that usual care for these patients normally involves monitoring for just a few days while in hospital, but this picks up less than 5% of AFib occurrences.
Baseline characteristics of patients in the STROKE AF study showed that the enrolled population was at high risk for stroke, with a CHADSVASC score of 5. But the index strokes were generally small; the median National Institutes of Health Stroke Scale score was 2.
Results at 12 months, reported 2 years ago, showed a 12.5% incidence of AFib with continuous monitoring versus 1.8% with standard of care (hazard ratio, 7.7; P < .001), rates similar to that found in the CRYSTAL-AF study.
By 3 years, the rate of detected AFib had risen to 21.7% in the continuous monitoring arm versus 2.4% in the control arm (HR, 10.0; P < .001).
“At 12 months, we were seven times more likely to detect AF[ib] with continuous monitoring in these patients, and by 3 years, it was 10 times more likely that AF would be detected with continuous monitoring. I think we’ve settled the question of the best way to find AF[ib] in these patients – it is with an inserted device,” Dr. Schwamm said.
“We have also shown that this is not a transient rise in AFib after the stroke which then diminishes over the next few years. It is a continuous and progressive detection of AF[ib].”
Dr. Schwamm pointed out that 88% of the recorded AFib episodes were asymptomatic. “So relying on patients self-reporting symptoms when deciding who to monitor is unreliable and not a sensible strategy.”
The median time to the first adjudicated AFib episode at 12-month follow-up was 99 days; at the 3-year follow-up, it was 284 days.
“This shows that 30 days of monitoring with an external patch is not sufficient to exclude the presence of AF[ib]. And this really argues for a strategy of immediate insertion of cardiac monitor placement if your goal is to look for AF[ib],” Dr. Schwamm commented.
Is this clinically relevant AFib?
Dr. Schwamm acknowledged that there is a question of whether device-detected AFib should be thought about in the same way as clinically detected AFib with respect to future stroke risk.
He noted that, in this study, 67.4% of patients for whom AFib was detected by continuous monitoring (31 of 46 patients) had at least one episode of AFib that lasted more than 1 hour.
“This is not a trivial little squiggle of something on an EKG which then goes away. This is of significant duration that the cardiologist who adjudicated these rhythm strips felt confident was AF[ib].”
He added: “AF[ib] lasting more than 1 hour crosses the threshold for most practitioners I know to feel confident in treating the patient with anticoagulation. If it was symptomatic AF, this wouldn’t even be a question.”
Dr. Schwamm made the point that device-detected A AFib F has been accepted as worthy of treatment in patients after cryptogenic stroke.
“If we are honest with ourselves and if we have no hesitation in starting anticoagulation in a patient with cryptogenic stroke who has had device-detected AF 6 months later, should we decide that if the patient has had a lacunar stroke, we can ignore that same device-detected fibrillation?”
He put forward the idea that, at some level, all stroke is cryptogenic. “We never know for sure what the cause was. We have hypotheses, we have associations, but we don’t really know. So how much should we weigh that presumptive etiology in terms of how we interpret a rhythm disturbance of fibrillation?”
When looking for predictors of AFib in this study, the investigators found that patients were more likely to have an episode of AFib detected if they had one of the four following risk factors: congestive heart failure, left atrial enlargement, obesity, or QRS prolongation.
“In patients with any one of those four factors, 30% of those had device-detected AF[ib]. These are same predictors of AF[ib] that we are all accustomed to,” Dr. Schwamm said.
Shared decision-making
Dr. Schwamm said in an interview that, in his practice, for these patients, the decision as to whether to use continuous monitoring is made with the patient through shared decision-making.
“We discuss the chance that they could have AF[ib], and I suggest that it might be worth looking for it, but there are factors to be considered. There is a cost to the device, and reimbursement may depend on insurance coverage. Also, some patients may have strong feelings about having the chip implanted in their body.”
He says implanting the chip is easy. “It takes longer to check in at the front desk than to put the device in. It is injected under the skin. It just needs two stitches and a Band-Aid.” The device connects with a smartphone, and the results are interpreted by a cardiologist.
Dr. Schwamm pointed out that the optimal antithrombotic regimen for these patients in whom AFib is detected remains uncertain and should be the focus of future research.
“Do we just stick to antiplatelet therapy or advance to anticoagulation? In moving to an anticoagulant, are we providing less effective prevention for the atherosclerotic stroke risk at the expense of reducing the AF[ib]-related stroke risk? That may be a reasonable trade-off because we know the disability from AF[ib]-associated stroke is much higher.
“Or perhaps the optimal therapy is aspirin plus low-dose anticoagulant? Or left atrial appendage closure and an antiplatelet for patients at a higher risk of bleeding?” he said. “These are the really important questions we need to start asking.”
He added that he hopes a future study will address these questions, but he noted that it would have to be a large study, that it would have to first identify these patients and then randomly assign them to anticoagulation or to no treatment. “That is quite a major undertaking.”
In the highlights presentation, Dr. Jovin said he was uncertain of which of these patients in whom AFib is detected would benefit from anticoagulation. He said he would also like to see a randomized trial on this. But he added: “This would be challenging, as there is the issue of whether there would be equipoise to allow us to randomize to a placebo.”
Dr. Sansing agreed. “I think it would be a hard sell. I would have to think carefully about randomizing a patient to anticoagulation therapy or no therapy who has been found to have AF[ib].”
Dr. Schwamm noted that the current STROKE-AF study was not designed or powered to detect differences in stroke recurrence rates and that there was no difference in stroke recurrence rates between the two arms. There was also no randomization with regard to treatment; choice of medication was left to the discretion of the treating physician.
But he noted that only for 3 of the 34 patients with recurrent stroke in the continuous-monitor arm was AFib detected prior to the recurrent stroke, and only one of those three was receiving anticoagulation at the time of the recurrent stroke.
“These strokes were occurring in patients who did not have device-detected AF[ib],” Dr. Schwamm said. “This is because the population in this study were loaded with stroke risk factors and are at risk of recurrent stroke, but we don’t have the opportunity in this study to really understand the significance of the recurrent strokes.”
The STROKE AF trial was funded by Medtronic. Dr. Schwamm is a consultant to Medtronic.
A version of this article originally appeared on Medscape.com.
In the STROKE AF study, among patients who had a stroke presumably caused by atherosclerosis, the rate of atrial fibrillation (AFib) was almost 22% at 3 years, as detected by continuous monitoring.
The 3-year results from the study were presented by Lee H. Schwamm, MD, of Massachusetts General Hospital, Boston, at the International Stroke Conference presented by the American Stroke Association, a division of the American Heart Association.
Dr. Schwamm said the high rate of AFib detection in this study suggests that continuous monitoring for AFib should be considered for a larger population of stroke patients, rather than just those with cryptogenic stroke.
“We found a much higher rate of AF[ib] than we expected in this population of patients who have had an atherosclerotic stroke,” Dr. Schwamm said in an interview.
“These AF[ib] occurrences were found by a device, so they are known as ‘device-documented AF[ib].’ The patient is not generally aware of symptoms, but 67% of the AF[ib] episodes lasted for more than 1 hour, showing that this is not trivial AF[ib]. This is meaningful AF[ib],” he said.
Dr. Schwamm said the major question is whether these cases of AFib that are detected with a device warrant treatment with anticoagulation. He noted that, in this study, clinicians decided to provide anticoagulation to 70%-80% of patients in whom AFib was detected.
“If we think it deserves treatment, then we have to look for it. And if we care about finding AF[ib], we have no choice but to monitor continuously,” he said.
“If this data doesn’t convince you that AF[ib] is present in this population, I don’t think any data will. Because it is consistent, it accumulates over time and looks remarkably similar to a set of data that we have all become very comfortable with – the CRYSTAL-AF study in patients with cryptogenic stroke,” he stated.
Dr. Schwamm noted that the STROKE AF trial was not based on the cause of the index stroke; rather, it was asking whether there are risk factors that could contribute to the 25% stroke recurrence rate in this population that are not covered in current guidelines.
“I’m really trying to move away from the anchor that I was trained in, which is to figure out the cause of the last stroke to help decide how to prevent the next stroke, towards more of a probabilistic model – of what is all the information I have at my disposal and how do I act on it to prevent the next stroke? We have to start thinking differently about building models for future stroke risk and determining therapy based on that,” he commented.
Changing practice
ISC 2023 program chair Tudor Jovin, MD, Cooper Neurological Institute, Cherry Hill, N.J., and moderator of the session at which the results were presented, discussed the STROKE AF results in a highlights presentation.
“To me as clinician, these results are even more relevant than those at 12 months,” Dr. Jovin said. “The lesson I took is that AF[ib] is even more prevalent than we thought. The burden of AF[ib] is significant in these patients, and it doesn’t seem to be limited to a particular time. These are very thought-provoking results which are going to change clinical practice. I think the threshold for long-term monitoring will be lower.”
Comoderator Lauren Sansing, MD, Yale University, New Haven, Conn., added: “This study shows that the longer we monitor, the more patients with AF[ib] we are likely to pick up. And because in two-thirds of patients with AF[ib], it lasted longer than 1 hour, I do believe this was clinically relevant AF[ib]. The question now is, do we monitor everyone? I think it puts the burden on us to search for AF[ib] in our patients.”
In his presentation, Dr. Schwamm explained that, on the basis of the CRYSTAL-AF study, insertable cardiac monitoring devices are frequently used to identify poststroke AFib in patients with cryptogenic stroke. In the device-monitored arm of that study, AFib was detected in 12.4% of patients over 12 months versus 2.0% in the control arm.
“However, we don’t know how often AF[ib] is detected in other presumed stroke types – largely those due to atherosclerosis,” he said.
He pointed out that, at present, long-term monitoring post stroke for the detection of AFib is not currently recommended for patients with ischemic stroke, owing to presumed small-vessel occlusion or large-artery atherosclerosis.
“In these patients, we are not suspecting AF[ib] because we believe the cause of the stroke was not embolic. But we wanted to investigate what the AF[ib] risk is in these patients, who often have multiple stroke risk factors,” he said.
The trial enrolled 496 patients at 33 centers in the United States. Eligible patients were aged 60 years or older or aged 50-59 years with at least one additional stroke risk factor and had an index stroke that was attributed to large-artery or small-vessel disease. Patients were randomly assigned either to continuous monitoring with the Reveal LINQ device (Medtronic) or to the control arm following site-specific standard of care for AFib detection.
Dr. Schwamm noted that usual care for these patients normally involves monitoring for just a few days while in hospital, but this picks up less than 5% of AFib occurrences.
Baseline characteristics of patients in the STROKE AF study showed that the enrolled population was at high risk for stroke, with a CHADSVASC score of 5. But the index strokes were generally small; the median National Institutes of Health Stroke Scale score was 2.
Results at 12 months, reported 2 years ago, showed a 12.5% incidence of AFib with continuous monitoring versus 1.8% with standard of care (hazard ratio, 7.7; P < .001), rates similar to that found in the CRYSTAL-AF study.
By 3 years, the rate of detected AFib had risen to 21.7% in the continuous monitoring arm versus 2.4% in the control arm (HR, 10.0; P < .001).
“At 12 months, we were seven times more likely to detect AF[ib] with continuous monitoring in these patients, and by 3 years, it was 10 times more likely that AF would be detected with continuous monitoring. I think we’ve settled the question of the best way to find AF[ib] in these patients – it is with an inserted device,” Dr. Schwamm said.
“We have also shown that this is not a transient rise in AFib after the stroke which then diminishes over the next few years. It is a continuous and progressive detection of AF[ib].”
Dr. Schwamm pointed out that 88% of the recorded AFib episodes were asymptomatic. “So relying on patients self-reporting symptoms when deciding who to monitor is unreliable and not a sensible strategy.”
The median time to the first adjudicated AFib episode at 12-month follow-up was 99 days; at the 3-year follow-up, it was 284 days.
“This shows that 30 days of monitoring with an external patch is not sufficient to exclude the presence of AF[ib]. And this really argues for a strategy of immediate insertion of cardiac monitor placement if your goal is to look for AF[ib],” Dr. Schwamm commented.
Is this clinically relevant AFib?
Dr. Schwamm acknowledged that there is a question of whether device-detected AFib should be thought about in the same way as clinically detected AFib with respect to future stroke risk.
He noted that, in this study, 67.4% of patients for whom AFib was detected by continuous monitoring (31 of 46 patients) had at least one episode of AFib that lasted more than 1 hour.
“This is not a trivial little squiggle of something on an EKG which then goes away. This is of significant duration that the cardiologist who adjudicated these rhythm strips felt confident was AF[ib].”
He added: “AF[ib] lasting more than 1 hour crosses the threshold for most practitioners I know to feel confident in treating the patient with anticoagulation. If it was symptomatic AF, this wouldn’t even be a question.”
Dr. Schwamm made the point that device-detected A AFib F has been accepted as worthy of treatment in patients after cryptogenic stroke.
“If we are honest with ourselves and if we have no hesitation in starting anticoagulation in a patient with cryptogenic stroke who has had device-detected AF 6 months later, should we decide that if the patient has had a lacunar stroke, we can ignore that same device-detected fibrillation?”
He put forward the idea that, at some level, all stroke is cryptogenic. “We never know for sure what the cause was. We have hypotheses, we have associations, but we don’t really know. So how much should we weigh that presumptive etiology in terms of how we interpret a rhythm disturbance of fibrillation?”
When looking for predictors of AFib in this study, the investigators found that patients were more likely to have an episode of AFib detected if they had one of the four following risk factors: congestive heart failure, left atrial enlargement, obesity, or QRS prolongation.
“In patients with any one of those four factors, 30% of those had device-detected AF[ib]. These are same predictors of AF[ib] that we are all accustomed to,” Dr. Schwamm said.
Shared decision-making
Dr. Schwamm said in an interview that, in his practice, for these patients, the decision as to whether to use continuous monitoring is made with the patient through shared decision-making.
“We discuss the chance that they could have AF[ib], and I suggest that it might be worth looking for it, but there are factors to be considered. There is a cost to the device, and reimbursement may depend on insurance coverage. Also, some patients may have strong feelings about having the chip implanted in their body.”
He says implanting the chip is easy. “It takes longer to check in at the front desk than to put the device in. It is injected under the skin. It just needs two stitches and a Band-Aid.” The device connects with a smartphone, and the results are interpreted by a cardiologist.
Dr. Schwamm pointed out that the optimal antithrombotic regimen for these patients in whom AFib is detected remains uncertain and should be the focus of future research.
“Do we just stick to antiplatelet therapy or advance to anticoagulation? In moving to an anticoagulant, are we providing less effective prevention for the atherosclerotic stroke risk at the expense of reducing the AF[ib]-related stroke risk? That may be a reasonable trade-off because we know the disability from AF[ib]-associated stroke is much higher.
“Or perhaps the optimal therapy is aspirin plus low-dose anticoagulant? Or left atrial appendage closure and an antiplatelet for patients at a higher risk of bleeding?” he said. “These are the really important questions we need to start asking.”
He added that he hopes a future study will address these questions, but he noted that it would have to be a large study, that it would have to first identify these patients and then randomly assign them to anticoagulation or to no treatment. “That is quite a major undertaking.”
In the highlights presentation, Dr. Jovin said he was uncertain of which of these patients in whom AFib is detected would benefit from anticoagulation. He said he would also like to see a randomized trial on this. But he added: “This would be challenging, as there is the issue of whether there would be equipoise to allow us to randomize to a placebo.”
Dr. Sansing agreed. “I think it would be a hard sell. I would have to think carefully about randomizing a patient to anticoagulation therapy or no therapy who has been found to have AF[ib].”
Dr. Schwamm noted that the current STROKE-AF study was not designed or powered to detect differences in stroke recurrence rates and that there was no difference in stroke recurrence rates between the two arms. There was also no randomization with regard to treatment; choice of medication was left to the discretion of the treating physician.
But he noted that only for 3 of the 34 patients with recurrent stroke in the continuous-monitor arm was AFib detected prior to the recurrent stroke, and only one of those three was receiving anticoagulation at the time of the recurrent stroke.
“These strokes were occurring in patients who did not have device-detected AF[ib],” Dr. Schwamm said. “This is because the population in this study were loaded with stroke risk factors and are at risk of recurrent stroke, but we don’t have the opportunity in this study to really understand the significance of the recurrent strokes.”
The STROKE AF trial was funded by Medtronic. Dr. Schwamm is a consultant to Medtronic.
A version of this article originally appeared on Medscape.com.
In the STROKE AF study, among patients who had a stroke presumably caused by atherosclerosis, the rate of atrial fibrillation (AFib) was almost 22% at 3 years, as detected by continuous monitoring.
The 3-year results from the study were presented by Lee H. Schwamm, MD, of Massachusetts General Hospital, Boston, at the International Stroke Conference presented by the American Stroke Association, a division of the American Heart Association.
Dr. Schwamm said the high rate of AFib detection in this study suggests that continuous monitoring for AFib should be considered for a larger population of stroke patients, rather than just those with cryptogenic stroke.
“We found a much higher rate of AF[ib] than we expected in this population of patients who have had an atherosclerotic stroke,” Dr. Schwamm said in an interview.
“These AF[ib] occurrences were found by a device, so they are known as ‘device-documented AF[ib].’ The patient is not generally aware of symptoms, but 67% of the AF[ib] episodes lasted for more than 1 hour, showing that this is not trivial AF[ib]. This is meaningful AF[ib],” he said.
Dr. Schwamm said the major question is whether these cases of AFib that are detected with a device warrant treatment with anticoagulation. He noted that, in this study, clinicians decided to provide anticoagulation to 70%-80% of patients in whom AFib was detected.
“If we think it deserves treatment, then we have to look for it. And if we care about finding AF[ib], we have no choice but to monitor continuously,” he said.
“If this data doesn’t convince you that AF[ib] is present in this population, I don’t think any data will. Because it is consistent, it accumulates over time and looks remarkably similar to a set of data that we have all become very comfortable with – the CRYSTAL-AF study in patients with cryptogenic stroke,” he stated.
Dr. Schwamm noted that the STROKE AF trial was not based on the cause of the index stroke; rather, it was asking whether there are risk factors that could contribute to the 25% stroke recurrence rate in this population that are not covered in current guidelines.
“I’m really trying to move away from the anchor that I was trained in, which is to figure out the cause of the last stroke to help decide how to prevent the next stroke, towards more of a probabilistic model – of what is all the information I have at my disposal and how do I act on it to prevent the next stroke? We have to start thinking differently about building models for future stroke risk and determining therapy based on that,” he commented.
Changing practice
ISC 2023 program chair Tudor Jovin, MD, Cooper Neurological Institute, Cherry Hill, N.J., and moderator of the session at which the results were presented, discussed the STROKE AF results in a highlights presentation.
“To me as clinician, these results are even more relevant than those at 12 months,” Dr. Jovin said. “The lesson I took is that AF[ib] is even more prevalent than we thought. The burden of AF[ib] is significant in these patients, and it doesn’t seem to be limited to a particular time. These are very thought-provoking results which are going to change clinical practice. I think the threshold for long-term monitoring will be lower.”
Comoderator Lauren Sansing, MD, Yale University, New Haven, Conn., added: “This study shows that the longer we monitor, the more patients with AF[ib] we are likely to pick up. And because in two-thirds of patients with AF[ib], it lasted longer than 1 hour, I do believe this was clinically relevant AF[ib]. The question now is, do we monitor everyone? I think it puts the burden on us to search for AF[ib] in our patients.”
In his presentation, Dr. Schwamm explained that, on the basis of the CRYSTAL-AF study, insertable cardiac monitoring devices are frequently used to identify poststroke AFib in patients with cryptogenic stroke. In the device-monitored arm of that study, AFib was detected in 12.4% of patients over 12 months versus 2.0% in the control arm.
“However, we don’t know how often AF[ib] is detected in other presumed stroke types – largely those due to atherosclerosis,” he said.
He pointed out that, at present, long-term monitoring post stroke for the detection of AFib is not currently recommended for patients with ischemic stroke, owing to presumed small-vessel occlusion or large-artery atherosclerosis.
“In these patients, we are not suspecting AF[ib] because we believe the cause of the stroke was not embolic. But we wanted to investigate what the AF[ib] risk is in these patients, who often have multiple stroke risk factors,” he said.
The trial enrolled 496 patients at 33 centers in the United States. Eligible patients were aged 60 years or older or aged 50-59 years with at least one additional stroke risk factor and had an index stroke that was attributed to large-artery or small-vessel disease. Patients were randomly assigned either to continuous monitoring with the Reveal LINQ device (Medtronic) or to the control arm following site-specific standard of care for AFib detection.
Dr. Schwamm noted that usual care for these patients normally involves monitoring for just a few days while in hospital, but this picks up less than 5% of AFib occurrences.
Baseline characteristics of patients in the STROKE AF study showed that the enrolled population was at high risk for stroke, with a CHADSVASC score of 5. But the index strokes were generally small; the median National Institutes of Health Stroke Scale score was 2.
Results at 12 months, reported 2 years ago, showed a 12.5% incidence of AFib with continuous monitoring versus 1.8% with standard of care (hazard ratio, 7.7; P < .001), rates similar to that found in the CRYSTAL-AF study.
By 3 years, the rate of detected AFib had risen to 21.7% in the continuous monitoring arm versus 2.4% in the control arm (HR, 10.0; P < .001).
“At 12 months, we were seven times more likely to detect AF[ib] with continuous monitoring in these patients, and by 3 years, it was 10 times more likely that AF would be detected with continuous monitoring. I think we’ve settled the question of the best way to find AF[ib] in these patients – it is with an inserted device,” Dr. Schwamm said.
“We have also shown that this is not a transient rise in AFib after the stroke which then diminishes over the next few years. It is a continuous and progressive detection of AF[ib].”
Dr. Schwamm pointed out that 88% of the recorded AFib episodes were asymptomatic. “So relying on patients self-reporting symptoms when deciding who to monitor is unreliable and not a sensible strategy.”
The median time to the first adjudicated AFib episode at 12-month follow-up was 99 days; at the 3-year follow-up, it was 284 days.
“This shows that 30 days of monitoring with an external patch is not sufficient to exclude the presence of AF[ib]. And this really argues for a strategy of immediate insertion of cardiac monitor placement if your goal is to look for AF[ib],” Dr. Schwamm commented.
Is this clinically relevant AFib?
Dr. Schwamm acknowledged that there is a question of whether device-detected AFib should be thought about in the same way as clinically detected AFib with respect to future stroke risk.
He noted that, in this study, 67.4% of patients for whom AFib was detected by continuous monitoring (31 of 46 patients) had at least one episode of AFib that lasted more than 1 hour.
“This is not a trivial little squiggle of something on an EKG which then goes away. This is of significant duration that the cardiologist who adjudicated these rhythm strips felt confident was AF[ib].”
He added: “AF[ib] lasting more than 1 hour crosses the threshold for most practitioners I know to feel confident in treating the patient with anticoagulation. If it was symptomatic AF, this wouldn’t even be a question.”
Dr. Schwamm made the point that device-detected A AFib F has been accepted as worthy of treatment in patients after cryptogenic stroke.
“If we are honest with ourselves and if we have no hesitation in starting anticoagulation in a patient with cryptogenic stroke who has had device-detected AF 6 months later, should we decide that if the patient has had a lacunar stroke, we can ignore that same device-detected fibrillation?”
He put forward the idea that, at some level, all stroke is cryptogenic. “We never know for sure what the cause was. We have hypotheses, we have associations, but we don’t really know. So how much should we weigh that presumptive etiology in terms of how we interpret a rhythm disturbance of fibrillation?”
When looking for predictors of AFib in this study, the investigators found that patients were more likely to have an episode of AFib detected if they had one of the four following risk factors: congestive heart failure, left atrial enlargement, obesity, or QRS prolongation.
“In patients with any one of those four factors, 30% of those had device-detected AF[ib]. These are same predictors of AF[ib] that we are all accustomed to,” Dr. Schwamm said.
Shared decision-making
Dr. Schwamm said in an interview that, in his practice, for these patients, the decision as to whether to use continuous monitoring is made with the patient through shared decision-making.
“We discuss the chance that they could have AF[ib], and I suggest that it might be worth looking for it, but there are factors to be considered. There is a cost to the device, and reimbursement may depend on insurance coverage. Also, some patients may have strong feelings about having the chip implanted in their body.”
He says implanting the chip is easy. “It takes longer to check in at the front desk than to put the device in. It is injected under the skin. It just needs two stitches and a Band-Aid.” The device connects with a smartphone, and the results are interpreted by a cardiologist.
Dr. Schwamm pointed out that the optimal antithrombotic regimen for these patients in whom AFib is detected remains uncertain and should be the focus of future research.
“Do we just stick to antiplatelet therapy or advance to anticoagulation? In moving to an anticoagulant, are we providing less effective prevention for the atherosclerotic stroke risk at the expense of reducing the AF[ib]-related stroke risk? That may be a reasonable trade-off because we know the disability from AF[ib]-associated stroke is much higher.
“Or perhaps the optimal therapy is aspirin plus low-dose anticoagulant? Or left atrial appendage closure and an antiplatelet for patients at a higher risk of bleeding?” he said. “These are the really important questions we need to start asking.”
He added that he hopes a future study will address these questions, but he noted that it would have to be a large study, that it would have to first identify these patients and then randomly assign them to anticoagulation or to no treatment. “That is quite a major undertaking.”
In the highlights presentation, Dr. Jovin said he was uncertain of which of these patients in whom AFib is detected would benefit from anticoagulation. He said he would also like to see a randomized trial on this. But he added: “This would be challenging, as there is the issue of whether there would be equipoise to allow us to randomize to a placebo.”
Dr. Sansing agreed. “I think it would be a hard sell. I would have to think carefully about randomizing a patient to anticoagulation therapy or no therapy who has been found to have AF[ib].”
Dr. Schwamm noted that the current STROKE-AF study was not designed or powered to detect differences in stroke recurrence rates and that there was no difference in stroke recurrence rates between the two arms. There was also no randomization with regard to treatment; choice of medication was left to the discretion of the treating physician.
But he noted that only for 3 of the 34 patients with recurrent stroke in the continuous-monitor arm was AFib detected prior to the recurrent stroke, and only one of those three was receiving anticoagulation at the time of the recurrent stroke.
“These strokes were occurring in patients who did not have device-detected AF[ib],” Dr. Schwamm said. “This is because the population in this study were loaded with stroke risk factors and are at risk of recurrent stroke, but we don’t have the opportunity in this study to really understand the significance of the recurrent strokes.”
The STROKE AF trial was funded by Medtronic. Dr. Schwamm is a consultant to Medtronic.
A version of this article originally appeared on Medscape.com.
FROM ISC 2023
Differences in brain structure linked to social disadvantage
Brain volume disparities among young children of different races may be attributable to adverse childhood experiences related to socioeconomic conditions and structural racism, new research suggests.
Investigators from the Belmont, Mass.–based McLean Hospital, an affiliate of Mass General Brigham, found that 9- and 10-year-old children of different racial and socioeconomic backgrounds have subtle neurobiological differences in gray matter volume in certain brain regions associated with trauma and stress.
Lead investigator Nathaniel Harnett, PhD, of the department of psychiatry at Harvard Medical School, Boston, believes this research shows evidence that “structural racism” – broad socioeconomic disadvantages that lead to poverty and emotional trauma – may affect brain structures and growth and ultimately may lead to psychiatric illness.
“For clinicians, I think the take-home message is that we really need to be more aware about the ways in which the disproportionate burden of stress might impact some groups,” Dr. Harnett told this news organization.
“This in turn can affect the way they respond either to later stress or maybe even treatment outcomes.” He added that other brain regions and compensatory mechanisms are likely to be involved, and more work needs to explore these connections.
The study was published online in the American Journal of Psychiatry.
‘Toxic stressor’
Dr. Harnett and colleagues used MRI and survey data from the 2019 Adolescent Brain Cognitive Development (ABCD) study involving over 12,000 children from 21 sites across the United States.
Participating children provided information about emotional and physical conflicts in the household. The ABCD study also surveyed the parents about their race and ethnicity, parental education, employment, and family income. Another factor in the analysis was neighborhood disadvantage, based on the Area Deprivation Index utilizing 17 socioeconomic indicators from the U.S. Census, including poverty and housing.
Comparing brain MRI findings from approximately 7,300 White children and 1,800 Black children in the ABCD study, Dr. Harnett’s group found that Black children had lower gray matter volume in the amygdala, hippocampus, and other subregions of the prefrontal cortex.
Experience of adversity was the “sole factor” explaining brain volume differences, with household income being the predominant factor.
Compared with White children, Black children were three times less likely to have parents who were currently employed. In addition, White parents were more likely than Black parents to have higher education at 75.2% versus 40.6%. Black families had significantly lower household income than White families and experienced more family conflict, material hardship, neighborhood disadvantage, and traumatic events.
The researchers analyzed race-related differences in posttraumatic stress disorder symptoms and the relationship with adversity and found that Black children had significantly greater PTSD symptom severity, and that symptom severity was “further predicted by adversity.”
“Taken together, early-life adversity may act as a toxic stressor that disproportionately impacts Black children as a result of their significantly greater exposure to adversity and contributes to differential neural development of key threat-processing regions,” the investigators write.
“These parts of the brain are involved in what we typically call threat learning,” Dr. Harnett explained. “Threat learning is basically learning to recognize potential dangers in our environment and selecting behaviors to keep us safe, whether we’re going to run away from a danger or face it head on. When you have chronic exposure to things that can be dangerous or can make you feel unsafe, that might have an impact on how these brain regions develop, with potential implications for how these regions function later on in life.”
A consequence of toxic stress
This study is part of a growing body of work on the influence of “toxic stress” and other forms of PTSD on brain architecture. The authors note that prolonged exposure to adverse experiences leads to excessive activation of stress-response systems and accumulation of stress hormones. This disrupts immune and metabolic regulatory systems that influence the developing structures of the brain.
The study helps to contradict the “pseudoscientific falsehood” of biologic race-related differences in brain volume, instead emphasizing the role of adversity brought on by structural racism, the authors add.
In an accompanying editor’s note, the publication’s Editor-in-Chief Ned H. Kalin, MD, called childhood adversity, maltreatment, and stress, “significant risk factors for the development of psychopathology.”
These findings are “critically important, as they speak to the need for psychiatry as a field to be outspoken about the detrimental psychological impacts of race-related disparities in childhood adversity, to call out the fact that these disparities stem from structural racism, and to vigorously support rectifying efforts by pursuing policy changes,” he stated in a news release.
Social construct?
Joan Luby, MD, coauthor of an accompanying editorial, said she and her coauthor “really appreciate the study and think the findings are overall very consistent with the emerging literature, increasing the confidence [in the findings].”
Dr. Luby, a professor of child psychiatry and director of the Early Emotional Development Program, Washington University, St. Louis, noted that she “takes issue” with the fact that the study “makes inferences regarding race, when we think those inferences aren’t well justified, are misinterpretations, and could be misleading.”
Race is a “social construct” and there are many sources of adversity that the authors didn’t measure in the study and are likely the source of any remaining variance they found, including experiences of structural racism and discrimination,” said Dr. Luby, who was not involved in the study.
“How people look doesn’t have any bearing on their inherent biological characteristics, and more [needs to be studied] on how they experience the psychosocial environment and how the psychosocial environment rejects or reacts to them.”
These psychosocial issues “have to be taken into account and measured in a very comprehensive way,” she added.
The ABCD study was supported by the National Institutes of Health and additional federal partners. Dr. Harnett reports no relevant financial relationships. The other authors’ disclosures are listed on the original paper. Dr. Luby receives royalties from Guilford Press. Her coauthor reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Brain volume disparities among young children of different races may be attributable to adverse childhood experiences related to socioeconomic conditions and structural racism, new research suggests.
Investigators from the Belmont, Mass.–based McLean Hospital, an affiliate of Mass General Brigham, found that 9- and 10-year-old children of different racial and socioeconomic backgrounds have subtle neurobiological differences in gray matter volume in certain brain regions associated with trauma and stress.
Lead investigator Nathaniel Harnett, PhD, of the department of psychiatry at Harvard Medical School, Boston, believes this research shows evidence that “structural racism” – broad socioeconomic disadvantages that lead to poverty and emotional trauma – may affect brain structures and growth and ultimately may lead to psychiatric illness.
“For clinicians, I think the take-home message is that we really need to be more aware about the ways in which the disproportionate burden of stress might impact some groups,” Dr. Harnett told this news organization.
“This in turn can affect the way they respond either to later stress or maybe even treatment outcomes.” He added that other brain regions and compensatory mechanisms are likely to be involved, and more work needs to explore these connections.
The study was published online in the American Journal of Psychiatry.
‘Toxic stressor’
Dr. Harnett and colleagues used MRI and survey data from the 2019 Adolescent Brain Cognitive Development (ABCD) study involving over 12,000 children from 21 sites across the United States.
Participating children provided information about emotional and physical conflicts in the household. The ABCD study also surveyed the parents about their race and ethnicity, parental education, employment, and family income. Another factor in the analysis was neighborhood disadvantage, based on the Area Deprivation Index utilizing 17 socioeconomic indicators from the U.S. Census, including poverty and housing.
Comparing brain MRI findings from approximately 7,300 White children and 1,800 Black children in the ABCD study, Dr. Harnett’s group found that Black children had lower gray matter volume in the amygdala, hippocampus, and other subregions of the prefrontal cortex.
Experience of adversity was the “sole factor” explaining brain volume differences, with household income being the predominant factor.
Compared with White children, Black children were three times less likely to have parents who were currently employed. In addition, White parents were more likely than Black parents to have higher education at 75.2% versus 40.6%. Black families had significantly lower household income than White families and experienced more family conflict, material hardship, neighborhood disadvantage, and traumatic events.
The researchers analyzed race-related differences in posttraumatic stress disorder symptoms and the relationship with adversity and found that Black children had significantly greater PTSD symptom severity, and that symptom severity was “further predicted by adversity.”
“Taken together, early-life adversity may act as a toxic stressor that disproportionately impacts Black children as a result of their significantly greater exposure to adversity and contributes to differential neural development of key threat-processing regions,” the investigators write.
“These parts of the brain are involved in what we typically call threat learning,” Dr. Harnett explained. “Threat learning is basically learning to recognize potential dangers in our environment and selecting behaviors to keep us safe, whether we’re going to run away from a danger or face it head on. When you have chronic exposure to things that can be dangerous or can make you feel unsafe, that might have an impact on how these brain regions develop, with potential implications for how these regions function later on in life.”
A consequence of toxic stress
This study is part of a growing body of work on the influence of “toxic stress” and other forms of PTSD on brain architecture. The authors note that prolonged exposure to adverse experiences leads to excessive activation of stress-response systems and accumulation of stress hormones. This disrupts immune and metabolic regulatory systems that influence the developing structures of the brain.
The study helps to contradict the “pseudoscientific falsehood” of biologic race-related differences in brain volume, instead emphasizing the role of adversity brought on by structural racism, the authors add.
In an accompanying editor’s note, the publication’s Editor-in-Chief Ned H. Kalin, MD, called childhood adversity, maltreatment, and stress, “significant risk factors for the development of psychopathology.”
These findings are “critically important, as they speak to the need for psychiatry as a field to be outspoken about the detrimental psychological impacts of race-related disparities in childhood adversity, to call out the fact that these disparities stem from structural racism, and to vigorously support rectifying efforts by pursuing policy changes,” he stated in a news release.
Social construct?
Joan Luby, MD, coauthor of an accompanying editorial, said she and her coauthor “really appreciate the study and think the findings are overall very consistent with the emerging literature, increasing the confidence [in the findings].”
Dr. Luby, a professor of child psychiatry and director of the Early Emotional Development Program, Washington University, St. Louis, noted that she “takes issue” with the fact that the study “makes inferences regarding race, when we think those inferences aren’t well justified, are misinterpretations, and could be misleading.”
Race is a “social construct” and there are many sources of adversity that the authors didn’t measure in the study and are likely the source of any remaining variance they found, including experiences of structural racism and discrimination,” said Dr. Luby, who was not involved in the study.
“How people look doesn’t have any bearing on their inherent biological characteristics, and more [needs to be studied] on how they experience the psychosocial environment and how the psychosocial environment rejects or reacts to them.”
These psychosocial issues “have to be taken into account and measured in a very comprehensive way,” she added.
The ABCD study was supported by the National Institutes of Health and additional federal partners. Dr. Harnett reports no relevant financial relationships. The other authors’ disclosures are listed on the original paper. Dr. Luby receives royalties from Guilford Press. Her coauthor reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Brain volume disparities among young children of different races may be attributable to adverse childhood experiences related to socioeconomic conditions and structural racism, new research suggests.
Investigators from the Belmont, Mass.–based McLean Hospital, an affiliate of Mass General Brigham, found that 9- and 10-year-old children of different racial and socioeconomic backgrounds have subtle neurobiological differences in gray matter volume in certain brain regions associated with trauma and stress.
Lead investigator Nathaniel Harnett, PhD, of the department of psychiatry at Harvard Medical School, Boston, believes this research shows evidence that “structural racism” – broad socioeconomic disadvantages that lead to poverty and emotional trauma – may affect brain structures and growth and ultimately may lead to psychiatric illness.
“For clinicians, I think the take-home message is that we really need to be more aware about the ways in which the disproportionate burden of stress might impact some groups,” Dr. Harnett told this news organization.
“This in turn can affect the way they respond either to later stress or maybe even treatment outcomes.” He added that other brain regions and compensatory mechanisms are likely to be involved, and more work needs to explore these connections.
The study was published online in the American Journal of Psychiatry.
‘Toxic stressor’
Dr. Harnett and colleagues used MRI and survey data from the 2019 Adolescent Brain Cognitive Development (ABCD) study involving over 12,000 children from 21 sites across the United States.
Participating children provided information about emotional and physical conflicts in the household. The ABCD study also surveyed the parents about their race and ethnicity, parental education, employment, and family income. Another factor in the analysis was neighborhood disadvantage, based on the Area Deprivation Index utilizing 17 socioeconomic indicators from the U.S. Census, including poverty and housing.
Comparing brain MRI findings from approximately 7,300 White children and 1,800 Black children in the ABCD study, Dr. Harnett’s group found that Black children had lower gray matter volume in the amygdala, hippocampus, and other subregions of the prefrontal cortex.
Experience of adversity was the “sole factor” explaining brain volume differences, with household income being the predominant factor.
Compared with White children, Black children were three times less likely to have parents who were currently employed. In addition, White parents were more likely than Black parents to have higher education at 75.2% versus 40.6%. Black families had significantly lower household income than White families and experienced more family conflict, material hardship, neighborhood disadvantage, and traumatic events.
The researchers analyzed race-related differences in posttraumatic stress disorder symptoms and the relationship with adversity and found that Black children had significantly greater PTSD symptom severity, and that symptom severity was “further predicted by adversity.”
“Taken together, early-life adversity may act as a toxic stressor that disproportionately impacts Black children as a result of their significantly greater exposure to adversity and contributes to differential neural development of key threat-processing regions,” the investigators write.
“These parts of the brain are involved in what we typically call threat learning,” Dr. Harnett explained. “Threat learning is basically learning to recognize potential dangers in our environment and selecting behaviors to keep us safe, whether we’re going to run away from a danger or face it head on. When you have chronic exposure to things that can be dangerous or can make you feel unsafe, that might have an impact on how these brain regions develop, with potential implications for how these regions function later on in life.”
A consequence of toxic stress
This study is part of a growing body of work on the influence of “toxic stress” and other forms of PTSD on brain architecture. The authors note that prolonged exposure to adverse experiences leads to excessive activation of stress-response systems and accumulation of stress hormones. This disrupts immune and metabolic regulatory systems that influence the developing structures of the brain.
The study helps to contradict the “pseudoscientific falsehood” of biologic race-related differences in brain volume, instead emphasizing the role of adversity brought on by structural racism, the authors add.
In an accompanying editor’s note, the publication’s Editor-in-Chief Ned H. Kalin, MD, called childhood adversity, maltreatment, and stress, “significant risk factors for the development of psychopathology.”
These findings are “critically important, as they speak to the need for psychiatry as a field to be outspoken about the detrimental psychological impacts of race-related disparities in childhood adversity, to call out the fact that these disparities stem from structural racism, and to vigorously support rectifying efforts by pursuing policy changes,” he stated in a news release.
Social construct?
Joan Luby, MD, coauthor of an accompanying editorial, said she and her coauthor “really appreciate the study and think the findings are overall very consistent with the emerging literature, increasing the confidence [in the findings].”
Dr. Luby, a professor of child psychiatry and director of the Early Emotional Development Program, Washington University, St. Louis, noted that she “takes issue” with the fact that the study “makes inferences regarding race, when we think those inferences aren’t well justified, are misinterpretations, and could be misleading.”
Race is a “social construct” and there are many sources of adversity that the authors didn’t measure in the study and are likely the source of any remaining variance they found, including experiences of structural racism and discrimination,” said Dr. Luby, who was not involved in the study.
“How people look doesn’t have any bearing on their inherent biological characteristics, and more [needs to be studied] on how they experience the psychosocial environment and how the psychosocial environment rejects or reacts to them.”
These psychosocial issues “have to be taken into account and measured in a very comprehensive way,” she added.
The ABCD study was supported by the National Institutes of Health and additional federal partners. Dr. Harnett reports no relevant financial relationships. The other authors’ disclosures are listed on the original paper. Dr. Luby receives royalties from Guilford Press. Her coauthor reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Be aware of hepatic encephalopathy, dementia overlap in older patients with cirrhosis
, according to a new study involving U.S. veterans.
The overlap between dementia and HE was also independent of alcohol use, brain injury, age, and other metabolic risk factors.
“The aging of patients with cirrhosis leads us to encounter several individuals who may be prone to both of these diseases,” senior author Jasmohan Bajaj, MD, a professor of gastroenterology, hepatology, and nutrition at Virginia Commonwealth University Medical Center and GI section of the Central Virginia Veterans Healthcare System in Richmond, said in an interview.
“Given the epidemic of metabolic syndrome and alcohol, consider excluding cirrhosis in your patient [for] whom the presumptive diagnosis is dementia, since they could have concomitant HE,” he said.
“On the flip side, in those with HE who have predominant long-term memory issues and persistent cognitive changes, consider consulting a neuropsychiatrist or neurologist to ensure there is a resolution of the underlying disease process,” Dr. Bajaj added.
The study was published online in The American Journal of Gastroenterology.
Analyzing associations
HE is a common decompensating event in patients with cirrhosis. Because of the aging population of patients with cirrhosis, however, it’s important to differentiate HE from nonhepatic etiologies of cognitive impairment, such as dementia, the authors note.
Using data from the VA Corporate Data Warehouse, Dr. Bajaj and colleagues identified veterans with cirrhosis who received VA care between October 2019 and September 2021 and compared baseline characteristics between the cohorts based on the presence or absence of dementia. The research team then evaluated factors associated with having a diagnosis of dementia, adjusting for demographics, comorbid illnesses, cirrhosis etiology, and cirrhosis complications.
Investigators identified 71,522 veterans with diagnostic codes for cirrhosis who were engaged in VA care in 2019. They were mostly men (96.2%) and had a median age of 66. The most common etiologies of cirrhosis were alcohol and hepatitis C, followed by nonalcoholic steatohepatitis (NASH). The group also included veterans with predominantly compensated cirrhosis and a median MELD-Na score of 9. The MELD-Na score gauges the severity of chronic liver disease using values such as serum bilirubin, serum creatinine, and the international normalized ratio for prothrombin time and sodium to predict survival.
Among those with cirrhosis, 5,647 (7.9%) also had dementia diagnosis codes. This rate is higher than the prevalence of dementia in the general population and equivalent to the rate of dementia in veterans without cirrhosis who are older than 65, the authors note.
In general, veterans with dementia tended to be older, to be White, to live in an urban area, and to have higher MELD-Na scores, and they were more frequently diagnosed with alcohol-related cirrhosis, alcohol and tobacco use disorder, diabetes, chronic kidney disease, chronic heart failure, brain trauma, and cerebrovascular disease.
In a multivariable analysis, the presence of any decompensating event was significantly associated with dementia. In subsequent analyses of individual decompensating events, however, the strongest association was with HE, while ascites or variceal bleeding did not add to the risk.
When HE was defined as patients who filled prescriptions for lactulose or rifaximin, the frequency of patients with HE decreased from 13.7% to 10.9%. In an analysis with HE as the decompensating event, the association between HE and dementia remained significant compared to when HE was defined by diagnostic codes alone.
“We were surprised by the high proportion of patients with dementia who also had cirrhosis, and given the genuine difficulty that clinicians have with defining HE vs. dementia, we were not very surprised at that overlap,” Dr. Bajaj said.
“We were also surprised at the specificity of this overlap only with HE and not with other decompensating events, which was also independent of head injury, alcohol use, and PTSD,” he added.
Additional research needed
Future research should look at the characteristics of HE, including the number of episodes or breakthrough episodes, and should focus on objective biomarkers to differentiate dementia and HE, the study authors write.
“The distinction and study of potential overlapping features among HE and dementia is important because HE is often treatable with medications and reverses after liver transplant, while this does not occur with dementia,” they add.
Dr. Bajaj and colleagues call for a greater awareness of disease processes and complications in older patients with cirrhosis, particularly since diagnostic imprecision can lead to patient and family confusion, distrust, and ineffective treatment.
The study will help physicians better understand the important overlap between dementia and HE, said Eric Orman, MD, an associate professor of medicine at Indiana University, Indianapolis.
Dr. Orman, who wasn’t involved with this study, has researched recent trends in the characteristics and outcomes of patients with newly diagnosed cirrhosis and has found that the proportion of older adults has increased, as well as those with alcoholic cirrhosis and NASH, which has implications for future patient care.
“It is important to recognize that both dementia and HE can occur either separately or concurrently in individuals with cirrhosis,” Dr. Orman told this news organization. “When seeing patients with cognitive impairment, having a high index of suspicion for both conditions is critical to ensure appropriate diagnosis and treatment.”
The study’s findings “represent the tip of the iceberg,” Neal Parikh, MD, an assistant professor of neurology and neuroscience at Weill Cornell Medicine in New York, said in an interview. “There is a tremendous amount left to be discovered regarding the role of the liver in brain health.”
Dr. Parikh, who wasn’t associated with this study, has researched the impact of chronic liver conditions on cognitive impairment and dementia. He is working on a project that addresses HE in detail.
“There is growing recognition of a so-called ‘liver-brain axis,’ with several researchers, including my group, showing that a range of chronic liver conditions may detrimentally impact cognitive function and increase the risk of dementia,” he said. “Studying the specific contributions of cirrhosis is critical for understanding the role of hepatic encephalopathy in age-related cognitive decline.”
The study received no financial support. The authors reported no potential competing interests.
A version of this article first appeared on Medscape.com.
, according to a new study involving U.S. veterans.
The overlap between dementia and HE was also independent of alcohol use, brain injury, age, and other metabolic risk factors.
“The aging of patients with cirrhosis leads us to encounter several individuals who may be prone to both of these diseases,” senior author Jasmohan Bajaj, MD, a professor of gastroenterology, hepatology, and nutrition at Virginia Commonwealth University Medical Center and GI section of the Central Virginia Veterans Healthcare System in Richmond, said in an interview.
“Given the epidemic of metabolic syndrome and alcohol, consider excluding cirrhosis in your patient [for] whom the presumptive diagnosis is dementia, since they could have concomitant HE,” he said.
“On the flip side, in those with HE who have predominant long-term memory issues and persistent cognitive changes, consider consulting a neuropsychiatrist or neurologist to ensure there is a resolution of the underlying disease process,” Dr. Bajaj added.
The study was published online in The American Journal of Gastroenterology.
Analyzing associations
HE is a common decompensating event in patients with cirrhosis. Because of the aging population of patients with cirrhosis, however, it’s important to differentiate HE from nonhepatic etiologies of cognitive impairment, such as dementia, the authors note.
Using data from the VA Corporate Data Warehouse, Dr. Bajaj and colleagues identified veterans with cirrhosis who received VA care between October 2019 and September 2021 and compared baseline characteristics between the cohorts based on the presence or absence of dementia. The research team then evaluated factors associated with having a diagnosis of dementia, adjusting for demographics, comorbid illnesses, cirrhosis etiology, and cirrhosis complications.
Investigators identified 71,522 veterans with diagnostic codes for cirrhosis who were engaged in VA care in 2019. They were mostly men (96.2%) and had a median age of 66. The most common etiologies of cirrhosis were alcohol and hepatitis C, followed by nonalcoholic steatohepatitis (NASH). The group also included veterans with predominantly compensated cirrhosis and a median MELD-Na score of 9. The MELD-Na score gauges the severity of chronic liver disease using values such as serum bilirubin, serum creatinine, and the international normalized ratio for prothrombin time and sodium to predict survival.
Among those with cirrhosis, 5,647 (7.9%) also had dementia diagnosis codes. This rate is higher than the prevalence of dementia in the general population and equivalent to the rate of dementia in veterans without cirrhosis who are older than 65, the authors note.
In general, veterans with dementia tended to be older, to be White, to live in an urban area, and to have higher MELD-Na scores, and they were more frequently diagnosed with alcohol-related cirrhosis, alcohol and tobacco use disorder, diabetes, chronic kidney disease, chronic heart failure, brain trauma, and cerebrovascular disease.
In a multivariable analysis, the presence of any decompensating event was significantly associated with dementia. In subsequent analyses of individual decompensating events, however, the strongest association was with HE, while ascites or variceal bleeding did not add to the risk.
When HE was defined as patients who filled prescriptions for lactulose or rifaximin, the frequency of patients with HE decreased from 13.7% to 10.9%. In an analysis with HE as the decompensating event, the association between HE and dementia remained significant compared to when HE was defined by diagnostic codes alone.
“We were surprised by the high proportion of patients with dementia who also had cirrhosis, and given the genuine difficulty that clinicians have with defining HE vs. dementia, we were not very surprised at that overlap,” Dr. Bajaj said.
“We were also surprised at the specificity of this overlap only with HE and not with other decompensating events, which was also independent of head injury, alcohol use, and PTSD,” he added.
Additional research needed
Future research should look at the characteristics of HE, including the number of episodes or breakthrough episodes, and should focus on objective biomarkers to differentiate dementia and HE, the study authors write.
“The distinction and study of potential overlapping features among HE and dementia is important because HE is often treatable with medications and reverses after liver transplant, while this does not occur with dementia,” they add.
Dr. Bajaj and colleagues call for a greater awareness of disease processes and complications in older patients with cirrhosis, particularly since diagnostic imprecision can lead to patient and family confusion, distrust, and ineffective treatment.
The study will help physicians better understand the important overlap between dementia and HE, said Eric Orman, MD, an associate professor of medicine at Indiana University, Indianapolis.
Dr. Orman, who wasn’t involved with this study, has researched recent trends in the characteristics and outcomes of patients with newly diagnosed cirrhosis and has found that the proportion of older adults has increased, as well as those with alcoholic cirrhosis and NASH, which has implications for future patient care.
“It is important to recognize that both dementia and HE can occur either separately or concurrently in individuals with cirrhosis,” Dr. Orman told this news organization. “When seeing patients with cognitive impairment, having a high index of suspicion for both conditions is critical to ensure appropriate diagnosis and treatment.”
The study’s findings “represent the tip of the iceberg,” Neal Parikh, MD, an assistant professor of neurology and neuroscience at Weill Cornell Medicine in New York, said in an interview. “There is a tremendous amount left to be discovered regarding the role of the liver in brain health.”
Dr. Parikh, who wasn’t associated with this study, has researched the impact of chronic liver conditions on cognitive impairment and dementia. He is working on a project that addresses HE in detail.
“There is growing recognition of a so-called ‘liver-brain axis,’ with several researchers, including my group, showing that a range of chronic liver conditions may detrimentally impact cognitive function and increase the risk of dementia,” he said. “Studying the specific contributions of cirrhosis is critical for understanding the role of hepatic encephalopathy in age-related cognitive decline.”
The study received no financial support. The authors reported no potential competing interests.
A version of this article first appeared on Medscape.com.
, according to a new study involving U.S. veterans.
The overlap between dementia and HE was also independent of alcohol use, brain injury, age, and other metabolic risk factors.
“The aging of patients with cirrhosis leads us to encounter several individuals who may be prone to both of these diseases,” senior author Jasmohan Bajaj, MD, a professor of gastroenterology, hepatology, and nutrition at Virginia Commonwealth University Medical Center and GI section of the Central Virginia Veterans Healthcare System in Richmond, said in an interview.
“Given the epidemic of metabolic syndrome and alcohol, consider excluding cirrhosis in your patient [for] whom the presumptive diagnosis is dementia, since they could have concomitant HE,” he said.
“On the flip side, in those with HE who have predominant long-term memory issues and persistent cognitive changes, consider consulting a neuropsychiatrist or neurologist to ensure there is a resolution of the underlying disease process,” Dr. Bajaj added.
The study was published online in The American Journal of Gastroenterology.
Analyzing associations
HE is a common decompensating event in patients with cirrhosis. Because of the aging population of patients with cirrhosis, however, it’s important to differentiate HE from nonhepatic etiologies of cognitive impairment, such as dementia, the authors note.
Using data from the VA Corporate Data Warehouse, Dr. Bajaj and colleagues identified veterans with cirrhosis who received VA care between October 2019 and September 2021 and compared baseline characteristics between the cohorts based on the presence or absence of dementia. The research team then evaluated factors associated with having a diagnosis of dementia, adjusting for demographics, comorbid illnesses, cirrhosis etiology, and cirrhosis complications.
Investigators identified 71,522 veterans with diagnostic codes for cirrhosis who were engaged in VA care in 2019. They were mostly men (96.2%) and had a median age of 66. The most common etiologies of cirrhosis were alcohol and hepatitis C, followed by nonalcoholic steatohepatitis (NASH). The group also included veterans with predominantly compensated cirrhosis and a median MELD-Na score of 9. The MELD-Na score gauges the severity of chronic liver disease using values such as serum bilirubin, serum creatinine, and the international normalized ratio for prothrombin time and sodium to predict survival.
Among those with cirrhosis, 5,647 (7.9%) also had dementia diagnosis codes. This rate is higher than the prevalence of dementia in the general population and equivalent to the rate of dementia in veterans without cirrhosis who are older than 65, the authors note.
In general, veterans with dementia tended to be older, to be White, to live in an urban area, and to have higher MELD-Na scores, and they were more frequently diagnosed with alcohol-related cirrhosis, alcohol and tobacco use disorder, diabetes, chronic kidney disease, chronic heart failure, brain trauma, and cerebrovascular disease.
In a multivariable analysis, the presence of any decompensating event was significantly associated with dementia. In subsequent analyses of individual decompensating events, however, the strongest association was with HE, while ascites or variceal bleeding did not add to the risk.
When HE was defined as patients who filled prescriptions for lactulose or rifaximin, the frequency of patients with HE decreased from 13.7% to 10.9%. In an analysis with HE as the decompensating event, the association between HE and dementia remained significant compared to when HE was defined by diagnostic codes alone.
“We were surprised by the high proportion of patients with dementia who also had cirrhosis, and given the genuine difficulty that clinicians have with defining HE vs. dementia, we were not very surprised at that overlap,” Dr. Bajaj said.
“We were also surprised at the specificity of this overlap only with HE and not with other decompensating events, which was also independent of head injury, alcohol use, and PTSD,” he added.
Additional research needed
Future research should look at the characteristics of HE, including the number of episodes or breakthrough episodes, and should focus on objective biomarkers to differentiate dementia and HE, the study authors write.
“The distinction and study of potential overlapping features among HE and dementia is important because HE is often treatable with medications and reverses after liver transplant, while this does not occur with dementia,” they add.
Dr. Bajaj and colleagues call for a greater awareness of disease processes and complications in older patients with cirrhosis, particularly since diagnostic imprecision can lead to patient and family confusion, distrust, and ineffective treatment.
The study will help physicians better understand the important overlap between dementia and HE, said Eric Orman, MD, an associate professor of medicine at Indiana University, Indianapolis.
Dr. Orman, who wasn’t involved with this study, has researched recent trends in the characteristics and outcomes of patients with newly diagnosed cirrhosis and has found that the proportion of older adults has increased, as well as those with alcoholic cirrhosis and NASH, which has implications for future patient care.
“It is important to recognize that both dementia and HE can occur either separately or concurrently in individuals with cirrhosis,” Dr. Orman told this news organization. “When seeing patients with cognitive impairment, having a high index of suspicion for both conditions is critical to ensure appropriate diagnosis and treatment.”
The study’s findings “represent the tip of the iceberg,” Neal Parikh, MD, an assistant professor of neurology and neuroscience at Weill Cornell Medicine in New York, said in an interview. “There is a tremendous amount left to be discovered regarding the role of the liver in brain health.”
Dr. Parikh, who wasn’t associated with this study, has researched the impact of chronic liver conditions on cognitive impairment and dementia. He is working on a project that addresses HE in detail.
“There is growing recognition of a so-called ‘liver-brain axis,’ with several researchers, including my group, showing that a range of chronic liver conditions may detrimentally impact cognitive function and increase the risk of dementia,” he said. “Studying the specific contributions of cirrhosis is critical for understanding the role of hepatic encephalopathy in age-related cognitive decline.”
The study received no financial support. The authors reported no potential competing interests.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF GASTROENTEROLOGY
Algorithm can spot signs of autism in babies, study says
, a study from Duke University, Durham, N.C., says.
“We can use the first 30 days of a child’s health care experience to say, ‘This child is really at risk,’ ” said David Mandell, DSc, a professor of psychiatry at the University of Pennsylvania, Philadelphia, in USA Today. He was not involved in the research.
Researchers analyzed electronic medical records of 45,000 children treated in the Duke University Health System as infants between 2006 and 2020. They created an algorithm that could predict which babies later developed autism. These babies were more likely to have been to an ophthalmologist or neurologist; had stomach or gastrointestinal issues; or received physical therapy.
“A huge number of factors across the infant’s entire health profile” went into the models, said study coauthor Matthew Engelhard, MD, an assistant professor of biostatistics and bioinformatics at Duke University. “Each one of those factors contributes incrementally.”
USA Today said the team “paid particular attention to how the model performed in groups of children who are often overlooked by traditional screening methods and, therefore, miss the advantages of early diagnosis, including girls, children of color, and children with combined diagnoses of autism and ADHD,” according to Dr. Engelhard.
The study could lead to the algorithm being used with other tools to diagnose and help children earlier, said study author Geraldine Dawson, PhD, who directs the Duke Center for Autism and Brain Development.
“We need to be thinking about autism as not only a behavioral health condition but also a condition that involves physical health,” she said. “This is one way to take advantage of that information: in doing a better job at early detection.”
Autism is a complicated condition that includes communication and behavior challenges involving a range of symptoms and skills. It can be minor or a disability that requires full-time care.
A version of this article first appeared on WebMD.com.
, a study from Duke University, Durham, N.C., says.
“We can use the first 30 days of a child’s health care experience to say, ‘This child is really at risk,’ ” said David Mandell, DSc, a professor of psychiatry at the University of Pennsylvania, Philadelphia, in USA Today. He was not involved in the research.
Researchers analyzed electronic medical records of 45,000 children treated in the Duke University Health System as infants between 2006 and 2020. They created an algorithm that could predict which babies later developed autism. These babies were more likely to have been to an ophthalmologist or neurologist; had stomach or gastrointestinal issues; or received physical therapy.
“A huge number of factors across the infant’s entire health profile” went into the models, said study coauthor Matthew Engelhard, MD, an assistant professor of biostatistics and bioinformatics at Duke University. “Each one of those factors contributes incrementally.”
USA Today said the team “paid particular attention to how the model performed in groups of children who are often overlooked by traditional screening methods and, therefore, miss the advantages of early diagnosis, including girls, children of color, and children with combined diagnoses of autism and ADHD,” according to Dr. Engelhard.
The study could lead to the algorithm being used with other tools to diagnose and help children earlier, said study author Geraldine Dawson, PhD, who directs the Duke Center for Autism and Brain Development.
“We need to be thinking about autism as not only a behavioral health condition but also a condition that involves physical health,” she said. “This is one way to take advantage of that information: in doing a better job at early detection.”
Autism is a complicated condition that includes communication and behavior challenges involving a range of symptoms and skills. It can be minor or a disability that requires full-time care.
A version of this article first appeared on WebMD.com.
, a study from Duke University, Durham, N.C., says.
“We can use the first 30 days of a child’s health care experience to say, ‘This child is really at risk,’ ” said David Mandell, DSc, a professor of psychiatry at the University of Pennsylvania, Philadelphia, in USA Today. He was not involved in the research.
Researchers analyzed electronic medical records of 45,000 children treated in the Duke University Health System as infants between 2006 and 2020. They created an algorithm that could predict which babies later developed autism. These babies were more likely to have been to an ophthalmologist or neurologist; had stomach or gastrointestinal issues; or received physical therapy.
“A huge number of factors across the infant’s entire health profile” went into the models, said study coauthor Matthew Engelhard, MD, an assistant professor of biostatistics and bioinformatics at Duke University. “Each one of those factors contributes incrementally.”
USA Today said the team “paid particular attention to how the model performed in groups of children who are often overlooked by traditional screening methods and, therefore, miss the advantages of early diagnosis, including girls, children of color, and children with combined diagnoses of autism and ADHD,” according to Dr. Engelhard.
The study could lead to the algorithm being used with other tools to diagnose and help children earlier, said study author Geraldine Dawson, PhD, who directs the Duke Center for Autism and Brain Development.
“We need to be thinking about autism as not only a behavioral health condition but also a condition that involves physical health,” she said. “This is one way to take advantage of that information: in doing a better job at early detection.”
Autism is a complicated condition that includes communication and behavior challenges involving a range of symptoms and skills. It can be minor or a disability that requires full-time care.
A version of this article first appeared on WebMD.com.
FROM JAMA NETWORK OPEN
Novel neuroprotective agent promising in stroke
preliminary results of a first-in-human study show.
The findings illustrate that it is possible to improve outcomes for stroke patients “not only with reperfusion therapy but with neuroprotectants,” study author Macarena Hernandez, PhD, associate professor, University Complutense, Madrid, told this news organization.
Dr. Hernandez said she hopes these positive results will spur investigation into other neuroprotective agents.
The findings were presented at the International Stroke Conference presented by the American Stroke Association, a division of the American Heart Association.
Best doses
The study investigated ApTOLL, which blocks the TOLL-like receptor 4 (TLR4) that induces inflammation after a stroke. Previous studies found that ApTOLL protected brain tissue in animal models of stroke.
The phase 1B part of the study found no safety issues and determined the best two doses to be used in phase 2A were 0.05 mg/kg and 0.2 mg/kg.
The analysis included 139 patients at 14 centers in Spain and France (mean age, about 70 years; 42% women) who had a large-vessel occlusion and were eligible for endovascular therapy.
“Our aim was to have a very homogeneous population” to try to replicate in humans what had worked in animals, another study author, Marc Ribó, MD, interventional neurologist, Hospital Vall d’Hebron, Barcelona, told this news organization.
Study participants had an Alberta Stroke Program Early CT Score (ASPECTS) of 5-10, and estimated infarct core volume on CT-perfusion was 5-70 mL. All were treated within 6 hours of stroke onset.
Researchers randomly assigned patients to receive the low dose of the drug, the high dose of the drug, or placebo. The drug was administered intravenously over a 30-minute period just prior to the groin puncture for the thrombectomy procedure.
“So, the drug had already started to work when they underwent the usual standard practice, the thrombectomy,” said Dr. Ribó.
Those who were eligible also received tissue plasminogen activator.
The primary endpoint was safety, including death, symptomatic intracranial hemorrhage (SICH), and recurrent stroke.
Lower mortality
At 90 days, there was a statistically significant lower mortality rate in the high-dose group, compared with the group that received placebo (4.76% vs. 18.18%).
The mortality rate was 26.19% in the low-dose group, but Dr. Ribó stressed that this dose was a quarter of the higher dose and so performed “much more like placebo.”
The higher dose also yielded a better SICH outcome (4.76% of patients vs. 7.27% for placebo and 7.14% for the lower dose). And it was superior in terms of brain edema (2.4% of the population vs. 7.3% for the placebo and 4.8% for the low-dose groups).
About 7.1% of the high-dose group, 3.7% of the placebo group, and 4.8% of the low-dose group had a recurrent transient ischemic attack or stroke.
A secondary efficacy endpoint was infarct volume on MRI at 72 hours. Here, for the higher-dose group, mean infarct volume was reduced, compared with the patients who received placebo (–29.31 cc; 90% confidence interval, –49.28 to –9.34).
This higher dose was also superior for the secondary outcome of National Institutes of Health Stroke Scale score at 72 hours and for the disability outcome on the modified Rankin Score (mRS).
Clear shift in disability
“There was a clear shift toward less disability across levels of the mRS score in the high-dose group at 90 days,” said Dr. Ribó.
He added that he and his colleagues are “very happy” with these results, as they reflect “a consistency” of outcomes.
“We observed that the infarct volumes were lower in the high-dose group, and that led to a significant lower NIH score, meaning less clinical neurological symptoms at 72 hours, and finally, this led to less disability at 90 days.”
These results are “very exciting,” Dr. Hernandez added. “This is the first neuroprotectant that has demonstrated this acute effect in reducing deaths, in reducing the infarct volume and improving functionality long-term in patients treated with the higher dose.”
Dr. Ribó noted the treatment would eventually be used in addition to reperfusion therapy. “It’s not competing with reperfusion treatment; it’s an additional layer” of treatment.
Although it would initially be offered only to patients eligible for thrombectomy, researchers will explore the drug’s effectiveness for other stroke patients, said Dr. Ribó. “We wanted to secure this indication, and from there, progressively expand to other profiles of stroke patients, and even to patients with intracranial hemorrhage.”
The study confirmed the safety of the drug. “There were no safety issues at all,” said Dr. Ribó. “We were initially concerned that an anti-inflammatory in these patients could lead to higher rates of infections, but this was absolutely not the case.”
The next step is to confirm the effects in a larger, multicenter study, which is planned to launch at the end of this year, said Dr. Hernandez.
‘Very robust results’
In a comment, Philip B. Gorelick, MD, professor of neurology, Northwestern University, Chicago, said that, while this was a small early-phase study, the results are “very robust.”
“The authors demonstrated proof of a neuroprotective effect; they showed at 90 days that the death rates were substantially reduced by about four times – 4% vs. 18% – and the size of the damaged tissue at about 72 hours was reduced by 40%,” said Dr. Gorelick, who did not participate in the study.
He also noted that the disability was “less pronounced” at 90 days in the 0.2 mg/kg group.
“So overall, these are very encouraging results,” said Dr. Gorelick. “We have had a lot of difficulty finding neuroprotectant drugs that work, and this drug, in combination with endovascular therapy, seems to be very promising.”
However, he stressed the drug “is not ready for prime-time practice.”
“The proof in the pudding will be in the large-scale main phase 3 trials,” he added.
The study was funded by aptaTargets. Dr. Hernandez is chief scientific officer at aptaTargets. Dr. Ribó is an adviser at AptaTargets; a consultant at Medtronic; has ownership interest in Anaconda and NoraHealth; is a consultant for Cerenovus and Philips; and has stock options at Methink. Dr. Gorelick has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
preliminary results of a first-in-human study show.
The findings illustrate that it is possible to improve outcomes for stroke patients “not only with reperfusion therapy but with neuroprotectants,” study author Macarena Hernandez, PhD, associate professor, University Complutense, Madrid, told this news organization.
Dr. Hernandez said she hopes these positive results will spur investigation into other neuroprotective agents.
The findings were presented at the International Stroke Conference presented by the American Stroke Association, a division of the American Heart Association.
Best doses
The study investigated ApTOLL, which blocks the TOLL-like receptor 4 (TLR4) that induces inflammation after a stroke. Previous studies found that ApTOLL protected brain tissue in animal models of stroke.
The phase 1B part of the study found no safety issues and determined the best two doses to be used in phase 2A were 0.05 mg/kg and 0.2 mg/kg.
The analysis included 139 patients at 14 centers in Spain and France (mean age, about 70 years; 42% women) who had a large-vessel occlusion and were eligible for endovascular therapy.
“Our aim was to have a very homogeneous population” to try to replicate in humans what had worked in animals, another study author, Marc Ribó, MD, interventional neurologist, Hospital Vall d’Hebron, Barcelona, told this news organization.
Study participants had an Alberta Stroke Program Early CT Score (ASPECTS) of 5-10, and estimated infarct core volume on CT-perfusion was 5-70 mL. All were treated within 6 hours of stroke onset.
Researchers randomly assigned patients to receive the low dose of the drug, the high dose of the drug, or placebo. The drug was administered intravenously over a 30-minute period just prior to the groin puncture for the thrombectomy procedure.
“So, the drug had already started to work when they underwent the usual standard practice, the thrombectomy,” said Dr. Ribó.
Those who were eligible also received tissue plasminogen activator.
The primary endpoint was safety, including death, symptomatic intracranial hemorrhage (SICH), and recurrent stroke.
Lower mortality
At 90 days, there was a statistically significant lower mortality rate in the high-dose group, compared with the group that received placebo (4.76% vs. 18.18%).
The mortality rate was 26.19% in the low-dose group, but Dr. Ribó stressed that this dose was a quarter of the higher dose and so performed “much more like placebo.”
The higher dose also yielded a better SICH outcome (4.76% of patients vs. 7.27% for placebo and 7.14% for the lower dose). And it was superior in terms of brain edema (2.4% of the population vs. 7.3% for the placebo and 4.8% for the low-dose groups).
About 7.1% of the high-dose group, 3.7% of the placebo group, and 4.8% of the low-dose group had a recurrent transient ischemic attack or stroke.
A secondary efficacy endpoint was infarct volume on MRI at 72 hours. Here, for the higher-dose group, mean infarct volume was reduced, compared with the patients who received placebo (–29.31 cc; 90% confidence interval, –49.28 to –9.34).
This higher dose was also superior for the secondary outcome of National Institutes of Health Stroke Scale score at 72 hours and for the disability outcome on the modified Rankin Score (mRS).
Clear shift in disability
“There was a clear shift toward less disability across levels of the mRS score in the high-dose group at 90 days,” said Dr. Ribó.
He added that he and his colleagues are “very happy” with these results, as they reflect “a consistency” of outcomes.
“We observed that the infarct volumes were lower in the high-dose group, and that led to a significant lower NIH score, meaning less clinical neurological symptoms at 72 hours, and finally, this led to less disability at 90 days.”
These results are “very exciting,” Dr. Hernandez added. “This is the first neuroprotectant that has demonstrated this acute effect in reducing deaths, in reducing the infarct volume and improving functionality long-term in patients treated with the higher dose.”
Dr. Ribó noted the treatment would eventually be used in addition to reperfusion therapy. “It’s not competing with reperfusion treatment; it’s an additional layer” of treatment.
Although it would initially be offered only to patients eligible for thrombectomy, researchers will explore the drug’s effectiveness for other stroke patients, said Dr. Ribó. “We wanted to secure this indication, and from there, progressively expand to other profiles of stroke patients, and even to patients with intracranial hemorrhage.”
The study confirmed the safety of the drug. “There were no safety issues at all,” said Dr. Ribó. “We were initially concerned that an anti-inflammatory in these patients could lead to higher rates of infections, but this was absolutely not the case.”
The next step is to confirm the effects in a larger, multicenter study, which is planned to launch at the end of this year, said Dr. Hernandez.
‘Very robust results’
In a comment, Philip B. Gorelick, MD, professor of neurology, Northwestern University, Chicago, said that, while this was a small early-phase study, the results are “very robust.”
“The authors demonstrated proof of a neuroprotective effect; they showed at 90 days that the death rates were substantially reduced by about four times – 4% vs. 18% – and the size of the damaged tissue at about 72 hours was reduced by 40%,” said Dr. Gorelick, who did not participate in the study.
He also noted that the disability was “less pronounced” at 90 days in the 0.2 mg/kg group.
“So overall, these are very encouraging results,” said Dr. Gorelick. “We have had a lot of difficulty finding neuroprotectant drugs that work, and this drug, in combination with endovascular therapy, seems to be very promising.”
However, he stressed the drug “is not ready for prime-time practice.”
“The proof in the pudding will be in the large-scale main phase 3 trials,” he added.
The study was funded by aptaTargets. Dr. Hernandez is chief scientific officer at aptaTargets. Dr. Ribó is an adviser at AptaTargets; a consultant at Medtronic; has ownership interest in Anaconda and NoraHealth; is a consultant for Cerenovus and Philips; and has stock options at Methink. Dr. Gorelick has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
preliminary results of a first-in-human study show.
The findings illustrate that it is possible to improve outcomes for stroke patients “not only with reperfusion therapy but with neuroprotectants,” study author Macarena Hernandez, PhD, associate professor, University Complutense, Madrid, told this news organization.
Dr. Hernandez said she hopes these positive results will spur investigation into other neuroprotective agents.
The findings were presented at the International Stroke Conference presented by the American Stroke Association, a division of the American Heart Association.
Best doses
The study investigated ApTOLL, which blocks the TOLL-like receptor 4 (TLR4) that induces inflammation after a stroke. Previous studies found that ApTOLL protected brain tissue in animal models of stroke.
The phase 1B part of the study found no safety issues and determined the best two doses to be used in phase 2A were 0.05 mg/kg and 0.2 mg/kg.
The analysis included 139 patients at 14 centers in Spain and France (mean age, about 70 years; 42% women) who had a large-vessel occlusion and were eligible for endovascular therapy.
“Our aim was to have a very homogeneous population” to try to replicate in humans what had worked in animals, another study author, Marc Ribó, MD, interventional neurologist, Hospital Vall d’Hebron, Barcelona, told this news organization.
Study participants had an Alberta Stroke Program Early CT Score (ASPECTS) of 5-10, and estimated infarct core volume on CT-perfusion was 5-70 mL. All were treated within 6 hours of stroke onset.
Researchers randomly assigned patients to receive the low dose of the drug, the high dose of the drug, or placebo. The drug was administered intravenously over a 30-minute period just prior to the groin puncture for the thrombectomy procedure.
“So, the drug had already started to work when they underwent the usual standard practice, the thrombectomy,” said Dr. Ribó.
Those who were eligible also received tissue plasminogen activator.
The primary endpoint was safety, including death, symptomatic intracranial hemorrhage (SICH), and recurrent stroke.
Lower mortality
At 90 days, there was a statistically significant lower mortality rate in the high-dose group, compared with the group that received placebo (4.76% vs. 18.18%).
The mortality rate was 26.19% in the low-dose group, but Dr. Ribó stressed that this dose was a quarter of the higher dose and so performed “much more like placebo.”
The higher dose also yielded a better SICH outcome (4.76% of patients vs. 7.27% for placebo and 7.14% for the lower dose). And it was superior in terms of brain edema (2.4% of the population vs. 7.3% for the placebo and 4.8% for the low-dose groups).
About 7.1% of the high-dose group, 3.7% of the placebo group, and 4.8% of the low-dose group had a recurrent transient ischemic attack or stroke.
A secondary efficacy endpoint was infarct volume on MRI at 72 hours. Here, for the higher-dose group, mean infarct volume was reduced, compared with the patients who received placebo (–29.31 cc; 90% confidence interval, –49.28 to –9.34).
This higher dose was also superior for the secondary outcome of National Institutes of Health Stroke Scale score at 72 hours and for the disability outcome on the modified Rankin Score (mRS).
Clear shift in disability
“There was a clear shift toward less disability across levels of the mRS score in the high-dose group at 90 days,” said Dr. Ribó.
He added that he and his colleagues are “very happy” with these results, as they reflect “a consistency” of outcomes.
“We observed that the infarct volumes were lower in the high-dose group, and that led to a significant lower NIH score, meaning less clinical neurological symptoms at 72 hours, and finally, this led to less disability at 90 days.”
These results are “very exciting,” Dr. Hernandez added. “This is the first neuroprotectant that has demonstrated this acute effect in reducing deaths, in reducing the infarct volume and improving functionality long-term in patients treated with the higher dose.”
Dr. Ribó noted the treatment would eventually be used in addition to reperfusion therapy. “It’s not competing with reperfusion treatment; it’s an additional layer” of treatment.
Although it would initially be offered only to patients eligible for thrombectomy, researchers will explore the drug’s effectiveness for other stroke patients, said Dr. Ribó. “We wanted to secure this indication, and from there, progressively expand to other profiles of stroke patients, and even to patients with intracranial hemorrhage.”
The study confirmed the safety of the drug. “There were no safety issues at all,” said Dr. Ribó. “We were initially concerned that an anti-inflammatory in these patients could lead to higher rates of infections, but this was absolutely not the case.”
The next step is to confirm the effects in a larger, multicenter study, which is planned to launch at the end of this year, said Dr. Hernandez.
‘Very robust results’
In a comment, Philip B. Gorelick, MD, professor of neurology, Northwestern University, Chicago, said that, while this was a small early-phase study, the results are “very robust.”
“The authors demonstrated proof of a neuroprotective effect; they showed at 90 days that the death rates were substantially reduced by about four times – 4% vs. 18% – and the size of the damaged tissue at about 72 hours was reduced by 40%,” said Dr. Gorelick, who did not participate in the study.
He also noted that the disability was “less pronounced” at 90 days in the 0.2 mg/kg group.
“So overall, these are very encouraging results,” said Dr. Gorelick. “We have had a lot of difficulty finding neuroprotectant drugs that work, and this drug, in combination with endovascular therapy, seems to be very promising.”
However, he stressed the drug “is not ready for prime-time practice.”
“The proof in the pudding will be in the large-scale main phase 3 trials,” he added.
The study was funded by aptaTargets. Dr. Hernandez is chief scientific officer at aptaTargets. Dr. Ribó is an adviser at AptaTargets; a consultant at Medtronic; has ownership interest in Anaconda and NoraHealth; is a consultant for Cerenovus and Philips; and has stock options at Methink. Dr. Gorelick has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ISC 2023
How a concussion led a former football player/WWE star to a pioneering neuroscience career
On Oct. 5, 2022, at 10:24 a.m., Chris Nowinski, PhD, cofounder of the Boston-based Concussion Legacy Foundation (CLF), was in his home office when the email came through.
“I pounded my desk, shouted YES! and went to find my wife so I could pick her up and give her a big hug,” he recalled. “It was the culmination of 15 years of research and hard work.”
Robert Cantu, MD, who has been studying head trauma for 50+ years and has published more than 500 papers about it, compares the announcement to the 1964 Surgeon General’s report that linked cigarette smoking with lung cancer and heart disease. With the NIH and the Centers of Disease Control and Prevention (CDC) now in agreement about the risks of participating in impact sports and activities, he said, “We’ve reached a tipping point that should finally prompt deniers such as the NHL, NCAA, FIFA, World Rugby, the International Olympic Committee, and other [sports organizations] to remove all unnecessary head trauma from their sports.”
“A lot of the credit for this must go to Chris,” added Dr. Cantu, medical director and director of clinical research at the Cantu Concussion Center at Emerson Hospital in Concord, Mass. “Clinicians like myself can reach only so many people by writing papers and giving speeches at medical conferences. For this to happen, the message needed to get out to parents, athletes, and society in general. And Chris was the vehicle for doing that.”
Dr. Nowinski didn’t set out to be the messenger. He played football at Harvard in the late 1990s, making second-team All-Ivy as a defensive tackle his senior year. In 2000, he enrolled in Killer Kowalski’s Wrestling Institute and eventually joined Vince McMahon’s World Wrestling Entertainment (WWE).
There he played the role of 295-pound villain “Chris Harvard,” an intellectual snob who dressed in crimson tights and insulted the crowd’s IQ. “Roses are red. Violets are blue. The reason I’m talking so slowly is because no one in [insert name of town he was appearing in] has passed grade 2!”
“I’d often apply my education during a match,” he wrote in his book, “Head Games: Football’s Concussion Crisis.“ In a match in Bridgeport, Conn., I assaulted [my opponent] with a human skeleton, ripped off the skull, got down on bended knee, and began reciting Hamlet. Those were good times.”
Those good times ended abruptly, however, during a match with Bubba Ray Dudley at the Hartford Civic Center in Connecticut in 2003. Even though pro wrestling matches are rehearsed, and the blows aren’t real, accidents happen. Mr. Dudley mistakenly kicked Dr. Nowinski in the jaw with enough force to put him on his back and make the whole ring shake.
“Holy shit, kid! You okay?” asked the referee. Before a foggy Dr. Nowinski could reply, 300-pound Mr. Dudley crashed down on him, hooked his leg, and the ref began counting, “One! Two! …” Dr. Nowinski instinctively kicked out but had forgotten the rest of the script. He managed to finish the match and stagger backstage.
His coherence and awareness gradually returned, but a “throbbing headache” persisted. A locker room doctor said he might have a concussion and recommended he wait to see how he felt before wrestling in Albany, N.Y., the next evening.
The following day the headache had subsided, but he still felt “a little strange.” Nonetheless, he told the doctor he was fine and strutted out to again battle Bubba Ray, this time in a match where he eventually got thrown through a ringside table and suffered the Dudley Death Drop. Afterward, “I crawled backstage and laid down. The headache was much, much worse.”
An event and a process
Dr. Nowinski continued to insist he was “fine” and wrestled a few more matches in the following days before finally acknowledging something was wrong. He’d had his bell rung numerous times in football, but this was different. Even more worrisome, none of the doctors he consulted could give him any definitive answers. He finally found his way to Emerson Hospital, where Dr. Cantu was the chief of neurosurgery.
“I remember that day vividly,” said Dr. Cantu. “Chris was this big, strapping, handsome guy – a hell of an athlete whose star was rising. He didn’t realize that he’d suffered a series of concussions and that trying to push through them was the worst thing he could be doing.”
Concussions and their effects were misunderstood by many athletes, coaches, and even physicians back then. It was assumed that the quarter inch of bone surrounding the adult brain provided adequate protection from common sports impacts and that any aftereffects were temporary. A common treatment was smelling salts and a pat on the back as the athlete returned to action.
However, the brain floats inside the skull in a bath of cerebral fluid. Any significant impact causes it to slosh violently from side to side, damaging tissue, synapses, and cells resulting in inflammation that can manifest as confusion and brain fog.
“A concussion is actually not defined by a physical injury,” explained Dr. Nowinski, “but by a loss of brain function that is induced by trauma. Concussion is not just an event, but also a process.” It’s almost as if the person has suffered a small seizure.
Fortunately, most concussion symptoms resolve within 2 weeks, but in some cases, especially if there’s been additional head trauma, they can persist, causing anxiety, depression, anger, and/or sleep disorders. Known as postconcussion syndrome (PCS), this is what Dr. Nowinski was unknowingly suffering from when he consulted Dr. Cantu.
In fact, one night it an Indianapolis hotel, weeks after his initial concussion, he awoke to find himself on the floor and the room in shambles. His girlfriend was yelling his name and shaking him. She told him he’d been having a nightmare and had suddenly started screaming and tearing up the room. “I didn’t remember any of it,” he said.
Dr. Cantu eventually advised Dr. Nowinski against ever returning to the ring or any activity with the risk for head injury. Research shows that sustaining a single significant concussion increases the risk of subsequent more-severe brain injuries.
“My diagnosis could have sent Chris off the deep end because he could no longer do what he wanted to do with this life,” said Dr. Cantu. “But instead, he used it as a tool to find meaning for his life.”
Dr. Nowinski decided to use his experience as a teaching opportunity, not just for other athletes but also for sports organizations and the medical community.
His book, which focused on the NFL’s “tobacco-industry-like refusal to acknowledge the depths of the problem,” was published in 2006. A year later, Dr. Nowinski partnered with Dr. Cantu to found the Sports Legacy Institute, which eventually became the Concussion Legacy Foundation (CLF).
Cold calling for brain donations
Robert Stern, PhD, is another highly respected authority in the study of neurodegenerative disease. In 2007, he was directing the clinical core of Boston University’s Alzheimer’s Disease Center. After giving a lecture to a group of financial planners and elder-law attorneys one morning, he got a request for a private meeting from a fellow named Chris Nowinski.
“I’d never heard of him, but I agreed,” recalled Dr. Stern, a professor of neurology, neurosurgery, anatomy, and neurobiology at Boston University. “A few days later, this larger-than-life guy walked into our conference room at the BU School of Medicine, exuding a great deal of passion, intellect, and determination. He told me his story and then started talking about the long-term consequences of concussions in sports.”
Dr. Stern had seen patients with dementia pugilistica, the old-school term for CTE. These were mostly boxers with cognitive and behavioral impairment. “But I had not heard about football players,” he said. “I hadn’t put the two together. And as I was listening to Chris, I realized if what he was saying was true then it was not only a potentially huge public health issue, but it was also a potentially huge scientific issue in the field of neurodegenerative disease.”
Dr. Nowinski introduced Dr. Stern to Dr. Cantu, and together with Ann McKee, MD, professor of neurology and pathology at BU, they cofounded the Center for the Study of Traumatic Encephalopathy (CSTE) in 2008. It was the first center of its kind devoted to the study of CTE in the world.
One of Dr. Nowinski’s first jobs at the CSTE was soliciting and procuring brain donations. Since CTE is generally a progressive condition that can take decades to manifest, autopsy was the only way to detect it.
The brains of two former Pittsburgh Steelers, Mike Webster and Terry Long, had been examined after their untimely deaths. After immunostaining, investigators found both former NFL players had “protein misfolds” characteristic of CTE.
This finding drew a lot of public and scientific attention, given that Mr. Long died by suicide and Mr. Webster was homeless when he died of a heart attack. But more scientific evidence was needed to prove a causal link between the head trauma and CTE.
Dr. Nowinski scoured obituaries looking for potential brains to study. When he found one, he would cold call the family and try to convince them to donate it to science. The first brain he secured for the center belonged to John Grimsley, a former NFL linebacker who in 2008 died at age 45 of an accidental gunshot wound. Often, Dr. Nowinski would even be the courier, traveling to pick up the brain after it had been harvested.
Over the next 10 years, Dr. Nowinski and his research team secured 500 brain donations. The research that resulted was staggering. In the beginning only 45 cases of CTE had been identified in the world, but in the first 111 NFL players who were autopsied, 110 had the disorder.
Of the first 53 college football players autopsied, 48 had CTE. Although Dr. Nowinski’s initial focus was football, evidence of CTE was soon detected among athletes in boxing, hockey, soccer, and rugby, as well as in combat veterans. However, the National Football League and other governing sports bodies initially denied any connection between sport-related head trauma and CTE.
Cumulative damage
In 2017, after 7 years of study, Dr. Nowinski earned a PhD in neurology. As the scientific evidence continued to accumulate, two shifts occurred that Dr. Stern said represent Dr. Nowinski’s greatest contributions. First, concussion is now widely recognized as an acute brain injury with symptoms that need to be immediately diagnosed and addressed.
“This is a completely different story from where things were just 10 years ago,” said Dr. Stern, “and Chris played a central role, if not the central role, in raising awareness about that.”
All 50 states and the District of Columbia now have laws regarding sports-related concussion. And there are brain banks in Australia, Canada, New Zealand, Brazil, and the United Kingdom studying CTE. More than 2,500 athletes in a variety of sports, including NASCAR’s Dale Earnhardt Jr. and NFL hall of famer Nick Buoniconti, have publicly pledged to donate their brains to science after their deaths.
Second, said Dr. Stern, we now know that although concussions can contribute to CTE, they are not the sole cause. It’s repetitive subconcussive trauma, without symptoms of concussion, that do the most damage.
“These happen during every practice and in every game,” said Dr. Stern. In fact, it’s estimated that pro football players suffer thousands of subconcussive incidents over the course of their careers. So, a player doesn’t have to see stars or lose consciousness to suffer brain damage; small impacts can accumulate over time.
Understanding this point is crucial for making youth sports safer. “Chris has played a critical role in raising awareness here, too,” said Dr. Stern. “Allowing our kids to get hit in the head over and over can put them at greater risk for later problems, plus it just doesn’t make common sense.”
“The biggest misconception surrounding head trauma in sports,” said Dr. Nowinski, “is the belief among players, coaches, and even the medical and scientific communities that if you get hit in the head and don’t have any symptoms then you’re okay and there hasn’t been any damage. That couldn’t be further from the truth. We now know that people are suffering serious brain injuries due to the accumulated effect of subconcussive impacts, and we need to get the word out about that.”
A major initiative from the Concussion Legacy Foundation called “Stop Hitting Kids in the Head” has the goal of convincing every sport to eliminate repetitive head impacts in players under age 14 – the time when the skull and brain are still developing and most vulnerable – by 2026. In fact, Dr. Nowinski wrote that “there could be a lot of kids who are misdiagnosed and medicated for various behavioral or emotional problems that may actually be head injury–related.”
Starting in 2009, the NFL adopted a series of rule changes designed to better protect its players against repeated head trauma. Among them is a ban on spearing or leading with the helmet, penalties for hitting defenseless players, and more stringent return-to-play guidelines, including concussion protocols.
The NFL has also put more emphasis on flag football options for youngsters and, for the first time, showcased this alternative in the 2023 Pro Bowl. But Dr. Nowinski is pressuring the league to go further. “While acknowledging that the game causes CTE, the NFL still underwrites recruiting 5-year-olds to play tackle football,” he said. “In my opinion, that’s unethical, and it needs to be addressed.”
WWE one of the most responsive organizations
Dr. Nowinski said WWE has been one of the most responsive sports organizations for protecting athletes. A doctor is now ringside at every match as is an observer who knows the script, thereby allowing for instant medical intervention if something goes wrong. “Since everyone is trying to look like they have a concussion all the time, it takes a deep understanding of the business to recognize a real one,” he said.
But this hasn’t been the case with other sports. “I am eternally disappointed in the response of the professional sports industry to the knowledge of CTE and long-term concussion symptoms,” said Dr. Nowinski.
“For example, FIFA [international soccer’s governing body] still doesn’t allow doctors to evaluate [potentially concussed] players on the sidelines and put them back in the game with a free substitution [if they’re deemed okay]. Not giving players proper medical care for a brain injury is unethical,” he said. BU’s Center for the Study of Traumatic Encephalopathy diagnosed the first CTE case in soccer in 2012, and in 2015 Dr. Nowinski successfully lobbied U.S. Soccer to ban heading the ball before age 11.
“Unfortunately, many governing bodies have circled the wagons in denying their sport causes CTE,” he continued. “FIFA, World Rugby, the NHL, even the NCAA and International Olympic Committee refuse to acknowledge it and, therefore, aren’t taking any steps to prevent it. They see it as a threat to their business model. Hopefully, now that the NIH and CDC are aligned about the risks of head impact in sports, this will begin to change.”
Meanwhile, research is continuing. Scientists are getting closer to being able to diagnose CTE in living humans, with ongoing studies using PET scans, blood markers, and spinal fluid markers. In 2019, researchers identified tau proteins specific to CTE that they believe are distinct from those of Alzheimer’s and other neurodegenerative diseases. Next step would be developing a drug to slow the development of CTE once detected.
Nonetheless, athletes at all levels in impact sports still don’t fully appreciate the risks of repeated head trauma and especially subconcussive blows. “I talk to former NFL and college players every week,” said Dr. Stern. “Some tell me, ‘I love the sport, it gave me so much, and I would do it again, but I’m not letting my grandchildren play.’ But others say, ‘As long as they know the risks, they can make their own decision.’ “
Dr. Nowinski has a daughter who is 4 and a son who’s 2. Both play soccer but, thanks to dad, heading isn’t allowed in their age groups. If they continue playing sports, Dr. Nowinski said he’ll make sure they understand the risks and how to protect themselves. This is a conversation all parents should have with their kids at every level to make sure they play safe, he added.
Those in the medical community can also volunteer their time to explain head trauma to athletes, coaches, and school administrators to be sure they understand its seriousness and are doing everything to protect players.
As you watch this year’s Super Bowl, Dr. Nowinski and his team would like you to keep something in mind. Those young men on the field for your entertainment are receiving mild brain trauma repeatedly throughout the game.
Even if it’s not a huge hit that gets replayed and makes everyone gasp, even if no one gets ushered into the little sideline tent for a concussion screening, even if no one loses consciousness, brain damage is still occurring. Watch the heads of the players during every play and think about what’s going on inside their skulls regardless of how big and strong those helmets look.
A version of this article first appeared on Medscape.com.
On Oct. 5, 2022, at 10:24 a.m., Chris Nowinski, PhD, cofounder of the Boston-based Concussion Legacy Foundation (CLF), was in his home office when the email came through.
“I pounded my desk, shouted YES! and went to find my wife so I could pick her up and give her a big hug,” he recalled. “It was the culmination of 15 years of research and hard work.”
Robert Cantu, MD, who has been studying head trauma for 50+ years and has published more than 500 papers about it, compares the announcement to the 1964 Surgeon General’s report that linked cigarette smoking with lung cancer and heart disease. With the NIH and the Centers of Disease Control and Prevention (CDC) now in agreement about the risks of participating in impact sports and activities, he said, “We’ve reached a tipping point that should finally prompt deniers such as the NHL, NCAA, FIFA, World Rugby, the International Olympic Committee, and other [sports organizations] to remove all unnecessary head trauma from their sports.”
“A lot of the credit for this must go to Chris,” added Dr. Cantu, medical director and director of clinical research at the Cantu Concussion Center at Emerson Hospital in Concord, Mass. “Clinicians like myself can reach only so many people by writing papers and giving speeches at medical conferences. For this to happen, the message needed to get out to parents, athletes, and society in general. And Chris was the vehicle for doing that.”
Dr. Nowinski didn’t set out to be the messenger. He played football at Harvard in the late 1990s, making second-team All-Ivy as a defensive tackle his senior year. In 2000, he enrolled in Killer Kowalski’s Wrestling Institute and eventually joined Vince McMahon’s World Wrestling Entertainment (WWE).
There he played the role of 295-pound villain “Chris Harvard,” an intellectual snob who dressed in crimson tights and insulted the crowd’s IQ. “Roses are red. Violets are blue. The reason I’m talking so slowly is because no one in [insert name of town he was appearing in] has passed grade 2!”
“I’d often apply my education during a match,” he wrote in his book, “Head Games: Football’s Concussion Crisis.“ In a match in Bridgeport, Conn., I assaulted [my opponent] with a human skeleton, ripped off the skull, got down on bended knee, and began reciting Hamlet. Those were good times.”
Those good times ended abruptly, however, during a match with Bubba Ray Dudley at the Hartford Civic Center in Connecticut in 2003. Even though pro wrestling matches are rehearsed, and the blows aren’t real, accidents happen. Mr. Dudley mistakenly kicked Dr. Nowinski in the jaw with enough force to put him on his back and make the whole ring shake.
“Holy shit, kid! You okay?” asked the referee. Before a foggy Dr. Nowinski could reply, 300-pound Mr. Dudley crashed down on him, hooked his leg, and the ref began counting, “One! Two! …” Dr. Nowinski instinctively kicked out but had forgotten the rest of the script. He managed to finish the match and stagger backstage.
His coherence and awareness gradually returned, but a “throbbing headache” persisted. A locker room doctor said he might have a concussion and recommended he wait to see how he felt before wrestling in Albany, N.Y., the next evening.
The following day the headache had subsided, but he still felt “a little strange.” Nonetheless, he told the doctor he was fine and strutted out to again battle Bubba Ray, this time in a match where he eventually got thrown through a ringside table and suffered the Dudley Death Drop. Afterward, “I crawled backstage and laid down. The headache was much, much worse.”
An event and a process
Dr. Nowinski continued to insist he was “fine” and wrestled a few more matches in the following days before finally acknowledging something was wrong. He’d had his bell rung numerous times in football, but this was different. Even more worrisome, none of the doctors he consulted could give him any definitive answers. He finally found his way to Emerson Hospital, where Dr. Cantu was the chief of neurosurgery.
“I remember that day vividly,” said Dr. Cantu. “Chris was this big, strapping, handsome guy – a hell of an athlete whose star was rising. He didn’t realize that he’d suffered a series of concussions and that trying to push through them was the worst thing he could be doing.”
Concussions and their effects were misunderstood by many athletes, coaches, and even physicians back then. It was assumed that the quarter inch of bone surrounding the adult brain provided adequate protection from common sports impacts and that any aftereffects were temporary. A common treatment was smelling salts and a pat on the back as the athlete returned to action.
However, the brain floats inside the skull in a bath of cerebral fluid. Any significant impact causes it to slosh violently from side to side, damaging tissue, synapses, and cells resulting in inflammation that can manifest as confusion and brain fog.
“A concussion is actually not defined by a physical injury,” explained Dr. Nowinski, “but by a loss of brain function that is induced by trauma. Concussion is not just an event, but also a process.” It’s almost as if the person has suffered a small seizure.
Fortunately, most concussion symptoms resolve within 2 weeks, but in some cases, especially if there’s been additional head trauma, they can persist, causing anxiety, depression, anger, and/or sleep disorders. Known as postconcussion syndrome (PCS), this is what Dr. Nowinski was unknowingly suffering from when he consulted Dr. Cantu.
In fact, one night it an Indianapolis hotel, weeks after his initial concussion, he awoke to find himself on the floor and the room in shambles. His girlfriend was yelling his name and shaking him. She told him he’d been having a nightmare and had suddenly started screaming and tearing up the room. “I didn’t remember any of it,” he said.
Dr. Cantu eventually advised Dr. Nowinski against ever returning to the ring or any activity with the risk for head injury. Research shows that sustaining a single significant concussion increases the risk of subsequent more-severe brain injuries.
“My diagnosis could have sent Chris off the deep end because he could no longer do what he wanted to do with this life,” said Dr. Cantu. “But instead, he used it as a tool to find meaning for his life.”
Dr. Nowinski decided to use his experience as a teaching opportunity, not just for other athletes but also for sports organizations and the medical community.
His book, which focused on the NFL’s “tobacco-industry-like refusal to acknowledge the depths of the problem,” was published in 2006. A year later, Dr. Nowinski partnered with Dr. Cantu to found the Sports Legacy Institute, which eventually became the Concussion Legacy Foundation (CLF).
Cold calling for brain donations
Robert Stern, PhD, is another highly respected authority in the study of neurodegenerative disease. In 2007, he was directing the clinical core of Boston University’s Alzheimer’s Disease Center. After giving a lecture to a group of financial planners and elder-law attorneys one morning, he got a request for a private meeting from a fellow named Chris Nowinski.
“I’d never heard of him, but I agreed,” recalled Dr. Stern, a professor of neurology, neurosurgery, anatomy, and neurobiology at Boston University. “A few days later, this larger-than-life guy walked into our conference room at the BU School of Medicine, exuding a great deal of passion, intellect, and determination. He told me his story and then started talking about the long-term consequences of concussions in sports.”
Dr. Stern had seen patients with dementia pugilistica, the old-school term for CTE. These were mostly boxers with cognitive and behavioral impairment. “But I had not heard about football players,” he said. “I hadn’t put the two together. And as I was listening to Chris, I realized if what he was saying was true then it was not only a potentially huge public health issue, but it was also a potentially huge scientific issue in the field of neurodegenerative disease.”
Dr. Nowinski introduced Dr. Stern to Dr. Cantu, and together with Ann McKee, MD, professor of neurology and pathology at BU, they cofounded the Center for the Study of Traumatic Encephalopathy (CSTE) in 2008. It was the first center of its kind devoted to the study of CTE in the world.
One of Dr. Nowinski’s first jobs at the CSTE was soliciting and procuring brain donations. Since CTE is generally a progressive condition that can take decades to manifest, autopsy was the only way to detect it.
The brains of two former Pittsburgh Steelers, Mike Webster and Terry Long, had been examined after their untimely deaths. After immunostaining, investigators found both former NFL players had “protein misfolds” characteristic of CTE.
This finding drew a lot of public and scientific attention, given that Mr. Long died by suicide and Mr. Webster was homeless when he died of a heart attack. But more scientific evidence was needed to prove a causal link between the head trauma and CTE.
Dr. Nowinski scoured obituaries looking for potential brains to study. When he found one, he would cold call the family and try to convince them to donate it to science. The first brain he secured for the center belonged to John Grimsley, a former NFL linebacker who in 2008 died at age 45 of an accidental gunshot wound. Often, Dr. Nowinski would even be the courier, traveling to pick up the brain after it had been harvested.
Over the next 10 years, Dr. Nowinski and his research team secured 500 brain donations. The research that resulted was staggering. In the beginning only 45 cases of CTE had been identified in the world, but in the first 111 NFL players who were autopsied, 110 had the disorder.
Of the first 53 college football players autopsied, 48 had CTE. Although Dr. Nowinski’s initial focus was football, evidence of CTE was soon detected among athletes in boxing, hockey, soccer, and rugby, as well as in combat veterans. However, the National Football League and other governing sports bodies initially denied any connection between sport-related head trauma and CTE.
Cumulative damage
In 2017, after 7 years of study, Dr. Nowinski earned a PhD in neurology. As the scientific evidence continued to accumulate, two shifts occurred that Dr. Stern said represent Dr. Nowinski’s greatest contributions. First, concussion is now widely recognized as an acute brain injury with symptoms that need to be immediately diagnosed and addressed.
“This is a completely different story from where things were just 10 years ago,” said Dr. Stern, “and Chris played a central role, if not the central role, in raising awareness about that.”
All 50 states and the District of Columbia now have laws regarding sports-related concussion. And there are brain banks in Australia, Canada, New Zealand, Brazil, and the United Kingdom studying CTE. More than 2,500 athletes in a variety of sports, including NASCAR’s Dale Earnhardt Jr. and NFL hall of famer Nick Buoniconti, have publicly pledged to donate their brains to science after their deaths.
Second, said Dr. Stern, we now know that although concussions can contribute to CTE, they are not the sole cause. It’s repetitive subconcussive trauma, without symptoms of concussion, that do the most damage.
“These happen during every practice and in every game,” said Dr. Stern. In fact, it’s estimated that pro football players suffer thousands of subconcussive incidents over the course of their careers. So, a player doesn’t have to see stars or lose consciousness to suffer brain damage; small impacts can accumulate over time.
Understanding this point is crucial for making youth sports safer. “Chris has played a critical role in raising awareness here, too,” said Dr. Stern. “Allowing our kids to get hit in the head over and over can put them at greater risk for later problems, plus it just doesn’t make common sense.”
“The biggest misconception surrounding head trauma in sports,” said Dr. Nowinski, “is the belief among players, coaches, and even the medical and scientific communities that if you get hit in the head and don’t have any symptoms then you’re okay and there hasn’t been any damage. That couldn’t be further from the truth. We now know that people are suffering serious brain injuries due to the accumulated effect of subconcussive impacts, and we need to get the word out about that.”
A major initiative from the Concussion Legacy Foundation called “Stop Hitting Kids in the Head” has the goal of convincing every sport to eliminate repetitive head impacts in players under age 14 – the time when the skull and brain are still developing and most vulnerable – by 2026. In fact, Dr. Nowinski wrote that “there could be a lot of kids who are misdiagnosed and medicated for various behavioral or emotional problems that may actually be head injury–related.”
Starting in 2009, the NFL adopted a series of rule changes designed to better protect its players against repeated head trauma. Among them is a ban on spearing or leading with the helmet, penalties for hitting defenseless players, and more stringent return-to-play guidelines, including concussion protocols.
The NFL has also put more emphasis on flag football options for youngsters and, for the first time, showcased this alternative in the 2023 Pro Bowl. But Dr. Nowinski is pressuring the league to go further. “While acknowledging that the game causes CTE, the NFL still underwrites recruiting 5-year-olds to play tackle football,” he said. “In my opinion, that’s unethical, and it needs to be addressed.”
WWE one of the most responsive organizations
Dr. Nowinski said WWE has been one of the most responsive sports organizations for protecting athletes. A doctor is now ringside at every match as is an observer who knows the script, thereby allowing for instant medical intervention if something goes wrong. “Since everyone is trying to look like they have a concussion all the time, it takes a deep understanding of the business to recognize a real one,” he said.
But this hasn’t been the case with other sports. “I am eternally disappointed in the response of the professional sports industry to the knowledge of CTE and long-term concussion symptoms,” said Dr. Nowinski.
“For example, FIFA [international soccer’s governing body] still doesn’t allow doctors to evaluate [potentially concussed] players on the sidelines and put them back in the game with a free substitution [if they’re deemed okay]. Not giving players proper medical care for a brain injury is unethical,” he said. BU’s Center for the Study of Traumatic Encephalopathy diagnosed the first CTE case in soccer in 2012, and in 2015 Dr. Nowinski successfully lobbied U.S. Soccer to ban heading the ball before age 11.
“Unfortunately, many governing bodies have circled the wagons in denying their sport causes CTE,” he continued. “FIFA, World Rugby, the NHL, even the NCAA and International Olympic Committee refuse to acknowledge it and, therefore, aren’t taking any steps to prevent it. They see it as a threat to their business model. Hopefully, now that the NIH and CDC are aligned about the risks of head impact in sports, this will begin to change.”
Meanwhile, research is continuing. Scientists are getting closer to being able to diagnose CTE in living humans, with ongoing studies using PET scans, blood markers, and spinal fluid markers. In 2019, researchers identified tau proteins specific to CTE that they believe are distinct from those of Alzheimer’s and other neurodegenerative diseases. Next step would be developing a drug to slow the development of CTE once detected.
Nonetheless, athletes at all levels in impact sports still don’t fully appreciate the risks of repeated head trauma and especially subconcussive blows. “I talk to former NFL and college players every week,” said Dr. Stern. “Some tell me, ‘I love the sport, it gave me so much, and I would do it again, but I’m not letting my grandchildren play.’ But others say, ‘As long as they know the risks, they can make their own decision.’ “
Dr. Nowinski has a daughter who is 4 and a son who’s 2. Both play soccer but, thanks to dad, heading isn’t allowed in their age groups. If they continue playing sports, Dr. Nowinski said he’ll make sure they understand the risks and how to protect themselves. This is a conversation all parents should have with their kids at every level to make sure they play safe, he added.
Those in the medical community can also volunteer their time to explain head trauma to athletes, coaches, and school administrators to be sure they understand its seriousness and are doing everything to protect players.
As you watch this year’s Super Bowl, Dr. Nowinski and his team would like you to keep something in mind. Those young men on the field for your entertainment are receiving mild brain trauma repeatedly throughout the game.
Even if it’s not a huge hit that gets replayed and makes everyone gasp, even if no one gets ushered into the little sideline tent for a concussion screening, even if no one loses consciousness, brain damage is still occurring. Watch the heads of the players during every play and think about what’s going on inside their skulls regardless of how big and strong those helmets look.
A version of this article first appeared on Medscape.com.
On Oct. 5, 2022, at 10:24 a.m., Chris Nowinski, PhD, cofounder of the Boston-based Concussion Legacy Foundation (CLF), was in his home office when the email came through.
“I pounded my desk, shouted YES! and went to find my wife so I could pick her up and give her a big hug,” he recalled. “It was the culmination of 15 years of research and hard work.”
Robert Cantu, MD, who has been studying head trauma for 50+ years and has published more than 500 papers about it, compares the announcement to the 1964 Surgeon General’s report that linked cigarette smoking with lung cancer and heart disease. With the NIH and the Centers of Disease Control and Prevention (CDC) now in agreement about the risks of participating in impact sports and activities, he said, “We’ve reached a tipping point that should finally prompt deniers such as the NHL, NCAA, FIFA, World Rugby, the International Olympic Committee, and other [sports organizations] to remove all unnecessary head trauma from their sports.”
“A lot of the credit for this must go to Chris,” added Dr. Cantu, medical director and director of clinical research at the Cantu Concussion Center at Emerson Hospital in Concord, Mass. “Clinicians like myself can reach only so many people by writing papers and giving speeches at medical conferences. For this to happen, the message needed to get out to parents, athletes, and society in general. And Chris was the vehicle for doing that.”
Dr. Nowinski didn’t set out to be the messenger. He played football at Harvard in the late 1990s, making second-team All-Ivy as a defensive tackle his senior year. In 2000, he enrolled in Killer Kowalski’s Wrestling Institute and eventually joined Vince McMahon’s World Wrestling Entertainment (WWE).
There he played the role of 295-pound villain “Chris Harvard,” an intellectual snob who dressed in crimson tights and insulted the crowd’s IQ. “Roses are red. Violets are blue. The reason I’m talking so slowly is because no one in [insert name of town he was appearing in] has passed grade 2!”
“I’d often apply my education during a match,” he wrote in his book, “Head Games: Football’s Concussion Crisis.“ In a match in Bridgeport, Conn., I assaulted [my opponent] with a human skeleton, ripped off the skull, got down on bended knee, and began reciting Hamlet. Those were good times.”
Those good times ended abruptly, however, during a match with Bubba Ray Dudley at the Hartford Civic Center in Connecticut in 2003. Even though pro wrestling matches are rehearsed, and the blows aren’t real, accidents happen. Mr. Dudley mistakenly kicked Dr. Nowinski in the jaw with enough force to put him on his back and make the whole ring shake.
“Holy shit, kid! You okay?” asked the referee. Before a foggy Dr. Nowinski could reply, 300-pound Mr. Dudley crashed down on him, hooked his leg, and the ref began counting, “One! Two! …” Dr. Nowinski instinctively kicked out but had forgotten the rest of the script. He managed to finish the match and stagger backstage.
His coherence and awareness gradually returned, but a “throbbing headache” persisted. A locker room doctor said he might have a concussion and recommended he wait to see how he felt before wrestling in Albany, N.Y., the next evening.
The following day the headache had subsided, but he still felt “a little strange.” Nonetheless, he told the doctor he was fine and strutted out to again battle Bubba Ray, this time in a match where he eventually got thrown through a ringside table and suffered the Dudley Death Drop. Afterward, “I crawled backstage and laid down. The headache was much, much worse.”
An event and a process
Dr. Nowinski continued to insist he was “fine” and wrestled a few more matches in the following days before finally acknowledging something was wrong. He’d had his bell rung numerous times in football, but this was different. Even more worrisome, none of the doctors he consulted could give him any definitive answers. He finally found his way to Emerson Hospital, where Dr. Cantu was the chief of neurosurgery.
“I remember that day vividly,” said Dr. Cantu. “Chris was this big, strapping, handsome guy – a hell of an athlete whose star was rising. He didn’t realize that he’d suffered a series of concussions and that trying to push through them was the worst thing he could be doing.”
Concussions and their effects were misunderstood by many athletes, coaches, and even physicians back then. It was assumed that the quarter inch of bone surrounding the adult brain provided adequate protection from common sports impacts and that any aftereffects were temporary. A common treatment was smelling salts and a pat on the back as the athlete returned to action.
However, the brain floats inside the skull in a bath of cerebral fluid. Any significant impact causes it to slosh violently from side to side, damaging tissue, synapses, and cells resulting in inflammation that can manifest as confusion and brain fog.
“A concussion is actually not defined by a physical injury,” explained Dr. Nowinski, “but by a loss of brain function that is induced by trauma. Concussion is not just an event, but also a process.” It’s almost as if the person has suffered a small seizure.
Fortunately, most concussion symptoms resolve within 2 weeks, but in some cases, especially if there’s been additional head trauma, they can persist, causing anxiety, depression, anger, and/or sleep disorders. Known as postconcussion syndrome (PCS), this is what Dr. Nowinski was unknowingly suffering from when he consulted Dr. Cantu.
In fact, one night it an Indianapolis hotel, weeks after his initial concussion, he awoke to find himself on the floor and the room in shambles. His girlfriend was yelling his name and shaking him. She told him he’d been having a nightmare and had suddenly started screaming and tearing up the room. “I didn’t remember any of it,” he said.
Dr. Cantu eventually advised Dr. Nowinski against ever returning to the ring or any activity with the risk for head injury. Research shows that sustaining a single significant concussion increases the risk of subsequent more-severe brain injuries.
“My diagnosis could have sent Chris off the deep end because he could no longer do what he wanted to do with this life,” said Dr. Cantu. “But instead, he used it as a tool to find meaning for his life.”
Dr. Nowinski decided to use his experience as a teaching opportunity, not just for other athletes but also for sports organizations and the medical community.
His book, which focused on the NFL’s “tobacco-industry-like refusal to acknowledge the depths of the problem,” was published in 2006. A year later, Dr. Nowinski partnered with Dr. Cantu to found the Sports Legacy Institute, which eventually became the Concussion Legacy Foundation (CLF).
Cold calling for brain donations
Robert Stern, PhD, is another highly respected authority in the study of neurodegenerative disease. In 2007, he was directing the clinical core of Boston University’s Alzheimer’s Disease Center. After giving a lecture to a group of financial planners and elder-law attorneys one morning, he got a request for a private meeting from a fellow named Chris Nowinski.
“I’d never heard of him, but I agreed,” recalled Dr. Stern, a professor of neurology, neurosurgery, anatomy, and neurobiology at Boston University. “A few days later, this larger-than-life guy walked into our conference room at the BU School of Medicine, exuding a great deal of passion, intellect, and determination. He told me his story and then started talking about the long-term consequences of concussions in sports.”
Dr. Stern had seen patients with dementia pugilistica, the old-school term for CTE. These were mostly boxers with cognitive and behavioral impairment. “But I had not heard about football players,” he said. “I hadn’t put the two together. And as I was listening to Chris, I realized if what he was saying was true then it was not only a potentially huge public health issue, but it was also a potentially huge scientific issue in the field of neurodegenerative disease.”
Dr. Nowinski introduced Dr. Stern to Dr. Cantu, and together with Ann McKee, MD, professor of neurology and pathology at BU, they cofounded the Center for the Study of Traumatic Encephalopathy (CSTE) in 2008. It was the first center of its kind devoted to the study of CTE in the world.
One of Dr. Nowinski’s first jobs at the CSTE was soliciting and procuring brain donations. Since CTE is generally a progressive condition that can take decades to manifest, autopsy was the only way to detect it.
The brains of two former Pittsburgh Steelers, Mike Webster and Terry Long, had been examined after their untimely deaths. After immunostaining, investigators found both former NFL players had “protein misfolds” characteristic of CTE.
This finding drew a lot of public and scientific attention, given that Mr. Long died by suicide and Mr. Webster was homeless when he died of a heart attack. But more scientific evidence was needed to prove a causal link between the head trauma and CTE.
Dr. Nowinski scoured obituaries looking for potential brains to study. When he found one, he would cold call the family and try to convince them to donate it to science. The first brain he secured for the center belonged to John Grimsley, a former NFL linebacker who in 2008 died at age 45 of an accidental gunshot wound. Often, Dr. Nowinski would even be the courier, traveling to pick up the brain after it had been harvested.
Over the next 10 years, Dr. Nowinski and his research team secured 500 brain donations. The research that resulted was staggering. In the beginning only 45 cases of CTE had been identified in the world, but in the first 111 NFL players who were autopsied, 110 had the disorder.
Of the first 53 college football players autopsied, 48 had CTE. Although Dr. Nowinski’s initial focus was football, evidence of CTE was soon detected among athletes in boxing, hockey, soccer, and rugby, as well as in combat veterans. However, the National Football League and other governing sports bodies initially denied any connection between sport-related head trauma and CTE.
Cumulative damage
In 2017, after 7 years of study, Dr. Nowinski earned a PhD in neurology. As the scientific evidence continued to accumulate, two shifts occurred that Dr. Stern said represent Dr. Nowinski’s greatest contributions. First, concussion is now widely recognized as an acute brain injury with symptoms that need to be immediately diagnosed and addressed.
“This is a completely different story from where things were just 10 years ago,” said Dr. Stern, “and Chris played a central role, if not the central role, in raising awareness about that.”
All 50 states and the District of Columbia now have laws regarding sports-related concussion. And there are brain banks in Australia, Canada, New Zealand, Brazil, and the United Kingdom studying CTE. More than 2,500 athletes in a variety of sports, including NASCAR’s Dale Earnhardt Jr. and NFL hall of famer Nick Buoniconti, have publicly pledged to donate their brains to science after their deaths.
Second, said Dr. Stern, we now know that although concussions can contribute to CTE, they are not the sole cause. It’s repetitive subconcussive trauma, without symptoms of concussion, that do the most damage.
“These happen during every practice and in every game,” said Dr. Stern. In fact, it’s estimated that pro football players suffer thousands of subconcussive incidents over the course of their careers. So, a player doesn’t have to see stars or lose consciousness to suffer brain damage; small impacts can accumulate over time.
Understanding this point is crucial for making youth sports safer. “Chris has played a critical role in raising awareness here, too,” said Dr. Stern. “Allowing our kids to get hit in the head over and over can put them at greater risk for later problems, plus it just doesn’t make common sense.”
“The biggest misconception surrounding head trauma in sports,” said Dr. Nowinski, “is the belief among players, coaches, and even the medical and scientific communities that if you get hit in the head and don’t have any symptoms then you’re okay and there hasn’t been any damage. That couldn’t be further from the truth. We now know that people are suffering serious brain injuries due to the accumulated effect of subconcussive impacts, and we need to get the word out about that.”
A major initiative from the Concussion Legacy Foundation called “Stop Hitting Kids in the Head” has the goal of convincing every sport to eliminate repetitive head impacts in players under age 14 – the time when the skull and brain are still developing and most vulnerable – by 2026. In fact, Dr. Nowinski wrote that “there could be a lot of kids who are misdiagnosed and medicated for various behavioral or emotional problems that may actually be head injury–related.”
Starting in 2009, the NFL adopted a series of rule changes designed to better protect its players against repeated head trauma. Among them is a ban on spearing or leading with the helmet, penalties for hitting defenseless players, and more stringent return-to-play guidelines, including concussion protocols.
The NFL has also put more emphasis on flag football options for youngsters and, for the first time, showcased this alternative in the 2023 Pro Bowl. But Dr. Nowinski is pressuring the league to go further. “While acknowledging that the game causes CTE, the NFL still underwrites recruiting 5-year-olds to play tackle football,” he said. “In my opinion, that’s unethical, and it needs to be addressed.”
WWE one of the most responsive organizations
Dr. Nowinski said WWE has been one of the most responsive sports organizations for protecting athletes. A doctor is now ringside at every match as is an observer who knows the script, thereby allowing for instant medical intervention if something goes wrong. “Since everyone is trying to look like they have a concussion all the time, it takes a deep understanding of the business to recognize a real one,” he said.
But this hasn’t been the case with other sports. “I am eternally disappointed in the response of the professional sports industry to the knowledge of CTE and long-term concussion symptoms,” said Dr. Nowinski.
“For example, FIFA [international soccer’s governing body] still doesn’t allow doctors to evaluate [potentially concussed] players on the sidelines and put them back in the game with a free substitution [if they’re deemed okay]. Not giving players proper medical care for a brain injury is unethical,” he said. BU’s Center for the Study of Traumatic Encephalopathy diagnosed the first CTE case in soccer in 2012, and in 2015 Dr. Nowinski successfully lobbied U.S. Soccer to ban heading the ball before age 11.
“Unfortunately, many governing bodies have circled the wagons in denying their sport causes CTE,” he continued. “FIFA, World Rugby, the NHL, even the NCAA and International Olympic Committee refuse to acknowledge it and, therefore, aren’t taking any steps to prevent it. They see it as a threat to their business model. Hopefully, now that the NIH and CDC are aligned about the risks of head impact in sports, this will begin to change.”
Meanwhile, research is continuing. Scientists are getting closer to being able to diagnose CTE in living humans, with ongoing studies using PET scans, blood markers, and spinal fluid markers. In 2019, researchers identified tau proteins specific to CTE that they believe are distinct from those of Alzheimer’s and other neurodegenerative diseases. Next step would be developing a drug to slow the development of CTE once detected.
Nonetheless, athletes at all levels in impact sports still don’t fully appreciate the risks of repeated head trauma and especially subconcussive blows. “I talk to former NFL and college players every week,” said Dr. Stern. “Some tell me, ‘I love the sport, it gave me so much, and I would do it again, but I’m not letting my grandchildren play.’ But others say, ‘As long as they know the risks, they can make their own decision.’ “
Dr. Nowinski has a daughter who is 4 and a son who’s 2. Both play soccer but, thanks to dad, heading isn’t allowed in their age groups. If they continue playing sports, Dr. Nowinski said he’ll make sure they understand the risks and how to protect themselves. This is a conversation all parents should have with their kids at every level to make sure they play safe, he added.
Those in the medical community can also volunteer their time to explain head trauma to athletes, coaches, and school administrators to be sure they understand its seriousness and are doing everything to protect players.
As you watch this year’s Super Bowl, Dr. Nowinski and his team would like you to keep something in mind. Those young men on the field for your entertainment are receiving mild brain trauma repeatedly throughout the game.
Even if it’s not a huge hit that gets replayed and makes everyone gasp, even if no one gets ushered into the little sideline tent for a concussion screening, even if no one loses consciousness, brain damage is still occurring. Watch the heads of the players during every play and think about what’s going on inside their skulls regardless of how big and strong those helmets look.
A version of this article first appeared on Medscape.com.